WO2025014963A1 - Inhibitors of p97 protein and methods of use - Google Patents
Inhibitors of p97 protein and methods of use Download PDFInfo
- Publication number
- WO2025014963A1 WO2025014963A1 PCT/US2024/037229 US2024037229W WO2025014963A1 WO 2025014963 A1 WO2025014963 A1 WO 2025014963A1 US 2024037229 W US2024037229 W US 2024037229W WO 2025014963 A1 WO2025014963 A1 WO 2025014963A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- alkyl
- hydrogen
- group
- alkylene
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D519/00—Heterocyclic compounds containing more than one system of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring system not provided for in groups C07D453/00 or C07D455/00
Definitions
- the invention relates generally to compounds and compositions useful for preventing and/or treating a disease or condition associated with activity of p97 protein, e.g., cancer or a neurodegenerative disorder.
- the ubiquitin-proteasome system (UPS) and autophagy processes are crucial to the regulation of protein degradation, which maintains proteostasis, or protein homeostasis. Mutations in protein coding sequences and aneuploidy can cause disruptions to protein synthesis due to an increase in damaged or deregulated proteins, which in turn causes stress on the UPS system as the UPS system must clear out the higher frequency of these proteins. Dysregulation of protein homeostasis is often associated with cancer and the development of neurodegenerative disease (Cheng, J. et al. (2016) AGING CELL, 17(5): el280). In cancer cells, this dysregulation of protein homeostasis may occur as a result of dysregulated proliferation, causing the cancer cells to have a greater reliance on protein quality control (PQC) mechanisms to clear increased amounts of damaged or deregulated proteins of the cancer environment.
- PQC protein quality control
- AAA unfoldases are a subset of AAA+ enzymes that assemble as hexamers to unfold substrate proteins.
- p97 also known as Valosin-containing protein, is an AAA unfoldase and plays an essential role in the protein homeostasis regulatory network.
- p97 is known to play a role in lysosomal degradation, but has also been shown to have roles in autophagy, endosomal sorting, regulating protein degradation at the outer mitochondrial membrane, and key chromatin-associated processes.
- p97 has also been shown to be required for the turnover of cytosolic substrates of the UPS (Janiesch, P. C. et al. (2007) NATURE CELL BIOL., 9: 379-390; Cao, K. et al. (2003) CELL, 115: 355-367; Fu, X. et al.
- cancer cells may be particularly dependent on ERAD to clear the higher rate of damaged and dysregulated proteins (Boelens, J. et al. (2007) IN Vivo, 21: 215-226).
- p97 is known to be highly expressed in multiple cancers (Y amamoto, S. et al.
- Described herein are compounds and compositions useful for preventing and/or treating a disease or condition associated with activity of p97 protein, e.g, cancer or a neurodegenerative disorder.
- the present disclosure provides a compound of Formula (I): or a pharmaceutically acceptable salt thereof, wherein the variables R 1 , R 2 , R 3 , R 4 , R 3 , R 6 , R 7 , m and n are as defined herein.
- the present disclosure provides a compound of Formula (II): or a pharmaceutically acceptable salt thereof, wherein R 1 , R 2 , R 3 (including R 8 and L 1 ), R 4 , R 5 , R 6 , R 7 , m and n are as defined herein.
- composition comprising a compound as described herein or a pharmaceutically acceptable salt thereof and a pharmaceutically acceptable excipient.
- a method of treating a disease or condition associated with p97 protein activity in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a compound as described herein or a pharmaceutically acceptable salt thereof or a pharmaceutical composition as described herein.
- the disease or condition associated with p97 protein activity is cancer, e.g., multiple myeloma or acute myeloid leukemia.
- the disease or condition associated with p97 protein activity' is a neurodegenerative disorder, e.g., ALS, Charcot-Marie-Tooth disease type 2Y, and Inclusion body myopathy with early- onset Paget disease and frontotemporal dementia (IBMPFD).
- ALS ALS
- Charcot-Marie-Tooth disease type 2Y Charcot-Marie-Tooth disease type 2Y
- IBMPFD Inclusion body myopathy with early- onset Paget disease and frontotemporal dementia
- the present disclosure provides compounds and compositions useful for preventing and/or treating a disease or condition described herein, e.g., a disease, disorder, or condition associated with activity of p97 protein, e.g.. cancer or a neurodegenerative disorder.
- a disease or condition described herein e.g., a disease, disorder, or condition associated with activity of p97 protein, e.g.. cancer or a neurodegenerative disorder.
- the compounds described herein can comprise one or more asymmetric centers, and thus can exist in various isomeric forms, e.g., enantiomers and/or diastereomers.
- the compounds described herein can be in the form of an individual enantiomer, diastereomer or geometric isomer, or can be in the form of a mixture of stereoisomers, including racemic mixtures and mixtures enriched in one or more stereoisomer.
- Isomers can be isolated from mixtures by methods known to those skilled in the art. including chiral high pressure liquid chromatography (HPLC) and the formation and crystallization of chiral salts; or preferred isomers can be prepared by asymmetric syntheses.
- HPLC high pressure liquid chromatography
- a pure enantiomeric compound is substantially free from other enantiomers or stereoisomers of the compound (i.e.. in enantiomeric excess).
- an “S” form of the compound is substantially free from the “R” form of the compound and is, thus, in enantiomeric excess of the “R” form.
- enantiomerically pure or “pure enantiomer” denotes that the compound comprises more than 75% by weight, more than 80% by weight, more than 85% by weight, more than 90% by weight, more than 91% by weight, more than 92% by weight, more than 93% by weight, more than 94% by weight, more than 95% by weight, more than 96% by weight, more than 97% by weight, more than 98% by weight, more than 98.5% by weight, more than 99% by weight, more than 99.2% by weight, more than 99.5% by weight, more than 99.6% by weight, more than 99.7% by weight, more than 99.8% by weight or more than 99.9% by weight, of the enantiomer.
- the weights are based upon total weight of all enantiomers or stereoisomers of the compound.
- an enantiomerically pure compound can be present with other active or inactive ingredients.
- a pharmaceutical composition comprising enantiomerically pure R-compound can comprise, for example, about 90% excipient and about 10% enantiomerically pure R-compound.
- the enantiomerically pure R-compound in such compositions can, for example, comprise, at least about 95% by weight R-compound and at most about 5% by weight S-compound, by total weight of the compound.
- a pharmaceutical composition comprising enantiomerically pure S-compound can comprise, for example, about 90% excipient and about 10% enantiomerically pure S-compound.
- the enantiomerically pure S-compound in such compositions can, for example, comprise, at least about 95% by weight S-compound and at most about 5% by weight R-compound, by total weight of the compound.
- the active ingredient can be formulated with little or no excipient or carrier.
- Compound described herein may also comprise one or more isotopic substitutions.
- H may be in any isotopic form, including 1 H, 2 H (D or deuterium), and 3 H (T or tritium);
- C may be in any isotopic form, including 12 C, 13 C, and 14 C;
- O may be in any isotopic form, including 16 O and 18 O;
- F may be in any isotopic form, including 18 F and 19 F; and the like.
- C1–6 alkyl is intended to encompass, C1, C2, C3, C4, C 5 , C 6 , C 1–6 , C 1–5 , C 1–4 , C 1–3 , C 1–2 , C 2–6 , C 2–5 , C 2–4 , C 2–3 , C 3–6 , C 3–5 , C 3–4 , C 4–6 , C 4–5 , and C 5–6 alkyl.
- alkyl refers to a radical of a straight–chain or branched saturated hydrocarbon group, e.g., having 1 to 20 carbon atoms (“C 1–20 alkyl”).
- an alkyl group has 1 to 10 carbon atoms (“C1–10 alkyl”). In some embodiments, an alkyl group has 1 to 9 carbon atoms (“C 1–9 alkyl”). In some embodiments, an alkyl group has 1 to 8 carbon atoms (“C1–8 alkyl”). In some embodiments, an alkyl group has 1 to 7 carbon atoms (“C 1–7 alkyl”). In some embodiments, an alkyl group has 1 to 6 carbon atoms (“C 1–6 alkyl”). In some embodiments, an alkyl group has 1 to 5 carbon atoms (“C1–5 alkyl”). In some embodiments, an alkyl group has 1 to 4 carbon atoms (“C 1–4 alkyl”).
- an alkyl group has 1 to 3 carbon atoms (“C1–3 alkyl”). In some embodiments, an alkyl group has 1 to 2 carbon atoms (“C1-2 alkyl”). In some embodiments, an alkyl group has 1 carbon atom (“C 1 alkyl”). Examples of C 1–6 alkyl groups include methyl, ethyl, propyl, isopropyl, butyl, isobutyl, pentyl, hexyl, and the like.
- alkenyl refers to a radical of a straight–chain or branched hydrocarbon group having from 2 to 20 carbon atoms, one or more carbon–carbon double bonds (e.g., 1, 2, 3, or 4 carbon–carbon double bonds), and optionally one or more carbon– carbon triple bonds (e.g., 1, 2, 3, or 4 carbon–carbon triple bonds) (“C2–20 alkenyl”).
- alkynyl refers to a radical of a straight–chain or branched hydrocarbon group having from 2 to 20 carbon atoms, one or more carbon–carbon triple bonds (e.g., 1, 2, 3, or 4 carbon–carbon triple bonds), and optionally one or more carbon– carbon double bonds (e.g., 1, 2, 3, or 4 carbon–carbon double bonds) (“C 2–20 alkynyl”).
- alkylene refers to a divalent radical of an alkyl. When a range or number of carbons is provided for a particular “alkylene” group, it is understood that the range or number refers to the range or number of carbons in the linear carbon divalent chain.
- Alkylene may be substituted or unsubstituted with one or more substituents as described herein.
- heteroalkylene refers to a divalent radical of a heteroalkyl.
- the term “heteroalkyl” as used herein refers to an “alkyl” group in which at least one carbon atom has been replaced with a hetero atom, e.g., O, N, or S atom.
- the heteroalkyl may be, for example, an –O-C1-C10alkyl group, an -C1-C6alkylene-O-C1-C6alkyl group, or a C1-C6 alkylene-OH group.
- the “heteroalkyl” may be 2-10 membered heteroalkyl, indicating that the heteroalkyl contains from 2 to 10 atoms selected from the group consisting of carbon, oxygen, nitrogen, and sulfur.
- aryl refers to a radical of a monocyclic or polycyclic (e.g., bicyclic or tricyclic) 4n+2 aromatic ring system (e.g., having 6, 10, or 14 ⁇ electrons shared in a cyclic array) having 6–14 ring carbon atoms and zero heteroatoms provided in the aromatic ring system (“C6–14 aryl”).
- heteroaryl refers to a radical of a 5–10 membered monocyclic or bicyclic 4n+2 aromatic ring system (e.g., having 6 or 10 electrons shared in a cyclic array) having ring carbon atoms and 1–4 ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from nitrogen, oxygen and sulfur (“5–10 membered heteroaryl”).
- heteroaryl groups that contain one or more nitrogen atoms, the point of attachment can be a carbon or nitrogen atom, as valency permits.
- Heteroaryl bicyclic ring systems can include one or more heteroatoms in one or both rings.
- Exemplary 5–membered heteroaryl groups containing three heteroatoms include, without limitation, triazolyl, oxadiazolyl, and thiadiazolyl.
- Exemplary 5–membered heteroaryl groups containing four heteroatoms include, without limitation, tetrazolyl.
- Exemplary 6–membered heteroaryl groups containing one heteroatom include, without limitation, pyridinyl.
- Exemplary 6–membered heteroaryl groups containing two heteroatoms include, without limitation, pyridazinyl, pyrimidinyl, and pyrazinyl.
- Exemplary 6–membered heteroaryl groups containing three or four heteroatoms include, without limitation, triazinyl and tetrazinyl, respectively.
- Exemplary 7–membered heteroaryl groups containing one heteroatom include, without limitation, azepinyl, oxepinyl, and thiepinyl.
- Exemplary 5,6–bicyclic heteroaryl groups include, without limitation, indolyl, isoindolyl, indazolyl, benzotriazolyl, benzothiophenyl, isobenzothiophenyl, benzofuranyl, benzoisofuranyl, benzimidazolyl, benzoxazolyl, benzisoxazolyl, benzoxadiazolyl, benzthiazolyl, benzisothiazolyl, benzthiadiazolyl, indolizinyl, and purinyl.
- Exemplary 6,6– bicyclic heteroaryl groups include, without limitation, naphthyridinyl, pteridinyl, quinolinyl, isoquinolinyl, cinnolinyl, quinoxalinyl, phthalazinyl, and quinazolinyl.
- Examples of representative heteroaryls include the following: . w y , , , , ; independently hydrogen, C 1 -C 8 alkyl, C 3 -C 10 carbocyclyl, 4-10 membered heterocyclyl, C 6 -C 10 aryl, and 5- 10 membered heteroaryl.
- carbocyclyl or “carbocyclic” refers to a radical of a non– aromatic cyclic hydrocarbon group having from 3 to 10 ring carbon atoms (“C 3–10 carbocyclyl”) and zero heteroatoms in the non–aromatic ring system.
- a carbocyclyl group has 3 to 8 ring carbon atoms (“C 3–8 carbocyclyl”).
- cycloalkyl refers to a monovalent saturated cyclic, bicyclic, or bridged cyclic (e.g., adamantyl) hydrocarbon group of 3-12, 3-8, 4-8, or 4-6 carbons, referred to herein, e.g., as “C 4-8 cycloalkyl,” derived from a cycloalkane.
- exemplary cycloalkyl groups include, but are not limited to, cyclohexanes, cyclopentanes, cyclobutanes and cyclopropanes.
- cycloalkyl groups are optionally substituted at one or more ring positions with, for example, alkanoyl, alkoxy, alkyl, haloalkyl, alkenyl, alkynyl, amido, amidino, amino, aryl, arylalkyl, azido, carbamate, carbonate, carboxy, cyano, cycloalkyl, ester, ether, formyl, halogen, haloalkyl, heteroaryl, heterocyclyl, hydroxyl, imino, ketone, nitro, phosphate, phosphonato, phosphinato, sulfate, sulfide, sulfonamido, sulfonyl or thiocarbonyl.
- Cycloalkyl groups can be fused to other cycloalkyl, aryl, or heterocyclyl groups. In certain embodiments, the cycloalkyl group is not substituted, i.e., it is unsubstituted.
- heterocyclyl or “heterocyclic” refers to a radical of a 3– to 10– membered non–aromatic ring system having ring carbon atoms and 1 to 4 ring heteroatoms, wherein each heteroatom is independently selected from nitrogen, oxygen, sulfur, boron, phosphorus, and silicon (“3–10 membered heterocyclyl”).
- heterocyclyl groups that contain one or more nitrogen atoms, the point of attachment can be a carbon or nitrogen atom, as valency permits.
- a heterocyclyl group can either be monocyclic (“monocyclic heterocyclyl”) or a fused, bridged or spiro ring system such as a bicyclic system (“bicyclic heterocyclyl”), and can be saturated or can be partially unsaturated.
- Heterocyclyl bicyclic ring systems can include one or more heteroatoms in one or both rings.
- Heterocyclyl also includes ring systems wherein the heterocyclyl ring, as defined above, is fused with one or more carbocyclyl or cycloalkyl groups wherein the point of attachment is either on the carbocyclyl, cycloalkyl, or heterocyclyl ring, or ring systems wherein the heterocyclyl ring, as defined above, is fused with one or more aryl or heteroaryl groups, wherein the point of attachment is on the heterocyclyl ring, and in such instances, the number of ring members continue to designate the number of ring members in the heterocyclyl ring system.
- heterocyclylene refers to a divalent radical of a heterocyclyl.
- a heterocyclyl group is a 5–6 membered non–aromatic ring system having ring carbon atoms and 1–4 ring heteroatoms, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur (“5–6 membered heterocyclyl”).
- the 5–6 membered heterocyclyl has 1–3 ring heteroatoms selected from nitrogen, oxygen, and sulfur.
- the 5–6 membered heterocyclyl has 1–2 ring heteroatoms selected from nitrogen, oxygen, and sulfur.
- the 5–6 membered heterocyclyl has one ring heteroatom selected from nitrogen, oxygen, and sulfur.
- Exemplary 3–membered heterocyclyl groups containing one heteroatom include, without limitation, azirdinyl, oxiranyl, thiorenyl.
- Exemplary 4–membered heterocyclyl groups containing one heteroatom include, without limitation, azetidinyl, oxetanyl and thietanyl.
- Exemplary 5–membered heterocyclyl groups containing one heteroatom include, without limitation, tetrahydrofuranyl, dihydrofuranyl, tetrahydrothiophenyl, dihydrothiophenyl, pyrrolidinyl, dihydropyrrolyl and pyrrolyl–2,5–dione.
- Exemplary 5– membered heterocyclyl groups containing two heteroatoms include, without limitation, dioxolanyl, oxasulfuranyl, disulfuranyl, and oxazolidin-2-one.
- Exemplary 5–membered heterocyclyl groups containing three heteroatoms include, without limitation, triazolinyl, oxadiazolinyl, and thiadiazolinyl.
- Exemplary 6–membered heterocyclyl groups containing one heteroatom include, without limitation, piperidinyl, tetrahydropyranyl, dihydropyridinyl, and thianyl.
- Exemplary 6–membered heterocyclyl groups containing two heteroatoms include, without limitation, piperazinyl, morpholinyl, dithianyl, dioxanyl. Exemplary 6– membered heterocyclyl groups containing two heteroatoms include, without limitation, triazinanyl. Exemplary 7–membered heterocyclyl groups containing one heteroatom include, without limitation, azepanyl, oxepanyl and thiepanyl. Exemplary 8–membered heterocyclyl groups containing one heteroatom include, without limitation, azocanyl, oxecanyl and thiocanyl.
- Exemplary 5-membered heterocyclyl groups fused to a C6 aryl ring include, without limitation, indolinyl, isoindolinyl, dihydrobenzofuranyl, dihydrobenzothienyl, benzoxazolinonyl, and the like.
- Exemplary 6-membered heterocyclyl groups fused to an aryl ring include, without limitation, tetrahydroquinolinyl, tetrahydroisoquinolinyl, and the like.
- Hetero when used to describe a compound or a group present on a compound means that one or more carbon atoms in the compound or group have been replaced by a nitrogen, oxygen, or sulfur heteroatom. Hetero may be applied to any of the hydrocarbyl groups described above such as alkyl, e.g., heteroalkyl; carbocyclyl, e.g., heterocyclyl; aryl, e.g,. heteroaryl; and the like having from 1 to 5, and particularly from 1 to 3 heteroatoms.
- halo or “halogen” refers to fluoro (F), chloro (Cl), bromo (Br) and iodo (I).
- substituted means that at least one hydrogen present on a group (e.g., a carbon or nitrogen atom) is replaced with a permissible substituent, e.g., a substituent which upon substitution results in a stable compound, e.g., a compound which does not spontaneously undergo transformation such as by rearrangement, cyclization, elimination, or other reaction.
- a “substituted” group has a substituent at one or more substitutable positions of the group, and when more than one position in any given structure is substituted, the substituent is either the same or different at each position.
- Suitable monovalent substituents on R ⁇ are independently, for example, deuterium, halogen, -(CH 2 ) 0-2 R ⁇ , -(haloR ⁇ ), -(CH 2 ) 0-2 OH, -(CH 2 ) 0-2 OR ⁇ , -(CH2)0-2CH(OR ⁇ )2; -O(haloR ⁇ ), -CN, -N3, -(CH2)0-2C(O)R ⁇ , -(CH2)0-2C(O)OH, -(CH 2 ) 0-2 C(O)OR ⁇ , -(CH 2 ) 0-2 SR ⁇ , -(CH 2 ) 0-2 SH, -(CH 2 ) 0-2 NH 2 , -(CH 2 ) 0-2 NHR ⁇ , -(CH2)
- Suitable divalent substituents that are bound to vicinal substitutable carbons of an “optionally substituted” group include: -O(C(R * )2)2-3O-, where each independent occurrence of R * is selected from hydrogen, C1-6 alkyl, C1-6 alkenyl, C 1-6 alkynyl, and carbocyclyl, which may be substituted as defined below, or an unsubstituted 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur.
- Suitable substituents on the alkyl, alkenyl, alkynyl, or carbocyclyl group of R * include halogen, -R ⁇ , -(haloR ⁇ ), -OH, -OR ⁇ , -O(haloR ⁇ ), -CN, -C(O)OH, -C(O)OR ⁇ , -NH2, -NHR ⁇ , -NR ⁇ 2, or -NO2, where each R ⁇ is unsubstituted or where preceded by “halo” is substituted only with one or more halogens, and is independently C 1-6 alkyl, C 1-6 alkenyl, C1-6 alkynyl, -CH2Ph, -O(CH2)0-1Ph, or a 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur.
- Suitable substituents on a substitutable nitrogen of an “optionally substituted” group include -R ⁇ , -NR ⁇ 2, -C(O)R ⁇ , -C(O)OR ⁇ , -C(O)C(O)R ⁇ , -C(O)CH2C(O)R ⁇ , -S(O) 2 R ⁇ , -S(O) 2 NR ⁇ 2 , -C(S)NR ⁇ 2 , -C(NH)NR ⁇ 2 , or -N(R ⁇ )S(O) 2 R ⁇ ; where each R ⁇ is independently hydrogen, C1-6 alkyl, C1-6 alkenyl, C1-6 alkynyl, which may be substituted as defined below, unsubstituted -OPh, or an unsubstituted 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur
- a substitutable nitrogen can be substituted with four substituents (or have four bonds to less than four substituents) such that the nitrogen atom will be positively charged or its cation, i.e., N + .
- Such substitution of a substitutable nitrogen can be of a ring nitrogen, for example, a ring nitrogen of a heterocyclic group containing nitrogen as a ring atom.
- one of the substituents of a cationic nitrogen atom is a hydroxyl group
- the hydroxyl group can be deprotonated and represented by a negatively charged oxygen atom, i.e., O-.
- a general formula for the substitution can be represented by RN + (O-)(R’)R’’, where R, R’ and R’’ represent carbon or other atoms, groups and/or moieties to which the nitrogen atom is bound.
- Suitable substituents on the alkyl, alkenyl, alkynyl, or carbocyclyl group of R ⁇ are independently, for example, halogen, -R ⁇ , -(haloR ⁇ ), -OH, -OR ⁇ , -O(haloR ⁇ ), -CN, -C(O)OH, -C(O)OR ⁇ , -NH2, -NHR ⁇ , -NR ⁇ 2, or -NO2, where each R ⁇ is unsubstituted or where preceded by “halo” is substituted only with one or more halogens, and is independently C1-6 alkyl,C1-6 alkenyl, C1-6 alkynyl, -
- compositions specifying a percentage are by weight unless otherwise specified. Further, if a variable is not accompanied by a definition, then the previous definition of the variable controls.
- pharmaceutically acceptable refers to those compounds, materials, compositions, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio.
- pharmaceutically acceptable carrier and “pharmaceutically acceptable excipient” are used interchangeably herein and refer to buffers, carriers, and excipients suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio.
- Pharmaceutically acceptable carriers include any of the standard pharmaceutical carriers, such as a phosphate buffered saline solution, water, emulsions (e.g., such as an oil/water or water/oil emulsions), and various types of wetting agents.
- the compositions also can include stabilizers and preservatives.
- carriers, stabilizers, and adjuvants see e.g., Adeboye Adejare, Remington: The Science and Practice of Pharmacy (23d ed.2020).
- pharmaceutically acceptable salt refers to those salts which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of humans and lower animals without undue toxicity, irritation, allergic response and the like, and are commensurate with a reasonable benefit/risk ratio.
- Pharmaceutically acceptable salts are well known in the art. For example, Berge et al., describes pharmaceutically acceptable salts in detail in J. PHARMACEUTICAL SCIENCES (1977) 66:1–19, and Gould, Salt selection for basic drugs, INTERNATIONAL JOURNAL OF PHARMACEUTICS, 33 (1986) 201-217.
- Pharmaceutically acceptable salts of the compounds of this invention include those derived from suitable inorganic and organic acids and bases.
- Examples of pharmaceutically acceptable, nontoxic acid addition salts are salts of an amino group formed with inorganic acids such as hydrochloric acid, hydrobromic acid, phosphoric acid, sulfuric acid and perchloric acid or with organic acids such as acetic acid, oxalic acid, maleic acid, tartaric acid, citric acid, succinic acid or malonic acid or by using other methods used in the art such as ion exchange.
- inorganic acids such as hydrochloric acid, hydrobromic acid, phosphoric acid, sulfuric acid and perchloric acid
- organic acids such as acetic acid, oxalic acid, maleic acid, tartaric acid, citric acid, succinic acid or malonic acid or by using other methods used in the art such as ion exchange.
- salts include adipate, alginate, ascorbate, aspartate, benzenesulfonate, benzoate, bisulfate, borate, butyrate, camphorate, camphorsulfonate, citrate, cyclopentanepropionate, digluconate, dodecylsulfate, ethanesulfonate, formate, fumarate, glucoheptonate, glycerophosphate, gluconate, hemisulfate, heptanoate, hexanoate, hydroiodide, 2–hydroxy–ethanesulfonate, lactobionate, lactate, laurate, lauryl sulfate, malate, maleate, malonate, methanesulfonate, 2–naphthalenesulfonate, nicotinate, nitrate, oleate, oxalate, palmitate, pamoate, pect
- Pharmaceutically acceptable salts derived from appropriate bases include alkali metal, alkaline earth metal, ammonium and N + (C 1–4 alkyl) 4 salts.
- Representative alkali or alkaline earth metal salts include sodium, lithium, potassium, calcium, magnesium, and the like.
- Further pharmaceutically acceptable salts include, when appropriate, nontoxic ammonium, quaternary ammonium, and amine cations formed using counterions such as halide, hydroxide, carboxylate, sulfate, phosphate, nitrate, lower alkyl sulfonate, and aryl sulfonate.
- a “subject” to which administration is contemplated includes, but is not limited to, humans (e.g., a male or female of any age group, e.g., a pediatric subject (e.g., infant, child, adolescent) or adult subject (e.g., young adult, middle–aged adult or senior adult)) and/or a non-human animal, e.g., a mammal such as primates (e.g., cynomolgus monkeys, rhesus monkeys), cattle, pigs, horses, sheep, goats, rodents, cats, and/or dogs.
- the subject is a human.
- the subject is a non- human animal.
- the terms “human,” “patient,” and “subject” are used interchangeably herein.
- Disease, disorder, and condition are used interchangeably herein.
- “treat,” “treating,” and “treatment” refer to the treatment of a disease, disorder, or symptom or manifestation of such in a subject, e.g., in a human. This includes: (a) preventing a disease or disorder, (b) inhibiting the disease, disorder, etc., i.e., slowing or arresting its progress or development; and (b) relieving the disease, disorder, etc., i.e., causing regression of the disease state.
- “prevent”, “preventing” and “prevention” refer to causing a disease, disorder, or symptom or manifestation of such not to occur for at least a period of time in at least some subjects.
- the term “effective amount” as used herein refers to the amount of a compound, e.g., a compound or composition described herein, sufficient to elicit a beneficial or desired result or biological effect.
- the effective amount of a compound or composition described herein may vary depending on such factors as the desired biological endpoint, the pharmacokinetics of the compound, the disease being treated, the mode of administration, and the age, weight, health, and condition of the subject.
- an effective amount can be administered in one or more administrations, applications, or dosages and is not intended to be limited to a particular formulation or administration route.
- an effective amount may be an amount of a compound or composition sufficient to achieve one or more of the following: (i) inhibit an activity of p97 protein; or (ii) treat or prevent a disease or condition associated with an activity of p97 protein, e.g., cancer, or a symptom or manifestation thereof.
- a “therapeutically effective amount” of a compound or composition is an amount sufficient to provide a therapeutic benefit in the treatment of a disease or condition, or to delay or minimize one or more symptoms associated with the disease, disorder or condition.
- a therapeutically effective amount of a compound or composition described herein may vary depending on such factors as the desired biological endpoint, the pharmacokinetics of the compound, the disease being treated, the mode of administration, and the age, weight, health, and condition of the subject.
- a therapeutically effective amount of a compound or composition can be administered in one or more administrations, applications, or dosages and is not intended to be limited to a particular formulation or administration route.
- a therapeutically effective amount of a compound or composition also includes an amount of therapeutic agent, alone or in combination with other therapies, which provides a therapeutic benefit in the treatment of the disease, disorder or condition.
- a therapeutically effective amount can encompass an amount of a compound or composition that improves overall therapy, reduces or avoids symptoms or causes of disease or condition, or enhances the therapeutic efficacy of another therapeutic agent.
- variable or parameters are disclosed in groups or in ranges. It is specifically intended that the description include each and every individual subcombination of the members of such groups and ranges.
- an integer in the range of 0 to 40 is specifically intended to individually disclose 0, 1, 2. 3, 4, 5, 6, 7, 8. 9. 10. 1 1. 12. 13. 14. 15, 16, 17, 18, 19, 20, 21, 22, 23, 24. 25. 26. 27. 28. 29. 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, and 40
- an integer in the range of 1 to 20 is specifically intended to individually disclose 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, and 20.
- each R 1 is independently selected from the group consisting of halogen, C 1-6 alkyl, C 1- 6 alkoxy, C1-6 haloalkyl, and C1-6 alkylene-OH;
- R 2 is selected from hydrogen and C1-6 alkyl;
- R 3 is selected from the group consisting of hydrogen, 3-10 membered heterocyclyl, - C(O)-(3-10 membered heterocyclyl), and C 1-6 alkyl optionally substituted with a substituent selected from the group consisting of C1-6 alkoxy, -CN, -C(O)N(R a )2, -NR b S(O)2C1-6 alkyl, - S(O) 2 N(R b ) 2, 5-6 membered heteroaryl, 3-10 membered heterocyclyl, and -C(O)-(3-10 membered heterocyclyl), wherein the 3-10 member
- each R 1 is independently selected from the group consisting of halogen, C 1-6 alkyl, C 1- 6 alkoxy, C1-6 haloalkyl, and C1-6 alkylene-OH;
- R 2 is selected from hydrogen and C1-6 alkyl;
- R 3 is selected from the group consisting of hydrogen, 3-10 membered heterocyclyl, - C(O)-(3-10 membered heterocyclyl), -L 1 -R 8 , and C 1-6 alkyl optionally substituted with a substituent selected from the group consisting of C1-6 alkoxy, -CN, -C(O)N(R a )2, - NR b S(O) 2 C 1-6 alkyl, -S(O) 2 N(R b ) 2, 5-6 membered heteroaryl, 3-10 membered heterocyclyl, and -C(O)-(3-10 membered heterocyclyl), wherein the 3
- R 2 is hydrogen.
- R 3 is selected from the group consisting of hydrogen, methyl, [00075] ce ta e bod e ts, s 1 bodiments, R 3 is hydrogen. [00077] In some embodiments, R 3 is -L 1 -R 8 .
- L 1 is a linker moiety selected from the group consisting of: -(C1-10 alkylene)-C(O)-, -(2-10 membered heteroalkylene)-C(O)-, -(2-10 membered heteroalkylene)-C(O)-(C 1-10 alkylene), and -(C 1-10 alkylene)-(3-7 membered heterocyclylene)- (C1-10 alkylene).
- R 8 is a moiety derived from an immunomodulatory imide drug.
- Immunomodulatory imide drugs are a class of drugs that adjust immune responses which contain an imide group.
- the immunomodulatory imide drug is a thalidomide analog, e.g., a thalidomide analog selected from the group consisting of pomalidomide and lenalidomide.
- R 8 is selected from: . ethyl.
- R 4 is methyl.
- R 4 is hydrogen.
- R 5 is hydrogen.
- R 6 is selected from hydrogen and methyl.
- R 6 is methyl.
- R 6 is at the 2-position of the indolyl.
- each R 7 is independently selected from the group consisting of hydrogen, C1-6 alkyl, -(C1-6 alkylene)-OH, -(C1-6 alkylene)-N(R b )2, -CN, and - C(O)NH2. In some embodiments, R 7 is -C(O)NH2. [00085] In other embodiments, R 7 is at the 4-position of the indolyl. [00086] In some embodiments, n is 1. In other embodiments, n is 2. [00087] In certain embodiments, m is 0. In some embodiments, m is 1. In other embodiments, m is 2.
- R 1 is selected from the group consisting of halogen, -CH2- OH, CHF2, CF3, -methyl, and -OMe.
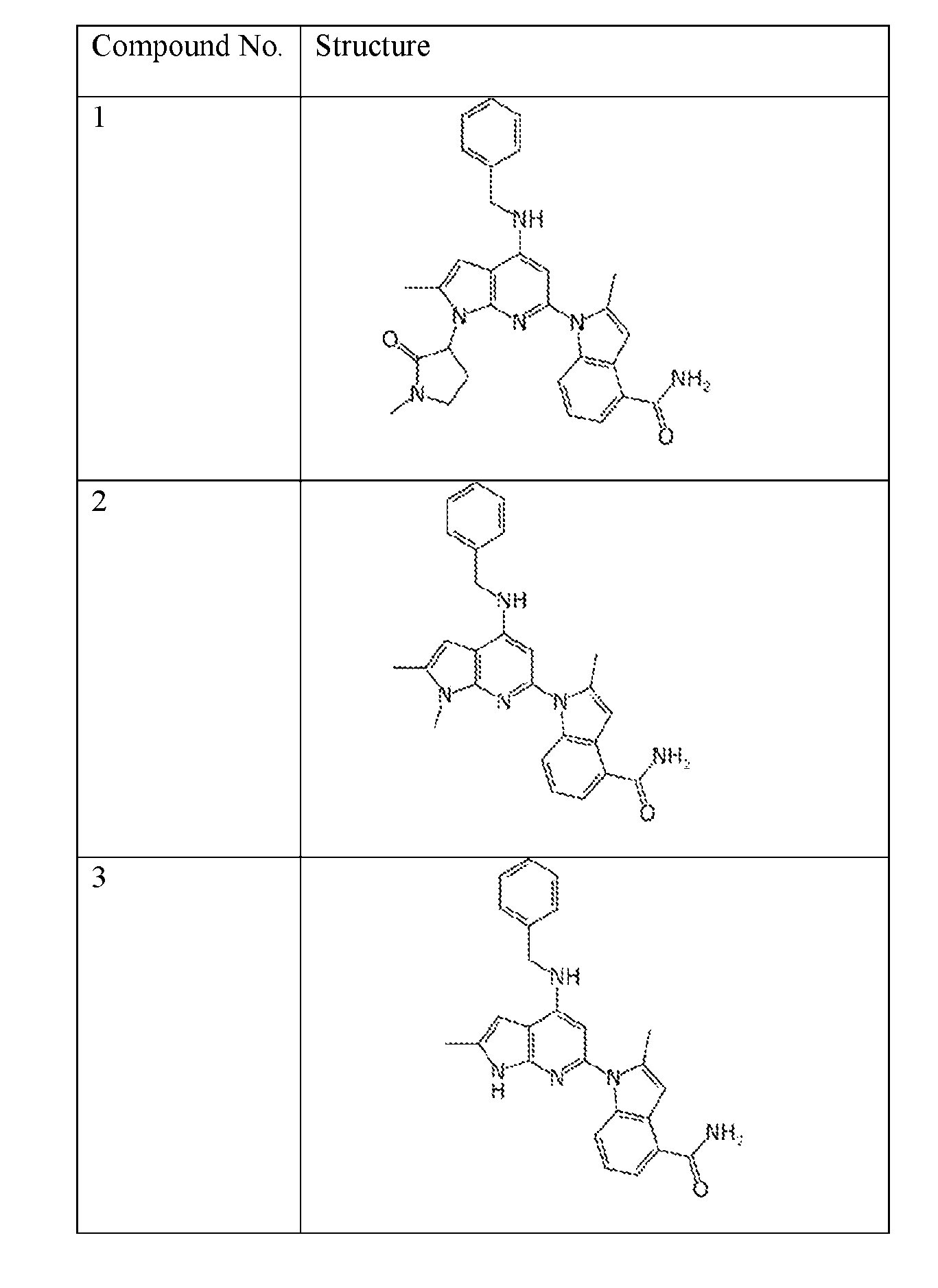
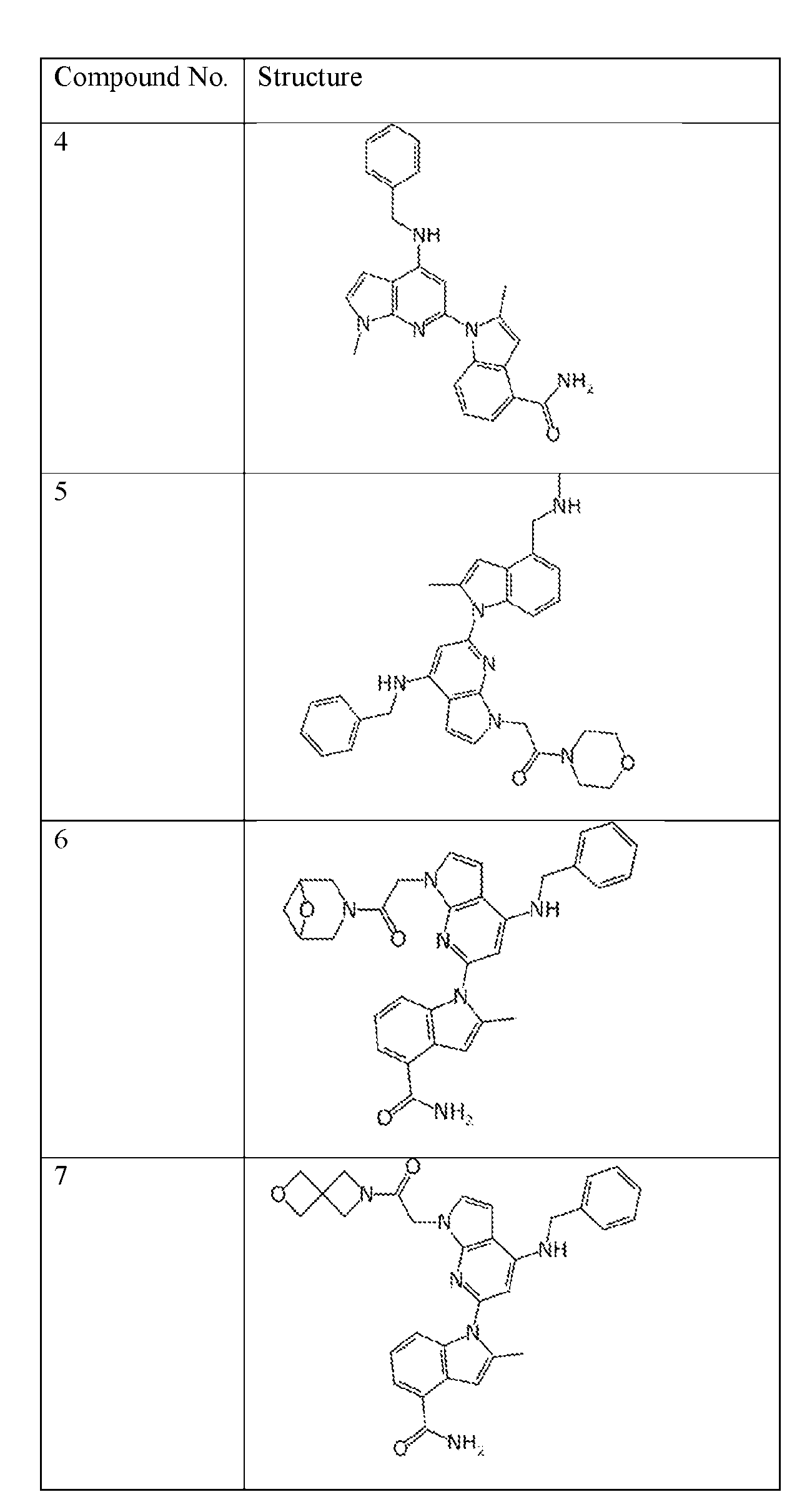
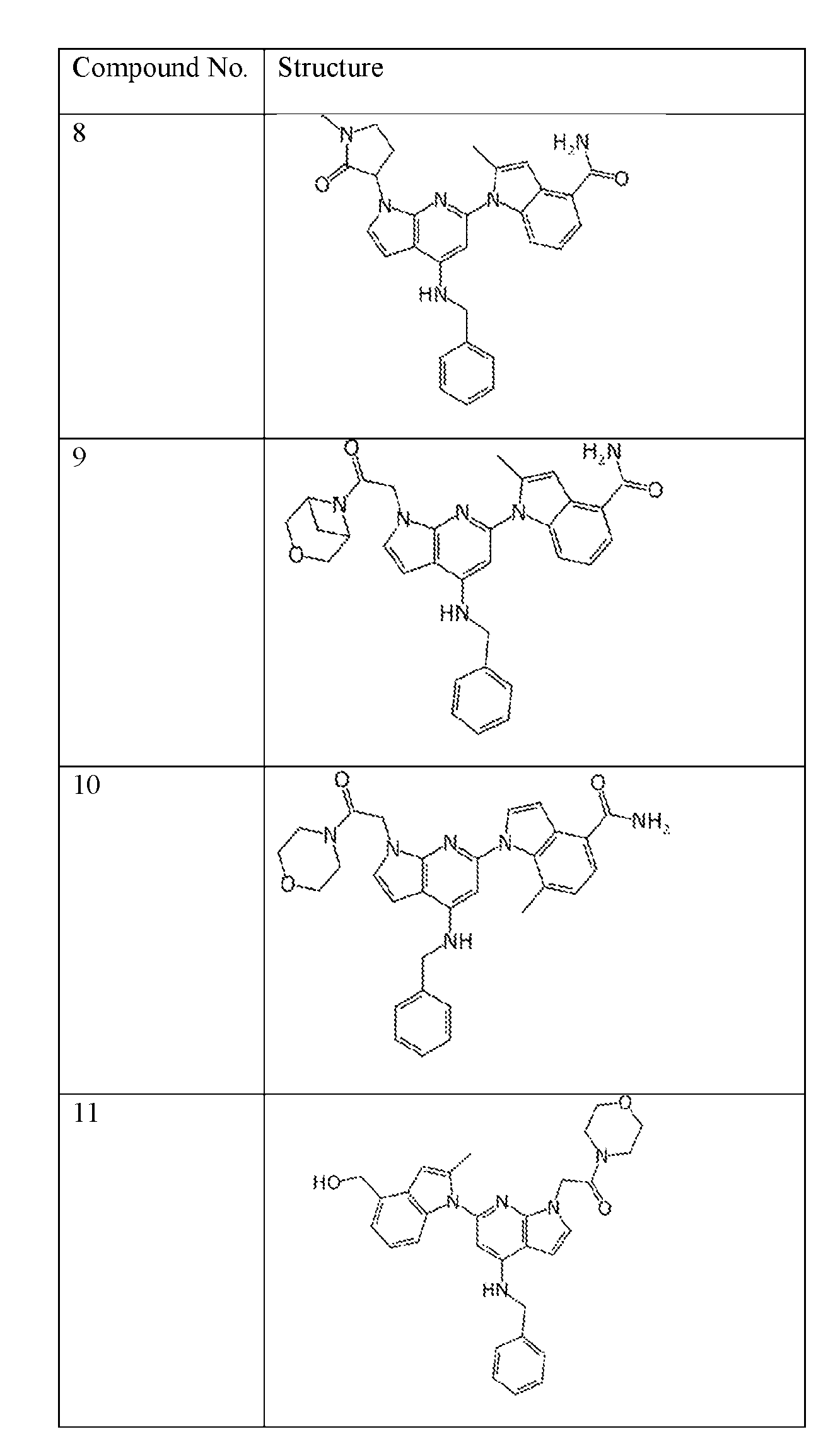
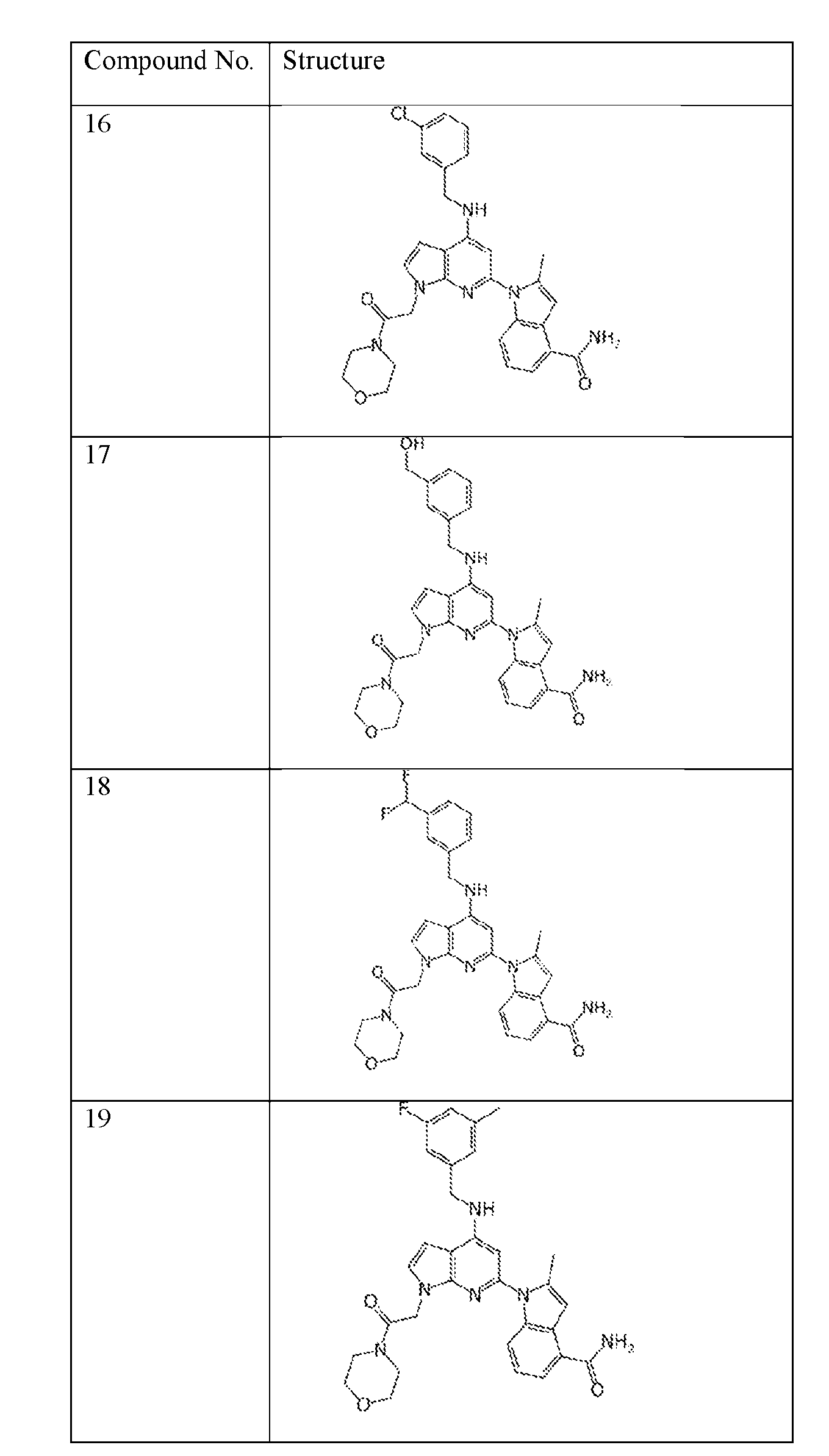
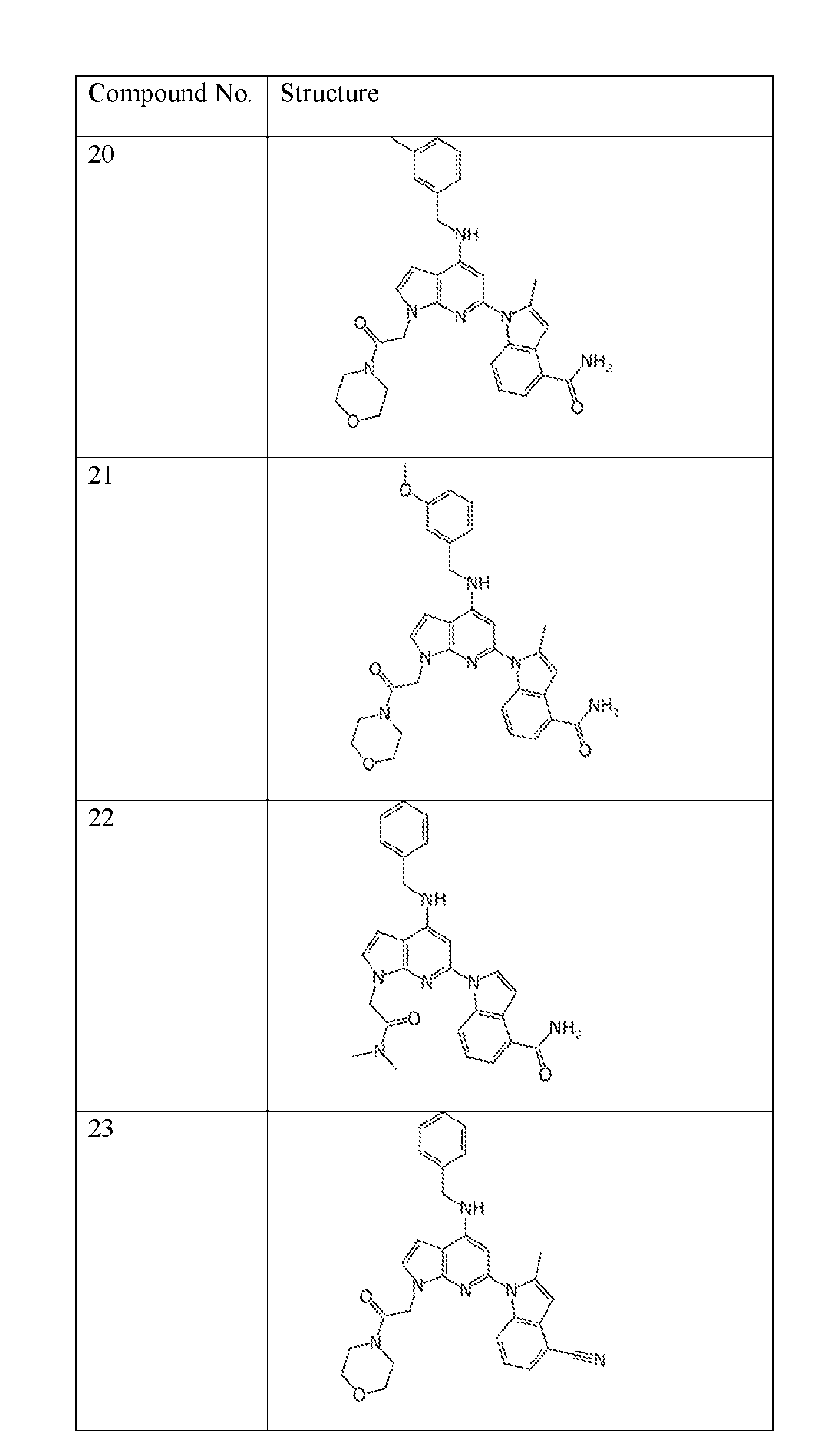
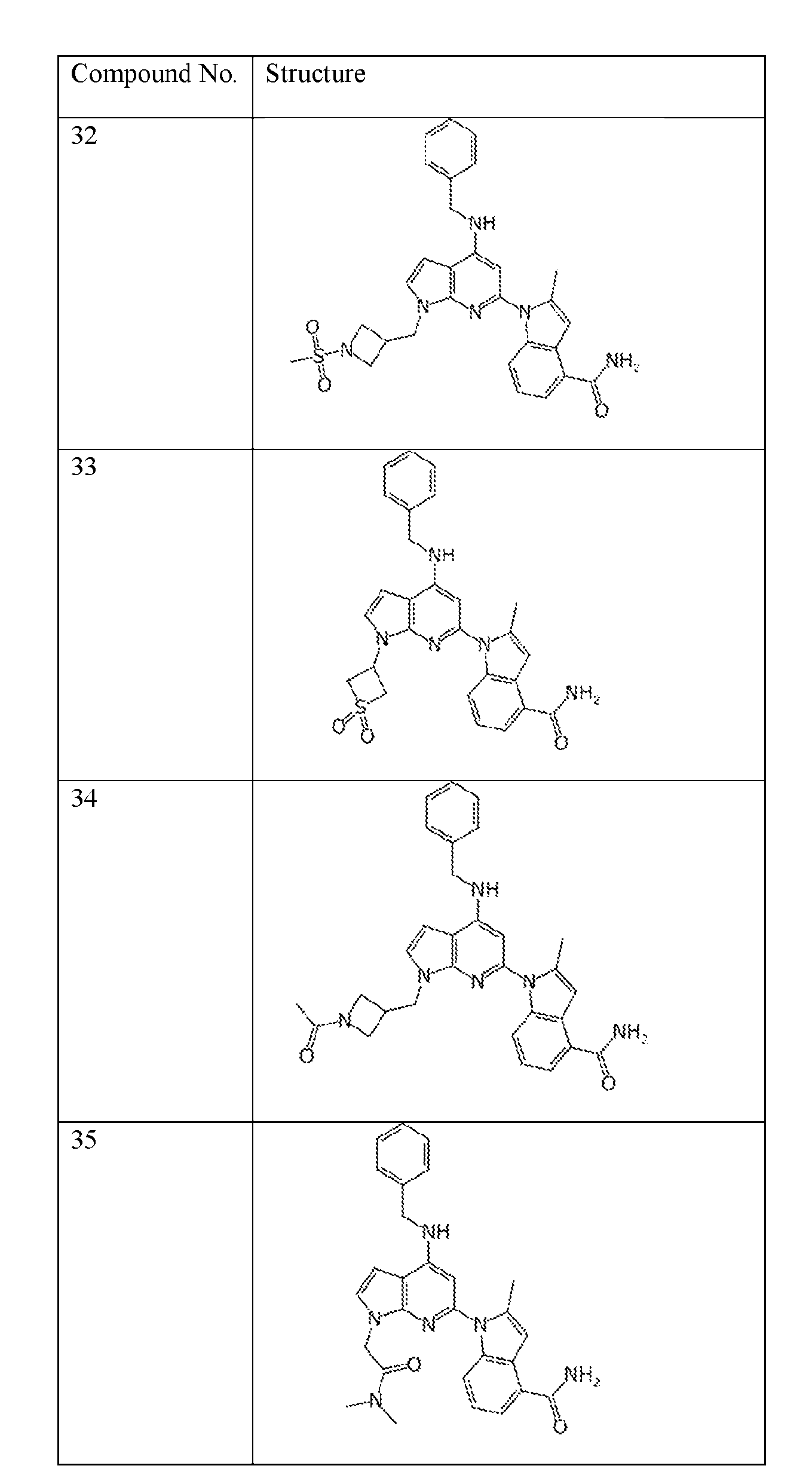
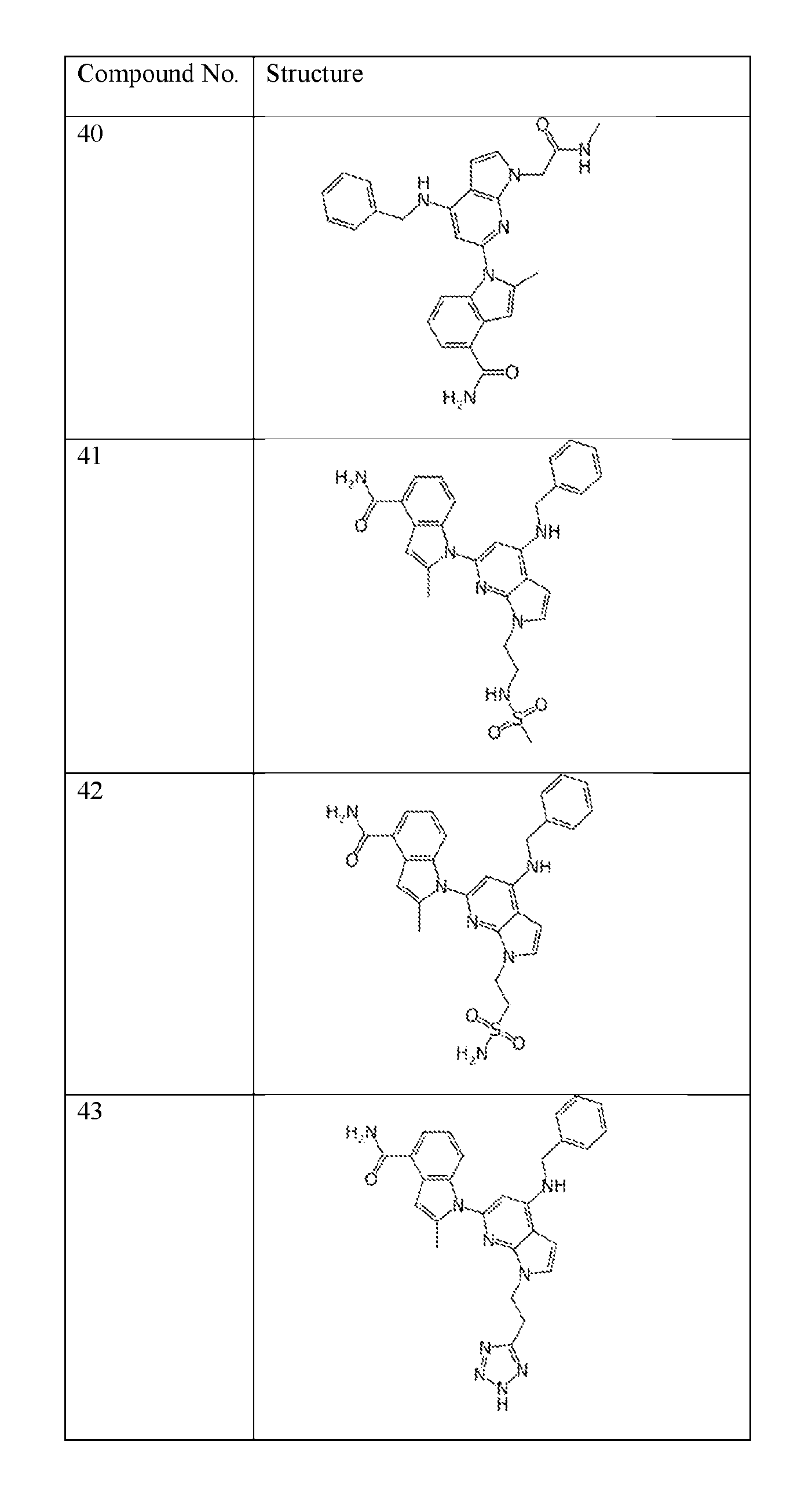
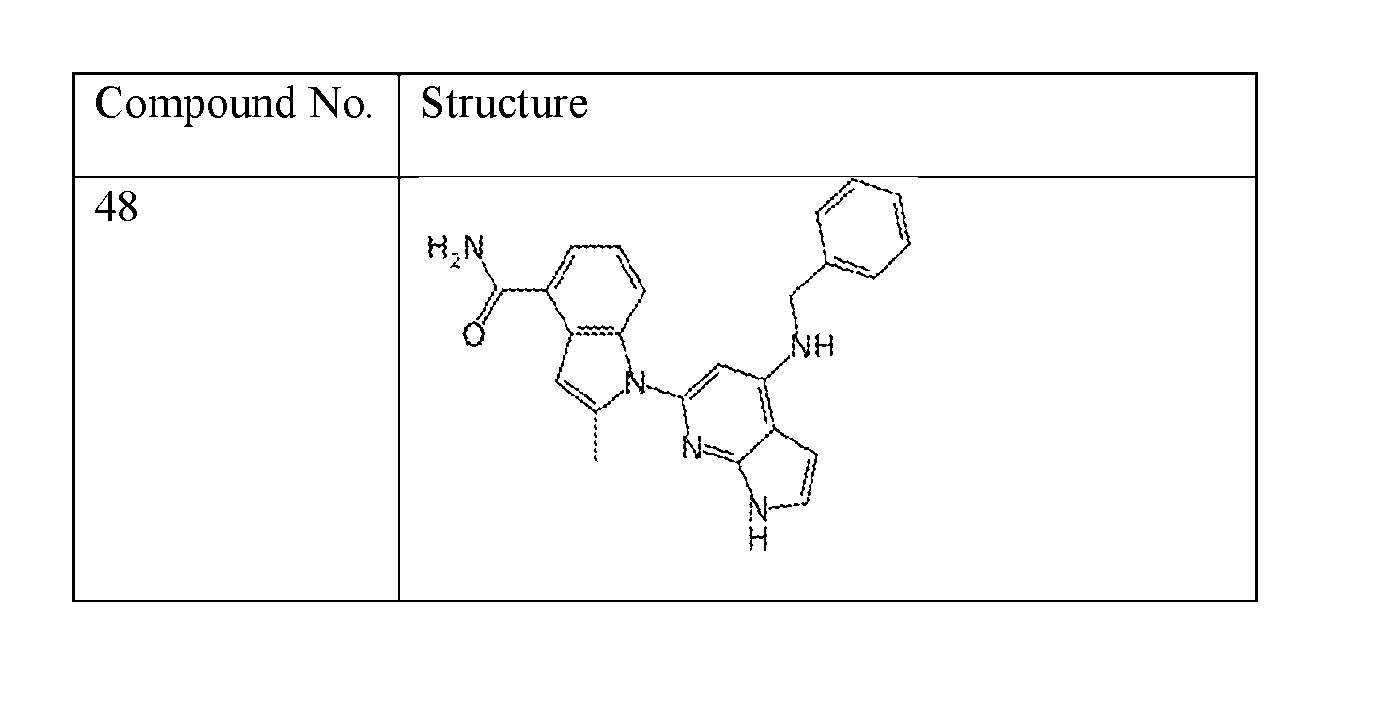
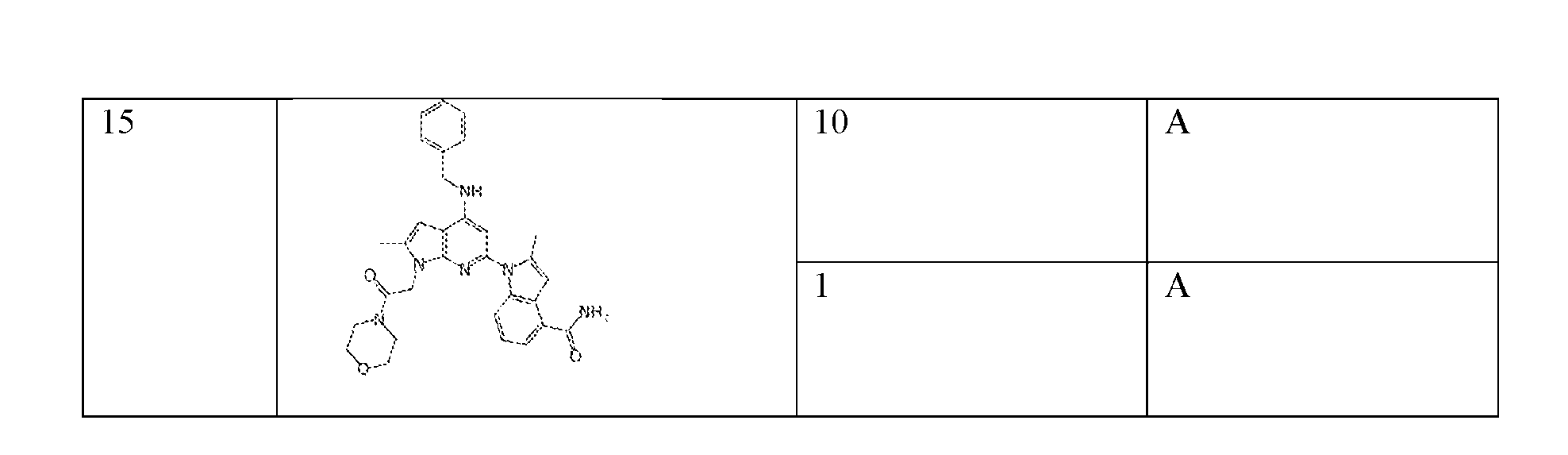
- the compound of Formula (I) or (II) is selected from Table 1 or a pharmaceutically acceptable salt thereof. Table 1.
- a commercially available 4-6-dihalosubstituted-7-azaindole is typically used as a starting material or a starting material may be prepared from a commercially available material by methods known to one of ordinary skill in the art.
- the nitrogen of the 5- membered ring of the 7-azaindole core is reacted with a protecting group under reaction conditions suitable for the protecting group (e.g., sodium hydride as a base in DMF solvent with SEM-chloride or benzenesulfonyl chloride as protecting group reagents).
- R 4 of the protected compound is functionalized (e.g...
- Protecting group removal e.g., TFA for SEM
- R 3 group introduction e.g., 2- chloroacetyl group, with a simple example being 2-chloroacetamide
- R 3 group introduction e.g., 2- chloroacetyl group, with a simple example being 2-chloroacetamide
- R 3 group introduction e.g., 2- chloroacetyl group, with a simple example being 2-chloroacetamide
- R 3 group introduction e.g., 2- chloroacetyl group, with a simple example being 2-chloroacetamide
- R 3 group introduction e.g., 2- chloroacetyl group, with a simple example being 2-chloroacetamide
- R 3 is -L'-R 8
- a linker group is introduced at R 3 , and then the pendant functionality of the linker (typically an acid or an amide) is reacted with an appropriately functionalized immunomodulatory imide compound (such as pomalidomide or
- compositions that contain, as the active ingredient, one or more of the compounds described, or a pharmaceutically acceptable salt or ester thereof, and one or more pharmaceutically acceptable excipients, carriers, including inert solid diluents and fillers, diluents, including sterile aqueous solution and various organic solvents, permeation enhancers, solubilizers and adjuvants.
- the pharmaceutical compositions may be administered alone or in combination with other therapeutic agents.
- Such compositions are prepared in a manner well known in the pharmaceutical art (see, e.g., Adeboye Adejare, Remington: The Science and Practice of Pharmacy (23 d ed. 2020)).
- a pharmaceutical composition may contain formulation materials for modifying, maintaining or preserving, for example, the pH, osmolarity, viscosity, clarity, color, isotonicity, odor, sterility, stability, rate of dissolution or release, adsorption or penetration of the composition.
- suitable formulation materials include, but are not limited to, amino acids (such as glycine, glutamine, asparagine, arginine or lysine); antimicrobials; antioxidants (such as ascorbic acid, sodium sulfite or sodium hydrogen-sulfite); buffers (such as borate, bicarbonate, Tris-HCl, citrates, phosphates or other organic acids); bulking agents (such as mannitol or glycine); chelating agents (such as ethylenediamine tetraacetic acid (EDTA)); complexing agents (such as caffeine, polyvinylpyrrolidone, beta-cyclodextrin or hydroxypropyl-beta-cyclodextrin); fillers; monosaccharides; disaccharides; and other carbohydrates (such as glucose, mannose or dextrins); proteins (such as serum albumin, gelatin or immunoglobulins); coloring, flavoring and diluting agents; emulsifying agents;
- amino acids
- sorbitan esters such as polysorbate 20, polysorbate, triton, tromethamine, lecithin, cholesterol, tyloxapal
- stability enhancing agents such as sucrose or sorbitol
- tonicity enhancing agents such as alkali metal halides, preferably sodium or potassium chloride, mannitol sorbitol
- delivery vehicles diluents; and/or excipients (see Adeboye Adejare (2020) supra).
- compositions may be administered in either single or multiple doses by any of the accepted modes of administration of agents having similar utilities, including, orally, topically, by rectal, buccal, intranasal or transdermal routes, intravenously, intraperitoneally, parenterally, intramuscularly, subcutaneously, or as an inhalant, or via an impregnated or coated device, e.g., a stent.
- One mode for administration is parenteral, particularly by injection.
- the forms in which the novel compounds or compositions described herein may be incorporated for administration by injection include aqueous or oil suspensions, or emulsions, with sesame oil, com oil, cottonseed oil, or peanut oil, as well as elixirs, mannitol, dextrose, or a sterile aqueous solution, and similar pharmaceutical vehicles.
- Aqueous solutions in saline are also conventionally used for injection, but less preferred in the context of the present invention.
- Ethanol, glycerol, propylene glycol, liquid polyethylene glycol, and the like (and suitable mixtures thereof), cyclodextrin derivatives, and vegetable oils may also be employed.
- the proper fluidity can be maintained, for example, by the use of a coating, such as lecithin, by the maintenance of the required particle size in the case of dispersion and by the use of surfactants.
- Sterile injectable solutions are prepared by incorporating a compound according to the present invention in the required amount in the appropriate solvent with various other ingredients as enumerated above, as required, followed by filtered sterilization.
- dispersions are prepared by incorporating the various sterilized active ingredients into a sterile vehicle which contains the basic dispersion medium and the required other ingredients from those enumerated above.
- the preferred methods of preparation are vacuum-drying and freeze- drying techniques which yield a powder of the active ingredient plus any additional desired ingredient from a previously sterile-filtered solution thereof.
- Oral administration is another route for administration of compounds in accordance with the invention. Administration may be via capsule or enteric coated tablets, or the like.
- the active ingredient is usually diluted by an excipient and/or enclosed within such a carrier that can be in the form of a capsule, sachet, paper or other container.
- the excipient serves as a diluent, it can be in the form of a solid, semi-solid, or liquid material, which acts as a vehicle, carrier or medium for the active ingredient.
- compositions can be in the form of tablets, pills, powders, lozenges, sachets, cachets, elixirs, suspensions, emulsions, solutions, syrups, aerosols (as a solid or in a liquid medium), ointments containing, for example, up to 10% by w eight of the active compound, soft and hard gelatin capsules, sterile injectable solutions, and sterile packaged powders.
- excipients include lactose, dextrose, sucrose, sorbitol, mannitol, starches, gum acacia, calcium phosphate, alginates, tragacanth, gelatin, calcium silicate, microcrystalline cellulose, polyvinylpyrrolidone, cellulose, sterile water, syrup, and methyl cellulose.
- the formulations can additionally include: lubricating agents such as talc, magnesium stearate, and mineral oil; wetting agents; emulsifying and suspending agents; preserving agents such as methyl and propylhydroxy-benzoates; sweetening agents; and flavoring agents.
- the compounds or compositions described herein can be formulated so as to provide quick, sustained or delayed release of the active ingredient after administration to the patient by employing procedures known in the art.
- Controlled release drug delivery systems for oral administration include osmotic pump systems and dissolutional systems containing polymer- coated reservoirs or drug-polymer matrix formulations. Examples of controlled release systems are given in U.S. Pat. Nos.3,845,770; 4,326,525; 4,902,514; and 5,616,345.
- the compounds or compositions described herein can be delivered by transdermal delivery devices (“patches”).
- transdermal patches may be used to provide continuous or discontinuous infusion of the compounds of the present invention in controlled amounts.
- the construction and use of transdermal patches for the delivery of pharmaceutical agents is well known in the art (see, e.g., U.S. Pat. Nos.5,023,252, 4,992,445 and 5,001,139).
- Such patches may be constructed for continuous, pulsatile, or on demand delivery of pharmaceutical agents.
- the compositions are preferably formulated in a unit dosage form.
- unit dosage forms refers to physically discrete units suitable as unitary dosages for human subjects and other mammals, each unit containing a predetermined quantity of active material calculated to produce the desired therapeutic effect, in association with a suitable pharmaceutical excipient (e.g., a tablet, capsule, ampoule).
- a suitable pharmaceutical excipient e.g., a tablet, capsule, ampoule.
- the compounds are generally administered in a pharmaceutically effective amount.
- each dosage unit may contain from 1 mg to 2 g (e.g., 1 mg to 1 g, 1 mg to 500 mg, 1 mg to 100 mg) of a compound described herein, and for parenteral administration, from 0.1 to 700 mg (e.g., from 0.1 mg to 300 mg, from 0.1 mg to 100 mg, from 0.1 mg to 10 mg) of a compound described herein.
- the reference to compound includes the compound either alone or in combination with a pharmaceutically acceptable salt. It will be understood, however, that the amount of the compound actually administered usually will be determined by a physician, in the light of the relevant circumstances, including the condition to be treated, the chosen route of administration, the actual compound administered and its relative activity , the age, weight, and response of the individual patient, the severity’ of the patient’s symptoms, and the like.
- the principal active ingredient is mixed with a pharmaceutical excipient to form a solid preformulation composition containing a homogeneous mixture of a compound of the present invention.
- a pharmaceutical excipient for preparing solid compositions such as tablets, the principal active ingredient is mixed with a pharmaceutical excipient to form a solid preformulation composition containing a homogeneous mixture of a compound of the present invention.
- these preformulation compositions as homogeneous, it is meant that the active ingredient is dispersed evenly throughout the composition so that the composition may be readily subdivided into equally effective unit dosage forms such as tablets, pills and capsules.
- the tablets or pills of the present invention may be coated or otherwise compounded to provide a dosage form affording the advantage of prolonged action, or to protect from the acid conditions of the stomach.
- the tablet or pill can comprise an inner dosage and an outer dosage component, the latter being in the form of an envelope over the former.
- the two components can be separated by an enteric layer that serves to resist disintegration in the stomach and permit the inner component to pass intact into the duodenum or to be delayed in release.
- enteric layers or coatings such materials including a number of polymeric acids and mixtures of polymeric acids with such materials as shellac, cetyl alcohol, and cellulose acetate.
- compositions for inhalation or insufflation include solutions and suspensions in pharmaceutically acceptable, aqueous or organic solvents, or mixtures thereof, and powders.
- the liquid or solid compositions may contain suitable pharmaceutically acceptable excipients as described supra.
- the compositions are administered by the oral or nasal respiratory route for local or systemic effect.
- Compositions in preferably pharmaceutically acceptable solvents may be nebulized by use of inert gases. Nebulized solutions may be inhaled directly from the nebulizing device or the nebulizing device may be attached to a facemask tent, or intermittent positive pressure breathing machine. Solution, suspension, or powder compositions may be administered, preferably orally or nasally, from devices that deliver the formulation in an appropriate manner.
- a pharmaceutical composition comprising a disclosed compound, e.g., a compound of Formula (I) or (II), or pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable carrier.
- the compounds and compositions described herein can be used in a method of inhibiting p97 protein and a method of treating a disease or condition associated with an activity of p97 protein, e.g., cancer or a neurodegenerative disorder.
- One aspect of the present disclosure includes methods of inhibiting p97 protein in a subject in need thereof.
- the present disclosure provides a method of treating a disease or condition associated with an activity of p97 protein, e.g., cancer, in a subject in need thereof.
- the treatment of the subject in need thereof comprises administering a compound of the present disclosure (e.g., a compound of Formula (I) or (II) or a pharmaceutically acceptable salt thereof) to the subject in need thereof.
- the disease or condition is cancer.
- abnormal cell growth as used herein and unless otherwise indicated, refers to cell growth that is independent of normal regulatory mechanisms (e.g., loss of contact inhibition).
- Abnormal cell growth can refer to cell growth of epithelial cells (e.g, carcinomas, adenocarcinomas), mesenchymal cells (e.g. sarcomas (e.g. Ewing’s sarcoma)), hematopoetic cells (e.g, lymphomas, leukemias, myelodysplasias) or other cells (e.g, melanoma, mesothelioma, and other tumors of unknown origin).
- a “neoplastic disorder” is a disease or disorder characterized by cells that have the capacity for autonomous growth or replication, e.g., an abnormal state or condition characterized by proliferative cell growth.
- An abnormal mass of tissue as a result of abnormal cell growth or division, or a neoplasm can be benign, pre-malignant or malignant.
- exemplary neoplastic disorders include: carcinoma, sarcoma, metastatic disorders (e.g, tumors arising from prostate, colon, lung, breast and liver origin), hematopoietic neoplastic disorders, e.g, leukemias, metastatic tumors.
- Treatment with the compound may be in an amount effective to ameliorate at least one symptom of the neoplastic disorder, e.g, reduced cell proliferation, reduced tumor mass, etc.
- the methods and compositions described herein may be useful in the prevention and treatment of a cancer, including, e.g, solid tumors, soft tissue tumors, and metastases thereof.
- a cancer including, e.g, solid tumors, soft tissue tumors, and metastases thereof.
- the disclosed methods are also useful in treating non-solid cancers.
- Exemplary' solid tumors include malignancies (e.g, sarcomas, adenocarcinomas, and carcinomas) of the various organ systems, such as those of lung, lymphoid, gastrointestinal (e.g, colon, rectum. or colorectum), brain, pancreas, genitourinary tracts (e.g, renal, urothelial, or testicular tumors), pharynx, prostate, and ovary.
- malignancies e.g, sarcomas, adenocarcinomas, and carcinomas
- gastrointestinal e.g, colon, rectum. or colorectum
- Exemplary adenocarcinomas include colorectal cancers, renal-cell carcinoma, liver cancer (e.g.. hepatocellular carcinoma), lung cancer (e.g., non-small cell lung cancer (NSCLC)), pancreatic (e.g., metastatic pancreatic adenocarcinoma) and cancer of the small intestine.
- the cancer can be colorectal cancer, NSCLC, or pancreatic cancer.
- the cancer may include brain metastasis.
- the cancer is multiple myeloma.
- the cancer is acute myeloid leukemia.
- the cancer can include mesothelioma; neurofibromatosis; e.g., neurofibromatosis type 2, neurofibromatosis type 1 , renal cancer; lung cancer, non-small cell lung cancer; thyroid cancer; a nervous system tumor; schwannoma; meningioma; schwannomatosis; neuroma acoustic; adenoid cystic carcinoma; ependymoma; ependymal tumors, or any other tumor which exhibits decreased merlin expression and/or mutation, and/or deletion and/or promotor hypermethylation of the NF-2 gene.
- the cancer is renal cancer.
- the cancer can include cancers characterized as comprising cancer stem cells, cancer associated mesenchymal cells, or tumor initiating cancer cells.
- the cancer can include cancers that have been characterized as being enriched with cancer stem cells, cancer associated mesenchymal cells, or tumor initiating cancer cells (e.g.. a tumor enriched with cells that have undergone an epithelial -to-mesenchymal transition or a metastatic tumor).
- the cancer can be a primary tumor, i.e., located at the anatomical site of tumor growth initiation.
- the cancer can also be metastatic, /. ⁇ ?., appearing at least a second anatomical site other than the anatomical site of tumor growth initiation.
- the cancer can be a recurrent cancer, i.e., cancer that returns following treatment, and after a period of time in which the cancer w as undetectable.
- the recurrent cancer can be anatomically located locally to the original tumor, e.g., anatomically near the original tumor; regionally to the original tumor, e.g., in a lymph node located near the original tumor; or distantly to the original tumor, e.g., anatomically in a region remote from the original tumor.
- the cancer can be brain or cerebrospinal fluid (CSF)-related metastasis.
- CSF cerebrospinal fluid
- the cancer can also include for example, but is not limited to, epithelial cancers, lung, pancreatic, colorectal (e.g.. metastatic colorectal), prostate, head and neck, melanoma, acute myelogenous leukemia, and glioblastoma.
- epithelial cancers include triple negative breast cancer, basal-like breast cancer, claudin-low' breast cancer, invasive, inflammatory, metaplastic, and advanced HER-2 positive or ER-positive cancers resistant to therapy.
- the cancer can also include lung adenocarcinoma, colorectal cancer (CRC), uveal melanoma, uterine endometrioid carcinoma, bladder urothelial carcinoma, breast invasive lobular carcinoma, cervical squamous cell carcinoma, cutaneous melanoma, endocervical adenocarcinoma, hepatocellular carcinoma, pancreatic adenocarcinoma, biphasic type pleural mesothelioma, renal clear cell carcinoma, renal clear cell carcinoma, stomach adenocarcinoma, tubular stomach adenocarcinoma, head and neck squamous cell carcinoma, uterine carcinosarcoma, or uterine malignant mixed Mullerian tumor.
- CRC colorectal cancer
- uveal melanoma uterine endometrioid carcinoma
- bladder urothelial carcinoma breast invasive lobular carcinoma
- cervical squamous cell carcinoma cutaneous mela
- the cancer is unresectable or metastatic melanoma, melanoma with lymph node involvement or metastatic disease who have undergone complete resection, metastatic non-small cell lung cancer and progression on or after platinum-based chemotherapy, metastatic small cell lung cancer with progression after platinum-based chemotherapy and at least one other line of therapy, advanced renal cell carcinoma who have received prior anti angiogenic therapy, advanced renal cell carcinoma, classical Hodgkin lymphoma, recurrent or metastatic squamous cell carcinoma of the head and neck with disease progression on or after a platinum-based therapy, urothelial carcinoma (e.g., locally advanced or metastatic), microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) metastatic colorectal cancer, or hepatocellular carcinoma.
- urothelial carcinoma e.g., locally advanced or metastatic
- MSI-H microsatellite instability-high
- dMMR mismatch repair deficient
- cancers include primary mediastinal large B-cell lymphoma, mantle cell lymphoma, gastric cancer, esophageal cancer, cervical cancer, merkel cell carcinoma, abdominal, esophagus, glioma, tongue, neuroblastoma, osteosarcoma, retinoblastoma, Wilm’s tumor, multiple myeloma, skin, lymphoma, blood and bone marrow cancers (e.g., advanced hematological malignancies, leukemia, e.g.. acute myeloid leukemia (e.g, primary’ or secondary), acute lymphoblastic leukemia, acute lymphocytic leukemia.
- leukemia e.g. acute myeloid leukemia (e.g, primary’ or secondary
- acute lymphoblastic leukemia e.g., acute lymphocytic leukemia.
- T cell leukemia hematological malignancies, advanced myeloproliferative disorders, myelodysplastic syndrome, relapsed or refractory’ multiple myeloma, advanced myeloproliferative disorders
- retinal bladder, cervical, kidney, endometrial, meningioma, uterine, lung, small cell lung cancer, non-small cell lung cancer, nasopharyngeal carcinoma, neuroblastoma, solid tumor, hematologic malignancy, squamous cell carcinoma, testicular, thyroid, mesothelioma, brain vulval, sarcoma, intestine, oral, endocrine, salivary, spermatocyte seminoma, sporadic medullary thyroid carcinoma, non-proliferating testes cells, cancers related to malignant mast cells, non-Hodgkin’s lymphoma, and diffuse large B cell lymphoma.
- the tumor is a solid tumor.
- Methods described herein can reduce, ameliorate or altogether eliminate the disorder, and/or its associated symptoms, to keep it from becoming worse, to slow the rate of progression, or to minimize the rate of recurrence of the disorder once it has been initially eliminated (e.g., to avoid a relapse).
- a suitable dose and therapeutic regimen may vary' depending upon the specific compounds, combinations, and/or pharmaceutical compositions used and the mode of delivery of the compounds, combinations, and/or pharmaceutical compositions.
- the method increases the average length of survival, increases the average length of progression-free survival, and/or reduces the rate of recurrence, of subjects treated with the combinations described herein in a statistically significant manner.
- the cancer is bone cancer, pancreatic cancer, uterine cancer, ovarian cancer (e.g, unresectable low-grade ovarian, advanced or metastatic ovarian cancer), rectal cancer, cancer of the anal region, stomach cancer, colon cancer, breast cancer (e.g., triple-negative breast cancer (e.g, breast cancer which does not express the genes for the estrogen receptor, progesterone receptor, and Her2/neu)), uterine cancer, carcinoma of the fallopian tubes, carcinoma of the endometrium, carcinoma of the cervix, carcinoma of the vagina, carcinoma of the vulva, Hodgkin’s Disease, cancer of the small intestine, cancer of the endocrine system, cancer of the thyroid gland, cancer of the parathyroid gland, cancer of the adrenal gland, sarcoma of soft tissue, cancer of the urethra, cancer of the penis, prostate cancer, chronic or acute leukemia, lymphocytic lymphomas, cancer of the ureter, renal cell carcinoma,
- ovarian cancer e
- a disease or condition associated with an activity of p97 protein is a neurodegenerative disorder.
- the neurodegenerative disorders include, for example, inclusion body myopathy (IBM), Paget's disease of the bone (PDB), frontotemporal dementia (FTD) and amyotrophic lateral sclerosis (ALS)).
- neurodegenerative diseases include, for example, amyotrophic lateral sclerosis (ALS), Charcot-Marie-Tooth disease type 2Y, and Inclusion body myopathy with early-onset Paget disease and frontotemporal dementia (IBMPFD).
- Neurodegenerative disorders also include subjects having p97 mutations, and symptoms manifesting as, for example, Parkinsonism, ataxia, cataracts, dilated cardiomyopathy, hepatic fibrosis, and hearing loss.
- a disease or condition associated with an activity of p97 protein is an infectious disease.
- the compound of the disclosure e.g, a compound of Formula (I) or (II)
- viruses e.g, poliovirus, herpes simplex virus (HSV), cytomegalovirus (CMV), or influenza.
- the compound of the disclosure may be included in a pharmaceutical formulation such as those disclosed herein, and may be administered in any pharmaceutically acceptable manner, including methods of administration described herein.
- An effective amount of a compound useful in the methods of the present invention may be administered to a subject in need thereof by any of a number of well-known methods for administering pharmaceutical compounds.
- the compound may be administered systemically or locally.
- the compound is administered intravenously.
- the compounds useful in the methods of the present technology may be administered via rapid intravenous bolus injection.
- the compound is administered as a constant rate intravenous infusion.
- the compound may also be administered orally, topically, intranasally, intramuscularly, subcutaneously, or transdermally.
- Other routes of administration include intracerebroventricularly or intrathecally. Intracerebroventricularly refers to administration into the ventricular system of the brain. Intrathecally refers to administration into the space under the arachnoid membrane of the spinal cord.
- the compounds useful in the methods of the present technology may also be administered to a subject in need thereof by sustained or controlled release, as is known in the art.
- Sustained release administration is a method of drug delivery to achieve a certain level of the drug over a particular period of time. The level typically is measured by serum or plasma concentration by, for example, nuclear magnetic resonance spectroscopy (NMR) or liquid chromatography mass spectrometry (LCMS).
- NMR nuclear magnetic resonance spectroscopy
- LCMS liquid chromatography mass spectrometry
- the compounds provided herein may be isolated and purified by known standard procedures. Such procedures include recrystallization, filtration, flash chromatography, trituration, high pressure liquid chromatography (HPLC). or supercritical fluid chromatography (SFC). Note that flash chromatography may either be performed manually or via an automated system.
- the compounds provided herein may be characterized by known standard procedures, such as nuclear magnetic resonance spectroscopy (NMR) or liquid chromatography mass spectrometry (LCMS). NMR chemical shifts are reported in part per million (ppm) and are generated using methods well known to those of skill in the art.
- the analytical HPLC chromatograms were performed using an Agilent 1100/1200 series instrument with DAD detector (190 nm to 300 nm) and a thermostatted column compartment set to 35 °C.
- the mass spectra were recorded with a Waters SQD detector with the desolvation temperature set to 500 °C, the source temperature set to 150 °C and the desolvation gas flow set to 1000 L/h.
- the mass spectrometer was equipped with an electrospray ion source (ESI) operated in a positive, or negative, ion mode and was set to scan between m/z 100-750 with a scan time of 0.2 s.
- ESI electrospray ion source
- Method A05 is a 3.50 minute method using a Kinetex EVO Cl 8 (5 pM, 4.6 x 50 mm) column, a flow rate of 2.20 mL/min, and a low pH buffer gradient of 5% to 100% of ACN in H2O (10 mM ammonium formate, adjusted to pH 4 with formic acid).
- the % ACN is kept stable at 5% from 0.00-0.50 minutes and gradually increased to 100% from 0.50-2.50 minutes. From 2.50-3.50 minutes, the % ACN is kept stable at 100%.
- Method A50 is a 3.50 minute method using a Kinetex EVO C18 (5 pM, 4.6 x 50 mm) column, a flow rate of 2.20 mL/min. and a low pH buffer gradient of 5% to 100% of ACN in H2O (10 mM ammonium formate, adjusted to pH 4 with formic acid).
- the % ACN is kept stable at 50% from 0.00-0.50 minutes and gradually increased to 100% from 0.50-2.50 minutes. From 2.50-3.50 minutes, the % ACN is kept stable at 100%.
- Method B05 is a 3.50 minute method using a Gemini NX-C18 (5 pM, 4.6 x 50 mm) column, a flow rate of 2.20 mL/min, and a high pH buffer gradient of 5 % to 100 % of ACN in H2O (10 mM ammonium bicarbonate, adjusted to pH 10 with ammonium hydroxide).
- the % ACN is kept stable at 5% from 0.00-0.50 minutes and gradually increased to 100% from 0.50-2.50 minutes. From 2.50-3.50 minutes, the % ACN is kept stable at 100%.
- Method B50 is a 3.50 minute method using a Gemini NX-C18 (5 pM, 4.6 x 50 mm) column, a flow rate of 2.20 mL/min. and a high pH buffer gradient of 50% to 100% of ACN in H2O (10 mM ammonium bicarbonate, adjusted to pH 10 with ammonium hydroxide).
- the % ACN is kept stable at 50% from 0.00-0.50 minutes and gradually increased to 100% from 0.50-2.50 minutes. From 2.50-3.50 minutes, the % ACN is kept stable at 100%.
- the analytical QA/QC chromatograms were also performed using a Waters AcquiW UPLC instrument with DAD detector (200 nm to 320 nm) and a thermostatted column compartment set to 35 °C.
- the mass spectra were recorded with a Waters SQD detector with the desolvation temperature set to 450 °C, the source temperature set to 150 °C and the desolvation gas flow set to 1000 L/hr.
- the mass spectrometer was equipped with an electrospray ion source (ESI) operated in a positive, or negative, ion mode and was set to scan between m/z 100-2000 with a scan time of 0.2 s.
- ESI electrospray ion source
- Step 2 N-benzyl-6-chloro-1-(2-trimethylsilylethoxymethyl)pyrrolo[2,3-b]pyridin-4- amine
- N-Ethyl-N-isopropyl-propan-2-amine (18.2 g, 141 mmol) was added to a mixture of 2-[(4,6-dichloropyrrolo[2,3-b]pyridin-1-yl)methoxy]ethyl-trimethyl-silane (14.9 g, 47.0 mmol) and benzylamine (10.1 g, 94.0 mmol) in NMP (30.0 mL). The mixture was stirred at 165 °C for 18 hr.
- the mixture was cooled to 23 °C and diluted with an aqueous solution of HCl (1 M, 100 mL).
- the aqueous phase was extracted with EtOAc (350 mL).
- the combined organic phases were washed with saturated aqueous NaHCO 3 (150 mL) and brine (150 mL), dried (Na2SO4), filtered, and concentrated.
- the residue was purified by silica gel chromatography (220 g cartridge) with hexanes and EtOAc (0-25%) to provide the title compound as a solid (11.3 g, 62%).
- Step 4 1-[4-(benzylamino)-1-(2-trimethylsilylethoxymethyl)pyrrolo[2,3-b]pyridin-6-yl]- 2-methyl-indole-4-carboxamide
- (1E)-Acetaldehyde oxime (3.21 g, 54.4 mmol) was added to a mixture of 1-[4- (benzylamino)-1-(2-trimethylsilylethoxymethyl)pyrrolo[2,3-b]pyridin-6-yl]-2-methyl-indole- 4-carbonitrile (46.0 %, 12.0 g, 10.9 mmol), palladium acetate (0.488 g, 2.17 mmol), and triphenylphosphine (1.14 g, 4.35 mmol) in a mixture of EtOH/H2O (8:1 v/v, 180 mL) at 23 °C.
- Step 5 1-[4-(benzylamino)-1H-pyrrolo[2,3-b]pyridin-6-yl]-2-methyl-indole-4- carboxamide
- TFA 50.0 mL
- 1-[4-(benzylamino)-1-(2- trimethylsilylethoxymethyl)pyrrolo[2,3-b]pyridin-6-yl]-2-methyl-indole-4-carboxamide 5.09 g, 9.68 mmol
- the mixture was stirred at 23 °C for 3 h and concentrated.
- Step 6 2-[4-(benzylamino)-6-(4-carbamoyl-2-methyl-indol-1-yl)pyrrolo[2,3-b]pyridin-1- yl]acetic acid
- Sodium hydride 60.0%, 26 mg, 0.68 mmol
- 1-[4- (benzylamino)-1H-pyrrolo[2,3-b]pyridin-6-yl]-2-methyl-indole-4-carboxamide (245 mg, 0.620 mmol) in dry DMF (5.00 mL) under N2 at 23 °C.
- Step 7 1-[4-(benzylamino)-1-[2-(morpholin-4-yl)-2-oxoethyl]-1H-pyrrolo[2,3-b]pyridin- 6-yl]-2-methyl-1H-indole-4-carboxamide
- HATU 419.0 mg, 1.10 mmol
- DIPEA 0.377 mL, 2.21 mmol
- 2-[4-(benzylamino)-6-(4-carbamoyl-2-methyl-indol-1-yl)pyrrolo[2,3-b]pyridin-1- yl]acetic acid 250 mg, 0.551 mmol
- morpholine (0.121 mL, 1.38 mmol
- Step 4 1-[1-(Benzenesulfonyl)-4-(benzylamino)-2-methyl-1H-pyrrolo[2,3-b]pyridin-6- yl]-2-methyl-1H-indole-4-carbonitrile
- 1-(Benzenesulfonyl)-N-benzyl-6-chloro-2-methyl-1H-pyrrolo[2,3-b]pyridin-4- amine 570 mg, 1.38 mmol
- 2-methyl-1H-indole-4- carbonitrile 216 mg, 1.38 mmol
- t-BuONa 206 mg, 2.14 mmol
- [PdCl(cinnamyl)]2 143 mg, 0.28 mmol
- BippyPhos 295 mg, 0.58 mmol
- Step 5 1-[1-(Benzenesulfonyl)-4-(benzylamino)-2-methyl-1H-pyrrolo[2,3-b]pyridin-6- yl]-2-methyl-1H-indole-4-carboxamide
- Acetaldoxime 108 ⁇ L, 1.80 mmol
- Pd(OAc)2 (16.9 mg, 0.08 mmol)
- TPP 22.6 mg, 0.09 mmol
- Step 6 1-[4-(Benzylamino)-2-methyl-1H-pyrrolo[2,3-b]pyridin-6-yl]-2-methyl-1H- indole-4-carboxamide
- Step 7 1-[4-(Benzylamino)-2-methyl-1-[2-(morpholin-4-yl)-2-oxoethyl]-1H-pyrrolo[2,3- b]pyridin-6-yl]-2-methyl-1H-indole-4-carboxamide
- Efficacy of exemplary compounds in inhibition of p97 ATPase activity was evaluated using a p97 ATPase assay, such as the ADP-Glo TM assay (Promega).
- the p97 ATPase assay consisted of 10 ⁇ L reaction volumes in 384-well white, opaque plates. Compounds were prepared from DMSO stocks in assay buffer (40 mM Tris pH 7.5, 20 mM MgCl2, 0.1 mg/mL BSA) and preincubated with 110 nM p97 for 30 min at room temperature.
- Mia-Paca2 stable cells with CHOP Luciferase reporter were acquired from Signosis.
- HCT116 cells were cultured in McCoy's 5a Medium Modified supplemented with 10% FBS
- RPMI-8226 cells were cultured in RPMI-1640 supplemented with 10% FBS
- Mia-Paca2 CHOP cells were cultured in DMEM supplemented with 10% FBS, 2.5% horse serum and Hygromycin B. All cells were incubated at 37 ⁇ C, 5% CO2.
- Efficacy of exemplary compounds in cellular increase of CHOP expression The effect on p97 cellular function and unfolded protein response pathway was measured by CHOP expression as a readout.
- Mia-Paca2 cells stably expressing a CHOP Luciferase reporter were plated with 6,500 cells per well in 384-well white, TC-treated plates. Cells were incubated for 24 h before treatment with test compounds (0.1% DMSO in final volume) or vehicle (DMSO) for 8 h. The plate was then equilibrated to room temperature before addition of 1:1 volume of Bright-Glo® Luciferase reagent (Promega). The contents were mixed for 2 min on an orbital shaker before measuring luminescence on a microplate reader, such as a Cytation 5 microplate reader.
- % luminescence normalized to DMSO control as a function of compound concentration from triplicate wells was fit with a 4-parameter logistic curve using GraphPad Prism or CDD Vault to derive EC50 values.
- RPMI-8226 p97i sensitive
- HCT116 p97i insensitive cells were plated at 6,500 cells per well in 384-well white, TC-treated plates. After incubating for 24 h the cells were treated with vehicle (DMSO) or test compounds (0.1% DMSO in final volume) for 24 hrs. The plate was then equilibrated to room temperature before addition of a 1:1 volume of CellTiter-Glo® reagent (Promega).
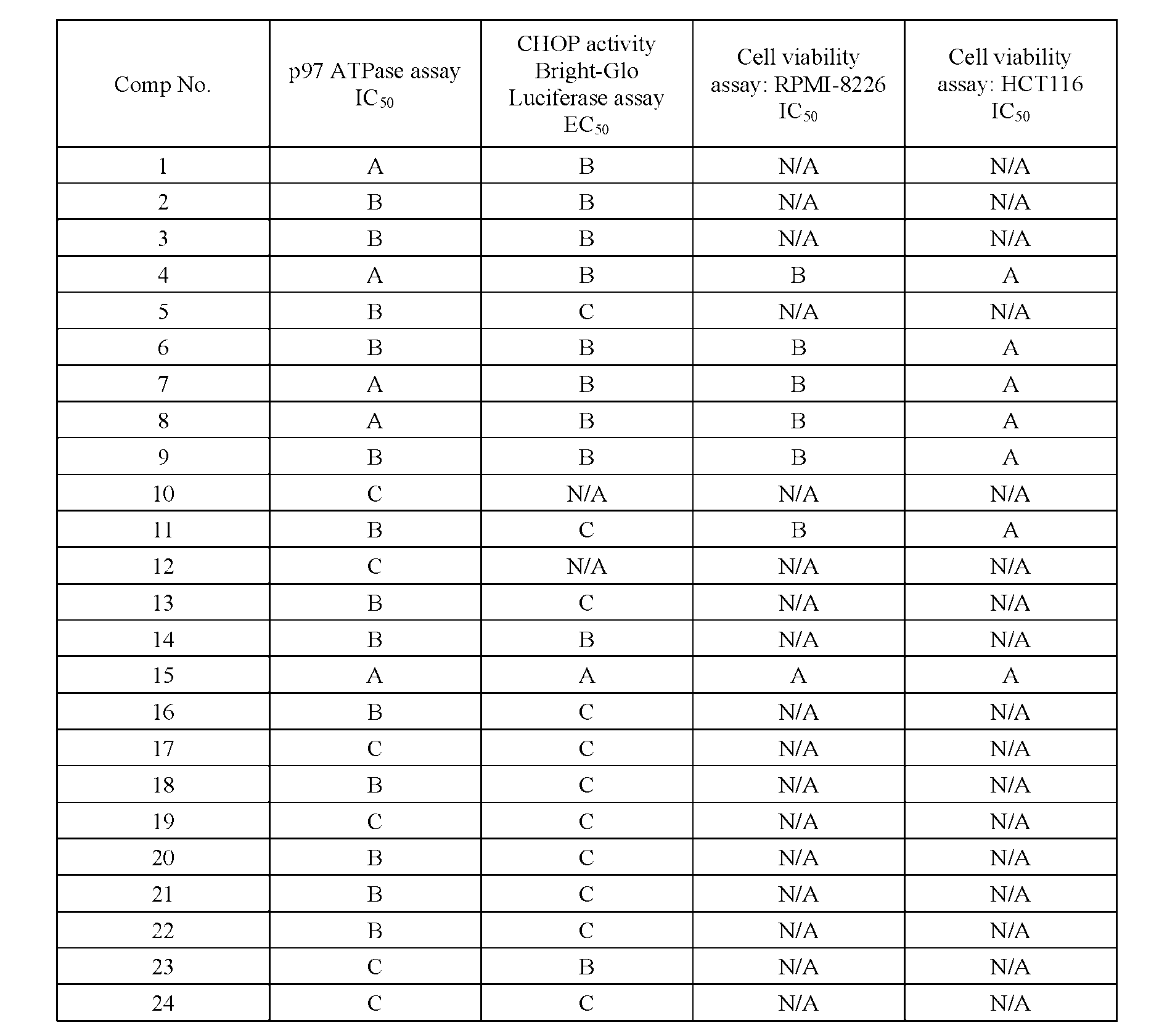
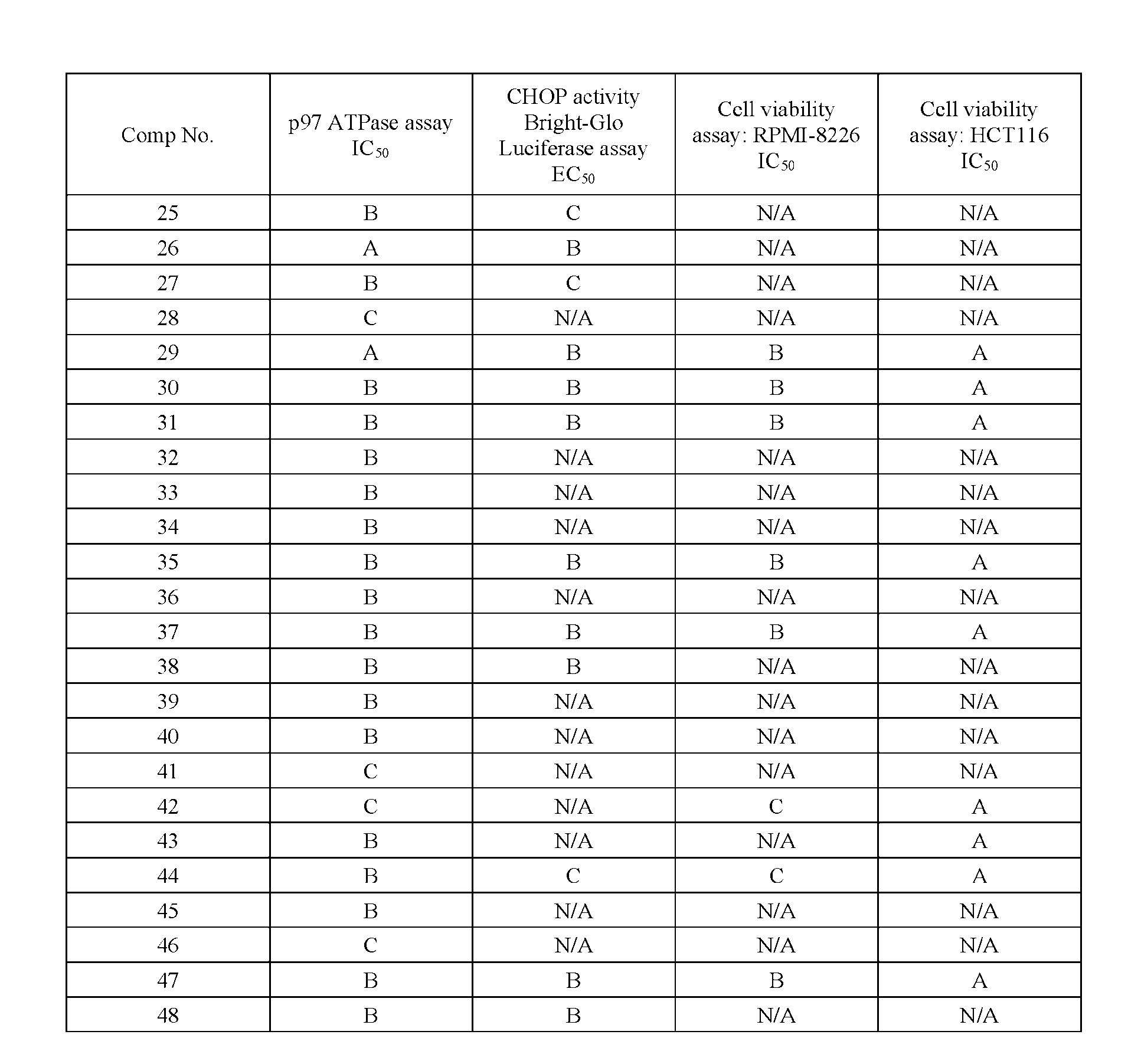
- % viability was calculated by normalizing to DMSO control (max) and no cells/background control (min). % viability as a function of compound concentration from triplicate wells was fit with a 4-parameter logistic curve using GraphPad Prism or CDD Vault to derive IC50 values. [000155] Results from these assays are summarized in Table 2 below.
- A indicates IC 50 of less than 1 ⁇ M
- B indicates IC 50 of between 1 ⁇ M to 10 ⁇ M
- C indicates IC50 of greater than 10 ⁇ M
- HCT116 CellTiter-Glo® cell viability assay “A” indicates IC 50 of greater than 10 ⁇ M. Table 2.
Landscapes
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biomedical Technology (AREA)
- Neurosurgery (AREA)
- Neurology (AREA)
- Hospice & Palliative Care (AREA)
- Psychiatry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Described herein are compounds and compositions useful for preventing and/or treating a disease or condition, e.g., a disease or condition associated with activity of p97 protein, e.g., cancer or a neurodegenerative disorder. In one aspect, the present disclosure provides a compound of formula (I) or a pharmaceutically acceptable salt thereof, wherein the variables R1, R2, R3, R4, R5, R6, R7, m and n are as defined herein.
Description
INHIBITORS OF P97 PROTEIN AND METHODS OF USE
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application claims the benefit and priority to U.S. Provisional Patent Application No. 63/512,750. filed July 10, 2023, the entire contents of which are incorporated herein by reference.
FIELD OF THE INVENTION
[0002] The invention relates generally to compounds and compositions useful for preventing and/or treating a disease or condition associated with activity of p97 protein, e.g., cancer or a neurodegenerative disorder.
BACKGROUND
[0003] The ubiquitin-proteasome system (UPS) and autophagy processes are crucial to the regulation of protein degradation, which maintains proteostasis, or protein homeostasis. Mutations in protein coding sequences and aneuploidy can cause disruptions to protein synthesis due to an increase in damaged or deregulated proteins, which in turn causes stress on the UPS system as the UPS system must clear out the higher frequency of these proteins. Dysregulation of protein homeostasis is often associated with cancer and the development of neurodegenerative disease (Cheng, J. et al. (2018) AGING CELL, 17(5): el280). In cancer cells, this dysregulation of protein homeostasis may occur as a result of dysregulated proliferation, causing the cancer cells to have a greater reliance on protein quality control (PQC) mechanisms to clear increased amounts of damaged or deregulated proteins of the cancer environment.
[0004] AAA unfoldases are a subset of AAA+ enzymes that assemble as hexamers to unfold substrate proteins. p97, also known as Valosin-containing protein, is an AAA unfoldase and plays an essential role in the protein homeostasis regulatory network. p97 is known to play a role in lysosomal degradation, but has also been shown to have roles in autophagy, endosomal sorting, regulating protein degradation at the outer mitochondrial membrane, and key chromatin-associated processes. These findings show that p97 not only has a role in lysosomal degradation, but also functions in protecting cells from protein stress and ensuring genome stability during proliferation (Meyer et al. (2012) NATURE CELL BIOL.. 14: 1 17-123).
[0005] One particular role of p97 is in assisting in the turnover of misfolded secretory proteins via the UPS (Carvalho, P. et al. (2006) CELL, 126: 361 -373). In this process, which
is known as ERAD (for endoplasmic reticulum-associated degradation), proteins that fail to fold within the ER are retrotranslocated in a p97-dependent manner into the cytoplasm to be degraded by the UPS (Y e, Y. et al. (2004) NATURE, 429: 841-847). p97 has also been shown to be required for the turnover of cytosolic substrates of the UPS (Janiesch, P. C. et al. (2007) NATURE CELL BIOL., 9: 379-390; Cao, K. et al. (2003) CELL, 115: 355-367; Fu, X. et al.
(2003) J. CELL BIOL., 163: 21-26). Furthermore, cancer cells may be particularly dependent on ERAD to clear the higher rate of damaged and dysregulated proteins (Boelens, J. et al. (2007) IN Vivo, 21: 215-226).
[0006] p97 is known to be highly expressed in multiple cancers (Y amamoto, S. et al.
(2005) ANN. SURG. ONCOL., 12: 925-934; Yamamoto, S. et al. (2004) CLIN. CANCER RES., 10: 5558-5565; Yamamoto, S. et al. (2004) ANN. SURG. ONCOL., 11 : 697-704; Yamamoto, S. et al. (2004) ANN. SURG. ONCOL., 11 : 165-172) suggesting that its role in maintaining protein homeostasis may be rate-limiting for the development of at least some cancers. Additionally, inhibition of p97 causes a problematic increase in protein concentration within the target cell, which often causes apoptosis or halts cellular grow th and mitosis. Despite the advances made to date in the treatment of cancers, neurodegenerative disorders and other diseases and conditions associated with an activity of p97, there is an ongoing need for additional therapies for treating such diseases and conditions.
SUMMARY
[0007] Described herein are compounds and compositions useful for preventing and/or treating a disease or condition associated with activity of p97 protein, e.g, cancer or a neurodegenerative disorder.
[0008] In one aspect, the present disclosure provides a compound of Formula (I):
or a pharmaceutically acceptable salt thereof, wherein the variables R1, R2, R3, R4, R3, R6, R7, m and n are as defined herein.
[0009] In another aspect, the present disclosure provides a compound of Formula (II):
or a pharmaceutically acceptable salt thereof, wherein R1, R2, R3 (including R8 and L1), R4, R5, R6, R7, m and n are as defined herein.
[00010] Also provided herein is a pharmaceutical composition comprising a compound as described herein or a pharmaceutically acceptable salt thereof and a pharmaceutically acceptable excipient.
[00011] Provided herein, in part, is a method of treating a disease or condition associated with p97 protein activity in a subject in need thereof, the method comprising administering to the subject a therapeutically effective amount of a compound as described herein or a pharmaceutically acceptable salt thereof or a pharmaceutical composition as described herein.
[00012] In some embodiments, the disease or condition associated with p97 protein activity is cancer, e.g., multiple myeloma or acute myeloid leukemia. In some embodiments, the disease or condition associated with p97 protein activity' is a neurodegenerative disorder, e.g., ALS, Charcot-Marie-Tooth disease type 2Y, and Inclusion body myopathy with early- onset Paget disease and frontotemporal dementia (IBMPFD).
[00013] Other objects and advantages will become apparent to those skilled in the art from a consideration of the ensuing Detailed Description, Examples, and Claims.
DETAILED DESCRIPTION
[00014] As generally described herein, the present disclosure provides compounds and compositions useful for preventing and/or treating a disease or condition described herein, e.g., a disease, disorder, or condition associated with activity of p97 protein, e.g.. cancer or a neurodegenerative disorder.
I. DEFINITIONS
Chemical Definitions
[00015] Definitions of specific functional groups and chemical terms are described in more detail below. The chemical elements are identified in accordance with the Periodic Table of the Elements, CAS version, Handbook of Chemistry and Physics, 75th Ed., inside cover, and specific functional groups are generally defined as described therein.
Additionally, general principles of organic chemistry, as well as specific functional moieties and reactivity, are described in Thomas Sorrell, Organic Chemistry, University Science Books, Sausalito, 1999; Smith and March, March ’s Advanced Organic Chemistry, 5th Edition, John Wiley & Sons, Inc., New York, 2001; Larock, Comprehensive Organic Transformations, VCH Publishers, Inc., New York, 1989; and Carruthers, Some Modern Methods of Organic Synthesis. 3rd Edition, Cambridge University Press, Cambridge, 1987.
[00016] The compounds described herein can comprise one or more asymmetric centers, and thus can exist in various isomeric forms, e.g., enantiomers and/or diastereomers. For example, the compounds described herein can be in the form of an individual enantiomer, diastereomer or geometric isomer, or can be in the form of a mixture of stereoisomers, including racemic mixtures and mixtures enriched in one or more stereoisomer. Isomers can be isolated from mixtures by methods known to those skilled in the art. including chiral high pressure liquid chromatography (HPLC) and the formation and crystallization of chiral salts; or preferred isomers can be prepared by asymmetric syntheses. See, for example, Jacques et al., ENANTIOMERS, RACEMATES AND RESOLUTIONS (Wiley Interscience, New York, 1981); When et al., TETRAHEDRON 33:2725 (1977); Eliel, STEREOCHEMISTRY OF CARBON COMPOUNDS (McGraw-Hill. NY, 1962); and Wilen, TABLES OF RESOLVING AGENTS AND OPTICAL RESOLUTIONS p. 268 (E.L. Eliel, Ed., Univ, of Notre Dame Press, Notre Dame, IN 1972). The invention additionally encompasses compounds described herein as individual isomers substantially free of other isomers, and alternatively, as mixtures of various isomers.
[00017] As used herein a pure enantiomeric compound is substantially free from other enantiomers or stereoisomers of the compound (i.e.. in enantiomeric excess). In other words, an “S” form of the compound is substantially free from the “R” form of the compound and is, thus, in enantiomeric excess of the “R” form. The term “enantiomerically pure” or “pure enantiomer” denotes that the compound comprises more than 75% by weight, more than 80% by weight, more than 85% by weight, more than 90% by weight, more than 91% by weight, more than 92% by weight, more than 93% by weight, more than 94% by weight, more than 95% by weight, more than 96% by weight, more than 97% by weight, more than 98% by weight, more than 98.5% by weight, more than 99% by weight, more than 99.2% by weight, more than 99.5% by weight, more than 99.6% by weight, more than 99.7% by weight, more than 99.8% by weight or more than 99.9% by weight, of the enantiomer. In certain embodiments, the weights are based upon total weight of all enantiomers or stereoisomers of the compound.
[00018] In the compositions provided herein, an enantiomerically pure compound can be present with other active or inactive ingredients. For example, a pharmaceutical composition comprising enantiomerically pure R-compound can comprise, for example, about 90% excipient and about 10% enantiomerically pure R-compound. In certain embodiments, the enantiomerically pure R-compound in such compositions can, for example, comprise, at least about 95% by weight R-compound and at most about 5% by weight S-compound, by total weight of the compound. For example, a pharmaceutical composition comprising enantiomerically pure S-compound can comprise, for example, about 90% excipient and about 10% enantiomerically pure S-compound. In certain embodiments, the enantiomerically pure S-compound in such compositions can, for example, comprise, at least about 95% by weight S-compound and at most about 5% by weight R-compound, by total weight of the compound. In certain embodiments, the active ingredient can be formulated with little or no excipient or carrier.
[00019] Compound described herein may also comprise one or more isotopic substitutions. For example, H may be in any isotopic form, including 1H, 2H (D or deuterium), and 3H (T or tritium); C may be in any isotopic form, including 12C, 13C, and 14C; O may be in any isotopic form, including 16O and 18O; F may be in any isotopic form, including 18F and 19F; and the like.
[00020] The following terms are intended to have the meanings presented therewith below and are useful in understanding the description and intended scope of the present invention.
When describing the invention, which may include compounds and pharmaceutically acceptable salts thereof, pharmaceutical compositions containing such compounds and methods of using such compounds and compositions, the following terms, if present, have the following meanings unless otherwise indicated. It should also be understood that when described herein any of the moieties defined forth below may be substituted with a variety of substituents, and that the respective definitions are intended to include such substituted moieties within their scope as set out below. Unless otherwise stated, the term “substituted” is to be defined as set out below. It should be further understood that the terms “groups” and “radicals” can be considered interchangeable when used herein. The articles “a” and “an” may be used herein to refer to one or to more than one (i.e. at least one) of the grammatical objects of the article. By way of example “an analogue” means one analogue or more than one analogue. [00021] When a range of values is listed, it is intended to encompass each value and sub– range within the range. For example, “C1–6 alkyl” is intended to encompass, C1, C2, C3, C4, C5, C6, C1–6, C1–5, C1–4, C1–3, C1–2, C2–6, C2–5, C2–4, C2–3, C3–6, C3–5, C3–4, C4–6, C4–5, and C5–6 alkyl. [00022] As used herein, “alkyl” refers to a radical of a straight–chain or branched saturated hydrocarbon group, e.g., having 1 to 20 carbon atoms (“C1–20 alkyl”). In some embodiments, an alkyl group has 1 to 10 carbon atoms (“C1–10 alkyl”). In some embodiments, an alkyl group has 1 to 9 carbon atoms (“C1–9 alkyl”). In some embodiments, an alkyl group has 1 to 8 carbon atoms (“C1–8 alkyl”). In some embodiments, an alkyl group has 1 to 7 carbon atoms (“C1–7 alkyl”). In some embodiments, an alkyl group has 1 to 6 carbon atoms (“C1–6 alkyl”). In some embodiments, an alkyl group has 1 to 5 carbon atoms (“C1–5 alkyl”). In some embodiments, an alkyl group has 1 to 4 carbon atoms (“C1–4 alkyl”). In some embodiments, an alkyl group has 1 to 3 carbon atoms (“C1–3 alkyl”). In some embodiments, an alkyl group has 1 to 2 carbon atoms (“C1-2 alkyl”). In some embodiments, an alkyl group has 1 carbon atom (“C1 alkyl”). Examples of C1–6 alkyl groups include methyl, ethyl, propyl, isopropyl, butyl, isobutyl, pentyl, hexyl, and the like. [00023] As used herein, “alkenyl” refers to a radical of a straight–chain or branched hydrocarbon group having from 2 to 20 carbon atoms, one or more carbon–carbon double bonds (e.g., 1, 2, 3, or 4 carbon–carbon double bonds), and optionally one or more carbon– carbon triple bonds (e.g., 1, 2, 3, or 4 carbon–carbon triple bonds) (“C2–20 alkenyl”).
[00024] As used herein, “alkynyl” refers to a radical of a straight–chain or branched hydrocarbon group having from 2 to 20 carbon atoms, one or more carbon–carbon triple bonds (e.g., 1, 2, 3, or 4 carbon–carbon triple bonds), and optionally one or more carbon– carbon double bonds (e.g., 1, 2, 3, or 4 carbon–carbon double bonds) (“C2–20 alkynyl”). [00025] As used herein, “alkylene,” refers to a divalent radical of an alkyl. When a range or number of carbons is provided for a particular “alkylene” group, it is understood that the range or number refers to the range or number of carbons in the linear carbon divalent chain. “Alkylene” group may be substituted or unsubstituted with one or more substituents as described herein. [00026] As used herein, “heteroalkylene” refers to a divalent radical of a heteroalkyl. The term “heteroalkyl” as used herein refers to an “alkyl” group in which at least one carbon atom has been replaced with a hetero atom, e.g., O, N, or S atom. The heteroalkyl may be, for example, an –O-C1-C10alkyl group, an -C1-C6alkylene-O-C1-C6alkyl group, or a C1-C6 alkylene-OH group. In certain embodiments, the “heteroalkyl” may be 2-10 membered heteroalkyl, indicating that the heteroalkyl contains from 2 to 10 atoms selected from the group consisting of carbon, oxygen, nitrogen, and sulfur. [00027] As used herein, “aryl” refers to a radical of a monocyclic or polycyclic (e.g., bicyclic or tricyclic) 4n+2 aromatic ring system (e.g., having 6, 10, or 14 π electrons shared in a cyclic array) having 6–14 ring carbon atoms and zero heteroatoms provided in the aromatic ring system (“C6–14 aryl”). In some embodiments, an aryl group has six ring carbon atoms (“C6 aryl”; e.g., phenyl). In some embodiments, an aryl group has ten ring carbon atoms (“C10 aryl”; e.g., naphthyl such as 1–naphthyl and 2–naphthyl). In some embodiments, an aryl group has fourteen ring carbon atoms (“C14 aryl”; e.g., anthracyl). [00028] As used herein, “heteroaryl” refers to a radical of a 5–10 membered monocyclic or bicyclic 4n+2 aromatic ring system (e.g., having 6 or 10 electrons shared in a cyclic array) having ring carbon atoms and 1–4 ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from nitrogen, oxygen and sulfur (“5–10 membered heteroaryl”). In heteroaryl groups that contain one or more nitrogen atoms, the point of attachment can be a carbon or nitrogen atom, as valency permits. Heteroaryl bicyclic ring systems can include one or more heteroatoms in one or both rings. “Heteroaryl” includes ring systems wherein the heteroaryl ring, as defined above, is fused with one or more carbocyclyl or heterocyclyl groups wherein the point of attachment is on the heteroaryl ring, and in such instances, the number of ring members continue to designate the number of
ring members in the heteroaryl ring system. “Heteroaryl” also includes ring systems wherein the heteroaryl ring, as defined above, is fused with one or more aryl groups wherein the point of attachment is either on the aryl or heteroaryl ring, and in such instances, the number of ring members designates the number of ring members in the fused (aryl/heteroaryl) ring system. Bicyclic heteroaryl groups wherein one ring does not contain a heteroatom (e.g., indolyl, quinolinyl, carbazolyl, and the like) the point of attachment can be on either ring, i.e., either the ring bearing a heteroatom (e.g., 2–indolyl) or the ring that does not contain a heteroatom (e.g., 5–indolyl). [00029] In some embodiments, a heteroaryl group is a 5–10 membered aromatic ring system having ring carbon atoms and 1–4 ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur (“5–10 membered heteroaryl”). In some embodiments, a heteroaryl group is a 5–8 membered aromatic ring system having ring carbon atoms and 1–4 ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur (“5–8 membered heteroaryl”). In some embodiments, a heteroaryl group is a 5–6 membered aromatic ring system having ring carbon atoms and 1–4 ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur (“5–6 membered heteroaryl”). In some embodiments, the 5–6 membered heteroaryl has 1–3 ring heteroatoms selected from nitrogen, oxygen, and sulfur. In some embodiments, the 5–6 membered heteroaryl has 1–2 ring heteroatoms selected from nitrogen, oxygen, and sulfur. In some embodiments, the 5–6 membered heteroaryl has 1 ring heteroatom selected from nitrogen, oxygen, and sulfur. [00030] Exemplary 5–membered heteroaryl groups containing one heteroatom include, without limitation, pyrrolyl, furanyl and thiophenyl. Exemplary 5–membered heteroaryl groups containing two heteroatoms include, without limitation, imidazolyl, pyrazolyl, oxazolyl, isoxazolyl, thiazolyl, and isothiazolyl. Exemplary 5–membered heteroaryl groups containing three heteroatoms include, without limitation, triazolyl, oxadiazolyl, and thiadiazolyl. Exemplary 5–membered heteroaryl groups containing four heteroatoms include, without limitation, tetrazolyl. Exemplary 6–membered heteroaryl groups containing one heteroatom include, without limitation, pyridinyl. Exemplary 6–membered heteroaryl groups containing two heteroatoms include, without limitation, pyridazinyl, pyrimidinyl, and pyrazinyl. Exemplary 6–membered heteroaryl groups containing three or four heteroatoms include, without limitation, triazinyl and tetrazinyl, respectively. Exemplary 7–membered
heteroaryl groups containing one heteroatom include, without limitation, azepinyl, oxepinyl, and thiepinyl. Exemplary 5,6–bicyclic heteroaryl groups include, without limitation, indolyl, isoindolyl, indazolyl, benzotriazolyl, benzothiophenyl, isobenzothiophenyl, benzofuranyl, benzoisofuranyl, benzimidazolyl, benzoxazolyl, benzisoxazolyl, benzoxadiazolyl, benzthiazolyl, benzisothiazolyl, benzthiadiazolyl, indolizinyl, and purinyl. Exemplary 6,6– bicyclic heteroaryl groups include, without limitation, naphthyridinyl, pteridinyl, quinolinyl, isoquinolinyl, cinnolinyl, quinoxalinyl, phthalazinyl, and quinazolinyl. [00031] Examples of representative heteroaryls include the following: . w
y , , , , ; independently hydrogen, C1-C8 alkyl, C3-C10 carbocyclyl, 4-10 membered heterocyclyl, C6-C10 aryl, and 5- 10 membered heteroaryl. [00032] As used herein, “carbocyclyl” or “carbocyclic” refers to a radical of a non– aromatic cyclic hydrocarbon group having from 3 to 10 ring carbon atoms (“C3–10 carbocyclyl”) and zero heteroatoms in the non–aromatic ring system. In some embodiments, a carbocyclyl group has 3 to 8 ring carbon atoms (“C3–8 carbocyclyl”). [00033] The term “cycloalkyl” refers to a monovalent saturated cyclic, bicyclic, or bridged cyclic (e.g., adamantyl) hydrocarbon group of 3-12, 3-8, 4-8, or 4-6 carbons, referred to herein, e.g., as “C4-8cycloalkyl,” derived from a cycloalkane. Exemplary cycloalkyl groups include, but are not limited to, cyclohexanes, cyclopentanes, cyclobutanes and cyclopropanes. Unless specified otherwise, cycloalkyl groups are optionally substituted at one or more ring positions with, for example, alkanoyl, alkoxy, alkyl, haloalkyl, alkenyl, alkynyl, amido,
amidino, amino, aryl, arylalkyl, azido, carbamate, carbonate, carboxy, cyano, cycloalkyl, ester, ether, formyl, halogen, haloalkyl, heteroaryl, heterocyclyl, hydroxyl, imino, ketone, nitro, phosphate, phosphonato, phosphinato, sulfate, sulfide, sulfonamido, sulfonyl or thiocarbonyl. Cycloalkyl groups can be fused to other cycloalkyl, aryl, or heterocyclyl groups. In certain embodiments, the cycloalkyl group is not substituted, i.e., it is unsubstituted. [00034] As used herein, “heterocyclyl” or “heterocyclic” refers to a radical of a 3– to 10– membered non–aromatic ring system having ring carbon atoms and 1 to 4 ring heteroatoms, wherein each heteroatom is independently selected from nitrogen, oxygen, sulfur, boron, phosphorus, and silicon (“3–10 membered heterocyclyl”). In heterocyclyl groups that contain one or more nitrogen atoms, the point of attachment can be a carbon or nitrogen atom, as valency permits. A heterocyclyl group can either be monocyclic (“monocyclic heterocyclyl”) or a fused, bridged or spiro ring system such as a bicyclic system (“bicyclic heterocyclyl”), and can be saturated or can be partially unsaturated. Heterocyclyl bicyclic ring systems can include one or more heteroatoms in one or both rings. “Heterocyclyl” also includes ring systems wherein the heterocyclyl ring, as defined above, is fused with one or more carbocyclyl or cycloalkyl groups wherein the point of attachment is either on the carbocyclyl, cycloalkyl, or heterocyclyl ring, or ring systems wherein the heterocyclyl ring, as defined above, is fused with one or more aryl or heteroaryl groups, wherein the point of attachment is on the heterocyclyl ring, and in such instances, the number of ring members continue to designate the number of ring members in the heterocyclyl ring system. [00035] As used herein, “heterocyclylene” refers to a divalent radical of a heterocyclyl. [00036] In some embodiments, a heterocyclyl group is a 5–10 membered non–aromatic ring system having ring carbon atoms and 1–4 ring heteroatoms, wherein each heteroatom is independently selected from nitrogen, oxygen, sulfur, boron, phosphorus, and silicon (“5–10 membered heterocyclyl”). In some embodiments, a heterocyclyl group is a 5–8 membered non–aromatic ring system having ring carbon atoms and 1–4 ring heteroatoms, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur (“5–8 membered heterocyclyl”). In some embodiments, a heterocyclyl group is a 5–6 membered non–aromatic ring system having ring carbon atoms and 1–4 ring heteroatoms, wherein each heteroatom is independently selected from nitrogen, oxygen, and sulfur (“5–6 membered heterocyclyl”). In some embodiments, the 5–6 membered heterocyclyl has 1–3 ring heteroatoms selected from nitrogen, oxygen, and sulfur. In some embodiments, the 5–6 membered heterocyclyl has 1–2
ring heteroatoms selected from nitrogen, oxygen, and sulfur. In some embodiments, the 5–6 membered heterocyclyl has one ring heteroatom selected from nitrogen, oxygen, and sulfur. [00037] Exemplary 3–membered heterocyclyl groups containing one heteroatom include, without limitation, azirdinyl, oxiranyl, thiorenyl. Exemplary 4–membered heterocyclyl groups containing one heteroatom include, without limitation, azetidinyl, oxetanyl and thietanyl. Exemplary 5–membered heterocyclyl groups containing one heteroatom include, without limitation, tetrahydrofuranyl, dihydrofuranyl, tetrahydrothiophenyl, dihydrothiophenyl, pyrrolidinyl, dihydropyrrolyl and pyrrolyl–2,5–dione. Exemplary 5– membered heterocyclyl groups containing two heteroatoms include, without limitation, dioxolanyl, oxasulfuranyl, disulfuranyl, and oxazolidin-2-one. Exemplary 5–membered heterocyclyl groups containing three heteroatoms include, without limitation, triazolinyl, oxadiazolinyl, and thiadiazolinyl. Exemplary 6–membered heterocyclyl groups containing one heteroatom include, without limitation, piperidinyl, tetrahydropyranyl, dihydropyridinyl, and thianyl. Exemplary 6–membered heterocyclyl groups containing two heteroatoms include, without limitation, piperazinyl, morpholinyl, dithianyl, dioxanyl. Exemplary 6– membered heterocyclyl groups containing two heteroatoms include, without limitation, triazinanyl. Exemplary 7–membered heterocyclyl groups containing one heteroatom include, without limitation, azepanyl, oxepanyl and thiepanyl. Exemplary 8–membered heterocyclyl groups containing one heteroatom include, without limitation, azocanyl, oxecanyl and thiocanyl. Exemplary 5-membered heterocyclyl groups fused to a C6 aryl ring (also referred to herein as a 5,6-bicyclic heterocyclic ring) include, without limitation, indolinyl, isoindolinyl, dihydrobenzofuranyl, dihydrobenzothienyl, benzoxazolinonyl, and the like. Exemplary 6-membered heterocyclyl groups fused to an aryl ring (also referred to herein as a 6,6-bicyclic heterocyclic ring) include, without limitation, tetrahydroquinolinyl, tetrahydroisoquinolinyl, and the like. [00038] “Hetero” when used to describe a compound or a group present on a compound means that one or more carbon atoms in the compound or group have been replaced by a nitrogen, oxygen, or sulfur heteroatom. Hetero may be applied to any of the hydrocarbyl groups described above such as alkyl, e.g., heteroalkyl; carbocyclyl, e.g., heterocyclyl; aryl, e.g,. heteroaryl; and the like having from 1 to 5, and particularly from 1 to 3 heteroatoms. [00039] As used herein, “halo” or “halogen” refers to fluoro (F), chloro (Cl), bromo (Br) and iodo (I). In certain embodiments, the halo group is either fluoro or chloro.
[00040] As used herein, “haloalkyl” refers to an alkyl group substituted with one or more halogen atoms. [00041] As used herein, “oxo” refers to -C=O. [00042] In general, the term “substituted,” whether preceded by the term “optionally” or not, means that at least one hydrogen present on a group (e.g., a carbon or nitrogen atom) is replaced with a permissible substituent, e.g., a substituent which upon substitution results in a stable compound, e.g., a compound which does not spontaneously undergo transformation such as by rearrangement, cyclization, elimination, or other reaction. Unless otherwise indicated, a “substituted” group has a substituent at one or more substitutable positions of the group, and when more than one position in any given structure is substituted, the substituent is either the same or different at each position. [00043] Unless otherwise indicated, an “optionally substituted” group may have a suitable substituent at each substitutable position of the group, and when more than one position in any given structure may be substituted with more than one substituent selected from a specified group, the substituent may be either the same or different at each position. Combinations of substituents envisioned under this invention are preferably those that result in the formation of stable or chemically feasible compounds. [00044] Suitable monovalent substituents on a substitutable carbon atom of an “optionally substituted” group (such as an alkyl, alkenyl, alkynyl, alkylene, alkenylene, alkynylene or the carbon atom of a carbocyclyl, aryl, heterocyclyl or heteroaryl) are independently, for example, halogen; -(CH2)0-4R ^; -(CH2)0-4OR ^; -O-(CH2)0-4C(O)OR°; -(CH2)0-4CH(OR ^)2; -(CH2)0-4SR ^; -(CH2)0-4Ph, which may be substituted with R°; -(CH2)0-4O(CH2)0-1Ph, which may be substituted with R°; -CH=CHPh, which may be substituted with -NO2; -CN; -N3; - (CH2)0-4N(R ^)2; -(CH2)0-4N(R ^)C(O)R ^; -N(R ^)C(S)R ^; -(CH2)0-4N(R ^) C(O)NR ^2; -N(R ^)C(S)NR ^2; -(CH2)0-4N(R ^)C(O)OR ^; -N(R ^)N(R ^)C(O)R ^; -N(R ^)N(R ^)C(O)NR ^2; -N(R ^)N(R ^)C(O)OR ^; -(CH2)0-4C(O)R ^; -C(S)R ^; -(CH2)0-4C(O)OR ^; -(CH2)0-4C(O)SR ^; -(CH2)0-4C(O)OSiR ^3; -(CH2)0-4OC(O)R ^; -OC(O)(CH2)0-4SR ^-, SC(S)SR°; -(CH2)0-4SC(O)R ^; -(CH2)0-4C(O)NR ^2; -C(S)NR ^2; -C(S)SR°; -(CH2)0-4OC(O)NR ^2; -C(O)N(OR ^)R ^; -C(O)C(O)R ^; -C(O)CH2C(O)R ^;-C(NOR ^)R ^; -(CH2)0-4SSR ^; -(CH2)0-4S(O)2R ^; -(CH2)0-4S(O)2OR ^; -(CH2)0-4OS(O)2R ^; -S(O)2NR ^2; -(CH2)0-4S(O)R ^; -N(R ^)S(O)2NR ^2; -N(R ^)S(O)2R ^; -N(OR ^)R ^; -C(NH)NR ^2; -P(O)2R ^; -P(O)R ^2;
-OP(O)R ^2; -OP(O)(OR ^)2; -SiR3; -(C1-4 straight or branched alkylene)O-N(R ^)2; or -(C1-4 straight or branched alkylene)C(O)O-N(R ^)2, where each R ^ may be substituted as defined below and is independently hydrogen, C1-6 alkyl, C1-6 alkenyl, C1-6 alkynyl, -CH2Ph, -O(CH2)0-1Ph, or a 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur, or, notwithstanding the definition above, two independent occurrences of R ^, taken together with their intervening atom(s), form a 3-12-membered saturated, partially unsaturated, or aryl mono- or bicyclic ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur, which may be substituted as defined below. [00045] Suitable monovalent substituents on R ^ (or the ring formed by taking two independent occurrences of R ^ together with their intervening atoms), are independently, for example, deuterium, halogen, -(CH2)0-2R ^, -(haloR ^), -(CH2)0-2OH, -(CH2)0-2OR ^, -(CH2)0-2CH(OR ^)2; -O(haloR ^), -CN, -N3, -(CH2)0-2C(O)R ^, -(CH2)0-2C(O)OH, -(CH2)0-2C(O)OR ^, -(CH2)0-2SR ^, -(CH2)0-2SH, -(CH2)0-2NH2, -(CH2)0-2NHR ^, -(CH2)0-2NR ^2, -NO2, -SiR ^3, -OSiR ^3, -C(O)SR ^, -(C1-4 straight or branched alkylene)C(O)OR ^, or -SSR ^, where each R ^ is unsubstituted or where preceded by “halo” is substituted only with one or more halogens, and is independently selected from C1-4 alkyl, C1-4 alkenyl, C1-4 alkynyl, -CH2Ph, -O(CH2)0-1Ph, or a 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur. Suitable divalent substituents on a saturated carbon atom of R ^ include =O and =S. [00046] Suitable divalent substituents on a saturated carbon atom of an “optionally substituted” group include the following: =O, =S, =NNR*2, =NNHC(O)R*, =NNHC(O)OR*, =NNHS(O)2R*, =NR*, =NOR*, -O(C(R* 2))2-3O-, or -S(C(R* 2))2-3S-, where each independent occurrence of R* is selected from hydrogen, C1-6 alkyl, C1-6 alkenyl, C1-6 alkynyl, and carbocyclyl, which may be substituted as defined below, or an unsubstituted 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur. Suitable divalent substituents that are bound to vicinal substitutable carbons of an “optionally substituted” group include: -O(C(R*)2)2-3O-, where each independent occurrence of R* is selected from hydrogen, C1-6 alkyl, C1-6 alkenyl, C1-6 alkynyl, and carbocyclyl, which may be substituted as defined below, or an unsubstituted 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur.
[00047] Suitable substituents on the alkyl, alkenyl, alkynyl, or carbocyclyl group of R* include halogen, -R ^, -(haloR ^), -OH, -OR ^, -O(haloR ^), -CN, -C(O)OH, -C(O)OR ^, -NH2, -NHR ^, -NR ^2, or -NO2, where each R ^ is unsubstituted or where preceded by “halo” is substituted only with one or more halogens, and is independently C1-6 alkyl, C1-6 alkenyl, C1-6 alkynyl, -CH2Ph, -O(CH2)0-1Ph, or a 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur. [00048] Suitable substituents on a substitutable nitrogen of an “optionally substituted” group include -R†, -NR†2, -C(O)R†, -C(O)OR†, -C(O)C(O)R†, -C(O)CH2C(O)R†, -S(O)2R†, -S(O)2NR† 2, -C(S)NR† 2, -C(NH)NR† 2, or -N(R†)S(O)2R†; where each R† is independently hydrogen, C1-6 alkyl, C1-6 alkenyl, C1-6 alkynyl, which may be substituted as defined below, unsubstituted -OPh, or an unsubstituted 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur, or, notwithstanding the definition above, two independent occurrences of R†, taken together with their intervening atom(s) form an unsubstituted 3-12-membered saturated, partially unsaturated, or aryl mono- or bicyclic ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur. In certain embodiments, it should be understood that a substitutable nitrogen can be substituted with four substituents (or have four bonds to less than four substituents) such that the nitrogen atom will be positively charged or its cation, i.e., N+. Such substitution of a substitutable nitrogen can be of a ring nitrogen, for example, a ring nitrogen of a heterocyclic group containing nitrogen as a ring atom. In particular embodiments, where one of the substituents of a cationic nitrogen atom is a hydroxyl group, the hydroxyl group can be deprotonated and represented by a negatively charged oxygen atom, i.e., O-. In such cases, a general formula for the substitution can be represented by RN+(O-)(R’)R’’, where R, R’ and R’’ represent carbon or other atoms, groups and/or moieties to which the nitrogen atom is bound. [00049] Suitable substituents on the alkyl, alkenyl, alkynyl, or carbocyclyl group of R† are independently, for example, halogen, -R ^, -(haloR ^), -OH, -OR ^, -O(haloR ^), -CN, -C(O)OH, -C(O)OR ^, -NH2, -NHR ^, -NR ^2, or -NO2, where each R ^ is unsubstituted or where preceded by “halo” is substituted only with one or more halogens, and is independently C1-6 alkyl,C1-6 alkenyl, C1-6 alkynyl, -CH2Ph, -O(CH2)0-1Ph, or a 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur.
[00050] These and other exemplary substituents are described in more detail in the Detailed Description, Examples, and Claims. The invention is not intended to be limited in any manner by the above exemplary listing of substituents. Other Definitions [00051] In the claims articles such as “a,” “an,” and “the” may mean one or more than one unless indicated to the contrary or otherwise evident from the context. Claims or descriptions that include “or” between one or more members of a group are considered satisfied if one, more than one, or all of the group members are present in, employed in, or otherwise relevant to a given product or process unless indicated to the contrary or otherwise evident from the context. Also contemplated are embodiments in which exactly one member of the group is present in, employed in, or otherwise relevant to a given product or process. Also contemplated are embodiments in which more than one, or all of the group members are present in, employed in, or otherwise relevant to a given product or process. [00052] Where the use of the term “about” is before a quantitative value, the present invention also includes the specific quantitative value itself, unless specifically stated otherwise. As used herein, the term “about” refers to a ±10% variation from the nominal value unless otherwise indicated or inferred. [00053] As a general matter, compositions specifying a percentage are by weight unless otherwise specified. Further, if a variable is not accompanied by a definition, then the previous definition of the variable controls. [00054] The term “pharmaceutically acceptable” refers to those compounds, materials, compositions, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio. [00055] The terms “pharmaceutically acceptable carrier” and “pharmaceutically acceptable excipient” are used interchangeably herein and refer to buffers, carriers, and excipients suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio. Pharmaceutically acceptable carriers include any of the standard pharmaceutical carriers, such as a phosphate buffered saline solution, water, emulsions (e.g., such as an oil/water or water/oil emulsions), and various
types of wetting agents. The compositions also can include stabilizers and preservatives. For examples of carriers, stabilizers, and adjuvants, see e.g., Adeboye Adejare, Remington: The Science and Practice of Pharmacy (23d ed.2020). [00056] The term “pharmaceutically acceptable salt” refers to those salts which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of humans and lower animals without undue toxicity, irritation, allergic response and the like, and are commensurate with a reasonable benefit/risk ratio. Pharmaceutically acceptable salts are well known in the art. For example, Berge et al., describes pharmaceutically acceptable salts in detail in J. PHARMACEUTICAL SCIENCES (1977) 66:1–19, and Gould, Salt selection for basic drugs, INTERNATIONAL JOURNAL OF PHARMACEUTICS, 33 (1986) 201-217. Pharmaceutically acceptable salts of the compounds of this invention include those derived from suitable inorganic and organic acids and bases. Examples of pharmaceutically acceptable, nontoxic acid addition salts are salts of an amino group formed with inorganic acids such as hydrochloric acid, hydrobromic acid, phosphoric acid, sulfuric acid and perchloric acid or with organic acids such as acetic acid, oxalic acid, maleic acid, tartaric acid, citric acid, succinic acid or malonic acid or by using other methods used in the art such as ion exchange. Other pharmaceutically acceptable salts include adipate, alginate, ascorbate, aspartate, benzenesulfonate, benzoate, bisulfate, borate, butyrate, camphorate, camphorsulfonate, citrate, cyclopentanepropionate, digluconate, dodecylsulfate, ethanesulfonate, formate, fumarate, glucoheptonate, glycerophosphate, gluconate, hemisulfate, heptanoate, hexanoate, hydroiodide, 2–hydroxy–ethanesulfonate, lactobionate, lactate, laurate, lauryl sulfate, malate, maleate, malonate, methanesulfonate, 2–naphthalenesulfonate, nicotinate, nitrate, oleate, oxalate, palmitate, pamoate, pectinate, persulfate, 3–phenylpropionate, phosphate, picrate, pivalate, propionate, stearate, succinate, sulfate, tartrate, thiocyanate, p–toluenesulfonate, undecanoate, valerate salts, and the like. Pharmaceutically acceptable salts derived from appropriate bases include alkali metal, alkaline earth metal, ammonium and N+(C1–4alkyl)4 salts. Representative alkali or alkaline earth metal salts include sodium, lithium, potassium, calcium, magnesium, and the like. Further pharmaceutically acceptable salts include, when appropriate, nontoxic ammonium, quaternary ammonium, and amine cations formed using counterions such as halide, hydroxide, carboxylate, sulfate, phosphate, nitrate, lower alkyl sulfonate, and aryl sulfonate. [00057] As used herein, a “subject” to which administration is contemplated includes, but is not limited to, humans (e.g., a male or female of any age group, e.g., a pediatric subject
(e.g., infant, child, adolescent) or adult subject (e.g., young adult, middle–aged adult or senior adult)) and/or a non-human animal, e.g., a mammal such as primates (e.g., cynomolgus monkeys, rhesus monkeys), cattle, pigs, horses, sheep, goats, rodents, cats, and/or dogs. In certain embodiments, the subject is a human. In certain embodiments, the subject is a non- human animal. The terms “human,” “patient,” and “subject” are used interchangeably herein. [00058] Disease, disorder, and condition are used interchangeably herein. [00059] As used herein, “treat,” “treating,” and “treatment” refer to the treatment of a disease, disorder, or symptom or manifestation of such in a subject, e.g., in a human. This includes: (a) preventing a disease or disorder, (b) inhibiting the disease, disorder, etc., i.e., slowing or arresting its progress or development; and (b) relieving the disease, disorder, etc., i.e., causing regression of the disease state. As used herein, “prevent”, “preventing” and “prevention” refer to causing a disease, disorder, or symptom or manifestation of such not to occur for at least a period of time in at least some subjects. [00060] The term “effective amount” as used herein refers to the amount of a compound, e.g., a compound or composition described herein, sufficient to elicit a beneficial or desired result or biological effect. As will be appreciated by those of ordinary skill in this art, the effective amount of a compound or composition described herein may vary depending on such factors as the desired biological endpoint, the pharmacokinetics of the compound, the disease being treated, the mode of administration, and the age, weight, health, and condition of the subject. An effective amount can be administered in one or more administrations, applications, or dosages and is not intended to be limited to a particular formulation or administration route. For example, an effective amount may be an amount of a compound or composition sufficient to achieve one or more of the following: (i) inhibit an activity of p97 protein; or (ii) treat or prevent a disease or condition associated with an activity of p97 protein, e.g., cancer, or a symptom or manifestation thereof. [00061] As used herein, and unless otherwise specified, a “therapeutically effective amount” of a compound or composition is an amount sufficient to provide a therapeutic benefit in the treatment of a disease or condition, or to delay or minimize one or more symptoms associated with the disease, disorder or condition. As will be appreciated by those of ordinary skill in this art, a therapeutically effective amount of a compound or composition described herein may vary depending on such factors as the desired biological endpoint, the pharmacokinetics of the compound, the disease being treated, the mode of administration, and the age, weight, health, and condition of the subject. A therapeutically effective amount
of a compound or composition can be administered in one or more administrations, applications, or dosages and is not intended to be limited to a particular formulation or administration route. A therapeutically effective amount of a compound or composition also includes an amount of therapeutic agent, alone or in combination with other therapies, which provides a therapeutic benefit in the treatment of the disease, disorder or condition. In addition, a therapeutically effective amount can encompass an amount of a compound or composition that improves overall therapy, reduces or avoids symptoms or causes of disease or condition, or enhances the therapeutic efficacy of another therapeutic agent.
[00062] At various places in the present specification, variable or parameters are disclosed in groups or in ranges. It is specifically intended that the description include each and every individual subcombination of the members of such groups and ranges. For example, an integer in the range of 0 to 40 is specifically intended to individually disclose 0, 1, 2. 3, 4, 5, 6, 7, 8. 9. 10. 1 1. 12. 13. 14. 15, 16, 17, 18, 19, 20, 21, 22, 23, 24. 25. 26. 27. 28. 29. 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, and 40, and an integer in the range of 1 to 20 is specifically intended to individually disclose 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, and 20.
[00063] Throughout the description, where compounds or compositions are described as having, including, or comprising specific components, or where processes and methods are described as having, including, or comprising specific steps, it is contemplated that, additionally, there are compounds or compositions of the present invention that consist essentially of, or consist of, the recited components, and that there are processes and methods according to the present invention that consist essentially of, or consist of, the recited processing steps. Similarly, throughout the description, where compounds or compositions are described as consisting essentially of specific components, or where processes and methods are described as consisting essentially of specific steps, it is contemplated that, additionally, there are compounds or compositions of the present invention that consist of the recited components, and that there are processes and methods according to the present invention that consist of the recited processing steps.
[00064] In the application, where an element or component is said to be included in and/or selected from a list of recited elements or components, it should be understood that the element or component can be any one of the recited elements or components, or the element or component can be selected from a group consisting of two or more of the recited elements or components.
[00065] Further, it should be understood that elements and/or features of a composition or a method described herein can be combined in a variety of ways without departing from the spirit and scope of the present invention, whether explicit or implicit herein. For example, where reference is made to a particular compound, that compound can be used in various embodiments of compositions of the present invention and/or in methods of the present invention, unless otherwise understood from the context. In other words, within this application, embodiments have been described and depicted in a way that enables a clear and concise application to be written and drawn, but it is intended and will be appreciated that embodiments may be variously combined or separated without parting from the present teachings and invention(s). For example, it will be appreciated that all features described and depicted herein can be applicable to all aspects of the invention(s) described and depicted herein.
[00066] It should be understood that the expression “at least one of’ includes individually each of the recited objects after the expression and the various combinations of two or more of the recited objects unless otherwise understood from the context and use. The expression “and/or” in connection with three or more recited objects should be understood to have the same meaning unless otherwise understood from the context.
[00067] The use of the term “include.” “includes,” “including,” “have.” “has,” “having,” “contain.” “contains.” or “containing,” including grammatical equivalents thereof, should be understood generally as open-ended and non-limiting, for example, not excluding additional unrecited elements or steps, unless otherwise specifically stated or understood from the context.
[00068] It should be understood that the order of steps or order for performing certain actions is immaterial so long as the present invention remain operable. Moreover, two or more steps or actions may be conducted simultaneously.
[00069] The use of any and all examples, or exemplary language herein, for example, “such as” or “including,” is intended merely to illustrate better the present invention and does not pose a limitation on the scope of the invention unless claimed. No language in the specification should be construed as indicating any non-claimed element as essential to the practice of the present invention.
[00070] The claims encompasses all variations, combinations, and permutations in which one or more limitations, elements, clauses, and descriptive terms from one or more of the
listed claims is introduced into another claim. For example, any claim that is dependent on another claim can be modified to include one or more limitations found in any other claim that is dependent on the same base claim. Where elements are presented as lists, e.g., in Markush group format, each subgroup of the elements is also disclosed, and any element(s) can be removed from the group. II. COMPOUNDS AND COMPOSITIONS [00071] In one aspect, the present disclosure provides a compound of Formula (I): :
each R1 is independently selected from the group consisting of halogen, C1-6 alkyl, C1- 6 alkoxy, C1-6 haloalkyl, and C1-6 alkylene-OH; R2 is selected from hydrogen and C1-6 alkyl; R3 is selected from the group consisting of hydrogen, 3-10 membered heterocyclyl, - C(O)-(3-10 membered heterocyclyl), and C1-6 alkyl optionally substituted with a substituent selected from the group consisting of C1-6 alkoxy, -CN, -C(O)N(Ra)2, -NRbS(O)2C1-6 alkyl, - S(O)2N(Rb)2, 5-6 membered heteroaryl, 3-10 membered heterocyclyl, and -C(O)-(3-10 membered heterocyclyl), wherein the 3-10 membered heterocyclyl is optionally substituted with 1, 2, or 3 substituents each independently selected from the group consisting of C1-6 alkyl, oxo, -C(O)-C1-6 alkyl, -S(O)2C1-6 alkyl, and -N(Rb)2, R4 is selected from hydrogen and C1-6 alkyl; R5 is selected from hydrogen and C1-6 alkyl; R6 is selected from hydrogen and C1-6 alkyl;
each R7 is independently selected from the group consisting of hydrogen, C1-6 alkyl, - (C1-6 alkylene)-OH, -(C1-6 alkylene)-C1-6 alkoxy, -(C1-6 alkylene)-N(Rb)2, C1-6 alkoxy, -CN, - C(O)N(Ra)2, and halogen; each Ra is independently selected from the group consisting of hydrogen, C1-6 alkyl, and C1-6 alkoxy; each Rb is independently selected from hydrogen or C1-6 alkyl; n is 1 or 2; and m is selected from 0, 1, and 2. [00072] In another aspect, the present disclosure provides a compound of Formula (II):
each R1 is independently selected from the group consisting of halogen, C1-6 alkyl, C1- 6 alkoxy, C1-6 haloalkyl, and C1-6 alkylene-OH; R2 is selected from hydrogen and C1-6 alkyl; R3 is selected from the group consisting of hydrogen, 3-10 membered heterocyclyl, - C(O)-(3-10 membered heterocyclyl), -L1-R8, and C1-6 alkyl optionally substituted with a substituent selected from the group consisting of C1-6 alkoxy, -CN, -C(O)N(Ra)2, - NRbS(O)2C1-6 alkyl, -S(O)2N(Rb)2, 5-6 membered heteroaryl, 3-10 membered heterocyclyl, and -C(O)-(3-10 membered heterocyclyl), wherein the 3-10 membered heterocyclyl is optionally substituted with 1, 2, or 3 substituents each independently selected from the group consisting of C1-6 alkyl, oxo, -C(O)-C1-6 alkyl, -S(O)2C1-6 alkyl, and -N(Rb)2, R4 is selected from hydrogen and C1-6 alkyl;
R5 is selected from hydrogen and C1-6 alkyl; R6 is selected from hydrogen and C1-6 alkyl; each R7 is independently selected from the group consisting of hydrogen, C1-6 alkyl, - (C1-6 alkylene)-OH, -(C1-6 alkylene)-C1-6 alkoxy, -(C1-6 alkylene)-N(Rb)2, C1-6 alkoxy, -CN, - C(O)N(Ra)2, and halogen; L1 is a linker moiety; R8 is a moiety derived from an immunomodulatory imide drug; each Ra is independently selected from the group consisting of hydrogen, C1-6 alkyl, and C1-6 alkoxy; each Rb is independently selected from hydrogen or C1-6 alkyl; n is 1 or 2; and m is selected from 0, 1, and 2. [00073] In some embodiments, R2 is hydrogen. [00074] In other embodiments, R3 is selected from the group consisting of hydrogen, methyl,
[00075] ce ta e bod e ts, s 1
bodiments, R3 is hydrogen.
[00077] In some embodiments, R3 is -L1-R8. [00078] In certain embodiments, L1 is a linker moiety selected from the group consisting of: -(C1-10 alkylene)-C(O)-, -(2-10 membered heteroalkylene)-C(O)-, -(2-10 membered heteroalkylene)-C(O)-(C1-10 alkylene), and -(C1-10 alkylene)-(3-7 membered heterocyclylene)- (C1-10 alkylene). [00079] In other embodiments, R8 is a moiety derived from an immunomodulatory imide drug. Immunomodulatory imide drugs are a class of drugs that adjust immune responses which contain an imide group. In some embodiments, the immunomodulatory imide drug is a thalidomide analog, e.g., a thalidomide analog selected from the group consisting of pomalidomide and lenalidomide. In certain embodiments, R8 is selected from: . ethyl. In other embodiments, R4 is
methyl. In some embodiments, R4 is hydrogen. [00081] In certain embodiments, R5 is hydrogen. [00082] In some embodiments, R6 is selected from hydrogen and methyl. In certain embodiments, R6 is methyl. [00083] In other embodiments, R6 is at the 2-position of the indolyl. [00084] In some embodiments, each R7 is independently selected from the group consisting of hydrogen, C1-6 alkyl, -(C1-6 alkylene)-OH, -(C1-6 alkylene)-N(Rb)2, -CN, and - C(O)NH2. In some embodiments, R7 is -C(O)NH2. [00085] In other embodiments, R7 is at the 4-position of the indolyl. [00086] In some embodiments, n is 1. In other embodiments, n is 2.
[00087] In certain embodiments, m is 0. In some embodiments, m is 1. In other embodiments, m is 2. [00088] In some embodiments, R1 is selected from the group consisting of halogen, -CH2- OH, CHF2, CF3, -methyl, and -OMe. [00089] In some embodiments, the compound of Formula (I) or (II) is selected from Table 1 or a pharmaceutically acceptable salt thereof. Table 1. Compound No. Structure
III. COMPOUND SYNTHESIS
General Synthetic Schemes
[00090] Exemplary methods for preparing compounds described herein are illustrated in the following synthetic schemes. These schemes are given for the purpose of illustrating the scope of the disclosure, and should not be regarded in any manner as limiting the scope or the spirit of the claimed invention.
[00091] The synthetic route illustrated in Scheme 1 depicts an exemplary procedure for preparing a compound of Formula (I) or (II).
SCHEME 1
1. Optional functionalization of R7
(e.g., reaction of ON with MeC=NOH, Pd(OAc)2)
2. PG removal
3. R3 introduction
(e.g., R'C(O)CH2CI)
[00092] A commercially available 4-6-dihalosubstituted-7-azaindole is typically used as a starting material or a starting material may be prepared from a commercially available material by methods known to one of ordinary skill in the art. The nitrogen of the 5- membered ring of the 7-azaindole core is reacted with a protecting group under reaction conditions suitable for the protecting group (e.g., sodium hydride as a base in DMF solvent with SEM-chloride or benzenesulfonyl chloride as protecting group reagents). Optionally. R4 of the protected compound is functionalized (e.g.. a reaction with lithium di-isopropylamide and Mel). The protected compound is then reacted with an optionally substituted benzylamine (commercially available or may be prepared from a commercially available material by methods known to one of ordinary skill in the art) regioselectively to provide compound A. A palladium catalyzed addition of an optionally substituted indole (typically 4- cyanoindole), which is commercially available or may be prepared from a commercially available material by methods know n to one of ordinary skill in the art, provides compound B. Optionally, R7 of compound B is functionalized (e.g., a reaction of the nitrile with acetaldoxime in the presence of palladium acetate affords the primary’ amide). Protecting group removal (e.g., TFA for SEM) optionally followed by R3 group introduction (e.g., 2- chloroacetyl group, with a simple example being 2-chloroacetamide) provides a compound of Formula (I). When R3 is -L'-R8, a linker group is introduced at R3, and then the pendant functionality of the linker (typically an acid or an amide) is reacted with an appropriately functionalized immunomodulatory imide compound (such as pomalidomide or lenalidomide) via standard amide coupling chemistry' methods to provide a compound of Formula (II).
[00093] In addition to the general synthetic schemes, specific syntheses of the compounds disclosed herein are set forth in the various Examples.
IV. PHARMACEUTICAL COMPOSITIONS AND ROUTES OF ADMINISTRATION
[00094] The compounds provided in accordance with the present disclosure, e.g., a compound of Formula (I) or (II) or a pharmaceutically acceptable salt thereof, are usually administered in the form of pharmaceutical compositions. Provided herein are pharmaceutical compositions that contain, as the active ingredient, one or more of the compounds described, or a pharmaceutically acceptable salt or ester thereof, and one or more pharmaceutically acceptable excipients, carriers, including inert solid diluents and fillers, diluents, including sterile aqueous solution and various organic solvents, permeation enhancers, solubilizers and adjuvants. The pharmaceutical compositions may be
administered alone or in combination with other therapeutic agents. Such compositions are prepared in a manner well known in the pharmaceutical art (see, e.g., Adeboye Adejare, Remington: The Science and Practice of Pharmacy (23 d ed. 2020)).
[00095] In certain embodiments, a pharmaceutical composition may contain formulation materials for modifying, maintaining or preserving, for example, the pH, osmolarity, viscosity, clarity, color, isotonicity, odor, sterility, stability, rate of dissolution or release, adsorption or penetration of the composition. In such embodiments, suitable formulation materials include, but are not limited to, amino acids (such as glycine, glutamine, asparagine, arginine or lysine); antimicrobials; antioxidants (such as ascorbic acid, sodium sulfite or sodium hydrogen-sulfite); buffers (such as borate, bicarbonate, Tris-HCl, citrates, phosphates or other organic acids); bulking agents (such as mannitol or glycine); chelating agents (such as ethylenediamine tetraacetic acid (EDTA)); complexing agents (such as caffeine, polyvinylpyrrolidone, beta-cyclodextrin or hydroxypropyl-beta-cyclodextrin); fillers; monosaccharides; disaccharides; and other carbohydrates (such as glucose, mannose or dextrins); proteins (such as serum albumin, gelatin or immunoglobulins); coloring, flavoring and diluting agents; emulsifying agents; hydrophilic polymers (such as polyvinylpyrrolidone); low molecular weight polypeptides; salt-forming counterions (such as sodium); preservatives (such as benzalkonium chloride, benzoic acid, salicylic acid, thimerosal, phenethyl alcohol, methylparaben, propylparaben, chlorhexidine, sorbic acid or hydrogen peroxide); solvents (such as glycerin, propylene glycol or polyethylene glycol); sugar alcohols (such as mannitol or sorbitol); suspending agents; surfactants or wetting agents (such as pluronics. PEG. sorbitan esters, polysorbates such as polysorbate 20, polysorbate, triton, tromethamine, lecithin, cholesterol, tyloxapal); stability enhancing agents (such as sucrose or sorbitol); tonicity enhancing agents (such as alkali metal halides, preferably sodium or potassium chloride, mannitol sorbitol); delivery vehicles; diluents; and/or excipients (see Adeboye Adejare (2020) supra).
[00096] The pharmaceutical compositions may be administered in either single or multiple doses by any of the accepted modes of administration of agents having similar utilities, including, orally, topically, by rectal, buccal, intranasal or transdermal routes, intravenously, intraperitoneally, parenterally, intramuscularly, subcutaneously, or as an inhalant, or via an impregnated or coated device, e.g., a stent.
[00097] One mode for administration is parenteral, particularly by injection. The forms in which the novel compounds or compositions described herein may be incorporated for
administration by injection include aqueous or oil suspensions, or emulsions, with sesame oil, com oil, cottonseed oil, or peanut oil, as well as elixirs, mannitol, dextrose, or a sterile aqueous solution, and similar pharmaceutical vehicles. Aqueous solutions in saline are also conventionally used for injection, but less preferred in the context of the present invention. Ethanol, glycerol, propylene glycol, liquid polyethylene glycol, and the like (and suitable mixtures thereof), cyclodextrin derivatives, and vegetable oils may also be employed. The proper fluidity can be maintained, for example, by the use of a coating, such as lecithin, by the maintenance of the required particle size in the case of dispersion and by the use of surfactants. The prevention of the action of microorganisms can be brought about by various antibacterial and antifungal agents, for example, parabens, chlorobutanol, phenol, sorbic acid, thimerosal. and the like.
[00098] Sterile injectable solutions are prepared by incorporating a compound according to the present invention in the required amount in the appropriate solvent with various other ingredients as enumerated above, as required, followed by filtered sterilization. Generally, dispersions are prepared by incorporating the various sterilized active ingredients into a sterile vehicle which contains the basic dispersion medium and the required other ingredients from those enumerated above. In the case of sterile powders for the preparation of sterile injectable solutions, the preferred methods of preparation are vacuum-drying and freeze- drying techniques which yield a powder of the active ingredient plus any additional desired ingredient from a previously sterile-filtered solution thereof.
[00099] Oral administration is another route for administration of compounds in accordance with the invention. Administration may be via capsule or enteric coated tablets, or the like. In making the pharmaceutical compositions that include at least one compound described herein, the active ingredient is usually diluted by an excipient and/or enclosed within such a carrier that can be in the form of a capsule, sachet, paper or other container. When the excipient serves as a diluent, it can be in the form of a solid, semi-solid, or liquid material, which acts as a vehicle, carrier or medium for the active ingredient. Thus, the compositions can be in the form of tablets, pills, powders, lozenges, sachets, cachets, elixirs, suspensions, emulsions, solutions, syrups, aerosols (as a solid or in a liquid medium), ointments containing, for example, up to 10% by w eight of the active compound, soft and hard gelatin capsules, sterile injectable solutions, and sterile packaged powders.
[000100] Some examples of suitable excipients include lactose, dextrose, sucrose, sorbitol, mannitol, starches, gum acacia, calcium phosphate, alginates, tragacanth, gelatin, calcium
silicate, microcrystalline cellulose, polyvinylpyrrolidone, cellulose, sterile water, syrup, and methyl cellulose. The formulations can additionally include: lubricating agents such as talc, magnesium stearate, and mineral oil; wetting agents; emulsifying and suspending agents; preserving agents such as methyl and propylhydroxy-benzoates; sweetening agents; and flavoring agents. [000101] The compounds or compositions described herein, e.g., a compound of Formula (I) or (II) or a pharmaceutically acceptable salt thereof, can be formulated so as to provide quick, sustained or delayed release of the active ingredient after administration to the patient by employing procedures known in the art. Controlled release drug delivery systems for oral administration include osmotic pump systems and dissolutional systems containing polymer- coated reservoirs or drug-polymer matrix formulations. Examples of controlled release systems are given in U.S. Pat. Nos.3,845,770; 4,326,525; 4,902,514; and 5,616,345. Similarly, the compounds or compositions described herein can be delivered by transdermal delivery devices (“patches”). Such transdermal patches may be used to provide continuous or discontinuous infusion of the compounds of the present invention in controlled amounts. The construction and use of transdermal patches for the delivery of pharmaceutical agents is well known in the art (see, e.g., U.S. Pat. Nos.5,023,252, 4,992,445 and 5,001,139). Such patches may be constructed for continuous, pulsatile, or on demand delivery of pharmaceutical agents. [000102] The compositions are preferably formulated in a unit dosage form. The term “unit dosage forms” refers to physically discrete units suitable as unitary dosages for human subjects and other mammals, each unit containing a predetermined quantity of active material calculated to produce the desired therapeutic effect, in association with a suitable pharmaceutical excipient (e.g., a tablet, capsule, ampoule). The compounds are generally administered in a pharmaceutically effective amount. For oral administration, each dosage unit may contain from 1 mg to 2 g (e.g., 1 mg to 1 g, 1 mg to 500 mg, 1 mg to 100 mg) of a compound described herein, and for parenteral administration, from 0.1 to 700 mg (e.g., from 0.1 mg to 300 mg, from 0.1 mg to 100 mg, from 0.1 mg to 10 mg) of a compound described herein. The reference to compound includes the compound either alone or in combination with a pharmaceutically acceptable salt. It will be understood, however, that the amount of the compound actually administered usually will be determined by a physician, in the light of the relevant circumstances, including the condition to be treated, the chosen route of
administration, the actual compound administered and its relative activity , the age, weight, and response of the individual patient, the severity’ of the patient’s symptoms, and the like.
[000103] For preparing solid compositions such as tablets, the principal active ingredient is mixed with a pharmaceutical excipient to form a solid preformulation composition containing a homogeneous mixture of a compound of the present invention. When referring to these preformulation compositions as homogeneous, it is meant that the active ingredient is dispersed evenly throughout the composition so that the composition may be readily subdivided into equally effective unit dosage forms such as tablets, pills and capsules.
[000104] The tablets or pills of the present invention may be coated or otherwise compounded to provide a dosage form affording the advantage of prolonged action, or to protect from the acid conditions of the stomach. For example, the tablet or pill can comprise an inner dosage and an outer dosage component, the latter being in the form of an envelope over the former. The two components can be separated by an enteric layer that serves to resist disintegration in the stomach and permit the inner component to pass intact into the duodenum or to be delayed in release. A variety of materials can be used for such enteric layers or coatings, such materials including a number of polymeric acids and mixtures of polymeric acids with such materials as shellac, cetyl alcohol, and cellulose acetate.
[000105] Compositions for inhalation or insufflation include solutions and suspensions in pharmaceutically acceptable, aqueous or organic solvents, or mixtures thereof, and powders. The liquid or solid compositions may contain suitable pharmaceutically acceptable excipients as described supra. Preferably, the compositions are administered by the oral or nasal respiratory route for local or systemic effect. Compositions in preferably pharmaceutically acceptable solvents may be nebulized by use of inert gases. Nebulized solutions may be inhaled directly from the nebulizing device or the nebulizing device may be attached to a facemask tent, or intermittent positive pressure breathing machine. Solution, suspension, or powder compositions may be administered, preferably orally or nasally, from devices that deliver the formulation in an appropriate manner.
[000106] In some embodiments, provided herein is a pharmaceutical composition comprising a disclosed compound, e.g., a compound of Formula (I) or (II), or pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable carrier.
V. METHODS OF USE
[000107] The compounds and compositions described herein can be used in a method of inhibiting p97 protein and a method of treating a disease or condition associated with an activity of p97 protein, e.g., cancer or a neurodegenerative disorder.
[000108] One aspect of the present disclosure includes methods of inhibiting p97 protein in a subject in need thereof. In another aspect, the present disclosure provides a method of treating a disease or condition associated with an activity of p97 protein, e.g., cancer, in a subject in need thereof. In some embodiments, the treatment of the subject in need thereof comprises administering a compound of the present disclosure (e.g., a compound of Formula (I) or (II) or a pharmaceutically acceptable salt thereof) to the subject in need thereof. In certain embodiments the disease or condition is cancer.
[000109] The compounds and compositions described above and herein can be used to treat an abnormal cell growth or a neuroplastic disorder in a subject in need thereof. Abnormal cell growth, as used herein and unless otherwise indicated, refers to cell growth that is independent of normal regulatory mechanisms (e.g., loss of contact inhibition).
[000110] Abnormal cell growth can refer to cell growth of epithelial cells (e.g, carcinomas, adenocarcinomas), mesenchymal cells (e.g. sarcomas (e.g. Ewing’s sarcoma)), hematopoetic cells (e.g, lymphomas, leukemias, myelodysplasias) or other cells (e.g, melanoma, mesothelioma, and other tumors of unknown origin). A “neoplastic disorder” is a disease or disorder characterized by cells that have the capacity for autonomous growth or replication, e.g., an abnormal state or condition characterized by proliferative cell growth. An abnormal mass of tissue as a result of abnormal cell growth or division, or a neoplasm, can be benign, pre-malignant or malignant. Exemplary neoplastic disorders include: carcinoma, sarcoma, metastatic disorders (e.g, tumors arising from prostate, colon, lung, breast and liver origin), hematopoietic neoplastic disorders, e.g, leukemias, metastatic tumors. Treatment with the compound may be in an amount effective to ameliorate at least one symptom of the neoplastic disorder, e.g, reduced cell proliferation, reduced tumor mass, etc.
[000111] The methods and compositions described herein may be useful in the prevention and treatment of a cancer, including, e.g, solid tumors, soft tissue tumors, and metastases thereof. The disclosed methods are also useful in treating non-solid cancers. Exemplary' solid tumors include malignancies (e.g, sarcomas, adenocarcinomas, and carcinomas) of the various organ systems, such as those of lung, lymphoid, gastrointestinal (e.g, colon, rectum.
or colorectum), brain, pancreas, genitourinary tracts (e.g, renal, urothelial, or testicular tumors), pharynx, prostate, and ovary. Exemplary adenocarcinomas include colorectal cancers, renal-cell carcinoma, liver cancer (e.g.. hepatocellular carcinoma), lung cancer (e.g., non-small cell lung cancer (NSCLC)), pancreatic (e.g., metastatic pancreatic adenocarcinoma) and cancer of the small intestine. In some embodiments, the cancer can be colorectal cancer, NSCLC, or pancreatic cancer. In certain embodiments, the cancer may include brain metastasis. In some embodiments, the cancer is multiple myeloma. In other embodiments, the cancer is acute myeloid leukemia.
[000112] The cancer can include mesothelioma; neurofibromatosis; e.g., neurofibromatosis type 2, neurofibromatosis type 1 , renal cancer; lung cancer, non-small cell lung cancer; thyroid cancer; a nervous system tumor; schwannoma; meningioma; schwannomatosis; neuroma acoustic; adenoid cystic carcinoma; ependymoma; ependymal tumors, or any other tumor which exhibits decreased merlin expression and/or mutation, and/or deletion and/or promotor hypermethylation of the NF-2 gene. In some embodiments, the cancer is renal cancer.
[000113] The cancer can include cancers characterized as comprising cancer stem cells, cancer associated mesenchymal cells, or tumor initiating cancer cells. The cancer can include cancers that have been characterized as being enriched with cancer stem cells, cancer associated mesenchymal cells, or tumor initiating cancer cells (e.g.. a tumor enriched with cells that have undergone an epithelial -to-mesenchymal transition or a metastatic tumor).
[000114] The cancer can be a primary tumor, i.e., located at the anatomical site of tumor growth initiation. The cancer can also be metastatic, /.<?., appearing at least a second anatomical site other than the anatomical site of tumor growth initiation. The cancer can be a recurrent cancer, i.e., cancer that returns following treatment, and after a period of time in which the cancer w as undetectable. The recurrent cancer can be anatomically located locally to the original tumor, e.g., anatomically near the original tumor; regionally to the original tumor, e.g., in a lymph node located near the original tumor; or distantly to the original tumor, e.g., anatomically in a region remote from the original tumor. In some embodiments, the cancer can be brain or cerebrospinal fluid (CSF)-related metastasis.
[000115] The cancer can also include for example, but is not limited to, epithelial cancers, lung, pancreatic, colorectal (e.g.. metastatic colorectal), prostate, head and neck, melanoma, acute myelogenous leukemia, and glioblastoma. Exemplary breast cancers include triple negative breast cancer, basal-like breast cancer, claudin-low' breast cancer, invasive,
inflammatory, metaplastic, and advanced HER-2 positive or ER-positive cancers resistant to therapy.
[000116] The cancer can also include lung adenocarcinoma, colorectal cancer (CRC), uveal melanoma, uterine endometrioid carcinoma, bladder urothelial carcinoma, breast invasive lobular carcinoma, cervical squamous cell carcinoma, cutaneous melanoma, endocervical adenocarcinoma, hepatocellular carcinoma, pancreatic adenocarcinoma, biphasic type pleural mesothelioma, renal clear cell carcinoma, renal clear cell carcinoma, stomach adenocarcinoma, tubular stomach adenocarcinoma, head and neck squamous cell carcinoma, uterine carcinosarcoma, or uterine malignant mixed Mullerian tumor.
[000117] In some embodiments, the cancer is unresectable or metastatic melanoma, melanoma with lymph node involvement or metastatic disease who have undergone complete resection, metastatic non-small cell lung cancer and progression on or after platinum-based chemotherapy, metastatic small cell lung cancer with progression after platinum-based chemotherapy and at least one other line of therapy, advanced renal cell carcinoma who have received prior anti angiogenic therapy, advanced renal cell carcinoma, classical Hodgkin lymphoma, recurrent or metastatic squamous cell carcinoma of the head and neck with disease progression on or after a platinum-based therapy, urothelial carcinoma (e.g., locally advanced or metastatic), microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) metastatic colorectal cancer, or hepatocellular carcinoma.
[000118] Other cancers include primary mediastinal large B-cell lymphoma, mantle cell lymphoma, gastric cancer, esophageal cancer, cervical cancer, merkel cell carcinoma, abdominal, esophagus, glioma, tongue, neuroblastoma, osteosarcoma, retinoblastoma, Wilm’s tumor, multiple myeloma, skin, lymphoma, blood and bone marrow cancers (e.g., advanced hematological malignancies, leukemia, e.g.. acute myeloid leukemia (e.g, primary’ or secondary), acute lymphoblastic leukemia, acute lymphocytic leukemia. T cell leukemia, hematological malignancies, advanced myeloproliferative disorders, myelodysplastic syndrome, relapsed or refractory’ multiple myeloma, advanced myeloproliferative disorders), retinal, bladder, cervical, kidney, endometrial, meningioma, uterine, lung, small cell lung cancer, non-small cell lung cancer, nasopharyngeal carcinoma, neuroblastoma, solid tumor, hematologic malignancy, squamous cell carcinoma, testicular, thyroid, mesothelioma, brain vulval, sarcoma, intestine, oral, endocrine, salivary, spermatocyte seminoma, sporadic medullary thyroid carcinoma, non-proliferating testes cells, cancers related to malignant mast cells, non-Hodgkin’s lymphoma, and diffuse large B cell lymphoma.
[000119] In some embodiments, the tumor is a solid tumor. In some embodiments, the solid tumor is locally advanced or metastatic. In some embodiments, the solid tumor is refractory (e.g., resistant) after standard therapy.
[000120] Methods described herein can reduce, ameliorate or altogether eliminate the disorder, and/or its associated symptoms, to keep it from becoming worse, to slow the rate of progression, or to minimize the rate of recurrence of the disorder once it has been initially eliminated (e.g., to avoid a relapse). A suitable dose and therapeutic regimen may vary' depending upon the specific compounds, combinations, and/or pharmaceutical compositions used and the mode of delivery of the compounds, combinations, and/or pharmaceutical compositions. In some embodiments, the method increases the average length of survival, increases the average length of progression-free survival, and/or reduces the rate of recurrence, of subjects treated with the combinations described herein in a statistically significant manner.
[000121] In some embodiments, the cancer is bone cancer, pancreatic cancer, uterine cancer, ovarian cancer (e.g, unresectable low-grade ovarian, advanced or metastatic ovarian cancer), rectal cancer, cancer of the anal region, stomach cancer, colon cancer, breast cancer (e.g., triple-negative breast cancer (e.g, breast cancer which does not express the genes for the estrogen receptor, progesterone receptor, and Her2/neu)), uterine cancer, carcinoma of the fallopian tubes, carcinoma of the endometrium, carcinoma of the cervix, carcinoma of the vagina, carcinoma of the vulva, Hodgkin’s Disease, cancer of the small intestine, cancer of the endocrine system, cancer of the thyroid gland, cancer of the parathyroid gland, cancer of the adrenal gland, sarcoma of soft tissue, cancer of the urethra, cancer of the penis, prostate cancer, chronic or acute leukemia, lymphocytic lymphomas, cancer of the ureter, renal cell carcinoma, carcinoma of the renal pelvis, neoplasms of the central nervous system (CNS), primary CNS lymphoma, spinal axis tumors, brain stem glioma, pituitary’ adenoma, mesothelioma (e.g, malignant pleural mesothelioma, e.g., surgical resectable malignant pleural mesothelioma) or a combination of one or more of the foregoing cancers.
[000122] Diseases in which p97 activity is increased through mutation or overexpression are also targets for therapeutic intervention.
[000123] In some embodiments, a disease or condition associated with an activity of p97 protein is a neurodegenerative disorder. The neurodegenerative disorders include, for example, inclusion body myopathy (IBM), Paget's disease of the bone (PDB), frontotemporal dementia (FTD) and amyotrophic lateral sclerosis (ALS)). In certain embodiments,
neurodegenerative diseases include, for example, amyotrophic lateral sclerosis (ALS), Charcot-Marie-Tooth disease type 2Y, and Inclusion body myopathy with early-onset Paget disease and frontotemporal dementia (IBMPFD). Neurodegenerative disorders also include subjects having p97 mutations, and symptoms manifesting as, for example, Parkinsonism, ataxia, cataracts, dilated cardiomyopathy, hepatic fibrosis, and hearing loss.
[000124] In other embodiments, a disease or condition associated with an activity of p97 protein is an infectious disease. The compound of the disclosure (e.g, a compound of Formula (I) or (II)) may be useful for antiviral therapy for certain viruses, e.g, poliovirus, herpes simplex virus (HSV), cytomegalovirus (CMV), or influenza.
[000125] The compound of the disclosure (e.g., a compound of Formula (I) or (II)) may be included in a pharmaceutical formulation such as those disclosed herein, and may be administered in any pharmaceutically acceptable manner, including methods of administration described herein.
[000126] An effective amount of a compound useful in the methods of the present invention, for example in a pharmaceutical composition, may be administered to a subject in need thereof by any of a number of well-known methods for administering pharmaceutical compounds. The compound may be administered systemically or locally. In one embodiment, the compound is administered intravenously. For example, the compounds useful in the methods of the present technology may be administered via rapid intravenous bolus injection. In some embodiments, the compound is administered as a constant rate intravenous infusion. The compound may also be administered orally, topically, intranasally, intramuscularly, subcutaneously, or transdermally. Other routes of administration include intracerebroventricularly or intrathecally. Intracerebroventricularly refers to administration into the ventricular system of the brain. Intrathecally refers to administration into the space under the arachnoid membrane of the spinal cord.
[000127] The compounds useful in the methods of the present technology may also be administered to a subject in need thereof by sustained or controlled release, as is known in the art. Sustained release administration is a method of drug delivery to achieve a certain level of the drug over a particular period of time. The level typically is measured by serum or plasma concentration by, for example, nuclear magnetic resonance spectroscopy (NMR) or liquid chromatography mass spectrometry (LCMS).
EXAMPLES
[000128] In order that the invention described herein may be more fully understood, the following examples are set forth. The synthetic and biological examples described in this application are offered to illustrate the compounds, pharmaceutical compositions and methods provided herein and are not to be construed in any way as limiting their scope.
[000129] The compounds provided herein can be prepared from readily available starting materials using the following general methods and procedures. It will be appreciated that where typical or preferred process conditions (e g., reaction temperatures, times, mole ratios of reactants, solvents, pressures, etc.) are given, other process conditions can also be used unless otherwise stated. Optimal reaction conditions may vary with the particular reactants or solvent used, but such conditions can be determined by one skilled in the art by routine optimization.
[000130] Additionally, as will be apparent to those skilled in the art, conventional protecting groups may be necessary' to prevent certain functional groups from undergoing undesired reactions. The choice of a suitable protecting group for a particular functional group as well as suitable conditions for protection and deprotection are well known in the art. For example, numerous protecting groups, and their introduction and removal, are described in T. W. Greene and P. G. M. Wuts, Protecting Groups in Organic Synthesis, Second Edition, Wiley, New York, 1991, and references cited therein.
[000131] The compounds provided herein may be isolated and purified by known standard procedures. Such procedures include recrystallization, filtration, flash chromatography, trituration, high pressure liquid chromatography (HPLC). or supercritical fluid chromatography (SFC). Note that flash chromatography may either be performed manually or via an automated system. The compounds provided herein may be characterized by known standard procedures, such as nuclear magnetic resonance spectroscopy (NMR) or liquid chromatography mass spectrometry (LCMS). NMR chemical shifts are reported in part per million (ppm) and are generated using methods well known to those of skill in the art.
[000132] All temperatures are in degrees Celsius (°C) and are uncorrected. Silica gel chromatography was performed on Teledyne Isco instruments using pre-packaged disposable SiCh stationary phase columns with eluent flow-rate ranges of 15 to 200 mL/min, UV
detection (254 and 280 nm). Reverse phase purification was carried out using Cl 8 columns, UV detection (214 and 254 nm).
[000133] The analytical HPLC chromatograms were performed using an Agilent 1100/1200 series instrument with DAD detector (190 nm to 300 nm) and a thermostatted column compartment set to 35 °C. The mass spectra were recorded with a Waters SQD detector with the desolvation temperature set to 500 °C, the source temperature set to 150 °C and the desolvation gas flow set to 1000 L/h. The mass spectrometer was equipped with an electrospray ion source (ESI) operated in a positive, or negative, ion mode and was set to scan between m/z 100-750 with a scan time of 0.2 s. Products and intermediates were analyzed by HPLC/MS on one of 4 methods (A05/A50/B05/B50). Method A05 is a 3.50 minute method using a Kinetex EVO Cl 8 (5 pM, 4.6 x 50 mm) column, a flow rate of 2.20 mL/min, and a low pH buffer gradient of 5% to 100% of ACN in H2O (10 mM ammonium formate, adjusted to pH 4 with formic acid). The % ACN is kept stable at 5% from 0.00-0.50 minutes and gradually increased to 100% from 0.50-2.50 minutes. From 2.50-3.50 minutes, the % ACN is kept stable at 100%. Method A50 is a 3.50 minute method using a Kinetex EVO C18 (5 pM, 4.6 x 50 mm) column, a flow rate of 2.20 mL/min. and a low pH buffer gradient of 5% to 100% of ACN in H2O (10 mM ammonium formate, adjusted to pH 4 with formic acid). The % ACN is kept stable at 50% from 0.00-0.50 minutes and gradually increased to 100% from 0.50-2.50 minutes. From 2.50-3.50 minutes, the % ACN is kept stable at 100%. Method B05 is a 3.50 minute method using a Gemini NX-C18 (5 pM, 4.6 x 50 mm) column, a flow rate of 2.20 mL/min, and a high pH buffer gradient of 5 % to 100 % of ACN in H2O (10 mM ammonium bicarbonate, adjusted to pH 10 with ammonium hydroxide). The % ACN is kept stable at 5% from 0.00-0.50 minutes and gradually increased to 100% from 0.50-2.50 minutes. From 2.50-3.50 minutes, the % ACN is kept stable at 100%. Method B50 is a 3.50 minute method using a Gemini NX-C18 (5 pM, 4.6 x 50 mm) column, a flow rate of 2.20 mL/min. and a high pH buffer gradient of 50% to 100% of ACN in H2O (10 mM ammonium bicarbonate, adjusted to pH 10 with ammonium hydroxide).
The % ACN is kept stable at 50% from 0.00-0.50 minutes and gradually increased to 100% from 0.50-2.50 minutes. From 2.50-3.50 minutes, the % ACN is kept stable at 100%.
[000134] The analytical QA/QC chromatograms were also performed using a Waters AcquiW UPLC instrument with DAD detector (200 nm to 320 nm) and a thermostatted column compartment set to 35 °C. The mass spectra were recorded with a Waters SQD detector with the desolvation temperature set to 450 °C, the source temperature set to 150 °C
and the desolvation gas flow set to 1000 L/hr. The mass spectrometer was equipped with an electrospray ion source (ESI) operated in a positive, or negative, ion mode and was set to scan between m/z 100-2000 with a scan time of 0.2 s. Products were analyzed by UPLC/MS on one of 3 methods (10 mM ammonium formate pH 4/10 mM ammonium bicarbonate pH 10/0.1% TFA). All 3 methods are 10.00 minute methods using an Acquity UPLC BEH C18 (1.7 µM, 2.1 x 50 mm) column and a flow rate of 0.700 mL/min. The gradients are 5%- 100% ACN from 0.00-9.00 minutes and then kept stable at 100% ACN from 9.00-10.00 minutes. [000135] The 1H NMR spectra were recorded on a Bruker Avance II 300 MHz instrument and a Bruker Avance DRX-500 MHz instrument. The chemical shifts are reported in parts- per-million and are referenced to solvent peaks, which in 1H NMR appear at 7.26 ppm for CDCl3, 2.50 for DMSO-d6, and 3.31 ppm for CD3OD. List of abbreviations ACN Acetonitrile ADP Adenosine diphosphate ATP Adenosine triphosphate BSA Bovine serum albumin CDCl3 Deuterated chloroform CD3OD Deuterated methanol CO2 Carbon dioxide DAD Diode-array detection DCM Dichloromethane DIPEA Diisopropylethylamine DMEM Dulbecco’s modified eagle medium DMF N,N’-dimethylformamide DMSO Dimethyl sulfoxide DMSO-d6 Deuterated dimethyl sulfoxide EC50 Half maximal effective concentration ES Electrospray ionization EtOAc Ethyl acetate EtOH Ethanol FBS Fetal bovine serum g Gram(s)
h Hour(s) H2O Water HATU Hexafluorophosphate azabenzotriazole tetramethyl uronium HCl Hydrogen chloride His-tag Polyhistidine tag Hz Hertz HPLC High-performance liquid chromatography IC50 Half-maximal inhibitory concentration L Liter(s) LCMS Liquid chromatography-mass spectrometry LDA Lithium diisopropylamide M Molar m/z Mass-to-charge ratio MeI Methyl iodide MeOH Methanol MgSO4 Magnesium sulfate MHz Megahertz min Minute(s) mL Milliliter(s) mM Millimolar mmol Millimole(s) N2 Nitrogen NaH Sodium hydride NaHCO3 Sodium bicarbonate NaOH Sodium hydroxide Na2SO4 Sodium sulfate NH4Cl Ammonium chloride NH4OH Ammonium hydroxide Ni-NTA Nickel-nitriloacetic acid nm Nanometer(s) nM Nanomolar NMP N-methyl-2-pyrrolidone NMR Nuclear magnetic resonance N-term N-terminus PdCl Palladium chloride
Pd(OAc)2 Palladium acetate TPP or PPh3 Triphenylphosphine QA/QC Quality assurance/Quality control rt Room temperature s Second(s) SEM 2-(Trimethylsilyl)ethoxymethyl t-BuONa Sodium tert-butoxide TC Tissue culture TFA Trifluoroacetic acid THF Tetrahydrofuran tR Retention time Tris Tris(hydroxymethyl)aminomethane UPLC Ultra-performance liquid chromatography UV Ultraviolet v/v Volume per volume μL Microliter(s) Example 1: Preparation of 1-[4-(benzylamino)-1-[2-(morpholin-4-yl)-2-oxoethyl]-1H- pyrrolo[2,3-b]pyridin-6-yl]-2-methyl-1H-indole-4-carboxamide (Compound 29) Step 1: 2-[(4,6-dichloropyrrolo[2,3-b]pyridin-1-yl)methoxy]ethyl-trimethyl-silane [000136] A solution of 4,6-dichloro-1H-pyrrolo[2,3-b]pyridine (10.0 g, 53.5 mmol) in anhydrous DMF (80.0 mL) was added to a suspension of NaH (60% oil dispersion, 3.42 g,
85.5 mmol) in anhydrous DMF (80.0 mL) at 0 °C. The mixture was stirred at 0 °C for 15 min, and 2-(chloromethoxy)ethyl-trimethyl-silane (10.7 g, 64.2 mmol) was added at 0 °C. The mixture was warmed to 23 °C and stirred for 2 h. The mixture was cooled to 0 °C and diluted with a saturated aqueous solution of NH4Cl (500 mL) and EtOAc (800 mL). The phases were separated, and the organic phase was washed with water (2 x 500 mL) and brine (2 x 250 mL), dried (Na2SO4), filtered, and concentrated. The residue was purified by silica gel chromatography (220 g cartridge) with hexanes and EtOAc (0-20%) to provide the title compound as an oil (14.92 g, 88% yield).1H NMR (300 MHz, CDCl3) δ 7.36 (d, J = 3.6 Hz, 1H), 7.16 (d, J = 0.7 Hz, 1H), 6.67 – 6.56 (m, 1H), 5.62 (s, 2H), 3.61 – 3.42 (m, 2H), 1.00 – 0.81 (m, 2H), -0.05 (d, J = 0.8 Hz, 9H). Step 2: N-benzyl-6-chloro-1-(2-trimethylsilylethoxymethyl)pyrrolo[2,3-b]pyridin-4- amine [000137] N-Ethyl-N-isopropyl-propan-2-amine (18.2 g, 141 mmol) was added to a mixture of 2-[(4,6-dichloropyrrolo[2,3-b]pyridin-1-yl)methoxy]ethyl-trimethyl-silane (14.9 g, 47.0 mmol) and benzylamine (10.1 g, 94.0 mmol) in NMP (30.0 mL). The mixture was stirred at 165 °C for 18 hr. The mixture was cooled to 23 °C and diluted with an aqueous solution of HCl (1 M, 100 mL). The aqueous phase was extracted with EtOAc (350 mL). The combined organic phases were washed with saturated aqueous NaHCO3 (150 mL) and brine (150 mL), dried (Na2SO4), filtered, and concentrated. The residue was purified by silica gel chromatography (220 g cartridge) with hexanes and EtOAc (0-25%) to provide the title compound as a solid (11.3 g, 62%).1H NMR (300 MHz, CDCl3) δ 7.49 – 7.30 (m, 5H), 7.10 (dd, J = 3.7, 0.9 Hz, 1H), 6.35 (dd, J = 3.7, 0.9 Hz, 1H), 6.29 (s, 1H), 5.57 (d, J = 0.9 Hz, 2H), 4.79 (d, J = 5.7 Hz, 1H), 4.50 (d, J = 5.5 Hz, 2H), 3.54 (td, J = 8.1, 0.9 Hz, 2H), 0.99 – 0.80 (m, 2H), -0.05 (d, J = 1.0 Hz, 9H). m/z: ES+ [M+H]+ = 388.4; LCMS (A05); tR = 3.03 min.
Step 3: 1-[4-(benzylamino)-1-methyl-pyrrolo[2,3-b]pyridin-6-yl]-2-methyl-indole-4- carbonitrile [000138] Degassed anhydrous toluene (200 mL) was added to a mixture of N-benzyl-6- chloro-1-(2-trimethylsilylethoxymethyl)pyrrolo[2,3-b]pyridin-4-amine (8.00 g, 20.6 mmol), 2-methyl-1H-indole-4-carbonitrile (75.0 %, 5.58 g, 26.8 mmol), t-BuONa (3,963 mg, 41.2 mmol), BippyPhos (3,134 mg, 6.19 mmol), and [(Cinnamyl)PdCl]2 (1602 mg, 3.09 mmol). The mixture was stirred at 110 °C for 3 h, cooled to 23 °C, and filtered through Celite washing with EtOAc (100 mL). The filtrate was concentrated. The residue was purified by silica gel chromatography (120 g cartridge) using hexanes and EtOAc (0-20%) to provide the title compound as an oil (12.0 g, 53%, 46% pure).1H NMR (300 MHz, CDCl3) δ 7.43 (d, J = 4.1 Hz, 2H), 7.24 (d, J = 7.8 Hz, 9H), 6.54 (d, J = 3.7 Hz, 1H), 5.62 (s, 2H), 5.04 (s, 2H), 3.59 – 3.48 (m, 2H), 2.40 (s, 3H), 1.43 (s, 9H), 0.88 (t, J = 8.2 Hz, 2H). m/z: ES+ [M+H]+ = 508.3; LCMS (A05); tR = 2.85 min. Step 4: 1-[4-(benzylamino)-1-(2-trimethylsilylethoxymethyl)pyrrolo[2,3-b]pyridin-6-yl]- 2-methyl-indole-4-carboxamide [000139] (1E)-Acetaldehyde oxime (3.21 g, 54.4 mmol) was added to a mixture of 1-[4- (benzylamino)-1-(2-trimethylsilylethoxymethyl)pyrrolo[2,3-b]pyridin-6-yl]-2-methyl-indole- 4-carbonitrile (46.0 %, 12.0 g, 10.9 mmol), palladium acetate (0.488 g, 2.17 mmol), and triphenylphosphine (1.14 g, 4.35 mmol) in a mixture of EtOH/H2O (8:1 v/v, 180 mL) at 23
°C. The flask was sealed, and the mixture was stirred at 110 °C for 2 h. The mixture was cooled to 23 °C and filtered through Celite washing with EtOAc (2 x 20 mL). The filtrate was concentrated, and the residue was purified by silica gel chromatography (220 g cartridge) with hexanes and ethyl acetate (0-100%) to provide the title compound as a solid (5.09 g, 89%). m/z: ES+ [M+H]+ = 526.2; LCMS (A05); tR = 2.53 min. Step 5: 1-[4-(benzylamino)-1H-pyrrolo[2,3-b]pyridin-6-yl]-2-methyl-indole-4- carboxamide [000140] TFA (50.0 mL) was added to a solution of 1-[4-(benzylamino)-1-(2- trimethylsilylethoxymethyl)pyrrolo[2,3-b]pyridin-6-yl]-2-methyl-indole-4-carboxamide (5.09 g, 9.68 mmol) in anhydrous DCM (150 mL) at 0 °C. The mixture was stirred at 23 °C for 3 h and concentrated. The residue was dissolved in 1,4-dioxane (150 mL), and a saturated aqueous solution of NH4OH (50 mL) was added. The mixture was stirred at 60 °C for 16 h and concentrated. The residue was purified by silica gel chromatography (80 g cartridge) with ethyl acetate in hexanes (0-100%) to provide the title compound as a solid (3.09 g, 81%).1H NMR (300 MHz, DMSO-d6) δ 11.39 (s, 1H), 7.68 (t, J = 6.0 Hz, 2H), 7.45 – 7.23 (m, 6H), 7.19 (t, J = 2.9 Hz, 2H), 6.99 – 6.83 (m, 2H), 6.80 (d, J = 1.3 Hz, 1H), 6.72 (dd, J = 3.5, 2.1 Hz, 1H), 6.01 (s, 1H), 4.52 (d, J = 6.0 Hz, 2H), 2.25 (s, 3H). m/z: ES+ [M+H]+ = 396.3; LCMS (A05); tR = 2.36 min.
Step 6: 2-[4-(benzylamino)-6-(4-carbamoyl-2-methyl-indol-1-yl)pyrrolo[2,3-b]pyridin-1- yl]acetic acid [000141] Sodium hydride (60.0%, 26 mg, 0.68 mmol) was added to a solution of 1-[4- (benzylamino)-1H-pyrrolo[2,3-b]pyridin-6-yl]-2-methyl-indole-4-carboxamide (245 mg, 0.620 mmol) in dry DMF (5.00 mL) under N2 at 23 °C. After stirring for 30 min, a solution of tert-butyl 2-bromoacetate (133 mg, 0.681 mmol) in dry DMF (2.00 mL) was added. The mixture was stirred at 23 °C for 1 h. Water (1.00 mL) was added to the mixture, and the solvent was concentrated. The residue was dissolved in DCM (3.00 mL) and TFA (3.00 mL) was added. After stirring the mixture for 15 h, the mixture was concentrated. The residue (500 mg) was taken to the next step without purification. m/z: ES+ [M+H]+ = 454.1; LCMS (A05); tR = 2.05 min. Step 7: 1-[4-(benzylamino)-1-[2-(morpholin-4-yl)-2-oxoethyl]-1H-pyrrolo[2,3-b]pyridin- 6-yl]-2-methyl-1H-indole-4-carboxamide [000142] HATU (419.0 mg, 1.10 mmol) was added to a mixture of DIPEA (0.377 mL, 2.21 mmol), 2-[4-(benzylamino)-6-(4-carbamoyl-2-methyl-indol-1-yl)pyrrolo[2,3-b]pyridin-1- yl]acetic acid (250 mg, 0.551 mmol) and morpholine (0.121 mL, 1.38 mmol) in dry DMF (5.00 mL) under N2 at 23 °C. The mixture was stirred at 23 °C for 2 h. The mixture was concentrated, and the residue was purified using reverse phase HPLC (40 g, C18 column)
with 10-80% ACN in Water (10 mM ammonium formate) as eluent to obtain the title product (230 mg, 80%) as a solid.1H NMR (500 MHz, DMSO-d6) δ 7.75 (s, 1H), 7.68 (s, 1H), 7.42 (d, J = 7.3 Hz, 1H), 7.36 (dt, J = 14.9, 7.3 Hz, 4H), 7.28 (t, J = 7.0 Hz, 1H), 7.18 (d, J = 3.5 Hz, 2H), 6.96 (d, J = 8.1 Hz, 1H), 6.86 (t, J = 7.8 Hz, 1H), 6.81 (s, 1H), 6.73 (d, J = 3.4 Hz, 1H), 6.03 (s, 1H), 5.05 (s, 2H), 4.54 (d, J = 5.9 Hz, 2H), 3.54 (s, 6H), 3.41 (s, 2H), 2.24 (s, 3H). m/z: ES+ [M+H]+ 523.8; LCMS (A05); tR = 2.16 min. Example 2: Preparation of 1-[4-(benzylamino)-1-[2-(morpholin-4-yl)-2-oxoethyl]-1H- pyrrolo[2,3-b]pyridin-6-yl]-2-methyl-1H-indole-4-carboxamide (Compound 15) Step 1: 1-(Benzenesulfonyl)-4,6-dichloro-1H-pyrrolo[2,3-b]pyridine [000143] 4,6-Dichloro-1h-pyrrolo[2,3-b]pyridine (20.0 g, 107 mmol) was dissolved in anhydrous DMF (320 mL) and NaH (60% oil dispersion, 6.42 g, 160 mmol) was added at 0 C. The mixture was stirred at 0 °C for 15 min, and benzenesulfonyl chloride (16.4 mL, 128 mmol) was added. The mixture was slowly warmed and stirred at 23 °C for 16 hours. The mixture was cooled to 0 °C, and diluted with a saturated aqueous solution of NH4Cl (100 mL), and the aqueous phase was extracted with DCM (2 x 100 mL). The combined organic phases were washed with water and brine, dried (MgSO4), filtered, and concentrated. The residue was triturated with ACN to provide the title compound as a solid (30.5 g, 87% yield). m/z: ES+ [M+H]+ = 327; LCMS (A05); tR = 2.84 min.
Step 2: 1-(Benzenesulfonyl)-4,6-dichloro-2-methyl-1H-pyrrolo[2,3-b]pyridine [000144] 1-(Benzenesulfonyl)-4,6-dichloro-1H-pyrrolo[2,3-b]pyridine (5.00 g, 15.3 mmol) was dissolved in dry THF (75 mL) and added dropwise to a freshly prepared solution of LDA (prepared by the addition of n-BuLi (2.5 M in hexane, 12.2 mL, 30.6 mmol) to a solution of diisopropylamine (4.30 mL, 30.6 mmol) in dry THF (45 mL) followed by stirring for 30 min at -78°C). The mixture was warmed to 0 °C, and MeI (1.90 mL, 30.6 mmol) was added dropwise. The mixture was stirred for 3 h at 0 °C and 23 °C for 16 hours. The mixture was diluted with a saturated aqueous solution of NH4Cl (100 mL) and extracted with EtOAc (2 x 100 mL). The combined organic phases were washed with water and brine, dried (MgSO4), filtered, and concentrated. The residue was purified by silica gel chromatography using 0- 20% EtOAc in cyclohexane to provide the title compound as a solid (1.18 g 23% yield). m/z: ES+ [M+H]+ = 341; LCMS (A05); tR = 1.23 min. Step 3: 1-(Benzenesulfonyl)-N-benzyl-6-chloro-2-methyl-1H-pyrrolo[2,3-b]pyridin-4- amine [000145] 1-(Benzenesulfonyl)-4,6-dichloro-2-methyl-1H-pyrrolo[2,3-b]pyridine (1.0g, 2.93 mmol) was dissolved in NMP (9 mL), and DIPEA (1.53 mL, 8.79 mmol) and benzylamine (6, 961 μL, 8.79 mmol) were added. The mixture was stirred at 200 °C for 16 hours. The mixture was cooled to 23 °C and diluted with water. The aqueous phase was extracted with cyclohexane (100 mL). The combined organic phase was dried (MgSO4), filtered, and concentrated. The residue was purified by silica gel chromatography using 0-20% EtOAc in
cyclohexane to provide the title compound as a solid (570 mg, 47% yield). m/z: ES+ [M+H]+ = 412; LCMS (A05); tR = 1.15 min. Step 4: 1-[1-(Benzenesulfonyl)-4-(benzylamino)-2-methyl-1H-pyrrolo[2,3-b]pyridin-6- yl]-2-methyl-1H-indole-4-carbonitrile [000146] 1-(Benzenesulfonyl)-N-benzyl-6-chloro-2-methyl-1H-pyrrolo[2,3-b]pyridin-4- amine (570 mg, 1.38 mmol) was dissolved in dry toluene (7 mL), 2-methyl-1H-indole-4- carbonitrile (216 mg, 1.38 mmol), t-BuONa (206 mg, 2.14 mmol), [PdCl(cinnamyl)]2 (143 mg, 0.28 mmol), and BippyPhos (295 mg, 0.58 mmol) were added at 23 °C under nitrogen. The mixture was stirred at 110 °C for 2 h. The mixture was cooled to 23 °C and filtered through a short pad of Celite, washing with EtOAc. The filtrate was concentrated, and the residue was purified by silica gel chromatography using 0-25% EtOAc in cyclohexane to provide the title compound as a solid (191 mg, 26% yield). m/z: ES+ [M+H]+ = 532; LCMS (A05); tR = 1.23 min. Step 5: 1-[1-(Benzenesulfonyl)-4-(benzylamino)-2-methyl-1H-pyrrolo[2,3-b]pyridin-6- yl]-2-methyl-1H-indole-4-carboxamide [000147] Acetaldoxime (108 ^L, 1.80 mmol), 1-[1-(Benzenesulfonyl)-4-(benzylamino)-2- methyl-1H-pyrrolo[2,3-b]pyridin-6-yl]-2-methyl-1H-indole-4-carbonitrile (191 mg, 0.36 mmol), Pd(OAc)2 (16.9 mg, 0.08 mmol), and TPP (22.6 mg, 0.09 mmol) were dissolved in a
mixture of EtOH/H2O (8:1 v/v, 8 mL) at 23 °C under nitrogen, and the flask was sealed. The mixture was stirred at 110 °C for 3 hours. The mixture was cooled to room temperature, filtered through a short pad of Celite, washing with EtOAc. The filtrate was concentrated, and the residue was purified by silica gel chromatography using 0-5% MeOH in DCM to provide the title compound as a solid (120 mg, 61%). m/z: ES+ [M+H]+ = 550; LCMS (A05); tR = 0.99 min. Step 6: 1-[4-(Benzylamino)-2-methyl-1H-pyrrolo[2,3-b]pyridin-6-yl]-2-methyl-1H- indole-4-carboxamide [000148] 1-[1-(Benzenesulfonyl)-4-(benzylamino)-2-methyl-1H-pyrrolo[2,3-b]pyridin-6- yl]-2-methyl-1H-indole-4-carboxamide (300 mg (70% purity), 0.38 mmol) was dissolved in 1,4-dioxane (3 mL), and aqueous solution of NaOH (2 M, 955 μL, 1.91 mmol) was added. The mixture was stirred at 80 °C for 16 hours. The mixture was concentrated and the residue was purified by reverse phase HPLC (40 g, C18 column) with 10-80% ACN in Water (10 mM ammonium formate) to provide the title compound as a solid (87 mg, 55% yield). 1H NMR (500 MHz, DMSO-d6) δ 11.21 (s, 1H), 7.68 (s, 1H), 7.47 (t, J = 6.1 Hz, 1H), 7.40 (d, J = 7.2 Hz, 1H), 7.38 – 7.30 (m, 4H), 7.27 (t, J = 6.4 Hz, 1H), 7.18 (s, 1H), 6.92 (d, J = 8.1 Hz, 1H), 6.86 (t, J = 7.7 Hz, 1H), 6.78 (s, 1H), 6.37 (s, 1H), 5.96 (s, 1H), 4.49 (d, J = 6.0 Hz, 2H), 2.35 (s, 3H), 2.23 (s, 3H). m/z: ES+ [M+H]+ = 410; LCMS (A05); tR = 4.67 min.
Step 7: 1-[4-(Benzylamino)-2-methyl-1-[2-(morpholin-4-yl)-2-oxoethyl]-1H-pyrrolo[2,3- b]pyridin-6-yl]-2-methyl-1H-indole-4-carboxamide [000149] 1-[4-(Benzylamino)-2-methyl-1H-pyrrolo[2,3-b]pyridin-6-yl]-2-methyl-1H- indole-4-carboxamide (40 mg, 0.10 mmol) was dissolved in dry DMF (0.8 mL), and NaH (60% oil dispersion, 4.30 mg, 0.11 mmol) was added. The mixture was stirred at rt for 30 min. A solution of N-(chloroacetyl)morpholine (13.6 μL, 0.11 mmol) in dry DMF (0.4 mL) was added, and the mixture was stirred at 23 °C for 1 h. The mixture was concentrated and the residue was purified by reverse phase HPLC (40 g, C18 column) with 10-80% ACN in Water (10 mM ammonium formate) to provide the title compound as a solid (11 mg, 21% yield).1H NMR (500 MHz, DMSO-d6) δ 7.68 (s, 1H), 7.59 (t, J = 6.1 Hz, 1H), 7.41 (d, J = 7.2 Hz, 1H), 7.35 (q, J = 7.5 Hz, 4H), 7.28 (t, J = 6.8 Hz, 1H), 7.18 (s, 1H), 6.91 (d, J = 8.1 Hz, 1H), 6.85 (t, J = 7.7 Hz, 1H), 6.79 (s, 1H), 6.47 (s, 1H), 6.00 (s, 1H), 5.01 (d, J = 9.0 Hz, 2H), 4.52 (d, J = 5.2 Hz, 2H), 3.56 (s, 4H), 3.50 (s, 2H), 3.41 (s, 2H), 2.31 (s, 3H), 2.23 (s, 3H). m/z: ES+ [M+H]+ = 537; LCMS (A05); tR = 4.39 min. Example 3: Biological Assays [000150] Protein expression and purification: Full length p97/VCP with N-term His-tag was expressed in BL21 E. coli expression system and purified using Ni-NTA chromatography followed by dialysis overnight in buffer containing Apyrase. The dialyzed protein was then concentrated and with size exclusion chromatography. Fractions consisting of hexameric p97 were pooled and concentrated for biochemical studies. [000151] Efficacy of exemplary compounds in inhibition of p97 ATPase activity: The effect of compound inhibition on p97 enzymatic activity was evaluated using a p97 ATPase assay, such as the ADP-GloTM assay (Promega). The p97 ATPase assay consisted of 10 µL reaction volumes in 384-well white, opaque plates. Compounds were prepared from DMSO stocks in assay buffer (40 mM Tris pH 7.5, 20 mM MgCl2, 0.1 mg/mL BSA) and
preincubated with 110 nM p97 for 30 min at room temperature. 50 µM ATP was then added to initiate an ATPase reaction with 0.1% DMSO in final volume. After 1 h incubation at room temperature, conversion of ATP to ADP was detected using ADP-GloTM reagents according to the manufacturer’s protocol. Luminescence was measured using a microplate reader, such as a Cytation 5 microplate reader. % activity was calculated by normalizing to DMSO control (max) and no ATP control (min). % activity as a function of compound concentration from triplicate wells was fit with a 4-parameter logistic curve equation using GraphPad Prism or CDD Vault to derive IC50 values. [000152] Cell culture: HCT116 and RPMI-8226 cells were acquired from American Type Culture Collection (ATCC). Mia-Paca2 stable cells with CHOP Luciferase reporter were acquired from Signosis. HCT116 cells were cultured in McCoy's 5a Medium Modified supplemented with 10% FBS, RPMI-8226 cells were cultured in RPMI-1640 supplemented with 10% FBS, and Mia-Paca2 CHOP cells were cultured in DMEM supplemented with 10% FBS, 2.5% horse serum and Hygromycin B. All cells were incubated at 37 ^C, 5% CO2. [000153] Efficacy of exemplary compounds in cellular increase of CHOP expression: The effect on p97 cellular function and unfolded protein response pathway was measured by CHOP expression as a readout. Mia-Paca2 cells stably expressing a CHOP Luciferase reporter were plated with 6,500 cells per well in 384-well white, TC-treated plates. Cells were incubated for 24 h before treatment with test compounds (0.1% DMSO in final volume) or vehicle (DMSO) for 8 h. The plate was then equilibrated to room temperature before addition of 1:1 volume of Bright-Glo® Luciferase reagent (Promega). The contents were mixed for 2 min on an orbital shaker before measuring luminescence on a microplate reader, such as a Cytation 5 microplate reader. % luminescence normalized to DMSO control as a function of compound concentration from triplicate wells was fit with a 4-parameter logistic curve using GraphPad Prism or CDD Vault to derive EC50 values. [000154] Efficacy of exemplary compounds in inhibition of cell viability: RPMI-8226 (p97i sensitive) and HCT116 (p97i insensitive) cells were plated at 6,500 cells per well in 384-well white, TC-treated plates. After incubating for 24 h the cells were treated with vehicle (DMSO) or test compounds (0.1% DMSO in final volume) for 24 hrs. The plate was then equilibrated to room temperature before addition of a 1:1 volume of CellTiter-Glo® reagent (Promega). The contents were mixed for 10 min on an orbital shaker to induce lysis before measuring luminescence on a microplate reader, such as a Cytation 5 microplate reader. % viability was calculated by normalizing to DMSO control (max) and no
cells/background control (min). % viability as a function of compound concentration from triplicate wells was fit with a 4-parameter logistic curve using GraphPad Prism or CDD Vault to derive IC50 values. [000155] Results from these assays are summarized in Table 2 below. In this table, for the p97 ATPase assay, “A” indicates IC50 of less than 10 nM; “B” indicates IC50 of between 10 nM and 100 nM; and “C” indicates IC50 of greater than 100 nM. For the CHOP expression Bright-Glo® Luciferase assay, “A” indicates EC50 of less than 1 μM; “B” indicates EC50 of between 1 μM to 10 μM; and “C” indicates EC50 of greater than 10 μM. For the RPMI-8226 CellTiter-Glo® cell viability assay, “A” indicates IC50 of less than 1 μM; “B” indicates IC50 of between 1 μM to 10 μM; and “C” indicates IC50 of greater than 10 μM. For the HCT116 CellTiter-Glo® cell viability assay, “A” indicates IC50 of greater than 10 μM. Table 2. Assay Results CHOP activity p97 A Bright-Glo Cell viability Cell viability C N TPase assay RPMI 8226 HCT116
CHOP acti it
PDE6 off-target activity of exemplary compounds was assessed using a cGMP phosphodiesterase assay, such as the cGMP phosphodiesterase assay at Eurofins Panlabs. Assay procedures were performed according to methods outlined by the manufacturer. Briefly, the assay consisted of 0.2 ^g/mL bovine retinal rod PDE6 activated with trypsin and preincubated with a given compound at 10 ^M or 1 ^M (1% DMSO in final volume) for 15 mins at 25 ^C. Then, 100 μM cGMP and 0.03 μM [3H]cGMP was added to initiate the reaction, incubated for 20 mins at 25 ^C, and then terminated at 100 ^C. Following the conversion of [3H]GMP to [3H]guanosine by nucleosidase, the amount of [3H]guanosine was quantified using scintillation counting by any known method in the art. [000157] Results from the assay are summarized in Table 3 below. In this table, “A” indicates PDE6 inhibition of less than 10%; “B” indicates PDE6 inhibition of between 10%
and 40%; “C” indicates PDE6 inhibition of between 40% to 70%; and “D” indicates PDE6 inhibition of greater than 70%. For comparative purposes, Table 3 also includes the PDE6 inhibition activity of a prior art compound referred to as CB-5083.
INCORPORATION BY REFERENCE
[000158] The entire disclosure of each of the patent and scientific documents referred to herein is incorporated by reference for all purposes. This application refers to various issued patents, published patent applications, journal articles, and other publications, all of which are incorporated herein by reference.
[000159] If there is a conflict between any of the incorporated references and the instant specification, the specification shall control. In addition, any particular embodiment of the present invention that falls within the prior art may be explicitly excluded from any one or more of the claims. Because such embodiments are deemed to be known to one of ordinary skill in the art, they may be excluded even if the exclusion is not set forth explicitly herein. Any particular embodiment of the invention can be excluded from any claim, for any reason, whether or not related to the existence of prior art.
EQUIVALENTS
[000160] The invention may be embodied in other specific forms without departing from the spirit or essential characteristics thereof. The foregoing embodiments are therefore to be considered in all respects illustrative rather than limiting on the invention described herein. Scope of the invention is thus indicated by the appended claims rather than by the foregoing description, and all changes that come within the meaning and range of equivalency of the claims are intended to be embraced therein.
Claims
CLAIMS What is claimed is: 1. A compound of Formula (I): (I), or a pharmaceutically acceptable salt thereof, wherein: each R1 is independently selected from the group consisting of halogen, C1-6 alkyl, C1- 6 alkoxy, C1-6 haloalkyl, and C1-6 alkylene-OH; R2 is selected from hydrogen and C1-6 alkyl; R3 is selected from the group consisting of hydrogen, 3-10 membered heterocyclyl, - C(O)-(3-10 membered heterocyclyl), and C1-6 alkyl optionally substituted with a substituent selected from the group consisting of C1-6 alkoxy, -CN, -C(O)N(Ra)2, -NRbS(O)2C1-6 alkyl, - S(O)2N(Rb)2, 5-6 membered heteroaryl, 3-10 membered heterocyclyl, and -C(O)-(3-10 membered heterocyclyl), wherein the 3-10 membered heterocyclyl is optionally substituted with 1, 2, or 3 substituents each independently selected from the group consisting of C1-6 alkyl, oxo, -C(O)-C1-6 alkyl, -S(O)2C1-6 alkyl, and -N(Rb)2, R4 is selected from hydrogen and C1-6 alkyl; R5 is selected from hydrogen and C1-6 alkyl; R6 is selected from hydrogen and C1-6 alkyl; each R7 is independently selected from the group consisting of hydrogen, C1-6 alkyl, - (C1-6 alkylene)-OH, -(C1-6 alkylene)-C1-6 alkoxy, -(C1-6 alkylene)-N(Rb)2, C1-6 alkoxy, -CN, - C(O)N(Ra)2, and halogen;
each Ra is independently selected from the group consisting of hydrogen, C1-6 alkyl, and C1-6 alkoxy; each Rb is independently selected from hydrogen or C1-6 alkyl; n is 1 or 2; and m is selected from 0, 1, and 2. 2. A compound of Formula (II): (II), or a pharmaceutically acceptable salt thereof, wherein: each R1 is independently selected from the group consisting of halogen, C1-6 alkyl, C1- 6 alkoxy, C1-6 haloalkyl, and C1-6 alkylene-OH; R2 is selected from hydrogen and C1-6 alkyl; R3 is selected from the group consisting of hydrogen, 3-10 membered heterocyclyl, - C(O)-(3-10 membered heterocyclyl), -L1-R8, and C1-6 alkyl optionally substituted with a substituent selected from the group consisting of C1-6 alkoxy, -CN, -C(O)N(Ra)2, - NRbS(O)2C1-6 alkyl, -S(O)2N(Rb)2, 5-6 membered heteroaryl, 3-10 membered heterocyclyl, and -C(O)-(3-10 membered heterocyclyl), wherein the 3-10 membered heterocyclyl is optionally substituted with 1,
2, or 3 substituents each independently selected from the group consisting of C1-6 alkyl, oxo, -C(O)-C1-6 alkyl, -S(O)2C1-6 alkyl, and -N(Rb)2, R4 is selected from hydrogen and C1-6 alkyl; R5 is selected from hydrogen and C1-6 alkyl; R6 is selected from hydrogen and C1-6 alkyl;
each R7 is independently selected from the group consisting of hydrogen, C1-6 alkyl, - (C1-6 alkylene)-OH, -(C1-6 alkylene)-C1-6 alkoxy, -(C1-6 alkylene)-N(Rb)2, C1-6 alkoxy, -CN, - C(O)N(Ra)2, and halogen; L1 is a linker moiety; R8 is a moiety derived from an immunomodulatory imide drug; each Ra is independently selected from the group consisting of hydrogen, C1-6 alkyl, and C1-6 alkoxy; each Rb is independently selected from hydrogen or C1-6 alkyl; n is 1 or 2; and m is selected from 0, 1, and 2.
3. The compound of claim 1 or 2, wherein R2 is hydrogen.
4. The compound of any one of claims 1-3, wherein R3 is selected from the group consisting of hydrogen, methyl, , , , , , , , O N , , , , , , , , , , , , , , , and .
5. The compound of any one of claims 1-4, wherein R3 is .
6. The compound of any one of claims 1-5, wherein R3 is hydrogen.
7. The compound of any one of claims 1-6, wherein R4 is hydrogen or methyl.
8. The compound of any one of claims 1-7, wherein R4 is methyl.
9. The compound of any one of claims 1-8, wherein R4 is hydrogen.
10. The compound of any one of claims 1-9, wherein R5 is hydrogen.
11. The compound of any one of claims 1-10, wherein R6 is selected from hydrogen and methyl.
12. The compound of any one of claims 1-11, wherein R6 is methyl.
13. The compound of any one of claims 1-12, wherein R6 is at the 2-position of the indolyl.
14. The compound of any one of claims 1-13, wherein each R7 is independently selected from the group consisting of hydrogen, C1-6 alkyl, -(C1-6 alkylene)-OH, -(C1-6 alkylene)- N(Rb)2, -CN, and -C(O)NH2.
15. The compound of any one of claims 1-14, wherein R7 is -C(O)NH2.
16. The compound of any one of claims 1-15, wherein R7 is at the 4-position of the indolyl.
17. The compound of any one of claims 2-16, wherein L1 is a linker moiety selected from the group consisting of: -(C1-10 alkylene)-C(O)-, -(2-10 membered heteroalkylene)-C(O)-, - (2-10 membered heteroalkylene)-C(O)-(C1-10 alkylene), and -(C1-10 alkylene)-(3-7 membered heterocyclylene)-(C1-10 alkylene).
18. The compound of any one of claims 2-17, wherein the immunomodulatory imide drug is a thalidomide analog.
19. The compound of any one of claims 2-18, wherein the immunomodulatory imide drug is selected from pomalidomide and lenalidomide.
20. The compound of claim 19, wherein R8 is selected from: and .
21. The compound of any one of claims 1-20, wherein n is 1.
22. The compound of any one of claims 1-21, wherein m is 0.
23. The compound of claim 1 or 2, wherein the compound is selected from Table 1 or a pharmaceutically acceptable salt thereof.
24. A pharmaceutical composition comprising a compound of any one of claims 1-23 or a pharmaceutically acceptable salt thereof and a pharmaceutically acceptable excipient.
25. A method of treating a disease or condition associated with p97 protein activity in a subject in need thereof, the method comprising administering to the subject a therapeutically effective amount of a compound of any one of claims 1 -23 or a pharmaceutically acceptable salt thereof or a pharmaceutical composition of claim 24.
26. The method of claim 25, wherein the disease or condition associated with p97 protein activity7 is cancer.
27. The method of claim 26, wherein the cancer is multiple myeloma or acute myeloid leukemia.
28. The method of claim 25, wherein the disease or condition associated with p97 protein activity is a neurodegenerative disorder.
29. The method of claim 28, wherein the neurodegenerative disorder is ALS, Charcot-Marie- Tooth disease type 2Y, or Inclusion body myopathy with early -onset Paget disease and frontotemporal dementia (TBMPFD).
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363512750P | 2023-07-10 | 2023-07-10 | |
| US63/512,750 | 2023-07-10 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025014963A1 true WO2025014963A1 (en) | 2025-01-16 |
Family
ID=91966542
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/037229 Pending WO2025014963A1 (en) | 2023-07-10 | 2024-07-09 | Inhibitors of p97 protein and methods of use |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025014963A1 (en) |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3845770A (en) | 1972-06-05 | 1974-11-05 | Alza Corp | Osmatic dispensing device for releasing beneficial agent |
| US4326525A (en) | 1980-10-14 | 1982-04-27 | Alza Corporation | Osmotic device that improves delivery properties of agent in situ |
| US4902514A (en) | 1988-07-21 | 1990-02-20 | Alza Corporation | Dosage form for administering nilvadipine for treating cardiovascular symptoms |
| US4992445A (en) | 1987-06-12 | 1991-02-12 | American Cyanamid Co. | Transdermal delivery of pharmaceuticals |
| US5001139A (en) | 1987-06-12 | 1991-03-19 | American Cyanamid Company | Enchancers for the transdermal flux of nivadipine |
| US5023252A (en) | 1985-12-04 | 1991-06-11 | Conrex Pharmaceutical Corporation | Transdermal and trans-membrane delivery of drugs |
| US5616345A (en) | 1983-12-22 | 1997-04-01 | Elan Corporation Plc | Controlled absorption diltiazen formulation for once-daily administration |
| WO2014015291A1 (en) * | 2012-07-20 | 2014-01-23 | Han-Jie Zhou | FUSED PYRIMIDINES AS INHIBITORS OF p97 COMPLEX |
| WO2016200840A1 (en) * | 2015-06-08 | 2016-12-15 | Cleave Biosciences, Inc. | Mono and bicyclic ring boronic acid, ester and salt compounds as inhibitors of p97 complex |
-
2024
- 2024-07-09 WO PCT/US2024/037229 patent/WO2025014963A1/en active Pending
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3845770A (en) | 1972-06-05 | 1974-11-05 | Alza Corp | Osmatic dispensing device for releasing beneficial agent |
| US4326525A (en) | 1980-10-14 | 1982-04-27 | Alza Corporation | Osmotic device that improves delivery properties of agent in situ |
| US5616345A (en) | 1983-12-22 | 1997-04-01 | Elan Corporation Plc | Controlled absorption diltiazen formulation for once-daily administration |
| US5023252A (en) | 1985-12-04 | 1991-06-11 | Conrex Pharmaceutical Corporation | Transdermal and trans-membrane delivery of drugs |
| US4992445A (en) | 1987-06-12 | 1991-02-12 | American Cyanamid Co. | Transdermal delivery of pharmaceuticals |
| US5001139A (en) | 1987-06-12 | 1991-03-19 | American Cyanamid Company | Enchancers for the transdermal flux of nivadipine |
| US4902514A (en) | 1988-07-21 | 1990-02-20 | Alza Corporation | Dosage form for administering nilvadipine for treating cardiovascular symptoms |
| WO2014015291A1 (en) * | 2012-07-20 | 2014-01-23 | Han-Jie Zhou | FUSED PYRIMIDINES AS INHIBITORS OF p97 COMPLEX |
| WO2016200840A1 (en) * | 2015-06-08 | 2016-12-15 | Cleave Biosciences, Inc. | Mono and bicyclic ring boronic acid, ester and salt compounds as inhibitors of p97 complex |
Non-Patent Citations (21)
| Title |
|---|
| ADEBOYE ADEJARE, REMINGTON: THE SCIENCE AND PRACTICE OF PHARMACY, 2020 |
| BERGE ET AL., J. PHARMACEUTICAL SCIENCES, vol. 66, 1977, pages 2725 - 19 |
| CAO, K. ET AL., CELL, vol. 115, 2003, pages 355 - 367 |
| CARRUTHERS: "Some Modern Methods of Organic Synthesis", 1987, CAMBRIDGE UNIVERSITY PRESS |
| CARVALHO, P. ET AL., CELL, vol. 126, 2006, pages 361 - 373 |
| CHENG, J ET AL., AGING CELL, vol. 17, no. 5, 2018, pages el280 |
| ELIEL: "STEREOCHEMISTRY OF CARBON COMPOUNDS", 1962, MCGRAW-HILL |
| FU, X., J. CELL BIOL., vol. 163, 2003, pages 21 - 26 |
| GOULD: "Salt selection for basic drugs", INTERNATIONAL JOURNAL OF PHARMACEUTICS, vol. 33, 1986, pages 201 - 217, XP025813036, DOI: 10.1016/0378-5173(86)90055-4 |
| JACQUES ET AL.: "ENANTIOMERS, RACEMATES AND RESOLUTIONS", 1981, WILEY INTERSCIENCE |
| JANIESCH, P. C. ET AL., NATURE CELL BIOL., vol. 9, 2007, pages 379 - 390 |
| LAROCK: "Comprehensive Organic Transformations", 1989, VCH PUBLISHERS, INC. |
| MEYER ET AL., NATURE CELL BIOL., vol. 14, 2012, pages 117 - 123 |
| SMITHMARCH: "March's Advanced Organic Chemistry", 2001, JOHN WILEY & SONS, INC. |
| T. W. GREENEP. G. M. WUTS: "Protecting Groups in Organic Synthesis", 1991, WILEY |
| THOMAS SORRELL: "Handbook of Chemistry and Physics", 1999, UNIVERSITY SCIENCE BOOKS |
| WILEN: "TABLES OF RESOLVING AGENTS AND OPTICAL RESOLUTIONS", 1972, UNIV. OF NOTRE DAME PRESS, pages: 268 |
| YAMAMOTO, S. ET AL., ANN. SURG. ONCOL., vol. 11, 2004, pages 165 - 172 |
| YAMAMOTO, S. ET AL., CLIN. CANCER RES., vol. 10, 2004, pages 5558 - 5565 |
| YAMAMOTO, S., ANN. SURG. ONCOL., vol. 12, 2005, pages 925 - 934 |
| YE, Y. ET AL., NATURE, vol. 429, 2004, pages 841 - 847 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP4592295A1 (en) | Pyrimidine-fused ring compound, and preparation method therefor and use thereof | |
| CN116554151B (en) | Kinesin KIF18A inhibitor and application thereof | |
| EP3184521B1 (en) | Indazole compounds as fgfr kinase inhibitors, preparation and use thereof | |
| CN107253963B (en) | Pyridone and azapyridone compounds and methods of use | |
| CN105051043B (en) | Novel heterocyclic derivatives as modulators of kinase activity | |
| EP4129996A1 (en) | Novel aminopyrimidine egfr inhibitor | |
| CN118005631A (en) | Tetrahydro-pyrido[3,4-b]indole estrogen receptor modulators and uses thereof | |
| BRPI0616722A2 (en) | compound or a pharmaceutically acceptable salt or an in vivo hydrolysable ester thereof, process for preparing it, pharmaceutical composition, use of a compound or a pharmaceutically acceptable salt or an in vivo hydrolysable ester thereof, and methods for producing an anti-oxidizing effect. -proliferation and an inhibitory effect cdk2, to treat cancer, and to treat diseases | |
| WO2020103897A1 (en) | Heterocyclic fused pyrimidine derivative, pharmaceutical composition thereof, and application thereof | |
| EP3849984B1 (en) | 1-isopropyl-3-methyl-8- (pyridin-3-yl) -1, 3-dihydro-2h-imidazo [4, 5-c] cinnolin-2-one as selective modulators of ataxia telangiectasia mutated (atm) kinase and uses thereof | |
| KR102667331B1 (en) | Aminopyridine derivatives and their use as selective alk-2 inhibitors | |
| CA2998705A1 (en) | 1-phenylpyrrolidin-2-one derivatives as perk inhibitors | |
| CN120794975A (en) | Novel heterocyclic compounds as CDK8/19 inhibitors | |
| KR20250036135A (en) | KAT6 targeting compounds having a ubiquitin ligase binding moiety | |
| WO2022251588A1 (en) | Heterobifunctional compounds and methods of treating disease | |
| EP3617204A1 (en) | Indoleamine 2,3-dioxygenase inhibitor and application | |
| CA3117319A1 (en) | Fused bicyclic heterocycles as thereapeutic agents | |
| CN113365696A (en) | Pharmaceutical compounds and their use as inhibitors of ubiquitin-specific protease 19(USP19) | |
| CN117402161A (en) | Sulfoxide imine compound with FGFR inhibition effect, pharmaceutical composition containing sulfoxide imine compound and application of sulfoxide imine compound | |
| WO2022048498A1 (en) | Usp7 inhibitor | |
| WO2025014963A1 (en) | Inhibitors of p97 protein and methods of use | |
| CN105102435A (en) | Novel imidazol-piperidinyl derivatives as modulators of kinase activity | |
| EP4308569A1 (en) | Selective modulators of ataxia telangiectasia mutated (atm) kinase and uses thereof | |
| CN109836427B (en) | Pyrimidopyrimidinones and their applications | |
| CN115279757A (en) | Pyrimidine bicyclic compounds as antiviral agents for the treatment and prevention of HIV infection |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24746591 Country of ref document: EP Kind code of ref document: A1 |