WO2024254212A2 - Anti-vista antibody rna or nucleic acid conjugates (arcs or ancs), compositions containing, and therapeutic use thereof - Google Patents
Anti-vista antibody rna or nucleic acid conjugates (arcs or ancs), compositions containing, and therapeutic use thereof Download PDFInfo
- Publication number
- WO2024254212A2 WO2024254212A2 PCT/US2024/032660 US2024032660W WO2024254212A2 WO 2024254212 A2 WO2024254212 A2 WO 2024254212A2 US 2024032660 W US2024032660 W US 2024032660W WO 2024254212 A2 WO2024254212 A2 WO 2024254212A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cell
- disease
- arc
- cancer
- nucleotides
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
- C12N15/1138—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing against receptors or cell surface proteins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6801—Drug-antibody or immunoglobulin conjugates defined by the pharmacologically or therapeutically active agent
- A61K47/6803—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates
- A61K47/6807—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates the drug or compound being a sugar, nucleoside, nucleotide, nucleic acid, e.g. RNA antisense
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6849—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a receptor, a cell surface antigen or a cell surface determinant
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6875—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody being a hybrid immunoglobulin
- A61K47/6877—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody being a hybrid immunoglobulin the antibody being an immunoglobulin containing regions, domains or residues from different species
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
- C07K16/2827—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily against B7 molecules, e.g. CD80, CD86
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
- C12N15/1137—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing against enzymes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Y—ENZYMES
- C12Y207/00—Transferases transferring phosphorus-containing groups (2.7)
- C12Y207/01—Phosphotransferases with an alcohol group as acceptor (2.7.1)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2239/00—Indexing codes associated with cellular immunotherapy of group A61K40/00
- A61K2239/38—Indexing codes associated with cellular immunotherapy of group A61K40/00 characterised by the dose, timing or administration schedule
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/24—Immunoglobulins specific features characterized by taxonomic origin containing regions, domains or residues from different species, e.g. chimeric, humanized or veneered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/52—Constant or Fc region; Isotype
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/52—Constant or Fc region; Isotype
- C07K2317/524—CH2 domain
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/55—Fab or Fab'
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/71—Decreased effector function due to an Fc-modification
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/77—Internalization into the cell
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/11—Antisense
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/14—Type of nucleic acid interfering nucleic acids [NA]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/35—Nature of the modification
- C12N2310/351—Conjugate
- C12N2310/3513—Protein; Peptide
Definitions
- the present invention disclosed herein relates to anti-VISTA antibody RNA or nucleic acid conjugates (ARCs or ANCs) which specifically deliver at least one nucleic acid, e.g., RNAs or DNAs or nucleic acid/protein complexes into immune cells and the use of such ARCs or ANCs as therapeutics, e.g., for treating autoimmune and inflammatory conditions, or for treating cancer and/or symptoms associated therewith elicited by specific immune cell types.
- ARCs or ANCs anti-VISTA antibody RNA or nucleic acid conjugates
- RNA function is a developing area of therapeutic interest.
- Drugs that affect mRNA stability like antisense oligonucleotides and short interfering RNAs are one way to modulate RNA function.
- Another group of oligonucleotides can modulate RNA function by altering the processing of pre-mRNA to include or exclude specific regions of pre-mRNAs from the ultimate gene product: the encoded protein. Accordingly, oligonucleotide therapeutics represent a means of modulating protein expression in disease states and as such have utility as therapeutics.
- RNAs to target cells by incorporation into ANCs or ARCs is known.
- existing ANCs or ARCs do not target immune cell types.
- the present invention relates to anti-VISTA antibody RNA conjugates (ARCs) which may be used to specifically deliver nucleic acids, e.g., RNAs or DNAs or nucleic acid/protein complexes into immune cells.
- ARCs anti-VISTA antibody RNA conjugates
- the present invention relates to the use of such ARCs as therapeutics, e.g., for treating autoimmune, inflammatory and cancer conditions.
- the present invention provides an antibody-RNA or antibody- nucleic acid conjugate (“ARC” or “ANC”) which comprises (i) an antibody or antibody fragment which binds to an antigen specifically or predominantly expressed by one or more immune cell types; and to which is directly or indirectly conjugated (ii) one or more nucleic acids, preferably RNA or DNA oligonucleotides (“payload” or “payloads”) comprised of wildtype or modified nucleotides, which oligonucleotides specifically bind to a target gene expressed by an immune cell, optionally an immunomodulatory gene or to an RNA encoded thereby; and optionally (iii) a cleavable or non-cleavable linker or adaptor, e.g., a peptide intervening said (i) antibody or antibody fragment and said (ii) one or more nucleic acids; wherein such ARC or ANC when in contact with an immune cell which expresses the antigen bound by the (i) an antibody or antibody fragment
- the ARC or ANC of any of the foregoing comprises one or more payloads which comprise one or more modified nucleotides, optionally at least one phosphonate and/or ribose modified nucleotide which facilitates direct or indirect attachment of the one or more payloads to the antibody or antibody fragment, optionally via a peptide linker, further optionally a cleavable or non-cleavable linker or adaptor, e.g., a peptide intervening said (i) antibody or antibody fragment and (ii) a payload.
- a payloads which comprise one or more modified nucleotides, optionally at least one phosphonate and/or ribose modified nucleotide which facilitates direct or indirect attachment of the one or more payloads to the antibody or antibody fragment, optionally via a peptide linker, further optionally a cleavable or non-cleavable linker or adaptor, e.g., a peptide intervening said
- the ARC or ANC of any of the foregoing comprises a payload which is directly or indirectly conjugated to the antibody or antibody fragment via a reactive amine, optionally comprised on a lysine residue on the antibody or antibody fragment and/or on a peptide linking the (i) antibody or antibody fragment to (ii) one or more payloads.
- the ARC or ANC of any of the foregoing comprises an antibody or antibody fragment which binds to VISTA, preferably human VISTA.
- the ARC or ANC of any of the foregoing comprises an antibody or antibody fragment that binds to VISTA, preferably human VISTA, and comprises the same VH and VL CDRs as any one of anti-human VISTA antibodies comprising the sequences in Figure 14 or in Appendix 1 or Appendix 3.
- the antibody or antibody fragment in the ARC or ANC binds to VISTA, preferably human VISTA, and comprises the same VH and/or VL regions and CDRs as any one of anti-human VISTA antibodies comprising the VH and/or VL sequences in Figure 14 or an antibody or antibody fragment comprising VH and/or VL regions possessing a least 90, 95 or 99% sequence identity to the VH and/or VL regions as any one of anti-human VISTA antibodies comprising the VH and/or VL sequences in Figure 14; or an antibody or antibody fragment which comprises the VH and/or VL sequences in Appendix 1 or Appendix 3; and which antibody or antibody fragment optionally comprises IgGl, lgG2, lgG3 or lgG4 constant domain polypeptides, further optionally IgGl constant domain polypeptides, still further optionally IgGl constant domain polypeptides having the sequences contained in Appendix 1 or
- the antibody or antibody fragment on the ANC or ARC comprises a human Fc region, optionally human IgGl, lgG2, lgG3 or lgG4, further optionally modified to impair complement and/or FcR binding and/or to enhance FcRn binding.
- the ARC or ANC of any of the foregoing comprises one or more of a short interfering RNA (siRNA), antisense oligonucleotide (ASO), short hairpin RNA (shRNA), microRNA (miRNA), double-stranded RNA (dsRNA), transfer RNA (tRNA), ribosomal RNA (rRNA), or heterogeneous nuclear RNA (hnRNA).
- siRNA short interfering RNA
- ASO antisense oligonucleotide
- shRNA microRNA
- miRNA microRNA
- dsRNA double-stranded RNA
- tRNA transfer RNA
- rRNA ribosomal RNA
- hnRNA heterogeneous nuclear RNA
- the ARC or ANC of any of the foregoing comprises a polynucleic acid molecule which is from about 10 to about 1000, 10 to about 500, 10 to about 400, 10 to about 300, 10 to about 200, 10 to about 150, 10 to about 100, 10 to about 50, about 10 to about 30, about 15 to about 30, about 18 to about 25, about 18 to about 24, about 19 to about 23, or about 20 to about 22 nucleotides in length; or which comprises a polynucleic acid molecule of about 50 nucleotides, about 45 nucleotides, about 40 nucleotides, about 35 nucleotides, about 30 nucleotides, about 25 nucleotides, about 20 nucleotides, about 19 nucleotides, about 18 nucleotides, about 17 nucleotides, about 16 nucleotides, about 15 nucleotides, about 14 nucleotides, about 13 nucleotides, about 12 nucleotides, about 11 nucleot
- the ARC or ANC of any of the foregoing comprises a first polynucleotide and a second polynucleotide, optionally wherein the first polynucleotide is a sense strand or passenger strand and/or the second polynucleotide is an antisense strand or guide strand.
- the ARC or ANC of any of the foregoing comprises an siRNA, ASO, tRNA, rRNA, or a mRNA.
- the ARC or ANC of any of the foregoing comprises, or is encapsulated in or conjugated to a lipid nanoparticle.
- the ARC or ANC of any of the foregoing comprises at least one payload targets an immunomodulator selected from a cytokine, chemokine, interleukin, interferon, tumor necrosis factor, or receptor of any of the foregoing.
- the ARC or ANC of any of the foregoing comprises a payload which targets an RNA or DNA sequence encoding an immunomodulator selected from is IL- 2, IL-3, IL-4, IL-5, IL-6, IL-7, IL-8, IL-10, IL-11, IL-12, 11-15, IL-17, IL-18, IL-22, IL-37, IL-1 ⁇ , TGF- ⁇ , IFN ⁇ , IFN ⁇ , IFN ⁇ , TNF- ⁇ , TNF- ⁇ , GM-CSF, phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha (PIK3CA), RAR related orphan receptor C (RORC) or any of the molecules having the sequences identified in Figure 1 or Figure 2.
- an immunomodulator selected from is IL- 2, IL-3, IL-4, IL-5, IL-6, IL-7, IL-8, IL-10, IL-11, IL-12, 11-15, IL-17, IL
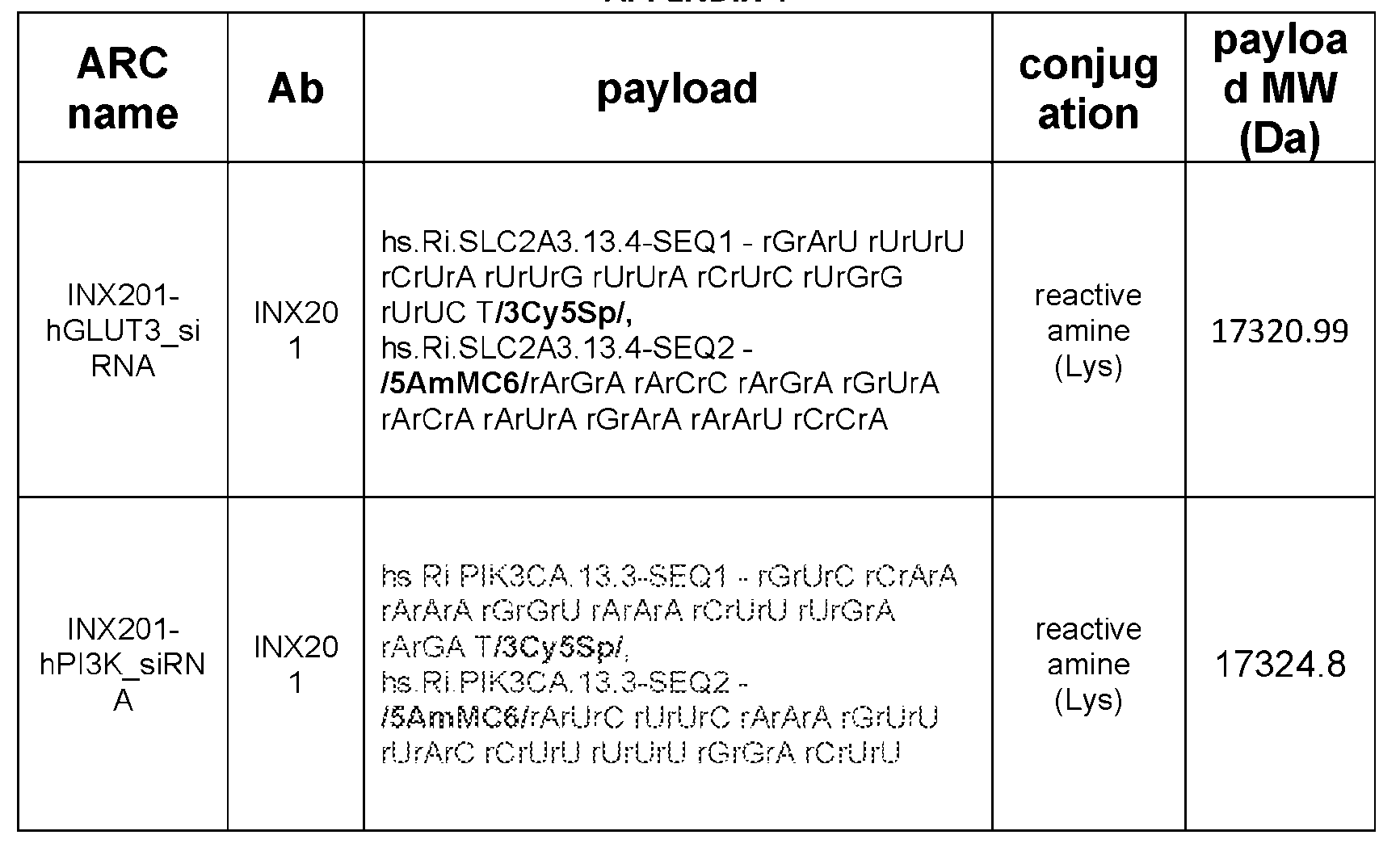
- the ARC or ANC of any of the foregoing comprises an siRNA payload which targets an RNA or DNA encoding GLUT3 or PIK3CA, optionally a sequence in Appendix 2 or 4.
- the ARC or ANC of any of the foregoing comprises an antibody or antibody fragment which binds to at least one immune cell selected from PMBCs, a T cell, a T cell progenitor cell, a CD4+ T cell, a helperT cell, a regulatory T cell, a CD8+T cell, a naive T cell, an effectorT cell, a memory T cell, a stem cell memory T (TSCM) cell, a central memory T (TCM) cell, an effector memoryT (TEM) cell, a terminally differentiated effector memory T cell, a tumor-infiltrating lymphocyte (TIL), an immature T cell, a mature T cell, a cytotoxic T cell, a mucosa-associated invariant T (MAIT) cell, a TH1 cell, a TH2 cell, a TH3 cell, a TH17 cell, a TH9 cell, a TH22 cell, a follicular help
- the ARC or ANC of any of the foregoing comprises an antibody or antibody fragment that binds to a myeloid cell and/or a T cell.
- the ARC or ANC of any of the foregoing comprises an antibody or antibody fragment that binds to a T cell orT cell progenitor cell or NK cell.
- the ARC or ANC of any of the foregoing comprises at least one nucleic acid payload, optionally an RNA or DNA, further optionally an siRNA or antisense RNA, that binds to a gene or nucleic acid, optionally an RNA or DNA, encoding an antigen selected from the group consisting of: (1) 17-IA, 4-1BB, 4Dc, 6- keto-PGFIa, 8-iso-PGF2a, 8- oxo-dG, Al Adenosine Receptor, A33, ACE, ACE-2, Activin, Activin A, Activin AB, Activin B, Activin C, Activin RIA, Activin RIA ALK-2, Activin RIB ALK-4, Activin RIIA, Activin RUB, ADAM, ADAM10, ADAM12, ADAM 15, ADAM 17/T ACE, ADAMS, ADAM9, ADAMTS, ADAMTS4, ADA
- the ARC or ANC of any of the foregoing comprises a nucleic acid payload, optionally an RNA, further optionally an siRNA or antisense RNA, having a payload comprising a sequence selected from those recited in Appendix 2 or Appendix 4; or which comprises an INX-201 ARC selected from those recited in Appendix 2 or which comprises an INX-201 ARC selected from those comprising the amino acid sequences and payload sequences recited Appendix 4.
- the ARC or ANC of any of the foregoing comprises at least 2 different RNA payloads which target the same or different immunomodulatory genes or mRNAs, optionally an immune target as disclosed above.
- the nucleic acid is linked to the antibody or antibody fragment via a cleavable or non-cleavable linker.
- the ARC or ANC of any of the foregoing is used to deliver one or more gene editing nucleic acids (e.g., CRISPR guide RNA (gRNA or sgRNA)) and optionally an CRISPR-associated endonuclease or a nucleic acid which encodes for a CRISPR- associated endonuclease.
- CRISPR guide RNA gRNA or sgRNA
- CRISPR-associated endonuclease or a nucleic acid which encodes for a CRISPR- associated endonuclease.
- the ARC or ANC of any of the foregoing comprises a PD of at least 1 day, 2 days, 3 days, 4 days, 5 days, a week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, or 6 weeks or longer.
- the ARC or ANC of any of the foregoing does not elicit any appreciable toxicity to non-target cells.
- the invention provides a composition comprising an ARC or ANC of any of the foregoing, and a pharmaceutically acceptable carrier or excipient, wherein said ARCs or ANCs are optionally comprised in or on a lipid nanoparticle.
- the invention provides a method of therapy or prophylaxis, which comprises the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof.
- the invention relates to the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof for treatment or prevention of a neoplastic, proliferative, neurodegenerative, neuroinflammatory, infectious, autoimmune, allergic, or inflammatory condition or a pathologic symptom associated with any of said conditions.
- the invention relates to the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof for treatment or prevention of an autoimmune disease, e.g., one involving myeloid orT cells.
- the invention relates to the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof for treatment or prevention of a neoplastic, proliferative, neurodegenerative, neuroinflammatory, infectious, autoimmune or inflammatory disease and/or to prevent or inhibit at least one pathologic symptom associated therewith.
- the invention relates to the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof for treatment or prevention of an autoimmune disease selected from one or more of Acromegaly, Acquired aplastic anemia, Acquired hemophilia, Agammaglobulinemia, primary, Alopecia areata, Ankylosing spondylitis (AS), Anti-NMDA receptor encephalitis, Antiphospholipid syndrome (APS)
- an autoimmune disease
- Juvenile polymyositis Juvenile dermatomyositis
- juvenile myositis Kawasaki disease, Lambert-Eaton myasthenic syndrome (LEMS), Leukocytoclastic vasculitis, Lichen planus, Lichen sclerosus, Ligneous conjunctivitis.
- LEMS Lambert-Eaton myasthenic syndrome
- Linear IgA disease (LAD)
- Paroxysmal nocturnal hemoglobinuria PNH
- Peripheral uveitis/pars planitis PANS/PANDAS, Parsonage-Turner syndrome
- Pemphigus gestationis / herpes gestationis
- Pemphigus foliaceus Pemphigus vulgaris
- Pernicious anemia POEMS syndrome
- Polyarteritis nodosa Polymyalgia rheumatica
- Polymyositis Postural orthostatic tachycardia syndrome (POTS)
- PBC Primary biliary cirrhosis
- PSC Primary biliary cholangitis
- Psoriasis Palmoplantar Pustulosis
- Psoriatic arthritis Pulmonary fibrosis, idiopathic (IPF), Pure red cell aplasia (PRCA), Pyoderma gangrenosum, Rasmussen's encephalitis,
- Restless leg syndrome (RLS) / Willis-Ekbom disease, Rheumatic fever, Rheumatoid arthritis, Sarcoidosis, Schmidt syndrome / autoimmune polyendocrine syndrome type II, Scleritis, Scleroderma, Sclerosing Mesenteritis / Mesenteric Panniculitis, Serpiginous choroidopathy, Sjogren's syndrome, Stiff person syndrome (SPS), Small fiber sensory neuropathy, Systemic lupus erythematosus (SLE), Subacute bacterial endocarditis (SBE), Subacute cutaneous lupus, Susac syndrome, Sydenham's chorea, Sympathetic ophthalmia, Takayasu's arteritis (vasculitis), Testicular autoimmunity (vasculitis, orchitis), Tolosa-Hunt syndrome, Transverse myelitis (TM), Tubulointerstitial nephritis uveitis syndrome (TINU), Ulcerative
- the invention relates to the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof for treatment or prevention of Addison disease, arthritis, celiac disease, lupus, Grave's disease, myasthenia gravis, multiple sclerosis, ITP, rheumatoid arthritis, colitis, inflammatory bowel disease, pernicious anemia, Hashimoto's thyroiditis Sjogren's disease, asthma, type 2 diabetes, and autoimmune type I diabetes and/or to prevent or inhibit at least one pathologic symptom associated therewith.
- Addison disease arthritis, celiac disease, lupus, Grave's disease, myasthenia gravis, multiple sclerosis, ITP, rheumatoid arthritis, colitis, inflammatory bowel disease, pernicious anemia, Hashimoto's thyroiditis Sjogren's disease, asthma, type 2 diabetes, and autoimmune type I diabetes and/or to prevent or inhibit at least one
- the invention relates to the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof for treatment or prevention of an inflammatory disease selected from the group consisting of Fatty liver disease, Endometriosis, Type 2 diabetes, mellitus, Type 1 diabetes mellitus, Inflammatory bowel disease (IBD), Asthma, Rheumatoid arthritis, asthma, Obesity, Fibromyalgia, Lupus SLE, osteoarthritis, Rheumatoid Arthritis, Shingles Herpes Zoster, and Vasculitis and/or to prevent or inhibit at least one pathologic symptom associated therewith.
- an inflammatory disease selected from the group consisting of Fatty liver disease, Endometriosis, Type 2 diabetes, mellitus, Type 1 diabetes mellitus, Inflammatory bowel disease (IBD), Asthma, Rheumatoid arthritis, asthma, Obesity, Fibromyalgia,
- the invention relates to the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof for treatment or prevention of a neurodegenerative or neuroinflammatory disease, e.g., Alzheimer's disease, Amyotrophic lateral sclerosis, Friedreich ataxia, Huntington's disease, Lewy body disease, aphasia, Parkinson's disease or Spinal muscular atrophy and/or to prevent or inhibit at least one pathologic symptom associated therewith.
- a neurodegenerative or neuroinflammatory disease e.g., Alzheimer's disease, Amyotrophic lateral sclerosis, Friedreich ataxia, Huntington's disease, Lewy body disease, aphasia, Parkinson's disease or Spinal muscular atrophy and/or to prevent or inhibit at least one pathologic symptom associated therewith.
- the invention relates to the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof for treatment or prevention of cancer or to prevent cancer reoccurrence and/or to inhibit least one pathologic symptom associated with specific immune cell types.
- the invention relates to the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof for treatment or prevention of a solid tumor and/or to prevent or inhibit at least one pathologic symptom associated therewith.
- the invention relates to the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof for treatment or prevention of a hematologic malignancy and/or to prevent or inhibit at least one pathologic symptom associated therewith.
- the invention relates to the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof for treatment or prevention of a relapsed or refractory cancer, or a metastatic cancer, optionally a relapsed or refractory solid tumor, or a metastatic solid tumor, a relapsed or refractory hematologic malignancy, ora metastatic hematologic malignancy.
- the invention relates to the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof for treatment or prevention of a solid tumor selected from anal cancer, appendix cancer, bile duct cancer (i.e., cholangiocarcinoma), bladder cancer, brain tumor, breast cancer, cervical cancer, colon cancer, cancer of Unknown Primary (CUP), esophageal cancer, eye cancer, fallopian tube cancer, gastroenterological cancer, kidney cancer, liver cancer, lung cancer, medulloblastoma, melanoma, oral cancer, ovarian cancer, pancreatic cancer, parathyroid disease, penile cancer, pituitary tumor, prostate cancer, rectal cancer, skin cancer, stomach cancer, testicular cancer, throat cancer, thyroid cancer, uterine cancer, vaginal cancer, or vulvar cancer and/or to prevent or inhibit at least one pathologic symptom associated therewith.
- a solid tumor selected from anal cancer, appendix cancer, bile
- the invention relates to the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof for treatment or prevention of a hematologic malignancy, optionally a leukemia, a lymphoma, a myeloma, a non-Hodgkin's lymphoma, or a Hodgkin's lymphoma.
- the hematologic malignancy comprises chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma (SLL), high risk CLL, a non-CLL/SLL lymphoma, prolymphocytic leukemia (PLL), follicular lymphoma (FL), diffuse large B-cell lymphoma (DLBCL), mantle cell lymphoma (MCL), Waldenstrom's macroglobulinemia, multiple myeloma, extranodal marginal zone B cell lymphoma, nodal marginal zone B cell lymphoma, Burkitt's lymphoma, non-Burkitt high grade B cell lymphoma, primary mediastinal B-cell lymphoma (PMBL), immunoblastic large cell lymphoma, precursor B -lymphoblastic lymphoma, B cell prolymphocytic leukemia, lymphoplasmacytic lymphoma, splenic marginal zone lymphoma, plasma cell my
- the invention relates to the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof for treatment or prevention of a hematologic malignancy selected from chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma (SLL), high risk CLL, a non-CLL/SLL lymphoma, prolymphocytic leukemia (PLL), follicular lymphoma (FL), diffuse large B-cell lymphoma (DLBCL), mantle cell lymphoma (MCL), Waldenstrom's macroglobulinemia, multiple myeloma, extranodal marginal zone B cell lymphoma, nodal marginal zone B cell lymphoma, Burkitt's lymphoma, non-Burkitt high grade B cell lymphoma, primary mediastinal B-cell lymphoma (PMBL), immunoblastic large cell lymphoma, precursor B-ly
- CLL chronic lymph
- the invention relates to the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof for treatment or prevention of an autoimmune disease selected from the group consisting of Addison disease, arthritis, celiac disease, lupus, Grave's disease, myasthenia gravis, multiple sclerosis, ITP, rheumatoid arthritis, colitis, inflammatory bowel disease, pernicious anemia, Hashimoto's thyroiditis Sjogren's disease, asthma, type 2 diabetes, and autoimmune type I diabetes and/or to prevent or inhibit at least one pathologic symptom associated therewith.
- an autoimmune disease selected from the group consisting of Addison disease, arthritis, celiac disease, lupus, Grave's disease, myasthenia gravis, multiple sclerosis, ITP, rheumatoid arthritis, colitis, inflammatory bowel disease, pernicious anemia, Hashimoto's thyroiditis Sjogren's disease
- the invention relates to the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof for treatment of cancer, and the ARC or ANC comprises a nucleic acid, optionally an ASO or siRNA which modulates or blocks the expression of any of PD-1, PD-L1, PD-L2, CTLA-4, B7-1, B7-2, LAG-3, HHLA2, TNFRSF12A, HLA-G, NECTIN2, TNFRSF25, TNFSF14, LAIR1, TNFSF15, TNFSF4, KIR2DL4, PDCD1, LGALS9, VSIr (VISTA) and PVR.
- a nucleic acid optionally an ASO or siRNA which modulates or blocks the expression of any of PD-1, PD-L1, PD-L2, CTLA-4, B7-1, B7-2, LAG-3, HHLA2, TNFRSF12A, HLA-G, NECTIN2, TNFRSF25,
- the invention relates to the administration of an ARC or ANC of any of the foregoing, or a composition containing, to a subject in need thereof for inhibiting or treating immune senescence associated with disease or aging, and the ARC or ANC optionally comprises a nucleic acid, further optionally an ASO or siRNA which modulates or blocks the expression of any of PD-1, PD-L1, PD-L2, CTLA-4, B7-1, B7-2, LAG-3, HHLA2, TNFRSF12A, HLA-G, NECTIN2, TNFRSF25, TNFSF14, LAIR1, TNFSF15, TNFSF4, KIR2DL4, PDCD1, LGALS9, VSIr (VISTA) and PVR.
- a nucleic acid further optionally an ASO or siRNA which modulates or blocks the expression of any of PD-1, PD-L1, PD-L2, CTLA-4, B7-1, B7-2, LAG-3, HHLA2, TNFRSF12A
- Figure 1 contains a list of immunologically relevant siRNAs and ASO payloads which are embraced by the invention and which have been tested.
- K562-VISTA cells were transfected with 200 nM siRNA (or ASO) alone, and target knockdown was analyzed by qRTPCR using the ddct method and reported as fold over control. Scrambled siRNA or ASO controls were also ordered from IDT. Payloads marked with (*) were selected for conjugation to a delivery vehicle (anti-VISTA Mab INX201).
- Figure 2 contains a list of other immunologically relevant siRNAs and ASO payloads which have been identified and tested.
- K562-VISTA cells were transfected with 200 nM siRNA (or ASO) alone, and target knockdown was analyzed by qRTPCR using the ddct method and reported as fold over control. Scrambled siRNA or ASO controls were also ordered from IDT. Payloads marked with (*) were selected for conjugation to a delivery vehicle (anti-VISTA Mab INX201). Only CD39 ARC is described in this report from the second round of payloads; other ARC'S conjugation is planned/ongoing.
- Figure 3 contains SDS-PAGE results for exemplary ARCs according to the invention (INX201 ARCs) confirming effective RNA conjugation.
- INX201 ARCs were resolved on reducing SDS-PAGE followed by silver stain.
- the HC or LC shift with siRNA is more profound than ASO, based on the molecular weight of the payload ( ⁇ 17 vs. ⁇ 6 kDa).
- Figure 4A-B shows that an exemplary ARC according to the invention efficiently binds to and provides for effective internalization from the surface and intracellular retention of siRNA inside K562-VISTA cells.
- Free INX201 line with circles
- eGFP ARC line with squares
- Figure 5A-C shows that an exemplary ARCs according to the invention (INX201
- ARC's knockdown protein expression is equivalent to transfection in vitro.
- K562-VISTA WT cells or eGFP+ cell pools were used for this study.
- A) cells were treated for 28 hours with no drug (left bar) or 200 nM eGFP ARC (right bar).
- the maximal levels of eGFP protein knockdown were established by transfecting eGFP siRNA into the same cells ( ⁇ 50%, measured at 24 hours). ARC-mediated knockdown was similar to the maximal possible knockdown levels (based on siRNA sequence).
- B) Dash line represents the maximal level of CD45 protein knockdown expected, based on the payload (siRNA) potency, which is 50%, as measured from free transfected CD45 siRNA at 48 hours (right bar); cells treated for 72 hours with 200 nM eGFP ARC (middle bar) or with no drug (left bar).
- FIG. 6A-C shows that exemplary ARCs according to the invention (INX201 ARCs) inhibit TNFa from PBMC.
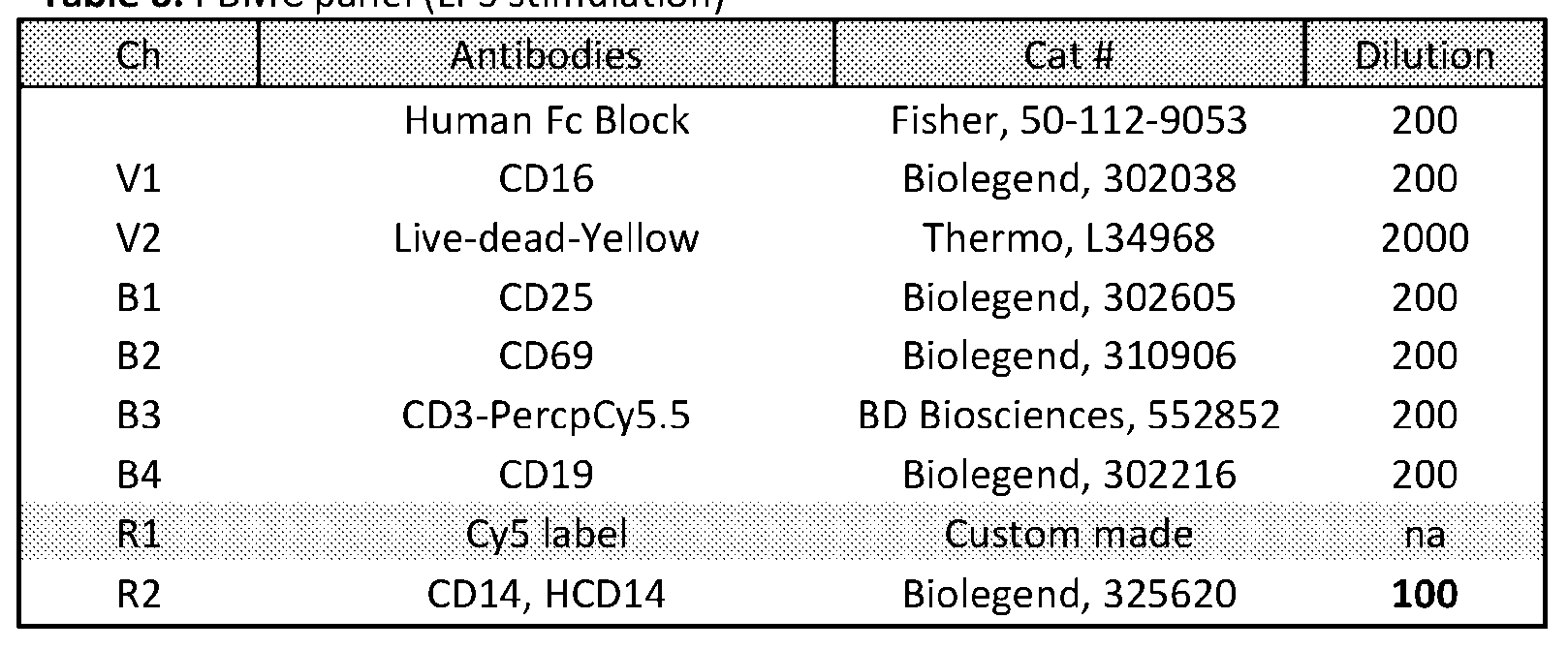
- Human PBMCs were activated with (A) 10 ng/ml LPS or (B) anti- CD3/CD28 beads, bead to T cell ratio 1:2, and treated with TNFa ARC (0-200 nM) or free RNA (200-1000 nM) for 48 hours (LPS) or 72 hours (beads). There was an efficient dosedependent reduction in TNFa levels with ARC but not free siRNA.
- FIG. 7A-B shows that an exemplary ARC according to the invention (INX201 ARC) slow down the proliferation of T cells.
- Human PBMCs were activated with anti-CD3/CD28 beads, bead to T cell ratio 1:2, and treated with (A) TN Fa ARC (0-200 nM) or free RNA (200- 1000 nM) or (B) PI3K ARC (0-200 nM) or free RNA (200-1000 nM) for 72 hours.
- Proliferation was analyzed by cell trace violet dilution, visualized by flow cytometry; no ARC was visualized as 0.1 nM (due to log scale).
- the curves were generated from inhibitor vs. response three-parameter non-linear regression analysis (GraphPad Prism 9). A single technical replicate per concentration point was used.
- FIG. 8A-B shows that an exemplary ARC according to the invention reduced PBMC activation (INX201-BTK ARC).
- Human PBMCs were activated with (A) 10 ng/ml LPS or (B) anti-CD3/CD28 beads, bead to T cell ratio 1:2, and treated with BTK ARC (0-200 nM, triangles) or free RNA (200-1000 nM, stars), or free INX201 (square) for 48 hours (LPS) or 72 hours (beads).
- the curves were generated from inhibitor vs. response three-parameter nonlinear regression analysis (GraphPad Prism 9).
- CD69 levels were measured in A; % newly proliferating cells in B; A single technical replicate per concentration point was used; no ARC was visualized as 0.1 nM (due to log scale); unstim - unstimulated cells; MFI - mean fluorescent intensity.
- Figure 9A-B shows that an exemplary ARC according to the invention reduced cytokine production (INX201-Glut1 ARC).
- Purified human T cells were activated with anti- CD3/CD28 beads, bead to a T cell ratio 1:2, and treated with Glutl ARC (0-200 nM) for 72 hours. A single technical replicate per concentration point was used. Two human donors were tested: donor 1 - solid line, donor 2 - dashed line; no ARC was visualized as 0.1 nM (due to log scale).
- A) IFNg and B) IL17A were measured by Luminex.
- Figure 10A-C also shows that an exemplary ARC according to the invention reduced cytokine production (INX201-Glutl ARC).
- Purified human T cells were activated with anti- CD3/CD28 beads, bead to a T cell ratio 1:2, and treated with RORC ARC (0-200 nM) for 72 hours. A single technical replicate per concentration point was used. Two human donors were tested: donorl - solid line, donor 2 - dashed line; no ARC was visualized as 0.1 nM (due to log scale).
- A) IFNg, B) IL-6, and C) IL12p40 were measured by Luminex.
- FIG 11 shows that an exemplary ARC according to the invention (INX201-CD39 ASO) successfully targets human PBMC and enhances immune responses in human PBMC.
- Human PBMCs were activated with anti-CD3/CD28 beads and treated with CD39 ARC (0-200 nM, triangles) or free INX201 (0-200 nM, circles) for 72 hours.
- the curves were generated from inhibitor vs. response three-parameter non-linear regression analysis (GraphPad Prism 9). A single technical replicate per concentration point was used; no ARC was visualized as 0.1 nM (due to the log scale).
- Figure 12A-B schematically shows how the inventive ARC platform may be optimized by using anti-VISTA Fab instead of a Mab.
- B) K562-VISTA cell-based competition assay, where the pre-bound increasing concentrations of INX201 Mab (squares) or INX201 Fab (circles) block available VISTA on the cell surface (hence reduced VISTA MFI measurement by INX201-AF488; the curves were generated from inhibitor vs. response three-parameter non-linear regression analysis (GraphPad Prism 9). n 2 technical replicate per concentration point was used; no Ab was visualized as 0.0001 nM (due to log scale).
- Figure 13 shows that exemplary ARCs according to the invention do not impact T cell viability.
- Human PBMCs were activated with anti-CD3/CD28 beads and treated with PI3K ARC (0-200 nM, triangles) or free INX201 (0-200 nM, squares) for 72 hours.
- the curves were generated from inhibitor vs. response three-parameter non-linear regression analysis (GraphPad Prism 9). A single technical replicate per concentration point was used; no ARC was visualized as 0.1 nM (due to the log scale); unstim - unstimulated cells.
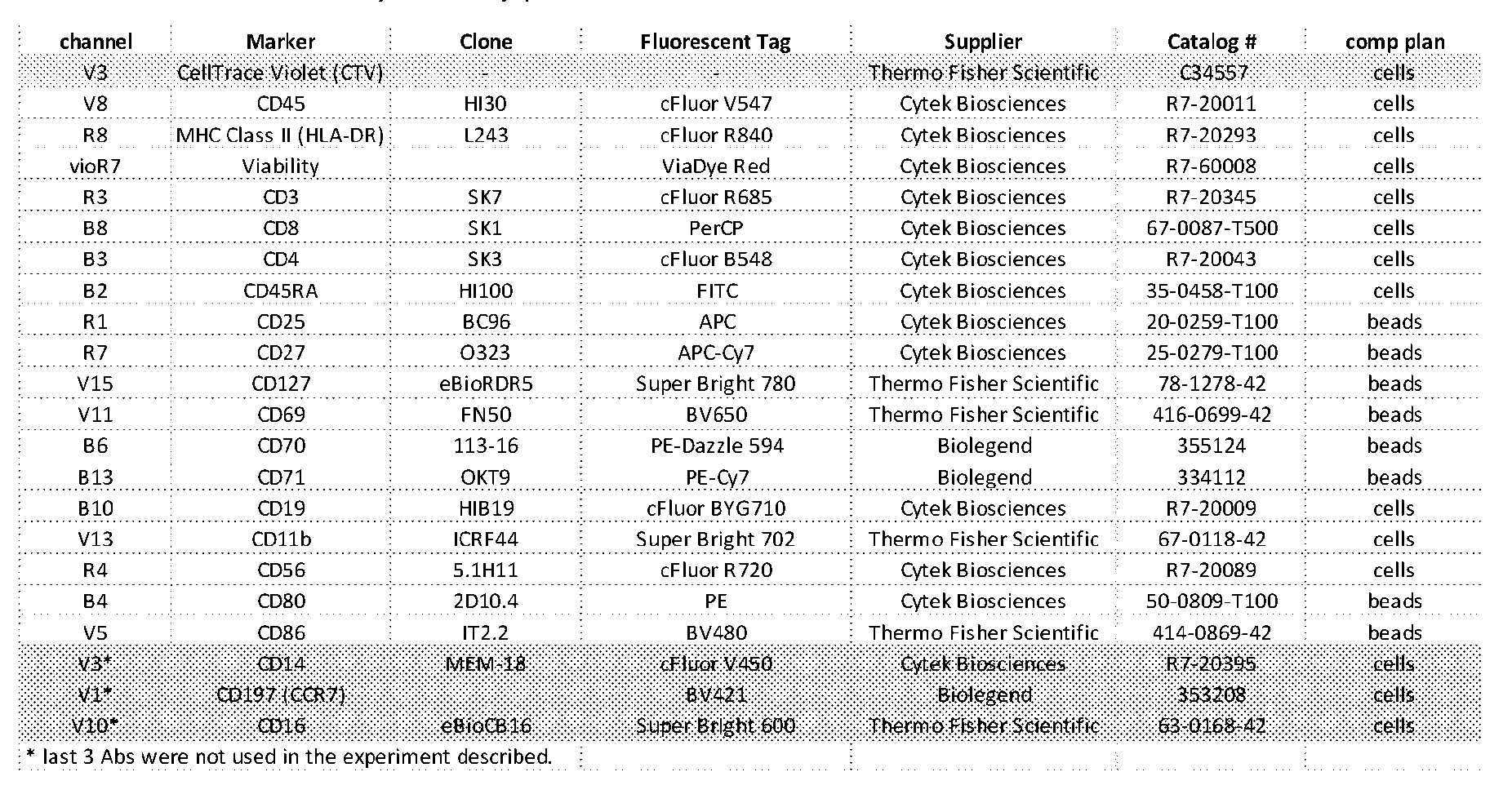
- Figure 14 shows the CDR and variable sequences at exemplary anti-human VISTA antibodies which may be used in ARCs or ANCs according to the invention.
- Figure 15 shows that INX201 (anti-VISTA) PI3K ARC, but not isotype control IgG1 PI3K ARC specifically knockdown PI3K expression.
- K562-VISTA cells were transfected with 40-200 nM were treated with 40-200 nM INX201-PI3K ARC or isotype control lgGl-PI3K ARC, and target knockdown was analyzed by qRTPCR using the ddct method and reported as % target suppression, where 0% suppression were INX201 alone samples;
- PI3K ARC anti-human VISTA mAb conjugated to PI3K siRNA;
- PI3K isotype ARC IgG control conjugated to PI3K siRNA; INX201, mAb unconjugated: naked anti-human VISTA mAb. A single technical replicate per concentration point was used.
- Figure 16A-B shows that human PI3K and GLUT3 ARCs are functional in vitro.
- Human PBMCs were activated with anti-CD3/CD28 beads for 72 hours, bead to T cell ratio 1:2, and treated with (A) PI3K ARC or free INX201 (0-200 nM), or (B) GLUT3 ARC or free INX201 (0- 200 nM).
- the curves were generated from inhibitor vs. response three-parameter non-linear regression analysis (GraphPad Prism 10).
- IL5/IL13/TNFa/IL17F cytokine levels were measured by Luminex.
- CD69 levels were measured in live CD45+/HLA-DR-/CD56- /CD3+/CD4+/CD45RA-/CD27- T effector memory cells; CD25 levels are measured in live CD45+/HLA-DR-/CD56-/CD3 cells.
- FIG. 17A-C shows that PI3K ARC reduces inflammatory cytokine responses in xeno- GvHD.
- C) Heat map based on Z-score is shown for cytokine levels at 4 hours post LPS stimulation (n 6 per group).
- FIG. 18 shows that PI3K ARC diminishes LPS induced T cell proliferation in vivo.
- Statistical analysis was performed by Student's t-test. Data represented as mean ⁇ SEM, * - p ⁇ 0.05.
- FIG. 19 shows that PI3K ARC does not reduce percent of regulatory T cells in vivo.
- Statistical analysis was performed by Student's t-test. Data represented as mean ⁇ SEM ns - not significant.
- Figure 20 shows that GLUT3 ARC reduces inflammatory cytokine responses in xeno- GvHD.
- Statistical analysis was performed by Student's t-test. Data represented as mean ⁇ SEM, ** - p ⁇ 0.01; **** - p ⁇ 0.0001.
- the present invention disclosed herein relates to anti-VISTA antibody oligonucleotide conjugates (ARCs or ANCs) which specifically deliver RNAs to immune cells and use thereof as therapeutics, e.g., for treating autoimmune and inflammatory conditions.
- ARCs or ANCs anti-VISTA antibody oligonucleotide conjugates
- antibody RNA conjugate or “antibody nucleic acid conjugate” or “ARC' or “ANC” herein generally refers to a conjugate comprising (i) an antibody or antibody fragment (such as a Fab) which specifically binds specifically or preferentially to one or more targeted immune cell types, to which is directly or indirectly attached (e.g., via a cleavable or non-cleavable peptide linker or other cleavable or non-cleavable linker) to (ii) one or more nucleic acids, typically oligonucleotides, e.g., DNAs or RNAs, which oligonucleotides may be comprised of wild-type or modified nucleotides, and which oligonucleotides specifically target immunomodulator genes or RNAs encoded thereby and which modulate the expression and/or activity of the immunomodulator protein encoded thereby.
- an antibody or antibody fragment such as a Fab
- oligonucleotides e.g
- the antibody or antibody fragment is an internalizing antibody, i.e., when it binds to the target antigen on target immune cells it internalizes the immune cell and delivers the oligonucleotide cargo comprised on the ARC or ANC into the immune cell.
- the antibody or antibody fragment is an internalizing antibody or antibody fragment which specifically binds to VISTA, preferably human VISTA.
- alkyl refers to saturated, straight- or branched-chain hydrocarbon moieties containing, in certain embodiments, between one and six, or one and eight carbon atoms, respectively.
- Examples of Ci-6-alkyl moieties include, but are not limited to, methyl, ethyl, propyl, isopropyl, «-butyl, tert-butyl, neopentyl, n-hexyl moieties; and examples of Ci- s-alkyl moieties include, but are not limited to, methyl, ethyl, propyl, isopropyl, «-butyl, tertbutyl, neopentyl, n-hexyl, heptyl, and octyl moieties.
- C x-y The number of carbon atoms in an alkyl substituent can be indicated by the prefix "C x-y ,” where x is the minimum and y is the maximum number of carbon atoms in the substituent.
- a C x chain means an alkyl chain containing x carbon atoms.
- heteroalkyl by itself or in combination with another term means, unless otherwise stated, a stable straight or branched chain alkyl group consisting of the stated number of carbon atoms and one or two heteroatoms selected from the group consisting of O, N, and S, and wherein the nitrogen and sulfur atoms may be optionally oxidized and the nitrogen heteroatom may be optionally quaternized.
- the heteroatom(s) may be placed at any position of the heteroalkyl group, including between the rest of the heteroalkyl group and the fragment to which it is attached, as well as attached to the most distal carbon atom in the heteroalkyl group.
- Up to two heteroatoms may be consecutive, such as, for example, -CH 2 - H-OCH, or -CH 2 -CH 2 -S-S-CH.
- aryl employed alone or in combination with other terms, means, unless otherwise stated, a carbocyclic aromatic system containing one or more rings (typically one, two, or three rings), wherein such rings may be attached together in a pendent manner, such as a biphenyl, or may be fused, such as naphthalene.
- aryl groups include phenyl, anthracyl, and naphthyl.
- examples of an aryl group may include phenyl (e.g., C 6 -aryl) and biphenyl (e.g., Ci 2 -aryl).
- aryl groups have from six to sixteen carbon atoms.
- aryl groups have from six to twelve carbon atoms (e.g., C 6 -i 2 -aryl).
- aryl groups have six carbon atoms (e.g., Ce-aryl).
- heteroaryl or “heteroaromatic” refers to a heterocycle having aromatic character.
- Heteroaryl substituents may be defined by the number of carbon atoms, e.g., Ci-9-heteroaryl indicates the number of carbon atoms contained in the heteroaryl group without including the number of heteroatoms.
- a Ci-9- heteroaryl will include an additional one to four heteroatoms.
- a polycyclic heteroaryl may include one or more rings that are partially saturated.
- heteroaryls include pyridyl, pyrazinyl, pyrimidinyl (including, e.g., 2- and 4-pyrimidinyl), pyridazinyl, thienyl, furyl, pyrrolyl (including, e.g., 2-pyrrolyl), imidazolyl, thiazolyl, oxazolyl, pyrazolyl (including, e.g., 3- and 5-pyrazolyl), isothiazolyl, 1,2,3-triazolyl, 1,2,4-triazolyl, 1,3,4-triazolyl, tetrazolyl, 1,2,3-thiadiazolyl, 1,2,3-oxadiazolyl, 1,3,4-thiadiazolyl and 1,3,4-oxadiazolyl.
- Non-limiting examples of polycyclic heterocycles and heteroaryls include indolyl (including, e.g., 3-, 4-, 5-, 6- and 7-indolyl), indolinyl, quinolyl, tetrahydroquinolyl, isoquinolyl (including, e.g., 1- and 5-isoquinolyl), 1, 2, 3, 4-tetra hydroisoquinolyl, cinnolinyl, quinoxalinyl (including, e.g., 2- and 5-quinoxalinyl), quinazolinyl, phthalazinyl,l,8-naphthyridinyl, 1,4- benzodioxanyl, coumarin, dihydrocoumarin, 1,5-naphthyridinyl, benzofuryl (including, e.g., 3-, 4-, 5-, 6- and 7-benzofuryl), 2,3-dihydrobenzofuryl, 1,2-benzisoxazo
- protecting group or "chemical protecting group” refers to chemical moieties that block some or all reactive moieties of a compound and prevent such moieties from participating in chemical reactions until the protective group is removed, for example, those moieties listed and described in T.W. Greene, P.G.M. Wuts, Protective Groups in Organic Synthesis, 3rd ed. John Wiley & Sons (1999). It may be advantageous, where different protecting groups are employed, that each (different) protective group be removable by a different means. Protective groups that are cleaved under totally disparate reaction conditions allow differential removal of such protecting groups. For example, protective groups can be removed by acid, base, and hydrogenolysis.
- Groups such as trityl, monomethoxytrityl, dimethoxytrityl, acetal and tert-butyldimethylsilyl are acid labile and may be used to protect carboxy and hydroxy reactive moieties in the presence of amino groups protected with Cbz groups, which are removable by hydrogenolysis, and Fmoc groups, which are base labile.
- Carboxylic acid moieties may be blocked with base labile groups such as, without limitation, methyl, or ethyl, and hydroxy reactive moieties may be blocked with base labile groups such as acetyl in the presence of amines blocked with acid labile groups such as tert-butyl carbamate or with carbamates that are both acid and base stable but hydrolytically removable.
- base labile groups such as, without limitation, methyl, or ethyl
- hydroxy reactive moieties may be blocked with base labile groups such as acetyl in the presence of amines blocked with acid labile groups such as tert-butyl carbamate or with carbamates that are both acid and base stable but hydrolytically removable.
- Carboxylic acid and hydroxyl reactive moieties may also be blocked with hydrolytically removable protective groups such as the benzyl group, while amine groups may be blocked with base labile groups such as Fmoc. A particularly useful amine protecting group is trifluoroacetamide.
- Carboxylic acid reactive moieties may be blocked with oxidatively-removable protective groups such as 2,4-dimethoxybenzyl, while coexisting amino groups may be blocked with fluoride labile silyl carbamates.
- Allyl blocking groups are useful in the presence of acid- and base-protecting groups since the former are stable and can be subsequently removed by metal or pi-acid catalysts.
- an allyl-blocked carboxylic acid can be deprotected with a palladium(O)- catalyzed reaction in the presence of acid labile t-butyl carbamate or base-labile acetate amine protecting groups.
- Yet another form of protecting group is a resin to which a compound or intermediate may be attached. As long as the residue is attached to the resin, that functional group is blocked and cannot react. Once released from the resin, the functional group is available to react.
- nucleobase refers to the heterocyclic ring portion of a nucleoside, nucleotide, and/or morpholino subunit. Nucleobases may be naturally occurring, or may be modified or analogs of these naturally occurring nucleobases, e.g., one or more nitrogen atoms of the nucleobase may be independently at each occurrence replaced by carbon.
- Exemplary analogs include hypoxanthine (the base component of the nucleoside inosine); 2, 6-diaminopurine; 5-methyl cytosine; C5-propynyl-modified pyrimidines; 10-(9-(aminoethoxy)phenoxazinyl) (G-clamp) and the like.
- base pairing moieties include, but are not limited to, uracil, thymine, adenine, cytosine, guanine and hypoxanthine having their respective amino groups protected by acyl protecting groups, 2-fluorouracil, 2-fluorocytosine, 5-bromouracil, 5- iodouracil, 2, 6-diaminopurine, azacytosine, pyrimidine analogs such as pseudoisocytosine and pseudouracil and other modified nucleobases such as 8 -substituted purines, xanthine, or hypoxanthine (the latter two being the natural degradation products).
- base pairing moieties include, but are not limited to, expanded- size nucleobases in which one or more benzene rings has been added. Nucleic base replacements described in the Glen Research catalog (www.glenresearch.com); Krueger AT et al, Acc. Chem. Res., 2007, 40, 141-150; Kool, ET, Acc. Chem. Res., 2002, 35, 936-943; Benner S.A., et al., Nat. Rev. Genet, 2005, 6, 553-543; Romesberg, F.E., et al, Curr. Opin. Chem. Biol, 2003, 7, 723-733; Hirao, L, Curr. Opin. Chem. Biol, 2006, 10, 622-627, the contents of which are incorporated herein by reference, are contemplated as useful for the synthesis of the oligomers described herein.
- oligonucleotide or “oligomer” refer to a compound comprising a plurality of linked nucleosides, nucleotides, or a combination of both nucleosides and nucleotides.
- an oligonucleotide is a morpholino oligonucleotide.
- morpholino oligonucleotide or “PMO” refers to a modified oligonucleotide having morpholino subunits linked together by phosphora midate or phosphorodiamidate linkages, joining the morpholino nitrogen of one subunit to the 5'- exocyclic carbon of an adjacent subunit.
- Each morpholino subunit comprises a nucleobase- pairing moiety effective to bind, by nucleobase-specific hydrogen bonding, to a nucleobase in a target.
- antisense oligomer refers to a sequence of subunits, each bearing a base-pairing moiety, linked by intersubunit linkages that allow the basepairing moieties to hybridize to a target sequence in a nucleic acid (typically an RNA) by Watson-Crick base pairing, to form a nucleic acid:oligomer heteroduplex within the target sequence.
- the oligomer may have exact (perfect) or near (sufficient) sequence complementarity to the target sequence; variations in sequence near the termini of an oligomer are generally preferable to variations in the interior.
- Such an antisense oligomer can be designed to block or inhibit translation of mRNA or to inhibit/alter natural or abnormal pre-mRNA splice processing, and may be said to be "directed to" or "targeted against” a target sequence with which it hybridizes.
- the target sequence is typically a region including an AUG start codon of an mRNA, a Translation Suppressing Oligomer, or splice site of a pre-processed mRNA, a Splice Suppressing Oligomer (SSO).
- the target sequence for a splice site may include an mRNA sequence having its 5' end 1 to about 25 base pairs downstream of a normal splice acceptor junction in a preprocessed mRNA.
- a target sequence may be any region of a preprocessed mRNA that includes a splice site or is contained entirely within an exon coding sequence or spans a splice acceptor or donor site.
- An oligomer is more generally said to be "targeted against” a biologically relevant target, such as a protein, virus, or bacteria, when it is targeted against the nucleic acid of the target as described above.
- the antisense oligonucleotide and the target RNA are complementary to each other when a sufficient number of corresponding positions in each molecule are occupied by nucleotides which can hydrogen bond with each other, such that stable and specific binding occurs between the oligonucleotide and the target.
- “specifically hybridizable” and “complementary” are terms which are used to indicate a sufficient degree of complementarity or precise pairing such that stable and specific binding occurs between the oligonucleotide and the target. It is understood in the art that the sequence of an oligonucleotide need not be 100% complementary to that of its target sequence to be specifically hybridizable.
- An oligonucleotide is specifically hybridizable when binding of the oligonucleotide to the target molecule interferes with the normal function of the target RNA, and there is a sufficient degree of complementarity to avoid non-specific binding of the antisense oligonucleotide to non-target sequences under conditions in which specific binding is desired, i.e., under physiological conditions in the case of in vivo assays or therapeutic treatment, and in the case of in vitro assays, under conditions in which the assays are performed.
- Oligonucleotides may also include nucleobase (often referred to in the art simply as "base”) modifications or substitutions.
- Oligonucleotides containing a modified or substituted base include oligonucleotides in which one or more purine or pyrimidine bases most commonly found in nucleic acids are replaced with less common or non-natural bases.
- the nucleobase is covalently linked at the N9 atom of the purine base, or at the N1 atom of the pyrimidine base, to the morpholine ring of a nucleotide or nucleoside.
- Purine bases comprise a pyrimidine ring fused to an imidazole ring.
- Adenine and guanine are the two purine nucleobases most commonly found in nucleic acids. These may be substituted with other naturally-occurring purines, including but not limited to N6- methyladenine, N2-methylguanine, hypoxanthine, and 7-methylguanine.
- Pyrimidine bases comprise a six-membered pyrimidine ring.
- Cytosine, uracil, and thymine are the pyrimidine bases most commonly found in nucleic acids. These may be substituted with other naturally-occurring pyrimidines, including but not limited to 5- methylcytosine, 5-hydroxymethylcytosine, pseudouracil, and 4-thiouracil.
- the oligonucleotides described herein contain thymine bases in place of uracil.
- modified or substituted bases include, but are not limited to, 2,6- diaminopurine, orotic acid, agmatidine, lysidine, 2-thiopyrimidine (e.g. 2-thiouracil, 2- thiothymine), G-clamp and its derivatives, 5-substituted pyrimidine (e.g.
- 5-halouracil 5- propynyluracil, 5-propynylcytosine, 5-aminomethyluracil, 5-hydroxymethyluracil, 5- aminomethylcytosine, 5-hydroxy methylcytosine, Super T), 7-deazaguanine, 7 -deazaadenine, 7-aza-2,6-diaminopurine, 8-aza-7-deazaguanine, 8-aza-7-deazaadenine, 8-aza-7-deaza-2,6- diaminopurine, Super G, Super A, and N4-ethylcytosine, or derivatives thereof; N2- cyclopentylguanine (cPent-G), N2-cyclopentyl-2-aminopurine (cPent-AP), and N2-propyl-2- aminopurine (Pr-AP), pseudouracil or derivatives thereof; and degenerate or universal bases, like 2,6-difluorotoluene or absent bases like abasic sites (e.
- Pseudouracil is a naturally occurring isomerized version of uracil, with a C-glycoside rather than the regular N-glycoside as in uridine.
- nucleobases reportedly are particularly useful for increasing the binding affinity of the antisense oligonucleotides. These include 5-substituted pyrimidines, 6-aza pyrimidines and N-2, N-6 and 0-6 substituted purines, including 2- aminopropyladenine, 5-propynyluracil and 5-propynylcytosine.
- nucleobases may include 5-methylcytosine substitutions, which have been shown to increase nucleic acid duplex stability by 0.6-1.2°C.
- antisense oligonucleotides may contain three or more (e.g., 3, 4, 5, 6 or more) consecutive guanine bases.
- a string of three or more consecutive guanine bases can result in aggregation of the oligonucleotides, complicating purification.
- one or more of the consecutive guanines can be substituted with hypoxanthine. The substitution of hypoxanthine for one or more guanines in a string of three or more consecutive guanine bases can reduce aggregation of the antisense oligonucleotide, thereby facilitating purification.
- the oligonucleotides disclosed herein are synthesized and do not include antisense compositions of biological origin.
- the molecules of the disclosure may also be mixed, encapsulated, conjugated or otherwise associated with other molecules, molecule structures or mixtures of compounds, as for example, liposomes, receptor targeted molecules, oral, rectal, topical or other formulations, for assisting in uptake, distribution, or absorption, or a combination thereof.
- complementarity refers to oligonucleotides (i.e., a sequence of nucleotides) related by base-pairing rules.
- sequence “T-G-A (5 -3') is complementary to the sequence “T-C-A (5'-3').”
- Complementarity may be “partial,” in which only some of the nucleic acids' bases are matched according to base pairing rules. Or, there may be “complete,” “total,” or “perfect” (100%) complementarity between the nucleic acids. The degree of complementarity between nucleic acid strands has significant effects on the efficiency and strength of hybridization between nucleic acid strands.
- an oligomer may hybridize to a target sequence at about 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 99% or 100% complementarity. Variations at any location within the oligomer are included.
- variations in sequence near the termini of an oligomer are generally preferable to variations in the interior, and if present are typically within about 6, 5, 4, 3, 2, or 1 nucleotides of the 5'-terminus, 3'-terminus, or both termini.
- peptide refers to a compound comprising a plurality of linked amino acids, e.g., which may be used to link a desired moiety, e.g., an oligonucleotide to an antibody or antibody fragment, typically an antibody or antibody fragment which specifically binds to one or more immune cell types, e.g., immune cells which are involved in an autoimmune or inflammatory disease condition.
- the term "internalizing antibody or antibody fragment” in the present invention generally refers to an antibody which internalizes immune cells when it binds to its target antigen and thereupon delivers one or more payloads which are attached thereto, e.g., an oligonucleotide (RNA or DNA comprised of wild-type or modified nucleotides), into targeted immune cells.
- the internalizing antibody or antibody fragment is an antibody which binds to VISTA, preferably human VISTA.
- cell penetrating peptide and “CPP” are used interchangeably and refer to cationic cell penetrating peptides, also called transport peptides, carrier peptides, or peptide transduction domains. Such peptides have the capability of inducing or enhancing cell penetration of a given cell culture population.
- treatment refers to the application of one or more specific procedures used for the amelioration of a disease.
- the specific procedure is the administration of one or more pharmaceutical agents.
- Treatment includes, but is not limited to, administration of a pharmaceutical composition, and may be performed either prophylactically or subsequent to the initiation of a pathologic event or contact with an etiologic agent. Treatment includes any desirable effect on the symptoms or pathology of a disease or condition, and may include, for example, minimal changes or improvements in one or more measurable markers of the disease or condition being treated.
- prophylactic treatments which can be directed to reducing the rate of progression of the disease or condition being treated, delaying the onset of that disease or condition, or reducing the severity of its onset.
- An “effective amount” or “therapeutically effective amount” refers to an amount of therapeutic compound, such as an antisense oligomer, administered to a mammalian subject, either as a single dose or as part of a series of doses, which is effective to produce a desired therapeutic effect.
- amelioration means a lessening of severity of at least one indicator of a condition or disease.
- amelioration includes a delay or slowing in the progression of one or more indicators of a condition or disease.
- the severity of indicators may be determined by subjective or objective measures which are known to those skilled in the art.
- salts refers to derivatives of the disclosed oligonucleotides wherein the parent oligonucleotide is modified by converting an existing acid or base moiety to its salt form. Lists of suitable salts are found in Remington's Pharmaceutical Sciences, 17th ed., Mack Publishing Company, Easton, Pa., 1985, p. 1418 and Journal of Pharmaceutical Science, 66, 2 (1977), each of which is incorporated herein by reference in its entirety.
- An oligonucleotides herein generally refers to a polynucleic acid molecule which when delivered into target immune cells modulates the expression and/or the activity of a protein which modulates immunity, which gene is expressed by an immune cell.
- a polynucleic acid molecule described herein modulates the expression of an immunomodulatory gene which may be wild type or may comprise one or more mutations, e.g., those which corelate to a disease condition associated with expression of the gene.
- the polynucleic acid molecule hybridizes to a target region of wild type DNA or RNA encoding the immunomodulatory gene or a fragment thereof.
- the polynucleic acid molecule is a polynucleic acid molecule that hybridizes to a target region of DNA or RNA encoded thereby comprising a mutation (e.g., a substitution, a deletion, or an addition).
- a mutation e.g., a substitution, a deletion, or an addition.
- the immunomodulatory gene and the RNA is selected from those identified in Figure 1 or Figure 2, or in Appendix 2 and Appendix 4.
- a polynucleic acid molecule hybridizes to a target region of a DNA or RNA encoding the targeted immunomodulatory protein which comprises one or more mutations.
- the RNA comprises short interfering RNA (siRNA), short hairpin RNA (shRNA), microRNA (miRNA), double-stranded RNA (dsRNA), transfer RNA (tRNA), ribosomal RNA (rRNA), or heterogeneous nuclear RNA (hnRNA).
- RNA comprises shRNA.
- RNA comprises miRNA.
- RNA comprises dsRNA.
- RNA comprises tRNA.
- RNA comprises rRNA.
- RNA comprises hnRNA.
- the RNA comprises siRNA.
- the polynucleic acid molecule comprises siRNA.
- the polynucleic acid molecule is about 19 nucleotides in length. In some instances, the polynucleic acid molecule is about 18 nucleotides in length. In some instances, the polynucleic acid molecule is about 17 nucleotides in length. In some instances, the polynucleic acid molecule is about 16 nucleotides in length. In some instances, the polynucleic acid molecule is about 15 nucleotides in length. In some instances, the polynucleic acid molecule is about 14 nucleotides in length. In some instances, the polynucleic acid molecule is about 13 nucleotides in length. In some instances, the polynucleic acid molecule is about 12 nucleotides in length.
- a polynucleic acid molecule is a first polynucleotide.
- the first polynucleotide is from about 10 to about 50 nucleotides in length. In some instances, the first polynucleotide is from about 10 to about 30, from about 15 to about 30, from about 18 to about 25, from about 18 to about 24, from about 19 to about 23, or from about 20 to about 22 nucleotides in length.
- a first polynucleotide is about 50 nucleotides in length. In some instances, the first polynucleotide is about 45 nucleotides in length. In some instances, the first polynucleotide is about 40 nucleotides in length. In some instances, the first polynucleotide is about 35 nucleotides in length. In some instances, the first polynucleotide is about 30 nucleotides in length. In some instances, the first polynucleotide is about 25 nucleotides in length. In some instances, the first polynucleotide is about 20 nucleotides in length.
- the first polynucleotide is about 19 nucleotides in length. In some instances, the first polynucleotide is about 18 nucleotides in length. In some instances, the first polynucleotide is about 17 nucleotides in length. In some instances, the first polynucleotide is about 16 nucleotides in length. In some instances, the first polynucleotide is about 15 nucleotides in length. In some instances, the first polynucleotide is about 14 nucleotides in length. In some instances, the first polynucleotide is about 13 nucleotides in length. In some instances, the first polynucleotide is about 12 nucleotides in length.
- the first polynucleotide is about 11 nucleotides in length. In some instances, the first polynucleotide is about 10 nucleotides in length. In some instances, the first polynucleotide is from about 10 to about 50 nucleotides in length. In some instances, the first polynucleotide is from about 10 to about 45 nucleotides in length. In some instances, the first polynucleotide is from about 10 to about 40 nucleotides in length. In some instances, the first polynucleotide is from about 10 to about 35 nucleotides in length. In some instances, the first polynucleotide is from about 10 to about 30 nucleotides in length.
- the first polynucleotide is from about 10 to about 25 nucleotides in length. In some instances, the first polynucleotide is from about 10 to about 20 nucleotides in length. In some instances, the first polynucleotide is from about 15 to about 25 nucleotides in length. In some instances, the first polynucleotide is from about 15 to about 30 nucleotides in length. In some instances, the first polynucleotide is from about 12 to about 30 nucleotides in length.
- a second polynucleotide is about 50 nucleotides in length. In some instances, the second polynucleotide is about 45 nucleotides in length. In some instances, the second polynucleotide is about 40 nucleotides in length. In some instances, the second polynucleotide is about 35 nucleotides in length. In some instances, the second polynucleotide is about 30 nucleotides in length. In some instances, the second polynucleotide is about 25 nucleotides in length. In some instances, the second polynucleotide is about 20 nucleotides in length.
- the second polynucleotide is about 11 nucleotides in length. In some instances, the second polynucleotide is about 10 nucleotides in length. In some instances, the second polynucleotide is from about 10 to about 50 nucleotides in length. In some instances, the second polynucleotide is from about 10 to about 45 nucleotides in length. In some instances, the second polynucleotide is from about 10 to about 40 nucleotides in length. In some instances, the second polynucleotide is from about 10 to about 35 nucleotides in length. In some instances, the second polynucleotide is from about 10 to about 30 nucleotides in length.
- the second polynucleotide is from about 10 to about 25 nucleotides in length. In some instances, the second polynucleotide is from about 10 to about 20 nucleotides in length. In some instances, the second polynucleotide is from about 15 to about 25 nucleotides in length. In some instances, the second polynucleotide is from about 15 to about 30 nucleotides in length. In some instances, the second polynucleotide is from about 12 to about 30 nucleotides in length.
- a polynucleic acid molecule comprises a first polynucleotide and a second polynucleotide.
- the polynucleic acid molecule further comprises a blunt terminus, an overhang, or a combination thereof.
- the blunt terminus is a 5' blunt terminus, a 3' blunt terminus, or both.
- the overhang is a 5' overhang, 3' overhang, or both.
- the overhang comprises 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 non-base pairing nucleotides.
- the overhang comprises 1, 2, 3, 4, 5, or 6 non-base pairing nucleotides.
- the overhang comprises 1, 2, 3, or 4 non-base pairing nucleotides. In some cases, the overhang comprises 1 non-base pairing nucleotide. In some cases, the overhang comprises 2 non-base pairing nucleotides. In some cases, the overhang comprises 3 non-base pairing nucleotides. In some cases, the overhang comprises 4 non-base pairing nucleotides.
- the sequence of a polynucleic acid molecule is at least 40%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99%, or 99.5% complementary to a target sequence. In some embodiments, the sequence of the polynucleic acid molecule is at least 50% complementary to a target sequence. In some embodiments, the sequence of the polynucleic acid molecule is at least 60% complementary to a target sequence. In some embodiments, the sequence of the polynucleic acid molecule is at least 70% complementary to a target sequence. In some embodiments, the sequence of the polynucleic acid molecule is at least 80% complementary to a target sequence.
- the sequence of the polynucleic acid molecule is at least 90% complementary to a target sequence. In some embodiments, the sequence of the polynucleic acid molecule is at least 95% complementary to a target sequence. In some embodiments, the sequence of the polynucleic acid molecule is at least 99% complementary to a target sequence. In some instances, the sequence of the polynucleic acid molecule is 100% complementary to a target sequence.
- the specificity of a polynucleic acid molecule that hybridizes to a target sequence described herein is a 95%, 98%, 99%, 99.5% or 100% sequence complementarity of the polynucleic acid molecule to a target sequence.
- the hybridization is a high stringent hybridization condition.
- a polynucleic acid molecule has reduced off-target effect.
- off-target or “off-target effects” refer to any instance in which a polynucleic acid polymer directed against a given target causes an unintended effect by interacting either directly or indirectly with another mRNA sequence, a DNA sequence or a cellular protein or other moiety.
- an "off-target effect” occurs when there is a simultaneous degradation of other transcripts due to partial homology or complementarity between that other transcript and the sense and/or antisense strand of the polynucleic acid molecule.
- a polynucleic acid molecule comprises natural, synthetic, or artificial nucleotide analogues or bases.
- the polynucleic acid molecule comprises combinations of DNA, RNA and/or nucleotide analogues.
- the synthetic or artificial nucleotide analogues or bases comprise modifications at one or more of ribose moiety, phosphate moiety, nucleoside moiety, ora combination thereof.
- nucleotide analogues or artificial nucleotide base comprise a nucleic acid with a modification at a 2' hydroxyl group of the ribose moiety.
- the modification includes an H, OR, R, halo, SH, SR, NH2, NHR, NR2, or CN, wherein R is an alkyl moiety.
- Exemplary alkyl moiety includes, but is not limited to, halogens, sulfurs, thiols, thioethers, thioesters, amines (primary, secondary, or tertiary), amides, ethers, esters, alcohols and oxygen.
- the alkyl moiety further comprises a modification.
- the modification comprises an azo group, a keto group, an aldehyde group, a carboxyl group, a nitro group, a nitroso, group, a nitrile group, a heterocycle (e.g., imidazole, hydrazino or hydroxylamino) group, an isocyanate or cyanate group, or a sulfur containing group (e.g., sulfoxide, sulfone, sulfide, or disulfide).
- the alkyl moiety further comprises a hetero substitution.
- the carbon of the heterocyclic group is substituted by a nitrogen, oxygen, or sulfur.
- the heterocyclic substitution includes but is not limited to, morpholino, imidazole, and pyrrolidino.
- the modification at the 2' hydroxyl group is a 2'-O-methyl modification or a 2'-O-methoxyethyl (2'-O-MOE) modification.
- the 2'-O- methyl modification adds a methyl group to the 2' hydroxyl group of the ribose moiety whereas the 2'O-methoxyethyl modification adds a methoxyethyl group to the 2' hydroxyl group of the ribose moiety.
- the modification at the 2' hydroxyl group is a 2'-O-aminopropyl modification in which an extended amine group comprising a propyl linker binds the amine group to the 2' oxygen.
- this modification neutralizes the phosphate derived overall negative charge of the oligonucleotide molecule by introducing one positive charge from the amine group per sugar and thereby improves cellular uptake properties due to its zwitterionic properties.
- the modification at the 2' hydroxyl group is a locked or bridged ribose modification (e.g., locked nucleic acid or LNA) in which the oxygen molecule bound at the 2' carbon is linked to the 4' carbon by a methylene group, thus forming a 2'-C,4'-C-oxy- methylene-linked bicyclic ribonucleotide monomer.
- LNA locked nucleic acid
- the modification at the 2' hydroxyl group comprises ethylene nucleic acids (ENA) such as for example 2'-4'-ethylene-bridged nucleic acid, which locks the sugar conformation into a C 3 '-endo sugar puckering conformation.
- ENA ethylene nucleic acids
- the bridged nucleic acids class of modified nucleic acids that also comprises LNA.
- additional modifications at the 2' hydroxyl group include 2'- deoxy, T-deoxy-2'-fluoro, 2'-O-aminopropyl (2'-O-AP), 2'-O-dimethylaminoethyl (2'-O- DMAOE), 2'-O- dimethylaminopropyl (2'-O-DMAP), T-O- dimethylaminoethyloxyethyl (2'-O- DMAEOE), or 2'-O- N-methylacetamido (2'-O-NMA).
- nucleotide analogues comprise modified bases such as, but not limited to, 5-propynyluridine, 5-propynylcytidine, 6- methyladenine, 6-methylguanine, N, N, - dimethyladenine, 2-propyladenine, 2-propylguanine, 2-aminoadenine, 1- methylinosine, 3- methyluridine, 5-methylcytidine, 5-methyluridine and other nucleotides having a modification at the 5 position, 5- (2- amino) propyl uridine, 5-halocytidine, 5- halouridine, 4-a cetylcytidine, 1- methyladenosine, 2-methyladenosine, 3-methylcytidine, 6- methyluridine, 2- methylguanosine, 7- methylguanosine, 2, 2-dimethylguanosine, 5- methy la mi noethyluridine, 5-methyloxyuridine, deazanucleot
- Modified nucleotides also include those nucleotides that are modified with respect to the sugar moiety, as well as nucleotides having sugars or analogs thereof that are not ribosyl.
- the sugar moieties in some cases are, or are based on, mannoses, arabinoses, glucopyranoses, galactopyranoses, 4'-thioribose, and other sugars, heterocycles, or carbocycles.
- the term nucleotide also includes what are known in the art as universal bases.
- universal bases include but are not limited to 3-nitropyrrole, 5- nitroindole, or nebularine.
- nucleotide analogues further comprise morpholines, peptide nucleic acids (PNAs), methylphosphonate nucleotides, thiolphosphonate nucleotides, 2'- fluoro N3- P5'-phosphoramidites, 1', 5'- anhydrohexitol nucleic acids (HNAs), ora combination thereof.
- PNAs peptide nucleic acids
- HNAs anhydrohexitol nucleic acids
- Morpholino or phosphorodiamidate morpholino oligo comprises synthetic molecules whose structure mimics natural nucleic acid structure but deviates from the normal sugar and phosphate structures.
- the five member ribose ring is substituted with a six member morpholino ring containing four carbons, one nitrogen and one oxygen.
- the ribose monomers are linked by a phosphordiamidate group instead of a phosphate group.
- the backbone alterations remove all positive and negative charges making morpholines neutral molecules capable of crossing cellular membranes without the aid of cellular delivery agents such as those used by charged oligonucleotides.
- peptide nucleic acid does not contain sugar ring or phosphate linkage and the bases are attached and appropriately spaced by oligoglycine-like molecules, therefore, eliminating a backbone charge.
- modified internucleotide linkage includes, but is not limited to, phosphorothioates; phosphorodithioates; methylphosphonates; 5'- alkylenephosphonates; 5'-methyl phosphonate; 3'-alkylene phosphonates; borontrifluoridates; borano phosphate esters and selenophosphates of 3'-5'linkage or 2'- 5'linkage; phosphotriesters; thionoalkylphosphotriesters; hydrogen phosphonate linkages; alkyl phosphonates; alkylphosphonothioates; arylphosphonothioates; phosphoroselenoates; phosphorodiselenoates; phosphinates; phosphoramidates; 3'- alkylphosphoramidates; aminoalkylphosphoramidates; thionophosphoramidates;
- the modification is a methyl or thiol modification such as methylphosphonate orthiolphosphonate modification.
- exemplary thiolphosphonate nucleotides and methylphosphonate nucleotides are known on the art and include 2'-fluoro N3-P5'-phosphoramidites.
- a modified nucleotide includes, but is not limited to, hexitol nucleic acid (or 1', 5'- anhydrohexitol nucleic acids (HNA)).
- one or more modifications further optionally include modifications of the ribose moiety, phosphate backbone and the nucleoside, or modifications of the nucleotide analogues at the 3' or the 5' terminus.
- the 3' terminus optionally includes a 3' cationic group, or by inverting the nucleoside at the 3'- terminus with a 3'-3' linkage.
- the 3'-terminus is optionally conjugated with an aminoalkyl group, e.g., a 3' C5-aminoalkyl dT.
- the 3'- terminus is optionally conjugated with an abasic site, e.g., with an apurinic or apyrimidinic site.
- the 5 '-terminus is conjugated with an aminoalkyl group, e.g., a 5'-O- aIkylamino substituent.
- the 5' -terminus is conjugated with an abasic site, e.g., with an apurinic or apyrimidinic site.
- a polynucleic acid molecule comprises one or more artificial nucleotide analogues described herein. In some instances, the polynucleic acid molecule comprises 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 20, 25, or more artificial nucleotide analogues described herein.
- the artificial nucleotide analogues include 2'-O-methyl, 2'-O-methoxyethyl (2'-O-MOE), 2'-O-aminopropyl, 2'-deoxy, T-deoxy-2'-fluoro, 2'-O-aminopropyl (2'-O-AP), 2'-O-dimethylaminoethyl (2'-O-DMAOE), 2'- O-dimethylaminopropyl (2'-O-DMAP), T-O- dimethylaminoethyloxyethyl (2'-O-DMAEOE), or 2'-O-N-methylacetamido (2'-O-NMA) modified, LNA, ENA, PNA, HNA, morpholino, methylphosphonate nucleotides, thiolphosphonate nucleotides, 2'-fluoro N3-P5'- phosphoramidites, or a combination thereof.
- the polynucleic acid molecule comprises 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 20, 25, or more of the artificial nucleotide analogues selected from 2'-O-methyl, 2'-O-methoxyethyl (2'-O- MOE), 2'-O-a mi no propyl, 2'-deoxy, T-deoxy-2'-fluoro, 2'-O-aminopropyl (2'-O-AP), 2'-O- dimethylaminoethyl (2'-O-DMAOE), 2'-O-dimethylaminopropyl (2'-O-DMAP), T-O- dimethylaminoethyloxyethyl (2'-O-DMAEOE), or 2'-O-N-methylacetamido (2'-0-NMA) modified, LNA, ENA, PNA, HNA, morpholino, methylphosphonate nucleotides, thiolphosphonate nucleot
- the polynucleic acid molecule comprises 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 20, 25, or more of 2'-O-methyl modified nucleotides. In some instances, the polynucleic acid molecule comprises 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 20, 25, or more of 2'-O-methoxy ethyl (2'-O-MOE) modified nucleotides. In some instances, the polynucleic acid molecule comprises 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 20, 25, or more of thiolphosphonate nucleotides.
- a polynucleic acid molecule comprises at least one of: from about 5% to about 100% modification, from about 10% to about 100% modification, from about 20% to about 100%) modification, from about 30% to about 100% modification, from about 40% to about 100%) modification, from about 50% to about 100% modification, from about 60% to about 100% modification, from about 70% to about 100% modification, from about 80% to about 100% modification, and from about 90% to about 100% modification.
- a polynucleic acid molecule comprises at least one of: from about 10% to about 90%) modification, from about 20% to about 90% modification, from about 30% to about 90%) modification, from about 40% to about 90% modification, from about 50% to about 90% modification, from about 60% to about 90% modification, from about 70% to about 90% modification, and from about 80% to about 100% modification.
- a polynucleic acid molecule comprises at least one of: from about 10% to about 80%) modification, from about 20% to about 80% modification, from about 30% to about 80%) modification, from about 40% to about 80% modification, from about 50% to about 80% modification, from about 60% to about 80% modification, and from about 70% to about 80% modification.
- a polynucleic acid molecule comprises at least one of: from about 10% to about 70%) modification, from about 20% to about 70% modification, from about 30% to about 70%) modification, from about 40% to about 70% modification, from about 50% to about 70% modification, and from about 60% to about 70% modification.
- a polynucleic acid molecule comprises at least one of: from about 10% to about 60%) modification, from about 20% to about 60% modification, from about 30% to about 60%) modification, from about 40% to about 60% modification, and from about 50% to about 60% modification.
- a polynucleic acid molecule comprises at least one of: from about 10% to about 50%) modification, from about 20% to about 50% modification, from about 30% to about 50% modification, and from about 40% to about 50% modification.
- a polynucleic acid molecule comprises at least one of: from about 10% to about 40%) modification, from about 20% to about 40% modification, and from about 30% to about 40%) modification.
- a polynucleic acid molecule comprises at least one of: from about 10% to about 30%) modification, and from about 20% to about 30% modification.
- a polynucleic acid molecule comprises from about 10% to about 20% modification. In some cases, a polynucleic acid molecule comprises from about 15% to about 90%, from about 20% to about 80%, from about 30% to about 70%, or from about 40% to about 60% modifications.
- a polynucleic acid molecule comprises at least about 15%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 99% modification.
- a polynucleic acid molecule comprises at least about 1, about 2, about 3, about 4, about 5, about 6, about 7, about 8, about 9, about 10, about 11, about 12, about 13, about 14, about 15, about 16, about 17, about 18, about 19, about 20, about 21, about 22, or more modifications.
- a polynucleic acid molecule comprises at least about 1, about 2, about 3, about 4, about 5, about 6, about 7, about 8, about 9, about 10, about 11, about 12, about 13, about 14, about 15, about 16, about 17, about 18, about 19, about 20, about 21, about 22 or more modified nucleotides.
- a polynucleic acid molecule comprises an artificial nucleotide analogue described herein. In some instances, about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, or 100% of a polynucleic acid molecule comprise an artificial nucleotide analogue.
- a polynucleic acid molecule comprise an artificial nucleotide analogue. In some instances, about 5% of a polynucleic acid molecule comprise an artificial nucleotide analogue. In some instances, about 10% of a polynucleic acid molecule comprise an artificial nucleotide analogue.
- a polynucleic acid molecule is assembled from two separate polynucleotides wherein one polynucleotide comprises the sense strand and the second polynucleotide comprises the antisense strand of the polynucleic acid molecule.
- the sense strand is connected to the antisense strand via a linker molecule, which in some instances is a polynucleotide linker or a non-nucleotide linker.
- a polynucleic acid molecule comprises a sense strand and antisense strand, wherein pyrimidine nucleotides in the sense strand comprise 2'-O- methylpyrimidine nucleotides and purine nucleotides in the sense strand comprise 2'-deoxy purine nucleotides.
- a polynucleic acid molecule comprises a sense strand and antisense strand, wherein pyrimidine nucleotides present in the sense strand comprise 2'-deoxy-2'-fluoro pyrimidine nucleotides and wherein purine nucleotides present in the sense strand comprise 2'-deoxy purine nucleotides.
- a polynucleic acid molecule comprises a sense strand and antisense strand, wherein the pyrimidine nucleotides when present in said antisense strand are 2'-deoxy-2'-fluoro pyrimidine nucleotides and the purine nucleotides when present in said antisense strand are 2'-O-methyl purine nucleotides.
- a polynucleic acid molecule comprises a sense strand and antisense strand, wherein the pyrimidine nucleotides when present in said antisense strand are 2'-deoxy-2'-fluoro pyrimidine nucleotides and wherein the purine nucleotides when present in said antisense strand comprise 2'-deoxy-purine nucleotides.
- a polynucleic acid molecule comprises a sense strand and antisense strand, wherein the sense strand includes a terminal cap moiety at the 5'-end, the 3'-end, or both of the 5' and 3' ends of the sense strand.
- the terminal cap moiety is an inverted deoxy abasic moiety.
- a polynucleic acid molecule comprises a sense strand and an antisense strand, wherein the antisense strand comprises a phosphate backbone modification at the 3' end of the antisense strand.
- the phosphate backbone modification is a phosphorothioate.
- a polynucleic acid molecule comprises a sense strand and an antisense strand, wherein the antisense strand comprises a glyceryl modification at the 3' end of the antisense strand.
- a polynucleic acid molecule comprises a sense strand and an antisense strand, in which the sense strand comprises one or more, for example, about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, or more phosphorothioate internucleotide linkages, and/or one or more (e.g., about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more) 2'-deoxy, 2'-O-methyl, 2'-deoxy-2'-fluoro, and/or about one or more (e.g., about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more) universal base modified nucleotides, and optionally a terminal cap molecule at the 3'-end, the 5'-end, or both of the 3'- and 5'-ends of the sense strand; and in which the antisense strand comprises about 1 to about 10 or more, specifically about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, or
- one or more, for example about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, or more, pyrimidine nucleotides of the sense and/or antisense strand are chemically-modified with 2'-deoxy, 2'-O-methyl and/or 2'-deoxy-2'-fluoro nucleotides, with or without one or more, for example about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, or more, phosphorothioate internucleotide linkages and/or a terminal cap molecule at the 3'-end, the 5'-end, or both of the 3'- and 5'- ends, being present in the same or different strand.
- a polynucleic acid molecule comprises a sense strand and an antisense strand, in which the sense strand comprises about 1 to about 25, for example, about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, or more phosphorothioate internucleotide linkages, and/or one or more (e.g., about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, or more) 2'-deoxy, 2'-O-methyl, 2'-deoxy-2'-fluoro, and/or one or more (e.g., about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, or more) universal base modified nucleotides, and optionally a terminal cap molecule at the 3 -end, the 5'-end, or both of the 3'- and 5'-ends of the sense strand; and in which the antisense strand comprises about 1 to about 25 or more, for example about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20,
- one or more, for example about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, or more, pyrimidine nucleotides of the sense and/ or antisense strand are chemically-modified with 2'-deoxy, 2'-O-methyl and/or 2'-deoxy-2'-fluoro nucleotides, with or without about 1 to about 25 or more, for example about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, or more phosphorothioate internucleotide linkages and/or a terminal cap molecule at the 3'-end, the 5'-end, or both of the 3'- and 5'-ends, being present in the same or different strand.
- a polynucleic acid molecule comprises a sense strand and an antisense strand, in which the antisense strand comprises one or more, for example, about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, or more phosphorothioate internucleotide linkages, and/or about one or more (e.g., about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more) 2'-deoxy, 2'-O-methyl, 2'-deoxy-2'-fluoro, and/or one or more (e.g., about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more) universal base modified nucleotides, and optionally a terminal cap molecule at the 3'-end, the 5'-end, or both of the 3'- and 5'-ends of the sense strand; and wherein the antisense strand comprises about 1 to about 10 or more, specifically about 1, 2,
- one or more, for example about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, or more pyrimidine nucleotides of the sense and/or antisense strand are chemically-modified with 2'-deoxy, 2'- O-methyl and/or 2'-deoxy-2'-fluoro nucleotides, with or without one or more, for example, about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more phosphorothioate internucleotide linkages and/or a terminal cap molecule at the 3'-end, the 5'-end, or both of the 3' and 5'-ends, being present in the same or different strand.
- a polynucleic acid molecule comprises a sense strand and an antisense strand, in which the antisense strand comprises about 1 to about 25 or more, for example, about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, or more phosphorothioate internucleotide linkages, and/or one or more (e.g., about 1, 2, 3, 4, 5, 6,
- the antisense strand comprises about 1 to about 25 or more, for example about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, or more phosphorothioate internucleotide linkages, and/or one or more (e.g., about 1, 2, 3, 4, 5, 6,
- pyrimidine nucleotides of the sense and/or antisense strand are chemically- modified with 2'-deoxy, 2'-O-methyl and/or 2'-deoxy-2'-fluoro nucleotides, with or without about 1 to about 5, for example about 1, 2, 3, 4, 5 or more phosphorothioate internucleotide linkages and/or a terminal cap molecule at the 3'-end, the 5'-end, or both of the 3'- and 5'-ends, being present in the same or different strand.
- a polynucleic acid molecule described herein is a chemically- modified short interfering nucleic acid molecule having about 1 to about 25, for example, about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20 or more phosphorothioate internucleotide linkages in each strand of the polynucleic acid molecule.
- a polynucleic acid molecule described herein comprises 2'- 5' internucleotide linkages.
- the 2'-5' internucleotide linkage(s) is at the 3'- end, the 5'-end, or both of the 3'- and 5' -ends of one or both sequence strands.
- the 2'-5' internucleotide linkage(s) is present at various other positions within one or both sequence strands, for example, about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, or more including every internucleotide linkage of a pyrimidine nucleotide in one or both strands of the polynucleic acid molecule comprise a 2'-5' internucleotide linkage, or about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, or more including every internucleotide linkage of a purine nucleotide in one or both strands of the polynucleic acid molecule comprise a 2'-5' internucleotide linkage.
- a polynucleic acid molecule is a single stranded polynucleic acid molecule that mediates RNAi activity in a cell or reconstituted in vitro system, wherein the polynucleic acid molecule comprises a single stranded polynucleotide having complementarity to a target nucleic acid sequence, and wherein one or more pyrimidine nucleotides present in the polynucleic acid are 2'-deoxy-2'-fluoro pyrimidine nucleotides (e.g., wherein all pyrimidine nucleotides are 2'-deoxy-2'-fluoro pyrimidine nucleotides or alternately a plurality of pyrimidine nucleotides are 2'-deoxy-2'-fluoro pyrimidine nucleotides), and wherein one or more purine nucleotides present in the polynucleic acid are 2'-deoxy purine nucleotides (e.g., wherein all
- one or more artificial nucleotide analogues are resistant toward nucleases such as for example ribonuclease such as RNase H, deoxyribu nuclease such as DNase, or exonuclease such as 5'-3' exonuclease and 3'-5' exonuclease when compared to natural polynucleic acid molecules.
- nucleases such as for example ribonuclease such as RNase H, deoxyribu nuclease such as DNase, or exonuclease such as 5'-3' exonuclease and 3'-5' exonuclease when compared to natural polynucleic acid molecules.
- artificial nucleotide analogues comprising 2'-O-methyl, 2'-O-methoxyethyl (2'-O-MOE), 2'-O-aminopropyl, 2'-deoxy, T- deoxy-2'-fluoro, 2'-O-aminopropyl (2'-O-AP), 2'-O-dimethylaminoethyl (2'-O-DMAOE), 2'-O- dimethylaminopropyl (2'-O-DMAP), T-O- dimethylaminoethyloxyethyl (2'-O-DMAEOE), or 2'- O-N-methylacetamido (2'-O-NMA) modified, LNA, ENA, PNA, HNA, morpholino, methylphosphonate nucleotides, thiolphosphonate nucleotides, 2'-fluoro N3-P5'- phosphoramidites, or combinations thereof are resistant toward nucle
- 2'-O-methyl modified polynucleic acid molecule is nuclease resistant (e.g., RNase H, DNase, 5'-3' exonuclease or 3'-5' exonuclease resistant).
- 2'0-methoxyethyl (2'-O-MOE) modified polynucleic acid molecule is nuclease resistant (e.g., RNase H, DNase, 5'-3' exonuclease or 3'-5' exonuclease resistant).
- 2'-O-aminopropyl modified polynucleic acid molecule is nuclease resistant (e.g., RNase H, DNase, 5'-3' exonuclease or 3'-5' exonuclease resistant).
- 2'-deoxy modified polynucleic acid molecule is nuclease resistant (e.g., RNase H, DNase, 5'-3' exonuclease or 3'-5' exonuclease resistant).
- T-deoxy-2'-fluoro modified polynucleic acid molecule is nuclease resistant (e.g., RNase H, DNase, 5'-3' exonuclease or 3'-5' exonuclease resistant).
- 2'-O- aminopropyl (2'-O-AP) modified polynucleic acid molecule is nuclease resistant (e.g., RNase H, DNase, 5'-3' exonuclease or 3'-5' exonuclease resistant).
- 2'-O- dimethylaminoethyl (2 -O-DMAOE) modified polynucleic acid molecule is nuclease resistant (e.g., RNase H, DNase, 5'-3' exonuclease or 3'-5' exonuclease resistant).
- 2'-O-dimethylaminopropyl (2'-O-DMAP) modified polynucleic acid molecule is nuclease resistant (e.g., RNase H, DNase, 5'-3' exonuclease or 3'-5' exonuclease resistant).
- T-O- dimethylaminoethyloxyethyl (2'-O-DMAEOE) modified polynucleic acid molecule is nuclease resistant (e.g., RNase H, DNase, 5'-3' exonuclease or 3'-5' exonuclease resistant).
- 2'-O-N-methylacetamido (2'-O-NMA) modified polynucleic acid molecule is nuclease resistant (e.g., RNase H, DNase, 5'-3' exonuclease or 3'-5' exonuclease resistant).
- LNA modified polynucleic acid molecule is nuclease resistant (e.g., RNase H, DNase, 5'-3' exonuclease or 3'-5" exonuclease resistant).
- ENA modified polynucleic acid molecule is nuclease resistant (e.g., RNase H, DNase, 5'-3' exonuclease or 3'-5' exonuclease resistant).
- HNA modified polynucleic acid molecule is nuclease resistant (e.g., RNase H, DNase, 5'-3' exonuclease or 3'-5' exonuclease resistant).
- morpholinos are nuclease resistant (e.g., RNase H, DNase, 5'-3' exonuclease or 3'-5' exonuclease resistant).
- PNA modified polynucleic acid molecule is resistant to nucleases (e.g., RNase H, DNase, 5'-3' exonuclease or 3'- 5' exonuclease resistant).
- methyl phosphonate nucleotides modified polynucleic acid molecule is nuclease resistant (e.g., RNase H, DNase, 5'-3' exonuclease or 3'-5' exonuclease resistant).
- thiolphosphonate nucleotides modified polynucleic acid molecule is nuclease resistant (e.g., RNase H, DNase, 5'-3' exonuclease or3'-5' exonuclease resistant).
- polynucleic acid molecule comprising 2'-fluoro N3-P5'-phosphoramidites is nuclease resistant (e.g., RNase H, DNase, 5'-3' exonuclease or3'-5' exonuclease resistant).
- the 5' conjugates described herein inhibit 5'-3' exonucleo lytic cleavage.
- the 3' conjugates described herein inhibit 3'-5' exonucleolytic cleavage.
- one or more artificial nucleotide analogues have increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- the one or more of the artificial nucleotide analogues comprising 2'-O-methyl, 2'- O-methoxyethyl (2'-O-MOE), 2'-O-aminopropyl, 2'-deoxy, T-deoxy-2'-fluoro, 2'-O- aminopropyl (2'-O-AP), 2'-O-dimethylaminoethyl (2'-O-DMAOE), 2'-O-dimethylaminopropyl (2'-O-DMAP), T-O- dimethylaminoethyloxyethyl (2'-O-DMAEOE), or 2'-O-N-methylacetamido (2'-O-NMA) modified, LNA, ENA, PNA, HNA, morpholino, methyl phosphon
- 2'-O-methyl modified polynucleic acid molecule has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- 2'-O-methoxy ethyl (2'-O-MOE)-modified polynucleic acid molecule has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- 2'-O-aminopropyl modified polynucleic acid molecule has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- 2'-deoxy modified polynucleic acid molecule has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- T-deoxy-2'-fluoro modified polynucleic acid molecule has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- 2'-O- aminopropyl (2'-O-AP)-modified polynucleic acid molecule has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- 2'-O-dimethylaminoethyl (2'-O-DMAOE)-modified polynucleic acid molecule has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- 2'-O-dimethylaminopropyl (2'-O-DMAP)- modified polynucleic acid molecule has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- T-O- dimethylaminoethyloxyethyl (2'-O-DMAEOE)-modified polynucleic acid molecule has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- 2'-O-N-methylacetamido (2'-O-NMA)-modified polynucleic acid molecule has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- LNA-modified polynucleic acid molecule has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- ENA-modified polynucleic acid molecule has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- PNA-modified polynucleic acid molecule has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- HNA-modified polynucleic acid molecule has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- morpholino-modified polynucleic acid molecule has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- methylphosphonate nucleotide-modified polynucleic acid molecule has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- thiolphosphonate nucleotide-modified polynucleic acid molecule has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- polynucleic acid molecule comprising 2'-fluoro N3-P5'- phosphoramidites has increased binding affinity toward their mRNA target relative to an equivalent natural polynucleic acid molecule.
- the increased affinity is illustrated with a lower Kd, a higher melt temperature (Tm), or a combination thereof.
- a polynucleic acid molecule is a chirally pure (or stereo pure) polynucleic acid molecule, or a polynucleic acid molecule comprising a single enantiomer.
- the polynucleic acid molecule comprises L-nucleotide.
- the polynucleic acid molecule comprises D-nucleotides.
- a polynucleic acid molecule composition comprises less than 30%, 25%, 20%, 15%, 10%, 5%, 4%, 3%, 2%, 1%, or less of its mirror enantiomer.
- a polynucleic acid molecule composition comprises less than 30%, 25%, 20%, 15%, 10%, 5%, 4%, 3%, 2%, 1%, or less of a racemic mixture.
- a polynucleic acid molecule may be further modified to include an aptamer-conjugating moiety.
- the aptamer conjugating moiety is a DNA aptamer-conjugating moiety.
- the aptamer conjugating moiety is Alphamer (Centauri Therapeutics), which comprises an aptamer portion that recognizes a specific cell-surface target and a portion that presents a specific epitopes for attaching to circulating antibodies.
- a polynucleic acid molecule described herein is further modified to include an aptamer conjugating moiety as described in: U.S. Patent Nos: 8,604,184, 8,591,910, and 7,850,975.
- a polynucleic acid molecule described herein is modified to increase its stability.
- the polynucleic acid molecule is RNA (e.g., siRNA), and the polynucleic acid molecule is modified to increase its stability.
- the polynucleic acid molecule is modified by one or more of the modifications described above to increase its stability.
- the polynucleic acid molecule is modified at the 2' hydroxyl position, such as by 2'-O-methyl, 2'-O-methoxyethyl (2'-O-MOE), 2'-O-aminopropyl, 2'-deoxy, T-deoxy-2'-fluoro, 2'-O-aminopropyl (2'-O-AP), 2'-O- dimethylaminoethyl (2'-O-DMAOE), 2'-O-dimethylaminopropyl (2'-O-DMAP), T-O- dimethylaminoethyloxyethyl (2'-O-DMAEOE), or 2'-O-N-methylacetamido (2'-O-NMA) modification or by a locked or bridged ribose conformation (e.g., LNA or ENA).
- a locked or bridged ribose conformation e.g., LNA or ENA
- the polynucleic acid molecule is modified by 2'-O-methyl and/or 2'-O-methoxyethyl ribose. In some cases, the polynucleic acid molecule also includes morpholinos, PNAs, HNA, methylphosphonate nucleotides, thiolphosphonate nucleotides, and/or 2'-fluoro N3-P5'- phosphoramidites to increase its stability. In some instances, the polynucleic acid molecule is a chirally pure (or stereo pure) polynucleic acid molecule. In some instances, the chirally pure (or stereo pure) polynucleic acid molecule is modified to increase its stability. Suitable modifications to the RNA to increase stability for delivery will be apparent to the skilled person.
- a polynucleic acid molecule in an ARC or ANC according to the invention has RNAi activity that modulates expression of RNA encoded by the target immunomodulator gene.
- a polynucleic acid molecule described herein is a double-stranded siRNA molecule that down-regulates expression of the target immunomodulatory protein, wherein one of the strands of the double-stranded siRNA molecule comprises a nucleotide sequence that is complementary to a nucleotide sequence of the immunomodulator gene or RNA encoded by the immunomodulator gene or a portion thereof, and wherein the second strand of the double-stranded siRNA molecule comprises a nucleotide sequence substantially similar to the nucleotide sequence of the immunomodulator gene or RNA encoded by the immunomodulator gene or a portion thereof.
- a polynucleic acid molecule described herein is a double-stranded siRNA molecule that down-regulates expression of the immunomodulator gene, wherein each strand of the siRNA molecule comprises about 15 to 25, 18 to 24, or 19 to about 23 nucleotides, and wherein each strand comprises at least about 14, 17, or 19 nucleotides that are complementary to the nucleotides of the other strand.
- a polynucleic acid molecule described herein is a double-stranded siRNA molecule that down-regulates expression of the immunomodulator gene, wherein each strand of the siRNA molecule comprises about 19 to about 23 nucleotides, and wherein each strand comprises at least about 19 nucleotides that are complementary to the nucleotides of the other strand.
- the RNAi activity occurs within a cell. In other instances, the RNAi activity occurs in a reconstituted in vitro system.
- a polynucleic acid molecule in an ARC or ANC according to the invention is a double-stranded polynucleotide molecule comprising self-complementary sense and antisense regions, wherein the antisense region comprises a nucleotide sequence that is complementary to a nucleotide sequence in a target nucleic acid molecule or a portion thereof and the sense region has a nucleotide sequence corresponding to the target nucleic acid sequence or a portion thereof.
- the polynucleic acid molecule is assembled from two separate polynucleotides, where one strand is the sense strand and the other is the antisense strand, wherein the antisense and sense strands are self- complementary (e.g., each strand comprises a nucleotide sequence that is complementary to a nucleotide sequence in the other strand; such as where the antisense strand and sense strand form a duplex or double stranded structure, for example wherein the double stranded region is about 19, 20, 21, 22, 23, or more base pairs); the antisense strand comprises nucleotide sequence that is complementary to a nucleotide sequence in a target nucleic acid molecule or a portion thereof and the sense strand comprises a nucleotide sequence corresponding to the target nucleic acid sequence or a portion thereof Alternatively, the polynucleic acid molecule is assembled from a single oligonucleotide, where the self-complementary sense and antisense regions
- a polynucleic acid molecule in an ARC or ANC according to the invention is a polynucleotide with a duplex, asymmetric duplex, hairpin or asymmetric hairpin secondary structure, having self-complementary sense and antisense regions, wherein the antisense region comprises a nucleotide sequence that is complementary to a nucleotide sequence in a separate target nucleic acid molecule or a portion thereof and the sense region has a nucleotide sequence corresponding to the target nucleic acid sequence or a portion thereof.
- the polynucleic acid molecule is a circular singlestranded polynucleotide having two or more loop structures and a stem comprising self- complementary sense and antisense regions, wherein the antisense region comprises nucleotide sequence that is complementary to a nucleotide sequence in a target nucleic acid molecule ora portion thereof and the sense region has a nucleotide sequence corresponding to the target nucleic acid sequence or a portion thereof, and wherein the circular polynucleotide is processed either in vivo or in vitro to generate an active polynucleic acid molecule capable of mediating RNAi.
- the polynucleic acid molecule also comprises a single stranded polynucleotide having a nucleotide sequence complementary to a nucleotide sequence in a target nucleic acid molecule or a portion thereof (for example, where such polynucleic acid molecule does not require the presence within the polynucleic acid molecule of a nucleotide sequence corresponding to the target nucleic acid sequence or a portion thereof), wherein the single stranded polynucleotide further comprises a terminal phosphate group, such as a 5 '-phosphate (see for example Martinez et al., 2002, Cell, 110, 563-574 and Schwarz et al., 2002, Molecular Cell, 10, 537- 568), or 5 ',3'-diphosphate.
- a terminal phosphate group such as a 5 '-phosphate (see for example Martinez et al., 2002, Cell, 110, 563-574 and Schwarz et al., 2002, Molecular Cell
- an asymmetric duplex is a linear polynucleic acid molecule comprising an antisense region, a loop portion that comprises nucleotides or nonnucleotides, and a sense region that comprises fewer nucleotides than the antisense region to the extent that the sense region has enough complimentary nucleotides to base pair with the antisense region and form a duplex with loop.
- an asymmetric hairpin polynucleic acid molecule comprises an antisense region having length sufficient to mediate RNAi in a cell or in vitro system (e.g.
- the asymmetric hairpin polynucleic acid molecule also comprises a 5 '-terminal phosphate group that is chemically modified.
- the loop portion of the asymmetric hairpin polynucleic acid molecule comprises nucleotides, non-nucleotides, linker molecules, or conjugate molecules.
- an asymmetric duplex is a polynucleic acid molecule having two separate strands comprising a sense region and an antisense region, wherein the sense region comprises fewer nucleotides than the antisense region to the extent that the sense region has enough complimentary nucleotides to base pair with the antisense region and form a duplex.
- an asymmetric duplex polynucleic acid molecule comprises an antisense region having length sufficient to mediate RNAi in a cell or in vitro system (e.g. about 19 to about 22 nucleotides) and a sense region having about 3 to about 18 nucleotides that are complementary to the antisense region.
- a universal base refers to nucleotide base analogs that form base pairs with each of the natural DNA/RNA. bases with little discrimination between them.
- Non-limiting examples of universal bases include C-phenyl, C-naphthyl and other aromatic derivatives, inosine, azole carboxamides, and nitroazole derivatives such as 3-mtropyrrole, 4-nitroindoie, 5-nitroindole, and 6-nitroindole as known in the art (see for example Loakes, 2001, Nucleic Acids Research, 29, 2437-2447).
- the subject ARCs or ANCs comprise one or more polynucleic acid molecules, typically RNAs, which polynucleic acid molecules may be synthesized as disclosed herein or using other known methods.
- a polynucleic acid molecule described herein is constructed using chemical synthesis and/or enzymatic ligation reactions using procedures known in the art.
- a polynucleic acid molecule is chemically synthesized using naturally occurring nucleotides or variously modified nucleotides designed to increase the biological stability of the molecules or to increase the physical stability of the duplex formed between the polynucleic acid molecule and target nucleic acids.
- Exemplary methods include those described in: U.S. Patent Nos. 5,142,047; 5,185,444; 5,889,136;
- the polynucleic acid molecule is produced biologically using an expression vector into which a polynucleic acid molecule has been subcloned in an antisense orientation
- a polynucleic acid molecule is synthesized via a tandem synthesis methodology, wherein both strands are synthesized as a single contiguous oligonucleotide fragment or strand separated by a cleavable linker which is subsequently cleaved to provide separate fragments or strands that hybridize and permit purification of the duplex.
- a polynucleic acid molecule is also assembled from two distinct nucleic acid strands or fragments wherein one fragment includes the sense region and the second fragment includes the antisense region of the molecule.
- an ARC or ANC according to the invention or a pharmaceutical composition containing as described herein is used for the treatment of a disease or disorder, typically an autoimmune or inflammatory disorder, cancer or a symptom associated therewith elicited by specific immune cell types.
- a disease or disorder typically an autoimmune or inflammatory disorder, cancer or a symptom associated therewith elicited by specific immune cell types.
- the ARC or ANC or a composition containing is used for the treatment of an autoimmune disease, e.g., one involving myeloid orT cells.
- the ARC or ANC or a composition containing is used for the treatment of a neoplastic, proliferative, neurodegenerative, neuroinflammatory, infectious, autoimmune or inflammatory disease or symptom thereof.
- the ARC or ANC or a composition containing is used to treat an autoimmune disease is selected from one or more of Acromegaly, Acquired aplastic anemia, Acquired hemophilia, Agammaglobulinemia, primary, Alopecia areata, Ankylosing spondylitis (AS), Anti-NMDA receptor encephalitis, Antiphospholipid syndrome (APS)
- Juvenile polymyositis Juvenile dermatomyositis
- juvenile myositis Kawasaki disease, Lambert-Eaton myasthenic syndrome (LEMS), Leukocytoclastic vasculitis, Lichen planus, Lichen sclerosus, Ligneous conjunctivitis, Linear IgA disease (LAD)
- the ARC or ANC is used for treatment of an autoimmune disease selected from the group consisting of Addison disease, arthritis, celiac disease, lupus, Grave's disease, myasthenia gravis, multiple sclerosis, ITP, rheumatoid arthritis, colitis, inflammatory bowel disease, pernicious anemia, Hashimoto's thyroiditis Sjogren's disease, asthma, type 2 diabetes, and autoimmune type I diabetes.
- an autoimmune disease selected from the group consisting of Addison disease, arthritis, celiac disease, lupus, Grave's disease, myasthenia gravis, multiple sclerosis, ITP, rheumatoid arthritis, colitis, inflammatory bowel disease, pernicious anemia, Hashimoto's thyroiditis Sjogren's disease, asthma, type 2 diabetes, and autoimmune type I diabetes.
- the ARC or ANC or a composition containing is used for treatment of an inflammatory disease selected from the group consisting of Fatty liver disease, Endometriosis, Type 2 diabetes, mellitus, Type 1 diabetes mellitus, Inflammatory bowel disease (IBD), Asthma, Rheumatoid arthritis, asthma, Obesity, Fibromyalgia, Lupus SLE, osteoarthritis, Rheumatoid Arthritis, Shingles Herpes Zoster, and Vasculitis.
- an inflammatory disease selected from the group consisting of Fatty liver disease, Endometriosis, Type 2 diabetes, mellitus, Type 1 diabetes mellitus, Inflammatory bowel disease (IBD), Asthma, Rheumatoid arthritis, asthma, Obesity, Fibromyalgia, Lupus SLE, osteoarthritis, Rheumatoid Arthritis, Shingles Herpes Zoster, and Vasculitis.
- the ANC or ARC or a composition containing is used to treat a neurodegenerative or neuroinflammatory disease, e.g., Alzheimer's disease, Amyotrophic lateral sclerosis, Friedreich ataxia, Huntington's disease, Lewy body disease, aphasia, Parkinson's disease or Spinal muscular atrophy.
- a neurodegenerative or neuroinflammatory disease e.g., Alzheimer's disease, Amyotrophic lateral sclerosis, Friedreich ataxia, Huntington's disease, Lewy body disease, aphasia, Parkinson's disease or Spinal muscular atrophy.
- the ARC or ANC or a composition containing is used for treatment of cancer or a symptom thereof associated with specific immune cell types.
- an ARC or ANC comprising a polynucleic acid molecule or a pharmaceutical composition described herein is used for the treatment of cancer.
- the cancer is a solid tumor.
- the cancer is a hematologic malignancy.
- the cancer is a relapsed or refractory cancer, or a metastatic cancer.
- the solid tumor is a relapsed or refractory solid tumor, or a metastatic solid tumor.
- the hematologic malignancy is a relapsed or refractory hematologic malignancy, or a metastatic hematologic malignancy.
- the cancer is a solid tumor.
- Exemplary solid tumor includes, but is not limited to, anal cancer, appendix cancer, bile duct cancer (i.e., cholangiocarcinoma), bladder cancer, brain tumor, breast cancer, cervical cancer, colon cancer, cancer of Unknown Primary (CUP), esophageal cancer, eye cancer, fallopian tube cancer, gastroenterological cancer, kidney cancer, liver cancer, lung cancer, medulloblastoma, melanoma, oral cancer, ovarian cancer, pancreatic cancer, parathyroid disease, penile cancer, pituitary tumor, prostate cancer, rectal cancer, skin cancer, stomach cancer, testicular cancer, throat cancer, thyroid cancer, uterine cancer, vaginal cancer, or vulvar cancer.
- CUP Unknown Primary
- a polynucleic acid molecule or a pharmaceutical composition described herein is used for the treatment of a solid tumor.
- a polynucleic acid molecule or a pharmaceutical composition described herein is used for the treatment of anal cancer, appendix cancer, bile duct cancer (i.e., cholangiocarcinoma), bladder cancer, brain tumor, breast cancer, cervical cancer, colon cancer, cancer of Unknown Primary (CUP), esophageal cancer, eye cancer, fallopian tube cancer, gastroenterological cancer, kidney cancer, liver cancer, lung cancer, medulloblastoma, melanoma, oral cancer, ovarian cancer, pancreatic cancer, parathyroid disease, penile cancer, pituitary tumor, prostate cancer, rectal cancer, skin cancer, stomach cancer, testicular cancer, throat cancer, thyroid cancer, uterine cancer, vaginal cancer, or vulvar cancer.
- the solid tumor is a relapsed or refractory solid tumor
- the cancer is a hematologic malignancy.
- the hematologic malignancy is a leukemia, a lymphoma, a myeloma, a non-Hodgkin's lymphoma, or a Hodgkin's lymphoma.
- the hematologic malignancy comprises chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma (SLL), high risk CLL, a non-CLL/SLL lymphoma, prolymphocytic leukemia (PLL), follicular lymphoma (FL), diffuse large B-cell lymphoma (DLBCL), mantle cell lymphoma (MCL), Waldenstrom's macroglobulinemia, multiple myeloma, extranodal marginal zone B cell lymphoma, nodal marginal zone B cell lymphoma, Burkitt's lymphoma, non-Burkitt high grade B cell lymphoma, primary mediastinal B-cell lymphoma (PMBL), immunoblastic large cell lymphoma, precursor B -lymphoblastic lymphoma, B cell prolymphocytic leukemia, lymphoplasmacytic lymphoma, splenic marginal zone lymphoma, plasma cell my
- a polynucleic acid molecule or a pharmaceutical composition described herein is used for the treatment of a hematologic malignancy. In some instances, a polynucleic acid molecule or a pharmaceutical composition described herein is used for the treatment of a leukemia, a lymphoma, a myeloma, a non-Hodgkin's lymphoma, or a Hodgkin's lymphoma.
- the hematologic malignancy comprises chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma (SLL), high risk CLL, a non-CLL/SLL lymphoma, prolymphocytic leukemia (PLL), follicular lymphoma (FL), diffuse large B-cell lymphoma (DLBCL), mantle cell lymphoma (MCL), Waldenstrom's macroglobulinemia, multiple myeloma, extranodal marginal zone B cell lymphoma, nodal marginal zone B cell lymphoma, Burkitt's lymphoma, non-Burkitt high grade B cell lymphoma, primary mediastinal B-cell lymphoma (PMBL), immunoblastic large cell lymphoma, precursor B- lymphoblastic lymphoma, B cell prolymphocytic leukemia, lymphoplasmacytic lymphoma, splenic marginal zone lymphoma, plasma cell myel
- the pharmaceutical formulations comprising an ARC or ANC according to the invention are administered to a subject by multiple administration routes including, but not limited to, parenteral (e.g., intravenous, subcutaneous, intramuscular), oral, intranasal, buccal, rectal, or transdermal administration routes.
- parenteral e.g., intravenous, subcutaneous, intramuscular
- oral e.g., intranasal
- buccal e.g., transdermal administration routes
- transdermal administration routes e.g., transdermal administration routes.
- the pharmaceutical composition describe herein is formulated for parenteral (e.g., intravenous, subcutaneous, intramuscular) administration.
- the pharmaceutical composition describe herein is formulated for oral administration.
- the pharmaceutical composition describe herein is formulated for intranasal administration.
- the pharmaceutical formulations include, but are not limited to, aqueous liquid dispersions, self-emulsifying dispersions, solid solutions, liposomal dispersions, aerosols, solid dosage forms, powders, immediate release formulations, controlled release formulations, fast melt formulations, tablets, capsules, pills, delayed release formulations, extended release formulations, pulsatile release formulations, multiparticulate formulations (e.g., nanoparticle formulations), and mixed immediate and controlled release formulations.
- aqueous liquid dispersions self-emulsifying dispersions, solid solutions, liposomal dispersions, aerosols, solid dosage forms, powders, immediate release formulations, controlled release formulations, fast melt formulations, tablets, capsules, pills, delayed release formulations, extended release formulations, pulsatile release formulations, multiparticulate formulations (e.g., nanoparticle formulations), and mixed immediate and controlled release formulations.
- the pharmaceutical formulation includes multiparticulate formulations.
- the pharmaceutical formulation includes nanoparticle formulations.
- nanoparticles comprise cMAP, cyclodextrin, or lipids.
- nanoparticles comprise solid lipid nanoparticles, polymeric nanoparticles, selfemulsifying nanoparticles, liposomes, microemulsions, or micellar solutions.
- Additional exemplary nanoparticles include, but are not limited to, paramagnetic nanoparticles, superparamagnetic nanoparticles, metal nanoparticles, fullerene-like materials, inorganic nanotubes, dendrimers (such as with covalently attached metal chelates), nanofibers, nanohorns, nano-onions, nanorods, nanoropes, and quantum dots.
- a nanoparticle is a metal nanoparticle, e.g., a nanoparticle of scandium, titanium, vanadium, chromium, manganese, iron, cobalt, nickel, copper, zinc, yttrium, zirconium, niobium, molybdenum, ruthenium, rhodium, palladium, silver, cadmium, hafnium, tantalum, tungsten, rhenium, osmium, iridium, platinum, gold, gadolinium, aluminum, gallium, indium, tin, thallium, lead, bismuth, magnesium, calcium, strontium, barium, lithium, sodium, potassium, boron, silicon, phosphorus, germanium, arsenic, antimony, and combinations, alloys, or oxides thereof.
- a metal nanoparticle e.g., a nanoparticle of scandium, titanium, vanadium, chromium, manganese, iron, cobalt,
- a nanoparticle includes a core or a core and a shell, as in a coreshell nanoparticle.
- a nanoparticle is further coated with molecules for attachment of functional elements (e.g., with one or more of a polynucleic acid molecule or binding moiety described herein).
- a coating comprises chondroitin sulfate, dextran sulfate, carboxymethyl dextran, alginic acid, pectin, carrageenan, fucoidan, agaropectin, porphyran, karaya gum, gellan gum, xanthan gum, hyaluronic acids, glucosamine, galactosamine, chitin (or chitosan), polyglutamic acid, polyaspartic acid, lysozyme, cytochrome C, ribonuclease, trypsinogen, chymotrypsinogen, a-chymotrypsin, polylysine, polyarginine, histone, protamine, ovalbumin, dextrin, or
- a nanoparticle has at least one dimension of less than about 500nm, 400nm, 300nm, 200nm, or lOOnm.
- the nanoparticle formulation comprises paramagnetic nanoparticles, superparamagnetic nanoparticles, metal nanoparticles, fullerene-like materials, inorganic nanotubes, dendrimers (such as with covalently attached metal chelates), nanofibers, nanohorns, nano-onions, nanorods, nanoropes or quantum dots.
- a polynucleic acid molecule or a binding moiety described herein is conjugated either directly or indirectly to the nanoparticle. In some instances, at least 1, 5, 10, 15, 20, 30, 40, 50, 60, 70, 80, 90, 100, or more poly nucleic acid molecules or binding moieties described herein are conjugated either directly or indirectly to a nanoparticle.
- the pharmaceutical formulation comprise a delivery vector, e.g., a recombinant vector, for the delivery of the polynucleic acid molecule into cells.
- the recombinant vector is DNA plasmid.
- the recombinant vector is a viral vector.
- Exemplary viral vectors include vectors derived from adeno-associated virus, retrovirus, adenovirus, or alphavirus.
- the recombinant vectors capable of expressing the polynucleic acid molecules provide stable expression in target cells.
- viral vectors are used that provide for transient expression of polynucleic acid molecules.
- the pharmaceutical formulations include a carrier or carrier materials selected on the basis of compatibility with the composition disclosed herein, and the release profile properties of the desired dosage form.
- exemplary carrier materials include, e.g., binders, suspending agents, disintegration agents, filling agents, surfactants, solubilizers, stabilizers, lubricants, wetting agents, diluents, and the like.
- Pharmaceutically compatible carrier materials include, but are not limited to, acacia, gelatin, colloidal silicon dioxide, calcium glycerophosphate, calcium lactate, maltodextrin, glycerine, magnesium silicate, polyvinylpyrrollidone (PVP), cholesterol, cholesterol esters, sodium caseinate, soy lecithin, taurocholic acid, phosphotidylcholine, sodium chloride, tricalcium phosphate, dipotassium phosphate, cellulose and cellulose conjugates, sugars sodium stearoyl lactylate, carrageenan, monoglyceride, diglyceride, pregelatinized starch, and the like.
- PVP polyvinylpyrrollidone
- the pharmaceutical formulations further include pH adjusting agents or buffering agents, which include acids such as acetic, boric, citric, lactic, phosphoric, and hydrochloric acids; bases such as sodium hydroxide, sodium phosphate, sodium borate, sodium citrate, sodium acetate, sodium lactate and tris- hydroxymethylaminomethane; and buffers such as citrate/dextrose, sodium bicarbonate, and ammonium chloride.
- acids such as acetic, boric, citric, lactic, phosphoric, and hydrochloric acids
- bases such as sodium hydroxide, sodium phosphate, sodium borate, sodium citrate, sodium acetate, sodium lactate and tris- hydroxymethylaminomethane
- buffers such as citrate/dextrose, sodium bicarbonate, and ammonium chloride.
- acids, bases and buffers are included in an amount required to maintain pH of the composition in an acceptable range.
- the pharmaceutical formulation includes one or more salts in an amount required to bring osmolality of the composition into an acceptable range.
- salts include those having sodium, potassium, or ammonium cations and chloride, citrate, ascorbate, borate, phosphate, bicarbonate, sulfate, thiosulfate, or bisulfite anions; suitable salts include sodium chloride, potassium chloride, sodium thiosulfate, sodium bisulfite, and ammonium sulfate.
- the pharmaceutical formulations further include diluent which are used to stabilize compounds because they provide a more stable environment.
- Salts dissolved in buffered solutions are utilized as diluents in the art, including, but not limited to a phosphate-buffered saline solution.
- diluents increase bulk of the composition to facilitate compression or create sufficient bulk for homogenous blend for capsule filling.
- Such compounds include e.g., lactose, starch, mannitol, sorbitol, dextrose, microcrystalline cellulose such as Avicel ® ; dibasic calcium phosphate, dicalcium phosphate dihydrate; tricalcium phosphate, calcium phosphate; anhydrous lactose, spray-dried lactose; pregelatinized starch, compressible sugar, such as Di-Pac ® (Amstar); mannitol, hydroxypropylmethylcellulose, hydroxypropylmethylcellulose acetate stearate, sucrose-based diluents, confectioner's sugar; monobasic calcium sulfate monohydrate, calcium sulfate dihydrate; calcium lactate trihydrate, dextrates; hydrolyzed cereal solids, amylose; powdered cellulose, calcium carbonate; glycine, kaolin; mannitol, sodium chloride; inositol, bentonite, and the like.
- the pharmaceutical formulations include disintegration agents or disintegrants to facilitate the breakup or disintegration of a substance.
- disintegrate includes both the dissolution and dispersion of the dosage form when contacted with gastrointestinal fluid.
- disintegration agents include a starch, e.g., a natural starch such as corn starch or potato starch, a pregelatinized starch such as National 1551 or Amijel ® , or sodium starch glycolate such as Promogel ® or Explotab ® ; a cellulose such as a wood product, methylcrystalline cellulose, e.g., Avicel ® , Avicel ® PH101, Avicel ® PH102, Avicel ® PH105, Elcema ® P100, Emcocel ® , Vivacel ® , MingTia ® , and Solka-Floc ® , methylcellulose, croscarmellose, ora cross-linked cellulose, such as cross-linked sodium carboxymethylcellulose (Ac-Di-Sol ® ), cross-linked carboxymethylcellulose, or cross-linked croscarmellose; a cross-linked starch such as sodium starch glycolate, a cross-linked polymer such as
- the pharmaceutical formulations include filling agents such as lactose, calcium carbonate, calcium phosphate, dibasic calcium phosphate, calcium sulfate, microcrystalline cellulose, cellulose powder, dextrose, dextrates, dextran, starches, pregelatinized starch, sucrose, xylitol, lactitol, mannitol, sorbitol, sodium chloride, polyethylene glycol, and the like.
- lactose calcium carbonate, calcium phosphate, dibasic calcium phosphate, calcium sulfate, microcrystalline cellulose, cellulose powder, dextrose, dextrates, dextran, starches, pregelatinized starch, sucrose, xylitol, lactitol, mannitol, sorbitol, sodium chloride, polyethylene glycol, and the like.
- Lubricants and glidants are also optionally included in the pharmaceutical formulations described herein for preventing, reducing, or inhibiting adhesion or friction of materials.
- Exemplary lubricants include, e.g., stearic acid, calcium hydroxide, talc, sodium stearyl fumerate, a hydrocarbon such as mineral oil, or hydrogenated vegetable oil such as hydrogenated soybean oil (Sterotex ® ), higher fatty acids and their alkali-metal and alkaline earth metal salts, such as aluminum, calcium, magnesium, zinc, stearic acid, sodium stearates, glycerol, talc, waxes, Stearowet ® , boric acid, sodium benzoate, sodium acetate, sodium chloride, leucine, a polyethylene glycol (e.g., PEG-4000) ora methoxypolyethylene glycol such as CarbowaxTM, sodium oleate, sodium benzoate, glyceryl behenate, polyethylene
- Solubilizers include compounds such as triacetin, triethylcitrate, ethyl oleate, ethyl caprylate, sodium lauryl sulfate, sodium doccusate, vitamin E TPGS, dimethylacetamide, N- methyl pyrrolidone, N-hydroxyethylpyrrolidone, polyvinylpyrrolidone, hydroxypropylmethyl cellulose, hydroxypropyl cyclodextrins, ethanol, n-butanol, isopropyl alcohol, cholesterol, bile salts, polyethylene glycol 200-600, glycofurol, transcutol, propylene glycol, dimethyl isosorbide and the like.
- Stabilizers include compounds such as any antioxidation agents, buffers, acids, preservatives and the like.
- Suspending agents include compounds such as polyvinylpyrrolidone (e.g., polyvinylpyrrolidone K12, polyvinylpyrrolidone K17, polyvinylpyrrolidone K25, or polyvinylpyrrolidone K30), vinyl pyrrolidone/vinyl acetate copolymer (S630), polyethylene glycol (e.g., the polyethylene glycol has a molecular weight of about 300 to about 6000, or about 3350 to about 4000, or about 7000 to about 5400), sodium carboxymethylcellulose, methylcellulose, hydroxypropylmethylcellulose, hydroxymethylcellulose acetate stearate, polysorbate-80, hydroxyethylcellulose, sodium alginate, gums (such as, e.g., gum tragacanth and gum acacia, guar gum, xanthans, including xanthan gum), sugars, cellulosics (such as, e.g., gum
- Surfactants include compounds such as sodium lauryl sulfate, sodium docusate, Tween 60 or 80, triacetin, vitamin E TPGS, sorbitan monooleate, polyoxyethylene sorbitan monooleate, polysorbates, polaxomers, bile salts, glyceryl monostearate, copolymers of ethylene oxide and propylene oxide, e.g., Pluronic ® (BASF), and the like.
- compounds such as sodium lauryl sulfate, sodium docusate, Tween 60 or 80, triacetin, vitamin E TPGS, sorbitan monooleate, polyoxyethylene sorbitan monooleate, polysorbates, polaxomers, bile salts, glyceryl monostearate, copolymers of ethylene oxide and propylene oxide, e.g., Pluronic ® (BASF), and the like.
- Pluronic ® Pluronic ®
- Additional surfactants include polyoxyethylene fatty acid glycerides and vegetable oils, e.g., polyoxyethylene (60) hydrogenated castor oil; and polyoxyethylene alkylethers and alkylphenyl ethers, e.g., octoxynol 10, octoxynol 40. Sometimes, surfactants are included to enhance physical stability or for other purposes.
- Viscosity enhancing agents include, e.g., methyl cellulose, xanthan gum, carboxymethyl cellulose, hydroxypropyl cellulose, hydroxypropylmethyl cellulose, hydroxypropylmethyl cellulose acetate stearate, hydroxypropy I methyl cellulose phthalate, carbomer, polyvinyl alcohol, alginates, acacia, chitosans, and combinations thereof.
- Wetting agents include compounds such as oleic acid, glyceryl monostearate, sorbitan monooleate, sorbitan monolaurate, triethanolamine oleate, polyoxyethylene sorbitan monooleate, polyoxyethylene sorbitan monolaurate, sodium docusate, sodium oleate, sodium lauryl sulfate, sodium doccusate, triacetin, Tween 80, vitamin E TPGS, ammonium salts, and the like.
- the pharmaceutical compositions comprising an ARC or ANC according to the invention are administered for therapeutic applications.
- the pharmaceutical composition is administered once per day, twice per day, three times per day or more.
- the pharmaceutical composition is administered daily, every day, every alternate day, five days a week, once a week, every other week, two weeks per month, three weeks per month, once a month, twice a month, three times per month, or more.
- the pharmaceutical composition is administered for at least 1 month, 2 months, 3 months, 4 months, 5 months, 6 months, 7 months, 8 months, 9 months, 10 months, 11 months, 12 months, 18 months, 2 years, 3 years, or more.
- one or more pharmaceutical compositions in an ARC or ANC according to the invention are administered simultaneously, sequentially, or at an interval period of time. In some embodiments, one or more pharmaceutical compositions are administered simultaneously. In some cases, one or more pharmaceutical compositions are administered sequentially. In additional cases, one or more pharmaceutical compositions are administered at an interval period of time (e.g., the first administration of a first pharmaceutical composition is on day one followed by an interval of at least 1, 2, 3, 4, 5, or more days prior to the administration of at least a second pharmaceutical composition).
- two or more different pharmaceutical compositions are coadministered. In some instances, the two or more different pharmaceutical compositions are co-administered simultaneously. In some cases, the two or more different pharmaceutical compositions are co-administered sequentially without a gap of time between administrations. In other cases, the two or more different pharmaceutical compositions are co-administered sequentially with a gap of about 0.5 hour, 1 hour, 2 hour, 3 hour, 12 hours, 1 day, 2 days, or more between administrations.
- the administration of the composition is given continuously; alternatively, the dose of the composition being administered is temporarily reduced or temporarily suspended for a certain length of time (i.e., a "drug holiday").
- the length of the drug holiday varies between 2 days and 1 year, including by way of example only, 2 days, 3 days, 4 days, 5 days, 6 days, 7 days, 10 days, 12 days, 15 days, 20 days, 28 days, 35 days, 50 days, 70 days, 100 days, 120 days, 150 days, 180 days, 200 days, 250 days, 280 days, 300 days, 320 days, 350 days, or 365 days.
- the dose reduction during a drug holiday is from 1096- 100%, including, by way of example only, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, or 100%.
- a maintenance dose is administered if necessary. Subsequently, the dosage or the frequency of administration, or both, are optionally reduced, as a function of the symptoms, to a level at which the improved disease, disorder or condition is retained.
- the amount of a given agent that corresponds to such an amount varies depending upon factors such as the particular compound, the severity of the disease, the identity (e.g., weight) of the subject or host in need of treatment, but nevertheless is routinely determined in a manner known in the art according to the particular circumstances surrounding the case, including, e.g., the specific agent being administered, the route of administration, and the subject or host being treated.
- the desired dose is conveniently presented in a single dose or as divided doses administered simultaneously (or over a short period of time) or at appropriate intervals, for example as two, three, four or more sub-doses per day.
- toxicity and therapeutic efficacy of such therapeutic regimens are determined by standard pharmaceutical procedures in cell cultures or experimental animals, including, but not limited to, the determination of the LD50 (the dose lethal to 50% of the population) and the ED50 (the dose therapeutically effective in 50% of the population).
- the dose ratio between the toxic and therapeutic effects is the therapeutic index and it is expressed as the ratio between LD50 and ED50.
- Compounds exhibiting high therapeutic indices are preferred.
- the data obtained from cell culture assays and animal studies are used in formulating a range of dosage for use in human.
- the dosage of such compounds lies preferably within a range of circulating concentrations that include the ED50 with minimal toxicity. The dosage varies within this range depending upon the dosage form employed and the route of administration utilized.
- kits and articles of manufacture for use with one or more of the compositions and methods described herein.
- Such kits include a carrier, package, or container that is compartmentalized to receive one or more containers such as vials, tubes, and the like, each of the container(s) comprising one of the separate elements to be used in a method described herein.
- Suitable containers include, for example, bottles, vials, syringes, and test tubes.
- the containers are formed from a variety of materials such as glass or plastic.
- the articles of manufacture provided herein contain packaging materials.
- packaging materials include, but are not limited to, blister packs, bottles, tubes, bags, containers, bottles, and any packaging material suitable for a selected formulation and intended mode of administration and treatment.
- the container(s) include an nucleic acid molecule described herein which specifically binds to an immunomodulator gene or RNA encoded thereby.
- kits optionally include an identifying description or label or instructions relating to its use in the methods described herein.
- a kit typically includes labels listing contents and/or instructions for use and package inserts with instructions for use. A set of instructions will also typically be included.
- a label is on or associated with the container.
- a label is on a container when letters, numbers, or other characters forming the label are attached, molded or etched into the container itself; a label is associated with a container when it is present within a receptacle or carrier that also holds the container, e.g., as a package insert.
- a label is used to indicate that the contents are to be used for a specific therapeutic application. The label also indicates directions for use of the contents, such as in the methods described herein.
- the pharmaceutical compositions are presented in a pack or dispenser device which contains one or more unit dosage forms containing a compound provided herein.
- the pack for example, contains metal or plastic foil, such as a blister pack.
- the pack or dispenser device is accompanied by instructions for administration.
- the pack or dispenser is also accompanied with a notice associated with the container in a form prescribed by a governmental agency regulating the manufacture, use, or sale of pharmaceuticals, which notice is reflective of approval by the agency of the form of the drug for human or veterinary administration.
- a notice associated with the container in a form prescribed by a governmental agency regulating the manufacture, use, or sale of pharmaceuticals which notice is reflective of approval by the agency of the form of the drug for human or veterinary administration.
- Such notice for example, is the labeling approved by the U.S. Food and Drug Administration for prescription drugs, or the approved product insert.
- RNA-ARC direct antibody-RNA conjugate
- ASO antisense oligonucleotides
- RNA-ARCs may be used to specifically inhibit mRNAs for key immune pathways, including PIK3CA and TNF as well as other known and novel pathways involved in immune regulation, tolerance, and metabolism.
- siRNAs inhibiting important immunological targets Glutl, PI3K, BTK, TNF, RORC
- ARCs bind their functionality and inhibit activation of the respective immune pathways, making them attractive targets in autoimmune disease treatment space.
- ASO- mediated inhibition of CD39 an ectonucleotidase involved in generating extracellular adenosine through ATP hydrolysis, results in an increased activation and cytokine production by T cells [PMID: 30871609].
- ARC-delivered CD39 retained its activity and should be considered a promising approach in cancer treatment.
- INX201 a humanized anti-human VISTA antibody on a human IgGl/kappa backbone with L234A/L235A/E269R/K322A silencing mutations in the Fc region, was used as a payload delivery vehicle.
- a humanized anti-human VISTA antibody on a human IgGl/kappa backbone with L234A/L235A/E269R/K322A silencing mutations in the Fc region was used as a payload delivery vehicle.
- ARC has similar binding and internalization properties as compared to free INX201 Mab
- siRNA payloads specific to immunologically relevant targets, (https://www.idtdna.com/pages/products/functional-genomics/dsirnas- and-trifecta-rnai-kits), which knock-down genes in regular transfection setting (inhibit target mRNA levels to ⁇ 30%) retain their function when conjugated to a Mab and delivered into the target cells;
- siRNA conjugated to an anti-VISTA Mab INX201 facilitated siRNA accumulation and function inside K562-VISTA cells
- siRNA conjugated to an anti-VISTA Mab INX201 had function in human PBMC and T cells, manifested in reduced proliferation and cytokine production;
- CD39 ASO conjugated to an anti-VISTA Mab INX201 had function in human PBMC, manifested in increased cytokine production.
- Fc heavy chain constant region (hinge/CH2/CH3) of an antibody
- MFI mean fluorescent intensity (signal used in flow cytometry)
- peripheral blood mononuclear cells xeno-GvHD xenogeneic graft versus host disease (autoimmune disease animal model)
- ASO antisense oligonucleotides siRNA small interfering RNA (dsRNA of 20-24 nucleotides)
- IgGl anti-VISTA Mab INX201.
- This Mab is a humanized anti-human VISTA antibody on a human IgGl/kappa backbone with L234A/L235A/E269R/K322A silencing mutations in the Fc region, was used as a payload delivery vehicle.
- K562-VISTA cell line assays binding, internalization, transfections, qPCR
- K562-VISTA VISTA expressing cells, made in-house from WT K562, ATCC CCL-243
- DNA pRP[Exp]-CAG>3xNLS/EGFP
- Vector ID VB900137-6122yyc (VectorBuilder, MaxH VB900137-6122yyc), for generation of GFP+ cell pool; the vector contains PuroR puromycin resistance gene
- Test articles free INX201 or INX201 ARCs, for example (INX201-eGFP_siRNA_Cy5)
- the base medium for this cell line is ATCC-formulated Iscove's Modified Dulbecco's Medium, Catalog No. 30-2005. To make the complete growth medium, add the following components to the base medium: FBS to a final concentration of 10%
- Cultures can be maintained by the addition or replacement of fresh medium. Start new cultures at 1 x 10 5 viable cells/mL. Subculture at 1 x 10 6 cells/ml
- Buffer RLT Plus may form a precipitate during storage. If necessary, redissolve by warming, and then place at room temperature.
- Buffer RPE is supplied as a concentrate. Before using it for the first time, add four volumes of ethanol (96-100%), as indicated on the bottle.
- the RNA segment to be transcribed and later amplified can be at least 3 kb long.
- incubation time may be increased to 60 minutes.
- the reaction volume is 20 ⁇ l.
- the reaction volume is 20 ⁇ l.
- the reaction volume is 20 ⁇ l.
- Human peripheral blood was obtained from apheresis cones provided by volunteer donors at the DHMC blood donor program.
- the cone blood was diluted at 1:4 in PBS and carefully deposited on top of 13 ml of Histopaque 1077. After 20 min centrifugation at 850 g
- the mononuclear cells from the Histopaque / PBS interfaced were collected. After one wash in PBS, the PBMC were resuspended at 100x10 6 cells/ml for freezing (90% DMSO, 10% FBS) or at 10x10 6 cells/ml for experiments
- RNA conjugates using the following payloads: RORC, PIK3CA, TNFa, BTK, Glutl, CD45 (siRNA); CD39 ASO
- Anti-CD3/CD28 beads - stimulation of human PBMCs orT cells CD3/CD28 T-cell Activator Dynabeads were used to activate human T cells for 3 days.
- a. Mix Dynabeads by vortexing and aliquot 2.5 ⁇ l/well of beads in a 2 ml Eppendorf tube.
- b. Add media to 2 ml, vortex, and place the tube on the magnet.
- d Add beads directly to the cells and plate in a U bottom 96-well plate, and add 100 ⁇ l of media to the total volume of 200 ⁇ l.
- Cell number 0.5 million/well for PBMCs; 0.1 million/well for T cells. Bead to T cell ratio 1:2.
- PBMCs were stimulated with 10 ng/ml LPS (Ams bio)
- Applicant has selected a variety of immunologically relevant targets, including ones with (1) existing approved drugs against (TNFa); (2) validated pathways with on-target toxicity concerns (PI3K), as well as (3) novel targets (Glutl), see Table 2.
- TNFa existing approved drugs against
- PI3K validated pathways with on-target toxicity concerns
- Glutl novel targets
- the first class of targets is used for proof of concept studies (TNFa), whereas the other targets corroborate the intrinsic advantages of the subject ARCs/ANCs which preferentially target myeloid and lymphoid cells.
- RNA-ARCs should minimize or eliminate toxicities associated with non-immune tissue targeting (PI3K) and/or broad expression of the target (Glutl) or the nature of the target, such as it being transcription factor (RORC), which toxicities heretofore have limited or precluded the successful development of biologies and/or small molecule inhibitors against these types of "difficult" targets (TNFa [PMID: 34301319, PMID: 29158574]; BTK [PMID: 27192942, PMID: 31431692, PMID: 33122850, PMID: 36903645]; PI3K [PMID: 31928691, PMID: 34127844, PMID: 26093105]; RORC [PMID: 30010338, PMID: 34040108]; Glutl [PMID: 35878663, PMID: 34018847]; CD39 [PMID: 30871609]).
- Table 9 Exemplary Payloads
- siRNA or ASO unconjugated payloads (1-4 per target) were ordered from IDT, (https://www.idtdna.com/pages/products/functional-genomics/dsirnas-and-trifecta-rnai- kits).
- K562-VISTA cells were transfected with 200 nM siRNA (or ASO) alone, and target knockdown was analyzed by qRTPCR using the ddct method and reported as fold over control.
- Scrambled siRNA or ASO controls were also ordered from IDT ( Figures 1-2). Payloads marked with (*) were selected for conjugation to a delivery vehicle (anti-VISTA Mab INX201).
- Anti-VISTA Mab and payloads used for conjugations are described in Tables 1-2 and Appendices 1-2.
- Oligo Conjugation Kit (Abeam, #ab218260) was used for all conjugations. All RNA oligos were purified by HPLC and resuspended at 100 ⁇ M. Lysine-based conjugation via the amine-reactive group, a widely utilized non-specific conjugation strategy was used. All payloads were synthesized at IDT and contained a 3'-sense strand Cy5 label and 5'-amine on the antisense strand (see Appendix 2).
- ARC conjugation was analyzed by SDS-PAGE and subsequent silver stain to visualize protein. Briefly, ARCs were mixed with reducing 2x Laemmli Sample Buffer (Bio- Rad, # 1610737) and incubated at 80°C for 5 min. Reduced samples were resolved using 4- 15% Mini-Protean TGX Precast Gel (Bio-Rad, #4561083) following the manufacturer's instructions. After the electrophoresis, the gel was rinsed with Mili-Q water and stained for protein using SilverQuest reagents (Thermo Fisher, #LC6070). CD45 siRNA ARC and SOCS1 ASO ARC were analyzed as representative ARCs for both siRNA and ASO payloads. Both showed expected conjugation patterns (Figure 3). Nanodrop measurements performed for ASO ARC were: 37.2 ⁇ M for RNA and 8.0 ⁇ M for protein; therefore, the DAR was ⁇ 4.65.
- Binding (to VISTA target) and internalization were compared between free INX201 and INX201 eGFP siRNA ARC.
- K562-VISTA cells were incubated with 200 nM of free Mab or ARC. Binding and Internalization were measured in a time course. Binding to target was equivalent between free antibody and ARC as evident as a zero hour time point (visualized as 0.1 h, Figure 4A). Internalization was also similar between free INX201 antibody and ARC and was rapid and efficient, as previously described by us. Specifically, within 30 minutes, over 90% of antibodies were internalized in both ARC and free Ab samples. It is evident by the lack of antibodies detected on the cell surface in the time course assay (Figure 4A).
- Cy5 label present on siRNA, was used to detect RNA inside the cells (given the lack of antibody detection on the cell surface due to internalization in the ARC samples, we assume that most of Cy5 detection is intracellular). As evidenced by a time course experiment, Cy5 was readily detected inside K562-VISTA cells and was preserved for the duration of the experiment (24 hours).
- K562-VISTA cells were transfected with plasmid DNA expressing eGFP, and a GFP+ cell pool was used 14-28 days after transfection. The cell pool was treated with no drug or eGFP ARC. eGFP or CD45 protein levels were measured by flow cytometry (Table 10).
- Table 10 Flow cytometry panel/channels used for ARC QC in K562 cells.
- the dash line represents the maximal level of CD45 protein knockdown expected, based on the payload (siRNA) potency, which is 50%, as measured from free transfected CD45 siRNA at 48 hours (right bar); cells treated for 72 hours with 200 nM eGFP ARC (middle bar) or with no drug (left bar).
- siRNA payload
- FIG. 5C CD45 levels measured in a repeat experiment. Cells were treated for 72 hours with no drug (left bar), free siRNA, no transfection (middle bar), and 200 nM ARC (right bar).
- TNFa protein measured by Luminex
- TNFa ARC was efficiently knocked down by TNFa ARC in a dose-dependent manner for both LPS-stimulated PBMCs (Figure 6A) and for anti-CD3/CD28-stimulated PBMCs ( Figure 6B).
- the level of knockdown was >80% (LPS stimulation) and ⁇ 50% (anti-CD3/CD28 stimulation).
- anti- CD3/CD28 stimulation we used cells collected at 72 hours for qRTPCR to validate that the target TNFa was knocked down at the mRNA level. In this case, we saw about 60% target knockdown on RNA level in PBMCs, which correlated well with the 50% reduction in TNFa cytokine levels (Figure 6C).
- TNFa ARC but not free TNFa siRNA
- PI3K ARC but not free PI3K siRNA
- Figure 7B also slowed down the proliferation of T cells in a dose-dependent manner
- BTK ARC was tested in human PBMCs activated with LPS or anti-CD3/CD28 dynabeads as described above.
- BTK ARC but not free BTK siRNA, was able to reduce activation of CD16+ monocytes as measured by CD69 levels at 48 hours post drug addiction (Figure 8A). CD69 levels were reduced to levels comparable to unstimulated cells.
- BTK ARC also slowed down the proliferation of T cells as measured at 72 hours post-stimulation ( Figure 8B). Free INX201 antibody or free untransfected siRNA payload had no impact on these assays.
- CD69 levels were measured in A; % newly proliferating cells in B; A single technical replicate per concentration point was used; no ARC was visualized as 0.1 nM (due to log scale); unstim - unstimulated cells; MFI - mean fluorescent intensity.
- RORC ARC was also tested on purified human T cells, stimulated with anti-CD3/CD28 dynabeads for 72 hours. Following incubation, media was collected, and cytokine levels were measured by Luminex for two human donors. IFNg, IL6, and IL12p40 levels were reduced 2-4 fold in a dose-dependent manner when T cells were treated with RORC ARC (Figure 1QA-C).
- a 96-well flat-bottom plate (Thermo Scientific Nunc Immuno Maxisorp, cat# 442404) was coated with hIX50 (human VISTA ECD, produced at Aragen Bioscience for ImmuNext) at 20nM in PBS for one hour at room temperature (RT).
- hIX50 human VISTA ECD, produced at Aragen Bioscience for ImmuNext
- the wells were washed three times with PT (PBS with 0.05% Tween 20) and then blocked with PTB (PBS with 0.05% Tween 20 and 1% BSA) for 30 minutes at RT.
- INX201 Fab was diluted from 1000 nM to 0.02 nM in PTB and then added to the wells for 1 hour at RT, or INX201 Mab was diluted from 20 nM to 0.002 nM in PTB and then added to the wells for 1 hour at RT. Following incubation, the wells were washed three times with PT, then mouse anti-human kappa coupled to HRP (SouthernBiotech, cat# 9230-05) was used as a detection reagent at a dilution of 1/2000 and incubated for 1.5 hours at RT. Following three washes, the ELISA reaction was revealed using TMB (Thermo Scientific, cat# 34028) as a colorimetric substrate. After a few minutes at RT, the reaction was stopped with IM H2S04. OD450 was read on Molecular devices Spectramax M3 plate Reader and analyzed with SoftMaxPro software.
- Figure 12 shows platform optimization using an anti-VISTA Fab instead of Mab.
- the experiment in Figure 12A compares binding to human VISTA ECD between INX201 Mab (squares) and INX201 Fab (circles) by ELISA.
- Figure 13 shows that the exemplary ARC does not impact T cell viability in experiments wherein human PBMCs were activated with anti-CD3/CD28 beads and treated with PI3K ARC (0-200 nM, triangles) or free INX201 (0-200 nM, squares) for 72 hours.
- the curves were generated from inhibitor vs. response three-parameter non-linear regression analysis (GraphPad Prism 9). A single technical replicate per concentration point was used; no ARC was visualized as 0.1 nM (due to the log scale); unstim - unstimulated cells.
- Example 2 In vivo evaluation of anti-VISTA antibody RNA conjugates (ARCs) in GVHD
- Table 11 shows the anti-VISTA and isotype control MAbs, and ARCs used for delivery of payloads.
- the antisense strand of siRNA represents an active drug. Once delivered inside cells of interest via our VISTA delivery platform, it binds to and triggers degradation of its target mRNA, thereby reducing the level of target mRNA and protein.
- the GVHD model was used to assess the efficacy of anti-VISTA antibody RNA conjugates (ARCs) because it is a well accepted autoimmune and inflammatory model.
- ARCs anti-VISTA antibody RNA conjugates
- humanized mouse models of xenogeneic Graft versus Host Disease (GvHD) allow the study of immunomodulatory compounds specific to human drug targets in vivo and their effects on markers of inflammation such as inflammatory cytokines.
- PBMCs peripheral blood mononuclear cells
- mice GVHD model combines the features of the NOD/ShiLtJ background, the severe combined immune deficiency mutation (scid), and IL2 receptor gamma chain deficiency. As a result, these mice lack mature T cells, B cells, or functional NK cells, and are deficient in cytokine signaling, leading to better engraftment of human hematopoietic stem cells and peripheral blood mononuclear cells than any other published mouse strain.
- Example 2 The same procedures disclosed in Example 1 were used for the Human PBMC and T cell activation and proliferation assays.
- Example 2 The same procedures disclosed in Example 1 were used for the flow cytometry - based cell activation analysis.
- the In vitro flow cytometry panel used in the analysis is set forth in Table 12.
- mice were 8-week-old male NSG purchased from Jackson Laboratory.
- mice per group were enrolled.
- mice were injected intravenously (i.v.) with 10x10 6 human PBMCs premixed with the antibodies in 200 ⁇ l of PBS via tail vein injection.
- mice were bled, plasma was collected for cytokine analysis and T cell numbers were evaluated using flow cytometry. Mice were weighed thrice a week to monitor disease progression and euthanized if their weight dropped below 75% of their initial weight.
- Human peripheral blood was obtained from apheresis cones provided by volunteer donors at the DHMC blood donor program.
- the cone blood was diluted at 1:4 in PBS and carefully deposited on top of 13 ml of Histopaque 1077. After 20 min centrifugation at 850 g (room temperature, no brake on deceleration), the mononuclear cells from the Histopaque / PBS interfaced were collected. After 1 wash in PBS, the PBMC were resuspended in PBS at 100x10 6 cells/ml for injection.
- INX201 alone or INX201 antibody RNA conjugate (ARC) was injected at 5 mg/kg (mixed with hPBMC). A single dose of ARC (or Ab) was used in all of the experiments described in this example.
- mice were injected with 10 million PBMCs per mouse. Plasma was collected for cytokine measurements by Luminex MILLIP LEX at early time points (4 hours to 7 days post stimulation).
- Anti-VISTA Mab payloads used for conjugations and ARCs are described in Table 11 and Appendixes 1-2. Oligo Conjugation Kit (Abeam, #ab218260) was used for all conjugations. All RNA oligos were purified by HPLC, resuspended at 100 ⁇ M and annealed. Lysine-based conjugation via the amine-reactive group, one of the most widely utilized nonspecific conjugation strategies, was used. All payloads were synthesized at IDT and contained a 3'-sense strand Cy5 label and 5'-amine on the antisense strand (see appendix 2). Drug-to-antibody ratio (DAR) was assessed by UV/Vis spectroscopy (Nanodrop) and was estimated to be 1.0 for both ARCs described.
- DAR Drug-to-antibody ratio
- K562-VISTA cells (described in ARC_01) were treated with 40-200 nM INX201-PI3K ARC or isotype control lgGl-PI3K ARC (table 1). PI3K knockdown was assessed at 24 hours by qRTPCR. INX201-PI3K ARC achieved 70% target knockdown, while INX201 Ab alone or isotype control PI3K ARC showed no PI3K knockdown ( Figure 15) indicating specificity.
- ARCs were tested in vitro using human PBMCs activated with anti-CD3/CD28 dynabeads for 72 hours as described above. Following incubation, media was collected, and cytokine levels were measured by Luminex. PI3K ARC, but not free INX201 antibody reduced pro-inflammatory cytokine production. Specifically, PI3K ARC reduced ILS and IL13 levels 3-6 fold; TNFa and IL17F levels ⁇ 2 fold, all as compared to free INX201 levels ( Figure 16A). [0300] As can be seen from Figure 16A, GLUT3 ARC, but not free INX201 Ab reduced T cell activation in a dose dependent manner.
- human PI3K and GLUT3 ARCs are demonstrated to be functional in vitro.
- human PBMCs were activated with anti-CD3/CD28 beads for 72 hours, bead to T cell ratio 1:2, and treated with (A) PI3K ARC or free INX201 (0-200 nM), or (B) GLUT3 ARC or free INX201 (0-200 nM).
- the curves were generated from inhibitor vs. response three-parameter non-linear regression analysis (GraphPad Prism 10). I L5/IL13/TNFa/IL17F cytokine levels were measured by Luminex.
- CD69 levels were measured in live CD45+/HLA-DR-/CD56-/CD3+/CD4+/CD45RA- /CD27-T effector memory cells; CD25 levels are measured in live CD45+/HLA-DR-/CD56- /CD3 cells.
- PI3K ARC treatment decreases human cytokine responses and T cell proliferation in vivo
- PI3K ARC diminished the production of IL6 and TNFa up to 3-fold, as compared to INX201 alone at 4 hours post LPS injection.
- PI3K ARC treatment also reduced delayed IFNg responses on day 7 (Figure 17B).
- PI3K ARC overall had a global impact on cytokine production, generally reducing most of them at early time point (4h) post LPS stimulation as shown in Figure 17C. Reductions in pro-inflammatory cytokines (Figure 3C) did not reach statistically significant levels, due to variability in responses.
- PI3K ARC did not show any impact on human cytokine production in groups without LPS stimulation (data not shown).
- FIG. 17 The experiments in Figure 17, clearly show that PI3K ARC reduces inflammatory cytokine responses in xeno-GvHD mice.
- Figure 17 A shows a schematic of the experiment in the xeno-GvHD LPS stimulation model.
- Figure 17B shows changes in plasma human cytokine levels on day 7 (IFNg) or 4 hours (IL6, TNFa). Statistical analysis was performed by Student's t-test.
- PI3K ARC does not reduce percent of regulatory T cells in vivo
- Treg cell levels in the blood of NSG mice in the same xeno-GvHD model we monitored Treg cell levels in the blood of NSG mice in the same xeno-GvHD model.
- NSG mice were injected i.v. with 10 million hPBMC (mixed with human INX201 alone or PI3K ARC, at 5 mg/kg).
- hPBMC mixed with human INX201 alone or PI3K ARC, at 5 mg/kg
- LPS was administered i.p.
- GLUT3 ARC treatment decreases human cytokine responses in vivo
- GLUT3 and PI3K ARCs are functional in vivo o GLUT3 treatment decreases human cytokine responses in vivo (xeno-GvHD) o PI3K ARC treatment decreases human cytokine responses in vivo o PI3K ARC treatment decreases T cell proliferation in vivo o PI3K ARC does not reduce percent of regulatory T cells in vivo
- ARCs may be used as therapeutics or prophylactics
- autoimmune diseases and inflammatory diseases e.g., those with a prominent myeloid and/or T cell component (based on where VISTA is expressed and, therefore it is where the drug will be specifically delivered).
- Potential indications include diseases where limiting activation and growth of autoreactive effector T cells might be beneficial to control/decrease disease.
- Our targeting technology can help remove on-target toxicities associated with target expression in non-immune tissues; it can help drug previous undruggable targets due to either too broad an expression profile of the target or localization of the target (transcription factors).
- rheumatoid arthritis, colitis, or systemic lupus erythematosus include autoimmune indications appropriate for ARC therapeutics.
- an siRNA or ASO according to the invention can be administered in order to act on a target such as an Immune-checkpoint inhibitor protein (e.g., PD-1, PD-L1, CTLA-4, B7-1, B7-2, LAG-3, HHLA2, TNFRSF12A, HLA-G, NECTIN2, TNFRSF25, TNFSF14, LAIR1, TNFSF15, TNFSF4, KIR2DL4, PDCD1, LGALS9, and PVR) which blocks or suppresses anti-tumor responses from being elicited in the host. Removing or inhibiting this blockade will enable the host to activate the immune system and innate antitumor immunity more efficiently.
- an Immune-checkpoint inhibitor protein e.g., PD-1, PD-L1, CTLA-4, B7-1, B7-2, LAG-3, HHLA2, TNFRSF12A, HLA-G, NECTIN2, TNFRSF25, TNFSF14, LAIR1, TNFSF15
- RNAs e.g., siRNAs and ASOs
- ARC has similar binding and internalization properties as compared to free INX201 Mab
- exemplary siRNA payloads specific to immunologically relevant targets, retain their function when conjugated to an anti-VISTA Mab and delivered into the target cells;
- siRNA conjugated to an anti-VISTA Mab INX201 facilitated siRNA accumulation and function inside K562-VISTA cells
- siRNA conjugated to an anti-VISTA Mab INX201 had function in human PBMCs and T cells, and effectively reduced proliferation and cytokine production (siRNA for five targets are described in the subject example);
- CD39 ASO conjugated to an anti-VISTA Mab INX201 functioned in human PBMCs, i.e., it provided for increased cytokine production, showing that the inventive ARCs are promising for cancer treatment;
- exemplary PI3K and GLUT3 ARCs - INX201 antibody RNA conjugates are functional in vitro, i.e., reduce T cell cytokine responses, reduce T Cell CD69/CD25 activation profile;
- exemplary GLUT3 and PI3K ARCs are functional in vivo, e.g., GLUT3 treatment decreases human cytokine responses in vivo (xeno-GvHD), PI3K ARC treatment decreases human cytokine responses in vivo, PI3K ARC treatment decreases T cell proliferation in vivo, and PI3K ARC does not reduce percent of regulatoryT cells in vivo.
- ARCs or ANCs comprising an exemplary anti-VISTA antibody and siRNA payloads which target GLUT3 or PIK3CA as therapeutics in vitro and in vivo in the GVHD model
- inventive ARCs or ANCs have much broader applicability. This can be corroborated in additional experiments such as the following:
- Table A lists sequences present in Figure 14 of the subject application and in Figure 14 of the U.S. provisional priority applications (U.S. Appl. No. 63/506,177 and 63/611,302, identified in the RELATED APPLICATIONS section on page 1 above, all of which are herein incorporated by reference in their entirety), but cannot be included in the 1143260_008613_SL.xml file submitted herewith due to the length of the sequences.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Genetics & Genomics (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Immunology (AREA)
- Molecular Biology (AREA)
- Biomedical Technology (AREA)
- Biochemistry (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- General Engineering & Computer Science (AREA)
- Biotechnology (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- Biophysics (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Microbiology (AREA)
- Physics & Mathematics (AREA)
- Plant Pathology (AREA)
- Virology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Cell Biology (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
Claims
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2024285600A AU2024285600A1 (en) | 2023-06-05 | 2024-06-05 | Anti-vista antibody rna or nucleic acid conjugates (arcs or ancs), compositions containing, and therapeutic use thereof |
| IL325028A IL325028A (en) | 2023-06-05 | 2025-11-30 | Anti-vista antibody rna or nucleic acid conjugates (arcs or ancs), compositions containing, and therapeutic use thereof |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363506177P | 2023-06-05 | 2023-06-05 | |
| US63/506,177 | 2023-06-05 | ||
| US202363611302P | 2023-12-18 | 2023-12-18 | |
| US63/611,302 | 2023-12-18 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2024254212A2 true WO2024254212A2 (en) | 2024-12-12 |
| WO2024254212A3 WO2024254212A3 (en) | 2025-02-20 |
Family
ID=93794697
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/032660 Pending WO2024254212A2 (en) | 2023-06-05 | 2024-06-05 | Anti-vista antibody rna or nucleic acid conjugates (arcs or ancs), compositions containing, and therapeutic use thereof |
Country Status (3)
| Country | Link |
|---|---|
| AU (1) | AU2024285600A1 (en) |
| IL (1) | IL325028A (en) |
| WO (1) | WO2024254212A2 (en) |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3442579A4 (en) * | 2016-04-15 | 2019-12-18 | Immunext Inc. | ANTI-HUMAN ANTI-VISTA ANTIBODIES AND USES THEREOF |
| US20240287147A1 (en) * | 2020-11-30 | 2024-08-29 | Fred Hutchinson Cancer Center | Pd-l1 binding peptides and peptide complexes and methods of use thereof |
-
2024
- 2024-06-05 WO PCT/US2024/032660 patent/WO2024254212A2/en active Pending
- 2024-06-05 AU AU2024285600A patent/AU2024285600A1/en active Pending
-
2025
- 2025-11-30 IL IL325028A patent/IL325028A/en unknown
Also Published As
| Publication number | Publication date |
|---|---|
| WO2024254212A3 (en) | 2025-02-20 |
| AU2024285600A1 (en) | 2026-01-08 |
| IL325028A (en) | 2026-01-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7583767B2 (en) | Engineered antibody FC variants for enhanced serum half-life | |
| JP7592034B2 (en) | Nucleic Acid Polypeptide Compositions and Uses Thereof | |
| JP2024050610A (en) | Nucleic Acid Polypeptide Compositions and Uses Thereof | |
| KR20220019259A (en) | UNA amidite and uses thereof | |
| WO2019136180A2 (en) | Heteroduplex nucleic acid molecules and uses thereof | |
| JP2021167353A (en) | Compositions and methods for treating autoimmune diseases and cancers | |
| KR20180095719A (en) | Chimeric proteins and methods of immunotherapy | |
| JP7774447B2 (en) | High affinity anti-CD3 antibodies and methods for producing and using same | |
| KR20220035368A (en) | Engineered PH-dependent anti-CD3 antibodies, and methods of making and using the same | |
| US20190358263A1 (en) | Compositions and Methods for Cell Therapy | |
| EP3256490A1 (en) | Engineered immunoglobulin fc polypeptides displaying improved complement activation | |
| TWI895351B (en) | Anti-CD137 antigen binding molecules for the treatment of cancer | |
| WO2020246563A1 (en) | Antibody cleavage site-binding molecule | |
| WO2022025220A1 (en) | Pharmaceutical composition including cell expressing chimeric receptor | |
| AU2024285600A1 (en) | Anti-vista antibody rna or nucleic acid conjugates (arcs or ancs), compositions containing, and therapeutic use thereof | |
| KR20250023497A (en) | Engineered FCRIIB selective IGG1 FC variants and uses thereof | |
| WO2017191504A1 (en) | Validation of therapeutic t-cells | |
| HK40090466A (en) | Pharmaceutical composition including cell expressing chimeric receptor | |
| HK40111428A (en) | Anti-cd137 antigen-binding molecule and utilization thereof | |
| CN116848141A (en) | Molecules that act specifically in tissues where cell death is observed | |
| HK40110891A (en) | Anti-cd137 antigen-binding molecule and utilization thereof | |
| HK40044036B (en) | Anti-cd137 antigen-binding molecule and utilization thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: AU2024285600 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 827852 Country of ref document: NZ |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112025026579 Country of ref document: BR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2024819971 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 827852 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020267000253 Country of ref document: KR |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2024285600 Country of ref document: AU Date of ref document: 20240605 Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24819971 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11202508210W Country of ref document: SG |
|
| WWP | Wipo information: published in national office |
Ref document number: 11202508210W Country of ref document: SG |