WO2025021790A2 - Multispecific antibodies - Google Patents
Multispecific antibodies Download PDFInfo
- Publication number
- WO2025021790A2 WO2025021790A2 PCT/EP2024/070818 EP2024070818W WO2025021790A2 WO 2025021790 A2 WO2025021790 A2 WO 2025021790A2 EP 2024070818 W EP2024070818 W EP 2024070818W WO 2025021790 A2 WO2025021790 A2 WO 2025021790A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- antibody
- multispecific antibody
- use according
- podxl
- tfr
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2896—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against molecules with a "CD"-designation, not provided for elsewhere
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2881—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against CD71
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/40—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against enzymes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/21—Immunoglobulins specific features characterized by taxonomic origin from primates, e.g. man
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/31—Immunoglobulins specific features characterized by aspects of specificity or valency multispecific
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/60—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments
- C07K2317/62—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments comprising only variable region components
- C07K2317/622—Single chain antibody (scFv)
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/77—Internalization into the cell
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
Definitions
- the present invention relates to multispecific antibodies binding to target proteins expressed on the Blood Brain Barrier (BBB) and their use to transport compounds across the BBB.
- BBB Blood Brain Barrier
- Brain penetration of neurological disorder drugs such as e.g. large biotherapeutic drugs or small molecule drugs having a low brain penetration, is strictly limited by the extensive and impermeable blood-brain-barrier (BBB) together with the other cell component in the neurovascular unit (NVU).
- BBB blood-brain-barrier
- NNU neurovascular unit
- Many strategies to overcome this obstacle have been tested and one is to utilize transcytosis pathways mediated by endogenous receptors expressed on the brain capillary endothelium (blood-brain-barrier- receptor).
- Recombinant proteins such as monoclonal antibodies or peptides have been designed against these receptors to enable receptor-mediated delivery of biotherapeutics to the brain.
- strategies to maximize brain uptake while minimizing miss-sorting within the brain endothelial cells (BECs), and the extent of accumulation within certain organelles (especially organelles that leads to degradation of the biotherapeutic) in BECs remain unexplored.
- BBB receptors i.e., transferrin receptor, insulin receptor and the like
- anti-transferrin receptor antibodies and methods of use are reported. It is further reported that targeting a BBB receptor with a traditional specific high-af- finity antibody generally resulted in limited increase in BBB transport. It was later found that the magnitude of antibody uptake into and distribution in the CNS is inversely related to its binding affinity for the BBB receptor amongst the anti-BBB antibodies studied. For example, a low-affinity antibody to transferrin receptor (TfR) dosed at therapeutic dose levels greatly improves BBB transport and CNS retention of the anti-TfR antibody relative to a higher- affinity anti-TfR antibody, and makes it possible to more readily attain therapeutic concentrations in the CNS (Atwal et al., Sci. Transl. Med.
- TfR transferrin receptor
- the present invention provides a multispecific antibody for use in transporting a compound across the Blood Brain Barrier (BBB), wherein the antibody binds to at least two target proteins selected from the group consisting of transferrin receptor (TFRC), TfR, CD98 (SLC3A2) and PODXL.
- BBB Blood Brain Barrier
- the antibody binds to TfR and CD98.
- the antibody binds to TfR and PODXL.
- the antibody binds to CD98 and PODXL.
- the antibody is a human antibody.
- the compound is a therapeutic compound.
- the antibody is coupled to the therapeutic compound.
- the target proteins are human proteins.
- the antibody comprises a first antigen binding site which binds to TfR and a second antigen binding site which binds to CD98.
- the antibody comprises a first antigen binding site which binds to TfR and a second antigen binding site which binds to PODXL.
- the antibody comprises a first antigen binding site which binds to CD98 and a second antigen binding site which binds to PODXL.
- the therapeutic compound is a neurological disorder drug.
- the therapeutic compound forms one portion of the multispecific antibody.
- the therapeutic compound forms one or two antigen binding sites of the multispecific antibody. In an embodiment of the present invention, the therapeutic compound which forms one or two antigen binding site of the multispecific antibody recognizes a brain antigen.
- the brain antigen is selected from the group consisting of beta-secretase 1 (BACE1), Abeta, epidermal growth factor receptor (EGFR), human epidermal growth factor receptor 2 (HER2), tau, apolipoprotein E (ApoE), alpha-synuclein, CD20, huntingtin, prion protein (PrP), leucine rich repeat kinase 2 (LRRK2), parkin, presenilin 1, presenilin 2, gamma secretase, death receptor 6 (DR6), amyloid precursor protein (APP), p75 neurotrophin receptor (p75NTR), TREM2, MS4A, TrkB, and caspase 6.
- BACE1 beta-secretase 1
- EGFR epidermal growth factor receptor
- HER2 human epidermal growth factor receptor 2
- tau tau
- ApoE apolipoprotein E
- alpha-synuclein CD20
- huntingtin huntingtin
- the antibody has a monovalent binding mode for each target protein.
- the TfR binding site has an affinity KD (nM) in the range of 100 - 1000 and the CD98 binding site has an affinity KD (nM) in the range of 0.1 - 100.
- the TfR binding site has an affinity KD (nM) in the range of 100 - 1000 and the PODXL binding site has an affinity KD (nM) in the range ofO.l - 100.
- the PODXL binding site has an affinity KD (nM) in the range of 0.1 - 100 and the CD98 binding site has an affinity KD (nM) in the range ofO.l - 100.
- the TfR binding site has an affinity KD (nM) in the range of 0.01 - 9 and the CD98 binding site has an affinity KD (nM) in the range of 0.1 - 100.
- the present invention provides an antibody for use in therapy, wherein the antibody is transporting a compound across the Blood Brain Barrier (BBB), wherein the antibody binds to at least the target protein PODXL.
- BBB Blood Brain Barrier
- the antibody is a human antibody.
- the compound is a therapeutic compound.
- the antibody is coupled to the therapeutic compound.
- the target proteins are human proteins.
- the therapeutic compound is a neurological disorder drug.
- the antibody is a multipecific antibody and the therapeutic compound forms one portion of the multispecific antibody.
- the therapeutic compound forms one or two antigen binding sites of the multispecific antibody.
- the therapeutic compound which forms one antigen binding site of the multispecific antibody recognizes a brain antigen.
- the brain antigen is selected from the group consisting of beta-secretase 1 (BACE1), Abeta, epidermal growth factor receptor (EGFR), human epidermal growth factor receptor 2 (HER2), tau, apolipoprotein E (ApoE), alpha-synuclein, CD20, huntingtin, prion protein (PrP), leucine rich repeat kinase 2 (LRRK2), parkin, presenilin 1, presenilin 2, gamma secretase, death receptor 6 (DR6), amyloid precursor protein (APP), p75 neurotrophin receptor (p75NTR), TREM2, MS4A, TrkB, and caspase 6.
- BACE1 beta-secretase 1
- EGFR epidermal growth factor receptor
- HER2 human epidermal growth factor receptor 2
- tau tau
- ApoE apolipoprotein E
- alpha-synuclein CD20
- huntingtin huntingtin
- the antibody has a monovalent binding mode for each target protein.
- Fig. 1 overview of the bispecific antibodies of the invention
- Fig. 2 - 4 Dose response of the bispecific antibodies on hCMECD3 cells:
- Fig. 5 2D transcytosis assay setup
- Fig. 6a -k Transcytosis & recycling of the bispecific antibodies of the present invention
- Fig. 7a - d Comparison of maximum transcytosis of bispecific antibodies directed to TfR/PODXL TfR/CD98 and PODXL/CD98 after 5 hours.
- Fig. 8 Comparison of maximum transcytosis of bispecific antibodies directed to TfR/PODXL TfR/CD98 and PODXL /CD98 after 20 hours.
- Fig. 9a- c Experimental setup of human BBB spheroids
- Fig. Ila - d signal quantification in human BBB spheroids.
- DP47 is a dummy binder.
- Fig. 12 Graphic overview of transcytosis rate for the bispecific antibodies of the invention: P1AI8549 (701-PODXL-low/1026-TFR-low) shows increased transcytosis compared to the controls P1AI8552 (701-PODXL-low/Dummy), P1AI8553 (1026-TFR-low/Dummy) and to the compared one-arm brain shuttle Pl AF3732 (1026-TFR-low).
- Fig. 13 Transcytosis and recycling of monospecific PODXL antibodies. DETAILED DESCRIPTION OF EMBODIMENTS OF THE INVENTION
- the "blood-brain barrier” or “BBB” refers to the physiological barrier between the peripheral circulation and the brain and spinal cord which is formed by tight junctions within the brain capillary endothelial plasma membranes, creating a tight barrier that restricts the transport of molecules into the brain, even very small molecules such as urea (60 Daltons).
- the BBB within the brain, the blood-spinal cord barrier within the spinal cord, and the blood- retinal barrier within the retina are contiguous capillary barriers within the CNS, and are herein collectively referred to an the blood-brain barrier or BBB.
- the BBB also encompasses the blood-CSF barrier (choroid plexus) where the barrier is comprised of ependymal cells rather than capillary endothelial cells.
- knobs into holes dimerization modules and their use in antibody engineering are described in Carter P.; Ridgway J.B.B.; Presta L.G.: Immunotechnology, Volume 2, Number 1, February 1996 , pp. 73-73(1)).
- central nervous system or “CNS” refers to the complex of nerve tissues that control bodily function, and includes the brain and spinal cord.
- R/BBB blood-brain barrier receptor
- IGF-R insulinlike growth factor receptor
- LRP1 low density lipoprotein receptors
- LRP8 heparin-binding epidermal growth factor-like growth factor
- HB-EGF heparin-binding epidermal growth factor-like growth factor
- Binding affinity refers to the strength of the sum total of nonco valent interactions between a single binding site of a molecule (e.g., an antibody) and its binding partner (e.g., an antigen).
- binding affinity refers to intrinsic binding affinity which reflects a 1 :1 interaction between members of a binding pair (e.g., antibody and antigen).
- KD dissociation constant
- Affinity can be measured by common methods known in the art. A preferred method for measuring affinity is Surface Plasmon Resonance (SPR).
- the "monovalent binding entity” refers to a molecule able to bind specifically and in a monovalent binding mode to an R/BBB.
- the blood brain shuttle and/or conjugate of the present invention are characterized by the presence of a single unit of a monovalent binding entity i.e. the blood brain shuttle and/or conjugate of the present invention comprise one unit of the monovalent binding entity.
- the monovalent binding entity includes but is not limited to proteins, poly-peptides, peptides and antibody fragments including Fab, Fab', Fv fragments, single-chain antibody molecules such as e.g. single chain Fab, scFv.
- the monovalent binding entity can for example be a scaffold protein engineered using state of the art technologies like phage display or immunization.
- the monovalent binding entity can also be a peptide.
- the "monovalent binding mode” refers to a specific binding to the R/BBB where the interaction between the monovalent binding entity and the R/BBB take place through one single epitope.
- the monovalent binding mode prevents any dimerization/multimerization of the R/BBB due to a single epitope interaction point.
- the monovalent binding mode prevents that the intracellular sorting of the R/BBB is changed.
- epitope includes any polypeptide determinant capable of specific binding to an antibody.
- epitope determinant include chemically active surface groupings of molecules such as amino acids, sugar side chains, phosphoryl, or sulfonyl, and, in certain embodiments, may have specific three dimensional structural characteristics, and or specific charge characteristics.
- An epitope is a region of an antigen that is bound by an antibody.
- TfR transferrin receptor
- the "transferrin receptor” (“TfR”) is a transmembrane glycoprotein (with a molecular weight of about 180,000) composed of two disulphide-bonded sub-units (each of apparent molecular weight of about 90,000) involved in iron uptake in vertebrates.
- the TfR herein is human TfR comprising the amino acid sequence as in Schneider et al. Nature 311 : 675 - 678 (1984), for example.
- CD98 (also referred to as CD98 heavy chain; 42F heavy chain; SLC3A2) is a type II transmembrane glycoprotein.
- the human CD98hc sequence is set forth in UNIPROT Accession No. P08195.
- the protein comprises a 75 amino acid N-terminal intracellular cytoplasmic domain, a single transmembrane domain, and a 426 amino acid C-terminal extracellular domain (Parmacek et al., Nucleic Acids Res. 17: 1915-1931, 1989).
- CD98 covalently links via a disulfide bond to one of several light chains (SLC7A5, 6, 7, 8, 10, or 1 1), which are L-type amino acid transporters.
- CD98 also associates with integral [3 subunits, thereby regulating integrin signaling that controls cell proliferation, survival, migration, and epithelial adhesion/polarity (Cai et al., J. Cell Sci. 1 18: 889-899, 2005).
- PODXL also known as podocalyxin-like protein-1, PCLP1 or PCX
- PCLP1 podocalyxin-like protein-1
- PCX podocalyxin-like protein-1
- PODXL Preserving the basic structure of family members CD34 and endoglycan, PODXL consists of a highly conserved cytoplasmic domain with a C-terminal PDZ binding region (DTHL motif), a single-pass transmembrane domain, and an extensively O-glycosylated and sialylated extracellular domain
- DTHL motif C-terminal PDZ binding region
- a "neurological disorder” as used herein refers to a disease or disorder which affects the CNS and/or which has an etiology in the CNS.
- CNS diseases or disorders include, but are not limited to, neuropathy, amyloidosis, cancer, an ocular disease or disorder, viral or microbial infection, inflammation, ischemia, neurodegenerative disease, seizure, behavioral disorders, and a lysosomal storage disease.
- the CNS will be understood to include the eye, which is normally sequestered from the rest of the body by the blood-retina barrier.
- neurological disorders include, but are not limited to, neurodegenerative diseases (including, but not limited to, Lewy body disease, postpoliomyelitis syndrome, Shy-Draeger syndrome, olivopontocerebellar atrophy, Parkinson's disease, multiple system atrophy, stria- tonigral degeneration, tauopathies (including, but not limited to, Alzheimer disease and supranuclear palsy), prion diseases (including, but not limited to, bovine spongiform encephalopathy, scrapie, Creutzfeldt- Jakob syndrome, kuru, Gerstmann-Straussler-Scheinker disease, chronic wasting disease, and fatal familial insomnia), bulbar palsy, motor neuron disease, and nervous system heterodegenerative disorders (including, but not limited to, Canavan disease, Huntington's disease, neuronal ceroid- lipofuscinosis, Alexander's disease, Tourette's syndrome, Menkes kinky hair syndrome, Cockayne syndrome, Halervorden-Spatz syndrome,
- Neurological disorder drug is a drug or therapeutic agent that treats one or more neurological disorder(s).
- Neurological disorder drugs of the invention include, but are not limited to, small molecule compounds, antibodies, peptides, proteins, natural ligands of one or more CNS target(s), modified versions of natural ligands of one or more CNS target(s), aptamers, inhibitory nucleic acids (i.e., small inhibitory RNAs (siRNA) and short hairpin RNAs (shRNA)), ribozymes, and small molecules, or active fragments of any of the foregoing.
- siRNA small inhibitory RNAs
- shRNA short hairpin RNAs
- Exemplary neurological disorder drugs of the invention include, but are not limited to: antibodies, aptamers, proteins, peptides, inhibitory nucleic acids and small molecules and active fragments of any of the foregoing that either are themselves or specifically recognize and/or act upon (i.e., inhibit, activate, or detect) a CNS antigen or target molecule such as, but not limited to, amyloid precursor protein or portions thereof, amyloid beta, beta-secretase, gamma-secretase, tau, alpha-synuclein, parkin, huntingtin, DR6, presenilin, ApoE, glioma or other CNS cancer markers, and neurotrophins.
- a CNS antigen or target molecule such as, but not limited to, amyloid precursor protein or portions thereof, amyloid beta, beta-secretase, gamma-secretase, tau, alpha-synuclein, parkin, huntingtin, DR6, presenilin, ApoE
- BDNF Brain- derived neurotrophic factor
- Neurogen-2 Chronic brain injury
- FGF-2 Fibroblast growth factor 2
- EGFR Anti-Epidermal Growth Factor Receptor Brain cancer
- EGFR G
- imaging agent is a compound that has one or more properties that permit its presence and/or location to be detected directly or indirectly.
- imaging agents include proteins and small molecule compounds incorporating a labeled entity that permits detection.
- a “CNS antigen” or "brain target” is an antigen and/or molecule expressed in the CNS, including the brain, which can be targeted with an antibody or small molecule.
- antigen and/or molecule include, without limitation: beta-secretase 1 (BACE1), amyloid beta (Abeta), epidermal growth factor receptor (EGFR), human epidermal growth factor receptor 2 (HER2), Tau, apolipoprotein E4 (ApoE4), alpha-synuclein, CD20, huntingtin, prion protein (PrP), leucine rich repeat kinase 2 (LRRK2), parkin, presenilin 1, presenilin 2, gamma secretase, death receptor 6 (DR6), amyloid precursor protein (APP), p75 neurotro- phin receptor (p75NTR), and caspase 6.
- BACE1 beta-secretase 1
- Abeta amyloid beta
- EGFR epidermal growth factor receptor
- a “native sequence” protein herein refers to a protein comprising the amino acid sequence of a protein found in nature, including naturally occurring variants of the protein.
- the term as used herein includes the protein as isolated from a natural source thereof or as recom- binantly produced.
- antibody herein is used in the broadest sense and specifically covers monoclonal antibodies, polyclonal antibodies, multispecific antibodies ⁇ e.g. bispecific antibodies) formed from at least two intact antibodies, and antibody fragments so long as they exhibit the desired biological activity.
- Antibody fragments herein comprise a portion of an intact antibody which retains the ability to bind antigen.
- Examples of antibody fragments include Fab, Fab', F(ab')2, and Fv fragments; diabodies; linear antibodies; single-chain antibody molecules such as e.g. single chain Fab, scFv and multispecific antibodies formed from antibody fragments.
- the “Single chain Fab” format is e.g. described in Hust M. et al. BMC Biotechnol. 2007 Mar 8;7:14.
- the term "monoclonal antibody” as used herein refers to an antibody obtained from a population of substantially homogeneous antibodies, i.e., the individual antibodies comprising the population are identical and/or bind the same epitope, except for possible variants that may arise during production of the monoclonal antibody, such variants generally being present in minor amounts.
- each monoclonal antibody is directed against a single determinant on the antigen.
- the monoclonal antibodies are advantageous in that they are uncontaminated by other immunoglobulins.
- the modifier "monoclonal” indicates the character of the antibody as being obtained from a substantially homogeneous population of antibodies, and is not to be construed as requiring production of the antibody by any particular method.
- the monoclonal antibodies to be used in accordance with the present invention may be made by the hy- bridoma method first described by Kohler et al., Nature, 256:495 (1975), or may be made by recombinant DNA methods (see, e.g., U.S. Patent No. 4,816,567).
- the "monoclonal antibodies” may also be isolated from phage antibody libraries using the techniques described in Clackson et al., Nature, 352:624-628 (1991) and Marks et al., J. Mol. Biol., 222:581-597 (1991), for example.
- Specific examples of monoclonal antibodies herein include chimeric antibodies, humanized antibodies, and human antibodies, including antigen-binding fragments thereof.
- the monoclonal antibodies herein specifically include "chimeric" antibodies (immunoglobulins) in which a portion of the heavy and/or light chain is identical with or homologous to corresponding sequences in antibodies derived from a particular species or belonging to a particular antibody class or subclass, while the remainder of the chain(s) is identical with or homologous to corresponding sequences in antibodies derived from another species or belonging to another antibody class or subclass, as well as fragments of such antibodies, so long as they exhibit the desired biological activity (U.S. Patent No. 4,816,567; Morrison et al, Proc. Natl. Acad. Sci. USA, 81 :6851-6855 (1984)).
- chimeric antibodies immunoglobulins in which a portion of the heavy and/or light chain is identical with or homologous to corresponding sequences in antibodies derived from a particular species or belonging to a particular antibody class or subclass, while the remainder of the chain(s) is identical with or homologous to corresponding sequences in
- Chimeric antibodies of interest herein include "primatized" antibodies comprising variable domain antigen-binding sequences derived from a non-human primate ⁇ e.g. Old World Monkey, such as baboon, rhesus or cynomolgus monkey) and human constant region sequences (US Pat No. 5,693,780).
- a non-human primate e.g. Old World Monkey, such as baboon, rhesus or cynomolgus monkey
- human constant region sequences US Pat No. 5,693,780
- Humanized forms of non-human ⁇ e.g., murine antibodies are chimeric antibodies that contain minimal sequence derived from non-human immunoglobulin.
- humanized antibodies are human immunoglobulins (recipient antibody) in which residues from a hypervariable region of the recipient are replaced by residues from a hypervariable region of a non-human species (donor antibody) such as mouse, rat, rabbit or nonhuman primate having the desired specificity, affinity, and capacity.
- donor antibody such as mouse, rat, rabbit or nonhuman primate having the desired specificity, affinity, and capacity.
- framework region (FR) residues of the human immunoglobulin are replaced by corresponding non-human residues.
- humanized antibodies may comprise residues that are not found in the recipient antibody or in the donor antibody. These modifications are made to further refine antibody performance.
- the humanized antibody will comprise substantially all of at least one, and typically two, variable domains, in which all or substantially all of the hypervariable regions correspond to those of a non-human immunoglobulin and all or substantially all of the FRs are those of a human immunoglobulin sequence, except for FR substitution(s) as noted above.
- the humanized antibody optionally also will comprise at least a portion of an immunoglobulin constant region, typically that of a human immunoglobulin. For further details, see Jones et al, Nature 321 :522-525 (1986); Riechmann et al, Nature 332:323-329 (1988); and Presta, Curr. Op. Struct. Biol 2:593-596 (1992).
- a "human antibody” herein is one comprising an amino acid sequence structure that corresponds with the amino acid sequence structure of an antibody obtainable from a human B- cell, and includes antigen-binding fragments of human antibodies.
- Such antibodies can be identified or made by a variety of techniques, including, but not limited to: production by transgenic animals ⁇ e.g., mice) that are capable, upon immunization, of producing human antibodies in the absence of endogenous immunoglobulin production (see, e.g., Jakobovits et al, Proc. Natl Acad. Sci.
- a “multispecific antibody” herein is an antibody having binding specificities for at least two different epitopes.
- Exemplary multispecific antibodies may bind both an R/BBB and a brain antigen.
- Multispecific antibodies can be prepared as full-length antibodies or antibody fragments (e.g. F(ab')2 bispecific antibodies).
- Engineered antibodies with two, three or more (e.g. four) functional antigen binding sites are also contemplated (see, e.g., US Appln No. US 2002/0004587 Al, Miller et al.).
- Multispecific antibodies can be prepared as full length antibodies or antibody fragments.
- Antibodies herein include "amino acid sequence variants" with altered antigen-binding or biological activity.
- amino acid alterations include antibodies with enhanced affinity for antigen (e.g. "affinity matured” antibodies), and antibodies with altered Fc region, if present, e.g. with altered (increased or diminished) antibody dependent cellular cytotoxicity (ADCC) and/or complement dependent cytotoxicity (CDC) (see, for example, WO 00/42072, Presta, L. and WO 99/51642, Iduosogie et al); and/or increased or diminished serum half-life (see, for example, WO00/42072, Presta, L.).
- ADCC antibody dependent cellular cytotoxicity
- CDC complement dependent cytotoxicity
- an "affinity modified variant” has one or more substituted hypervariable region or framework residues of a parent antibody (e.g. of a parent chimeric, humanized, or human antibody) that alter (increase or reduce) affinity.
- a parent antibody e.g. of a parent chimeric, humanized, or human antibody
- the resulting variant(s) selected for further development will have reduced affinity for the R/BBB according to the present invention.
- a convenient way for generating such substitutional variants uses phage display. Briefly, several hypervariable region sites (e.g. 6-7 sites) are mutated to generate all possible amino substitutions at each site. The antibody variants thus generated are displayed in a monovalent fashion from filamentous phage particles as fusions to the gene III product of Ml 3 packaged within each particle.
- the phage-displayed variants are then screened for their biological activity (e.g. binding affinity).
- alanine scanning mutagenesis can be performed to identify hypervariable region residues contributing significantly to antigen binding.
- the antibody herein may be a "glycosylation variant" such that any carbohydrate attached to the Fc region, if present, is altered.
- a glycoslation variant such that any carbohydrate attached to the Fc region, if present, is altered.
- antibodies with a mature carbohydrate structure that lacks fucose attached to an Fc region of the antibody are described in US Pat Appl No US 2003/0157108 (Presta, L ). See also US 2004/0093621 (KyowaHakko Kogyo Co., Ltd).
- Antibodies with a bisecting N-acetylglucosamine (GlcNAc) in the carbohydrate attached to an Fc region of the antibody are referenced in WO 2003/011878, Jean-Mai- ret et al. and US Patent No.
- the hypervariable region comprises amino acid residues from a "complementarity determining region" or "CDR" (e.g. residues 24- 34 (LI), 50-56 (L2) and 89-97 (L3) in the light chain variable domain and 31-35 (HI), 50-65 (H2) and 95-102 (H3) in the heavy chain variable domain; Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, MD. (1991)) and/or those residues from a "hypervariable loop" (e.g.
- a “full length antibody” is one which comprises an antigen-binding variable region as well as a light chain constant domain (CL) and heavy chain constant domains, CHI, CH2 and CH3.
- the constant domains may be native sequence constant domains (e.g. human native sequence constant domains) or amino acid sequence variants thereof.
- naked antibody is an antibody (as herein defined) that is not conjugated to a heterologous molecule, such as a cytotoxic entity, polymer, or radiolabel.
- Antibody effector functions refer to those biological activities attributable to the Fc region (a native sequence Fc region or amino acid sequence variant Fc region) of an antibody. Examples of antibody effector functions include Clq binding, complement dependent cytotoxicity (CDC), Fc receptor binding, antibody-dependent cell-mediated cytotoxicity (ADCC), etc. In one embodiment, the antibody herein essentially lacks effector function.
- ADCC antibody-dependent cellular cytotoxicity
- CDC complement-dependent cytotoxicity
- Antibodies of subclass IgGl, IgG2, and IgG3 usually show complement activation including Clq and C3 binding, whereas IgG4 does not activate the complement system and does not bind Clq and/or C3.
- full length antibodies can be assigned to different "classes". There are five major classes of full length antibodies: IgA, IgD, IgE, IgG, and IgM, and several of these may be further divided into “subclasses” (isotypes), e.g., IgGl, IgG2, IgG3, IgG4, IgA, and IgA2.
- the heavychain constant domains that correspond to the different classes of antibodies are called alpha, delta, epsilon, gamma, and mu, respectively.
- the subunit structures and three-dimensional configurations of different classes of immunoglobulins are well known.
- recombinant antibody refers to an antibody (e.g. a chimeric, humanized, or human antibody or antigen-binding fragment thereof) that is expressed by a recombinant host cell comprising nucleic acid encoding the antibody.
- host cells include: (1) mammalian cells, for example, Chinese Hamster Ovary (CHO), COS, myeloma cells (including YO and NSO cells), baby hamster kidney (BHK), Hela and Vero cells; (2) insect cells, for example, sf9, sf21 and Tn5; (3) plant cells, for example plants belonging to the genus Nicotiana (e.g.
- Nicotianatabacum (4) yeast cells, for example, those belonging to the genus Saccharomyces (e.g. Saccharomyces cerevisiae) or the genus Aspergillus (e.g. Aspergillus niger); (5) bacterial cells, for example Escherichia, coli cells or Bacillus subtilis cells, etc.
- yeast cells for example, those belonging to the genus Saccharomyces (e.g. Saccharomyces cerevisiae) or the genus Aspergillus (e.g. Aspergillus niger)
- bacterial cells for example Escherichia, coli cells or Bacillus subtilis cells, etc.
- binding affinity is generally determined using a standard assay, such as Scatchard analysis, or surface plasmon resonance technique (e.g. using BIACORE®).
- an "antibody that binds to the same epitope” as a reference antibody refers to an antibody that blocks binding of the reference antibody to its antigen in a competition assay by 50% or more, and conversely, the reference antibody blocks binding of the antibody to its antigen in a competition assay by 50% or more.
- imaging agent is a compound that has one or more properties that permit its presence and/or location to be detected directly or indirectly.
- imaging agents include proteins and small molecule compounds incorporating a labeled entity that permits detection.
- a “label” is a marker coupled with the antibody herein and used for detection or imaging.
- labels include: radiolabel, a fluorophore, a chromophore, or an affinity tag.
- the label is a radiolabel used for medical imaging, for example tc99m or 1123, or a spin label for nuclear magnetic resonance (NMR) imaging (also known as magnetic resonance imaging, mri), such as iodine- 123 again, iodine-131, indium-i ll, fluo- rine- 19, carbon-13, nitrogen-15, oxygen-17, gadolinium, manganese, iron, etc.
- NMR nuclear magnetic resonance
- cytotoxic agent refers to a substance that inhibits or prevents a cellular function and/or causes cell death or destruction.
- Cytotoxic agents include, but are not limited to, radioactive isotopes (e.g., At211, 1131, 1125, Y90, Rel86, Rel88, Sml53, Bi212, P32, Pb212 and radioactive isotopes of Lu); chemotherapeutic agents or drugs (e.g., methotrexate, adriamicin, vinca alkaloids (vincristine, vinblastine, etoposide), doxorubicin, melphalan, mitomycin C, chlorambucil, daunorubicin or other intercalating agents); growth inhibitory agents; enzymes and fragments thereof such as nucleolytic enzymes; antibiotics; toxins such as small molecule toxins or enzymatically active toxins of bacterial, fungal, plant or animal origin, including fragments and/or variants
- an "effective amount" of an agent refers to an amount effective, at dosages and for periods of time necessary, to achieve the desired therapeutic or prophylactic result.
- Fc region herein is used to define a C-terminal region of an immunoglobulin heavy chain that contains at least a portion of the constant region.
- the Fc region comprises the CH2 and CH3 domains of an immunoglobulin.
- the term includes native sequence Fc regions and variant Fc regions.
- a human IgG heavy chain Fc region extends from Cys226, or from Pro230, to the carboxyl-terminus of the heavy chain.
- the C- terminal lysine (Lys447) of the Fc region may or may not be present.
- EU numbering system also called the EU index, as described in Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, MD, 1991.
- FR Framework or "FR” refers to variable domain residues other than hypervariable region (HVR) residues.
- the FR of a variable domain generally consists of four FR domains: FR1, FR2, FR3, and FR4. Accordingly, the HVR and FR sequences generally appear in the following sequence in VH (or VL): FR1-H1(L1)-FR2-H2(L2)-FR3-H3(L3)-FR4.
- CH2-CH3 Ig entity refers to a protein entity derived from immunoglobulin CH2 or CH3 domains.
- the “CH2-CH3 Ig entity” comprises two “CH2- CH3” polypeptides forming a dimer.
- the immunoglobulin can be IgG, IgA, IgD, IgE or IgM.
- the CH2-CH3 Ig entity derived from an IgG immunoglobulin is referred to herein as “CH2-CH3 IgG entity”.
- the term includes native sequence of CH2-CH3 domains and variant CH2-CH3 domains.
- the “CH2-CH3 Ig entity” derives from human heavy chain CH2-CH3 IgG domain which extends from Cys226, or from Pro230, to the carboxyl-terminus of the heavy chain.
- the C-terminal lysine (Lys447) of the Fc region may or may not be present.
- numbering of amino acid residues in the CH2-CH3 domain region or constant region is according to the EU numbering system, also called the EU index, as described in Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, MD, 1991.
- conjugated is fusion protein of the present invention conjugated to one or more heterologous molecule(s), including but not limited to a label, neurological disorder drug or cytotoxic agent.
- a "linker” as used herein refers to a chemical linker or a single chain peptide linker that covalently connects the different entities of the multispecific antibody and/or the fusion protein and/or the conjugate of the present invention.
- the linker connects for example the compound to the monovalent binding entity.
- the monovalent binding entity comprises a CH2-CH3 Ig entity and a sFab directed to the blood brain barrier receptor, then the linker connects the scFab to the C-terminal end of the CH3-CH2 Ig entity.
- the linker connecting the brain effector entity to the monovalent binding entity (first linker) and the linker connecting the scFab to the C-terminal end of the CH2-CH3 Ig domain (second linker) can be the same or different.
- Single chain peptide linkers comprised of from one to twenty amino acids joined by peptide bonds, can be used.
- the amino acids are selected from the twenty naturally-occurring amino acids.
- one or more of the amino acids are selected from glycine, alanine, proline, asparagine, glutamine and lysine.
- the linker is a chemical linker.
- said linker is a single chain peptide with an amino acid sequence with a length of at least 25 amino acids, preferably with a length of 32 to 50 amino acids.
- said linker is (648)4 (Seq. Id. No. 17).
- said linker is (GiSjeGz (Seq. Id. No. 13).
- Conjugation may be performed using a variety of chemical linkers.
- the monovalent binding entity or the fusion protein and the compound may be conjugated using a variety of bifunctional protein coupling agents such as N-succinimidyl-3-(2-pyridyldithio) propionate (SPDP), succinimidyl-4-(N-maleimidomethyl) cyclohexane-l-carboxylate (SMCC), iminothiolane (IT), bifunctional derivatives of imidoesters (such as dimethyl adip- imidate HC1), active esters (such as disuccinimidyl sub erate), aldehydes (such as glutaraldehyde), bis-azido compounds (such as bis (p- azidobenzoyl) hexanediamine), bis-diazonium derivatives (such as bis-(p-diazoniumbenzoyl)- ethylenediamine), diisocyanates (such as tolu
- the linker may be a "cleavable linker" facilitating release of the effector entity upon delivery to the brain.
- a "cleavable linker” facilitating release of the effector entity upon delivery to the brain.
- an acid- labile linker, peptidase-sensitive linker, photola- bile linker, dimethyl linker or disulfide- containing linker (Chari et al, Cancer Res. 52: 127- 131 (1992); U.S. Patent No. 5,208,020) may be used.
- Covalent conjugation can either be direct or via a linker.
- direct conjugation is by construction of a protein fusion (i.e., by genetic fusion of the two genes encoding the monovalent binding entity towards the R/BBB and effector entity and expressed as a single protein).
- direct conjugation is by formation of a covalent bond between a reactive group on one of the two portions of the monovalent binding entity against the R/BBB and a corresponding group or acceptor on the compound.
- direct conjugation is by modification (i.e., genetic modification) of one of the two molecules to be conjugated to include a reactive group (as non-limiting examples, a sulfhydryl group or a carboxyl group) that forms a covalent attachment to the other molecule to be conjugated under appropriate conditions.
- a reactive group as non-limiting examples, a sulfhydryl group or a carboxyl group
- a molecule i.e., an amino acid
- a desired reactive group i.e., a cysteine residue
- a monovalent binding entity and a effector entity may be conjugated using a variety of bifunctional protein coupling agents such as N- succinimidyl-3 -(2 -pyridyldithio) propionate (SPDP), succinimidyl-4-(N-maleimidomethyl) cyclohexane-l-carboxylate (SMCC), iminothiolane (IT), bifunctional derivatives of imidoesters (such as dimethyl adipimidate HC1), active esters (such as disuccinimidyl suberate), aldehydes (such as glutaraldehyde), bis-azido compounds (such as bis (p- azidobenzoyl) hexanediamine), bis-diazonium derivatives (such as bis-(p-dia- zoniumbenzoyl)- ethylenediamine), diisocyanates (such as toluene 2,6-diisocyanate), and bis- active protein coup
- Peptide linkers com- prised of from one to twenty amino acids joined by peptide bonds
- the amino acids are selected from the twenty naturally-occurring amino acids.
- one or more of the amino acids are selected from glycine, alanine, proline, asparagine, glutamine and lysine.
- the linker may be a "cleavable linker" facilitating release of the effector entity upon delivery to the brain.
- an acid- labile linker, peptidase-sensitive linker, photolabile linker, dimethyl linker or disulfide- containing linker (Chari et al, Cancer Res. 52: 127-131 (1992); U.S. Patent No. 5,208,020) may be used.
- a “label” is a marker coupled with the fusion protein herein and used for detection or imaging.
- labels include: radiolabel, a fluorophore, a chromophore, or an affinity tag.
- the label is a radiolabel used for medical imaging, for example tc99m or 1123, or a spin label for nuclear magnetic resonance (NMR) imaging (also known as magnetic resonance imaging, mri), such as iodine- 123 again, iodine-131, indium- 111, fluorine- 19, carbon- 13, nitrogen-15, oxygen- 17, gadolinium, manganese, iron, etc.
- NMR nuclear magnetic resonance
- Mammals include, but are not limited to, domesticated animals (e.g., cows, sheep, cats, dogs, and horses), primates (e.g., humans and non-human primates such as monkeys), rabbits, and rodents (e.g., mice and rats).
- domesticated animals e.g., cows, sheep, cats, dogs, and horses
- primates e.g., humans and non-human primates such as monkeys
- rabbits e.g., mice and rats
- rodents e.g., mice and rats.
- the individual or subject is a human.
- an “isolated” antibody is one which has been separated from a component of its natural environment.
- an antibody is purified to greater than 95% or 99% purity as determined by, for example, electrophoretic (e.g., SDS-PAGE, isoelectric focusing (IEF), capillary electrophoresis) or chromatographic (e.g., ion exchange or reverse phase HPLC).
- electrophoretic e.g., SDS-PAGE, isoelectric focusing (IEF), capillary electrophoresis
- chromatographic e.g., ion exchange or reverse phase HPLC
- pharmaceutical formulation refers to a preparation which is in such form as to permit the biological activity of an active ingredient contained therein to be effective, and which contains no additional components which are unacceptably toxic to a subject to which the formulation would be administered.
- a “pharmaceutically acceptable carrier” refers to an ingredient in a pharmaceutical formulation, other than an active ingredient, which is nontoxic to a subject.
- a pharmaceutically acceptable carrier includes, but is not limited to, a buffer, excipient, stabilizer, or preservative.
- treatment refers to clinical intervention in an attempt to alter the natural course of the individual being treated, and can be performed either for prophylaxis or during the course of clinical pathology. Desirable effects of treatment include, but are not limited to, preventing occurrence or recurrence of disease, alleviation of symptoms, diminishment of any direct or indirect pathological consequences of the disease, preventing metastasis, decreasing the rate of disease progression, amelioration or palliation of the disease state, and remission or improved prognosis.
- antibodies of the invention are used to delay development of a disease or to slow the progression of a disease.
- a neurological drug may be selected that is an analgesic including, but not limited to, anarcotic/opioid analgesic (i.e., morphine, fentanyl, hydrocodone, meperidine, methadone, oxymorphone, pentazocine, propoxyphene, tramadol, codeine and oxycodone), a nonsteroidal anti-inflammatory drug (NSAID) (i.e., ibuprofen, naproxen, diclofenac, diflunisal, etodolac, fenoprofen, flurbiprofen, indomethacin, ketorolac, mefenamic acid, meloxicam, nabumetone, oxaprozin, piroxicam, sulindac, and tolmetin), a corticosteroid (i.e., cortisone, prednisone, prednisolone, dexamethasone
- NSAID non
- a neurological drug may be selected that is an anti- vertigo agent including, but not limited to, meclizine, diphenhydramine, promethazine and diazepam.
- a neurological drug may be selected that is an anti-nausea agent including, but not limited to, promethazine, chlorpromazine, prochlorperazine, trimethobenzamide, and metoclopramide.
- a neurological drug may be selected that is a growth hormone or neurotrophic factor; examples include but are not limited to brain-derived neurotrophic factor (BDNF), nerve growth factor (NGF), neurotrophin-4/5, fibroblast growth factor (FGF)-2 and other FGFs, neurotrophin (NT)-3, erythropoietin (EPO), hepatocyte growth factor (HGF), epidermal growth factor (EGF), transforming growth factor (TGF)-alpha, TGF- beta, vascular endothelial growth factor (VEGF), interleukin-1 receptor antagonist (IL-lra), ciliary neurotrophic factor (CNTF), glial-derived neurotrophic factor (GDNF), neurturin, platelet-derived growth factor (PDGF), heregulin, neuregulin, artemin, persephin, interleukins, glial cell line derived neurotrophic factor (GFR), granulocyte-colony stimulating factor (CSF), granulocyte-
- BDNF brain-
- a neurological drug may be selected that is a chemotherapeutic agent.
- chemotherapeutic agents include alkylating agents such as thi- otepa and CYTOXAN® cyclosphosphamide; alkyl sulfonates such as busulfan, improsulfan and piposulfan; aziridines such as benzodopa, carboquone, meturedopa, and uredopa; ethyl- enimines and methylamelamines including altretamine, triethylenemelamine, trie- tylenephosphoramide, triethiylenethiophosphor-amide and trimethylolomelamine; acetogenins (especially bullatacin and bullatacinone); delta-9-tetrahydrocannabinol (dronabinol, MARINOL®); beta-lapachone; lapachol; colchicines; betulinic acid; a camptothecin (including
- calicheamicin especially calicheamicin gammall and calicheamicin omegall
- dynemicin including dynemicin A; an esperamicin; as well as neocarzinostatin chromophore and related chromoprotein enediyne antiobiotic chromophores), aclacinomysins, actinomycin, authramycin, azaserine, bleomycins, cactinomycin, carabicin, carminomycin, carzinophilin, chromomycinis, dactinomycin, dauno- rubicin, detorubicin, 6-diazo-5-oxo-L-norleucine, ADRIAMYCIN® doxorubicin (including morpholino-doxorubicin, cyanomorph
- chemotherapeutic agents are anti-hormonal agents that act to regulate, reduce, block, or inhibit the effects of hormones that can promote the growth of cancer, and are often in the form of systemic, or whole-body treatment. They may be hormones themselves.
- anti-estrogens and selective estrogen receptor modulators include, for example, tamoxifen (including NOLVADEX® tamoxifen), EVISTA® raloxifene, droloxifene, 4-hydroxytamoxifen, trioxifene, keoxifene, LY 117018, onapristone, and FARESTON® toremifene; anti-progesterones; estrogen receptor down-regulators (ERDs); agents that function to suppress or shut down the ovaries, for example, leutinizing hormone-releasing hormone (LHRH) agonists such as LUPRON® and ELI- GARD® leuprolide acetate, goserelin acetate, buserelin acetate and tripterelin; other anti- androgens such as flutamide, nilutamide and bicalutamide; and aromatase inhibitors that inhibit the enzyme aromatase, which regulates estrogen production in
- LHRH le
- chemotherapeutic agents includes bisphosphonates such as clodronate (for example, BONEFOS® or OSTAC®), DIDROCAL® etidronate, NE-58095, ZOMETA® zoledronic acid/zoledronate, FOSAMAX® alendronate, ARE- DIA® pamidronate, SKELID® tiludronate, or ACTONEL® risedronate; as well as troxacita- bine (a 1,3 -dioxolane nucleoside cytosine analog); antisense oligonucleotides, particularly those that inhibit expression of genes in signaling pathways implicated in aberrant cell proliferation, such as, for example, PKC-alpha, Raf, H-Ras, and epidermal growth factor receptor (EGF-R); vaccines such as THERATOPE® vaccine and gene therapy vaccines, for example, ALLOVECTIN® vaccine, LEUVECTIN® vaccine, and VAXID® vaccine; LUR
- anti-cancer immunoglobulins including, but not limited to, trastuzumab, bevacizumab, alemtuxumab, cetuximab, gemtuzumab ozogamicin, ibritumomab tiuxetan, panitumumab and rituximab.
- antibodies in conjunction with a toxic label may be used to target and kill desired cells (i.e., cancer cells), including, but not limited to, tositumomab with a radiolabel.
- a neurological drug may be selected that is an anti- angiogenic ophthalmic agent (i.e., bevacizumab, ranibizumab and pegaptanib), an ophthalmic glaucoma agent (i.e., carbachol, epinephrine, demecarium bromide, apraclonidine, brimoni- dine, brinzolamide, levobunolol, timolol, betaxolol, dorzolamide, bimatoprost, carteolol, metipranolol, dipivefrin, travoprost and latano prost), a carbonic anhydrase inhibitor (i.e., methazolamide and acetazolamide), an ophthalmic antihistamine (i.e., naphazoline, phenylephrine and tetrahydrozoline), an ocular lubricant, an ophthalmic agent (i.e.,
- felbamate carbonic anhydrase inhibitor anticonvulsants (i.e., acetazolamide, topiramate and zonisamide), dibenzazepine anticonvulsants (i.e., rufinamide, carbamazepine, and oxcarbazepine), fatty acid derivative anticonvulsants (i.e., divalproex and valproic acid), gamma-aminobutyric acid analogs (i.e., pregabalin, gabapentin and vigabatrin), gamma-aminobutyric acid reuptake inhibitors (i.e., ti- agabine), gamma-aminobutyric acid transaminase inhibitors (i.e., vigabatrin), hydantoin anticonvulsants (i.e.
- phenytoin, ethotoin, fosphenytoin and mephenytoin miscellaneous anticonvulsants (i.e., lacosamide and magnesium sulfate), progestins (i.e., progesterone), oxazoli- dinedione anticonvulsants (i.e., paramethadione and trimethadione), pyrrolidine anticonvulsants (i.e., levetiracetam), succinimide anticonvulsants (i.e., ethosuximide and methsuximide), triazine anticonvulsants (i.e., lamotrigine), and urea anticonvulsants (i.e., phenacemide and pheneturide).
- progestins i.e., progesterone
- oxazoli- dinedione anticonvulsants i.e., paramethadione and trimethadione
- a neurological drug may be selected that is itself or otherwise mimics the activity of the enzyme that is impaired in the disease.
- Exemplary recombinant enzymes for the treatment of lysosomal storage disorders include, but are not limited to those set forth in e.g., U.S. Patent Application publication no.
- 2005/0142141 i.e., alpha-L- iduronidase, iduronate-2-sulphatase, N-sulfatase, alpha-N-acetylglucosaminidase, N- acetyl- galactosamine-6-sulfatase, beta-galactosidase, aryl sulphatase B, beta-glucuronidase, acid al- pha-glucosidase, gluco cerebrosidase, alpha-galactosidase A, hexosaminidase A, acid sphingomyelinase, beta-galactocerebrosidase, beta-galactosidase, arylsulfatase A, acid ceramidase, aspartoacylase, palmitoyl-protein thioesterase 1 and trip eptidyl amino peptidase 1).
- a neurological drug may be selected that includes, but is not limited to, an antibody or other binding molecule (including, but not limited to a small molecule, a peptide, an aptamer, or other protein binder) that specifically binds to a target selected from: beta secretase, tau, presenilin, amyloid precursor protein or portions thereof, amyloid beta peptide or oligomers or fibrils thereof, death receptor 6 (DR6), receptor for advanced glycation endproducts (RAGE), parkin, and huntingtin; a cholinesterase inhibitor (i.e., galantamine, donepezil, rivastigmine and tacrine); an NMD A receptor antagonist (i.e., memantine), a monoamine depletor (i.e., tetrab enazine); an ergoloid mesylate; an anticholinergic antiparkinsonism agent (i.e., procyclidine, diphenhydramine, trihe
- a neurological drug may be selected that includes, but is not limited to, an antiviral compound (including, but not limited to, an adamantane antiviral (i.e., rimantadine and amantadine), an antiviral interferon (i.e., peginterferon alfa-2b), a chem- okine receptor antagonist (i.e., maraviroc), an integrase strand transfer inhibitor (i.e., ralte- gravir), a neuraminidase inhibitor (i.e., oseltamivir and zanamivir), a non-nucleoside reverse transcriptase inhibitor (i.e., efavirenz, etravirine, delavirdine and nevirapine), a nucleoside reverse transcriptase inhibitors (tenofovir, abacavir, lamivudine, zidovudine, stavudin
- an antiviral compound including,
- a neurological drug may be selected that includes, but is not limited to, a thrombolytic (i.e., urokinase,reteplase, reteplase and tenecteplase), a platelet aggregation inhibitor (i.e., aspirin, cilostazol, clopidogrel, prasugrel and dipyridamole), a statin (i.e., lovastatin, pravastatin, fiuvastatin, rosuvastatin, atorvastatin, simvastatin, cerivastatin and pitavas- tatin), and a compound to improve blood flow or vascular flexibility, including, e.g., blood pressure medications.
- a thrombolytic i.e., urokinase,reteplase, reteplase and tenecteplase
- a platelet aggregation inhibitor i.e., aspirin, cil
- a neurological drug may be selected from a behavior- modifying compound including, but not limited to, an atypical antipsychotic (i.e., risperidone, olanzapine, apripiprazole, quetiapine, paliperidone, asenapine, clozapine, iloperidone and ziprasidone), a pheno thiazine antipsychotic (i.e., prochlorperazine, chlorpromazine, fluphena- zine, perphenazine, trifluoperazine, thioridazine and mesoridazine), a thioxanthene (i.e., thiothixene), a miscellaneous antipsychotic (i.e., pimozide, lithium, molindone, haloperidol and loxapine), a selective serotonin reuptake inhibitor (i.e., citalopram, e
- a neurological drug may be selected that addresses the inflammation itself (i.e., anon-steroidal anti-inflammatory agent such as ibuprofen or naproxen), or one which treats the underlying cause of the inflammation (i.e., an anti-viral or anti-cancer agent).
- anon-steroidal anti-inflammatory agent such as ibuprofen or naproxen
- an anti-viral or anti-cancer agent i.e., an anti-viral or anti-cancer agent
- the compound is an intact or full-length antibody.
- intact antibodies can be assigned to different classes. There are five major classes of intact antibodies: IgA, IgD, IgE, IgG, and IgM, and several of these may be further divided into subclasses (isotypes), e.g., IgGl, IgG2, IgG3, IgG4, IgA, and IgA2.
- the heavy chain constant domains that correspond to the different classes of antibodies are called a, 6, e, y, and p, respectively.
- the subunit structures and three-dimensional configurations of different classes of immunoglobulins are well known.
- the intact antibody lacks effector function.
- the antibody is a chimeric, humanized, or human antibody or antigen-binding fragment thereof.
- Assays for evaluating uptake of systemically administered multispecific antibody and/or conjugate and other biological activity of multispecific antibody and/or conjugate can be performed as disclosed in the examples or as known for the multispecific antibody and/or conjugate of interest.
- Mesuring the concentration within the parenchyma space of CNS can also be used using for example microdialysis or the capillary depletion method combined with ELISA or radioactivity measurements of labeled multispecific antibody and/or conjugate.
- Therapeutic formulations of the antibody or conjugate used in accordance with the present invention are prepared for storage by mixing with optional pharmaceutically acceptable carriers, excipients or stabilizers ⁇ Remington’s Pharmaceutical Sciences 16th edition, Osol, A. Ed. (1980)), in the form of lyophilized formulations or aqueous solutions.
- Acceptable carriers, excipients, or stabilizers are nontoxic to recipients at the dosages and concentrations employed, and include buffers such as phosphate, citrate, and other organic acids; anti- oxidants including ascorbic acid and methionine; preservatives (such as octadecyldimethylbenzyl ammonium chloride; hexamethonium chloride; benzalkonium chloride, benzetho- nium chloride; phenol, butyl or benzyl alcohol; alkyl parabens such as methyl or propyl paraben; catechol; resorcinol; cyclohexanol; 3-pentanol; and m-cresol); low molecular weight (less than about 10 residues) polypeptides; proteins, such as serum albumin, gelatin, or immunoglobulins; hydrophilic polymers such as polyvinylpyrrolidone; amino acids such as glycine, glutamine, asparagine
- the formulation herein may also contain more than one active compound as necessary, optionally those with complementary activities that do not adversely affect each other.
- the type and effective amounts of such medicaments depend, for example, on the amount of multispecific antibody and/or conjugate present in the formulation, and clinical parameters of the subjects. Exemplary such medicaments are discussed below.
- the active ingredients may also be entrapped in microcapsules prepared, for example, by coacervation techniques or by interfacial polymerization, for example, hydroxymethylcellulose or gelatin-micro capsules and poly-(methylmethacylate) micro capsules, respectively, in colloidal drug delivery systems (for example, liposomes, albumin microspheres, microemulsions, nano-particles and nanocapsules) or in macroemulsions.
- colloidal drug delivery systems for example, liposomes, albumin microspheres, microemulsions, nano-particles and nanocapsules
- Sustained-release preparations may be prepared. Suitable examples of sustained- release preparations include semi-permeable matrices of solid hydrophobic polymers containing the antibody, which matrices are in the form of shaped articles, e.g. films, or microcapsules. Examples of sustained-release matrices include polyesters, hydrogels (for example, poly(2- hydroxyethyl-methacrylate), or poly(vinylalcohol)), polylactides (U.S. Pat. No.
- copolymers of L-glutamic acid and y ethyl-L-glutamate copolymers of L-glutamic acid and y ethyl-L-glutamate, non-degradable ethylene- vinyl acetate, degradable lactic acid-glycolic acid copolymers such as the LUPRON DEPOTTM (injectable microspheres composed of lactic acid-glycolic acid copolymer and leuprolide acetate), and poly-D-(-)-3-hydroxybutyric acid.
- the formulations to be used for in vivo administration must be sterile. This is readily accomplished by filtration through sterile filtration membranes. In one embodiment the formulation is isotonic.
- the multispecific antibody or the conjugate of the invention may be utilized in a variety of in vivo methods.
- the invention provides a method of transporting a therapeutic compound across the BBB comprising exposing multispecific antibody and/or conjugate to the BBB such that the multispecific antibody transports the therapeutic compound coupled thereto across the BBB.
- the invention provides a method of transporting a neurological disorder drug across the BBB comprising exposing the conjugate to the BBB such that the multispecific antibody transports the neurological disorder drug coupled thereto across the BBB.
- the BBB here is in a mammal (e.g. a human), e.g.
- neurological disorder including, without limitation: Alzheimer's disease (AD), stroke, dementia, muscular dystrophy (MD), multiple sclerosis (MS), amyotrophic lateral sclerosis (ALS), cystic fibrosis, Angelman's syndrome, Liddle syndrome, Parkinson's disease, Pick's disease, Paget's disease, cancer, traumatic brain injury, etc.
- neurological disorder is selected from: a neuropathy, amyloidosis, cancer (e.g. involving the CNS or brain), an ocular disease or disorder, a viral or microbial infection, inflammation (e.g. of the CNS or brain), ischemia, neurodegenerative disease, seizure, behavioral disorder, lysosomal storage disease, etc.
- Neuropathy disorders are diseases or abnormalities of the nervous system characterized by inappropriate or uncontrolled nerve signaling or lack thereof, and include, but are not limited to, chronic pain (including nociceptive pain), pain caused by an injury to body tissues, including cancer-related pain, neuropathic pain (pain caused by abnormalities in the nerves, spinal cord, or brain), and psychogenic pain (entirely or mostly related to a psychological disorder), headache, migraine, neuropathy, and symptoms and syndromes often accompanying such neuropathy disorders such as vertigo or nausea.
- chronic pain including nociceptive pain
- pain caused by an injury to body tissues including cancer-related pain
- neuropathic pain pain caused by abnormalities in the nerves, spinal cord, or brain
- psychogenic pain include, but are not limited to, headache, migraine, neuropathy, and symptoms and syndromes often accompanying such neuropathy disorders such as vertigo or nausea.
- Amyloidoses are a group of diseases and disorders associated with extracellular proteinaceous deposits in the CNS, including, but not limited to, secondary amyloidosis, age- related amyloidosis, Alzheimer's Disease (AD), mild cognitive impairment (MCI), Lewy body dementia, Down's syndrome, hereditary cerebral hemorrhage with amyloidosis (Dutch type); the Guam Parkinson-Dementia complex, cerebral amyloid angiopathy, Huntington's disease, progressive supranuclear palsy, multiple sclerosis; Creutzfeld Jacob disease, Parkinson's disease, transmissible spongiform encephalopathy, HIV-related dementia, amyotropic lateral sclerosis (ALS), inclusion-body myositis (IBM), and ocular diseases relating to beta-amyloid deposition (i.e., macular degeneration, drusen-related optic neuropathy, and cataract).
- AD Alzheimer's Disease
- MCI mild cognitive impairment
- Lewy body dementia Down's
- Cancers of the CNS are characterized by aberrant proliferation of one or more CNS cell (i.e., a neural cell) and include, but are not limited to, glioma, glioblastoma multiforme, meningioma, astrocytoma, acoustic neuroma, chondroma, oligodendroglioma, medulloblastomas, ganglioglioma, Schwannoma, neurofibroma, neuroblastoma, and extradural, intramedullary or intradural tumors.
- glioma glioblastoma multiforme
- meningioma astrocytoma
- acoustic neuroma chondroma
- oligodendroglioma oligodendroglioma
- medulloblastomas ganglioglioma
- Schwannoma neurofibroma
- neurofibroma neuroblastoma
- Viral or microbial infections of the CNS include, but are not limited to, infections by viruses (i.e., influenza, HIV, poliovirus, rubella, ), bacteria (i.e., Neisseria sp., Streptococcus sp., Pseudomonas sp., Proteus sp., E. coli, S.
- viruses i.e., influenza, HIV, poliovirus, rubella,
- bacteria i.e., Neisseria sp., Streptococcus sp., Pseudomonas sp., Proteus sp., E. coli, S.
- aureus Pneumococcus sp., Meningococcus sp., Haemophilus sp., and Mycobacterium tuberculosis
- fungi i.e., yeast, Cryptococcus neoformans
- parasites i.e., toxoplasma gondii
- amoebas resulting in CNS pathophysiologies including, but not limited to, meningitis, encephalitis, myelitis, vasculitis and abscess, which can be acute or chronic.
- Inflammation of the CNS is inflammation that is caused by an injury to the CNS, which can be a physical injury (i.e., due to accident, surgery, brain trauma, spinal cord injury, concussion) or an injury due to or related to one or more other diseases or disorders of the CNS (i.e., abscess, cancer, viral or microbial infection).
- an injury to the CNS which can be a physical injury (i.e., due to accident, surgery, brain trauma, spinal cord injury, concussion) or an injury due to or related to one or more other diseases or disorders of the CNS (i.e., abscess, cancer, viral or microbial infection).
- Ischemia of the CNS refers to a group of disorders relating to aberrant blood flow or vascular behavior in the brain or the causes therefor, and includes, but is not limited to: focal brain ischemia, global brain ischemia, stroke (i.e., subarachnoid hemorrhage and intracerebral hemorrhage), and aneurysm.
- Neurodegenerative diseases are a group of diseases and disorders associated with neural cell loss of function or death in the CNS, and include, but are not limited to: adrenoleukodystrophy, Alexander's disease, Alper's disease, amyotrophic lateral sclerosis, ataxia telangiectasia, Batten disease, cockayne syndrome, corticobasal degeneration, degeneration caused by or associated with an amyloidosis, Friedreich's ataxia, frontotemporal lobar degeneration, Kennedy's disease, multiple system atrophy, multiple sclerosis, primary lateral sclerosis, progressive supranuclear palsy, spinal muscular atrophy, transverse myelitis, Refsum's disease, and spinocerebellar ataxia.
- Seizure diseases and disorders of the CNS involve inappropriate and/or abnormal electrical conduction in the CNS, and include, but are not limited to: epilepsy (i.e. , absence seizures, atonic seizures, benign Rolandic epilepsy, childhood absence, clonic seizures, complex partial seizures, frontal lobe epilepsy, febrile seizures, infantile spasms, juvenile myoclonic epilepsy, juvenile absence epilepsy, Lennox-Gastaut syndrome, Landau-Kleffher Syndrome, Dravef s syndrome, Otahara syndrome, West syndrome, myoclonic seizures, mitochondrial disorders, progressive myoclonic epilepsies, psychogenic seizures, reflex epilepsy, Rasmussen's Syndrome, simple partial seizures, secondarily generalized seizures, temporal lobe epilepsy, tonicionic seizures, tonic seizures, psychomotor seizures, limbic epilepsy, partialonset seizures, generalized-onset seizures, status epilepticus, abdominal epilepsy, akinetic seizures, autonomic seizures, massive bilateral myo
- Behavioral disorders are disorders of the CNS characterized by aberrant behavior on the part of the afflicted subject and include, but are not limited to: sleep disorders (i.e., insomnia, parasomnias, night terrors, circadian rhythm sleep disorders, and narcolepsy), mood disorders (i.e., depression, suicidal depression, anxiety, chronic affective disorders, phobias, panic attacks, obsessive-compulsive disorder, attention deficit hyperactivity disorder (ADHD), attention deficit disorder (ADD), chronic fatigue syndrome, agoraphobia, post-traumatic stress disorder, bipolar disorder), eating disorders (i.e., anorexia or bulimia), psychoses, developmental behavioral disorders (i.e., autism, Ret's syndrome, Aspberger's syndrome), personality disorders and psychotic disorders (i.e., schizophrenia, delusional disorder, and the like).
- sleep disorders i.e., insomnia, parasomnias, night terrors, circadian rhythm sleep disorders, and narcolepsy
- mood disorders i.
- Lysosomal storage disorders are metabolic disorders which are in some cases associated with the CNS or have CNS-specific symptoms; such disorders include, but are not limited to: Tay-Sachs disease, Gaucher's disease, Fabry disease, mucopolysaccharidosis (types I, II, III, IV, V, VI and VII), glycogen storage disease, GM1 -gangliosidosis, metachromatic leukodystrophy, Farber's disease, Canavan's leukodystrophy, and neuronal ceroid lipofuscinoses types 1 and 2, Niemann-Pick disease, Pompe disease, and Krabbe's disease.
- the multispecific antibody and/or conjugate of the invention can be used either alone or in combination with other agents in a therapy.
- the multispecific antibody and/or conjugate of the invention may be co-administered with at least one additional therapeutic agent.
- an additional therapeutic agent is a therapeutic agent effective to treat the same or a different neurological disorder as the multispecific antibody and/or conjugate of the invention is being employed to treat.
- Exemplary additional therapeutic agents include, but are not limited to: the various neurological drugs described above, cholinesterase inhibitors (such as donepezil, galantamine, rovastigmine, and tacrine), NMDA receptor antagonists (such as memantine), amyloid beta peptide aggregation inhibitors, antioxidants, y-secretase modulators, nerve growth factor (NGF) mimics or NGF gene therapy, PPARy agonists, HMS-CoA reductase inhibitors (statins), ampakines, calcium channel block- ers, GABA receptor antagonists, glycogen synthase kinase inhibitors, intravenous immunoglobulin, muscarinic receptor agonists, nicrotinic receptor modulators, active or passive amyloid beta peptide immunization, phosphodiesterase inhibitors, serotonin receptor antagonists and anti-amyloid beta peptide antibodies.
- the at least one additional therapeutic agent is selected for its ability to mitigate one or more side effects of
- Such combination therapies noted above encompass combined administration (where two or more therapeutic agents are included in the same or separate formulations), and separate administration, in which case, administration of the multispecific antibody and/or conjugate of the invention can occur prior to, simultaneously, and/or following, administration of the additional therapeutic agent and/or adjuvant.
- Multispecific antibodys and/or conjugates of the invention can also be used in combination with other interventional therapies such as, but not limited to, radiation therapy, behavioral therapy, or other therapies known in the art and appropriate for the neurological disorder to be treated or prevented.
- the multispecific antibody and/or conjugate of the invention (and any additional therapeutic agent) can be administered by any suitable means, including parenteral, intrapulmonary, and intranasal, and, if desired for local treatment, intralesional administration.
- Parenteral infusions include intramuscular, intravenous, intraarterial, intraperitoneal, or subcutaneous administration.
- Dosing can be by any suitable route, e.g. by injections, such as intravenous or subcutaneous injections, depending in part on whether the administration is brief or chronic.
- Various dosing schedules including but not limited to monovalent or multiple administrations over various time- points, bolus administration, and pulse infusion are contemplated herein.
- Multispecific antibody and/or conjugates of the invention would be formulated, dosed, and administered in a fashion consistent with good medical practice.
- Factors for consideration in this context include the particular disorder being treated, the particular mammal being treated, the clinical condition of the individual patient, the cause of the disorder, the site of delivery of the agent, the method of administration, the scheduling of administration, and other factors known to medical practitioners.
- the multispecific antibody and/or conjugates of the invention need not be, but is optionally formulated with one or more agents currently used to prevent or treat the disorder in question. The effective amount of such other agents depends on the amount of multispecific antibody and/or conjugate present in the formulation, the type of disorder or treatment, and other factors discussed above. These are generally used in the same dosages and with administration routes as described herein, or about from 1 to 99% of the dosages described herein, or in any dosage and by any route that is empirically/clinically determined to be appropriate.
- the appropriate dosage of multispecific antibody and/or conjugate of the invention (when used alone or in combination with one or more other additional therapeutic agents) will depend on the type of disease to be treated, the type of multispecific antibody and/or conjugate, the severity and course of the disease, whether the antibody is administered for preventive or therapeutic purposes, previous therapy, the patient's clinical history and response to the multispecific antibody and/or conjugate, and the discretion of the attending physician.
- the multispecific antibody and/or conjugate is suitably administered to the patient at one time or over a series of treatments. Depending on the type and severity of the disease, about 1 pg/kg to 15 mg/kg (e.g.
- 0.1 mg/kg- lOmg/kg) of multispecific antibody and/or conjugate can be an initial candidate dosage for administration to the patient, whether, for example, by one or more separate administrations, or by continuous inmultimeric.
- One typical daily dosage might range from about 1 pg/kg to 100 mg/kg or more, depending on the factors mentioned above.
- the treatment would generally be sustained until a desired suppression of disease symptoms occurs.
- One exemplary dosage of the antibody would be in the range from about 0.05 mg/kg to about 10 mg/kg.
- one or more doses of about 0.5 mg/kg, 2.0 mg/kg, 4.0 mg/kg or 10 mg/kg (or any combination thereof) may be administered to the patient.
- Such doses may be administered intermittently, e.g. every week or every three weeks (e.g. such that the patient receives from about two to about twenty, or e.g. about six doses of the antibody).
- An initial higher loading dose, followed by one or more lower doses may be administered.
- other dosage regimens may be useful. The progress of this therapy is easily monitored by conventional techniques and assays.
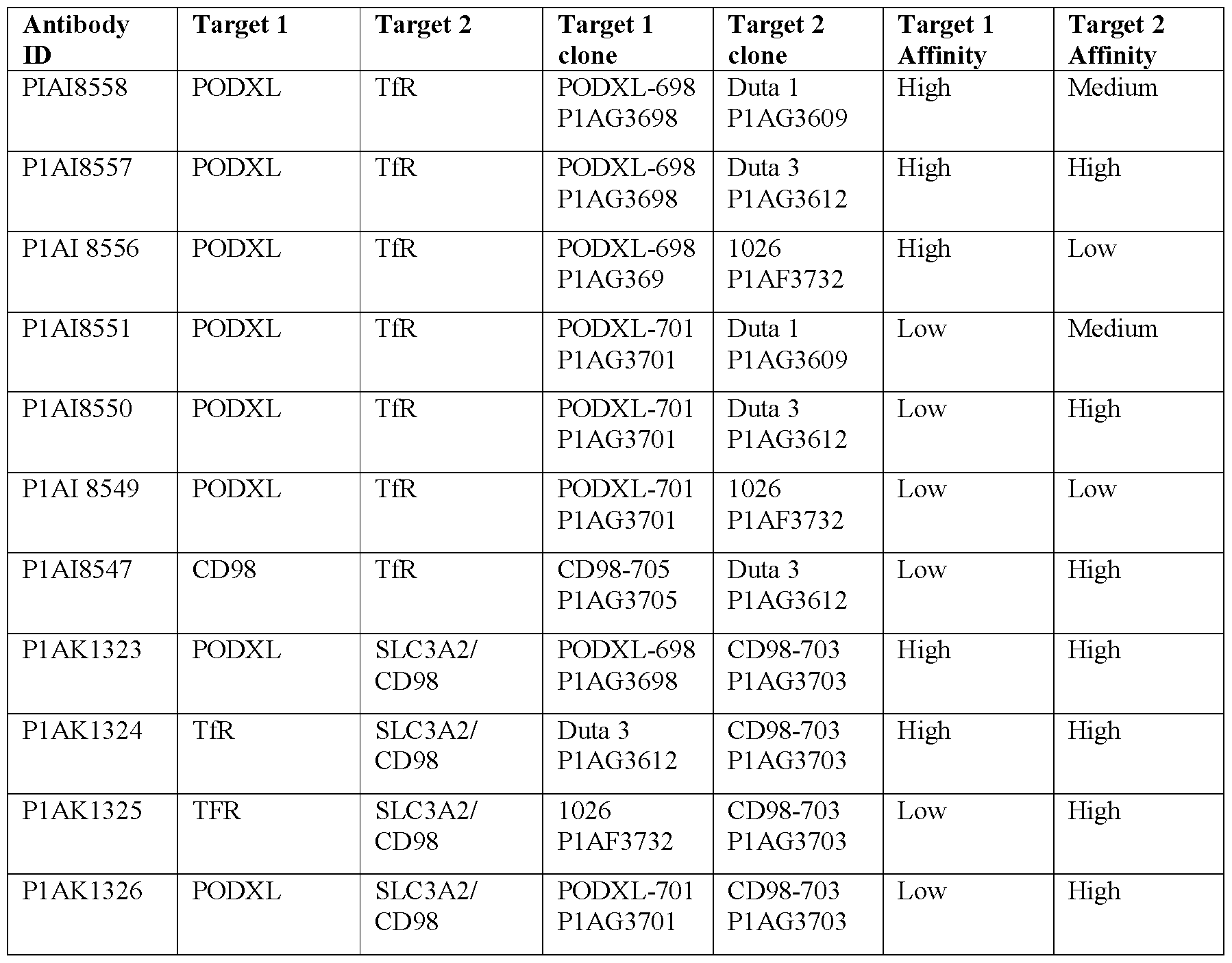
- Target 1 is recognized by Light chain A and Heavy chain K
- Target 2 is recognized by Light chain B and Heavy chain H Table 2: Bispecific antibodies with target clone affinities
- Figure 2a shows the results of the binding assay on hCMECD3 for bispecific antibodies P1AI8556 (PODXL-698-high/TfR-1026-low), P1AI8559 (PODXL-698-high/Dummy), P1AI8549 (PODXL-701 -low/TfR-1026-low), P1AI8553 (Dummy/TfR-1026-low) and P1AI8552 (PODXL-701-low/Dummy).
- the binding assay showed highest binding to hCMECD3 cells for P1AI8556 (698-PODXL - high/TFR - low).
- TFR- low with 698-PODXL-high increased the binding properties compared to both monospecific binder controls P1AI8559 (698-PODXL-high/Dummy) and P1AI8553 (1026-TfR- low/Dummy).
- Figure 2b shows the results of the binding assay on hCMECD3 for bispecific antibodies P1AI8557 (PODXL - 698 - high / TfR- Duta 3- high), P1AI8550 (PODXL-701-low/ TfR- Duta 3- low) and Pl AI8547 (CD98-705-low/TfR-Duta 3-high).
- the binding assay showed highest binding to hCMECD3 cells for Pl AI8557.
- the combination of PODXL - 698 - high / TfR- Duta 3- high increased the binding properties compared to both monospecific binder controls P1AI8559 (698-PODXL/Dummy).
- Figure 2c shows the results of the binding assay on hCMECD3 for bispecific antibodies P1AI8558 (PODXL-698-high / TfR- Duta 1- medium), P1AI8559 (PODXL-698-high/ Dummy), P1AI8555 (Dummy/TfR-Duta 1-medium), P1AI8551 (PODXL-701-low/TfR-Duta 1 -medium), P1AI8552 (PODXL-701-low/Dummy), P1AG3698 (PODXL-698-high- onearmed) and P1AG3701 (PODXL-701 -low-onearmed).
- P1AI8558 PODXL-698-high / TfR- Duta 1- medium
- P1AI8559 PODXL-698-high/ Dummy
- P1AI8555 Dummy/TfR-Duta 1-medium
- P1AI8551 PODXL-701-low/TfR-
- the binding assay showed highest binding to hCMECD3 cells for Pl AG3698, Pl AI8559 and Pl AI8558.
- the combination of PODXL-698-high / TfR-Duta 1-medium increased the binding properties compared to the monospecific TfR binder control P1AI8555 (Dummy/Duta- 1-medium) but not to the PODXL binder controls P1AI8558 (PODXL-698-high / TfR- Duta 1- medium) and P1AG3698 (PODXL-698-high-onearmed).
- PODXL-701 -low / TfR-Duta 1-medium increased the binding affinity compared to both monospecific controls P1AI8552 (PODXL- 701 -low/Dummy) and P1AI8555 (Dummy/TfR-Duta 1-medium).
- Figure 2d shows the results of the binding assay on hCMECD3 for bispecific antibodies P1AK1324 (TfR-Duta 3 -high/CD98 -703 -high) and P1AK1327 (Dummy/CD98-703-high).
- the binding assay showed the highest binding to hCMECD3 cells for Pl AK1327 (Dummy/CD98-703-high).
- the combination of TfR-Duta 3-high/CD98-703-high P1AK1324) decreased the binding affinity compared to the monospecific control P1AK1327 (Dummy/CD98-703-high)
- FIG. 3 shows the results of the binding assay on hCMEDCD3 for bispecific antibodies P1AI8549 (PODXL-701 -low/TfR-1026-low), P1AI8553 (Dummy/TfR) and P1AI8552 (PODXL-701 -low/Dummy).
- Pl AI8553 (1026-TFR-low/Dummy) showed the lowest binding to hCMECD3 cells.
- this Tfr- binder is combined with the 701-PODXL binder this increase the binding strength: Pl AI8549 (701-PODXL-low/1026-TFR-low).
- P1AI8552 (701-PODXL-low/Dummy) showed as a monospecific binder higher binding strength than the bispecific Pl AI8549 (PODXL-701- low/TfR-1026-lo w).
- Figure 4 shows the results of the binding assay on hCMEDCD3 for bispecific antibodies P1AI8559 (PODXL-698-high/Dummy), P1AG3701 (PODXL-701 -low/None), P1AG3698 (PODXL-698-high/None), P1AI8552 (PODXL-701 -low/Dummy), P1AG3705 (CD98-705- low), P1AI4681 and P1AI3732.
- the one-arm version of 698-PODXL-high behaves similarly to the 2-arm version where 698-PODXL-high is combined with a non-binder (P1AI8559 (PODXL-698-high/Dummy). From that one can conclude, that the shown effects are not due to the antibody format (one arm vs. two arm IgG).
- Figure 6a and 6b show an overview of the transcytosis assay results for the following bispecific antibodies and control antibodies: P1AI8549 (PODXL-701-low/TfR-1026-low), P1AI8552 (PODXL-701/Dummy), P1AI8553 (Dummy/TfR.-1026-low), P1AI8556 (PODXL- 698-high/TfR-1026-low) agree P1AI8559 (PODXL-698-high/Dummy), P1AK1323 (PODXL-698- high/CD98-703 -high) , P1AK1326 (PODXL-701-low/CD98-703-high), P1AK1327 (Dummy/CD98-703-high), and P1AK1325 (TfR-1026-low/CD98-703-high).
- Experimental setup hCMECD3; loading: 20.1 pg/ml, conjugate 2.67 pg/ml.
- Figure 6c shows the results of the transcytosis assay for a selection of bispecific antibodies, namely: P1AI8549 (PODXL-701-low/TfR-1026-low), P1AI8556 (PODXL-698- high/TfR.-1026-low), P1AK1323 (PODXL-698-high/CD98-703-high) and P1AK1325 (TfR- 1026-low/CD98-703-high).
- Experimental setup hCMECD3; loading: 20.1 pg/ml, conjugate 2.67 pg/ml.
- Figure 6d shows the results of the transcytosis assay for a selection of bispecific antibodies, namely: P1AI8552 (PODXL-701/Dummy), P1AI8553 (Dummy/TfR.-1026-low), P1AI8559 (PODXL-698-high/Dummy) and P1AK1327 (Dummy/CD98-703-high).
- Experimental setup hCMECD3; loading: 20.1 pg/ml, conjugate 2.67 pg/ml.
- Fig. 6e shows the results of the transcytosis assay for bispecific antibody P1AI8549 (PODXL-701-low/Tfr-1026-low) + Controls.
- Experimental setup hCMECD3; loading: 20.1 pg/ml, conjugate 2.67 pg/ml.
- Fig. 6f shows the results of the transcytosis assay for bispecific antibody P1AI8550 (PODXL-701-low/TfR-1026-low) + Controls.
- Experimental setup hCMECD3; loading: 20.1 pg/ml, conjugate 2.67 pg/ml.
- Fig. 6g shows the results of the transcytosis assay for bispecific antibody Pl AI8551 (CD98-705-low/TfR-duta 1 - medium) + Controls.
- Fig. 6h shows the results of the transcytosis assay for bispecific antibody P 1 AI8547 (PODXL-701-low/Tfr-Duta 3 - high) + Controls.
- Fig. 6i shows the results of the transcytosis assay for bispecific antibody Pl AI8557 (PODXL-698-high/Tfr-Duta 3 - high) + Controls.
- Fig. 6j shows the results of the transcytosis assay for bispecific antibody Pl AI8558 (PODXL-698-high/TfR.-Duta 1 - medium) + Controls.
- Fig. 6k shows the results of the transcytosis assay for bispecific antibody PIAK1324 (TfR-Duta 3 - high/CD98 - 703-high) + Controls.
- P1AI8556 (PODXL-698-high/TfR-1026-low) shows increased transcytosis compared to both monospecific controls: Pl AI8553 (1026-TFR-low/Dummy) and Pl AI8559 (698-PODXL- high/Dummy).
- P1AK1323 (PODXL-698-high/CD98-703-high), where P1AK1327 (Dummy/CD98-703 -high) represents the monospecific 703-CD98 control and P1AI8559 (698-PODXL-high/Dummy) the other control.
- P1AK1326 (PODXL-701-low/CD98 -703 -high), shows increased transcytosis compared to the control P1AI8552 (701-PODXL-low/Dummy).
- P1AK1325 (1026-TFR-low/ CD98-703-high) shows increased transcytosis compared to the control P1AI8553 (1026-TFR-low/Dummy)
- P1AI8549 (701-PODXL-low/1026-TFR-low) shows increased transcytosis compared to the control P1AI8552 (701-PODXL-low/Dummy).
- Figure 7 shows the results of the transcytosis assay results for bispecific antibodies targeting TfR/PODXL TfR/CD98 and PODXL/CD98 after 5h trafficking.
- Pl AK1325 (1026-TFR-low/ CD98 -703 -high) shows the highest transcytosis, followed by P1AK1323 (698-PODXL-high/703-CD98-high).
- P1AK1326 (701- PODX-lowL/703-CD98-high
- P1AI8559 (698-PODXL-high/Dummy
- P1AI8556 (698- PODXL-high/1026-TFR)-low show also high levels of transcytosis.
- Figure 8 shows the results of the transcytosis assay for bispecific antibodies targeting TfR/PODXL TfR/CD98 and PODXL/CD98 after 20h trafficking. After 20h of intracellular transport Pl AK1323 (698-PODXL-high/703-CD98-high) shows the highest transcytosis, followed by P1AK1325 (1026-TFR-low/ CD98-703-high).
- P1AK1326 (701-PODXL-low/703-CD98-high), Pl AI8559 (698- PODXL-high/Dummy) and P1AI8556 (698-PODXL-high/1026-TFR-low) show also high levels of transcytosis.
- Figure 13 shows the results of the transcytosis assay for monospecific antibodies targeting PODXL.
- Pl AG3700 shows the highest transcytosis.
- Expi293 cells were transiently transfected in a 1.0 L production scale with plasmids in a ratio of 1 : 1 : 1 : 1 encoding the protein. Protein was purified by affinity chromatography
- hCMECD3 cells were incubated for Ih with the antibodies at concentrations of 1000 nM, 500 nM, 250 nM, 125 nM, 63 nM, 31 nM, 16 nM, 8 nM, 4 nM, 2 nM and 1 nM.
- the plate Before seeding the cells, the plate was filled with prewarmed cell culture medium (EBM-2 Basal medium; Lonza; CC-3156 and EGM-2 MV SingleQuots; Lonza; CC-4147), 1.6 ml basolateral and 0.4 ml apical.
- EBM-2 Basal medium Lonza; CC-3156 and EGM-2 MV SingleQuots; Lonza; CC-4147
- HCMECD3 cells were thawed and 8000 cells/filter were seeded on the apical part of the transwell insert and cultured at 37 °C and 5 % CO2 for 12 days with changing the medium every 2-3 days.
- the samples were measured via ELISA.
- 384-well maxisorp plates (Thermo Scientific; 460372) were coated with 30 pl of AffiniPure Goat Anti-Human IgG Fcy-specific coating antibody (BIOZOL; 109-005-098) in a concentration of 1 pg/ml overnight at 4 °C.
- the coating antibody was removed and replaced by 100 pl PBS w/o Ca2+ & Mg2+ (PAN Biotech; P04-991500) + 0,5 % BSA (Thermo Scientific; 37525) + 0,05 % Tween 20 (anatrace; T1003).
- a standard curve of the respective antibody was prepared in EBM-2 Basal medium (Lonza; CC-3156) or RIPA buffer (Sigma Aldrich; R0278) with concentrations of 0 ng/ml, 0.0004 ng/ml, 0.0011 ng/ml 0.004 ng/ml, 0.011 ng/ml, 0.03 ng/ml, 0.1 ng/ml, 0.3 ng/ml, 0.9 ng/ml, 2.77 ng/ml, 8.33 ng/ml and 25 ng/ml.
- microcavities of 600 pm in diameter and 720 pm in depth imprinted in polyethylene glycol (PEG) hydrogels were typically used (GRI3D® 96-well plate, SunBioscience).
- HA Astrocyte Growth Medium
- AGM Astrocyte Growth Medium
- AM Astrocyte Medium
- AGS Astrocytes Growth Supplement
- PGM Pericyte Growth Medium
- PM Pericytes Medium
- PGS Pericytes Growth Supplement
- hCMEC/D3, Merck Human cerebral microvascular endothelial cells
- EBM-2 Endothelial Basal Medium
- hydrocortisone GA-1000, 2% FBS, hEGF, VEGF, hFGF-B, R3-IGF-1, ascorbic acid and heparin
- EBM-2 SingleQuots Supplements Lonza
- Cells were grown in T-75 flasks, coated with 2 pg/cm2 of polyl-lysine for HBVP and HA. The growth medium was changed every two days.
- SM Spheroid Medium
- HA, HBVP were detached by 0.05% trypsin/EDTA (ThermoFisher Scientific) and hCMEC/D3 with 0.25% trypsin/EDTA and resuspended in warm SM.
- the cells were resuspended at the appropriate concentration to target 1000 cells per micro well in a 1 : 1 : 1 ratio (i.e. a total of 3000 cells per micro well) in a seeding volume of 60 pL per well. 150 uL of media was added after 20 min.
- the cells were grown in a humidified incubator at 37 °C with 5% CO2 for 48 h (with a medium refresh after 24h) to allow self-assembly of the multicellular spheroids
- BBB spheroid arrays were incubated with the tested antibodies targeting the human transferrin receptor and a non-targeting human IgG as a control in SM for 4 h at 37 °C with 5% CO2. After incubation, BBB spheroids were washed thoroughly 6 times for 5 min with pre-warmed SM in the incubator and then they were fixed in 4% paraformaldehyde (PF A) for 20 min at room temperature (RT). Samples were washed thoroughly with DPBS and permeabilized and blocked with 0.6% Triton-X + 10% donkey serum in DPBS for 1 h at RT.
- PF A paraformaldehyde
- BBB spheroids were flushed from the plate with washing buffer (DPBS with 0.1% Triton-X) and transferred into 1.5-mL Protein LoBind tubes. After 2-3 min, when spheroids were settled to the bottom of the tube, supernatant was removed.
- Anti-Human FcY (H+L) secondary antibody Jackson ImmunoResearch 709-545-098; 488 fluorescently labelled
- DAPI 1 pg/mL, Sigma-Aldrich
- Spheroids were imaged using a Leica Microsystems, Thunder Imager 3D Assay with a 20 x /0.55 Ph2 dry objective.
- the images were acquired with a 2x2 binning in a 16 bit format.
- Two different channels were set up — one for DAPI (UV laser), and one that matches the fluorophore of the secondary antibody used, in this case AlexaFluor 488 (488 laser).
- a z- stack covering a total depth of 8.5 pm, using 8 steps with the core placed at the center (1.21 pm step size) were used.
- the macro opens an individual z-stack.
- the macro converts the multi-channel z-stack into a multi-channel maximum projection image. It then splits the multi-channel maximum projection image into individual channel images, and takes the DAPI maximum projection image to create a mask via thresholding.
- the macro then converts the mask into a region of interest (ROI) based on its size and shape.
- ROI region of interest
- the ROIs are then reduced to 75%, to cover only the core of the spheroid, and exclude measurements from the endothelial surface of the spheroid.
- the shrunk ROIs are overlaid on top of the channel of interest (488 channel), and relevant measurements are calculated. Fluorescence intensity is reported per pm2, by dividing raw integrated density over area (pm2).
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Immunology (AREA)
- General Health & Medical Sciences (AREA)
- Biophysics (AREA)
- Biochemistry (AREA)
- Genetics & Genomics (AREA)
- Medicinal Chemistry (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Life Sciences & Earth Sciences (AREA)
- Peptides Or Proteins (AREA)
Abstract
The present invention relates to a multispecific antibody for use in transporting a com- pound across the Blood Brain Barrier (BBB), wherein the antibody binds to at least two target proteins selected from the group consisting of transferrin receptor (TFRC), CD98 (SLC3A2) and PODXL.
Description
Multispecific antibodies
FIELD OF THE INVENTION
The present invention relates to multispecific antibodies binding to target proteins expressed on the Blood Brain Barrier (BBB) and their use to transport compounds across the BBB.
BACKGROUND
Brain penetration of neurological disorder drugs such as e.g. large biotherapeutic drugs or small molecule drugs having a low brain penetration, is strictly limited by the extensive and impermeable blood-brain-barrier (BBB) together with the other cell component in the neurovascular unit (NVU). Many strategies to overcome this obstacle have been tested and one is to utilize transcytosis pathways mediated by endogenous receptors expressed on the brain capillary endothelium (blood-brain-barrier- receptor). Recombinant proteins such as monoclonal antibodies or peptides have been designed against these receptors to enable receptor-mediated delivery of biotherapeutics to the brain. However, strategies to maximize brain uptake while minimizing miss-sorting within the brain endothelial cells (BECs), and the extent of accumulation within certain organelles (especially organelles that leads to degradation of the biotherapeutic) in BECs, remain unexplored.
Monoclonal antibodies and other biotherapeutics have huge therapeutic potential for treatment of pathology in the central nervous system (CNS). However, their route into the brain is prevented by the BBB. Previous studies have illustrated that a very small percentage (approximately 0.1 %) of an IgG injected in the bloodstream are able to penetrate into the CNS compartment (Felgenhauer, Klin. Wschr. 52 (1974) 1158-1164). This will certainly limit any pharmacological effect due to the low concentration within CNS of the antibody.
It was previously found that the percentage of the antibody that distributes into the CNS could be improved by exploiting BBB receptors (i.e., transferrin receptor, insulin receptor and the like) (see, e.g., WO 95/02421).
Therefore, there is a need for delivery systems of neurological disorder drugs across the BBB to shuttle the drugs into the brain efficiently.
In WO 2014/033074 a blood-brain-barrier shuttle is reported.
In WO 2014/189973 anti-transferrin receptor antibodies and methods of use are reported. It is further reported that targeting a BBB receptor with a traditional specific high-af- finity antibody generally resulted in limited increase in BBB transport. It was later found that the magnitude of antibody uptake into and distribution in the CNS is inversely related to its binding affinity for the BBB receptor amongst the anti-BBB antibodies studied. For example, a low-affinity antibody to transferrin receptor (TfR) dosed at therapeutic dose levels greatly improves BBB transport and CNS retention of the anti-TfR antibody relative to a higher- affinity anti-TfR antibody, and makes it possible to more readily attain therapeutic concentrations in the CNS (Atwal et al., Sci. Transl. Med. 3 (2011) 84ra43). Proof of such BBB transport was achieved using a bispecific antibody that binds both TfR and the amyloid precursor protein (APP) cleavage enzyme, -secretase (BACE1). A single systemic dose of the bispecific anti-TfR/BACEl antibody engineered using a low-affinity antibody not only resulted in significant antibody uptake in brain, but also dramatically reduced levels of brain A i-40 com-
pared to monospecific anti- BACE1 alone, suggesting that BBB penetrance affects the potency of anti-BACEl (Atwal et al., Sci. Transl. Med. 3 (2011) 84ra43; Yu et al., Sci. Transl. Med. 3 (2011) 84ra44).
There is a need for delivery systems of compounds across the BBB to shuttle the compounds into the brain efficiently.
SUMMARY
In a first aspect, the present invention provides a multispecific antibody for use in transporting a compound across the Blood Brain Barrier (BBB), wherein the antibody binds to at least two target proteins selected from the group consisting of transferrin receptor (TFRC), TfR, CD98 (SLC3A2) and PODXL.
In an embodiment of the present invention, the antibody binds to TfR and CD98.
In an embodiment of the present invention, the antibody binds to TfR and PODXL.
In an embodiment of the present invention, the antibody binds to CD98 and PODXL.
In an embodiment of the present invention, the antibody is a human antibody.
In an embodiment of the present invention, the compound is a therapeutic compound.
In an embodiment of the present invention, the antibody is coupled to the therapeutic compound.
In an embodiment of the present invention, the target proteins are human proteins.
In an embodiment of the present invention, the antibody comprises a first antigen binding site which binds to TfR and a second antigen binding site which binds to CD98.
In an embodiment of the present invention, the antibody comprises a first antigen binding site which binds to TfR and a second antigen binding site which binds to PODXL.
In an embodiment of the present invention, the antibody comprises a first antigen binding site which binds to CD98 and a second antigen binding site which binds to PODXL.
In an embodiment of the present invention, the therapeutic compound is a neurological disorder drug.
In an embodiment of the present invention, the therapeutic compound forms one portion of the multispecific antibody.
In an embodiment of the present invention, the therapeutic compound forms one or two antigen binding sites of the multispecific antibody.
In an embodiment of the present invention, the therapeutic compound which forms one or two antigen binding site of the multispecific antibody recognizes a brain antigen.
In an embodiment of the present invention, the brain antigen is selected from the group consisting of beta-secretase 1 (BACE1), Abeta, epidermal growth factor receptor (EGFR), human epidermal growth factor receptor 2 (HER2), tau, apolipoprotein E (ApoE), alpha-synuclein, CD20, huntingtin, prion protein (PrP), leucine rich repeat kinase 2 (LRRK2), parkin, presenilin 1, presenilin 2, gamma secretase, death receptor 6 (DR6), amyloid precursor protein (APP), p75 neurotrophin receptor (p75NTR), TREM2, MS4A, TrkB, and caspase 6.
In an embodiment of the present invention, the antibody has a monovalent binding mode for each target protein.
In an embodiment of the present invention, the TfR binding site has an affinity KD (nM) in the range of 100 - 1000 and the CD98 binding site has an affinity KD (nM) in the range of 0.1 - 100.
In an embodiment of the present invention, the TfR binding site has an affinity KD (nM) in the range of 100 - 1000 and the PODXL binding site has an affinity KD (nM) in the range ofO.l - 100.
In an embodiment of the present invention, the PODXL binding site has an affinity KD (nM) in the range of 0.1 - 100 and the CD98 binding site has an affinity KD (nM) in the range ofO.l - 100.
In an embodiment of the present invention, the TfR binding site has an affinity KD (nM) in the range of 0.01 - 9 and the CD98 binding site has an affinity KD (nM) in the range of 0.1 - 100.
In a second aspect, the present invention provides an antibody for use in therapy, wherein the antibody is transporting a compound across the Blood Brain Barrier (BBB), wherein the antibody binds to at least the target protein PODXL.
In an embodiment of the present invention, the antibody is a human antibody.
In an embodiment of the present invention, the compound is a therapeutic compound.
In an embodiment of the present invention, the antibody is coupled to the therapeutic compound.
In an embodiment of the present invention, the target proteins are human proteins.
In an embodiment of the present invention, the therapeutic compound is a neurological disorder drug.
In an embodiment of the present invention, the antibody is a multipecific antibody and the therapeutic compound forms one portion of the multispecific antibody.
In an embodiment of the present invention, the therapeutic compound forms one or two antigen binding sites of the multispecific antibody.
In an embodiment of the present invention, the therapeutic compound which forms one antigen binding site of the multispecific antibody recognizes a brain antigen.
In an embodiment of the present invention, the brain antigen is selected from the group consisting of beta-secretase 1 (BACE1), Abeta, epidermal growth factor receptor (EGFR), human epidermal growth factor receptor 2 (HER2), tau, apolipoprotein E (ApoE), alpha-synuclein, CD20, huntingtin, prion protein (PrP), leucine rich repeat kinase 2 (LRRK2), parkin, presenilin 1, presenilin 2, gamma secretase, death receptor 6 (DR6), amyloid precursor protein (APP), p75 neurotrophin receptor (p75NTR), TREM2, MS4A, TrkB, and caspase 6.
In an embodiement of the present invention, the antibody has a monovalent binding mode for each target protein.
SHORT DESCRIPTION OF THE FIGURES
Fig. 1: overview of the bispecific antibodies of the invention
Fig. 2 - 4: Dose response of the bispecific antibodies on hCMECD3 cells:
Fig. 5: 2D transcytosis assay setup
Fig. 6a -k: Transcytosis & recycling of the bispecific antibodies of the present invention
Fig. 7a - d: Comparison of maximum transcytosis of bispecific antibodies directed to TfR/PODXL TfR/CD98 and PODXL/CD98 after 5 hours.
Fig. 8:Comparison of maximum transcytosis of bispecific antibodies directed to TfR/PODXL TfR/CD98 and PODXL /CD98 after 20 hours.
Fig. 9a- c: Experimental setup of human BBB spheroids
Fig. 10: Results of human BBB spheroids
Fig. Ila - d: signal quantification in human BBB spheroids. DP47 is a dummy binder.
Fig. 12: Graphic overview of transcytosis rate for the bispecific antibodies of the invention: P1AI8549 (701-PODXL-low/1026-TFR-low) shows increased transcytosis compared to the controls P1AI8552 (701-PODXL-low/Dummy), P1AI8553 (1026-TFR-low/Dummy) and to the compared one-arm brain shuttle Pl AF3732 (1026-TFR-low).
Fig. 13: Transcytosis and recycling of monospecific PODXL antibodies.
DETAILED DESCRIPTION OF EMBODIMENTS OF THE INVENTION
DEFINITIONS
The "blood-brain barrier" or "BBB" refers to the physiological barrier between the peripheral circulation and the brain and spinal cord which is formed by tight junctions within the brain capillary endothelial plasma membranes, creating a tight barrier that restricts the transport of molecules into the brain, even very small molecules such as urea (60 Daltons). The BBB within the brain, the blood-spinal cord barrier within the spinal cord, and the blood- retinal barrier within the retina are contiguous capillary barriers within the CNS, and are herein collectively referred to an the blood-brain barrier or BBB. The BBB also encompasses the blood-CSF barrier (choroid plexus) where the barrier is comprised of ependymal cells rather than capillary endothelial cells.
The knobs into holes dimerization modules and their use in antibody engineering are described in Carter P.; Ridgway J.B.B.; Presta L.G.: Immunotechnology, Volume 2, Number 1, February 1996 , pp. 73-73(1)).
The "central nervous system" or "CNS" refers to the complex of nerve tissues that control bodily function, and includes the brain and spinal cord.
A "blood-brain barrier receptor" (abbreviated "R/BBB" herein) is an extracellular membrane-linked receptor protein expressed on brain endothelial cells which is capable of transporting molecules across the BBB or be used to transport exogenous administrated molecules. Examples of R/BBB herein include: transferrin receptor (TfR), insulin receptor, insulinlike growth factor receptor (IGF-R), low density lipoprotein receptors including without limitation low density lipoprotein receptor-related protein 1 (LRP1) and low density lipoprotein receptor-related protein 8 (LRP8), and heparin-binding epidermal growth factor-like growth factor (HB-EGF). An exemplary R/BBB herein is transferrin receptor (TfR).
“Affinity” refers to the strength of the sum total of nonco valent interactions between a single binding site of a molecule (e.g., an antibody) and its binding partner (e.g., an antigen). Unless indicated otherwise, as used herein, “binding affinity” refers to intrinsic binding affinity which reflects a 1 :1 interaction between members of a binding pair (e.g., antibody and antigen). The affinity of a molecule X for its partner Y can generally be represented by the dissociation constant (KD). Affinity can be measured by common methods known in the art. A preferred method for measuring affinity is Surface Plasmon Resonance (SPR).
The "monovalent binding entity" refers to a molecule able to bind specifically and in a monovalent binding mode to an R/BBB. The blood brain shuttle and/or conjugate of the present invention are characterized by the presence of a single unit of a monovalent binding entity i.e. the blood brain shuttle and/or conjugate of the present invention comprise one unit of the monovalent binding entity. The monovalent binding entity includes but is not limited to proteins, poly-peptides, peptides and antibody fragments including Fab, Fab', Fv fragments, single-chain antibody molecules such as e.g. single chain Fab, scFv. The monovalent binding entity can for example be a scaffold protein engineered using state of the art technologies like phage display or immunization. The monovalent binding entity can also be a peptide.
The "monovalent binding mode" refers to a specific binding to the R/BBB where the interaction between the monovalent binding entity and the R/BBB take place through one single epitope. The monovalent binding mode prevents any dimerization/multimerization of the
R/BBB due to a single epitope interaction point. The monovalent binding mode prevents that the intracellular sorting of the R/BBB is changed.
The term "epitope" includes any polypeptide determinant capable of specific binding to an antibody. In certain embodiments, epitope determinant include chemically active surface groupings of molecules such as amino acids, sugar side chains, phosphoryl, or sulfonyl, and, in certain embodiments, may have specific three dimensional structural characteristics, and or specific charge characteristics. An epitope is a region of an antigen that is bound by an antibody.
The "transferrin receptor" ("TfR") is a transmembrane glycoprotein (with a molecular weight of about 180,000) composed of two disulphide-bonded sub-units (each of apparent molecular weight of about 90,000) involved in iron uptake in vertebrates. In one embodiment, the TfR herein is human TfR comprising the amino acid sequence as in Schneider et al. Nature 311 : 675 - 678 (1984), for example.
CD98 (also referred to as CD98 heavy chain; 42F heavy chain; SLC3A2) is a type II transmembrane glycoprotein. The human CD98hc sequence is set forth in UNIPROT Accession No. P08195. The protein comprises a 75 amino acid N-terminal intracellular cytoplasmic domain, a single transmembrane domain, and a 426 amino acid C-terminal extracellular domain (Parmacek et al., Nucleic Acids Res. 17: 1915-1931, 1989). CD98 covalently links via a disulfide bond to one of several light chains (SLC7A5, 6, 7, 8, 10, or 1 1), which are L-type amino acid transporters. This interaction is required for the cell surface expression and amino acid transport function of the light chains. CD98 also associates with integral [3 subunits, thereby regulating integrin signaling that controls cell proliferation, survival, migration, and epithelial adhesion/polarity (Cai et al., J. Cell Sci. 1 18: 889-899, 2005).
PODXL, also known as podocalyxin-like protein-1, PCLP1 or PCX, is a type I transmembrane protein of the sialomucin family of CD34. The human PODXL sequence is set forth in UNIPOROT Accession No. 000592. Preserving the basic structure of family members CD34 and endoglycan, PODXL consists of a highly conserved cytoplasmic domain with a C-terminal PDZ binding region (DTHL motif), a single-pass transmembrane domain, and an extensively O-glycosylated and sialylated extracellular domain A "neurological disorder" as used herein refers to a disease or disorder which affects the CNS and/or which has an etiology in the CNS. Exemplary CNS diseases or disorders include, but are not limited to, neuropathy, amyloidosis, cancer, an ocular disease or disorder, viral or microbial infection, inflammation, ischemia, neurodegenerative disease, seizure, behavioral disorders, and a lysosomal storage disease. For the purposes of this application, the CNS will be understood to include the eye, which is normally sequestered from the rest of the body by the blood-retina barrier. Specific examples of neurological disorders include, but are not limited to, neurodegenerative diseases (including, but not limited to, Lewy body disease, postpoliomyelitis syndrome, Shy-Draeger syndrome, olivopontocerebellar atrophy, Parkinson's disease, multiple system atrophy, stria- tonigral degeneration, tauopathies (including, but not limited to, Alzheimer disease and supranuclear palsy), prion diseases (including, but not limited to, bovine spongiform encephalopathy, scrapie, Creutzfeldt- Jakob syndrome, kuru, Gerstmann-Straussler-Scheinker disease, chronic wasting disease, and fatal familial insomnia), bulbar palsy, motor neuron disease, and nervous system heterodegenerative disorders (including, but not limited to, Canavan disease, Huntington's disease, neuronal ceroid- lipofuscinosis, Alexander's disease, Tourette's syndrome, Menkes kinky hair syndrome, Cockayne syndrome, Halervorden-Spatz syndrome, lafora disease, Rett syndrome, hepatolenticular degeneration, Lesch-Nyhan syndrome, and Unverricht-Lundborg syndrome), dementia (including, but not limited to, Pick's disease, and
spinocerebellar ataxia), cancer (e.g. of the CNS and/or brain, including brain metastases resulting from cancer elsewhere in the body).
A "neurological disorder drug" is a drug or therapeutic agent that treats one or more neurological disorder(s). Neurological disorder drugs of the invention include, but are not limited to, small molecule compounds, antibodies, peptides, proteins, natural ligands of one or more CNS target(s), modified versions of natural ligands of one or more CNS target(s), aptamers, inhibitory nucleic acids (i.e., small inhibitory RNAs (siRNA) and short hairpin RNAs (shRNA)), ribozymes, and small molecules, or active fragments of any of the foregoing. Exemplary neurological disorder drugs of the invention are described herein and include, but are not limited to: antibodies, aptamers, proteins, peptides, inhibitory nucleic acids and small molecules and active fragments of any of the foregoing that either are themselves or specifically recognize and/or act upon (i.e., inhibit, activate, or detect) a CNS antigen or target molecule such as, but not limited to, amyloid precursor protein or portions thereof, amyloid beta, beta-secretase, gamma-secretase, tau, alpha-synuclein, parkin, huntingtin, DR6, presenilin, ApoE, glioma or other CNS cancer markers, and neurotrophins. Non-limiting examples of neurological disorder drugs and the corresponding disorders they may be used to treat: Brain- derived neurotrophic factor (BDNF), Chronic brain injury (Neurogenesis), Fibroblast growth factor 2 (FGF-2), Anti-Epidermal Growth Factor Receptor Brain cancer, (EGFR)-antibody, Glial cell-line derived neural factor Parkinson's disease, (GDNF), Brain-derived neurotrophic factor (BDNF) Amyotrophic lateral sclerosis, depression, Lysosomal enzyme Lysosomal storage disorders of the brain, Ciliary neurotrophic factor (CNTF) Amyotrophic lateral sclerosis, Neuregulin-1 Schizophrenia, Anti-HER2 antibody (e.g. trastuzumab) Brain metastasis from HER2 -positive cancer.
An "imaging agent" is a compound that has one or more properties that permit its presence and/or location to be detected directly or indirectly. Examples of such imaging agents include proteins and small molecule compounds incorporating a labeled entity that permits detection.
A "CNS antigen" or "brain target" is an antigen and/or molecule expressed in the CNS, including the brain, which can be targeted with an antibody or small molecule. Examples of such antigen and/or molecule include, without limitation: beta-secretase 1 (BACE1), amyloid beta (Abeta), epidermal growth factor receptor (EGFR), human epidermal growth factor receptor 2 (HER2), Tau, apolipoprotein E4 (ApoE4), alpha-synuclein, CD20, huntingtin, prion protein (PrP), leucine rich repeat kinase 2 (LRRK2), parkin, presenilin 1, presenilin 2, gamma secretase, death receptor 6 (DR6), amyloid precursor protein (APP), p75 neurotro- phin receptor (p75NTR), and caspase 6. In one embodiment, the antigen is BACE1.
A "native sequence" protein herein refers to a protein comprising the amino acid sequence of a protein found in nature, including naturally occurring variants of the protein. The term as used herein includes the protein as isolated from a natural source thereof or as recom- binantly produced.
The term "antibody" herein is used in the broadest sense and specifically covers monoclonal antibodies, polyclonal antibodies, multispecific antibodies {e.g. bispecific antibodies) formed from at least two intact antibodies, and antibody fragments so long as they exhibit the desired biological activity.
"Antibody fragments" herein comprise a portion of an intact antibody which retains the ability to bind antigen. Examples of antibody fragments include Fab, Fab', F(ab')2, and Fv fragments; diabodies; linear antibodies; single-chain antibody molecules such as e.g. single
chain Fab, scFv and multispecific antibodies formed from antibody fragments. The “Single chain Fab” format is e.g. described in Hust M. et al. BMC Biotechnol. 2007 Mar 8;7:14.
The term "monoclonal antibody" as used herein refers to an antibody obtained from a population of substantially homogeneous antibodies, i.e., the individual antibodies comprising the population are identical and/or bind the same epitope, except for possible variants that may arise during production of the monoclonal antibody, such variants generally being present in minor amounts. In contrast to polyclonal antibody preparations that typically include different antibodies directed against different determinants (epitopes), each monoclonal antibody is directed against a single determinant on the antigen. In addition to their specificity, the monoclonal antibodies are advantageous in that they are uncontaminated by other immunoglobulins. The modifier "monoclonal" indicates the character of the antibody as being obtained from a substantially homogeneous population of antibodies, and is not to be construed as requiring production of the antibody by any particular method. For example, the monoclonal antibodies to be used in accordance with the present invention may be made by the hy- bridoma method first described by Kohler et al., Nature, 256:495 (1975), or may be made by recombinant DNA methods (see, e.g., U.S. Patent No. 4,816,567). The "monoclonal antibodies" may also be isolated from phage antibody libraries using the techniques described in Clackson et al., Nature, 352:624-628 (1991) and Marks et al., J. Mol. Biol., 222:581-597 (1991), for example. Specific examples of monoclonal antibodies herein include chimeric antibodies, humanized antibodies, and human antibodies, including antigen-binding fragments thereof. The monoclonal antibodies herein specifically include "chimeric" antibodies (immunoglobulins) in which a portion of the heavy and/or light chain is identical with or homologous to corresponding sequences in antibodies derived from a particular species or belonging to a particular antibody class or subclass, while the remainder of the chain(s) is identical with or homologous to corresponding sequences in antibodies derived from another species or belonging to another antibody class or subclass, as well as fragments of such antibodies, so long as they exhibit the desired biological activity (U.S. Patent No. 4,816,567; Morrison et al, Proc. Natl. Acad. Sci. USA, 81 :6851-6855 (1984)). Chimeric antibodies of interest herein include "primatized" antibodies comprising variable domain antigen-binding sequences derived from a non-human primate {e.g. Old World Monkey, such as baboon, rhesus or cynomolgus monkey) and human constant region sequences (US Pat No. 5,693,780).
"Humanized" forms of non-human {e.g., murine) antibodies are chimeric antibodies that contain minimal sequence derived from non-human immunoglobulin. For the most part, humanized antibodies are human immunoglobulins (recipient antibody) in which residues from a hypervariable region of the recipient are replaced by residues from a hypervariable region of a non-human species (donor antibody) such as mouse, rat, rabbit or nonhuman primate having the desired specificity, affinity, and capacity. In some instances, framework region (FR) residues of the human immunoglobulin are replaced by corresponding non-human residues. Furthermore, humanized antibodies may comprise residues that are not found in the recipient antibody or in the donor antibody. These modifications are made to further refine antibody performance. In general, the humanized antibody will comprise substantially all of at least one, and typically two, variable domains, in which all or substantially all of the hypervariable regions correspond to those of a non-human immunoglobulin and all or substantially all of the FRs are those of a human immunoglobulin sequence, except for FR substitution(s) as noted above. The humanized antibody optionally also will comprise at least a portion of an
immunoglobulin constant region, typically that of a human immunoglobulin. For further details, see Jones et al, Nature 321 :522-525 (1986); Riechmann et al, Nature 332:323-329 (1988); and Presta, Curr. Op. Struct. Biol 2:593-596 (1992).
A "human antibody" herein is one comprising an amino acid sequence structure that corresponds with the amino acid sequence structure of an antibody obtainable from a human B- cell, and includes antigen-binding fragments of human antibodies. Such antibodies can be identified or made by a variety of techniques, including, but not limited to: production by transgenic animals {e.g., mice) that are capable, upon immunization, of producing human antibodies in the absence of endogenous immunoglobulin production (see, e.g., Jakobovits et al, Proc. Natl Acad. Sci. USA, 90:2551 (1993); Jakobovits et al, Nature, 362:255-258 (1993); Bruggermann et al, Year in Immuno., 7:33 (1993); and US Patent Nos. 5,591,669, 5,589,369 and 5,545,807)); selection from phage display libraries expressing human antibodies or human antibody fragments (see, for example, McCafferty et al, Nature 348:552-553 (1990); Johnson et al, Current Opinion in Structural Biology 3:564-571 (1993); Clackson et al, Nature, 352:624-628 (1991); Marks et al, J. Mol. Biol. 222:581-597 (1991); Griffith et al, EMBO J. 12:725-734 (1993);US Patent Nos. 5,565,332 and 5,573,905); generation via in vitro activated B cells (see US Patents 5,567,610 and 5,229,275); and isolation from human antibody producing hybridomas.
A "multispecific antibody" herein is an antibody having binding specificities for at least two different epitopes. Exemplary multispecific antibodies may bind both an R/BBB and a brain antigen. Multispecific antibodies can be prepared as full-length antibodies or antibody fragments (e.g. F(ab')2 bispecific antibodies). Engineered antibodies with two, three or more (e.g. four) functional antigen binding sites are also contemplated (see, e.g., US Appln No. US 2002/0004587 Al, Miller et al.). Multispecific antibodies can be prepared as full length antibodies or antibody fragments.
Antibodies herein include "amino acid sequence variants" with altered antigen-binding or biological activity. Examples of such amino acid alterations include antibodies with enhanced affinity for antigen (e.g. "affinity matured" antibodies), and antibodies with altered Fc region, if present, e.g. with altered (increased or diminished) antibody dependent cellular cytotoxicity (ADCC) and/or complement dependent cytotoxicity (CDC) (see, for example, WO 00/42072, Presta, L. and WO 99/51642, Iduosogie et al); and/or increased or diminished serum half-life (see, for example, WO00/42072, Presta, L.).
An "affinity modified variant" has one or more substituted hypervariable region or framework residues of a parent antibody (e.g. of a parent chimeric, humanized, or human antibody) that alter (increase or reduce) affinity. In one embodiment, the resulting variant(s) selected for further development will have reduced affinity for the R/BBB according to the present invention. A convenient way for generating such substitutional variants uses phage display. Briefly, several hypervariable region sites (e.g. 6-7 sites) are mutated to generate all possible amino substitutions at each site. The antibody variants thus generated are displayed in a monovalent fashion from filamentous phage particles as fusions to the gene III product of Ml 3 packaged within each particle. The phage-displayed variants are then screened for their biological activity (e.g. binding affinity). In order to identify candidate hypervariable region sites for modification, alanine scanning mutagenesis can be performed to identify hypervariable region residues contributing significantly to antigen binding. Alternatively, or additionally, it may be beneficial to analyze a crystal structure of the antigen-antibody complex to identify
contact points between the antibody and its target. Such contact residues and neighboring residues are candidates for substitution according to the techniques elaborated herein. Once such variants are generated, the panel of variants is subjected to screening and antibodies with altered affinity may be selected for further development.
The antibody herein may be a "glycosylation variant" such that any carbohydrate attached to the Fc region, if present, is altered. For example, antibodies with a mature carbohydrate structure that lacks fucose attached to an Fc region of the antibody are described in US Pat Appl No US 2003/0157108 (Presta, L ). See also US 2004/0093621 (KyowaHakko Kogyo Co., Ltd). Antibodies with a bisecting N-acetylglucosamine (GlcNAc) in the carbohydrate attached to an Fc region of the antibody are referenced in WO 2003/011878, Jean-Mai- ret et al. and US Patent No. 6,602,684, Umana et al. Antibodies with at least one galactose residue in the oligosaccharide attached to an Fc region of the antibody are reported in WO 1997/30087, Patel et al. See, also, WO 1998/58964 (Raju, S.) and WO 1999/22764 (Raju, S.) concerning antibodies with altered carbohydrate attached to the Fc region thereof. See also US 2005/0123546 (Umana et al.) describing antibodies with modified glycosylation. The term "hypervariable region" when used herein refers to the amino acid residues of an antibody that are responsible for antigen binding. The hypervariable region comprises amino acid residues from a "complementarity determining region" or "CDR" (e.g. residues 24- 34 (LI), 50-56 (L2) and 89-97 (L3) in the light chain variable domain and 31-35 (HI), 50-65 (H2) and 95-102 (H3) in the heavy chain variable domain; Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, MD. (1991)) and/or those residues from a "hypervariable loop" (e.g. residues 26-32 (LI), 50-52 (L2) and 91-96 (L3) in the light chain variable domain and 26-32 (HI), 53-55 (H2) and 96-101 (H3) in the heavy chain variable domain; Chothia and Lesk J. Mol. Biol. 196:901- 917 (1987)). "Framework" or "FR" residues are those variable domain residues other than the hypervariable region residues as herein defined.
A "full length antibody" is one which comprises an antigen-binding variable region as well as a light chain constant domain (CL) and heavy chain constant domains, CHI, CH2 and CH3. The constant domains may be native sequence constant domains (e.g. human native sequence constant domains) or amino acid sequence variants thereof.
A "naked antibody" is an antibody (as herein defined) that is not conjugated to a heterologous molecule, such as a cytotoxic entity, polymer, or radiolabel.
Antibody "effector functions" refer to those biological activities attributable to the Fc region (a native sequence Fc region or amino acid sequence variant Fc region) of an antibody. Examples of antibody effector functions include Clq binding, complement dependent cytotoxicity (CDC), Fc receptor binding, antibody-dependent cell-mediated cytotoxicity (ADCC), etc. In one embodiment, the antibody herein essentially lacks effector function.
The term “antibody-dependent cellular cytotoxicity (ADCC)” refers to lysis of human target cells by an antibody in the presence of effector cells. The term “complement-dependent cytotoxicity (CDC)” denotes a process initiated by binding of complement factor Clq to the Fc part of most IgG antibody subclasses. Binding of Clq to an antibody is caused by defined protein-protein interactions at the so called binding site. Such Fc part binding sites are known in the state of the art. Such Fc part binding sites are, e.g., characterized by the amino acids L234, L235, D270, N297, E318, K320, K322, P331, and P329 (numbering according to EU
index of Kabat). Antibodies of subclass IgGl, IgG2, and IgG3 usually show complement activation including Clq and C3 binding, whereas IgG4 does not activate the complement system and does not bind Clq and/or C3.
Depending on the amino acid sequence of the constant domain of their heavy chains, full length antibodies can be assigned to different "classes". There are five major classes of full length antibodies: IgA, IgD, IgE, IgG, and IgM, and several of these may be further divided into "subclasses" (isotypes), e.g., IgGl, IgG2, IgG3, IgG4, IgA, and IgA2. The heavychain constant domains that correspond to the different classes of antibodies are called alpha, delta, epsilon, gamma, and mu, respectively. The subunit structures and three-dimensional configurations of different classes of immunoglobulins are well known. The term "recombinant antibody", as used herein, refers to an antibody (e.g. a chimeric, humanized, or human antibody or antigen-binding fragment thereof) that is expressed by a recombinant host cell comprising nucleic acid encoding the antibody. Examples of "host cells" for producing recombinant antibodies include: (1) mammalian cells, for example, Chinese Hamster Ovary (CHO), COS, myeloma cells (including YO and NSO cells), baby hamster kidney (BHK), Hela and Vero cells; (2) insect cells, for example, sf9, sf21 and Tn5; (3) plant cells, for example plants belonging to the genus Nicotiana (e.g. Nicotianatabacum); (4) yeast cells, for example, those belonging to the genus Saccharomyces (e.g. Saccharomyces cerevisiae) or the genus Aspergillus (e.g. Aspergillus niger); (5) bacterial cells, for example Escherichia, coli cells or Bacillus subtilis cells, etc.
As used herein, "specifically binding" or "binds specifically to" refers to an antibody selectively or preferentially binding to an antigen. The binding affinity is generally determined using a standard assay, such as Scatchard analysis, or surface plasmon resonance technique (e.g. using BIACORE®).
An "antibody that binds to the same epitope" as a reference antibody refers to an antibody that blocks binding of the reference antibody to its antigen in a competition assay by 50% or more, and conversely, the reference antibody blocks binding of the antibody to its antigen in a competition assay by 50% or more.
An "imaging agent" is a compound that has one or more properties that permit its presence and/or location to be detected directly or indirectly. Examples of such imaging agents include proteins and small molecule compounds incorporating a labeled entity that permits detection.
A “label” is a marker coupled with the antibody herein and used for detection or imaging. Examples of such labels include: radiolabel, a fluorophore, a chromophore, or an affinity tag. In one embodiment, the label is a radiolabel used for medical imaging, for example tc99m or 1123, or a spin label for nuclear magnetic resonance (NMR) imaging (also known as magnetic resonance imaging, mri), such as iodine- 123 again, iodine-131, indium-i ll, fluo- rine- 19, carbon-13, nitrogen-15, oxygen-17, gadolinium, manganese, iron, etc.
The term "cytotoxic agent" as used herein refers to a substance that inhibits or prevents a cellular function and/or causes cell death or destruction. Cytotoxic agents include, but are not limited to, radioactive isotopes (e.g., At211, 1131, 1125, Y90, Rel86, Rel88, Sml53, Bi212, P32, Pb212 and radioactive isotopes of Lu); chemotherapeutic agents or drugs (e.g., methotrexate, adriamicin, vinca alkaloids (vincristine, vinblastine, etoposide), doxorubicin, melphalan, mitomycin C, chlorambucil, daunorubicin or other intercalating agents); growth inhibitory agents; enzymes and fragments thereof such as nucleolytic enzymes; antibiotics;
toxins such as small molecule toxins or enzymatically active toxins of bacterial, fungal, plant or animal origin, including fragments and/or variants thereof.
An "effective amount" of an agent, e.g., a pharmaceutical formulation, refers to an amount effective, at dosages and for periods of time necessary, to achieve the desired therapeutic or prophylactic result.
The term "Fc region" herein is used to define a C-terminal region of an immunoglobulin heavy chain that contains at least a portion of the constant region. The Fc region comprises the CH2 and CH3 domains of an immunoglobulin. The term includes native sequence Fc regions and variant Fc regions. In one embodiment, a human IgG heavy chain Fc region extends from Cys226, or from Pro230, to the carboxyl-terminus of the heavy chain. However, the C- terminal lysine (Lys447) of the Fc region may or may not be present. Unless otherwise specified herein, numbering of amino acid residues in the Fc region or constant region is according to the EU numbering system, also called the EU index, as described in Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, MD, 1991.
"Framework" or "FR" refers to variable domain residues other than hypervariable region (HVR) residues. The FR of a variable domain generally consists of four FR domains: FR1, FR2, FR3, and FR4. Accordingly, the HVR and FR sequences generally appear in the following sequence in VH (or VL): FR1-H1(L1)-FR2-H2(L2)-FR3-H3(L3)-FR4.
The term “CH2-CH3 Ig entity” as used herein refers to a protein entity derived from immunoglobulin CH2 or CH3 domains. The “CH2-CH3 Ig entity” comprises two “CH2- CH3” polypeptides forming a dimer. The immunoglobulin can be IgG, IgA, IgD, IgE or IgM. In one embodiment, the CH2-CH3 Ig entity derived from an IgG immunoglobulin and is referred to herein as “CH2-CH3 IgG entity”. The term includes native sequence of CH2-CH3 domains and variant CH2-CH3 domains. In one embodiment, the “CH2-CH3 Ig entity” derives from human heavy chain CH2-CH3 IgG domain which extends from Cys226, or from Pro230, to the carboxyl-terminus of the heavy chain. However, the C-terminal lysine (Lys447) of the Fc region may or may not be present. Unless otherwise specified herein, numbering of amino acid residues in the CH2-CH3 domain region or constant region is according to the EU numbering system, also called the EU index, as described in Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, MD, 1991.
An "conjugate" is fusion protein of the present invention conjugated to one or more heterologous molecule(s), including but not limited to a label, neurological disorder drug or cytotoxic agent.
A " linker" as used herein refers to a chemical linker or a single chain peptide linker that covalently connects the different entities of the multispecific antibody and/or the fusion protein and/or the conjugate of the present invention. The linker connects for example the compound to the monovalent binding entity. For example, if the monovalent binding entity comprises a CH2-CH3 Ig entity and a sFab directed to the blood brain barrier receptor, then the linker connects the scFab to the C-terminal end of the CH3-CH2 Ig entity. The linker connecting the brain effector entity to the monovalent binding entity (first linker) and the linker connecting the scFab to the C-terminal end of the CH2-CH3 Ig domain (second linker) can be the same or different.
Single chain peptide linkers, comprised of from one to twenty amino acids joined by peptide bonds, can be used. In certain embodiments, the amino acids are selected from the
twenty naturally-occurring amino acids. In certain other embodiments, one or more of the amino acids are selected from glycine, alanine, proline, asparagine, glutamine and lysine. In other embodiments, the linker is a chemical linker. In certain embodiments, said linker is a single chain peptide with an amino acid sequence with a length of at least 25 amino acids, preferably with a length of 32 to 50 amino acids. In one embodiment said linker is (GxS)n with G = glycine, S = serine, (x =3, n= 8, 9 or 10 and m= 0, 1 , 2 or 3) or (x = 4 and n= 6, 7 or 8 and m= 0, 1, 2 or 3), preferably with x = 4, n= 6 or 7 and m= 0, 1, 2 or 3, more preferably with x = 4, n= 7 and m= 2. In one embodiment said linker is (648)4 (Seq. Id. No. 17). In one embodiment said linker is (GiSjeGz (Seq. Id. No. 13).
Conjugation may be performed using a variety of chemical linkers. For example, the monovalent binding entity or the fusion protein and the compound may be conjugated using a variety of bifunctional protein coupling agents such as N-succinimidyl-3-(2-pyridyldithio) propionate (SPDP), succinimidyl-4-(N-maleimidomethyl) cyclohexane-l-carboxylate (SMCC), iminothiolane (IT), bifunctional derivatives of imidoesters (such as dimethyl adip- imidate HC1), active esters (such as disuccinimidyl sub erate), aldehydes (such as glutaraldehyde), bis-azido compounds (such as bis (p- azidobenzoyl) hexanediamine), bis-diazonium derivatives (such as bis-(p-diazoniumbenzoyl)- ethylenediamine), diisocyanates (such as toluene 2,6-diisocyanate), and bis-active fluorine compounds (such as l,5-difhioro-2,4-dinitroben- zene). The linker may be a "cleavable linker" facilitating release of the effector entity upon delivery to the brain. For example, an acid- labile linker, peptidase-sensitive linker, photola- bile linker, dimethyl linker or disulfide- containing linker (Chari et al, Cancer Res. 52: 127- 131 (1992); U.S. Patent No. 5,208,020) may be used.
Covalent conjugation can either be direct or via a linker. In certain embodiments, direct conjugation is by construction of a protein fusion (i.e., by genetic fusion of the two genes encoding the monovalent binding entity towards the R/BBB and effector entity and expressed as a single protein). In certain embodiments, direct conjugation is by formation of a covalent bond between a reactive group on one of the two portions of the monovalent binding entity against the R/BBB and a corresponding group or acceptor on the compound. In certain embodiments, direct conjugation is by modification (i.e., genetic modification) of one of the two molecules to be conjugated to include a reactive group (as non-limiting examples, a sulfhydryl group or a carboxyl group) that forms a covalent attachment to the other molecule to be conjugated under appropriate conditions. As one non-limiting example, a molecule (i.e., an amino acid) with a desired reactive group (i.e., a cysteine residue) may be introduced into, e.g., the monovalent binding entity towards the R/BBB antibody and a disulfide bond formed with the neurological drug. Methods for covalent conjugation of nucleic acids to proteins are also known in the art (i.e., photocrosslinking, see, e.g., Zatsepin et al. Russ. Chem. Rev. 74: 77-95 (2005)) Conjugation may also be performed using a variety of linkers. For example, a monovalent binding entity and a effector entity may be conjugated using a variety of bifunctional protein coupling agents such as N- succinimidyl-3 -(2 -pyridyldithio) propionate (SPDP), succinimidyl-4-(N-maleimidomethyl) cyclohexane-l-carboxylate (SMCC), iminothiolane (IT), bifunctional derivatives of imidoesters (such as dimethyl adipimidate HC1), active esters (such as disuccinimidyl suberate), aldehydes (such as glutaraldehyde), bis-azido compounds (such as bis (p- azidobenzoyl) hexanediamine), bis-diazonium derivatives (such as bis-(p-dia- zoniumbenzoyl)- ethylenediamine), diisocyanates (such as toluene 2,6-diisocyanate), and bis- active fluorine compounds (such as l,5-difluoro-2,4-dinitrobenzene). Peptide linkers, com-
prised of from one to twenty amino acids joined by peptide bonds, may also be used. In certain such embodiments, the amino acids are selected from the twenty naturally-occurring amino acids. In certain other such embodiments, one or more of the amino acids are selected from glycine, alanine, proline, asparagine, glutamine and lysine. The linker may be a "cleavable linker" facilitating release of the effector entity upon delivery to the brain. For example, an acid- labile linker, peptidase-sensitive linker, photolabile linker, dimethyl linker or disulfide- containing linker (Chari et al, Cancer Res. 52: 127-131 (1992); U.S. Patent No. 5,208,020) may be used.
A "label" is a marker coupled with the fusion protein herein and used for detection or imaging. Examples of such labels include: radiolabel, a fluorophore, a chromophore, or an affinity tag. In one embodiment, the label is a radiolabel used for medical imaging, for example tc99m or 1123, or a spin label for nuclear magnetic resonance (NMR) imaging (also known as magnetic resonance imaging, mri), such as iodine- 123 again, iodine-131, indium- 111, fluorine- 19, carbon- 13, nitrogen-15, oxygen- 17, gadolinium, manganese, iron, etc. An "individual" or "subject" is a mammal. Mammals include, but are not limited to, domesticated animals (e.g., cows, sheep, cats, dogs, and horses), primates (e.g., humans and non-human primates such as monkeys), rabbits, and rodents (e.g., mice and rats). In certain embodiments, the individual or subject is a human.
An "isolated" antibody is one which has been separated from a component of its natural environment. In some embodiments, an antibody is purified to greater than 95% or 99% purity as determined by, for example, electrophoretic (e.g., SDS-PAGE, isoelectric focusing (IEF), capillary electrophoresis) or chromatographic (e.g., ion exchange or reverse phase HPLC). For review of methods for assessment of antibody purity, see, e.g., Flatman et al, J. Chromatogr. B 848:79-87 (2007).
The term "pharmaceutical formulation" refers to a preparation which is in such form as to permit the biological activity of an active ingredient contained therein to be effective, and which contains no additional components which are unacceptably toxic to a subject to which the formulation would be administered.
A "pharmaceutically acceptable carrier" refers to an ingredient in a pharmaceutical formulation, other than an active ingredient, which is nontoxic to a subject., A pharmaceutically acceptable carrier includes, but is not limited to, a buffer, excipient, stabilizer, or preservative.
As used herein, "treatment" (and grammatical variations thereof such as "treat" or "treating") refers to clinical intervention in an attempt to alter the natural course of the individual being treated, and can be performed either for prophylaxis or during the course of clinical pathology. Desirable effects of treatment include, but are not limited to, preventing occurrence or recurrence of disease, alleviation of symptoms, diminishment of any direct or indirect pathological consequences of the disease, preventing metastasis, decreasing the rate of disease progression, amelioration or palliation of the disease state, and remission or improved prognosis. In some embodiments, antibodies of the invention are used to delay development of a disease or to slow the progression of a disease.
For a neuropathy disorder, a neurological drug may be selected that is an analgesic including, but not limited to, anarcotic/opioid analgesic (i.e., morphine, fentanyl, hydrocodone, meperidine, methadone, oxymorphone, pentazocine, propoxyphene, tramadol, codeine and oxycodone), a nonsteroidal anti-inflammatory drug (NSAID) (i.e., ibuprofen, naproxen, diclofenac, diflunisal, etodolac, fenoprofen, flurbiprofen, indomethacin, ketorolac, mefenamic
acid, meloxicam, nabumetone, oxaprozin, piroxicam, sulindac, and tolmetin), a corticosteroid (i.e., cortisone, prednisone, prednisolone, dexamethasone, methylprednisolone and triamcinolone), an anti-migraine agent (i.e., sumatriptin, almo triptan, frovatriptan, sumatriptan, rizatriptan, eletriptan, zolmitriptan, dihydro ergotamine, eletriptan and ergotamine), acetaminophen, a salicylate (i.e., aspirin, choline salicylate, magnesium salicylate, diflunisal, and salsalate), an anti-convulsant (i.e., carbamazepine, clonazepam, gabapentin, lamotrigine, pregabalin, tiaga- bine, and topiramate), an anaesthetic (i.e., isoflurane, trichloroethylene, halothane, sevoflu- rane, benzocaine, chloroprocaine, cocaine, cyclomethycaine, dimethocaine, propoxycaine, procaine, novocaine, proparacaine, tetracaine, articaine, bupivacaine, carticaine, cinchocaine, etidocaine, levobupivacaine, lidocaine, mepivacaine, piperocaine, prilocaine, ropivacaine, trimecaine, saxitoxin and tetrodo toxin), and a cox-2-inhibitor (i.e., celecoxib, rofecoxib, and valdecoxib). For a neuropathy disorder with vertigo involvement, a neurological drug may be selected that is an anti- vertigo agent including, but not limited to, meclizine, diphenhydramine, promethazine and diazepam. For a neuropathy disorder with nausea involvement, a neurological drug may be selected that is an anti-nausea agent including, but not limited to, promethazine, chlorpromazine, prochlorperazine, trimethobenzamide, and metoclopramide. For a neurodegenerative disease, a neurological drug may be selected that is a growth hormone or neurotrophic factor; examples include but are not limited to brain-derived neurotrophic factor (BDNF), nerve growth factor (NGF), neurotrophin-4/5, fibroblast growth factor (FGF)-2 and other FGFs, neurotrophin (NT)-3, erythropoietin (EPO), hepatocyte growth factor (HGF), epidermal growth factor (EGF), transforming growth factor (TGF)-alpha, TGF- beta, vascular endothelial growth factor (VEGF), interleukin-1 receptor antagonist (IL-lra), ciliary neurotrophic factor (CNTF), glial-derived neurotrophic factor (GDNF), neurturin, platelet-derived growth factor (PDGF), heregulin, neuregulin, artemin, persephin, interleukins, glial cell line derived neurotrophic factor (GFR), granulocyte-colony stimulating factor (CSF), granulocyte-macro phage-CSF, netrins, cardiotrophin-1, hedgehogs, leukemia inhibitory factor (LIF), midkine, pleiotrophin, bone morphogenetic proteins (BMPs), netrins, saposins, semaphorins, and stem cell factor (SCF). For cancer, a neurological drug may be selected that is a chemotherapeutic agent. Examples of chemotherapeutic agents include alkylating agents such as thi- otepa and CYTOXAN® cyclosphosphamide; alkyl sulfonates such as busulfan, improsulfan and piposulfan; aziridines such as benzodopa, carboquone, meturedopa, and uredopa; ethyl- enimines and methylamelamines including altretamine, triethylenemelamine, trie- tylenephosphoramide, triethiylenethiophosphor-amide and trimethylolomelamine; acetogenins (especially bullatacin and bullatacinone); delta-9-tetrahydrocannabinol (dronabinol, MARINOL®); beta-lapachone; lapachol; colchicines; betulinic acid; a camptothecin (including the synthetic analogue topotecan (HYCAMTIN®), CPT-11 (irinotecan, CAMP- TOSAR®), acetylcamptothecin, scopolectin, and 9-aminocamptothecin); bryostatin; callysta- tin; CC-1065 (including its adozelesin, carzelesin and bizelesin synthetic analogues); podo- phyllotoxin; podophyllinic acid; teniposide; cryptophycins (particularly cryptophycin 1 and cryptophycin 8); dolastatin; duocarmycin (including the synthetic analogues, KW-2189 and CB1-TM1); eleutherobin; pancratistatin; a sarcodictyin; spongistatin; nitrogen mustards such as chlorambucil, chlomaphazine, cholophosphamide, estramustine, ifosfamide, mechlor- ethamme, mechlorethamme oxide hydrochloride, melphalan, novembichin, phenesterine, prednimustine, trofosfamide, uracil mustard; nitrosureas such as carmustine, chlorozotocin, fotemustine, lomustine, nimustine, and ranimnustine; antibiotics such as the enediyne antibiotics (e. g., calicheamicin, especially calicheamicin gammall and calicheamicin omegall (see,
e.g., Agnew, Chem Inti. Ed. Engl, 33: 183-186 (1994)); dynemicin, including dynemicin A; an esperamicin; as well as neocarzinostatin chromophore and related chromoprotein enediyne antiobiotic chromophores), aclacinomysins, actinomycin, authramycin, azaserine, bleomycins, cactinomycin, carabicin, carminomycin, carzinophilin, chromomycinis, dactinomycin, dauno- rubicin, detorubicin, 6-diazo-5-oxo-L-norleucine, ADRIAMYCIN® doxorubicin (including morpholino-doxorubicin, cyanomorpholino- doxorubicin, 2-pyrrolino-doxorubicin and deoxydoxorubicin), epirubicin, esorubicin, idarubicin, marcellomycin, mitomycins such as mitomycin C, mycophenolic acid, nogalamycin, olivomycins, peplomycin, potfiromycin, puromy- cin, quelamycin, rodorubicin, streptonigrin, streptozocin, tubercidin, ubenimex, zinostatin, zo- rubicin; anti-metabolites such as methotrexate and 5 -fluorouracil (5-FU); folic acid analogues such as denopterin, methotrexate, pteropterin, trimetrexate; purine analogs such as fludarabine, 6 -mercaptopurine, thiamiprine, thioguanine; pyrimidine analogs such as ancitabine, azacitidine, 6-azauridine, carmofur, cytarabine, dideoxyuridine, doxifluridine, enocitabine, floxuridine; androgens such as calusterone, dromostanolone propionate, epitiostanol, mepiti- ostane, testolactone; anti- adrenals such as aminoglutethimide, mitotane, trilostane; folic acid replenisher such as frolinic acid; aceglatone; aldophosphamide glycoside; aminolevulinic acid; eniluracil; amsacrine; bestrabucil; bisantrene; edatraxate; defofamine; demecolcine; dia- ziquone; elfomithine; elliptinium acetate; an epothilone; etoglucid; gallium nitrate; hydroxyurea; lentinan; lonidainine; maytansinoids such as maytansine and ansamitocins; mito- guazone; mitoxantrone; mopidanmol; nitraerine; pentostatin; phenamet; pirarubicin; losoxan- trone; 2-ethylhydrazide; procarbazine; PSK® polysaccharide complex (JHS Natural Products, Eugene, OR); razoxane; rhizoxin; sizofiran; spirogermanium; tenuazonic acid; triaziquone; 2, 2', 2"- trichlorotriethylamine; trichothecenes (especially T-2 toxin, verracurin A, roridin A and anguidine); urethan; vindesine (ELDISINE®, FILDESIN®); dacarbazine; mannomustine; mitobronitol; mitolactol; pipobroman; gacytosine; arabinoside ("Ara-C"); thiotepa; taxoids, e.g., TAXOL® paclitaxel (Bristol-Myers Squibb Oncology, Princeton, N.J.), ABRAX- ANETM Cremophor-free, albumin-engineered nanoparticle formulation of paclitaxel (American Pharmaceutical Partners, Schaumberg, Illinois), and TAXOTERE® doxetaxel (Rhone- Poulenc Rorer, Antony, France); chloranbucil; gemcitabine (GEMZAR®); 6-thioguanine; mercaptopurine; methotrexate; platinum analogs such as cisplatin and carboplatin; vinblastine (VELBAN®); platinum; etoposide (VP- 16); ifosfamide; mitoxantrone; vincristine (ONCOVIN®); oxaliplatin; leucovovin; vinorelbine (NAVELBINE®); novantrone; edatrexate; daunomycin; aminopterin; ibandronate; topoisomerase inhibitor RFS 2000; difluorometlhy- lomithine (DMFO); retinoids such as retinoic acid; capecitabine (XELODA®); pharmaceutically acceptable salts, acids or derivatives of any of the above; as well as combinations of two or more of the above such as CHOP, an abbreviation for a combined therapy of cyclophosphamide, doxorubicin, vincristine, and prednisolone, and FOLFOX, an abbreviation for a treatment regimen with oxaliplatin (ELOXATINTM) combined with 5-FU and leucovovin.
Also included in this definition of chemotherapeutic agents are anti-hormonal agents that act to regulate, reduce, block, or inhibit the effects of hormones that can promote the growth of cancer, and are often in the form of systemic, or whole-body treatment. They may be hormones themselves. Examples include anti-estrogens and selective estrogen receptor modulators (SERMs), including, for example, tamoxifen (including NOLVADEX® tamoxifen), EVISTA® raloxifene, droloxifene, 4-hydroxytamoxifen, trioxifene, keoxifene, LY 117018, onapristone, and FARESTON® toremifene; anti-progesterones; estrogen receptor
down-regulators (ERDs); agents that function to suppress or shut down the ovaries, for example, leutinizing hormone-releasing hormone (LHRH) agonists such as LUPRON® and ELI- GARD® leuprolide acetate, goserelin acetate, buserelin acetate and tripterelin; other anti- androgens such as flutamide, nilutamide and bicalutamide; and aromatase inhibitors that inhibit the enzyme aromatase, which regulates estrogen production in the adrenal glands, such as, for example, 4(5)-imidazoles, aminoglutethimide, MEGASE® megestrol acetate, AROMASIN® exemestane, formestanie, fadrozole, RIVISOR® vorozole, FEMARA® letrozole, and ARIM- IDEX® anastrozole. In addition, such definition of chemotherapeutic agents includes bisphosphonates such as clodronate (for example, BONEFOS® or OSTAC®), DIDROCAL® etidronate, NE-58095, ZOMETA® zoledronic acid/zoledronate, FOSAMAX® alendronate, ARE- DIA® pamidronate, SKELID® tiludronate, or ACTONEL® risedronate; as well as troxacita- bine (a 1,3 -dioxolane nucleoside cytosine analog); antisense oligonucleotides, particularly those that inhibit expression of genes in signaling pathways implicated in aberrant cell proliferation, such as, for example, PKC-alpha, Raf, H-Ras, and epidermal growth factor receptor (EGF-R); vaccines such as THERATOPE® vaccine and gene therapy vaccines, for example, ALLOVECTIN® vaccine, LEUVECTIN® vaccine, and VAXID® vaccine; LURTO- TECAN® topoisomerase 1 inhibitor; ABARELLX® rmRH; lapatinib ditosylate (an ErbB-2 and EGFR dual tyrosine kinase small-molecule inhibitor also known as GW572016); and pharmaceutically acceptable salts, acids or derivatives of any of the above.
Another group of compounds that may be selected as neurological drugs for cancer treatment or prevention are anti-cancer immunoglobulins (including, but not limited to, trastuzumab, bevacizumab, alemtuxumab, cetuximab, gemtuzumab ozogamicin, ibritumomab tiuxetan, panitumumab and rituximab). In some instances, antibodies in conjunction with a toxic label may be used to target and kill desired cells (i.e., cancer cells), including, but not limited to, tositumomab with a radiolabel.
For an ocular disease or disorder, a neurological drug may be selected that is an anti- angiogenic ophthalmic agent (i.e., bevacizumab, ranibizumab and pegaptanib), an ophthalmic glaucoma agent (i.e., carbachol, epinephrine, demecarium bromide, apraclonidine, brimoni- dine, brinzolamide, levobunolol, timolol, betaxolol, dorzolamide, bimatoprost, carteolol, metipranolol, dipivefrin, travoprost and latano prost), a carbonic anhydrase inhibitor (i.e., methazolamide and acetazolamide), an ophthalmic antihistamine (i.e., naphazoline, phenylephrine and tetrahydrozoline), an ocular lubricant, an ophthalmic steroid (i.e., fluoro- metholone, prednisolone, loteprednol, dexamethasone, difluprednate, rimexolone, fluocino- lone, medrysone and triamcinolone), an ophthalmic anesthetic (i.e., lidocaine, proparacaine and tetracaine), an ophthalmic anti -infective (i.e., levofloxacin, gatifloxacin, ciprofloxacin, moxif oxacin, chloramphenicol, bacitracin/polymyxin b, sulfacetamide, tobramycin, azithromycin, besifloxacin, norfloxacin, sulfisoxazole, gentamicin, idoxuridine, erythromycin, natamycin, gramicidin, neomycin, ofloxacin, trif uridine, ganciclovir, vidarabine), an ophthalmic anti-inflammatory agent (i.e., nepafenac, ketorolac, flurbiprofen, suprofen, cyclosporine, triamcinolone, diclofenac and bromfenac), and an ophthalmic antihistamine or decongestant (i.e., ketotifen, olopatadine, epinastine, naphazoline, cromolyn, tetrahydrozoline, pemirolast, bepotastine, naphazoline, phenylephrine, nedocromil, lodox amide, phenylephrine, emedas- tine and azelastine).For a seizure disorder, a neurological drug may be selected that is an anticonvulsant or antiepileptic including, but not limited to, barbiturate anticonvulsants (i.e., primidone, metharbital, mephobarbital, allobarbital, amobarbital, aprobarbital, alphenal, barbital, brallobarbital and phenobarbital), benzodiazepine anticonvulsants (i.e., diazepam,
clonazepam, and lorazepam), carbamate anticonvulsants (i.e. felbamate), carbonic anhydrase inhibitor anticonvulsants (i.e., acetazolamide, topiramate and zonisamide), dibenzazepine anticonvulsants (i.e., rufinamide, carbamazepine, and oxcarbazepine), fatty acid derivative anticonvulsants (i.e., divalproex and valproic acid), gamma-aminobutyric acid analogs (i.e., pregabalin, gabapentin and vigabatrin), gamma-aminobutyric acid reuptake inhibitors (i.e., ti- agabine), gamma-aminobutyric acid transaminase inhibitors (i.e., vigabatrin), hydantoin anticonvulsants (i.e. phenytoin, ethotoin, fosphenytoin and mephenytoin), miscellaneous anticonvulsants (i.e., lacosamide and magnesium sulfate), progestins (i.e., progesterone), oxazoli- dinedione anticonvulsants (i.e., paramethadione and trimethadione), pyrrolidine anticonvulsants (i.e., levetiracetam), succinimide anticonvulsants (i.e., ethosuximide and methsuximide), triazine anticonvulsants (i.e., lamotrigine), and urea anticonvulsants (i.e., phenacemide and pheneturide).
For a lysosomal storage disease, a neurological drug may be selected that is itself or otherwise mimics the activity of the enzyme that is impaired in the disease. Exemplary recombinant enzymes for the treatment of lysosomal storage disorders include, but are not limited to those set forth in e.g., U.S. Patent Application publication no. 2005/0142141 (i.e., alpha-L- iduronidase, iduronate-2-sulphatase, N-sulfatase, alpha-N-acetylglucosaminidase, N- acetyl- galactosamine-6-sulfatase, beta-galactosidase, aryl sulphatase B, beta-glucuronidase, acid al- pha-glucosidase, gluco cerebrosidase, alpha-galactosidase A, hexosaminidase A, acid sphingomyelinase, beta-galactocerebrosidase, beta-galactosidase, arylsulfatase A, acid ceramidase, aspartoacylase, palmitoyl-protein thioesterase 1 and trip eptidyl amino peptidase 1).
For amyloidosis, a neurological drug may be selected that includes, but is not limited to, an antibody or other binding molecule (including, but not limited to a small molecule, a peptide, an aptamer, or other protein binder) that specifically binds to a target selected from: beta secretase, tau, presenilin, amyloid precursor protein or portions thereof, amyloid beta peptide or oligomers or fibrils thereof, death receptor 6 (DR6), receptor for advanced glycation endproducts (RAGE), parkin, and huntingtin; a cholinesterase inhibitor (i.e., galantamine, donepezil, rivastigmine and tacrine); an NMD A receptor antagonist (i.e., memantine), a monoamine depletor (i.e., tetrab enazine); an ergoloid mesylate; an anticholinergic antiparkinsonism agent (i.e., procyclidine, diphenhydramine, trihexylphenidyl, benztropine, biperiden and trihexyphenidyl); a dopaminergic antiparkinsonism agent (i.e., entacapone, selegiline, pramipexole, bromocriptine, rotigotine, selegiline, ropinirole, rasagiline, apomorphine, car- bidopa, levodopa, pergolide, tolcapone and amantadine); a tetrabenazine; an anti-inflammatory (including, but not limited to, a nonsteroidal anti-inflammatory drug (i.e., indomethicin and other compounds listed above); a hormone (i.e., estrogen, progesterone and leuprolide); a vitamin (i.e., folate and nicotinamide); a dimebolin; a homotaurine (i.e., 3- aminopropanesulfonic acid; 3 APS); a serotonin receptor activity modulator (i.e., xaliproden); an, an interferon, and a glucocorticoid.
For a viral or microbial disease, a neurological drug may be selected that includes, but is not limited to, an antiviral compound (including, but not limited to, an adamantane antiviral (i.e., rimantadine and amantadine), an antiviral interferon (i.e., peginterferon alfa-2b), a chem- okine receptor antagonist (i.e., maraviroc), an integrase strand transfer inhibitor (i.e., ralte- gravir), a neuraminidase inhibitor (i.e., oseltamivir and zanamivir), a non-nucleoside reverse transcriptase inhibitor (i.e., efavirenz, etravirine, delavirdine and nevirapine), a nucleoside reverse transcriptase inhibitors (tenofovir, abacavir, lamivudine, zidovudine, stavudine,
entecavir, emtricitabine, adefovir, zalcitabine, telbivudine and didanosine), a protease inhibitor (i.e., darunavir, atazanavir, fosamprenavir, tipranavir, ritonavir, nelfmavir, amprenavir, indinavir and saquinavir), a purine nucleoside (i.e., valacyclovir, famciclovir, acyclovir, ribavirin, ganciclovir, valganciclovir and cidofovir), and a miscellaneous antiviral (i.e., enfuvir- tide, foscamet, palivizumab and fomivirsen)), an antibiotic (including, but not limited to, an aminopenicillin (i.e., amoxicillin, ampicillin, oxacillin, nafcillin, cioxacillin, dicl oxacillin, flu- coxacillin, temocillin, azlocillin, carbenicillin, ticarcillin, mezlocillin, piperacillin and bacampicillin), a cephalosporin (i.e., cefazolin, cephalexin, cephalothin, cefamandole, ceftriaxone, cefotaxime, cefpodoxime, ceftazidime, cefadroxil, cephradine, loracarbef, cefotetan, cefuroxime, cefprozil, cefaclor, and cefoxitin), a carbapenem/penem (i.e., imipenem, mero- penem, ertapenem, faropenem and doripenem), amonobactam (i.e., aztreonam, tigemonam, norcardicin A and tabtoxinine-beta-lactam, a beta-lactamase inhibitor (i.e., clavulanic acid, tazobactam and sulbactam) in conjunction with another beta-lactam antibiotic, an aminoglycoside (i.e., amikacin, gentamicin, kanamycin, neomycin, netilmicin, streptomycin, tobramycin, and paromomycin), an ansamycin (i.e., geldanamycin and herbimycin), a carbacephem (i.e., loracarbel), a glycopeptides (i.e., teicoplanin and vancomycin), a macrolide (i.e., azithromycin, clarithromycin, dirithromycin, erythromycin, roxithromycin, troleandomycin, telithro- mycin and spectinomycin), a monobactam (i.e., aztreonam), a quinolone (i.e., ciprofloxacin, enoxacin, gatifloxacin, levofloxacin, lomefloxacin, moxifloxacin, norfloxacin, ofloxacin, trovafloxacin, grepafloxacin, sparfloxacin and temafloxacin), a sulfonamide (i.e., mafenide, sulfonamidochrysoidine, sulfacetamide, sulfadiazine, sulfamethizole, sulfanilamide, sulfasalazine, sulfisoxazole, trimethoprim, trimethoprim and sulfamethoxazole), a tetracycline (i.e., tetracycline, demeclocy cline, doxycycline, minocycline and oxytetracycline), an antineo- plastic or cytotoxic antibiotic (i.e., doxorubicin, mitoxantrone, bleomycin, daunorubicin, dactinomycin, epirubicin, idarubicin, plicamycin, mitomycin, pentostatin and valrubicin) and a miscellaneous antibacterial compound (i.e., bacitracin, colistin and polymyxin B)), an antifungal (i.e., metronidazole, nitazoxanide, imidazole, chloroquine, iodoquinol and paromomycin), and an antiparasitic (including, but not limited to, quinine, chloroquine, amodiaquine, pyrimethamine, sulphadoxine, proguanil, mefloquine, atovaquone, primaquine, artemesinin, halofantrine, doxycycline, clindamycin, mebendazole, pyrantel pamoate, thiabendazole, diethyl carb amazine, ivermectin, rifampin, amphotericin B, melarsoprol, efornithine and albendazole). For ischemia, a neurological drug may be selected that includes, but is not limited to, a thrombolytic (i.e., urokinase, alteplase, reteplase and tenecteplase), a platelet aggregation inhibitor (i.e., aspirin, cilostazol, clopidogrel, prasugrel and dipyridamole), a statin (i.e., lovastatin, pravastatin, fiuvastatin, rosuvastatin, atorvastatin, simvastatin, cerivastatin and pitavas- tatin), and a compound to improve blood flow or vascular flexibility, including, e.g., blood pressure medications.
For a behavioral disorder, a neurological drug may be selected from a behavior- modifying compound including, but not limited to, an atypical antipsychotic (i.e., risperidone, olanzapine, apripiprazole, quetiapine, paliperidone, asenapine, clozapine, iloperidone and ziprasidone), a pheno thiazine antipsychotic (i.e., prochlorperazine, chlorpromazine, fluphena- zine, perphenazine, trifluoperazine, thioridazine and mesoridazine), a thioxanthene (i.e., thiothixene), a miscellaneous antipsychotic (i.e., pimozide, lithium, molindone, haloperidol and loxapine), a selective serotonin reuptake inhibitor (i.e., citalopram, escitalopram, paroxetine, fluoxetine and sertraline), a serotonin-norepinephrine reuptake inhibitor (i.e., duloxetine, venlafaxine, desvenlafaxine, a tricyclic antidepressant (i.e., doxepin, clomipramine, amoxapine,
nortriptyline, amitriptyline, trimipramine, imipramine, protriptyline and desipramine), a tetracyclic antidepressant (i.e., mirtazapine and maprotiline), a phenylpiperazine antidepressant (i.e., trazodone and nefazodone), a monoamine oxidase inhibitor (i.e., isocarboxazid, phenelzine, selegiline and tranylcypromine), a benzodiazepine (i.e., alprazolam, estazolam, flu- razeptam, clonazepam, lorazepam and diazepam), a norepinephrine-dopamine reuptake inhibitor (i.e., bupropion), a CNS stimulant (i.e., phentermine, diethylpropion, methamphetamine, dextroamphetamine, amphetamine, methylphenidate, dexmethylphenidate, lisdexamfetamine, modafinil, pemoline, phendimetrazme, benzphetamine, phendimetrazme, armodafmil, diethylpropion, caffeine, atomoxetine, doxapram, and mazindol), an anxiolytic/sedative/hypnotic (including, but not limited to, a barbiturate (i.e., secobarbital, phenobarbital and mephobarbi- tal), a benzodiazepine (as described above), and a miscellaneous anxiolytic/sedative/hypnotic (i.e. diphenhydramine, sodium oxybate, zaleplon, hydroxyzine, chloral hydrate, aolpidem, buspirone, doxepin, eszopiclone, ramelteon, meprobamate and ethclorvynol)), a secretin (see, e.g., Ratliff- Schaub et al. Autism 9: 256-265 (2005)), an opioid peptide (see, e.g., Cowen et al, J. Neurochem. 89:273-285 (2004)), and a neuropeptide (see, e.g., Hethwa et al. Am. J. Physiol. 289: E301-305 (2005)).
For CNS inflammation, a neurological drug may be selected that addresses the inflammation itself (i.e., anon-steroidal anti-inflammatory agent such as ibuprofen or naproxen), or one which treats the underlying cause of the inflammation (i.e., an anti-viral or anti-cancer agent).
In another embodiment, the compound is an intact or full-length antibody. Depending on the amino acid sequence of the constant domain of their heavy chains, intact antibodies can be assigned to different classes. There are five major classes of intact antibodies: IgA, IgD, IgE, IgG, and IgM, and several of these may be further divided into subclasses (isotypes), e.g., IgGl, IgG2, IgG3, IgG4, IgA, and IgA2. The heavy chain constant domains that correspond to the different classes of antibodies are called a, 6, e, y, and p, respectively. The subunit structures and three-dimensional configurations of different classes of immunoglobulins are well known. In one embodiment, the intact antibody lacks effector function.
Techniques for generating antibodies are known and examples provided above in the definitions section of this document. In one embodiment, the antibody is a chimeric, humanized, or human antibody or antigen-binding fragment thereof.
Assays for evaluating uptake of systemically administered multispecific antibody and/or conjugate and other biological activity of multispecific antibody and/or conjugate can be performed as disclosed in the examples or as known for the multispecific antibody and/or conjugate of interest. Mesuring the concentration within the parenchyma space of CNS can also be used using for example microdialysis or the capillary depletion method combined with ELISA or radioactivity measurements of labeled multispecific antibody and/or conjugate.
PHARMACEUTICAL FORMULATIONS
Therapeutic formulations of the antibody or conjugate used in accordance with the present invention are prepared for storage by mixing with optional pharmaceutically acceptable carriers, excipients or stabilizers {Remington’s Pharmaceutical Sciences 16th edition, Osol, A. Ed. (1980)), in the form of lyophilized formulations or aqueous solutions. Acceptable carriers, excipients, or stabilizers are nontoxic to recipients at the dosages and concentrations employed, and include buffers such as phosphate, citrate, and other organic acids; anti-
oxidants including ascorbic acid and methionine; preservatives (such as octadecyldimethylbenzyl ammonium chloride; hexamethonium chloride; benzalkonium chloride, benzetho- nium chloride; phenol, butyl or benzyl alcohol; alkyl parabens such as methyl or propyl paraben; catechol; resorcinol; cyclohexanol; 3-pentanol; and m-cresol); low molecular weight (less than about 10 residues) polypeptides; proteins, such as serum albumin, gelatin, or immunoglobulins; hydrophilic polymers such as polyvinylpyrrolidone; amino acids such as glycine, glutamine, asparagine, histidine, arginine, or lysine; monosaccharides, disaccharides, and other carbohydrates including glucose, mannose, or dextrins; chelating agents such as EDTA; sugars such as sucrose, mannitol, trehalose or sorbitol; salt-forming counter-ions such as sodium; metal complexes (e.g. Zn-protein complexes); and/or non-ionic surfactants such as TWEEN™, PLURONICS™ or polyethylene glycol (PEG).
The formulation herein may also contain more than one active compound as necessary, optionally those with complementary activities that do not adversely affect each other. The type and effective amounts of such medicaments depend, for example, on the amount of multispecific antibody and/or conjugate present in the formulation, and clinical parameters of the subjects. Exemplary such medicaments are discussed below.
The active ingredients may also be entrapped in microcapsules prepared, for example, by coacervation techniques or by interfacial polymerization, for example, hydroxymethylcellulose or gelatin-micro capsules and poly-(methylmethacylate) micro capsules, respectively, in colloidal drug delivery systems (for example, liposomes, albumin microspheres, microemulsions, nano-particles and nanocapsules) or in macroemulsions. Such techniques are disclosed in Remington's Pharmaceutical Sciences 16th edition, Osol, A. Ed. (1980).
Sustained-release preparations may be prepared. Suitable examples of sustained- release preparations include semi-permeable matrices of solid hydrophobic polymers containing the antibody, which matrices are in the form of shaped articles, e.g. films, or microcapsules. Examples of sustained-release matrices include polyesters, hydrogels (for example, poly(2- hydroxyethyl-methacrylate), or poly(vinylalcohol)), polylactides (U.S. Pat. No. 3,773,919), copolymers of L-glutamic acid and y ethyl-L-glutamate, non-degradable ethylene- vinyl acetate, degradable lactic acid-glycolic acid copolymers such as the LUPRON DEPOT™ (injectable microspheres composed of lactic acid-glycolic acid copolymer and leuprolide acetate), and poly-D-(-)-3-hydroxybutyric acid.
The formulations to be used for in vivo administration must be sterile. This is readily accomplished by filtration through sterile filtration membranes. In one embodiment the formulation is isotonic.
The multispecific antibody or the conjugate of the invention may be utilized in a variety of in vivo methods. For example, the invention provides a method of transporting a therapeutic compound across the BBB comprising exposing multispecific antibody and/or conjugate to the BBB such that the multispecific antibody transports the therapeutic compound coupled thereto across the BBB. In another example, the invention provides a method of transporting a neurological disorder drug across the BBB comprising exposing the conjugate to the BBB such that the multispecific antibody transports the neurological disorder drug coupled thereto across the BBB. In one embodiment, the BBB here is in a mammal (e.g. a human), e.g. one which has a neurological disorder, including, without limitation: Alzheimer's disease (AD), stroke, dementia, muscular dystrophy (MD), multiple sclerosis (MS), amyotrophic lateral sclerosis (ALS), cystic fibrosis, Angelman's syndrome, Liddle syndrome, Parkinson's disease, Pick's disease, Paget's disease, cancer, traumatic brain injury, etc.
In one embodiment, neurological disorder is selected from: a neuropathy, amyloidosis, cancer (e.g. involving the CNS or brain), an ocular disease or disorder, a viral or microbial infection, inflammation (e.g. of the CNS or brain), ischemia, neurodegenerative disease, seizure, behavioral disorder, lysosomal storage disease, etc.
Neuropathy disorders are diseases or abnormalities of the nervous system characterized by inappropriate or uncontrolled nerve signaling or lack thereof, and include, but are not limited to, chronic pain (including nociceptive pain), pain caused by an injury to body tissues, including cancer-related pain, neuropathic pain (pain caused by abnormalities in the nerves, spinal cord, or brain), and psychogenic pain (entirely or mostly related to a psychological disorder), headache, migraine, neuropathy, and symptoms and syndromes often accompanying such neuropathy disorders such as vertigo or nausea.
Amyloidoses are a group of diseases and disorders associated with extracellular proteinaceous deposits in the CNS, including, but not limited to, secondary amyloidosis, age- related amyloidosis, Alzheimer's Disease (AD), mild cognitive impairment (MCI), Lewy body dementia, Down's syndrome, hereditary cerebral hemorrhage with amyloidosis (Dutch type); the Guam Parkinson-Dementia complex, cerebral amyloid angiopathy, Huntington's disease, progressive supranuclear palsy, multiple sclerosis; Creutzfeld Jacob disease, Parkinson's disease, transmissible spongiform encephalopathy, HIV-related dementia, amyotropic lateral sclerosis (ALS), inclusion-body myositis (IBM), and ocular diseases relating to beta-amyloid deposition (i.e., macular degeneration, drusen-related optic neuropathy, and cataract).
Cancers of the CNS are characterized by aberrant proliferation of one or more CNS cell (i.e., a neural cell) and include, but are not limited to, glioma, glioblastoma multiforme, meningioma, astrocytoma, acoustic neuroma, chondroma, oligodendroglioma, medulloblastomas, ganglioglioma, Schwannoma, neurofibroma, neuroblastoma, and extradural, intramedullary or intradural tumors.
Viral or microbial infections of the CNS include, but are not limited to, infections by viruses (i.e., influenza, HIV, poliovirus, rubella, ), bacteria (i.e., Neisseria sp., Streptococcus sp., Pseudomonas sp., Proteus sp., E. coli, S. aureus, Pneumococcus sp., Meningococcus sp., Haemophilus sp., and Mycobacterium tuberculosis) and other microorganisms such as fungi (i.e., yeast, Cryptococcus neoformans), parasites (i.e., toxoplasma gondii) or amoebas resulting in CNS pathophysiologies including, but not limited to, meningitis, encephalitis, myelitis, vasculitis and abscess, which can be acute or chronic. Inflammation of the CNS is inflammation that is caused by an injury to the CNS, which can be a physical injury (i.e., due to accident, surgery, brain trauma, spinal cord injury, concussion) or an injury due to or related to one or more other diseases or disorders of the CNS (i.e., abscess, cancer, viral or microbial infection).
Ischemia of the CNS, as used herein, refers to a group of disorders relating to aberrant blood flow or vascular behavior in the brain or the causes therefor, and includes, but is not limited to: focal brain ischemia, global brain ischemia, stroke (i.e., subarachnoid hemorrhage and intracerebral hemorrhage), and aneurysm.
Neurodegenerative diseases are a group of diseases and disorders associated with neural cell loss of function or death in the CNS, and include, but are not limited to: adrenoleukodystrophy, Alexander's disease, Alper's disease, amyotrophic lateral sclerosis, ataxia telangiectasia, Batten disease, cockayne syndrome, corticobasal degeneration, degeneration caused by or associated with an amyloidosis, Friedreich's ataxia, frontotemporal lobar degeneration,
Kennedy's disease, multiple system atrophy, multiple sclerosis, primary lateral sclerosis, progressive supranuclear palsy, spinal muscular atrophy, transverse myelitis, Refsum's disease, and spinocerebellar ataxia.
Seizure diseases and disorders of the CNS involve inappropriate and/or abnormal electrical conduction in the CNS, and include, but are not limited to: epilepsy (i.e. , absence seizures, atonic seizures, benign Rolandic epilepsy, childhood absence, clonic seizures, complex partial seizures, frontal lobe epilepsy, febrile seizures, infantile spasms, juvenile myoclonic epilepsy, juvenile absence epilepsy, Lennox-Gastaut syndrome, Landau-Kleffher Syndrome, Dravef s syndrome, Otahara syndrome, West syndrome, myoclonic seizures, mitochondrial disorders, progressive myoclonic epilepsies, psychogenic seizures, reflex epilepsy, Rasmussen's Syndrome, simple partial seizures, secondarily generalized seizures, temporal lobe epilepsy, tonicionic seizures, tonic seizures, psychomotor seizures, limbic epilepsy, partialonset seizures, generalized-onset seizures, status epilepticus, abdominal epilepsy, akinetic seizures, autonomic seizures, massive bilateral myoclonus, catamenial epilepsy, drop seizures, emotional seizures, focal seizures, gelastic seizures, Jacksonian March, Lafora Disease, motor seizures, multifocal seizures, nocturnal seizures, photosensitive seizure, pseudo seizures, sensory seizures, subtle seizures, sylvan seizures, withdrawal seizures, and visual reflex seizures).
Behavioral disorders are disorders of the CNS characterized by aberrant behavior on the part of the afflicted subject and include, but are not limited to: sleep disorders (i.e., insomnia, parasomnias, night terrors, circadian rhythm sleep disorders, and narcolepsy), mood disorders (i.e., depression, suicidal depression, anxiety, chronic affective disorders, phobias, panic attacks, obsessive-compulsive disorder, attention deficit hyperactivity disorder (ADHD), attention deficit disorder (ADD), chronic fatigue syndrome, agoraphobia, post-traumatic stress disorder, bipolar disorder), eating disorders (i.e., anorexia or bulimia), psychoses, developmental behavioral disorders (i.e., autism, Ret's syndrome, Aspberger's syndrome), personality disorders and psychotic disorders (i.e., schizophrenia, delusional disorder, and the like).
Lysosomal storage disorders are metabolic disorders which are in some cases associated with the CNS or have CNS-specific symptoms; such disorders include, but are not limited to: Tay-Sachs disease, Gaucher's disease, Fabry disease, mucopolysaccharidosis (types I, II, III, IV, V, VI and VII), glycogen storage disease, GM1 -gangliosidosis, metachromatic leukodystrophy, Farber's disease, Canavan's leukodystrophy, and neuronal ceroid lipofuscinoses types 1 and 2, Niemann-Pick disease, Pompe disease, and Krabbe's disease.
The multispecific antibody and/or conjugate of the invention can be used either alone or in combination with other agents in a therapy. For instance, the multispecific antibody and/or conjugate of the invention may be co-administered with at least one additional therapeutic agent. In certain embodiments, an additional therapeutic agent is a therapeutic agent effective to treat the same or a different neurological disorder as the multispecific antibody and/or conjugate of the invention is being employed to treat. Exemplary additional therapeutic agents include, but are not limited to: the various neurological drugs described above, cholinesterase inhibitors (such as donepezil, galantamine, rovastigmine, and tacrine), NMDA receptor antagonists (such as memantine), amyloid beta peptide aggregation inhibitors, antioxidants, y-secretase modulators, nerve growth factor (NGF) mimics or NGF gene therapy, PPARy agonists, HMS-CoA reductase inhibitors (statins), ampakines, calcium channel block-
ers, GABA receptor antagonists, glycogen synthase kinase inhibitors, intravenous immunoglobulin, muscarinic receptor agonists, nicrotinic receptor modulators, active or passive amyloid beta peptide immunization, phosphodiesterase inhibitors, serotonin receptor antagonists and anti-amyloid beta peptide antibodies. In certain embodiments, the at least one additional therapeutic agent is selected for its ability to mitigate one or more side effects of the neurological drug.
Such combination therapies noted above encompass combined administration (where two or more therapeutic agents are included in the same or separate formulations), and separate administration, in which case, administration of the multispecific antibody and/or conjugate of the invention can occur prior to, simultaneously, and/or following, administration of the additional therapeutic agent and/or adjuvant. Multispecific antibodys and/or conjugates of the invention can also be used in combination with other interventional therapies such as, but not limited to, radiation therapy, behavioral therapy, or other therapies known in the art and appropriate for the neurological disorder to be treated or prevented. The multispecific antibody and/or conjugate of the invention (and any additional therapeutic agent) can be administered by any suitable means, including parenteral, intrapulmonary, and intranasal, and, if desired for local treatment, intralesional administration. Parenteral infusions include intramuscular, intravenous, intraarterial, intraperitoneal, or subcutaneous administration.
Dosing can be by any suitable route, e.g. by injections, such as intravenous or subcutaneous injections, depending in part on whether the administration is brief or chronic. Various dosing schedules including but not limited to monovalent or multiple administrations over various time- points, bolus administration, and pulse infusion are contemplated herein.
Multispecific antibody and/or conjugates of the invention would be formulated, dosed, and administered in a fashion consistent with good medical practice. Factors for consideration in this context include the particular disorder being treated, the particular mammal being treated, the clinical condition of the individual patient, the cause of the disorder, the site of delivery of the agent, the method of administration, the scheduling of administration, and other factors known to medical practitioners. The multispecific antibody and/or conjugates of the invention need not be, but is optionally formulated with one or more agents currently used to prevent or treat the disorder in question. The effective amount of such other agents depends on the amount of multispecific antibody and/or conjugate present in the formulation, the type of disorder or treatment, and other factors discussed above. These are generally used in the same dosages and with administration routes as described herein, or about from 1 to 99% of the dosages described herein, or in any dosage and by any route that is empirically/clinically determined to be appropriate.
For the prevention or treatment of disease, the appropriate dosage of multispecific antibody and/or conjugate of the invention (when used alone or in combination with one or more other additional therapeutic agents) will depend on the type of disease to be treated, the type of multispecific antibody and/or conjugate, the severity and course of the disease, whether the antibody is administered for preventive or therapeutic purposes, previous therapy, the patient's clinical history and response to the multispecific antibody and/or conjugate, and the discretion of the attending physician. The multispecific antibody and/or conjugate is suitably administered to the patient at one time or over a series of treatments. Depending on the type and severity of the disease, about 1 pg/kg to 15 mg/kg (e.g. 0.1 mg/kg- lOmg/kg) of multispecific antibody and/or conjugate can be an initial candidate dosage for administration to the patient,
whether, for example, by one or more separate administrations, or by continuous inmultimeric. One typical daily dosage might range from about 1 pg/kg to 100 mg/kg or more, depending on the factors mentioned above. For repeated administrations over several days or longer, depending on the condition, the treatment would generally be sustained until a desired suppression of disease symptoms occurs. One exemplary dosage of the antibody would be in the range from about 0.05 mg/kg to about 10 mg/kg. Thus, one or more doses of about 0.5 mg/kg, 2.0 mg/kg, 4.0 mg/kg or 10 mg/kg (or any combination thereof) may be administered to the patient. Such doses may be administered intermittently, e.g. every week or every three weeks (e.g. such that the patient receives from about two to about twenty, or e.g. about six doses of the antibody). An initial higher loading dose, followed by one or more lower doses may be administered. However, other dosage regimens may be useful. The progress of this therapy is easily monitored by conventional techniques and assays.
Table 1 : Bispecific antibody amino acid sequences
Target 1 is recognized by Light chain A and Heavy chain K Target 2 is recognized by Light chain B and Heavy chain H
Table 2: Bispecific antibodies with target clone affinities
Table 3: Antibody affinities based on SPR measurements
EXAMPLES
Dose response of bispecific antibodies on hCMECD3 cells:
Figure 2a shows the results of the binding assay on hCMECD3 for bispecific antibodies P1AI8556 (PODXL-698-high/TfR-1026-low), P1AI8559 (PODXL-698-high/Dummy), P1AI8549 (PODXL-701 -low/TfR-1026-low), P1AI8553 (Dummy/TfR-1026-low) and P1AI8552 (PODXL-701-low/Dummy). The binding assay showed highest binding to hCMECD3 cells for P1AI8556 (698-PODXL - high/TFR - low). The combination of TFR-
low with 698-PODXL-high increased the binding properties compared to both monospecific binder controls P1AI8559 (698-PODXL-high/Dummy) and P1AI8553 (1026-TfR- low/Dummy).
Figure 2b shows the results of the binding assay on hCMECD3 for bispecific antibodies P1AI8557 (PODXL - 698 - high / TfR- Duta 3- high), P1AI8550 (PODXL-701-low/ TfR- Duta 3- low) and Pl AI8547 (CD98-705-low/TfR-Duta 3-high). The binding assay showed highest binding to hCMECD3 cells for Pl AI8557. The combination of PODXL - 698 - high / TfR- Duta 3- high increased the binding properties compared to both monospecific binder controls P1AI8559 (698-PODXL/Dummy).
Figure 2c shows the results of the binding assay on hCMECD3 for bispecific antibodies P1AI8558 (PODXL-698-high / TfR- Duta 1- medium), P1AI8559 (PODXL-698-high/ Dummy), P1AI8555 (Dummy/TfR-Duta 1-medium), P1AI8551 (PODXL-701-low/TfR-Duta 1 -medium), P1AI8552 (PODXL-701-low/Dummy), P1AG3698 (PODXL-698-high- onearmed) and P1AG3701 (PODXL-701 -low-onearmed). The binding assay showed highest binding to hCMECD3 cells for Pl AG3698, Pl AI8559 and Pl AI8558. The combination of PODXL-698-high / TfR-Duta 1-medium increased the binding properties compared to the monospecific TfR binder control P1AI8555 (Dummy/Duta- 1-medium) but not to the PODXL binder controls P1AI8558 (PODXL-698-high / TfR- Duta 1- medium) and P1AG3698 (PODXL-698-high-onearmed). The combination of PODXL-701 -low / TfR-Duta 1-medium increased the binding affinity compared to both monospecific controls P1AI8552 (PODXL- 701 -low/Dummy) and P1AI8555 (Dummy/TfR-Duta 1-medium).
Figure 2d shows the results of the binding assay on hCMECD3 for bispecific antibodies P1AK1324 (TfR-Duta 3 -high/CD98 -703 -high) and P1AK1327 (Dummy/CD98-703-high). The binding assay showed the highest binding to hCMECD3 cells for Pl AK1327 (Dummy/CD98-703-high). The combination of TfR-Duta 3-high/CD98-703-high (P1AK1324) decreased the binding affinity compared to the monospecific control P1AK1327 (Dummy/CD98-703-high)
Figure 3 shows the results of the binding assay on hCMEDCD3 for bispecific antibodies P1AI8549 (PODXL-701 -low/TfR-1026-low), P1AI8553 (Dummy/TfR) and P1AI8552 (PODXL-701 -low/Dummy). For the lower binding antibodies one could see that Pl AI8553 (1026-TFR-low/Dummy) showed the lowest binding to hCMECD3 cells. When this Tfr- binder is combined with the 701-PODXL binder this increase the binding strength: Pl AI8549 (701-PODXL-low/1026-TFR-low). But P1AI8552 (701-PODXL-low/Dummy) showed as a monospecific binder higher binding strength than the bispecific Pl AI8549 (PODXL-701- low/TfR-1026-lo w).
Figure 4 shows the results of the binding assay on hCMEDCD3 for bispecific antibodies P1AI8559 (PODXL-698-high/Dummy), P1AG3701 (PODXL-701 -low/None), P1AG3698 (PODXL-698-high/None), P1AI8552 (PODXL-701 -low/Dummy), P1AG3705 (CD98-705- low), P1AI4681 and P1AI3732. The one-arm version of 698-PODXL-high (Pl AG3698) behaves similarly to the 2-arm version where 698-PODXL-high is combined with a non-binder (P1AI8559 (PODXL-698-high/Dummy). From that one can conclude, that the shown effects are not due to the antibody format (one arm vs. two arm IgG).
Transcytosis assay results for bispecific antibodies
Figure 6a and 6b show an overview of the transcytosis assay results for the following bispecific antibodies and control antibodies: P1AI8549 (PODXL-701-low/TfR-1026-low), P1AI8552 (PODXL-701/Dummy), P1AI8553 (Dummy/TfR.-1026-low), P1AI8556 (PODXL- 698-high/TfR-1026-low)„ P1AI8559 (PODXL-698-high/Dummy), P1AK1323 (PODXL-698- high/CD98-703 -high) , P1AK1326 (PODXL-701-low/CD98-703-high), P1AK1327 (Dummy/CD98-703-high), and P1AK1325 (TfR-1026-low/CD98-703-high). Experimental setup: hCMECD3; loading: 20.1 pg/ml, conjugate 2.67 pg/ml.
Figure 6c shows the results of the transcytosis assay for a selection of bispecific antibodies, namely: P1AI8549 (PODXL-701-low/TfR-1026-low), P1AI8556 (PODXL-698- high/TfR.-1026-low), P1AK1323 (PODXL-698-high/CD98-703-high) and P1AK1325 (TfR- 1026-low/CD98-703-high). Experimental setup: hCMECD3; loading: 20.1 pg/ml, conjugate 2.67 pg/ml.
Figure 6d shows the results of the transcytosis assay for a selection of bispecific antibodies, namely: P1AI8552 (PODXL-701/Dummy), P1AI8553 (Dummy/TfR.-1026-low), P1AI8559 (PODXL-698-high/Dummy) and P1AK1327 (Dummy/CD98-703-high). Experimental setup: hCMECD3; loading: 20.1 pg/ml, conjugate 2.67 pg/ml.
Fig. 6e shows the results of the transcytosis assay for bispecific antibody P1AI8549 (PODXL-701-low/Tfr-1026-low) + Controls. Experimental setup: hCMECD3; loading: 20.1 pg/ml, conjugate 2.67 pg/ml.
Fig. 6f shows the results of the transcytosis assay for bispecific antibody P1AI8550 (PODXL-701-low/TfR-1026-low) + Controls. Experimental setup: hCMECD3; loading: 20.1 pg/ml, conjugate 2.67 pg/ml.
Fig. 6g shows the results of the transcytosis assay for bispecific antibody Pl AI8551 (CD98-705-low/TfR-duta 1 - medium) + Controls. Experimental setup: hCMECD3; loading:
20.1 pg/ml, conjugate 2.67 pg/ml.
Fig. 6h shows the results of the transcytosis assay for bispecific antibody P 1 AI8547 (PODXL-701-low/Tfr-Duta 3 - high) + Controls. Experimental setup: hCMECD3; loading:
20.1 pg/ml, conjugate 2.67 pg/ml.
Fig. 6i shows the results of the transcytosis assay for bispecific antibody Pl AI8557 (PODXL-698-high/Tfr-Duta 3 - high) + Controls. Experimental setup: hCMECD3; loading:
20.1 pg/ml, conjugate 2.67 pg/ml.
Fig. 6j shows the results of the transcytosis assay for bispecific antibody Pl AI8558 (PODXL-698-high/TfR.-Duta 1 - medium) + Controls. Experimental setup: hCMECD3; loading: 20.1 pg/ml, conjugate 2.67 pg/ml.
Fig. 6k shows the results of the transcytosis assay for bispecific antibody PIAK1324 (TfR-Duta 3 - high/CD98 - 703-high) + Controls. Experimental setup: hCMECD3; loading: 20.1 pg/ml, conjugate 2.67 pg/ml.
For the transcytosis similar effects than for the binding properties are seen P1AI8556 (PODXL-698-high/TfR-1026-low) shows increased transcytosis compared to both monospecific controls: Pl AI8553 (1026-TFR-low/Dummy) and Pl AI8559 (698-PODXL- high/Dummy).
The same can be observed for P1AK1323 (PODXL-698-high/CD98-703-high), where P1AK1327 (Dummy/CD98-703 -high) represents the monospecific 703-CD98 control and P1AI8559 (698-PODXL-high/Dummy) the other control.
P1AK1326 (PODXL-701-low/CD98 -703 -high), shows increased transcytosis compared to the control P1AI8552 (701-PODXL-low/Dummy).
P1AK1325 (1026-TFR-low/ CD98-703-high) shows increased transcytosis compared to the control P1AI8553 (1026-TFR-low/Dummy)
P1AI8549 (701-PODXL-low/1026-TFR-low) shows increased transcytosis compared to the control P1AI8552 (701-PODXL-low/Dummy).
Figure 7 shows the results of the transcytosis assay results for bispecific antibodies targeting TfR/PODXL TfR/CD98 and PODXL/CD98 after 5h trafficking. After 5h of intracellular transport Pl AK1325 (1026-TFR-low/ CD98 -703 -high) shows the highest transcytosis, followed by P1AK1323 (698-PODXL-high/703-CD98-high). Furthermore P1AK1326 (701- PODX-lowL/703-CD98-high), P1AI8559 (698-PODXL-high/Dummy) and P1AI8556 (698- PODXL-high/1026-TFR)-low show also high levels of transcytosis.
Figure 8 shows the results of the transcytosis assay for bispecific antibodies targeting TfR/PODXL TfR/CD98 and PODXL/CD98 after 20h trafficking. After 20h of intracellular transport Pl AK1323 (698-PODXL-high/703-CD98-high) shows the highest transcytosis, followed by P1AK1325 (1026-TFR-low/ CD98-703-high).
Furthermore P1AK1326 (701-PODXL-low/703-CD98-high), Pl AI8559 (698- PODXL-high/Dummy) and P1AI8556 (698-PODXL-high/1026-TFR-low) show also high levels of transcytosis.
Figure 13 shows the results of the transcytosis assay for monospecific antibodies targeting PODXL. Pl AG3700 shows the highest transcytosis. Experimental setup: hCMECD3; loading: 20.1 pg/ml.
MATERIAL & METHODS
Antibody production, purification & analysis:
Expi293 cells were transiently transfected in a 1.0 L production scale with plasmids in a ratio of 1 : 1 : 1 : 1 encoding the protein. Protein was purified by affinity chromatography
(MabSelect SuRe), ion exchange chromatography (POROS XS), and size exclusion chromatography. For quality control the endotoxin levels, (<0.5 EU/mg (kinetic LAL)), the
monomer content >96 % (aSEC, after lx Freeze/Thaw) and the purity (capillary gel electrophoresis) of the antibodies were analyzed.
Binding Assay:
For the FACS binding assay hCMECD3 cells were incubated for Ih with the antibodies at concentrations of 1000 nM, 500 nM, 250 nM, 125 nM, 63 nM, 31 nM, 16 nM, 8 nM, 4 nM, 2 nM and 1 nM. After washing the cells with PBS (DPBS w/o Calcium, w/o Magnesium, sterile; PANBiotech; P04-36500) + 5 % FBS (FBS Premium, Australia origin fetal bovine serum gamma irradiated, 0.2um sterile filtered; PANBiotech; P30-2006) a secondary antibody (R-Phycoerythrin AffiniPure F(ab')2 Fragment Donkey Anti-Human IgG (H+L); Jackson Immuno Research; 709-116-149) was added (1:100 diluted). After 1 hour of incubation and after washing the cells with PBS (DPBS w/o Calcium, w/o Magnesium, sterile; PANBiotech; P04- 36500) + 5 % FBS (FBS Premium, Australia origin fetal bovine serum gamma irradiated, 0.2um sterile filtered; PANBiotech; P30-2006), the cells were analyzed with flow cytometry (BD FACSCelesta Cell Analyzer) for PE intensity. The results were analyzed with the softwares FlowJo and Graphpad Prism.
Transcytosis Assay:
For the transcytosis assay 12-well cell culture inserts (cellQART; 9310402) were coated with 400 pl collagen (Coming; 354236) and 200 pl Fibronectin (Gibco; 33016-015) both in a concentration of 50 pg/ml for Ih. After washing the inserts two times with DPBS CaC12, CaMg2 (Gibco; 14040-091) and stored at 4°C for up to one week until seeding of cells. Before seeding the cells, the plate was filled with prewarmed cell culture medium (EBM-2 Basal medium; Lonza; CC-3156 and EGM-2 MV SingleQuots; Lonza; CC-4147), 1.6 ml basolateral and 0.4 ml apical. HCMECD3 cells were thawed and 8000 cells/filter were seeded on the apical part of the transwell insert and cultured at 37 °C and 5 % CO2 for 12 days with changing the medium every 2-3 days. On day 12, fresh cell culture medium was added basolateral and the apical medium was exchanged by a previously prepared antibody dilution (21.0 pg/ml) and incubated for 1 h at 37 °C and 5 % CO2 to allow intracellular uptake. After washing 5 times basolateral and apical with DPBS CaCh, CaMg2 + 2.5 % fetal bovine serum (Lonza; CC-4102B), fresh cell culture medium was added and the cells are incubated at 37 °C and 5 % CO2 for 0 h, 1 h, 2 h, 4 h, 5 h and 20 h. At every time point, samples were taken of the apical and basolateral supernatant and cell lysates were made, each from two replicates. For the cell lysates, the whole cell culture medium was removed and cells were lysed in RIP A buffer (Sigma Aldrich; R0278). The samples were stored at -80°C and the IgG amount was measured via huIgG ELISA and analyzed using Excel and XLfit. huIgG ELISA
To measure the IgG amount in the supernatants and cell lysates of the transcytosis assay, the samples were measured via ELISA. For this, 384-well maxisorp plates (Thermo Scientific; 460372) were coated with 30 pl of AffiniPure Goat Anti-Human IgG Fcy-specific coating antibody (BIOZOL; 109-005-098) in a concentration of 1 pg/ml overnight at 4 °C. At the day of the ELISA, the coating antibody was removed and replaced by 100 pl PBS w/o Ca2+ & Mg2+ (PAN Biotech; P04-991500) + 0,5 % BSA (Thermo Scientific; 37525) + 0,05 % Tween 20 (anatrace; T1003). A standard curve of the respective antibody was prepared in
EBM-2 Basal medium (Lonza; CC-3156) or RIPA buffer (Sigma Aldrich; R0278) with concentrations of 0 ng/ml, 0.0004 ng/ml, 0.0011 ng/ml 0.004 ng/ml, 0.011 ng/ml, 0.03 ng/ml, 0.1 ng/ml, 0.3 ng/ml, 0.9 ng/ml, 2.77 ng/ml, 8.33 ng/ml and 25 ng/ml. In addition a 1 :3 dilution series was prepared with every sample starting with the undiluted sample up to a 1 :729 dilution in EBM-2 Basal medium (Lonza; CC-3156) or RIPA buffer (Sigma Aldrich; R0278), for supernatant or cell lysates, respectively. The coated and blocked 384-well plate was washed four times with lOx PBST Buffer (Serva; 42597.01) 1 :10 diluted in millipore H2O. The standard curve and diluted samples were transferred to the 384-well plate and incubated at room temperature shaking at 450 rpm for 2 h. After four times of washing with PBST, 30 pl of Bio- tin-SP AffiniPure F(ab’)2 Fragment Goat Anti-Human IgG (BIOZOL; 109-066-097) was added in a concentration of 50 ng/ml and incubated at room temperature shaking at 450 rpm for 1 h. After washing six times with PBST, 30 pl streptavidin poly-hrp40 conjugate (Fitzgerald; 65R-S104HRP) was added in a concentration of 10 ng/ml and incubated at room temperature, shaking at 450 rpm for 20 min protected from light. After washing six times with PBST, 100 pl of SuperSignal ELISA Pico Chemiluminescence Substrate (Thermo; 37069) was added, incubated at room temperature for 5 min protected from light and the luminescence was measured with Tecan Infinite F200 (TEC AN) without shaking, with an attenuation of GDI and an integration time of 100 ms. Calculations were performed using Excel and XLfit.
Spheroid Assay:
References:
Simonneau et al., Fluids Barriers CNS (2021) Kassianidou and Simonneau et al., Bio Pro toe (2022)
Fabrication of hydrogel-based U-bottom microwell arrays
For the generation of high-throughput BBB spheroids, microcavities of 600 pm in diameter and 720 pm in depth imprinted in polyethylene glycol (PEG) hydrogels were typically used (GRI3D® 96-well plate, SunBioscience).
Culture conditions
Primary human astrocytes (HA, ScienCell Research Laboratories) were grown in Astrocyte Growth Medium (AGM) composed of Astrocyte Medium (AM, ScienCell Research Laboratories) supplemented with 2% FBS, 1% Astrocytes Growth Supplement (AGS, ScienCell Research Laboratories) and 1% penicillin/streptomycin. Human brain microvascular pericytes (HBVP, ScienCell Research Laboratories) were cultured in Pericyte Growth Medium (PGM) composed of Pericytes Medium (PM, ScienCell Research Laboratories) supplemented with 2% FBS, 1% Pericytes Growth Supplement (PGS, ScienCell Research Laboratories) and 1% penicillin/streptomycin. Human cerebral microvascular endothelial cells (hCMEC/D3, Merck) were maintained in culture using Endothelial Basal Medium (EBM-2, Lonza) supplemented with hydrocortisone, GA-1000, 2% FBS, hEGF, VEGF, hFGF-B, R3-IGF-1, ascorbic acid and heparin (EGM-2 SingleQuots Supplements, Lonza). Cells were grown in T-75 flasks, coated with 2 pg/cm2 of polyl-lysine for HBVP and HA. The growth medium was changed every two days. For experimental use, cells were grown until 90% confluence before subculturing; HA and HBVP were maintained between passages pl and 4 and hCMEC/D3 cells
were used for 10 passages. For the generation of BBB spheroids, EGM-2 medium without VEGF was used, and named hereinafter Spheroid Medium (SM).
BBB spheroid generation
HA, HBVP were detached by 0.05% trypsin/EDTA (ThermoFisher Scientific) and hCMEC/D3 with 0.25% trypsin/EDTA and resuspended in warm SM. The cells were resuspended at the appropriate concentration to target 1000 cells per micro well in a 1 : 1 : 1 ratio (i.e. a total of 3000 cells per micro well) in a seeding volume of 60 pL per well. 150 uL of media was added after 20 min. The cells were grown in a humidified incubator at 37 °C with 5% CO2 for 48 h (with a medium refresh after 24h) to allow self-assembly of the multicellular spheroids
Transcytosis assay with BBB spheroid arrays
After 48 h of assembly, BBB spheroid arrays were incubated with the tested antibodies targeting the human transferrin receptor and a non-targeting human IgG as a control in SM for 4 h at 37 °C with 5% CO2. After incubation, BBB spheroids were washed thoroughly 6 times for 5 min with pre-warmed SM in the incubator and then they were fixed in 4% paraformaldehyde (PF A) for 20 min at room temperature (RT). Samples were washed thoroughly with DPBS and permeabilized and blocked with 0.6% Triton-X + 10% donkey serum in DPBS for 1 h at RT.
Immunofluorescence staining of BBB spheroids
BBB spheroids were flushed from the plate with washing buffer (DPBS with 0.1% Triton-X) and transferred into 1.5-mL Protein LoBind tubes. After 2-3 min, when spheroids were settled to the bottom of the tube, supernatant was removed. Anti-Human FcY (H+L) secondary antibody (Jackson ImmunoResearch 709-545-098; 488 fluorescently labelled) and DAPI (1 pg/mL, Sigma-Aldrich) were then added to the spheroids and incubated for 1 h at RT on a rotary shaker in the dark. Finally, the samples were washed again with washing buffer, transferred to cover glasses, and mounted with Fluoromount (Electron Microscopy Science).
Imaging of BBB spheroids
Spheroids were imaged using a Leica Microsystems, Thunder Imager 3D Assay with a 20 x /0.55 Ph2 dry objective. The images were acquired with a 2x2 binning in a 16 bit format. Two different channels were set up — one for DAPI (UV laser), and one that matches the fluorophore of the secondary antibody used, in this case AlexaFluor 488 (488 laser). A z- stack covering a total depth of 8.5 pm, using 8 steps with the core placed at the center (1.21 pm step size) were used.
At least 10 spheroids per condition per experiment were acquired. Instant Computational Clearing (ICC) algorithm by Leica was then applied to the images.
Data analysis of transcytosis assay
The code used for analysis is available here: https://github.com/phag- ozyt/Fiji/blob/fb365d7cl275a6b013dc32b982504dfef9653d42/MIP75ROI.
Briefly, after receiving user input, the macro opens an individual z-stack. The macro converts the multi-channel z-stack into a multi-channel maximum projection image. It then splits the multi-channel maximum projection image into individual channel images, and takes the DAPI maximum projection image to create a mask via thresholding. The macro then converts the mask into a region of interest (ROI) based on its size and shape. The ROIs are then reduced to 75%, to cover only the core of the spheroid, and exclude measurements from the endothelial surface of the spheroid. The shrunk ROIs are overlaid on top of the channel of interest (488 channel), and relevant measurements are calculated. Fluorescence intensity is reported per pm2, by dividing raw integrated density over area (pm2).
Claims
1. A multispecific antibody for use in transporting a compound across the Blood Brain Barrier (BBB), wherein the antibody binds to at least two target proteins selected from the group consisting of transferrin receptor (TFRC, TfR), CD98 (SLC3A2) and PODXL.
2. The multispecific antibody for use according to claim 1, wherein the antibody binds to TfR and CD98.
3. The multispecific antibody for use according to claim 1, wherein the antibody binds to TfR and PODXL.
4. The multispecific antibody for use according to claim 1, wherein the antibody binds to CD98 and PODXL.
5. The multispecific antibody for use according to claims 1 - 4, wherein the antibody is a human antibody.
6. The multispecific antibody for use according to claims 1 - 5, wherein the compound is a therapeutic compound.
7. The multispecific antibody for use according to claim 6, wherein the antibody is coupled to the therapeutic compound.
8. The multispecific antibody for use according to claims 1 - 7, wherein the target proteins are human proteins.
9. The multispecific antibody for use according to claims 1 - 8, wherein the antibody comprises a first antigen binding site which binds to TfR and a second antigen binding site which binds to CD98.
10. The multispecific antibody for use according to claims 1 - 8, wherein the antibody comprises a first antigen binding site which binds to TfR and a second antigen binding site which binds to PODXL.
11. The multispecific antibody for use according to claims 1 - 8, wherein the antibody comprises a first antigen binding site which binds to CD98 and a second antigen binding site which binds to PODXL.
12. The multispecific antibody for use according to claims 6 - 11, wherein the therapeutic compound is a neurological disorder drug.
13. The multispecific antibody for use according to claims 6 - 12, wherein the therapeutic compound forms one portion of the multispecific antibody.
14. The multispecific antibody for use according to claim 13, wherein the therapeutic compound forms one or two antigen binding sites of the multispecific antibody.
15. The multispecific antibody for use according to claims 13 or 14, wherein the therapeutic compound which forms one antigen binding site of the multispecific antibody recognizes a brain antigen.
16. The multispecific antibody for use according to claim 15, wherein the brain antigen is selected from the group consisting of beta-secretase 1 (BACE1), Abeta, epidermal growth factor receptor (EGFR), human epidermal growth factor receptor 2 (HER2), tau, apolipoprotein E (ApoE), alpha-synuclein, CD20, huntingtin, prion protein (PrP), leucine rich repeat kinase 2 (LRRK2), parkin, presenilin 1, presenilin 2, gamma secretase, death receptor 6 (DR6), amyloid precursor protein (APP), p75 neurotrophin receptor (p75NTR), TREM2, MS4A, TrkB, and caspase 6.
17. The multispecific antibody for use according to claims 1 - 16, wherein the antibody has a monovalent binding mode for each target protein.
18. The multispecific antibody for use according to claim 2, wherein the TfR binding site has an affinity KD (nM) in the range of 100 - 1000 and the CD98 binding site has an affinity KD (nM) in the range of 0.1 - 100.
19. The multispecific antibody for use according to claim 3, wherein the TfR binding site has an affinity KD (nM) in the range of 100 - 1000 and the PODXL binding site has an affinity KD (nM) in the range of 0.1 - 100.
20. The multispecific antibody for use according to claim 4, wherein the PODXL binding site has an affinity KD (nM) in the range of 0.1 - 100 and the CD98 binding site has an affinity KD (nM) in the range of 0.1 - 100.
21. The multispecific antibody for use according to claim 2, wherein the TfR binding site has an affinity KD (nM) in the range of 0.01 - 9 and the CD98 binding site has an affinity KD (nM) in the range of 0.1 - 100.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23187260.7 | 2023-07-24 | ||
| EP23187260 | 2023-07-24 | ||
| EP23213859.4 | 2023-12-04 | ||
| EP23213859 | 2023-12-04 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2025021790A2 true WO2025021790A2 (en) | 2025-01-30 |
| WO2025021790A3 WO2025021790A3 (en) | 2025-02-27 |
Family
ID=92106610
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/070818 Pending WO2025021790A2 (en) | 2023-07-24 | 2024-07-23 | Multispecific antibodies |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025021790A2 (en) |
Citations (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3773919A (en) | 1969-10-23 | 1973-11-20 | Du Pont | Polylactide-drug mixtures |
| US4816567A (en) | 1983-04-08 | 1989-03-28 | Genentech, Inc. | Recombinant immunoglobin preparations |
| US5208020A (en) | 1989-10-25 | 1993-05-04 | Immunogen Inc. | Cytotoxic agents comprising maytansinoids and their therapeutic use |
| US5229275A (en) | 1990-04-26 | 1993-07-20 | Akzo N.V. | In-vitro method for producing antigen-specific human monoclonal antibodies |
| WO1995002421A1 (en) | 1993-07-16 | 1995-01-26 | Alkermes, Inc. | Transferrin receptor specific ligand-neuropharmaceutical agent fusion proteins |
| US5545807A (en) | 1988-10-12 | 1996-08-13 | The Babraham Institute | Production of antibodies from transgenic animals |
| US5565332A (en) | 1991-09-23 | 1996-10-15 | Medical Research Council | Production of chimeric antibodies - a combinatorial approach |
| US5567610A (en) | 1986-09-04 | 1996-10-22 | Bioinvent International Ab | Method of producing human monoclonal antibodies and kit therefor |
| US5573905A (en) | 1992-03-30 | 1996-11-12 | The Scripps Research Institute | Encoded combinatorial chemical libraries |
| US5589369A (en) | 1992-02-11 | 1996-12-31 | Cell Genesys Inc. | Cells homozygous for disrupted target loci |
| US5591669A (en) | 1988-12-05 | 1997-01-07 | Genpharm International, Inc. | Transgenic mice depleted in a mature lymphocytic cell-type |
| WO1997030087A1 (en) | 1996-02-16 | 1997-08-21 | Glaxo Group Limited | Preparation of glycosylated antibodies |
| US5693780A (en) | 1991-07-25 | 1997-12-02 | Idec Pharmaceuticals Corporation | Recombinant antibodies for human therapy |
| WO1998058964A1 (en) | 1997-06-24 | 1998-12-30 | Genentech, Inc. | Methods and compositions for galactosylated glycoproteins |
| WO1999022764A1 (en) | 1997-10-31 | 1999-05-14 | Genentech, Inc. | Methods and compositions comprising glycoprotein glycoforms |
| WO1999051642A1 (en) | 1998-04-02 | 1999-10-14 | Genentech, Inc. | Antibody variants and fragments thereof |
| WO2000042072A2 (en) | 1999-01-15 | 2000-07-20 | Genentech, Inc. | Polypeptide variants with altered effector function |
| US20020004587A1 (en) | 2000-04-11 | 2002-01-10 | Genentech, Inc. | Multivalent antibodies and uses therefor |
| WO2003011878A2 (en) | 2001-08-03 | 2003-02-13 | Glycart Biotechnology Ag | Antibody glycosylation variants having increased antibody-dependent cellular cytotoxicity |
| US6602684B1 (en) | 1998-04-20 | 2003-08-05 | Glycart Biotechnology Ag | Glycosylation engineering of antibodies for improving antibody-dependent cellular cytotoxicity |
| US20030157108A1 (en) | 2001-10-25 | 2003-08-21 | Genentech, Inc. | Glycoprotein compositions |
| US20040093621A1 (en) | 2001-12-25 | 2004-05-13 | Kyowa Hakko Kogyo Co., Ltd | Antibody composition which specifically binds to CD20 |
| US20050123546A1 (en) | 2003-11-05 | 2005-06-09 | Glycart Biotechnology Ag | Antigen binding molecules with increased Fc receptor binding affinity and effector function |
| US20050142141A1 (en) | 2002-11-27 | 2005-06-30 | Pardridge William M. | Delivery of enzymes to the brain |
| WO2014033074A1 (en) | 2012-08-29 | 2014-03-06 | F. Hoffmann-La Roche Ag | Blood brain barrier shuttle |
| WO2014189973A2 (en) | 2013-05-20 | 2014-11-27 | Genentech, Inc. | Anti-transferrin receptor antibodies and methods of use |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2714738B1 (en) * | 2011-05-24 | 2018-10-10 | Zyngenia, Inc. | Multivalent and monovalent multispecific complexes and their uses |
| CN107207591A (en) * | 2014-12-10 | 2017-09-26 | 豪夫迈·罗氏有限公司 | Blood-brain barrier receptor antibody and application method |
| AU2016333175B2 (en) * | 2015-10-01 | 2023-08-24 | The Centre For Drug Research And Development | Anti-podocalyxin antibodies and methods of using the same |
| EP4132589A4 (en) * | 2020-04-08 | 2024-05-08 | Aliada Therapeutics, Inc. | COMPOSITIONS AND METHODS FOR DELIVERY OF THE BLOOD-BRAIN BARRIER |
-
2024
- 2024-07-23 WO PCT/EP2024/070818 patent/WO2025021790A2/en active Pending
Patent Citations (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3773919A (en) | 1969-10-23 | 1973-11-20 | Du Pont | Polylactide-drug mixtures |
| US4816567A (en) | 1983-04-08 | 1989-03-28 | Genentech, Inc. | Recombinant immunoglobin preparations |
| US5567610A (en) | 1986-09-04 | 1996-10-22 | Bioinvent International Ab | Method of producing human monoclonal antibodies and kit therefor |
| US5545807A (en) | 1988-10-12 | 1996-08-13 | The Babraham Institute | Production of antibodies from transgenic animals |
| US5591669A (en) | 1988-12-05 | 1997-01-07 | Genpharm International, Inc. | Transgenic mice depleted in a mature lymphocytic cell-type |
| US5208020A (en) | 1989-10-25 | 1993-05-04 | Immunogen Inc. | Cytotoxic agents comprising maytansinoids and their therapeutic use |
| US5229275A (en) | 1990-04-26 | 1993-07-20 | Akzo N.V. | In-vitro method for producing antigen-specific human monoclonal antibodies |
| US5693780A (en) | 1991-07-25 | 1997-12-02 | Idec Pharmaceuticals Corporation | Recombinant antibodies for human therapy |
| US5565332A (en) | 1991-09-23 | 1996-10-15 | Medical Research Council | Production of chimeric antibodies - a combinatorial approach |
| US5589369A (en) | 1992-02-11 | 1996-12-31 | Cell Genesys Inc. | Cells homozygous for disrupted target loci |
| US5573905A (en) | 1992-03-30 | 1996-11-12 | The Scripps Research Institute | Encoded combinatorial chemical libraries |
| WO1995002421A1 (en) | 1993-07-16 | 1995-01-26 | Alkermes, Inc. | Transferrin receptor specific ligand-neuropharmaceutical agent fusion proteins |
| WO1997030087A1 (en) | 1996-02-16 | 1997-08-21 | Glaxo Group Limited | Preparation of glycosylated antibodies |
| WO1998058964A1 (en) | 1997-06-24 | 1998-12-30 | Genentech, Inc. | Methods and compositions for galactosylated glycoproteins |
| WO1999022764A1 (en) | 1997-10-31 | 1999-05-14 | Genentech, Inc. | Methods and compositions comprising glycoprotein glycoforms |
| WO1999051642A1 (en) | 1998-04-02 | 1999-10-14 | Genentech, Inc. | Antibody variants and fragments thereof |
| US6602684B1 (en) | 1998-04-20 | 2003-08-05 | Glycart Biotechnology Ag | Glycosylation engineering of antibodies for improving antibody-dependent cellular cytotoxicity |
| WO2000042072A2 (en) | 1999-01-15 | 2000-07-20 | Genentech, Inc. | Polypeptide variants with altered effector function |
| US20020004587A1 (en) | 2000-04-11 | 2002-01-10 | Genentech, Inc. | Multivalent antibodies and uses therefor |
| WO2003011878A2 (en) | 2001-08-03 | 2003-02-13 | Glycart Biotechnology Ag | Antibody glycosylation variants having increased antibody-dependent cellular cytotoxicity |
| US20030157108A1 (en) | 2001-10-25 | 2003-08-21 | Genentech, Inc. | Glycoprotein compositions |
| US20040093621A1 (en) | 2001-12-25 | 2004-05-13 | Kyowa Hakko Kogyo Co., Ltd | Antibody composition which specifically binds to CD20 |
| US20050142141A1 (en) | 2002-11-27 | 2005-06-30 | Pardridge William M. | Delivery of enzymes to the brain |
| US20050123546A1 (en) | 2003-11-05 | 2005-06-09 | Glycart Biotechnology Ag | Antigen binding molecules with increased Fc receptor binding affinity and effector function |
| WO2014033074A1 (en) | 2012-08-29 | 2014-03-06 | F. Hoffmann-La Roche Ag | Blood brain barrier shuttle |
| WO2014189973A2 (en) | 2013-05-20 | 2014-11-27 | Genentech, Inc. | Anti-transferrin receptor antibodies and methods of use |
Non-Patent Citations (33)
| Title |
|---|
| "Agnew, Chem Intl. Ed. Engl", vol. 33, 1994, pages: 183 - 186 |
| "Remington's Pharmaceutical Sciences", 1980 |
| "UNIPROT", Database accession no. P08195 |
| ATWAL ET AL., SCI. TRANSL. MED., vol. 3, 2011, pages 84 - 44 |
| BRUGGERMANN ET AL., YEAR IN IMMUNO., vol. 7, 1993, pages 33 |
| CAI ET AL., J. CELL SCI., vol. 1, no. 18, 2005, pages 889 - 899 |
| CARTER P.RIDGWAY J.B.BPRESTA L.G., IMMUNOTECHNOLOGY, vol. 2, no. 1, February 1996 (1996-02-01), pages 73 - 73 |
| CHARI ET AL., CANCER RES., vol. 52, 1992, pages 127 - 131 |
| CHOTHIALESK, J. MOL. BIOL., vol. 196, 1987, pages 901 - 917 |
| CLACKSON ET AL., NATURE, vol. 352, 1991, pages 624 - 628 |
| COWEN ET AL., J. NEUROCHEM., vol. 89, 2004, pages 273 - 285 |
| FELGENHAUER, KLIN. WSCHR., vol. 52, 1974, pages 1158 - 1164 |
| FLATMAN ET AL., J. CHROMATOGR. B, vol. 848, 2007, pages 79 - 87 |
| GRIFFITH ET AL., EMBO J., vol. 12, 1993, pages 725 - 734 |
| HETHWA ET AL., AM. J. PHYSIOL., vol. 289, 2005, pages E301 - 305 |
| HUST M. ET AL., BMC BIOTECHNOL., vol. 7, 8 March 2007 (2007-03-08), pages 14 |
| JAKOBOVITS ET AL., NATURE, vol. 362, 1993, pages 255 - 258 |
| JAKOBOVITS ET AL., PROC. NATL ACAD. SCI. USA, vol. 90, 1993, pages 2551 |
| JOHNSON ET AL., CURRENT OPINION IN STRUCTURAL BIOLOGY, vol. 3, 1993, pages 564 - 571 |
| JONES ET AL., NATURE, vol. 321, 1986, pages 522 - 525 |
| KABAT ET AL.: "Sequences of Proteins of Immunological Interest", 1991, NATIONAL INSTITUTES OF HEALTH, BETHESDA |
| KASSIANIDOUSIMONNEAU ET AL., BIO PROTOC, 2022 |
| KOHLER ET AL., NATURE, vol. 256, 1975, pages 495 |
| MARKS ET AL., J. MOL. BIOL., vol. 222, 1991, pages 581 - 597 |
| MCCAFFERTY ET AL., NATURE, vol. 348, 1990, pages 552 - 553 |
| MORRISON ET AL., PROC. NATL. ACAD. SCI. USA, vol. 81, 1984, pages 6851 - 6855 |
| PARMACEK ET AL., NUCLEIC ACIDS RES., vol. 17, 1989, pages 1915 - 1931 |
| PRESTA, CURR. OP. STRUCT. BIOL, vol. 2, 1992, pages 593 - 596 |
| RATLIFF-SCHAUB ET AL., AUTISM, vol. 9, 2005, pages 256 - 265 |
| RIECHMANN ET AL., NATURE, vol. 332, 1988, pages 323 - 329 |
| SCHNEIDER ET AL., NATURE, vol. 311, 1984, pages 675 - 678 |
| SIMONNEAU ET AL., FLUIDS BARRIERS CNS, 2021 |
| ZATSEPIN ET AL., RUSS. CHEM. REV., vol. 74, 2005, pages 77 - 95 |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2025021790A3 (en) | 2025-02-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10941215B2 (en) | Low affinity blood brain barrier receptor antibodies and uses thereof | |
| EP2890712B1 (en) | Blood brain barrier shuttle | |
| US11167038B2 (en) | Methods for improving safety of blood-brain barrier transport | |
| WO2025021790A2 (en) | Multispecific antibodies |