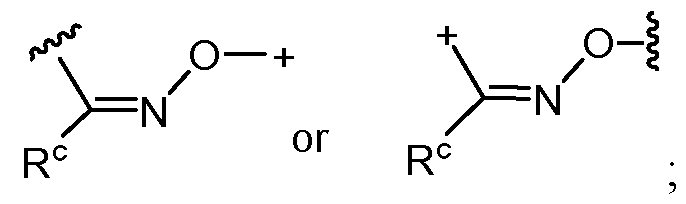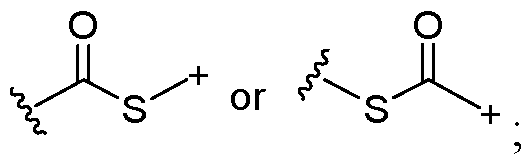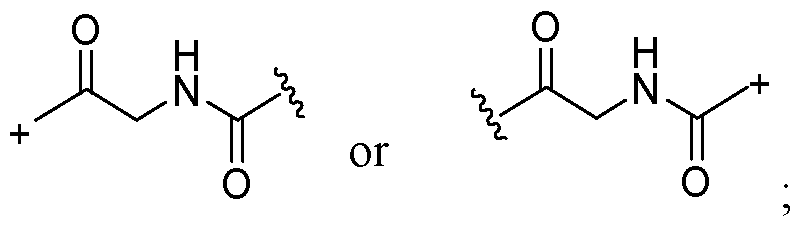WO2024241086A1 - Pegylated bovine interferon lambda and methods of use thereof - Google Patents
Pegylated bovine interferon lambda and methods of use thereof Download PDFInfo
- Publication number
- WO2024241086A1 WO2024241086A1 PCT/IB2023/061789 IB2023061789W WO2024241086A1 WO 2024241086 A1 WO2024241086 A1 WO 2024241086A1 IB 2023061789 W IB2023061789 W IB 2023061789W WO 2024241086 A1 WO2024241086 A1 WO 2024241086A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- amino acid
- protein
- boifnz
- acid sequence
- seq
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/52—Cytokines; Lymphokines; Interferons
- C07K14/555—Interferons [IFN]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/56—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule
- A61K47/59—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes
- A61K47/60—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes the organic macromolecular compound being a polyoxyalkylene oligomer, polymer or dendrimer, e.g. PEG, PPG, PEO or polyglycerol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
Definitions
- This invention relates to conjugated proteins.
- the invention relates to engineered bovine interferon lambda-3 protein conjugated to a water-soluble polymer moiety via a non-natural amino acid incorporated into the protein, and methods of use thereof.
- Infectious diseases associated with viral pathogens have a profound negative economic impact upon food animal production. In addition to negatively impacting the health and welfare of the animals, these diseases increase production costs and reduce profits for livestock producers.
- Two significant infectious diseases of beef and dairy cattle associated with viruses include bovine respiratory disease and foot and mouth disease.
- Bovine respiratory disease has been referred to as a “disease complex” since it is typically associated with both viral and bacterial pathogens that interact with one another to produce the acute clinical disease (see Taylor J.D. et al. (2010) Can. Vet. J. 51(10): 1095— 1102).
- Single or mixed viral infections that occur during periods of stress, such as weaning and commingling of animals from different sources prior to shipping to feedyards, include bovine viral diarrhea virus (BVDV), infectious bovine rhinotracheitis virus (IBR), parainfluenza-3 (PI3) and bovine respiratory syncytial virus (BRSV) (Gagea M.I. et al. (2006) J. Vet. Diagn. Invest. 18: 18— 28).
- BVDV bovine viral diarrhea virus
- IBR infectious bovine rhinotracheitis virus
- PI3 parainfluenza-3
- BRSV bovine respiratory syncytial virus
- Vaccination represents the primary method of controlling respiratory viral infections in beef and dairy cattle (Urban-Chmiel R. and Grooms, D.L. (2012) J. Livestock Sci. 3:27-36).
- Effective vaccination protocols require the use of vaccines that can elicit an antibody response in the host animal to neutralize the specific viral pathogen affecting the herd.
- viruses mutate over time, resulting in changes in their susceptibility to the antibodies generated in response to vaccines, the vaccines must be updated periodically to ensure that the antibodies are capable of neutralizing the viruses that are present in the current environment.
- the efficacy of viral vaccination can also be compromised by the timing of administration of the vaccine relative to the period(s) of stress when viral infections are most prevalent (Richeson J.T. et al.
- FMDV foot-and-mouth disease virus
- Interferons are naturally occurring cytokines that function as a first line of defense against viral infection.
- Type I IFNs IFN-a/p
- Type I IFNs bind to receptors on many different cell types, resulting in the expression of gene products that control cell death and impair viral replication.
- IFNs also activate both innate and adaptive immune cells (Lopusna K., et al. (2013) Acta. Virologica 57: 171-179).
- Type III IFNs are the most recently described IFNs (IFN- I, IFN-X2, IFN-Z.3) and have been identified in several species other than humans, including cattle and swine (Segundo 2011, Id. Sang, Y. et al. (2010) J. Interferon Cytokine Res. 30: 1-7). Type III IFNs are structurally distinct from type I IFNs ( ⁇ 5% amino acid sequence identity) but possess intrinsic antiviral activity (Donnelly R and Kotenko S. (2010) Interferon & Cytokine Res. 30:555- 564). Expression of Type III IFN receptors is limited to epithelial cells, suggesting a pivotal role in protection of mucosal surfaces (Lin J.D. et al. (2016) PLOS Pathog. 12(4):el005600).
- FMDV is highly sensitive to the action of IFNs (Chinsangaram et al. (1999) J. Virol. 73:9891-9898; Chinsangaram et al. (2001) J. Virol. 75:5498-5503; Moraes et al. (2007) J. Virol. 81 :7124-7135; U.S. Patent No. 8,906,384 B2; the entire contents of each of which are hereby incorporated by reference herein in their entirety).
- vaccination is currently being used to control rapidly spreading FMDV, the development of a protective adaptive immune response in vaccinated animals still takes 5 to 7 days.
- IFNs Interferon Cytokine Res. 23:371-380; Perez-Martin (2012) J. Virol. 86:4477-4487). It is known in the art that IFNs are generally short-lived species in circulation (see, e.g., Bansal, R. et al. (2011) J. Controlled Release 154(3):233-240). Indeed, the relatively short half-life of IFN protein in vivo has hampered efforts to induce the protective response that is needed to control FMD in cattle. [0011] Methods of modulating the biophysical properties and/or biological activity of a biotherapeutic via conjugation with another moiety are known in the art, including PEGylation.
- PEGylation involves the covalent attachment of poly(ethylene glycol) (PEG) to a biotherapeutic, and can be used to modulate a target biotherapeutic’ s water solubility, bioavailability, half-life, immunogenicity and/or biological activity.
- PEG poly(ethylene glycol)
- Methods and reagents for PEGylation have been described, for example, in U.S. Pat. Nos. 6,610,281 and 6,602,498, and “Polyethylene Glycol and Derivatives for Advanced PEGylation”, Nektar Molecular Engineering Catalog, 2003, pp. 1-17, the entire contents of each of which are hereby incorporated by reference herein in their entirety.
- PEGylated porcine IFN-alpha has been studied for the treatment of FMD in swine (Diaz-San Segundo et al. (2021) Frontiers in Microbiology, V12; Article 668890), but unfortunately, IFN- alpha does not protect cattle against FMD or other diseases.
- PEG moieties are typically covalently linked to a target biotherapeutic via its naturally-occuring amino acids, such as lysine, cysteine, histidine or the N-terminus, and/or to any carbohydrate moieties present in the biotherapeutic.
- Reactive sites that may seem suitable for PEGylation may play a significant role in receptor binding; thus, indiscriminate attachment of polymer chains such as PEG to such reactive sites on a biotherapeutic can lead to a significant reduction or even total loss of its biological activity (Clark, R. et al. (1996) J. Biol. Chem. 271 :21969-21977).
- PEG derivatives can also undergo side reactions with residues other than those targeted for modification, which can create complex, heterogeneous mixtures of PEG-derivatized biotherapeutics having reduced biological activity.
- compositions and methods for controlling BRD and FMD outbreaks Such compositions and methods would block viral replication during the lag period required for cloven-hoofed animals to develop a vaccine-stimulated adaptive immune response.
- present disclosure fulfills this need.
- conjugated proteins comprising engineered bovine interferon 3 (boIFNZ.3) protein joined to water-soluble polymer(s) through one or more non-natural amino acids, and methods of making the same. Also described are methods of treating a disease or condition in a mammal via administration of a conjugated protein of the present disclosure, or a composition comprising a conjugated protein of the present disclosure.
- BoIFNZ.3 engineered bovine interferon 3
- the present disclosure provides a conjugated protein comprising: an engineered bovine interferon 3 (boIFNZ.3) protein comprising a non-natural amino acid; and a water-soluble polymer; wherein the water-soluble polymer is conjugated to the engineered boIFNz.3 protein.
- the water-soluble polymer is conjugated to the engineered boIFNZ.3 protein via the non-natural amino acid.
- the water-soluble polymer is conjugated to the engineered boIFNz.3 protein via a covalent linkage with the non-natural amino acid.
- the water-soluble polymer is one or more water-soluble polymers
- the non-natural amino acid is one or more non-natural amino acids
- each of the one or more water-soluble polymers is conjugated to the engineered boIFNz.3 protein via each of the one or more non-natural amino acids.
- the one or more water-soluble polymers is one water-soluble polymer
- the one or more non-natural amino acids is one non-natural amino acid.
- the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24. In some embodiments, the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with: (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine; or (b) the engineered boIFNz.3 protein (i) has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the engineered boIFNz.3 protein amino acid sequence further comprises an N-terminal methionine.
- the engineered boIFNz.3 protein amino acid sequence shares at least 90% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNZ.3 protein amino acid sequence shares at least 90% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 96% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 96% identity with SEQ ID NO: 19.
- the engineered boIFNz.3 protein amino acid sequence shares at least 97% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 97% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 98% identity with SEQ ID NO:
- the engineered boIFNz.3 protein amino acid sequence shares at least 98% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO:
- the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 19.
- the non-natural amino acid is selected from the group consisting of 4-acetyl-L-phenylalanine (para-acetyl-L-phenylalanine (pAF)), 3-O-(N-acetyl-beta-D- glucosaminyl)-L-threonine, N4-(P-N-Acetyl-D-glucosaminyl)-L-asparagine, O-allyl-L-tyrosine, alpha-N-acetylgalactosamine-O-L-serine, alpha-N-acetylgalactosamine-O-L-threonine, 2- aminooctanoic acid, 2-amino-L-phenylalanine, 3-amino-L-phenylalanine, 4-amino-L- phenylalanine, 2-amino-L-tyrosine, 3-amino-L-tyrosine,
- the water-soluble polymer is selected from the group consisting of a carbohydrate, a polypeptide and a polyalkylene glycol; and derivatives thereof.
- the water-soluble polymer is a polyalkylene glycol or a derivative thereof.
- the polyalkylene glycol, or the derivative thereof is a polyethylene glycol (PEG), or a derivative thereof.
- the polyalkylene glycol or derivative thereof is a linear PEG or a derivative thereof.
- the PEG or the derivative thereof has a molecular weight of at least about 0.1 kDa and at most about 100 kDa.
- the PEG or the derivative thereof has a molecular weight of at most about 50 kDa. In some embodiments, the PEG or the derivative thereof has a molecular weight within a range of about 10 kDa to about 50 kDa, about 20 kDa to about 40 kDa, or about 25 kDa to about 35 kDa. In some embodiments, the PEG or the derivative thereof has a molecular weight of about 30 kDa. In some embodiments, prior to the conjugation with the engineered boIFNZ.3 protein, the water-soluble polymer is a PEG derivative comprising an aminooxy group.
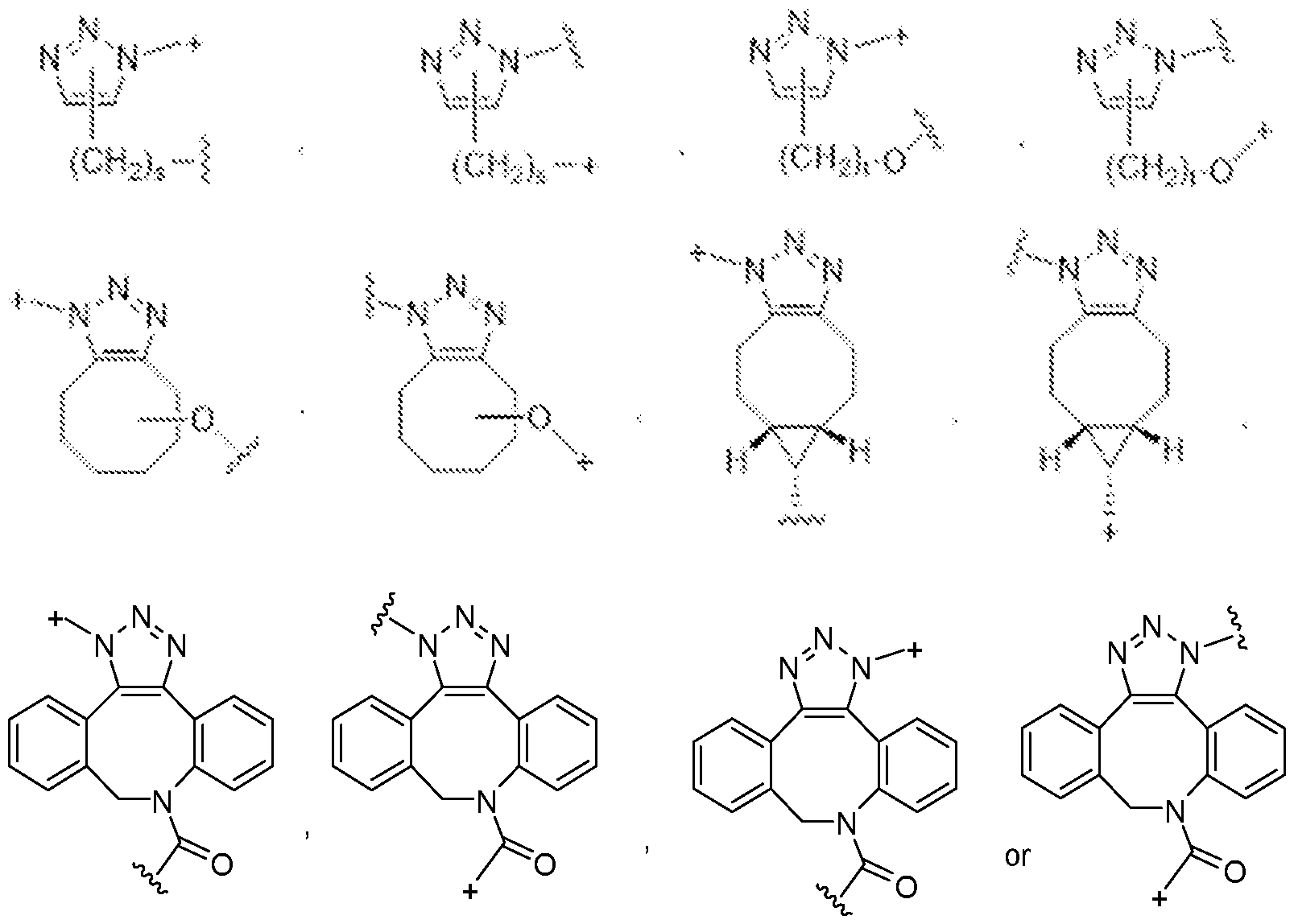
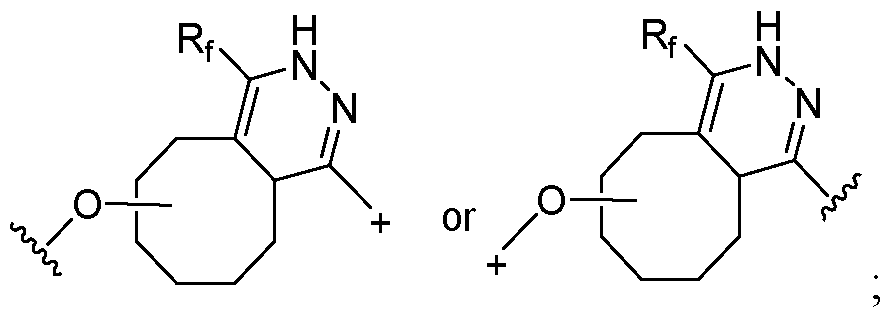
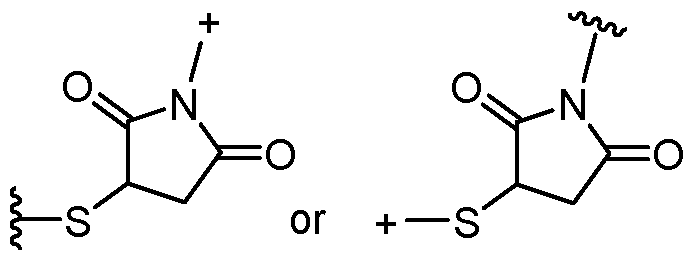
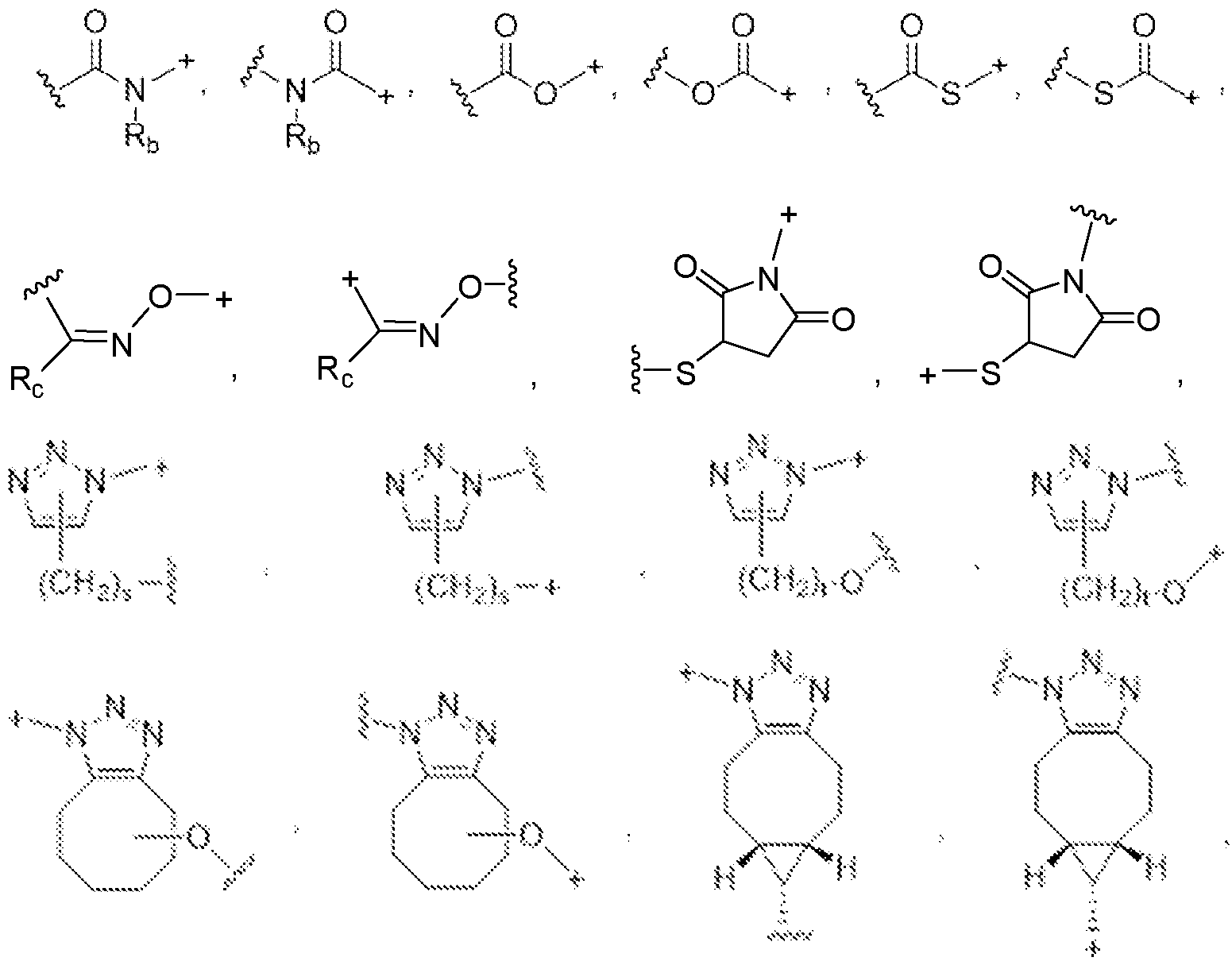
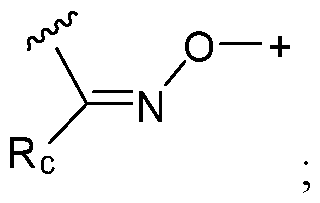
- the covalent linkage conjugating the water-soluble polymer to the non-natural amino acid comprises an amide, an ester, a thioester, a disulfide, an oxime, an imine, a pyrrolidine-2, 5-dione, a 1,2,3-triazole or a 1,4-dihydropyridazine.
- the 1,2,3-triazole is fused to an 8-membered ring.
- the 1,4-dihydropyridazine is fused to an 8-membered ring.
- the covalent linkage comprises an oxime.
- the engineered boIFNz.3 protein is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA synthetase.
- the non-natural amino acid is site- specifically incorporated into the protein.
- the conjugated boIFNz.3 protein is characterized as having a plasma elimination half-life.
- the conjugated boIFNz.3 protein plasma elimination half-life is greater than the plasma elimination half-life of the unconjugated engineered boIFNz.3 protein (e.g., the corresponding unconjugated engineered boIFNz.3 protein).
- the conjugated boIFNz.3 protein plasma elimination half-life is greater than the plasma elimination half-life of wild-type boIFNz.3 protein.
- the conjugated boIFNZ.3 protein plasma elimination half-life is greater than the plasma elimination half-life of wild-type boIFNz.3 protein having the amino acid sequence of SEQ ID NO: 1.
- the conjugated boIFNz.3 protein plasma elimination half-life is at least about 10 hours in a rat.
- the conjugated boIFNz.3 protein plasma elimination halflife is at least about 10 hours in a Sprague-Dawley rat after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein to the rat, wherein the dose is within a range of about 0.1 mg/kg to about 1.0 mg/kg body weight of the rat.
- the conjugated boIFNz.3 protein plasma elimination half-life is at least about 10 hours in a Sprague-Dawley rat after administration of a single subcutaneous dose, wherein the dose is about 0.25 mg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 40 hours in cattle. In some embodiments, the conjugated boIFNz.3 protein plasma elimination halflife is at least about 50 hours in cattle.
- the conjugated boIFNz.3 protein plasma elimination half-life is at least about 40 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is within a range of about 50 pg/kg to about 500 pg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 50 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is within a range of about 50 pg/kg to about 500 pg/kg.
- the conjugated boIFNz.3 protein plasma elimination half-life is at least about 40 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is within a range of about 75 pg/kg to about 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 50 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is within a range of about 75 pg/kg to about 150 pg/kg.
- the conjugated boIFNz.3 protein plasma elimination half-life is at least about 40 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is about 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 50 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is about 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 40 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is about 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 50 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is about 150 pg/kg.
- the conjugated boIFNz.3 protein plasma elimination half-life is at least about 60 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is within a range of about 75 pg/kg to about 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 60 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is about 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least 60 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is about 150 pg/kg.
- the present disclosure provides a pharmaceutical composition comprising a conjugated boIFNZ.3 protein of the present disclosure and a pharmaceutically acceptable excipient or carrier.
- the present disclosure provides an engineered boIFNz.3 protein; wherein the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with: (i) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; or (ii) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N- terminal methionine; and wherein each said amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the engineered boIFNz.3 protein comprises an amino acid sequence that shares at least 90% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, but which shares less than 100% identity with SEQ ID NO: 1; optionally wherein the engineered boIFNZ.3 protein amino acid sequence further comprises an N-terminal methionine.
- the engineered boIFNz.3 protein comprises a non-natural amino acid.
- the amino acid sequence shares at least 90% identity with SEQ ID NO: 5.
- the amino acid sequence shares at least 90% identity with SEQ ID NO: 5 further comprising the N-terminal methionine.
- the amino acid sequence shares at least 95% identity with SEQ ID NO: 5.
- the amino acid sequence shares at least 95% identity with SEQ ID NO: 5 further comprising the N-terminal methionine. In some embodiments, the amino acid sequence shares at least 96% identity with SEQ ID NO: 5. In some embodiments, the amino acid sequence shares at least 96% identity with SEQ ID NO: 5 further comprising the N-terminal methionine. In some embodiments, the amino acid sequence shares at least 97% identity with SEQ ID NO: 5. In some embodiments, the amino acid sequence shares at least 97% identity with SEQ ID NO: 5 further comprising the N-terminal methionine. In some embodiments, the amino acid sequence shares at least 98% identity with SEQ ID NO: 5.
- the amino acid sequence shares at least 98% identity with SEQ ID NO: 5 further comprising the N-terminal methionine. In some embodiments, the amino acid sequence shares at least 99% identity with SEQ ID NO: 5. In some embodiments, the amino acid sequence shares at least 99% identity with SEQ ID NO: 5 further comprising the N-terminal methionine.
- the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 5. In some embodiments, the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 5 further comprising an N-terminal methionine. In some embodiments, the engineered boIFNZ.3 protein does not contain a non-natural amino acid.
- the engineered boIFNz.3 protein can be optionally defined as comprising an amino acid that shares the given percentage identity (e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99% identity with SEQ ID NO: 5), optionally wherein the engineered boIFNZ.3 protein amino acid sequence further comprises an N- terminal methionine.
- the present disclosure provides an engineered boIFNz.3 protein comprising a non-natural amino acid; wherein the engineered boIFNZ.3 protein has an amino acid sequence that shares at least 90% identity with: (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with: (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24, or (ii) has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the engineered boIFNz.3 protein amino acid sequence further comprises an N-terminal methionine.
- the engineered boIFNz.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 18.
- the engineered boIFNz.3 protein amino acid sequence shares at least 96% identity with SEQ ID NO: 18.
- the engineered boIFNz.3 protein amino acid sequence shares at least 97% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 98% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 18. In some other embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 96% identity with SEQ ID NO: 19.
- the engineered boIFNz.3 protein amino acid sequence shares at least 97% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 98% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least
- the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 19.
- the non-natural amino acid is para-acetyl-L-phenylalanine.
- the engineered boIFNz.3 protein is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA, and the non-natural amino acid is site- specifically incorporated into the protein.
- the present disclosure provides a pharmaceutical composition
- a pharmaceutical composition comprising an engineered boIFNZ.3 protein of the present disclosure and a pharmaceutically acceptable excipient or carrier.
- the present disclosure provides a nucleic acid having the nucleotide sequence of SEQ ID NO: 27.
- the present disclosure provides a nucleic acid molecule having a nucleotide sequence encoding any of the engineered boIFNz.3 proteins referred to herein (e.g. a nucleic acid molecule encoding any one of SEQ ID NOS: 2 to 24).
- the present disclosure provides vector comprising a nucleic acid molecule encoding any one of SEQ ID NOS: 2 to 24.
- the vector comprises a nucleic acid molecule encoding SEQ ID NO: 4.
- the vector comprises a nucleic acid molecule encoding SEQ ID NO: 5.
- the vector comprises a nucleic acid molecule encoding SEQ ID NO: 6.
- the vector comprises a nucleic acid molecule encoding SEQ ID NO: 7.
- the vector comprises a nucleic acid molecule encoding SEQ ID NO: 18.
- the vector comprises a nucleic acid molecule encoding SEQ ID NO: 19.
- the present disclosure provides a method of treating a disease or condition in a mammal.
- the method is a prophylactic method.
- the method comprises administering to the mammal a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a conjugated boIFNz.3 protein of the present disclosure.
- the mammal is a ruminant mammal.
- the ruminant mammal is bovine.
- the ruminant mammal is a cattle.
- the ruminant mammal is a calf.
- the method of treating a disease or condition in a mammal provides the mammal with protection against the disease or condition.
- the method induces systemic antiviral activity in the mammal.
- the method induces systemic antiviral activity in the mammal, such as systemic antiviral activity against a virus that is the cause of or is associated with the disease or condition.
- the method provides protection against infection by a pathogen, such as a virus, that is the cause of or is associated with the disease or condition.
- the method provides protection against the disease or condition.
- the method provides protection against symptoms of the disease or condition.
- the method reduces a rate of infection by the causative or associated virus. In some embodiments, the method reduces a degree of infection by the causative or associated virus. In some embodiments, the method inhibits replication of the causative or associated virus. In some embodiments, the method delays onset of the disease or condition. In some embodiments, the method reduces severity of the disease or condition. In some embodiments, the method reduces severity of symptoms of the disease or condition. In some embodiments, the method prevents the disease or condition from occurring. In some embodiments, the method prevents infection by the causative or associated virus. In some embodiments, the method reduces the rate of transmission of the causative or associated virus from one ruminant mammal to another ruminant mammal or mammals.
- the method prevents the transmission of the causative or associated virus from one ruminant mammal to another ruminant mammal or mammals.
- the method further comprises administering a vaccine to the ruminant mammal, wherein the vaccine is capable of preventing the disease or condition.
- the method induces (i) anti-viral activity against the causative or associated virus; (ii) adjuvanted adaptive immune responses against the causative or associated virus; and/or (iii) expression of interferon-stimulated genes correlated with systemic control of viral replication in a mammal susceptible to infection by the causative or associated virus in vivo; thereby delaying, reducing severity and/or preventing the disease or condition.
- the disease or condition being treated is a bovine respiratory disease.
- the disease or condition is associated with bovine viral diarrhea virus (BVDV), infectious bovine rhinotracheitis virus (IBR), parainfluenza-3 (PI3) or bovine respiratory syncytial virus (BRSV); or a combination thereof.
- BVDV bovine viral diarrhea virus
- IBR infectious bovine rhinotracheitis virus
- PI3 parainfluenza-3
- BRSV bovine respiratory syncytial virus
- the disease or condition is associated with BVDV, PI3 or BRSV; or a combination thereof.
- the method of treating a disease or condition in a mammal further comprises administering to the mammal a vaccine that is capable of preventing infection by BVDV, IBR, PI3 or BRSV.
- the disease or condition being treated is foot and mouth disease.
- the disease or condition is associated with foot and mouth disease virus (FMDV).
- the method of treating a disease or condition in a mammal further comprises administering to the mammal a vaccine that is capable of preventing infection by FMDV.
- the method of treating the disease or condition comprises administering a conjugated boIFNZ.3 protein of the present disclosure to a ruminant mammal, wherein the dose is within a range of about 50 pg/kg to about 500 pg/kg of the body weight of the mammal.
- the dose is within a range of about 75 pg/kg to about 150 pg/kg of the body weight of the mammal. In some embodiments, the dose is within a range of about 100 pg/kg to about 200 pg/kg of the body weight of the mammal.
- the ruminant mammal is bovine. In some embodiments, the ruminant mammal is a cattle. In some embodiments, the ruminant mammal is a calf.
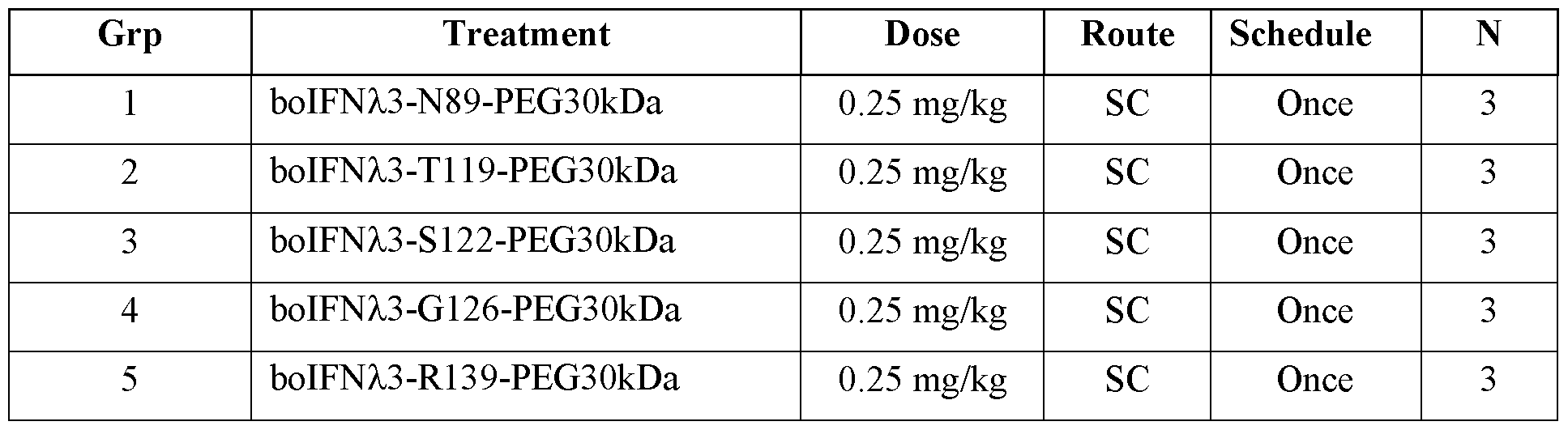
- FIG. 1 shows E. coli plasmid map (A) pKGOlOO for expression of wild type boIFNZ.3; and (B) pKG0168 for expression of engineered boIFNZ.3 containing a non-naturally encoded amino acid.
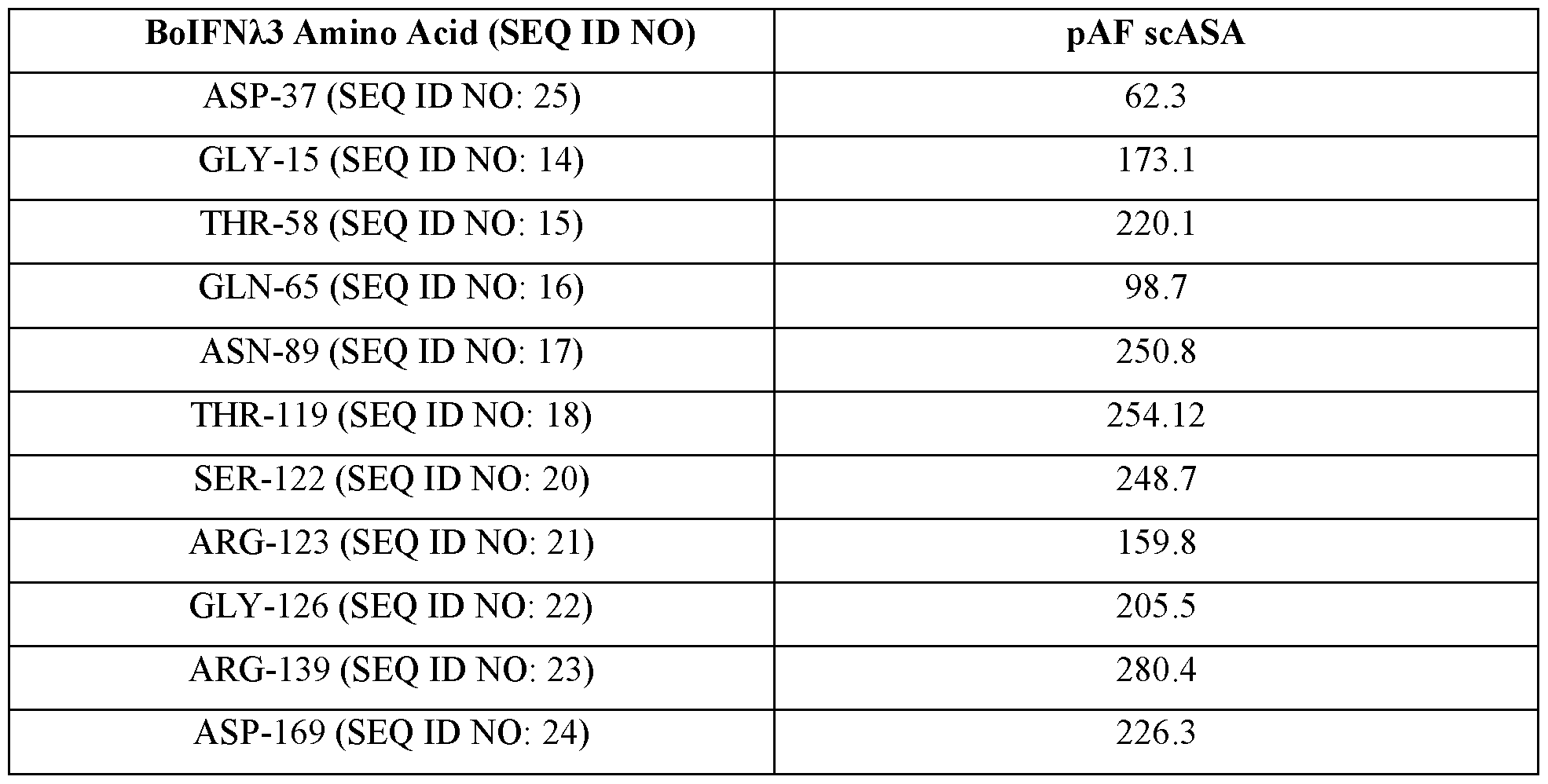
- FIG. 2 shows a boIFNZ.3 3 -dimensional model with amino acids selected for non-natural amino acid incorporation.
- FIG. 3 shows real-time quantitative polymerase chain reaction (RT-qPCR) of ISG15 after treatment with boIFNk3-PEG variants in MDBK cells.
- FIG. 4 shows MDBK ISRE-luciferase reporter assay after treatment with PEGylated boIFN/3 (boIFN/3-PEG) variants for 7 hours.
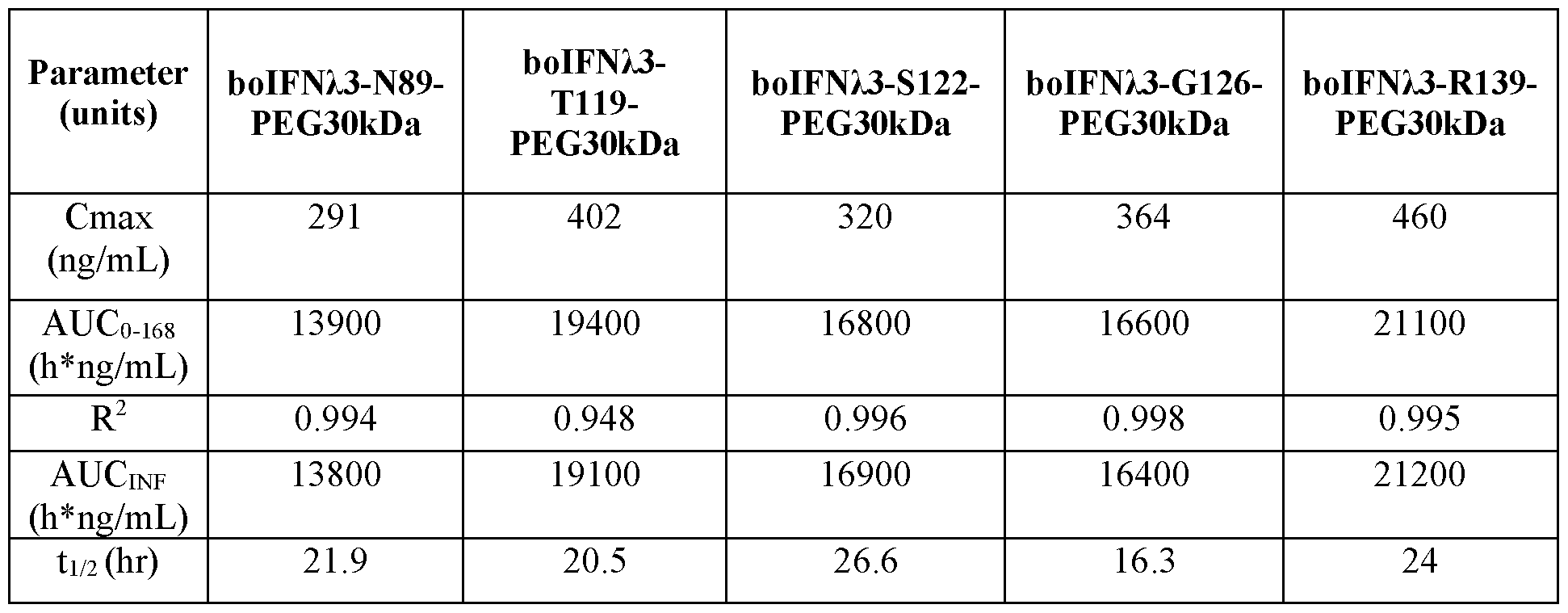
- FIG. 5 shows mean plasma concentration versus time profiles for boIFN/3-PEG variants in male Sprague-Dawley rats.
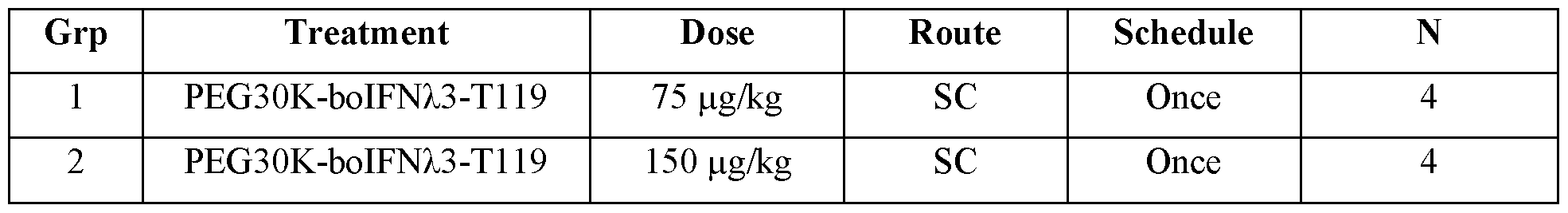
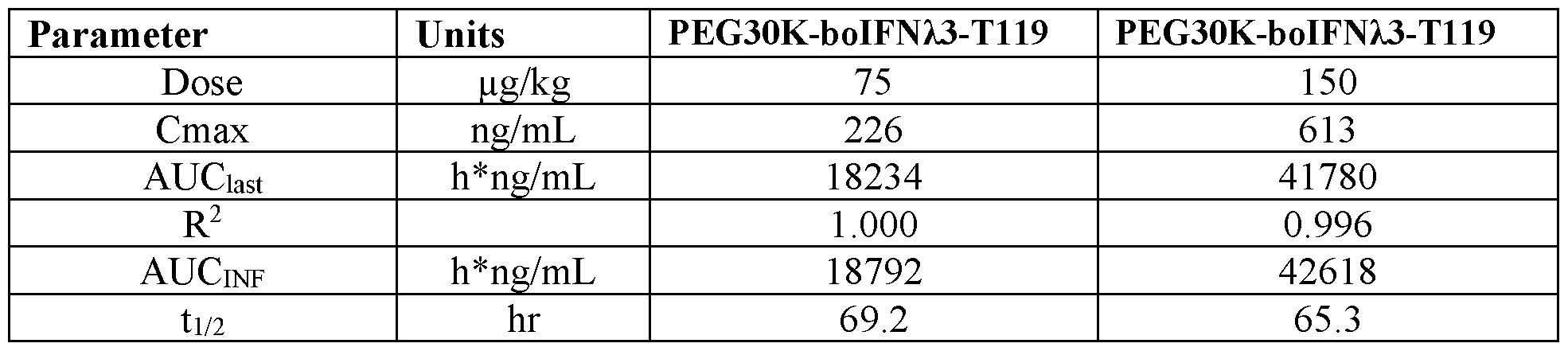
- FIG. 6 shows mean plasma concentration versus time profiles for boIFN/3-T I 19- PEG30kDa in Holstein-Friesian Calves.
- FIG. 7 shows shows thermal melting temperatures of (A) boIFNk3-pAF variants and (B) boIFNX3-pAF-PEG variants.
- the present disclosure provides a conjugated bovine interferon lambda-3 (boIFNX3) protein, which is sometimes referred to herein simply as a “conjugated protein.”
- the conjugated protein comprises an engineered boIFNz.3 protein conjugated to a moiety that is capable of extending boIFNz.3 protein elimination half-life, such as a water-soluble polymer.
- a water-soluble polymer is conjugated to the engineered boIFNz.3 protein via a nonnatural amino acid in the protein.
- the water-soluble polymer is a polyalkylene glycol, such as a polyethylene glycol (PEG) or PEG derivative.
- compositions containing the conjugated boIFNz.3 protein can be used in ruminant mammals such as cattle as a method of treating (e.g., protecting the mammals from) a disease or condition associated with or caused by a virus.
- the disease or condition is foot and mouth disease, or any disease or condition associated with foot and mouth disease virus.
- the disease or condition is a bovine respiratory disease, or any disease or condition associated with bovine viral diarrhea virus, infectious bovine rhinotracheitis virus, parainfluenza- 3 and/or bovine respiratory syncytial virus.
- the method of treatment can further include inoculation of the ruminant mammal with a vaccine that is suitable for the treatment of the virus that causes, or is associated, with the disease or condition.
- administration of a conjugated boIFNz.3 protein of the present disclosure, or a composition containing a conjugated boIFNZ.3 protein of the present disclosure can reduce the incidence and/or severity of, and/or prevent the disease or condition.
- administration of a conjugated boIFNz.3 protein of the present disclosure, or a composition containing a conjugated boIFNz.3 protein of the present disclosure can reduce the incidence and/or severity of, and/or prevent the disease or condition.
- administration of a conjugated boIFNz.3 protein of the present disclosure, or a composition containing a conjugated boIFNz.3 protein of the present disclosure, with further administration of a vaccine can reduce the incidence and/or severity of, and/or prevent the disease or condition.
- the present disclosure also provides engineered boIFNz.3 proteins and their amino acid sequences, including engineered boIFNz.3 proteins suitable for conjugation to moieties that are capable of extending boIFNz.3 protein elimination half-life.
- engineered boIFNZ.3 proteins and their amino acid sequences wherein the engineered boIFNz.3 proteins contain one or more non-natural amino acids suitable for conjugation to moieties that are capable of extending boIFNz.3 protein elimination half-life.
- the moieties that are capable of extending boIFNz.3 protein elimination half-life are water-soluble polymers.
- the engineered boIFNz.3 proteins of the present disclosure can be obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA synthetase, which allow for site-specific incorporation of the nonnatural amino acid into boIFNz.3 protein.
- the one or more non-natural amino acids present in engineered boIFNz.3 proteins or conjugates containing them can be one or more non-naturally encoded amino acids.
- Novel nucleic acid sequences encoding the engineered boIFNz.3 proteins are also provided.
- adaptive immunity refers to a type of immunity that develops when a subject's immune system responds to a foreign substance or microorganism, such as after an infection or vaccination.
- the function of an adaptive immune response is to destroy invading pathogens and any toxic molecules they produce.
- biotherapeutic refers to a therapeutic agent produced using biological means.
- the biological means include recombinant DNA technology.
- Biotherapeutics of the present disclosure include biologically active proteins, such as engineered boIFNz.3 proteins, and conjugated boIFNz.3 proteins and variants thereof. Biotherapeutics also include vaccines.
- BTD bovine respiratory disease
- BRSV bovine respiratory syncytial virus
- BVDB bovine viral diarrhea virus
- IBR infectious bovine rhinotracheitis virus
- PI-3 parainfluenza-3 virus
- canonical amino acid refers to any one of the 20 common amino acids that are genetically encoded in humans (i.e., alanine (“A” or “Ala”), arginine (“R” or “Arg”), asparagine (“N” or “Asn”), aspartic acid (“D” or Asp”), cysteine (“C” or “Cys”), glutamic acid (“E” or “Glu”), glutamine (“Q” or “Gin”), glycine (“G” or “Gly”), histidine (“H” or “His”), isoleucine (“I” or “He”), leucine (“L” or “Leu”), lysine (“K” or “Lys”), methionine (“M” or “Met”), phenylalanine (“F” or “Phe”), proline (“P” or “Pro”), serine (“S” or “Ser”), threonine (“T” or “Thr”), try
- cloven-hoofed animals refers to members of the mammalian order Artiodactyla.
- Non-limiting examples of cloven-hoofed animals include cattle, deer, pigs, antelopes, gazelles, goats and sheep.
- conjugated protein refers to a protein to which another chemical group or molecule has been attached.
- the chemical group or molecule is a moiety that is capable of extending protein elimination half-life, such as a half-life extender moiety.
- the elimination half-life of the conjugated protein can be greater than the elimination half-life of the corresponding or similar protein in its unconjugated form.
- engineered protein refers to a protein created and/or generated by design.
- the engineered protein can be a protein “variant” as disclosed herein.
- the engineered protein can be a deletion mutant.
- the engineered protein can be a protein or variant containing a non-natural amino acid, or more particularly, a non-naturally encoded amino acid, as disclosed herein.
- Methods of engineering a protein to contain a non-natural amino acid, such as a non- naturally encoded amino acid are disclosed herein.
- An engineered protein of the disclosure can be a product of such a method.
- foot and mouth disease encompasses disease symptoms in swine, cattle, sheep, and goats caused by a foot and mouth disease virus (FMDV) infection.
- FMDV foot and mouth disease virus
- examples of such symptoms include, but are not limited to, fever, lameness and vesicular lesions on the feet, tongue, snout and teats.
- the term “foot and mouth disease virus” or “FMDV” as used herein refers to a highly contagious pathogenic member of the Picornaviridae family that causes vesicular lesions in cattle, swine and other cloven-hoofed animals.
- the term “half-life” as used herein refers to the time required for any specified property to decrease by half. Typically, the specified property is the concentration of a substance in the body, or a compartment thereof, wherein the substance is a conjugated protein of the present disclosure, or the corresponding protein or a comparable protein in its unconjugated form.
- the term “halflife” can be referred to herein as “ti/2” or “T1/2”. In some embodiments, a half-life refers to a “terminal half-life” as further disclosed herein.
- half-life extender refers to molecules (or moieties, e.g., monovalent or bivalent moieties) that can be used to increase the size of a biotherapeutic.
- the half-life extender can be used to increase the hydrodynamic volume of a biotherapeutic.
- a half-life extender can thus be used to increase the half-life of the biotherapeutic to which it is attached.
- a half-life extender can be a water-soluble polymer.
- a biotherapeutic include conjugation with a water-soluble polymer (e.g., PEGylation), introduction of glycosylation site(s), and fusion to plasma protein(s) (e.g., serum albumin; see, e.g., Kontermann R.E. (2016) Expert Opin. Biol. Therap. 16(7):903-915, the entire contents of which are hereby incorporated by reference herein in their entirety).
- a water-soluble polymer e.g., PEGylation
- glycosylation site(s) e.g., serum albumin; see, e.g., Kontermann R.E. (2016) Expert Opin. Biol. Therap. 16(7):903-915, the entire contents of which are hereby incorporated by reference herein in their entirety.
- plasma protein(s) e.g., serum albumin; see, e.g., Kontermann R.E. (2016) Expert Opin. Biol. Therap. 16(7):903-915, the entire contents of
- a half-life extender molecule or moiety of the present disclosure is a molecule or moiety that is capable of extending the half-life of a protein of the present disclosure, such as extending the halflife of a boIFNZ.3 protein or variant thereof.
- nucleic acids or polypeptide sequences refer to two or more sequences or subsequences that are the same. Identity can be expressed as a percentage. For example, two sequences that are exactly the same share 100% identity.
- Sequences are "substantially identical” if they have a percentage of nucleotides, or amino acid residues, that are the same (i.e., about 60% identity, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, or about 95% identity) over a specified region, when the nucleotides, or amino acid residues, are compared and aligned for maximum correspondence over a comparison window, or designated region as measured, e.g., using sequence comparison algorithms or other algorithms available to persons of ordinary skill in the art. For example, sequence alignment programs such as BLAST can be used to align two or more sequences, or sequences can be aligned by manual alignment and visual inspection. This definition also refers to the complement of a test sequence.
- the identity can exist over a region that is at least about 50 amino acids or nucleotides in length, or over a region that is 75-100 amino acids or nucleotides in length, or, where not specified, across the entire sequence of a polynucleotide or polypeptide, e.g., across the full sequence that is specified.
- isolated when applied to a nucleic acid or protein, denotes that the nucleic acid or protein is free of at least some of the cellular components with which it is associated in the natural state, or that the nucleic acid or protein has been concentrated to a level greater than the concentration of its in vivo or in vitro production. It can be in a homogeneous state. Isolated substances can be in either a dry or semi-dry state, or in solution, including but not limited to, an aqueous solution. It can be a component of a pharmaceutical composition that comprises additional pharmaceutically acceptable carriers and/or excipients.
- Purity and homogeneity are typically determined using analytical chemistry techniques such as polyacrylamide gel electrophoresis or high-performance liquid chromatography.
- a protein which is the predominant species present in a preparation is substantially purified.
- an isolated gene is separated from open reading frames which flank the gene and encode a protein other than the gene of interest.
- the term "purified” denotes that a nucleic acid or protein gives rise to substantially one band in an electrophoretic gel. Particularly, it may mean that the nucleic acid or protein is at least 75% pure, at least 80% pure, at least 85% pure, at least 90% pure, at least 95% pure, at least 99% or greater pure.
- moiety that is capable of extending protein elimination half-life refers to a moiety that, when conjugated to a protein, can increase the protein elimination half-life.
- the elimination half-life is a blood, plasma, serum, or whole body elimination half-life.
- a moiety that is capable of extending protein elimination half-life includes a half-life extender moiety, which include a water-soluble polymer such as PEG.
- HESylation refers to coupling a biotherapeutic with hydroxy alkyl starch, or more particularly, hydroxy ethyl starch (HES), or a derivative thereof.
- the term “elimination half-life” as used herein refers to a pharmacokinetic parameter that is defined as the period of time that it takes for the concentration of a biotherapeutic in the plasma or serum of a subject, or the total amount in the whole body of a subject, to be reduced by about 50%.
- concentration of the biotherapeutic in the plasma or serum, or in the whole body, of the subject will be half of the starting concentration.
- the period of time that it takes for the concentration of the biotherapeutic to be reduced by about 50% commences at or about the time of administration of the biotherapeutic to the subject.
- immune response refers to the production of molecules such as cytokines (e.g., interferons, chemokines, etc., and/or antibodies and/or cells (such as T lymphocytes)) that are directed against, or assist in the decomposition or inhibition of, a particular infectious agent, such as a virus or an antigenic epitope or particular antigenic epitopes.
- cytokines e.g., interferons, chemokines, etc.
- antibodies and/or cells such as T lymphocytes
- an effective immunoprotective response refers to an immune response that is directed against an infectious agent such as a virus as a whole and/or one or more antigenic epitopes of a pathogen so as to protect against infection by the pathogen in a treated animal.
- An effective immunoprotective response can be induced in animals that have not previously been infected with the pathogen and/or are not infected with the pathogen, e.g., at the time of treatment with a biotherapeutic of the present disclosure and/or a vaccine.
- An effective immunoprotective response can also be induced in an animal already infected with the pathogen, e.g., at the time of treatment with a biotherapeutic of the present disclosure and/or a vaccine.
- modulated serum half-life and “modulated plasma half-life” refer to positive or negative changes in the circulating half-life of a conjugated protein molecule in serum or plasma, respectively, relative to its unconjugated form.
- modulated therapeutic half-life refers to a positive or negative change in the half-life of the therapeutically effective amount of a conjugated protein relative to its unconjugated form.
- non-natural amino acid refers to non-proteinogenic amino acids, meaning that they are not among the 20 amino acids attached to tRNAs in living human cells used to polymerize proteins. Some non-natural amino acids do occur in nature, e.g., in some non-human species, but most are chemically synthesized.
- citrulline is a non- proteinogenic amino acid produced in vivo through the oxidation of arginine, while p-benzoyl- phenylalanine is an example of a non-natural amino acid that is not found in nature at all.
- Nonlimiting examples of non-natural amino acids are disclosed herein. Some non-natural amino acids are derived from the 20 canonical amino acids, e.g., via chemical modifications. Non-natural amino acids of the present disclosure include non-naturally encoded amino acids. Non-natural amino acid-containing polypeptides presented herein may include isotopically-labelled compounds with one or more atoms replaced by an atom having an atomic mass or mass number different from the atomic mass or mass number usually found in nature. Examples of isotopes that can be incorporated into the present compounds include isotopes of hydrogen, carbon, nitrogen, oxygen, sulfur, fluorine and chlorine, such as 2 H, 3 H, 13 C, 14 C, 15 N, 17 O, 18 O, 35 S, 18 F, 36 C1, respectively.
- isotopically-labelled compounds described herein for example those into which radioactive isotopes such as 3 H and 14 C are incorporated, may be useful in drug and/or substrate tissue distribution assays. Further, substitution with isotopes such as deuterium, i.e., 2 H, can afford certain therapeutic advantages resulting from greater metabolic stability, for example, increased in vivo half-life or reduced dosage requirements.
- non-naturally encoded amino acid refers to a non-natural amino acid that has been incorporated into an amino acid sequence via a change in the genetic code of a system (e.g., an organism) that encodes the amino acid sequence.
- a system e.g., an organism
- Methods of incorporating a nonnatural amino acid into an amino acid sequence via a change in the genetic code are disclosed herein.
- nucleic acid refers to deoxyribonucleotides, deoxyribonucleosides, ribonucleosides, or ribonucleotides and polymers thereof in either single- or double-stranded form. Unless specifically limited, the term encompasses nucleic acids containing known analogues of natural nucleotides which have similar binding properties as the reference nucleic acid and are metabolized in a manner similar to naturally occurring nucleotides.
- oligonucleotide analogs including peptidonucleic acid (PNA), analogs of DNA used in antisense technology (phosphorothioates, phosphoroamidates, and the like).
- PNA peptidonucleic acid
- a nucleic acid having a particular nucleotide sequence also implicitly encompasses conservatively modified variants thereof (including but not limited to, degenerate codon substitutions) and complementary sequences as well as the sequence explicitly indicated.
- degenerate codon substitutions may be achieved by generating sequences in which the third position of one or more selected (or all) codons is substituted with mixed-base and/or deoxyinosine residues (Batzer et al., Nucleic Acid Res. 19:5081 (1991); Ohtsuka et al., J. Biol. Chem. 260:2605-2608 (1985); Rossolini et al., Mol. Cell. Probes 8:91-98 (1994)).
- a nucleic acid encoding a polypeptide of the present invention may be obtained by a process comprising the steps of screening a library under stringent hybridization conditions with a labeled probe having a nucleotide sequence of the invention, or a fragment thereof, and isolating full-length cDNA and genomic clones containing said nucleotide sequence.
- Such hybridization techniques are well known to the skilled artisan.
- PEGylation refers to coupling a biotherapeutic with polyethylene glycol (PEG), or a derivative thereof.
- PEGylated refers to a PEG-biotherapeutic conjugate, wherein the biotherapeutic is covalently linked to at least one PEG moiety.
- polyalkylene glycol poly(alkene glycol),” “polyoxyalkylene glycol” and “polyglycol” are used interchangeably herein, and as used herein refers to linear, branched and multiarm polymeric polyether polyols.
- Non-limiting examples of polyalkylene glycols of the present disclosure include polyethylene glycol (PEG), polypropylene glycol, polybutylene glycol, and derivatives thereof.
- polypeptide and protein as used herein have their ordinary meaning in the art and are used interchangeably herein unless expressly indicated otherwise.
- prevent has its ordinary meaning in the art, that is, to “prevent” means to keep something (e.g., a disease or condition, a viral infection, or both) from happening or arising.
- prophylaxis or “prophylactic treatment” as used herein includes preventing a disease or condition in a mammal from occurring and/or preventing a disease or condition from getting worse.
- prophylaxis refers to preventing a disease or condition from occurring, e.g., by administering a biotherapeutic of the present disclosure to a mammal prior to the onset of the disease or condition, and/or prior to the onset of symptoms of the disease or condition.
- prophylaxis refers to protection against infection by a pathogen in a mammal.
- the term “protection against infection” as used herein includes not only the absolute prevention of infection by a pathogen, but also any detectable reduction in a degree or rate of infection by a pathogen, or any detectable reduction in severity of a disease or any symptom or condition resulting from infection by the pathogen in an animal, such as a vaccinated animal, as compared to an infected animal, such as an unvaccinated infected animal, e.g., using methods known to a person of ordinary skill in the art and/or via assessment by a veterinarian or physician.
- a method of treating a disease or condition can include providing protection against the disease or condition.
- refolding refers to any process, reaction or method which transforms disulfide bond containing polypeptides from an improperly folded or unfolded state to a native or properly folded conformation with respect to disulfide bonds.
- ruminant mammal refers to members of the mammalian order Artiodactyla.
- Non-limiting examples of ruminant mammals are cattle, all domesticated and wild bovines, deer, giraffes, antelopes, gazelles, goats and sheep.
- the ruminant mammal is bovine.
- Bovine as used herein refers to a diverse group of hoofed animals (ungulates), including cattle, bison, African buffalo, water buffalo and some antelope.
- bovine can refer to an individual animal (singular form), or to a group of animals (plural form).
- the ruminant mammal is a cattle.
- “cattle” refers to an individual animal (singlular form) or a group of animals (plural form).
- a cattle is a member of the genus Bos, which includes domestic cattle (Bos Taurus).
- the cattle may be, e.g., beef cattle (raised for meat production) or dairy cattle (used for milk production).
- the ruminant mammal is a calf.
- a calf is less than 12 months old.
- a calf is less than 6 months old.
- a calf is between 4 and 6 months old.
- subject refers to an animal who is the object of treatment, observation or experiment.
- animal is a ruminant mammal, e.g., as defined herein.
- terminal half-life refers to the time required to divide the concentration of an agent (e.g., a conjugated protein of the present disclosure) in a body or body compartment (e.g., plasma or serum) by two after reaching pseudo-equilibrium, and not the time required to eliminate half the administered dose (see, e.g., Toutain P.L. and Bousquet-Melou A., J. Vetinary Pharmacology and Therapeutics (2004) 27(6):427-439).
- clearance e.g., plasma clearance
- the terminal half-life does not reflect the elimination process; rather, the terminal half-life reflects the rate and extent of absorption. Terminal half-life is particularly relevant in the case of multiple dosing regimens.
- therapeutically effective amount refers to an amount sufficient to treat, prevent or at least partially arrest symptoms, or at least partially protect against the development of symptoms, of a disease, disorder or condition, or an amount sufficient to induce a desired response in a subject, including but not limited to inducing an immune response and/or protection from an infectious disease.
- a therapeutically effective amount will depend on the severity and course of the disease, disorder or condition, previous therapy, the subject's health status and response to the drugs, and the judgment of the treating physician or veterinarian. It is considered well within the skill of a person of ordinary skill in the art to determine such therapeutically effective amounts by routine experimentation e.g., a dose escalation trial).
- treating refers to prophylaxic treatment and can also include or refer to therapeutic treatment, unless expressly indicated otherwise.
- the term “unusual genetically encoded amino acid that is not present in humans” as used herein refers to amino acids that are used in the biosynthesis of proteins in non-humans. Nonlimiting examples include selenocysteine and pyrrolysine, the latter of which is produced in some methanogenic archaea and bacteria.
- variant refers to a protein derived from the corresponding native (also referred to herein as “wild type”) protein by deletion (also referred to herein as “truncation”) or addition of one or more amino acids to the N-terminus and/or C-terminus of the native protein; deletion or addition of one or more amino acids at one or more sites in the native protein; and/or substitution of one or more amino acids at one or more sites in the native protein.
- variants may result from, for example, protein engineering.
- a protein variant of the present disclosure can be an engineered protein. Protein variants encompassed by the present disclosure are biologically active, that is, they possess the desired IFNz.3 biological activity, as described herein.
- Non-limiting examples of the desired IFNZ.3 biological activity of an IFNZ.3 protein variant include the ability of the IFNZ.3 protein variant to induce mRNA expression of ISG15, and/or the ability of the IFNZ.3 protein variant to inhibit viral replication.
- the desired IFNZ.3 biological activity is the ability to induce mRNA expression of ISG15. Methods of determining induction of mRNA expression of ISG15 are disclosed herein.
- a IFNZ.3 protein variant is considered to have the desired IFNZ.3 activity if it induces mRNA expression of ISG15 at a level that is at least about 50%, about 60%, about 70%, about 80%, about 90% or about 95% of the level of mRNA expression of ISG15 that is induced by the IFNZ.3 molecule of SEQ ID NO: 1, e.g., if it induces mRNA expression of ISG15 in MDBK cells at a level that is at least about 50%, about 60%, about 70%, about 80%, about 90% or about 95% of the level of mRNA expression of ISG15 that is induced by the IFNz.3 molecule of SEQ ID NO: 1 after treatment of the MDBK cells with the IFNZ.3 protein variant at a concentration of 100 ng/ml, and wherein mRNA expression is determined 6 hours after the treatment.
- the desired IFNZ.3 biological activity is the ability to inhibit viral replication. In some embodiments, the desired IFNZ.3 biological activity is the ability to inhibit the replication of bovine viral diarrhea virus (BVDV). Methods of determining the ability to inhibit the replication of BVDV are disclosed herein.
- BVDV bovine viral diarrhea virus
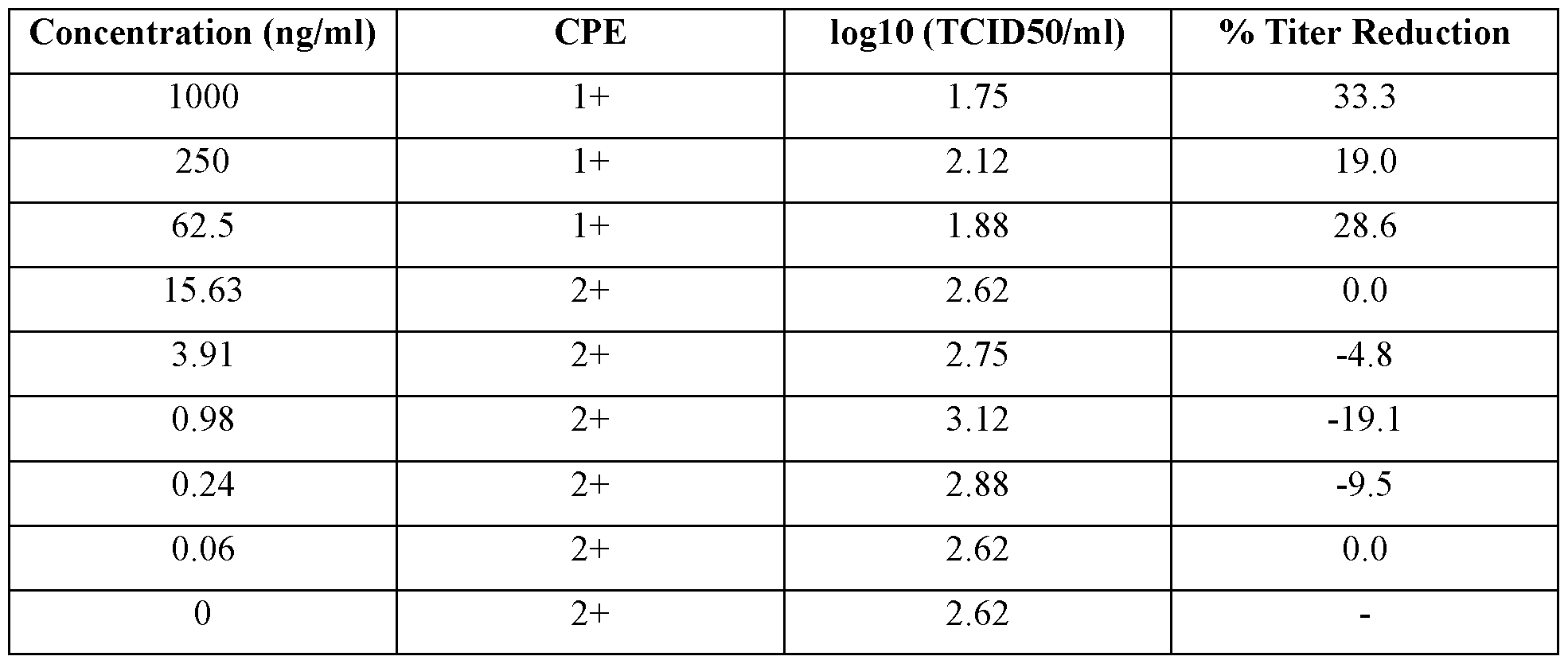
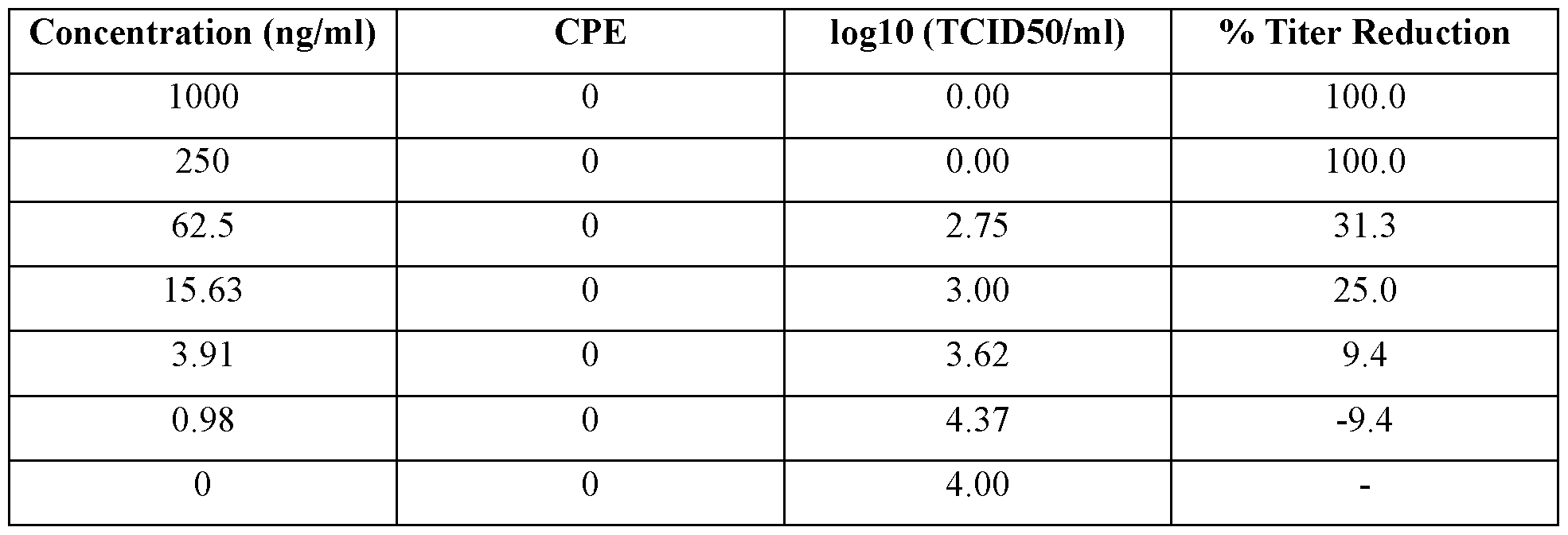
- a IFNZ.3 protein variant is considered to have the desired IFNz.3 activity if it inhibits the replication of BVDV by at least about 50%, about 60%, about 70%, about 80%, about 90% or about 95%, e.g., if it inhibits the replication of BVDV, such as a cytopathic genotype la BVDV strain, in MDBK cells by at least about 50%, about 60%, about 70%, about 80%, about 90% or about 95% after treatment of the MDBK cells with the IFNZ.3 protein variant at a concentration of > 250 ng/ml, and wherein the inhibition of BVDV replication is determined 24 hours after the treatment.
- the desired IFNZ.3 biological activity is the ability to inhibit the replication of foot and mouth disease virus (FMDV).
- FMDV foot and mouth disease virus
- Methods of determining the ability to inhibit the replication of FMDV are disclosed herein.
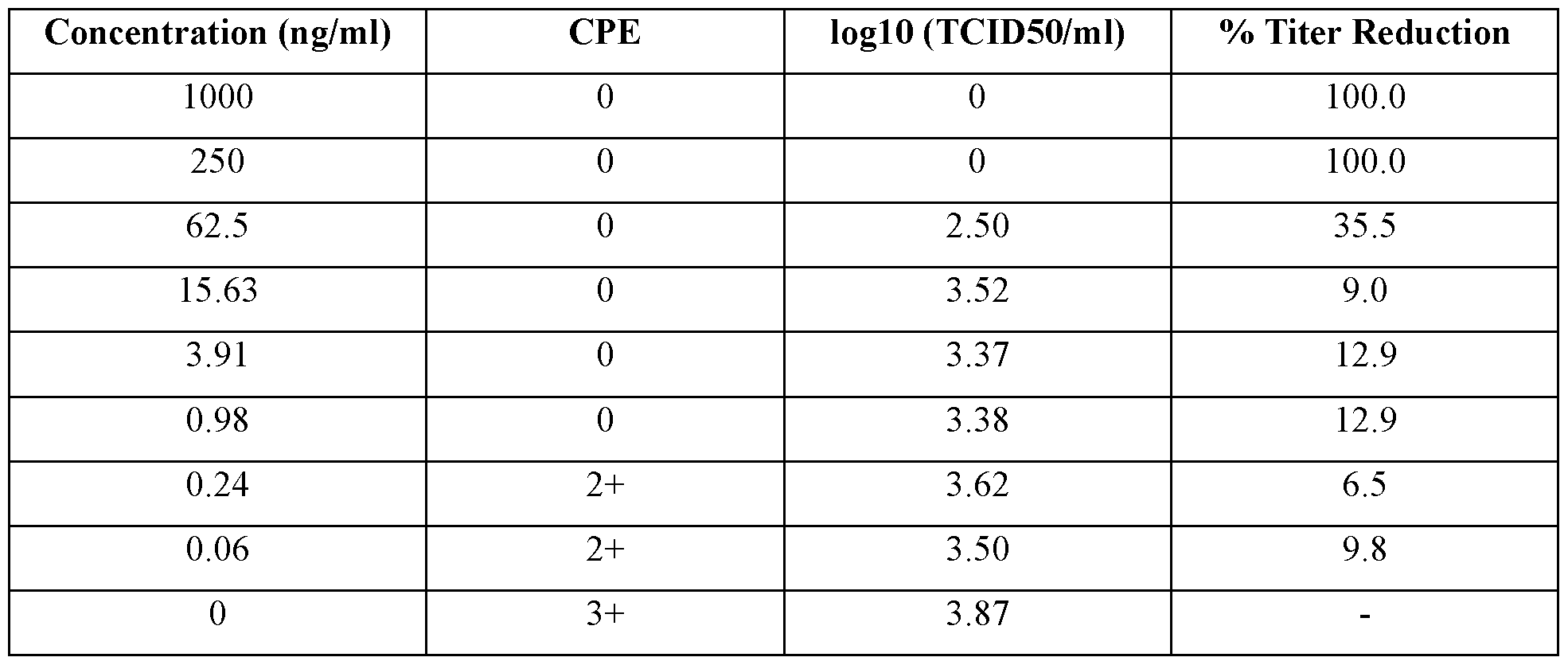
- a IFNZ.3 protein variant is considered to have the desired IFNz.3 activity if it inhibits the replication of FMDV by at least about 50%, about 60%, about 70%, about 80%, about 90% or about 95%, e.g., if it inhibits the replication of FMDV, such as South African Territories (SAT)l FMDV, in MDBK cells by at least about 50%, about 60%, about 70%, about 80%, about 90% or about 95% after treatment of the MDBK cells with > 10 ng/ml concentrations of the IFNX3 protein variant, and wherein the inhibition of FMDV replication is determined approximately 48 hours after the treatment.
- SAT South African Territories
- the biologically active variant may differ from the corresponding wild type protein by as few as 1 to 15 amino acid residues, as few as 1 to 10 amino acid residues, as few as 1 to 5 amino acid residues, as few as 1 to 3 amino acid residues, or by 1 amino acid residue.
- viremia refers to the presence of virus(es) in the bloodstream and/or the entry of virus(es) into the bloodstream.
- Methods of measuring viremia in a subject are known to a person of ordinary skill in the art. Such methods include drawing blood sample(s) from the subject and testing the sample (e.g., for viral RNA) using a polymerase chain reaction (PCR), such as quantitative reverse transcription PCR (RT-PCR) (e.g., see Zhang Z. and Alexandersen S. (2003). J Virol Methods, 111 :95-100; and Stenfeldt C. and Belsham G.J. (2012) Veterinary Microbiology, 154:230-239).
- PCR polymerase chain reaction
- RT-PCR quantitative reverse transcription PCR
- Viremia can include primary viremia, which refers to the initial spread of virus in the blood from a primary site of infection, and/or secondary viremia, which occurs when primary viremia has resulted in infection of additional tissues via the bloodstream and then reenters the bloodstream.
- primary viremia refers to the initial spread of virus in the blood from a primary site of infection
- secondary viremia which occurs when primary viremia has resulted in infection of additional tissues via the bloodstream and then reenters the bloodstream.
- water-soluble polymer refers to any polymer that is soluble in aqueous solvents.
- a water-soluble polymer derivative of the present disclosure includes a water- soluble polymer comprising a reactive group that is capable of reacting with a non-natural amino acid.
- Water-soluble polymers may or may not have their own biological activity.
- a water-soluble polymer of the present disclosure can be a polyalkylene glycol.
- polypeptides containing non-naturally encoded amino acid can be metabolized upon administration to an organism in need to produce a metabolite for use to provide a desired effect, for example, a desired therapeutic effect.
- active metabolites of polypeptides containing non-naturally encoded amino acids are active metabolites of polypeptides containing non-naturally encoded amino acids.
- non-naturally encoded amino acid polypeptides may exist as tautomers.
- the non-naturally encoded amino acid polypeptides described herein can exist in unsolvated as well as solvated forms with pharmaceutically acceptable solvents such as water, ethanol, and the like.
- the solvated forms are also considered to be disclosed herein.
- Those of ordinary skill in the art will recognize that some of the compounds herein can exist in several tautomeric forms. All such tautomeric forms are considered as part of the compositions described herein.
- MS mass spectroscopy
- NMR nuclear magnetic resonance
- HPLC high performance liquid chromatography
- reference to the term “having an amino acid sequence” or “has an amino acid sequence” may encompass “comprises an amino acid sequence” or “comprising an amino acid sequence”.
- a protein having an amino acid sequence of SEQ ID NO: 2 or that has an amino acid sequence of SEQ ID NO: 2 may comprise the amino acid sequence of SEQ ID NO: 2.
- reference to the term “having an amino acid sequence” or “has an amino acid sequence” may encompass “consisting of an amino acid sequence”.
- a protein having an amino acid sequence of SEQ ID NO: 2 may consist of the amino acid sequence of SEQ ID NO: 2.
- boIFNz.3 proteins and variants thereof including boIFN/3 variants comprising one or more non-naturally encoded amino acids.
- the boIFNZ.3 variants of the present disclosure are engineered proteins that can be produced in cells equipped with a translation system comprising an orthogonal tRNA and an orthogonal aminoacyl tRNA synthetase, thereby allowing for the site-specific incorporation of non-natural amino acids into boIFNZ.3 protein, as disclosed herein.
- the present disclosure further provides conjugated boIFNz.3 proteins.
- Conjugated boIFNZ.3 proteins of the present disclosure comprise engineered boIFNz.3 proteins containing one or more non-natural amino acid, wherein the engineered boIFNz.3 protein is conjugated to a moiety that is capable of extending boIFNz.3 protein elimination half-life.
- a half-life extender moiety such as a water-soluble polymer, can be conjugated to an engineered boIFNz.3 protein via a stable covalent linkage to the non-natural amino acid. The stable covalent linkage can reduce or eliminate the potential for cleavage of the half-life extender moiety from the protein in vivo.
- the stable linkage provides the conjugated protein with an increased and consistent elimination half-life, improved pharmacodynamics and/or an improved therapeutic profile compared to other IFNs, or to other conjugated IFNs that do not contain a stable covalent linkage joining the half-life extender moiety to the IFN via a site-specifically incorporated non-natural amino acid.
- the present disclosure also provides pharmaceutical compositions containing engineered boIFNZ.3 proteins, or conjugated boIFNz.3 proteins or variants thereof. Also provided are methods of treating (e.g., preventing) diseases or conditions, such as BRD and FMD, in ruminant mammals including cattle, via the administration of a conjugated boIFNz.3 protein of the present disclosure.
- the treatment can include combination therapy by further administration of a vaccine.
- a conjugated boIFNz.3 protein of the present disclosure has an elimination half-life that exceeds that of typical IFNs and can provide early and sustained IFN activity in vivo. Consequently, the presently disclosed conjugated boIFN/3 proteins can provide early and sustained anti-viral activity in treated animals, thereby protecting the treated animals from infection by viruses associated with BRD or FMD. While vaccination is a useful tool for preventing diseases such as FMD, development of protective adaptive immune response takes about 5 to 7 days post-vaccination (Diaz-San Segundo F. et al. (2021) Frontiers in Microbiology V12; Article 668890; Diaz-San Segundo F. et al.
- the protection from viral infection provided by administration of a conjugated boIFNZ.3 of the present disclosure, alone or in combination with a FMD vaccine, can protect treated animals for at least 5 days postadministration.
- treatment of an animal with a combination of a conjugated boIFNZ.3 protein of the present disclosure and a vaccine can provide the treated animal with protection against viral infection prior to the development of adaptive immune response, thereby filling the gap between time of vaccination and development of antibody-mediated anti-viral activity in vivo.
- treatment with a conjugated boIFNz.3 protein of the present disclosure has the potential to induce long-term protection against viral infection.
- the present disclosure provides engineered boIFNz.3 proteins, including boIFNz.3 protein deletion variants.
- BoIFNz.3 proteins and variants of the present disclosure are characterized by features including their amino acid sequence and/or elimination half-life in vivo.
- the present disclosure includes engineered boIFNz.3 proteins and variants produced using the compositions and methods disclosed herein.
- An excipient (including but not limited to, a pharmaceutically acceptable excipient) can also be present with the protein or variant.
- Table 1 discloses boIFNz.3 protein amino acid sequences, including wild type protein and variant sequences, wherein the variant sequences are N-terminal truncations of the wild type sequence.
- the amino acid sequences can contain a non-natural amino acid.
- Table 1 further discloses some nucleic acid sequences encoding amino acids sequences of the present disclosure.
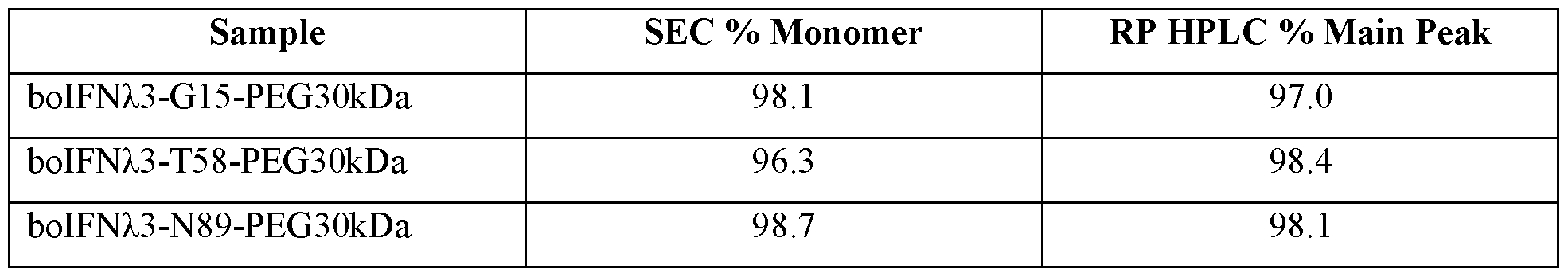
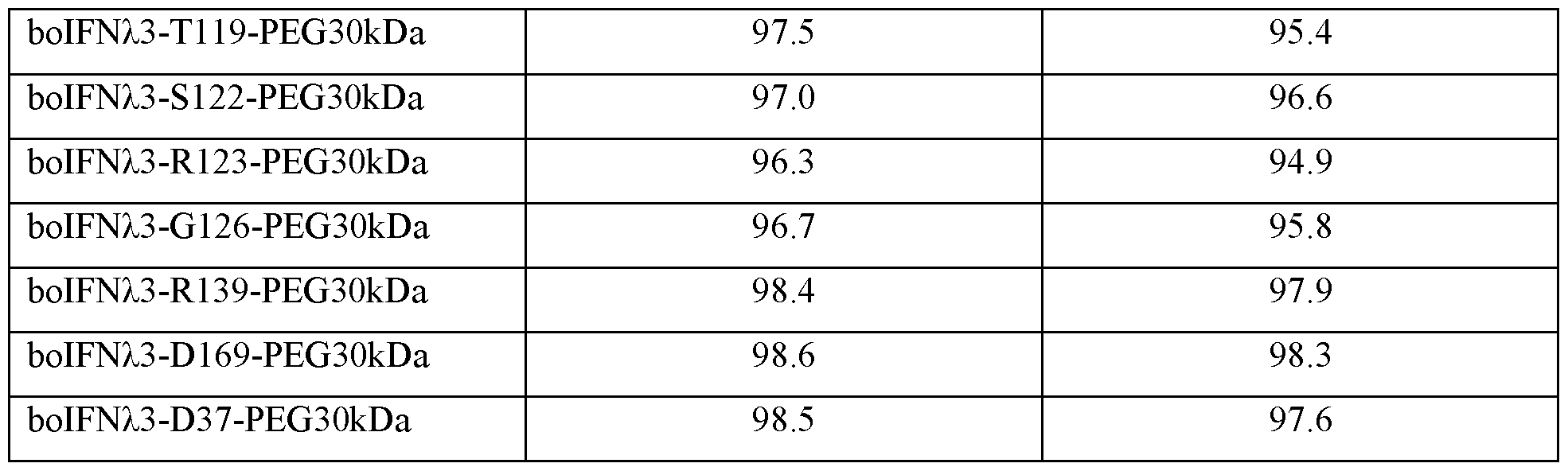
- Table 1 Bovine IFNZ.3 protein amino acid sequences that can include one or more non- naturally encoded amino acids (nnAA), and bovine IFNz.3 nucleic acid sequences.
- amino acid sequences in Table 1 wherein: (i) any amino acid is replaced with any nnAA; (ii) any amino acid is replaced with pyrrolysine; (iii) X is replaced with any nnAA; (iv) X is pAF; (v) X is pyrrolysine; and all the foregoing, wherein the amino acid sequence further contains an N-terminal methionine residue, unless such a residue is expressly shown.
- amino acid position refers to the position in the corresponding wild type sequence (SEQ ID NO: 1) with no
- a boIFNZ.3 protein variant of the present disclosure is an N- terminal deletion variant.
- a boIFNZ.3 protein variant of the present disclosure can have an amino acid sequence disclosed in Table 1, wherein the amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 1, wherein each sequence optionally further comprises an N-terminal methionine, and wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNz.3 protein variant amino acid sequence shares at most about 99%, or at most about 98% identity with SEQ ID NO: 1, wherein each sequence optionally further comprises an N-terminal methionine. In some embodiments, the boIFNZ.3 protein variant amino acid sequence shares at most about 98% identity with SEQ ID NO: 1. In some embodiments, the boIFNZ.3 protein variant amino acid sequence shares at most about 99% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant of the present disclosure is an N- terminal deletion variant having an amino acid sequence disclosed in Table 1, wherein the boIFNz.3 protein variant amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 1, and wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant amino acid sequence shares at most about 99%, or at most about 98% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant amino acid sequence shares at most about 98% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant amino acid sequence shares at most about 99% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant of the present disclosure is an N- terminal deletion variant having an amino acid sequence disclosed in Table 1, wherein each sequence further comprises an N-terminal methionine, and wherein the boIFNZ.3 protein variant amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 1, and shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant amino acid sequence of Table 1 that further comprises an N-terminal methionine shares at most about 99%, or at most about 98% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant amino acid sequence of Table 1 that further comprises an N-terminal methionine shares at most about 98% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant amino acid sequence of Table 1 that further comprises an N-terminal methionine shares at most about 99% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant of the present disclosure can have an amino acid sequence disclosed in Table 1, wherein the amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with (i) SEQ ID NO: 2, 3, 4, 5, 6, 7,
- SEQ ID NO: 2 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNX3 protein variant of the present disclosure can have an amino acid sequence disclosed in Table 1, wherein the amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8,
- amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant of the present disclosure can have an amino acid sequence disclosed in Table 1, wherein the amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; and wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant of the present disclosure can have an amino acid sequence that shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1; optionally wherein the boIFNz.3 protein variant further comprises an N-terminal methionine.
- a boIFNz.3 protein variant of the present disclosure can have an amino acid sequence, wherein the boIFNz.3 protein variant amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant of the present disclosure can have an amino acid sequence, wherein the boIFNz.3 protein variant amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing amino acid sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNX3 protein variant has an amino acid sequence that shares at least 75% identity with (i) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; or (ii) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNX3 protein variant has an amino acid sequence that shares at least 75% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNX3 protein variant has an amino acid sequence that shares at least 75% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNX3 protein variant has an amino acid sequence that shares at least 75% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 75% identity with SEQ ID NO: 5; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 75% identity with SEQ ID NO: 5, wherein the boIFNZ.3 protein variant further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNX3 protein variant has an amino acid sequence that shares at least 85% identity with (i) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; or (ii) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNX3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNX3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNX3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 5; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 5, wherein the sequence further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNX3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- a boIFNZ.3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 5; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 5, wherein the boIFNX3 protein variant further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNX3 protein variant has an amino acid sequence that shares at least 90% identity with (i) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; or (ii) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNX3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNX3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 5; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 5, wherein the sequence further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNX3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 5, wherein the boIFNZ.3 protein variant further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with (i) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; or (ii) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 5; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 5, wherein the sequence further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 5, wherein the boIFNZ.3 protein variant further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with (i) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; or (ii) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 5; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNX3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 5, wherein the sequence optionally further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNX3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNX3 protein variant amino acid sequence further comprises an N-terminal methionine.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNX3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 5; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 5, wherein the boIFNZ.3 protein variant further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNX3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 5; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNX3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 5, wherein the sequence further comprises an N- terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNX3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 5, wherein the boIFNZ.3 protein variant further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 5; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNX3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 5, wherein the sequence further comprises an N- terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNX3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 5, wherein the boIFNZ.3 protein variant further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNX3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 5; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNX3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 5, wherein the sequence further comprises an N- terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNX3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 5, wherein the boIFNZ.3 protein variant further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 99% identity with (i) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; or (ii) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNX3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNX3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- a boIFNz.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNz.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 5, wherein the boIFNZ.3 protein variant further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 2; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 2; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 2, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 3; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 3; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 3, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 4; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 4; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 4, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 5; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 5; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 5, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 6; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 6; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 6, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 7; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 7; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 7, wherein amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 8; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 8; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 8, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 9; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 9; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 9, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 10; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 10; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 10, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 11; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 11; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 11, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 proteinv variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 12; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 12; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 12, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 13, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 2. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 2, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N- terminal methionine.
- the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 3. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 3, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N- terminal methionine.
- the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 4. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 4, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N- terminal methionine.
- the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 5. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 5, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N- terminal methionine.
- the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 6. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 6, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N- terminal methionine.
- the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 7. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 7, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N- terminal methionine.
- the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 8. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 8, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N- terminal methionine.
- the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 9. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 9, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N- terminal methionine.
- the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 10. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 10, wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine. [00201] In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 11. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 11, wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 12. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 12, wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 13. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 13, wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- any definition of a protein or variant can equally be applied to the boIFNZ.3 protein portion of a conjugate, and vice versa.
- a boIFNZ.3 protein or variant of the present disclosure can comprise one or more nonnatural amino acids, as further disclosed herein.
- a boIFNZ.3 protein or variant of the present disclosure can comprise an unusual genetically encoded amino acid that is not present in humans.
- the unusual genetically encoded amino acid can be selenocysteine or pyrrolysine, the latter of which is known to be used in the biosynthesis of proteins in some methanogenic archaea and bacteria.
- the present disclosure also provides a nucleic acid having a nucleotide sequence disclosed in Table 1, wherein the nucleotide sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 26.
- the nucleic acid has a nucleotide sequence that shares at least 75% identity with SEQ ID NO: 26.
- the nucleic acid has a nucleotide sequence that shares at least 85% identity with SEQ ID NO: 26.
- the nucleic acid has a nucleotide sequence that shares at least 90% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 95% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 96% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 97% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 98% identity with SEQ ID NO: 26.
- the nucleic acid has a nucleotide sequence that shares at least 99% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares less than 100% identity with SEQ ID NO: 26.
- the present disclosure also provides a nucleic acid having a nucleotide sequence wherein the nucleotide sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 26.
- the nucleic acid has a nucleotide sequence that shares at least 75% identity with SEQ ID NO: 26.
- the nucleic acid has a nucleotide sequence that shares at least 85% identity with SEQ ID NO: 26.
- the nucleic acid has a nucleotide sequence that shares at least 90% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 95% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 96% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 97% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 98% identity with SEQ ID NO: 26.
- the nucleic acid has a nucleotide sequence that shares at least 99% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares less than 100% identity with SEQ ID NO: 26.
- the present disclosure provides a boIFN+3 protein variant that is characterized as having an amino acid sequence comprising at least one non-natural amino acid.
- the boIFN+3 protein variant can be further characterized by its elimination half-life.
- BoIFN/3 protein variants containing at least one non-natural amino acid can be used for the treatment of a disease or condition in a mammal, and/or can be used for producing protein conjugates of the present disclosure.
- the invention includes boIFN+3 protein variants produced using the compositions and methods of the invention.
- An excipient (including but not limited to, a pharmaceutically acceptable excipient) can also be present with the protein or variant thereof.
- non-natural amino acids into proteins permits the introduction of reactive chemical moieties as alternatives to those present in canonical amino acids, such as the epsilon -NEE of lysine, the sulfhydryl -SH of cysteine, the hydroxyl -OH of serine, and the imino group of histidine.
- reactive chemical moieties such as the epsilon -NEE of lysine, the sulfhydryl -SH of cysteine, the hydroxyl -OH of serine, and the imino group of histidine.
- Certain chemical functional groups are known to be inert to the functional groups found in the 20 canonical amino acids but react cleanly and efficiently with other moieties to form stable linkages.
- azide and acetylene groups which are not present in canonical amino acids, are known in the art to undergo a Huisgen [3+2] cycloaddition reaction in aqueous conditions in the presence of a catalytic amount of copper. See, e.g., Tomoe, et al., (2002) J. Org. Chem. 67 :3057 -3064; and Rostovtsev, et al., (2002) Angew. Chem. Int. Ed. 41 :2596-2599.
- an azide moiety By introducing an azide moiety into a protein structure, one is able to incorporate a functional group that is chemically inert to amines, sulfhydryls, carboxylic acids and hydroxyl groups found in proteins, but that reacts smoothly and efficiently with an acetylene moiety to form a cycloaddition product. Importantly, in the absence of the acetylene moiety, the azide remains chemically inert and unreactive in the presence of other protein side chains and under physiological conditions.
- the introduction of a non-natural amino acid into a protein provides it with alternative chemical moieties that can be used to selectively conjugate additional molecules to the protein.
- the present disclosure provides the introduction of at least one nonnatural amino acid into boIFNZ.3 protein or a variant thereof, allowing for selective conjugation to occur with an additional molecule, such as a molecule that extends its elimination half-life.
- the non-natural amino acids used in the methods and compositions described herein have at least one of the following properties: (1) at least one functional group on the sidechain of the non-natural amino acid has at least one characteristic and/or activity and/or reactivity orthogonal to the chemical reactivity of the canonical amino acids, or at least orthogonal to the chemical reactivity of the canonical amino acids present in the polypeptide that includes the non-natural amino acid; (2) the introduced non-natural amino acids are substantially chemically inert toward the canonical amino acids; and (3) the non-natural amino acid can be stably incorporated into a polypeptide, preferably with the stability commensurate with the canonical amino acids or under typical physiological conditions, and further preferably such incorporation can occur in a cell-based system, such as cell culture.
- the one or more non-natural amino acids is 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 non-natural amino acids. In some embodmients, the one or more non-natural amino acids is 1, 2, 3, 4 or 5 non-natural amino acids. In some embodiments, the one or more non-natural amino acids is 1, 2 or 3 non-natural amino acids. In some embodiments, the one or more non-natural amino acids is one (1) non-natural amino acid.
- the one or more non-natural amino acids may be one or more non-naturally encoded amino acids.
- the one or more non-naturally encoded amino acids may be encoded by a codon that does not code for one of the twenty canonical amino acids.
- the one or more non-natural amino acids may be encoded by a nonsense codon (stop codon).
- the stop codon may be an amber codon.
- the amber codon may comprise a UAG sequence.
- the stop codon may be an ochre codon.
- the ochre codon may comprise a UAA sequence.
- the stop codon may be an opal or umber codon.
- the opal or umber codon may comprise a UGA sequence.
- Non-natural amino acids of the present disclosure include, but are not limited to, 1) substituted phenylalanine and tyrosine analogues, such as 4-amino-L-phenylalanine, 4-acetyl-L- phenylalanine, 4-azido-L-phenylalanine, 4-nitro-L-phenylalanine, 3 -m ethoxy -L-phenylalanine, 4- isopropyl-L-phenylalanine, 3-nitro-L-tyrosine, O-methyl-L-tyrosine and O-phosphotyrosine; 2) amino acids that can be photo-cross-linked, e.g., amino acids with aryl azide or benzophenone groups, such as 4-azidophenylalanine or 4-benzoylphenylalanine; 3) amino acids that have unique chemical reactivity, such as 4-acet
- non-natural amino acids include, but are not limited to, a p-acetylphenylalanine (e.g., 4-acetyl-L-phenylalanine), a 4-boronophenylalanine (pBoF) (e.g., 4-borono-L-phenylalanine, a 4-propargyloxyphenylalanine (pPrF) (e.g., 4- propargyloxy-L-phenylalanine), an O-methyltyrosine (e.g., O-methyl-L-tyrosine), a 3-(2- naphthyl)alanine (NapA) (e.g., 3-(2-naphthyl)-L-alanine), a 3 -methylphenylalanine (e.g., 3- methyl-L-phenylalanine), an O-allyltyrosine (e.g., O-allyl-L-tyrosine), an O-is
- the one or more non-natural amino acids can be p- acetylphenylalanine.
- the one or more non-natural amino acids can be p-acetyl-L-phenylalanine (pAF).
- one or more non-natural amino acids is selected from the group consisting of 4-acetylphenylalanine, 3-O-(N-acetyl-beta-D-glucosaminyl)threonine, N4-( -N- Acetyl-D-glucosaminyl)asparagine, O-allyltyrosine, alpha-N-acetylgalactosamine-O-serine, alpha-N-acetylgalactosamine-O-threonine, 2-aminooctanoic acid, 2-aminophenylalanine, 3- aminophenylalanine, 4-aminophenylalanine, 2-aminotyrosine, 3 -aminotyrosine, 4- azidophenylalanine, 4-benzoylphenylalanine, (2,2-bipyridin-5yl)alanine, 3-boronophenylalanine, 4-boronophen
- one or more non-natural amino acids is selected from the group consisting of 4-acetyl-L-phenylalanine (para-acetyl-L-phenylalanine (pAF)), 3-O-(N- acetyl-beta-D-glucosaminyl)-L-threonine, N4-(P-N-Acetyl-D-glucosaminyl)-L-asparagine, O- allyl-L-tyrosine, alpha-N-acetylgalactosamine-O-L-serine, alpha-N-acetylgalactosamine-O-L- threonine, 2-aminooctanoic acid, 2-amino-L-phenylalanine, 3-amino-L-phenylalanine, 4-amino- L-phenylalanine, 2-amino-L-tyrosine, 3-amino-
- the non-natural amino acid includes an aminooxy functional group or a functional group that can be transformed into an aminooxy group by reacting with a reagent, preferably under conditions that do not destroy the biological properties of the polypeptide that includes the non-natural amino acid (unless of course such a destruction of biological properties is the purpose of the modification/transformation), or where the transformation can occur under aqueous conditions at a pH between about 4 and about 8.
- the non-natural amino acid includes a carbonyl (e.g., an acyl) functional group or a functional group that can be transformed into a carbonyl (e.g., an acyl) group by reacting with a reagent, preferably under conditions that do not destroy the biological properties of the polypeptide that includes the non-natural amino acid (unless of course such a destruction of biological properties is the purpose of the modification/transformation), or where the transformation can occur under aqueous conditions at a pH between about 4 and about 8. Any number of non-natural amino acids can be introduced into the polypeptide.
- a carbonyl e.g., an acyl
- a functional group that can be transformed into a carbonyl (e.g., an acyl) group by reacting with a reagent, preferably under conditions that do not destroy the biological properties of the polypeptide that includes the non-natural amino acid (unless of course such a destruction of biological properties is the purpose of the modification/transformation), or where the transformation can occur under
- Non-natural amino acids may also include protected or masked oximes or protected or masked groups that can be transformed into an oxime group after deprotection of the protected group or unmasking of the masked group.
- Oxime-based non-natural amino acids may be synthesized by methods well known in the art (see for example WO 2013/185117 and WO 2005/074650, the entire contents of each of which are hereby incorporated by reference herein in their entirety), including reaction of a carbonyl-containing non-natural amino acid with a hydroxylamine or aminooxy-containing reagent.
- Non-natural amino acids may also include protected or masked carbonyl groups, which can be transformed into a carbonyl group (e.g., an acyl group) after deprotection of the protected group or unmasking of the masked group and thereby are available to react with aminooxy groups to form oxime groups.
- a carbonyl group e.g., an acyl group
- One non-natural amino acid containing an acyl group is p-acetylphenylalanine.
- the present invention contemplates incorporation of one or more non-natural amino acids into boIFNZ.3 protein.
- One or more non-natural amino acids may be incorporated at a particular position which does not disrupt activity of the protein. This can be achieved by making "conservative" substitutions, including but not limited to, substituting hydrophobic amino acids with non-natural hydrophobic amino acids, bulky amino acids with non-natural bulky amino acids, hydrophilic amino acids with non-natural hydrophilic amino acids and/or inserting the non-natural amino acid into a location that is not required for activity.
- Selection of desired sites may be based on producing a non-natural amino acid polypeptide (which may be further modified or remain unmodified) having any desired property or activity, including but not limited to a receptor binding modulator, receptor activity modulator, modulator of binding to binder partners, binding partner activity modulator, binding partner conformation modulator, dimer or multimer formation, no change to activity or property compared to the native molecule, or manipulating any physical or chemical property of the protein such as solubility, aggregation or stability.
- the sites identified as critical to biological activity may also be good candidates for substitution with a non-natural amino acid, again depending on the desired activity sought for the protein.
- the structure and activity of naturally-occurring mutants of a protein that contain deletions can also be examined to determine regions of the protein that are likely to be tolerant of substitution with a non-natural amino acid. Once residues that are likely to be intolerant to substitution with non-natural amino acids have been eliminated, the impact of proposed substitutions at each of the remaining positions can be examined using methods including, but not limited to, the three-dimensional structure of the relevant protein, and any associated ligands or binding proteins.
- X-ray crystallographic and NMR structures of many polypeptides are available in the Protein Data Bank (PDB; see world wide web for rcsb.org), a centralized database containing three-dimensional structural data of large molecules of proteins and nucleic acids, which can be used to identify amino acid positions that can be substituted with non-natural amino acids.
- PDB Protein Data Bank
- models may be made investigating the secondary and tertiary structure of polypeptides, if three-dimensional structural data are not available.
- those of ordinary skill in the art can readily identify amino acid positions that can be substituted with non-natural amino acids.
- Exemplary sites of incorporation of a non-natural amino acid include, but are not limited to, those that are excluded from potential receptor binding regions, or regions for binding to binding proteins or ligands may be fully or partially solvent exposed, have minimal or no hydrogen-bonding interactions with nearby residues, may be minimally exposed to nearby reactive residues, and/or may be in regions that are highly flexible as predicted by the three-dimensional crystal structure of a particular polypeptide with its associated receptor, ligand or binding proteins.
- a wide variety of non-natural amino acids can be substituted for, or incorporated into, a given position in a polypeptide.
- a particular non-natural amino acid may be selected for incorporation based on an examination of the three-dimensional crystal structure of a polypeptide with its associated ligand, receptor and/or binding proteins, with a preference for conservative substitutions.
- non-natural amino acid site selection can be based on surface exposure/site accessibility within the protein. Hydrophobic or neutral amino acid sites can be selected to maintain the charge on the protein. In some embodiments, non-naturally encoded amino acid site selection is based on surface exposure.
- one possible site is an amino acid having a solvent accessible surface area ratio of 30% or more, 40% or more, 50% or more, 60% or more, 70% or more, 80% or more, 90% or more, or 95% or more.
- one possible site is an amino acid having a solvent accessible surface area ratio of about 30%, about 40%, about 50%, about 60%, about 70%, about 80%, about 90%, or about 95%, or more.
- the solvent accessible surface area can be calculated based on the DSSP program (Kabsch W. and Sander C. (1983) Biopolymers, 22:2577-2637), using a crystalline structure analyzing data file of proteins registered in the PDB.
- the ratio of the solvent accessible surface area of the amino acid residues of interest can be calculated by dividing the protein structural solvent accessible surface area calculated in the above by the solvent accessible surface area of alanine-X-alanine (wherein X represents the amino acid residue(s) of interest).
- X represents the amino acid residue(s) of interest.
- the solvent accessibility of an amino acid can be determined by a solvent accessibility test in which a functional group on the amino acid (e.g., a thiol, amino, or carbonyl group) is functionalized when treated with an electrophilic reagent or a nucleophilic reagent, or the like. Based on the test results, the functional group (e.g., the thiol, amino, or carbonyl group) can be called, for example, at least 50% solvent accessible when at least 50% of the functional group is functionalized in the test.
- a functional group on the amino acid e.g., a thiol, amino, or carbonyl group
- the functional group e.g., the thiol, amino, or carbonyl group
- the non-naturally encoded amino acid site is at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% solvent accessible.
- solvent accessibility test include, but are not limited to, propargylation of a surface thiol group, or a- bromopyruvate reacting with a surface thiol group, etc.
- a site for non-natural amino acid incorporation can be selected by modeling a desired non-natural amino acid at each amino acid site, e.g., based on a protein crystalline structure of a corresponding protein, and assessing side chain accessible surface area (scASA) of the desired non-natural amino acid.
- scASA side chain accessible surface area
- non-natural amino acid site selection is based on scASA.
- one possible site is an amino acid having an scASA value of about 98 angstroms 2 (A 2 ) or more, about 125 A 2 or more, about 150 A 2 or more, about 175 A 2 or more, about 200 A 2 or more, about 225 A 2 or more, about 250 A 2 or more, or about 275 A 2 or more.
- a protein includes at least one non-natural amino acid and at least one post-translational modification that is made by a eukaryotic cell, where the post-translational modification is not made by a prokaryotic cell.
- the post-translation modification can include acetylation, acylation, lipid-modification, palmitoylation, palmitate addition, phosphorylation, glycolipid-linkage modification, glycosylation, and the like.
- a protein with at least one non-natural amino acid comprising a first reactive group undergoes at least one post-translational modification.
- the post-translational modification is made in a eukaryotic cell or in a non- eukaryotic cell.
- the post-translational modification is made in vitro.
- the post-translational modification is made in vitro and in vivo.
- the post-translational modification is through the non-natural amino acid.
- the molecule to be attached to the protein comprises a second reactive group that is capable of reacting with the first reactive group of the non-natural amino acid, and the molecule is attached to the protein utilizing chemistry methodology that is known to one of ordinary skill in the art to be suitable for the particular reactive groups.
- the post-translational modification can be through a nucleophilic-electrophilic reaction.
- Most reactions currently used for the selective modification of proteins involve covalent bond formation between nucleophilic and electrophilic reaction partners. Selectivity in these cases is determined by the number and accessibility of the nucleophilic residues in the protein.
- other more selective reactions can be used such as the reaction of a non-natural keto-amino acid with hydrazides or aminooxy compounds.
- the first reactive group is a keto moiety (including but not limited to, the acetyl group of the non-natural amino acid paraacetylphenylalanine, or para-acetyl-L-phenylalanine) and the second reactive group is an aminooxy moiety.
- the first reactive group is an azido moiety (including but not limited to, the azido group of the non-natural amino acid / /ra-azido-L-phenylalanine) and the second reactive group is an alkynyl moiety.
- a post-translational modification to the protein comprises attachment of any desirable compound or substance, including but not limited to a moiety that is capable of extending a protein’s elimination half-life, as disclosed herein.
- the non-natural amino acid is site-specifically incorporated into the protein or variant.
- auxotrophic strains may also be used in place of engineered tRNA and synthetase.
- orthogonal tRNA synthetases are used as disclosed in, for example, WO 2002/085923, WO 2002/086075, WO 2004/035743, WO 2007/021297, WO 2006/068802 and WO 2006/069246; the entire contents of each of which are hereby incorporated by reference herein in their entirety.
- Incorporating one or more non-natural amino acids into the protein or variant thereof may comprise modifying one or more amino acid residues in the protein or variant.
- Modifying the one or more amino acid residues in the protein or variant may comprise mutating one or more nucleotides in the nucleic acid encoding the protein or variant. Mutating the one or more nucleotides in the nucleic acid encoding the protein or variant may comprise altering a codon encoding an amino acid to a nonsense codon. Incorporating one or more non-natural amino acids into the protein or variant may comprise modifying one or more nucleotides in the nucleic acid to introduce one or more amber codons into the nucleotide sequence. The one or more non-natural amino acids may be incorporated into the protein or variant in response to an amber codon. The one or more non-natural amino acids may be site-specifically incorporated into the protein or variant.
- Incorporating one or more non-natural amino acids into the protein or variant may comprise incorporating one or more genetically encoded non-natural amino acids with orthogonal chemical reactivity relative to the 20 canonical amino acids to site-specifically modify the protein or variant.
- Incorporating the one or more non-natural amino acids may comprise use of a tRNA/aminoacyl-tRNA synthetase pair to site-specifically incorporate one or more non-natural amino acids at defined sites in the protein or variant in response to one or more amber nonsense codon.
- the boIFNZ.3 protein variants of the invention can be generated using modified tRNA and tRNA synthetases to add to or substitute amino acids that are not encoded in naturally- occurring systems.
- Methods for generating tRNAs and tRNA synthetases which use amino acids that are not encoded in naturally-occurring systems are described in, e.g., U.S. Patent Nos. 7,045,337 and 7,083,970, the contents of each of which are hereby incorporated by reference herein in their entirety. These methods involve generating a translational machinery that functions independently of the synthetases and tRNAs endogenous to the translation system (and are therefore sometimes referred to as “orthogonal”).
- the translation system comprises an orthogonal tRNA (O-tRNA) and an orthogonal aminoacyl tRNA synthetase (O-RS).
- O-tRNA orthogonal tRNA
- O-RS orthogonal aminoacyl tRNA synthetase
- Use of O- tRNA/aminoacyl-tRNA synthetases involves selection of a specific codon which encodes the non- naturally encoded amino acid (a selector codon). While any codon can be used, it is generally desirable to select a codon that is rarely or never used in the cell in which the O-tRNA/aminoacyl- tRNA synthetase is expressed.
- exemplary codons include nonsense codon such as stop codons (amber, ochre, and opal), four or more base codons and other natural three-base codons that are rarely used or unused.
- Specific selector codon(s) can be introduced into appropriate positions in boIFNZ.3 sequence using mutagenesis methods known in the art (including but not limited to, site-specific mutagenesis, cassette mutagenesis, restriction selection mutagenesis, etc.).
- the O-RS preferentially aminoacylates the O-tRNA with at least one non-natural amino acid in the translation system and the O-tRNA recognizes at least one selector codon that is not recognized by other tRNAs in the system.
- the translation system thus inserts the non-naturally encoded amino acid into a protein produced in the system, in response to an encoded selector codon, thereby "substituting" an amino acid into a position in the encoded polypeptide.
- a boIFNZ.3 protein variant of the present disclosure containing a non-natural amino acid is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA synthetase.
- the non-natural amino acid is site-specifically incorporated.
- orthogonal tRNAs and aminoacyl tRNA synthetases have been described in the art for inserting particular synthetic amino acids into polypeptides and are generally suitable for use in the present invention.
- keto-specific O-tRNA/aminoacyl- tRNA synthetases are described in Wang, L. et al. (2003) Proc. Natl. Acad. Sci. USA 100:56-61 and Zhang, Z. et al. (2003) Biochem. 42(22):6735-6746.
- Exemplary O-RS, or portions thereof are encoded by polynucleotide sequences and include amino acid sequences disclosed in U.S. Patent Nos. 7,045,337 and 7,083,970.
- O-tRNA molecules for use with the O-RSs are also described in U.S. Patent Nos. 7,045,337 and 7,083,970. Additional examples of O- tRNA/aminoacyl-tRNA synthetase pairs are described in WO 2005/007870, WO 2005/007624 and WO 2005/019415, the entire contents of each of which are hereby incorporated by reference herein in their entirety.
- An example of an azide-specific O-tRNA/aminoacyl-tRNA synthetase system is described in Chin, J. W. et al. (2002) J. Am. Chem. Soc. 124:9026-9027.
- Exemplary O-RS sequences for /?-azido-L-Phe include, but are not limited to, nucleotide sequences SEQ ID NOs: 14-16 and 29-32 and amino acid sequences SEQ ID NOs: 46-48 and 61-64 as disclosed in U.S. Patent No. 7,083,970.
- Exemplary O-tRNA sequences suitable for use in the present invention include, but are not limited to, nucleotide sequences SEQ ID NOs: 1-3 as disclosed in U.S. Patent No. 7,083,970.
- Other examples of O-tRNA/aminoacyl-tRNA synthetase pairs specific to particular non-naturally encoded amino acids are described in U.S. Patent No. 7,045,337.
- O-RS and O-tRNA that incorporate both keto- and azide-containing amino acids in S. cerevisiae are described in Chin, J. W., et al., Science 301 :964-967 (2003).
- Several other orthogonal pairs have been reported. Glutaminyl (see, e.g., Liu, D. R., and Schultz, P. G. (1999) Proc. Natl. Acad. Sci. U. S. A. 96:4780-4785), aspartyl (see, e.g., Pastrnak, M., et al., (2000) Helv. Chim.
- the one or more non-natural amino acids may be produced through selective reaction of one or more natural amino acids.
- the selective reaction may be mediated by one or more enzymes.
- the selective reaction of one or more cysteines with formylglycine generating enzyme may produce one or more formylglycines as described in Rabuka et al. (2012) Nature Protocols 7: 1052-1067.
- the one or more non-natural amino acids may be produced through a method involving a chemical reaction to form a linker.
- the chemical reaction to form the linker may include a bioorthogonal reaction.
- the chemical reaction to form the linker may include click chemistry. See, for example W02006/050262, the entire contents of which are hereby incorporated by reference herein in their entirety.
- the present disclosure provides a boIFNZ.3 protein or variant thereof comprising an unusual genetically-encoded amino acid that is not present in humans, such as selenocysteine or pyrrolysine.
- the boIFNZ.3 protein or variant thereof comprises selenocysteine.
- the boIFNZ.3 protein or variant thereof comprises pyrrolysine.
- the boIFNZ.3 protein or variant thereof comprising the unusual genetically encoded amino acid does not contain a non-natural amino acid.
- one or more non-naturally encoded amino acids are incorporated into one or more of the following positions in wild type boIFNZ.3 (SEQ ID NO: 1) or into the corresponding amino acid positions in a boIFNZ.3 variant having an amino acid sequence of SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13: before position 1 (i.e.
- the non naturally encoded amino acid may substitute for the amino acid present at the relevant position in any one of the recited SEQ ID NOs.
- one or more non-naturally encoded amino acids are incorporated into one or more of the following positions of wild type boIFNZ.3 (SEQ ID NO: 1), or into the corresponding amino acid positions in a boIFNZ.3 protein variant having an amino acid sequence of SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13: position 15, 58, 65, 89, 119, 122, 123, 126, 139 and/or 169.
- one non-naturally encoded amino acid is incorporated into position 119 of wild type boIFNZ.3 (SEQ ID NO: 1), or into the corresponding amino acid position in a boIFNZ.3 protein variant having an amino acid sequence of SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13.
- a boIFNZ.3 protein variant of the present disclosure can have an amino acid sequence disclosed in Table 1, wherein the amino acid sequence comprises a nonnatural amino acid, and wherein the amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24 or 25; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23, 24 or 25, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- a boIFNZ.3 protein variant of the present disclosure can have an amino acid sequence disclosed in Table 1, wherein the amino acid sequence comprises a nonnatural amino acid, and wherein the amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or
- each of the foregoing sequences further comprises an N-terminal methionine.
- a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the amino acid sequence shares at least 75% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 75% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) shares at least 75% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the conjugated boIFNZ.3 has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 75% identity with SEQ ID NO: 18.
- the conjugated boIFNZ.3 has an amino acid sequence comprising a non- natural amino acid, wherein the amino acid sequence shares at least 75% identity with SEQ ID NO: 19.
- a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the amino acid sequence shares at least 80% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 80% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) shares at least 80% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence comprising a nonnatural amino acid, wherein the amino acid sequence shares at least 80% identity with SEQ ID NO: 18.
- the boIFNZ.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 80% identity with SEQ ID NO: 19.
- a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the amino acid sequence shares at least 85% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 85% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) shares at least 85% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence comprising a non- natural amino acid, wherein the amino acid sequence shares at least 85% identity with SEQ ID NO: 18.
- the boIFNZ.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 85% identity with SEQ ID NO: 19.
- a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the amino acid sequence shares at least 90% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence comprising a non- natural amino acid, wherein the amino acid sequence shares at least 90% identity with SEQ ID NO: 18.
- the boIFNZ.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 90% identity with SEQ ID NO: 19.
- a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the amino acid sequence shares at least 95% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 95% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24 or (ii) shares at least 95% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence comprising a non- natural amino acid, wherein the amino acid sequence shares at least 95% identity with SEQ ID NO: 18.
- the boIFNz.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 95% identity with SEQ ID NO: 19.
- a boIFNz.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the amino acid sequence shares at least 96% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- a boIFNz.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 96% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24 or (ii) shares at least 96% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNz.3 protein variant has an amino acid sequence comprising a non- natural amino acid, wherein the amino acid sequence shares at least 96% identity with SEQ ID NO: 18.
- the boIFNz.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 96% identity with SEQ ID NO: 19.
- a boIFNz.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the amino acid sequence shares at least 97% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 97% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) shares at least 97% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence comprising a nonnatural amino acid, wherein the amino acid sequence shares at least 97% identity with SEQ ID NO: 18.
- the boIFNZ.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 97% identity with SEQ ID NO: 19.
- a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the amino acid sequence shares at least 98% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 98% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24, or (ii) shares at least 98% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence comprising a non- natural amino acid, wherein the amino acid sequence shares at least 98% identity with SEQ ID NO: 18.
- the boIFNZ.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 98% identity with SEQ ID NO: 19.
- a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the amino acid sequence shares at least 99% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 99% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24, or (ii) shares at least 99% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
- the boIFNZ.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 18.
- the non-natural amino acid is pAF.
- the boIFNZ.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 19.
- the non-natural amino acid is pAF.
- the boIFNZ.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 14, 16, 17, 18, 19, 20, 21, 22 or 23.
- the non- natural amino acid is pAF.
- the boIFNZ.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 17, 18, 20 or 21.
- the non-natural amino acid is pAF.
- the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 14. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 14, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
- the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 15. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 15, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
- the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 16. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 16, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
- the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 17. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 17, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF. [00257] In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 18. In some embodiments, the non-natural amino acid is pAF.
- the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 19.
- the non-natural amino acid is pAF.
- the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 20. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 20, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
- the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 21. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 21, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
- the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 22. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 22, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
- the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 23. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 23, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
- the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 24. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 24, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
- the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 25. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 25, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
- any definition of a protein or variant can equally be applied to the protein portion of a conjugate, and vice versa.
- the present disclosure also provides a nucleic acid having a nucleotide sequence disclosed in Table 1, wherein the nucleotide sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 27.
- the present disclosure also provides a nucleic acid having a nucleotide sequence, wherein the nucleotide sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 27.
- the nucleic acid has a nucleotide sequence that shares at least 75% identity with SEQ ID NO: 27.
- the nucleic acid has a nucleotide sequence that shares at least 80% identity with SEQ ID NO: 27.
- the nucleic acid has a nucleotide sequence that shares at least 85% identity with SEQ ID NO: 27. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 90% identity with SEQ ID NO: 27. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 95% identity with SEQ ID NO: 27. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 96% identity with SEQ ID NO: 27. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 97% identity with SEQ ID NO: 27.
- the nucleic acid has a nucleotide sequence that shares at least 98% identity with SEQ ID NO: 27. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 99% identity with SEQ ID NO: 27. In some embodiments, the nucleic acid has a nucleotide sequence that shares less than 100% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has the nucleotide sequence of SEQ ID NO: 27.
- the present disclosure provides a conjugated protein comprising an engineered boIFNz.3 protein, such as a boIFN/3 protein variant, and a moiety that is capable of extending the engineered boIFNZ.3 protein elimination half-life.
- the engineered boIFNZ.3 protein of said protein conjugate is characterized as having an amino acid sequence comprising a non-natural amino acid.
- the moiety that is capable of extending the engineered boIFNZ.3 protein elimination half-life is conjugated to the protein via the non-natural amino acid.
- the conjugated protein is characterized as having an elimination half-life.
- the conjugated protein elimination half-life is extended (i.e., longer) than the half-life of the corresponding unconjugated engineered boIFNz.3 protein, or a comparable unconjugated protein, such as another unconjugated IFN protein. In some embodiments, the conjugated protein elimination half-life is extended (i.e., longer) than the half-life of unconjugated wild-type boIFNz.3 protein, such as the wild-type boIFNz.3 protein having the amino acid sequence of SEQ ID NO: 1.
- a moiety of the present disclosure that is capable of extending protein elimination halflife is capable of modifying one or more characteristics of a biotherapeutic agent relative to its unconjugated form, including but not limited to its half-life for elimination from the body or a compartment thereof.
- conjugation of a biotherapeutic agent with such a moiety can modify characteristics including, but not limited to, its pharmacodynamics, pharmacokinetics, water solubility, serum half-life, plasma half-life, therapeutic half-life, bioavailability, renal clearance, biological activity, circulation time, immunogenicity, allergenicity, physical association characteristics including, but not limited to, aggregation and multimer formation, receptor binding, binding to one or more binding partners, and/or receptor dimerization or multimerization.
- characteristics including, but not limited to, its pharmacodynamics, pharmacokinetics, water solubility, serum half-life, plasma half-life, therapeutic half-life, bioavailability, renal clearance, biological activity, circulation time, immunogenicity, allergenicity, physical association characteristics including, but not limited to, aggregation and multimer formation, receptor binding, binding to one or more binding partners, and/or receptor dimerization or multimerization.
- conjugation of a biotherapeutic agent such as a boIFNZ.3 protein variant
- a moiety that is capable of extending its half-life can provide the agent with improved pharmacodynamics, improved pharmacokinetics, increased water solubility, increased or modulated serum half-life, increased or modulated plasma half-life, increased or modulated therapeutic half-life, increased bioavailability, reduced or modulated renal clearance, modulated biological activity, extended circulation time, reduced or modulated immunogenicity, reduced or modulated allergenicity, modulated physical association characteristics including, but not limited to, aggregation and multimer formation, altered receptor binding, altered binding to one or more binding partners, and/or altered receptor dimerization or multimerization, relative to its unconjugated form.
- a moiety that is capable of extending boIFNZ.3 protein elimination half-life is a water-soluble polymer.
- HESylation involves coupling a biotherapeutic with hydroxyalkylstarch, and in particular, hydroxy ethyl starch (HES).
- HES hydroxy ethyl starch
- Disclosures related to HESylation technology include U.S. Patent Application Publication Numbers US20050063943, US20060121073, US20010100163, US20050234230, US20050238723, US20060019877, US20070134197, US20070087961 and U.S. Patent No.
- PEGylation involves coupling a biological macromolecule, such as a biotherapeutic agent, with polyethylene glycol (PEG) or a derivative thereof (e.g., see Harris J.M. and Chess R.B. (2003) Nature Reviews Drug Discovery, 2:214-221; Roberts M.J. and Harris J.M (2002) Adv. Drug Delivery Reviews, 54(4):459-476).
- PEG polyethylene glycol
- a second and equally important complication of existing methods for protein PEGylation is that the PEG derivatives can undergo undesired side reactions with residues other than those desired.
- the PEG derivatives directed at the epsilon -NH2 group of lysine also react with cysteine, histidine or other residues. This can create complex, heterogeneous mixtures of PEG-derivatized bioactive molecules at the risk of attenuating or even destroying the desired activity of the bioactive molecule.
- considerable effort has been directed toward the development of activated PEG reagents that target other amino acid side chains, including cysteine and histidine, or the N-terminus (see, e.g., U.S. Pat.
- a cysteine residue can be introduced site-selectively into the structure of proteins using site-directed mutagenesis and other techniques known in the art, and the resulting free sulfhydryl moiety can be reacted with PEG derivatives that bear thiol-reactive functional groups.
- This approach is complicated, however, in that the introduction of a free sulfhydryl group can complicate the expression, folding and stability of the resulting protein.
- biotherapeutic proteins it would be desirable to have a means to site-specifically and predictably introduce chemical functional groups into biotherapeutic proteins, thereby enabling the selective conjugation of the therapeutic protein with one or more moieties that are capable of extending its elimination half-life (and/or capable of modulating other characteristics, as disclosed herein) while simultaneously being compatible with (i.e., not engaging in undesired side reactions with) sulfhydryls, epsilon amino groups and other chemical functional groups that are present in the protein.
- compositions comprising a boIFNz.3 protein variant site-specifically conjugated to a moiety that is capable of extending boIFNZ.3 protein elimination half-life.
- the moiety is site-specifically conjugated to the boIFNZ.3 protein variant via a non-natural amino acid in the protein sequence.
- the non-natural amino acid contains chemical functional groups that are inert to the functional groups found in the 20 canonical amino acids but react cleanly and efficiently with other functional groups, such as those present in water-soluble polymers and derivatives thereof, as provided herein, to form stable linkages in a site-specific manner.
- the moiety that is capable of extending boIFNz.3 protein elimination half-life is one or more moieties
- the non-natural amino acid is one or more non-natural amino acids
- each of the one or more moieties is conjugated to the boIFNz.3 protein variant via each of the one or more non-natural amino acids.
- the one or more moieties is one moiety
- the one or more non-natural amino acids is one non-natural amino acid.
- the one, or the one or more, non-natural amino acids is a non-naturally encoded amino acid.
- the non-natural amino acid is para-acetyl-L- phenylalanine.
- the non-natural amino acid is para-azido-L- phenylalanine.
- the moiety that is capable of extending the boIFNZ.3 protein elimination halflife is a water-soluble polymer.
- the present disclosure provides for boIFNz.3 protein variant site-specifically conjugated to one or more water-soluble polymers via one or more non-natural amino acids in the protein sequence.
- the one or more water-soluble polymers is one water-soluble polymer, and the one or more non-natural amino acids is one non- natural amino acid.
- Water-soluble polymers of the present disclosure include, but are not limited to, polyethylene glycol (PEG), polyethylene glycol propionaldehyde, mono C1-C10 alkoxy or aryloxy derivatives thereof (described in U.S. PatentNo.
- the present disclosure includes derivatives of any of the foregoing water- soluble polymers.
- Such water-soluble polymer derivatives include water-soluble polymers comprising reactive group(s) that are capable of reacting with complementary reactive groups present in non-natural amino acids of the present disclosure.
- a water- soluble polymer derivative of the present disclosure includes PEG derivatives comprising reactive aminooxy groups that are capable of reacting with carbonyl groups of non-natural amino acids (e.g., acyl groups of the non-natural amino acid pAF).
- Water-soluble polymers and their derivatives may or may not have their own biological activity.
- boIFNZ.3 protein variants conjugated to water-soluble polymer(s) including polyalkylene glycols and derivatives thereof include polyalkylene glycols and derivatives thereof.
- polyalkylene glycols include, but are not limited to, polyethylene glycol (PEG), polypropylene glycol, polybutylene glycol, and derivatives thereof.
- PEG polyethylene glycol
- Other exemplary embodiments are listed, for example, in commercial supplier catalogs, such as Shearwater Corporation's catalog “Polyethylene Glycol and Derivatives for Biomedical Applications” (2001).
- such polyalkylene glycols can have average molecular weights within a range of about 0.1 kDa to about 100 kDa.
- such polyalkylene glycols include, but are not limited to, polyalkylene glycols having a molecular weight within a range of about 100 Da to about 100,000 Da or more.
- the molecular weight (which can be an average molecular weight) of a polyalkylene glycol of the present disclosure may be within a range of about 100 Da to about 100,000 Da, including but not limited to, about 100,000 Da, about 95,000 Da, about 90,000 Da, about 85,000 Da, about 80,000 Da, about 75,000 Da, about 70,000 Da, about 65,000 Da, about 60,000 Da, about 55,000 Da, about 50,000 Da, about 45,000 Da, about 40,000 Da, about 35,000 Da, about 30,000 Da, about 25,000 Da, about 20,000 Da, about 15,000 Da, about 10,000 Da, about 9,000 Da, about 8,000 Da, about 7,000 Da, about 6,000 Da, about 5,000 Da, about 4,000 Da, about 3,000 Da, about 2,000 Da, about 1,000 Da, about 900 Da, about 800 Da, about 700 Da, about
- the molecular weight of the polyalkylene glycol is within a range of about 100 Da to about 50,000 Da. In some embodiments, the molecular weight of the polyalkylene glycol is within a range of about 100 Da to about 40,000 Da. In some embodiments, the molecular weight of the poly alkylene glycol is within a range of about 1,000 Da to about 40,000 Da. In some embodiments, the molecular weight of the polyalkylene glycol is within a range of about 2,000 to about 50,000 Da. In some embodiments, the molecular weight of the polyalkylene glycol is within a range of about 5,000 Da to about 40,000 Da.
- the molecular weight of the polyalkylene glycols is within a range of about 10,000 Da to about 40,000 Da. In some embodiments, the molecular weight of the polyalkylene glycol is at most about 100 kDa. In some embodiments, the molecular weight of the polyalkylene glycol is at most about 50 kDa. In some embodiments, the molecular weight of the polyalkylene glycol is at least about 0.1 kDa and at most about 100 kDa. In some embodiments, the molecular weight of the polyalkylene glycol is within a range of about 10 kDa to about 50 kDa.
- the molecular weight of the polyalkylene glycol is within a range of about 20 kDa to about 40 kDa. In some embodiments, the molecular weight of the polyalkylene glycol is within a range of about 25 kDa to about 35 kDa. In some embodiments, the molecular weight of the polyalkylene glycol is about 25 kDa, about 26 kDa, about 27 kDa, about 28 kDa, about 29 kDa, about 30 kDa, about 31 kDa, about 32, kDa, about 33 kDa, about 34 kDa or about 35 kDa.
- the molecular weight of the polyalkylene glycol is about 30 kDa.
- the polyalkylene glycol is a linear chain polyalkylene glycol.
- the polyalkylene glycol is a branched chain polyalkylene glycol.
- the present disclosure includes derivatives of any of the foregoing polyalkylene glycols.
- polyalkylene glycol derivatives include polyalkylene glycol derivatives comprising reactive group(s) that are capable of reacting with complementary reactive groups present in non-natural amino acids present in proteins of the present disclosure.
- a polyalkylene glycol derivative includes polyalkylene glycol derivatives comprising reactive aminooxy groups that are capable of reacting with carbonyl groups of non-natural amino acids (e.g., acyl groups of the non-natural amino acid pAF) present in proteins of the present disclosure.
- the present disclosure provides a boIFNZ.3 protein variant conjugated to a water-soluble polymer via a non-natural amino acid, wherein the water-soluble polymer is a polyalkylene glycol derivative, wherein, prior to the conjugation with the boIFNZ.3 protein variant, said polyalkylene glycol derivative comprises an aminooxy group.
- the non-natural amino acid is pAF
- the covalent linkage comprises an oxime.
- the non-natural amino acid is pAF
- the covalent linkage consists of an oxime.
- boIFNZ.3 protein variant conjugated to a water-soluble polymer via a non-natural amino acid wherein the water-soluble polymer is a polyalkylene glycol derivative, wherein, prior to the conjugation with the boIFNZ.3 protein variant, said polyalkylene glycol derivative comprises an azide group.
- the non-natural amino acid that reacts with the azide group comprises an alkynyl group, such as a cyclooctynyl group.
- the non-natural amino acid comprises the azide
- the water-soluble polymer comprises the alkyne.
- the non-natural amino acid comprising the azide is p-azido-L-phenylalanine.
- a boIFNZ.3 protein variant of the present disclosure is conjugated to a polyalkylene glycol or derivatives thereof, wherein the polyalkylene glycol is a polyethylene glycol (PEG) or a derivative thereof.
- PEGs include, but are not limited to, PEGs having an average molecular weight within a range of about 100 Da to about 100,000 Da or more.
- the molecular weight (which can be an average molecular weight) of a PEG of the present disclosure may be within a range of about 100 Da to about 100,000 Da, including but not limited to, about 100,000 Da, about 95,000 Da, about 90,000 Da, about 85,000 Da, about 80,000 Da, about 75,000 Da, about 70,000 Da, about 65,000 Da, about 60,000 Da, about 55,000 Da, about 50,000 Da, about 45,000 Da, about 40,000 Da, about 35,000 Da, about 30,000 Da, about 25,000 Da, about 20,000 Da, about 15,000 Da, about 10,000 Da, about 9,000 Da, about 8,000 Da, about 7,000 Da, about 6,000 Da, about 5,000 Da, about 4,000 Da, about 3,000 Da, about 2,000 Da, about 1,000 Da, about 900 Da, about 800 Da, about 700 Da, about 600 Da, about 500 Da, 400 Da, about 300 Da, about 200 Da or about 100 Da.
- molecular weight of the PEG is within a range of about 100 Da to about 50,000 Da. In some embodiments, the molecular weight of the PEG is within a range of about 100 Da to about 40,000 Da. In some embodiments, the molecular weight of the PEG is within a range of about 1,000 Da to about 40,000 Da. In some embodiments, the molecular weight of the PEG is within a range of about 2,000 to about 50,000 Da. In some embodiments, the molecular weight of the PEG is within a range of about 5,000 Da to about 40,000 Da. In some embodiments, the molecular weight of the PEG is within a range of about 10,000 Da to about 40,000 Da.
- the molecular weight of the PEG is at most about 100 kDa. In some embodiments, the molecular weight of the PEG is at most about 50 kDa. In some embodiments, the molecular weight of the PEG is at least about 0.1 kDa and at most about 100 kDa. In some embodiments, the molecular weight of the PEG is within a range of about 10 kDa to about 50 kDa. In some embodiments, the molecular weight of the PEG is within a range of about 20 kDa to about 40 kDa. In some embodiments, the molecular weight of the PEG is within a range of about 25 kDa to about 35 kDa.
- the molecular weight of the PEG is about 25 kDa, about 26 kDa, about 27 kDa, about 28 kDa, about 29 kDa, about 30 kDa, about 31 kDa, about 32, kDa, about 33 kDa, about 34 kDa or about 35 kDa. In some embodiments, the molecular weight of the PEG is about 30 kDa. In some embodiments, there is provided any one of the foregoing PEGs, wherein the PEG is a linear chain PEG. In some other embodiments, there is provided any one of the foregoing PEGs, wherein the PEG is a branched chain PEG.
- any one of the foregoing PEGs wherein the PEG is a multiarm PEG.
- the present disclosure includes derivatives of any of the foregoing PEGs.
- PEG derivatives include PEG derivatives comprising reactive group(s) that are capable of reacting with complementary reactive groups present in non-natural amino acids present in proteins of the present disclosure.
- a PEG derivative of the present disclosure includes PEG derivatives comprising reactive aminooxy groups that are capable of reacting with carbonyl groups of non-natural amino acids (e.g., acyl groups of the non-natural amino acid pAF) present in proteins of the present disclosure.
- Activated and functionalized PEGs can be obtained from commercial suppliers such as Biopharma PEG (Biochempeg) Scientific Inc., Watertown, MA and MilliporeSigma, Burlington, MA.
- the present disclosure provides boIFNZ.3 protein variant conjugated to a water-soluble polymer via a non-natural amino acid, wherein the water-soluble polymer is a PEG derivative, wherein, prior to the conjugation with the boIFN/3 protein variant, said PEG derivative comprises an aminooxy group.
- the non-natural amino acid is pAF
- the covalent linkage comprises an oxime.
- the non-natural amino acid is pAF, and upon conjugation with the PEG derivative, the covalent linkage consists of an oxime.
- the present disclosure provides a conjugated protein comprising: an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid; and a water-soluble polymer; wherein the water-soluble polymer is conjugated to the engineered boIFNZ.3 protein.
- the water-soluble polymer is conjugated to the engineered boIFNz.3 protein via the non-natural amino acid.
- the water- soluble polymer is conjugated to the engineered boIFNz.3 protein via a covalent linkage with the non-natural amino acid.
- the water-soluble polymer is one or more water-soluble polymers
- the non-natural amino acid is one or more non-natural amino acids
- each of the one or more water-soluble polymers is conjugated to the engineered boIFNz.3 protein via each of the one or more non-natural amino acids.
- the one or more water-soluble polymers is one water-soluble polymer
- the one or more non-natural amino acids is one non- natural amino acid.
- each of the one or more water-soluble polymers is independently selected from the group consisting of a carbohydrate, a polypeptide and a polyalkylene glycol; and derivatives thereof.
- each water-soluble polymer is a polyalkylene glycol or a derivative thereof.
- the polyalkylene glycol, or the derivative, thereof is a linear polyalkylene glycol, or a derivative thereof.
- the polyalkylene glycol, or the derivative, thereof is a branched polyalkylene glycol, or a derivative thereof.
- the polyalkylene glycol, or the derivative, thereof is a multiarm polyalkylene glycol, or a derivative thereof.
- the polyalkylene glycol is a polyalkylene glycol derivative, wherein, prior to the conjugation with the engineered boIFNz.3 protein, the polyalkylene glycol derivative comprises a group that is capable of forming an oxime. In some embodiments, the polyalkylene glycol is a polyalkylene glycol derivative, wherein, prior to the conjugation with the engineered boIFNZ.3 protein, the polyalkylene glycol derivative comprises an aminooxy group. In some embodiments, the covalent linkage conjugating the engineered boIFNZ.3 protein with the polyalkylene glycol derivative comprises an oxime. In some embodiments, the covalent linkage conjugating the engineered boIFNZ.3 protein with the polyalkylene glycol derivative consists of an oxime.
- the polyalkylene glycol or the derivative thereof has a molecular weight of at least about 0.1 kDa. In some embodiments, the polyalkylene glycol or the derivative thereof has a molecular weight of at most about 100 kDa. In some embodiments, the polyalkylene glycol or the derivative thereof has a molecular weight of at most about 50 kDa.
- the polyalkylene glycol or derivative thereof is a PEG, or a derivative thereof.
- the PEG or the derivative thereof is a linear PEG or a derivative thereof.
- the PEG is a PEG derivative, wherein, prior to the conjugation with the engineered boIFNZ.3 protein, the PEG derivative comprises a group that is capable of forming an oxime.
- the PEG is a PEG derivative, wherein, prior to the conjugation with the engineered boIFNZ.3 protein, the PEG derivative comprises an aminooxy group.
- the covalent linkage conjugating the engineered boIFNZ.3 protein with the PEG derivative comprises, or consists of, an oxime.
- the conjugated protein comprises a non-natural amino acid selected from the group consisting of 4-acetyl-L-phenylalanine (para-acetyl-L-phenylalanine (pAF)), 3-O-(N-acetyl-beta-D-glucosaminyl)-L-threonine, N4-(P-N-Acetyl-D-glucosaminyl)-L- asparagine, O-allyl-L-tyrosine, alpha-N-acetylgalactosamine-O-L-serine, alpha-N- acetylgalactosamine-O-L-threonine, 2-aminooctanoic acid, 2-amino-L-phenylalanine, 3-amino-L- phenylal anine, 4-amino-L-phenylalanine, 2-amino-L-tyrosine, 3-amino
- the engineered boIFNz.3 protein comprising a non-natural amino acid is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA synthetase.
- the non-natural amino acid is site-specifically incorporated.
- a water-soluble polymer is conjugated to an engineered boIFNz.3 protein of the present disclosure by reacting a water-soluble polymer containing a reactive group with the engineered boIFNz.3 protein containing one or more natural or non-natural amino acids.
- the conjugation reaction provides a protein conjugate, wherein the water-soluble polymer is conjugated to a natural or non-natural amino acid of the engineered boIFNz.3 protein via a covalent linkage.
- the covalent linkage can be a product of the reactive group of the water-soluble polymer and an additional moiety present in the natural or non-natural amino acid, wherein the additional moiety can react to form the covalent linkage with the reactive group of the water-soluble polymer.
- Non-limiting examples of reactions and linkages formed between water-soluble polymers and natural or non-natural amino acids that are present in an engineered boIFNz.3 protein of the present disclosure include those disclosed in the following paragraphs A to I.
- A. (i) Reaction of a water-soluble polymer comprising reactive group -N3 with a boIFNk3protein, wherein the protein contains a non-natural amino acid comprising an alkynyl group, thereby providing a linkage comprising a 1,2,3-triazolyl moiety; or (ii) reaction of a water- soluble polymer comprising a reactive alkynyl group with a boIFNZ.3 protein, wherein the protein contains a non-natural amino acid comprising -N3, thereby providing a linkage comprising a 1,2,3- triazolyl moiety.
- the alkynyl group is a cyclooctynyl group.
- the non-natural amino acid is p-azido-L-phenylalanine.
- the linkage comprising the 1,2,3-triazolyl moiety has the following structure: wherein: each s is independently 0 or an integer from 1 to 50; each t is independently 0 or an integer from 1 to 50; each + denotes connection to the water-soluble polymer; and each wavy line denotes connection to the protein.
- each s is independently 0, 1, 2, 3, 4, 5 or 6.
- each t is independently 0, 1, 2, 3, 4, 5 or 6.
- B (i) Reaction of a water-soluble polymer comprising a reactive tetrazinyl group with a boIFNZ.3 protein, wherein the protein contains a non-natural amino acid comprising an (E)- cyclooctenyl group, thereby providing a linkage comprising a 1,4-dihydropyridazinyl moiety; or (ii) reaction of a water-soluble polymer comprising a reactive (E)-cyclooctenyl group with a boIFNZ.3 protein, wherein the protein contains a non-natural amino acid comprising a tetrazinyl group, thereby providing a linkage comprising a 1,4-dihydropyridazinyl moiety.
- the linkage comprising the 1,4-dihydropyridazinyl moiety has the following structure: wherein: each Rf is independently H or alkyl; each + denotes connection to a water-soluble polymer; and each wavy line denotes connection to the protein. In some embodiments, each Rf is independently unsubstituted C1-C6 alkyl.
- the carbonyl or ketone group is -C(O)Rc, wherein Rc is unsubstituted C1-C6 alkyl.
- Rc is methyl.
- the linkage comprising the oxime moiety has the following structure: wherein: each Rc is independently unsubstituted C1-C6 alkyl; each + denotes connection to a water-soluble polymer; and each wavy line denotes connection to the protein. In some embodiments, each Rc is methyl.
- the natural amino acid is cysteine.
- the linkage comprising the a pyrrolidine- 2, 5-dione moiety, such as a 3-(kl-sulfaneyl)pyrrolidine-2, 5-dione moiety has the following structure: wherein: each + denotes connection to a water-soluble polymer; and each wavy line denotes connection to the protein.
- the natural amino acid is aspartic acid or glutamic acid. In some other embodiments, the natural amino acid is lysine.
- the reaction is a peptide coupling reaction or other well-known method of forming an amide, each of which can be performed using methods readily understood by a person of ordinary skill in the art.
- the linkage comprising the amide moiety has the following structure: wherein: each Rb is independently H or alkyl; each + denotes connection to a water-soluble polymer; and each wavy line denotes connection to the protein. In some embodiments, each Rb is independently alkyl, wherein the alkyl is unsubstituted C1-C6 alkyl.
- the natural amino acid is aspartic acid or glutamic acid. In some other embodiments, the natural amino acid is serine, threonine or tyrosine. Methods of forming such esters linkages can be performed using methods readily understood by a person of ordinary skill in the art.
- the linkage comprising the ester moiety has the following structure: wherein: each + denotes connection to a water-soluble polymer; and each wavy line denotes connection to the protein.
- G (i) Reaction of a water-soluble polymer comprising a thiol group (-SH) with a boIFNZ.3 protein, wherein the protein contains a natural or non-natural amino acid comprising a carboxylic acid group, a protected carboxylic acid, or an activated ester group, thereby providing a linkage comprising a thioester moiety; or (ii) reaction of a water-soluble polymer comprising a carboxylic acid group, a protected carboxylic acid, or an activated ester group with a boIFNZ.3 protein, wherein the protein contains a natural or non-natural amino acid comprising a thiol group, thereby providing a linkage comprising a thioester moiety.
- the natural amino acid is aspartic acid or glutamic acid. In some other embodiments, the natural amino acid is cysteine. Methods of forming such thioesters linkages can be performed using methods readily understood by a person of ordinary skill in the art. In some embodiments, the linkage comprising the ester moiety has the following structure: wherein: each + denotes connection to a water-soluble polymer; and each wavy line denotes connection to the protein.
- the natural amino acid is aspartic acid or glutamic acid.
- Methods of forming such linkages can be performed using methods readily understood by a person of ordinary skill in the art.
- the linkage has the following structure: wherein: each + denotes connection to water-soluble polymer; and each wavy line denotes connection to the protein.
- the conjugated protein of the present disclosure comprises a covalent linkage that joins an engineered boIFN/3 protein to a water-soluble polymer.
- the covalent linkage comprises an amide, an ester, a thioester, a disulfide, an oxime, an imine, a pyrrolidine-2, 5-dione, a 1,2, 3 -triazole or a 1,4-dihydropyridazine.
- the 1,2,3-triazole is fused to an 8-membered ring.
- the 1,4- dihydropyridazine is fused to an 8-membered ring.
- the covalent linkage that joins the engineered boIFNZ.3 protein to the water-soluble polymer comprises, or consists of, one of the following groups:
- the covalent linkage comprises an oxime. In some embodiments, the covalent linkage consists of an oxime. In some embodiments, the covalent linkage has the following structure: wherein + denotes the connection to the water-soluble polymer; the wavyline denotes connection to the engineered boIFNZ.3 protein; and Rc is unsubstituted Ci-Ce alkyl. In some embodiments, Rc is methyl. Rc can be the methyl group of the non-natural amino acid pAF that is present in the engineered boIFNZ.3 protein amino acid sequence.
- the present disclosure also provides a protein conjugate comprising a boIFNZ.3 protein or variant thereof, wherein the boIFNZ.3 protein or variant thereof comprises an unusual genetically-encoded amino acid that is not present in humans.
- unusual genetically-encoded amino acid that is not present in humans is selenocysteine.
- unusual genetically-encoded amino acid that is not present in humans is pyrrolysine.
- the boIFNZ.3 protein or variant thereof comprising the unusual genetically encoded amino acid is conjugated to a water-soluble polymer via the unusual genetically encoded amino acid.
- the boIFNZ.3 protein or variant thereof comprising the unusual genetically encoded amino acid conjugated to the water-soluble polymer via the unusual genetically encoded amino acid, does not contain a non-natural amino acid.
- the water-soluble polymer can be a PEG or a derivative thereof.
- a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein having an amino acid sequence disclosed in Table 1, wherein the amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 1, wherein each said amino acid sequence optionally further comprises an N-terminal methionine.
- the engineered boIFNZ.3 protein amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the conjugated protein comprises an engineered boIFNz.3 protein, wherein the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 1 wherein at least one amino acid of SEQ ID NO: 1 is replaced with a different amino acid.
- the different amino acid is a non-natural amino acid.
- the non-natural amino acid is pAF.
- one amino acid of SEQ ID NO: 1 is replaced with one non-natural amino acid.
- the one non-natural amino acid is pAF.
- a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein having an amino acid sequence that shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 1, optionally wherein the engineered boIFNz.3 protein amino acid sequence further comprises an N-terminal methionine.
- the engineered boIFNz.3 protein amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
- the engineered boIFNZ.3 protein amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with a sequence listed in Table 1, wherein X is para-acetyl -L-phenylalanine (pAF).
- pAF para-acetyl -L-phenylalanine
- a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein, wherein the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 1, wherein one or more amino acid of SEQ ID NO: 1 is replaced with a different amino acid.
- the different amino acid is a non-natural amino acid.
- the non-natural amino acid is pAF.
- one amino acid of SEQ ID NO: 1 is replaced with one non-natural amino acid.
- the one non-natural amino acid is pAF.
- a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein having an amino acid sequence disclosed in Table 1, wherein the amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with (i) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; or (ii) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N- terminal methionine; and wherein the engineered boIFNz.3 protein amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, each of SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13 has one or more non-natural amino acid incorporated.
- a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein having an amino acid sequence that shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the engineered boIFNz.3 protein amino acid sequence shares less than 100% identity with SEQ ID NO: 1, and optionally wherein the engineered boIFNz.3 protein amino acid sequence further comprises an N-terminal methionine.
- the engineered boIFNZ.3 protein has an amino acid sequence in which one or more non-natural amino acid is incorporated e.g., has the amino acid sequence of any of SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13 in which one or more non-natural amino acid is incorporated.
- the one or more non-natural amino acid is pAF.
- the engineered boIFNz.3 protein has the amino acid sequence of any of SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13 in which one non-natural amino acid incorporated.
- the one non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 90% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24 further comprises an N-terminal methionine.
- a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine.
- the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
- the amino acid sequence shares at least 90% identity with a sequence listed in Table 1, wherein X is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 90% identity with SEQ ID NO: 18.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 90% identity with SEQ ID NO: 19.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 95% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- a conjugated protein of the present disclosure can comprise an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 95% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 95% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine.
- the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
- the amino acid sequence shares at least 95% identity with a sequence listed in Table 1, wherein X is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 95% identity with SEQ ID NO:
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 95% identity with SEQ ID NO:
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 96%, at least 97% or at least 98% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- a conjugated protein of the present disclosure can comprise an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid and having (i) an amino acid sequence that shares at least 96% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) an amino acid sequence that shares at least 96% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- a conjugated protein of the present disclosure can comprise an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 96% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 96% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine.
- the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
- the amino acid sequence shares at least 96% identity with a sequence listed in Table 1, wherein X is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 96% identity with SEQ ID NO:
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 96% identity with SEQ ID NO:
- a conjugated protein of the present disclosure can comprise an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid and having (i) an amino acid sequence that shares at least 97% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) an amino acid sequence that shares at least 97% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- a conjugated protein of the present disclosure can comprise an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid and having (i) an amino acid sequence that shares at least 97% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) an amino acid sequence that shares at least 97% identity with SEQ ID NO: 14, 15, 16, 17, 18,
- the engineered boIFNZ.3 protein further comprises an N-terminal methionine.
- the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
- the amino acid sequence shares at least 97% identity with a sequence listed in Table 1, wherein X is pAF.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g. an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 97% identity with SEQ ID NO: 18.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 97% identity with SEQ ID NO: 19.
- an engineered boIFNz.3 protein having an amino acid sequence e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid
- a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid and having (i) an amino acid sequence that shares at least 98% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) an amino acid sequence that shares at least 98% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid and having (i) an amino acid sequence that shares at least 98% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) an amino acid sequence that shares at least 98% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine.
- the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
- the amino acid sequence shares at least 98% identity with a sequence listed in Table 1, wherein X is pAF.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 98% identity with SEQ ID NO: 18.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 98% identity with SEQ ID NO: 19.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
- a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid and having (i) an amino acid sequence that shares at least 99% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) an amino acid sequence that shares at least 99% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine.
- the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
- the amino acid sequence shares at least 99% identity with a sequence listed in Table 1, wherein X is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 14.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 14, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 14, wherein the sequence optionally further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 14.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 14, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 14, and wherein the engineered boIFNz.3 protein optionally further comprises an N- terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 14.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 14, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine.
- the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 15.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 15, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 15, wherein the sequence optionally further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 15.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 15, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 15, and wherein the engineered boIFNz.3 protein optionally further comprises an N- terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 15.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 15, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine.
- the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 16.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 16, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 16, wherein the sequence optionally further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 16.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 16, and wherein the engineered boIFN/3 protein further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non -natural amino acid) that shares at least 99% identity with SEQ ID NO: 16, and wherein the engineered boIFNz.3 protein optionally further comprises an N- terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non -natural amino acid) that shares at least 99% identity with SEQ ID NO: 16.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 16, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine.
- the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 17.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 17, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 17, wherein the sequence optionally further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 17.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 17, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNZ.3 protein having an amino acid sequence comprising a non -natural amino acid) that shares at least 99% identity with SEQ ID NO: 17, and wherein the engineered boIFNX3 protein optionally further comprises an N- terminal methionine.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNZ.3 protein having an amino acid sequence comprising a non -natural amino acid) that shares at least 99% identity with SEQ ID NO: 17.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 17, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine.
- the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 18.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 18.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 18.
- the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 19.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 19.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 19.
- the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 20.
- the conjugated protein comprises an engineered boIFNX3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 20, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 20, wherein the sequence optionally further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 20.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 20, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 20, and wherein the engineered boIFNz.3 protein optionally further comprises an N- terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 20.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 20, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine.
- the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 21.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 21, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 21, wherein the sequence optionally further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 21.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 21, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non -natural amino acid) that shares at least 99% identity with SEQ ID NO: 21, and wherein the engineered boIFNz.3 protein optionally further comprises an N- terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non -natural amino acid) that shares at least 99% identity with SEQ ID NO: 21.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 21, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine.
- the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 22.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 22, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 22, wherein the sequence optionally further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 22.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 22, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 22, and wherein the engineered boIFNZ.3 protein optionally further comprises an N- terminal methionine.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNZ.3 protein having an amino acid sequence comprising a non -natural amino acid) that shares at least 99% identity with SEQ ID NO: 22.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO:
- the engineered boIFNZ.3 protein further comprises an N-terminal methionine.
- the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 23.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 23, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 23, wherein the sequence optionally further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 23.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 23, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 23, and wherein the engineered boIFNZ.3 protein optionally further comprises an N- terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 23.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO:
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 24.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 24, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 24, wherein the sequence optionally further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 24.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 24, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 24, and wherein the engineered boIFNZ.3 protein optionally further comprises an N- terminal methionine.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 24.
- the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 24, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine.
- the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
- the conjugated protein comprises an engineered boIFNz.3 protein having the amino acid sequence of SEQ ID NO: 14 comprising a non-natural amino acid.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNz.3 protein having the amino acid sequence of SEQ ID NO: 14 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 15 comprising a non-natural amino acid.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 15 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 16 comprising a non-natural amino acid.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 16 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 17 comprising a non-natural amino acid.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 17 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 18 comprising a non-natural amino acid.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence of SEQ ID NO: 19 comprising a non-natural amino acid.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 20 comprising a non-natural amino acid.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 20 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 21 comprising a non-natural amino acid.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 21 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 22 comprising a non-natural amino acid.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 22 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 23 comprising a non-natural amino acid.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 23 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 24 comprising a non-natural amino acid.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 24 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 25 comprising a non-natural amino acid.
- the non-natural amino acid is pAF.
- the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 25 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine.
- the non-natural amino acid is pAF.
- the non-natural amino acid in the boIFNz.3 protein may be pAF.
- the present disclosure provides a conjugated protein comprising: an engineered boIFNz.3 protein having an amino acid sequence comprising a non- natural amino acid; and a water-soluble polymer; wherein: the water-soluble polymer is a polyethylene glycol (PEG) or a derivative thereof, which is conjugated to the engineered boIFNz.3 protein via a covalent linkage with the non-natural amino acid; and the engineered boIFNz.3 protein amino acid sequence shares at least 90% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 18.
- PEG polyethylene glycol
- the engineered boIFNz.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 18.
- the engineered boIFNz.3 protein amino acid sequence shares at least 96% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 97% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 98% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFN 3 protein has the amino acid sequence of SEQ ID NO: 18. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine.
- the engineered boIFNz.3 protein is an isolated protein. In some embodiments, the engineered boIFNz.3 protein is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA synthetase.
- the non-natural amino acid is site-specifically incorporated and non-naturally encoded.
- the PEG or the derivative thereof has a molecular weight of at least about 0.1 kDa and at most about 100 kDa. In some embodiments, the PEG or the derivative thereof has a molecular weight of at most about 50 kDa.
- the PEG or the derivative thereof has a molecular weight within a range of about 10 kDa to about 50 kDa, about 20 kDa to about 40 kDa, or about 25 kDa to about 35 kDa. In some embodiments, the PEG or the derivative thereof has a molecular weight of about 30 kDa. In some embodiments, the PEG or the derivative thereof is a linear PEG or a linear PEG derivative. In some embodiments, the linear PEG derivative is a linear PEG comprising a reactive moiety that is capable of reacting with a non-natural amino acid. In some embodiments, the linear PEG derivative is a linear PEG comprising a terminal aminooxy group.
- the linear PEG derivative is a linear PEG comprising a terminal alkyne. In some embodiments, the linear PEG derivative is a linear PEG comprising a terminal azido group. In some embodiments, the PEG is a PEG derivative, such as a linear PEG derivative, wherein, prior to the conjugation with the engineered boIFNz.3 protein, the PEG derivative comprises an aminooxy group. In some embodiments, the covalent linkage conjugating the engineered boIFNz.3 protein with the PEG derivative comprises, or consists of, an oxime. A conjugated boIFNz.3 protein of the present disclosure has an extended elimination half-life in vivo, as further disclosed herein.
- the present disclosure provides a conjugated protein comprising: an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid; and a water-soluble polymer; wherein: the water-soluble polymer is a polyethylene glycol (PEG) or a derivative thereof, which is conjugated to the engineered boIFNz.3 protein via a covalent linkage with the non-natural amino acid; and the engineered boIFNz.3 protein amino acid sequence shares at least 90% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 19.
- PEG polyethylene glycol
- the engineered boIFNz.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 19.
- the engineered boIFNz.3 protein amino acid sequence shares at least 96% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 97% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 98% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 19. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine.
- the engineered boIFNz.3 protein is an isolated protein. In some embodiments, the engineered boIFNz.3 protein is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA synthetase.
- the non-natural amino acid is site-specifically incorporated and non-naturally encoded.
- the PEG or the derivative thereof has a molecular weight of at least about 0.1 kDa and at most about 100 kDa. In some embodiments, the PEG or the derivative thereof has a molecular weight of at most about 50 kDa.
- the PEG or the derivative thereof has a molecular weight within a range of about 10 kDa to about 50 kDa, about 20 kDa to about 40 kDa, or about 25 kDa to about 35 kDa. In some embodiments, the PEG or the derivative thereof has a molecular weight of about 30 kDa. In some embodiments, the PEG or the derivative thereof is a linear PEG or a linear PEG derivative. In some embodiments, the linear PEG derivative is a linear PEG comprising a reactive moiety that is capable of reacting with a non-natural amino acid. In some embodiments, the linear PEG derivative is a linear PEG comprising a terminal aminooxy group.
- the linear PEG derivative is a linear PEG comprising a terminal alkyne. In some embodiments, the linear PEG derivative is a linear PEG comprising a terminal azido group. In some embodiments, the PEG is a PEG derivative, such as a linear PEG derivative, wherein, prior to the conjugation with the engineered boIFNZ.3 protein, the PEG derivative comprises an aminooxy group. In some embodiments, the covalent linkage conjugating the engineered boIFN/3 protein with the PEG derivative comprises, or consists of, an oxime.
- a conjugated boIFNz.3 protein of the present disclosure has an extended elimination half-life in vivo, as further disclosed herein.
- the present invention provides a conjugated protein comprising: an engineered boIFNz.3 protein having an amino acid sequence comprising a nonnatural amino acid, wherein the non-natural amino acid is para-acetyl-L-phenyalanine, wherein the engineered boIFNz.3 protein has an amino acid sequence of SEQ ID NO: 18; and a water soluble polymer, wherein the water soluble polymer is PEG or a derivative thereof, wherein the PEG or derivative thereof has a molecular weight of about 30kDa, optionally wherein the PEG or derivative thereof is a linear PEG or a linear PEG derivative; wherein the water-soluble polymer is conjugated to the engineered boIFNz.3 protein via the non-natural amino acid, optionally via a covalent linkage, optionally wherein the covalent linkage comprises, or consists of, an oxime.
- the present invention provides a conjugated protein comprising: an engineered boIFNz.3 protein having an amino acid sequence comprising a non- natural amino acid, wherein the non-natural amino acid is para-acetyl-L-phenyalanine, wherein the engineered boIFNz.3 protein has an amino acid sequence of SEQ ID NO: 19; and a water soluble polymer, wherein the water soluble polymer is PEG or a derivative thereof, wherein the PEG or derivative thereof has a molecular weight of about 30kDa, optionally wherein the PEG or derivative thereof is a linear PEG or a linear PEG derivative; wherein the water-soluble polymer is conjugated to the engineered boIFNz.3 protein via the non-natural amino acid, optionally via a covalent linkage, optionally wherein the covalent linkage comprises, or consists of, an oxime.
- Unconjugated IFNs are generally short-lived species in circulation, with typical half-lives occurring within minutes, and less than one hour, as understood by a person skilled in the art.
- Bansal et al. have shown that IFNy plasma concentrations in mice were reduced by over 90% within about 30 minutes after administration (Bansal R. et al. (2011) J. Controlled Release 154(3):233-240; see, e.g., Bansal at Figure 4A).
- the rapid clearance of unconjugated boIFNz.3 protein has hindered efforts to induce the protective immune response that is needed to control devastating viral infections in ruminant mammals.
- the present disclosure provides conjugated proteins and uses thereof with properties that can overcome these limitations.
- the improved pharmacokinetic profiles of the conjugated boIFNZ.3 proteins of the present disclosure support their use in providing treated subjects with protection against infection by pathogenic viruses, such as those associated with BRD and FMD.
- the protection can begin prior to the onset of an adaptive immune response that occurs after the administration of a vaccine.
- An improved pharmacokinetic profile can further enable an improved dosing regimen or avoid toxic effects.
- the elimination half-life of a biotherapeutic protein can refer to the time it takes for the concentration of the protein in the blood, plasma, serum or body of a subject to be reduced by 50%. Thus, after one half-life, the concentration of the protein in the blood, plasma, serum or body of the subject, respectively, will be half of the starting concentration.
- the elimination half-life is a blood elimination half-life. In some other embodiments, the elimination half-life is a plasma elimination half-life. In some other embodiments, the elimination half-life is a serum elimination half-life. In yet some other embodiments, the elimination half-life is a whole body elimination half-life.
- elimination half-life in plasma can be measured by taking blood samples at various time points after administration of the protein (whether conjugated or unconjugated) and determining the concentration of that protein in each sample. Correlation of the plasma concentration with time allows calculation of the elimination half-life in plasma. Elimination halflife for a conjugated protein, an unconjugated protein, or both, can be evaluated for an individual subject, between individual subjects, within a group of subjects, between groups of subjects, and/or between an individual subject and a group of subjects, depending upon study design and other factors, as understood by persons of ordinary skill in the art.
- a conjugated boIFNz.3 protein of the present disclosure can have an elimination half-life that is greater than the elimination half-life of the unconjugated engineered boIFNZ.3 protein (e.g., the corresponding unconjugated boIFNZ.3 protein, or of a related unconjugated protein, such as another unconjugated IFN protein (e.g., an unconjugated IFNy or IFNZ.).
- the related unconjugated protein is wild-type boIFNz.3 protein having the amino acid sequence of SEQ ID NO: 1.
- the conjugated boIFNz.3 protein has an elimination half-life that is at least about 5% greater than the elimination half-life of the unconjugated boIFNz.3 protein (e.g., the corresponding unconjugated boIFNz.3 protein), or of the related unconjugated protein.
- the conjugated boIFNz.3 protein can have an elimination half-life that is at least about 10%, at least about 20%, at least about 30%, at least about 40%, at least about 50%, at least about 60%, at least about 70%, at least about 80%, at least about 90% or at least about 100% greater than the elimination half-life of the unconjugated boIFNz.3 protein (e.g., the corresponding unconjugated boIFNZ.3 protein), or of the related unconjugated protein.
- the unconjugated boIFNz.3 protein e.g., the corresponding unconjugated boIFNZ.3 protein
- the conjugated boIFNz.3 protein can have an elimination half-life that is at least about 5-fold, about 10-fold (1000%), about 20-fold, about 30- fold or about 40-fold greater than the elimination half-life of the unconjugated boIFNz.3 protein (e.g., the corresponding unconjugated boIFNZ.3 protein), or of the related unconjugated protein.
- the elimination half life can be in a mammal (e.g., as determined in a mammal).
- Suitable mammals include rodents (e.g., a rat, such as a Sprague- Dawley rat), ruminant mammals, cattle (e.g., bovine, bovine calf, such as Holstein Friesian Calf).
- rodents e.g., a rat, such as a Sprague- Dawley rat
- ruminant mammals e.g., cattle
- bovine, bovine calf such as Holstein Friesian Calf
- the elimination half life is a terminal half life.
- the elimination half-life can be in a Sprague-Dawley rat, e.g., after subcutaneous or another form of administration to the Sprague-Dawley rat, as disclosed in the Examples herein (e.g., as determined in the Sprague-Dawley rat).
- the elimination half-life can be in a Holstein-Friesian Calf, e.g., after subcutaneous or another form of administration to the Holstein-Friesian Calf, also disclosed in the Examples herein (e.g., as determined in the Frieisan Calf).
- the conjugated boIFNz.3 protein can have an elimination half-life in a Holstein-Friesian Calf that is at least about 50-fold greater than the elimination half-life of the corresponding unconjugated boIFNZ.3 protein, or of a related unconjugated protein. In some embodiments, the conjugated boIFNz.3 protein can have an elimination half-life in a Holstein-Friesian Calf that is at least about 60-fold greater than the elimination half-life of the corresponding unconjugated boIFNZ.3 protein, or of a related unconjugated protein.
- the conjugated boIFNz.3 protein can have an elimination half-life in a Holstein-Friesian Calf that is at least about 65-fold greater than the elimination half-life of the corresponding unconjugated boIFNZ.3 protein, or of a related unconjugated protein.
- the corresponding unconjugated boIFNZ.3 protein may in some embodiments differ from the relevant conjugated boIFNz.3 protein in that the unconjugated version does not contain a water soluble polmer as defined herein (and optionally differs from the unconjugated version only in this respect).
- the elimination half-life is a blood elimination half-life.
- the blood elimination half life is a terminal blood half life.
- the elimination half-life is a plasma elimination half-life.
- the plasma elimination half life is a terminal plasma half life.
- the elimination half-life is a serum elimination half-life.
- the serum elimination half life is a terminal serum half life.
- the elimination half-life is a half-life of elimination from the body (whole body elimination half-life).
- the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 10 hours in a rat after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein to the rat.
- the single subcutaneous dose is a dose within a range of about 0.1 mg/kg to about 10 mg/kg of the body weight of the rat.
- the half life is observed in a Sprague-Dawley rat.
- the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 10 hours in a Sprague-Dawley rat after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein of about 0.25 mg/kg (e.g., 0.25 mg/kg) to the Sprague-Dawley rat. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 15 hours in a Sprague-Dawley rat after administration of a single subcutaneous dose of the conjugated boIFNZ.3 protein of about 0.25 mg/kg (e.g., 0.25 mg/kg).
- the conjugated boIFNZ.3 protein can have a plasma elimination half-life within a range of about 15 hours to about 30 hours in a Sprague-Dawley rat after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein of about 0.25 mg/kg (e.g., 0.25 mg/kg). In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of about 16 hours, about 17 hours, about 18 hours, about 19 hours or about 20 hours in a Sprague-Dawley rat after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein of about 0.25 mg/kg (e.g., 0.25 mg/kg).
- the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 10 hours in cattle after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein to the cattle. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 20 hours in cattle after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein to the cattle. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 30 hours in cattle after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein to the cattle.
- the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 40 hours in cattle after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein to the cattle. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 50 hours in cattle after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein to the cattle. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination halflife of at least about 60 hours in cattle after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein to the cattle.
- the single subcutaneous dose is within a range of about 50 pg/kg to about 500 pg/kg of the body weight of the cattle. In some embodiments, the single subcutaneous dose is within a range of about 75 pg/kg to about 150 pg/kg of the body weight of the cattle. In some embodiments, the single subcutaneous dose is about 75 pg/kg of the body weight of the cattle. In some embodiments, the single subcutaneous dose is about 150 pg/kg of the body weight of the cattle. In some embodiments, the single subcutaneous dose is within a range of about 100 pg/kg to about 200 pg/kg of the body weight of the cattle.
- the half life is observed in a Holstein-F resian calf.
- the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 10 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 75 pg/kg to the Holstein-Fresian calf.
- the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 20 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 75 pg/kg.
- the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 30 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 40 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 50 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 75 pg/kg.
- the conjugated boIFNz.3 protein can have a plasma elimination halflife within a range of about 50 hours to about 75 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 60 hours in a Holstein- Fresian calf after administration of a single subcutaneous dose of 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 65 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 75 pg/kg.
- the conjugated boIFNz.3 protein can have a plasma elimination half-life of about 61 hours, about 62 hours, about 63 hours, about 64 hours, about 65 hours, about 66 hours, about 67 hours, about 68 hours or about 69 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 75 pg/kg.
- the conjugated boIFNX3 protein can have a plasma elimination half-life of at least about 70 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 75 pg/kg.
- the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 10 hours in a Holstein- Fresian calf after administration of a single subcutaneous dose of about 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 20 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 30 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 150 pg/kg.
- the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 40 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 50 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life within a range of about 50 hours to about 75 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 150 pg/kg.
- the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 60 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 65 hours in a Holstein- Fresian calf after administration of a single subcutaneous dose of 150 pg/kg.
- the conjugated boIFNz.3 protein can have a plasma elimination half-life of about 61 hours, about 62 hours, about 63 hours, about 64 hours, about 65 hours, about 66 hours, about 67 hours, about 68 hours or about 69 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 70 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 150 pg/kg.
- the engineered boIFNz.3 proteins and conjugated boIFNz.3 proteins of the disclosure can be employed for therapeutic uses, including but not limited to, in combination with a suitable pharmaceutical carrier.
- Such compositions can comprise a therapeutically effective amount of the engineered boIFNz.3 protein, or the conjugated boIFNz.3 protein, and a pharmaceutically acceptable carrier or excipient.
- a carrier or excipient includes, but is not limited to, saline, buffered saline, dextrose, water, glycerol, ethanol, and/or combinations thereof, or any other suitable carrier or excipient, including those further disclosed herein.
- the formulation is made to suit the mode of administration.
- compositions may be in a water-soluble form, such as being present as pharmaceutically acceptable salts, which is meant to include both acid and base addition salts.
- Pharmaceutically acceptable carriers are determined in part by the particular composition being administered, as well as by the particular method used to administer the composition. Accordingly, there is a wide variety of suitable formulations of pharmaceutical compositions of the present disclosure.
- the formulations can be presented in unit-dose or multidose sealed containers, such as ampules and vials.
- Formulations suitable for administration include aqueous and non-aqueous solutions, isotonic sterile solutions, which can contain antioxidants, buffers, bacteriostats, and solutes that render the formulation isotonic, and aqueous and non-aqueous sterile suspensions that can include suspending agents, solubilizers, thickening agents, stabilizers, and preservatives. Solutions and suspensions can be prepared from sterile powders, granules, and tablets.
- compositions and formulations of the disclosure may comprise a pharmaceutically acceptable carrier, excipient or stabilizer.
- Pharmaceutically acceptable carriers are determined in part by the particular composition being administered, as well as by the particular method used to administer the composition. Accordingly, there is a wide variety of suitable formulations of pharmaceutical compositions (including optional pharmaceutically acceptable carriers, excipients, or stabilizers) of the present disclosure (see, e.g., Remington ’s Pharmaceutical Sciences, 17 th ed. 1985).
- Suitable carriers include but are not limited to, buffers containing succinate, phosphate, borate, HEPES, citrate, histidine, imidazole, acetate, bicarbonate, and other organic acids; antioxidants including but not limited to, ascorbic acid; low molecular weight polypeptides including but not limited to those less than about 10 residues; proteins, including but not limited to, serum albumin, gelatin, or immunoglobulins; hydrophilic polymers including but not limited to, polyvinylpyrrolidone; amino acids including but not limited to, glycine, glutamine, asparagine, arginine, histidine or histidine derivatives, methionine, glutamate, or lysine; monosaccharides, disaccharides, and other carbohydrates, including but not limited to, trehalose, sucrose, glucose, mannose, or dextrins; chelating agents including but not limited to, EDTA and edentate disodium; divalent metal ions including but not limited
- Suitable surfactants include for example but are not limited to polyethers based upon poly(ethylene oxide)-poly(propylene oxide)- poly(ethylene oxide), i.e., (PEO-PPO-PEO), or polypropylene oxide)-poly(ethylene oxide)- poly(propylene oxide), i.e., (PPO-PEO-PPO), or a combination thereof.
- PEO-PPO-PEO and PPO- PEO-PPO are commercially available under the trade names PluronicsTM, R-PluronicsTM, TetronicsTM and R-TetronicsTM (BASF Wyandotte Corp., Wyandotte, Mich.) and are further described in U.S. Pat. No.
- ethylene/polypropylene block polymers may be suitable surfactants.
- a surfactant or a combination of surfactants may be used to stabilize PEGylated boIFNz.3 against one or more stresses including but not limited to stress that results from agitation. Some of the above may be referred to as “bulking agents.” Some may also be referred to as “tonicity modifiers.”
- Antimicrobial preservatives may also be applied for product stability and antimicrobial effectiveness; suitable preservatives include but are not limited to, benzyl alcohol, benzalkonium chloride, metacresol, methyl/propyl parabene, cresol, and phenol, or a combination thereof.
- U.S. Patent No. 7,144,574 which is incorporated by reference herein, describe additional materials that may be suitable in pharmaceutical compositions and formulations of the invention and other delivery preparations.
- compositions comprising one or more protein or conjugated protein of the disclosure can be tested in one or more appropriate in vitro and/or in vivo models of disease, to confirm efficacy, tissue metabolism, and to estimate dosages, according to methods known to those of ordinary skill in the art, i.e., in a relevant assay.
- Administration is by any of the routes normally used for introducing a molecule into ultimate contact with blood or tissue cells.
- the proteins or conjugated proteins of the disclosure are administered in any suitable manner.
- the proteins or conjugated proteins of the disclosure can be administered in any suitable manner with one or more pharmaceutically acceptable carriers or excipients. Suitable methods of administering such conjugated proteins in the context of the present disclosure to a subject are available, and, although more than one route can be used to administer a particular composition, a particular route can often provide a more immediate and more effective action or reaction than another route.
- proteins or conjugated proteins of the disclosure may be administered by any conventional route suitable for proteins or peptides, including, but not limited to parenterally, e.g., injections including, but not limited to, subcutaneously, intramuscularly or intravenously or any other form of injections or infusions.
- Protein and conjugated protein compositions can be administered by a number of routes including, but not limited to oral, intravenous, intraperitoneal, intramuscular, transdermal, subcutaneous, topical, sublingual, or rectal means.
- the boIFNZ.3 protein or variant thereof, or pharmaceutical composition comprising the boIFNZ.3 protein or variant thereof is administered subcutaneously.
- the conjugated boIFNZ.3 protein, or pharmaceutical composition comprising the conjugated boIFNZ.3 protein is administered subcutaneously. In some embodiments, the boIFNZ.3 protein or variant thereof, or pharmaceutical composition comprising the boIFNZ.3 protein or variant thereof, is administered intramuscularly. In some embodiments, the conjugated boIFNZ.3 protein, or pharmaceutical composition comprising the conjugated boIFNZ.3 protein, is administered intramuscularly. Compositions comprising proteins or conjugated proteins can also be administered via liposomes. Such administration routes and appropriate formulations are generally known to those of skill in the art. The protein or conjugated protein may be used alone or in combination with other suitable components such as a pharmaceutical carrier. The conjugated protein may be used in combination with other agents or therapeutics. In some embodiments, the conjugated protein is used in combination with a vaccine. In some embodiments, the conjugated protein is used in combination with an antibiotic.
- the dose administered to a subject is sufficient to have a beneficial therapeutic response in the subject over time, or other appropriate activity, depending on the application.
- the dose is determined by the efficacy of the particular protein or conjugated protein, or formulation containing the protein or conjugated protein, and the activity, stability, or plasma or serum or plasma half-life of the protein or conjugated protein employed, and the condition of the subject, as well as the body weight or surface area of the subject to be treated.
- the size of the dose is also determined by the existence, nature, and extent of any adverse side-effects that accompany the administration of a particular conjugated protein, formulation, or the like in a particular subject. Administration can be accomplished via single or divided doses.
- the present disclosure also provides a pharmaceutical composition comprising a protein or conjugated protein of the present disclosure and an antibiotic for the treatment of a condition in a mammal, such as bovine respiratory disease.
- the antibiotic is a penicillin, a cephalosporin, a macrolide, a tetracycline or a fluoroquinolone antibiotic; or a combination thereof.
- the antibiotic is a cephalosporin antibiotic.
- the antibiotic is a macrolide antibiotic.
- Non-limiting examples of penicillin, cephalosporin, macrolide and tetracycline antibiotics that can be used in combination with a protein or conjugated protein of the present disclosure, or that can be formulated in combination with a protein or conjugated protein of the present disclosure, are disclosed herein.
- the present disclosure also provides a pharmaceutical composition comprising a protein or conjugated protein of the present disclosure and a vaccine for the treatment of a condition in a mammal, such as a disease or condition associated with BRDV or FMDV.
- the engineered boIFNz.3 proteins, conjugated boIFNz.3 proteins, and compositions containing the engineered boIFNz.3 proteins or conjugated boIFNz.3 proteins of the present disclosure are useful for prophylactic treatment of a viral-associated disease or condition in a mammal, or more particularly, a ruminant mammal.
- the disclosure includes a method of treating a ruminant mammal that is at risk for, is having, or has had, a viral or a virus-associated disease or condition.
- the disease or condition is foot and mouth disease (FMD).
- the disease or condition is bovine respiratory disease (BRD).
- the present disclosure provides a method of treating a disease or condition in a ruminant mammal.
- the method of treatment can include providing protection against the disease or condition.
- the method of treatment comprises administering a therapeutically effective amount of a conjugated boIFN/3 protein, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein, to a ruminant mammal.
- the ruminant mammal is bovine.
- the ruminant mammal is cattle.
- the ruminant mammal is a bovine calf.
- the conjugated boIFNz.3 protein comprises an engineered boIFNz.3 protein having an amino acid sequence of SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24, or any other boIFNz.3 protein, comprising one or more non-natural amino acids.
- the engineered boIFNz.3 protein is conjugated to a water-soluble polymer via the non-natural amino acid.
- the water-soluble polymer is a polyalkylene glycol or a derivative thereof.
- the water-soluble polymer is a PEG or a derivative thereof.
- the PEG or derivative thereof has a molecular weight of about 30kDa.
- the one or more non-natural amino acids is one non-natural amino acid.
- the non-natural amino acid is pAF.
- the conjugated boIFNz.3 protein comprises an engineered boIFNZ.3 protein having an amino acid sequence of SEQ ID NO: 18.
- the conjugated boIFNz.3 protein comprises an engineered boIFNz.3 protein having an amino acid sequence of SEQ ID NO: 19.
- the disease or condition is foot and mouth disease (FMD).
- the disease or condition is bovine respiratory disease (BRD). Suitable conjugates and engineered boIFNz.3 proteins are as disclosed elsewhere herein.
- the present disclosure provides a method of treating a disease or condition in a ruminant mammal, wherein the method comprises administering a vaccine to the ruminant mammal, and administering a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein.
- the present disclosure provides a method of treating a disease or condition in a ruminant mammal, wherein the method comprises administering the combination of a therapeutically effective amount of conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein, and a vaccine to the ruminant mammal.
- a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure can be administered as a single or divided dose to a subject, such as a ruminant mammal.
- a conjugated boIFNz.3 protein dose is at least about 1 pg/kg of the subject. In some embodiments, the dose is at least about 5 pg/kg of the subject. In some embodiments, the dose is at least about 10 pg/kg of the subject. In some embodiments, the dose is at least about 25 pg/kg of the subject. In some embodiments, the dose is at least about 50 pg/kg of the subject. In some embodiments, the dose is at least about 75 pg/kg of the subject.
- the dose is at least about 100 pg/kg of the subject. In some embodiments, the dose is at most about 1,000 pg/kg of the subject. In some embodiments, the dose is at most about 750 pg/kg of the subject. In some embodiments, the dose is at most about 500 pg/kg of the subject. In some embodiments, the dose is at most about 400 pg/kg of the subject. In some embodiments, the dose is at most about 300 pg/kg of the subject. In some embodiments, the dose is at most about 250 pg/kg of the subject. In some embodiments, the dose is at most about 200 pg/kg of the subject. In some embodiments, the dose is at most about 150 pg/kg of the subject.
- the dose is within a range of about 5 pg/kg to about 1000 pg/kg of the subject. In some embodiments, the dose is within a range of about 5 pg/kg to about 750 pg/kg of the subject. In some embodiments, the dose is within a range of about 5 pg/kg to about 500 pg/kg of the subject. In some embodiments, the dose is within a range of about 5 pg/kg to about 400 pg/kg of the subject. In some embodiments, the dose is within a range of about 5 pg/kg to about 300 pg/kg of the subject.
- the dose is within a range of about 5 pg/kg to about 250 pg/kg of the subject. In some embodiments, the dose is within a range of about 5 pg/kg to about 200 pg/kg of the subject. In some embodiments, the dose is within a range of about 5 pg/kg to about 150 pg/kg of the subject. In some embodiments, the dose is within a range of about 10 pg/kg to about 1000 pg/kg of the subject. In some embodiments, the dose is within a range of about 10 pg/kg to about 750 gg/kg of the subject. In some embodiments, the dose is within a range of about 10 gg/kg to about 500 gg/kg of the subject.
- the dose is within a range of about 10 gg/kg to about 400 gg/kg of the subject. In some embodiments, the dose is within a range of about 10 gg/kg to about 300 gg/kg of the subject. In some embodiments, the dose is within a range of about 10 gg/kg to about 250 gg/kg of the subject. In some embodiments, the dose is within a range of about 25 gg/kg to about 1000 gg/kg of the subject. In some embodiments, the dose is within a range of about 25 gg/kg to about 750 gg/kg of the subject. In some embodiments, the dose is within a range of about 25 gg/kg to about 500 gg/kg of the subject.
- the dose is within a range of about 25 gg/kg to about 400 gg/kg of the subject. In some embodiments, the dose is within a range of about 25 gg/kg to about 300 gg/kg of the subject. In some embodiments, the dose is within a range of about 25 gg/kg to about 250 gg/kg of the subject. In some embodiments, the dose is within a range of about 25 gg/kg to about 200 gg/kg of the subject. In some embodiments, the dose is within a range of about 25 gg/kg to about 150 gg/kg of the subject. In some embodiments, the dose is within a range of about 50 gg/kg to about 1000 gg/kg of the subject.
- the dose is within a range of about 50 gg/kg to about 750 gg/kg of the subject. In some embodiments, the dose is within a range of about 50 gg/kg to about 500 gg/kg of the subject. In some embodiments, the dose is within a range of about 50 gg/kg to about 400 gg/kg of the subject. In some embodiments, the dose is within a range of about 50 gg/kg to about 300 gg/kg of the subject. In some embodiments, the dose is within a range of about 50 gg/kg to about 250 gg/kg of the subject. In some embodiments, the dose is within a range of about 50 gg/kg to about 200 gg/kg of the subject.
- the dose is within a range of about 50 gg/kg to about 150 gg/kg of the subject. In some embodiments, the dose is within a range of about 75 gg/kg to about 1000 gg/kg of the subject. In some embodiments, the dose is within a range of about 75 gg/kg to about 750 gg/kg of the subject. In some embodiments, the dose is within a range of about 75 gg/kg to about 500 gg/kg of the subject. In some embodiments, the dose is within a range of about 75 gg/kg to about 400 gg/kg of the subject. In some embodiments, the dose is within a range of about 75 gg/kg to about 300 gg/kg of the subject.
- the dose is within a range of about 75 gg/kg to about 250 gg/kg of the subject. In some embodiments, the dose is within a range of about 75 gg/kg to about 200 gg/kg of the subject. In some embodiments, the dose is within a range of about 75 gg/kg to about 150 gg/kg of the subject. In some embodiments, the dose is within a range of about 50 gg/kg to about 500 gg/kg of the subject. In some embodiments, the dose is within a range of about 75 gg/kg to about 250 gg/kg of the subject. In some embodiments, the dose is within a range of about 100 gg/kg to about 200 gg/kg of the subject.
- the dose is about 50 gg/kg of the subject. In some embodiments, the dose is about 55 gg/kg of the subject. In some embodiments, the dose is about 60 gg/kg of the subject. In some embodiments, the dose is about 65 gg/kg of the subject. In some embodiments, the dose is about 70 gg/kg of the subject. In some embodiments, the dose is about 75 gg/kg of the subject. In some embodiments, the dose is about 80 gg/kg of the subject. In some embodiments, the dose is about 85 gg/kg of the subject. In some embodiments, the dose is about 90 gg/kg of the subject. In some embodiments, the dose is about 95 gg/kg of the subject.
- the dose is about 100 gg/kg of the subject. In some embodiments, the dose is about 105 gg/kg of the subject. In some embodiments, the dose is about 110 gg/kg of the subject. In some embodiments, the dose is about 115 gg/kg of the subject. In some embodiments, the dose is about 120 gg/kg of the subject. In some embodiments, the dose is about 125 gg/kg of the subject. In some embodiments, the dose is about 130 gg/kg of the subject. In some embodiments, the dose is about 135 gg/kg of the subject. In some embodiments, the dose is about 140 gg/kg of the subject. In some embodiments, the dose is about 145 gg/kg of the subject.
- the dose is about 150 gg/kg of the subject. In some embodiments, the dose is about 155 gg/kg of the subject. In some embodiments, the dose is about 160 gg/kg of the subject. In some embodiments, the dose is about 165 gg/kg of the subject. In some embodiments, the dose is about 170 gg/kg of the subject. In some embodiments, the dose is about 175 gg/kg of the subject. In some embodiments, the dose is about 180 gg/kg of the subject. In some embodiments, the dose is about 185 gg/kg of the subject. In some embodiments, the dose is about 190 gg/kg of the subject. In some embodiments, the dose is about 195 gg/kg of the subject. In some embodiments, the dose is about 200 gg/kg of the subject. In some embodiments, the doses is administered subcutaneously. In some other embodiments, the dose is administered intramuscularly.
- FMDV is a highly contagious pathogen among cloven-hoofed animals, including cattle.
- Current vaccines against FMDV do not induce protective antibody titers until 5 to 7 days post-vaccination, rendering vaccinated animals susceptible to infection and transmission (Diaz-San Segundo F. et al. (2011) Virology 413:283-292).
- a treatment that induces early protection against FMD (e.g., protection that begins prior to the induction of protective antibody titers in response to a vaccine), thereby closing the susceptibility gap between vaccination and the onset of adaptive immunity.
- the present disclosure provides a method of treating FMD in a ruminant mammal, wherein the method comprises administering a therapeutically effective amount of conjugated boIFNZ.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNZ.3 protein, to the ruminant mammal.
- the ruminant mammal is bovine.
- the ruminant mammal is a cattle.
- the ruminant mammal is a bovine calf.
- the method of treatment can provide protection against FMD, provide protection against FMD symptoms, provide protection against FMDV infection, reduce a rate of infection by FMDV, reduce a degree of infection by FMDV, inhibit FMDV replication, delay onset of FMD, reduce severity of FMD, reduce severity of FMD symptoms, prevent FMD and/or prevent infection by FMDV.
- the method of treatment can reduce the rate of transmission, or prevent the transmission, of FMDV from one ruminant mammal to another ruminant mammal or mammals.
- the method further comprises administering a FMDV vaccine to the ruminant mammal.
- the method of treating FMD provides the ruminant mammal with early protection against FMD, i.e., protection against FMDV infection throughout an early protection period.
- the early protection period begins within about 1 day from the time of administration of the conjugated boIFN/3 protein to the mammal and can last for at least about 5 to 7 days.
- the early protection period begins prior to the induction of protective antibody titers in the mammal in response to a vaccine.
- the early protection period is about 1 day, about 2 days, about 3 days, about 4 days, about 5 days, about 6 days or about 7 days or more.
- the early protection period is a period of about 1 day.
- the early protection period is a period of about 2 days. In some embodiments, the early protection period is a period of about 3 days. In some embodiments, the early protection period is a period of about 4 days. In some embodiments, the early protection period is a period of about 5 days. In some embodiments, the early protection period is a period of about 6 days. In some embodiments, the early protection period is a period of about 7 days. In some embodiments, the early protection period is about 1 day to about 5 days. In some embodiments, the early protection period is about 1 day to about 7 days.
- the method of treating FMD further provides the ruminant mammal with long-term protection against FMD, including long-term protection from FMDV infection, i.e., protection against FMDV throughout a long-term protection period.
- the long-term protection period is at least about 2 weeks. In some embodiments, the long-term protection period is at least about 3 weeks. In some embodiments, the long-term protection period is at least about 4 weeks. In some embodiments, the long-term protection period is at least about a month. In some embodiments, the long-term protection period is at least about 2 months. In some embodiments, the long-term protection period is at least about 3 months.
- the long-term protection period is about 100 days, or is about 120 days, or more.
- An early and/or long-term protection against FMD can be characterized, for example, by an absence of viremia, including a negative result in a test for the virus using methods known to a person of skill in the art.
- the protection against FMD is characterized as an absence of a clinical FMD symptom.
- the absence of a clinical FMD symptom is an absence of fever, an absence of vesicular lesions (blisters, ulcers and/or sores) in the mouth, tongue, muzzle, nostrils, teats and/or feet, and/or an absence of lameness, e.g., as determined by a veterinarian or veterinary scientist.
- the ruminant mammal is bovine, and the fever is characterized by an elevated body temperature (e.g., a body temperature within a range of about 103 °F to about 106 °F in a bovid).
- the method of treating FMD comprises combination therapy.
- the present disclosure provides a method of treating FMD in a ruminant mammal, wherein the method comprises administering a vaccine to the ruminant mammal and administering a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein to the ruminant mammal.
- the present disclosure provides a method of treating FMD in a ruminant mammal, wherein the method comprises administering the combination of a therapeutically effective amount of a conjugated boIFNZ.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein, and a vaccine to the ruminant mammal.
- the method of treating FMD comprises administering a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNZ.3 protein, and a FMD vaccine to a ruminant mammal, thereby providing the ruminant mammal with early and long-term protection against FMD.
- the vaccine is a FMD antigen.
- the vaccine is an inactivated whole FMDV antigen.
- the vaccine is a modified live vaccine.
- the FMD vaccine is an adenovirus type 5 (Ad5) FMD (Ad5-FMD) vaccine (e.g., as disclosed in US patent no. 8,906,384, the entire contents of which are hereby incorporated by reference herein in their entirety).
- Ad5-FMD adenovirus type 5
- FMDV is highly variable, with seven serotypes and dozens of subtypes, each one may require a specific vaccine.
- emerging strains of FMDV may require specific new vaccines to be developed.
- the method of treatment comprises administering a therapeutically effective amount of the conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein, and a FMD vaccine that is effective in the treatment of one or more of the FMDV serotypes and/or subtypes, or an emerging FMDV strain.
- the method provides the ruminant mammal with early protection against FMD.
- the early protection occurs throughout an early protection period.
- the early protection period begins prior to the induction of protective antibody titers that can be induced via vaccination.
- the early protection period precedes the induction of protective antibody titers post-vaccination.
- the early protection period is a period of about 1 day, about 2 days, about 3 days, about 4 days, about 5 days, about 6 days or about 7 days or more.
- the early protection period is a period of about 1 day.
- the early protection period is a period of about 2 days.
- the early protection period is a period of about 3 days.
- the early protection period is a period of about 4 days.
- the early protection period is a period of about 5 days.
- the early protection period is a period of about 6 days.
- the early protection period is a period of about 7 days. In some embodiments, the early protection period is a period of about 1 day to about 5 days. In some embodiments, the early protection period is a period of about 1 day to about 7 days. In some embodiments, the method of treatment further provides the ruminant mammal with long-term protection against FMD, including protection from FMDV infection. In some embodiments, the long-term protection period is at least about 2 weeks. In some embodiments, the long-term protection period is at least about 3 weeks. In some embodiments, the long-term protection period is at least about 4 weeks. In some embodiments, the long-term protection period is at least about a month. In some embodiments, the long-term protection period is at least about 2 months.
- the long-term protection period is at least about 3 months. In some embodiments, the long-term protection period is about 100 days, or is about 120 days, or more.
- the early and/or long-term protection against FMD can be characterized, for example, by an absence of viremia. Absence of viremia can include a negative result in a test for the virus using methods known to a person of skill in the art, or methods disclosed herein. In some embodiments, the early and/or long-term protection against FMD is characterized as an absence of a clinical FMD symptom.
- the absence of a clinical FMD symptom is an absence of fever, an absence of vesicular lesions (blisters, ulcers and/or sores) in the mouth, tongue, muzzle, nostrils, teats and/or feet, and/or an absence of lameness, e.g., as determined by a veterinarian or veterinary scientist.
- the ruminant mammal is bovine, and the fever is characterized by an elevated body temperature (e.g., a body temperature within a range of about 103 °F to about 106 °F in a bovid).
- the conjugated boIFNz.3 protein can be administered prior to, after, or at about the same time as the administration of the vaccine, as determined by a person of skill in the art, or by a veterinarian or veterinary scientist. In some embodiments, the conjugated boIFNZ.3 protein or composition is administered before the administration of the vaccine.
- the conjugated boIFNz.3 protein or composition is administered before but within about a week of the administration of the vaccine, e.g., up to about one week, about 6 days, about 5 days, about 4 days, about 3 days, about 2 days or about 1 day (e.g., up to about 24 hours, e.g., up to about 20 hours, about 16 hours, about 12 hours, about 8 hours or about 4 hours) before the administration of the vaccine.
- the conjugated boIFNz.3 protein or composition is administered about 0-7 days, about 1-6 days, about 2-5 days or 3-4 days, or is administered about 4 hours to about 7 days, about 8 hours to about 6 days, about 12 hours to about
- the conjugated boIFNz.3 protein or composition is administered about 1 day before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 2 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 3 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 4 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 5 days before the administration of the vaccine.
- the conjugated boIFNz.3 protein or composition is administered about 6 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 7 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein is administered after the administration of the vaccine. For example, in some embodiments, the conjugated boIFNz.3 protein is administered after but within about one week of the vaccine, e.g., up to about 6 days, about 5 days, about 4 days, about 3 days, about 2 days, or about 1 day after the administration of the vaccine. For example, in some embodiments, the conjugated boIFNz.3 protein or composition is administered about 0-7 days, about 1-6 days, about 2-5 days or 3-4 days, or is administered about 4 hours to about 7 days, about 8 hours to about
- the conjugated boIFNz.3 protein is administered at about the same time as the administration of the vaccine (e.g., within about 4 hours, about 3 hours, about 2 hours, about 1 hour or about 30 minutes of the administration of the vaccine).
- the method of treating the ruminant mammal for FMD comprises administering a composition comprising both FMD vaccine and the conjugated boIFNZ.3 protein to the ruminant mammal.
- the administration is a subcutaneous administration.
- the administration is an intramuscular administration.
- the present disclosure also provides a method of inducing systemic antiviral activity in a ruminant mammal, the method comprising administering to a ruminant mammal a therapeutically effective amount of a conjugated protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated protein.
- the method further comprises administering a vaccine that is capable of preventing FMDV.
- the method induces anti-FMDV activity, thereby preventing FMD.
- the method induces adjuvanted adaptive immune response against FMDV, thereby preventing FMD.
- the method induces expression of interferon-stimulated genes correlated with systemic control of viral replication in an animal susceptible to FMDV in vivo, thereby preventing FMD.
- the present disclosure also provides a method of inducing systemic antiviral activity in a ruminant mammal, the method comprising administering a vaccine to the ruminant mammal, wherein the method further comprises administering a therapeutically effective amount of a conjugated protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated protein to the ruminant mammal.
- the present disclosure also provides a method of inducing systemic antiviral activity in a ruminant mammal, the method comprising administering a combination of a therapeutically effective amount of a conjugated boIFNZ.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFN/3 protein, and a vaccine to the ruminant mammal.
- a method of inducing systemic antiviral activity in a ruminant mammal comprising administering a combination of a therapeutically effective amount of a conjugated boIFNZ.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFN/3 protein, and a vaccine to the ruminant mammal.
- Appropriate doses of each of the conjugated protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated protein and the vaccine, as well as appropriate timings are as disclosed elsewhere herein.
- IFNs can provide protection against other important domestic livestock diseases such as bovine viral diarrhea virus (BVDV) as well as bovine respiratory syncytial virus (BRSV), infectious bovine rhinotracheitis virus (IBR), bovine coronaviruses and parainfluenza-3 viruses (PI-3) among others. These viruses are among the initial causes of the bovine respiratory disease (BRD) complex that open the door to severe bacterial infections of the lung.
- BRD bovine respiratory disease
- a nonantibiotic-based early intervention strategy to prevent and control BRD is highly desirable.
- BoIFNZ.3 is most active in the upper respiratory track in cattle and thus represents an opportunity for a targeted approach to treating or preventing BRD.
- the present disclosure provides a method of treating BRD in a ruminant mammal, wherein the method comprises administering a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNZ.3 protein, to the ruminant mammal.
- the ruminant mammal is bovine.
- the ruminant mammal is a cattle.
- the ruminant mammal is a bovine calf.
- the method of treatment can provide protection against BRD, provide protection against BRD symptoms, provide protection against infections associated with BRD, including viral infections associated with BRD, reduce a rate of infection by a virus associated with BRD, reduce a degree of infection by a virus associated with BRD, inhibit replication of a virus associated with BRD, reduce severity of BRD, reduce severity of BRD symptoms, prevent BRD and/or prevent infection by a virus associated with BRD.
- the method of treatment can reduce the rate of transmission, or prevent the transmission, of a virus associated with BRD from one ruminant mammal to another ruminant mammal or mammals.
- the method further comprises administering a vaccine for the treatment of a virus associated with BRD to the ruminant mammal.
- the virus associated with BRD is selected from the group consisting of bovine viral diarrhea virus (BVDV), bovine respiratory syncytial virus (BRSV), infectious bovine rhinotracheitis virus (IBR), a bovine coronavirus and a parainfluenza-3 virus (PI-3); and combinations thereof.
- the virus associated with BRD is selected from the group consisting of BVDV, BRSV, a bovine coronavirus and PI-3; and combinations thereof.
- the method of treating BRD provides the ruminant mammal with early protection against BRD.
- the early protection occurs throughout an early protection period.
- the early protection period begins prior to the induction of protective antibody titers that can be induced via vaccination.
- the early protection period begins prior to the induction of protective antibody titers post-vaccination.
- the early protection period is a period of about 1 day, about 2 days, about 3 days, about 4 days, about 5 days, about 6 days or about 7 days or more.
- the early protection period is a period of about 1 day.
- the early protection period is a period of about 2 days.
- the early protection period is a period of about 3 days. In some embodiments, the early protection period is a period of about 4 days. In some embodiments, the early protection period is a period of about 5 days. In some embodiments, the early protection period is a period of about 6 days. In some embodiments, the early protection period is a period of about 7 days. In some embodiments, the early protection period is a period of about 1 day to about 5 days. In some embodiments, the early protection period is a period of about 1 day to about 7 days. In some embodiments, the method further provides the ruminant mammal with long-term protection against BRD, including protection from a virus associated with BRD.
- the long-term protection occurs during a long-term protection period that begins after the administration of the conjugated protein.
- the long-term protection period can coincide with the early protection period.
- the long-term protection period is at least about 2 weeks.
- the long-term protection period is at least about 3 weeks.
- the long-term protection period is at least about 4 weeks.
- the long-term protection period is at least about a month.
- the long-term protection period is at least about 2 months.
- the long-term protection period is at least about 3 months.
- the long-term protection period is about 100 days, or about 120 days or more.
- the method of treating BRD provides the ruminant mammal with both early protection and long-term protection against BRD.
- the early and/or longterm protection against BRD is an early and/or long-term protection against infection by a virus associated with BRD.
- the early and/or long-term protection is a prevention of infection by a virus associated with BRD.
- the early and/or long-term protection against BRD is characterized as an absence of viremia.
- the early and/or long-term protection against BRD is characterized as an absence of a clinical BRD symptom.
- the method of treating BRD comprises combination therapy.
- the present disclosure provides a method of treating BRD in a ruminant mammal, wherein the method comprises administering a vaccine to the ruminant mammal, and administering a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein to the ruminant mammal.
- the present disclosure provides a method of treating BRD in a ruminant mammal, wherein the method comprises administering the combination of a therapeutically effective amount of conjugated boIFNZ.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein, and a vaccine to the ruminant mammal.
- the method of treating BRD comprises administering a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNZ.3 protein, and a vaccine for the treatment of a virus associated with BRD to a ruminant mammal, thereby providing the ruminant mammal with protection against BRD.
- the vaccine is a BVDV antigen, a BRSV antigen, an IBR antigen, a PI-3 antigen or a bovine coronavirus antigen.
- the vaccine is an inactivated whole BVDV antigen, an inactivated whole BRSV antigen, an inactivated whole IBR antigen, an inactivated whole PI-3 antigen or an inactivated whole bovine coronavirus antigen.
- the vaccine is a modified live vaccine.
- the method provides the ruminant mammal with early protection against BRD.
- the early protection occurs throughout an early protection period.
- the early protection period is a time period that precedes the induction of protective antibody titers that can be induced via the vaccination.
- the early protection period is a time period that begins prior to the induction of protective antibody titers post-vaccination.
- the early protection period is a period of about 1 day, about 2 days, about 3 days, about 4 days, about 5 days, about 6 days or about 7 days or more. In some embodiments, the early protection period is a period of about 1 day. In some embodiments, the early protection period is a period of about 2 days. In some embodiments, the early protection period is a period of about 3 days. In some embodiments, the early protection period is a period of about 4 days. In some embodiments, the early protection period is a period of about 5 days. In some embodiments, the early protection period is a period of about 6 days. In some embodiments, the early protection period is a period of about 7 days. In some embodiments, the early protection period is a period of about 1 day to about 5 days.
- the early protection period is a period of about 1 day to about 7 days.
- the method further provides the ruminant mammal with long-term protection against FMD.
- the long-term protection against BRD occurs during a long-term protection period after the administration.
- the long-term protection period can coincide with the early protection period.
- the long-term protection period is at least about 2 weeks.
- the long-term protection period is at least about 3 weeks.
- the long-term protection period is at least about 4 weeks.
- the long-term protection period is at least about a month. In some embodiments, the long-term protection period is at least about 2 months.
- the long-term protection period is at least about 3 months. In some embodiments, the long-term protection period is about 100 days, or about 120 days or more.
- the method provides the ruminant mammal with both early protection and long-term protection against BRD.
- the early and/or long-term protection against BRD is a protection against infection by a virus associated with BRD. In some embodiments the early and/or long-term protection is a prevention of infection by a virus associated with BRD.
- the early and/or long-term protection against BRD is characterized as an absence of viremia. In some embodiments, the early and/or long-term protection against BRD is characterized as an absence of a clinical BRD symptom.
- the virus associated with BRD is selected from the group consisting of bovine viral diarrhea virus (BVDV), bovine respiratory syncytial virus (BRSV), infectious bovine rhinotracheitis virus (IBR), a bovine coronavirus and a parainfluenza-3 virus (PI-3); and combinations thereof.
- the virus associated with BRD is selected from the group consisting of BVDV, BRSV, a bovine coronavirus and PI-3; and combinations thereof.
- the conjugated boIFNz.3 protein can be administered prior to, after, or at about the same time as the administration of the vaccine, as determined by a person of skill in the art, or by a veterinarian or veterinary scientist.
- the conjugated boIFNZ.3 protein or composition is administered before the administration of the vaccine.
- the conjugated boIFNz.3 protein or composition is administered before but within about one week of the administration of the vaccine, e.g., up to about one week, about 6 days, about 5 days, about 4 days, about 3 days, about 2 days or about 1 day (e.g., up to about 24 hours, e.g., up to about 20 hours, about 16 hours, about 12 hours, about 8 hours or about 4 hours) before the administration of the vaccine.
- the conjugated boIFNz.3 protein or composition is administered about 0-7 days, about 1-6 days, about 2-5 days or about 3- 4 days, or is administered about 4 hours to about 7 days, about 8 hours to about 6 days, about 12 hours to about 5 days, about 24 hours to about 4 days, about 36 hours to about 3 days, before the administration of the vaccine.
- the conjugated boIFNz.3 protein or composition is administered about 1 day before the administration of the vaccine.
- the conjugated boIFNz.3 protein or composition is administered about 2 days before the administration of the vaccine.
- the conjugated boIFNz.3 protein or composition is administered about 3 days before the administration of the vaccine.
- the conjugated boIFNz.3 protein or composition is administered about 4 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 5 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 6 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 7 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein is administered after the administration of the vaccine.
- the conjugated boIFNz.3 protein is administered after but within about one week of the administration of the vaccine, e.g., up to about 6 days, about 5 days, about 4 days, about 3 days, about 2 days, or about 1 day after the administration of the vaccine.
- the conjugated boIFNz.3 protein or composition is administered about 0-7 days, about 1-6 days, about 2-5 days or about 3-4 days after the administration of the vaccine, or is administered about 4 hours to about 7 days, about 8 hours to about 6 days, about 12 hours to about 5 days, about 24 hours to about 4 days or about 36 hours to about 3 days after the administration of the vaccine.
- the conjugated boIFNz.3 protein is administered at about the same time as the administration of the vaccine (e.g., within about 4 hours, about 3 hours, about 2 hours, about 1 hour or about 30 minutes of the administration of the vaccine).
- the method of treating BRD further comprises administering an antibiotic to the ruminant mammal.
- the present disclosure provides a method of treating BRD in a ruminant mammal, wherein the method comprises administering an antibiotic to the ruminant mammal, wherein the method further comprises administering a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein to the ruminant mammal.
- the present disclosure provides a method of treating BRD in a ruminant mammal, wherein the method comprises administering the combination of a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein, and an antibiotic to the ruminant mammal.
- the antibiotic is a penicillin, a cephalosporin, a macrolide, a tetracycline or a fluoroquinolone antibiotic; or a combination thereof.
- the antibiotic is a cephalosporin antibiotic.
- the antibiotic is a macrolide antibiotic.
- the conjugated boIFNz.3 protein can be administered prior to, after, or at about the same time as the administration of the antibiotic and/or vaccine, as determined by a person of skill in the art, or by a veterinarian or veterinary scientist.
- the conjugated boIFNz.3 protein or composition is administered before the administration of the antibiotic.
- the conjugated boIFNz.3 protein or composition is administered before but within about a week of the administration of the antibiotic, e.g., up to about one week, about 6 days, about 5 days, about 4 days, about 3 days, about 2 days or 1 about day (e.g., up to about 24 hours, e.g.
- the conjugated boIFNz.3 protein or composition is administered about 0-7 days, about 1-6 days, about 2-5 days or about 3- 4 days before the administration of the antibiotic, or about 4 hours to about 7 days, about 8 hours to about 6 days, about 12 hours to about 5 days, about 24 hours to about 4 days or about 36 hours to about 3 days before the administration of the antibiotic.
- the conjugated boIFNz.3 protein is administered after the administration of the antibiotic.
- the conjugated boIFNZ.3 protein is administered after but within about one week of the administration of the antibiotic e.g., up to about 6 days, about 5 days, about 4 days, about 3 days, about 2 days or about 1 day after the administration of the antibiotic.
- the conjugated boIFNZ.3 protein or composition is administered about 0-7 days, about 1-6 days, about 2-5 days or about 3-4 days, about 4 hours to about 7 days, about 8 hours to about 6 days, about 12 hours to about 5 days, about 24 hours to about 4 days or about 36 hours to about 3 days after the administration of the antibiotic.
- the conjugated boIFNz.3 protein is administered at about the same time as the administration of the antibiotic (e.g., within about 4 hours, about 3 hours, about 2 hours, about 1 hour or about 30 minutes of the administration of the antibiotic).
- the antibiotic is a penicillin.
- the penicillin is selected from the group consisting of an aminopenicillin (including ampicillin and amoxicillin), aztreonam, carbenicillin, a carboxypenicillin (including carbenicillin, its acid-stable indanyl ester, and ticarcillin), clavulanate-potentiated amoxicillin, cioxacillin, floxacillin, hetacillin, imipenem, an isoxazolyl penicillin (including oxacillin, cioxacillin, dicloxacillin and flucloxacillin), mecillinam, meropenem, methicillin, nafcillin, oxacillin, penicillin G (benzylpenicillin), penicillin V (phenoxymethyl-penicillin), phenethicillin, a piperazine penicillin (e.g., piperacillin); pivampicillin, sulficillin, sulfam, a carb
- the antibiotic is a macrolide.
- the macrolide antibiotic is selected from the group consisting of azithromycin, erythromycin, erythromycin A, erythromycin B, erythromycin C, erythromycin D, erythromycin E, gamithromycin, tilmicosin, tulathromycin, tildipirosin and tylosin; and combinations thereof.
- the antibiotic is a cephalosporin.
- the cephalosporin is selected from the group consisting of cefachlor, cefadroxil, cefamandole, cefazolin, cefepime, cefoperazone, ceforanide, cefotaxime, cefotiam, cefpodoxime, ceftazidime, cefovecin, cefoxitin (a cephamycin), cefsulodin, ceftiofur, ceftriaxone, cefuroxime, cephalexin, cephaloridine, cephalothin, cephapirin and cephradine; and combinations thereof.
- the antibiotic is a tetracycline.
- the tetracycline is selected from the group consisting of chlortetracycline, demethylchlortetracycline, doxycycline, a glycylcycline (including tigecycline), lymecycline, methacycline, minocycline, oxytetracycline, rolitetracycline and tetracycline; and combinations thereof.
- the antibiotic is a fluoroquinolone antibiotic.
- the fluoroquinolone antibiotic is Enrofloxacin (Baytril).
- the present disclosure also provides a method of inducing systemic antiviral activity in a ruminant mammal, the method comprising administering to a ruminant mammal a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure or a pharmaceutical composition comprising a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, and further comprising administering a vaccine for treatment of BRD.
- the present disclosure also provides a method of inducing systemic antiviral activity in a ruminant mammal, the method comprising administering a vaccine to a ruminant mammal, wherein the method further comprises administering a therapeutically effective amount of a conjugated protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated protein to the ruminant mammal.
- the present disclosure also provides a method of inducing systemic antiviral activity in a ruminant mammal, the method comprising administering a combination of a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein, and a vaccine to the ruminant mammal.
- the vaccine for the treatment of BRD is a vaccine for the treatment of BVDV, BRSV, IBR, a bovine coronavirus and/or PI-3.
- the method induces anti-BVDV, anti-BRSV, anti-IBR, anti-bovine coronavirus and/or anti-PI-3 activity, thereby delaying, reducing severity and/or preventing BRD.
- the method induces adjuvanted adaptive immune response against BRD, thereby delaying, reducing severity and/or preventing BRD.
- the method induces expression of interferon- stimulated genes correlated with systemic control of viral replication in an animal susceptible to a BRD-associated virus in vivo, thereby delaying, reducing severity and/or preventing BRD.
- engineered boIFNz.3 proteins and/or conjugated boIFNz.3 proteins of the present disclosure may reduce morbidity in cattle or other livestock species as well.
- Example 1 boIFNz.3 expression system
- This example describes expression systems and methods used for boIFNz.3 polypeptides comprising wild type amino acid sequence (SEQ ID NO: 1; Diaz-San Segundo F. et al. (2011) Virology 413:283-292; GenBank: ADP05157.1), modified amino acid sequences (N-terminal deletion variants of SEQ ID NOs: 2 to 13), and amino acid sequences containing a non-natural amino acid (SEQ ID NOs: 14 to 25).
- Host cells were transformed with constructs for orthogonal tRNA, orthogonal aminoacyl tRNA synthetase, and a polynucleotide encoding boIFNZ.3 polypeptide as in SEQ ID NOs: 2 to 13, or a polynucleotide comprising a selector codon encoding the amino acid sequences shown in SEQ ID NOs: 14 to 25 (e.g., the polynucleotide of SEQ ID NO: 27 encoding amino acid SEQ ID NO: 18).
- E. coli expression vector construction and sequence verification cloning and expression of wild type boIFNz.3, modified boIFNZ.3 sequences (N-terminal deletion variants), and boIFNZ.3 containing a non-naturally encoded amino acid in E. coli.
- BoIFNz.3 expression plasmids were constructed either by recombination-based cloning method using Gibson Assembly kit (New England Biolabs (NEB), Ipswich, MA) or by using QuikChange mutagenesis kit (Agilent Technologies, Santa Clara, CA) in E. coli NEB5a cloning strain (New England Biolabs, MA) as described below.
- the E. coli expression plasmids are shown in FIGs. 1 A and IB.
- Gibson Assembly The primers for amplifying various gene of interests (GOIs) containing Donor fragments had about 18-24 base pair (bp) overlap sequence at their 5'-termini with the Acceptor vector sequences for homologous recombination and were synthesized at Integrated DNA Technologies (IDT) (San Diego, CA). The PCR fragments were amplified using high fidelity DNA polymerase mix, Pfu Ultra II Hotstart PCR Master Mix (Cat. No: 600852, Agilent Technologies).
- the PCR products were digested with Dpnl restriction enzyme (NEB# R0176L) for 2 hours at 37 °C to remove plasmid background followed by column purification using Qiagen PCR column purification kit (Qiagen, Valencia, CA; # 28104) and quantitated by Nanodrop (ThermoFisher, Carlsbad, CA).
- the Acceptor vectors were linearized by digesting with unique restriction enzymes (NEB, MA) within the vector for 3 to 5 hours at supplier recommended temperatures, PCR column purified and quantitated.
- the Donor inserts and appropriately prepared Acceptor vectors were mixed at a 3:1 molar ratio, incubated at 50 °C for 15 min, using Gibson Assembly kit (NEB # E261 IS), and then used for transformation into E. coli NEB5a strain (New England Biolabs, Ipswitch, MA (NEB); NEB # 2987).
- the recombinants were recovered by plating Gibson Assembly mix on to lysogeny broth (LB) agar plates containing appropriate antibiotics. The next day, 4 to 6 well-isolated single colonies were inoculated into 5 mL LB + 50 pg/mL kanamycin sulfate (Sigma, St Louis, MO; cat# K0254) media and grown overnight at 37 °C.
- the recombinant plasmids were isolated using Qiagen plasmid DNA mini -prep kit (Qiagen #27104) and verified by DNA sequencing (Eton Biosciences, San Diego, CA). The complete GOI region plus 100 bp upstream and 100 bp downstream sequences were verified by using gene-specific sequencing primers.
- QuickChange Mutagenesis All Amber variants containing TAG stop codon were created by using QuickChange Lightning site directed mutagenesis kit (Agilent Technologies # 201519). All QCM oligonucleotides were designed using QuickChange Web Portal (Agilent Technologies), and ordered from IDT (San Diego, CA).
- the QCM PCR mix contained 5 pl of lOx buffer, 2.5 pl of dNTP Mix, 1 pl (100 ng) of plasmid template, 1 pl of oligo mix (10 pM concentration each), 1 pl of QuickChange Lightning enzyme, 2.5 pl of Quick solution and 37 pl of distilled water (DW). The DNA was amplified using the PCR program recommended by the kit for 18 cycles only.
- AXID Expression strain construction and verification: To prepare AXID production strains, chemically competent E. coli W3110B60 host cells were transformed with sequence- verified plasmid DNA (50 ng), the recombinant cells were selected on 2xYT+l% glucose agar plates containing 50 pg/mL kanamycin sulfate (Sigma, cat# K0254), and incubated overnight at 37 °C. A single colony from the fresh transformation plate was then propagated thrice on 2xYT+l% glucose agar plates containing 50 pg/mL kanamycin sulfate by sequential triplestreaking and incubating overnight at 37 °C.
- a single colony from the third-streaked plate was inoculated into 20 mL Super Broth (Fisher-OptigrowTM, #BP1432-1OB1) containing 50 pg/mL kanamycin sulfate (Sigma, cat# K0254) and incubated overnight at 37 °C and 250 rpm. The overnight grown culture was then diluted with glycerol to a final glycerol concentration of 20% (w/v) (KIC, Ref# 67790-GL99UK). This cell suspension was then dispensed into 1 mL aliquots into several cryovials and frozen at -80 °C as AXID production strain vials.
- glycerol vials of the AXID production strains were further validated by DNA sequencing and phenotypic characterization of antibiotic resistance markers.
- the plasmid was sequence verified. Twenty mL LB containing 50 pg/mL kanamycin sulfate was inoculated with a stab from a glycerol vial of the AXID clone and grown at 37 °C, 250 rpm overnight.
- the plasmid DNA was isolated using Qiagen Miniprep Kit (#27104) and the presence of intact GOI ORF in the isolated plasmid was confirmed by DNA sequencing (Eton Biosciences, CA).
- Expression system The amino acid sequences for boIFNZ.3 are shown in Table 1.
- a translation system that comprises an orthogonal tRNA (O-tRNA) and an orthogonal aminoacyl tRNA synthetase (O-RS) (see, W02006/068802 and W02007/021297) was used to express wild type boIFNz.3 sequence (see plasmid map pKGOlOO, FIG. 1A), boIFNz.3 sequences with N- terminal modifications, and boIFNZ.3 sequences containing a non-naturally encoded amino acid (see plasmid map pKG0168, FIG. IB).
- the O-RS preferentially aminoacylates the O-tRNA with a non-naturally encoded amino acid.
- the translation system inserts the non-naturally encoded amino acid into boIFNz.3 or boIFNz.3 variants, in response to an encoded selector codon.
- Suitable O-RS and O-tRNA sequences are described in W02006/068802 entitled “Compositions of Aminoacyl-tRNA Synthetase and Uses Thereof’ and W02007/021297 entitled “Compositions of tRNA and Uses Thereof’, the contents of each of which are hereby incorporated by reference in their entirety herein.
- Example 2 Deletion of N-terminal amino acids for improved expression of boIFNZ.3
- N-terminal amino acids 1-12 were sequentially removed from boIFNZ.3 wild type sequence (SEQ ID NO: 1; Diaz-San Segundo F. et al. (2011) Virology 413:283-292; GenBank: ADP05157.1) using QuickChange Mutagenesis (QCM) as described in Example 1 to provide variants of SEQ ID NOs: 2 to 13 (Table 1).
- QCM QuickChange Mutagenesis
- the AXID production strain, also described in Example 1, was used to test for boIFN 3 expression in shake flask experiments.
- plasmids for the expression of modified boIFNz.3 polypeptides were transformed into W3110B60 E. coli cells. Protein expression was induced by the addition of arabinose and cultures were incubated for 4 to 6 hours at 37 °C. An aliquot from the harvested cells were taken and analyzed for improved expression by SDS-PAGE; improved expression was seen with A4 and A5 N-terminal deletion variants (SEQ ID NO: 5 and SEQ ID NO: 6, respectively; data not shown).
- Recoverable titers post-purification were as follows: boIFNz.3 wild type: not detectable; boIFNz.3 Al : 8.5 mg/L; boIFNz.3 A3: 97.6 mg/L; boIFNz.3 A4: 107.6 mg/L; boIFNz.3 A5: 109.6 mg/L; and boIFNZ.3 A6: 64.6 mg/L.
- Additional constructs to increase boIFNz.3 expression in E. coli To increase the production of boIFNz.3 in E. coli, the following expression parameters could be further optimized in addition to DNA sequence optimization based on E. coli codon usage reported herein: testing different promoters besides T7 bacteriophage promoter such as arabinose B (araB), pTrc and bacteriophage T5 promoters; stabilization ofboIFNX3 mRNA; screening of different E.
- T7 bacteriophage promoter such as arabinose B (araB), pTrc and bacteriophage T5 promoters
- stabilization ofboIFNX3 mRNA Screen of different E.
- coli host strains besides the standard W3110B60 strain; production process parameters optimization such as temperature, culture media, inducer concentration, etc.; transcriptional and translational control elements optimization such as start and stop codons, ribosome binding site (RBS), transcriptional terminators etc; plasmid copy number and plasmid stability optimization; translational initiation region (TIR) optimization.
- production process parameters optimization such as temperature, culture media, inducer concentration, etc.
- transcriptional and translational control elements optimization such as start and stop codons, ribosome binding site (RBS), transcriptional terminators etc
- plasmid copy number and plasmid stability optimization translational initiation region (TIR) optimization.
- TIR translational initiation region
- Inclusion body prep Inclusion bodies containing boIFNz.3 N-terminal deletion variants were isolated through a series of wash steps. Frozen cell paste was re-suspended in wash buffer I (50 mM Tris, pH 8.0; 1% triton X-100; 100 mM NaCl, 1 mM EDTA) to a concentration of 10% (w/v) and homogenized at 4 °C followed by centrifugation (10,000g, 30 min, 4 °C). The supernatant was discarded, and the inclusion body pellet was re-suspended in an additional volume of wash buffer I followed by centrifuged at 15,000g for 30 min at 4 °C.
- wash buffer I 50 mM Tris, pH 8.0; 1% triton X-100; 100 mM NaCl, 1 mM EDTA
- the supernatant was discarded, and the inclusion body pellet was re-suspended in an additional volume of wash buffer I followed by centrifuged at
- the supernatant was discarded, and the inclusion body pellet was re-suspended in wash buffer II (50 mM Tris, pH 8.0; 100 mM NaCl, 1 mM EDTA) followed by centrifuged at 10,000g for 30 min at 4 °C. The supernatant was discarded, and the inclusion body pellet was re-suspended in water followed by centrifugation at 10,000g for 30 min at 4°C. Washed inclusion bodies were stored at -80 °C until further use.
- wash buffer II 50 mM Tris, pH 8.0; 100 mM NaCl, 1 mM EDTA
- BoIFNZ.3 Fractions containing boIFNZ.3 were collected and buffer exchanged into 30 mM sodium acetate, 200 mM glycine, 2.5% trehalose, pH 4.0. BoIFNZ.3 was concentrated to 1-10 mg/mL, 0.22 pM filtered, and stored at -80 °C. Purified samples were characterized by SDS-PAGE (data not shown), reversed phase HPLC (RP), and size exclusion chromatograph (SEC); (Table 2).
- Example 4 Real-time quantitative polymerase chain reaction (RT-qPCR) of ISG15 after treatment with boIFNz.3 variants in Madin-Darby bovine kidney (MDBK) cells
- Interferon-stimulated gene product 15 (ISG15), a ubiquitin-like protein involved in interferon induced antiviral response, was evaluated in an MDBK cell-based assay for induction levels based on treatment with boIFNz.3 variants.
- MDBK cells were treated with boIFNz.3 variants at the following concentrations: 100, 20, 4, 0.8, 0.16 ng/mL, and cells were isolated 6 hours post treatment to evaluate relative mRNA expression of ISG15.
- Total RNA was isolated using TRIzol Reagent (Thermo Fisher Scientific) and reverse transcribed into cDNA using TaqManTM Fast Advanced Master Mix (Applied Biosystems) according to manufacturer's protocol. Relative fold changes in ISG15 gene expression were evaluated and normalized against control. N-terminal deletion variants up to 12 amino acids (variants Al to A12; Table 1) exhibited mRNA expression of ISG15 (data not shown).
- the sites to be used in generating boIFNz.3 muteins were selected by modeling nonnatural amino acid para-acetyl-L-phenylalanine (pAF) at each amino acid site of full length human IFNz.3 protein crystal structure and assessing side chain accessible surface area (scASA) of the non-natural amino acid using Molecular Operating Environment (MOE) (Chemical Computing Group). Lead sites selected for pAF incorporation, with their associated scASA values, are disclosed in Table 3. Sites known to be important for receptor binding were avoided.
- FIG. 2 shows the boIFNZ.3 model with amino acids selected for pAF incorporation.
- Amino acid residue numbers are based on wild type sequence (SEQ ID NO: 1; Table 1).
- Example 6 E. coli shake flask expression of pAF site variants and high cell density fermentation
- Shake-flask expression testing The AXID production strain as described in Example 1 was used to test for boIFNz.3 expression in shake flask experiments. Briefly, an inoculum from the AXID glycerol vial was put into a 5 mL of Super Broth (Fisher-OptigrowTM, #BP1432-1OB1) media containing 50 pg/mL of kanamycin sulfate (Sigma, MO) and grown overnight at 37°C with shaking.
- Super Broth Fisher-OptigrowTM, #BP1432-1OB1
- kanamycin sulfate Sigma, MO
- the overnight culture was diluted 1 : 100 in Super Broth (Fisher-OptigrowTM, #BP1432- 10B1) media containing 50 pg/mL of kanamycin sulfate (Sigma, MO) and grown at 37 °C with shaking.
- kanamycin sulfate Sigma, MO
- the culture density reached an OD600 of 0.6-0.8, it was induced with 0.2% arabinose and non-natural amino acid pAF added, followed by harvesting after about 3 to 5 hours of production. An aliquot from the harvested cells was taken and analyzed by SDS-PAGE (data not shown).
- Optimal expression of boIFNZ.3 was standardized by varying temperature, duration of induction and inducer concentration.
- High cell density fermentations The fermentation process for production of boIFNZ.3 consisted of two stages: (i) inoculum preparation and (ii) fermentor production.
- the inoculum was started from a single glycerol vial, thawed, diluted 1 : 1000 (v/v) into 50 mL of defined seed medium in a 250 mL baffled Erlenmeyer flask, and incubated at 37 °C and 250 rpm. Prior to use, the fermentor was cleaned and autoclaved. A specified amount of basal medium was added to the fermentor and steam sterilized.
- kanamycin sulfate solution Specified amounts of kanamycin sulfate solution, feed medium and P2000 antifoam were added to the basal medium prior to inoculation. All solutions added to the fermentor after autoclaving were either 0.2 pm filtered or autoclaved prior to aseptic addition.
- the fermentor was batched with 4 L of chemically defined medium that utilizes glycerol as a carbon source.
- the seed culture was added to the fermentor to an initial OD600nm of 0.05.
- Dissolved oxygen was maintained at 30% air saturation using agitation from 480 to 1200 rpm and oxygen enrichment with a head pressure of 6 psig and air flow of 5 slpm.
- the temperature and pH were controlled at 37 °C and 7.0, respectively.
- L-Ala-pAcF (also referred to as L-Ala-pAF)
- dipeptide was added at 0.4 g/L.
- the culture was induced with L-arabinose at a final concentration of 2 g/L.
- the culture was harvested at 6 h post induction.
- Example 7 Purification of boIFNZ.3 N-terminal A4 deletion variants containing nonnatural amino acid
- Inclusion body prep Inclusion bodies were isolated through a series of wash steps. Frozen cell paste was re-suspended in wash buffer I (50 mM Tris, pH 8.0; 1% triton X-100; 100 mM NaCl, 1 mM EDTA) to a concentration of 10% (w/v) and homogenized at 4 °C followed by centrifugation (10,000g, 30 minutes, 4 °C). The supernatant was discarded, and the inclusion body pellet was re-suspended in an additional volume of wash buffer I followed by centrifugation at 15,000g for 30 minutes at 4 °C.
- wash buffer I 50 mM Tris, pH 8.0; 1% triton X-100; 100 mM NaCl, 1 mM EDTA
- the supernatant was discarded, and the inclusion body pellet was re-suspended in an additional volume of wash buffer I followed by centrifugation at 15,000g for 30 minutes at 4 °C.
- boIFN/3 was refolded by diluting solubilized material 1 : 10 into 20 mM tris, 1.0 M arginine, pH 8.0, 1 mM cysteine at 4 °C. After addition of solubilized material, the refolding reaction was allowed to incubate at RT for 16-24 hours.
- Example 8 Site Specific Conjugation of boIFNz.3 with PEG and boIFNX3-PEG Purification.
- BoIFNz.3 variants (SEQ ID NOS: 14-18 and 20-25) containing non-natural amino acid pAF were buffer exchanged into conjugation buffer (30 mM sodium acetate, 200 mM glycine, 2.5% trehalose, pH 4.0) and concentrated to 1-10 mg/mL. A final of 100 mM acetic hydrazide was added to the reactions followed by an 8-molar excess of aminooxy functionalized 30kDa PEG (NOF America Corp.). The conjugation reactions were incubated for 18-20 hours at 25-30 °C.
- the PEGylated boIFNz.3 was diluted 1 :10 with 30 mM sodium acetate, pH 5.0 and loaded over a Capto SP Impres column equilibrated with 25 mM potassium phosphate, pH 6.5.
- the loaded column was washed with 2-5 CV 25mM potassium phosphate, pH 6.5 and boIFNX3-PEG was eluted with a linear gradient over 20 CV to 25 mM potassium phosphate, pH 6.5; 0.5M NaCl.
- Fractions containing boIFNX3-PEG were collected and buffer exchanged into 20 mM potassium phosphate, pH 6.5; 100 mM NaCl; 2.5% trehalose.
- BoIFNz.3 was concentrated to 1-10 mg/mL, 0.22 pM filtered, and stored at -80 °C. Purified samples were characterized by SDS- PAGE, followed by reversed phase (RP) HPLC and size exclusion chromatography analysis (Table 4).
- Example 9 Real-time quantitative polymerase chain reaction (RT-qPCR) of ISG15 after treatment with PEGylated boIFNZ.3 variants in MDBK cells
- MDBK cells were treated with boIFNX3-PEG variants (500, 50, and 5 ng/mL). Cells were isolated 6 hours post treatment to evaluate relative mRNA expression of ISG15. Total RNA was isolated using TRIzol Reagent (Thermo Fisher Scientific) and reverse transcribed into cDNA using TaqManTM Fast Advanced Master Mix (Applied Biosystems) according to manufacturer's protocol. Relative fold changes in ISG15 gene expression were evaluated and normalized against non-treated control cells (FIG. 3).
- Example 10 MDBK ISRE-luciferase reporter assay.
- MDBK cells transfected with pGL4.45[luc2P/ISRE/Hygro] Vector (Promega, Cat. # E414A) were treated with boIFNX3-PEG variants (1000, 200, 40, 8, 16., 0.32, 0.64, and 0.0002 ng/mL).
- boIFNX3-PEG variants 1000, 200, 40, 8, 16., 0.32, 0.64, and 0.0002 ng/mL.
- luciferase luminescence was evaluate using ONE-GloTM Luciferase Assay System according to manufactures instructions (Promega, Cat. # E6120). Luciferase luminescence was normalized to untreated cells (FIG. 4). Variants that retained activity in this reporter assay were advanced to further testing.
- T119 (SEQ ID NO: 18) is exemplified in the following in vitro and in vivo studies.
- Example 11 In Vitro Antiviral Testing - Bovine Respiratory Disease Viruses
- Viral strains were purchased from the USDA National Veterinary Services Laboratory. Bovine viral diarrhea virus, Singer strain (cytopathic, genotype la (NVSL 140BDV); BRSV strain A51908 (NVSL 110BDV) and IBRV, also called bovine herpesvirus 1, Colorado strain (NVSL 050BDV) were propagated and titrated in Madin-Darby bovine kidney (MDBK) cells maintained in minimum essential medium supplemented with 10% fetal bovine serum, 2 mM L-glutamine, 0.05 mg/ml gentamicin, 10 unit/ml penicillin, 10 pg/ml streptomycin, and 0.25 pg/ml amphotericin.
- MDBK Madin-Darby bovine kidney
- BoIFN/3-TI 19-PEG30KDa prepared according to Example 8, was serially diluted in cell culture medium to generate concentrations of 1000, 250, 62.5, 15.63, 3.91, 0.98, 0.06 and 0 ng/ml.
- MDBK cells were cultured in 24-well plates (1 plate per virus). Duplicate wells containing each concentration of boIFN/3-T I I 9-PEG30KDa were prepared for each plate. Cells were cultured in the presence of boIFNX3-T119-PEG30KDa for approximately 18 hr prior to viral inoculation. Culture medium was removed from each well of MDBK cells and replaced with 300 pl of medium containing individual viruses.
- the supernatants of the cell lysates were serially diluted in culture medium and titrated in fresh MDBK cells cultured in 96-well plates (triplicate wells were used for each dilution). After 3-4 days, plates were observed for CPE rinsed and fixed with 80% cold acetone. Fifty pl of fluorescent antibody conjugates specific for each virus were added to each corresponding well and the plates were incubated for 1 hr at 37 °C. The antibody conjugate was discarded, and plates were washed with phosphate buffered saline.
- TCID50 tissue culture infective dose
- boIFN/3-T I 19-PEG30KDa completely inhibited replication of a cytopathic, genotype la BVDV strain at concentrations > 250 ng/ml, and reduced replication of BRSV by 20% to 30% at concentrations > 62.5 ng/ml.
- Example 12 In Vitro Antiviral Testing - Bovine Viral Diarrhea Virus Strains
- Bovine viral diarrhea virus (BVDV) strains were purchased from the USDA National Veterinary Service Laborator. Singer strain (cytopathic, genotype la (NVSL140BDV) and BVDV Draper strain (noncytopathic, genotype lb (NVSL 141BDV) were propagated and titrated in MDBK cells maintained in minimum essential medium supplemented with 10% fetal bovine serum, 2 mM L-glutamine, 0.05 mg/ml gentamicin, 10 unit/ml penicillin, 10 pg/ml streptomycin, and 0.25 pg/ml amphotericin.
- BVDV Bovine viral diarrhea virus
- BoIFN/3-TI 19-PEG30KDa prepared according to Example 8, was serially diluted in cell culture medium to generate concentrations of 1000, 250, 62.5, 15.63, 3.91, 0.98, 0.06 and 0 ng/ml.
- MDBK cells were cultured in 24-well plates (1 plate per virus strain). Duplicate wells containing each concentration of boIFN/3-T I 19-PEG30KDa were prepared for each plate. Cells were cultured in the presence of boIFN/3-T I 19-PEG30KDa for approximately 18 hr prior to viral inoculation. Culture medium was removed from each well of MDBK cells and replaced with 300 pl of medium containing individual viral strains.
- CPE cytopathic effect
- TCID50 tissue culture infective dose
- boIFNX3-T119-PEG30KDa completely inhibited replication of the cytopathic, genotype la and non-cytopathic, genotype lb strains of BVDV at concentrations > 250 ng/ml.
- boIFN/3-T I I 9-PEG30KDa exhibits antiviral activity against both cytopathic and non-cytopathic strains of BVDV associated with disease in cattle.
- Example 13 In Vitro Antiviral Testing - Cytopathic, Genotype 2 Bovine Viral Diarrhea Virus
- Cytopathic, genotype 2 strain 125 of BVDV was purchased from the USDA National Veterinary Service Laboratory. The virus was propagated and titrated in MDBK cells maintained in minimum essential medium supplemented with 10% fetal bovine serum, 2 mM L-glutamine, 0.05 mg/ml gentamicin, 10 unit/ml penicillin, 10 pg/ml streptomycin, and 0.25 pg/ml amphotericin.
- BoIFNX3-Tl 19-PEG30KDa prepared according to Example 8, was serially diluted in cell culture medium to generate concentrations of 1000, 500, 250 and 0 ng/ml.
- MDBK cells were cultured in 24-well plates. Triplicate wells containing each concentration of boIFNX3-PEG were prepared. Cells were cultured in the presence of boIFNX3-T119-PEG30KDa for approximately 18 hr prior to viral inoculation. Culture medium was removed from each well of MDBK cells and replaced with 300 pl of medium containing virus. Plates were incubated at 37 °C in 5% CO2 for 1 hr.
- the virus inoculum was removed from each well and 2 ml of culture medium containing the same concentrations of boIFNX3-Tl 19-PEG30KDa used initially was added to each corresponding well.
- the plates were incubated at 37 °C in 5% CO2 incubator for 24 hr, and cytopathic effect (CPE) development was recorded using a scale of 0-4 (0: no CPE; 1 : 25% CPE; 2: 50% CPE; 3: 75% CPE; 4: 100% CPE).
- CPE cytopathic effect
- the supernatants of the cell lysates were serially diluted in culture medium and titrated in fresh MDBK cells cultured in 96-well plates (triplicate wells were used for each dilution). After 3-4 days, plates were observed for CPE and the cells rinsed once and fixed with 80% cold acetone. Fifty pl of mouse-source anti-BVDV monoclonal antibody was added to each well and plates were incubated at 37 °C for 1 hr. The primary antibody was discarded, and the plates were washed with phosphate buffered saline (PBS).
- PBS phosphate buffered saline
- Fifty pl of the secondary antibody conjugate (goat anti-mouse IgG conjugated to FITC) was added to the wells and plates were incubated at 37 °C for 1 hr. The antibody conjugate was discarded, and plates were washed with PBS. Plates were read under fluorescence microscopy and virus infectious titers were calculated according to the Reed and Muench method (based on CPE and immunofluorescence staining results) and expressed as the 50% tissue culture infective dose (TCID50)/ml.
- Example 14 In Vitro Antiviral Testing - Vesicular Stomatitis Virus New Jersey
- VSV Vesicular Stomatitis virus
- PIADC Plum Island Animal Disease Center
- Example 15 In Vitro Antiviral Testing - Foot and Mouth Disease Virus
- Baby Hamster kidney cells strain 21 (BHK-21; ATCC CCL-10) were used to propoagate South African Territories (SAT)l of FMDV that was used for these experiments. Briefly, MDBK cells were seeded at 1.5xl0 5 cells/well and incubated at 37 °C and 5% CO2. After 24 hr, cells were treated with serial 2-fold dilutions of boIFNX3-T119-PEG30KDa in cell culture medium to generate concentrations from 0-157 ng/ml and incubated at 37 °C and 5% CO2.
- Example 17 Pharmacokinetic (PK) Analysis of boIFNZ.3-T I 19-PEG30kDa in Holstein- Friesian Calves.
- PK Pharmacokinetic
- the study design is summarized in Table 15. The study included 15 time points (0, 0.5, 1, 3, 6, 12, 24, 36, 48, 72, 96, 120, 168, 240, and 336 hours). Bioanalysis of plasma samples were performed using MSD platform utilizing electrochemiluminescence assay (ECLA).
- ECLA electrochemiluminescence assay
- PK data analysis was performed using WinNonlin software. The results are summarized in FIG. 6, which depicts the mean plasma concentration versus time profiles, and Table 16. PEG30kDa-boIFNZ.3-T I 19 showed dose-proportional increases in Cmax and exposure. Terminal half-life was similar between groups, ranging from 65-69 hours.
- Example 18 Differential scanning fluorimetry of boIFNZ.3-pAF and boIFNX3-pAF- PEG30kDa variants.
- Tm values were determined by calculating the temperature derivative of the melting curve.
- the resulting derivative curve was processed with the peak fitting algorithm in Bio-Rad CFX Manager 3.1, applying a sigmoidal baseline and fitting the peak to determine the Tm and its standard error.
- the data shows the thermal stability of boIFN/3-pAF and boIFNX3-pAF-PEG30kDa variants. Variants with thermal stability similar to wild type boIFNz.3 were preferred variants for further development.
- a conjugated protein comprising: an engineered bovine interferon Z.3 (boIFNZ.3) protein having an amino acid sequence comprising a non-natural amino acid; and a water-soluble polymer; wherein the water-soluble polymer is conjugated to the engineered boIFNz.3 protein.
- boIFNZ.3 engineered bovine interferon Z.3
- conjugated protein of any one of embodiments 1 to 8, wherein the non-natural amino acid is selected from the group consisting of 4-acetyl-L-phenylalanine (para-acetyl-L- phenylalanine (pAF)), 3-O-(N-acetyl-beta-D-glucosaminyl)-L-threonine, N4-( -N-Acetyl-D- glucosaminyl)-L-asparagine, O-allyl-L-tyrosine, alpha-N-acetylgalactosamine-O-L-serine, alpha- N-acetylgalactosamine-O-L-threonine, 2-aminooctanoic acid, 2-amino-L-phenylalanine, 3- amino-L-phenylalanine, 4-amino-L-phenylalanine, 2-amino-L-tyrosine,
- the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with:
- (ii) has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the engineered boIFNz.3 protein amino acid sequence further comprises an N-terminal methionine.
- conjugated protein of embodiment 16 wherein the covalent linkage comprises an amide, an ester, a thioester, a disulfide, an oxime, an imine, a pyrrolidine-2, 5-dione, a 1,2,3- triazole or a 1,4-dihydropyridazine, wherein the 1,2, 3 -triazole and the 1,4-dihydropyridazine are each optionally fused to an 8-membered ring.
- the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 18 or 19;
- the water-soluble polymer is a polyethylene glycol (PEG) or a derivative thereof; and the PEG or the derivative thereof is conjugated to the engineered boIFNz.3 protein via a covalent linkage with the non-natural amino acid.
- conjugated protein of any one of embodiments 1 to 38, wherein the engineered boIFNZ.3 protein is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA synthetase.
- conjugated protein of any one of embodiments 1 to 40 wherein the conjugated protein is characterized as having a plasma elimination half-life; and (i) the conjugated protein plasma elimination half-life is greater than the plasma elimination half-life of the unconjugated engineered boIFNz.3 protein; (ii) the conjugated protein plasma elimination half-life is greater than the plasma elimination half-life of wild-type boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 1; (iii) the conjugated protein plasma elimination half-life is at least about 10 hours in a Sprague-Dawley rat after administration of a single subcutaneous dose of 0.25 mg/kg; or (iv) the conjugated protein plasma elimination half-life is at least about 50 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 75 pg/kg.
- conjugated protein of any one of embodiments 1 to 40 wherein (i) the conjugated protein is characterized as having a plasma elimination half-life in a mammal, wherein the conjugated protein plasma elimination half-life is at least about 10-fold greater than the plasma elimination half-life of the corresponding unconjugated boIFN/3 protein in the mammal, or (ii) the conjugated protein is characterized as having a plasma elimination half-life in a mammal; wherein the conjugated protein plasma elimination half-life is at least about 10-fold greater than the plasma elimination half-life of wild-type boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 1 in the mammal.
- conjugated protein of any one of embodiments 1 to 40 wherein the conjugated protein is characterized as having a plasma elimination half-life in a mammal; wherein the conjugated protein plasma elimination half-life is at least about 10-fold greater than the plasma elimination half-life of an unconjugated IF NX protein or an unconjugated IFNy protein in the mammal.
- conjugated protein of any one of embodiments 1 to 40 or 45 wherein the conjugated protein is characterized as having a plasma elimination half-life in a mammal, wherein the mammal is a Holstein-Fresian calf, and wherein the conjugated protein plasma elimination half-life is at least about is at least about 50-fold greater than the plasma elimination half-life of (i) the corresponding unconjugated boIFNZ.3 protein; (ii) wild-type boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 1; (iii) an unconjugated IFNk protein; or (iv) an unconjugated IFNy protein in the mammal.
- conjugated protein of any one of embodiments 1 to 40 wherein the conjugated protein plasma elimination half-life is at least about 60 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 75 pg/kg.
- a pharmaceutical composition comprising a conjugated protein of any one of embodiments 1 to 49 and a pharmaceutically acceptable excipient or carrier.
- An engineered bovine interferon Z.3 (boIFNZ.3) protein a) wherein the engineered boIFNz.3 optionally comprises a non-natural amino acid; and wherein the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with:
- each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1; or b) wherein the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, and which shares less than 100% identity with SEQ ID NO: 1; and optionally wherein the boIFNz.3 protein further comprises an N-terminal methionine; and wherein: the boIFNz.3 protein amino acid sequence comprises a non-natural amino acid; or the boIFNz.3 protein amino acid sequence does not comprise a non-natural amino acid.
- the engineered boIFNz.3 protein of embodiment 51 wherein a) the engineered boIFNZ.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 5, or with SEQ ID NO: 5 further comprising the N-terminal methionine or b) the engineered boIFNz.3 protein has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 5, and optionally further comprises an N-terminal methionine.
- the engineered boIFNz.3 protein of embodiment 51 or 52, wherein the boIFNz.3 protein amino acid sequence comprises a non-natural amino acid optionally wherein: a) the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 5, in which a non- natural amino acid is substituted for the amino acid at one or more position of that sequence, preferably a non-natural amino acid is substituted for the amino acid at one position of that sequence; b) the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 5 further comprising the N-terminal methionine, in which a non-natural amino acid is substituted for the amino acid at one or more position of that sequence, preferably a non-natural amino acid is substituted for the amino acid at one position of that sequence; c) the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 6, in which a non- natural amino acid is substituted for the amino acid at one or more
- a vector comprising a nucleic acid encoding any one of SEQ ID NOs: 2 to 24.
- a vector comprising a nucleic acid encoding SEQ ID NO: 5.
- a vector comprising a nucleic acid encoding SEQ ID NO: 18.
- a vector comprising a nucleic acid encoding SEQ ID NO: 19.
- An engineered bovine interferon Z.3 (boIFNz.3) protein comprising a non-natural amino acid; wherein (a) the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with:
- (ii) has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNz.3 protein amino acid sequence further comprises an N-terminal methionine.
- a prophylactic method of treating a disease or condition in a mammal comprising administering to the mammal a conjugated protein of any one of embodiments 1 to 49 or a pharmaceutical composition of embodiment 50.
- BVDV bovine viral diarrhea virus
- PI3 parainfluenza-3
- BRSV bovine respiratory syncytial virus
- antibiotic is a penicillin, a cephalosporin, a macrolide, a tetracycline antibiotic or a fluoroquinolone antibiotic; or a combination thereof.
- the antibiotic is a cephalosporin
- the cephalosporin is selected from the group consisting of cefachlor, cefadroxil, cefamandole, cefazolin, cefepime, cefoperazone, ceforanide, cefotaxime, cefotiam, cefpodoxime, ceftazidime, cefovecin, cefoxitin (a cephamycin), cefsulodin, ceftiofur, ceftriaxone, cefuroxime, cephalexin, cephaloridine, cephalothin, cephapirin and cephradine; and combinations thereof.
- the antibiotic is a macrolide
- the macrolide is selected from the group consisting of azithromycin, erythromycin, erythromycin A, erythromycin B, erythromycin C, erythromycin D, erythromycin E, gamithromycin, tilmicosin, tulathromycin, tildipirosin and tylosin; and combinations thereof.
- a pharmaceutical composition comprising a conjugated protein of any one of embodiments 1 to 49 and an antibiotic for the treatment of a bovine respiratory disease.
- a method of inducing systemic antiviral activity in a mammal comprising administering to the mammal a conjugated protein of any one of embodiments 1 to 49 or a pharmaceutical composition of embodiment 50.
- the method of embodiment 110, wherein the vaccine is a FMD antigen, an inactivated whole FMDV antigen, a modified live vaccine or an adenovirus type 5 (Ad5) FMD (Ad5-FMD) vaccine.
- Ad5-FMD adenovirus type 5
- 112. The method of embodiment 110 or 111, wherein the method induces (i) anti- FMDV activity; (ii) adjuvanted adaptive immune responses against FMDV; and/or (iii) expression of interferon-stimulated genes correlated with systemic control of viral replication in an animal susceptible to FMDV in vivo; thereby delaying, reducing severity and/or preventing foot and mouth disease.
- the protection begins within about 1 day after the administration of the conjugated protein
- the protection begins within about 2 days after the administration of the conjugated protein
- the protection begins within about 3 days after the administration of the conjugated protein.
- 116 The method of embodiment 114 or 115, wherein the method comprises administration of a FMDV vaccine, and the protection begins prior to the induction of protective antibody titers in the mammal, wherein the protective antibody titers are induced by the administration of the vaccine.
- 123 The method of embodiment 122, wherein the virus associated with BRD is selected from the group consisting of bovine viral diarrhea virus (BVDV), bovine respiratory syncytial virus (BRSV), a bovine coronavirus and a parainfluenza-3 virus (PI-3); and combinations thereof.
- BVDV bovine viral diarrhea virus
- BRSV bovine respiratory syncytial virus
- PI-3 parainfluenza-3 virus
- 124 The method of embodiment 121, 122 or 123, wherein the protection is characterized by an absence of viremia.
- Conditional language such as “can,” “could,” “might,” or “rnay,” unless specifically stated otherwise, or otherwise understood within the context as used, is generally intended to convey that certain embodiments include, while other embodiments do not include, certain features, elements, and/or steps. Thus, such conditional language is not generally intended to imply that features, elements, and/or steps are in any way required for one or more embodiments or that one or more embodiments necessarily include logic for deciding, with or without user input or prompting, whether these features, elements, and/or steps are included or are to be performed in any particular embodiment.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Biochemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Engineering & Computer Science (AREA)
- Veterinary Medicine (AREA)
- Toxicology (AREA)
- Zoology (AREA)
- Gastroenterology & Hepatology (AREA)
- Epidemiology (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Peptides Or Proteins (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
A long-acting conjugated protein comprising a bovine interferon lambda-3 protein conjugated to a water-soluble polymer moiety via a non-naturally encoded amino acid in the protein is described. Also described are methods of protecting ruminant mammals from foot and mouth disease or bovine respiratory disease by administering the long-acting conjugated protein. Bovine interferon lambda-3 protein variants, and variants containing non-naturally encoded amino acids suitable for conjugation to moieties that are capable of extending bovine interferon lambda-3 protein elimination half-life, such as water-soluble polymers, are also described.
Description
PEGYLATED BOVINE INTERFERON LAMBDA AND METHODS OF USE THEREOF
REFERENCE TO RELATED APPLICATIONS
[0001] The present application claims priority to U.S. Provisional Application No. 63/504,133, filed on May 24, 2023, the entire contents of which are hereby incorporated herein in their entirety.
SEQUENCE LISTING
[0002] The present application contains a Sequence Listing which has been submitted in XML format and is hereby incorporated by reference in its entirety. The XML copy, created on October 3, 2023, is named P086609WO_Sequence_Listing and is 34,541 bytes in size.
FIELD OF THE INVENTION
[0003] This invention relates to conjugated proteins. In particular, the invention relates to engineered bovine interferon lambda-3 protein conjugated to a water-soluble polymer moiety via a non-natural amino acid incorporated into the protein, and methods of use thereof.
BACKGROUND OF THE INVENTION
[0004] Infectious diseases associated with viral pathogens have a profound negative economic impact upon food animal production. In addition to negatively impacting the health and welfare of the animals, these diseases increase production costs and reduce profits for livestock producers. Two significant infectious diseases of beef and dairy cattle associated with viruses include bovine respiratory disease and foot and mouth disease.
[0005] Bovine respiratory disease (BRD) has been referred to as a “disease complex” since it is typically associated with both viral and bacterial pathogens that interact with one another to produce the acute clinical disease (see Taylor J.D. et al. (2010) Can. Vet. J. 51(10): 1095— 1102). Single or mixed viral infections that occur during periods of stress, such as weaning and commingling of animals from different sources prior to shipping to feedyards, include bovine viral diarrhea virus (BVDV), infectious bovine rhinotracheitis virus (IBR), parainfluenza-3 (PI3) and bovine respiratory syncytial virus (BRSV) (Gagea M.I. et al. (2006) J. Vet. Diagn. Invest. 18: 18— 28). The viruses, in addition to environmental stressors, contribute to suppression of an animal’s immune responses (Lillie L.E. (1974) Can. Vet. J. 15:233-242). The reduction in protective immunity results in increased susceptibility to secondary invasion of the respiratory tract by bacteria that can establish pneumonia in the lungs of affected animals, leading to morbidity and mortality. Cattle arriving in feedyards typically exhibit 15-45% morbidity and 1-15% mortality due to respiratory disease (Kelly A.P. and Janzen E.D. (1986) Can. Vet. J. 27(12):496-500).
[0006] It is widely recognized that elimination of these viral and bacterial pathogens from the environment is impossible to achieve. Therefore, control of BRD is currently focused on preventing the pathogens from establishing infections, or diagnosis and treatment of secondary bacterial pneumonia as quickly and effectively as possible.
[0007] Vaccination represents the primary method of controlling respiratory viral infections in beef and dairy cattle (Urban-Chmiel R. and Grooms, D.L. (2012) J. Livestock Sci. 3:27-36). Effective vaccination protocols require the use of vaccines that can elicit an antibody response in the host animal to neutralize the specific viral pathogen affecting the herd. Because viruses mutate over time, resulting in changes in their susceptibility to the antibodies generated in response to vaccines, the vaccines must be updated periodically to ensure that the antibodies are capable of neutralizing the viruses that are present in the current environment. The efficacy of viral vaccination can also be compromised by the timing of administration of the vaccine relative to the period(s) of stress when viral infections are most prevalent (Richeson J.T. et al. (2008) J. An. Sci. 86(4):999-1005). If animals become infected prior to completion of the vaccination protocol or before protective antibody titers induced by vaccination are present, viral induced disease is still likely to occur. Similarly, administration of vaccines to very young animals can be problematic due to the presence of maternal antibodies absorbed from the colostrum of the dam, which limit the ability of the calf to generate a protective immune response following vaccination (Ellis J. et al. (2001) J. Am. Vet. Med. Assoc. 219:351-356). Treatment of the secondary bacterial infections is typically accomplished through the administration of antibiotic therapy. Hopes that antibiotics would allow complete control of the disease have not been realized. Development of antimicrobial resistance to antibiotics commonly used to control respiratory disease bacteria has been documented (Welsh R.D. et al. (2004) J. Vet. Diagn. Invest. 16:426-431). Additionally, consumer preferences and regulatory agency trends are moving towards identifying alternatives to antibiotics to reduce the likelihood of antimicrobial resistance development against antibiotics used in human health.
[0008] Another devastating disease that can impact cloven-hoofed animals is foot and mouth disease (FMD), which is caused by foot-and-mouth disease virus (FMDV), the most contagious pathogen among these livestock, including cattle. Animals infected with FMDV develop acute disease that spreads rapidly and is characterized by fever, lameness and lesions on the feet, tongue, snout and teats (Grubman and Baxt (2004) Clin Microbiol Rev, 17:465-493). Cattle infected with FMDV exhibit high morbidity, but low mortality. In regions other than North America, Western Europe, New Zealand and Australia, FMDV is enzootic. Disease control is achieved by prohibition of animal movement, slaughter of symptomatic and in-contact animals, disinfection of
contaminated facilities and vaccination with a killed viral vaccine. Current vaccines do not induce protective antibody titers until 5-7 days post-vaccination; thus, vaccinated animals remain susceptible to infection with FMDV during this period (Diaz-San Segundo F. et al. (2011) Virology 413:283-292). Due to technical limitations, it is difficult to differentiate FMDV vaccinated animals from infected animals. Therefore, there are significant restrictions on movement of animals and animal-derived products from FMDV positive countries (OIE, 2007. Terrestrial animal health code. Foot and Mouth Disease, Chapter 2.2.10). Acute losses associated with the shortcomings of existing control measures, along with severe trade restrictions limiting exports, would result in catastrophic economic damage should a FMDV outbreak occur in a FMDV-free region.
[0009] Interferons (IFNs) are naturally occurring cytokines that function as a first line of defense against viral infection. Type I IFNs (IFN-a/p) were the first to be discovered and are produced and secreted by virus-infected cells of many types. Type I IFNs bind to receptors on many different cell types, resulting in the expression of gene products that control cell death and impair viral replication. These IFNs also activate both innate and adaptive immune cells (Lopusna K., et al. (2013) Acta. Virologica 57: 171-179). Type III IFNs are the most recently described IFNs (IFN- I, IFN-X2, IFN-Z.3) and have been identified in several species other than humans, including cattle and swine (Segundo 2011, Id. Sang, Y. et al. (2010) J. Interferon Cytokine Res. 30: 1-7). Type III IFNs are structurally distinct from type I IFNs (~5% amino acid sequence identity) but possess intrinsic antiviral activity (Donnelly R and Kotenko S. (2010) Interferon & Cytokine Res. 30:555- 564). Expression of Type III IFN receptors is limited to epithelial cells, suggesting a pivotal role in protection of mucosal surfaces (Lin J.D. et al. (2016) PLOS Pathog. 12(4):el005600).
[0010] FMDV is highly sensitive to the action of IFNs (Chinsangaram et al. (1999) J. Virol. 73:9891-9898; Chinsangaram et al. (2001) J. Virol. 75:5498-5503; Moraes et al. (2007) J. Virol. 81 :7124-7135; U.S. Patent No. 8,906,384 B2; the entire contents of each of which are hereby incorporated by reference herein in their entirety). Although vaccination is currently being used to control rapidly spreading FMDV, the development of a protective adaptive immune response in vaccinated animals still takes 5 to 7 days. It would be advantageous to control FMD outbreaks by attenuating viral replication during this lag period, which might be achieved by inducing early and sustained IFN activity. In some studies, expression of type I, II or III IFNs after administration of an Ad5 vector platform was shown to provide early protection against FMDV in swine (Chinsangaram et al. (2003) J. Virol. 77:1621-1625; Moraes (2007), Id.; Dias et al. (2011) J. Interferon Cytokine Res. 31 :227-236; Perez-Martin et al. (2014) J. Interferon Cytokine Res. 34:810-821), but only partial protection was achieved in cattle (Wu et al. (2003) J. Interferon
Cytokine Res. 23:371-380; Perez-Martin (2012) J. Virol. 86:4477-4487). It is known in the art that IFNs are generally short-lived species in circulation (see, e.g., Bansal, R. et al. (2011) J. Controlled Release 154(3):233-240). Indeed, the relatively short half-life of IFN protein in vivo has hampered efforts to induce the protective response that is needed to control FMD in cattle. [0011] Methods of modulating the biophysical properties and/or biological activity of a biotherapeutic via conjugation with another moiety are known in the art, including PEGylation. PEGylation involves the covalent attachment of poly(ethylene glycol) (PEG) to a biotherapeutic, and can be used to modulate a target biotherapeutic’ s water solubility, bioavailability, half-life, immunogenicity and/or biological activity. Methods and reagents for PEGylation have been described, for example, in U.S. Pat. Nos. 6,610,281 and 6,602,498, and “Polyethylene Glycol and Derivatives for Advanced PEGylation”, Nektar Molecular Engineering Catalog, 2003, pp. 1-17, the entire contents of each of which are hereby incorporated by reference herein in their entirety. PEGylated porcine IFN-alpha has been studied for the treatment of FMD in swine (Diaz-San Segundo et al. (2021) Frontiers in Microbiology, V12; Article 668890), but unfortunately, IFN- alpha does not protect cattle against FMD or other diseases. Moreover, PEG moieties are typically covalently linked to a target biotherapeutic via its naturally-occuring amino acids, such as lysine, cysteine, histidine or the N-terminus, and/or to any carbohydrate moieties present in the biotherapeutic. Reactive sites that may seem suitable for PEGylation may play a significant role in receptor binding; thus, indiscriminate attachment of polymer chains such as PEG to such reactive sites on a biotherapeutic can lead to a significant reduction or even total loss of its biological activity (Clark, R. et al. (1996) J. Biol. Chem. 271 :21969-21977). PEG derivatives can also undergo side reactions with residues other than those targeted for modification, which can create complex, heterogeneous mixtures of PEG-derivatized biotherapeutics having reduced biological activity.
[0012] One technology which promises to overcome many of these limitations is the incorporation of non-natural amino acids into proteins; see, e.g., Wang L. et al. (2001) Science 292:498-500; Chin J. et al. (2003) Science 301 :964-7; Chin J. W. et al. (2002) Proc. Natl. Acad. Sci. U.S. A. 99: 11020-11024; Chin J. W. et al. (2002) J. Am. Chem. Soc. 124:9026-9027; Chin J.W. and Schultz P.G. (2002) ChemBioChem 3(11): 1135-1137; Wang L. and Schultz P.G. (2002), Chem. Comm. 1 : 1-11; Tian F. et al. (2014) Proc. Natl. Acad. Sci. U.S.A 111(5): 1766-1771; and U.S. patent no. 7,083,970; the entire contents of each of which are hereby incorporated by reference herein in their entirety. These and other studies have demonstrated that it is possible to site- specifically introduce into a protein a non-natural amino acid containing a chemical functional group that is not found in the 20 common, genetically-encoded amino acids, that is chemically
inert to all of the functional groups found in the 20 common, genetically-encoded amino acids, and that can be used to react efficiently and selectively to form stable covalent linkages with moieties, such as water-soluble polymer moieities, that are chosen for conjugation with the protein (e.g., biotherapeutic) of interest.
[0013] There remains a need for compositions and methods for controlling BRD and FMD outbreaks. Such compositions and methods would block viral replication during the lag period required for cloven-hoofed animals to develop a vaccine-stimulated adaptive immune response. The present disclosure fulfills this need.
SUMMARY OF THE INVENTION
[0014] Disclosed herein are conjugated proteins comprising engineered bovine interferon 3 (boIFNZ.3) protein joined to water-soluble polymer(s) through one or more non-natural amino acids, and methods of making the same. Also described are methods of treating a disease or condition in a mammal via administration of a conjugated protein of the present disclosure, or a composition comprising a conjugated protein of the present disclosure.
[0015] In some general aspects, the present disclosure provides a conjugated protein comprising: an engineered bovine interferon 3 (boIFNZ.3) protein comprising a non-natural amino acid; and a water-soluble polymer; wherein the water-soluble polymer is conjugated to the engineered boIFNz.3 protein. In some embodiments, the water-soluble polymer is conjugated to the engineered boIFNZ.3 protein via the non-natural amino acid. In some embodiments, the water-soluble polymer is conjugated to the engineered boIFNz.3 protein via a covalent linkage with the non-natural amino acid. In some embodiments, the water-soluble polymer is one or more water-soluble polymers, the non-natural amino acid is one or more non-natural amino acids, and each of the one or more water-soluble polymers is conjugated to the engineered boIFNz.3 protein via each of the one or more non-natural amino acids. In some other embodiments, the one or more water-soluble polymers is one water-soluble polymer, and the one or more non-natural amino acids is one non- natural amino acid.
[0016] In some embodiments, the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24. In some embodiments, the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine. In some embodiments (a) the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with: (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine; or
(b) the engineered boIFNz.3 protein (i) has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the engineered boIFNz.3 protein amino acid sequence further comprises an N-terminal methionine. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 90% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNZ.3 protein amino acid sequence shares at least 90% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 96% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 96% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 97% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 97% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 98% identity with SEQ ID NO:
18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 98% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO:
19. In some embodiments, the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 19.
[0017] In some embodiments, the non-natural amino acid is selected from the group consisting of 4-acetyl-L-phenylalanine (para-acetyl-L-phenylalanine (pAF)), 3-O-(N-acetyl-beta-D- glucosaminyl)-L-threonine, N4-(P-N-Acetyl-D-glucosaminyl)-L-asparagine, O-allyl-L-tyrosine, alpha-N-acetylgalactosamine-O-L-serine, alpha-N-acetylgalactosamine-O-L-threonine, 2- aminooctanoic acid, 2-amino-L-phenylalanine, 3-amino-L-phenylalanine, 4-amino-L- phenylalanine, 2-amino-L-tyrosine, 3-amino-L-tyrosine, 4-azido-L-phenylalanine, 4-benzoyl-L- phenylalanine, (2,2-bipyridin-5yl)-L-alanine, 3-borono-L-phenylalanine, 4-borono-L- phenylalanine, 4-bromo-L-phenylalanine, p-carboxymethyl-L-phenylalanine, 4-carboxy-L- phenylalanine, p-cyano-L-phenylalanine, 3,4-dihydroxy-L-phenylalanine (L-DOPA), 4-ethynyl- L-phenylalanine, 2-fluoro-L-phenylalanine, 3-fluoro-L-phenylalanine, 4-fluoro-L-phenylalanine, O-(3-O-D-galactosyl-N-acetyl-beta-D-galactosaminyl)-L-serine, L-homoglutamine, (8-
hydroxyquinolin-3-yl)-L-alanine, 4-iodo-L-phenylalanine, 4-isopropyl-L-phenylalanine, 0-i- propyl-L-tyrosine, 3-isopropyl-L-tyrosine, O-mannopyranosyl-L-serine, 2-methoxy-L- phenylalanine, 3 -m ethoxy -L-phenylalanine, 4-m ethoxy -L-phenylalanine, 3-methyl-L- phenylalanine, O-methyl-L-tyrosine, 3-(2-naphthyl)-L-alanine, 5-nitro-L-histidine, 4-nitro-L- histidine, 4-nitro-L-leucine, 2-nitro-L-phenylalanine, 3-nitro-L-phenylalanine, 4-nitro-L- phenylalanine, 4-nitro-L-tryptophan, 5-nitro-L-tryptophan, 6-nitro-L-tryptophan, 7-nitro-L- tryptophan, 2-nitro-L-tyrosine, 3-nitro-L-tyrosine, O-phospho-L-serine, O-phospho-L-tyrosine, 4- propargyloxy -L-phenylalanine, O-2-propyn-l-yl-L-tyrosine, 4-sulfo-L-phenylalanine and O- sulfo-L-tyrosine. In some embodiments, the non-natural amino acid is para-acetyl-L- phenylalanine (pAF).
[0018] In some embodiments, the water-soluble polymer is selected from the group consisting of a carbohydrate, a polypeptide and a polyalkylene glycol; and derivatives thereof. In some embodiments, the water-soluble polymer is a polyalkylene glycol or a derivative thereof. In some embodiments, the polyalkylene glycol, or the derivative thereof, is a polyethylene glycol (PEG), or a derivative thereof. In some embodiments, the polyalkylene glycol or derivative thereof is a linear PEG or a derivative thereof. In some embodiments, the PEG or the derivative thereof has a molecular weight of at least about 0.1 kDa and at most about 100 kDa. In some embodiments, the PEG or the derivative thereof has a molecular weight of at most about 50 kDa. In some embodiments, the PEG or the derivative thereof has a molecular weight within a range of about 10 kDa to about 50 kDa, about 20 kDa to about 40 kDa, or about 25 kDa to about 35 kDa. In some embodiments, the PEG or the derivative thereof has a molecular weight of about 30 kDa. In some embodiments, prior to the conjugation with the engineered boIFNZ.3 protein, the water-soluble polymer is a PEG derivative comprising an aminooxy group.
[0019] In some embodiments, the covalent linkage conjugating the water-soluble polymer to the non-natural amino acid comprises an amide, an ester, a thioester, a disulfide, an oxime, an imine, a pyrrolidine-2, 5-dione, a 1,2,3-triazole or a 1,4-dihydropyridazine. In some embodiments, the 1,2,3-triazole is fused to an 8-membered ring. In some embodiments, the 1,4-dihydropyridazine is fused to an 8-membered ring. In some embodiments, the covalent linkage comprises an oxime.
[0020] In some embodiments, the engineered boIFNz.3 protein is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA synthetase. In some embodiments, the non-natural amino acid is site- specifically incorporated into the protein.
[0021] In some embodiments, the conjugated boIFNz.3 protein is characterized as having a plasma elimination half-life. In some embodiments, the conjugated boIFNz.3 protein plasma elimination
half-life is greater than the plasma elimination half-life of the unconjugated engineered boIFNz.3 protein (e.g., the corresponding unconjugated engineered boIFNz.3 protein). In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is greater than the plasma elimination half-life of wild-type boIFNz.3 protein. In some embodiments, the conjugated boIFNZ.3 protein plasma elimination half-life is greater than the plasma elimination half-life of wild-type boIFNz.3 protein having the amino acid sequence of SEQ ID NO: 1. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 10 hours in a rat. In some embodiments, the conjugated boIFNz.3 protein plasma elimination halflife is at least about 10 hours in a Sprague-Dawley rat after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein to the rat, wherein the dose is within a range of about 0.1 mg/kg to about 1.0 mg/kg body weight of the rat. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 10 hours in a Sprague-Dawley rat after administration of a single subcutaneous dose, wherein the dose is about 0.25 mg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 40 hours in cattle. In some embodiments, the conjugated boIFNz.3 protein plasma elimination halflife is at least about 50 hours in cattle. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 40 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is within a range of about 50 pg/kg to about 500 pg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 50 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is within a range of about 50 pg/kg to about 500 pg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 40 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is within a range of about 75 pg/kg to about 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 50 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is within a range of about 75 pg/kg to about 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 40 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is about 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 50 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is about 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 40 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is about 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma
elimination half-life is at least about 50 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is about 150 pg/kg.
[0022] In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 60 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is within a range of about 75 pg/kg to about 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least about 60 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is about 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein plasma elimination half-life is at least 60 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose, wherein the dose is about 150 pg/kg.
[0023] In some other general aspects, the present disclosure provides a pharmaceutical composition comprising a conjugated boIFNZ.3 protein of the present disclosure and a pharmaceutically acceptable excipient or carrier.
[0024] In some other general aspects, the present disclosure provides an engineered boIFNz.3 protein; wherein the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with: (i) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; or (ii) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N- terminal methionine; and wherein each said amino acid sequence shares less than 100% identity with SEQ ID NO: 1. Alternatively the engineered boIFNz.3 protein comprises an amino acid sequence that shares at least 90% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, but which shares less than 100% identity with SEQ ID NO: 1; optionally wherein the engineered boIFNZ.3 protein amino acid sequence further comprises an N-terminal methionine. In some embodiments, the engineered boIFNz.3 protein comprises a non-natural amino acid. In some embodiments, the amino acid sequence shares at least 90% identity with SEQ ID NO: 5. In some embodiments, the amino acid sequence shares at least 90% identity with SEQ ID NO: 5 further comprising the N-terminal methionine. In some embodiments, the amino acid sequence shares at least 95% identity with SEQ ID NO: 5. In some embodiments, the amino acid sequence shares at least 95% identity with SEQ ID NO: 5 further comprising the N-terminal methionine. In some embodiments, the amino acid sequence shares at least 96% identity with SEQ ID NO: 5. In some embodiments, the amino acid sequence shares at least 96% identity with SEQ ID NO: 5 further comprising the N-terminal methionine. In some embodiments, the amino acid sequence shares at least 97% identity with SEQ ID NO: 5. In some embodiments, the amino acid sequence shares at least 97% identity with SEQ ID NO: 5 further comprising the N-terminal methionine. In some embodiments, the amino acid sequence shares at least 98% identity with SEQ ID NO: 5. In some
embodiments, the amino acid sequence shares at least 98% identity with SEQ ID NO: 5 further comprising the N-terminal methionine. In some embodiments, the amino acid sequence shares at least 99% identity with SEQ ID NO: 5. In some embodiments, the amino acid sequence shares at least 99% identity with SEQ ID NO: 5 further comprising the N-terminal methionine. In some embodiments, the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 5. In some embodiments, the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 5 further comprising an N-terminal methionine. In some embodiments, the engineered boIFNZ.3 protein does not contain a non-natural amino acid. In all cases the engineered boIFNz.3 protein can be optionally defined as comprising an amino acid that shares the given percentage identity (e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99% identity with SEQ ID NO: 5), optionally wherein the engineered boIFNZ.3 protein amino acid sequence further comprises an N- terminal methionine.
[0025] In yet some other general aspects, the present disclosure provides an engineered boIFNz.3 protein comprising a non-natural amino acid; wherein the engineered boIFNZ.3 protein has an amino acid sequence that shares at least 90% identity with: (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine. Alternatively, the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with: (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24, or (ii) has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the engineered boIFNz.3 protein amino acid sequence further comprises an N-terminal methionine. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 96% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 97% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 98% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 18. In some other embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 96% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 97% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 98% identity with SEQ ID NO:
19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least
99% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 19. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine. In some embodiments, the engineered boIFNz.3 protein is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA, and the non-natural amino acid is site- specifically incorporated into the protein.
[0026] In some other general aspects, the present disclosure provides a pharmaceutical composition comprising an engineered boIFNZ.3 protein of the present disclosure and a pharmaceutically acceptable excipient or carrier.
[0027] In some other general aspects, the present disclosure provides a nucleic acid having the nucleotide sequence of SEQ ID NO: 27. In other aspects, the present disclosure provides a nucleic acid molecule having a nucleotide sequence encoding any of the engineered boIFNz.3 proteins referred to herein (e.g. a nucleic acid molecule encoding any one of SEQ ID NOS: 2 to 24).
[0028] In yet some other general aspects, the present disclosure provides vector comprising a nucleic acid molecule encoding any one of SEQ ID NOS: 2 to 24. In some embodiments, the vector comprises a nucleic acid molecule encoding SEQ ID NO: 4. In some embodiments, the vector comprises a nucleic acid molecule encoding SEQ ID NO: 5. In some embodiments, the vector comprises a nucleic acid molecule encoding SEQ ID NO: 6. In some embodiments, the vector comprises a nucleic acid molecule encoding SEQ ID NO: 7. In some embodiments, the vector comprises a nucleic acid molecule encoding SEQ ID NO: 18. In some embodiments, the vector comprises a nucleic acid molecule encoding SEQ ID NO: 19.
[0029] In some other general aspects, the present disclosure provides a method of treating a disease or condition in a mammal. In some embodiments, the method is a prophylactic method. In some embodiments, the method comprises administering to the mammal a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a conjugated boIFNz.3 protein of the present disclosure. In some embodiments, the mammal is a ruminant mammal. In some embodiments, the ruminant mammal is bovine. In some embodiments, the ruminant mammal is a cattle. In some embodiments, the ruminant mammal is a calf.
[0030] In some embodiments, the method of treating a disease or condition in a mammal provides the mammal with protection against the disease or condition. In some embodiments, the method induces systemic antiviral activity in the mammal. In some embodiments, the method induces systemic antiviral activity in the mammal, such as systemic antiviral activity against a virus that is the cause of or is associated with the disease or condition. In some embodiments, the
method provides protection against infection by a pathogen, such as a virus, that is the cause of or is associated with the disease or condition. In some embodiments, the method provides protection against the disease or condition. In some embodiments, the method provides protection against symptoms of the disease or condition. In some embodiments, the method reduces a rate of infection by the causative or associated virus. In some embodiments, the method reduces a degree of infection by the causative or associated virus. In some embodiments, the method inhibits replication of the causative or associated virus. In some embodiments, the method delays onset of the disease or condition. In some embodiments, the method reduces severity of the disease or condition. In some embodiments, the method reduces severity of symptoms of the disease or condition. In some embodiments, the method prevents the disease or condition from occurring. In some embodiments, the method prevents infection by the causative or associated virus. In some embodiments, the method reduces the rate of transmission of the causative or associated virus from one ruminant mammal to another ruminant mammal or mammals. In some embodiments, the method prevents the transmission of the causative or associated virus from one ruminant mammal to another ruminant mammal or mammals. In some embodiments, the method further comprises administering a vaccine to the ruminant mammal, wherein the vaccine is capable of preventing the disease or condition. In some embodiments the method induces (i) anti-viral activity against the causative or associated virus; (ii) adjuvanted adaptive immune responses against the causative or associated virus; and/or (iii) expression of interferon-stimulated genes correlated with systemic control of viral replication in a mammal susceptible to infection by the causative or associated virus in vivo; thereby delaying, reducing severity and/or preventing the disease or condition.
[0031] In some embodiments, the disease or condition being treated is a bovine respiratory disease. In some embodiments, the disease or condition is associated with bovine viral diarrhea virus (BVDV), infectious bovine rhinotracheitis virus (IBR), parainfluenza-3 (PI3) or bovine respiratory syncytial virus (BRSV); or a combination thereof. In some embodiments, the disease or condition is associated with BVDV, PI3 or BRSV; or a combination thereof. In some embodiments, the method of treating a disease or condition in a mammal further comprises administering to the mammal a vaccine that is capable of preventing infection by BVDV, IBR, PI3 or BRSV.
[0032] In some other embodiments, the disease or condition being treated is foot and mouth disease. In some embodiments, the disease or condition is associated with foot and mouth disease virus (FMDV). In some embodiments, the method of treating a disease or condition in a mammal further comprises administering to the mammal a vaccine that is capable of preventing infection by FMDV.
[0033] In some embodiments, the method of treating the disease or condition comprises administering a conjugated boIFNZ.3 protein of the present disclosure to a ruminant mammal, wherein the dose is within a range of about 50 pg/kg to about 500 pg/kg of the body weight of the mammal. In some embodiments, the dose is within a range of about 75 pg/kg to about 150 pg/kg of the body weight of the mammal. In some embodiments, the dose is within a range of about 100 pg/kg to about 200 pg/kg of the body weight of the mammal. In some embodiments, the ruminant mammal is bovine. In some embodiments, the ruminant mammal is a cattle. In some embodiments, the ruminant mammal is a calf.
INCORPORATION BY REFERENCE
[0034] All publications, patents, patent applications and/or other documents mentioned herein are incorporated herein by reference in their entirety for all purposes and to the same extent as if each individual publication, patent, patent application and/or other document was specifically and individually indicated to be incorporated by reference for all purposes, and for the purpose of describing and disclosing, for example, the compositions and other methodologies that are described in the publications, patents, patent applications and/or other documents, which might be used in connection with the presently described inventions. The publications discussed herein are provided solely for their disclosure prior to the filing date of the present application.
BRIEF DESCRIPTION OF THE DRAWINGS
[0035] FIG. 1 shows E. coli plasmid map (A) pKGOlOO for expression of wild type boIFNZ.3; and (B) pKG0168 for expression of engineered boIFNZ.3 containing a non-naturally encoded amino acid.
[0036] FIG. 2 shows a boIFNZ.3 3 -dimensional model with amino acids selected for non-natural amino acid incorporation.
[0037] FIG. 3 shows real-time quantitative polymerase chain reaction (RT-qPCR) of ISG15 after treatment with boIFNk3-PEG variants in MDBK cells.
[0038] FIG. 4 shows MDBK ISRE-luciferase reporter assay after treatment with PEGylated boIFN/3 (boIFN/3-PEG) variants for 7 hours.
[0039] FIG. 5 shows mean plasma concentration versus time profiles for boIFN/3-PEG variants in male Sprague-Dawley rats.
[0040] FIG. 6 shows mean plasma concentration versus time profiles for boIFN/3-T I 19- PEG30kDa in Holstein-Friesian Calves.
[0041] FIG. 7 shows shows thermal melting temperatures of (A) boIFNk3-pAF variants and (B) boIFNX3-pAF-PEG variants.
DETAILED DESCRIPTION
[0042] The present disclosure provides a conjugated bovine interferon lambda-3 (boIFNX3) protein, which is sometimes referred to herein simply as a “conjugated protein.” The conjugated protein comprises an engineered boIFNz.3 protein conjugated to a moiety that is capable of extending boIFNz.3 protein elimination half-life, such as a water-soluble polymer. In some embodiments, a water-soluble polymer is conjugated to the engineered boIFNz.3 protein via a nonnatural amino acid in the protein. In some embodiments, the water-soluble polymer is a polyalkylene glycol, such as a polyethylene glycol (PEG) or PEG derivative. Also provided are antiviral compositions containing the conjugated boIFNz.3 protein. In some embodiments, a conjugated boIFNz.3 protein of the present disclosure, or a composition containing the conjugated boIFNz.3 protein of the present disclosure, can be used in ruminant mammals such as cattle as a method of treating (e.g., protecting the mammals from) a disease or condition associated with or caused by a virus. In some embodiments, the disease or condition is foot and mouth disease, or any disease or condition associated with foot and mouth disease virus. In some other embodiments, the disease or condition is a bovine respiratory disease, or any disease or condition associated with bovine viral diarrhea virus, infectious bovine rhinotracheitis virus, parainfluenza- 3 and/or bovine respiratory syncytial virus. The method of treatment can further include inoculation of the ruminant mammal with a vaccine that is suitable for the treatment of the virus that causes, or is associated, with the disease or condition. As provided herein, administration of a conjugated boIFNz.3 protein of the present disclosure, or a composition containing a conjugated boIFNZ.3 protein of the present disclosure, can reduce the incidence and/or severity of, and/or prevent the disease or condition. In some aspects, administration of a conjugated boIFNz.3 protein of the present disclosure, or a composition containing a conjugated boIFNz.3 protein of the present disclosure, can reduce the incidence and/or severity of, and/or prevent the disease or condition. In some aspects, administration of a conjugated boIFNz.3 protein of the present disclosure, or a composition containing a conjugated boIFNz.3 protein of the present disclosure, with further administration of a vaccine, can reduce the incidence and/or severity of, and/or prevent the disease or condition. The present disclosure also provides engineered boIFNz.3 proteins and their amino acid sequences, including engineered boIFNz.3 proteins suitable for conjugation to moieties that are capable of extending boIFNz.3 protein elimination half-life. Also provided are engineered boIFNZ.3 proteins and their amino acid sequences, wherein the engineered boIFNz.3 proteins contain one or more non-natural amino acids suitable for conjugation to moieties that are capable of extending boIFNz.3 protein elimination half-life. In some embodiments, the moieties that are capable of extending boIFNz.3 protein elimination half-life are water-soluble polymers. The
engineered boIFNz.3 proteins of the present disclosure can be obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA synthetase, which allow for site-specific incorporation of the nonnatural amino acid into boIFNz.3 protein. Thus, the one or more non-natural amino acids present in engineered boIFNz.3 proteins or conjugates containing them can be one or more non-naturally encoded amino acids. Novel nucleic acid sequences encoding the engineered boIFNz.3 proteins are also provided.
[0043] Definitions
[0044] As used herein and in the appended claims, the singular forms “a,” “an,” and “the” include plural reference unless the context clearly indicates otherwise.
[0045] As used herein and in the appended claims, the term “comprising” is open ended, and the broadest reasonable interpretation of the term applies. The present disclosure contemplates alternative embodiments wherein the term “consisting of’ can be used in place of any recitation of the term “comprising” herein.
[0046] Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood to a person of ordinary skill in the art to which this invention belongs. Although any methods, devices and materials similar or equivalent to those described herein can be used in the practice or testing of the invention, the preferred methods, devices and materials are now described.
[0047] The term “about” will be understood by persons of ordinary skill in the art and will vary to some extent depending on the context in which it is used.
[0048] The term “adaptive immunity” as used herein refers to a type of immunity that develops when a subject's immune system responds to a foreign substance or microorganism, such as after an infection or vaccination. The function of an adaptive immune response is to destroy invading pathogens and any toxic molecules they produce.
[0049] The term “biotherapeutic” as used herein refers to a therapeutic agent produced using biological means. Typically, the biological means include recombinant DNA technology. Biotherapeutics of the present disclosure include biologically active proteins, such as engineered boIFNz.3 proteins, and conjugated boIFNz.3 proteins and variants thereof. Biotherapeutics also include vaccines.
[0050] The term “bovine respiratory disease” or “BRD” as used herein refers to respiratory disease in cattle affecting the lower or upper respiratory tract, particularly during periods of stress, and is typically considered a disease complex associated with both viral and bacterial pathogens that interact with one another to produce acute clinical disease. For example, the disease can be
associated with multiple bacteria, including Mannheimia haemolytica, Pasteurella multocida, Histophilus somni and/or Mycoplasma bovis, and with viruses, including bovine herpesvirus, bovine respiratory syncytial virus (BRSV), bovine viral diarrhea virus (BVDB), infectious bovine rhinotracheitis virus (IBR), parainfluenza-3 (PI-3) virus and/or bovine respiratory coronavirus.
[0051] The term “canonical amino acid” as used herein refers to any one of the 20 common amino acids that are genetically encoded in humans (i.e., alanine (“A” or “Ala”), arginine (“R” or “Arg”), asparagine (“N” or “Asn”), aspartic acid (“D” or Asp”), cysteine (“C” or “Cys”), glutamic acid (“E” or “Glu”), glutamine (“Q” or “Gin”), glycine (“G” or “Gly”), histidine (“H” or “His”), isoleucine (“I” or “He”), leucine (“L” or “Leu”), lysine (“K” or “Lys”), methionine (“M” or “Met”), phenylalanine (“F” or “Phe”), proline (“P” or “Pro”), serine (“S” or “Ser”), threonine (“T” or “Thr”), tryptophan (“W” or “Trp”), tyrosine (“Y” or “Tyr”) and valine (“V” or “Vai”)). “Canonical amino acids” refers to the collective 20 common amino acids recited above.
[0052] The term “cloven-hoofed animals” as used herein refers to members of the mammalian order Artiodactyla. Non-limiting examples of cloven-hoofed animals include cattle, deer, pigs, antelopes, gazelles, goats and sheep.
[0053] The term “conjugated protein” as used herein refers to a protein to which another chemical group or molecule has been attached. Typically, the chemical group or molecule is a moiety that is capable of extending protein elimination half-life, such as a half-life extender moiety. Thus, the elimination half-life of the conjugated protein can be greater than the elimination half-life of the corresponding or similar protein in its unconjugated form.
[0054] The term “engineered protein” as used herein refers to a protein created and/or generated by design. The engineered protein can be a protein “variant” as disclosed herein. The engineered protein can be a deletion mutant. The engineered protein can be a protein or variant containing a non-natural amino acid, or more particularly, a non-naturally encoded amino acid, as disclosed herein. Methods of engineering a protein to contain a non-natural amino acid, such as a non- naturally encoded amino acid, are disclosed herein. An engineered protein of the disclosure can be a product of such a method.
[0055] The term “foot and mouth disease” or “FMD” as used herein encompasses disease symptoms in swine, cattle, sheep, and goats caused by a foot and mouth disease virus (FMDV) infection. Examples of such symptoms include, but are not limited to, fever, lameness and vesicular lesions on the feet, tongue, snout and teats.
[0056] The term “foot and mouth disease virus” or “FMDV” as used herein refers to a highly contagious pathogenic member of the Picornaviridae family that causes vesicular lesions in cattle, swine and other cloven-hoofed animals.
[0057] The term “half-life” as used herein refers to the time required for any specified property to decrease by half. Typically, the specified property is the concentration of a substance in the body, or a compartment thereof, wherein the substance is a conjugated protein of the present disclosure, or the corresponding protein or a comparable protein in its unconjugated form. The term “halflife” can be referred to herein as “ti/2” or “T1/2”. In some embodiments, a half-life refers to a “terminal half-life” as further disclosed herein.
[0058] The terms “half-life extender,” “half-life extender molecule” and “half-life extender moiety” as used herein refer to molecules (or moieties, e.g., monovalent or bivalent moieties) that can be used to increase the size of a biotherapeutic. Thus, the half-life extender can be used to increase the hydrodynamic volume of a biotherapeutic. A half-life extender can thus be used to increase the half-life of the biotherapeutic to which it is attached. A half-life extender can be a water-soluble polymer. Strategies for extending the half-life of a biotherapeutic include conjugation with a water-soluble polymer (e.g., PEGylation), introduction of glycosylation site(s), and fusion to plasma protein(s) (e.g., serum albumin; see, e.g., Kontermann R.E. (2016) Expert Opin. Biol. Therap. 16(7):903-915, the entire contents of which are hereby incorporated by reference herein in their entirety). Non-limiting examples of half-life extenders include PEG, hydroxyethyl starch (HES), serum albumin, and derivatives thereof, as disclosed herein. Thus, a half-life extender molecule or moiety of the present disclosure is a molecule or moiety that is capable of extending the half-life of a protein of the present disclosure, such as extending the halflife of a boIFNZ.3 protein or variant thereof.
[0059] The terms “identical” or “identity” or “shared identity” in the context of two or more nucleic acids or polypeptide sequences, as used herein, refer to two or more sequences or subsequences that are the same. Identity can be expressed as a percentage. For example, two sequences that are exactly the same share 100% identity. Sequences are "substantially identical" if they have a percentage of nucleotides, or amino acid residues, that are the same (i.e., about 60% identity, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, or about 95% identity) over a specified region, when the nucleotides, or amino acid residues, are compared and aligned for maximum correspondence over a comparison window, or designated region as measured, e.g., using sequence comparison algorithms or other algorithms available to persons of ordinary skill in the art. For example, sequence alignment programs such as BLAST can be used to align two or more sequences, or sequences can be aligned by manual alignment and visual inspection. This definition also refers to the complement of a test sequence. The identity can exist over a region that is at least about 50 amino acids or nucleotides in length, or over a region that is
75-100 amino acids or nucleotides in length, or, where not specified, across the entire sequence of a polynucleotide or polypeptide, e.g., across the full sequence that is specified.
[0060] The term "isolated," when applied to a nucleic acid or protein, denotes that the nucleic acid or protein is free of at least some of the cellular components with which it is associated in the natural state, or that the nucleic acid or protein has been concentrated to a level greater than the concentration of its in vivo or in vitro production. It can be in a homogeneous state. Isolated substances can be in either a dry or semi-dry state, or in solution, including but not limited to, an aqueous solution. It can be a component of a pharmaceutical composition that comprises additional pharmaceutically acceptable carriers and/or excipients. Purity and homogeneity are typically determined using analytical chemistry techniques such as polyacrylamide gel electrophoresis or high-performance liquid chromatography. A protein which is the predominant species present in a preparation is substantially purified. In particular, an isolated gene is separated from open reading frames which flank the gene and encode a protein other than the gene of interest. The term "purified" denotes that a nucleic acid or protein gives rise to substantially one band in an electrophoretic gel. Particularly, it may mean that the nucleic acid or protein is at least 75% pure, at least 80% pure, at least 85% pure, at least 90% pure, at least 95% pure, at least 99% or greater pure.
[0061] The term “moiety that is capable of extending protein elimination half-life” as used herein refers to a moiety that, when conjugated to a protein, can increase the protein elimination half-life. In some embodiments, the elimination half-life is a blood, plasma, serum, or whole body elimination half-life. A moiety that is capable of extending protein elimination half-life includes a half-life extender moiety, which include a water-soluble polymer such as PEG.
[0062] The term “HESylation” as used herein refers to coupling a biotherapeutic with hydroxy alkyl starch, or more particularly, hydroxy ethyl starch (HES), or a derivative thereof.
[0063] The term “elimination half-life” as used herein refers to a pharmacokinetic parameter that is defined as the period of time that it takes for the concentration of a biotherapeutic in the plasma or serum of a subject, or the total amount in the whole body of a subject, to be reduced by about 50%. Thus, after one half-life, the concentration of the biotherapeutic in the plasma or serum, or in the whole body, of the subject will be half of the starting concentration. Typically, the period of time that it takes for the concentration of the biotherapeutic to be reduced by about 50% (e.g., in the blood, plasma or serum, or in the whole body) commences at or about the time of administration of the biotherapeutic to the subject.
[0064] The term "immune response" as used herein refers to the production of molecules such as cytokines (e.g., interferons, chemokines, etc., and/or antibodies and/or cells (such as T
lymphocytes)) that are directed against, or assist in the decomposition or inhibition of, a particular infectious agent, such as a virus or an antigenic epitope or particular antigenic epitopes.
[0065] The terms "effective immunoprotective response," "immunoprotection" and like terms, as used herein, refer to an immune response that is directed against an infectious agent such as a virus as a whole and/or one or more antigenic epitopes of a pathogen so as to protect against infection by the pathogen in a treated animal. An effective immunoprotective response can be induced in animals that have not previously been infected with the pathogen and/or are not infected with the pathogen, e.g., at the time of treatment with a biotherapeutic of the present disclosure and/or a vaccine. An effective immunoprotective response can also be induced in an animal already infected with the pathogen, e.g., at the time of treatment with a biotherapeutic of the present disclosure and/or a vaccine.
[0066] The terms “modulated serum half-life” and “modulated plasma half-life” refer to positive or negative changes in the circulating half-life of a conjugated protein molecule in serum or plasma, respectively, relative to its unconjugated form.
[0067] The term “modulated therapeutic half-life” as used herein refers to a positive or negative change in the half-life of the therapeutically effective amount of a conjugated protein relative to its unconjugated form.
[0068] The term “mutein” as used herein refers to a protein with an altered amino acid sequence. [0069] The term “non-natural amino acid” as used herein refers to non-proteinogenic amino acids, meaning that they are not among the 20 amino acids attached to tRNAs in living human cells used to polymerize proteins. Some non-natural amino acids do occur in nature, e.g., in some non-human species, but most are chemically synthesized. By way of example only, citrulline is a non- proteinogenic amino acid produced in vivo through the oxidation of arginine, while p-benzoyl- phenylalanine is an example of a non-natural amino acid that is not found in nature at all. Nonlimiting examples of non-natural amino acids are disclosed herein. Some non-natural amino acids are derived from the 20 canonical amino acids, e.g., via chemical modifications. Non-natural amino acids of the present disclosure include non-naturally encoded amino acids. Non-natural amino acid-containing polypeptides presented herein may include isotopically-labelled compounds with one or more atoms replaced by an atom having an atomic mass or mass number different from the atomic mass or mass number usually found in nature. Examples of isotopes that can be incorporated into the present compounds include isotopes of hydrogen, carbon, nitrogen, oxygen, sulfur, fluorine and chlorine, such as 2H, 3H, 13C, 14C, 15N, 17O, 18O, 35S, 18F, 36C1, respectively. Certain isotopically-labelled compounds described herein, for example those into which radioactive isotopes such as 3H and 14C are incorporated, may be useful in drug and/or
substrate tissue distribution assays. Further, substitution with isotopes such as deuterium, i.e., 2H, can afford certain therapeutic advantages resulting from greater metabolic stability, for example, increased in vivo half-life or reduced dosage requirements.
[0070] The term “non-naturally encoded amino acid” as used herein refers to a non-natural amino acid that has been incorporated into an amino acid sequence via a change in the genetic code of a system (e.g., an organism) that encodes the amino acid sequence. Methods of incorporating a nonnatural amino acid into an amino acid sequence via a change in the genetic code are disclosed herein.
[0071] The term "nucleic acid" as used herein refers to deoxyribonucleotides, deoxyribonucleosides, ribonucleosides, or ribonucleotides and polymers thereof in either single- or double-stranded form. Unless specifically limited, the term encompasses nucleic acids containing known analogues of natural nucleotides which have similar binding properties as the reference nucleic acid and are metabolized in a manner similar to naturally occurring nucleotides. Unless specifically limited otherwise, the term also refers to oligonucleotide analogs including peptidonucleic acid (PNA), analogs of DNA used in antisense technology (phosphorothioates, phosphoroamidates, and the like). Unless otherwise indicated, a nucleic acid having a particular nucleotide sequence also implicitly encompasses conservatively modified variants thereof (including but not limited to, degenerate codon substitutions) and complementary sequences as well as the sequence explicitly indicated. Specifically, degenerate codon substitutions may be achieved by generating sequences in which the third position of one or more selected (or all) codons is substituted with mixed-base and/or deoxyinosine residues (Batzer et al., Nucleic Acid Res. 19:5081 (1991); Ohtsuka et al., J. Biol. Chem. 260:2605-2608 (1985); Rossolini et al., Mol. Cell. Probes 8:91-98 (1994)). A nucleic acid encoding a polypeptide of the present invention may be obtained by a process comprising the steps of screening a library under stringent hybridization conditions with a labeled probe having a nucleotide sequence of the invention, or a fragment thereof, and isolating full-length cDNA and genomic clones containing said nucleotide sequence. Such hybridization techniques are well known to the skilled artisan.
[0072] The term “PEGylation” as used herein refers to coupling a biotherapeutic with polyethylene glycol (PEG), or a derivative thereof. The term “PEGylated” as used herein refers to a PEG-biotherapeutic conjugate, wherein the biotherapeutic is covalently linked to at least one PEG moiety.
[0073] The terms “polyalkylene glycol,” “poly(alkene glycol),” “polyoxyalkylene glycol” and “polyglycol” are used interchangeably herein, and as used herein refers to linear, branched and multiarm polymeric polyether polyols. Non-limiting examples of polyalkylene glycols of the
present disclosure include polyethylene glycol (PEG), polypropylene glycol, polybutylene glycol, and derivatives thereof.
[0074] The terms “polypeptide” and “protein” as used herein have their ordinary meaning in the art and are used interchangeably herein unless expressly indicated otherwise.
[0075] The term “prevent” has its ordinary meaning in the art, that is, to “prevent” means to keep something (e.g., a disease or condition, a viral infection, or both) from happening or arising.
[0076] The term “prophylaxis” or “prophylactic treatment” as used herein includes preventing a disease or condition in a mammal from occurring and/or preventing a disease or condition from getting worse. In some embodiments, prophylaxis refers to preventing a disease or condition from occurring, e.g., by administering a biotherapeutic of the present disclosure to a mammal prior to the onset of the disease or condition, and/or prior to the onset of symptoms of the disease or condition. In some embodiments, prophylaxis refers to protection against infection by a pathogen in a mammal.
[0077] The term “protection against infection” as used herein includes not only the absolute prevention of infection by a pathogen, but also any detectable reduction in a degree or rate of infection by a pathogen, or any detectable reduction in severity of a disease or any symptom or condition resulting from infection by the pathogen in an animal, such as a vaccinated animal, as compared to an infected animal, such as an unvaccinated infected animal, e.g., using methods known to a person of ordinary skill in the art and/or via assessment by a veterinarian or physician. Where the term “method of treating a disease or condition” is used herein, such a method can include providing protection against the disease or condition.
[0078] The term “refolding" as used herein refers to any process, reaction or method which transforms disulfide bond containing polypeptides from an improperly folded or unfolded state to a native or properly folded conformation with respect to disulfide bonds.
[0079] The term “ruminant mammal” as used herein refers to members of the mammalian order Artiodactyla. Non-limiting examples of ruminant mammals are cattle, all domesticated and wild bovines, deer, giraffes, antelopes, gazelles, goats and sheep. In some embodiments, the ruminant mammal is bovine. “Bovine” as used herein refers to a diverse group of hoofed animals (ungulates), including cattle, bison, African buffalo, water buffalo and some antelope. As used herein, “bovine” can refer to an individual animal (singular form), or to a group of animals (plural form). In some embodiments, the ruminant mammal is a cattle. As used herein, “cattle” refers to an individual animal (singlular form) or a group of animals (plural form). In some embodiments, a cattle is a member of the genus Bos, which includes domestic cattle (Bos Taurus). The cattle may be, e.g., beef cattle (raised for meat production) or dairy cattle (used for milk production). In
some embodiments, the ruminant mammal is a calf. In some embodiments, a calf is less than 12 months old. In some embodiments, a calf is less than 6 months old. In some embodiments, a calf is between 4 and 6 months old.
[0080] The term "subject" as used herein, refers to an animal who is the object of treatment, observation or experiment. In some embodiments, the animal is a ruminant mammal, e.g., as defined herein.
[0081] The term “terminal half-life” as used herein refers to the time required to divide the concentration of an agent (e.g., a conjugated protein of the present disclosure) in a body or body compartment (e.g., plasma or serum) by two after reaching pseudo-equilibrium, and not the time required to eliminate half the administered dose (see, e.g., Toutain P.L. and Bousquet-Melou A., J. Vetinary Pharmacology and Therapeutics (2004) 27(6):427-439). As understood by a person of ordinary skill in the art, when the absorptiom process is not a limiting factor, half-life is controlled by clearance (e.g., plasma clearance) and the extent of distribution. However, when the absorption process is a limiting factor, the terminal half-life does not reflect the elimination process; rather, the terminal half-life reflects the rate and extent of absorption. Terminal half-life is particularly relevant in the case of multiple dosing regimens.
[0082] The term “therapeutically effective amount” as used herein refers to an amount sufficient to treat, prevent or at least partially arrest symptoms, or at least partially protect against the development of symptoms, of a disease, disorder or condition, or an amount sufficient to induce a desired response in a subject, including but not limited to inducing an immune response and/or protection from an infectious disease. A therapeutically effective amount will depend on the severity and course of the disease, disorder or condition, previous therapy, the subject's health status and response to the drugs, and the judgment of the treating physician or veterinarian. It is considered well within the skill of a person of ordinary skill in the art to determine such therapeutically effective amounts by routine experimentation e.g., a dose escalation trial).
[0083] The term “treating” as used herein refers to prophylaxic treatment and can also include or refer to therapeutic treatment, unless expressly indicated otherwise.
[0084] The term “unusual genetically encoded amino acid that is not present in humans” as used herein refers to amino acids that are used in the biosynthesis of proteins in non-humans. Nonlimiting examples include selenocysteine and pyrrolysine, the latter of which is produced in some methanogenic archaea and bacteria.
[0085] The term "variant" as used herein refers to a protein derived from the corresponding native (also referred to herein as “wild type”) protein by deletion (also referred to herein as “truncation”) or addition of one or more amino acids to the N-terminus and/or C-terminus of the native protein;
deletion or addition of one or more amino acids at one or more sites in the native protein; and/or substitution of one or more amino acids at one or more sites in the native protein. Such variants may result from, for example, protein engineering. Thus, a protein variant of the present disclosure can be an engineered protein. Protein variants encompassed by the present disclosure are biologically active, that is, they possess the desired IFNz.3 biological activity, as described herein. Non-limiting examples of the desired IFNZ.3 biological activity of an IFNZ.3 protein variant include the ability of the IFNZ.3 protein variant to induce mRNA expression of ISG15, and/or the ability of the IFNZ.3 protein variant to inhibit viral replication. In some embodiments, the desired IFNZ.3 biological activity is the ability to induce mRNA expression of ISG15. Methods of determining induction of mRNA expression of ISG15 are disclosed herein. For example, in some embodiments, a IFNZ.3 protein variant is considered to have the desired IFNZ.3 activity if it induces mRNA expression of ISG15 at a level that is at least about 50%, about 60%, about 70%, about 80%, about 90% or about 95% of the level of mRNA expression of ISG15 that is induced by the IFNZ.3 molecule of SEQ ID NO: 1, e.g., if it induces mRNA expression of ISG15 in MDBK cells at a level that is at least about 50%, about 60%, about 70%, about 80%, about 90% or about 95% of the level of mRNA expression of ISG15 that is induced by the IFNz.3 molecule of SEQ ID NO: 1 after treatment of the MDBK cells with the IFNZ.3 protein variant at a concentration of 100 ng/ml, and wherein mRNA expression is determined 6 hours after the treatment. In some embodiments, the desired IFNZ.3 biological activity is the ability to inhibit viral replication. In some embodiments, the desired IFNZ.3 biological activity is the ability to inhibit the replication of bovine viral diarrhea virus (BVDV). Methods of determining the ability to inhibit the replication of BVDV are disclosed herein. For example, in some embodiments, a IFNZ.3 protein variant is considered to have the desired IFNz.3 activity if it inhibits the replication of BVDV by at least about 50%, about 60%, about 70%, about 80%, about 90% or about 95%, e.g., if it inhibits the replication of BVDV, such as a cytopathic genotype la BVDV strain, in MDBK cells by at least about 50%, about 60%, about 70%, about 80%, about 90% or about 95% after treatment of the MDBK cells with the IFNZ.3 protein variant at a concentration of > 250 ng/ml, and wherein the inhibition of BVDV replication is determined 24 hours after the treatment. In some other embodiments, the desired IFNZ.3 biological activity is the ability to inhibit the replication of foot and mouth disease virus (FMDV). Methods of determining the ability to inhibit the replication of FMDV are disclosed herein. For example, in some embodiments, a IFNZ.3 protein variant is considered to have the desired IFNz.3 activity if it inhibits the replication of FMDV by at least about 50%, about 60%, about 70%, about 80%, about 90% or about 95%, e.g., if it inhibits the replication of FMDV, such as South African Territories (SAT)l FMDV, in MDBK cells by at least
about 50%, about 60%, about 70%, about 80%, about 90% or about 95% after treatment of the MDBK cells with > 10 ng/ml concentrations of the IFNX3 protein variant, and wherein the inhibition of FMDV replication is determined approximately 48 hours after the treatment. The biologically active variant may differ from the corresponding wild type protein by as few as 1 to 15 amino acid residues, as few as 1 to 10 amino acid residues, as few as 1 to 5 amino acid residues, as few as 1 to 3 amino acid residues, or by 1 amino acid residue.
[0086] The term “viremia” as used herein refers to the presence of virus(es) in the bloodstream and/or the entry of virus(es) into the bloodstream. Methods of measuring viremia in a subject are known to a person of ordinary skill in the art. Such methods include drawing blood sample(s) from the subject and testing the sample (e.g., for viral RNA) using a polymerase chain reaction (PCR), such as quantitative reverse transcription PCR (RT-PCR) (e.g., see Zhang Z. and Alexandersen S. (2003). J Virol Methods, 111 :95-100; and Stenfeldt C. and Belsham G.J. (2012) Veterinary Microbiology, 154:230-239). Viremia can include primary viremia, which refers to the initial spread of virus in the blood from a primary site of infection, and/or secondary viremia, which occurs when primary viremia has resulted in infection of additional tissues via the bloodstream and then reenters the bloodstream.
[0087] The term “water-soluble polymer” as used herein refers to any polymer that is soluble in aqueous solvents. A water-soluble polymer derivative of the present disclosure includes a water- soluble polymer comprising a reactive group that is capable of reacting with a non-natural amino acid. Water-soluble polymers may or may not have their own biological activity. In a non-limiting example, a water-soluble polymer of the present disclosure can be a polyalkylene glycol.
[0088] All isomers including but not limited to diastereomers, enantiomers, and mixtures thereof are considered as part of the compositions described herein. In additional or further embodiments, the polypeptides containing non-naturally encoded amino acid can be metabolized upon administration to an organism in need to produce a metabolite for use to provide a desired effect, for example, a desired therapeutic effect. In further or additional embodiments are active metabolites of polypeptides containing non-naturally encoded amino acids.
[0089] In some situations, non-naturally encoded amino acid polypeptides may exist as tautomers. In addition, the non-naturally encoded amino acid polypeptides described herein can exist in unsolvated as well as solvated forms with pharmaceutically acceptable solvents such as water, ethanol, and the like. The solvated forms are also considered to be disclosed herein. Those of ordinary skill in the art will recognize that some of the compounds herein can exist in several tautomeric forms. All such tautomeric forms are considered as part of the compositions described herein.
[0090] Unless otherwise indicated, conventional methods of mass spectroscopy (MS), nuclear magnetic resonance (NMR), high performance liquid chromatography (HPLC), protein chemistry, biochemistry, recombinant DNA techniques and pharmacology, within the skill of the art are employed.
[0091] In some embodiments, reference to the term “having an amino acid sequence” or “has an amino acid sequence” may encompass “comprises an amino acid sequence” or “comprising an amino acid sequence”. For example, a protein having an amino acid sequence of SEQ ID NO: 2 or that has an amino acid sequence of SEQ ID NO: 2 may comprise the amino acid sequence of SEQ ID NO: 2. Accordingly, reference to the term “having an amino acid sequence” or “has an amino acid sequence” may encompass “consisting of an amino acid sequence”. For example, a protein having an amino acid sequence of SEQ ID NO: 2 may consist of the amino acid sequence of SEQ ID NO: 2.
[0092] Introduction
The present disclosure provides boIFNz.3 proteins and variants thereof, including boIFN/3 variants comprising one or more non-naturally encoded amino acids. The boIFNZ.3 variants of the present disclosure are engineered proteins that can be produced in cells equipped with a translation system comprising an orthogonal tRNA and an orthogonal aminoacyl tRNA synthetase, thereby allowing for the site-specific incorporation of non-natural amino acids into boIFNZ.3 protein, as disclosed herein.
[0093] The present disclosure further provides conjugated boIFNz.3 proteins. Conjugated boIFNZ.3 proteins of the present disclosure comprise engineered boIFNz.3 proteins containing one or more non-natural amino acid, wherein the engineered boIFNz.3 protein is conjugated to a moiety that is capable of extending boIFNz.3 protein elimination half-life. A half-life extender moiety, such as a water-soluble polymer, can be conjugated to an engineered boIFNz.3 protein via a stable covalent linkage to the non-natural amino acid. The stable covalent linkage can reduce or eliminate the potential for cleavage of the half-life extender moiety from the protein in vivo. Accordingly, the stable linkage provides the conjugated protein with an increased and consistent elimination half-life, improved pharmacodynamics and/or an improved therapeutic profile compared to other IFNs, or to other conjugated IFNs that do not contain a stable covalent linkage joining the half-life extender moiety to the IFN via a site-specifically incorporated non-natural amino acid.
[0094] The present disclosure also provides pharmaceutical compositions containing engineered boIFNZ.3 proteins, or conjugated boIFNz.3 proteins or variants thereof. Also provided are methods of treating (e.g., preventing) diseases or conditions, such as BRD and FMD, in ruminant mammals
including cattle, via the administration of a conjugated boIFNz.3 protein of the present disclosure. The treatment can include combination therapy by further administration of a vaccine.
[0095] As disclosed herein, a conjugated boIFNz.3 protein of the present disclosure has an elimination half-life that exceeds that of typical IFNs and can provide early and sustained IFN activity in vivo. Consequently, the presently disclosed conjugated boIFN/3 proteins can provide early and sustained anti-viral activity in treated animals, thereby protecting the treated animals from infection by viruses associated with BRD or FMD. While vaccination is a useful tool for preventing diseases such as FMD, development of protective adaptive immune response takes about 5 to 7 days post-vaccination (Diaz-San Segundo F. et al. (2021) Frontiers in Microbiology V12; Article 668890; Diaz-San Segundo F. et al. (2016) Virology V499:340-349). The protection from viral infection provided by administration of a conjugated boIFNZ.3 of the present disclosure, alone or in combination with a FMD vaccine, can protect treated animals for at least 5 days postadministration. Thus, treatment of an animal with a combination of a conjugated boIFNZ.3 protein of the present disclosure and a vaccine can provide the treated animal with protection against viral infection prior to the development of adaptive immune response, thereby filling the gap between time of vaccination and development of antibody-mediated anti-viral activity in vivo. Additionally, as disclosed herein, treatment with a conjugated boIFNz.3 protein of the present disclosure has the potential to induce long-term protection against viral infection.
[0096] Bovine IFNX3 protein deletion variants
[0097] The present disclosure provides engineered boIFNz.3 proteins, including boIFNz.3 protein deletion variants. BoIFNz.3 proteins and variants of the present disclosure are characterized by features including their amino acid sequence and/or elimination half-life in vivo. The present disclosure includes engineered boIFNz.3 proteins and variants produced using the compositions and methods disclosed herein. An excipient (including but not limited to, a pharmaceutically acceptable excipient) can also be present with the protein or variant.
[0098] Table 1 discloses boIFNz.3 protein amino acid sequences, including wild type protein and variant sequences, wherein the variant sequences are N-terminal truncations of the wild type sequence. The amino acid sequences can contain a non-natural amino acid. Table 1 further discloses some nucleic acid sequences encoding amino acids sequences of the present disclosure. [0099] Table 1 Bovine IFNZ.3 protein amino acid sequences that can include one or more non- naturally encoded amino acids (nnAA), and bovine IFNz.3 nucleic acid sequences. Also disclosed are: all of the amino acid sequences in Table 1, wherein: (i) any amino acid is replaced with any nnAA; (ii) any amino acid is replaced with pyrrolysine; (iii) X is replaced with any nnAA; (iv) X is pAF; (v) X is pyrrolysine; and all the foregoing, wherein the amino acid sequence further
contains an N-terminal methionine residue, unless such a residue is expressly shown. Where an amino acid and its corresponding position is listed in the Description column, the amino acid position refers to the position in the corresponding wild type sequence (SEQ ID NO: 1) with no
“X” denotes a nnAA; underscore indicates the amber stop codon
[00100] In some embodiments, a boIFNZ.3 protein variant of the present disclosure is an N- terminal deletion variant. In some embodiments, a boIFNZ.3 protein variant of the present disclosure can have an amino acid sequence disclosed in Table 1, wherein the amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 1, wherein each sequence optionally further comprises an N-terminal methionine, and wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some other embodiments, the boIFNz.3 protein variant amino acid sequence shares at most about 99%, or at most about 98% identity with SEQ ID NO: 1, wherein each sequence optionally further comprises an N-terminal methionine. In some embodiments, the boIFNZ.3 protein variant amino acid sequence shares at most about 98% identity with SEQ ID NO: 1. In some embodiments, the boIFNZ.3 protein variant amino acid sequence shares at most about 99% identity with SEQ ID NO: 1.
[00101] In some embodiments, a boIFNz.3 protein variant of the present disclosure is an N- terminal deletion variant having an amino acid sequence disclosed in Table 1, wherein the boIFNz.3 protein variant amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 1, and wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNZ.3 protein variant amino acid sequence shares at most about 99%, or at most about 98% identity with SEQ ID NO: 1. In some embodiments, the boIFNZ.3 protein variant amino acid sequence shares at most about 98% identity with SEQ ID NO: 1. In some other embodiments, the boIFNZ.3 protein variant amino acid sequence shares at most about 99% identity with SEQ ID NO: 1.
[00102] In some other embodiments, a boIFNz.3 protein variant of the present disclosure is an N- terminal deletion variant having an amino acid sequence disclosed in Table 1, wherein each sequence further comprises an N-terminal methionine, and wherein the boIFNZ.3 protein variant amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%
identity with SEQ ID NO: 1, and shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNZ.3 protein variant amino acid sequence of Table 1 that further comprises an N-terminal methionine shares at most about 99%, or at most about 98% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant amino acid sequence of Table 1 that further comprises an N-terminal methionine shares at most about 98% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant amino acid sequence of Table 1 that further comprises an N-terminal methionine shares at most about 99% identity with SEQ ID NO: 1.
[00103] In some other embodiments, a boIFNz.3 protein variant of the present disclosure can have an amino acid sequence disclosed in Table 1, wherein the amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with (i) SEQ ID NO: 2, 3, 4, 5, 6, 7,
8, 9, 10, 11, 12 or 13; or (ii) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00104] In some embodiments, a boIFNX3 protein variant of the present disclosure can have an amino acid sequence disclosed in Table 1, wherein the amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8,
9, 10, 11, 12 or 13; and wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00105] In some other embodiments, a boIFNz.3 protein variant of the present disclosure can have an amino acid sequence disclosed in Table 1, wherein the amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; and wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00106] In some other embodiments, a boIFNz.3 protein variant of the present disclosure can have an amino acid sequence that shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1; optionally wherein the boIFNz.3 protein variant further comprises an N-terminal methionine. In some other embodiments, a boIFNz.3 protein variant of the present disclosure can have an amino acid
sequence, wherein the boIFNz.3 protein variant amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some other embodiments, a boIFNz.3 protein variant of the present disclosure can have an amino acid sequence, wherein the boIFNz.3 protein variant amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing amino acid sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00107] In some embodiments, a boIFNX3 protein variant has an amino acid sequence that shares at least 75% identity with (i) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; or (ii) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00108] In some embodiments, a boIFNX3 protein variant has an amino acid sequence that shares at least 75% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00109] In some embodiments, a boIFNX3 protein variant has an amino acid sequence that shares at least 75% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00110] In some embodiments, the boIFNX3 protein variant has an amino acid sequence that shares at least 75% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00111] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 75% identity with SEQ ID NO: 5; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00112] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 75% identity with SEQ ID NO: 5, wherein the boIFNZ.3 protein variant further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00113] In some embodiments, a boIFNX3 protein variant has an amino acid sequence that shares at least 85% identity with (i) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; or (ii) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00114] In some embodiments, a boIFNX3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00115] In some embodiments, a boIFNX3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00116] In some embodiments, the boIFNX3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 5; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00117] In some other embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 5, wherein the sequence further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00118] In some embodiments, a boIFNX3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, a boIFNZ.3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, the boIFNz.3 protein variant has an
amino acid sequence that shares at least 85% identity with SEQ ID NO: 5; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 85% identity with SEQ ID NO: 5, wherein the boIFNX3 protein variant further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00119] In some embodiments, a boIFNX3 protein variant has an amino acid sequence that shares at least 90% identity with (i) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; or (ii) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00120] In some embodiments, a boIFNX3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00121] In some other embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00122] In some embodiments, the boIFNX3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 5; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00123] In some other embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 5, wherein the sequence further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00124] In some embodiments, a boIFNX3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing
sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 5, wherein the boIFNZ.3 protein variant further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00125] In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with (i) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; or (ii) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00126] In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00127] In some other embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00128] In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 5; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00129] In some other embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 5, wherein the sequence further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00130] In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally
wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 5, wherein the boIFNZ.3 protein variant further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00131] In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with (i) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; or (ii) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00132] In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00133] In some other embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00134] In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 5; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00135] In some embodiments, the boIFNX3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 5, wherein the sequence optionally further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00136] In some embodiments, a boIFNX3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNX3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNX3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 5; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 5, wherein the boIFNZ.3 protein variant further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00137] In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00138] In some other embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00139] In some embodiments, the boIFNX3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 5; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00140] In some embodiments, the boIFNX3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 5, wherein the sequence further comprises an N- terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00141] In some embodiments, a boIFNX3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 96% identity with SEQ ID NO: 5, wherein the boIFNZ.3 protein variant further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00142] In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00143] In some other embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each
of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00144] In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 5; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00145] In some embodiments, the boIFNX3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 5, wherein the sequence further comprises an N- terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00146] In some embodiments, a boIFNX3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 97% identity with SEQ ID NO: 5, wherein the boIFNZ.3 protein variant further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00147] In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00148] In some other embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00149] In some embodiments, the boIFNX3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 5; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00150] In some embodiments, the boIFNX3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 5, wherein the sequence further comprises an N- terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00151] In some embodiments, a boIFNX3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 98% identity with SEQ ID NO: 5, wherein the boIFNZ.3 protein variant further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00152] In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 99% identity with (i) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; or (ii) SEQ ID NO:
2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00153] In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00154] In some embodiments, a boIFNX3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00155] In some embodiments, a boIFNX3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, a boIFNz.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 5; wherein the boIFNz.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 5, wherein the boIFNZ.3 protein variant further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00156] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 2; wherein the boIFNZ.3 protein variant amino acid
sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00157] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 2; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00158] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 2, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00159] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 3; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00160] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 3; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00161] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 3, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00162] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 4; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00163] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 4; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00164] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 4, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00165] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 5; wherein the boIFNZ.3 protein variant amino acid
sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00166] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 5; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00167] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 5, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00168] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 6; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00169] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 6; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00170] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 6, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00171] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 7; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00172] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 7; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00173] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 7, wherein amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00174] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 8; wherein the amino acid sequence shares less than
100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00175] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 8; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00176] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 8, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00177] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 9; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00178] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 9; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00179] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 9, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00180] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 10; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00181] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 10; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00182] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 10, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00183] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 11; wherein the amino acid sequence shares less
than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00184] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 11; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00185] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 11, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00186] In some embodiments, the boIFNZ.3 proteinv variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 12; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00187] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 12; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00188] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 12, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the boIFNZ.3 protein variant amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00189] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1, optionally wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00190] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 13; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00191] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence that shares at least 99% identity with SEQ ID NO: 13, wherein the amino acid sequence further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1.
[00192] In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 2. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ
ID NO: 2, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N- terminal methionine.
[00193] In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 3. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 3, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N- terminal methionine.
[00194] In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 4. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 4, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N- terminal methionine.
[00195] In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 5. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 5, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N- terminal methionine.
[00196] In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 6. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 6, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N- terminal methionine.
[00197] In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 7. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 7, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N- terminal methionine.
[00198] In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 8. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 8, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N- terminal methionine.
[00199] In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 9. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 9, wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N- terminal methionine.
[00200] In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 10. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 10, wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00201] In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 11. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 11, wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00202] In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 12. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 12, wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00203] In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 13. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 13, wherein the boIFNz.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00204] In the section above any definition of a protein or variant can equally be applied to the boIFNZ.3 protein portion of a conjugate, and vice versa.
[00205] A boIFNZ.3 protein or variant of the present disclosure can comprise one or more nonnatural amino acids, as further disclosed herein.
[00206] A boIFNZ.3 protein or variant of the present disclosure can comprise an unusual genetically encoded amino acid that is not present in humans. The unusual genetically encoded amino acid can be selenocysteine or pyrrolysine, the latter of which is known to be used in the biosynthesis of proteins in some methanogenic archaea and bacteria.
[00207] The present disclosure also provides a nucleic acid having a nucleotide sequence disclosed in Table 1, wherein the nucleotide sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 75% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 85% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 90% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 95% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 96% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 97% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 98% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide
sequence that shares at least 99% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares less than 100% identity with SEQ ID NO: 26.
[00208] The present disclosure also provides a nucleic acid having a nucleotide sequence wherein the nucleotide sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 75% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 85% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 90% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 95% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 96% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 97% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 98% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 99% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has a nucleotide sequence that shares less than 100% identity with SEQ ID NO: 26.
[00209] Bovine IFNX3 protein variants containing non-natural amino acids
[00210] The present disclosure provides a boIFN+3 protein variant that is characterized as having an amino acid sequence comprising at least one non-natural amino acid. The boIFN+3 protein variant can be further characterized by its elimination half-life. BoIFN/3 protein variants containing at least one non-natural amino acid can be used for the treatment of a disease or condition in a mammal, and/or can be used for producing protein conjugates of the present disclosure. The invention includes boIFN+3 protein variants produced using the compositions and methods of the invention. An excipient (including but not limited to, a pharmaceutically acceptable excipient) can also be present with the protein or variant thereof.
[00211] The incorporation of non-natural amino acids into proteins permits the introduction of reactive chemical moieties as alternatives to those present in canonical amino acids, such as the epsilon -NEE of lysine, the sulfhydryl -SH of cysteine, the hydroxyl -OH of serine, and the imino group of histidine. Certain chemical functional groups are known to be inert to the functional groups found in the 20 canonical amino acids but react cleanly and efficiently with other moieties to form stable linkages. For example, azide and acetylene groups, which are not present in canonical amino acids, are known in the art to undergo a Huisgen [3+2] cycloaddition reaction in aqueous conditions in the presence of a catalytic amount of copper. See, e.g., Tomoe, et al.,
(2002) J. Org. Chem. 67 :3057 -3064; and Rostovtsev, et al., (2002) Angew. Chem. Int. Ed. 41 :2596-2599. By introducing an azide moiety into a protein structure, one is able to incorporate a functional group that is chemically inert to amines, sulfhydryls, carboxylic acids and hydroxyl groups found in proteins, but that reacts smoothly and efficiently with an acetylene moiety to form a cycloaddition product. Importantly, in the absence of the acetylene moiety, the azide remains chemically inert and unreactive in the presence of other protein side chains and under physiological conditions. Thus, the introduction of a non-natural amino acid into a protein provides it with alternative chemical moieties that can be used to selectively conjugate additional molecules to the protein. The present disclosure provides the introduction of at least one nonnatural amino acid into boIFNZ.3 protein or a variant thereof, allowing for selective conjugation to occur with an additional molecule, such as a molecule that extends its elimination half-life.
[00212] Typically, the non-natural amino acids used in the methods and compositions described herein have at least one of the following properties: (1) at least one functional group on the sidechain of the non-natural amino acid has at least one characteristic and/or activity and/or reactivity orthogonal to the chemical reactivity of the canonical amino acids, or at least orthogonal to the chemical reactivity of the canonical amino acids present in the polypeptide that includes the non-natural amino acid; (2) the introduced non-natural amino acids are substantially chemically inert toward the canonical amino acids; and (3) the non-natural amino acid can be stably incorporated into a polypeptide, preferably with the stability commensurate with the canonical amino acids or under typical physiological conditions, and further preferably such incorporation can occur in a cell-based system, such as cell culture.
[00213] In some embodiments, the one or more non-natural amino acids is 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 non-natural amino acids. In some embodmients, the one or more non-natural amino acids is 1, 2, 3, 4 or 5 non-natural amino acids. In some embodiments, the one or more non-natural amino acids is 1, 2 or 3 non-natural amino acids. In some embodiments, the one or more non-natural amino acids is one (1) non-natural amino acid.
[00214] The one or more non-natural amino acids may be one or more non-naturally encoded amino acids. The one or more non-naturally encoded amino acids may be encoded by a codon that does not code for one of the twenty canonical amino acids. The one or more non-natural amino acids may be encoded by a nonsense codon (stop codon). The stop codon may be an amber codon. The amber codon may comprise a UAG sequence. The stop codon may be an ochre codon. The ochre codon may comprise a UAA sequence. The stop codon may be an opal or umber codon. The opal or umber codon may comprise a UGA sequence. The one or more non-natural amino acids may be encoded by a four-base codon.
[00215] Non-natural amino acids of the present disclosure include, but are not limited to, 1) substituted phenylalanine and tyrosine analogues, such as 4-amino-L-phenylalanine, 4-acetyl-L- phenylalanine, 4-azido-L-phenylalanine, 4-nitro-L-phenylalanine, 3 -m ethoxy -L-phenylalanine, 4- isopropyl-L-phenylalanine, 3-nitro-L-tyrosine, O-methyl-L-tyrosine and O-phosphotyrosine; 2) amino acids that can be photo-cross-linked, e.g., amino acids with aryl azide or benzophenone groups, such as 4-azidophenylalanine or 4-benzoylphenylalanine; 3) amino acids that have unique chemical reactivity, such as 4-acetyl-L-phenylalanine, 3-acetyl-L-phenylalanine, O-allyl-L- tyrosine, O-2-propyn-l-yl-L-tyrosine, N-(ethylthio)thiocarbonyl-L-phenylalanine and p-(3- oxobutanoyl)-L-phenylalanine; 4) heavy-atom-containing amino acids, e.g., for phasing in X-ray crystallography, such as 4-iodo-L-phenylalanine or 4-bromo-L-phenylalanine; 5) a redox-active amino acid, such as 3, 4-dihydroxy -L-phenylalanine; 6) a fluorinated amino acid, such as a 2- fluorophenyl alanine (e.g., 2-fluoro-L-phenylalanine), a 3 -fluorophenylalanine (e.g., 3-fluoro-L- phenylalanine) or a 4-fluorophenylalanine (e.g., 4-fluoro-L-phenylalanine; 7) a fluorescent amino acid, such as an amino acid containing a naphthyl, dansyl or 7-aminocoumarin side chain; 8) a photocleavable or photoisomerizable amino acid, such as an amino acid comprising an azobenzyl or nitrobenzyl, e.g., cysteine, serine or tyrosine comprising azobenzyl or nitrobenzyl; 9) a P-amino acid (e.g., a p2 or p3 amino acid); 10) a homo-amino acid, such as homoglutamine (e.g., betahomoglutamine) or homophenylalanine (e.g., beta-homophenylalanine); 11) a proline or pyruvic acid derivative; 12) a 3-substituted alanine derivative; 14) a glycine derivative; 15) a linear core amino acid; 16) a diamino acid; 17) a D-amino acid; 18) an N-m ethyl amino acid; 19) a phosphotyrosine mimetic, such as a carboxymethylphenylalanine (pCmF) (e.g., 4-carboxymethyl- L-phenylalanine); 20) 2-aminooctanoic acid; and 21) an amino acid comprising a saccharide moiety, such asN-acetyl-L-glucosaminyl-L-serine, beta-N-acetylglucosamine-O-serine, N-acetyl- L-galactosaminyl-L-serine, alpha-N-acetylgalactosamine-O-serine, O-(3-O-D-galactosyl-N- acetyl-beta-D-galactosaminyl)-L-serine, N-acetyl-L-glucosaminyl-L-threonine, alpha-N- acetylgalactosamine-O-threonine, 3-O-(N-acetyl-beta-D-glucosaminyl)-L-threonine, N-acetyl-L- glucosaminyl-L-asparagine, N4-(P-N-Acetyl-D-glucosaminyl)-L-asparagine and O-(mannosyl)- L-serine; an amino acid wherein the naturally-occurring N- or O- linkage between the amino acid and the saccharide is replaced by a covalent linkage not commonly found in nature, including but not limited to, an alkene, an oxime, a thioether, an amide and the like; or an amino acid containing saccharides that are not commonly found in naturally-occurring proteins, such as 2-deoxy -glucose, 2-deoxy -galactose and the like. Specific examples of non-natural amino acids include, but are not limited to, a p-acetylphenylalanine (e.g., 4-acetyl-L-phenylalanine), a 4-boronophenylalanine (pBoF) (e.g., 4-borono-L-phenylalanine, a 4-propargyloxyphenylalanine (pPrF) (e.g., 4-
propargyloxy-L-phenylalanine), an O-methyltyrosine (e.g., O-methyl-L-tyrosine), a 3-(2- naphthyl)alanine (NapA) (e.g., 3-(2-naphthyl)-L-alanine), a 3 -methylphenylalanine (e.g., 3- methyl-L-phenylalanine), an O-allyltyrosine (e.g., O-allyl-L-tyrosine), an O-isopropyltyrosine (e.g., O-isopropyl-L-tyrosine), a dopamine (e.g., L-Dopa), a 4-isopropylphenylalanine (e.g., 4- isopropyl-L-phenylalanine), a 4-azidophenylalanine (pAz) (e.g., 4-azido-L-phenylalanine), a 4- benzoylphenylalanine (pBpF) (e.g., 4-benzoyl-L-phenylalanine), an O-phosphoserine (e.g., O- phospho-L-serine), an O-phosphotyrosine (e.g., O-phospho-L-tyrosine), a 4-iodophenylalanine (pIF) (e.g., 4-iodo-L-phenylalanine, a 4-bromophenylalanine (e.g., 4-bromo-L-phenylalanine), a 4-aminophenylalanine (e.g., 4-amino-L-phenylalanine), a 4-cyanophenylalanine (pCNF) (e.g., 4- cyano-L-phenylalanine, a (8-hydroxyquinolin-3-yl)alanine (HQA) (e.g., (8-hydroxyquinolin-3- yl)-L-alanine), a (2,2-bipyridin-5-yl)alanine (BipyA) (e.g., (2,2-bipyridin-5-yl)-L-alanine), and the like. Additional non-natural amino acids are disclosed in Liu et al. (2010) Annu Rev Biochem, 79:413-44; Wang et al. (2005) Angew Chem Int Ed, 44:34-66; and Published International Application Nos.: WO 2012/166560, WO 2012/166559, WO 2011/028195, WO 2010/037062, WO 2008/083346, WO 2008/077079, WO 2007/094916, WO 2007/079130, WO 2007/070659 and WO 2007/059312, the entire contents of each of which are hereby incorporated by reference herein in their entirety. In some embodiments, the one or more non-natural amino acids can be p- acetylphenylalanine. In some more particular embodiments, the one or more non-natural amino acids can be p-acetyl-L-phenylalanine (pAF).
[00216] In some embodiments, one or more non-natural amino acids is selected from the group consisting of 4-acetylphenylalanine, 3-O-(N-acetyl-beta-D-glucosaminyl)threonine, N4-( -N- Acetyl-D-glucosaminyl)asparagine, O-allyltyrosine, alpha-N-acetylgalactosamine-O-serine, alpha-N-acetylgalactosamine-O-threonine, 2-aminooctanoic acid, 2-aminophenylalanine, 3- aminophenylalanine, 4-aminophenylalanine, 2-aminotyrosine, 3 -aminotyrosine, 4- azidophenylalanine, 4-benzoylphenylalanine, (2,2-bipyridin-5yl)alanine, 3-boronophenylalanine, 4-boronophenylalanine, 4-bromophenylalanine, p-carboxymethylphenylalanine, 4- carboxyphenylalanine, p-cyanophenylalanine, 3,4-dihydroxyphenylalanine, 4- ethynylphenylalanine, 2-fluorophenylalanine, 3 -fluorophenylalanine, 4-fluorophenylalanine, O- (3-O-D-galactosyl-N-acetyl-beta-D-galactosaminyl)serine, homoglutamine, (8-hydroxyquinolin-
3-yl)alanine, 4-iodophenylalanine, 4-isopropylphenylalanine, O-i-propyltyrosine, 3- isopropyltyrosine, O-mannopyranosyl serine, 2-methoxyphenylalanine, 3 -methoxyphenylalanine,
4-methoxyphenylalanine, 3 -methylphenylalanine, O-methyltyrosine, 3-(2-naphthyl)alanine, 5- nitrohistidine, 4-nitrohistidine, 4-nitroleucine, 2-nitrophenylalanine, 3 -nitrophenylalanine, 4- nitrophenylalanine, 4-nitrotryptophan, 5 -nitrotryptophan, 6-nitrotryptophan, 7-nitrotry ptophan, 2-
nitrotyrosine, 3 -nitrotyrosine, O-phosphoserine, O-phosphotyrosine, 4- propargyloxyphenylalanine, O-2-propyn-l-yltyrosine, 4-sulfophenylalanine and O-sulfotyrosine. [00217] In some further embodiments, one or more non-natural amino acids is selected from the group consisting of 4-acetyl-L-phenylalanine (para-acetyl-L-phenylalanine (pAF)), 3-O-(N- acetyl-beta-D-glucosaminyl)-L-threonine, N4-(P-N-Acetyl-D-glucosaminyl)-L-asparagine, O- allyl-L-tyrosine, alpha-N-acetylgalactosamine-O-L-serine, alpha-N-acetylgalactosamine-O-L- threonine, 2-aminooctanoic acid, 2-amino-L-phenylalanine, 3-amino-L-phenylalanine, 4-amino- L-phenylalanine, 2-amino-L-tyrosine, 3-amino-L-tyrosine, 4-azido-L-phenylalanine, 4-benzoyl- L-phenylalanine, (2,2-bipyridin-5yl)-L-alanine, 3-borono-L-phenylalanine, 4-borono-L- phenylalanine, 4-bromo-L-phenylalanine, p-carboxymethyl-L-phenylalanine, 4-carboxy-L- phenylalanine, p-cyano-L-phenylalanine, 3,4-dihydroxy-L-phenylalanine (L-DOPA), 4-ethynyl- L-phenylalanine, 2-fluoro-L-phenylalanine, 3-fluoro-L-phenylalanine, 4-fluoro-L-phenylalanine, O-(3-O-D-galactosyl-N-acetyl-beta-D-galactosaminyl)-L-serine, L-homoglutamine, (8- hydroxyquinolin-3-yl)-L-alanine, 4-iodo-L-phenylalanine, 4-isopropyl-L-phenylalanine, O-i- propyl-L-tyrosine, 3-isopropyl-L-tyrosine, O-mannopyranosyl-L-serine, 2-methoxy-L- phenylalanine, 3 -m ethoxy -L-phenylalanine, 4-m ethoxy -L-phenylalanine, 3-methyl-L- phenylalanine, O-methyl-L-tyrosine, 3-(2-naphthyl)-L-alanine, 5-nitro-L-histidine, 4-nitro-L- histidine, 4-nitro-L-leucine, 2-nitro-L-phenylalanine, 3-nitro-L-phenylalanine, 4-nitro-L- phenylalanine, 4-nitro-L-tryptophan, 5-nitro-L-tryptophan, 6-nitro-L-tryptophan, 7-nitro-L- tryptophan, 2-nitro-L-tyrosine, 3-nitro-L-tyrosine, O-phospho-L-serine, O-phospho-L-tyrosine, 4- propargyloxy -L-phenylalanine, O-2-propyn-l-yl-L-tyrosine, 4-sulfo-L-phenylalanine and O- sulfo-L-tyrosine.
[00218] In some embodiments, the non-natural amino acid includes an aminooxy functional group or a functional group that can be transformed into an aminooxy group by reacting with a reagent, preferably under conditions that do not destroy the biological properties of the polypeptide that includes the non-natural amino acid (unless of course such a destruction of biological properties is the purpose of the modification/transformation), or where the transformation can occur under aqueous conditions at a pH between about 4 and about 8. In some embodiments, the non-natural amino acid includes a carbonyl (e.g., an acyl) functional group or a functional group that can be transformed into a carbonyl (e.g., an acyl) group by reacting with a reagent, preferably under conditions that do not destroy the biological properties of the polypeptide that includes the non-natural amino acid (unless of course such a destruction of biological properties is the purpose of the modification/transformation), or where the transformation can occur under aqueous conditions at a pH between about 4 and about 8. Any number of non-natural amino acids can be
introduced into the polypeptide. Non-natural amino acids may also include protected or masked oximes or protected or masked groups that can be transformed into an oxime group after deprotection of the protected group or unmasking of the masked group. Oxime-based non-natural amino acids may be synthesized by methods well known in the art (see for example WO 2013/185117 and WO 2005/074650, the entire contents of each of which are hereby incorporated by reference herein in their entirety), including reaction of a carbonyl-containing non-natural amino acid with a hydroxylamine or aminooxy-containing reagent. Non-natural amino acids may also include protected or masked carbonyl groups, which can be transformed into a carbonyl group (e.g., an acyl group) after deprotection of the protected group or unmasking of the masked group and thereby are available to react with aminooxy groups to form oxime groups. One non-natural amino acid containing an acyl group is p-acetylphenylalanine. The synthesis of p-acetyl-(+/-)- phenylalanine and m-acetyl-(+/-)-phenylalanine is described in Zhang, Z., et al., Biochemistry 42: 6735-6746 (2003), and the synthesis of p-acetyl-L-phenylalanine is described in WO2015/153761A2; the entire contents of each of which are hereby incorporated by reference in their entirety. Other carbonyl-containing amino acids can be similarly prepared.
[00219] The present invention contemplates incorporation of one or more non-natural amino acids into boIFNZ.3 protein. One or more non-natural amino acids may be incorporated at a particular position which does not disrupt activity of the protein. This can be achieved by making "conservative" substitutions, including but not limited to, substituting hydrophobic amino acids with non-natural hydrophobic amino acids, bulky amino acids with non-natural bulky amino acids, hydrophilic amino acids with non-natural hydrophilic amino acids and/or inserting the non-natural amino acid into a location that is not required for activity.
[00220] A variety of biochemical and structural approaches can be employed to select the desired sites for substitution with a non-naturally encoded amino acid within boIFNZ.3. It is readily apparent to those of ordinary skill in the art that any position of the protein or variant is suitable for selection to incorporate a non-natural amino acid, and selection may be based on rational design or by random selection for any or no particular desired purpose. Selection of desired sites may be based on producing a non-natural amino acid polypeptide (which may be further modified or remain unmodified) having any desired property or activity, including but not limited to a receptor binding modulator, receptor activity modulator, modulator of binding to binder partners, binding partner activity modulator, binding partner conformation modulator, dimer or multimer formation, no change to activity or property compared to the native molecule, or manipulating any physical or chemical property of the protein such as solubility, aggregation or stability. Alternatively, the sites identified as critical to biological activity may also be good candidates for substitution with
a non-natural amino acid, again depending on the desired activity sought for the protein. Another alternative would be to simply make serial substitutions in each position on the protein chain with a non-natural amino acid and observe the effect on the activities of the protein. It is readily apparent to those of ordinary skill in the art that any means, technique or method for selecting a position for substitution with a non-natural amino acid into any protein is suitable for use in the methods, techniques and compositions described herein.
[00221] The structure and activity of naturally-occurring mutants of a protein that contain deletions can also be examined to determine regions of the protein that are likely to be tolerant of substitution with a non-natural amino acid. Once residues that are likely to be intolerant to substitution with non-natural amino acids have been eliminated, the impact of proposed substitutions at each of the remaining positions can be examined using methods including, but not limited to, the three-dimensional structure of the relevant protein, and any associated ligands or binding proteins. X-ray crystallographic and NMR structures of many polypeptides are available in the Protein Data Bank (PDB; see world wide web for rcsb.org), a centralized database containing three-dimensional structural data of large molecules of proteins and nucleic acids, which can be used to identify amino acid positions that can be substituted with non-natural amino acids. In addition, models may be made investigating the secondary and tertiary structure of polypeptides, if three-dimensional structural data are not available. Thus, those of ordinary skill in the art can readily identify amino acid positions that can be substituted with non-natural amino acids.
[00222] Exemplary sites of incorporation of a non-natural amino acid include, but are not limited to, those that are excluded from potential receptor binding regions, or regions for binding to binding proteins or ligands may be fully or partially solvent exposed, have minimal or no hydrogen-bonding interactions with nearby residues, may be minimally exposed to nearby reactive residues, and/or may be in regions that are highly flexible as predicted by the three-dimensional crystal structure of a particular polypeptide with its associated receptor, ligand or binding proteins. A wide variety of non-natural amino acids can be substituted for, or incorporated into, a given position in a polypeptide. By way of example, a particular non-natural amino acid may be selected for incorporation based on an examination of the three-dimensional crystal structure of a polypeptide with its associated ligand, receptor and/or binding proteins, with a preference for conservative substitutions.
[00223] One of ordinary skill in the art recognizes that such analysis of boIFNZ.3 protein enables the determination of which amino acid residues are surface exposed compared to amino acid residues that are buried within the tertiary structure of the protein. Therefore, it is an embodiment of the present invention to substitute a non-naturally encoded amino acid for an amino acid that is
a surface exposed residue. Thus, non-natural amino acid site selection can be based on surface exposure/site accessibility within the protein. Hydrophobic or neutral amino acid sites can be selected to maintain the charge on the protein. In some embodiments, non-naturally encoded amino acid site selection is based on surface exposure. For example, one possible site is an amino acid having a solvent accessible surface area ratio of 30% or more, 40% or more, 50% or more, 60% or more, 70% or more, 80% or more, 90% or more, or 95% or more. In some embodiments, one possible site is an amino acid having a solvent accessible surface area ratio of about 30%, about 40%, about 50%, about 60%, about 70%, about 80%, about 90%, or about 95%, or more. The solvent accessible surface area can be calculated based on the DSSP program (Kabsch W. and Sander C. (1983) Biopolymers, 22:2577-2637), using a crystalline structure analyzing data file of proteins registered in the PDB.
[00224] The ratio of the solvent accessible surface area of the amino acid residues of interest can be calculated by dividing the protein structural solvent accessible surface area calculated in the above by the solvent accessible surface area of alanine-X-alanine (wherein X represents the amino acid residue(s) of interest). In a case in which two or more PDB files are present for one species of protein, any one or more of them can be used in the present invention.
[00225] Alternatively, the solvent accessibility of an amino acid can be determined by a solvent accessibility test in which a functional group on the amino acid (e.g., a thiol, amino, or carbonyl group) is functionalized when treated with an electrophilic reagent or a nucleophilic reagent, or the like. Based on the test results, the functional group (e.g., the thiol, amino, or carbonyl group) can be called, for example, at least 50% solvent accessible when at least 50% of the functional group is functionalized in the test. In some embodiments, the non-naturally encoded amino acid site is at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% solvent accessible. Examples of solvent accessibility test include, but are not limited to, propargylation of a surface thiol group, or a- bromopyruvate reacting with a surface thiol group, etc.
[00226] Alternatively, a site for non-natural amino acid incorporation can be selected by modeling a desired non-natural amino acid at each amino acid site, e.g., based on a protein crystalline structure of a corresponding protein, and assessing side chain accessible surface area (scASA) of the desired non-natural amino acid. In some embodiments, non-natural amino acid site selection is based on scASA. For example, one possible site is an amino acid having an scASA value of about 98 angstroms2 (A2) or more, about 125 A2 or more, about 150 A2 or more, about 175 A2 or more, about 200 A2 or more, about 225 A2 or more, about 250 A2 or more, or about 275 A2 or more.
[00227] When producing proteins or polypeptides of interest with at least one non-natural amino acid in eukaryotic cells, the proteins or polypeptides will typically include eukaryotic post- translational modifications. Accordingly, in certain embodiments, a protein includes at least one non-natural amino acid and at least one post-translational modification that is made by a eukaryotic cell, where the post-translational modification is not made by a prokaryotic cell. In a non-limiting example, the post-translation modification can include acetylation, acylation, lipid-modification, palmitoylation, palmitate addition, phosphorylation, glycolipid-linkage modification, glycosylation, and the like. Some post-translational modifications can occur at the N-terminus or C -terminus of the polypeptide but are not limited as such.
[00228] In certain embodiments of the disclosure, a protein with at least one non-natural amino acid comprising a first reactive group undergoes at least one post-translational modification. In certain embodiments, the post-translational modification is made in a eukaryotic cell or in a non- eukaryotic cell. In other embodiments the post-translational modification is made in vitro. In another embodiment, the post-translational modification is made in vitro and in vivo. Thus, in certain embodiments, the post-translational modification is through the non-natural amino acid. The molecule to be attached to the protein comprises a second reactive group that is capable of reacting with the first reactive group of the non-natural amino acid, and the molecule is attached to the protein utilizing chemistry methodology that is known to one of ordinary skill in the art to be suitable for the particular reactive groups. For example, the post-translational modification can be through a nucleophilic-electrophilic reaction. Most reactions currently used for the selective modification of proteins involve covalent bond formation between nucleophilic and electrophilic reaction partners. Selectivity in these cases is determined by the number and accessibility of the nucleophilic residues in the protein. In proteins of the invention, other more selective reactions can be used such as the reaction of a non-natural keto-amino acid with hydrazides or aminooxy compounds. See, e.g., Cornish, et al. (1996) J. Am. Chem. Soc., 118:8150-8151; Mahal, et al. (1997) Science, 276: 1125-1128,; Wang, et al. (2001) Science 292:498-500; Chin, et al. (2002) J. Am. Chem. Soc. 124:9026-9027; Chin, et al. (2002) Proc. Natl. Acad. Sci., 99: 11020-11024; Wang, et al. (2003) Proc. Natl. Acad. Sci., 100:56-61; Zhang, et al. (2003) Biochemistry, 42:6735- 6746; and Chin, et al. (2003) Science, 301 :964-7; the contents of each of which are hereby incorporated by reference herein in their entirety. In some embodiments, the first reactive group is a keto moiety (including but not limited to, the acetyl group of the non-natural amino acid paraacetylphenylalanine, or para-acetyl-L-phenylalanine) and the second reactive group is an aminooxy moiety. In another example, the first reactive group is an azido moiety (including but not limited to, the azido group of the non-natural amino acid / /ra-azido-L-phenylalanine) and the
second reactive group is an alkynyl moiety. In one embodiment, a post-translational modification to the protein comprises attachment of any desirable compound or substance, including but not limited to a moiety that is capable of extending a protein’s elimination half-life, as disclosed herein. [00229] In some embodiments disclosed herein, the non-natural amino acid is site-specifically incorporated into the protein or variant. Methods for incorporating a non-natural amino acid into a molecule, for example, proteins, polypeptides or peptides, are disclosed herein, e.g., in the Examples, and in U.S. patent nos.: 7,332,571, 7,928,163, 7,696,312, 8,008,456, 8,048,988, 8,809,511, 8,859,802, 8,791,231, 8,476,411 and 9,637,411; and in PCT publication nos: WO 2018/223108, WO 2010/011735 and WO 2005/074650; the contents of each of which are hereby incorporated by reference herein in their entirety. The one or more non-natural amino acids may be incorporated by methods known in the art. For example, cell-based or cell-free systems may be used, and auxotrophic strains may also be used in place of engineered tRNA and synthetase. In certain embodiments, orthogonal tRNA synthetases are used as disclosed in, for example, WO 2002/085923, WO 2002/086075, WO 2004/035743, WO 2007/021297, WO 2006/068802 and WO 2006/069246; the entire contents of each of which are hereby incorporated by reference herein in their entirety. Incorporating one or more non-natural amino acids into the protein or variant thereof may comprise modifying one or more amino acid residues in the protein or variant. Modifying the one or more amino acid residues in the protein or variant may comprise mutating one or more nucleotides in the nucleic acid encoding the protein or variant. Mutating the one or more nucleotides in the nucleic acid encoding the protein or variant may comprise altering a codon encoding an amino acid to a nonsense codon. Incorporating one or more non-natural amino acids into the protein or variant may comprise modifying one or more nucleotides in the nucleic acid to introduce one or more amber codons into the nucleotide sequence. The one or more non-natural amino acids may be incorporated into the protein or variant in response to an amber codon. The one or more non-natural amino acids may be site-specifically incorporated into the protein or variant. Incorporating one or more non-natural amino acids into the protein or variant may comprise incorporating one or more genetically encoded non-natural amino acids with orthogonal chemical reactivity relative to the 20 canonical amino acids to site-specifically modify the protein or variant. Incorporating the one or more non-natural amino acids may comprise use of a tRNA/aminoacyl-tRNA synthetase pair to site-specifically incorporate one or more non-natural amino acids at defined sites in the protein or variant in response to one or more amber nonsense codon.
[00230] Thus, the boIFNZ.3 protein variants of the invention can be generated using modified tRNA and tRNA synthetases to add to or substitute amino acids that are not encoded in naturally-
occurring systems. Methods for generating tRNAs and tRNA synthetases which use amino acids that are not encoded in naturally-occurring systems are described in, e.g., U.S. Patent Nos. 7,045,337 and 7,083,970, the contents of each of which are hereby incorporated by reference herein in their entirety. These methods involve generating a translational machinery that functions independently of the synthetases and tRNAs endogenous to the translation system (and are therefore sometimes referred to as “orthogonal”). Typically, the translation system comprises an orthogonal tRNA (O-tRNA) and an orthogonal aminoacyl tRNA synthetase (O-RS). Use of O- tRNA/aminoacyl-tRNA synthetases involves selection of a specific codon which encodes the non- naturally encoded amino acid (a selector codon). While any codon can be used, it is generally desirable to select a codon that is rarely or never used in the cell in which the O-tRNA/aminoacyl- tRNA synthetase is expressed. For example, exemplary codons include nonsense codon such as stop codons (amber, ochre, and opal), four or more base codons and other natural three-base codons that are rarely used or unused. Specific selector codon(s) can be introduced into appropriate positions in boIFNZ.3 sequence using mutagenesis methods known in the art (including but not limited to, site-specific mutagenesis, cassette mutagenesis, restriction selection mutagenesis, etc.). Typically, the O-RS preferentially aminoacylates the O-tRNA with at least one non-natural amino acid in the translation system and the O-tRNA recognizes at least one selector codon that is not recognized by other tRNAs in the system. The translation system thus inserts the non-naturally encoded amino acid into a protein produced in the system, in response to an encoded selector codon, thereby "substituting" an amino acid into a position in the encoded polypeptide.
[00231] Thus, in some embodiments, a boIFNZ.3 protein variant of the present disclosure containing a non-natural amino acid is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA synthetase. Thus, in some embodiments, the non-natural amino acid is site-specifically incorporated.
[00232] A wide variety of orthogonal tRNAs and aminoacyl tRNA synthetases have been described in the art for inserting particular synthetic amino acids into polypeptides and are generally suitable for use in the present invention. For example, keto-specific O-tRNA/aminoacyl- tRNA synthetases are described in Wang, L. et al. (2003) Proc. Natl. Acad. Sci. USA 100:56-61 and Zhang, Z. et al. (2003) Biochem. 42(22):6735-6746. Exemplary O-RS, or portions thereof, are encoded by polynucleotide sequences and include amino acid sequences disclosed in U.S. Patent Nos. 7,045,337 and 7,083,970. Corresponding O-tRNA molecules for use with the O-RSs are also described in U.S. Patent Nos. 7,045,337 and 7,083,970. Additional examples of O- tRNA/aminoacyl-tRNA synthetase pairs are described in WO 2005/007870, WO 2005/007624 and
WO 2005/019415, the entire contents of each of which are hereby incorporated by reference herein in their entirety. An example of an azide-specific O-tRNA/aminoacyl-tRNA synthetase system is described in Chin, J. W. et al. (2002) J. Am. Chem. Soc. 124:9026-9027. Exemplary O-RS sequences for /?-azido-L-Phe include, but are not limited to, nucleotide sequences SEQ ID NOs: 14-16 and 29-32 and amino acid sequences SEQ ID NOs: 46-48 and 61-64 as disclosed in U.S. Patent No. 7,083,970. Exemplary O-tRNA sequences suitable for use in the present invention include, but are not limited to, nucleotide sequences SEQ ID NOs: 1-3 as disclosed in U.S. Patent No. 7,083,970. Other examples of O-tRNA/aminoacyl-tRNA synthetase pairs specific to particular non-naturally encoded amino acids are described in U.S. Patent No. 7,045,337. O-RS and O-tRNA that incorporate both keto- and azide-containing amino acids in S. cerevisiae are described in Chin, J. W., et al., Science 301 :964-967 (2003). Several other orthogonal pairs have been reported. Glutaminyl (see, e.g., Liu, D. R., and Schultz, P. G. (1999) Proc. Natl. Acad. Sci. U. S. A. 96:4780-4785), aspartyl (see, e.g., Pastrnak, M., et al., (2000) Helv. Chim. Acta 83:2277- 2286), and tyrosyl (see, e.g., Ohno, S., et al., (1998) J. Biochem. (Tokyo, Jpn.) 124: 1065-1068; and Kowal, A. K., et al., (2001) Proc. Natl. Acad. Sci. U. S. A. 98:2268-2273) systems derived from S. cerevisiae tRNAs and synthetases have been described for the potential incorporation of unnatural amino acids in E. coli. Systems derived from the E. coli glutaminyl (see, e.g., Kowal, A. K., et al., (2001) Proc. Natl. Acad. Sci. U. S. A. 98:2268-2273) and tyrosyl (see, e.g., Edwards, H., and Schimmel, P. (1990) Mol. Cell. Biol. 10: 1633-1641) synthetases have been described for use in S. cerevisiae. The E. coli tyrosyl system has been used for the incorporation of 3-iodo-L- tyrosine in vivo, in mammalian cells. See, Sakamoto, K., et al., (2002) Nucleic Acids Res. 30:4692-4699. Additional methods for incorporating non-natural amino acids include, but are not limited to, methods disclosed in Chatterjee A. et al. (2013) 52(10): 1828-1837; Kazane et al. (2013) J Am Chem Soc, 135(l):340-6; Kim et al. (2012) J Am Chem Soc, 134(24):9918-21; Johnson et al. (2011) Nat Chem Biol, 7(11):779-86; and Hutchins et al. (2011) J Mol Biol, 406(4):595-603. [00233] The one or more non-natural amino acids may be produced through selective reaction of one or more natural amino acids. The selective reaction may be mediated by one or more enzymes. In non-limiting examples, the selective reaction of one or more cysteines with formylglycine generating enzyme (FGE) may produce one or more formylglycines as described in Rabuka et al. (2012) Nature Protocols 7: 1052-1067. The one or more non-natural amino acids may be produced through a method involving a chemical reaction to form a linker. The chemical reaction to form the linker may include a bioorthogonal reaction. The chemical reaction to form the linker may include click chemistry. See, for example W02006/050262, the entire contents of which are hereby incorporated by reference herein in their entirety.
[00234] In some other embodiments, the present disclosure provides a boIFNZ.3 protein or variant thereof comprising an unusual genetically-encoded amino acid that is not present in humans, such as selenocysteine or pyrrolysine. In some embodiments, the boIFNZ.3 protein or variant thereof comprises selenocysteine. In some embodiments, the boIFNZ.3 protein or variant thereof comprises pyrrolysine. In some embodiments, the boIFNZ.3 protein or variant thereof comprising the unusual genetically encoded amino acid does not contain a non-natural amino acid.
[00235] In some embodiments, one or more non-naturally encoded amino acids are incorporated into one or more of the following positions in wild type boIFNZ.3 (SEQ ID NO: 1) or into the corresponding amino acid positions in a boIFNZ.3 variant having an amino acid sequence of SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13: before position 1 (i.e. at the N-terminus), 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100, 101, 102, 103, 104, 105, 106, 107,
108, 109, 110, 111, 112, 113, 114, 115, 116, 117, 118, 119, 120, 121, 122, 123, 124, 125, 126,
127, 128, 129, 130, 131, 132, 133, 134, 135, 136, 137, 138, 139, 140, 141, 142, 143, 144, 145,
146, 147, 148, 149, 150, 151, 152, 153, 154, 155, 156, 157, 158, 159, 160, 161, 162, 163, 164,
165, 166, 167, 168, 169, 170, 171, 172; and/or added to the carboxyl terminus of the protein; and any combination thereof. The non naturally encoded amino acid may substitute for the amino acid present at the relevant position in any one of the recited SEQ ID NOs.
[00236] In some embodiments, one or more non-naturally encoded amino acids are incorporated into one or more of the following positions of wild type boIFNZ.3 (SEQ ID NO: 1), or into the corresponding amino acid positions in a boIFNZ.3 protein variant having an amino acid sequence of SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13: position 15, 58, 65, 89, 119, 122, 123, 126, 139 and/or 169.
[00237] In some embodiments, one non-naturally encoded amino acid is incorporated into position 119 of wild type boIFNZ.3 (SEQ ID NO: 1), or into the corresponding amino acid position in a boIFNZ.3 protein variant having an amino acid sequence of SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13.
[00238] In some embodiments, a boIFNZ.3 protein variant of the present disclosure can have an amino acid sequence disclosed in Table 1, wherein the amino acid sequence comprises a nonnatural amino acid, and wherein the amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22,
23, 24 or 25; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23, 24 or 25, wherein each of the foregoing sequences further comprises an N-terminal methionine.
[00239] In some embodiments, a boIFNZ.3 protein variant of the present disclosure can have an amino acid sequence disclosed in Table 1, wherein the amino acid sequence comprises a nonnatural amino acid, and wherein the amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or
24, optionally wherein each of the foregoing sequences further comprises an N-terminal methionine.
[00240] In some embodiments, there is provided a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the amino acid sequence shares at least 75% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine. In some embodiments, there is provided a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 75% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) shares at least 75% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, the conjugated boIFNZ.3 has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 75% identity with SEQ ID NO: 18. In some embodiments, the conjugated boIFNZ.3 has an amino acid sequence comprising a non- natural amino acid, wherein the amino acid sequence shares at least 75% identity with SEQ ID NO: 19.
[00241] In some embodiments, there is provided a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the amino acid sequence shares at least 80% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine. In some embodiments, there is provided a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 80% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) shares at least 80% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine. In
some embodiments, the boIFNZ.3 protein variant has an amino acid sequence comprising a nonnatural amino acid, wherein the amino acid sequence shares at least 80% identity with SEQ ID NO: 18. In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 80% identity with SEQ ID NO: 19.
[00242] In some embodiments, there is provided a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the amino acid sequence shares at least 85% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine. In some embodiments, there is provided a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 85% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) shares at least 85% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence comprising a non- natural amino acid, wherein the amino acid sequence shares at least 85% identity with SEQ ID NO: 18. In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 85% identity with SEQ ID NO: 19.
[00243] In some embodiments, there is provided a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the amino acid sequence shares at least 90% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine. In some embodiments, there is provided a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence comprising a non- natural amino acid, wherein the amino acid sequence shares at least 90% identity with SEQ ID NO: 18. In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence
comprising a non-natural amino acid, wherein the amino acid sequence shares at least 90% identity with SEQ ID NO: 19.
[00244] In some embodiments, there is provided a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the amino acid sequence shares at least 95% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine. In some embodiments, there is provided a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 95% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24 or (ii) shares at least 95% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence comprising a non- natural amino acid, wherein the amino acid sequence shares at least 95% identity with SEQ ID NO: 18. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 95% identity with SEQ ID NO: 19.
[00245] In some embodiments, there is provided a boIFNz.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the amino acid sequence shares at least 96% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine. In some embodiments, there is provided a boIFNz.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 96% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24 or (ii) shares at least 96% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence comprising a non- natural amino acid, wherein the amino acid sequence shares at least 96% identity with SEQ ID NO: 18. In some embodiments, the boIFNz.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 96% identity with SEQ ID NO: 19.
[00246] In some embodiments, there is provided a boIFNz.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the
amino acid sequence shares at least 97% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine. In some embodiments, there is provided a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 97% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) shares at least 97% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence comprising a nonnatural amino acid, wherein the amino acid sequence shares at least 97% identity with SEQ ID NO: 18. In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 97% identity with SEQ ID NO: 19.
[00247] In some embodiments, there is provided a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the amino acid sequence shares at least 98% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine. In some embodiments, there is provided a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 98% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24, or (ii) shares at least 98% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine. In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence comprising a non- natural amino acid, wherein the amino acid sequence shares at least 98% identity with SEQ ID NO: 18. In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 98% identity with SEQ ID NO: 19.
[00248] In some embodiments, there is provided a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid sequence comprises a non-natural amino acid, and wherein the amino acid sequence shares at least 99% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine. In some embodiments, there is provided a boIFNZ.3 protein variant having an amino acid sequence, wherein the amino acid
sequence comprises a non-natural amino acid and wherein the amino acid sequence (i) shares at least 99% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24, or (ii) shares at least 99% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein variant amino acid sequence further comprises an N-terminal methionine.
[00249] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 18. In some embodiments, the non-natural amino acid is pAF.
[00250] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 19. In some embodiments, the non-natural amino acid is pAF.
[00251] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 14, 16, 17, 18, 19, 20, 21, 22 or 23. In some embodiments, the non- natural amino acid is pAF.
[00252] In some embodiments, the boIFNZ.3 protein variant has an amino acid sequence comprising a non-natural amino acid, wherein the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 17, 18, 20 or 21. In some embodiments, the non-natural amino acid is pAF.
[00253] In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 14. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 14, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00254] In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 15. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 15, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00255] In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 16. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 16, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00256] In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 17. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 17, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00257] In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 18. In some embodiments, the non-natural amino acid is pAF.
[00258] In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 19. In some embodiments, the non-natural amino acid is pAF.
[00259] In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 20. In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 20, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00260] In some embodiments, the boIFNZ.3 protein variant has the amino acid sequence of SEQ ID NO: 21. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 21, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00261] In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 22. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 22, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00262] In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 23. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 23, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00263] In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 24. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 24, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00264] In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 25. In some embodiments, the boIFNz.3 protein variant has the amino acid sequence of SEQ ID NO: 25, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00265] In the section above any definition of a protein or variant can equally be applied to the protein portion of a conjugate, and vice versa.
[00266] The present disclosure also provides a nucleic acid having a nucleotide sequence disclosed in Table 1, wherein the nucleotide sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 27. The present disclosure also
provides a nucleic acid having a nucleotide sequence, wherein the nucleotide sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 27. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 75% identity with SEQ ID NO: 27. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 80% identity with SEQ ID NO: 27. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 85% identity with SEQ ID NO: 27. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 90% identity with SEQ ID NO: 27. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 95% identity with SEQ ID NO: 27. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 96% identity with SEQ ID NO: 27. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 97% identity with SEQ ID NO: 27. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 98% identity with SEQ ID NO: 27. In some embodiments, the nucleic acid has a nucleotide sequence that shares at least 99% identity with SEQ ID NO: 27. In some embodiments, the nucleic acid has a nucleotide sequence that shares less than 100% identity with SEQ ID NO: 26. In some embodiments, the nucleic acid has the nucleotide sequence of SEQ ID NO: 27.
[00267] Conjugated bovine IFN 3 proteins
[00268] The present disclosure provides a conjugated protein comprising an engineered boIFNz.3 protein, such as a boIFN/3 protein variant, and a moiety that is capable of extending the engineered boIFNZ.3 protein elimination half-life. In some embodiments, the engineered boIFNZ.3 protein of said protein conjugate is characterized as having an amino acid sequence comprising a non-natural amino acid. In some embodiments, the moiety that is capable of extending the engineered boIFNZ.3 protein elimination half-life is conjugated to the protein via the non-natural amino acid. In some embodiments, the conjugated protein is characterized as having an elimination half-life. In some embodiments, the conjugated protein elimination half-life is extended (i.e., longer) than the half-life of the corresponding unconjugated engineered boIFNz.3 protein, or a comparable unconjugated protein, such as another unconjugated IFN protein. In some embodiments, the conjugated protein elimination half-life is extended (i.e., longer) than the half-life of unconjugated wild-type boIFNz.3 protein, such as the wild-type boIFNz.3 protein having the amino acid sequence of SEQ ID NO: 1.
[00269] A moiety of the present disclosure that is capable of extending protein elimination halflife, such as a half-life extender molecule or moiety, is capable of modifying one or more characteristics of a biotherapeutic agent relative to its unconjugated form, including but not limited
to its half-life for elimination from the body or a compartment thereof. By way of example only, conjugation of a biotherapeutic agent (such as a boIFNZ.3 protein variant) with such a moiety can modify characteristics including, but not limited to, its pharmacodynamics, pharmacokinetics, water solubility, serum half-life, plasma half-life, therapeutic half-life, bioavailability, renal clearance, biological activity, circulation time, immunogenicity, allergenicity, physical association characteristics including, but not limited to, aggregation and multimer formation, receptor binding, binding to one or more binding partners, and/or receptor dimerization or multimerization. Thus, conjugation of a biotherapeutic agent, such as a boIFNZ.3 protein variant, with a moiety that is capable of extending its half-life can provide the agent with improved pharmacodynamics, improved pharmacokinetics, increased water solubility, increased or modulated serum half-life, increased or modulated plasma half-life, increased or modulated therapeutic half-life, increased bioavailability, reduced or modulated renal clearance, modulated biological activity, extended circulation time, reduced or modulated immunogenicity, reduced or modulated allergenicity, modulated physical association characteristics including, but not limited to, aggregation and multimer formation, altered receptor binding, altered binding to one or more binding partners, and/or altered receptor dimerization or multimerization, relative to its unconjugated form.
[00270] In some embodiments, a moiety that is capable of extending boIFNZ.3 protein elimination half-life is a water-soluble polymer.
[00271] Methods of modulating biotherapeutic agent characteristics via conjugation with another molecule or moiety are known in the art, including HESylation and PEGylation. HESylation involves coupling a biotherapeutic with hydroxyalkylstarch, and in particular, hydroxy ethyl starch (HES). By varying different parameters, such as the HES molecular weight, a wide range of HES conjugates can be customized. Disclosures related to HESylation technology include U.S. Patent Application Publication Numbers US20050063943, US20060121073, US20010100163, US20050234230, US20050238723, US20060019877, US20070134197, US20070087961 and U.S. Patent No. 7,285,661, the entire contents of each of which are hereby incorporated by reference herein in their entirety. But HES shares a common disadvantage with other presently available polymers: its polydispersity. PEGylation involves coupling a biological macromolecule, such as a biotherapeutic agent, with polyethylene glycol (PEG) or a derivative thereof (e.g., see Harris J.M. and Chess R.B. (2003) Nature Reviews Drug Discovery, 2:214-221; Roberts M.J. and Harris J.M (2002) Adv. Drug Delivery Reviews, 54(4):459-476). Much of the art in the field of protein derivatization with PEG has been directed to developing PEG derivatives for attachment to the epsilon -NH2 moiety of lysine residues present in proteins (see, e.g., “Polyethylene Glycol and Derivatives for Advanced PEGylation”, Nektar Molecular Engineering Catalog (2003) pp. 1-
17). These PEG derivatives share a common limitation of non-selectively among the lysine residues present on the surfaces of proteins. This can be a significant limitation in instances where a lysine residue is important to protein activity, including cases where a lysine residue plays a role in mediating the interaction of the protein with other biological molecules, as in the case of receptor binding sites. A second and equally important complication of existing methods for protein PEGylation is that the PEG derivatives can undergo undesired side reactions with residues other than those desired. In some instances, the PEG derivatives directed at the epsilon -NH2 group of lysine also react with cysteine, histidine or other residues. This can create complex, heterogeneous mixtures of PEG-derivatized bioactive molecules at the risk of attenuating or even destroying the desired activity of the bioactive molecule. In addition to lysine residues, considerable effort has been directed toward the development of activated PEG reagents that target other amino acid side chains, including cysteine and histidine, or the N-terminus (see, e.g., U.S. Pat. No. 6,610,281 and “Polyethylene Glycol and Derivatives for Advanced PEGylation”, Nektar Molecular Engineering Catalog (2003) pp. 1-17). A cysteine residue can be introduced site-selectively into the structure of proteins using site-directed mutagenesis and other techniques known in the art, and the resulting free sulfhydryl moiety can be reacted with PEG derivatives that bear thiol-reactive functional groups. This approach is complicated, however, in that the introduction of a free sulfhydryl group can complicate the expression, folding and stability of the resulting protein. Thus, it would be desirable to have a means to site-specifically and predictably introduce chemical functional groups into biotherapeutic proteins, thereby enabling the selective conjugation of the therapeutic protein with one or more moieties that are capable of extending its elimination half-life (and/or capable of modulating other characteristics, as disclosed herein) while simultaneously being compatible with (i.e., not engaging in undesired side reactions with) sulfhydryls, epsilon amino groups and other chemical functional groups that are present in the protein.
[00272] The present disclosure provides for such means, and accordingly, for compositions comprising a boIFNz.3 protein variant site-specifically conjugated to a moiety that is capable of extending boIFNZ.3 protein elimination half-life. The moiety is site-specifically conjugated to the boIFNZ.3 protein variant via a non-natural amino acid in the protein sequence. As disclosed herein, the non-natural amino acid contains chemical functional groups that are inert to the functional groups found in the 20 canonical amino acids but react cleanly and efficiently with other functional groups, such as those present in water-soluble polymers and derivatives thereof, as provided herein, to form stable linkages in a site-specific manner.
[00273] In some embodiments, the moiety that is capable of extending boIFNz.3 protein elimination half-life is one or more moieties, the non-natural amino acid is one or more non-natural
amino acids, and each of the one or more moieties is conjugated to the boIFNz.3 protein variant via each of the one or more non-natural amino acids. In some embodiments, the one or more moieties is one moiety, and the one or more non-natural amino acids is one non-natural amino acid. In some embodiments, the one, or the one or more, non-natural amino acids is a non-naturally encoded amino acid. In some embodiments, the non-natural amino acid is para-acetyl-L- phenylalanine. In some other embodiments, the non-natural amino acid is para-azido-L- phenylalanine.
[00274] Typically, the moiety that is capable of extending the boIFNZ.3 protein elimination halflife is a water-soluble polymer. Thus, the present disclosure provides for boIFNz.3 protein variant site-specifically conjugated to one or more water-soluble polymers via one or more non-natural amino acids in the protein sequence. In some embodiments, the one or more water-soluble polymers is one water-soluble polymer, and the one or more non-natural amino acids is one non- natural amino acid.
[00275] Water-soluble polymers of the present disclosure include, but are not limited to, polyethylene glycol (PEG), polyethylene glycol propionaldehyde, mono C1-C10 alkoxy or aryloxy derivatives thereof (described in U.S. PatentNo. 5,252,714, the entire contents of which are hereby incorporated by reference herein in their entirety), monomethoxy-polyethylene glycol, polyvinyl pyrrolidone, polyvinyl alcohol, polyamino acids, divinylether maleic anhydride, N-(2- hydroxypropyl)-methacrylamide, dextran, dextran derivatives including dextran sulfate, polypropylene glycol, polypropylene oxide/ethylene oxide copolymer, polyoxyethylated polyol, polyethers (which include PEGs and derivatives thereof), heparin, heparin fragments, polysaccharides, oligosaccharides, glycans, cellulose and cellulose derivatives, including but not limited to methylcellulose and carboxymethyl cellulose, albumin (including serum albumin), starch and starch derivatives (including hydroxy alkyl starch, or more particularly, hydroxy ethyl starch (HES)), polypeptides, polyalkylene glycol and derivatives thereof (including but not limited to polyethylene glycol (PEG), polypropylene glycol, polybutylene glycol, and derivatives thereof), copolymers of polyalkylene glycols and derivatives thereof, polyoxyalkylene glycol and derivatives thereof, copolymers of polyoxyalkylene glycol and derivatives thereof, polyvinyl ethyl ethers, and alpha-beta-poly(N-2-hydroxyethyl)-DL-aspartamide, and the like, or combinations thereof. The present disclosure includes derivatives of any of the foregoing water- soluble polymers. Such water-soluble polymer derivatives include water-soluble polymers comprising reactive group(s) that are capable of reacting with complementary reactive groups present in non-natural amino acids of the present disclosure. In a non-limiting example, a water- soluble polymer derivative of the present disclosure includes PEG derivatives comprising reactive
aminooxy groups that are capable of reacting with carbonyl groups of non-natural amino acids (e.g., acyl groups of the non-natural amino acid pAF). Water-soluble polymers and their derivatives may or may not have their own biological activity.
[00276] More particularly, the present disclosure provides boIFNZ.3 protein variants conjugated to water-soluble polymer(s) including polyalkylene glycols and derivatives thereof. Such polyalkylene glycols include, but are not limited to, polyethylene glycol (PEG), polypropylene glycol, polybutylene glycol, and derivatives thereof. Other exemplary embodiments are listed, for example, in commercial supplier catalogs, such as Shearwater Corporation's catalog “Polyethylene Glycol and Derivatives for Biomedical Applications” (2001). By way of example only, such polyalkylene glycols can have average molecular weights within a range of about 0.1 kDa to about 100 kDa. By way of further example, such polyalkylene glycols include, but are not limited to, polyalkylene glycols having a molecular weight within a range of about 100 Da to about 100,000 Da or more. Thus, the molecular weight (which can be an average molecular weight) of a polyalkylene glycol of the present disclosure may be within a range of about 100 Da to about 100,000 Da, including but not limited to, about 100,000 Da, about 95,000 Da, about 90,000 Da, about 85,000 Da, about 80,000 Da, about 75,000 Da, about 70,000 Da, about 65,000 Da, about 60,000 Da, about 55,000 Da, about 50,000 Da, about 45,000 Da, about 40,000 Da, about 35,000 Da, about 30,000 Da, about 25,000 Da, about 20,000 Da, about 15,000 Da, about 10,000 Da, about 9,000 Da, about 8,000 Da, about 7,000 Da, about 6,000 Da, about 5,000 Da, about 4,000 Da, about 3,000 Da, about 2,000 Da, about 1,000 Da, about 900 Da, about 800 Da, about 700 Da, about 600 Da, about 500 Da, 400 Da, about 300 Da, about 200 Da or about 100 Da. In some embodiments, the molecular weight of the polyalkylene glycol is within a range of about 100 Da to about 50,000 Da. In some embodiments, the molecular weight of the polyalkylene glycol is within a range of about 100 Da to about 40,000 Da. In some embodiments, the molecular weight of the poly alkylene glycol is within a range of about 1,000 Da to about 40,000 Da. In some embodiments, the molecular weight of the polyalkylene glycol is within a range of about 2,000 to about 50,000 Da. In some embodiments, the molecular weight of the polyalkylene glycol is within a range of about 5,000 Da to about 40,000 Da. In some embodiments, the molecular weight of the polyalkylene glycols is within a range of about 10,000 Da to about 40,000 Da. In some embodiments, the molecular weight of the polyalkylene glycol is at most about 100 kDa. In some embodiments, the molecular weight of the polyalkylene glycol is at most about 50 kDa. In some embodiments, the molecular weight of the polyalkylene glycol is at least about 0.1 kDa and at most about 100 kDa. In some embodiments, the molecular weight of the polyalkylene glycol is within a range of about 10 kDa to about 50 kDa. In some embodiments, the molecular weight of the polyalkylene glycol
is within a range of about 20 kDa to about 40 kDa. In some embodiments, the molecular weight of the polyalkylene glycol is within a range of about 25 kDa to about 35 kDa. In some embodiments, the molecular weight of the polyalkylene glycol is about 25 kDa, about 26 kDa, about 27 kDa, about 28 kDa, about 29 kDa, about 30 kDa, about 31 kDa, about 32, kDa, about 33 kDa, about 34 kDa or about 35 kDa. In some embodiments, the molecular weight of the polyalkylene glycol is about 30 kDa. In some embodiments, there is provided any one of the foregoing polyalkylene glycols, wherein the polyalkylene glycol is a linear chain polyalkylene glycol. In some other embodiments, there is provided any one of the foregoing polyalkylene glycols, wherein the polyalkylene glycol is a branched chain polyalkylene glycol. In some other embodiments, there is provided any one of the foregoing polyalkylene glycols, wherein the polyalkylene glycol is a multiarm polyalkylene glycol. The present disclosure includes derivatives of any of the foregoing polyalkylene glycols. Such polyalkylene glycol derivatives include polyalkylene glycol derivatives comprising reactive group(s) that are capable of reacting with complementary reactive groups present in non-natural amino acids present in proteins of the present disclosure. In a non-limiting example, a polyalkylene glycol derivative includes polyalkylene glycol derivatives comprising reactive aminooxy groups that are capable of reacting with carbonyl groups of non-natural amino acids (e.g., acyl groups of the non-natural amino acid pAF) present in proteins of the present disclosure. Thus, the present disclosure provides a boIFNZ.3 protein variant conjugated to a water-soluble polymer via a non-natural amino acid, wherein the water-soluble polymer is a polyalkylene glycol derivative, wherein, prior to the conjugation with the boIFNZ.3 protein variant, said polyalkylene glycol derivative comprises an aminooxy group. In some embodiments, the non-natural amino acid is pAF, and upon conjugation with the polyalkylene glycol derivative, the covalent linkage comprises an oxime. In some embodiments, the non-natural amino acid is pAF, and upon conjugation with the polyalkylene glycol derivative, the covalent linkage consists of an oxime. In some other embodiments, there is provided boIFNZ.3 protein variant conjugated to a water-soluble polymer via a non-natural amino acid, wherein the water-soluble polymer is a polyalkylene glycol derivative, wherein, prior to the conjugation with the boIFNZ.3 protein variant, said polyalkylene glycol derivative comprises an azide group. In some embodiments, the non-natural amino acid that reacts with the azide group comprises an alkynyl group, such as a cyclooctynyl group. In some embodiments, the non-natural amino acid comprises the azide, and the water-soluble polymer comprises the alkyne. In some embodiments the non-natural amino acid comprising the azide is p-azido-L-phenylalanine.
[00277] In some more particular embodiments, a boIFNZ.3 protein variant of the present disclosure is conjugated to a polyalkylene glycol or derivatives thereof, wherein the polyalkylene
glycol is a polyethylene glycol (PEG) or a derivative thereof. By way of example, such PEGs include, but are not limited to, PEGs having an average molecular weight within a range of about 100 Da to about 100,000 Da or more. Thus, the molecular weight (which can be an average molecular weight) of a PEG of the present disclosure may be within a range of about 100 Da to about 100,000 Da, including but not limited to, about 100,000 Da, about 95,000 Da, about 90,000 Da, about 85,000 Da, about 80,000 Da, about 75,000 Da, about 70,000 Da, about 65,000 Da, about 60,000 Da, about 55,000 Da, about 50,000 Da, about 45,000 Da, about 40,000 Da, about 35,000 Da, about 30,000 Da, about 25,000 Da, about 20,000 Da, about 15,000 Da, about 10,000 Da, about 9,000 Da, about 8,000 Da, about 7,000 Da, about 6,000 Da, about 5,000 Da, about 4,000 Da, about 3,000 Da, about 2,000 Da, about 1,000 Da, about 900 Da, about 800 Da, about 700 Da, about 600 Da, about 500 Da, 400 Da, about 300 Da, about 200 Da or about 100 Da. In some embodiments molecular weight of the PEG is within a range of about 100 Da to about 50,000 Da. In some embodiments, the molecular weight of the PEG is within a range of about 100 Da to about 40,000 Da. In some embodiments, the molecular weight of the PEG is within a range of about 1,000 Da to about 40,000 Da. In some embodiments, the molecular weight of the PEG is within a range of about 2,000 to about 50,000 Da. In some embodiments, the molecular weight of the PEG is within a range of about 5,000 Da to about 40,000 Da. In some embodiments, the molecular weight of the PEG is within a range of about 10,000 Da to about 40,000 Da. In some embodiments, the molecular weight of the PEG is at most about 100 kDa. In some embodiments, the molecular weight of the PEG is at most about 50 kDa. In some embodiments, the molecular weight of the PEG is at least about 0.1 kDa and at most about 100 kDa. In some embodiments, the molecular weight of the PEG is within a range of about 10 kDa to about 50 kDa. In some embodiments, the molecular weight of the PEG is within a range of about 20 kDa to about 40 kDa. In some embodiments, the molecular weight of the PEG is within a range of about 25 kDa to about 35 kDa. In some embodiments, the molecular weight of the PEG is about 25 kDa, about 26 kDa, about 27 kDa, about 28 kDa, about 29 kDa, about 30 kDa, about 31 kDa, about 32, kDa, about 33 kDa, about 34 kDa or about 35 kDa. In some embodiments, the molecular weight of the PEG is about 30 kDa. In some embodiments, there is provided any one of the foregoing PEGs, wherein the PEG is a linear chain PEG. In some other embodiments, there is provided any one of the foregoing PEGs, wherein the PEG is a branched chain PEG. In some other embodiments, there is provided any one of the foregoing PEGs, wherein the PEG is a multiarm PEG. The present disclosure includes derivatives of any of the foregoing PEGs. Such PEG derivatives include PEG derivatives comprising reactive group(s) that are capable of reacting with complementary reactive groups present in non-natural amino acids present in proteins of the present disclosure. In a non-limiting
example, a PEG derivative of the present disclosure includes PEG derivatives comprising reactive aminooxy groups that are capable of reacting with carbonyl groups of non-natural amino acids (e.g., acyl groups of the non-natural amino acid pAF) present in proteins of the present disclosure. Activated and functionalized PEGs, including aminooxy PEG derivatives, can be obtained from commercial suppliers such as Biopharma PEG (Biochempeg) Scientific Inc., Watertown, MA and MilliporeSigma, Burlington, MA. Thus, the present disclosure provides boIFNZ.3 protein variant conjugated to a water-soluble polymer via a non-natural amino acid, wherein the water-soluble polymer is a PEG derivative, wherein, prior to the conjugation with the boIFN/3 protein variant, said PEG derivative comprises an aminooxy group. In some embodiments, the non-natural amino acid is pAF, and upon conjugation with the PEG derivative, the covalent linkage comprises an oxime. In some embodiments, the non-natural amino acid is pAF, and upon conjugation with the PEG derivative, the covalent linkage consists of an oxime.
[00278] In some aspects, the present disclosure provides a conjugated protein comprising: an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid; and a water-soluble polymer; wherein the water-soluble polymer is conjugated to the engineered boIFNZ.3 protein. In some embodiments, the water-soluble polymer is conjugated to the engineered boIFNz.3 protein via the non-natural amino acid. In some embodiments, the water- soluble polymer is conjugated to the engineered boIFNz.3 protein via a covalent linkage with the non-natural amino acid. In some embodiments, the water-soluble polymer is one or more water- soluble polymers, the non-natural amino acid is one or more non-natural amino acids, and each of the one or more water-soluble polymers is conjugated to the engineered boIFNz.3 protein via each of the one or more non-natural amino acids. In some embodiments, the one or more water-soluble polymers is one water-soluble polymer, and the one or more non-natural amino acids is one non- natural amino acid.
[00279] In some embodiments, each of the one or more water-soluble polymers is independently selected from the group consisting of a carbohydrate, a polypeptide and a polyalkylene glycol; and derivatives thereof. In some embodiments, each water-soluble polymer is a polyalkylene glycol or a derivative thereof. In some embodiments, the polyalkylene glycol, or the derivative, thereof is a linear polyalkylene glycol, or a derivative thereof. In some embodiments, the polyalkylene glycol, or the derivative, thereof is a branched polyalkylene glycol, or a derivative thereof. In some embodiments, the polyalkylene glycol, or the derivative, thereof is a multiarm polyalkylene glycol, or a derivative thereof. In some embodiments, the polyalkylene glycol is a polyalkylene glycol derivative, wherein, prior to the conjugation with the engineered boIFNz.3 protein, the polyalkylene glycol derivative comprises a group that is capable of forming an oxime. In some
embodiments, the polyalkylene glycol is a polyalkylene glycol derivative, wherein, prior to the conjugation with the engineered boIFNZ.3 protein, the polyalkylene glycol derivative comprises an aminooxy group. In some embodiments, the covalent linkage conjugating the engineered boIFNZ.3 protein with the polyalkylene glycol derivative comprises an oxime. In some embodiments, the covalent linkage conjugating the engineered boIFNZ.3 protein with the polyalkylene glycol derivative consists of an oxime.
[00280] In some embodiments, the polyalkylene glycol or the derivative thereof has a molecular weight of at least about 0.1 kDa. In some embodiments, the polyalkylene glycol or the derivative thereof has a molecular weight of at most about 100 kDa. In some embodiments, the polyalkylene glycol or the derivative thereof has a molecular weight of at most about 50 kDa.
[00281] In some embodiments, the polyalkylene glycol or derivative thereof is a PEG, or a derivative thereof. In some embodiments, the PEG or the derivative thereof is a linear PEG or a derivative thereof. In some embodiments, the PEG is a PEG derivative, wherein, prior to the conjugation with the engineered boIFNZ.3 protein, the PEG derivative comprises a group that is capable of forming an oxime. In some embodiments, the PEG is a PEG derivative, wherein, prior to the conjugation with the engineered boIFNZ.3 protein, the PEG derivative comprises an aminooxy group. In some embodiments, the covalent linkage conjugating the engineered boIFNZ.3 protein with the PEG derivative comprises, or consists of, an oxime.
[00282] In some embodiments, the conjugated protein comprises a non-natural amino acid selected from the group consisting of 4-acetyl-L-phenylalanine (para-acetyl-L-phenylalanine (pAF)), 3-O-(N-acetyl-beta-D-glucosaminyl)-L-threonine, N4-(P-N-Acetyl-D-glucosaminyl)-L- asparagine, O-allyl-L-tyrosine, alpha-N-acetylgalactosamine-O-L-serine, alpha-N- acetylgalactosamine-O-L-threonine, 2-aminooctanoic acid, 2-amino-L-phenylalanine, 3-amino-L- phenylal anine, 4-amino-L-phenylalanine, 2-amino-L-tyrosine, 3-amino-L-tyrosine, 4-azido-L- phenylalanine, 4-b enzoy 1 -L-pheny 1 al anine, (2,2-bipyridin-5yl)-L-alanine, 3-borono-L- phenylalanine, 4-borono-L-phenylalanine, 4-bromo-L-phenylalanine, p-carboxymethyl-L- phenylalanine, 4-carboxy-L-phenylalanine, p-cyano-L-phenylalanine, 3,4-dihydroxy-L- phenylalanine (L-DOPA), 4-ethynyl-L-phenylalanine, 2-fluoro-L-phenylalanine, 3-fluoro-L- phenylalanine, 4-fluoro-L-phenylalanine, O-(3-O-D-galactosyl-N-acetyl-beta-D-galactosaminyl)- L-serine, L-homoglutamine, (8-hydroxyquinolin-3-yl)-L-alanine, 4-iodo-L-phenylalanine, 4- isopropyl-L-phenylalanine, O-i -propyl -L-tyrosine, 3-isopropyl-L-tyrosine, O-mannopyranosyl-L- serine, 2-m ethoxy -L-phenylalanine, 3 -methoxy -L-phenylalanine, 4-m ethoxy -L-phenylalanine, 3- methyl-L-phenylalanine, O-methyl-L-tyrosine, 3-(2-naphthyl)-L-alanine, 5-nitro-L-histidine, 4- nitro-L-histidine, 4-nitro-L-leucine, 2-nitro-L-phenylalanine, 3-nitro-L-phenylalanine, 4-nitro-L-
phenylalanine, 4-nitro-L-tryptophan, 5-nitro-L-tryptophan, 6-nitro-L-tryptophan, 7-nitro-L- tryptophan, 2-nitro-L-tyrosine, 3-nitro-L-tyrosine, O-phospho-L-serine, O-phospho-L-tyrosine, 4- propargyloxy-L-phenylalanine, O-2-propyn-l-yl-L-tyrosine, 4-sulfo-L-phenylalanine and O- sulfo-L-tyrosine. In some embodiments, the non-natural amino acid is para-acetyl-L- phenylalanine (pAF). In some embodiments, the non-natural amino acid is a non-naturally encoded amino acid.
[00283] In some embodiments, the engineered boIFNz.3 protein comprising a non-natural amino acid is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA synthetase. Thus, in some embodiments, the non-natural amino acid is site-specifically incorporated.
[00284] Methods of conjugating a water-soluble polymer to a protein or peptide are known to a person of ordinary skill in the art, and the present disclosure provides for such methods, including methods disclosed in the Examples herein.
[00285] In some embodiments, a water-soluble polymer is conjugated to an engineered boIFNz.3 protein of the present disclosure by reacting a water-soluble polymer containing a reactive group with the engineered boIFNz.3 protein containing one or more natural or non-natural amino acids. The conjugation reaction provides a protein conjugate, wherein the water-soluble polymer is conjugated to a natural or non-natural amino acid of the engineered boIFNz.3 protein via a covalent linkage. The covalent linkage can be a product of the reactive group of the water-soluble polymer and an additional moiety present in the natural or non-natural amino acid, wherein the additional moiety can react to form the covalent linkage with the reactive group of the water-soluble polymer. Several reviews and monographs on the functionalization and conjugation of the water-soluble polymer PEG are available, as are examples of cytokine PEGylation. See, for example, Harris, J.M. (1985) J. Macromolecular Sci., Part C, Polymer Reviews, 25(3):325-373; Scouten, W.H. (1987) Methods in Enzymology, 135:30-65; Wong S.S. and Wong L-J.C. (1992) Enzyme Microb. Technol. 14(11):866-874; Zeuzem, S. et al. (2003) Seminars in Liver Disease 23:023-028; Zalipsky, S. (1995) Bioconjugate Chem. 6(2): 150-165; Bansal R. et al. (2011) J. Controlled Release 154(3):233-240; and WO2017222940A1; the contents of each of which are hereby incorporated herein by reference in their entirety.
[00286] Non-limiting examples of reactions and linkages formed between water-soluble polymers and natural or non-natural amino acids that are present in an engineered boIFNz.3 protein of the present disclosure, include those disclosed in the following paragraphs A to I.
[00287] A. (i) Reaction of a water-soluble polymer comprising reactive group -N3 with a boIFNk3protein, wherein the protein contains a non-natural amino acid comprising an alkynyl
group, thereby providing a linkage comprising a 1,2,3-triazolyl moiety; or (ii) reaction of a water- soluble polymer comprising a reactive alkynyl group with a boIFNZ.3 protein, wherein the protein contains a non-natural amino acid comprising -N3, thereby providing a linkage comprising a 1,2,3- triazolyl moiety. In some embodiments, the alkynyl group is a cyclooctynyl group. In some embodiments, the non-natural amino acid is p-azido-L-phenylalanine. In some embodiments, the linkage comprising the 1,2,3-triazolyl moiety has the following structure:
wherein: each s is independently 0 or an integer from 1 to 50; each t is independently 0 or an integer from 1 to 50; each + denotes connection to the water-soluble polymer; and each wavy line denotes connection to the protein. In some embodiments, each s is independently 0, 1, 2, 3, 4, 5 or 6. In some embodiments, each t is independently 0, 1, 2, 3, 4, 5 or 6.
[00288] B. (i) Reaction of a water-soluble polymer comprising a reactive tetrazinyl group with a boIFNZ.3 protein, wherein the protein contains a non-natural amino acid comprising an (E)- cyclooctenyl group, thereby providing a linkage comprising a 1,4-dihydropyridazinyl moiety; or (ii) reaction of a water-soluble polymer comprising a reactive (E)-cyclooctenyl group with a boIFNZ.3 protein, wherein the protein contains a non-natural amino acid comprising a tetrazinyl group, thereby providing a linkage comprising a 1,4-dihydropyridazinyl moiety. In some embodiments, the linkage comprising the 1,4-dihydropyridazinyl moiety has the following structure:
wherein: each Rf is independently H or alkyl; each + denotes connection to a water-soluble polymer; and each wavy line denotes connection to the protein. In some embodiments, each Rf is independently unsubstituted C1-C6 alkyl.
[00289] C. (i) Reaction of a water-soluble polymer comprising an -0NH2 group with a boIFN/3 protein, wherein the protein contains a natural or non-natural amino acid comprising a carbonylcontaining group (e.g., a ketone group), thereby providing a linkage comprising an oxime moiety; or (ii) reaction of a water-soluble polymer comprising a carbonyl (e.g., ketone) group with a boIFNZ.3 protein, wherein the protein contains a non-natural amino acid comprising an -0NH2 group, thereby providing a linkage comprising an oxime moiety. In some embodiments, the carbonyl or ketone group is -C(O)Rc, wherein Rc is unsubstituted C1-C6 alkyl. In some embodiments, Rc is methyl. In some embodiments, the linkage comprising the oxime moiety has the following structure:
wherein: each Rc is independently unsubstituted C1-C6 alkyl; each + denotes connection to a water-soluble polymer; and each wavy line denotes connection to the protein. In some embodiments, each Rc is methyl.
[00290] D. (i) Reaction of a water-soluble polymer comprising a maleimide group with a boIFNZ.3 protein, wherein the protein contains a natural or non-natural amino acid comprising a thiol (-SH), thereby providing a linkage comprising a pyrrolidine-2, 5-dione moiety, such as a 3- (kl-sulfaneyl)pyrrolidine-2, 5-dione moiety; or (ii) reaction of a water-soluble polymer comprising comprising a thiol (-SH) group with a boIFNZ.3 protein, wherein the protein contains a non-natural amino acid comprising a maleimide group, thereby providing a linkage comprising a pyrrolidine- 2, 5-dione moiety, such as a 3 - -sulfaneyl)pyrrolidine-2, 5-dione moiety. In some embodiments, the natural amino acid is cysteine. In some embodiments, the linkage comprising the a pyrrolidine- 2, 5-dione moiety, such as a 3-(kl-sulfaneyl)pyrrolidine-2, 5-dione moiety, has the following structure:
wherein: each + denotes connection to a water-soluble polymer; and each wavy line denotes connection to the protein.
[00291] E. (i) Reaction of a water-soluble polymer comprising a primary or secondary amine with a boIFNZ.3 protein, wherein the protein contains a natural or non-natural amino acid comprising a carboxylic acid group, a protected carboxylic acid, or an activated ester group, thereby providing a linkage comprising an amide moiety; or (ii) reaction of a water-soluble polymer comprising a carboxylic acid group, a protected carboxylic acid, or an activated ester group with a boIFNZ.3 protein, wherein the protein contains a natural or non-natural amino acid comprising a primary or secondary amine, thereby providing a linkage comprising an amide moiety. In some embodiments, the natural amino acid is aspartic acid or glutamic acid. In some other embodiments, the natural amino acid is lysine. In some embodiments, the reaction is a peptide coupling reaction or other well-known method of forming an amide, each of which can be performed using methods readily understood by a person of ordinary skill in the art. In some embodiments, the linkage comprising the amide moiety has the following structure:
wherein: each Rb is independently H or alkyl; each + denotes connection to a water-soluble polymer; and each wavy line denotes connection to the protein. In some embodiments, each Rb is independently alkyl, wherein the alkyl is unsubstituted C1-C6 alkyl.
[00292] F. (i) Reaction of a water-soluble polymer comprising a hydroxyl group (-OH) with a boIFNZ.3 protein, wherein the protein contains a natural or non-natural amino acid comprising a carboxylic acid group, a protected carboxylic acid, or an activated ester group, thereby providing a linkage comprising an ester moiety; or (ii) reaction of a water-soluble polymer comprising a carboxylic acid group, a protected carboxylic acid, or an activated ester group with a boIFNZ.3 protein, wherein the protein contains a natural or non-natural amino acid comprising a hydroxyl group, thereby providing a linkage comprising an ester moiety. In some embodiments, the natural amino acid is aspartic acid or glutamic acid. In some other embodiments, the natural amino acid is serine, threonine or tyrosine. Methods of forming such esters linkages can be performed using
methods readily understood by a person of ordinary skill in the art. In some embodiments, the linkage comprising the ester moiety has the following structure:
wherein: each + denotes connection to a water-soluble polymer; and each wavy line denotes connection to the protein.
[00293] G. (i) Reaction of a water-soluble polymer comprising a thiol group (-SH) with a boIFNZ.3 protein, wherein the protein contains a natural or non-natural amino acid comprising a carboxylic acid group, a protected carboxylic acid, or an activated ester group, thereby providing a linkage comprising a thioester moiety; or (ii) reaction of a water-soluble polymer comprising a carboxylic acid group, a protected carboxylic acid, or an activated ester group with a boIFNZ.3 protein, wherein the protein contains a natural or non-natural amino acid comprising a thiol group, thereby providing a linkage comprising a thioester moiety. In some embodiments, the natural amino acid is aspartic acid or glutamic acid. In some other embodiments, the natural amino acid is cysteine. Methods of forming such thioesters linkages can be performed using methods readily understood by a person of ordinary skill in the art. In some embodiments, the linkage comprising the ester moiety has the following structure:
wherein: each + denotes connection to a water-soluble polymer; and each wavy line denotes connection to the protein.
[00294] H. Reaction of a water-soluble polymer comprising a -C(O)CH2NH2 group with a boIFNZ.3 protein, wherein the protein contains a natural or non-natural amino acid comprising a carboxylic acid group, a protected carboxylic acid, or an activated ester group, thereby providing a linkage comprising a -C(O)CH2NHC(O)- moiety; or (ii) reaction of a water-soluble polymer comprising a carboxylic acid group, a protected carboxylic acid, or an activated ester group with a boIFNZ.3 protein, wherein the protein contains a non-natural amino acid comprising a - C(O)CH2NH2 group, thereby providing a linkage comprising a -C(O)CH2NHC(O)- moiety. In some embodiments, the natural amino acid is aspartic acid or glutamic acid. Methods of forming such linkages can be performed using methods readily understood by a person of ordinary skill in the art. In some embodiments, the linkage has the following structure:
wherein: each + denotes connection to water-soluble polymer; and each wavy line denotes connection to the protein.
[00295] I. Reaction of a water-soluble polymer comprising a thiol group (-SH) with a boIFNZ.3 protein, wherein the protein contains a natural or non-natural amino acid comprising a thiol group, thereby providing a linkage comprising a disulfide. In some embodiments, the natural amino acid is cysteine. Methods of forming disulfide linkages can be performed using methods readily understood by a person of ordinary skill in the art.
[00296] Thus, in some embodiments, the conjugated protein of the present disclosure comprises a covalent linkage that joins an engineered boIFN/3 protein to a water-soluble polymer. In some embodiments, the covalent linkage comprises an amide, an ester, a thioester, a disulfide, an oxime, an imine, a pyrrolidine-2, 5-dione, a 1,2, 3 -triazole or a 1,4-dihydropyridazine. In some embodiments, the 1,2,3-triazole is fused to an 8-membered ring. In some embodiments, the 1,4- dihydropyridazine is fused to an 8-membered ring. In some embodiments, the covalent linkage that joins the engineered boIFNZ.3 protein to the water-soluble polymer comprises, or consists of, one of the following groups:
wherein: each Rb is independently H or unsubstituted alkyl; each Rc is unsubstituted alkyl; each Rf is independently H or unsubstituted alkyl; each s is independently 0, 1, 2, 3, 4, 5 or 6; each t is independently 0, 1, 2, 3, 4, 5 or 6; each + denotes connection to water-soluble polymer; and each wavy line denotes connection to the engineered boIFNZ.3 protein.
[00297] In some embodiments, the covalent linkage comprises an oxime. In some embodiments, the covalent linkage consists of an oxime. In some embodiments, the covalent linkage has the following structure:
wherein + denotes the connection to the water-soluble polymer; the wavyline denotes connection to the engineered boIFNZ.3 protein; and Rc is unsubstituted Ci-Ce alkyl. In some embodiments, Rc is methyl. Rc can be the methyl group of the non-natural amino acid pAF that is present in the engineered boIFNZ.3 protein amino acid sequence.
[00298] The present disclosure also provides a protein conjugate comprising a boIFNZ.3 protein or variant thereof, wherein the boIFNZ.3 protein or variant thereof comprises an unusual genetically-encoded amino acid that is not present in humans. In some embodiments, unusual genetically-encoded amino acid that is not present in humans is selenocysteine. In some embodiments, unusual genetically-encoded amino acid that is not present in humans is pyrrolysine. In some embodiments, the boIFNZ.3 protein or variant thereof comprising the unusual genetically encoded amino acid is conjugated to a water-soluble polymer via the unusual genetically encoded amino acid. In some embodiments, the boIFNZ.3 protein or variant thereof comprising the unusual genetically encoded amino acid conjugated to the water-soluble polymer via the unusual genetically encoded amino acid, does not contain a non-natural amino acid. In any of the foregoing
embodiments, wherein the boIFNZ.3 protein or variant thereof comprises an unusual genetically- encoded amino acid, the water-soluble polymer can be a PEG or a derivative thereof.
[00299] In some embodiments, a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein having an amino acid sequence disclosed in Table 1, wherein the amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 1, wherein each said amino acid sequence optionally further comprises an N-terminal methionine. In some embodiments, the engineered boIFNZ.3 protein amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein, wherein the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 1 wherein at least one amino acid of SEQ ID NO: 1 is replaced with a different amino acid. In some embodiments, the different amino acid is a non-natural amino acid. In some embodiments, the non-natural amino acid is pAF. In some embodiments, one amino acid of SEQ ID NO: 1 is replaced with one non-natural amino acid. In some embodiments, the one non-natural amino acid is pAF.
[00300] In some embodiments, a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein having an amino acid sequence that shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 1, optionally wherein the engineered boIFNz.3 protein amino acid sequence further comprises an N-terminal methionine. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, the engineered boIFNZ.3 protein amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with a sequence listed in Table 1, wherein X is para-acetyl -L-phenylalanine (pAF).
[00301] In some embodiments, a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein, wherein the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 1, wherein one or more amino acid of SEQ ID NO: 1 is replaced with a different amino acid. In some embodiments, the different amino acid is a non-natural amino acid. In some embodiments, the non-natural amino acid is pAF. In some embodiments, one amino acid of SEQ ID NO: 1 is replaced with one non-natural amino acid. In some embodiments, the one non-natural amino acid is pAF.
[00302] In some other embodiments, a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein having an amino acid sequence disclosed in Table 1, wherein the
amino acid sequence shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with (i) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; or (ii) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N- terminal methionine; and wherein the engineered boIFNz.3 protein amino acid sequence shares less than 100% identity with SEQ ID NO: 1. In some embodiments, each of SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13 has one or more non-natural amino acid incorporated.
[00303] In some other embodiments, a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein having an amino acid sequence that shares at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; wherein the engineered boIFNz.3 protein amino acid sequence shares less than 100% identity with SEQ ID NO: 1, and optionally wherein the engineered boIFNz.3 protein amino acid sequence further comprises an N-terminal methionine. In some embodiments, the engineered boIFNZ.3 protein has an amino acid sequence in which one or more non-natural amino acid is incorporated e.g., has the amino acid sequence of any of SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13 in which one or more non-natural amino acid is incorporated. In some embodiments, the one or more non-natural amino acid is pAF. In some embodiments, the engineered boIFNz.3 protein has the amino acid sequence of any of SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13 in which one non-natural amino acid incorporated. In some embodiments, the one non-natural amino acid is pAF.
[00304] In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 90% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24 further comprises an N-terminal methionine. In some other embodiments, a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine (pAF). In some
embodiments, the amino acid sequence shares at least 90% identity with a sequence listed in Table 1, wherein X is pAF. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 90% identity with SEQ ID NO: 18. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 90% identity with SEQ ID NO: 19.
[00305] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 95% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine. In some other embodiments, a conjugated protein of the present disclosure can comprise an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 95% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 95% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine (pAF). In some embodiments, the amino acid sequence shares at least 95% identity with a sequence listed in Table 1, wherein X is pAF. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 95% identity with SEQ ID NO:
18. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 95% identity with SEQ ID NO:
19.
[00306] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 96%, at least 97% or at least 98% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine.
[00307] In some other embodiments, a conjugated protein of the present disclosure can comprise an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid and having (i) an amino acid sequence that shares at least 96% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) an amino acid sequence that shares at least 96% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine. In some other embodiments, a conjugated protein of the present disclosure can comprise an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 96% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 96% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine (pAF). In some embodiments, the amino acid sequence shares at least 96% identity with a sequence listed in Table 1, wherein X is pAF. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 96% identity with SEQ ID NO:
18. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 96% identity with SEQ ID NO:
19.
[00308] In some other embodiments, a conjugated protein of the present disclosure can comprise an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid and having (i) an amino acid sequence that shares at least 97% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) an amino acid sequence that shares at least 97% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine. In some other embodiments, a conjugated protein of the present disclosure can comprise an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid and having (i) an amino acid sequence that shares at least 97% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) an amino acid sequence that shares at least 97% identity with SEQ ID NO: 14, 15, 16, 17, 18,
20. 21, 22, 23 or 24, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine
(pAF). In some embodiments, the amino acid sequence shares at least 97% identity with a sequence listed in Table 1, wherein X is pAF. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g. an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 97% identity with SEQ ID NO: 18. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 97% identity with SEQ ID NO: 19.
[00309] In some other embodiments, a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid and having (i) an amino acid sequence that shares at least 98% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) an amino acid sequence that shares at least 98% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine. In some other embodiments, a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid and having (i) an amino acid sequence that shares at least 98% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) an amino acid sequence that shares at least 98% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine (pAF). In some embodiments, the amino acid sequence shares at least 98% identity with a sequence listed in Table 1, wherein X is pAF. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 98% identity with SEQ ID NO: 18. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 98% identity with SEQ ID NO: 19.
[00310] In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with (i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine. In some other embodiments, a conjugated protein of the present disclosure can comprise an engineered boIFNz.3 protein having an amino
acid sequence comprising a non-natural amino acid and having (i) an amino acid sequence that shares at least 99% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or (ii) an amino acid sequence that shares at least 99% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine (pAF). In some embodiments, the amino acid sequence shares at least 99% identity with a sequence listed in Table 1, wherein X is pAF.
[00311] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 14. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 14, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00312] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 14, wherein the sequence optionally further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 14. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 14, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 14, and wherein the engineered boIFNz.3 protein optionally further comprises an N- terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 14. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 14, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
[00313] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 15. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 15, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00314] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 15, wherein the sequence optionally further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 15. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 15, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 15, and wherein the engineered boIFNz.3 protein optionally further comprises an N- terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 15. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 15, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
[00315] In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 16. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 16, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00316] In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 16, wherein the sequence optionally further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 16. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 16, and wherein the engineered boIFN/3 protein further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non -natural amino acid) that shares at least 99% identity with SEQ ID NO: 16, and wherein the engineered boIFNz.3 protein optionally further comprises an N- terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non -natural amino acid) that shares at least 99% identity with SEQ ID NO: 16. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 16, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
[00317] In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 17. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 17, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00318] In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 17, wherein the sequence optionally further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 17. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 17, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered
boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNZ.3 protein having an amino acid sequence comprising a non -natural amino acid) that shares at least 99% identity with SEQ ID NO: 17, and wherein the engineered boIFNX3 protein optionally further comprises an N- terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNZ.3 protein having an amino acid sequence comprising a non -natural amino acid) that shares at least 99% identity with SEQ ID NO: 17. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 17, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
[00319] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 18. In some embodiments, the non-natural amino acid is pAF.
[00320] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 18. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 18. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
[00321] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 19. In some embodiments, the non-natural amino acid is pAF.
[00322] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 19. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g. an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 19. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
[00323] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 20. In some embodiments, the conjugated
protein comprises an engineered boIFNX3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 20, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00324] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 20, wherein the sequence optionally further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 20. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 20, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 20, and wherein the engineered boIFNz.3 protein optionally further comprises an N- terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 20. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 20, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
[00325] In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 21. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 21, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00326] In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 21, wherein the sequence optionally further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that
shares at least 99% identity with SEQ ID NO: 21. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 21, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non -natural amino acid) that shares at least 99% identity with SEQ ID NO: 21, and wherein the engineered boIFNz.3 protein optionally further comprises an N- terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non -natural amino acid) that shares at least 99% identity with SEQ ID NO: 21. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 21, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
[00327] In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 22. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 22, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00328] In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 22, wherein the sequence optionally further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 22. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 22, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 22, and wherein the engineered boIFNZ.3 protein optionally further comprises an N- terminal methionine. In some embodiments, the conjugated protein comprises an engineered
boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNZ.3 protein having an amino acid sequence comprising a non -natural amino acid) that shares at least 99% identity with SEQ ID NO: 22. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO:
22, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
[00329] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 23. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 23, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00330] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 23, wherein the sequence optionally further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 23. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 23, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 23, and wherein the engineered boIFNZ.3 protein optionally further comprises an N- terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 23. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO:
23, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
[00331] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 24. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence comprising a non-natural amino acid, wherein the amino acid sequence shares at least 99% identity with SEQ ID NO: 24, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00332] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 24, wherein the sequence optionally further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 24. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence that shares at least 99% identity with SEQ ID NO: 24, and wherein the engineered boIFNZ.3 protein further comprises an N-terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 24, and wherein the engineered boIFNZ.3 protein optionally further comprises an N- terminal methionine. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 24. In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having an amino acid sequence (e.g., an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid) that shares at least 99% identity with SEQ ID NO: 24, and wherein the engineered boIFNz.3 protein further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine (pAF).
[00333] In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having the amino acid sequence of SEQ ID NO: 14 comprising a non-natural amino acid. In some embodiments, the non-natural amino acid is pAF.
[00334] In some embodiments, the conjugated protein comprises an engineered boIFNz.3 protein having the amino acid sequence of SEQ ID NO: 14 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00335] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 15 comprising a non-natural amino acid. In some embodiments, the non-natural amino acid is pAF.
[00336] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 15 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00337] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 16 comprising a non-natural amino acid. In some embodiments, the non-natural amino acid is pAF.
[00338] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 16 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00339] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 17 comprising a non-natural amino acid. In some embodiments, the non-natural amino acid is pAF.
[00340] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 17 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00341] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 18 comprising a non-natural amino acid. In some embodiments, the non-natural amino acid is pAF.
[00342] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having an amino acid sequence of SEQ ID NO: 19 comprising a non-natural amino acid. In some embodiments, the non-natural amino acid is pAF.
[00343] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 20 comprising a non-natural amino acid. In some embodiments, the non-natural amino acid is pAF.
[00344] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 20 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00345] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 21 comprising a non-natural amino acid. In some embodiments, the non-natural amino acid is pAF.
[00346] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 21 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00347] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 22 comprising a non-natural amino acid. In some embodiments, the non-natural amino acid is pAF.
[00348] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 22 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00349] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 23 comprising a non-natural amino acid. In some embodiments, the non-natural amino acid is pAF.
[00350] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 23 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00351] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 24 comprising a non-natural amino acid. In some embodiments, the non-natural amino acid is pAF.
[00352] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 24 comprising a non-natural amino acid, wherein the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00353] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 25 comprising a non-natural amino acid. In some embodiments, the non-natural amino acid is pAF.
[00354] In some embodiments, the conjugated protein comprises an engineered boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 25 comprising a non-natural amino acid, wherein
the amino acid sequence further comprises an N-terminal methionine. In some embodiments, the non-natural amino acid is pAF.
[00355] In all embodiments referred to above, the non-natural amino acid in the boIFNz.3 protein may be pAF.
[00356] In some more particular aspects, the present disclosure provides a conjugated protein comprising: an engineered boIFNz.3 protein having an amino acid sequence comprising a non- natural amino acid; and a water-soluble polymer; wherein: the water-soluble polymer is a polyethylene glycol (PEG) or a derivative thereof, which is conjugated to the engineered boIFNz.3 protein via a covalent linkage with the non-natural amino acid; and the engineered boIFNz.3 protein amino acid sequence shares at least 90% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 96% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 97% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 98% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO: 18. In some embodiments, the engineered boIFN 3 protein has the amino acid sequence of SEQ ID NO: 18. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine. In some embodiments, the engineered boIFNz.3 protein is an isolated protein. In some embodiments, the engineered boIFNz.3 protein is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA synthetase. Thus, in some embodiments, the non-natural amino acid is site-specifically incorporated and non-naturally encoded. In some embodiments, the PEG or the derivative thereof has a molecular weight of at least about 0.1 kDa and at most about 100 kDa. In some embodiments, the PEG or the derivative thereof has a molecular weight of at most about 50 kDa. In some embodiments, the PEG or the derivative thereof has a molecular weight within a range of about 10 kDa to about 50 kDa, about 20 kDa to about 40 kDa, or about 25 kDa to about 35 kDa. In some embodiments, the PEG or the derivative thereof has a molecular weight of about 30 kDa. In some embodiments, the PEG or the derivative thereof is a linear PEG or a linear PEG derivative. In some embodiments, the linear PEG derivative is a linear PEG comprising a reactive moiety that is capable of reacting with a non-natural amino acid. In some embodiments, the linear PEG derivative is a linear PEG comprising a terminal aminooxy group. In some embodiments, the linear PEG derivative is a linear PEG comprising a terminal alkyne. In some embodiments, the linear PEG derivative is a linear PEG comprising a terminal azido group.
In some embodiments, the PEG is a PEG derivative, such as a linear PEG derivative, wherein, prior to the conjugation with the engineered boIFNz.3 protein, the PEG derivative comprises an aminooxy group. In some embodiments, the covalent linkage conjugating the engineered boIFNz.3 protein with the PEG derivative comprises, or consists of, an oxime. A conjugated boIFNz.3 protein of the present disclosure has an extended elimination half-life in vivo, as further disclosed herein.
[00357] In some other more particular aspects, the present disclosure provides a conjugated protein comprising: an engineered boIFNz.3 protein having an amino acid sequence comprising a non-natural amino acid; and a water-soluble polymer; wherein: the water-soluble polymer is a polyethylene glycol (PEG) or a derivative thereof, which is conjugated to the engineered boIFNz.3 protein via a covalent linkage with the non-natural amino acid; and the engineered boIFNz.3 protein amino acid sequence shares at least 90% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 96% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 97% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 98% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO: 19. In some embodiments, the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 19. In some embodiments, the non-natural amino acid is para-acetyl-L-phenylalanine. In some embodiments, the engineered boIFNz.3 protein is an isolated protein. In some embodiments, the engineered boIFNz.3 protein is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA synthetase. Thus, in some embodiments, the non-natural amino acid is site-specifically incorporated and non-naturally encoded. In some embodiments, the PEG or the derivative thereof has a molecular weight of at least about 0.1 kDa and at most about 100 kDa. In some embodiments, the PEG or the derivative thereof has a molecular weight of at most about 50 kDa. In some embodiments, the PEG or the derivative thereof has a molecular weight within a range of about 10 kDa to about 50 kDa, about 20 kDa to about 40 kDa, or about 25 kDa to about 35 kDa. In some embodiments, the PEG or the derivative thereof has a molecular weight of about 30 kDa. In some embodiments, the PEG or the derivative thereof is a linear PEG or a linear PEG derivative. In some embodiments, the linear PEG derivative is a linear PEG comprising a reactive moiety that is capable of reacting with a non-natural amino acid. In some embodiments, the linear PEG derivative is a linear PEG comprising a terminal aminooxy group.
In some embodiments, the linear PEG derivative is a linear PEG comprising a terminal alkyne. In some embodiments, the linear PEG derivative is a linear PEG comprising a terminal azido group. In some embodiments, the PEG is a PEG derivative, such as a linear PEG derivative, wherein, prior to the conjugation with the engineered boIFNZ.3 protein, the PEG derivative comprises an aminooxy group. In some embodiments, the covalent linkage conjugating the engineered boIFN/3 protein with the PEG derivative comprises, or consists of, an oxime. A conjugated boIFNz.3 protein of the present disclosure has an extended elimination half-life in vivo, as further disclosed herein.
[00358] In a preferred embodiment, the present invention provides a conjugated protein comprising: an engineered boIFNz.3 protein having an amino acid sequence comprising a nonnatural amino acid, wherein the non-natural amino acid is para-acetyl-L-phenyalanine, wherein the engineered boIFNz.3 protein has an amino acid sequence of SEQ ID NO: 18; and a water soluble polymer, wherein the water soluble polymer is PEG or a derivative thereof, wherein the PEG or derivative thereof has a molecular weight of about 30kDa, optionally wherein the PEG or derivative thereof is a linear PEG or a linear PEG derivative; wherein the water-soluble polymer is conjugated to the engineered boIFNz.3 protein via the non-natural amino acid, optionally via a covalent linkage, optionally wherein the covalent linkage comprises, or consists of, an oxime.
[00359] In another preferred embodiment, the present invention provides a conjugated protein comprising: an engineered boIFNz.3 protein having an amino acid sequence comprising a non- natural amino acid, wherein the non-natural amino acid is para-acetyl-L-phenyalanine, wherein the engineered boIFNz.3 protein has an amino acid sequence of SEQ ID NO: 19; and a water soluble polymer, wherein the water soluble polymer is PEG or a derivative thereof, wherein the PEG or derivative thereof has a molecular weight of about 30kDa, optionally wherein the PEG or derivative thereof is a linear PEG or a linear PEG derivative; wherein the water-soluble polymer is conjugated to the engineered boIFNz.3 protein via the non-natural amino acid, optionally via a covalent linkage, optionally wherein the covalent linkage comprises, or consists of, an oxime.
[00360] Modulation of boIFN 3 protein elimination half-life
[00361] Unconjugated IFNs are generally short-lived species in circulation, with typical half-lives occurring within minutes, and less than one hour, as understood by a person skilled in the art. For example, Bansal et al. have shown that IFNy plasma concentrations in mice were reduced by over 90% within about 30 minutes after administration (Bansal R. et al. (2011) J. Controlled Release 154(3):233-240; see, e.g., Bansal at Figure 4A). The rapid clearance of unconjugated boIFNz.3 protein has hindered efforts to induce the protective immune response that is needed to control devastating viral infections in ruminant mammals. The present disclosure provides conjugated
proteins and uses thereof with properties that can overcome these limitations. The improved pharmacokinetic profiles of the conjugated boIFNZ.3 proteins of the present disclosure support their use in providing treated subjects with protection against infection by pathogenic viruses, such as those associated with BRD and FMD. The protection can begin prior to the onset of an adaptive immune response that occurs after the administration of a vaccine. An improved pharmacokinetic profile can further enable an improved dosing regimen or avoid toxic effects.
[00362] As is understood by a person of ordinary skill in the art, the elimination half-life of a biotherapeutic protein, whether conjugated or unconjugated, can refer to the time it takes for the concentration of the protein in the blood, plasma, serum or body of a subject to be reduced by 50%. Thus, after one half-life, the concentration of the protein in the blood, plasma, serum or body of the subject, respectively, will be half of the starting concentration. In some embodiments, the elimination half-life is a blood elimination half-life. In some other embodiments, the elimination half-life is a plasma elimination half-life. In some other embodiments, the elimination half-life is a serum elimination half-life. In yet some other embodiments, the elimination half-life is a whole body elimination half-life. Methods for evaluating pharmacokinetics and protein elimination halflife are known in the art (see, e.g., the Examples herein; Rosenbaum S.E. (Ed), (2016) Basic Pharmacokinetics and Pharmacodynamics: An Integrated Textbook and Computer Simulations, 2nd Edition (ISBN: 978-1-119-14315-4); Mansoor A, Mahabadi N. Volume of Distribution. [Updated 2022 Jul 25], In: StatPearls [Internet], Treasure Island (FL): StatPearls Publishing; 2023, available from the worldwide web at ncbi.nlm.nih.gov/books/NBK545280/; and Bansal (2011) (TtZ.). By way of example, elimination half-life in plasma can be measured by taking blood samples at various time points after administration of the protein (whether conjugated or unconjugated) and determining the concentration of that protein in each sample. Correlation of the plasma concentration with time allows calculation of the elimination half-life in plasma. Elimination halflife for a conjugated protein, an unconjugated protein, or both, can be evaluated for an individual subject, between individual subjects, within a group of subjects, between groups of subjects, and/or between an individual subject and a group of subjects, depending upon study design and other factors, as understood by persons of ordinary skill in the art.
[00363] In some embodiments, a conjugated boIFNz.3 protein of the present disclosure can have an elimination half-life that is greater than the elimination half-life of the unconjugated engineered boIFNZ.3 protein (e.g., the corresponding unconjugated boIFNZ.3 protein, or of a related unconjugated protein, such as another unconjugated IFN protein (e.g., an unconjugated IFNy or IFNZ.). In some embodiments, the related unconjugated protein is wild-type boIFNz.3 protein having the amino acid sequence of SEQ ID NO: 1. In some embodiments, the conjugated boIFNz.3
protein has an elimination half-life that is at least about 5% greater than the elimination half-life of the unconjugated boIFNz.3 protein (e.g., the corresponding unconjugated boIFNz.3 protein), or of the related unconjugated protein. In some embodiments, the conjugated boIFNz.3 protein can have an elimination half-life that is at least about 10%, at least about 20%, at least about 30%, at least about 40%, at least about 50%, at least about 60%, at least about 70%, at least about 80%, at least about 90% or at least about 100% greater than the elimination half-life of the unconjugated boIFNz.3 protein (e.g., the corresponding unconjugated boIFNZ.3 protein), or of the related unconjugated protein. In some embodiments, the conjugated boIFNz.3 protein can have an elimination half-life that is at least about 5-fold, about 10-fold (1000%), about 20-fold, about 30- fold or about 40-fold greater than the elimination half-life of the unconjugated boIFNz.3 protein (e.g., the corresponding unconjugated boIFNZ.3 protein), or of the related unconjugated protein. [00364] In some embodiments, the elimination half life can be in a mammal (e.g., as determined in a mammal). Examples of suitable mammals include rodents (e.g., a rat, such as a Sprague- Dawley rat), ruminant mammals, cattle (e.g., bovine, bovine calf, such as Holstein Friesian Calf). In some embodiments, the elimination half life is a terminal half life.
[00365] In some embodiments, the elimination half-life can be in a Sprague-Dawley rat, e.g., after subcutaneous or another form of administration to the Sprague-Dawley rat, as disclosed in the Examples herein (e.g., as determined in the Sprague-Dawley rat). In some other embodiments, the elimination half-life can be in a Holstein-Friesian Calf, e.g., after subcutaneous or another form of administration to the Holstein-Friesian Calf, also disclosed in the Examples herein (e.g., as determined in the Frieisan Calf). In some embodiments, the conjugated boIFNz.3 protein can have an elimination half-life in a Holstein-Friesian Calf that is at least about 50-fold greater than the elimination half-life of the corresponding unconjugated boIFNZ.3 protein, or of a related unconjugated protein. In some embodiments, the conjugated boIFNz.3 protein can have an elimination half-life in a Holstein-Friesian Calf that is at least about 60-fold greater than the elimination half-life of the corresponding unconjugated boIFNZ.3 protein, or of a related unconjugated protein. In some embodiments, the conjugated boIFNz.3 protein can have an elimination half-life in a Holstein-Friesian Calf that is at least about 65-fold greater than the elimination half-life of the corresponding unconjugated boIFNZ.3 protein, or of a related unconjugated protein. The corresponding unconjugated boIFNZ.3 protein may in some embodiments differ from the relevant conjugated boIFNz.3 protein in that the unconjugated version does not contain a water soluble polmer as defined herein (and optionally differs from the unconjugated version only in this respect). The corresponding unconjugated version may therefore have the same amino acid sequence as the relevant conjugated boIFNz.3 protein (optionally
including any non-natural amino acids). In some embodiments, the elimination half-life is a blood elimination half-life. In some embodiments, the blood elimination half life is a terminal blood half life. In some embodiments, the elimination half-life is a plasma elimination half-life. In some embodiments, the plasma elimination half life is a terminal plasma half life. In some embodiments, the elimination half-life is a serum elimination half-life. In some embodiments, the serum elimination half life is a terminal serum half life. In some embodiments, the elimination half-life is a half-life of elimination from the body (whole body elimination half-life).
[00366] In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 10 hours in a rat after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein to the rat. In some embodiments, the single subcutaneous dose is a dose within a range of about 0.1 mg/kg to about 10 mg/kg of the body weight of the rat. In some embodiments, the half life is observed in a Sprague-Dawley rat. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 10 hours in a Sprague-Dawley rat after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein of about 0.25 mg/kg (e.g., 0.25 mg/kg) to the Sprague-Dawley rat. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 15 hours in a Sprague-Dawley rat after administration of a single subcutaneous dose of the conjugated boIFNZ.3 protein of about 0.25 mg/kg (e.g., 0.25 mg/kg). In some embodiments, the conjugated boIFNZ.3 protein can have a plasma elimination half-life within a range of about 15 hours to about 30 hours in a Sprague-Dawley rat after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein of about 0.25 mg/kg (e.g., 0.25 mg/kg). In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of about 16 hours, about 17 hours, about 18 hours, about 19 hours or about 20 hours in a Sprague-Dawley rat after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein of about 0.25 mg/kg (e.g., 0.25 mg/kg).
[00367] In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 10 hours in cattle after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein to the cattle. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 20 hours in cattle after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein to the cattle. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 30 hours in cattle after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein to the cattle. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 40 hours in cattle after administration of a single
subcutaneous dose of the conjugated boIFNz.3 protein to the cattle. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 50 hours in cattle after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein to the cattle. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination halflife of at least about 60 hours in cattle after administration of a single subcutaneous dose of the conjugated boIFNz.3 protein to the cattle. In some embodiments, the single subcutaneous dose is within a range of about 50 pg/kg to about 500 pg/kg of the body weight of the cattle. In some embodiments, the single subcutaneous dose is within a range of about 75 pg/kg to about 150 pg/kg of the body weight of the cattle. In some embodiments, the single subcutaneous dose is about 75 pg/kg of the body weight of the cattle. In some embodiments, the single subcutaneous dose is about 150 pg/kg of the body weight of the cattle. In some embodiments, the single subcutaneous dose is within a range of about 100 pg/kg to about 200 pg/kg of the body weight of the cattle. In some embodiments, the half life is observed in a Holstein-F resian calf. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 10 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 75 pg/kg to the Holstein-Fresian calf. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 20 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 30 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 40 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 50 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination halflife within a range of about 50 hours to about 75 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 60 hours in a Holstein- Fresian calf after administration of a single subcutaneous dose of 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 65 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of about 61 hours, about 62 hours, about 63 hours, about 64 hours, about 65 hours, about 66 hours, about 67 hours, about 68 hours or about 69 hours in a Holstein-Fresian calf after administration of a single
subcutaneous dose of 75 pg/kg. In some embodiments, the conjugated boIFNX3 protein can have a plasma elimination half-life of at least about 70 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 75 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 10 hours in a Holstein- Fresian calf after administration of a single subcutaneous dose of about 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 20 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 30 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 40 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 50 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life within a range of about 50 hours to about 75 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 60 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 65 hours in a Holstein- Fresian calf after administration of a single subcutaneous dose of 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of about 61 hours, about 62 hours, about 63 hours, about 64 hours, about 65 hours, about 66 hours, about 67 hours, about 68 hours or about 69 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 150 pg/kg. In some embodiments, the conjugated boIFNz.3 protein can have a plasma elimination half-life of at least about 70 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 150 pg/kg.
[00368] Pharmaceutical compositions and administration of engineered boIFN 3 protein and conjugates thereof
[00369] The engineered boIFNz.3 proteins and conjugated boIFNz.3 proteins of the disclosure can be employed for therapeutic uses, including but not limited to, in combination with a suitable pharmaceutical carrier. Such compositions can comprise a therapeutically effective amount of the engineered boIFNz.3 protein, or the conjugated boIFNz.3 protein, and a pharmaceutically acceptable carrier or excipient. Such a carrier or excipient includes, but is not limited to, saline,
buffered saline, dextrose, water, glycerol, ethanol, and/or combinations thereof, or any other suitable carrier or excipient, including those further disclosed herein. The formulation is made to suit the mode of administration. In general, methods of administering biotherapeutic proteins are known to those of ordinary skill in the art and can be applied to administration of the proteins and conjugated proteins of the present disclosure. Compositions may be in a water-soluble form, such as being present as pharmaceutically acceptable salts, which is meant to include both acid and base addition salts. Pharmaceutically acceptable carriers are determined in part by the particular composition being administered, as well as by the particular method used to administer the composition. Accordingly, there is a wide variety of suitable formulations of pharmaceutical compositions of the present disclosure. The formulations can be presented in unit-dose or multidose sealed containers, such as ampules and vials.
[00370] Formulations suitable for administration include aqueous and non-aqueous solutions, isotonic sterile solutions, which can contain antioxidants, buffers, bacteriostats, and solutes that render the formulation isotonic, and aqueous and non-aqueous sterile suspensions that can include suspending agents, solubilizers, thickening agents, stabilizers, and preservatives. Solutions and suspensions can be prepared from sterile powders, granules, and tablets.
[00371] The pharmaceutical compositions and formulations of the disclosure may comprise a pharmaceutically acceptable carrier, excipient or stabilizer. Pharmaceutically acceptable carriers are determined in part by the particular composition being administered, as well as by the particular method used to administer the composition. Accordingly, there is a wide variety of suitable formulations of pharmaceutical compositions (including optional pharmaceutically acceptable carriers, excipients, or stabilizers) of the present disclosure (see, e.g., Remington ’s Pharmaceutical Sciences, 17th ed. 1985). Suitable carriers include but are not limited to, buffers containing succinate, phosphate, borate, HEPES, citrate, histidine, imidazole, acetate, bicarbonate, and other organic acids; antioxidants including but not limited to, ascorbic acid; low molecular weight polypeptides including but not limited to those less than about 10 residues; proteins, including but not limited to, serum albumin, gelatin, or immunoglobulins; hydrophilic polymers including but not limited to, polyvinylpyrrolidone; amino acids including but not limited to, glycine, glutamine, asparagine, arginine, histidine or histidine derivatives, methionine, glutamate, or lysine; monosaccharides, disaccharides, and other carbohydrates, including but not limited to, trehalose, sucrose, glucose, mannose, or dextrins; chelating agents including but not limited to, EDTA and edentate disodium; divalent metal ions including but not limited to, zinc, cobalt, or copper; sugar alcohols including but not limited to, mannitol or sorbitol; salt-forming counter ions including but not limited to, sodium and sodium chloride; fillers such as microcrystalline cellulose, lactose, corn
and other starches; binding agents; sweeteners and other flavoring agents; coloring agents; and/or nonionic surfactants including but not limited to Tween™ (including but not limited to, Tween 80 (polysorbate 80) and Tween 20 (polysorbate 20), Pluronics™ and other pluronic acids, including but not limited to, pluronic acid F68 (poloxamer 188), or PEG. Suitable surfactants include for example but are not limited to polyethers based upon poly(ethylene oxide)-poly(propylene oxide)- poly(ethylene oxide), i.e., (PEO-PPO-PEO), or polypropylene oxide)-poly(ethylene oxide)- poly(propylene oxide), i.e., (PPO-PEO-PPO), or a combination thereof. PEO-PPO-PEO and PPO- PEO-PPO are commercially available under the trade names Pluronics™, R-Pluronics™, Tetronics™ and R-Tetronics™ (BASF Wyandotte Corp., Wyandotte, Mich.) and are further described in U.S. Pat. No. 4,820,352 incorporated herein in its entirety by reference. Other ethylene/polypropylene block polymers may be suitable surfactants. A surfactant or a combination of surfactants may be used to stabilize PEGylated boIFNz.3 against one or more stresses including but not limited to stress that results from agitation. Some of the above may be referred to as “bulking agents.” Some may also be referred to as “tonicity modifiers.” Antimicrobial preservatives may also be applied for product stability and antimicrobial effectiveness; suitable preservatives include but are not limited to, benzyl alcohol, benzalkonium chloride, metacresol, methyl/propyl parabene, cresol, and phenol, or a combination thereof. U.S. Patent No. 7,144,574, which is incorporated by reference herein, describe additional materials that may be suitable in pharmaceutical compositions and formulations of the invention and other delivery preparations.
[00372] Therapeutic compositions comprising one or more protein or conjugated protein of the disclosure can be tested in one or more appropriate in vitro and/or in vivo models of disease, to confirm efficacy, tissue metabolism, and to estimate dosages, according to methods known to those of ordinary skill in the art, i.e., in a relevant assay.
[00373] Administration is by any of the routes normally used for introducing a molecule into ultimate contact with blood or tissue cells. The proteins or conjugated proteins of the disclosure are administered in any suitable manner. The proteins or conjugated proteins of the disclosure can be administered in any suitable manner with one or more pharmaceutically acceptable carriers or excipients. Suitable methods of administering such conjugated proteins in the context of the present disclosure to a subject are available, and, although more than one route can be used to administer a particular composition, a particular route can often provide a more immediate and more effective action or reaction than another route. Thus, the proteins or conjugated proteins of the disclosure may be administered by any conventional route suitable for proteins or peptides, including, but not limited to parenterally, e.g., injections including, but not limited to, subcutaneously, intramuscularly or intravenously or any other form of injections or infusions.
Protein and conjugated protein compositions can be administered by a number of routes including, but not limited to oral, intravenous, intraperitoneal, intramuscular, transdermal, subcutaneous, topical, sublingual, or rectal means. In some embodiments, the boIFNZ.3 protein or variant thereof, or pharmaceutical composition comprising the boIFNZ.3 protein or variant thereof, is administered subcutaneously. In some embodiments, the conjugated boIFNZ.3 protein, or pharmaceutical composition comprising the conjugated boIFNZ.3 protein, is administered subcutaneously. In some embodiments, the boIFNZ.3 protein or variant thereof, or pharmaceutical composition comprising the boIFNZ.3 protein or variant thereof, is administered intramuscularly. In some embodiments, the conjugated boIFNZ.3 protein, or pharmaceutical composition comprising the conjugated boIFNZ.3 protein, is administered intramuscularly. Compositions comprising proteins or conjugated proteins can also be administered via liposomes. Such administration routes and appropriate formulations are generally known to those of skill in the art. The protein or conjugated protein may be used alone or in combination with other suitable components such as a pharmaceutical carrier. The conjugated protein may be used in combination with other agents or therapeutics. In some embodiments, the conjugated protein is used in combination with a vaccine. In some embodiments, the conjugated protein is used in combination with an antibiotic.
[00374] The dose administered to a subject, in the context of the present disclosure, is sufficient to have a beneficial therapeutic response in the subject over time, or other appropriate activity, depending on the application. The dose is determined by the efficacy of the particular protein or conjugated protein, or formulation containing the protein or conjugated protein, and the activity, stability, or plasma or serum or plasma half-life of the protein or conjugated protein employed, and the condition of the subject, as well as the body weight or surface area of the subject to be treated. The size of the dose is also determined by the existence, nature, and extent of any adverse side-effects that accompany the administration of a particular conjugated protein, formulation, or the like in a particular subject. Administration can be accomplished via single or divided doses.
[00375] The present disclosure also provides a pharmaceutical composition comprising a protein or conjugated protein of the present disclosure and an antibiotic for the treatment of a condition in a mammal, such as bovine respiratory disease. In some embodiments, the antibiotic is a penicillin, a cephalosporin, a macrolide, a tetracycline or a fluoroquinolone antibiotic; or a combination thereof. In some embodiments, the antibiotic is a cephalosporin antibiotic. In some embodiments, the antibiotic is a macrolide antibiotic. Non-limiting examples of penicillin, cephalosporin, macrolide and tetracycline antibiotics that can be used in combination with a protein or conjugated protein of the present disclosure, or that can be formulated in combination with a protein or conjugated protein of the present disclosure, are disclosed herein.
[00376] The present disclosure also provides a pharmaceutical composition comprising a protein or conjugated protein of the present disclosure and a vaccine for the treatment of a condition in a mammal, such as a disease or condition associated with BRDV or FMDV.
[00377] Therapeutic uses of engineered boIFN 3 proteins and conjugates thereof
[00378] The engineered boIFNz.3 proteins, conjugated boIFNz.3 proteins, and compositions containing the engineered boIFNz.3 proteins or conjugated boIFNz.3 proteins of the present disclosure, are useful for prophylactic treatment of a viral-associated disease or condition in a mammal, or more particularly, a ruminant mammal. The disclosure includes a method of treating a ruminant mammal that is at risk for, is having, or has had, a viral or a virus-associated disease or condition. In some embodiments, the disease or condition is foot and mouth disease (FMD). In some embodiments, the disease or condition is bovine respiratory disease (BRD).
[00379] Thus, in some embodiments, the present disclosure provides a method of treating a disease or condition in a ruminant mammal. The method of treatment can include providing protection against the disease or condition. In some embodiments, the method of treatment comprises administering a therapeutically effective amount of a conjugated boIFN/3 protein, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein, to a ruminant mammal. In some embodiments, the ruminant mammal is bovine. In some embodiments, the ruminant mammal is cattle. In some embodiments, the ruminant mammal is a bovine calf. In some embodiments, the conjugated boIFNz.3 protein comprises an engineered boIFNz.3 protein having an amino acid sequence of SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24, or any other boIFNz.3 protein, comprising one or more non-natural amino acids. In some embodiments, the engineered boIFNz.3 protein is conjugated to a water-soluble polymer via the non-natural amino acid. In some embodiments, the water-soluble polymer is a polyalkylene glycol or a derivative thereof. In some embodiments, the water-soluble polymer is a PEG or a derivative thereof. In some embodiments, the PEG or derivative thereof has a molecular weight of about 30kDa. In some embodiments, the one or more non-natural amino acids is one non-natural amino acid. In some embodiments, the non-natural amino acid is pAF. In some embodiments, the conjugated boIFNz.3 protein comprises an engineered boIFNZ.3 protein having an amino acid sequence of SEQ ID NO: 18. In some embodiments, the conjugated boIFNz.3 protein comprises an engineered boIFNz.3 protein having an amino acid sequence of SEQ ID NO: 19. In some embodiments, the disease or condition is foot and mouth disease (FMD). In some embodiments, the disease or condition is bovine respiratory disease (BRD). Suitable conjugates and engineered boIFNz.3 proteins are as disclosed elsewhere herein.
[00380] In some embodiments, the present disclosure provides a method of treating a disease or condition in a ruminant mammal, wherein the method comprises administering a vaccine to the ruminant mammal, and administering a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein. In some embodiments, the present disclosure provides a method of treating a disease or condition in a ruminant mammal, wherein the method comprises administering the combination of a therapeutically effective amount of conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein, and a vaccine to the ruminant mammal.
[00381] A therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure can be administered as a single or divided dose to a subject, such as a ruminant mammal. In some embodiments, a conjugated boIFNz.3 protein dose is at least about 1 pg/kg of the subject. In some embodiments, the dose is at least about 5 pg/kg of the subject. In some embodiments, the dose is at least about 10 pg/kg of the subject. In some embodiments, the dose is at least about 25 pg/kg of the subject. In some embodiments, the dose is at least about 50 pg/kg of the subject. In some embodiments, the dose is at least about 75 pg/kg of the subject. In some embodiments, the dose is at least about 100 pg/kg of the subject. In some embodiments, the dose is at most about 1,000 pg/kg of the subject. In some embodiments, the dose is at most about 750 pg/kg of the subject. In some embodiments, the dose is at most about 500 pg/kg of the subject. In some embodiments, the dose is at most about 400 pg/kg of the subject. In some embodiments, the dose is at most about 300 pg/kg of the subject. In some embodiments, the dose is at most about 250 pg/kg of the subject. In some embodiments, the dose is at most about 200 pg/kg of the subject. In some embodiments, the dose is at most about 150 pg/kg of the subject. In some embodiments, the dose is within a range of about 5 pg/kg to about 1000 pg/kg of the subject. In some embodiments, the dose is within a range of about 5 pg/kg to about 750 pg/kg of the subject. In some embodiments, the dose is within a range of about 5 pg/kg to about 500 pg/kg of the subject. In some embodiments, the dose is within a range of about 5 pg/kg to about 400 pg/kg of the subject. In some embodiments, the dose is within a range of about 5 pg/kg to about 300 pg/kg of the subject. In some embodiments, the dose is within a range of about 5 pg/kg to about 250 pg/kg of the subject. In some embodiments, the dose is within a range of about 5 pg/kg to about 200 pg/kg of the subject. In some embodiments, the dose is within a range of about 5 pg/kg to about 150 pg/kg of the subject. In some embodiments, the dose is within a range of about 10 pg/kg to about 1000 pg/kg of the subject. In some embodiments, the dose is within a range of about 10 pg/kg to
about 750 gg/kg of the subject. In some embodiments, the dose is within a range of about 10 gg/kg to about 500 gg/kg of the subject. In some embodiments, the dose is within a range of about 10 gg/kg to about 400 gg/kg of the subject. In some embodiments, the dose is within a range of about 10 gg/kg to about 300 gg/kg of the subject. In some embodiments, the dose is within a range of about 10 gg/kg to about 250 gg/kg of the subject. In some embodiments, the dose is within a range of about 25 gg/kg to about 1000 gg/kg of the subject. In some embodiments, the dose is within a range of about 25 gg/kg to about 750 gg/kg of the subject. In some embodiments, the dose is within a range of about 25 gg/kg to about 500 gg/kg of the subject. In some embodiments, the dose is within a range of about 25 gg/kg to about 400 gg/kg of the subject. In some embodiments, the dose is within a range of about 25 gg/kg to about 300 gg/kg of the subject. In some embodiments, the dose is within a range of about 25 gg/kg to about 250 gg/kg of the subject. In some embodiments, the dose is within a range of about 25 gg/kg to about 200 gg/kg of the subject. In some embodiments, the dose is within a range of about 25 gg/kg to about 150 gg/kg of the subject. In some embodiments, the dose is within a range of about 50 gg/kg to about 1000 gg/kg of the subject. In some embodiments, the dose is within a range of about 50 gg/kg to about 750 gg/kg of the subject. In some embodiments, the dose is within a range of about 50 gg/kg to about 500 gg/kg of the subject. In some embodiments, the dose is within a range of about 50 gg/kg to about 400 gg/kg of the subject. In some embodiments, the dose is within a range of about 50 gg/kg to about 300 gg/kg of the subject. In some embodiments, the dose is within a range of about 50 gg/kg to about 250 gg/kg of the subject. In some embodiments, the dose is within a range of about 50 gg/kg to about 200 gg/kg of the subject. In some embodiments, the dose is within a range of about 50 gg/kg to about 150 gg/kg of the subject. In some embodiments, the dose is within a range of about 75 gg/kg to about 1000 gg/kg of the subject. In some embodiments, the dose is within a range of about 75 gg/kg to about 750 gg/kg of the subject. In some embodiments, the dose is within a range of about 75 gg/kg to about 500 gg/kg of the subject. In some embodiments, the dose is within a range of about 75 gg/kg to about 400 gg/kg of the subject. In some embodiments, the dose is within a range of about 75 gg/kg to about 300 gg/kg of the subject. In some embodiments, the dose is within a range of about 75 gg/kg to about 250 gg/kg of the subject. In some embodiments, the dose is within a range of about 75 gg/kg to about 200 gg/kg of the subject. In some embodiments, the dose is within a range of about 75 gg/kg to about 150 gg/kg of the subject. In some embodiments, the dose is within a range of about 50 gg/kg to about 500 gg/kg of the subject. In some embodiments, the dose is within a range of about 75 gg/kg to about 250 gg/kg of the subject. In some embodiments, the dose is within a range of about 100 gg/kg to about 200 gg/kg of the subject. In some embodiments, the dose is about 50 gg/kg of the
subject. In some embodiments, the dose is about 55 gg/kg of the subject. In some embodiments, the dose is about 60 gg/kg of the subject. In some embodiments, the dose is about 65 gg/kg of the subject. In some embodiments, the dose is about 70 gg/kg of the subject. In some embodiments, the dose is about 75 gg/kg of the subject. In some embodiments, the dose is about 80 gg/kg of the subject. In some embodiments, the dose is about 85 gg/kg of the subject. In some embodiments, the dose is about 90 gg/kg of the subject. In some embodiments, the dose is about 95 gg/kg of the subject. In some embodiments, the dose is about 100 gg/kg of the subject. In some embodiments, the dose is about 105 gg/kg of the subject. In some embodiments, the dose is about 110 gg/kg of the subject. In some embodiments, the dose is about 115 gg/kg of the subject. In some embodiments, the dose is about 120 gg/kg of the subject. In some embodiments, the dose is about 125 gg/kg of the subject. In some embodiments, the dose is about 130 gg/kg of the subject. In some embodiments, the dose is about 135 gg/kg of the subject. In some embodiments, the dose is about 140 gg/kg of the subject. In some embodiments, the dose is about 145 gg/kg of the subject. In some embodiments, the dose is about 150 gg/kg of the subject. In some embodiments, the dose is about 155 gg/kg of the subject. In some embodiments, the dose is about 160 gg/kg of the subject. In some embodiments, the dose is about 165 gg/kg of the subject. In some embodiments, the dose is about 170 gg/kg of the subject. In some embodiments, the dose is about 175 gg/kg of the subject. In some embodiments, the dose is about 180 gg/kg of the subject. In some embodiments, the dose is about 185 gg/kg of the subject. In some embodiments, the dose is about 190 gg/kg of the subject. In some embodiments, the dose is about 195 gg/kg of the subject. In some embodiments, the dose is about 200 gg/kg of the subject. In some embodiments, the doses is administered subcutaneously. In some other embodiments, the dose is administered intramuscularly.
[00382] As disclosed herein, FMDV is a highly contagious pathogen among cloven-hoofed animals, including cattle. Current vaccines against FMDV do not induce protective antibody titers until 5 to 7 days post-vaccination, rendering vaccinated animals susceptible to infection and transmission (Diaz-San Segundo F. et al. (2011) Virology 413:283-292). Thus, there remains a need for a treatment that induces early protection against FMD (e.g., protection that begins prior to the induction of protective antibody titers in response to a vaccine), thereby closing the susceptibility gap between vaccination and the onset of adaptive immunity.
[00383] In some embodiments, the present disclosure provides a method of treating FMD in a ruminant mammal, wherein the method comprises administering a therapeutically effective amount of conjugated boIFNZ.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNZ.3 protein, to the ruminant mammal. In some embodiments, the ruminant mammal is bovine. In some embodiments, the
ruminant mammal is a cattle. In some embodiments, the ruminant mammal is a bovine calf. In some embodiments, the method of treatment can provide protection against FMD, provide protection against FMD symptoms, provide protection against FMDV infection, reduce a rate of infection by FMDV, reduce a degree of infection by FMDV, inhibit FMDV replication, delay onset of FMD, reduce severity of FMD, reduce severity of FMD symptoms, prevent FMD and/or prevent infection by FMDV. In some embodiments, the method of treatment can reduce the rate of transmission, or prevent the transmission, of FMDV from one ruminant mammal to another ruminant mammal or mammals. In some embodiments, the method further comprises administering a FMDV vaccine to the ruminant mammal.
[00384] In some embodiments, the method of treating FMD provides the ruminant mammal with early protection against FMD, i.e., protection against FMDV infection throughout an early protection period. In some embodiments, the early protection period begins within about 1 day from the time of administration of the conjugated boIFN/3 protein to the mammal and can last for at least about 5 to 7 days. In some embodiments, the early protection period begins prior to the induction of protective antibody titers in the mammal in response to a vaccine. In some embodiments, the early protection period is about 1 day, about 2 days, about 3 days, about 4 days, about 5 days, about 6 days or about 7 days or more. In some embodiments, the early protection period is a period of about 1 day. In some embodiments, the early protection period is a period of about 2 days. In some embodiments, the early protection period is a period of about 3 days. In some embodiments, the early protection period is a period of about 4 days. In some embodiments, the early protection period is a period of about 5 days. In some embodiments, the early protection period is a period of about 6 days. In some embodiments, the early protection period is a period of about 7 days. In some embodiments, the early protection period is about 1 day to about 5 days. In some embodiments, the early protection period is about 1 day to about 7 days. In some embodiments, the method of treating FMD further provides the ruminant mammal with long-term protection against FMD, including long-term protection from FMDV infection, i.e., protection against FMDV throughout a long-term protection period. In some embodiments, the long-term protection period is at least about 2 weeks. In some embodiments, the long-term protection period is at least about 3 weeks. In some embodiments, the long-term protection period is at least about 4 weeks. In some embodiments, the long-term protection period is at least about a month. In some embodiments, the long-term protection period is at least about 2 months. In some embodiments, the long-term protection period is at least about 3 months. In some embodiments, the long-term protection period is about 100 days, or is about 120 days, or more. An early and/or long-term protection against FMD can be characterized, for example, by an absence of viremia, including a
negative result in a test for the virus using methods known to a person of skill in the art. In some embodiments, the protection against FMD is characterized as an absence of a clinical FMD symptom. In some embodiments, the absence of a clinical FMD symptom is an absence of fever, an absence of vesicular lesions (blisters, ulcers and/or sores) in the mouth, tongue, muzzle, nostrils, teats and/or feet, and/or an absence of lameness, e.g., as determined by a veterinarian or veterinary scientist. In some embodiments, the ruminant mammal is bovine, and the fever is characterized by an elevated body temperature (e.g., a body temperature within a range of about 103 °F to about 106 °F in a bovid).
[00385] In some embodiments, the method of treating FMD comprises combination therapy. In some embodiments, the present disclosure provides a method of treating FMD in a ruminant mammal, wherein the method comprises administering a vaccine to the ruminant mammal and administering a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein to the ruminant mammal. In some embodiments, the present disclosure provides a method of treating FMD in a ruminant mammal, wherein the method comprises administering the combination of a therapeutically effective amount of a conjugated boIFNZ.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein, and a vaccine to the ruminant mammal. Thus, in some embodiments, the method of treating FMD comprises administering a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNZ.3 protein, and a FMD vaccine to a ruminant mammal, thereby providing the ruminant mammal with early and long-term protection against FMD. In some embodiments, the vaccine is a FMD antigen. In some embodiments, the vaccine is an inactivated whole FMDV antigen. In some embodiments, the vaccine is a modified live vaccine. In some embodiments, the FMD vaccine is an adenovirus type 5 (Ad5) FMD (Ad5-FMD) vaccine (e.g., as disclosed in US patent no. 8,906,384, the entire contents of which are hereby incorporated by reference herein in their entirety). As FMDV is highly variable, with seven serotypes and dozens of subtypes, each one may require a specific vaccine. Furthermore, emerging strains of FMDV may require specific new vaccines to be developed. Thus, in some embodiments, the method of treatment comprises administering a therapeutically effective amount of the conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein, and a FMD vaccine that is effective in the treatment of one or more of the FMDV serotypes and/or subtypes, or an emerging FMDV strain. In some embodiments, the
method provides the ruminant mammal with early protection against FMD. In some embodiments, the early protection occurs throughout an early protection period. In some embodiments, the early protection period begins prior to the induction of protective antibody titers that can be induced via vaccination. Thus, in some embodiments, the early protection period precedes the induction of protective antibody titers post-vaccination. In some embodiments, the early protection period is a period of about 1 day, about 2 days, about 3 days, about 4 days, about 5 days, about 6 days or about 7 days or more. In some embodiments, the early protection period is a period of about 1 day. In some embodiments, the early protection period is a period of about 2 days. In some embodiments, the early protection period is a period of about 3 days. In some embodiments, the early protection period is a period of about 4 days. In some embodiments, the early protection period is a period of about 5 days. In some embodiments, the early protection period is a period of about 6 days. In some embodiments, the early protection period is a period of about 7 days. In some embodiments, the early protection period is a period of about 1 day to about 5 days. In some embodiments, the early protection period is a period of about 1 day to about 7 days. In some embodiments, the method of treatment further provides the ruminant mammal with long-term protection against FMD, including protection from FMDV infection. In some embodiments, the long-term protection period is at least about 2 weeks. In some embodiments, the long-term protection period is at least about 3 weeks. In some embodiments, the long-term protection period is at least about 4 weeks. In some embodiments, the long-term protection period is at least about a month. In some embodiments, the long-term protection period is at least about 2 months. In some embodiments, the long-term protection period is at least about 3 months. In some embodiments, the long-term protection period is about 100 days, or is about 120 days, or more. The early and/or long-term protection against FMD can be characterized, for example, by an absence of viremia. Absence of viremia can include a negative result in a test for the virus using methods known to a person of skill in the art, or methods disclosed herein. In some embodiments, the early and/or long-term protection against FMD is characterized as an absence of a clinical FMD symptom. In some embodiments, the absence of a clinical FMD symptom is an absence of fever, an absence of vesicular lesions (blisters, ulcers and/or sores) in the mouth, tongue, muzzle, nostrils, teats and/or feet, and/or an absence of lameness, e.g., as determined by a veterinarian or veterinary scientist. In some embodiments, the ruminant mammal is bovine, and the fever is characterized by an elevated body temperature (e.g., a body temperature within a range of about 103 °F to about 106 °F in a bovid). The conjugated boIFNz.3 protein can be administered prior to, after, or at about the same time as the administration of the vaccine, as determined by a person of skill in the art, or by a veterinarian or veterinary scientist. In some embodiments, the conjugated boIFNZ.3 protein or
composition is administered before the administration of the vaccine. For example, in some embodiments, the conjugated boIFNz.3 protein or composition is administered before but within about a week of the administration of the vaccine, e.g., up to about one week, about 6 days, about 5 days, about 4 days, about 3 days, about 2 days or about 1 day (e.g., up to about 24 hours, e.g., up to about 20 hours, about 16 hours, about 12 hours, about 8 hours or about 4 hours) before the administration of the vaccine. For example, in some embodiments, the conjugated boIFNz.3 protein or composition is administered about 0-7 days, about 1-6 days, about 2-5 days or 3-4 days, or is administered about 4 hours to about 7 days, about 8 hours to about 6 days, about 12 hours to about
5 days, about 24 hours to about 4 days, about 36 hours to aboyut 3 days, before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 1 day before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 2 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 3 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 4 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 5 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 6 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 7 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein is administered after the administration of the vaccine. For example, in some embodiments, the conjugated boIFNz.3 protein is administered after but within about one week of the vaccine, e.g., up to about 6 days, about 5 days, about 4 days, about 3 days, about 2 days, or about 1 day after the administration of the vaccine. For example, in some embodiments, the conjugated boIFNz.3 protein or composition is administered about 0-7 days, about 1-6 days, about 2-5 days or 3-4 days, or is administered about 4 hours to about 7 days, about 8 hours to about
6 days, about 12 hours to about 5 days, about 24 hours to about 4 days or about 36 hours to about 3 days after the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein is administered at about the same time as the administration of the vaccine (e.g., within about 4 hours, about 3 hours, about 2 hours, about 1 hour or about 30 minutes of the administration of the vaccine). In some embodiments, the method of treating the ruminant mammal for FMD comprises administering a composition comprising both FMD vaccine and the conjugated boIFNZ.3 protein to the ruminant mammal. In some embodiments, the administration is a
subcutaneous administration. In some embodiments, the administration is an intramuscular administration.
[00386] The present disclosure also provides a method of inducing systemic antiviral activity in a ruminant mammal, the method comprising administering to a ruminant mammal a therapeutically effective amount of a conjugated protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated protein. In some embodiments, the method further comprises administering a vaccine that is capable of preventing FMDV. In some embodiments, the method induces anti-FMDV activity, thereby preventing FMD. In some embodiments, the method induces adjuvanted adaptive immune response against FMDV, thereby preventing FMD. In some embodiments, the method induces expression of interferon-stimulated genes correlated with systemic control of viral replication in an animal susceptible to FMDV in vivo, thereby preventing FMD.
[00387] The present disclosure also provides a method of inducing systemic antiviral activity in a ruminant mammal, the method comprising administering a vaccine to the ruminant mammal, wherein the method further comprises administering a therapeutically effective amount of a conjugated protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated protein to the ruminant mammal. The present disclosure also provides a method of inducing systemic antiviral activity in a ruminant mammal, the method comprising administering a combination of a therapeutically effective amount of a conjugated boIFNZ.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFN/3 protein, and a vaccine to the ruminant mammal. Appropriate doses of each of the conjugated protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated protein and the vaccine, as well as appropriate timings are as disclosed elsewhere herein.
[00388] Beyond FMD, IFNs can provide protection against other important domestic livestock diseases such as bovine viral diarrhea virus (BVDV) as well as bovine respiratory syncytial virus (BRSV), infectious bovine rhinotracheitis virus (IBR), bovine coronaviruses and parainfluenza-3 viruses (PI-3) among others. These viruses are among the initial causes of the bovine respiratory disease (BRD) complex that open the door to severe bacterial infections of the lung. A nonantibiotic-based early intervention strategy to prevent and control BRD is highly desirable. BoIFNZ.3 is most active in the upper respiratory track in cattle and thus represents an opportunity for a targeted approach to treating or preventing BRD.
[00389] Thus, in some embodiments, the present disclosure provides a method of treating BRD in a ruminant mammal, wherein the method comprises administering a therapeutically effective
amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNZ.3 protein, to the ruminant mammal. In some embodiments, the ruminant mammal is bovine. In some embodiments, the ruminant mammal is a cattle. In some embodiments, the ruminant mammal is a bovine calf. In some embodiments, the method of treatment can provide protection against BRD, provide protection against BRD symptoms, provide protection against infections associated with BRD, including viral infections associated with BRD, reduce a rate of infection by a virus associated with BRD, reduce a degree of infection by a virus associated with BRD, inhibit replication of a virus associated with BRD, reduce severity of BRD, reduce severity of BRD symptoms, prevent BRD and/or prevent infection by a virus associated with BRD. In some embodiments, the method of treatment can reduce the rate of transmission, or prevent the transmission, of a virus associated with BRD from one ruminant mammal to another ruminant mammal or mammals. In some embodiments, the method further comprises administering a vaccine for the treatment of a virus associated with BRD to the ruminant mammal. In some embodiments, the virus associated with BRD is selected from the group consisting of bovine viral diarrhea virus (BVDV), bovine respiratory syncytial virus (BRSV), infectious bovine rhinotracheitis virus (IBR), a bovine coronavirus and a parainfluenza-3 virus (PI-3); and combinations thereof. In some embodiments, the virus associated with BRD is selected from the group consisting of BVDV, BRSV, a bovine coronavirus and PI-3; and combinations thereof.
[00390] In some embodiments, the method of treating BRD provides the ruminant mammal with early protection against BRD. In some embodiments, the early protection occurs throughout an early protection period. In some embodiments, the early protection period begins prior to the induction of protective antibody titers that can be induced via vaccination. Thus, in some embodiments, the early protection period begins prior to the induction of protective antibody titers post-vaccination. In some embodiments, the early protection period is a period of about 1 day, about 2 days, about 3 days, about 4 days, about 5 days, about 6 days or about 7 days or more. In some embodiments, the early protection period is a period of about 1 day. In some embodiments, the early protection period is a period of about 2 days. In some embodiments, the early protection period is a period of about 3 days. In some embodiments, the early protection period is a period of about 4 days. In some embodiments, the early protection period is a period of about 5 days. In some embodiments, the early protection period is a period of about 6 days. In some embodiments, the early protection period is a period of about 7 days. In some embodiments, the early protection period is a period of about 1 day to about 5 days. In some embodiments, the early protection period is a period of about 1 day to about 7 days. In some embodiments, the method further
provides the ruminant mammal with long-term protection against BRD, including protection from a virus associated with BRD. In some embodiments, the long-term protection occurs during a long-term protection period that begins after the administration of the conjugated protein. The long-term protection period can coincide with the early protection period. In some embodiments, the long-term protection period is at least about 2 weeks. In some embodiments, the long-term protection period is at least about 3 weeks. In some embodiments, the long-term protection period is at least about 4 weeks. In some embodiments, the long-term protection period is at least about a month. In some embodiments, the long-term protection period is at least about 2 months. In some embodiments, the long-term protection period is at least about 3 months. In some embodiments, the long-term protection period is about 100 days, or about 120 days or more. In some embodiments, the method of treating BRD provides the ruminant mammal with both early protection and long-term protection against BRD. In some embodiments, the early and/or longterm protection against BRD is an early and/or long-term protection against infection by a virus associated with BRD. In some embodiments the early and/or long-term protection is a prevention of infection by a virus associated with BRD. In some embodiments, the early and/or long-term protection against BRD is characterized as an absence of viremia. In some embodiments, the early and/or long-term protection against BRD is characterized as an absence of a clinical BRD symptom.
[00391] In some embodiments, the method of treating BRD comprises combination therapy. In some embodiments, the present disclosure provides a method of treating BRD in a ruminant mammal, wherein the method comprises administering a vaccine to the ruminant mammal, and administering a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein to the ruminant mammal. In some embodiments, the present disclosure provides a method of treating BRD in a ruminant mammal, wherein the method comprises administering the combination of a therapeutically effective amount of conjugated boIFNZ.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein, and a vaccine to the ruminant mammal. Thus, in some embodiments, the method of treating BRD comprises administering a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNZ.3 protein, and a vaccine for the treatment of a virus associated with BRD to a ruminant mammal, thereby providing the ruminant mammal with protection against BRD. In some embodiments, the vaccine is a BVDV antigen, a BRSV antigen, an IBR antigen, a PI-3 antigen or
a bovine coronavirus antigen. In some embodiments, the vaccine is an inactivated whole BVDV antigen, an inactivated whole BRSV antigen, an inactivated whole IBR antigen, an inactivated whole PI-3 antigen or an inactivated whole bovine coronavirus antigen. In some embodiments, the vaccine is a modified live vaccine. In some embodiments, the method provides the ruminant mammal with early protection against BRD. In some embodiments, the early protection occurs throughout an early protection period. In some embodiments, the early protection period is a time period that precedes the induction of protective antibody titers that can be induced via the vaccination. Thus, in some embodiments, the early protection period is a time period that begins prior to the induction of protective antibody titers post-vaccination. In some embodiments, the early protection period is a period of about 1 day, about 2 days, about 3 days, about 4 days, about 5 days, about 6 days or about 7 days or more. In some embodiments, the early protection period is a period of about 1 day. In some embodiments, the early protection period is a period of about 2 days. In some embodiments, the early protection period is a period of about 3 days. In some embodiments, the early protection period is a period of about 4 days. In some embodiments, the early protection period is a period of about 5 days. In some embodiments, the early protection period is a period of about 6 days. In some embodiments, the early protection period is a period of about 7 days. In some embodiments, the early protection period is a period of about 1 day to about 5 days. In some embodiments, the early protection period is a period of about 1 day to about 7 days. In some embodiments, the method further provides the ruminant mammal with long-term protection against FMD. In some embodiments, the long-term protection against BRD occurs during a long-term protection period after the administration. The long-term protection period can coincide with the early protection period. In some embodiments, the long-term protection period is at least about 2 weeks. In some embodiments, the long-term protection period is at least about 3 weeks. In some embodiments, the long-term protection period is at least about 4 weeks. In some embodiments, the long-term protection period is at least about a month. In some embodiments, the long-term protection period is at least about 2 months. In some embodiments, the long-term protection period is at least about 3 months. In some embodiments, the long-term protection period is about 100 days, or about 120 days or more. In some embodiments, the method provides the ruminant mammal with both early protection and long-term protection against BRD. In some embodiments, the early and/or long-term protection against BRD is a protection against infection by a virus associated with BRD. In some embodiments the early and/or long-term protection is a prevention of infection by a virus associated with BRD. In some embodiments, the early and/or long-term protection against BRD is characterized as an absence of viremia. In some embodiments, the early and/or long-term protection against BRD is characterized as an absence of a clinical BRD
symptom. In some embodiments, the virus associated with BRD is selected from the group consisting of bovine viral diarrhea virus (BVDV), bovine respiratory syncytial virus (BRSV), infectious bovine rhinotracheitis virus (IBR), a bovine coronavirus and a parainfluenza-3 virus (PI-3); and combinations thereof. In some embodiments, the virus associated with BRD is selected from the group consisting of BVDV, BRSV, a bovine coronavirus and PI-3; and combinations thereof. The conjugated boIFNz.3 protein can be administered prior to, after, or at about the same time as the administration of the vaccine, as determined by a person of skill in the art, or by a veterinarian or veterinary scientist. In some embodiments, the conjugated boIFNZ.3 protein or composition is administered before the administration of the vaccine. For example, in some embodiments, the conjugated boIFNz.3 protein or composition is administered before but within about one week of the administration of the vaccine, e.g., up to about one week, about 6 days, about 5 days, about 4 days, about 3 days, about 2 days or about 1 day (e.g., up to about 24 hours, e.g., up to about 20 hours, about 16 hours, about 12 hours, about 8 hours or about 4 hours) before the administration of the vaccine. For example, in some embodiments, the conjugated boIFNz.3 protein or composition is administered about 0-7 days, about 1-6 days, about 2-5 days or about 3- 4 days, or is administered about 4 hours to about 7 days, about 8 hours to about 6 days, about 12 hours to about 5 days, about 24 hours to about 4 days, about 36 hours to about 3 days, before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 1 day before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 2 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 3 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 4 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 5 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 6 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein or composition is administered about 7 days before the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein is administered after the administration of the vaccine. For example, in some embodiments, the conjugated boIFNz.3 protein is administered after but within about one week of the administration of the vaccine, e.g., up to about 6 days, about 5 days, about 4 days, about 3 days, about 2 days, or about 1 day after the administration of the vaccine. For example, in some embodiments, the conjugated boIFNz.3 protein or composition is administered about 0-7 days, about 1-6 days, about 2-5 days or about 3-4 days after the
administration of the vaccine, or is administered about 4 hours to about 7 days, about 8 hours to about 6 days, about 12 hours to about 5 days, about 24 hours to about 4 days or about 36 hours to about 3 days after the administration of the vaccine. In some embodiments, the conjugated boIFNz.3 protein is administered at about the same time as the administration of the vaccine (e.g., within about 4 hours, about 3 hours, about 2 hours, about 1 hour or about 30 minutes of the administration of the vaccine).
[00392] In some embodiments, the method of treating BRD further comprises administering an antibiotic to the ruminant mammal. In some embodiments, the present disclosure provides a method of treating BRD in a ruminant mammal, wherein the method comprises administering an antibiotic to the ruminant mammal, wherein the method further comprises administering a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein to the ruminant mammal. In some embodiments, the present disclosure provides a method of treating BRD in a ruminant mammal, wherein the method comprises administering the combination of a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein, and an antibiotic to the ruminant mammal. In some embodiments, the antibiotic is a penicillin, a cephalosporin, a macrolide, a tetracycline or a fluoroquinolone antibiotic; or a combination thereof. In some embodiments, the antibiotic is a cephalosporin antibiotic. In some embodiments, the antibiotic is a macrolide antibiotic. The conjugated boIFNz.3 protein can be administered prior to, after, or at about the same time as the administration of the antibiotic and/or vaccine, as determined by a person of skill in the art, or by a veterinarian or veterinary scientist. In some embodiments, the conjugated boIFNz.3 protein or composition is administered before the administration of the antibiotic. For example, in some embodiments, the conjugated boIFNz.3 protein or composition is administered before but within about a week of the administration of the antibiotic, e.g., up to about one week, about 6 days, about 5 days, about 4 days, about 3 days, about 2 days or 1 about day (e.g., up to about 24 hours, e.g. up to about 20 hours, about 16 hours, about 12 hours, about 8 hours or about 4 hours) before the administration of the antibiotic. For example, in some embodiments, the conjugated boIFNz.3 protein or composition is administered about 0-7 days, about 1-6 days, about 2-5 days or about 3- 4 days before the administration of the antibiotic, or about 4 hours to about 7 days, about 8 hours to about 6 days, about 12 hours to about 5 days, about 24 hours to about 4 days or about 36 hours to about 3 days before the administration of the antibiotic. In some other embodiments, the conjugated boIFNz.3 protein is administered after the administration of the antibiotic. For
example, in some embodiments, the conjugated boIFNZ.3 protein is administered after but within about one week of the administration of the antibiotic e.g., up to about 6 days, about 5 days, about 4 days, about 3 days, about 2 days or about 1 day after the administration of the antibiotic. For example, in some embodiments, the conjugated boIFNZ.3 protein or composition is administered about 0-7 days, about 1-6 days, about 2-5 days or about 3-4 days, about 4 hours to about 7 days, about 8 hours to about 6 days, about 12 hours to about 5 days, about 24 hours to about 4 days or about 36 hours to about 3 days after the administration of the antibiotic. In some embodiments, the conjugated boIFNz.3 protein is administered at about the same time as the administration of the antibiotic (e.g., within about 4 hours, about 3 hours, about 2 hours, about 1 hour or about 30 minutes of the administration of the antibiotic).
[00393] In some embodiments, the antibiotic is a penicillin. In some embodiments, the penicillin is selected from the group consisting of an aminopenicillin (including ampicillin and amoxicillin), aztreonam, carbenicillin, a carboxypenicillin (including carbenicillin, its acid-stable indanyl ester, and ticarcillin), clavulanate-potentiated amoxicillin, cioxacillin, floxacillin, hetacillin, imipenem, an isoxazolyl penicillin (including oxacillin, cioxacillin, dicloxacillin and flucloxacillin), mecillinam, meropenem, methicillin, nafcillin, oxacillin, penicillin G (benzylpenicillin), penicillin V (phenoxymethyl-penicillin), phenethicillin, a piperazine penicillin (e.g., piperacillin); pivampicillin, sulbactam-potentiated ampicillin, talampicillin, tazobactam-potentiated piperacillin, temocillin, ticarcillin, a ureido-penicillin (including azlocillin and mezlocillin) and streptocillin; and combinations thereof.
[00394] In some embodiments, the antibiotic is a macrolide. In some embodiments, the macrolide antibiotic is selected from the group consisting of azithromycin, erythromycin, erythromycin A, erythromycin B, erythromycin C, erythromycin D, erythromycin E, gamithromycin, tilmicosin, tulathromycin, tildipirosin and tylosin; and combinations thereof.
[00395] In some embodiments, the antibiotic is a cephalosporin. In some embodiments, the cephalosporin is selected from the group consisting of cefachlor, cefadroxil, cefamandole, cefazolin, cefepime, cefoperazone, ceforanide, cefotaxime, cefotiam, cefpodoxime, ceftazidime, cefovecin, cefoxitin (a cephamycin), cefsulodin, ceftiofur, ceftriaxone, cefuroxime, cephalexin, cephaloridine, cephalothin, cephapirin and cephradine; and combinations thereof.
[00396] In some embodiments, the antibiotic is a tetracycline. In some embodiments, the tetracycline is selected from the group consisting of chlortetracycline, demethylchlortetracycline, doxycycline, a glycylcycline (including tigecycline), lymecycline, methacycline, minocycline, oxytetracycline, rolitetracycline and tetracycline; and combinations thereof.
[00397] In some embodiments, the antibiotic is a fluoroquinolone antibiotic. In some embodiments, the fluoroquinolone antibiotic is Enrofloxacin (Baytril).
[00398] The present disclosure also provides a method of inducing systemic antiviral activity in a ruminant mammal, the method comprising administering to a ruminant mammal a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure or a pharmaceutical composition comprising a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, and further comprising administering a vaccine for treatment of BRD. The present disclosure also provides a method of inducing systemic antiviral activity in a ruminant mammal, the method comprising administering a vaccine to a ruminant mammal, wherein the method further comprises administering a therapeutically effective amount of a conjugated protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated protein to the ruminant mammal. The present disclosure also provides a method of inducing systemic antiviral activity in a ruminant mammal, the method comprising administering a combination of a therapeutically effective amount of a conjugated boIFNz.3 protein of the present disclosure, or a pharmaceutical composition comprising a therapeutically effective amount of the conjugated boIFNz.3 protein, and a vaccine to the ruminant mammal.
[00399] In some embodiments, the vaccine for the treatment of BRD is a vaccine for the treatment of BVDV, BRSV, IBR, a bovine coronavirus and/or PI-3. In some embodiments, the method induces anti-BVDV, anti-BRSV, anti-IBR, anti-bovine coronavirus and/or anti-PI-3 activity, thereby delaying, reducing severity and/or preventing BRD. In some embodiments, the method induces adjuvanted adaptive immune response against BRD, thereby delaying, reducing severity and/or preventing BRD. In some embodiments, the method induces expression of interferon- stimulated genes correlated with systemic control of viral replication in an animal susceptible to a BRD-associated virus in vivo, thereby delaying, reducing severity and/or preventing BRD.
[00400] Use of engineered boIFNz.3 proteins and/or conjugated boIFNz.3 proteins of the present disclosure may reduce morbidity in cattle or other livestock species as well.
EXAMPLES
[00401] It is understood that the examples and embodiments described herein are for illustrative purposes only and that various modifications or changes in light thereof will be suggested to persons skilled in the art and are to be included within the spirit and purview of this application and scope of the appended claims.
[00402] Example 1 : boIFNz.3 expression system
[00403] This example describes expression systems and methods used for boIFNz.3 polypeptides comprising wild type amino acid sequence (SEQ ID NO: 1; Diaz-San Segundo F. et al. (2011) Virology 413:283-292; GenBank: ADP05157.1), modified amino acid sequences (N-terminal deletion variants of SEQ ID NOs: 2 to 13), and amino acid sequences containing a non-natural amino acid (SEQ ID NOs: 14 to 25). Host cells were transformed with constructs for orthogonal tRNA, orthogonal aminoacyl tRNA synthetase, and a polynucleotide encoding boIFNZ.3 polypeptide as in SEQ ID NOs: 2 to 13, or a polynucleotide comprising a selector codon encoding the amino acid sequences shown in SEQ ID NOs: 14 to 25 (e.g., the polynucleotide of SEQ ID NO: 27 encoding amino acid SEQ ID NO: 18).
[00404] E. coli expression vector construction and sequence verification: cloning and expression of wild type boIFNz.3, modified boIFNZ.3 sequences (N-terminal deletion variants), and boIFNZ.3 containing a non-naturally encoded amino acid in E. coli. BoIFNz.3 expression plasmids were constructed either by recombination-based cloning method using Gibson Assembly kit (New England Biolabs (NEB), Ipswich, MA) or by using QuikChange mutagenesis kit (Agilent Technologies, Santa Clara, CA) in E. coli NEB5a cloning strain (New England Biolabs, MA) as described below. The E. coli expression plasmids are shown in FIGs. 1 A and IB.
[00405] Gibson Assembly: The primers for amplifying various gene of interests (GOIs) containing Donor fragments had about 18-24 base pair (bp) overlap sequence at their 5'-termini with the Acceptor vector sequences for homologous recombination and were synthesized at Integrated DNA Technologies (IDT) (San Diego, CA). The PCR fragments were amplified using high fidelity DNA polymerase mix, Pfu Ultra II Hotstart PCR Master Mix (Cat. No: 600852, Agilent Technologies). The PCR products were digested with Dpnl restriction enzyme (NEB# R0176L) for 2 hours at 37 °C to remove plasmid background followed by column purification using Qiagen PCR column purification kit (Qiagen, Valencia, CA; # 28104) and quantitated by Nanodrop (ThermoFisher, Carlsbad, CA). The Acceptor vectors were linearized by digesting with unique restriction enzymes (NEB, MA) within the vector for 3 to 5 hours at supplier recommended temperatures, PCR column purified and quantitated. The Donor inserts and appropriately prepared Acceptor vectors were mixed at a 3:1 molar ratio, incubated at 50 °C for 15 min, using Gibson Assembly kit (NEB # E261 IS), and then used for transformation into E. coli NEB5a strain (New England Biolabs, Ipswitch, MA (NEB); NEB # 2987).
[00406] The recombinants were recovered by plating Gibson Assembly mix on to lysogeny broth (LB) agar plates containing appropriate antibiotics. The next day, 4 to 6 well-isolated single colonies were inoculated into 5 mL LB + 50 pg/mL kanamycin sulfate (Sigma, St Louis, MO; cat# K0254) media and grown overnight at 37 °C. The recombinant plasmids were isolated using
Qiagen plasmid DNA mini -prep kit (Qiagen #27104) and verified by DNA sequencing (Eton Biosciences, San Diego, CA). The complete GOI region plus 100 bp upstream and 100 bp downstream sequences were verified by using gene-specific sequencing primers.
[00407] QuickChange Mutagenesis (QCM): All Amber variants containing TAG stop codon were created by using QuickChange Lightning site directed mutagenesis kit (Agilent Technologies # 201519). All QCM oligonucleotides were designed using QuickChange Web Portal (Agilent Technologies), and ordered from IDT (San Diego, CA). The QCM PCR mix contained 5 pl of lOx buffer, 2.5 pl of dNTP Mix, 1 pl (100 ng) of plasmid template, 1 pl of oligo mix (10 pM concentration each), 1 pl of QuickChange Lightning enzyme, 2.5 pl of Quick solution and 37 pl of distilled water (DW). The DNA was amplified using the PCR program recommended by the kit for 18 cycles only.
[00408] After completion of the PCR reaction, the mix was digested with Dpnl enzyme that came with the kit (Agilent Technologies) for 2-3 hour at 37 °C and ran on a gel to check the presence of amplified PCR product. Thereafter, 2.5 to 5 pl of PCR product was transformed into E.coli NEB5a strain. The recombinant plasmids from 4 to 6 colonies were then isolated and sequence verified as described in Gibson Assembly section above.
[00409] Expression strain (AXID) construction and verification: To prepare AXID production strains, chemically competent E. coli W3110B60 host cells were transformed with sequence- verified plasmid DNA (50 ng), the recombinant cells were selected on 2xYT+l% glucose agar plates containing 50 pg/mL kanamycin sulfate (Sigma, cat# K0254), and incubated overnight at 37 °C. A single colony from the fresh transformation plate was then propagated thrice on 2xYT+l% glucose agar plates containing 50 pg/mL kanamycin sulfate by sequential triplestreaking and incubating overnight at 37 °C. Finally, a single colony from the third-streaked plate was inoculated into 20 mL Super Broth (Fisher-OptigrowTM, #BP1432-1OB1) containing 50 pg/mL kanamycin sulfate (Sigma, cat# K0254) and incubated overnight at 37 °C and 250 rpm. The overnight grown culture was then diluted with glycerol to a final glycerol concentration of 20% (w/v) (KIC, Ref# 67790-GL99UK). This cell suspension was then dispensed into 1 mL aliquots into several cryovials and frozen at -80 °C as AXID production strain vials.
[00410] After the generation of glycerol vials of the AXID production strains as described above, they were further validated by DNA sequencing and phenotypic characterization of antibiotic resistance markers. To confirm that the AXID production strain vial had the correct plasmid in the production host, the plasmid was sequence verified. Twenty mL LB containing 50 pg/mL kanamycin sulfate was inoculated with a stab from a glycerol vial of the AXID clone and grown at 37 °C, 250 rpm overnight. The plasmid DNA was isolated using Qiagen Miniprep Kit (#27104)
and the presence of intact GOI ORF in the isolated plasmid was confirmed by DNA sequencing (Eton Biosciences, CA).
[00411] To further verify the strain genotype of the AXID production strains, cells from the same vial were streaked onto four separate plates of LB: LB containing 50 pg/mL kanamycin sulfate, LB containing 15 pg/mL tetracycline, LB containing 34 pg/mL chloramphenicol and LB containing 75 pg/mL trimethoprim. They were then checked for positive growth on all of these plates, as expected with the strain genotype of W3110B60 production host strain.
[00412] Expression system: The amino acid sequences for boIFNZ.3 are shown in Table 1. A translation system that comprises an orthogonal tRNA (O-tRNA) and an orthogonal aminoacyl tRNA synthetase (O-RS) (see, W02006/068802 and W02007/021297) was used to express wild type boIFNz.3 sequence (see plasmid map pKGOlOO, FIG. 1A), boIFNz.3 sequences with N- terminal modifications, and boIFNZ.3 sequences containing a non-naturally encoded amino acid (see plasmid map pKG0168, FIG. IB). The O-RS preferentially aminoacylates the O-tRNA with a non-naturally encoded amino acid. In turn the translation system inserts the non-naturally encoded amino acid into boIFNz.3 or boIFNz.3 variants, in response to an encoded selector codon. Suitable O-RS and O-tRNA sequences are described in W02006/068802 entitled “Compositions of Aminoacyl-tRNA Synthetase and Uses Thereof’ and W02007/021297 entitled “Compositions of tRNA and Uses Thereof’, the contents of each of which are hereby incorporated by reference in their entirety herein.
[00413] The transformation of E. coli with plasmids containing the wild type or modified boIFNZ.3 variant polynucleotide sequence and the orthogonal aminoacyl tRNA synthetase/tRNA pair (specific for the desired non-naturally encoded amino acid) allows the site-specific incorporation of non-naturally encoded amino acid into the boIFNz.3 polypeptide. Expression of boIFNZ.3 variant polypeptides is under the control of the T7 promoter and induced by the addition of arabinose in the media (see plasmid map pKGOlOO; FIG. 1 A and pKG0168; FIG. IB).
[00414] Example 2: Deletion of N-terminal amino acids for improved expression of boIFNZ.3 [00415] N-terminal amino acids 1-12 were sequentially removed from boIFNZ.3 wild type sequence (SEQ ID NO: 1; Diaz-San Segundo F. et al. (2011) Virology 413:283-292; GenBank: ADP05157.1) using QuickChange Mutagenesis (QCM) as described in Example 1 to provide variants of SEQ ID NOs: 2 to 13 (Table 1). The AXID production strain, also described in Example 1, was used to test for boIFN 3 expression in shake flask experiments. Briefly, plasmids for the expression of modified boIFNz.3 polypeptides were transformed into W3110B60 E. coli cells. Protein expression was induced by the addition of arabinose and cultures were incubated for 4 to 6 hours at 37 °C. An aliquot from the harvested cells were taken and analyzed for improved
expression by SDS-PAGE; improved expression was seen with A4 and A5 N-terminal deletion variants (SEQ ID NO: 5 and SEQ ID NO: 6, respectively; data not shown). Recoverable titers post-purification were as follows: boIFNz.3 wild type: not detectable; boIFNz.3 Al : 8.5 mg/L; boIFNz.3 A3: 97.6 mg/L; boIFNz.3 A4: 107.6 mg/L; boIFNz.3 A5: 109.6 mg/L; and boIFNZ.3 A6: 64.6 mg/L.
[00416] Suppression with pAF: Plasmids for the expression boIFNz.3 polypeptides were transformed into W3110B60 E. coli cells. Para-acetyl-L-phenylalanine (pAF) was added to the cells, and protein expression was induced by the addition of arabinose. SDS-PAGE analysis of the expression of boIFNz.3 polypeptide was performed, and the boIFNz.3 polypeptides were observed. Lanes were run for comparison between the original wild type boIFNz.3 polypeptide and for the pAF-substituted boIFNz.3 polypeptides, a boIFNz.3 with, for example, a pAF substitution made at a particular amino acid residue. Cultures were incubated for 4 to 6 hours at 37 °C.
[00417] Additional constructs to increase boIFNz.3 expression in E. coli: To increase the production of boIFNz.3 in E. coli, the following expression parameters could be further optimized in addition to DNA sequence optimization based on E. coli codon usage reported herein: testing different promoters besides T7 bacteriophage promoter such as arabinose B (araB), pTrc and bacteriophage T5 promoters; stabilization ofboIFNX3 mRNA; screening of different E. coli host strains besides the standard W3110B60 strain; production process parameters optimization such as temperature, culture media, inducer concentration, etc.; transcriptional and translational control elements optimization such as start and stop codons, ribosome binding site (RBS), transcriptional terminators etc; plasmid copy number and plasmid stability optimization; translational initiation region (TIR) optimization.
[00418] Example 3: Purification of boIFNz.3 N-terminal deletion variants
[00419] Inclusion body prep. Inclusion bodies containing boIFNz.3 N-terminal deletion variants were isolated through a series of wash steps. Frozen cell paste was re-suspended in wash buffer I (50 mM Tris, pH 8.0; 1% triton X-100; 100 mM NaCl, 1 mM EDTA) to a concentration of 10% (w/v) and homogenized at 4 °C followed by centrifugation (10,000g, 30 min, 4 °C). The supernatant was discarded, and the inclusion body pellet was re-suspended in an additional volume of wash buffer I followed by centrifuged at 15,000g for 30 min at 4 °C. The supernatant was discarded, and the inclusion body pellet was re-suspended in wash buffer II (50 mM Tris, pH 8.0; 100 mM NaCl, 1 mM EDTA) followed by centrifuged at 10,000g for 30 min at 4 °C. The supernatant was discarded, and the inclusion body pellet was re-suspended in water followed by
centrifugation at 10,000g for 30 min at 4°C. Washed inclusion bodies were stored at -80 °C until further use.
[00420] Refolding. Inclusion bodies were solubilized by resuspension in 20 mM tris, pH 8.0; 6.0 M guanidine hydrochloride. Under mixing, boIFNZ.3 variants were refolded by diluting solubilized material 1 : 10 into 20 mM tris, 1.0M arginine, pH 8.0, 1 mM cysteine at 4 °C. After addition of solubilized material, the refolding reaction was allowed to incubate at RT for 16-24 hours.
[00421] Column Purification. The refold reaction was pH adjusted to pH 5.5 with acetic acid followed by a 5-fold dilution with water. The pH adjusted reaction mixture was conditioned by filtering through a 0.22 pm filter and loaded over a Capto SP Impres (GE Healthcare) column equilibrated with 25 mM potassium phosphate, pH 6.5. The column was washed with 3-5 column volumes (CV) 25 mM potassium phosphate, pH 6.5 and eluted with a linear gradient over 20 CV to 25mM potassium phosphate, pH 6.5; 0.5M NaCl. Fractions containing boIFNZ.3 were collected and buffer exchanged into 30 mM sodium acetate, 200 mM glycine, 2.5% trehalose, pH 4.0. BoIFNZ.3 was concentrated to 1-10 mg/mL, 0.22 pM filtered, and stored at -80 °C. Purified samples were characterized by SDS-PAGE (data not shown), reversed phase HPLC (RP), and size exclusion chromatograph (SEC); (Table 2).
[00422] Table 2. Analytical size exclusion and reversed phase characterization of boIFNz.3 variants.
[00423] Example 4: Real-time quantitative polymerase chain reaction (RT-qPCR) of ISG15 after treatment with boIFNz.3 variants in Madin-Darby bovine kidney (MDBK) cells
[00424] Interferon-stimulated gene product 15 (ISG15), a ubiquitin-like protein involved in interferon induced antiviral response, was evaluated in an MDBK cell-based assay for induction levels based on treatment with boIFNz.3 variants. MDBK cells were treated with boIFNz.3 variants at the following concentrations: 100, 20, 4, 0.8, 0.16 ng/mL, and cells were isolated 6 hours post treatment to evaluate relative mRNA expression of ISG15. Total RNA was isolated using TRIzol Reagent (Thermo Fisher Scientific) and reverse transcribed into cDNA using TaqMan™ Fast
Advanced Master Mix (Applied Biosystems) according to manufacturer's protocol. Relative fold changes in ISG15 gene expression were evaluated and normalized against control. N-terminal deletion variants up to 12 amino acids (variants Al to A12; Table 1) exhibited mRNA expression of ISG15 (data not shown).
[00425] Example 5: Site selection for incorporation of non-natural amino acid
[00426] The sites to be used in generating boIFNz.3 muteins were selected by modeling nonnatural amino acid para-acetyl-L-phenylalanine (pAF) at each amino acid site of full length human IFNz.3 protein crystal structure and assessing side chain accessible surface area (scASA) of the non-natural amino acid using Molecular Operating Environment (MOE) (Chemical Computing Group). Lead sites selected for pAF incorporation, with their associated scASA values, are disclosed in Table 3. Sites known to be important for receptor binding were avoided. FIG. 2 shows the boIFNZ.3 model with amino acids selected for pAF incorporation.
[00427] Table 3. Calculated scASA values of pAF incorporated into amino acid sites in boIFNL3.
[00428] Example 6: E. coli shake flask expression of pAF site variants and high cell density fermentation
[00429] Shake-flask expression testing: The AXID production strain as described in Example 1 was used to test for boIFNz.3 expression in shake flask experiments. Briefly, an inoculum from the AXID glycerol vial was put into a 5 mL of Super Broth (Fisher-OptigrowTM, #BP1432-1OB1) media containing 50 pg/mL of kanamycin sulfate (Sigma, MO) and grown overnight at 37°C with shaking. The overnight culture was diluted 1 : 100 in Super Broth (Fisher-OptigrowTM, #BP1432- 10B1) media containing 50 pg/mL of kanamycin sulfate (Sigma, MO) and grown at 37 °C with
shaking. When the culture density reached an OD600 of 0.6-0.8, it was induced with 0.2% arabinose and non-natural amino acid pAF added, followed by harvesting after about 3 to 5 hours of production. An aliquot from the harvested cells was taken and analyzed by SDS-PAGE (data not shown). Optimal expression of boIFNZ.3 was standardized by varying temperature, duration of induction and inducer concentration.
[00430] High cell density fermentations: The fermentation process for production of boIFNZ.3 consisted of two stages: (i) inoculum preparation and (ii) fermentor production. The inoculum was started from a single glycerol vial, thawed, diluted 1 : 1000 (v/v) into 50 mL of defined seed medium in a 250 mL baffled Erlenmeyer flask, and incubated at 37 °C and 250 rpm. Prior to use, the fermentor was cleaned and autoclaved. A specified amount of basal medium was added to the fermentor and steam sterilized. Specified amounts of kanamycin sulfate solution, feed medium and P2000 antifoam were added to the basal medium prior to inoculation. All solutions added to the fermentor after autoclaving were either 0.2 pm filtered or autoclaved prior to aseptic addition. [00431] The fermentor was batched with 4 L of chemically defined medium that utilizes glycerol as a carbon source. The seed culture was added to the fermentor to an initial OD600nm of 0.05. Dissolved oxygen was maintained at 30% air saturation using agitation from 480 to 1200 rpm and oxygen enrichment with a head pressure of 6 psig and air flow of 5 slpm. The temperature and pH were controlled at 37 °C and 7.0, respectively. When the culture reached an OD600nm of 35 ± 5, feeding commenced at a rate of 0.25 mL/L/min. Consequently, L-Ala-pAcF, (also referred to as L-Ala-pAF), dipeptide was added at 0.4 g/L. Fifteen minutes after the addition of dipeptide, the culture was induced with L-arabinose at a final concentration of 2 g/L. The culture was harvested at 6 h post induction.
[00432] Example 7: Purification of boIFNZ.3 N-terminal A4 deletion variants containing nonnatural amino acid
[00433] Inclusion body prep. Inclusion bodies were isolated through a series of wash steps. Frozen cell paste was re-suspended in wash buffer I (50 mM Tris, pH 8.0; 1% triton X-100; 100 mM NaCl, 1 mM EDTA) to a concentration of 10% (w/v) and homogenized at 4 °C followed by centrifugation (10,000g, 30 minutes, 4 °C). The supernatant was discarded, and the inclusion body pellet was re-suspended in an additional volume of wash buffer I followed by centrifugation at 15,000g for 30 minutes at 4 °C. The supernatant was discarded, and the inclusion body pellet was re-suspended in wash buffer II (50 mM Tris, pH 8.0; 100 mM NaCl, 1 mM EDTA) followed by centrifuged at 10,000g for 30 minutes at 4°C. The supernatant was discarded, and the inclusion body pellet was re-suspended in water followed by centrifugation at 10,000g for 30 minutes at 4 °C. Washed inclusion bodies were stored at -80 °C until further use.
[00434] Refolding. Inclusion bodies were solubilized by resuspension in 20 mM tris, pH 8.0; 6.0 M guanidine hydrochloride. Under mixing, boIFN/3 was refolded by diluting solubilized material 1 : 10 into 20 mM tris, 1.0 M arginine, pH 8.0, 1 mM cysteine at 4 °C. After addition of solubilized material, the refolding reaction was allowed to incubate at RT for 16-24 hours.
[00435] Column purification. The refolding reaction was pH adjusted to pH 5.5 with acetic acid followed by a 5-fold dilution with water. The material was 0.22pm filtered and loaded over a Capto SP Impres (GE Healthcare) column equilibrated with 25 mM potassium phosphate, pH 6.5, then washed with 3-5CV 25 mM potassium phosphate, pH 6.5 and eluted with a linear gradient over 20 CV to 25 mM potassium phosphate, pH 6.5; 0.5 M NaCl. Fractions containing bovine IFN-Z.3 were collected and buffer exchanged into 30 mM sodium acetate, 200 mM glycine, 2.5% trehalose, pH 4.0. BoIFNz.3 variants containing non-natural amino acid pAF were concentrated to 1-10 mg/mL, 0.22 pM filtered and stored at -80 °C.
[00436] Example 8: Site Specific Conjugation of boIFNz.3 with PEG and boIFNX3-PEG Purification.
[00437] BoIFNz.3 variants (SEQ ID NOS: 14-18 and 20-25) containing non-natural amino acid pAF were buffer exchanged into conjugation buffer (30 mM sodium acetate, 200 mM glycine, 2.5% trehalose, pH 4.0) and concentrated to 1-10 mg/mL. A final of 100 mM acetic hydrazide was added to the reactions followed by an 8-molar excess of aminooxy functionalized 30kDa PEG (NOF America Corp.). The conjugation reactions were incubated for 18-20 hours at 25-30 °C. Following conjugation, the PEGylated boIFNz.3 was diluted 1 :10 with 30 mM sodium acetate, pH 5.0 and loaded over a Capto SP Impres column equilibrated with 25 mM potassium phosphate, pH 6.5. The loaded column was washed with 2-5 CV 25mM potassium phosphate, pH 6.5 and boIFNX3-PEG was eluted with a linear gradient over 20 CV to 25 mM potassium phosphate, pH 6.5; 0.5M NaCl. Fractions containing boIFNX3-PEG were collected and buffer exchanged into 20 mM potassium phosphate, pH 6.5; 100 mM NaCl; 2.5% trehalose. BoIFNz.3 was concentrated to 1-10 mg/mL, 0.22 pM filtered, and stored at -80 °C. Purified samples were characterized by SDS- PAGE, followed by reversed phase (RP) HPLC and size exclusion chromatography analysis (Table 4).
[00438] Table 4. Analytical size exclusion and reversed phase characterization of boIFNz.3- PEG30kDa variants.
[00439] Example 9: Real-time quantitative polymerase chain reaction (RT-qPCR) of ISG15 after treatment with PEGylated boIFNZ.3 variants in MDBK cells
[00440] MDBK cells were treated with boIFNX3-PEG variants (500, 50, and 5 ng/mL). Cells were isolated 6 hours post treatment to evaluate relative mRNA expression of ISG15. Total RNA was isolated using TRIzol Reagent (Thermo Fisher Scientific) and reverse transcribed into cDNA using TaqMan™ Fast Advanced Master Mix (Applied Biosystems) according to manufacturer's protocol. Relative fold changes in ISG15 gene expression were evaluated and normalized against non-treated control cells (FIG. 3).
[00441] Example 10: MDBK ISRE-luciferase reporter assay.
[00442] MDBK cells transfected with pGL4.45[luc2P/ISRE/Hygro] Vector (Promega, Cat. # E414A) were treated with boIFNX3-PEG variants (1000, 200, 40, 8, 16., 0.32, 0.64, and 0.0002 ng/mL). Post 7 hours treatment, luciferase luminescence was evaluate using ONE-Glo™ Luciferase Assay System according to manufactures instructions (Promega, Cat. # E6120). Luciferase luminescence was normalized to untreated cells (FIG. 4). Variants that retained activity in this reporter assay were advanced to further testing.
[00443] Among the variants discussed in the previous examples, T119 (SEQ ID NO: 18) is exemplified in the following in vitro and in vivo studies.
[00444] Example 11 : In Vitro Antiviral Testing - Bovine Respiratory Disease Viruses
[00445] The antiviral activity of boIFN/A-T I 19-PEG30KDa against viruses associated with bovine respiratory disease was evaluated in vitro.
[00446] Viral strains were purchased from the USDA National Veterinary Services Laboratory. Bovine viral diarrhea virus, Singer strain (cytopathic, genotype la (NVSL 140BDV); BRSV strain A51908 (NVSL 110BDV) and IBRV, also called bovine herpesvirus 1, Colorado strain (NVSL 050BDV) were propagated and titrated in Madin-Darby bovine kidney (MDBK) cells maintained in minimum essential medium supplemented with 10% fetal bovine serum, 2 mM L-glutamine, 0.05 mg/ml gentamicin, 10 unit/ml penicillin, 10 pg/ml streptomycin, and 0.25 pg/ml amphotericin.
[00447] BoIFN/3-TI 19-PEG30KDa, prepared according to Example 8, was serially diluted in cell culture medium to generate concentrations of 1000, 250, 62.5, 15.63, 3.91, 0.98, 0.06 and 0 ng/ml. MDBK cells were cultured in 24-well plates (1 plate per virus). Duplicate wells containing each concentration of boIFN/3-T I I 9-PEG30KDa were prepared for each plate. Cells were cultured in the presence of boIFNX3-T119-PEG30KDa for approximately 18 hr prior to viral inoculation. Culture medium was removed from each well of MDBK cells and replaced with 300 pl of medium containing individual viruses. Plates were incubated at 37 °C in 5% CO2 for 1 hr. The virus inoculum was removed from each well and 2 ml of culture medium containing the same concentrations of boIFN/3-T I 19-PEG30KDa used initially was added to each corresponding well. [00448] The plates were incubated at 37 °C in 5% CO2 incubator for 24 hr and virus-induced cytopathic effect (CPE) development was recorded using a scale of 0-4 (0: no CPE; 1 : 25% CPE; 2: 50% CPE; 3: 75% CPE; 4: 100% CPE). Plates were frozen and thawed once to generate cell lysates which were harvested. Following centrifugation, the supernatants of the cell lysates were serially diluted in culture medium and titrated in fresh MDBK cells cultured in 96-well plates (triplicate wells were used for each dilution). After 3-4 days, plates were observed for CPE rinsed and fixed with 80% cold acetone. Fifty pl of fluorescent antibody conjugates specific for each virus were added to each corresponding well and the plates were incubated for 1 hr at 37 °C. The antibody conjugate was discarded, and plates were washed with phosphate buffered saline. Plates were read under fluorescence microscopy and virus infectious titers were calculated according to the Reed and Muench method (based on CPE and immunofluorescence staining results) and expressed as the 50% tissue culture infective dose (TCID50)/ml.
[00449] The impact of boIFNX3-Tl 19-PEG30KDa on bovine respiratory disease virus-induced cytopathic effect and viral titers is presented in Tables 5-7 below.
[00450] Table 5. Antiviral activity of boIFNX3-Tl 19-PEG30KDa against BVDV genotype la.
[00451] Table 6. Antiviral activity of boIFN/A-T I 19-PEG30KDa against BRSV.
[00453] The data indicated that boIFN/3-T I 19-PEG30KDa completely inhibited replication of a cytopathic, genotype la BVDV strain at concentrations > 250 ng/ml, and reduced replication of BRSV by 20% to 30% at concentrations > 62.5 ng/ml. These results indicate boIFNX3-Tl 19- PEG30KDa eliminates or reduces the ability of some important viruses associated with bovine respiratory disease to replicate in vitro.
[00454] Example 12: In Vitro Antiviral Testing - Bovine Viral Diarrhea Virus Strains
[00455] The antiviral activity of boIFN/.3-T I 19-PEG30KDa against cytopathic and non- cytopathic genotype la and lb strains of BVDV was evaluated in vitro.
[00456] Bovine viral diarrhea virus (BVDV) strains were purchased from the USDA National Veterinary Service Laborator. Singer strain (cytopathic, genotype la (NVSL140BDV) and BVDV
Draper strain (noncytopathic, genotype lb (NVSL 141BDV) were propagated and titrated in MDBK cells maintained in minimum essential medium supplemented with 10% fetal bovine serum, 2 mM L-glutamine, 0.05 mg/ml gentamicin, 10 unit/ml penicillin, 10 pg/ml streptomycin, and 0.25 pg/ml amphotericin.
[00457] BoIFN/3-TI 19-PEG30KDa, prepared according to Example 8, was serially diluted in cell culture medium to generate concentrations of 1000, 250, 62.5, 15.63, 3.91, 0.98, 0.06 and 0 ng/ml. MDBK cells were cultured in 24-well plates (1 plate per virus strain). Duplicate wells containing each concentration of boIFN/3-T I 19-PEG30KDa were prepared for each plate. Cells were cultured in the presence of boIFN/3-T I 19-PEG30KDa for approximately 18 hr prior to viral inoculation. Culture medium was removed from each well of MDBK cells and replaced with 300 pl of medium containing individual viral strains. Plates were incubated at 37 °C in 5% CO2 for 1 hr. The virus inoculum was removed from each well and 2 ml of culture medium containing the same concentrations of boIFNX3-Tl 19-PEG30KDa used initially was added to each corresponding well.
[00458] The plates were incubated at 37°C in 5% CO2 incubator for 24 hr and cytopathic effect (CPE) development was recorded using a scale of 0-4 (0: no CPE; 1 : 25% CPE; 2: 50% CPE; 3: 75% CPE; 4: 100% CPE). Plates were frozen and thawed once to generate cell lysates which were harvested. Following centrifugation, the supernatants of the cell lysates were serially diluted in culture medium and titrated in fresh MDBK cells cultured in 96-well plates (triplicate wells were used for each dilution). After 3 to 4 days, plates were observed for CPE and the cells rinsed once and fixed with 80% cold acetone. Fifty pl of mouse-source, anti-BVDV monoclonal antibody was added to each well and plates were incubated at 37°C for 1 hr. The primary antibody was discarded and the plates were washed with phosphate buffered saline (PBS). Fifty pl of the secondary antibody conjugate (goat anti-mouse IgG conjugated to FITC) was added to the wells and plates were incubated at 37°C for 1 hr. The antibody conjugate was discarded, and plates were washed with PBS. Plates were read under fluorescence microscopy and virus infectious titers were calculated according to the Reed and Muench method (based on CPE and immunofluorescence staining results) and expressed as the 50% tissue culture infective dose (TCID50)/ml.
[00459] The impact of boIFNX3-T119-PEG30KDa on bovine viral diarrhea virus-induced cytopathic effect and viral titers is presented in Tables 8 and 9 below.
[00460] Table 8. Antiviral activity of boIFNX3-Tl 19-PEG30KDa against the cytopathic, genotype la Singer strain of BVDV.
[00461] Table 9. Antiviral activity of boIFNX3-Tl 19-PEG30KDa against the non-cytopathic, genotype lb Draper strain of BVDV.
[00462] The data indicated that boIFNX3-T119-PEG30KDa completely inhibited replication of the cytopathic, genotype la and non-cytopathic, genotype lb strains of BVDV at concentrations > 250 ng/ml. These results showed that boIFN/3-T I I 9-PEG30KDa exhibits antiviral activity against both cytopathic and non-cytopathic strains of BVDV associated with disease in cattle.
[00463] Example 13: In Vitro Antiviral Testing - Cytopathic, Genotype 2 Bovine Viral Diarrhea Virus
[00464] The antiviral activity of boIFN/.3-T I 19-PEG30KDa against a cytopathic, genotype 2 strain of BVDV was evaluated in vitro.
[00465] Cytopathic, genotype 2 strain 125 of BVDV was purchased from the USDA National Veterinary Service Laboratory. The virus was propagated and titrated in MDBK cells maintained in minimum essential medium supplemented with 10% fetal bovine serum, 2 mM L-glutamine, 0.05 mg/ml gentamicin, 10 unit/ml penicillin, 10 pg/ml streptomycin, and 0.25 pg/ml amphotericin.
[00466] BoIFNX3-Tl 19-PEG30KDa, prepared according to Example 8, was serially diluted in cell culture medium to generate concentrations of 1000, 500, 250 and 0 ng/ml. MDBK cells were cultured in 24-well plates. Triplicate wells containing each concentration of boIFNX3-PEG were prepared. Cells were cultured in the presence of boIFNX3-T119-PEG30KDa for approximately
18 hr prior to viral inoculation. Culture medium was removed from each well of MDBK cells and replaced with 300 pl of medium containing virus. Plates were incubated at 37 °C in 5% CO2 for 1 hr. The virus inoculum was removed from each well and 2 ml of culture medium containing the same concentrations of boIFNX3-Tl 19-PEG30KDa used initially was added to each corresponding well. The plates were incubated at 37 °C in 5% CO2 incubator for 24 hr, and cytopathic effect (CPE) development was recorded using a scale of 0-4 (0: no CPE; 1 : 25% CPE; 2: 50% CPE; 3: 75% CPE; 4: 100% CPE). Cell plates were frozen and thawed once to generate cell lysates which were harvested. Following centrifugation, the supernatants of the cell lysates were serially diluted in culture medium and titrated in fresh MDBK cells cultured in 96-well plates (triplicate wells were used for each dilution). After 3-4 days, plates were observed for CPE and the cells rinsed once and fixed with 80% cold acetone. Fifty pl of mouse-source anti-BVDV monoclonal antibody was added to each well and plates were incubated at 37 °C for 1 hr. The primary antibody was discarded, and the plates were washed with phosphate buffered saline (PBS). Fifty pl of the secondary antibody conjugate (goat anti-mouse IgG conjugated to FITC) was added to the wells and plates were incubated at 37 °C for 1 hr. The antibody conjugate was discarded, and plates were washed with PBS. Plates were read under fluorescence microscopy and virus infectious titers were calculated according to the Reed and Muench method (based on CPE and immunofluorescence staining results) and expressed as the 50% tissue culture infective dose (TCID50)/ml.
[00467] The impact of boIFNX3-Tl 19-PEG30KDa on cytopathic, genotype 2 BVDV viral titers is presented in Table 10.
[00468] Table 10. Antiviral activity of boIFNX3-Tl 19-PEG30KDa against the cytopathic, genotype 2 strain 125 of BVDV.
[00469] These results confirm the antiviral activity of boIFNX3-Tl 19-PEG30KDa against a cytopathic, genotype 2 strain of BVDV. BoIFN/.3-T I 19-PEG30KDa completely prevented replication of the virus at concentrations > 250 ng/ml. These results combined with the results shown in Example 12 (e.g., Table 8) confirmed the ability of boIFNX3-Tl 19-PEG30KDa to
prevent replication of both cytopathic and non-cytopathic, genotype 1 and 2 BVDV strains which represent the clinically relevant BVDV pathogenic strains associated with diseases of cattle.
[00470] Example 14: In Vitro Antiviral Testing - Vesicular Stomatitis Virus New Jersey
[00471] Vesicular Stomatitis virus (VSV) is a member of the Rhabdoviridae family which causes vesicular lesions primarily in cattle, horses and occasionally, swine. The two most prevalent serotypes of VSV in the United States of America are serotypes VSV-Indiana (VSV-IN) and VSV- New Jersey (VSV-NJ). The antiviral activity of boIFNX3-Tl 19-PEG30KDa against a VSV-NJ was evaluated in vitro at the Plum Island Animal Disease Center (PIADC).
[00472] Serotype New Jersey of VSV provided by the Foreign Animal Disease Diagnostic Laboratory at PIADC was used for these experiments. Briefly, MDBK-t2 cells were seeded at 1.5 x 105 cells/well and incubated at 37 °C and 5% CO2. After 24 hr, cells were treated with serial 2- fold dilutions of boIFNX3-Tl 19-PEG30KDa in cell culture medium to generate concentrations from 0-5000 ng/ml and incubated at 37 °C and 5% CO2.
[00473] Cells were cultured in the presence of boIFNX3-T119-PEG30KDa that was prepared according to Example 8, for approximately 24 hr prior to viral inoculation. Culture medium containing boIFNX3-Tl 19-PEG30KDa was removed from each well of MDBK-t2 cells and replaced with cell culture medium containing VSV-NJ at a multiplicity of infection of 0.1. Plates were incubated at 37 °C in 5% CO2 for approximately 48 hr, and VSV-NJ titers were determined using a tetrazolium dye (MTT) reduction assay.
[00474] The impact of boIFNX3-Tl 19-PEG30KDa on VSV-NJ viral titers is presented in Table 11.
[00475] Table 11. Antiviral activity of boIFNX3-Tl 19-PEG30KDa against the New Jersey strain of VSV.
[00476] The data indicated that boIFNX3-T119-PEG30KDa completely inhibited in vitro replication of VSV-NJ at 5000 ng/ml. BoIFN/3-T I 19-PEG30KDa reduced in vitro replication of VSV-NJ by > 97% at concentrations > 1250 ng/ml. These results indicated that boIFNX3-Tl 19- PEG30KDa exhibits antiviral activity against VSV-NJ associated with vesicular disease in cattle.
[00477] Example 15: In Vitro Antiviral Testing - Foot and Mouth Disease Virus
[00478] The antiviral activity of boIFN/.3-T I 19-PEG30KDa against FMDV was evaluated in vitro at PI ADC.
[00479] Baby Hamster kidney cells strain 21 (BHK-21; ATCC CCL-10) were used to propoagate South African Territories (SAT)l of FMDV that was used for these experiments. Briefly, MDBK cells were seeded at 1.5xl05 cells/well and incubated at 37 °C and 5% CO2. After 24 hr, cells were treated with serial 2-fold dilutions of boIFNX3-T119-PEG30KDa in cell culture medium to generate concentrations from 0-157 ng/ml and incubated at 37 °C and 5% CO2.
[00480] Cells were cultured in the presence of boIFN/A-T I 19-PEG30KDa for approximately 24 hr prior to viral inoculation. Culture medium containing boIFN/A-T I 19-PEG30KDa was removed from each well of MDBK cells and replaced with cell culture medium containing FMDV SAT1 at a multiplicity of infection of 0.1. Plates were incubated at 37 °C in 5% CO2 for approximately 48 hr, and viral titers (TCID50/ml) were determined by endpoint dilution on BHKS cells.
[00481] The impact of boIFNX3-Tl 19-PEG30KDa on FMDV SAT1 viral titers is presented in Table 12.
[00483] The data indicated that boIFNX3-T119-PEG30KDa inhibited in vitro replication of FMDV SAT1 by > 99% at concentration > 39.06 ng/ml. These results showed that boIFNX3-Tl 19-PEG30KDa exhibits antiviral activity against FMDV SAT1 associated with vesicular disease in cattle.
[00484] Example 16: Pharmacokinetic (PK) study of boIFN/3-PEG in male Sprague-Dawley rats.
[00485] Five (5) groups of male Sprague-Dawley rats were administered a single subcutaneous bolus dose of PEGylated boIFN/A variants boIFNX3-N89-PEG30kDa, boIFNX3-T119- PEG30kDa, boIFNX3-S122-PEG30kDa, boIFNX3-G126-PEG30kDa or boIFNX3-R139-
PEG30kDa, and the plasma concentration was assessed over time. The study design is summarized in Table 13. The study included 9 time points (0, 3, 6, 24, 48, 72, 96, 168, and 264 hours). Bioanalysis of plasma samples were performed using ELISA assay. PK data analysis was performed using WinNonlin software. The results are summarized in FIG. 5, which depicts the mean plasma concentration versus time profiles, and Table 14. BoIFNX3-PEG exposure was similar (within 2-fold) following all treatments. In terms of AUC, exposure was lowest to highest in the following order: N89<G126<S122<T119<R139.
[00486] Table 13. PK study design of boIFNX3 -PEG variants in Sprague-Dawley rats
[00487] Table 14. BoIFNX3-PEG variant PK parameters in Sprague-Dawley rats
[00488] Example 17: Pharmacokinetic (PK) Analysis of boIFNZ.3-T I 19-PEG30kDa in Holstein- Friesian Calves.
[00489] Two (2) groups of Holstein-Friesian calves each consisting of 2 male/2 female individuals, aged 4-6 months were administered a single subcutaneous injection of boIFN/3-T I 19- PEG30kDa at doses of 75 and 150 pg/kg, and plasma concentration was assessed over time. The study design is summarized in Table 15. The study included 15 time points (0, 0.5, 1, 3, 6, 12, 24, 36, 48, 72, 96, 120, 168, 240, and 336 hours). Bioanalysis of plasma samples were performed using MSD platform utilizing electrochemiluminescence assay (ECLA). PK data analysis was performed using WinNonlin software. The results are summarized in FIG. 6, which depicts the mean plasma concentration versus time profiles, and Table 16. PEG30kDa-boIFNZ.3-T I 19 showed dose-proportional increases in Cmax and exposure. Terminal half-life was similar between groups, ranging from 65-69 hours.
[00492] Example 18: Differential scanning fluorimetry of boIFNZ.3-pAF and boIFNX3-pAF- PEG30kDa variants.
[00493] Differential scanning fluorimetry was performed using Bio-Rad Cl 000 Thermal Cycler RT-PCR instruments with the excitation and emission wavelengths set to 491 and 586 nm, respectively. Solution volumes were 25 pL in 96-well plates. Assay buffer for thermal stability measurements was 25 mM potassium phosphate (pH 6.5), 100 mM NaCl for pAF variants (FIG. 7 A), and 20 mM potassium phosphate (pH 6.5), lOOmM NaCl, 2.5% trehalose for PEG variants (FIG. 7B). The temperature scan rate was fixed at 1 °C/min. Protein and SYPRO orange (Thermo Fisher) concentrations were at 25 pM and 5X respectively. Data analysis was performed in BioRad CFX Manager 3.1 software. Tm values were determined by calculating the temperature derivative of the melting curve. The resulting derivative curve was processed with the peak fitting algorithm in Bio-Rad CFX Manager 3.1, applying a sigmoidal baseline and fitting the peak to determine the Tm and its standard error. The data shows the thermal stability of boIFN/3-pAF and
boIFNX3-pAF-PEG30kDa variants. Variants with thermal stability similar to wild type boIFNz.3 were preferred variants for further development.
[00494] Further non-limiting embodiments of the present disclosure are listed below.
[00495] 1. A conjugated protein comprising: an engineered bovine interferon Z.3 (boIFNZ.3) protein having an amino acid sequence comprising a non-natural amino acid; and a water-soluble polymer; wherein the water-soluble polymer is conjugated to the engineered boIFNz.3 protein.
[00496] 2 The conjugated protein of embodiment 1, wherein the water-soluble polymer is conjugated to the engineered boIFNz.3 protein via the non-natural amino acid.
[00497] 3. The conjugated protein of embodiment 1 or 2, wherein the water-soluble polymer is one or more water-soluble polymers, the non-natural amino acid is one or more non-natural amino acids, and each of the one or more water-soluble polymers is conjugated to the engineered boIFNz.3 protein via each of the one or more non-natural amino acids.
[00498] 4. The conjugated protein of embodiment 1 or 2, wherein the one or more water-soluble polymers is one water-soluble polymer, and the one or more non-natural amino acids is one non- natural amino acid.
[00499] 5. The conjugated protein of any one of embodiments 1 to 4, wherein the water-soluble polymer is selected from the group consisting of a carbohydrate, a polypeptide and a polyalkylene glycol; and derivatives thereof.
[00500] 6. The conjugated protein of embodiment 5, wherein the water-soluble polymer is a polyalkylene glycol or a derivative thereof.
[00501] 7. The conjugated protein of embodiment 6, wherein the polyalkylene glycol or a derivative thereof, wherein the polyalkylene glycol, or the derivative, thereof is a linear, branched or multiarm polyalkylene glycol, or a derivative thereof.
[00502] 8. The conjugated protein of embodiment 6 or 7, wherein the polyalkylene glycol, or the derivative thereof, is a polyethylene glycol (PEG), or a derivative thereof.
[00503] 9. The conjugated protein of any one of embodiments 1 to 8, wherein the non-natural amino acid is selected from the group consisting of 4-acetyl-L-phenylalanine (para-acetyl-L- phenylalanine (pAF)), 3-O-(N-acetyl-beta-D-glucosaminyl)-L-threonine, N4-( -N-Acetyl-D- glucosaminyl)-L-asparagine, O-allyl-L-tyrosine, alpha-N-acetylgalactosamine-O-L-serine, alpha- N-acetylgalactosamine-O-L-threonine, 2-aminooctanoic acid, 2-amino-L-phenylalanine, 3- amino-L-phenylalanine, 4-amino-L-phenylalanine, 2-amino-L-tyrosine, 3-amino-L-tyrosine, 4- azido-L-phenylalanine, 4-benzoyl-L-phenylalanine, (2,2-bipyridin-5yl)-L-alanine, 3-borono-L-
phenylalanine, 4-borono-L-phenylalanine, 4-bromo-L-phenylalanine, p-carboxymethyl-L- phenylalanine, 4-carboxy-L-phenylalanine, p-cyano-L-phenylalanine, 3,4-dihydroxy-L- phenylalanine (L-DOPA), 4-ethynyl-L-phenylalanine, 2-fluoro-L-phenylalanine, 3-fluoro-L- phenylalanine, 4-fluoro-L-phenylalanine, O-(3-O-D-galactosyl-N-acetyl-beta-D-galactosaminyl)- L-serine, L-homoglutamine, (8-hydroxyquinolin-3-yl)-L-alanine, 4-iodo-L-phenylalanine, 4- isopropyl-L-phenylalanine, O-i -propyl -L-tyrosine, 3-isopropyl-L-tyrosine, O-mannopyranosyl-L- serine, 2-m ethoxy -L-phenylalanine, 3 -methoxy -L-phenylalanine, 4-m ethoxy -L-phenylalanine, 3- methyl-L-phenylalanine, O-methyl-L-tyrosine, 3-(2-naphthyl)-L-alanine, 5-nitro-L-histidine, 4- nitro-L-histidine, 4-nitro-L-leucine, 2-nitro-L-phenylalanine, 3-nitro-L-phenylalanine, 4-nitro-L- phenylalanine, 4-nitro-L-tryptophan, 5-nitro-L-tryptophan, 6-nitro-L-tryptophan, 7-nitro-L- tryptophan, 2-nitro-L-tyrosine, 3-nitro-L-tyrosine, O-phospho-L-serine, O-phospho-L-tyrosine, 4- propargyloxy -L-phenylalanine, O-2-propyn-l-yl-L-tyrosine, 4-sulfo-L-phenylalanine and O- sulfo-L-tyrosine.
[00504] 10. The conjugated protein of any one of embodiments 1 to 9, wherein
(a) the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with:
(i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or
(ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine; or
(b) the engineered boIFNz.3 protein:
(i) has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or
(ii) has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the engineered boIFNz.3 protein amino acid sequence further comprises an N-terminal methionine.
[00505] 11. The conjugated protein of embodiment 10, wherein the engineered boIFNz.3 protein amino acid sequence shares at least 90% identity with SEQ ID NO: 18 or 19.
[00506] 12. The conjugated protein of any one of embodiments 6 to 11, wherein the polyalkylene glycol or the derivative thereof has a molecular weight of at least about 0.1 kDa.
[00507] 13. The conjugated protein of embodiment 12, wherein the polyalkylene glycol or the derivative thereof has a molecular weight of at most about 100 kDa.
[00508] 14. The conjugated protein of embodiment 12, wherein the polyalkylene glycol or the derivative thereof has a molecular weight of at most about 50 kDa.
[00509] 15. The conjugated protein of any one of embodiments 6 to 14, wherein the polyalkylene glycol or derivative thereof is a linear PEG or a derivative thereof.
[00510] 16. The conjugated protein of any one of embodiments 1 to 15, wherein the water-soluble polymer is conjugated to the engineered boIFNz.3 protein via a covalent linkage with the nonnatural amino acid.
[00511] 17. The conjugated protein of embodiment 16, wherein the covalent linkage comprises an amide, an ester, a thioester, a disulfide, an oxime, an imine, a pyrrolidine-2, 5-dione, a 1,2,3- triazole or a 1,4-dihydropyridazine, wherein the 1,2, 3 -triazole and the 1,4-dihydropyridazine are each optionally fused to an 8-membered ring.
[00512] 18. The conjugated protein of embodiment 17, wherein the covalent linkage comprises an oxime.
[00513] 19. The conjugated protein of any one of embodiments 1 to 18, wherein the non-natural amino acid is para-acetyl-L-phenylalanine.
[00514] 20. The conjugated protein of any one of embodiments 11 to 19, wherein the engineered boIFNz.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 18 or 19.
[00515] 21. The conjugated protein of embodiment 20, wherein the engineered boIFNZ.3 protein amino acid sequence shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 18 or 19.
[00516] 22. The conjugated protein of embodiment 20, wherein the engineered boIFNZ.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO: 18 or 19.
[00517] 23. The conjugated protein of embodiment 1 or 20, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 18.
[00518] 24. The conjugated protein of embodiment 1 or 20, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 19.
[00519] 25. The conjugated protein of embodiment 2, wherein: the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 18 or 19; the water-soluble polymer is a polyethylene glycol (PEG) or a derivative thereof; and the PEG or the derivative thereof is conjugated to the engineered boIFNz.3 protein via a covalent linkage with the non-natural amino acid.
[00520] 26. The conjugated protein of embodiment 25, wherein the non-natural amino acid is para-acetyl-L-phenylalanine.
[00521] 27. The conjugated protein of embodiment 25 or 26, wherein the PEG or the derivative thereof has a molecular weight of at least about 0.1 kDa and at most about 100 kDa.
[00522] 28. The conjugated protein of embodiment 27, wherein the PEG or the derivative thereof has a molecular weight of at most about 50 kDa.
[00523] 29. The conjugated protein of embodiment 28, wherein the PEG or the derivative thereof has a molecular weight within a range of about 10 kDa to about 50 kDa, about 20 kDa to about 40 kDa, or about 25 kDa to about 35 kDa.
[00524] 30. The conjugated protein of embodiment 29, wherein the PEG or the derivative thereof has a molecular weight of about 30 kDa.
[00525] 31. The conjugated protein of any one of embodiments 25 to 30, wherein the PEG or the derivative thereof is a linear PEG or a linear PEG derivative.
[00526] 32. The conjugated protein of any one of embodiments 25 to 31, wherein the covalent linkage comprises an oxime.
[00527] 33. The conjugated protein of embodiment 32, wherein, prior to the conjugation with the engineered boIFNz.3 protein, the water-soluble polymer is a PEG derivative comprising an aminooxy group.
[00528] 34. The conjugated protein of any one of embodiments 25 to 33, wherein the engineered boIFNz.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 18 or 19.
[00529] 35. The conjugated protein of any one of embodiments 25 to 33, wherein the engineered boIFNz.3 protein amino acid sequence shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 18 or 19.
[00530] 36. The conjugated protein of any one of embodiments 25 to 33, wherein the engineered boIFNz.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO: 18 or 19.
[00531] 37. The conjugated protein of any one of embodiments 25 to 33, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 18.
[00532] 38. The conjugated protein of any one of embodiments 25 to 33, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 19.
[00533] 39. The conjugated protein of any one of embodiments 1 to 38, wherein the engineered boIFNZ.3 protein is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA synthetase.
[00534] 40. The conjugated protein of any one of embodiments 1 to 39, wherein the non-natural amino acid is site-specifically incorporated into the protein.
[00535] 41. The conjugated protein of any one of embodiments 1 to 40, wherein the conjugated protein is characterized as having a plasma elimination half-life; and (i) the conjugated protein plasma elimination half-life is greater than the plasma elimination half-life of the unconjugated engineered boIFNz.3 protein; (ii) the conjugated protein plasma elimination half-life is greater than
the plasma elimination half-life of wild-type boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 1; (iii) the conjugated protein plasma elimination half-life is at least about 10 hours in a Sprague-Dawley rat after administration of a single subcutaneous dose of 0.25 mg/kg; or (iv) the conjugated protein plasma elimination half-life is at least about 50 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of 75 pg/kg.
[00536] 42. The conjugated protein of any one of embodiments 1 to 40, wherein (i) the conjugated protein is characterized as having a plasma elimination half-life in a mammal, wherein the conjugated protein plasma elimination half-life is at least about 10-fold greater than the plasma elimination half-life of the corresponding unconjugated boIFN/3 protein in the mammal, or (ii) the conjugated protein is characterized as having a plasma elimination half-life in a mammal; wherein the conjugated protein plasma elimination half-life is at least about 10-fold greater than the plasma elimination half-life of wild-type boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 1 in the mammal.
[00537] 43. The conjugated protein of any one of embodiments 1 to 40, wherein the conjugated protein is characterized as having a plasma elimination half-life in a mammal; wherein the conjugated protein plasma elimination half-life is at least about 10-fold greater than the plasma elimination half-life of an unconjugated IF NX protein or an unconjugated IFNy protein in the mammal.
[00538] 44. The conjugated protein of any one of embodiments 41 to 43, wherein the plasma elimination half-life is in a Sprague-Dawley rat after subcutaneous administration of a single dose of about 0.25 mg/kg.
[00539] 45. The conjugated protein of any one of embodiments 41 to 43, wherein the plasma elimination half-life is in a Holstein-Fresian calf after subcutaneous administration of a single dose of about 75 pg/kg.
[00540] 46. The conjugated protein of any one of embodiments 1 to 40 or 45, wherein the conjugated protein is characterized as having a plasma elimination half-life in a mammal, wherein the mammal is a Holstein-Fresian calf, and wherein the conjugated protein plasma elimination half-life is at least about is at least about 50-fold greater than the plasma elimination half-life of (i) the corresponding unconjugated boIFNZ.3 protein; (ii) wild-type boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 1; (iii) an unconjugated IFNk protein; or (iv) an unconjugated IFNy protein in the mammal.
[00541] 47. The conjugated protein of any one of embodiments 1 to 40, wherein the conjugated protein is characterized as having a plasma elimination half-life of at least about 40 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 75 pg/kg.
[00542] 48. The conjugated protein of any one of embodiments 1 to 40, wherein the conjugated protein is characterized as having a plasma elimination half-life of at least about 50 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 75 pg/kg.
[00543] 49. The conjugated protein of any one of embodiments 1 to 40, wherein the conjugated protein plasma elimination half-life is at least about 60 hours in a Holstein-Fresian calf after administration of a single subcutaneous dose of about 75 pg/kg.
[00544] 50. A pharmaceutical composition comprising a conjugated protein of any one of embodiments 1 to 49 and a pharmaceutically acceptable excipient or carrier.
[00545] 51. An engineered bovine interferon Z.3 (boIFNZ.3) protein, a) wherein the engineered boIFNz.3 optionally comprises a non-natural amino acid; and wherein the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with:
(i) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13; or
(ii) SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, wherein each of the foregoing sequences further comprises an N-terminal methionine; wherein the amino acid sequence shares less than 100% identity with SEQ ID NO: 1; or b) wherein the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, and which shares less than 100% identity with SEQ ID NO: 1; and optionally wherein the boIFNz.3 protein further comprises an N-terminal methionine; and wherein: the boIFNz.3 protein amino acid sequence comprises a non-natural amino acid; or the boIFNz.3 protein amino acid sequence does not comprise a non-natural amino acid.
[00546] 52. The engineered boIFNz.3 protein of embodiment 51, wherein a) the engineered boIFNZ.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 5, or with SEQ ID NO: 5 further comprising the N-terminal methionine or b) the engineered boIFNz.3 protein has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 5, and optionally further comprises an N-terminal methionine.
[00547] 53. The engineered boIFNz.3 protein of embodiment 52, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 5.
[00548] 54. The engineered boIFNz.3 protein of embodiment 52, wherein the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 5 further comprising the N-terminal methionine.
[00549] 55. The engineered boIFNZ.3 protein of embodiment 51, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 6.
[00550] 56. The engineered boIFNz.3 protein of embodiment 51, wherein the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 6 further comprising the N-terminal methionine.
[00551] 57. The engineered boIFNz.3 protein of embodiment 51 or 52, wherein the engineered boIFNZ.3 protein amino acid sequence does not comprise a non-natural amino acid; or wherein the engineered boIFNz.3 protein amino acid sequence comprises only amino acids selected from the group consisting of the 20 canonical amino acids.
[00552] 58. The engineered boIFNz.3 protein of embodiment 51 or 52, wherein the boIFNz.3 protein amino acid sequence comprises a non-natural amino acid, optionally wherein: a) the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 5, in which a non- natural amino acid is substituted for the amino acid at one or more position of that sequence, preferably a non-natural amino acid is substituted for the amino acid at one position of that sequence; b) the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 5 further comprising the N-terminal methionine, in which a non-natural amino acid is substituted for the amino acid at one or more position of that sequence, preferably a non-natural amino acid is substituted for the amino acid at one position of that sequence; c) the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 6, in which a non- natural amino acid is substituted for the amino acid at one or more position of that sequence, preferably a non-natural amino acid is substituted for the amino acid at one position of that sequence; or d) the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 6 further comprising the N-terminal methionine, in which a non-natural amino acid is substituted for the amino acid at one or more position of that sequence, preferably a non-natural amino acid is substituted for the amino acid at one position of that sequence.
[00553] 59. The engineered boIFNz.3 protein of embodiment 51 or 58, wherein the non-natural amino acid is selected from the group consisting of 4-acetyl-L-phenylalanine (para-acetyl-L- phenylalanine (pAF)), 3-O-(N-acetyl-beta-D-glucosaminyl)-L-threonine, N4-( -N-Acetyl-D- glucosaminyl)-L-asparagine, O-allyl-L-tyrosine, alpha-N-acetylgalactosamine-O-L-serine, alpha- N-acetylgalactosamine-O-L-threonine, 2-aminooctanoic acid, 2-amino-L-phenylalanine, 3- amino-L-phenylalanine, 4-amino-L-phenylalanine, 2-amino-L-tyrosine, 3-amino-L-tyrosine, 4- azido-L-phenylalanine, 4-benzoyl-L-phenylalanine, (2,2-bipyridin-5yl)-L-alanine, 3-borono-L-
phenylalanine, 4-borono-L-phenylalanine, 4-bromo-L-phenylalanine, p-carboxymethyl-L- phenylalanine, 4-carboxy-L-phenylalanine, p-cyano-L-phenylalanine, 3,4-dihydroxy-L- phenylalanine (L-DOPA), 4-ethynyl-L-phenylalanine, 2-fluoro-L-phenylalanine, 3-fluoro-L- phenylalanine, 4-fluoro-L-phenylalanine, O-(3-O-D-galactosyl-N-acetyl-beta-D-galactosaminyl)- L-serine, L-homoglutamine, (8-hydroxyquinolin-3-yl)-L-alanine, 4-iodo-L-phenylalanine, 4- isopropyl-L-phenylalanine, O-i -propyl -L-tyrosine, 3-isopropyl-L-tyrosine, O-mannopyranosyl-L- serine, 2-m ethoxy -L-phenylalanine, 3 -methoxy -L-phenylalanine, 4-m ethoxy -L-phenylalanine, 3- methyl-L-phenylalanine, O-methyl-L-tyrosine, 3-(2-naphthyl)-L-alanine, 5-nitro-L-histidine, 4- nitro-L-histidine, 4-nitro-L-leucine, 2-nitro-L-phenylalanine, 3-nitro-L-phenylalanine, 4-nitro-L- phenylalanine, 4-nitro-L-tryptophan, 5-nitro-L-tryptophan, 6-nitro-L-tryptophan, 7-nitro-L- tryptophan, 2-nitro-L-tyrosine, 3-nitro-L-tyrosine, O-phospho-L-serine, O-phospho-L-tyrosine, 4- propargyloxy -L-phenylalanine, O-2-propyn-l-yl-L-tyrosine, 4-sulfo-L-phenylalanine and O- sulfo-L-tyrosine.
[00554] 60. The engineered boIFNZ.3 protein of embodiment 51, 58 or 59, wherein the nonnatural amino acid is para-acetyl-L-phenylalanine.
[00555] 61. The engineered boIFNZ.3 protein of any one of embodiments 51, 58, 59 or 60, wherein the non-natural amino acid is at a position selected from the group consisting of G15, T58, Q65, N89, T119, S122, R123, G126, R139 and D169, wherein the position number is based on the wild type boIFNZ.3 protein amino acid sequence of SEQ ID NO: 1.
[00556] 62. The engineered boIFNZ.3 protein of embodiment 61, wherein the non-natural amino acid is at position T119, wherein the position number is based on the wild type boIFNZ.3 protein amino acid sequence of SEQ ID NO: 1.
[00557] 63. The engineered boIFNZ.3 protein of embodiment 62, wherein the non-natural amino acid at position T119 is para-acetyl-L-phenylalanine.
[00558] 64. The engineered boIFNZ.3 protein of any one of embodiments 51 to 63, wherein the engineered boIFNZ.3 protein is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA.
[00559] 65. The engineered boIFNZ.3 protein of any one of embodiments 51 to 64, wherein the non-natural amino acid is site-specifically incorporated.
[00560] 66. A nucleic acid having the nucleotide sequence of SEQ ID NO: 27.
[00561] 67. A vector comprising a nucleic acid encoding any one of SEQ ID NOs: 2 to 24.
[00562] 68. A vector comprising a nucleic acid encoding SEQ ID NO: 5.
[00563] 69. A vector comprising a nucleic acid encoding SEQ ID NO: 18.
[00564] 70. A vector comprising a nucleic acid encoding SEQ ID NO: 19.
[00565] 71. An engineered bovine interferon Z.3 (boIFNz.3) protein comprising a non-natural amino acid; wherein (a) the engineered boIFNz.3 protein has an amino acid sequence that shares at least 90% identity with:
(i) SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24, or
(ii) SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, wherein each of the foregoing sequences further comprises an N-terminal methionine; or
(b) the engineered boIFNz.3 protein
(i) has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24, or
(ii) has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNz.3 protein amino acid sequence further comprises an N-terminal methionine.
[00566] 72. The engineered boIFNz.3 protein of embodiment 71, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 14, or has the amino acid sequence of SEQ ID NO: 14 further comprising the N-terminal methionine.
[00567] 73. The engineered boIFNz.3 protein of embodiment 71, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 15, or has the amino acid sequence of SEQ ID NO: 15 further comprising the N-terminal methionine.
[00568] 74. The engineered boIFNz.3 protein of embodiment 71, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 16, or has the amino acid sequence of SEQ ID NO: 16 further comprising the N-terminal methionine.
[00569] 75. The engineered boIFNz.3 protein of embodiment 71, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 17, or has the amino acid sequence of SEQ ID NO: 17 further comprising the N-terminal methionine.
[00570] 76. The engineered boIFNz.3 protein of embodiment 71, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 18 or SEQ ID NO: 19.
[00571] 77. The engineered boIFNz.3 protein of embodiment 71, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 20, or has the amino acid sequence of SEQ ID NO: 20 further comprising the N-terminal methionine.
[00572] 78. The engineered boIFNz.3 protein of embodiment 71, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 21, or has the amino acid sequence of SEQ ID NO: 21 further comprising the N-terminal methionine.
[00573] 79. The engineered boIFNZ.3 protein of embodiment 71, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 22, or has the amino acid sequence of SEQ ID NO: 22 further comprising the N-terminal methionine.
[00574] 80. The engineered boIFNz.3 protein of embodiment 71, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 23, or has the amino acid sequence of SEQ ID NO: 23 further comprising the N-terminal methionine.
[00575] 81. The engineered boIFNz.3 protein of embodiment 71, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 24, or has the amino acid sequence of SEQ ID NO: 24 further comprising the N-terminal methionine.
[00576] 82. The engineered boIFNz.3 protein of embodiment 71, wherein the engineered boIFNz.3 protein amino acid sequence shares at least 95%, at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 18.
[00577] 83. The engineered boIFNz.3 protein of embodiment 71, wherein the engineered boIFNz.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO: 18.
[00578] 84. The engineered boIFNz.3 protein of embodiment 71, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 18.
[00579] 85. The engineered boIFNz.3 protein of embodiment 71, wherein the engineered boIFNZ.3 protein amino acid sequence shares at least 95% or at least 98% identity with SEQ ID NO: 19.
[00580] 86. The engineered boIFNz.3 protein of embodiment 71, wherein the engineered boIFNz.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO: 19.
[00581] 87. The engineered boIFNz.3 protein of embodiment 71, wherein the engineered boIFNz.3 protein has the amino acid sequence SEQ ID NO: 19.
[00582] 88. The engineered boIFNz.3 protein of any one of embodiments 71 to 87, wherein the non-natural amino acid is selected from the group consisting of 4-acetyl-L-phenylalanine (para- acetyl-L-phenylalanine (pAF)), 3-O-(N-acetyl-beta-D-glucosaminyl)-L-threonine, N4-( -N- Acetyl-D-glucosaminyl)-L-asparagine, O-allyl-L-tyrosine, alpha-N-acetylgalactosamine-O-L- serine, alpha-N-acetylgalactosamine-O-L-threonine, 2-aminooctanoic acid, 2-amino-L- phenylalanine, 3-amino-L-phenylalanine, 4-amino-L-phenylalanine, 2-amino-L-tyrosine, 3- amino-L-tyrosine, 4-azido-L-phenylalanine, 4-benzoyl-L-phenylalanine, (2,2-bipyridin-5yl)-L- alanine, 3-borono-L-phenylalanine, 4-borono-L-phenylalanine, 4-bromo-L-phenylalanine, p- carboxymethyl-L-phenylalanine, 4-carboxy-L-phenylalanine, p-cyano-L-phenylalanine, 3,4- dihydroxy-L-phenylalanine (L-DOPA), 4-ethynyl-L-phenylalanine, 2-fluoro-L-phenylalanine, 3- fluoro-L-phenylalanine, 4-fluoro-L-phenylalanine, O-(3-O-D-galactosyl-N-acetyl-beta-D-
galactosaminyl)-L-serine, L-homoglutamine, (8-hydroxyquinolin-3-yl)-L-alanine, 4-iodo-L- phenylalanine, 4-isopropyl-L-phenylalanine, O-i-propyl-L-tyrosine, 3-isopropyl-L-tyrosine, O- mannopyranosyl-L-serine, 2-methoxy-L-phenylalanine, 3 -m ethoxy -L-phenylalanine, 4-methoxy- L-phenylalanine, 3-methyl-L-phenylalanine, O-methyl-L-tyrosine, 3-(2-naphthyl)-L-alanine, 5- nitro-L-histidine, 4-nitro-L-histidine, 4-nitro-L-leucine, 2-nitro-L-phenylalanine, 3-nitro-L- phenylalanine, 4-nitro-L-phenylalanine, 4-nitro-L-tryptophan, 5-nitro-L-tryptophan, 6-nitro-L- tryptophan, 7 -nitro-L-try ptophan, 2-nitro-L-tyrosine, 3-nitro-L-tyrosine, O-phospho-L-serine, O- phospho-L-tyrosine, 4-propargyloxy -L-phenylalanine, O-2-propyn-l-yl-L-tyrosine, 4-sulfo-L- phenylalanine and O-sulfo-L-tyrosine.
[00583] 89. The engineered boIFNZ.3 protein of embodiment 88, wherein the non-natural amino acid is para-acetyl-L-phenylalanine.
[00584] 90. The engineered boIFNZ.3 protein of any one of embodiments 71 to 89, wherein the engineered boIFNZ.3 protein is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA, and the non-natural amino acid is site-specifically incorporated.
[00585] 91. A prophylactic method of treating a disease or condition in a mammal, the method comprising administering to the mammal a conjugated protein of any one of embodiments 1 to 49 or a pharmaceutical composition of embodiment 50.
[00586] 92. The method of embodiment 91, wherein the mammal is a ruminant mammal.
[00587] 93. The method of embodiment 92, wherein the ruminant mammal is bovine.
[00588] 94. The method of embodiment 91, 92 or 93, wherein the disease or condition is a bovine respiratory disease.
[00589] 95. The method of any one of embodiments 91 to 94, wherein the disease or condition is associated with bovine viral diarrhea virus (BVDV), parainfluenza-3 (PI3) or bovine respiratory syncytial virus (BRSV); or a combination thereof.
[00590] 96. The method of embodiment 91, 92 or 93, wherein the disease or condition is foot and mouth disease.
[00591] 97. The method of any one of embodiments 91 to 93 or 96, wherein the disease or condition is associated with foot and mouth disease virus (FMDV).
[00592] 98. The method of any one of embodiments 91 to 97, wherein the method further comprises administering a vaccine.
[00593] 99. The method of embodiment 98, wherein the disease or condition is foot and mouth disease, and the vaccine is suitable for the treatment of FMDV.
[00594] 100. The method of embodiment 98, wherein the disease or condition is bovine respiratory disease, and the vaccine is suitable for the treatment of bovine viral diarrhea virus (BVDV), parainfluenza-3 (PI3) or bovine respiratory syncytial virus (BRSV); or a combination thereof.
[00595] 101. The method of embodiment 94, 95 or 100, wherein the method further comprises administering an antibiotic to the ruminant mammal.
[00596] 102. The method of embodiment 101, wherein the antibiotic is a penicillin, a cephalosporin, a macrolide, a tetracycline antibiotic or a fluoroquinolone antibiotic; or a combination thereof.
[00597] 103. The method of embodiment 102, wherein the antibiotic is a cephalosporin, optionally, wherein the cephalosporin is selected from the group consisting of cefachlor, cefadroxil, cefamandole, cefazolin, cefepime, cefoperazone, ceforanide, cefotaxime, cefotiam, cefpodoxime, ceftazidime, cefovecin, cefoxitin (a cephamycin), cefsulodin, ceftiofur, ceftriaxone, cefuroxime, cephalexin, cephaloridine, cephalothin, cephapirin and cephradine; and combinations thereof.
[00598] 104. The method of embodiment 102, wherein the antibiotic is a macrolide, optionally, wherein the macrolide is selected from the group consisting of azithromycin, erythromycin, erythromycin A, erythromycin B, erythromycin C, erythromycin D, erythromycin E, gamithromycin, tilmicosin, tulathromycin, tildipirosin and tylosin; and combinations thereof.
[00599] 105. A pharmaceutical composition comprising a conjugated protein of any one of embodiments 1 to 49 and an antibiotic for the treatment of a bovine respiratory disease.
[00600] 106. A method of inducing systemic antiviral activity in a mammal, the method comprising administering to the mammal a conjugated protein of any one of embodiments 1 to 49 or a pharmaceutical composition of embodiment 50.
[00601] 107. The method of embodiment 106, wherein the mammal is a ruminant mammal.
[00602] 108. The method of embodiment 107, wherein the ruminant mammal is bovine.
[00603] 109. The method of embodiment 106, 107 or 108, wherein the method further comprises administering a vaccine, optionally a vaccine for a virus associated with FMDV or BRD.
[00604] 110. The method of embodiment 109, wherein the vaccine is suitable for the treatment of foot and mouth disease virus (FMDV).
[00605] 111. The method of embodiment 110, wherein the vaccine is a FMD antigen, an inactivated whole FMDV antigen, a modified live vaccine or an adenovirus type 5 (Ad5) FMD (Ad5-FMD) vaccine.
[00606] 112. The method of embodiment 110 or 111, wherein the method induces (i) anti- FMDV activity; (ii) adjuvanted adaptive immune responses against FMDV; and/or (iii) expression of interferon-stimulated genes correlated with systemic control of viral replication in an animal susceptible to FMDV in vivo; thereby delaying, reducing severity and/or preventing foot and mouth disease.
[00607] 113. The method of any one of embodiments 91 to 97, wherein the method provides the mammal with protection against infection by a virus associated with the disease or condition.
[00608] 114. The method of embodiment 113, wherein disease or condition is FMD, and the virus is a FMD virus (FMDV).
[00609] 115. The method of embodiment 113 or 114, wherein:
(i) the protection begins within about 1 day after the administration of the conjugated protein;
(ii) the protection begins within about 2 days after the administration of the conjugated protein; or
(iii) the protection begins within about 3 days after the administration of the conjugated protein. [00610] 116. The method of embodiment 114 or 115, wherein the method comprises administration of a FMDV vaccine, and the protection begins prior to the induction of protective antibody titers in the mammal, wherein the protective antibody titers are induced by the administration of the vaccine.
[00611] 117. The method of embodiment 114 or 115, wherein the method comprises administration of a FMDV vaccine, and the protection begins prior to vaccine-induced onset of adaptive immunity in the mammal.
[00612] 118. The method of any one of embodiments 113 to 117, wherein the protection is characterized by an absence of viremia.
[00613] 119. The method of any one of embodiments 113 to 118, wherein the protection is characterized by an absence of a clinical FMD symptom.
[00614] 120. The method of embodiment 119, wherein the absence of a clinical FMD symptom is an absence of fever, an absence of vesicular lesions (blisters, ulcers and/or sores) in the mouth, tongue, muzzle, nostrils, teats and/or feet, and/or an absence of lameness.
[00615] 121. The method of embodiment 113, wherein the disease or condition is BRD.
[00616] 122. The method of embodiment 121, wherein the protection is protection against infection by a virus associated with BRD.
[00617] 123. The method of embodiment 122, wherein the virus associated with BRD is selected from the group consisting of bovine viral diarrhea virus (BVDV), bovine respiratory syncytial virus (BRSV), a bovine coronavirus and a parainfluenza-3 virus (PI-3); and combinations thereof.
[00618] 124. The method of embodiment 121, 122 or 123, wherein the protection is characterized by an absence of viremia.
[00619] 125. The method of any one of embodiments 121 to 124, wherein the protection is characterized by an absence of a clinical BRD symptom.
[00620] 126. The method of any one of embodiments 121 to 125, wherein the protection begins within about 1 day after the administration of the conjugated protein.
[00621] 127. The method of any one of embodiments 121 to 126, wherein the method comprises administration of a vaccine against a virus associated with BRD, and the protection begins prior to the induction of protective antibody titers in the mammal, wherein the protective antibody titers are induced by the administration of the vaccine.
[00622] 128. The method of embodiment 121 to 126, wherein the method comprises administration of a vaccine against a virus associated with BRD, and the protection begins prior to vaccine-induced onset of adaptive immunity in the mammal.
[00623] 129. The method of any one of embodiments 113 to 128, wherein the protection occurs over a period of time of at least about 3 days.
[00624] 130. The method of embodiment 129, wherein the protection occurs over a period of time of at least about 5 days.
[00625] 131. The method of any one of embodiments 113 to 128, wherein the protection occurs over a period of time of about 1 day, about 2 days, about 3 days, about 4 days, about 5 days, about 6 days or about 7 days.
[00626] 132. The method of any one of embodiments 113 to 128, wherein the protection occurs over a period of time of about 1 day to about 5 days.
[00627] 133. The method of any one of embodiments 113 to 128, wherein the protection occurs over a period of time of at least about 7 days.
[00628] 134. The method of any one of embodiments 91 to 104 and 106-133, wherein the conjugated protein is administered to a ruminant mammal at a dose within a range of about 50 pg/kg to about 500 pg/kg of the body weight of the ruminant mammal.
[00629] 135. The method of embodiment 134, wherein the dose is within a range of about 75 pg/kg to about 300 pg/kg.
[00630] 136. The method of embodiment 134, wherein the dose is within a range of about 75 pg/kg to about 200 pg/kg.
[00631] 137. The method of embodiment 134, wherein the dose is within a range of about 75 pg/kg to about 150 pg/kg.
[00632] 138. The method of embodiment 134, wherein the dose is within a range of about 100 gg/kg to about 200 gg/kg.
[00633] 139. The method of embodiment 134, wherein the dose is about 75 gg/kg.
[00634] 140. The method of embodiment 134, wherein the dose is about 125 gg/kg.
[00635] While certain embodiments of the inventions have been described, these embodiments have been presented by way of example only and are not intended to limit the scope of the disclosure. Indeed, the novel compositions, methods and systems described herein may be embodied in a variety of other forms. Furthermore, various omissions, substitutions and changes in the compositions, systems and methods described herein may be made without departing from the spirit of the disclosure. The accompanying claims and their equivalents are intended to cover such forms or modifications as would fall within the scope and spirit of the disclosure. Accordingly, the scope of the present inventions is defined only by reference to the appended claims.
[00636] Features, materials, characteristics, or groups described in conjunction with a particular aspect, embodiment, or example are to be understood to be applicable to any other aspect, embodiment or example described in this section or elsewhere in this specification unless incompatible therewith. All of the features disclosed in this specification (including any accompanying claims, abstract and drawings), and/or all of the compositions, steps of any method or process so disclosed, may be combined in any combination, except combinations where at least some of such features and/or steps are mutually exclusive. The protection is not restricted to the details of any foregoing embodiments. The protection extends to any novel one, or any novel combination, of the features disclosed in this specification (including any accompanying claims, abstract and drawings), or to any novel one, or any novel combination, of the compositions, steps of any method or process so disclosed.
[00637] Furthermore, certain features that are described in this disclosure in the context of separate implementations can also be implemented in combination in a single implementation. Conversely, various features that are described in the context of a single implementation can also be implemented in multiple implementations separately or in any suitable subcombination. Although features may be described above as acting in certain combinations, one or more features from a claimed combination can, in some cases, be excised from the combination, and the combination may be claimed as a subcombination or variation of a subcombination.
[00638] The features and attributes of the specific embodiments disclosed above may be combined in different ways to form additional embodiments, all of which fall within the scope of the present disclosure. Also, the separation of various system components in the implementations
described above should not be understood as requiring such separation in all implementations, and it should be understood that the described components and systems can generally be integrated together in a single product or packaged into multiple products.
[00639] For purposes of this disclosure, certain aspects, advantages, and novel features are described herein. Not necessarily all such advantages may be achieved in accordance with any particular embodiment. Thus, for example, those skilled in the art will recognize that the disclosure may be embodied or carried out in a manner that achieves one advantage or a group of advantages as taught herein without necessarily achieving other advantages as may be taught or suggested herein.
[00640] Conditional language, such as "can," "could," "might," or "rnay," unless specifically stated otherwise, or otherwise understood within the context as used, is generally intended to convey that certain embodiments include, while other embodiments do not include, certain features, elements, and/or steps. Thus, such conditional language is not generally intended to imply that features, elements, and/or steps are in any way required for one or more embodiments or that one or more embodiments necessarily include logic for deciding, with or without user input or prompting, whether these features, elements, and/or steps are included or are to be performed in any particular embodiment.
[00641] Conjunctive language such as the phrase "at least one of X, Y, and Z," unless specifically stated otherwise, is otherwise understood with the context as used in general to convey that an item, term, etc. may be either X, Y, or Z. Thus, such conjunctive language is not generally intended to imply that certain embodiments require the presence of at least one of X, at least one of Y, and at least one of Z.
[00642] Language of degree used herein, such as the terms "approximately," "about," "generally," and "substantially" as used herein represent a value, amount, or characteristic close to the stated value, amount, or characteristic that still performs a desired function or achieves a desired result. For example, the terms "approximately", "about", "generally," and "substantially" may refer to an amount that is within less than 10% of, within less than 5% of, within less than 1% of: within less than 0.1% of, and within less than 0.01% of the stated amount.
[00643] The scope of the present disclosure is not intended to be limited by the specific disclosures of preferred embodiments in this section or elsewhere in this specification, and may be defined by claims as presented in this section or elsewhere in this specification or as presented in the future. The language of the claims is to be interpreted broadly based on the language employed in the claims and not limited to the examples described in the present specification or during the prosecution of the application, which examples are to be construed as non-exclusive.
Claims
1. A conjugated protein comprising: an engineered bovine interferon Z.3 (boIFNz.3) protein having an amino acid sequence comprising a non-natural amino acid; and a water-soluble polymer; wherein the water-soluble polymer is conjugated to the engineered boIFNz.3 protein.
2. The conjugated protein of claim 1, wherein the water-soluble polymer is conjugated to the engineered boIFNz.3 protein via the non-natural amino acid.
3. The conjugated protein of claim 1 or 2, wherein the water-soluble polymer is one or more water- soluble polymers, the non-natural amino acid is one or more non-natural amino acids, and each of the one or more water-soluble polymers is conjugated to the engineered boIFNz.3 protein via each of the one or more non-natural amino acids.
4. The conjugated protein of claim 1 or 2, wherein the one or more water-soluble polymers is one water-soluble polymer, and the one or more non-natural amino acids is one non-natural amino acid.
5. The conjugated protein of any one of claims 1 to 4, wherein the water-soluble polymer is selected from the group consisting of a carbohydrate, a polypeptide and a polyalkylene glycol; and derivatives thereof.
6. The conjugated protein of claim 5, wherein the water-soluble polymer is a polyalkylene glycol or a derivative thereof.
7. The conjugated protein of claim 6, wherein the polyalkylene glycol, or the derivative thereof, is a linear, branched or multiarm polyalkylene glycol, or a derivative thereof.
8. The conjugated protein of claim 6 or 7, wherein the polyalkylene glycol, or the derivative thereof, is a polyethylene glycol (PEG), or a derivative thereof.
9. The conjugated protein of any one of claims 1 to 8, wherein the non-natural amino acid is selected from the group consisting of 4-acetyl-L-phenylalanine (para-acetyl-L-phenylalanine (pAF)), 3-O-(N-acetyl-beta-D-glucosaminyl)-L-threonine, N4-(P-N-Acetyl-D-glucosaminyl)-L- asparagine, O-allyl-L-tyrosine, alpha-N-acetylgalactosamine-O-L-serine, alpha-N- acetylgalactosamine-O-L-threonine, 2-aminooctanoic acid, 2-amino-L-phenylalanine, 3-amino-L- phenylalanine, 4-amino-L-phenylalanine, 2-amino-L-tyrosine, 3-amino-L-tyrosine, 4-azido-L-
phenylalanine, 4-b enzoy 1 -L-pheny 1 al anine, (2,2-bipyridin-5yl)-L-alanine, 3-borono-L- phenylalanine, 4-borono-L-phenylalanine, 4-bromo-L-phenylalanine, p-carboxymethyl-L- phenylalanine, 4-carboxy-L-phenylalanine, p-cyano-L-phenylalanine, 3,4-dihydroxy-L- phenylalanine (L-DOPA), 4-ethynyl-L-phenylalanine, 2-fluoro-L-phenylalanine, 3-fluoro-L- phenylalanine, 4-fluoro-L-phenylalanine, O-(3-O-D-galactosyl-N-acetyl-beta-D-galactosaminyl)-
L-serine, L-homoglutamine, (8-hydroxyquinolin-3-yl)-L-alanine, 4-iodo-L-phenylalanine, 4- isopropyl-L-phenylalanine, O-i -propyl -L-tyrosine, 3-isopropyl-L-tyrosine, O-mannopyranosyl-L- serine, 2-m ethoxy -L-phenylalanine, 3 -methoxy -L-phenylalanine, 4-m ethoxy -L-phenylalanine, 3- methyl-L-phenylalanine, O-methyl-L-tyrosine, 3-(2-naphthyl)-L-alanine, 5-nitro-L-histidine, 4- nitro-L-histidine, 4-nitro-L-leucine, 2-nitro-L-phenylalanine, 3-nitro-L-phenylalanine, 4-nitro-L- phenylalanine, 4-nitro-L-tryptophan, 5-nitro-L-tryptophan, 6-nitro-L-tryptophan, 7-nitro-L- tryptophan, 2-nitro-L-tyrosine, 3-nitro-L-tyrosine, O-phospho-L-serine, O-phospho-L-tyrosine, 4- propargyloxy -L-phenylalanine, O-2-propyn-l-yl-L-tyrosine, 4-sulfo-L-phenylalanine and O- sulfo-L-tyrosine.
10. The conjugated protein of any one of claims 1 to 9, wherein the engineered boIFNZ.3 protein
(i) has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24; or
(ii) has an amino acid sequence that shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the engineered boIFNZ.3 protein amino acid sequence further comprises an N-terminal methionine.
11. The conjugated protein of claim 10, wherein the engineered boIFNZ.3 protein amino acid sequence shares at least 90% identity with SEQ ID NO: 18 or 19.
12. The conjugated protein of any one of claims 6 to 11, wherein the polyalkylene glycol or the derivative thereof has a molecular weight of at least about 0.1 kDa.
13. The conjugated protein of claim 12, wherein the polyalkylene glycol or the derivative thereof has a molecular weight of at most about 100 kDa.
14. The conjugated protein of claim 12, wherein the polyalkylene glycol or the derivative thereof has a molecular weight of at most about 50 kDa.
15. The conjugated protein of any one of claims 6 to 14, wherein the polyalkylene glycol or derivative thereof is a linear PEG or a derivative thereof.
16. The conjugated protein of any one of claims 1 to 15, wherein the water-soluble polymer is conjugated to the engineered boIFNz.3 protein via a covalent linkage with the non-natural amino acid.
17. The conjugated protein of claim 16, wherein the covalent linkage comprises an amide, an ester, a thioester, a disulfide, an oxime, an imine, a pyrrolidine-2, 5-dione, a 1,2, 3 -triazole or a 1,4- dihydropyridazine, wherein the 1,2,3-triazole and the 1,4-dihydropyridazine are each optionally fused to an 8-membered ring.
18. The conjugated protein of claim 17, wherein the covalent linkage comprises an oxime.
19. The conjugated protein of any one of claims 1 to 18, wherein the non-natural amino acid is para-acetyl -L-phenylalanine.
20. The conjugated protein of any one of claims 11 to 19, wherein the engineered boIFNZ.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 18 or 19.
21. The conjugated protein of claim 20, wherein the engineered boIFNz.3 protein amino acid sequence shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 18 or 19.
22. The conjugated protein of claim 20, wherein the engineered boIFNz.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO: 18 or 19.
23. The conjugated protein of claim 1 or 20, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 18.
24. The conjugated protein of claim 1 or 20, wherein the engineered boIFNz.3 protein has the amino acid sequence of SEQ ID NO: 19.
25. The conjugated protein of claim 2, wherein: the engineered boIFNz.3 protein amino acid sequence shares at least 90% identity with SEQ ID NO: 18 or 19; the water-soluble polymer is a polyethylene glycol (PEG) or a derivative thereof; and the PEG or the derivative thereof is conjugated to the engineered boIFNz.3 protein via a covalent linkage with the non-natural amino acid.
26. The conjugated protein of claim 25, wherein the non-natural amino acid is para-acetyl-L- phenylalanine.
27. The conjugated protein of claim 25 or 26, wherein the PEG or the derivative thereof has a molecular weight of at least about 0.1 kDa and at most about 100 kDa.
28. The conjugated protein of claim 27, wherein the PEG or the derivative thereof has a molecular weight of at most about 50 kDa.
29. The conjugated protein of claim 28, wherein the PEG or the derivative thereof has a molecular weight within a range of about 10 kDa to about 50 kDa, about 20 kDa to about 40 kDa, or about 25 kDa to about 35 kDa.
30. The conjugated protein of claim 29, wherein the PEG or the derivative thereof has a molecular weight of about 30 kDa.
31. The conjugated protein of any one of claims 25 to 30, wherein the PEG or the derivative thereof is a linear PEG or a linear PEG derivative.
32. The conjugated protein of any one of claims 25 to 31, wherein the covalent linkage comprises an oxime.
33. The conjugated protein of claim 32, wherein, prior to the conjugation with the engineered boIFNZ.3 protein, the water-soluble polymer is a PEG derivative comprising an aminooxy group.
34. The conjugated protein of any one of claims 25 to 33, wherein the engineered boIFNZ.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 18 or 19.
35. The conjugated protein of any one of claims 25 to 33, wherein the engineered boIFNZ.3 protein amino acid sequence shares at least 96%, at least 97% or at least 98% identity with SEQ ID NO: 18 or 19.
36. The conjugated protein of any one of claims 25 to 33, wherein the engineered boIFNZ.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO: 18 or 19.
37. The conjugated protein of any one of claims 25 to 33, wherein the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 18.
38. The conjugated protein of any one of claims 25 to 33, wherein the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 19.
39. The conjugated protein of any one of claims 1 to 38, wherein the engineered boIFNZ.3 protein is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA synthetase.
40. The conjugated protein of any one of claims 1 to 39, wherein the non-natural amino acid is site-specifically incorporated into the protein.
41. The conjugated protein of any one of claims 1 to 40, wherein the conjugated protein is characterized as having a plasma elimination half-life in a mammal, wherein the conjugated protein plasma elimination half-life is at least about 10-fold greater than the plasma elimination half-life of the corresponding unconjugated boIFNZ.3 protein in the mammal.
42. The conjugated protein of any one of claims 1 to 40, wherein the conjugated protein is characterized as having a plasma elimination half-life in a mammal; wherein the conjugated protein plasma elimination half-life is at least about 10-fold greater than the plasma elimination half-life of wild-type boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 1 in the mammal.
43. The conjugated protein of any one of claims 1 to 40, wherein the conjugated protein is characterized as having a plasma elimination half-life in a mammal; wherein the conjugated protein plasma elimination half-life is at least about 10-fold greater than the plasma elimination half-life of an unconjugated IF NX protein or an unconjugated IFNy protein in the mammal.
44. The conjugated protein of any one of claims 41 to 43, wherein the plasma elimination halflife is in a Sprague-Dawley rat after subcutaneous administration of a single dose of about 0.25 mg/kg.
45. The conjugated protein of any one of claims 41 to 43, wherein the plasma elimination halflife is in a Holstein-Fresian calf after subcutaneous administration of a single dose of about 75 Fg/kg-
46. The conjugated protein of any one of claims 1 to 40 or 45, wherein the conjugated protein is characterized as having a plasma elimination half-life in a mammal, wherein the mammal is a Holstein-Fresian calf, and wherein the conjugated protein plasma elimination half-life is at least about 50-fold greater than the plasma elimination half-life of (i) the corresponding unconjugated boIFNZ.3 protein; (ii) wild-type boIFNZ.3 protein having the amino acid sequence of SEQ ID NO: 1; (iii) an unconjugated IFNk protein; or (iv) an unconjugated IFNY protein in the mammal.
47. The conjugated protein of any one of claims 1 to 40, wherein the conjugated protein is characterized as having a plasma elimination half-life of at least about 40 hours in a Holstein- Fresian calf after administration of a single subcutaneous dose of about 75 pg/kg.
48. The conjugated protein of any one of claims 1 to 40, wherein the conjugated protein is characterized as having a plasma elimination half-life of at least about 50 hours in a Holstein- Fresian calf after administration of a single subcutaneous dose of about 75 pg/kg.
49. The conjugated protein of any one of claims 1 to 40, wherein the conjugated protein is characterized as having a plasma elimination half-life of at least about 60 hours in a Holstein- Fresian calf after administration of a single subcutaneous dose of about 75 pg/kg.
50. A pharmaceutical composition comprising a conjugated protein of any one of claims 1 to 49 and a pharmaceutically acceptable excipient or carrier.
51. An engineered bovine interferon 3 (boIFNZ.3) protein having an amino acid sequence that shares at least 90% identity with SEQ ID NO: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12 or 13, and which shares less than 100% identity with SEQ ID NO: 1, and optionally wherein the boIFNZ.3 protein further comprises an N-terminal methionine; and wherein: the boIFNZ.3 protein amino acid sequence comprises a non-natural amino acid; or the boIFNZ.3 protein amino acid sequence does not comprise a non-natural amino acid.
52. The engineered boIFNZ.3 protein of claim 51, wherein the engineered boIFNZ.3 protein has an amino acid sequence that shares at least 95% identity with SEQ ID NO: 5, and optionally further comprises an N-terminal methionine.
53. The engineered boIFNZ.3 protein of claim 52, wherein the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 5.
54. The engineered boIFNZ.3 protein of claim 52, wherein the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 5 further comprising the N-terminal methionine.
55. The engineered boIFNZ.3 protein of claim 51, wherein the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 6.
56. The engineered boIFNZ.3 protein of claim 51, wherein the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 6 further comprising the N-terminal methionine.
57. The engineered boIFNZ.3 protein of any one of claims 51 to 56, wherein the amino acid sequence does not comprise a non-natural amino acid.
58. The engineered boIFNZ.3 protein of claim 51 or 52, wherein the amino acid sequence comprises a non-natural amino acid, optionally wherein:
a) the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 5, in which a nonnatural amino acid is substituted for the amino acid at one or more position of that sequence, preferably a non-natural amino acid is substituted for the amino acid at one position of that sequence; b) the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 5 further comprising the N-terminal methionine, in which a non-natural amino acid is substituted for the amino acid at one or more position of that sequence, preferably a non-natural amino acid is substituted for the amino acid at one position of that sequence; c) the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 6, in which a nonnatural amino acid is substituted for the amino acid at one or more position of that sequence, preferably a non-natural amino acid is substituted for the amino acid at one position of that sequence; or d) the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 6 further comprising the N-terminal methionine, in which a non-natural amino acid is substituted for the amino acid at one or more position of that sequence, preferably a non-natural amino acid is substituted for the amino acid at one position of that sequence.
59. The engineered boIFNZ.3 protein of claim 51, 52 or 58, wherein the non-natural amino acid is selected from the group consisting of 4-acetyl-L-phenylalanine (para-acetyl-L-phenylalanine (pAF)), 3-O-(N-acetyl-beta-D-glucosaminyl)-L-threonine, N4-(P-N-Acetyl-D-glucosaminyl)-L- asparagine, O-allyl-L-tyrosine, alpha-N-acetylgalactosamine-O-L-serine, alpha-N- acetylgalactosamine-O-L-threonine, 2-aminooctanoic acid, 2-amino-L-phenylalanine, 3-amino-L- phenylalanine, 4-amino-L-phenylalanine, 2-amino-L-tyrosine, 3-amino-L-tyrosine, 4-azido-L- phenylalanine, 4-b enzoy 1 -L-pheny 1 al anine, (2,2-bipyridin-5yl)-L-alanine, 3-borono-L- phenylalanine, 4-borono-L-phenylalanine, 4-bromo-L-phenylalanine, p-carboxymethyl-L- phenylalanine, 4-carboxy-L-phenylalanine, p-cyano-L-phenylalanine, 3,4-dihydroxy-L- phenylalanine (L-DOPA), 4-ethynyl-L-phenylalanine, 2-fluoro-L-phenylalanine, 3-fluoro-L- phenylalanine, 4-fluoro-L-phenylalanine, O-(3-O-D-galactosyl-N-acetyl-beta-D-galactosaminyl)- L-serine, L-homoglutamine, (8-hydroxyquinolin-3-yl)-L-alanine, 4-iodo-L-phenylalanine, 4- isopropyl-L-phenylalanine, O-i -propyl -L-tyrosine, 3-isopropyl-L-tyrosine, O-mannopyranosyl-L- serine, 2-m ethoxy -L-phenylalanine, 3 -methoxy -L-phenylalanine, 4-m ethoxy -L-phenylalanine, 3- methyl-L-phenylalanine, O-methyl-L-tyrosine, 3-(2-naphthyl)-L-alanine, 5-nitro-L-histidine, 4- nitro-L-histidine, 4-nitro-L-leucine, 2-nitro-L-phenylalanine, 3-nitro-L-phenylalanine, 4-nitro-L- phenylalanine, 4-nitro-L-tryptophan, 5-nitro-L-tryptophan, 6-nitro-L-tryptophan, 7-nitro-L-
tryptophan, 2-nitro-L-tyrosine, 3-nitro-L-tyrosine, O-phospho-L-serine, O-phospho-L-tyrosine, 4- propargyloxy-L-phenylalanine, O-2-propyn-l-yl-L-tyrosine, 4-sulfo-L-phenylalanine and O- sulfo-L-tyrosine.
60. The engineered boIFNZ.3 protein of any one of claims 51, 52, 58 or 59, wherein the non-natural amino acid is para-acetyl-L-phenylalanine.
61. The engineered boIFNZ.3 protein of any one of claims 51, 52, 58, 59 or 60, wherein the nonnatural amino acid is at a position selected from the group consisting of G15, T58, Q65, N89, T119, S122, R123, G126, R139 and D169, wherein the position number is based on the wild type boIFNZ.3 protein amino acid sequence of SEQ ID NO: 1.
62. The engineered boIFNZ.3 protein of claim 61, wherein the non-natural amino acid is at position T119, wherein the position number is based on the wild type boIFNZ.3 protein amino acid sequence of SEQ ID NO: 1.
63. The engineered boIFNZ.3 protein of claim 62, wherein the non-natural amino acid at position T119 is para-acetyl-L-phenylalanine.
64. The engineered boIFNZ.3 protein of any one of claims 51 to 63, wherein the engineered boIFNZ.3 protein is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA.
65. The engineered boIFNZ.3 protein of any one of claims 51 to 64, wherein the non-natural amino acid is site-specifically incorporated.
66. A nucleic acid having the nucleotide sequence of SEQ ID NO: 27.
67. A vector comprising a nucleic acid encoding any one of SEQ ID NOS: 2 to 24.
68. A vector comprising a nucleic acid encoding SEQ ID NO: 5.
69. A vector comprising a nucleic acid encoding SEQ ID NO: 18.
70. A vector comprising a nucleic acid encoding SEQ ID NO: 19.
71. An engineered bovine interferon X3 (boIFNZ.3) protein having an amino acid sequence comprising a non-natural amino acid; wherein the engineered boIFNZ.3 protein amino acid sequence: a) shares at least 90% identity withSEQ ID NO: 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24, or
b) shares at least 90% identity with SEQ ID NO: 14, 15, 16, 17, 18, 20, 21, 22, 23 or 24, and wherein the boIFNZ.3 protein further comprises an N-terminal methionine.
72. The engineered boIFNZ.3 protein of claim 71, wherein the engineered boIFNZ.3 protein amino acid sequence shares at least 95% identity with SEQ ID NO: 18.
73. The engineered boIFNZ.3 protein of claim 71, wherein the engineered boIFNZ.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO: 18.
74. The engineered boIFNZ.3 protein of claim 71, wherein the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 18.
75. The engineered boIFNz.3 protein of claim 71, wherein the amino acid sequence shares at least 95% identity with SEQ ID NO: 19.
76. The engineered boIFNz.3 protein of claim 71, wherein the engineered boIFNZ.3 protein amino acid sequence shares at least 99% identity with SEQ ID NO: 19.
77. The engineered boIFNz.3 protein of claim 71, wherein the engineered boIFNZ.3 protein has the amino acid sequence of SEQ ID NO: 19.
78. The engineered boIFNz.3 protein of any one of claims 71 to 77, wherein the non-natural amino acid is selected from the group consisting of 4-acetyl-L-phenylalanine (para-acetyl -L- phenylalanine (pAF)), 3-O-(N-acetyl-beta-D-glucosaminyl)-L-threonine, N4-( -N-Acetyl-D- glucosaminyl)-L-asparagine, O-allyl-L-tyrosine, alpha-N-acetylgalactosamine-O-L-serine, alpha- N-acetylgalactosamine-O-L-threonine, 2-aminooctanoic acid, 2-amino-L-phenylalanine, 3- amino-L-phenylalanine, 4-amino-L-phenylalanine, 2-amino-L-tyrosine, 3-amino-L-tyrosine, 4- azido-L-phenylalanine, 4-benzoyl-L-phenylalanine, (2,2-bipyridin-5yl)-L-alanine, 3-borono-L- phenylalanine, 4-borono-L-phenylalanine, 4-bromo-L-phenylalanine, p-carboxymethyl-L- phenylalanine, 4-carboxy-L-phenylalanine, p-cyano-L-phenylalanine, 3,4-dihydroxy-L- phenylalanine (L-DOPA), 4-ethynyl-L-phenylalanine, 2-fluoro-L-phenylalanine, 3-fluoro-L- phenylalanine, 4-fluoro-L-phenylalanine, O-(3-O-D-galactosyl-N-acetyl-beta-D-galactosaminyl)- L-serine, L-homoglutamine, (8-hydroxyquinolin-3-yl)-L-alanine, 4-iodo-L-phenylalanine, 4- isopropyl-L-phenylalanine, O-i -propyl -L-tyrosine, 3-isopropyl-L-tyrosine, O-mannopyranosyl-L- serine, 2-m ethoxy -L-phenylalanine, 3 -methoxy -L-phenylalanine, 4-m ethoxy -L-phenylalanine, 3- methyl-L-phenylalanine, O-methyl-L-tyrosine, 3-(2-naphthyl)-L-alanine, 5-nitro-L-histidine, 4- nitro-L-histidine, 4-nitro-L-leucine, 2-nitro-L-phenylalanine, 3-nitro-L-phenylalanine, 4-nitro-L- phenylalanine, 4-nitro-L-tryptophan, 5-nitro-L-tryptophan, 6-nitro-L-tryptophan, 7-nitro-L- tryptophan, 2-nitro-L-tyrosine, 3-nitro-L-tyrosine, O-phospho-L-serine, O-phospho-L-tyrosine, 4-
propargyloxy -L-phenylalanine, O-2-propyn-l-yl-L-tyrosine, 4-sulfo-L-phenylalanine and O- sulfo-L-tyrosine.
79. The engineered boIFNZ.3 protein of claim 78, wherein the non-natural amino acid is paraacetyl -L-pheny 1 al anine .
80. The engineered boIFNZ.3 protein of any one of claims 71 to 79, wherein the engineered boIFNZ.3 protein is obtained from a cell comprising a translation system, wherein the translation system comprises an orthogonal tRNA and an orthogonal aminoacyl tRNA, and the non-natural amino acid is site-specifically incorporated.
81. A prophylactic method of treating a disease or condition in a mammal, the method comprising administering to the mammal a conjugated protein of any one of claims 1 to 49 or a pharmaceutical composition of claim 50.
82. The method of claim 81, wherein the mammal is a ruminant mammal.
83. The method of claim 82, wherein the ruminant mammal is bovine.
84. The method of claim 81, 82 or 83, wherein the disease or condition is a bovine respiratory disease.
85. The method of any one of claims 81 to 84, wherein the disease or condition is associated with bovine viral diarrhea virus (BVDV), parainfluenza-3 (PI3) or bovine respiratory syncytial virus (BRSV); or a combination thereof.
86. The method of claim 81, 82 or 83, wherein the disease or condition is foot and mouth disease.
87. The method of any one of claim 81 to 83 or 86, wherein the disease or condition is associated with foot and mouth disease virus (FMDV).
88. The method of any one of claims 81 to 87, wherein the method further comprises administering a vaccine.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363504133P | 2023-05-24 | 2023-05-24 | |
| US63/504,133 | 2023-05-24 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024241086A1 true WO2024241086A1 (en) | 2024-11-28 |
Family
ID=88975494
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2023/061789 Pending WO2024241086A1 (en) | 2023-05-24 | 2023-11-22 | Pegylated bovine interferon lambda and methods of use thereof |
Country Status (2)
| Country | Link |
|---|---|
| AR (1) | AR131133A1 (en) |
| WO (1) | WO2024241086A1 (en) |
Citations (41)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4820352A (en) | 1983-01-10 | 1989-04-11 | Bausch & Lomb Incorporated | Cleaning and conditioning solutions for contact lenses and methods of use |
| US5252714A (en) | 1990-11-28 | 1993-10-12 | The University Of Alabama In Huntsville | Preparation and use of polyethylene glycol propionaldehyde |
| WO2002086075A2 (en) | 2001-04-19 | 2002-10-31 | The Scripps Research Institute | Methods and composition for the production of orthoganal trna-aminoacyltrna synthetase pairs |
| US6602498B2 (en) | 2000-02-22 | 2003-08-05 | Shearwater Corporation | N-maleimidyl polymer derivatives |
| US6610281B2 (en) | 1993-11-12 | 2003-08-26 | Shearwater Corporation | Water soluble activated polymers containing an active ethyl sulfone moiety for modification of surfaces and molecules |
| WO2004035743A2 (en) | 2002-10-16 | 2004-04-29 | The Scripps Research Institute | Site specific incorporation of keto amino acids into proteins |
| WO2005007870A2 (en) | 2003-07-07 | 2005-01-27 | The Scripps Research Institute | COMPOSITIONS OF ORTHOGONAL LEUCYL-tRNA AND AMINOACYL-tRNA SYNTHETASE PAIRS AND USES THEREOF |
| WO2005007624A2 (en) | 2003-07-07 | 2005-01-27 | The Scripps Research Institute | Compositions of orthogonal glutamyl-trna and aminoacyl trna synthetase pairs and uses thereof |
| WO2005019415A2 (en) | 2003-07-07 | 2005-03-03 | The Scripps Research Institute | Compositions of orthogonal lysyl-trna and aminoacyl-trna synthetase pairs and uses thereof |
| US20050063943A1 (en) | 2001-03-16 | 2005-03-24 | Klaus Sommermeyer | Conjugated of hydroxyalkyl starch and an active agent |
| WO2005074650A2 (en) | 2004-02-02 | 2005-08-18 | Ambrx, Inc. | Modified human four helical bundle polypeptides and their uses |
| US20050234230A1 (en) | 2002-09-11 | 2005-10-20 | Norbert Zander | Hydroxyalkyl starch derivatives |
| WO2006050262A2 (en) | 2004-11-01 | 2006-05-11 | The Regents Of The University Of California | Compositions and methods for modification of biomolecules |
| US20060121073A1 (en) | 2004-07-12 | 2006-06-08 | Sandhya Goyal | Topical gel formulation comprising insecticide and its preparation thereof |
| WO2006068802A2 (en) | 2004-12-22 | 2006-06-29 | Ambrx, Inc. | COMPOSITIONS OF AMINOACYL-tRNA SYNTHETASE AND USES THEREOF |
| WO2006069246A2 (en) | 2004-12-22 | 2006-06-29 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| US7144574B2 (en) | 1999-08-27 | 2006-12-05 | Maxygen Aps | Interferon β variants and conjugates |
| WO2007012033A2 (en) * | 2005-07-20 | 2007-01-25 | Zymogenetics, Inc. | Il28 and il29 truncated cysteine mutants and antiviral methods of using same |
| WO2007021297A1 (en) | 2005-08-18 | 2007-02-22 | Ambrx, Inc. | COMPOSITIONS OF tRNA AND USES THEREOF |
| US20070087961A1 (en) | 2004-03-11 | 2007-04-19 | Wolfram Eichner | Conjugates of hydroxyalkyl starch and erythropoietin |
| WO2007059312A2 (en) | 2005-11-16 | 2007-05-24 | Ambrx, Inc. | Methods and compositions comprising non-natural amino acids |
| US20070134197A1 (en) | 2004-03-11 | 2007-06-14 | Wolfram Eichner | Conjugates of hydroxyalkyl starch and a protein, prepared by reductive amination |
| WO2007070659A2 (en) | 2005-12-14 | 2007-06-21 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| WO2007079130A2 (en) | 2005-12-30 | 2007-07-12 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| WO2007094916A2 (en) | 2006-01-19 | 2007-08-23 | Ambrx, Inc. | Non-natural amino acid polypeptides having modulated immunogenicity |
| US7285661B2 (en) | 2002-02-20 | 2007-10-23 | Fresenius Kabi Deutschland Gmbh | Starch derivatives, starch active substance conjugates, method for the production thereof and their use as medicaments |
| WO2008077079A1 (en) | 2006-12-18 | 2008-06-26 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| WO2008083346A1 (en) | 2006-12-28 | 2008-07-10 | Ambrx, Inc. | Phenazine and quinoxaline substituted amino acids and polypeptides |
| WO2010011735A2 (en) | 2008-07-23 | 2010-01-28 | Ambrx, Inc. | Modified bovine g-csf polypeptides and their uses |
| WO2010037062A1 (en) | 2008-09-26 | 2010-04-01 | Ambrx, Inc. | Non-natural amino acid replication-dependent microorganisms and vaccines |
| WO2012166559A1 (en) | 2011-05-27 | 2012-12-06 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acid linked dolastatin derivatives |
| WO2012166560A1 (en) | 2011-05-27 | 2012-12-06 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acid linked dolastatin derivatives |
| WO2013185117A1 (en) | 2012-06-07 | 2013-12-12 | Ambrx, Inc. | Prostate-specific membrane antigen antibody drug conjugates |
| US8906384B2 (en) | 2010-12-22 | 2014-12-09 | The United States Of America As Represented By The Secretary Of Agriculture | Antiviral activity of bovine type III interferon against foot-and-mouth disease virus |
| WO2015153761A2 (en) | 2014-04-02 | 2015-10-08 | University Of Rochester | Macrocyclic peptidomimetics for alpha-helix mimicry |
| US9637411B2 (en) | 2010-12-29 | 2017-05-02 | Sumco Corporation | Vitreous silica crucible and method of manufacturing the same |
| WO2017222940A1 (en) | 2016-06-20 | 2017-12-28 | Eli Lilly And Company | Pegylated porcine interferon and methods of use thereof |
| WO2018064574A1 (en) * | 2016-09-30 | 2018-04-05 | The Board Of Trustees Of The Leland Stanford Junior University | Variant type iii interferons and synthekines |
| WO2018223108A1 (en) | 2017-06-02 | 2018-12-06 | Ambrx, Inc. | Methods and compositions for promoting non-natural amino acid-containing protein production |
| CN110272479A (en) * | 2018-03-14 | 2019-09-24 | 江苏科技大学 | 3 interferon mutant of ox λ and its preparation method and application |
| WO2020041636A1 (en) * | 2018-08-23 | 2020-02-27 | Exalt Therapeutics, Llc | Modified ifnl3 polypeptides comprising a pharmacokinetic enhancing moiety and their uses |
-
2023
- 2023-11-22 WO PCT/IB2023/061789 patent/WO2024241086A1/en active Pending
- 2023-11-22 AR ARP230103138A patent/AR131133A1/en unknown
Patent Citations (56)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4820352A (en) | 1983-01-10 | 1989-04-11 | Bausch & Lomb Incorporated | Cleaning and conditioning solutions for contact lenses and methods of use |
| US5252714A (en) | 1990-11-28 | 1993-10-12 | The University Of Alabama In Huntsville | Preparation and use of polyethylene glycol propionaldehyde |
| US6610281B2 (en) | 1993-11-12 | 2003-08-26 | Shearwater Corporation | Water soluble activated polymers containing an active ethyl sulfone moiety for modification of surfaces and molecules |
| US7144574B2 (en) | 1999-08-27 | 2006-12-05 | Maxygen Aps | Interferon β variants and conjugates |
| US6602498B2 (en) | 2000-02-22 | 2003-08-05 | Shearwater Corporation | N-maleimidyl polymer derivatives |
| US20050063943A1 (en) | 2001-03-16 | 2005-03-24 | Klaus Sommermeyer | Conjugated of hydroxyalkyl starch and an active agent |
| US7045337B2 (en) | 2001-04-19 | 2006-05-16 | The Scripps Research Institute | In vivo incorporation of unnatural amino acids |
| WO2002086075A2 (en) | 2001-04-19 | 2002-10-31 | The Scripps Research Institute | Methods and composition for the production of orthoganal trna-aminoacyltrna synthetase pairs |
| US7083970B2 (en) | 2001-04-19 | 2006-08-01 | The Scripps Research Institute | Methods and compositions for the production of orthogonal tRNA-aminoacyl tRNA synthetase pairs |
| WO2002085923A2 (en) | 2001-04-19 | 2002-10-31 | The Scripps Research Institute | In vivo incorporation of unnatural amino acids |
| US7285661B2 (en) | 2002-02-20 | 2007-10-23 | Fresenius Kabi Deutschland Gmbh | Starch derivatives, starch active substance conjugates, method for the production thereof and their use as medicaments |
| US20060019877A1 (en) | 2002-09-11 | 2006-01-26 | Conradt Harald S | Hasylated polypeptides |
| US20050234230A1 (en) | 2002-09-11 | 2005-10-20 | Norbert Zander | Hydroxyalkyl starch derivatives |
| US20050238723A1 (en) | 2002-09-11 | 2005-10-27 | Norbert Zander | Method of producing hydroxyalkyl starch derivatives |
| WO2004035743A2 (en) | 2002-10-16 | 2004-04-29 | The Scripps Research Institute | Site specific incorporation of keto amino acids into proteins |
| WO2005019415A2 (en) | 2003-07-07 | 2005-03-03 | The Scripps Research Institute | Compositions of orthogonal lysyl-trna and aminoacyl-trna synthetase pairs and uses thereof |
| WO2005007624A2 (en) | 2003-07-07 | 2005-01-27 | The Scripps Research Institute | Compositions of orthogonal glutamyl-trna and aminoacyl trna synthetase pairs and uses thereof |
| WO2005007870A2 (en) | 2003-07-07 | 2005-01-27 | The Scripps Research Institute | COMPOSITIONS OF ORTHOGONAL LEUCYL-tRNA AND AMINOACYL-tRNA SYNTHETASE PAIRS AND USES THEREOF |
| WO2005074650A2 (en) | 2004-02-02 | 2005-08-18 | Ambrx, Inc. | Modified human four helical bundle polypeptides and their uses |
| US20070087961A1 (en) | 2004-03-11 | 2007-04-19 | Wolfram Eichner | Conjugates of hydroxyalkyl starch and erythropoietin |
| US20070134197A1 (en) | 2004-03-11 | 2007-06-14 | Wolfram Eichner | Conjugates of hydroxyalkyl starch and a protein, prepared by reductive amination |
| US20060121073A1 (en) | 2004-07-12 | 2006-06-08 | Sandhya Goyal | Topical gel formulation comprising insecticide and its preparation thereof |
| WO2006050262A2 (en) | 2004-11-01 | 2006-05-11 | The Regents Of The University Of California | Compositions and methods for modification of biomolecules |
| WO2006068802A2 (en) | 2004-12-22 | 2006-06-29 | Ambrx, Inc. | COMPOSITIONS OF AMINOACYL-tRNA SYNTHETASE AND USES THEREOF |
| US8791231B2 (en) | 2004-12-22 | 2014-07-29 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| US8008456B2 (en) | 2004-12-22 | 2011-08-30 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| US7696312B2 (en) | 2004-12-22 | 2010-04-13 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| US8048988B2 (en) | 2004-12-22 | 2011-11-01 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| WO2006069246A2 (en) | 2004-12-22 | 2006-06-29 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| US8859802B2 (en) | 2004-12-22 | 2014-10-14 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| US7928163B2 (en) | 2004-12-22 | 2011-04-19 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| US7332571B2 (en) | 2004-12-22 | 2008-02-19 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| US8809511B2 (en) | 2004-12-22 | 2014-08-19 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| US8476411B2 (en) | 2004-12-22 | 2013-07-02 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| WO2007012033A2 (en) * | 2005-07-20 | 2007-01-25 | Zymogenetics, Inc. | Il28 and il29 truncated cysteine mutants and antiviral methods of using same |
| WO2007021297A1 (en) | 2005-08-18 | 2007-02-22 | Ambrx, Inc. | COMPOSITIONS OF tRNA AND USES THEREOF |
| WO2007059312A2 (en) | 2005-11-16 | 2007-05-24 | Ambrx, Inc. | Methods and compositions comprising non-natural amino acids |
| WO2007070659A2 (en) | 2005-12-14 | 2007-06-21 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| WO2007079130A2 (en) | 2005-12-30 | 2007-07-12 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| WO2007094916A2 (en) | 2006-01-19 | 2007-08-23 | Ambrx, Inc. | Non-natural amino acid polypeptides having modulated immunogenicity |
| WO2011028195A2 (en) | 2006-12-18 | 2011-03-10 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| WO2008077079A1 (en) | 2006-12-18 | 2008-06-26 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acids and polypeptides |
| WO2008083346A1 (en) | 2006-12-28 | 2008-07-10 | Ambrx, Inc. | Phenazine and quinoxaline substituted amino acids and polypeptides |
| WO2010011735A2 (en) | 2008-07-23 | 2010-01-28 | Ambrx, Inc. | Modified bovine g-csf polypeptides and their uses |
| WO2010037062A1 (en) | 2008-09-26 | 2010-04-01 | Ambrx, Inc. | Non-natural amino acid replication-dependent microorganisms and vaccines |
| US8906384B2 (en) | 2010-12-22 | 2014-12-09 | The United States Of America As Represented By The Secretary Of Agriculture | Antiviral activity of bovine type III interferon against foot-and-mouth disease virus |
| US9637411B2 (en) | 2010-12-29 | 2017-05-02 | Sumco Corporation | Vitreous silica crucible and method of manufacturing the same |
| WO2012166560A1 (en) | 2011-05-27 | 2012-12-06 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acid linked dolastatin derivatives |
| WO2012166559A1 (en) | 2011-05-27 | 2012-12-06 | Ambrx, Inc. | Compositions containing, methods involving, and uses of non-natural amino acid linked dolastatin derivatives |
| WO2013185117A1 (en) | 2012-06-07 | 2013-12-12 | Ambrx, Inc. | Prostate-specific membrane antigen antibody drug conjugates |
| WO2015153761A2 (en) | 2014-04-02 | 2015-10-08 | University Of Rochester | Macrocyclic peptidomimetics for alpha-helix mimicry |
| WO2017222940A1 (en) | 2016-06-20 | 2017-12-28 | Eli Lilly And Company | Pegylated porcine interferon and methods of use thereof |
| WO2018064574A1 (en) * | 2016-09-30 | 2018-04-05 | The Board Of Trustees Of The Leland Stanford Junior University | Variant type iii interferons and synthekines |
| WO2018223108A1 (en) | 2017-06-02 | 2018-12-06 | Ambrx, Inc. | Methods and compositions for promoting non-natural amino acid-containing protein production |
| CN110272479A (en) * | 2018-03-14 | 2019-09-24 | 江苏科技大学 | 3 interferon mutant of ox λ and its preparation method and application |
| WO2020041636A1 (en) * | 2018-08-23 | 2020-02-27 | Exalt Therapeutics, Llc | Modified ifnl3 polypeptides comprising a pharmacokinetic enhancing moiety and their uses |
Non-Patent Citations (75)
| Title |
|---|
| "Basic Pharmacokinetics and Pharmacodynamics: An Integrated Textbook and Computer Simulations", 2016, STATPEARLS PUBLISHING |
| "GenBank", Database accession no. ADP05157.1 |
| "Polyethylene Glycol and Derivatives for Advanced PEGylation", NEKTAR MOLECULAR ENGINEERING CATALOG, 2003, pages 1 - 17 |
| "Remington's Pharmaceutical Sciences", 1985 |
| BANSAL, R. ET AL., J. CONTROLLED RELEASE, vol. 154, no. 3, 2011, pages 233 - 240 |
| BATZER ET AL., NUCLEIC ACID RES., vol. 19, 1991, pages 5081 |
| CHIN ET AL., PROC. NATL. ACAD. SCI., vol. 99, 2002, pages 11020 - 11024 |
| CHIN J. W. ET AL., PROC. NATL. ACAD. SCI. U.S. A., vol. 99, 2002, pages 11020 - 11024 |
| CHIN J.W.SCHULTZ P.G., CHEMBIOCHEM, vol. 3, no. 11, 2002, pages 1135 - 1137 |
| CHIN, J. W. ET AL., J. AM. CHEM. SOC., vol. 124, 2002, pages 9026 - 9027 |
| CHIN, J. W. ET AL., SCIENCE, vol. 301, 2003, pages 964 - 967 |
| CHINSANGARAM ET AL., J. VIROL., vol. 73, 1999, pages 9891 - 9898 |
| CHINSANGARAM ET AL., J. VIROL., vol. 75, 2001, pages 5498 - 5503 |
| CHINSANGARAM ET AL., J. VIROL., vol. 77, 2003, pages 1621 - 1625 |
| CLARK, R. ET AL., J. BIOL. CHEM., vol. 271, 1996, pages 21969 - 21977 |
| CORNISH ET AL., J. AM. CHEM. SOC., vol. 118, 1996, pages 8150 - 8151 |
| DIAZ-SAN SEGUNDO F. ET AL., FRONTIERS IN MICROBIOLOGY, vol. 12, 2021 |
| DIAZ-SAN SEGUNDO F. ET AL., VIROLOGY, vol. 413, 2011, pages 283 - 292 |
| DIAZ-SAN SEGUNDO F. ET AL., VIROLOGY, vol. 499, 2016, pages 340 - 349 |
| DONNELLY RKOTENKO S., INTERFERON & CYTOKINE RES., vol. 30, 2010, pages 555 - 564 |
| EDWARDS, H.SCHIMMEL, P., MOL. CELL. BIOL., vol. 10, 1990, pages 1633 - 1641 |
| ELLIS J. ET AL., J. AM. VET. MED. ASSOC., vol. 219, 2001, pages 351 - 356 |
| GAGEA M.I. ET AL., J. VET. DIAGN. INVEST., vol. 18, 2006, pages 18 - 28 |
| GRUBMANBAXT, CLIN MICROBIOL REV, vol. 17, 2004, pages 465 - 493 |
| HARRIS J.M.CHESS R.B., NATURE REVIEWS DRUG DISCOVERY, vol. 2, 2003, pages 214 - 221 |
| HARRIS, J.M., J. MACROMOLECULAR SCI., PART C, POLYMER REVIEWS, vol. 25, no. 3, 1985, pages 325 - 373 |
| HUTCHINS ET AL., J MOL BIOL, vol. 406, no. 4, 2011, pages 595 - 603 |
| JOHNSON ET AL., NAT CHEM BIOL, vol. 7, no. 11, 2011, pages 779 - 86 |
| KABSCH W.SANDER C., BIOPOLYMERS, vol. 22, 1983, pages 2577 - 2637 |
| KAZANE ET AL., J AM CHEM SOC, vol. 135, no. 1, 2013, pages 340 - 6 |
| KELLY A.P.JANZEN E.D., CAN. VET. J., vol. 27, no. 12, 1986, pages 496 - 500 |
| KIM ET AL., J AM CHEM SOC, vol. 134, no. 24, 2012, pages 9918 - 21 |
| KONTERMANN R.E., EXPERT OPIN. BIOL. THERAP., vol. 16, no. 7, 2016, pages 903 - 915 |
| KOWAL, A. K. ET AL., PROC. NATL. ACAD. SCI. U. S. A., vol. 98, 2001, pages 2268 - 2273 |
| LILLIE L.E., CAN. VET. J., vol. 15, 1974, pages 233 - 242 |
| LIN J.D. ET AL., PLOS PATHOG., vol. 12, no. 4, 2016, pages e1005600 |
| LIU ET AL., ANNU REV BIOCHEM, vol. 79, 2010, pages 413 - 44 |
| LIU, D. R.SCHULTZ, P. G., PROC. NATL. ACAD. SCI. U. S. A., vol. 96, 1999, pages 4780 - 4785 |
| LOPUSNA K., ACTA. VIROLOGICA, vol. 57, 2013, pages 171 - 179 |
| MAHAL, SCIENCE, vol. 276, 1997, pages 1125 - 1128 |
| MORAES ET AL., J. VIROL., vol. 81, 2007, pages 7124 - 7135 |
| MORAESDIAS ET AL., J. INTERFERON CYTOKINE RES., vol. 31, 2011, pages 227 - 236 |
| NATURE PROTOCOLS, vol. 7, 2012, pages 1052 - 1067 |
| OHNO, S. ET AL., J. BIOCHEM. (TOKYO, JPN., vol. 124, 1998, pages 1065 - 1068 |
| OHTSUKA ET AL., J. BIOL. CHEM., vol. 260, 1985, pages 2605 - 2608 |
| OIE: "Foot and Mouth Disease", 2007, article "Terrestrial animal health code" |
| PASTRNAK, M. ET AL., HELV. CHIM. ACTA, vol. 83, 2000, pages 2277 - 2286 |
| PEREZ-MARTIN ET AL., J. INTERFERON CYTOKINE RES., vol. 34, 2014, pages 810 - 821 |
| PEREZ-MARTIN, J. VIROL., vol. 86, 2012, pages 4477 - 4487 |
| RICHESON J.T. ET AL., J. AN. SCI., vol. 86, no. 4, 2008, pages 999 - 1005 |
| ROBERTS M.J.HARRIS J.M, ADV. DRUG DELIVERY REVIEWS, vol. 54, no. 4, 2002, pages 459 - 476 |
| ROSSOLINI ET AL., MOL. CELL. PROBES, vol. 8, 1994, pages 91 - 98 |
| ROSTOVTSEV ET AL., ANGEW. CHEM. INT. ED., vol. 41, 2002, pages 2596 - 2599 |
| SAKAMOTO, K. ET AL., NUCLEIC ACIDS RES., vol. 30, 2002, pages 4692 - 4699 |
| SANG, Y. ET AL., J. INTERFERON CYTOKINE RES., vol. 30, 2010, pages 1 - 7 |
| SCOUTEN, W.H., METHODS IN ENZYMOLOGY, vol. 135, 1987, pages 30 - 65 |
| STENFELDT C.BELSHAM G.J., VETERINARY MICROBIOLOGY, vol. 154, 2012, pages 230 - 239 |
| TAYLOR J.D. ET AL., CAN. VET. J., vol. 51, no. 10, 2010, pages 1095 - 1102 |
| TIAN F. ET AL., PROC. NATL. ACAD. SCI. U.S.A, vol. 111, no. 5, 2014, pages 1766 - 1771 |
| TOMOE ET AL., J. ORG. CHEM., vol. 67, 2002, pages 3057 - 3064 |
| TOUTAIN P.L.BOUSQUET-MELOU A., J. VETINARY PHARMACOLOGY AND THERAPEUTICS, vol. 27, no. 6, 2004, pages 427 - 439 |
| URBAN-CHMIEL R.GROOMS, D.L., J. LIVESTOCK SCI., vol. 3, 2012, pages 27 - 36 |
| WANG ET AL., ANGEW CHEM INT ED, vol. 44, 2005, pages 34 - 66 |
| WANG ET AL., PROC. NATL. ACAD. SCI., vol. 100, 2003, pages 56 - 61 |
| WANG L. ET AL., SCIENCE, vol. 292, 2001, pages 498 - 500 |
| WANG L.SCHULTZ P.G., CHEM. COMM., vol. 1, 2002, pages 1 - 11 |
| WANG, L. ET AL., PROC. NATL. ACAD. SCI. USA, vol. 100, 2003, pages 56 - 61 |
| WELSH R.D. ET AL., J. VET. DIAGN. INVEST., vol. 16, 2004, pages 426 - 431 |
| WONG S.S.WONG L-J.C., ENZYME MICROB. TECHNOL., vol. 14, no. 11, 1992, pages 866 - 874 |
| WU ET AL., J. INTERFERON CYTOKINE RES., vol. 23, 2003, pages 371 - 380 |
| ZALIPSKY, S., BIOCONJUGATE CHEM., vol. 6, no. 2, 1995, pages 150 - 165 |
| ZEUZEM, S. ET AL., SEMINARS IN LIVER DISEASE, vol. 23, 2003, pages 023 - 028 |
| ZHANG Z.ALEXANDERSEN S., J VIROL METHODS, vol. 111, 2003, pages 95 - 100 |
| ZHANG, Z. ET AL., BIOCHEM., vol. 42, no. 22, 2003, pages 6735 - 6746 |
| ZHANG, Z. ET AL., BIOCHEMISTRY, vol. 42, 2003, pages 6735 - 6746 |
Also Published As
| Publication number | Publication date |
|---|---|
| AR131133A1 (en) | 2025-02-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7441892B2 (en) | Pegylated porcine interferon and its use | |
| EP4389145A2 (en) | Interleukin-2 polypeptide conjugates and their uses | |
| WO2020168017A1 (en) | Compositions containing, methods and uses of antibody-tlr agonist conjugates | |
| EA019968B1 (en) | Modified bovine g-csf polypeptides and their uses | |
| US20220009986A1 (en) | Interleukin-10 polypeptide conjugates, dimers thereof, and their uses | |
| IL296099A (en) | Interleukin-2 polypeptide conjugates and methods of use thereof | |
| WO2022212899A1 (en) | Anti-her2 antibody-drug conjugates and uses thereof | |
| JP2021502990A (en) | Methods for reducing the reconstitution time of spray-dried protein formulations | |
| WO2024241086A1 (en) | Pegylated bovine interferon lambda and methods of use thereof | |
| JP2013538828A (en) | Formulation for bovine granulocyte colony-stimulating factor and variants thereof | |
| HK40113033A (en) | Interleukin-2 polypeptide conjugates and their uses | |
| HK40074343A (en) | Interleukin-2 polypeptide conjugates and methods of use thereof | |
| HK40057215A (en) | Interleukin-2 polypeptide conjugates and their uses | |
| HK40057215B (en) | Interleukin-2 polypeptide conjugates and their uses | |
| CN117440833A (en) | anti-HER 2 antibody-drug conjugates and uses thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 23813886 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |