WO2024264003A2 - Antibodies that bind interleukin 13 and tslp or tslpr and methods of use - Google Patents
Antibodies that bind interleukin 13 and tslp or tslpr and methods of use Download PDFInfo
- Publication number
- WO2024264003A2 WO2024264003A2 PCT/US2024/035140 US2024035140W WO2024264003A2 WO 2024264003 A2 WO2024264003 A2 WO 2024264003A2 US 2024035140 W US2024035140 W US 2024035140W WO 2024264003 A2 WO2024264003 A2 WO 2024264003A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- seq
- nos
- cdr
- antibody
- sequence
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/24—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against cytokines, lymphokines or interferons
- C07K16/244—Interleukins [IL]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/31—Immunoglobulins specific features characterized by aspects of specificity or valency multispecific
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
Definitions
- Interleukin (IL)-13 is a T helper cell subclass 2 (Th2) cytokine and belongs to a family of type I cytokines, exhibiting pleiotropic effects across multiple cellular pathways. IL-13 is involved in the differentiation of na ⁇ ve T cells into Th2 cells.
- IL-13 promotes B-cell proliferation and induces immunoglobulin isotype class switching to IgG4 and IgE when co- stimulated with CD40/CD40L. It also up-regulates Fc ⁇ RI, and thus, helps in IgE priming of mast cells. In monocytes/macrophages, IL-13 up-regulates expression of CD23 and MHC class I and class II antigens, down-regulates the expression of CD14, inhibits antibody- dependent cytotoxicity, and promotes eosinophil survival, activation, and recruitment. IL-13 also manifests important functions on nonhematopoietic cells, such as smooth muscle cells, epithelial cells, endothelial cells, and fibroblast cells.
- IL-13 enhances proliferation and cholinergic-induced contractions of smooth muscles.
- IL-13 is a potent inducer of chemokine production, alters mucociliary differentiation, decreases ciliary beat frequency of ciliated epithelial cells, and results in goblet cell metaplasia.
- IL-13 is a potent inducer of vascular cell adhesion molecule 1 (VCAM-1), which is important for recruitment of eosinophils.
- VCAM-1 vascular cell adhesion molecule 1
- IL-13 reduces the expression of barrier integrity molecules, such as filaggrin and loricrin, while stimulating CCL26 and CCL2 secretion responsible for the recruitment of several inflammatory cells of myeloid lineages.
- Thymic stromal lymphopoietin is an epithelial cell derived cytokine produced in response to pro-inflammatory stimuli.
- TSLP has been discovered to promote allergic inflammatory responses primarily through its activity on dendritic and mast cells.
- Human TSLP expression has been reported to be increased in asthmatic airways correlating to disease severity.
- TSLP protein levels are detectable in the concentrated bronchoalveolar lavage (BAL) fluid of asthma patients, and other patients suffering from allergic disorders.
- BAL concentrated bronchoalveolar lavage
- TSLP binds to a heterodimeric receptor consisting of the TSLP receptor (TSLPR) and an IL-7 receptor ⁇ chain (IL-7R ⁇ ) in dendritic cells, thereby activating the dendritic cells.
- the dendritic cells express inflammatory chemokines such as thymus and activation regulated chemokines (TARC (CCL17)), macrophage-derived chemokines (MDC (CCL22)), and the like.
- TARC thymus and activation regulated chemokines
- MDC macrophage-derived chemokines
- Activation of dendritic cells by TSLP through the TSLP receptor is associated with disease pathology, including allergic inflammatory diseases, such as asthma, and autoimmune disease, such as systemic sclerosis.
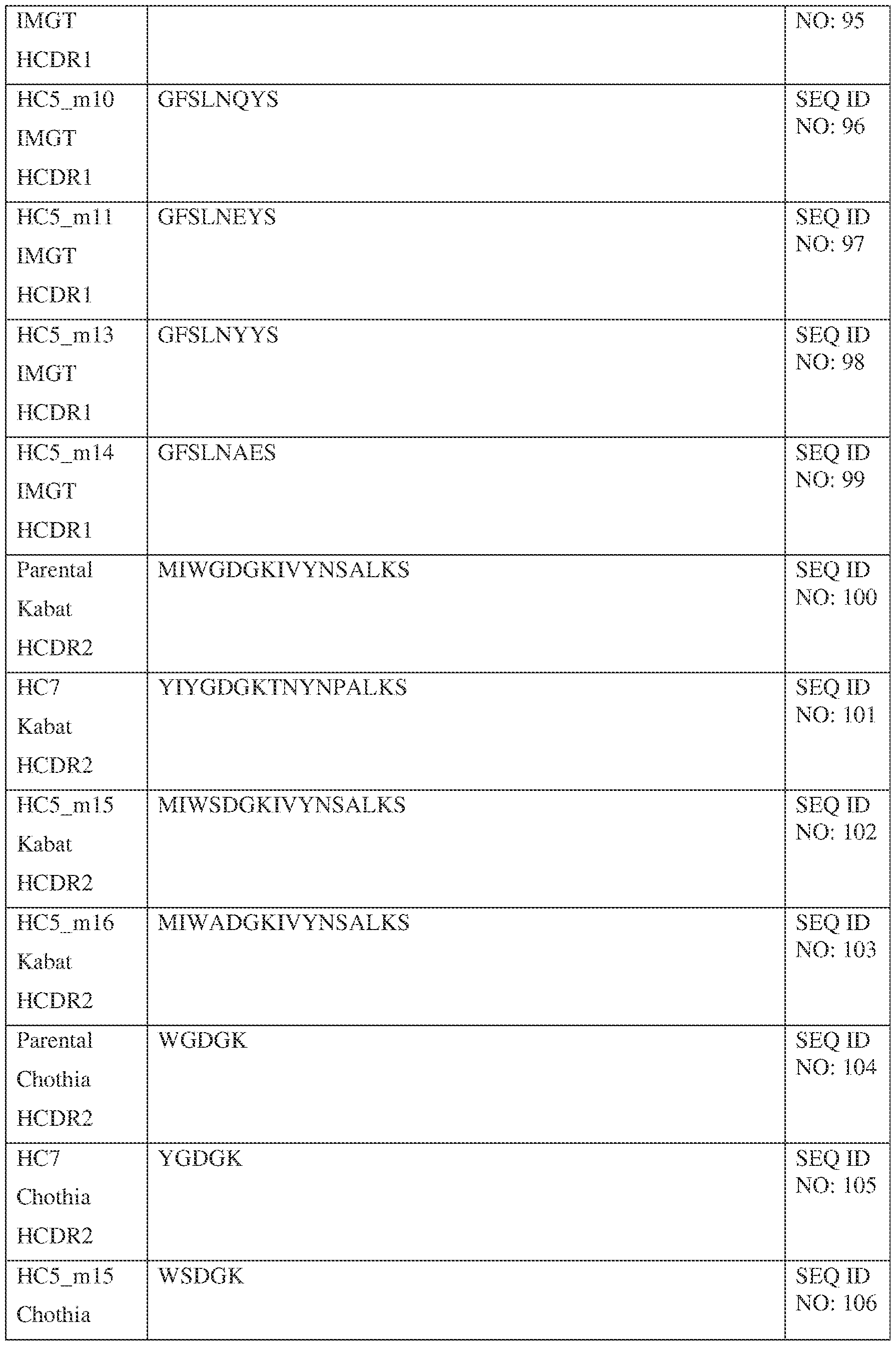
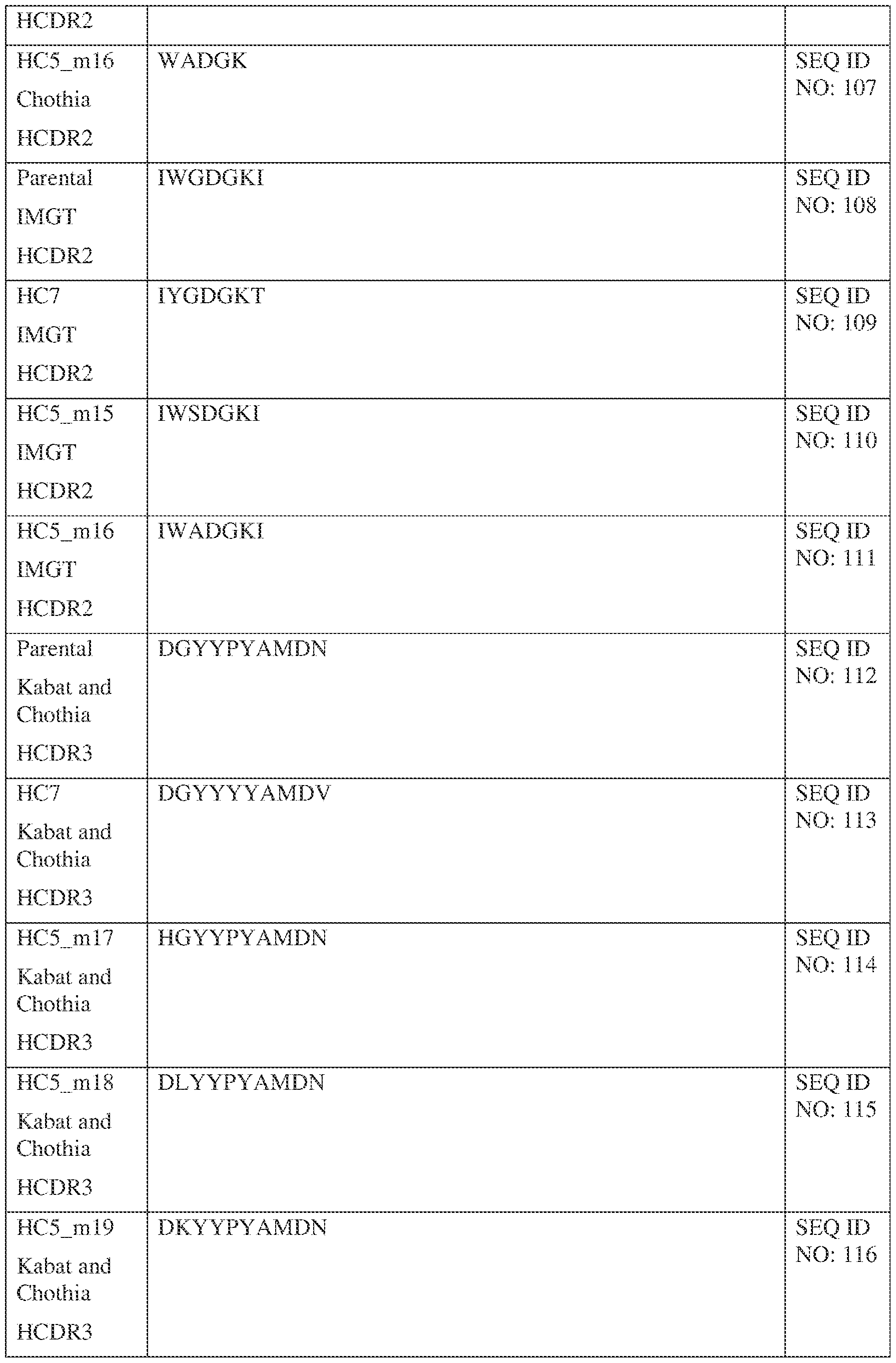
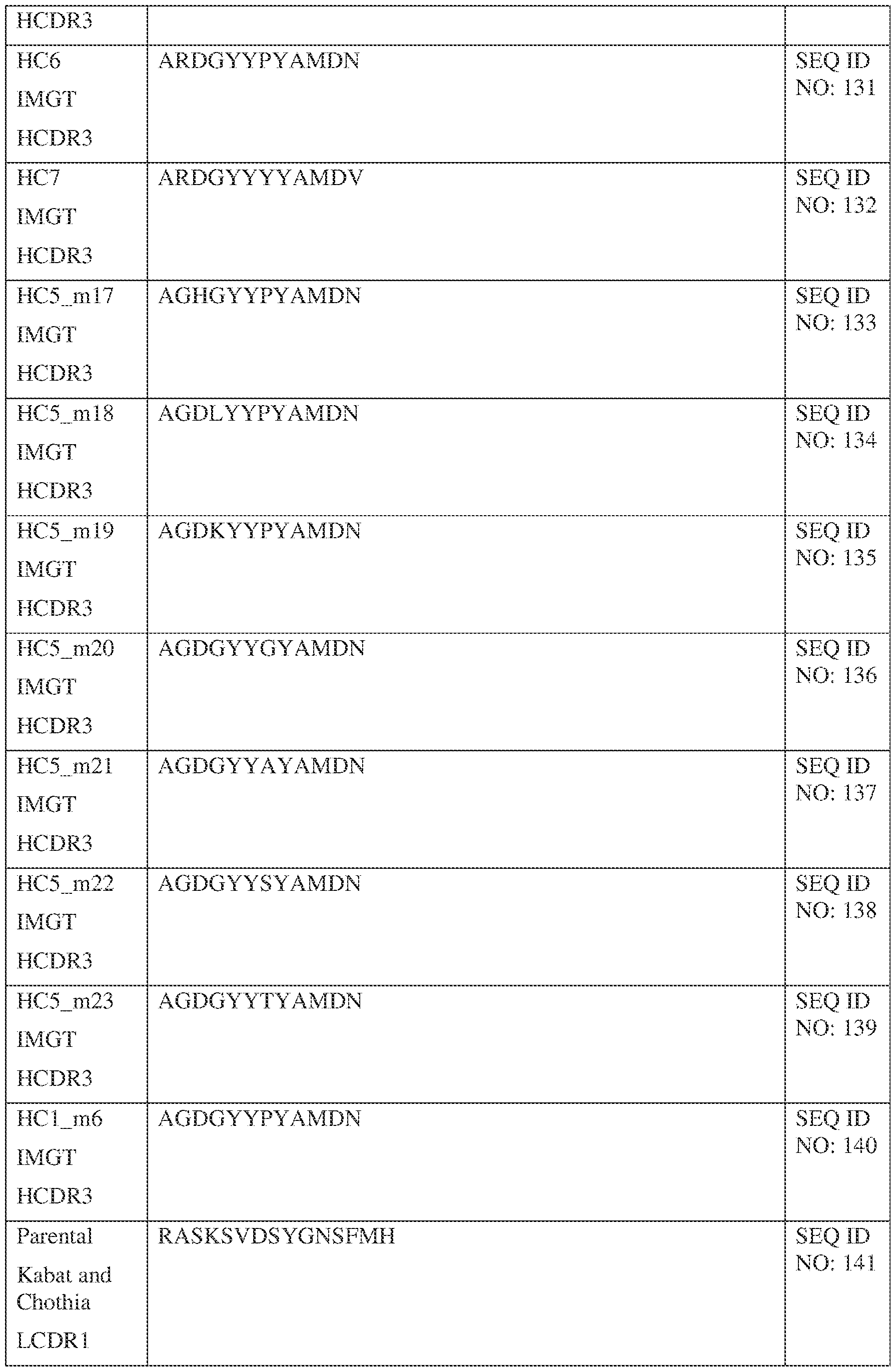
- a bispecific antibody comprising a first antigen binding site and a second antigen binding site, wherein the first antigen binding site binds Interleukin 13 (IL-13) and comprises a first variable heavy (VH) chain sequence comprising three first heavy chain CDR sequences, a first CDR-H1, a first CDR-H2, and a first CDR-H3; and a first variable light (VL) chain sequence comprising three first light chain CDR sequences, a first CDR-L1, a first CDR-L2, and a first CDR-L3; wherein: the first CDR-H1 comprises a sequence selected from the sequences set forth in SEQ ID NOs: 58-99 and 121;the first CDR-H2 comprises a sequence selected from the sequences set forth in SEQ ID NOs: 100-111;the first CDR-H3 comprises a sequence selected from the sequences set forth in SEQ ID NOs: 112-120 and
- the second CDR-L1 comprises a sequence selected from the sequences set forth in SEQ ID NOs: 637-645.
- the second CDR-L2 comprises a sequence selected from the sequences set forth in SEQ ID NOs: 646-651;
- the second CDR-L3 comprises a sequence selected from the sequences set forth in SEQ ID NOs: 652-657.
- the first CDR-H1 comprises a sequence selected from the sequences set forth in SEQ ID NOs: 58, 68, and 85;
- the first CDR-H2 comprises a sequence selected from the sequences set forth in SEQ ID NOs: 100, 104; and 108;
- the first CDR-H3 comprises a sequence selected from the sequences set forth in SEQ ID NOs: 112 and 130;
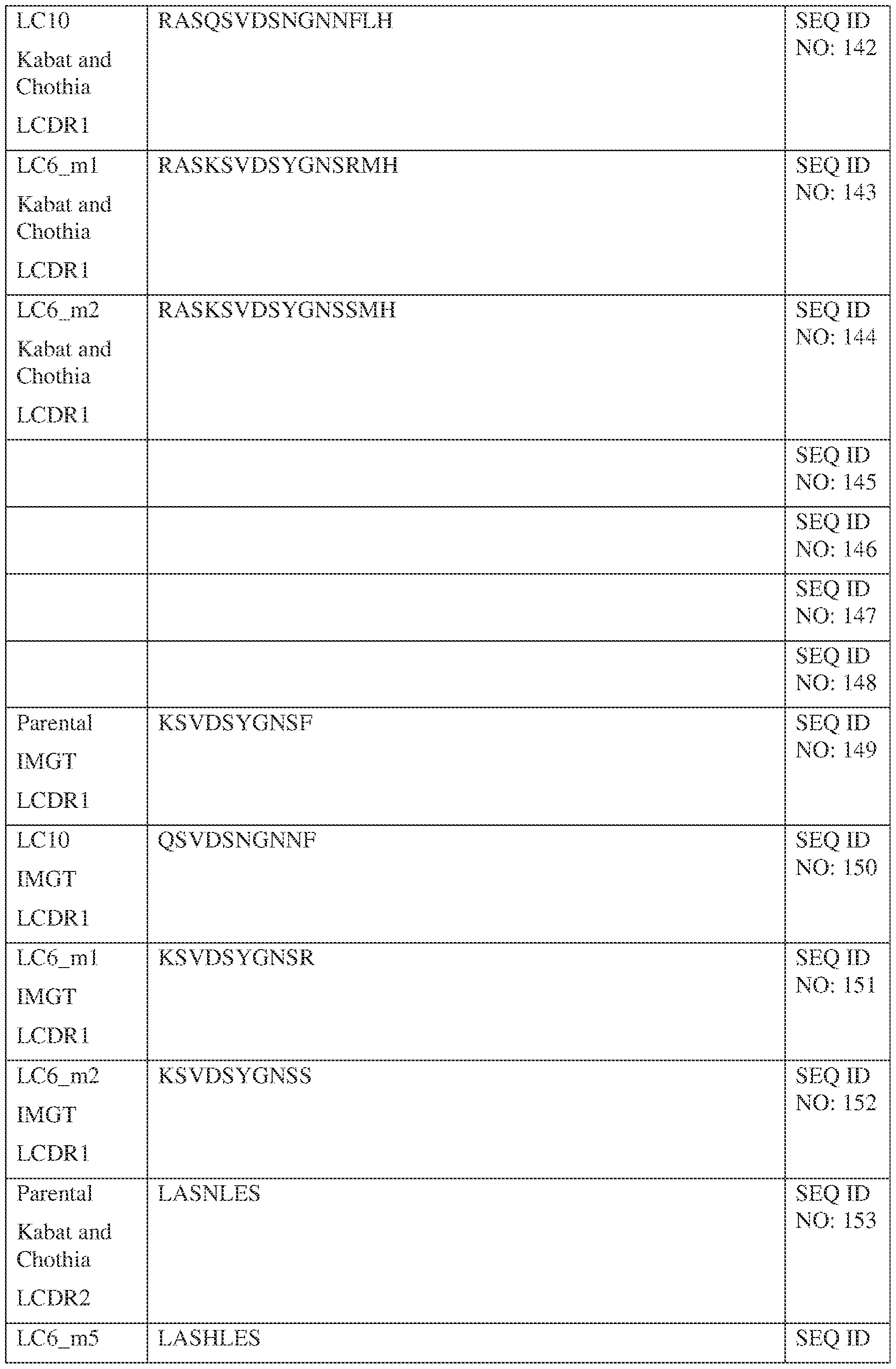
- the first CDR-L1 comprises a sequence selected from the sequences set forth in SEQ ID NOs: 141 and 589;
- the first CDR-L2 comprises a sequence selected from the sequences set forth in SEQ ID NOs: 153 and LAS;
- the first CDR-L3 comprises a sequence set forth in SEQ ID NOs: 165;
- the second CDR-H1 comprises a sequence selected from the sequences set forth in SEQ ID NOs: 610-618;
- the second CDR-H2 comprises a sequence selected from the sequences set forth in SEQ ID NOs: 619
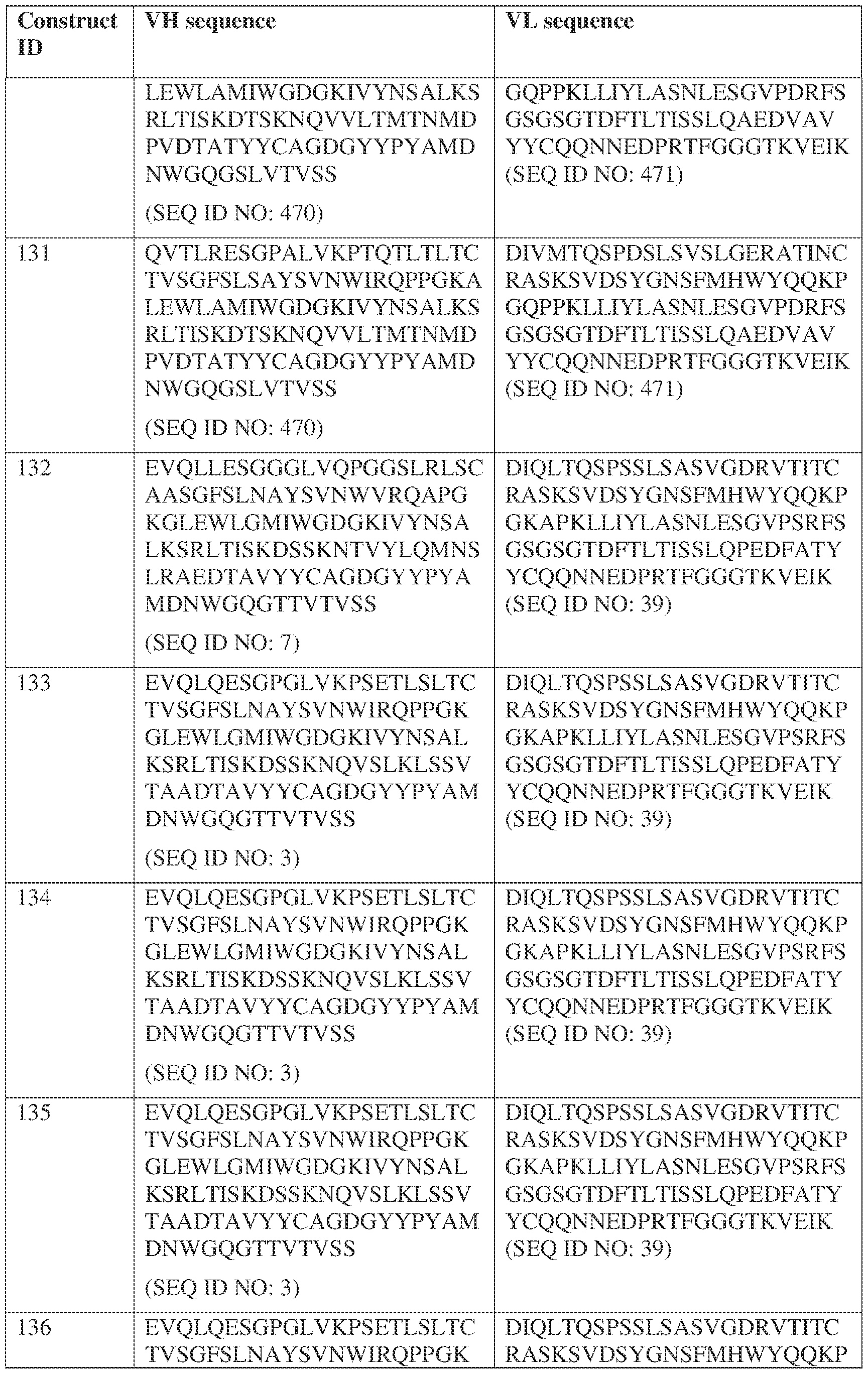
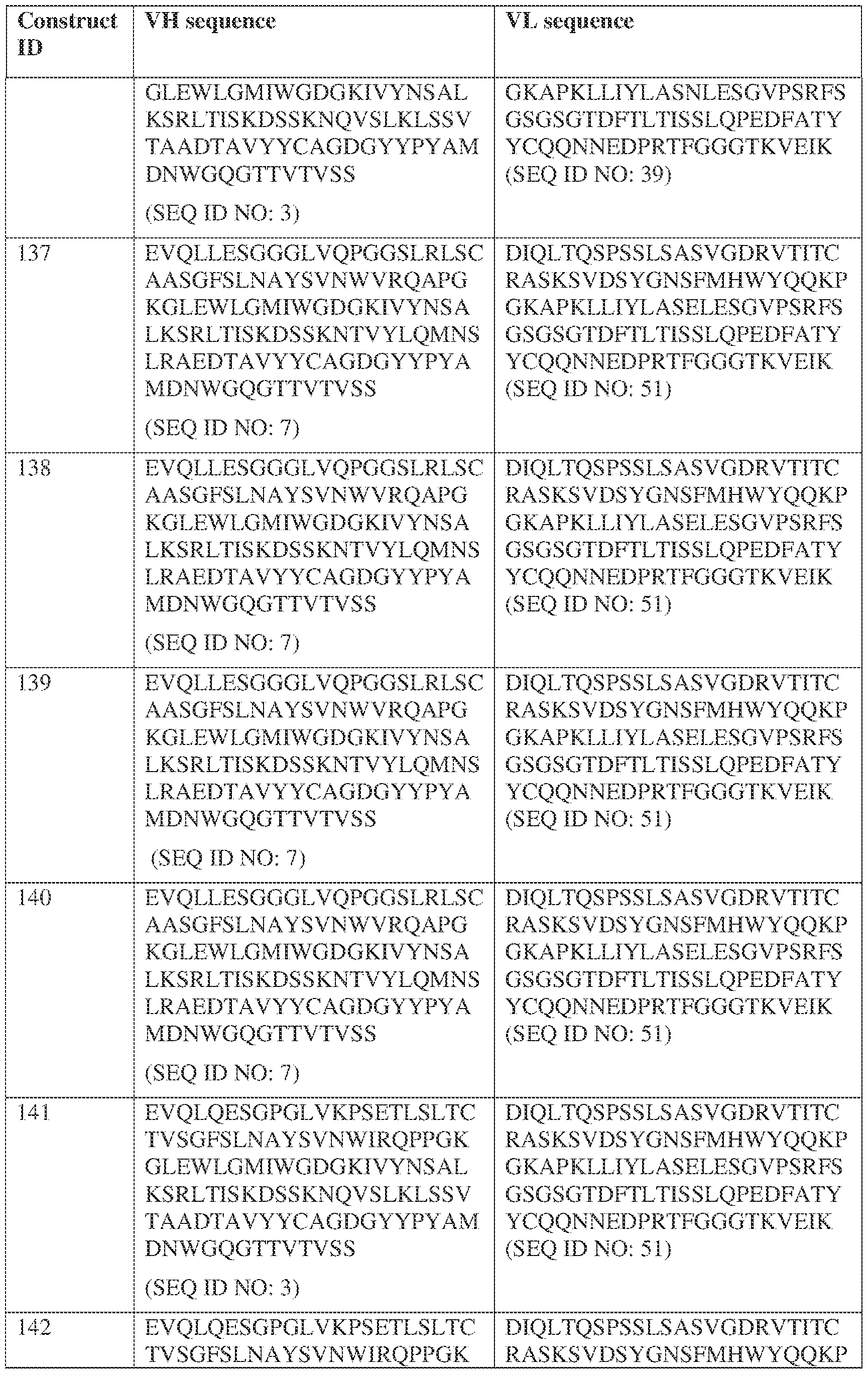
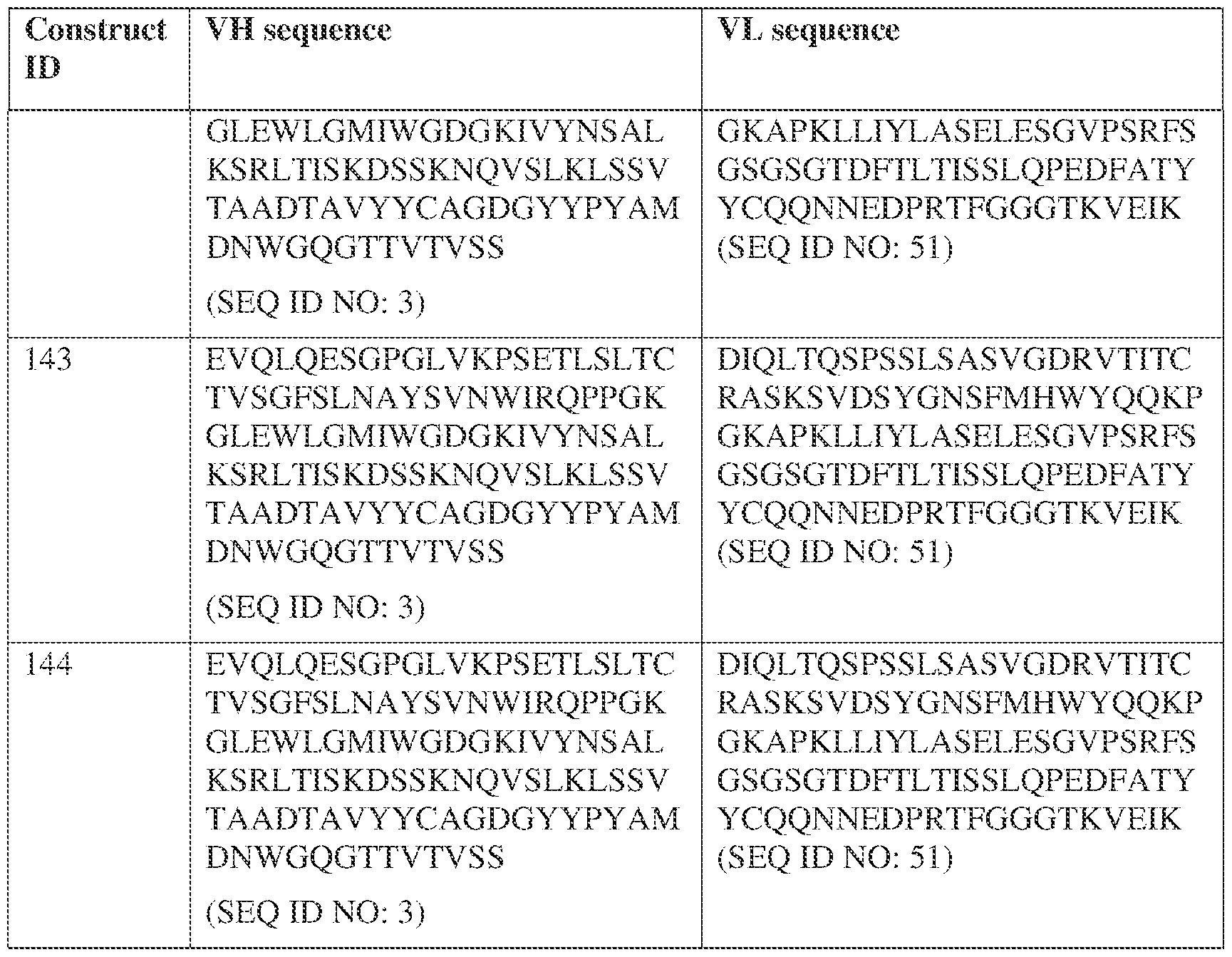
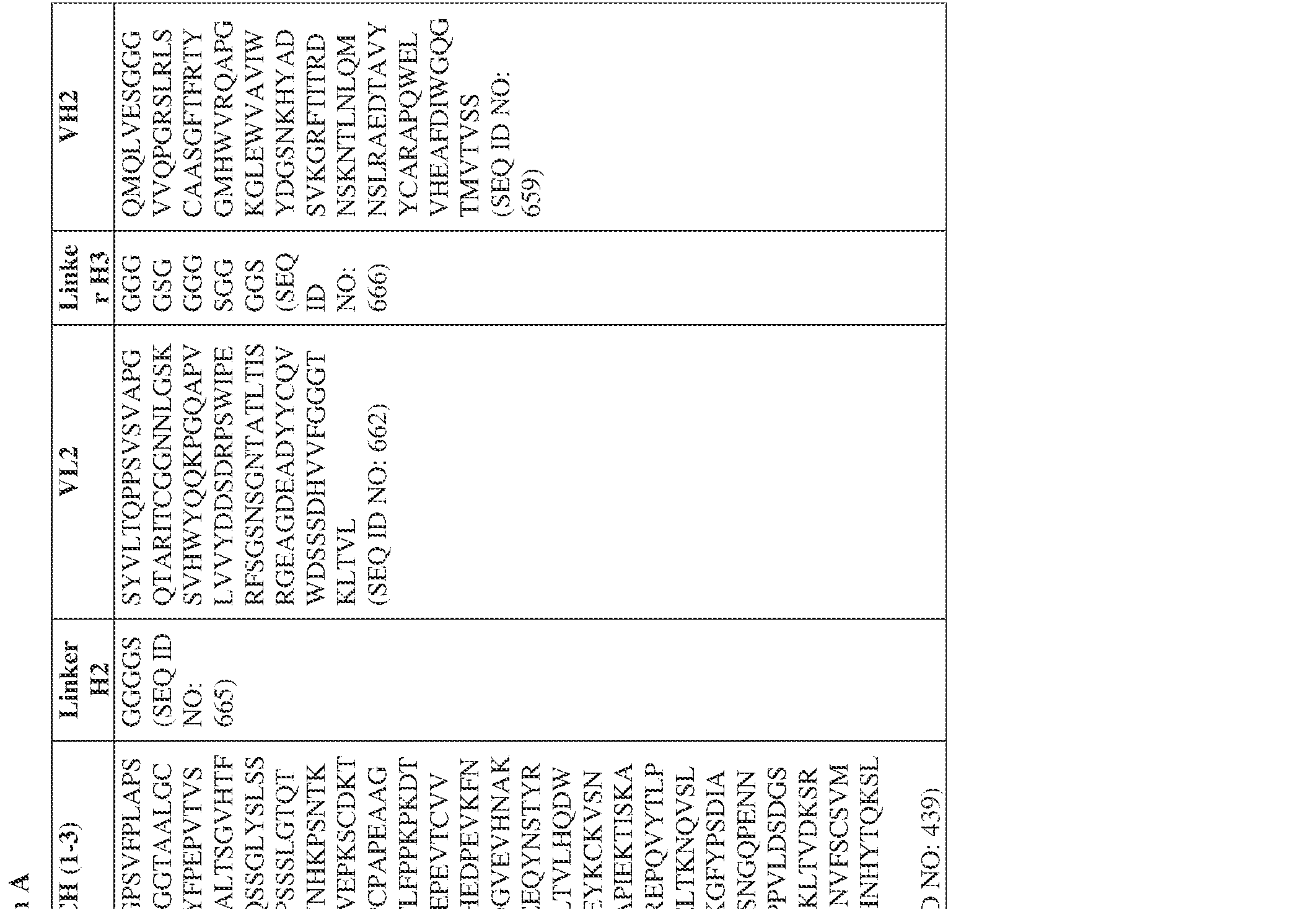
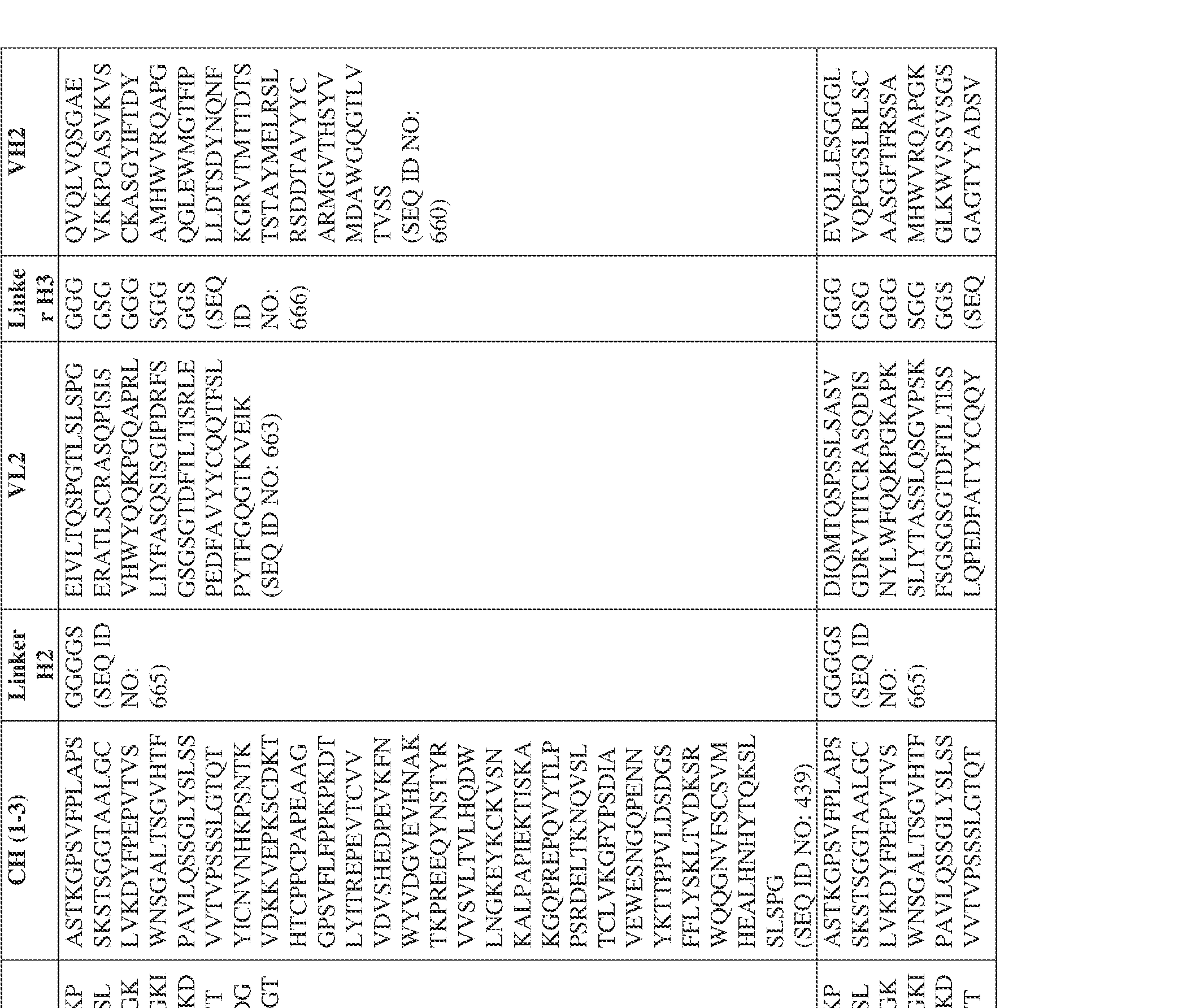
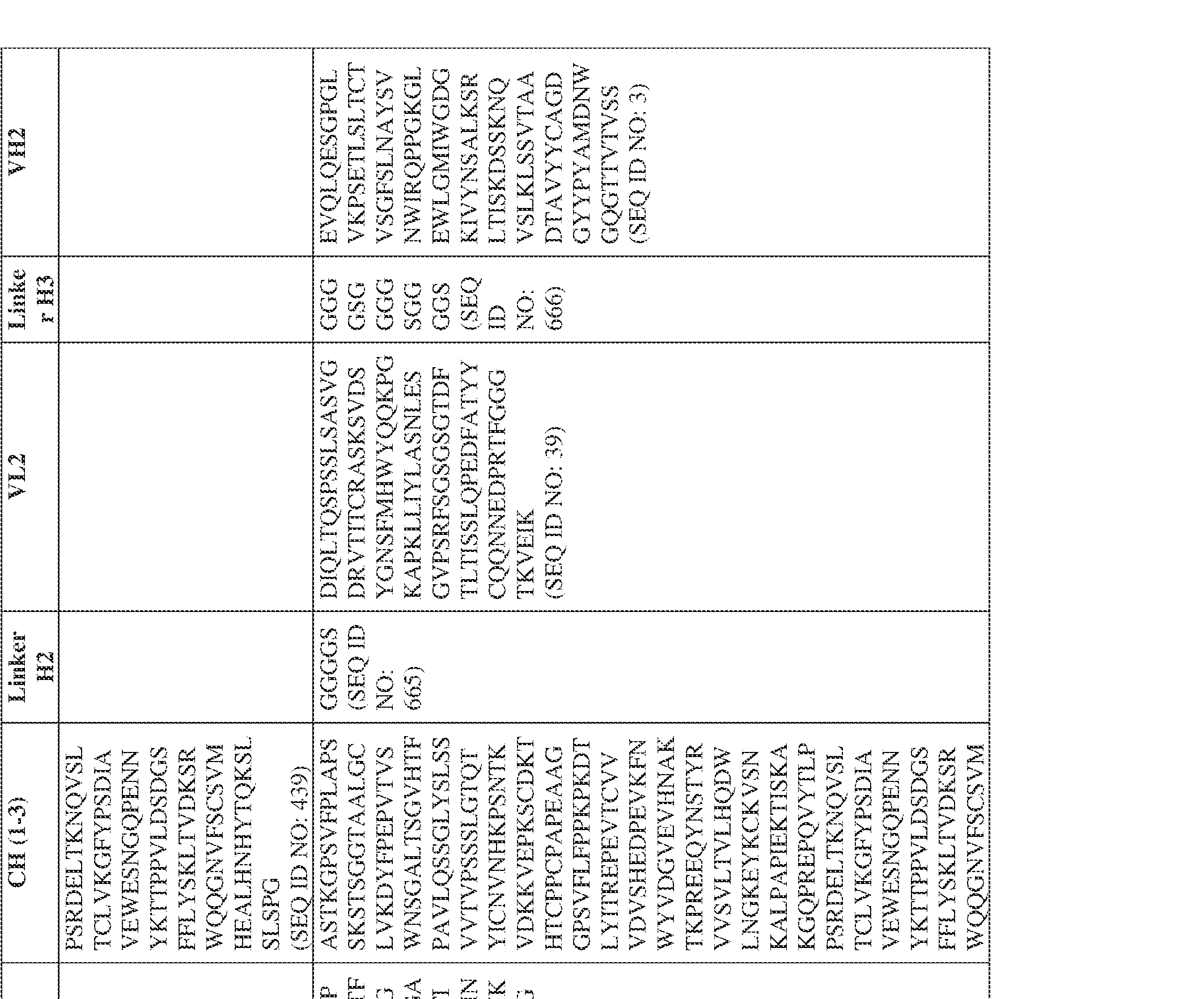
- the antibody comprises a first VH sequence selected from SEQ ID NOs: 1-32, 470, and 688 and a second VH sequence selected from SEQ ID NOs: 659-661, 682, 684, 686, and 688.
- the antibody comprises a first VH sequence comprising SEQ ID NO: 3 and a second VH sequence selected from SEQ ID NOs: 659-661, 682, 684, 686, and 688.
- the antibody comprises a first VL sequence selected from SEQ ID NOs: 33-57, 471, and 687 and a second VL sequence selected from SEQ ID NOs: 662-664, 681, 683, and 685.
- the bispecific antibody comprises a first VL comprises a first VL sequence comprising SEQ ID NO: 39 and a second VL sequence selected from SEQ ID NOs: 662-664, 681, 683, and 685. [0013] In some embodiments, the bispecific antibody comprises a first VH sequence selected from SEQ ID NOs: 1-32, 470, and 688 and a second VH sequence selected from IPTS/128623945.3 3 Attorney Docket No.
- the bispecific antibody comprises a first VH sequence comprising SEQ ID NOs: 3 and a second VH sequence selected from SEQ ID NOs: 659-661, 682, 684, 686, and 688; and a first VL sequence comprising SEQ ID NOs: 39 and a second VL sequence selected from SEQ ID NOs: 662-664, 681, 683, and 685.
- the bispecific antibody is a humanized, human, or chimeric antibody.
- the bispecific antibody is a humanized antibody.
- the bispecific antibody comprises a heavy chain human constant region of a class selected from IgG, IgA, IgD, IgE, and IgM.
- the human Fc region comprises a human heavy chain constant region of the class IgG and a subclass selected from IgG1, IgG2, IgG3, and IgG4.
- the human Fc region comprises a human IgG1 Fc.
- the human Fc region comprises a human IgG4 Fc.
- the human Fc region comprises a human IgG2 Fc.
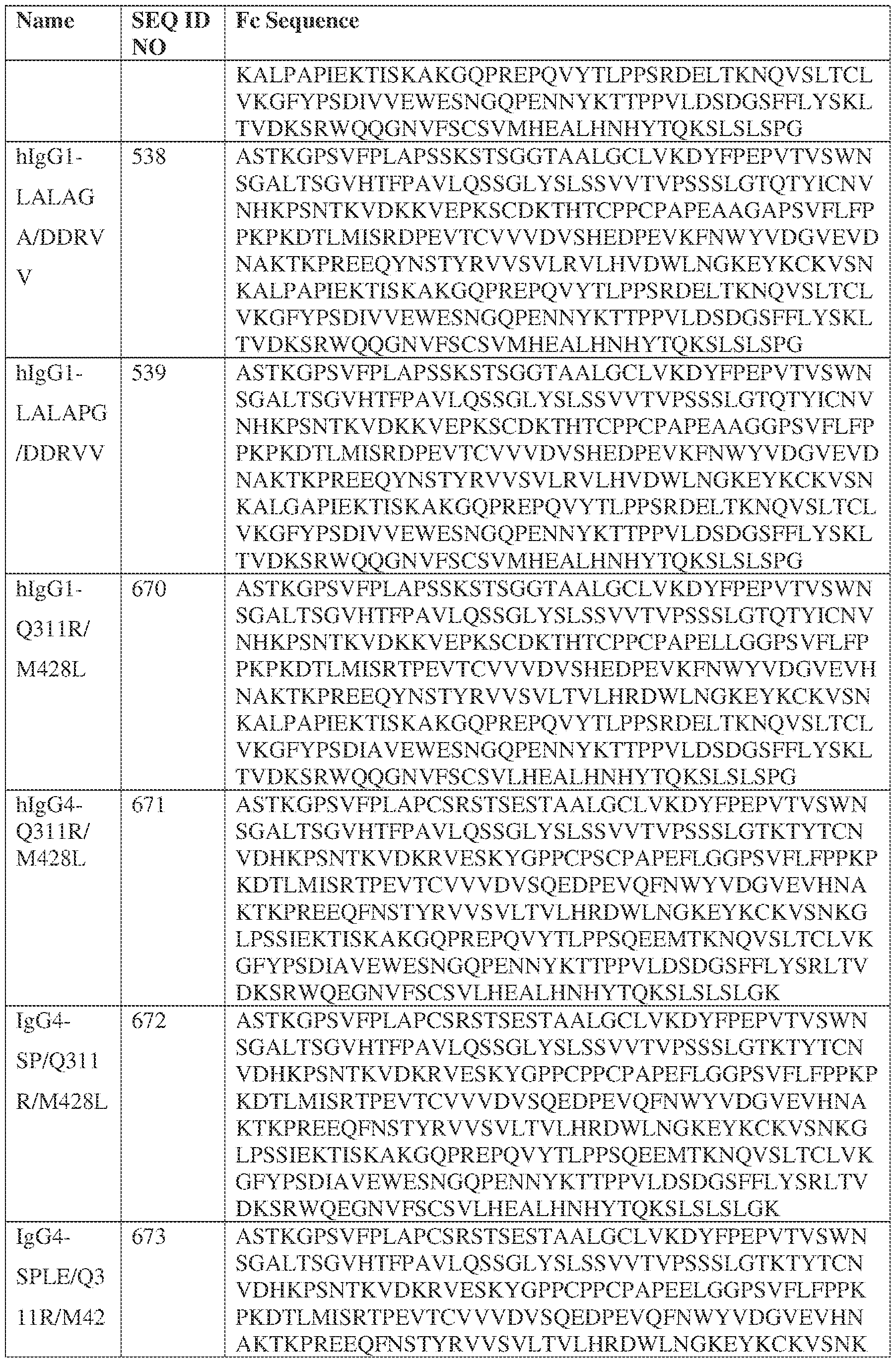
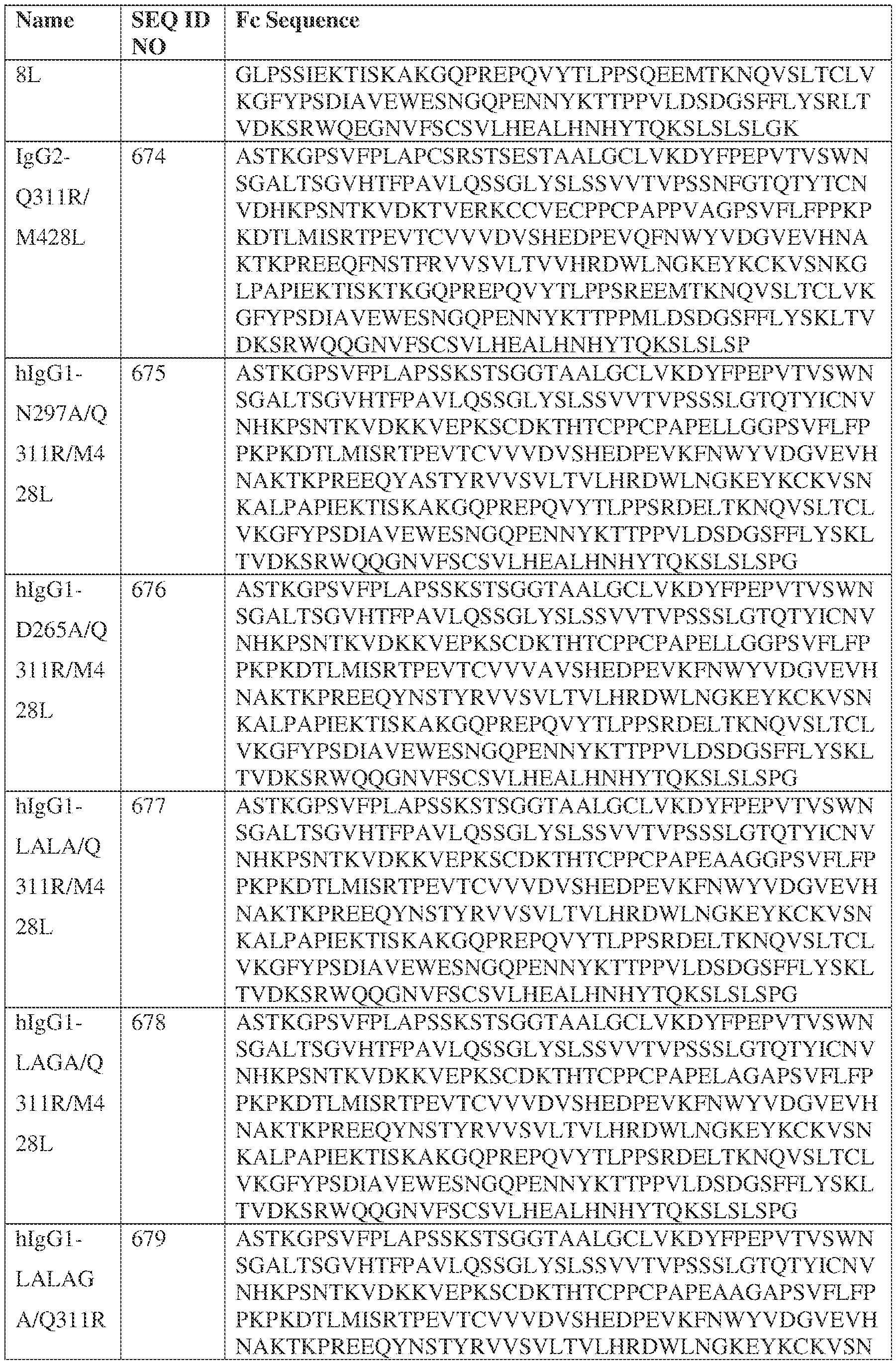
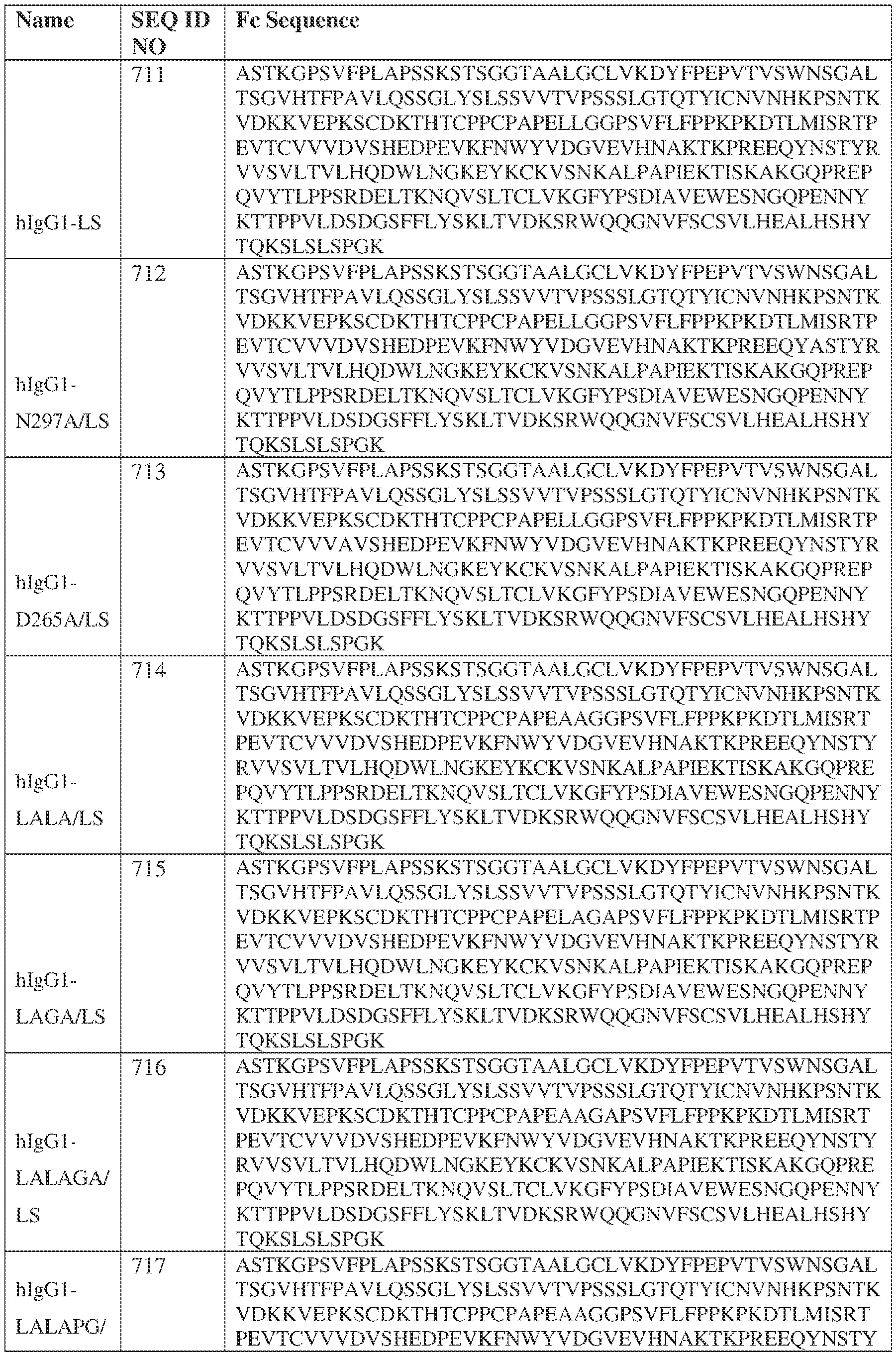
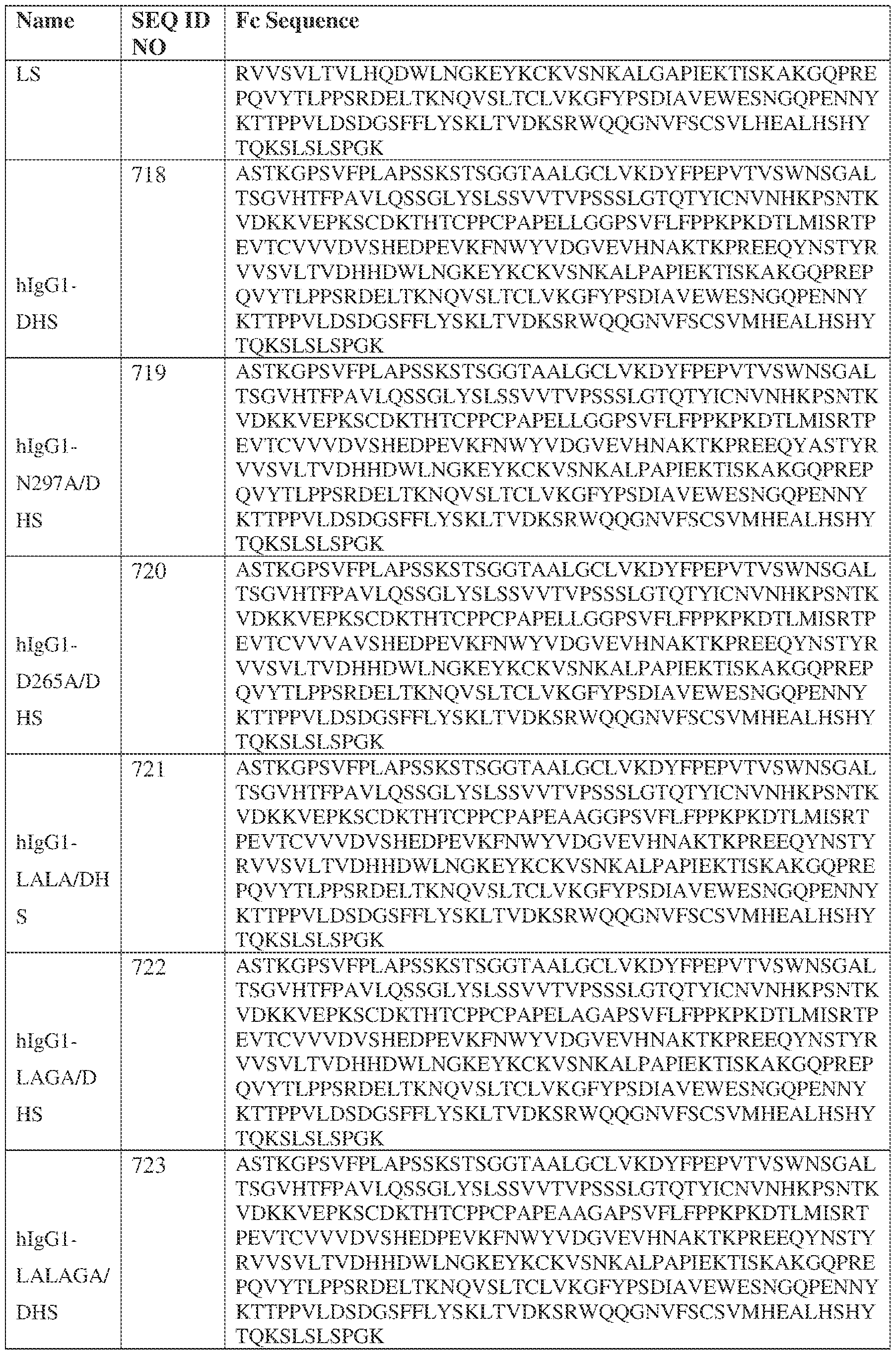
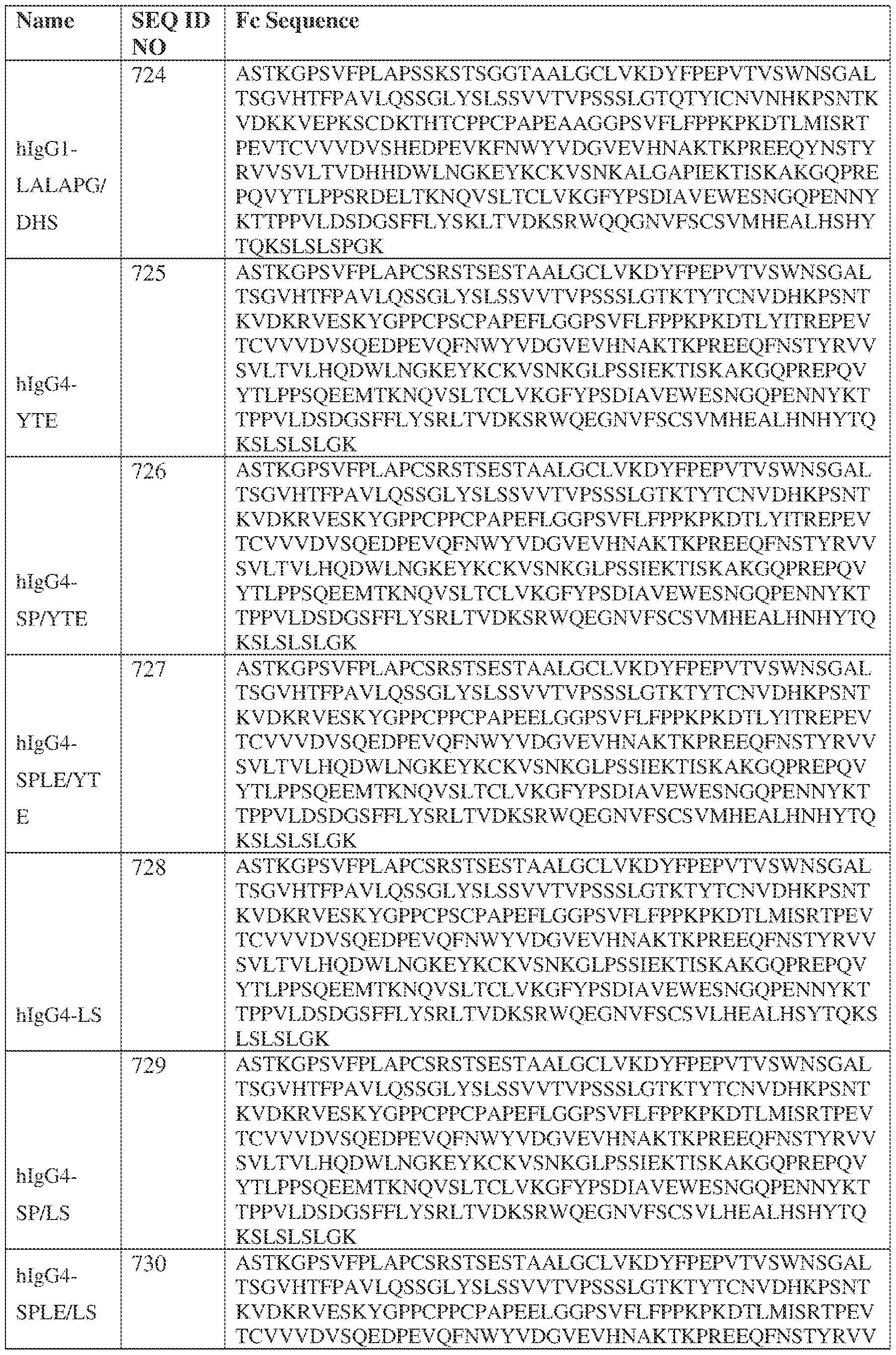
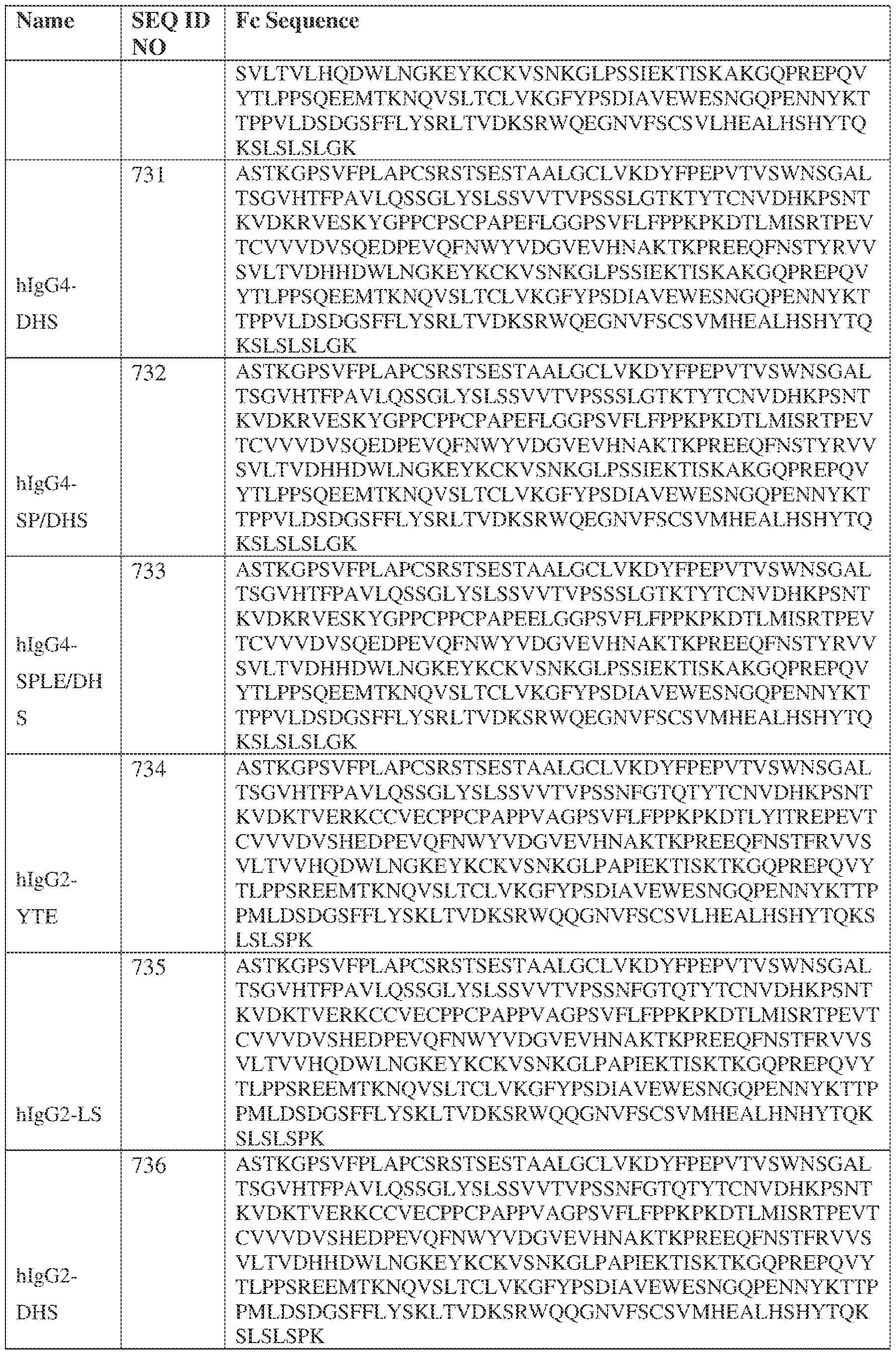
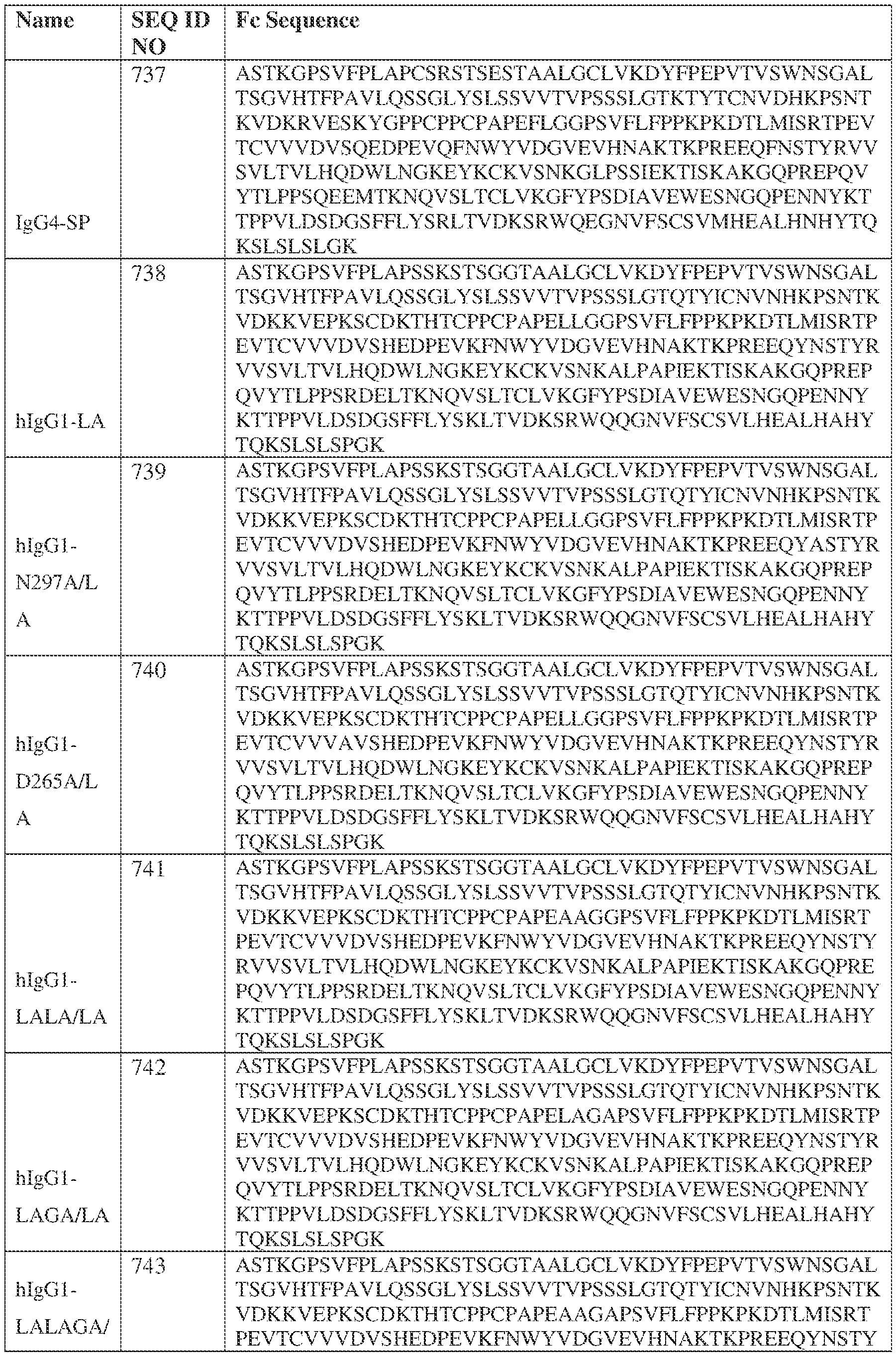
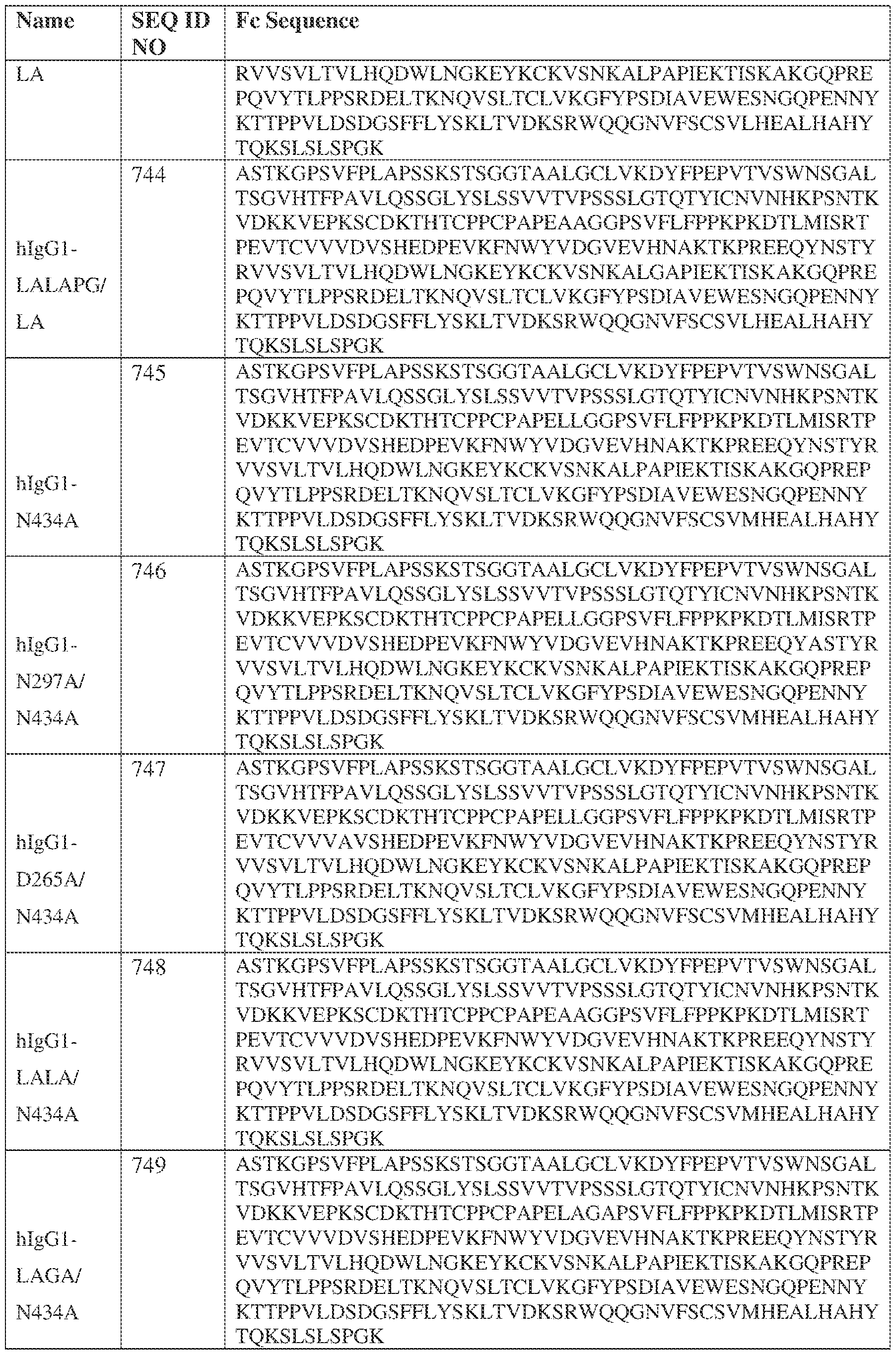
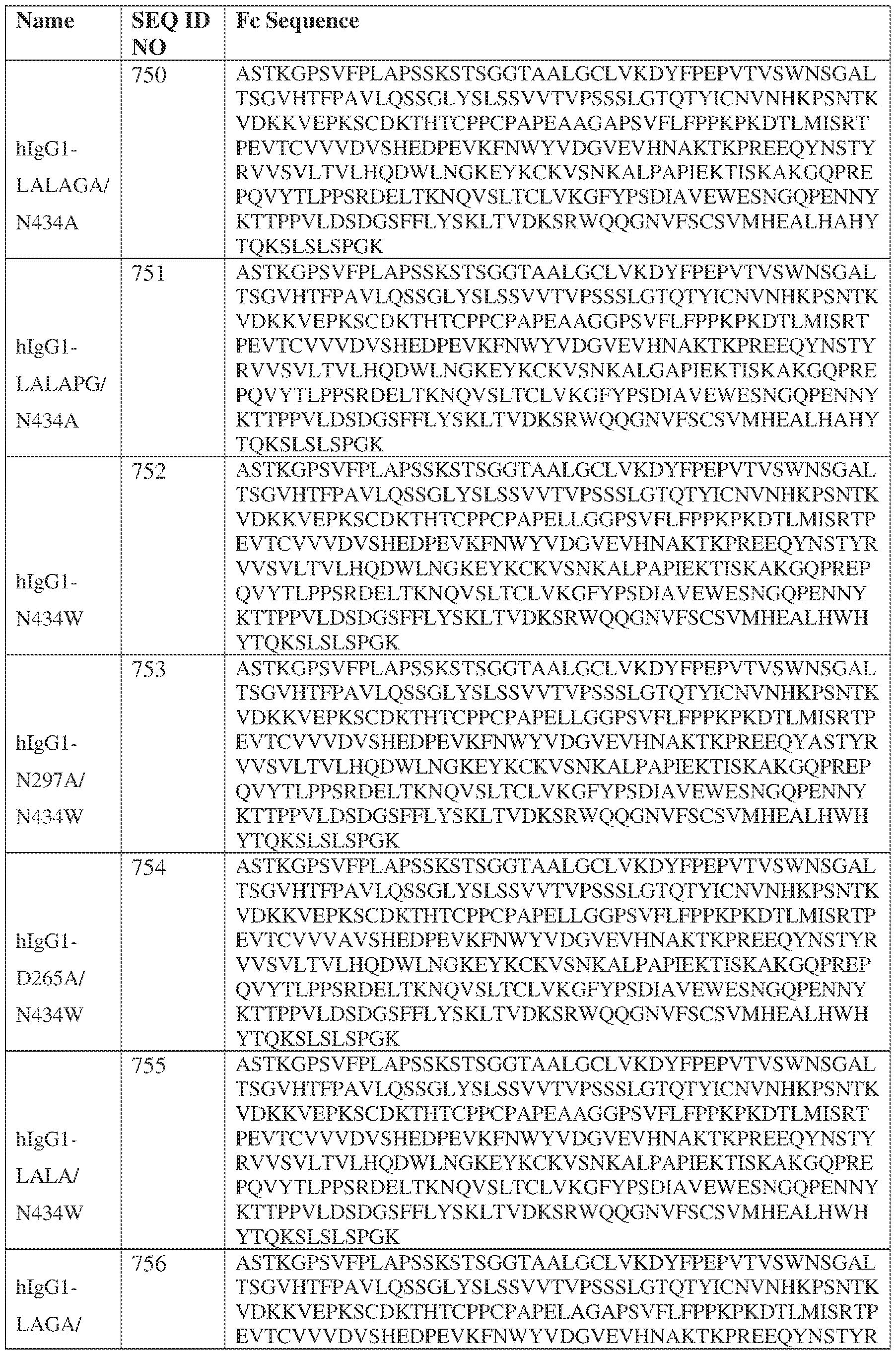
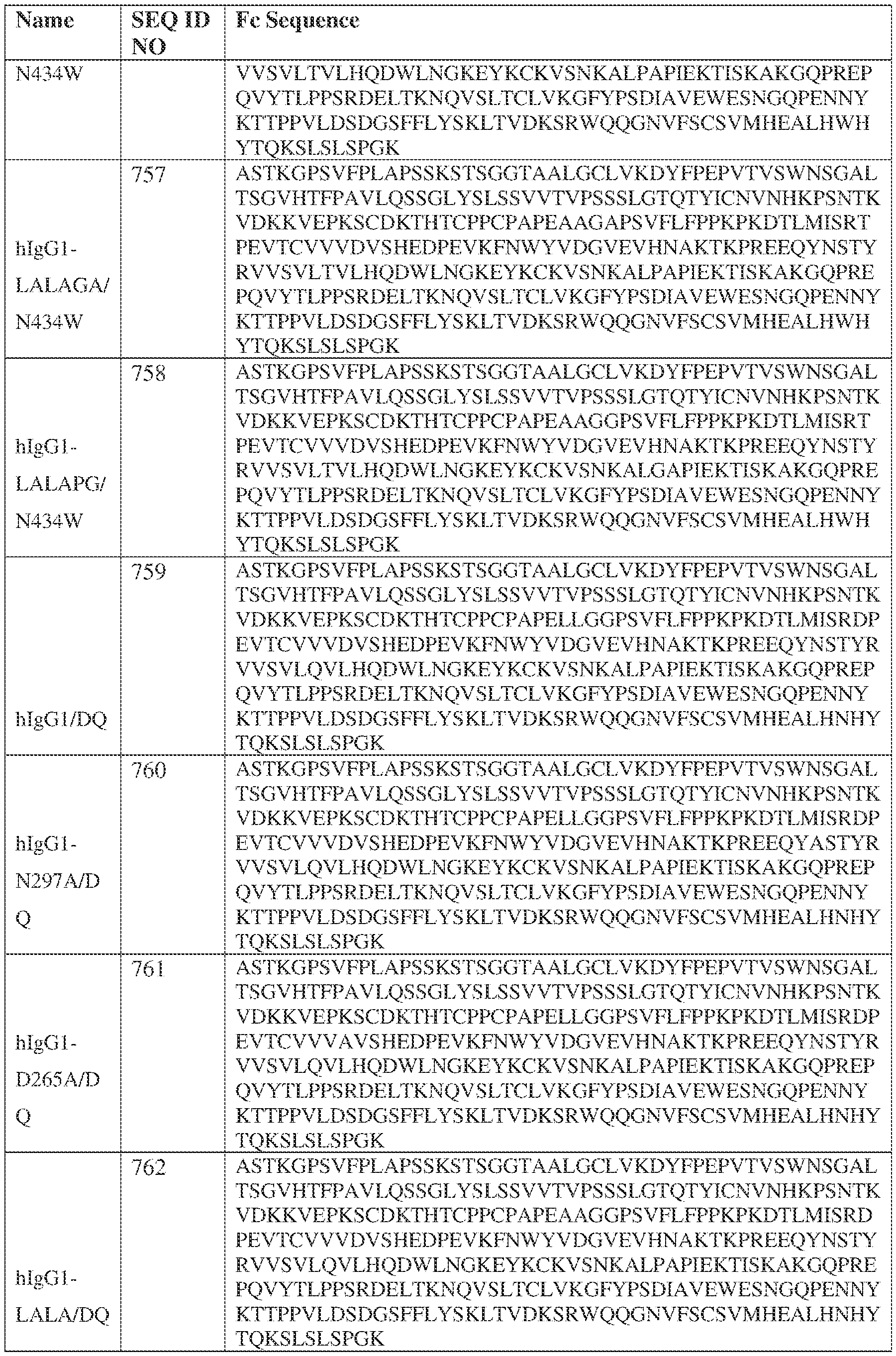
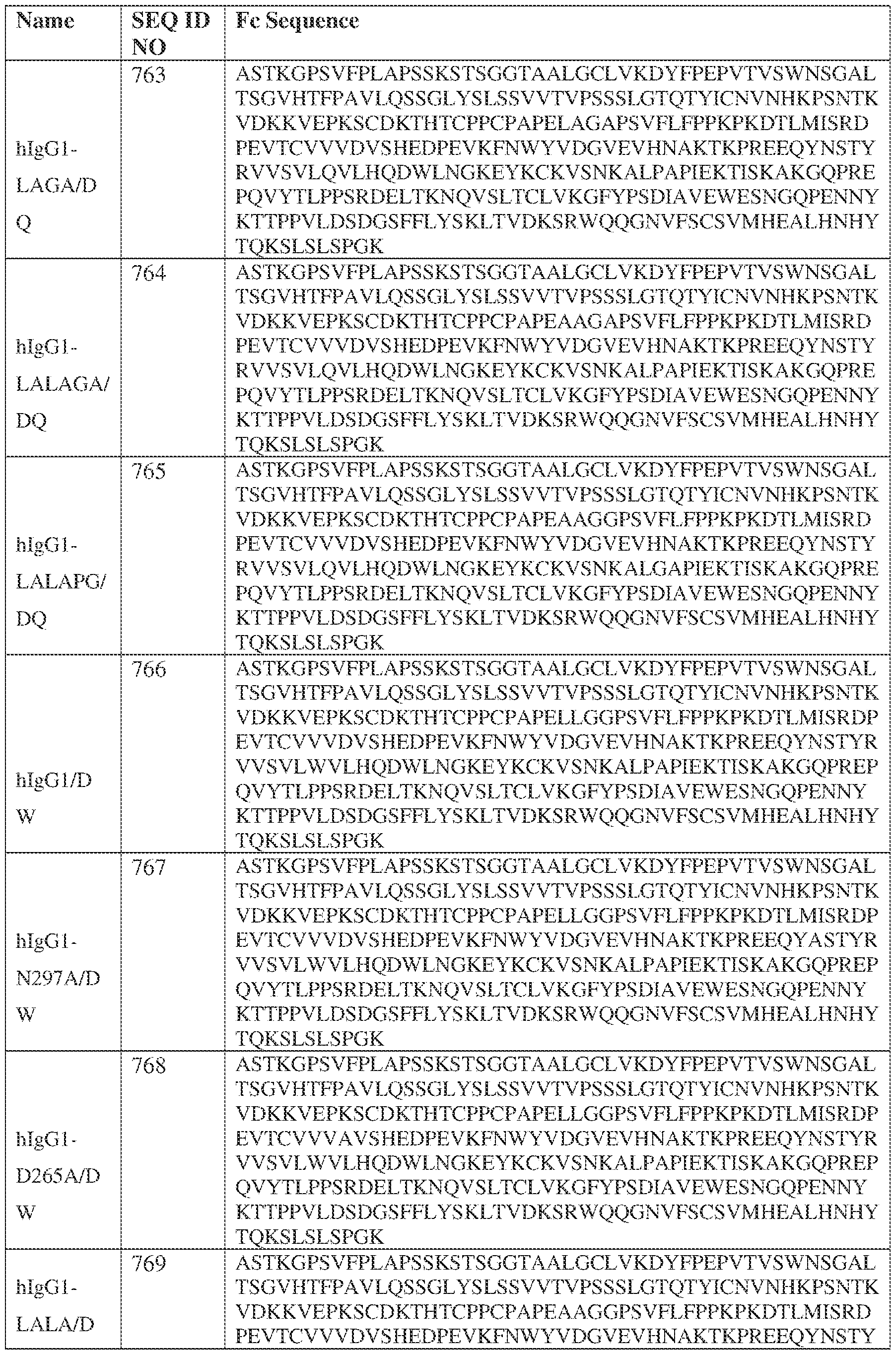
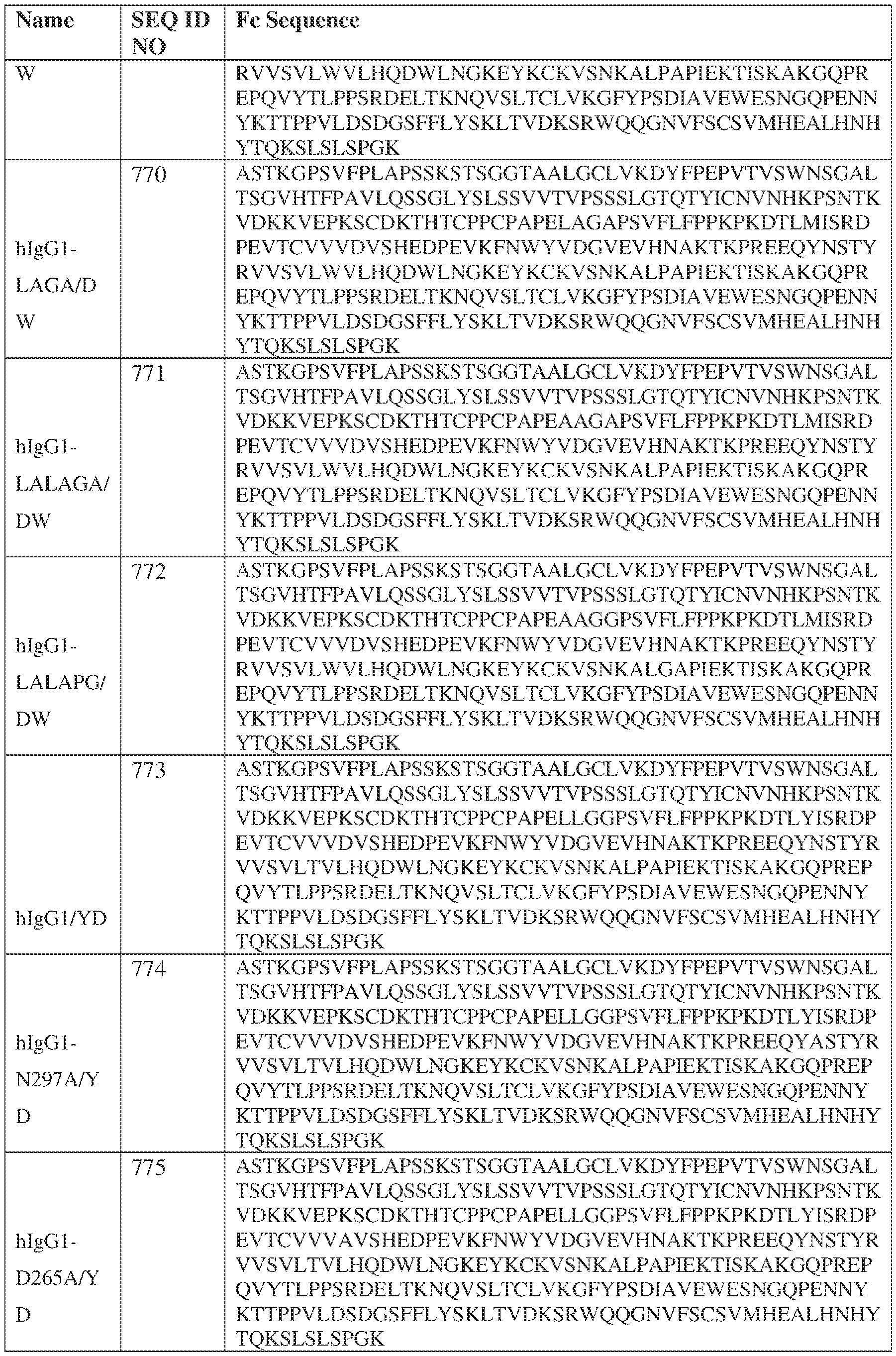
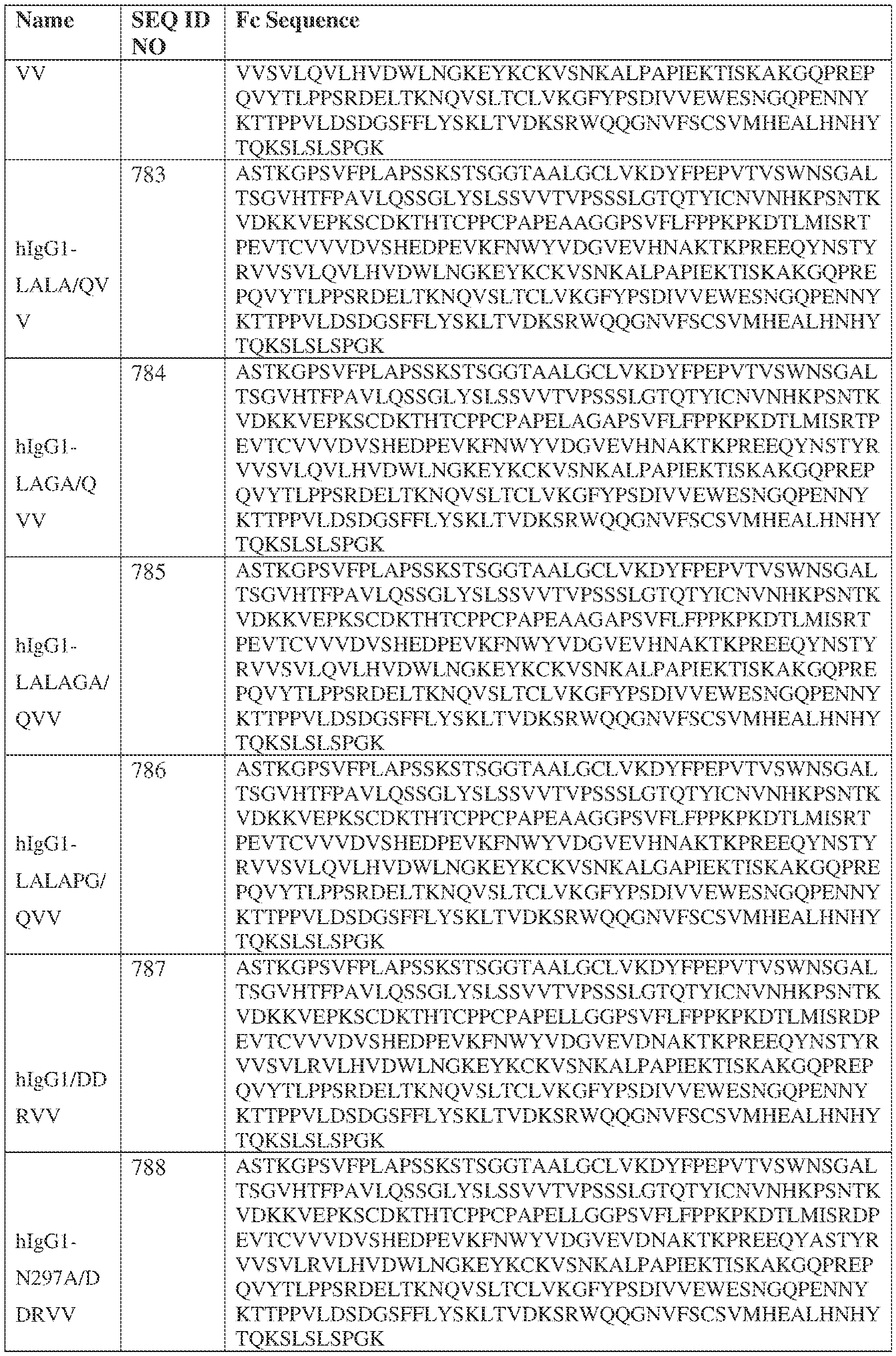
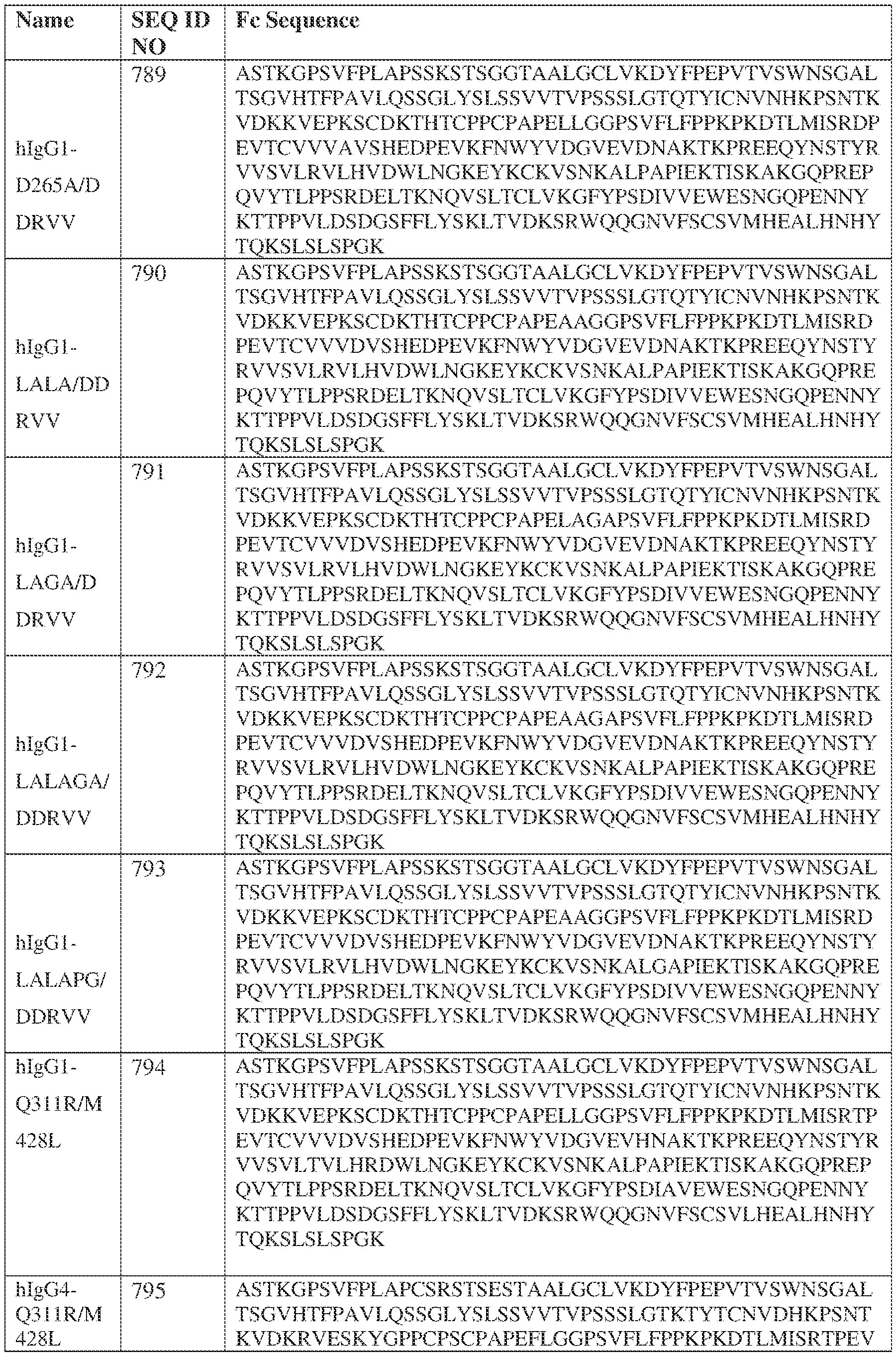
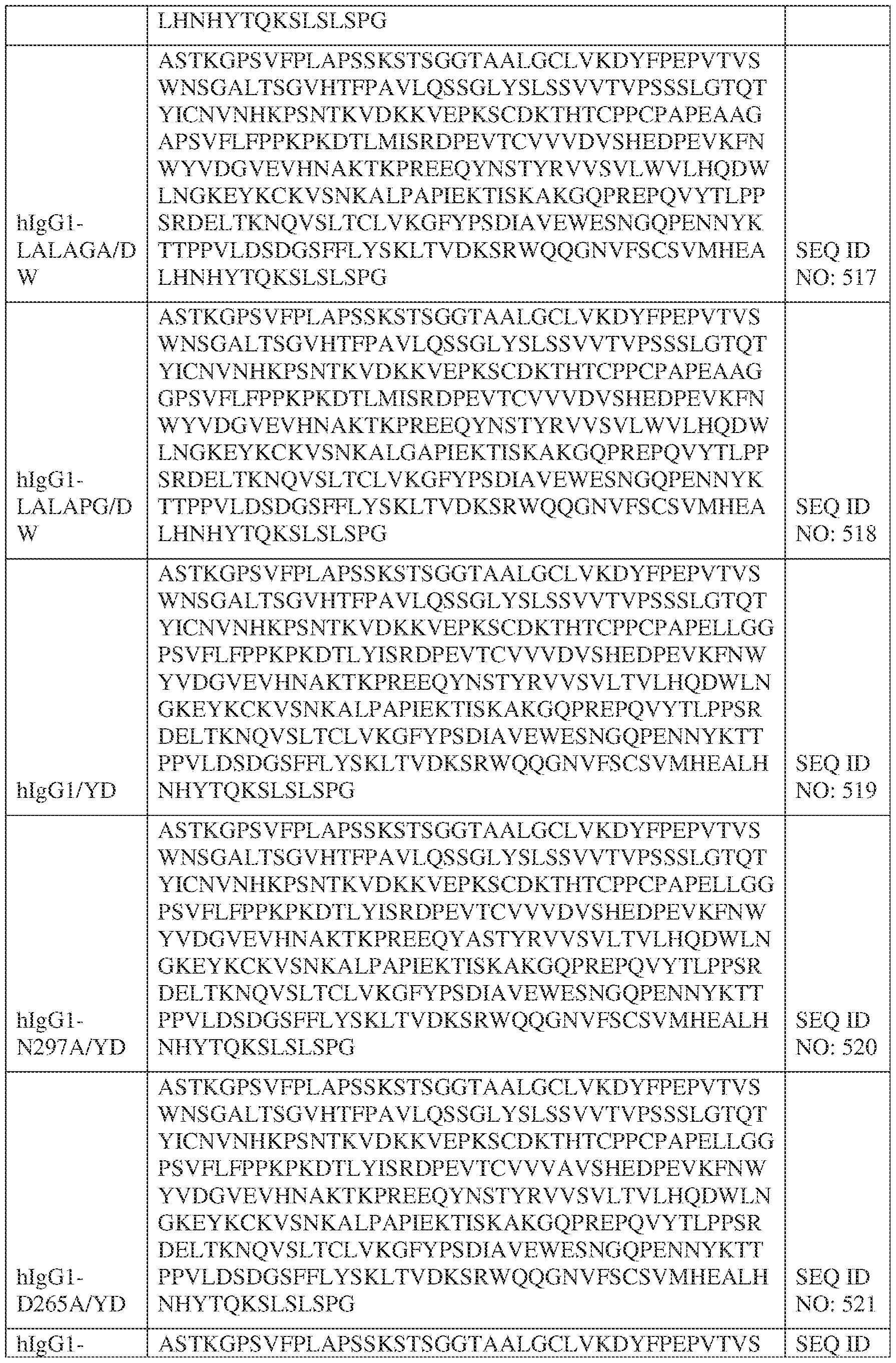
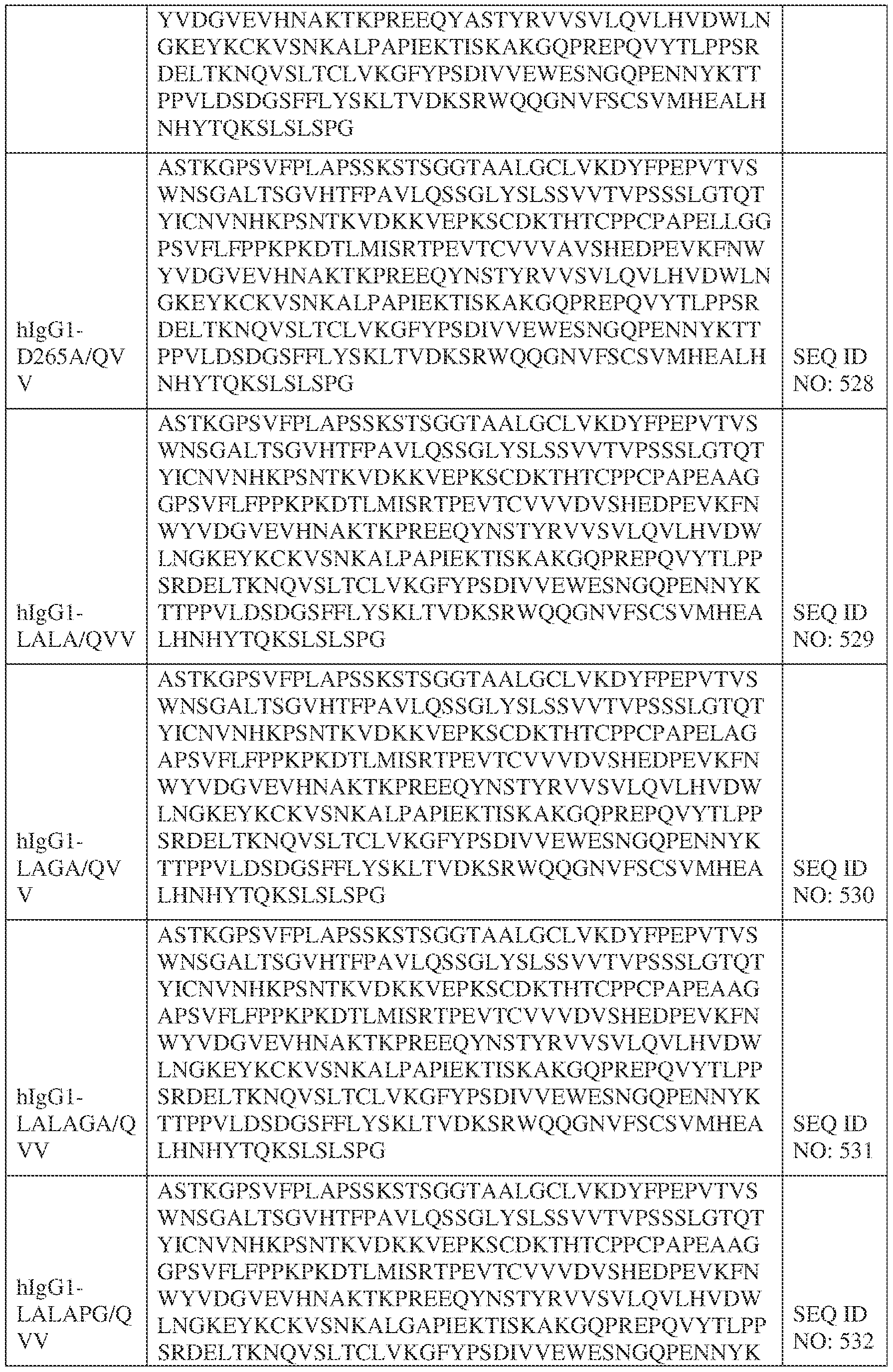
- the heavy chain comprises an Fc sequence selected from the sequences set forth in SEQ ID NOs: 425-468, 484-539, 670-680, 689-691, and 693-804. [0023] In some embodiments, the heavy chain comprises a constant heavy chain sequence set forth in SEQ ID NO: 439.
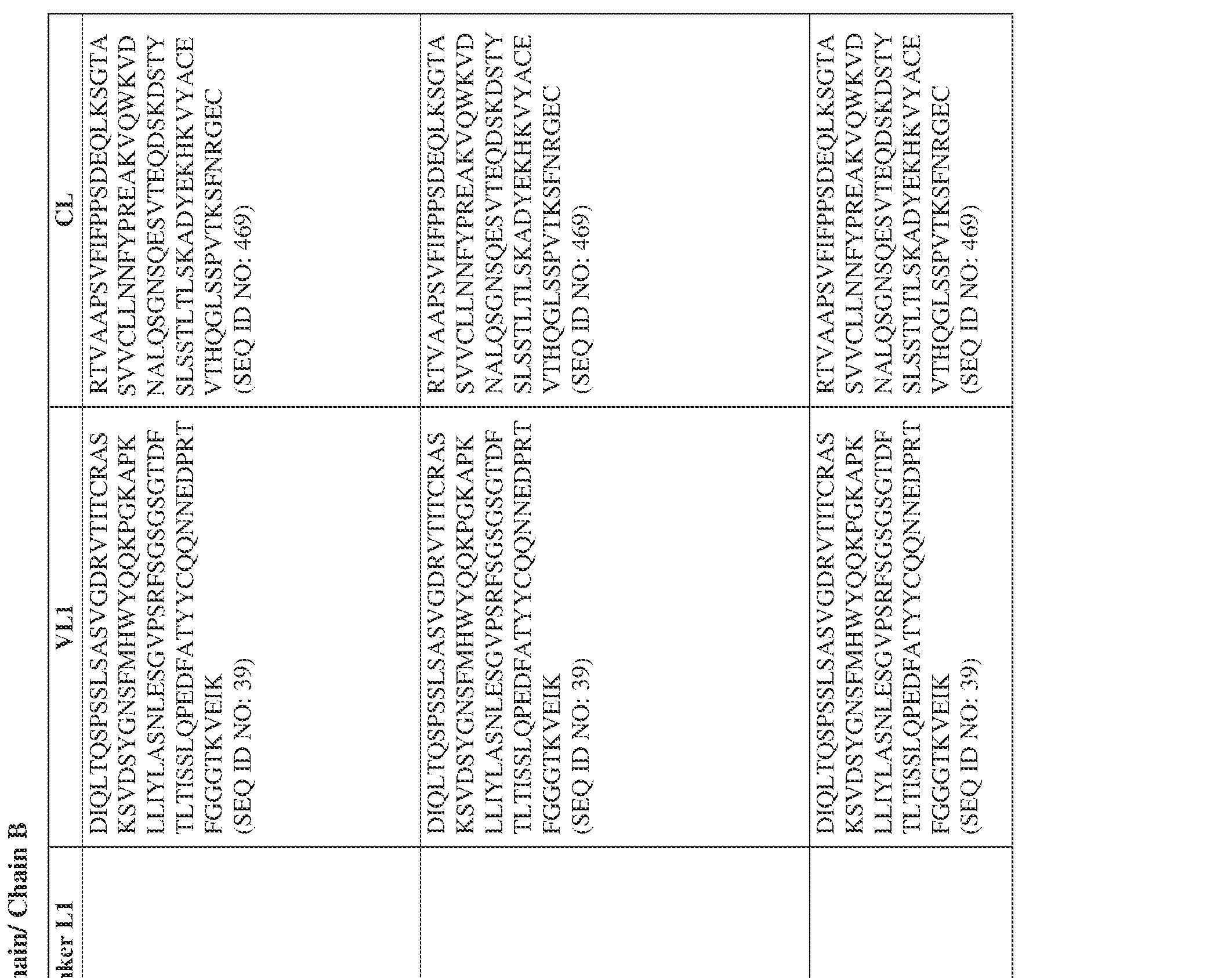
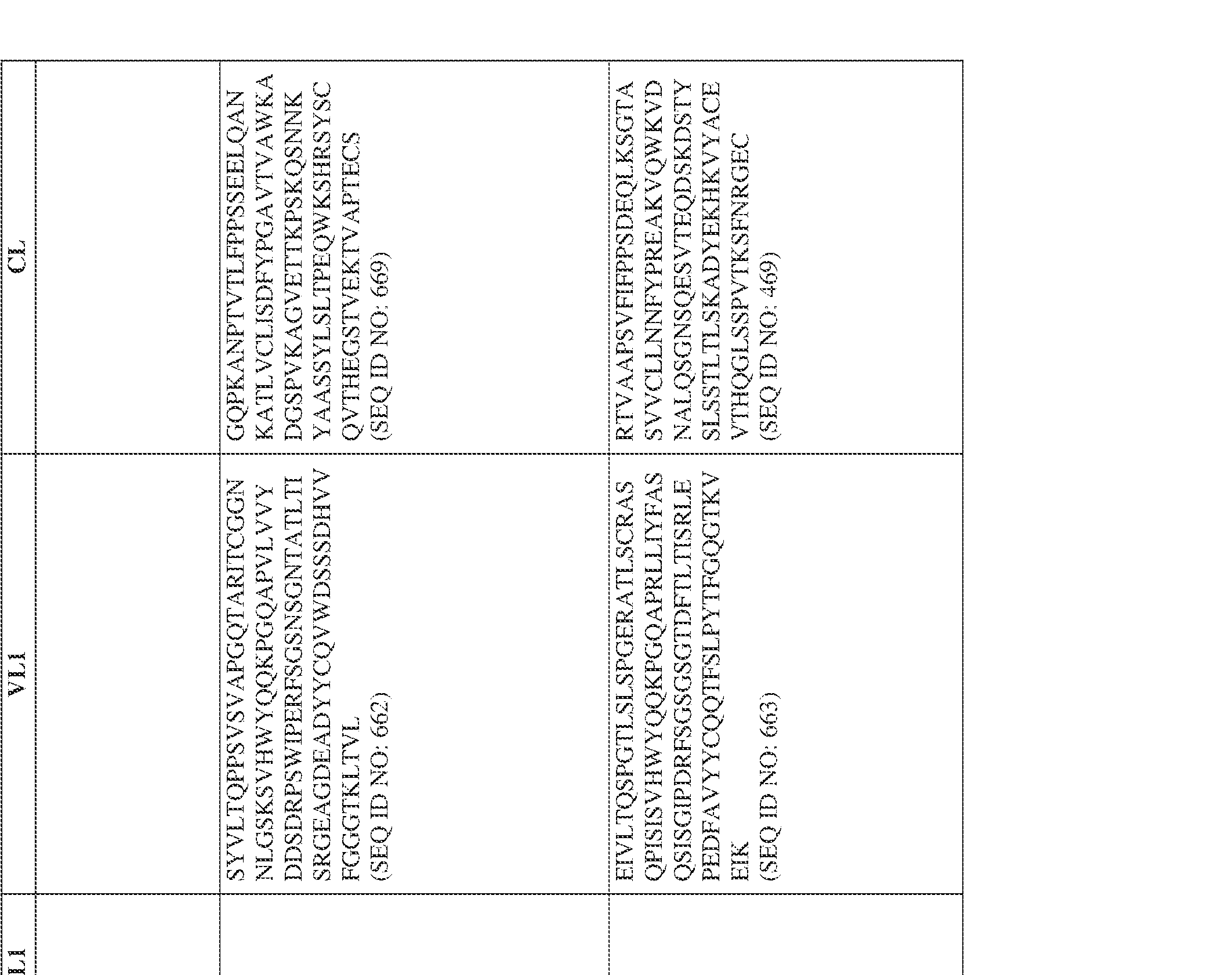
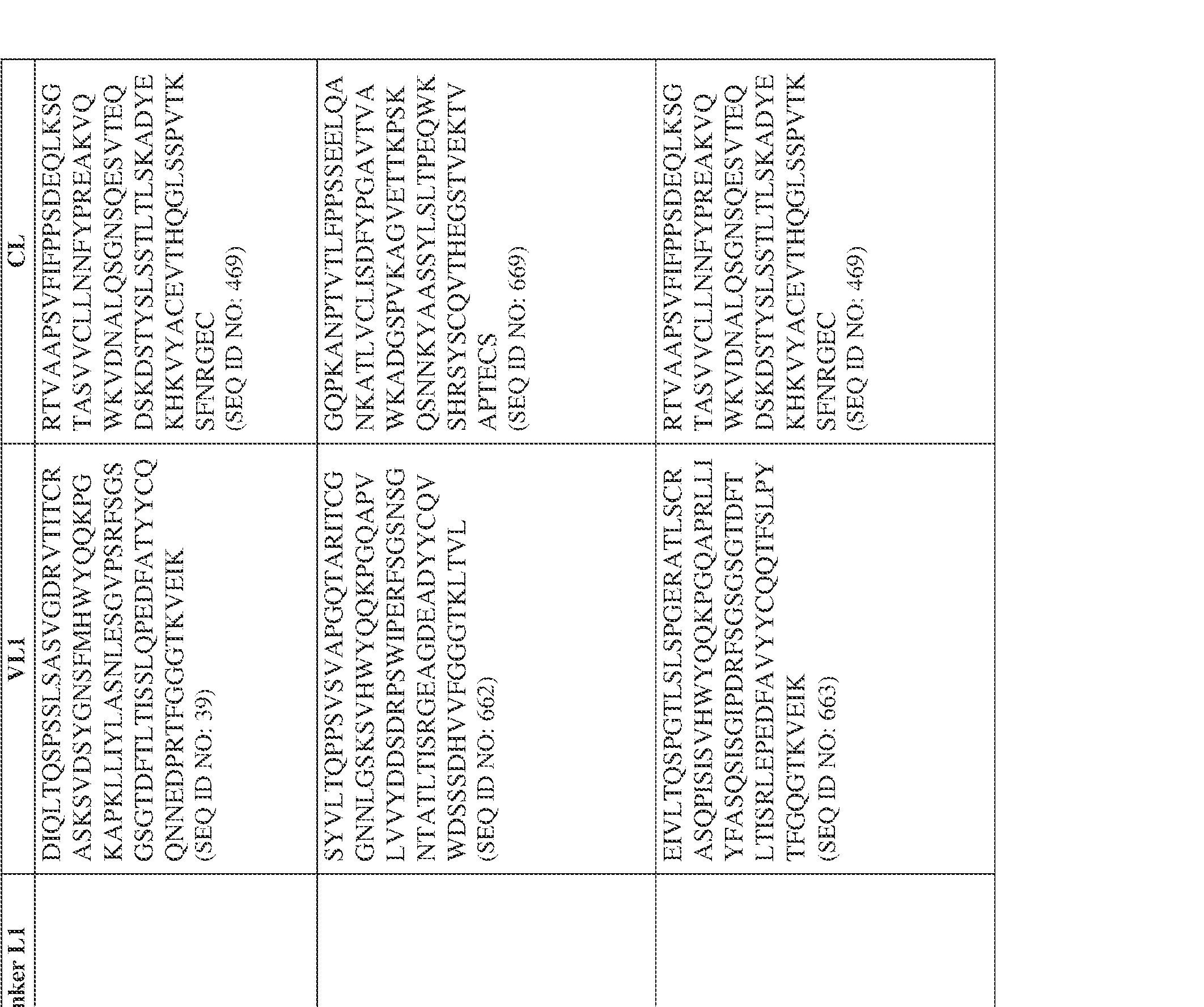
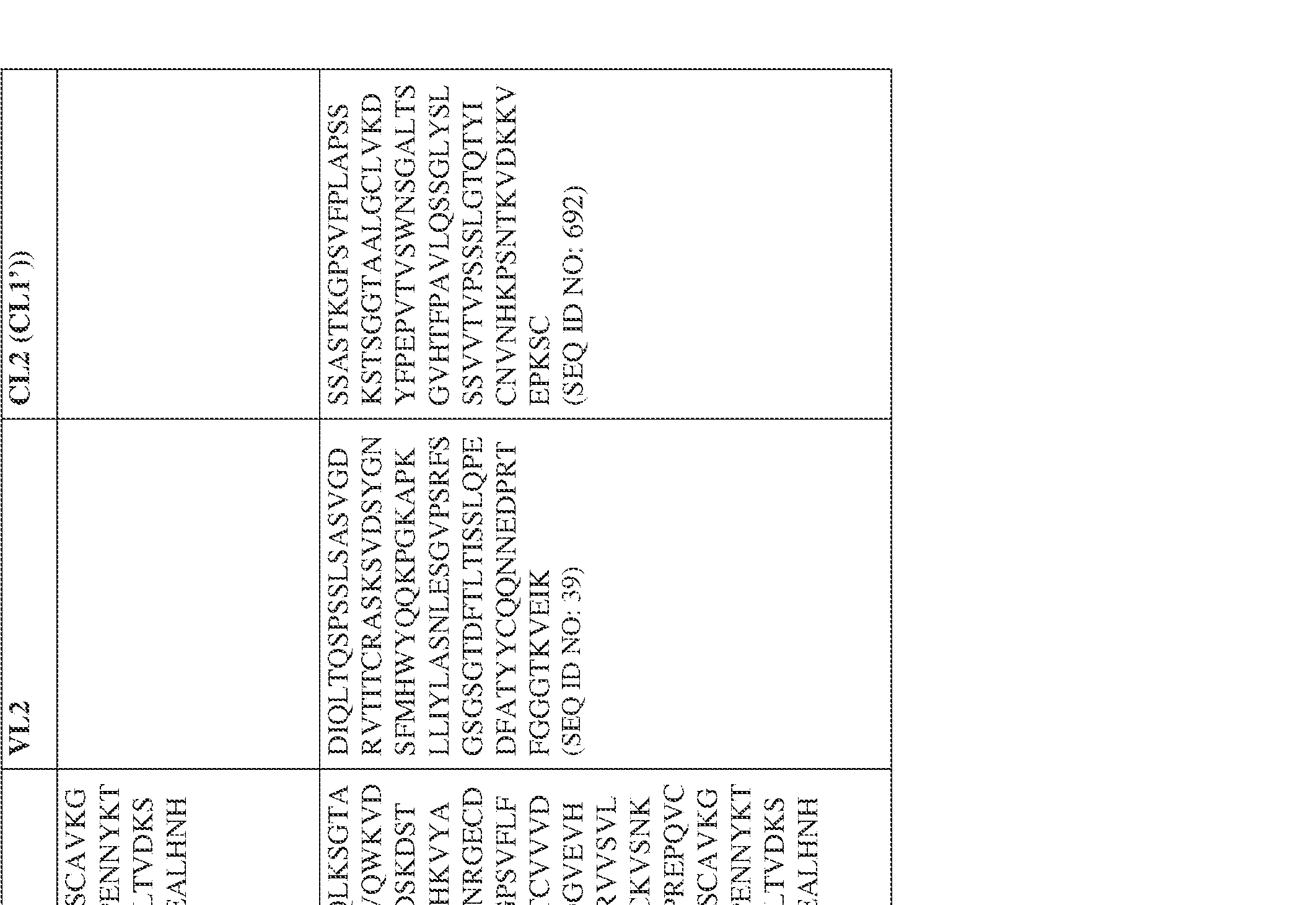
- the bispecific antibody comprises a heavy chain/chain A from Table 11a or Table 11b, a light chain/chain B from Table 12a or Table 12b, and, optionally, an Fc sequence from Table 13 (SEQ ID NOs: 425-468, 484-539, 670-680, 689- 691, and 693-804).
- the Fc region comprises one or more amino acid substitutions, wherein the one or more substitutions result in a change in antibody half-life, ADCC activity, ADCP activity, or CDC activity as compared to an otherwise equivalent antibody comprising an Fc without the one or more substitutions.
- the change is (a) an increase in antibody half-life and (b) a decrease in ADCC activity, ADCP activity, or CDC activity as compared to an otherwise equivalent antibody comprising an Fc without the one or more substitutions.
- the one or more amino acid substitutions results in increased antibody half-life compared to an antibody comprising a wild-type Fc region.
- IPTS/128623945.3 4 Attorney Docket No. PRG-013WO [0028]
- any of the isolated bispecific antibody described herein are for use in the treatment of an inflammatory disorder or disease.
- an isolated polynucleotide or set of polynucleotides encoding the bispecific antibody of any of the above claims, a VH thereof, a VL thereof, a light chain thereof, a heavy chain thereof, or an antigen-binding portion thereof, and optionally, wherein the polynucleotide or set of polynucleotides comprises cDNA.
- vector or set of vectors comprising the polynucleotide or set of polynucleotides described herein.
- a host cell comprising the polynucleotide or set of polynucleotides or the vector or set of vectors described herein.
- a method of producing an antibody comprising expressing the bispecific antibody with the host cell described herein and isolating the expressed bispecific antibody.
- a pharmaceutical composition comprising the bispecific antibody of any one of the bispecific antibodies described herein and a pharmaceutically acceptable excipient.
- a kit comprising the bispecific antibody of any one of the bispecific antibodies described herein or a pharmaceutical composition described herein and instructions for use.
- a method for treating an inflammatory disorder or disease in a mammalian subject in need thereof comprising administering to the mammalian subject a therapeutically effective amount the bispecific antibody of any one of the bispecific antibodies described herein or a pharmaceutical composition described herein.
- the isolated bispecific antibody is used in the treatment of an inflammatory disorder or disease.
- the isolated bispecific antibody is used in the treatment of atopic dermatitis.
- the treatment reduces disease severity in a subject and wherein disease severity is assessed by an Atopic Dermatitis Disease Severity Outcome Measure.
- the isolated bispecific antibody is used in the treatment of asthma.
- the isolated bispecific antibody is used in the treatment of idiopathic pulmonary fibrosis. In certain embodiments, the isolated bispecific antibody is used in the treatment of alopecia areata. In certain embodiments, the isolated bispecific antibody is used in the treatment of chronic sinusitis with nasal polyps. In certain embodiments, the isolated bispecific antibody is used in the treatment of Chronic IPTS/128623945.3 5 Attorney Docket No. PRG-013WO Rhinosinusitis without Nasal Polyps (CRSsNP). In certain embodiments, the isolated bispecific antibody is used in the treatment of eosinophilic esophagitis (EoE).
- EoE eosinophilic esophagitis
- the isolated bispecific antibody is used in the treatment of an Eosinophilic gastrointestinal disorder or disease (ENID) selected from the group consisting of Eosinophilic Gastritis (EoG), Eosinophilic Enteritis (EoN), Eosinophilic Colitis (EoC), and Eosinophilic Gastroenteritis (EGE).
- Eosinophilic gastrointestinal disorder or disease selected from the group consisting of Eosinophilic Gastritis (EoG), Eosinophilic Enteritis (EoN), Eosinophilic Colitis (EoC), and Eosinophilic Gastroenteritis (EGE).
- Eosinophilic Gastritis EoG
- EoN Eosinophilic Enteritis
- EoC Eosinophilic Colitis
- EGE Eosinophilic Gastroenteritis
- the isolated bispecific antibody is used in the treatment of Churg-Strauss syndrome/Eosinophilic gran
- the isolated bispecific antibody is used in the treatment of Chronic Spontaneous Urticaria (CSU). In certain embodiments, the isolated bispecific antibody is used in the treatment of Chronic Pruritis of Unknown Origin (CPUO). In certain embodiments, the isolated bispecific antibody is used in the treatment of Bullous Pemphigoid (BP). In certain embodiments, the isolated bispecific antibody is used in the treatment of Cold Inducible Urticaria (ColdU). In certain embodiments, the isolated bispecific antibody is used in the treatment of Allergic Fungal Rhinosinusitis (AFRS). In certain embodiments, the isolated bispecific antibody is used in the treatment of Allergic Bronchopulmonary Aspergillosis (ABPA).
- the isolated bispecific antibody is used in the treatment of Chronic Obstructive Pulmonary Disease (COPD).
- COPD Chronic Obstructive Pulmonary Disease
- the isolated bispecific antibody is used in the treatment of inflammatory bowel disease, such as Crohn disease or ulcerative colitis.
- the isolated bispecific antibody is used in the treatment of psoriasis.
- the isolated bispecific antibody is used in the treatment of lupus.
- the isolated bispecific antibody is used in the treatment of rheumatoid arthritis.
- described herein is an isolated polynucleotide or set of polynucleotides encoding an antibody described herein, a VH thereof, a VL thereof, a light chain thereof, a heavy chain thereof, or an antigen-binding portion thereof, and optionally, wherein the polynucleotide or set of polynucleotides comprises cDNA.
- described herein is a vector or set of vectors comprising the polynucleotide or set of polynucleotides.
- described herein is a host cell comprising the polynucleotide or set of polynucleotides or the vector or set of vectors.
- described herein is a method of producing an antibody, the method comprising expressing the antibody with the host cell described herein and isolating the expressed antibody. IPTS/128623945.3 6 Attorney Docket No. PRG-013WO [0039] In certain aspects, described herein is a pharmaceutical composition comprising a bispecific antibody described herein and a pharmaceutically acceptable excipient. [0040] In certain aspects, described herein is a kit comprising a bispecific antibody described herein or a pharmaceutical composition described herein and instructions for use.
- a method for treating an inflammatory disorder or disease in a mammalian subject in need thereof comprising administering to the mammalian subject a therapeutically effective amount a bispecific antibody described herein or a pharmaceutical composition described herein.
- the inflammatory disorder or disease is atopic dermatitis.
- the inflammatory disorder or disease is asthma.
- the inflammatory disorder or disease is idiopathic pulmonary fibrosis.
- the inflammatory disorder or disease is alopecia areata.
- the inflammatory disorder or disease is chronic sinusitis with nasal polyps.
- the inflammatory disorder or disease is Chronic Rhinosinusitis without Nasal Polyps (CRSsNP).
- the inflammatory disorder or disease is eosinophilic esophagitis (EoE).
- the inflammatory disorder or disease is an Eosinophilic gastrointestinal disorder or disease (ENID) selected from the group consisting of Eosinophilic Gastritis (EoG), Eosinophilic enteritis (EoN), Eosinophilic colitis (EoC), and Eosinophilic Gastroenteritis (EGE).
- EoG Eosinophilic Gastritis
- EoN Eosinophilic enteritis
- EoC Eosinophilic colitis
- EGE Eosinophilic Gastroenteritis
- the inflammatory disorder or disease is Churg-Strauss syndrome/Eosinophilic granulomatosis with polyangiitis (EGPA).
- the inflammatory disorder or disease is Prurigo Nodularis (PN).
- the inflammatory disorder or disease is Chronic Spontaneous Urticaria (CSU).
- the inflammatory disorder or disease is Chronic Pruritis of Unknown Origin (CPUO).
- the inflammatory disorder or disease is Bullous Pemphigoid (BP).
- the inflammatory disorder or disease is Cold Inducible Urticaria (ColdU).
- the inflammatory disorder or disease is Allergic Fungal Rhinosinusitis (AFRS).
- the inflammatory disorder or disease is Allergic Bronchopulmonary Aspergillosis (ABPA). In certain embodiments, the inflammatory disorder or disease is Chronic Obstructive Pulmonary Disease (COPD). In certain embodiments, the inflammatory disorder or disease is inflammatory bowel disease, such as Crohn disease or ulcerative colitis. In certain embodiments, the inflammatory disorder or disease is psoriasis. In certain embodiments, the inflammatory disorder or disease is lupus. In certain embodiments, the inflammatory disorder or disease is rheumatoid arthritis. IPTS/128623945.3 7 Attorney Docket No.
- described herein is a method for treating a pathology associated with elevated levels of IL-13 in a mammalian subject in need thereof, the method comprising administering to the mammalian subject a therapeutically effective amount of a bispecific antibody described herein or a pharmaceutical composition described herein.
- described herein is a method of reducing biological activity of IL-13 in a mammalian subject in need thereof, the method comprising administering to the mammalian subject a therapeutically effective amount a bispecific antibody described herein or a pharmaceutical composition described herein.
- described herein is a method of inhibiting the TH2 type allergic response in a mammalian subject in need thereof, the method comprising administering to the mammalian subject a therapeutically effective amount of a bispecific antibody described herein or a pharmaceutical composition described herein.
- described herein is a method of reducing levels of Thymus and Activation Regulated Chemokine (TARC)/CCL17 in a mammalian subject in need thereof, the method comprising administering to the mammalian subject a therapeutically effective amount of a bispecific antibody described herein or a pharmaceutical composition described herein.
- TARC Thymus and Activation Regulated Chemokine
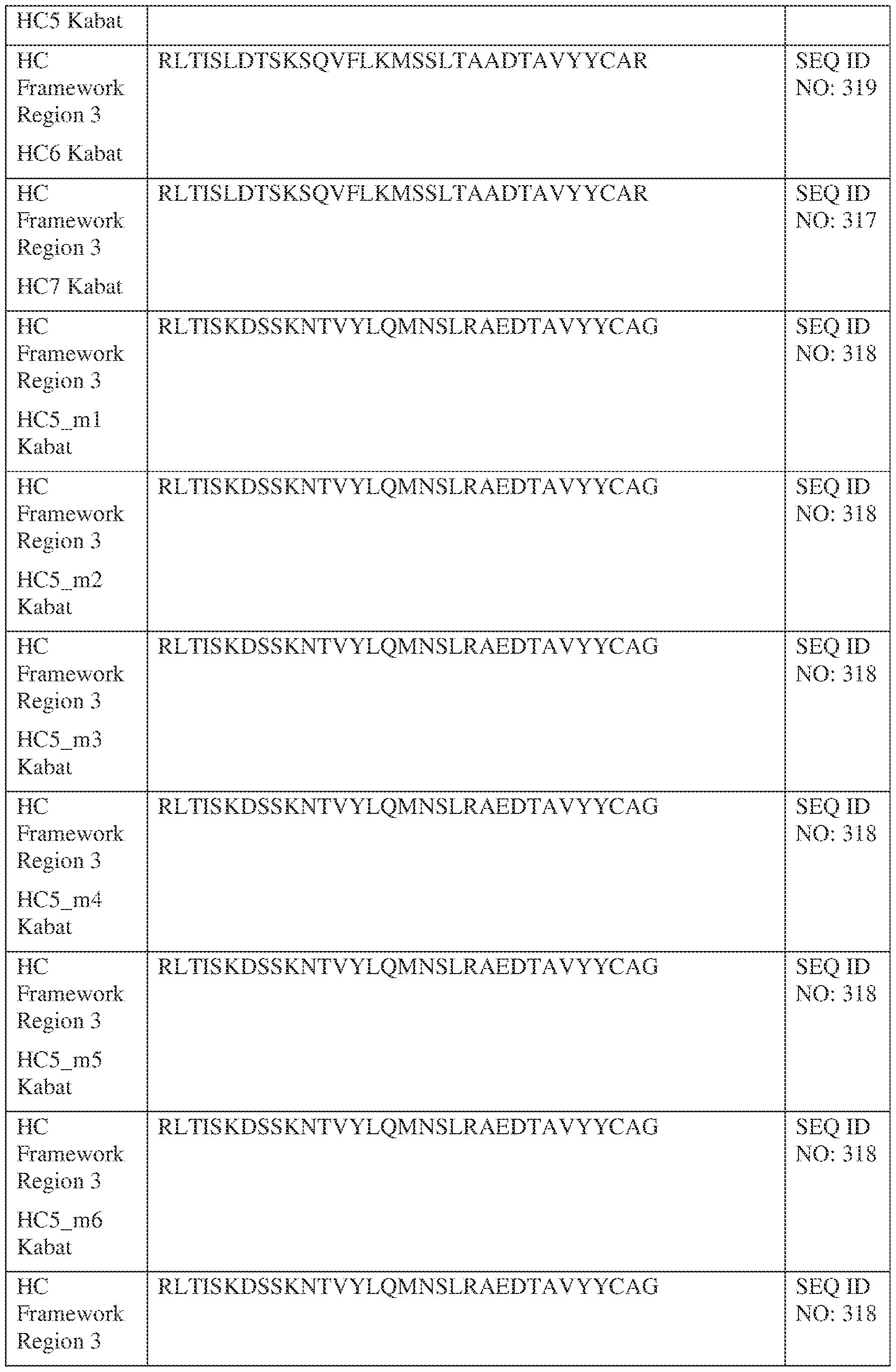
- FIG. 1 depicts an exemplary IgG-scFv bispecific antibody format.
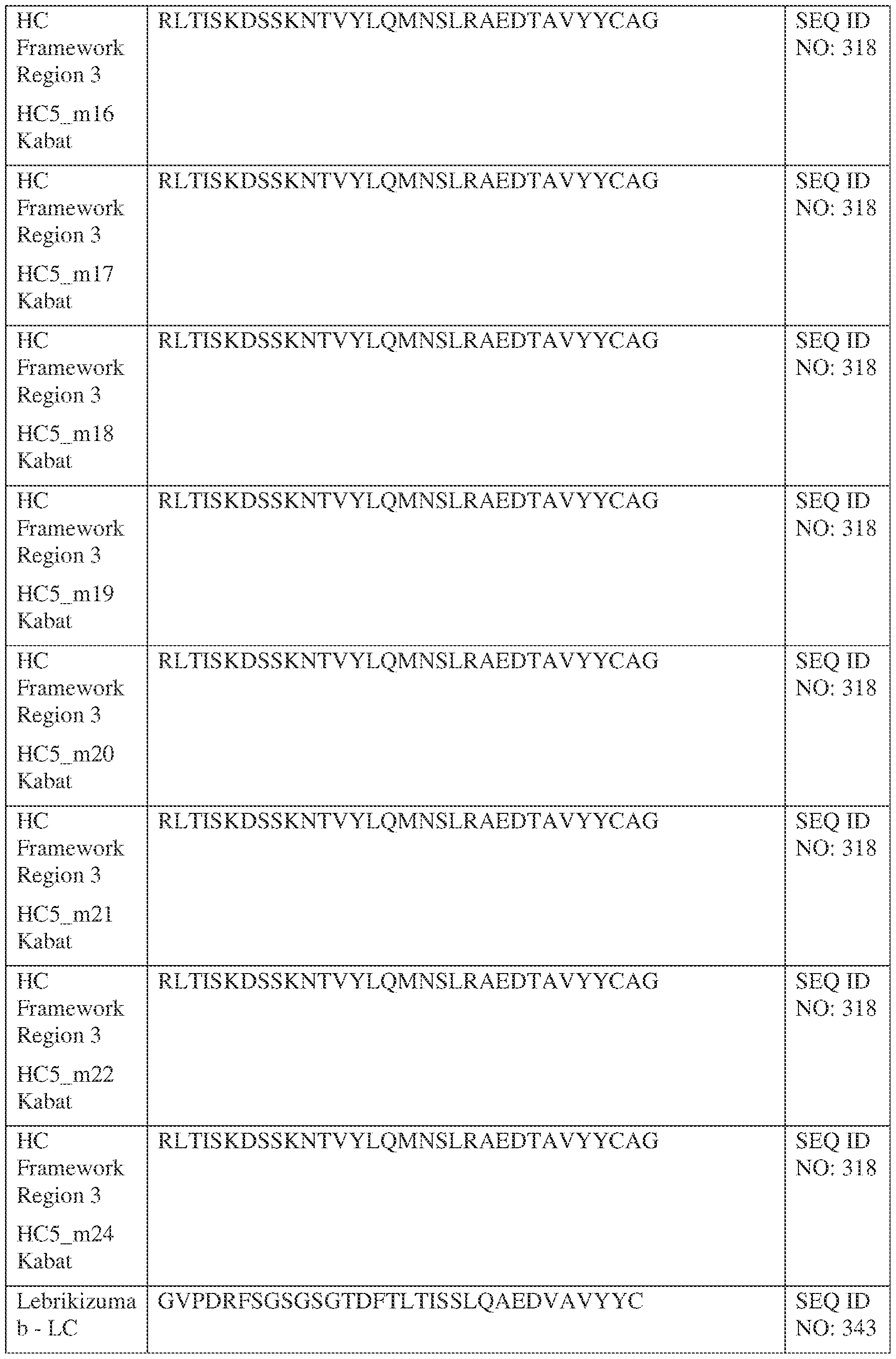
- Figure 2 depicts an exemplary DVD-Ig bispecific antibody format.
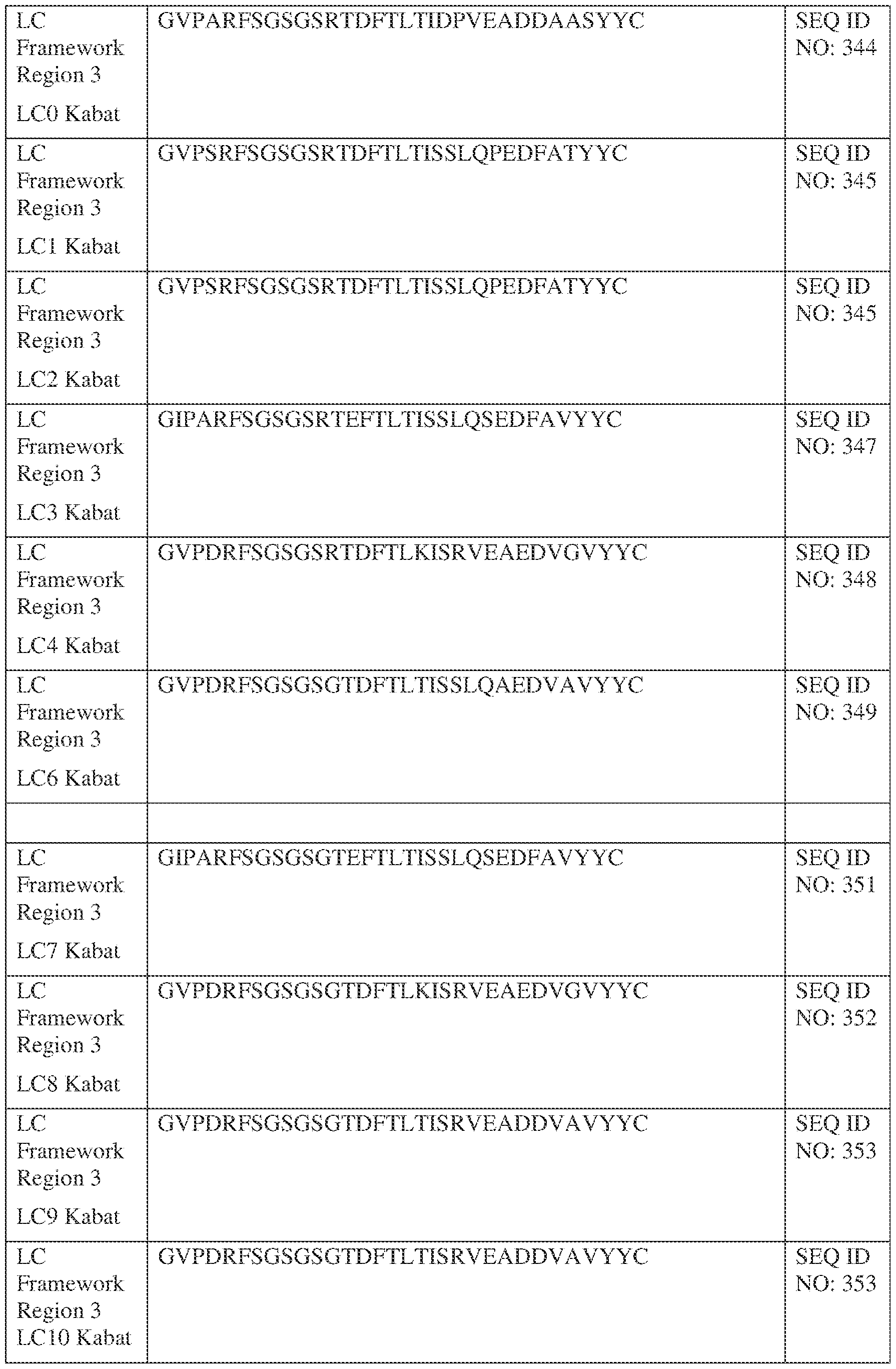
- Figure 3 depicts an exemplary CrossMab bispecific antibody format.
- the instant disclosure relates, in part, to antibodies (e.g., bispecific antibodies) that bind (1) IL-13 and (2) either TSLP or a TSLP receptor (TSLPR).
- TSLPR TSLP receptor
- all terms of art, notations and other scientific terminology used herein are intended to have the meanings commonly understood by those of skill in the art. In some cases, terms with commonly understood meanings are defined herein for clarity and/or for ready reference, and the inclusion of such definitions herein should not necessarily be construed to represent a difference over what is generally understood in the art.
- compositions described herein include “comprising,” “consisting,” and “consisting essentially of” aspects and embodiments. [0055] For all compositions described herein, and all methods using a composition described herein, the compositions can either comprise the listed components or steps, or can “consist essentially of” the listed components or steps.
- composition when a composition is described as “consisting essentially of” the listed components, the composition contains the components listed, and may contain other components which do not substantially affect the condition being treated, but do not contain any other components which substantially affect the condition being treated other than those components expressly listed; or, if the composition does contain extra components other than those listed which substantially affect the condition being treated, the composition does not contain a sufficient concentration or amount of the extra components to substantially affect the condition being treated.
- a method is described as “consisting essentially of” the listed steps, the method contains the steps listed, and may contain other steps that do not substantially affect the condition being treated, but the method does not contain any other steps which substantially affect the condition being treated other than those steps expressly listed.
- composition when a composition is described as “consisting essentially of” a component, the composition may additionally contain any amount of pharmaceutically acceptable carriers, vehicles, or diluents and other such components which do not substantially affect the condition being treated.
- IPTS/128623945.3 9 Attorney Docket No. PRG-013WO [0056]
- vector refers to a nucleic acid molecule capable of propagating another nucleic acid to which it is linked.
- the term includes the vector as a self- replicating nucleic acid structure as well as the vector incorporated into the genome of a host cell into which it has been introduced. Certain vectors are capable of directing the expression of nucleic acids to which they are operatively linked.
- host cell Such vectors are referred to herein as “expression vectors.”
- host cell refers to cells into which an exogenous nucleic acid has been introduced, and the progeny of such cells.
- Host cells include “transformants” (or “transformed cells”) and “transfectants” (or “transfected cells”), which each include the primary transformed or transfected cell and progeny derived therefrom.
- Such progeny may not be completely identical in nucleic acid content to a parent cell, and may contain mutations.
- a “recombinant host cell” or “host cell” refers to a cell that includes an exogenous polynucleotide, regardless of the method used for insertion, for example, direct uptake, transduction, f-mating, or other methods known in the art to create recombinant host cells.
- the term “eukaryote” refers to organisms belonging to the phylogenetic domain Eucarya such as animals (including but not limited to, mammals, insects, reptiles, birds, etc.), ciliates, plants (including but not limited to, monocots, dicots, algae, etc.), fungi, yeasts, flagellates, microsporidia, protists, etc.
- prokaryote refers to prokaryotic organisms.
- a non-eukaryotic organism can belong to the Eubacteria (including but not limited to, Escherichia coli, Thermus thermophilus, Bacillus stearothermophilus, Pseudomonas fluorescens, Pseudomonas aeruginosa, Pseudomonas putida, etc.) phylogenetic domain, or the Archaea (including but not limited to, Methanococcus jannaschii, Methanobacterium thermoautotrophicum, Halobacterium such as Haloferax volcanii and Halobacterium species NRC-1, Archaeoglobus fulgidus, Pyrococcus furiosus, Pyrococcus horikoshii, Aeuropyrum pernix, etc.) phylogenetic domain.
- Eubacteria including but not limited to, Escherichia coli, Thermus thermophilus,
- an “effective amount” or “therapeutically effective amount” as used herein refers to an amount of therapeutic compound, such as an anti-IL-13 antibody, administered to an individual, either as a single dose or as part of a series of doses, which is effective to produce or contribute to a desired therapeutic effect, either alone or in combination with another therapeutic modality. Examples of a desired therapeutic effect is enhancing an immune response, slowing or delaying tumor development; stabilization of disease; amelioration of one or more symptoms. An effective amount may be given in one or more dosages. IPTS/128623945.3 10 Attorney Docket No.
- PRG-013WO refers to clinical intervention in an attempt to alter the natural course of a disease or condition in a subject in need thereof. Treatment can be performed during the course of clinical pathology. Desirable effects of treatment include preventing recurrence of disease, alleviation of symptoms, diminishment of any direct or indirect pathological consequences of the disease, preventing metastasis, decreasing the rate of disease progression, amelioration or palliation of the disease state, and remission or improved prognosis.
- the term “sufficient amount” means an amount sufficient to produce a desired effect, e.g., an amount sufficient to modulate an immune response in a subject.
- the term “subject” or “individual” means a mammalian subject. Exemplary subjects include humans, monkeys, dogs, cats, mice, rats, cows, horses, camels, goats, rabbits, and sheep. In certain embodiments, the subject is a human. In some embodiments the subject has a disease or condition that can be treated with an antibody provided herein. In some embodiments, the disease or condition is a cancer. In some embodiments, the disease or condition is a viral infection. [0064] The term “in vitro” refers to processes that occur in a living cell growing separate from a living organism, e.g., growing in tissue culture.
- the term “in vivo” refers to processes that occur in a living organism.
- the term “package insert” is used to refer to instructions customarily included in commercial packages of therapeutic or diagnostic products (e.g., kits) that contain information about the indications, usage, dosage, administration, combination therapy, contraindications and/or warnings concerning the use of such therapeutic or diagnostic products.
- the term “pharmaceutical composition” refers to a preparation which is in such form as to permit the biological activity of an active ingredient contained therein to be effective in treating a subject, and which contains no additional components which are unacceptably toxic to the subject in the amounts provided in the pharmaceutical composition.
- co-administration include the administration of two or more therapeutic agents either simultaneously, concurrently or sequentially within no specific time limits.
- the agents are present in the cell or in the subject's body at the same time or exert their biological or therapeutic effect at the same time.
- the therapeutic agents are in the same composition or unit dosage form.
- the therapeutic agents are in separate IPTS/128623945.3 11 Attorney Docket No. PRG-013WO compositions or unit dosage forms.
- a first agent can be administered prior to the administration of a second therapeutic agent.
- modulate and “modulation” refer to reducing or inhibiting or, alternatively, activating or increasing, a recited variable.
- increase and “activate” refer to an increase of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 75%, 80%, 85%, 90%, 95%, 100%, 2-fold, 3-fold, 4-fold, 5-fold, 10-fold, 20-fold, 50-fold, 100-fold, or greater in a recited variable.
- the terms “reduce” and “inhibit” refer to a decrease of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 75%, 80%, 85%, 90%, 95%, 2-fold, 3-fold, 4-fold, 5-fold, 10-fold, 20-fold, 50-fold, 100-fold, or greater in a recited variable.
- the term “about” indicates and encompasses an indicated value and a range above and below that value. In certain embodiments, the term “about” indicates the designated value ⁇ 10%, ⁇ 5%, or ⁇ 1%. In certain embodiments, where applicable, the term “about” indicates the designated value(s) ⁇ one standard deviation of that value(s).
- the term “agonize” refers to the activation of receptor signaling to induce a biological response associated with activation of the receptor.
- An “agonist” is an entity that binds to and agonizes a receptor.
- the term “antagonize” refers to the inhibition of receptor signaling to inhibit a biological response associated with activation of the receptor.
- An “antagonist” is an entity that binds to and antagonizes a receptor.
- amino acid refers to the twenty common naturally occurring amino acids.
- Naturally occurring amino acids include alanine (Ala; A), arginine (Arg; R), asparagine (Asn; N), aspartic acid (Asp; D), cysteine (Cys; C); glutamic acid (Glu; E), glutamine (Gln; Q), Glycine (Gly; G); histidine (His; H), isoleucine (Ile; I), leucine (Leu; L), lysine (Lys; K), methionine (Met; M), phenylalanine (Phe; F), proline (Pro; P), serine (Ser; S), threonine (Thr; T), tryptophan (Trp; W), tyrosine (Tyr; Y), and valine (Val; V).
- Naturally occurring amino acids include alanine (Ala; A), arginine (Arg; R), asparagine (Asn; N), as
- affinity refers to the strength of the sum total of non-covalent interactions between a single binding site of a molecule (e.g., an antibody) and its binding partner (e.g., an antigen or epitope).
- affinity IPTS/128623945.3 12 Attorney Docket No. PRG-013WO refers to intrinsic binding affinity, which reflects a 1:1 interaction between members of a binding pair (e.g., antibody and antigen or epitope).
- kd (sec-1), as used herein, refers to the dissociation rate constant of a particular antibody-antigen interaction. This value is also referred to as the koff value.
- ka M-1 ⁇ sec-1
- KD Kd/ka.
- affinity of an antibody is described in terms of the KD for an interaction between such antibody and its antigen. For clarity, as known in the art, a smaller KD value indicates a higher affinity interaction, while a larger KD value indicates a lower affinity interaction.
- KA (M-1), as used herein, refers to the association equilibrium constant of a particular antibody-antigen interaction.
- KA ka/kd.
- antibody is used herein in its broadest sense and includes certain types of immunoglobulin molecules comprising one or more antigen-binding domains that specifically bind to an antigen or epitope. An antibody specifically includes intact antibodies (e.g., intact immunoglobulins), antibody fragments, and multi-specific antibodies.
- a “anti-IL-13 antibody,” “IL-13 antibody,” or “IL-13 specific antibody” is an antibody, as provided herein, which specifically binds to the antigen IL-13.
- epitope means a portion of an antigen that specifically binds to an antibody.
- epitope means a portion of an antigen that specifically binds to an antibody.
- hypervariable region or “HVR,” as used herein, refers to each of the regions of an antibody variable domain which are hypervariable in sequence and/or form structurally defined loops (“hypervariable loops”).
- antigen-binding domain means the portion of an antibody that is capable of specifically binding to an antigen or epitope.
- chimeric antibody refers to an antibody in which a portion of the heavy and/or light chain is derived from a particular source or species, while the remainder of the heavy and/or light chain is derived from a different source or species.
- human antibody refers to an antibody which possesses an amino acid sequence corresponding to that of an antibody produced by a human or a human cell, or derived from a non-human source that utilizes a human antibody repertoire or human antibody-encoding sequences (e.g., obtained from human sources or designed de novo). Human antibodies specifically exclude humanized antibodies. IPTS/128623945.3 13 Attorney Docket No.
- humanized antibody refers to a protein having a sequence that differs from the sequence of an antibody derived from a non-human species by one or more amino acid substitutions, deletions, and/or additions, such that the humanized antibody is less likely to induce an immune response, and/or induces a less severe immune response, as compared to the non-human species antibody, when it is administered to a human subject.
- multispecific antibody refers to an antibody that comprises two or more different antigen-binding domains that collectively specifically bind two or more different epitopes.
- a “bispecific antibody” is an antibody that comprises two different antigen binding domains that each bind different epitopes.
- a “monospecific antibody” is an antibody that comprises one or more binding sites that specifically bind to a single epitope.
- An example of a monospecific antibody is a naturally occurring IgG molecule which, while divalent (i.e., having two antigen-binding domains), recognizes the same epitope at each of the two antigen-binding domains. The binding specificity may be present in any suitable valency.
- the term “monoclonal antibody” refers to an antibody from a population of substantially homogeneous antibodies. A population of substantially homogeneous antibodies comprises antibodies that are substantially similar and that bind the same epitope(s), except for variants that may normally arise during production of the monoclonal antibody.
- a monoclonal antibody is typically obtained by a process that includes the selection of a single antibody from a plurality of antibodies.
- the selection process can be the selection of a unique clone from a plurality of clones, such as a pool of hybridoma clones, phage clones, yeast clones, bacterial clones, or other recombinant DNA clones.
- the selected antibody can be further altered, for example, to improve affinity for the target (“affinity maturation”), to humanize the antibody, to improve its production in cell culture, and/or to reduce its immunogenicity in a subject.
- the term “single-chain” refers to a molecule comprising amino acid monomers linearly linked by peptide bonds.
- the C-terminus of the Fab light chain is connected to the N-terminus of the Fab heavy chain in the single-chain Fab molecule.
- an scFv has a variable domain of light chain (VL) connected from its C-terminus to the N-terminal end of a variable domain of heavy chain (VH) by a polypeptide chain.
- VL variable domain of light chain
- VH variable domain of heavy chain
- the scFv comprises of polypeptide chain where in the C-terminal end of the VH is connected to the N-terminal end of VL by a polypeptide chain.
- the “Fab fragment” (also referred to as fragment antigen-binding) contains the constant domain (CL) of the light chain and the first constant domain (CH1) of the heavy chain along with the variable domains VL and VH on the light and heavy chains respectively.
- the variable domains comprise the complementarity determining loops (CDR, also referred to as hypervariable region) that are involved in antigen-binding.
- CDR complementarity determining loops
- Fab ⁇ fragments differ from Fab fragments by the addition of a few residues at the carboxy terminus of the heavy chain CH1 domain including one or more cysteines from the antibody hinge region.
- F(ab’)2 fragments contain two Fab’ fragments joined, near the hinge region, by disulfide bonds. F(ab’)2 fragments may be generated, for example, by recombinant methods or by pepsin digestion of an intact antibody. The F(ab’) fragments can be dissociated, for example, by treatment with ß-mercaptoethanol. [0098] “Fv” fragments comprise a non-covalently-linked dimer of one heavy chain variable domain and one light chain variable domain. [0099] “Single-chain Fv” or “sFv” or “scFv” includes the VH and VL domains of an antibody, wherein these domains are present in a single polypeptide chain.
- the Fv polypeptide further comprises a polypeptide linker between the VH and VL domains which enables the scFv to form the desired structure for antigen-binding.
- a polypeptide linker between the VH and VL domains which enables the scFv to form the desired structure for antigen-binding.
- an Fc domain may be attached to the C-terminal of the scFv.
- the Fc domain may follow the VH or VL, depending on the orientation of the variable domains in the scFv (i.e., VH-VL or VL- VH ). Any suitable Fc domain known in the art or described herein may be used.
- the Fc domain comprises an IgG4 Fc domain.
- single domain antibody or “sdAb” refers to a molecule in which one variable domain of an antibody specifically binds to an antigen without the presence of the other variable domain. Single domain antibodies, and fragments thereof, are described in Arabi Ghahroudi et al.
- Single domain antibodies are also known as sdAbs or nanobodies. Sdabs are fairly stable and easy to express as fusion partner with the Fc chain of an antibody (Harmsen MM, IPTS/128623945.3 15 Attorney Docket No. PRG-013WO De Haard HJ (2007) “Properties, production, and applications of camelid single-domain antibody fragments” Appl. Microbiol Biotechnol.77(1): 13-22).
- full length antibody “intact antibody,” and “whole antibody” are used herein interchangeably to refer to an antibody having a structure substantially similar to a naturally occurring antibody structure and having heavy chains that comprise an Fc region.
- a “full length antibody” is an antibody that comprises two heavy chains and two light chains.
- antibody fragment refers to an antibody that comprises a portion of an intact antibody, such as the antigen-binding or variable region of an intact antibody.
- Antibody fragments include, for example, Fv fragments, Fab fragments, F(ab’)2 fragments, Fab’ fragments, scFv (sFv) fragments, and scFv-Fc fragments.
- Fc domain or “Fc region” herein is used to define a C-terminal region of an immunoglobulin heavy chain that contains at least a portion of the constant region. The term includes native sequence Fc regions and variant Fc regions.
- substantially purified refers to a construct described herein, or variant thereof that may be substantially or essentially free of components that normally accompany or interact with the protein as found in its naturally occurring environment, i.e.
- a native cell, or host cell in the case of recombinantly produced antibody that in certain embodiments, is substantially free of cellular material includes preparations of protein having less than about 30%, less than about 25%, less than about 20%, less than about 15%, less than about 10%, less than about 5%, less than about 4%, less than about 3%, less than about 2%, or less than about 1% (by dry weight) of contaminating protein.
- percent identity in the context of two or more nucleic acid or polypeptide sequences, refer to two or more sequences or subsequences that have a specified percentage of nucleotides or amino acid residues that are the same, when compared and aligned for maximum correspondence, as measured using one of the sequence comparison algorithms described below (e.g., using publicly available computer software such as BLAST, BLASTP, BLASTN, BLAST-2, ALIGN, MEGALIGN (DNASTAR), CLUSTALW, CLUSTAL OMEGA, or MUSCLE software or other algorithms available to persons of skill) or by visual inspection. Software for performing BLAST analyses (Altschul et al. (1990) J. Mol.
- Biol.215:403-410) is publicly available through the National Center for Biotechnology Information (ncbi.nlm.nih.gov). Those skilled in the art can determine appropriate parameters for aligning sequences, including any algorithms needed to achieve maximal alignment over the full length of the sequences being compared. Depending on the application, the percent IPTS/128623945.3 16 Attorney Docket No. PRG-013WO “identity” can exist over a region of the sequence being compared, e.g., over a functional domain, or, alternatively, exist over the full length of the two sequences to be compared. [00107] For sequence comparison, typically one sequence acts as a reference sequence to which test sequences are compared.
- test and reference sequences are input into a computer, subsequence coordinates are designated, if necessary, and sequence algorithm program parameters are designated.
- sequence comparison algorithm calculates the percent sequence identity for the test sequence(s) relative to the reference sequence, based on the designated program parameters.
- Optimal alignment of sequences for comparison can be conducted, e.g., by the local homology algorithm of Smith & Waterman (1981) Adv. Appl. Math.2:482, by the homology alignment algorithm of Needleman & Wunsch (1970) J. Mol. Biol.48:443, by the search for similarity method of Pearson & Lipman (1988) Proc. Nat’l. Acad. Sci.
- a range of 1 to 50 is understood to include any number, combination of numbers, or sub-range from the group consisting of 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, and 50.
- the recognized immunoglobulin (antibody) genes include the kappa, lambda, alpha, gamma, delta, epsilon, and mu constant region genes, as well as the myriad immunoglobulin variable region genes.
- Light chains are classified as either kappa or lambda.
- the “class” of an antibody or immunoglobulin refers to the type of constant domain or constant region possessed by its heavy chain.
- IgA immunoglobulin structural unit
- IgD immunoglobulin D
- IgE immunoglobulin E
- IgG immunoglobulin G
- IgM immunoglobulin M
- subclasses e.g., IgG1, IgG2, IgG3, IgG4, IgA1, and IgA2.
- the heavy chain constant domains IPTS/128623945.3 17 Attorney Docket No. PRG-013WO that correspond to the different classes of immunoglobulins are called ⁇ , ⁇ , ⁇ , ⁇ , and ⁇ , respectively.
- An exemplary immunoglobulin structural unit is composed of two pairs of polypeptide chains, each pair having one “light” (about 25 kD) and one “heavy” chain (about 50-70 kD).
- the N-terminal domain of each chain defines a variable region of about 100 to 110 or more amino acids primarily responsible for antigen recognition.
- VL variable light chain
- VH variable heavy chain
- the IgG1 heavy chain comprises of the VH, CH1, CH2, and CH3 domains respectively from the N- to C-terminus.
- the light chain comprises of the VL and CL domains from N- to C-terminus.
- the IgG1 heavy chain comprises a hinge between the CH1 and CH2 domains.
- the immunoglobulin constructs comprise at least one immunoglobulin domain from IgG, IgM, IgA, IgD, or IgE connected to a therapeutic polypeptide.
- the immunoglobulin domain found in an antibody provided herein is from or derived from an immunoglobulin based construct such as a diabody or a nanobody.
- the immunoglobulin constructs described herein comprise at least one immunoglobulin domain from a heavy chain antibody such as a camelid antibody.
- the immunoglobulin constructs provided herein comprise at least one immunoglobulin domain from a mammalian antibody such as a bovine antibody, a human antibody, a camelid antibody, a mouse antibody, or any chimeric antibody.
- the antibodies provided herein comprise a heavy chain.
- the heavy chain is an IgA.
- the heavy chain is an IgD. In one embodiment, the heavy chain is an IgE. In one embodiment, the heavy chain is an IgG. In one embodiment, the heavy chain is an IgM. In one embodiment, the heavy chain is an IgG1. In one embodiment, the heavy chain is an IgG2. In one embodiment, the heavy chain is an IgG3. In one embodiment, the heavy chain is an IgG4. In one embodiment, the heavy chain is an IgA1. In one embodiment, the heavy chain is an IgA2. [00114] In some embodiments, an antibody is an IgG1 antibody. In some embodiments, an antibody is an IgG3 antibody. In some embodiments, an antibody is an IgG2 antibody.
- an antibody is an IgG4 antibody.
- native four-chain antibodies comprise six hypervariable regions (HVRs); three in the VH (H1, H2, and H3), and three in the VL (L1, L2, and L3).
- HVRs generally comprise amino acid residues from the hypervariable loops and/or from the complementarity determining regions (CDRs), the latter being of highest sequence variability and/or involved in antigen recognition.
- CDRs complementarity determining regions
- CDRs complementarity determining regions
- HVRs are also referred to as CDRs, and these terms are used herein interchangeably in reference to portions of the variable region that form the antigen-binding regions. This particular region has been described by Kabat et al. (1983) U.S. Dept. of Health and Human Services, Sequences of Proteins of Immunological Interest and by Chothia et al. (1987) J Mol Biol 196:901-917, where the definitions include overlapping or subsets of amino acid residues when compared against each other. Nevertheless, application of either definition to refer to a CDR of an antibody or variants thereof is intended to be within the scope of the term as defined and used herein. The exact residue numbers which encompass a particular CDR will vary depending on the sequence and size of the CDR.
- the amino acid sequence boundaries of a CDR can be determined by one of skill in the art using any of a number of known numbering schemes, including those described by Kabat et al., supra (“Kabat” numbering scheme); Al-Lazikani et al. (1997) J. Mol. Biol., 273:927-948 (“Chothia” numbering scheme); MacCallum et al. (1996) J. Mol. Biol.262:732- 745 (“Contact” numbering scheme); Lefranc et al. (2003) Dev. Comp.
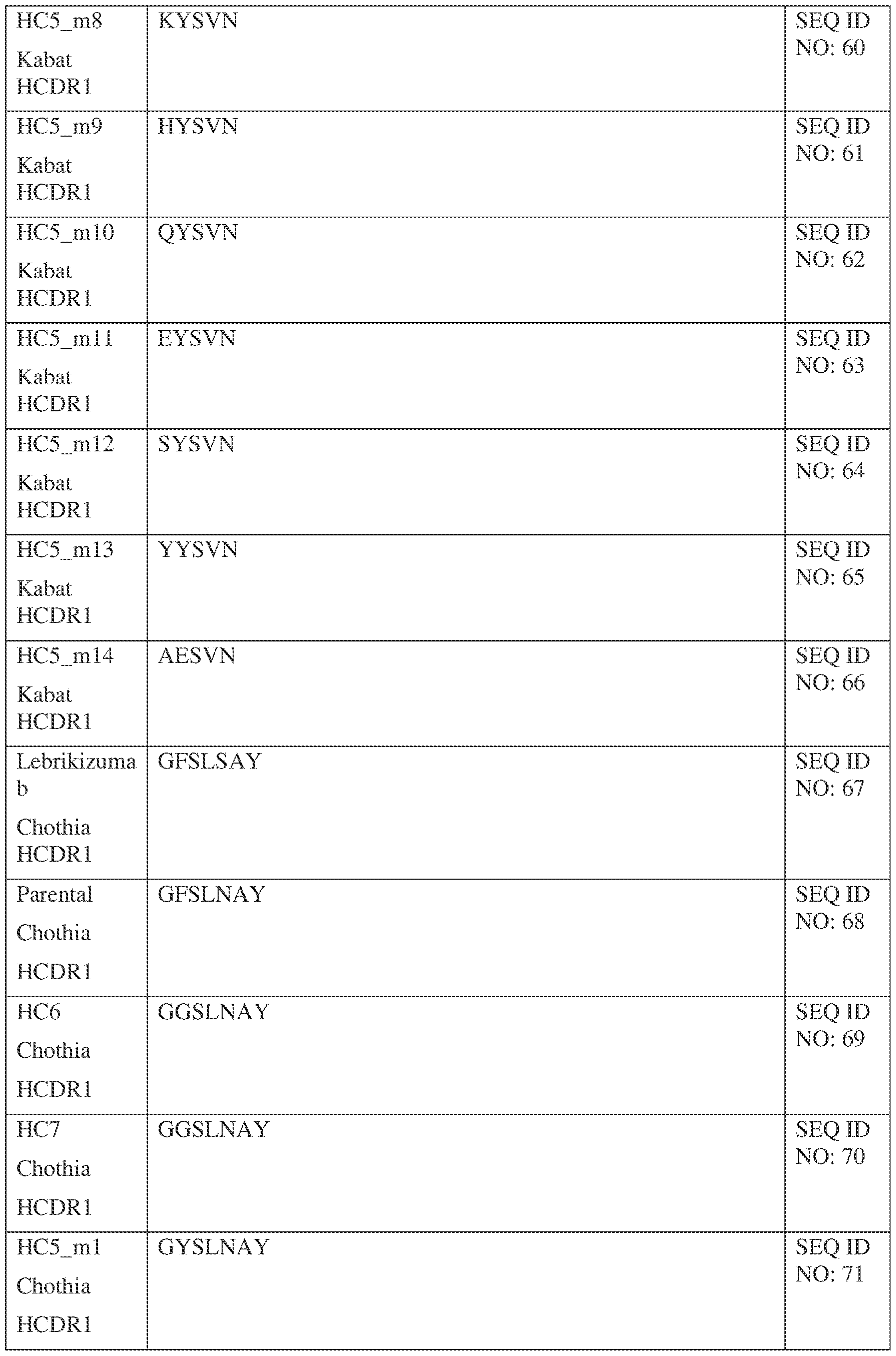
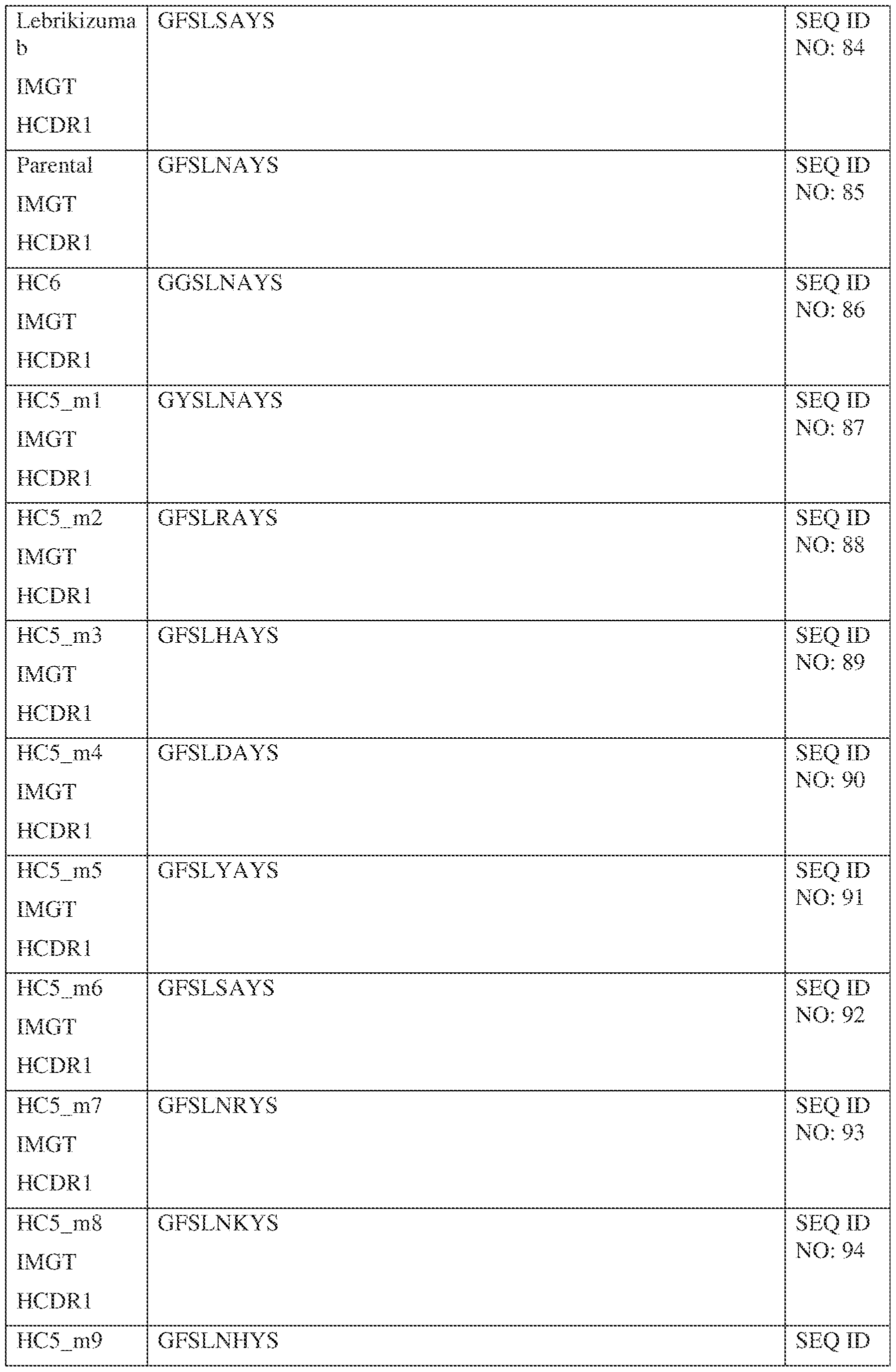
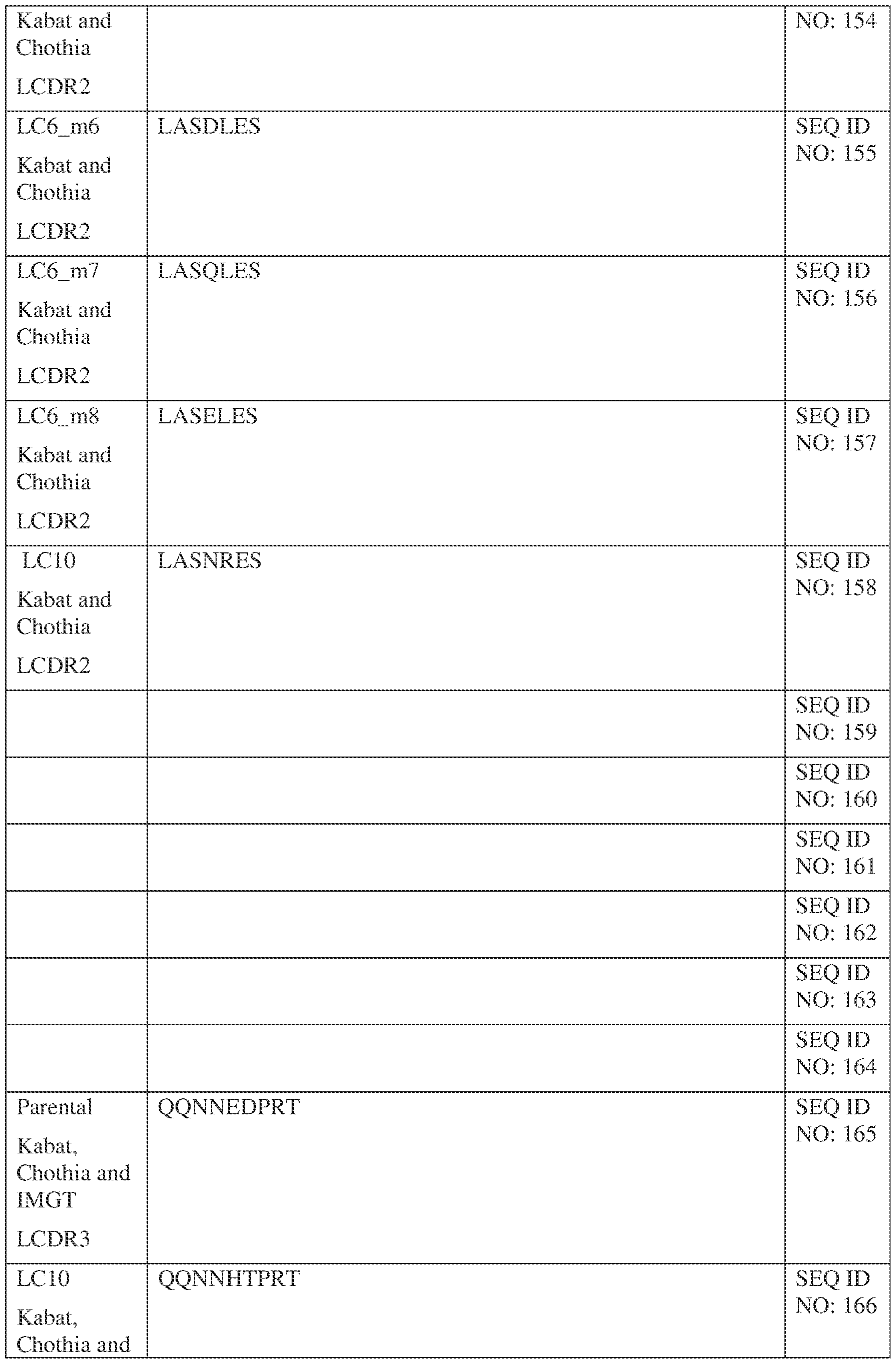
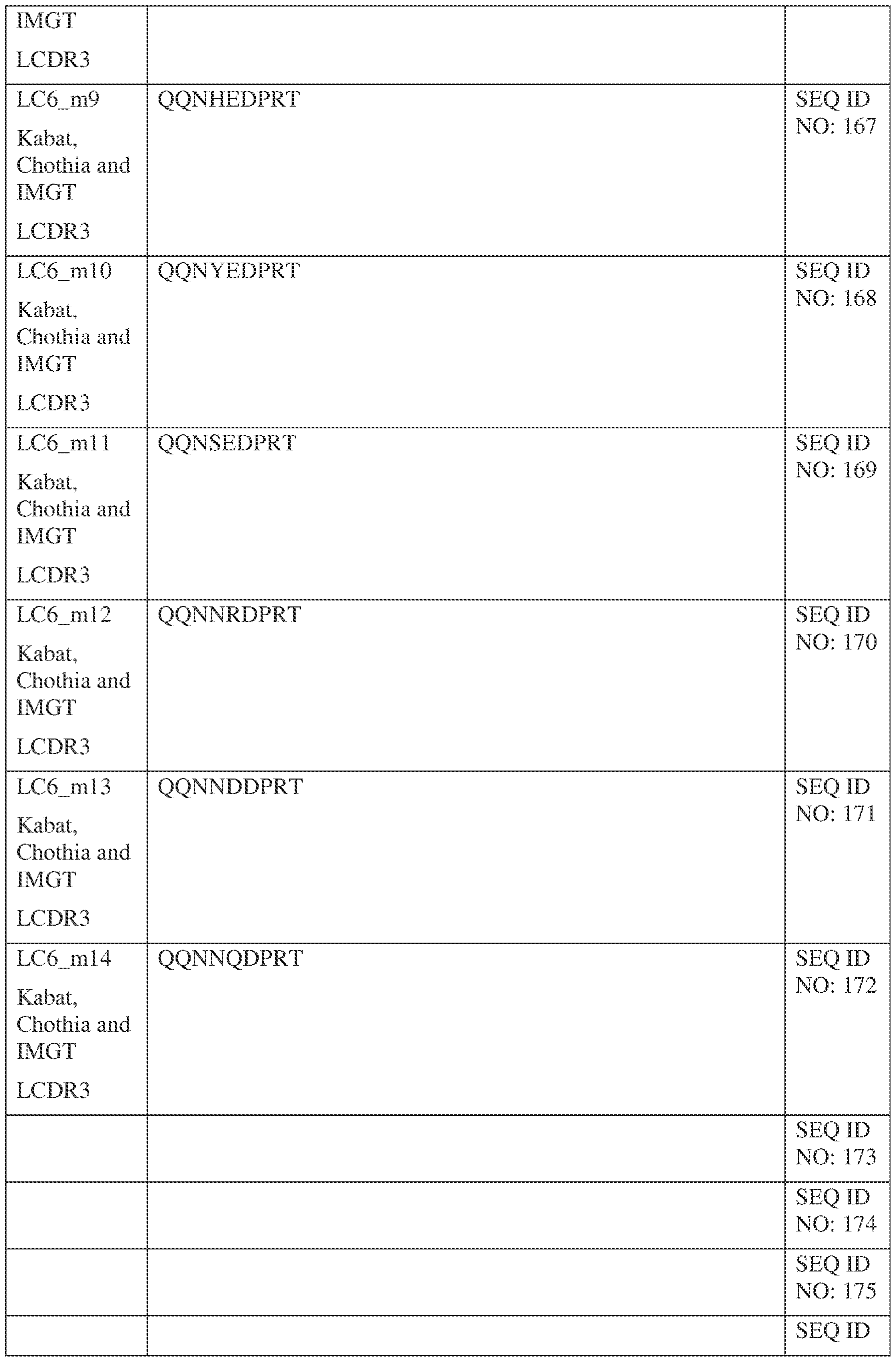
- Table 1 provides the positions of CDR-L1, CDR-L2, CDR-L3, CDR-H1, CDR- H2, and CDR-H3 as identified by the Kabat and Chothia schemes.
- residue numbering is provided using both the Kabat and Chothia numbering schemes.
- CDRs may be assigned, for example, using antibody numbering software, such as Abnum, available at www.bioinf.org.uk/abs/abnum/, and described in Abhinandan and Martin (2008) Immunology, 45:3832-3839, incorporated by reference in its entirety.
- Table 1 Residues in CDRs according to Kabat and Chothia numbering schemes. IPTS/128623945.3 19 Attorney Docket No. PRG-013WO * The C-terminus of CDR-H1, when numbered using the Kabat numbering convention, varies between H32 and H34, depending on the length of the CDR.
- the “EU numbering scheme” is generally used when referring to a residue in an antibody heavy chain constant region (e.g., as reported in Kabat et al., supra). Unless stated otherwise, the EU numbering scheme is used to refer to residues in antibody heavy chain constant regions described herein.
- an antigen-binding domain is an antigen-binding domain formed by a VH-VL dimer of an antibody.
- Another example of an antigen-binding domain is an antigen-binding domain formed by diversification of certain loops from the tenth fibronectin type III domain of an Adnectin.
- An antigen-binding domain can include CDRs 1, 2, and 3 from a heavy chain in that order; and CDRs 1, 2, and 3 from a light chain in that order.
- Epitopes frequently consist of surface-accessible amino acid residues and/or sugar side chains and may have specific three-dimensional structural characteristics, as well as specific charge characteristics. Conformational and non-conformational epitopes are distinguished in that the binding to the former but not the latter may be lost in the presence of denaturing solvents.
- An epitope may comprise amino acid residues that are directly involved in the binding and other amino acid residues, which are not directly involved in the binding.
- the epitope to which an antibody binds can be determined using known techniques for epitope determination such as, for example, testing for antibody binding to IL-13 variants with different point-mutations or to chimeric IL-13 variants.
- a target antigen bound by an antibody of interest e.g., IL-13, TSLP, or TSLPR
- a routine cross-blocking assay such as that described in Antibodies, A Laboratory Manual, Cold Spring Harbor Laboratory, Ed Harlow and David Lane (1988), can be performed.
- epitope mapping can be performed by methods known in the art.
- Chimeric antibodies are antibodies in which a portion of the heavy and/or light chain is derived from a particular source or species, while the remainder of the heavy and/or light chain is derived from a different source or species.
- Human antibodies are antibodies which possesses an amino acid sequence corresponding to that of an antibody produced by a human or a human cell, or derived from a IPTS/128623945.3 20 Attorney Docket No. PRG-013WO non-human source that utilizes a human antibody repertoire or human antibody-encoding sequences (e.g., obtained from human sources or designed de novo). Human antibodies specifically exclude humanized antibodies.
- a humanized antibody has a sequence that differs from the sequence of an antibody derived from a non-human species by one or more amino acid substitutions, deletions, and/or additions, such that the humanized antibody is less likely to induce an immune response, and/or induces a less severe immune response, as compared to the non- human species antibody, when it is administered to a human subject.
- certain amino acids in the framework and constant domains of the heavy and/or light chains of the non-human species antibody are mutated to produce the humanized antibody.
- the constant domain(s) from a human antibody are fused to the variable domain(s) of a non-human species.
- one or more amino acid residues in one or more CDR sequences of a non-human antibody are changed to reduce the likely immunogenicity of the non-human antibody when it is administered to a human subject, wherein the changed amino acid residues either are not critical for immunospecific binding of the antibody to its antigen, or the changes to the amino acid sequence that are made are conservative changes, such that the binding of the humanized antibody to the antigen is not significantly worse than the binding of the non-human antibody to the antigen.
- Examples of how to make humanized antibodies can be found in U.S. Pat. Nos.6,054,297, 5,886,152 and 5,877,293. For further details, see Jones et al. (1986) Nature 321:522-525; Riechmann et al.
- the two or more different epitopes may be epitopes on the same antigen (e.g., a single IL-13) or on different antigens (e.g., different IL-13 molecules, or a IL-13 molecule and a non- IL-13 molecule).
- a multi-specific antibody binds two different epitopes (i.e., a “bispecific antibody”). In some embodiments, a multi-specific antibody binds three different epitopes (i.e., a “trispecific antibody”).
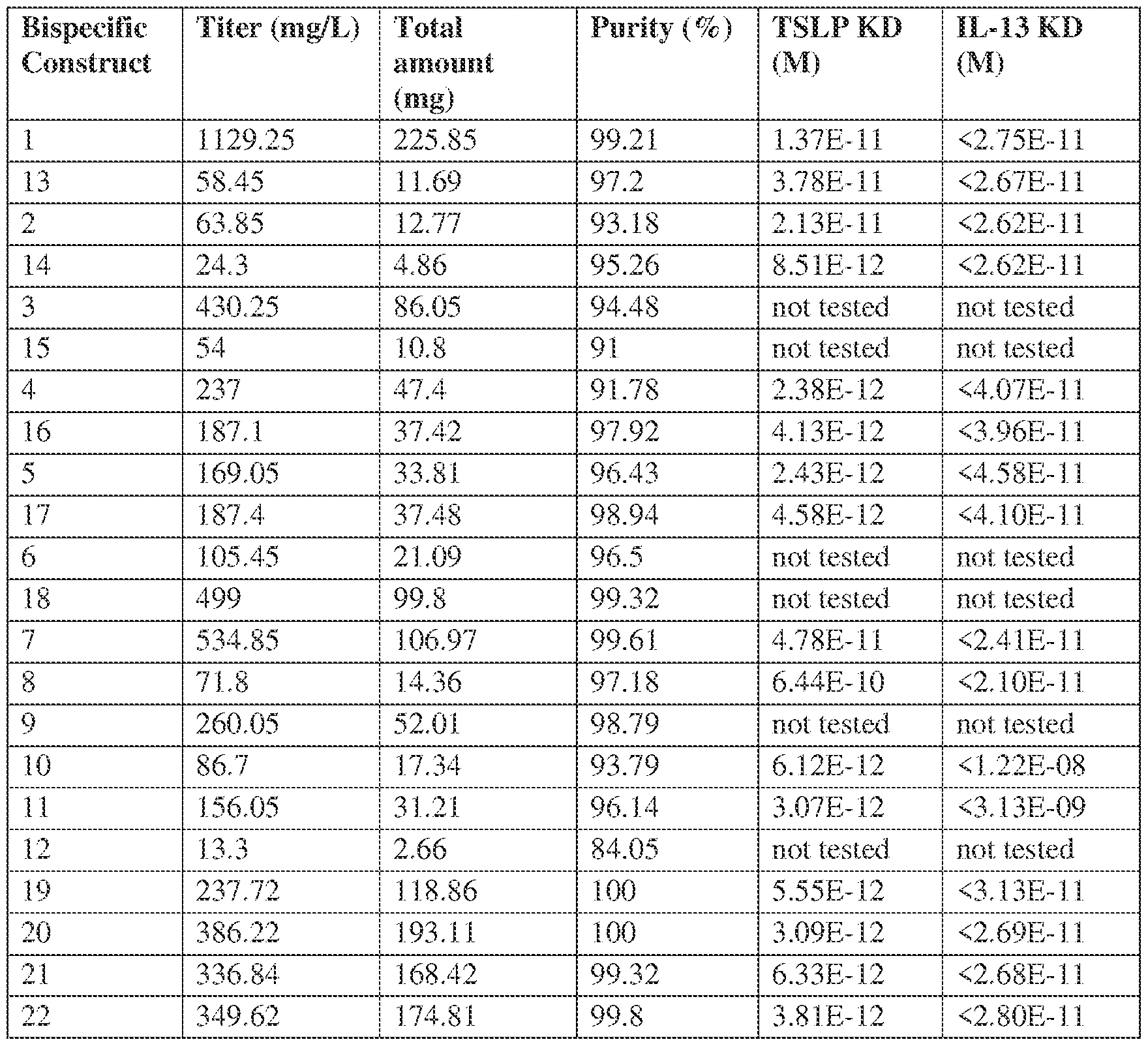
- Anti-IL-13/TSLP or TSLPR antibodies can include those described herein such as the clones set forth in the drawings and/or tables.
- the bispecific antibody comprises an alternative scaffold.
- the bispecific antibody consists of an alternative scaffold.
- the bispecific antibody consists essentially of an alternative scaffold.
- the bispecific antibody comprises an antibody fragment.
- the bispecific antibody consists of an IPTS/128623945.3 21 Attorney Docket No. PRG-013WO antibody fragment.
- the bispecific antibody consists essentially of an antibody fragment.
- the bispecific antibodies are monoclonal antibodies.
- the bispecific antibodies are polyclonal antibodies.
- the bispecific antibodies are produced by hybridomas. In other embodiments, the bispecific antibodies are produced by recombinant cells engineered to express the desired variable and constant domains. [00131] In some embodiments the bispecific antibodies may be single chain antibodies or other antibody derivatives retaining the antigen specificity and the lower hinge region or a variant thereof. [00132] In some embodiments the bispecific antibodies may be polyfunctional antibodies, recombinant antibodies, human antibodies, humanized antibodies, fragments or variants thereof. In particular embodiments, the antibody fragment or a derivative thereof is selected from a Fab fragment, a Fab ⁇ 2 fragment, a CDR, and scFv. [00133] In some embodiments, the bispecific antibodies are capable of forming an immune complex.
- an immune complex can be a tumor cell covered by bispecific antibodies.
- sequence comparison typically one sequence acts as a reference sequence to which test sequences are compared.
- test and reference sequences are input into a computer, subsequence coordinates are designated, if necessary, and sequence algorithm program parameters are designated.
- sequence comparison algorithm then calculates the percent sequence identity for the test sequence(s) relative to the reference sequence, based on the designated program parameters.
- Bispecific Antibody Structure [00135]
- the present application provides antibodies and compositions comprising a bispecific antibody that binds IL-13 and TSLP or TSLPR.
- the bispecific antibodies disclosed here can be of any structure known in the art.
- the bispecific antibody can take any format shown in but not limited to the examples below.
- One format is a bispecific antibody that includes a first immunoglobulin heavy chain, a second immunoglobulin heavy chain and an immunoglobulin light chain.
- the first immunoglobulin heavy chain includes a first Fc (hinge-CH2-CH3) domain, a first variable heavy chain domain and an optional first CH1 heavy chain domain.
- the immunoglobulin light chain includes a variable light chain domain and a constant light chain IPTS/128623945.3 22 Attorney Docket No. PRG-013WO domain; together with the first immunoglobulin heavy chain, the immunoglobulin light chain forms an antigen-binding site that binds IL-13.
- the second immunoglobulin heavy chain comprises a second Fc (hinge-CH2-CH3) domain, a second variable heavy chain domain and a second CH1 heavy chain domain that may pair with an immunoglobulin light chain identical to the one that pairs with the first immunoglobulin heavy chain, except that when immunoglobulin light chain is paired with the second immunoglobulin heavy chain, the resulting antigen binding site binds to TSLP or TSLPR.
- Another exemplary format, as shown in Figure 1 involves a bispecific antibody that includes two sets of immunoglobulin light chain and immunoglobulin heavy chain pairs (IgG-scFv form).
- the first light chain/heavy chain pair includes a first immunoglobulin heavy chain, a immunoglobulin light chain and an scFv.
- the first immunoglobulin heavy chain includes, from N- to C-terminus, a first VH1 domain, a first CH1 domain, a first Fc (hinge-CH2-CH3) domain fused via either a linker or an antibody hinge to a single chain Fv (scFv).
- linkers can be used for linking the scFv to the first Fc domain (linker H2) or between the VL2 and the VH2 of the scFv itself (see black line connecting VH2 and VL2 of scFv in inset of Figure 1, referred to as linker H3 herein).
- the immunoglobulin light chain includes, from N- to C-terminus, a first variable light chain (VL1) domain and a first constant light chain (CL) domain.
- the first light chain/heavy chain pair binds to a second light chain/heavy chain pair to form two Fab regions and an Fc domain comprising the first Fc domain and a second Fc domain from the second heavy chain.
- the C-terminus of each of the first Fc domain and the second Fc domain is linked to an scFv.
- the Fab region can bind to IL-13 and the scFv region can bind to TSLP or TSLPR, or the Fab region can bind to TSLP or TSLPR, and the scFv region can bind to IL-13.
- the bispecific antibody is in the dual-variable domain immunoglobulin (DVD-IgTM) form, as shown in Figure 2.
- the DVD-IgTM combines the target binding domains of two monoclonal antibodies via flexible, naturally occurring linkers, and yields a tetravalent IgG-like molecule.
- the DVD-Ig format includes two sets of immunoglobulin light chain and immunoglobulin heavy chain pairs.
- the first heavy chain includes, from N- to C-terminus, a first heavy chain variable domain (VH1), a second heavy chain variable domain (VH’), and a first heavy chain constant domain (CH1-CH2-CH3).
- the VH1 and VH’ domains are connected by a linker.
- the first light chain includes, from N- to C-terminus, a first light chain variable domain (VL1), a second IPTS/128623945.3 23 Attorney Docket No. PRG-013WO light chain variable domain (VL’), and a constant light chain domain (CL).
- VL first light chain variable domain
- VL second IPTS/128623945.3 23
- CL constant light chain domain
- the VL’ is fused to the VL2 by a linker.
- the bispecific antibody is in the CrossMab format, as shown in Figure 3.
- the CrossMab form is a bispecific antibody that includes two sets of immunoglobulin light chain and immunoglobulin heavy chain pairs.
- the first light chain/heavy chain pair includes a first immunoglobulin heavy chain (HC1) with an Fc domain and an immunoglobulin light chain (LC1).
- the first immunoglobulin heavy chain includes, from N- to C-terminus, a first VH domain (VH1) and a first constant heavy chain domain (CH1-CH2-CH3)
- the first immunoglobulin light chain includes, from N- to C- terminus, a first VL domain and a first constant light chain (CL) domain
- the second immunoglobulin heavy chain (HC2) includes, from N- to C-terminus, a second VH domain, (VH2) and a second constant heavy chain domain (CL’-CH2-CH3)
- the second immunoglobulin light chain (LC2) includes a second VL domain (VL2) and a second constant heavy chain (CH1’) domain.
- the CH3 domains for the HC1 and HC2 can, in some embodiments, comprise mutations to promote heterodimerization (e.g., knob and hole mutations).
- the bispecific antibody is in the Triomab form, which is a trifunctional, bispecific antibody that maintains an IgG-like shape. This chimera consists of two half antibodies, each with one light and one heavy chain, that originate from two parental antibodies.
- the bispecific antibody is the KiH Common Light Chain (LC) form, which involves the knobs-into-holes (KIHs) technology.
- LC KiH Common Light Chain
- the KIH involves engineering C H 3 domains to create either a “knob” or a “hole” in each heavy chain to promote heterodimerization.
- the concept behind the “Knobs-into-Holes (KiH)” Fc technology was to introduce a “knob” in one CH3 domain (CH3A) by substitution of a small residue with a bulky one (e.g., T366W CH3A in EU numbering).
- a complementary “hole” surface was created on the other CH3 domain (CH3B) by replacing the closest neighboring residues to the knob with smaller ones (e.g., T366S/L368A/Y407VCH3B).
- the “hole” mutation was optimized by structured-guided phage library screening (Atwell et al. (1997) “Stable heterodimers from remodeling the domain interface of a homodimer using a phage display library,” J. Mol. Biol.270(1):26–35).
- X-ray crystal structures of KiH Fc variants (Elliott et al. (2014) “Antiparallel conformation of knob and hole aglycosylated half-antibody homodimers is mediated by a CH2-CH3 hydrophobic interaction,” J. Mol. Biol.426(9):1947–57; Mimoto et al. (2014) “Crystal structure of a novel IPTS/128623945.3 24 Attorney Docket No.
- the bispecific antibody is in the Orthogonal Fab interface (Ortho-Fab) form.
- Orthogonal Fab interface Ortho-Fab
- the bispecific antibody is in the 2-in-1 Ig format.
- the bispecific antibody is in the ES form, which is a heterodimeric construct containing two different Fabs binding to targets 1 and target 2 fused to the Fc. Heterodimerization is ensured by electrostatic steering mutations in the Fc.
- the bispecific antibody is in the ⁇ -Body form, which is an heterodimeric constructs with two different Fabs fused to Fc stabilized by heterodimerization mutations: Fab1 targeting antigen 1 contains kappa LC, while second Fab targeting antigen 2 contains lambda LC.
- the bispecific antibody is in Fab Arm Exchange form (antibodies that exchange Fab arms by swapping a heavy chain and attached light chain (half- molecule) with a heavy-light chain pair from another molecule, which results in bispecific antibodies).
- the bispecific antibody is in the SEED Body form.
- the strand-exchange engineered domain (SEED) platform was designed to generate asymmetric and bispecific antibody-like molecules, a capability that expands therapeutic applications of natural antibodies.
- This protein engineered platform is based on exchanging structurally related sequences of immunoglobulin within the conserved CH3 domains.
- the SEED design allows efficient generation of AG/GA heterodimers, while disfavoring homodimerization of AG and GA SEED CH3 domains.
- the bispecific antibody is in the LuZ-Y form, in which a leucine zipper is used to induce heterodimerization of two different HCs. (Wranik et al. (2012) J.
- the bispecific antibody is in the Cov-X-Body form.
- bispecific CovX-Bodies two different peptides are joined together using a branched azetidinone linker and fused to the scaffold antibody under mild conditions in a site-specific manner. Whereas the pharmacophores are responsible for functional activities, the antibody scaffold imparts long half-life and Ig-like distribution.
- the pharmacophores can be chemically optimized or replaced with other pharmacophores to generate optimized or unique bispecific antibodies. (Doppalapudi et al.
- the bispecific antibody is in an Oasc-Fab heterodimeric form that includes Fab binding to target 1, and scFab binding to target 2 fused to Fc. Heterodimerization is ensured by mutations in the Fc.
- the bispecific antibody is in a DuetMab form, which is an heterodimeric construct containing two different Fabs binding to antigens 1 and 2, and Fc stabilized by heterodimerization mutations.
- Fab 1 and 2 contain differential S-S bridges that ensure correct LC and HC pairing.
- the bispecific antibody is in a CrossmAb form, which is an heterodimeric construct with two different Fabs binding to targets 1 and 2, fused to Fc stabilized by heterodimerization. CL and CH1 domains and VH and VL domains are switched, e.g., CH1 is fused in-line with VL, while CL is fused in-line with VH.
- the bispecific antibody is n a Fit-Ig form, which is an homodimeric constructs where Fab binding to antigen 2 is fused to the N terminus of HC of Fab that binds to antigen 1. The construct contains wild-type Fc.
- a bispecific antibody provided herein comprises a first VH sequence selected from SEQ ID NOs: 1-32, 470, and 688 and a second VH sequence selected from SEQ ID NOs: 659-661, 682, 684, 686, and 688.
- a bispecific antibody provided herein comprises a first VH sequence comprising SEQ ID NO: 3 and a second VH sequence selected from SEQ ID NOs: 659-661, 682, 684, 686, and 688.
- a bispecific antibody provided herein comprises a first VH sequence having at least about 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% identity to a VH sequence selected from SEQ ID NOs: 1-32, 470, and 688 and a second VH sequence selected from SEQ ID NOs: 659-661, 682, 684, 686, and 688.
- a bispecific antibody provided herein comprises a first VH sequence having at least about 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% identity to a VH sequence comprising SEQ ID NO: 3 and a second VH sequence selected from SEQ ID NOs: 659-661, 682, 684, 686, and 688.
- a bispecific antibody provided herein comprises a first VH sequence selected from SEQ ID NOs: 1-32, 470, and 688 and a second VH sequence selected from SEQ ID NOs: 659-661, 682, 684, 686, and 688, with up to 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 amino acid substitutions.
- a bispecific antibody provided herein comprises a first VH sequence comprising SEQ ID NO: 3 and a second VH sequence selected from SEQ ID NOs: 659-661, 682, 684, 686, and 688, with up to 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 amino acid substitutions.
- the amino acid substitutions are conservative amino acid substitutions.
- the bispecific antibodies described in this paragraph are referred to herein as “variants.”
- such variants are derived from a sequence provided herein, for example, by affinity maturation, site directed mutagenesis, random mutagenesis, or any other method known in the art or described herein.
- such variants are not derived from a sequence provided herein and may, for example, be isolated de novo according to the methods provided herein for obtaining bispecific antibodies.
- a bispecific antibody provided herein comprises a first VL sequence selected from SEQ ID NOs: 33-57, 471, and 687 and a second VL sequence selected from 662-664, 681, 683, and 685.
- a bispecific antibody provided herein comprises a first VL sequence comprising SEQ ID NO: 39 and a second VL sequence selected from SEQ ID NOs: 662-664, 681, 683, and 685.
- PRG-013WO sequence comprising SEQ ID NO: 39 and a second VL sequence selected from SEQ ID NOs: 662-664, 681, 683, and 685.
- a bispecific antibody provided herein comprises a first VL sequence selected from SEQ ID NOs: 33-57, 471, and 687, and a second VL sequence selected from SEQ ID NOs: 662-664, 681, 683, and 685, with up to 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 amino acid substitutions.
- a bispecific antibody provided herein comprises a first VL sequence comprising SEQ ID NO: 39 and a second VL sequence selected from SEQ ID NOs: 662-664, 681, 683, and 685, with up to 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 amino acid substitutions. Table 1a.
- a bispecific antibody comprises a first VH sequence selected from SEQ ID NOs: 1-32, 470, and 688 and a second VH sequence selected from 659-661, 682, 684, 686, and 688; and a first VL sequence selected from SEQ ID NOs: 33-57, 471, and 687, and a second VL sequence selected from SEQ ID NOs: 662-664, 681, IPTS/128623945.3 28 Attorney Docket No. PRG-013WO 683, and 685.
- a bispecific antibody provided herein comprises a first VH sequence comprising SEQ ID NOs: 3 and a second VH sequence selected from 659-661, 682, 684, 686, and 688; and a first VL sequence comprising SEQ ID NOs: 39 and a second VL sequence selected from 662-664, 681, 683, and 685.
- any of SEQ ID NOs: 1-32, 470, and 688 can be combined with any of SEQ ID NOs: 33-57, 471, and 687, and any of SEQ ID NOs: 659-661, 682, 684, 686, and 688 can be combined with any of SEQ ID NOs: 662-664, 681, 683, and 685.
- a bispecific antibody provided herein comprises a first VH sequence having at least about 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% identity to a VH sequence provided in selected from SEQ ID NOs: 1-32, 470, and 688 and a second VH sequence selected from SEQ ID NOs: 659-661, 682, 684, 686, and 688; and a first VL sequence having at least about 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% identity to a VL sequence provided in SEQ ID NOs: 33-57, 471, and 687 and a second VL sequence selected from SEQ ID NOs: 662-664, 681, 683, and 685.
- a bispecific antibody provided herein comprises a first VH sequence having at least about 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% identity to a VH sequence comprising SEQ ID NO: 3 and a second VH sequence selected from SEQ ID NOs 659-661, 682, 684, 686, and 688; and a first VL sequence having at least about 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% identity to a VL sequence comprising SEQ ID NO: 39 and a second VL sequence selected from SEQ ID NOs: 662-664, 681, 683, and 685.
- a bispecific antibody provided herein comprises a first VH sequence provided in SEQ ID NOs: 1-32, 470, and 688 and a second VH sequence selected from SEQ ID NOs: 659-661, 682, 684, 686, and 688, with up to 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 amino acid substitutions; and a first VL sequence provided in SEQ ID NOs: 33-57, 471, and 687 and a second VL sequence selected from SEQ ID NOs: 662-664, 681, 683, and 685, with up to 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 amino acid substitutions.
- a bispecific antibody provided herein comprises a first VH sequence comprising SEQ ID NO: 3 and a second VH sequence selected from SEQ ID NOs: 659-661, 682, 684, 686, and 688, with up to 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 amino acid substitutions; and a first VL sequence comprising SEQ ID NO: 39 and a second VL sequence selected from SEQ ID NOs: 662-664, 681, 683, and 685, with up to 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 amino acid substitutions
- the amino acid substitutions are conservative amino acid substitutions.
- the antibodies described in IPTS/128623945.3 29 Attorney Docket No. PRG-013WO this paragraph are referred to herein as “variants.”
- such variants are derived from a sequence provided herein, for example, by affinity maturation, site directed mutagenesis, random mutagenesis, or any other method known in the art or described herein.
- such variants are not derived from a sequence provided herein and may, for example, be isolated de novo according to the methods provided herein for obtaining antibodies.
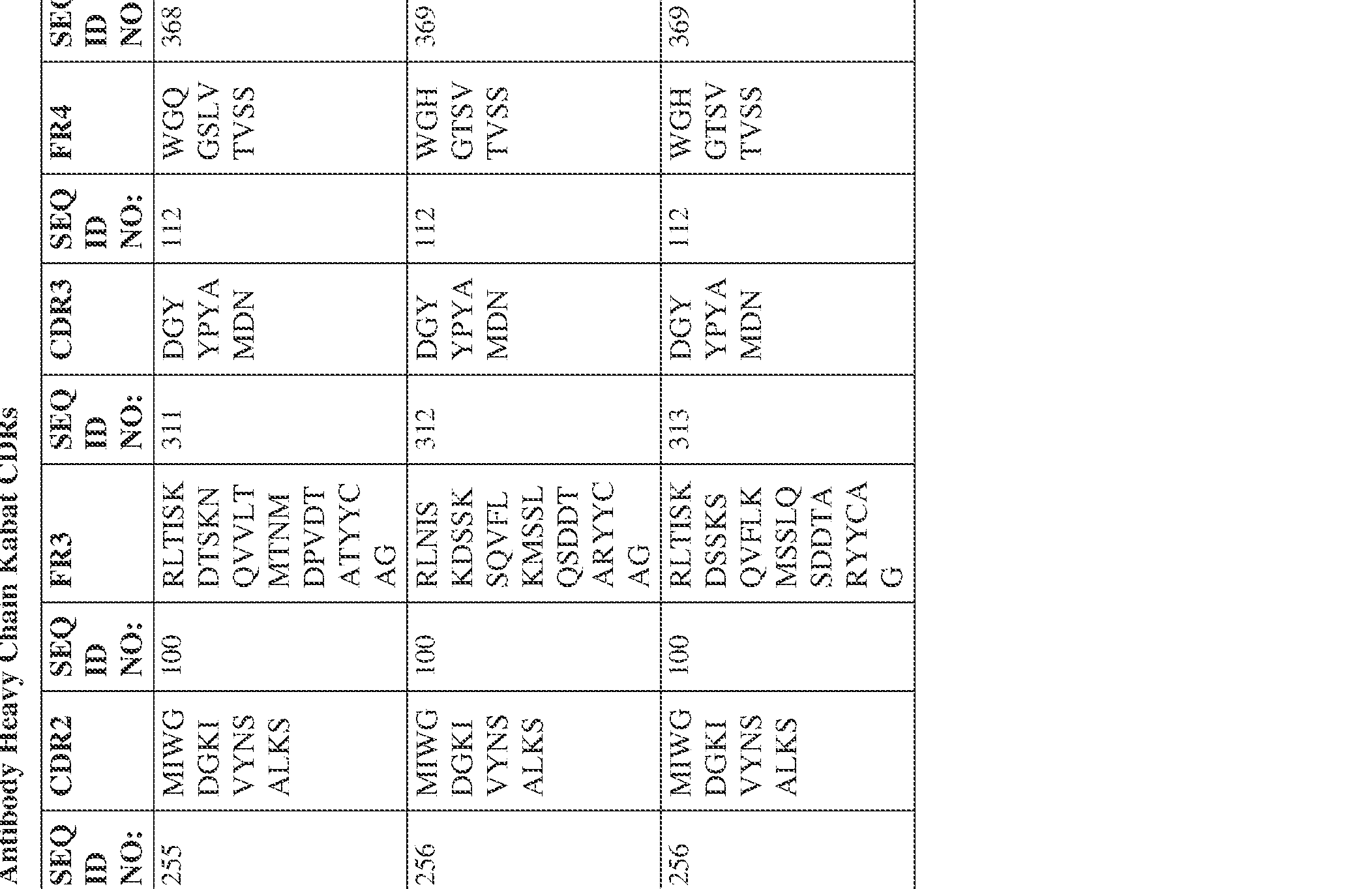
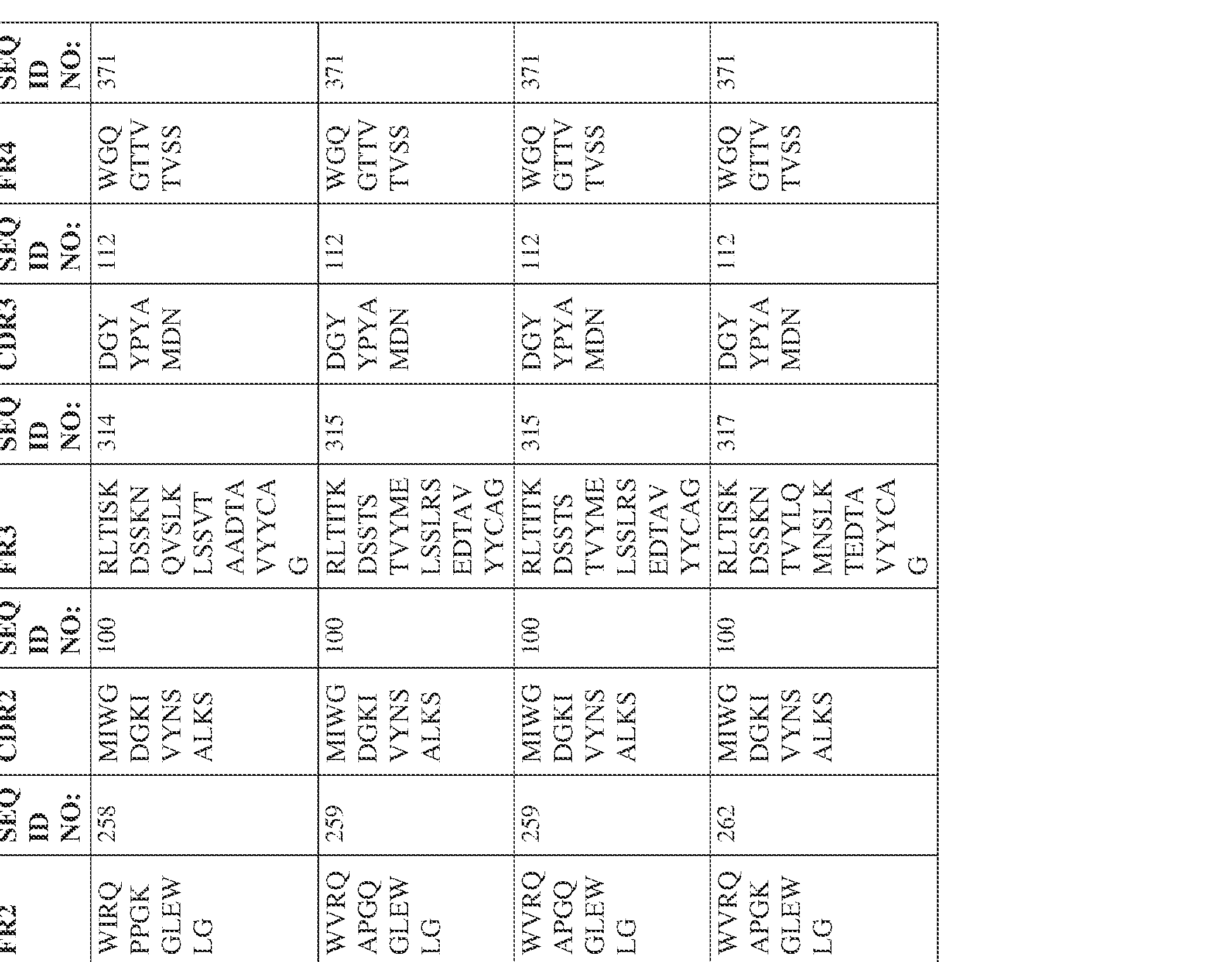
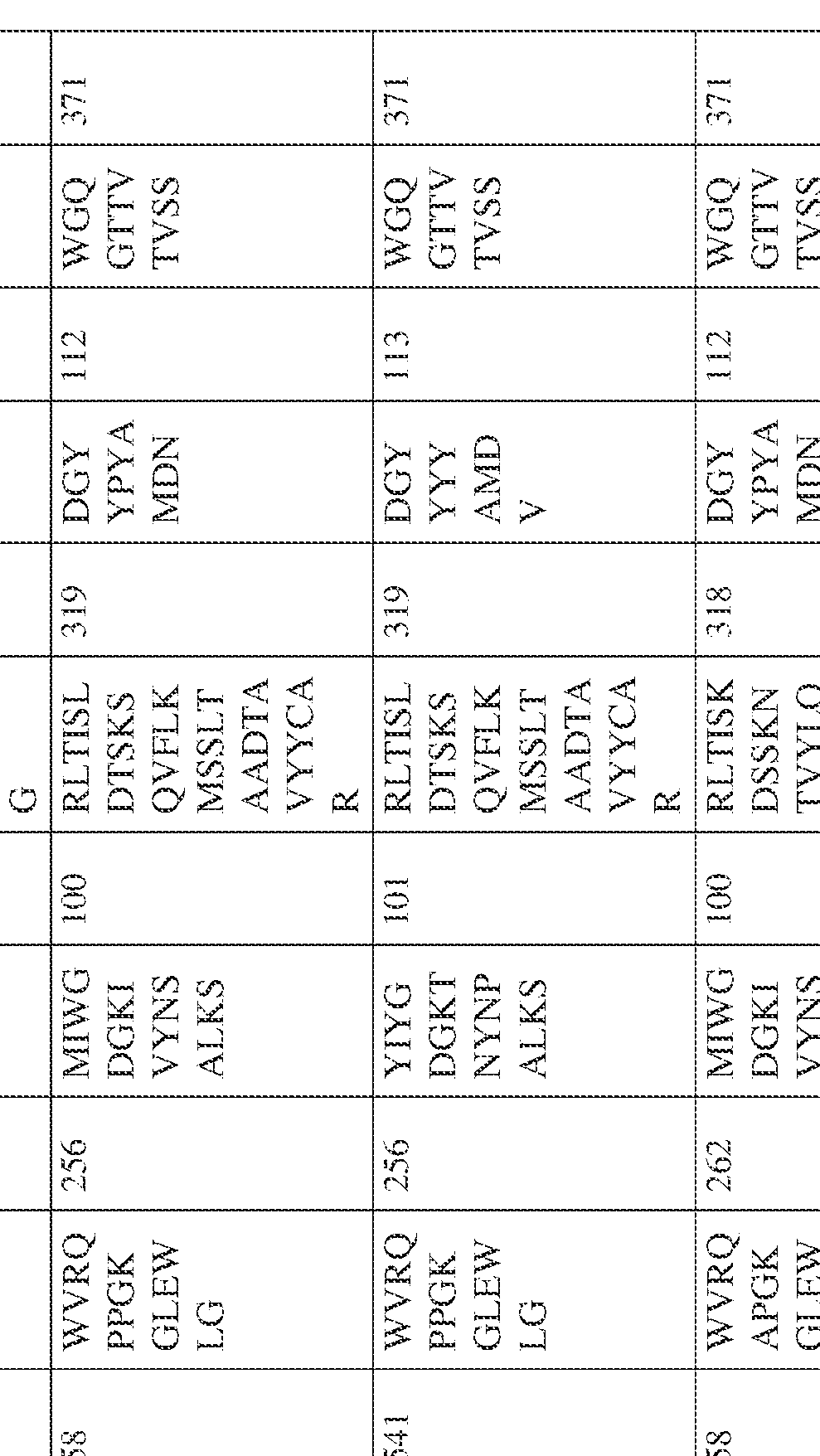
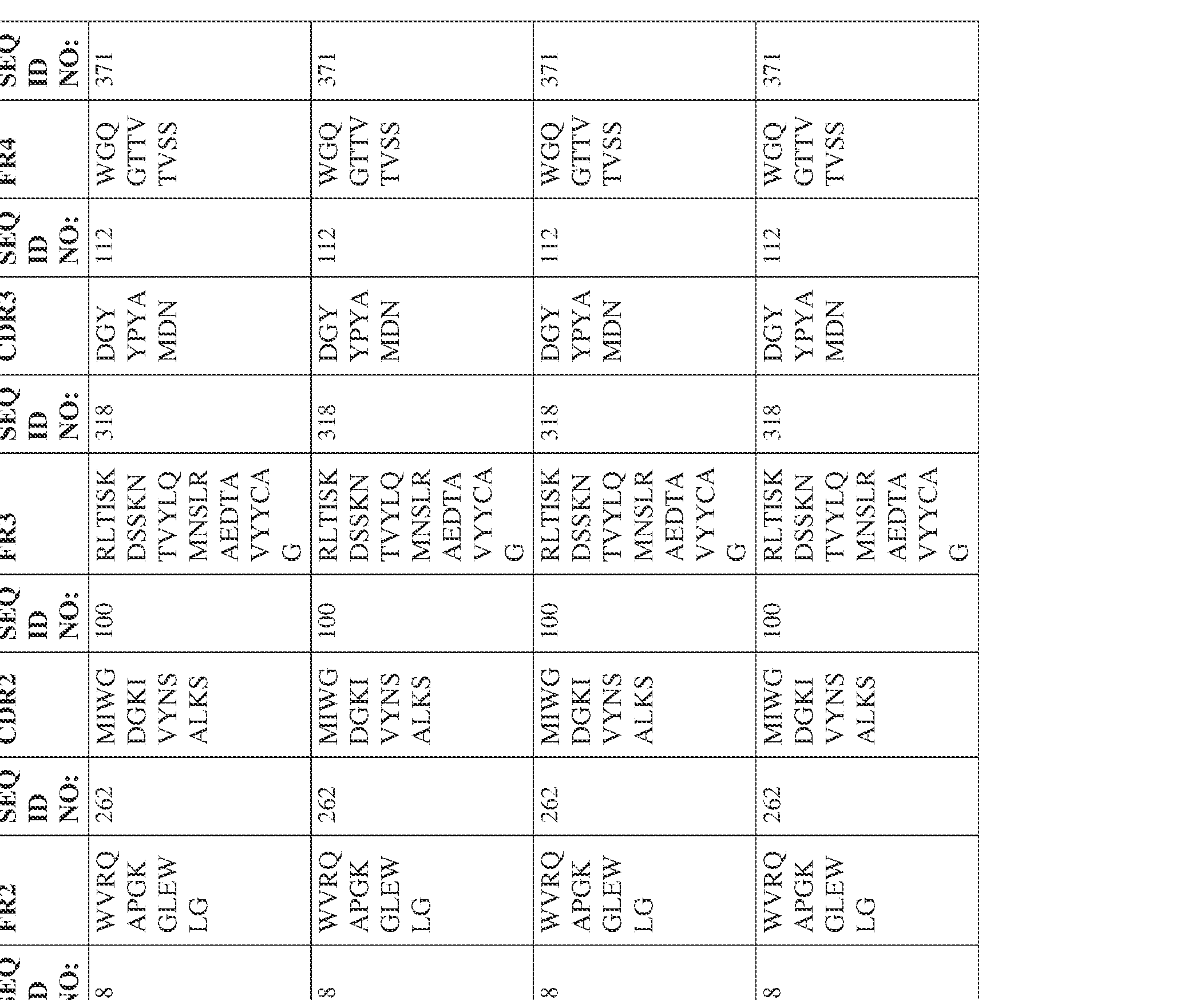
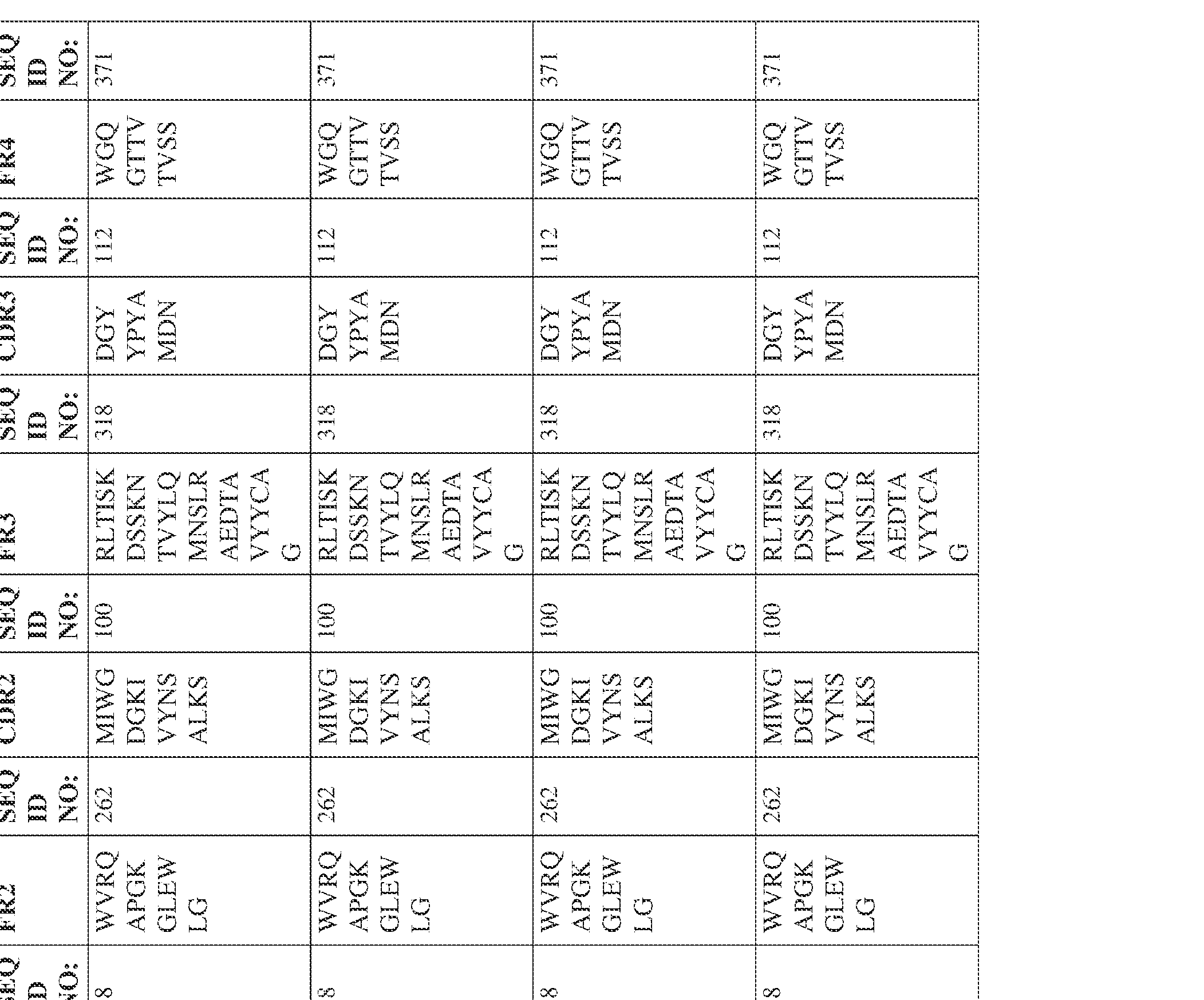
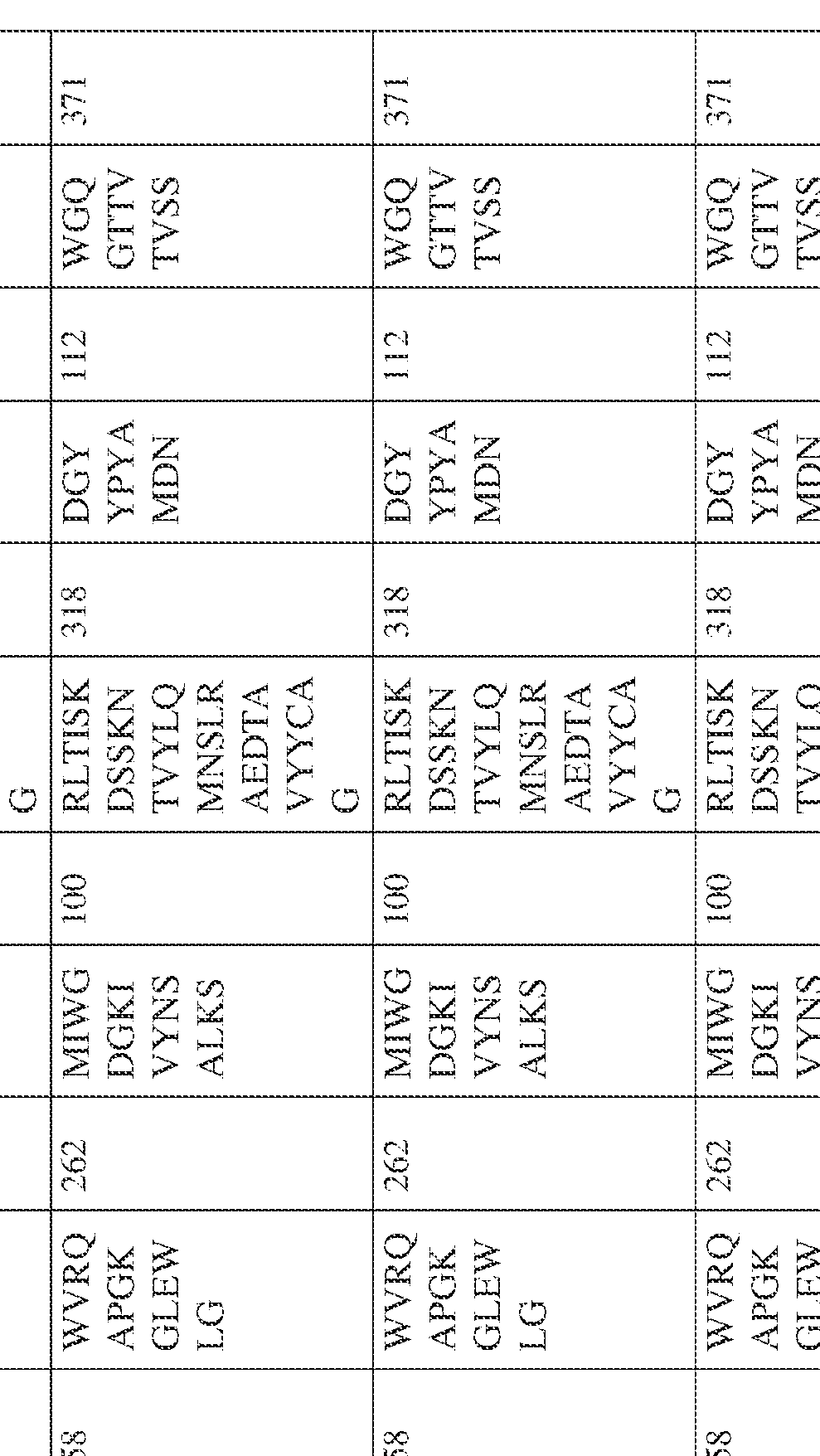
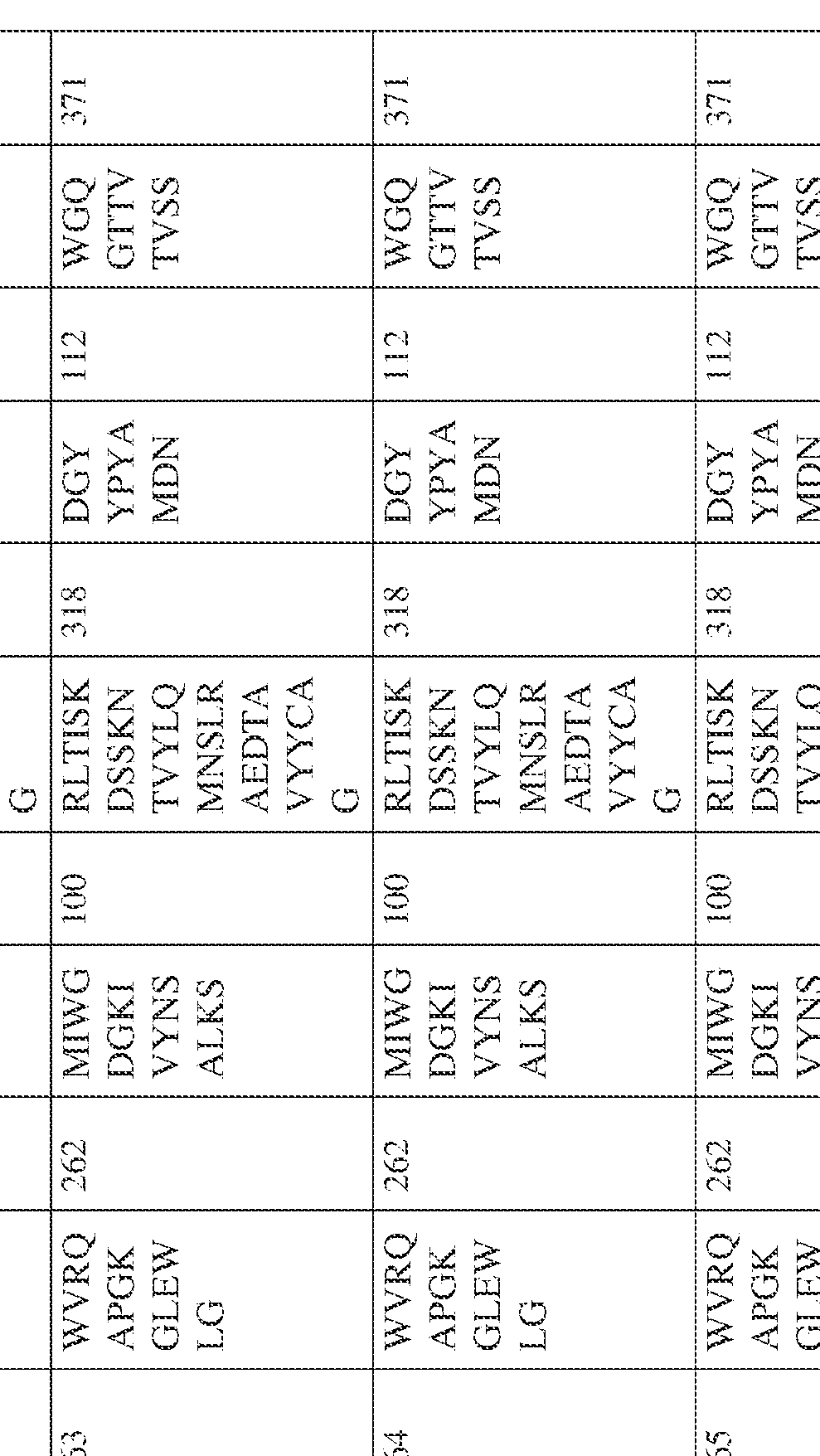
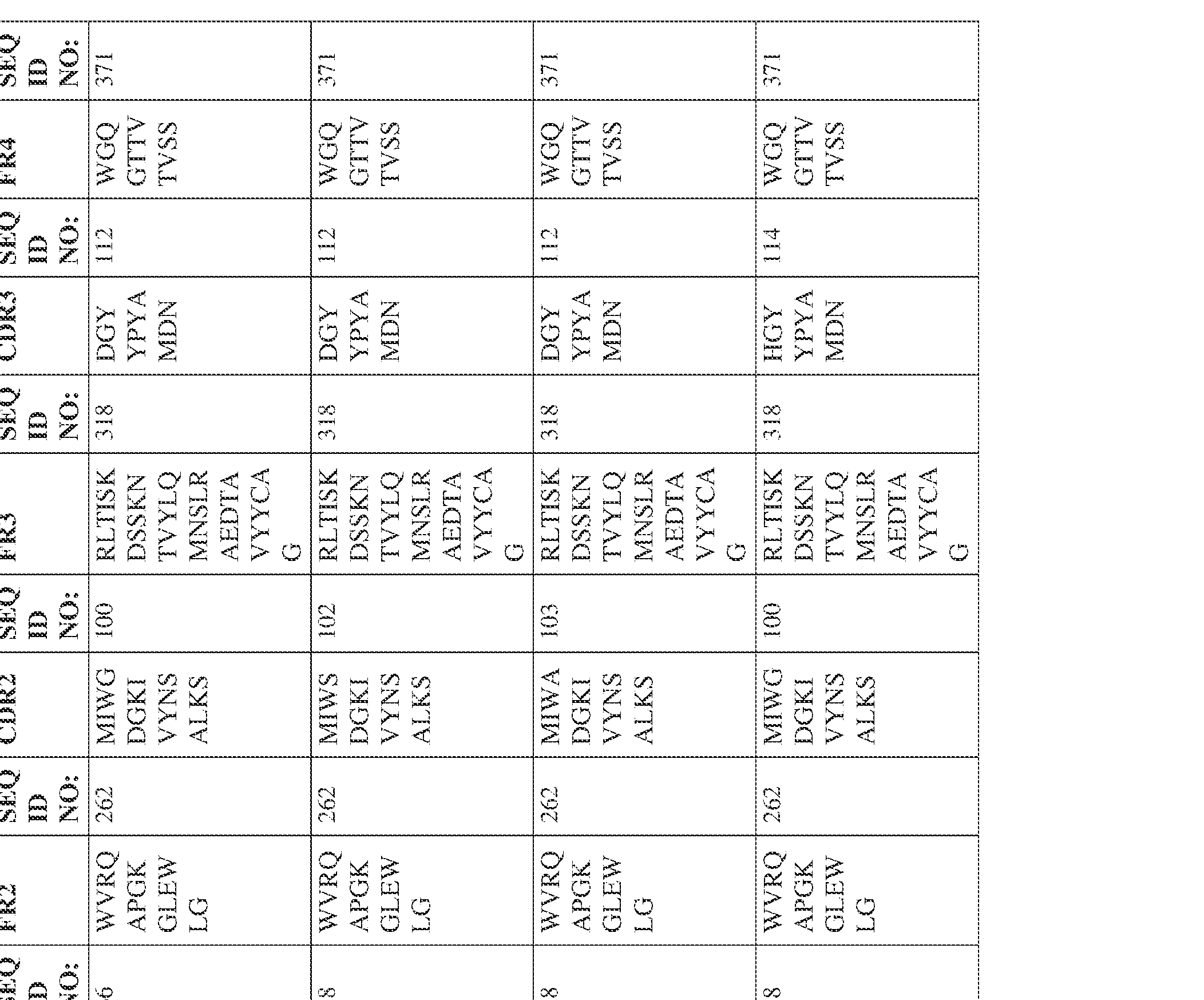
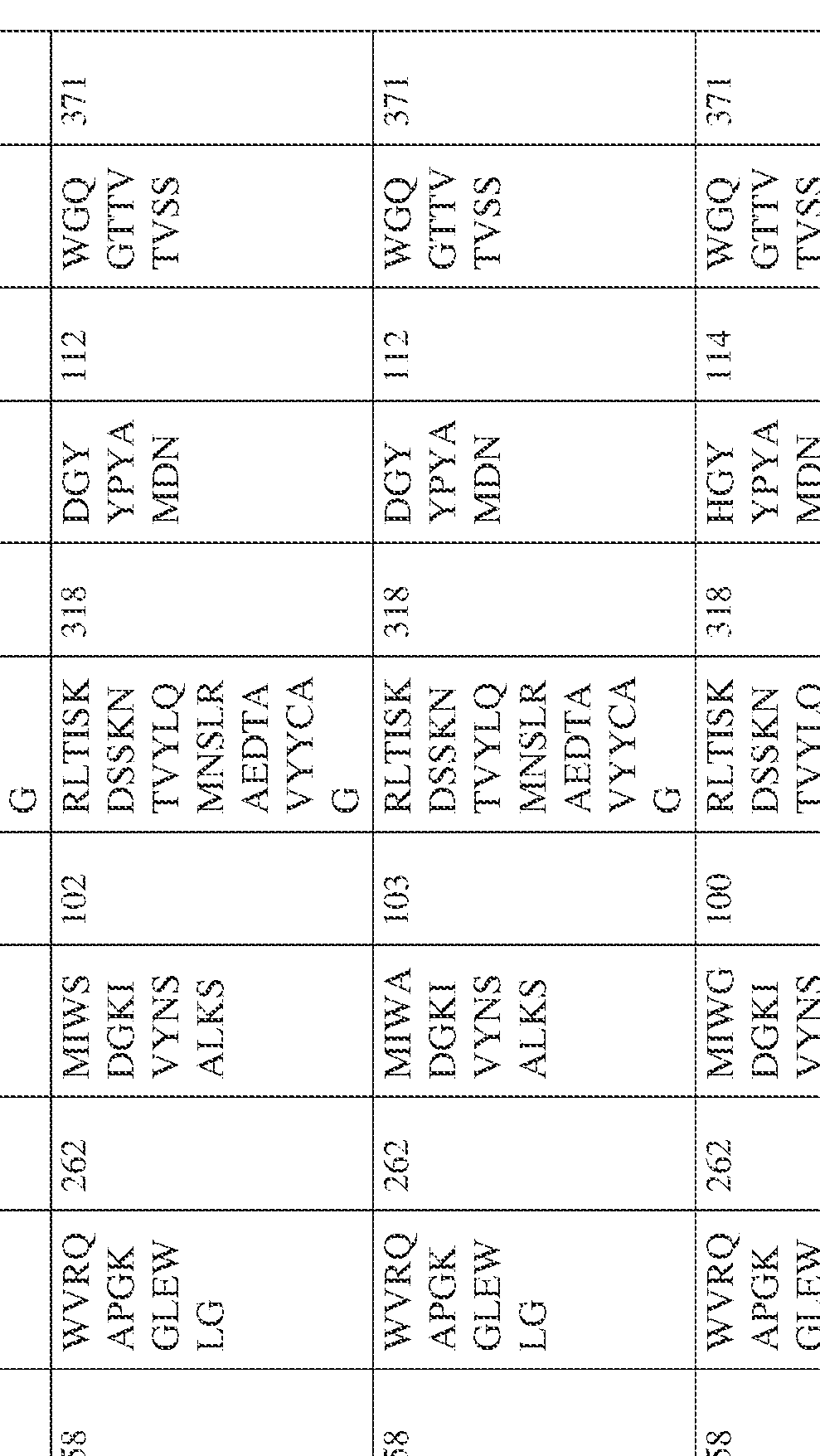
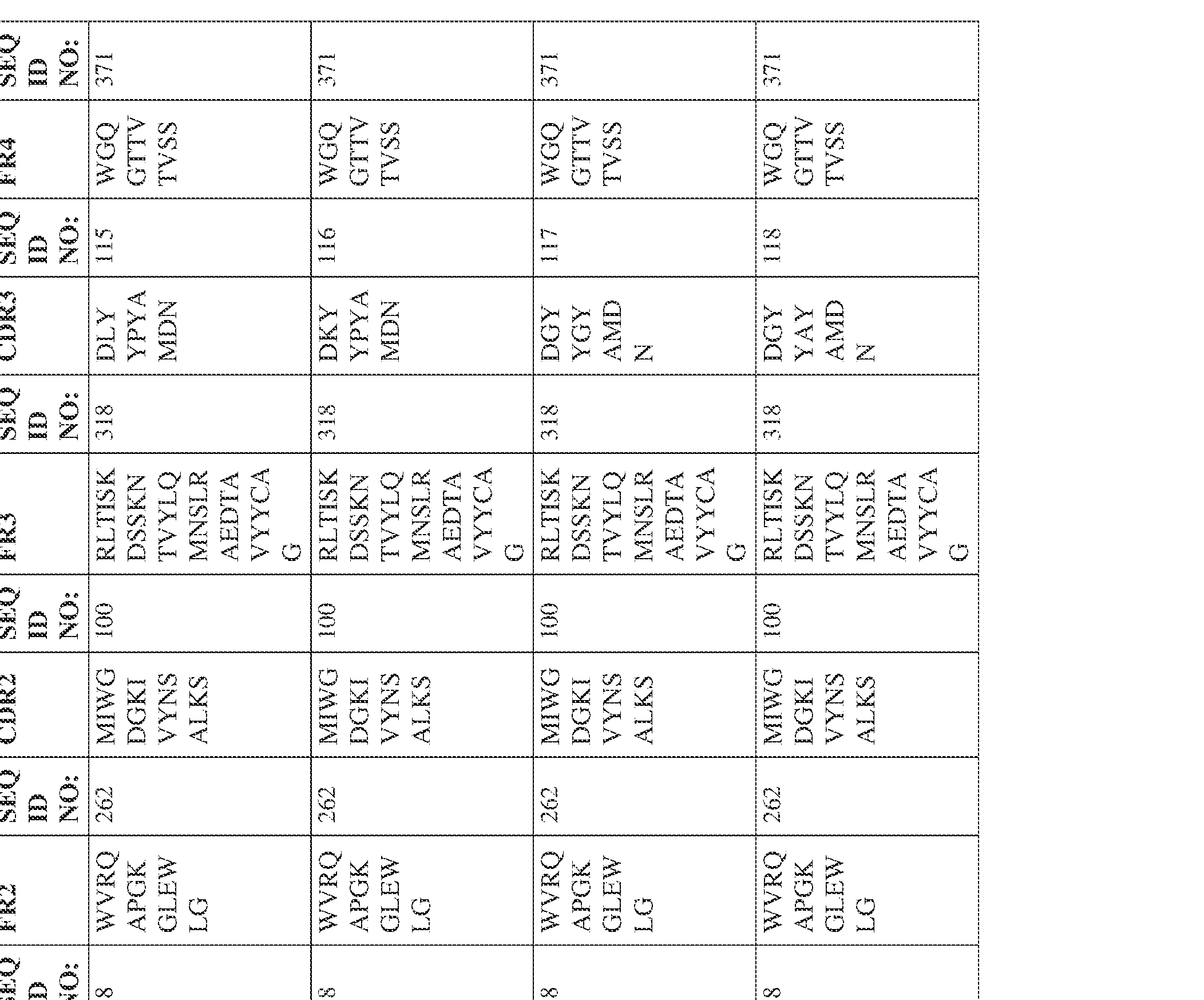
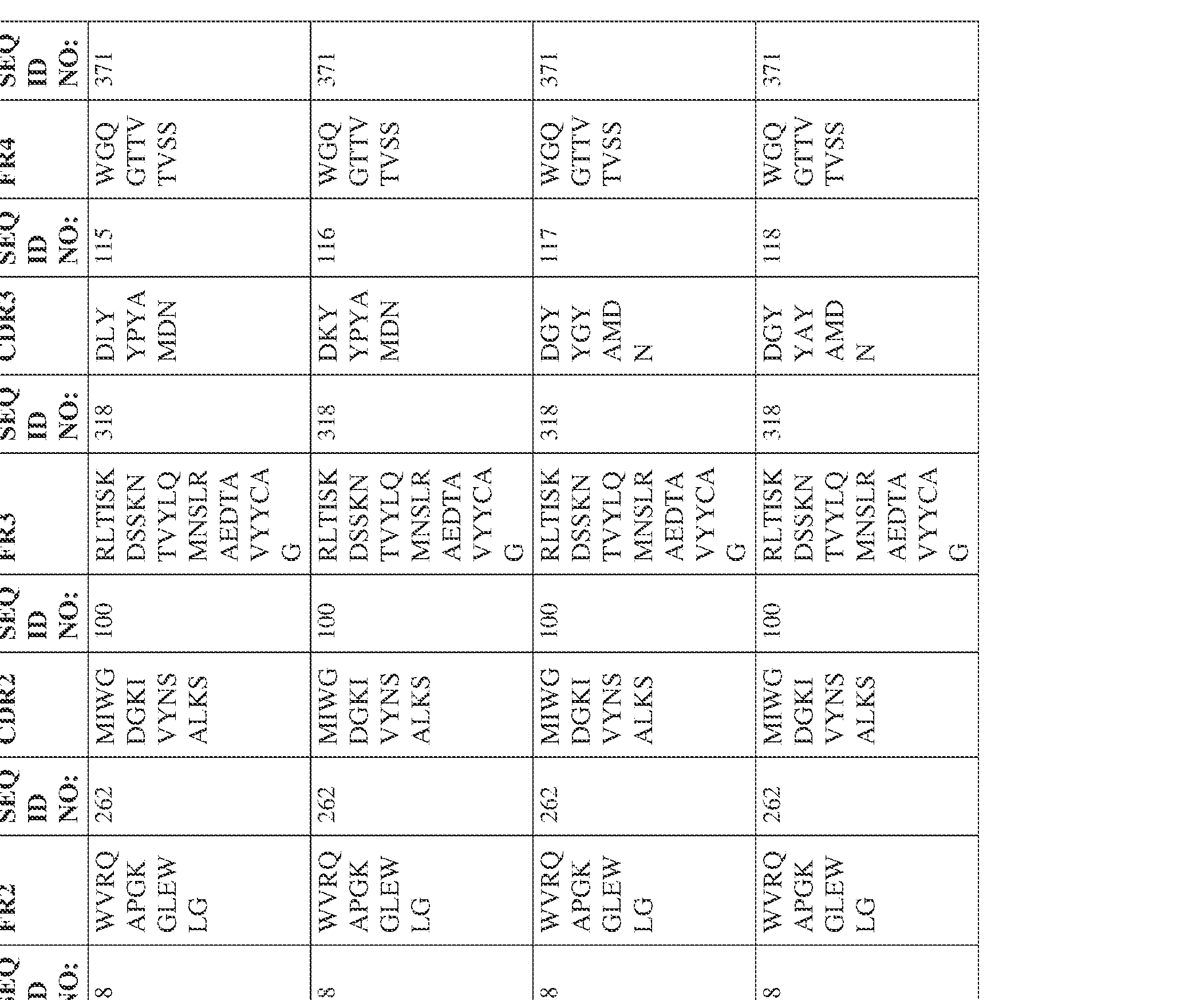
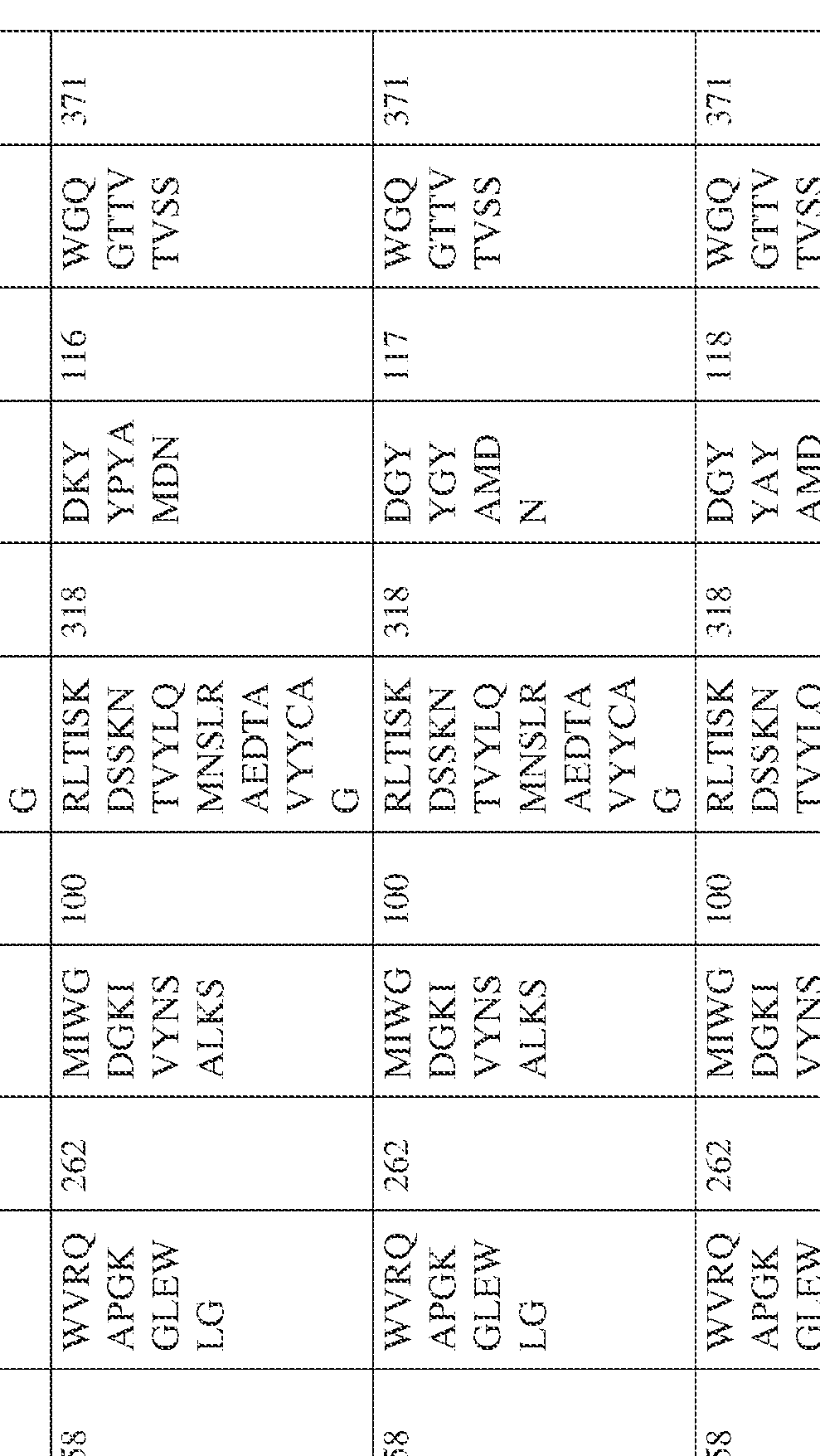
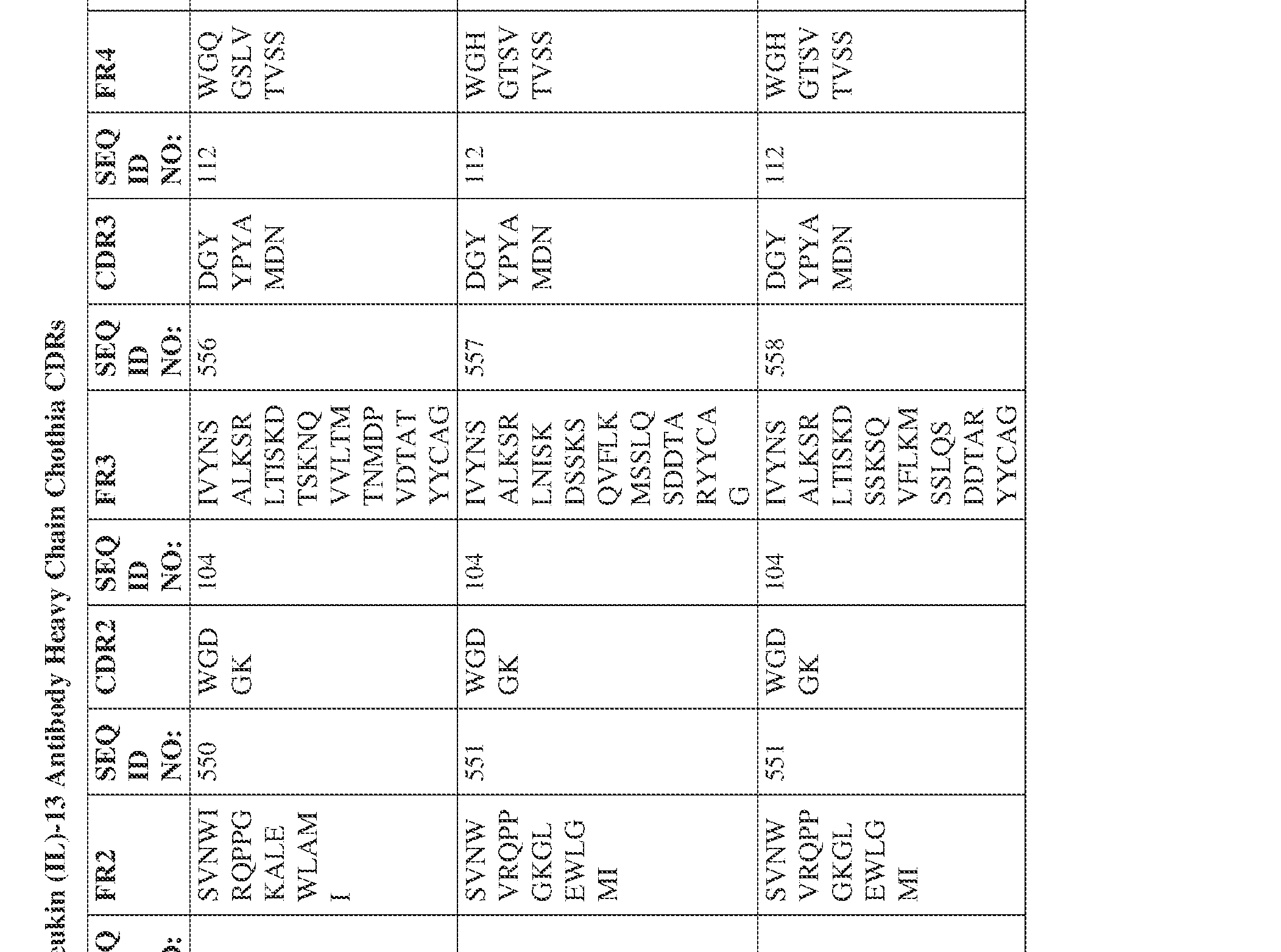
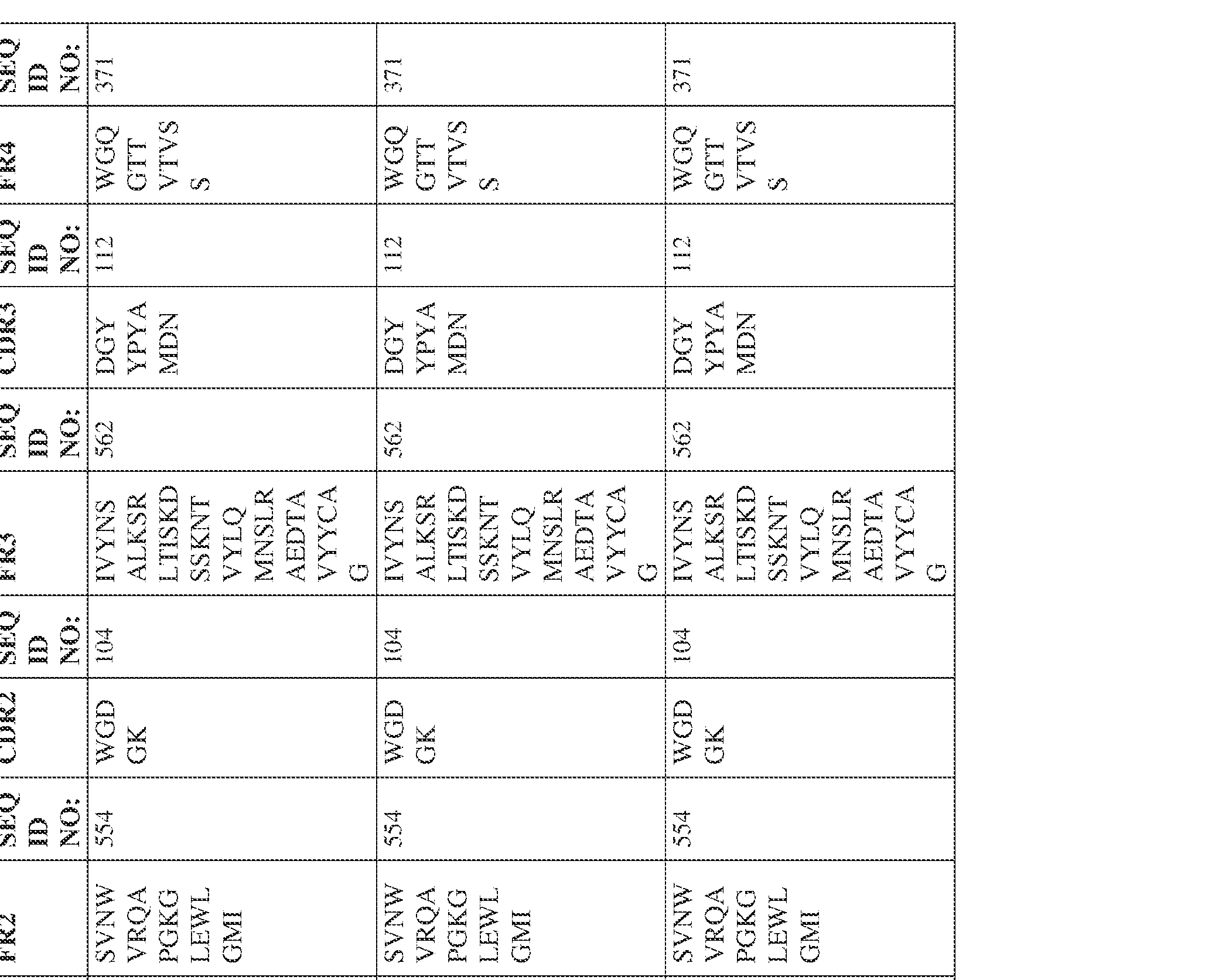
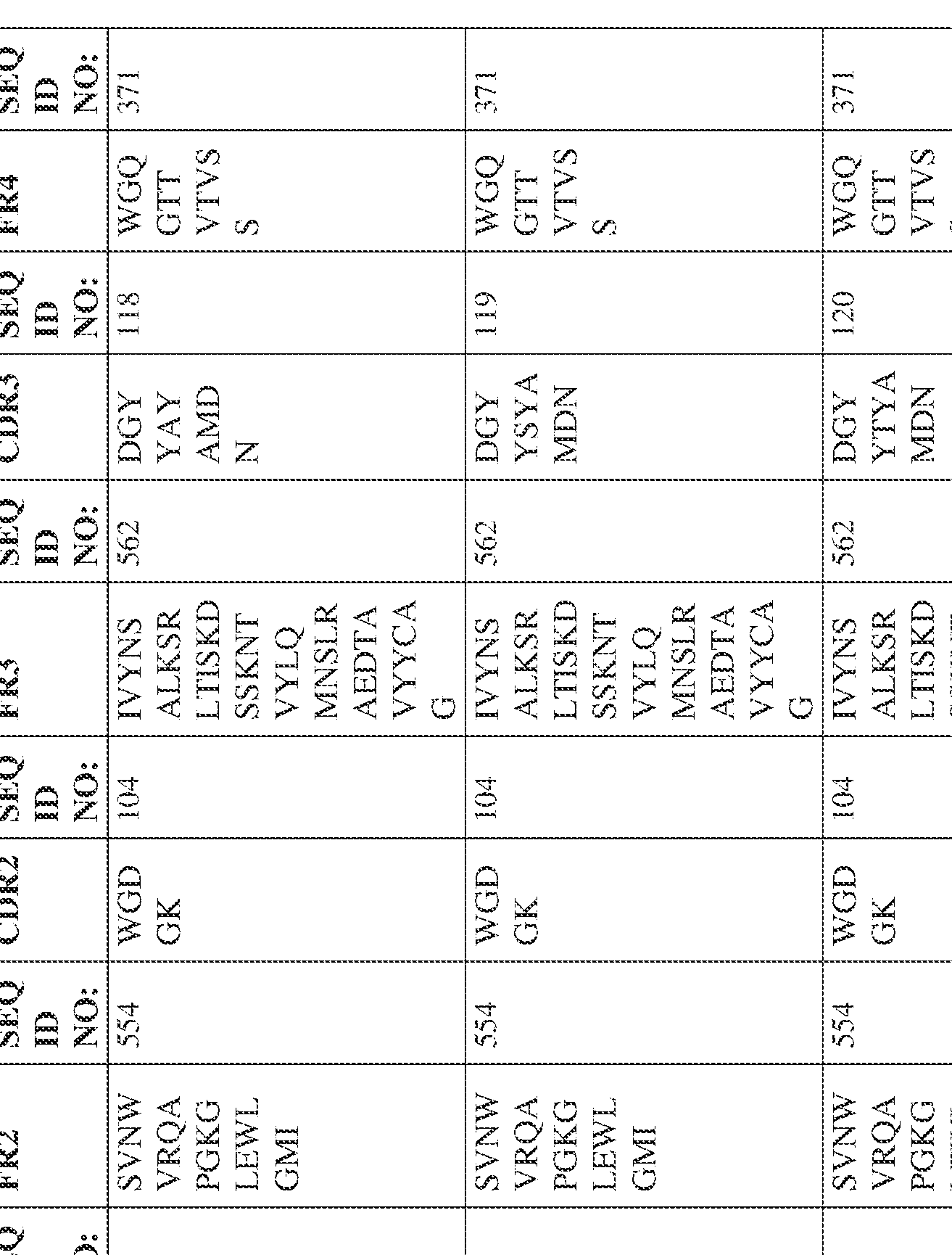
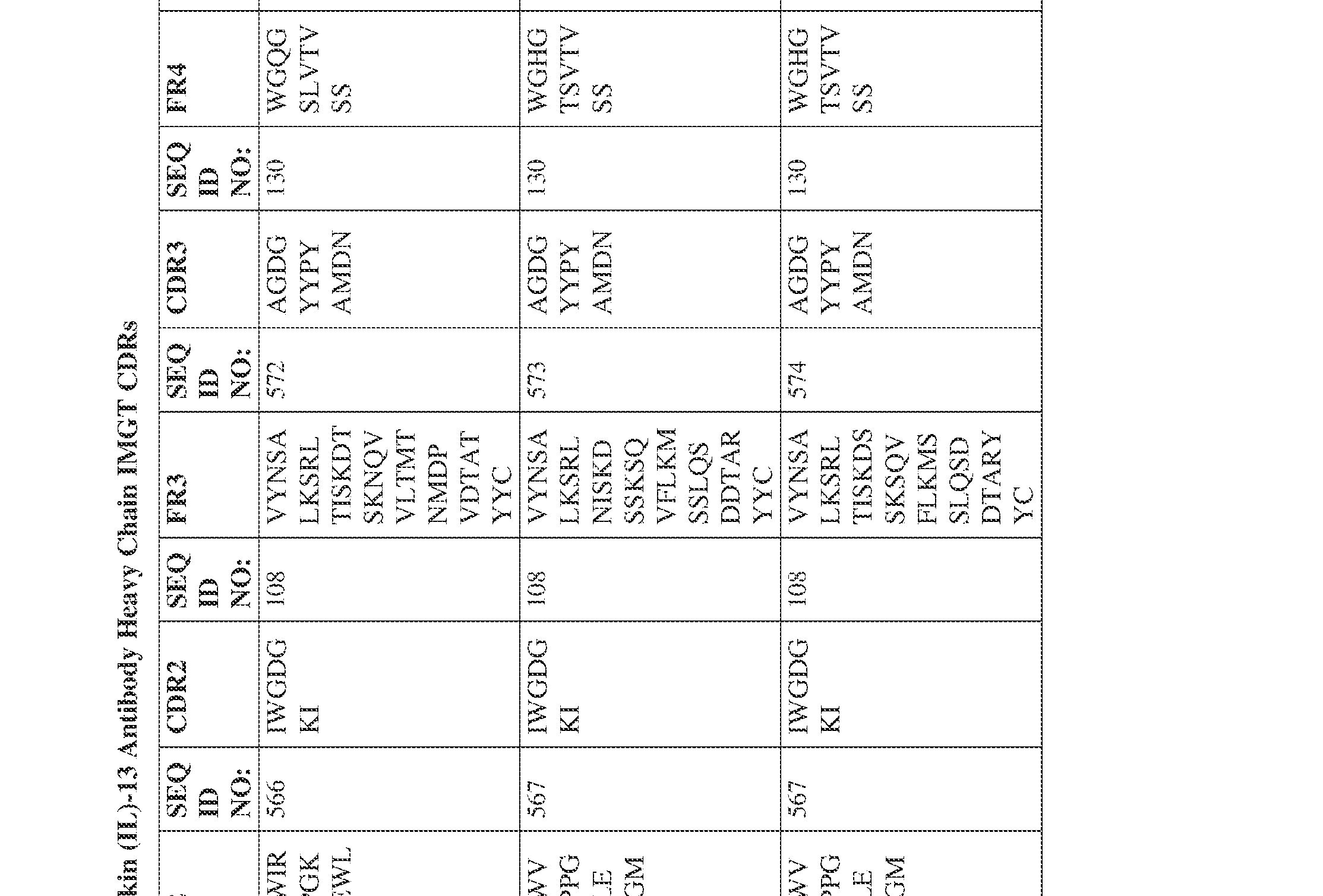
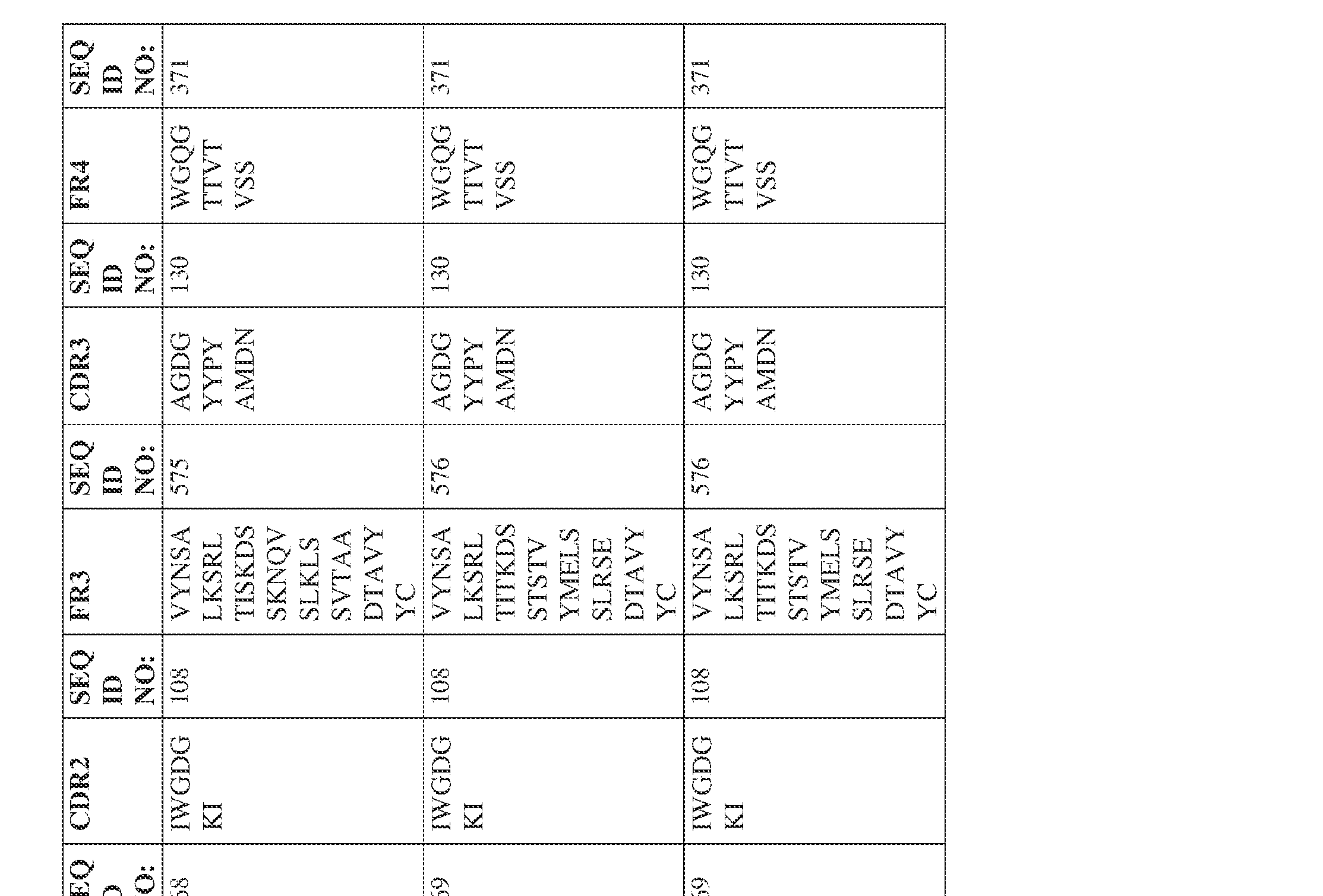
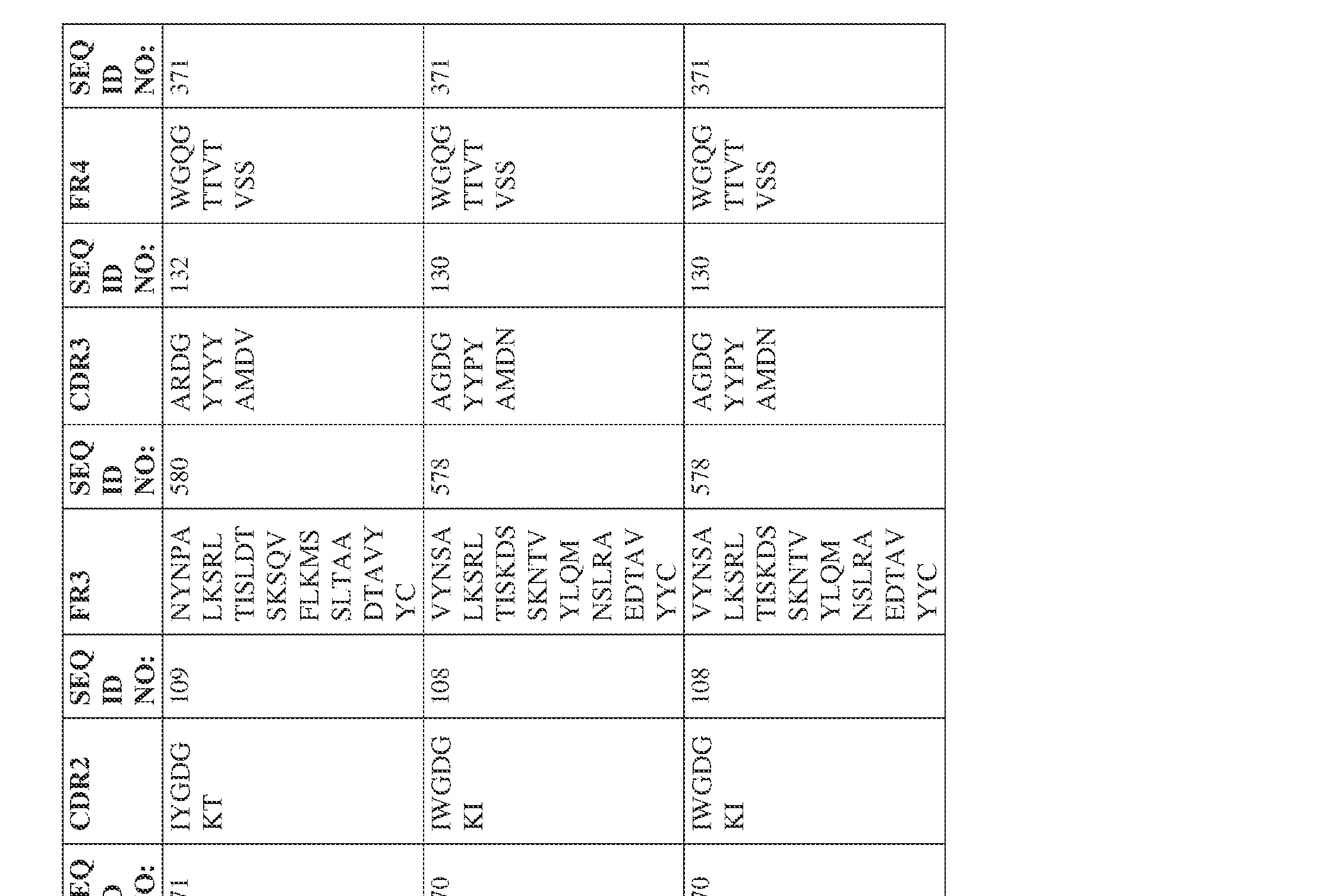
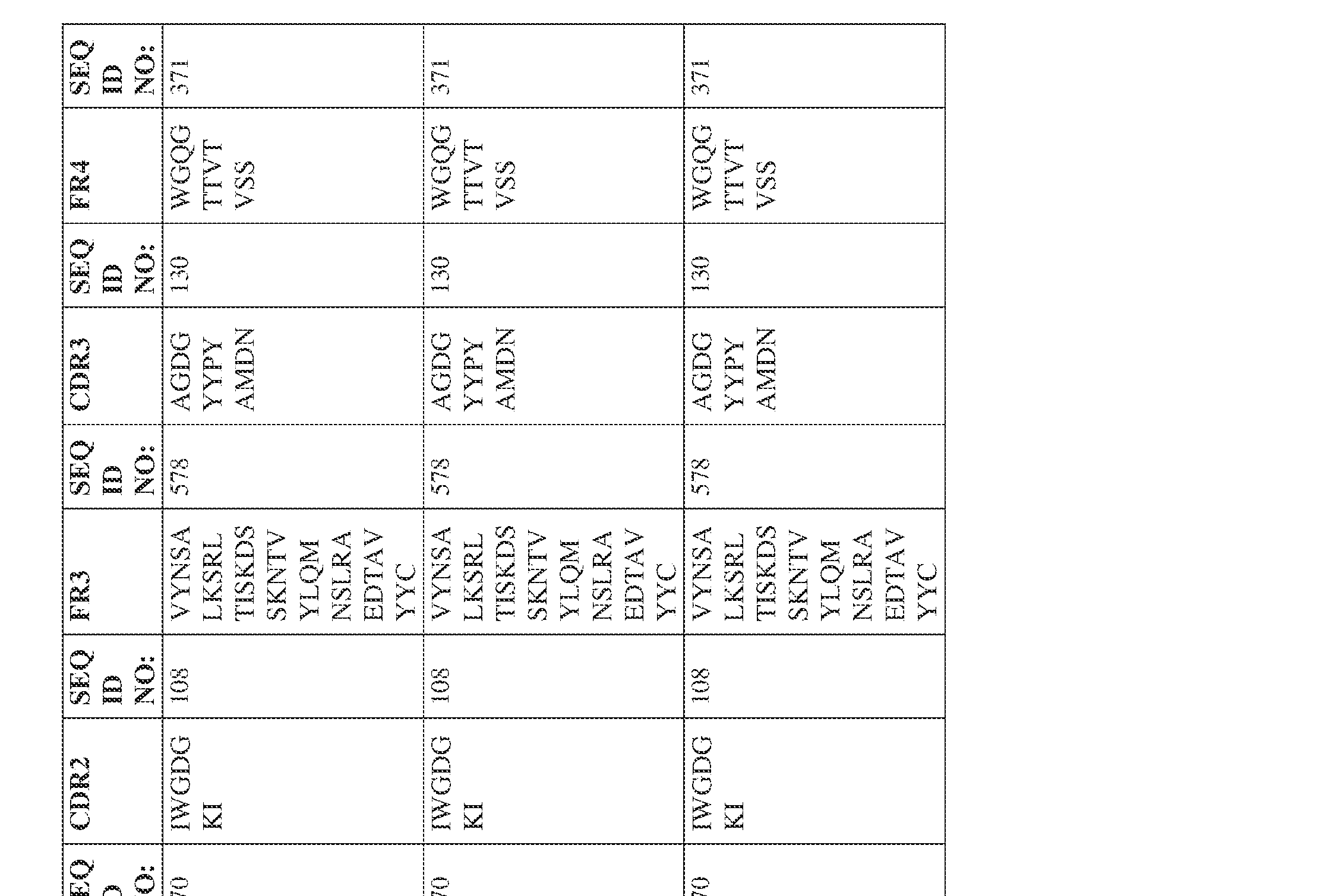
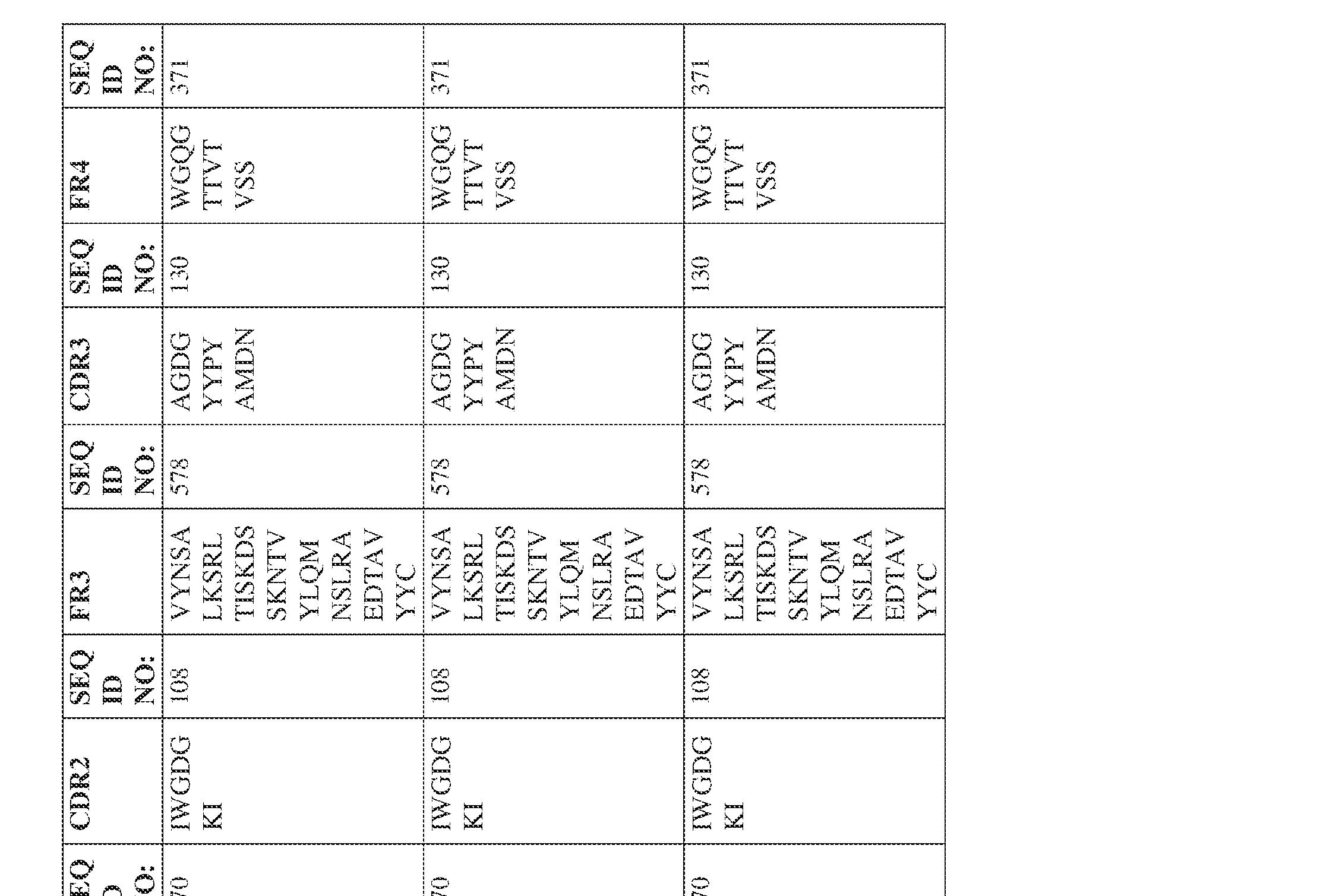
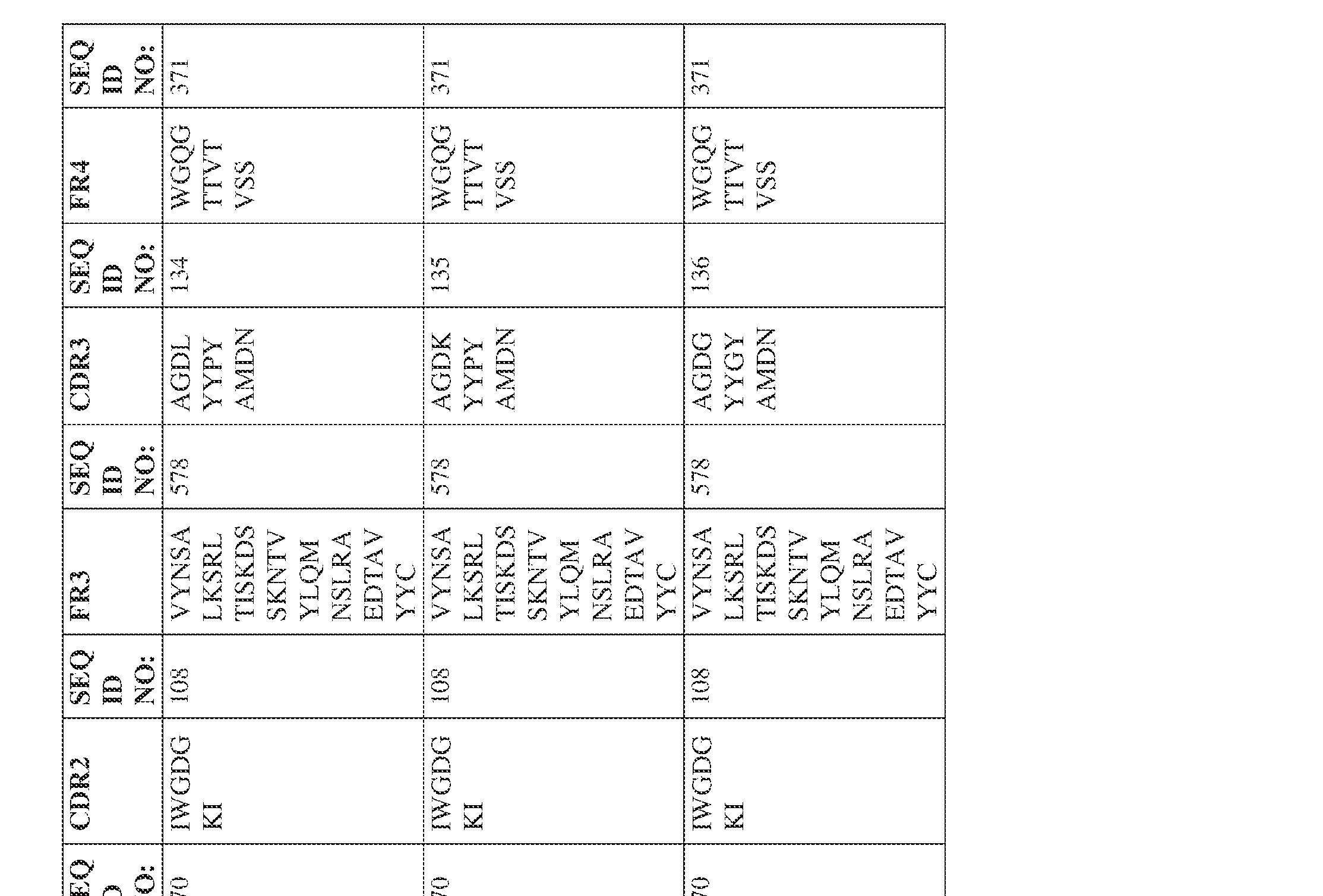
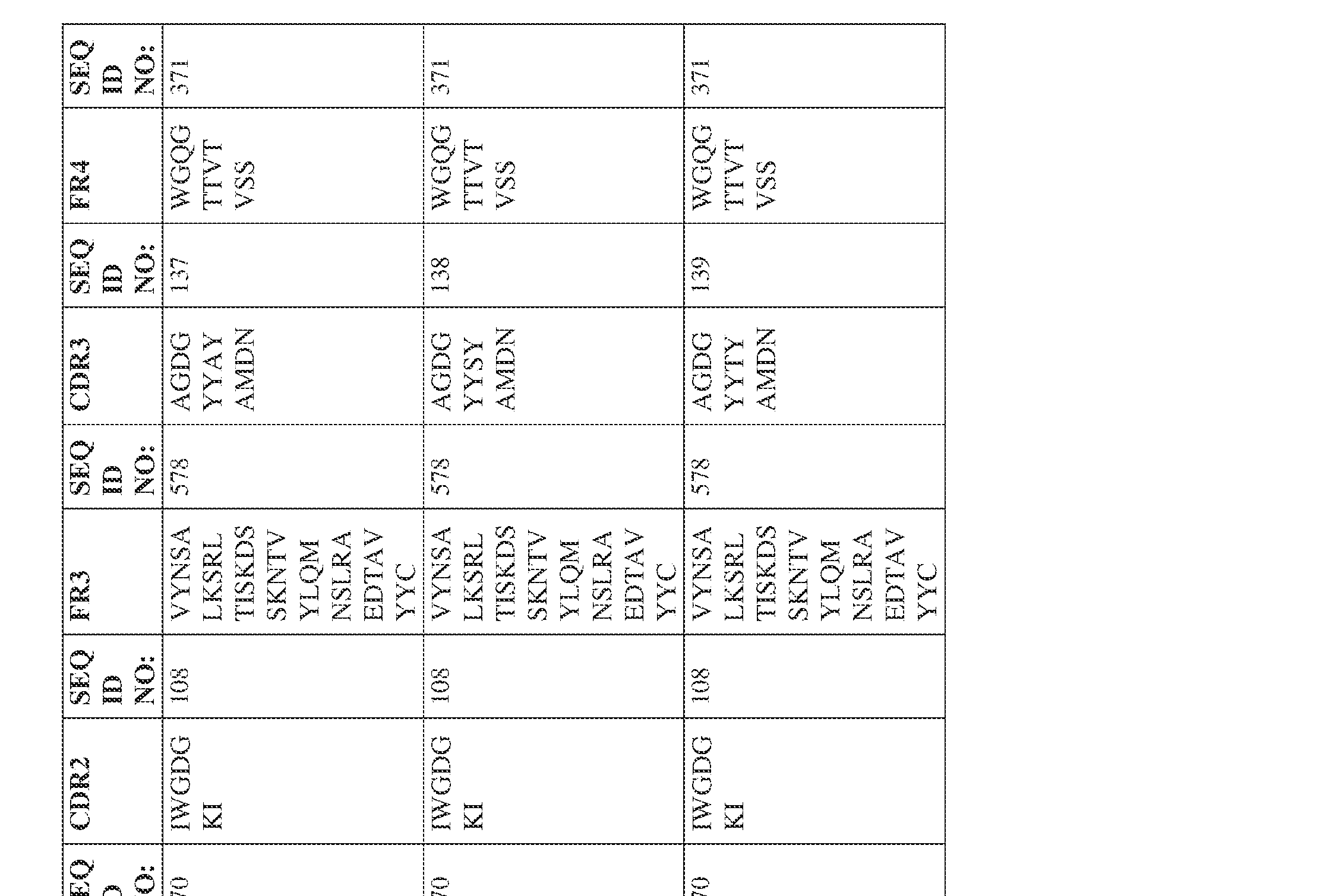
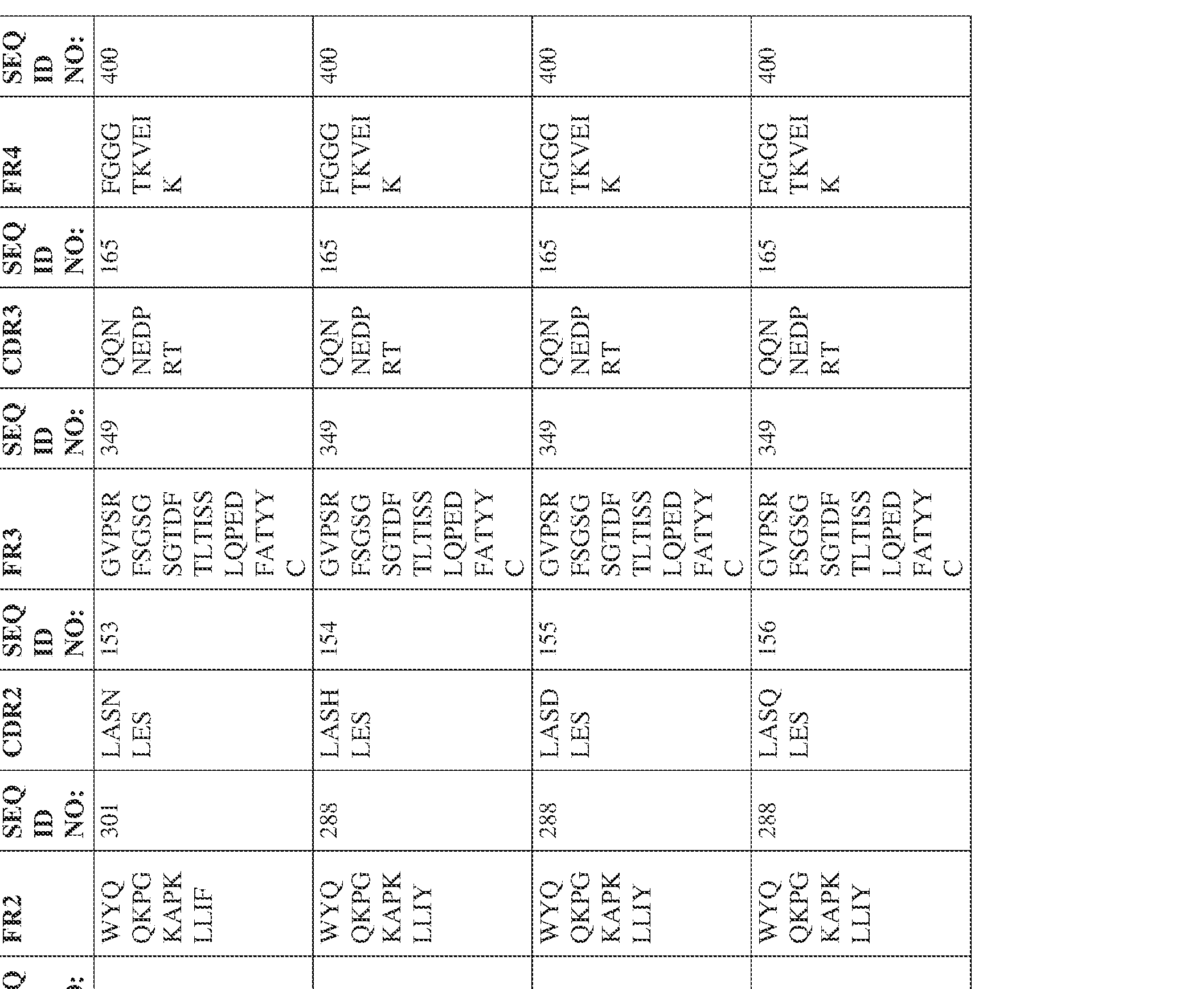
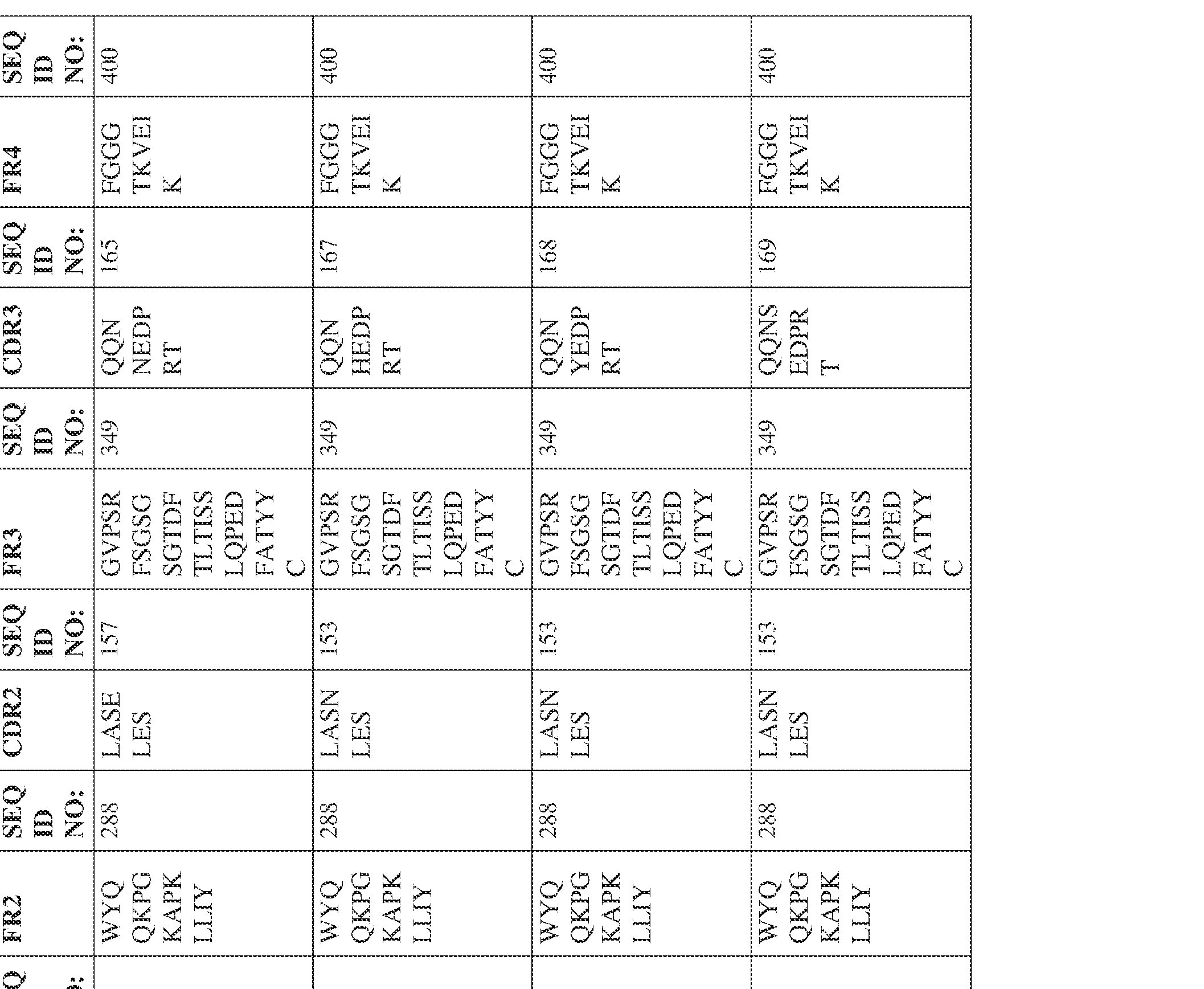
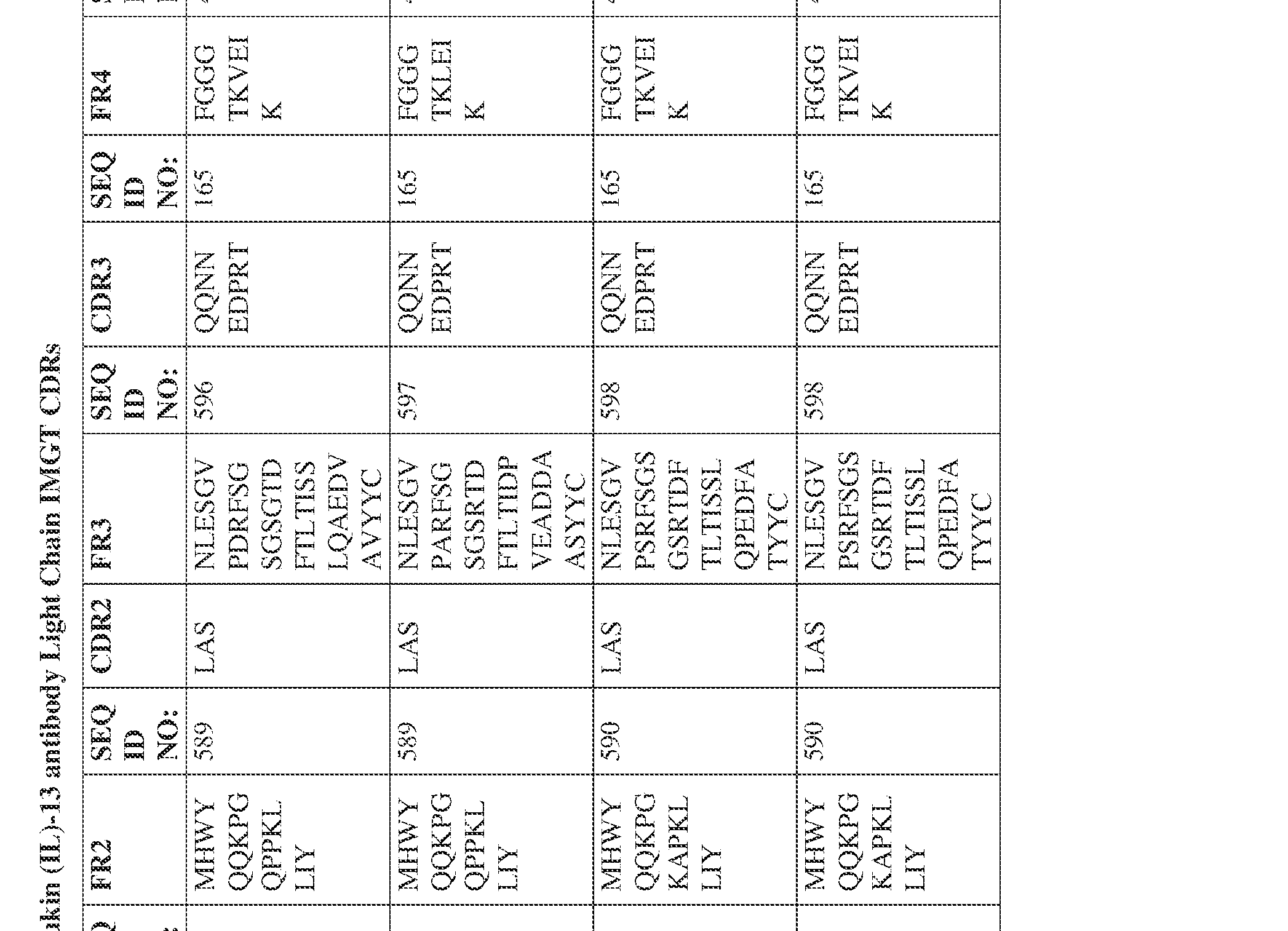
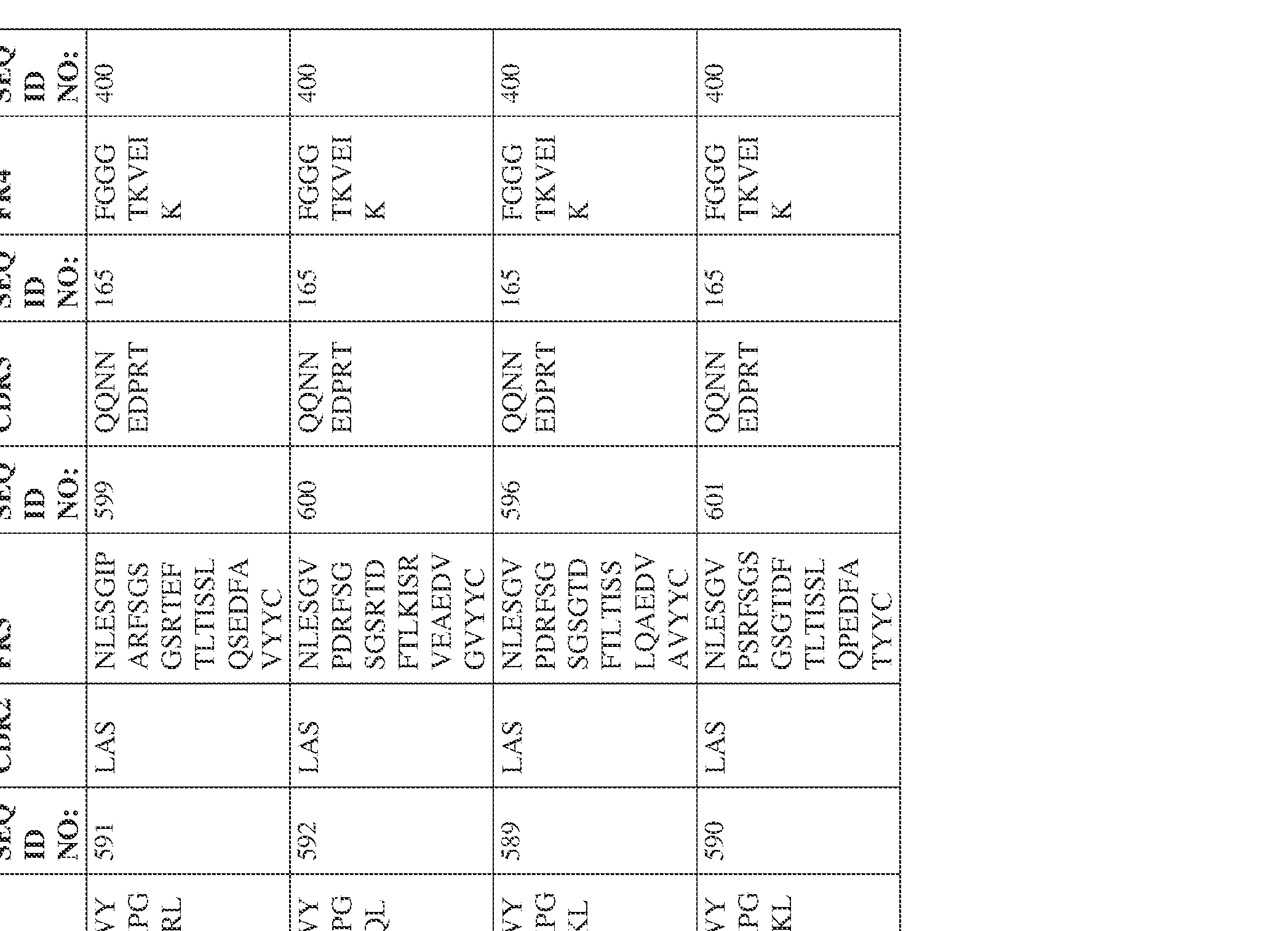
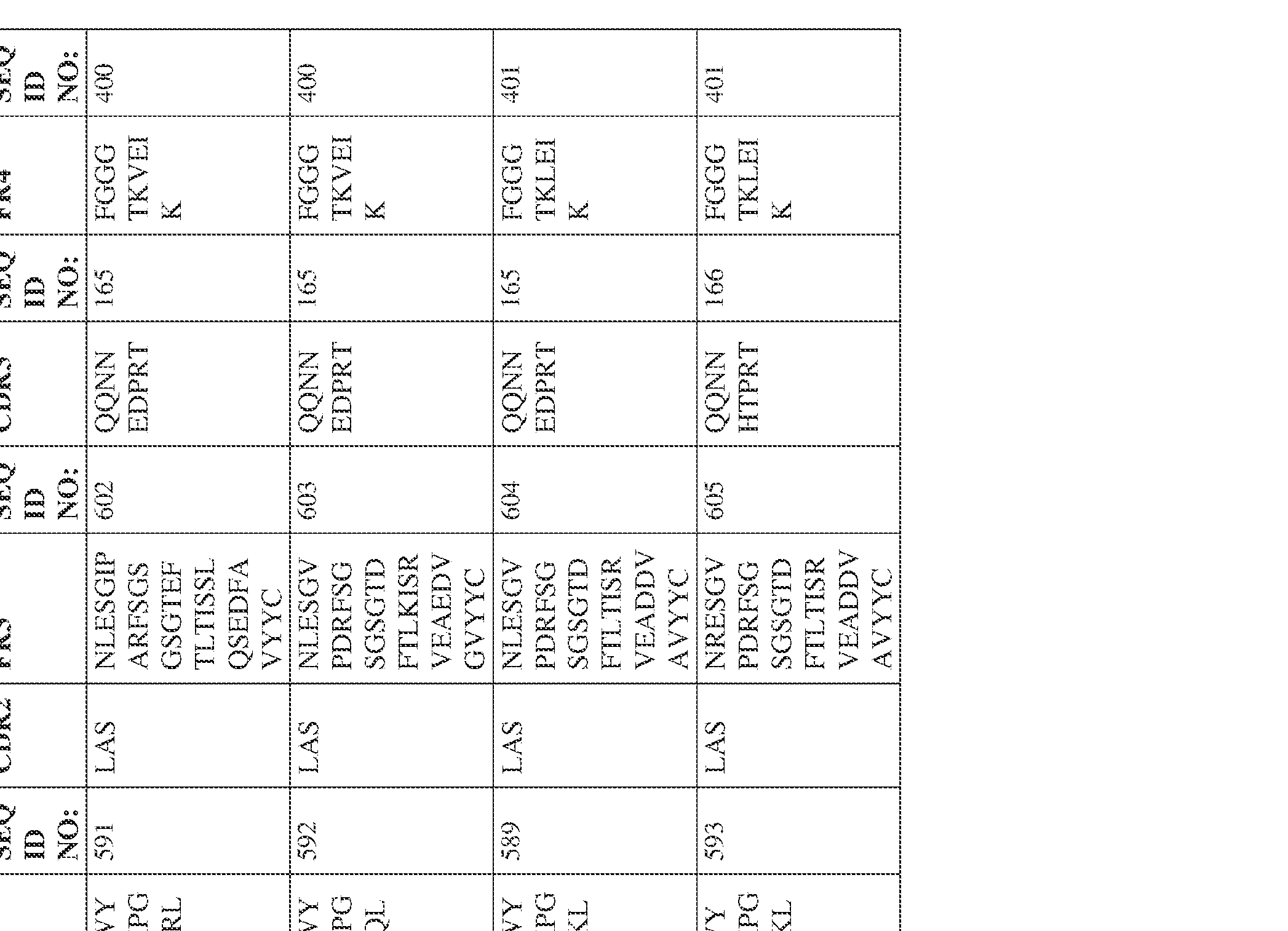
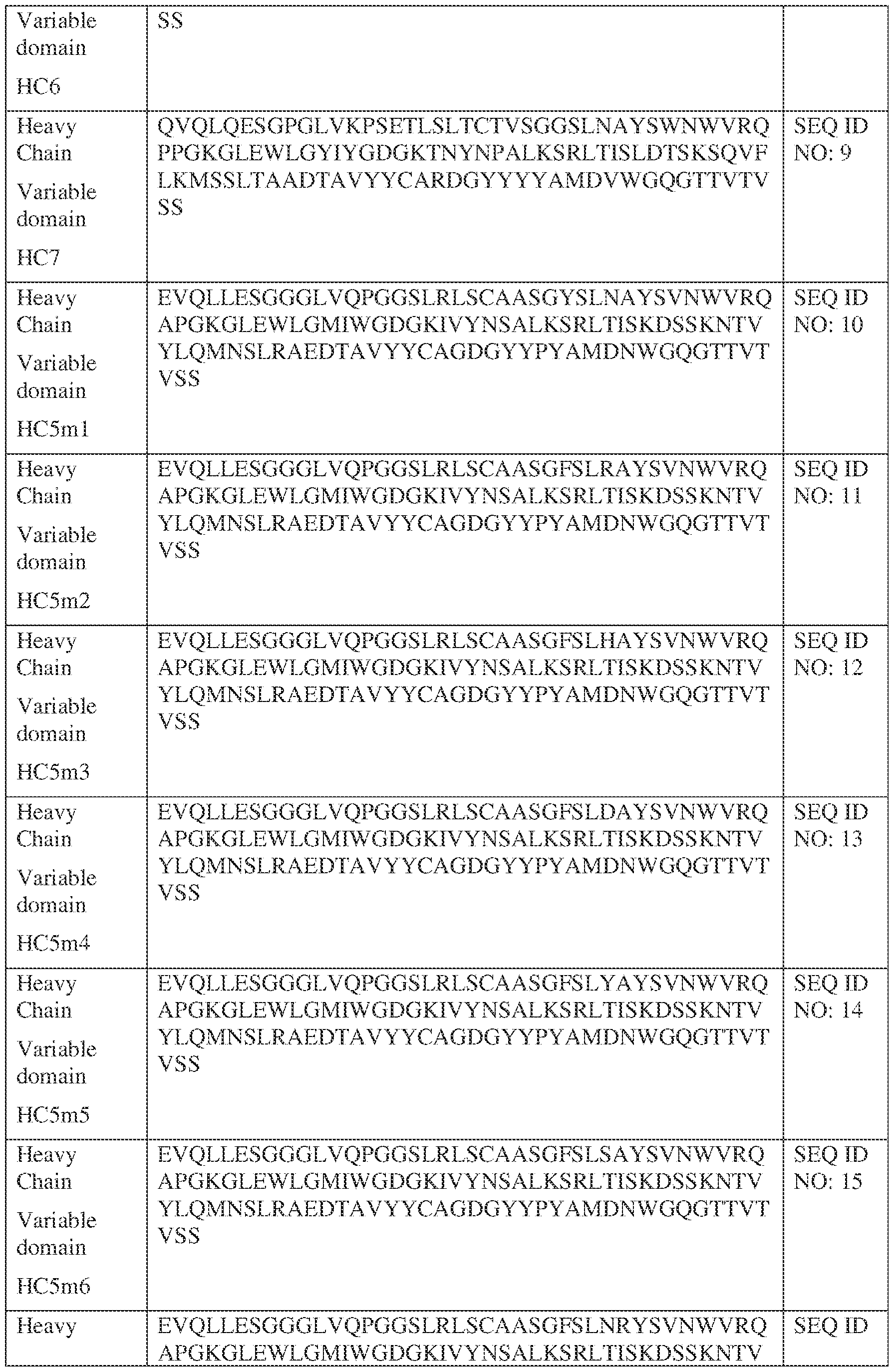
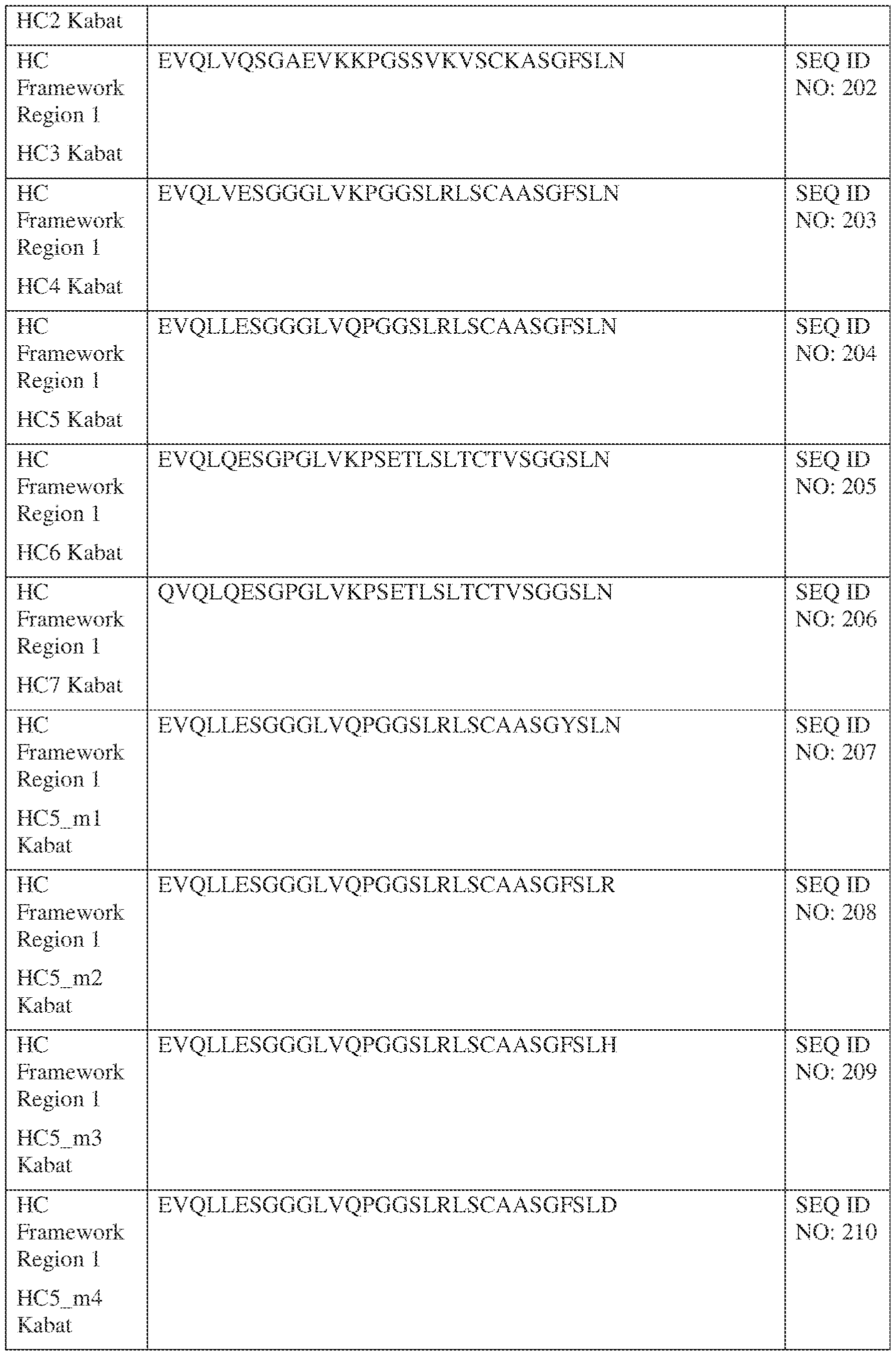
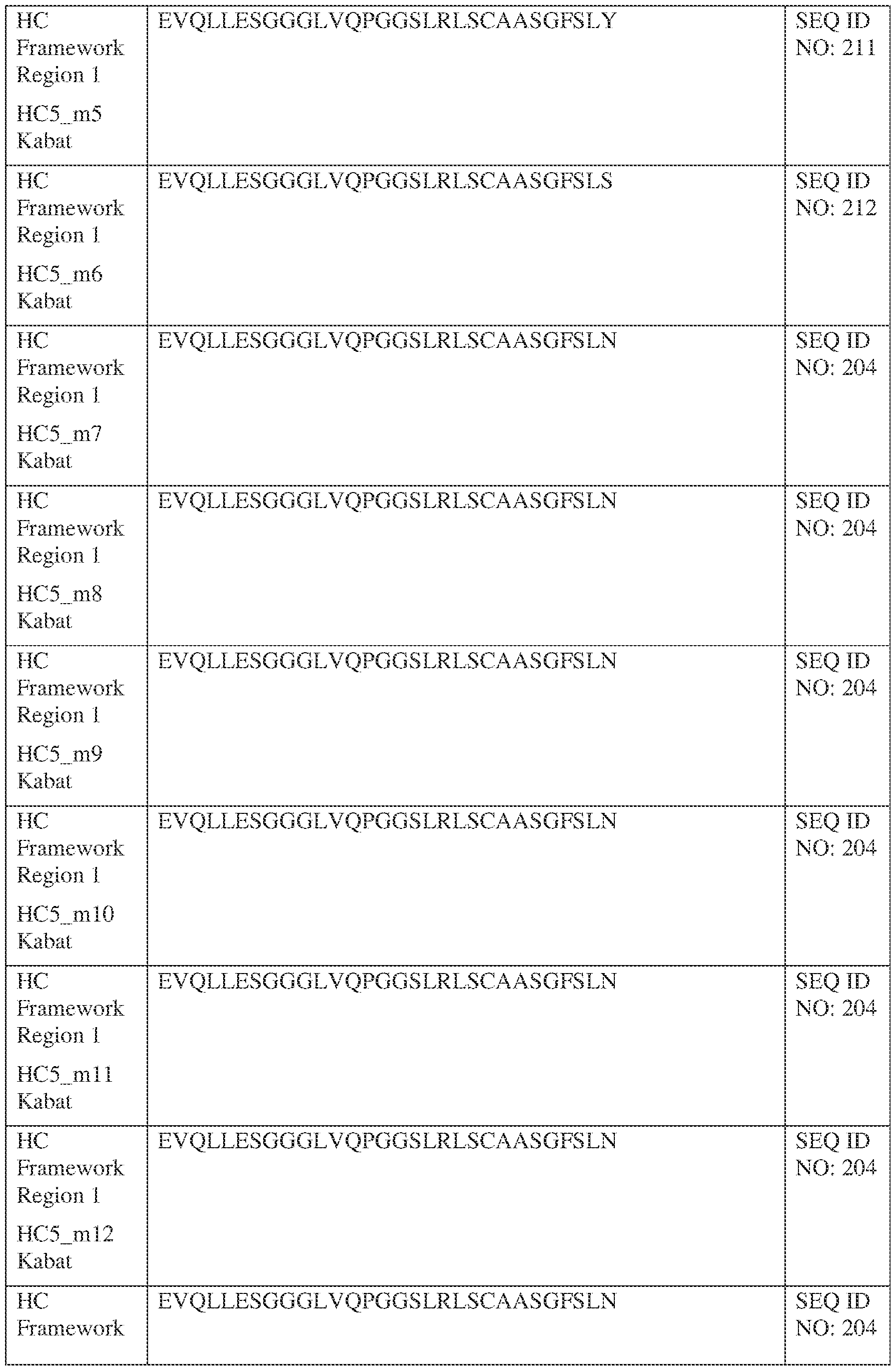
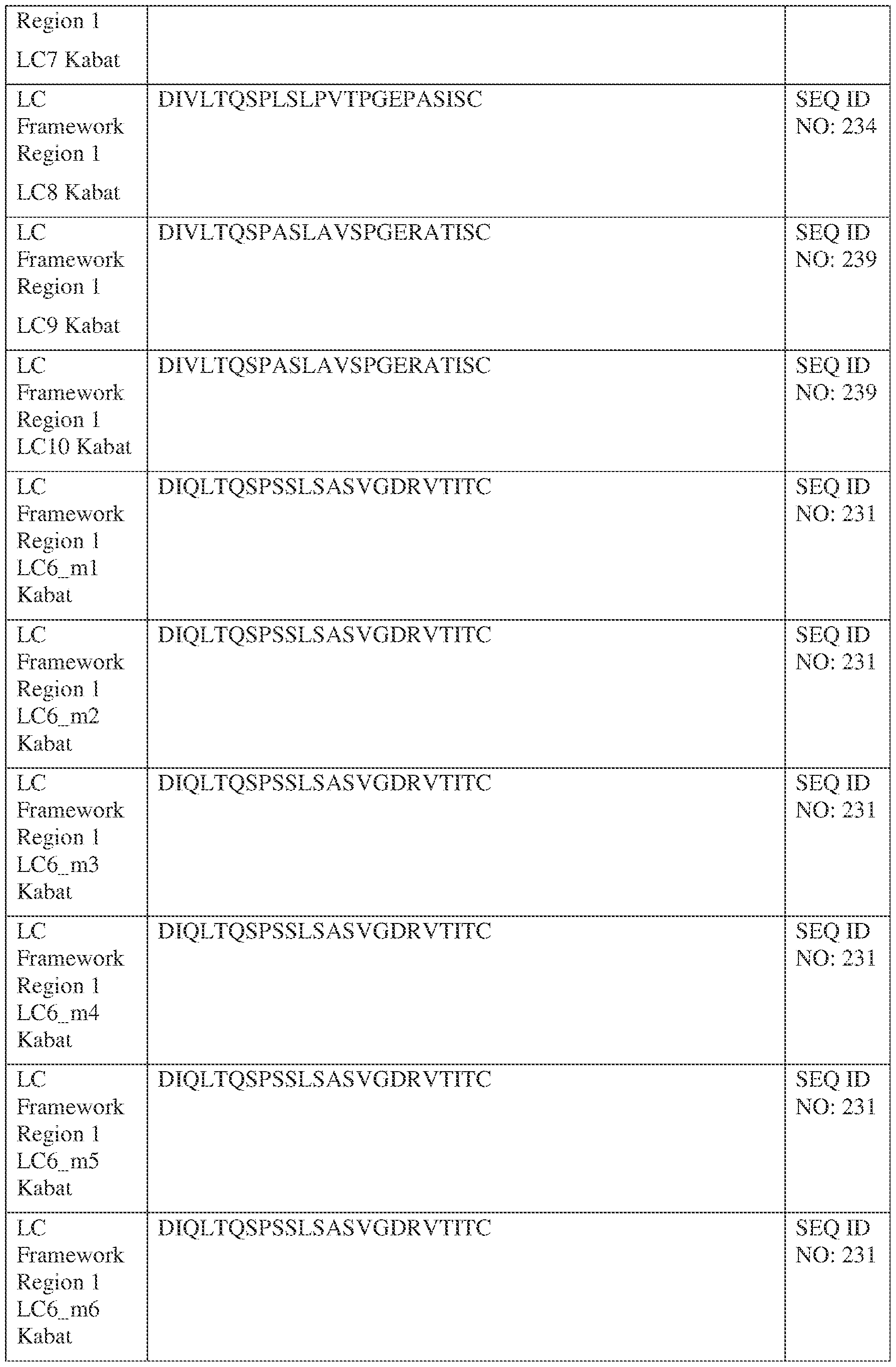
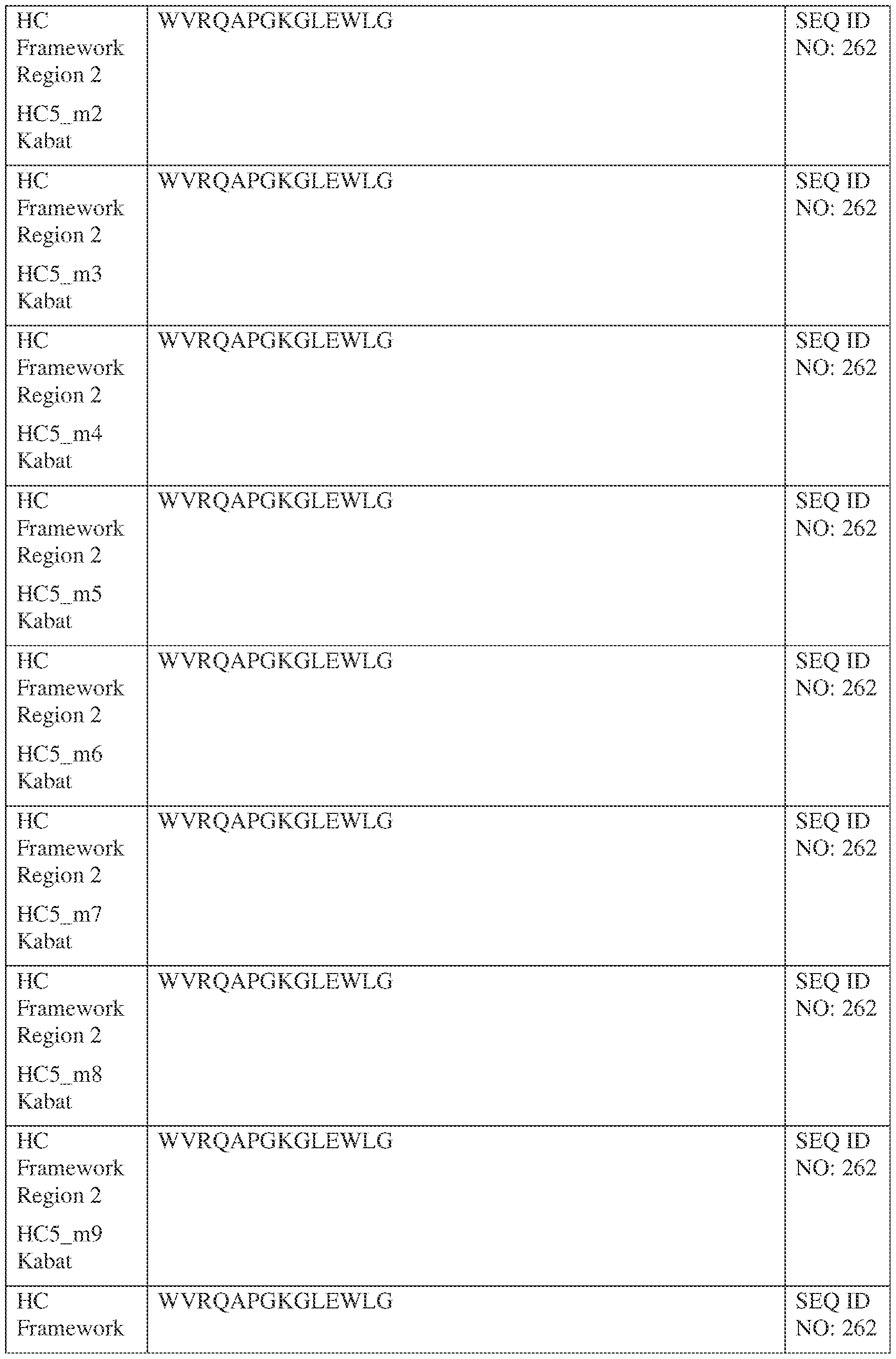
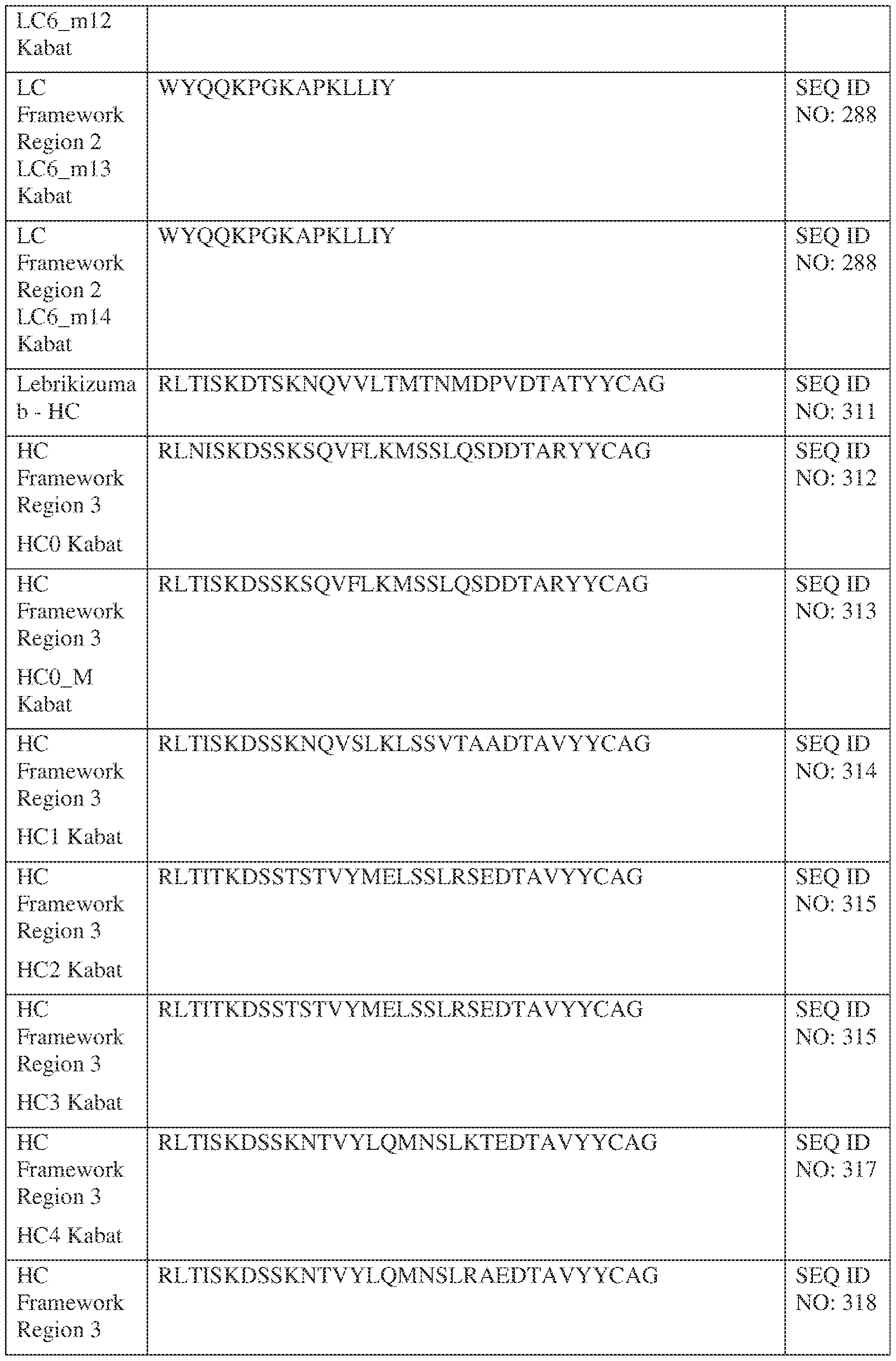
- the isolated bispecific antibody comprises a heavy chain variable domain comprising a framework region sequence selected from a sequence set forth in SEQ ID NOs: 198-229, 255-256, 258-259, 261-285, 311-315, 317-342, 368-369, 371-399, and 540-580.
- the isolated bispecific antibody comprises a heavy chain variable domain comprising 1, 2, 3, or 4 framework region sequences selected from a sequence set forth in SEQ ID NOs: 198-229, 255-256, 258-259, 261-285, 311-315, 317-342, 368-369, 371-399, and 540-580.
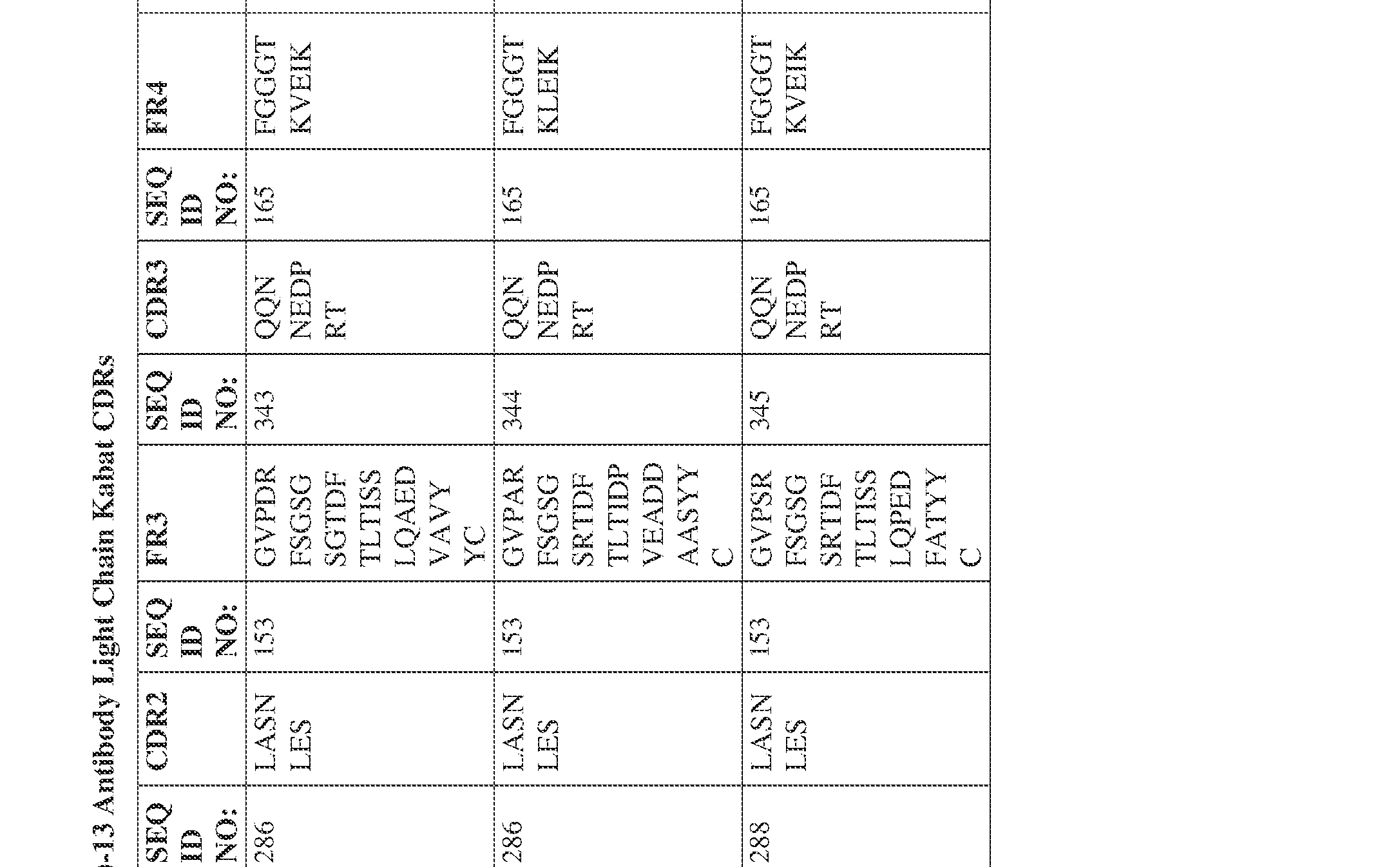
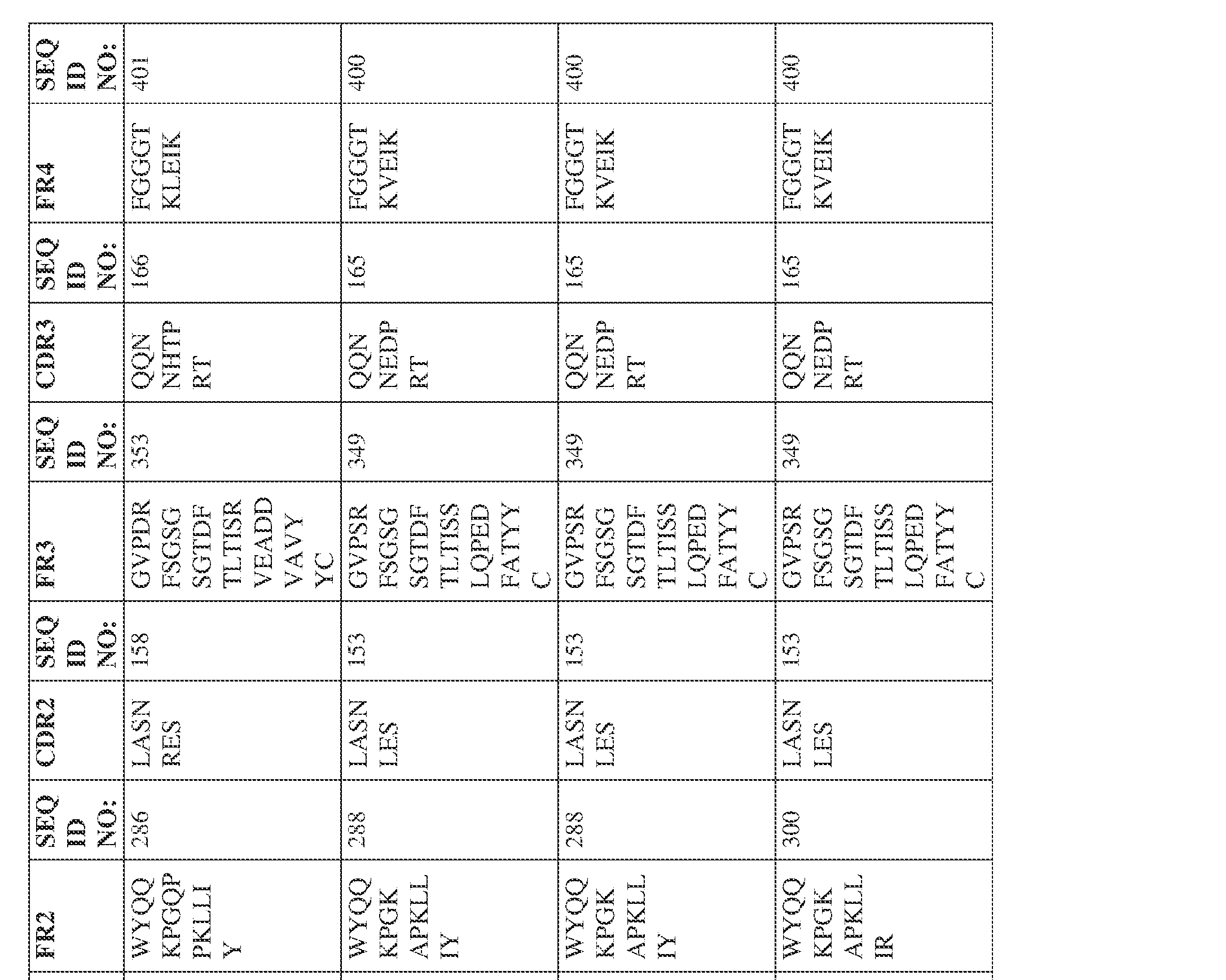
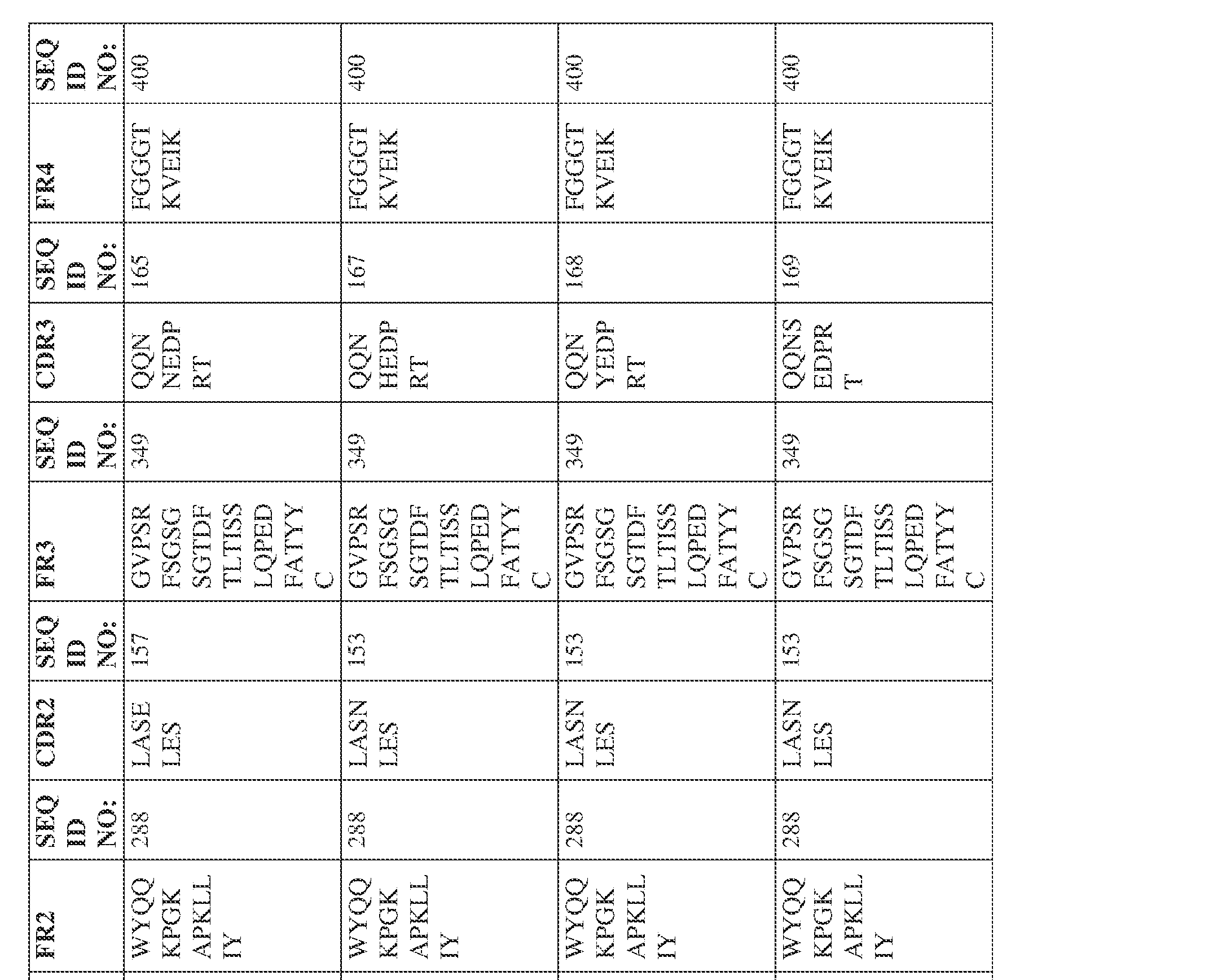
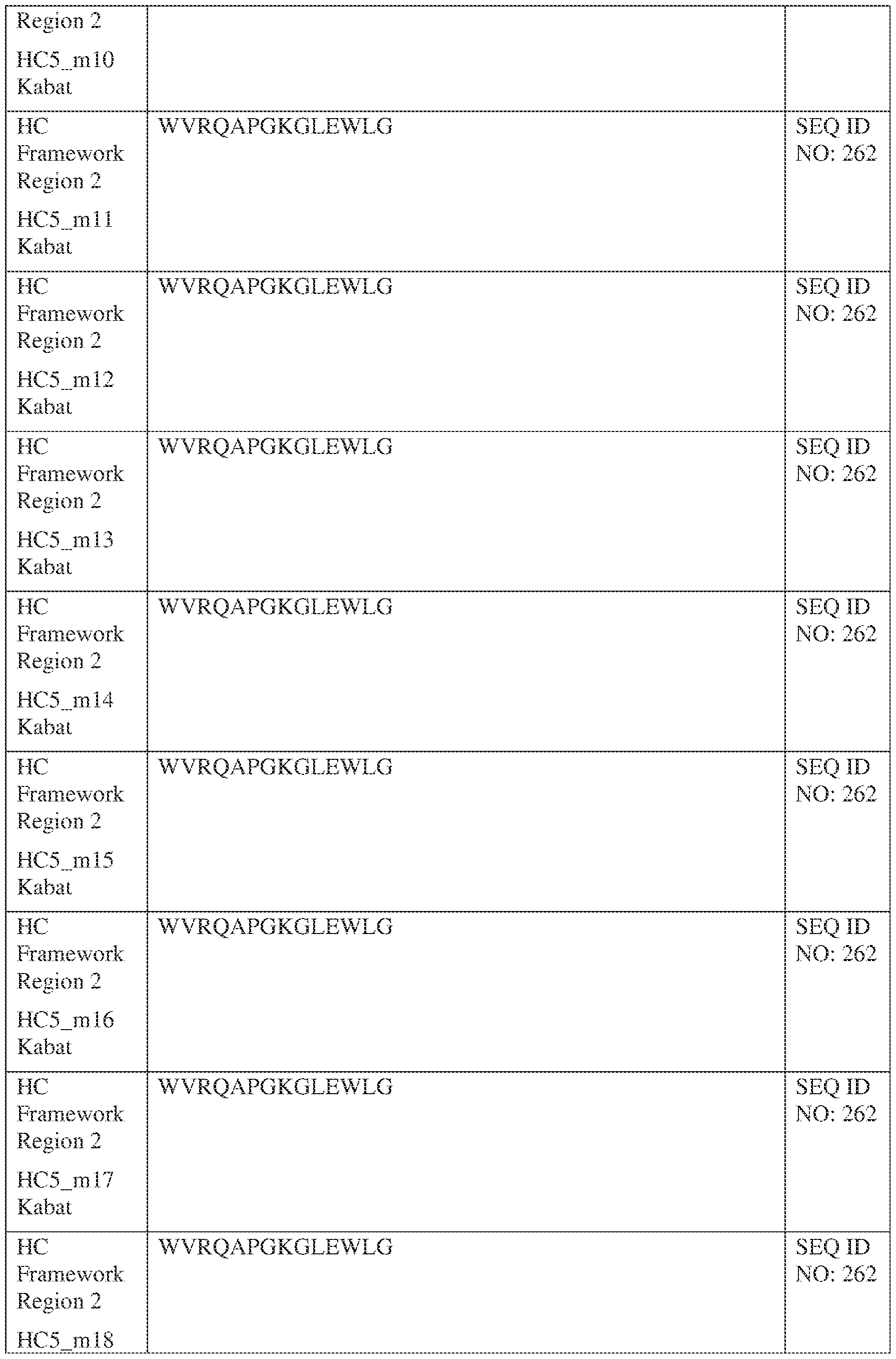
- the isolated bispecific antibody comprises a light chain variable domain comprising a framework region sequence selected from a sequence set forth in SEQ ID NOs: 230-231, 233-235, 239, 241-254, 286, 288, 290-291, 293, 296-310, 343-345, 347, 400-424, and 581-609.
- the isolated bispecific antibody comprises a light chain variable domain comprising 1, 2, 3, or 4 framework region sequences selected from a sequence set forth in SEQ ID NOs: 230-231, 233-235, 239, 241-254, 286, 288, 290-291, 293, 296-310, 343-345, 347, 400-424, and 581-609.
- the isolated bispecific antibody comprises a heavy chain variable domain comprising 1, 2, 3, or 4 framework region sequences selected from a sequence set forth in SEQ ID NOs: 198-229, 255-256, 258-259, 261-285, 311-315, 317-342, 368-369, 371-399, and 540-580, and comprises a light chain variable domain comprising 1, 2, 3, or 4 framework region sequences selected from a sequence set forth in SEQ ID NOs: 230- 231, 233-235, 239, 241-254, 286, 288, 290-291, 293, 296-310, 343-345, 347, 400-424, and 581-609. Table 2.
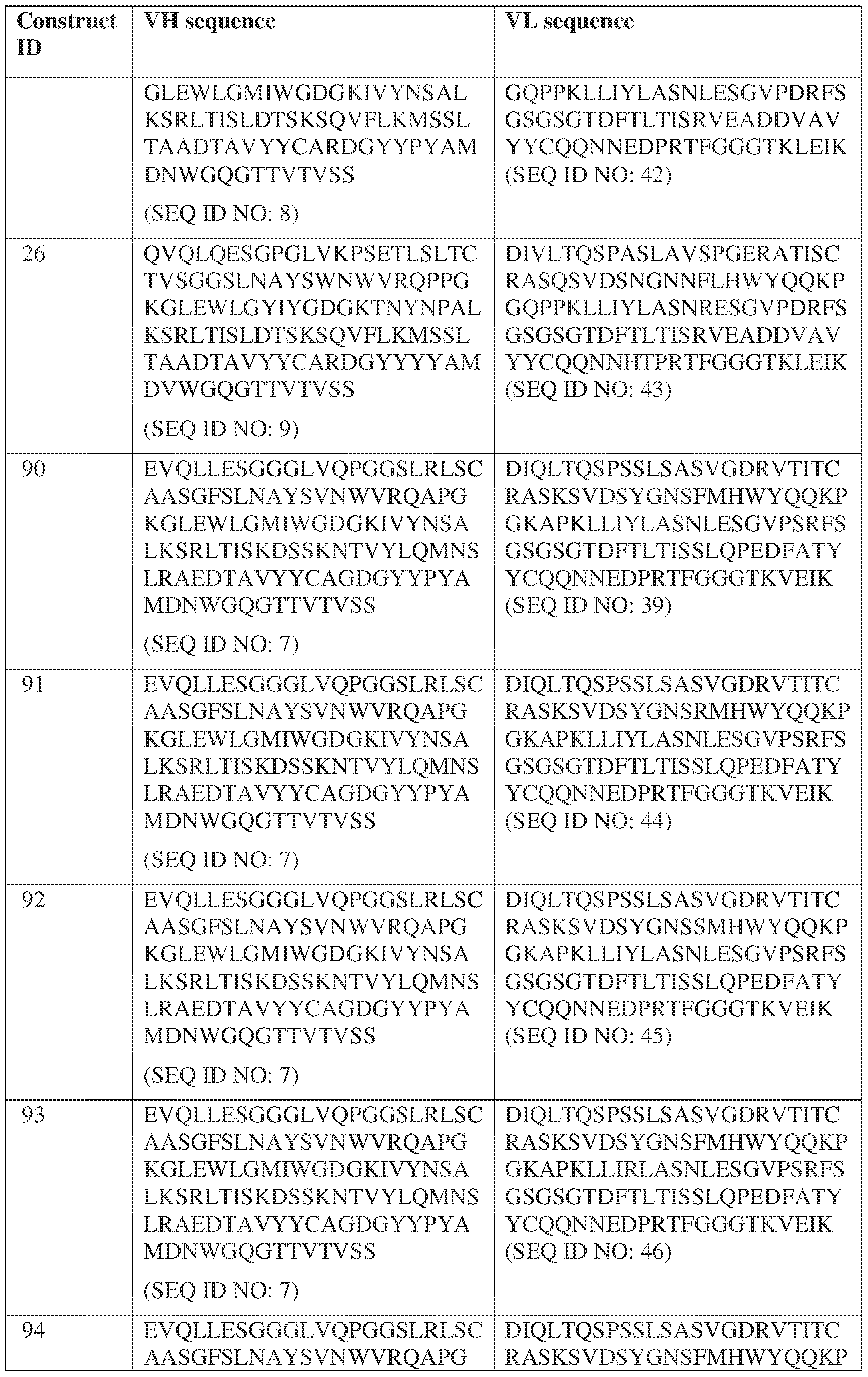
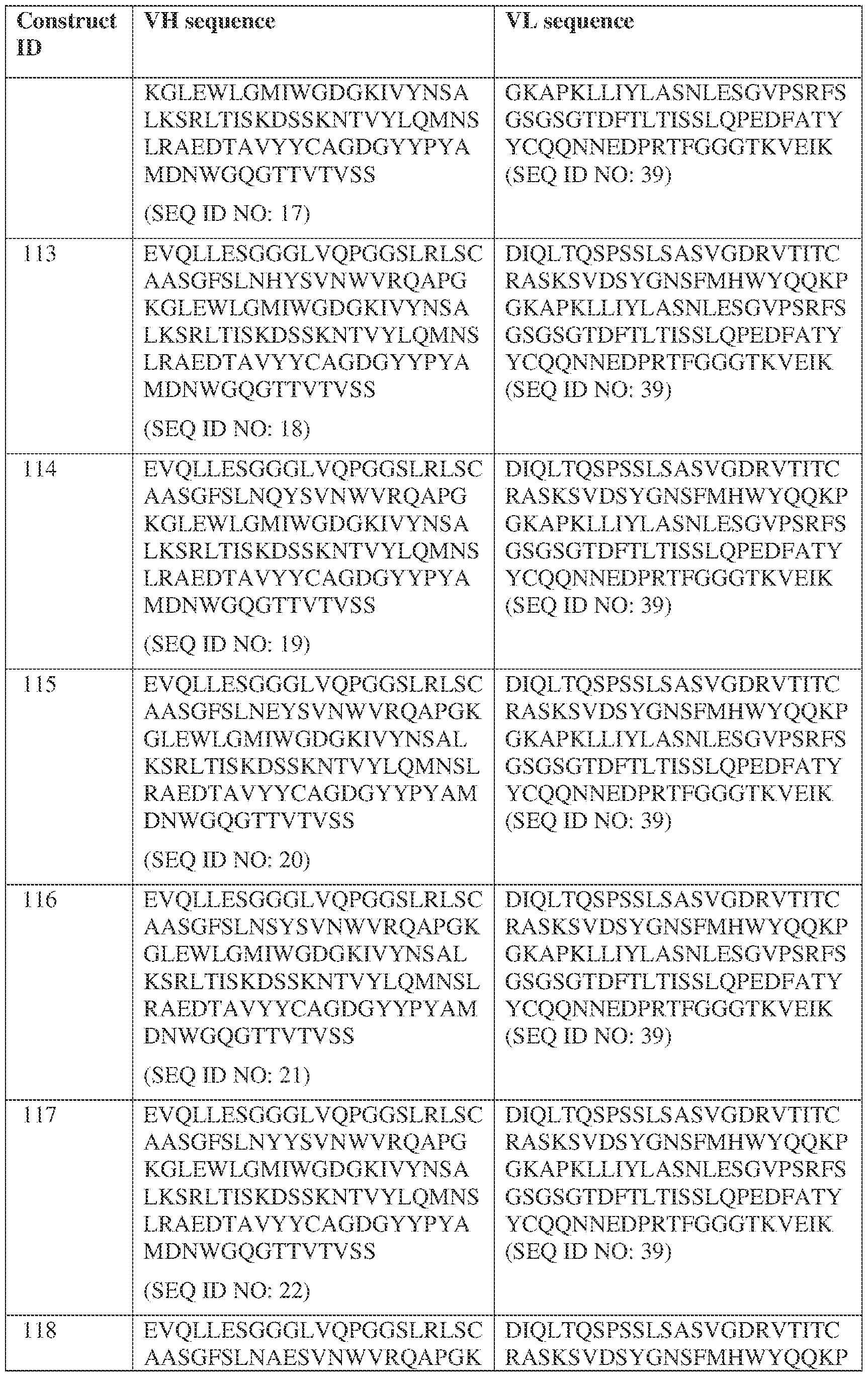
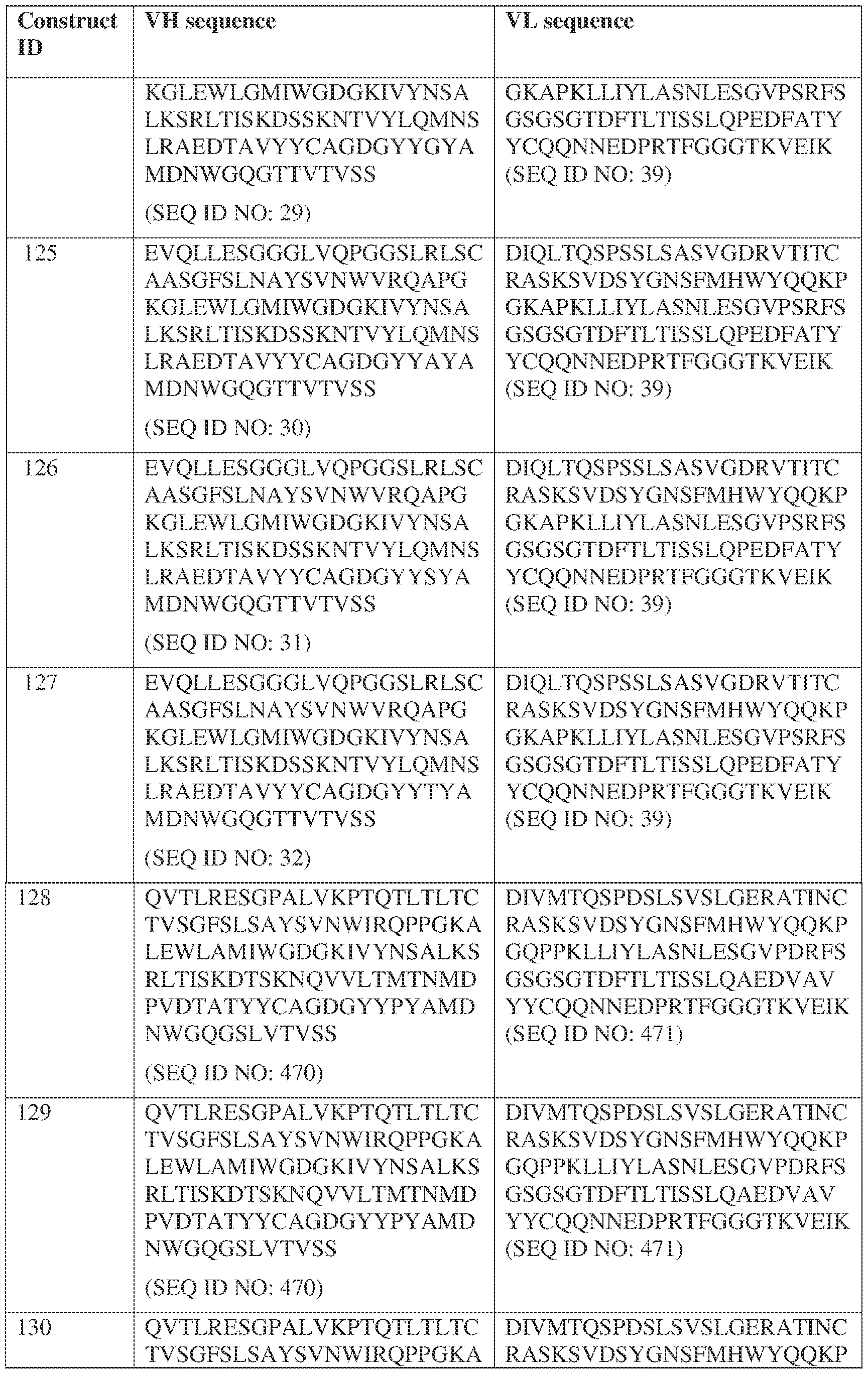
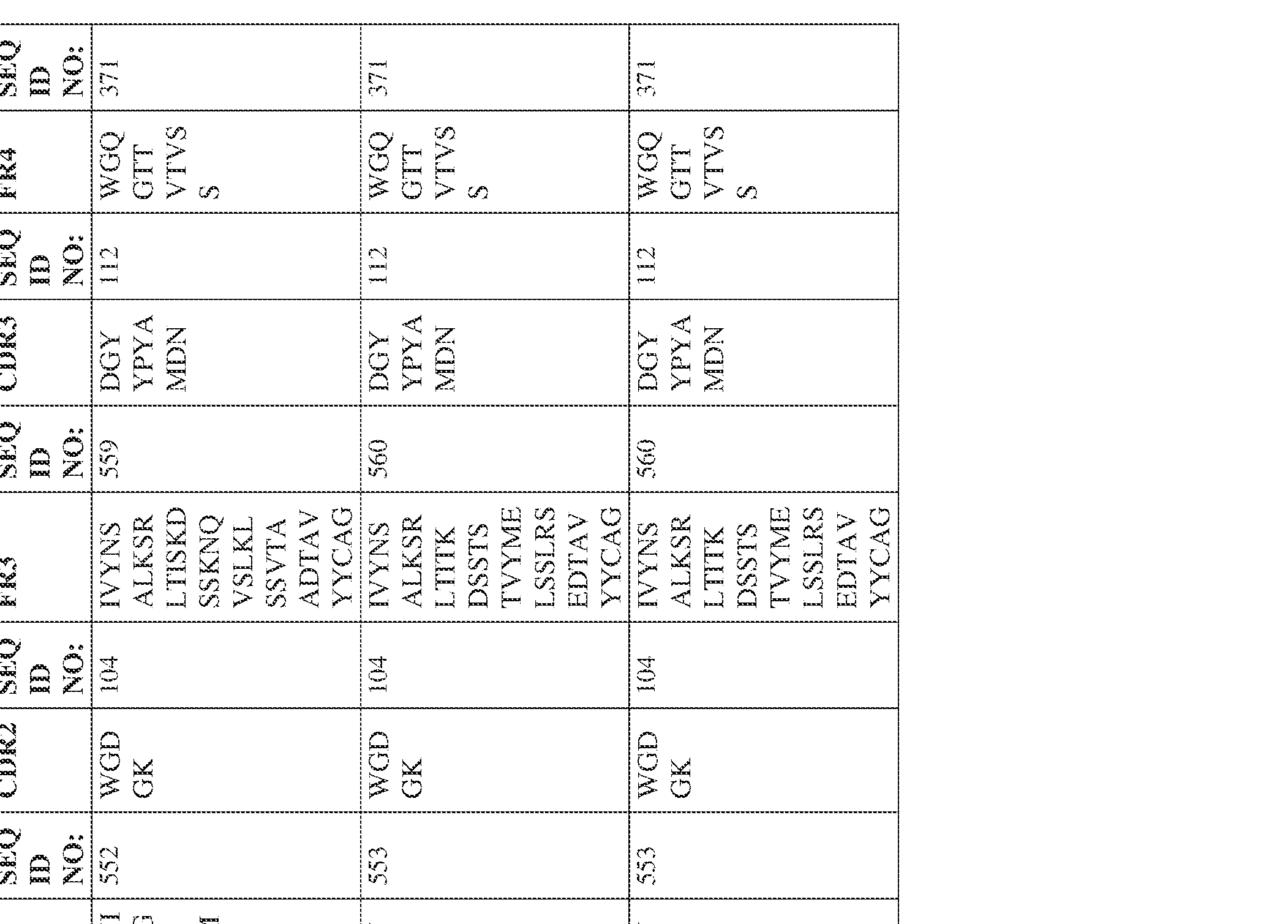
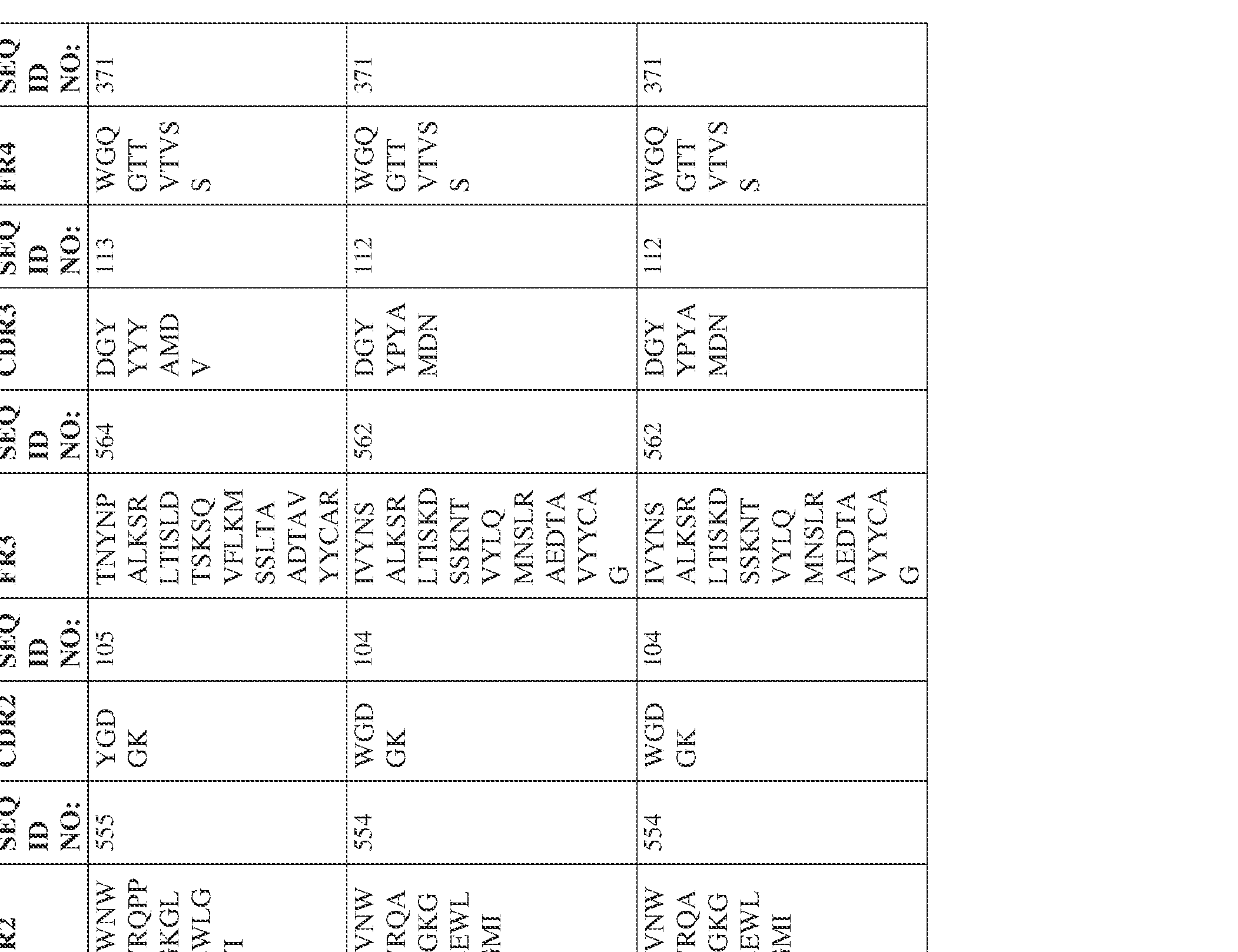
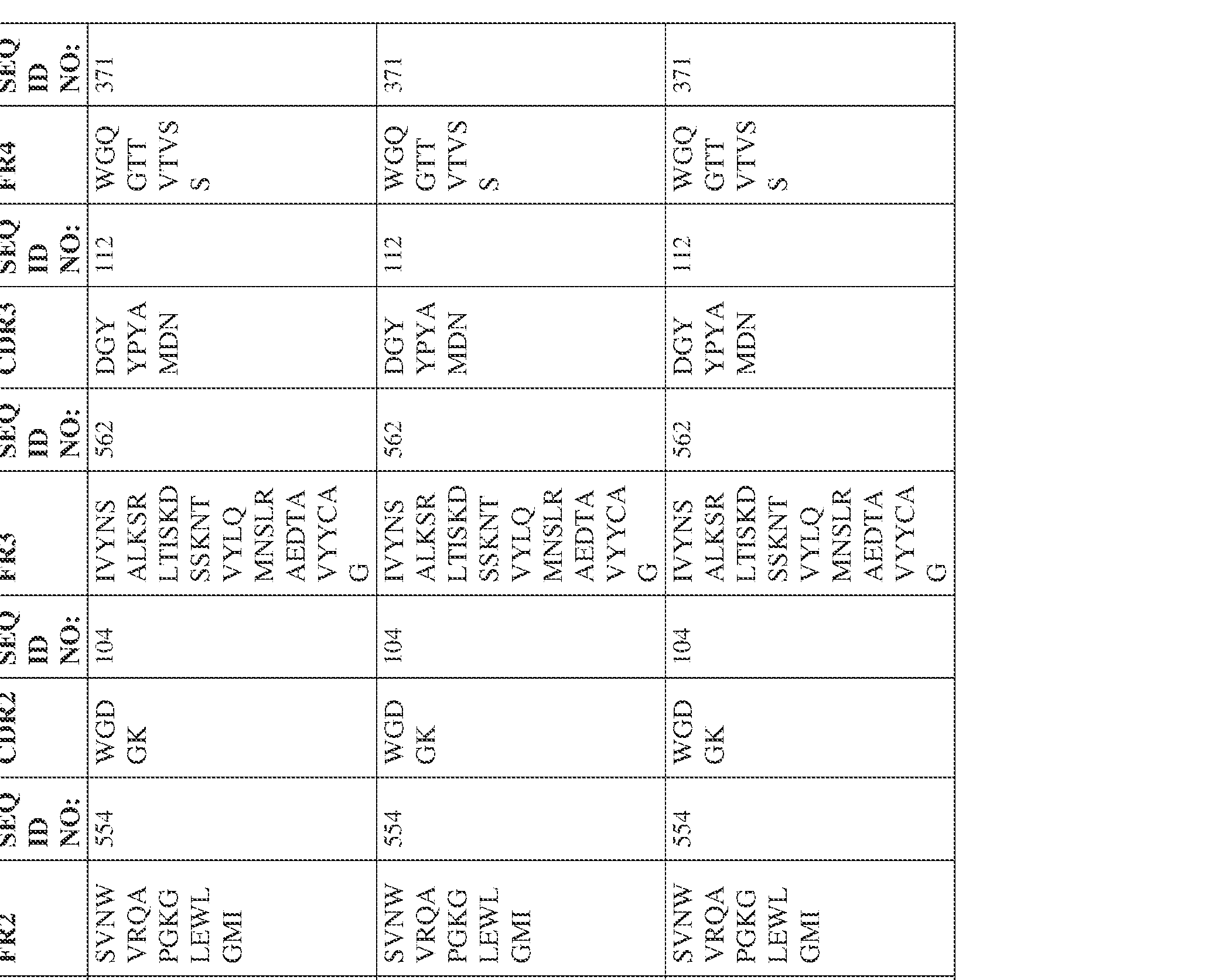
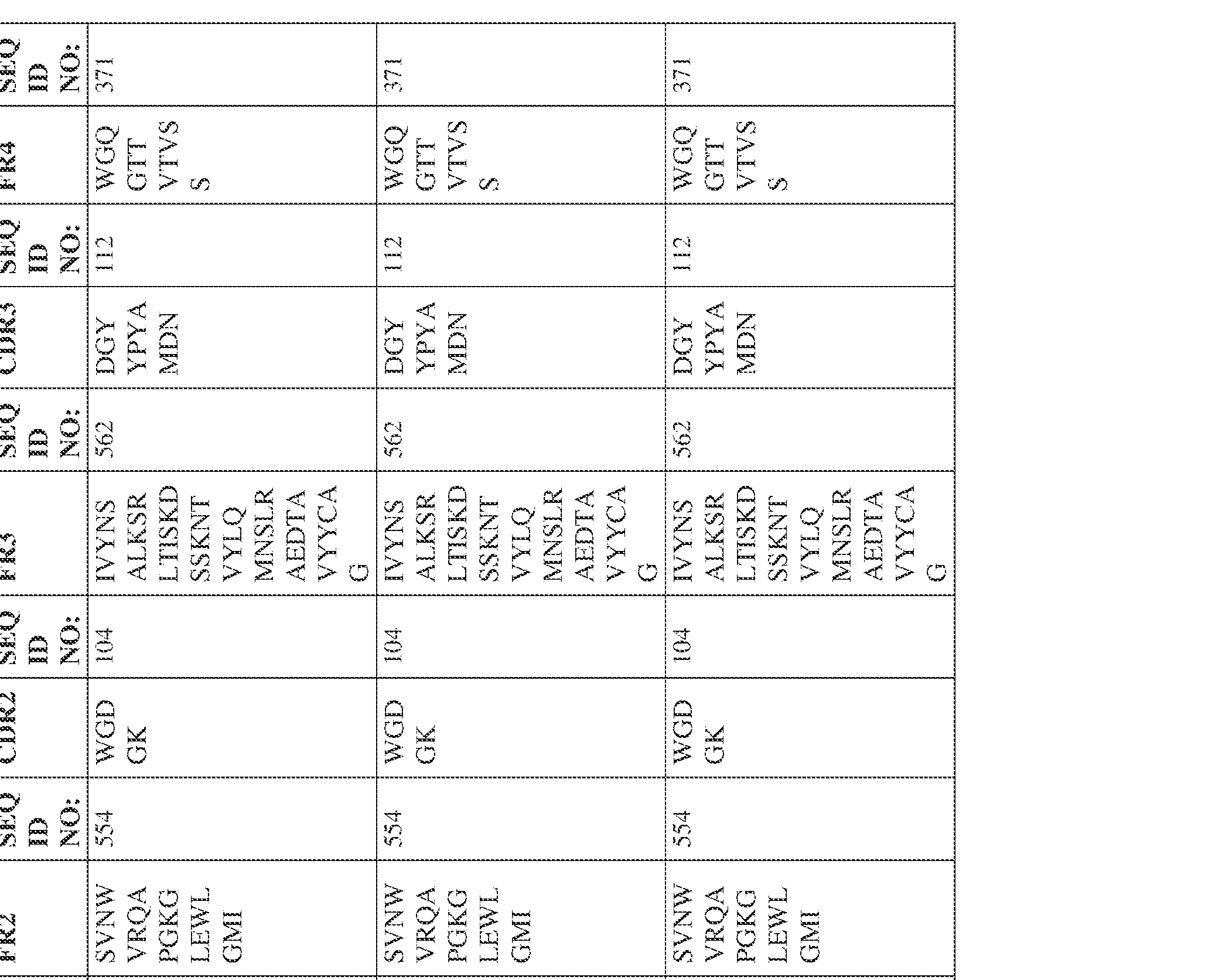
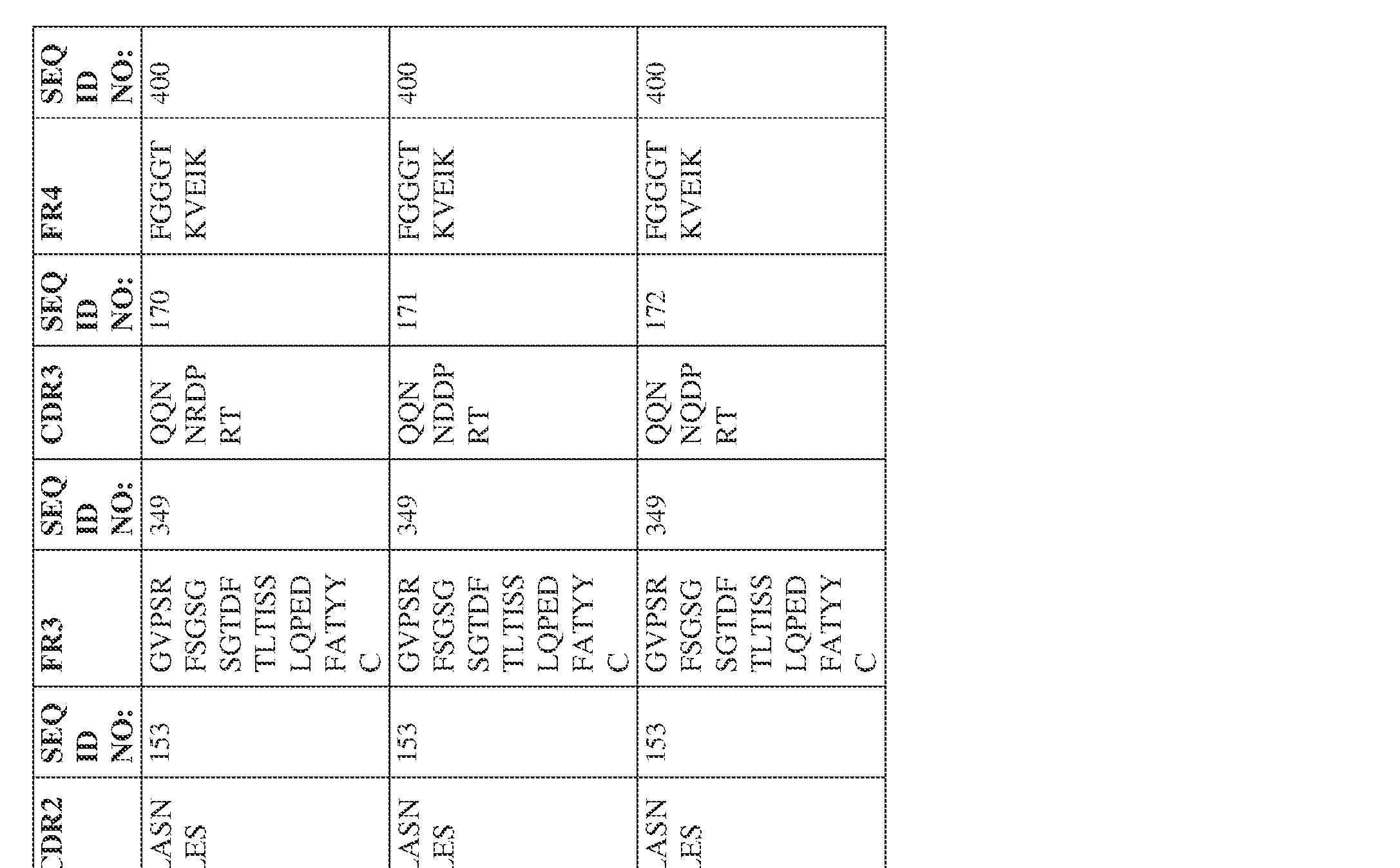
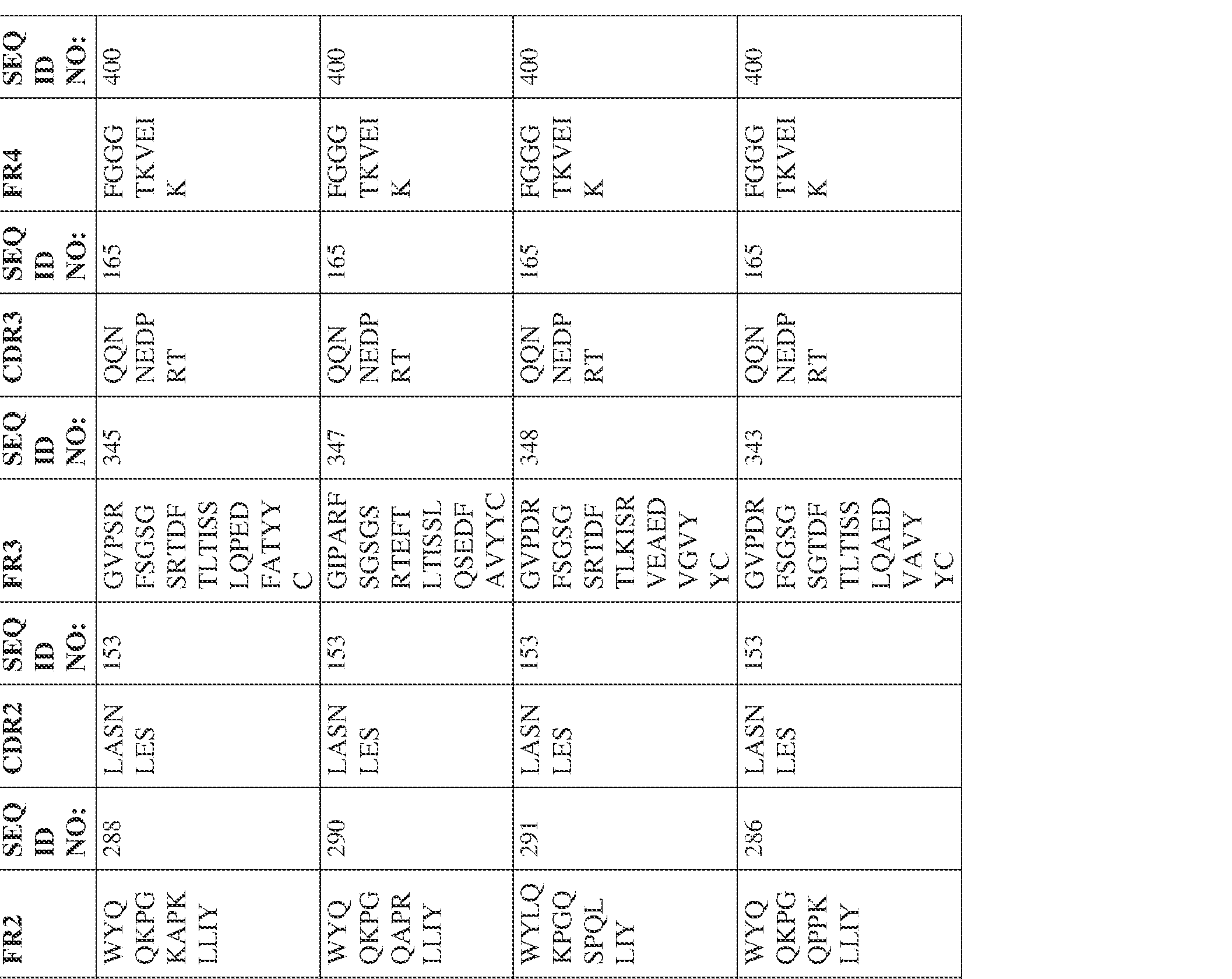
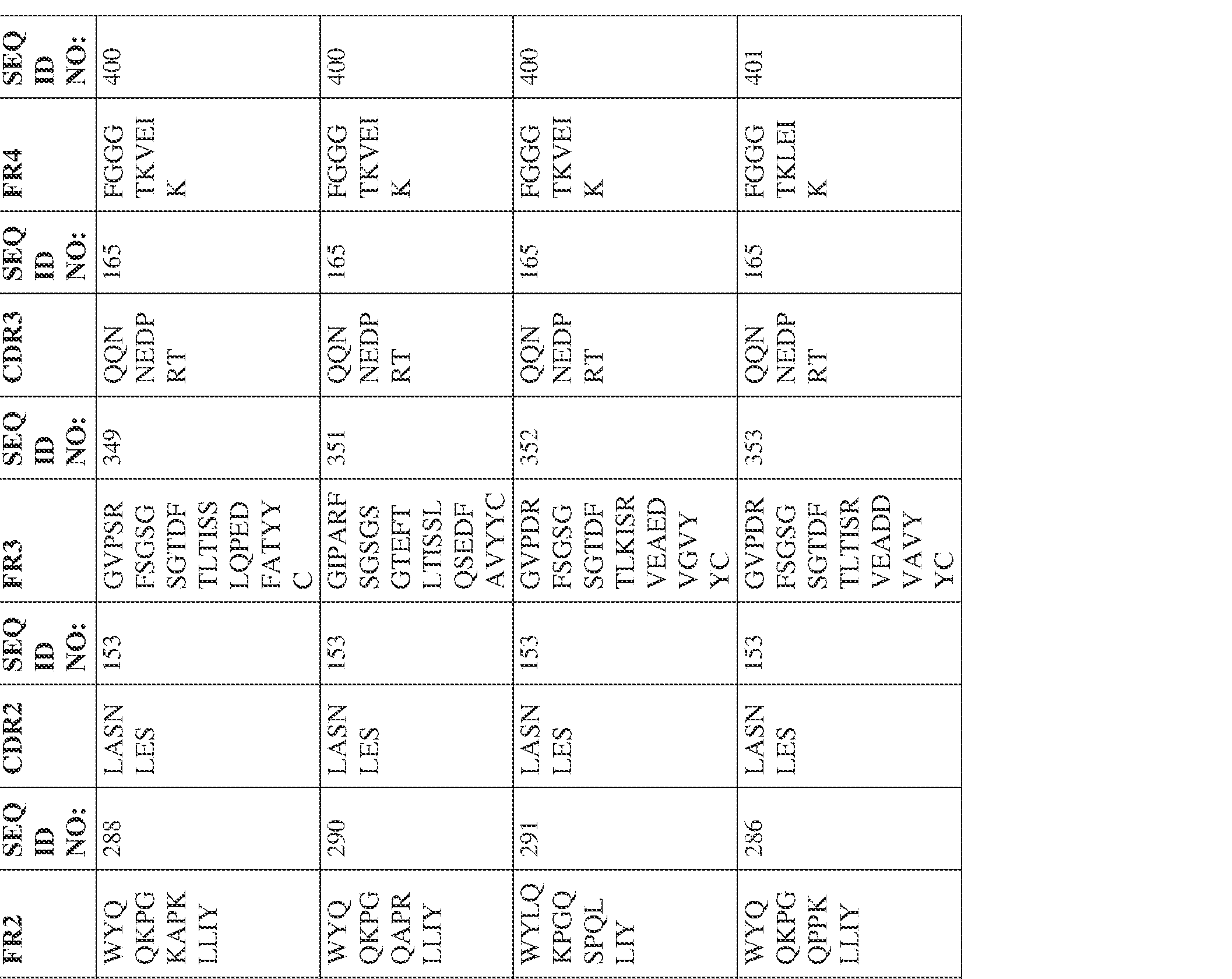
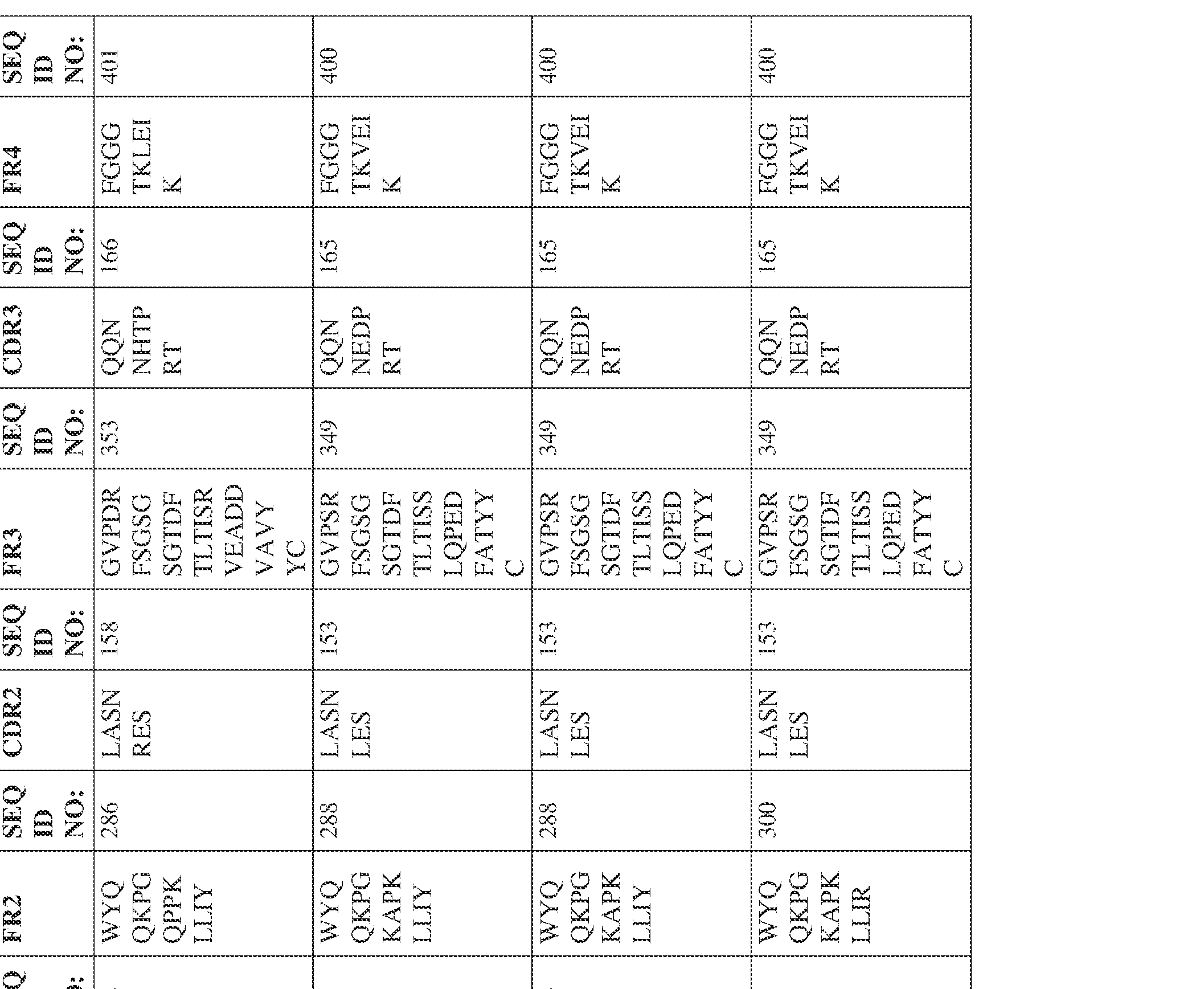
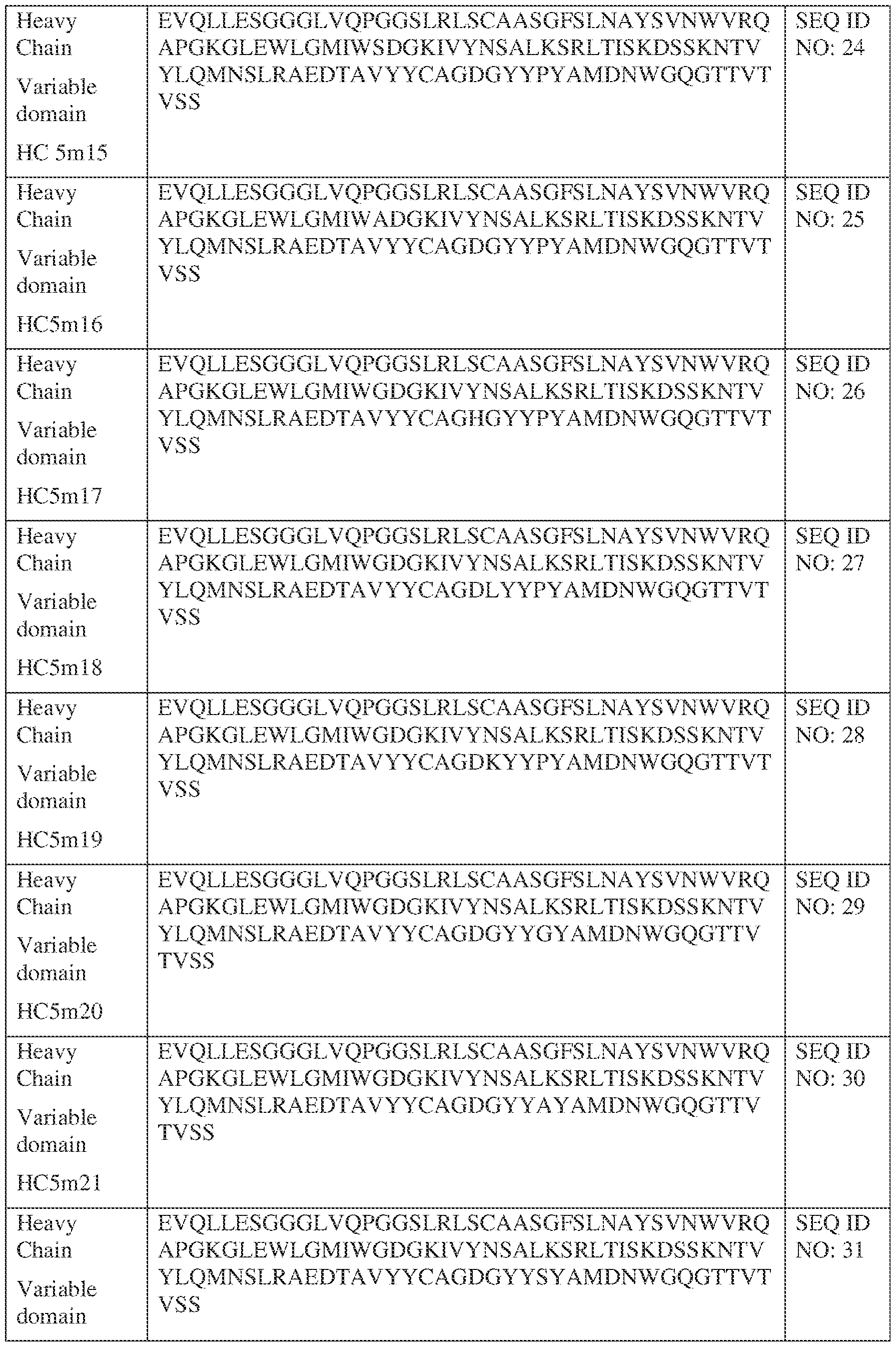
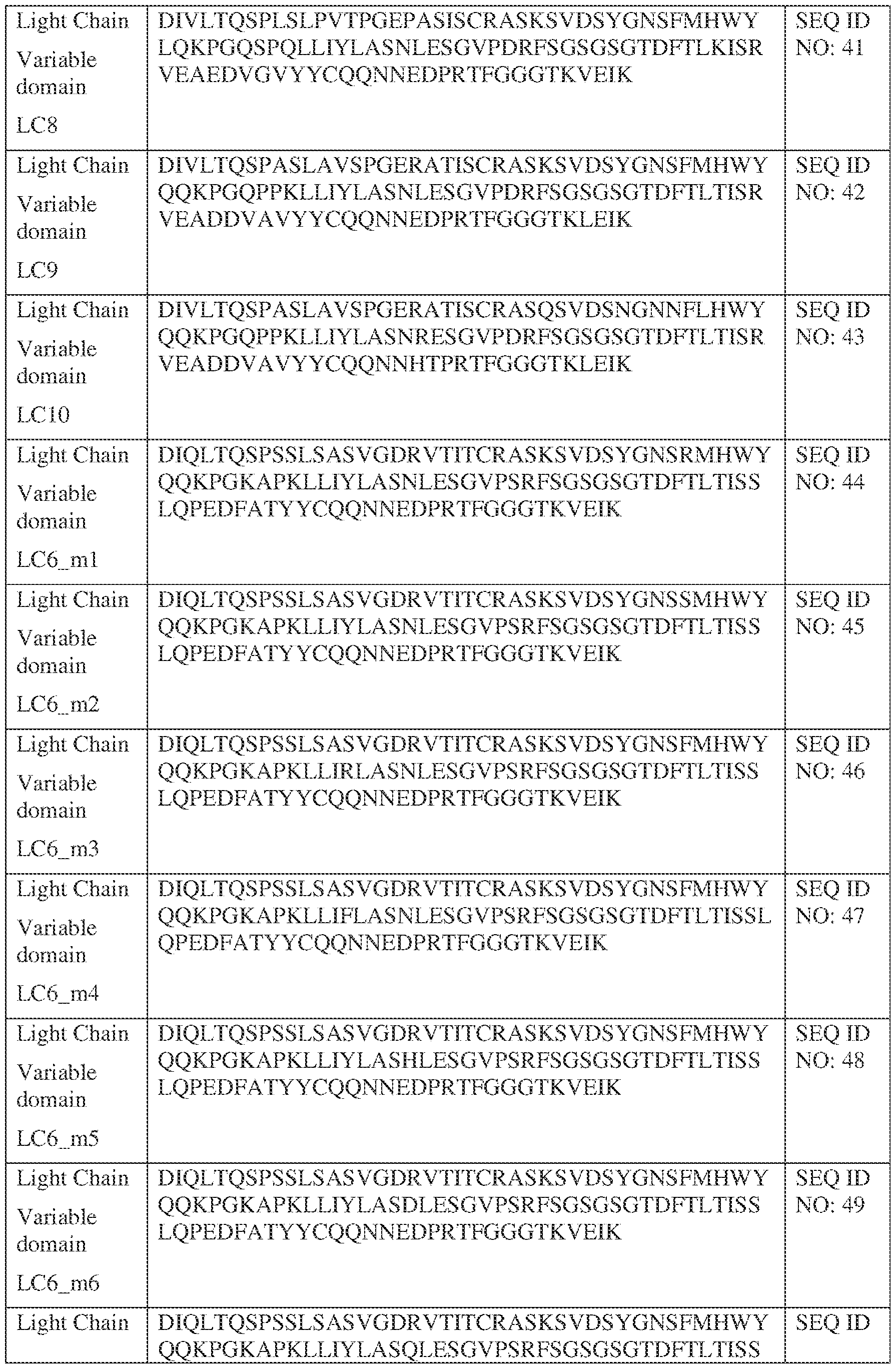
- Anti-interleukin (IL)-13 antibody VH-VL sequences IPTS/128623945.3 30 Attorney Docket No. PRG-013WO IPTS/128623945.3 31 Attorney Docket No. PRG-013WO IPTS/128623945.3 32 Attorney Docket No. PRG-013WO IPTS/128623945.3 33 Attorney Docket No. PRG-013WO IPTS/128623945.3 34 Attorney Docket No. PRG-013WO IPTS/128623945.3 35 Attorney Docket No. PRG-013WO IPTS/128623945.3 36 Attorney Docket No. PRG-013WO IPTS/128623945.3 36 Attorney Docket No. PRG-013WO IPTS/128623945.3 37 Attorney Docket No. PRG-013WO IPTS/128623945.3 38 Attorney Docket No.
- PRG-013WO IPTS/128623945.3 39 Attorney Docket No. PRG-013WO IPTS/128623945.3 40 Attorney Docket No. PRG-013WO IPTS/128623945.3 41 Attorney Docket No. PRG-013WO IPTS/128623945.3 42 Attorney Docket No. PRG-013WO IPTS/128623945.3 43 Attorney Docket No.
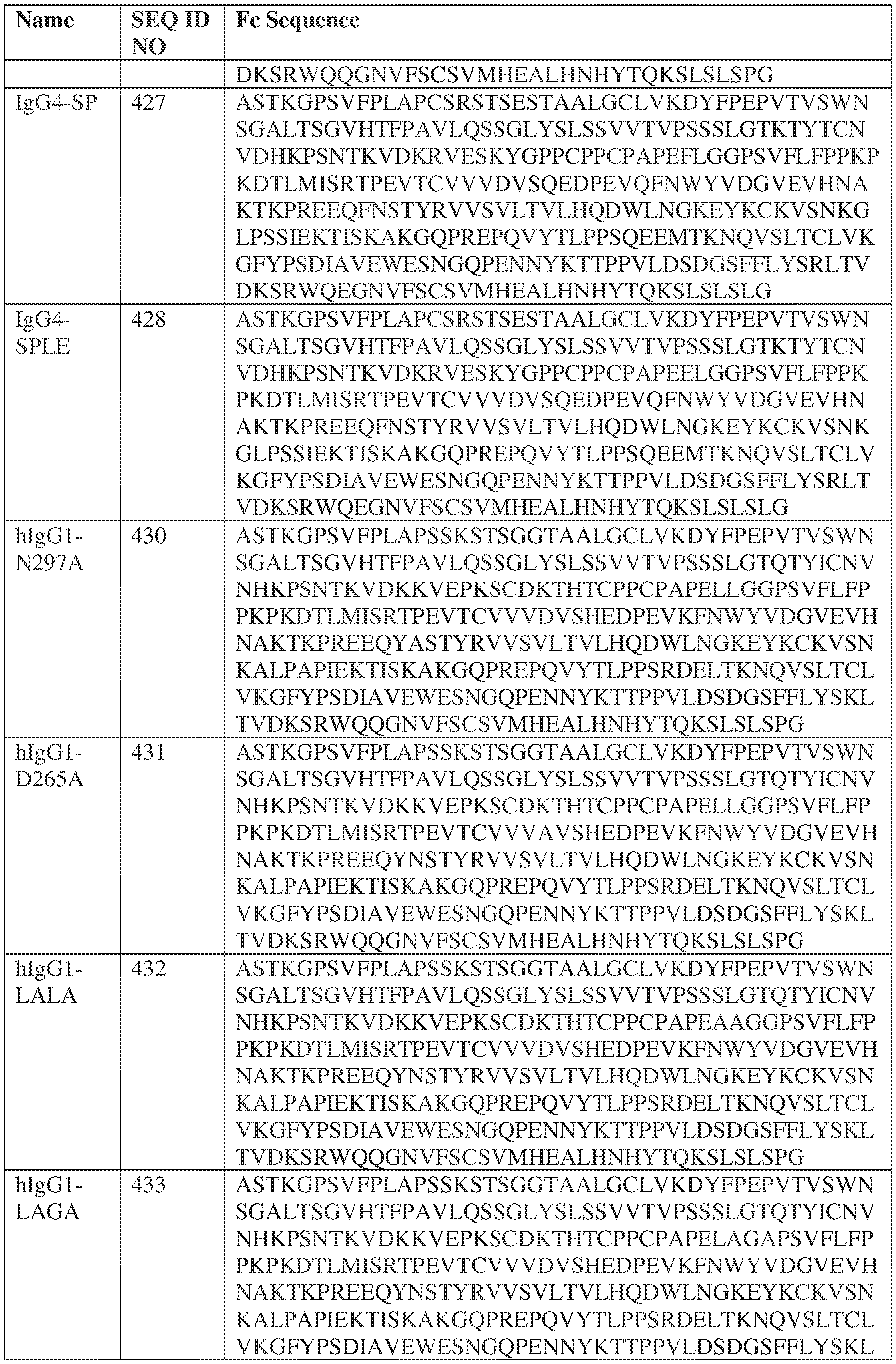
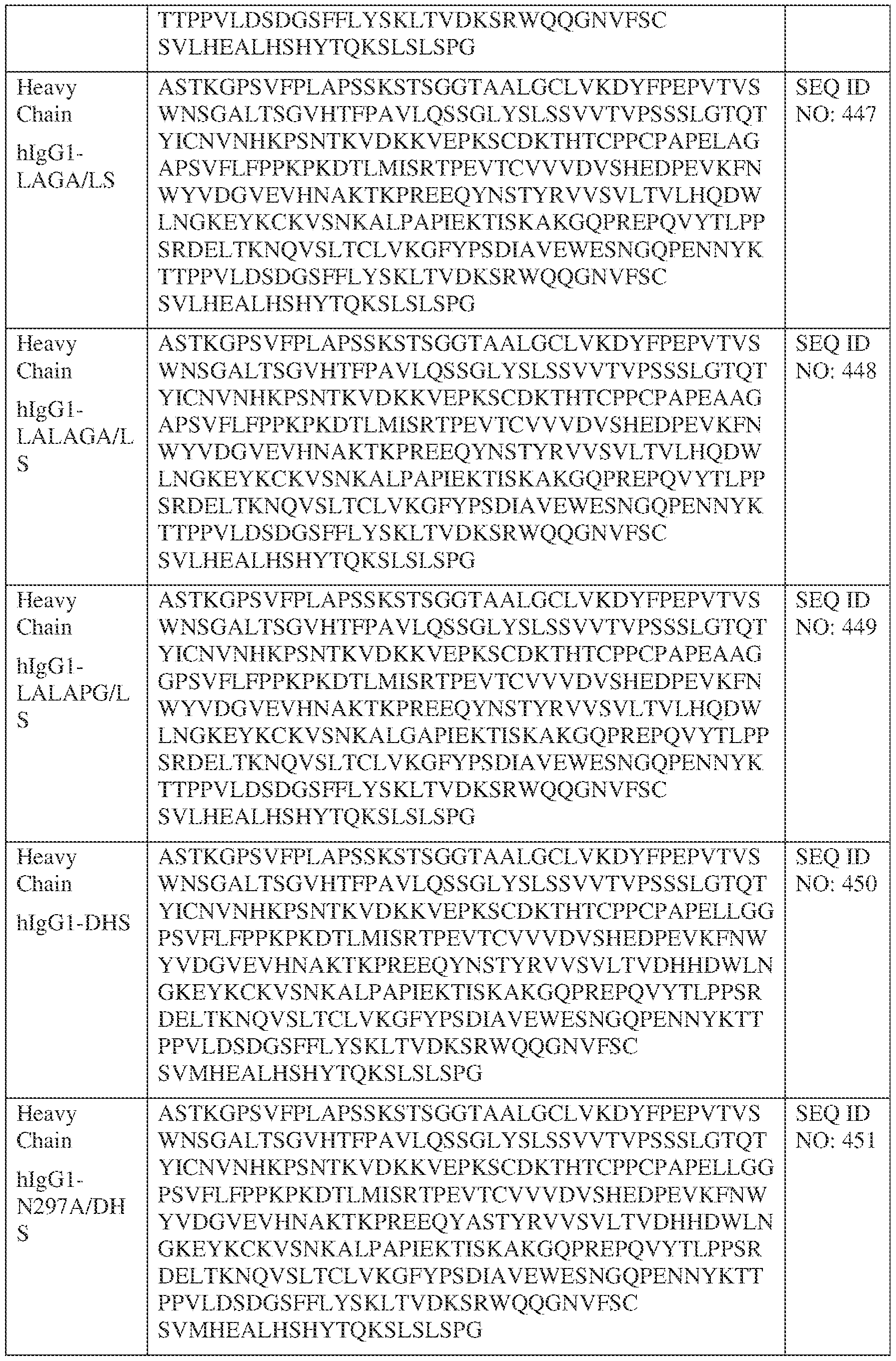
- such a IgG4-SP HC constant domain has the sequence: ASTKGPSVFPLAPCSRSTSESTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQ SSGLYSLSSVVTVPSSSLGTKTYTCNVDHKPSNTKVDKRVESKYGPPCPPCPAPEFLG GPSVFLFPPKPKDTLMISRTPEVTCVVVDVSQEDPEVQFNWYVDGVEVHNAKTKPRE EQFNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKGLPSSIEKTISKAKGQPREPQVYT LPPSQEEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGSFFLYS RLTVDKSRWQEGNVFSCSVMHEALHNHYTQKSLSLSLGK (SEQ ID NO: 427).
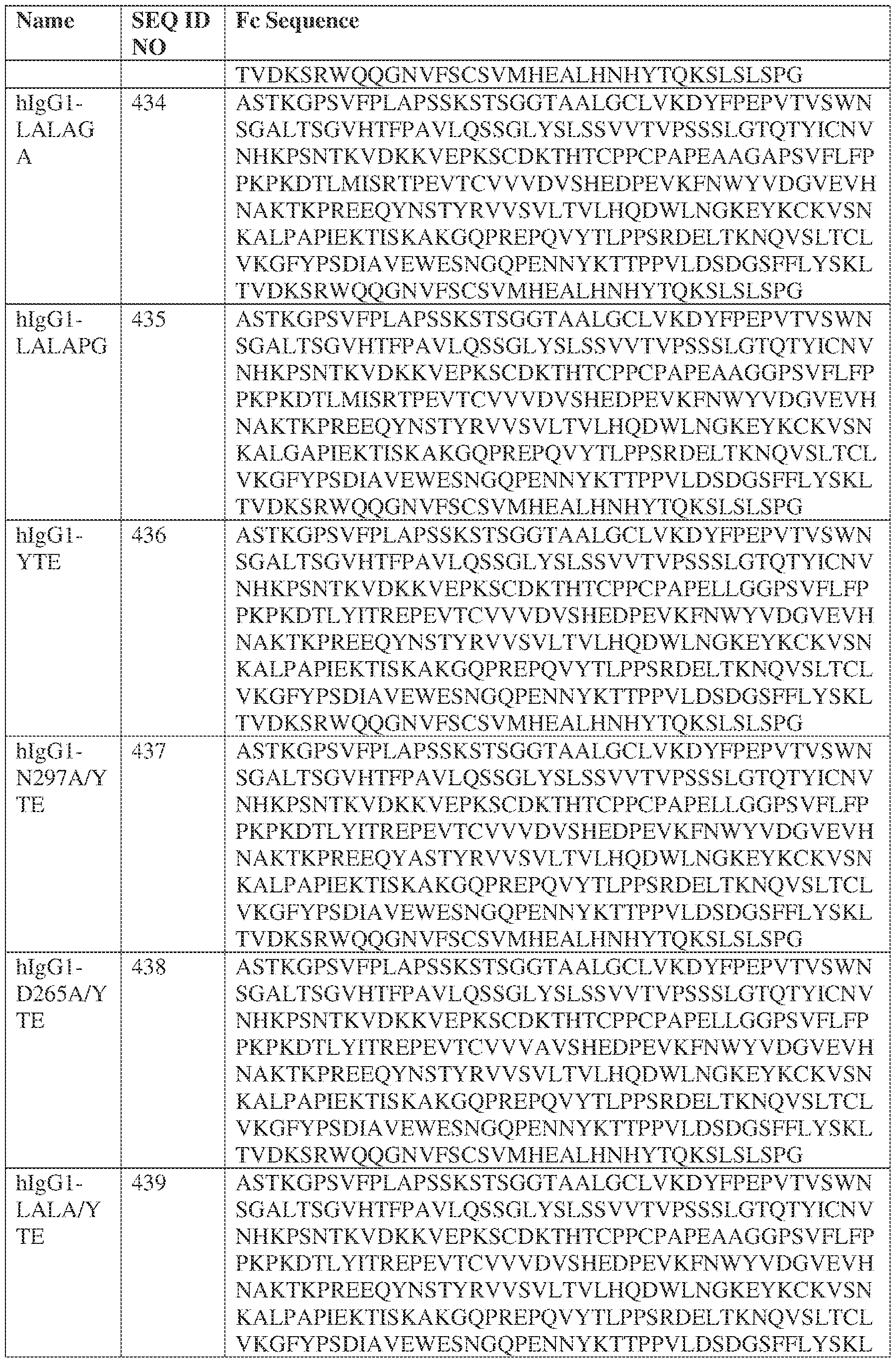
- such a hIgG1-LALA-YTE HC constant domain has the sequence: ASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVL QSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAP EAAGGPSVFLFPPKPKDTLYITREPEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAK TKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAPIEKTISKAKGQPRE PQVYTLPPSRDELTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDG SFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSPG (SEQ ID NO: 439).
- such a hIgG1-LAGA YTE HC constant domain has the sequence: ASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVL QSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAP ELAGAPSVFLFPPKPKDTLYITREPEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKT KPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAPIEKTISKAKGQPREP QVYTLPPSRDELTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGS FFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSPG (SEQ ID NO: 440).
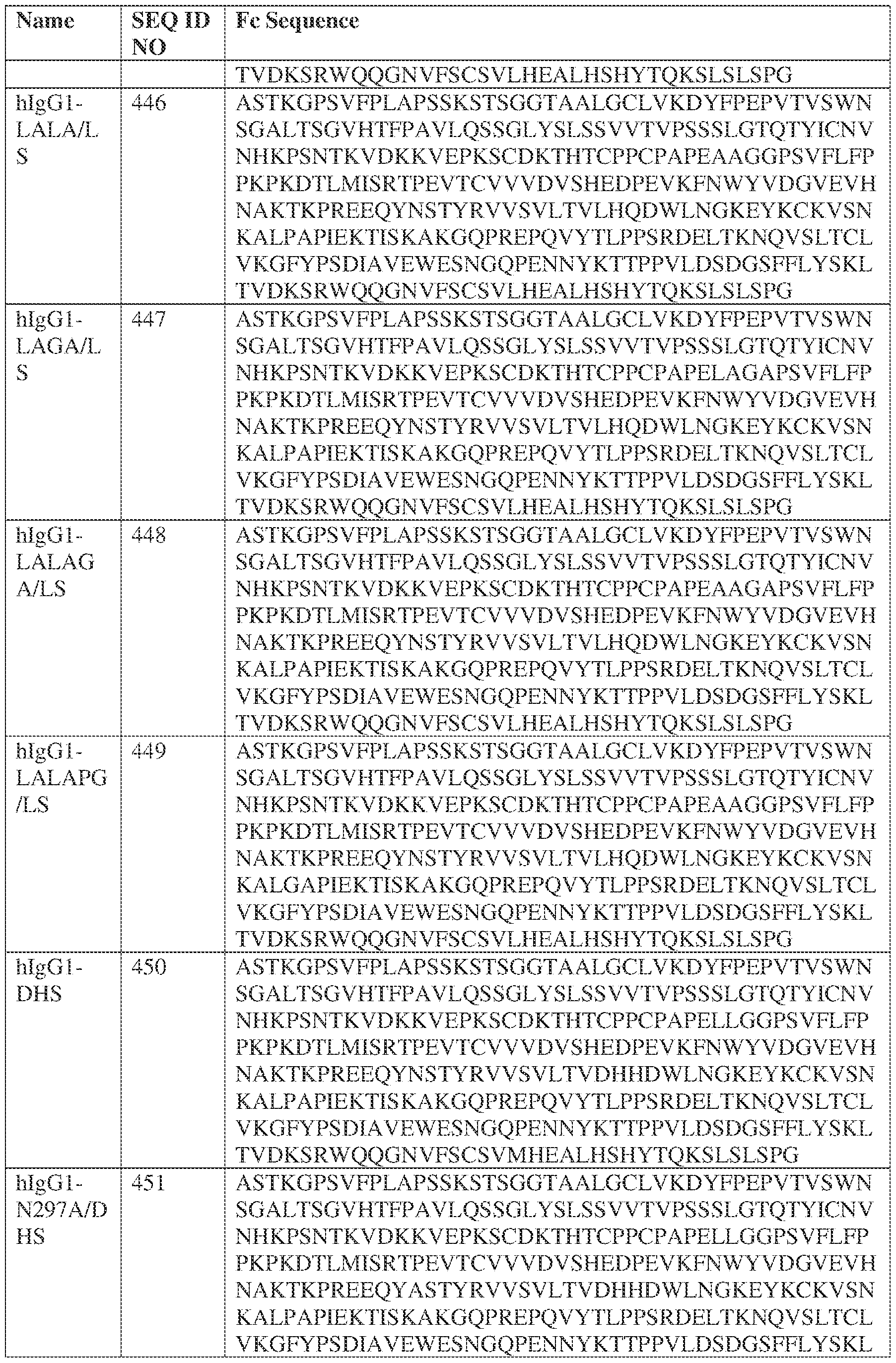
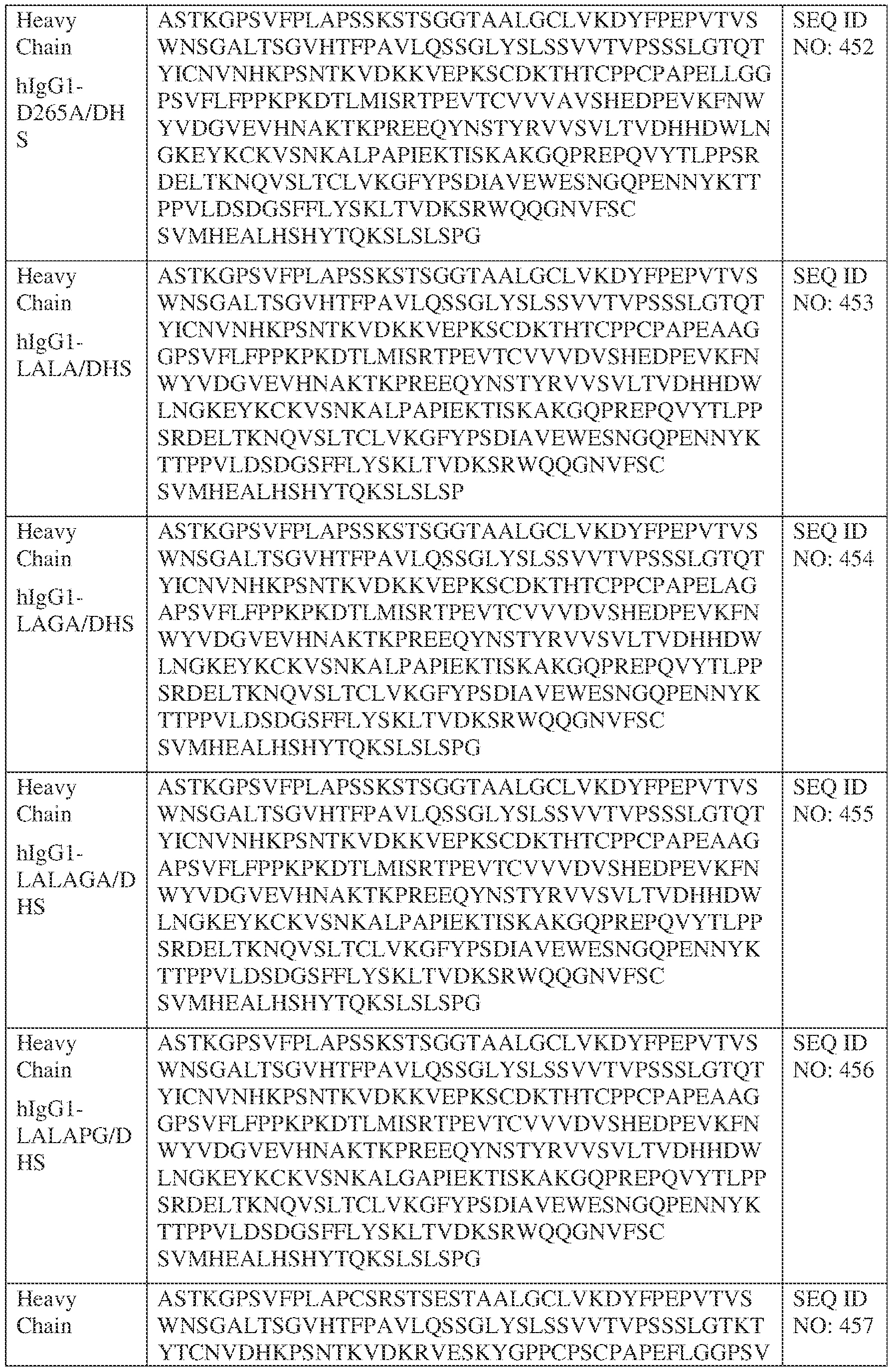
- such a hIgG1-LALA-LS HC constant domain has the sequence: [00165] ASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGV HTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHT CPPCPAPEAAGGPSVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWYVDGV EVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAPIEKTISK AKGQPREPQVYTLPPSRDELTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTP PVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVLHEALHSHYTQKSLSPG (SEQ ID NO: 446).
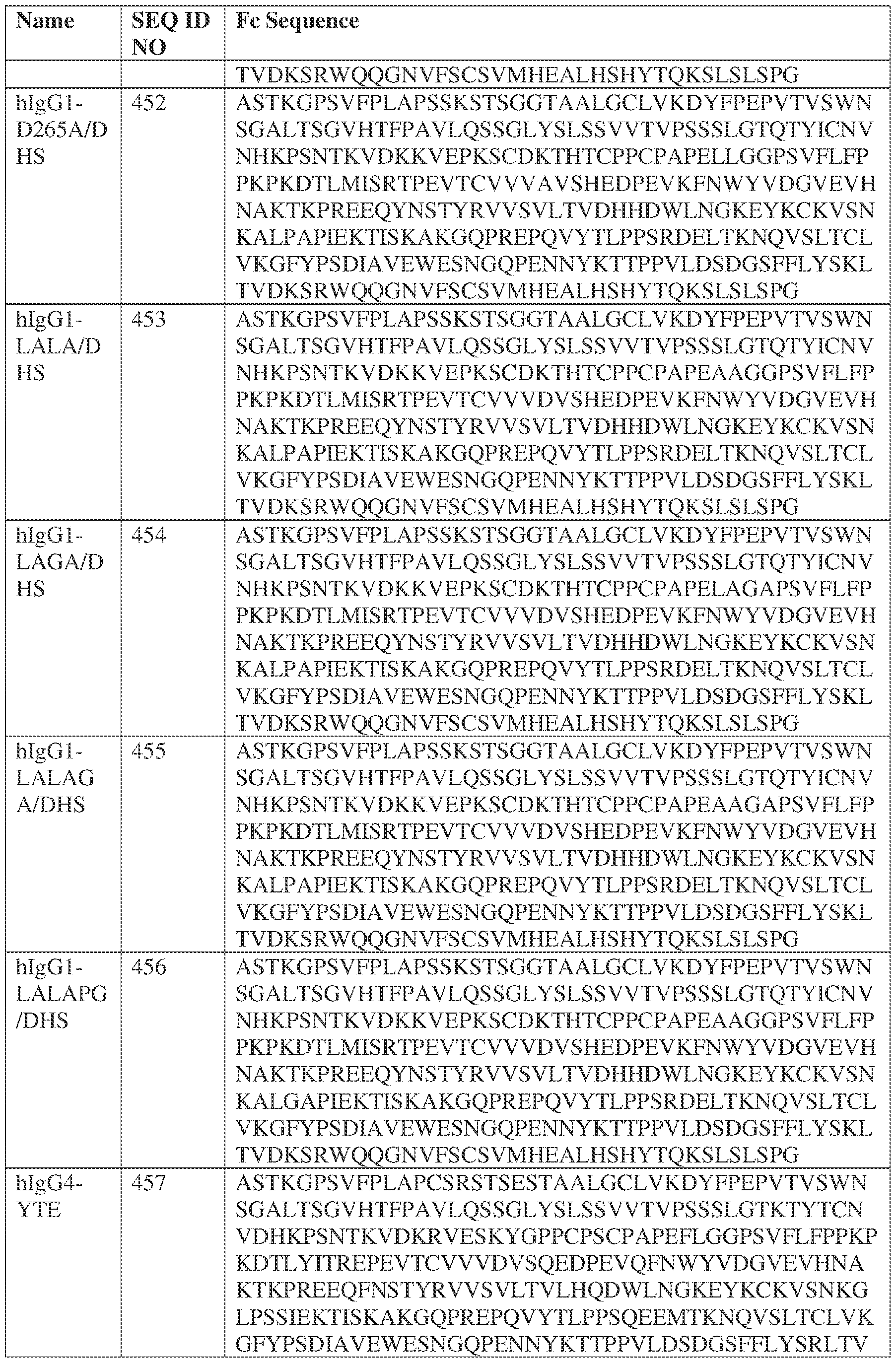
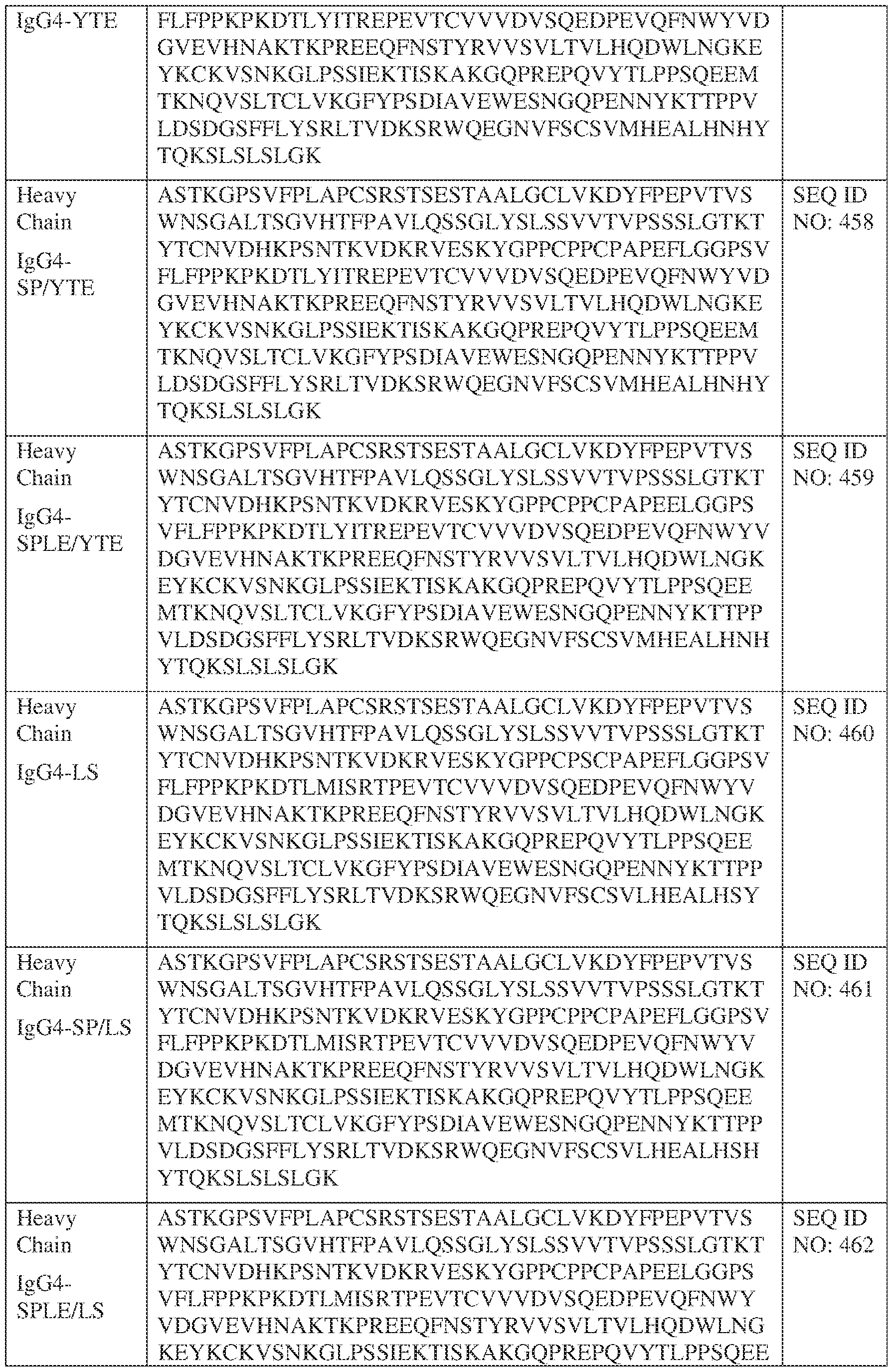
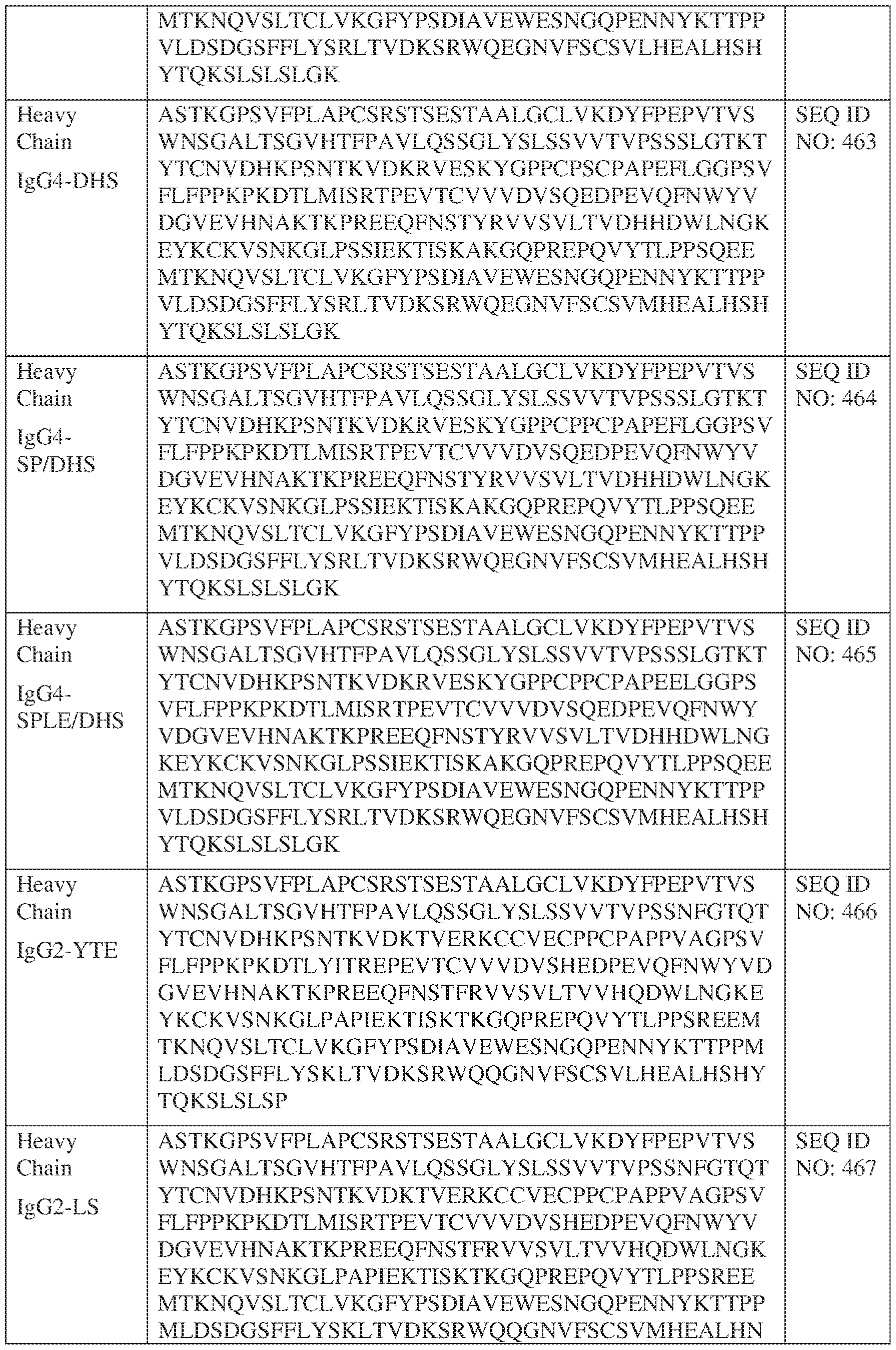
- such a IgG4-YTE HC constant domain has the sequence: ASTKGPSVFPLAPCSRSTSESTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQ SSGLYSLSSVVTVPSSSLGTKTYTCNVDHKPSNTKVDKRVESKYGPPCPSCPAPEFLG GPSVFLFPPKPKDTLYITREPEVTCVVVDVSQEDPEVQFNWYVDGVEVHNAKTKPRE EQFNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKGLPSSIEKTISKAKGQPREPQVYT LPPSQEEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGSFFLYS RLTVDKSRWQEGNVFSCSVMHEALHNHYTQKSLSLSLGK (SEQ ID NO: 457).
- such a IgG4-LS HC constant domain has the sequence: ASTKGPSVFPLAPCSRSTSESTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQ SSGLYSLSSVVTVPSSSLGTKTYTCNVDHKPSNTKVDKRVESKYGPPCPSCPAPEFLG GPSVFLFPPKPKDTLMISRTPEVTCVVVDVSQEDPEVQFNWYVDGVEVHNAKTKPRE EQFNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKGLPSSIEKTISKAKGQPREPQVYT LPPSQEEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGSFFLYS RLTVDKSRWQEGNVFSCSVLHEALHSYTQKSLSLSLGK (SEQ ID NO: 460).
- such a human kappa LC constant domain has the sequence: IPTS/128623945.3 45 Attorney Docket No. PRG-013WO RTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQESVT EQDSKDSTYSLSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFNRGEC (SEQ ID NO: 469).
- a bispecific antibody provided herein comprises a first antigen binding site comprising three CDRs of a first VH domain selected from SEQ ID NOs: 1-32, 470, and 688, such as any of the CDRs listed in Table 3, Table 4, or Table 5, below, and a second antigen binding site comprising three CDRs of a second VH domain selected from SEQ ID NOs: 659-661, 682, 684, 686, and 688, such as any of the CDRs listed in Table 3a.
- the CDRs are Exemplary CDRs.
- the CDRs are Kabat CDRs.
- the CDRs are Chothia CDRs.
- the CDRs are IMGT CDRs. In some embodiments, the CDRs are AbM CDRs. In some embodiments, the CDRs are Contact CDRs. [00170] In some embodiments, the first antigen binding site comprises CDRs having at least about 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% or 100% identity with a CDR-H1, CDR-H2, or CDR-H3 of SEQ ID NOs: 58-140, and the second antigen binding site comprises CDRs having at least about 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% or 100% identity with a CDR-H1, CDR-H2, or CDR-H3 of SEQ ID NOs: 610-636.
- the amino acid substitutions are conservative amino acid substitutions.
- the antibodies described in this paragraph are referred to herein as “variants.”
- such variants are derived from a sequence provided herein, for example, by affinity maturation, site directed mutagenesis, random mutagenesis, or any other method known in the art or described herein.
- such variants are not derived from a sequence provided herein and may, for example, be isolated de novo according to the methods provided herein for obtaining antibodies.
- a bispecific antibody provided herein comprises a first antigen binding site comprising one to three CDRs of a VL domain of SEQ ID NOs: 33-57, 471, and 687, such as any of the CDRs listed in Table 6, Table 7, or Table 8, and a second antigen binding comprising one to three CDRs of a VL domain of SEQ ID NOs: 662-664, 681, 683, and 685, such as any of the CDRs listed in Table 3a, below.
- a bispecific antibody provided herein comprises a first antigen binding site comprising three CDRs of a VL domain of SEQ ID NOs: 33-57, 471, and 687, such as any of the CDRs listed IPTS/128623945.3 46 Attorney Docket No. PRG-013WO in Table 6, Table 7, or Table 8, and a second antigen binding comprising three CDRs of a VL domain of SEQ ID NOs: 662-664, 681, 683, and 685, such as any of the CDRs listed in Table 3a, below.
- the CDRs are Exemplary CDRs.
- the CDRs are Kabat CDRs.
- the CDRs are Chothia CDRs.
- the CDRs are IMGT CDRs. In some embodiments, the CDRs are AbM CDRs. In some embodiments, the CDRs are Contact CDRs.
- the first antigen binding site comprises CDRs having at least about 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% or 100% identity with a CDR-L1, CDR-L2, or CDR-L3 of SEQ ID NOs: 141-188
- the second antigen binding site comprises CDRs having at least about 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% or 100% identity with a CDR-L1, CDR-L2, or CDR-L3 of SEQ ID NOs: 637-657.
- the amino acid substitutions are conservative amino acid substitutions.
- the antibodies described in this paragraph are referred to herein as “variants.”
- such variants are derived from a sequence provided herein, for example, by affinity maturation, site directed mutagenesis, random mutagenesis, or any other method known in the art or described herein.
- such variants are not derived from a sequence provided herein and may, for example, be isolated de novo according to the methods provided herein for obtaining antibodies.
- a bispecific antibody provided herein comprises a first antigen binding site comprising three CDRs of a VH domain selected from SEQ ID NOs: 1- 32, 470, and 688 and three CDRs of a VL domain of SEQ ID NOs: 33-57, 471, and 687, and a second antigen binding site comprising three CDRs of a VH domain selected from SEQ ID NOs: 659-661, 682, 684, 686, and 688 and three CDRs of a VL domain of SEQ ID NOs: 662- 664, 681, 683, and 685.
- a bispecific antibody provided herein comprises a first antigen binding site comprising three CDRs of a VH domain comprising SEQ ID NO: 3 and three CDRs of a VL domain comprising SEQ ID NO: 39, and a second antigen binding site comprising three CDRs of a VH domain selected from SEQ ID NOs: 659-661, 682, 684, 686, and 688 and three CDRs of a VL domain of SEQ ID NOs: 662-664, 681, 683, and 685.
- the CDRs are Exemplary CDRs.
- the CDRs are Kabat CDRs.
- the CDRs are Chothia CDRs.
- a bispecific antibody provided herein comprises a first antigen binding site comprising a CDR-H3 selected from SEQ ID NOs: 112-120 and 130- 140, a CDR-H2 of SEQ ID NOs: 100-111, a CDR-H1 selected from SEQ ID NOs: 58-99 and 121, a CDR-L3 selected from SEQ ID NOs: 165-172, a CDR-L2 selected from SEQ ID NOs: 153-158 and LAS, and a CDR-L1 selected from SEQ ID NOs: 141-144 and 149-152, and a second antigen binding site comprising a CDR-H3 selected from SEQ ID NOs: 628-6
- a bispecific antibody provided herein comprises a first antigen binding site comprising a CDR-H3 selected from SEQ ID NOs: 112 and 130, a CDR-H2 of SEQ ID NOs: 100, 104, 108, a CDR- H1 selected from SEQ ID NOs: 58, 68, and 85, a CDR-L3 comprising SEQ ID NO: 165, a CDR-L2 selected from SEQ ID NOs: 153 and LAS, and a CDR-L1 selected from SEQ ID NOs: 141 and 589, and a second antigen binding site comprising a CDR-H3 selected from SEQ ID NOs: 628-636, a CDR-H2 of SEQ ID NOs: 619-627, a CDR-H1 selected from SEQ ID NOs: 610-618, a CDR-L3 selected from SEQ ID NOs: 652-657, a CDR-L2 selected from SEQ ID NOs: 646
- the CDR-H3 has at least about 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% identity with a CDR-H3 selected from SEQ ID NOs: 112 and 130
- the CDR-H2 has at least about 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% identity with a CDR-H2 of SEQ ID NOs: 100, 104, 108
- the CDR- H1 has at least about 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% identity with a CDR-H1 selected from SEQ ID NOs: 58, 68, and 85
- the CDR-L3 has at least about 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% identity with a CDR- L3 comprising a CDR
- the CDR-L2 has at least about 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% identity with a CDR-L2 selected from SEQ ID NOs: 646-651
- the CDR-L1 has at least about 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% identity with a CDR-L1 selected from SEQ ID NOs: 637-645 in the second antigen binding site.
- the amino acid substitutions are conservative amino acid substitutions.
- the antibodies described in this disclosure are referred to herein as “variants” or “clones”.
- such variants or clones are derived from a sequence provided herein, for example, by affinity maturation, site directed mutagenesis, random mutagenesis, or any other method known in the art or described herein.
- such variants or cones are not derived from a sequence provided herein and may, for example, be isolated de novo according to the methods provided herein for obtaining antibodies.
- the antibodies disclosed herein do not include antibodies disclosed in U.S. Pat. No.9,067,994.
- IPTS/128623945.3 49 Attorney Docket No. PRG-013WO Q : 3 1- ) L QKW Q W Q W I ( 2 n R R I G E RK i F L VG E RK L VG E W P P A A L W P P G G L W P P P L G G L k u e : l Q r e t E S D I O N 8 5 8 5 8 5 n I -i t R S S S n D YN YN YN A C 1 A V A V A V .
- PRG-013WO Q E O 8 9 9 S D I N 6 3 6 3 6 3 e l r e t E S D I O N 7 6 8 6 8 6 n I -i 1 t R L S Y LY LY n D F S F A S F A A .
- PRG-013WO Q E O 8 N 6 9 9 S D I 3 6 3 6 3 u e 2 W P E W P L G l L W P L G L r R e F N L M N t V P Q V Q G N R K W V Q G A A R K W n I -i Q : t E O n S D I N 4 8 5 8 5 8 A .
- PRG-013WO Q E S D I O 0 1 0 N 0 4 0 4 0 4 ) LI ( n Q i Q P Q I P k 2 L Q I Q Q L Q Q K L L Y G L Y G L GK u e R P K P K Y P P Y l F W K P Y W K P Y W K A I r e t n Q : I 1 1 -i E t S D I O N 4 1 1 4 1 4 1 n A .
- PRG-013WO Q E O 0 1 0 S D I N 0 4 0 4 0 4 - ) LI ( 2 Q G Y P K i K P Y Q G P KY Q G P K P Y P I Y P I Y A I n k R F W Q Q L L W K Q P Q L L W K Q K L L u e l r Q : e t E O 1 1 1 n S D I N 4 1 4 1 4 1 I -i t 1 R K S S S S S N K S K S S S n H NH NH D DG D A .
- PRG-013WO Q E S D I O 8 8 8 N 8 2 8 2 8 2 2 Q G R Y P K P Y I Q G P K P Y I Q G P K P Y I F W K Q A K L Y L W K Q A K L Y L W K Q A K L L L L .
- PRG-013WO Q E O 0 N 0 1 0 0 0 S D I 4 4 0 4 0 4 u e l Q : r e t E S D I O 9 N 4 9 1 4 9 1 4 9 1 4 1 n i -i S t 1 D F S S D F S S D F S n R V S N V N V N V N A D G .
- the format of the bispecific antibodies disclosed herein have any of the bispecific antibody formats described herein.
- the format of the bispecific antibodies disclosed herein is selected from the group consisting of (a) Single chain Fv (scFv), (b) tandem scFv format of bispecific T cell engager (BiTE), (c) disulfide-linked diabody format of dual affinity retargeting (DART) bsAb, (d) tandem diabody (TandAb), (e) conventional immunoglobulin G (IgG), (f) IgGs with additional binding units such as scFv, (g) dual variable domain immunoglobulin (DVD-Ig), (h) quadromab bsAb, (i) knobs-into-holes (KiH) bsAb with a common light chain, (j) KiH- CrossMabCH1-CL, and (k) bsAb by controlled Fab arm exchange (c
- the format of the bispecific antibodies disclosed herein is selected from the group consisting of IgG-scFv and DVD-Ig. In some embodiments, the format of the bispecific antibodies disclosed herein is IgG-scFv. In some embodiments, the format of the bispecific antibodies disclosed herein is DVD-Ig. In some embodiments, the format of the bispecific antibodies disclosed herein is kiH- CrossMabCH1-CL.
- a Heavy Chain/Chain A construct in Table 11a or Table 11b with an alias and construct number is combined with the Light Chain/Chain B construct having the same alias and construct number in Table 12a or Table 12b.
- the abbreviations used to describe the first VH sequence, heavy chain constant region, first VL sequence, second VH sequence, linkers, and light chain constant domain in the paragraphs below correspond to the abbreviations used in column titles of Table 11a, Table 11b, Table 12a, and Table 12b.
- the format of the bispecific antibodies disclosed herein is IgG-scFv.
- the bispecific format comprises a first VH sequence (VH1) comprising SEQ ID NO: 3, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and linker H2 comprising SEQ ID NO: 665, a first VL sequence (VL1) comprising SEQ ID NO: 662, a linker H3 comprising SEQ ID NO: 666, a second VH (VH2) sequence comprising SEQ ID NO: 659, a second VL sequence (VL2) comprising SEQ ID NO: 39, and a light chain constant domain (CL) comprising SEQ ID NO: 469.
- VH1 comprising SEQ ID NO: 3
- linker H2 comprising SEQ ID NO: 665
- VL1 comprising SEQ ID NO: 662
- linker H3 comprising SEQ ID NO: 666
- VH2 VH2 sequence comprising SEQ ID NO: 659
- VL2 comprising SEQ ID NO: 39
- CL light chain constant domain
- the bispecific format comprises a first VH sequence (VH1) comprising SEQ ID NO: 3, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and linker H2 comprising SEQ ID NO: 665, a first VL sequence (VL2) comprising SEQ ID NO: 663, a linker H3 comprising SEQ ID NO: 666, a second VH (VH2) sequence comprising SEQ ID NO: 660, a second VL sequence (VL1) comprising SEQ ID NO: 39, and a light chain constant domain (CL) comprising SEQ ID NO: 469.
- the bispecific format comprises a first VH sequence (VH1) comprising SEQ ID NO: 3, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and linker H2 comprising SEQ ID NO: 665, a first VL sequence (VL2) comprising SEQ ID NO: 664, a linker H3 comprising SEQ ID NO: 666, a second VH (VH2) sequence comprising SEQ ID NO: 661, a second VL sequence (VL1) comprising SEQ ID NO: 39, and a light chain constant domain (CL) comprising SEQ ID NO: 469.
- VH1 comprising SEQ ID NO: 3
- VL2 comprising SEQ ID NO: 665
- VL2 comprising SEQ ID NO: 664
- linker H3 comprising SEQ ID NO: 666
- VH2 VH2 sequence comprising SEQ ID NO: 661
- VL1 comprising SEQ ID NO: 39
- CL light chain constant domain
- the bispecific format comprises a first VH sequence (VH1) comprising SEQ ID NO: 659, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and linker H2 comprising SEQ ID NO: 665, a first VL sequence (VL2) comprising SEQ ID NO: 39, a linker H3 comprising SEQ ID NO: 666, a second VH (VH2) sequence comprising SEQ ID NO: 3, a second VL sequence (VL1) comprising SEQ ID NO: 662, and a light chain constant domain (CL) comprising SEQ ID NO: 669.
- the bispecific format comprises a first VH sequence (VH1) comprising SEQ ID NO: 660, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and linker H2 comprising SEQ ID NO: 665, a first VL sequence (VL2) comprising SEQ ID NO: 39, a linker H3 comprising SEQ ID NO: 666, a second VH (VH2) sequence comprising SEQ ID NO: 3, a second VL sequence (VL1) comprising SEQ ID NO: 663, and a light chain constant domain (CL) comprising SEQ ID NO: 469.
- the bispecific format comprises a first VH sequence (VH1) comprising SEQ ID NO: 661, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and linker H2 comprising SEQ ID NO: 665, a first VL sequence (VL2) comprising SEQ ID NO: 39, a linker H3 comprising SEQ ID NO: 666, a second VH (VH2) sequence comprising SEQ ID NO: 3, a second VL sequence (VL1) comprising SEQ ID NO: 664, and a light chain constant domain (CL) comprising SEQ ID NO: 469.
- IPTS/128623945.3 104 Attorney Docket No.
- the bispecific format comprises a first VH sequence (VL’) comprising SEQ ID NO: 3, a linker comprising SEQ ID NO: 667, a second VH sequence (VL1) comprising SEQ ID NO: LAS, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and a first VL sequence (VL’) comprising SEQ ID NO: 39, a linker comprising SEQ ID NO: 668, a second VL (VL1) sequence comprising SEQ ID NO: 662, and a light chain constant domain (CL) comprising SEQ ID NO: 469.
- VL VH sequence
- the bispecific format comprises a first VH sequence (VL’) comprising SEQ ID NO: 3, a linker comprising SEQ ID NO: 667, a second VH sequence (VL1) comprising SEQ ID NO: 660, and a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and a first VL sequence (VL’) comprising SEQ ID NO: 39, a linker comprising SEQ ID NO: 668, a second VL (VL1) sequence comprising SEQ ID NO: 663, and a light chain constant domain (CL) comprising SEQ ID NO: 469.
- VL VH sequence
- the bispecific format comprises a first VH sequence (VL’) comprising SEQ ID NO: 3, a linker comprising SEQ ID NO: 667, a second VH sequence (VL1) comprising SEQ ID NO: 661, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and a first VL sequence (VL’) comprising SEQ ID NO: 39, a linker comprising SEQ ID NO: 668, a second VL (VL1) sequence comprising SEQ ID NO: 664, and a light chain constant domain (CL) comprising SEQ ID NO: 469.
- the bispecific format comprises a first VH sequence (VL’) comprising SEQ ID NO: 659, a linker comprising SEQ ID NO: 667, a second VH sequence (VL1) comprising SEQ ID NO: 3, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and a first VL sequence (VL’) comprising SEQ ID NO: 662, a linker comprising SEQ ID NO: 668, a second VL (VL1) sequence comprising SEQ ID NO:39, and a light chain constant domain (CL) comprising SEQ ID NO: 469.
- VL VH sequence
- VL1 comprising SEQ ID NO: 659
- VL1 comprising SEQ ID NO: 667
- VL1 comprising SEQ ID NO: 3
- CH(1-3) heavy chain constant region
- CL light chain constant domain
- the bispecific format comprises a first VH sequence (VL’) comprising SEQ ID NO: 660, a linker comprising SEQ ID NO: 667, a second VH sequence (VL1) comprising SEQ ID NO: 3, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and a first VL sequence (VL’) comprising SEQ ID NO: 663, a linker comprising SEQ ID NO: 668, a second VL (VL1) sequence comprising SEQ ID NO: 39, and a light chain constant domain (CL) comprising SEQ ID NO: 469.
- VL VH sequence
- the bispecific format comprises a first VH sequence (VL’) comprising SEQ ID NO: 661, a linker comprising SEQ ID NO: 667, a second VH sequence (VL1) comprising SEQ ID NO: 3, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and a first VL sequence (VL’) comprising SEQ ID NO: 664, a linker comprising SEQ ID NO: 668, a second VL (VL1) sequence comprising SEQ ID NO: 39, and a light chain constant domain (CL) comprising SEQ ID NO: 469.
- VL VH sequence
- VL1 comprising SEQ ID NO: 661
- VL1 comprising SEQ ID NO: 3
- CH(1-3) heavy chain constant region
- CL light chain constant domain
- the format of the bispecific antibodies disclosed herein is IgG-scFv.
- the bispecific format comprises a first VH sequence (VH1) comprising SEQ ID NO: 3, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and linker H2 comprising SEQ ID NO: 665, a first VL sequence (VL2) comprising SEQ ID NO: 681, a linker H3 comprising SEQ ID NO: 666, a second VH (VH2) sequence comprising SEQ ID NO: 682, a second VL sequence (VL1) comprising SEQ ID NO: 39, and a light chain constant domain (CL) comprising SEQ ID NO: 469.
- VH1 comprising SEQ ID NO: 3
- CH(1-3) heavy chain constant region
- CL light chain constant domain
- the bispecific format comprises a first VH sequence (VH1) comprising SEQ ID NO: 3, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and linker H2 comprising SEQ ID NO: 665, a first VL sequence (VL2) comprising SEQ ID NO: 683, a linker H3 comprising SEQ ID NO: 666, a second VH (VH2) sequence comprising SEQ ID NO: 684, a second VL sequence (VL1) comprising SEQ ID NO: 39, and a light chain constant domain (CL) comprising SEQ ID NO: 469.
- VH1 comprising SEQ ID NO: 3
- linker H2 comprising SEQ ID NO: 665
- VL2 comprising SEQ ID NO: 683
- linker H3 comprising SEQ ID NO: 666
- VH2 sequence comprising SEQ ID NO: 684
- VL1 comprising SEQ ID NO: 39
- CL light chain constant domain
- the bispecific format comprises a first VH sequence (VH1) comprising SEQ ID NO: 3, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and linker H2 comprising SEQ ID NO: 665, a first VL sequence (VL2) comprising SEQ ID NO: 685, a linker H3 comprising SEQ ID NO: 666, a second VH (VH2) sequence comprising SEQ ID NO: 686, a second VL sequence (VL1) comprising SEQ ID NO: 39, and a light chain constant domain (CL) comprising SEQ ID NO: 469.
- VH1 comprising SEQ ID NO: 3
- linker H2 comprising SEQ ID NO: 665
- VL2 comprising SEQ ID NO: 685
- linker H3 comprising SEQ ID NO: 666
- VH2 VH2 sequence comprising SEQ ID NO: 686
- VL1 comprising SEQ ID NO: 39
- CL light chain constant domain
- the bispecific format comprises a first VH sequence (VH1) comprising SEQ ID NO: 659, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and linker H2 comprising SEQ ID NO: 665, a first VL sequence (VL2) comprising SEQ ID NO: 687, a linker H3 comprising SEQ ID NO: 666, a second VH (VH2) sequence comprising SEQ ID NO: 688, a second VL sequence (VL1) comprising SEQ ID NO: 662, and a light chain constant domain (CL) comprising SEQ ID NO: 669.
- IPTS/128623945.3 106 Attorney Docket No.
- the bispecific format comprises a first VH sequence (VH1) comprising SEQ ID NO: 660, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and linker H2 comprising SEQ ID NO: 665, a first VL sequence (VL2) comprising SEQ ID NO: 687, a linker H3 comprising SEQ ID NO: 666, a second VH (VH2) sequence comprising SEQ ID NO: 688, a second VL sequence (VL1) comprising SEQ ID NO: 663, and a light chain constant domain (CL) comprising SEQ ID NO: 469.
- the bispecific format comprises a first VH sequence (VH1) comprising SEQ ID NO: 661, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 439, and linker H2 comprising SEQ ID NO: 665, a first VL sequence (VL2) comprising SEQ ID NO: 687, a linker H3 comprising SEQ ID NO: 666, a second VH (VH2) sequence comprising SEQ ID NO: 688, a second VL sequence (VL1) comprising SEQ ID NO: 664, and a light chain constant domain (CL) comprising SEQ ID NO: 469.
- the bispecific format comprises a first VH sequence (VH1) comprising SEQ ID NO: 3, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 689, a first VL sequence (VL1) comprising SEQ ID NO: 39, and a light chain constant domain (CL) comprising SEQ ID NO: 469, a second VH sequence (VH2) comprising SEQ ID NO: 659, a heavy chain constant domain (CH2) comprising SEQ ID NO: 690, a second VL sequence (VL2) comprising SEQ ID NO: 662, and a second light chain constant domain comprising SEQ ID NO: 692.
- the bispecific format comprises a first VH sequence (VH1) comprising SEQ ID NO: 3, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 689, a first VL sequence (VL1) comprising SEQ ID NO: 39, and a light chain constant domain (CL) comprising SEQ ID NO: 469, a second VH sequence (VH2) comprising SEQ ID NO: 660, a heavy chain constant domain (CH2) comprising SEQ ID NO: 691, a second VL sequence (VL2) comprising SEQ ID NO: 663, and a second light chain constant domain comprising SEQ ID NO: 692.
- the bispecific format comprises a first VH sequence (VH1) comprising SEQ ID NO: 659, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 689, a first VL sequence (VL1) comprising SEQ ID NO: 662, and a light chain constant domain (CL) comprising SEQ ID NO: 669, a second VH sequence (VH2) comprising SEQ ID NO: 3, a heavy chain constant domain (CH2) comprising SEQ ID NO: 691, a second VL sequence (VL2) comprising SEQ ID NO: 39, and a second light chain constant domain comprising SEQ ID NO: 692.
- IPTS/128623945.3 107 Attorney Docket No.
- the bispecific format comprises a first VH sequence (VH1) comprising SEQ ID NO: 660, a heavy chain constant region (CH(1-3)) comprising SEQ ID NO: 689, a first VL sequence (VL1) comprising SEQ ID NO: 663, and a light chain constant domain (CL) comprising SEQ ID NO: 469, a second VH sequence (VH2) comprising SEQ ID NO: 3, a heavy chain constant domain (CH2) comprising SEQ ID NO: 691, a second VL sequence (VL2) comprising SEQ ID NO: 39, and a second light chain constant domain comprising SEQ ID NO: 692.
- IPTS/128623945.3 108 Attorney Docket No.
- bispecific antibodies that bind (1) IL-13 and (2) TSLP or TSLPR comprising modified Fc regions. Unless otherwise specified herein, numbering of amino acid residues in the Fc region or constant region is according to the EU numbering system, also called the EU index, as described in Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, MD, 1991. [00204] In some embodiments, the bispecific antibody comprises a modified Fc comprising one or more modifications.
- the one or more modifications are located in a Fc from IgG1 (e.g., human IgG1 (hIgG1). In some embodiments, the one or more modifications are located in a Fc from IgG4 (e.g., human IgG4 (hIgG4). In some embodiments, the one or more modifications are located in a Fc from IgG2. In some embodiments, the one or more modifications promote selective binding of Fc-gamma receptors. In any embodiment, a constant heavy chain region can include a C-terminal lysine. [00205] Amino acid sequences of exemplary Fc regions are provided in Table 13. Table 13. Fc Sequences IPTS/128623945.3 139 Attorney Docket No.
- PRG-013WO IPTS/128623945.3 140 Attorney Docket No. PRG-013WO IPTS/128623945.3 141 Attorney Docket No. PRG-013WO IPTS/128623945.3 142 Attorney Docket No. PRG-013WO IPTS/128623945.3 143 Attorney Docket No. PRG-013WO IPTS/128623945.3 144 Attorney Docket No. PRG-013WO IPTS/128623945.3 145 Attorney Docket No. PRG-013WO IPTS/128623945.3 146 Attorney Docket No. PRG-013WO IPTS/128623945.3 147 Attorney Docket No. PRG-013WO IPTS/128623945.3 148 Attorney Docket No.
- PRG-013WO IPTS/128623945.3 149 Attorney Docket No. PRG-013WO IPTS/128623945.3 150 Attorney Docket No. PRG-013WO IPTS/128623945.3 151 Attorney Docket No. PRG-013WO IPTS/128623945.3 152 Attorney Docket No. PRG-013WO IPTS/128623945.3 153 Attorney Docket No. PRG-013WO IPTS/128623945.3 154 Attorney Docket No. PRG-013WO IPTS/128623945.3 155 Attorney Docket No. PRG-013WO IPTS/128623945.3 156 Attorney Docket No. PRG-013WO IPTS/128623945.3 157 Attorney Docket No.
- PRG-013WO IPTS/128623945.3 158 Attorney Docket No. PRG-013WO IPTS/128623945.3 159 Attorney Docket No. PRG-013WO IPTS/128623945.3 160 Attorney Docket No. PRG-013WO IPTS/128623945.3 161 Attorney Docket No. PRG-013WO IPTS/128623945.3 162 Attorney Docket No. PRG-013WO IPTS/128623945.3 163 Attorney Docket No. PRG-013WO IPTS/128623945.3 164 Attorney Docket No. PRG-013WO IPTS/128623945.3 165 Attorney Docket No. PRG-013WO IPTS/128623945.3 166 Attorney Docket No.
- PRG-013WO IPTS/128623945.3 167 Attorney Docket No. PRG-013WO IPTS/128623945.3 168 Attorney Docket No. PRG-013WO IPTS/128623945.3 169 Attorney Docket No. PRG-013WO IPTS/128623945.3 170 Attorney Docket No. PRG-013WO IPTS/128623945.3 171 Attorney Docket No. PRG-013WO IPTS/128623945.3 172 Attorney Docket No. PRG-013WO IPTS/128623945.3 173 Attorney Docket No. PRG-013WO IPTS/128623945.3 174 Attorney Docket No.
- any bispecific antibody described herein may comprise a Fc comprising one or more modifications with any one of the Fc modifications described herein.
- any bispecific antibody described herein can comprise any one of the Fc sequences in Table 13 (SEQ ID NOs: 425-468, 484-539, 670-680, 689-691, and 693-804).
- the CH(1-3) domain of a bispecific antibody in Table 11a or Table 11b is substituted with any one of the Fc sequences in Table 13(SEQ ID NOs: 425- 468, 484-539, 670-680, 689-691, and 693-804).
- one or more modifications in the modified Fc is selected from the group consisting of: S298A, E333A, K334A, K326A, F243L, R292P, Y300L, V305I, P396L, F243L, R292P, Y300L, L235V, P396L, F243L, S239D, I332E, A330L, S267E, L328F, D265S, S239E, K326A, A327H, G237F, K326E, G236A, D270L, H268D, S324T, L234F, N325L, V266L, and S267D.
- one or more modifications in the modified Fc is selected from the group consisting of S228P, M252Y, S254T, T256E, T256D, T250Q, H285D, T307A, T307Q, T307R, T307W, L309D, Q411H, IPTS/128623945.3 175 Attorney Docket No. PRG-013WO Q311V, A378V, E380A, M428L, N434A, N434S, N297A, D265A, L234A, L235A, and N434W.
- the modified Fc comprises a specific combination of amino acid substitutions selected from the group consisting of: L234A/L235A; V234A/G237A; L235A/G237A/E318A; S228P/L236E; H268Q/V309L/A330S/A331S; C220S/C226S/C229S/P238S; C226S/C229S/E3233P/L235V/L235A; L234F/L235E/P331S; C226S/P230S; L234A/G237A; L234A/L235A/G237A; and L234A/L235A/P329G.
- the modified Fc comprises a specific combination of amino acid substitutions selected from the group consisting of M428L/N434S (LS); M252Y/S254T/T256E (YTE); T250Q/M428L; T307A/E380A/N434A; T256D/T307Q (DQ); T256D/T307W (DW); M252Y/T256D (YD); T307Q/Q311V/A378V (QVV); T256D/H285D/T307R/Q311V/A378V (DDRVV); L309D/Q311H/N434S (DHS); S228P/L235E (SPLE); L234A/L235A (LALA); M428L/N434A (LA); L234A/G237A (LAGA); L234A/L235A/G237A (LALAGA); L234A
- the modified Fc comprises a specific combination of amino acid substitutions selected from the group consisting of M428L/N434S (LS) and M252Y/S254T/T256E (YTE).
- the modified Fc comprises M428L/N434S (LS) (e.g., SEQ ID NO: 93 SEQ ID NO: 110, SEQ ID NO: 117) modifications.
- the modified Fc comprises M252Y/S254T/T256E (YTE) modifications.
- the bispecific antibodies described herein includes modifications to improve its ability to mediate effector function. Such modifications are IPTS/128623945.3 176 Attorney Docket No.
- the bispecific antibodies provided herein comprises a Fc domain (e.g., IgG1) with reduced fucose content at position Asn 297 (EU numbering) compared to a naturally occurring Fc domain.
- Fc domains are known to have improved ADCC.
- such antibodies do not comprise any fucose at position Asn 297.
- the bispecific antibodies described herein comprises an Fc region with one or more amino acid substitutions which improve ADCC, such as a substitution at one or more of positions 298, 333, and 334 of the Fc region.
- the bispecific antibodies provided herein comprises an Fc region with one or more amino acid substitutions at positions 239, 332, and 330.
- the Fc comprises an amino acid sequence having at least 80% sequence identity with the amino acid sequence according to any one of SEQ ID NOs in Table 13.
- the Fc comprises an amino acid sequence having at least 85% sequence identity with the amino acid sequence according to any one of SEQ ID NOs in Table 13.
- the Fc comprises an amino acid sequence having at least 90% sequence identity with the amino acid sequence according to any one of SEQ ID NOs in Table 13. In some embodiments, the Fc comprises an amino acid sequence having at least 95% sequence identity with the amino acid sequence according to any one of SEQ ID NOs in Table 13. In some embodiments, the Fc comprises an amino acid sequence having at least 96% sequence identity with the amino acid sequence according to any one of SEQ ID NOs in Table 13. In some embodiments, the Fc comprises an amino acid sequence having at least 97% sequence identity with the amino acid sequence according to any one of SEQ ID NOs in Table 13. In some embodiments, the Fc comprises an amino acid sequence having at least 98% sequence identity with the amino acid sequence according to any one of SEQ ID NOs in Table 13.
- the Fc comprises an amino acid sequence having at least 99% sequence identity with the amino acid sequence according to any one of SEQ ID NOs in Table 13. In some embodiments, the Fc comprises the amino acid sequence according to any one of SEQ ID NOs in Table 13.
- the bispecific antibodies described herein comprise an Fc region with at least one galactose residue in the oligosaccharide attached to the Fc region. Such variants may have improved CDC function. IPTS/128623945.3 177 Attorney Docket No. PRG-013WO [00215] In some embodiments, the bispecific antibodies described herein comprise one or more alterations that improves or diminishes C1q binding and/or CDC.
- the Fc region comprises one or more amino acid substitutions, wherein the one or more substitutions result in an increase in one or more of antibody half-life, ADCC activity, ADCP activity, or CDC activity compared with the Fc without the one or more substitutions.
- the one or more amino acid substitutions results in increased antibody half-life at pH 6.0 compared to an antibody comprising a wild-type Fc region.
- the bispecific antibody has an increased half-life that is about 10,000-fold, 1,000-fold, 500-fold, 100-fold, 50-fold, 20-fold, 10-fold, 9-fold, 8-fold, 7-fold, 6-fold, 5-fold, 4.5-fold, 4-fold, 3.5-fold, 3-fold, 2.5-fold, 2- fold, 1.95-fold, 1.9-fold, 1.85-fold, 1.8-fold, 1.75-fold, 1.7-fold, 1.65-fold, 1.6-fold, 1.55-fold, 1.50-fold, 1.45-fold, 1.4-fold, 1.35-fold, 1.3-fold, 1.25-fold, 1.2-fold, 1.15-fold, 1.1-fold, or 1.05-fold longer compared to an antibody comprising a wild-type Fc region.
- the Fc region comprises one or more amino acid substitutions, wherein the one or more substitutions result in a decrease in one or more of ADCC activity, ADCP activity, or CDC activity compared with the Fc without the one or more substitutions.
- the Fc region binds an Fc ⁇ Receptor selected from the group consisting of: Fc ⁇ RI, Fc ⁇ RIIa, Fc ⁇ RIIb, Fc ⁇ RIIc, Fc ⁇ RIIIa, and Fc ⁇ RIIIb.
- the Fc region binds an Fc ⁇ Receptor with higher affinity at pH 6.0 compared to an antibody comprising a wild-type Fc region.
- the bispecific antibodies described herein comprise an extended half-life (i.e., serum half-life). In some embodiments, the bispecific antibodies described herein comprise a half-life of at least about 14, 28, 42, 56, 70, 84, 96, or more than 96 weeks.
- the TSLP or TSLPR binding protein described herein comprises a half-life in a range of about 14 days to about 96 days, about 14 days to about 84 days, about 14 days to about 70 days, about 14 days to about 56 days, about 14 days to about 42 days, about 14 days to about 28 days, of about 28 days to about 96 days, about 28 days to about 84 days, about 28 days to about 70 days, about 28 days to about 56 days, about 28 days to about 42 days, of about 42 days to about 96 days, about 42 days to about 84 days, about 42 days to about 70 days, or about 42 days to about 56 days.
- the bispecific antibodies described herein comprise a half-life in a range of about 42 days to about 56 days.
- the bispecific antibodies described herein comprise a half-life of at least about 50 days. In some embodiments, the bispecific antibodies described IPTS/128623945.3 178 Attorney Docket No. PRG-013WO herein comprise a half-life of about 50 days. Methods of measuring half-life are known in the art. In some embodiments, the half-life is measured in a non-human primate. In some embodiments, the half-life is measured in a human. In some embodiments, the half-life is measured following intravenous administration. In some embodiments, the half-life is measured following subcutaneous administration. [00220] In some embodiments, the bispecific antibodies described herein have a half-life that is at least 20% longer than a comparator antibody.
- the comparator antibody comprises the same complementarity determining regions and variable regions but different Fc regions.
- the half-life of the bispecific antibodies described herein is at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, or at least 90% longer than the half-life of the comparator antibody.
- the half-life of the bispecific antibodies described herein is longer than the half-life of the comparator antibody by at least 2 fold, at least 3 fold, at least 4 fold, at least 5 fold, at least 6 fold, at least 7 fold, at least 8 fold, at least 9 fold, or at least 10 fold.
- Binding [00221] The affinity of a molecule X for its partner Y can be represented by the dissociation equilibrium constant (K D ).
- K D dissociation equilibrium constant
- the kinetic components that contribute to the dissociation equilibrium constant are described in more detail below.
- Affinity can be measured by common methods known in the art, including those described herein, such as surface plasmon resonance (SPR) technology (e.g., BIACORE®) or biolayer interferometry (e.g., FORTEBIO®).
- SPR surface plasmon resonance
- BIACORE® BIACORE®
- FORTEBIO® biolayer interferometry
- the terms “bind,” “specific binding,” “specifically binds to,” “specific for,” “selectively binds,” and “selective for” a particular antigen (e.g., a polypeptide target) or an epitope on a particular antigen mean binding that is measurably different from a non-specific or non-selective interaction (e.g., with a non-target molecule).
- Specific binding can be measured, for example, by measuring binding to a target molecule (i.e., IL-13. TSLP, or TSLPR) and comparing it to binding to a non-target molecule.
- Specific binding can also be determined by competition with a control molecule that mimics the epitope recognized on the target molecule. In that case, specific binding is indicated if the binding of the antibody to the target molecule is competitively inhibited by the control molecule.
- the affinity of a bispecific antibody disclosed here in for a non-target molecule is less than about 50% of the affinity for IL-13 TSLP, or TSLPR. In some embodiments, the affinity of a bispecific antibody disclosed here in for a non-target molecule is less than about 40% of the affinity for IL-13 TSLP, or TSLPR. IPTS/128623945.3 179 Attorney Docket No.
- the affinity of a bispecific antibody disclosed here in for a non-target molecule is less than about 40% of the affinity for IL-13 TSLP, or TSLPR. In some embodiments, the affinity of a bispecific antibody disclosed here in for a non-target molecule is less than about 20% of the affinity for IL-13 TSLP, or TSLPR. In some embodiments, the affinity of a bispecific antibody disclosed here in for a non-target molecule is less than about 10% of the affinity for IL-13 TSLP, or TSLPR. In some embodiments, the affinity of a bispecific antibody disclosed here in for a non-target molecule is less than about 1% of the affinity for IL-13 TSLP, or TSLPR.
- the affinity of a bispecific antibody disclosed here in for a non-target molecule is less than about 0.1% of the affinity for IL-13 TSLP, or TSLPR.
- the term “competes with” or “cross-competes with” indicates that the two or more antibodies compete for binding to an antigen (e.g., IL-13).
- IL-13 is coated on a surface and contacted with a first anti-IL-13 antibody, after which a second anti-IL-13 antibody is added.
- a first anti-IL-13 antibody is coated on a surface and contacted with IL-13, and then a second anti-IL-13 antibody is added.
- the antibodies compete with each other.
- the term “competes with” also includes combinations of antibodies where one antibody reduces binding of another antibody, but where no competition is observed when the antibodies are added in the reverse order.
- the first and second antibodies inhibit binding of each other, regardless of the order in which they are added.
- one antibody reduces binding of another antibody to its antigen by at least 25%, at least 50%, at least 60%, at least 70%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99% as measured in a competitive binding assay.
- a skilled artisan can select the concentrations of the antibodies used in the competition assays based on the affinities of the antibodies for IL-13 and the valency of the antibodies.
- the assays described in this definition are illustrative, and a skilled artisan can utilize any suitable assay to determine if antibodies compete with each other. Suitable assays are described, for example, in Cox et al., “Immunoassay Methods,” in Assay Guidance Manual [Internet], Updated December 24, 2014 (ncbi.nlm.nih.gov/books/NBK92434/; accessed September 29, 2015); Silman et al., Cytometry, 2001, 44:30-37; and Finco et al., J. Pharm.
- test antibody competes with a reference antibody if an excess of a test antibody (e.g., at least 2x, 5x, 10x, 20x, or 100x) inhibits or blocks binding of the reference antibody by, e.g., at least 50%, 60%, 70%, 75%, 80%, 85%, 90%, 95%, or 99% as measured in a competitive binding assay.
- a test antibody e.g., at least 2x, 5x, 10x, 20x, or 100x
- Antibodies identified by competition assay include antibodies binding to the same epitope as the reference antibody and antibodies binding to an adjacent epitope sufficiently proximal to the epitope bound by the reference antibody for steric hindrance to occur.
- a second, competing antibody can be identified that competes for binding to IL-13 with a first antibody described herein.
- the second antibody can block or inhibit binding of the first antibody by, e.g., at least 50%, 60%, 70%, 75%, 80%, 85%, 90%, 95%, or 99% as measured in a competitive binding assay.
- the second antibody can displace the first antibody by greater than 50%, 60%, 70%, 75%, 80%, 85%, 90%, 95%, or 99%.
- the antibody binds a human IL-13 and TSLP or TSLPR.
- the bispecific antibody binds an IL-13 sequence set forth in SEQ ID NOs: 472-475.
- the bispecific antibody is cross-reactive to cynomolgus monkey IL-13.
- the bispecific antibody is cross-reactive to cynomolgus monkey IL-13.
- the bispecific antibody binds to an IL-13 sequence set forth in SEQ ID NOs: 472-475 with a K D of less than or equal to about 1, 2, 3, 4, 5, 6, 7, 8, 9 x 10 -9 M, as measured by SPR. In certain embodiments, the bispecific antibody binds to an IL-13 sequence set forth in SEQ ID NOs: 472-475 with a K D of less than or equal to about 1 x 10 -10 M, as measured by SPR. In certain embodiments, the bispecific antibody binds to human IL-13 with a K D of less than or equal to about 1 x 10 -9 M, as measured by SPR.
- a bispecific antibody provided herein binds IL-13 with a K D of less than or equal to about 0.01, 0.02, 0.03, 0.04, 0.05, 0.06, 0.07, 0.08, 0.09, 0.1, 0.2, 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9, 1, 1.1, 1.2, 1.3, 1.4, 1.5, 1.6, 1.7, 1.8, 1.9, 1.95, 2, 2.5, 3, 3.5, 4, 4.5, 5, 6, 7, 8, 9, or 10 x 10 -8 M, as measured by ELISA or any other suitable method known in the art.
- an antibody provided herein binds IL-13 with a K D of less than or equal to about 0.01, 0.02, 0.03, 0.04, 0.05, 0.06, 0.07, 0.08, 0.09, 0.1, 0.2, 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9, 1, 1.1, 1.2, 1.3, 1.4, 1.5, 1.6, 1.7, 1.8, 1.9, 1.95, 2, 2.5, 3, 3.5, 4, 4.5, 5, 6, 7, 8, 9, or 10 x 10 -9 M, as measured by ELISA or any other suitable method known in the art.
- IPTS/128623945.3 181 Attorney Docket No.
- the K D of the antibody provided herein for the binding of IL-13, TSLP, or TSLPR is between about 0.001-0.01, 0.01-0.1, 0.01-0.05, 0.05-0.1, 0.1-0.5, 0.5-1, 0.25-0.75, 0.25-0.5, 0.5-0.75, 0.75-1, 0.75-2, 1.1-1.2, 1.2-1.3, 1.3-1.4, 1.4-1.5, 1.5-1.6, 1.6-1.7, 1.7-1.8, 1.8-1.9, 1.9-2, 1-2, 1-5, 2-7, 3-8, 3-5, 4-6, 5-7, 6-8, 7-9, 7-10, or 5-10 x 10 -8 M, as measured by ELISA or any other suitable method known in the art.
- an antibody provided herein binds IL-13, TSLP, or TSLPR with a KD of less than or equal to about 1 x 10 -8 M, or less than or equal to above 1 x 10 -9 M as measured by ELISA or any other suitable method known in the art.
- the antibody provided herein binds IL-13, TSLP, or TSLPR with a KD of less than or equal to about 10, 9, 8, 7, 6, 5, 4.5, 4, 3.5, 3, 2.5, 2, 1.98, 1.95, 1.9, 1.85, 1.8, 1.75, 1.7, 1.65, 1.6, 1.55, 1.50, 1.45, 1.4, 1.3, 1.2, 1.1, 1, 0.9, 0.85, 0.8, 0.75, 0.7, 0.65, 0.6, 0.55, 0.5, 0.45, 0.4, 0.35, 0.3, 0.25, 0.2, 0.15, 0.1, 0.05, 0.01, 0.005, 0.001, 0.0005, or 0.0001 x 10 -8 M, or less, as measured by ELISA or any other suitable method known in the art .
- the antibody provided herein binds IL-13, TSLP, or TSLPR with a K D between 5-3, 4-2, 3-1, 1.9-1.8, 1.8-1.7, 1.7-1.6, 1.6-1.5, 1.9-1.5, 1.5-1, 1-0.8, 1-0.5, 0.9-0.6, 0.7-0.4, 0.6-0.2, 0.5-0.3, 0.3-0.2, 0.2-0.1, 0.1-0.01, 0.01-0.001, or 0.001-0.0001 x 10 -8 M as measured by ELISA or any other suitable method known in the art.
- Function [00233] “Effector functions” refer to those biological activities mediated by the Fc region of an antibody, which activities may vary depending on the antibody isotype.
- compositions comprising the bispecific antibodies including pharmaceutical compositions comprising any one or more of the bispecific antibodies described herein with one or more pharmaceutically acceptable excipients.
- the composition is sterile.
- compositions generally comprise an effective amount of a bispecific antibody.
- compositions can comprise, in addition to one or more of the bispecific antibodies disclosed herein, a pharmaceutically acceptable excipient, carrier, buffer, stabilizer IPTS/128623945.3 182 Attorney Docket No. PRG-013WO or other materials well known to those skilled in the art. Such materials should be non-toxic and should not interfere with the efficacy of the active ingredient.
- the precise nature of the carrier or other material can depend on the route of administration, e.g., oral, intravenous, cutaneous or subcutaneous, nasal, intramuscular, intraperitoneal routes.
- Pharmaceutical compositions for oral administration can be in tablet, capsule, powder or liquid form.
- a tablet can include a solid carrier such as gelatin or an adjuvant.
- Liquid pharmaceutical compositions generally include a liquid carrier such as water, petroleum, animal or vegetable oils, mineral oil or synthetic oil. Physiological saline solution, dextrose or other saccharide solution or glycols such as ethylene glycol, propylene glycol or polyethylene glycol can be included. [00237] For intravenous, cutaneous or subcutaneous injection, or injection at the site of affliction, the active ingredient will be in the form of a parenterally acceptable aqueous solution which is pyrogen-free and has suitable pH, isotonicity and stability.
- bispecific anti-IL-13/TSLP or TSLPR antibodies that is to be given to an individual, administration is preferably in a “therapeutically effective amount” or “prophylactically effective amount” (as the case can be, although prophylaxis can be considered therapy), this being sufficient to show benefit to the individual.
- a “therapeutically effective amount” or “prophylactically effective amount” as the case can be, although prophylaxis can be considered therapy
- Prescription of treatment is within the responsibility of general practitioners and other medical doctors, and typically takes account of the disorder to be treated, the condition of the individual patient, the site of delivery, the method of administration and other factors known to practitioners. Examples of the techniques and protocols mentioned above can be found in Remington’s Pharmaceutical Sciences, 16 th edition, Osol, A. (ed), 1980.
- a composition can be administered alone or in combination with other treatments, either simultaneously or sequentially dependent upon the condition to be treated. Methods IPTS/128623945.3 183 Attorney Docket No. PRG-013WO Methods of Preparation [0001] Methods for making bispecific antibodies are known in the art.
- Bispecific antibodies include cross-linked or “heteroconjugate” or “heterodimer” antibodies.
- one of the antibodies in the heterodimer can be coupled to avidin, the other to biotin.
- Heterodimer antibodies may be made using any convenient cross-linking method. Suitable cross-linking agents are well known in the art, and are disclosed in U.S. Pat.
- bispecific antibodies described herein can be produced using recombinant methods and compositions, e.g., as described in U.S. Pat. No.4,816,567.
- an isolated nucleic acid encoding a bispecific antibody described herein is provided.
- Such a nucleic acid may encode an amino acid sequence comprising the VL sequence(s) and/or an amino acid sequence comprising the VH sequence(s) of the antibody (e.g., the light and/or heavy chains of the antibody).
- one or more vectors e.g., expression vectors comprising such nucleic acids are provided.
- the nucleic acid is provided in a multicistronic vector.
- a host cell comprising such nucleic acid is provided.
- a host cell comprises (e.g., has been transformed with): (1) a vector comprising a nucleic acid that encodes an amino acid sequence comprising the VL sequence(s) of the antibody and an amino acid sequence comprising the VH sequence(s) of the antigen-binding polypeptide construct, or (2) a first vector comprising a nucleic acid that encodes an amino acid sequence comprising the VL sequence(s) of the antigen-binding polypeptide construct and a second vector comprising a nucleic acid that encodes an amino acid sequence comprising the VH sequence(s) of the antigen-binding polypeptide construct.
- the host cell is eukaryotic, e.g., a Chinese Hamster Ovary (CHO) cell, or human embryonic kidney (HEK) cell, or lymphoid cell (e.g., Y0, NS0, Sp20 cell).
- a method of making a bispecific antibody comprises culturing a host cell comprising nucleic acid encoding the bispecific antibody, as provided above, under conditions suitable for expression of the antibody, and optionally recovering the antibody from the host cell (or host cell culture medium).
- nucleic acid encoding an antibody is isolated and inserted into one or more vectors for further cloning and/or expression in a host cell.
- nucleic acid may be readily isolated and sequenced using conventional procedures (e.g., by using oligonucleotide probes that are capable of binding specifically to genes encoding the heavy and light chains of the antibody).
- the protein in certain embodiments is present at about 30%, about 25%, about 20%, about 15%, about 10%, about 5%, about 4%, about 3%, about 2%, or about 1% or less of the dry weight of the cells.
- the protein in certain embodiments, is present in the culture medium at about 5 g/L, about 4 g/L, about 3 g/L, about 2 g/L, about 1 g/L, about 750 mg/L, about 500 mg/L, about 250 mg/L, about 100 mg/L, about 50 mg/L, about 10 mg/L, or about 1 mg/L or less of the dry weight of the cells.
- substantially purified antibody produced by the methods described herein has a purity level of at least about 30%, at least about 35%, at least about 40%, at least about 45%, at least about 50%, at least about 55%, at least about 60%, at least about 65%, at least about 70%, specifically, a purity level of at least about 75%, 80%, 85%, and more specifically, a purity level of at least about 90%, a purity level of at least about 95%, a purity level of at least about 99% or greater as determined by appropriate methods such as SDS/PAGE analysis, RP-HPLC, SEC, and capillary electrophoresis.
- Suitable host cells for cloning or expression of antibody-encoding vectors include prokaryotic or eukaryotic cells described herein.
- Recombinant host cells or host cells are cells that include an exogenous polynucleotide, regardless of the method used for insertion, for example, direct uptake, transduction, f-mating, or other methods known in the art to create recombinant host cells.
- the exogenous polynucleotide may be maintained as a nonintegrated vector, for example, a plasmid, or alternatively, may be integrated into the host genome.
- Host cells can include CHO, derivatives of CHO, NS0, Sp2O, CV-1, VERO-76, HeLa, HepG2, Per.C6, or BHK.
- antibody may be produced in bacteria, in particular when glycosylation and Fc effector function are not needed.
- U.S. Pat. Nos.5,648,237, 5,789,199, and 5,840,523. See also Charlton, Methods in Molecular Biology, Vol.248 (B.K.C. Lo, ed., Humana Press, Totowa, N.J., 2003), pp.245-254, describing expression of antibody fragments in E.
- the antibody may be isolated from the bacterial cell paste in a soluble fraction and can be further purified.
- eukaryotic microbes such as filamentous fungi or yeast are suitable cloning or expression hosts for antibody-encoding vectors, including fungi and yeast strains whose glycosylation pathways have been “humanized,” resulting in the production of an antibody with a partially or fully human glycosylation pattern. See Gerngross, Nat. Biotech.22:1409-1414 (2004), and Li et al., Nat. Biotech.24:210-215 (2006).
- Suitable host cells for the expression of glycosylated antibodies are also derived from multicellular organisms (invertebrates and vertebrates). Examples of invertebrate cells include plant and insect cells. Numerous baculoviral strains have been identified which may be used in conjunction with insect cells, particularly for transfection of Spodoptera frugiperda cells.
- Plant cell cultures can also be utilized as hosts. See, e.g., U.S. Pat. Nos.5,959,177, 6,040,498, 6,420,548, 7,125,978, and 6,417,429 (describing PLANTIBODIESTM technology for producing antibodies in transgenic plants).
- Vertebrate cells may also be used as hosts.
- mammalian cell lines that are adapted to grow in suspension may be useful.
- useful mammalian host cell lines are monkey kidney CV1 line transformed by SV40 (COS-7); human embryonic kidney line (293 or 293 cells as described, e.g., in Graham et al., J. Gen Virol. 36:59 (1977)); baby hamster kidney cells (BHK); mouse sertoli cells (TM4 cells as described, e.g., in Mather, Biol.
- monkey kidney cells (CV1); African green monkey kidney cells (VERO-76); human cervical carcinoma cells (HELA); canine kidney cells (MDCK; buffalo rat liver cells (BRL 3A); human lung cells (W138); human liver cells (Hep G2); mouse mammary tumor (MMT 060562); TRI cells, as described, e.g., in Mather et al., Annals N.Y. Acad. Sci.383:44-68 (1982); MRC 5 cells; and FS4 cells.
- Other useful mammalian host cell lines include Chinese hamster ovary (CHO) cells, including DHFR ⁇ CHO cells (Urlaub et al., Proc. Natl. Acad. Sci.
- the antibodies described herein are produced in stable mammalian cells, by a method comprising: transfecting at least one stable mammalian cell with: nucleic acid encoding the antibody, in a predetermined ratio; and expressing the nucleic IPTS/128623945.3 186 Attorney Docket No. PRG-013WO acid in the at least one mammalian cell.
- the predetermined ratio of nucleic acid is determined in transient transfection experiments to determine the relative ratio of input nucleic acids that results in the highest percentage of the antibody in the expressed product.
- the method of producing a glycosylated antibody in stable mammalian cells described herein said method comprising identifying and purifying the desired glycosylated antibody.
- the said identification is by one or both of liquid chromatography and mass spectrometry.
- the antibodies can be purified or isolated after expression.
- Proteins may be isolated or purified in a variety of ways known to those skilled in the art.
- Standard purification methods include chromatographic techniques, including ion exchange, hydrophobic interaction, affinity, sizing or gel filtration, and reversed-phase, carried out at atmospheric pressure or at high pressure using systems such as FPLC and HPLC.
- Purification methods also include electrophoretic, immunological, precipitation, dialysis, and chromatofocusing techniques. Ultrafiltration and diafiltration techniques, in conjunction with protein concentration, are also useful.
- a variety of natural proteins bind Fc and antibodies, and these proteins can find use in the present invention for purification of antibodies.
- the bacterial proteins A and G bind to the Fc region.
- the bacterial protein L binds to the Fab region of some antibodies.
- Purification can often be enabled by a particular fusion partner.
- antibodies may be purified using glutathione resin if a GST fusion is employed, Ni +2 affinity chromatography if a His-tag is employed or immobilized anti-flag antibody if a flag-tag is used.
- suitable purification techniques see, e.g., incorporated entirely by reference Protein Purification: Principles and Practice, 3 rd Ed., Scopes, Springer-Verlag, NY, 1994, incorporated entirely by reference.
- the degree of purification necessary will vary depending on the use of the antibodies. In some instances, no purification is necessary.
- the antibodies are purified using Anion Exchange Chromatography including, but not limited to, chromatography on Q-sepharose, DEAE sepharose, poros HQ, poros DEAF, Toyopearl Q, Toyopearl QAE, Toyopearl DEAE, Resource/Source Q and DEAE, Fractogel Q and DEAE columns.
- Anion Exchange Chromatography including, but not limited to, chromatography on Q-sepharose, DEAE sepharose, poros HQ, poros DEAF, Toyopearl Q, Toyopearl QAE, Toyopearl DEAE, Resource/Source Q and DEAE, Fractogel Q and DEAE columns.
- IPTS/128623945.3 187 Attorney Docket No.
- the proteins described herein are purified using Cation Exchange Chromatography including, but not limited to, SP-sepharose, CM sepharose, poros HS, poros CM, Toyopearl SP, Toyopearl CM, Resource/Source S and CM, Fractogel S and CM columns and their equivalents and comparables.
- Cation Exchange Chromatography including, but not limited to, SP-sepharose, CM sepharose, poros HS, poros CM, Toyopearl SP, Toyopearl CM, Resource/Source S and CM, Fractogel S and CM columns and their equivalents and comparables.
- antibodies described herein can be chemically synthesized using techniques known in the art (e.g., see Creighton, 1983, Proteins: Structures and Molecular Principles, W. H. Freeman & Co., N.Y and Hunkapiller et al., Nature, 310:105-111 (1984)).
- polypeptide corresponding to a fragment of a polypeptide can be synthesized by use of a peptide synthesizer.
- nonclassical amino acids or chemical amino acid analogs can be introduced as a substitution or addition into the polypeptide sequence.
- Non-classical amino acids include, but are not limited to, to the D-isomers of the common amino acids, 2,4diaminobutyric acid, alpha-amino isobutyric acid, 4aminobutyric acid, Abu, 2-amino butyric acid, g-Abu, e-Ahx, 6amino hexanoic acid, Aib, 2-amino isobutyric acid, 3-amino propionic acid, ornithine, norleucine, norvaline, hydroxyproline, sarcosine, citrulline, homocitrulline, cysteic acid, t-butylglycine, t-butylalanine, phenylglycine, cyclohexylalanine, alanine, fluoro-amino acids, designer amino acids such as methyl amino acids, C-methyl amino acids, N-methyl amino acids, and amino acid analogs in general.
- an antibody described herein has an aggregation temperature greater than about 69 °C, greater than about 70 °C, greater than about 71 °C, greater than about 72 °C, greater than about 73 °C, greater than about 74 °C, greater than about 75 °C, or greater than about 76 °C, for example, between about 69 °C and about 77 °C, between about 70 °C and about 76 °C, between about 71 °C and about 75 °C.
- aggregation temperature is measured using DSF.
- an antibody described herein has reduced hydrophobicity as compared to lebrikizumab as measured by hydrophobic interaction chromatography (HIC). In certain embodiments, the antibody exhibits an HIC retention time that is less than about 15.2 min. In certain embodiments, the antibody exhibits an HIC retention time that is between about 13 min and about 15 min.
- Methods of Use [00259] In an aspect, the present application provides methods of contacting IL-13 with an bispecific antibody described herein, which results in inhibition of IL-13 binding to an IL-13 receptor expressed on a cell and/or the inhibition of TSLP binding to TSLPR on a cell. IPTS/128623945.3 188 Attorney Docket No.
- the present application provides methods of using the bispecific antibodies described herein for treatment of a disorder or disease in a subject.
- described herein is a method for treating a subject in need thereof with a bispecific antibody, the method comprising administering to a mammalian subject a therapeutically effective amount of a bispecific antibody or pharmaceutical composition comprising a bispecific antibody described herein.
- the present application provides methods of treating a disorder or disease associated with elevated levels of IL-13 and/or IgE and/or TSLP in a subject.
- described herein are methods for treating a pathology associated with IL-13 and/or TSLP activity, the method comprising administering to a mammalian subject a therapeutically effective amount a bispecific antibody or a pharmaceutical composition comprising a bispecific antibody described herein.
- described herein is a method for treating an inflammatory disorder or disease in a mammalian subject in need thereof, the method comprising administering to the mammalian subject a therapeutically effective amount an antibody described herein or a pharmaceutical composition described herein.
- the inflammatory disorder or disease is atopic dermatitis.
- the inflammatory disorder or disease is asthma.
- the inflammatory disorder or disease is idiopathic pulmonary fibrosis. In certain embodiments of the methods described herein, the inflammatory disorder or disease is alopecia areata. In certain embodiments, the inflammatory disorder or disease is chronic sinusitis with nasal polyps. In certain embodiments, the inflammatory disorder or disease is Chronic Rhinosinusitis without Nasal Polyps (CRSsNP). In certain embodiments, the inflammatory disorder or disease is eosinophilic esophagitis (EoE).
- the inflammatory disorder or disease is an Eosinophilic gastrointestinal disorder or disease (ENID) selected from the group consisting of Eosinophilic Gastritis (EoG), Eosinophilic enteritis (EoN), Eosinophilic colitis (EoC), and Eosinophilic Gastroenteritis (EGE).
- EoG Eosinophilic Gastritis
- EoN Eosinophilic enteritis
- EoC Eosinophilic colitis
- EGE Eosinophilic Gastroenteritis
- the inflammatory disorder or disease is Churg-Strauss syndrome/Eosinophilic granulomatosis with polyangiitis (EGPA).
- the inflammatory disorder or disease is Prurigo Nodularis (PN).
- the inflammatory disorder or disease is Chronic Spontaneous Urticaria (CSU).
- the inflammatory disorder or disease is Chronic Pruritis of Unknown Origin (CPUO).
- the inflammatory disorder or disease is Bullous Pemphigoid (BP).
- the inflammatory disorder or disease is Cold Inducible Urticaria (ColdU).
- the inflammatory disorder or disease is Allergic Fungal Rhinosinusitis (AFRS).
- AFRS Allergic Fungal Rhinosinusitis
- ABPA Allergic Bronchopulmonary Aspergillosis
- the inflammatory disorder or disease is Chronic Obstructive Pulmonary Disease (COPD).
- COPD Chronic Obstructive Pulmonary Disease
- the inflammatory disorder or disease is inflammatory bowel disease, such as Crohn disease or ulcerative colitis. In certain embodiments, the inflammatory disorder or disease is psoriasis. In certain embodiments, the inflammatory disorder or disease is lupus. In certain embodiments, the inflammatory disorder or disease is rheumatoid arthritis. [00263] In certain aspects, described herein are methods for treating a pathology associated with elevated levels of IL-13 in a mammalian subject in need thereof, the method comprising administering to the mammalian subject a therapeutically effective amount an antibody or a pharmaceutical composition described herein.
- described herein are methods of reducing biological activity of IL-13 and/or TSLP in a mammalian subject in need thereof, the method comprising administering to the mammalian subject a therapeutically effective amount an antibody or a pharmaceutical composition described herein.
- described herein are methods for inhibiting the TH2 type allergic response in a mammalian subject in need thereof, the method comprising administering to the mammalian subject a therapeutically effective amount of a bispecific antibody or a pharmaceutical composition described herein.
- described herein are methods for inhibiting IL-13-induced phosphorylation of STAT6 in a cell, the method comprising contacting the cell with an antibody described herein.
- described herein are methods for inhibiting IL-13-induced CD23 expression in a cell, the method comprising contacting the cell with a bispecific antibody described herein.
- described herein are methods for inhibiting IL-13-induced secretion of CCL2 and CCL26 from a cell, the method comprising contacting the cell with a bispecific antibody described herein.
- described herein are methods for inhibiting IL-13-induced NTRK1 expression in a cell, the method comprising contacting the cell with a bispecific antibody described herein.
- described herein are methods for reducing levels of Thymus and Activation Regulated Chemokine (TARC)/CCL17 in a mammalian subject in need IPTS/128623945.3 190 Attorney Docket No. PRG-013WO thereof, the method comprising administering to the mammalian subject a therapeutically effective amount a bispecific antibody or a pharmaceutical composition described herein.
- TARC Thymus and Activation Regulated Chemokine
- the methods provided herein are useful for the treatment of a disease or disorder in an individual.
- the individual is a human and the antibody is an anti-IL-13 antibody described herein.
- an antibody is administered intravenously, intramuscularly, subcutaneously, topically, orally, transdermally, intraperitoneally, intraorbitally, by implantation, by inhalation, intrathecally, intraventricularly, or intranasally.
- An effective amount of a bispecific antibody may be administered for the treatment of a disease or disorder.
- an antibody provided herein is administered with at least one additional therapeutic agent.
- Any suitable additional therapeutic or immunotherapeutic agent may be administered with an antibody provided herein.
- Additional therapeutic agents include agents that are used to treat or prevent a disease or disorder such as, but not limited to, an inflammatory disease or disorder associated with elevated levels of IL-13 and/or IgE and/or TSLP.
- the additional therapeutic agent can be administered by any suitable means.
- an antibody provided herein and the additional therapeutic agent are included in the same pharmaceutical composition. In some embodiments, an antibody provided herein and the additional therapeutic agent are included in different pharmaceutical compositions. [00276] In embodiments where a bispecific antibody provided herein and the additional therapeutic agent are included in different pharmaceutical compositions, administration of the bispecific antibody can occur prior to, simultaneously, and/or following, administration of the additional therapeutic agent. In some embodiments, administration of an antibody provided herein and the additional therapeutic agent occur within about one month of each other. In IPTS/128623945.3 191 Attorney Docket No. PRG-013WO some embodiments, administration of a bispecific antibody provided herein and the additional therapeutic agent occur within about one week of each other.
- kits and Articles of Manufacture The present application provides kits comprising any one or more of the bispecific antibody compositions described herein and instructions for use. In some embodiments, the kits further contain a component selected from any of secondary antibodies, reagents for immunohistochemistry analysis, pharmaceutically acceptable excipient and instruction manual and any combination thereof.
- the kit comprises a pharmaceutical composition comprising any one or more of the bispecific antibody compositions described herein, with one or more pharmaceutically acceptable excipients.
- the present application also provides articles of manufacture comprising any one of the bispecific antibody compositions or kits described herein. Examples of an article of manufacture include vials (including sealed vials).
- EXAMPLES [00279] Provided herein are examples of specific embodiments for carrying out the present invention. The examples are offered for illustrative purposes only, and are not intended to limit the scope of the present invention in any way. Efforts have been made to ensure accuracy with respect to numbers used (e.g., amounts, temperatures, etc.), but some experimental error and deviation should, of course, be allowed for.
- CHO cells at approximately 5.5 ⁇ 10 6 /mL in a shake flask were used as the host. Transfection was initiated by adding a mixture of 1 mg/L DNA and 7 mg/L PEI in OptiMEMTM medium (Invitrogen) to the cells followed by gentle mixing. Cells were then cultured in an incubator shaker at 120 rpm, 37°C, and 8% CO2, for 9 days. Feeding with peptone and glucose was carried out 24 h later and every 2-3 days thereafter depending on the cell density and viability. The cell culture was terminated on day 9 when cell viability reduced to ⁇ 80%. The conditioned medium was harvested for protein purification.
- OptiMEMTM medium Invitrogen
- a Biacore 8K SPR system (GE HealthCare) equipped with Series S Sensor Chip Protein G (Cytiva, Cat.29179315) was used to determine the binding kinetic rate and affinity constants at 25 ⁇ C and in a running buffer of HBS-EP+ (10 mM HEPES pH 7.4, 150 mM NaCl, 3 mM EDTA, 0.05% Surfactant P20). Following a stabilization period in running buffer, the bispecific constructs (diluted to 1 ⁇ g/mL) were captured onto flow cell 2 (active) for 60 sec at a flow rate of 10 ⁇ L/min.
- Recombinant Human IL13, TSLP and/or TSLPR Protein, His Tag was prepared at concentrations of 0, 0.39, 0.78, 1.56, 3.13, 6.25, 12.5 and 0 nM and injected over flow cell 1 (reference) and flow cell 2 (active) for 180 sec at a flow rate of 30 ⁇ L/min.
- Recombinant Cynomolgus IL13, TSLP and/or TSLPR Protein, His Tag was prepared at concentrations of 0, 0.39, 0.78, 1.56, 3.13, 6.25, 12.5, 25 and 0 nM and injected over flow cell 1 (reference) and flow cell 2 (active) for 180 sec at a flow rate of 30 ⁇ L/min.
- HEK293 previously transduced to stably express both hIL-13/IL4a and/or hTSLP/TSLPR are cultured and harvested. Cells are seeded at 200,000 cells in 100 ⁇ L per well. Cells are washed and the supernatant is discarded. A 100 ⁇ L mixture of biotinylated hIL-13 and/or hTSLP and purified bispecific antibody (1:1 by volume) is made and incubated for 1 hour.
- the incubated mixture IPTS/128623945.3 194 Attorney Docket No. PRG-013WO is added to resuspend the cells, resulting in a final concentration of 0.05 ug/mL of hIL-13 and/or hTSLP and 0-100 nM of purified bispecific antibody.
- the cells are stained in this mixture at 4 °C for 1 hour. Cells are then washed and stained with 100 ⁇ L of Alexa Fluor 488-conjugated streptavidin at a 1:1000 dilution to detect binding of biotinylated hIL-13 on the cell surface. Cells are incubated at 4 °C for 1 hour, protected from light.
- MFI median fluorescence intensity
- test articles Serial diluted test articles (duplicate, 1/5 diluted from 10 nM, 7 dose + Blank) are mixed with 5 ng/ml Biotin- TSLP for 30 min at RT. Then, test antibody (TA)-TSLP mixture is added to blocked plates and incubated for 1 h at 37°C. After washing, plates are incubated with Streptavidin-HRP for 1 h at 37°C. After wash, TMB is added to each well and incubated at room temperature until color developed (approximately 10 min). Reactions are stopped by addition of 1N HCl and optical density (OD) is read at 450 nm. Inhibition% is calculated as 1- (OD450 of sample/OD450 of ‘Ligand only’).
- IC50 of TAs is calculated through non-linear regression Inhibition of IL-13 or TSLP Binding to IL-13/IL4a or TSLP/TSLPR Overexpressing Cells [00288] IL-13 and/or TSLP binding to cells overexpressing IL-13/IL4R ⁇ and/or TSLP/TSLPR is used to evaluate the functional blockade of antibodies against this binding interaction. It is expected that the cell-line-based assays will show that the bispecific antibodies will exhibit a low IC50 and inhibit IL-13 and/or TSLP binding on an IL-13/IL4R ⁇ or TSLP/TSLPR overexpression cell line.
- Example 1 Engineered bispecific antibodies exhibit improved affinity and potency of blockade of IL-13/IL4a and/or TSLP/TSLPR Results IPTS/128623945.3 195 Attorney Docket No. PRG-013WO Determination of Antibody Affinity to IL-13 and/or TSLP or TSLPR [00289] Using the methods described above, the affinity of bispecific antibodies to IL-13 and/or TSLP/TSLPR and the binding kinetics thereof were assessed using surface plasmon resonance (SPR). [00290] As measure by SPR, the bispecific antibodies bound to human IL-13 and/or TSLP with sub-nanomolar affinity. The results are summarized in Table 14. Table 14. IPTS/128623945.3 196 Attorney Docket No.
- PRG-013WO Informal sequence listing IPTS/128623945.3 197 Attorney Docket No. PRG-013WO IPTS/128623945.3 198 Attorney Docket No. PRG-013WO IPTS/128623945.3 199 Attorney Docket No. PRG-013WO IPTS/128623945.3 200 Attorney Docket No. PRG-013WO IPTS/128623945.3 201 Attorney Docket No. PRG-013WO IPTS/128623945.3 202 Attorney Docket No. PRG-013WO IPTS/128623945.3 203 Attorney Docket No. PRG-013WO IPTS/128623945.3 204 Attorney Docket No. PRG-013WO IPTS/128623945.3 205 Attorney Docket No.
- PRG-013WO IPTS/128623945.3 206 Attorney Docket No. PRG-013WO IPTS/128623945.3 207 Attorney Docket No. PRG-013WO IPTS/128623945.3 208 Attorney Docket No. PRG-013WO IPTS/128623945.3 209 Attorney Docket No. PRG-013WO IPTS/128623945.3 210 Attorney Docket No. PRG-013WO IPTS/128623945.3 211 Attorney Docket No. PRG-013WO IPTS/128623945.3 212 Attorney Docket No. PRG-013WO IPTS/128623945.3 213 Attorney Docket No. PRG-013WO IPTS/128623945.3 214 Attorney Docket No.
- PRG-013WO IPTS/128623945.3 215 Attorney Docket No. PRG-013WO IPTS/128623945.3 216
- Attorney Docket No. PRG-013WO IPTS/128623945.3 220 Attorney Docket No. PRG-013WO IPTS/128623945.3 221
- PRG-013WO IPTS/128623945.3 224 Attorney Docket No. PRG-013WO IPTS/128623945.3 225 Attorney Docket No. PRG-013WO IPTS/128623945.3 226 Attorney Docket No. PRG-013WO IPTS/128623945.3 227 Attorney Docket No. PRG-013WO IPTS/128623945.3 228 Attorney Docket No. PRG-013WO IPTS/128623945.3 229 Attorney Docket No. PRG-013WO IPTS/128623945.3 230 Attorney Docket No. PRG-013WO IPTS/128623945.3 231 Attorney Docket No. PRG-013WO IPTS/128623945.3 232 Attorney Docket No.
- PRG-013WO IPTS/128623945.3 233 Attorney Docket No. PRG-013WO IPTS/128623945.3 234 Attorney Docket No. PRG-013WO IPTS/128623945.3 235 Attorney Docket No. PRG-013WO IPTS/128623945.3 236 Attorney Docket No. PRG-013WO IPTS/128623945.3 237 Attorney Docket No. PRG-013WO IPTS/128623945.3 238 Attorney Docket No. PRG-013WO IPTS/128623945.3 239 Attorney Docket No. PRG-013WO IPTS/128623945.3 240 Attorney Docket No. PRG-013WO IPTS/128623945.3 241 Attorney Docket No.
- PRG-013WO IPTS/128623945.3 242 Attorney Docket No. PRG-013WO IPTS/128623945.3 243 Attorney Docket No. PRG-013WO IPTS/128623945.3 244 Attorney Docket No. PRG-013WO IPTS/128623945.3 245 Attorney Docket No. PRG-013WO IPTS/128623945.3 246 Attorney Docket No. PRG-013WO IPTS/128623945.3 247 Attorney Docket No. PRG-013WO IPTS/128623945.3 248 Attorney Docket No. PRG-013WO IPTS/128623945.3 249 Attorney Docket No. PRG-013WO IPTS/128623945.3 250 Attorney Docket No.
- PRG-013WO IPTS/128623945.3 251 Attorney Docket No. PRG-013WO IPTS/128623945.3 252 Attorney Docket No. PRG-013WO IPTS/128623945.3 253 Attorney Docket No. PRG-013WO IPTS/128623945.3 254 Attorney Docket No. PRG-013WO IPTS/128623945.3 255 Attorney Docket No. PRG-013WO IPTS/128623945.3 256 Attorney Docket No. PRG-013WO IPTS/128623945.3 257 Attorney Docket No. PRG-013WO IPTS/128623945.3 258 Attorney Docket No. PRG-013WO IPTS/128623945.3 259 Attorney Docket No. PRG-013WO IPTS/128623945.3 260
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Life Sciences & Earth Sciences (AREA)
- Genetics & Genomics (AREA)
- Medicinal Chemistry (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Immunology (AREA)
- Peptides Or Proteins (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
Description
Claims
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2024313862A AU2024313862A1 (en) | 2023-06-22 | 2024-06-21 | Antibodies that bind interleukin 13 and tslp or tslpr and methods of use |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363522631P | 2023-06-22 | 2023-06-22 | |
| US63/522,631 | 2023-06-22 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2024264003A2 true WO2024264003A2 (en) | 2024-12-26 |
| WO2024264003A3 WO2024264003A3 (en) | 2025-02-20 |
Family
ID=93936320
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/035140 Pending WO2024264003A2 (en) | 2023-06-22 | 2024-06-21 | Antibodies that bind interleukin 13 and tslp or tslpr and methods of use |
Country Status (2)
| Country | Link |
|---|---|
| AU (1) | AU2024313862A1 (en) |
| WO (1) | WO2024264003A2 (en) |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ES2967485T3 (en) * | 2003-12-23 | 2024-04-30 | Genentech Inc | New anti-IL 13 antibodies and their uses |
| US7982016B2 (en) * | 2007-09-10 | 2011-07-19 | Amgen Inc. | Antigen binding proteins capable of binding thymic stromal lymphopoietin |
| TWI861302B (en) * | 2019-12-09 | 2024-11-11 | 比利時商艾伯霖克斯公司 | Polypeptides comprising immunoglobulin single variable domains targeting il-13 and tslp |
-
2024
- 2024-06-21 WO PCT/US2024/035140 patent/WO2024264003A2/en active Pending
- 2024-06-21 AU AU2024313862A patent/AU2024313862A1/en active Pending
Also Published As
| Publication number | Publication date |
|---|---|
| WO2024264003A3 (en) | 2025-02-20 |
| AU2024313862A1 (en) | 2026-01-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12358979B2 (en) | Antibodies that bind interleukin 13 and methods of use | |
| JP2024525188A (en) | Antibodies that bind to human fibrin or fibrinogen gamma C domain and methods of use - Patents.com | |
| AU2024261144A1 (en) | Antibodies that bind ox40l and methods of use | |
| US20240279357A1 (en) | ANTIBODIES WHICH BIND HUMAN FIBRIN OR FIBRINOGEN yC DOMAIN AND METHODS OF USE | |
| EP4665757A2 (en) | Antibodies that bind interleukin 4 receptor alpha and methods of use | |
| US12103967B2 (en) | Bispecific anti-CCL2 antibodies | |
| WO2022159776A1 (en) | Antibodies which bind human fibrin and methods of use | |
| WO2025166234A1 (en) | Methods of administering antibodies that bind interleukin 4 receptor alpha | |
| WO2024076993A2 (en) | Multi-specific antigen binding proteins which bind human fibrin or fibrinogen ?c domain and vascular endothelial growth factor and methods of use | |
| EP4598573A1 (en) | Multi-specific antigen binding proteins which bind human fibrin or fibrinogen ?c domain and vascular endothelial growth factor and methods of use | |
| AU2024313862A1 (en) | Antibodies that bind interleukin 13 and tslp or tslpr and methods of use | |
| WO2025265071A1 (en) | Antibodies that bind tslp and methods of use | |
| WO2026036080A1 (en) | Compositions and methods comprising combinations of tslp and il-13 antibodies | |
| WO2026036115A2 (en) | Compositions and methods comprising combinations of tslp and ox40l antibodies | |
| WO2026036112A1 (en) | Compositions and methods comprising combinations of tslp and interleukin 4 receptor alpha antibodies | |
| KR20240021859A (en) | Bispecific anti-CCL2 antibody | |
| WO2025212751A1 (en) | Multi-specific antigen-binding proteins which bind human fibrin yc or fibrinogen yc domain and vascular endothelial growth factor and methods of use |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24826790 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: AU2024313862 Country of ref document: AU |
|
| ENP | Entry into the national phase |
Ref document number: 2024313862 Country of ref document: AU Date of ref document: 20240621 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2024826790 Country of ref document: EP |