WO2025017392A1 - Customized zirconia gel article, zirconia dental restoration and process of production - Google Patents
Customized zirconia gel article, zirconia dental restoration and process of production Download PDFInfo
- Publication number
- WO2025017392A1 WO2025017392A1 PCT/IB2024/056101 IB2024056101W WO2025017392A1 WO 2025017392 A1 WO2025017392 A1 WO 2025017392A1 IB 2024056101 W IB2024056101 W IB 2024056101W WO 2025017392 A1 WO2025017392 A1 WO 2025017392A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- sol
- zirconia
- gel
- article
- dental restoration
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K6/00—Preparations for dentistry
- A61K6/15—Compositions characterised by their physical properties
- A61K6/17—Particle size
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61C—DENTISTRY; APPARATUS OR METHODS FOR ORAL OR DENTAL HYGIENE
- A61C13/00—Dental prostheses; Making same
- A61C13/0003—Making bridge-work, inlays, implants or the like
- A61C13/0022—Blanks or green, unfinished dental restoration parts
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61C—DENTISTRY; APPARATUS OR METHODS FOR ORAL OR DENTAL HYGIENE
- A61C13/00—Dental prostheses; Making same
- A61C13/08—Artificial teeth; Making same
- A61C13/082—Cosmetic aspects, e.g. inlays; Determination of the colour
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61C—DENTISTRY; APPARATUS OR METHODS FOR ORAL OR DENTAL HYGIENE
- A61C13/00—Dental prostheses; Making same
- A61C13/08—Artificial teeth; Making same
- A61C13/083—Porcelain or ceramic teeth
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61C—DENTISTRY; APPARATUS OR METHODS FOR ORAL OR DENTAL HYGIENE
- A61C13/00—Dental prostheses; Making same
- A61C13/08—Artificial teeth; Making same
- A61C13/09—Composite teeth, e.g. front and back section; Multilayer teeth
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61C—DENTISTRY; APPARATUS OR METHODS FOR ORAL OR DENTAL HYGIENE
- A61C5/00—Filling or capping teeth
- A61C5/70—Tooth crowns; Making thereof
- A61C5/73—Composite crowns
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61C—DENTISTRY; APPARATUS OR METHODS FOR ORAL OR DENTAL HYGIENE
- A61C5/00—Filling or capping teeth
- A61C5/70—Tooth crowns; Making thereof
- A61C5/77—Methods or devices for making crowns
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K6/00—Preparations for dentistry
- A61K6/70—Preparations for dentistry comprising inorganic additives
- A61K6/78—Pigments
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K6/00—Preparations for dentistry
- A61K6/80—Preparations for artificial teeth, for filling teeth or for capping teeth
- A61K6/802—Preparations for artificial teeth, for filling teeth or for capping teeth comprising ceramics
- A61K6/804—Preparations for artificial teeth, for filling teeth or for capping teeth comprising ceramics comprising manganese oxide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K6/00—Preparations for dentistry
- A61K6/80—Preparations for artificial teeth, for filling teeth or for capping teeth
- A61K6/802—Preparations for artificial teeth, for filling teeth or for capping teeth comprising ceramics
- A61K6/811—Preparations for artificial teeth, for filling teeth or for capping teeth comprising ceramics comprising chromium oxide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K6/00—Preparations for dentistry
- A61K6/80—Preparations for artificial teeth, for filling teeth or for capping teeth
- A61K6/802—Preparations for artificial teeth, for filling teeth or for capping teeth comprising ceramics
- A61K6/813—Preparations for artificial teeth, for filling teeth or for capping teeth comprising ceramics comprising iron oxide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K6/00—Preparations for dentistry
- A61K6/80—Preparations for artificial teeth, for filling teeth or for capping teeth
- A61K6/802—Preparations for artificial teeth, for filling teeth or for capping teeth comprising ceramics
- A61K6/818—Preparations for artificial teeth, for filling teeth or for capping teeth comprising ceramics comprising zirconium oxide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K6/00—Preparations for dentistry
- A61K6/80—Preparations for artificial teeth, for filling teeth or for capping teeth
- A61K6/802—Preparations for artificial teeth, for filling teeth or for capping teeth comprising ceramics
- A61K6/82—Preparations for artificial teeth, for filling teeth or for capping teeth comprising ceramics comprising hafnium oxide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K6/00—Preparations for dentistry
- A61K6/80—Preparations for artificial teeth, for filling teeth or for capping teeth
- A61K6/802—Preparations for artificial teeth, for filling teeth or for capping teeth comprising ceramics
- A61K6/822—Preparations for artificial teeth, for filling teeth or for capping teeth comprising ceramics comprising rare earth metal oxides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K6/00—Preparations for dentistry
- A61K6/80—Preparations for artificial teeth, for filling teeth or for capping teeth
- A61K6/884—Preparations for artificial teeth, for filling teeth or for capping teeth comprising natural or synthetic resins
- A61K6/887—Compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/622—Forming processes; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/624—Sol-gel processing
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/622—Forming processes; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/626—Preparing or treating the powders individually or as batches ; preparing or treating macroscopic reinforcing agents for ceramic products, e.g. fibres; mechanical aspects section B
- C04B35/62605—Treating the starting powders individually or as mixtures
- C04B35/62645—Thermal treatment of powders or mixtures thereof other than sintering
- C04B35/62655—Drying, e.g. freeze-drying, spray-drying, microwave or supercritical drying
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/622—Forming processes; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/626—Preparing or treating the powders individually or as batches ; preparing or treating macroscopic reinforcing agents for ceramic products, e.g. fibres; mechanical aspects section B
- C04B35/62605—Treating the starting powders individually or as mixtures
- C04B35/6269—Curing of mixtures
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B41/00—After-treatment of mortars, concrete, artificial stone or ceramics; Treatment of natural stone
- C04B41/80—After-treatment of mortars, concrete, artificial stone or ceramics; Treatment of natural stone of only ceramics
- C04B41/81—Coating or impregnation
- C04B41/85—Coating or impregnation with inorganic materials
- C04B41/86—Glazes; Cold glazes
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/32—Metal oxides, mixed metal oxides, or oxide-forming salts thereof, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3231—Refractory metal oxides, their mixed metal oxides, or oxide-forming salts thereof
- C04B2235/3244—Zirconium oxides, zirconates, hafnium oxides, hafnates, or oxide-forming salts thereof
- C04B2235/3246—Stabilised zirconias, e.g. YSZ or cerium stabilised zirconia
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/60—Aspects relating to the preparation, properties or mechanical treatment of green bodies or pre-forms
- C04B2235/602—Making the green bodies or pre-forms by moulding
- C04B2235/6023—Gel casting
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/60—Aspects relating to the preparation, properties or mechanical treatment of green bodies or pre-forms
- C04B2235/602—Making the green bodies or pre-forms by moulding
- C04B2235/6026—Computer aided shaping, e.g. rapid prototyping
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/65—Aspects relating to heat treatments of ceramic bodies such as green ceramics or pre-sintered ceramics, e.g. burning, sintering or melting processes
- C04B2235/656—Aspects relating to heat treatments of ceramic bodies such as green ceramics or pre-sintered ceramics, e.g. burning, sintering or melting processes characterised by specific heating conditions during heat treatment
- C04B2235/6562—Heating rate
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/65—Aspects relating to heat treatments of ceramic bodies such as green ceramics or pre-sintered ceramics, e.g. burning, sintering or melting processes
- C04B2235/66—Specific sintering techniques, e.g. centrifugal sintering
- C04B2235/661—Multi-step sintering
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/74—Physical characteristics
- C04B2235/75—Products with a concentration gradient
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/74—Physical characteristics
- C04B2235/76—Crystal structural characteristics, e.g. symmetry
- C04B2235/762—Cubic symmetry, e.g. beta-SiC
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/74—Physical characteristics
- C04B2235/76—Crystal structural characteristics, e.g. symmetry
- C04B2235/765—Tetragonal symmetry
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/74—Physical characteristics
- C04B2235/77—Density
- C04B2235/775—Products showing a density-gradient
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/74—Physical characteristics
- C04B2235/78—Grain sizes and shapes, product microstructures, e.g. acicular grains, equiaxed grains, platelet-structures
- C04B2235/781—Nanograined materials, i.e. having grain sizes below 100 nm
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/74—Physical characteristics
- C04B2235/78—Grain sizes and shapes, product microstructures, e.g. acicular grains, equiaxed grains, platelet-structures
- C04B2235/785—Submicron sized grains, i.e. from 0,1 to 1 micron
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/80—Phases present in the sintered or melt-cast ceramic products other than the main phase
- C04B2235/85—Intergranular or grain boundary phases
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/96—Properties of ceramic products, e.g. mechanical properties such as strength, toughness, wear resistance
- C04B2235/9646—Optical properties
- C04B2235/9661—Colour
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/01—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on oxide ceramics
- C04B35/48—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on oxide ceramics based on zirconium or hafnium oxides, zirconates, zircon or hafnates
- C04B35/486—Fine ceramics
Definitions
- the invention relates to a customized zirconia gel article having the shape of a dental restoration and a dental restoration, in particular a monolithic zirconia ceramic dental restoration obtained therefrom, and a process of producing a zirconia ceramic dental restoration.
- the zirconia ceramic dental restoration is highly aesthetic and has adequate physical-mechanical properties.
- the zirconia ceramic dental restoration can be produced without conducting a powdercompacting step or a milling step.
- the zirconia dental restoration may also feature tailored color and translucency variations that allow it to mimic the aesthetics of natural teeth.
- US 9,039,947 B2 (Jahns et al.) describes a method for making a layered dental appliance, the method comprising: providing a mold comprising a negative of an outer shape of the layered dental appliance; positioning a slurry in the mold, forming a first layer of the layered dental appliance, the slurry comprising: (i) at least one of a glass powder and a glass ceramic powder, present in an amount of 60 wt.% to 85 wt.%, (ii) a liquid, and (iii) an inorganic binder precursor; providing a solid structure comprising a positive shape of a second layer of the layered dental appliance, wherein the solid structure includes a stamp; pressing the solid structure into the slurry in the mold; and removing the solid structure from the slurry to form a cavity comprising a negative of the second layer in the slurry.
- US 10,028,809 B2 (Jahns et al.) describes a porous dental milling block comprising at least two geometrically defined Material Sections A and B, Material Section A comprising a tetragonal zirconia crystal phase in an amount A-T in wt.% and a cubic zirconia crystal phase in an amount A- C in wt.%, Material Section B comprising a tetragonal zirconia crystal phase in an amount B-T in wt.% and a cubic zirconia crystal phase in an amount B-C in wt.%, wherein (amount of tetragonal phase A-T in wt.%) / (amount of cubic phase content A-C in wt.%) > 1 and (amount of tetragonal phase content B-T in wt.%) / (amount of cubic phase content B-C in wt.%) ⁇ 1, the material of Material Sections A and B showing a N2 adsorption and/or
- US 2022/0380260 Al (Shah et al.) relates to an additive manufacturing method of making a ceramic article, the method comprising: a) obtaining a photopolymerizable sol comprising a plurality of ceramic particles distributed in the photopolymerizable sol, wherein the ceramic particles have an average particle size diameter of 1 nanometer (nm) to 100 nm; b) selectively polymerizing the photopolymerizable sol using actinic radiation and continuous movement of a build substrate through the photopolymerizable sol to form a gelled article; c) extracting solvent from the gelled article to form an aerogel article or a xerogel article; d) heat treating the aerogel article or the xerogel article to form a porous ceramic article; e) sintering the porous ceramic article to form a sintered ceramic article; wherein the sintered ceramic article exhibits a density of 98% or greater with respect to a theoretical density of the ceramic material and wherein the sintered ceramic article exhibits
- US 10,759,707 B2 (Mayr et al.) describes a process for producing a ceramic article, the process comprising: providing a printing sol, wherein the printing sol comprising solvent, nano-sized particles, radiation curable monomer(s) and photoinitiator, and wherein the printing sol have a viscosity of less than 500 mPa*s at 23 °C.; processing the printing sol as construction material in an additive manufacturing process to obtain a 3-dimensional article being in a gel state, the 3- dimensional article having a Volume A, transferring the 3-dimensional article being in a gel state to a 3-dimensional article being in a dry state, namely an aerogel or xerogel; and applying a heat treatment step to obtain a sintered 3 ceramic article, the ceramic article having a Volume F; wherein Volume A of the 3-dimensional article in a gel state being more than 500% of Volume F of the ceramic article in its sintered state.
- US 10,532,008 B2 (Balasubramanian et al.) describes a shaded zirconia ceramic material for use in dental applications comprising an yttria-stabilized zirconia material comprising from 4.7 mol % to 5.1 mol % yttria, a coloring agent comprising at least one metal selected from: a) Tb, measured as Tb4O?, in an amount from 0.005 wt.% to 0.025 wt.%; b) Cr, measured as C O?. in an amount from 0.0002 wt.% to 0.0009 wt.%; c) Er, measured as E O?.
- the dental restoration should be sufficiently strong and highly translucent in specific regions. Ideally, the dental restorations should be provided in a “ready-to-use” form, that is without the need for an additional glazing step.
- the invention relates to a customized zirconia gel article, a dental restoration and a process of producing it as described in the claims and the description.
- the invention relates to a customized zirconia gel article having the shape of a dental restoration, the customized zirconia gel article comprising a transition area comprising a transition gel, the transition area having a color gradient, the material composition of the transition gel comprising crystalline zirconia particles, a crystal phase stabilizer component, a cured organic component, optionally a photo-initiator, a liquid, and a coloring component, wherein the crystal phase stabilizer component is contained in the crystalline zirconia particles and the coloring component can be contained in the crystalline zirconia particles or be present as a separate component in the gel.
- the invention also relates to a zirconia dental restoration which can be obtained from the customized zirconia gel article.
- Fig. 1 shows a photograph of a dental restoration according to example 1 of the invention.
- Fig. 2 shows a schematic view of a process for producing the dental restoration of example 1.
- Fig. 3 shows a photograph of a dental restoration according to example 2 of the invention.
- Fig. 4 shows a schematic view of a process for producing the dental restoration of example 2.
- Fig. 5 shows a photograph of a dental restoration according to example 3 of the invention.
- Fig. 6 shows a schematic view of a process for producing the dental restoration of example 3.
- Fig. 7 shows a photograph of a dental restoration according to comparative example 1.
- Fig. 8 shows a photograph of a dental restoration according to comparative example 2.
- a “hardenable or curable or polymerizable component” is any component which can be cured or solidified in the presence of a photo-initiator by radiation-induced polymerization or by heattreatment.
- a hardenable component may contain only one, two, three or more polymerizable groups. Typical examples of polymerizable groups include unsaturated carbon groups, such as a vinyl group being present i.a. in a (methyl) acrylate group.
- (meth)acryl is a shorthand term referring to "acryl” and/or “methacryl”.
- hardening or “curing” a composition are used interchangeably and refer to polymerization and/or crosslinking reactions including, for example, photo-polymerization reactions and chemical -polymerization techniques (e. g., ionic reactions or chemical reactions forming radicals effective to polymerize ethylenically unsaturated compounds) involving one or more materials included in the composition.
- chemical -polymerization techniques e. g., ionic reactions or chemical reactions forming radicals effective to polymerize ethylenically unsaturated compounds
- Dental article means an article which is to be used in the dental or orthodontic field, especially as or for producing a dental restoration.
- a dental article has typically two different surface portions, an outer surface and an inner surface.
- the outer surface is the surface which is not in permanent contact with the surface of a tooth.
- the inner surface is the surface which is used for attaching or fixing the dental article to a tooth. If the dental article has the shape of a dental crown or dental veneer, the inner surface has typically a concave shape, whereas the outer surface has typically a convex shape.
- a dental article should not contain components which are detrimental to the patient's health and thus free of hazardous and toxic components being able to migrate out of the dental or orthodontic article.
- Dental restoration means dental articles which are used for restoring a tooth to be treated.
- Examples of dental restorations include crowns, bridges, inlays, onlays, veneers, orthodontic articles and parts thereof.
- the dental restoration described in the present text does not have the shape of a milling block.
- Examples of orthodontic articles include brackets, buccal tubes, attachments, cleats and buttons and parts thereof.
- a “monolithic zirconia ceramic dental restoration” means a dental restoration which does not comprise ceramic phases other than zirconia or hafnia.
- a monolithic zirconia ceramic dental restoration does not comprise glass or glass ceramic materials or phases.
- a monolithic restoration comprises crystalline phases of zirconia, wherein these phases may include soluble dopant ions that effect the zirconia phase composition, translucency and color.
- Solid refers to a continuous liquid phase containing discrete particles having sizes in a range of 1 nm to 100 nm or 1 nm to 50 nm, a so called “colloidal solution”.
- the sols described in the present text are translucent and do show a so-called “Tyndall effect” or “Tyndall scattering”. The size of the particles is below the wavelength of the visible light (400 to 700 nm).
- a "gel” means a wet solid-like material in which a network of interconnected nanostructures spans the volume of a liquid medium. Gels tend to be mostly liquid in composition and typically exhibit the density of a liquid as result but have cohesiveness like a solid. A sol can become a gel when the solid nanoparticles dispersed in it can join together to form a network of particles that spans the liquid.
- Coloring components or ions shall mean components or ions which have an absorption in the spectrum visible to the human eye (e.g., from 380 to 780 nm), which results in a colored solution (visible to the human eye), if the coloring ions are dissolved in water (e.g., about 0.6 mol/1) and/or cause a coloring effect in the zirconia article containing coloring ions or components.
- a translucent composition lets light partially permeate through although it is not fully transparent, i.e. showing a significant volume scattering of the transmitted light.
- the reciprocal property of translucency is opacity (O).
- T Transmission
- I Intensity of permeated light
- I Intensity of light before permeation). So, opacity values less than about 0.9 for a 1 mm thick platelet with a diameter of 15 mm are regarded as translucent (e.g., for a measurement with a Color i7 device, X-Rite corporation USA, measurement mode: remission contrast ratio).
- Opacity can be measured by various means: in transmission, in remission, and in remission using the contrast ratio method.
- a “particle” means a substance being a solid having a shape which can be geometrically determined. The shape can be regular or irregular. Particles can typically be analysed with respect to e.g., particle size and particle size distribution.
- primary particle size refers to the size of a non-associated single particle, which is considered to be a primary particle.
- a dental ceramic article is classified as “pre-sintered” if the dental ceramic article has been treated with heat (temperature range of 900 to l,100°C) for 1 to 3 h to such an extent that the raw breaking resistance of the dental ceramic measured according to the “punch on three ball test” ISO 6872:2015 is within a range of 15 to 55 MPa or 30 to 50 MPa.
- a pre-sintered dental ceramic article usually has a porous structure and its density (usually 3.0 g/cm 3 for a 3 mol% yttrium stabilized zirconia ceramic) is less compared to a completely sintered dental ceramic framework (usually about 6.1 g/cm 3 for a 3 mol% yttrium stabilized zirconia ceramic).
- Ceramic zirconia article shall mean a 3 -dimensional article wherein at least one the x, y, z dimension is at least about 5 mm, the article being comprised of at least 80 wt.% or at least 90 wt.% zirconia.
- Ceramic means an inorganic non-metallic material that is produced by application of heat. Ceramics are usually hard and brittle and, in contrast to glasses or glass ceramics, display an essentially purely crystalline structure. “Crystalline” means a solid composed of atoms arranged in a pattern periodic in three dimensions (i.e., has long range crystal structure as determined by X-ray diffraction). Crystal structures include tetragonal, monocline, cubic zirconia and mixtures thereof.
- Diafdtration is a technique that uses ultrafdtration membranes to completely remove, replace, or lower the concentration of salts or solvents from solutions containing organic molecules. The process selectively utilizes permeable (porous) membrane filters to separate the components of solutions and suspensions based on their molecular size.
- aerogel shall mean a three-dimensional low density solid (i.e., less than 20 % of the density of an article having a porosity of less than 0.1 vol.%).
- An aerogel is a porous material derived from a gel, in which the liquid component of the gel has been replaced with a gas. The solvent removal is often done under supercritical conditions. During this process the network does not substantially shrink and a highly porous, low-density material can be obtained.
- Density means the ratio of mass to volume of an object.
- the unit of density is typically g/cm 3 .
- the density of an object can be calculated e.g., by determining its volume (e.g., by calculation or applying the Archimedes principle or method) and measuring its mass.
- tubular reactor refers to the portion of a continuous hydrothermal reactor system that is heated (i.e., the heated zone).
- the tubular reactor can be in any suitable shape.
- the shape of the tubular reactor is often selected based on the desired length of the tubular reactor and the method used to heat the tubular reactor.
- the tubular reactor can be straight, U-shaped, or coiled.
- the interior potion of the tubular reactor can be empty or can contain baffles, balls, or other known mixing techniques.
- Calcining refers to a process of heating a solid material to drive off at least 90 wt.% of volatile chemically bond components (e.g., organic components) (vs., for example, drying, in which physically bonded water is driven off by heating). Calcining is done at a temperature below a temperature needed for conducting a pre-sintering step.
- volatile chemically bond components e.g., organic components
- sintering or “firing” are used interchangeably.
- a pre-sintered ceramic article shrinks during a sintering step, that is, if an adequate temperature is applied.
- the sintering temperature to be applied depends on the ceramic material chosen. For zirconia based ceramics a typical sintering temperature range is l,100°C to l,550°C.
- Sintering typically includes the densification of a porous material to a less porous material (or a material having less cells) having a higher density, in some cases sintering may also include changes of the material phase composition (for example, a partial conversion of an amorphous phase toward a crystalline phase).
- Sintering of firing means making objects from a compressed powder by heating the material (typically below its melting point - solid state sintering) until its particles adhere to each other.
- Additional manufacturing means processes used to make 3 -dimensional articles.
- An example of an additive manufacturing technique is vat polymerisation, in particular stereolithography (SLA), digital light processing (DLP), continuous liquid interface production (CLIP) and volumetric additive manufacturing (VAM).
- SLA stereolithography
- DLP digital light processing
- CLIP continuous liquid interface production
- VAM volumetric additive manufacturing
- the articles can be of almost any shape or geometry and are produced from a 3 -dimensional model or other electronic data source.
- Ambient conditions mean the conditions which the composition described in the present text is usually subjected to during storage and handling. Ambient conditions may, for example, be a pressure of 900 to 1,100 mbar, a temperature of 10 to 40 °C and a relative humidity of 10 to 100 %. In the laboratory ambient conditions are typically adjusted to 20 to 25 °C and 1 ,000 to 1 ,025 mbar (at maritime level).
- additive(s) means that the term should include the singular and plural form.
- additive(s) means one additive and more additives (e.g. 2, 3, 4, etc.).
- a composition is “essentially or substantially free of’ a certain component, if the composition does not contain said component as an essential feature. Thus, said component is not wilfully added to the composition either as such or in combination with other components or ingredient of other components.
- a composition being essentially free of a certain component usually does not contain that component at all. However, sometimes the presence of a small amount of the said component is not avoidable e.g., due to impurities contained in the raw materials used.
- the customized zirconia gel article, the zirconia ceramic dental restoration and the process for production described in the present text are advantageous for a variety of reasons.
- the coloring concept proposed in the present text better matches with the dental situation in the mouth of a patient. Further, an undesired diffusion-controlled color gradient from the outer surface area to the inner surface area of a dental restoration can be avoided.
- dental restorations obtained by milling from a compacted zirconia dental mill blank have a larger average grain size and require a higher sintering temperature.
- the invention enables the manufacturing of dental restorations with a vertically arrangement of differently colored areas.
- a dental restoration obtained from a customized zirconia gel article comprising two different gels and comprising a diffusion-controlled transition gel area between the two gels allows for a more natural color gradient from the inner surface area of the dental restoration to the outer surface area.
- the invention also enables the manufacturing of individualized, that is, customized dental restorations.
- the convex surface of the outer surface area of the dental restoration corresponds to the concave inner surface of the respective mould used for receiving the curable sols.
- the concave surface of the inner surface area of the dental restoration corresponds to the convex outer surface of the respective mould used for receiving the curable sols.
- the inner surface of such a mould can easily be shaped or manufactured based on information associated with the dental situation of the patient. This process can therefore take advantage of both geometric and tooth shade data from an individual patient to design and produce restorations with excellent fit and aesthetics.
- the invention also enables the production of filigree dental restorations, in particular dental restoration with thin side walls.
- the invention is directed to a customized zirconia gel article having the shape of a dental restoration.
- the zirconia gel article comprises a transition area comprising a transition gel, wherein the transition area has a color gradient.
- the shape of the transition area is not particularly limited, but typically non-planar.
- the customized zirconia gel article comprises at least three areas, an inner surface area comprising gel Gi, an outer surface area comprising gel G2 and a transition area comprising transition gel G1/2.
- the areas of the zirconia gel article originate from curing the sols used for producing the customized zirconia gel article and have essentially corresponding volumes.
- the geometry of the inner surface, the outer surface or of both, the inner surface and the outer surface of the respective areas is typically based on information associated with the dental situation of a patient. That is, the geometry of at least one of the surfaces of the zirconia gel article is customized.
- the material compositions of the gels each comprise crystalline zirconia particles, a crystal phase stabilizer component, a cured organic component, in particular a radiation-cured organic component, optionally a photo-initiator, a liquid, and optionally a coloring component.
- a radiation-curable sol comprising radiation-curable components and a photo-initiator is often preferred.
- the crystal phase stabilizer component is contained in the crystalline zirconia particles and the optional coloring component can be contained in the crystalline zirconia particles or be present as a separate component.
- At least one gel and the transition gel contain a coloring component, wherein gel Gi is different from gel G2. If desired, one or more coloring components can be used.
- the transition gel comprises a mixture of the material compositions of gels Gi and G2.
- the customized zirconia gel article may have the shape of a dental crown, bridge, onlay, or veneer.
- the size of the customized zirconia gel article is typically larger than the size of the dental restoration, e.g., by a factor of at least 2 in each dimension.
- the transition area can be planar or non-planar.
- a non- planar transition gel area can be preferred as such a geometry often better mimics the structure of a natural tooth.
- the content of coloring components in the transition area is typically lower than the content of the coloring components in the inner surface area.
- the invention also relates to a process for producing the customized zirconia gel article and related dental restoration described in the present text.
- sol Si for the production at least two different curable sols are used, sol Si and sol S2.
- sols are placed into a mould having a non-planar inner bottom surface and/or on top of each other.
- the placing of the sols can be done either manually or automated, e.g., with the help of a robot.
- the placing of the sols into the mould can be done by various methods, including casting, using an inkjet-printing technology, a gradient printing technology using vat polymerization, or any other suitable technique.
- the non-planar inner bottom surface of the mould typically comprises a section with a concave region, a convex region, or a mixed concave/convex region.
- a mixed concave/convex region can be advantageous for producing dental restorations with a so-called mamelon wave geometry.
- the concave and/or convex regions are typically adapted to the shape of a tooth surface.
- the non-planar bottom surface of the mould may comprise one or more sections with a shape which is based on information associated with the dental situation of a patient.
- Such a shape facilitates the manufacturing of customized dental restoration.
- the information associated with the dental situation of a patient is so-called digital data can be obtained and provided by means known to the skilled person, e.g., by photographing or scanning the dental situation in the mouth of a patient or by using information stored in a database.
- the amounts of the sols to be used in the process is rather low.
- the sols can be used in various amounts. For producing a customized dental restoration only small amounts of sols are typically needed.
- Useful amounts may be in the range of 0.0001 to 1 ml or 0.01 to 0.5 ml.
- the individual sols are typically used in different amounts with respect to volume.
- sol Si is used in a larger amount than sol S2 and larger than optionally sols SN, e.g., by a ratio larger than 2/1 or 5/1 or 10/1 with respect to volume.
- Sol Si is typically used for forming the outer surface area of the dental restoration, whereas sol S2 and optional sols SN are used for individualizing the inner surface area of the dental restoration, which often requires a smaller amount of material.
- Sol Si, sol S2 and optional sols SN differ from each other with respect to the following properties alone or in combination: (i) content of optional coloring components, (ii) content of crystal phase stabilizing components, and/or (iii) content of crystalline zirconia particles, wherein a properties (i) and (ii) and the combination of properties (i) and (ii) is sometimes preferred.
- the process for producing the customized zirconia gel article may comprise in addition the following steps before step (a): providing sols with different contents of stabilizing and/or coloring components, mixing at least two of these sols to obtain curable sol Si, curable sol S2, and optionally curable sols SN-
- the chemical compositions of curable sol Si, curable sol S2, and optionally curable sols SN can be adjusted to match with information associated with the dental situation of a patient. This may facilitate the production of even better customized dental restorations. If desired, the curable sol S2 and/or sols SN are placed in contact with the curable sol Si only in one or more individual areas of the top surface of sol Si.
- At least two different curable sols S2 can be placed in contact with the curable sol Si only in one or more individual areas of the top surface of sol Si.
- Using a more opaque sol Si for the inner surface of the dental article can be advantageous to mask a dark surface of a tooth stump without effecting the depth of reflections on the outer surface of the dental article.
- the viscosity of each sol is typically in a range of 50 to 3,000 mPa*s or 60 to 2,000 mPa* at 23°C. Such a viscosity was found to be advantageous for a variety of reasons: It facilitates the placing of the sols into the mould by either casting or inkjet-printing. The viscosity is low enough to enable a diffusion-controlled mixing of the sols in the interface or transition area leading to a smooth color gradient from the inner surface area to the outer surface area. The viscosity is high enough to avoid an undesired blending of the sols when placing them into the mould.
- sol Si, sol S2 and optional sols SN can be further characterized by the following properties alone or in combination: being translucent in a wavelength range of 420 to 600 nm for a path length of 10 mm; showing a transmission of at least 5% at a wavelength of 420 nm for a path length of 10 mm; having a pH value in the range of 1 to 6.
- Translucent sols show less scattering of light, which is used for polymerizing the curable components contained in the sol.
- the increased translucency allows for a shallower cure gradient, which may also allow for a more uniform cure across the entire structure to be obtained, as lower energy doses are required to cure through a translucent material.
- a transmission in such a range was found to be sufficiently high for enabling the radiation -curing of the sol within an adequate time frame for the desired thickness.
- the mould into which the sols are casted has a non-planar inner bottom surface.
- the respective mould used for producing the dental restorations is customized too, in particular as regards its inner bottom surface.
- the mould or the inner bottom surface can be produced by various techniques including thermoforming, 3d-printing, or casting e.g., via lab duplication techniques in silicone, hydrocolloid, etc.
- Dental thermoforming works by applying heat and pressure to a plastic sheet which is positioned on top of a 3d-model produced (e.g., by using an additive-manufacturing technique) specifically for that patient.
- the mould can directly be produced by additive -manufacturing using the information associated with the dental situation of a patient.
- Additive-manufacturing technique which can be used include vat polymerization, stereolithography (SLA), digital light processing (DLP), robocasting (RC), material jetting (MJ), binder jetting (BJ), and volumetric additive manufacturing (VAM).
- SLA stereolithography
- DLP digital light processing
- RC robocasting
- MJ material jetting
- BJ binder jetting
- VAM volumetric additive manufacturing
- the information associated with the dental situation of a patient can be obtained by using e.g., an intraoral scanner.
- Various intraoral scanners are meanwhile commercially available on the market (e.g., from 3Shape, Planmeca, and others).
- the information can be obtained from and matched with data stored in a tooth library.
- an adequate tooth color can be determined by using either a tooth shade guide (e.g., VITA Toothguide 3D-MASTERTM), or an electronic device (e.g., VITA EasyshadeTM LITE, 3 Shape TriosTM 3).
- a tooth shade guide e.g., VITA Toothguide 3D-MASTERTM
- an electronic device e.g., VITA EasyshadeTM LITE, 3 Shape TriosTM 3
- the designing and manufacturing processes may also include Computer-Aided-Design (CAD) and/or Computer- Aided-Manufacturing (CAM). Mould dimensions are typically scaled up to account for shrinkage between the casting process and subsequent binder removal and sintering processes.
- CAD Computer-Aided-Design
- CAM Computer- Aided-Manufacturing
- moulds Any suitable material for the mould can be used. Suitable materials include PET, and polyurethanes. Alternatively, moulds may be formed by casting silicone or hydrocolloid or other conventional dental laboratory duplicating materials.
- Moulds may be of frangible (e.g., polymer) or degradable (e.g., wax) materials to accommodate undercut features.
- Moulds may be single component or may be assembled from multiple components and may contain parting lines, slides, etc. to accommodate production and reuse.
- the curable sols comprise crystalline zirconia particles, preferably in an amount of 25 to 65 wt.%, a crystal phase stabilizer component, preferably in an amount of 2 to 8 mol% calculated as oxide with respect to the total crystalline oxide particles, a curable organic component, preferably in an amount of 2 to 30 wt.%, optionally a photo-initiator, preferably in an amount of 0.01 to 3 wt.%, a liquid, preferably in an amount of 25 to 70 wt.%, optionally coloring ions, preferably in an amount of 0 to 2 wt.%, optionally inhibitor components, preferably in an amount of 0 to 0.5 wt.%, wt.% with respect to the weight of the curable sol, wherein the crystal phase stabilizer component and/or the optional coloring ions can also be contained in the crystalline zirconia particles.
- the crystalline zirconia particles in the sol typically have a primary particle size in a range of 2 nm to 50 nm (in some embodiments, 5 nm to 50 nm, 2 nm to 25 nm, 5 nm to 25 nm, 2 nm to 15 nm, or even 5 nm to 15 nm).
- the crystalline zirconia particles are typically present in the following amounts: at least 25 or at least 30 or at least 35 wt.%; at most 65 or at most 60 or at most 55 wt.%; from 25 to 65, or 30 to 60, or 35 to 55 wt.%; wt.% with respect to the weight of the sol.
- a sol with a high crystalline zirconia particles content can be advantageous as the efforts for removing the liquid in later process steps is reduced.
- a crystalline zirconia particles content in the range of 35 to 65 wt.% can be preferred.
- Crystal phase stabilizing components which can be used include ions selected from Ce, Mg, Ca, Y, La, rare earth elements or combinations thereof, wherein the use of Y is sometimes preferred.
- the crystal phase stabilizer component calculated as oxide is typically present in the following amounts: at least 2 or at least 3 or at least 4 mol%; at most 7 or at most 6 or at most 5 mol%; from 2 to 7, or 3 to 6, or 4 to 5 mol%; mol% with respect to the total crystalline oxide particles.
- the crystal phase stabilizing components are typically contained in the crystalline zirconia articles.
- the curable organic component being present in the sol can be described as first, second, third, etc. monomer.
- Zirconia particles having been surface-modified with a polymerizable agent can be polymerized, if desired, to provide a composition comprising crosslinked zirconia particles.
- a first monomer can function as a polymerizable surface modification agent. Multiple first monomers can be used.
- Surface modification agents may be represented by the formula A-B, where the A group is capable of attaching to the surface of a zirconia-based particle and the B group is radiation curable.
- Group A can be attached to the surface of the zirconia-based particle by adsorption, formation of an ionic bond, formation of a covalent bond, or a combination thereof.
- Group A examples include acidic moieties (like carboxylic acid groups, phosphoric acid groups, sulfonic acid groups and anions thereof) and silanes.
- Group B comprises a radiation curable moiety.
- Group B examples include vinyl, in particular acryl or methacryl moieties.
- Suitable surface modifying agents comprise polymerizable carboxylic acids and/or anions thereof, polymerizable sulfonic acids and/or anions thereof, polymerizable phosphoric acids and/or anions thereof, and polymerizable silanes. Suitable surface modification agents are further described, for example, in WO 2009/085926 Al (Kolb et al.).
- a radically polymerizable surface modifier is a polymerizable surface modification agent comprising an acidic moiety or anion thereof, e.g. a carboxylic acid group.
- exemplary acidic radically polymerizable surface modifiers include acrylic acid, methacrylic acid, beta-carboxyethyl acrylate, and mono-2-(methacryloxyethyl)succinate.
- Exemplary radically polymerizable surface modifiers can be reaction products of hydroxylcontaining polymerizable monomers with cyclic anhydrides such as succinic anhydride, maleic anhydride and phthalic anhydride.
- Exemplary polymerization hydroxyl-containing monomers include hydroxyethyl acrylate, hydroxyethyl methacrylate, hydroxypropyl acrylate, hydroxypropyl methacrylate, hydroxyl butyl acrylate, and hydroxybutyl methacrylate.
- Acryloxy and methacryloxy functional polyethylene oxide, and polypropylene oxide may also be used as the polymerizable hydroxyl-containing monomers.
- An exemplary radically polymerizable surface modifier for imparting both polar character and reactivity to the zirconia-containing nanoparticles is mono(methacryloxypolyethyleneglycol) succinate.
- Another example of a radically polymerizable surface modifier is a polymerizable silane.
- Exemplary polymerizable silanes include methacryloxyalkyltrialkoxysilanes or acryloxyalkyltri- alkoxysilanes (e.g., 3-methacryloxypropyltrimethoxysilane, 3-acryloxypropyltrimethoxysilane, and 3-(methacryloxy)propyltriethoxysilane); methacryloxyalkylalkyldialkoxysilanes or acryloxyalkyl- alkyldialkoxysilanes (e.g., 3-(methacryloxy)propylmethyldimethoxysilane and 3-(acryloxy- propyl)methyldimethoxy silane); methacryloxy alkyldialkylalkoxysilanes or acyrloxy alkyldialkylalkoxysilanes (e.g.
- mercaptoalkyltrialkoxylsilanes e.g., 3 -mercaptopropyltrimethoxy silane
- aryltrialkoxysilanes e.g., styrylethyltrimethoxy silane
- vinylsilanes e.g., vinylmethyldiacetoxysilane, vinyldimethylethoxysilane, vinylmethyl- diethoxysilane, vinyltrimethoxy silane, vinyltriethoxy silane, vinyltriacetoxysilane, vinyl- triisopropoxy silane, and vinyltris(2- methoxy ethoxy)silane).
- the sol described in the present text comprises one or more second monomers comprising at least two radiation-curable moieties. Those second monomer(s) may act as crosslinker(s) during the gel-forming step.
- any suitable second monomer that does not have a surface modification group can be used. That is, the optional second monomer does not have a carboxylic acid group or a silyl group.
- the second monomers are often polar monomers (e.g., non-acidic polar monomers), monomers having a plurality of polymerizable groups, alkyl (meth)acrylates and mixtures thereof.
- the presence of the monomer having a plurality of polymerizable groups tends to enhance the strength of the gel composition formed when the sol is polymerized.
- the number of polymerizable groups can be in a range of 2 to 6 or even higher. In many embodiments, the number of polymerizable groups is in a range of 2 to 5 or 2 to 4.
- the polymerizable groups are typically (meth)acryloyl groups.
- Exemplary monomers with two (meth)acryloyl groups include 1,2-ethanediol diacrylate, 1,3- propanediol diacrylate, 1,9 -nonanediol diacrylate, 1,12 -dodecanediol diacrylate, 1 ,4-butanediol diacrylate, 1,6-hexanediol diacrylate, butylene glycol diacrylate, bisphenol A diacrylate, diethylene glycol diacrylate, triethylene glycol diacrylate, tetraethylene glycol diacrylate, tripropylene glycol diacrylate, polyethylene glycol diacrylate, polypropylene glycol diacrylate, poly- ethylene/polypropylene copolymer diacrylate, polybutadiene di(meth)acrylate, propoxylated glycerine tri(meth)acrylate, and neopentylglycol hydroxypivalate diacrylate modified caprolactone.
- Exemplary monomers with three or four (meth)acryloyl groups include trimethylolpropane triacrylate (e.g., commercially available as TMPTA-NTM from Cytec Industries, Inc. (Smyrna, GA, USA) and as SR-351TM from Sartomer (Exton, PA, USA)), pentaerythritol triacrylate (e.g., commercially as SR-444TM from Sartomer), ethoxylated (3) trimethylolpropane triacrylate (e.g., commercially available as SR-454TM from Sartomer), ethoxylated (4) pentaerythritol tetraacrylate (e.g., commercially available as SR-494TM from Sartomer), tris(2-hydroxyethylisocyanurate) triacrylate (e.g., commercially available as SR-368TM from Sartomer), a mixture of pentaerythritol triacrylate and pentaerythr
- Exemplary monomers with five or six (meth)acryloyl groups include dipentaerythritol pentaacrylate (e.g., commercially available as SR-399TM from Sartomer) and a hexa-functional urethane acrylate (e.g., commercially available as CN975TM from Sartomer).
- dipentaerythritol pentaacrylate e.g., commercially available as SR-399TM from Sartomer
- a hexa-functional urethane acrylate e.g., commercially available as CN975TM from Sartomer
- the optional second monomer is a polar monomer.
- polar monomer refers to a monomer having a free radical polymerizable group and a polar group.
- the polar group is typically non-acidic and often contains a hydroxyl group, a primary amido group, a secondary amido group, a tertiary amido group, an amino group, or an ether group (i.e., a group containing at least one alkylene-oxy-alkylene group of formula -R-O-R- where each R is an alkylene having 1 to 4 carbon atoms).
- Suitable optional polar monomers having a hydroxyl group include, but are not limited to, hydroxyalkyl (meth)acrylates (e.g., 2-hydroxyethyl (meth)acrylate, 2-hydroxypropyl (meth)acrylate, 3 -hydroxypropyl (meth)acrylate, and 4-hydroxybutyl (meth)acrylate), and hydroxyalkyl (meth) acrylamides (e.g., 2-hydroxyethyl (meth)acrylamide or 3 -hydroxypropyl (meth)acrylamide), ethoxylated hydroxyethyl (meth) acrylate (e.g., monomers commercially available from Sartomer (Exton, PA, USA) as CD570TM, CD571TM, and CD572TM), and aryloxy substituted hydroxyalkyl (meth)acrylates (e.g., 2-hydroxy-2-phenoxypropyl (meth) acrylate).
- hydroxyalkyl (meth)acrylates e.g
- Exemplary polar monomers with a primary amido group include (meth)acrylamide.
- Exemplary polar monomers with secondary amido groups include, but are not limited to, N-alkyl (meth)acrylamides such as N-methyl (meth)acrylamide, N-ethyl (meth)acrylamide, N-isopropyl (meth)acrylamide, N-tert-octyl (meth)acrylamide, and N-octyl (meth)acrylamide.
- Exemplary polar monomers with a tertiary amido group include, but are not limited to, N-vinyl caprolactam, N- vinyl-2-pyrrolidone, (meth)acryloyl morpholine, and N,N-dialkyl (meth)acrylamides such as N,N- dimethyl (meth)acrylamide, N,N-diethyl (meth)acrylamide, N,N-dipropyl (meth)acrylamide, and N,N-dibutyl (meth)acrylamide.
- Polar monomers with an amino group include various N,N-dialkylaminoalkyl (meth)acrylates and N,N-dialkylaminoalkyl (meth)acrylamides.
- Examples include, but are not limited to, N,N-dimethyl aminoethyl (meth) acrylate, N,N-dimethylaminoethyl (meth) acrylamide, N,N-dimethylaminopropyl (meth)acrylate, N,N-dimethylaminopropyl (meth)acrylamide, N,N-diethylaminoethyl (methacrylate, N,N-diethylaminoethyl (meth)acrylamide, N,N-diethylaminopropyl (meth)acrylate, and N,N -diethylaminopropyl (meth) acrylamide .
- Exemplary polar monomers with an ether group include, but are not limited to, alkoxylated alkyl (meth)acrylates such as ethoxyethoxyethyl (meth)acrylate, 2 -methoxy ethyl (meth)acrylate, and 2- ethoxyethyl (meth)acrylate; and poly( alkylene oxide) (meth)acrylates such as poly(ethylene oxide) (meth)acrylates, and poly(propylene oxide) (meth)acrylates.
- the poly(alkylene oxide) acrylates are often referred to as poly(alkylene glycol) (meth)acrylates.
- These monomers can have any suitable end group such as a hydroxyl group or an alkoxy group. For example, when the end group is a methoxy group, the monomer can be referred to as methoxy poly(ethylene glycol) (meth)acrylate.
- Suitable alkyl (meth)acrylates that can be used as a second monomer can have an alkyl group with a linear, branched, or cyclic structure.
- suitable alkyl (meth)acrylates include, but are not limited to, methyl (meth)acrylate, ethyl (meth)acrylate, n-propyl (meth)acrylate, isopropyl (meth)acrylate, n-butyl (meth) acrylate, isobutyl (meth)acrylate, n-pentyl (meth)acrylate, 2- methylbutyl (meth)acrylate, n-hexyl (meth)acrylate, cyclohexyl (meth)acrylate, 4-methyl-2-pentyl (meth)acrylate, 2-ethylhexyl (meth) acrylate, 2-methylhexyl (meth)acrylate, n-octyl (meth)acrylate, isooc
- the second monomer(s) can be characterized by the following parameters alone or in combination: (i) soluble in the liquid contained in the sol; (ii) bearing at least one or two or three curable moieties; (iii) bearing radiation curable moieties selected from vinyl, acryl or methacryl moieties; (iv) molecular weight (Mw): 70 to 5,000 g/mol; or 70 to 1,000 g/mol; or 100 to 500 g/mol, wherein the combination of parameters (ii) and (iii), or (ii) and (iv), or (ii), (iii) and (iv) is sometimes preferred.
- the curable component is typically present in the following amounts: at least 2 or at least 5 or at least 10 wt.%; at most 30 or at most 25 or at most 20 wt.%; from 2 to 30 or 5 to 25 or 10 to 20 wt.%; wt.% with respect to the weight of the sol.
- the sol described in the present text comprises one or more photo-initiator(s).
- the nature and structure of the photo-initiator is not particularly limited, either, unless the desired result cannot be achieved.
- the photo-initiator(s) can be characterized by at least one or more, sometimes all of the following parameters: soluble in the liquid contained in the sol; and/or showing radiation absorption within a range of 200 to 500 nm or 300 to 450 nm.
- the photo-initiator should be able to start or initiate the curing or hardening reaction of the radiation curable component(s) being present in the sol.
- photo-initiator(s) can be used: a) two-component system where a radical is generated through abstraction of a hydrogen atom form a donor compound; b) one component system where two radicals are generated by cleavage.
- photo-initiators according to type (a) typically contain a moiety selected from benzophenone, xanthone or quinone in combination with an aliphatic amine.
- photo-initiators according to type (b) typically contain a moiety selected form benzoin ether, acetophenone, benzoyl oxime or acyl phosphine.
- Exemplary UV initiators include 1 -hydroxy cyclohexyl benzophenone (previously available from Ciba Specialty Chemicals Corp as IRGACURE 184TM), 4-(2-hydroxyethoxy)phenyl-(2-hydroxy-2- propyl) ketone (previously available from Ciba Specialty Chemicals Corp as IRGACURE 2529TM, 2-hydroxy-2-methylpropiophenone (previously available from Ciba Specialty Chemicals Corp, as DAROCURE Di l lTM) and bis(2,4,6-trimethylbenzoyl)-phenylphosphineoxide (previously available from Ciba Specialty Chemicals Corp as IRGACURE 819TM).
- the photo-initiator is typically present in the following amounts: at least 0.01 or at least 0.1 or at least 0.2 wt.%; at most 3 or at most 2 or at most 1 wt.%; from 0.01 to 3 or 0.1 to 2 or 0.2 to 1 wt.%; wt.% with respect to the weight of the sol.
- the solvent can be characterized by the following features alone or in combination: a) boiling point: above 100°C or above 120°C or above 150°C; b) molecular weight: 25 to 300 g/mol; c) viscosity: 0.2 to 10 mPa*s (23°C); d) miscible with water; e) soluble in supercritical carbon dioxide or liquid carbon dioxide.
- a combination of the following features is sometimes preferred: a) and b), or a), b) and c), or a), b), c) and d).
- a liquid with a boiling point above 100°C or 150°C can be beneficial for reducing the evaporation of the liquid during the process.
- a liquid with a molecular weight and/or viscosity in the above range can be beneficial as it helps in adjusting the viscosity of the sol.
- the molecular weight size can also affect the diffusion constant and how easily the liquid can be removed.
- Using a mixture of different liquids can be beneficial as it allows adjusting the viscosity or post processing properties, e.g., removal of excess sol after casting.
- the liquid should also be easily removable during the further processing steps needed for the realization of a ceramic article.
- the liquid should not interfere with or negatively influence the polymerization of the curable components being present in the sol.
- liquid not bearing polymerizable moieties can be beneficial.
- the liquid typically bears one or more polar moieties, including ether, alcohol or carboxy moieties.
- the liquid is often a glycol or polyglycol, mono-ether glycol or mono-ether polyglycol, di-ether glycol or di-ether polyglycol, ether ester glycol or ether ester polyglycol, carbonate, amide, or sulfoxide (e.g., dimethyl sulfoxide).
- Suitable glycols or polyglycols, mono-ether glycols or mono-ether polyglycols, di-ether glycols or di-ether poly glycols, and ether ester glycols or ether ester polyglycols are often of the following Formula (I).
- each R 1 independently is hydrogen, alkyl, aryl, or acyl.
- Suitable alkyl groups often have 1 to 10 carbon atoms, 1 to 6 carbon atoms, or 1 to 4 carbon atoms.
- Suitable aryl groups often have 6 to 10 carbon atoms and are often phenyl or phenyl substituted with an alkyl group having 1 to 4 carbon atoms.
- Suitable acyl groups are often of formula -(CO)R a where R a is an alkyl having 1 to 10 carbon atoms, 1 to 6 carbon atoms, 1 to 4 carbon atoms, 2 carbon atoms, or 1 carbon atom.
- the acyl is often an acetyl group (-(COjCHs).
- each R 2 is typically ethylene or propylene.
- the variable n is at least 1 and can be in a range of 1 to 10, 1 to 6, 1 to 4, or 1 to 3.
- Glycols or polyglycols of Formula (I) have two R 1 groups equal to hydrogen.
- glycols include, but are not limited to, ethylene glycol, propylene glycol, diethylene glycol, dipropylene glycol, triethylene glycol, and tripropylene glycol.
- Mono-ether glycols or mono-ether polyglycols of Formula (I) have a first R 1 group equal to hydrogen and a second R 1 group equal to alkyl or aryl.
- mono-ether glycols or monoether polyglycols include, but are not limited to, ethylene glycol monohexyl ether, ethylene glycol monophenyl ether, propylene glycol monobutyl ether, diethylene glycol monomethyl ether, diethylene glycol monoethyl ether, diethylene glycol monopropyl ether, diethylene glycol monobutyl ether, diethylene glycol monohexyl ether, dipropylene glycol monomethyl ether, dipropylene glycol monoethyl ether, dipropylene glycol monopropyl ether, triethylene glycol monomethyl ether, triethylene glycol monoethyl ether, triethylene glycol monobutyl ether, tripropylene glycol monomethyl ether, and tripropylene glycol monobutyl
- Di-ether glycols or di-ether polyglycols of Formula (I) have two R 1 group equal to alkyl or aryl.
- Examples of di-ether glycols or di-ether polyglycols include, but are not limited to, ethylene glycol dipropyl ether, ethylene glycol dibutyl ether, dipropylene glycol dibutyl ether, diethylene glycol dimethyl ether, diethylene glycol diethyl ether, triethylene glycol dimethyl ether, tetraethylene glycol dimethyl ether, and pentaethylene glycol dimethyl ether.
- Ether ester glycols or ether ester polyglycols of Formula (I) have a first R 1 group equal to an alkyl or aryl and a second R 1 group equal to an acyl.
- ether ester glycols or ether ester polyglycols include, but are not limited to, ethylene glycol butyl ether acetate, diethylene glycol butyl ether acetate, and diethylene glycol ethyl ether acetate.
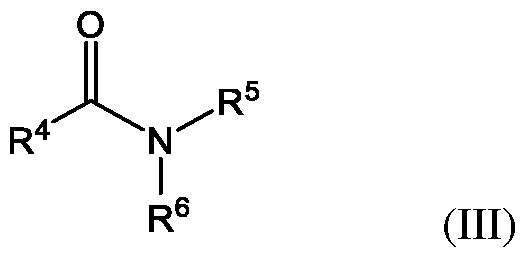
- R 3 is hydrogen or an alkyl such as an alkyl having 1 to 4 carbon atoms, 1 to 3 carbon atoms, or 1 carbon atom. Examples include ethylene carbonate and propylene carbonate. Yet other suitable organic solvents are amides of Formula (III).
- group R 4 is hydrogen, alkyl, or combines with R 5 to form a five-membered ring including the carbonyl attached to R 4 and the nitrogen atom attached to R 5 .
- Group R 5 is hydrogen, alkyl, or combines with R 4 to form a five-membered ring including the carbonyl attached to R 4 and the nitrogen atom attached to R 5 .
- Group R 6 is hydrogen or alkyl. Suitable alkyl groups for R 4 , R 5 , and R 6 have 1 to 6 carbon atoms, 1 to 4 carbon atoms, 1 to 3 carbon atoms, or 1 carbon atom.
- amide organic solvents of Formula (III) include, but are not limited to, formamide, N,N-dimethylformamide, N,N-dimethylacetamide, N,N-diethylacetamide, N-methyl-2-pyrrolidone, and N-ethyl-2-pyrrolidone.
- solvents which can be used include mono alcohols (e.g. C2 to Cs alcohols, including primary, secondary and tertiary alcohols), poly alcohols (e.g. ethylene glycol, propylene glycol, glycerine, diethylene glycol ethyl ether (CarbitolTM), l-methoxy-2-propanol, N-methyl pyrrolidone, acetonitrile, chlorobenzene, 1,4-dioxane, ethyl acetate, methyl ethyl ketone, tetrahydrofuran, toluene, xylene and mixtures thereof.
- mono alcohols e.g. C2 to Cs alcohols, including primary, secondary and tertiary alcohols
- poly alcohols e.g. ethylene glycol, propylene glycol, glycerine, diethylene glycol ethyl ether (CarbitolTM), l-methoxy-2-propan
- solvents are sometimes preferred: ethanol, 1 -methoxy-2-propanol, N-methyl pyrrolidone, diethylene glycol ethyl ether, and mixtures thereof.
- suitable solvents may also include low boiling alcohols (below 100°C; like methanol, ethanol, propanol) and mixtures thereof or preferably the same solvent(s) described above.
- the liquid is typically present in the following amounts: at least 25 or at least 30 or at least 35 wt.%; at most 70 or at most 65 or at most 60 wt.%; from 25 to 70, or 30 to 65, or 35 to 60 wt.%; wt.% with respect to the weight of the sol.
- Coloring components which can be used include in particular components comprising ions selected from Fe, Mn, Er, Pr, Tb, Cr, Co, Mo and/or Nd.
- these components are typically present as oxides in the sintered article.
- the coloring ions are typically present in the following amounts: 0, or at least 0.1, or at least 0.2 wt.%; at most 2, or at most 1, or at most 0.8 wt.%; from 0 to 2, or 0.1 to 1, or 0.2 to 0.8 wt.%; wt.% total coloring ions with respect to the weight of the sol.
- the sol described in the present text comprises one or more inhibitor(s).
- the nature and structure of the inhibitor(s) is not particularly limited, either, unless the desired result cannot be achieved.
- An inhibitor may extend the shelf life of the sol, help prevent undesired side reactions, and adjust the polymerization process of the radiation curable component(s) present in the sol.
- Adding one or more inhibitor(s) to the sol may further help to improving the accuracy or detail resolution of the surface of the ceramic article.
- adding inhibitor(s) to the printing sol described in the present text may help to enhance the resolution and accuracy of the SLA process by attenuating or avoiding unwanted scattering effects, as well as increase the shelf life of the sol.
- the inhibitor(s) should be soluble in the solvent contained in the sol. Inhibitors which can be used often comprise a phenol moiety.
- inhibitor(s) which can be used include: butylhydroxy toluol (Ionol), p- methoxyphenol (MOP), hydroquinone monomethylether (MEHQ), 2,6-di-tert-butyl-4-methyl- phenol (BHT), phenothiazine, 2,2,6,6-tetramethyl-piperidine-l-oxyl radical (TEMPO) and mixtures thereof.
- Ionol butylhydroxy toluol
- MOP p- methoxyphenol
- MEHQ hydroquinone monomethylether
- BHT 2,6-di-tert-butyl-4-methyl- phenol
- TEMPO 2,2,6,6-tetramethyl-piperidine-l-oxyl radical
- the inhibitor is typically present in the following amounts: 0, or at least 0.001, or at least 0.01 wt.%; at most 0.5, or at most 0.02, or at most 0.01 wt.%; from 0 to 0.5, or 0.001 to 0.05, or 0.01 to 0.02 wt.%; wt.% with respect to the weight of the sol.
- sols described in the present text can be obtained as follows:
- a starting sol containing nano-sized zirconia particles is provided.
- a precursor solution is typically prepared by combining a zirconium salt (e.g., acetate) solution and a solvent (e.g., water).
- a phase stabilizing agent e.g., yttrium acetate
- the resulting composition is pumped e.g., through a hydrothermal reactor. Suitable hydrothermal reactors are described e.g., in US 5,453,262 (Dawson et al.) and US 5,652,192 (Matson et al.).
- the content of tetragonal and/or cubic phase of the zirconia crystallites can be adjusted by varying the amount of phase stabilizing components added during the production method.
- the starting sol is typically concentrated.
- At least a portion of the aqueous-based medium is removed from the zirconia-based sol.
- the zirconia-based sol is subjected to dialysis or diafiltration.
- the content of the crystalline nano-sized zirconia particles in the concentrated starting sol is typically in a range of 25 to 65 wt.%.
- the zirconia-based sol can be subjected to a solvent exchange process.
- organic liquid having a higher boiling point than water can be added to the effluent.
- organic liquids that are suitable for use in a solvent exchange method include 1 -methoxylpropanol, N-methyl pyrrolidone or diethylene glycol ethyl ether.
- the water then can be removed by a method such as distillation, rotary evaporation, or oven drying.
- a zirconia-based sol comprises zirconia-based particles dispersed and/or suspended (i.e., dispersed, suspended, or a combination thereof) in an aqueous/organic matrix.
- the curable component(s) the photo-initiator(s), optionally organic dye(s), inhibitor(s), coloring component(s), and other additive(s), if desired.
- the preparation of the sol is typically conducted under safe light conditions to avoid an undesired early polymerization.
- Sol Si, sol S2 and optional sols SN can also be produced by mixing so-called master batches with different contents of stabilizing and/or coloring components.
- the chemical composition of curable sol Si, curable sol S2, and optionally curable sols SN can be adjusted, if desired, to match with information associated with the dental situation of a patient.
- sol Si sol S2 and optional sols SN-
- At least sol S2 contains coloring components, wherein sol S2 is typically less light-transmissive than sol Si.
- sol Si, sol S2 and optional sol SN differ from each other with respect to the following properties alone or in combination: (i) content of coloring components, (ii) content of crystal phase stabilizer components, (iii) content of crystalline zirconia particles, (iv) viscosity, wherein a combination of (i) and (ii), or (i) and (iii), or (ii) and (iii) is sometimes preferred.
- the difference in viscosity between the sols is typically greater than a ratio of 1.2 or 1.5 or 2.
- the process for producing the customized zirconia gel article further comprises a curing step, in particular a radiation-curing step.
- the radiation-curing step is typically conducted under the following conditions: wavelength: 300 to 500 nm, or 365 to 460 nm; duration: 1 s to 5 min; temperature: 10 to 30°C.
- the curable components contained in the sol are at least partially crosslinked leading to the formation of a gel article.
- the process described in the present text may also comprise a post-curing step.
- the zirconia gel article can be post-cured by applying radiation or heat.
- Such a step may help to improve the stability of the zirconia gel article by further increasing the degree of polymerization.
- the post-curing step can be characterized by the following features alone or in combination: (i) applying radiation with wavelength of 200 to 500 nm or 350 to 450 nm; (ii) applying a heating step with a temperature below the temperature at which drying will occur or which is used for de-bindering or calcining; e.g., 30 to 110°C or 40 to 80°C.
- a post-curing step is typically conducted after the step of radiation-curing the curable zirconia sols.
- the process may comprise further steps.
- These steps may yield intermediate products such as an aerogel article and/or a pre-sintered zirconia article.
- One additional step may relate to the transforming of the zirconia gel article into a zirconia aerogel article.
- the transforming step can be achieved by a process comprising an extraction step.
- a zirconia aerogel article can be formed by removing liquid from the zirconia gel article without excessive shrinkage (e.g., not greater than about 30 or 40 vol.%).
- Water if present, can be removed from the zirconia gel article e.g., via alcohol exchange to provide an at least partially de-watered gel.
- the zirconia gel article is then converted or transformed to a zirconia aerogel article by removing the alcohol, if present, from the partially de-watered zirconia gel article via e.g., super critical extraction.
- the zirconia aerogel article can be characterized by the following features alone or in combination:
- (a) comprising crystalline zirconia particles having an average primary particle size in a range of 2 nm to 50 nm or 2 nm to 30 nm or 2 to 20 nm;
- a combination of the features (a) and (b), or (a), (b) and (c), or (b), (d) and € can be preferred.
- the extraction step can be characterized by the following features alone or in combination:
- a combination of features (a), (b) and (d) is sometimes preferred.
- the aerogels contain some residual organic liquid.
- the residual liquid can be up to 6 wt.% based on the total weight of the aerogel article.
- the aerogel article can contain up to 5 wt.%, up to 4 wt.%, up to 3 wt.%, up to 2 wt.%, or up to 1 wt.% organic liquid.
- the removal of organic liquid results in the formation of pores within the dried structure.
- the pores are sufficiently large to allow gases from the decomposition products of the polymeric material to escape without cracking the structure when the dried structure is further heated to burnout the organic material and to form a sintered article.
- the process further comprises one or more heat -treatment steps.
- the heat-treatment may yield a calcined dental zirconia article, or a pre-sintered dental zirconia article, or a sintered dental zirconia article.
- the heat treatment can be conducted in one or more steps and at different locations.
- a heating step is conducted to remove organic residues which are still present in the zirconia article before final sintering. Removing organic residues before sintering reduces the risks of cracks during sintering.
- the heating step is typically conducted at a temperature below 800°C or below 700°C or below 600°C.
- a typical temperature range is from 400 to 800°C or from 500 to 700°C.
- Such a heating step is typically conducted for a time needed to combust the organic components in the zirconia article.
- a typical time frame is from 5 to 100 h or from 10 to 50 h.
- pre-sintered dental zirconia article For obtaining a pre-sintered dental zirconia article the following conditions are typically applied alone or in combination: (i) temperature: 900 to l,100°C or 950 to l,090°C or 975 to l,080°C; (ii) atmosphere: air or inert gas (e.g., nitrogen, argon); (iii) dwell time: 0 to 24h, or 0.1 to 5h; (iv) duration: until a density of 40 to 60% of the density of the sintered material has been reached.
- atmosphere air or inert gas (e.g., nitrogen, argon)
- dwell time 0 to 24h, or 0.1 to 5h
- duration until a density of 40 to 60% of the density of the sintered material has been reached.
- a combination of conditions (i) and (ii); or (i) and (iii); or (i) and (iv); or (i) and (ii) and (iv) is sometimes preferred.
- the dwell time (that is the time during which the aerogel article is kept at that temperature) is helpful as well to tune strength and/or hardness to the specific needs of the chosen machining technology. If the dwell time is too long, the dental article may become too hard to be machined under reasonable conditions, if desired.
- a sintered dental zirconia article For obtaining a sintered dental zirconia article the following conditions are typically applied: (i) temperature: 1,100 to l,350°C or 1,200 to l,300°C; (ii) atmosphere: air or inert gas (e.g., nitrogen, argon); (iii) duration: until the porosity of the zirconia article is less than 0.1 or less than 0.05 vol.%; (iv) dwell time: 1 to 24 h or 2 to 12 h; (v) pressure: ambient pressure.
- temperature 1,100 to l,350°C or 1,200 to l,300°C
- atmosphere air or inert gas (e.g., nitrogen, argon)
- duration until the porosity of the zirconia article is less than 0.1 or less than 0.05 vol.%
- dwell time 1 to 24 h or 2 to 12 h
- pressure ambient pressure.
- the porous dental ceramic article is sintered to its final shape, thereby undergoing changes with regard to dimension, density, hardness, bending strength and/or grain size.
- the heating temperature and dwell time (that is, the time period during which a particular temperature is kept) are typically correlated. A higher temperature typically requires only a short dwell time.
- the heat-treatment steps can be characterized as regular firing conditions (e.g., heating rate of 10 to 30°C/min up to at least l,200°C) or speed-firing conditions (e.g., heating rate of 60 to 350°C/min up to at least l,200°C).
- regular firing conditions e.g., heating rate of 10 to 30°C/min up to at least l,200°C
- speed-firing conditions e.g., heating rate of 60 to 350°C/min up to at least l,200°C.
- the temperature needed for obtaining a fully-sintered article is usually not more than 1 ,350°C, sometimes not more than 1 ,300°C.
- the dental restoration described in the present text can be produced more economically as less heat is needed for sintering.
- an extraction step can be conducted in addition.
- Such an extraction step may help to remove or exchange ions from the article, such as sulfate ions which might be present at a certain stage of the process.
- a pre-sintered zirconia article can be soaked in a basic solution such as an aqueous solution of ammonium hydroxide.
- the pre-sintered zirconia article is removed from the solution and washed thoroughly with water.
- the article can be soaked in water for any desired period of time such as at least 30 min, at least 1 h, at least 2 h, or at least 4 h.
- the soaking in water can be repeated several times, if desired, by replacing the water with fresh water.
- the article After soaking, the article is typically dried in an oven to remove the water.
- the article can be dried by heating in an oven set at a temperature equal to at least 80°C, at least 90°C, or at least 100°C.
- the temperature can be in a range of 80°C to 150°C, 90°C to 150°C, or 90°C to 125 °C for at least 30 min, at least 60 min, or at least 120 min.
- An ion-exchange step can typically be characterized by the following features alone or in combination: duration: 5 to 24 hrs; medium: aqueous ammonium hydroxide liquid (e.g., concentration IN); and/or temperature: 20 to 25°C.
- the invention is also directed to a zirconia ceramic dental restoration, in particular to a monolithic zirconia ceramic dental restoration, which can be obtained or is obtainable from the customized zirconia gel article.
- the zirconia ceramic dental restoration has typically the shape of a dental crown, bridge, inlay, onlay, or veneer.
- the dental restoration has a shape which allows its use by the practitioner without the need for further adjustments or modifications. In particular, there is no need for an additional glazing and/or milling step.
- a front tooth has an outer or labial surface.
- the outer surface of a front tooth corresponds to the outer surface of a veneer.
- a dental restoration like a veneer has also an inner surface which corresponds to the outer surface of a prepared tooth in labial direction.
- the zirconia ceramic dental restoration described in the present text comprises an outer surface area and an inner surface area.
- the outer surface of the outer surface area, or the inner surface of the inner surface area, or both surfaces may comprise a section having a shape which is based on information associated with the dental situation of a patient.
- Both, the outer surface area and the inner surface area comprise ceramic components and stabilizing components.
- At least one region of the inner surface area comprises coloring components or a different concentration of stabilizing components.
- At least one region of the outer surface area may comprise coloring components or a different concentration of stabilizing components, as well.
- the material of the inner surface area of the zirconia ceramic dental restoration is typically less light-transmissive than the outer surface area in at least one region of the inner surface area. It was found that this arrangement can more closely mimic the natural tooth layers of a relatively opaque dentin core covered by a relatively translucent enamel layer.
- the material of the inner surface area of the zirconia ceramic dental restoration is less light-transmissive than the material of the outer surface area in at most three individual regions of the inner surface area.
- a difference in light-transmission can be determined or analysed with a microscope on a cross-section of the respective area of the zirconia ceramic dental restoration. If desired, the light transmittance can also be assessed by comparing the respective contrast ratio reflectance (CR-R) as further outlined in the example section.
- CR-R contrast ratio reflectance
- Contrast ratio is related to light transmission. Contrast ration is the ratio of luminence observed when a test article of a prescribed thickness is measured over a black background vs. the same article measured over a white background. An article with high light transmittance will change in luminance with a different background, an article with low transmittance will change less.
- the inner surface area and the outer surface area are contacting each other at an interface area or transition area, which is typically a non-planar.
- Such a transition zone can be advantageous as it allows for a smoother transition of the two material compositions of the inner surface area and the outer surface area.
- the range and material composition of the interface can be modified and adjusted.
- the content of coloring components in the interface area or transition zone is typically lower than the content of the coloring components in the inner surface area.
- the difference in content of coloring components can be determined on a sample cut from the zirconia ceramic dental restoration to be analyzed by XRF technology.
- the inner surface area and/or the outer surface area typically have a thickness in the range of 0.02 to 3 mm or 0.05 to 2 mm.
- the material of the zirconia ceramic dental restoration comprises ceramic components, stabilizing components and coloring components.
- the main ceramic component is zirconia. Besides zirconia, hafnia is typically present as well, but in smaller amounts.
- Alumina is typically not present and/or has not been wilfully added.
- Crystal phase stabilizing components which can be present include oxides of Ce, Mg, Ca, La and Y, wherein the use of Y is sometimes preferred.
- Crystal phase stabilizing components calculated as oxides are typically present in the following amounts: at least 1, 2 or 3 mol%; at most 7, 6 or 5 mol%; from 1 to 7 mol%, or 2 to 6, or 3 to 5 mol%; mol% with respect to the zirconia ceramic dental restoration.
- Coloring components which are present in the zirconia ceramic dental restoration include oxides selected from Fe, Mn, Er, Pr, Tb, Cr, Co, Mo, Nb and mixtures thereof, wherein the combination of Fe, Mn, Er and Pr is sometime preferred.
- the material of the zirconia ceramic dental restoration may comprise or essentially consist of or consist of the following components:
- Coloring components selected from oxides of Fe, Mn, Er, Pr, Tb, Cr, Co, Mo, Nb or mixtures thereof in an amount of 0.001 to 1 mol%, mol% with respect to the zirconia ceramic dental restoration.
- the material of the zirconia ceramic dental restoration may comprise or essentially consist of or consist of the following components:
- HfCF in an amount of 0 to 2 mol%
- Y2O3 in an amount of 3 to 6 mol%, coloring components selected from oxides of Fe, Mn, Er, Pr, Tb, Cr, Co, Mo, Nb or mixtures thereof in an amount of 0.01 to 0.8 mol%, mol% with respect to the zirconia ceramic dental restoration.
- the material of the zirconia ceramic dental restoration typically has a tetragonal crystal phase content of at least 40 vol.% or at least 45 vol.% or at least 50 vol.%.
- a high tetragonal crystal phase content typically goes along with a high strength of the material.
- the nano-sized zirconia materials described in the present text may have a rather high tetragonal phase content even if the material has a rather high content of stabilizing components (e.g., above 4, 5 or 6 mol% with respect to the zirconia ceramic dental restoration).
- the material of the zirconia ceramic dental restoration has an average grain size of 80 to 300 nm for a material with a content of stabilizing components in a range of 3 to 5.0 mol%, or 150 to 550 nm for a material with a content of stabilizing components in the range of 5.1 to 8 mol%, mol% with respect to the zirconia ceramic dental restoration.
- the material of the zirconia ceramic dental restoration is to be regarded as a very fine grained material as it results from the processing of compositions comprising nano-sized components.
- the grain size of the zirconia ceramic body is significantly smaller than the grain size of a zirconia material with the same content of stabilizer but made by compacting a respective powder and sintering the compact at higher temperatures.
- a zirconia material with such an average grain size has advantageous optical properties, in particular as regards transmission.
- the zirconia ceramic dental restoration can in addition be characterized by the following features alone or in combination: a) porosity: less than 0.1 vol.%; b) thickness between outer and inner surface: 0.5 to 0.02 mm at at least one section of the monolithic zirconia ceramic dental restoration; c) translucency: at least 40 % for at least one region of the monolithic zirconia ceramic dental restoration, if determined on a sample having a thickness of 1 mm with a light having a wavelength in the range of 360 to 780 nm; d) opalescence: at least 9 determined on a sample having a thickness of 1 mm; e) contrast ratio differing from each other by at least 5% in two sections of the monolithic zirconia ceramic dental restoration normalized to a thickness of 1 mm and spaced apart from each other by 3 mm.
- a material with a porosity of less than 0.1 vol.% or less than 0.05 vol.% is considered as dense.
- the zirconia ceramic dental restoration and related process described in the present text has or allows the manufacturing of ceramic articles with very thin sections. It was found that a dental restoration with such a thin and filigree structure cannot be produced by other means, such as milling or 3d-printing.
- a section is to be understood as a volume that is distinguishable and that typically corresponds to an anatomical feature (i.e. dental situation) on a tooth.
- Mamelons are an example.
- Three mamelon features typically span the width of a tooth, so they have visible features that range in size from tens to hundreds of microns. Sections are visually distinguishable when viewed by a dentally- trained eye without supplemental magnification.
- a section of a zirconia ceramic dental restoration is typically to be understood to have a size in one, two or three dimensions of at least 100 or 150 or 200 pm.
- Zirconia ceramic dental restoration with such thin section cannot be efficiently produced by milling.
- the zirconia ceramic dental restoration is highly translucent at at least one region of the dental restoration, e.g., the occlusal or incisal region.
- the opalescence of the material of the dental restoration is in a region which basically corresponds to the opalescence of natural teeth.
- the manufacturing process described in the present text allows the production of customized zirconia ceramic dental restorations where the contrast ratio of different sections of the restoration may differ already over a small distance.
- the process described in the present text does typically not require the following steps alone or in combination: a zirconia powder compacting step; a milling step; a step of applying a coloring liquid; and/or a step of applying a glazing layer.
- a process for producing a dental restoration comprising, essentially consisting of, or consisting of the following steps:
- a post-curing step e.g., by heating the zirconia gel article to a temperature in the range of 35 to 80 °C or by lightcuring, in particular for the purpose of increasing the stability of the zirconia gel article,
- sol Si, sol S2 and optional sols SN each comprising crystalline zirconia particles, a crystal phase stabilizer component, a radiation-curable organic component, a photo-initiator, a liquid, and wherein at least one sol contains coloring components, wherein the crystal phase stabilizer component is contained in the crystalline zirconia particles and the coloring component, if present, can be contained in the crystalline zirconia particles or be present as a separate component in the sol, wherein the viscosity of each sol is in a range of 50 to 3,000 mPa*s at 23°C; and wherein sol Si, sol S2 and optional sols SN differ from each other with respect to the following properties alone or in combination: content of coloring components, content of crystal phase stabilizing components, content of crystalline zirconia particles.
- a dental restoration in particular obtainable or obtained by the process described in the present text, the dental ceramic zirconia article being characterized by the following properties: thickness between outer and inner surface in at least one section: 0.5 to 0.02 mm, difference in optical properties of at least two sections regarding colour and/or translucency: DE > 2 and/or %CR > 2, determined on individually colored 1 mm thick samples, opalescence: at least 10,
- HfCE content 0 to 2 mol%
- AI2O3 content 0 to 0.1 mol%, mol.% with respect to the dental ceramic zirconia article.
- a customized zirconia gel article having the shape of a dental restoration comprising at least 3 areas, an inner surface area comprising gel Gl, an outer surface area comprising gel G2, and a transition area comprising transition gel G1/2, the material compositions of the gels each comprising crystalline zirconia particles, a crystal phase stabilizer component, a cured organic component, optionally a photo-initiator, a liquid, and wherein at least one of the gels comprise a color component, wherein the crystal phase stabilizer component is contained in the crystalline zirconia particles and the coloring component, if present, can be contained in the crystalline zirconia particles or be present as a separate component in the gel, wherein gel Gi is different from gel G2, wherein the transition area has a color gradient, and wherein the transition gel comprises a mixture of the material compositions of gels Gi and G2.
- Dried zirconia samples were ground by hand using an agate mortar and pestle. A liberal amount of the sample was applied by spatula to a glass microscope slide on which a section of double-sided adhesive tape had been adhered. The sample was pressed into the adhesive on the tape by forcing the sample against the adhesive with the spatula blade. Excess sample was removed by scraping the sample area with the edge of the spatula blade, leaving a thin layer of particles adhered to the adhesive. Loosely adhered materials remaining after the scraping were removed by forcefully tapping the microscope slide against a hard surface. In a similar manner, corundum (Linde 1.0 pm alumina polishing powder, Lot Number C062, Union Carbide, Indianapolis, IN) was prepared and used to calibrate the X-ray diffractometer for instrumental broadening.
- X-ray diffraction scans were obtained using a Philips vertical diffractometer having a reflection geometry, copper K a radiation, and a proportional detector registry of the scattered radiation.
- the diffractometer was fitted with variable incident beam slits, fixed diffracted beam slits, and a graphite diffracted beam monochromator.
- the survey scan was recorded from 25 to 55 degrees two theta (2q) using a step size of 0.04 degrees and a dwell time of 8 seconds.
- X-ray generator settings of 45 kV and 35 mA were used. Data for the corundum standard was collected on three separate areas of several individual corundum mounts. Likewise, data was collected on three separate areas of the thin layer sample mount.
- the observed diffraction peaks were identified by comparison to reference diffraction patterns contained within the International Center for Diffraction Data (ICDD) powder diffraction database (sets 1-47, ICDD, Newton Square, PA, USA).
- ICDD International Center for Diffraction Data
- the diffraction peaks for the samples were attributed to either cubic/tetragonal (C/T) or monoclinic (M) forms of zirconia.
- C/T cubic/tetragonal
- M monoclinic
- Peak widths for the observed diffraction maxima due to corundum were measured by profile fitting. The relationship between mean corundum peak widths and corundum peak position (2q) was determined by fitting a polynomial to these data to produce a continuous function used to evaluate the instrumental breadth at any peak position within the corundum testing range. Peak widths for the observed diffraction maxima due to zirconia were measured by profile fitting the observed diffraction peaks. The following peak widths were evaluated depending on the zirconia phase found to be present:
- C/T Cubic/Tetragonal (C/T): (1 1 1) Monoclinic (M): (-1 1 1), and (l 1 1)
- Crystallite Size (D) Kl/b (cos q)
- K is the form factor (here 0.9)
- 1 is the wavelength (1.540598 A)
- b is the calculated peak width after correction for instrumental broadening (in radians)
- q equals half the peak position (scattering angle)
- b is equal to [calculated peak FWHM - instrumental breadth] (converted to radians) where FWHM is full width at half maximum.
- the cubic/tetragonal (C/T) mean crystallite size was measured as the average of three measurements using (1 1 1) peak. That is,
- PCS Photon Correlation Spectroscopy
- Particle size measurements were made using a light scattering particle sizer equipped with a red laser having a 633 nm wavelength of light (obtained under the trade designation “ZETA SIZER - Nano Series, Model ZEN3600” from Malvern Instruments Inc., Westborough, MA). Each sample was analyzed in a one centimeter square polystyrene sample cuvette. The sample cuvette was filled with about 1 gram of deionized water, and then a few drops (about 0.1 gram) of the zirconia-based sol were added. The composition (e.g., sample) within each sample cuvette was mixed by drawing the composition into a clean pipette and discharging the composition back into the sample cuvette several times.
- the sample cuvette was then placed in the instrument and equilibrated at 25°C.
- the instrument parameters were set as follows: dispersant refractive index 1.330, dispersant viscosity 0.8872 MPa-second, material refractive index 2.10, and material absorption value 0.10 units.
- the automatic size-measurement procedure was then run. The instrument automatically adjusted the laser-beam position and attenuator setting to obtain the best measurement of particle size.
- the light scattering particle sizer illuminated the sample with a laser and analyzed the intensity fluctuations of the light scattered from the particles at an angle of 173 degrees.
- the method of Photon Correlation Spectroscopy (PCS) was used by the instrument to calculate the particle size.
- PCS uses the fluctuating light intensity to measure Brownian motion of the particles in the liquid.
- the particle size is then calculated to be the diameter of sphere that moves at the measured speed.
- the intensity of the light scattered by the particle is proportional to the sixth power of the particle diameter.
- the Z-average size or cumulant mean is a mean calculated from the intensity distribution and the calculation is based on assumptions that the particles are mono-modal, mono-disperse, and spherical.
- the mean of the Intensity Distribution is calculated based on the assumption that the particles are spherical. Both the Z-average size and the Intensity Distribution mean are more sensitive to larger particles than smaller ones.
- the Volume Distribution gives the percentage of the total volume of particles corresponding to particles in a given size range.
- the volume-average size is the size of a particle that corresponds to the mean of the Volume Distribution. Since the volume of a particle is proportional to the third power of the diameter, this distribution is less sensitive to larger particles than the Z-average size. Thus, the volume-average will typically be a smaller value than the Z-average size.
- the oxide content was measured via thermal gravimetric analysis (obtained under the trade designation “TGA Q500” from TA Instruments, New Castle, DE, USA). The sample (about 50 mg) was loaded into the TGA and the temperature was taken to 900°C in air. The oxide content of the sample was equal to the residual weight after heating to 900°C.
- the density of the sintered material was measured by the Archimedes technique. The measurements were made on a precision balance (identified as “XSE204” from Mettler-Toledo, LLC, Columbus, OH, USA) using a density determination kit (identified as “Density Determination Kit for Excellence XP/XS Analytical Balances” from Mettler-Toledo, LLC, Columbus, OH, USA). The sample was first weighed in air (A), then immersed in water and weighed (B). The water was distilled and deionized. Three drops of a wetting agent (obtained under trade designation “PERVITRO 75%” from Mettler-Toledo, LLC, Columbus, OH, USA) was added to 250 ml of water.
- a wetting agent obtained under trade designation “PERVITRO 75%” from Mettler-Toledo, LLC, Columbus, OH, USA
- CR-R value is the opacity using the contrast ratio method
- _E is the color difference.
- Recording was done using a Photospectrometer Color i7 (X-Rite Corp.) with an optical configuration of d/8° using D65 calibrated illumination, where a spectral range between 360 and 750nm with lOnm wavelength interval was recorded. For this measurement reflectance mode with 10mm aperture was used. Color evaluation was configured with 10° observer, exclusion of specular and inclusion of UV irradiation. For these measurements, fully sintered ceramic test pieces of approximately 1 mm thickness were used. The samples were measured using both a light backing and a dark backing. L*, a* and b* values are automatically calculated from the spectra by the X- Rite software.
- CR-R is also done automatically by the software, using a formula at the ratio of dark backing to light backing measurement. CR-R is expressed as a percentage. The higher the level of CR-R, the more opaque the material is, and the lower the level of CR-R, the more translucent the material is.
- viscosity can be measured using a capillary viscosity method. Fluid is placed in a cylinder of a diameter D of 10 mm, a piston is moved at a constant speed v of 2.9 mm/min in order to press the sol through a die of a length L of 38.1 mm and a diameter d of 0.58 mm. The force F is measured to move a piston at constant speed, calibrated to piston friction. A standard equipment, Zwick, Ulm, was used to perform these measurements.
- the pH value of can be determined as follows: 1.0 g of a component is dispersed in 10 ml de-ionized water and stirred for about 5 min. A calibrated pH electrode is dipped into the suspension and the pH value is determined during stirring.
- the elemental composition can be determined by X-ray fluorescence spectrometry (XRF), e.g., with the ZSX Primus II from Rigaku, Japan. This method is especially suited for the analysis of solids, e.g., zirconia ceramics or glass materials.
- XRF X-ray fluorescence spectrometry
- the OP value can be determined with a sphere benchtop spectral photometer Color i7800 (x-rite, Michigan USA), applying the formula described above using samples of 1.0 mm in height and 15 mm in diameter.
- Average Grain Size can be determined according to ASTM El 12 using the planimetric intercept procedure to determine f .
- the average grain size is the average value of the grain size of 10 lines.
- the average grain size of a line is the line length in a SEM picture divided by the number of grain boundary intercept multiplied by the shape factor 1.56.
- the porosity of a zirconia article can be determined from the water uptake after soaking the article in water.
- the water uptake is the weight of the soaked article divided by the weight of the dry article.
- the volume of the porosity is calculated from the water uptake and the density of the water at the measure temperature.
- Zirconia-based sols Sol-I(a)-Sol-III(a) were prepared as described in WO 2016/191534 Al (Examples Section - Processing: Preparation of Sol-Si) except that the feed composition was varied. Yttrium acetate, iron acetate and erbium acetate were added to the precursor solution in appropriate amounts.
- the target compositions of zirconia in Sol-I(a)-Sol-III(a) are listed in Table 1 , below.
- Sol-I(a)-Sol-II(a) were further processed to increase their concentration and/or remove acetic acid (AcOH).
- a combination of one or more of ultrafiltration, diafiltration and distillation were used.
- the diafiltration and ultrafiltration were performed using a dialysis membrane (Membra-CelTM MD77 14x100 CLR). Distillation was performed using rotary evaporation.
- Diethylene glycol monoethyl ether-based sols Sol-I(b)-Sol-III(b), were prepared from Sol-I(a)- Sol-III(a) by adding 2-[2-(2-methoxyethoxy)ethoxy]acetic acid (MEEAA) (3.3 wt.% with respect to the grams of oxide in the sol) as well as the appropriate amount of diethylene glycol monoethyl ether (adjusted to the intended final oxide concentration in the sol of 53 wt.%) and concentrating each sol via rotary evaporation. 282 g of Sol 1(b), 135 g of Sol 11(b) and 70 g of Sol 111(b) were made.
- MEEAA 2-[2-(2-methoxyethoxy)ethoxy]acetic acid
- Casting Sols CSol2 was obtained from Sol 11(b) and Casting Sol CSol3 was obtained from Sol 111(b).
- a body for an outer tooth shape was made from scan data (.stl) of an anterior tooth by CAD/CAM milling.
- the size of the mold needed to be scaled, depending on the shrinkage factor of the complete procedure.
- the shrinkage factor was determined experimentally by measuring the size of a casted body (e.g., 2.4 cm) and the final sintered body (e.g., 1.3 cm).
- the shrinkage factor was also calculated from the solid contents of the casting sols.
- a mold was formed by a vacuum thermoforming process using a BIOSTARTM VII device (Scheu, Germany) and COPYPLAST® mold material. Caution was taken to prevent light exposure by performing procedures in a UV-filtered room (yellow lights), or in low-light conditions when UV-filtering was not available.
- a second mould can be placed on top of the casted sol to provide a second, customized outer shape.
- the shape of the mould may be based on information associated with the dental situation of a patient.
- Curing was done by applying UV light (Osram Dulux S Blue 9W/71, Germany) for 15 s to 3 min. After curing, the gel sample was immediately removed from the mold and submerged in diethylene glycol monoethyl ether.
- the casted gel body was dried via supercritical fluid extraction, e.g., as described in the Method for Supercritical Extraction of Gels in the examples section of WO 2016/191534 Al (Mayr et al).
- the dried gel body was placed on a bed of zirconia beads in an alumina crucible.
- the crucible was covered with alumina plates and then fired in air according to the following schedule:
- the pre-sintered body was placed in a glass jar containing EON NH4OH at a depth of about 2.5 cm and soaked for at least 16 hours.
- the NH4OH was then poured off and the jar was filled with distilled water.
- the body was soaked in distilled water for 1 h.
- the water was then replaced with fresh distilled water. This step was repeated until the pH of the soak water was equal to that of fresh distilled water.
- the body was then dried at 90- 125 °C for a minimum of 15 min.
- the pre-sintered, ion-exchanged body was placed in a furnace and sintered in air according to the following schedule:
- Fig. 2 (1) to (4) shows a mold with a customized concave inner surface. Within the mold there is CSol 1 at the bottom (1) and small volumes of Csol 2 are applied on different areas on top of Csoll (2), (3). Shown is also the transition area formed by diffusion and or mixing of Csol 1 and Csol 2 (4).
- Example 2 exhibit a prototype veneer casted with about 0.31 ml of Cast sol Csoll first, on the right incisal side of the mold, about 0.22 ml Cast sol Csol3 at the left incisal side of the mold and about 0.7 ml of Csol2 at the body side of the mold, resulting in a color difference between the three zones.
- the sols mixed inherently due to self-diffusion.
- a light curing was done (about 2 min).
- the body had dimensions of 13 mm max length, 9.5 mm width and 2 mm max thickness and a weight of 0.66 g.
- XRD 77% tetragonal and 23% cubic crystallite phases were determined.
- SEM grain size was determined to 300 nm of the region Csol3.
- a picture of this prototype veneer is shown in Fig. 3.
- Fig. 4 shows a mold with a customized concave inner surface. Within the mold there is Csol 1 at the bottom (1). Different volumes of Csol 2 (2) and Csol 3 (3) are placed on top of or in contact with Csoll. Shown are also transition areas formed by diffusion and or mixing of Csol 1 with Csol 2 and Csol 3, respectively (4).
- Fig. 6 shows a mold with a customized concave inner surface. Within the mold there is Csol 1 at the bottom (1) and Csol 2 is placed on top of Csoll (2). A second mould with a convex outer surface is placed on top of Csol 2 (3) resulting in a mixing of the two sols in the transition area. After curing (4) a customized gel article having the shape of a dental restoration is obtained (5).
- CE1 is a veneer milled from a 3M LavaTM Esthetic Fluorescent Full-Contour Zirconia Disc (3M Oral Care) and finalized according to the instruction of use provided by the manufacturer. For this kind of material, the vendor reports an average grain size of approximately 1 pm. The veneer is not further glazed.
- the material of the disc has a build-in shade gradient, compared to the inventive examples above the appearance of the milled and sintered veneer of CE1 appeared rather homogeneous with respect to its color.
- CE2 is a veneer which was 3D-printed from a zirconia nanosol essentially corresponding to Sol- Ila), that is a sol without coloring components. The appearance of the veneer is homogeneous with respect to its color. As 3d-printer an Asiga Max 43 UV device was used. A picture of this sample is shown in Fig. 8.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Engineering & Computer Science (AREA)
- Ceramic Engineering (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Epidemiology (AREA)
- Plastic & Reconstructive Surgery (AREA)
- Manufacturing & Machinery (AREA)
- Dentistry (AREA)
- Inorganic Chemistry (AREA)
- Organic Chemistry (AREA)
- Structural Engineering (AREA)
- Materials Engineering (AREA)
- Dispersion Chemistry (AREA)
- Physics & Mathematics (AREA)
- Thermal Sciences (AREA)
- Dental Preparations (AREA)
Abstract
The invention relates to a customized zirconia gel article having the shape of a dental restoration, the customized zirconia gel article comprising a transition area comprising a transition gel, the transition area having a color gradient, the material composition of the transition gel comprising crystalline zirconia particles, a crystal phase stabilizer component, a cured organic component, optionally a photo-initiator, a liquid, and a coloring component, wherein the crystal phase stabilizer component is contained in the crystalline zirconia particles and the optional coloring component can be contained in the crystalline zirconia particles or be present as a separate component in the gel. Further, the invention relates to a process for producing such a customized zirconia gel article.
Description
CUSTOMIZED ZIRCONIA GEL ARTICLE, ZIRCONIA DENTAL RESTORATION
AND PROCESS OF PRODUCTION
Field of Invention
The invention relates to a customized zirconia gel article having the shape of a dental restoration and a dental restoration, in particular a monolithic zirconia ceramic dental restoration obtained therefrom, and a process of producing a zirconia ceramic dental restoration.
The zirconia ceramic dental restoration is highly aesthetic and has adequate physical-mechanical properties. The zirconia ceramic dental restoration can be produced without conducting a powdercompacting step or a milling step. The zirconia dental restoration may also feature tailored color and translucency variations that allow it to mimic the aesthetics of natural teeth.
Various methods for producing dental mill blanks and dental restorations from mill blanks or by other processes are meanwhile described in the literature.
US 9,039,947 B2 (Jahns et al.) describes a method for making a layered dental appliance, the method comprising: providing a mold comprising a negative of an outer shape of the layered dental appliance; positioning a slurry in the mold, forming a first layer of the layered dental appliance, the slurry comprising: (i) at least one of a glass powder and a glass ceramic powder, present in an amount of 60 wt.% to 85 wt.%, (ii) a liquid, and (iii) an inorganic binder precursor; providing a solid structure comprising a positive shape of a second layer of the layered dental appliance, wherein the solid structure includes a stamp; pressing the solid structure into the slurry in the mold; and removing the solid structure from the slurry to form a cavity comprising a negative of the second layer in the slurry.
US 10,028,809 B2 (Jahns et al.) describes a porous dental milling block comprising at least two geometrically defined Material Sections A and B, Material Section A comprising a tetragonal zirconia crystal phase in an amount A-T in wt.% and a cubic zirconia crystal phase in an amount A- C in wt.%, Material Section B comprising a tetragonal zirconia crystal phase in an amount B-T in wt.% and a cubic zirconia crystal phase in an amount B-C in wt.%, wherein (amount of tetragonal phase A-T in wt.%) / (amount of cubic phase content A-C in wt.%) > 1 and (amount of tetragonal phase content B-T in wt.%) / (amount of cubic phase content B-C in wt.%) < 1, the material of Material Sections A and B showing a N2 adsorption and/or desorption behavior of isotherm IV according to IUPAC classification.
US 2022/0380260 Al (Shah et al.) relates to an additive manufacturing method of making a ceramic article, the method comprising: a) obtaining a photopolymerizable sol comprising a plurality of ceramic particles distributed in the photopolymerizable sol, wherein the ceramic particles have an average particle size diameter of 1 nanometer (nm) to 100 nm; b) selectively
polymerizing the photopolymerizable sol using actinic radiation and continuous movement of a build substrate through the photopolymerizable sol to form a gelled article; c) extracting solvent from the gelled article to form an aerogel article or a xerogel article; d) heat treating the aerogel article or the xerogel article to form a porous ceramic article; e) sintering the porous ceramic article to form a sintered ceramic article; wherein the sintered ceramic article exhibits a density of 98% or greater with respect to a theoretical density of the ceramic material and wherein the sintered ceramic article exhibits an opacity of 80% or less.
US 10,759,707 B2 (Mayr et al.) describes a process for producing a ceramic article, the process comprising: providing a printing sol, wherein the printing sol comprising solvent, nano-sized particles, radiation curable monomer(s) and photoinitiator, and wherein the printing sol have a viscosity of less than 500 mPa*s at 23 °C.; processing the printing sol as construction material in an additive manufacturing process to obtain a 3-dimensional article being in a gel state, the 3- dimensional article having a Volume A, transferring the 3-dimensional article being in a gel state to a 3-dimensional article being in a dry state, namely an aerogel or xerogel; and applying a heat treatment step to obtain a sintered 3 ceramic article, the ceramic article having a Volume F; wherein Volume A of the 3-dimensional article in a gel state being more than 500% of Volume F of the ceramic article in its sintered state.
US 10,532,008 B2 (Balasubramanian et al.) describes a shaded zirconia ceramic material for use in dental applications comprising an yttria-stabilized zirconia material comprising from 4.7 mol % to 5.1 mol % yttria, a coloring agent comprising at least one metal selected from: a) Tb, measured as Tb4O?, in an amount from 0.005 wt.% to 0.025 wt.%; b) Cr, measured as C O?. in an amount from 0.0002 wt.% to 0.0009 wt.%; c) Er, measured as E O?. in an amount from 0.022 wt.% to 0.3 wt.%; and Co, measured as CO3O4, in an amount from 0 wt.% to 0.0001 wt.%, and optionally, alumina in an amount from 0 wt.% to 0.25 wt.%, wherein the shaded zirconia ceramic exhibits a flexural strength of at least 800 MPa when fully sintered.
Summary of Invention
However, there is still a desire for a highly aesthetic dental restoration which can be customized or individualized to the dental situation in the mouth of a patient at an early manufacturing step.
The dental restoration should be sufficiently strong and highly translucent in specific regions. Ideally, the dental restorations should be provided in a “ready-to-use” form, that is without the need for an additional glazing step.
If possible, a powder compacting and/or milling step should not be needed, either.
One or more of these objections can be addressed by the invention described in the present text.
The invention relates to a customized zirconia gel article, a dental restoration and a process of producing it as described in the claims and the description.
In particular, the invention relates to a customized zirconia gel article having the shape of a dental restoration, the customized zirconia gel article comprising a transition area comprising a transition gel, the transition area having a color gradient, the material composition of the transition gel comprising crystalline zirconia particles, a crystal phase stabilizer component, a cured organic component, optionally a photo-initiator, a liquid, and a coloring component, wherein the crystal phase stabilizer component is contained in the crystalline zirconia particles and the coloring component can be contained in the crystalline zirconia particles or be present as a separate component in the gel.
Further, the invention relates to a process for producing the customized zirconia gel article described in the present text and claims, the process comprising the steps of placing a curable sol Si in an amount Ai into a mould with a non-planar inner bottom surface, placing a curable sol S 2 in an amount A2 in contact with curable sol Si, optionally placing curable sols SN in amounts AN in contact with either sol Si and/or sol S2 with N = 1, 2, 3, 4 conducting a curing step, sol Si, sol S2 and optional sols SN each comprising crystalline zirconia particles, a crystal phase stabilizer component, a curable organic component, a photo-initiator, optinally a coloring component, and a liquid, wherein the crystal phase stabilizer component is contained in the crystalline zirconia particles and the optional coloring component can be contained in the crystalline zirconia particles or be present as a separate component in the sol, wherein at least sol S2 contains a coloring component, wherein the viscosity of each sol is in a range of 50 to 3,000 mPa*s at 23°C; and wherein sol Si, sol S2 and optional sols SN differ from each other with respect to the following properties alone or in combination: content of coloring components, content of crystal phase stabilizing components, content of crystalline zirconia particles.
The invention also relates to a zirconia dental restoration which can be obtained from the customized zirconia gel article.
Figures
Fig. 1 shows a photograph of a dental restoration according to example 1 of the invention. Fig. 2 shows a schematic view of a process for producing the dental restoration of example 1. Fig. 3 shows a photograph of a dental restoration according to example 2 of the invention. Fig. 4 shows a schematic view of a process for producing the dental restoration of example 2. Fig. 5 shows a photograph of a dental restoration according to example 3 of the invention. Fig. 6 shows a schematic view of a process for producing the dental restoration of example 3. Fig. 7 shows a photograph of a dental restoration according to comparative example 1. Fig. 8 shows a photograph of a dental restoration according to comparative example 2.
Detailed Description
Unless defined differently, for this description the following terms shall have the given meaning:
A “hardenable or curable or polymerizable component” is any component which can be cured or solidified in the presence of a photo-initiator by radiation-induced polymerization or by heattreatment. A hardenable component may contain only one, two, three or more polymerizable groups. Typical examples of polymerizable groups include unsaturated carbon groups, such as a vinyl group being present i.a. in a (methyl) acrylate group.
As used herein, "(meth)acryl" is a shorthand term referring to "acryl" and/or "methacryl”. For example, a "(meth) acryloxy" group is a shorthand term referring to either an acryloxy group (i.e., CH2=CH-C(0)-0-) and/or a methacryloxy group (i.e., CH2=C(CH3)-C(O)-O-).
As used herein, "hardening" or "curing" a composition are used interchangeably and refer to polymerization and/or crosslinking reactions including, for example, photo-polymerization reactions and chemical -polymerization techniques (e. g., ionic reactions or chemical reactions forming radicals effective to polymerize ethylenically unsaturated compounds) involving one or more materials included in the composition.
“Dental article” means an article which is to be used in the dental or orthodontic field, especially as or for producing a dental restoration. A dental article has typically two different surface portions, an outer surface and an inner surface. The outer surface is the surface which is not in permanent contact with the surface of a tooth. In contrast thereto, the inner surface is the surface which is used for attaching or fixing the dental article to a tooth. If the dental article has the shape of a dental crown or dental veneer, the inner surface has typically a concave shape, whereas the outer surface has typically a convex shape. A dental article should not contain components which are detrimental to the patient's health and thus free of hazardous and toxic components being able to migrate out of the dental or orthodontic article.
“Dental restoration” means dental articles which are used for restoring a tooth to be treated. Examples of dental restorations include crowns, bridges, inlays, onlays, veneers, orthodontic articles and parts thereof. The dental restoration described in the present text does not have the shape of a milling block.
Examples of orthodontic articles include brackets, buccal tubes, attachments, cleats and buttons and parts thereof.
A “monolithic zirconia ceramic dental restoration” means a dental restoration which does not comprise ceramic phases other than zirconia or hafnia. In particular, a monolithic zirconia ceramic dental restoration does not comprise glass or glass ceramic materials or phases. A monolithic restoration comprises crystalline phases of zirconia, wherein these phases may include soluble dopant ions that effect the zirconia phase composition, translucency and color.
“Sol” refers to a continuous liquid phase containing discrete particles having sizes in a range of 1 nm to 100 nm or 1 nm to 50 nm, a so called “colloidal solution”. The sols described in the present text are translucent and do show a so-called “Tyndall effect” or “Tyndall scattering”. The size of the particles is below the wavelength of the visible light (400 to 700 nm).
A "gel” means a wet solid-like material in which a network of interconnected nanostructures spans the volume of a liquid medium. Gels tend to be mostly liquid in composition and typically exhibit the density of a liquid as result but have cohesiveness like a solid. A sol can become a gel when the solid nanoparticles dispersed in it can join together to form a network of particles that spans the liquid.
“Coloring components or ions” shall mean components or ions which have an absorption in the spectrum visible to the human eye (e.g., from 380 to 780 nm), which results in a colored solution (visible to the human eye), if the coloring ions are dissolved in water (e.g., about 0.6 mol/1) and/or cause a coloring effect in the zirconia article containing coloring ions or components.
A translucent composition lets light partially permeate through although it is not fully transparent, i.e. showing a significant volume scattering of the transmitted light. The reciprocal property of translucency is opacity (O). O = 1/T = 1/10 (T = Transmission, I = Intensity of permeated light, I = Intensity of light before permeation). So, opacity values less than about 0.9 for a 1 mm thick platelet with a diameter of 15 mm are regarded as translucent (e.g., for a measurement with a Color i7 device, X-Rite corporation USA, measurement mode: remission contrast ratio). Opacity can be measured by various means: in transmission, in remission, and in remission using the contrast ratio method.
A “particle” means a substance being a solid having a shape which can be geometrically determined. The shape can be regular or irregular. Particles can typically be analysed with respect to e.g., particle size and particle size distribution.
The term “primary particle size” refers to the size of a non-associated single particle, which is considered to be a primary particle.
A dental ceramic article is classified as “pre-sintered” if the dental ceramic article has been treated with heat (temperature range of 900 to l,100°C) for 1 to 3 h to such an extent that the raw breaking resistance of the dental ceramic measured according to the “punch on three ball test” ISO 6872:2015 is within a range of 15 to 55 MPa or 30 to 50 MPa. A pre-sintered dental ceramic article usually has a porous structure and its density (usually 3.0 g/cm3 for a 3 mol% yttrium stabilized zirconia ceramic) is less compared to a completely sintered dental ceramic framework (usually about 6.1 g/cm3 for a 3 mol% yttrium stabilized zirconia ceramic).
“Ceramic zirconia article” shall mean a 3 -dimensional article wherein at least one the x, y, z dimension is at least about 5 mm, the article being comprised of at least 80 wt.% or at least 90 wt.% zirconia.
“Ceramic” means an inorganic non-metallic material that is produced by application of heat. Ceramics are usually hard and brittle and, in contrast to glasses or glass ceramics, display an essentially purely crystalline structure.
“Crystalline” means a solid composed of atoms arranged in a pattern periodic in three dimensions (i.e., has long range crystal structure as determined by X-ray diffraction). Crystal structures include tetragonal, monocline, cubic zirconia and mixtures thereof.
“Diafdtration” is a technique that uses ultrafdtration membranes to completely remove, replace, or lower the concentration of salts or solvents from solutions containing organic molecules. The process selectively utilizes permeable (porous) membrane filters to separate the components of solutions and suspensions based on their molecular size.
The term “aerogel” shall mean a three-dimensional low density solid (i.e., less than 20 % of the density of an article having a porosity of less than 0.1 vol.%). An aerogel is a porous material derived from a gel, in which the liquid component of the gel has been replaced with a gas. The solvent removal is often done under supercritical conditions. During this process the network does not substantially shrink and a highly porous, low-density material can be obtained.
“Density” means the ratio of mass to volume of an object. The unit of density is typically g/cm3. The density of an object can be calculated e.g., by determining its volume (e.g., by calculation or applying the Archimedes principle or method) and measuring its mass.
The term “tubular reactor” refers to the portion of a continuous hydrothermal reactor system that is heated (i.e., the heated zone). The tubular reactor can be in any suitable shape. The shape of the tubular reactor is often selected based on the desired length of the tubular reactor and the method used to heat the tubular reactor. For example, the tubular reactor can be straight, U-shaped, or coiled. The interior potion of the tubular reactor can be empty or can contain baffles, balls, or other known mixing techniques.
The term “calcining” refers to a process of heating a solid material to drive off at least 90 wt.% of volatile chemically bond components (e.g., organic components) (vs., for example, drying, in which physically bonded water is driven off by heating). Calcining is done at a temperature below a temperature needed for conducting a pre-sintering step.
The terms “sintering” or “firing” are used interchangeably. A pre-sintered ceramic article shrinks during a sintering step, that is, if an adequate temperature is applied. The sintering temperature to be applied depends on the ceramic material chosen. For zirconia based ceramics a typical sintering temperature range is l,100°C to l,550°C. Sintering typically includes the densification of a porous material to a less porous material (or a material having less cells) having a higher density, in some cases sintering may also include changes of the material phase composition (for example, a partial conversion of an amorphous phase toward a crystalline phase). Sintering of firing means making objects from a compressed powder by heating the material (typically below its melting point - solid state sintering) until its particles adhere to each other.
“Additive manufacturing” means processes used to make 3 -dimensional articles. An example of an additive manufacturing technique is vat polymerisation, in particular stereolithography (SLA),
digital light processing (DLP), continuous liquid interface production (CLIP) and volumetric additive manufacturing (VAM).
The articles can be of almost any shape or geometry and are produced from a 3 -dimensional model or other electronic data source.
“Ambient conditions” mean the conditions which the composition described in the present text is usually subjected to during storage and handling. Ambient conditions may, for example, be a pressure of 900 to 1,100 mbar, a temperature of 10 to 40 °C and a relative humidity of 10 to 100 %. In the laboratory ambient conditions are typically adjusted to 20 to 25 °C and 1 ,000 to 1 ,025 mbar (at maritime level).
As used herein, “a”, “an”, “the”, “at least one” and “one or more” are used interchangeably. Also herein, the recitations of numerical ranges by endpoints include all numbers subsumed within that range (e.g., 1 to 5 includes 1, 1.5, 2, 2.75, 3, 3.80, 4, 5, etc.).
Adding an “(s)” to a term means that the term should include the singular and plural form. E.g., the term “additive(s)” means one additive and more additives (e.g. 2, 3, 4, etc.).
Unless otherwise indicated, all numbers expressing quantities of ingredients, measurement of physical properties such as described below and so forth used in the specification and claims are to be understood as being modified in all instances by the term “about”.
The terms “comprise” or “contain” and variations thereof do not have a limiting meaning where these terms appear in the description and claims. “Consisting essentially of’ means that specific further components can be present, namely those which do not materially affect the essential characteristic of the article or composition. “Consisting of’ means that no further components should be present. The term “comprise” shall include also the terms “consist essentially of’ and “consists of’.
A composition is “essentially or substantially free of’ a certain component, if the composition does not contain said component as an essential feature. Thus, said component is not wilfully added to the composition either as such or in combination with other components or ingredient of other components. A composition being essentially free of a certain component usually does not contain that component at all. However, sometimes the presence of a small amount of the said component is not avoidable e.g., due to impurities contained in the raw materials used.
The customized zirconia gel article, the zirconia ceramic dental restoration and the process for production described in the present text are advantageous for a variety of reasons.
It was found that for achieving a highly aesthetic dental restoration it would be advantageous, if the interior area of a dental restoration shows a color gradient and is less transmissive than the exterior area at least with respect to some regions.
Compared to the application of coloring liquids on the outer surface area of a dental restoration, the coloring concept proposed in the present text better matches with the dental situation in the mouth of a patient.
Further, an undesired diffusion-controlled color gradient from the outer surface area to the inner surface area of a dental restoration can be avoided.
It was also found that in particular with using nano-sized zirconia materials not only highly translucent articles can be manufactured more easily but also that a lower temperature for sintering can be used.
In contrast, dental restorations obtained by milling from a compacted zirconia dental mill blank have a larger average grain size and require a higher sintering temperature.
In addition, the invention enables the manufacturing of dental restorations with a vertically arrangement of differently colored areas.
This is in contrast to dental restorations obtained by milling from dental mill blanks with a color gradient having rather a horizontally layered structure.
Further, a dental restoration obtained from a customized zirconia gel article comprising two different gels and comprising a diffusion-controlled transition gel area between the two gels allows for a more natural color gradient from the inner surface area of the dental restoration to the outer surface area.
The invention also enables the manufacturing of individualized, that is, customized dental restorations.
Depending on its curvature, the convex surface of the outer surface area of the dental restoration corresponds to the concave inner surface of the respective mould used for receiving the curable sols.
Similarly, the concave surface of the inner surface area of the dental restoration corresponds to the convex outer surface of the respective mould used for receiving the curable sols.
The inner surface of such a mould can easily be shaped or manufactured based on information associated with the dental situation of the patient. This process can therefore take advantage of both geometric and tooth shade data from an individual patient to design and produce restorations with excellent fit and aesthetics.
The invention also enables the production of filigree dental restorations, in particular dental restoration with thin side walls.
The invention is directed to a customized zirconia gel article having the shape of a dental restoration.
The zirconia gel article comprises a transition area comprising a transition gel, wherein the transition area has a color gradient.
The shape of the transition area is not particularly limited, but typically non-planar.
According to a more specific embodiment, the customized zirconia gel article comprises at least three areas, an inner surface area comprising gel Gi, an outer surface area comprising gel G2 and a transition area comprising transition gel G1/2.
The areas of the zirconia gel article originate from curing the sols used for producing the customized zirconia gel article and have essentially corresponding volumes.
The geometry of the inner surface, the outer surface or of both, the inner surface and the outer surface of the respective areas is typically based on information associated with the dental situation of a patient. That is, the geometry of at least one of the surfaces of the zirconia gel article is customized.
The material compositions of the gels each comprise crystalline zirconia particles, a crystal phase stabilizer component, a cured organic component, in particular a radiation-cured organic component, optionally a photo-initiator, a liquid, and optionally a coloring component.
Using a radiation-curable sol comprising radiation-curable components and a photo-initiator is often preferred.
The crystal phase stabilizer component is contained in the crystalline zirconia particles and the optional coloring component can be contained in the crystalline zirconia particles or be present as a separate component.
At least one gel and the transition gel contain a coloring component, wherein gel Gi is different from gel G2. If desired, one or more coloring components can be used.
The transition gel comprises a mixture of the material compositions of gels Gi and G2.
Like the dental restoration which can be obtained from the customized zirconia gel article, the customized zirconia gel article may have the shape of a dental crown, bridge, onlay, or veneer.
However, the size of the customized zirconia gel article is typically larger than the size of the dental restoration, e.g., by a factor of at least 2 in each dimension.
The transition area can be planar or non-planar. For highly individualized dental restorations a non- planar transition gel area can be preferred as such a geometry often better mimics the structure of a natural tooth.
The content of coloring components in the transition area is typically lower than the content of the coloring components in the inner surface area.
The invention also relates to a process for producing the customized zirconia gel article and related dental restoration described in the present text.
For the production at least two different curable sols are used, sol Si and sol S2. Optionally and if desired, further sols SN can be used, with N = 1, 2, 3, 4, . . . or N = 1, 2, 3, 4, . . . 10.
These sols are placed into a mould having a non-planar inner bottom surface and/or on top of each other.
The placing of the sols can be done either manually or automated, e.g., with the help of a robot.
The placing of the sols into the mould can be done by various methods, including casting, using an inkjet-printing technology, a gradient printing technology using vat polymerization, or any other suitable technique.
The non-planar inner bottom surface of the mould typically comprises a section with a concave region, a convex region, or a mixed concave/convex region. A mixed concave/convex region can be advantageous for producing dental restorations with a so-called mamelon wave geometry.
The concave and/or convex regions are typically adapted to the shape of a tooth surface.
The non-planar bottom surface of the mould may comprise one or more sections with a shape which is based on information associated with the dental situation of a patient.
Such a shape facilitates the manufacturing of customized dental restoration.
The information associated with the dental situation of a patient is so-called digital data can be obtained and provided by means known to the skilled person, e.g., by photographing or scanning the dental situation in the mouth of a patient or by using information stored in a database.
As the process described in the present text is in particular suitable for producing an individual dental restoration, the amounts of the sols to be used in the process is rather low.
The sols can be used in various amounts. For producing a customized dental restoration only small amounts of sols are typically needed.
Useful amounts may be in the range of 0.0001 to 1 ml or 0.01 to 0.5 ml.
The individual sols are typically used in different amounts with respect to volume.
Usually, sol Si is used in a larger amount than sol S2 and larger than optionally sols SN, e.g., by a ratio larger than 2/1 or 5/1 or 10/1 with respect to volume.
Sol Si is typically used for forming the outer surface area of the dental restoration, whereas sol S2 and optional sols SN are used for individualizing the inner surface area of the dental restoration, which often requires a smaller amount of material.
Sol Si, sol S2 and optional sols SN differ from each other with respect to the following properties alone or in combination: (i) content of optional coloring components, (ii) content of crystal phase stabilizing components, and/or (iii) content of crystalline zirconia particles, wherein a properties (i) and (ii) and the combination of properties (i) and (ii) is sometimes preferred.
The process for producing the customized zirconia gel article may comprise in addition the following steps before step (a): providing sols with different contents of stabilizing and/or coloring components, mixing at least two of these sols to obtain curable sol Si, curable sol S2, and optionally curable sols SN-
If desired, the chemical compositions of curable sol Si, curable sol S2, and optionally curable sols SN can be adjusted to match with information associated with the dental situation of a patient. This may facilitate the production of even better customized dental restorations.
If desired, the curable sol S2 and/or sols SN are placed in contact with the curable sol Si only in one or more individual areas of the top surface of sol Si.
If desired, at least two different curable sols S2 can be placed in contact with the curable sol Si only in one or more individual areas of the top surface of sol Si.
If differently colored zirconia sols are used, differently colored surface areas can be realized.
This allows the provision of customized dental articles adapted to the individual situation in the mouth of a patient.
Using a more opaque sol Si for the inner surface of the dental article can be advantageous to mask a dark surface of a tooth stump without effecting the depth of reflections on the outer surface of the dental article.
The viscosity of each sol is typically in a range of 50 to 3,000 mPa*s or 60 to 2,000 mPa* at 23°C. Such a viscosity was found to be advantageous for a variety of reasons: It facilitates the placing of the sols into the mould by either casting or inkjet-printing. The viscosity is low enough to enable a diffusion-controlled mixing of the sols in the interface or transition area leading to a smooth color gradient from the inner surface area to the outer surface area. The viscosity is high enough to avoid an undesired blending of the sols when placing them into the mould.
If desired, sol Si, sol S2 and optional sols SN can be further characterized by the following properties alone or in combination: being translucent in a wavelength range of 420 to 600 nm for a path length of 10 mm; showing a transmission of at least 5% at a wavelength of 420 nm for a path length of 10 mm; having a pH value in the range of 1 to 6.
It was found that using a translucent sol can be beneficial for improving the accuracy or detail resolution of the surface of the ceramic article. Translucent sols show less scattering of light, which is used for polymerizing the curable components contained in the sol. The increased translucency allows for a shallower cure gradient, which may also allow for a more uniform cure across the entire structure to be obtained, as lower energy doses are required to cure through a translucent material.
A transmission in such a range was found to be sufficiently high for enabling the radiation -curing of the sol within an adequate time frame for the desired thickness.
The mould into which the sols are casted has a non-planar inner bottom surface.
As the dental restorations described in the present text are customized, the respective mould used for producing the dental restorations is customized too, in particular as regards its inner bottom surface.
As desired, the mould or the inner bottom surface can be produced by various techniques including thermoforming, 3d-printing, or casting e.g., via lab duplication techniques in silicone, hydrocolloid, etc.
Dental thermoforming works by applying heat and pressure to a plastic sheet which is positioned on top of a 3d-model produced (e.g., by using an additive-manufacturing technique) specifically for that patient.
Alternatively, the mould can directly be produced by additive -manufacturing using the information associated with the dental situation of a patient.
Additive-manufacturing technique which can be used include vat polymerization, stereolithography (SLA), digital light processing (DLP), robocasting (RC), material jetting (MJ), binder jetting (BJ), and volumetric additive manufacturing (VAM).
The information associated with the dental situation of a patient can be obtained by using e.g., an intraoral scanner. Various intraoral scanners are meanwhile commercially available on the market (e.g., from 3Shape, Planmeca, and others). Alternatively, the information can be obtained from and matched with data stored in a tooth library.
Similarly, if desired, an adequate tooth color can be determined by using either a tooth shade guide (e.g., VITA Toothguide 3D-MASTER™), or an electronic device (e.g., VITA Easyshade™ LITE, 3 Shape Trios™ 3).
The designing and manufacturing processes may also include Computer-Aided-Design (CAD) and/or Computer- Aided-Manufacturing (CAM). Mould dimensions are typically scaled up to account for shrinkage between the casting process and subsequent binder removal and sintering processes.
Any suitable material for the mould can be used. Suitable materials include PET, and polyurethanes. Alternatively, moulds may be formed by casting silicone or hydrocolloid or other conventional dental laboratory duplicating materials.
Moulds may be of frangible (e.g., polymer) or degradable (e.g., wax) materials to accommodate undercut features.
Moulds may be single component or may be assembled from multiple components and may contain parting lines, slides, etc. to accommodate production and reuse.
The curable sols comprise crystalline zirconia particles, preferably in an amount of 25 to 65 wt.%, a crystal phase stabilizer component, preferably in an amount of 2 to 8 mol% calculated as oxide with respect to the total crystalline oxide particles, a curable organic component, preferably in an amount of 2 to 30 wt.%, optionally a photo-initiator, preferably in an amount of 0.01 to 3 wt.%, a liquid, preferably in an amount of 25 to 70 wt.%, optionally coloring ions, preferably in an amount of 0 to 2 wt.%, optionally inhibitor components, preferably in an amount of 0 to 0.5 wt.%, wt.% with respect to the weight of the curable sol, wherein the crystal phase stabilizer component and/or the optional coloring ions can also be contained in the crystalline zirconia particles.
The crystalline zirconia particles in the sol typically have a primary particle size in a range of 2 nm to 50 nm (in some embodiments, 5 nm to 50 nm, 2 nm to 25 nm, 5 nm to 25 nm, 2 nm to 15 nm, or even 5 nm to 15 nm).
The crystalline zirconia particles are typically present in the following amounts: at least 25 or at least 30 or at least 35 wt.%; at most 65 or at most 60 or at most 55 wt.%; from 25 to 65, or 30 to 60, or 35 to 55 wt.%; wt.% with respect to the weight of the sol.
Using a sol with a high crystalline zirconia particles content can be advantageous as the efforts for removing the liquid in later process steps is reduced. Thus, a crystalline zirconia particles content in the range of 35 to 65 wt.% can be preferred.
Crystal phase stabilizing components which can be used include ions selected from Ce, Mg, Ca, Y, La, rare earth elements or combinations thereof, wherein the use of Y is sometimes preferred.
The crystal phase stabilizer component calculated as oxide is typically present in the following amounts: at least 2 or at least 3 or at least 4 mol%; at most 7 or at most 6 or at most 5 mol%; from 2 to 7, or 3 to 6, or 4 to 5 mol%; mol% with respect to the total crystalline oxide particles.
For being able to act as crystal phase stabilizer, the crystal phase stabilizing components are typically contained in the crystalline zirconia articles.
The curable organic component being present in the sol can be described as first, second, third, etc. monomer.
Zirconia particles having been surface-modified with a polymerizable agent can be polymerized, if desired, to provide a composition comprising crosslinked zirconia particles.
A first monomer can function as a polymerizable surface modification agent. Multiple first monomers can be used.
Surface modification agents may be represented by the formula A-B, where the A group is capable of attaching to the surface of a zirconia-based particle and the B group is radiation curable.
Group A can be attached to the surface of the zirconia-based particle by adsorption, formation of an ionic bond, formation of a covalent bond, or a combination thereof.
Examples for Group A include acidic moieties (like carboxylic acid groups, phosphoric acid groups, sulfonic acid groups and anions thereof) and silanes.
Group B comprises a radiation curable moiety.
Examples for Group B include vinyl, in particular acryl or methacryl moieties.
Suitable surface modifying agents comprise polymerizable carboxylic acids and/or anions thereof, polymerizable sulfonic acids and/or anions thereof, polymerizable phosphoric acids and/or anions thereof, and polymerizable silanes. Suitable surface modification agents are further described, for example, in WO 2009/085926 Al (Kolb et al.).
An example of a radically polymerizable surface modifier is a polymerizable surface modification agent comprising an acidic moiety or anion thereof, e.g. a carboxylic acid group.
Exemplary acidic radically polymerizable surface modifiers include acrylic acid, methacrylic acid, beta-carboxyethyl acrylate, and mono-2-(methacryloxyethyl)succinate.
Exemplary radically polymerizable surface modifiers can be reaction products of hydroxylcontaining polymerizable monomers with cyclic anhydrides such as succinic anhydride, maleic anhydride and phthalic anhydride. Exemplary polymerization hydroxyl-containing monomers include hydroxyethyl acrylate, hydroxyethyl methacrylate, hydroxypropyl acrylate, hydroxypropyl methacrylate, hydroxyl butyl acrylate, and hydroxybutyl methacrylate. Acryloxy and methacryloxy functional polyethylene oxide, and polypropylene oxide may also be used as the polymerizable hydroxyl-containing monomers.
An exemplary radically polymerizable surface modifier for imparting both polar character and reactivity to the zirconia-containing nanoparticles is mono(methacryloxypolyethyleneglycol) succinate.
Another example of a radically polymerizable surface modifier is a polymerizable silane.
Exemplary polymerizable silanes include methacryloxyalkyltrialkoxysilanes or acryloxyalkyltri- alkoxysilanes (e.g., 3-methacryloxypropyltrimethoxysilane, 3-acryloxypropyltrimethoxysilane, and 3-(methacryloxy)propyltriethoxysilane); methacryloxyalkylalkyldialkoxysilanes or acryloxyalkyl- alkyldialkoxysilanes (e.g., 3-(methacryloxy)propylmethyldimethoxysilane and 3-(acryloxy- propyl)methyldimethoxy silane); methacryloxy alkyldialkylalkoxysilanes or acyrloxy alkyldialkylalkoxysilanes (e.g. , 3 -(methacryloxy)propyldimethylethoxysilane) ; mercaptoalkyltrialkoxylsilanes (e.g., 3 -mercaptopropyltrimethoxy silane); aryltrialkoxysilanes (e.g., styrylethyltrimethoxy silane); vinylsilanes (e.g., vinylmethyldiacetoxysilane, vinyldimethylethoxysilane, vinylmethyl- diethoxysilane, vinyltrimethoxy silane, vinyltriethoxy silane, vinyltriacetoxysilane, vinyl- triisopropoxy silane, and vinyltris(2- methoxy ethoxy)silane).
According to one embodiment, the sol described in the present text comprises one or more second monomers comprising at least two radiation-curable moieties. Those second monomer(s) may act as crosslinker(s) during the gel-forming step.
Any suitable second monomer that does not have a surface modification group can be used. That is, the optional second monomer does not have a carboxylic acid group or a silyl group. The second monomers are often polar monomers (e.g., non-acidic polar monomers), monomers having a plurality of polymerizable groups, alkyl (meth)acrylates and mixtures thereof.
The presence of the monomer having a plurality of polymerizable groups tends to enhance the strength of the gel composition formed when the sol is polymerized.
The number of polymerizable groups can be in a range of 2 to 6 or even higher. In many embodiments, the number of polymerizable groups is in a range of 2 to 5 or 2 to 4. The polymerizable groups are typically (meth)acryloyl groups.
Exemplary monomers with two (meth)acryloyl groups include 1,2-ethanediol diacrylate, 1,3- propanediol diacrylate, 1,9 -nonanediol diacrylate, 1,12 -dodecanediol diacrylate, 1 ,4-butanediol
diacrylate, 1,6-hexanediol diacrylate, butylene glycol diacrylate, bisphenol A diacrylate, diethylene glycol diacrylate, triethylene glycol diacrylate, tetraethylene glycol diacrylate, tripropylene glycol diacrylate, polyethylene glycol diacrylate, polypropylene glycol diacrylate, poly- ethylene/polypropylene copolymer diacrylate, polybutadiene di(meth)acrylate, propoxylated glycerine tri(meth)acrylate, and neopentylglycol hydroxypivalate diacrylate modified caprolactone. Exemplary monomers with three or four (meth)acryloyl groups include trimethylolpropane triacrylate (e.g., commercially available as TMPTA-N™ from Cytec Industries, Inc. (Smyrna, GA, USA) and as SR-351™ from Sartomer (Exton, PA, USA)), pentaerythritol triacrylate (e.g., commercially as SR-444™ from Sartomer), ethoxylated (3) trimethylolpropane triacrylate (e.g., commercially available as SR-454™ from Sartomer), ethoxylated (4) pentaerythritol tetraacrylate (e.g., commercially available as SR-494™ from Sartomer), tris(2-hydroxyethylisocyanurate) triacrylate (e.g., commercially available as SR-368™ from Sartomer), a mixture of pentaerythritol triacrylate and pentaerythritol tetraacrylate (e.g., commercially available from Cytec Industries, Inc., as PETIA™ with an approximately 1:1 ratio of tetraacrylate to triacrylate and under the trade designation PETA-K™ with an approximately 3:1 ratio of tetraacrylate to triacrylate), pentaerythritol tetraacrylate (e.g., commercially available as SR-295™ from Sartomer), and ditrimethylolpropane tetraacrylate (e.g., commercially available as SR-355™ from Sartomer).
Exemplary monomers with five or six (meth)acryloyl groups include dipentaerythritol pentaacrylate (e.g., commercially available as SR-399™ from Sartomer) and a hexa-functional urethane acrylate (e.g., commercially available as CN975™ from Sartomer).
In some embodiments, the optional second monomer is a polar monomer. As used herein, the term “polar monomer” refers to a monomer having a free radical polymerizable group and a polar group. The polar group is typically non-acidic and often contains a hydroxyl group, a primary amido group, a secondary amido group, a tertiary amido group, an amino group, or an ether group (i.e., a group containing at least one alkylene-oxy-alkylene group of formula -R-O-R- where each R is an alkylene having 1 to 4 carbon atoms).
Suitable optional polar monomers having a hydroxyl group include, but are not limited to, hydroxyalkyl (meth)acrylates (e.g., 2-hydroxyethyl (meth)acrylate, 2-hydroxypropyl (meth)acrylate, 3 -hydroxypropyl (meth)acrylate, and 4-hydroxybutyl (meth)acrylate), and hydroxyalkyl (meth) acrylamides (e.g., 2-hydroxyethyl (meth)acrylamide or 3 -hydroxypropyl (meth)acrylamide), ethoxylated hydroxyethyl (meth) acrylate (e.g., monomers commercially available from Sartomer (Exton, PA, USA) as CD570™, CD571™, and CD572™), and aryloxy substituted hydroxyalkyl (meth)acrylates (e.g., 2-hydroxy-2-phenoxypropyl (meth) acrylate).
Exemplary polar monomers with a primary amido group include (meth)acrylamide. Exemplary polar monomers with secondary amido groups include, but are not limited to, N-alkyl (meth)acrylamides such as N-methyl (meth)acrylamide, N-ethyl (meth)acrylamide, N-isopropyl (meth)acrylamide, N-tert-octyl (meth)acrylamide, and N-octyl (meth)acrylamide. Exemplary polar
monomers with a tertiary amido group include, but are not limited to, N-vinyl caprolactam, N- vinyl-2-pyrrolidone, (meth)acryloyl morpholine, and N,N-dialkyl (meth)acrylamides such as N,N- dimethyl (meth)acrylamide, N,N-diethyl (meth)acrylamide, N,N-dipropyl (meth)acrylamide, and N,N-dibutyl (meth)acrylamide.
Polar monomers with an amino group include various N,N-dialkylaminoalkyl (meth)acrylates and N,N-dialkylaminoalkyl (meth)acrylamides. Examples include, but are not limited to, N,N-dimethyl aminoethyl (meth) acrylate, N,N-dimethylaminoethyl (meth) acrylamide, N,N-dimethylaminopropyl (meth)acrylate, N,N-dimethylaminopropyl (meth)acrylamide, N,N-diethylaminoethyl (methacrylate, N,N-diethylaminoethyl (meth)acrylamide, N,N-diethylaminopropyl (meth)acrylate, and N,N -diethylaminopropyl (meth) acrylamide .
Exemplary polar monomers with an ether group include, but are not limited to, alkoxylated alkyl (meth)acrylates such as ethoxyethoxyethyl (meth)acrylate, 2 -methoxy ethyl (meth)acrylate, and 2- ethoxyethyl (meth)acrylate; and poly( alkylene oxide) (meth)acrylates such as poly(ethylene oxide) (meth)acrylates, and poly(propylene oxide) (meth)acrylates. The poly(alkylene oxide) acrylates are often referred to as poly(alkylene glycol) (meth)acrylates. These monomers can have any suitable end group such as a hydroxyl group or an alkoxy group. For example, when the end group is a methoxy group, the monomer can be referred to as methoxy poly(ethylene glycol) (meth)acrylate.
Suitable alkyl (meth)acrylates that can be used as a second monomer can have an alkyl group with a linear, branched, or cyclic structure. Examples of suitable alkyl (meth)acrylates include, but are not limited to, methyl (meth)acrylate, ethyl (meth)acrylate, n-propyl (meth)acrylate, isopropyl (meth)acrylate, n-butyl (meth) acrylate, isobutyl (meth)acrylate, n-pentyl (meth)acrylate, 2- methylbutyl (meth)acrylate, n-hexyl (meth)acrylate, cyclohexyl (meth)acrylate, 4-methyl-2-pentyl (meth)acrylate, 2-ethylhexyl (meth) acrylate, 2-methylhexyl (meth)acrylate, n-octyl (meth)acrylate, isooctyl (meth)acrylate, 2-octyl (meth)acrylate, isononyl (meth)acrylate, isoamyl (meth)acrylate, 3,3,5-trimethylcyclohexyl (meth)acrylate, n-decyl (meth)acrylate, isodecyl (meth)acrylate, isobornyl (meth)acrylate, 2-propylheptyl (meth)acrylate, isotridecyl (meth)acrylate, isostearyl (meth)acrylate, octadecyl (meth) acrylate, 2-octyldecyl (meth)acrylate, dodecyl (meth)acrylate, lauryl (meth)acrylate, and heptadecanyl (meth) acrylate.
In certain embodiments the second monomer(s) can be characterized by the following parameters alone or in combination: (i) soluble in the liquid contained in the sol; (ii) bearing at least one or two or three curable moieties; (iii) bearing radiation curable moieties selected from vinyl, acryl or methacryl moieties; (iv) molecular weight (Mw): 70 to 5,000 g/mol; or 70 to 1,000 g/mol; or 100 to 500 g/mol, wherein the combination of parameters (ii) and (iii), or (ii) and (iv), or (ii), (iii) and (iv) is sometimes preferred.
Using radiation curable component(s) as described above having a molecular weight in the above range facilitates the provision of a sol having the desired viscosity. Lower molecular weight components are typically also better soluble than high molecular weight components.
The curable component is typically present in the following amounts: at least 2 or at least 5 or at least 10 wt.%; at most 30 or at most 25 or at most 20 wt.%; from 2 to 30 or 5 to 25 or 10 to 20 wt.%; wt.% with respect to the weight of the sol.
The sol described in the present text comprises one or more photo-initiator(s).
The nature and structure of the photo-initiator is not particularly limited, either, unless the desired result cannot be achieved.
In certain embodiments the photo-initiator(s) can be characterized by at least one or more, sometimes all of the following parameters: soluble in the liquid contained in the sol; and/or showing radiation absorption within a range of 200 to 500 nm or 300 to 450 nm.
The photo-initiator should be able to start or initiate the curing or hardening reaction of the radiation curable component(s) being present in the sol.
The following classes of photo-initiator(s) can be used: a) two-component system where a radical is generated through abstraction of a hydrogen atom form a donor compound; b) one component system where two radicals are generated by cleavage.
Examples of photo-initiators according to type (a) typically contain a moiety selected from benzophenone, xanthone or quinone in combination with an aliphatic amine.
Examples of photo-initiators according to type (b) typically contain a moiety selected form benzoin ether, acetophenone, benzoyl oxime or acyl phosphine.
Exemplary UV initiators include 1 -hydroxy cyclohexyl benzophenone (previously available from Ciba Specialty Chemicals Corp as IRGACURE 184™), 4-(2-hydroxyethoxy)phenyl-(2-hydroxy-2- propyl) ketone (previously available from Ciba Specialty Chemicals Corp as IRGACURE 2529™, 2-hydroxy-2-methylpropiophenone (previously available from Ciba Specialty Chemicals Corp, as DAROCURE Di l l™) and bis(2,4,6-trimethylbenzoyl)-phenylphosphineoxide (previously available from Ciba Specialty Chemicals Corp as IRGACURE 819™).
The photo-initiator is typically present in the following amounts: at least 0.01 or at least 0.1 or at least 0.2 wt.%; at most 3 or at most 2 or at most 1 wt.%; from 0.01 to 3 or 0.1 to 2 or 0.2 to 1 wt.%; wt.% with respect to the weight of the sol.
The nature and structure of the liquid is not particularly limited unless the desired result cannot be achieved. In certain embodiments the solvent can be characterized by the following features alone or in combination: a) boiling point: above 100°C or above 120°C or above 150°C; b) molecular weight: 25 to 300 g/mol; c) viscosity: 0.2 to 10 mPa*s (23°C); d) miscible with water; e) soluble in supercritical carbon dioxide or liquid carbon dioxide.
A combination of the following features is sometimes preferred: a) and b), or a), b) and c), or a), b), c) and d).
Using a liquid with a boiling point above 100°C or 150°C can be beneficial for reducing the evaporation of the liquid during the process.
Using a liquid with a molecular weight and/or viscosity in the above range can be beneficial as it helps in adjusting the viscosity of the sol. The molecular weight size can also affect the diffusion constant and how easily the liquid can be removed.
Using a mixture of different liquids can be beneficial as it allows adjusting the viscosity or post processing properties, e.g., removal of excess sol after casting.
The liquid should also be easily removable during the further processing steps needed for the realization of a ceramic article.
Further, the liquid should not interfere with or negatively influence the polymerization of the curable components being present in the sol.
In this respect, using liquid not bearing polymerizable moieties can be beneficial.
To enhance the dissolving capability or property of the liquid, the liquid typically bears one or more polar moieties, including ether, alcohol or carboxy moieties.
According to one embodiment, the liquid is often a glycol or polyglycol, mono-ether glycol or mono-ether polyglycol, di-ether glycol or di-ether polyglycol, ether ester glycol or ether ester polyglycol, carbonate, amide, or sulfoxide (e.g., dimethyl sulfoxide).
Suitable glycols or polyglycols, mono-ether glycols or mono-ether polyglycols, di-ether glycols or di-ether poly glycols, and ether ester glycols or ether ester polyglycols are often of the following Formula (I).
R’O- R^jn-R1 (I)
In Formula (I), each R1 independently is hydrogen, alkyl, aryl, or acyl. Suitable alkyl groups often have 1 to 10 carbon atoms, 1 to 6 carbon atoms, or 1 to 4 carbon atoms. Suitable aryl groups often have 6 to 10 carbon atoms and are often phenyl or phenyl substituted with an alkyl group having 1 to 4 carbon atoms. Suitable acyl groups are often of formula -(CO)Ra where Ra is an alkyl having 1 to 10 carbon atoms, 1 to 6 carbon atoms, 1 to 4 carbon atoms, 2 carbon atoms, or 1 carbon atom. The acyl is often an acetyl group (-(COjCHs). In Formula (I), each R2 is typically ethylene or propylene. The variable n is at least 1 and can be in a range of 1 to 10, 1 to 6, 1 to 4, or 1 to 3.
Glycols or polyglycols of Formula (I) have two R1 groups equal to hydrogen. Examples of glycols include, but are not limited to, ethylene glycol, propylene glycol, diethylene glycol, dipropylene glycol, triethylene glycol, and tripropylene glycol.
Mono-ether glycols or mono-ether polyglycols of Formula (I) have a first R1 group equal to hydrogen and a second R1 group equal to alkyl or aryl. Examples of mono-ether glycols or monoether polyglycols include, but are not limited to, ethylene glycol monohexyl ether, ethylene glycol monophenyl ether, propylene glycol monobutyl ether, diethylene glycol monomethyl ether, diethylene glycol monoethyl ether, diethylene glycol monopropyl ether, diethylene glycol monobutyl ether, diethylene glycol monohexyl ether, dipropylene glycol monomethyl ether,
dipropylene glycol monoethyl ether, dipropylene glycol monopropyl ether, triethylene glycol monomethyl ether, triethylene glycol monoethyl ether, triethylene glycol monobutyl ether, tripropylene glycol monomethyl ether, and tripropylene glycol monobutyl ether.
Di-ether glycols or di-ether polyglycols of Formula (I) have two R1 group equal to alkyl or aryl. Examples of di-ether glycols or di-ether polyglycols include, but are not limited to, ethylene glycol dipropyl ether, ethylene glycol dibutyl ether, dipropylene glycol dibutyl ether, diethylene glycol dimethyl ether, diethylene glycol diethyl ether, triethylene glycol dimethyl ether, tetraethylene glycol dimethyl ether, and pentaethylene glycol dimethyl ether.
Ether ester glycols or ether ester polyglycols of Formula (I) have a first R1 group equal to an alkyl or aryl and a second R1 group equal to an acyl. Examples of ether ester glycols or ether ester polyglycols include, but are not limited to, ethylene glycol butyl ether acetate, diethylene glycol butyl ether acetate, and diethylene glycol ethyl ether acetate.
In Formula (II), R3 is hydrogen or an alkyl such as an alkyl having 1 to 4 carbon atoms, 1 to 3 carbon atoms, or 1 carbon atom. Examples include ethylene carbonate and propylene carbonate. Yet other suitable organic solvents are amides of Formula (III).
In Formula (III), group R4 is hydrogen, alkyl, or combines with R5 to form a five-membered ring including the carbonyl attached to R4 and the nitrogen atom attached to R5. Group R5 is hydrogen, alkyl, or combines with R4 to form a five-membered ring including the carbonyl attached to R4 and the nitrogen atom attached to R5. Group R6 is hydrogen or alkyl. Suitable alkyl groups for R4, R5, and R6 have 1 to 6 carbon atoms, 1 to 4 carbon atoms, 1 to 3 carbon atoms, or 1 carbon atom. Examples of amide organic solvents of Formula (III) include, but are not limited to, formamide, N,N-dimethylformamide, N,N-dimethylacetamide, N,N-diethylacetamide, N-methyl-2-pyrrolidone, and N-ethyl-2-pyrrolidone.
Specific examples of solvents which can be used include mono alcohols (e.g. C2 to Cs alcohols, including primary, secondary and tertiary alcohols), poly alcohols (e.g. ethylene glycol, propylene glycol, glycerine, diethylene glycol ethyl ether (Carbitol™), l-methoxy-2-propanol, N-methyl pyrrolidone, acetonitrile, chlorobenzene, 1,4-dioxane, ethyl acetate, methyl ethyl ketone, tetrahydrofuran, toluene, xylene and mixtures thereof.
The following solvents are sometimes preferred: ethanol, 1 -methoxy-2-propanol, N-methyl pyrrolidone, diethylene glycol ethyl ether, and mixtures thereof. In some situations, suitable solvents may also include low boiling alcohols (below 100°C; like methanol, ethanol, propanol) and mixtures thereof or preferably the same solvent(s) described above.
The liquid is typically present in the following amounts: at least 25 or at least 30 or at least 35 wt.%; at most 70 or at most 65 or at most 60 wt.%; from 25 to 70, or 30 to 65, or 35 to 60 wt.%; wt.% with respect to the weight of the sol.
Coloring components which can be used include in particular components comprising ions selected from Fe, Mn, Er, Pr, Tb, Cr, Co, Mo and/or Nd.
If present, these components are typically present as oxides in the sintered article.
The coloring ions are typically present in the following amounts: 0, or at least 0.1, or at least 0.2 wt.%; at most 2, or at most 1, or at most 0.8 wt.%; from 0 to 2, or 0.1 to 1, or 0.2 to 0.8 wt.%; wt.% total coloring ions with respect to the weight of the sol.
According to a further embodiment, the sol described in the present text comprises one or more inhibitor(s).
The nature and structure of the inhibitor(s) is not particularly limited, either, unless the desired result cannot be achieved.
An inhibitor may extend the shelf life of the sol, help prevent undesired side reactions, and adjust the polymerization process of the radiation curable component(s) present in the sol.
Adding one or more inhibitor(s) to the sol may further help to improving the accuracy or detail resolution of the surface of the ceramic article.
In particular it was found that adding inhibitor(s) to the printing sol described in the present text may help to enhance the resolution and accuracy of the SLA process by attenuating or avoiding unwanted scattering effects, as well as increase the shelf life of the sol.
The inhibitor(s) should be soluble in the solvent contained in the sol. Inhibitors which can be used often comprise a phenol moiety.
Specific examples of inhibitor(s) which can be used include: butylhydroxy toluol (Ionol), p- methoxyphenol (MOP), hydroquinone monomethylether (MEHQ), 2,6-di-tert-butyl-4-methyl- phenol (BHT), phenothiazine, 2,2,6,6-tetramethyl-piperidine-l-oxyl radical (TEMPO) and mixtures thereof.
The inhibitor is typically present in the following amounts: 0, or at least 0.001, or at least 0.01 wt.%; at most 0.5, or at most 0.02, or at most 0.01 wt.%; from 0 to 0.5, or 0.001 to 0.05, or 0.01 to 0.02 wt.%; wt.% with respect to the weight of the sol.
Generally, the sols described in the present text can be obtained as follows:
A starting sol containing nano-sized zirconia particles is provided.
A precursor solution is typically prepared by combining a zirconium salt (e.g., acetate) solution and a solvent (e.g., water). A phase stabilizing agent (e.g., yttrium acetate) is added and dissolved in the
precursor solution. The resulting composition is pumped e.g., through a hydrothermal reactor. Suitable hydrothermal reactors are described e.g., in US 5,453,262 (Dawson et al.) and US 5,652,192 (Matson et al.).
When subjected to hydrothermal treatment, the various dissolved salts undergo hydrolysis and condensation reactions to form zirconia-based particles. These reactions are often accompanied with the release of an acidic by-product (e.g., acetic acid).
The content of tetragonal and/or cubic phase of the zirconia crystallites can be adjusted by varying the amount of phase stabilizing components added during the production method.
The starting sol is typically concentrated.
To obtain a more concentrated sol, at least a portion of the aqueous-based medium is removed from the zirconia-based sol.
According to one embodiment, the zirconia-based sol is subjected to dialysis or diafiltration.
The content of the crystalline nano-sized zirconia particles in the concentrated starting sol is typically in a range of 25 to 65 wt.%.
In some embodiments, the zirconia-based sol can be subjected to a solvent exchange process.
An organic liquid having a higher boiling point than water can be added to the effluent. Examples of organic liquids that are suitable for use in a solvent exchange method include 1 -methoxylpropanol, N-methyl pyrrolidone or diethylene glycol ethyl ether. The water then can be removed by a method such as distillation, rotary evaporation, or oven drying.
A zirconia-based sol comprises zirconia-based particles dispersed and/or suspended (i.e., dispersed, suspended, or a combination thereof) in an aqueous/organic matrix.
To the starting sol the other components are added: the curable component(s), the photo-initiator(s), optionally organic dye(s), inhibitor(s), coloring component(s), and other additive(s), if desired.
The preparation of the sol is typically conducted under safe light conditions to avoid an undesired early polymerization.
Sol Si, sol S2 and optional sols SN can also be produced by mixing so-called master batches with different contents of stabilizing and/or coloring components.
As the sols to be used for producing customized dental restorations, the chemical composition of curable sol Si, curable sol S2, and optionally curable sols SN can be adjusted, if desired, to match with information associated with the dental situation of a patient.
Thus, depending on the desired color of the customized dental restoration to be produced, different amounts of differently colored master batches can be mixed to obtain sol Si, sol S2 and optional sols SN-
At least sol S2 contains coloring components, wherein sol S2 is typically less light-transmissive than sol Si.
Further, sol Si, sol S2 and optional sol SN differ from each other with respect to the following properties alone or in combination: (i) content of coloring components, (ii) content of crystal phase
stabilizer components, (iii) content of crystalline zirconia particles, (iv) viscosity, wherein a combination of (i) and (ii), or (i) and (iii), or (ii) and (iii) is sometimes preferred.
If there is difference in viscosity, the difference in viscosity between the sols is typically greater than a ratio of 1.2 or 1.5 or 2.
The process for producing the customized zirconia gel article further comprises a curing step, in particular a radiation-curing step.
The radiation-curing step is typically conducted under the following conditions: wavelength: 300 to 500 nm, or 365 to 460 nm; duration: 1 s to 5 min; temperature: 10 to 30°C.
By conducing a radiation-curing step, the curable components contained in the sol are at least partially crosslinked leading to the formation of a gel article.
The process described in the present text may also comprise a post-curing step.
If desired, the zirconia gel article can be post-cured by applying radiation or heat.
Such a step may help to improve the stability of the zirconia gel article by further increasing the degree of polymerization.
If present, the post-curing step can be characterized by the following features alone or in combination: (i) applying radiation with wavelength of 200 to 500 nm or 350 to 450 nm; (ii) applying a heating step with a temperature below the temperature at which drying will occur or which is used for de-bindering or calcining; e.g., 30 to 110°C or 40 to 80°C.
A post-curing step is typically conducted after the step of radiation-curing the curable zirconia sols. For producing a dental restoration, the process may comprise further steps.
These steps may yield intermediate products such as an aerogel article and/or a pre-sintered zirconia article.
One additional step may relate to the transforming of the zirconia gel article into a zirconia aerogel article.
The transforming step can be achieved by a process comprising an extraction step.
A zirconia aerogel article can be formed by removing liquid from the zirconia gel article without excessive shrinkage (e.g., not greater than about 30 or 40 vol.%).
Water, if present, can be removed from the zirconia gel article e.g., via alcohol exchange to provide an at least partially de-watered gel. The zirconia gel article is then converted or transformed to a zirconia aerogel article by removing the alcohol, if present, from the partially de-watered zirconia gel article via e.g., super critical extraction.
If desired, the zirconia aerogel article can be characterized by the following features alone or in combination:
(a) comprising crystalline zirconia particles having an average primary particle size in a range of 2 nm to 50 nm or 2 nm to 30 nm or 2 to 20 nm;
(b) content of crystalline zirconia particles: at least 85 mol%;
(c) having an organic content of at least 3 wt.% or within a range of 3 to 40 wt.%;
(d) length of two of the x, y, z dimensions: at least 1 or at least 2 or at least 3 mm;
(e) density: 0.5 to 1.5 g/cm3.
A combination of the features (a) and (b), or (a), (b) and (c), or (b), (d) and € can be preferred.
If applied, the extraction step can be characterized by the following features alone or in combination:
(a) temperature: 20 to 100°C or 30 to 80°C or 15 to 150°C;
(b) pressure: 5 to 200 MPa or 10 to 100 MPa or 1 to 20 MPa or 5 to 15 MPa;
(c) duration: 2 to 175 h or 5 to 25 h or 1 to 5 h;
(d) extraction or drying medium: carbon dioxide in its supercritical stage.
A combination of features (a), (b) and (d) is sometimes preferred.
With conducting a supercritical extraction step most of the liquid in the printed gel article can be removed.
In some embodiments, the aerogels contain some residual organic liquid. The residual liquid can be up to 6 wt.% based on the total weight of the aerogel article. E.g., the aerogel article can contain up to 5 wt.%, up to 4 wt.%, up to 3 wt.%, up to 2 wt.%, or up to 1 wt.% organic liquid.
The removal of organic liquid results in the formation of pores within the dried structure. Preferably, the pores are sufficiently large to allow gases from the decomposition products of the polymeric material to escape without cracking the structure when the dried structure is further heated to burnout the organic material and to form a sintered article.
For producing the dental restoration, the process further comprises one or more heat -treatment steps.
Depending on the heat-treatment conditions the heat-treatment may yield a calcined dental zirconia article, or a pre-sintered dental zirconia article, or a sintered dental zirconia article.
The heat treatment can be conducted in one or more steps and at different locations.
For obtaining a calcined dental zirconia article, a heating step is conducted to remove organic residues which are still present in the zirconia article before final sintering. Removing organic residues before sintering reduces the risks of cracks during sintering.
The heating step is typically conducted at a temperature below 800°C or below 700°C or below 600°C. A typical temperature range is from 400 to 800°C or from 500 to 700°C.
Such a heating step is typically conducted for a time needed to combust the organic components in the zirconia article. A typical time frame is from 5 to 100 h or from 10 to 50 h.
For obtaining a pre-sintered dental zirconia article the following conditions are typically applied alone or in combination: (i) temperature: 900 to l,100°C or 950 to l,090°C or 975 to l,080°C; (ii) atmosphere: air or inert gas (e.g., nitrogen, argon); (iii) dwell time: 0 to 24h, or 0.1 to 5h; (iv) duration: until a density of 40 to 60% of the density of the sintered material has been reached.
A combination of conditions (i) and (ii); or (i) and (iii); or (i) and (iv); or (i) and (ii) and (iv) is sometimes preferred.
The dwell time (that is the time during which the aerogel article is kept at that temperature) is helpful as well to tune strength and/or hardness to the specific needs of the chosen machining technology. If the dwell time is too long, the dental article may become too hard to be machined under reasonable conditions, if desired.
For obtaining a sintered dental zirconia article the following conditions are typically applied: (i) temperature: 1,100 to l,350°C or 1,200 to l,300°C; (ii) atmosphere: air or inert gas (e.g., nitrogen, argon); (iii) duration: until the porosity of the zirconia article is less than 0.1 or less than 0.05 vol.%; (iv) dwell time: 1 to 24 h or 2 to 12 h; (v) pressure: ambient pressure.
A combination of conditions (i) and (iii); or (i) and (v); or (i), (iii) and (v) is sometimes preferred.
It is, however, also possible to provide a pre-sintered dental zirconia article first which is later sintered to a sintered dental zirconia article. If desired, this can be done at a different location.
During the heating step(s) the porous dental ceramic article is sintered to its final shape, thereby undergoing changes with regard to dimension, density, hardness, bending strength and/or grain size.
The heating temperature and dwell time (that is, the time period during which a particular temperature is kept) are typically correlated. A higher temperature typically requires only a short dwell time.
Depending on the heating rate which is applied, the heat-treatment steps can be characterized as regular firing conditions (e.g., heating rate of 10 to 30°C/min up to at least l,200°C) or speed-firing conditions (e.g., heating rate of 60 to 350°C/min up to at least l,200°C).
However, due to the use of nano-sized zirconia materials, the temperature needed for obtaining a fully-sintered article is usually not more than 1 ,350°C, sometimes not more than 1 ,300°C.
Thus, the dental restoration described in the present text can be produced more economically as less heat is needed for sintering.
If desired, an extraction step can be conducted in addition.
Such an extraction step may help to remove or exchange ions from the article, such as sulfate ions which might be present at a certain stage of the process.
E.g., a pre-sintered zirconia article can be soaked in a basic solution such as an aqueous solution of ammonium hydroxide.
After soaking, the pre-sintered zirconia article is removed from the solution and washed thoroughly with water. The article can be soaked in water for any desired period of time such as at least 30 min, at least 1 h, at least 2 h, or at least 4 h. The soaking in water can be repeated several times, if desired, by replacing the water with fresh water.
After soaking, the article is typically dried in an oven to remove the water. For example, the article can be dried by heating in an oven set at a temperature equal to at least 80°C, at least 90°C, or at least 100°C. For example, the temperature can be in a range of 80°C to 150°C, 90°C to 150°C, or 90°C to 125 °C for at least 30 min, at least 60 min, or at least 120 min.
An ion-exchange step can typically be characterized by the following features alone or in combination: duration: 5 to 24 hrs; medium: aqueous ammonium hydroxide liquid (e.g., concentration IN); and/or temperature: 20 to 25°C.
The invention is also directed to a zirconia ceramic dental restoration, in particular to a monolithic zirconia ceramic dental restoration, which can be obtained or is obtainable from the customized zirconia gel article.
Like the customized zirconia gel article, the zirconia ceramic dental restoration has typically the shape of a dental crown, bridge, inlay, onlay, or veneer.
That is, the dental restoration has a shape which allows its use by the practitioner without the need for further adjustments or modifications. In particular, there is no need for an additional glazing and/or milling step.
A front tooth has an outer or labial surface. The outer surface of a front tooth corresponds to the outer surface of a veneer.
A dental restoration like a veneer has also an inner surface which corresponds to the outer surface of a prepared tooth in labial direction.
Correspondingly, the zirconia ceramic dental restoration described in the present text comprises an outer surface area and an inner surface area.
The outer surface of the outer surface area, or the inner surface of the inner surface area, or both surfaces may comprise a section having a shape which is based on information associated with the dental situation of a patient.
Both, the outer surface area and the inner surface area comprise ceramic components and stabilizing components.
In addition, at least one region of the inner surface area comprises coloring components or a different concentration of stabilizing components.
If desired, at least one region of the outer surface area may comprise coloring components or a different concentration of stabilizing components, as well.
The material of the inner surface area of the zirconia ceramic dental restoration is typically less light-transmissive than the outer surface area in at least one region of the inner surface area. It was found that this arrangement can more closely mimic the natural tooth layers of a relatively opaque dentin core covered by a relatively translucent enamel layer.
In one aspect, the material of the inner surface area of the zirconia ceramic dental restoration is less light-transmissive than the material of the outer surface area in at most three individual regions of the inner surface area.
Thus, there is no need for the whole material of the inner surface area to be colored. The coloring of the material of the inner surface area is typically done only in those regions which are desired.
If desired, a difference in light-transmission can be determined or analysed with a microscope on a cross-section of the respective area of the zirconia ceramic dental restoration.
If desired, the light transmittance can also be assessed by comparing the respective contrast ratio reflectance (CR-R) as further outlined in the example section.
Contrast ratio is related to light transmission. Contrast ration is the ratio of luminence observed when a test article of a prescribed thickness is measured over a black background vs. the same article measured over a white background. An article with high light transmittance will change in luminance with a different background, an article with low transmittance will change less.
The inner surface area and the outer surface area are contacting each other at an interface area or transition area, which is typically a non-planar.
Thus, there is an interface area or transition zone where the material compositions of the inner surface area and the outer surface area overlap with each other.
Such a transition zone can be advantageous as it allows for a smoother transition of the two material compositions of the inner surface area and the outer surface area.
Depending on the radiation-curing conditions and/or and sequence of steps and/or the viscosity of the sols which are used, the range and material composition of the interface can be modified and adjusted.
The content of coloring components in the interface area or transition zone is typically lower than the content of the coloring components in the inner surface area.
If desired, the difference in content of coloring components can be determined on a sample cut from the zirconia ceramic dental restoration to be analyzed by XRF technology.
The inner surface area and/or the outer surface area typically have a thickness in the range of 0.02 to 3 mm or 0.05 to 2 mm.
The material of the zirconia ceramic dental restoration comprises ceramic components, stabilizing components and coloring components.
The main ceramic component is zirconia. Besides zirconia, hafnia is typically present as well, but in smaller amounts.
Alumina is typically not present and/or has not been wilfully added.
Crystal phase stabilizing components which can be present include oxides of Ce, Mg, Ca, La and Y, wherein the use of Y is sometimes preferred.
Crystal phase stabilizing components calculated as oxides are typically present in the following amounts: at least 1, 2 or 3 mol%; at most 7, 6 or 5 mol%; from 1 to 7 mol%, or 2 to 6, or 3 to 5 mol%; mol% with respect to the zirconia ceramic dental restoration.
Coloring components which are present in the zirconia ceramic dental restoration include oxides selected from Fe, Mn, Er, Pr, Tb, Cr, Co, Mo, Nb and mixtures thereof, wherein the combination of Fe, Mn, Er and Pr is sometime preferred.
The material of the zirconia ceramic dental restoration may comprise or essentially consist of or consist of the following components:
ZrC>2 in an amount of 70 to 98 mol%;
HfCF in an amount of 0 to 2 mol%;
Y2O3 in an amount of 2 to 7 mol%;
Coloring components selected from oxides of Fe, Mn, Er, Pr, Tb, Cr, Co, Mo, Nb or mixtures thereof in an amount of 0.001 to 1 mol%, mol% with respect to the zirconia ceramic dental restoration.
According to a further embodiment, the material of the zirconia ceramic dental restoration may comprise or essentially consist of or consist of the following components:
Z1O2 in an amount of 90 to 98 mol%,
HfCF in an amount of 0 to 2 mol%,
Y2O3 in an amount of 3 to 6 mol%, coloring components selected from oxides of Fe, Mn, Er, Pr, Tb, Cr, Co, Mo, Nb or mixtures thereof in an amount of 0.01 to 0.8 mol%, mol% with respect to the zirconia ceramic dental restoration.
It was found that a higher Y2O3 content typically leads to an increase of the cubic crystal phase in the zirconia ceramic material after having sintered the material to final density. A higher content of the cubic crystal phase may contribute to a better translucency.
The material of the zirconia ceramic dental restoration typically has a tetragonal crystal phase content of at least 40 vol.% or at least 45 vol.% or at least 50 vol.%.
A high tetragonal crystal phase content typically goes along with a high strength of the material. Surprisingly it was found that in its sintered stage the nano-sized zirconia materials described in the present text may have a rather high tetragonal phase content even if the material has a rather high content of stabilizing components (e.g., above 4, 5 or 6 mol% with respect to the zirconia ceramic dental restoration).
Further, the material of the zirconia ceramic dental restoration has an average grain size of 80 to 300 nm for a material with a content of stabilizing components in a range of 3 to 5.0 mol%, or 150 to 550 nm for a material with a content of stabilizing components in the range of 5.1 to 8 mol%, mol% with respect to the zirconia ceramic dental restoration.
The material of the zirconia ceramic dental restoration is to be regarded as a very fine grained material as it results from the processing of compositions comprising nano-sized components.
Due to the process described in the present text, the grain size of the zirconia ceramic body is significantly smaller than the grain size of a zirconia material with the same content of stabilizer but made by compacting a respective powder and sintering the compact at higher temperatures.
It was found that a zirconia material with such an average grain size has advantageous optical properties, in particular as regards transmission.
Further, if desired the zirconia ceramic dental restoration can in addition be characterized by the following features alone or in combination: a) porosity: less than 0.1 vol.%;
b) thickness between outer and inner surface: 0.5 to 0.02 mm at at least one section of the monolithic zirconia ceramic dental restoration; c) translucency: at least 40 % for at least one region of the monolithic zirconia ceramic dental restoration, if determined on a sample having a thickness of 1 mm with a light having a wavelength in the range of 360 to 780 nm; d) opalescence: at least 9 determined on a sample having a thickness of 1 mm; e) contrast ratio differing from each other by at least 5% in two sections of the monolithic zirconia ceramic dental restoration normalized to a thickness of 1 mm and spaced apart from each other by 3 mm.
A combination of the following features is sometimes preferred: b) and c); c) and d); d) and e); c), d) and e). If desired, these properties can be determined as described in the example section.
A material with a porosity of less than 0.1 vol.% or less than 0.05 vol.% is considered as dense.
The zirconia ceramic dental restoration and related process described in the present text has or allows the manufacturing of ceramic articles with very thin sections. It was found that a dental restoration with such a thin and filigree structure cannot be produced by other means, such as milling or 3d-printing.
A section is to be understood as a volume that is distinguishable and that typically corresponds to an anatomical feature (i.e. dental situation) on a tooth. Mamelons are an example. Three mamelon features typically span the width of a tooth, so they have visible features that range in size from tens to hundreds of microns. Sections are visually distinguishable when viewed by a dentally- trained eye without supplemental magnification.
A section of a zirconia ceramic dental restoration is typically to be understood to have a size in one, two or three dimensions of at least 100 or 150 or 200 pm.
Zirconia ceramic dental restoration with such thin section cannot be efficiently produced by milling.
The zirconia ceramic dental restoration is highly translucent at at least one region of the dental restoration, e.g., the occlusal or incisal region.
It was also found that the opalescence of the material of the dental restoration is in a region which basically corresponds to the opalescence of natural teeth.
Further, the manufacturing process described in the present text allows the production of customized zirconia ceramic dental restorations where the contrast ratio of different sections of the restoration may differ already over a small distance.
The process described in the present text does typically not require the following steps alone or in combination: a zirconia powder compacting step; a milling step; a step of applying a coloring liquid; and/or a step of applying a glazing layer.
Thus, there is no need for conducting a step of compacting powders, in particular zirconia powders, to obtain a dental milling block which is later machined.
As the dental restoration is provided by a casting step, there is also no need for conducing a milling step, that is a step where a dental milling block is machined in a milling device.
As the zirconia ceramic dental restoration already contains coloring components, there is also no need for applying a coloring liquid later.
Due to the presence of a translucent outer surface area, there is also no need for applying a glazing layer on the zirconia ceramic dental restoration.
Further embodiments or aspects relating to the customized zirconia gel article, the dental restoration, and the processes described in the present text are given below.
Embodiment 1
A process for producing a dental restoration, the process comprising, essentially consisting of, or consisting of the following steps:
(a) placing a curable sol Si in an amount Ai into a mould with a non-planar inner bottom surface,
(b) placing a curable sol S 2 in an amount A2 in contact with curable sol Si,
(c) optionally placing curable sols SN in amounts AN in contact with either sol Si and/or sol S2 with N = 1, 2, 3, 4 ... 10;
(d) optionally, applying a second mould on top of sol S2 and/or optional sols SN,
(e) conducting a radiation-curing step for obtaining a zirconia gel article,
(f) optionally conducting a post-curing step (e.g., by heating the zirconia gel article to a temperature in the range of 35 to 80 °C or by lightcuring), in particular for the purpose of increasing the stability of the zirconia gel article,
(g) optionally soaking the zirconia gel article with another liquid (e.g., diethylene glycol ethyl ether or ethanol), in particular for exchanging the liquid,
(h) transforming the zirconia gel article to a zirconia aerogel article, preferably by applying a supercritical drying step, in particular for the purpose of removing liquid,
(i) heat-treating the zirconia aerogel article to a temperature in the range of 400 to 800°C, in particular for the purpose of removing residual organic components form the zirconia aerogel article,
(j) heat-treating the 3-dim article of the previous step to a temperature in the range of 800 to l,050°C, in particular for the purpose of obtaining a pre-sintered article,
(k) optionally conducting an extraction step, in particular for the purpose of removing ions from the pre-sintered article,
(l) sintering the pre-sintered article to obtain a sintered zirconia ceramic article, sol Si, sol S2 and optional sols SN each comprising crystalline zirconia particles, a crystal phase stabilizer component, a radiation-curable organic component,
a photo-initiator, a liquid, and wherein at least one sol contains coloring components, wherein the crystal phase stabilizer component is contained in the crystalline zirconia particles and the coloring component, if present, can be contained in the crystalline zirconia particles or be present as a separate component in the sol, wherein the viscosity of each sol is in a range of 50 to 3,000 mPa*s at 23°C; and wherein sol Si, sol S2 and optional sols SN differ from each other with respect to the following properties alone or in combination: content of coloring components, content of crystal phase stabilizing components, content of crystalline zirconia particles.
Embodiment 2
A dental restoration, in particular obtainable or obtained by the process described in the present text, the dental ceramic zirconia article being characterized by the following properties: thickness between outer and inner surface in at least one section: 0.5 to 0.02 mm, difference in optical properties of at least two sections regarding colour and/or translucency: DE > 2 and/or %CR > 2, determined on individually colored 1 mm thick samples, opalescence: at least 10,
Z1 O2 content: 90 to 98 mol%,
HfCE content: 0 to 2 mol%,
Y2O3 content: 3 to 6.5 mol%,
AI2O3 content: 0 to 0.1 mol%, mol.% with respect to the dental ceramic zirconia article.
Embodiment 3
A customized zirconia gel article having the shape of a dental restoration, the customized zirconia gel article comprising at least 3 areas, an inner surface area comprising gel Gl, an outer surface area comprising gel G2, and a transition area comprising transition gel G1/2, the material compositions of the gels each comprising crystalline zirconia particles, a crystal phase stabilizer component, a cured organic component,
optionally a photo-initiator, a liquid, and wherein at least one of the gels comprise a color component, wherein the crystal phase stabilizer component is contained in the crystalline zirconia particles and the coloring component, if present, can be contained in the crystalline zirconia particles or be present as a separate component in the gel, wherein gel Gi is different from gel G2, wherein the transition area has a color gradient, and wherein the transition gel comprises a mixture of the material compositions of gels Gi and G2.
The components, elements or features mentioned in these embodiments correspond to the components, elements or features described in the present text.
The complete disclosures of the patents, patent documents, and publications cited herein are incorporated by reference in their entirety as if each were individually incorporated. Various modifications and alterations to this invention will become apparent to those skilled in the art without departing from the scope and spirit of this invention. The above specification, examples and data provide a description of the manufacture and use of the compositions and methods of the invention. The invention is not limited to the embodiments disclosed herein. One skilled in the art will appreciate that many alternative embodiments of the invention can be made without departing from the spirit and scope of thereof.
Examples
Objects and advantages of this disclosure are further illustrated by the following examples. The particular materials and amounts recited in these examples, as well as other conditions and details, should not be construed to unduly limit this disclosure.
Unless otherwise indicated, all parts and percentages are on a weight basis, all water is de -ionized water, and all molecular weights are weight average molecular weight. Moreover, unless otherwise indicated all experiments were conducted at ambient conditions (23°C; 1013 mbar).
Methods
Method for Crystalline Structure and Size (XRD Analysis)
Dried zirconia samples were ground by hand using an agate mortar and pestle. A liberal amount of the sample was applied by spatula to a glass microscope slide on which a section of double-sided adhesive tape had been adhered. The sample was pressed into the adhesive on the tape by forcing the sample against the adhesive with the spatula blade. Excess sample was removed by scraping the sample area with the edge of the spatula blade, leaving a thin layer of particles adhered to the adhesive. Loosely adhered materials remaining after the scraping were removed by forcefully tapping the microscope slide against a hard surface. In a similar manner, corundum (Linde 1.0 pm
alumina polishing powder, Lot Number C062, Union Carbide, Indianapolis, IN) was prepared and used to calibrate the X-ray diffractometer for instrumental broadening.
X-ray diffraction scans were obtained using a Philips vertical diffractometer having a reflection geometry, copper Ka radiation, and a proportional detector registry of the scattered radiation. The diffractometer was fitted with variable incident beam slits, fixed diffracted beam slits, and a graphite diffracted beam monochromator. The survey scan was recorded from 25 to 55 degrees two theta (2q) using a step size of 0.04 degrees and a dwell time of 8 seconds. X-ray generator settings of 45 kV and 35 mA were used. Data for the corundum standard was collected on three separate areas of several individual corundum mounts. Likewise, data was collected on three separate areas of the thin layer sample mount.
The observed diffraction peaks were identified by comparison to reference diffraction patterns contained within the International Center for Diffraction Data (ICDD) powder diffraction database (sets 1-47, ICDD, Newton Square, PA, USA). The diffraction peaks for the samples were attributed to either cubic/tetragonal (C/T) or monoclinic (M) forms of zirconia. For zirconia-based particles, the (111) peak for the cubic phase and (101) peak for the tetragonal phase could not be separated so these phases were reported together. The amounts of each zirconia form were evaluated on a relative basis and the form of zirconia having the most intense diffraction peak was assigned the relative intensity value of 100. The strongest line of the remaining crystalline zirconia form was scaled relative to the most intense line and given a value between 1 and 100.
Peak widths for the observed diffraction maxima due to corundum were measured by profile fitting. The relationship between mean corundum peak widths and corundum peak position (2q) was determined by fitting a polynomial to these data to produce a continuous function used to evaluate the instrumental breadth at any peak position within the corundum testing range. Peak widths for the observed diffraction maxima due to zirconia were measured by profile fitting the observed diffraction peaks. The following peak widths were evaluated depending on the zirconia phase found to be present:
Cubic/Tetragonal (C/T): (1 1 1) Monoclinic (M): (-1 1 1), and (l 1 1)
A Pearson VII peak shape model with Kai and Ka2 wavelength components and linear background model were used for all measurements. Widths were calculated as the peak full width at half maximum (FWHM) having units of degrees. The profile fitting was accomplished by use of the capabilities of the JADE diffraction software suite. Sample peak widths were evaluated for the three separate data collections obtained for the same thin layer sample mount.
Sample peaks were corrected for instrumental broadening by interpolation of instrumental breadth values from corundum instrument calibration and corrected peak widths converted to units of radians. The Scherrer equation was used to calculate the primary crystal size.
Crystallite Size (D) = Kl/b (cos q)
In the Scherrer equation, K is the form factor (here 0.9), 1 is the wavelength (1.540598 A), b is the calculated peak width after correction for instrumental broadening (in radians), and q equals half the peak position (scattering angle), b is equal to [calculated peak FWHM - instrumental breadth] (converted to radians) where FWHM is full width at half maximum. The cubic/tetragonal (C/T) mean crystallite size was measured as the average of three measurements using (1 1 1) peak. That is,
C/T mean Crystallite Size = [D(l 1 1) are l + D(1 1 1) area2 + D(1 1 1) areas] / 3.
Method for Photon Correlation Spectroscopy (PCS)
Particle size measurements were made using a light scattering particle sizer equipped with a red laser having a 633 nm wavelength of light (obtained under the trade designation “ZETA SIZER - Nano Series, Model ZEN3600” from Malvern Instruments Inc., Westborough, MA). Each sample was analyzed in a one centimeter square polystyrene sample cuvette. The sample cuvette was filled with about 1 gram of deionized water, and then a few drops (about 0.1 gram) of the zirconia-based sol were added. The composition (e.g., sample) within each sample cuvette was mixed by drawing the composition into a clean pipette and discharging the composition back into the sample cuvette several times. The sample cuvette was then placed in the instrument and equilibrated at 25°C. The instrument parameters were set as follows: dispersant refractive index 1.330, dispersant viscosity 0.8872 MPa-second, material refractive index 2.10, and material absorption value 0.10 units. The automatic size-measurement procedure was then run. The instrument automatically adjusted the laser-beam position and attenuator setting to obtain the best measurement of particle size.
The light scattering particle sizer illuminated the sample with a laser and analyzed the intensity fluctuations of the light scattered from the particles at an angle of 173 degrees. The method of Photon Correlation Spectroscopy (PCS) was used by the instrument to calculate the particle size. PCS uses the fluctuating light intensity to measure Brownian motion of the particles in the liquid. The particle size is then calculated to be the diameter of sphere that moves at the measured speed. The intensity of the light scattered by the particle is proportional to the sixth power of the particle diameter. The Z-average size or cumulant mean is a mean calculated from the intensity distribution and the calculation is based on assumptions that the particles are mono-modal, mono-disperse, and spherical. Related functions calculated from the fluctuating light intensity are the Intensity Distribution and its mean. The mean of the Intensity Distribution is calculated based on the assumption that the particles are spherical. Both the Z-average size and the Intensity Distribution mean are more sensitive to larger particles than smaller ones.
The Volume Distribution gives the percentage of the total volume of particles corresponding to particles in a given size range. The volume-average size is the size of a particle that corresponds to the mean of the Volume Distribution. Since the volume of a particle is proportional to the third
power of the diameter, this distribution is less sensitive to larger particles than the Z-average size. Thus, the volume-average will typically be a smaller value than the Z-average size.
Method for Measuring Oxide Content
The oxide content was measured via thermal gravimetric analysis (obtained under the trade designation “TGA Q500” from TA Instruments, New Castle, DE, USA). The sample (about 50 mg) was loaded into the TGA and the temperature was taken to 900°C in air. The oxide content of the sample was equal to the residual weight after heating to 900°C.
Method for Measuring Archimedes Density
The density of the sintered material was measured by the Archimedes technique. The measurements were made on a precision balance (identified as “XSE204” from Mettler-Toledo, LLC, Columbus, OH, USA) using a density determination kit (identified as “Density Determination Kit for Excellence XP/XS Analytical Balances” from Mettler-Toledo, LLC, Columbus, OH, USA). The sample was first weighed in air (A), then immersed in water and weighed (B). The water was distilled and deionized. Three drops of a wetting agent (obtained under trade designation “PERVITRO 75%” from Mettler-Toledo, LLC, Columbus, OH, USA) was added to 250 ml of water. The density was calculated using the balance density function, which used the formula p = (A/(A-B)) (PO-PL) + PL, where po is the density of water and PL is the density of air (0.0012 g/cm3). The relative density can be calculated by reference to the theoretical density (pt) of the material, prei = (p/pt)*100.
Method for Measuring Contrast Ratio Reflectance (CR-R) and DE
CR-R value is the opacity using the contrast ratio method, and _E is the color difference. Recording was done using a Photospectrometer Color i7 (X-Rite Corp.) with an optical configuration of d/8° using D65 calibrated illumination, where a spectral range between 360 and 750nm with lOnm wavelength interval was recorded. For this measurement reflectance mode with 10mm aperture was used. Color evaluation was configured with 10° observer, exclusion of specular and inclusion of UV irradiation. For these measurements, fully sintered ceramic test pieces of approximately 1 mm thickness were used. The samples were measured using both a light backing and a dark backing. L*, a* and b* values are automatically calculated from the spectra by the X- Rite software. The calculation of CR-R is also done automatically by the software, using a formula at the ratio of dark backing to light backing measurement. CR-R is expressed as a percentage. The higher the level of CR-R, the more opaque the material is, and the lower the level of CR-R, the more translucent the material is.
Viscosity
If desired, viscosity can be measured using a capillary viscosity method. Fluid is placed in a cylinder of a diameter D of 10 mm, a piston is moved at a constant speed v of 2.9 mm/min in order to press the sol through a die of a length L of 38.1 mm and a diameter d of 0.58 mm. The force F is measured to move a piston at constant speed, calibrated to piston friction. A standard equipment, Zwick, Ulm, was used to perform these measurements.
Viscosity r| = r/y
Shear Rate y = 4*Vdot/7t*d3
Flowrate V<|<1|= Ap*v = v * 7i * D2 / 4
T = F / (4*(L/d)) pH value
If desired, the pH value of can be determined as follows: 1.0 g of a component is dispersed in 10 ml de-ionized water and stirred for about 5 min. A calibrated pH electrode is dipped into the suspension and the pH value is determined during stirring.
Elemental Composition
If desired, the elemental composition can be determined by X-ray fluorescence spectrometry (XRF), e.g., with the ZSX Primus II from Rigaku, Japan. This method is especially suited for the analysis of solids, e.g., zirconia ceramics or glass materials.
Opalescence
An article is considered opalescent, if the calculated opalescent value OP meets the following condition:
OP = [(ClEaT* - ClEaR*)2 + (ClEbT* - CIEbR*)2]1/2 = at least 10, or 10 to 40 or 15 to 40, wherein (CIEar* - CIEaR*) is the difference between transmission and reflectance modes in red- green coordinate a*, and (CIEbi* - CIEbR*) is the difference between transmission and reflectance modes in yellow-blue color coordinate b*.
The OP value can be determined with a sphere benchtop spectral photometer Color i7800 (x-rite, Michigan USA), applying the formula described above using samples of 1.0 mm in height and 15 mm in diameter.
Average Grain Size
Average Grain Size can be determined according to ASTM El 12 using the planimetric intercept procedure to determine f . The average grain size is the average value of the grain size of 10 lines.
The average grain size of a line is the line length in a SEM picture divided by the number of grain boundary intercept multiplied by the shape factor 1.56.
Porosity of Zirconia Article The porosity of a zirconia article can be determined from the water uptake after soaking the article in water. The water uptake is the weight of the soaked article divided by the weight of the dry article. The volume of the porosity is calculated from the water uptake and the density of the water at the measure temperature. Materials
Sol Preparation
Zirconia-based sols Sol-I(a)-Sol-III(a) were prepared as described in WO 2016/191534 Al (Examples Section - Processing: Preparation of Sol-Si) except that the feed composition was varied. Yttrium acetate, iron acetate and erbium acetate were added to the precursor solution in appropriate amounts. The target compositions of zirconia in Sol-I(a)-Sol-III(a) are listed in Table 1 , below.
Sol-I(a)-Sol-II(a) were further processed to increase their concentration and/or remove acetic acid (AcOH). A combination of one or more of ultrafiltration, diafiltration and distillation were used. The diafiltration and ultrafiltration were performed using a dialysis membrane (Membra-Cel™ MD77 14x100 CLR). Distillation was performed using rotary evaporation.
Diethylene glycol monoethyl ether-based sols, Sol-I(b)-Sol-III(b), were prepared from Sol-I(a)- Sol-III(a) by adding 2-[2-(2-methoxyethoxy)ethoxy]acetic acid (MEEAA) (3.3 wt.% with respect to the grams of oxide in the sol) as well as the appropriate amount of diethylene glycol monoethyl ether (adjusted to the intended final oxide concentration in the sol of 53 wt.%) and concentrating each sol via rotary evaporation. 282 g of Sol 1(b), 135 g of Sol 11(b) and 70 g of Sol 111(b) were made.
Preparation of Casting Sols
With respect to the weight of solid zirconia in each Sols (Sol 1(b), Sol 11(b), Sol 111(b)), 9.8 wt.% acrylic acid, 5% HEAA and 0.2 wt.% Irgacure 819 were added and stirred for Ih.
Casting Sols CSol2 was obtained from Sol 11(b) and Casting Sol CSol3 was obtained from Sol 111(b).
In order make a sol providing a bright dental incisal color, portions of 36.5 g of Sol-I(b), 3.2 g of Sol-II(b) and 0.24 g of Sol-III(b), were mixed and sired for 5 min to obtain Casting Sol CSoll. The viscosity was determined to 570 mPa*s using capillary viscosity method.
Method for Sectioned Casting
To cast sectioned objects from ceramic sols, the following procedure was used. A body for an outer tooth shape was made from scan data (.stl) of an anterior tooth by CAD/CAM milling. The size of
the mold needed to be scaled, depending on the shrinkage factor of the complete procedure. The shrinkage factor was determined experimentally by measuring the size of a casted body (e.g., 2.4 cm) and the final sintered body (e.g., 1.3 cm). The shrinkage factor was also calculated from the solid contents of the casting sols. A mold was formed by a vacuum thermoforming process using a BIOSTAR™ VII device (Scheu, Germany) and COPYPLAST® mold material. Caution was taken to prevent light exposure by performing procedures in a UV-filtered room (yellow lights), or in low-light conditions when UV-filtering was not available.
Small amounts of sols were dosed by hand using a pipette or a dosing robot (Vieweg, VR3203) at room temperature.
If desired, a second mould can be placed on top of the casted sol to provide a second, customized outer shape. The shape of the mould may be based on information associated with the dental situation of a patient.
Curing was done by applying UV light (Osram Dulux S Blue 9W/71, Germany) for 15 s to 3 min. After curing, the gel sample was immediately removed from the mold and submerged in diethylene glycol monoethyl ether.
Method for Supercritical Fluid Extraction
The casted gel body was dried via supercritical fluid extraction, e.g., as described in the Method for Supercritical Extraction of Gels in the examples section of WO 2016/191534 Al (Mayr et al).
Method for Burnout and Pre-sinter
The dried gel body was placed on a bed of zirconia beads in an alumina crucible. The crucible was covered with alumina plates and then fired in air according to the following schedule:
1- Heat from 20°C to 170°C at 18°C/hour rate,
2- Heat from 220°C to 244°C at 1 °C/hour rate,
3- Heat from 244°C to 400°C at 6°C/hour rate,
4- Heat from 400°C to l,000°C at 60°C/hour rate,
5- Hold temperature of 1000°C for Ih
6- Cool from l,000°C to 20°C at 120°C/hour rate.
Method for Ion Exchange
The pre-sintered body was placed in a glass jar containing EON NH4OH at a depth of about 2.5 cm and soaked for at least 16 hours. The NH4OH was then poured off and the jar was filled with distilled water. The body was soaked in distilled water for 1 h. The water was then replaced with fresh distilled water. This step was repeated until the pH of the soak water was equal to that of fresh distilled water. The body was then dried at 90- 125 °C for a minimum of 15 min.
Method for Sintering
The pre-sintered, ion-exchanged body was placed in a furnace and sintered in air according to the following schedule:
1- Heat from 25°C to 400°C at 5.7°C/s rate,
2- Heat from 400°C to 1255°C at 0.4°C/s rate,
3- Hold at 1,255°C for 800 s,
4- Cool down from 1,245°C to 700°C at -1.2°C/s rate.
5- Remove from the furnace and perform natural cooling to room temperature
Examples
All samples were casted using a mold in 2.3 cm max. length and 1.8 cm max with. All samples were dried according to the method for supercritical fluid extraction, and subsequently processed according to the method for burnout and pre-sinter, the method for ion exchange, and the method for sintering to form fully dense ceramic articles.
CSol 1 : L* =84. a*= -3, b* = 7, CR = 60
CSol 2: L* =62, a*=12, b* = 28, CR = 80
CSol3: L* = 69, a*=23, b*= - 1, CR = 45
Example 1
Example 1 exhibits a prototype veneer casted with about 1.07 ml of Cast sol CSoll first. Then 3 droplets (about 0.02 ml, 0.06 ml and 0.25 ml) of CSol3 were carefully casted on top of CSoll, resulting in a color difference between the zones. At the interface the sols mixed inherently due to self-diffusion. After that, light curing (about 3 min) was done. After finalization of the process, the body had dimensions of 13 mm max length, 9.5 mm width and 2 mm max. thickness and a weight of 0.74 g. The color difference between the zones was DE = 26, determined from the references. Using XRD, 77% tetragonal and 23% cubic crystallite phases were determined. Using SEM, grain size was determined to 214 nm in the region of CSoll. A picture of the prototype veneer is shown in Fig. 1.
The sol casting process for this prototype veneer is schematically shown in Fig. 2 (1) to (4). Fig. 2 (1) to (4) shows a mold with a customized concave inner surface. Within the mold there is CSol 1 at the bottom (1) and small volumes of Csol 2 are applied on different areas on top of Csoll (2), (3). Shown is also the transition area formed by diffusion and or mixing of Csol 1 and Csol 2 (4).
Example 2 (Ex 2)
Example 2 exhibit a prototype veneer casted with about 0.31 ml of Cast sol Csoll first, on the right incisal side of the mold, about 0.22 ml Cast sol Csol3 at the left incisal side of the mold and about 0.7 ml of Csol2 at the body side of the mold, resulting in a color difference between the three
zones. At the interface the sols mixed inherently due to self-diffusion. A light curing was done (about 2 min). After finalization of the process, the body had dimensions of 13 mm max length, 9.5 mm width and 2 mm max thickness and a weight of 0.66 g. The color difference between the zones was DEI,2 = 26, DE2.3 = 34 and DE1 = 31, determined from the references. Using XRD, 77% tetragonal and 23% cubic crystallite phases were determined. Using SEM, grain size was determined to 300 nm of the region Csol3. A picture of this prototype veneer is shown in Fig. 3.
The sol casting process for this prototype veneer is schematically shown in Fig. 4 (1) to (4). Fig. 4 shows a mold with a customized concave inner surface. Within the mold there is Csol 1 at the bottom (1). Different volumes of Csol 2 (2) and Csol 3 (3) are placed on top of or in contact with Csoll. Shown are also transition areas formed by diffusion and or mixing of Csol 1 with Csol 2 and Csol 3, respectively (4).
Example 3 (Ex 3)
Example 3 exhibits prototype veneers casted into a first mould, with about 0,57 g of Cast sol Csoll first and about 0.71 g of Csol2 at the top side. A second mould was placed to form a second, defined surface. A light curing was done (about 2 min). Second mould was removed and avter that, the veneer was removed from the first mould. After finalization of the process, the body had 1,4 mm max thickness. The color difference between the zones was DE1.2 = 26, determined from the references. A picture of this sample is shown in Fig. 5.
The sol casting process for this prototype veneer is schematically shown in Fig. 6 (1) to (5). Fig. 6 shows a mold with a customized concave inner surface. Within the mold there is Csol 1 at the bottom (1) and Csol 2 is placed on top of Csoll (2). A second mould with a convex outer surface is placed on top of Csol 2 (3) resulting in a mixing of the two sols in the transition area. After curing (4) a customized gel article having the shape of a dental restoration is obtained (5).
Comparative Example 1 (CE1)
CE1 is a veneer milled from a 3M Lava™ Esthetic Fluorescent Full-Contour Zirconia Disc (3M Oral Care) and finalized according to the instruction of use provided by the manufacturer. For this kind of material, the vendor reports an average grain size of approximately 1 pm. The veneer is not further glazed.
Although the material of the disc has a build-in shade gradient, compared to the inventive examples above the appearance of the milled and sintered veneer of CE1 appeared rather homogeneous with respect to its color.
A picture of this sample is shown in Fig. 7
Comparative Example 2 (CE2)
CE2 is a veneer which was 3D-printed from a zirconia nanosol essentially corresponding to Sol- Ila), that is a sol without coloring components. The appearance of the veneer is homogeneous with respect to its color. As 3d-printer an Asiga Max 43 UV device was used. A picture of this sample is shown in Fig. 8.
Claims
1. A customized zirconia gel article having the shape of a dental restoration, the customized zirconia gel article comprising a transition area comprising a transition gel, the transition area having a color gradient, the material composition of the transition gel comprising crystalline zirconia particles, a crystal phase stabilizer component, a cured organic component, optionally a photo-initiator, a liquid, and a coloring component, wherein the crystal phase stabilizer component is contained in the crystalline zirconia particles and the coloring component can be contained in the crystalline zirconia particles or be present as a separate component in the gel.
2. The customized zirconia gel article according to any of the preceding claims, the transition area being non-planar.
3. The customized zirconia gel article according to the preceding claim comprising at least 3 areas, an inner surface area comprising gel Gi, an outer surface area comprising gel G2, and the transition area comprising transition gel G1/2, the geometry of the inner surface, the outer surface or of both, the inner surface and the outer surface of the respective areas being based on information associated with the dental situation of a patient.
4. The customized zirconia gel article according to the preceding claim, the content of coloring components in the transition area being lower than the content of the coloring components in the inner surface area.
5. A process for producing the customized zirconia gel article according to any of claims 1 to 4, the process comprising the steps of
(a) placing a curable sol Si in an amount Ai into a mould with a non-planar inner bottom surface,
(b) placing a curable sol S 2 in an amount A2 in contact with curable sol Si,
(c) optionally placing curable sols SN in amounts AN in contact with either sol Si and/or sol S2 with N = 1, 2, 3, 4 ... ,
(d) conducting a curing step, sol Si, sol S2 and optional sols SN each comprising crystalline zirconia particles, a crystal phase stabilizer component, a curable organic component, a photo-initiator, optionally a coloring component, and a liquid, wherein at least sol S2 contains a coloring component, wherein the crystal phase stabilizer component is contained in the crystalline zirconia particles and the coloring component, if present, is contained in the crystalline zirconia particles or present as a separate component in the sol, wherein the viscosity of each sol is in a range of 50 to 3,000 mPa*s at 23°C as measured according to the description; and wherein sol Si, sol S2 and optional sols SN differ from each other with respect to the following properties alone or in combination: content of coloring components, content of crystal phase stabilizing components, content of crystalline zirconia particles.
6. The process according to claim 5, the non-planar inner bottom surface of the mould comprising a section with a concave region and/or convex region.
7. The process according to any of claims 5 to 6, the non-planar bottom surface of the mould comprising a section with a geometry which is based on information associated with the dental situation of a patient.
8. The process according to any of claims 5 to 7 comprising in addition the following steps before step (a): providing sols with different contents of stabilizing and/or coloring components, mixing at least two of these sols to obtain curable sol Si, curable sol S2, and optionally curable sols SN-
9. The process according to any of claims 5 to 8, wherein the chemical compositions of curable sol Si, curable sol S2, and optionally curable sols SN are adjusted to match with information associated with the dental situation of a patient.
10. The process according to any of claims 5 to 9, wherein amount Ai and amount A2 are different with respect to volume.
11. The process according to any of claims 5 to 10, the placing of the sols being conducted by either of the following methods: casting; inkjet-printing; or gradient printing using vat polymerization.
12. The process according to any of claims 5 to 11 not comprising the following steps alone or in combination: a powder compacting step; a milling step; a step of applying a coloring liquid; and/or a step of applying a glazing layer.
13. A dental restoration obtained or obtainable by the process according to any of claims 5 to 12, comprising in addition the steps of optionally conducting an extracting step, and conducting one or more heat-treatment steps.
14. The dental restoration in particular according to the preceding claim, having an outer surface area and an inner surface area, the zirconia ceramic dental restoration comprising stabilizing components and coloring components, the inner surface area comprising a material having a higher contrast ratio than the material of the outer surface area in at least one region of the inner surface area, the material of the zirconia ceramic dental restoration having a tetragonal crystal phase content of at least 40 vol.%, and an average grain size of
80 to 300 nm for a material with a content of stabilizing components in a range of 3 to 5.0 mol%,
150 to 550 nm for a material with a content of stabilizing components in the range of 5.1 to 8 mol%, vol.% and mol% with respect to the dental restoration, wherein the average grain size is determined according to ASTM El 12 and according to the description.
15. The dental restoration according to any of claims 13 to 14 comprising at least one section having a thickness in the range of 0.5 to 0.02 mm.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23186020 | 2023-07-18 | ||
| EP23186020.6 | 2023-07-18 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025017392A1 true WO2025017392A1 (en) | 2025-01-23 |
Family
ID=87419173
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2024/056101 Pending WO2025017392A1 (en) | 2023-07-18 | 2024-06-21 | Customized zirconia gel article, zirconia dental restoration and process of production |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025017392A1 (en) |
Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5453262A (en) | 1988-12-09 | 1995-09-26 | Battelle Memorial Institute | Continuous process for production of ceramic powders with controlled morphology |
| US5652192A (en) | 1992-07-10 | 1997-07-29 | Battelle Memorial Institute | Catalyst material and method of making |
| WO2009085926A2 (en) | 2007-12-28 | 2009-07-09 | 3M Innovative Properties Company | Method of making zirconia-containing nanoparticles |
| WO2014062375A1 (en) * | 2012-10-17 | 2014-04-24 | 3M Innovative Properties Company | Multi sectional dental zirconia milling block, process of production and use thereof |
| US9039947B2 (en) | 2009-09-30 | 2015-05-26 | 3M Innovative Properties Company | Methods for making layered dental appliances from the outside in |
| WO2016191534A1 (en) | 2015-05-28 | 2016-12-01 | 3M Innovative Properties Company | Sol containing nano zirconia particles for use in additive manufacturing processes for the production of 3-dimensional articles |
| US10532008B2 (en) | 2017-02-22 | 2020-01-14 | James R. Glidewell Dental Ceramics, Inc. | High strength and translucency dental zirconia ceramic materials, devices and methods |
| US10759707B2 (en) | 2015-05-28 | 2020-09-01 | 3M Innovative Properties Company | Additive manufacturing process for producing ceramic articles using a sol containing nano-sized particles |
| EP3936081A1 (en) * | 2019-03-06 | 2022-01-12 | Kuraray Noritake Dental Inc. | Zirconia molded article capable of being fired quickly and calcinated body |
| US20220096215A1 (en) * | 2020-09-30 | 2022-03-31 | Ivoclar Vivadent Ag | Process For The Preparation Of A Dental Shaped Body |
| US20220380260A1 (en) | 2019-08-06 | 2022-12-01 | 3M Innovative Properties Company | Continuous Additive Manufacturing Method for Making Ceramic Articles, and Ceramic Articles |
-
2024
- 2024-06-21 WO PCT/IB2024/056101 patent/WO2025017392A1/en active Pending
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5453262A (en) | 1988-12-09 | 1995-09-26 | Battelle Memorial Institute | Continuous process for production of ceramic powders with controlled morphology |
| US5652192A (en) | 1992-07-10 | 1997-07-29 | Battelle Memorial Institute | Catalyst material and method of making |
| WO2009085926A2 (en) | 2007-12-28 | 2009-07-09 | 3M Innovative Properties Company | Method of making zirconia-containing nanoparticles |
| US9039947B2 (en) | 2009-09-30 | 2015-05-26 | 3M Innovative Properties Company | Methods for making layered dental appliances from the outside in |
| WO2014062375A1 (en) * | 2012-10-17 | 2014-04-24 | 3M Innovative Properties Company | Multi sectional dental zirconia milling block, process of production and use thereof |
| US10028809B2 (en) | 2012-10-17 | 2018-07-24 | 3M Innovative Properties Company | Multi sectional dental zirconia milling block, process of production and use thereof |
| WO2016191534A1 (en) | 2015-05-28 | 2016-12-01 | 3M Innovative Properties Company | Sol containing nano zirconia particles for use in additive manufacturing processes for the production of 3-dimensional articles |
| US10759707B2 (en) | 2015-05-28 | 2020-09-01 | 3M Innovative Properties Company | Additive manufacturing process for producing ceramic articles using a sol containing nano-sized particles |
| US10532008B2 (en) | 2017-02-22 | 2020-01-14 | James R. Glidewell Dental Ceramics, Inc. | High strength and translucency dental zirconia ceramic materials, devices and methods |
| EP3936081A1 (en) * | 2019-03-06 | 2022-01-12 | Kuraray Noritake Dental Inc. | Zirconia molded article capable of being fired quickly and calcinated body |
| US20220380260A1 (en) | 2019-08-06 | 2022-12-01 | 3M Innovative Properties Company | Continuous Additive Manufacturing Method for Making Ceramic Articles, and Ceramic Articles |
| US20220096215A1 (en) * | 2020-09-30 | 2022-03-31 | Ivoclar Vivadent Ag | Process For The Preparation Of A Dental Shaped Body |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3303255B1 (en) | Additive manufacturing process for producing ceramic articles using a sol containing nano-sized particles | |
| EP3303254B1 (en) | Use of a sol containing nano zirconia particles in an additive manufacturing process and a process | |
| EP3583083B1 (en) | Zirconia article with high alumina content, process of production and use thereof | |
| RU2698021C1 (en) | Dental preparation for milling, method of its production and use | |
| CN104717937B (en) | Including the dental blank of pre-sintered porous oxidation zirconia material, its preparation method and the dental articles being formed by described dental blank | |
| US12528743B2 (en) | Continuous additive manufacturing method for making ceramic articles, and ceramic articles | |
| KR102583377B1 (en) | Methods for making gel compositions, shaped gel articles and sintered articles | |
| EP3504017A1 (en) | Coloured curable composition for additive manufacturing processes, 3-dim composite article and use thereof | |
| WO2025017392A1 (en) | Customized zirconia gel article, zirconia dental restoration and process of production | |
| CN121532364A (en) | Customized zirconia gel products, zirconia dental restorations and manufacturing methods | |
| WO2024231752A1 (en) | Process for producing a zirconia dental article | |
| WO2024180438A1 (en) | Ceramic veneers and continuous additive manufacturing method for making ceramic veneers |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24738396 Country of ref document: EP Kind code of ref document: A1 |