WO2025021943A1 - Quinazolinone, benzoxazinone and benzoxazepinone derivatives as protein kinase inhibitors - Google Patents
Quinazolinone, benzoxazinone and benzoxazepinone derivatives as protein kinase inhibitors Download PDFInfo
- Publication number
- WO2025021943A1 WO2025021943A1 PCT/EP2024/071181 EP2024071181W WO2025021943A1 WO 2025021943 A1 WO2025021943 A1 WO 2025021943A1 EP 2024071181 W EP2024071181 W EP 2024071181W WO 2025021943 A1 WO2025021943 A1 WO 2025021943A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- independently
- occurrence
- compound
- hydrogen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
Definitions
- the present invention is in the field of medicinal chemistry and pharmaceuticals.
- Protein phosphorylation is the most common form of reversible post- translational modification, with an estimated 50% of all proteins undergoing phosphorylation.
- the phosphorylation state of any given protein is controlled by the coordinated action of specific kinases and phosphatases that add and remove phosphate, respectively.
- protein kinases are a kind of protein phosphotransferases bringing the phosphate of ATP to the specific amino acid residue. They may conventionally be divided into five classes: tyrosine protein kinases, serine/threonine protein kinases, histidine protein kinases, tryptophan protein kinases and aspartyl/glutamoyl protein kinases.
- tumours the abnormal oncogenic activation of protein kinases derives from multiple types of genetic and epigenetic changes. These alterations result in increased specific activity of the kinase itself, its overexpression, or the loss of negative regulation leading to uncontrolled cellular growth and sustained malignant behaviour.
- the signalling networks operating in cancer cells can also contribute to innate or acquired resistance to treatment, since they are able to create the most common or rare oncogenic mutations different from tumour to tumour.
- the search for small-molecule inhibitors targeting the altered protein kinase molecules in tumour cells has become a major research focus in the academia and pharmaceutical companies.
- Such inhibitors can be products that are derived (isolated) from sources such as plants, animals or microorganisms, or can be small- molecules that are designed (synthetized).
- WO 2004/022572 discloses classes of biologically active compounds interacting with kinases, and the preparation of these compounds.
- pexidartinib is utilized to inhibit the colony-stimulating factor-1 receptor (CSF1 R), the KIT proto-oncogene receptor tyrosine kinase (KIT) and the FMS-like tyrosine kinase 3 (FLT3) in, for example, the treatments of patients with symptomatic tenosynovial giant cell tumors (TGCT); edicotinib to inhibit the CSF1 R and currently in phase II for acute myeloid leukemia, cognition disorders or Crohn’s disease; or nintedanib to inhibit the endothelial growth factor receptor (VEGFR), fibroplast growth factor receptor (FGFR), platelet-derived growth factor receptor (PDGFR) and CSF1 R in, for example, the treatment of idiopathic pulmonary fibrosis.
- CSF1 R colony-stimulating factor-1 receptor
- KIT KIT proto-oncogene receptor tyrosine kinase
- FLT3
- WO 201 1/090738 A2 discloses compounds that are able to inhibit B-RAF and B-RAF mutations and methods for treating diseases related to B-RAF and B-RAF mutation modulation.
- US 2009/0325945 describes active compounds, specifically, certain imidazo[4,5-b]pyridin-2-one and oxazolo[4,5-b]pyridin-2-one compounds and analogs inhibiting RAF (e.g., B-RAF) activity in a cell, in vitro or in vivo, inhibiting receptor tyrosine kinase (RTK) activity, such as FGFR, Tie, VEGFR and/or Eph activity, for example, FGFR-1 , FGFR-2, FGFR-3, Tie2, VEGFR-2 and/or EphB2 activity, in a cell, in vitro or in vivo.
- RAF e.g., B-RAF
- RTK receptor tyrosine kinase
- the present invention further relates to a pharmaceutical composition comprising a carrier, and as active ingredient an effective amount of a compound as defined in any one of the embodiments presented herein.
- the present invention relates to a compound as defined in any one of the embodiments presented herein, for use as a medicament.
- the present invention relates to a compound as defined in any one of the embodiments presented herein for use in the treatment of a disease selected from cancer, metabolic disorders (such as diabetes), inflammatory and autoimmune disorders (such as inflammatory bowel diseases, e.g.
- Crohn’s disease and ulcerative colitis inflammatory pulmonary diseases, rheumatoid arthritis, lupus nephritis, systemic lupus erythematosus and psoriasis and psoriasis arthritis), neurological disorders (such as Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, Charcot-Marie-Tooth neuropathy, amyotrophic lateral sclerosis and epilepsy), atherosclerosis and cardiovascular diseases, Sjogren Syndrome, renal allograft rejection, viral induced diseases, circulatory diseases, bone osteolysis and osteoporosis, osteoarthritis, sarcopenia, Langerhans cell histiocytosis, spinal cord injury, endometriosis, asthma and allergic asthma, eye diseases (such as retinopathies, age-related macular degeneration and uveitis) chronic and neuropathic pain, and fibro- proliferative diseases.
- neurological disorders such as Alzheimer’s disease, Parkinson’s disease, multiple
- the present invention relates to a compound as defined in any one of the embodiments presented herein, for use in the treatment of pain sensitization.
- the present invention further relates to a method of inhibiting protein kinase activity in a warm-blooded animal said method comprising the administration to an animal in need thereof, of a kinase-inhibitory effective amount of a compound according to any one of the embodiments presented herein.
- the present invention further relates to a method of treating a disease selected from cancer, metabolic disorders (such as diabetes), inflammatory and autoimmune disorders (such as inflammatory bowel diseases, e.g.
- each of R 1 in compound (C) of formula (I), independently from each other and at each occurrence, is selected from hydrogen or C 1-6 alkyl. More preferably, each of R 1 , independently from each other and at each occurrence, is selected from hydrogen, or C 1-4 alkyl. Even more preferably, each of R 1 , independently from each other and at each occurrence, is C 1-4 alkyl.
- each of R1, independently from each other and at each occurrence is methyl;
- each of X in compound (C) of formula (I), independently from each other and at each occurrence is selected from O, S, or NR7, wherein R7 is selected from hydrogen or C1-6 alkyl. More preferably, each of X, independently from each other and at each occurrence is selected from O, S, or NR 7 , wherein R 7 is selected from hydrogen or C 1-4 alkyl. Even more preferably, each of X, independently from each other and at each occurrence is selected from O, S, or NH. Even more preferably, each of X, independently from each other and at each occurrence is O.
- each of Y in compound (C) of formula (I), independently from each other and at each occurrence is selected from the group consisting of O, S, and NR 7 , wherein R 7 is selected from hydrogen or C 1-6 alkyl. More preferably, each of Y independently from each other and at each occurrence is selected from the group consisting of O, S, and NR 7 , wherein R 7 is selected from hydrogen or C 1-4 alkyl. Even more preferably, each of Y independently from each other and at each occurrence is selected from the group consisting of O, S, and NH. Even more preferably, each of Y independently from each other and at each occurrence is selected from O, or NH.
- each of R5 and R5’ in compound (C) of formula (I), independently from each other and at each occurrence is selected from hydrogen or C1-6 alkyl. More preferably, each of R5, and R5’, independently from each other and at each occurrence is selected from hydrogen or C1-4 alkyl. Even more preferably, each of R5, and R5’, independently from each other and at each occurrence is selected from hydrogen or methyl.
- each of n5 in compound (C) of formula (I), independently from each other and at each occurrence is an integer equal to 0 or 1. More preferably, each of n5, independently from each other and at each occurrence, is an integer equal to 0.
- R6 in compound (C) of formula (I) is selected from the group consisting of halo, NO2, C1-6 alkyl, CF3, CN, OR11, SR11, N(R11)2, COR11, C(O)OR11, CON(R11)2, OC(O)R11, and wherein R11 is independently selected from the group consisting of hydrogen, C1-6 alkyl, and CF3. More preferably, R 6 is selected from the group consisting of halo, NO 2 , C 1-6 alkyl, CF 3 , CN, OR 11 , and N(R 11 ) 2 , and wherein R 11 is independently selected from the group consisting of hydrogen, and C 1-6 alkyl.
- R 6 is selected from the group consisting of halo, NO 2 , C 1-6 alkyl, CF 3 , CN, OR 11 , and N(R 11 ) 2 , and wherein R 11 is independently selected from the group consisting of hydrogen, and C 1-4 alkyl. Even more preferably, R 6 is halo.
- each of n 6 in compound (C) of formula (I), independently from each other and at each occurrence is an integer equal to 0 or 1. More preferably, each of n 5 , independently from each other and at each occurrence, is an integer equal to 0.
- each of Z in compound (C) of formula (I), independently from each other and at each occurrence is selected from the group consisting of O, S, and NR 7 , wherein R 7 is selected from hydrogen or C 1-6 alkyl. More preferably, each of Z independently from each other and at each occurrence is selected from the group consisting of O, S, and NR7, wherein R7 is selected from hydrogen or C1-4 alkyl. Even more preferably, each of Z independently from each other and at each occurrence is NR7, wherein R7 is selected from hydrogen or C1-4 alkyl. Even more preferably, each of Z, independently from each other and at each occurrence, is NR7, wherein R7 is selected from hydrogen or methyl.
- each of z in compound (C) of formula (I), independently from each other and at each occurrence, is an integer equal to 0, 1, 2 or 3. More preferably, z, independently from each other and at each occurrence, is an integer equal to 0, 1, or 2;
- each of A in compound (C) of formula (I), independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C 1-6 alkyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl, wherein said alkyl, alkenyl, alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl are optionally substituted with one or more substituents independently selected from the group consisting of halo, NO 2 , C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, cycloalkyl, heterocyclyl
- each of A independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl, wherein said alkyl, alkenyl, alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl are optionally substituted with one or more substituents independently selected from the group consisting of halo, C1-6 alkyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, CF3, CN, OR11, N(R11)2, OC(R11)2O, OC(R11)2C(R11)2O, S(O)R12, SO2R12, SO2N(R11)2, COR11, C(O)OR11, CON(R11)2, OC(O)R11, and OCON(R11)2; and wherein each of R11
- each of A independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl, wherein said alkyl, alkenyl, alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl are optionally substituted with one or more substituents independently selected from the group consisting of halo, C 1-6 alkyl, CF 3 , CN, OR 11 , N(R 11 ) 2 , OC(R 11 ) 2 O, SO 2 N(R 11 ) 2 ; and wherein each of R 11 and R 12 , independently from each other and at each occurrence, is selected from the group consisting of hydrogen or C 1-4 alkyl such as methyl, ethyl, propyl, isopropyl, butyl, tert-butyl and isobutyl
- each of halo is F, Cl, Br, or I
- each of R is hydrogen or C1-4 alkyl such as methyl, ethyl, propyl, isopropyl, butyl, tert-butyl is isobutyl, preferably R is hydrogen, methyl, ethyl, 2-methylpropyl or tert-butyl.
- the compound (C) according to formula (I), or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof preferably is a compound of formulae (I-a-1) [compound (C) of class (I-a) herein-after]: Formula (I-a-1) wherein R 1 , X, Y, R 5 , R 5 ’, R 4 , R 4 ’, z and A have the same meaning as defined above for formula (I).
- the compound (C) according to formula (I), or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof preferably is a compound chosen among those of formulae (I-b-1) to (I-b-3) [compound (C) of class (I-b) herein-after]: Formula (I-b-1) Formula (I-b-2) Formula (I-b-3) wherein R1, X, R5, R4, R4’, z and A have the same meaning as defined above for formula (I).
- the compound (C) according to formula (I), or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof preferably is a compound according to formula (I-c-1) [compound (C) of class (I-c) herein-after]: Formula (I-c-1) wherein R 1 , X, Z, R 4 , R 4 ’, z and A have the same meaning as defined above for formula (I).
- the compound (C) according to formula (I), or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof preferably is a compound of formulae (I-d-1) [compound (C) of class (I-d) herein-after]: Formula (I-d-1) wherein R1, X, Y, R5, n5, R4, R4’, z and A have the same meaning as defined above for formula (I).
- the compound (C) according to formula (I), or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof preferably is a compound of formulae (I-e-1) [compound (C) of class (I-e) herein-after]: Formula (I-e-1) wherein R1, X, Y, Z, R5, n5, R4, R4’, z and A have the same meaning as defined above for formula (I).
- the compound (C) according to formula (I), or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof preferably is a compound of formulae (I-f-1) [compound (C) of class (I-f) herein-after]: Formula (I-f-1) wherein R1, X, Y, Z, R6 and n6 have the same meaning as defined above for formula (I).
- R1, X, Y, Z, R6 and n6 have the same meaning as defined above for formula (I).
- X is O
- Y is O
- Z is NR7.
- Preferred compounds (C) of class (I-a) are thus compounds of formula (I-a-1-1) herein below: Formula (I-a-1-1) wherein R 1 , R 5 , R 5 ’, R 4 , R 4 ’, z and A have the same meaning as defined above for formula (I).
- the compounds (C) of class (I-a) are selected from those of formula (I-a-1-1).
- Preferred compounds (C) of class (I-b) are thus selected from those of formulae (I-b-1-1) to (I-b-3-1) herein below: Formula (I-b-1-1) Formula (I-b-2-1) Formula (I-b-3-1) wherein R1, R5, R4, R4’, z and A have the same meaning as defined above for formula (I).
- the compounds (C) of class (I-b) are selected from those of formula (I-b-1-1), (I-b-2-1) or (I-b-3-1).
- Preferred compounds (C) of class (I-c) are thus compounds of formula (I-c-1-1) herein-below: Formula (I-c-1-1) wherein R 1 , R 4 , R 4 ’, z and A have the same meaning as defined above for formula (I), and wherein R 7 is selected from hydrogen or C 1-6 alkyl, wherein said alkyl is optionally substituted with halogen atom, preferably R 7 is hydrogen or methyl, more preferably R 7 is hydrogen.
- the compounds (C) of class (I-c) are selected from those of formula (I-c-1-1).
- Preferred compounds (C) of class (I-d) are thus compounds of formula (I-d-1-1) herein-below: Formula (I-d-1-1) wherein R1, R5, n5, R4, R4’, z and A have the same meaning as defined above for formula (I).
- the compounds (C) of class (I-d) are selected from those of formula (I-d-1-1).
- Preferred compounds (C) of class (I-e) are thus compounds of formula (I-e-1-1) herein-below: Formula (I-e-1-1) wherein R 1 , R 5 , n 5 , R 4 , R 4 ’, z and A have the same meaning as defined above for formula (I), and wherein R7 is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with halogen atom, preferably R7 is hydrogen or methyl, more preferably R7 is hydrogen.
- the compounds (C) of class (I-e) are selected from those of formula (I-e-1-1).
- Preferred compounds (C) of class (I-f) are thus compounds of formula (I- f-1-1) herein-below: Formula (I-f-1-1) wherein R1, R6, and n6 have the same meaning as defined above for formula (I), and wherein R 7 is selected from hydrogen or C 1-6 alkyl, wherein said alkyl is optionally substituted with halogen atom, preferably R 7 is hydrogen or methyl, more preferably R 7 is hydrogen.
- the compounds (C) of class (I-f) are selected from those of formula (I-f-1-1).
- the compound (C) of class (I-a) is selected from those of formula (I-a-1-1-1) herein-below: Formula (I-a-1-1-1) wherein R1, R5, R5’, R4, R4’, z and A have the same meaning as defined above for formula (I).
- the compound (C) of formula (I-b-1-1) of class (I-b) is selected from those of formula (I-b-1-1-1) to formula (I-b-1-1-3) herein-below: Formula (I-b-1-1-1) Formula (I-b-1-1-2) Formula (I-b-1-1-3) wherein R 1 , R 5 , R 4 , R 4 ’, z and A have the same meaning as defined above for formula (I).
- the compound (C) of formula (I-b-2-1) of class (I-b) is selected from those of formulae (I-b-2-1-1) to (I- b-2-1-3) herein-below: Formula (I-b-2-1-1) Formula (I-b-2-1-2) Formula (I-b-2-1-3) wherein R 1 , R 5 , R 4 , R 4 ’, z and A have the same meaning as defined above for formula (I).
- the compound (C) of formula (I-b-3-1) of class (I-b) is selected from those of formula (I-b-3-1-1) to (I- b-3-1-3) herein-below: Formula (I-b-3-1-1) Formula (I-b-3-1-2) Formula (I-b-3-1-3) wherein R1, R5, R4, R4’, z and A have the same meaning as defined above for formula (I).
- the compound (C) of class (I-c) is selected from those of formula (I-c-1-1-1) herein-below: Formula (I-c-1-1-1) wherein R1, R4, R4’, z and A have the same meaning as defined above for formula (I), and wherein R7 is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with halogen atom, preferably R7 is hydrogen or methyl, more preferably R7 is hydrogen.
- the compound (C) of class (I-d) is selected from those of formula (I-d-1-1-1) herein-below: Formula (I-d-1-1-1) wherein R 1 , R 5 , n 5 , R 4 , R 4 ’, z and A have the same meaning as defined above for formula (I).
- the compound (C) of class (I-f) is selected from those of formula (I-f-1-1-1) herein-below: Formula (I-f-1-1-1) wherein R1, R6 and n6 have the same meaning as defined above for formula (I).
- the compound (C) of formula (I-a-1-1-1) of class (I-a) according to the present invention is a compound chosen among those of formula (I-a-1-1-1-1) to (I-a-1-1-1-6) herein below: Formula (I-a-1-1-1-1) Formula (I-a-1-1-1-2) Formula (I-a-1-1-1-3) Formula (I-a-1-1-1-4) Formula (I-a-1-1-1-5) Formula (I-a-1-1-1-6) wherein R4, R4’ and z have the same meaning as defined above for formula (I) and wherein: - each of R 8 independently from each other and at each occurrence is selected from the group consisting of hydrogen, halo and CF3.
- the compound (C) of formula (I-b-1-1-1) of class (I-b) according to the present invention is selected from those of formula (I-b-1-1-1-1) to (I-b-1-1-1-13) herein below: Formula (I-b-1-1-1-1) Formula (I-b-1-1-1-2) Formula (I-b-1-1-1-3) Formula (I-b-1-1-1-4) Formula (I-b-1-1-1-5) Formula (I-b-1-1-1-1-6) Formula (I-b-1-1-1-7) Formula (I-b-1-1-1-1-8) Formula (I-b-1-1-1-9) Formula (I-b-1-1-1-10) Formula (I-b-1-1-1-11) Formula (I-b-1-1-12) Formula (I-b-1-1-13) wherein R4, R4’ and z have the same meaning as defined above for formula
- the compound (C) of Formula (I-b-1-1-2) of class (I-b) according to the present invention is selected from those of formula (I-b-1-1-2-1) herein below: Formula (I-b-1-1-2-1) wherein R 4 , R 4 ’ and z have the same meaning as defined above for formula (I) and wherein halo is selected from the group consisting of F, Cl, Br and I.
- the compound (C) of Formula (I-b-1-1-3) of class (I-b-1) according to the present invention is selected from those of formula (I-b-1-1-3-1) herein below: Formula (I-b-1-1-3-1) wherein R4, R4’ and z have the same meaning as defined above for formula (I) and wherein halo is selected from the group consisting of F, Cl, Br and I.
- the compound (C) of Formula (I-b-2-1-1) of class (I-b-2) according to the present invention is selected from those of formula (I-b-2-1-1-1) herein below: H O R 4 N O Halo N N N N N R 4 ' H 3 C z Formula (I-b-2-1-1-1) wherein R 4 , R 4 ’ and z have the same meaning as defined above for formula (I) and wherein halo is selected from the group consisting of F, Cl, Br and I.
- the compound (C) of Formula (I-b-2-1-2) of class (I-b-2) according to the present invention is selected from those of formula (I-b-2-1-2-1) herein below: Formula (I-b-2-1-2-1) wherein R 4 , R 4 ’ and z have the same meaning as defined above for formula (I) and wherein halo is selected from the group consisting of F, Cl, Br and I.
- the compound (C) of Formula (I-b-2-1-3) of class (I-b-2) according to the present invention is selected from those of formula (I-b-2-1-3-1) herein below: Formula (I-b-2-1-3-1) wherein R4, R4’ and z have the same meaning as defined above for formula (I) and wherein halo is selected from the group consisting of F, Cl, Br and I.
- the compound (C) of Formula (I-b-3-1-1) of class (I-b-3) according to the present invention is selected from those of formula (I-b-3-1-1-1) herein below: Formula (I-b-3-1-1-1) wherein R 4 , R 4 ’ and z have the same meaning as defined above for formula (I) and wherein halo is selected from the group consisting of F, Cl, Br and I.
- the compound (C) of Formula (I-b-3-1-2) of class (I-b) according to the present invention is selected from those of formula (I-b-3-1-2-1) herein below: Formula (I-b-3-1-2-1) wherein R4, R4’ and z have the same meaning as defined above for formula (I) and wherein halo is selected from the group consisting of F, Cl, Br and I.
- the compound (C) of Formula (I-b-3-1-3) of class (I-b) according to the present invention is selected from those of formula (I-b-3-1-3-1) herein below: Formula (I-b-3-1-3-1) wherein R 4 , R 4 ’ and z have the same meaning as defined above for formula (I) and wherein halo is selected from the group consisting of F, Cl, Br and I.
- the compound (C) of Formula (I-c-1-1-1) of class (I-c) according to the present invention is selected from those of formula (I-c-1-1-1) or (I-c-1-1-1-2) herein below:
- Formula (I-c-1-1-1-1) Formula (I-c-1-1-1-2) wherein R 4 , R 4 ’ and z have the same meaning as defined above for formula (I) and wherein: - each of R 7 ’ , independently from each other and at each occurrence is H or CH 3 ;
- - each of U is independently C-R 12c or N;
- - each of R 12c independently from each other and at each occurrence, is hydrogen, halo or OCH 3 ;
- - n 12c is an integer equal to 0, 1 or 2.
- the compound (C) of Formula (I-d-1-1-1) of class (I-d) according to the present invention is selected from those of formula (I-d-1-1-1) or (I-d-1-1-1-2) herein below:
- Formula (I-d-1-1-1-1) Formula (I-d-1-1-1-2) wherein R 4 , R 4 ’ and z have the same meaning as defined above for formula (I) and wherein: - each of U is independently C-R 12d or N; - each of R 12d , independently from each other and at each occurrence, is hydrogen, halo or CF 3 ; - n 12d is an integer equal to 0, 1 or 2.
- the compound (C) of formula (I-f-1-1-1) of class (I-f) is selected from those of formula (I-f-1-1-1-1) herein-below: Formula (I-f-1-1-1-1) wherein halo is selected from the group consisting of F, Cl, Br and I.
- the compound (C) of formula (I-a-1-1-1-3) of class (I-a) is preferably selected from those of formula (I-a-1-1- 1-3-0) herein-below: Formula (I-a-1-1-1-3-0) wherein: - each of R 80 independently from each other and at each occurrence is selected from hydrogen or halo; - n 80 is an integer equal to 0 or 1.
- the compound (C) of formula (I-a-1-1-1-3) of class (I-a) is preferably selected from those of formula (I-a-1-1- 1-3-1) herein-below: Formula (I-a-1-1-1-3-1) wherein: - each of R 5 and R 5 ’, independently from each other and at each occurrence is hydrogen or methyl; - each of R 81 independently from each other and at each occurrence is selected from the group consisting of hydrogen, halo and CF 3 ; - n81 is an integer equal to 0, 1 or 2.
- the compound (C) of formula (I-a-1-1-1-3) of class (I-a) is preferably selected from those of formula (I-a-1-1- 1-3-2) herein-below: Formula (I-a-1-1-1-3-2) wherein each of R82 independently from each other and at each occurrence is selected from hydrogen or halo.
- the compound (C) of formula (I-a-1-1-1-6) of class (I-a) is preferably selected from those of formula (I-a-1-1- 1-6-2) herein-below: Formula (I-a-1-1-1-6-2)
- the compound (C) of formula (I-b-1-1-1-4) of class (I-b) is preferably selected from those of formula (I-a-1-1- 1-4-0) herein-below: Formula (I-b-1-1-1-4-0) wherein each of R 110 , independently from each other and at each occurrence is selected from hydrogen or halo.
- the compound (C) of formula (I-b-1-1-1-1) of class (I-b) is preferably selected from those of the following formulae herein-below: Formula (I-b-1-1-1-1-2) Formula (I-b-1-1-1-4-2) Formula (I-b-1-1-1-1-4-2a) Formula (I-b-1-1-1-5-2) Formula (I-b-1-1-1-5-2a) Formula (I-b-1-1-1-6-2) Formula (I-b-1-1-1-7-2) wherein: - R 92 is selected from hydrogen, N(CH 3 ) 2 or ethyl; - R 42 is selected from hydrogen or ethyl; - each of R 112 , independently from each other and at each occurrence is selected from the group consisting of hydrogen, halo, and CF 3 ; - each of R 102 , independently from each other and at each occurrence is selected from hydrogen or methyl; - each of n 102 , independently from each other and at each occurrence is an integer
- the compound (C) of formula (I-b-2-1-1-1) of class (I-b) is preferably selected from those of the following formulae herein-below: Formula (I-b-2-1-1-1-1) wherein halo is selected from the group consisting of F, Cl, Br and I.
- the compound (C) of formula (I-b-2-1-2-1) of class (I-b) is preferably selected from those of the following formulae herein-below: Formula (I-b-2-1-2-1-1) wherein halo is selected from the group consisting of F, Cl, Br and I.
- the compound (C) of formula (I-b-2-1-3-1) of class (I-b) is preferably selected from those of the following formulae herein-below: Formula (I-b-2-1-3-1-1) wherein halo is selected from the group consisting of F, Cl, Br and I.
- the compound (C) of formula (I-b-3-1-1-1) of class (I-b) is preferably selected from those of the following formulae herein-below: Formula (I-b-3-1-1-1-1) wherein halo is selected from the group consisting of F, Cl, Br and I.
- the compound (C) of formula (I-b-3-1-2-1) of class (I-b) is preferably selected from those of the following formulae herein-below: Formula (I-b-3-1-2-1-1) wherein halo is selected from the group consisting of F, Cl, Br and I.
- the compound (C) of formula (I-b-3-1-3-1) of class (I-b) is preferably selected from those of the following formulae herein-below: Formula (I-b-3-1-3-1-1) wherein halo is selected from the group consisting of F, Cl, Br and I.
- the compound (C) of formula (I-c-1-1-1-2) of class (I-c) is preferably selected from those of formulae (I-c-1- 1-2-1) herein-below: Formula (I-c-1-1-1-2-1) wherein: - each of R7’, independently from each other and at each occurrence is H or CH3; - each of U is independently selected from C-R12’ or N; - each of R12c1, independently from each other and at each occurrence, is hydrogen, halo or OCH3; - n12c1 is an integer equal to 0, 1 or 2.
- the compound (C) of formula (I-d-1-1-1-1) of class (I-d) is preferably selected from those of formulae (I-d-1- 1-1-0) or (I-d-1-1-1-1-0) herein-below: Formula (I-d-1-1-1-1-0) wherein: - each of U is independently C-R 12d0 or N; - each of R 12d0 , independently from each other and at each occurrence, is selected from the group consisting of hydrogen, halo, or CF 3 ; - n 12d0 is an integer equal to 0, 1 or 2; In one embodiment of the present invention, the compound (C) of formula (I-d-1-1-1-1) of class (I-d) is preferably selected from those of formulae (I-d-1-1- 1-1-1) herein-below: Formula (I-d-1-1-1-1-1-1) wherein: - each of U is independently C-R12d1 or N; - each of R12d1, independently from each other and at
- the compound (C) according to general formula (I-a-1) is a compound chosen among those of formulae (II) to (XXIX) herein below: Formula (I) Formula (II) Formula (III) Formula (IV) Formula (V) Formula (VI) Formula (VII) Formula (VIII) Formula (IX) Formula (X) Formula (XI) Formula (XII) Formula (XIII) Formula (XIV) Formula (XV) Formula (XVI) Formula (XVII) Formula (XVIII) Formula (XIX) Formula (XX) Formula (XXI) Formula (XXII) Formula (XXIII) Formula (XXIV) Formula (XXV) Formula (XXVI) Formula (XVII) Formula (XXVIII) Formula (XXIX) In a preferred embodiment of the present invention, the compound (C) according to general formula (I-b-1) is a compound chosen among those of formulae (XXX) to (CIII) herein below:
- each of n6, independently from each other and at each occurrence, is an integer equal to 0, 1 or 2;
- - each of Z independently from each other and at each occurrence is selected from the group consisting of O, S and NR 7 , wherein R 7 is selected from hydrogen or C 1-6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom;
- - each of z, independently from each other and at each occurrence is an integer equal to 0, 1, 2, 3 or 4;
- - each of A independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, cycloalkyl
- halo - alone or in combination means all halogens, that is, chloro (Cl), bromo (Br), fluoro (F), iodo (I).
- alkyl - alone or in combination means an alkane-derived radical containing from 1 to 15 carbon atoms, unless otherwise specified, for example C F-G alkyl defines a straight or branched alkyl radical having from F to G carbon atoms, e.g.
- C 1-4 alkyl defines a straight or branched alkyl radical having from 1 to 4 carbon atoms such as for example methyl, ethyl, 1-propyl, 2-propyl, I-butyl, 2-butyl, 2-methyl-1-propyl.
- An alkyl group may be a straight chain alkyl or branched alkyl.
- straight or branched alkyl groups containing from 1- 10, more preferably 1 to 8, even more preferably 1-6 and most preferably 1-4, carbon atoms, such as methyl, ethyl, propyl, isopropyl, butyl, t-butyl and the like.
- Alkyl also includes a straight chain or branched alkyl group that contains or is interrupted by a cycloalkyl portion.
- the straight chain or branched alkyl group is attached at any available point to produce a stable compound. Examples of this include, but are not limited to, 4-(isopropyl)-cyclohexylethyl or 2-methyl- cyclopropylpentyl.
- alkenyl - alone or in combination means a straight or branched hydrocarbon containing 2-15, more preferably 2-10, even more preferably 2-8, most preferably 2-4, carbon atoms, unless otherwise specified and at least one, preferably 1-3, more preferably 1-2, most preferably one, carbon to carbon double bond.
- alkenyl groups include ethenyl, propenyl, isopropenyl, butenyl, cyclohexenyl, cyclohexenylalkyl and the like.
- Alkenyl also includes a straight chain or branched alkenyl group that contains or is interrupted by a cycloalkyl portion. Carbon to carbon double bonds may be either contained within a cycloalkyl portion, with the exception of cyclopropyl, or within a straight chain or branched portion.
- alkynyl - alone or in combination means a straight or branched hydrocarbon containing 2-15 more preferably 2-10, even more preferably 2-8, most preferably 2-4, carbon atoms containing at least one, preferably one, carbon to carbon triple bond.
- alkynyl groups include ethynyl, propynyl, butynyl and the like.
- aryl - alone or in combination means phenyl, naphthyl or anthracenyl optionally carbocyclic fused with a cycloalkyl or heterocyclyl of preferably 5-7, more preferably 5-6, ring members and/or optionally substituted with 1 to 5 groups or substituent.
- An aryl may be optionally substituted whereby the substituent is attached at one point to the aryl or whereby the substituent is attached at two points to the aryl to form a bicyclic system e.g. benzodioxole, benzodioxan, benzimidazole.
- heteroaryl - alone or in combination means a monocyclic aromatic ring structure containing 5 or 6 ring atoms, or a bicyclic aromatic group having 8 to 10 atoms, containing 1-3, heteroatoms independently selected from the group O, S, and N, and optionally substituted with 1 to 5 groups or substituents.
- Heteroaryl is also intended to include oxidized S or N, such as sulfinyl, sulfonyl and N-oxide of a tertiary ring nitrogen.
- a carbon or nitrogen atom is the point of attachment of the heteroaryl ring structure such that a stable aromatic ring is retained.
- heteroaryl includes, but is not limited to, pyridyl, furanyl, thiophenyl, thiazolyl, isothiazolyl, triazolyl, imidazolyl, isoxazolyl, pyrrolyl, pyrazolyl, pyrimidinyl, benzofuranyl, isobenzofuranyl, benzothiazolyl, benzoisothiazolyl, benzotriazolyl, indolyl, isoindolyl, benzoxazolyl, quinolyl, isoquinolyl, benzimidazolyl, benzisoxazolyl, benzothiophenyl, dibenzofuran, and benzodiazepin-2-one-5-yl, and the like.
- heterocyclyl - alone or in combination is intended to denote a saturated, partially unsaturated or completely unsaturated monocycle, bicycle, or tricycle having 3 to 12 carbon atoms and containing 1 or 2 heteroatoms each independently selected from O, S, P or N, and are optionally benzo fused or fused heteroaryl of 5-6 ring members and/or are optionally substituted as in the case of cycloalkyl.
- Heterocycyl is also intended to include oxidized S or N, such as sulfinyl, sulfonyl and N-oxide of a tertiary ring nitrogen. The point of attachment is at a carbon or nitrogen atom.
- cycloalkyl refers to a cyclic or polycyclic alkyl group containing 3 to 7 carbon atoms.
- cycloalkyl groups are monocyclic, bicyclic or tricyclic ring systems of 3-6, ring members per ring, such as cyclopropyl, cyclopentyl, cyclohexyl, adamantyl and the like.
- aralkyl refers to organic compounds containing an aromatic nucleus to which an alkyl radical is bonded.
- alkyl radicals include methyl, ethyl, propyl, butyl, octyl, etc. radicals.
- aralkyl is thus seen to include aralkyl hydrocarbons such as the alkyl benzenes, and the various alkyl naphthalenes. From this definition of the term aralkyl compound it is seen that the term includes compounds such as benzyl, the three isomeric xylyls, the two isomeric trimethyl benzenes, ethyl benzene, p-methyl biphenyl, a-methyl naphthalene, etc.
- the present invention further relates to a pharmaceutical composition
- a pharmaceutical composition comprising a carrier, and as active ingredient an effective amount of a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (I-a-1), (I-b-1)- (I-b-3), (I-c-1), (I-d-1), (I-e-1), (I-f-1), (I-a- 1-1), (I-b-1-1), (I-b-2-1), (I-b-3-1), (I-c-1-1), (I-d-1-1), (I-e-1-1), (I-f-1), (I-a-1-1- 1), (I-b-1-1-1)- (I-b-1-1-3), (I-b-2-1-1)- (I-b-2-1-3), (I-b-3-1-1)- (I-b-3-1-3), (I-c-1- 1-1), (I-d-1-1), (I-f-1-1), (I-a-1-1-1)- (
- the present invention relates to a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (I-a-1), (I-b-1)- (I-b-3), (I-c-1), (I-d-1), (I-e-1), (I-f-1), (I-a-1-1), (I-b-1-1), (I-b-2-1), (I-b-3-1), (I-c-1-1), (I-d-1-1), (I-e-1-1), (I-f-1), (I-a-1-1-1), (I-b-1-1-1)- (I-b-1-1-3), (I-b-2-1-1)- (I-b-2-1-3), (I-b-3-1-1)- (I-b-3-1-3), (I-c-1-1), (I-d-1-1), (I-f-1-1), (I-a-1-1-1-1)- (I-a-1-1-1-6), (I-b-1-1-1-1)- (I-b-1-1-1-1-1-1
- the present invention relates to a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1), (l-b-1)- (l-b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1 -1), (l-e-1-1), (l-f-1 -1), (l-a-1-1 -1), (l-b-1-1-1)- (l-b-1-1-3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3-1 -1 )- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1 -1 -1 ), (l-f-1 -1 ), (l-a-1-1 -1 ), (l-b-1-1)- (
- Crohn’s disease and ulcerative colitis inflammatory pulmonary diseases, rheumatoid arthritis, lupus nephritis, systemic lupus erythematosus and psoriasis and psoriasis arthritis), neurological disorders (such as Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, Charcot-Marie-Tooth neuropathy, amyotrophic lateral sclerosis and epilepsy), atherosclerosis and cardiovascular diseases, Sjogren Syndrome, renal allograft rejection, viral induced diseases, circulatory diseases, bone osteolysis and osteoporosis, osteoarthritis, sarcopenia, Langerhans cell histiocytosis, spinal cord injury, endometriosis, asthma and allergic asthma, eye diseases (such as retinopathies, age-related macular degeneration and uveitis) chronic and neuropathic pain, and fibro- proliferative diseases.
- neurological disorders such as Alzheimer’s disease, Parkinson’s disease, multiple
- the present invention further relates to a method of inhibiting protein kinase activity in a warm-blooded animal said method comprising the administration to an animal in need thereof, of a kinase-inhibitory effective amount of a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- ( l-b-3) , (l-c-1 ), (I- d-1), (l-e-1), (l-f-1 ), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1-1 ), (I- e-1), (l-f-1 -1 ), (l-a-1 -1 -1 ), (l-b-1 -1 )- (l-b-1 -1 -3), (l-b-2-1 -1 )- (l-b-2-1 -1 )- (l-
- the present invention further relates to a method of inhibiting protein kinase activity in a warm-blooded animal said method comprising the administration to an animal in need thereof, of a kinase-inhibitory effective amount of a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- (l-b-3), (l-c-1 ), (I- d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1-1), (I- e-1-1), (l-f-1 -1 ), (l-a-1 -1 -1 ), (l-b-1-1-1)- (l-b-1 -1 -3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3- 1-1)- (
- the present invention further relates to a method of treating a disease selected from cancer, metabolic disorders (such as diabetes), inflammatory and autoimmune disorders (such as inflammatory bowel diseases, e.g. Crohn’s disease and ulcerative colitis, inflammatory pulmonary diseases, rheumatoid arthritis, lupus nephritis, systemic lupus erythematosus and psoriasis and psoriasis arthritis), neurological disorders (such as Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, Charcot-Marie-Tooth neuropathy, amyotrophic lateral sclerosis and epilepsy), atherosclerosis and cardiovascular diseases, Sjogren Syndrome, renal allograft rejection, viral induced diseases, circulatory diseases, bone osteolysis and osteoporosis, osteoarthritis, sarcopenia, Langerhans cell histiocytosis, spinal cord injury, endometriosis, asthma and allergic asthma, eye diseases (such as retinopathies, age-
- radical positions on any molecular moiety used in the definitions may be anywhere on such moiety as long as it is chemically stable.
- Radicals used in the definitions of the variables include all possible isomers unless otherwise indicated.
- pyridyl includes 2-pyridyl, 3- pyridyl and 4-pyridyl
- pentyl includes 1 -pentyl, 2-pentyl and 3-pentyl.
- One embodiment comprises the compounds (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1), (l-b-1)- (l-b-3), (l-c-1), (l-d-1), (l-e-1 ), (l-f-1), (l-a- 1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1-1), (l-e-1), (l-f-1), (l-a-1 -1 -
- Another embodiment comprises the compounds (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- (I- b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1- 1), (l-d-1-1 ), (l-e-1 -1 ), (l-f-1 -1 ), (l-a-1-1 -1), (l-b-1 -1 -1)- (l-b-1 -1 -3), (l-b-2-1 -1 )- (I- b-2-1-3), (l-b-3-1-1)- (l-b-3-1-3), (l-c-1-1), (l-d-1-1), (l-f-1-1), (l-a-1 -1 -1 -1 )- (l-a-1 -1-1-6-
- stereochemically isomeric forms as used herein defines all the possible compounds made up of the same atoms bonded by the same sequence of bonds but having different three-dimensional structures which are not interchangeable, which the compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1), (l-b-1 )- (l-b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1 ), (l-c-1-1 ), (l-d-1 -1), (l-e-1), (l-f-1 -1), (l-a-1-1 -1), (l-b-1-1-1)- (l-b-1-1-3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3-1 -1 )- (l-b-3-1 -3), (l-b-b-3-1
- Said mixture may contain all diastereomers and/or enantiomers of the basic molecular structure of said compound. All stereochemically isomeric forms of the compounds of the present invention both in pure form or mixed with each other are intended to be embraced within the scope of the present invention.
- stereoisomerically pure concerns compounds or intermediates having a stereoisomeric excess of at least 80% (i.e. minimum 90% of one isomer and maximum 10% of the other possible isomers) up to a stereoisomeric excess of 100% (i.e. 100% of one isomer and none of the other), more in particular, compounds or intermediates having a stereoisomeric excess of 90% up to 100%, even more in particular having a stereoisomeric excess of 94% up to 100% and most in particular having a stereoisomeric excess of 97% up to 100%.
- enantiomerically pure and “diastereomerically pure” should be understood in a similar way, but then having regard to the enantiomeric excess, and the diastereomeric excess, respectively, of the mixture in question.
- Pure stereoisomeric forms of the compounds and intermediates of this invention may be obtained by the application procedures known in the art.
- enantiomers may be separated from each other by the selective crystallization of their diastereomeric salts with optically active acids or bases. Examples thereof are tartaric acid, dibenzoyltartaric acid, ditoluoyltartaric acid and camphorsulfonic acid.
- enantiomers may be separated by chromatographic techniques using chiral stationary phases.
- Said pure stereochemically isomeric forms may also be derived from the corresponding pure stereochemically isomeric forms of the appropriate starting materials, provided that the reaction occurs stereospecifically.
- a specific stereoisomer is desired, said compound will be synthesized by stereospecific methods of preparation. These methods will advantageously employ enantiomerically pure starting materials.
- a person skilled in the art is able to determine the absolute configuration of such compounds using art-known methods such as, for example, X-ray diffraction.
- the present invention is also intended to include all isotopes of atoms occurring on the present to a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- (l-b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1 ), (l-a-1 -1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c- 1-1), (l-d-1-1), (l-e-1-1), (l-f-1-1), (l-a-1-1-1 ), (l-b-1 -1 -1 )- (l-b-1-1-3), (l-b-2-1 -1 )- (l-b-2-1-3), (l-b-3-1-1 )- (l-b-3-1-3), (l-c-1-1 -1 ), (l-d-1 -1 -1), (l-f-1 -1), (l
- Isotopes include those atoms having the same atomic number but different mass numbers.
- isotopes of hydrogen include tritium and deuterium.
- isotopes of carbon include C-13 and C-14.
- the pharmaceutically acceptable acid and base addition salts as mentioned hereinabove are meant to comprise the therapeutically active non- toxic acid and base addition salt forms that the compounds (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1), (l-b-1)- (l-b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1 ), (l-d-1 -1), (l-e-1-1), (l-f-1 -1), (l-a-1-1 -1), (l-b-1-1-1)- (l-b-1-1-3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3-1 -1 )- (l-b-3-1 -3), (l-c-1 -1 ), (l-d-1 -1 ), (l-d
- the pharmaceutically acceptable acid addition salts can conveniently be obtained by treating the base form with such appropriate acid in an anion form.
- Appropriate anions comprise, for example, trifluoroacetate, acetate, benzenesulfonate, benzoate, bicarbonate, bitartrate, bromide, calcium edetate, camsyiate, carbonate, chloride, citrate, dihydrochloride, edetate, edisylate, estolate, esylate, fumarate, gluceptate, gluconate, glutamate, glycollylarsanilate, hexylresorcinate, hydrabamine, hydrobromide, hydrochloride, hydroxynaphthoate, iodide, isethionate, lactate, lactobionate, malate, maleate, mandelate, mesylate, methylbromide, methylnitrate, methylsulfate, mucate, napsylate, n
- Appropriate basic salts comprise those formed with organic cations such as benzathine, chloroprocaine, choline, diethanolamine, ethylenediamine, meglumine, procaine, and the like; and those formed with metallic cations such as aluminum, calcium, lithium, magnesium, potassium, sodium, zinc, and the like. Conversely said salt forms can be converted by treatment with an appropriate acid into the free form.
- addition salt as used hereinabove also comprises the solvates which the compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- (l-b-3), (l-c-1 ), (I- d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1-1), (I- e-1), (l-f-1 -1 ), (l-a-1 -1 -1 ), (l-b-1-1-1)- (l-b-1 -1 -3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3- 1-1)- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1 -1 -1 ), (l-f-1 -1 ), (l-a-1
- the present invention concerns a pharmaceutical composition
- a pharmaceutical composition comprising a therapeutically effective amount of a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- (l-b-3), (l-c-1), (l-d-1), (l-e-1 ), (l-f-1 ), (l-a- 1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1-1), (l-e-1), (l-f-1), (l-a-1 -1 -
- a therapeutically effective amount in this context is an amount sufficient to prophylactically act against, to stabilize or reduce illnesses mediated by protein kinases in ill subjects or subjects being at risk of being ill, in particular a protein kinase selected from the group consisting of CSF1R, FLT3, Kit, PDGFRB (PDGFR beta), PDGFRA (PDGFR alfa), ABL1, ACVR1B (ALK4), AKT1 (PKB alpha), AMPK A1/B1/G1, AURKA (Aurora A), BTK, CDK1/cyclin B, CHEK1 (CHK1), CSNK1G2 (CK1 gamma 2), CSNK2A1 (CK2 alpha 1), DYRK3, EGFR (ErbB1), EPHA2, ERBB2 (HER2), FGFR1, FRAP1 (mTOR), GSK3B (GSK3 beta), IGF1 R, IKBKB (IKK beta), INSR, IRAK4, JAK3, KDR
- illnesses mediated by protein kinases include in particular of illnesses mediated by protein kinases include in particular cancer, metabolic disorders (such as diabetes), inflammatory and autoimmune disorders (such as inflammatory bowel diseases, e.g. Crohn’s disease and ulcerative colitis, inflammatory pulmonary diseases, rheumatoid arthritis, lupus nephritis, systemic lupus erythematosus and psoriasis and psoriasis arthritis), neurological disorders (such as Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, Charcot-Marie-Tooth neuropathy, amyotrophic lateral sclerosis and epilepsy), atherosclerosis and cardiovascular diseases, Sjogren Syndrome, renal allograft rejection, viral induced diseases, circulatory diseases, bone osteolysis and osteoporosis, osteoarthritis, sarcopenia, Langerhans cell histiocytosis, spinal cord injury, endometriosis, asthma and allergic asthma, eye diseases (such
- this invention relates to a process of preparing a pharmaceutical composition as specified herein, which comprises intimately mixing a pharmaceutically acceptable carrier with a therapeutically effective amount of a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- ( l-b-3) , (l-c-1 ), (I- d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1-1), (I- e-1-1), (l-f-1 -1 ), (l-a-1 -1 -1 ), (l-b-1 -1 -1 )- (l-b-1 -1 -3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3- 1-1)- (l-b-3-1),
- compositions usually employed for systemically administering drugs there may be cited all compositions usually employed for systemically administering drugs.
- an effective amount of the particular compound, optionally in addition salt form or metal complex, as the active ingredient is combined in intimate admixture with a pharmaceutically acceptable carrier, which carrier may take a wide variety of forms depending on the form of preparation desired for administration.
- a pharmaceutically acceptable carrier which carrier may take a wide variety of forms depending on the form of preparation desired for administration.
- These pharmaceutical compositions are desirable in unitary dosage form suitable, particularly, for administration orally, rectally, percutaneously, or by parenteral injection.
- tablets and capsules represent the most advantageous oral dosage unit forms, in which case solid pharmaceutical carriers are obviously employed.
- the carrier will usually comprise sterile water, at least in large part, though other ingredients, for example, to aid solubility, may be included.
- injectable solutions for example, may be prepared in which the carrier comprises saline solution, glucose solution or a mixture of saline and glucose solution.
- injectable suspensions may also be prepared in which case appropriate liquid carriers, suspending agents and the like may be employed.
- solid form preparations which are intended to be converted, shortly before use, to liquid form preparations.
- the carrier optionally comprises a penetration enhancing agent and/or a suitable wetting agent, optionally combined with suitable additives of any nature in minor proportions, which additives do not introduce a significant deleterious effect on the skin.
- the present invention also provides a pharmaceutical composition adapted for administration by inhalation or insufflation through the mouth comprising a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- (l-b-3), (l-c-1 ), (l-d-1 ), (l-e-1 ), (l-f-1 ), (l-a-1 -1 ), (l-b-1 -1 ), (l-b-2-1 ), (l-b-3-1 ), (l-c-1 -1 ), (l-d-1 -1 ), (I- e-1 -1 ), (l-f-1 -1 ), (l-a-1 -1 -1 ), (l-b-1 -1 )- (l-b-1 -1 -3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3- 1 -1 )- (l-b-3-1 -3), (l-b
- Unit dosage form refers to physically discrete units suitable as unitary dosages, each unit containing a predetermined quantity of active ingredient calculated to produce the desired therapeutic effect in association with the required pharmaceutical carrier.
- unit dosage forms are tablets (including scored or coated tablets), capsules, pills, suppositories, powder packets, wafers, injectable solutions or suspensions and the like, and segregated multiples thereof.
- Illnesses and diseases treatable using the compounds and methods of the present invention include protein kinase mediated diseases like like cancer, metabolic disorders (such as diabetes), inflammatory and autoimmune disorders (such as inflammatory bowel diseases, e.g. Crohn’s disease and ulcerative colitis, inflammatory pulmonary diseases, rheumatoid arthritis, lupus nephritis, systemic lupus erythematosus and psoriasis and psoriasis arthritis), neurological disorders (such as Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, Charcot-Marie- Tooth neuropathy, amyotrophic lateral sclerosis and epilepsy), atherosclerosis and cardiovascular diseases, Sjogren Syndrome, renal allograft rejection, viral induced diseases, circulatory diseases, bone osteolysis and osteoporosis, osteoarthritis, sarcopenia, Langerhans cell histiocytosis, spinal cord injury, endometriosis, asthma and allergic asthma,
- Many of the compounds of this invention may show a favourable pharmacokinetic profile and have attractive properties in terms of bioavailability, including an acceptable half-life, AUC (area under the curve) and peak values and lacking unfavourable phenomena such as insufficient quick onset and tissue retention.
- the combinations of the present invention may be used as medicaments.
- Said use as a medicine or method of treatment comprises the systemic administration to ill subjects of an amount effective to combat the conditions associated with the illnesses. Consequently, the combinations of the present invention can be used in the manufacture of a medicament useful for treating, preventing or combating illness or disease associated with protein kinases including cancer, metabolic disorders (such as diabetes), inflammatory and autoimmune disorders (such as inflammatory bowel diseases, e.g.
- Crohn’s disease and ulcerative colitis inflammatory pulmonary diseases, rheumatoid arthritis, lupus nephritis, systemic lupus erythematosus and psoriasis and psoriasis arthritis), neurological disorders (such as Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, Charcot-Marie-Tooth neuropathy, amyotrophic lateral sclerosis and epilepsy), atherosclerosis and cardiovascular diseases, Sjogren Syndrome, renal allograft rejection, viral induced diseases, circulatory diseases, bone osteolysis and osteoporosis, osteoarthritis, sarcopenia, Langerhans cell histiocytosis, spinal cord injury, endometriosis, asthma and allergic asthma, eye diseases (such as retinopathies, age-related macular degeneration and uveitis) chronic and neuropathic pain, and fibro- proliferative diseases.
- neurological disorders such as Alzheimer’s disease, Parkinson’s disease, multiple
- terapéuticaally effective amount means that amount of active compound or component or pharmaceutical agent that elicits the biological or medicinal response in a tissue, system, animal or human that is being sought, in the light of the present invention, by a researcher, veterinarian, medical doctor or other clinician, which includes alleviation of the symptoms of the disease being treated.
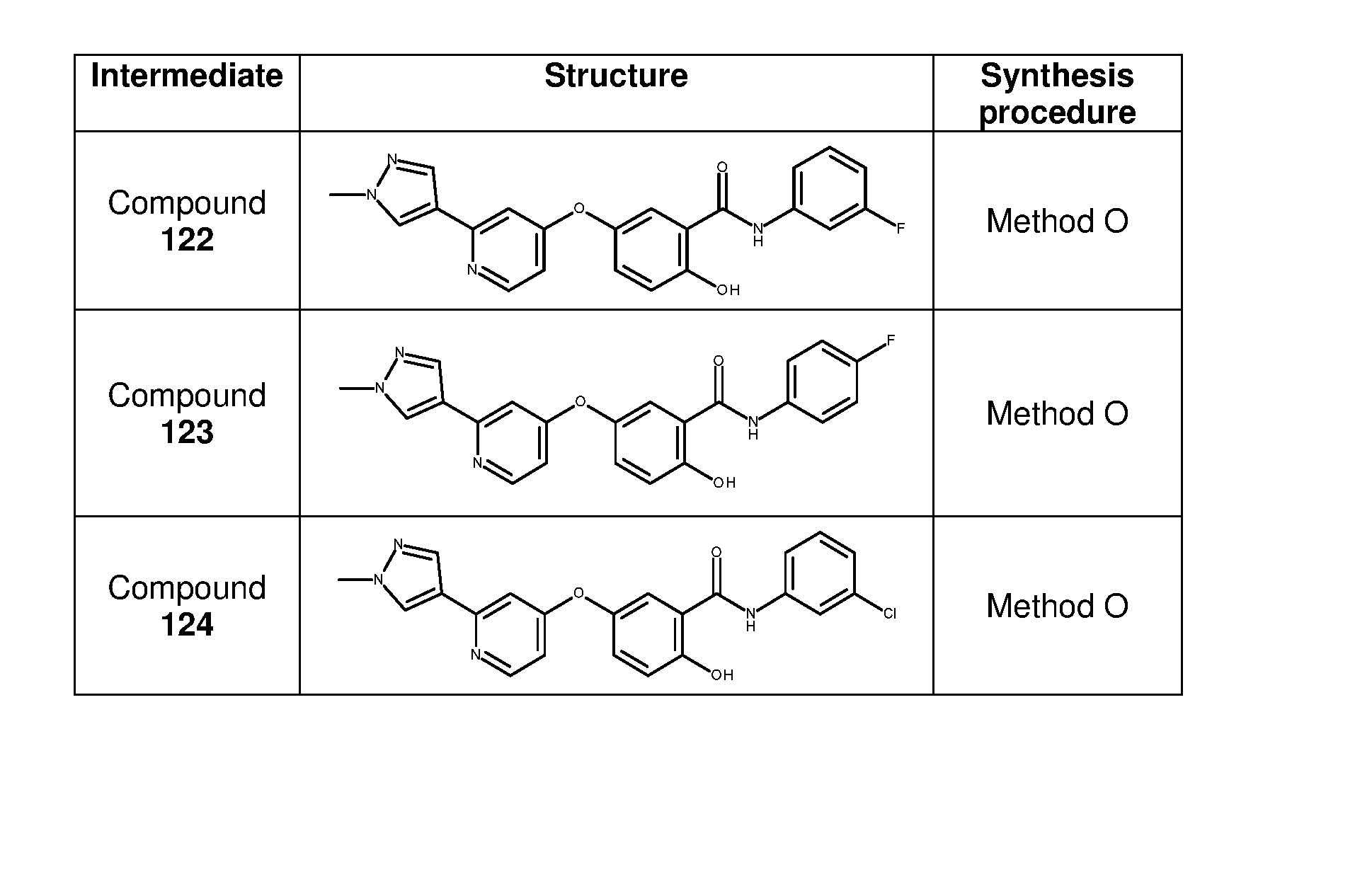
- Example 1 General procedure for the preparation of analogues 4-66 Cs 2 CO 3 , DMF 100°C Method A Pd(PPh 3 ) 4 , Cs 2 CO 3 1M, dioxane, 100°C, 1h Method B 1 2 Vilsmeier reagent, CH 2 Cl 2 , rt, 1h30 Method C RNH 2 , AcOH, MW, 100°C, 20 min Method D 4-66 3 Method A: To a solution of methyl 2-amino-5-hydroxybenzoate (527 mg, 3.15 mmol) in DMF (12 mL) under nitrogen was added solid cesium carbonate (2.569 g, 7.88 mmol) followed by 2-chloro-4-nitropyridine derivative (500 mg, 3.15 mmol).
- Method B To a stirred solution of intermediate 1 (400 mg, 1.44 mmol) in dioxane (7 mL) were added under nitrogen 1-methyl-4-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-1H-pyrazole (448 mg, 2.15 mmol), Cs 2 CO 3 1M (4.306 mL, 4.31 mmol) and Pd(PPh 3 ) 4 (166 mg, 0.14 mmol). The reaction mixture was stirred at 100°C for 1h.
- ESI-MS 440.10 (M+H) + .
- 5-(aminomethyl)-N-methyl-pyridin-2-amine was prepared by the following procedure: To a mixture 6-(methylamino)nicotinonitrile (251 mg, 1.8 mmol) in ammonia (solution 7N in MeOH, 15 mL) was added Raney nickel (slurry in water, 1g). After stirring overnight under a hydrogen atmosphere, the reaction mixture was filtered through a pad of Celite, rinsed with MeOH, and concentrated under reduced pressure to provide the expected compound which was used without further purification.
- ESI-MS 138.20 (M+H) + .
- ESI-MS 442.10 (M+H) + .
- (1-isopropylimidazol-4-yl)methanamine was prepared by hydrogenation of 1- isopropyl-1H-imidazole-4-carbonitrile (WO 2009/070485) according to example 22.
- 2-(cis-2,6-dimethylpiperidin-1-yl)ethanamine was prepared by the following procedure: To a solution of cis-2,6-dimethylpiperidine (100 mg, 0.88 mmol) and potassium carbonate (245 mg, 1.77 mmol) in acetonitrile (4.5 mL) was added 2- bromoacetonitrile (74 ⁇ L, 1.06 mmol). The resulting mixture was stirred at 60°C for 2h. The reaction mixture was diluted with DCM and washed with water. Organic layers were dried over Na 2 SO 4 , filtered and concentrated under reduced pressure.
- 2-(4,4-difluoro-1-piperidyl)ethanamine was prepared by the following procedure: To a solution of 4,4-difluoropiperidine (200 mg, 1.65 mmol) and potassium carbonate (456 mg, 3.30 mmol) in acetonitrile (5 mL) was added tert-butyl (2- bromoethyl)carbamate (444 mg, 1.98 mmol). The resulting mixture was stirred at 60°C for 4h. The reaction mixture was diluted with DCM and washed with water. Organic layers were dried over Na2SO4, filtered and concentrated under reduced pressure.
- the reaction mixture was diluted with EtOAc and washed twice with a saturated solution of NH 4 Cl.
- the organic layer was dried over Na 2 SO 4 , filtered and concentrated under reduced pressure.
- the crude product was purified by flash column chromatography (DCM/MeOH from 100/0 to 95/5) and reverse phase chromatography (H 2 O/MeOH: 0 to 100%) to give the expected compound.
- Example 3 General procedure for the preparation of analogues 79-87 3-Chlorobenzylamine, Vilsmeier reagent, AcOH, CH 2 Cl 2 , rt, 1h30 MW, 100°C, 20 min Method C Method D 1 77 78 1) Benzophenone imine, Pd 2 dba 3 , BINAP, t-BuONa, toluene, 90°C Boronic ester, Amine, 2) HCl (4M), dioxane, rt Pd(PPh 3 ) 4 , Cs 2 CO 3 1M, Pd 2 dba 3 , Xantphos, dioxane, 100°C Cs 2 CO 3 , dioxane, 100°C Method H Method B Method G R 2 COOH, EDC.HCl, DMAP, CH 2 Cl 2 , rt Method I 85-87 84 79-83 Preparation of methyl 5-[(2-chloro-4-pyridyl)oxy]-2
- reaction mixture was concentrated under reduced pressure and purified by flash column chromatography (DCM/MeOH from 100/0 to 97/3) and reverse phase chromatography (H2O/MeOH from 100/0 to 0/100) to give 69 mg of 6-[(2-amino- 4-pyridyl)oxy]-3-[(3-chlorophenyl)methyl]quinazolin-4-one 84 as a white powder in 34% yield.
- ESI-MS 378.90 (M+H) + .
- Example 4 General procedure for the preparation of analogues 104-119 NH2 NH2 NH2 O OH NH 4 OH, HOBt, NH2 O NaOH 2N, MeOH, O EDC.HCl, NMM, O 50°C, 2h DMF, rt, 2h O O O Method J
- Method K N N N N N N N N 2 N 88 N 89 RCOCl, TEA, RCOOH, DMAP, THF, rt, 2h HOBt, EDC.HCl, Method L DMF, rt, Method M O R N R N R NH N NH O Cs 2 CO 3 , MeI, O t-BuOK, t-BuOH, 80°C, 1 NH2 DMF h O , rt O O Method F Method N O N N N N N N N N N 118-119 104-117 N 90-103
- Method J To a solution of intermediate 2 (582 mg, 1.79 mmol) in MeOH (6 m
- Method K To a solution of 88 (138 mg, 0.45 mmol) in DMF (4 mL) cooled to 0°C with an ice bath were added HOBt (136 mg, 0.89 mmol), EDC.HCl (171 mg, 0.89 mmol) and N-methylmorpholine (98 ⁇ L, 0.89 mmol). Ammonium hydroxide (30%, 0.77 mmol) was then added dropwise. The ice bath was removed, and the reaction mixture was allowed to stir at room temperature for 2h. The reaction mixture was concentrated under reduced pressure.
- Example 5 General procedure for the preparation of analogues 145-167 Preparation of methyl 5-[(2-chloro-4-pyridyl)oxy]-2-hydroxy-benzoate (120): Intermediate 120 was synthesized from 2-chloro-4-nitropyridine (3.15 mmol) and methyl 2,5-dihydroxybenzoate (3.15 mmol) as a white powder in 60% yield according to the general method A.
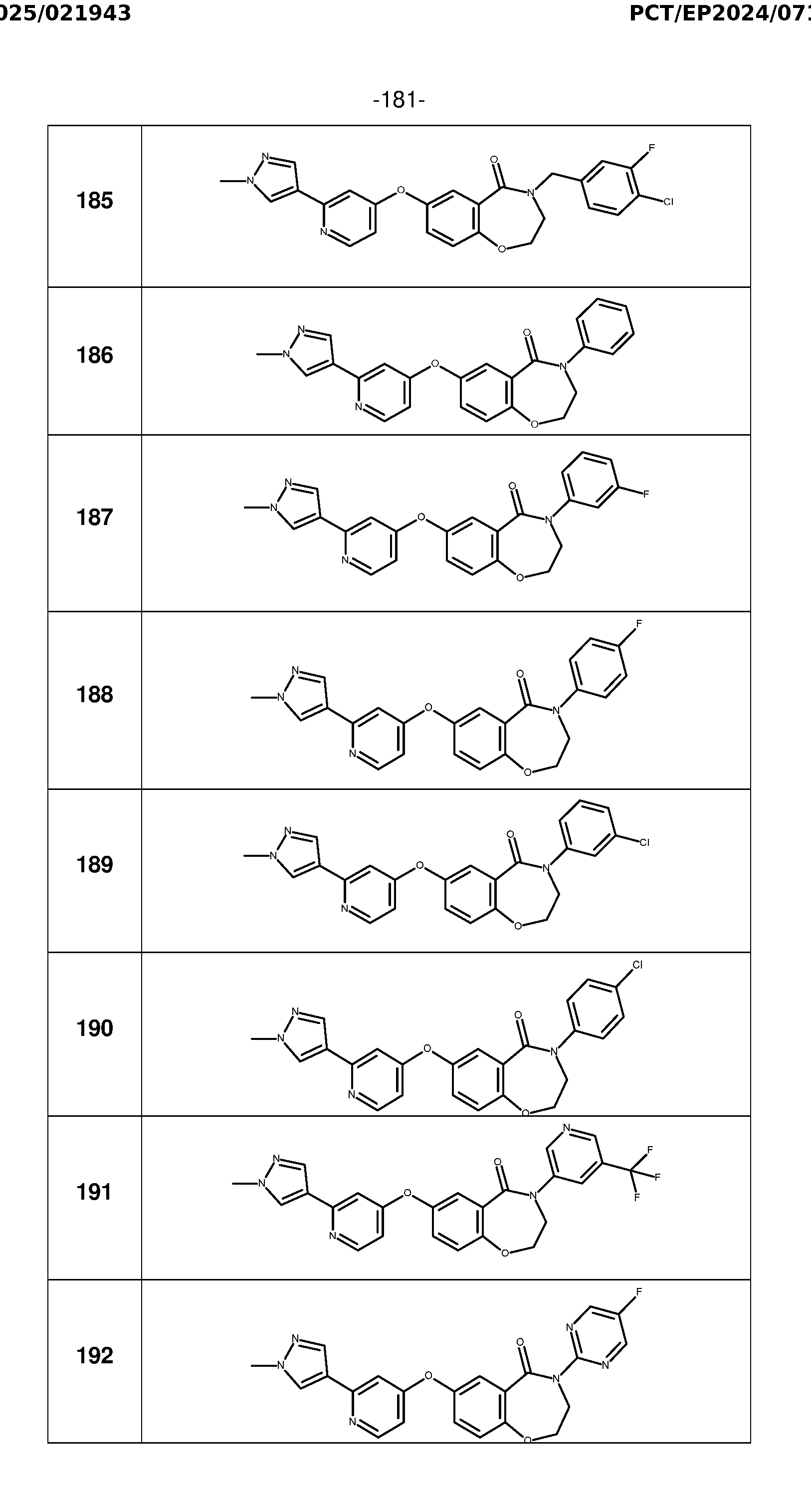
- Example 7 General procedure for the preparation of analogues 178-194 Method T: To a solution of derivative 169 (250 mg, 0.81 mmol) in DMF (13 mL) under nitrogen was added potassium carbonate (1.113 g, 8.05 mmol) and 1,2- dibromoethane (0.555 mL, 6.45 mmol). The reaction mixture was stirred at room temperature overnight. The reaction mixture was diluted with EtOAc and washed twice with a saturated solution of NH4Cl. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure.
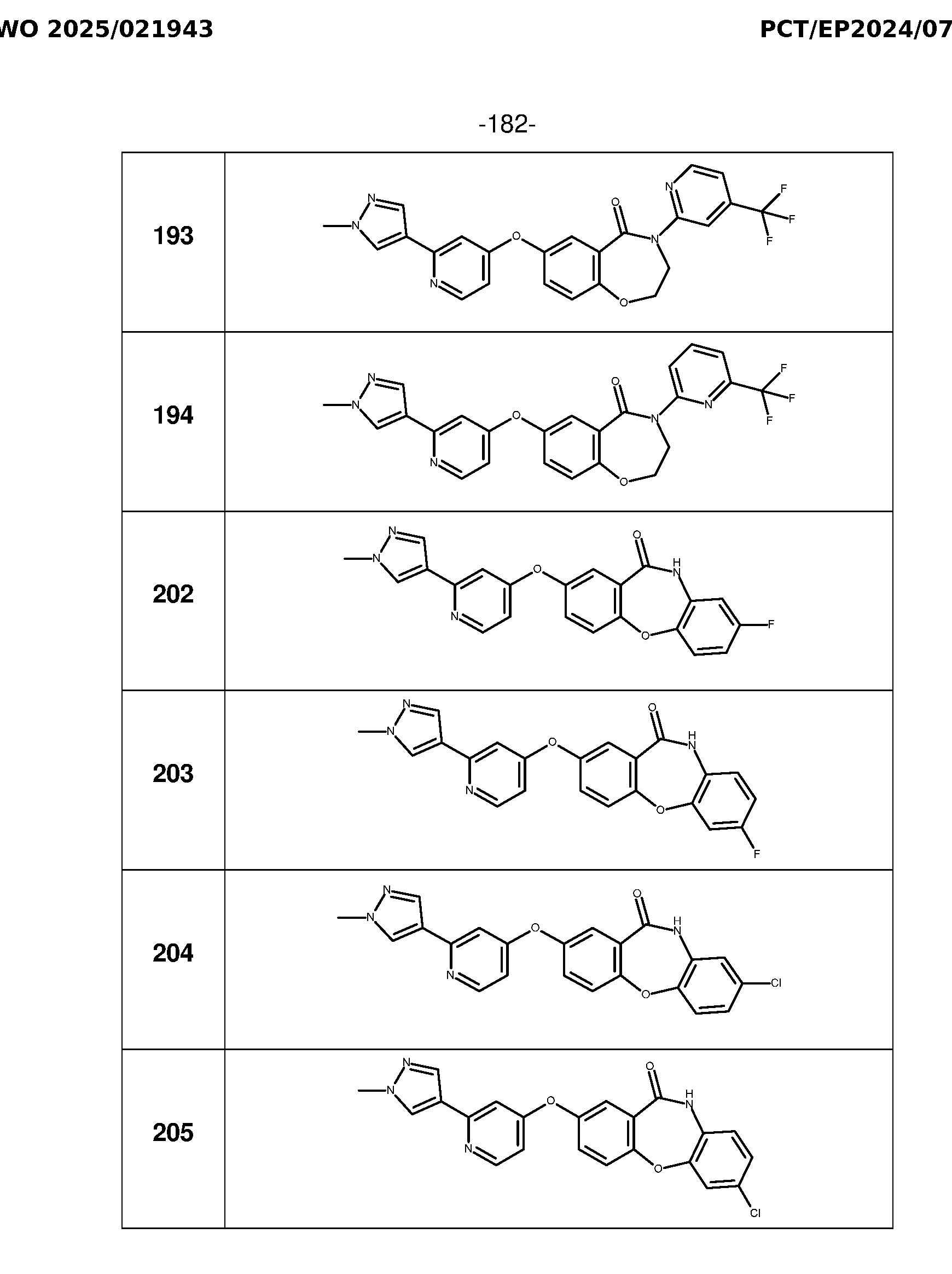
- Example 8 General procedure for the preparation of analogues 202-205 Cs 2 CO 3 , DMF 100°C Method A Pd(PPh 3 ) 4 , Cs 2 CO 3 1M, dioxane, 100°C, 1h Method B 195 196 NaOH 2N, MeOH, 50°C, 2h Method J RNH 2 , DBU, DMF EDC.HCl, 150°C HOBt, DMF, rt, Method W Method O 202-205 198-201 197 Preparation of methyl 5-[(2-chloro-4-pyridyl)oxy]-2-fluoro-benzoate (195): Intermediate 195 was synthesized from 2-chloro-4-nitropyridine (1.24 mmol) and methyl 2-fluoro-5-hydroxybenzoate (1.24 mmol) as a white powder in 88% yield according to the general method A.
- Example 9 Cell-based assays: Biological assay measuring cell proliferation in cell lines Compounds were evaluated in different cancer cell lines (Molm-13, M-NFS-60, HL-60 and P815) and in PDGFR ⁇ -BaF3 stable cell line. For each, cell proliferation were measured. The protocols of these assays are described below.
- MOLM-13 Exponential growing MOLM-13 cells (DSMZ, ACC-554) were seeded at 2.10 ⁇ 4 per 200 ⁇ L of complete medium.20 ⁇ L of test compound dilution were added to each well and the plates were incubated for 72 h at 37 °C, 5% CO2.
- WST-1 solution Cell Proliferation Reagent WST-1, Roche Applied Science.
- optical densities were measured at 450 nm and 620 nm for the background on microplate reader (Envision 2105, Perkinelmer).
- M-NFS-60 Exponential growing M-NFS-60 cells (ATCC, CRL-1838) were seeded at 10 ⁇ 4 per 200 ⁇ L of complete medium with beta-mercaptoethanol and M-CSF (62 ng/mL) or IL34 (500 ng/mL). Twenty ⁇ L of test compound dilution were added to each well and the plates were incubated for 72 h at 37 °C, 5% CO 2 . Untreated cells and positive control (0.5% triton X-100, for the last 15 min) served as reference for maximum and minimum viability.
- HL-60 Exponential growing HL-60 cells (DSMZ, ACC-3) were seeded at 2.10 ⁇ 4 per 200 ⁇ L of complete RPMI medium.20 ⁇ L of test compound dilution were added to each well and the plates were incubated for 72 h at 37 °C, 5% CO 2 .
- WST-1 solution Cell Proliferation Reagent WST-1, Roche Applied Science.
- optical densities were measured at 450 nm and 620 nm for the background on microplate reader (Envision 2105, Perkinelmer). IC50 were measured and some biological results of these assays are presented in the following table.
- P-815 Exponential growing P-815 cells (DSMZ, ACC-1) were seeded at 2.10 ⁇ 4 per 200 ⁇ L of complete RPMI medium. Twenty ⁇ L of test compound dilution were added to each well and the plates were incubated for 72 h at 37 °C, 5% CO2. Untreated cells and positive control (0,5% triton X-100, for the last 15 min) served as reference for maximum and minimum viability. At the end of incubation 100 ⁇ L of supernatant were removed and replaced by 10 ⁇ L of WST- 1 solution (Cell Proliferation Reagent WST-1, Roche Applied Science).
- BaF3-PDGFR ⁇ Exponential growing BaF3 cells stably transfected with a plasmid encoding the fusion gene GFP-ETV6-PDGFRA (ABMGood, T3082) were seeded at 5.10 ⁇ 3 per 200 ⁇ L of complete RPMI medium. Twenty ⁇ L of test compound dilution were added to each well and the plates were incubated for 72 h at 37 °C, 5% CO2.
- WST-1 solution Cell Proliferation Reagent WST-1, Roche Applied Science.
- optical densities were measured at 450 nm and 620 nm for the background on microplate reader (Envision 2105, Perkinelmer). IC50 were measured and some biological results of these assays are presented in the following table.
- HEK-CSF1R-STAT5-Luc Exponential growing HEK293T cells (ATCC® CRL- 3216TM), ectopically expressing human CSF1R receptor (Origene) and five copies of a STAT5 response element (STAT5 RE, promega) that drives transcription of the luciferase reporter were seeded at 5.10 ⁇ 3 per 20 ⁇ L of complete DMEM medium.
- test compound dilution 2 ⁇ L were added to each well and stimulated with 600 ng/ml of M-CSF.
- the plates were incubated for 24 h at 37 °C, 5% CO2. Unstimulated and stimulated cells served as reference for maximum and minimum induction.
- 25 ⁇ L of Steady-Glo® Luciferase Assay System (Promega) were added after 5 min of lysis, luminescence was measured on microplate reader (Envision 2105, Perkinelmer).
- HEK-CSF1R-WST-1 Exponential growing HEK293T cells (ATCC® CRL- 3216TM), were seeded at 5.10 ⁇ 3 per 200 ⁇ L of complete DMEM medium. The next day, twenty ⁇ L of test compound dilution were added to each well and the plates were incubated for 72 h at 37 °C, 5% CO2. Untreated cells and positive control (0,5% triton X-100, for the last 15 min) served as reference for maximum and minimum viability. At the end of incubation 100 ⁇ L of supernatant were removed and replaced by 10 ⁇ L of WST-1 solution (Cell Proliferation Reagent WST-1, Roche Applied Science).
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The present invention relates to a compound suitable for use as a kinase inhibitor.
Description
QUINAZOLINONE, BENZOXAZINONE AND BENZOXAZEPINONE DERIVATIVES AS PROTEIN KINASE INHIBITORS
Field of the invention
The present invention is in the field of medicinal chemistry and pharmaceuticals.
Background of the invention
Protein phosphorylation is the most common form of reversible post- translational modification, with an estimated 50% of all proteins undergoing phosphorylation. The phosphorylation state of any given protein is controlled by the coordinated action of specific kinases and phosphatases that add and remove phosphate, respectively. Particularly, protein kinases are a kind of protein phosphotransferases bringing the phosphate of ATP to the specific amino acid residue. They may conventionally be divided into five classes: tyrosine protein kinases, serine/threonine protein kinases, histidine protein kinases, tryptophan protein kinases and aspartyl/glutamoyl protein kinases.
Signaling networks that employ phosphorylation to modulate target activities have been shown to be critically involved in all aspects of cellular function, the abnormal activation of protein phosphorylation is frequently either a driver or direct consequence of the disease. Kinase signaling pathway dysregulation is associated with cancer, inflammatory disease, cardiovascular disease, neurodegenerative disease, and metabolic disease, through the constitutive activation of many downstream pathways, such as phosphatidyl- inositol 3-kinase/v-akt murine thymoma viral oncogene homolog 1 (PIK3/AKT), mitogen-activated protein kinase/extracellular signal regulated kinase (MAPK/ERK) and signal transducer and activator of transcription 5 (STAT5). Consequently, protein kinases represent important therapeutic targets.
In tumours, the abnormal oncogenic activation of protein kinases derives from multiple types of genetic and epigenetic changes. These alterations result in increased specific activity of the kinase itself, its overexpression, or the loss of negative regulation leading to uncontrolled cellular growth and sustained malignant behaviour. The signalling networks operating in cancer cells can also contribute to innate or acquired resistance
to treatment, since they are able to create the most common or rare oncogenic mutations different from tumour to tumour. Hence, the search for small-molecule inhibitors targeting the altered protein kinase molecules in tumour cells has become a major research focus in the academia and pharmaceutical companies.
Such inhibitors can be products that are derived (isolated) from sources such as plants, animals or microorganisms, or can be small- molecules that are designed (synthetized).
WO 2004/022572 discloses classes of biologically active compounds interacting with kinases, and the preparation of these compounds.
In cancerology, there are currently multiple examples of small molecule kinase inhibitors with both selectivity and suitable pharmaceutical properties that have produced meaningful clinical benefit. For instance, pexidartinib is utilized to inhibit the colony-stimulating factor-1 receptor (CSF1 R), the KIT proto-oncogene receptor tyrosine kinase (KIT) and the FMS-like tyrosine kinase 3 (FLT3) in, for example, the treatments of patients with symptomatic tenosynovial giant cell tumors (TGCT); edicotinib to inhibit the CSF1 R and currently in phase II for acute myeloid leukemia, cognition disorders or Crohn’s disease; or nintedanib to inhibit the endothelial growth factor receptor (VEGFR), fibroplast growth factor receptor (FGFR), platelet-derived growth factor receptor (PDGFR) and CSF1 R in, for example, the treatment of idiopathic pulmonary fibrosis.
There is still a great need to develop potent inhibitors of protein kinase that are useful in treating the various protein kinase-related conditions.
In this sense, WO 201 1/090738 A2 discloses compounds that are able to inhibit B-RAF and B-RAF mutations and methods for treating diseases related to B-RAF and B-RAF mutation modulation.
US 2009/0325945 describes active compounds, specifically, certain imidazo[4,5-b]pyridin-2-one and oxazolo[4,5-b]pyridin-2-one compounds and analogs inhibiting RAF (e.g., B-RAF) activity in a cell, in vitro or in vivo, inhibiting receptor tyrosine kinase (RTK) activity, such as FGFR, Tie, VEGFR and/or Eph activity, for example, FGFR-1 , FGFR-2, FGFR-3, Tie2, VEGFR-2 and/or EphB2 activity, in a cell, in vitro or in vivo.
US 2015/0182526: This document describes therapeutic compounds for
treating proliferative disorders, cancer, etc., and more specifically certain pyrido[2,3-b]pyrazin-8-substituted compounds, which, inter alia, inhibit RAF (e.g., B-RAF) activity and inhibit receptor tyrosine kinase (RTK) activity. However, despite the growing effort in developing new protein kinase inhibitors-based therapies, there is still a need for protein kinase inhibitors which may overcome the disadvantages of current protein kinase therapies such as side effects, limited efficacy, the emerging of resistance, and compliance failures. Summary of the invention The inventors have surprisingly found that the use of protein kinase inhibitors according to the invention allows to provide an improved treatment of dysregulated protein kinase related diseases, by developing a therapy that is more effective, that reduces side effects, that limits the emerging of resistance and that facilitates compliance. Therefore, the present invention provides a compound suitable for use as a protein kinase inhibitor according to formula (I) [compounds (C) herein after], or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof Formula (I) wherein: - each of R1, independently from each other and at each occurrence, is selected from hydrogen, or C1-6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of W, independently from each other and at each occurrence is a bond, or is selected from the group consisting of O, N(R11), and N(R11)(C=O); wherein each of R11, independently from each other and at each occurrence is selected from the group consisting of hydrogen and C1-6 alkyl; - each of X, independently from each other and at each occurrence is selected from O, S, or NR7, wherein R7 is selected from hydrogen or C1-
6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of R2, independently from each other and at each occurrence is a moiety selected from the group consisting of (R2-a) to (R2-f): (R2-a) (R2-b) (R2-c) (R2-d) (R2-e) (R2-f) wherein: - each of Y independently from each other and at each occurrence is selected from the group consisting of O, S, and NR7, wherein R7 is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of R5, and R5’, independently from each other and at each occurrence is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of n5, independently from each other and at each occurrence, is an integer equal to 0, 1 or 2; - R6 is selected from the group consisting of halo, NO2, C1-6 alkyl, C2-4 alkenyl, C2-4 alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, CF3, CN, OR11, SR11, N(R11)2,, NR11COR12, COR11, C(O)OR11, CON(R11)2,
OC(O)R11, and OCON(R11)2, wherein each of R11 and R12, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and CF3, wherein said alkyl, alkenyl, and alkynyl substituents are optionally substituted with at least one halogen atom. - each of n6, independently from each other and at each occurrence, is an integer equal to 0, 1 or 2; - each of Z independently from each other and at each occurrence is selected from the group consisting of O, S and NR7, wherein R7 is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of R4 and R4’, independently from each other and at each occurrence, are selected from hydrogen, or C1-6 alkyl, wherein said alkyl is optionally substituted with a halogen atom, or R4-C-R4’ can form C=O; - each of z, independently from each other and at each occurrence, is an integer equal to 0, 1, 2, 3 or 4; - each of A, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl, wherein said alkyl, alkenyl, alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl are optionally substituted with one or more substituents independently selected from the group consisting of halo, NO2, C1-6 alkyl, C2-4 alkenyl, C2-4 alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, CF3, CN, OR11, SR11, N(R11)2, OC(R11)2O, OC(R11)2C(R11)2O, S(O)R12, SO2R12, SO2N(R11)2, S(O)3R11, P(=O)(OR11)2, P(=O)(R11)2, NR11COR12, COR11, C(O)OR11, CON(R11)2, OC(O)R11, and OCON(R11)2, and each optional alkyl, alkenyl, alkynyl, cycloalkyl, aryl, heterocyclyl, and heteroaryl substituents is further optionally substituted with halo, NO2, CF3, N(R11)2, COR11, CON(R11)2, OC(O)R11, CN, or OR11; and wherein each of R11 and R12, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and CF3, wherein said alkyl, alkenyl, and alkynyl, substituents are optionally substituted at least one halogen atom;
wherein said cycloalkyl is a monocyclic, bicyclic or tricyclic ring system of 3-6 ring members per ring; said heterocyclyl is a saturated, partially saturated or completely unsaturated monocycle, bicycle or tricycle containing 3 to 12 carbon atoms and 1 or 2 heteroatoms independently selected from O or N; said aryl is phenyl, naphthyl or anthracenyl optionally carbocyclic fused with a cycloalkyl or heterocyclyl of 5-7 ring members; said heteroaryl is a monocyclic ring structure containing 5 or 6 ring atoms, or a bicyclic aromatic group having 8 to 10 atoms, containing 1-3 heteroatoms independently selected from O or N. The present invention further relates to a pharmaceutical composition comprising a carrier, and as active ingredient an effective amount of a compound as defined in any one of the embodiments presented herein. The present invention relates to a compound as defined in any one of the embodiments presented herein, for use as a medicament. The present invention relates to a compound as defined in any one of the embodiments presented herein for use in the treatment of a disease selected from cancer, metabolic disorders (such as diabetes), inflammatory and autoimmune disorders (such as inflammatory bowel diseases, e.g. Crohn’s disease and ulcerative colitis, inflammatory pulmonary diseases, rheumatoid arthritis, lupus nephritis, systemic lupus erythematosus and psoriasis and psoriasis arthritis), neurological disorders (such as Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, Charcot-Marie-Tooth neuropathy, amyotrophic lateral sclerosis and epilepsy), atherosclerosis and cardiovascular diseases, Sjogren Syndrome, renal allograft rejection, viral induced diseases, circulatory diseases, bone osteolysis and osteoporosis, osteoarthritis, sarcopenia, Langerhans cell histiocytosis, spinal cord injury, endometriosis, asthma and allergic asthma, eye diseases (such as retinopathies, age-related macular degeneration and uveitis) chronic and neuropathic pain, and fibro- proliferative diseases. The present invention relates to a compound as defined in any one of the embodiments presented herein, for use in the treatment of pain sensitization. The present invention further relates to a method of inhibiting protein kinase activity in a warm-blooded animal said method comprising the administration to an animal in need thereof, of a kinase-inhibitory effective
amount of a compound according to any one of the embodiments presented herein. The present invention further relates to a method of treating a disease selected from cancer, metabolic disorders (such as diabetes), inflammatory and autoimmune disorders (such as inflammatory bowel diseases, e.g. Crohn’s disease and ulcerative colitis, inflammatory pulmonary diseases, rheumatoid arthritis, lupus nephritis, systemic lupus erythematosus and psoriasis and psoriasis arthritis), neurological disorders (such as Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, Charcot-Marie-Tooth neuropathy, amyotrophic lateral sclerosis and epilepsy), atherosclerosis and cardiovascular diseases, Sjogren Syndrome, renal allograft rejection, viral induced diseases, circulatory diseases, bone osteolysis and osteoporosis, osteoarthritis, sarcopenia, Langerhans cell histiocytosis, spinal cord injury, endometriosis, asthma and allergic asthma, eye diseases (such as retinopathies, age-related macular degeneration and uveitis) chronic and neuropathic pain, and fibro-proliferative diseases in a warm-blooded animal said method comprising the administration to an animal in need thereof of an effective amount of a compound according to any one of the embodiments presented herein. Detailed description of the invention A first aspect of the present invention relates to a compound suitable for use as a protein kinase inhibitor according to formula (I) [compounds (C) herein after], or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof Formula (I) wherein: - each of R1, independently from each other and at each occurrence, is selected from hydrogen, or C1-6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of W, independently from each other and at each occurrence is a bond, or is selected from the group consisting of O, N(R11), and
N(R11)(C=O); wherein each of R11, independently from each other and at each occurrence is selected from the group consisting of hydrogen and C1-6 alkyl; - each of X, independently from each other and at each occurrence is selected from O, S, or NR7, wherein R7 is selected from hydrogen or C1- 6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of R2, independently from each other and at each occurrence is a moiety selected from the group consisting of (R2-a) to (R2-f): (R2-a) (R2-b) (R2-c) (R2-d) (R2-e) (R2-f) wherein: - each of Y independently from each other and at each occurrence is selected from the group consisting of O, S, and NR7, wherein R7 is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of R5, and R5’, independently from each other and at each occurrence is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom;
- each of n5, independently from each other and at each occurrence, is an integer equal to 0, 1 or 2; - R6 is selected from the group consisting of halo, NO2, C1-6 alkyl, C2-4 alkenyl, C2-4 alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, CF3, CN, OR11, SR11, N(R11)2, NR11COR12, COR11, C(O)OR11, CON(R11)2, OC(O)R11, and OCON(R11)2, wherein each of R11 and R12, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and CF3, wherein said alkyl, alkenyl, and alkynyl substituents are optionally substituted with at least one halogen atom. - each of n6, independently from each other and at each occurrence, is an integer equal to 0, 1 or 2; - each of Z independently from each other and at each occurrence is selected from the group consisting of O, S and NR7, wherein R7 is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of R4 and R4’, independently from each other and at each occurrence, are selected from hydrogen, or C1-6 alkyl, wherein said alkyl is optionally substituted with a halogen atom, or R4-C-R4’ can form C=O; - each of z, independently from each other and at each occurrence, is an integer equal to 0, 1, 2, 3 or 4; - each of A, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl, wherein said alkyl, alkenyl, alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl are optionally substituted with one or more substituents independently selected from the group consisting of halo, NO2, C1-6 alkyl, C2-4 alkenyl, C2-4 alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, CF3, CN, OR11, SR11, N(R11)2, OC(R11)2O, OC(R11)2C(R11)2O, S(O)R12, SO2R12, SO2N(R11)2, S(O)3R11, P(=O)(OR11)2, P(=O)(R11)2, NR11COR12, COR11, C(O)OR11, CON(R11)2, OC(O)R11, and OCON(R11)2, and each optional alkyl, alkenyl, alkynyl, cycloalkyl, aryl, heterocyclyl, and heteroaryl substituents is further optionally substituted with halo, NO2, CF3, N(R11)2, COR11, CON(R11)2, OC(O)R11, CN, or OR11; and
wherein each of R11 and R12, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and CF3, wherein said alkyl, alkenyl, and alkynyl, substituents are optionally substituted at least one halogen atom; wherein said cycloalkyl is a monocyclic, bicyclic or tricyclic ring system of 3- 6 ring members per ring; said heterocyclyl is a saturated, partially saturated or completely unsaturated monocycle, bicycle or tricycle containing 3 to 12 carbon atoms and 1 or 2 heteroatoms independently selected from O or N; said aryl is phenyl, naphthyl or anthracenyl optionally carbocyclic fused with a cycloalkyl or heterocyclyl of 5-7 ring members; said heteroaryl is a monocyclic ring structure containing 5 or 6 ring atoms, or a bicyclic aromatic group having 8 to 10 atoms, containing 1-3 heteroatoms independently selected from O or N. In a preferred embodiment of the present invention, each of R1 in compound (C) of formula (I), independently from each other and at each occurrence, is selected from hydrogen or C1-6 alkyl. More preferably, each of R1, independently from each other and at each occurrence, is selected from hydrogen, or C1-4 alkyl. Even more preferably, each of R1, independently from each other and at each occurrence, is C1-4 alkyl. Even more preferably, each of R1, independently from each other and at each occurrence, is methyl; In a preferred embodiment of the present invention, each of W in compound (C) of formula (I), independently from each other and at each occurrence is a bond, or is selected from the group consisting of O, N(R11), and N(R11)(C=O); wherein each of R11, independently from each other and at each occurrence is selected from the group consisting of hydrogen and C1-4 alkyl. More preferably, each of W, independently from each other and at each occurrence is a bond, or is selected from the group consisting of O, N(H), and N(H)(C=O). In a preferred embodiment of the present invention, each of X in compound (C) of formula (I), independently from each other and at each
occurrence is selected from O, S, or NR7, wherein R7 is selected from hydrogen or C1-6 alkyl. More preferably, each of X, independently from each other and at each occurrence is selected from O, S, or NR7, wherein R7 is selected from hydrogen or C1-4 alkyl. Even more preferably, each of X, independently from each other and at each occurrence is selected from O, S, or NH. Even more preferably, each of X, independently from each other and at each occurrence is O. In a preferred embodiment of the present invention, each of Y in compound (C) of formula (I), independently from each other and at each occurrence is selected from the group consisting of O, S, and NR7, wherein R7 is selected from hydrogen or C1-6 alkyl. More preferably, each of Y independently from each other and at each occurrence is selected from the group consisting of O, S, and NR7, wherein R7 is selected from hydrogen or C1-4 alkyl. Even more preferably, each of Y independently from each other and at each occurrence is selected from the group consisting of O, S, and NH. Even more preferably, each of Y independently from each other and at each occurrence is selected from O, or NH. In a preferred embodiment of the present invention, each of R5 and R5’ in compound (C) of formula (I), independently from each other and at each occurrence is selected from hydrogen or C1-6 alkyl. More preferably, each of R5, and R5’, independently from each other and at each occurrence is selected from hydrogen or C1-4 alkyl. Even more preferably, each of R5, and R5’, independently from each other and at each occurrence is selected from hydrogen or methyl. In a preferred embodiment of the present invention, each of n5 in compound (C) of formula (I), independently from each other and at each occurrence is an integer equal to 0 or 1. More preferably, each of n5, independently from each other and at each occurrence, is an integer equal to 0. In a preferred embodiment of the present invention, R6 in compound (C) of formula (I), is selected from the group consisting of halo, NO2, C1-6 alkyl, CF3,
CN, OR11, SR11, N(R11)2, COR11, C(O)OR11, CON(R11)2, OC(O)R11, and wherein R11 is independently selected from the group consisting of hydrogen, C1-6 alkyl, and CF3. More preferably, R6 is selected from the group consisting of halo, NO2, C1-6 alkyl, CF3, CN, OR11, and N(R11)2, and wherein R11 is independently selected from the group consisting of hydrogen, and C1-6 alkyl. Even more preferably, R6 is selected from the group consisting of halo, NO2, C1-6 alkyl, CF3, CN, OR11, and N(R11)2, and wherein R11 is independently selected from the group consisting of hydrogen, and C1-4 alkyl. Even more preferably, R6 is halo. In a preferred embodiment of the present invention, each of n6 in compound (C) of formula (I), independently from each other and at each occurrence, is an integer equal to 0 or 1. More preferably, each of n5, independently from each other and at each occurrence, is an integer equal to 0. In a preferred embodiment of the present invention, each of Z in compound (C) of formula (I), independently from each other and at each occurrence is selected from the group consisting of O, S, and NR7, wherein R7 is selected from hydrogen or C1-6alkyl. More preferably, each of Z independently from each other and at each occurrence is selected from the group consisting of O, S, and NR7, wherein R7 is selected from hydrogen or C1-4 alkyl. Even more preferably, each of Z independently from each other and at each occurrence is NR7, wherein R7 is selected from hydrogen or C1-4 alkyl. Even more preferably, each of Z, independently from each other and at each occurrence, is NR7, wherein R7 is selected from hydrogen or methyl. In a preferred embodiment of the present invention, each of R4 and R4’ in compound (C) of formula (I), independently from each other and at each occurrence are selected from hydrogen or C1-6 alkyl, or R4-C-R4’ can form C=O. More preferably, each of R4 and R4’, independently from each other and at each occurrence, are selected from hydrogen or C1-4 alkyl, or R4-C-R4’ can form C=O.
Even more preferably, each of R4 and R4’, independently from each other and at each occurrence, are selected from the group consisting of hydrogen, methyl, ethyl, and butyl, or R4-C-R4’ can form C=O. In a preferred embodiment of the present invention, each of z in compound (C) of formula (I), independently from each other and at each occurrence, is an integer equal to 0, 1, 2 or 3. More preferably, z, independently from each other and at each occurrence, is an integer equal to 0, 1, or 2; In a preferred embodiment of the present invention, each of A in compound (C) of formula (I), independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl, wherein said alkyl, alkenyl, alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl are optionally substituted with one or more substituents independently selected from the group consisting of halo, NO2, C1-6 alkyl, C2-4 alkenyl, C2-4 alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, CF3, CN, OR11, SR11, N(R11)2, OC(R11)2O, OC(R11)2C(R11)2O, S(O)R12, SO2R12, SO2N(R11)2, S(O)3R11, NR11COR12, COR11, C(O)OR11, CON(R11)2, OC(O)R11, and OCON(R11)2, and each optional alkyl, alkenyl, alkynyl, cycloalkyl, aryl, heterocyclyl, and heteroaryl substituents is further optionally substituted with halo, NO2, CF3, N(R11)2, CN, or OR11; and wherein each of R11 and R12, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and CF3. More preferably, each of A, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl, wherein said alkyl, alkenyl, alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl are optionally substituted with one or more substituents independently selected from the group consisting of halo, C1-6 alkyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, CF3, CN, OR11, N(R11)2, OC(R11)2O, OC(R11)2C(R11)2O, S(O)R12, SO2R12, SO2N(R11)2, COR11, C(O)OR11, CON(R11)2, OC(O)R11, and OCON(R11)2; and wherein each of R11 and R12, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, and CF3.
Even more preferably, each of A, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl, wherein said alkyl, alkenyl, alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl are optionally substituted with one or more substituents independently selected from the group consisting of halo, C1-6 alkyl, CF3, CN, OR11, N(R11)2, OC(R11)2O, SO2N(R11)2; and wherein each of R11 and R12, independently from each other and at each occurrence, is selected from the group consisting of hydrogen or C1-4 alkyl such as methyl, ethyl, propyl, isopropyl, butyl, tert-butyl and isobutyl. In one embodiment of the present invention, A in compound (C) of formula (I) is independently selected from the following moieties:
wherein each of halo is F, Cl, Br, or I, and each of R is hydrogen or C1-4 alkyl such as methyl, ethyl, propyl, isopropyl, butyl, tert-butyl is isobutyl, preferably R is hydrogen, methyl, ethyl, 2-methylpropyl or tert-butyl. According to one embodiment of the present invention, the compound (C) according to formula (I), or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, preferably is a compound of formulae (I-a-1) [compound (C) of class (I-a) herein-after]: Formula (I-a-1) wherein R1, X, Y, R5, R5’, R4, R4’, z and A have the same meaning as defined above for formula (I). According to one embodiment of the present invention, the compound (C) according to formula (I), or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, preferably is a compound chosen among those of formulae (I-b-1) to (I-b-3) [compound (C) of class (I-b) herein-after]: Formula (I-b-1) Formula (I-b-2) Formula (I-b-3) wherein R1, X, R5, R4, R4’, z and A have the same meaning as defined above for formula (I).
According to one embodiment of the present invention, the compound (C) according to formula (I), or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, preferably is a compound according to formula (I-c-1) [compound (C) of class (I-c) herein-after]: Formula (I-c-1) wherein R1, X, Z, R4, R4’, z and A have the same meaning as defined above for formula (I). According to one embodiment of the present invention, the compound (C) according to formula (I), or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, preferably is a compound of formulae (I-d-1) [compound (C) of class (I-d) herein-after]: Formula (I-d-1) wherein R1, X, Y, R5, n5, R4, R4’, z and A have the same meaning as defined above for formula (I). According to one embodiment of the present invention, the compound (C) according to formula (I), or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, preferably is a compound of formulae (I-e-1) [compound (C) of class (I-e) herein-after]: Formula (I-e-1)
wherein R1, X, Y, Z, R5, n5, R4, R4’, z and A have the same meaning as defined above for formula (I). According to one embodiment of the present invention, the compound (C) according to formula (I), or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, preferably is a compound of formulae (I-f-1) [compound (C) of class (I-f) herein-after]: Formula (I-f-1) wherein R1, X, Y, Z, R6 and n6 have the same meaning as defined above for formula (I). In compounds (C) according to the present invention, preferably X is O, Y is O, and Z is NR7. Preferred compounds (C) of class (I-a) are thus compounds of formula (I-a-1-1) herein below: Formula (I-a-1-1) wherein R1, R5, R5’, R4, R4’, z and A have the same meaning as defined above for formula (I). In one embodiment of the present invention, the compounds (C) of class (I-a) are selected from those of formula (I-a-1-1). Preferred compounds (C) of class (I-b) are thus selected from those of formulae (I-b-1-1) to (I-b-3-1) herein below: Formula (I-b-1-1)
Formula (I-b-2-1) Formula (I-b-3-1) wherein R1, R5, R4, R4’, z and A have the same meaning as defined above for formula (I). In one embodiment of the present invention, the compounds (C) of class (I-b) are selected from those of formula (I-b-1-1), (I-b-2-1) or (I-b-3-1). Preferred compounds (C) of class (I-c) are thus compounds of formula (I-c-1-1) herein-below: Formula (I-c-1-1) wherein R1, R4, R4’, z and A have the same meaning as defined above for formula (I), and wherein R7 is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with halogen atom, preferably R7 is hydrogen or methyl, more preferably R7 is hydrogen. In one embodiment of the present invention, the compounds (C) of class (I-c) are selected from those of formula (I-c-1-1). Preferred compounds (C) of class (I-d) are thus compounds of formula (I-d-1-1) herein-below: Formula (I-d-1-1) wherein R1, R5, n5, R4, R4’, z and A have the same meaning as defined above for formula (I).
In one embodiment of the present invention, the compounds (C) of class (I-d) are selected from those of formula (I-d-1-1). Preferred compounds (C) of class (I-e) are thus compounds of formula (I-e-1-1) herein-below: Formula (I-e-1-1) wherein R1, R5, n5, R4, R4’, z and A have the same meaning as defined above for formula (I), and wherein R7 is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with halogen atom, preferably R7 is hydrogen or methyl, more preferably R7 is hydrogen. In one embodiment of the present invention, the compounds (C) of class (I-e) are selected from those of formula (I-e-1-1). Preferred compounds (C) of class (I-f) are thus compounds of formula (I- f-1-1) herein-below: Formula (I-f-1-1) wherein R1, R6, and n6 have the same meaning as defined above for formula (I), and wherein R7 is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with halogen atom, preferably R7 is hydrogen or methyl, more preferably R7 is hydrogen. In one embodiment of the present invention, the compounds (C) of class (I-f) are selected from those of formula (I-f-1-1). In a preferred embodiment of the present invention, the compound (C) of class (I-a) is selected from those of formula (I-a-1-1-1) herein-below: Formula (I-a-1-1-1)
wherein R1, R5, R5’, R4, R4’, z and A have the same meaning as defined above for formula (I). In a preferred embodiment of the present invention, the compound (C) of formula (I-b-1-1) of class (I-b) is selected from those of formula (I-b-1-1-1) to formula (I-b-1-1-3) herein-below: Formula (I-b-1-1-1) Formula (I-b-1-1-2) Formula (I-b-1-1-3) wherein R1, R5, R4, R4’, z and A have the same meaning as defined above for formula (I). In a preferred embodiment of the present invention, the compound (C) of formula (I-b-2-1) of class (I-b) is selected from those of formulae (I-b-2-1-1) to (I- b-2-1-3) herein-below: Formula (I-b-2-1-1) Formula (I-b-2-1-2)
Formula (I-b-2-1-3) wherein R1, R5, R4, R4’, z and A have the same meaning as defined above for formula (I). In a preferred embodiment of the present invention, the compound (C) of formula (I-b-3-1) of class (I-b) is selected from those of formula (I-b-3-1-1) to (I- b-3-1-3) herein-below: Formula (I-b-3-1-1) Formula (I-b-3-1-2) Formula (I-b-3-1-3) wherein R1, R5, R4, R4’, z and A have the same meaning as defined above for formula (I). In a preferred embodiment of the present invention, the compound (C) of class (I-c) is selected from those of formula (I-c-1-1-1) herein-below: Formula (I-c-1-1-1) wherein R1, R4, R4’, z and A have the same meaning as defined above for formula (I), and wherein R7 is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with halogen atom, preferably R7 is hydrogen or methyl, more preferably R7 is hydrogen.
In a preferred embodiment of the present invention, the compound (C) of class (I-d) is selected from those of formula (I-d-1-1-1) herein-below: Formula (I-d-1-1-1) wherein R1, R5, n5, R4, R4’, z and A have the same meaning as defined above for formula (I). In a preferred embodiment of the present invention, the compound (C) of class (I-f) is selected from those of formula (I-f-1-1-1) herein-below: Formula (I-f-1-1-1) wherein R1, R6 and n6 have the same meaning as defined above for formula (I). In a preferred embodiment of the present invention, the compound (C) of formula (I-a-1-1-1) of class (I-a) according to the present invention is a compound chosen among those of formula (I-a-1-1-1-1) to (I-a-1-1-1-6) herein below: Formula (I-a-1-1-1-1) Formula (I-a-1-1-1-2) Formula (I-a-1-1-1-3)
Formula (I-a-1-1-1-4) Formula (I-a-1-1-1-5) Formula (I-a-1-1-1-6) wherein R4, R4’ and z have the same meaning as defined above for formula (I) and wherein: - each of R8 independently from each other and at each occurrence is selected from the group consisting of hydrogen, halo and CF3. - n8 is an integer equal to 0, 1 or 2; - R5 and R5’, independently from each other and at each occurrence is hydrogen or methyl. In a preferred embodiment of the present invention, the compound (C) of formula (I-b-1-1-1) of class (I-b) according to the present invention is selected from those of formula (I-b-1-1-1-1) to (I-b-1-1-1-13) herein below: Formula (I-b-1-1-1-1) Formula (I-b-1-1-1-2)
Formula (I-b-1-1-1-3) Formula (I-b-1-1-1-4) Formula (I-b-1-1-1-5) Formula (I-b-1-1-1-6) Formula (I-b-1-1-1-7) Formula (I-b-1-1-1-8) Formula (I-b-1-1-1-9) Formula (I-b-1-1-1-10) Formula (I-b-1-1-1-11)
Formula (I-b-1-1-1-12) Formula (I-b-1-1-1-13) wherein R4, R4’ and z have the same meaning as defined above for formula (I) and wherein: - R9 is selected from the group consisting of methyl, isopropyl, isobutyl, and ethyl, wherein said methyl, isopropyl, isobutyl and ethyl may be optionally substituted with N(CH3)2, - each of R10, independently from each other and at each occurrence is selected from the group consisting of hydrogen, halo, methyl and CF3; - n10 is an integer equal to 0, 1 or 2; - each of R11, independently from each other and at each occurrence is selected from the group consisting of hydrogen, halo, methyl, OCH3, CF3, and SO2NHCH3; - n11 is an integer equal to 0, 1 or 2; - each of R12, independently from each other and at each occurrence is selected from the group consisting of hydrogen, CF3, and N(R121)2, wherein each of R121, independently from each other and at each occurrence is hydrogen or methyl; - n12 is an integer equal to 0 or 1; - R7b is selected from methyl or isopropyl; - each of E, independently from each other and at each occurrence, is selected from CH or N; - each of B independently from each other and at each occurrence, is selected from the group consisting of N-R13, C(RB)2 and O, wherein each of RB, independently from each other and at each occurrence is selected from H or halo; - each of R13, independently from each other and at each occurrence is selected from hydrogen or methyl; - the dash bond represents an optional double bond.
In a preferred embodiment of the present invention, the compound (C) of Formula (I-b-1-1-2) of class (I-b) according to the present invention is selected from those of formula (I-b-1-1-2-1) herein below: Formula (I-b-1-1-2-1) wherein R4, R4’ and z have the same meaning as defined above for formula (I) and wherein halo is selected from the group consisting of F, Cl, Br and I. In a preferred embodiment of the present invention, the compound (C) of Formula (I-b-1-1-3) of class (I-b-1) according to the present invention is selected from those of formula (I-b-1-1-3-1) herein below: Formula (I-b-1-1-3-1) wherein R4, R4’ and z have the same meaning as defined above for formula (I) and wherein halo is selected from the group consisting of F, Cl, Br and I. In a preferred embodiment of the present invention, the compound (C) of Formula (I-b-2-1-1) of class (I-b-2) according to the present invention is selected from those of formula (I-b-2-1-1-1) herein below: H O R4 N O Halo N N N N N R4' H3C z Formula (I-b-2-1-1-1) wherein R4, R4’ and z have the same meaning as defined above for formula (I) and wherein halo is selected from the group consisting of F, Cl, Br and I. In a preferred embodiment of the present invention, the compound (C) of Formula (I-b-2-1-2) of class (I-b-2) according to the present invention is selected from those of formula (I-b-2-1-2-1) herein below:
Formula (I-b-2-1-2-1) wherein R4, R4’ and z have the same meaning as defined above for formula (I) and wherein halo is selected from the group consisting of F, Cl, Br and I. In a preferred embodiment of the present invention, the compound (C) of Formula (I-b-2-1-3) of class (I-b-2) according to the present invention is selected from those of formula (I-b-2-1-3-1) herein below: Formula (I-b-2-1-3-1) wherein R4, R4’ and z have the same meaning as defined above for formula (I) and wherein halo is selected from the group consisting of F, Cl, Br and I. In a preferred embodiment of the present invention, the compound (C) of Formula (I-b-3-1-1) of class (I-b-3) according to the present invention is selected from those of formula (I-b-3-1-1-1) herein below: Formula (I-b-3-1-1-1) wherein R4, R4’ and z have the same meaning as defined above for formula (I) and wherein halo is selected from the group consisting of F, Cl, Br and I. In a preferred embodiment of the present invention, the compound (C) of Formula (I-b-3-1-2) of class (I-b) according to the present invention is selected from those of formula (I-b-3-1-2-1) herein below: Formula (I-b-3-1-2-1) wherein R4, R4’ and z have the same meaning as defined above for formula (I) and wherein halo is selected from the group consisting of F, Cl, Br and I. In a preferred embodiment of the present invention, the compound (C) of Formula (I-b-3-1-3) of class (I-b) according to the present invention is
selected from those of formula (I-b-3-1-3-1) herein below: Formula (I-b-3-1-3-1) wherein R4, R4’ and z have the same meaning as defined above for formula (I) and wherein halo is selected from the group consisting of F, Cl, Br and I. In a preferred embodiment of the present invention, the compound (C) of Formula (I-c-1-1-1) of class (I-c) according to the present invention is selected from those of formula (I-c-1-1-1-1) or (I-c-1-1-1-2) herein below: Formula (I-c-1-1-1-1) Formula (I-c-1-1-1-2) wherein R4, R4’ and z have the same meaning as defined above for formula (I) and wherein: - each of R7’, independently from each other and at each occurrence is H or CH3; - each of U is independently C-R12c or N; - each of R12c, independently from each other and at each occurrence, is hydrogen, halo or OCH3; - n12c is an integer equal to 0, 1 or 2. In a preferred embodiment of the present invention, the compound (C) of Formula (I-d-1-1-1) of class (I-d) according to the present invention is selected from those of formula (I-d-1-1-1-1) or (I-d-1-1-1-2) herein below: Formula (I-d-1-1-1-1)
Formula (I-d-1-1-1-2) wherein R4, R4’ and z have the same meaning as defined above for formula (I) and wherein: - each of U is independently C-R12d or N; - each of R12d, independently from each other and at each occurrence, is hydrogen, halo or CF3; - n12d is an integer equal to 0, 1 or 2. In a preferred embodiment of the present invention, the compound (C) of formula (I-f-1-1-1) of class (I-f) is selected from those of formula (I-f-1-1-1-1) herein-below: Formula (I-f-1-1-1-1) wherein halo is selected from the group consisting of F, Cl, Br and I. In one embodiment of the present invention, the compound (C) of formula (I-a-1-1-1-3) of class (I-a) is preferably selected from those of formula (I-a-1-1- 1-3-0) herein-below: Formula (I-a-1-1-1-3-0) wherein: - each of R80 independently from each other and at each occurrence is selected from hydrogen or halo; - n80 is an integer equal to 0 or 1. In one embodiment of the present invention, the compound (C) of formula (I-a-1-1-1-3) of class (I-a) is preferably selected from those of formula (I-a-1-1- 1-3-1) herein-below:
Formula (I-a-1-1-1-3-1) wherein: - each of R5 and R5’, independently from each other and at each occurrence is hydrogen or methyl; - each of R81 independently from each other and at each occurrence is selected from the group consisting of hydrogen, halo and CF3; - n81 is an integer equal to 0, 1 or 2. In one embodiment of the present invention, the compound (C) of formula (I-a-1-1-1-3) of class (I-a) is preferably selected from those of formula (I-a-1-1- 1-3-2) herein-below: Formula (I-a-1-1-1-3-2) wherein each of R82 independently from each other and at each occurrence is selected from hydrogen or halo. In one embodiment of the present invention, the compound (C) of formula (I-a-1-1-1-6) of class (I-a) is preferably selected from those of formula (I-a-1-1- 1-6-2) herein-below: Formula (I-a-1-1-1-6-2) In one embodiment of the present invention, the compound (C) of formula (I-b-1-1-1-4) of class (I-b) is preferably selected from those of formula (I-a-1-1- 1-4-0) herein-below: Formula (I-b-1-1-1-4-0) wherein each of R110, independently from each other and at each occurrence is selected from hydrogen or halo.
In one embodiment of the present invention, the compound (C) of formula (I-b-1-1-1-2) of class (I-b) is preferably selected from those of the following formulae herein-below: Formula (I-b-1-1-1-2-1) Formula (I-b-1-1-1-4-1) Formula (I-b-1-1-1-5-1) Formula (I-b-1-1-1-7-1) Formula (I-b-1-1-1-8-1) Formula (I-b-1-1-1-12-1) Formula (I-b-1-1-1-13-1) wherein: - each of R101, independently from each other and at each occurrence is selected from the group consisting of hydrogen, halo, and CF3; - n101 is an integer equal to 0 or 1; - R4’’ is hydrogen or methyl;
- each of R111, independently from each other and at each occurrence is selected from the group consisting of hydrogen, halo, OCH3, CF3, and SO2NHCH3; - n111 is an integer equal to 0, 1 or 2; - each of R121, independently from each other and at each occurrence is selected from the group consisting of hydrogen, CF3, and N(R1211)2, wherein each of R1211, independently from each other and at each occurrence is hydrogen or methyl; - n121 is an integer equal to 0 or 1; - R7b1 is selected from methyl, or isopropyl; - each of B independently from each other and at each occurrence, is selected from N-CH3 or O; - the dash bond represents an optional double bond. In one embodiment of the present invention, the compound (C) of formula (I-b-1-1-1-1) of class (I-b) is preferably selected from those of the following formulae herein-below: Formula (I-b-1-1-1-1-2) Formula (I-b-1-1-1-4-2) Formula (I-b-1-1-1-4-2a) Formula (I-b-1-1-1-5-2)
Formula (I-b-1-1-1-5-2a) Formula (I-b-1-1-1-6-2) Formula (I-b-1-1-1-7-2) wherein: - R92 is selected from hydrogen, N(CH3)2 or ethyl; - R42 is selected from hydrogen or ethyl; - each of R112, independently from each other and at each occurrence is selected from the group consisting of hydrogen, halo, and CF3; - each of R102, independently from each other and at each occurrence is selected from hydrogen or methyl; - each of n102, independently from each other and at each occurrence is an integer equal to 0, 1 or 2; - each of B independently from each other and at each occurrence, is selected from C-(RB)2 or O, wherein each of RB, independently from each other and at each occurrence is selected from H or halo. In one embodiment of the present invention, the compound (C) of formula (I-b-2-1-1-1) of class (I-b) is preferably selected from those of the following formulae herein-below: Formula (I-b-2-1-1-1-1) wherein halo is selected from the group consisting of F, Cl, Br and I. In one embodiment of the present invention, the compound (C) of formula (I-b-2-1-2-1) of class (I-b) is preferably selected from those of the following formulae herein-below:
Formula (I-b-2-1-2-1-1) wherein halo is selected from the group consisting of F, Cl, Br and I. In one embodiment of the present invention, the compound (C) of formula (I-b-2-1-3-1) of class (I-b) is preferably selected from those of the following formulae herein-below: Formula (I-b-2-1-3-1-1) wherein halo is selected from the group consisting of F, Cl, Br and I. In one embodiment of the present invention, the compound (C) of formula (I-b-3-1-1-1) of class (I-b) is preferably selected from those of the following formulae herein-below: Formula (I-b-3-1-1-1-1) wherein halo is selected from the group consisting of F, Cl, Br and I. In one embodiment of the present invention, the compound (C) of formula (I-b-3-1-2-1) of class (I-b) is preferably selected from those of the following formulae herein-below: Formula (I-b-3-1-2-1-1) wherein halo is selected from the group consisting of F, Cl, Br and I. In one embodiment of the present invention, the compound (C) of formula (I-b-3-1-3-1) of class (I-b) is preferably selected from those of the following formulae herein-below: Formula (I-b-3-1-3-1-1) wherein halo is selected from the group consisting of F, Cl, Br and I.
In one embodiment of the present invention, the compound (C) of formula (I-c-1-1-1-2) of class (I-c) is preferably selected from those of formulae (I-c-1-1- 1-2-1) herein-below: Formula (I-c-1-1-1-2-1) wherein: - each of R7’, independently from each other and at each occurrence is H or CH3; - each of U is independently selected from C-R12’ or N; - each of R12c1, independently from each other and at each occurrence, is hydrogen, halo or OCH3; - n12c1 is an integer equal to 0, 1 or 2. In one embodiment of the present invention, the compound (C) of formula (I-d-1-1-1-1) of class (I-d) is preferably selected from those of formulae (I-d-1-1- 1-1-0) or (I-d-1-1-1-1-0) herein-below: Formula (I-d-1-1-1-1-0) wherein: - each of U is independently C-R12d0 or N; - each of R12d0, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, halo, or CF3; - n12d0 is an integer equal to 0, 1 or 2; In one embodiment of the present invention, the compound (C) of formula (I-d-1-1-1-1) of class (I-d) is preferably selected from those of formulae (I-d-1-1- 1-1-1) herein-below: Formula (I-d-1-1-1-1-1) wherein: - each of U is independently C-R12d1 or N;
- each of R12d1, independently from each other and at each occurrence, is hydrogen, halo or CF3; - n12d1 is an integer equal to 0, 1 or 2. In a preferred embodiment of the present invention, the compound (C) according to general formula (I-a-1) is a compound chosen among those of formulae (II) to (XXIX) herein below: Formula (I) Formula (II) Formula (III) Formula (IV) Formula (V) Formula (VI) Formula (VII)
Formula (VIII) Formula (IX) Formula (X) Formula (XI) Formula (XII) Formula (XIII) Formula (XIV) Formula (XV)
Formula (XVI) Formula (XVII) Formula (XVIII) Formula (XIX) Formula (XX) Formula (XXI) Formula (XXII) Formula (XXIII)
Formula (XXIV) Formula (XXV) Formula (XXVI) Formula (XXVII) Formula (XXVIII) Formula (XXIX) In a preferred embodiment of the present invention, the compound (C) according to general formula (I-b-1) is a compound chosen among those of formulae (XXX) to (CIII) herein below: Formula (XXX)
Formula (XXXI) Formula (XXXII) Formula (XXXIII) Formula (XXXIV) Formula (XXXV) Formula (XXXVI) Formula (XXXVII) Formula (XXXVIII)
Formula (XXXIX) Formula (XL) Formula (XLI) Formula (XLII) Formula (XLIII) Formula (XLIV) Formula (XLV) Formula (XLVI)
Formula (XLVII) Formula (XLVIII) Formula (XLIX) Formula (L) Formula (LI) Formula (LII) Formula (LIII) Formula (LIV)
Formula (LV) Formula (LVI) Formula (LVII) Formula (LVIII) Formula (LIX) Formula (LX) Formula (LXI) Formula (LXII)
Formula (LXIII) Formula (LXIV) Formula (LXV) Formula (LXVI) Formula (LXVII) Formula (LXVIII) Formula (LXIX) Formula (LXX)
Formula (LXXI) Formula (LXXII) Formula (LXXIII) Formula (LXXIV) Formula (LXXV) Formula (LXXVI) Formula (LXXVII) Formula (LXXVIII)
Formula (LXXIX) Formula (LXXX) Formula (LXXXI) Formula (LXXXII) Formula (LXXXIII) Formula (LXXXIV) Formula (LXXXV) Formula (LXXXVI)
Formula (LXXXVII) Formula (LXXXVIII) Formula (LXXXIX) Formula (XC) Formula (XCI) Formula (XCII) Formula (XCIII) Formula (XCIV)
Formula (XCV) Formula (XCVI) Formula (XCVII) Formula (XCVIII) Formula (XCIX) Formula (C) Formula (CI) Formula (CII)
Formula (CIII) In a preferred embodiment of the present invention, the compound (C) according to general formula (I-b-2) is a compound chosen among those of formulae (CIV) to (CVI) herein below: Formula (CIV) Formula (CV) Formula (CVI) In a preferred embodiment of the present invention, the compound (C) according to general formula (I-b-3) is a compound chosen among those of formulae (CVII) to (CIX) herein below: Formula (CVII) Formula (CVIII) Formula (CIX)
In a preferred embodiment of the present invention, the compound (C) according to general formula (I-c-1) is a compound chosen among those of formulae (CX) to (CXXV) herein below: Formula (CX) Formula (CXI) Formula (CXII) Formula (CXIII) Formula (CXIV) Formula (CXV) Formula (CXVI) Formula (CXVII)
Formula (CXVIII) Formula (CXIX) Formula (CXX) Formula (CXXI) Formula (CXXII) Formula (CXXIII) Formula (CXXIV) Formula (CXXV)
In a preferred embodiment of the present invention, the compound (C) according to general formula (I-d-1) is a compound chosen among those of formulae (CXXVI) to (CXLII) herein below: Formula (CXXVI) Formula (CXXVII) Formula (CXXVIII) Formula (CXXIX) Formula (CXXX) Formula (CXXXI) Formula (CXXXII)
Formula (CXXXIII) Formula (CXXXIV) Formula (CXXXV) Formula (CXXXVI) Formula (CXXXVII) Formula (CXXXVIII) Formula (CXXXIX)
Formula (CXL) Formula (CXLI) Formula (CXLII) In a preferred embodiment of the present invention, the compound (C) according to general formula (I-f-1) is a compound chosen among those of formulae (CXLIII) to (CXLVI) herein below: Formula (CXLIII) Formula (CXLIV) Formula (CXLV)
Formula (CXLVI) The present invention further relates to an in vitro method of inhibiting protein kinase activity which comprises contacting a protein kinase with a compound of formulae (I) to (VII) [compound (C), herein after], as defined above, or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, R1 N N X W R2 N Formula (I) wherein: - each of R1, independently from each other and at each occurrence, is selected from hydrogen, or C1-6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of W, independently from each other and at each occurrence is a bond, or is selected from the group consisting of O, N(R11), and N(R11)(C=O); wherein each of R11, independently from each other and at each occurrence is selected from the group consisting of hydrogen and C1-6 alkyl; - each of X, independently from each other and at each occurrence is selected from O, S, or NR7, wherein R7 is selected from hydrogen or C1- 6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of R2, independently from each other and at each occurrence is a moiety selected from the group consisting of (R2-a) to (R2-f): O R4' N A Y R5' R4 R5 z (R2-a)
(R2-b) (R2-c) (R2-d) (R2-e) (R2-f) wherein: - each of Y independently from each other and at each occurrence is selected from the group consisting of O, S, and NR7, wherein R7 is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of R5, and R5’, independently from each other and at each occurrence is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of n5, independently from each other and at each occurrence, is an integer equal to 0, 1 or 2; - R6 is selected from the group consisting of halo, NO2, C1-6 alkyl, C2-4 alkenyl, C2-4 alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, CF3, CN, OR11, SR11, N(R11)2,, NR11COR12, COR11, C(O)OR11, CON(R11)2, OC(O)R11, and OCON(R11)2, wherein each of R11 and R12, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and CF3, wherein said alkyl, alkenyl, and alkynyl substituents are optionally substituted with at least one halogen atom.
- each of n6, independently from each other and at each occurrence, is an integer equal to 0, 1 or 2; - each of Z independently from each other and at each occurrence is selected from the group consisting of O, S and NR7, wherein R7 is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of R4 and R4’, independently from each other and at each occurrence, are selected from hydrogen, or C1-6 alkyl, wherein said alkyl is optionally substituted with a halogen atom, or R4-C-R4’ can form C=O; - each of z, independently from each other and at each occurrence, is an integer equal to 0, 1, 2, 3 or 4; - each of A, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl, wherein said alkyl, alkenyl, alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl are optionally substituted with one or more substituents independently selected from the group consisting of halo, NO2, C1-6 alkyl, C2-4 alkenyl, C2-4 alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, CF3, CN, OR11, SR11, N(R11)2, OC(R11)2O, OC(R11)2C(R11)2O, S(O)R12, SO2R12, SO2N(R11)2, S(O)3R11, P(=O)(OR11)2, P(=O)(R11)2, NR11COR12, COR11, C(O)OR11, CON(R11)2, OC(O)R11, and OCON(R11)2, and each optional alkyl, alkenyl, alkynyl, cycloalkyl, aryl, heterocyclyl, and heteroaryl substituents is further optionally substituted with halo, NO2, CF3, N(R11)2, COR11, CON(R11)2, OC(O)R11, CN, or OR11; and wherein each of R11 and R12, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and CF3, wherein said alkyl, alkenyl, and alkynyl, substituents are optionally substituted at least one halogen atom; wherein said cycloalkyl is a monocyclic, bicyclic or tricyclic ring system of 3- 6 ring members per ring; said heterocyclyl is a saturated, partially saturated or completely unsaturated monocycle, bicycle or tricycle containing 3 to 12 carbon atoms and 1 or 2 heteroatoms independently selected from O or N; said aryl is phenyl, naphthyl or anthracenyl optionally carbocyclic fused with a cycloalkyl or heterocyclyl of 5-7 ring members; said heteroaryl is a
monocyclic ring structure containing 5 or 6 ring atoms, or a bicyclic aromatic group having 8 to 10 atoms, containing 1-3 heteroatoms independently selected from O or N. It is further understood that all definitions and preferences as described for compound (C) above equally apply for this embodiment and all further embodiments, as described below. As used in the foregoing and hereinafter, the following definitions apply unless otherwise noted. The term halo - alone or in combination means all halogens, that is, chloro (Cl), bromo (Br), fluoro (F), iodo (I). The term alkyl - alone or in combination means an alkane-derived radical containing from 1 to 15 carbon atoms, unless otherwise specified, for example CF-G alkyl defines a straight or branched alkyl radical having from F to G carbon atoms, e.g. C1-4 alkyl defines a straight or branched alkyl radical having from 1 to 4 carbon atoms such as for example methyl, ethyl, 1-propyl, 2-propyl, I-butyl, 2-butyl, 2-methyl-1-propyl. An alkyl group may be a straight chain alkyl or branched alkyl. Preferably, straight or branched alkyl groups containing from 1- 10, more preferably 1 to 8, even more preferably 1-6 and most preferably 1-4, carbon atoms, such as methyl, ethyl, propyl, isopropyl, butyl, t-butyl and the like. Alkyl also includes a straight chain or branched alkyl group that contains or is interrupted by a cycloalkyl portion. The straight chain or branched alkyl group is attached at any available point to produce a stable compound. Examples of this include, but are not limited to, 4-(isopropyl)-cyclohexylethyl or 2-methyl- cyclopropylpentyl. The term alkenyl - alone or in combination means a straight or branched hydrocarbon containing 2-15, more preferably 2-10, even more preferably 2-8, most preferably 2-4, carbon atoms, unless otherwise specified and at least one, preferably 1-3, more preferably 1-2, most preferably one, carbon to carbon double bond. Examples of alkenyl groups include ethenyl, propenyl, isopropenyl, butenyl, cyclohexenyl, cyclohexenylalkyl and the like. Alkenyl also includes a straight chain or branched alkenyl group that contains or is interrupted by a cycloalkyl portion. Carbon to carbon double bonds may be either contained within a cycloalkyl portion, with the exception of cyclopropyl, or within a straight chain or branched portion.
The term alkynyl - alone or in combination means a straight or branched hydrocarbon containing 2-15 more preferably 2-10, even more preferably 2-8, most preferably 2-4, carbon atoms containing at least one, preferably one, carbon to carbon triple bond. Examples of alkynyl groups include ethynyl, propynyl, butynyl and the like. The term aryl - alone or in combination means phenyl, naphthyl or anthracenyl optionally carbocyclic fused with a cycloalkyl or heterocyclyl of preferably 5-7, more preferably 5-6, ring members and/or optionally substituted with 1 to 5 groups or substituent. An aryl may be optionally substituted whereby the substituent is attached at one point to the aryl or whereby the substituent is attached at two points to the aryl to form a bicyclic system e.g. benzodioxole, benzodioxan, benzimidazole. The term heteroaryl - alone or in combination means a monocyclic aromatic ring structure containing 5 or 6 ring atoms, or a bicyclic aromatic group having 8 to 10 atoms, containing 1-3, heteroatoms independently selected from the group O, S, and N, and optionally substituted with 1 to 5 groups or substituents. Heteroaryl is also intended to include oxidized S or N, such as sulfinyl, sulfonyl and N-oxide of a tertiary ring nitrogen. A carbon or nitrogen atom is the point of attachment of the heteroaryl ring structure such that a stable aromatic ring is retained. More specifically the term heteroaryl includes, but is not limited to, pyridyl, furanyl, thiophenyl, thiazolyl, isothiazolyl, triazolyl, imidazolyl, isoxazolyl, pyrrolyl, pyrazolyl, pyrimidinyl, benzofuranyl, isobenzofuranyl, benzothiazolyl, benzoisothiazolyl, benzotriazolyl, indolyl, isoindolyl, benzoxazolyl, quinolyl, isoquinolyl, benzimidazolyl, benzisoxazolyl, benzothiophenyl, dibenzofuran, and benzodiazepin-2-one-5-yl, and the like. The term heterocyclyl - alone or in combination is intended to denote a saturated, partially unsaturated or completely unsaturated monocycle, bicycle, or tricycle having 3 to 12 carbon atoms and containing 1 or 2 heteroatoms each independently selected from O, S, P or N, and are optionally benzo fused or fused heteroaryl of 5-6 ring members and/or are optionally substituted as in the case of cycloalkyl. Heterocycyl is also intended to include oxidized S or N, such as sulfinyl, sulfonyl and N-oxide of a tertiary ring nitrogen. The point of attachment is at a carbon or nitrogen atom. In each case the heterocyclyl may be condensed with an aryl to form a bicyclic ring system.
The term cycloalkyl refers to a cyclic or polycyclic alkyl group containing 3 to 7 carbon atoms. Preferably, cycloalkyl groups are monocyclic, bicyclic or tricyclic ring systems of 3-6, ring members per ring, such as cyclopropyl, cyclopentyl, cyclohexyl, adamantyl and the like. The term aralkyl refers to organic compounds containing an aromatic nucleus to which an alkyl radical is bonded. These alkyl radicals include methyl, ethyl, propyl, butyl, octyl, etc. radicals. The term aralkyl is thus seen to include aralkyl hydrocarbons such as the alkyl benzenes, and the various alkyl naphthalenes. From this definition of the term aralkyl compound it is seen that the term includes compounds such as benzyl, the three isomeric xylyls, the two isomeric trimethyl benzenes, ethyl benzene, p-methyl biphenyl, a-methyl naphthalene, etc. The present invention further relates to a pharmaceutical composition comprising a carrier, and as active ingredient an effective amount of a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (I-a-1), (I-b-1)- (I-b-3), (I-c-1), (I-d-1), (I-e-1), (I-f-1), (I-a- 1-1), (I-b-1-1), (I-b-2-1), (I-b-3-1), (I-c-1-1), (I-d-1-1), (I-e-1-1), (I-f-1-1), (I-a-1-1- 1), (I-b-1-1-1)- (I-b-1-1-3), (I-b-2-1-1)- (I-b-2-1-3), (I-b-3-1-1)- (I-b-3-1-3), (I-c-1- 1-1), (I-d-1-1-1), (I-f-1-1-1), (I-a-1-1-1-1)- (I-a-1-1-1-6), (I-b-1-1-1-1)- (I-b-1-1-1- 13), (I-b-1-1-2-1), (I-b-1-1-3-1), (I-b-2-1-1-1), (I-b-2-1-2-1-1), (I-b-2-1-2-1), (I-b-2- 1-3-1), (I-b-3-1-1-1), (I-b-3-1-2-1), (I-b-3-1-3-1), (I-c-1-1-1-1), (I-c-1-1-1-2), (I-d- 1-1-1-1), (I-d-1-1-1-2), (I-f-1-1-1-1), (I-a-1-1-1-3-0), (I-a-1-1-1-3-1), (I-a-1-1-1-3- 2), (I-a-1-1-1-6-2), (I-b-1-1-1-4-0), (I-b-1-1-1-2-1), (I-b-1-1-1-4-1), (I-b-1-1-1-5-1), (I-b-1-1-1-7-1), (I-b-1-1-1-8-1), (I-b-1-1-1-12-1), (I-b-1-1-1-13-1), (I-b-1-1-1-1-2), (I-b-1-1-1-4-2), (I-b-1-1-1-4-2a), (I-b-1-1-1-5-2), (I-b-1-1-1-6-2), (I-b-1-1-1-7-2), (I-b-2-1-1-1-1), (I-b-2-1-3-1-1), (I-b-3-1-1-1-1), (I-b-3-1-2-1-1), (I-b-3-1-3-1-1), (I- c-1-1-1-2-1), (I-d-1-1-1-1-0), and (I-d-1-1-1-1-1), as specified herein, and as defined in any one of the embodiments presented herein. The present invention relates to a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (I-a-1), (I-b-1)- (I-b-3), (I-c-1), (I-d-1), (I-e-1), (I-f-1), (I-a-1-1), (I-b-1-1), (I-b-2-1), (I-b-3-1), (I-c-1-1), (I-d-1-1), (I-e-1-1), (I-f-1-1), (I-a-1-1-1), (I-b-1-1-1)- (I-b-1-1-3), (I-b-2-1-1)- (I-b-2-1-3), (I-b-3-1-1)- (I-b-3-1-3), (I-c-1-1-1), (I-d-1-1-1), (I-f-1-1-1), (I-a-1-1-1-1)- (I-a-1-1-1-6), (I-b-1-1-1-1)- (I-b-1-1-1-13), (I-b-1-1-2-1),
(l-b-1 -1-3-1), (l-b-2-1 -1-1), (l-b-2-1 -2-1-1 ), (l-b-2-1 -2-1 ), (l-b-2-1 -3-1 ), (l-b-3-1 -1- 1), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1-1 -1-1), (l-c-1-1-1-2), (l-d-1 -1 -1 -1 ), (l-d-1-1- 1-2), (l-f-1 -1-1 -1), (l-a-1-1 -1 -3-0), (l-a-1-1 -1-3-1), (l-a-1 -1 -1 -3-2), (l-a-1 -1 -1 -6-2), (l-b-1 -1 -1-4-0), (l-b-1-1 -1 -2-1 ), (l-b-1 -1 -1-4-1 ), (l-b-1 -1 -1 -5-1 ), (l-b-1 -1 -1 -7-1 ), (I- b-1-1-1-8-1), (l-b-1 -1 -1 -12-1 ), (l-b-1 -1 -1 -13-1 ), (l-b-1 -1 -1 -1 -2), (l-b-1 -1-1 -4-2), (I- b-1-1-1-4-2a), (l-b-1 -1-1 -5-2), (l-b-1 -1 -1 -6-2), (l-b-1 -1-1 -7-2), (l-b-2-1 -1 -1 -1 ), (I- b-2-1-3-1-1), (l-b-3-1 -1-1 -1 ), (l-b-3-1 -2-1 -1 ), (l-b-3-1 -3-1-1), (l-c-1 -1 -1 -2-1 ), (l-d- 1 -1 -1 -1 -0), and (l-d-1 -1-1-1 -1 ), as specified herein, and as defined in any one of the embodiments presented herein, for use as a medicament.
The present invention relates to a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1), (l-b-1)- (l-b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1 -1), (l-e-1-1), (l-f-1 -1), (l-a-1-1 -1), (l-b-1-1-1)- (l-b-1-1-3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3-1 -1 )- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1 -1 -1 ), (l-f-1-1 -1 ), (l-a-1-1 -1 -1)- (l-a-1 -1 -1-6), (l-b-1-1 -1 -1)- (l-b-1 -1 -1 -13), (l-b-1 -1 -2-1 ), (l-b-1 -1 -3-1 ), (l-b-2-1 -1-1), (l-b-2-1 -2-1-1 ), (l-b-2-1 -2-1 ), (l-b-2-1 -3-1), (l-b-3-1 -1- 1), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1-1 -1-1), (l-c-1-1-1-2), (l-d-1 -1 -1 -1 ), (l-d-1-1- 1-2), (l-f-1 -1-1 -1), (l-a-1 -1-1 -3-0), (l-a-1-1 -1-3-1), (l-a-1 -1-1 -3-2), (l-a-1 -1-1 -6-2), (l-b-1 -1-1 -4-0), (l-b-1-1 -1 -2-1 ), (l-b-1 -1 -1-4-1 ), (l-b-1 -1 -1 -5-1 ), (l-b-1 -1 -1 -7-1 ), (I- b-1-1-1-8-1), (l-b-1 -1 -1 -12-1 ), (l-b-1 -1 -1 -13-1 ), (l-b-1 -1 -1 -1 -2), (l-b-1 -1-1 -4-2), (I- b-1-1-1-4-2a), (l-b-1 -1-1 -5-2), (l-b-1 -1 -1 -6-2), (l-b-1 -1-1 -7-2), (l-b-2-1 -1 -1 -1 ), (I- b-2-1-3-1-1), (l-b-3-1 -1-1 -1 ), (l-b-3-1 -2-1 -1 ), (l-b-3-1 -3-1-1), (l-c-1 -1 -1 -2-1 ), (l-d- 1 -1 -1 -1 -0), and (l-d-1 -1-1-1 -1 ), as specified herein, and as defined in any one of the embodiments presented herein, for use in the treatment of a disease selected from from cancer, metabolic disorders (such as diabetes), inflammatory and autoimmune disorders (such as inflammatory bowel diseases, e.g. Crohn’s disease and ulcerative colitis, inflammatory pulmonary diseases, rheumatoid arthritis, lupus nephritis, systemic lupus erythematosus and psoriasis and psoriasis arthritis), neurological disorders (such as Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, Charcot-Marie-Tooth neuropathy, amyotrophic lateral sclerosis and epilepsy), atherosclerosis and cardiovascular diseases, Sjogren Syndrome, renal allograft rejection, viral induced diseases, circulatory diseases, bone osteolysis and osteoporosis, osteoarthritis, sarcopenia, Langerhans cell histiocytosis, spinal cord injury, endometriosis,
asthma and allergic asthma, eye diseases (such as retinopathies, age-related macular degeneration and uveitis) chronic and neuropathic pain, and fibro- proliferative diseases.
The present invention further relates to a method of inhibiting protein kinase activity in a warm-blooded animal said method comprising the administration to an animal in need thereof, of a kinase-inhibitory effective amount of a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- ( l-b-3) , (l-c-1 ), (I- d-1), (l-e-1), (l-f-1 ), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1-1 ), (I- e-1-1), (l-f-1 -1 ), (l-a-1 -1 -1 ), (l-b-1 -1 -1 )- (l-b-1 -1 -3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3- 1-1)- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1-1-1), (l-f-1 -1 -1 ), (l-a-1 -1 -1 -1 )- (l-a-1 -1 -1 -6), (I- b-1-1-1-1 )- (l-b-1-1 -1-13), (l-b-1-1-2-1), (l-b-1 -1-3-1), (l-b-2-1 -1 -1 ), (l-b-2-1 -2-1 - 1), (l-b-2-1 -2-1), (l-b-2-1 -3-1), (l-b-3-1 -1-1), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1 -1 - 1-1), (l-c-1 -1 -1 -2), (l-d-1-1-1-1), (l-d-1-1-1-2), (l-f-1 -1 -1 -1 ), (l-a-1 -1 -1 -3-0), (l-a-1 - 1-1 -3-1 ), (l-a-1 -1-1 -3-2), (l-a-1 -1-1 -6-2), (l-b-1 -1-1 -4-0), (l-b-1 -1 -1 -2-1 ), (l-b-1-1 - 1-4-1), (l-b-1-1-1-5-1), (l-b-1 -1 -1-7-1), (l-b-1-1-1 -8-1), (l-b-1 -1 -1 -12-1 ), (l-b-1-1- 1-13-1), (l-b-1 -1 -1 -1 -2), (l-b-1 -1-1 -4-2), (l-b-1 -1-1-4-2a), (l-b-1 -1 -1 -5-2), (l-b-1 -1 - 1-6-2), (l-b-1 -1-1 -7-2), (l-b-2-1 -1-1 -1 ), (l-b-2-1 -3-1 -1 ), (l-b-3-1 -1 -1 -1 ), (l-b-3-1 -2- 1-1), (l-b-3-1 -3-1-1), (l-c-1 -1-1 -2-1), (l-d-1-1 -1-1-0), and (l-d-1-1-1-1-1), as specified herein, and according to any one of the embodiments presented herein.
The present invention further relates to a method of inhibiting protein kinase activity in a warm-blooded animal said method comprising the administration to an animal in need thereof, of a kinase-inhibitory effective amount of a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- (l-b-3), (l-c-1 ), (I- d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1-1), (I- e-1-1), (l-f-1 -1 ), (l-a-1 -1 -1 ), (l-b-1-1-1)- (l-b-1 -1 -3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3- 1-1)- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1-1-1), (l-f-1 -1 -1 ), (l-a-1 -1 -1 -1 )- (l-a-1 -1 -1 -6), (I- b-1-1-1-1 )- (l-b-1-1 -1-13), (l-b-1-1-2-1), (l-b-1 -1-3-1), (l-b-2-1 -1 -1 ), (l-b-2-1 -2-1 - 1), (l-b-2-1 -2-1), (l-b-2-1 -3-1), (l-b-3-1 -1-1), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1 -1 - 1-1), (l-c-1 -1 -1 -2), (l-d-1-1-1-1), (l-d-1-1-1-2), (l-f-1 -1 -1 -1 ), (l-a-1 -1 -1 -3-0), (l-a-1 - 1-1 -3-1 ), (l-a-1 -1-1 -3-2), (l-a-1 -1-1 -6-2), (l-b-1 -1-1 -4-0), (l-b-1 -1 -1 -2-1 ), (l-b-1-1 - 1-4-1), (l-b-1-1-1-5-1), (l-b-1 -1 -1-7-1), (l-b-1-1-1 -8-1), (l-b-1 -1 -1 -12-1 ), (l-b-1-1-
1 -13-1 ), (l-b-1 -1 -1 -1 -2), (l-b-1 -1 -1 -4-2), (l-b-1 -1 -1 -4-2a), (l-b-1 -1 -1 -5-2), (l-b-1 -1 - 1 -6-2), (l-b-1 -1 -1 -7-2), (l-b-2-1 -1 -1 -1 ), (l-b-2-1 -3-1 -1 ), (l-b-3-1 -1 -1 -1 ), (l-b-3-1 -2- 1 -1 ), (l-b-3-1 -3-1 -1 ), (l-c-1-1-1 -2-1), (l-d-1 -1-1 -1 -0), and (l-d-1 -1 -1 -1 -1 ), as specified herein, and according to any one of the embodiments presented herein, wherein the protein kinase is selected from the group consisting of CSF1 R, FLT3, Kit, PDGFRB (PDGFR beta), PDGFRA (PDGFR alfa), ABL1 , ACVR1 B (ALK4), AKT1 (PKB alpha), AMPK A1/B1/G1 , AURKA (Aurora A), BTK, CDK1/cyclin B, CHEK1 (CHK1 ), CSNK1 G2 (CK1 gamma 2), CSNK2A1 (CK2 alpha 1 ), DYRK3, EGFR (ErbB1 ), EPHA2, ERBB2 (HER2), FGFR1 , FRAP1 (mTOR), GSK3B (GSK3 beta), IGF1 R, IKBKB (IKK beta), INSR, IRAK4, JAK3, KDR (VEGFR2), LCK, MAP2K1 (MEK1 ), MAP4K4 (HGK), MAPK1 (ERK2), MAPK14 (p38 alpha), MAPK3 (ERK1 ), MAPK8 (JNK1 ), MARK2, MET (cMet), NEK1 , PAK4, PHKG2, PIM1 , PLK1 , PRKACA (PKA), PRKCB1 (PKC beta I), ROCK1 , RPS6KA3 (RSK2), RPS6KB1 (p70S6K), SRC, SYK, and TEK (Tie2). Preferably, the protein kinase is selected from the group consisting of CSF1 R, FLT3, Kit, PDGFRB (PDGFR beta), PDGFRA (PDGFR alpha).
The present invention further relates to a method of treating a disease selected from cancer, metabolic disorders (such as diabetes), inflammatory and autoimmune disorders (such as inflammatory bowel diseases, e.g. Crohn’s disease and ulcerative colitis, inflammatory pulmonary diseases, rheumatoid arthritis, lupus nephritis, systemic lupus erythematosus and psoriasis and psoriasis arthritis), neurological disorders (such as Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, Charcot-Marie-Tooth neuropathy, amyotrophic lateral sclerosis and epilepsy), atherosclerosis and cardiovascular diseases, Sjogren Syndrome, renal allograft rejection, viral induced diseases, circulatory diseases, bone osteolysis and osteoporosis, osteoarthritis, sarcopenia, Langerhans cell histiocytosis, spinal cord injury, endometriosis, asthma and allergic asthma, eye diseases (such as retinopathies, age-related macular degeneration and uveitis) chronic and neuropathic pain, and fibro- proliferative diseases, in a warm-blooded animal said method comprising the administration to an animal in need thereof of an effective amount of a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- (l-b-3), (l-c-1 ), (l-d-1 ), (l-e-1 ), (l-f-1 ), (l-a- 1 -1 ), (l-b-1-1), (l-b-2-1 ), (l-b-3-1 ), (l-c-1-1), (l-d-1-1), (l-e-1-1), (l-f-1-1), (l-a-1 -1 -
1), (l-b-1 -1-1)- (l-b-1-1-3), (l-b-2-1-1)- (l-b-2-1-3), (l-b-3-1-1)- (l-b-3-1-3), (l-c-1 - 1-1), (l-d-1-1 -1), (l-f-1-1-1 ), (l-a-1 -1-1-1)- (l-a-1-1-1 -6), (l-b-1 -1 -1 -1 )- (l-b-1 -1 -1 - 13), (l-b-1 -1 -2-1 ), (l-b-1 -1 -3-1 ), (l-b-2-1 -1 -1 ), (l-b-2-1 -2-1 -1 ), (l-b-2-1 -2-1 ), (l-b-2- 1-3-1), (l-b-3-1-1 -1), (l-b-3-1-2-1), (l-b-3-1 -3-1), (l-c-1 -1 -1 -1 ), (l-c-1 -1 -1 -2), (l-d- 1 -1-1-1), (l-d-1 -1-1-2), (l-f-1 -1-1-1), (l-a-1 -1-1 -3-0), (l-a-1 -1-1 -3-1 ), (l-a-1 -1 -1 -3-
2), (l-a-1 -1-1 -6-2), (l-b-1 -1 -1 -4-0), (l-b-1 -1 -1 -2-1 ), (l-b-1 -1 -1 -4-1 ), (l-b-1 -1 -1 -5-1 ), (l-b-1 -1-1-7-1 ), (l-b-1 -1-1 -8-1 ), (l-b-1-1-1-12-1), (l-b-1 -1 -1-13-1), (l-b-1 -1 -1 -1 -2), (l-b-1 -1-1 -4-2), (l-b-1 -1-1-4-2a), (l-b-1 -1-1 -5-2), (l-b-1 -1-1 -6-2), (l-b-1 -1-1 -7-2), (l-b-2-1 -1-1 -1 ), (l-b-2-1 -3-1-1 ), (l-b-3-1 -1-1 -1 ), (l-b-3-1 -2-1 -1 ), (l-b-3-1 -3-1-1), (I- c-1-1-1-2-1), (l-d-1 -1-1 -1-0), and (l-d-1 -1-1 -1-1), as specified herein, and according to any one of the embodiments presented herein.
It should be noted that the radical positions on any molecular moiety used in the definitions may be anywhere on such moiety as long as it is chemically stable.
Radicals used in the definitions of the variables include all possible isomers unless otherwise indicated. For instance, pyridyl includes 2-pyridyl, 3- pyridyl and 4-pyridyl; pentyl includes 1 -pentyl, 2-pentyl and 3-pentyl.
When any variable occurs more than one time in any constituent, each definition is independent. Whenever used hereinafter, the term "compounds (C) of formulae (I) to (VII) ", or "the present compounds" or similar terms, it is meant to include all the compounds (C) of formulae (I), N-oxides, addition salts, and stereochemically isomeric forms. One embodiment comprises the compounds (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1), (l-b-1)- (l-b-3), (l-c-1), (l-d-1), (l-e-1 ), (l-f-1), (l-a- 1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1-1), (l-e-1-1), (l-f-1-1), (l-a-1 -1 -
1), (l-b-1 -1-1)- (l-b-1-1-3), (l-b-2-1-1)- (l-b-2-1-3), (l-b-3-1-1)- (l-b-3-1-3), (l-c-1 - 1-1), (l-d-1-1 -1), (l-f-1-1-1), (l-a-1 -1-1-1)- (l-a-1-1-1 -6), (l-b-1-1-1-1)- (l-b-1 -1 -1 - 13), (l-b-1 -1 -2-1 ), (l-b-1 -1 -3-1 ), (l-b-2-1 -1 -1 ), (l-b-2-1 -2-1 -1 ), (l-b-2-1 -2-1 ), (l-b-2- 1-3-1), (l-b-3-1-1 -1), (l-b-3-1-2-1), (l-b-3-1 -3-1), (l-c-1 -1 -1 -1 ), (l-c-1 -1 -1 -2), (l-d- 1 -1-1-1), (l-d-1 -1-1-2), (l-f-1 -1-1-1), (l-a-1 -1-1 -3-0), (l-a-1 -1-1 -3-1 ), (l-a-1 -1 -1 -3-
2), (l-a-1 -1-1 -6-2), (l-b-1 -1 -1 -4-0), (l-b-1 -1 -1 -2-1 ), (l-b-1 -1 -1 -4-1 ), (l-b-1 -1 -1 -5-1 ), (l-b-1 -1-1-7-1 ), (l-b-1 -1-1 -8-1 ), (l-b-1-1-1-12-1), (l-b-1 -1 -1-13-1), (l-b-1 -1 -1 -1 -2), (l-b-1 -1-1 -4-2), (l-b-1 -1-1-4-2a), (l-b-1 -1-1 -5-2), (l-b-1 -1-1 -6-2), (l-b-1 -1-1 -7-2), (l-b-2-1 -1-1 -1 ), (l-b-2-1 -3-1-1 ), (l-b-3-1 -1-1 -1 ), (l-b-3-1 -2-1 -1 ), (l-b-3-1 -3-1-1), (I-
c-1-1-1-2-1), (l-d-1 -1 -1 -1 -0), and (l-d-1 -1 -1 -1 -1 ), as specified herein, as well as the N-oxides, salts, as the possible stereoisomeric forms thereof. Another embodiment comprises the compounds (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- (I- b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1- 1), (l-d-1-1 ), (l-e-1 -1 ), (l-f-1 -1 ), (l-a-1-1 -1), (l-b-1 -1 -1)- (l-b-1 -1 -3), (l-b-2-1 -1 )- (I- b-2-1-3), (l-b-3-1-1)- (l-b-3-1-3), (l-c-1-1-1), (l-d-1-1-1), (l-f-1-1-1), (l-a-1 -1 -1 -1 )- (l-a-1 -1-1-6), (l-b-1 -1-1-1)- (l-b-1-1 -1-13), (l-b-1 -1 -2-1 ), (l-b-1 -1 -3-1 ), (l-b-2-1 -1 - 1), (l-b-2-1 -2-1-1), (l-b-2-1 -2-1), (l-b-2-1 -3-1), (l-b-3-1 -1 -1 ), (l-b-3-1 -2-1), (l-b-3- 1-3-1), (l-c-1 -1 -1 -1 ), (l-c-1 -1-1 -2), (l-d-1 -1 -1 -1 ), (l-d-1 -1 -1 -2), (l-f-1 -1 -1 -1 ), (l-a-1 - 1-1 -3-0), (l-a-1 -1 -1-3-1), (l-a-1 -1-1 -3-2), (l-a-1 -1-1 -6-2), (l-b-1 -1-1 -4-0), (l-b-1-1 - 1-2-1), (l-b-1-1 -1 -4-1 ), (l-b-1-1 -1-5-1 ), (l-b-1-1 -1-7-1 ), (l-b-1 -1 -1 -8-1 ), (l-b-1-1 -1- 12-1), (l-b-1-1 -1-13-1), (l-b-1 -1-1-1-2), (l-b-1 -1-1 -4-2), (l-b-1 -1-1-4-2a), (l-b-1 -1 - 1-5-2), (l-b-1 -1-1 -6-2), (l-b-1-1 -1-7-2), (l-b-2-1 -1 -1 -1 ), (l-b-2-1 -3-1 -1 ), (l-b-3-1 -1- 1-1), (l-b-3-1 -2-1 -1 ), (l-b-3-1 -3-1-1), (l-c-1 -1 -1 -2-1 ), (l-d-1 -1 -1 -1 -0), and (l-d-1 -1 - 1 -1 -1 ), specified herein, as well as the salts as the possible stereoisomeric forms thereof.
The compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- ( l-b-3) , (l-c-1 ), (I- d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1-1), (I- e-1-1), (l-f-1 -1 ), (l-a-1 -1 -1 ), (l-b-1 -1 -1 )- (l-b-1 -1 -3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3- 1-1)- (l-b-3-1-3), (l-c-1-1-1), (l-d-1-1-1), (l-f-1-1-1), (l-a-1 -1 -1 -1 )- (l-a-1 -1 -1 -6), (I- b-1-1-1-1 )- (l-b-1-1 -1-13), (l-b-1-1-2-1), (l-b-1 -1-3-1), (l-b-2-1 -1 -1 ), (l-b-2-1 -2-1 - 1), (l-b-2-1 -2-1), (l-b-2-1 -3-1), (l-b-3-1 -1-1), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1 -1 - 1-1), (l-c-1 -1 -1 -2), (l-d-1 -1 -1 -1 ), (l-d-1 -1 -1 -2), (l-f-1 -1 -1 -1 ), (l-a-1 -1 -1 -3-0), (l-a-1 - 1-1 -3-1 ), (l-a-1 -1-1 -3-2), (l-a-1 -1-1 -6-2), (l-b-1 -1-1 -4-0), (l-b-1 -1 -1 -2-1 ), (l-b-1-1 - 1-4-1), (l-b-1 -1 -1 -5-1 ), (l-b-1 -1 -1-7-1), (l-b-1-1-1 -8-1), (l-b-1 -1 -1 -12-1 ), (l-b-1-1- 1-13-1), (l-b-1 -1 -1 -1 -2), (l-b-1 -1-1 -4-2), (l-b-1 -1-1-4-2a), (l-b-1 -1 -1 -5-2), (l-b-1 -1 - 1-6-2), (l-b-1 -1-1 -7-2), (l-b-2-1 -1-1 -1 ), (l-b-2-1 -3-1 -1 ), (l-b-3-1 -1 -1 -1 ), (l-b-3-1 -2- 1-1), (l-b-3-1 -3-1-1), (l-c-1 -1-1 -2-1), (l-d-1 -1-1 -1-0), and (l-d-1 -1 -1 -1 -1 ), as specified herein, have several centers of chirality and exist as stereochemically isomeric forms. The term "stereochemically isomeric forms" as used herein defines all the possible compounds made up of the same atoms bonded by the same sequence of bonds but having different three-dimensional structures
which are not interchangeable, which the compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1), (l-b-1 )- (l-b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1 ), (l-c-1-1 ), (l-d-1 -1), (l-e-1-1), (l-f-1 -1), (l-a-1-1 -1), (l-b-1-1-1)- (l-b-1-1-3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3-1 -1 )- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1 -1 -1 ), (l-f-1-1 -1 ), (l-a-1-1 -1 -1)- (l-a-1 -1 -1-6), (l-b-1-1 -1 -1)- (l-b-1 -1 -1 -13), (l-b-1 -1 -2-1 ), (l-b-1 -1 -3-1 ), (l-b-2-1 -1-1), (l-b-2-1 -2-1-1 ), (l-b-2-1 -2-1 ), (l-b-2-1 -3-1), (l-b-3-1 -1- 1), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1-1 -1-1), (l-c-1 -1 -1 -2), (l-d-1 -1 -1 -1 ), (l-d-1-1- 1-2), (l-f-1 -1-1 -1), (l-a-1 -1-1 -3-0), (l-a-1-1 -1-3-1 ), (l-a-1 -1-1 -3-2), (l-a-1 -1-1 -6-2), (l-b-1 -1-1 -4-0), (l-b-1-1 -1 -2-1 ), (l-b-1 -1 -1-4-1 ), (l-b-1 -1 -1 -5-1 ), (l-b-1 -1 -1 -7-1 ), (I- b-1-1-1-8-1), (l-b-1 -1 -1 -12-1 ), (l-b-1 -1 -1 -13-1 ), (l-b-1 -1 -1 -1 -2), (l-b-1 -1-1 -4-2), (I- b-1-1-1-4-2a), (l-b-1 -1-1 -5-2), (l-b-1 -1 -1 -6-2), (l-b-1 -1-1 -7-2), (l-b-2-1 -1 -1 -1 ), (I- b-2-1-3-1-1), (l-b-3-1 -1-1 -1 ), (l-b-3-1 -2-1 -1 ), (l-b-3-1 -3-1-1), (l-c-1 -1 -1 -2-1 ), (l-d- 1-1 -1-1-0), and (l-d-1 -1-1 -1-1), as specified herein, may possess.
Unless otherwise mentioned or indicated, the chemical designation of a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1), (l-b-1)- (l-b-3), (l-c-1), (l-d-1), (l-e- 1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1 -1 ), (l-e-1-1), (l-f-
1-1), (l-a-1-1-1), (l-b-1-1-1 )- (l-b-1-1-3), (l-b-2-1-1)- (l-b-2-1-3), (l-b-3-1-1)- (l-b- 3-1-3), (l-c-1-1 -1 ), (l-d-1-1 -1 ), (l-f-1 -1-1), (l-a-1 -1-1 -1 )- (l-a-1-1 -1-6), (l-b-1-1 -1 - 1)- (l-b-1 -1 -1 -13), (l-b-1 -1-2-1), (l-b-1-1-3-1), (l-b-2-1-1-1), (l-b-2-1 -2-1 -1 ), (l-b-
2-1-2-1), (l-b-2-1 -3-1), (l-b-3-1 -1-1), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1 -1 -1 -1 ), (I- c-1-1-1-2), (l-d-1-1-1 -1), (l-d-1 -1-1-2), (l-f-1-1-1-1), (l-a-1 -1-1 -3-0), (l-a-1 -1 -1 -3- 1), (l-a-1 -1-1 -3-2), (l-a-1 -1 -1 -6-2), (l-b-1 -1 -1 -4-0), (l-b-1 -1 -1 -2-1 ), (l-b-1 -1 -1 -4-1 ), (l-b-1 -1-1-5-1 ), (l-b-1 -1-1 -7-1 ), (l-b-1 -1 -1 -8-1 ), (l-b-1-1-1-12-1), (l-b-1 -1 -1 -13-1 ), (l-b-1 -1 -1 -1 -2), (l-b-1 -1-1 -4-2), (l-b-1 -1-1-4-2a), (l-b-1 -1 -1 -5-2), (l-b-1 -1 -1 -6-2), (l-b-1 -1 -1-7-2), (l-b-2-1 -1 -1-1 ), (l-b-2-1 -3-1 -1 ), (l-b-3-1 -1 -1 -1 ), (l-b-3-1 -2-1-1), (I- b-3-1-3-1-1), (l-c-1-1-1-2-1), (l-d-1-1-1-1-0), and (l-d-1 -1 -1 -1 -1 ), as specified herein, encompasses the mixture of all possible stereochemically isomeric forms, which said compound may possess. Said mixture may contain all diastereomers and/or enantiomers of the basic molecular structure of said compound. All stereochemically isomeric forms of the compounds of the present invention both in pure form or mixed with each other are intended to be embraced within the scope of the present invention.
Pure stereoisomeric forms of the compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1), (l-b-1 )- (l-b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1 -1 ), (l-b-2-1), (l-b-3-1 ), (l-c-1 -1 ), (l-d-1 -1), (l-e-1-1), (l-f-1 -1), (l-a-1-1 -1), (l-b-1-1 -1)- (l-b-1 -1 -3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3-1 -1 )- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1 -1 -1 ), (l-f-1 -1 -1 ), (l-a-1-1 -1 -1)- (l-a-1 -1 -1-6), (l-b-1-1 -1 -1)- (l-b-1 -1 -1 -13), (l-b-1 -1 -2-1 ), (l-b-1 -1 -3-1 ), (l-b-2-1 -1-1), (l-b-2-1 -2-1-1 ), (l-b-2-1 -2-1 ), (l-b-2-1 -3-1 ), (l-b-3-1 -1- 1 ), (l-b-3-1 -2-1 ), (l-b-3-1 -3-1 ), (l-c-1-1 -1-1), (l-c-1 -1 -1 -2), (l-d-1 -1 -1 -1 ), (l-d-1 -1 - 1 -2), (l-f-1 -1-1 -1), (l-a-1 -1 -1 -3-0), (l-a-1-1 -1-3-1 ), (l-a-1 -1 -1 -3-2), (l-a-1 -1 -1 -6-2), (l-b-1 -1 -1 -4-0), (l-b-1-1 -1 -2-1 ), (l-b-1 -1 -1-4-1 ), (l-b-1 -1 -1 -5-1 ), (l-b-1 -1 -1 -7-1 ), (I- b-1 -1 -1 -8-1 ), (l-b-1 -1 -1 -12-1 ), (l-b-1 -1 -1 -13-1 ), (l-b-1 -1 -1 -1 -2), (l-b-1 -1 -1 -4-2), (I- b-1 -1 -1 -4-2a), (l-b-1 -1 -1 -5-2), (l-b-1 -1 -1 -6-2), (l-b-1 -1 -1 -7-2), (l-b-2-1 -1 -1 -1 ), (I- b-2-1 -3-1 -1 ), (l-b-3-1 -1-1 -1 ), (l-b-3-1 -2-1 -1 ), (l-b-3-1 -3-1 -1 ), (l-c-1 -1 -1 -2-1 ), (l-d- 1 -1 -1 -1 -0), and (l-d-1 -1 -1 -1 -1 ), as specified herein, and intermediates as mentioned herein are defined as isomers substantially free of other enantiomeric or diastereomeric forms of the same basic molecular structure of said compounds or intermediates. In particular, the term “stereoisomerically pure” concerns compounds or intermediates having a stereoisomeric excess of at least 80% (i.e. minimum 90% of one isomer and maximum 10% of the other possible isomers) up to a stereoisomeric excess of 100% (i.e. 100% of one isomer and none of the other), more in particular, compounds or intermediates having a stereoisomeric excess of 90% up to 100%, even more in particular having a stereoisomeric excess of 94% up to 100% and most in particular having a stereoisomeric excess of 97% up to 100%. The terms “enantiomerically pure” and “diastereomerically pure” should be understood in a similar way, but then having regard to the enantiomeric excess, and the diastereomeric excess, respectively, of the mixture in question.
Pure stereoisomeric forms of the compounds and intermediates of this invention may be obtained by the application procedures known in the art. For instance, enantiomers may be separated from each other by the selective crystallization of their diastereomeric salts with optically active acids or bases. Examples thereof are tartaric acid, dibenzoyltartaric acid, ditoluoyltartaric acid and camphorsulfonic acid. Alternatively, enantiomers may be separated by chromatographic techniques using chiral stationary phases. Said pure
stereochemically isomeric forms may also be derived from the corresponding pure stereochemically isomeric forms of the appropriate starting materials, provided that the reaction occurs stereospecifically. Preferably, if a specific stereoisomer is desired, said compound will be synthesized by stereospecific methods of preparation. These methods will advantageously employ enantiomerically pure starting materials.
The diastereomeric racemates of the compounds (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1), (l-b-1 )- (l-b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1 ), (l-c-1-1 ), (l-d-1 -1), (l-e-1-1), (l-f-1 -1), (l-a-1-1 -1), (l-b-1-1-1)- (l-b-1-1-3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3-1 -1 )- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1 -1 -1 ), (l-f-1-1 -1 ), (l-a-1-1 -1 -1)- (l-a-1 -1 -1-6), (l-b-1-1 -1 -1)- (l-b-1 -1 -1 -13), (l-b-1 -1 -2-1 ), (l-b-1 -1 -3-1 ), (l-b-2-1 -1-1), (l-b-2-1 -2-1-1 ), (l-b-2-1 -2-1 ), (l-b-2-1 -3-1), (l-b-3-1 -1- 1), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1-1 -1-1), (l-c-1 -1 -1 -2), (l-d-1 -1 -1 -1 ), (l-d-1-1- 1-2), (l-f-1 -1-1 -1), (l-a-1 -1-1 -3-0), (l-a-1-1 -1-3-1 ), (l-a-1 -1-1 -3-2), (l-a-1 -1-1 -6-2), (l-b-1 -1-1 -4-0), (l-b-1-1 -1 -2-1 ), (l-b-1 -1 -1-4-1 ), (l-b-1 -1 -1 -5-1 ), (l-b-1 -1 -1 -7-1 ), (I- b-1-1-1-8-1), (l-b-1 -1 -1 -12-1 ), (l-b-1 -1 -1 -13-1 ), (l-b-1 -1 -1 -1 -2), (l-b-1 -1-1 -4-2), (I- b-1-1-1-4-2a), (l-b-1 -1-1 -5-2), (l-b-1 -1 -1 -6-2), (l-b-1 -1-1 -7-2), (l-b-2-1 -1 -1 -1 ), (I- b-2-1-3-1-1), (l-b-3-1 -1-1 -1 ), (l-b-3-1 -2-1 -1 ), (l-b-3-1 -3-1-1), (l-c-1 -1 -1 -2-1 ), (l-d- 1-1 -1-1-0), and (l-d-1 -1-1 -1-1), as specified herein, can be obtained separately by conventional methods. Appropriate physical separation methods that may advantageously be employed are, for example, selective crystallization and chromatography, e.g. column chromatography.
For some of the compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1), (l-b-1)- (I- b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1- 1), (l-d-1-1), (l-e-1-1), (l-f-1-1), (l-a-1-1 -1), (l-b-1 -1 -1)- (l-b-1 -1 -3), (l-b-2-1 -1 )- (I- b-2-1-3), (l-b-3-1-1)- (l-b-3-1-3), (l-c-1-1-1), (l-d-1-1-1), (l-f-1-1-1), (l-a-1 -1 -1 -1 )- (l-a-1 -1-1-6), (l-b-1 -1-1-1)- (l-b-1-1 -1-13), (l-b-1 -1 -2-1 ), (l-b-1 -1 -3-1 ), (l-b-2-1 -1 - 1), (l-b-2-1 -2-1-1), (l-b-2-1 -2-1), (l-b-2-1 -3-1), (l-b-3-1 -1 -1 ), (l-b-3-1 -2-1), (l-b-3- 1-3-1), (l-c-1 -1 -1 -1 ), (l-c-1 -1-1 -2), (l-d-1 -1 -1 -1 ), (l-d-1 -1 -1 -2), (l-f-1 -1 -1 -1 ), (l-a-1 - 1-1 -3-0), (l-a-1 -1 -1-3-1), (l-a-1 -1-1 -3-2), (l-a-1 -1-1 -6-2), (l-b-1 -1-1 -4-0), (l-b-1-1 - 1-2-1), (l-b-1-1 -1 -4-1 ), (l-b-1-1 -1-5-1 ), (l-b-1-1 -1-7-1 ), (l-b-1 -1 -1 -8-1 ), (l-b-1-1 -1- 12-1), (l-b-1-1 -1-13-1), (l-b-1 -1-1-1-2), (l-b-1 -1-1 -4-2), (l-b-1 -1-1-4-2a), (l-b-1 -1 -
1-5-2), (l-b-1-1 -1 -6-2), (l-b-1-1 -1-7-2), (l-b-2-1 -1 -1 -1 ), (l-b-2-1 -3-1 -1 ), (l-b-3-1 -1- 1-1), (l-b-3-1 -2-1 -1 ), (l-b-3-1 -3-1-1), (l-c-1 -1 -1 -2-1 ), (l-d-1 -1 -1 -1 -0), and (l-d-1 -1 - 1-1-1), as specified herein, N-oxides, salts, solvates, and the intermediates used in the preparation thereof, the absolute stereochemical configuration was not experimentally determined.
A person skilled in the art is able to determine the absolute configuration of such compounds using art-known methods such as, for example, X-ray diffraction.
The present invention is also intended to include all isotopes of atoms occurring on the present to a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- (l-b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1 ), (l-a-1 -1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c- 1-1), (l-d-1-1), (l-e-1-1), (l-f-1-1), (l-a-1-1-1 ), (l-b-1 -1 -1 )- (l-b-1-1-3), (l-b-2-1 -1 )- (l-b-2-1-3), (l-b-3-1-1 )- (l-b-3-1-3), (l-c-1-1 -1 ), (l-d-1 -1 -1), (l-f-1 -1-1), (l-a-1-1 -1 - 1)- (l-a-1-1-1 -6), (l-b-1-1-1-1)- (l-b-1 -1-1-13), (l-b-1 -1 -2-1 ), (l-b-1 -1 -3-1 ), (l-b-2- 1-1-1), (l-b-2-1-2-1 -1), (l-b-2-1 -2-1), (l-b-2-1 -3-1), (l-b-3-1 -1 -1 ), (l-b-3-1 -2-1), (I- b-3-1-3-1), (l-c-1 -1-1 -1 ), (l-c-1 -1-1 -2), (l-d-1-1 -1-1 ), (l-d-1-1 -1-2), (l-f-1 -1 -1 -1 ), (I- a-1-1 -1-3-0), (l-a-1-1 -1-3-1 ), (l-a-1 -1 -1 -3-2), (l-a-1 -1 -1 -6-2), (l-b-1 -1-1 -4-0), (l-b- 1-1 -1-2-1 ), (l-b-1 -1 -1-4-1 ), (l-b-1 -1 -1-5-1 ), (l-b-1 -1 -1-7-1), (l-b-1 -1 -1 -8-1 ), (l-b-1- 1-1-12-1), (l-b-1-1-1-13-1), (l-b-1-1-1 -1-2), (l-b-1-1 -1-4-2), (l-b-1 -1-1-4-2a), (l-b- 1-1 -1-5-2), (l-b-1 -1 -1-6-2), (l-b-1 -1 -1-7-2), (l-b-2-1 -1 -1 -1 ), (l-b-2-1 -3-1 -1 ), (l-b-3- 1-1 -1 -1 ), (l-b-3-1 -2-1 -1 ), (l-b-3-1 -3-1-1), (l-c-1 -1 -1 -2-1 ), (l-d-1 -1 -1 -1 -0), and (l-d-
1-1 -1-1-1), as specified herein. Isotopes include those atoms having the same atomic number but different mass numbers. By way of general example and without limitation, isotopes of hydrogen include tritium and deuterium. Isotopes of carbon include C-13 and C-14.
For therapeutic use, salts of the compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1)- (l-b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3- 1), (l-c-1-1), (l-d-1-1), (l-e-1-1), (l-f-1-1), (l-a-1-1-1), (l-b-1-1-1)- (l-b-1-1-3), (l-b-
2-1-1)- (l-b-2-1-3), (l-b-3-1-1)- (l-b-3-1-3), (l-c-1 -1 -1 ), (l-d-1 -1 -1 ), (l-f-1 -1 -1 ), (l-a- 1-1-1-1)- (l-a-1 -1-1-6), (l-b-1-1-1-1)- (l-b-1-1 -1-13), (l-b-1 -1 -2-1 ), (l-b-1 -1 -3-1 ), (I- b-2-1-1-1), (l-b-2-1 -2-1 -1 ), (l-b-2-1-2-1), (l-b-2-1 -3-1 ), (l-b-3-1 -1 -1 ), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1-1 -1 -1), (l-c-1-1 -1 -2), (l-d-1 -1 -1 -1), (l-d-1 -1 -1-2), (l-f-1 -1 -1 -1 ),
(l-a-1 -1 -1-3-0), (l-a-1-1 -1 -3-1 ), (l-a-1 -1 -1 -3-2), (l-a-1 -1 -1 -6-2), (l-b-1 -1 -1 -4-0), (I- b-1-1 -1-2-1 ), (l-b-1-1 -1-4-1 ), (l-b-1-1 -1-5-1 ), (l-b-1-1 -1-7-1 ), (l-b-1 -1 -1 -8-1 ), (l-b- 1-1-1-12-1), (l-b-1-1-1 -13-1 ), (l-b-1-1 -1-1-2), (l-b-1 -1-1 -4-2), (l-b-1 -1-1-4-2a), (I- b-1-1 -1-5-2), (l-b-1-1 -1-6-2), (l-b-1-1 -1-7-2), (l-b-2-1 -1 -1 -1 ), (l-b-2-1 -3-1 -1 ), (l-b- 3-1 -1-1 -1 ), (l-b-3-1 -2-1 -1), (l-b-3-1 -3-1-1), (l-c-1 -1 -1 -2-1 ), (l-d-1 -1 -1 -1 -0), and (I- d-1-1-1-1-1), as specified herein, are those wherein the counter-ion is pharmaceutically acceptable, which salts can be referred to as pharmaceutically acceptable acid and base addition salts. However, salts of acids and bases that are non-pharmaceutically acceptable may also find use, for example, in the preparation or purification of a pharmaceutically acceptable compound. All salts, whether pharmaceutically acceptable or not, are included within the ambit of the present invention.
The pharmaceutically acceptable acid and base addition salts as mentioned hereinabove are meant to comprise the therapeutically active non- toxic acid and base addition salt forms that the compounds (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1), (l-b-1)- (l-b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1 ), (l-d-1 -1), (l-e-1-1), (l-f-1 -1), (l-a-1-1 -1), (l-b-1-1-1)- (l-b-1-1-3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3-1 -1 )- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1 -1 -1 ), (l-f-1-1 -1 ), (l-a-1-1 -1 -1)- (l-a-1 -1 -1-6), (l-b-1-1 -1 -1)- (l-b-1 -1 -1 -13), (l-b-1 -1 -2-1 ), (l-b-1 -1 -3-1 ), (l-b-2-1 -1-1), (l-b-2-1 -2-1-1 ), (l-b-2-1 -2-1 ), (l-b-2-1 -3-1), (l-b-3-1 -1- 1), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1-1 -1-1), (l-c-1 -1 -1 -2), (l-d-1 -1 -1 -1 ), (l-d-1-1- 1-2), (l-f-1 -1-1 -1), (l-a-1 -1-1 -3-0), (l-a-1-1 -1-3-1), (l-a-1 -1-1 -3-2), (l-a-1 -1-1 -6-2), (l-b-1 -1-1 -4-0), (l-b-1-1 -1 -2-1 ), (l-b-1 -1 -1-4-1 ), (l-b-1 -1 -1 -5-1 ), (l-b-1 -1 -1 -7-1 ), (I- b-1-1-1-8-1), (l-b-1 -1 -1 -12-1 ), (l-b-1 -1 -1 -13-1 ), (l-b-1 -1 -1 -1 -2), (l-b-1 -1-1 -4-2), (I- b-1-1-1-4-2a), (l-b-1 -1-1 -5-2), (l-b-1 -1 -1 -6-2), (l-b-1 -1-1 -7-2), (l-b-2-1 -1 -1 -1 ), (I- b-2-1-3-1-1), (l-b-3-1 -1-1 -1 ), (l-b-3-1 -2-1 -1 ), (l-b-3-1 -3-1-1), (l-c-1 -1 -1 -2-1 ), (l-d- 1-1 -1-1-0), and (l-d-1 -1-1 -1-1), as specified herein, are able to form. The pharmaceutically acceptable acid addition salts can conveniently be obtained by treating the base form with such appropriate acid in an anion form. Appropriate anions comprise, for example, trifluoroacetate, acetate, benzenesulfonate, benzoate, bicarbonate, bitartrate, bromide, calcium edetate, camsyiate, carbonate, chloride, citrate, dihydrochloride, edetate, edisylate, estolate, esylate, fumarate, gluceptate, gluconate, glutamate, glycollylarsanilate,
hexylresorcinate, hydrabamine, hydrobromide, hydrochloride, hydroxynaphthoate, iodide, isethionate, lactate, lactobionate, malate, maleate, mandelate, mesylate, methylbromide, methylnitrate, methylsulfate, mucate, napsylate, nitrate, pamoate (embonate), pantothenate, phosphate/diphosphate, polygalacturonate, salicylate, stearate, subacetate, succinate, sulfate, tannate, tartrate, teoclate, triethiodide, and the like. The counterion of choice can be introduced using ion exchange resins. Conversely said salt forms can be converted by treatment with an appropriate base into the free base form.
The compounds (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- ( l-b-3) , (l-c-1 ), (I- d-1), (l-e-1), (l-f-1 ), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1-1 ), (I- e-1-1), (l-f-1 -1 ), (l-a-1 -1 -1 ), (l-b-1 -1 -1 )- (l-b-1 -1 -3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3- 1-1)- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1-1-1), (l-f-1 -1 -1 ), (l-a-1 -1 -1 -1 )- (l-a-1 -1 -1 -6), (I- b-1-1-1-1 )- (l-b-1-1 -1-13), (l-b-1-1-2-1 ), (l-b-1 -1-3-1), (l-b-2-1 -1 -1 ), (l-b-2-1 -2-1 - 1), (l-b-2-1 -2-1), (l-b-2-1 -3-1), (l-b-3-1 -1-1), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1 -1 - 1-1), (l-c-1 -1 -1 -2), (l-d-1-1-1-1), (l-d-1-1-1-2), (l-f-1 -1 -1 -1 ), (l-a-1 -1 -1 -3-0), (l-a-1 - 1-1 -3-1 ), (l-a-1 -1-1 -3-2), (l-a-1 -1-1 -6-2), (l-b-1 -1-1 -4-0), (l-b-1 -1 -1 -2-1 ), (l-b-1-1 - 1-4-1), (l-b-1 -1 -1 -5-1 ), (l-b-1 -1 -1-7-1), (l-b-1-1-1 -8-1), (l-b-1 -1 -1 -12-1 ), (l-b-1-1- 1-13-1), (l-b-1 -1 -1 -1 -2), (l-b-1 -1-1 -4-2), (l-b-1 -1-1-4-2a), (l-b-1 -1 -1 -5-2), (l-b-1 -1 - 1-6-2), (l-b-1 -1-1 -7-2), (l-b-2-1 -1-1 -1 ), (l-b-2-1 -3-1 -1 ), (l-b-3-1 -1 -1 -1 ), (l-b-3-1 -2- 1-1), (l-b-3-1 -3-1-1), (l-c-1 -1-1 -2-1), (l-d-1-1 -1-1-0), and (l-d-1-1-1-1-1), as specified herein, containing an acidic proton may also be converted into their nontoxic metal or amine addition salt forms by treatment with appropriate organic and inorganic bases in a cation form. Appropriate basic salts comprise those formed with organic cations such as benzathine, chloroprocaine, choline, diethanolamine, ethylenediamine, meglumine, procaine, and the like; and those formed with metallic cations such as aluminum, calcium, lithium, magnesium, potassium, sodium, zinc, and the like. Conversely said salt forms can be converted by treatment with an appropriate acid into the free form.
The term addition salt as used hereinabove also comprises the solvates which the compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- (l-b-3), (l-c-1 ), (I- d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1-1), (I- e-1-1), (l-f-1 -1 ), (l-a-1 -1 -1 ), (l-b-1-1-1)- (l-b-1 -1 -3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3-
1-1)- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1 -1 -1 ), (l-f-1 -1 -1 ), (l-a-1 -1 -1 -1 )- (l-a-1 -1 -1 -6), (I- b-1-1-1-1 )- (l-b-1-1 -1-13), (l-b-1-1-2-1), (l-b-1 -1-3-1), (l-b-2-1 -1 -1 ), (l-b-2-1 -2-1 - 1), (l-b-2-1 -2-1), (l-b-2-1 -3-1), (l-b-3-1 -1-1), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1 -1 - 1-1), (l-c-1 -1 -1 -2), (l-d-1 -1 -1 -1 ), (l-d-1 -1 -1 -2), (l-f-1 -1 -1 -1 ), (l-a-1 -1 -1 -3-0), (l-a-1 - 1-1 -3-1 ), (l-a-1 -1-1 -3-2), (l-a-1 -1-1 -6-2), (l-b-1 -1-1 -4-0), (l-b-1 -1 -1 -2-1 ), (l-b-1-1 - 1-4-1), (l-b-1-1-1-5-1), (l-b-1 -1 -1-7-1), (l-b-1-1-1 -8-1), (l-b-1 -1 -1 -12-1 ), (l-b-1-1- 1-13-1), (l-b-1 -1 -1 -1 -2), (l-b-1 -1-1 -4-2), (l-b-1 -1-1-4-2a), (l-b-1 -1 -1 -5-2), (l-b-1 -1 - 1-6-2), (l-b-1 -1-1 -7-2), (l-b-2-1 -1-1 -1 ), (l-b-2-1 -3-1 -1 ), (l-b-3-1 -1 -1 -1 ), (l-b-3-1 -2- 1-1), (l-b-3-1 -3-1-1), (l-c-1 -1-1 -2-1), (l-d-1 -1-1 -1-0), and (l-d-1 -1 -1 -1 -1 ), as specified herein, as well as the salts thereof, are able to form. Such solvates are for example hydrates, alcoholates and the like.
The N-oxide forms of the present compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1), (l-b-1)- (l-b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1 -1), (l-e-1-1), (l-f-1 -1), (l-a-1-1 -1), (l-b-1-1-1)- (l-b-1-1-3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3-1 -1 )- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1 -1 -1 ), (l-f-1-1 -1 ), (l-a-1-1 -1 -1)- (l-a-1 -1 -1-6), (l-b-1-1 -1 -1)- (l-b-1 -1 -1 -13), (l-b-1-1-2-1), (l-b-1 -1 -3-1 ), (l-b-2-1 -1-1), (l-b-2-1 -2-1-1 ), (l-b-2-1 -2-1 ), (l-b-2-1 -3-1), (l-b-3-1 -1- 1), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1-1 -1-1), (l-c-1 -1 -1 -2), (l-d-1 -1 -1 -1 ), (l-d-1-1- 1-2), (l-f-1 -1-1 -1), (l-a-1 -1-1 -3-0), (l-a-1-1 -1-3-1 ), (l-a-1 -1-1 -3-2), (l-a-1 -1-1 -6-2), (l-b-1 -1-1 -4-0), (l-b-1-1 -1 -2-1 ), (l-b-1 -1 -1-4-1 ), (l-b-1-1-1-5-1), (l-b-1 -1 -1 -7-1 ), (I- b-1-1-1-8-1), (l-b-1 -1 -1 -12-1 ), (l-b-1 -1 -1 -13-1 ), (l-b-1 -1 -1 -1 -2), (l-b-1 -1-1 -4-2), (I- b-1-1-1-4-2a), (l-b-1 -1-1 -5-2), (l-b-1 -1 -1 -6-2), (l-b-1 -1-1 -7-2), (l-b-2-1 -1 -1 -1 ), (I- b-2-1-3-1-1), (l-b-3-1 -1-1 -1 ), (l-b-3-1 -2-1 -1 ), (l-b-3-1 -3-1-1), (l-c-1 -1 -1 -2-1 ), (l-d-
1-1 -1-1-0), and (l-d-1 -1-1 -1-1), as specified herein, are meant to comprise the compounds of formula (I) wherein one or several nitrogen atoms are oxidized to the so-called N-oxide.
It will be appreciated that the compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1)- (l-b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3- 1), (l-c-1-1), (l-d-1 -1), (l-e-1-1), (l-f-1-1), (l-a-1 -1-1 ), (l-b-1-1-1)- (l-b-1-1-3), (l-b-
2-1-1)- (l-b-2-1 -3), (l-b-3-1 -1 )- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1 -1 -1 ), (l-f-1 -1 -1 ), (l-a- 1-1-1-1)- (l-a-1 -1-1-6), (l-b-1 -1 -1-1)- (l-b-1-1 -1-13), (l-b-1-1-2-1), (l-b-1 -1 -3-1 ), (I- b-2-1-1-1), (l-b-2-1 -2-1 -1 ), (l-b-2-1 -2-1 ), (l-b-2-1 -3-1 ), (l-b-3-1 -1 -1 ), (l-b-3-1 -2-1),
(l-b-3-1 -3-1 ), (l-c-1-1 -1 -1), (l-c-1-1 -1 -2), (l-d-1-1-1-1), (l-d-1-1-1-2), (l-f-1 -1 -1 -1 ), (l-a-1 -1 -1-3-0), (l-a-1-1 -1 -3-1 ), (l-a-1 -1 -1 -3-2), (l-a-1 -1 -1 -6-2), (l-b-1 -1 -1 -4-0), (I- b-1-1 -1-2-1 ), (l-b-1-1 -1-4-1 ), (l-b-1-1 -1-5-1 ), (l-b-1-1 -1-7-1 ), (l-b-1 -1 -1 -8-1 ), (l-b- 1-1-1-12-1), (l-b-1-1-1 -13-1 ), (l-b-1-1 -1-1-2), (l-b-1 -1-1 -4-2), (l-b-1 -1-1-4-2a), (I- b-1-1 -1-5-2), (l-b-1-1 -1-6-2), (l-b-1-1 -1-7-2), (l-b-2-1 -1 -1 -1 ), (l-b-2-1 -3-1 -1 ), (l-b- 3-1 -1-1 -1 ), (l-b-3-1 -2-1 -1), (l-b-3-1 -3-1-1), (l-c-1 -1 -1 -2-1 ), (l-d-1-1-1-1-0), and (I- d-1-1-1-1-1), as specified herein, may have metal binding, chelating, complex forming properties and therefore may exist as metal complexes or metal chelates. Such metalated derivatives of the compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1), (l-b-1)- (l-b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1 -1), (l-e-1-1), (l-f-1 -1), (l-a-1-1 -1), (l-b-1-1-1)- (l-b-1-1-3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3-1 -1 )- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1 -1 -1 ), (l-f-1-1 -1 ), (l-a-1-1 -1 -1)- (l-a-1 -1 -1-6), (l-b-1-1 -1 -1)- (l-b-1 -1 -1 -13), (l-b-1 -1 -2-1 ), (l-b-1 -1 -3-1 ), (l-b-2-1 -1-1), (l-b-2-1 -2-1-1 ), (l-b-2-1 -2-1 ), (l-b-2-1 -3-1), (l-b-3-1 -1- 1), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1-1 -1-1), (l-c-1 -1 -1 -2), (l-d-1-1-1-1), (l-d-1-1- 1-2), (l-f-1 -1-1 -1), (l-a-1 -1-1 -3-0), (l-a-1-1 -1-3-1 ), (l-a-1 -1-1 -3-2), (l-a-1 -1-1 -6-2), (l-b-1 -1-1 -4-0), (l-b-1-1 -1 -2-1 ), (l-b-1 -1 -1-4-1 ), (l-b-1 -1 -1 -5-1 ), (l-b-1 -1 -1 -7-1 ), (I- b-1-1-1-8-1), (l-b-1 -1 -1 -12-1 ), (l-b-1 -1 -1 -13-1 ), (l-b-1 -1 -1 -1 -2), (l-b-1 -1-1 -4-2), (I- b-1-1-1-4-2a), (l-b-1 -1-1 -5-2), (l-b-1 -1 -1 -6-2), (l-b-1 -1-1 -7-2), (l-b-2-1 -1 -1 -1 ), (I- b-2-1-3-1-1), (l-b-3-1 -1-1 -1 ), (l-b-3-1 -2-1 -1 ), (l-b-3-1 -3-1-1), (l-c-1 -1 -1 -2-1 ), (l-d- 1-1-1-1-0), and (l-d-1 -1-1 -1-1), as specified herein, are intended to be included within the scope of the present invention.
Some of the compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1), (l-b-1)- (I- b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1- 1), (l-d-1-1), (l-e-1-1), (l-f-1-1), (l-a-1-1 -1), (l-b-1-1-1)- (l-b-1 -1 -3), (l-b-2-1 -1 )- (I- b-2-1-3), (l-b-3-1-1)- (l-b-3-1-3), (l-c-1-1-1), (l-d-1-1-1), (l-f-1-1-1), (l-a-1 -1 -1 -1 )- (l-a-1 -1-1-6), (l-b-1 -1-1-1)- (l-b-1-1 -1-13), (l-b-1 -1 -2-1 ), (l-b-1 -1 -3-1 ), (l-b-2-1 -1 - 1), (l-b-2-1 -2-1-1), (l-b-2-1 -2-1), (l-b-2-1 -3-1), (l-b-3-1 -1 -1 ), (l-b-3-1 -2-1), (l-b-3- 1-3-1), (l-c-1 -1 -1 -1 ), (l-c-1 -1-1 -2), (l-d-1-1-1-1), (l-d-1-1-1-2), (l-f-1 -1 -1 -1 ), (l-a-1 - 1-1 -3-0), (l-a-1 -1 -1-3-1), (l-a-1 -1-1 -3-2), (l-a-1 -1-1 -6-2), (l-b-1 -1-1 -4-0), (l-b-1-1 - 1-2-1), (l-b-1-1 -1 -4-1 ), (l-b-1-1 -1-5-1 ), (l-b-1-1 -1-7-1 ), (l-b-1 -1 -1 -8-1 ), (l-b-1-1 -1- 12-1), (l-b-1-1 -1-13-1), (l-b-1 -1-1-1-2), (l-b-1 -1-1 -4-2), (l-b-1 -1-1-4-2a), (l-b-1 -1 -
1-5-2), (l-b-1-1 -1 -6-2), (l-b-1-1 -1-7-2), (l-b-2-1 -1 -1 -1 ), (l-b-2-1 -3-1 -1 ), (l-b-3-1 -1- 1-1), (l-b-3-1 -2-1 -1 ), (l-b-3-1 -3-1-1), (l-c-1 -1 -1 -2-1 ), (l-d-1-1-1-1-0), and (l-d-1 -1 - 1-1-1), as specified herein, may also exist in their tautomeric form. Such forms although not explicitly indicated in the above formula are intended to be included within the scope of the present invention.
In a further aspect, the present invention concerns a pharmaceutical composition comprising a therapeutically effective amount of a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- (l-b-3), (l-c-1), (l-d-1), (l-e-1 ), (l-f-1 ), (l-a- 1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1-1), (l-e-1-1), (l-f-1-1), (l-a-1 -1 -
1), (l-b-1 -1-1)- (l-b-1 -1-3), (l-b-2-1 -1)- (l-b-2-1 -3), (l-b-3-1 -1)- (l-b-3-1 -3), (l-c-1 - 1-1), (l-d-1-1 -1), (l-f-1-1-1), (l-a-1 -1-1-1)- (l-a-1-1-1 -6), (l-b-1 -1 -1 -1 )- (l-b-1 -1 -1 - 13), (l-b-1 -1 -2-1 ), (l-b-1 -1 -3-1 ), (l-b-2-1 -1 -1 ), (l-b-2-1 -2-1 -1 ), (l-b-2-1 -2-1 ), (l-b-2- 1-3-1), (l-b-3-1-1 -1), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1 -1 -1 -1 ), (l-c-1 -1 -1 -2), (l-d- 1 -1-1-1), (l-d-1 -1-1-2), (l-f-1 -1-1-1), (l-a-1 -1-1 -3-0), (l-a-1 -1-1 -3-1 ), (l-a-1 -1 -1 -3-
2), (l-a-1 -1-1 -6-2), (l-b-1 -1 -1 -4-0), (l-b-1 -1 -1 -2-1 ), (l-b-1 -1 -1 -4-1 ), (l-b-1 -1 -1 -5-1 ), (l-b-1 -1-1-7-1 ), (l-b-1 -1-1 -8-1 ), (l-b-1-1-1-12-1), (l-b-1 -1 -1-13-1), (l-b-1 -1 -1 -1 -2), (l-b-1 -1-1 -4-2), (l-b-1 -1-1-4-2a), (l-b-1 -1-1 -5-2), (l-b-1 -1-1 -6-2), (l-b-1 -1-1 -7-2), (l-b-2-1 -1-1 -1 ), (l-b-2-1 -3-1-1 ), (l-b-3-1 -1-1 -1 ), (l-b-3-1 -2-1 -1 ), (l-b-3-1 -3-1-1), (I- c-1-1-1-2-1), (l-d-1-1-1-1-0), and (l-d-1 -1-1 -1-1), as specified herein, and a pharmaceutically acceptable carrier. A therapeutically effective amount in this context is an amount sufficient to prophylactically act against, to stabilize or reduce illnesses mediated by protein kinases in ill subjects or subjects being at risk of being ill, in particular a protein kinase selected from the group consisting of CSF1R, FLT3, Kit, PDGFRB (PDGFR beta), PDGFRA (PDGFR alfa), ABL1, ACVR1B (ALK4), AKT1 (PKB alpha), AMPK A1/B1/G1, AURKA (Aurora A), BTK, CDK1/cyclin B, CHEK1 (CHK1), CSNK1G2 (CK1 gamma 2), CSNK2A1 (CK2 alpha 1), DYRK3, EGFR (ErbB1), EPHA2, ERBB2 (HER2), FGFR1, FRAP1 (mTOR), GSK3B (GSK3 beta), IGF1 R, IKBKB (IKK beta), INSR, IRAK4, JAK3, KDR (VEGFR2), LCK, MAP2K1 (MEK1), MAP4K4 (HGK), MAPK1 (ERK2), MAPK14 (p38 alpha), MAPK3 (ERK1), MAPK8 (JNK1), MARK2, MET (cMet), NEK1, PAK4, PHKG2, PIM1, PLK1, PRKACA (PKA), PRKCB1 (PKC beta I), ROCK1, RPS6KA3 (RSK2), RPS6KB1 (p70S6K), SRC, SYK, and TEK (Tie2). Preferably, the protein kinase is selected from the group consisting of
CSF1R, FLT3, Kit, PDGFRB (PDGFR beta), PDGFRA (PDGFR alpha).
Examples of illnesses mediated by protein kinases include in particular of illnesses mediated by protein kinases include in particular cancer, metabolic disorders (such as diabetes), inflammatory and autoimmune disorders (such as inflammatory bowel diseases, e.g. Crohn’s disease and ulcerative colitis, inflammatory pulmonary diseases, rheumatoid arthritis, lupus nephritis, systemic lupus erythematosus and psoriasis and psoriasis arthritis), neurological disorders (such as Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, Charcot-Marie-Tooth neuropathy, amyotrophic lateral sclerosis and epilepsy), atherosclerosis and cardiovascular diseases, Sjogren Syndrome, renal allograft rejection, viral induced diseases, circulatory diseases, bone osteolysis and osteoporosis, osteoarthritis, sarcopenia, Langerhans cell histiocytosis, spinal cord injury, endometriosis, asthma and allergic asthma, eye diseases (such as retinopathies, age-related macular degeneration and uveitis) chronic and neuropathic pain, and fibro-proliferative diseases.
In still a further aspect, this invention relates to a process of preparing a pharmaceutical composition as specified herein, which comprises intimately mixing a pharmaceutically acceptable carrier with a therapeutically effective amount of a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- ( l-b-3) , (l-c-1 ), (I- d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1-1), (I- e-1-1), (l-f-1 -1 ), (l-a-1 -1 -1 ), (l-b-1 -1 -1 )- (l-b-1 -1 -3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3- 1-1)- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1-1-1), (l-f-1 -1 -1 ), (l-a-1 -1 -1 -1 )- (l-a-1 -1 -1 -6), (I- b-1-1-1-1 )- (l-b-1-1 -1-13), (l-b-1-1-2-1), (l-b-1 -1-3-1), (l-b-2-1 -1 -1 ), (l-b-2-1 -2-1 - 1), (l-b-2-1 -2-1), (l-b-2-1 -3-1), (l-b-3-1 -1-1), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1 -1 - 1-1), (l-c-1 -1 -1 -2), (l-d-1-1-1-1), (l-d-1-1-1-2), (l-f-1 -1 -1 -1 ), (l-a-1 -1 -1 -3-0), (l-a-1 - 1-1 -3-1 ), (l-a-1 -1-1 -3-2), (l-a-1 -1-1 -6-2), (l-b-1 -1-1 -4-0), (l-b-1 -1 -1 -2-1 ), (l-b-1-1 - 1-4-1), (l-b-1 -1 -1 -5-1 ), (l-b-1 -1 -1-7-1), (l-b-1-1-1 -8-1), (l-b-1 -1 -1 -12-1 ), (l-b-1-1- 1-13-1), (l-b-1 -1 -1 -1 -2), (l-b-1 -1-1 -4-2), (l-b-1 -1 -1 -4-2a), (l-b-1 -1 -1 -5-2), (l-b-1 -1 - 1-6-2), (l-b-1 -1-1 -7-2), (l-b-2-1 -1-1 -1 ), (l-b-2-1 -3-1 -1 ), (l-b-3-1 -1 -1 -1 ), (l-b-3-1 -2- 1-1), (l-b-3-1 -3-1-1), (l-c-1 -1-1 -2-1), (l-d-1-1 -1-1-0), and (l-d-1-1-1-1-1), as specified herein.
Therefore, the compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1), (l-b-1)- (I-
b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1- 1 ), (l-d-1-1 ), (l-e-1 -1 ), (l-f-1 -1 ), (l-a-1-1 -1), (l-b-1 -1 -1)- (l-b-1 -1 -3), (l-b-2-1 -1 )- (I- b-2-1-3), (l-b-3-1-1)- (l-b-3-1-3), (l-c-1-1-1), (l-d-1-1-1), (l-f-1-1-1), (l-a-1 -1 -1 -1 )- (l-a-1 -1 -1 -6), (l-b-1 -1-1-1)- (l-b-1-1 -1-13), (l-b-1 -1 -2-1 ), (l-b-1 -1 -3-1 ), (l-b-2-1 -1 - 1 ), (l-b-2-1 -2-1-1), (l-b-2-1 -2-1 ), (l-b-2-1 -3-1 ), (l-b-3-1 -1 -1 ), (l-b-3-1 -2-1 ), (l-b-3- 1 -3-1 ), (l-c-1 -1 -1 -1 ), (l-c-1 -1 -1 -2), (l-d-1 -1 -1 -1 ), (l-d-1 -1 -1 -2), (l-f-1 -1 -1 -1 ), (l-a-1 - 1-1 -3-0), (l-a-1 -1 -1-3-1), (l-a-1 -1 -1 -3-2), (l-a-1 -1 -1 -6-2), (l-b-1 -1 -1 -4-0), (l-b-1-1 - 1 -2-1 ), (l-b-1-1 -1 -4-1 ), (l-b-1-1 -1-5-1 ), (l-b-1-1 -1-7-1 ), (l-b-1 -1 -1 -8-1 ), (l-b-1-1 -1- 12-1 ), (l-b-1-1 -1-13-1), (l-b-1 -1-1-1-2), (l-b-1 -1 -1 -4-2), (l-b-1 -1 -1 -4-2a), (l-b-1 -1 - 1 -5-2), (l-b-1 -1 -1 -6-2), (l-b-1-1 -1-7-2), (l-b-2-1 -1 -1 -1 ), (l-b-2-1 -3-1 -1 ), (l-b-3-1 -1- 1 -1 ), (l-b-3-1 -2-1 -1 ), (l-b-3-1 -3-1 -1 ), (l-c-1 -1 -1 -2-1 ), (l-d-1 -1 -1 -1 -0), and (l-d-1 -1 - 1 -1 -1 ), as specified herein, may be formulated into various pharmaceutical forms for administration purposes. As appropriate compositions there may be cited all compositions usually employed for systemically administering drugs. To prepare the pharmaceutical compositions of this invention, an effective amount of the particular compound, optionally in addition salt form or metal complex, as the active ingredient is combined in intimate admixture with a pharmaceutically acceptable carrier, which carrier may take a wide variety of forms depending on the form of preparation desired for administration. These pharmaceutical compositions are desirable in unitary dosage form suitable, particularly, for administration orally, rectally, percutaneously, or by parenteral injection. For example, in preparing the compositions in oral dosage form, any of the usual pharmaceutical media may be employed such as, for example, water, glycols, oils, alcohols and the like in the case of oral liquid preparations such as suspensions, syrups, elixirs, emulsions and solutions; or solid carriers such as starches, sugars, kaolin, lubricants, binders, disintegrating agents and the like in the case of powders, pills, capsules, and tablets.
Because of their ease in administration, tablets and capsules represent the most advantageous oral dosage unit forms, in which case solid pharmaceutical carriers are obviously employed. For parenteral compositions, the carrier will usually comprise sterile water, at least in large part, though other ingredients, for example, to aid solubility, may be included. Injectable solutions, for example, may be prepared in which the carrier comprises saline solution, glucose solution or a mixture of saline and glucose solution. Injectable
suspensions may also be prepared in which case appropriate liquid carriers, suspending agents and the like may be employed. Also included are solid form preparations, which are intended to be converted, shortly before use, to liquid form preparations. In the compositions suitable for percutaneous administration, the carrier optionally comprises a penetration enhancing agent and/or a suitable wetting agent, optionally combined with suitable additives of any nature in minor proportions, which additives do not introduce a significant deleterious effect on the skin.
The compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- ( l-b-3) , (l-c-1 ), (I- d-1), (l-e-1), (l-f-1 ), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1-1 ), (I- e-1-1), (l-f-1 -1 ), (l-a-1 -1 -1 ), (l-b-1 -1 -1 )- (l-b-1 -1 -3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3-
1-1)- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1-1-1), (l-f-1 -1 -1 ), (l-a-1 -1 -1 -1 )- (l-a-1 -1 -1 -6), (I- b-1-1-1-1 )- (l-b-1-1 -1-13), (l-b-1-1-2-1 ), (l-b-1 -1-3-1), (l-b-2-1 -1 -1 ), (l-b-2-1 -2-1 - 1), (l-b-2-1 -2-1), (l-b-2-1 -3-1), (l-b-3-1 -1-1), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1 -1 - 1-1), (l-c-1 -1 -1 -2), (l-d-1-1-1-1), (l-d-1-1-1-2), (l-f-1 -1 -1 -1 ), (l-a-1 -1 -1 -3-0), (l-a-1 - 1-1 -3-1 ), (l-a-1 -1-1 -3-2), (l-a-1 -1-1 -6-2), (l-b-1 -1-1 -4-0), (l-b-1 -1 -1 -2-1 ), (l-b-1-1 - 1-4-1), (l-b-1-1-1-5-1), (l-b-1 -1 -1-7-1), (l-b-1-1-1 -8-1), (l-b-1 -1 -1 -12-1 ), (l-b-1-1- 1-13-1), (l-b-1 -1 -1 -1 -2), (l-b-1 -1-1 -4-2), (l-b-1 -1-1-4-2a), (l-b-1 -1 -1 -5-2), (l-b-1 -1 - 1-6-2), (l-b-1 -1-1 -7-2), (l-b-2-1 -1-1 -1 ), (l-b-2-1 -3-1 -1 ), (l-b-3-1 -1 -1 -1 ), (l-b-3-1 -2- 1-1), (l-b-3-1 -3-1-1), (l-c-1 -1-1 -2-1), (l-d-1-1 -1-1-0), and (l-d-1-1-1-1-1), as specified herein, of the present invention may also be administered via oral inhalation or insufflation by means of methods and formulations employed in the art for administration via this way. Thus, in general the compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1), (l-b-1)- (l-b-3), (l-c-1), (l-d-1), (l-e-1), (l-f-1), (l-a-1-1), (l-b-1- 1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1-1), (l-e-1-1), (l-f-1-1), (l-a-1-1-1), (l-b-1 -1 - 1)- (l-b-1-1-3), (l-b-2-1 -1)- (l-b-2-1-3), (l-b-3-1 -1)- (l-b-3-1-3), (l-c-1-1-1), (l-d-1-
1-1), (l-f-1 -1 -1 ), (l-a-1 -1 -1 -1 )- (l-a-1 -1 -1 -6), (l-b-1 -1 -1 -1 )- (l-b-1 -1 -1 -13), (l-b-1-1-
2-1), (l-b-1 -1-3-1), (l-b-2-1 -1-1), (l-b-2-1 -2-1-1), (l-b-2-1 -2-1), (l-b-2-1 -3-1), (l-b-
3-1-1-1), (l-b-3-1 -2-1), (l-b-3-1 -3-1), (l-c-1 -1 -1 -1 ), (l-c-1 -1 -1 -2), (l-d-1-1-1-1), (I- d-1-1-1-2), (l-f-1 -1-1-1), (l-a-1 -1-1 -3-0), (l-a-1-1-1 -3-1), (l-a-1 -1-1 -3-2), (l-a-1-1- 1-6-2), (l-b-1 -1-1 -4-0), (l-b-1-1 -1-2-1 ), (l-b-1 -1 -1 -4-1 ), (l-b-1-1-1-5-1), (l-b-1-1 -1- 7-1), (l-b-1 -1 -1 -8-1 ), (l-b-1 -1 -1 -12-1 ), (l-b-1 -1 -1 -13-1 ), (l-b-1 -1 -1 -1 -2), (l-b-1 -1 -1 -
4-2), (l-b-1 -1-1-4-2a), (l-b-1 -1 -1 -5-2), (l-b-1 -1 -1 -6-2), (l-b-1 -1 -1 -7-2), (l-b-2-1 -1- 1 -1 ), (l-b-2-1 -3-1 -1 ), (l-b-3-1 -1-1-1 ), (l-b-3-1 -2-1-1), (l-b-3-1 -3-1 -1 ), (l-c-1 -1 -1 -2- 1 ), (l-d-1 -1 -1 -1 -0), and (l-d-1 -1 -1 -1 -1 ), as specified herein, may be administered to the lungs in the form of a solution, a suspension or a dry powder, a solution being preferred. Any system developed for the delivery of solutions, suspensions or dry powders via oral inhalation or insufflation are suitable for the administration of the present compounds.
Thus, the present invention also provides a pharmaceutical composition adapted for administration by inhalation or insufflation through the mouth comprising a compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- (l-b-3), (l-c-1 ), (l-d-1 ), (l-e-1 ), (l-f-1 ), (l-a-1 -1 ), (l-b-1 -1 ), (l-b-2-1 ), (l-b-3-1 ), (l-c-1 -1 ), (l-d-1 -1 ), (I- e-1 -1 ), (l-f-1 -1 ), (l-a-1 -1 -1 ), (l-b-1 -1 -1 )- (l-b-1 -1 -3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3- 1 -1 )- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1 -1 -1 ), (l-f-1 -1 -1 ), (l-a-1 -1 -1 -1 )- (l-a-1 -1 -1 -6), (I- b-1-1-1-1 )- (l-b-1-1 -1-13), (l-b-1-1 -2-1 ), (l-b-1 -1-3-1), (l-b-2-1 -1 -1 ), (l-b-2-1 -2-1 - 1 ), (l-b-2-1 -2-1 ), (l-b-2-1 -3-1 ), (l-b-3-1 -1-1), (l-b-3-1 -2-1 ), (l-b-3-1 -3-1 ), (l-c-1 -1 - 1 -1 ), (l-c-1 -1 -1 -2), (l-d-1 -1 -1 -1 ), (l-d-1 -1 -1 -2), (l-f-1 -1 -1 -1 ), (l-a-1 -1 -1 -3-0), (l-a-1 - 1-1 -3-1 ), (l-a-1 -1 -1 -3-2), (l-a-1 -1 -1 -6-2), (l-b-1 -1 -1 -4-0), (l-b-1 -1 -1 -2-1 ), (l-b-1-1 - 1 -4-1 ), (l-b-1 -1 -1 -5-1 ), (l-b-1 -1 -1-7-1), (l-b-1-1-1 -8-1), (l-b-1 -1 -1 -12-1 ), (l-b-1 -1 - 1 -13-1 ), (l-b-1 -1 -1 -1 -2), (l-b-1 -1 -1 -4-2), (l-b-1 -1 -1 -4-2a), (l-b-1 -1 -1 -5-2), (l-b-1 -1 - 1 -6-2), (l-b-1 -1 -1 -7-2), (l-b-2-1 -1-1 -1 ), (l-b-2-1 -3-1 -1 ), (l-b-3-1 -1 -1 -1 ), (l-b-3-1 -2- 1 -1 ), (l-b-3-1 -3-1 -1 ), (l-c-1 -1-1 -2-1), (l-d-1 -1-1 -1-0), and (l-d-1 -1 -1 -1 -1 ), as specified herein, and a pharmaceutically acceptable carrier. Preferably, the compounds of the present invention are administered via inhalation of a solution in nebulized or aerosolized doses.
It is especially advantageous to formulate the aforementioned pharmaceutical compositions in unit dosage form for ease of administration and uniformity of dosage. Unit dosage form as used herein refers to physically discrete units suitable as unitary dosages, each unit containing a predetermined quantity of active ingredient calculated to produce the desired therapeutic effect in association with the required pharmaceutical carrier. Examples of such unit dosage forms are tablets (including scored or coated tablets), capsules, pills, suppositories, powder packets, wafers, injectable solutions or suspensions and the like, and segregated multiples thereof.
The compound (C) of formula (I), as specified herein, or a compound of any of the subgroups of compounds of formula (l-a-1 ), (l-b-1 )- ( l-b-3) , (l-c-1 ), (I- d-1 ), (l-e-1), (l-f-1 ), (l-a-1-1), (l-b-1-1), (l-b-2-1), (l-b-3-1), (l-c-1-1), (l-d-1 -1 ), (I- e-1 -1 ), (l-f-1 -1 ), (l-a-1 -1 -1 ), (l-b-1 -1 -1 )- (l-b-1 -1 -3), (l-b-2-1 -1 )- (l-b-2-1 -3), (l-b-3- 1 -1 )- (l-b-3-1 -3), (l-c-1 -1 -1 ), (l-d-1 -1 -1 ), (l-f-1 -1 -1 ), (l-a-1 -1 -1 -1 )- (l-a-1 -1 -1 -6), (I- b-1-1-1-1 )- (l-b-1-1 -1-13), (l-b-1-1 -2-1 ), (l-b-1 -1-3-1), (l-b-2-1 -1 -1 ), (l-b-2-1 -2-1 - 1 ), (l-b-2-1 -2-1 ), (l-b-2-1 -3-1 ), (l-b-3-1 -1-1), (l-b-3-1 -2-1 ), (l-b-3-1 -3-1 ), (l-c-1 -1 - 1 -1 ), (l-c-1 -1 -1 -2), (l-d-1 -1 -1 -1 ), (l-d-1 -1 -1 -2), (l-f-1 -1 -1 -1 ), (l-a-1 -1 -1 -3-0), (l-a-1 - 1-1 -3-1 ), (l-a-1 -1 -1 -3-2), (l-a-1 -1 -1 -6-2), (l-b-1 -1 -1 -4-0), (l-b-1 -1 -1 -2-1 ), (l-b-1-1 - 1 -4-1 ), (l-b-1 -1 -1 -5-1 ), (l-b-1 -1 -1-7-1), (l-b-1-1-1 -8-1), (l-b-1 -1 -1 -12-1 ), (l-b-1 -1 - 1 -13-1 ), (l-b-1 -1 -1 -1 -2), (l-b-1 -1 -1 -4-2), (l-b-1 -1 -1 -4-2a), (l-b-1 -1 -1 -5-2), (l-b-1 -1 - 1 -6-2), (l-b-1 -1 -1 -7-2), (l-b-2-1 -1-1 -1 ), (l-b-2-1 -3-1 -1 ), (l-b-3-1 -1 -1 -1 ), (l-b-3-1 -2- 1 -1 ), (l-b-3-1 -3-1 -1 ), (l-c-1 -1-1 -2-1), (l-d-1-1 -1 -1-0), and (l-d-1 -1 -1 -1 -1 ), as specified herein, show kinase inhibition properties. Illnesses and diseases treatable using the compounds and methods of the present invention include protein kinase mediated diseases like like cancer, metabolic disorders (such as diabetes), inflammatory and autoimmune disorders (such as inflammatory bowel diseases, e.g. Crohn’s disease and ulcerative colitis, inflammatory pulmonary diseases, rheumatoid arthritis, lupus nephritis, systemic lupus erythematosus and psoriasis and psoriasis arthritis), neurological disorders (such as Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, Charcot-Marie- Tooth neuropathy, amyotrophic lateral sclerosis and epilepsy), atherosclerosis and cardiovascular diseases, Sjogren Syndrome, renal allograft rejection, viral induced diseases, circulatory diseases, bone osteolysis and osteoporosis, osteoarthritis, sarcopenia, Langerhans cell histiocytosis, spinal cord injury, endometriosis, asthma and allergic asthma, eye diseases (such as retinopathies, age-related macular degeneration and uveitis) chronic and neuropathic pain, and fibro-proliferative diseases. Many of the compounds of this invention may show a favourable pharmacokinetic profile and have attractive properties in terms of bioavailability, including an acceptable half-life, AUC (area under the curve) and peak values and lacking unfavourable phenomena such as insufficient quick onset and tissue retention.
The combinations of the present invention may be used as medicaments. Said use as a medicine or method of treatment comprises the systemic
administration to ill subjects of an amount effective to combat the conditions associated with the illnesses. Consequently, the combinations of the present invention can be used in the manufacture of a medicament useful for treating, preventing or combating illness or disease associated with protein kinases including cancer, metabolic disorders (such as diabetes), inflammatory and autoimmune disorders (such as inflammatory bowel diseases, e.g. Crohn’s disease and ulcerative colitis, inflammatory pulmonary diseases, rheumatoid arthritis, lupus nephritis, systemic lupus erythematosus and psoriasis and psoriasis arthritis), neurological disorders (such as Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, Charcot-Marie-Tooth neuropathy, amyotrophic lateral sclerosis and epilepsy), atherosclerosis and cardiovascular diseases, Sjogren Syndrome, renal allograft rejection, viral induced diseases, circulatory diseases, bone osteolysis and osteoporosis, osteoarthritis, sarcopenia, Langerhans cell histiocytosis, spinal cord injury, endometriosis, asthma and allergic asthma, eye diseases (such as retinopathies, age-related macular degeneration and uveitis) chronic and neuropathic pain, and fibro- proliferative diseases.
The term "therapeutically effective amount" as used herein means that amount of active compound or component or pharmaceutical agent that elicits the biological or medicinal response in a tissue, system, animal or human that is being sought, in the light of the present invention, by a researcher, veterinarian, medical doctor or other clinician, which includes alleviation of the symptoms of the disease being treated.
Examples Example 1: General procedure for the preparation of analogues 4-66 Cs2CO3, DMF 100°C Method A Pd(PPh3)4, Cs2CO31M, dioxane, 100°C, 1h Method B 1 2 Vilsmeier reagent, CH2Cl2, rt, 1h30 Method C RNH2, AcOH, MW, 100°C, 20 min Method D 4-66 3 Method A: To a solution of methyl 2-amino-5-hydroxybenzoate (527 mg, 3.15 mmol) in DMF (12 mL) under nitrogen was added solid cesium carbonate (2.569 g, 7.88 mmol) followed by 2-chloro-4-nitropyridine derivative (500 mg, 3.15 mmol). The reaction mixture was stirred at 100°C for 1h. After cooling at room temperature, a saturated aqueous solution of NH4Cl was added and the aqueous layer was extracted with EtOAc. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. The crude was purified by flash column chromatography (cyclohexane/EtOAc from100/0 to 70/30) to give 732 mg of methyl 2-amino-5-[(2-chloro-4-pyridyl)oxy]benzoate 1 as a white powder in 83% yield. ESI-MS: 278.90 (M+H)+. Method B: To a stirred solution of intermediate 1 (400 mg, 1.44 mmol) in dioxane (7 mL) were added under nitrogen 1-methyl-4-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-1H-pyrazole (448 mg, 2.15 mmol), Cs2CO31M (4.306 mL, 4.31 mmol) and Pd(PPh3)4 (166 mg, 0.14 mmol). The reaction mixture was stirred at 100°C for 1h. The solvent was removed under vacuum, and the crude was purified by flash column chromatography (CH2Cl2/MeOH from 100/0 to 95/5), to give 466 mg of methyl 2-amino-5-{[2-(1-methylpyrazol-4-yl)-4-
pyridyl]oxy}benzoate 2 as a beige powder in quantitative yield. ESI-MS: 324.95 (M+H)+. Method C: To a solution of intermediate 2 (466 mg, 1.44 mmol) in DCM (7 mL) under nitrogen was added Vilsmeier reagent (276 mg, 2.15 mmol). The reaction mixture was stirred at room temperature for 2h. The reaction mixture was diluted with DCM and washed with a saturated solution of NaHCO3. Organic layers were dried over Na2SO4, filtered and concentrated under reduced pressure. Intermediate 3 was obtained as an orange oil in quantitative yield and directly used in the next step without purification. ESI-MS: 380.05 (M+H)+. Method D: Compound 3 (30 mg, 0.08 mmol) and appropriate amine derivative (1.5-10 equiv.) were dissolved in acetic acid (0.5 mL) in an oven-dried screw- cap test tube. The reaction mixture was stirred under microwave irradiation at 100°C for 20 minutes or under conventional heating at 100°C for 1h. The solvent was removed under vacuum, and the crude was purified by flash column chromatography (CH2Cl2/MeOH from 100/0 to 95/5) and reverse phase chromatography (H2O/MeOH from 100/0 to 0/100) to give the expected compound. The following compounds are examples illustrating Method D: 3-cyclohexyl-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}quinazolin-4-one (4): Compound 4 was synthesized from intermediate 3 (0.08 mmol) and cyclohexylamine (0.16 mmol) as a white solid in 19% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.49 (s, 1H), 8.42 (d, J = 5.6, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.80 (d, J = 9.0, 1H), 7.76 (d, J = 2.5, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.31 (d, J = 2.1, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 4.66-4.58 (m, 1H), 3.85 (s, 3H), 1.90-1.80 (m, 6H), 1.70-1.66 (m, 1H), 1.47-1.18 (m, 3H). ESI-MS: 402.10 (M+H)+.
3-(3-fluorophenyl)-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}quinazolin-4-one (5): Compound 5 was synthesized from intermediate 3 (0.08 mmol) and 3- fluoroaniline (0.16 mmol) as a white solid in 11% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.44 (d, J = 5.6, 1H), 8.40 (s, 1H), 8.28 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.88 (d, J = 8.8, 1H), 7.81 (d, J = 2.5, 1H), 7.76 (dd, J = 8.8, 2.9, 1H), 7.68-7.59 (m, 1H), 7.56- 7.52 (m, 1H), 7.44-7.37 (m, 2H), 7.35 (d, J = 2.1, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 3.86 (s, 3H). ESI-MS: 414.05 (M+H)+. 3-(4-fluorophenyl)-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}quinazolin-4-one (6): Compound 6 was synthesized from intermediate 3 (0.08 mmol) and 4- fluoroaniline (0.16 mmol) as a white solid in 15% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.44 (d, J = 5.6, 1H), 8.37 (s, 1H), 8.28 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.88 (d, J = 8.9, 1H), 7.81 (d, J = 2.5, 1H), 7.75 (dd, J = 8.8, 2.9, 1H), 7.64-7.60 (m, 2H), 7.45-7.39 (m, 2H), 7.34 (d, J = 2.0, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 3.86 (s, 3H). ESI-MS: 414.00 (M+H)+. 3-benzyl-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}quinazolin-4-one (7): Compound 7 was synthesized from intermediate 3 (0.09 mmol) and benzylamine (0.19 mmol) as a white solid in 37% yield according to the general method D.1H
NMR (400 MHz, DMSO-d6) δ (ppm): 8.61 (s, 1H), 8.41 (d, J = 5.6, 1H), 8.26 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.82 (d, J = 8.9, 1H), 7.76 (d, J = 2.5, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.40-7.27 (m, 6H), 6.78 (dd, J = 5.7, 2.4, 1H), 5.21 (s, 2H), 3.85 (s, 3H). ESI-MS: 410.00 (M+H)+. 3-[(3-fluorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}quinazolin- 4-one (8): Compound 8 was synthesized from intermediate 3 (0.08 mmol) and 3- fluorobenzylamine (0.12 mmol) as a white solid in 41% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.61 (s, 1H), 8.42 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.82 (d, J = 8.8, 1H), 7.76 (d, J = 2.8, 1H), 7.69 (dd, J = 8.8, 2.8, 1H), 7.43-7.37 (m, 1H), 7.32 (d, J = 2.3, 1H), 7.26- 7.20 (m, 2H), 7.14 (td, J = 8.6, 2.2, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 5.21 (s, 2H), 3.85 (s, 3H). ESI-MS: 428.10 (M+H)+. 3-[(4-fluorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}quinazolin- 4-one (9): Compound 9 was synthesized from intermediate 3 (0.08 mmol) and 4- fluorobenzylamine (0.12 mmol) as a white solid in 44% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.62 (s, 1H), 8.42 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.6, 1H), 7.81 (d, J = 8.8, 1H), 7.75 (d, J = 2.7, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.48-7.44 (m, 2H), 7.32 (d, J = 2.3, 1H), 7.21-7.16 (m, 2H), 6.78 (dd, J = 5.7, 2.4, 1H), 5.18 (s, 2H), 3.85 (s, 3H). ESI-MS: 428.10 (M+H)+.
3-[(3-chlorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (10): Compound 10 was synthesized from intermediate 3 (0.08 mmol) and 3- chlorobenzylamine (0.12 mmol) as a white solid in 34% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.63 (s, 1H), 8.42 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.82 (d, J = 8.8, 1H), 7.76 (d, J = 2.7, 1H), 7.69 (dd, J = 8.8, 2.8, 1H), 7.49 (s, 1H), 7.40-7.31 (m, 4H), 6.78 (dd, J = 5.7, 2.4, 1H), 5.20 (s, 2H), 3.85 (s, 3H). ESI-MS: 444.05 (M+H)+. 3-[(4-chlorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (11): Compound 11 was synthesized from intermediate 3 (0.08 mmol) and 4- chlorobenzylamine (0.12 mmol) as a white solid in 44% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.61 (s, 1H), 8.42 (d, J = 5.6, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.6, 1H), 7.82 (d, J = 8.8, 1H), 7.75 (d, J = 2.6, 1H), 7.69 (dd, J = 8.8, 2.9, 1H), 7.42 (s, 4H), 7.32 (d, J = 2.2, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 5.19 (s, 2H), 3.85 (s, 3H). ESI-MS: 444.05 (M+H)+.
3-[(3,5-difluorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (12): Compound 12 was synthesized from intermediate 3 (0.08 mmol) and 3,5- difluorobenzylamine (0.12 mmol) as a white solid in 40% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.59 (s, 1H), 8.42 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.83 (d, J = 8.8, 1H), 7.76 (d, J = 2.6, 1H), 7.70 (dd, J = 8.8, 2.9, 1H), 7.32 (d, J = 2.1, 1H), 7.22-7.10 (m, 3H), 6.78 (dd, J = 5.7, 2.4, 1H), 5.21 (s, 2H), 3.85 (s, 3H). ESI-MS: 446.00 (M+H)+. 3-[(3,4-difluorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (13): Compound 13 was synthesized from intermediate 3 (0.09 mmol) and 3,4- difluorobenzylamine (0.19 mmol) as a white solid in 59% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.61 (s, 1H), 8.42 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.82 (d, J = 8.8, 1H), 7.75 (d, J = 2.6, 1H), 7.69 (dd, J = 8.8, 2.9, 1H), 7.55-7.49 (m, 1H), 7.45-7.38 (m, 1H), 7.32 (d, J = 2.1, 1H), 7.27-7.23 (m, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 5.18 (s, 2H), 3.85 (s, 3H). ESI-MS: 446.05 (M+H)+.
3-[(4-chloro-3-fluorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (14): Compound 14 was synthesized from intermediate 3 (0.08 mmol) and 4-chloro- 3-fluorobenzylamine (0.12 mmol) as a white solid in 50% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.60 (s, 1H), 8.42 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.83 (d, J = 8.8, 1H), 7.75 (d, J = 2.5, 1H), 7.69 (dd, J = 8.8, 2.9, 1H), 7.58 (t, J = 8.1, 1H), 7.48 (dd, J = 10.3, 2.0, 1H), 7.32 (d, J = 2.1, 1H), 7.25 (dd, J = 8.3, 1.5, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 5.20 (s, 2H), 3.85 (s, 3H). ESI-MS: 462.00 (M+H)+. 3-[(2-methoxyphenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (15): Compound 15 was synthesized from intermediate 3 (0.08 mmol) and 2- methoxybenzylamine (0.12 mmol) as a white solid in 43% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.47 (s, 1H), 8.42 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.7, 1H), 7.81 (d, J = 8.8, 1H), 7.73 (d, J = 2.5, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.32-7.27 (m, 2H), 7.16 (dd, J = 7.5, 1.6, 1H), 7.03 (d, J = 8.4, 1H), 6.90 (td, J = 7.5, 1.0, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 5.12 (s, 2H), 3.85 (s, 3H), 3.84 (s, 3H). ESI-MS: 440.05 (M+H)+.
6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-{[3- (trifluoromethyl)phenyl]methyl}quinazolin-4-one (16): Compound 16 was synthesized from intermediate 3 (0.08 mmol) and 3- (trifluoromethyl)benzylamine (0.12 mmol) as a white solid in 63% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.67 (s, 2H), 8.50 (dd, J = 4.8, 1.6, 1H), 8.42 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.7, 1H), 7.83-7.79 (m, 2H), 7.75 (d, J = 2.5, 1H), 7.69 (dd, J = 8.8, 2.9, 1H), 7.38 (ddd, J = 7.9, 4.8, 0.8, 1H), 7.31 (d, J = 2.1, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 5.23 (s, 2H), 3.85 (s, 3H). ESI-MS: 478.00 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-{[4- (trifluoromethyl)phenyl]methyl}quinazolin-4-one (17): Compound 17 was synthesized from intermediate 3 (0.08 mmol) and 4- (trifluoromethyl)benzylamine (0.12 mmol) as a white solid in 42% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.64 (s, 1H), 8.42 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.6, 1H), 7.84 (d, J = 8.8, 1H), 7.75-7.68 (m, 4H), 7.59 (d, J = 8.1, 2H), 7.32 (d, J = 2.3, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 5.30 (s, 2H), 3.85 (s, 3H). ESI-MS: 478.05 (M+H)+.
6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-(3-pyridylmethyl)quinazolin-4-one (18): Compound 18 was synthesized from intermediate 3 (0.08 mmol) and 3- (aminomethyl)pyridine (0.12 mmol) as a white solid in 63% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.68 (s, 1H), 8.42 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.7, 1H), 7.84-7.81 (m, 2H), 7.76 (d, J = 2.6, 1H), 7.71-7.66 (m, 3H), 7.62-7.57 (m, 1H), 7.32 (d, J = 2.1, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 5.29 (s, 2H), 3.85 (s, 3H). ESI-MS: 411.00 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-(4-pyridylmethyl)quinazolin-4-one (19): Compound 19 was synthesized from intermediate 3 (0.08 mmol) and 4- (aminomethyl)pyridine (0.12 mmol) as a white solid in 41% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.59 (s, 1H), 8.54- 8.52 (m, 2H), 8.42 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.85 (d, J = 9.0, 1H), 7.75 (d, J = 2.5, 1H), 7.71 (dd, J = 8.8, 2.9, 1H), 7.34-7.29 (m, 3H), 6.78 (dd, J = 5.7, 2.4, 1H), 5.24 (s, 2H), 3.85 (s, 3H). ESI-MS: 411.05 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-{[6-(trifluoromethyl)-3- pyridyl]methyl}quinazolin-4-one (20): Compound 20 was synthesized from intermediate 3 (0.08 mmol) and [6-
(trifluoromethyl)-3-pyridyl]methanamine (0.12 mmol) as a white solid in 45% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.85 (d, J = 1.5, 1H), 8.68 (s, 1H), 8.41 (d, J = 5.7, 1H), 8.26 (s, 1H), 8.08 (dd, J = 8.1, 1.7, 1H), 7.97 (s, 1H), 7.89 (d, J = 8.1, 1H), 7.83 (d, J = 8.8, 1H), 7.74 (d, J = 2.8, 1H), 7.70 (dd, J = 8.8, 2.8, 1H), 7.32 (d, J = 2.4, 1H), 6.76 (dd, J = 5.7, 2.4, 1H), 5.34 (s, 2H), 3.85 (s, 3H). ESI-MS: 479.05 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-{[2-(trifluoromethyl)-4- pyridyl]methyl}quinazolin-4-one (21): Compound 21 was synthesized from intermediate 3 (0.08 mmol) and [2- (trifluoromethyl)-4-pyridyl]methanamine (0.12 mmol) as a white solid in 13% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.72 (d, J = 5.0, 1H), 8.62 (s, 1H), 8.42 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98-7.94 (m, 2H), 7.85 (d, J = 8.7, 1H), 7.75-7.69 (m, 2H), 7.61 (d, J = 4.9, 1H), 7.33 (d, J = 2.3, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 5.34 (s, 2H), 3.85 (s, 3H). ESI-MS: 479.05 (M+H)+. 3-{[6-(methylamino)-3-pyridyl]methyl}-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (22): Compound 22 was synthesized from intermediate 3 (0.08 mmol) and 5- (aminomethyl)-N-methyl-pyridin-2-amine (0.12 mmol) as a white solid in 32% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.60 (s, 1H), 8.41 (d, J = 5.7, 1H), 8.26 (s, 1H), 8.11 (d, J = 2.2, 1H), 7.98 (d, J = 0.5, 1H), 7.79 (d, J = 8.8, 1H), 7.75 (d, J = 2.8, 1H), 7.66 (dd, J = 8.8, 2.9, 1H), 7.47 (dd, J = 8.6, 2.4, 1H), 7.31 (d, J = 2.3, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 6.53 (q, J = 4.7, 1H), 6.39 (d, J = 8.5, 1H), 5.00 (s, 2H), 3.85 (s, 3H), 2.72 (d, J = 4.8, 3H). ESI-MS: 440.10 (M+H)+.
5-(aminomethyl)-N-methyl-pyridin-2-amine was prepared by the following procedure: To a mixture 6-(methylamino)nicotinonitrile (251 mg, 1.8 mmol) in ammonia (solution 7N in MeOH, 15 mL) was added Raney nickel (slurry in water, 1g). After stirring overnight under a hydrogen atmosphere, the reaction mixture was filtered through a pad of Celite, rinsed with MeOH, and concentrated under reduced pressure to provide the expected compound which was used without further purification. ESI-MS: 138.20 (M+H)+. N-methyl-3-[(6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-4-oxo-quinazolin-3- yl)methyl]benzene sulfonamide (23): Compound 23 was synthesized from intermediate 3 (0.08 mmol) and 3- (aminomethyl)-N-methyl-benzenesulfonamide (0.12 mmol) as a white solid in 10% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.66 (s, 1H), 8.42 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.6, 1H), 7.83 (d, J = 8.8, 1H), 7.79 (s, 1H), 7.76 (d, J = 2.7, 1H), 7.71-7.68 (m, 2H), 7.66-7.56 (m, 2H), 7.46 (bs, 1H), 7.32 (d, J = 2.3, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 5.30 (s, 2H), 3.85 (s, 3H), 2.39 (s, 3H). ESI-MS: 503.10 (M+H)+. N-methyl-4-([6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-4-oxo-quinazolin-3- yl)methyl]benzene sulfonamide (24): Compound 24 was synthesized from intermediate 3 (0.08 mmol) and 4- (aminomethyl)-N-methyl-benzenesulfonamide (0.12 mmol) as a white solid in 23% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ
(ppm): 8.63 (s, 1H), 8.42 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.84 (d, J = 8.8, 1H), 7.76-7.74 (m, 3H), 7.70 (dd, J = 8.8, 2.8, 1H), 7.57 (d, J = 8.4, 2H), 7.45 (bs, 1H), 7.32 (d, J = 2.3, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 5.29 (s, 2H), 3.85 (s, 3H), 2.38 (s, 3H). ESI-MS: 503.10 (M+H)+. 3-(cyclohexylmethyl)-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}quinazolin-4- one (25): Compound 25 was synthesized from intermediate 3 (0.08 mmol) and 1- cyclohexyl methanamine (0.12 mmol) as a white solid in 21% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.42 (d, J = 5.6, 1H), 8.36 (s, 1H), 8.28 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.80 (d, J = 8.9, 1H), 7.75 (d, J = 2.5, 1H), 7.67 (dd, J = 8.8, 2.9, 1H), 7.33 (d, J = 2.1, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 3.6-3.83 (m, 5H), 1.84-1.56 (m, 6H), 1.23-0.94 (m, 5H). ESI-MS: 416.10 (M+H)+. 3-[(1R)-1-cyclohexylethyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}quinazolin- 4-one (26): Compound 26 was synthesized from intermediate 3 (0.08 mmol) and (R)-1- cyclohexylethanamine (0.12 mmol) as a white solid in 42% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43-8.40 (m, 2H), 8.28 (s, 1H), 7.99 (d, J = 0.7, 1H), 7.80 (d, J = 9.0, 1H), 7.75 (d, J = 2.6, 1H), 7.67 (dd, J = 8.8, 2.9, 1H), 7.34 (d, J = 2.1, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 4.62- 4.54 (m, 1H), 3.86 (s, 3H), 1.89-1.58 (m, 5H), 1.43 (d, J = 7.0, 3H), 1.30-0.83 (m, 6H). ESI-MS: 430.10 (M+H)+.
3-[(1S)-1-cyclohexylethyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}quinazolin- 4-one (27): Compound 27 was synthesized from intermediate 3 (0.08 mmol) and (S)-1- cyclohexylethanamine (0.12 mmol) as a white solid in 15% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43-8.41 (m, 2H), 8.28 (s, 1H), 7.99 (s, 1H), 7.80 (d, J = 8.8, 1H), 7.75 (d, J = 2.8, 1H), 7.67 (dd, J = 8.8, 2.8, 1H), 7.34 (d, J = 2.3, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 4.63-4.52 (m, 1H), 3.86 (s, 3H), 1.88-1.58 (m, 5H), 1.43 (d, J = 7.0, 3H), 1.33-0.80 (m, 6H). ESI-MS: 430.10 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-{[4- (trifluoromethyl)cyclohexyl]methyl}quinazolin-4-one (28): Compound 28 was synthesized from intermediate 3 (0.08 mmol) and [4- (trifluoromethyl)cyclohexyl]methanamine (0.16 mmol) as a white solid in 44% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.6, 1H), 8.37 (s, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.80 (d, J = 8.9, 1H), 7.75 (d, J = 2.6, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.33 (d, J = 2.1, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 3.88-3.85 (m, 5H), 2.27-2.18 (m, 1H), 1.88-1.69 (m, 5H), 1.25-1.06 (m, 4H). ESI-MS: 484.10 (M+H)+.
6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-(tetrahydropyran-4- ylmethyl)quinazolin-4-one (29): Compound 29 was synthesized from intermediate 3 (0.08 mmol) and 4- (aminomethyl)tetrahydropyran (0.16 mmol) as a white solid in 42% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.42 (d, J = 5.7, 1H), 8.38 (s, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.6, 1H), 7.80 (d, J = 8.8, 1H), 7.75 (d, J = 2.7, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.33 (d, J = 2.2, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 3.90-3.81 (m, 7H), 3.23 (td, J = 11.6, 1.7, 2H), 2.08-2.02 (m, 1H), 1.47 (dd, J = 12.6, 1.8, 2H), 1.29 (ddd, J = 16.2, 12.3, 4.4, 2H). ESI-MS: 418.05 (M+H)+. 3-[(1-methyl-4-piperidyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (30): Compound 30 was synthesized from intermediate 3 (0.09 mmol) and (1-methyl- 4-piperidinyl)methanamine (0.19 mmol) as a white solid in 25% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.7, 1H), 8.38 (s, 1H), 8.28 (s, 1H), 7.99 (d, J = 0.6, 1H), 7.80 (d, J = 8.8, 1H), 7.76 (d, J = 2.7, 1H), 7.68 (dd, J = 8.8, 2.8, 1H), 7.33 (d, J = 2.3, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 3.89-3.86 (m, 5H), 2.75 (d, J = 11.4, 2H), 2.13 (s, 3H), 1.80- 1.71 (m, 3H), 1.51 (d, J = 11.2, 2H), 1.32-1.22 (m, 2H). ESI-MS: 431.10 (M+H)+.
3-isobutyl-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}quinazolin-4-one (31): Compound 31 was synthesized from intermediate 3 (0.08 mmol) and isobutylamine (0.79 mmol) as a white solid in 47% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.42 (d, J = 5.7, 1H), 8.38 (s, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.80 (d, J = 8.8, 1H), 7.76 (d, J = 2.8, 1H), 7.68 (dd, J = 8.8, 2.8, 1H), 7.33 (d, J = 2.3, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 3.86 (s, 3H), 3.82 (d, J = 7.3, 2H), 2.16-2.05 (m, 1H), 0.90 (d, J = 6.7, 6H). ESI-MS: 376.00 (M+H)+. 3-[2-(dimethylamino)ethyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (32): Compound 32 was synthesized from intermediate 3 (0.08 mmol) and N,N- dimethylethylenediamine (0.16 mmol) as a white solid in 64% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.6, 1H), 8.33 (s, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.79 (d, J = 8.9, 1H), 7.75 (d, J = 2.6, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.33 (d, J = 2.1, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 4.07 (t, J = 6.1, 2H), 3.86 (s, 3H), 2.55 (t, J = 6.1, 2H), 2.19 (s, 6H). ESI-MS: 391.00 (M+H)+. 3-indan-1-yl-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}quinazolin-4-one (33): Compound 33 was synthesized from intermediate 3 (0.08 mmol) and 2,3- dihydro-1H-inden-1-amine (0.12 mmol) as a white solid in 35% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J =
5.6, 1H), 8.28 (s, 1H), 8.00-7.98 (m, 2H), 7.81-7.78 (m, 2H), 7.70 (dd, J = 8.9, 2.7, 1H), 7.39 (d, J = 7.6, 1H), 7.34-7.30 (m, 2H), 7.23 (t, J = 7.3, 1H), 7.17 (d, J = 7.5, 1H), 6.79 (dd, J = 5.7, 2.4, 1H), 6.32-6.28 (m, 1H), 3.86 (s, 3H), 3.24- 3.16 (m, 1H), 3.04-2.96 (m, 1H), 2.71-2.62 (m, 1H), 2.30-2.20 (m, 1H). ESI-MS: 436.05 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-(2-phenylethyl)quinazolin-4-one (34): Compound 34 was synthesized from intermediate 3 (0.08 mmol) and 2- phenylethylamine (0.12 mmol) as a white solid in 43% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.7, 1H), 8.28 (s, 1H), 8.19 (s, 1H), 7.99 (d, J = 0.6, 1H), 7.78 (d, J = 2.1, 1H), 7.76 (d, J = 3.6, 1H), 7.67 (dd, J = 8.9, 2.8, 1H), 7.34-7.20 (m, 6H), 6.79 (dd, J = 5.7, 2.4, 1H), 4.22 (t, J = 7.4, 2H), 3.86 (s, 3H), 3.02 (t, J = 7.4, 2H). ESI-MS: 424.05 (M+H)+. 3-[2-(3-fluorophenyl)ethyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}quinazolin- 4-one (35): Compound 35 was synthesized from intermediate 3 (0.08 mmol) and 2-(3- fluorophenyl)ethylamine (0.16 mmol) as a white solid in 31% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.6, 1H), 8.27 (s, 1H), 8.22 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.79-7.76 (m, 2H), 7.67 (dd, J = 8.5, 3.1, 1H), 7.36-7.30 (m, 2H), 7.15-7.11 (m, 1H), 7.08-7.03 (m, 2H), 6.78 (dd, J = 5.7, 2.4, 1H), 4.23 (t, J = 7.3, 2H), 3.86 (s, 3H), 3.05 (t, J = 7.3, 2H). ESI-MS: 442.05 (M+H)+.
3-[2-(4-fluorophenyl)ethyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}quinazolin- 4-one (36): Compound 36 was synthesized from intermediate 3 (0.08 mmol) and 4- fluorophenethylamine (0.16 mmol) as a white solid in 40% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.6, 1H), 8.28 (s, 1H), 8.20 (s, 1H), 7.99 (d, J = 0.7, 1H), 7.79-7.76 (m, 2H), 7.67 (dd, J = 8.6, 3.1, 1H), 7.34 (d, J = 2.0, 1H), 7.29-7.25 (m, 2H), 7.15-7.10 (m, 2H), 6.78 (dd, J = 5.7, 2.4, 1H), 4.20 (t, J = 7.3, 2H) 3.86 (s, 3H), 3.01 (t, J = 7.3, 2H). ESI-MS: 442.05 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-(2-morpholinoethyl)quinazolin-4- one (37): Compound 37 was synthesized from intermediate 3 (0.08 mmol) and 4-(2- aminoethyl)morpholine (0.16 mmol) as a white solid in 58% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.7, 1H), 8.34 (s, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.6, 1H), 7.80 (d, J = 8.8, 1H), 7.75 (d, J = 2.7, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.33 (d, J = 2.3, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 4.09 (t, J = 6.0, 2H), 3.86 (s, 3H), 3.54-3.51 (m, 4H), 2.60 (t, J = 6.0, 2H), 2.45-2.41 (m, 4H). ESI-MS: 433.10 (M+H)+.
3-[(1-methylpyrazol-4-yl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (38): Compound 38 was synthesized from intermediate 3 (0.08 mmol) and (1- methylpyrazol-4-yl)methanamine (0.16 mmol) as a white solid in 33% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.55 (s, 1H), 8.43 (d, J = 5.6, 1H), 8.26 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.79 (d, J = 8.9, 1H), 7.76-7.74 (m, 2H), 7.67 (dd, J = 8.8, 2.9, 1H), 7.48 (d, J = 0.6, 1H), 7.31 (d, J = 2.1, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 5.02 (s, 2H), 3.85 (s, 3H), 3.77 (s, 3H). ESI-MS: 414.05 (M+H)+. 3-[(1-methylpyrazol-3-yl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (39): Compound 39 was synthesized from intermediate 3 (0.08 mmol) and (1- methylpyrazol-3-yl)methanamine (0.16 mmol) as a white solid in 51% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.47 (s, 1H), 8.42 (d, J = 5.6, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.80 (d, J = 8.9, 1H), 7.75 (d, J = 2.6, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.62 (d, J = 2.1, 1H), 7.32 (d, J = 2.1, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 6.21 (d, J = 2.2, 1H), 5.15 (s, 2H), 3.85 (s, 3H), 3.77 (s, 3H). ESI-MS: 414.05 (M+H)+. 3-[(2-methylpyrazol-3-yl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (40): Compound 40 was synthesized from intermediate 3 (0.08 mmol) and (2-
methylpyrazol-3-yl)methanamine (0.12 mmol) as a white solid in 36% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.55 (s, 1H), 8.42 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.82 (d, J = 8.8, 1H), 7.77 (d, J = 2.8, 1H), 7.70 (dd, J = 8.8, 2.8, 1H), 7.32 (t, J = 2.2, 2H), 6.78 (dd, J = 5.7, 2.4, 1H), 6.22 (d, J = 1.8, 1H), 5.28 (s, 2H), 3.92 (s, 3H), 3.85 (s, 3H). ESI-MS: 414.05 (M+H)+. 3-[(1-isopropylimidazol-4-yl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (41): Compound 41 was synthesized from intermediate 3 (0.08 mmol) and (1- isopropylimidazol-4-yl)methanamine (0.12 mmol) as a white solid in 23% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.49 (s, 1H), 8.41 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.80 (d, J = 8.8, 1H), 7.75 (d, J = 2.8, 1H), 7.67 (dd, J = 8.8, 2.8, 1H), 7.64 (d, J = 1.2, 1H), 7.31 (d, J = 2.3, 1H), 7.26 (s, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 5.05 (s, 2H), 4.35 (hept, J = 6.7, 1H), 3.85 (s, 3H), 1.36 (d, J = 6.7, 6H). ESI-MS: 442.10 (M+H)+. (1-isopropylimidazol-4-yl)methanamine was prepared by hydrogenation of 1- isopropyl-1H-imidazole-4-carbonitrile (WO 2009/070485) according to example 22. ESI-MS: 410.35 (M+H)+. 3-[(3,5-dimethylisoxazol-4-yl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (42): Compound 42 was synthesized from intermediate 3 (0.08 mmol) and (3,5- dimethylisoxazol-4-yl)methanamine (0.12 mmol) as a white solid in 30% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.58
(s, 1H), 8.41 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.81 (d, J = 8.8, 1H), 7.73 (d, J = 2.8, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.32 (d, J = 2.3, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 4.98 (s, 2H), 3.85 (s, 3H), 2.46 (s, 3H), 2.23 (s, 3H). ESI-MS: 429.05 (M+H)+. 3-[(1-methyl-2-oxo-3-pyridyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (43): Compound 43 was synthesized from intermediate 3 (0.08 mmol) and 3- (aminomethyl)-1-methyl-pyridin-2-one (0.12 mmol) as a white solid in 29% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.55 (s, 1H), 8.41 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.5, 1H), 7.79 (d, J = 8.8, 1H), 7.72 (d, J = 2.7, 1H), 7.70-7.65 (m, 2H), 7.44 (dd, J = 6.9, 1.9, 1H), 7.31 (d, J = 2.3, 1H), 6.76 (dd, J = 5.7, 2.4, 1H), 6.22 (t, J = 6.8, 1H), 4.93 (s, 2H), 3.85 (s, 3H), 3.44 (s, 3H). ESI-MS: 441.05 (M+H)+. 3-[(1-methyl-2-oxo-3-piperidyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (44): Compound 44 was synthesized from intermediate 3 (0.08 mmol) and 3- (aminomethyl)-1-methyl-piperidin-2-one (0.12 mmol) as a white solid in 34% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.42 (d, J = 5.7, 1H), 8.34 (s, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.79 (d, J = 8.8, 1H), 7.75 (d, J = 2.8, 1H), 7.68 (dd, J = 8.8, 2.8, 1H), 7.33 (d, J = 2.3, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 4.42 (dd, J = 13.5, 6.4, 1H), 3.97 (dd, J = 13.5, 7.8, 1H), 3.86 (s, 3H), 3.27-3.23 (m, 2H), 2.81-2.75 (m, 4H), 1.89-1.80 (m, 1H), 1.76-1.62 (m, 2H), 1.56-1.46 (m, 1H). ESI-MS: 445.05 (M+H)+.
-3-[(1-methylindazol-6-yl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (45): Compound 45 was synthesized from intermediate 3 (0.08 mmol) and (1- methylindazol-6-yl)methanamine (0.12 mmol) as a white solid in 44% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.65 (s, 1H), 8.41 (d, J = 5.7, 1H), 8.26 (s, 1H), 8.02 (d, J = 0.8, 1H), 7.97 (s, 1H), 7.82 (d, J = 8.8, 1H), 7.77 (d, J = 2.8, 1H), 7.73 (d, J = 8.4, 1H), 7.70-7.67 (m, 2H), 7.32 (d, J = 2.3, 1H), 7.17 (dd, J = 8.4, 1.2, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 5.34 (s, 2H), 4.02 (s, 3H), 3.85 (s, 3H). ESI-MS: 464.10 (M+H)+. 3-[(1-methylindazol-5-yl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (46): Compound 46 was synthesized from intermediate 3 (0.08 mmol) and (1- methylindazol-5-yl)methanamine (0.12 mmol) as a white solid in 49% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.66 (s, 1H), 8.41 (d, J = 5.7, 1H), 8.26 (s, 1H), 8.03 (d, J = 0.7, 1H), 7.97 (s, 1H), 7.82-7.79 (m, 2H), 7.76 (d, J = 2.8, 1H), 7.67 (dd, J = 8.8, 2.9, 1H), 7.62 (d, J = 8.7, 1H), 7.49 (dd, J = 8.7, 1.5, 1H), 7.31 (d, J = 2.3, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 5.30 (s, 2H), 4.01 (s, 3H), 3.85 (s, 3H). ESI-MS: 464.10 (M+H)+.
3-(1,3-benzodioxol-5-ylmethyl)-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (47): Compound 47 was synthesized from intermediate 3 (0.09 mmol) and 3,4- (methylenedioxy)benzylamine (0.14 mmol) as a white solid in 31% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.58 (s, 1H), 8.41 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.80 (d, J = 8.9, 1H), 7.76 (d, J = 2.6, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.31 (d, J = 2.1, 1H), 7.02 (d, J = 1.4, 1H), 6.93-6.86 (m, 2H), 6.78 (dd, J = 5.7, 2.4, 1H), 5.99 (s, 2H), 5.09 (s, 2H), 3.85 (s, 3H). ESI-MS: 454.05 (M+H)+. 3-[2-(1,3-benzodioxol-5-yl)ethyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (48): Compound 48 was synthesized from intermediate 3 (0.11 mmol) and 3,4- methylenedioxyphenethylamine (0.16 mmol) as a white solid in 29% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.9, 1H), 8.28 (s, 1H), 8.18 (s, 1H), 7.99 (d, J = 0.7, 1H), 7.78-7.63 (m, 2H), 7.67 (dd, J = 9.0, 2.7, 1H), 7.34 (d, J = 2.1, 1H), 6.87 (d, J = 1.6, 1H), 6.82- 6.77 (m, 2H), 6.64 (dd, J = 7.9, 1.6, 1H), 5.98 (s, 2H), 4.17 (t, J = 7.3, 2H), 3.86 (s, 3H), 2.93 (t, J = 7.2, 2H). ESI-MS: 468.10 (M+H)+.
6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-[2-(2-pyridyl)ethyl]quinazolin-4-one (49): Compound 49 was synthesized from intermediate 3 (0.11 mmol) and 2-(2- pyridyl)ethylamine (0.16 mmol) as a white solid in 45% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.50 (ddd, J = 4.8, 1.8, 0.9, 1H), 8.43 (d, J = 5.7, 1H), 8.28 (s, 1H), 8.21 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.77-7.75 (m, 2H), 7.71 (td, J = 7.7, 1.9, 1H), 7.67 (dd, J = 8.9, 2.8, 1H), 7.33 (d, J = 2.2, 1H), 7.29 (d, J = 7.8, 1H), 7.24 (ddd, J = 7.5, 4.9, 1.1, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 4.38 (t, J = 7.1, 2H), 3.86 (s, 3H), 3.20 (t, J = 7.1, 2H). ESI-MS: 425.05 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-[2-(3-pyridyl)ethyl]quinazolin-4-one (50): Compound 50 was synthesized from intermediate 3 (0.11 mmol) and 2-(pyridin- 3-yl)ethanamine (0.16 mmol) as a white solid in 60% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.44-8.42 (m, 3H), 8.28 (s, 1H), 8.23 (s, 1H), 7.99 (d, J = 0.5, 1H), 7.79-7.76 (m, 2H), 7.69-7.66 (m, 2H), 7.35-7.31 (m, 2H), 6.79 (dd, J = 5.7, 2.4, 1H), 4.25 (t, J = 7.3, 2H), 3.86 (s, 3H), 3.06 (t, J = 7.3, 2H). ESI-MS: 425.05 (M+H)+.
6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-[2-(4-pyridyl)ethyl]quinazolin-4-one (51): Compound 51 was synthesized from intermediate 3 (0.11 mmol) and (2-pyridin- 4-ylethyl)amine dihydrochloride (0.16 mmol) as a white solid in 31% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.49- 8.47 (m, 2H), 8.43 (d, J = 5.7, 1H), 8.28 (s, 1H), 8.26 (s, 1H), 7.99 (d, J = 0.7, 1H), 7.79-7.75 (m, 2H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.34 (d, J = 2.1, 1H), 7.29- 7.27 (m, 2H), 6.78 (dd, J = 5.7, 2.4, 1H), 4.26 (t, J = 7.3, 2H), 3.86 (s, 3H), 3.06 (t, J = 7.3, 2H). ESI-MS: 425.05 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-phenacyl-quinazolin-4-one (52): Compound 52 was synthesized from intermediate 3 (0.11 mmol) and 2- aminoacetophenone hydrochloride (0.16 mmol) as a white solid in 13% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.7, 1H), 8.37 (s, 1H), 8.28 (s, 1H), 8.12-8.09 (m, 2H), 7.99 (d, J = 0.7, 1H), 7.87 (dd, J = 8.7, 0.5, 1H), 7.78-7.72 (m, 3H), 7.65-7.60 (m, 2H), 7.35 (d, J = 2.1, 1H), 6.81 (dd, J = 5.7, 2.4, 1H), 5.66 (s, 2H), 3.86 (s, 3H). ESI-MS: 438.05 (M+H)+. 3-(2-ethylbutyl)-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}quinazolin-4-one (53): Compound 53 was synthesized from intermediate 3 (0.11 mmol) and 2-ethyl-1- butylamine (0.16 mmol) as a white solid in 13% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.7, 1H), 8.38 (s,
1H), 8.28 (s, 1H), 7.99 (d, J = 0.7, 1H), 7.80 (d, J = 8.9, 1H), 7.75 (d, J = 2.6, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.34 (d, J = 2.1, 1H), 6.79 (dd, J = 5.7, 2.4, 1H), 3.89 (d, J = 7.3, 2H), 3.86 (s, 3H), 1.82-1.75 (m, 1H), 1.33-1.25 (m, 4H), 0.87 (t, J = 7.4, 6H). ESI-MS: 404.05 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-[2-(1-piperidyl)ethyl]quinazolin-4- one (54): Compound 54 was synthesized from intermediate 3 (0.11 mmol) and N-(2- aminoethyl)piperidine (0.16 mmol) as a white solid in 57% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.42 (d, J = 5.7, 1H), 8.30 (s, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.79 (d, J = 8.9, 1H), 7.75 (d, J = 2.6, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.33 (d, J = 2.1, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 4.07 (t, J = 6.1, 2H), 3.86 (s, 3H), 2.55 (t, J = 6.1, 2H), 2.42-2.36 (m, 4H), 1.48-1.33 (m, 6H). ESI-MS: 431.10 (M+H)+. 3-{2-[(2R,6S)-2,6-dimethyl-1-piperidyl]ethyl}-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy} quinazolin-4-one (55): Compound 55 was synthesized from intermediate 3 (0.14 mmol) and 2-(cis-2,6- dimethylpiperidin-1-yl)ethanamine (0.20 mmol) as a white solid in 15% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.7, 1H), 8.36 (s, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.80 (d, J = 8.9, 1H), 7.75 (d, J = 2.6, 1H), 7.67 (dd, J = 8.8, 2.9, 1H), 7.33 (d, J = 2.0, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 3.96 (t, J = 7.0, 2H), 3.86 (s, 3H), 2.81 (t, J = 7.0, 2H), 2.46-2.40 (m, 2H), 1.61-1.45 (m, 3H), 1.30-1.22 (m, 1H), 1.15-1.05 (m, 2H), 1.01 (d, J = 6.2, 6H). ESI-MS: 459.15 (M+H)+.
2-(cis-2,6-dimethylpiperidin-1-yl)ethanamine was prepared by the following procedure: To a solution of cis-2,6-dimethylpiperidine (100 mg, 0.88 mmol) and potassium carbonate (245 mg, 1.77 mmol) in acetonitrile (4.5 mL) was added 2- bromoacetonitrile (74 µL, 1.06 mmol). The resulting mixture was stirred at 60°C for 2h. The reaction mixture was diluted with DCM and washed with water. Organic layers were dried over Na2SO4, filtered and concentrated under reduced pressure. The crude was purified by flash column chromatography (CH2Cl2/MeOH from 100/0 to 93/7) to give 126 mg of 2-[(2R,6S)-2,6-dimethyl-1- piperidyl]acetonitrile as a yellow oil in 94% yield. ESI-MS: 153.00 (M+H)+. To a mixture a previous intermediate (126 mg) in MeOH (8 mL) was added Raney nickel (slurry in water, 30 mg). After stirring overnight under a hydrogen atmosphere, the reaction mixture was filtered through a pad of Celite, rinsed with MeOH, and concentrated under reduced pressure to provide 32 mg of the expected compound (25% yield) which was used directly without further purification. ESI-MS: 157.05 (M+H)+. 3-(2-cyclohexylethyl)-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}quinazolin-4- one (56): Compound 56 was synthesized from intermediate 3 (0.11 mmol) and 2- cyclohexylethanamine (0.16 mmol) as a white solid in 38% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43-8.41 (m, 2H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.80 (d, J = 8.9, 1H), 7.75 (d, J = 2.5, 1H), 7.67 (dd, J = 8.8, 2.9, 1H), 7.32 (d, J = 2.0, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 4.02- 3.98 (m, 2H), 3.85 (s, 3H), 1.77-1.55 (m, 7H), 1.33-1.07 (m, 4H), 0.99-0.89 (m, 2H). ESI-MS: 430.10 (M+H)+.
6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-(2-pyrrolidin-1-ylethyl)quinazolin-4- one (57): Compound 57 was synthesized from intermediate 3 (0.11 mmol) and pyrrolidinoethylamine (0.16 mmol) as a white solid in 46% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.42 (d, J = 5.7, 1H), 8.34 (s, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.79 (d, J = 9.0, 1H), 7.76 (d, J = 2.5, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.33 (d, J = 2.0, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 4.09 (t, J = 6.1, 2H), 3.86 (s, 3H), 2.72 (t, J = 6.1, 2H), 1.68-1.64 (m, 4H). ESI-MS: 417.10 (M+H)+. 3-[2-(4,4-difluoro-1-piperidyl)ethyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (58): Compound 58 was synthesized from intermediate 3 (0.11 mmol) and 2-(4,4- difluoro-1-piperidyl)ethanamine (hydrochloride salt 0.21 mmol) as a white solid in 24% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.42 (d, J = 5.7, 1H), 8.36 (s, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.80 (d, J = 8.9, 1H), 7.76 (d, J = 2.6, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.33 (d, J = 2.1, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 4.09 (t, J = 6.0, 2H), 3.86 (s, 3H), 2.70- 2.66 (m, 2H), 2.59-2.55 (m, 4H), 1.95-1.85 (m, 4H). ESI-MS: 467.10 (M+H)+. 2-(4,4-difluoro-1-piperidyl)ethanamine was prepared by the following procedure: To a solution of 4,4-difluoropiperidine (200 mg, 1.65 mmol) and potassium carbonate (456 mg, 3.30 mmol) in acetonitrile (5 mL) was added tert-butyl (2- bromoethyl)carbamate (444 mg, 1.98 mmol). The resulting mixture was stirred at 60°C for 4h. The reaction mixture was diluted with DCM and washed with
water. Organic layers were dried over Na2SO4, filtered and concentrated under reduced pressure. The crude was purified by flash column chromatography (CH2Cl2/MeOH from 100/0 to 93/7) to give 297 mg of tert-butyl N-[2-(4,4-difluoro- 1-piperidyl)ethyl]carbamate as a yellow oil in 68% yield. ESI-MS: 265.00 (M+H)+. To a mixture a previous intermediate (297 mg) in dioxane (2 mL) was added HCl 4M in dioxane (4 mL) and the reaction mixture was stirred at room temperature overnight. The mixture was concentrated under reduced pressure to provide 251 mg of the expected compound as hydrochloride salt (white powder, 100% yield) which was used directly without further purification. ESI-MS: 165.05 (M+H)+. 3-(cyclopentylmethyl)-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}quinazolin-4- one (59): Compound 59 was synthesized from intermediate 3 (0.11 mmol) and (aminomethyl)cyclopentane (0.16 mmol) as a white solid in 33% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43-8.41 (m, 2H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.80 (d, J = 8.8, 1H), 7.76 (d, J = 2.5, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.33 (d, J = 2.1, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 3.93 (d, J = 7.6, 2H), 3.86 (s, 3H), 2.39-2.33 (m, 1H), 1.69-1.45 (m, 6H), 1.33- 1.23 (m, 2H). ESI-MS: 402.05 (M+H)+. 3-[2-(2,2-dimethylpyrrolidin-1-yl)ethyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (60): Compound 60 was synthesized from intermediate 3 (0.11 mmol) and 2-(2,2- dimethylpyrrolidin-1-yl)ethanamine (hydrochloride salt, 0.21 mmol) as a white solid in 35% yield according to the general method D. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.7, 1H), 8.27 (s, 1H), 8.24 (s, 1H), 7.97 (d, J =
0.7, 1H), 7.78 (d, J = 8.8, 1H), 7.75 (d, J = 2.7, 1H), 7.66 (dd, J = 8.8, 2.9, 1H), 7.33 (d, J = 2.2, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 4.00 (t, J = 5.8, 2H), 3.86 (s, 3H), 2.86-2.82 (m, 2H), 2.64 (t, J = 5.8, 2H), 1.72-1.64 (m, 2H), 1.50-1.45 (m, 2H), 0.72 (s, 6H). ESI-MS: 445.10 (M+H)+. 2-(2,2-dimethylpyrrolidin-1-yl)ethanamine was prepared from 2,2- dimethylpyrrolidine (1.00 mmol) and tert-butyl (2-bromoethyl)carbamate (1.51 mmol) according to procedure described for compound 58. ESI-MS: 143.05 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-{2-[(2R)-2-methylpyrrolidin-1- yl]ethyl}quinazolin-4-one (61): Compound 61 was synthesized from intermediate 3 (0.11 mmol) and 2-[(2R)-2- methylpyrrolidin-1-yl]ethanamine (hydrochloride salt,0.21 mmol) as a white solid in 36% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.7, 1H), 8.32 (s, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.5, 1H), 7.79 (d, J = 8.8, 1H), 7.76 (d, J = 2.8, 1H), 7.67 (dd, J = 8.8, 2.9, 1H), 7.33 (d, J = 2.3, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 4.18-4.11 (m, 1H), 4.00-3.93 (m, 1H), 3.86 (s, 3H), 3.25-3.18 (m, 1H), 3.06-2.99 (m, 1H), 2.43-2.32 (m, 2H), 2.19 (q, J = 8.6, 1H), 1.88-1.79 (m, 1H), 1.68-1.61 (m, 2H), 1.26-1.16 (m, 1H), 0.80 (d, J = 6.0, 3H). ESI-MS: 431.10 (M+H)+. 2-[(2R)-2-methylpyrrolidin-1-yl]ethanamine was prepared from (R)-2- methylpyrrolidine (1.17 mmol) and tert-butyl (2-bromoethyl)carbamate (1.76 mmol) according to procedure described for compound 58. ESI-MS: 129.05 (M+H)+.
6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-{2-[(2S)-2-methylpyrrolidin-1- yl]ethyl}quinazolin-4-one (62): Compound 62 was synthesized from intermediate 3 (0.11 mmol) and 2-[(2S)-2- methylpyrrolidin-1-yl]ethanamine (hydrochloride salt, 0.21 mmol) as a white solid in 47% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.7, 1H), 8.32 (s, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.79 (d, J = 8.8, 1H), 7.76 (d, J = 2.8, 1H), 7.67 (dd, J = 8.8, 2.9, 1H), 7.33 (d, J = 2.3, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 4.18-4.11 (m, 1H), 4.01-3.93 (m, 1H), 3.86 (s, 3H), 3.25-3.19 (m, 1H), 3.06-2.99 (m, 1H), 2.43-2.32 (m, 2H), 2.19 (q, J = 8.6, 1H), 1.88-1.79 (m, 1H), 1.68-1.61 (m, 2H), 1.26-1.16 (m, 1H), 0.80 (d, J = 6.0, 3H). ESI-MS: 431.10 (M+H)+. 2-[(2S)-2-methylpyrrolidin-1-yl]ethanamine was prepared from (S)-2- methylpyrrolidine (1.17 mmol) and tert-butyl (2-bromoethyl)carbamate (1.76 mmol) according to procedure described for compound 58. ESI-MS: 129.05 (M+H)+. 3-{2-[(2R,6R)-2,6-dimethylmorpholin-4-yl]ethyl}-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy} quinazolin-4-one (63): Compound 63 was synthesized from intermediate 3 (0.11 mmol) and 2-[(2R,6R)- 2,6-dimethylmorpholin-4-yl]ethanamine (hydrochloride salt, 0.21 mmol) as a white solid in 21% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.7, 1H), 8.32 (s, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.80 (d, J = 8.8, 1H), 7.75 (d, J = 2.8, 1H), 7.68 (dd, J = 8.8, 2.8, 1H), 7.33 (d, J = 2.3, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 4.14-4.03 (m, 2H), 3.86-3.81 (m, 5H), 2.63-2.56 (m, 1H), 2.47-2.42 (m, 3H), 2.20-2.15 (m, 2H), 1.06 (d, J = 6.4, 6H). ESI-MS: 461.15 (M+H)+.
2-[(2R,6R)-2,6-dimethylmorpholin-4-yl]ethanamine was prepared from trans- 2,6-dimethyl morpholine (0.43 mmol) and tert-butyl (2-bromoethyl)carbamate (0.65 mmol) according to procedure described for compound 58. ESI-MS: 159.00 (M+H)+. 3-{2-[(3R,5S)-3,5-dimethylmorpholin-4-yl]ethyl}-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy} quinazolin-4-one (64): Compound 64 was synthesized from intermediate 3 (0.11 mmol) and 2-[(3S,5R)- 3,5-dimethylmorpholin-4-yl]ethanamine (hydrochloride salt, 0.21mmol) as a white solid in 25% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.7, 1H), 8.40 (s, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.5, 1H), 7.80 (d, J = 8.8, 1H), 7.76 (d, J = 2.8, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.33 (d, J = 2.3, 1H), 6.77 (dd, J = 5.7, 2.4, 1H), 3.99 (t, J = 7.0, 2H), 3.86 (s, 3H), 3.59 (dd, J = 11.1, 2.8, 2H), 2.98 (t, J = 10.7, 2H), 2.84 (t, J = 7.0, 2H), 2.57- 2.51 (m, 2H), 0.87 (d, J = 6.2, 6H). ESI-MS: 461.10 (M+H)+. 2-[(3S,5R)-3,5-dimethylmorpholin-4-yl]ethanamine was prepared from (3R,5S)- rel-3,5-dimethylmorpholine (0.43 mmol) and tert-butyl (2-bromoethyl)carbamate (0.87 mmol) according to procedure described for compound 58. ESI-MS: 159.05 (M+H)+. 3-{2-[(2S,6R)-2,6-dimethylmorpholin-4-yl]ethyl}-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy} quinazolin-4-one (65): Compound 65 was synthesized from intermediate 3 (0.11 mmol) and 2-[(2S,6R)- 2,6-dimethylmorpholin-4-yl]ethanamine (hydrochloride salt, 0.21 mmol) as a white solid in 21% yield according to the general method D.1H NMR (400 MHz,
DMSO-d6) δ (ppm): 8.43 (d, J = 5.7, 1H), 8.34 (s, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.80 (d, J = 8.9, 1H), 7.75 (d, J = 2.6, 1H), 7.68 (dd, J = 8.8, 2.9, 1H), 7.33 (d, J = 2.1, 1H), 6.78 (dd, J = 5.7, 2.4, 1H), 4.09 (t, J = 6.0, 2H), 3.86 (s, 3H), 3.51-3.43 (m, 2H), 2.82 (d, J = 10.4, 2H), 2.57 (t, J = 6.0, 2H), 1.67 (t, J = 10.7, 2H), 1.03 (d, J = 6.3, 6H). ESI-MS: 461.15 (M+H)+. 2-[(2S,6R)-2,6-dimethylmorpholin-4-yl]ethanamine was prepared from cis-2,6- dimethyl morpholine (1.73 mmol) and tert-butyl (2-bromoethyl)carbamate (2.60 mmol) according to procedure described for compound 58. ESI-MS: 159.05 (M+H)+. 3-(2-cyclohexyl-2-oxo-ethyl)-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (66): Compound 66 was synthesized from intermediate 3 (0.11 mmol) and 2-amino- 1-cyclohexylethanone hydrochloride (0.21 mmol) as a white solid in 28% yield according to the general method D.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.42 (d, J = 5.7, 1H), 8.28 (s, 1H), 8.24 (s, 1H), 7.99 (d, J = 0.5, 1H), 7.83 (dd, J = 8.4, 0.7, 1H), 7.73-7.69 (m, 2H), 7.34 (d, J = 2.3, 1H), 6.79 (dd, J = 5.7, 2.4, 1H), 5.03 (s, 2H), 3.86 (s, 3H), 2.66-2.59 (m, 1H), 1.92-1.87 (m, 2H), 1.75-1.61 (m, 3H), 1.37-1.13 (m, 5H). ESI-MS: 444.15 (M+H)+. Example 2: General procedure for the preparation of analogues 68-76
Method E: Intermediate 2 (600 mg, 1.85 mmol) was suspended in formamide (4 mL) in an oven-dried screw-cap test tube. The reaction mixture was stirred at 140°C overnight. After cooling at room temperature, the mixture was concentrated under vacuum. The crude was purified by flash column chromatography (DCM/MeOH from 100/0 to 90/10) and reverse phase chromatography (H2O/MeOH: 0 to 100%) to give 151 mg of 6-{[2-(1- methylpyrazol-4-yl)-4-pyridyl]oxy}-3H-quinazolin-4-one 67 as a white powder in 26% yield. ESI-MS: 320.00 (M+H)+. Method F: To a solution of derivative 67 (1 equiv.) in DMF (10 mL/mmol) under nitrogen were added Cs2CO3 (1.2 equiv.) and alkyl bromide derivative (1.2 equiv.). The reaction mixture was stirred at room temperature until completion. The reaction mixture was diluted with EtOAc and washed twice with a saturated solution of NH4Cl. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. The crude product was purified by flash column chromatography (DCM/MeOH from 100/0 to 95/5) and reverse phase chromatography (H2O/MeOH: 0 to 100%) to give the expected compound. The following compounds are examples illustrating Method F: 3-[2-(3-chlorophenyl)-2-oxo-ethyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (68): Compound 68 was synthesized from intermediate 67 (0.07 mmol) and 2-bromo- 1-(3-chlorophenyl)ethanone (0.08 mmol) as a white solid in 73% yield according to the general method F.1H NMR (600 MHz, DMSO-d6) δ (ppm): 8.43 (dd, J = 5.6, 0.4, 1H), 8.35 (s, 1H), 8.28 (s, 1H), 8.12 (t, J = 1.8, 1H), 8.06 (ddd, J = 7.8, 1.6, 1.0, 1H), 7.99 (d, J = 0.7, 1H), 7.88-7.85 (m, 1H), 7.83 (ddd, J = 8.0, 2.2, 1.0, 1H), 7.76 (dd, J = 2.8, 0.4, 1H), 7.74 (dd, J = 8.8, 2.9, 1H), 7.66 (t, J = 7.9, 1H), 7.35-7.34 (m, 1H), 6.81 (dd, J = 5.7, 2.4, 1H), 5.67 (s, 2H), 3.86 (s, 3H). ESI-MS: 472.10 (M+H)+.
3-[2-(4-chlorophenyl)-2-oxo-ethyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (69): Compound 69 was synthesized from intermediate 67 (0.07 mmol) and 2-bromo- 4-chloroacetophenone (0.08 mmol) as a white solid in 56% yield according to the general method F.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.7, 1H), 8.36 (s, 1H), 8.28 (s, 1H), 8.12 (d, J = 8.6, 2H), 7.99 (s, 1H), 7.87 (d, J = 8.6, 1H), 7.75-7.69 (m, 4H), 7.35 (d, J = 2.3, 1H), 6.81 (dd, J = 5.6, 2.4, 1H), 5.65 (s, 2H), 3.86 (s, 3H). ESI-MS: 472.05 (M+H)+. 3-[2-(3-fluorophenyl)-2-oxo-ethyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (70): Compound 70 was synthesized from intermediate 67 (0.06 mmol) and 2-bromo- 1-(3-fluorophenyl)ethan-1-one (0.08 mmol) as a white solid in 65% yield according to the general method F.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.7, 1H), 8.36 (s, 1H), 8.28 (s, 1H), 7.99-7.95 (m, 2H), 7.92-7.85 (m, 2H), 7.77-7.66 (m, 3H), 7.62 (td, J = 8.4, 1.8, 1H), 7.35 (d, J = 2.3, 1H), 6.81 (dd, J = 5.7, 2.4, 1H), 5.66 (s, 2H), 3.86 (s, 3H). ESI-MS: 456.10 (M+H)+.
3-[2-(4-fluorophenyl)-2-oxo-ethyl]-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (71): Compound 71 was synthesized from intermediate 67 (0.06 mmol) and 2-bromo- 1-(4-fluorophenyl)ethanone (0.08 mmol) as a white solid in 65% yield according to the general method F.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.7, 1H), 8.36 (s, 1H), 8.28 (s, 1H), 8.21-8.17 (m, 2H), 7.99 (s, 1H), 7.87 (d, J = 8.7, 1H), 7.76-7.72 (m, 2H), 7.49-7.44 (m, 2H), 7.35 (d, J = 2.3, 1H), 6.81 (dd, J = 5.7, 2.4, 1H), 5.65 (s, 2H), 3.86 (s, 3H). ESI-MS: 456.15 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-{2-oxo-2-[3- (trifluoromethyl)phenyl]ethyl} quinazolin-4-one (72): Compound 72 was synthesized from intermediate 67 (0.07 mmol) and 3- (trifluoromethyl) phenacyl bromide (0.08 mmol) as a white solid in 29% yield according to the general method F.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.44- 8.36 (m, 4H), 8.28 (s, 1H), 8.13 (d, J = 8.0, 1H), 7.99 (s, 1H), 7.90-7.86 (m, 2H), 7.77-7.72 (m, 2H), 7.35 (d, J = 2.3, 1H), 6.81 (dd, J = 5.7, 2.4, 1H), 5.75 (s, 2H), 3.86 (s, 3H). ESI-MS: 506.15 (M+H)+.
6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-{2-oxo-2-[4- (trifluoromethyl)phenyl]ethyl} quinazolin-4-one (73): Compound 73 was synthesized from intermediate 67 (0.06 mmol) and 2-bromo- 1-[4-(trifluoromethyl)phenyl]ethan-1-one (0.08 mmol) as a white solid in 75% yield according to the general method F.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.43 (d, J = 5.7, 1H), 8.37 (s, 1H), 8.31-8.28 (m, 3H), 8.02-7.98 (m, 3H), 7.87 (d, J = 8.9, 1H), 7.77-7.72 (m, 2H), 7.35 (d, J = 2.3, 1H), 6.81 (dd, J = 5.7, 2.4, 1H), 5.71 (s, 2H), 3.86 (s, 3H). ESI-MS: 506.15 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-[2-oxo-2-(2- pyridyl)ethyl]quinazolin-4-one (74): Compound 74 was synthesized from intermediate 67 (0.06 mmol) and 2- (bromoacetyl)pyridine hydrobromide (0.08 mmol) as a white solid in 52% yield according to the general method F.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.84 (d, J = 4.7, 1H), 8.43 (d, J = 5.7, 1H), 8.40 (s, 1H), 8.28 (s, 1H), 8.10 (td, J = 7.7, 1.7, 1H), 8.03 (d, J = 7.7, 1H), 7.99 (s, 1H), 7.87 (d, J = 8.5, 1H), 7.79 (ddd, J = 7.5, 4.8, 1.3, 1H), 7.75-7.71 (m, 2H), 7.35 (d, J = 2.3, 1H), 6.80 (dd, J = 5.7, 2.4, 1H), 5.75 (s, 2H), 3.86 (s, 3H). ESI-MS: 439.10 (M+H)+.
6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-[2-oxo-2-(3- pyridyl)ethyl]quinazolin-4-one (75): Compound 75 was synthesized from intermediate 67 (0.06 mmol) and 3-(2- bromoacetyl)pyridine hydrobromide (0.08 mmol) as a white solid in 37% yield according to the general method F.1H NMR (400 MHz, DMSO-d6) δ (ppm): 9.28 (d, J = 2.0, 1H), 8.90 (dd, J = 4.8, 1.5, 1H), 8.45-8.42 (m, 2H), 8.36 (s, 1H), 8.28 (s, 1H), 7.99 (s, 1H), 7.87 (d, J = 8.6, 1H), 7.77-7.72 (m, 2H), 7.67 (dd, J = 8.0, 4.8, 1H), 7.35 (d, J = 2.3, 1H), 6.81 (dd, J = 5.7, 2.4, 1H), 5.70 (s, 2H), 3.86 (s, 3H). ESI-MS: 439.10 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-[2-oxo-2-(4- pyridyl)ethyl]quinazolin-4-one (76): Compound 76 was synthesized from intermediate 67 (0.06 mmol) and 2-bromo- 1-(pyridin-4-yl)ethanone hydrobromide (0.08 mmol) as a white solid in 19% yield according to the general method F.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.92- 8.90 (m, 2H), 8.43 (d, J = 5.7, 1H), 8.36 (s, 1H), 8.28 (s, 1H), 7.99-7.95 (m, 3H), 7.87 (dd, J = 8.5, 0.6, 1H), 7.77-7.72 (m, 2H), 7.35 (d, J = 2.2, 1H), 6.81 (dd, J = 5.7, 2.4, 1H), 5.68 (s, 2H), 3.86 (s, 3H). ESI-MS: 439.05 (M+H)+.
Example 3: General procedure for the preparation of analogues 79-87 3-Chlorobenzylamine, Vilsmeier reagent, AcOH, CH2Cl2, rt, 1h30 MW, 100°C, 20 min Method C Method D 1 77 78 1) Benzophenone imine, Pd2dba3, BINAP, t-BuONa, toluene, 90°C Boronic ester, Amine, 2) HCl (4M), dioxane, rt Pd(PPh3)4, Cs2CO31M, Pd2dba3, Xantphos, dioxane, 100°C Cs2CO3, dioxane, 100°C Method H Method B Method G R2COOH, EDC.HCl, DMAP, CH2Cl2, rt Method I 85-87 84 79-83 Preparation of methyl 5-[(2-chloro-4-pyridyl)oxy]-2-[(E) dimethylaminomethyleneamino] benzoate (77): Intermediate 77 was synthesized from intermediate 1 (1.12 mmol) and Vilsmeier reagent (1.79 mmol) as a yellow oil in quantitative yield according to the general method C. ESI-MS: 333.95 (M+H)+. Preparation of 3-[(3-chlorophenyl)methyl]-6-[(2-chloro-4-pyridyl)oxy]quinazolin- 4-one (78): Intermediate 78 was synthesized from intermediate 77 (1.19 mmol) and 3- chlorobenzylamine (1.79 mmol) as a white powder in 72% yield according to the general method D. ESI-MS: 397.85 (M+H)+.
The following compounds are examples illustrating Method B: 3-[(3-chlorophenyl)methyl]-6-{[2-(1-methylpyrazol-3-yl)-4- pyridyl]oxy}quinazolin-4-one (79): Compound 79 was synthesized from intermediate 78 (0.06 mmol) and 1-methyl- 3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole (0.08 mmol) as a white solid in 47% yield according to the general method B.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.63 (s, 1H), 8.50 (d, J = 5.7, 1H), 7.85 (d, J = 8.8, 1H), 7.80 (d, J = 2.7, 1H), 7.75-7.71 (m, 2H), 7.50 (s, 1H), 7.41-7.33 (m, 4H), 6.99 (dd, J = 5.6, 2.5, 1H), 6.79 (d, J = 2.2, 1H), 5.20 (s, 2H), 3.84 (s, 3H). ESI-MS: 444.15 (M+H)+. 3-[(3-chlorophenyl)methyl]-6-{[2-(2-methylpyrazol-3-yl)-4- pyridyl]oxy}quinazolin-4-one (80): Compound 80 was synthesized from intermediate 78 (0.06 mmol) and 1-methyl- 1H-pyrazole-5-boronic acid pinacol ester (0.08 mmol) as a white solid in 54% yield according to the general method B.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.63 (s, 1H), 8.58 (d, J = 5.7, 1H), 7.84 (d, J = 8.9, 1H), 7.80 (d, J = 2.7, 1H), 7.73 (dd, J = 8.8, 2.9, 1H), 7.50-7.45 (m, 3H), 7.41-7.33 (m, 3H), 6.97 (dd, J = 5.7, 2.4, 1H), 6.78 (d, J = 2.0, 1H), 5.20 (s, 2H), 4.13 (s, 3H). ESI-MS: 444.00 (M+H)+. Method G: To a solution of 78 (25 mg, 0.06 mmol) in dioxane (1 mL) under nitrogen were added amine derivative (2 equiv.), Pd2dba3 (0.1 equiv.), Xantphos (0.2 equiv.) and Cs2CO3 (2 equiv.). The mixture was stirred at 100°C for 2 h. The reaction mixture was concentrated under reduced pressure. The crude was purified by flash column chromatography (DCM/MeOH from 100/0 to 90/10) and
reverse phase chromatography (H2O/MeOH from 100/0 to 0/100) to give the expected compound. The following compounds are examples illustrating Method G: 3-[(3-chlorophenyl)methyl]-6-({2-[(1-methylpyrazol-3-yl)amino]-4- pyridyl}oxy)quinazolin-4-one (81): Compound 81 was synthesized from intermediate 78 (0.06 mmol) and 1-methyl- 1H-pyrazol-3-amine (0.13 mmol) as a white solid in 52% yield according to the general method G.1H NMR (400 MHz, DMSO-d6) δ (ppm): 9.23 (bs, 1H), 8.62 (s, 1H), 8.04 (d, J = 5.7, 1H), 7.82 (d, J = 8.8, 1H), 7.74 (d, J = 2.8, 1H), 7.67 (dd, J = 8.8, 2.8, 1H), 7.49 (s, 1H), 7.45 (d, J = 2.2, 1H), 7.41-7.33 (m, 3H), 6.97 (d, J = 2.1, 1H), 6.37 (dd, J = 5.7, 2.3, 1H), 6.16 (d, J = 2.2, 1H), 5.20 (s, 2H), 3.60 (s, 3H). ESI-MS: 459.00 (M+H)+. 3-[(3-chlorophenyl)methyl]-6-({2-[(1-methylpyrazol-4-yl)amino]-4- pyridyl}oxy)quinazolin-4-one (82): Compound 82 was synthesized from intermediate 78 (0.06 mmol) and 1-methyl- 1H-pyrazol-4-ylamine (0.13 mmol) as a white solid in 58% yield according to the general method G.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.73 (bs, 1H), 8.62 (s, 1H), 8.04 (d, J = 5.8, 1H), 7.88 (s, 1H), 7.82 (d, J = 8.8, 1H), 7.74 (d, J = 2.7, 1H), 7.67 (dd, J = 8.8, 2.8, 1H), 7.49 (s, 1H), 7.41-7.32 (m, 4H), 6.35 (dd, J = 5.8, 2.2, 1H), 6.11 (d, J = 2.2, 1H), 5.20 (s, 2H), 3.77 (s, 3H). ESI-MS: 459.00 (M+H)+.
3-[(3-chlorophenyl)methyl]-6-({2-[(2-methylpyrazol-3-yl)amino]-4- pyridyl}oxy)quinazolin-4-one (83): Compound 83 was synthesized from intermediate 78 (0.06 mmol) and 1-methyl- 1H-pyrazol-5-amine (0.13 mmol) as a white solid in 55% yield according to the general method G.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.74 (bs, 1H), 8.63 (s, 1H), 8.05 (d, J = 5.8, 1H), 7.83 (d, J = 8.8, 1H), 7.77 (d, J = 2.7, 1H), 7.69 (dd, J = 8.8, 2.8, 1H), 7.49 (s, 1H), 7.42-7.33 (m, 3H), 7.27 (d, J = 1.9, 1H), 6.49 (dd, J = 5.8, 2.2, 1H), 6.24 (d, J = 2.2, 1H), 6.19 (d, J = 1.9, 1H), 5.20 (s, 2H), 3.61 (s, 3H). ESI-MS: 459.00 (M+H)+. Method H: To a solution of 78 (213 mg, 0.54 mmol) in toluene (6 mL) under nitrogen were added benzophenone imine (135 µL, 0.80 mmol), Pd2dba3 (49 mg, 0.05 mmol), Binap (67 mg, 0.11 mmol) and t-BuONa (77 mg, 0.08 mmol). The mixture was stirred at 90°C for 2h30. The reaction mixture was concentrated under reduced pressure. The crude was suspended in HCl 4M in dioxane and the reaction mixture was stirred at room temperature for 30 minutes. The reaction mixture was concentrated under reduced pressure and purified by flash column chromatography (DCM/MeOH from 100/0 to 97/3) and reverse phase chromatography (H2O/MeOH from 100/0 to 0/100) to give 69 mg of 6-[(2-amino- 4-pyridyl)oxy]-3-[(3-chlorophenyl)methyl]quinazolin-4-one 84 as a white powder in 34% yield. ESI-MS: 378.90 (M+H)+. Method I: To a suspension of appropriate intermediate 84 (23 mg, 0.06 mmol) in DCM (3 mL) under nitrogen were added DMAP (4.4 equiv.), EDC.HCl (4 equiv.) and appropriate carboxylic acid (3 equiv.). The reaction mixture was stirred at room temperature overnight. The reaction mixture was diluted with DCM and washed twice with a saturated solution of NH4Cl. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. The crude product was purified by flash column chromatography (DCM/MeOH from
100/0 to 95/5) and reverse phase chromatography (H2O/MeOH from 100/0 to 0/100) to give the expected compound. The following compounds are examples illustrating Method I: N-(4-{3-[(3-chlorophenyl)methyl]-4-oxo-quinazolin-6-yl}oxy-2-pyridyl)-1-methyl- pyrazole-3-carboxamide (85): Compound 85 was synthesized from intermediate 84 (0.06 mmol) and 1-methyl- 1H-pyrazole-3-carboxylic acid (0.19 mmol) as a white solid in 80% yield according to the general method I.1H NMR (400 MHz, DMSO-d6) δ (ppm): 9.58 (bs, 1H), 8.64 (s, 1H), 8.27 (d, J = 5.9, 1H), 7.87-7.83 (m, 2H), 7.82 (d, J = 2.6, 1H), 7.78 (d, J = 2.2, 1H), 7.73 (dd, J = 8.8, 2.8, 1H), 7.50 (s, 1H), 7.41-7.33 (m, 3H), 6.82 (dd, J = 5.7, 2.4, 1H), 6.80 (d, J = 2.3, 1H), 5.20 (s, 2H), 3.95 (s, 3H). ESI-MS: 487.00 (M+H)+. N-(4-{3-[(3-chlorophenyl)methyl]-4-oxo-quinazolin-6-yl}oxy-2-pyridyl)-1-methyl- pyrazole-4-carboxamide (86): Compound 86 was synthesized from intermediate 84 (0.06 mmol) and 1-methyl- 1H-pyrazole-4-carboxylic acid (0.19 mmol) as a white solid in 14% yield according to the general method I.1H NMR (400 MHz, DMSO-d6) δ (ppm): 10.59 (bs, 1H), 8.63 (s, 1H), 8.39 (s, 1H), 8.28 (d, J = 5.9, 1H), 8.08 (d, J = 0.7, 1H), 7.85-7.82 (m, 2H), 7.79 (d, J = 2.6, 1H), 7.71 (dd, J = 8.8, 2.8, 1H), 7.49 (s, 1H), 7.41-7.33 (m, 3H), 6.79 (dd, J = 5.7, 2.4, 1H), 5.20 (s, 2H), 3.86 (s, 3H). ESI-MS: 487.00 (M+H)+.
N-(4-{3-[(3-chlorophenyl)methyl]-4-oxo-quinazolin-6-yl}oxy-2-pyridyl)-2-methyl- pyrazole-3-carboxamide (87): Compound 87 was synthesized from intermediate 84 (0.06 mmol) and 1-methyl- 1H-pyrazole-5-carboxylic acid (0.19 mmol) as a white solid in 80% yield according to the general method I.1H NMR (400 MHz, DMSO-d6) δ (ppm): 10.91 (bs, 1H), 8.63 (s, 1H), 8.32 (d, J = 5.7, 1H), 7.85 (d, J = 8.9, 1H), 7.80-7.79 (m, 2H), 7.73 (dd, J = 8.8, 2.8, 1H), 7.50-7.49 (m, 2H), 7.41-7.33 (m, 3H), 7.26 (d, J = 2.1, 1H), 6.84 (dd, J = 5.7, 2.3, 1H), 5.20 (s, 2H), 4.03 (s, 3H). ESI-MS: 487.05 (M+H)+. Example 4: General procedure for the preparation of analogues 104-119 NH2 NH2 NH2 O OH NH4OH, HOBt, NH2 O NaOH 2N, MeOH, O EDC.HCl, NMM, O 50°C, 2h DMF, rt, 2h O O O Method J Method K N N N N N N N 2 N 88 N 89 RCOCl, TEA, RCOOH, DMAP, THF, rt, 2h HOBt, EDC.HCl, Method L DMF, rt, Method M O R N R N R NH N NH O Cs2CO3, MeI, O t-BuOK, t-BuOH, 80°C, 1 NH2 DMF h O , rt O O Method F Method N O N N N N N N N N 118-119 104-117 N 90-103 Method J: To a solution of intermediate 2 (582 mg, 1.79 mmol) in MeOH (6 mL) was added a solution of NaOH 2N (1.79 mL, 3.59 mmol). The reaction mixture was stirred at 50°C for 2h. MeOH was evaporated under reduced pressure and the residue was then acidified with concentrated HCl until pH = 2-3. The mixture was evaporated under reduced pressure and purified by reverse phase
chromatography (H2O/MeOH from 100/0 to 0/100) to give 445 mg of 2-amino-5- {[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}benzoic acid 88 as a white powder in 80% yield. ESI-MS: 310.95 (M+H)+. Method K: To a solution of 88 (138 mg, 0.45 mmol) in DMF (4 mL) cooled to 0°C with an ice bath were added HOBt (136 mg, 0.89 mmol), EDC.HCl (171 mg, 0.89 mmol) and N-methylmorpholine (98 µL, 0.89 mmol). Ammonium hydroxide (30%, 0.77 mmol) was then added dropwise. The ice bath was removed, and the reaction mixture was allowed to stir at room temperature for 2h. The reaction mixture was concentrated under reduced pressure. The residue was purified by flash column chromatography (DCM/MeOH from 100/0 to 92/8) and reverse phase chromatography (H2O/MeOH from 100/0 to 0/100) to give 112 mg of 2- amino-5-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}benzamide 89 as a white powder in 81% yield. ESI-MS: 309.95 (M+H)+. Method L: To a suspension of intermediate 89 (30 mg, 0.10 mmol) in THF (1 mL) under nitrogen were added TEA (1.3 equiv.) and appropriate acyl chloride (1.2 equiv.). The reaction mixture was stirred at room temperature for 2h. The reaction mixture was diluted with EtOAc and washed twice with a saturated solution of NH4Cl. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. The crude was directly used in the next step without purification. Method M: To a solution of intermediate 89 (30 mg, 0.10 mmol) in DMF (3 mL) were added DMAP (2 equiv.), HOBt (2 equiv.), EDC.HCl (2 equiv.) and appropriate carboxylic acid (1.5 equiv.). The reaction mixture was stirred at room temperature overnight. The reaction mixture was diluted with EtOAc and washed twice with a saturated solution of NH4Cl. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by flash column chromatography (DCM/MeOH from 100/0 to 90/10) or directly used in the next step without purification.
The following table illustrates intermediates 89-102 prepared from method L and M:
Method N: To a solution of appropriate amide derivative 90-103 (1 equiv.) in t-
BuOH (15 mL/mmol) was added t-BuOK (2 equiv.), and the reaction mixture was stirred at 80°C for 1h. The reaction mixture was diluted with EtOAc and washed twice with a saturated solution of NH4Cl. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. The crude product was purified by flash column chromatography (DCM/MeOH from 100/0 to 95/5) and reverse phase chromatography (H2O/MeOH: 0 to 100%) to give the expected compound. The following compounds are examples illustrating Method N: 2-[(3-fluorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3H- quinazolin-4-one (104): Compound 104 was synthesized from intermediate 90 (0.10 mmol) and t-BuOK (0.19 mmol) as a white solid in 32% yield according to the general method N.1H NMR (400 MHz, DMSO-d6) δ (ppm): 12.54 (bs, 1H), 8.41 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.6, 1H), 7.72 (d, J = 8.9, 1H), 7.69 (d, J = 2.7, 1H), 7.62 (dd, J = 8.8, 2.9, 1H), 7.41-7.35 (m, 1H), 7.28 (d, J = 2.2, 1H), 7.26-7.21 (m, 2H), 7.10 (ddd, J = 8.3, 2.6, 1.2, 1H), 6.76 (dd, J = 5.7, 2.4, 1H), 3.98 (s, 2H), 3.85 (s, 3H). ESI-MS: 428.00 (M+H)+. 2-[(4-fluorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3H- quinazolin-4-one (105): Compound 105 was synthesized from intermediate 91 (0.10 mmol) and t-BuOK (0.19 mmol) as a white solid in 53% yield according to the general method N.1H NMR (400 MHz, DMSO-d6) δ (ppm): 12.56 (bs, 1H), 8.41 (d, J = 5.7, 1H), 8.25
(s, 1H), 7.97 (d, J = 0.7, 1H), 7.71 (d, J = 8.9, 1H), 7.68 (d, J = 2.5, 1H), 7.61 (dd, J = 8.8, 2.9, 1H), 7.46-7.40 (m, 2H), 7.28 (d, J = 2.2, 1H), 7.19-7.13 (m, 2H), 6.75 (dd, J = 5.7, 2.4, 1H), 3.94 (s, 2H), 3.85 (s, 3H). ESI-MS: 428.00 (M+H)+. 2-[(3-chlorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3H- quinazolin-4-one (106): Compound 106 was synthesized from intermediate 92 (0.10 mmol) and t-BuOK (0.19 mmol) as a white solid in 23% yield according to the general method N.1H NMR (400 MHz, DMSO-d6) δ (ppm): 12.54 (bs, 1H), 8.41 (d, J = 5.6, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.6, 1H), 7.72 (d, J = 8.9, 1H), 7.68 (d, J = 2.7, 1H), 7.62 (dd, J = 8.8, 2.9, 1H), 7.49 (s, 1H), 7.40-7.31 (m, 3H), 7.28 (d, J = 2.2, 1H), 6.76 (dd, J = 5.7, 2.4, 1H), 3.97 (s, 2H), 3.85 (s, 3H). ESI-MS: 444.00 (M+H)+. 2-[(4-chlorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3H- quinazolin-4-one (107): Compound 107 was synthesized from intermediate 93 (0.10 mmol) and t-BuOK (0.19 mmol) as a white solid in 35% yield according to the general method N.1H NMR (400 MHz, DMSO-d6) δ (ppm): 12.54 (bs, 1H), 8.41 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.7, 1H), 7.71 (d, J = 8.8, 1H), 7.68 (d, J = 2.6, 1H), 7.61 (dd, J = 8.8, 2.9, 1H), 7.43-7.38 (m, 4H), 7.28 (d, J = 2.1, 1H), 6.76 (dd, J = 5.7, 2.4, 1H), 3.95 (s, 2H), 3.85 (s, 3H). ESI-MS: 444.00 (M+H)+.
2-benzyl-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3H-quinazolin-4-one (108): Compound 108 was synthesized from intermediate 94 (0.10 mmol) and t-BuOK (0.19 mmol) as a white solid in 27% yield according to the general method N.1H NMR (400 MHz, DMSO-d6) δ (ppm): 12.54 (bs, 1H), 8.40 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.7, 1H), 7.72 (d, J = 9.0, 1H), 7.68 (d, J = 2.6, 1H), 7.61 (dd, J = 8.8, 2.9, 1H), 7.41-7.38 (m, 2H), 7.35-7.31 (m, 2H), 7.28-7.23 (m, 2H), 6.76 (dd, J = 5.7, 2.4, 1H), 3.95 (s, 2H), 3.85 (s, 3H). ESI-MS: 410.05 (M+H)+. 2-[(3,5-difluorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3H- quinazolin-4-one (109): Compound 109 was synthesized from intermediate 95 (0.10 mmol) and t-BuOK (0.19 mmol) as a white solid in 42% yield according to the general method N.1H NMR (400 MHz, DMSO-d6) δ (ppm): 12.53 (bs, 1H), 8.41 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.7, 1H), 7.72 (d, J = 8.8, 1H), 7.69 (d, J = 2.5, 1H), 7.62 (dd, J = 8.8, 2.9, 1H), 7.28 (d, J = 2.1, 1H), 7.17-7.10 (m, 3H), 6.76 (dd, J = 5.7, 2.4, 1H), 4.00 (s, 2H), 3.85 (s, 3H). ESI-MS: 446.00 (M+H)+. 2-[(3,4-difluorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3H- quinazolin-4-one (110): Compound 110 was synthesized from intermediate 96 (0.10 mmol) and t-BuOK
(0.19 mmol) as a white solid in 23% yield according to the general method N.1H NMR (400 MHz, DMSO-d6) δ (ppm): 12.52 (bs, 1H), 8.41 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.7, 1H), 7.71 (d, J = 8.8, 1H), 7.68 (d, J = 2.5, 1H), 7.61 (dd, J = 8.8, 2.9, 1H), 7.50-7.36 (m, 2H), 7.29 (d, J = 2.1, 1H), 7.25-7.21 (m, 1H), 6.76 (dd, J = 5.7, 2.4, 1H), 3.96 (s, 2H), 3.85 (s, 3H). ESI-MS: 446.00 (M+H)+. 2-[(4-chloro-3-fluoro-phenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}- 3H-quinazolin-4-one (111): Compound 111 was synthesized from intermediate 97 (0.10 mmol) and t-BuOK (0.19 mmol) as a white solid in 22% yield according to the general method N.1H NMR (400 MHz, DMSO-d6) δ (ppm): 12.53 (bs, 1H), 8.41 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.7, 1H), 7.71 (d, J = 8.8, 1H), 7.68 (d, J = 2.5, 1H), 7.61 (dd, J = 8.8, 2.9, 1H), 7.56 (t, J = 8.1, 1H), 7.45 (dd, J = 10.5, 1.9, 1H), 7.29 (d, J = 2.1, 1H), 7.25 (dd, J = 8.3, 1.4, 1H), 6.75 (dd, J = 5.7, 2.4, 1H), 3.99 (s, 2H), 3.85 (s, 3H). ESI-MS: 462.00 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2-(3-pyridylmethyl)-3H-quinazolin-4- one (112): Compound 112 was synthesized from intermediate 98 (0.11 mmol) and t-BuOK (0.21 mmol) as a white solid in 68% yield according to the general method N.1H NMR (400 MHz, DMSO-d6) δ (ppm): 12.58 (bs, 1H), 8.61 (d, J = 1.8, 1H), 8.48 (dd, J = 4.8, 1.5, 1H), 8.41 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.81-7.78 (m, 1H), 7.71-7.68 (m, 2H), 7.61 (dd, J = 8.8, 2.9, 1H), 7.37 (dd, J = 7.7, 4.8, 1H), 7.28 (d, J = 2.3, 1H), 6.76 (dd, J = 5.7, 2.4, 1H), 4.00 (s, 2H), 3.85 (s, 3H). ESI-MS: 411.05 (M+H)+.
6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2-(4-pyridylmethyl)-3H-quinazolin-4- one (113): Compound 113 was synthesized from intermediate 99 (0.09 mmol) and t-BuOK (0.17 mmol) as a white solid in 6% yield according to the general method N.1H NMR (400 MHz, DMSO-d6) δ (ppm): 12.53 (bs, 1H), 8.53-8.51 (m, 2H), 8.41 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.5, 1H), 7.72-7.68 (m, 2H), 7.62 (dd, J = 8.8, 2.9, 1H), 7.39 (d, J = 6.0, 2H), 7.29 (d, J = 2.3, 1H), 6.76 (dd, J = 5.7, 2.4, 1H), 4.00 (s, 2H), 3.85 (s, 3H). ESI-MS: 411.00 (M+H)+. 2-[(6-methoxy-3-pyridyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3H- quinazolin-4-one (114): Compound 114 was synthesized from intermediate 100 (0.13 mmol) and t-BuOK (0.25 mmol) as a white solid in 40% yield according to the general method N.1H NMR (400 MHz, DMSO-d6) δ (ppm): 12.53 (bs, 1H), 8.40 (d, J = 5.7, 1H), 8.26 (s, 1H), 8.18 (d, J = 2.2, 1H), 7.97 (s, 1H), 7.73-7.67 (m, 3H), 7.61 (dd, J = 8.8, 2.9, 1H), 7.28 (d, J = 2.3, 1H), 6.80 (d, J = 8.5, 1H), 6.75 (dd, J = 5.7, 2.4, 1H), 3.90 (s, 2H), 3.85 (s, 3H), 3.83 (s, 3H). ESI-MS: 441.10 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2-(2-phenylethyl)-3H-quinazolin-4- one (115): Compound 115 was synthesized from intermediate 101 (0.09 mmol) and t-BuOK
(0.17 mmol) as a white solid in 11% yield according to the general method N.1H NMR (400 MHz, DMSO-d6) δ (ppm): 12.39 (bs, 1H), 8.42 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.73 (d, J = 8.9, 1H), 7.69 (d, J = 2.6, 1H), 7.62 (dd, J = 8.8, 2.9, 1H), 7.32-7.28 (m, 5H), 7.23-7.17 (m, 1H), 6.76 (dd, J = 5.7, 2.4, 1H), 3.86 (s, 3H), 3.08-3.04 (m, 2H), 2.93-2.89 (m, 2H). ESI-MS: 424.05 (M+H)+. 2-(cyclohexylmethyl)-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3H-quinazolin- 4-one (116): Compound 116 was synthesized from intermediate 102 (0.10 mmol) and t-BuOK (0.19 mmol) as a white solid in 5% yield according to the general method N.1H NMR (400 MHz, DMSO-d6) δ (ppm): 12.22 (bs, 1H), 8.41 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.72-7.67 (m, 2H), 7.61 (dd, J = 8.8, 2.8, 1H), 7.30 (d, J = 2.2, 1H), 6.76 (dd, J = 5.6, 2.3, 1H), 3.85 (s, 3H), 1.91-1.85 (m, 1H), 1.74-1.60 (m, 5H), 1.30-1.12 (m, 3H), 1.05-0.96 (m, 2H). ESI-MS: 416.10 (M+H)+. 2-isobutyl-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3H-quinazolin-4-one (117): Compound 117 was synthesized from intermediate 103 (0.09 mmol) and t-BuOK (0.17 mmol) as a white solid in 30% yield according to the general method N.1H NMR (400 MHz, DMSO-d6) δ (ppm): 12.26 (bs, 1H), 8.41 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.72 (d, J = 8.8, 1H), 7.68 (d, J = 2.6, 1H), 7.62 (dd, J = 8.8, 2.9, 1H), 7.31 (d, J = 2.3, 1H), 6.76 (dd, J = 5.7, 2.4, 1H), 3.86 (s, 3H), 2.24-2.14 (m, 1H), 0.95 (d, J = 6.6, 6H). ESI-MS: 376.00 (M+H)+.
Method M: To a solution of derivative 106-107 (1 equiv.) in DMF (10 mL/mmol) under nitrogen were added Cs2CO3 (1.2 equiv.) and iodomethane (1.2 equiv.). The reaction mixture was stirred at room temperature until completion. The reaction mixture was diluted with EtOAc and washed twice with a saturated solution of NH4Cl. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. The crude product was purified by flash column chromatography (DCM/MeOH from 100/0 to 95/5) and reverse phase chromatography (H2O/MeOH: 0 to 100%) to give the expected compound. The following compounds are examples illustrating Method F: 2-[(3-chlorophenyl)methyl]-3-methyl-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (118): Compound 118 was synthesized from intermediate 106 (0.04 mmol) and MeI (0.05 mmol) as a white solid in 48% yield according to the general method F.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.42 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.7, 1H), 7.73-7.71 (m, 2H), 7.64 (dd, J = 8.9, 2.8, 1H), 7.42-7.33 (m, 3H), 7.31-7.30 (m, 1H), 7.27 (dt, J = 7.3, 1.5, 1H), 6.76 (dd, J = 5.7, 2.4, 1H), 4.33 (s, 2H), 3.85 (s, 3H), 3.48 (s, 3H). ESI-MS: 458.05 (M+H)+. 2-[(4-chlorophenyl)methyl]-3-methyl-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}quinazolin-4-one (119): Compound 119 was synthesized from intermediate 107 (0.05 mmol) and MeI (0.06 mmol) as a white solid in 78% yield according to the general method F.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.42 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.6, 1H), 7.73-7.71 (m, 2H), 7.64 (dd, J = 8.9, 2.7, 1H), 7.44-7.40 (m, 2H),
7.36-7.30 (m, 3H), 6.76 (dd, J = 5.7, 2.4, 1H), 4.31 (s, 2H), 3.86 (s, 3H), 3.47 (s, 3H). ESI-MS: 458.05 (M+H)+. Example 5: General procedure for the preparation of analogues 145-167 Preparation of methyl 5-[(2-chloro-4-pyridyl)oxy]-2-hydroxy-benzoate (120): Intermediate 120 was synthesized from 2-chloro-4-nitropyridine (3.15 mmol) and methyl 2,5-dihydroxybenzoate (3.15 mmol) as a white powder in 60% yield according to the general method A. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 10.50 (bs, 1H), 8.27 (d, J = 5.8, 1H), 7.54 (d, J = 3.0, 1H), 7.42 (dd, J = 9.0, 3.1, 1H), 7.11 (d, J = 9.0, 1H), 6.99 (d, J = 2.2, 1H), 6.93 (dd, J = 5.8, 2.3, 1H), 3.87 (s, 3H). ESI-MS: 279.90 (M+H)+.
Preparation of 2-hydroxy-5-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}benzoic acid (121): Intermediate 121 was synthesized from intermediate 120 (1.90 mmol) and 1- methyl-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole (2.85 mmol) as a white powder in 90% yield according to the general method B. ESI-MS: 311.90 (M+H)+. Method O: To a solution of intermediate 121 (1 equiv.) in DMF (10 mL/mmol) were added the appropriate amine derivative (1.5 equiv.), HOBt (1.2 equiv.), and EDC.HCl (1.2 equiv.). The reaction mixture was stirred at room temperature overnight. The reaction mixture was diluted with EtOAc and washed twice with a saturated solution of NH4Cl. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by flash column chromatography (DCM/MeOH from 100/0 to 90/10) to give the expected compound. The following table illustrates intermediates 122-144 prepared from method O:
Method P1: To a solution of appropriate derivative 122-125 (1 equiv.) in DMF (10 mL/mmol) were added Cs2CO3 (8 equiv.) and dibromomethane (4 equiv.), and the reaction mixture was stirred at 90°C until completion. The reaction mixture was diluted with EtOAc and washed twice with a saturated solution of NH4Cl. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. The crude product was purified by flash column chromatography (DCM/MeOH from 100/0 to 95/5) and reverse phase
chromatography (H2O/MeOH: 0 to 100%) to give the expected compound. The following compounds are examples illustrating Method P1: 3-(3-fluorophenyl)-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2H-1,3- benzoxazin-4-one (145): Compound 145 was synthesized from intermediate 122 (0.04 mmol) as a white solid in 40% yield according to the general method P1. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.39 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.59 (d, J = 2.9, 1H), 7.53-7.47 (m, 2H), 7.35-7.25 (m, 4H), 7.19-7.14 (m, 1H), 6.69 (dd, J = 5.7, 2.4, 1H), 5.76 (s, 2H), 3.86 (s, 3H). ESI-MS: 417.05 (M+H)+. 3-(4-fluorophenyl)-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2H-1,3- benzoxazin-4-one (146): Compound 146 was synthesized from intermediate 123 (0.06 mmol) as a white solid in 42% yield according to the general method P1. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.39 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.58 (d, J = 2.9, 1H), 7.49-7.42 (m, 3H), 7.33-7.25 (m, 4H), 6.68 (dd, J = 5.7, 2.4, 1H), 5.71 (s, 2H), 3.86 (s, 3H). ESI-MS: 417.05 (M+H)+.
3-(3-chlorophenyl)-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2H-1,3- benzoxazin-4-one (147): N O N O N Cl N O Compound 147 was synthesized from intermediate 124 (0.06 mmol) as a white solid in 25% yield according to the general method P1. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.39 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.59 (d, J = 2.9, 1H), 7.54 (t, J = 2.0, 1H), 7.51-7.46 (m, 2H), 7.40 (d, J = 1.9, 1H), 7.38-7.37 (m, 1H), 7.29 (d, J = 8.8, 1H), 7.26 (d, J = 2.4, 1H), 6.68 (dd, J = 5.7, 2.4, 1H), 5.76 (s, 2H), 3.86 (s, 3H). ESI-MS: 433.00 (M+H)+. 3-(4-chlorophenyl)-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2H-1,3- benzoxazin-4-one (148): Compound 148 was synthesized from intermediate 125 (0.05 mmol as a white solid in 45% yield according to the general method P1. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.39 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.58 (d, J = 2.9, 1H), 7.54-7.41 (m, 5H), 7.29 (d, J = 8.8, 1H), 7.26 (d, J = 2.3, 1H), 6.68 (dd, J = 5.7, 2.3, 1H), 5.74 (s, 2H), 3.86 (s, 3H). ESI-MS: 433.00 (M+H)+. Method P2: To a solution of appropriate derivative 126-144 (1 equiv.) in DMF (10 mL/mmol) were added NaOH (8 equiv.) and dibromomethane (4 equiv.), and the reaction mixture was stirred at 90°C until completion. The reaction mixture was diluted with EtOAc and washed twice with a saturated solution of NH4Cl. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. The crude product was purified by flash column chromatography (DCM/MeOH from 100/0 to 95/5) and reverse phase chromatography (H2O/MeOH: 0 to 100%) to give the expected compound.
The following compounds are examples illustrating Method P2: 3-[(3-fluorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2H-1,3- benzoxazin-4-one (149): Compound 149 was synthesized from intermediate 126 (0.07 mmol) as a white solid in 45% yield according to the general method P2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.53 (d, J = 3.0, 1H), 7.44-7.38 (m, 2H), 7.26 (d, J = 2.3, 1H), 7.22-7.10 (m, 4H), 6.66 (dd, J = 5.7, 2.4, 1H), 5.41 (s, 2H), 4.72 (s, 2H), 3.86 (s, 3H). ESI-MS: 431.05 (M+H)+. 3-[(4-fluorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2H-1,3- benzoxazin-4-one (150): Compound 150 was synthesized from intermediate 127 (0.07 mmol) as a white solid in 29% yield according to the general method P2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.52 (d, J = 3.0, 1H), 7.42-7.38 (m, 3H), 7.25 (d, J = 2.4, 1H), 7.22-7.16 (m, 3H), 6.66 (dd, J = 5.7, 2.4, 1H), 5.37 (s, 2H), 4.68 (s, 2H), 3.86 (s, 3H). ESI-MS: 431.10 (M+H)+. 3-[(3-chlorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2H-1,3- benzoxazin-4-one (151): Compound 151 was synthesized from intermediate 128 (0.05 mmol) as a white
solid in 42% yield according to the general method P2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.52 (d, J = 3.0, 1H), 7.44- 7.36 (m, 5H), 7.25 (d, J = 2.4, 1H), 7.20 (d, J = 8.8, 1H), 6.66 (dd, J = 5.7, 2.4, 1H), 5.38 (s, 2H), 4.69 (s, 2H), 3.86 (s, 3H). ESI-MS: 447.00 (M+H)+. 3-[(4-chlorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2H-1,3- benzoxazin-4-one (152): Compound 152 was synthesized from intermediate 129 (0.07 mmol) as a white solid in 41% yield according to the general method P2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.6, 1H), 7.53 (d, J = 3.0, 1H), 7.43-4.31 (m, 5H), 7.26 (d, J = 2.3, 1H), 7.21 (d, J = 8.8, 1H), 6.66 (dd, J = 5.7, 2.4, 1H), 5.41 (s, 2H), 4.70 (s, 2H), 3.86 (s, 3H). ESI-MS: 447.05 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-{[3-(trifluoromethyl)phenyl]methyl}- 2H-1,3-benzoxazin-4-one (153): Compound 153 was synthesized from intermediate 130 (0.07 mmol) as a white solid in 39% yield according to the general method P2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.72 (s, 1H), 7.68-7.59 (m, 3H), 7.53 (d, J = 2.8, 1H), 7.42 (dd, J = 8.8, 2.9, 1H), 7.26 (d, J = 1.9, 1H), 7.21 (d, J = 8.8, 1H), 6.66 (dd, J = 5.6, 2.2, 1H), 5.44 (s, 2H), 4.79 (s, 2H), 3.86 (s, 3H). ESI-MS: 481.05 (M+H)+.
6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-{[4-(trifluoromethyl)phenyl]methyl}- 2H-1,3-benzoxazin-4-one (154): Compound 154 was synthesized from intermediate 131 (0.08 mmol) as a white solid in 46% yield according to the general method P2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.74 (d, J = 8.1, 2H), 7.57 (d, J = 8.1, 2H), 7.53 (d, J = 3.0, 1H), 7.42 (dd, J = 8.8, 3.0, 1H), 7.26 (d, J = 2.3, 1H), 7.21 (d, J = 8.8, 1H), 6.66 (dd, J = 5.7, 2.4, 1H), 5.42 (s, 2H), 4.80 (s, 2H), 3.86 (s, 3H). ESI-MS: 481.05 (M+H)+. 3-[(3,5-difluorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2H- 1,3-benzoxazin-4-one (155): Compound 155 was synthesized from intermediate 132 (0.07 mmol) as a white solid in 25% yield according to the general method P2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.6, 1H), 7.53 (d, J = 3.0, 1H), 7.42 (dd, J = 8.8, 3.0, 1H), 7.26 (d, J = 2.2, 1H), 7.22 (d, J = 8.8, 1H), 7.20-7.13 (m, 1H), 7.11-7.04 (m, 2H), 6.66 (dd, J = 5.7, 2.4, 1H), 5.43 (s, 2H), 4.72 (s, 2H), 3.86 (s, 3H). ESI-MS: 449.05 (M+H)+.
3-[(3,4-difluorophenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2H- 1,3-benzoxazin-4-one (156): Compound 156 was synthesized from intermediate 133 (0.12 mmol) as a white solid in 29% yield according to the general method P2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.52 (d, J = 2.9, 1H), 7.47-7.39 (m, 3H), 7.26-7.19 (m, 3H), 6.66 (dd, J = 5.7, 2.3, 1H), 5.40 (s, 2H), 4.68 (s, 2H), 3.86 (s, 3H). ESI-MS: 449.00 (M+H)+. 3-[(4-chloro-3-fluoro-phenyl)methyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}- 2H-1,3-benzoxazin-4-one (157): Compound 157 was synthesized from intermediate 134 (0.12 mmol) as a white solid in 33% yield according to the general method P2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.59 (t, J = 8.0, 1H), 7.52 (d, J = 2.9, 1H), 7.43-7.38 (m, 2H), 7.26-7.19 (m, 3H), 6.66 (dd, J = 5.7, 2.4, 1H), 5.41 (s, 2H), 4.71 (s, 2H), 3.86 (s, 3H). ESI-MS: 465.05 (M+H)+. 3-(cyclohexylmethyl)-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2H-1,3- benzoxazin-4-one (158): Compound 158 was synthesized from intermediate 135 (0.11 mmol) as a white solid in 42% yield according to the general method P2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.37 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.47 (d, J =
3.0, 1H), 7.38 (dd, J = 8.8, 3.0, 1H), 7.25 (d, J = 2.3, 1H), 7.18 (d, J = 8.8, 1H), 6.65 (dd, J = 5.7, 2.4, 1H), 5.33 (s, 2H), 3.86 (s, 3H), 1.68-1.60 (m, 6H), 1.23- 1.08 (m, 3H), 0.97-0.89 (m, 2H). ESI-MS: 419.10 (M+H)+. 3-[2-(3-fluorophenyl)ethyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2H-1,3- benzoxazin-4-one (159): Compound 159 was synthesized from intermediate 136 (0.11 mmol) as a white solid in 27% yield according to the general method P2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.37 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.46 (d, J = 2.8, 1H), 7.40-7.31 (m, 2H), 7.24 (s, 1H), 7.19-7.10 (m, 3H), 7.04 (t, J = 8.8, 1H), 6.65-6.64 (m, 1H), 5.30 (s, 2H), 3.86 (s, 3H), 3.71 (t, J = 7.2, 2H), 2.90 (t, J = 7.2, 2H). ESI-MS: 445.15 (M+H)+. 3-[2-(4-fluorophenyl)ethyl]-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2H-1,3- benzoxazin-4-one (160): Compound 160 was synthesized from intermediate 137 (0.14 mmol) as a white solid in 45% yield according to the general method P2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.46 (d, J = 2.7, 1H), 7.38 (dd, J = 8.8, 3.0, 1H), 7.31 (dd, J = 8.1, 5.8, 2H), 7.24 (d, J = 1.9, 1H), 7.18-7.09 (m, 3H), 6.64 (dd, J = 5.7, 2.3, 1H), 5.29 (s, 2H), 3.86 (s, 3H), 3.68 (t, J = 7.4, 2H), 2.87 (t, J = 7.4, 2H). ESI-MS: 445.10 (M+H)+.
6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-[2-(2-pyridyl)ethyl]-2H-1,3- benzoxazin-4-one (161): Compound 161 was synthesized from intermediate 138 (0.16 mmol) as a white solid in 34% yield according to the general method qP2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.50 (d, J = 4.8, 1H), 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.71 (td, J = 7.7, 1.8, 1H), 7.46 (d, J = 3.0, 1H), 7.37 (dd, J = 8.8, 3.0, 1H), 7.32 (d, J = 7.7, 1H), 7.25-7.21 (m, 2H), 7.16 (d, J = 8.8, 1H), 6.65 (dd, J = 5.7, 2.4, 1H), 5.29 (s, 2H), 3.86-3.82 (m, 5H), 3.05 (t, J = 7.3, 2H). ESI-MS: 428.05 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-[2-(3-pyridyl)ethyl]-2H-1,3- benzoxazin-4-one (162): Compound 162 was synthesized from intermediate 139 (0.14 mmol) as a white solid in 30% yield according to the general method P2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.48 (d, J = 1.9, 1H), 8.42 (dd, J = 4.8, 1.6, 1H), 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.70 (dt, J = 7.8, 1.9, 1H), 7.45 (d, J = 3.0, 1H), 7.38 (dd, J = 8.8, 3.0, 1H), 7.32 (dd, J = 7.6, 4.6, 1H), 7.24 (d, J = 2.4, 1H), 7.18 (d, J = 8.8, 1H), 6.64 (dd, J = 5.7, 2.4, 1H), 5.32 (s, 2H), 3.86 (s, 3H), 3.72 (t, J = 7.3, 2H), 2.91 (t, J = 7.3, 2H). ESI-MS: 428.05 (M+H)+.
6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-(2-phenylethyl)-2H-1,3- benzoxazin-4-one (163): Compound 163 was synthesized from intermediate 140 (0.18 mmol) as a white solid in 22% yield according to the general method P2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.47 (d, J = 3.0, 1H), 7.38 (dd, J = 8.8, 3.0, 1H), 7.32-7.16 (m, 7H), 6.65 (dd, J = 5.7, 2.4, 1H), 5.28 (s, 2H), 3.86 (s, 3H), 3.70 (t, J = 7.3, 2H), 2.88 (t, J = 7.3, 2H). ESI-MS: 427.10 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-(2-pyrrolidin-1-ylethyl)-2H-1,3- benzoxazin-4-one (164): Compound 164 was synthesized from intermediate 141 (0.09 mmol) as a white solid in 21% yield according to the general method P2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.37 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.47 (d, J = 2.9, 1H), 7.38 (dd, J = 8.8, 3.0, 1H), 7.24 (d, J = 2.3, 1H), 7.18 (d, J = 8.8, 1H), 6.65 (dd, J = 5.7, 2.4, 1H), 5.38 (s, 2H), 3.86 (s, 3H), 3.59 (t, J = 6.4, 2H), 2.59 (t, J = 6.4, 2H), 1.66 (s, 4H). ESI-MS: 420.15 (M+H)+. 3-(2-cyclohexylethyl)-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2H-1,3- benzoxazin-4-one (165): Compound 165 was synthesized from intermediate 142 (0.11 mmol) as a white solid in 48% yield according to the general method P2. 1H NMR (400 MHz,
DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.6, 1H), 7.47 (d, J = 2.9, 1H), 7.38 (dd, J = 8.8, 3.0, 1H), 7.24 (d, J = 2.3, 1H), 7.18 (d, J = 8.8, 1H), 6.65 (dd, J = 5.7, 2.4, 1H), 5.34 (s, 2H), 3.86 (s, 3H), 3.50-3.46 (m, 2H), 1.74-1.57 (m, 5H), 1.49-1.41 (m, 2H), 1.30-1.10 (m, 4H), 0.95-0.85 (m, 2H). ESI-MS: 433.15 (M+H)+. 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3-[2-(1-piperidyl)ethyl]-2H-1,3- benzoxazin-4-one (166): Compound 166 was synthesized from intermediate 143 (0.14 mmol) as a white solid in 23% yield according to the general method P2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.47 (d, J = 3.0, 1H), 7.38 (dd, J = 8.8, 3.0, 1H), 7.24 (d, J = 2.3, 1H), 7.18 (d, J = 8.8, 1H), 6.65 (dd, J = 5.7, 2.4, 1H), 5.38 (s, 2H), 3.86 (s, 3H), 3.58 (t, J = 6.3, 2H), 2.44- 2.36 (m, 6H), 1.49-1.4 (m, 4H), 1.39-1.34 (m, 2H). ESI-MS: 434.15 (M+H)+. 3-(cyclopentylmethyl)-6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2H-1,3- benzoxazin-4-one (167): Compound 167 was synthesized from intermediate 144 (0.11 mmol) as a white solid in 52% yield according to the general method P2. 1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.6, 1H), 7.48 (d, J = 2.9, 1H), 7.38 (dd, J = 8.8, 3.0, 1H), 7.25 (d, J = 2.2, 1H), 7.18 (d, J = 8.8, 1H), 6.65 (dd, J = 5.7, 2.4, 1H), 5.36 (s, 2H), 3.86 (s, 3H), 3.40 (d, J = 7.6, 2H), 2.24-2.14 (m, 1H), 1.71-1.48 (m, 6H), 1.26-1.17 (m, 2H). ESI-MS: 405.15 (M+H)+.
Example 6: General procedure for the preparation of analogues 171-175 Method Q: To a solution of 2,5-dihydroxybenzamide (1.074 g, 6.78 mmol) in DMF (20 mL) under nitrogen was added solid potassium carbonate (1.407 g, 10.16 mmol) followed by 2-chloro-4-nitropyridine derivative (1.037 g, 6.78 mmol). The reaction mixture was stirred at room temperature overnight. The reaction mixture was diluted with EtOAc and washed twice with a saturated solution of NH4Cl. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. The crude was purified by flash column chromatography (DCM/EtOAc from100/0 to 60/40) to give 915 mg of 5-[(2- chloro-4-pyridyl)oxy]-2-hydroxy-benzamide 168 as a beige powder in 51% yield. ESI-MS: 264.85 (M+H)+. Preparation of 2-hydroxy-5-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}benzamide (169): Intermediate 169 was synthesized from intermediate 168 (4.19 mmol) and 1- methyl-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole (6.29 mmol) as a beige powder in 78% yield according to the general method B. ESI-MS: 310.90 (M+H)+.
Method R: To a solution of intermediate 169 (150 mg, 0.48 mmol) in 2,2- dimethoxypropane (3 mL) under nitrogen was added p-toluenesulfonic acid (92 mg, 0.48 mmol). The reaction mixture was stirred at 80°C overnight. The reaction mixture was diluted with EtOAc and washed twice with a saturated solution of NaHCO3. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. The crude was purified by flash column chromatography (DCM/MeOH from100/0 to 94/6) to give 127 mg of 2,2-dimethyl- 6-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3H-1,3-benzoxazin-4-one 170 as a yellow powder in 75% yield. ESI-MS: 351.00 (M+H)+. Method S: To a solution of derivative 170 (1 equiv.) in THF (10 mL/mmol) under nitrogen was added NaH (60% in oil, 1.5 equiv.) and the reaction mixture was stirred at room temperature for 30 minutes. Appropriate benzyl bromide was then added (1.2-1.7 equiv.) and the reaction mixture was stirred at room temperature overnight. The reaction mixture was diluted with EtOAc and washed twice with a saturated solution of NH4Cl. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. The crude product was purified by flash column chromatography (DCM/MeOH from 100/0 to 95/5) and reverse phase chromatography (H2O/MeOH: 0 to 100%) to give the expected compound. The following compounds are examples illustrating Method S: 3-[(3-fluorophenyl)methyl]-2,2-dimethyl-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}-1,3-benzoxazin-4-one (171): Compound 171 was synthesized from intermediate 170 (0.09 mmol) and 3- fluorobenzyl bromide (0.15 mmol) as a white solid in 36% yield according to the general method S.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.39 (d, J = 5.7, 1H), 8.28 (s, 1H), 7.99 (d, J = 0.7, 1H), 7.54 (d, J = 3.0, 1H), 7.43-7.37 (m, 2H), 7.29 (d, J = 2.1, 1H), 7.18-7.06 (m, 4H), 6.70 (dd, J = 5.7, 2.4, 1H), 4.80 (s, 2H), 3.87 (s, 3H), 1.59 (s, 6H). ESI-MS: 459.20 (M+H)+.
3-[(4-fluorophenyl)methyl]-2,2-dimethyl-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}-1,3-benzoxazin-4-one (172): Compound 172 was synthesized from intermediate 170 (0.09 mmol) and 4- fluorobenzyl bromide (0.15 mmol) as a white solid in 28% yield according to the general method S.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.39 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.6, 1H), 7.53 (d, J = 3.0, 1H), 7.41 (dd, J = 8.8, 3.0, 1H), 7.38-7.33 (m, 2H), 7.28 (d, J = 2.3, 1H), 7.20-7.11 (m, 3H), 6.68 (dd, J = 5.7, 2.4, 1H), 4.77 (s, 2H), 3.87 (s, 3H), 1.57 (s, 6H). ESI-MS: 459.05 (M+H)+. 3-[(3-chlorophenyl)methyl]-2,2-dimethyl-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}-1,3-benzoxazin-4-one (173): Compound 173 was synthesized from intermediate 170 (0.05 mmol) and 3- chlorobenzyl bromide (0.08 mmol) as a white solid in 27% yield according to the general method S.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.53 (d, J = 3.0, 1H), 7.41 (dd, J = 8.8, 3.0, 1H), 7.39-7.31 (m, 3H), 7.30-7.26 (m, 2H), 7.13 (d, J = 8.8, 1H), 6.69 (dd, J = 5.7, 2.4, 1H), 4.78 (s, 2H), 3.86 (s, 3H), 1.58 (s, 6H). ESI-MS: 475.05 (M+H)+.
3-[(4-chlorophenyl)methyl]-2,2-dimethyl-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}-1,3-benzoxazin-4-one (174): Compound 174 was synthesized from intermediate 170 (0.09 mmol) and 4- chlorobenzyl bromide (0.15 mmol) as a white solid in 34% yield according to the general method S.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.52 (d, J = 3.0, 1H), 7.43-7.39 (m, 3H), 7.36- 7.32 (m, 2H), 7.28 (d, J = 2.2, 1H), 7.13 (d, J = 8.8, 1H), 6.68 (dd, J = 5.7, 2.4, 1H), 4.77 (s, 2H), 3.87 (s, 3H), 1.57 (s, 6H). ESI-MS: 475.05 (M+H)+. 3-[(3,5-difluorophenyl)methyl]-2,2-dimethyl-6-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}-1,3-benzoxazin-4-one (175): Compound 175 was synthesized from intermediate 170 (0.09 mmol) and 3,5- difluorobenzyl bromide (0.15 mmol) as a white solid in 10% yield according to the general method S.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.6, 1H), 7.53 (d, J = 3.0, 1H), 7.44-7.32 (m, 3H), 7.28 (d, J = 2.3, 1H), 7.20-7.11 (m, 2H), 6.69 (dd, J = 5.7, 2.4, 1H), 4.76 (s, 2H), 3.87 (s, 3H), 1.58 (s, 6H). ESI-MS: 477.20 (M+H)+.
Example 7: General procedure for the preparation of analogues 178-194 Method T: To a solution of derivative 169 (250 mg, 0.81 mmol) in DMF (13 mL) under nitrogen was added potassium carbonate (1.113 g, 8.05 mmol) and 1,2- dibromoethane (0.555 mL, 6.45 mmol). The reaction mixture was stirred at room temperature overnight. The reaction mixture was diluted with EtOAc and washed twice with a saturated solution of NH4Cl. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. The crude was purified by flash column chromatography (DCM/MeOH from100/0 to 93/7) to give 228 mg of 2-(2-bromoethoxy)-5-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}benzamide 176 as a white powder in 68% yield. ESI-MS: 416.95-418.90 (M+H)+. Method U: To a suspension of NaH (60% in oil, 24 mg, 0.60 mmol) in DMF (2.5 mL) under nitrogen was slowly added a solution of derivative 176 (228 mg, 0.55 mmol) in DMF (2.5 mL). The resulting solution was stirred at room temperature for 2h. The reaction mixture was diluted with EtOAc and washed twice with a saturated solution of NH4Cl. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. The crude was purified by flash column chromatography (DCM/MeOH from100/0 to 93/7) to give 111 mg of 7- {[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-3,4-dihydro-2H-1,4-benzoxazepin-5- one 177 as a white powder in 60% yield.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.46 (t, J = 5.2, 1H), 8.37 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.48 (d, J = 3.1, 1H), 7.32 (dd, J = 8.8, 3.1, 1H), 7.25 (d, J = 2.3, 1H), 7.13 (d, J = 8.8, 1H),
6.64 (dd, J = 5.7, 2.4, 1H), 4.30-4.33 (m, 2H), 3.86 (s, 3H), 3.38-3.35 (m, 2H). ESI-MS: 336.95 (M+H)+. The following compounds are examples illustrating Method S: 4-[(3-fluorophenyl)methyl]-7-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2,3- dihydro-1,4-benzoxazepin-5-one (178): Compound 178 was synthesized from intermediate 177 (0.06 mmol) and 3- fluorobenzyl bromide (0.07 mmol) as a white solid in 76% yield according to the general method S.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.6, 1H), 7.43-7.38 (m, 2H), 7.33 (dd, J = 8.7, 3.1, 1H), 7.27 (d, J = 2.3, 1H), 7.21-7.09 (m, 4H), 6.67 (dd, J = 5.7, 2.4, 1H), 4.77 (s, 2H), 4.26 (t, J = 5.1, 2H), 3.86 (s, 3H), 3.62 (t, J = 5.1, 2H). ESI-MS: 445.00 (M+H)+. 4-[(4-fluorophenyl)methyl]-7-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2,3- dihydro-1,4-benzoxazepin-5-one (179): Compound 179 was synthesized from intermediate 177 (0.07 mmol) and 4- fluorobenzyl bromide (0.07 mmol) as a white solid in 34% yield according to the general method S.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.7, 1H), 7.43-7.37 (m, 3H), 7.33 (dd, J = 8.7, 3.1, 1H), 7.26 (d, J = 2.1, 1H), 7.22-7.12 (m, 3H), 6.67 (dd, J = 5.7, 2.4, 1H), 4.73 (s, 2H), 4.23 (t, J = 5.1, 2H), 3.86 (s, 3H), 3.59 (t, J = 5.1, 2H). ESI-MS: 445.05 (M+H)+.
4-[(3-chlorophenyl)methyl]-7-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2,3- dihydro-1,4-benzoxazepin-5-one (180): Compound 180 was synthesized from intermediate 177 (0.06 mmol) and 3- chlorobenzyl bromide (0.07 mmol) as a white solid in 84% yield according to the general method S.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.43-7.41 (m, 2H), 7.40-7.31 (m, 4H), 7.27 (d, J = 2.3, 1H), 7.15 (d, J = 8.7, 1H), 6.68 (dd, J = 5.7, 2.4, 1H), 4.76 (s, 2H), 4.26 (t, J = 5.1, 2H), 3.86 (s, 3H), 3.63 (t, J = 5.1, 2H). ESI-MS: 461.05 (M+H)+. 4-[(4-chlorophenyl)methyl]-7-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2,3- dihydro-1,4-benzoxazepin-5-one (181): N O N O N Cl N O Compound 181 was synthesized from intermediate 177 (0.06 mmol) and 4- chlorobenzyl bromide (0.07 mmol) as a white solid in 58% yield according to the general method S.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.44-7.36 (m, 5H), 7.33 (dd, J = 8.7, 3.1, 1H), 7.27 (d, J = 2.3, 1H), 7.14 (d, J = 8.7, 1H), 6.67 (dd, J = 5.7, 2.4, 1H), 4.74 (s, 2H), 4.24 (t, J = 5.1, 2H), 3.87 (s, 3H), 3.60 (t, J = 5.1, 2H). ESI-MS: 461.05 (M+H)+. 4-[(3,5-difluorophenyl)methyl]-7-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2,3- dihydro-1,4-benzoxazepin-5-one (182): Compound 182 was synthesized from intermediate 177 (0.06 mmol) and 3,5-
difluorobenzyl bromide (0.07 mmol) as a white solid in 73% yield according to the general method S.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.6, 1H), 7.44 (d, J = 3.0, 1H), 7.34 (dd, J = 8.7, 3.1, 1H), 7.27 (d, J = 2.2, 1H), 7.19-7.13 (m, 2H), 7.10-7.04 (m, 2H), 6.67 (dd, J = 5.7, 2.4, 1H), 4.77 (s, 2H), 4.29 (t, J = 5.1, 2H), 3.86 (s, 3H), 3.65 (t, J = 5.1, 2H). ESI-MS: 463.05 (M+H)+. 7-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-4-{[3-(trifluoromethyl)phenyl]methyl}- 2,3-dihydro-1,4-benzoxazepin-5-one (183): Compound 183 was synthesized from intermediate 177 (0.06 mmol) and 1- (bromomethyl)-3-(trifluoromethyl)benzene (0.07 mmol) as a white solid in 52% yield according to the general method S.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (d, J = 0.6, 1H), 7.72 (s, 1H), 7.68-7.58 (m, 3H), 7.42 (d, J = 3.0, 1H), 7.34 (dd, J = 8.7, 3.1, 1H), 7.27 (d, J = 2.3, 1H), 7.16 (d, J = 8.7, 1H), 6.67 (dd, J = 5.7, 2.4, 1H), 4.85 (s, 2H), 4.25 (t, J = 5.1, 2H), 3.86 (s, 3H), 3.65 (t, J = 5.1, 2H). ESI-MS: 495.10 (M+H)+. 4-[(3,4-difluorophenyl)methyl]-7-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2,3- dihydro-1,4-benzoxazepin-5-one (184): Compound 184 was synthesized from intermediate 177 (0.06 mmol) and 3,4- difluorobenzyl bromide (0.07 mmol) as a white solid in 85% yield according to the general method S.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.46-7.38 (m, 3H), 7.34 (dd, J = 8.7, 3.1, 1H), 7.26 (d, J = 2.2, 1H), 7.24-7.19 (m, 1H), 7.15 (d, J = 8.7, 1H), 6.67 (dd, J = 5.7,
2.4, 1H), 4.73 (s, 2H), 4.26 (t, J = 5.1, 2H), 3.86 (s, 3H), 3.62 (t, J = 5.1, 2H). ESI-MS: 463.00 (M+H)+. 4-[(4-chloro-3-fluoro-phenyl)methyl]-7-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}- 2,3-dihydro-1,4-benzoxazepin-5-one (185): Compound 185 was synthesized from intermediate 177 (0.06 mmol) and 4- (bromomethyl)-1-chloro-2-fluorobenzene (0.07 mmol) as a white solid in 96% yield according to the general method S.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.38 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.97 (s, 1H), 7.58 (t, J = 8.0, 1H), 7.43 (d, J = 3.0, 1H), 7.39 (dd, J = 10.3, 1.7, 1H), 7.34 (dd, J = 8.7, 3.1, 1H), 7.27 (d, J = 2.3, 1H), 7.23 (dd, J = 8.3, 1.4, 1H), 7.15 (d, J = 8.7, 1H), 6.67 (dd, J = 5.7, 2.4, 1H), 4.75 (s, 2H), 4.27 (t, J = 5.1, 2H), 3.86 (s, 3H), 3.63 (t, J = 5.1, 2H). ESI-MS: 479.00 (M+H)+. Method V: To a solution of 177 (20 mg, 0.06 mmol) in dioxane (1 mL) were added bromo or iodo derivative (1.1-2 equiv.), CuI (0.5 equiv.), DMEDA (0.7 equiv.) and K2CO3 (6 equiv.). The mixture was stirred at 120°C until completion. The reaction mixture was concentrated under reduced pressure. The crude was purified by flash column chromatography (DCM/MeOH from 100/0 to 95/5) and reverse phase chromatography (H2O/MeOH from 100/0 to 0/100) to give the expected compound.
The following compounds are examples illustrating Method V: 7-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-4-phenyl-2,3-dihydro-1,4- benzoxazepin-5-one (186): Compound 186 was synthesized from intermediate 177 (0.06 mmol) and iodobenzene (0.07 mmol) as a white solid in 41% yield according to the general method V.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.39 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.47-7.36 (m, 6H), 7.33-7.27 (m, 2H), 7.21 (d, J = 8.7, 1H), 6.69 (dd, J = 5.7, 2.4, 1H), 4.47 (t, J = 5.1, 2H), 3.99 (t, J = 5.1, 2H), 3.87 (s, 3H). ESI-MS: 413.05 (M+H)+. 4-(3-fluorophenyl)-7-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2,3-dihydro-1,4- benzoxazepin-5-one (187): Compound 187 was synthesized from intermediate 177 (0.06 mmol) and 1- fluoro-3-iodobenzene (0.09 mmol) as a white solid in 68% yield according to the general method V.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.39 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.6, 1H), 7.51-7.34 (m, 4H), 7.31-7.28 (m, 1H), 7.28 (d, J = 2.3, 1H), 7.21 (d, J = 8.7, 1H), 7.18-7.12 (m, 1H), 6.69 (dd, J = 5.7, 2.4, 1H), 4.49 (t, J = 5.1, 2H), 4.01 (t, J = 5.1, 2H), 3.87 (s, 3H). ESI-MS: 431.05 (M+H)+.
4-(4-fluorophenyl)-7-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2,3-dihydro-1,4- benzoxazepin-5-one (188): Compound 188 was synthesized from intermediate 177 (0.06 mmol) and 1- fluoro-4-iodobenzene (0.09 mmol) as a white solid in 92% yield according to the general method V.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.39 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.98 (d, J = 0.6, 1H), 7.48-7.36 (m, 4H), 7.30-7.24 (m, 3H), 7.21 (d, J = 8.7, 1H), 6.68 (dd, J = 5.7, 2.4, 1H), 4.47 (t, J = 5.1, 2H), 3.96 (t, J = 5.1, 2H), 3.86 (s, 3H). ESI-MS: 431.00 (M+H)+. 4-(3-chlorophenyl)-7-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2,3-dihydro-1,4- benzoxazepin-5-one (189): Compound 189 was synthesized from intermediate 177 (0.06 mmol) and 1- chloro-3-iodobenzene (0.09 mmol) as a white solid in 42% yield according to the general method V.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.39 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.56 (t, J = 1.9, 1H), 7.50-7.36 (m, 5H), 7.28 (d, J = 2.3, 1H), 7.21 (d, J = 8.7, 1H), 6.69 (dd, J = 5.7, 2.4, 1H), 4.49 (t, J = 5.1, 2H), 4.00 (t, J = 5.1, 2H), 3.87 (s, 3H). ESI-MS: 447.00 (M+H)+.
4-(4-chlorophenyl)-7-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2,3-dihydro-1,4- benzoxazepin-5-one (190): Compound 190 was synthesized from intermediate 177 (0.06 mmol) and 1- chloro-4-iodobenzene (0.09 mmol) as a white solid in 65% yield according to the general method V.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.39 (d, J = 5.7, 1H), 8.26 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.51-7.44 (m, 4H), 7.42-7.37 (m, 2H), 7.27 (d, J = 2.1, 1H), 7.21 (d, J = 8.7, 1H), 6.69 (dd, J = 5.7, 2.4, 1H), 4.47 (t, J = 5.1, 2H), 3.99 (t, J = 5.1, 2H), 3.86 (s, 3H). ESI-MS: 447.00 (M+H)+. 7-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-4-[5-(trifluoromethyl)-3-pyridyl]-2,3- dihydro-1,4-benzoxazepin-5-one (191): Compound 191 was synthesized from intermediate 177 (0.06 mmol) and 3- bromo-5-(trifluoromethyl)pyridine (0.07 mmol) as a white solid in 70% yield according to the general method V.1H NMR (400 MHz, DMSO-d6) δ (ppm): 9.00 (d, J = 2.2, 1H), 8.90 (d, J = 0.8, 1H), 8.39 (d, J = 5.7, 1H), 8.35 (bs, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.47 (d, J = 3.0, 1H), 7.43 (dd, J = 8.7, 3.1, 1H), 7.28 (d, J = 2.3, 1H), 7.24 (d, J = 8.7, 1H), 6.69 (dd, J = 5.7, 2.4, 1H), 4.56 (t, J = 5.0, 2H), 4.12 (t, J = 5.0, 2H), 3.87 (s, 3H). ESI-MS: 482.05 (M+H)+.
4-(5-fluoropyrimidin-2-yl)-7-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-2,3- dihydro-1,4-benzoxazepin-5-one (192): Compound 192 was synthesized from intermediate 177 (0.06 mmol) and 2- bromo-5-fluoropyrimidine (0.07 mmol) as a white solid in 16% yield according to the general method V.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.95 (s, 2H), 8.39 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.44-7.41 (m, 2H), 7.28-7.23 (m, 2H), 6.72 (dd, J = 5.7, 2.4, 1H), 4.41 (t, J = 5.2, 2H), 4.26 (t, J = 5.1, 2H), 3.87 (s, 3H). ESI-MS: 433.00 (M+H)+. 7-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-4-[4-(trifluoromethyl)-2-pyridyl]-2,3- dihydro-1,4-benzoxazepin-5-one (193): Compound 193 was synthesized from intermediate 177 (0.06 mmol) and 2- bromo-4-(trifluoromethyl)pyridine (0.07 mmol) as a white solid in 61% yield according to the general method V.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.76 (d, J = 5.2, 1H), 8.45 (d, J = 0.7, 1H), 8.40 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (d, J = 0.7, 1H), 7.65 (dd, J = 5.1, 0.9, 1H), 7.48-7.42 (m, 2H), 7.29 (d, J = 2.1, 1H), 7.24 (d, J = 8.8, 1H), 6.73 (dd, J = 5.7, 2.4, 1H), 4.50-4.42 (m, 4H), 3.87 (s, 3H). ESI-MS: 482.05 (M+H)+.
7-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-4-[6-(trifluoromethyl)-2-pyridyl]-2,3- dihydro-1,4-benzoxazepin-5-one (194): Compound 194 was synthesized from intermediate 177 (0.06 mmol) and 2- bromo-6-trifluoromethylpyridine (0.07 mmol) as a white solid in 57% yield according to the general method V.1H NMR (400 MHz, DMSO-d6) δ (ppm): 8.39 (d, J = 5.6, 1H), 8.34 (d, J = 8.5, 1H), 8.27 (s, 1H), 8.19-8.14 (m, 1H), 7.98 (d, J = 0.7, 1H), 7.77 (d, J = 7.2, 1H), 7.46-7.42 (m, 2H), 7.29-7.28 (m, 1H), 7.25 (dd, J = 8.4, 0.7, 1H), 6.73 (dd, J = 5.7, 2.4, 1H), 4.46 (t, J = 5.0, 2H), 4.39 (t, J = 5.0, 2H), 3.87 (s, 3H). ESI-MS: 482.05 (M+H)+. Example 8: General procedure for the preparation of analogues 202-205 Cs2CO3, DMF 100°C Method A Pd(PPh3)4, Cs2CO31M, dioxane, 100°C, 1h Method B 195 196 NaOH 2N, MeOH, 50°C, 2h Method J RNH2, DBU, DMF EDC.HCl, 150°C HOBt, DMF, rt, Method W Method O 202-205 198-201 197
Preparation of methyl 5-[(2-chloro-4-pyridyl)oxy]-2-fluoro-benzoate (195): Intermediate 195 was synthesized from 2-chloro-4-nitropyridine (1.24 mmol) and methyl 2-fluoro-5-hydroxybenzoate (1.24 mmol) as a white powder in 88% yield according to the general method A. ESI-MS: 281.85 (M+H)+. Preparation of ethyl 2-fluoro-5-{[2-(1-methylpyrazol-4-yl)-4- pyridyl]oxy}benzoate (196): Intermediate 196 was synthesized from intermediate 195 (1.10 mmol) and 1- methyl-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole (1.32 mmol) as a white powder in quantitative yield according to the general method B. ESI-MS: 327.95 (M+H)+. Preparation of 2-fluoro-5-[[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy]benzoic acid (197): Intermediate 197 was synthesized from intermediate 196 (1.10 mmol) and NaOH (3.00 mmol) as a white powder in 89% yield according to the general method J. ESI-MS: 313.90 (M+H)+.
The following table illustrates intermediates 198-201 prepared from method O: Intermediate Structure Synthesis procedure Compound 198 Method O Compound 199 Method O Compound 200 Method O Compound 201 Method O Method W: To a solution of appropriate intermediate 198-201 (90 mg, 0.21 mmol) in DMF (3 mL) was added DBU (2.5 equiv.) and the mixture was stirred at 150°C for 2h. After cooling at room temperature, a saturated aqueous solution of NH4Cl was added and the aqueous layer was extracted with EtOAc. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. The crude was purified by flash column chromatography (DCM/MeOH from 100/0 to 93/7) and reverse phase chromatography (H2O/MeOH from 100/0 to 0/100) to give the expected compound.
The following compounds are examples illustrating Method W: 3-fluoro-8-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-5H- benzo[b][1,4]benzoxazepin-6-one (202): Compound 202 was synthesized from intermediate 198 (0.21 mmol) DBU (0.53 mmol) as a white solid in 44% yield according to the general method W.1H NMR (400 MHz, DMSO-d6) δ (ppm): 10.72 (bs, 1H), 8.38 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.47-7.40 (m, 4H), 7.30 (d, J = 2.3, 1H), 7.03-6.96 (m, 2H), 6.69 (dd, J = 5.7, 2.4, 1H), 3.85 (s, 3H). ESI-MS: 402.95 (M+H)+. 2-fluoro-8-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-5H- benzo[b][1,4]benzoxazepin-6-one (203): Compound 203 was synthesized from intermediate 199 (0.23 mmol) DBU (0.59 mmol) as a white solid in 74% yield according to the general method W.1H NMR (400 MHz, DMSO-d6) δ (ppm): 10.64 (bs, 1H), 8.38 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.50-7.44 (m, 3H), 7.36 (dd, J = 9.0, 2.8, 1H), 7.31 (d, J = 2.3, 1H), 7.20 (dd, J = 8.9, 5.9, 1H), 7.11 (td, J = 8.5, 2.8, 1H), 6.70 (dd, J = 5.7, 2.4, 1H), 3.85 (s, 3H). ESI-MS: 403.05 (M+H)+.
3-chloro-8-[[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy]-5H- benzo[b][1,4]benzoxazepin-6-one (204): Compound 204 was synthesized from intermediate 200 (0.10 mmol) DBU (0.24 mmol) as a white solid in 43% yield according to the general method W.1H NMR (400 MHz, DMSO-d6) δ (ppm): 10.73 (bs, 1H), 8.38 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.47-7.40 (m, 4H), 7.30 (d, J = 2.3, 1H), 7.23-7.21 (m, 2H), 6.70 (dd, J = 5.7, 2.4, 1H), 3.85 (s, 3H). ESI-MS: 419.00 (M+H)+. 2-chloro-8-{[2-(1-methylpyrazol-4-yl)-4-pyridyl]oxy}-5H- benzo[b][1,4]benzoxazepin-6-one (205): Compound 205 was synthesized from intermediate 201 (0.07 mmol) DBU (0.17 mmol) as a white solid in 68% yield according to the general method W.1H NMR (400 MHz, DMSO-d6) δ (ppm): 10.73 (bs, 1H), 8.38 (d, J = 5.7, 1H), 8.27 (s, 1H), 7.98 (s, 1H), 7.56 (d, J = 2.3, 1H), 7.49-7.44 (m, 3H), 7.32-7.28 (m, 2H), 7.18 (d, J = 8.6, 1H), 6.70 (dd, J = 5.7, 2.4, 1H), 3.85 (s, 3H). ESI-MS: 419.00 (M+H)+.
Table 1: Examples of compounds according to the invention
Example 9: Cell-based assays: Biological assay measuring cell proliferation in cell lines Compounds were evaluated in different cancer cell lines (Molm-13, M-NFS-60, HL-60 and P815) and in PDGFRα-BaF3 stable cell line. For each, cell proliferation were measured. The protocols of these assays are described below. MOLM-13: Exponential growing MOLM-13 cells (DSMZ, ACC-554) were seeded at 2.10^4 per 200 μL of complete medium.20 μL of test compound dilution were added to each well and the plates were incubated for 72 h at 37 °C, 5% CO2.
Untreated cells and positive control (0,5% triton X-100, for the last 15 min) served as reference for maximum and minimum viability. At the end of incubation 100 μL of supernatant were removed and replaced by 10 μL of WST-1 solution (Cell Proliferation Reagent WST-1, Roche Applied Science). After 3 h incubation at 37 °C, 5% CO2, optical densities were measured at 450 nm and 620 nm for the background on microplate reader (Envision 2105, Perkinelmer). M-NFS-60: Exponential growing M-NFS-60 cells (ATCC, CRL-1838) were seeded at 10^4 per 200 μL of complete medium with beta-mercaptoethanol and M-CSF (62 ng/mL) or IL34 (500 ng/mL). Twenty μL of test compound dilution were added to each well and the plates were incubated for 72 h at 37 °C, 5% CO2. Untreated cells and positive control (0.5% triton X-100, for the last 15 min) served as reference for maximum and minimum viability. At the end of incubation 100 μL of supernatant were removed and replaced by 10 μL of WST-1 solution (Cell Proliferation Reagent WST-1, Roche Applied Science). After 3 h incubation at 37 °C, 5% CO2, optical densities were measured at 450 nm and 620 nm for the background on microplate reader (Envision 2105, Perkinelmer). HL-60: Exponential growing HL-60 cells (DSMZ, ACC-3) were seeded at 2.10^4 per 200 μL of complete RPMI medium.20 μL of test compound dilution were added to each well and the plates were incubated for 72 h at 37 °C, 5% CO2. Untreated cells and positive control (0.5% triton X-100, for the last 15 min) served as reference for maximum and minimum viability. At the end of incubation 100 μL of supernatant were removed and replaced by 10 μL of WST-1 solution (Cell Proliferation Reagent WST-1, Roche Applied Science). After 3 h incubation at 37 °C, 5% CO2, optical densities were measured at 450 nm and 620 nm for the background on microplate reader (Envision 2105, Perkinelmer). IC50 were measured and some biological results of these assays are presented in the following table. P-815: Exponential growing P-815 cells (DSMZ, ACC-1) were seeded at 2.10^4 per 200 μL of complete RPMI medium. Twenty μL of test compound dilution were added to each well and the plates were incubated for 72 h at 37 °C, 5% CO2. Untreated cells and positive control (0,5% triton X-100, for the last 15 min) served as reference for maximum and minimum viability. At the end of incubation 100 μL of supernatant were removed and replaced by 10 μL of WST- 1 solution (Cell Proliferation Reagent WST-1, Roche Applied Science). After 3 h
incubation at 37 °C, 5% CO2, optical densities were measured at 450 nm and 620 nm for the background on microplate reader (Envision 2105, Perkinelmer). BaF3-PDGFRα: Exponential growing BaF3 cells stably transfected with a plasmid encoding the fusion gene GFP-ETV6-PDGFRA (ABMGood, T3082) were seeded at 5.10^3 per 200 μL of complete RPMI medium. Twenty μL of test compound dilution were added to each well and the plates were incubated for 72 h at 37 °C, 5% CO2. Untreated cells and positive control (0,5% triton X-100, for the last 15 min) served as reference for maximum and minimum viability. At the end of incubation 100 μL of supernatant were removed and replaced by 10 μL of WST-1 solution (Cell Proliferation Reagent WST-1, Roche Applied Science). After 3 h incubation at 37 °C, 5% CO2, optical densities were measured at 450 nm and 620 nm for the background on microplate reader (Envision 2105, Perkinelmer). IC50 were measured and some biological results of these assays are presented in the following table.
Table 2: Results biological cell-based assay measuring cell proliferation in cell lines
Example 10: Cell-based assays: Biological assay measuring cell proliferation in non-cancer cell lines CSF1R receptor has been expressed in HEK cell lines following the protocols below. HEK-CSF1R-STAT5-Luc: Exponential growing HEK293T cells (ATCC® CRL- 3216™), ectopically expressing human CSF1R receptor (Origene) and five copies of a STAT5 response element (STAT5 RE, promega) that drives transcription of the luciferase reporter were seeded at 5.10^3 per 20 μL of complete DMEM medium. The next day, 2.25 μL of test compound dilution were added to each well and stimulated with 600 ng/ml of M-CSF. The plates were incubated for 24 h at 37 °C, 5% CO2. Unstimulated and stimulated cells served as reference for maximum and minimum induction. At the end of incubation 25 μL of Steady-Glo® Luciferase Assay System (Promega) were added after 5 min of lysis, luminescence was measured on microplate reader (Envision 2105, Perkinelmer). HEK-CSF1R-WST-1: Exponential growing HEK293T cells (ATCC® CRL- 3216™), were seeded at 5.10^3 per 200 μL of complete DMEM medium. The next day, twenty μL of test compound dilution were added to each well and the plates were incubated for 72 h at 37 °C, 5% CO2. Untreated cells and positive control (0,5% triton X-100, for the last 15 min) served as reference for maximum and minimum viability. At the end of incubation 100 μL of supernatant were removed and replaced by 10 μL of WST-1 solution (Cell Proliferation Reagent WST-1, Roche Applied Science). After 3 h incubation at 37 °C, 5% CO2, optical densities were measured at 450 nm and 620 nm for the background, on microplate reader (Envision 2105, Perkinelmer). IC50 were measured and some biological results of these assays are presented in the following table.
Table 3: Results biological cell-based assay measuring cell proliferation in non-cancer cell lines
Claims
CLAIMS 1. A compound according to general formula (I) [compound (C) hereinafter], or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, Formula (I) wherein: - each of R1, independently from each other and at each occurrence, is selected from hydrogen, or C1-6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of W, independently from each other and at each occurrence is a bond, or is selected from the group consisting of O, N(R11), and N(R11)(C=O); wherein each of R11, independently from each other and at each occurrence is selected from the group consisting of hydrogen and C1-6 alkyl; - each of X, independently from each other and at each occurrence is selected from O, S, or NR7, wherein R7 is selected from hydrogen or C1- 6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of R2, independently from each other and at each occurrence is a moiety selected from the group consisting of (R2-a) to (R2-f): (R2-a) (R2-b) (R2-c)
(R2-d) (R2-e) (R2-f) wherein: - each of Y independently from each other and at each occurrence is selected from the group consisting of O, S, and NR7, wherein R7 is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of R5, and R5’, independently from each other and at each occurrence is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom; - each of n5, independently from each other and at each occurrence, is an integer equal to 0, 1 or 2; - R6 is selected from the group consisting of halo, NO2, C1-6 alkyl, C2-4 alkenyl, C2-4 alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, CF3, CN, OR11, SR11, N(R11)2,, NR11COR12, COR11, C(O)OR11, CON(R11)2, OC(O)R11, and OCON(R11)2, wherein each of R11 and R12, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and CF3, wherein said alkyl, alkenyl, and alkynyl substituents are optionally substituted with at least one halogen atom. - each of n6, independently from each other and at each occurrence, is an integer equal to 0, 1 or 2; - each of Z independently from each other and at each occurrence is selected from the group consisting of O, S and NR7, wherein R7 is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with at least one halogen atom;
- each of R4 and R4’, independently from each other and at each occurrence, are selected from hydrogen, or C1-6 alkyl, wherein said alkyl is optionally substituted with a halogen atom, or R4-C-R4’ can form C=O; - each of z, independently from each other and at each occurrence, is an integer equal to 0, 1, 2, 3 or 4; - each of A, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl, wherein said alkyl, alkenyl, alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl are optionally substituted with one or more substituents independently selected from the group consisting of halo, NO2, C1-6 alkyl, C2-4 alkenyl, C2-4 alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, CF3, CN, OR11, SR11, N(R11)2, OC(R11)2O, OC(R11)2C(R11)2O, S(O)R12, SO2R12, SO2N(R11)2, S(O)3R11, P(=O)(OR11)2, P(=O)(R11)2, NR11COR12, COR11, C(O)OR11, CON(R11)2, OC(O)R11, and OCON(R11)2, and each optional alkyl, alkenyl, alkynyl, cycloalkyl, aryl, heterocyclyl, and heteroaryl substituents is further optionally substituted with halo, NO2, CF3, N(R11)2, COR11, CON(R11)2, OC(O)R11, CN, or OR11; and wherein each of R11 and R12, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and CF3, wherein said alkyl, alkenyl, and alkynyl, substituents are optionally substituted at least one halogen atom; wherein said cycloalkyl is a monocyclic, bicyclic or tricyclic ring system of 3- 6 ring members per ring; said heterocyclyl is a saturated, partially saturated or completely unsaturated monocycle, bicycle or tricycle containing 3 to 12 carbon atoms and 1 or 2 heteroatoms independently selected from O or N; said aryl is phenyl, naphthyl or anthracenyl optionally carbocyclic fused with a cycloalkyl or heterocyclyl of 5-7 ring members; said heteroaryl is a monocyclic ring structure containing 5 or 6 ring atoms, or a bicyclic aromatic group having 8 to 10 atoms, containing 1-3 heteroatoms independently selected from O or N.
2. The compound (C) according to claim 1, or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, wherein: - each of R1, independently from each other and at each occurrence, is selected from hydrogen or C1-6 alkyl; - each of W, independently from each other and at each occurrence is a bond, or is selected from the group consisting of O, N(R11), and N(R11)(C=O); wherein each of R11, independently from each other and at each occurrence is selected from the group consisting of hydrogen and C1-4 alkyl. - each of X, independently from each other and at each occurrence is selected from O, S, or NR7, wherein R7 is selected from hydrogen or C1- 6 alkyl. - each of R2, independently from each other and at each occurrence is a moiety selected from the group consisting of (R2-a) to (R2-f): (R2-a) (R2-b) (R2-c) (R2-d) (R2-e) (R2-f)
wherein: - each of Y independently from each other and at each occurrence is selected from the group consisting of O, S, and NR7, wherein R7 is selected from hydrogen or C1-6 alkyl; - each of R5, and R5’, independently from each other and at each occurrence is selected from hydrogen or C1-6 alkyl; - each of n5, independently from each other and at each occurrence is an integer equal to 0 or 1; - R6 is selected from the group consisting of halo, NO2, C1-6 alkyl, CF3, CN, OR11, SR11, N(R11)2, COR11, C(O)OR11, CON(R11)2, OC(O)R11, and wherein R11is independently selected from the group consisting of hydrogen, C1-6 alkyl, and CF3. - each of n6, independently from each other and at each occurrence, is an integer equal to 0 or 1; - each of Z, independently from each other and at each occurrence is selected from the group consisting of O, S, and NR7, wherein R7 is selected from hydrogen or C1-6 alkyl; - each of R4 and R4’, independently from each other and at each occurrence are selected from hydrogen or C1-6 alkyl, or R4-C-R4’ can form C=O. - each of z, independently from each other and at each occurrence, is an integer equal to 0, 1, 2 or 3; - each of A, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl, wherein said alkyl, alkenyl, alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, and aralkyl are optionally substituted with one or more substituents independently selected from the group consisting of halo, NO2, C1-6 alkyl, C2-4 alkenyl, C2-4 alkynyl, cycloalkyl, heterocyclyl, aryl, heteroaryl, CF3, CN, OR11, SR11, N(R11)2, OC(R11)2O, OC(R11)2C(R11)2O, S(O)R12, SO2R12, SO2N(R11)2, S(O)3R11, NR11COR12, COR11, C(O)OR11, CON(R11)2, OC(O)R11, and OCON(R11)2, and each optional alkyl, alkenyl, alkynyl, cycloalkyl, aryl, heterocyclyl, and heteroaryl substituents is further optionally substituted with halo, NO2, CF3, N(R11)2, CN, or OR11; and
wherein each of R11 and R12, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and CF3 wherein said cycloalkyl is a monocyclic, bicyclic or tricyclic ring system of 3-6 ring members per ring; said heterocyclyl is a saturated, partially saturated or completely unsaturated monocycle, bicycle or tricycle containing 3 to 12 carbon atoms and 1 or 2 heteroatoms independently selected from O or N; said aryl is phenyl, naphthyl or anthracenyl optionally carbocyclic fused with a cycloalkyl or heterocyclyl of 5-7 ring members; said heteroaryl is a monocyclic ring structure containing 5 or 6 ring atoms, or a bicyclic aromatic group having 8 to 10 atoms, containing 1-3 heteroatoms independently selected from O or N.
3. The compound (C) according to general formula (I), and according to claim 1, or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, wherein said compound (C) is chosen among those of formula (I-a-1) [compounds (C) of class (I-a)], or of formula (I-b-1) to (I-b-3) [compounds (C) of class (I-b)] or of formula (I-c-1) [compounds (C) of class (I-c)], or of formula (I-d-1) [compounds (C) of class (I-d)], or of formula (I-e-1) [compounds (C) of class (I-e)], or of formula (I- f-1) [compounds (C) of class (I-f)] herein after: Formula (I-a-1) Formula (I-b-1) Formula (I-b-2)
Formula (I-b-3) Formula (I-c-1) Formula (I-d-1) Formula (I-e-1) Formula (I-f-1) wherein R1, X, Y, Z, R5, n5, R5’, R4, R4’, z, A, R6 and n6 are as defined as in claim 1 or in claim 2.
4. The compound (C) according to general formula (I), and according to claim 1, or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, wherein said compound (C) is chosen among those of formula (I-a-1-1) [compounds (C) of class (I-a)], or of formula (I-b-1-1), (1-b-2-1) or (I-b-3-1) [compounds (C) of class (I-b)], or of formula (I-c-1-1) [compounds (C) of class (I-c)], or of formula (I-d-1-
1) [compounds (C) of class (I-d)], or of formula (I-e-1-1) [compounds (C) of class (I-e)], or of formula (I-f-1-1) [compounds (C) of class (I-f)] herein after: Formula (I-a-1-1) Formula (I-b-1-1) Formula (I-b-2-1) Formula (I-b-3-1) Formula (I-c-1-1) Formula (I-d-1-1) Formula (I-e-1-1)
Formula (I-f-1-1) wherein R1, R5, n5, R5’, R4, R4’, z, A, R6 and n6 are as defined as in claim 1 or in claim 2; and wherein R7 is selected from hydrogen or C1-6 alkyl, wherein said alkyl is optionally substituted with halogen atom, preferably R7 is hydrogen or methyl, more preferably R7 is hydrogen.
5. The compound (C) according to general formula (I), and according to claim 1, or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, wherein said compound (C) is chosen among those of formula (I-a-1-1-1-1) to (I-a-1-1-1-6) [compounds (C) of class (I-a)], herein after: Formula (I-a-1-1-1-1) Formula (I-a-1-1-1-2) Formula (I-a-1-1-1-3) Formula (I-a-1-1-1-4)
Formula (I-a-1-1-1-5) Formula (I-a-1-1-1-6) wherein R4, R4’ and z are as defined as in claim 1 or in claim 2, and wherein: - each of R8 independently from each other and at each occurrence is selected from the group consisting of hydrogen, halo and CF3. - n8 is an integer equal to 0, 1 or 2; - R5 and R5’, independently from each other and at each occurrence is hydrogen or methyl.
6. The compound (C) according to general formula (I), and according to claim 1, or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, wherein said compound (C) is chosen among those of formula (I-b-1-1-1-1) to (I-b-1-1-1-13) [compounds (C) of class (I-a)], herein after: Formula (I-b-1-1-1-1) Formula (I-b-1-1-1-2) Formula (I-b-1-1-1-3)
Formula (l-b-1 -1 -1 -4)
Formula (l-b-1 -1 -1 -5)
Formula (l-b-1 -1 -1 -6)
Formula (l-b-1 -1 -1 -7)
Formula (l-b-1 -1 -1 -8)
Formula (l-b-1 -1 -1 -9)
Formula (l-b-1 -1 -1 -10)
Formula (l-b-1 -1 -1 -11 )
Formula (l-b-1 -1 -1 -12)
H3C N z Formula (l-b-1 -1 -1 -13) wherein FU, FU’ and z are as defined as in claim 1 or in claim 2, and wherein:
R9 is selected from the group consisting of methyl, isopropyl, isobutyl, and ethyl, wherein said methyl, isopropyl, isobutyl and ethyl may be optionally substituted with N(CH3)2,
- each of Rw, independently from each other and at each occurrence is selected from the group consisting of hydrogen, halo, methyl and CF3;
- nw is an integer equal to 0 or 1 ;
- each of Rn, independently from each other and at each occurrence is selected from the group consisting of hydrogen, halo, methyl, OCH3, CF3, and SO2NHCH3;
- nn is an integer equal to 0, 1 or 2;
- each of RI2, independently from each other and at each occurrence is selected from the group consisting of hydrogen, CF3, and N(RI2I)2, wherein each of RI2I , independently from each other and at each occurrence is hydrogen or methyl;
- ni2 is an integer equal to 0 or 1 ;
R?b is selected from methyl or isopropyl;
- each of E, independently from each other and at each occurrence, is selected from CH or N;
- each of B independently from each other and at each occurrence, is selected from the group consisting of N-RI3, C(RB)2 and O, wherein each of RB, independently from each other and at each occurrence is selected from H or halo;
- each of RI3, independently from each other and at each occurrence is selected from hydrogen or methyl;
- the dash bond represents an optional double bond.
7. The compound (C) according to general formula (I), and according to claim 1 , or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, wherein said
compound (C) is chosen among those of formula (l-b-1 -1-2-1), (l-b-1 -1-3-1), (I- b-2-1-2-1), (l-b-2-1 -3-1 ), (l-b-3-1 -1 -1 ), (l-b-3-1 -2-1 ) or (l-b-3-1 -3-1 ) [compounds (C) of class (l-b)], herein after:
Formula (l-b-1 -1-3-1)
Halo
N
H3C Formula (l-b-2-1 -1-1)
R4 Halo
H,C
R4' - z Formula (l-b-2-1 -2-1)
R4 Halo
R4'
CH3
Formula (l-b-2-1 -3-1)
Formula (l-b-3-1 -1-1)
Halo
Formula (l-b-3-1 -2-1)
Formula (l-b-3-1 -3-1 ) wherein R4, R4’ and z are as defined as in claim 1 or in claim 2, and wherein halo is selected from the group consisting of F, Cl, Br and I.
8. The compound (C) according to general formula (I), and according to claim 1 , or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, wherein said compound (C) is chosen among those of formula (l-c-1 -1 -1 -1 ) or (l-c-1 -1 -1 -2) [compounds (C) of class (l-c)], herein after wherein R4, R4’ and z are as defined as in claim 1 or in claim 2, and wherein:
- each of R7’, independently from each other and at each occurrence is H or CH3;
- each of U is independently C-R12C or N;
- each of Ri2c, independently from each other and at each occurrence, is hydrogen, halo or OCH3;
- ni2c is an integer equal to 0, 1 or 2.
9. The compound (C) according to general formula (I), and according to claim 1 , or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, wherein said compound (C) is chosen among those of formula (l-d-1 -1 -1 -1 ) or (l-d-1 -1 -1 -2) [compounds (C) of class (l-d)], herein after
Formula (l-d-1 -1 -1 -1 )
Formula (l-d-1 -1 -1 -2) wherein R4, RT and z are as defined as in claim 1 or in claim 2, and wherein:
- each of U is independently C, C-Ri2d or N;
- each of Ri2d, independently from each other and at each occurrence, is hydrogen, halo or CF3;
- ni2d is an integer equal to 0, 1 or 2.
10. The compound (C) according to general formula (I), and according to claim 1 , or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, wherein said compound (C) is chosen among those of formula (l-f-1 -1 -1 -1 ) [compounds (C) of class (l-f)], herein after
Formula (l-f-1 -1 -1 -1 ) wherein halo is selected from the group consisting of F, Cl, Br and I.
1 1. The compound (C) according to general formula (I), and according to claim 1 , or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, wherein said compound (C) is chosen among those of formula (l-a-1 -1 -1 -3-0), (l-a-1 -1 -1 -3-1 ), (l-a-1 -1 -1 -3-2), or (l-a-1 -1 -1 -6-2) herein after:
H3C-N o
Formula (l-a-1 -1 -1 -3-0)
Formula (l-a-1 -1 -1 -3-1 )
H3C-N
^82
Formula (l-a-1 -1 -1 -3-2)
H3c— N
Formula (l-a-1 -1 -1 -6-2) wherein:
- each of R8o independently from each other and at each occurrence is selected from hydrogen or halo;
- n8o is an integer equal to 0 or 1 ;
- each of Rs and Rs’, independently from each other and at each occurrence is hydrogen or methyl;
- each of R8I independently from each other and at each occurrence is selected from the group consisting of hydrogen, halo and CF3;
- n8i is an integer equal to 0, 1 or 2;
- each of R82 independently from each other and at each occurrence is selected from hydrogen or halo.
12. The compound (C) according to general formula (I), and according to claim 1 , or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, wherein said compound (C) is chosen among those of formula (l-b-1 -1 -1 -4-0), (l-b-1 -1 -1 -2-1 ), (l-b-1 -1 -1 -4-1 ), (l-b-1 -1 -1 -5-1 ), (l-b-1 -1 -1 -7-1 ), (l-b-1 -1 -1 -8-1 ), (l-b-1 -1 -1 -12-1 ) or (l-b-1 -1 -1 -13-1 ) herein after
KK Rno
'3'
Formula (l-b-1 -1 -1 -4-0)
Formula (l-b-1 -1 -1 -2-1 )
Formula (l-b-1 -1 -1 -4-1 )
Formula (l-b-1 -1 -1 -5-1 )
Formula (l-b-1 -1 -1 -7-1 )
Formula (l-b-1 -1 -1 -8-1 )
Formula (l-b-1 -1 -1 -12-1 )
Formula (l-b-1 -1 -1 -13-1 ) wherein :
- each of Ruo, independently from each other and at each occurrence is selected from hydrogen or halo;
- each of Rwi, independently from each other and at each occurrence is selected from the group consisting of hydrogen, halo, and CF3;
- nioi is an integer equal to 0 or 1 ;
R4” is hydrogen or methyl; - each of R111, independently from each other and at each occurrence is selected from the group consisting of hydrogen, halo, OCH3, CF3, and SO2NHCH3;
- rim is an integer equal to 0, 1 or 2;
- each of RI2I , independently from each other and at each occurrence is selected from the group consisting of hydrogen, CF3, and N(RI2H)2, wherein each of RI2H , independently from each other and at each occurrence is hydrogen or methyl;
- ni2i is an integer equal to 0 or 1 ;
R?bi is selected from methyl, or isopropyl;
- each of B independently from each other and at each occurrence, is selected from N-CH3, or O;
- the dash bond represents an optional double bond.
13. The compound (C) according to general formula (I), and according to claim 1 , or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, wherein said compound (C) is chosen among those of formula (l-b-1 -1 -1 -1 -2), (l-b-1 -1 -1 -4-2), (l-b-1 -1 -1 -4-2a), (l-b-1 -1 -1 -5-2), (l-b-1 -1 -1 -5-2a), (l-b-1 -1 -1 -6-2), (l-b-1 -1 -1 -7-2),
(l-b-2-1 -1-1 -1 ), (l-b-2-1 -2-1 -1 ) or (l-b-2-1 -3-1 -1 ), (l-b-3-1 -1 -1 -1 ), (l-b-3-1 -2-1 -1 ), or (l-b-3-1 -3-1 -1 ) herein after:
Formula (l-b-1 -1 -1 -1 -2)
Formula (l-b-1 -1 -1 -4-2)
Formula (l-b-1 -1 -1 -4-2a)
Formula (l-b-1 -1 -1 -5-2)
Formula (l-b-1 -1 -1 -5-2a)
Formula (l-b-1 -1-1 -6-2)
Formula (l-b-1 -1-1 -7-2)
Formula (l-b-2-1 -1 -1 -1 )
Formula (l-b-2-1 -2-1-1)
Formula (l-b-2-1 -3-1-1)
Formula (l-b-3-1 -1 -1 -1 )
Formula (l-b-3-1 -2-1-1)
Formula (l-b-3-1 -3-1-1) wherein:
R92 is selected from hydrogen, N(CH3)2 or ethyl;
R42 is selected from hydrogen or ethyl;
- each of Rn2, independently from each other and at each occurrence is selected from the group consisting of hydrogen, halo, and CF3;
- each of RI02, independently from each other and at each occurrence is selected from hydrogen or methyl;
- each of ni02, independently from each other and at each occurrence is an integer equal to 0, 1 or 2;
- each of B independently from each other and at each occurrence, is selected from C-(RB)2 or O, wherein each of RB, independently from each other and at each occurrence is selected from H or halo.
- halo is selected from the group consisting of F, Cl, Br and I.
14. The compound (C) according to general formula (I), and according to claim 1 , or the N-oxide, pharmaceutically acceptable salt, pharmaceutically acceptable solvate, or stereoisomer thereof, wherein said compound (C) is chosen among those of formula (l-c-1 -1 -1 -2-1 ), (l-d-1 -1 -1 -1 -0), (l-d-1 -1 -1 -1 -1 ) herein after
Formula (l-c-1 -1 -1 -2-1 )
Formula (l-d-1 -1 -1 -1 -0)
Formula (l-d-1 -1 -1 -1 -1 ) wherein:
- each of R7’, independently from each other and at each occurrence is H or CH3;
- each of U is independently selected from C-RI2CI , C-Ri2do, C-Ri2di or N;
- each of RI2CI , independently from each other and at each occurrence, is hydrogen, halo or OCH3;
- ni2d is an integer equal to 0, 1 or 2;
- each of Ri2do, independently from each other and at each occurrence, is selected from the group consisting of hydrogen, halo, or CF3;
- ni2do is an integer equal to 0, 1 or 2; - each of Ri2di, independently from each other and at each occurrence, is hydrogen, halo or CF3;
- ni2di is an integer equal to 0, 1 or 2.
15. A pharmaceutical composition comprising a carrier, and as active ingredient the compound (C), as defined according to any one of claims 1 to 14.
16. A compound (C) as defined according to any one of claims 1 to 14, for use as a medicament.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23187940 | 2023-07-26 | ||
| EP23187940.4 | 2023-07-26 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025021943A1 true WO2025021943A1 (en) | 2025-01-30 |
Family
ID=87517203
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/071181 Pending WO2025021943A1 (en) | 2023-07-26 | 2024-07-25 | Quinazolinone, benzoxazinone and benzoxazepinone derivatives as protein kinase inhibitors |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025021943A1 (en) |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004022572A1 (en) | 2002-09-06 | 2004-03-18 | Alchemia Limited | Compounds that interact with kinases |
| WO2009070485A1 (en) | 2007-11-29 | 2009-06-04 | Boehringer Ingelheim International Gmbh | DERIVATIVES OF 6,7-DIHYDRO-5H-IMIDAZO[1,2-α]IMIDAZOLE-3- CARBOXYLIC ACID AMIDES |
| WO2009089042A1 (en) * | 2008-01-09 | 2009-07-16 | Signal Pharmaceuticals, Llc | Pyrazole pyrazine amine compounds as kinase inhibitors, compositions thereof and methods of treatment therewith |
| US20090325945A1 (en) | 2006-04-26 | 2009-12-31 | Dan Niculescu-Duvaz | Imidazo[4, 5-b]pyridin-2-one and oxazolo[4, 5-b]pyridin-2-one compounds and analogs thereof as cancer therapeutic compounds |
| WO2011032277A1 (en) * | 2009-09-18 | 2011-03-24 | Boehringer Ingelheim International Gmbh | Quinazolinone derivatives as viral polymerase inhibitors |
| WO2011090738A2 (en) | 2009-12-29 | 2011-07-28 | Dana-Farber Cancer Institute, Inc. | Type ii raf kinase inhibitors |
| WO2015042077A1 (en) * | 2013-09-22 | 2015-03-26 | Calitor Sciences, Llc | Substituted aminopyrimidine compounds and methods of use |
| US20150182526A1 (en) | 2007-12-19 | 2015-07-02 | Institute Of Cancer Research: Royal Cancer Hospital (The) | Pyrido[2,3-b]pyrazin-8-substituted compounds and their use |
| WO2021023888A1 (en) * | 2019-08-08 | 2021-02-11 | B.C.I. Pharma | Isoquinoline derivatives as protein kinase inhibitors |
-
2024
- 2024-07-25 WO PCT/EP2024/071181 patent/WO2025021943A1/en active Pending
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004022572A1 (en) | 2002-09-06 | 2004-03-18 | Alchemia Limited | Compounds that interact with kinases |
| US20090325945A1 (en) | 2006-04-26 | 2009-12-31 | Dan Niculescu-Duvaz | Imidazo[4, 5-b]pyridin-2-one and oxazolo[4, 5-b]pyridin-2-one compounds and analogs thereof as cancer therapeutic compounds |
| WO2009070485A1 (en) | 2007-11-29 | 2009-06-04 | Boehringer Ingelheim International Gmbh | DERIVATIVES OF 6,7-DIHYDRO-5H-IMIDAZO[1,2-α]IMIDAZOLE-3- CARBOXYLIC ACID AMIDES |
| US20150182526A1 (en) | 2007-12-19 | 2015-07-02 | Institute Of Cancer Research: Royal Cancer Hospital (The) | Pyrido[2,3-b]pyrazin-8-substituted compounds and their use |
| WO2009089042A1 (en) * | 2008-01-09 | 2009-07-16 | Signal Pharmaceuticals, Llc | Pyrazole pyrazine amine compounds as kinase inhibitors, compositions thereof and methods of treatment therewith |
| WO2011032277A1 (en) * | 2009-09-18 | 2011-03-24 | Boehringer Ingelheim International Gmbh | Quinazolinone derivatives as viral polymerase inhibitors |
| WO2011090738A2 (en) | 2009-12-29 | 2011-07-28 | Dana-Farber Cancer Institute, Inc. | Type ii raf kinase inhibitors |
| WO2015042077A1 (en) * | 2013-09-22 | 2015-03-26 | Calitor Sciences, Llc | Substituted aminopyrimidine compounds and methods of use |
| WO2021023888A1 (en) * | 2019-08-08 | 2021-02-11 | B.C.I. Pharma | Isoquinoline derivatives as protein kinase inhibitors |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2020324561B2 (en) | Quinoline derivatives as protein kinase inhibitors | |
| CA3017972C (en) | Compounds and methods for kinase modulation and indications therefore | |
| DK3009431T3 (en) | 2- (2,4,5-SUBSTITUTED ANILIN) -PYRIMIDINE DERIVATIVES AS EGFR MODULATORS USED FOR CANCER TREATMENT | |
| SG192686A1 (en) | Compounds and methods for kinase modulation, and indications therefor | |
| CN112313207B (en) | Cyano-substituted pyridine and cyano-substituted pyrimidine compounds, and preparation methods and applications thereof | |
| AU2017260298B2 (en) | Adenine derivatives as protein kinase inhibitors | |
| WO2020150545A1 (en) | Pyrazole derivatives as modulators of the wnt/b-catenin signaling pathway | |
| US11174255B2 (en) | Pyrido[2,3-d]pyrimidin-7-ones and related compounds as inhibitors of protein kinases | |
| WO2025021943A1 (en) | Quinazolinone, benzoxazinone and benzoxazepinone derivatives as protein kinase inhibitors | |
| AU2023385378A1 (en) | Pyridine derivatives as protein kinase inhibitors | |
| CA3022896C (en) | Adenine derivatives as protein kinase inhibitors | |
| AU2015261672B2 (en) | 2 - (2, 4, 5 - substituted -anilino) pyrimidine derivatives as egfr modulators useful for treating cancer | |
| EA048250B1 (en) | QUINOLINE DERIVATIVES AS PROTEIN KINASE INHIBITORS | |
| HK1256370B (en) | 2-(2,4,5-substituted-anilino)pyrimidine derivatives as egfr modulators useful for treating cancer | |
| HK1221216B (en) | 2-(2,4,5-substituted-anilino)pyrimidine derivatives as egfr modulators useful for treating cancer | |
| HK1192549B (en) | 2 - (2, 4, 5 - substituted -anilino) pyrimidine derivatives as egfr modulators useful for treating cancer |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24745462 Country of ref document: EP Kind code of ref document: A1 |