WO2025026908A1 - Serum epha1 as biomarker for endometriosis - Google Patents
Serum epha1 as biomarker for endometriosis Download PDFInfo
- Publication number
- WO2025026908A1 WO2025026908A1 PCT/EP2024/071268 EP2024071268W WO2025026908A1 WO 2025026908 A1 WO2025026908 A1 WO 2025026908A1 EP 2024071268 W EP2024071268 W EP 2024071268W WO 2025026908 A1 WO2025026908 A1 WO 2025026908A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- endometriosis
- epha1
- subject
- uterine
- level
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- G01N33/5755—
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/68—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids
- G01N33/689—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids related to pregnancy or the gonads
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2333/00—Assays involving biological materials from specific organisms or of a specific nature
- G01N2333/90—Enzymes; Proenzymes
- G01N2333/91—Transferases (2.)
- G01N2333/912—Transferases (2.) transferring phosphorus containing groups, e.g. kinases (2.7)
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2800/00—Detection or diagnosis of diseases
- G01N2800/36—Gynecology or obstetrics
- G01N2800/364—Endometriosis, i.e. non-malignant disorder in which functioning endometrial tissue is present outside the uterine cavity
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2800/00—Detection or diagnosis of diseases
- G01N2800/52—Predicting or monitoring the response to treatment, e.g. for selection of therapy based on assay results in personalised medicine; Prognosis
-
- G—PHYSICS
- G16—INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR SPECIFIC APPLICATION FIELDS
- G16H—HEALTHCARE INFORMATICS, i.e. INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR THE HANDLING OR PROCESSING OF MEDICAL OR HEALTHCARE DATA
- G16H50/00—ICT specially adapted for medical diagnosis, medical simulation or medical data mining; ICT specially adapted for detecting, monitoring or modelling epidemics or pandemics
- G16H50/20—ICT specially adapted for medical diagnosis, medical simulation or medical data mining; ICT specially adapted for detecting, monitoring or modelling epidemics or pandemics for computer-aided diagnosis, e.g. based on medical expert systems
Definitions
- Serum EphA1 as biomarker for endometriosis
- the present invention relates to methods of diagnosing whether a subject has endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain, methods of assessing the risk of a subject having endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain, to methods of determining the therapeutic effect of a treatment regimen for endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain, and methods of monitoring endometriosis progression, uterine/pelvic pathology progression and/or endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain progression in a subject, by determining the amount or concentration of EphA1 in a sample of the subject, and comparing the determined level to
- Endometriosis is a chronic disorder defined by the growth of endometrial glands and stromalike lesions outside the uterus (Liu et al., 2011).
- the lesions can be peritoneal lesions, superficial implants or cysts on the ovary, or deep infiltrating disease. It arises from eutopic endometrial cells characterized by increased proliferation and adhesion properties (Liu et al., 2011).
- the increased cell viability in eutopic endometrium is a consequence of reduced apoptosis and an increase in cell proliferation (Johnson et al., 2005).
- Endometriosis affects 5- 8% of all women of reproductive age and 70% of women with chronic pelvic pain.
- endometriosis has been estimated at 176 million women worldwide (Adamson et al. J Endometr. 2010; 2: 3-6). For many of these women there is often a delay in diagnosis of endometriosis resulting in unnecessary suffering and reduced quality of life. In patients aged 18-45 years, there is a diagnostic delay of endometriosis of 7-10 years. As most women with endometriosis report the onset of symptoms during adolescence, early referral, diagnosis, identification of disease and treatment may mitigate pain while also potentially preventing disease progression.
- Barriers to early diagnosis include the interventional nature of current diagnostic techniques such as laparoscopy, the high cost of diagnosis and treatment in adolescent patients and presentation of confounding symptoms such as cyclic and non-cyclic pain (Parasar et al. Curr Obstet Gynecol Rep. 2017; 6: 34-41).
- Non-invasive diagnosis of endometriosis would allow earlier diagnosis and treatment, with the potential to improve quality of life and reduce the societal costs related to endometriosis, and has therefore been selected as a research priority by the World Endometriosis Society (WES) and the World Endometriosis Research Foundation (WERF) (Fassbender et al., Springer, Peripheral Blood Biomarkers for Endometriosis. 2017).
- WERF World Endometriosis Research Foundation
- a non-invasive tool to diagnose endometriosis could facilitate earlier diagnosis and intervention that could ultimately improve quality of life and preserve fertility (Parasar et al. Curr Obstet Gynecol Rep. 2017; 6: 34-41).
- CA-125 Blood based biomarkers are essential to help reduce the time delay of diagnosing endometriosis that require laparoscopy.
- CA-125 is one of the most commonly used blood biomarkers, however, its diagnostic utility is limited to endometriosis rASRM stages III and IV (Nisenblat et al., Cochrane Database of Systematic Reviews. 2016;5: CD012179). As such the use of CA-125 is not recommended in clinical guidelines for the diagnosis of endometriosis (ESHRE Guideline Endometriosis Human Reproduction Open, 2022). Same applies to similar disorders like uterine and pelvic pathology as well as endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
- Endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain is a component of endometriosis-associated pain (Coxon et al., /s there a Neuropathic-Like Component to Endometriosis-Associated Pain? Resuits From a Large Cohort Questionnaire Study, Front Pain Res (Lausanne), 2021 ; 2: 743812; doi: 10.3389/fpain.2021.743812).
- Ephrin (Eph) receptor family comprises the largest family in receptor tyrosine kinases and can be divided into two groups based on their structures and receptor-ligand specificity.
- EphA consists of nine type-A Eph receptors (EphA1-8, and EphAIO) and five type-B Eph receptors (EphB1-4, 6).
- Eph receptors bind to the ephrin ligands which are also divided into ephrin- A (ephrin-A1-6) and ephrin-B (ephrin- B1-3) based on their structures.
- ephrins Binding of ephrins to the Eph receptors activates signaling cascades that regulate several biological processes such as cellular proliferation, differentiation, migration, angiogenesis, and vascular remodeling. There is evidence implicating ephrins and the Eph receptors in the modulation of folliculogenesis, ovulation, embryo transport, implantation, and placentation (Adu-Gyamfi et al. Biology of Reproduction, 2021).
- Ephrin type-A receptor 1 (EphA1) is a receptor tyrosine kinase which binds to the ephrin-A family ligands residing on adjacent cells. Both EphA1 and Ephrin-A are membrane bound with their binding occurring by direct cell-cell interaction leading to contact-dependent bidirectional signaling into neighboring cells. Forward signaling occurs in Eph receptorexpressing cells whereas reverse signaling occurs in ephrin-expressing cells.
- EphA receptors are involved in regulating tumor growth, invasiveness, angiogenesis, and metastasis by altering cell proliferation, motility, invasion, and migration (Zhang X. Front Oncol. 2021 ;11 :619949).
- Fujii et al. (Hum Reprod 2011 ;26:299-306) has shown by RT-PCR that mRNA of EPHA1 was expressed in endometrial epithelial cell fractions in the proliferative and secretory phases of the menstrual cycle. EphA1 protein was expressed on the endometrial luminal surface and glandular epithelial cells demonstrated by immunohistochemistry.
- the levels/presence of a biomarker can differ when measured in the tissue or in blood serum.
- the complement component C7 and complement component C4 showed overexpression in ectopic endometrium of women with endometriosis compared to eutopic endometrium of control women without endometriosis (Ahn et al. Fertil Steril 2016; Eyster et al. Fertil Steril 2007).
- no increase of serum complement component C7 protein nor complement component C4 protein was found in circulating blood (Hever et al. PNAS 2007).
- Brain-derived neurotrophic factor (BDNF) mRNA expression levels were higher in ovarian endometriotic lesions than in eutopic endometrium (Wang et al.
- biomarkers in tissue does not translate 1 :1 to significantly different levels of these biomarkers in circulating blood.
- the present invention therefore, provides means and methods complying with these needs.
- the present invention relates to a method for assessing endometriosis in a subject, the method comprising the steps of: a) determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject, b) comparing the level of the extracellular protein part of EphA1 to at least one appropriate reference value of a level of the extracellular protein part of EphA1, c) identifying a subject as having endometriosis if the comparison in step b) indicates that the subject has a decreased level of the extracellular protein part of EphA1 compared to the appropriate reference value, wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
- r-ASRM revised scoring system of the American Society for Reproductive Medicine
- the present invention relates to a method for assessing uterine/pelvic pathology in a subject, the method comprising the steps of: a) determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject, b) comparing the level of the extracellular protein part of EphA1 to at least one appropriate reference value of a level of the extracellular protein part of EphA1, c) identifying a subject as having uterine/pelvic pathology if the comparison in step b) indicates that the subject has a decreased level of the extracellular protein part of EphA1 compared to the appropriate reference value,
- the invention in a third aspect relates to a method for assessing endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject, the method comprising the steps of: a) determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject, b) comparing the level of the extracellular protein part of EphA1 to at least one appropriate reference value of a level of the extracellular protein part of EphA1, c) identifying a subject as having endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain if the comparison in step b) indicates that the subject has a decreased level of the extracellular protein part of EphA1 compared to the appropriate reference value, wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
- r-ASRM revised scoring system of the
- the present invention relates to a method for monitoring endometriosis progression, uterine/pelvic pathology progression and/or progression of endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject, the method comprising the steps of: i. determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject in accordance with method steps a) to b) of the first aspect, ii. repeating step i. using a biological fluid sample obtained from the subject during or after treatment for a time interval; and iii. comparing the levels of the extracellular protein part of EphA1 identified in i.
- a change in the levels of extracellular protein part of EphA1 from i. to ii. is indicative of a change in endometriosis progression , uterine/pelvic pathology progression and/or progression of endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in the subject, wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
- the present invention relates to a method for determining the therapeutic effect of a treatment regimen for endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject, the method comprising the steps of: i. determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject in accordance with method steps a) to b) of the first aspect, ii. repeating step i. using a biological fluid sample obtained from the subject during or after treatment for a time interval; and iii. comparing levels of the extracellular protein part of EphA1 identified in i.
- stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
- the present invention relates to a computer-implemented method for assessing a patient with suspected endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain comprising the steps of: a) receiving a value for level of a first biomarker in a biological fluid sample of the subject, said first biomarker being the extracellular protein part of EphA1 ; b) receiving a value for the level of a second biomarker in a sample of the subject, wherein said second biomarker is CA125, c) receiving a value for the level of dysmenorrhea according to the VAS and/or lower abdominal pain according to the VAS, d) comparing the values for the levels of steps (a) - (c) to references for said biomarkers and the amount of dysmenorrhea and/or calculating a score for assessing the subject with suspected endometriosis, uterine/pelvic
- EphA1 EphA1 in serum samples of control groups versus endometriosis case groups (rASRM stage I, endometriosis rASRM stage II, endometriosis rASRM stage III and endometriosis rASRM stage IV). Serum EphA1 peptide levels were measured using advanced mass spectrometry based proteomics technology.
- Receiver operator characteristic (ROC) analysis is shown describing the area under the curve (AUC) of the ROC analysis and the associated 95% confidence interval.
- AUC area under the curve
- N depicts the number of samples tested (cases plus controls, numbers in each group vary depending on the analyte).
- FIGS 3a and b Boxplot analysis (a) and ROC analysis (b): EphA1 is decreased in serum samples of women with early endometriosis rASRM stages l/ll “Case” compared to control women without endometriosis and without uterine and pelvic pathology (“Ctrl”).
- FIG. 4a and b Boxplot analysis (a) and ROC analysis (b): EphA1 is decreased in serum samples of women with early endometriosis rASRM stage I “Case” compared to women without endometriosis and without uterine and pelvic pathology (“Ctrl”).
- FIG. 5a and b Boxplot analysis (a) and ROC analysis (b): EphA1 is decreased in serum samples of women with early endometriosis rASRM stage II “Case” compared to women without endometriosis and without uterine and pelvic pathology (“Ctrl”).
- FIG. 6a and b Boxplot analysis (a) and ROC analysis (b): EphA1 is decreased in serum samples of women with endometriosis stage III “Case” compared to control women without endometriosis and without uterine and pelvic pathology (“Ctrl”).
- FIG 7a and b Boxplot analysis (a) and ROC analysis (b): EphA1 is decreased in serum samples of women with endometriosis rASRM stage IV “Case” compared to controls without endometriosis and without uterine and pelvic pathology (“Ctrl”).
- Figure 8a and b Boxplot analysis (a) and ROC analysis (b) of CA-125. Comparison to CA-125 levels measured in the same sample set as EphA1 , using the Roche Elecsys CA-125 immunoassay method. CA-125 in serum samples of women with early endometriosis rASRM stage I “Case” compared to controls without endometriosis and without uterine and pelvic pathology “Ctrl”.
- Figure 9a and b Boxplot analysis (a) and ROC analysis (b) of CA-125 in serum samples of women with early endometriosis rASRM stage II “Case” compared to controls without endometriosis and without uterine and pelvic pathology “Ctrl”.
- the Transmembrane domain links the Extracellular domain and the Intracellular domain.
- the Intracellular domain is composed of the Juxtamembrane domain, the Kinase domain, and the SAM domain.
- An unbiased proteomics discovery was performed in serum samples of women with endometriosis or other uterine pelvic pathology and symptoms (pain symptoms such as dysmenorrhea, pelvic pain, dyspareunia, dysuria, dyschezia, other menstrual-cycle dependent pain symptoms) and women without endometriosis (controls). Details for the unbiased proteomics discovery using advanced mass spectrometry (MS)-based proteomics including automated sample preparation procedures are described in Example 4 - Materials and Methods.
- MS mass spectrometry
- the invention is based on the surprising finding that decreased EphA1 levels in serum, in particular decreased levels of the extracellular protein part of EphA1 , is associated with endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain.
- serum from women with endometriosis (cases) and controls (without endometriosis) peptides were identified of the EphA1 extracellular protein part detected using advanced mass spectrometry proteomics technology.
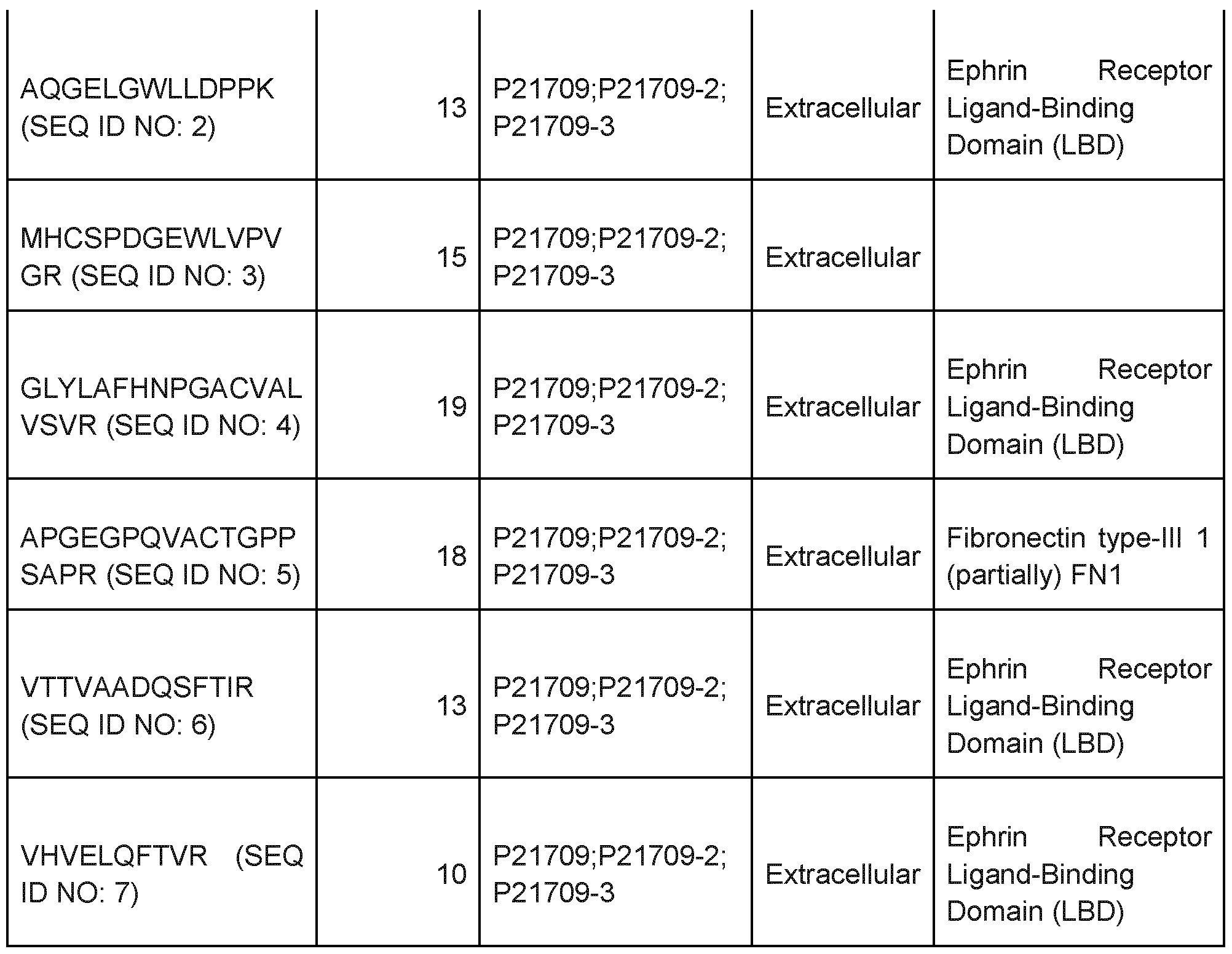
- seven peptides of EphA1 were detected using this mass spectrometry technology which all belong to the extracellular protein part of the EphA1 (Table 1).
- the structure of the EphA1 protein is shown in Figure 10. EphA1 levels are decreased at all rASRM stages I, II, III and IV, which confers serum EphA1 a diagnostic potential for early detection of endometriosis.
- EphA1 measured in serum is decreased in women with endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain compared to controls.
- EphA1 has the advantage of a non-invasive blood-based test that identifies women with early stages of endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
- the inventors have investigated the levels of EphA1 in serum obtained from women with endometriosis. Surprisingly, they found that a decreased level of EphA1 can be detected in serum samples from women with endometriosis. In particular, the fact that already in women with early stages of endometriosis decreased EphA1 levels can be detected make this marker a helpful tool to diagnose endometriosis at early stages. Assays that enable the determination of the level of EphA1 in such biological fluids may therefore be useful for endometriosis risk stratification, diagnosis, prognosis and patient stratification for treatment.
- EphA1 levels in particular levels of the extracellular protein part of EphA1 , in serum provides a means for diagnosing endometriosis and risk stratification of having endometriosis. This also allows the monitoring of endometriosis progression and/or the evaluation of treatment regimen. In particular, the data show that a diagnosis of early stages of endometriosis is possible by determining EphA1 levels.
- EphA1 levels in serum provides a means for detecting early stages of endometriosis and control samples more accurately than CA-125.
- the inventors have investigated the levels of EphA1 in serum obtained from women with uterine/pelvic pathology. Surprisingly, they found that a decreased level of EphA1 can be detected in serum samples from women with uterine/pelvic pathology. This makes this marker a helpful tool to diagnose uterine/pelvic pathology at early stages. Assays that enable the determination of the level of EphA1 in such biological fluids may therefore be useful for end uterine/pelvic pathology risk stratification, diagnosis, prognosis and patient stratification for treatment.
- EphA1 levels in particular levels of the extracellular protein part of EphA1 , in serum provides a means for diagnosing uterine/pelvic pathology and risk stratification of having uterine/pelvic pathology. This also allows the monitoring of uterine/pelvic pathology progression and/or the evaluation of treatment regimen.
- Concentrations, levels, amounts, and other numerical data may be expressed or presented herein in a “range” format. It is to be understood that such a range format is used merely for convenience and brevity and thus should be interpreted flexibly to include not only the numerical values explicitly recited as the limits of the range, but also to include all the individual numerical values or sub-ranges encompassed within that range as if each numerical value and sub-range is explicitly recited. As an illustration, a numerical range of "150 mg to 600 mg” should be interpreted to include not only the explicitly recited values of 150 mg to 600 mg, but to also include individual values and sub-ranges within the indicated range. Thus, included in this numerical range are individual values such as 150, 160, 170, 180, 190, ...
- the methods described are in vitro methods that are performed using a sample that has already been obtained from the subject (i.e. , the sample is provided for the method, and the steps taken to obtain the sample from the subject are not included as part of the method).
- the methods may therefore include the step of providing a biological fluid sample from a subject.
- Directly obtaining a sample means performing a process (e.g., performing a physical method such as extraction) to obtain the sample.
- Indirectly obtaining a sample refers to receiving the sample from another party or source (e.g., a third party laboratory that directly acquired the sample).
- test samples comprise providing a biological fluid sample (for example a blood sample) from a subject.
- a biological fluid sample for example a blood sample
- test samples are also referred to as “test samples”.
- biological (fluid) sample As used herein, the terms "biological (fluid) sample”, “test sample”, “sample” are used interchangeably, and variations thereof refer to a sample obtained or derived from a subject.
- the sample is, or comprises, a biological fluid (also referred to herein as a bodily fluid) sample.
- samples include but are not limited to fluid samples such as blood, serum, plasma, synovial fluid, interstitial fluid, capillary blood, peritoneal fluid, menstrual fluid, urine, saliva, and lymphatic fluid. Analysis of a sample may be accomplished on chemical basis. Chemical analysis includes but is not limited to the detection of the presence or absence of specific indicators or alterations in their amount, concentration or level.
- the sample is an in vitro sample, it will be analyzed in vitro and not transferred back into the body.
- a blood sample may be a whole blood sample, or a processed blood sample e.g., serum, plasma etc.
- Methods for obtaining biological fluid samples (e.g., whole blood, serum, plasma, etc) from a subject are well known in the art. For example, methods for obtaining blood samples from a subject are well known and include established techniques used in phlebotomy.
- the obtained blood samples may be further processed using standard techniques to obtain e.g., a serum sample, or a plasma sample.
- methods for obtaining biological fluid samples from a subject are typically low-invasive or non-invasive.
- a whole blood sample is defined as a blood sample drawn from the body and from which (substantially) no constituents (such as platelets or plasma) have been removed.
- the relative ratio of constituents in a whole blood sample is substantially the same as a blood in the body.
- “substantially the same” allows for a very small change in the relative ratio of the constituents of whole blood e.g., a change of up to 5%, up to 4%, up to 3%, up to 2%, up to 1 % etc.
- Whole blood contains both the cell and fluid portions of blood.
- a whole blood sample may therefore also be defined as a blood sample with (substantially) all of its cellular components in plasma, wherein the cellular components (i.e. , at least comprising the requisite white blood cells, red blood cells, platelets of blood) are intact.
- the biological fluid sample is serum.
- biomarker refers to a substance within a biological system that is used as an indicator of a biological state of said system.
- the term termed “biomarker” is sometimes also applied to means for the detection of said endogenous substances (e.g. antibodies, nucleic acid probes etc, imaging systems).
- biomarker shall be only applied for the substance, not for the detection means.
- biomarkers can be any kind of molecule present in a living organism, such as a nucleic acid (DNA, mRNA, miRNA, rRNA etc.), a protein (cell surface receptor, cytosolic protein etc.), a metabolite or hormone (blood sugar, insulin, estrogen, etc.), a molecule characteristic of a certain modification of another molecule (e.g. sugar moieties or phosphoryl residues on proteins, methyl-residues on genomic DNA) or a substance that has been internalized by the organism or a metabolite of such a substance.
- a nucleic acid DNA, mRNA, miRNA, rRNA etc.
- a protein cell surface receptor, cytosolic protein etc.
- a metabolite or hormone blood sugar, insulin, estrogen, etc.
- a molecule characteristic of a certain modification of another molecule e.g. sugar moieties or phosphoryl residues on proteins, methyl-residues on genomic DNA
- a biomarker is an organic biomolecule (e.g., a protein, polypeptide, peptide, isomeric form thereof, immunologically detectable fragment thereof, corresponding nucleic acid molecule (e.g., mRNA, cDNA etc)) which is differentially present in a sample taken from a subject having a disease as compared with a subject not having the disease.
- a biomarker is differentially present if the mean or median level of the biomarker in the different groups is calculated to be statistically relevant. Common tests for statistical significance include, among others, t-test (e.g., student t-test), ANOVA, Kruskal-Wallis, Wilcoxon, Mann- Whitney, Receiver Operating Characteristic (ROC curve), accuracy and odds ratio.
- Biomarkers alone or in combination, provide measures of relative risk that a subject belongs to one phenotypic status or another.
- the biomarker referred to herein is measured at the protein level.
- EphA1 EPH receptor A1
- ephrin type-A receptor 1 is a protein that in humans is encoded by the EPHA 1 gene. This gene belongs to the ephrin receptor subfamily of the protein-tyrosine kinase family. EPH and EPH-related receptors have been implicated in mediating developmental events, particularly in the nervous system. Receptors in the EPH subfamily typically have an extracellular part, a transmembrane part, and a cytoplasmic part. The extracellular part contains a ligand binding domain (Ephrin-binding domain), a cysteine- rich EGF-like motif, and two fibronectin type III repeats.
- Ephrin-binding domain a ligand binding domain
- cysteine- rich EGF-like motif a cysteine- rich EGF-like motif
- two fibronectin type III repeats two fibronectin type III repeats.
- the cytoplasmic part contains a protein kinase domain, a SAM domain and a PDZ-binding domain.
- the ephrin receptors are divided into two groups (EphA and EphB receptors) based on the similarity of their extracellular domain sequences and their affinities for binding ephrin-A and ephrin-B ligands (Adu-Gyamfi et al. Biology of Reproduction 2021 , Darling and Lamb, Frontiers in Immunology 2019).
- EphA1 can be accessed via UniProt (see UniProtKB - P21709 EPHA1_HUMAN). There are three isoforms described for EphA1 UniProtKB - P21709-1 , UniProtKB - P21709-2, and UniProtKB - P21709-3.
- EphA1 is a receptor tyrosine kinase which binds to the ephrin-A family ligands residing on adjacent cells. Both EphA1 and Ephrin-A are membrane bound with their binding occurring by direct cell-cell interaction leading to contact-dependent bidirectional signaling into neighboring cells. Forward signaling occurs in Eph receptor-expressing cells whereas reverse signaling occurs in ephrin-expressing cells.
- EphA1 protein Using serum from women with endometriosis (cases) and controls (without endometriosis) seven peptides were identified of the EphA1 protein detected using advanced mass spectrometry proteomics technology. The seven identified peptides described in Table 1 are all part of the extracellular part of the EphA1 protein being part of the ligand binding domain (Ephrin binding domain) and fibronectin type III repeats.
- the EphA1 protein consists of an extracellular domain, a transmembrane domain, and an intracellular domain ( Figure 10).
- the extracellular part of Eph receptors can be proteolytically cleaved and released to the circulating blood (shedding of extracellular part of Eph receptors). Soluble circulating EphA1 is discussed to be involved in immune cell trafficking.
- the determined level of the extracellular protein part of EphA1 is representative of the level of EphA1 in the sample.
- the expressions “EphA1 level” and “level of the extracellular protein part of EphA1” and “EphA1 extracellular protein part level” are used interchangeable herein.
- the methods provided herein refer to “determining” the level of one or more proteins. As would be clear to a person of skill in the art, the level of one or more proteins is typically “determined” by measuring the level of the protein in the sample. The term “determining” can therefore be replaced with the term “measuring” or “determining by measuring” herein.
- determining also refers to assessing/determining whether a woman suffers from endometriosis. Accordingly, assessing/determining as used herein includes diagnosing endometriosis, assessing the risk that a subject suffers from endometriosis, selecting for therapy of endometriosis, monitoring a patient suffering from endometriosis or being treated for endometriosis, by determining the amount or concentration of EphA1 in a sample of the patient, and comparing the determined amount or concentration to a reference.
- the assessment referred to in accordance with the present invention is the assessment of the presence of endometriosis.
- the assessment referred to in accordance with the present invention is to diagnose endometriosis, uteri ne/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain in a subject.
- the assessment referred to in accordance with the present invention is to stratify the risk of a subject to suffer from endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
- measurement preferably comprises a qualitative, a semi-quantitative or a quantitative measurement.
- determining methods may include sending a clinical sample(s) to a commercial laboratory for measurement of the biomarker levels in the biological fluid sample, or the use of commercially available assay kits for measuring the biomarker levels in the biological fluid sample. Exemplary kits and suppliers will be apparent to a person of skill in the art.
- biomarkers may be determined, detected and/or quantified using ELISA assays or lateral flow devices, such as for point-of-care use, as well as spot check colorimetric tests.
- level or “amount” as used herein encompass the absolute amount of a biomarker as referred to herein, the relative amount or concentration of the said biomarker as well as any value or parameter which correlates thereto or can be derived therefrom.
- values or parameters comprise intensity signal values from all specific physical or chemical properties obtained from the said peptides by direct measurements, e.g., intensity values in mass spectra or NMR spectra.
- values or parameters which are obtained by indirect measurements specified elsewhere in this description e.g., response amounts measured from biological read out systems in response to the peptides or intensity signals obtained from specifically bound ligands. It is to be understood that values correlating to the aforementioned amounts or parameters can also be obtained by all standard mathematical operations.
- the level of biomarker present in the biological fluid sample may be determined by e.g. assaying the amount of protein biomarker present in the sample. Assays for measuring the amount of a specified protein are well known in the art and include direct or indirect measures.
- the level of protein biomarker in a sample may also be determined by determining the level of protein biomarker activity in a sample. Accordingly, protein “level” encompasses both the amount of protein per se, or its level of activity.
- the level of a protein biomarker in a biological fluid sample can be determined (e.g., measured) by any suitable methods and materials known in the art, including, for example, a process selected from the group consisting of mass spectrometry, immunoassays, enzymatic assays, spectrophotometry, colorimetry, fluorometry, bacterial assays, protein microarrays, compound separation techniques, or other known techniques for determining the presence and/or quantity of an analyte.
- ELISAs enzyme linked immunosorbent assays
- IA enzyme immunoassay
- RIA radioimmunoassay
- LFDs Lateral Flow Devices
- the level of a protein biomarker in a biological fluid sample is measured by ELISA or lateral flow.
- CA-125 the Carbohydrate antigen 125, sometimes named as Cancer Antigen 125 or Tumor Antigen 125, is a mucin-type glycoprotein, produced by the MUC16 gene, and associated with the cellular membrane.
- CA-125 is a biomarker for epithelial cell ovarian cancer being derived from coelomic epithelia including the endometrium, fallopian tube, ovary, and peritoneum. Diagnostic use of CA-125 is limited to endometriosis stages III and IV (moderate and severe endometriosis) with moderate sensitivity.
- Symptoms of a disease are implications of the disease noticeable by the tissue, organ or organism having such disease and include but are not limited to pain, weakness, tenderness, strain, stiffness, and spasm of the tissue, an organ or an individual.
- “Signs” or “signals” of a disease include but are not limited to the change or alteration such as the presence, absence, increase or elevation, decrease or decline, of specific indicators such as biomarkers or molecular markers, or the development, presence, or worsening of symptoms.
- Symptoms of pain include but are not limited to an unpleasant sensation that may be felt as a persistent or varying burning, throbbing, itching or stinging ache.
- disease and “disorder” are used interchangeably herein, referring to an abnormal condition, especially an abnormal medical condition such as an illness or injury, wherein a tissue, an organ or an individual is not able to efficiently fulfil its function anymore.
- a disease is associated with specific symptoms or signs indicating the presence of such disease. The presence of such symptoms or signs may thus, be indicative for a tissue, an organ or an individual suffering from a disease. An alteration of these symptoms or signs may be indicative for the progression of such a disease.
- a progression of a disease is typically characterised by an increase or decrease of such symptoms or signs which may indicate a "worsening" or “bettering” of the disease.
- the "worsening" of a disease is characterised by a decreasing ability of a tissue, organ or organism to fulfil its function efficiently, whereas the “bettering" of a disease is typically characterised by an increase in the ability of a tissue, an organ or an individual to fulfil its function efficiently.
- a tissue, an organ or an individual being at "risk of developing" a disease is in a healthy state but shows potential of a disease emerging.
- the risk of developing a disease is associated with early or weak signs or symptoms of such disease. In such case, the onset of the disease may still be prevented by treatment.
- Examples of a disease include but are not limited to inflammatory diseases, infectious diseases, cutaneous conditions, endocrine diseases, intestinal diseases, neurological disorders, joint diseases, genetic disorders, autoimmune diseases, traumatic diseases, and various types of cancer.
- Endometriosis is a chronic, hormone-dependent, inflammatory disease that is characterized by lesions of endometrial-like tissue outside of the uterus. Clinical presentation of endometriosis varies significantly from patient to patient. Endometriosis patients often present with symptoms such as intermenstrual bleeding, painful periods (dysmenorrhea), painful intercourse (dyspareunia), painful defecation (dyschezia) and painful urination (dysuria). Pelvic pain due to endometriosis is usually chronic (lasting >6 months) and is associated with dysmenorrhea (in 50 to 90% of cases), dyspareunia, deep pelvic pain, and lower abdominal pain with or without back and loin pain.
- the pain can occur unpredictably and intermittently throughout the menstrual cycle or it can be continuous, and it can be dull, throbbing, or sharp, and exacerbated by physical activity.
- Bladder- and bowel-associated symptoms are typically cyclic. Pain often worsens overtime and may change in character; infrequently, women report burning or hypersensitivity, symptoms that are suggestive of a neuropathic component. Often, endometriosis can be asymptomatic, only coming to a clinician’s attention during evaluation for infertility (Sinaii et al. Fertil Steril. 2008; 89(3): 538-545).
- endometriosis The most commonly affected sites of endometriosis are the pelvic organs and peritoneum, although other parts of the body such as the lungs are occasionally affected.
- the extent of the disease varies from a few, small lesions on otherwise normal pelvic organs to large, ovarian endometriotic cysts (endometriomas) and/or extensive fibrosis and adhesion formation causing marked distortion of pelvic anatomy. Based on the location, endometriotic lesions can be classified into peritoneal endometriosis, ovarian endometriotic cysts (endometrioma), deep nodules (deep infiltrating endometriosis; Kennedy et al. Hum Reprod. 2005; 20(10): 2698- 2704).
- Deep infiltrating endometriosis is considered to be any manifestation of endometriosis that is located other than in the superficial tissues of the rectovaginal septum and vaginal fornix, the pelvic wall, parametrium, bowel, uterus, or urinary bladder (Halis. et al. (2010). Deutsches Cardioeblatt International, 107(25), 446).
- Endometriosis can also involve the diaphragm (diaphragmatic endometriosis) or involve the thorax (thoracic endometriosis) (Nezhat et al. JSLS 2019).
- uterine/pelvic pathology and “uterine and pelvic pathology” are used interchangeably herein and comprise adenomyosis, uterine fibroids, ovarian cysts requiring surgery, and uterine/pelvic cancer such as ovarian cancer or endometrial cancer.
- endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain refers to neuropathic pain defined by the International Association for the Study of Pain as “pain caused by disease or lesion of the somatosensory nervous system,” contrasting with nociceptive pain which is defined as “pain that arises from actual or threatened damage to nonneural tissue and is due to the activation of nociceptors”.
- Neuropathic pain could be expected to arise in the context of endometriosis for a number of reasons. (Coxon et al., /s there a Neuropathic-Like Component to Endometriosis-Associated Pain? Results From a Large Cohort Questionnaire Study, Front Pain Res (Lausanne), 2021 ; 2: 743812; doi: 10.3389/fpain.2021.743812).
- rASRM stage or “rASRM staging” refers to the revised classification system established by the American Society for Reproductive Medicine (ASRM) describing the severity of endometriosis based on the findings at surgery (laparoscopy). The classification is based on the morphology of peritoneal and pelvic implants such as red, white and black lesions, percentage of involvement of each lesion should be included. Number, size, and location endometrial implants, plaques, endometriomas and adhesions should be noted. Endometriosis in bowel, urinary tract, fallopian tube, vagina, cervix, skin, or other locations should be documented per ASRM guidelines.
- Stages of endometriosis according to ASRM guidelines are stage I, II, III, and IV determined based on the point scores and correspond to minimal, mild, moderate and severe endometriosis.
- the rASRM stages I & II endometriosis are defined by superficial peritoneal endometriosis, possible presence of small deep lesions, absence of endometrioma and/or mild filmy adhesion.
- the rASRM stages III and IV endometriosis are defined by the presence of superficial peritoneal endometriosis, deep infiltrating endometriosis with moderate to extensive adhesions between the uterus and bowels and/or endometrioma cysts with moderate to extensive adhesions involving the ovaries and tubes.
- VAS Visual Analog Scale
- the VAS consists of a 10-cm long horizontal line with its extremes marked as ‘no pain’ and ‘worst pain imaginable’. Each patient ticks her pain level on the line and the distance from ‘no pain’ on the extreme left to the tick mark is measured in centimeters, yielding a pain score from 0 to 10. ‘No pain’ corresponds to a pain score of 0, ‘worst pain imaginable’ corresponds to a pain score of 10.
- VAS score the Visual Analog Scale, is an instrument to assess the intensity of pain.
- the VAS consists of a 10-cm long horizontal line with its extremes marked as ‘no pain’ and ‘worst pain imaginable’. Each patient ticks her pain level on the line and the distance from ‘no pain’ on the extreme left to the tick mark is measured in centimeters, yielding a pain score from 0 to
- the subject may be referred to herein as a patient.
- the terms “subject”, “individual”, and “patient” are used herein interchangeably and refer to an animal, preferably a mammal and, more typically to a human.
- the patient is preferably a human female.
- the patient is preferably a young or adolescent human female aged between 12- 24 years.
- the patient is a young or adolescent human female.
- the subject can be symptomatic (e.g., the subject presents symptoms associated with endometriosis), or the subject can be asymptomatic (e.g., the subject does not present symptoms associated with endometriosis).
- the subject may be diagnosed with, be at risk of developing or present with symptoms of endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
- the subject may have, or be suspected of having (e.g., present with symptoms or a history indicative or suggestive of) endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
- the subject has endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain (and the method diagnoses, identifies, (or detects) that the subject has endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain).
- diagnosis “identify”, and “detect” can be used interchangeably.
- the subject has early stage (stage I or stage II) endometriosis.
- the patient to be investigated by the method of the present invention shall be a patient having suspected endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
- the term “suspected endometriosis” as used herein means that the patient shall exhibit clinical parameters, signs and/or symptoms of endometriosis.
- the patient according to the invention is, typically, a patient that suffers from an endometriosis or is suspected to suffer from an endometriosis.
- EphA1 levels may be determined routinely as part of screening tests without any suspicion on endometriosis but to be able to detect symptom-free endometriosis at early stages.
- comparing refers to comparing the amount/level of the biomarker in the sample from the subject with the reference amount or reference value of the biomarker specified elsewhere in this description. It is to be understood that comparing as used herein usually refers to a comparison of corresponding parameters or values, e.g., an absolute amount is compared to an absolute reference amount while a concentration is compared to a reference concentration or an intensity signal obtained from the biomarker in a sample is compared to the same type of intensity signal obtained from a reference sample.
- the comparison may be carried out manually or computer assisted. Thus, the comparison may be carried out by a computing device.
- the value of the measured or detected amount of the biomarker in the sample from the subject and the reference amount can be, e.g., compared to each other and the said comparison can be automatically carried out by a computer program executing an algorithm for the comparison.
- the computer program carrying out the said evaluation will provide the desired assessment in a suitable output format.
- the value of the measured amount may be compared to values corresponding to suitable references which are stored in a database by a computer program.
- the computer program may further evaluate the result of the comparison, i.e. automatically provide the desired assessment in a suitable output format.
- the value of the measured amount may be compared to values corresponding to suitable references which are stored in a database by a computer program.
- the computer program may further evaluate the result of the comparison, i.e. automatically provides the desired assessment in a suitable output format.
- (appropriate) reference value refers to a sample which is analysed in a substantially identical manner as the sample of interest and whose information is compared to that of the sample of interest.
- the “(appropriate) reference value” is a predetermined reference.
- a reference sample thereby provides a standard allowing for the evaluation of the information obtained from the sample of interest.
- a control sample may be derived from a body fluid of a healthy individual, in particular serum or plasma for a non-invasive test, thereby providing a standard of a healthy status of a tissue, organ or individual. Differences between the status of the normal reference sample and the status of the sample of interest may be indicative of the presence or further progression of such disease or disorder.
- a control sample may be derived from an abnormal or diseased tissue, organ or individual thereby providing a standard of a diseased status of a tissue, organ or individual. Differences between the status of the normal or abnormal reference sample and the status of the sample of interest may be indicative of the absence or bettering of such disease or disorder.
- a reference sample may also be derived from the same tissue, organ, or individual as the sample of interest but has been taken at an earlier time point. Differences between the status of the earlier taken reference sample and the status of the sample of interest may be indicative of the progression of the disease, i.e. a bettering or worsening of the disease over time.
- the determined value can be compared to more than one (appropriate) reference values, which can be of different kind.
- the determined value can be compared to one or more values obtained from the same subject at earlier time points and in parallel it can be compared to one or more values obtained from other subjects (with a known stage of endometriosis).
- the control sample may be an internal or an external control sample.
- An internal control sample is used, i.e. the marker level(s) is(are) assessed in the test sample as well as in one or more other sample(s) taken from the same subject to determine if there are any changes in the level(s) of said marker(s).
- For an external control sample the presence or amount of a marker in a sample derived from the individual is compared to its presence or amount in an individual known to suffer from, or known to be at risk of, a given condition; or an individual known to be free of a given condition, i.e., "normal individual".
- such external control sample may be obtained from a single individual or may be obtained from a reference population that is age-matched and free of confounding diseases. Typically, samples from 100 well-characterized individuals from the appropriate reference population are used to establish a "reference value". However, reference population may also be chosen to consist of 20, 30, 50, 200, 500 or 1000 individuals. Healthy individuals represent a preferred reference population for establishing a control value.
- a marker concentration in a patient sample can be compared to a concentration known to be associated with a specific course of a certain disease. For example, it can be compared to a concentration known to be associated with a certain stage of endometriosis.
- the sample's marker concentration is directly or indirectly correlated with a diagnosis and the marker concentration is e.g. used to determine whether an individual is at risk for a certain suffering from that disease.
- the marker concentration can be compared to marker concentrations obtained from the same subject at an earlier time point.
- the sample's marker concentration can e.g., be compared to a marker concentration known to be associated with a response to therapy in a certain disease, the diagnosis of a certain disease, the assessment of the severity of a certain disease, the guidance for selecting an appropriate drug to a certain disease, in judging the risk of disease progression, or in the follow-up of patients.
- an appropriate control sample is chosen and a control or reference value for the marker established therein.
- the absolute marker values established in a control sample will be dependent on the assay used.
- corresponding subjects from which these samples are obtained are “symptomatic subjects” respectively.
- Symptomatic controls refer to control samples of subjects that suffer from symptoms that are usually associated with endometriosis (e.g., menstrual/abdominal pain, infertility, etc) but where, based on laparoscopy, endometriosis can be excluded and no tissue alterations (e.g., adenomyosis, uterine/ovarian cysts, fibroids or uterine/pelvic cancer such as ovarian cancer, endometrial cancer) can be observed, that is symptomatic controls do not have endometriosis or any other uterine/pelvic pathology (e.g., adenomyosis, uterine/ovarian cysts, fibroids, uterine/pelvic cancer such as ovarian cancer and endometrial cancer).
- endometriosis e.g., menstrual/abdominal pain, infertility, etc
- endometriosis e.g., menstrual/abdominal pain, infer
- Women with other “uterine/pelvic pathology” refer to a group of samples of subjects that have tissue alterations (e.g., adenomyosis, fibroids, uterine/ovarian cysts requiring surgery, and uterine/pelvic cancer) which however does not resemble endometriosis. Further, these subjects are most often symptomatic (e.g., menstrual/abdominal pain, infertility, etc).
- the control sample may be assayed at the same time, before or after, separately or simultaneously with the test sample.
- the control value that is used in the comparison with the test sample may be a value that is calculated as an average or median of more than one (e.g., two or more, five or more, ten or more, a group etc) of control samples.
- the control sample may be a sample that originated from (i.e., is a mix of) more than one (e.g., two or more, five or more, ten or more, a group etc) individual that is not suffering from endometriosis (or is a “symptomatic control”) and not from other uterine/pelvic pathology.
- control sample is obtained from a subject that is a “symptomatic control” not having endometriosis or another uterine/pelvic pathology (adenomyosis, fibroids, ovarian cysts requiring surgery, uterine/pelvic cancer).
- the level of biomarker (e.g., protein) in the biological fluid sample may be compared to a pre-determined reference level for the biomarker of interest.
- a “predetermined reference level” refers to a biomarker level obtained from a reference database, which may be used to generate a pre-determined cut off value, i.e., a score that is statistically predictive of endometriosis.
- the predetermined reference level is the average or median level of the biomarker in at least one individual not suffering from endometriosis from the same species.
- the predetermined reference value may be calculated as the average or median, taken from a group or population of individuals that are not suffering from endometriosis.
- the predetermined reference value may be calculated as the average or median, taken from a group or population of individuals that are “symptomatic controls”.
- the individual or the population of individuals can be the same age or in the same state or condition of health as the subject from which the test sample is obtained.
- the pre-determined reference level is therefore the average level of the biomarker in a control subject that does not have endometriosis.
- the pre- determined reference level is the average level of the biomarker in a subject that is a “symptomatic control”.
- control sample or predetermined reference are obtained from an individual or group of individuals that are distinct from the subject that is being tested (i.e., the subject from which the test sample is obtained/provided).
- the control or predetermined reference are used as a bench line to determine whether the tested subject has endometriosis.
- control or predetermined reference value may be obtained from the same individual as the test sample, but at an earlier time point.
- This is particularly relevant for the methods described herein that determine the progression in a subject, that determine the therapeutic effect of a treatment regimen for endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain, and/or that determine a subject’s compliance or adherence with a prescribed treatment regimen for endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain.
- the samples are taken from the same biological fluid of the same subject, wherein the biological fluid is blood, serum, plasma, capillary blood, interstitial fluid, peritoneal fluid, or menstrual fluid preferably the biological fluid sample is serum.
- control sample or predetermined reference level is used to determine any changes in the level of the biomarker(s) over a time interval for the same subject.
- the predetermined reference level or control sample can therefore be from the same subject that the test sample is obtained from, for example obtained at an earlier time point. This earlier time point can be before they were diagnosed with endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
- a pre-determined level can be single cut-off value, such as a median or mean. It can be a range of cut-off (or threshold) values, such as a confidence interval. It can be established based upon comparative groups, such as where the risk in one defined group is a fold higher, or lower, (e.g., approximately 2-fold, 4-fold, 8-fold, 16-fold or more) than the risk in another defined group.
- the reference could be a calculated reference, most preferably the average or median, for the relative or absolute amount of a biomarker of a population of individuals comprising the subject to be investigated. How to calculate a suitable reference value, preferably, the average or median, is well known in the art.
- the level of the protein biomarker in a subject being less than or equal to the level of the biomarker of the control sample or pre-determined reference level is indicative of a clinical status (e.g., indicative of endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain).
- the greater than, or the less than, that is sufficient to distinguish a subject from a control subject is a statistically significantly greater than, or a statistically significant less than.
- the "being equal” refers to being approximately equal (e.g., not statistically different).
- the pre-determined value can depend upon a particular population of subjects (e.g., human subjects) selected. For example, an apparently healthy population will have a different 'normal' range of the protein biomarker than will a population of subjects which have, or are likely to have, endometriosis. Accordingly, the pre-determined values selected may take into account the category (e.g., healthy, diseased, stage of disease) in which a subject (e.g., human subject) falls.
- the category e.g., healthy, diseased, stage of disease
- the level of the specific biomarker detected in a sample may be normalized by adjusting the measured level (amount or activity) of the biomarker using the level of a reference protein in the same sample, wherein the reference protein is not a marker itself (it is e.g., a protein that is constitutively expressed).
- This normalization allows the comparison of the biomarker level in one sample to another sample, or between samples from different sources. This normalized level can then optionally be compared to a reference value or control.
- the biomarker when measuring a protein biomarker in a whole blood sample the biomarker may be expressed as an absolute concentration or, alternatively, it may be normalized against a known protein constitutively expressed in whole blood such as albumin, immunoglobulins or plasma protein concentration.
- the biomarker when measuring a protein biomarker in a serum (or plasma) sample the biomarker may be expressed as an absolute concentration or, alternatively, it may be normalized against a known protein constitutively expressed in serum (or plasma).
- the biomarker level(s) in the test sample may be compared to the level of the same biomarker in a control sample or with a pre-determined reference level for the same biomarker to identify an increase or decrease in a level of the one or more biomarker in the sample of the subject.
- the subject may be identified as having endometriosis if the comparison (between biomarker level(s) in the control sample/predetermined reference value and the test sample of the subject) indicates that the subject has a decreased level of EphA1 compared to the control sample or the pre-determined reference level.
- the prediction is made within a predictive window of 6 month and two years. More typically, said predictive window is about a time window of about 6 months to 12 month for a non-invasive test dependent on the symptoms, such as pelvic pain.
- the assessment made in accordance with the present invention may usually not be correct for 100% of the investigated subjects.
- the term typically, requires that a statistically significant portion of subjects can be correctly assessed. Whether a portion is statistically significant can be determined without further ado by the person skilled in the art using various well known statistic evaluation tools, e.g., determination of confidence intervals, p-value determination, Student's t-test, Mann-Whitney test, etc. Details may be found in Dowdy and Wearden, Statistics for Research, John Wiley & Sons, New York 1983. Typically envisaged confidence intervals are at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%.
- the p-values are, typically, 0.2, 0.1 , 0.05.
- the terms “lowered” or “decreased” level of an indicator refer to the level of such indicator in the sample being reduced in comparison to the reference (value) or reference sample.
- the terms “decrease”, “decreased” “reduced”, “reduction” or “down- regulated”, “lower” are all used herein generally to mean a decrease by a statistically significant amount.
- “reduced”, “reduction”, “decreased” or “decrease” means a decrease by at least 10% as compared to a reference level/control, for example a decrease by at least about 20%, or at least about 30%, or at least about 40%, or at least about 50%, or at least about 60%, or at least about 70%, or at least about 80%, or at least about 90% or up to and including a 100% decrease (i.e.
- any decrease between 10-100% as compared to a reference level/control or at least about a 0.5-fold, or at least about a 1.0-fold, or at least about a 1.2-fold, or at least about a 1.5-fold, or at least about a 2-fold, or at least about a 3 -fold, or at least about a 4-fold, or at least about a 5-fold or at least about a 10-fold decrease, or any decrease between 1.0-fold and 10-fold or greater as compared to a reference level/control.
- an indicator/(bio)marker refers to the level of such indicator in the sample being higher in comparison to the reference (value) or reference sample.
- a protein that is detectable in higher amounts in a fluid sample of one individual suffering from a given disease than in the same fluid sample of individuals not suffering from said disease has an elevated level.
- the terms “increased”, “increase” or “up-regulated”, “higher” are all used herein to generally mean an increase by a statically significant amount; for the avoidance of any doubt, the terms “increased” or “increase” means an increase of at least 10% as compared to a reference level/control, for example an increase of at least about 20%, or at least about 30%, or at least about 40%, or at least about 50%, or at least about 60%, or at least about 70%, or at least about 80%, or at least about 90% or up to and including a 100% increase or any increase between 10-100% as compared to a reference level/control, or at least about a 0.5-fold, or at least about a 1.0-fold, or at least about a 1.2-fold, or at least about a 1 .5-fold, or at least about a 2-fold, or at least about a 3-fold, or at least about a 4-fold, or at least about a 5 -fold or at least about a 10-fold increase, or any increase between
- lower(ed) or “decreased” level of an indicator/(bio)marker refer to the level of such indicator in the sample being lower in comparison to the reference (value) or reference sample.
- a protein that is detectable in lower amounts in a fluid sample of one individual suffering from a given disease than in the same fluid sample of individuals not suffering from said disease has a lowered level.
- the terms “less”, “decrease” or “downregulated-regulated”, “lower” are all used herein to generally mean a decrease by a statistically significant amount; for the avoidance of any doubt, the terms “decreased” or “decrease” means a decrease of at least 10% as compared to a reference level/control, for example a decrease of at least about 20%, or at least about 30%, or at least about 40%, or at least about 50%, or at least about 60%, or at least about 70%, or at least about 80%, or at least about 90% or up to and including a 100% decrease or any decrease between 10-100% as compared to a reference level/control, or at least about a 0.5-fold, or at least about a 1.0-fold, or at least about a 1.2- fold, or at least about a 1 .5-fold, or at least about a 2-fold, or at least about a 3-fold, or at least about a 4-fold, or at least about a 5 -fold or at least about a 10-fold decrease, or any decrease
- immunoglobulin refers to immunity conferring glycoproteins of the immunoglobulin superfamily.
- surface immunoglobulins are attached to the membrane of effector cells by their transmembrane region and encompass molecules such as but not limited to B-cell receptors, T -cell receptors, class I and II major histocompatibility complex (MHC) proteins, beta-2 microglobulin ( ⁇ 2M), CD3, CD4 and CDS.
- MHC major histocompatibility complex
- ⁇ 2M beta-2 microglobulin
- CD3, CD4 and CDS CDS.
- antibody refers to secreted immunoglobulins which lack the transmembrane region and can thus, be released into the bloodstream and body cavities.
- Human antibodies are grouped into different isotypes based on the heavy chain they possess. There are five types of human lg heavy chains denoted by the Greek letters: a, y, 5, E, and p.- The type of heavy chain present defines the class of antibody, i.e. these chains are found in IgA, IgD, IgE, IgG, and IgM antibodies, respectively, each performing different roles, and directing the appropriate immune response against different types of antigens.
- Distinct heavy chains differ in size and composition; and may comprise approximately 450 amino acids (Janeway et al. (2001) Immunobiology, Garland Science).
- IgA is found in mucosal areas, such as the gut, respiratory tract and urogenital tract, as well as in saliva, tears, and breast milk and prevents colonization by pathogens (Underdown & Schiff (1986) Annu. Rev. Immunol. 4:389- 417).
- IgD mainly functions as an antigen receptor on B cells that have not been exposed to antigens and is involved in activating basophils and mast cells to produce antimicrobial factors (Geisberger et al. (2006) Immunology 118:429-437; Chen et al. (2009) Nat. Immunol.
- IgE is involved in allergic reactions via its binding to allergens triggering the release of histamine from mast cells and basophils. IgE is also involved in protecting against parasitic worms (Pier et al. (2004) Immunology, Infection, and Immunity, ASM Press). IgG provides the majority of antibody-based immunity against invading pathogens and is the only antibody isotype capable of crossing the placenta to give passive immunity to fetus (Pier et al. (2004) Immunology, Infection, and Immunity, ASM Press).
- IgG subclasses In humans there are four different IgG subclasses (IgGI, 2, 3, and 4), named in order of their abundance in serum with IgGI being the most abundant (-66%), followed by lgG2 (-23%), lgG3 (-7%) and IgG (-4%).
- the biological profile of the different IgG classes is determined by the structure of the respective hinge region.
- IgM is expressed on the surface of B cells in a monomeric form and in a secreted pentameric form with very high avidity. IgM is involved in eliminating pathogens in the early stages of B cell mediated (humoral) immunity before sufficient IgG is produced (Geisberger et al. (2006) Immunology 118:429-437).
- Antibodies are not only found as monomers but are also known to form dimers of two Ig units (e.g. IgA), tetramers of four Ig units (e.g. IgM of teleost fish), or pentamers of five Ig units (e.g. mammalian IgM).
- Antibodies are typically made of four polypeptide chains comprising two identical heavy chains and identical two light chains which are connected via disulfide bonds and resemble a "Y"-shaped macro-molecule. Each of the chains comprises a number of immunoglobulin domains out of which some are constant domains and others are variable domains. Immunoglobulin domains consist of a 2-layer sandwich of between 7 and 9 antiparallel --strands arranged in two --sheets.
- the heavy chain of an antibody comprises four Ig domains with three of them being constant (CH domains: CHI. CH2. CH3) domains and one of the being a variable domain (V H).
- the light chain typically comprises one constant Ig domain (CL) and one variable Ig domain (V L).
- the human IgG heavy chain is composed of four Ig domains linked from N- to C- terminus in the order VwCH1-CH2-CH3 (also referred to as VwCyl-Cy2-Cy3), whereas the human IgG light chain is composed of two immunoglobulin domains linked from N- to C- terminus in the order VL-CL, being either of the kappa or lambda type (VK-CK or VA.-CA.).
- the constant chain of human IgG comprises 447 amino acids. Throughout the present specification and claims, the numbering of the amino acid positions in an immunoglobulin are that of the "Ell index" as in Kabat, E.
- CH domains in the context of IgG are as follows: "CHI” refers to amino acid positions 118-220 according to the Ell index as in Kabat; "CH2” refers to amino acid positions 237-340 according to the Ell index as in Kabat; and "CH3” refers to amino acid positions 341-44 7 according to the Ell index as in Kabat.
- full-length antibody “intact antibody”, and “whole antibody” are used herein interchangeably to refer to an antibody in its substantially intact form, not antibody fragments as defined below.
- Papain digestion of antibodies produces two identical antigen binding fragments, called “Fab fragments” (also referred to as “Fab portion” or “Fab region”) each with a single antigen binding site, and a residual “Fe fragment” (also referred to as “Fe portion” or “Fe region”) whose name reflects its ability to crystallize readily.
- Fab fragments also referred to as “Fab portion” or “Fab region”
- Fe portion also referred to as “Fe portion” or “Fe region
- the Fe region is composed of two identical protein fragments, derived from the CH2 and CH3 domains of the antibody's two heavy chains; in IgM and IgE isotypes, the Fe regions contain three heavy chain constant domains (CH2-4) in each polypeptide chain.
- CH2-4 heavy chain constant domains
- smaller immunoglobulin molecules exist naturally or have been constructed artificially.
- the term "Fab 1 fragment” refers to a Fab fragment additionally comprise the hinge region of an Ig molecule whilst “F(ab')2 fragments” are understood to comprise two Fab' fragments being either chemically linked or connected via a disulfide bond. Whilst “single domain antibodies (sdAb )" (Desmyter et al.
- scFv single chain Fv
- di-scFvs Divalent single-chain variable fragments
- scFvA-scFvB Divalent single-chain variable fragments
- Bispecific diabodies are formed by expressing to chains with the arrangement VHA-VLB and VHB-VLA or VLA-VHB and VLB-VHA, respectively.
- Singlechain diabodies comprise a VHA-VLB and a VHB-VLA fragment which are linked by a linker peptide (P) of 12-20 amino acids, preferably 14 amino acids, (VHA-VLB-P-VHB-VLA).
- Bi-specific T-cell engagers (BiTEs)" are fusion proteins consisting of two scFvs of different antibodies wherein one of the scFvs binds to T cells via the CD3 receptor, and the other to a tumor cell via a tumor specific molecule (Kufer et al. (2004) Trends Biotechnol. 22:238-244).
- Dual affinity retargeting molecules (“DART” molecules) are diabodies additionally stabilized through a C-terminal disulfide bridge.
- antibody fragments refers to a portion of an intact antibody, preferably comprising the antigen-binding region thereof.
- Antibody fragments include but are not limited to Fab, Fab', F(ab')2, Fv fragments; diabodies; sdAb, nanobodies, scFv, di-scFvs, tandem scFvs, triabodies, diabodies, scDb, BiTEs, and DARTs.
- binding affinity generally refers to the strength of the sum total of noncovalent interactions between a single binding site of a molecule (e.g., an antibody) and its binding partner (e.g., an antigen). Unless indicated otherwise, as used herein, “binding affinity” refers to intrinsic binding affinity which reflects a 1 :1 interaction between members of a binding pair (e.g., antibody and antigen).
- the affinity of a molecule X for its partner Y can generally be represented by the dissociation constant (Kd). Affinity can be measured by common methods known in the art, including but not limited to surface plasmon resonance-based assay (such as the BIAcore assay as described in PCT Application Publication No.
- Low- affinity antibodies generally bind antigen slowly and tend to dissociate readily, whereas high- affinity antibodies generally bind antigen faster and tend to remain bound longer.
- a variety of methods of measuring binding affinity are known in the art, any of which can be used for purposes of the present invention.
- “Sandwich immunoassays” are broadly used in the detection of an analyte of interest.
- the analyte is “sandwiched” in between a first antibody and a second antibody.
- a sandwich assay requires that capture and detection antibody bind to different, nonoverlapping epitopes on an analyte of interest. By appropriate means such sandwich complex is measured and the analyte thereby quantified.
- a first antibody bound to the solid phase or capable of binding thereto and a detectably-labeled second antibody each bind to the analyte at different and non-overlapping epitopes.
- the first analyte-specific binding agent (e.g., an antibody) is either covalently or passively bound to a solid surface.
- the solid surface is typically glass or a polymer, the most commonly used polymers being cellulose, polyacrylamide, nylon, polystyrene, polyvinyl chloride, or polypropylene.
- the solid supports may be in the form of tubes, beads, discs of microplates, or any other surface suitable for conducting an immunoassay.
- the binding processes are well- known in the art and generally consist of cross-linking covalently binding or physically adsorbing, the polymer-antibody complex is washed in preparation for the test sample.
- an aliquot of the sample to be tested is then added to the solid phase complex and incubated for a period of time sufficient ⁇ e.g., 2-40 minutes or overnight if more convenient) and under suitable conditions (e.g., from room temperature to 40°C such as between 25° C and 37° C inclusive) to allow for binding between the first or capture antibody and the corresponding antigen.
- suitable conditions e.g., from room temperature to 40°C such as between 25° C and 37° C inclusive
- the solid phase, comprising the first or capture antibody and bound thereto the antigen can be washed, and incubated with a secondary or labeled antibody binding to another epitope on the antigen.
- the second antibody is linked to a reporter molecule which is used to indicate the binding of the second antibody to the complex of first antibody and the antigen of interest.
- An extremely versatile alternative sandwich assay format includes the use of a solid phase coated with the first partner of a binding pair, e.g., paramagnetic streptavidin-coated microparticles. Such microparticles are mixed and incubated with an analyte-specific binding agent bound to the second partner of the binding pair (e.g., a biotinylated antibody), a sample suspected of comprising or comprising the analyte, wherein said second partner of the binding pair is bound to said analyte-specific binding agent, and a second analyte-specific binding agent which is detectably labeled.
- an analyte-specific binding agent bound to the second partner of the binding pair e.g., a biotinylated antibody
- a sample suspected of comprising or comprising the analyte wherein said second partner of the binding pair is bound to said analyte-specific binding agent
- a second analyte-specific binding agent which is detectably labeled
- these components are incubated under appropriate conditions and for a period of time sufficient for binding the labeled antibody via the analyte, the analyte-specific binding agent (bound to) the second partner of the binding pair and the first partner of the binding pair to the solid phase microparticles.
- assay may include one or more washing step(s).
- detectably labeled encompasses labels that can be directly or indirectly detected.
- Directly detectable labels either provide a detectable signal or they interact with a second label to modify the detectable signal provided by the first or second label, e.g., to give FRET (fluorescence resonance energy transfer).
- Labels such as fluorescent dyes and luminescent (including chemiluminescent and electrochemiluminescent) dyes (Briggs et al "Synthesis of Functionalised Fluorescent Dyes and Their Coupling to Amines and Amino Acids," J. Chem. Soc., Perkin-Trans. 1 (1997) 1051-1058) provide a detectable signal and are generally applicable for labeling.
- detectably labeled refers to a label providing or inducible to provide a detectable signal, i.e. , to a fluorescent label, to a luminescent label (e.g., a chemiluminescent label or an electrochemiluminescent label), a radioactive label or a metalchelate based label, respectively.
- Fluorescent dyes are e.g., described by Briggs et al "Synthesis of Functionalized Fluorescent Dyes and Their Coupling to Amines and Amino Acids," J. Chem. Soc., Perkin-Trans. 1 (1997) 1051-1058).
- Fluorescent labels orfluorophores include rare earth chelates (europium chelates), fluorescein type labels including FITC, 5-carboxyfluorescein, 6-carboxy fluorescein; rhodamine type labels including TAMRA; dansyl; Lissamine; cyanines; phycoerythrins; Texas Red; and analogs thereof.
- the fluorescent labels can be conjugated to an aldehyde group comprised in target molecule using the techniques disclosed herein.
- Fluorescent dyes and fluorescent label reagents include those which are commercially available from Invitrogen/Molecular Probes (Eugene, Oregon, USA) and Pierce Biotechnology, Inc. (Rockford, III.).
- Luminescent dyes or labels can be further subcategorized into chemiluminescent and electrochemiluminescent dyes.
- chemiluminogenic labels include luminol, acridinium compounds, coelenterazine and analogues, dioxetanes, systems based on peroxyoxalic acid and their derivatives.
- acridinium based labels are used (a detailed overview is given in Dodeigne C. et al., Taianta 51 (2000) 415-439).
- Electrochemiluminescense proved to be very useful in analytical applications as a highly sensitive and selective method. It combines analytical advantages of chemiluminescent analysis (absence of background optical signal) with ease of reaction control by applying electrode potential.
- Ruthenium complexes especially [Ru (Bpy)3]2+ (which releases a photon at -620 nm) regenerating with TPA (Tripropylamine) in liquid phase or liquid-solid interface are used as ECL-labels.
- Electrochemiluminescent (ECL) assays provide a sensitive and precise measurement of the presence and concentration of an analyte of interest. Such techniques use labels or other reactants that can be induced to luminesce when electrochemically oxidized or reduced in an appropriate chemical environment. Such electrochemiluminescense is triggered by a voltage imposed on a working electrode at a particular time and in a particular manner. The light produced by the label is measured and indicates the presence or quantity of the analyte.
- ECL Electrochemiluminescent
- Radioactive labels make use of radioisotopes (radionuclides), such as 3H, 11 C, 14C, 18F, 32P, 35S, 64Cu, 68Gn, 86Y, 89Zr, 99TC, 1111n, 1231, 1241, 1251, 1311, 133Xe, 177Lu, 211 At, or 131 Bi.
- radioisotopes radioisotopes (radionuclides)
- the methods described herein can further comprise selecting, and optionally administering, a treatment regimen for the subject based on the diagnosis (i.e. , based on the comparison of the levels of the biomarkers with the reference values/levels/controls).
- Treatment can include, for example, surgery and, in some cases, therapy, or combinations thereof. However, in some cases, immediate treatment may not be required, and the subject may be selected for active surveillance.
- active surveillance As used herein, the terms “active surveillance”, “monitoring” and “watchful waiting” are used interchangeably herein to mean closely monitoring a patient's condition without giving any treatment until symptoms appear or change.
- treatment refers to therapeutic treatment, wherein the object is to slow down (lessen) the targeted condition, disorder or symptom.
- Treatment therefore encompasses a reduction, slowing or inhibition of the symptoms of endometriosis, for example of at least 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90% or 100% when compared to the symptoms before treatment.
- appropriate treatment may include pain medication, hormone treatments (such as hormonal contraceptives), gonadotropin-releasing hormone (GnRH) agonists, and/or surgery.
- hormone treatments such as hormonal contraceptives
- GnRH gonadotropin-releasing hormone
- the term “surgery” applies to surgical methods undertaken for removal of endometric tissue, like, for example, laparoscopy or nerve sparing surgery.
- the term “therapy” includes drug-based therapy, radiation, hormonal therapy, cryosurgery, chemotherapy, immunotherapy, biologic therapy, and high-intensity focused ultrasound.
- Drug-based therapy of endometriosis can for example be by inhibiting or targeting neurogenic inflammation and/or pain medication and/or hormonal therapy.
- the type of treatment will vary depending on the particular form and/or stage of endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain that the subject has, is suspected of having.
- the inventors have surprisingly identified the new protein biomarker EphA1 that is decreased in biological fluids, in particular serum, of women with endometriosis, especially in women with early stages of endometriosis.
- the biomarker EphA1 can be used for diagnosing endometriosis in a subject compared to a control (e.g., non-pathological subjects or symptomatic subjects).
- serum EphA1 can be used as a blood biomarker for early diagnosis and risk stratification of endometriosis. Furthermore, serum EphA1 can be used to select patients with disease stage I and stage II for early medical management of endometriosis. Therefore, it can significantly reduce endometriosis diagnostic delay, improve patients' lives, and reduce the economic burden.
- the biomarker can advantageously be used in any of the methods, kits, assays, or uses described herein.
- the present invention relates to a method for assessing endometriosis in a subject, the method comprising the steps of: a) determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject, b) comparing the level of the extracellular protein part of EphA1 to at least one appropriate reference value of a level of the extracellular protein part of EphA1, c) identifying a subject as having endometriosis if the comparison in step b) indicates that the subject has a decreased level of the extracellular protein part of EphA1 compared to the appropriate reference value, wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
- r-ASRM revised scoring system of the American Society for Reproductive Medicine
- the method is an in vitro method.
- the at least one appropriate reference value is i. a level of EphA1 in a SNF subject or in a SOF subject, or is ii. an average level of EphA1 in a group of SNF subjects or in a group of SOF subjects, or a combination thereof, or wherein the at least one appropriate reference value is iii. a predetermined value of a level of EphA1 in a SNF subject or SOF subject, or is iv. a predetermined average value of a level of EphA1 in a group of SNF subjects or in a group of SOF subjects, or a combination thereof.
- the inventors could detect a decrease of serum EphA1 in the early and late stages of endometriosis compared to the control levels.
- an amount of EphA1 lowered by 100% or more is indicative of the presence of endometrioses.
- an amount of EphA1 lowered by 150% or more is indicative of the presence of endometrioses.
- an amount of EphA1 lowered by 200% or more is indicative of endometrioses.
- an amount of EphA1 lowered by 10% or more is indicative of the presence or risk of having or developing endometriosis.
- the biological fluid sample is blood, serum, plasma, capillary blood, interstitial fluid, peritoneal fluid, or menstrual fluid, preferably the biological fluid sample is serum.
- the biological fluid sample is serum.
- the sample is an in vitro sample, i.e., it will be analyzed in vitro and not transferred back into the body of the subject.
- the method of the present invention is an in vitro method.
- the subject is a human subject.
- the patient is a female human subject.
- the subject is a young or adolescent human female.
- the subject is a subject who is capable of suffering from endometriosis due to the physical condition.
- the assessment is carried out without performing laparoscopy.
- the assessment is performed without assessing the presence or severity of endometriosis in the patient using laparoscopy and/ the rASRM staging.
- endometriosis is early endometriosis, in particular stage I endometriosis according to rASRM staging or stage II endometriosis according to rASRM staging.
- the protein level of EphA1 is determined, optionally using a process selected from: ELISA assay, immunoblotting, lateral flow assay, protein microarray and mass spectrometry.
- the level of EphA1 is determined by detecting the extracellular protein part of EphA1 by ELISA or by ECLIA.
- the extracellular protein part of EphA1 is detected by identification of at least one of the following peptides: WEPPADTGGR (SEQ ID NO: 1), AQGELGWLLDPPK (SEQ ID NO: 2), MHCSPDGEWLVPVGR (SEQ ID NO: 3), GLYLAFHNPGACVALVSVR (SEQ ID NO: 4), APGEGPQVACTGPPSAPR (SEQ ID NO: 5), VTTVAADQSFTIR (SEQ ID NO: 6), or VHVELQFTVR (SEQ ID NO: 7). Details are shown in Table 1.
- Table 1 Peptides identified of the EphA1 extracellular protein part detected using advanced mass spectrometry proteomics technology in serum samples of women with endometriosis (cases) and women without endometriosis (controls).
- the amount of EphA1 is determined using antibodies, in particular using monoclonal antibodies.
- step a) of determining the amount of EphA1 in a sample of the patient comprises performing an immunoassay.
- the immunoassay is performed either in a direct or indirect format.
- such immunoassays are selected from the group consisting of enzyme linked immunosorbent assay (ELISA), enzyme immunoassay (EIA), radioimmunoassay (RIA), or immuno assays based on detection of luminescence, fluorescence, chemiluminescence or electrochemiluminescence.
- step a) of determining the level of EphA1 in a sample of the subject comprises the steps of i) incubating the sample of the subject with one or more antibodies specifically binding to EphA1 , thereby generating a complex between the antibody and EphA1 , and ii) quantifying the complex formed in step i), thereby quantifying the amount of EphA1 in the sample of the subject.
- step i) the sample is incubated with two antibodies, specifically binding to EphA1 .
- the sample can be contacted with the first and the second antibody in any desired order, i.e., first antibody first and then the second antibody or second antibody first and then the first antibody, or simultaneously, for a time and under conditions sufficient to form a first anti- EphA1 antibody/ EphA1 /second anti- EphA1 antibody complex.
- the detection of the anti- EphA1 antibody I EphA1 complex can be performed by any appropriate means.
- the detection of the first anti- EphA1 antibody/ EphA1 /second anti- EphA1 antibody complex can be performed by any appropriate means.
- the person skilled in the art is absolutely familiar with such means/methods.
- a sandwich will be formed comprising a first antibody to EphA1 , EphA1 (analyte) and the second antibody to EphA1 , wherein the second antibody is detectably labeled.
- a sandwich will be formed comprising a first antibody to EphA1 , the EphA1 (analyte) and the second antibody to EphA1 , wherein the second antibody is detectably labeled and wherein the first anti- EphA1 antibody is capable of binding to a solid phase or is bound to a solid phase.
- the second antibody is directly or indirectly detectably labeled.
- the second antibody is detectably labeled with a luminescent dye, in particular a chemiluminescent dye or an electrochemiluminescent dye.
- the endometriosis assessed is selected from the group consisting of peritoneal endometriosis, endometrioma, and deep infiltrating endometriosis (DIE).
- DIE deep infiltrating endometriosis
- the endometriosis diagnosed is peritoneal endometriosis. In other particular embodiments the endometriosis diagnosed is peritoneal endometriosis of stage I or stage II according to rASRM staging.
- the methods further comprise selecting a treatment regimen for the subject based on the comparison of the level of EphA1 with the control sample or with the predetermined reference level.
- the method further comprises administering the selected treatment regimen to the subject, optionally wherein the selected treatment regimen comprises drug-based therapy and/or surgical treatment (laparoscopy).
- Drug-based therapy of endometriosis can for example be by pain medication hormone treatments, and/or surgery.
- the method further comprising the assessment of dysmenorrhea and/or lower abdominal pain in the patient.
- the presence of dysmenorrhea and/or lower abdominal pain is assessed according to the VAS scale.
- dysmenorrhea VAS score of 4 or higher indicated moderate or severe dysmenorrhea.
- scores of 3 or less indicate no or mild dysmenorrhea.
- the method further comprising determining the level of CA-125 in the biological fluid sample from the subject.
- the method comprises calculating • a ratio of the amount or concentration the extracellular protein part of EphA1 and the amount or concentration of CA-125, or
- an amount of EphA1 lowered by 15% or more, 20% or more, 30% or more, 40% or more, 50% or more is indicative of the presence or the risk of developing of uterine/pelvic pathology.
- an amount of EphA1 lowered by 100% or more is indicative of the presence of uterine/pelvic pathology.
- an amount of EphA1 lowered by 150% or more is indicative of the presence of uterine/pelvic pathology.
- an amount of EphA1 lowered by 200% or more is indicative of uterine/pelvic pathology.
- an amount of EphA1 lowered by 10% or more is indicative of the presence or risk of having or developing uterine/pelvic pathology.
- the biological fluid sample is blood, serum, plasma, capillary blood, interstitial fluid, peritoneal fluid, or menstrual fluid, preferably the biological fluid sample is serum.
- the biological fluid sample is serum.
- the protein level of EphA1 is determined, optionally using a process selected from: ELISA assay, immunoblotting, lateral flow assay, protein microarray and mass spectrometry.
- the level of EphA1 is determined by detecting the extracellular protein part of EphA1 by ELISA or by ECLIA.
- the extracellular protein part of EphA1 is detected by identification of at least one of the following peptides: WEPPADTGGR (SEQ ID NO: 1), AQGELGWLLDPPK (SEQ ID NO: 2), MHCSPDGEWLVPVGR (SEQ ID NO: 3), GLYLAFHNPGACVALVSVR (SEQ ID NO: 4), APGEGPQVACTGPPSAPR (SEQ ID NO: 5), VTTVAADQSFTIR (SEQ ID NO: 6), or VHVELQFTVR (SEQ ID NO: 7). Details are shown in Table 1.
- the methods further comprise selecting a treatment regimen for the subject based on the comparison of the level of EphA1 with the control sample or with the predetermined reference level.
- the method further comprises administering the selected treatment regimen to the subject, optionally wherein the selected treatment regimen comprises drug-based therapy and/or surgical treatment (laparoscopy).
- the invention relates to a method for assessing endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject, the method comprising the steps of: a) determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject, b) comparing the level of the extracellular protein part of EphA1 to at least one appropriate reference value of a level of the extracellular protein part of EphA1 , c) identifying a subject as having endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain if the comparison in step b) indicates that the subject has a decreased level of the extracellular protein part of EphA1 compared to the appropriate reference value, wherein the subject’s stage of endometriosis is classified as stage I or stage II endo
- the method is an in vitro method.
- the at least one appropriate reference value is i. a level of EphA1 in a SNF subject or in a SOF subject, or is ii. an average level of EphA1 in a group of SNF subjects or in a group of SOF subjects, or a combination thereof, or wherein the at least one appropriate reference value is iii. a predetermined value of a level of EphA1 in a SNF subject or SOF subject, or is iv. a predetermined average value of a level of EphA1 in a group of SNF subjects or in a group of SOF subjects, or a combination thereof.
- an amount of EphA1 lowered by 10% or more is indicative of the presence or risk of having or developing endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain.
- the biological fluid sample is blood, serum, plasma, capillary blood, interstitial fluid, peritoneal fluid, or menstrual fluid, preferably the biological fluid sample is serum.
- the biological fluid sample is serum.
- the protein level of EphA1 is determined, optionally using a process selected from: ELISA assay, immunoblotting, lateral flow assay, protein microarray and mass spectrometry.
- the level of EphA1 is determined by detecting the extracellular protein part of EphA1 by ELISA or by ECLIA.
- the extracellular protein part of EphA1 is detected by identification of at least one of the following peptides: WEPPADTGGR (SEQ ID NO: 1), AQGELGWLLDPPK (SEQ ID NO: 2), MHCSPDGEWLVPVGR (SEQ ID NO: 3), GLYLAFHNPGACVALVSVR (SEQ ID NO: 4), APGEGPQVACTGPPSAPR (SEQ ID NO: 5), VTTVAADQSFTIR (SEQ ID NO: 6), or VHVELQFTVR (SEQ ID NO: 7). Details are shown in Table 1.
- the methods further comprise selecting a treatment regimen for the subject based on the comparison of the level of EphA1 with the control sample or with the pre- determined reference level.
- the method further comprises administering the selected treatment regimen to the subject, optionally wherein the selected treatment regimen comprises drug-based therapy and/or surgical treatment (laparoscopy).
- the present invention relates to a method for monitoring endometriosis progression, uterine/pelvic pathology progression and/or progression of endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject, the method comprising the steps of: i. determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject in accordance with method steps a) to b) described above herein, ii. repeating step i. using a biological fluid sample obtained from the subject during or after treatment for a time interval; and iii. comparing the levels of the extracellular protein part of EphA1 identified in i.
- a change in the levels of extracellular protein part of EphA1 from i. to ii. is indicative of a change in endometriosis progression, uterine/pelvic pathology progression and/or progression of endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in the subject, wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
- a patient suffering from endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain is monitored to determine if the amount or concentration of EphA1 is changing over time in a sample of the patient.
- a patient suffering from endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain is monitored to determine if the amount or concentration of EphA1 is increasing, decreasing or not changing over time.
- a patient suffering from endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain is monitored if a decreased amount of EphA1 in the sample of the patient is determined.
- the method is an in vitro method.
- the biological fluid sample is serum.
- the method may be used to monitor the progression of any kind of endometriosis described herein.
- the method may be used to monitor the progression of any kind of uterine/pelvic pathology described herein.
- monitoring methods are performed on subjects that have not yet been treated for endometriosis (i.e. , they have not previously received endometriosis treatment (therapy or surgery)), for uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain.
- endometriosis i.e. , they have not previously received endometriosis treatment (therapy or surgery)
- uterine/pelvic pathology or endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain.
- Such subjects are described as “naive” subjects herein.
- therapy of endometriosis is selected from the group consisting of drug-based therapy or surgical therapy.
- the treatment regimen comprises surgical therapy, radiotherapy, immunotherapy, hormone therapy, ultrasound therapy, or combinations thereof.
- surgical therapy of endometriosis is laparoscopy or nerve sparing surgery.
- drug-based therapy of endometriosis is inhibiting or targeting neurogenic inflammation and/or pain medication and/or hormonal therapy.
- the term “computer-implemented” as used herein means that the method is carried out in an automated fashion on a data processing unit which is, typically, comprised in a computer or similar data processing device.
- the data processing unit shall receive values for the amount of the biomarkers. Such values can be the amounts, relative amounts or any other calculated value reflecting the amount as described elsewhere herein in detail. Accordingly, it is to be understood that the aforementioned method does not require the determination of amounts for the biomarkers but rather uses values for already predetermined amounts.
- the present invention also, in principle, contemplates a computer program, computer program product or computer readable storage medium having tangibly embedded said computer program, wherein the computer program comprises instructions which, when run on a data processing device or computer, carry out the method of the present invention as specified above.
- the present disclosure further encompasses: a computer or computer network comprising at least one processor, wherein the processor is adapted to perform the method according to one of the embodiments described in this description, a computer loadable data structure that is adapted to perform the method according to one of the embodiments described in this description while the data structure is being executed on a computer, a computer script, wherein the computer program is adapted to perform the method according to one of the embodiments described in this description while the program is being executed on a computer, a computer program comprising program means for performing the method according to one of the embodiments described in this description while the computer program is being executed on a computer or on a computer network, a computer program comprising program means according to the preceding embodiment, wherein the program means are stored on a storage medium readable to a computer, a storage medium, wherein a data structure is stored on the storage medium and wherein the data structure is adapted to perform the method according to one of the embodiments described in this description after having been loaded into a main and/or working storage of
- the methods according to the present invention can be combined with other tests, biomarkers, clinical data or further information that is useful to diagnose or classify endometriosis in order to obtain the most reliable result.
- CA-125 is routinely used as a biomarker for endometriosis. Accordingly, it may be advantageous to combine determining the level of EphA1 with CA-125 obtained from the subject in the context of the methods described herein.
- symptoms or clinical data that is used for diagnosing or classifying endometriosis can be used in combination with the determination of EphA1 levels.
- symptoms or clinical data can be but are not limited to age, dysmenorrhea, abdominal pain, or other biomarkers.
- kits for diagnosing or classifying the stage of endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject.
- the kits include reagents suitable for determining levels of a plurality of analytes in a test sample (e.g., reagents suitable for determining levels of the biomarker disclosed herein).
- the kits described herein typically comprise a detectably labelled agent that specifically binds to EphA1 protein.
- kit may additionally comprise a detectably labelled agent that specifically binds to CA- 125.
- kits described herein can take on a variety of forms.
- the kits will include reagents suitable for determining levels of a plurality of biomarkers (for example EphA1 and optionally CA-125) in a sample.
- a plurality of biomarkers for example EphA1 and optionally CA-125
- kits may contain one or more control samples or references.
- a comparison between the levels of the biomarkers in the subject and levels of the biomarkers in the control samples is indicative of a clinical status (e.g., diagnosis of endometriosis).
- the kits in some cases, will include written information (indicia) providing a reference (e.g., pre-determined values), wherein a comparison between the levels of the biomarker in the subject and the reference (pre- determined values) is indicative of a clinical status (e.g., diagnosis of endometriosis).
- the kits comprise software useful for comparing biomarker levels or occurrences with a reference (e.g., a prediction model).
- the software will be provided in a computer readable format such as a compact disc, but it also may be available for downloading via the internet.
- the kits are not so limited and other variations with will be apparent to one of ordinary skill in the art.
- the components of the kit may be housed in a container that is suitable for transportation. Details on the biomarker is given above and apply equally here.
- the biomarker may be protein.
- kits include the detectably labelled agent(s) on a continuous (e.g., solid) surface, such as a lateral flow surface.
- the detectably labelled agent(s) may be located in distinct (i.e., spatially separate) zones on a (e.g., solid) surface, such as a multiwall micro-titre plate (e.g., for an ELISA assay).
- a multiwall micro-titre plate e.g., for an ELISA assay
- the kit further comprises one or more reagents for detecting the detectably labelled agent.
- Suitable reagents are well known in the art and include but are not limited to standard reagents and buffers required to perform any one of the appropriate detection methods that may be used (and are well known in the art).
- the kit comprises one or more of the following: a multi-well plate, ball bearing(s), extraction buffer, extraction bottle and a lateral flow device lateral flow device.
- An assay device is also provided for diagnosing endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject.
- the device comprises a surface with at least one detectably labelled agent located thereon that specifically binds to EphA1 protein.
- Such a device may additionally comprise a detectably labelled agent that specifically binds to CA-125.
- two detectably labelled agents may be located in separate zones on the surface.
- the two detectably labelled agents may be located in distinct (i.e. , spatially separate) zones on a (e.g., solid) surface, such as a multiwell micro-titre plate.
- Detectably labelled agent(s) that specifically bind to the biomarker(s) of interest are described in detail elsewhere herein.
- the assay device comprises a surface upon which the detectably labelled agents are located.
- Appropriate surfaces include a continuous (e.g., solid) surface, such as a lateral flow surface, a dot blot surface, a dipstick surface or a surface suitable for performing surface plasmon resonance.
- Other appropriate surfaces include microtitre plates, multi-well plates etc. Other appropriate surfaces that are well known in the art may also form part of the assay device described herein.
- Appropriate assay device formats therefore include but are not limited to device formats suitable for performing any one of lateral flow, dot blot, ELISA, or surface plasmon resonance assays for detecting the presence, level or absence of the biomarker of interest.
- Biomarker levels and/or reference levels may be stored in a suitable data storage medium (e.g., a database) and are, thus, also available for future diagnoses. This also allows efficiently diagnosing prevalence for a disease because suitable reference results can be identified in the database once it has been confirmed (in the future) that the subject from which the corresponding reference sample was obtained did have endometriosis.
- a “database” comprises data collected (e.g., analyte and/or reference level information and /or patient information) on a suitable storage medium.
- the database may further comprise a database management system.
- the database management system is, preferably, a network-based, hierarchical or object-oriented database management system.
- the database may be a federal or integrated database. More preferably, the database will be implemented as a distributed (federal) system, e.g., as a Client-Server-System. More preferably, the database is structured as to allow a search algorithm to compare a test data set with the data sets comprised by the data collection. Specifically, by using such an algorithm, the database can be searched for similar or identical data sets being indicative of endometriosis (e.g., a query search). Thus, if an identical or similar data set can be identified in the data collection, the test data set will be associated with endometriosis. Consequently, the information obtained from the data collection can be used to diagnose endometriosis or based on a test data set obtained from a subject. More preferably, the data collection comprises characteristic values of all analytes comprised by any one of the groups recited above.
- the methods described herein may further include communication of the results or diagnoses (or both) to technicians, physicians or patients, for example.
- computers will be used to communicate results or diagnoses (or both) to interested parties, e.g., physicians and their patients.
- the results or diagnoses (or both) are communicated to the subject as soon as possible after the diagnosis is obtained.
- the results or diagnoses (or both) may be communicated to the subject by the subject's treating physician.
- the results or diagnoses (or both) may be sent to a subject by email or communicated to the subject by phone.
- a computer may be used to communicate the results or diagnoses by email or phone.
- the message containing results or diagnoses may be generated and delivered automatically to the subject using a combination of computer hardware and software which will be familiar to artisans skilled in telecommunications. Uses
- biomarker EphA1 as a biological fluid biomarker for endometriosis, in particular the early stages, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
- EphA1 may be used as biomarker for endometriosis generally.
- endometriosis generally refers to all forms of endometriosis, including but not limited to peritoneal endometriosis, endometrioma, and deep infiltrating endometriosis.
- EphA1 may also be combined with CA-125.
- EphA1 may be used as biomarker for uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
- kits, assay devices and uses provided herein may be used as part of a companion diagnostic e.g., as part of a medical device, often an in vitro device, which provides information that is essential for the safe and effective use of a corresponding drug or biological product (wherein the corresponding drug or biological product is for treating or preventing endometriosis).
- the present invention relates to the following aspects, wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM):
- An in vitro method for diagnosing endometriosis in a subject comprising the steps of: a) determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject, b) comparing the level of the extracellular protein part of EphA1 to at least one appropriate reference value of a level of the extracellular protein part of EphA1, c) identifying a subject as having endometriosis if the comparison in step b) indicates that the subject has a decreased level of the extracellular protein part of EphA1 compared to the appropriate reference value.
- the at least one appropriate reference value is i. a level of EphA1 in a non-pathological subject or in a symptomatic subject, or is ii. an average level of EphA1 in a group of non-pathological subjects or in a group of symptomatic subjects, or a combination thereof, or wherein the at least one appropriate reference value is iii. a predetermined value of a level of EphA1 in a non-pathological subject or symptomatic subject, or is iv. a predetermined average value of a level of EphA1 in a group of non- pathological subjects or in a group of symptomatic subjects, or a combination thereof.
- the method according to aspect 1 or 2 wherein the at least one appropriate reference value is
- r-ASRM a level of EphA1 in a non-pathological subject, in a symptomatic subject, or in a subject that has stage I, stage II, stage III or stage IV endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM), or is
- r-ASRM a predetermined value of a level of EphA1 in a non-pathological subject or in a symptomatic subject, or in a subject that has stage I, stage II, stage III or stage IV endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM), or is
- the biological fluid sample is blood, serum, plasma, capillary blood, interstitial fluid, peritoneal fluid, or menstrual fluid, preferably the biological fluid sample is serum.
- the subject is a human, preferably a female human.
- the protein level of EphA1 in particular its extracellular protein part, is determined, optionally using a process selected from: ELISA assay, immunoblotting, lateral flow assay, protein microarray and mass spectrometry.
- a process selected from: ELISA assay, immunoblotting, lateral flow assay, protein microarray and mass spectrometry optionally using a process selected from: ELISA assay, immunoblotting, lateral flow assay, protein microarray and mass spectrometry.
- the extracellular protein part of EphA1 is detected by identification of at least one of the following peptides: WEPPADTGGR (SEQ ID NO: 1), AQGELGWLLDPPK (SEQ ID NO: 2), MHCSPDGEWLVPVGR (SEQ ID NO: 3), GLYLAFHNPGACVALVSVR (SEQ ID NO: 4), APGEGPQVACTGPPSAPR (SEQ ID NO: 5), VTTVAADQSFTIR (SEQ ID NO: 6), or VHVELQFTVR (SEQ ID NO: 7).
- WEPPADTGGR SEQ ID NO: 1
- AQGELGWLLDPPK SEQ ID NO: 2
- MHCSPDGEWLVPVGR SEQ ID NO: 3
- GLYLAFHNPGACVALVSVR SEQ ID NO: 4
- APGEGPQVACTGPPSAPR SEQ ID NO: 5
- VTTVAADQSFTIR SEQ ID NO: 6
- VHVELQFTVR SEQ ID NO
- endometriosis is selected from the group consisting of peritoneal endometriosis, endometrioma, and deep infiltrating endometriosis.
- Serum EphA1 can be used for therapy selection and monitoring as a potential companion diagnostics biomarker.
- Example 2 Diagnostic performance of biomarker EphA1 compared to CA-125 in women with endometriosis
- the concentration of CA-125 was determined by a cobas e 601 analyzer. Detection of CA 125 II with a cobas e 601 analyzer is based on the Elecsys® Electro- ChemiLuminescence (ECL) technology.
- ECL Electro- ChemiLuminescence
- biotin-labelled and ruthenium-labelled antibodies are combined with the respective amount of undiluted sample and incubated on the analyzer.
- streptavidin-coated magnetic microparticles are added and incubated on the instrument in order to facilitate binding of the biotin-labelled immunological complexes. After this incubation step the reaction mixture is transferred into the measuring cell where the beads are magnetically captured on the surface of an electrode.
- ProCell M Buffer containing tripropylamine (TPA) for the subsequent ECL reaction is then introduced into the measuring cell in order to separate bound immunoassay complexes from the free remaining particles.
- Induction of voltage between the working and the counter electrode then initiates the reaction leading to emission of photons by the ruthenium complexes as well as TPA.
- the resulting electrochemiluminescent signal is recorded by a photomultiplier and converted into numeric values indicating concentration level of the respective analyte.
- Figs 8a and b and Figs 9a and b show CA-125 in serum samples of women with early endometriosis rASRM stage I (Fig. 8) and II (Fig. 9) “Case” compared to controls without endometriosis and without uterine and pelvic pathology “Ctrl”.
- Box plots in Figs. 8a and b and 9a and b for CA-125 were generated for controls and for the endometriosis stages I and II using a cobas e 601 analyzer based on Elecsys® ElectroChemiLuminescence (ECL) technology.).
- the data are presented using box and whisker plots, including the median (middle quartile), the inter-quartile range (which represents the middle 50% of scores for the group), the upper quartile (75% of scores fall below the upper quartile), the lower quartile (25% of scores fall below the lower quartile).
- the whiskers show the 5th percentile and the 95th percentile, respectively.
- serum EphA1 shows better diagnosis performance for the detection of early stages of endometriosis (Stage I, Stage II) compared to the reference biomarker CA-125.
- Serum EphA1 and serum CA-125 combined can be used as a blood biomarker for (early) diagnosis and risk stratification of endometriosis or other uterine/pelvic pathologies, in particular early diagnosis of rASRM stages 1 and 2 endometriosis.
- Serum EphA1 and serum CA-125 combined showed better identification of all cases (endometriosis and other uterine/pelvic pathologies) than the CA-125 reference biomarker alone.
- Serum EphA1 and serum CA-125 combined can be used to select patients for early medical management (drug therapy or surgery) of endometriosis. Therefore, it can significantly reduce endometriosis diagnostic delay, improve patients' lives, and reduce the economic burden.
- Serum EphA1 and serum CA-125 can be used for therapy selection and monitoring as a potential companion diagnostics biomarker.
- Samples were digested with Trypsin/Endoproteinase LysC to generate proteolytic peptides, which were subsequently subjected to high resolution liquid chromatography coupled to tandem mass spectrometry (LC-MS/MS) applying a data-independent acquisition method (DIA).
- LC-MS/MS tandem mass spectrometry
- DIA data-independent acquisition method
- PCA principal component analysis
- Serum samples were shipped by Roche on dry ice and stored at -80°C. For experimental processing, samples were thawed on ice and centrifuged with 2,000 x g for 15 min at 4°C. For documentation, a hemolysis score was determined before the supernatants were transferred into new tubes. 7 pl serum per sample were subjected to depletion of 14 high-abundant proteins using High Select Top14 Abundant Protein Depletion Mini Spin Columns (Thermo Scientific) according to the manufacturer's instructions. The depleted, lyophilized plasma protein samples were subjected to enzymatic cleavage and peptide clean-up using an MS sample preparation kit according to the manufacturer's instructions. The workflow was performed in 96 well plates using the Agilent BRAVO automated liquid handling platform. Serum control samples were used for each sample preparation batch and were treated equally.
- LC-MS/MS analysis was performed on an Orbitrap Exploris 480 mass spectrometer (Thermo Scientific) equipped with an Easy n-LC 1200 LIHPLC system (Thermo Scientific). Peptides were separated by a 125 min gradient before they were sprayed directly into the mass spectrometer using a nano-electrospray ion source (ProxeonBiosystems). The mass spectrometer was operated in the data independent mode (DIA) for single-shot analysis.
- DIA data independent mode
- the EphA1 protein is composed of an Extracellular domain, a Transmembrane domain and an Intracellular domain.
- the Extracellular domain contains the Ephrin-binding domain or Ephrin Receptor Ligand-Binding Domain (LBD), the Cysteine-rich region (including Sushi domain and EGF-like domain), and the Fibronectin-type III repeats (FN1 and FN2).
- the T ransmembrane domain (TM) links the Extracellular domain and the Intracellular domain.
- the Intracellular domain is composed of the Juxtamembrane domain, the Kinase domain, and the SAM domain.
- Figure 10 shows the structure of the EphA1 protein ( Figure adapted from Owens HE. Corrected Thesis. Characterisation of EphA1 and its potential role in Alzheimer’s Disease. 2019)
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Hematology (AREA)
- Chemical & Material Sciences (AREA)
- Urology & Nephrology (AREA)
- Molecular Biology (AREA)
- Biomedical Technology (AREA)
- Immunology (AREA)
- Microbiology (AREA)
- Food Science & Technology (AREA)
- Biotechnology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Reproductive Health (AREA)
- Pregnancy & Childbirth (AREA)
- Gynecology & Obstetrics (AREA)
- Cell Biology (AREA)
- Medicinal Chemistry (AREA)
- Physics & Mathematics (AREA)
- Analytical Chemistry (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- General Physics & Mathematics (AREA)
- Pathology (AREA)
- Investigating Or Analysing Biological Materials (AREA)
Abstract
The present invention relates to methods of diagnosing whether a subject has endometriosis, uterine/pelvic pathology and/or endometriosis and/or uterine/pelvic pathology associated neuropathic pain, to methods of determining the therapeutic effect of a treatment regimen for endometriosis, uterine/pelvic pathology and/or endometriosis and/or uterine/pelvic pathology associated neuropathic pain, and methods of monitoring endometriosis, uterine/pelvic pathology and/or endometriosis and/or uterine/pelvic pathology associated neuropathic pain progression in a subject, by determining the amount or concentration of EphA1 in a sample of the subject, and comparing the determined level to a reference value.
Description
Serum EphA1 as biomarker for endometriosis
The present invention relates to methods of diagnosing whether a subject has endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain, methods of assessing the risk of a subject having endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain, to methods of determining the therapeutic effect of a treatment regimen for endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain, and methods of monitoring endometriosis progression, uterine/pelvic pathology progression and/or endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain progression in a subject, by determining the amount or concentration of EphA1 in a sample of the subject, and comparing the determined level to a reference value. The present invention in particular refers to the early stages of endometriosis.
Backaround of the Invention
Endometriosis is a chronic disorder defined by the growth of endometrial glands and stromalike lesions outside the uterus (Liu et al., 2011). The lesions can be peritoneal lesions, superficial implants or cysts on the ovary, or deep infiltrating disease. It arises from eutopic endometrial cells characterized by increased proliferation and adhesion properties (Liu et al., 2011). The increased cell viability in eutopic endometrium is a consequence of reduced apoptosis and an increase in cell proliferation (Johnson et al., 2005). Endometriosis affects 5- 8% of all women of reproductive age and 70% of women with chronic pelvic pain. The prevalence of endometriosis has been estimated at 176 million women worldwide (Adamson et al. J Endometr. 2010; 2: 3-6). For many of these women there is often a delay in diagnosis of endometriosis resulting in unnecessary suffering and reduced quality of life. In patients aged 18-45 years, there is a diagnostic delay of endometriosis of 7-10 years. As most women with endometriosis report the onset of symptoms during adolescence, early referral, diagnosis, identification of disease and treatment may mitigate pain while also potentially preventing disease progression. Barriers to early diagnosis include the interventional nature of current diagnostic techniques such as laparoscopy, the high cost of diagnosis and treatment in
adolescent patients and presentation of confounding symptoms such as cyclic and non-cyclic pain (Parasar et al. Curr Obstet Gynecol Rep. 2017; 6: 34-41).
Gold standard for the diagnosis of endometriosis is laparoscopic visualization and subsequent histological confirmation. Until now, there are no universal non-invasive methods for the diagnosis of endometriosis, in particular for early, minimal and mild endometriosis (revised American Society for Reproductive Medicine rASRM stages l-ll; Hsu et al. Clin Obstet Gynecol 2010: 53: 413-419). During a diagnostic laparoscopy, a gynaecologist with training and skills in laparoscopic surgery for endometriosis should perform a systematic inspection of the pelvis (NICE guideline NG73, 2017, ESHRE Guideline Endometriosis Human Reproduction Open, 2022). Surgical visualization requires good expertise, training and skills for reliable diagnosis. The fact that laparoscopic surgery is needed for diagnosis, which is avoided by doctors as long as possible, is one main reason leading to a 7-10 year delay in diagnosis. The lack of a non-invasive diagnostic test significantly contributes to the long delay between onset of the symptoms and definitive diagnosis of endometriosis (Signorile and Baldi. J Cell Physiol 2014; 229: 1731-1735). Thus, there is an unmet medical need for a non-invasive test for the diagnosis of endometriosis, in particular for the diagnosis of early, minimal and mild endometriosis (revised American Society for Reproductive Medicine rASRM stages l-ll).
Non-invasive diagnosis of endometriosis would allow earlier diagnosis and treatment, with the potential to improve quality of life and reduce the societal costs related to endometriosis, and has therefore been selected as a research priority by the World Endometriosis Society (WES) and the World Endometriosis Research Foundation (WERF) (Fassbender et al., Springer, Peripheral Blood Biomarkers for Endometriosis. 2017). Thus, a non-invasive tool to diagnose endometriosis could facilitate earlier diagnosis and intervention that could ultimately improve quality of life and preserve fertility (Parasar et al. Curr Obstet Gynecol Rep. 2017; 6: 34-41).
Blood based biomarkers are essential to help reduce the time delay of diagnosing endometriosis that require laparoscopy. CA-125 is one of the most commonly used blood biomarkers, however, its diagnostic utility is limited to endometriosis rASRM stages III and IV (Nisenblat et al., Cochrane Database of Systematic Reviews. 2016;5: CD012179). As such the use of CA-125 is not recommended in clinical guidelines for the diagnosis of endometriosis (ESHRE Guideline Endometriosis Human Reproduction Open, 2022).
Same applies to similar disorders like uterine and pelvic pathology as well as endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
Endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain is a component of endometriosis-associated pain (Coxon et al., /s there a Neuropathic-Like Component to Endometriosis-Associated Pain? Resuits From a Large Cohort Questionnaire Study, Front Pain Res (Lausanne), 2021 ; 2: 743812; doi: 10.3389/fpain.2021.743812).
The Ephrin (Eph) receptor family comprises the largest family in receptor tyrosine kinases and can be divided into two groups based on their structures and receptor-ligand specificity. EphA consists of nine type-A Eph receptors (EphA1-8, and EphAIO) and five type-B Eph receptors (EphB1-4, 6). The Eph receptors bind to the ephrin ligands which are also divided into ephrin- A (ephrin-A1-6) and ephrin-B (ephrin- B1-3) based on their structures. Binding of ephrins to the Eph receptors activates signaling cascades that regulate several biological processes such as cellular proliferation, differentiation, migration, angiogenesis, and vascular remodeling. There is evidence implicating ephrins and the Eph receptors in the modulation of folliculogenesis, ovulation, embryo transport, implantation, and placentation (Adu-Gyamfi et al. Biology of Reproduction, 2021).
Specifically Ephrin type-A receptor 1 (EphA1) is a receptor tyrosine kinase which binds to the ephrin-A family ligands residing on adjacent cells. Both EphA1 and Ephrin-A are membrane bound with their binding occurring by direct cell-cell interaction leading to contact-dependent bidirectional signaling into neighboring cells. Forward signaling occurs in Eph receptorexpressing cells whereas reverse signaling occurs in ephrin-expressing cells.
Deregulated activation of the EphA family members was found in various human cancers, such as lung cancer, gastric cancer, hepatocellular carcinoma, esophageal squamous cell carcinoma, and prostate cancer. Evidence indicates that EphA receptors are involved in regulating tumor growth, invasiveness, angiogenesis, and metastasis by altering cell proliferation, motility, invasion, and migration (Zhang X. Front Oncol. 2021 ;11 :619949).
Fujii et al. (Hum Reprod 2011 ;26:299-306) has shown by RT-PCR that mRNA of EPHA1 was expressed in endometrial epithelial cell fractions in the proliferative and secretory phases of
the menstrual cycle. EphA1 protein was expressed on the endometrial luminal surface and glandular epithelial cells demonstrated by immunohistochemistry.
The levels/presence of a biomarker can differ when measured in the tissue or in blood serum. For example, the complement component C7 and complement component C4 showed overexpression in ectopic endometrium of women with endometriosis compared to eutopic endometrium of control women without endometriosis (Ahn et al. Fertil Steril 2016; Eyster et al. Fertil Steril 2007). However, no increase of serum complement component C7 protein nor complement component C4 protein was found in circulating blood (Hever et al. PNAS 2007). Brain-derived neurotrophic factor (BDNF) mRNA expression levels were higher in ovarian endometriotic lesions than in eutopic endometrium (Wang et al. Journal of Ovarian Research 2022). However, serum BDNF was not significantly different in women with endometriosis compared to control women without endometriosis (Perricos et al. Exp Biol Med (Maywood) 2018). Furthermore, in breast cancer, while it has observed that molecular markers such as CEA (O), ERp, CK19 and, c-Myc were significantly different between blood of normal and patients while there were no significant differences of these markers in tissue samples.
Therefore, the locally altered expression of biomarkers in tissue (mRNA or protein expression) does not translate 1 :1 to significantly different levels of these biomarkers in circulating blood.
There is a high need for a non-invasive diagnosis of endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain by using a biomarker, which allows a reliable and early risk assessment and/or identification of women showing signs and symptoms of endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
The present invention, therefore, provides means and methods complying with these needs.
Summary of the Invention
In a first aspect, the present invention relates to a method for assessing endometriosis in a subject, the method comprising the steps of: a) determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject,
b) comparing the level of the extracellular protein part of EphA1 to at least one appropriate reference value of a level of the extracellular protein part of EphA1, c) identifying a subject as having endometriosis if the comparison in step b) indicates that the subject has a decreased level of the extracellular protein part of EphA1 compared to the appropriate reference value, wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
In a second aspect the present invention relates to a method for assessing uterine/pelvic pathology in a subject, the method comprising the steps of: a) determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject, b) comparing the level of the extracellular protein part of EphA1 to at least one appropriate reference value of a level of the extracellular protein part of EphA1, c) identifying a subject as having uterine/pelvic pathology if the comparison in step b) indicates that the subject has a decreased level of the extracellular protein part of EphA1 compared to the appropriate reference value,
In a third aspect the invention relates to a method for assessing endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject, the method comprising the steps of: a) determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject, b) comparing the level of the extracellular protein part of EphA1 to at least one appropriate reference value of a level of the extracellular protein part of EphA1, c) identifying a subject as having endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain if the comparison in step b) indicates that the subject has a decreased level of the extracellular protein part of EphA1 compared to the appropriate reference value,
wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
In a fourth aspect the present invention relates to a method for monitoring endometriosis progression, uterine/pelvic pathology progression and/or progression of endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject, the method comprising the steps of: i. determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject in accordance with method steps a) to b) of the first aspect, ii. repeating step i. using a biological fluid sample obtained from the subject during or after treatment for a time interval; and iii. comparing the levels of the extracellular protein part of EphA1 identified in i. with the levels of extracellular protein part of EphA1 identified in ii., wherein a change in the levels of extracellular protein part of EphA1 from i. to ii. is indicative of a change in endometriosis progression , uterine/pelvic pathology progression and/or progression of endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in the subject, wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
In a fifth aspect, the present invention relates to a method for determining the therapeutic effect of a treatment regimen for endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject, the method comprising the steps of:
i. determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject in accordance with method steps a) to b) of the first aspect, ii. repeating step i. using a biological fluid sample obtained from the subject during or after treatment for a time interval; and iii. comparing levels of the extracellular protein part of EphA1 identified in i. with the levels of extracellular protein part of EphA1 identified in ii., and identifying that the treatment regimen has a therapeutic effect if the level of the extracellular protein part of EphA1 increased after treatment, wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
In a sixth aspect the present invention relates to a computer-implemented method for assessing a patient with suspected endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain comprising the steps of: a) receiving a value for level of a first biomarker in a biological fluid sample of the subject, said first biomarker being the extracellular protein part of EphA1 ; b) receiving a value for the level of a second biomarker in a sample of the subject, wherein said second biomarker is CA125, c) receiving a value for the level of dysmenorrhea according to the VAS and/or lower abdominal pain according to the VAS, d) comparing the values for the levels of steps (a) - (c) to references for said biomarkers and the amount of dysmenorrhea and/or calculating a score for assessing the subject with suspected endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain based on the levels of the biomarkers and the amount of dysmenorrhea; and
e) assessing said subject based on the comparison and/or the calculation made in step (d), wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
Brief description of the Figures
Embodiments of the invention are further described hereinafter with reference to the accompanying drawings.
Box plot analysis EphA1 : EphA1 in serum samples of control groups versus endometriosis case groups (rASRM stage I, endometriosis rASRM stage II, endometriosis rASRM stage III and endometriosis rASRM stage IV). Serum EphA1 peptide levels were measured using advanced mass spectrometry based proteomics technology.
Receiver operator characteristic (ROC) analysis is shown describing the area under the curve (AUC) of the ROC analysis and the associated 95% confidence interval. N depicts the number of samples tested (cases plus controls, numbers in each group vary depending on the analyte).
Figure 1 : Boxplot analysis: Serum EphA1 levels are decreased in cases of symptomatic women with endometriosis and women without endometriosis and with other uterine and pelvic pathologies (such as uterine fibroids, adenomyosis only, ovarian cysts requiring endometriosis, cancer) (“Case SOF”; SOF=Symptomatic Other Findings) compared to controls without endometriosis and without uterine and pelvic pathologies (“Ctrl SNF”; SNF=Symptomatic No Finding). Note: Case is referring to the rASRM stage of endometriosis (Case 1= rASRM Stage I endometriosis, Case ll= rASRM Stage II endometriosis, Case lll= rASRM Stage III endometriosis, Case IV= rASRM Stage IV endometriosis).
Figures 2a and b: Boxplot analysis (a) and ROC analysis (b): EphA1 is decreased in serum samples of women with other uterine and pelvic pathologies (such as uterine fibroids, adenomyosis only, ovarian cysts requiring surgery, cancer) (“Case SOF”; SOF=Symptomatic Other Findings) compared to control women without endometriosis and without uterine and pelvic pathology (“Ctrl”).
Figures 3a and b: Boxplot analysis (a) and ROC analysis (b): EphA1 is decreased in serum samples of women with early endometriosis rASRM stages l/ll “Case” compared to control women without endometriosis and without uterine and pelvic pathology (“Ctrl”).
Figure 4a and b: Boxplot analysis (a) and ROC analysis (b): EphA1 is decreased in serum samples of women with early endometriosis rASRM stage I “Case” compared to women without endometriosis and without uterine and pelvic pathology (“Ctrl”).
Figure 5a and b: Boxplot analysis (a) and ROC analysis (b): EphA1 is decreased in serum samples of women with early endometriosis rASRM stage II “Case” compared to women without endometriosis and without uterine and pelvic pathology (“Ctrl”).
Figure 6a and b: Boxplot analysis (a) and ROC analysis (b): EphA1 is decreased in serum samples of women with endometriosis stage III “Case” compared to control women without endometriosis and without uterine and pelvic pathology (“Ctrl”).
Figure 7a and b: Boxplot analysis (a) and ROC analysis (b): EphA1 is decreased in serum samples of women with endometriosis rASRM stage IV “Case” compared to controls without endometriosis and without uterine and pelvic pathology (“Ctrl”).
Figure 8a and b: Boxplot analysis (a) and ROC analysis (b) of CA-125. Comparison to CA-125 levels measured in the same sample set as EphA1 , using the Roche Elecsys CA-125 immunoassay method. CA-125 in serum samples of women with early endometriosis rASRM stage I “Case” compared to controls without endometriosis and without uterine and pelvic pathology “Ctrl”.
Figure 9a and b: Boxplot analysis (a) and ROC analysis (b) of CA-125 in serum samples of women with early endometriosis rASRM stage II “Case” compared to controls without endometriosis and without uterine and pelvic pathology “Ctrl”.
Figure 10: Structure of EphA 1 protein. The Extracellular domain contains the
Ephrin-binding domain, the Cysteine-rich region (including Sushi domain and EGF-like domain), and the Fibronectin-type III repeats (FN1 and FN2). The Transmembrane domain (TM) links the Extracellular domain and the Intracellular domain. The Intracellular domain is composed of the Juxtamembrane domain, the Kinase domain, and the SAM domain.
Detailed Description of the Invention
An unbiased proteomics discovery was performed in serum samples of women with endometriosis or other uterine pelvic pathology and symptoms (pain symptoms such as dysmenorrhea, pelvic pain, dyspareunia, dysuria, dyschezia, other menstrual-cycle dependent pain symptoms) and women without endometriosis (controls). Details for the unbiased proteomics discovery using advanced mass spectrometry (MS)-based proteomics including automated sample preparation procedures are described in Example 4 - Materials and Methods.
The invention is based on the surprising finding that decreased EphA1 levels in serum, in particular decreased levels of the extracellular protein part of EphA1 , is associated with endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain. Using serum from women with endometriosis (cases) and controls (without endometriosis) peptides were identified of the EphA1 extracellular protein part detected using advanced mass spectrometry proteomics technology. In total, seven
peptides of EphA1 were detected using this mass spectrometry technology which all belong to the extracellular protein part of the EphA1 (Table 1). The structure of the EphA1 protein is shown in Figure 10. EphA1 levels are decreased at all rASRM stages I, II, III and IV, which confers serum EphA1 a diagnostic potential for early detection of endometriosis.
The inventors show for the first time that EphA1 measured in serum is decreased in women with endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain compared to controls.
There is an unmet medical need for a non-invasive test for the reliable diagnosis and/or classification of endometriosis, in particular early endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain. EphA1 has the advantage of a non-invasive blood-based test that identifies women with early stages of endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
The inventors have investigated the levels of EphA1 in serum obtained from women with endometriosis. Surprisingly, they found that a decreased level of EphA1 can be detected in serum samples from women with endometriosis. In particular, the fact that already in women with early stages of endometriosis decreased EphA1 levels can be detected make this marker a helpful tool to diagnose endometriosis at early stages. Assays that enable the determination of the level of EphA1 in such biological fluids may therefore be useful for endometriosis risk stratification, diagnosis, prognosis and patient stratification for treatment.
The data presented herein show that determination of EphA1 levels, in particular levels of the extracellular protein part of EphA1 , in serum provides a means for diagnosing endometriosis and risk stratification of having endometriosis. This also allows the monitoring of endometriosis progression and/or the evaluation of treatment regimen. In particular, the data show that a diagnosis of early stages of endometriosis is possible by determining EphA1 levels.
The data presented herein also show that determination of EphA1 levels in serum provides a means for detecting early stages of endometriosis and control samples more accurately than CA-125.
The inventors have investigated the levels of EphA1 in serum obtained from women with uterine/pelvic pathology. Surprisingly, they found that a decreased level of EphA1 can be detected in serum samples from women with uterine/pelvic pathology. This makes this marker a helpful tool to diagnose uterine/pelvic pathology at early stages. Assays that enable the determination of the level of EphA1 in such biological fluids may therefore be useful for end uterine/pelvic pathology risk stratification, diagnosis, prognosis and patient stratification for treatment.
The data presented herein show that determination of EphA1 levels, in particular levels of the extracellular protein part of EphA1 , in serum provides a means for diagnosing uterine/pelvic pathology and risk stratification of having uterine/pelvic pathology. This also allows the monitoring of uterine/pelvic pathology progression and/or the evaluation of treatment regimen.
Definitions
The word "comprise", and variations such as "comprises" and "comprising", will be understood to imply the inclusion of a stated integer or step or group of integers or steps but not the exclusion of any other integer or step or group of integers or steps.
As used in this specification and the appended claims, the singular forms "a", "an", and "the" include plural referents, unless the content clearly dictates otherwise.
Concentrations, levels, amounts, and other numerical data may be expressed or presented herein in a “range” format. It is to be understood that such a range format is used merely for convenience and brevity and thus should be interpreted flexibly to include not only the numerical values explicitly recited as the limits of the range, but also to include all the individual numerical values or sub-ranges encompassed within that range as if each numerical value and sub-range is explicitly recited. As an illustration, a numerical range of "150 mg to 600 mg" should be interpreted to include not only the explicitly recited values of 150 mg to 600 mg, but to also include individual values and sub-ranges within the indicated range. Thus, included in this numerical range are individual values such as 150, 160, 170, 180, 190, ... 580, 590, 600 mg and sub-ranges such as from 150 to 200, 150 to 250, 250 to 300, 350 to 600, etc. This same principle applies to ranges reciting only one numerical value. Furthermore, such an interpretation should apply regardless of the breadth of the range or the characteristics being described.
The term “about” when used in connection with a numerical value is meant to encompass numerical values within a range having a lower limit that is 5% smaller than the indicated numerical value and having an upper limit that is 5% larger than the indicated numerical value.
In general, the methods described are in vitro methods that are performed using a sample that has already been obtained from the subject (i.e. , the sample is provided for the method, and the steps taken to obtain the sample from the subject are not included as part of the method). The methods may therefore include the step of providing a biological fluid sample from a subject.
As used herein, “provide”, "obtain" or "obtaining" can be any means whereby one comes into possession of the sample by "direct" or "indirect" means. Directly obtaining a sample means performing a process (e.g., performing a physical method such as extraction) to obtain the sample. Indirectly obtaining a sample refers to receiving the sample from another party or source (e.g., a third party laboratory that directly acquired the sample).
The methods provided herein comprise providing a biological fluid sample (for example a blood sample) from a subject. The samples being tested in the methods described herein are also referred to as “test samples”.
As used herein, the terms "biological (fluid) sample", “test sample”, "sample" are used interchangeably, and variations thereof refer to a sample obtained or derived from a subject. For the purposes described herein, the sample is, or comprises, a biological fluid (also referred to herein as a bodily fluid) sample.
Examples of samples include but are not limited to fluid samples such as blood, serum, plasma, synovial fluid, interstitial fluid, capillary blood, peritoneal fluid, menstrual fluid, urine, saliva, and lymphatic fluid. Analysis of a sample may be accomplished on chemical basis. Chemical analysis includes but is not limited to the detection of the presence or absence of specific indicators or alterations in their amount, concentration or level.
The sample is an in vitro sample, it will be analyzed in vitro and not transferred back into the body.
A blood sample may be a whole blood sample, or a processed blood sample e.g., serum, plasma etc. Methods for obtaining biological fluid samples (e.g., whole blood, serum, plasma, etc) from a subject are well known in the art. For example, methods for obtaining blood samples from a subject are well known and include established techniques used in phlebotomy. The obtained blood samples may be further processed using standard techniques to obtain e.g., a serum sample, or a plasma sample. Advantageously, methods for obtaining biological fluid samples from a subject are typically low-invasive or non-invasive.
A whole blood sample is defined as a blood sample drawn from the body and from which (substantially) no constituents (such as platelets or plasma) have been removed. In other words, the relative ratio of constituents in a whole blood sample is substantially the same as a blood in the body. In this context, “substantially the same” allows for a very small change in the relative ratio of the constituents of whole blood e.g., a change of up to 5%, up to 4%, up to 3%, up to 2%, up to 1 % etc. Whole blood contains both the cell and fluid portions of blood. A whole blood sample may therefore also be defined as a blood sample with (substantially) all of its cellular components in plasma, wherein the cellular components (i.e. , at least comprising the requisite white blood cells, red blood cells, platelets of blood) are intact.
In a preferred example, the biological fluid sample is serum.
Methods for analysing (and optionally isolating, enriching for or extracting) protein biomarkers from blood, plasma, serum, saliva, and urine samples have been described previously, see for example, Heitzer, E., Haque, I.S., Roberts, C.E.S. et al. Current and future perspectives of liquid biopsies in genomics-driven oncology. Nat Rev Genet 20, 71-88 (2019).
In the context of present invention, the term “biomarker” refers to a substance within a biological system that is used as an indicator of a biological state of said system. In the art, the term „biomarker“ is sometimes also applied to means for the detection of said endogenous substances (e.g. antibodies, nucleic acid probes etc, imaging systems). In the context of present invention, the term “biomarker" shall be only applied for the substance, not for the detection means. Thus, biomarkers can be any kind of molecule present in a living organism, such as a nucleic acid (DNA, mRNA, miRNA, rRNA etc.), a protein (cell surface receptor, cytosolic protein etc.), a metabolite or hormone (blood sugar, insulin, estrogen, etc.), a molecule characteristic of a certain modification of another molecule (e.g. sugar moieties or
phosphoryl residues on proteins, methyl-residues on genomic DNA) or a substance that has been internalized by the organism or a metabolite of such a substance. A biomarker is an organic biomolecule (e.g., a protein, polypeptide, peptide, isomeric form thereof, immunologically detectable fragment thereof, corresponding nucleic acid molecule (e.g., mRNA, cDNA etc)) which is differentially present in a sample taken from a subject having a disease as compared with a subject not having the disease. A biomarker is differentially present if the mean or median level of the biomarker in the different groups is calculated to be statistically relevant. Common tests for statistical significance include, among others, t-test (e.g., student t-test), ANOVA, Kruskal-Wallis, Wilcoxon, Mann- Whitney, Receiver Operating Characteristic (ROC curve), accuracy and odds ratio. Biomarkers, alone or in combination, provide measures of relative risk that a subject belongs to one phenotypic status or another.
Therefore, they are useful as markers for disease (diagnostics), therapeutic effectiveness of a drug and drug toxicity.
Typically, the biomarker referred to herein is measured at the protein level.
The term “EphA1”, EPH receptor A1 , or ephrin type-A receptor 1 is a protein that in humans is encoded by the EPHA 1 gene. This gene belongs to the ephrin receptor subfamily of the protein-tyrosine kinase family. EPH and EPH-related receptors have been implicated in mediating developmental events, particularly in the nervous system. Receptors in the EPH subfamily typically have an extracellular part, a transmembrane part, and a cytoplasmic part. The extracellular part contains a ligand binding domain (Ephrin-binding domain), a cysteine- rich EGF-like motif, and two fibronectin type III repeats. The cytoplasmic part contains a protein kinase domain, a SAM domain and a PDZ-binding domain. The ephrin receptors are divided into two groups (EphA and EphB receptors) based on the similarity of their extracellular domain sequences and their affinities for binding ephrin-A and ephrin-B ligands (Adu-Gyamfi et al. Biology of Reproduction 2021 , Darling and Lamb, Frontiers in Immunology 2019). The amino acid sequence of human EphA1 can be accessed via UniProt (see UniProtKB - P21709 EPHA1_HUMAN). There are three isoforms described for EphA1 UniProtKB - P21709-1 , UniProtKB - P21709-2, and UniProtKB - P21709-3.
EphA1 is a receptor tyrosine kinase which binds to the ephrin-A family ligands residing on adjacent cells. Both EphA1 and Ephrin-A are membrane bound with their binding occurring by
direct cell-cell interaction leading to contact-dependent bidirectional signaling into neighboring cells. Forward signaling occurs in Eph receptor-expressing cells whereas reverse signaling occurs in ephrin-expressing cells.
Using serum from women with endometriosis (cases) and controls (without endometriosis) seven peptides were identified of the EphA1 protein detected using advanced mass spectrometry proteomics technology.. The seven identified peptides described in Table 1 are all part of the extracellular part of the EphA1 protein being part of the ligand binding domain (Ephrin binding domain) and fibronectin type III repeats. The EphA1 protein consists of an extracellular domain, a transmembrane domain, and an intracellular domain (Figure 10). The extracellular part of Eph receptors can be proteolytically cleaved and released to the circulating blood (shedding of extracellular part of Eph receptors). Soluble circulating EphA1 is discussed to be involved in immune cell trafficking.
Therefore, the determined level of the extracellular protein part of EphA1 is representative of the level of EphA1 in the sample. The expressions “EphA1 level” and “level of the extracellular protein part of EphA1” and “EphA1 extracellular protein part level” are used interchangeable herein.
The methods provided herein refer to “determining” the level of one or more proteins. As would be clear to a person of skill in the art, the level of one or more proteins is typically “determined” by measuring the level of the protein in the sample. The term “determining” can therefore be replaced with the term “measuring” or “determining by measuring” herein.
The terms “determining” or “assessing” as used herein also refer to assessing/determining whether a woman suffers from endometriosis. Accordingly, assessing/determining as used herein includes diagnosing endometriosis, assessing the risk that a subject suffers from endometriosis, selecting for therapy of endometriosis, monitoring a patient suffering from endometriosis or being treated for endometriosis, by determining the amount or concentration of EphA1 in a sample of the patient, and comparing the determined amount or concentration to a reference. Typically, the assessment referred to in accordance with the present invention is the assessment of the presence of endometriosis.
Typically, the assessment referred to in accordance with the present invention is to diagnose endometriosis, uteri ne/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain in a subject.
Typically, the assessment referred to in accordance with the present invention is to stratify the risk of a subject to suffer from endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
The term "measurement", "measuring" or "determining" preferably comprises a qualitative, a semi-quantitative or a quantitative measurement.
Conventional "determining" methods may include sending a clinical sample(s) to a commercial laboratory for measurement of the biomarker levels in the biological fluid sample, or the use of commercially available assay kits for measuring the biomarker levels in the biological fluid sample. Exemplary kits and suppliers will be apparent to a person of skill in the art. In various examples, biomarkers may be determined, detected and/or quantified using ELISA assays or lateral flow devices, such as for point-of-care use, as well as spot check colorimetric tests.
The terms “level” or "amount" as used herein encompass the absolute amount of a biomarker as referred to herein, the relative amount or concentration of the said biomarker as well as any value or parameter which correlates thereto or can be derived therefrom. Such values or parameters comprise intensity signal values from all specific physical or chemical properties obtained from the said peptides by direct measurements, e.g., intensity values in mass spectra or NMR spectra. Moreover, encompassed are all values or parameters which are obtained by indirect measurements specified elsewhere in this description, e.g., response amounts measured from biological read out systems in response to the peptides or intensity signals obtained from specifically bound ligands. It is to be understood that values correlating to the aforementioned amounts or parameters can also be obtained by all standard mathematical operations.
The level of biomarker present in the biological fluid sample may be determined by e.g. assaying the amount of protein biomarker present in the sample. Assays for measuring the amount of a specified protein are well known in the art and include direct or indirect measures. The level of protein biomarker in a sample may also be determined by determining the level
of protein biomarker activity in a sample. Accordingly, protein “level” encompasses both the amount of protein per se, or its level of activity.
By way of example, the level of a protein biomarker in a biological fluid sample can be determined (e.g., measured) by any suitable methods and materials known in the art, including, for example, a process selected from the group consisting of mass spectrometry, immunoassays, enzymatic assays, spectrophotometry, colorimetry, fluorometry, bacterial assays, protein microarrays, compound separation techniques, or other known techniques for determining the presence and/or quantity of an analyte. Examples of relevant techniques include enzyme linked immunosorbent assays (ELISAs), immunoprecipitation, immunofluorescence, enzyme immunoassay (EIA), radioimmunoassay (RIA), Western blot analysis, and Lateral Flow (using e.g. Lateral Flow Devices (LFDs) utilizing a membrane bound antibody specific to the protein biomarker).
Preferably, the level of a protein biomarker in a biological fluid sample is measured by ELISA or lateral flow.
“CA-125”, the Carbohydrate antigen 125, sometimes named as Cancer Antigen 125 or Tumor Antigen 125, is a mucin-type glycoprotein, produced by the MUC16 gene, and associated with the cellular membrane. CA-125 is a biomarker for epithelial cell ovarian cancer being derived from coelomic epithelia including the endometrium, fallopian tube, ovary, and peritoneum. Diagnostic use of CA-125 is limited to endometriosis stages III and IV (moderate and severe endometriosis) with moderate sensitivity.
"Symptoms" of a disease are implications of the disease noticeable by the tissue, organ or organism having such disease and include but are not limited to pain, weakness, tenderness, strain, stiffness, and spasm of the tissue, an organ or an individual. "Signs" or "signals" of a disease include but are not limited to the change or alteration such as the presence, absence, increase or elevation, decrease or decline, of specific indicators such as biomarkers or molecular markers, or the development, presence, or worsening of symptoms. Symptoms of pain include but are not limited to an unpleasant sensation that may be felt as a persistent or varying burning, throbbing, itching or stinging ache.
The term "disease" and "disorder" are used interchangeably herein, referring to an abnormal condition, especially an abnormal medical condition such as an illness or injury, wherein a
tissue, an organ or an individual is not able to efficiently fulfil its function anymore. Typically, but not necessarily, a disease is associated with specific symptoms or signs indicating the presence of such disease. The presence of such symptoms or signs may thus, be indicative for a tissue, an organ or an individual suffering from a disease. An alteration of these symptoms or signs may be indicative for the progression of such a disease. A progression of a disease is typically characterised by an increase or decrease of such symptoms or signs which may indicate a "worsening" or "bettering" of the disease. The "worsening" of a disease is characterised by a decreasing ability of a tissue, organ or organism to fulfil its function efficiently, whereas the "bettering" of a disease is typically characterised by an increase in the ability of a tissue, an organ or an individual to fulfil its function efficiently. A tissue, an organ or an individual being at "risk of developing" a disease is in a healthy state but shows potential of a disease emerging. Typically, the risk of developing a disease is associated with early or weak signs or symptoms of such disease. In such case, the onset of the disease may still be prevented by treatment. Examples of a disease include but are not limited to inflammatory diseases, infectious diseases, cutaneous conditions, endocrine diseases, intestinal diseases, neurological disorders, joint diseases, genetic disorders, autoimmune diseases, traumatic diseases, and various types of cancer.
“Endometriosis” is a chronic, hormone-dependent, inflammatory disease that is characterized by lesions of endometrial-like tissue outside of the uterus. Clinical presentation of endometriosis varies significantly from patient to patient. Endometriosis patients often present with symptoms such as intermenstrual bleeding, painful periods (dysmenorrhea), painful intercourse (dyspareunia), painful defecation (dyschezia) and painful urination (dysuria). Pelvic pain due to endometriosis is usually chronic (lasting >6 months) and is associated with dysmenorrhea (in 50 to 90% of cases), dyspareunia, deep pelvic pain, and lower abdominal pain with or without back and loin pain. The pain can occur unpredictably and intermittently throughout the menstrual cycle or it can be continuous, and it can be dull, throbbing, or sharp, and exacerbated by physical activity. Bladder- and bowel-associated symptoms (nausea, distention, and early satiety) are typically cyclic. Pain often worsens overtime and may change in character; infrequently, women report burning or hypersensitivity, symptoms that are suggestive of a neuropathic component. Often, endometriosis can be asymptomatic, only coming to a clinician’s attention during evaluation for infertility (Sinaii et al. Fertil Steril. 2008; 89(3): 538-545). In women with endometriosis, there is a reduced monthly fecundity rate (2-
10%) compared with fertile couples (15-20%). Although endometriosis impairs fertility, it does not usually completely prevent conception (Fadhlaoui et al. Front Surg. 2014; 1 : 24).
The most commonly affected sites of endometriosis are the pelvic organs and peritoneum, although other parts of the body such as the lungs are occasionally affected. The extent of the disease varies from a few, small lesions on otherwise normal pelvic organs to large, ovarian endometriotic cysts (endometriomas) and/or extensive fibrosis and adhesion formation causing marked distortion of pelvic anatomy. Based on the location, endometriotic lesions can be classified into peritoneal endometriosis, ovarian endometriotic cysts (endometrioma), deep nodules (deep infiltrating endometriosis; Kennedy et al. Hum Reprod. 2005; 20(10): 2698- 2704). Deep infiltrating endometriosis is considered to be any manifestation of endometriosis that is located other than in the superficial tissues of the rectovaginal septum and vaginal fornix, the pelvic wall, parametrium, bowel, uterus, or urinary bladder (Halis. et al. (2010). Deutsches Arzteblatt International, 107(25), 446). Endometriosis can also involve the diaphragm (diaphragmatic endometriosis) or involve the thorax (thoracic endometriosis) (Nezhat et al. JSLS 2019).
As used herein the expressions “uterine/pelvic pathology” and “uterine and pelvic pathology” are used interchangeably herein and comprise adenomyosis, uterine fibroids, ovarian cysts requiring surgery, and uterine/pelvic cancer such as ovarian cancer or endometrial cancer.
The expression “endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain” as used herein refers to neuropathic pain defined by the International Association for the Study of Pain as “pain caused by disease or lesion of the somatosensory nervous system,” contrasting with nociceptive pain which is defined as “pain that arises from actual or threatened damage to nonneural tissue and is due to the activation of nociceptors”. Neuropathic pain could be expected to arise in the context of endometriosis for a number of reasons. (Coxon et al., /s there a Neuropathic-Like Component to Endometriosis-Associated Pain? Results From a Large Cohort Questionnaire Study, Front Pain Res (Lausanne), 2021 ; 2: 743812; doi: 10.3389/fpain.2021.743812).
The term “rASRM stage” or “rASRM staging” refers to the revised classification system established by the American Society for Reproductive Medicine (ASRM) describing the severity of endometriosis based on the findings at surgery (laparoscopy). The classification is
based on the morphology of peritoneal and pelvic implants such as red, white and black lesions, percentage of involvement of each lesion should be included. Number, size, and location endometrial implants, plaques, endometriomas and adhesions should be noted. Endometriosis in bowel, urinary tract, fallopian tube, vagina, cervix, skin, or other locations should be documented per ASRM guidelines. Stages of endometriosis according to ASRM guidelines are stage I, II, III, and IV determined based on the point scores and correspond to minimal, mild, moderate and severe endometriosis. The rASRM stages I & II endometriosis (minimal to mild endometriosis) are defined by superficial peritoneal endometriosis, possible presence of small deep lesions, absence of endometrioma and/or mild filmy adhesion. The rASRM stages III and IV endometriosis (moderate to severe endometriosis) are defined by the presence of superficial peritoneal endometriosis, deep infiltrating endometriosis with moderate to extensive adhesions between the uterus and bowels and/or endometrioma cysts with moderate to extensive adhesions involving the ovaries and tubes.
The term “VAS”, the Visual Analog Scale, is an instrument to assess the intensity of pain. The VAS consists of a 10-cm long horizontal line with its extremes marked as ‘no pain’ and ‘worst pain imaginable’. Each patient ticks her pain level on the line and the distance from ‘no pain’ on the extreme left to the tick mark is measured in centimeters, yielding a pain score from 0 to 10. ‘No pain’ corresponds to a pain score of 0, ‘worst pain imaginable’ corresponds to a pain score of 10. In women with endometriosis dysmenorrhea is associated with the highest perception of pain with a mean VAS score of about 6 (Cozzolino et al. Rev Bras Ginecol Obstet 2019; 41 (3): 170-175).
The subject may be referred to herein as a patient. The terms “subject”, “individual”, and “patient” are used herein interchangeably and refer to an animal, preferably a mammal and, more typically to a human. The patient is preferably a human female. There is a need for diagnosis of endometriosis at a young age, as it starts with the initiation of the menstruation. Therefore, the patient is preferably a young or adolescent human female aged between 12- 24 years. In an embodiment of the present invention, the patient is a young or adolescent human female. The subject can be symptomatic (e.g., the subject presents symptoms associated with endometriosis), or the subject can be asymptomatic (e.g., the subject does not present symptoms associated with endometriosis). The subject may be diagnosed with, be at risk of developing or present with symptoms of endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain. The
subject may have, or be suspected of having (e.g., present with symptoms or a history indicative or suggestive of) endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
Accordingly, in some examples, the subject has endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain (and the method diagnoses, identifies, (or detects) that the subject has endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain). In this context, the terms “diagnose” “identify”, and “detect” can be used interchangeably.
In particular examples, the subject has early stage (stage I or stage II) endometriosis.
The patient to be investigated by the method of the present invention shall be a patient having suspected endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
The term “suspected endometriosis” as used herein means that the patient shall exhibit clinical parameters, signs and/or symptoms of endometriosis. Thus, the patient according to the invention is, typically, a patient that suffers from an endometriosis or is suspected to suffer from an endometriosis. A patient having a suspicion of endometriosis with signs and symptoms: dysmenorrhea (painful menstrual periods), dysuria (painful urination), dyschezia (difficult or painful defecation), dyspareunia (pain during or after sexual intercourse) and chronic abdominal/pelvic pain independent of menstrual cycle, heavy menstrual bleeding, long menstrual periods, infertility, fatigue, cyclical lung problems (pneumothorax), cyclical cough, chest pain, or coughing of blood (haemoptysis), shoulder tip pain, painful rectal bleeding or the presence of blood in the urine (haematuria), and cyclical scar swelling and pain (EHRE Information on Endometriosis, 2022 www.eshre.eu/guidelines).
Alternatively, the EphA1 levels may be determined routinely as part of screening tests without any suspicion on endometriosis but to be able to detect symptom-free endometriosis at early stages.
By detecting decreased levels of EphA1 in a subject the suspicion that the subject suffers from endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-
associated neuropathic pain would be confirmed and there is a high risk that that the subject suffers from endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain . In particular, in cases where the subject already exhibits clinical parameters, signs and/or symptoms of endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain the determination of decreased EphA1 levels would confirm the presence of uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
The term "comparing" as used herein refers to comparing the amount/level of the biomarker in the sample from the subject with the reference amount or reference value of the biomarker specified elsewhere in this description. It is to be understood that comparing as used herein usually refers to a comparison of corresponding parameters or values, e.g., an absolute amount is compared to an absolute reference amount while a concentration is compared to a reference concentration or an intensity signal obtained from the biomarker in a sample is compared to the same type of intensity signal obtained from a reference sample. The comparison may be carried out manually or computer assisted. Thus, the comparison may be carried out by a computing device. The value of the measured or detected amount of the biomarker in the sample from the subject and the reference amount can be, e.g., compared to each other and the said comparison can be automatically carried out by a computer program executing an algorithm for the comparison. The computer program carrying out the said evaluation will provide the desired assessment in a suitable output format. For a computer- assisted comparison, the value of the measured amount may be compared to values corresponding to suitable references which are stored in a database by a computer program. The computer program may further evaluate the result of the comparison, i.e. automatically provide the desired assessment in a suitable output format. For a computer-assisted comparison, the value of the measured amount may be compared to values corresponding to suitable references which are stored in a database by a computer program. The computer program may further evaluate the result of the comparison, i.e. automatically provides the desired assessment in a suitable output format.
The terms “(appropriate) reference value”, "reference sample" or "control (sample)" as used herein, refers to a sample which is analysed in a substantially identical manner as the sample of interest and whose information is compared to that of the sample of interest. In one embodiment the “(appropriate) reference value” is a predetermined reference. A reference
sample thereby provides a standard allowing for the evaluation of the information obtained from the sample of interest. A control sample may be derived from a body fluid of a healthy individual, in particular serum or plasma for a non-invasive test, thereby providing a standard of a healthy status of a tissue, organ or individual. Differences between the status of the normal reference sample and the status of the sample of interest may be indicative of the presence or further progression of such disease or disorder. A control sample may be derived from an abnormal or diseased tissue, organ or individual thereby providing a standard of a diseased status of a tissue, organ or individual. Differences between the status of the normal or abnormal reference sample and the status of the sample of interest may be indicative of the absence or bettering of such disease or disorder.
A reference sample may also be derived from the same tissue, organ, or individual as the sample of interest but has been taken at an earlier time point. Differences between the status of the earlier taken reference sample and the status of the sample of interest may be indicative of the progression of the disease, i.e. a bettering or worsening of the disease over time.
The determined value can be compared to more than one (appropriate) reference values, which can be of different kind. For example the determined value can be compared to one or more values obtained from the same subject at earlier time points and in parallel it can be compared to one or more values obtained from other subjects (with a known stage of endometriosis).
The control sample may be an internal or an external control sample. An internal control sample is used, i.e. the marker level(s) is(are) assessed in the test sample as well as in one or more other sample(s) taken from the same subject to determine if there are any changes in the level(s) of said marker(s). For an external control sample the presence or amount of a marker in a sample derived from the individual is compared to its presence or amount in an individual known to suffer from, or known to be at risk of, a given condition; or an individual known to be free of a given condition, i.e., "normal individual".
It will be appreciated by the skilled artisan that such external control sample may be obtained from a single individual or may be obtained from a reference population that is age-matched and free of confounding diseases. Typically, samples from 100 well-characterized individuals from the appropriate reference population are used to establish a "reference value". However,
reference population may also be chosen to consist of 20, 30, 50, 200, 500 or 1000 individuals. Healthy individuals represent a preferred reference population for establishing a control value.
For example, a marker concentration in a patient sample can be compared to a concentration known to be associated with a specific course of a certain disease. For example, it can be compared to a concentration known to be associated with a certain stage of endometriosis. Usually, the sample's marker concentration is directly or indirectly correlated with a diagnosis and the marker concentration is e.g. used to determine whether an individual is at risk for a certain suffering from that disease. Alternatively the marker concentration can be compared to marker concentrations obtained from the same subject at an earlier time point. Alternatively, the sample's marker concentration can e.g., be compared to a marker concentration known to be associated with a response to therapy in a certain disease, the diagnosis of a certain disease, the assessment of the severity of a certain disease, the guidance for selecting an appropriate drug to a certain disease, in judging the risk of disease progression, or in the follow-up of patients. Depending on the intended diagnostic use an appropriate control sample is chosen and a control or reference value for the marker established therein. As also clear to the skilled artisan, the absolute marker values established in a control sample will be dependent on the assay used.
The most common control samples and/or reference values derived therefrom for the methods described herein are obtained from but not limited to “symptomatic controls” (also: SNF=symptomatic no findings). The corresponding subjects from which these samples are obtained are “symptomatic subjects” respectively.
“Symptomatic controls” (also: SNF=symptomatic no findings) refer to control samples of subjects that suffer from symptoms that are usually associated with endometriosis (e.g., menstrual/abdominal pain, infertility, etc) but where, based on laparoscopy, endometriosis can be excluded and no tissue alterations (e.g., adenomyosis, uterine/ovarian cysts, fibroids or uterine/pelvic cancer such as ovarian cancer, endometrial cancer) can be observed, that is symptomatic controls do not have endometriosis or any other uterine/pelvic pathology (e.g., adenomyosis, uterine/ovarian cysts, fibroids, uterine/pelvic cancer such as ovarian cancer and endometrial cancer).
Women with other “uterine/pelvic pathology” (SOF=symptomatic other findings) refer to a group of samples of subjects that have tissue alterations (e.g., adenomyosis, fibroids, uterine/ovarian cysts requiring surgery, and uterine/pelvic cancer) which however does not resemble endometriosis. Further, these subjects are most often symptomatic (e.g., menstrual/abdominal pain, infertility, etc).
The control sample may be assayed at the same time, before or after, separately or simultaneously with the test sample. The control value that is used in the comparison with the test sample may be a value that is calculated as an average or median of more than one (e.g., two or more, five or more, ten or more, a group etc) of control samples. Alternatively, the control sample may be a sample that originated from (i.e., is a mix of) more than one (e.g., two or more, five or more, ten or more, a group etc) individual that is not suffering from endometriosis (or is a “symptomatic control”) and not from other uterine/pelvic pathology.
In one example, the control sample is obtained from a subject that is a “symptomatic control” not having endometriosis or another uterine/pelvic pathology (adenomyosis, fibroids, ovarian cysts requiring surgery, uterine/pelvic cancer).
Alternatively, the level of biomarker (e.g., protein) in the biological fluid sample may be compared to a pre-determined reference level for the biomarker of interest. As used herein, a “predetermined reference level” refers to a biomarker level obtained from a reference database, which may be used to generate a pre-determined cut off value, i.e., a score that is statistically predictive of endometriosis. In one example, the predetermined reference level is the average or median level of the biomarker in at least one individual not suffering from endometriosis from the same species. The predetermined reference value may be calculated as the average or median, taken from a group or population of individuals that are not suffering from endometriosis. For example, the predetermined reference value may be calculated as the average or median, taken from a group or population of individuals that are “symptomatic controls”. The individual or the population of individuals can be the same age or in the same state or condition of health as the subject from which the test sample is obtained.
In one example, the pre-determined reference level is therefore the average level of the biomarker in a control subject that does not have endometriosis. In a further example the pre-
determined reference level is the average level of the biomarker in a subject that is a “symptomatic control”.
Typically, in methods for diagnosing endometriosis in a subject, the control sample or predetermined reference are obtained from an individual or group of individuals that are distinct from the subject that is being tested (i.e., the subject from which the test sample is obtained/provided). In such examples, the control or predetermined reference are used as a bench line to determine whether the tested subject has endometriosis.
In an alternative example, the control or predetermined reference value may be obtained from the same individual as the test sample, but at an earlier time point. This is particularly relevant for the methods described herein that determine the progression in a subject, that determine the therapeutic effect of a treatment regimen for endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain, and/or that determine a subject’s compliance or adherence with a prescribed treatment regimen for endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain. For this the samples are taken from the same biological fluid of the same subject, wherein the biological fluid is blood, serum, plasma, capillary blood, interstitial fluid, peritoneal fluid, or menstrual fluid preferably the biological fluid sample is serum.
In such examples, the control sample or predetermined reference level is used to determine any changes in the level of the biomarker(s) over a time interval for the same subject. The predetermined reference level or control sample can therefore be from the same subject that the test sample is obtained from, for example obtained at an earlier time point. This earlier time point can be before they were diagnosed with endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
A pre-determined level can be single cut-off value, such as a median or mean. It can be a range of cut-off (or threshold) values, such as a confidence interval. It can be established based upon comparative groups, such as where the risk in one defined group is a fold higher, or lower, (e.g., approximately 2-fold, 4-fold, 8-fold, 16-fold or more) than the risk in another defined group. It can be a range, for example, where a population of subjects (e.g., control subjects) is divided equally (or unequally) into groups, such as a low-risk group, a medium risk
group and a high-risk group, or into quartiles, the lowest quartile being subjects with the lowest risk and the highest quartile being subjects with the highest risk, or into n-quantiles (i.e., n regularly spaced intervals) the lowest of the n-quantiles being subjects with the lowest risk and the highest of the n-quantiles being subjects with the highest risk. Moreover, the reference could be a calculated reference, most preferably the average or median, for the relative or absolute amount of a biomarker of a population of individuals comprising the subject to be investigated. How to calculate a suitable reference value, preferably, the average or median, is well known in the art.
Thus, in some cases the level of the protein biomarker in a subject being less than or equal to the level of the biomarker of the control sample or pre-determined reference level is indicative of a clinical status (e.g., indicative of endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain).
Typically, but not necessarily, the greater than, or the less than, that is sufficient to distinguish a subject from a control subject is a statistically significantly greater than, or a statistically significant less than. In cases where the level of the biomarker in a subject being equal to the level of the biomarker in a control subject is indicative of (a stage of) endometriosis, the "being equal" refers to being approximately equal (e.g., not statistically different).
The pre-determined value can depend upon a particular population of subjects (e.g., human subjects) selected. For example, an apparently healthy population will have a different 'normal' range of the protein biomarker than will a population of subjects which have, or are likely to have, endometriosis. Accordingly, the pre-determined values selected may take into account the category (e.g., healthy, diseased, stage of disease) in which a subject (e.g., human subject) falls.
Appropriate ranges and categories can be selected with no more than routine experimentation by those of ordinary skill in the art.
Suitably, the level of the specific biomarker detected in a sample (e.g., a test sample, a control sample etc) may be normalized by adjusting the measured level (amount or activity) of the biomarker using the level of a reference protein in the same sample, wherein the reference protein is not a marker itself (it is e.g., a protein that is constitutively expressed). This normalization allows the comparison of the biomarker level in one sample to another sample,
or between samples from different sources. This normalized level can then optionally be compared to a reference value or control. For example, when measuring a protein biomarker in a whole blood sample the biomarker may be expressed as an absolute concentration or, alternatively, it may be normalized against a known protein constitutively expressed in whole blood such as albumin, immunoglobulins or plasma protein concentration.
For example, when measuring a protein biomarker in a serum (or plasma) sample the biomarker may be expressed as an absolute concentration or, alternatively, it may be normalized against a known protein constitutively expressed in serum (or plasma).
The biomarker level(s) in the test sample may be compared to the level of the same biomarker in a control sample or with a pre-determined reference level for the same biomarker to identify an increase or decrease in a level of the one or more biomarker in the sample of the subject.
In the methods described herein, the subject may be identified as having endometriosis if the comparison (between biomarker level(s) in the control sample/predetermined reference value and the test sample of the subject) indicates that the subject has a decreased level of EphA1 compared to the control sample or the pre-determined reference level.
Moreover, it will be understood that if the risk of the deterioration of the health condition is predicted, typically, the prediction is made within a predictive window of 6 month and two years. More typically, said predictive window is about a time window of about 6 months to 12 month for a non-invasive test dependent on the symptoms, such as pelvic pain.
As will be understood by those skilled in the art, the assessment made in accordance with the present invention, although preferred to be, may usually not be correct for 100% of the investigated subjects. The term, typically, requires that a statistically significant portion of subjects can be correctly assessed. Whether a portion is statistically significant can be determined without further ado by the person skilled in the art using various well known statistic evaluation tools, e.g., determination of confidence intervals, p-value determination, Student's t-test, Mann-Whitney test, etc. Details may be found in Dowdy and Wearden, Statistics for Research, John Wiley & Sons, New York 1983. Typically envisaged confidence intervals are at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%. The p-values are, typically, 0.2, 0.1 , 0.05.
The terms "lowered" or "decreased" level of an indicator refer to the level of such indicator in the sample being reduced in comparison to the reference (value) or reference sample. The terms "decrease", "decreased" "reduced", "reduction" or “down- regulated", “lower” are all used herein generally to mean a decrease by a statistically significant amount. However, for avoidance of doubt, "reduced", "reduction", "decreased" or "decrease" means a decrease by at least 10% as compared to a reference level/control, for example a decrease by at least about 20%, or at least about 30%, or at least about 40%, or at least about 50%, or at least about 60%, or at least about 70%, or at least about 80%, or at least about 90% or up to and including a 100% decrease (i.e. absent level as compared to a reference/control sample), or any decrease between 10-100% as compared to a reference level/control, or at least about a 0.5-fold, or at least about a 1.0-fold, or at least about a 1.2-fold, or at least about a 1.5-fold, or at least about a 2-fold, or at least about a 3 -fold, or at least about a 4-fold, or at least about a 5-fold or at least about a 10-fold decrease, or any decrease between 1.0-fold and 10-fold or greater as compared to a reference level/control.
The terms "elevated" or "increased" level of an indicator/(bio)marker refer to the level of such indicator in the sample being higher in comparison to the reference (value) or reference sample. E.g. a protein that is detectable in higher amounts in a fluid sample of one individual suffering from a given disease than in the same fluid sample of individuals not suffering from said disease, has an elevated level. The terms "increased", "increase" or "up-regulated", “higher” are all used herein to generally mean an increase by a statically significant amount; for the avoidance of any doubt, the terms "increased" or "increase" means an increase of at least 10% as compared to a reference level/control, for example an increase of at least about 20%, or at least about 30%, or at least about 40%, or at least about 50%, or at least about 60%, or at least about 70%, or at least about 80%, or at least about 90% or up to and including a 100% increase or any increase between 10-100% as compared to a reference level/control, or at least about a 0.5-fold, or at least about a 1.0-fold, or at least about a 1.2-fold, or at least about a 1 .5-fold, or at least about a 2-fold, or at least about a 3-fold, or at least about a 4-fold, or at least about a 5 -fold or at least about a 10-fold increase, or any increase between 1.0- fold and 10-fold or greater as compared to a reference level/control.
The terms "lower(ed)" or "decreased" level of an indicator/(bio)marker refer to the level of such indicator in the sample being lower in comparison to the reference (value) or reference sample.
E.g. a protein that is detectable in lower amounts in a fluid sample of one individual suffering from a given disease than in the same fluid sample of individuals not suffering from said disease, has a lowered level. The terms "less", "decrease" or "downregulated-regulated", “lower” are all used herein to generally mean a decrease by a statistically significant amount; for the avoidance of any doubt, the terms "decreased" or "decrease" means a decrease of at least 10% as compared to a reference level/control, for example a decrease of at least about 20%, or at least about 30%, or at least about 40%, or at least about 50%, or at least about 60%, or at least about 70%, or at least about 80%, or at least about 90% or up to and including a 100% decrease or any decrease between 10-100% as compared to a reference level/control, or at least about a 0.5-fold, or at least about a 1.0-fold, or at least about a 1.2- fold, or at least about a 1 .5-fold, or at least about a 2-fold, or at least about a 3-fold, or at least about a 4-fold, or at least about a 5 -fold or at least about a 10-fold decrease, or any decrease between 1.0-fold and 10-fold or greater as compared to a reference level/control.
The term "immunoglobulin (lg)" as used herein refers to immunity conferring glycoproteins of the immunoglobulin superfamily. "Surface immunoglobulins" are attached to the membrane of effector cells by their transmembrane region and encompass molecules such as but not limited to B-cell receptors, T -cell receptors, class I and II major histocompatibility complex (MHC) proteins, beta-2 microglobulin (~2M), CD3, CD4 and CDS.
Typically, the term "antibody" as used herein refers to secreted immunoglobulins which lack the transmembrane region and can thus, be released into the bloodstream and body cavities. Human antibodies are grouped into different isotypes based on the heavy chain they possess. There are five types of human lg heavy chains denoted by the Greek letters: a, y, 5, E, and p.- The type of heavy chain present defines the class of antibody, i.e. these chains are found in IgA, IgD, IgE, IgG, and IgM antibodies, respectively, each performing different roles, and directing the appropriate immune response against different types of antigens. Distinct heavy chains differ in size and composition; and may comprise approximately 450 amino acids (Janeway et al. (2001) Immunobiology, Garland Science). IgA is found in mucosal areas, such as the gut, respiratory tract and urogenital tract, as well as in saliva, tears, and breast milk and prevents colonization by pathogens (Underdown & Schiff (1986) Annu. Rev. Immunol. 4:389- 417). IgD mainly functions as an antigen receptor on B cells that have not been exposed to antigens and is involved in activating basophils and mast cells to produce antimicrobial factors (Geisberger et al. (2006) Immunology 118:429-437; Chen et al. (2009) Nat. Immunol. 10:889-
898). IgE is involved in allergic reactions via its binding to allergens triggering the release of histamine from mast cells and basophils. IgE is also involved in protecting against parasitic worms (Pier et al. (2004) Immunology, Infection, and Immunity, ASM Press). IgG provides the majority of antibody-based immunity against invading pathogens and is the only antibody isotype capable of crossing the placenta to give passive immunity to fetus (Pier et al. (2004) Immunology, Infection, and Immunity, ASM Press). In humans there are four different IgG subclasses (IgGI, 2, 3, and 4), named in order of their abundance in serum with IgGI being the most abundant (-66%), followed by lgG2 (-23%), lgG3 (-7%) and IgG (-4%). The biological profile of the different IgG classes is determined by the structure of the respective hinge region. IgM is expressed on the surface of B cells in a monomeric form and in a secreted pentameric form with very high avidity. IgM is involved in eliminating pathogens in the early stages of B cell mediated (humoral) immunity before sufficient IgG is produced (Geisberger et al. (2006) Immunology 118:429-437). Antibodies are not only found as monomers but are also known to form dimers of two Ig units (e.g. IgA), tetramers of four Ig units (e.g. IgM of teleost fish), or pentamers of five Ig units (e.g. mammalian IgM). Antibodies are typically made of four polypeptide chains comprising two identical heavy chains and identical two light chains which are connected via disulfide bonds and resemble a "Y"-shaped macro-molecule. Each of the chains comprises a number of immunoglobulin domains out of which some are constant domains and others are variable domains. Immunoglobulin domains consist of a 2-layer sandwich of between 7 and 9 antiparallel --strands arranged in two --sheets. Typically, the heavy chain of an antibody comprises four Ig domains with three of them being constant (CH domains: CHI. CH2. CH3) domains and one of the being a variable domain (V H). The light chain typically comprises one constant Ig domain (CL) and one variable Ig domain (V L). Exemplified, the human IgG heavy chain is composed of four Ig domains linked from N- to C- terminus in the order VwCH1-CH2-CH3 (also referred to as VwCyl-Cy2-Cy3), whereas the human IgG light chain is composed of two immunoglobulin domains linked from N- to C- terminus in the order VL-CL, being either of the kappa or lambda type (VK-CK or VA.-CA.). Exemplified, the constant chain of human IgG comprises 447 amino acids. Throughout the present specification and claims, the numbering of the amino acid positions in an immunoglobulin are that of the "Ell index" as in Kabat, E. A., Wu, T.T., Perry, H. M., Gottesman, K. S., and Foeller, C., (1991) Sequences of proteins of immunological interest, 5thed. U.S. Department of Health and Human Service, National Institutes of Health, Bethesda, MD. The "EU index as in Kabat" refers to the residue numbering of the human IgG IEU
antibody. Accordingly, CH domains in the context of IgG are as follows: "CHI" refers to amino acid positions 118-220 according to the Ell index as in Kabat; "CH2" refers to amino acid positions 237-340 according to the Ell index as in Kabat; and "CH3" refers to amino acid positions 341-44 7 according to the Ell index as in Kabat.
The terms "full-length antibody", "intact antibody", and "whole antibody" are used herein interchangeably to refer to an antibody in its substantially intact form, not antibody fragments as defined below. The terms particularly refer to an antibody with heavy chains that contain an Fc region.
Papain digestion of antibodies produces two identical antigen binding fragments, called "Fab fragments" (also referred to as "Fab portion" or "Fab region") each with a single antigen binding site, and a residual "Fe fragment" (also referred to as "Fe portion" or "Fe region") whose name reflects its ability to crystallize readily. The crystal structure of the human IgG Fe region has been determined (Deisenhofer (1981) Biochemistry 20:2361-2370). In IgG, IgA and IgD isotypes, the Fe region is composed of two identical protein fragments, derived from the CH2 and CH3 domains of the antibody's two heavy chains; in IgM and IgE isotypes, the Fe regions contain three heavy chain constant domains (CH2-4) in each polypeptide chain. In addition, smaller immunoglobulin molecules exist naturally or have been constructed artificially. The term "Fab1 fragment" refers to a Fab fragment additionally comprise the hinge region of an Ig molecule whilst "F(ab')2 fragments" are understood to comprise two Fab' fragments being either chemically linked or connected via a disulfide bond. Whilst "single domain antibodies (sdAb )" (Desmyter et al. (1996) Nat. Structure Biol. 3:803-811) and "Nanobodies" only comprise a single VH domain, "single chain Fv (scFv)" fragments comprise the heavy chain variable domain joined via a short linker peptide to the light chain variable domain (Huston et al. (1988) Proc. Natl. Acad. Sci. USA 85, 5879-5883). Divalent single-chain variable fragments (di-scFvs) can be engineered by linking two scFvs (scFvA-scFvB). This can be done by producing a single peptide chain with two VH and two VL regions, yielding "tandem scFvs" (VHA-VLA-VHB-VLB). Another possibility is the creation of scFvs with linkers that are too short for the two variable regions to fold together, forcing scFvs to dimerize. Usually, linkers with a length of 5 residues are used to generate these dimers. This type is known as "diabodies". Still shorter linkers (one or two amino acids) between a V H and V L domain lead to the formation of monospecific trimers, so-called "triabodies" or "tribadies". Bispecific diabodies are formed by expressing to chains with the arrangement VHA-VLB and
VHB-VLA or VLA-VHB and VLB-VHA, respectively. Singlechain diabodies (scDb) comprise a VHA-VLB and a VHB-VLA fragment which are linked by a linker peptide (P) of 12-20 amino acids, preferably 14 amino acids, (VHA-VLB-P-VHB-VLA). "Bi-specific T-cell engagers (BiTEs)" are fusion proteins consisting of two scFvs of different antibodies wherein one of the scFvs binds to T cells via the CD3 receptor, and the other to a tumor cell via a tumor specific molecule (Kufer et al. (2004) Trends Biotechnol. 22:238-244). Dual affinity retargeting molecules ("DART" molecules) are diabodies additionally stabilized through a C-terminal disulfide bridge.
Accordingly, the term "antibody fragments" refers to a portion of an intact antibody, preferably comprising the antigen-binding region thereof. Antibody fragments include but are not limited to Fab, Fab', F(ab')2, Fv fragments; diabodies; sdAb, nanobodies, scFv, di-scFvs, tandem scFvs, triabodies, diabodies, scDb, BiTEs, and DARTs.
The term "binding affinity" generally refers to the strength of the sum total of noncovalent interactions between a single binding site of a molecule (e.g., an antibody) and its binding partner (e.g., an antigen). Unless indicated otherwise, as used herein, “binding affinity” refers to intrinsic binding affinity which reflects a 1 :1 interaction between members of a binding pair (e.g., antibody and antigen). The affinity of a molecule X for its partner Y can generally be represented by the dissociation constant (Kd). Affinity can be measured by common methods known in the art, including but not limited to surface plasmon resonance-based assay (such as the BIAcore assay as described in PCT Application Publication No. W02005/012359); enzyme-linked immunoabsorbent assay (ELISA); and competition assays (e.g. RIA’s). Low- affinity antibodies generally bind antigen slowly and tend to dissociate readily, whereas high- affinity antibodies generally bind antigen faster and tend to remain bound longer. A variety of methods of measuring binding affinity are known in the art, any of which can be used for purposes of the present invention.
"Sandwich immunoassays" are broadly used in the detection of an analyte of interest. In such assay the analyte is “sandwiched” in between a first antibody and a second antibody. Typically, a sandwich assay requires that capture and detection antibody bind to different, nonoverlapping epitopes on an analyte of interest. By appropriate means such sandwich complex is measured and the analyte thereby quantified. In a typical sandwich-type assay, a first antibody bound to the solid phase or capable of binding thereto and a detectably-labeled
second antibody each bind to the analyte at different and non-overlapping epitopes. The first analyte-specific binding agent (e.g., an antibody) is either covalently or passively bound to a solid surface. The solid surface is typically glass or a polymer, the most commonly used polymers being cellulose, polyacrylamide, nylon, polystyrene, polyvinyl chloride, or polypropylene. The solid supports may be in the form of tubes, beads, discs of microplates, or any other surface suitable for conducting an immunoassay. The binding processes are well- known in the art and generally consist of cross-linking covalently binding or physically adsorbing, the polymer-antibody complex is washed in preparation for the test sample. An aliquot of the sample to be tested is then added to the solid phase complex and incubated for a period of time sufficient {e.g., 2-40 minutes or overnight if more convenient) and under suitable conditions (e.g., from room temperature to 40°C such as between 25° C and 37° C inclusive) to allow for binding between the first or capture antibody and the corresponding antigen. Following the incubation period, the solid phase, comprising the first or capture antibody and bound thereto the antigen can be washed, and incubated with a secondary or labeled antibody binding to another epitope on the antigen. The second antibody is linked to a reporter molecule which is used to indicate the binding of the second antibody to the complex of first antibody and the antigen of interest.
An extremely versatile alternative sandwich assay format includes the use of a solid phase coated with the first partner of a binding pair, e.g., paramagnetic streptavidin-coated microparticles. Such microparticles are mixed and incubated with an analyte-specific binding agent bound to the second partner of the binding pair (e.g., a biotinylated antibody), a sample suspected of comprising or comprising the analyte, wherein said second partner of the binding pair is bound to said analyte-specific binding agent, and a second analyte-specific binding agent which is detectably labeled. As obvious to the skilled person these components are incubated under appropriate conditions and for a period of time sufficient for binding the labeled antibody via the analyte, the analyte-specific binding agent (bound to) the second partner of the binding pair and the first partner of the binding pair to the solid phase microparticles. As appropriate such assay may include one or more washing step(s).
The term "detectably labeled" encompasses labels that can be directly or indirectly detected.
Directly detectable labels either provide a detectable signal or they interact with a second label to modify the detectable signal provided by the first or second label, e.g., to give FRET
(fluorescence resonance energy transfer). Labels such as fluorescent dyes and luminescent (including chemiluminescent and electrochemiluminescent) dyes (Briggs et al "Synthesis of Functionalised Fluorescent Dyes and Their Coupling to Amines and Amino Acids," J. Chem. Soc., Perkin-Trans. 1 (1997) 1051-1058) provide a detectable signal and are generally applicable for labeling. In one embodiment detectably labeled refers to a label providing or inducible to provide a detectable signal, i.e. , to a fluorescent label, to a luminescent label (e.g., a chemiluminescent label or an electrochemiluminescent label), a radioactive label or a metalchelate based label, respectively.
Numerous labels (also referred to as dyes) are available which can be generally grouped into the following categories, all of them together and each of them representing embodiments according the present disclosure:
(a) Fluorescent dyes
Fluorescent dyes are e.g., described by Briggs et al "Synthesis of Functionalized Fluorescent Dyes and Their Coupling to Amines and Amino Acids," J. Chem. Soc., Perkin-Trans. 1 (1997) 1051-1058).
Fluorescent labels orfluorophores include rare earth chelates (europium chelates), fluorescein type labels including FITC, 5-carboxyfluorescein, 6-carboxy fluorescein; rhodamine type labels including TAMRA; dansyl; Lissamine; cyanines; phycoerythrins; Texas Red; and analogs thereof. The fluorescent labels can be conjugated to an aldehyde group comprised in target molecule using the techniques disclosed herein. Fluorescent dyes and fluorescent label reagents include those which are commercially available from Invitrogen/Molecular Probes (Eugene, Oregon, USA) and Pierce Biotechnology, Inc. (Rockford, III.).
(b) Luminescent dyes
Luminescent dyes or labels can be further subcategorized into chemiluminescent and electrochemiluminescent dyes.
The different classes of chemiluminogenic labels include luminol, acridinium compounds, coelenterazine and analogues, dioxetanes, systems based on peroxyoxalic acid and their
derivatives. For immunodiagnostic procedures predominantly acridinium based labels are used (a detailed overview is given in Dodeigne C. et al., Taianta 51 (2000) 415-439).
The labels of major relevance used as electrochemiluminescent labels are the Ruthenium- and the Iridium-based electrochemiluminescent complexes, respectively. Electrochemiluminescense (ECL) proved to be very useful in analytical applications as a highly sensitive and selective method. It combines analytical advantages of chemiluminescent analysis (absence of background optical signal) with ease of reaction control by applying electrode potential. In general Ruthenium complexes, especially [Ru (Bpy)3]2+ (which releases a photon at -620 nm) regenerating with TPA (Tripropylamine) in liquid phase or liquid-solid interface are used as ECL-labels.
Electrochemiluminescent (ECL) assays provide a sensitive and precise measurement of the presence and concentration of an analyte of interest. Such techniques use labels or other reactants that can be induced to luminesce when electrochemically oxidized or reduced in an appropriate chemical environment. Such electrochemiluminescense is triggered by a voltage imposed on a working electrode at a particular time and in a particular manner. The light produced by the label is measured and indicates the presence or quantity of the analyte. For a fuller description of such ECL techniques, reference is made to US Patent No. 5,221 ,605,
US Patent No. 5,591 ,581 , US Patent No. 5,597,910, PCT published application W090/05296,
PCT published application WO92/14139, PCT published application W090/05301 , PCT published application WO96/24690, PCT published application US95/03190, PCT application
US97/16942, PCT published application US96/06763, PCT published application WO95/08644, PCT published application WO96/06946, PCT published application WO96/33411 , PCT published application W087/06706, PCT published application WO96/39534, PCT published application WO96/41175, PCT published application WO96/40978, PCT/US97/03653 and US patent application 08/437,348 (U.S. Patent No.
5,679,519). Reference is also made to a 1994 review of the analytical applications of ECL by
Knight, et al. (Analyst, 1994, 119: 879-890) and the references cited therein. In one embodiment the method according to the present description is practiced using an electrochemiluminescent label.
Recently also Iridium-based ECL-labels have been described (W02012107419).
(c) Radioactive labels make use of radioisotopes (radionuclides), such as 3H, 11 C, 14C, 18F, 32P, 35S, 64Cu, 68Gn, 86Y, 89Zr, 99TC, 1111n, 1231, 1241, 1251, 1311, 133Xe, 177Lu, 211 At, or 131 Bi.
(d) Metal-chelate complexes suitable as labels for imaging and therapeutic purposes are well- known in the art (US 2010/0111861 ; US 5,342,606; US 5,428,155; US 5,316,757; US 5,480,990; US 5,462,725; US 5,428,139; US 5,385,893; US 5,739,294; US 5,750,660; US 5,834,461 ; Hnatowich et al, J. Immunol. Methods 65 (1983) 147-157; Meares et al, Anal. Biochem. 142 (1984) 68-78; Mirzadeh et al, Bioconjugate Chem. 1 (1990) 59-65; Meares et al, J. Cancer (1990), Suppl. 10:21-26; Izard et al, Bioconjugate Chem. 3 (1992) 346-350; Nikula et al, Nucl. Med. Biol. 22 (1995) 387-90; Camera et al, Nucl. Med. Biol. 20 (1993) 955- 62; Kukis et al, J. Nucl. Med. 39 (1998) 2105-2110; Verel et al., J. Nucl. Med. 44 (2003) 1663- 1670; Camera et al, J. Nucl. Med. 21 (1994) 640-646; Ruegg et al, Cancer Res. 50 (1990) 4221-4226; Verel et al, J. Nucl. Med. 44 (2003) 1663-1670; Lee et al, Cancer Res. 61 (2001) 4474-4482; Mitchell, et al, J. Nucl. Med. 44 (2003) 1105-1112; Kobayashi et al Bioconjugate Chem. 10 (1999) 103-111 ; Miederer et al, J. Nucl. Med. 45 (2004) 129-137; DeNardo et al, Clinical Cancer Research 4 (1998) 2483-90; Blend et al, Cancer Biotherapy & Radiopharmaceuticals 18 (2003) 355-363; Nikula et al J. Nucl. Med. 40 (1999) 166-76; Kobayashi et al, J. Nucl. Med. 39 (1998) 829-36; Mardirossian et al, Nucl. Med. Biol. 20 (1993) 65-74; Roselli et al, Cancer Biotherapy & Radiopharmaceuticals, 14 (1999) 209-20).
The methods described herein can further comprise selecting, and optionally administering, a treatment regimen for the subject based on the diagnosis (i.e. , based on the comparison of the levels of the biomarkers with the reference values/levels/controls). Treatment can include, for example, surgery and, in some cases, therapy, or combinations thereof. However, in some cases, immediate treatment may not be required, and the subject may be selected for active surveillance.
As used herein, the terms “active surveillance”, “monitoring” and “watchful waiting” are used interchangeably herein to mean closely monitoring a patient's condition without giving any treatment until symptoms appear or change.
As used herein, the terms “treat”, “treating” and "treatment" are taken to include an intervention performed with the intention of altering the pathology of a condition, disorder or symptom (i.e.,
in this case endometriosis). Accordingly, "treatment" refers to therapeutic treatment, wherein the object is to slow down (lessen) the targeted condition, disorder or symptom. “Treatment” therefore encompasses a reduction, slowing or inhibition of the symptoms of endometriosis, for example of at least 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90% or 100% when compared to the symptoms before treatment. In the context of endometriosis, appropriate treatment may include pain medication, hormone treatments (such as hormonal contraceptives), gonadotropin-releasing hormone (GnRH) agonists, and/or surgery. (Longo, D.L et al. 2020. Sc. D. N Engl J Med, 382, pp.1244-56).
As used herein, the term “surgery” applies to surgical methods undertaken for removal of endometric tissue, like, for example, laparoscopy or nerve sparing surgery.
As used herein, the term “therapy” includes drug-based therapy, radiation, hormonal therapy, cryosurgery, chemotherapy, immunotherapy, biologic therapy, and high-intensity focused ultrasound. Drug-based therapy of endometriosis can for example be by inhibiting or targeting neurogenic inflammation and/or pain medication and/or hormonal therapy.
The type of treatment will vary depending on the particular form and/or stage of endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain that the subject has, is suspected of having.
Embodiments
The inventors have surprisingly identified the new protein biomarker EphA1 that is decreased in biological fluids, in particular serum, of women with endometriosis, especially in women with early stages of endometriosis.
The biomarker EphA1 can be used for diagnosing endometriosis in a subject compared to a control (e.g., non-pathological subjects or symptomatic subjects).
In particular, serum EphA1 can be used as a blood biomarker for early diagnosis and risk stratification of endometriosis. Furthermore, serum EphA1 can be used to select patients with disease stage I and stage II for early medical management of endometriosis. Therefore, it can
significantly reduce endometriosis diagnostic delay, improve patients' lives, and reduce the economic burden.
The biomarker can advantageously be used in any of the methods, kits, assays, or uses described herein.
Methods for assessing endometriosis in a subject
In a first aspect the present invention relates to a method for assessing endometriosis in a subject, the method comprising the steps of: a) determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject, b) comparing the level of the extracellular protein part of EphA1 to at least one appropriate reference value of a level of the extracellular protein part of EphA1, c) identifying a subject as having endometriosis if the comparison in step b) indicates that the subject has a decreased level of the extracellular protein part of EphA1 compared to the appropriate reference value, wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
In embodiments, a decreased level or amount or concentration of EphA1 in the fluid sample of the subject is indicative of the presence of endometriosis in the subject. In particular, an amount or concentration of EphA1 in the fluid sample of the subject is indicative of the presence of endometriosis in the subject if the amount or concentration of EphA1 in the fluid sample of the subject is less than the amount or concentration of EphA1 according to a reference value.
In one embodiment the method is an in vitro method.
In a particular embodiment the at least one appropriate reference value is i. a level of EphA1 in a SNF subject or in a SOF subject, or is
ii. an average level of EphA1 in a group of SNF subjects or in a group of SOF subjects, or a combination thereof, or wherein the at least one appropriate reference value is iii. a predetermined value of a level of EphA1 in a SNF subject or SOF subject, or is iv. a predetermined average value of a level of EphA1 in a group of SNF subjects or in a group of SOF subjects, or a combination thereof.
In a particular embodiment, the inventors could detect a decrease of serum EphA1 in the early and late stages of endometriosis compared to the control levels.
In particular, an amount of EphA1 lowered by 15% or more, 20% or more, 30% or more, 40% or more, 50% or more, is indicative of the presence or the risk of developing of endometrioses. In particular, an amount of EphA1 lowered by 100% or more, is indicative of the presence of endometrioses. In particular, an amount of EphA1 lowered by 150% or more, is indicative of the presence of endometrioses. In particular, an amount of EphA1 lowered by 200% or more, is indicative of endometrioses.
In a particular embodiment, an amount of EphA1 lowered by 10% or more is indicative of the presence or risk of having or developing endometriosis.
Suitably, the biological fluid sample is blood, serum, plasma, capillary blood, interstitial fluid, peritoneal fluid, or menstrual fluid, preferably the biological fluid sample is serum.
In one embodiment the biological fluid sample is serum.
In embodiments, the sample is an in vitro sample, i.e., it will be analyzed in vitro and not transferred back into the body of the subject. In embodiments, the method of the present invention is an in vitro method.
In particular embodiments, the subject is a human subject. In particular embodiments, the patient is a female human subject. In particular embodiments, the subject is a young or
adolescent human female. In particular, the subject is a subject who is capable of suffering from endometriosis due to the physical condition.
In particular, the assessment is carried out without performing laparoscopy. In particular, the assessment is performed without assessing the presence or severity of endometriosis in the patient using laparoscopy and/ the rASRM staging.
According to the invention, endometriosis is early endometriosis, in particular stage I endometriosis according to rASRM staging or stage II endometriosis according to rASRM staging.
In embodiments according to the invention, the protein level of EphA1 is determined, optionally using a process selected from: ELISA assay, immunoblotting, lateral flow assay, protein microarray and mass spectrometry.
Preferably the level of EphA1 is determined by detecting the extracellular protein part of EphA1 by ELISA or by ECLIA.
In a particular embodiment the extracellular protein part of EphA1 is detected by identification of at least one of the following peptides: WEPPADTGGR (SEQ ID NO: 1), AQGELGWLLDPPK (SEQ ID NO: 2), MHCSPDGEWLVPVGR (SEQ ID NO: 3), GLYLAFHNPGACVALVSVR (SEQ ID NO: 4), APGEGPQVACTGPPSAPR (SEQ ID NO: 5), VTTVAADQSFTIR (SEQ ID NO: 6), or VHVELQFTVR (SEQ ID NO: 7). Details are shown in Table 1.
Table 1 : Peptides identified of the EphA1 extracellular protein part detected using advanced mass spectrometry proteomics technology in serum samples of women with endometriosis (cases) and women without endometriosis (controls).
In embodiments, the amount of EphA1 is determined using antibodies, in particular using monoclonal antibodies. In embodiments, step a) of determining the amount of EphA1 in a sample of the patient comprises performing an immunoassay. In embodiments, the immunoassay is performed either in a direct or indirect format. In embodiments such immunoassays are selected from the group consisting of enzyme linked immunosorbent assay (ELISA), enzyme immunoassay (EIA), radioimmunoassay (RIA), or immuno assays based on detection of luminescence, fluorescence, chemiluminescence or electrochemiluminescence. In particular embodiments, step a) of determining the level of EphA1 in a sample of the subject comprises the steps of
i) incubating the sample of the subject with one or more antibodies specifically binding to EphA1 , thereby generating a complex between the antibody and EphA1 , and ii) quantifying the complex formed in step i), thereby quantifying the amount of EphA1 in the sample of the subject.
In particular embodiments, in step i) the sample is incubated with two antibodies, specifically binding to EphA1 . As obvious to the skilled artisan, the sample can be contacted with the first and the second antibody in any desired order, i.e., first antibody first and then the second antibody or second antibody first and then the first antibody, or simultaneously, for a time and under conditions sufficient to form a first anti- EphA1 antibody/ EphA1 /second anti- EphA1 antibody complex. As the skilled artisan will readily appreciate it is nothing but routine experimentation to establish the time and conditions that are appropriate or that are sufficient for the formation of a complex either between the specific anti- EphA1 antibody and the EphA1 antigen/analyte (= anti- EphA1 complex) or the formation of the secondary, or sandwich complex comprising the first antibody to EphA1 , EphA1 (the analyte) and the second anti - EphA1 antibody (=anti- EphA1 antibody/ EphA1 /second anti- EphA1 antibody complex).
The detection of the anti- EphA1 antibody I EphA1 complex can be performed by any appropriate means. The detection of the first anti- EphA1 antibody/ EphA1 /second anti- EphA1 antibody complex can be performed by any appropriate means. The person skilled in the art is absolutely familiar with such means/methods.
In certain embodiments a sandwich will be formed comprising a first antibody to EphA1 , EphA1 (analyte) and the second antibody to EphA1 , wherein the second antibody is detectably labeled.
In one embodiment a sandwich will be formed comprising a first antibody to EphA1 , the EphA1 (analyte) and the second antibody to EphA1 , wherein the second antibody is detectably labeled and wherein the first anti- EphA1 antibody is capable of binding to a solid phase or is bound to a solid phase.
In embodiments, the second antibody is directly or indirectly detectably labeled. In particular embodiments, the second antibody is detectably labeled with a luminescent dye, in particular a chemiluminescent dye or an electrochemiluminescent dye.
In embodiments, the endometriosis assessed is selected from the group consisting of peritoneal endometriosis, endometrioma, and deep infiltrating endometriosis (DIE).
In particular embodiments, the endometriosis diagnosed is peritoneal endometriosis. In other particular embodiments the endometriosis diagnosed is peritoneal endometriosis of stage I or stage II according to rASRM staging.
In another embodiment the methods further comprise selecting a treatment regimen for the subject based on the comparison of the level of EphA1 with the control sample or with the predetermined reference level. In particular embodiments, the method further comprises administering the selected treatment regimen to the subject, optionally wherein the selected treatment regimen comprises drug-based therapy and/or surgical treatment (laparoscopy). Drug-based therapy of endometriosis can for example be by pain medication hormone treatments, and/or surgery.
Depending on whether the diagnosis rather hints to a severe disease stage or not the person skilled in the art would be well aware of how to select the most appropriate and promising treatment regimen.
In embodiments, the method further comprising the assessment of dysmenorrhea and/or lower abdominal pain in the patient. In embodiments the presence of dysmenorrhea and/or lower abdominal pain is assessed according to the VAS scale. In embodiments, dysmenorrhea VAS score of 4 or higher indicated moderate or severe dysmenorrhea. In embodiments, scores of 3 or less indicate no or mild dysmenorrhea.
In embodiments, the method further comprising determining the level of CA-125 in the biological fluid sample from the subject.
When determining the level of EphA1 and CA-125 in one embodiment the method comprises calculating
• a ratio of the amount or concentration the extracellular protein part of EphA1 and the amount or concentration of CA-125, or
• a ratio of the amount or concentration of the extracellular protein part of EphA1 and dysmenorrhea, or
• a ratio of the amount or concentration the extracellular protein part of EphA1 and the amount or concentration of CA-125 and dysmenorrhea, or
• a ratio of the amount or concentration the extracellular protein part of EphA1 and lower abdominal pain according to the VAS scale.
Methods for assessing uterine/pelvic pathology in a subject
The inventors have surprisingly identified the new protein biomarker EphA1 that is decreased in biological fluids, in particular serum, of women with uterine/pelvic pathology.
The biomarker EphA1 can be used for diagnosing uterine/pelvic pathology in a subject compared to a control (e.g., SNF or SOF).
In particular, serum EphA1 can be used as a blood biomarker for early diagnosis and risk stratification of uterine/pelvic pathology. Furthermore, serum EphA1 can be used to select patients for early medical management of uterine/pelvic pathology. Therefore, it can significantly reduce uterine/pelvic pathology diagnostic delay, improve patients' lives, and reduce the economic burden.
Accordingly, in a second aspect the present invention relates to a method for assessing uterine/pelvic pathology in a subject, the method comprising the steps of: a) determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject, b) comparing the level of the extracellular protein part of EphA1 to at least one appropriate reference value of a level of the extracellular protein part of EphA1,
c) identifying a subject as having uterine/pelvic pathology if the comparison in step b) indicates that the subject has a decreased level of the extracellular protein part of EphA1 compared to the appropriate reference value.
In one embodiment the method is an in vitro method.
In a particular embodiment the at least one appropriate reference value is i. a level of EphA1 in a SNF subject or in a SOF subject, or is ii. an average level of EphA1 in a group of SNF subjects or in a group of SOF subjects, or a combination thereof, or wherein the at least one appropriate reference value is iii. a predetermined value of a level of EphA1 in a SNF subject or SOF subject, or is iv. a predetermined average value of a level of EphA1 in a group of SNF subjects or in a group of SOF subjects, or a combination thereof.
In particular, an amount of EphA1 lowered by 15% or more, 20% or more, 30% or more, 40% or more, 50% or more, is indicative of the presence or the risk of developing of uterine/pelvic pathology. In particular, an amount of EphA1 lowered by 100% or more, is indicative of the presence of uterine/pelvic pathology. In particular, an amount of EphA1 lowered by 150% or more, is indicative of the presence of uterine/pelvic pathology. In particular, an amount of EphA1 lowered by 200% or more, is indicative of uterine/pelvic pathology.
In a particular embodiment, an amount of EphA1 lowered by 10% or more is indicative of the presence or risk of having or developing uterine/pelvic pathology.
Suitably, the biological fluid sample is blood, serum, plasma, capillary blood, interstitial fluid, peritoneal fluid, or menstrual fluid, preferably the biological fluid sample is serum.
In one embodiment the biological fluid sample is serum.
In embodiments according to the invention, the protein level of EphA1 is determined, optionally using a process selected from: ELISA assay, immunoblotting, lateral flow assay, protein microarray and mass spectrometry.
Preferably the level of EphA1 is determined by detecting the extracellular protein part of EphA1 by ELISA or by ECLIA.
In a particular embodiment the extracellular protein part of EphA1 is detected by identification of at least one of the following peptides: WEPPADTGGR (SEQ ID NO: 1), AQGELGWLLDPPK (SEQ ID NO: 2), MHCSPDGEWLVPVGR (SEQ ID NO: 3), GLYLAFHNPGACVALVSVR (SEQ ID NO: 4), APGEGPQVACTGPPSAPR (SEQ ID NO: 5), VTTVAADQSFTIR (SEQ ID NO: 6), or VHVELQFTVR (SEQ ID NO: 7). Details are shown in Table 1.
Statements made with regards to determining the level of EphA1 made elsewhere herein (e.g. section referring to the assessment of endometriosis) equally apply here.
In embodiments the uterine/pelvic pathology is selected from the group consisting of adenomyosis, fibroids, ovarian cysts requiring surgery and uterine/pelvic cancer.
In another embodiment the methods further comprise selecting a treatment regimen for the subject based on the comparison of the level of EphA1 with the control sample or with the predetermined reference level. In particular embodiments, the method further comprises administering the selected treatment regimen to the subject, optionally wherein the selected treatment regimen comprises drug-based therapy and/or surgical treatment (laparoscopy).
Details of the biomarkers, combinations, samples, methods steps, subjects, types of endometriosis, treatments, reference values, etc are provided elsewhere and apply equally to this and all the other aspects.
Methods for assessing endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject
According to a third aspect the invention relates to a method for assessing endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject, the method comprising the steps of: a) determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject, b) comparing the level of the extracellular protein part of EphA1 to at least one appropriate reference value of a level of the extracellular protein part of EphA1 , c) identifying a subject as having endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain if the comparison in step b) indicates that the subject has a decreased level of the extracellular protein part of EphA1 compared to the appropriate reference value, wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
In one embodiment the method is an in vitro method.
In a particular embodiment the at least one appropriate reference value is i. a level of EphA1 in a SNF subject or in a SOF subject, or is ii. an average level of EphA1 in a group of SNF subjects or in a group of SOF subjects, or a combination thereof, or wherein the at least one appropriate reference value is iii. a predetermined value of a level of EphA1 in a SNF subject or SOF subject, or is iv. a predetermined average value of a level of EphA1 in a group of SNF subjects or in a group of SOF subjects, or a combination thereof.
In particular, an amount of EphA1 lowered by 15% or more, 20% or more, 30% or more, 40% or more, 50% or more, is indicative of the presence or the risk of developing of endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain. In particular, an amount of EphA1 lowered by 100% or more, is indicative of the presence of endometriosis- and/or
uterine/pelvic pathology-associated neuropathic pain. In particular, an amount of EphA1 lowered by 150% or more, is indicative of the presence of endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain. In particular, an amount of EphA1 lowered by 200% or more, is indicative of endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
In a particular embodiment, an amount of EphA1 lowered by 10% or more is indicative of the presence or risk of having or developing endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain.
Suitably, the biological fluid sample is blood, serum, plasma, capillary blood, interstitial fluid, peritoneal fluid, or menstrual fluid, preferably the biological fluid sample is serum.
In one embodiment the biological fluid sample is serum.
In embodiments according to the invention, the protein level of EphA1 is determined, optionally using a process selected from: ELISA assay, immunoblotting, lateral flow assay, protein microarray and mass spectrometry.
Preferably the level of EphA1 is determined by detecting the extracellular protein part of EphA1 by ELISA or by ECLIA.
In a particular embodiment the extracellular protein part of EphA1 is detected by identification of at least one of the following peptides: WEPPADTGGR (SEQ ID NO: 1), AQGELGWLLDPPK (SEQ ID NO: 2), MHCSPDGEWLVPVGR (SEQ ID NO: 3), GLYLAFHNPGACVALVSVR (SEQ ID NO: 4), APGEGPQVACTGPPSAPR (SEQ ID NO: 5), VTTVAADQSFTIR (SEQ ID NO: 6), or VHVELQFTVR (SEQ ID NO: 7). Details are shown in Table 1.
Statements made with regards to determining the level of EphA1 made elsewhere herein (e.g. section referring to the assessment of endometriosis) equally apply here.
In another embodiment the methods further comprise selecting a treatment regimen for the subject based on the comparison of the level of EphA1 with the control sample or with the pre-
determined reference level. In particular embodiments, the method further comprises administering the selected treatment regimen to the subject, optionally wherein the selected treatment regimen comprises drug-based therapy and/or surgical treatment (laparoscopy).
Details of the biomarkers, combinations, samples, methods steps, subjects, types of endometriosis, treatments, reference values, etc are provided elsewhere and apply equally to this and all the other aspects.
Methods for monitoring endometriosis progression, uterine/pelvic pathology progression and/or progression of endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject
In a fourth aspect the present invention relates to a method for monitoring endometriosis progression, uterine/pelvic pathology progression and/or progression of endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject, the method comprising the steps of: i. determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject in accordance with method steps a) to b) described above herein, ii. repeating step i. using a biological fluid sample obtained from the subject during or after treatment for a time interval; and iii. comparing the levels of the extracellular protein part of EphA1 identified in i. with the levels of extracellular protein part of EphA1 identified in ii., wherein a change in the levels of extracellular protein part of EphA1 from i. to ii. is indicative of a change in endometriosis progression, uterine/pelvic pathology progression and/or progression of endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in the subject, wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
In embodiments, a patient suffering from endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain is monitored to determine if the amount or concentration of EphA1 is changing over time in a sample of the patient. In particular, a patient suffering from endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain is monitored to determine if the amount or concentration of EphA1 is increasing, decreasing or not changing over time. In embodiments, a patient suffering from endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain is monitored if a decreased amount of EphA1 in the sample of the patient is determined.
In one embodiment the method is an in vitro method.
In one embodiment the biological fluid sample is serum.
The method may be used to monitor the progression of any kind of endometriosis described herein.
The method may be used to monitor the progression of any kind of uterine/pelvic pathology described herein.
Typically, such monitoring methods are performed on subjects that have not yet been treated for endometriosis (i.e. , they have not previously received endometriosis treatment (therapy or surgery)), for uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain. Such subjects are described as “naive” subjects herein.
However, such monitoring methods also encompass methods performed on subjects that have already been treated (e.g. for endometriosis).
Monitoring the progression of endometriosis in a subject over time assists in the earliest possible identification of disease progression (e.g., a worsening in disease status or disease symptoms). Such monitoring naturally involves the taking of repeated samples over time. The method may therefore be repeated at one or more time intervals for a particular subject and the results compared to monitor the development, progression or improvement in
endometriosis of that subject over time, wherein a change in the amount of level of the biomarker tested for in the biological fluid sample (e.g., serum) is indicative of a change in the progression of the endometriosis in the subject.
Same applies for monitoring uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
Disease progression may be indicated by a decrease in the level of EphA1 detected over time when the results of two or more time intervals are compared for the same subject. A “decrease” in the level of EphA1 encompasses no detection of EphA1 (i.e. , it is not present at detectable levels) at a later time interval when EphA1 was detected when the method was performed previously (i.e. at an earlier time interval) on the same subject (and an equivalent biological fluid sample type).
In other words, if the method is performed a plurality of times, disease progression may be indicated when the level of EphA1 detected at the later time interval(s) is equal or lower than that detected at the earlier time interval(s).
Suitable time intervals for monitoring disease progression can easily be identified by a person of skill in the art and will depend on the specific form of endometriosis being monitored. As a non-limiting example, the method may be repeated at least every week, month, six months, or at least every year, or whenever clinically needed, i.e., in case of a significant change in endometriosis symptoms.
Details of the biomarkers, combinations, samples, methods steps, subjects, types of endometriosis, treatments, reference values, etc are provided elsewhere and apply equally to this and all the other aspects.
Methods for determining the therapeutic effect of a treatment regimen for endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject
In a fifth aspect the present invention relates to a method for determining the therapeutic effect of a treatment regimen for endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject, the method comprising the steps of: i. determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject in accordance with method steps a) to b) as described above herein, ii. repeating step i. using a biological fluid sample obtained from the subject during or after treatment for a time interval; and iii. comparing levels of the extracellular protein part of EphA1 identified in i. with the levels of extracellular protein part of EphA1 identified in ii., and identifying that the treatment regimen has a therapeutic effect if the level of the extracellular protein part of EphA1 increased after treatment, wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
In one example, the change in level of EphA1 that is indicative of a therapeutic effect is an increase in EphA1 level after treatment. An “increase” in the level of EphA1 encompasses the detection of EphA1 at a later time interval when no EphA1 was detected (i.e., it is not present at detectable levels) when the method was performed previously (i.e. at an earlier time interval) on the same subject (and an equivalent biological fluid sample type).
In one embodiment the method is an in vitro method.
In one embodiment the biological fluid sample is serum.
Step i. may first be performed in accordance with the method using a biological fluid sample that was obtained from the subject at a time point before the treatment regimen for endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-
associated neuropathic pain began. Alternatively, step i. may first be performed using a biological fluid sample that was obtained from the subject at the same time as commencing the treatment regimen, or at a time point after the treatment regimen for endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain began. The method can therefore be used to determine the therapeutic effect of a treatment regimen for endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain from the outset (i.e., from the start of the regimen) or from a time point after the treatment regimen has started (i.e., determining the therapeutic effect of a treatment regimen for endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain during the treatment regimen itself).
In embodiments, an unaltered or decreasing amount or concentration of EphA1 in a sample of the subject being treated for endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain is indicative of the therapy being ineffective, i.e., an unaltered or decreasing amount or concentration of EphA1 in a sample of the subject being treated for endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain is indicative of persisting or recurring endometriosis. In particular, the treatment for endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain is ineffective if the amount of EphA1 is decreasing to 50% or less. In particular, the treatment for endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain is ineffective if the amount of EphA1 is decreasing to 100%.
Alternatively, in the case of endometriosis an unaltered level of EphA1 could either mean that the disease stagnates or that it has been progressed from stage I to stages II, III or IV where the EphA1 levels are comparable to stage I.
An improvement in disease status or symptoms (e.g., over a treatment period) may also be indicated by stabilized levels of EphA1 over time (compared to the level of EphA1 observed in the absence of treatment over the equivalent time period or compared to equivalent controls). In other cases EphA1 levels will be increased if the disease itself is improved; this
may also encompass improvement of symptoms such as pelvic pain or neuropathic pain associated with endometriosis or other uterine/pelvic pathology.
A treatment regimen may be identified as having a therapeutic effect if it results in a delay in disease progression or a delay in the development of symptoms (e.g., over a treatment period).
A treatment regimen may also be identified as having a therapeutic effect if it results in an improvement in disease status or symptoms (e.g., over a treatment period). Methods for determining if the treatment regimen has a therapeutic effect are well known in the art.
A treatment period refers to a time interval over which treatment occurs (e.g., 1 month, 3 months, 6 months, 1 year, 2 years, etc).
As would be clear to a person of skill in the art, the direction of change in EphA1 levels that is indicative of a therapeutic effect may depend on the disease status of the subject prior to treatment and the control/reference used.
The change in level of EphA1 can also be indicative of compliance or adherence with the prescribed treatment after treatment.
The trends for identifying that the subject has complied or adhered with the prescribed treatment regimen are equivalent to those described in detail above in respect of determining the therapeutic effect of a treatment regimen for endometriosis. This is because a “prescribed treatment regimen” is a recommended treatment regimen and therefore typically has a therapeutic effect (and thus, observation of the therapeutic effect on the biomarker levels is an indication of subject compliance or adherence with the prescribed treatment regimen).
In embodiments, the subject is monitored several times at different time points. In embodiments, the patient is monitored several times within a time frame of weeks, months, or years. In particular embodiments, a subject is monitored is once a month or once a year. In embodiments, a subject suffering from endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain is monitored once a month or once a year after diagnosis of endometriosis, uterine/pelvic pathology, or
endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain. In embodiments, a subject being treated for endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain is monitored once after therapy, in particular once after surgical therapy. In particular, the subject being treated for endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain is monitored once a month or once a year to determine the efficacy of treatment and/or the recurrence of endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
The method can also be useful as a screening tool for determining if specific regimens or treatment modalities have a therapeutic effect on endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain. The tested regimens or treatment modalities may be new regimens or treatment modalities, modified regimens or treatment modalities, or known regimens or treatment modalities that need further testing. In this context, a treatment modality is e.g., a drug or medicament that is useful or suspected to be useful in the treatment of endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
In embodiments, therapy of endometriosis is selected from the group consisting of drug-based therapy or surgical therapy. In embodiments the treatment regimen comprises surgical therapy, radiotherapy, immunotherapy, hormone therapy, ultrasound therapy, or combinations thereof. In preferred embodiments, surgical therapy of endometriosis is laparoscopy or nerve sparing surgery. In embodiments, drug-based therapy of endometriosis is inhibiting or targeting neurogenic inflammation and/or pain medication and/or hormonal therapy.
Same applies for uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain.
In particular embodiments, therapy is adapted if an unaltered or decreasing amount or concentration of EphA1 in a sample of the patient being treated for endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain is determined.
Details of the biomarkers, combinations, samples, methods steps, subjects, types of endometriosis, treatments, etc are provided elsewhere and apply equally to this aspect.
Accordingly, all aspects described in detail above for methods for assessing and/or monitoring endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain apply equally here.
Computer-implemented method for assessing a patient with endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain
In a further aspect the present invention relates to a computer-implemented method for assessing a patient with suspected endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain comprising the steps of: a) receiving a value for level of a first biomarker in a biological fluid sample of the subject, said first biomarker being the extracellular protein part of EphA1 ; b) receiving a value for the level of a second biomarker in a sample of the subject, wherein said second biomarker is CA125, c) receiving a value for the level of dysmenorrhea according to the VAS and/or lower abdominal pain according to the VAS, d) comparing the values for the levels of steps (a) - (c) to references for said biomarkers and the amount of dysmenorrhea and/or calculating a score for assessing the subject with suspected endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain based on the levels of the biomarkers and the amount of dysmenorrhea; and e) assessing said subject based on the comparison and/or the calculation made in step (d), wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
The term “computer-implemented” as used herein means that the method is carried out in an automated fashion on a data processing unit which is, typically, comprised in a computer or similar data processing device. The data processing unit shall receive values for the amount of the biomarkers. Such values can be the amounts, relative amounts or any other calculated value reflecting the amount as described elsewhere herein in detail. Accordingly, it is to be understood that the aforementioned method does not require the determination of amounts for the biomarkers but rather uses values for already predetermined amounts.
The present invention also, in principle, contemplates a computer program, computer program product or computer readable storage medium having tangibly embedded said computer program, wherein the computer program comprises instructions which, when run on a data processing device or computer, carry out the method of the present invention as specified above. Specifically, the present disclosure further encompasses: a computer or computer network comprising at least one processor, wherein the processor is adapted to perform the method according to one of the embodiments described in this description, a computer loadable data structure that is adapted to perform the method according to one of the embodiments described in this description while the data structure is being executed on a computer, a computer script, wherein the computer program is adapted to perform the method according to one of the embodiments described in this description while the program is being executed on a computer, a computer program comprising program means for performing the method according to one of the embodiments described in this description while the computer program is being executed on a computer or on a computer network, a computer program comprising program means according to the preceding embodiment, wherein the program means are stored on a storage medium readable to a computer, a storage medium, wherein a data structure is stored on the storage medium and wherein the data structure is adapted to perform the method according to one of the embodiments described in this description after having been loaded into a main and/or working storage of a computer or of a computer network,
a computer program product having program code means, wherein the program code means can be stored or are stored on a storage medium, for performing the method according to one of the embodiments described in this description, if the program code means are executed on a computer or on a computer network, a data stream signal, typically encrypted, comprising a data for parameters as defined herein elsewhere, and a data stream signal, typically encrypted, comprising the assessment provided by the methods of the present invention.
Combination with CA- 125, symptoms or clinical data
The methods according to the present invention can be combined with other tests, biomarkers, clinical data or further information that is useful to diagnose or classify endometriosis in order to obtain the most reliable result.
Despite its rather weak diagnosis performance CA-125 is routinely used as a biomarker for endometriosis. Accordingly, it may be advantageous to combine determining the level of EphA1 with CA-125 obtained from the subject in the context of the methods described herein.
Besides CA-125 other symptoms or clinical data that is used for diagnosing or classifying endometriosis can be used in combination with the determination of EphA1 levels. Such symptoms or clinical data can be but are not limited to age, dysmenorrhea, abdominal pain, or other biomarkers.
Kits and devices
In another aspect, kits are provided for diagnosing or classifying the stage of endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject. The kits include reagents suitable for determining levels of a plurality of analytes in a test sample (e.g., reagents suitable for determining levels of the biomarker disclosed herein).
The kits described herein typically comprise a detectably labelled agent that specifically binds to EphA1 protein.
Such a kit may additionally comprise a detectably labelled agent that specifically binds to CA- 125.
The kits described herein can take on a variety of forms. Typically, the kits will include reagents suitable for determining levels of a plurality of biomarkers (for example EphA1 and optionally CA-125) in a sample.
Optionally, the kits may contain one or more control samples or references. Typically, a comparison between the levels of the biomarkers in the subject and levels of the biomarkers in the control samples is indicative of a clinical status (e.g., diagnosis of endometriosis). Also, the kits, in some cases, will include written information (indicia) providing a reference (e.g., pre-determined values), wherein a comparison between the levels of the biomarker in the subject and the reference (pre- determined values) is indicative of a clinical status (e.g., diagnosis of endometriosis). In some cases, the kits comprise software useful for comparing biomarker levels or occurrences with a reference (e.g., a prediction model). Usually, the software will be provided in a computer readable format such as a compact disc, but it also may be available for downloading via the internet. However, the kits are not so limited and other variations with will be apparent to one of ordinary skill in the art.
The components of the kit may be housed in a container that is suitable for transportation. Details on the biomarker is given above and apply equally here. Suitably, the biomarker may be protein.
In some examples the kits include the detectably labelled agent(s) on a continuous (e.g., solid) surface, such as a lateral flow surface. Alternatively, in examples comprising more than one detectably labelled agent, the detectably labelled agent(s) may be located in distinct (i.e., spatially separate) zones on a (e.g., solid) surface, such as a multiwall micro-titre plate (e.g., for an ELISA assay). Other appropriate surfaces and containers that are well known in the art may also form part of the kits described herein.
In one example, the kit further comprises one or more reagents for detecting the detectably labelled agent. Suitable reagents are well known in the art and include but are not limited to
standard reagents and buffers required to perform any one of the appropriate detection methods that may be used (and are well known in the art).
In one example, the kit comprises one or more of the following: a multi-well plate, ball bearing(s), extraction buffer, extraction bottle and a lateral flow device lateral flow device.
An assay device is also provided for diagnosing endometriosis, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject.
Typically, the device comprises a surface with at least one detectably labelled agent located thereon that specifically binds to EphA1 protein.
Such a device may additionally comprise a detectably labelled agent that specifically binds to CA-125.
If two detectably labelled agents are used, they may be located in separate zones on the surface. In other words, the two detectably labelled agents may be located in distinct (i.e. , spatially separate) zones on a (e.g., solid) surface, such as a multiwell micro-titre plate. Detectably labelled agent(s) that specifically bind to the biomarker(s) of interest are described in detail elsewhere herein.
The assay device comprises a surface upon which the detectably labelled agents are located. Appropriate surfaces include a continuous (e.g., solid) surface, such as a lateral flow surface, a dot blot surface, a dipstick surface or a surface suitable for performing surface plasmon resonance. Other appropriate surfaces include microtitre plates, multi-well plates etc. Other appropriate surfaces that are well known in the art may also form part of the assay device described herein.
Appropriate assay device formats therefore include but are not limited to device formats suitable for performing any one of lateral flow, dot blot, ELISA, or surface plasmon resonance assays for detecting the presence, level or absence of the biomarker of interest.
Data storage aspects
Biomarker levels and/or reference levels may be stored in a suitable data storage medium (e.g., a database) and are, thus, also available for future diagnoses. This also allows efficiently diagnosing prevalence for a disease because suitable reference results can be identified in the database once it has been confirmed (in the future) that the subject from which the corresponding reference sample was obtained did have endometriosis. As used herein a "database" comprises data collected (e.g., analyte and/or reference level information and /or patient information) on a suitable storage medium. Moreover, the database, may further comprise a database management system. The database management system is, preferably, a network-based, hierarchical or object-oriented database management system. Furthermore, the database may be a federal or integrated database. More preferably, the database will be implemented as a distributed (federal) system, e.g., as a Client-Server-System. More preferably, the database is structured as to allow a search algorithm to compare a test data set with the data sets comprised by the data collection. Specifically, by using such an algorithm, the database can be searched for similar or identical data sets being indicative of endometriosis (e.g., a query search). Thus, if an identical or similar data set can be identified in the data collection, the test data set will be associated with endometriosis. Consequently, the information obtained from the data collection can be used to diagnose endometriosis or based on a test data set obtained from a subject. More preferably, the data collection comprises characteristic values of all analytes comprised by any one of the groups recited above.
The methods described herein may further include communication of the results or diagnoses (or both) to technicians, physicians or patients, for example. In certain examples, computers will be used to communicate results or diagnoses (or both) to interested parties, e.g., physicians and their patients.
In some examples, the results or diagnoses (or both) are communicated to the subject as soon as possible after the diagnosis is obtained. The results or diagnoses (or both) may be communicated to the subject by the subject's treating physician. Alternatively, the results or diagnoses (or both) may be sent to a subject by email or communicated to the subject by phone. A computer may be used to communicate the results or diagnoses by email or phone. In certain examples, the message containing results or diagnoses may be generated and delivered automatically to the subject using a combination of computer hardware and software which will be familiar to artisans skilled in telecommunications.
Uses
Also provided herein is the use of the biomarker EphA1 as a biological fluid biomarker for endometriosis, in particular the early stages, uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
In a preferred example EphA1 may be used as biomarker for endometriosis generally. In this context “endometriosis generally” refers to all forms of endometriosis, including but not limited to peritoneal endometriosis, endometrioma, and deep infiltrating endometriosis.
EphA1 may also be combined with CA-125.
In a preferred example EphA1 may be used as biomarker for uterine/pelvic pathology, or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain.
Details of the biomarkers, samples, methods, subjects, types of endometriosis etc are provided elsewhere and apply equally to this aspect.
Companion diagnostic
The methods kits, assay devices and uses provided herein may be used as part of a companion diagnostic e.g., as part of a medical device, often an in vitro device, which provides information that is essential for the safe and effective use of a corresponding drug or biological product (wherein the corresponding drug or biological product is for treating or preventing endometriosis).
In further embodiments, the present invention relates to the following aspects, wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM):
An in vitro method for diagnosing endometriosis in a subject, the method comprising the steps of: a) determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject, b) comparing the level of the extracellular protein part of EphA1 to at least one appropriate reference value of a level of the extracellular protein part of EphA1, c) identifying a subject as having endometriosis if the comparison in step b) indicates that the subject has a decreased level of the extracellular protein part of EphA1 compared to the appropriate reference value. The method according to aspect 1 , wherein the at least one appropriate reference value is i. a level of EphA1 in a non-pathological subject or in a symptomatic subject, or is ii. an average level of EphA1 in a group of non-pathological subjects or in a group of symptomatic subjects, or a combination thereof, or wherein the at least one appropriate reference value is iii. a predetermined value of a level of EphA1 in a non-pathological subject or symptomatic subject, or is iv. a predetermined average value of a level of EphA1 in a group of non- pathological subjects or in a group of symptomatic subjects, or a combination thereof. The method according to aspect 1 or 2, wherein the at least one appropriate reference value is
• a level of EphA1 in a non-pathological subject, in a symptomatic subject, or in a subject that has stage I, stage II, stage III or stage IV endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM), or is
• an average level of EphA1 in a group of subjects that are non-pathological subjects, that are symptomatic subjects, or a combination thereof, or in a group of subjects that have stage I, stage II, stage III or stage IV
endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM), or wherein the at least one appropriate reference value is
• a predetermined value of a level of EphA1 in a non-pathological subject or in a symptomatic subject, or in a subject that has stage I, stage II, stage III or stage IV endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM), or is
• a predetermined average value of a level of EphA1 in a group of non- pathological subjects, that are symptomatic subjects, or a combination thereof, or in a group of subjects that have stage I, stage II, stage III or stage IV endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM). The method according to any preceding claim, wherein the biological fluid sample is blood, serum, plasma, capillary blood, interstitial fluid, peritoneal fluid, or menstrual fluid, preferably the biological fluid sample is serum. The method according to any preceding aspect, wherein the subject is a human, preferably a female human. The method according to any preceding aspect, wherein the protein level of EphA1 , in particular its extracellular protein part, is determined, optionally using a process selected from: ELISA assay, immunoblotting, lateral flow assay, protein microarray and mass spectrometry. The method according to any preceding aspect, wherein the extracellular protein part of EphA1 is detected by mass spectrometry. The method according to any preceding aspect, wherein the extracellular protein part of EphA1 is detected by identification of at least one of the following
peptides: WEPPADTGGR (SEQ ID NO: 1), AQGELGWLLDPPK (SEQ ID NO: 2), MHCSPDGEWLVPVGR (SEQ ID NO: 3), GLYLAFHNPGACVALVSVR (SEQ ID NO: 4), APGEGPQVACTGPPSAPR (SEQ ID NO: 5), VTTVAADQSFTIR (SEQ ID NO: 6), or VHVELQFTVR (SEQ ID NO: 7).
9. The method according to any preceding aspect, wherein endometriosis is selected from the group consisting of peritoneal endometriosis, endometrioma, and deep infiltrating endometriosis.
10. The method of any preceding aspect, further comprising selecting a treatment regimen for the subject based on the comparison of the level of the extracellular protein part of EphAlwith the control sample or with the pre-determined reference level.
11. The method according to aspect 10, further comprising administering the selected treatment regimen to the subject, optionally wherein the selected treatment regimen comprises drug-based therapy and/or surgical treatment (laparoscopy) or combinations thereof.
12. An in vitro method for monitoring endometriosis progression in a subject, the method comprising the steps of: i. determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject in accordance with method steps a) to b) of any one of aspects 1 to 13, ii. repeating step i. using a biological fluid sample obtained from the subject during or after treatment for a time interval; and iii. comparing the levels of the extracellular protein part of EphA1 identified in i. with the levels of extracellular protein part of EphA1 identified in ii., wherein a change in the levels of extracellular protein part of EphA1 from i. to ii. is indicative of a change in endometriosis progression in the subject.
An in vitro method for determining the therapeutic effect of a treatment regimen for endometriosis in a subject, the method comprising the steps of: i. determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject in accordance with method steps a) to b) of any one of aspects 1 to 13, ii. repeating step i. using a biological fluid sample obtained from the subject during or after treatment for a time interval; and iii. comparing levels of the extracellular protein part of EphA1 identified in i. with the levels of extracellular protein part of EphA1 identified in ii., and identifying that the treatment regimen has a therapeutic effect if the level of the extracellular protein part of EphA1 increased after treatment. The method according to any of the previous claims, further comprising the assessment of dysmenorrhea according to the Visual Analog Scale (VAS) and/or lower abdominal pain according to VAS. The method according to any of the previous aspects, further comprising determining the level of CA-125 in the biological fluid sample from the subject. The method according to aspect 15, comprising calculating
• a ratio of the amount or concentration the extracellular protein part of EphA1 and the amount or concentration of CA-125, or
• a ratio of the amount or concentration of the extracellular protein part of EphA1 and dysmenorrhea, or
• a ratio of the amount or concentration the extracellular protein part of EphA1 and the amount or concentration of CA-125 and dysmenorrhea, or
• a ratio of the amount or concentration the extracellular protein part of EphA1 and lower abdominal pain according to the VAS scale.
17. A computer-implemented method for assessing a patient with suspected endometriosis comprising the steps of: a) receiving a value for level of a first biomarker in a biological fluid sample of the subject, said first biomarker being the extracellular protein part of EphA1 ; b) receiving a value for the level of a second biomarker in a sample of the subject, wherein said second biomarker is CA125, c) receiving a value for the level of dysmenorrhea according to the VAS and/or lower abdominal pain according to the VAS, d) comparing the values for the levels of steps (a) - (c) to references for said biomarkers and the amount of dysmenorrhea and/or calculating a score for assessing the subject with suspected endometriosis based on the levels of the biomarkers and the amount of dysmenorrhea; and e) assessing said subject based on the comparison and/or the calculation made in step (d).
18. Use of elevated EphA1 levels in a biological fluid sample as a biomarker for endometriosis.
19. The use according to aspect 18, wherein the biological fluid sample is blood or blood-derived, preferably serum.
20. The use according to aspects 18 and 19, wherein the use is to diagnose and/or classify endometriosis.
21. A kit for diagnosing and/or classifying endometriosis in a subject, comprising at least one detectably labelled agent that specifically binds to EphA1 protein.
22. The kit according to aspect 21 , further comprising one or more reagents for detecting the detectably labelled agent.
23. An assay device for diagnosing and/or classifying endometriosis in a subject, the device comprising a surface with at least one detectably labelled agent located thereon that specifically binds to EphA1 protein.
Aspects of the invention are demonstrated by the following non-limiting examples. The following examples and figures are provided to aid the understanding of the present invention, the true scope of which is set forth in the appended claims. It is understood that modifications can be made in the procedures set forth without departing from the spirit of the invention.
Examples
Example 1 : Diagnostic performance of biomarker EphA1 in women with endometriosis
For the measurements, a total of 164 serum samples from human females were analyzed (for clinical data it is referred to the corresponding section herein). The concentration of the analytes was determined by advanced proteomics technology based on mass spectrometry. The case group is comprised of patients diagnosed with endometriosis (peritoneal endometriosis with or without endometrioma and/or deep infiltrating endometriosis; rASRM stages l-l V) or other uterine/pelvic pathologies (e.g. adenomyosis, fibroids, etc.) diagnosed by laparoscopic with subsequent histological confirmation, and the control group including women with symptoms but without endometriosis or other uterine/pelvic pathologies.
The concentration of EphA1 in human serum was determined advanced proteomics technology based on mass spectrometry.
Results are shown in Figure 1 .
Serum EphA1 levels are decreased in stages I to IV (as well as in other uterine/pelvic pathological finding; Case SOF) compared to control groups (symptomatic controls with no uterine and pelvic pathology “Ctrl SNF”; SNF=Symptomatic No Findings).
As shown in Figs. 2a and b EphA1 is decreased in serum samples of women with other uterine/pelvic pathological findings (Case SOF) compared to symptomatic control women
without endometriosis and without uterine and pelvic pathologies “Ctrl SNF”; SNF=Symptomatic No Findings).
As shown in Figs. 3a and b EphA1 is decreased in serum samples of women with early endometriosis rASRM stages l/ll “Case” compared to control women without endometriosis and without uterine and pelvic pathology (“Ctrl”).
As shown in Figs. 4a and b EphA1 is decreased in serum samples of women with early endometriosis rASRM stage I “Case” compared to women without endometriosis and without uterine and pelvic pathology (“Ctrl”).
As shown in Figs. 5a and b EphA1 is decreased in serum samples of women with early endometriosis rASRM stage II “Case” compared to control women without endometriosis and without uterine and pelvic pathology (“Ctrl”).
As shown in Figs. 6a and b EphA1 is decreased in serum samples of women with endometriosis stage III “Case” compared to control women without endometriosis and without uterine and pelvic pathology (“Ctrl”).
As shown in Figs. 7a and b EphA1 is decreased in serum samples of women with endometriosis rASRM stage IV “Case” compared to controls without endometriosis and without uterine and pelvic pathology (“Ctrl”).
In the following Table 2 the model performance is determined by looking at the area under the curve (AUC). The best possible AUC is 1 while the lowest possible is 0.5. Optimal cut-offs were selected using Youden’s index (maximized sum of sensitivity plus specificity - 1).
Table 2: The diagnostic performance of EphA1 biomarker to discriminate women with histologically confirmed endometriosis and women without endometriosis (controls) using receiver operator characteristic (ROC) analysis is shown describing the area under the curve (AUC) of the ROC analysis and the associated 95% confidence interval. N depicts the number of samples tested (cases plus controls, numbers in each group vary depending on the analyte).
Serum EphA1 can be used as a blood biomarker for early diagnosis and risk stratification of endometriosis and other uterine/pelvic pathology (such as adenomyosis, fibroids, ovarian cysts requiring surgery, uterine/pelvic cancer such as ovarian cancer or endometrial cancer).
Serum EphA1 can be used to select patients with rASRM disease stage I and stage II for early medical management of endometriosis. Therefore, it can significantly reduce the delay to the diagnosis of endometriosis in women with signs and symptoms of endometriosis (such as dysmenorrhea, pelvic pain, infertility), improve patients' lives, and reduce the economic burden.
Serum EphA1 can be used for therapy selection and monitoring as a potential companion diagnostics biomarker.
Example 2: Diagnostic performance of biomarker EphA1 compared to CA-125 in women with endometriosis
In a further experiment the performance of the biomarker EphA1 was compared to the current standard biomarker CA-125.
The concentration of CA-125 was determined by a cobas e 601 analyzer. Detection of CA 125 II with a cobas e 601 analyzer is based on the Elecsys® Electro- ChemiLuminescence (ECL) technology. In brief, biotin-labelled and ruthenium-labelled antibodies are combined with the respective amount of undiluted sample and incubated on the analyzer. Subsequently, streptavidin-coated magnetic microparticles are added and incubated on the instrument in order to facilitate binding of the biotin-labelled immunological complexes. After this incubation step the reaction mixture is transferred into the measuring cell where the beads are magnetically captured on the surface of an electrode. ProCell M Buffer containing
tripropylamine (TPA) for the subsequent ECL reaction is then introduced into the measuring cell in order to separate bound immunoassay complexes from the free remaining particles. Induction of voltage between the working and the counter electrode then initiates the reaction leading to emission of photons by the ruthenium complexes as well as TPA. The resulting electrochemiluminescent signal is recorded by a photomultiplier and converted into numeric values indicating concentration level of the respective analyte.
The results are shown in Figures 8a and b and 9a and b and in Table 3 below. Note that for CA-125, slightly more sample measurements were available in some subgroups.
Figs 8a and b and Figs 9a and b show CA-125 in serum samples of women with early endometriosis rASRM stage I (Fig. 8) and II (Fig. 9) “Case” compared to controls without endometriosis and without uterine and pelvic pathology “Ctrl”.
Box plots in Figs. 8a and b and 9a and b for CA-125 were generated for controls and for the endometriosis stages I and II using a cobas e 601 analyzer based on Elecsys® ElectroChemiLuminescence (ECL) technology.). The data are presented using box and whisker plots, including the median (middle quartile), the inter-quartile range (which represents the middle 50% of scores for the group), the upper quartile (75% of scores fall below the upper quartile), the lower quartile (25% of scores fall below the lower quartile). The whiskers show the 5th percentile and the 95th percentile, respectively.
As can be seen from a comparison of Tables 2 and 3 serum EphA1 shows better diagnosis performance for the detection of early stages of endometriosis (Stage I, Stage II) compared to the reference biomarker CA-125.
In the following Table 4 the performance of EphA1 as biomarker in combination with CA-125 is determined by looking at the area under the curve (AUC) for early stage endometriosis (Stage I and II), for late stage endometriosis (Stage III and IV) and for all stages (Stage I to IV).
Table 4: Diagnostic performance of serum EphA1 biomarker in combination with biomarker CA-125 in women with endometriosis and controls. The measurement of CA-125 was performed using a cobas e 601 analyzer based on Elecsys® ElectroChemiLuminescence (ECL) technology. Due to low sample numbers, bivariate analysis for the individual stages (I, II, III, IV, SOF) were not performed. Only samples with both EphA1 and CA-125 measurements available were used.
Serum EphA1 and serum CA-125 combined can be used as a blood biomarker for (early) diagnosis and risk stratification of endometriosis or other uterine/pelvic pathologies, in particular early diagnosis of rASRM stages 1 and 2 endometriosis. Serum EphA1 and serum CA-125 combined showed better identification of all cases (endometriosis and other uterine/pelvic pathologies) than the CA-125 reference biomarker alone.
Serum EphA1 and serum CA-125 combined can be used to select patients for early medical management (drug therapy or surgery) of endometriosis. Therefore, it can significantly reduce endometriosis diagnostic delay, improve patients' lives, and reduce the economic burden.
Serum EphA1 and serum CA-125 combined can be used for therapy selection and monitoring as a potential companion diagnostics biomarker.
Example 3: Clinical Data on cohort (Cases and Controls):
1) Endometriosis (or other uterine pathology) Cases:
No contraceptive hormones/devices: 54.31 %
Taking contraceptive hormones/devices: 45.69% b) Endometriosis stage I other uterine/pelvic pathology findings:
2) Controls: No Findings, Severe Pain (“Symptomatic, No endometriosis and No uterine and pelvic pathology”) a) Total cohort number: 48 patients; Hormonal Contraceptives/ Devices:
No contraceptive hormones/devices: 58.33%
Taking contraceptive hormones/devices: 37.50%
Example 4:
Materials and Methods:
Proteomics Discovery Method used for detection of EphA1
Human serum samples from women with pain symptoms and with endometriosis or other uterine/pelvic pathologies such as adenomyosis and fibroids (cases) and symptomatic women without endometriosis (symptomatic controls) were analyzed using advanced mass spectrometry (MS)-based proteomics including automated sample preparation procedures. Serum samples were blinded for clinical information and randomized for proteome analysis.
Samples were digested with Trypsin/Endoproteinase LysC to generate proteolytic peptides, which were subsequently subjected to high resolution liquid chromatography coupled to tandem mass spectrometry (LC-MS/MS) applying a data-independent acquisition method (DIA). Based on the batch design, an in-depth spectral library from 50 representatively selected samples across all six depletion batches was generated by use of advanced neural network based data analysis tools.
For basic quality control, the number of identified protein groups per sample and conducted principal component analysis (PCA) was determined, which did not show an obvious separation of samples.
Protein intensity data was corrected for experimental batch effects connected to sample preparation to enable a correct comparison of sample. Moreover, serum contamination analysis was performed and compared to previously visually determined hemolysis scales.
4.1 Sample preparation
Serum samples were shipped by Roche on dry ice and stored at -80°C. For experimental processing, samples were thawed on ice and centrifuged with 2,000 x g for 15 min at 4°C. For documentation, a hemolysis score was determined before the supernatants were transferred into new tubes. 7 pl serum per sample were subjected to depletion of 14 high-abundant proteins using High Select Top14 Abundant Protein Depletion Mini Spin Columns (Thermo Scientific) according to the manufacturer's instructions. The depleted, lyophilized plasma protein samples were subjected to enzymatic cleavage and peptide clean-up using an MS sample preparation kit according to the manufacturer's instructions. The workflow was performed in 96 well plates using the Agilent BRAVO automated liquid handling platform. Serum control samples were used for each sample preparation batch and were treated equally.
4.2 Mass spectrometric analysis
LC-MS/MS analysis was performed on an Orbitrap Exploris 480 mass spectrometer (Thermo Scientific) equipped with an Easy n-LC 1200 LIHPLC system (Thermo Scientific). Peptides were separated by a 125 min gradient before they were sprayed directly into the mass spectrometer using a nano-electrospray ion source (ProxeonBiosystems). The mass spectrometer was operated in the data independent mode (DIA) for single-shot analysis.
4.3 Data processing
All raw files acquired in this study were processed with the DIA-NN software suite (version 1.8.0) for peptide/protein identification and quantification using a curated Uniprot database (Swissprot and varsplic including protein isoforms, version 2020_03). The false discovery rate (FDR) for protein identifications was set to 1%. A spectral library (DIA Speclib) was generated from 50 single-shot DIA files with the “FASTA digest for library free search” option and “Deep Learning” was enabled for RT estimation. “Unrelated runs” was enabled to determine mass accuracy and retention time scan window for each run separately.
The inference of protein groups from DiaNN output was performed using a custom algorithm based on Nesvizhskii & Aebersold. For protein quantification the MaxLFQ algorithm from the DIA-NN R package (https://github.com/vdemichev/diann-rpackage) was applied and Iog10 intensities were subjected to median scaling. Protein intensity data has been corrected for experimental batch effects connected to sample preparation (“depletion batch”) to enable a correct comparison of samples using the function removeBatchEffect from the LIMMA R- package (10.18129/B9.bioc.limma). For the principal component analysis (PCA) only proteins that had no missing values were taken into account.
4.4 Calculation of serum contamination indices
Calculation of erythrocyte and platelet contamination indices was based on the list of gene names provided by Geyer et al. 2019. The mean of corrected Iog10 protein intensities for proteins in the list that were expressed in all samples was calculated for each sample.
Example 5: Structure of EphA1 protein
The EphA1 protein is composed of an Extracellular domain, a Transmembrane domain and an Intracellular domain. The Extracellular domain contains the Ephrin-binding domain or Ephrin Receptor Ligand-Binding Domain (LBD), the Cysteine-rich region (including Sushi domain and EGF-like domain), and the Fibronectin-type III repeats (FN1 and FN2). The T ransmembrane domain (TM) links the Extracellular domain and the Intracellular domain. The Intracellular domain is composed of the Juxtamembrane domain, the Kinase domain, and the SAM domain. Figure 10 shows the structure of the EphA1 protein (Figure adapted from Owens HE. Corrected Thesis. Characterisation of EphA1 and its potential role in Alzheimer’s Disease. 2019)
Claims
1. A method for assessing endometriosis in a subject, the method comprising the steps of: a) determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject, b) comparing the level of the extracellular protein part of EphA1 to at least one appropriate reference value of a level of the extracellular protein part of EphA1, c) identifying a subject as having endometriosis if the comparison in step b) indicates that the subject has a decreased level of the extracellular protein part of EphA1 compared to the appropriate reference value, wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
2. The method according to claim 1, wherein endometriosis is selected from the group consisting of peritoneal endometriosis, endometrioma, and deep infiltrating endometriosis.
3. A method for assessing uterine/pelvic pathology in a subject, the method comprising the steps of: a) determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject, b) comparing the level of the extracellular protein part of EphA1 to at least one appropriate reference value of a level of the extracellular protein part of EphA1, c) identifying a subject as having uterine/pelvic pathology if the comparison in step b) indicates that the subject has a decreased level of the extracellular protein part of EphA1 compared to the appropriate reference value.
4. The method according to claim 3, wherein uterine/pelvic pathology is selected from the group consisting of adenomyosis, fibroids, ovarian cysts requiring surgery and uterine/pelvic cancer.
5. A method for assessing endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in a subject, the method comprising the steps of: a) determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject, b) comparing the level of the extracellular protein part of EphA1 to at least one appropriate reference value of a level of the extracellular protein part of EphA1 , c) identifying a subject as having endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain if the comparison in step b) indicates that the subject has a decreased level of the extracellular protein part of EphA1 compared to the appropriate reference value, wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
6. The method according to any preceding claim, wherein the biological fluid sample is blood, serum, plasma, capillary blood, interstitial fluid, peritoneal fluid, or menstrual fluid, preferably the biological fluid sample is serum.
7. The method of any preceding claim, further comprising selecting a treatment regimen for the subject based on the comparison of the level of the extracellular protein part of EphAlwith the control sample or with the pre-determined reference level.
8. The method of any preceding claim , further comprising administering the selected treatment regimen to the subject, optionally wherein the selected treatment regimen comprises drug-based therapy and/or surgical treatment (laparoscopy).
9. A method for monitoring endometriosis progression, uterine/pelvic pathology progression and/or progression of endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain in a subject, the method comprising the steps of: i. determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject in accordance with method steps a) to b) of any one of claims 1 to 8, ii. repeating step i. using a biological fluid sample obtained from the subject during or after treatment for a time interval; and iii. comparing the levels of the extracellular protein part of EphA1 identified in i. with the levels of extracellular protein part of EphA1 identified in ii., wherein a change in the levels of extracellular protein part of EphA1 from i. to ii. is indicative of a change in endometriosis progression, uterine/pelvic pathology progression and/or progression of endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain in the subject, wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
10. A method for determining the therapeutic effect of a treatment regimen for endometriosis, uterine/pelvic pathology and/or endometriosis and/or uterine/pelvic pathology associated neuropathic pain in a subject, the method comprising the steps of: i. determining the level of the extracellular protein part of EphA1 in a biological fluid sample from the subject in accordance with method steps a) to b) of any one of claims 1 to 8, ii. repeating step i. using a biological fluid sample obtained from the subject during or after treatment for a time interval; and iii. comparing levels of the extracellular protein part of EphA1 identified in i. with the levels of extracellular protein part of EphA1 identified in ii., and identifying that the treatment regimen has a therapeutic effect if the level of the extracellular protein part of EphA1 increased after treatment,
wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
11. The method according to any of the previous claims, further comprising the assessment of dysmenorrhea according to the Visual Analog Scale ( AS) and/or lower abdominal pain according to VAS.
12. The method according to any of the previous claims, further comprising determining the level of CA-125 in the biological fluid sample from the subject.
13. The method according to claim 12, comprising calculating
• a ratio of the amount or concentration the extracellular protein part of EphA1 and the amount or concentration of CA-125, or
• a ratio of the amount or concentration of the extracellular protein part of EphA1 and dysmenorrhea, or
• a ratio of the amount or concentration the extracellular protein part of EphA1 and the amount or concentration of CA-125 and dysmenorrhea, or
• a ratio of the amount or concentration the extracellular protein part of EphA1 and lower abdominal pain according to the VAS scale.
14. A computer-implemented method for assessing a patient with suspected endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology- associated neuropathic pain comprising the steps of: a) receiving a value for level of a first biomarker in a biological fluid sample of the subject, said first biomarker being the extracellular protein part of EphA1 ; b) receiving a value for the level of a second biomarker in a sample of the subject, wherein said second biomarker is CA125, c) receiving a value for the level of dysmenorrhea according to the VAS and/or lower abdominal pain according to the VAS, d) comparing the values for the levels of steps (a) - (c) to references for said biomarkers and the amount of dysmenorrhea and/or calculating a score for
assessing the subject with suspected endometriosis, uterine/pelvic pathology and/or endometriosis- and/or uterine/pelvic pathology-associated neuropathic pain based on the levels of the biomarkers and the amount of dysmenorrhea; and e) assessing said subject based on the comparison and/or the calculation made in step (d), wherein the subject’s stage of endometriosis is classified as stage I or stage II endometriosis according to the revised scoring system of the American Society for Reproductive Medicine (r-ASRM).
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23188461.0 | 2023-07-28 | ||
| EP23188461 | 2023-07-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025026908A1 true WO2025026908A1 (en) | 2025-02-06 |
Family
ID=87519981
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/071268 Pending WO2025026908A1 (en) | 2023-07-28 | 2024-07-26 | Serum epha1 as biomarker for endometriosis |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025026908A1 (en) |
Citations (33)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1987006706A1 (en) | 1986-04-30 | 1987-11-05 | Igen, Inc. | Electrochemiluminescent assays |
| WO1990005301A1 (en) | 1988-11-03 | 1990-05-17 | Igen, Inc. | Electrochemiluminescent assays |
| WO1990005296A1 (en) | 1988-11-03 | 1990-05-17 | Igen, Inc. | Electrochemiluminescent reaction utilizing amine-derived reductant |
| WO1992014139A1 (en) | 1991-02-06 | 1992-08-20 | Igen, Inc. | Methods and apparatus for improved luminescence assays |
| US5221605A (en) | 1984-10-31 | 1993-06-22 | Igen, Inc. | Luminescent metal chelate labels and means for detection |
| US5316757A (en) | 1984-10-18 | 1994-05-31 | Board Of Regents, The University Of Texas System | Synthesis of polyazamacrocycles with more than one type of side-chain chelating groups |
| US5342606A (en) | 1984-10-18 | 1994-08-30 | Board Of Regents, The University Of Texas System | Polyazamacrocyclic compounds for complexation of metal ions |
| US5385893A (en) | 1993-05-06 | 1995-01-31 | The Dow Chemical Company | Tricyclopolyazamacrocyclophosphonic acids, complexes and derivatives thereof, for use as contrast agents |
| WO1995008644A1 (en) | 1993-09-22 | 1995-03-30 | Igen, Inc. | Self-sustained sequence replication electrochemiluminescent nucleic acid assay |
| US5428139A (en) | 1991-12-10 | 1995-06-27 | The Dow Chemical Company | Bicyclopolyazamacrocyclophosphonic acid complexes for use as radiopharmaceuticals |
| US5462725A (en) | 1993-05-06 | 1995-10-31 | The Dow Chemical Company | 2-pyridylmethylenepolyazamacrocyclophosphonic acids, complexes and derivatives thereof, for use as contrast agents |
| US5480990A (en) | 1991-12-10 | 1996-01-02 | The Dow Chemical Company | Bicyclopolyazamacrocyclocarboxylic acid complexes for use as contrast agents |
| WO1996006946A1 (en) | 1994-08-26 | 1996-03-07 | Igen, Inc. | Biosensor for and method of electrogenerated chemiluminescent detection of nucleic acid adsorbed to a solid surface |
| WO1996024690A1 (en) | 1995-02-09 | 1996-08-15 | Igen, Inc. | Electrogenerated chemiluminescence labels for analysis and/or referencing |
| WO1996033411A1 (en) | 1995-04-18 | 1996-10-24 | Igen, Inc. | Electrochemiluminescence of rare earth metal chelates |
| WO1996039534A1 (en) | 1995-06-06 | 1996-12-12 | Igen, Inc. | Electrochemiluminescent enzyme biosensors |
| WO1996040978A1 (en) | 1995-06-07 | 1996-12-19 | Igen, Inc. | Electrochemiluminescent monitoring of compounds |
| WO1996041175A1 (en) | 1995-06-07 | 1996-12-19 | Igen, Inc. | Electrochemiluminescent enzyme immunoassay |
| US5591581A (en) | 1986-04-30 | 1997-01-07 | Igen, Inc. | Electrochemiluminescent rhenium moieties and methods for their use |
| US5597910A (en) | 1991-12-11 | 1997-01-28 | Igen, Inc. | Electrochemiluminescent label for DNA probe assays |
| US5679519A (en) | 1995-05-09 | 1997-10-21 | Oprandy; John J. | Multi-label complex for enhanced sensitivity in electrochemiluminescence assay |
| US5739294A (en) | 1991-12-10 | 1998-04-14 | The Dow Chemical Company | Bicyclopol yazamacrocyclophosphonic acid complexes for use as contrast agents |
| US5834461A (en) | 1993-07-29 | 1998-11-10 | American Cyanamid Company | Tricyclic benzazepine vasopressin antagonists |
| WO2005012359A2 (en) | 2003-08-01 | 2005-02-10 | Genentech, Inc. | Anti-vegf antibodies |
| US20100111861A1 (en) | 2008-10-31 | 2010-05-06 | Lili Liu | Detection and quantification of abasic site formation in vivo |
| WO2012107419A1 (en) | 2011-02-09 | 2012-08-16 | Roche Diagnostics Gmbh | New iridium-based complexes for ecl |
| US9503190B2 (en) | 2012-06-28 | 2016-11-22 | Chunghwa Telecom Co., Ltd. | Client-side dynamic multi-routing power distribution system of FTTx optical terminal equipment |
| US9606763B2 (en) | 2013-12-07 | 2017-03-28 | Lenovo (Singapore) Pte Ltd | Folding electronic device |
| US9703653B2 (en) | 2012-12-12 | 2017-07-11 | Kabushiki Kaisha Toshiba | Cloud system management apparatus, cloud system, reallocation method, and computer program product |
| US9716942B2 (en) | 2015-12-22 | 2017-07-25 | Bose Corporation | Mitigating effects of cavity resonance in speakers |
| US20220221472A1 (en) * | 2019-04-26 | 2022-07-14 | Chugai Seiyaku Kabushiki Kaisha | Method for diagnosing endometriosis, disease state monitoring method, and kit |
| WO2022207628A1 (en) * | 2021-03-30 | 2022-10-06 | F. Hoffmann-La Roche Ag | Scf as blood biomarker for the non-invasive diagnosis of endometriosis |
| WO2022243210A1 (en) * | 2021-05-17 | 2022-11-24 | F. Hoffmann-La Roche Ag | sFRP4 AS BLOOD BIOMARKER FOR THE NON-INVASIVE DIAGNOSIS OF ADENOMYOSIS |
-
2024
- 2024-07-26 WO PCT/EP2024/071268 patent/WO2025026908A1/en active Pending
Patent Citations (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5428155A (en) | 1984-10-18 | 1995-06-27 | Board Of Regents, The University Of Texas System | Synthesis of polyazamacrocycles with more than one type of side-chain chelating groups |
| US5342606A (en) | 1984-10-18 | 1994-08-30 | Board Of Regents, The University Of Texas System | Polyazamacrocyclic compounds for complexation of metal ions |
| US5316757A (en) | 1984-10-18 | 1994-05-31 | Board Of Regents, The University Of Texas System | Synthesis of polyazamacrocycles with more than one type of side-chain chelating groups |
| US5221605A (en) | 1984-10-31 | 1993-06-22 | Igen, Inc. | Luminescent metal chelate labels and means for detection |
| WO1987006706A1 (en) | 1986-04-30 | 1987-11-05 | Igen, Inc. | Electrochemiluminescent assays |
| US5591581A (en) | 1986-04-30 | 1997-01-07 | Igen, Inc. | Electrochemiluminescent rhenium moieties and methods for their use |
| WO1990005296A1 (en) | 1988-11-03 | 1990-05-17 | Igen, Inc. | Electrochemiluminescent reaction utilizing amine-derived reductant |
| WO1990005301A1 (en) | 1988-11-03 | 1990-05-17 | Igen, Inc. | Electrochemiluminescent assays |
| WO1992014139A1 (en) | 1991-02-06 | 1992-08-20 | Igen, Inc. | Methods and apparatus for improved luminescence assays |
| US5428139A (en) | 1991-12-10 | 1995-06-27 | The Dow Chemical Company | Bicyclopolyazamacrocyclophosphonic acid complexes for use as radiopharmaceuticals |
| US5480990A (en) | 1991-12-10 | 1996-01-02 | The Dow Chemical Company | Bicyclopolyazamacrocyclocarboxylic acid complexes for use as contrast agents |
| US5750660A (en) | 1991-12-10 | 1998-05-12 | The Dow Chemical Company | Bicyclopolyazamacrocyclophosphonic acid half esters |
| US5739294A (en) | 1991-12-10 | 1998-04-14 | The Dow Chemical Company | Bicyclopol yazamacrocyclophosphonic acid complexes for use as contrast agents |
| US5597910A (en) | 1991-12-11 | 1997-01-28 | Igen, Inc. | Electrochemiluminescent label for DNA probe assays |
| US5385893A (en) | 1993-05-06 | 1995-01-31 | The Dow Chemical Company | Tricyclopolyazamacrocyclophosphonic acids, complexes and derivatives thereof, for use as contrast agents |
| US5462725A (en) | 1993-05-06 | 1995-10-31 | The Dow Chemical Company | 2-pyridylmethylenepolyazamacrocyclophosphonic acids, complexes and derivatives thereof, for use as contrast agents |
| US5834461A (en) | 1993-07-29 | 1998-11-10 | American Cyanamid Company | Tricyclic benzazepine vasopressin antagonists |
| WO1995008644A1 (en) | 1993-09-22 | 1995-03-30 | Igen, Inc. | Self-sustained sequence replication electrochemiluminescent nucleic acid assay |
| WO1996006946A1 (en) | 1994-08-26 | 1996-03-07 | Igen, Inc. | Biosensor for and method of electrogenerated chemiluminescent detection of nucleic acid adsorbed to a solid surface |
| WO1996024690A1 (en) | 1995-02-09 | 1996-08-15 | Igen, Inc. | Electrogenerated chemiluminescence labels for analysis and/or referencing |
| WO1996033411A1 (en) | 1995-04-18 | 1996-10-24 | Igen, Inc. | Electrochemiluminescence of rare earth metal chelates |
| US5679519A (en) | 1995-05-09 | 1997-10-21 | Oprandy; John J. | Multi-label complex for enhanced sensitivity in electrochemiluminescence assay |
| WO1996039534A1 (en) | 1995-06-06 | 1996-12-12 | Igen, Inc. | Electrochemiluminescent enzyme biosensors |
| WO1996041175A1 (en) | 1995-06-07 | 1996-12-19 | Igen, Inc. | Electrochemiluminescent enzyme immunoassay |
| WO1996040978A1 (en) | 1995-06-07 | 1996-12-19 | Igen, Inc. | Electrochemiluminescent monitoring of compounds |
| WO2005012359A2 (en) | 2003-08-01 | 2005-02-10 | Genentech, Inc. | Anti-vegf antibodies |
| US20100111861A1 (en) | 2008-10-31 | 2010-05-06 | Lili Liu | Detection and quantification of abasic site formation in vivo |
| WO2012107419A1 (en) | 2011-02-09 | 2012-08-16 | Roche Diagnostics Gmbh | New iridium-based complexes for ecl |
| US9503190B2 (en) | 2012-06-28 | 2016-11-22 | Chunghwa Telecom Co., Ltd. | Client-side dynamic multi-routing power distribution system of FTTx optical terminal equipment |
| US9703653B2 (en) | 2012-12-12 | 2017-07-11 | Kabushiki Kaisha Toshiba | Cloud system management apparatus, cloud system, reallocation method, and computer program product |
| US9606763B2 (en) | 2013-12-07 | 2017-03-28 | Lenovo (Singapore) Pte Ltd | Folding electronic device |
| US9716942B2 (en) | 2015-12-22 | 2017-07-25 | Bose Corporation | Mitigating effects of cavity resonance in speakers |
| US20220221472A1 (en) * | 2019-04-26 | 2022-07-14 | Chugai Seiyaku Kabushiki Kaisha | Method for diagnosing endometriosis, disease state monitoring method, and kit |
| WO2022207628A1 (en) * | 2021-03-30 | 2022-10-06 | F. Hoffmann-La Roche Ag | Scf as blood biomarker for the non-invasive diagnosis of endometriosis |
| WO2022243210A1 (en) * | 2021-05-17 | 2022-11-24 | F. Hoffmann-La Roche Ag | sFRP4 AS BLOOD BIOMARKER FOR THE NON-INVASIVE DIAGNOSIS OF ADENOMYOSIS |
Non-Patent Citations (58)
| Title |
|---|
| ADAMSON ET AL., J ENDOMETR., vol. 2, 2010, pages 3 - 6 |
| ADU-GYAMFI ET AL., BIOLOGY OF REPRODUCTION, 2021 |
| AHN ET AL., FERTIL STERIL, 2016 |
| BLEND ET AL., CANCER BIOTHERAPY & RADIOPHARMACEUTICALS, vol. 18, 2003, pages 355 - 363 |
| BRIGGS ET AL.: "Synthesis of Functionalised Fluorescent Dyes and Their Coupling to Amines and Amino Acids", J. CHEM. SOC., PERKIN-TRANS., vol. 1, 1997, pages 1051 - 1058, XP002298160, DOI: 10.1039/a605012c |
| BRIGGS ET AL.: "Synthesis of Functionalized Fluorescent Dyes and Their Coupling to Amines and Amino Acids", J. CHEM. SOC., PERKIN-TRANS., vol. 1, 1997, pages 1051 - 1058, XP002298160, DOI: 10.1039/a605012c |
| CAMERA ET AL., J. NUCL. MED., vol. 21, 1994, pages 640 - 646 |
| CHEN ET AL., NAT. IMMUNOL., vol. 10, 2009, pages 889 - 898 |
| COXON ET AL.: "Is there a Neuropathic-Like Component to Endometriosis-Associated Pain? Results From a Large Cohort Questionnaire Study", FRONT PAIN RES (LAUSANNE, vol. 2, 2021, pages 743812 |
| COZZOLINO ET AL., REV BRAS GINECOL OBSTET, vol. 41, no. 3, 2019, pages 170 - 175 |
| DARLINGLAMB, FRONTIERS IN IMMUNOLOGY, 2019 |
| DEISENHOFER, BIOCHEMISTRY, vol. 20, 1981, pages 2361 - 2370 |
| DENARDO ET AL., CLINICAL CANCER RESEARCH, vol. 4, 1998, pages 2483 - 90 |
| DESMYTER ET AL., NAT. STRUCTURE BIOL., vol. 3, 1996, pages 803 - 811 |
| DODEIGNE C. ET AL., TALANTA, vol. 51, 2000, pages 415 - 439 |
| EYSTER ET AL., FERTIL STERIL, 2007 |
| FADHLAOUI ET AL., FRONT SURG., vol. 1, 2014, pages 24 |
| FASSBENDER ET AL., SPRINGER, PERIPHERAL BLOOD BIOMARKERS FOR ENDOMETRIOSIS, 2017 |
| FUJII ET AL., HUM REPROD, vol. 26, 2011, pages 299 - 306 |
| GEISBERGER ET AL., IMMUNOLOGY, vol. 118, 2006, pages 429 - 437 |
| HALIS. ET AL., DEUTSCHES ARZTEBLATT INTERNATIONAL, vol. 107, no. 25, 2010, pages 446 |
| HEITZER, E.HAQUE, I.S.ROBERTS, C.E.S. ET AL.: "Current and future perspectives of liquid biopsies in genomics-driven oncology", NAT REV GENET, vol. 20, 2019, pages 71 - 88, XP036675874, DOI: 10.1038/s41576-018-0071-5 |
| HEVER ET AL., PNAS, 2007 |
| HNATOWICH ET AL., J. IMMUNOL. METHODS, vol. 65, 1983, pages 147 - 157 |
| HSU ET AL., CLIN OBSTET GYNECOL, vol. 53, 2010, pages 413 - 419 |
| HUSTON ET AL., PROC. NATL. ACAD. SCI. USA, vol. 85, 1988, pages 5879 - 5883 |
| IZARD ET AL., BIOCONJUGATE CHEM., vol. 3, 1992, pages 346 - 350 |
| JANEWAY ET AL., IMMUNOBIOLOGY, GARLAND SCIENCE, 2001 |
| KABAT, E. A.WU, T.T.PERRY, H. M.GOTTESMAN, K. S.FOELLER, C.: "Sequences of proteins of immunological interest, 5thed. U.S. Department of Health and Human Service, National Institutes of Health, Bethesda, MD", THE ''EU INDEX AS IN KABAT'' REFERS TO THE RESIDUE NUMBERING OF THE HUMAN IGG IEU ANTIBODY, 1991 |
| KENNEDY ET AL., HUM REPROD., vol. 20, no. 10, 2005, pages 2698 - 2704 |
| KIMBER-TROJNAR ZANETA ET AL: "The Potential of Non-Invasive Biomarkers for Early Diagnosis of Asymptomatic Patients with Endometriosis", vol. 10, no. 13, 23 June 2021 (2021-06-23), CH, pages 2762, XP093111730, ISSN: 2077-0383, Retrieved from the Internet <URL:https://www.mdpi.com/2077-0383/10/13/2762/pdf> DOI: 10.3390/jcm10132762 * |
| KIYOMIDDINOVNA MUFTAYDINOVA SHAKHNOZA ET AL: "EXPRESSION OF THE TYROSINE KINASE RECEPTOR (EPHA1) IN THE EUTOPIC AND ECTOPIC ENDOMETRIUM OF PATIENTS WITH DEEP INFILTRATIVE ENDOMETRIOSIS USE OF MODERN DIGITAL TECHNOLOGIES", 15 December 2022 (2022-12-15), pages 1 - 6, XP093111345, Retrieved from the Internet <URL:https://dl.acm.org/doi/pdf/10.1145/3584202.3584263> [retrieved on 20231212] * |
| KNIGHT ET AL., ANALYST, vol. 119, 1994, pages 879 - 890 |
| KOBAYASHI ET AL., BIOCONJUGATE CHEM., vol. 10, 1999, pages 103 - 111 |
| KOBAYASHI ET AL., J. NUCL. MED., vol. 39, 1998, pages 2105 - 2110 |
| KUFER ET AL., TRENDS BIOTECHNOL., vol. 22, 2004, pages 238 - 244 |
| LEE ET AL., CANCER RES., vol. 61, 2001, pages 4474 - 4482 |
| LONGO, D.L ET AL., SC. D. N ENGL J MED, vol. 382, 2020, pages 1244 - 56 |
| MARDIROSSIAN ET AL., NUCL. MED. BIOL., vol. 20, 1993, pages 955 - 74 |
| MEARES ET AL., ANAL. BIOCHEM., vol. 142, 1984, pages 68 - 78 |
| MEARES ET AL., J. CANCER, vol. 10, 1990, pages 21 - 26 |
| MIEDERER ET AL., J. NUCL. MED., vol. 45, 2004, pages 129 - 137 |
| MIRZADEH ET AL., BIOCONJUGATE CHEM., vol. 1, 1990, pages 59 - 65 |
| MITCHELL ET AL., J. NUCL. MED., vol. 44, 2003, pages 1105 - 1112 |
| MOLDASSARINA RYMGUL S.: "Modern view on the diagnostics and treatment of adenomyosis", ARCHIVES OF GYNECOLOGY AND OBSTETRICS ;, vol. 308, no. 1, 15 April 2023 (2023-04-15), Berlin, DE, pages 171 - 181, XP093213667, ISSN: 1432-0711, Retrieved from the Internet <URL:https://link.springer.com/content/pdf/10.1007/s00404-023-06982-1.pdf> DOI: 10.1007/s00404-023-06982-1 * |
| NEZHAT ET AL., JSLS, 2019 |
| NIKULA ET AL., J. NUCL. MED., vol. 40, 1999, pages 166 - 76 |
| NIKULA ET AL., NUCL. MED. BIOL., vol. 22, 1995, pages 387 - 90 |
| PARASAR ET AL., CURR OBSTET GYNECOL REP., vol. 6, 2017, pages 34 - 41 |
| PERRICOS ET AL., EXP BIOL MED (MAYWOOD, 2018 |
| PIER ET AL., IMMUNOLOGY, INFECTION, AND IMMUNITY, 2004 |
| ROSELLI ET AL., CANCER BIOTHERAPY & RADIOPHARMACEUTICALS, vol. 14, 1999, pages 209 - 20 |
| RUEGG ET AL., CANCER RES., vol. 50, 1990, pages 4221 - 4226 |
| SIGNORILEBALDI, J CELL PHYSIOL, vol. 229, 2014, pages 1731 - 1735 |
| SINAII ET AL., FERTIL STERIL., vol. 89, no. 3, 2008, pages 538 - 545 |
| UNDERDOWNSCHIFF, ANNU. REV. IMMUNOL., vol. 4, 1986, pages 389 - 417 |
| WANG ET AL., JOURNAL OF OVARIAN RESEARCH, 2022 |
| ZHANG X., FRONT ONCOL., vol. 11, 2021, pages 619949 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20220137058A1 (en) | S100a6 as blood biomarker for the non-invasive diagnosis of endometriosis | |
| US20250130244A1 (en) | Method for diagnosing endometriosis and for classifying the stage of endometriosis | |
| US20220137068A1 (en) | S100a8 as blood biomarker for the non-invasive diagnosis of endometriosis | |
| JP7727015B2 (en) | sFRP4 as a blood biomarker for the non-invasive diagnosis of adenomyosis | |
| WO2022207628A1 (en) | Scf as blood biomarker for the non-invasive diagnosis of endometriosis | |
| US20240027474A1 (en) | Psp94 as blood biomarker for the non-invasive diagnosis of endometriosis | |
| US20220155315A1 (en) | S100a9 as blood biomarker for the non-invasive diagnosis of endometriosis | |
| US20220137069A1 (en) | S100a12 as blood biomarker for the non-invasive diagnosis of endometriosis | |
| WO2025026908A1 (en) | Serum epha1 as biomarker for endometriosis | |
| JP7315780B2 (en) | Substance P as a blood biomarker for non-invasive diagnosis of endometriosis | |
| CA2929810A1 (en) | Improved early prediction of preeclampsia | |
| CN112680514A (en) | Marker for liver cancer diagnosis and application thereof | |
| CN112695093A (en) | Application of gene combination in diagnosis of liver cancer | |
| CN112695092A (en) | Diagnostic system for determining risk of disease | |
| CN112501300A (en) | Molecular markers for cancer diagnosis | |
| CN112695090A (en) | Use of marker combinations |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24748936 Country of ref document: EP Kind code of ref document: A1 |