WO2024236167A1 - Il-13 receptor alpha 2 binding polypeptides - Google Patents
Il-13 receptor alpha 2 binding polypeptides Download PDFInfo
- Publication number
- WO2024236167A1 WO2024236167A1 PCT/EP2024/063669 EP2024063669W WO2024236167A1 WO 2024236167 A1 WO2024236167 A1 WO 2024236167A1 EP 2024063669 W EP2024063669 W EP 2024063669W WO 2024236167 A1 WO2024236167 A1 WO 2024236167A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- seq
- helix
- present disclosure
- amino acid
- hth
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
- C07K14/715—Receptors; Cell surface antigens; Cell surface determinants for cytokines; for lymphokines; for interferons
- C07K14/7155—Receptors; Cell surface antigens; Cell surface determinants for cytokines; for lymphokines; for interferons for interleukins [IL]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/33—Fusion polypeptide fusions for targeting to specific cell types, e.g. tissue specific targeting, targeting of a bacterial subspecies
Definitions
- the present invention provides IL- 13 receptor alpha 2 (IL-13Ra2) receptor binding polypeptides based on a helix-turn-helix scaffold structure (HTH polypeptide or Helix-turn-Helix polypeptide), fusion molecules comprising such polypeptides and pharmaceutical compositions containing the same, and methods of use thereof.
- IL-13Ra2 IL- 13 receptor alpha 2 receptor alpha 2 receptor binding polypeptides based on a helix-turn-helix scaffold structure (HTH polypeptide or Helix-turn-Helix polypeptide), fusion molecules comprising such polypeptides and pharmaceutical compositions containing the same, and methods of use thereof.
- IL-13Ra2 is overexpressed in a variety of human tumor types such as glioblastoma (Stupp, R. et al., N Engl J Med 352, 987-996 (2005); Zheng, S. et al., The journal of gene medicine 9, 151-160 (2007); Van Houdt, W. J. et al., J Neurosurg 104, 583-592 (2006)), renal cell carcinoma, pancreatic, melanoma, head and neck, mesothelioma and ovarian. In contrast, normal tissues express little to no IL-13Ra2, with the exception of the testes (Iliasov, I. V.
- IL-13Ra2 exists on the cell membrane, intracellular, and in soluble form and has an extremely high affinity for IL-13.
- the strong binding affinity of IL-13 to IL-13Ra2 was interpreted as a way to sequester IL-13 and provoke its downregulation.
- membrane bound IL-13Ra2 might has IL-13 mediated signaling capabilities in cancer metastasis (WO 2019/086676).
- IL-13Ra1 a different receptor with low affinity for IL-13, is expressed ubiquitously by many tissues making it a poor candidate for selective targeting of tumor-specific therapeutic applications (Chakravarti, A. et al., Journal of the American Society of Clinical Oncology 20, 1063-1068 (2002); Uematsu, M. et al., Journal of neuro-oncology 72, 231-238 (2005); Choi, B. D. et al., Proceedings of the National Academy of Sciences of the United States of America 110, 270-275 (2013)).
- IL-13Ra1 forms a heterodimer with the alpha chain of the IL-4 receptor (IL-4Ra) that is a signaling IL-13 receptor.
- IL- 13 receptors for cancer therapy has been the subject of numerous studies and different strategies, including immunotoxins, DNA vaccines and specific monoclonal antibodies.
- One approach for targeting IL-13Ra2 utilizes its cognate ligand IL-13, conjugated to cytotoxic molecules (W01996/029417).
- Some studies involved the use of IL-13 immunotoxin, containing a truncated version of Pseudomonas exotoxin, which was highly cytotoxic to renal cancer cells and other human solid tumors (Puri RK et al, Blood 1996; 87:4333- 9).
- IL-13 immunotoxin has been also used in a Phase III clinical trial with glioblastoma patients, showing small but significant effects on survival (Kunwar S. et al, Neuro Oncol 2010; 12:871-81).
- IL-13 also binds the low affinity receptor IL-13Ra1 , which is abundantly expressed in many normal tissues.
- other approaches were developed utilizing de novo designed muteins of IL-13 with altered binding affinities towards IL-13Ra2 or IL13Ra1 (WO1999/051643, WO2002/018422, WO2013/112871).
- Other approaches utilize targeting peptides obtained from synthetic phage libraries (WO 2010121125A1). However, these peptides bind to a region on IL-13Ra2 not involved in IL-13 binding.
- the present disclosure provides novel polypeptides based on a conformational constrained helix-turn-helix scaffold structure (HTH polypeptide) which specifically bind to IL-13Ra2.
- HTH polypeptide conformational constrained helix-turn-helix scaffold structure
- the polypeptides bind to human IL-13Ra2 and preferably cross-react with cynomolgus monkey and mouse IL-13Ra2.
- the polypeptides according to the present disclosure do not bind to IL- 13Ra1. In certain embodiments, the polypeptides according to the present disclosure do not bind to human IL-4Ra. In certain embodiments, the polypeptides according to the present disclosure do not bind to the human IL-13Ra1/IL-4Ra complex. In certain embodiments, said IL-13Ra1 or IL-4Ra is human or mouse IL-13Ra1 or IL-4Ra.
- the polypeptides according to the present disclosure bind to the binding region of IL-13 on IL-13Ra2.
- the polypeptides compete with IL-13 for binding to IL-13Ra2.
- the polypeptides may interfere with IL-13 mediated signaling through IL-13Ra2.
- the polypeptides according to the present disclosure are small in size ( ⁇ 6kDa), reveal low immunogenicity, extreme stability against thermal and chemical denaturation, relatively insensitiveness to changes in pH and to proteolytic degradation.
- the HTH polypeptides according to the present disclosure may optionally be fused or conjugated to one or more moieties, such as therapeutic agents or detectable tags.
- polypeptides according to the present disclosure combine favorable structural, functional and safety properties never observed before. These features make the polypeptides superior to the IL-13 peptide mimetic approaches known from the art and as such highly desirable for diagnostic and therapeutic use, such as for preventing and/or treating cancer.
- polypeptides of the present disclosure are comprised of a helix-turn-helix scaffold structure of the formula Helix-1 - Li - Helix-2, wherein Li is a linker and wherein Helix-1 and Helix 2 comprise a first and second a-helical peptide, wherein said first and said second a-helical peptide form an antiparallel coiled-coiled structure.
- the present disclosure pertains to an isolated polypeptide specific for IL-13 receptor alpha 2 (IL-13Ra2) comprising a helix-turn-helix scaffold structure.
- said helix-turn-helix scaffold structure has the formula Helix-1 - Li - Helix-2, wherein Li is a linker and wherein Helix-1 and Helix 2 comprise a first and second a-helical peptide.
- said first and said second a-helical peptide form an antiparallel coiled-coiled structure.
- the present disclosure pertains to an isolated polypeptide specific for IL-13 receptor alpha 2 (IL-13Ra2) comprising a helix-turn-helix scaffold structure of the formula Helix-1 - Li - Helix-2, wherein Li is a linker and wherein Helix-1 and Helix 2 comprise a first and second a-helical peptide, wherein each of said a-helical peptides comprises the amino acid sequence
- Vari is any natural occurring amino acid excluding G, P and C in Helix-1 and Helix-2,
- Var2 is K, R, T or I in Helix-1 and I or K in Helix-2,
- Var3 is R or Q in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2
- Var4 is Q or K in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2
- Var5 is F, Y or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
- Var6 is K, R or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
- X1 is Q in Helix-1 and A in Helix-2,
- X2 is E in Helix-1 and K in Helix-2, and
- X3 is K in Helix-1 and M or A in Helix-2
- the present disclosure pertains to an isolated polypeptide specific for IL-13 receptor alpha 2 (IL-13Ra2) comprising a helix-turn-helix scaffold structure of the formula Helix-1 - Li - Helix-2, wherein Li is a linker and wherein Helix-1 comprise a first a-helical peptide and Helix- 2 comprise a second a-helical peptide, wherein each of said a-helical peptides comprises the amino acid sequence of
- Vari is any natural occurring amino acid excluding G, P and C in Helix 1 and Helix 2,
- Var2 is K, R, T or I in Helixl and I or K in Helix-2,
- Var3 is R or Q in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
- Var4 is Q or K in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
- Var5 is F, Y or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
- Var6 is K, R or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
- X1 is Q in Helix-1 and A in Helix-2
- X2 is E in Helix-1 and K in Helix-2
- X3 is K in Helix-1 and M or A in Helix-2.
- the present disclosure pertains to an isolated polypeptide specific for IL-13 receptor alpha 2 (IL-13Ra2) comprising a structure of the formula Helix-1 - Li - Helix-2, wherein Li is a linker and wherein Helix-1 comprises the amino acid sequence of
- Vari is any natural occurring amino acid excluding G, P and C in Helix 1 and Helix 2,
- Var2 is K, R, T or I in Helixl and I or K in Helix-2,
- Var3 is R or Q in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
- Var4 is Q or K in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
- Var5 is F, Y or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
- Var6 is K, R or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
- X1 is Q in Helix-1 and A in Helix-2,
- X2 is E in Helix-1 and K in Helix-2, and
- X3 is K in Helix-1 and M or A in Helix-2.
- Var3 is A, R, E or Q in Helix-2
- Var4 is A, H, D, I, or E in Helix-2
- Var5 is A, E, I, L, T, K or D in Helix-2, and
- Var6 is A, Q, R or H in Helix-2
- Var2 is K in Helix-1 and I Helix-2
- Var3 is R in Helix-1 and Helix-2
- Var4 is Q in Helix-1 and H or D in Helix-2,
- Var5 is Y in Helix-1 and E or I in Helix-2, and
- Var6 is R in Helix-1 and Q or R in Helix-1.
- the present disclosure provides an isolated polypeptide specific for IL- 13Ra2 as disclosed herein, wherein the first and second a-helical peptide form an antiparallel coiled-coiled structure.
- the present disclosure provides an isolated polypeptide specific for IL-13Ra2 as disclosed herein, wherein Helix-1 and Helix-2 form an antiparallel coiled-coiled structure.
- the isolated polypeptide specific for IL- 13 receptor alpha 2 (IL-13Ra2) according to the present disclosure comprises a helix-turn- helix scaffold structure.
- the present disclosure provides an isolated polypeptide specific for IL-13Ra2 as disclosed herein, wherein Helix-1 comprises a first a- helical peptide and Helix-2 comprises a second a-helical peptide.
- Helix-1 comprises the amino acid sequence of SEQ ID NO: 197 and Helix-2 comprises the amino acid sequence of SEQ ID NO: 204.
- the first a-helical peptide comprises the amino acid sequence of SEQ ID NO: 197 and the second a-helical peptide comprises the amino acid sequence of SEQ ID NO: 204.
- the isolated polypeptide specific for IL-13Ra2 according to the present disclosure is cross- reactive to mouse and cynomolgus IL-13Ra2.
- the isolated polypeptide specific for IL-13Ra2 according to the present disclosure competes with IL-13 for binding to IL-13Ra2. In an embodiment, the isolated polypeptide according to the present disclosure does not bind to IL-13 receptor alpha 1 (IL- 13Ra1), IL-4 receptor alpha (IL-4Ra) or the IL-13Ra1/IL-4Ra complex.
- IL-13 receptor alpha 1 IL- 13Ra1
- IL-4Ra IL-4 receptor alpha
- the isolated polypeptide specific for IL-13Ra2 according to the present disclosure is internalized from the surface of a cell expressing IL-13Ra2. In an embodiment of the present disclosure, said internalization occurs upon binding of said polypeptide to IL-13Ra2 present of the surface of said cell expressing IL-13Ra2.
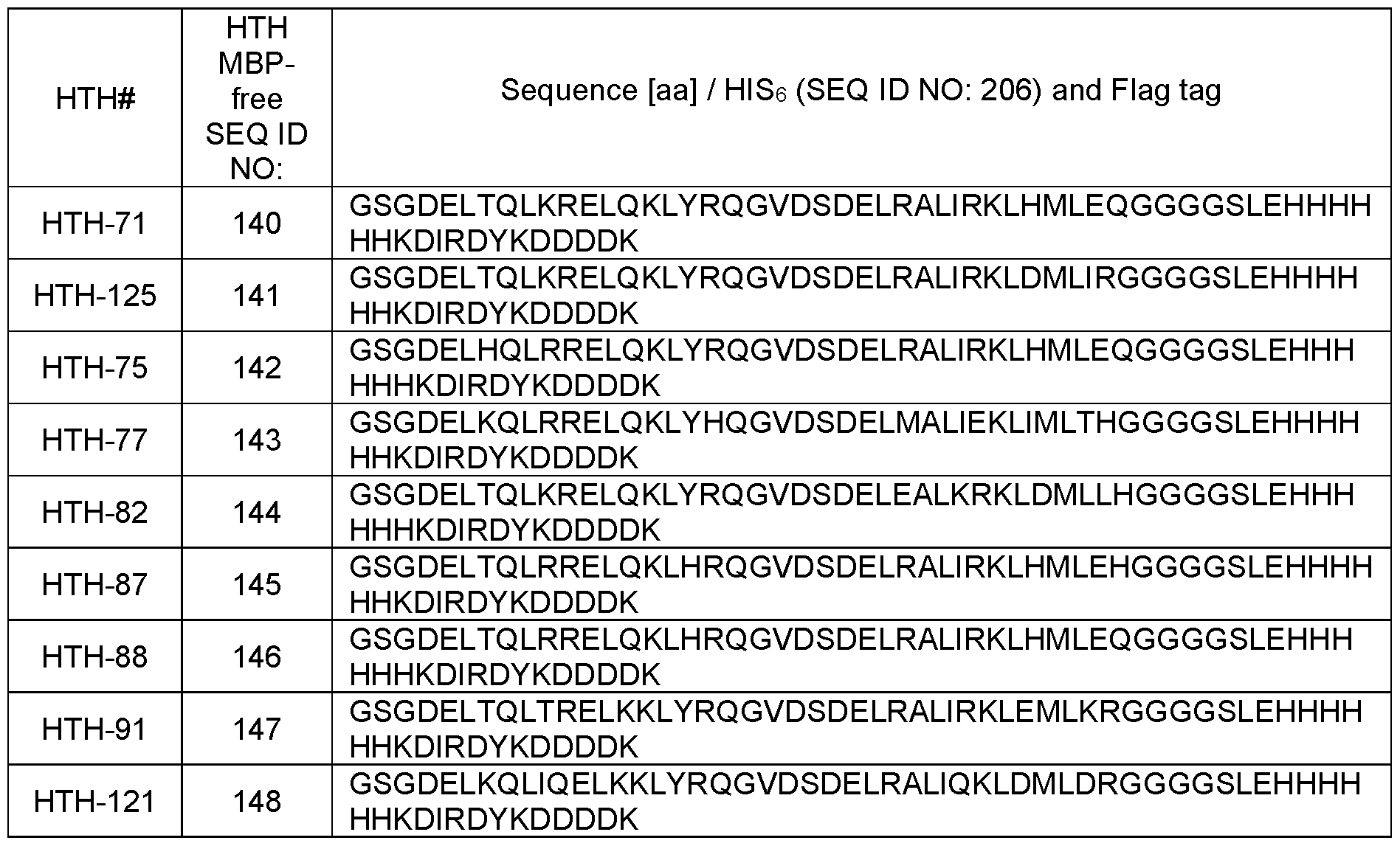
- the present disclosure provides an isolated polypeptide specific IL-13Ra2 as disclosed herein, wherein Helix-1 or the first a-helical peptide comprises of the amino sequence selected from the group consisting of: DELTQLKRELQKLYR (SEQ ID NO: 1),
- DELAQLKRELQKLYR SEQ ID NO: 2
- DELTQLKRELQKLYK SEQ ID NO: 3
- DELTQLTRELKKLYR SEQ ID NO: 8
- DELKQLIQELKKLYR SEQ ID NO: 9
- the present disclosure provides an isolated polypeptide specific for IL- 13Ra2 as disclosed herein, wherein Helix-2 or the second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQ (SEQ ID NO: 19), DELRALIRKLHALEQ (SEQ ID NO: 20), DELAALIRKLHMLEQ (SEQ ID NO: 21), DELRALIAKLHMLEQ (SEQ ID NO: 22), DELRALIRKLAMLEQ (SEQ ID NO: 23),
- DELRALIRKLHMLAQ (SEQ ID NO: 24), DELRALIRKLHMLEA (SEQ ID NO: 25),
- DELRALIRKLDMLIR SEQ ID NO: 26
- DELEALKRKLDMLLH SEQ ID NO: 27
- DELRALIRKLEMLKR SEQ ID NO: 30
- DELRALIQKLDMLDR SEQ ID NO: 31
- the present disclosure provides an isolated polypeptide specific for IL-13Ra2 as disclosed herein, wherein the linker Li comprises 1 to 30 amino acid residues.
- said linker Li comprises the amino acid sequence QGVDS (SEQ ID NO: 182) or QGVGS (SEQ ID NO: 183).
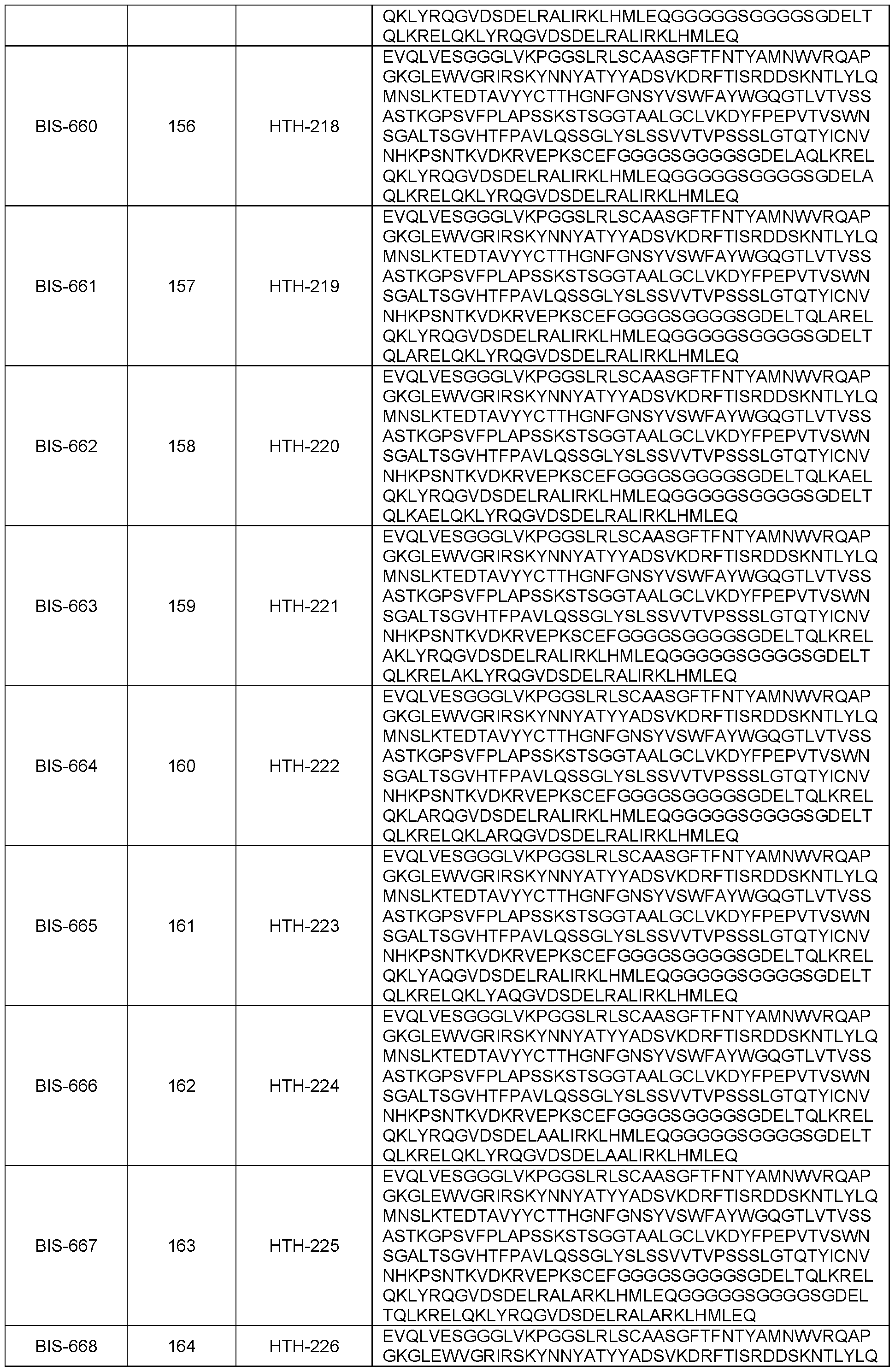
- the present disclosure provides an isolated polypeptide specific for IL-13Ra2 as disclosed herein, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of:
- DELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQ SEQ ID NO: 47
- DELTQLKRELQKLYRQGVDSDELRALIRKLHALEQ SEQ ID NO: 49
- DELTQLKRELQKLYRQGVDSDELRALIRKLDMLIR SEQ ID NO: 58
- DELTQLKRELQKLYRQGVDSDELEALKRKLDMLLH SEQ ID NO: 59
- DELHQLRRELQKLYRQGVDSDELRALIRKLHMLEQ SEQ ID NO: 60
- DELKQLRRELQKLYHQGVDSDELMALI EKLIM LTH SEQ ID NO: 61
- DELTQLRRELQKLHRQGVDSDELRALIRKLHMLEH SEQ ID NO: 62
- DELTQLRRELQKLHRQGVDSDELRALIRKLHMLEQ SEQ ID NO: 63
- the present disclosure provides an isolated polypeptide specific for IL- 13Ra2 as disclosed herein, wherein a G is present at the N- and/or C-terminus of the helix- turn-helix scaffold structure.
- the present disclosure provides an isolated polypeptide specific for IL- 13Ra2 as disclosed herein, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of:
- GDELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 66), GDELTQLKRELQKLYRQGVDSDELRALIRKLHALEQG (SEQ ID NO: 68), GDELTQLKRELQKLYRQGVDSDELRALIRKLDMLIRG (SEQ ID NO: 77), GDELTQLKRELQKLYRQGVDSDELEALKRKLDMLLHG (SEQ ID NO: 78), GDELHQLRRELQKLYRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 79), GDELKQLRRELQKLYHQGVDSDELMALIEKLIMLTHG (SEQ ID NO: 80), GDELTQLRRELQKLHRQGVDSDELRALIRKLHMLEHG (SEQ ID NO: 81), GDELTQLRRELQKLHRQGVDSDELRALIRKLHMLEQG (SEQ ID
- said additional moiety is a selected from the group consisting of a drug, a therapeutic agent, an antibody or antibody fragment, a peptide, a polypeptide, a cytotoxic molecule, a cytokine, an enzyme, a moiety being capable of binding a metal ion, a tag suitable for detection and/or purification, a homo- or hetero-association domain, a moiety which increases solubility of a protein, a moiety which comprises an enzymatic cleavage site, or a lipid, a liposome and a virus-like-particle.
- said additional moiety is fused to the isolated polypeptide specific for IL-13Ra2 via a peptide tether.
- said fusion molecule is capable of mediating killing of IL-13Ra2 expressing cells.
- said additional moiety is a radionuclide, a chemotherapeutic agent, a cytotoxic agent or a detectable group.
- said additional moiety is an antibody or antibody fragment.
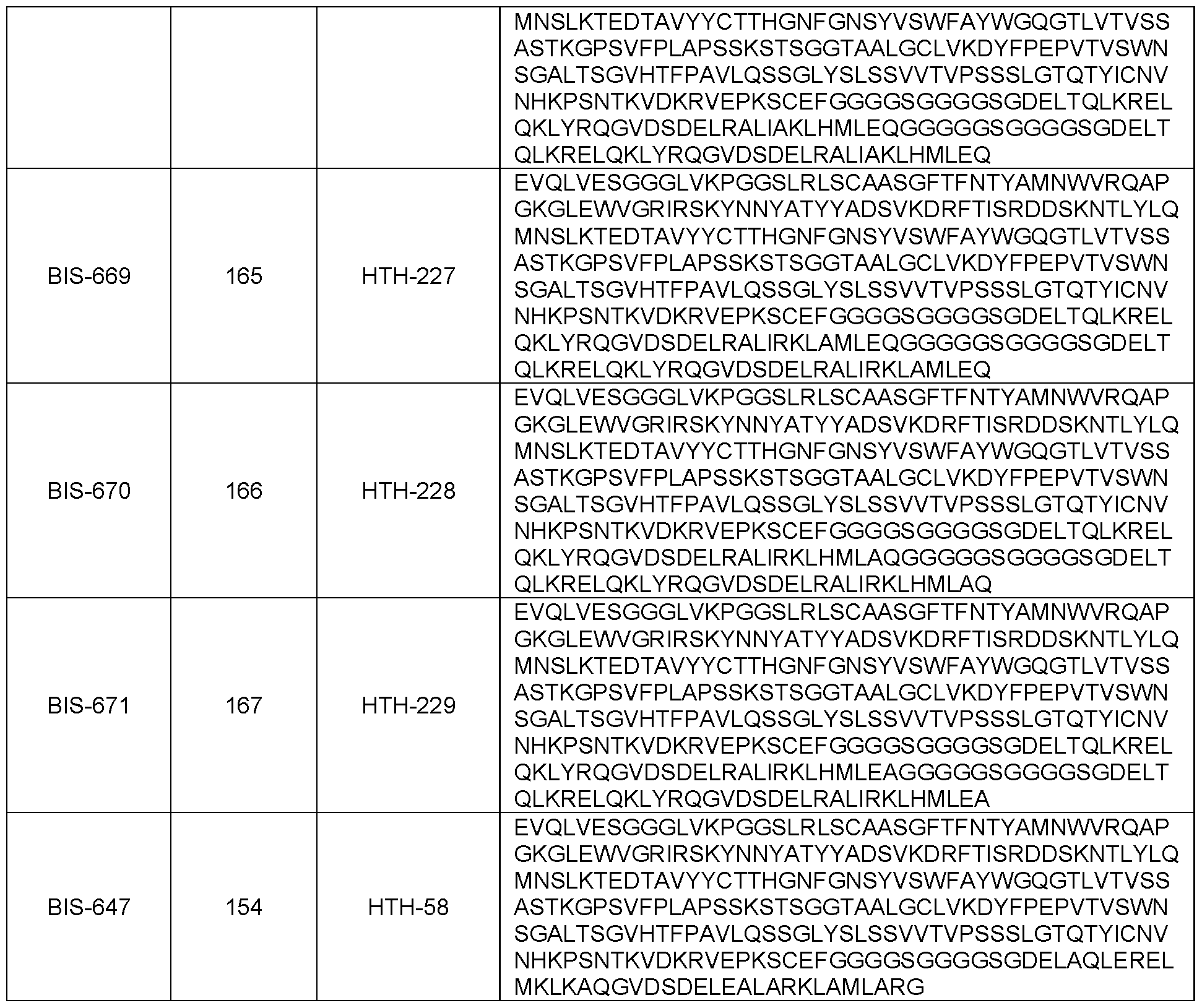
- said additional moiety is an antibody or antibody fragment specific for CD3.
- said fusion molecule is capable of re-directing cytotoxic activity of a T-cell to an IL-13Ra2 expressing cell.
- said antibody or antibody fragment specific for CD3 comprises a VH comprising the amino acid sequence of SEQ ID NO: 180 and a VL comprising the amino acid sequence of SEQ ID NO: 179.
- the present disclosure also provides the polypeptide or the fusion molecule according to the present disclosure for use in medicine.
- the present disclosure provides a nucleic acid composition comprising a nucleic acid sequence or a plurality of nucleic acid sequences encoding the polypeptide or the fusion molecules according to the present disclosure.
- the present disclosure provides a vector composition comprising a vector or a plurality of vectors comprising the nucleic acid composition according to the present disclosure.
- the present disclosure provides a host cell comprising the vector composition or the nucleic acid composition according to the present disclosure.
- the present disclosure also provides a pharmaceutical composition comprising the polypeptide or the fusion molecule according to the present disclosure and a pharmaceutically acceptable carrier or excipient.
- the present disclosure also provides methods for treating a subject suffering from a disease, such as cancer, by administering to said subject an effective amount of a polypeptide or the fusion molecule or the pharmaceutical composition according to the present disclosure.
- said subject is a human.
- Figure 1 Amino acid sequence alignment of isolated IL-13Ra2 specific helix-turn-helix (HTH) polypeptides and generated variants. Variable positions (Var x ) of the employed phage-library are indicated in black. Invariant positions on Helix-1 and Helix-2 but bearing distinct amino acids residues on Helix- 1 and Helix-2 are denotes as “X n ”.
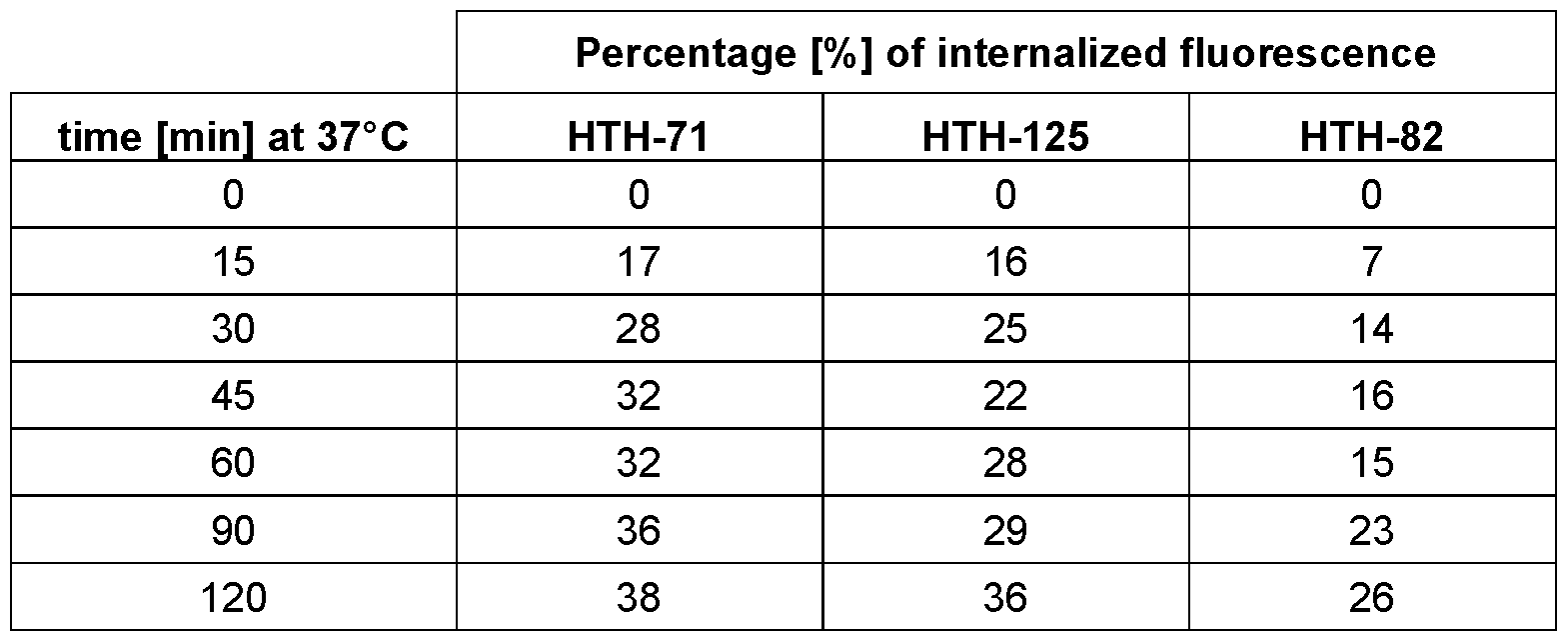
- Figure 2 Internalization rate of indirectly fluorescence-labeled IL-13Ra2 specific HTH- polypeptides after being exposed to CHO-cells stably expressing the cynomolgus IL-13Ra2 receptor.
- FIG. 3 Inhibition of in vitro cancer cell proliferation mediated by HTH polypeptide - MMAF drug conjugates (PDCs).
- A-375 (IL-13Ra2 positive) and A-549 (IL-13Ra2 negative) cancer cells were exposed to 3 different HTH-71 / MMAF drug conjugates.
- the number of viable cells was strongly reduced by the presence of PDCs in dose dependent manner whereas viability of A-549 cells was only affected at PDC concentrations of greater 100 nM.
- PDCs comprising two HTH-polypeptides (PDC-2, PDC-3) were significant more potent compared to PDC with only one HTH-polypeptide (PDC-1).
- Figure 4 Inhibition of in vitro cancer cell proliferation of A-375 cells mediated by two HTH polypeptide - MMAF drug conjugates (Figure A: PDC-1 ; Figure 4 B: PDC-2) in the presence of human IL-13.
- the inhibitory activity of both tested PDCs was reduced in presence of human IL-13 at IL-13 concentration of greater 50 pM.
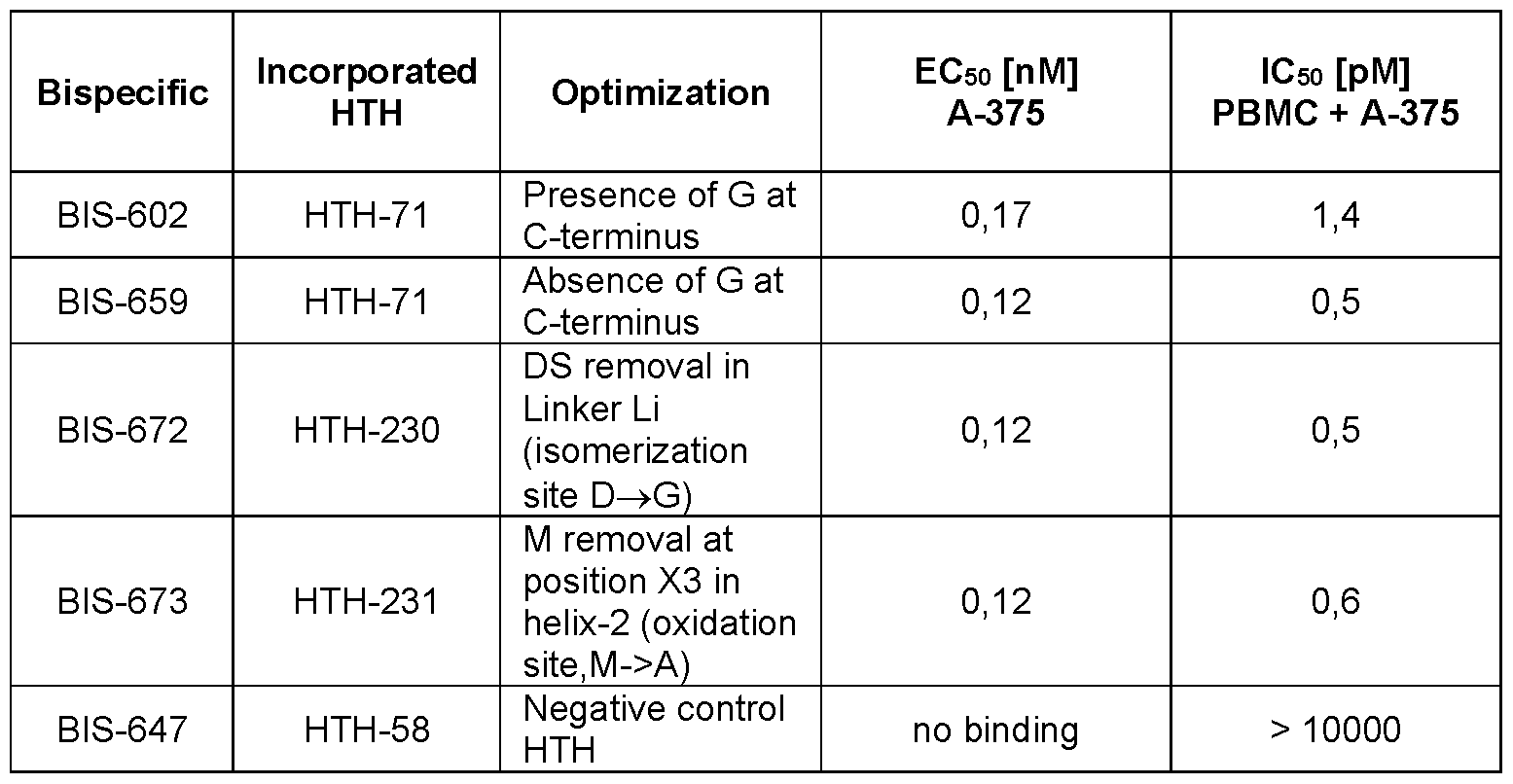
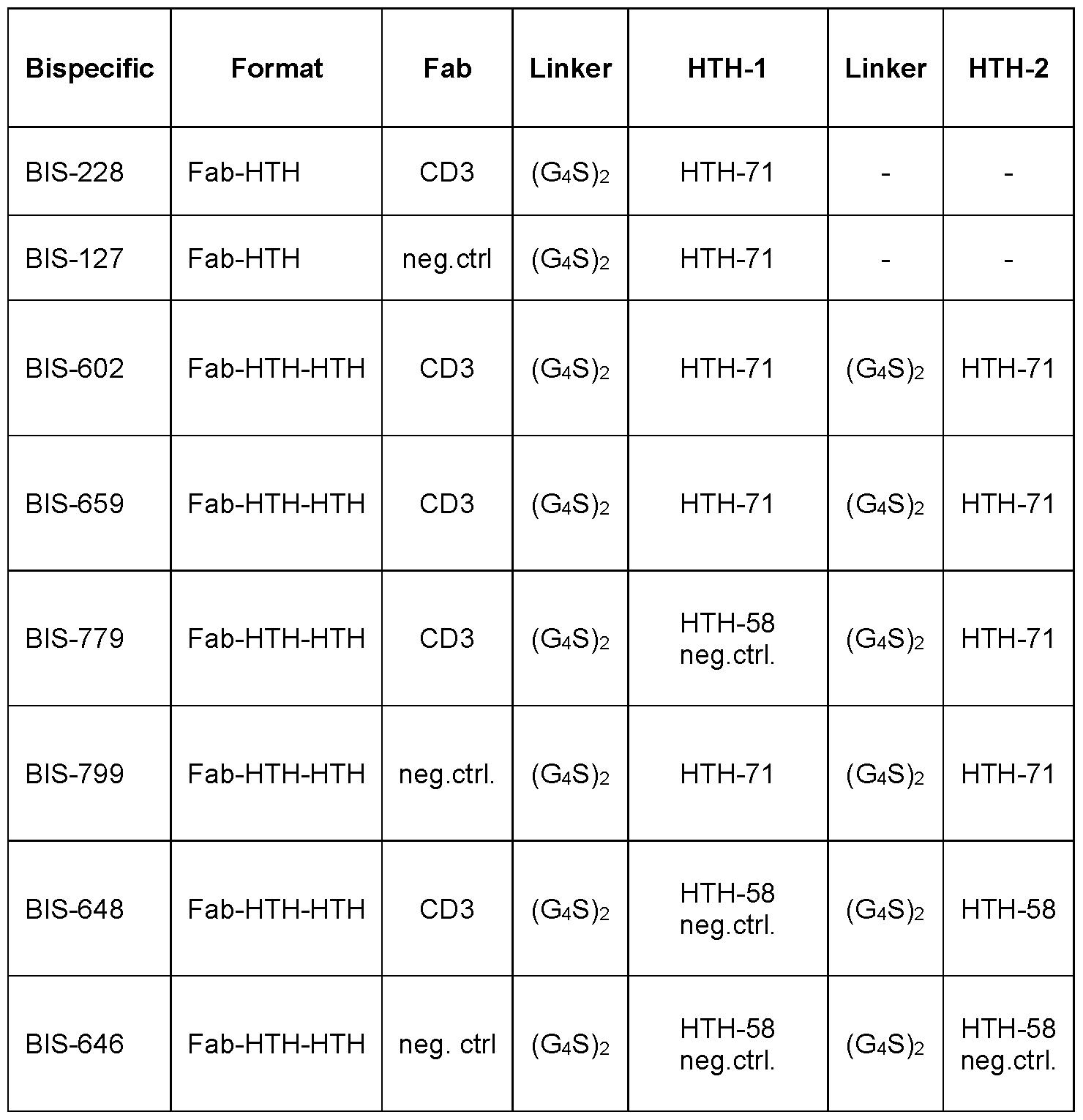
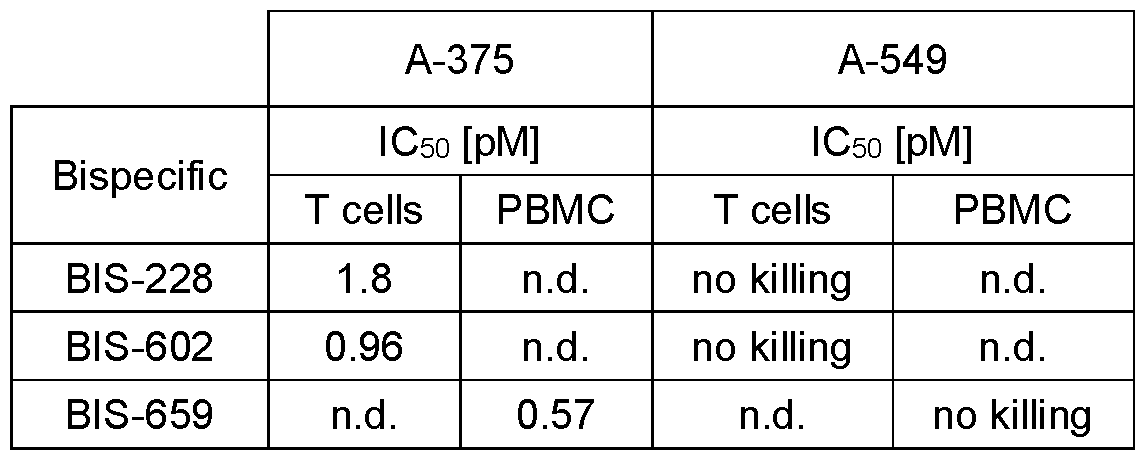
- Figure 5 Cytotoxicity assay of bispecific Fab-HTH polypeptide fusion molecules comprising a CD3 specific Fab and IL-13Ra2 specific HTH polypeptides according to the present disclosure on IL-13Ra2 expressing A-375 cells in presence of human derived T-cells or PBMCs.
- Figure 5A is a graph showing the relative fluorescence of A-375 cells as a function of BIS228 or BIS602 concentration in presence of human T-cells.
- Figure 5B indicates the same as Figure 5A but with results obtained for BIS659 in the presence of human PBMCs derived from one donor.
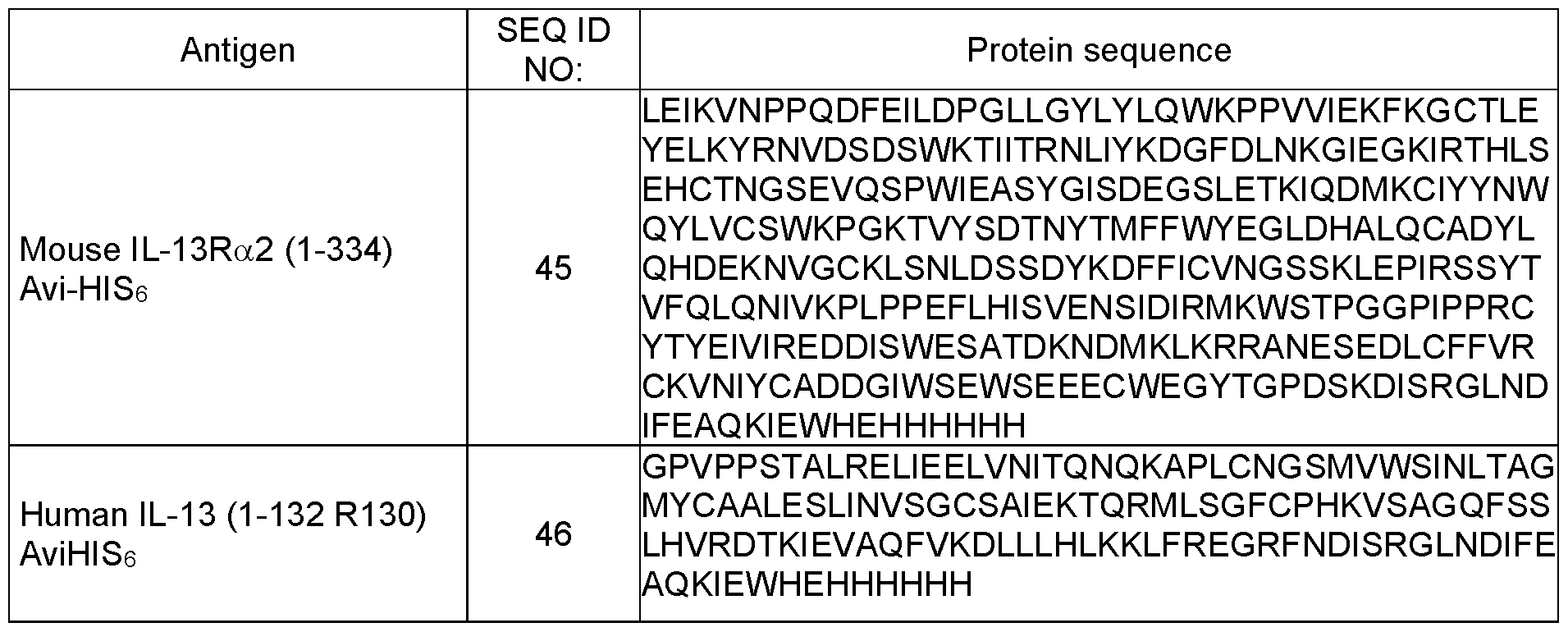
- IL-13Ra2 refers to a protein known as IL-13 receptor alpha 2.
- Human IL-13Ra2 (1-380) has the amino acid sequence of (Uniprot: Q14627):
- Cynomolgus monkey IL-13Ra2 (1-380) has the amino acid sequence of:
- Murine IL-13Ra2 (1-383) has the amino acid sequence of (UniProt: 088786):
- IL-13Ra1 refers to a protein known as IL-13 receptor alpha 1.
- Human IL-13Ra1 (1-427) has the amino acid sequence of (Uniprot: P78552):
- Mouse IL-13Ra1 (1-424) has the amino acid sequence of (Uniprot: 009030): MARPALLGELLVLLLWTATVGQVAAATEVQPPVTNLSVSVEN LCTI I WTWSPPEGAS PNCTLRYFSHFDDQQDKKIAPETHRKEELPLDEKICLQVGSQCSANESEKPSPLVKK CISPPEGDPESAVTELKCIWHNLSYMKCSWLPGRNTSPDTHYTLYYWYSSLEKSRQ CENIYREGQHIACSFKLTKVEPSFEHQNVQIMVKDNAGKIRPSCKIVSLTSYVKPDPP HIKHLLLKNGALLVQWKNPQNFRSRCLTYEVEVNNTQTDRHNILEVEEDKCQNSES DRNMEGTSCFQLPGVLADAVYTVRVRVKTNKLCFDDNKLWSDWSEAQSIGKEQNS TFYTTMLLTIPVFVAVAVIILLFYLKRLKIIIFPPIPDPG
- IL-13 refers to a protein known as Interleukin-13.
- Human IL-13 (1-146) has the amino acid sequence of (Uniprot: P35225):
- IL-4Ra refers to a protein known as lnterleukin-4 receptor subunit alpha.
- Human IL-4Ra (1-825) has the amino acid sequence of (Uniprot: P24394):
- Mouse IL-4Ra (1-810) has the amino acid sequence of (Uniprot: P16382):
- peptide is meant a short molecule having less than or equal to 20 amino acids.
- polypeptide means a molecule having more than 20 amino acids.
- a “fusion molecule” as meant herein refers to a polypeptide according to the present disclosure linked to at least further moiety. Such moiety may have a different property compared to the polypeptide. Such property may be a biological property, such as activity in vitro or in vivo. The property may also be a simple chemical or physical property, such as binding to a target molecule, catalysis of a reaction, etc..
- the fusion or conjugation may be covalent or non-covalent. Preferably, the fusion or conjugation is covalent.
- helix-turn-helix scaffold refers to a secondary structure of a polypeptide in which two a-helices are orientated in a parallel or an anti-parallel orientation, and in which the two a -helices are linked via a short stretch of amino acids.
- coiled-coil and “coiled-coil structure” are used interchangeably herein and will be clear to the person skilled in the art based on the common general knowledge and the description and further references cited herein.
- a coiled-coil structure is used by nature to stabilize a-helices in proteins.
- a coiled-coil is a structural motif in polypeptides or proteins in which 2 to 7 a-helices are coiled together.
- the coiled-coil formation of a-helical peptides is facilitated through a burial of hydrophobic side chains by arranging them on one side of the a-helices so that they are not accessed by polar water molecules.
- a typical coiled- coil motif (4-3 hydrophobic repeat) is a heptad repeat of amino acids from ‘a’ to ‘g’ so that ‘a’ and ‘d’ are hydrophobic.
- Particular reference in this regard is made to review papers concerning coiled-coil structures, such as for example, Cohen and Parry Proteins 1990, 7:1- 15; Kohn and Hodges Trends Biotechnol 1998, 16:379-389; Schneider et al Fold Des 1998, 3:R29-R40; Harbury et al.
- the tertiary structure of a a-helix is such that 7 amino acid residues in the primary sequence correspond to approximately 2 turns of the a-helix. Accordingly, a primary amino acid sequence giving rise to a a-helical conformation may be broken down into units of 7 residues each.
- the individual positions of a heptad unit are denoted by small letters, i.e. one heptad unit is for example represented by the sequence ‘abcdefg’, ‘bcdefga’, ‘cdefgab’, ‘defgabc’, ‘efgabcd’, ‘fgabcde’ or ‘gabcdef’.
- the ‘a’ and the ‘d’ position of a heptad unit assembled in an HTH scaffold of the present disclosure are of hydrophobic nature. These positions are typically either leucine, isoleucine or valine, and the parallel or the anti-parallel secondary structure of the HTH are formed by hydrophobic interactions via these positions between different heptad units present on two distinct a-helical peptides.
- anti-parallel refers to an HTH scaffold in which two a-helical peptides of an HTH scaffold are arranged such that the amino-terminal end (N-terminus) of one a-helical peptide is aligned with the carboxyl-terminal end (C-terminus) of the second a- helical peptide, and vice versa.
- N-terminus amino-terminal end
- C-terminus carboxyl-terminal end
- the heptad positions of a first helix is defined as ‘abcdefg’ as read from the amino- to the carboxyl-terminus
- the heptad positions of a second a-helix in an anti-parallel orientation would be defined as ‘gfedcba’ as read from the amino- to the carboxyl-terminus.
- parallel refers to an HTH scaffold in which the two a-helical peptides are aligned such that they have the same orientation such that the amino-terminal end of one helix is aligned with the amino-terminal end of the second a-helix, and vice versa.
- the relative orientation of the heptad ‘a-g’ positions of two interacting a-helical peptides aligned in parallel orientation is in the same direction.
- the heptad positions of a first helix is defined as ‘abcdefg’ from the amino-terminus to the carboxyl-terminus
- the heptad positions of a second helix in a parallel orientation would be also defined as ‘abcdefg’ as read from the amino- to the carboxyl-terminus.
- linker refers to an amino acid sequence that is part of the contiguous amino acid sequence of an HTH polypeptide, and covalently links the two a-helical peptide sequences of that polypeptide.
- single-chain refers to the HTH scaffold of the present disclosure, wherein the stabilizing coiled-coil structure is formed from different regions of a contiguous amino acid sequence of an HTH polypeptide chain folded back in an appropriate manner.
- solvent-oriented or “solvent-exposed” refers to the region of an entity which is directly exposed or which comes directly into contact with the solvent in the environment or the milieu in which it is present. In the context of the present disclosure it is the a -helix or a a- helical part of an HTH scaffold which is directly exposed or which comes directly into contact with the solvent in the environment or the milieu in which it is present. More particularly, in the context of a binding site, where one or more amino acids located in a solvent-oriented part of the HTH scaffold contribute to the binding site, the binding site is considered to be formed by a solvent-oriented part of the HTH scaffold.
- a “a-helical part” of a polypeptide refers to a part of a polypeptide of the present disclosure that has an a-helical secondary structure.
- the “hydrophobic core” of an HTH scaffold refers to the part on an HTH scaffold which is not directly exposed to the solvent in which it is present.
- a polypeptide of the present disclosure “binds specifically to”, “specifically binds to”, is “specific to/for” an antigen, if such polypeptide is able to discriminate between such antigen and one or more reference antigen(s), since binding specificity is not an absolute, but a relative property.
- the reference antigen(s) may be one or more closely related antigen(s), which are used as reference points.
- specific binding can be determined with a standard ELISA assay.
- Alternative methods comprise, but are not limited to Western blots, ELISA-, RIA-, ECL-, IRMA-tests and peptide scans. The scoring may be carried out by standard color development (e.g.
- binding specificity is performed by using not a single reference antigen, but a set of about three to five unrelated antigens, such as milk powder, BSA, transferrin or the like. Additionally, “specific binding” may relate to the ability to discriminate between different parts of its target antigen, e.g. different domains or regions of said target antigen, or between one or more key amino acid residues or stretches of amino acid residues of a target antigen.
- the “affinity” of a polypeptide is represented by the equilibrium constant for the dissociation of the polypeptide and the target protein of interest to which it binds. The lower the KD value, the stronger the binding strength between the said polypeptide and the target protein of interest to which it binds.
- the affinity can be expressed in terms of the affinity constant (KA), which corresponds to 1/KD.
- KA affinity constant
- the binding affinity of a polypeptide can be determined in a manner known to the skilled person, depending on the specific target protein of interest.
- the KD can be expressed as the ratio of the dissociation rate constant of a complex, denoted as koff (expressed in seconds -1 or s -1 ), to the rate constant of its association, denoted kon (expressed in molar -1 seconds -1 or M -1 s -1 ).
- koff expressed in seconds -1 or s -1
- kon expressed in molar -1 seconds -1 or M -1 s -1
- a KD value greater than about 1 millimolar is considered to indicate non-binding or non-specific binding.
- inhibitors or “inhibit” or “reduction” or “reduce” refer to a decrease or cessation of any phenotypic characteristic (such as binding or a biological activity or function) or to the decrease or cessation in the incidence, degree, or likelihood of that characteristic.
- the “inhibition”, “reduction” or “neutralization” needs not to be complete as long as it is detectable using an appropriate assay.
- by “reduce” or “inhibit” or “neutralize” is meant the ability to cause a decrease of 20% or greater.
- by “reduce” or “inhibit” or “neutralize” is meant the ability to cause a decrease of 50% or greater.
- inhibiting activity of a polypeptide of the present disclosure may be reversible or irreversible, but for pharmaceutical and pharmacological applications will typically occur reversibly.
- the inhibiting activity of a polypeptide of the present disclosure may be measured using a suitable in vitro, cellular or in vivo assay.
- EC50 refers to the concentration of an antibody or antibody fragment or ligand, which induces a response in an assay half way between the baseline and maximum. It therefore represents the antibody or ligand concentration at which 50% of the maximal effect is observed
- IC50 refers to the concentration of an antibody or antibody fragment that inhibits a response in an assay half way between the maximal response and the baseline. It represents the antibody concentration that reduces a given response by 50%.
- synthetic describes a molecule that is made outside of the human body by synthesis or synthesized.
- isolated refers to a compound which can be e.g. a polypeptide according to the present disclosure, that is substantially free of other polypeptides having different antigenic specificities. Moreover, an isolated polypeptide may be substantially free of other cellular material and/or chemicals.
- Consstrained refers to a polypeptide in which the three- dimensional structure is maintained substantially in one spatial arrangement over time. The polypeptides within the present disclosure have a constrained conformation. Methods of determining whether peptides or polypeptides are constrained are known in the art.
- amino acid residues will be indicated either by their full name or according to the standard three-letter or one-letter amino acid code. “Natural occurring amino acids” means the following amino acids:
- binding region refers to a particular site, part, domain or stretch of amino acid residues present on the polypeptides of the present disclosure that is responsible for binding to a target molecule.
- binding region consists of specific amino acids from the said polypeptide which are in contact with the target molecule.
- target antigen e.g. IL-13Ra2
- binding region consists of specific amino acids from said target molecule, which are in contact with the polypeptides of the present disclosure.
- vector refers to a polynucleotide molecule capable of transporting another polynucleotide to which it has been linked.
- Preferred vectors are those capable of autonomous replication and/or expression of nucleic acids to which they are linked.
- plasmid refers to a circular double stranded DNA loop into which additional DNA segments may be ligated.
- viral vector Another type of vector is a viral vector, wherein additional DNA segments may be ligated into the viral genome.
- Certain vectors are capable of autonomous replication in a host cell into which they are introduced (e.g., bacterial vectors having a bacterial origin of replication and mammalian vectors).
- vectors can be integrated into the genome of a host cell upon introduction into the host cell, and thereby are replicated along with the host genome.
- Vectors may be compatible with prokaryotic or eukaryotic cells.
- Prokaryotic vectors typically include a prokaryotic replicon which may include a prokaryotic promoter capable of directing the expression (transcription and translation) of the peptide in a bacterial host cell, such as Escherichia coli transformed therewith.
- a promoter is an expression control element formed by a DNA sequence that permits binding of RNA polymerase and transcription to occur.
- Promoter sequences compatible with bacterial hosts are typically provided in plasmid vectors containing convenience restriction sites for insertion of a DNA segment.
- vector plasmids examples include pUC8, pUC9, pBR322, and pBR329, pPL and pKK223, available commercially.
- "Expression vectors” are those vectors capable of directing the expression of nucleic acids to which they are operatively linked and is intended to include such other forms of expression vectors, such as viral vectors (e.g., replication defective retroviruses, adenoviruses and adeno-associated viruses), which serve equivalent functions.
- viral vectors e.g., replication defective retroviruses, adenoviruses and adeno-associated viruses
- recombinant host cell refers to a cell into which a recombinant expression vector has been introduced. It should be understood that such terms are intended to refer not only to the particular subject cell but to the progeny of such a cell. Because certain modifications may occur in succeeding generations due to either mutation or environmental influences, such progeny may not, in fact, be identical to the parent cell, but are still included within the scope of the term “host cell” as used herein.
- Typical host cells are prokaryotic (such as bacterial, including but not limited to E. coli) or eukaryotic (which includes yeast, mammalian cells, and more).
- Bacterial cells are preferred prokaryotic host cells and typically are a strain of Escherichia coli (E. coli) such as, for example, the E. coli strain DH5 available from Bethesda Research Laboratories, Inc., Bethesda, Md.
- E. coli Escherichia coli
- Preferred eukaryotic host cells include yeast and mammalian cells including murine and rodents, preferably vertebrate cells such as those from a mouse, rat, monkey or human cell line, for example HKB11 cells, PERC.6 cells, or CHO cells.
- antibody refers to a protein comprising at least two heavy (H) chains and two light (L) chains inter-connected by disulfide bonds which interacts with an antigen.
- Each heavy chain is comprised of a heavy chain variable region (abbreviated herein as VH) and a heavy chain constant region.
- the heavy chain constant region is comprised of three domains, CH1 , CH2 and CH3.
- Each light chain is comprised of a light chain variable region (abbreviated herein as VL) and a light chain constant region.
- the light chain constant region is comprised of one domain, CL.
- VH and VL regions can be further subdivided into regions of hypervariability, termed complementarity determining regions (CDR), interspersed with regions that are more conserved, termed framework regions (FR).
- CDR complementarity determining regions
- FR framework regions
- Each VH and VL is composed of three CDRs and four FR’s arranged from amino-terminus to carboxy-terminus in the following order: FR1 , CDR1 , FR2, CDR2, FR3, CDR3, and FR4.
- the variable regions of the heavy and light chains contain a binding domain that interacts with an antigen.
- the constant regions of the antibodies may mediate the binding of the immunoglobulin to host tissues or factors, including various cells of the immune system (e.g., effector cells) and the first component (Clq) of the classical complement system.
- antibody includes for example, monoclonal antibodies, human antibodies, humanized antibodies, camelised antibodies and chimeric antibodies.
- the antibodies can be of any isotype (e.g., IgG, IgE, IgM, IgD, IgA and IgY), class (e.g., lgG1 , lgG2, lgG3, lgG-4, lgA1 and lgA2) or subclass. Both the light and heavy chains are divided into regions of structural and functional homology.
- antibody fragment refers to one or more portions of an antibody that retain the ability to specifically interact with (e.g., by binding, steric hindrance, stabilizing spatial distribution) an antigen.
- binding fragments include, but are not limited to, a Fab fragment, a monovalent fragment consisting of the VL, VH, CL and CH1 domains; a F(ab)2 fragment, a bivalent fragment comprising two Fab fragments linked by a disulfide bridge at the hinge region; a Fd fragment consisting of the VH and CH1 domains; a Fv fragment consisting of the VL and VH domains of a single arm of an antibody; a dAb fragment (Ward et al., (1989) Nature 341 :544-546), which consists of a VH domain; and an isolated complementarity determining region (CDR).
- a Fab fragment a monovalent fragment consisting of the VL, VH, CL and CH1 domains
- F(ab)2 fragment a bi
- the two domains of the Fv fragment, VL and VH are coded for by separate genes, they can be joined, using recombinant methods, by a synthetic linker that enables them to be made as a single protein chain in which the VL and VH regions pair to form monovalent molecules (known as single chain Fv (scFv); see e.g., Bird et al., (1988) Science 242:423-426; and Huston et al., (1988) Proc. Natl. Acad. Sci. 85:5879-5883).
- single chain Fv single chain Fv
- Such single chain antibodies are also intended to be encompassed within the term “antibody fragment”.
- Antibody fragments are obtained using conventional techniques known to those of skill in the art, and the fragments are screened for utility in the same manner as are intact antibodies.
- Antibody fragments can also be incorporated into single domain antibodies, maxibodies, minibodies, intrabodies, diabodies, triabodies, tetrabodies, v- NAR and bis-scFv (see, e.g., Hollinger and Hudson, (2005) Nature Biotechnology 23:1126- 1136).
- Antibody fragments can be grafted into scaffolds based on polypeptides such as Fibronectin type III (Fn3) (see U.S. Pat. No. 6,703,199, which describes fibronectin polypeptide monobodies).
- Fn3 Fibronectin type III
- Antibody fragments can be incorporated into molecules comprising a pair of tandem Fv segments (VH-CH1-VH-CH1) which, together with complementary light chain polypeptides, form a pair of antigen-binding sites (Zapata et al., (1995) Protein Eng. 8:1057- 1062; and U.S. Pat. No. 5,641 ,870).
- polypeptides as used herein, includes all polypeptides according to the present disclosure that are prepared, expressed, created or segregated by means not existing in nature.
- polypeptides isolated from a host cell transformed to express the polypeptides polypeptides selected and isolated from a recombinant, HTH polypeptide library, and polypeptides prepared, expressed, created or isolated by any other means.
- the polypeptides disclosed herein are isolated from the HTH polypeptide library described in WO 2017/149117.
- compositions may be used for therapeutic or prophylactic applications.
- the present disclosure therefore, includes a pharmaceutical composition containing an antibody or antibody fragment as disclosed herein and a pharmaceutically acceptable carrier or excipient therefor.
- the present disclosure provides a method for treating cancer. Such method contains the steps of administering to a subject in need thereof an effective amount of the pharmaceutical composition that contains an antibody or antibody fragment as described herein.
- the present disclosure provides therapeutic methods comprising the administration of a therapeutically effective amount of an antibody or antibody fragment as disclosed herein to a subject in need of such treatment.
- a "therapeutically effective amount” or “effective amount”, as used herein, refers to the amount of a polypeptide according to the present disclosure necessary to elicit the desired biological response.
- the therapeutic effective amount is the amount of a IL-13Ra2 specific polypeptide disclosed herein necessary to treat and/or prevent a disease.
- administering includes but is not limited to delivery of a drug by an injectable form, such as, for example, an intravenous, intramuscular, intradermal or subcutaneous route or mucosal route, for example, as a nasal spray or aerosol for inhalation or as an ingestible solution, capsule or tablet.
- an injectable form such as, for example, an intravenous, intramuscular, intradermal or subcutaneous route or mucosal route, for example, as a nasal spray or aerosol for inhalation or as an ingestible solution, capsule or tablet.
- the administration is by an injectable form.
- treatment refers to clinical intervention in an attempt to alter the natural course of a disease in the subject being treated, and can be performed either for prophylaxis or during the course of clinical pathology.
- Desirable effects of treatment include, but are not limited to, preventing occurrence or recurrence of disease, alleviation of symptoms, diminishment of any direct or indirect pathological consequences of the disease, preventing metastasis, decreasing the rate of disease progression, amelioration or palliation of the disease state, and remission or improved prognosis.
- antibodies or antibody fragments according to the preset disclosure are used to delay development of a disease or to slow the progression of a disease.
- Preventing refers to a reduction in risk of acquiring or developing a disease (i.e. causing at least one of the clinical symptoms of the disease not to develop in a subject that may be exposed to a disease-causing agent, or predisposed to the disease in advance of disease onset). “Prevention” also refers to methods which aim to prevent the onset of a disease or its symptoms or which delay the onset of a disease or its symptoms.
- Subject or “species” or as used in this context refers to any mammal, including rodents, such as mouse or rat, and primates, such as cynomolgus monkey (Macaca fascicularis), rhesus monkey (Macaca mulatta) or humans (Homo sapiens).
- rodents such as mouse or rat
- primates such as cynomolgus monkey (Macaca fascicularis), rhesus monkey (Macaca mulatta) or humans (Homo sapiens).
- the subject is a primate, most preferably a human.
- engineered or “modified” as used herein includes manipulation of nucleic acids or polypeptides by synthetic means (e.g. by recombinant techniques, in vitro peptide synthesis, by enzymatic or chemical coupling of peptides or some combination of these techniques).
- the polypeptides according to the present disclosure are engineered or modified to improve one or more properties, such as antigen binding, stability, half-life, effector function, immunogenicity, safety and the like.
- Variant refers to a polypeptide that differs from a reference polypeptide by one or more modifications for example amino acid substitutions, insertions or deletions.
- amino acid mutation as used herein is meant to encompass amino acid substitutions, deletions, insertions, and modifications. Any combination of substitution, deletion, insertion, and modification can be made as long as the final construct possesses the desired characteristics.
- Amino acid sequence deletions and insertions include N-and/or C- terminal deletions and insertions of amino acids. Particular amino acid mutations are amino acid substitutions.
- Amino acid substitutions include replacement by non-naturally occurring amino acids or by naturally occurring amino acid derivatives of the twenty standard amino acids.
- Amino acid mutations can be generated using genetic or chemical methods well known in the art. Genetic methods may include site-directed mutagenesis, PCR, gene synthesis and the like. It is contemplated that methods of altering the side chain group of an amino acid residue by methods other than genetic engineering, such as chemical modification, may also be useful.
- the present disclosure provides a polypeptide specific for IL-13 receptor alpha 2 (IL-13Ra2) comprising a helix-turn-helix scaffold structure of the formula Helix-1 - Li - Helix-2, wherein Li is a linker and wherein Helix-1 and Helix-2 comprise a first and second a-helical peptide, wherein each of said a-helical peptides comprises the amino acid sequence of
- Vari is any natural occurring amino acid excluding G, P and C in Helix-1 and Helix-2,
- Var2 is K, R, T or I in Helix 1 and I or K in Helix-2,
- Var3 is R or Q in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
- Var4 is Q or K in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
- Var5 is F, Y or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
- Var6 is K, R or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
- X1 is Q in Helix-1 and A in Helix-2
- X2 is E in Helix-1 and K in Helix-2
- X3 is K in Helix-1 and M or A in Helix-2
- X3 is M in Helix-2. In another embodiment, X3 is A in Helix-2. In an embodiment, X3 is K in Helix-1 and A in Helix-2. In an embodiment, X3 is K in Helix-1 and M in Helix-2
- Vari is any natural occurring amino acid excluding G, P C in Helix 1.
- Vari is A, K, H or T in Helix-1.
- Vari is K in Helix-1.
- Vari is H in Helix-1.
- Vari is T in Helix-1.
- Vari is A in Helix-1.
- Vari is any natural occurring amino acid excluding G, P C in Helix 2.
- Vari is A, R, E or M in Helix-2.
- Vari is A in Helix-2.
- Vari is R in Helix-2.
- Vari is E in Helix-2.
- Vari is M in Helix-2.
- Var2 is I, K, R or T in Helix-1. In an embodiment, Var2 is I in Helix-1. In an embodiment, Var2 is R in Helix-1. In an embodiment, Var2 is T in Helix-1. In an embodiment, Var2 is K in Helix 1. In an embodiment, Var2 is I or K in Helix-2. In an embodiment, Var2 is I in Helix-2. In an embodiment, Var2 is K in Helix-2.
- Var3 is R or Q in Helix-1. In an embodiment, Var3 is Q in Helix-1. In an embodiment, Var3 is R in Helix-1. In an embodiment, Var3 is any natural occurring amino acid excluding G, P C in Helix-2. In an embodiment, Var3 is A, R, E or Q in Helix-2. In an embodiment, Var3 is R in Helix-2. In an embodiment, Var3 is Q in Helix-2. In an embodiment, Var3 is E in Helix-2, In an embodiment, Var3 is A in Helix-2.
- Var4 is K or Q in Helix-1. In an embodiment, Var4 is K in Helix-1. In an embodiment, Var4 is Q in Helix-1. In an embodiment, Var4 is any natural occurring amino acid excluding G, P C in Helix-2. In an embodiment, Var4 is Var4 is A, H, D, I, or E in Helix-2. In an embodiment, Var4 is H in Helix- 2, In an embodiment, Var4 is D in Helix-2. In an embodiment, Var4 is I in Helix-2. In an embodiment, Var4 is E in Helix-2. In an embodiment, Var4 is A in Helix- 2.
- Var5 is F, Y or H in Helix-1. In an embodiment, Var5 is F in Helix-1. In an embodiment, Var5 is Y in Helix-1. In an embodiment, Var5 is H in Helix-1. In an embodiment, Var5 is any natural occurring amino acid excluding G, P C in Helix-2. In an embodiment, Var5 is A, E, I, T, L, K or D in Helix-2. In an embodiment, Var5 is E or I in Helix-2. In an embodiment, Var5 is I in Helix-2. In an embodiment, Var5 is T in Helix-2. In an embodiment, Var5 is L in Helix-2. In an embodiment, Var5 is K in Helix-2. In an embodiment, Var5 is D in Helix-2. In an embodiment, Var5 is A in Helix-2.
- Var6 is K, R or H in Helix-1. In an embodiment, Var6 is K in Helix-1. In an embodiment, Var6 is R in Helix-1. In an embodiment, Var6 is H in Helix-1. In an embodiment. Var6 is any natural occurring amino acid excluding G, P C in Helix-2. In an embodiment, Var6 is A, H, Q or R in Helix-2. In an embodiment, Var6 is H in Helix-2. In an embodiment, Var6 is R in Helix-2. In an embodiment, Var6 is Q in Helix-2. In an embodiment, Var6 is A in Helix-2.
- first and second a-helical peptide comprised in the helix-turn- helix scaffold structure form an antiparallel coiled-coiled structure.
- first a-helical peptide is comprised in Helix-1 and the second a-helical peptide is comprised in Helix-2.
- Helix-1 and Helix-2 comprised in the helix-turn-helix scaffold structure form an antiparallel coiled-coiled structure.
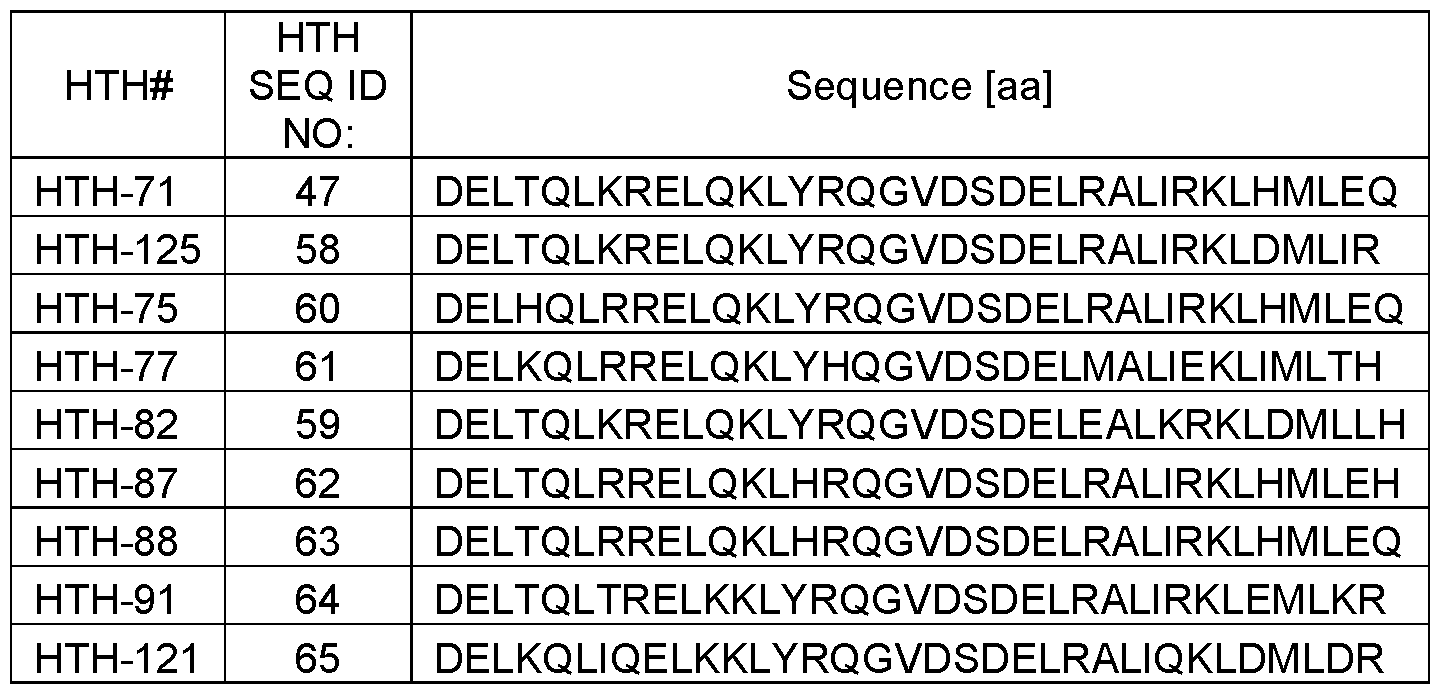
- said first a-helical peptide comprises an amino sequence selected from the group consisting of: DELTQLKRELQKLYR (SEQ ID NO: 1), DELAQLKRELQKLYR (SEQ ID NO: 2), DELTQLKRELQKLYK (SEQ ID NO: 3), DELTQLKRELQKLFR (SEQ ID NO: 4), DELHQLRRELQKLYR (SEQ ID NO: 5), DELKQLRRELQKLYH (SEQ ID NO: 6), DELTQLRRELQKLHR (SEQ ID NO: 7), DELTQLTRELKKLYR (SEQ ID NO: 8), and DELKQLIQELKKLYR (SEQ ID NO: 9).
- said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQ (SEQ ID NO: 19), DELRALIRKLHALEQ (SEQ ID NO: 20), DELAALIRKLHMLEQ (SEQ ID NO: 21), DELRALIAKLHMLEQ (SEQ ID NO: 22), DELRALIRKLAMLEQ (SEQ ID NO: 23), DELRALIRKLHMLAQ (SEQ ID NO: 24),DELRALIRKLHMLEA (SEQ ID NO: 25), DELRALIRKLDMLIR (SEQ ID NO: 26), DELEALKRKLDMLLH (SEQ ID NO: 27), DELMALIEKLIMLTH (SEQ ID NO: 28), DELRALIRKLHMLEH (SEQ ID NO: 29), DELRALIRKLEMLKR (SEQ ID NO: 30), and DELRALIQKLDMLDR (SEQ ID NO: 31).
- said first a-helical peptide comprises an amino sequence selected from the group consisting of: DELTQLKRELQKLYR (SEQ ID NO: 1), DELAQLKRELQKLYR (SEQ ID NO: 2), DELTQLKRELQKLYK (SEQ ID NO: 3), DELTQLKRELQKLFR (SEQ ID NO: 4), DELHQLRRELQKLYR (SEQ ID NO: 5), DELKQLRRELQKLYH (SEQ ID NO: 6),
- DELKQLIQELKKLYR SEQ ID NO: 9
- said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQ (SEQ ID NO: 19), DELRALIRKLHALEQ (SEQ ID NO: 20), DELAALIRKLHMLEQ (SEQ ID NO: 21),
- DELRALIRKLHMLAQ (SEQ ID NO: 24), DELRALIRKLHMLEA (SEQ ID NO: 25),
- DELRALIRKLDMLIR SEQ ID NO: 26
- DELEALKRKLDMLLH SEQ ID NO: 27
- DELRALIRKLEMLKR SEQ ID NO: 30
- DELRALIQKLDMLDR SEQ ID NO: 31
- said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQ (SEQ ID NO: 19),
- DELRALIRKLHALEQ SEQ ID NO: 20
- DELAALIRKLHMLEQ SEQ ID NO: 21
- DELRALIRKLHMLAQ (SEQ ID NO: 24), DELRALIRKLHMLEA (SEQ ID NO: 25),
- DELRALIRKLDMLIR SEQ ID NO: 26
- DELEALKRKLDMLLH SEQ ID NO: 27
- said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
- said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHALEQ (SEQ ID NO: 20).
- said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELAALIRKLHMLEQ (SEQ ID NO: 21).
- said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELRALIAKLHMLEQ (SEQ ID NO: 22).
- said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELRALIRKLAMLEQ (SEQ ID NO: 23).
- said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLAQ (SEQ ID NO: 24).
- said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEA (SEQ ID NO: 25).
- said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELRALIRKLDMLIR (SEQ ID NO: 26).
- said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELEALKRKLDMLLH (SEQ ID NO: 27).
- said first a-helical peptide comprises the amino sequence of DELAQLKRELQKLYR (SEQ ID NO: 2) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
- said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYK (SEQ ID NO: 3) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
- said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLFR (SEQ ID NO: 4) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
- said first a-helical peptide comprises the amino sequence of DELHQLRRELQKLYR (SEQ ID NO: 5) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
- said first a-helical peptide comprises the amino sequence of DELKQLRRELQKLYH (SEQ ID NO: 6) and said second a-helical peptide comprises the amino sequence of DELMALIEKLIMLTH (SEQ ID NO: 28).
- said first a-helical peptide comprises the amino sequence of DELTQLRRELQKLHR (SEQ ID NO: 7) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEH (SEQ ID NO: 29) or DELRALIRKLHMLEQ (SEQ ID NO: 19).
- said first a-helical peptide comprises the amino sequence of DELTQLRRELQKLHR (SEQ ID NO: 7) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEH (SEQ ID NO: 29).
- said first a-helical peptide comprises the amino sequence of DELTQLRRELQKLHR (SEQ ID NO: 7) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
- said first a-helical peptide comprises the amino sequence of DELTQLTRELKKLYR (SEQ ID NO: 8) and said second a-helical peptide comprises the amino sequence of DELRALIRKLEMLKR (SEQ ID NO: 30).
- said first a-helical peptide comprises the amino sequence of DELKQLIQELKKLYR (SEQ ID NO: 9) and said second a-helical peptide comprises the amino sequence of DELRALIQKLDMLDR (SEQ ID NO: 31).
- said first a-helical peptide comprises an amino sequence selected from the group consisting of: DELTQLKRELQKLYR (SEQ ID NO: 1), DELAQLKRELQKLYR (SEQ ID NO: 2), DELTQLKRELQKLYK (SEQ ID NO: 3), DELTQLKRELQKLFR (SEQ ID NO: 4), DELHQLRRELQKLYR (SEQ ID NO: 5), and DELTQLRRELQKLHR (SEQ ID NO: 7) and said second a-helical peptide comprises the amino sequence of: DELRALIRKLHMLEQ (SEQ ID NO: 19).
- said Helix-1 or said first a-helical peptide comprises an amino sequence selected from the group consisting of: DELTQLKRELQKLYR (SEQ ID NO: 1),
- DELAQLKRELQKLYR SEQ ID NO: 2
- DELTQLKRELQKLYK SEQ ID NO: 3
- DELTQLTRELKKLYR SEQ ID NO: 8
- DELKQLIQELKKLYR SEQ ID NO: 9
- said Helix-2 or said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQ (SEQ ID NO: 19),
- DELRALIRKLHALEQ SEQ ID NO: 20
- DELAALIRKLHMLEQ SEQ ID NO: 21
- DELRALIRKLHMLAQ (SEQ ID NO: 24),DELRALIRKLHMLEA (SEQ ID NO: 25), DELRALIRKLDMLIR (SEQ ID NO: 26), DELEALKRKLDMLLH (SEQ ID NO: 27),
- DELMALIEKLIMLTH SEQ ID NO: 28
- DELRALIRKLHMLEH SEQ ID NO: 29
- DELRALIRKLEMLKR SEQ ID NO: 30
- DELRALIQKLDMLDR SEQ ID NO: 31
- said Helix-1 or said first a-helical peptide comprises an amino sequence selected from the group consisting of: DELTQLKRELQKLYR (SEQ ID NO: 1),
- DELAQLKRELQKLYR SEQ ID NO: 2
- DELTQLKRELQKLYK SEQ ID NO: 3
- DELTQLKRELQKLFR (SEQ ID NO: 4), DELHQLRRELQKLYR (SEQ ID NO: 5), DELKQLRRELQKLYH (SEQ ID NO: 6), DELTQLRRELQKLHR (SEQ ID NO: 7), DELTQLTRELKKLYR (SEQ ID NO: 8), DELKQLIQELKKLYR (SEQ ID NO: 9), and said Helix- 2 or said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQ (SEQ ID NO: 19), DELRALIRKLHALEQ (SEQ ID NO: 20), DELAALIRKLHMLEQ (SEQ ID NO: 21), DELRALIAKLHMLEQ (SEQ ID NO: 22), DELRALIRKLAMLEQ (SEQ ID NO: 23), DELRALIRKLHMLAQ (SEQ ID NO: 24),
- DELRALIRKLHMLEA SEQ ID NO: 25
- DELRALIRKLDMLIR SEQ ID NO: 26
- DELRALIRKLHMLEH (SEQ ID NO: 29), DELRALIRKLEMLKR (SEQ ID NO: 30), and
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQ (SEQ ID NO: 19), DELRALIRKLHALEQ (SEQ ID NO: 20), DELAALIRKLHMLEQ (SEQ ID NO: 21), DELRALIAKLHMLEQ (SEQ ID NO: 22), DELRALIRKLAMLEQ (SEQ ID NO: 23),
- DELRALIRKLHMLAQ (SEQ ID NO: 24), DELRALIRKLHMLEA (SEQ ID NO: 25),
- DELRALIRKLDMLIR SEQ ID NO: 26
- DELEALKRKLDMLLH SEQ ID NO: 27
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHALEQ (SEQ ID NO: 20).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELAALIRKLHMLEQ (SEQ ID NO: 21).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIAKLHMLEQ (SEQ ID NO: 22).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLAMLEQ (SEQ ID NO: 23).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLAQ (SEQ ID NO: 24).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEA (SEQ ID NO: 25).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLDMLIR (SEQ ID NO: 26).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELEALKRKLDMLLH (SEQ ID NO: 27).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELAQLKRELQKLYR (SEQ ID NO: 2) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYK (SEQ ID NO: 3) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLFR (SEQ ID NO: 4) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELHQLRRELQKLYR (SEQ ID NO: 5) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELKQLRRELQKLYH (SEQ ID NO: 6) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELMALIEKLIMLTH (SEQ ID NO: 28).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLRRELQKLHR (SEQ ID NO: 7) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEH (SEQ ID NO: 29) or DELRALIRKLHMLEQ (SEQ ID NO: 19).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLRRELQKLHR (SEQ ID NO: 7) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEH (SEQ ID NO: 29).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLRRELQKLHR (SEQ ID NO: 7) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLTRELKKLYR (SEQ ID NO: 8) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLEMLKR (SEQ ID NO: 30).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of DELKQLIQELKKLYR (SEQ ID NO: 9) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIQKLDMLDR (SEQ ID NO: 31).
- said Helix-1 or said first a-helical peptide comprises an amino sequence selected from the group consisting of: DELTQLKRELQKLYR (SEQ ID NO: 1),
- DELAQLKRELQKLYR SEQ ID NO: 2
- DELTQLKRELQKLYK SEQ ID NO: 3
- DELTQLKRELQKLFR (SEQ ID NO: 4), DELHQLRRELQKLYR (SEQ ID NO: 5), and DELTQLRRELQKLHR (SEQ ID NO: 7) and said Helix-2 or said second a-helical peptide comprises the amino sequence of: DELRALIRKLHMLEQ (SEQ ID NO: 19).
- first and second a-helical peptide form an antiparallel coiled-coiled structure.
- Helix-1 and Helix-2 form an antiparallel coiled-coiled structure.
- the linker Li has a length of 1 to 50 amino acid residues. In another embodiment, the linker Li has a length of 1-30 amino acid residues. Yet in another embodiment, the linker Li has a length of 1-10 amino acid residues. In one embodiment, the linker Li has a length of 5 amino acids. In yet a further aspect of the present disclosure the linker Li comprises 1 to 30 amino acid residues. In an embodiment, the linker Li comprises the amino acid sequence QGVDS (SEQ ID NO: 182). In one embodiment, the linker Li comprises the amino acid sequence QGVGS (SEQ ID NO: 183). In one embodiment, the linker Li consists of the amino acid sequence QGVDS (SEQ ID NO: 182).
- the linker Li consists of the amino acid sequence or QGVGS (SEQ ID NO: 183). Amino acid residues may also be replaced, deleted or added, for example to aid in the expression of library members in a preferred host species, to facilitate cloning of the molecule, to increase the stability of the polypeptide or to increase helix packing and the like.
- G added to or being present at the N- and/or C-terminus of the Helix-turn-Helix scaffold structure may serve as a helix-breaker in situations where the Helix-turn-Helix scaffold structure is genetically fused to other moieties, such as affinity- or detection tags.
- it’s absence may be desired in situations where no moiety is fused to the HTH-scaffold structure.
- mass spectrometry analysis of HTH polypeptides bearing a C-terminal accessible G revealed that this terminal amino acid is partly removed by proteases during mammalian production (data not shown). Thus, removal of this G may be desirable to increase the homogeneity of recombinantly produced product.
- additional amino acid residues are added to the N- terminus and/or the C-terminus of the helix-turn-helix scaffold.
- a G is added to the N-terminus of the helix-turn- helix scaffold structure. In an embodiment of the present disclosure, a G is added to the C- terminus of the helix-turn-helix scaffold structure. In an embodiment of the present disclosure, a G is added to the N-terminus of the helix-turn-helix scaffold and a G is added to the C- terminus of the helix-turn-helix scaffold. In an embodiment of the present disclosure, a G is present at the N-terminus of the helix-turn-helix scaffold structure. In an embodiment of the present disclosure, a G is present at the C-terminus of the helix-turn-helix scaffold structure.
- a G is present at the N-terminus of the helix-turn- helix scaffold and a G is present to the C-terminus of the helix-turn-helix scaffold.
- a G is added to the N-terminus to the first a-helical peptide.
- a G is added to the C-terminus of the second a-helical peptide.
- a G is added to the N- terminus of the first a-helical peptide and a G is added to the C-terminus of the second a- helical peptide.
- a G is present at the N-terminus of the first a-helical peptide. In an embodiment of the present disclosure, a G is present at the C-terminus of the second a-helical peptide. In an embodiment of the present disclosure, a G is present at the N-terminus of the first a-helical peptide and a G is present at the C-terminus of the second a-helical peptide.
- said Helix-1 or said first a-helical peptide comprises an amino sequence selected from the group consisting of: GDELTQLKRELQKLYR (SEQ ID NO: 10), GDELAQLKRELQKLYR (SEQ ID NO: 11), GDELTQLKRELQKLYK (SEQ ID NO: 12), GDELTQLKRELQKLFR (SEQ ID NO: 13), GDELHQLRRELQKLYR (SEQ ID NO: 14), GDELKQLRRELQKLYH (SEQ ID NO: 15), GDELTQLRRELQKLHR (SEQ ID NO: 16).
- GDELTQLTRELKKLYR SEQ ID NO: 17
- GDELKQLIQELKKLYR SEQ ID NO: 18
- said Helix-2 or said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQG (SEQ ID NO: 32),
- DELRALIRKLHALEQG SEQ ID NO: 33
- DELAALIRKLHMLEQG SEQ ID NO: 34
- DELRALIRKLHMLAQG SEQ ID NO: 37
- DELRALIRKLHMLEAG SEQ ID NO: 38
- DELRALIRKLDMLIRG SEQ ID NO: 39
- DELEALKRKLDMLLHG SEQ ID NO: 40
- DELRALIRKLEMLKRG SEQ ID NO: 43
- DELRALIQKLDMLDRG SEQ ID NO: 44
- said Helix-1 or said first a-helical peptide comprises an amino sequence selected from the group consisting of: GDELTQLKRELQKLYR (SEQ ID NO: 10), GDELAQLKRELQKLYR (SEQ ID NO: 11), GDELTQLKRELQKLYK (SEQ ID NO: 12),
- GDELTQLKRELQKLFR (SEQ ID NO: 13), GDELHQLRRELQKLYR (SEQ ID NO: 14),
- GDELKQLRRELQKLYH (SEQ ID NO: 15), GDELTQLRRELQKLHR (SEQ ID NO: 16),
- GDELTQLTRELKKLYR SEQ ID NO: 17
- GDELKQLIQELKKLYR SEQ ID NO: 18
- said Helix-2 or said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQG (SEQ ID NO: 32), DELRALIRKLHALEQG (SEQ ID NO: 33), DELAALIRKLHMLEQG (SEQ ID NO: 34), DELRALIAKLHMLEQG (SEQ ID NO: 35), DELRALIRKLAMLEQG (SEQ ID NO: 36), DELRALIRKLHMLAQG (SEQ ID NO: 37), DELRALIRKLHMLEAG (SEQ ID NO: 38), DELRALIRKLDMLIRG (SEQ ID NO: 39), DELEALKRKLDMLLHG (SEQ ID NO: 40), DELMALIEKLIMLTHG (SEQ ID NO: 41), DELRALIRKLHMLEHG (SEQ ID NO:
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQG (SEQ ID NO: 32), DELRALIRKLHALEQG (SEQ ID NO: 33), DELAALIRKLHMLEQG (SEQ ID NO: 34), DELRALIAKLHMLEQG (SEQ ID NO: 35), DELRALIRKLAMLEQG (SEQ ID NO: 36), DELRALIRKLHMLAQG (SEQ ID NO: 37), DELRALIRKLHMLEAG (SEQ ID NO: 38), DELRALIRKLDMLIRG (SEQ ID NO: 39), and DELEALKRKLDMLLHG (SEQ ID NO: 40).
- DELRALIRKLHMLEQG SEQ ID NO: 32
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQG (SEQ ID NO: 32).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHALEQG (SEQ ID NO: 33).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELAALIRKLHMLEQG (SEQ ID NO: 34).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIAKLHMLEQG (SEQ ID NO: 35).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLAMLEQG (SEQ ID NO: 36).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLAQG (SEQ ID NO: 37).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEAG (SEQ ID NO: 38).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLDMLIRG (SEQ ID NO: 39).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELEALKRKLDMLLHG (SEQ ID NO: 40).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELAQLKRELQKLYR (SEQ ID NO: 11 and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQG (SEQ ID NO: 32).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYK (SEQ ID NO: 12) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQG (SEQ ID NO: 32).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLFR (SEQ ID NO: 13) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQG (SEQ ID NO: 32).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELHQLRRELQKLYR (SEQ ID NO: 14) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQG (SEQ ID NO: 32).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELKQLRRELQKLYH (SEQ ID NO: 15) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELMALIEKLIMLTHG (SEQ ID NO: 41).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLRRELQKLHR (SEQ ID NO: 16) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEHG (SEQ ID NO: 42) or DELRALIRKLHMLEQG (SEQ ID NO: 32).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLRRELQKLHR (SEQ ID NO: 16) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEHG (SEQ ID NO: 42).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLRRELQKLHR (SEQ ID NO: 16) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQG (SEQ ID NO: 32).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLTRELKKLYR (SEQ ID NO: 17) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLEMLKRG (SEQ ID NO: 43).
- said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELKQLIQELKKLYR (SEQ ID NO: 18) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIQKLDMLDRG (SEQ ID NO: 44).
- said Helix-1 or said first a-helical peptide comprises an amino sequence selected from the group consisting of: GDELTQLKRELQKLYR (SEQ ID NO: 10), GDELAQLKRELQKLYR (SEQ ID NO: 11), GDELTQLKRELQKLYK (SEQ ID NO: 12), GDELTQLKRELQKLFR (SEQ ID NO: 13), GDELHQLRRELQKLYR (SEQ ID NO: 14), and GDELTQLRRELQKLHR (SEQ ID NO: 16), and wherein said Helix-2 or said second a-helical peptide comprises the amino sequence of: DELRALIRKLHMLEQG (SEQ ID NO: 32).
- the first and second a-helical peptide form an antiparallel coiled-coiled structure.
- Helix-1 and Helix-2 form an antiparallel coiled-coiled structure.
- the linker Li has a length of 1 to 50 amino acids. In another embodiment, the linker Li has a length of 1-30 amino acids. Yet in another embodiment, the linker Li has a length of 1-10 amino acids. In one embodiment, the linker Li has a length of 5 amino acids. In yet a further aspect of the present disclosure the linker Li comprises 1 to 30 amino acid residues. In an embodiment, the linker Li comprises the amino acid sequence QGVDS (SEQ ID NO: 182). In one embodiment, the linker Li comprises the amino acid sequence QGVGS (SEQ ID NO: 183). In one embodiment, the linker Li consists of the amino acid sequence QGVDS (SEQ ID NO: 182). In one embodiment, the linker Li consists of the amino acid sequence or QGVGS (SEQ ID NO: 183).
- Helix-1 and Helix-2 are formed by a single polypeptide (a single-chain polypeptide) wherein the two a-helical peptides are either directly linked via a single peptide bond to each other, or are linked by a linker segment that does not substantially interfere with the association of Helix-1 and Helix-2 into a coiled-coil structure.
- a single polypeptide a single-chain polypeptide
- the linker Li comprises a non-helical region.
- the first and last residues of the nonhelical region can be any amino acid.
- one or both residues are helix breaking or helix destabilizing residues, such as G or P.
- Peptide linkers include for example, but are not limited to, G linkers, S linkers, mixed G/S linkers, G- and S-rich linkers, such as (GS) n (SEQ ID NO: 184), (G 4 S) n (SEQ ID NO: 185), (SG 4 )n (SEQ ID NO: 186), (GSGGS) n (SEQ ID NO: 187), (GGGS) n (SEQ ID NO: 188) or G 4 (SG 4 ) n (SEQ ID NO: 189), wherein n is an integer between 1 and 10, typically between 2 and 4, which are known to the skilled artisan.
- the linker Li has the amino acid sequence of GGGGSGGGGS (SEQ ID NO: 190).
- the two a-helical peptides are linked via a peptide linker Li which connects the C-terminus of Helix-1 to the N-terminus of Helix-2 thus resulting in a single-chain amino acid sequence for the HTH scaffold structure of the polypeptides according to the present disclosure.
- the HTH scaffold structure of the polypeptides according to the present disclosure comprises the general formula Helix-1 - Li - Helix-2.
- Helix-1 , Li and Helix-2 are covalently linked in a way that the C-terminus of Helix-1 is linked to the N-terminus of Li and that the C-terminus of Li is linked to the N-terminus of Helix-2.
- the arrangement from the N- to the C-terminus is as follows: Helix-1 , Li and Helix-2.
- the linker Li is absent.
- a peptide linker Li as used herein is not limited to only one of the aforementioned and exemplified peptide linkers but may comprise any combination of two or more such linker which are fused to each other.
- the linker has a length of 1 to 50 amino acid residues. In another embodiment, the linker has a length of 1-30 amino acid residues. Yet in another embodiment, the linker has a length of 1-10 amino acid residues. In one embodiment, the linker has a length of 5 amino acids. In yet a further aspect of the present disclosure the linker Li comprises 1 to 30 amino acid residues.
- the linker Li comprises the amino acid sequence QGVDS (SEQ ID NO: 182). In one embodiment, the linker Li comprises the amino acid sequence QGVGS (SEQ ID NO: 183). In one embodiment, the linker Li consists of the amino acid sequence QGVDS (SEQ ID NO: 182). In one embodiment, the linker Li consists of the amino acid sequence or QGVGS (SEQ ID NO: 183).
- the present disclosure provides an isolated polypeptide specific for IL- 13Ra2 according to the present disclosure, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of: DELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 47), DELTQLKRELQKLYRQGVGSDELRALIRKLHMLEQ (SEQ ID NO: 48), DELTQLKRELQKLYRQGVDSDELRALIRKLHALEQ (SEQ ID NO: 49), DELAQLKRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 50), DELTQLKRELQKLYRQGVDSDELAALIRKLHMLEQ (SEQ ID NO: 51), DELTQLKRELQKLYRQGVDSDELRALIAKLHMLEQ (SEQ ID NO: 52), DELTQLKRELQKLYRQGVDSDELRALIRKLAMLEQ (SEQ ID NO: 53),
- the present disclosure provides an isolated polypeptide specific for IL-13Ra2, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of:
- GDELTQLKRELQKLYRQGVGSDELRALIRKLHMLEQG (SEQ ID NO: 67), GDELTQLKRELQKLYRQGVDSDELRALIRKLHALEQG (SEQ ID NO: 68), GDELAQLKRELQKLYRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 69), GDELTQLKRELQKLYRQGVDSDELAALIRKLHMLEQG (SEQ ID NO: 70), GDELTQLKRELQKLYRQGVDSDELRALIAKLHMLEQG (SEQ ID NO: 71), GDELTQLKRELQKLYRQGVDSDELRALIRKLAMLEQG (SEQ ID NO: 72), GDELTQLKRELQKLYRQGVDSDELRALIRKLHMLAQG (SEQ ID NO: 73), GDELTQLKRELQKLYRQGVDSDELRALIRKLHMLEAG (SEQ ID
- the present disclosure provides an isolated polypeptide specific for IL-13Ra2, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of:
- GDELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 85), GDELTQLKRELQKLYRQGVGSDELRALIRKLHMLEQ (SEQ ID NO: 86), GDELTQLKRELQKLYRQGVDSDELRALIRKLHALEQ (SEQ ID NO: 87), GDELAQLKRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 88), GDELTQLKRELQKLYRQGVDSDELAALIRKLHMLEQ (SEQ ID NO: 89), GDELTQLKRELQKLYRQGVDSDELRALIAKLHMLEQ (SEQ ID NO: 90), GDELTQLKRELQKLYRQGVDSDELRALIRKLAMLEQ (SEQ ID NO: 91), GDELTQLKRELQKLYRQGVDSDELRALIRKLHMLAQ (SEQ ID NO: 92), GDE
- the present disclosure provides an isolated polypeptide specific for IL-13Ra2, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of:
- DELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 104), DELTQLKRELQKLYRQGVGSDELRALIRKLHMLEQG (SEQ ID NO: 105), DELTQLKRELQKLYRQGVDSDELRALIRKLHALEQG (SEQ ID NO: 106), DELAQLKRELQKLYRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 107), DELTQLKRELQKLYRQGVDSDELAALIRKLHMLEQG (SEQ ID NO: 108), DELTQLKRELQKLYRQGVDSDELRALIAKLHMLEQG (SEQ ID NO: 109), DELTQLKRELQKLYRQGVDSDELRALIRKLAMLEQG (SEQ ID NO: 110), DELTQLKRELQKLYRQGVDSDELRALIRKLHMLAQG (SEQ ID NO: 111), DE
- the present disclosure provides an isolated polypeptide specific for IL- 13Ra2 according to the present disclosure, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of: DELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 47), GDELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 66), GDELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 85), DELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 104), DELTQLKRELQKLYRQGVDSDELRALIRKLHALEQ (SEQ ID NO: 49), GDELTQLKRELQKLYRQGVDSDELRALIRKLHALEQG (SEQ ID NO: 68), GDELTQLKRELQKLYRQGVDSDELRALIRKLHALE
- said polypeptide is cross-reactive to mouse and cynomolgus IL-13Ra2.
- said polypeptide does not bind to IL-13 receptor alpha 1 (IL-13Ra1), IL-4 receptor alpha (IL-4Ra) or to the IL-13Ra1/ IL-4Ra complex. In an embodiment, said polypeptide competes with human IL-13 for binding to IL-13Ra2.
- the polypeptide specific for IL-13Ra2 according to the present disclosure is a recombinant polypeptide. In an embodiment, the polypeptide specific for IL-13Ra2 according to the present disclosure is a synthetic polypeptide. In an embodiment, the polypeptide specific for IL-13Ra2 according to the present disclosure is an isolated polypeptide. In a further embodiment, the polypeptide specific for IL-13Ra2 according to the present disclosure is an isolated recombinant polypeptide.
- the HTH scaffold structure of the polypeptides of present disclosure may be further stabilized by the introduction of negatively charged amino acid residues at the N-terminus of each a-helical peptide. This may stabilize the dipole moment of the a-helices of said peptides.
- a negatively charged amino acid residue can be D or E. In another embodiment, such a negatively charged amino acid residue is D.
- a polypeptide according to the present disclosure as may also be a functional variant of any of the specific polypeptides described herein.
- Such a variant polypeptide is a polypeptide that differs from a specific polypeptide described herein by one or more suitable amino acid modifications, such as substitutions, deletions, insertions, or terminal sequence additions, for instance in the invariant positions of the HTH scaffold structure, and/or the variable positions of the HTH scaffold structure in a single variant polypeptide.
- a functional variant polypeptide as used in the context of an IL-13Ra2 specific polypeptide still allows the polypeptide to retain at least a substantial proportion (at least about 50%, 60%, 70%, 80%, 90%, 95% or more) of the affinity and/or the specificity of the parent polypeptide and in some cases such an IL-13Ra2 specific polypeptide may be associated with greater affinity and/or specificity than the parent polypeptide.
- Such functional variants typically retain significant sequence identity to the parent polypeptide.
- the sequence of variable position variants may differ from the sequence of the variable position of the parent polypeptide sequences through mostly conservative amino acid substitutions; for instance at least about 35%, about 50% or more, about 60% or more, about 70% or more, about 75% or more, about 80% or more, about 85% or more, about 90% or more, about 95% or more of the substitutions in the variant are conservative amino acid residue replacements.
- conservative amino acid substitutions may be defined by substitutions within the classes of amino acids reflected in one or more of the following three tables:
- polypeptides of the present disclosure can be produced (e.g. recombinantly or organic synthesis) with additional amino acid residues added at their N- and the C-terminus in order to allow linkage of the N- and C terminal end of the polypeptides.
- the thus formed cyclized polypeptides may further increase the stability of the a-helical structure and improve resistance against proteolytic degradation by proteases.
- the polypeptides of the present disclosure can be cyclized, introducing e.g. a disulfide bridge or a reduction insensitive thioether linkage.
- a disulfide bonde can be formed under oxidizing conditions between N- and C-terminally introduced cysteine residues.
- a thioether bonde according to the present disclosure may be formed for instance between N-chloroacetyl glycine present at the N-terminus and a C residue present at the C-terminus.
- the polypeptides are cyclic polypeptides.
- the cyclic polypeptides are formed by a covalent bond.
- the covalent bond is a disulfide bond.
- the disulfide bond is formed by two C residues.
- the disulfide bond is formed between a C residue present at the N-terminus and a C residue present at the C-terminus of the polypeptides of the present disclosure.
- the covalent bond is a thioether bond. In certain embodiments of the present disclosure, the covalent bond is a thioether bond formed between N-chloroacetyl glycine and a C residue. In certain embodiments, the covalent bond is a thioether bond formed between N-chloroacetyl glycine present at the N-terminus and a C residues present at the C-terminus of the polypeptides of the present disclosure. In certain embodiments, the covalent bond is a thioether bond formed between N-chloroacetyl glycine present at the N-terminus and a C residues present at the C-terminus of the polypeptides of the present disclosure.
- polypeptides of the present disclosure can be synthesized by a variety of means, for example, by recombinant DNA technology or by organic chemical synthesis. Methods of peptide synthesis are known in the art.
- the coding sequences for the polypeptides can be recombinant DNA molecules, which are introduced into expression vectors or phage by operatively linking the DNA to the necessary expression control regions (e.g. regulatory regions) required for gene expression.
- the vectors can be introduced into the appropriate host cells such as prokaryotic (e.g., bacterial) or eukaryotic (e.g., yeast or mammalian) cells by methods well known in the art (see, e.g., "Current Protocol in Molecular Biology", Ausubel et al. (eds.), Greene Publishing Assoc, and John Wiley Interscience, New York, 1989 and 1992). Numerous cloning vectors are known to those of skill in the art, and the selection of an appropriate cloning vector is a matter of choice.
- prokaryotic e.g., bacterial
- eukaryotic e.g., yeast or mammalian cells
- Numerous cloning vectors are known to those of skill in the art, and the selection of an appropriate cloning vector is a matter of choice.
- the gene can be placed under the control of a promoter, ribosome binding site (for bacterial expression) and, optionally, an operator (collectively referred to herein as "control" elements), so that the DNA sequence encoding the desired protein is transcribed into RNA in the host cell transformed by a vector containing this expression construction.
- the coding sequence may or may not contain a signal peptide or leader sequence.
- the proteins of the present disclosure are produced by growing host cells transformed by an expression vector described above under conditions whereby the protein of interest is expressed. The polypeptide is then isolated from the host cells and purified. If the expression system secretes the protein into growth media, the protein can be purified directly from the media.
- polypeptides of the present disclosure can then be purified by a number of techniques as known to the person skilled in the art. It should be noted that the polypeptides of the present disclosure are not naturally occurring proteins. Typically, the polypeptides of the present disclosure are recombinant, synthetic or semi-synthetic amino acid sequences, polypeptides or proteins. Nucleic acids
- the present disclosure refers to a nucleic acid composition comprising a nucleic acid sequence or a plurality of nucleic acid sequences encoding an isolated polypeptide specific for IL-13Ra2 according to the present disclosure or a fusion molecule comprising said isolated polypeptide and at least one additional moiety.
- the present disclosure refers to a nucleic acid composition
- a nucleic acid composition comprising a nucleic acid sequence or a plurality of nucleic acid sequences encoding an isolated polypeptide specific for IL-13-Ra2 according to the present disclosure or a fusion molecule comprising said isolated polypeptide and at least one additional moiety, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 47 - 122.
- the present disclosure refers to a nucleic acid composition
- a nucleic acid composition comprising a nucleic acid sequence or a plurality of nucleic acid sequences encoding Helix-1 of an isolated polypeptide specific for IL-13-Ra2 or a fusion molecule comprising said Helix-1 and at least one additional moiety, wherein said Helix-1 comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 1 - 18.
- the present disclosure refers to a nucleic acid composition
- a nucleic acid composition comprising a nucleic acid sequence or a plurality of nucleic acid sequences encoding Helix-2 of an isolated polypeptide specific for IL-13Ra2 or a fusion molecule comprising said Helix-2 and at least one additional moiety, wherein said Helix-2 comprises an amino acid sequence selected from the group consisting of: SEQ ID NO 19 - 44.
- the present disclosure refers to a nucleic acid composition
- a nucleic acid composition comprising a nucleic acid sequence or a plurality of nucleic acid sequences encoding Helix-1 and Helix-2 of an isolated polypeptide specific for IL- 13Ra2 or a fusion molecule comprising said Helix-1 and Helix-2 and at least one additional moiety, wherein said Helix-1 comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 1 - 18, and wherein said Helix-2 comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 19 - 44.
- the present disclosure refers to a vector composition comprising a vector or plurality of vectors comprising a nucleic acid composition according to the present disclosure encoding an isolated polypeptide specific for IL-13Ra2 according to the present disclosure or a fusion molecule comprising said isolated polypeptide and at least one additional moiety.
- the present disclosure refers to a vector composition comprising a vector or plurality of vectors comprising a nucleic acid composition according to the present disclosure encoding an isolated polypeptide specific for IL-13Ra2 according to the present disclosure or a fusion molecule comprising said isolated polypeptide and at least one additional moiety.
- said polypeptide comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 47 - 122.
- the present disclosure refers to a vector composition
- a vector composition comprising a vector or plurality of vectors comprising a nucleic acid composition according to the present disclosure encoding Helix-1 of an isolated polypeptide specific for IL-13-Ra2 according to the present disclosure or a fusion molecule comprising said Helix-2 of an isolated polypeptide and at least one additional moiety, wherein said Helix-1 comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 1 - 18:
- the present disclosure refers to a vector composition comprising a vector or plurality of vectors comprising a nucleic acid composition according to the present disclosure encoding Helix-2 of an isolated polypeptide specific for IL-13-Ra2 according to the present disclosure or a fusion molecule comprising said Helix-2 of an isolated polypeptide and at least one additional moiety, wherein said Helix-2 comprises an amino acid sequence selected from the group consisting of: SEQ ID NO 19 - 44.
- the present disclosure refers to a vector composition
- a vector composition comprising a vector or plurality of vectors comprising a nucleic acid composition according to the present disclosure encoding Helix-1 and Helix-2 of an isolated polypeptide specific for IL-13-Ra2 according to the present disclosure or a fusion molecule comprising said Helix-1 and Helix-2 of an isolated polypeptide and at least one additional moiety, wherein said Helix-1 comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 1 - 18 and wherein said Helix-2 comprises an amino acid sequence selected from the group consisting of SEQ ID NO 19 - 44.
- the present disclosure refers to a host cell comprising a vector composition according to the present disclosure encoding an isolated polypeptide specific for IL-13Ra2 according to the present disclosure or a fusion molecule comprising said isolated polypeptide and at least one additional moiety.
- said polypeptide comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 47 - 122.
- the present disclosure refers to host cell comprising a vector composition according to the present disclosure encoding Helix-1 of an isolated polypeptide specific for IL- 13-Ra2 according to the present disclosure or a fusion molecule comprising said Helix-1 of an isolated polypeptide and at least one additional moiety, wherein said Helix-1 comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 1 - 18.
- the present disclosure refers to host cell comprising a vector composition according to the present disclosure encoding Helix-2 of an isolated polypeptide specific for IL- 13-Ra2 according to the present disclosure or a fusion molecule comprising said Helix-2 of an isolated polypeptide and at least one additional moiety, wherein said Helix-2 comprises an amino acid sequence selected from the group consisting of: SEQ ID NO 19 - 44.
- the present disclosure refers to a host cell comprising a vector composition according to the present disclosure encoding Helix-1 and Helix-2 of an isolated polypeptide specific for IL-13-Ra2 according to the present disclosure or a fusion molecule comprising said Helix- 1 and Helix-2 of an isolated polypeptide and at least one additional moiety, wherein said Helix-1 comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 1 - 18 and wherein said Helix-2 comprises an amino acid sequence selected from the group consisting of: SEQ ID NO 19 - 44.
- said host cell is able to express the polypeptide according to the present disclosure encoded by the vector composition.
- said host cell is an isolated cell.
- said isolated host cell is a mammalian cell.
- said mammalian cell is a human cell.
- said mammalian cell is a CHO cell.
- the present disclosure pertains to an isolated polypeptide specific for IL- 13Ra2 disclosed in SEQ ID NO: 47 - 122.
- the isolated polypeptide according to the present disclosure is specific for IL-13Ra2. In an embodiment, the isolated polypeptide according to the present disclosure is specific for human IL-13Ra2 encoded by the amino acid sequence of SEQ ID NO: 123. In an embodiment, the isolated polypeptide according to the present disclosure is specific for a polypeptide comprising the amino acid sequence of SEQ ID NO: 123. In an embodiment, the polypeptide according to the present disclosure specifically binds to the extracellular region human IL-13Ra2.
- the isolated polypeptide according to the present disclosure does not bind to IL-13Ra1. In an embodiment, the isolated polypeptide according to the present disclosure does not bind to human and/or mouse IL-13Ra1. In certain embodiment, the isolated polypeptide according to the present disclosure is not cross-reactive to IL-13Ra1. In an embodiment, the isolated polypeptide according to the present disclosure does not bind to human and/or mouse IL-13Ra1.
- the isolated polypeptide according to the present disclosure does not bind to IL-4Ra. In certain embodiment, the isolated polypeptide according to the present disclosure is not cross- reactive to IL-4Ra. In an embodiment, the isolated polypeptide according to the present disclosure does not bind to human and/or mouse IL-4Ra. In certain embodiment, the isolated polypeptide according to the present disclosure does not bind to IL- 13Ra1/IL-4Ra complex. In an embodiment, the isolated polypeptide according to the present disclosure does not bind to the human and/or mouse IL-13Ra1/IL-4Ra complex.
- said IL-13Ra1 , IL-4Ra or IL-13Ra1/IL-4Ra complex is expressed on cells.
- the present disclosure refers to an isolated polypeptide specific for IL- 13Ra2 according to the present disclosure, wherein said polypeptide binds to human IL-13Ra2 with an ECso concentration of 60 nM or less, 50 nM or less, 40 nM or less, 30 nM or less, 20 nM or less, 15 nM or less, 10 nM or less, 5 nM or less, 4 nM or less, 3 nM or less, 2 nM or less, 1 nM or less, 0.5 nM or less, 0.4 nM or less, 0.3 nM or less, 0.2 nM or less or 0.1 nM or less.
- the present disclosure pertains to an isolated polypeptide specific for IL- 13Ra2, wherein said polypeptide binds to cynomolgus IL-13Ra2 with an ECso concentration of 60 nM or less, 50 nM or less, 40 nM or less, 30 nM or less, 20 nM or less, 15 nM or less, 10 nM or less, 5 nM or less, 4 nM or less, 3 nM or less, 2 nM or less, 1 nM or less, 0.5 nM or less, 0.4 nM or less, 0.3 nM or less, 0.2 nM or less or 0.1 nM or less.
- the present disclosure refers to an isolated polypeptide specific for IL-13Ra2, wherein said polypeptide binds to mouse IL-13Ra2 with an ECso concentration of 60 nM or less, 50 nM or less, 40 nM or less, 30 nM or less, 20 nM or less, 15 nM or less, 10 nM or less, 5 nM or less, 4 nM or less, 3 nM or less, 2 nM or less, 1 nM or less, 0.5 nM or less, 0.4 nM or less, 0.3 nM or less, 0.2 nM or less or 0.1 nM or less.
- the present disclosure refers to an isolated polypeptide specific for IL- 13Ra2, wherein said polypeptide binds to human, cynomolgus and mouse IL-13Ra2 with an ECso concentration of 10 nM or less.
- said human IL-13Ra2 comprises the amino acid sequence of SEQ ID NO: 123.
- said mouse IL-13Ra2 comprises the amino acid sequence of SEQ ID NO:125.
- said cynomolgus IL-13Ra2 comprises the amino acid sequence of SEQ ID NO: 124.
- said human, cynomolgus or mouse IL-13Ra2 is expressed on cells.
- said human, cynomolgus or mouse IL-13Ra2 is expressed on engineered CHO cells expressing full-length human, cynomolgus, or mouse IL-13Ra2.
- said CHO cells are Flp-ln CHO cells.
- said human IL-13Ra2 is expressed on cancer cells.
- said human IL-13Ra2 is expressed on A-375 cells.
- said EC50 concentration is determined as described herein in Example 3. In certain embodiments, said ECso concentration is determined by FACS described herein in Example 3. In further embodiments, the polypeptide specific for IL-13Ra2 according to the present invention does substantially not bind to IL-13Ra1 , IL-4Ra or to the IL-13Ra1/IL-4Ra complex. In an embodiment, said polypeptide according to the present disclosure does substantially not bind to IL-13Ra1 , IL-4Ra or to the IL-13Ra1/IL-4Ra complex at a polypeptide concentration of 1 pM or less. In an embodiment, said binding is determined as described in Example 3. In an embodiment, said binding is determined in ELISA.
- said binding is determined by FACS.
- said IL-13Ra1, IL-4Ra or IL-13Ra1/IL-4Ra complex is expressed on cells.
- said IL-13Ra1 , IL-4Ra or IL-13Ra1/IL-4Ra complex is expressed on cancer cells.
- said IL-13Ra1 , IL-4Ra or IL- 13Ra1/IL-4Ra complex is expressed on A- 549 cells.
- the isolated polypeptide according to the present disclosure is cross- reactive to cynomolgus monkey (cynomolgus) IL-13Ra2. In certain embodiment, the isolated polypeptide according to the present disclosure is cross-reactive to mouse IL-13Ra2. In certain embodiments, the isolated polypeptide according to the present disclosure is cross- reactive to cynomolgus and mouse IL-13Ra2. In certain embodiments, the isolated polypeptide according to the present disclosure is specific for human, cynomolgus and mouse IL-13Ra2.
- the present disclosure refers to an isolated polypeptide specific for human IL- 13Ra2, wherein said polypeptide cross-reactively binds to cynomolgus and mouse IL-13Ra2.
- the isolated polypeptide according to the present disclosure specifically binds to the extracellular region of human, cynomolgus and mouse IL-13Ra2.
- IL-13Ra2 is internalized from the cell surface of cells, such as cancer cells, expressing IL-13Ra2, upon binding of the polypeptide according to the present invention to said cells.
- the polypeptide according to the present invention is internalized from the surface of a cell, such as a cancer cell, expressing IL-13Ra2, upon binding to the cells.
- the polypeptide of the present disclosure binds to the binding region of IL-13 on IL-13Ra2.
- the polypeptide of the present disclosure may compete with IL-13 for binding to IL-13Ra2.
- the disclosed polypeptides may interfere with IL-13 mediated signaling through IL-13Ra2.
- the polypeptide of the present disclosure does not compete with IL- 13 for binding to IL-13Ra1. In certain embodiments, the polypeptide of the present disclosure does not compete with IL-13 for binding to IL-4Ra. In certain embodiments, the polypeptide of the present disclosure does not compete with I L-13 for binding to the IL13Ra1/IL-4Ra complex. In certain embodiments, the polypeptide according to the present disclosure does not interfere with IL-13 mediated signaling through IL-13Ra1 or the IL13Ra1/IL-4Ra complex. In an embodiment, said IL-13 is human IL-13.
- HTH polypeptides provided by the present disclosure may be fused, coupled or conjugated to one or more other moieties to generate fusion molecules in accordance with any of a variety of techniques, such as those employed in the production of immunoconjugates, which are known to the person skilled in the art.
- fusion molecules are generally achieved by covalent bonds between the HTH polypeptides of the present disclosure and the one or more moieties.
- the fusion molecules are targeted only to cells expressing a target antigen for a HTH polypeptide according to the present invention. These targets may internalize in response to polypeptide binding. In certain embodiments, said target is IL-13Ra2. Accordingly, the present disclosure provides fusion molecules comprising an isolated polypeptide according to the present disclosure and at least one additional moiety.
- said additional moiety is a selected from the group consisting of a therapeutic agent, a cytotoxic agent, a bacterial toxin (such as Pseudomonas exotoxin A and Diphtheria toxin, or plant toxins, such as ricin), a cytokine, an antibody or antibody fragment, a peptide, a polypeptide, a reporter enzyme, a detectable group, a moiety being capable of binding a metal ion, a tag suitable for detection and/or purification, a targeting ligands, a homo- or hetero-association domain, a moiety which increases solubility of a protein, or a moiety which comprises an enzymatic cleavage site, a lipid, a liposome and virus-like-particles
- a “therapeutic agent” as used herein may be any therapeutic agent including, but not limited to, genetic materials or agents, radionuclides, chemotherapeutic agents, and cytotoxic agents (See, e.g., U.S. Patent No. 6,949,245 to Sliwkowski), and antimicrobial peptides.
- Radionuclide as described herein includes, but is not limited to 88 Ga 225 Ac, 210 At, Ba, 77 Br, 109 Cd, 51 Cr, 67 Cu, 165 Dy, 155 Eu, 153 Gd, 198 Au, 166 Ho, 113ra ln, 115111 ln, 123 l, 125 l, 131 l, 189 lr, 191 lr, 192 lr, 194 lr, 52 Fe, 55 Fe, 59 Fe, 177 Lu, 109 Pd, 32 P, 226 Ra, 186 Re, 188 Re, 153 Sm, 46 Sc, 47 Sc, 72 Se, 75 Se, 105 Ag, 89 Sr, 35 S, 177 Ta, 117 mSn, 121 Sn, 166 Yb, 169 Yb, 90 Y, 212 Bi, 213 Bi 119 Sb, 197 Hg, 97 Ru, 100 Pd, 101 mRh, and 212 Pb.
- “Chemotherapeutic agent” as used herein includes, but is not limited to, methotrexate, daunomycin, mitomycin C, cisplatin, vincristine, epirubicin, fluorouracil, verapamil, cyclophosphamide, cytosine arabinoside, aminopterin, bleomycin, mitomycin C, democolcine, etoposide, mithramycin, chlorambucil, melphalan, daunorubicin, doxorubicin, tamosifen, paclitaxel, vincristin, vinblastine, camptothecin, actinomycin D, and cytarabine.
- Cytotoxic agent includes, but is not limited to, maytansinoids and maytansinoid analogs (such as DM1 , DM4), taxoids, CC- 1065 and CC- 1065 analogs, dolastatin and dolastatin analogs, ricin (or more particularly the ricin A chain), aclacinomycin, Diphtheria toxin, Monensin, Verrucarin A, Abrin, Tricothecenes, and Pseudomonas exotoxin A, taxol, cytochalasin B, gramicidin D, ethidium bromide, emetine, mitomycin, etoposide, tenoposide, anti-mitotic agents, such as the vinca alkaloids (e.g., vincristine and vinblastine), colchicin, anthracyclines, such as doxorubicin and daunorubicin, dihydroxy anthracin dione, mitox
- ricin or
- Detectable group includes, but is not limited to, radiolabels, enzyme labels (e.g., horseradish peroxidase, alkaline phosphatase), gold beads, chemiluminescence labels, ligands (e.g., biotin, digoxin) and/or fluorescence labels (e.g., rhodamine, phycoerythrin, fluorescein, fluorescent proteins), a fluorescent protein including, but not limited to, a green fluorescent protein or one of its many modified forms, a nucleic acid segment in accordance with known techniques, and energy absorbing and energy emitting agents.
- enzyme labels e.g., horseradish peroxidase, alkaline phosphatase
- gold beads e.g., gold beads
- chemiluminescence labels e.g., ligands (e.g., biotin, digoxin) and/or fluorescence labels (e.g., rhodamine, phycoerythr
- the polypeptide according to the present disclosure is fused to one or more Monomethyl auristatin E (MMAE) and/or Monomethyl auristatin F (MMAF) molecules (e.g., about 1 to about 10 MMAE or MMAF molecules per targeting polypeptide).
- MMAE Monomethyl auristatin E
- MMAF Monomethyl auristatin F
- the HTH polypeptide conjugated to one or more maytansine molecules such as DM-1 or DM-4) (e.g., about 1 to about 10 maytansine molecules per targeting polypeptide).
- Enzymatically active toxins and fragments thereof which can be used are described above and include diphtheria A chain, nonbinding active fragments of diphtheria toxin, exotoxin A chain (from Pseiidomonas aeruginosa), ricin A chain, abrin A chain (from Corrybacterhim typhimuriae), modeccin A chain, alpha-sarcin, Aleurites fordii proteins, dianthin proteins, Phytolaca americana proteins (PAPI, PAPII, and PAP-S), momordica charantia inhibitor, curcin, crotin, sapaonaria officinalis inhibitor, gelonin, mitogellin, restrictocin, phenomycin, enomycin and the tricothecenes (see for example WO 93/21232)
- radioactive isotopes or radionuclides are available for the production of radioconjugated fusion molecules as described above.
- the polypeptide specific for IL13-Ra2 according to the present disclosure is fused to one or more radionuclides.
- the fusion molecules according to the present disclosure may be made using a variety of bi-functional protein coupling agents such as N-succinimidyl-3-(2- pyridyldithiol)propionate (SPDP), succinimidyl-4-(N- maleimidomethyl) cyclohexane-l- carboxylate, iminothiolane (IT), bifunctional derivatives of imidoesters (such as dimethyl adipimidate HCL), active esters (such as disuccinimidyl suberate), aldehydes (such as glutareldehyde), bis-azido compounds (such as bis(p- azidobenzoyl)hexanediamine), bis- diazonium derivatives (such as bis-(p-diazoniumbenzoyl)- ethylenediamine), diisocyanates (such as tolyene 2,6-diisocyanate), and bis-active fluorine compounds (such as l,
- SPDP
- Carbon- 14-labeled 1- isothiocyanatobenzyl-3-methyldiethylene triaminepentaacetic acid is an exemplary chelating agent for conjugation of radionucleotide to the targeting peptide (see WO 94/11026).
- 1,4,7,10-Tetraazacyclododecane- 1 ,4,7,10-tetraacetic acid is a further exemplary chelating agent for conjugation of radionucleotide to the polypeptides according to the present disclosure
- DOTA also known as tetraxetan
- the present disclosure provides a fusion molecules comprising an isolated polypeptide specific for IL-13Ra2 according to the present disclosure and at least one or more radionuclides.
- said radionuclide is contained within 1 ,4,7,10- tetraaza-
- TCMC 1.4.7.10-tetra(2-carbamoylmethyl)cyclododecane
- said radionuclide is 177 Lu. In an embodiment, said radionuclide is 255 Ac. In an embodiment, said radionuclide is 213 Bi.
- said polypeptide specific for IL-13Ra2 comprises an amino acid sequence selected from the group of SEQ ID NOs: 47 - 122.
- said polypeptide specific for IL-13Ra2 comprises an amino acid sequence selected from the group of: DELTQLKRELQKLYRQGVDSDELRALIRKLHALEQ (SEQ ID NO: 49), GDELTQLKRELQKLYRQGVDSDELRALIRKLHALEQG (SEQ ID NO: 68) GDELTQLKRELQKLYRQGVDSDELRALIRKLHALEQ (SEQ ID NO: 87), DELTQLKRELQKLYRQGVDSDELRALIRKLHALEQG (SEQ ID NO: 106), and GDELTQLKRELQKLYRQGVDSDELRALIRKLHALEQGGGGGSGGGGGGSGDELTQLKRELQK LYRQGVDSDELRALIRKLHALEQG (SEQ ID NO: 168)
- a fusion molecule according to the present disclosure may be made such that its individual components (e.g. toxin or antibody) are fused directly to each other or indirectly through a tether.
- the components of a fusion molecule according to the present disclosure are genetically fused to each other.
- Such fusion can be achieved by a number of strategies, which include, but are not limited to polypeptide fusion between the N- and C- terminus of two polypeptides, fusion via disulfide bonds, and fusion via chemical cross-linking reagents.
- tethers include for example, but are not limited to peptide or polypeptide tethers such as glycine tethers, serine tethers, mixed glycine/serine tethers, glycine- and serine-rich tethers, tethers composed of largely polar polypeptide fragments or tethers comprising an amino acid sequence forming a random coil conformation.
- peptide or polypeptide tethers such as glycine tethers, serine tethers, mixed glycine/serine tethers, glycine- and serine-rich tethers, tethers composed of largely polar polypeptide fragments or tethers comprising an amino acid sequence forming a random coil conformation.
- a peptide or polypeptide tether may be any suitable amino acid sequence having a length between 1 and 500 amino acid residues, such as between 1 and 100, between 1 and 50, between 1 and 10, or between 1 and 5 amino acid residues.
- the composition and length of a tether may be determined in accordance with methods well known in the art and may be tested for efficacy.
- the tether is non-immunogenic.
- the tether is a peptide tether.
- the tether is a peptide tether comprising one or more amino acid residues, joined by peptide bonds that are known in the art.
- the peptide tether should have a length that is adequate to fuse two polypeptides (or components) in such a way that they assume the correct conformation relative to one another so that they retain or obtain the desired activity.
- a peptide or polypeptide tether according to the present disclosure is from 1 to 70 amino residues in length, 1 to 50 amino acid residues in length, 1 to 40 amino residues in length, 1 to 30 amino acid residues in length, 1 to 20 amino acid residues in length, 1 to 10 amino acid residue in length, 1 to 5 amino acid residues in length.
- a peptide or polypeptide tether according to the present disclosure has a length of at least 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 25, 30, 35, 40, 45, 50, 60, 70 amino acids residues.
- a peptide or polypeptide tether according to the present disclosure has a length of 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 25, 30, 35, 40, 45, 50, 60, or 70 amino acids residues.
- the peptide or polypeptide tether may pre-dominantly comprise the following amino acid residues: Gly, Ser, Ala, or Thr.
- Suitable, non-immunogenic peptide tethers comprises glycineserine polymers for example, (GS) n (SEQ ID NO: 184), (G4S) n (SEQ ID NO: 185), (SG4)n (SEQ ID NO: 186), (GSGGS) n (SEQ ID NO: 187), (GGGS) n (SEQ ID NO: 188) or G 4 (SG 4 )n (SEQ ID NO: 189), wherein n is an integer between 1 and 10, typically between 2 and 4.
- a non- immunogenic peptide tether used herein may comprise glycine-alanine polymers, alanineserine polymers, and other flexible peptide tethers.
- a suitable peptide tether for fusing the polypeptide according to the present disclosure to the second polypeptide according to the present disclosure and/or to a moiety, such as an antibody or antibody fragment is a glycineserine polymer, such as (648)2 (SEQ ID NO: 190).
- Peptide tethers can be also derived from immunoglobulin light or heavy chain constant domain, such as CLK or CL domains or the CH1 domain, but not all residues of such a constant domain, for example only the first 5 - 12 amino acid residues.
- the peptide tether is not a immunoglobulin light or heavy chain constant domain.
- the peptide linker is not a CLK, CL , CH1 , CH2 or CH3 domain.
- Exemplary peptide tethers which may be used in an antigen-binding molecule are derived from immunoglobulin light or heavy chain constant domain are QPKAAP (SEQ ID NO: 191) or ASTKGP (SEQ ID NO: 192).
- peptide tethers can be derived from immunoglobulin heavy chains of any isotype, including for example Cy1 , Cy2, Cy3, Cy4, Ca1 , Ca2, C8, Cs, and Op.
- a peptide tether may also comprise an immunoglobulin hinge (e.g. a human I gG 1 hinge or part thereof) or any peptide derived from such hinge.
- the truncated hinge may not include one or more of its interchain cysteines. The presence of the interchain cysteines would allow for the formation of a dimeric peptide linker (or hinge region) by disulfide bridges.
- a peptide or polypeptide tethers as used herein is not limited to only one of the aforementioned and exemplified peptide tethers but my comprise any combination of two or more such tethers which are fused to each other.
- a peptide tether as used herein may be built from a glycine-serine polymer and an immunoglobulin hinge derived sequence in order to retain or obtain the desired activity.
- non-proteinaceous polymers including but not limited to polyethylene glycol (PEG), polypropylene glycol, polyoxyalkylenes, or copolymers of polyethylene glycol and polypropylene glycol, may be used as tethers.
- PEG polyethylene glycol
- polypropylene glycol polypropylene glycol
- polyoxyalkylenes polyoxyalkylenes
- copolymers of polyethylene glycol and polypropylene glycol may be used as tethers.
- the tether may be a cleavable tether facilitating release of the cytotoxic drug in the cell.
- a cleavable tether facilitating release of the cytotoxic drug in the cell.
- an acid-labile thether, peptidase-sensitive thether, dimethyl tether or disulfide- containing tether (Chari et al. (1992) Cancer Res. 52:127-131) may be used.
- the total number of amino acid residues in a polypeptide of the present disclosure can be in the range of 25 to 50000, in the range of 25-10000, in the range of 25 to 5000, in the range of 25-1000, in the range of 25-500, in the range of 25-250, in the range of 25-100, in the range of 25-50, or in the range of 25-35, depending mainly on the length of the flexible linkers interconnecting the two a-helical peptides and the additional moieties which may be linked to the helix-turn-helix scaffold structure.
- the polypeptides of the present disclosure are fused to a poly-histidine tag. In other embodiment, the polypeptides of the present disclosure are fused to a FLAG tag. Yet in another embodiment, the polypeptides are fused to a FLAG and poly-histidine tag. In certain embodiments, said poly-histidine and/or FLAG tag is fused to the carboxyl- and/or aminoterminus of the polypeptide of the present disclosure. In another embodiment, the polypeptides of the present disclosure are fused to a maltose binding protein (MBP). Maltose binding protein may increase the solubility during expression in bacterial hosts.
- MBP maltose binding protein
- the maltose binding protein domain is fused to the amino-terminus of the polypeptide.
- the bacterial host used for expression is Escherichia coli.
- an enzymatic cleavage side is present between the C-terminus of the maltose- binding domain and the N-terminus of the polypeptides of the present disclosure.
- said enzymatic cleavage side is a FXa cleavage side.
- the present disclosure provides a fusion molecule comprising an isolated polypeptide specific for IL-13Ra2 according to the present disclosure and an antibody or antibody fragment thereof.
- the polypeptides specific for IL-13Ra2 according to the present are fused to an antibody or an antibody fragment thereof.
- said antibody fragment is a Fab fragment.
- the antibody or antibody fragment thereof may have the same binding specificity or a different binding specificity as the polypeptides of the present disclosure.
- polypeptides specific for IL-13Ra2 according to the present disclosure may be fused to the antibody or an antibody fragment thereof, either by a direct linkage to the N- and/or C- terminus of the HTH polypeptides or by a peptide tether comprising one or more amino acid residues at the N- and/or C-terminus of the polypeptides.
- the polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the C-terminus of the heavy chain of an antibody.
- the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the N-terminus of the heavy chain of an antibody.
- the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the C-terminus of the light chain of an antibody. In an embodiment, the HTH polypeptide according to the present disclosure is fused to the N- terminus of the light chain of an antibody. In an embodiment, the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the C-terminus of a Fab heavy chain. In an embodiment, the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the C-terminus of the CH1 domain of a Fab.
- the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the N-terminus of a Fab heavy chain. In an embodiment, the HTH polypeptide specific for IL- 13Ra2 according to the present disclosure is fused to the N-terminus of the VH domain of a Fab. In an embodiment, the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the C-terminus of a Fab light chain. In an embodiment, the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the C-terminus of the CL domain of a Fab.
- the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the N-terminus of a Fab light chain. In an embodiment, the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the N-terminus of the VL domain of a Fab.
- the HTH polypeptide specific for IL-13Ra2 can be fused to an antibody or antibody fragment via a peptide tether comprising glycine-serine polymers, such as (GS) n (SEQ ID NO: 184), (G4S) n (SEQ ID NO: 185), (SG4)n (SEQ ID NO: 186), (GSGGS) n (SEQ ID NO: 187), (GGGS) n (SEQ ID NO: 188) or G 4 (SG 4 )n (SEQ ID NO: 189), wherein n is an integer between 1 and 10.
- a suitable peptide tether for fusing the polypeptide according to the present disclosure to an antibody or antibody fragment is a glycine-serine polymer, such as (648)2 (SEQ ID NO: 190).
- the present disclosure also provides a novel approach of simultaneously engaging CD3 and the cancer specific antigen IL-13Ra2 via a fusion molecule having the following properties: (a) binding specificity for CD3; and (b) binding specificity for IL-13Ra2.
- This fusion molecule according to the present disclosure may be designed to provide a tumor-target-dependent activation of CD3 on T-cells, via IL-13Ra2 expressed on tumor cells.
- CD3 is a proven T-cell stimulating antigen with therapeutic relevance.
- CD3 refers to an antigen which is expressed on T cells as part of the multi-molecular T cell receptor (TCR) and which consists of a homodimer or heterodimer formed from the association of two of four receptor chains: CD3-epsilon, CD3-delta, CD3-zeta, and CD3-gamma Human CD3 epsilon has the amino acid sequence as defined in UniProt: P07766 (SEQ ID NO: 193).
- the bispecific fusion molecule according to the present disclosure is capable of crosslinking a T-cell and an IL-13Ra2 expressing cell by simultaneous binding to an IL-13Ra2 expressing cell and a CD3 expressing T-cell.
- simultaneous binding results in lysis of the IL-13Ra2 expressing cell, particularly lysis of a tumor cell expressing IL-13Ra2.
- simultaneous binding results in activation of the T-cell.
- the simultaneous binding results in a cellular response of a T-lymphocyte, particularly a cytotoxic T-lymphocyte, selected from the group of: proliferation, differentiation, cytokine secretion, cytotoxic effector molecule release, cytotoxic activity, and expression of activation markers.
- a cytotoxic T-lymphocyte selected from the group of: proliferation, differentiation, cytokine secretion, cytotoxic effector molecule release, cytotoxic activity, and expression of activation markers.
- binding of the bispecific fusion molecule according to the present disclosure to CD3 without simultaneous binding to the target cell antigen does not result in T-cell activation.
- the bispecific fusion molecule according to the present disclosure is capable of re-directing cytotoxic activity of a T-cell to a IL-13Ra2 expressing cancer cell.
- a T-cell according to any of the embodiments according to the present disclosure is a cytotoxic T-cell.
- the T-cell is a CD4+ or a CD8+ T cell.
- the present disclosure provides a bispecific fusion molecule comprising at least one HTH polypeptide specific for IL-13Ra2 according to the present disclosure and at least one antibody or antibody fragment specific for CD3.
- the bispecific fusion molecule may comprise a second HTH polypeptide specific for IL-13Ra2 according to the present disclosure.
- the HTH polypeptides specific for IL-13Ra2 according to the present disclosure can be fused to the antibody Fab fragment as essentially described in Figure 6.
- a HTH polypeptides specific for IL-13Ra2 according to the present disclosure can be fused via a peptide tether to the C-terminus of the Fab heavy chain, the N-terminus of the Fab heavy chain, the C-terminus of the Fab light chain and/or the N-terminus of the Fab light chain.
- the HTH polypeptide is fused to the C-terminus of the Fab heavy chain ( Figure 6A).
- the HTH polypeptides specific for IL-13Ra2 according to the present disclosure is fused at its N-terminus or its C-terminus to the Fab.
- the HTH polypeptide is fused at its C-terminus to Fab heavy chain.
- the HTH polypeptides according to the present disclosure may be linked via a peptide tether between the C-terminus of the Fab heavy chain constant region (CH1) or the C-terminus of a Fab light chain constant region (CL) of the Fab.
- the peptide tether may be an unstructured (648)2 (SEQ ID NO: 190) tether.
- the Fab and/or the HTH polypeptide may be fused at its N-terminus and/or its C-terminus to another moiety.
- a first HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the heavy chain of a Fab
- a second HTH polypeptide may be fused to the light chain of the Fab.
- a second HTH polypeptide may be fused at its N-terminus and/or its C-terminus to the first HTH polypeptide fused to the Fab ( Figure 6B).
- said fusion may occur via an unstructured (648)2 (SEQ ID NO: 190) tether.
- the present disclosure provides a bispecific fusion molecule comprising a polypeptide specific for IL-13Ra2 according to the present disclosure an antibody or antibody fragment specific for CD3.
- said IL-13Ra2 specific polypeptide comprised in the bispecific fusion molecule according to the present disclosure is fused to the C-terminus of the Fab heavy chain.
- the Fab specific for CD3 comprises the variable heavy chain sequence of: EVQLVESGGGLVKPGGSLRLSCAASGFTFNTYAMNWVRQAPGKGLEWVGRIRSKYNNYAT YYADSVKDRFTISRDDSKNTLYLQMNSLKTEDTAVYYCTTHGNFGNSYVSWFAYWGQGTL VTVSS (SEQ ID NO: 180) and the variable light chain sequence of:
- the Fab specific for CD3 comprises the heavy chain sequence of: EVQLVESGGGLVKPGGSLRLSCAASGFTFNTYAMNWVRQAPGKGLEWVGRIRSKYNNYAT YYADSVKDRFTISRDDSKNTLYLQMNSLKTEDTAVYYCTTHGNFGNSYVSWFAYWGQGTL VTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAV LQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKRVEPKSC (SEQ ID NO: 178) and the light chain sequence of:
- the IL-13Ra2 specific polypeptide comprised in the bispecific fusion molecule according to the present disclosure comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 47 - 122.
- the bispecific fusion molecule according to the present disclosure is composed of at least 2 polypeptides, wherein a. a first polypeptide comprises the light chain of the Fab specific for CD3, b. a second polypeptide comprises from its N-terminus to its C-terminus i. a heavy chain of the Fab specific for CD3 ii. a peptide tether and iii. a polypeptide specific for IL-13Ra2 according to the present disclosure.
- the first polypeptide chain comprises the amino acid sequence of SEQ ID NO: 155.
- the second polypeptide chain comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 150, 151 , 156 - 167, 171 , and 173.
- the second polypeptide chain comprises the amino acid sequence of EVQLVESGGGLVKPGGSLRLSCAASGFTFNTYAMNWVRQAPGKGLEWVGRIRSKYNNYAT YYADSVKDRFTISRDDSKNTLYLQMNSLKTEDTAVYYCTTHGNFGNSYVSWFAYWGQGTL VTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAV LQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKRVEPKSCEFGGGGSGGGGSG DELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQGGGGGSGGGGSGDELTQLKRELQKL YRQGVDSDELRALIRKLHMLEQGGGGGGSGGGGSGDELTQLKRELQKL YRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 150)
- the second polypeptide chain comprises an amino acid sequence of EVQLVESGGGLVKPGGSLRLSCAASGFTFNTYAMNWVRQAPGKGLEWVGRIRSKYNNYAT YYADSVKDRFTISRDDSKNTLYLQMNSLKTEDTAVYYCTTHGNFGNSYVSWFAYWGQGTL VTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAV LQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKRVEPKSCEFGGGGSGGGGSG DELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQGGGGGSGGGGSGDELTQLKRELQKL YRQGVDSDELRALIRKLHMLEQ (GGGGGSGGGGSGDELTQLKRELQKL YRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 151)
- the bispecific fusion molecule according to the present disclosure bind to human IL-13Ra2 with an ECso concentration of 10 nM or less, such as 5 nM, 3 nM, 2 nM, 1 nM, 0.6 nM, 0.5 nM, 0.4 nM or 0.3 nM.
- said IL-13Ra2 is expressed on cells. In an embodiment, said IL-13Ra2 is human IL-13Ra2 expressed on cells. In an embodiment, said IL-13Ra2 is cynomolgus IL- 13Ra2 expressed on cells. In an embodiment, said IL-13Ra2 is mouse IL-13Ra2 expressed on cells. In an embodiment, said binding is measured in an FACS assay as described in Example 11. In certain embodiment, the bispecific fusion molecule according the present disclosure may be capable of co-stimulating T-cell responses in a functional T-cell activation assay as described in Example 12. In an embodiment, the bispecific fusion molecule according to the present disclosure may be capable to mediate killing of IL-13Ra2 expressing cancer cells.
- the bispecific fusion molecule according to the present disclosure may be capable to induce T-cell mediated killing of cancer cells as described in Example 12. In an embodiment, the bispecific fusion molecule according to the present disclosure may be capable to induce T-cell mediated killing of A-375 cells in vitro as described in Example 12. In an embodiment, the bispecific fusion molecule according to the disclosure may be capable to induce T-cell mediated killing of A-375 cells in vitro with an IC50 concentration of 10 nM or less, such as 5 nM, 3 nM, 2 nM, 1 nM, 0.6 nM, 0.5 nM, 0.4 nM or 0.3 nM.
- the isolated polypeptide specific for IL-13Ra2 or the fusion molecule according to the present disclosure may be used in therapeutic methods.
- the polypeptide or fusion molecule may be used for the treatment of a disease, such as cancer, an autoimmune disease or inflammatory disease.
- the present disclosure provides a method for the treatment of a disease.
- the present disclosure provides an isolated polypeptide or a fusion molecule according to the present disclosure for the treatment of a disease. In an embodiment, the present disclosure provides an isolated polypeptide or a fusion molecule according to the present disclosure for use in the treatment of a disease. In an embodiment, the present disclosure provides an isolated polypeptide or a fusion molecule according to the present disclosure for use in the treatment of a disease in a subject in need thereof. In an embodiment, the present disclosure provides the use of an isolated polypeptide or a fusion molecule according to the present disclosure for the manufacture of a medicament. In an embodiment, the present disclosure provides an isolated polypeptide or a fusion molecule according to the present disclosure for use in medicine.
- the present disclosure provides an isolated polypeptide or a fusion molecule according to the present disclosure for use as a medicament. In an embodiment, the present disclosure provides an isolated polypeptide or a fusion molecule according to the present disclosure for use in medicine. In an embodiment, the present disclosure provides an isolated polypeptide or a fusion molecule according to the present disclosure for use as a medicament for the treatment of a subject in need thereof. In an embodiment, the disease is associated with the undesired presence of IL13-Ra2, in particular human IL-13Ra2.
- the disease to be treated is a proliferative disease.
- the disease is cancer.
- cancers include bladder cancer, brain cancer (such as glioblastoma), head and neck cancer, pancreatic cancer, lung cancer, breast cancer, ovarian cancer, uterine cancer, cervical cancer, endometrial cancer, esophageal cancer, colon cancer, colorectal cancer, rectal cancer, gastric cancer, prostate cancer, blood cancer, sarcoma, skin cancer, squamous cell carcinoma, bone cancer, melanoma, renal cell carcinoma, and kidney cancer.
- the disease to be treated is an autoimmune or inflammatory disease.
- an autoimmune or inflammatory disease include rheumatoid arthritis (RA), psoriasis, psoriatic arthritis, systemic lupus erythematosus (SLE), lupus nephritis, type I diabetes, Grave's disease, Inflammatory bowel disease (IBD), Crohn's disease (CD), ulcerative colitis (UC), irritable bowel syndrome, multiple sclerosis (MS), autoimmune myocarditis, Kawasaki disease, coronary artery disease, chronic obstructive pulmonary disease (COPD), interstitial lung disease, autoimmune thyroiditis, scleroderma, systemic sclerosis, osteoarthritis, atoptic dermatitis, vitiligo, graft vs.
- RA rheumatoid arthritis
- psoriasis psoriatic arthritis
- SLE systemic lupus erythemat
- the present disclosure provides an isolated polypeptide or a fusion molecule specific for IL-13Ra2 according to the present disclosure for use in a method of treating a subject having a disease comprising administering to the subject a therapeutically effective amount of a polypeptide or a fusion molecule according to the present disclosure.
- the method further comprises administering to the subject a therapeutically effective amount of at least one additional therapeutic agent.
- the subject in need of treatment is typically a mammal, more specifically a human.
- an isolated polypeptide or a fusion molecule according to the present disclosure would be formulated, dosed, and administered in a way consistent with good medical practice.
- the present disclosure provides a pharmaceutical composition
- a pharmaceutical composition comprising a polypeptide or a fusion molecule according to the present disclosure and a pharmaceutically acceptable carrier or excipient.
- the pharmaceutical compositions may further comprise at least one other pharmaceutically active compound.
- the pharmaceutical composition according to the present disclosure can be used in the diagnosis, prevention and/or treatment of diseases associated with the undesired presence of IL-13Ra2, in particular human IL-13Ra2.
- the present disclosure provides a pharmaceutical composition comprising a polypeptide or a fusion molecule according to the present disclosure that is suitable for prophylactic, therapeutic and/or diagnostic use in a mammal, more particular in a human.
- a polypeptide or a fusion molecule according to the present disclosure may be formulated as a pharmaceutical composition comprising at least one polypeptide or fusion molecule according to the present disclosure and at least one pharmaceutically acceptable carrier or excipient, and optionally one or more further pharmaceutically active compounds.
- Such a formulation may be suitable for oral, parenteral, topical administration or for administration by inhalation.
- a pharmaceutical composition comprising at least one polypeptide or fusion molecule according to the present disclosure may be administered parenterally, such as intravenously, or intramuscularly, or subcutaneously.
- a polypeptide or fusion molecule according of the disclosure may be administered via a non- parenteral route, such as per-orally or topically.
- a pharmaceutical composition comprising a polypeptide or a fusion molecule according to the present disclosure is administered intravenously or subcutaneously.
- a polypeptide or a fusion molecule according to the present disclosure may be used in combination with one or more pharmaceutically active compounds that are or can be used for the prevention and/or treatment of the diseases in which a target antigen of interest is involved, as a result of which a synergistic effect may or may not be obtained.
- pharmaceutically active compounds that are or can be used for the prevention and/or treatment of the diseases in which a target antigen of interest is involved, as a result of which a synergistic effect may or may not be obtained. Examples of such compounds, as well as routes, methods and pharmaceutical formulations or compositions for administering them will be clear to the clinician.
- the present disclosure provides a pharmaceutical composition comprising a polypeptide or a fusion molecule according to the present disclosure for use in the prevention and/or treatment of a disease associated with the undesired presence of IL-13Ra2.
- the present disclosure provides a pharmaceutical composition comprising a polypeptide or a fusion molecule according to the present disclosure for the use as a medicament.
- the present disclosure provides a pharmaceutical composition comprising a polypeptide or a fusion molecule according to the present disclosure for use in the prevention and/or treatment of an autoimmune disease and/or inflammatory disease and/or cancer.
- the present disclosure provides a method for the treatment of an autoimmune disease and/or inflammatory disease and/or cancer in a subject in need thereof using a pharmaceutical composition comprising a polypeptide or a fusion molecule according to the present disclosure.
- a method of producing a polypeptide or a fusion molecule according to the present disclosure in a form suitable for administration in vivo comprising (a) obtaining a polypeptide or a fusion molecule according by a method disclosed herein, and (b) formulating said polypeptide or fusion molecule with at least one pharmaceutically acceptable carrier or excipient, whereby a preparation of a polypeptide or fusion molecule is formulated for administration in vivo.
- compositions according to the present disclosure comprise a therapeutically effective amount of one or more polypeptides or fusion molecules according to the present disclosure dissolved in a pharmaceutically acceptable carrier or excipient.
- the present disclosure provides the use of an isolated polypeptide specific for IL-13Ra2 according to the present disclosure for diagnosis tic purposes. In an embodiment, the present disclosure provides the use of an isolated polypeptide specific for IL-13Ra2 according to the present disclosure for the diagnosis of a disease. In an embodiment, the present disclosure provides the use of an isolated polypeptide specific for IL-13Ra2 according to the present disclosure for the detection of IL-13Ra2, in particular human IL-13Ra2. In an embodiment, the present disclosure provides a method for detecting IL-13Ra2 in a subject or a sample, comprising the step of contacting said subject or sample with an isolated polypeptide specific for IL-13Ra2 according to the present disclosure. In an embodiment, the present disclosure provides a method for diagnosing a disease in a subject, comprising the step of contacting said subject or sample with an isolated polypeptide specific for IL-13Ra2 according to the present disclosure.
- Example 1 Antigen generation and quality control
- Selected IL-13Ra2-specific HTH polypeptides of the present disclosure were chemically synthesized either with a biotin tag or fluorescence dye (PSL GmbH, Germany). Alternatively, peptides were conjugated to cytotoxic drugs (such as MMAE, MMAF, DM-1 or DM-4) (PSL GmbH, Germany). Peptides were purified and delivered as lyophilized material. The lyophilized peptides were stored at -80°C and reconstituted in an appropriate buffer when needed.
- Human IL-13 and mouse IL-13Ra2 Recombinant human IL-13 (hlL-13) or mouse IL-13Ra2 was produced in house (see Table 2).
- DNA encoding hlL-13 (Uniprot: P35225) or mouse IL-13Ra2 (Uniprot: 088786) was cloned in frame with an N-terminal VK leader sequence and a C-terminal AVI-6xHis-tag into a pMAX expression vector, which is a modified expression vectTable 1or based on pcDNA3.1 (Thermo Fisher).
- HEK293-6E cells were transiently transfected one day post seeding with a commercially available transfection reagent according to the manufacturer’s instructions.
- the cells were cultured for 3 days and the conditioned cell culture supernatant was harvested by centrifugation followed by sterile filtration (0.22 pm).
- the conditioned cell culture supernatants were harvested by centrifugation followed by sterile filtration (0.22 pm).
- the antigen was purified by IMAC using Protino Ni-NTA columns (Macherey-Nagel). All chromatography steps were performed using AKTA chromatography systems (GE Healthcare).
- the samples were buffer-exchanged to D-PBS using PD10 columns (GE Healthcare).
- a polishing preparative SEC step was performed in D-PBS using a Superdex 200 column (GE Healthcare).
- biotinylation of the protein was performed by in vitro biotinylation using the BirA Kit (Avidity) followed by a preparative SEC using a Superdex 200 column (GE Healthcare).
- the quality of the samples was analyzed by denaturing, reducing or non-reducing SDS-PAGE, Streptavidin-Shift Assay, HP-SEC and DLS.
- Table 2 Amino acid sequences of produced antigens.
- CHO Flp-ln cells (Invitrogen) stably expressing full-length cynomolgus IL13Ra2 and CHO Flp- In cells transiently expressing full-length human IL-13Ra2 or mouse IL-13Ra were generated in-house.
- various vector constructs encoding the respective full-length receptor were gene synthesized in-house and transfection of cells was performed according to the instructor’s manual (Thermofischer I Invitrogen).
- HTH polypeptide phage-display library described in WO 2017/149117 (MORPHOSYS AG) was used for selections against human IL-13Ra2.
- the library is based on a de novo generated helix-turn- helix scaffold structure, wherein two a-helical peptides form a conformational constrained coiled-coil structure.
- Each of the two a-helical peptides is diversified at six solvent exposed positions leading to a total number of 12 diversified positions.
- Human IL-13Ra2 was used as a human Ig-Fc fusion protein (R&D Systems; Catalog Number: 7147-IR).
- coli TG1 F’ with an GD600nm of 0.6-0.8 was added to the phage eluates of each selection and was incubated in an incubator without shaking. After infection, bacteria were plated out evenly on two large LB/Chloramphenicol/Glucose agar plates for each selection and incubated overnight at 37°C and Glycerol phage stocks were prepared. For the following panning rounds bacterial suspensions of each pool were collected and used to propagate phages for an additional panning round as described above. After each round of panning the phage titer was determined. The expected range goes from 1.0E+06 - 1.0E+07 phage/ml for the output titer.
- phage output pools were subcloned via PGR into an expression vector to facilitate the cytoplasmic expression of the polypeptides in E. coli.
- Expression of single clones resulted in the production of polypeptides that were N-terminally fused to the Maltose Binding Protein (MBP) and that included a Factor FXa protease cleavage site.
- MBP Maltose Binding Protein
- all polypeptides were fused C-terminally to a Hiss-Flag tag (HHHHHHKDIRDYKDDDDK (SEQ ID NO: 181)).
- ELISA and FACS screenings with crude bacterial cell extracts containing the HTH polypeptides obtained from the panning outputs were carried out.
- ELISA screenings this was done by capturing the Fc-tagged hlL-13Ra2/Fc (R&D Systems; Catalog Number: 7147-IR) protein or unrelated Fc-tagged proteins (such as human Ox40/Fc (R&D Systems)) on the surface of a microtiter plate pre-coated with an anti-human Fc specific antibody (Jackson Immuno Research) and by adding E. coli lysates containing the polypeptides obtained from the panning outputs.
- mouse IL-13Ra2 this was done by direct coating of the avi-His tagged protein on the surface of a microtiter plate. Bound polypeptides were detected by the encoded M BP-tag using anti- MBP detection (Abeam). To analyze polypeptide expression, anti-His capture (R&D Systems) and anti-MBP detection (Abeam) was applied.
- ELISA screening experiments also included counter antigens human, mouse IL-13Ra1/Fc (R&D Systems; Catalog Number: 146-IR, Catalog Number: 491-IR/CF) and human, mouse IL4Ra/Fc (R&D Systems; Catalog Number: 7700-4R, Catalog Number: 530-MR) which helped to identify polypeptides truly specific for the IL-13Ra2 protein.
- mouse anti hlL-13Ra2 IgG BioLegend
- mouse anti hlL-13Ra1 IgG R&D Systems
- mouse anti hlL- 4Ra IgG R&D Systems
- Fab alkaline phosphate conjugated goat anti mouse IgG
- FACS based cell screening experiments on the cancer cell line A-375 expressing hlL-13Ra2 but not hlL13Ra1/IL4Ra as well as cell binding studies on CHO cells engineered to stably express cynomolgus monkey IL-13Ra2 further confirmed cell binding and species cross-reactivity of the identified HTH polypeptides.
- HTH polypeptides were expressed in E. coli cells as a N-terminally tagged maltose-binding protein (MBP) that includes a Factor FXa protease cleavage and a C- terminally tagged (His)e-Flag-fusion protein ("(His)6" disclosed as SEQ ID NO: 206). Protein expression was induced by the addition of IPTG and cultures were further cultivated for 20-23 h.
- MBP N-terminally tagged maltose-binding protein
- His C- terminally tagged (His)e-Flag-fusion protein
- HTH polypeptide fusion molecules were purified by Dextrin-Sepharose affinity chromatography using a MBP-Trap column (GE- Healthcare) and optionally polished by cation exchange chromatography using a Hi-Trap SP FF column (GE LifeSciences).
- Table 3 Amino acid sequences of bacterial produced IL-13Ra2 specific helix-turn-helix polypeptides including tags.
- M BP-free HTH polypeptides the purified M BP-fusion molecules were buffer- exchanged by PD10 columns (GE Healthcare) into FXa-digest buffer (20 mM Tris/HCI pH 8.0; 100 mM NaCI; 2 mM CaCh).
- the HTH polypeptides were released from the maltose-binding protein by addition of Factor Xa (1 :100 (w/w)) and incubation O/N in a rotary shaker at room temperature.
- the released peptides were further purified by IMAC using either 1 ml Protino IMAC columns or self-packed 1 ml columns filled with Profinity IMAC Ni-charged resin. All affinity chromatography steps were performed using the AKTA Avant 25 preparative chromatography system for low-throughput applications and using the Gilson ASPEC GX-274 solid phase extraction workstation for high-throughput applications. Buffer exchange to PBS was performed using PD 10 columns (GE Healthcare). Samples were sterile filtered and HTH polypeptide concentration was determined by UV-spectrophotometry. The purity and integrity of the samples were analyzed in denaturing, reducing or non-reducing SDS-PAGE, SEC- HPLC and CD-spectroscopy. Purified HTH polypeptides were re-characterized in more detail for ELISA and cell binding, affinity, and functional activity in a relevant in vitro assay.
- Example 3 Characterization of IL-13Ra2 specific MBP-free HTH-polypeptides for ELISA and cell binding (FACS)
- Fc fusion proteins were captured at saturating concentration via an anti-human-Fc- capture (Jackson Immuno Research) specific antibody on Maxisorp plates. Avi-His fusion proteins were direct coated at saturating concentrations on Maxisorp plates. Bound M BP-free HTH polypeptide fusions were detected using a mouse anti-FLAG M2 conjugated to alkaline phosphatase (AP) secondary detection antibody (Sigma).
- AP alkaline phosphatase
- FACS Cells were adjusted to 2x10 6 cells/ml in PBS/3% FCS/0.02% NaN 3 (FACS buffer). FACS staining was performed in V-bottom 96-well microtiter and 1x 10 5 cells per well were mixed with purified HTH-polypeptides (diluted in FACS buffer) and incubated on ice for 1h. After 1 h incubation on ice cells were washed 4x with FACS buffer and taken up in 50 pl phycoerythrin-conjugated (PE) anti-FLAG M2 secondary antibody (Jackson Immuno Research), diluted 1 :100 in FACS buffer.
- PE phycoerythrin-conjugated
- HTH-71 and HTH-125 both sharing preferred binding properties, were selected as a lead and back-up candidate, respectively.
- both molecules share almost identical amino acid sequences (Table 1 and Figure 1) with the only difference in 3 amino acid positions located at variable positions Var4, Var5, and Var6 in Helix-2 indicating that these positions are less important for antigen binding. This assumption was later confirmed in Ala-scanning experiments for HTH-71 and additional variants designed for HTH-71 (see Example 4).
- highly specific HTH-polypeptides specifically recognizing IL-13Ra2 were identified. These molecules can be further developed for diagnostic, imaging and therapeutic intervention of various types of cancer.
- Table 5 Amino acid sequence of the core helix-turn-helix sequence of IL-13Ra2 specific HTHs identified in ELISA and FACS experiments.
- Table 6 Overview ELISA and cell binding results using recombinantly produced HTH polypeptides.
- Lead HTH polypeptide HTH-71 was subjected to protein optimization.
- Variants were designed to remove potential sites of oxidation and/or isomerization in the invariant positions of the HTH-scaffold structure of HTH-71. Isomerization and/or oxidation of HTH polypeptides can lead to product heterogeneity and loss of activity.
- a M residue found in the invariant HTH-scaffold structure position X3 in Helix- 2 of HTH-71 was mutated to A.
- a D residue present in the Linker sequence Li was mutated to G to prevent potential isomerization. All variants were generated by site-directed mutagenesis according to the manufacturer's recommendations.
- Variant HTH polypeptides were produced in mammalian cells as Fab-HTH- HTH fusion molecules (bivalent HTH binding to IL-13Ra2, monovalent Fab binding to CD3) using glycine-serine tethers as described in Example 10 and assayed for binding to A-375 cells and for T-cell mediated killing of A-375 cells as described in Example 11 and 12.
- Table 7 HTH scaffold amino acid sequence of generated variants of HTH-71 used in the Fab-HTH-HTH fusion molecules. As negative control, only one HTH (HTH-58) molecule was fused to the CD3-specific Fab heavy chain.
- Table 8 Amino acid sequences of the Fab heavy chain - HTH - HTH fusion polypeptide.
- Table 9 Amino acid sequence of the Fab light chain used for the Fab - HTH - HTH fusion molecules.
- Fab-HTH-HTH fusion molecules comprising the negative control HTH polypeptide HTH-58 revealed no binding or killing of A-375 cells.
- Fab-HTH-HTH fusion molecules comprising a sequence optimized HTH-71 variant revealed similar binding to and killing of A-375 cells when compared to the parent Fab-HTH-HTH fusion. Accordingly, the removal of the potential isomerization site in the Linker sequence and the removal of the potential oxidation site in Helix-2 does not substantially affect binding and functional activity of the HTH polypeptides of the present disclosure.
- Example 5 Identification of amino acid residues required for target binding - alanine scanning experiments.
- Table 13 Summary of the sequences and functional properties of the tested Ala-variants of HTH-71.
- Table 14 Overview of variable positions on Helix-1 and Helix-2 of HTH-71 being relevant and not relevant for binding to IL-13Ra2.
- HTH- 125 differs from HTH-71 in variable positions Var4, Var5 and Var6 on Helix-2 (see Table 15) but has similar binding characteristics as HTH-71 (see Example 2) supporting that these variant positions are not relevant for binding to the IL-13Ra2 receptor.
- Table 15 Comparison of variable positions on Helix-1 and Helix-2 of HTH-71 and HTH-125.
- HTH-71 Additional variants of HTH-71 were designed to confirm the results of Example 4 and to further elucidate the aforementioned identified relevant positions of HTH-71 for binding to IL-13Ra2.
- Hise single MBP-free Hise/Flag tagged polypeptides
- Single MBP-free Hise/Flag tagged polypeptides (“Hise” disclosed as SEQ ID NO: 206) with monovalent binding to IL-13Ra2 were generated and recombinantly produced in bacteria and purified as described Example 1.
- Purified HTH polypeptides were tested for ELISA and cell binding to recombinant IL-13Ra2 proteins from different species as described in Example 1.
- IL-13Ra2 specific HTH polypeptides The ability of identified IL-13Ra2 specific HTH polypeptides to deliver therapeutic agents into the cytoplasm of targeted cells expressing IL13Ra2 was assessed by analyzing their capacity to induce IL13Ra2 receptor internalization on target cells.
- IL-13Ra2 specific HTH polypeptides HTH-71 (SEQ ID NO: 66), HTH-125 (SEQ ID NO: 77), and HTH-82 (SEQ ID NO: 78), as well as one negative control HTH polypeptide were chemically synthesized by an external service provider (JPT, Germany).
- HTH polypeptides were either N-terminally biotinylated or alternatively directly conjugated to the fluorescence dye Atto488.
- HTH polypeptides were expressing the full-length cynomolgus IL13Ra2 or parental CHO cells as negative control cells were used.
- Cell bound biotinylated HTH polypeptides were incubated with Streptavidin-Alexa488 (Jackson) and the formed complex was incubated at 4°C and in parallel at 37°C for different time points to allow for receptor internalization. After incubation, cells were immediately placed on ice.
- the amount of internalized fluorescence was determined based on the signal intensities of unquenched and quenched samples at individual time point using the following formula:
- N1 Fluorescence of unquenched sample at time point x
- HTH- 71 shows the fasted and highest rate of internalized fluorescence with up to 38% internalized fluorescence after 120 minutes.
- the identified IL-13Ra2 specific HTH polypeptides of the present disclosure efficiently internalize into cells expressing IL-13Ra2 and as such are well suited to deliver therapeutic agents into cells expressing IL13Ra2.
- Table 17 Internalized fluorescence of HTH-bio/Strep-A-488 complexes in CHO cells stably transfected with full-length IL13Ra2 protein.
- Example 7 Design of HTH polypeptide drug conjugates (PDCs)
- IL-13Ra2 specific HTH polypeptide drug conjugates were generated and tested for toxin-mediated in vitro killing of A-375 cells and A-549 cells.
- PDCs IL-13Ra2 specific HTH polypeptide drug conjugates
- MMAF Monomethyl-Auristatin F
- Monomethyl auristatin F is an antimitotic agent, which inhibits cell division by blocking the polymerisation of tubulin.
- PDC-1 MMAF - PEG9 - HTH-71 (one HTH polypeptide, one toxin)
- PDC-2 MMAF - PEG9 - HTH-71 - (G 4 S) 2 - HTH-71 (two HTH polypeptides, one toxin) ("(G 4 S) 2 " disclosed as SEQ ID NO: 190)
- PDC-3 MMAF - PEG9 - HTH-71 - (G 4 S) 2 - HTH-71 - PEG9 - MMAF (two HTH polypeptides, two toxins) ("(G 4 S) 2 " disclosed as SEQ ID NO: 190)
- the ability of the IL-13Ra2 specific PDCs from Example 7 to inhibit growth of the cancer cell lines A-375 cells and A-549 cells was assessed in in vitro growth assays.
- Results are summarized in Table 20 and shown in Figure 3. All tested PDCs mediated potent and dose dependent cell killing of the IL-13Ra2 positive cancer cell line A-375 with IC50 values down to 12 pM. Killing of the IL-13Ra2 negative cancer cell line A-375 revealed an assay window of up to 40.000 fold for PDC-2.
- the identified IL-13Ra2 specific HTH polypeptides of the present disclosure are well suited to deliver therapeutic agents into targeted cells expressing IL-13Ra2.
- Example 9 In vitro PDC growth assays in presence of IL-13 (competition assays)
- Example 8 In order to determine the ability of IL-13 to compete with the IL13Ra2 specific polypeptides of the present disclosure for binding to IL13Ra2, the in vitro growth assay of Example 8 was performed in the presence of varying concentrations of human IL-13.
- the competition assays were basically performed as described in Example 6 with the difference that serially diluted PDC-1 and PDC-3 were pre-incubated with varying concentration of recombinant human IL- 13 (diluted in culture media) before being added to the cells.
- Results are summarized in Table 21 and shown in Figure 4. Presence of human IL-13 clearly inhibited the anti-proliferative effects of the tested PDCs on A-375 cells in a dose dependent manner, indicating that the HTH polypeptides of the present invention compete with IL-13 for binding to IL-13Ra2. However, unlike natural IL-13, the HTH polypeptides does not bind to IL- 13Ra1 and as such does not interfere with IL-13 mediated signaling over the IL-13Ra1/IL-4Ra receptor complex. Notably, despite the reported high affinity binding of human IL-13 to human IL-13Ra2, IL13 concentrations of up to 50 pM did not significantly altered the inhibitory activity of the tested PDC constructs.
- Table 21 Results of in vitro cell killing of PDCs in presence of recombinant human IL-13.
- Example 10 Design and production of bispecific Fab-HTH polypeptide fusion molecules with specificity for CD3 and IL-13Roc2
- Therapeutic delivery systems which not only specifically recognize cancer cells but also enable the recruitment of immune cells, such as cytotoxic T-cells to their respective site of action were designed. This approach enables to kill such tumor cells by e.g. activated T-cells.
- Construct 1 a Fab-HTH format ( Figure 6A) comprising one monoclonal Fab fragment specific for CD3 epsilon fused to one HTH polypeptide specific for IL-13Ra2.
- Figure 6A the N- terminus of the HTH polypeptide is fused to the C-terminus of the Fab heavy chain via a glycine-serine tether.
- Construct 2 a Fab-HTH-HTH format ( Figure 6B) comprising one monoclonal Fab fragment specific for CD3 epsilon fused to two HTH-polypeptides specific for IL-13Ra2.
- a first HTH polypeptide is fused with its N-terminus to the C-terminus of the Fab heavy chain via a peptide tether and a second HTH-polypeptide is fused via its N-terminus to the C-terminus of the first HTH polypeptide with a peptide tether.
- the DNA encoding the entire designed Fab-heavy chain constant region genetically fused to the HTH-polypeptide were synthesized as double-stranded DNA fragments. All synthesized DNA fragments were cloned into suited mammalian human Fab expression vectors by replacing the parental Fab heavy chain constant region using standard molecular biology methods.
- the VH and VL domains of a CD3 specific monoclonal antibody disclosed in WO 2019/034580 were used. This CD3 specific antibody binds to the N-terminus of the epsilon chain of CD3.
- HTH-71 the IL-13Ra2 specific HTH polypeptides
- SEQ ID NO: 01 the VH and VL domains of the in-house negative isotype control antibody MQR03207 with specificity for hen-egg lysozyme were used.
- HTH-58 SEQ ID NO: 149 comprising the HTH scaffold template sequence used for generation of the HTH-phage library disclosed in WO 2017/149117 was used.
- Table 22 Overview of generated bispecific Fab-HTH - HTH polypeptide fusion molecules (Table discloses "(G 4 S) 2 " as SEQ ID NO: 190)
- Table 23 Amino acid sequences of the generated bispecific antibody-HTH-polypeptide fusion molecules
- eukaryotic HKB11 or HEK 293 cells were transfected with pYMexlO eukaryotic expression vector DNA encoding both polypeptides chains of Fab-HTH polypeptide fusions.
- Cell culture supernatant was harvested on day 3 or day 6 post transfection and subjected to Capture select lgG-CH1 affinity chromatography (MabSelect SURE, GE Healthcare) for antibody-peptide purification. All samples were sterile filtered (0.2 pm pore size).
- Fab-HTH polypeptide fusions were analyzed under denaturing, reducing and non-reducing conditions using a Labchip System (Caliper GXII, Perkin Elmer) or on SDS-PAGE. Protein concentrations were determined by UV- spectrophotometry and HP-SEC was performed to analyze Fab-peptide fusion preparations in native state.
- Table 24 summarizes yields and final monomer content of 3 produced bispecific constructs.
- the constructs could be generated by the described production and purification method with yields between 24 - 40 mg/L and final monomer content between 95 - 99 %.
- Example 11 Binding of bispecific Fab-HTH polypeptide fusion molecules to IL13-Ra2 expressed on cells
- A-375 (ATCC® CRL-1619TM) cells Flp-ln CHO cells engineered to express full-length cynomolgus IL-13Ra2 as well as HEK-293 cells engineered to express full-length mouse IL-13Ra2 were used.
- the human CD3 positive T cell leukemia cell line, Jurkat was used to confirm binding of the Fab to CD3 (data not shown).
- Table 25 Cell binding results of bispecific Fab-HTH polypeptide fusion molecules to IL13Ra2 expressed on cells
- Example 12 T-cell mediated killing of cancer cells induced by bispecific Fab - HTH polypeptide fusion molecules
- the produced bispecific Fab - HTH fusion molecules of Example 10 and 11 were analyzed for their potential to induce T-cell mediated killing of IL-13Ra2 expressing tumor cells in the presence of human T-cells or PBMCs.
- Diluted blood was transferred to SepMate-50 tubes (StemCell Technologies, #15063) containing 15 ml of Lymphoprep density gradient medium (Stemcell Technologies, #07811) and centrifuged for 20 min at 1200 x g at room temperature. Supernatant was transferred into a 50 ml conical tube, diluted to 45 ml with PBS containing 2% fetal bovine serum and 2 mM EDTA and centrifuged for 5 min at 800x g. Supernatant was discarded, cell pellet resuspended in 1 ml PBS containing 2 % fetal bovine serum.
- Cell suspensions were pooled and transferred to a 50 ml tube and diluted to 30ml PBS containing 2 % fetal bovine serum. Cells were pelleted by centrifugation for 5 min at 800 x g. The cell pellet was resuspended in 2 ml of 1x Pharm Lyse Red Blood Cell lysing buffer (BD, #555899) and incubated at 4°C for 10 min. PBS containing 2 % fetal bovine serum was added to a final volume of 15 ml. Cells were pelleted for 10 min at 120 x g and the supernatant decanted. The cells were washed twice with PBS containing 2 % fetal bovine serum and counted (CASY TT device, Beckmann Coulter).
- PBS TT device Beckmann Coulter
- 5,000 IL-13Ra2 expressing A-375 cells or IL-13Ra2 negative A- 549 cells were suspended in culture medium supplemented with 10% FCS, seeded in black 96 well assay plates (Corning) and incubated over night at 37°C and 5% CO2.
- HTH polypeptides of the present disclosure can be used in various approaches to efficiently target IL-13Ra2 expressing cells.
- Table 26 Re-directed T-cell mediated killing of cancer cells by bispecific Fab-HTH polypeptide fusion molecules
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Biochemistry (AREA)
- Zoology (AREA)
- Gastroenterology & Hepatology (AREA)
- Toxicology (AREA)
- Immunology (AREA)
- Biophysics (AREA)
- General Health & Medical Sciences (AREA)
- Genetics & Genomics (AREA)
- Medicinal Chemistry (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Cell Biology (AREA)
- Peptides Or Proteins (AREA)
Abstract
The present invention relates to polypeptides that are conformational constrained in an antiparallel, helix-turn-helix arrangement and which specifically bind to IL-13Rα2 but not to IL-13Rα1 and compete with IL-13 for binding to IL-13Rα2. The invention further relates to fusion proteins and pharmaceutical compositions comprising said polypeptides and to their use for diagnostic and therapeutic purposes.
Description
IL-13 RECEPTOR ALPHA 2 BINDING POLYPEPTIDES
FIELD OF THE INVENTION
The present invention provides IL- 13 receptor alpha 2 (IL-13Ra2) receptor binding polypeptides based on a helix-turn-helix scaffold structure (HTH polypeptide or Helix-turn-Helix polypeptide), fusion molecules comprising such polypeptides and pharmaceutical compositions containing the same, and methods of use thereof.
BACKGROUND OF THE INVENTION
IL-13Ra2 is overexpressed in a variety of human tumor types such as glioblastoma (Stupp, R. et al., N Engl J Med 352, 987-996 (2005); Zheng, S. et al., The journal of gene medicine 9, 151-160 (2007); Van Houdt, W. J. et al., J Neurosurg 104, 583-592 (2006)), renal cell carcinoma, pancreatic, melanoma, head and neck, mesothelioma and ovarian. In contrast, normal tissues express little to no IL-13Ra2, with the exception of the testes (Iliasov, I. V. et al., British journal of cancer 100, 1154-1164 (2009)). IL-13Ra2 exists on the cell membrane, intracellular, and in soluble form and has an extremely high affinity for IL-13. The strong binding affinity of IL-13 to IL-13Ra2 was interpreted as a way to sequester IL-13 and provoke its downregulation. However, recent reports revealed that membrane bound IL-13Ra2 might has IL-13 mediated signaling capabilities in cancer metastasis (WO 2019/086676).
Notably, IL-13Ra1 , a different receptor with low affinity for IL-13, is expressed ubiquitously by many tissues making it a poor candidate for selective targeting of tumor-specific therapeutic applications (Chakravarti, A. et al., Journal of the American Society of Clinical Oncology 20, 1063-1068 (2002); Uematsu, M. et al., Journal of neuro-oncology 72, 231-238 (2005); Choi, B. D. et al., Proceedings of the National Academy of Sciences of the United States of America 110, 270-275 (2013)). IL-13Ra1 forms a heterodimer with the alpha chain of the IL-4 receptor (IL-4Ra) that is a signaling IL-13 receptor.
Targeting IL- 13 receptors for cancer therapy has been the subject of numerous studies and different strategies, including immunotoxins, DNA vaccines and specific monoclonal antibodies. One approach for targeting IL-13Ra2 utilizes its cognate ligand IL-13, conjugated to cytotoxic molecules (W01996/029417). Some studies involved the use of IL-13 immunotoxin, containing a truncated version of Pseudomonas exotoxin, which was highly cytotoxic to renal cancer cells and other human solid tumors (Puri RK et al, Blood 1996; 87:4333- 9). IL-13 immunotoxin has been also used in a Phase III clinical trial with glioblastoma
patients, showing small but significant effects on survival (Kunwar S. et al, Neuro Oncol 2010; 12:871-81).
However, the problem of these strategies is the relative lack of specificity, as IL-13 also binds the low affinity receptor IL-13Ra1 , which is abundantly expressed in many normal tissues. In order to circumvent the aforementioned shortcoming, other approaches were developed utilizing de novo designed muteins of IL-13 with altered binding affinities towards IL-13Ra2 or IL13Ra1 (WO1999/051643, WO2002/018422, WO2013/112871). Other approaches utilize targeting peptides obtained from synthetic phage libraries (WO 2010121125A1). However, these peptides bind to a region on IL-13Ra2 not involved in IL-13 binding.
Therefore, developing small but selective polypeptides with high specificity for IL-13Ra2 and the capability to interfere with IL-13 binding to the same is expected to yield therapeutically beneficial results in diseases where IL-13Ra2 is expressed, such as cancer, in particular brain cancer.
SUMMARY OF THE INVENTION
The present disclosure provides novel polypeptides based on a conformational constrained helix-turn-helix scaffold structure (HTH polypeptide) which specifically bind to IL-13Ra2. In an embodiment, the polypeptides bind to human IL-13Ra2 and preferably cross-react with cynomolgus monkey and mouse IL-13Ra2.
In other embodiments, the polypeptides according to the present disclosure do not bind to IL- 13Ra1. In certain embodiments, the polypeptides according to the present disclosure do not bind to human IL-4Ra. In certain embodiments, the polypeptides according to the present disclosure do not bind to the human IL-13Ra1/IL-4Ra complex. In certain embodiments, said IL-13Ra1 or IL-4Ra is human or mouse IL-13Ra1 or IL-4Ra.
Thus, in certain embodiments, the polypeptides according to the present disclosure do not compete with IL-13 for binding to IL-13Ra1 or for binding to the IL13Ra1/IL-4Ra complex. Thus, in certain embodiments, the disclosed polypeptides do not interfere with IL-13 mediated signaling through the IL-13Ra1/IL-4Ra complex.
In certain embodiments, the polypeptides according to the present disclosure bind to the binding region of IL-13 on IL-13Ra2. Thus, in some embodiments, the polypeptides compete with IL-13 for binding to IL-13Ra2. In certain embodiments, the polypeptides may interfere with IL-13 mediated signaling through IL-13Ra2.
In addition, the polypeptides according to the present disclosure are small in size (~6kDa), reveal low immunogenicity, extreme stability against thermal and chemical denaturation, relatively insensitiveness to changes in pH and to proteolytic degradation. The HTH polypeptides according to the present disclosure may optionally be fused or conjugated to one or more moieties, such as therapeutic agents or detectable tags.
Accordingly, the polypeptides according to the present disclosure combine favorable structural, functional and safety properties never observed before. These features make the polypeptides superior to the IL-13 peptide mimetic approaches known from the art and as such highly desirable for diagnostic and therapeutic use, such as for preventing and/or treating cancer.
The polypeptides of the present disclosure are comprised of a helix-turn-helix scaffold structure of the formula Helix-1 - Li - Helix-2, wherein Li is a linker and wherein Helix-1 and Helix 2 comprise a first and second a-helical peptide, wherein said first and said second a-helical peptide form an antiparallel coiled-coiled structure.
Accordingly, the present disclosure pertains to an isolated polypeptide specific for IL-13 receptor alpha 2 (IL-13Ra2) comprising a helix-turn-helix scaffold structure. In an embodiment, said helix-turn-helix scaffold structure has the formula Helix-1 - Li - Helix-2, wherein Li is a linker and wherein Helix-1 and Helix 2 comprise a first and second a-helical peptide. In an embodiment, said first and said second a-helical peptide form an antiparallel coiled-coiled structure.
Accordingly, the present disclosure pertains to an isolated polypeptide specific for IL-13 receptor alpha 2 (IL-13Ra2) comprising a helix-turn-helix scaffold structure of the formula Helix-1 - Li - Helix-2, wherein Li is a linker and wherein Helix-1 and Helix 2 comprise a first and second a-helical peptide, wherein each of said a-helical peptides comprises the amino acid sequence
D - E - L - Vari -X1 - L - Var2 - Var3 - X2 - L - Var4 - X3 - L - Var5 - Var6 (Helix-1 disclosed as SEQ ID NO: 197; Helix-2 disclosed as SEQ ID NO: 204), wherein
Vari is any natural occurring amino acid excluding G, P and C in Helix-1 and Helix-2,
Var2 is K, R, T or I in Helix-1 and I or K in Helix-2,
Var3 is R or Q in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
Var4 is Q or K in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
Var5 is F, Y or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
Var6 is K, R or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
X1 is Q in Helix-1 and A in Helix-2,
X2 is E in Helix-1 and K in Helix-2, and
X3 is K in Helix-1 and M or A in Helix-2
In an embodiment, the present disclosure pertains to an isolated polypeptide specific for IL-13 receptor alpha 2 (IL-13Ra2) comprising a helix-turn-helix scaffold structure of the formula Helix-1 - Li - Helix-2, wherein Li is a linker and wherein Helix-1 comprise a first a-helical peptide and Helix- 2 comprise a second a-helical peptide, wherein each of said a-helical peptides comprises the amino acid sequence of
D - E - L - Vari -X1 - L - Var2 - Var3 - X2 - L - Var4 - X3 - L - Var5 - Var6 (Helix-1 disclosed as SEQ ID NO: 197; Helix-2 disclosed as SEQ ID NO: 204), wherein,
Vari is any natural occurring amino acid excluding G, P and C in Helix 1 and Helix 2,
Var2 is K, R, T or I in Helixl and I or K in Helix-2,
Var3 is R or Q in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
Var4 is Q or K in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
Var5 is F, Y or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
Var6 is K, R or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
X1 is Q in Helix-1 and A in Helix-2,
X2 is E in Helix-1 and K in Helix-2, and
X3 is K in Helix-1 and M or A in Helix-2.
In an embodiment, the present disclosure pertains to an isolated polypeptide specific for IL-13 receptor alpha 2 (IL-13Ra2) comprising a structure of the formula Helix-1 - Li - Helix-2, wherein Li is a linker and wherein Helix-1 comprises the amino acid sequence of
D - E - L - Vari -X1 - L - Var2 - Var3 - X2 - L - Var4 - X3 - L - Var5 - Var6 (SEQ ID NO: 197) and wherein Helix- 2 comprises the amino acid sequence of
D - E - L - Vari -X1 - L - Var2 - Var3 - X2 - L - Var4 - X3 - L - Var5 - Var6 (SEQ ID NO: 204), wherein
Vari is any natural occurring amino acid excluding G, P and C in Helix 1 and Helix 2,
Var2 is K, R, T or I in Helixl and I or K in Helix-2,
Var3 is R or Q in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
Var4 is Q or K in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
Var5 is F, Y or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
Var6 is K, R or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
X1 is Q in Helix-1 and A in Helix-2,
X2 is E in Helix-1 and K in Helix-2, and
X3 is K in Helix-1 and M or A in Helix-2.
In a further embodiment of the present disclosure,
Vari is A, T, H or K in Helix-1 and A, R, E or M in Helix-2,
Var3 is A, R, E or Q in Helix-2,
Var4 is A, H, D, I, or E in Helix-2,
Var5 is A, E, I, L, T, K or D in Helix-2, and
Var6 is A, Q, R or H in Helix-2
In one further embodiment of the present disclosure,
Vari is T in Helix-1 and R in Helix-2,
Var2 is K in Helix-1 and I Helix-2,
Var3 is R in Helix-1 and Helix-2,
Var4 is Q in Helix-1 and H or D in Helix-2,
Var5 is Y in Helix-1 and E or I in Helix-2, and
Var6 is R in Helix-1 and Q or R in Helix-1.
In an embodiment, the present disclosure provides an isolated polypeptide specific for IL- 13Ra2 as disclosed herein, wherein the first and second a-helical peptide form an antiparallel coiled-coiled structure. In an embodiment, the present disclosure provides an isolated polypeptide specific for IL-13Ra2 as disclosed herein, wherein Helix-1 and Helix-2 form an antiparallel coiled-coiled structure. In an embodiment, the isolated polypeptide specific for IL- 13 receptor alpha 2 (IL-13Ra2) according to the present disclosure comprises a helix-turn- helix scaffold structure. In an embodiment, the present disclosure provides an isolated polypeptide specific for IL-13Ra2 as disclosed herein, wherein Helix-1 comprises a first a- helical peptide and Helix-2 comprises a second a-helical peptide.
In an embodiment of the present disclosure Helix-1 comprises the amino acid sequence of SEQ ID NO: 197 and Helix-2 comprises the amino acid sequence of SEQ ID NO: 204. In an embodiment of the present disclosure the first a-helical peptide comprises the amino acid sequence of SEQ ID NO: 197 and the second a-helical peptide comprises the amino acid sequence of SEQ ID NO: 204.
In an embodiment, the isolated polypeptide specific for IL-13Ra2 according to the present disclosure is cross- reactive to mouse and cynomolgus IL-13Ra2.
In an embodiment, the isolated polypeptide specific for IL-13Ra2 according to the present disclosure competes with IL-13 for binding to IL-13Ra2. In an embodiment, the isolated
polypeptide according to the present disclosure does not bind to IL-13 receptor alpha 1 (IL- 13Ra1), IL-4 receptor alpha (IL-4Ra) or the IL-13Ra1/IL-4Ra complex.
In an embodiment, the isolated polypeptide specific for IL-13Ra2 according to the present disclosure is internalized from the surface of a cell expressing IL-13Ra2. In an embodiment of the present disclosure, said internalization occurs upon binding of said polypeptide to IL-13Ra2 present of the surface of said cell expressing IL-13Ra2.
In an embodiment, the present disclosure provides an isolated polypeptide specific IL-13Ra2 as disclosed herein, wherein Helix-1 or the first a-helical peptide comprises of the amino sequence selected from the group consisting of: DELTQLKRELQKLYR (SEQ ID NO: 1),
DELAQLKRELQKLYR (SEQ ID NO: 2), DELTQLKRELQKLYK (SEQ ID NO: 3),
DELTQLKRELQKLFR (SEQ ID NO: 4), DELHQLRRELQKLYR (SEQ ID NO: 5),
DELKQLRRELQKLYH (SEQ ID NO: 6), DELTQLRRELQKLHR (SEQ ID NO: 7).
DELTQLTRELKKLYR (SEQ ID NO: 8), and DELKQLIQELKKLYR (SEQ ID NO: 9).
In an embodiment, the present disclosure provides an isolated polypeptide specific for IL- 13Ra2 as disclosed herein, wherein Helix-2 or the second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQ (SEQ ID NO: 19), DELRALIRKLHALEQ (SEQ ID NO: 20), DELAALIRKLHMLEQ (SEQ ID NO: 21), DELRALIAKLHMLEQ (SEQ ID NO: 22), DELRALIRKLAMLEQ (SEQ ID NO: 23),
DELRALIRKLHMLAQ (SEQ ID NO: 24), DELRALIRKLHMLEA (SEQ ID NO: 25),
DELRALIRKLDMLIR (SEQ ID NO: 26), DELEALKRKLDMLLH (SEQ ID NO: 27),
DELMALI EKLIM LTH (SEQ ID NO: 28), DELRALIRKLHMLEH (SEQ ID NO: 29),
DELRALIRKLEMLKR (SEQ ID NO: 30), and DELRALIQKLDMLDR (SEQ ID NO: 31).
In a further embodiment, the present disclosure provides an isolated polypeptide specific for IL-13Ra2 as disclosed herein, wherein the linker Li comprises 1 to 30 amino acid residues. In an embodiment, said linker Li comprises the amino acid sequence QGVDS (SEQ ID NO: 182) or QGVGS (SEQ ID NO: 183).
In a further embodiment, the present disclosure provides an isolated polypeptide specific for IL-13Ra2 as disclosed herein, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of:
DELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 47), DELTQLKRELQKLYRQGVDSDELRALIRKLHALEQ (SEQ ID NO: 49), DELTQLKRELQKLYRQGVDSDELRALIRKLDMLIR (SEQ ID NO: 58), DELTQLKRELQKLYRQGVDSDELEALKRKLDMLLH (SEQ ID NO: 59),
DELHQLRRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 60), DELKQLRRELQKLYHQGVDSDELMALI EKLIM LTH (SEQ ID NO: 61), DELTQLRRELQKLHRQGVDSDELRALIRKLHMLEH (SEQ ID NO: 62), DELTQLRRELQKLHRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 63), DELTQLTRELKKLYRQGVDSDELRALIRKLEMLKR (SEQ ID NO: 64), and DELKQLIQELKKLYRQGVDSDELRALIQKLDMLDR (SEQ ID NO: 65).
In an embodiment, the present disclosure provides an isolated polypeptide specific for IL- 13Ra2 as disclosed herein, wherein a G is present at the N- and/or C-terminus of the helix- turn-helix scaffold structure.
In an embodiment, the present disclosure provides an isolated polypeptide specific for IL- 13Ra2 as disclosed herein, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of:
GDELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 66), GDELTQLKRELQKLYRQGVDSDELRALIRKLHALEQG (SEQ ID NO: 68), GDELTQLKRELQKLYRQGVDSDELRALIRKLDMLIRG (SEQ ID NO: 77), GDELTQLKRELQKLYRQGVDSDELEALKRKLDMLLHG (SEQ ID NO: 78), GDELHQLRRELQKLYRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 79), GDELKQLRRELQKLYHQGVDSDELMALIEKLIMLTHG (SEQ ID NO: 80), GDELTQLRRELQKLHRQGVDSDELRALIRKLHMLEHG (SEQ ID NO: 81), GDELTQLRRELQKLHRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 82), GDELTQLTRELKKLYRQGVDSDELRALIRKLEMLKRG (SEQ ID NO: 83), GDELKQLIQELKKLYRQGVDSDELRALIQKLDMLDRG (SEQ ID NO: 84), GDELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 85), GDELTQLKRELQKLYRQGVDSDELRALIRKLHALEQ (SEQ ID NO: 87), GDELTQLKRELQKLYRQGVDSDELRALIRKLDMLIR (SEQ ID NO: 96), GDELTQLKRELQKLYRQGVDSDELEALKRKLDMLLH (SEQ ID NO: 97), GDELHQLRRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 98), GDELKQLRRELQKLYHQGVDSDELMALIEKLIMLTH (SEQ ID NO: 99), GDELTQLRRELQKLHRQGVDSDELRALIRKLHMLEH (SEQ ID NO: 100), GDELTQLRRELQKLHRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 101), GDELTQLTRELKKLYRQGVDSDELRALIRKLEMLKR (SEQ ID NO: 102), and GDELKQLIQELKKLYRQGVDSDELRALIQKLDMLDR (SEQ ID NO: 103).
In a further embodiment, the present disclosure provides a fusion molecule comprising a polypeptide specific for IL-13 receptor alpha 2 (IL-13Ra2) according to the present disclosure and at least one additional moiety.
In an embodiment, said additional moiety is a selected from the group consisting of a drug, a therapeutic agent, an antibody or antibody fragment, a peptide, a polypeptide, a cytotoxic molecule, a cytokine, an enzyme, a moiety being capable of binding a metal ion, a tag suitable for detection and/or purification, a homo- or hetero-association domain, a moiety which increases solubility of a protein, a moiety which comprises an enzymatic cleavage site, or a lipid, a liposome and a virus-like-particle. In an embodiment of the present disclosure, said additional moiety is fused to the isolated polypeptide specific for IL-13Ra2 via a peptide tether. In an embodiment of the present disclosure, said fusion molecule is capable of mediating killing of IL-13Ra2 expressing cells. In an embodiment of the present disclosure, said additional moiety is a radionuclide, a chemotherapeutic agent, a cytotoxic agent or a detectable group. In an embodiment of the present disclosure, said additional moiety is an antibody or antibody fragment. In an embodiment of the present disclosure, said additional moiety is an antibody or antibody fragment specific for CD3. In an embodiment of the present disclosure, said fusion molecule is capable of re-directing cytotoxic activity of a T-cell to an IL-13Ra2 expressing cell. In an embodiment of the present disclosure, said antibody or antibody fragment specific for CD3 comprises a VH comprising the amino acid sequence of SEQ ID NO: 180 and a VL comprising the amino acid sequence of SEQ ID NO: 179.
The present disclosure also provides the polypeptide or the fusion molecule according to the present disclosure for use in medicine.
The present disclosure provides a nucleic acid composition comprising a nucleic acid sequence or a plurality of nucleic acid sequences encoding the polypeptide or the fusion molecules according to the present disclosure. The present disclosure provides a vector composition comprising a vector or a plurality of vectors comprising the nucleic acid composition according to the present disclosure. The present disclosure provides a host cell comprising the vector composition or the nucleic acid composition according to the present disclosure.
The present disclosure also provides a pharmaceutical composition comprising the polypeptide or the fusion molecule according to the present disclosure and a pharmaceutically acceptable carrier or excipient. The present disclosure also provides methods for treating a subject suffering from a disease, such as cancer, by administering to said subject an effective
amount of a polypeptide or the fusion molecule or the pharmaceutical composition according to the present disclosure. Preferably, said subject is a human.
BRIEF DESCRIPTION OF THE DRAWINGS
Figure 1 : Amino acid sequence alignment of isolated IL-13Ra2 specific helix-turn-helix (HTH) polypeptides and generated variants. Variable positions (Varx) of the employed phage-library are indicated in black. Invariant positions on Helix-1 and Helix-2 but bearing distinct amino acids residues on Helix- 1 and Helix-2 are denotes as “Xn”.
Figure 2: Internalization rate of indirectly fluorescence-labeled IL-13Ra2 specific HTH- polypeptides after being exposed to CHO-cells stably expressing the cynomolgus IL-13Ra2 receptor.
Figure 3: Inhibition of in vitro cancer cell proliferation mediated by HTH polypeptide - MMAF drug conjugates (PDCs). A-375 (IL-13Ra2 positive) and A-549 (IL-13Ra2 negative) cancer cells were exposed to 3 different HTH-71 / MMAF drug conjugates. For A-375 cells, the number of viable cells was strongly reduced by the presence of PDCs in dose dependent manner whereas viability of A-549 cells was only affected at PDC concentrations of greater 100 nM. PDCs comprising two HTH-polypeptides (PDC-2, PDC-3) were significant more potent compared to PDC with only one HTH-polypeptide (PDC-1).
Figure 4: Inhibition of in vitro cancer cell proliferation of A-375 cells mediated by two HTH polypeptide - MMAF drug conjugates (Figure A: PDC-1 ; Figure 4 B: PDC-2) in the presence of human IL-13. The inhibitory activity of both tested PDCs was reduced in presence of human IL-13 at IL-13 concentration of greater 50 pM.
Figure 5: Cytotoxicity assay of bispecific Fab-HTH polypeptide fusion molecules comprising a CD3 specific Fab and IL-13Ra2 specific HTH polypeptides according to the present disclosure on IL-13Ra2 expressing A-375 cells in presence of human derived T-cells or PBMCs. Figure 5A is a graph showing the relative fluorescence of A-375 cells as a function of BIS228 or BIS602 concentration in presence of human T-cells. Figure 5B indicates the same as Figure 5A but with results obtained for BIS659 in the presence of human PBMCs derived from one donor.
Figure 6: Illustration of generated Fab-HTH-polypeptide (A) and Fab-HTH-HTH (B) fusion molecules.
DETAILED DESCRIPTION OF THE INVENTION
Definitions
The term “IL-13Ra2” refers to a protein known as IL-13 receptor alpha 2.
Human IL-13Ra2 (1-380) has the amino acid sequence of (Uniprot: Q14627):
MAFVCLAIGCLYTFLISTTFGCTSSSDTEIKVNPPQDFEIVDPGYLGYLYLQWQPPLSL DHFKECTVEYELKYRNIGSETWKTIITKNLHYKDGFDLNKGIEAKIHTLLPWQCTNGS EVQSSWAETTYWISPQGIPETKVQDMDCVYYNWQYLLCSWKPGIGVLLDTNYNLFY WYEGLDHALQCVDYIKADGQNIGCRFPYLEASDYKDFYICVNGSSENKPIRSSYFTF QLQNIVKPLPPVYLTFTRESSCEIKLKWSIPLGPIPARCFDYEIEIREDDTTLVTATVEN ETYTLKTTNETRQLCFWRSKVNIYCSDDGIWSEWSDKQCWEGEDLSKKTLLRFWL PFGFILILVIFVTGLLLRKPNTYPKMIPEFFCDT (SEQ ID NO: 123). Underlined = Signal sequence
Cynomolgus monkey IL-13Ra2 (1-380) has the amino acid sequence of:
MDFVYLAIRCLCTFLISTTFGYTSSSDTEIKVNPPQDFEIVDPGYLGYLYLQWQPPLSL DNFKECTVEYELKYRNIGSETWTTIITKNLHYKDGFDLNKGIEAKIHTLLPWQCTNGS EVQSSWAEATYWISPQGIPETKVQDMDCVYYNWQYLLCSWKPGIGVLLDTNYNLFY WYEGLDRALQCVDYIKVDGQNIGCRFPYLESSDYKDFYICVNGSSETKPIRSSYFTF QLQNIVKPLPPVCLTCTQESLYEIKLKWSIPLGPIPARCFVYEIEIREDDTTLVTTTVEN ETYTLKITNETRQLCFWRSKVNIYCSDDGIWSEWSDKQCWEVEELLKKTLLLFLLPF GFILILVIFVTGLLLCKRDSYPKMIPEFFCDR (SEQ ID NO: 124). Underlined = Signal sequence
Murine IL-13Ra2 (1-383) has the amino acid sequence of (UniProt: 088786):
MAFVHIRCLCFILLCTITGYSLEIKVNPPQDFEILDPGLLGYLYLQWKPPVVIEKFKGCT LEYELKYRNVDSDSWKTIITRNLIYKDGFDLNKGIEGKIRTHLSEHCTNGSEVQSPWI EASYGISDEGSLETKIQDMKCIYYNWQYLVCSWKPGKTVYSDTNYTMFFWYEGLDH ALQCADYLQHDEKNVGCKLSNLDSSDYKDFFICVNGSSKLEPIRSSYTVFQLQNIVK PLPPEFLHISVENSIDIRMKWSTPGGPIPPRCYTYEIVIREDDISWESATDKNDMKLKR RANESEDLCFFVRCKVNIYCADDGIWSEWSEEECWEGYTGPDSKIIFIVPVCLFFIFL LLLLCLIVEKEEPEPTLSLHVDLNKEVCAYEDTLC (SEQ ID NO: 125). Underlined = Signal sequence
The term “IL-13Ra1” refers to a protein known as IL-13 receptor alpha 1.
Human IL-13Ra1 (1-427) has the amino acid sequence of (Uniprot: P78552):
MEWPARLCGLWALLLCAGGGGGGGGAAPTETQPPVTNLSVSVENLCTVIWTWNPP EGASSNCSLWYFSHFGDKQDKKIAPETRRSIEVPLNERICLQVGSQCSTNESEKPSI LVEKCISPPEGDPESAVTELQCIWHNLSYMKCSWLPGRNTSPDTNYTLYYWHRSLE KIHQCENIFREGQYFGCSFDLTKVKDSSFEQHSVQIMVKDNAGKIKPSFNIVPLTSRV KPDPPHIKNLSFHNDDLYVQWENPQNFISRCLFYEVEVNNSQTETHNVFYVQEAKC ENPEFERNVENTSCFMVPGVLPDTLNTVRIRVKTNKLCYEDDKLWSNWSQEMSIGK KRNSTLYITMLLIVPVIVAGAIIVLLLYLKRLKIIIFPPIPDPGKIFKEMFGDQNDDTLHWK KYDIYEKQTKEETDSWLIENLKKASQ (SEQ ID NO: 126). Underlined = Signal sequence
Mouse IL-13Ra1 (1-424) has the amino acid sequence of (Uniprot: 009030):
MARPALLGELLVLLLWTATVGQVAAATEVQPPVTNLSVSVEN LCTI I WTWSPPEGAS PNCTLRYFSHFDDQQDKKIAPETHRKEELPLDEKICLQVGSQCSANESEKPSPLVKK CISPPEGDPESAVTELKCIWHNLSYMKCSWLPGRNTSPDTHYTLYYWYSSLEKSRQ CENIYREGQHIACSFKLTKVEPSFEHQNVQIMVKDNAGKIRPSCKIVSLTSYVKPDPP HIKHLLLKNGALLVQWKNPQNFRSRCLTYEVEVNNTQTDRHNILEVEEDKCQNSES DRNMEGTSCFQLPGVLADAVYTVRVRVKTNKLCFDDNKLWSDWSEAQSIGKEQNS TFYTTMLLTIPVFVAVAVIILLFYLKRLKIIIFPPIPDPGKIFKEMFGDQNDDTLHWKKYDI YEKQSKEETDSWLIENLKKAAP (SEQ ID NO: 127). Underlined = Signal sequence
The term “IL-13” refers to a protein known as Interleukin-13.
Human IL-13 (1-146) has the amino acid sequence of (Uniprot: P35225):
MHPLLNPLLLALGLMALLLTTVIALTCLGGFASPGPVPPSTALRELIEELVNITQNQKA PLCNGSM VWSI N LTAGMYCAALESLI N VSGCSAI EKTQRM LSGFCPH KVSAGQFSSL HVRDTKIEVAQFVKDLLLHLKKLFREGRFN (SEQ ID NO: 128) Underlined = Signal sequence
The term “IL-4Ra” refers to a protein known as lnterleukin-4 receptor subunit alpha.
Human IL-4Ra (1-825) has the amino acid sequence of (Uniprot: P24394):
MGWLCSGLLFPVSCLVLLQVASSGNMKVLQEPTCVSDYMSISTCEWKMNGPTNCS TELRLLYQLVFLLSEAHTCIPENNGGAGCVCHLLMDDVVSADNYTLDLWAGQQLLW KGSFKPSEHVKPRAPGNLTVHTNVSDTLLLTWSNPYPPDNYLYNHLTYAVNIWSEN DPADFRIYNVTYLEPSLRIAASTLKSGISYRARVRAWAQCYNTTWSEWSPSTKWHN SYREPFEQHLLLGVSVSCIVILAVCLLCYVSITKIKKEWWDQIPNPARSRLVAIIIQDAQ
GSQWEKRSRGQEPAKCPHWKNCLTKLLPCFLEHNMKRDEDPHKAAKEMPFQGSG KSAWCPVEISKTVLWPESISVVRCVELFEAPVECEEEEEVEEEKGSFCASPESSRDD FQEGREGIVARLTESLFLDLLGEENGGFCQQDMGESCLLPPSGSTSAHMPWDEFP SAGPKEAPPWGKEQPLHLEPSPPASPTQSPDNLTCTETPLVIAGNPAYRSFSNSLS QSPCPRELGPDPLLARHLEEVEPEMPCVPQLSEPTTVPQPEPETWEQILRRNVLQH
GAAAAPVSAPTSGYQEFVHAVEQGGTQASAWGLGPPGEAGYKAFSSLLASSAVS
PEKCGFGASSGEEGYKPFQDLIPGCPGDPAPVPVPLFTFGLDREPPRSPQSSHLPS SSPEHLGLEPGEKVEDMPKPPLPQEQATDPLVDSLGSGIVYSALTCHLCGHLKQCH GQEDGGQTPVMASPCCGCCCGDRSSPPTTPLRAPDPSPGGVPLEASLCPASLAPS GISEKSKSSSSFHPAPGNAQSSSQTPKIVNFVSVGPTYMRVS (SEQ ID NO: 129) Underlined = Signal sequence
Mouse IL-4Ra (1-810) has the amino acid sequence of (Uniprot: P16382):
MGRLCTKFLTSVGCLILLLVTGSGSIKVLGEPTCFSDYIRTSTCEWFLDSAVDCSSQL CLHYRLMFFEFSENLTCIPRNSASTVCVCHMEMNRPVQSDRYQMELWAEHRQLWQ GSFSPSGNVKPLAPDNLTLHTNVSDEWLLTWNNLYPSNNLLYKDLISMVNISREDNP AEFIVYNVTYKEPRLSFPINILMSGVYYTARVRVRSQILTGTWSEWSPSITWYNHFQL PLIQRLPLGVTISCLCIPLFCLFCYFSITKIKKIWWDQIPTPARSPLVAIIIQDAQVPLWD KQTRSQESTKYPHWKTCLDKLLPCLLKHRVKKKTDFPKAAPTKSLQSPGKAGWCP MEVSRTVLWPENVSVSVVRCMELFEAPVQNVEEEEDEIVKEDLSMSPENSGGCGF QESQADIMARLTENLFSDLLEAENGGLGQSALAESCSPLPSGSGQASVSWACLPM GPSEEATCQVTEQPSHPGPLSGSPAQSAPTLACTQVPLVLADNPAYRSFSDCCSPA PNPGELAPEQQQADHLEEEEPPSPADPHSSGPPMQPVESWEQILHMSVLQHGAAA GSTPAPAGGYQEFVQAVKQGAAQDPGVPGVRPSGDPGYKAFSSLLSSNGIRGDTA
AAGTDDGHGGYKPFQNPVPNQSPSSVPLFTFGLDTELSPSPLNSDPPKSPPECLGL
ELGLKGGDWVKAPPPADQVPKPFGDDLGFGIVYSSLTCHLCGHLKQHHSQEEGGQ SPIVASPGCGCCYDDRSPSLGSLSGALESCPEGIPPEANLMSAPKTPSNLSGEGKG PGHSPVPSQTTEVPVGALGIAVS (SEQ ID NO: 130) Underlined = Signal sequence
Throughout this specification, unless the context requires otherwise, the words "comprise", “have” and “include” and their respective variations such as "comprises", "comprising", “has”, “having”, “includes” and “including” will be understood to imply the inclusion of a stated element or integer or group of elements or integers but not the exclusion of any other element or integer or group of elements or integers.
By the term “peptide” is meant a short molecule having less than or equal to 20 amino acids.
The term “polypeptide” means a molecule having more than 20 amino acids.
A “fusion molecule" as meant herein refers to a polypeptide according to the present disclosure linked to at least further moiety. Such moiety may have a different property compared to the polypeptide. Such property may be a biological property, such as activity in vitro or in vivo. The property may also be a simple chemical or physical property, such as binding to a target molecule, catalysis of a reaction, etc.. The fusion or conjugation may be covalent or non-covalent. Preferably, the fusion or conjugation is covalent.
As used herein, the term “helix-turn-helix scaffold” or “HTH scaffold” refers to a secondary structure of a polypeptide in which two a-helices are orientated in a parallel or an anti-parallel orientation, and in which the two a -helices are linked via a short stretch of amino acids.
The terms “coiled-coil” and “coiled-coil structure” are used interchangeably herein and will be clear to the person skilled in the art based on the common general knowledge and the description and further references cited herein. In general, a coiled-coil structure is used by nature to stabilize a-helices in proteins. A coiled-coil is a structural motif in polypeptides or proteins in which 2 to 7 a-helices are coiled together. The coiled-coil formation of a-helical peptides is facilitated through a burial of hydrophobic side chains by arranging them on one side of the a-helices so that they are not accessed by polar water molecules. A typical coiled- coil motif (4-3 hydrophobic repeat) is a heptad repeat of amino acids from ‘a’ to ‘g’ so that ‘a’ and ‘d’ are hydrophobic. Particular reference in this regard is made to review papers concerning coiled-coil structures, such as for example, Cohen and Parry Proteins 1990, 7:1- 15; Kohn and Hodges Trends Biotechnol 1998, 16:379-389; Schneider et al Fold Des 1998, 3:R29-R40; Harbury et al. Science 1998, 282:1462-1467; Mason and Arndt Chem Bio Chem 2004, 5:170-176; Lupas and Gruber Adv Protein Chem 2005, 70:37-78; Woolfson Adv Protein Chem 2005, 70:79-112; Parry et al. J Struct Biol 2008, 163:258-269; McFarlane et al. Eur J Pharmacol 2009, 625:101-107.
The terms “heptad”, “heptad unit”, “heptad repeat” are used interchangeably herein and refer to a 7-mer peptide fragment that is repeated two or more times within a HTH scaffold. The tertiary structure of a a-helix is such that 7 amino acid residues in the primary sequence correspond to approximately 2 turns of the a-helix. Accordingly, a primary amino acid sequence giving rise to a a-helical conformation may be broken down into units of 7 residues each. The individual positions of a heptad unit are denoted by small letters, i.e. one heptad unit is for example represented by the sequence ‘abcdefg’, ‘bcdefga’, ‘cdefgab’, ‘defgabc’, ‘efgabcd’, ‘fgabcde’ or ‘gabcdef’. The ‘a’ and the ‘d’ position of a heptad unit assembled in an HTH scaffold of the present disclosure are of hydrophobic nature. These positions are typically either leucine, isoleucine or valine, and the parallel or the anti-parallel secondary structure of the HTH are formed by hydrophobic interactions via these positions between different heptad units present on two distinct a-helical peptides.
As used herein, the term “anti-parallel” refers to an HTH scaffold in which two a-helical peptides of an HTH scaffold are arranged such that the amino-terminal end (N-terminus) of one a-helical peptide is aligned with the carboxyl-terminal end (C-terminus) of the second a- helical peptide, and vice versa. Thus, the relative orientation of the heptad ‘a-g’ positions of two interacting a-helices aligned in an anti-parallel orientation is in the opposite direction. For instance, if the heptad positions of a first helix is defined as ‘abcdefg’ as read from the amino- to the carboxyl-terminus, the heptad positions of a second a-helix in an anti-parallel orientation would be defined as ‘gfedcba’ as read from the amino- to the carboxyl-terminus.
As used herein, the term “parallel” refers to an HTH scaffold in which the two a-helical peptides are aligned such that they have the same orientation such that the amino-terminal end of one helix is aligned with the amino-terminal end of the second a-helix, and vice versa. Thus, the relative orientation of the heptad ‘a-g’ positions of two interacting a-helical peptides aligned in parallel orientation is in the same direction. For instance, if the heptad positions of a first helix is defined as ‘abcdefg’ from the amino-terminus to the carboxyl-terminus, the heptad positions of a second helix in a parallel orientation would be also defined as ‘abcdefg’ as read from the amino- to the carboxyl-terminus.
The terms “linker”, “turn”, “linker sequence” or “turn sequence” are used interchangeably herein and refer to an amino acid sequence that is part of the contiguous amino acid sequence of an HTH polypeptide, and covalently links the two a-helical peptide sequences of that polypeptide.
As used herein, the term “single-chain” refers to the HTH scaffold of the present disclosure, wherein the stabilizing coiled-coil structure is formed from different regions of a contiguous amino acid sequence of an HTH polypeptide chain folded back in an appropriate manner.
The term “solvent-oriented” or “solvent-exposed” refers to the region of an entity which is directly exposed or which comes directly into contact with the solvent in the environment or the milieu in which it is present. In the context of the present disclosure it is the a -helix or a a- helical part of an HTH scaffold which is directly exposed or which comes directly into contact with the solvent in the environment or the milieu in which it is present. More particularly, in the context of a binding site, where one or more amino acids located in a solvent-oriented part of the HTH scaffold contribute to the binding site, the binding site is considered to be formed by a solvent-oriented part of the HTH scaffold.
A “a-helical part” of a polypeptide refers to a part of a polypeptide of the present disclosure that has an a-helical secondary structure.
The “hydrophobic core” of an HTH scaffold refers to the part on an HTH scaffold which is not directly exposed to the solvent in which it is present.
As used herein, a polypeptide of the present disclosure “binds specifically to”, “specifically binds to”, is “specific to/for” an antigen, if such polypeptide is able to discriminate between such antigen and one or more reference antigen(s), since binding specificity is not an absolute, but a relative property. The reference antigen(s) may be one or more closely related antigen(s), which are used as reference points. For example, specific binding can be determined with a standard ELISA assay. Alternative methods comprise, but are not limited to Western blots, ELISA-, RIA-, ECL-, IRMA-tests and peptide scans. The scoring may be carried out by standard color development (e.g. detection antibody with horseradish peroxide and tetramethyl benzidine with hydrogen peroxide). The reaction in certain wells is scored by the optical density, for example, at 450 nm. Typical background (=negative reaction) may be 0.1 OD; typical positive reaction may be 1 OD. This means the difference positive/negative can be more than 10-fold. Typically, determination of binding specificity is performed by using not a single reference antigen, but a set of about three to five unrelated antigens, such as milk powder, BSA, transferrin or the like. Additionally, “specific binding” may relate to the ability to discriminate between different parts of its target antigen, e.g. different domains or regions of said target antigen, or between one or more key amino acid residues or stretches of amino acid residues of a target antigen.
The “affinity” of a polypeptide is represented by the equilibrium constant for the dissociation of the polypeptide and the target protein of interest to which it binds. The lower the KD value,
the stronger the binding strength between the said polypeptide and the target protein of interest to which it binds. Alternatively, the affinity can be expressed in terms of the affinity constant (KA), which corresponds to 1/KD. The binding affinity of a polypeptide can be determined in a manner known to the skilled person, depending on the specific target protein of interest. It is generally known in the art that the KD can be expressed as the ratio of the dissociation rate constant of a complex, denoted as koff (expressed in seconds-1 or s-1), to the rate constant of its association, denoted kon (expressed in molar-1 seconds-1 or M-1 s-1). A KD value greater than about 1 millimolar is considered to indicate non-binding or non-specific binding.
The terms "inhibition" or "inhibit" or “reduction” or “reduce” refer to a decrease or cessation of any phenotypic characteristic (such as binding or a biological activity or function) or to the decrease or cessation in the incidence, degree, or likelihood of that characteristic. The “inhibition”, “reduction” or “neutralization” needs not to be complete as long as it is detectable using an appropriate assay. In some embodiments, by "reduce" or "inhibit" or “neutralize” is meant the ability to cause a decrease of 20% or greater. In another embodiment, by "reduce" or "inhibit" or “neutralize” is meant the ability to cause a decrease of 50% or greater. In yet another embodiment, by "reduce" or "inhibit" or “neutralize” is meant the ability to cause an overall decrease of 75%, 85%, 90%, 95%, or greater. The inhibiting activity of a polypeptide of the present disclosure may be reversible or irreversible, but for pharmaceutical and pharmacological applications will typically occur reversibly. The inhibiting activity of a polypeptide of the present disclosure may be measured using a suitable in vitro, cellular or in vivo assay.
The term “EC50” as used herein, refers to the concentration of an antibody or antibody fragment or ligand, which induces a response in an assay half way between the baseline and maximum. It therefore represents the antibody or ligand concentration at which 50% of the maximal effect is observed
The term “IC50” as used herein, refers to the concentration of an antibody or antibody fragment that inhibits a response in an assay half way between the maximal response and the baseline. It represents the antibody concentration that reduces a given response by 50%.
The term “synthetic” describes a molecule that is made outside of the human body by synthesis or synthesized.
The term "isolated” refers to a compound which can be e.g. a polypeptide according to the present disclosure, that is substantially free of other polypeptides having different antigenic specificities. Moreover, an isolated polypeptide may be substantially free of other cellular material and/or chemicals.
“Constrained” as used in the present disclosure refers to a polypeptide in which the three- dimensional structure is maintained substantially in one spatial arrangement over time. The polypeptides within the present disclosure have a constrained conformation. Methods of determining whether peptides or polypeptides are constrained are known in the art.
As used herein, amino acid residues will be indicated either by their full name or according to the standard three-letter or one-letter amino acid code. “Natural occurring amino acids” means the following amino acids:
Table 1 : Natural occurring amino acids
The terms “binding region”, “binding site” and “interaction site” as used herein refer to a particular site, part, domain or stretch of amino acid residues present on the polypeptides of the present disclosure that is responsible for binding to a target molecule. Such binding region consists of specific amino acids from the said polypeptide which are in contact with the target molecule. Alternatively, the terms refer to a particular site, part, domain or stretch of amino acids present on the target antigen (e.g. IL-13Ra2) that are recognized and bound by the polypeptides of the present disclosure. Such binding region consists of specific amino acids from said target molecule, which are in contact with the polypeptides of the present disclosure.
The term “vector” refers to a polynucleotide molecule capable of transporting another polynucleotide to which it has been linked. Preferred vectors are those capable of autonomous replication and/or expression of nucleic acids to which they are linked. One type of vector is a “plasmid”, which refers to a circular double stranded DNA loop into which additional DNA segments may be ligated. Another type of vector is a viral vector, wherein additional DNA segments may be ligated into the viral genome. Certain vectors are capable of autonomous replication in a host cell into which they are introduced (e.g., bacterial vectors having a bacterial origin of replication and mammalian vectors). Other vectors can be integrated into the genome of a host cell upon introduction into the host cell, and thereby are replicated along with the host genome. Vectors may be compatible with prokaryotic or eukaryotic cells. Prokaryotic vectors typically include a prokaryotic replicon which may include a prokaryotic promoter capable of directing the expression (transcription and translation) of the peptide in a bacterial host cell, such as Escherichia coli transformed therewith. A promoter is an expression control element formed by a DNA sequence that permits binding of RNA polymerase and transcription to occur. Promoter sequences compatible with bacterial hosts are typically provided in plasmid vectors containing convenience restriction sites for insertion of a DNA segment. Examples of such vector plasmids include pUC8, pUC9, pBR322, and pBR329, pPL and pKK223, available commercially. "Expression vectors" are those vectors capable of directing the expression of nucleic acids to which they are operatively linked and is intended to include such other forms of expression vectors, such as viral vectors (e.g., replication defective retroviruses, adenoviruses and adeno-associated viruses), which serve equivalent functions.
The term “recombinant host cell” (or “host cell”) refers to a cell into which a recombinant expression vector has been introduced. It should be understood that such terms are intended to refer not only to the particular subject cell but to the progeny of such a cell. Because certain modifications may occur in succeeding generations due to either mutation or environmental influences, such progeny may not, in fact, be identical to the parent cell, but are still included within the scope of the term “host cell” as used herein. Typical host cells are prokaryotic (such as bacterial, including but not limited to E. coli) or eukaryotic (which includes yeast, mammalian
cells, and more). Bacterial cells are preferred prokaryotic host cells and typically are a strain of Escherichia coli (E. coli) such as, for example, the E. coli strain DH5 available from Bethesda Research Laboratories, Inc., Bethesda, Md. Preferred eukaryotic host cells include yeast and mammalian cells including murine and rodents, preferably vertebrate cells such as those from a mouse, rat, monkey or human cell line, for example HKB11 cells, PERC.6 cells, or CHO cells.
The term “antibody” as used herein refers to a protein comprising at least two heavy (H) chains and two light (L) chains inter-connected by disulfide bonds which interacts with an antigen. Each heavy chain is comprised of a heavy chain variable region (abbreviated herein as VH) and a heavy chain constant region. The heavy chain constant region is comprised of three domains, CH1 , CH2 and CH3. Each light chain is comprised of a light chain variable region (abbreviated herein as VL) and a light chain constant region. The light chain constant region is comprised of one domain, CL. The VH and VL regions can be further subdivided into regions of hypervariability, termed complementarity determining regions (CDR), interspersed with regions that are more conserved, termed framework regions (FR). Each VH and VL is composed of three CDRs and four FR’s arranged from amino-terminus to carboxy-terminus in the following order: FR1 , CDR1 , FR2, CDR2, FR3, CDR3, and FR4. The variable regions of the heavy and light chains contain a binding domain that interacts with an antigen. The constant regions of the antibodies may mediate the binding of the immunoglobulin to host tissues or factors, including various cells of the immune system (e.g., effector cells) and the first component (Clq) of the classical complement system. The term “antibody” includes for example, monoclonal antibodies, human antibodies, humanized antibodies, camelised antibodies and chimeric antibodies. The antibodies can be of any isotype (e.g., IgG, IgE, IgM, IgD, IgA and IgY), class (e.g., lgG1 , lgG2, lgG3, lgG-4, lgA1 and lgA2) or subclass. Both the light and heavy chains are divided into regions of structural and functional homology.
The phrase “antibody fragment”, as used herein, refers to one or more portions of an antibody that retain the ability to specifically interact with (e.g., by binding, steric hindrance, stabilizing spatial distribution) an antigen. Examples of binding fragments include, but are not limited to, a Fab fragment, a monovalent fragment consisting of the VL, VH, CL and CH1 domains; a F(ab)2 fragment, a bivalent fragment comprising two Fab fragments linked by a disulfide bridge at the hinge region; a Fd fragment consisting of the VH and CH1 domains; a Fv fragment consisting of the VL and VH domains of a single arm of an antibody; a dAb fragment (Ward et al., (1989) Nature 341 :544-546), which consists of a VH domain; and an isolated complementarity determining region (CDR). Furthermore, although the two domains of the Fv fragment, VL and VH, are coded for by separate genes, they can be joined, using recombinant methods, by a synthetic linker that enables them to be made as a single protein chain in which
the VL and VH regions pair to form monovalent molecules (known as single chain Fv (scFv); see e.g., Bird et al., (1988) Science 242:423-426; and Huston et al., (1988) Proc. Natl. Acad. Sci. 85:5879-5883). Such single chain antibodies are also intended to be encompassed within the term “antibody fragment”. These antibody fragments are obtained using conventional techniques known to those of skill in the art, and the fragments are screened for utility in the same manner as are intact antibodies. Antibody fragments can also be incorporated into single domain antibodies, maxibodies, minibodies, intrabodies, diabodies, triabodies, tetrabodies, v- NAR and bis-scFv (see, e.g., Hollinger and Hudson, (2005) Nature Biotechnology 23:1126- 1136). Antibody fragments can be grafted into scaffolds based on polypeptides such as Fibronectin type III (Fn3) (see U.S. Pat. No. 6,703,199, which describes fibronectin polypeptide monobodies). Antibody fragments can be incorporated into molecules comprising a pair of tandem Fv segments (VH-CH1-VH-CH1) which, together with complementary light chain polypeptides, form a pair of antigen-binding sites (Zapata et al., (1995) Protein Eng. 8:1057- 1062; and U.S. Pat. No. 5,641 ,870).
The term "recombinant” polypeptide, as used herein, includes all polypeptides according to the present disclosure that are prepared, expressed, created or segregated by means not existing in nature. For example, polypeptides isolated from a host cell transformed to express the polypeptides, polypeptides selected and isolated from a recombinant, HTH polypeptide library, and polypeptides prepared, expressed, created or isolated by any other means. In an embodiment, the polypeptides disclosed herein are isolated from the HTH polypeptide library described in WO 2017/149117.
“Compositions” of the present disclosure may be used for therapeutic or prophylactic applications. The present disclosure, therefore, includes a pharmaceutical composition containing an antibody or antibody fragment as disclosed herein and a pharmaceutically acceptable carrier or excipient therefor. In a related aspect, the present disclosure provides a method for treating cancer. Such method contains the steps of administering to a subject in need thereof an effective amount of the pharmaceutical composition that contains an antibody or antibody fragment as described herein.
The present disclosure provides therapeutic methods comprising the administration of a therapeutically effective amount of an antibody or antibody fragment as disclosed herein to a subject in need of such treatment. A "therapeutically effective amount” or “effective amount”, as used herein, refers to the amount of a polypeptide according to the present disclosure necessary to elicit the desired biological response. In accordance with the subject disclosure, the therapeutic effective amount is the amount of a IL-13Ra2 specific polypeptide disclosed herein necessary to treat and/or prevent a disease.
"Administered" or “administration” includes but is not limited to delivery of a drug by an injectable form, such as, for example, an intravenous, intramuscular, intradermal or subcutaneous route or mucosal route, for example, as a nasal spray or aerosol for inhalation or as an ingestible solution, capsule or tablet. Preferably, the administration is by an injectable form.
As used herein, "treatment", "treat" or "treating" and the like refers to clinical intervention in an attempt to alter the natural course of a disease in the subject being treated, and can be performed either for prophylaxis or during the course of clinical pathology. Desirable effects of treatment include, but are not limited to, preventing occurrence or recurrence of disease, alleviation of symptoms, diminishment of any direct or indirect pathological consequences of the disease, preventing metastasis, decreasing the rate of disease progression, amelioration or palliation of the disease state, and remission or improved prognosis. In some embodiments, antibodies or antibody fragments according to the preset disclosure are used to delay development of a disease or to slow the progression of a disease.
‘Preventing’ or ‘prevention’ refers to a reduction in risk of acquiring or developing a disease (i.e. causing at least one of the clinical symptoms of the disease not to develop in a subject that may be exposed to a disease-causing agent, or predisposed to the disease in advance of disease onset). “Prevention” also refers to methods which aim to prevent the onset of a disease or its symptoms or which delay the onset of a disease or its symptoms.
“Subject” or “species” or as used in this context refers to any mammal, including rodents, such as mouse or rat, and primates, such as cynomolgus monkey (Macaca fascicularis), rhesus monkey (Macaca mulatta) or humans (Homo sapiens). Preferably, the subject is a primate, most preferably a human.
The terms "engineered" or “modified” as used herein includes manipulation of nucleic acids or polypeptides by synthetic means (e.g. by recombinant techniques, in vitro peptide synthesis, by enzymatic or chemical coupling of peptides or some combination of these techniques). Preferably, the polypeptides according to the present disclosure are engineered or modified to improve one or more properties, such as antigen binding, stability, half-life, effector function, immunogenicity, safety and the like.
"Variant" as used herein refers to a polypeptide that differs from a reference polypeptide by one or more modifications for example amino acid substitutions, insertions or deletions.
The term "amino acid mutation" as used herein is meant to encompass amino acid substitutions, deletions, insertions, and modifications. Any combination of substitution,
deletion, insertion, and modification can be made as long as the final construct possesses the desired characteristics. Amino acid sequence deletions and insertions include N-and/or C- terminal deletions and insertions of amino acids. Particular amino acid mutations are amino acid substitutions. Amino acid substitutions include replacement by non-naturally occurring amino acids or by naturally occurring amino acid derivatives of the twenty standard amino acids. Amino acid mutations can be generated using genetic or chemical methods well known in the art. Genetic methods may include site-directed mutagenesis, PCR, gene synthesis and the like. It is contemplated that methods of altering the side chain group of an amino acid residue by methods other than genetic engineering, such as chemical modification, may also be useful.
Embodiments
The present disclosure provides a polypeptide specific for IL-13 receptor alpha 2 (IL-13Ra2) comprising a helix-turn-helix scaffold structure of the formula Helix-1 - Li - Helix-2, wherein Li is a linker and wherein Helix-1 and Helix-2 comprise a first and second a-helical peptide, wherein each of said a-helical peptides comprises the amino acid sequence of
D - E - L - Vari -X1 - L - Var2 - Var3 - X2 - L - Var4 - X3 - L - Var5 - Var6 (Helix-1 disclosed as SEQ ID NO: 197; Helix-2 disclosed as SEQ ID NO: 204), wherein,
Vari is any natural occurring amino acid excluding G, P and C in Helix-1 and Helix-2,
Var2 is K, R, T or I in Helix 1 and I or K in Helix-2,
Var3 is R or Q in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
Var4 is Q or K in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
Var5 is F, Y or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
Var6 is K, R or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
X1 is Q in Helix-1 and A in Helix-2,
X2 is E in Helix-1 and K in Helix-2, and
X3 is K in Helix-1 and M or A in Helix-2
In an embodiment, X3 is M in Helix-2. In another embodiment, X3 is A in Helix-2. In an embodiment, X3 is K in Helix-1 and A in Helix-2. In an embodiment, X3 is K in Helix-1 and M in Helix-2
In an embodiment, Vari is any natural occurring amino acid excluding G, P C in Helix 1. In an embodiment, Vari is A, K, H or T in Helix-1. In an embodiment, Vari is K in Helix-1. In an embodiment, Vari is H in Helix-1. In an embodiment, Vari is T in Helix-1. In an embodiment, Vari is A in Helix-1.
In an embodiment, Vari is any natural occurring amino acid excluding G, P C in Helix 2. In an embodiment, Vari is A, R, E or M in Helix-2. In an embodiment, Vari is A in Helix-2. In an embodiment, Vari is R in Helix-2. In an embodiment, Vari is E in Helix-2. In an embodiment, Vari is M in Helix-2.
In an embodiment, Var2 is I, K, R or T in Helix-1. In an embodiment, Var2 is I in Helix-1. In an embodiment, Var2 is R in Helix-1. In an embodiment, Var2 is T in Helix-1. In an embodiment, Var2 is K in Helix 1. In an embodiment, Var2 is I or K in Helix-2. In an embodiment, Var2 is I in Helix-2. In an embodiment, Var2 is K in Helix-2.
In an embodiment, Var3 is R or Q in Helix-1. In an embodiment, Var3 is Q in Helix-1. In an embodiment, Var3 is R in Helix-1. In an embodiment, Var3 is any natural occurring amino acid excluding G, P C in Helix-2. In an embodiment, Var3 is A, R, E or Q in Helix-2. In an embodiment, Var3 is R in Helix-2. In an embodiment, Var3 is Q in Helix-2. In an embodiment, Var3 is E in Helix-2, In an embodiment, Var3 is A in Helix-2.
In an embodiment, Var4 is K or Q in Helix-1. In an embodiment, Var4 is K in Helix-1. In an embodiment, Var4 is Q in Helix-1. In an embodiment, Var4 is any natural occurring amino acid excluding G, P C in Helix-2. In an embodiment, Var4 is Var4 is A, H, D, I, or E in Helix-2. In an embodiment, Var4 is H in Helix- 2, In an embodiment, Var4 is D in Helix-2. In an embodiment, Var4 is I in Helix-2. In an embodiment, Var4 is E in Helix-2. In an embodiment, Var4 is A in Helix- 2.
In an embodiment, Var5 is F, Y or H in Helix-1. In an embodiment, Var5 is F in Helix-1. In an embodiment, Var5 is Y in Helix-1. In an embodiment, Var5 is H in Helix-1. In an embodiment, Var5 is any natural occurring amino acid excluding G, P C in Helix-2. In an embodiment, Var5 is A, E, I, T, L, K or D in Helix-2. In an embodiment, Var5 is E or I in Helix-2. In an embodiment,
Var5 is I in Helix-2. In an embodiment, Var5 is T in Helix-2. In an embodiment, Var5 is L in Helix-2. In an embodiment, Var5 is K in Helix-2. In an embodiment, Var5 is D in Helix-2. In an embodiment, Var5 is A in Helix-2.
In an embodiment, Var6 is K, R or H in Helix-1. In an embodiment, Var6 is K in Helix-1. In an embodiment, Var6 is R in Helix-1. In an embodiment, Var6 is H in Helix-1. In an embodiment. Var6 is any natural occurring amino acid excluding G, P C in Helix-2. In an embodiment, Var6 is A, H, Q or R in Helix-2. In an embodiment, Var6 is H in Helix-2. In an embodiment, Var6 is R in Helix-2. In an embodiment, Var6 is Q in Helix-2. In an embodiment, Var6 is A in Helix-2.
In a further embodiment, the first and second a-helical peptide comprised in the helix-turn- helix scaffold structure form an antiparallel coiled-coiled structure. In an embodiment, the first a-helical peptide is comprised in Helix-1 and the second a-helical peptide is comprised in Helix-2. In a further embodiment, Helix-1 and Helix-2 comprised in the helix-turn-helix scaffold structure form an antiparallel coiled-coiled structure.
In an embodiment, said first a-helical peptide comprises an amino sequence selected from the group consisting of: DELTQLKRELQKLYR (SEQ ID NO: 1), DELAQLKRELQKLYR (SEQ ID NO: 2), DELTQLKRELQKLYK (SEQ ID NO: 3), DELTQLKRELQKLFR (SEQ ID NO: 4), DELHQLRRELQKLYR (SEQ ID NO: 5), DELKQLRRELQKLYH (SEQ ID NO: 6), DELTQLRRELQKLHR (SEQ ID NO: 7), DELTQLTRELKKLYR (SEQ ID NO: 8), and DELKQLIQELKKLYR (SEQ ID NO: 9).
In an embodiment, said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQ (SEQ ID NO: 19), DELRALIRKLHALEQ (SEQ ID NO: 20), DELAALIRKLHMLEQ (SEQ ID NO: 21), DELRALIAKLHMLEQ (SEQ ID NO: 22), DELRALIRKLAMLEQ (SEQ ID NO: 23), DELRALIRKLHMLAQ (SEQ ID NO: 24),DELRALIRKLHMLEA (SEQ ID NO: 25), DELRALIRKLDMLIR (SEQ ID NO: 26), DELEALKRKLDMLLH (SEQ ID NO: 27), DELMALIEKLIMLTH (SEQ ID NO: 28), DELRALIRKLHMLEH (SEQ ID NO: 29), DELRALIRKLEMLKR (SEQ ID NO: 30), and DELRALIQKLDMLDR (SEQ ID NO: 31).
In an embodiment, said first a-helical peptide comprises an amino sequence selected from the group consisting of: DELTQLKRELQKLYR (SEQ ID NO: 1), DELAQLKRELQKLYR (SEQ ID NO: 2), DELTQLKRELQKLYK (SEQ ID NO: 3), DELTQLKRELQKLFR (SEQ ID NO: 4), DELHQLRRELQKLYR (SEQ ID NO: 5), DELKQLRRELQKLYH (SEQ ID NO: 6),
DELTQLRRELQKLHR (SEQ ID NO: 7), DELTQLTRELKKLYR (SEQ ID NO: 8),
DELKQLIQELKKLYR (SEQ ID NO: 9), and said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQ (SEQ ID NO: 19),
DELRALIRKLHALEQ (SEQ ID NO: 20), DELAALIRKLHMLEQ (SEQ ID NO: 21),
DELRALIAKLHMLEQ (SEQ ID NO: 22), DELRALIRKLAMLEQ (SEQ ID NO: 23),
DELRALIRKLHMLAQ (SEQ ID NO: 24), DELRALIRKLHMLEA (SEQ ID NO: 25),
DELRALIRKLDMLIR (SEQ ID NO: 26), DELEALKRKLDMLLH (SEQ ID NO: 27),
DELMALI EKLIM LTH (SEQ ID NO: 28), DELRALIRKLHMLEH (SEQ ID NO: 29),
DELRALIRKLEMLKR (SEQ ID NO: 30), and DELRALIQKLDMLDR (SEQ ID NO: 31).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQ (SEQ ID NO: 19),
DELRALIRKLHALEQ (SEQ ID NO: 20), DELAALIRKLHMLEQ (SEQ ID NO: 21),
DELRALIAKLHMLEQ (SEQ ID NO: 22), DELRALIRKLAMLEQ (SEQ ID NO: 23),
DELRALIRKLHMLAQ (SEQ ID NO: 24), DELRALIRKLHMLEA (SEQ ID NO: 25),
DELRALIRKLDMLIR (SEQ ID NO: 26), and DELEALKRKLDMLLH (SEQ ID NO: 27).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHALEQ (SEQ ID NO: 20).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELAALIRKLHMLEQ (SEQ ID NO: 21).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELRALIAKLHMLEQ (SEQ ID NO: 22).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELRALIRKLAMLEQ (SEQ ID NO: 23).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLAQ (SEQ ID NO: 24).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEA (SEQ ID NO: 25).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELRALIRKLDMLIR (SEQ ID NO: 26).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises the amino sequence of DELEALKRKLDMLLH (SEQ ID NO: 27).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELAQLKRELQKLYR (SEQ ID NO: 2) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYK (SEQ ID NO: 3) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLFR (SEQ ID NO: 4) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELHQLRRELQKLYR (SEQ ID NO: 5) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELKQLRRELQKLYH (SEQ ID NO: 6) and said second a-helical peptide comprises the amino sequence of DELMALIEKLIMLTH (SEQ ID NO: 28).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELTQLRRELQKLHR (SEQ ID NO: 7) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEH (SEQ ID NO: 29) or DELRALIRKLHMLEQ (SEQ ID NO: 19). In an embodiment, said first a-helical peptide comprises the amino sequence of DELTQLRRELQKLHR (SEQ ID NO: 7) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEH (SEQ ID NO: 29).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELTQLRRELQKLHR (SEQ ID NO: 7) and said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELTQLTRELKKLYR (SEQ ID NO: 8) and said second a-helical peptide comprises the amino sequence of DELRALIRKLEMLKR (SEQ ID NO: 30).
In an embodiment, said first a-helical peptide comprises the amino sequence of DELKQLIQELKKLYR (SEQ ID NO: 9) and said second a-helical peptide comprises the amino sequence of DELRALIQKLDMLDR (SEQ ID NO: 31).
In an embodiment, said first a-helical peptide comprises an amino sequence selected from the group consisting of: DELTQLKRELQKLYR (SEQ ID NO: 1), DELAQLKRELQKLYR (SEQ ID NO: 2), DELTQLKRELQKLYK (SEQ ID NO: 3), DELTQLKRELQKLFR (SEQ ID NO: 4), DELHQLRRELQKLYR (SEQ ID NO: 5), and DELTQLRRELQKLHR (SEQ ID NO: 7) and said second a-helical peptide comprises the amino sequence of: DELRALIRKLHMLEQ (SEQ ID NO: 19).
In an embodiment, said Helix-1 or said first a-helical peptide comprises an amino sequence selected from the group consisting of: DELTQLKRELQKLYR (SEQ ID NO: 1),
DELAQLKRELQKLYR (SEQ ID NO: 2), DELTQLKRELQKLYK (SEQ ID NO: 3),
DELTQLKRELQKLFR (SEQ ID NO: 4), DELHQLRRELQKLYR (SEQ ID NO: 5),
DELKQLRRELQKLYH (SEQ ID NO: 6), DELTQLRRELQKLHR (SEQ ID NO: 7),
DELTQLTRELKKLYR (SEQ ID NO: 8), and DELKQLIQELKKLYR (SEQ ID NO: 9).
In an embodiment, said Helix-2 or said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQ (SEQ ID NO: 19),
DELRALIRKLHALEQ (SEQ ID NO: 20), DELAALIRKLHMLEQ (SEQ ID NO: 21),
DELRALIAKLHMLEQ (SEQ ID NO: 22), DELRALIRKLAMLEQ (SEQ ID NO: 23),
DELRALIRKLHMLAQ (SEQ ID NO: 24),DELRALIRKLHMLEA (SEQ ID NO: 25), DELRALIRKLDMLIR (SEQ ID NO: 26), DELEALKRKLDMLLH (SEQ ID NO: 27),
DELMALIEKLIMLTH (SEQ ID NO: 28), DELRALIRKLHMLEH (SEQ ID NO: 29), DELRALIRKLEMLKR (SEQ ID NO: 30), and DELRALIQKLDMLDR (SEQ ID NO: 31).
In an embodiment, said Helix-1 or said first a-helical peptide comprises an amino sequence selected from the group consisting of: DELTQLKRELQKLYR (SEQ ID NO: 1),
DELAQLKRELQKLYR (SEQ ID NO: 2), DELTQLKRELQKLYK (SEQ ID NO: 3),
DELTQLKRELQKLFR (SEQ ID NO: 4), DELHQLRRELQKLYR (SEQ ID NO: 5),
DELKQLRRELQKLYH (SEQ ID NO: 6), DELTQLRRELQKLHR (SEQ ID NO: 7), DELTQLTRELKKLYR (SEQ ID NO: 8), DELKQLIQELKKLYR (SEQ ID NO: 9), and said Helix- 2 or said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQ (SEQ ID NO: 19), DELRALIRKLHALEQ (SEQ ID NO: 20), DELAALIRKLHMLEQ (SEQ ID NO: 21), DELRALIAKLHMLEQ (SEQ ID NO: 22), DELRALIRKLAMLEQ (SEQ ID NO: 23), DELRALIRKLHMLAQ (SEQ ID NO: 24),
DELRALIRKLHMLEA (SEQ ID NO: 25), DELRALIRKLDMLIR (SEQ ID NO: 26),
DELEALKRKLDMLLH (SEQ ID NO: 27), DELMALIEKLIMLTH (SEQ ID NO: 28),
DELRALIRKLHMLEH (SEQ ID NO: 29), DELRALIRKLEMLKR (SEQ ID NO: 30), and
DELRALIQKLDMLDR (SEQ ID NO: 31).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQ (SEQ ID NO: 19), DELRALIRKLHALEQ (SEQ ID NO: 20), DELAALIRKLHMLEQ (SEQ ID NO: 21), DELRALIAKLHMLEQ (SEQ ID NO: 22), DELRALIRKLAMLEQ (SEQ ID NO: 23),
DELRALIRKLHMLAQ (SEQ ID NO: 24), DELRALIRKLHMLEA (SEQ ID NO: 25),
DELRALIRKLDMLIR (SEQ ID NO: 26), and DELEALKRKLDMLLH (SEQ ID NO: 27).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHALEQ (SEQ ID NO: 20).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELAALIRKLHMLEQ (SEQ ID NO: 21).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIAKLHMLEQ (SEQ ID NO: 22).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLAMLEQ (SEQ ID NO: 23).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLAQ (SEQ ID NO: 24).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEA (SEQ ID NO: 25).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLDMLIR (SEQ ID NO: 26).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYR (SEQ ID NO: 1) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELEALKRKLDMLLH (SEQ ID NO: 27).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELAQLKRELQKLYR (SEQ ID NO: 2) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLYK (SEQ ID NO: 3) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLKRELQKLFR (SEQ ID NO: 4) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELHQLRRELQKLYR (SEQ ID NO: 5) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELKQLRRELQKLYH (SEQ ID NO: 6) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELMALIEKLIMLTH (SEQ ID NO: 28).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLRRELQKLHR (SEQ ID NO: 7) and said Helix-2 or said second a-helical peptide
comprises the amino sequence of DELRALIRKLHMLEH (SEQ ID NO: 29) or DELRALIRKLHMLEQ (SEQ ID NO: 19).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLRRELQKLHR (SEQ ID NO: 7) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEH (SEQ ID NO: 29).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLRRELQKLHR (SEQ ID NO: 7) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQ (SEQ ID NO: 19).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELTQLTRELKKLYR (SEQ ID NO: 8) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLEMLKR (SEQ ID NO: 30).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of DELKQLIQELKKLYR (SEQ ID NO: 9) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIQKLDMLDR (SEQ ID NO: 31).
In an embodiment, said Helix-1 or said first a-helical peptide comprises an amino sequence selected from the group consisting of: DELTQLKRELQKLYR (SEQ ID NO: 1),
DELAQLKRELQKLYR (SEQ ID NO: 2), DELTQLKRELQKLYK (SEQ ID NO: 3),
DELTQLKRELQKLFR (SEQ ID NO: 4), DELHQLRRELQKLYR (SEQ ID NO: 5), and DELTQLRRELQKLHR (SEQ ID NO: 7) and said Helix-2 or said second a-helical peptide comprises the amino sequence of: DELRALIRKLHMLEQ (SEQ ID NO: 19).
In an embodiment, the first and second a-helical peptide form an antiparallel coiled-coiled structure. In an embodiment, Helix-1 and Helix-2 form an antiparallel coiled-coiled structure.
In an embodiment, the linker Li has a length of 1 to 50 amino acid residues. In another embodiment, the linker Li has a length of 1-30 amino acid residues. Yet in another embodiment, the linker Li has a length of 1-10 amino acid residues. In one embodiment, the linker Li has a length of 5 amino acids. In yet a further aspect of the present disclosure the linker Li comprises 1 to 30 amino acid residues. In an embodiment, the linker Li comprises the amino acid sequence QGVDS (SEQ ID NO: 182). In one embodiment, the linker Li comprises the amino acid sequence QGVGS (SEQ ID NO: 183). In one embodiment, the linker Li consists of the amino acid sequence QGVDS (SEQ ID NO: 182). In one embodiment, the linker Li consists of the amino acid sequence or QGVGS (SEQ ID NO: 183).
Amino acid residues may also be replaced, deleted or added, for example to aid in the expression of library members in a preferred host species, to facilitate cloning of the molecule, to increase the stability of the polypeptide or to increase helix packing and the like.
For instance, G added to or being present at the N- and/or C-terminus of the Helix-turn-Helix scaffold structure may serve as a helix-breaker in situations where the Helix-turn-Helix scaffold structure is genetically fused to other moieties, such as affinity- or detection tags. However, it’s absence may be desired in situations where no moiety is fused to the HTH-scaffold structure. Indeed, mass spectrometry analysis of HTH polypeptides bearing a C-terminal accessible G revealed that this terminal amino acid is partly removed by proteases during mammalian production (data not shown). Thus, removal of this G may be desirable to increase the homogeneity of recombinantly produced product. In certain embodiments of the present disclosure, additional amino acid residues are added to the N- terminus and/or the C-terminus of the helix-turn-helix scaffold.
In an embodiment of the present disclosure, a G is added to the N-terminus of the helix-turn- helix scaffold structure. In an embodiment of the present disclosure, a G is added to the C- terminus of the helix-turn-helix scaffold structure. In an embodiment of the present disclosure, a G is added to the N-terminus of the helix-turn-helix scaffold and a G is added to the C- terminus of the helix-turn-helix scaffold. In an embodiment of the present disclosure, a G is present at the N-terminus of the helix-turn-helix scaffold structure. In an embodiment of the present disclosure, a G is present at the C-terminus of the helix-turn-helix scaffold structure. In an embodiment of the present disclosure, a G is present at the N-terminus of the helix-turn- helix scaffold and a G is present to the C-terminus of the helix-turn-helix scaffold. In an embodiment of the present disclosure, a G is added to the N-terminus to the first a-helical peptide. In an embodiment of the present disclosure, a G is added to the C-terminus of the second a-helical peptide. In an embodiment of the present disclosure, a G is added to the N- terminus of the first a-helical peptide and a G is added to the C-terminus of the second a- helical peptide. In an embodiment of the present disclosure, a G is present at the N-terminus of the first a-helical peptide. In an embodiment of the present disclosure, a G is present at the C-terminus of the second a-helical peptide. In an embodiment of the present disclosure, a G is present at the N-terminus of the first a-helical peptide and a G is present at the C-terminus of the second a-helical peptide.
Accordingly, in an embodiment, said Helix-1 or said first a-helical peptide comprises an amino sequence selected from the group consisting of: GDELTQLKRELQKLYR (SEQ ID NO: 10), GDELAQLKRELQKLYR (SEQ ID NO: 11), GDELTQLKRELQKLYK (SEQ ID NO: 12), GDELTQLKRELQKLFR (SEQ ID NO: 13), GDELHQLRRELQKLYR (SEQ ID NO: 14),
GDELKQLRRELQKLYH (SEQ ID NO: 15), GDELTQLRRELQKLHR (SEQ ID NO: 16). GDELTQLTRELKKLYR (SEQ ID NO: 17), and GDELKQLIQELKKLYR (SEQ ID NO: 18).
In an embodiment, said Helix-2 or said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQG (SEQ ID NO: 32),
DELRALIRKLHALEQG (SEQ ID NO: 33), DELAALIRKLHMLEQG (SEQ ID NO: 34),
DELRALIAKLHMLEQG (SEQ ID NO: 35), DELRALIRKLAMLEQG (SEQ ID NO: 36),
DELRALIRKLHMLAQG (SEQ ID NO: 37), DELRALIRKLHMLEAG (SEQ ID NO: 38),
DELRALIRKLDMLIRG (SEQ ID NO: 39), DELEALKRKLDMLLHG (SEQ ID NO: 40),
DELMALI EKLIM LTHG (SEQ ID NO: 41), DELRALIRKLHMLEHG (SEQ ID NO: 42),
DELRALIRKLEMLKRG (SEQ ID NO: 43), and DELRALIQKLDMLDRG (SEQ ID NO: 44).
In an embodiment, said Helix-1 or said first a-helical peptide comprises an amino sequence selected from the group consisting of: GDELTQLKRELQKLYR (SEQ ID NO: 10), GDELAQLKRELQKLYR (SEQ ID NO: 11), GDELTQLKRELQKLYK (SEQ ID NO: 12),
GDELTQLKRELQKLFR (SEQ ID NO: 13), GDELHQLRRELQKLYR (SEQ ID NO: 14),
GDELKQLRRELQKLYH (SEQ ID NO: 15), GDELTQLRRELQKLHR (SEQ ID NO: 16),
GDELTQLTRELKKLYR (SEQ ID NO: 17), and GDELKQLIQELKKLYR (SEQ ID NO: 18), and said Helix-2 or said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQG (SEQ ID NO: 32), DELRALIRKLHALEQG (SEQ ID NO: 33), DELAALIRKLHMLEQG (SEQ ID NO: 34), DELRALIAKLHMLEQG (SEQ ID NO: 35), DELRALIRKLAMLEQG (SEQ ID NO: 36), DELRALIRKLHMLAQG (SEQ ID NO: 37), DELRALIRKLHMLEAG (SEQ ID NO: 38), DELRALIRKLDMLIRG (SEQ ID NO: 39), DELEALKRKLDMLLHG (SEQ ID NO: 40), DELMALIEKLIMLTHG (SEQ ID NO: 41), DELRALIRKLHMLEHG (SEQ ID NO: 42), DELRALIRKLEMLKRG (SEQ ID NO: 43), and DELRALIQKLDMLDRG (SEQ ID NO: 44).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises an amino sequence selected from the group consisting of: DELRALIRKLHMLEQG (SEQ ID NO: 32), DELRALIRKLHALEQG (SEQ ID NO: 33), DELAALIRKLHMLEQG (SEQ ID NO: 34), DELRALIAKLHMLEQG (SEQ ID NO: 35), DELRALIRKLAMLEQG (SEQ ID NO: 36), DELRALIRKLHMLAQG (SEQ ID NO: 37), DELRALIRKLHMLEAG (SEQ ID NO: 38), DELRALIRKLDMLIRG (SEQ ID NO: 39), and DELEALKRKLDMLLHG (SEQ ID NO: 40).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQG (SEQ ID NO: 32).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHALEQG (SEQ ID NO: 33).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELAALIRKLHMLEQG (SEQ ID NO: 34).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIAKLHMLEQG (SEQ ID NO: 35).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLAMLEQG (SEQ ID NO: 36).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLAQG (SEQ ID NO: 37).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEAG (SEQ ID NO: 38).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLDMLIRG (SEQ ID NO: 39).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYR (SEQ ID NO: 10) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELEALKRKLDMLLHG (SEQ ID NO: 40).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELAQLKRELQKLYR (SEQ ID NO: 11 and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQG (SEQ ID NO: 32).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLYK (SEQ ID NO: 12) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQG (SEQ ID NO: 32).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLKRELQKLFR (SEQ ID NO: 13) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQG (SEQ ID NO: 32).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELHQLRRELQKLYR (SEQ ID NO: 14) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQG (SEQ ID NO: 32).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELKQLRRELQKLYH (SEQ ID NO: 15) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELMALIEKLIMLTHG (SEQ ID NO: 41).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLRRELQKLHR (SEQ ID NO: 16) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEHG (SEQ ID NO: 42) or DELRALIRKLHMLEQG (SEQ ID NO: 32).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLRRELQKLHR (SEQ ID NO: 16) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEHG (SEQ ID NO: 42).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLRRELQKLHR (SEQ ID NO: 16) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLHMLEQG (SEQ ID NO: 32).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELTQLTRELKKLYR (SEQ ID NO: 17) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIRKLEMLKRG (SEQ ID NO: 43).
In an embodiment, said Helix-1 or said first a-helical peptide comprises the amino sequence of GDELKQLIQELKKLYR (SEQ ID NO: 18) and said Helix-2 or said second a-helical peptide comprises the amino sequence of DELRALIQKLDMLDRG (SEQ ID NO: 44).
In an embodiment, said Helix-1 or said first a-helical peptide comprises an amino sequence selected from the group consisting of: GDELTQLKRELQKLYR (SEQ ID NO: 10), GDELAQLKRELQKLYR (SEQ ID NO: 11), GDELTQLKRELQKLYK (SEQ ID NO: 12), GDELTQLKRELQKLFR (SEQ ID NO: 13), GDELHQLRRELQKLYR (SEQ ID NO: 14), and GDELTQLRRELQKLHR (SEQ ID NO: 16), and wherein said Helix-2 or said second a-helical peptide comprises the amino sequence of: DELRALIRKLHMLEQG (SEQ ID NO: 32).
In an embodiment, the first and second a-helical peptide form an antiparallel coiled-coiled structure. In an embodiment, Helix-1 and Helix-2 form an antiparallel coiled-coiled structure.
In an embodiment, the linker Li has a length of 1 to 50 amino acids. In another embodiment, the linker Li has a length of 1-30 amino acids. Yet in another embodiment, the linker Li has a length of 1-10 amino acids. In one embodiment, the linker Li has a length of 5 amino acids. In yet a further aspect of the present disclosure the linker Li comprises 1 to 30 amino acid residues. In an embodiment, the linker Li comprises the amino acid sequence QGVDS (SEQ ID NO: 182). In one embodiment, the linker Li comprises the amino acid sequence QGVGS (SEQ ID NO: 183). In one embodiment, the linker Li consists of the amino acid sequence QGVDS (SEQ ID NO: 182). In one embodiment, the linker Li consists of the amino acid sequence or QGVGS (SEQ ID NO: 183).
Linker Li
In an embodiment of the present disclosure, Helix-1 and Helix-2 are formed by a single polypeptide (a single-chain polypeptide) wherein the two a-helical peptides are either directly linked via a single peptide bond to each other, or are linked by a linker segment that does not substantially interfere with the association of Helix-1 and Helix-2 into a coiled-coil structure.
Preferably, the linker Li comprises a non-helical region. The first and last residues of the nonhelical region can be any amino acid. Preferably, one or both residues are helix breaking or helix destabilizing residues, such as G or P.
Peptide linkers include for example, but are not limited to, G linkers, S linkers, mixed G/S linkers, G- and S-rich linkers, such as (GS)n (SEQ ID NO: 184), (G4S)n (SEQ ID NO: 185), (SG4)n (SEQ ID NO: 186), (GSGGS)n (SEQ ID NO: 187), (GGGS)n (SEQ ID NO: 188) or G4(SG4)n (SEQ ID NO: 189), wherein n is an integer between 1 and 10, typically between 2 and 4, which are known to the skilled artisan. In an embodiment, the linker Li has the amino acid sequence of GGGGSGGGGS (SEQ ID NO: 190).
In an embodiment, the two a-helical peptides are linked via a peptide linker Li which connects the C-terminus of Helix-1 to the N-terminus of Helix-2 thus resulting in a single-chain amino acid sequence for the HTH scaffold structure of the polypeptides according to the present disclosure. Thus, according to a specific embodiment of the present disclosure, the HTH scaffold structure of the polypeptides according to the present disclosure comprises the general formula Helix-1 - Li - Helix-2.
In an embodiment, Helix-1 , Li and Helix-2 are covalently linked in a way that the C-terminus of Helix-1 is linked to the N-terminus of Li and that the C-terminus of Li is linked to the N-terminus of Helix-2. In an embodiment, the arrangement from the N- to the C-terminus is as follows: Helix-1 , Li and Helix-2. In an embodiment of the present disclosure, the linker Li is absent.
It is understood that a peptide linker Li as used herein is not limited to only one of the aforementioned and exemplified peptide linkers but may comprise any combination of two or more such linker which are fused to each other.
In an embodiment of the present disclosure, the linker has a length of 1 to 50 amino acid residues. In another embodiment, the linker has a length of 1-30 amino acid residues. Yet in another embodiment, the linker has a length of 1-10 amino acid residues. In one embodiment, the linker has a length of 5 amino acids. In yet a further aspect of the present disclosure the linker Li comprises 1 to 30 amino acid residues.
In one embodiment, the linker Li comprises the amino acid sequence QGVDS (SEQ ID NO: 182). In one embodiment, the linker Li comprises the amino acid sequence QGVGS (SEQ ID NO: 183). In one embodiment, the linker Li consists of the amino acid sequence QGVDS (SEQ ID NO: 182). In one embodiment, the linker Li consists of the amino acid sequence or QGVGS (SEQ ID NO: 183).
In an embodiment, the present disclosure provides an isolated polypeptide specific for IL- 13Ra2 according to the present disclosure, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of: DELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 47), DELTQLKRELQKLYRQGVGSDELRALIRKLHMLEQ (SEQ ID NO: 48), DELTQLKRELQKLYRQGVDSDELRALIRKLHALEQ (SEQ ID NO: 49), DELAQLKRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 50), DELTQLKRELQKLYRQGVDSDELAALIRKLHMLEQ (SEQ ID NO: 51), DELTQLKRELQKLYRQGVDSDELRALIAKLHMLEQ (SEQ ID NO: 52), DELTQLKRELQKLYRQGVDSDELRALIRKLAMLEQ (SEQ ID NO: 53), DELTQLKRELQKLYRQGVDSDELRALIRKLHMLAQ (SEQ ID NO: 54), DELTQLKRELQKLYRQGVDSDELRALIRKLHMLEA (SEQ ID NO: 55), DELTQLKRELQKLYKQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 56), DELTQLKRELQKLFRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 57), DELTQLKRELQKLYRQGVDSDELRALIRKLDMLIR (SEQ ID NO: 58), DELTQLKRELQKLYRQGVDSDELEALKRKLDMLLH (SEQ ID NO: 59), DELHQLRRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 60), DELKQLRRELQKLYHQGVDSDELMALI EKLIM LTH (SEQ ID NO: 61),
DELTQLRRELQKLHRQGVDSDELRALIRKLHMLEH (SEQ ID NO: 62), DELTQLRRELQKLHRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 63), DELTQLTRELKKLYRQGVDSDELRALIRKLEMLKR (SEQ ID NO: 64), and DELKQLIQELKKLYRQGVDSDELRALIQKLDMLDR (SEQ ID NO: 65).
In a further embodiment, the present disclosure provides an isolated polypeptide specific for IL-13Ra2, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of:
GDELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 66),
GDELTQLKRELQKLYRQGVGSDELRALIRKLHMLEQG (SEQ ID NO: 67), GDELTQLKRELQKLYRQGVDSDELRALIRKLHALEQG (SEQ ID NO: 68), GDELAQLKRELQKLYRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 69), GDELTQLKRELQKLYRQGVDSDELAALIRKLHMLEQG (SEQ ID NO: 70), GDELTQLKRELQKLYRQGVDSDELRALIAKLHMLEQG (SEQ ID NO: 71), GDELTQLKRELQKLYRQGVDSDELRALIRKLAMLEQG (SEQ ID NO: 72), GDELTQLKRELQKLYRQGVDSDELRALIRKLHMLAQG (SEQ ID NO: 73), GDELTQLKRELQKLYRQGVDSDELRALIRKLHMLEAG (SEQ ID NO: 74), GDELTQLKRELQKLYKQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 75), GDELTQLKRELQKLFRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 76), GDELTQLKRELQKLYRQGVDSDELRALIRKLDMLIRG (SEQ ID NO: 77), GDELTQLKRELQKLYRQGVDSDELEALKRKLDMLLHG (SEQ ID NO: 78), GDELHQLRRELQKLYRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 79), GDELKQLRRELQKLYHQGVDSDELMALIEKLIMLTHG (SEQ ID NO: 80), GDELTQLRRELQKLHRQGVDSDELRALIRKLHMLEHG (SEQ ID NO: 81), GDELTQLRRELQKLHRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 82), GDELTQLTRELKKLYRQGVDSDELRALIRKLEMLKRG (SEQ ID NO: 83), and GDELKQLIQELKKLYRQGVDSDELRALIQKLDMLDRG (SEQ ID NO: 84).
In a further embodiment, the present disclosure provides an isolated polypeptide specific for IL-13Ra2, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of:
GDELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 85), GDELTQLKRELQKLYRQGVGSDELRALIRKLHMLEQ (SEQ ID NO: 86), GDELTQLKRELQKLYRQGVDSDELRALIRKLHALEQ (SEQ ID NO: 87), GDELAQLKRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 88), GDELTQLKRELQKLYRQGVDSDELAALIRKLHMLEQ (SEQ ID NO: 89), GDELTQLKRELQKLYRQGVDSDELRALIAKLHMLEQ (SEQ ID NO: 90),
GDELTQLKRELQKLYRQGVDSDELRALIRKLAMLEQ (SEQ ID NO: 91), GDELTQLKRELQKLYRQGVDSDELRALIRKLHMLAQ (SEQ ID NO: 92), GDELTQLKRELQKLYRQGVDSDELRALIRKLHMLEA (SEQ ID NO: 93), GDELTQLKRELQKLYKQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 94), GDELTQLKRELQKLFRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 95), GDELTQLKRELQKLYRQGVDSDELRALIRKLDMLIR (SEQ ID NO: 96), GDELTQLKRELQKLYRQGVDSDELEALKRKLDMLLH (SEQ ID NO: 97), GDELHQLRRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 98), GDELKQLRRELQKLYHQGVDSDELMALIEKLIMLTH (SEQ ID NO: 99), GDELTQLRRELQKLHRQGVDSDELRALIRKLHMLEH (SEQ ID NO: 100), GDELTQLRRELQKLHRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 101), GDELTQLTRELKKLYRQGVDSDELRALIRKLEMLKR (SEQ ID NO: 102), and GDELKQLIQELKKLYRQGVDSDELRALIQKLDMLDR (SEQ ID NO: 103).
In a further embodiment, the present disclosure provides an isolated polypeptide specific for IL-13Ra2, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of:
DELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 104), DELTQLKRELQKLYRQGVGSDELRALIRKLHMLEQG (SEQ ID NO: 105), DELTQLKRELQKLYRQGVDSDELRALIRKLHALEQG (SEQ ID NO: 106), DELAQLKRELQKLYRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 107), DELTQLKRELQKLYRQGVDSDELAALIRKLHMLEQG (SEQ ID NO: 108), DELTQLKRELQKLYRQGVDSDELRALIAKLHMLEQG (SEQ ID NO: 109), DELTQLKRELQKLYRQGVDSDELRALIRKLAMLEQG (SEQ ID NO: 110), DELTQLKRELQKLYRQGVDSDELRALIRKLHMLAQG (SEQ ID NO: 111), DELTQLKRELQKLYRQGVDSDELRALIRKLHMLEAG (SEQ ID NO: 112), DELTQLKRELQKLYKQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 113), DELTQLKRELQKLFRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 114), DELTQLKRELQKLYRQGVDSDELRALIRKLDMLIRG (SEQ ID NO: 115), DELTQLKRELQKLYRQGVDSDELEALKRKLDMLLHG (SEQ ID NO: 116), DELHQLRRELQKLYRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 117), DELKQLRRELQKLYHQGVDSDELMALI EKLIM LTHG (SEQ ID NO: 118), DELTQLRRELQKLHRQGVDSDELRALIRKLHMLEHG (SEQ ID NO: 119), DELTQLRRELQKLHRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 120), DELTQLTRELKKLYRQGVDSDELRALIRKLEMLKRG (SEQ ID NO: 121), and DELKQLIQELKKLYRQGVDSDELRALIQKLDMLDRG (SEQ ID NO: 122).
In an embodiment, the present disclosure provides an isolated polypeptide specific for IL- 13Ra2 according to the present disclosure, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of: DELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 47), GDELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 66), GDELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 85), DELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 104), DELTQLKRELQKLYRQGVDSDELRALIRKLHALEQ (SEQ ID NO: 49), GDELTQLKRELQKLYRQGVDSDELRALIRKLHALEQG (SEQ ID NO: 68), GDELTQLKRELQKLYRQGVDSDELRALIRKLHALEQ (SEQ ID NO: 87), and DELTQLKRELQKLYRQGVDSDELRALIRKLHALEQG (SEQ ID NO: 106).
In an embodiment, said polypeptide is cross-reactive to mouse and cynomolgus IL-13Ra2.
In an embodiment, said polypeptide does not bind to IL-13 receptor alpha 1 (IL-13Ra1), IL-4 receptor alpha (IL-4Ra) or to the IL-13Ra1/ IL-4Ra complex. In an embodiment, said polypeptide competes with human IL-13 for binding to IL-13Ra2.
In an embodiment, the polypeptide specific for IL-13Ra2 according to the present disclosure is a recombinant polypeptide. In an embodiment, the polypeptide specific for IL-13Ra2 according to the present disclosure is a synthetic polypeptide. In an embodiment, the polypeptide specific for IL-13Ra2 according to the present disclosure is an isolated polypeptide. In a further embodiment, the polypeptide specific for IL-13Ra2 according to the present disclosure is an isolated recombinant polypeptide.
In further embodiments, the HTH scaffold structure of the polypeptides of present disclosure may be further stabilized by the introduction of negatively charged amino acid residues at the N-terminus of each a-helical peptide. This may stabilize the dipole moment of the a-helices of said peptides. Such a negatively charged amino acid residue can be D or E. In another embodiment, such a negatively charged amino acid residue is D.
A polypeptide according to the present disclosure as may also be a functional variant of any of the specific polypeptides described herein. Such a variant polypeptide is a polypeptide that differs from a specific polypeptide described herein by one or more suitable amino acid modifications, such as substitutions, deletions, insertions, or terminal sequence additions, for instance in the invariant positions of the HTH scaffold structure, and/or the variable positions of the HTH scaffold structure in a single variant polypeptide.
A functional variant polypeptide as used in the context of an IL-13Ra2 specific polypeptide still allows the polypeptide to retain at least a substantial proportion (at least about 50%, 60%, 70%, 80%, 90%, 95% or more) of the affinity and/or the specificity of the parent polypeptide and in some cases such an IL-13Ra2 specific polypeptide may be associated with greater affinity and/or specificity than the parent polypeptide.
Such functional variants typically retain significant sequence identity to the parent polypeptide. The percent identity between two sequences is a function of the number of identical positions shared by the sequences (i.e., % homology = # of identical positions/total # of positions x 100), taking into account the number of gaps, and the length of each gap, which need to be introduced for optimal alignment of the two sequences.
The sequence of variable position variants may differ from the sequence of the variable position of the parent polypeptide sequences through mostly conservative amino acid substitutions; for instance at least about 35%, about 50% or more, about 60% or more, about 70% or more, about 75% or more, about 80% or more, about 85% or more, about 90% or more, about 95% or more of the substitutions in the variant are conservative amino acid residue replacements.
In the context of the present disclosure, conservative amino acid substitutions may be defined by substitutions within the classes of amino acids reflected in one or more of the following three tables:
Alternative conservative amino acid substitution classes:
Alternative physical and functional classification of amino acids:
Other conservative substitutions groupings include: V-L-l, F-Y, K-R, A-V, and N-Q.
Stabilized polypeptides of the disclosure
The polypeptides of the present disclosure can be produced (e.g. recombinantly or organic synthesis) with additional amino acid residues added at their N- and the C-terminus in order to allow linkage of the N- and C terminal end of the polypeptides. The thus formed cyclized polypeptides may further increase the stability of the a-helical structure and improve resistance against proteolytic degradation by proteases.
The polypeptides of the present disclosure can be cyclized, introducing e.g. a disulfide bridge or a reduction insensitive thioether linkage. A disulfide bonde can be formed under oxidizing conditions between N- and C-terminally introduced cysteine residues. A thioether bonde according to the present disclosure may be formed for instance between N-chloroacetyl glycine present at the N-terminus and a C residue present at the C-terminus.
In an embodiment of the present disclosure, the polypeptides are cyclic polypeptides. In an embodiment of the present disclosure, the cyclic polypeptides are formed by a covalent bond. In certain embodiments of the present disclosure, the covalent bond is a disulfide bond. In certain embodiments of the present disclosure, the disulfide bond is formed by two C residues. In certain embodiments of the present disclosure, the disulfide bond is formed between a C residue present at the N-terminus and a C residue present at the C-terminus of the polypeptides of the present disclosure.
In certain embodiments of the present disclosure, the covalent bond is a thioether bond. In certain embodiments of the present disclosure, the covalent bond is a thioether bond formed
between N-chloroacetyl glycine and a C residue. In certain embodiments, the covalent bond is a thioether bond formed between N-chloroacetyl glycine present at the N-terminus and a C residues present at the C-terminus of the polypeptides of the present disclosure. In certain embodiments, the covalent bond is a thioether bond formed between N-chloroacetyl glycine present at the N-terminus and a C residues present at the C-terminus of the polypeptides of the present disclosure.
Production
The polypeptides of the present disclosure can be synthesized by a variety of means, for example, by recombinant DNA technology or by organic chemical synthesis. Methods of peptide synthesis are known in the art. Alternatively, the coding sequences for the polypeptides can be recombinant DNA molecules, which are introduced into expression vectors or phage by operatively linking the DNA to the necessary expression control regions (e.g. regulatory regions) required for gene expression. The vectors can be introduced into the appropriate host cells such as prokaryotic (e.g., bacterial) or eukaryotic (e.g., yeast or mammalian) cells by methods well known in the art (see, e.g., "Current Protocol in Molecular Biology", Ausubel et al. (eds.), Greene Publishing Assoc, and John Wiley Interscience, New York, 1989 and 1992). Numerous cloning vectors are known to those of skill in the art, and the selection of an appropriate cloning vector is a matter of choice. The gene can be placed under the control of a promoter, ribosome binding site (for bacterial expression) and, optionally, an operator (collectively referred to herein as "control" elements), so that the DNA sequence encoding the desired protein is transcribed into RNA in the host cell transformed by a vector containing this expression construction. The coding sequence may or may not contain a signal peptide or leader sequence. Depending on the expression system and host selected, the proteins of the present disclosure are produced by growing host cells transformed by an expression vector described above under conditions whereby the protein of interest is expressed. The polypeptide is then isolated from the host cells and purified. If the expression system secretes the protein into growth media, the protein can be purified directly from the media. If the protein is not secreted, it is isolated from cell lysates or recovered from the cell membrane fraction. The selection of the appropriate growth conditions and recovery methods are within the skill of the art. The polypeptides of the present disclosure can then be purified by a number of techniques as known to the person skilled in the art. It should be noted that the polypeptides of the present disclosure are not naturally occurring proteins. Typically, the polypeptides of the present disclosure are recombinant, synthetic or semi-synthetic amino acid sequences, polypeptides or proteins.
Nucleic acids
In an embodiment, the present disclosure refers to a nucleic acid composition comprising a nucleic acid sequence or a plurality of nucleic acid sequences encoding an isolated polypeptide specific for IL-13Ra2 according to the present disclosure or a fusion molecule comprising said isolated polypeptide and at least one additional moiety.
In an embodiment, the present disclosure refers to a nucleic acid composition comprising a nucleic acid sequence or a plurality of nucleic acid sequences encoding an isolated polypeptide specific for IL-13-Ra2 according to the present disclosure or a fusion molecule comprising said isolated polypeptide and at least one additional moiety, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 47 - 122.
In an embodiment, the present disclosure refers to a nucleic acid composition comprising a nucleic acid sequence or a plurality of nucleic acid sequences encoding Helix-1 of an isolated polypeptide specific for IL-13-Ra2 or a fusion molecule comprising said Helix-1 and at least one additional moiety, wherein said Helix-1 comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 1 - 18.
In an embodiment, the present disclosure refers to a nucleic acid composition comprising a nucleic acid sequence or a plurality of nucleic acid sequences encoding Helix-2 of an isolated polypeptide specific for IL-13Ra2 or a fusion molecule comprising said Helix-2 and at least one additional moiety, wherein said Helix-2 comprises an amino acid sequence selected from the group consisting of: SEQ ID NO 19 - 44.
In an embodiment, the present disclosure refers to a nucleic acid composition comprising a nucleic acid sequence or a plurality of nucleic acid sequences encoding Helix-1 and Helix-2 of an isolated polypeptide specific for IL- 13Ra2 or a fusion molecule comprising said Helix-1 and Helix-2 and at least one additional moiety, wherein said Helix-1 comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 1 - 18, and wherein said Helix-2 comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 19 - 44.
Vectors
In an embodiment, the present disclosure refers to a vector composition comprising a vector or plurality of vectors comprising a nucleic acid composition according to the present disclosure encoding an isolated polypeptide specific for IL-13Ra2 according to the present disclosure or a fusion molecule comprising said isolated polypeptide and at least one additional moiety.
In an embodiment, the present disclosure refers to a vector composition comprising a vector or plurality of vectors comprising a nucleic acid composition according to the present disclosure encoding an isolated polypeptide specific for IL-13Ra2 according to the present disclosure or a fusion molecule comprising said isolated polypeptide and at least one additional moiety. In an embodiment, said polypeptide comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 47 - 122.
In an embodiment, the present disclosure refers to a vector composition comprising a vector or plurality of vectors comprising a nucleic acid composition according to the present disclosure encoding Helix-1 of an isolated polypeptide specific for IL-13-Ra2 according to the present disclosure or a fusion molecule comprising said Helix-2 of an isolated polypeptide and at least one additional moiety, wherein said Helix-1 comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 1 - 18:
In an embodiment, the present disclosure refers to a vector composition comprising a vector or plurality of vectors comprising a nucleic acid composition according to the present disclosure encoding Helix-2 of an isolated polypeptide specific for IL-13-Ra2 according to the present disclosure or a fusion molecule comprising said Helix-2 of an isolated polypeptide and at least one additional moiety, wherein said Helix-2 comprises an amino acid sequence selected from the group consisting of: SEQ ID NO 19 - 44.
In an embodiment, the present disclosure refers to a vector composition comprising a vector or plurality of vectors comprising a nucleic acid composition according to the present disclosure encoding Helix-1 and Helix-2 of an isolated polypeptide specific for IL-13-Ra2 according to the present disclosure or a fusion molecule comprising said Helix-1 and Helix-2 of an isolated polypeptide and at least one additional moiety, wherein said Helix-1 comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 1 - 18 and wherein said Helix-2 comprises an amino acid sequence selected from the group consisting of SEQ ID NO 19 - 44.
Host cell
In an embodiment, the present disclosure refers to a host cell comprising a vector composition according to the present disclosure encoding an isolated polypeptide specific for IL-13Ra2 according to the present disclosure or a fusion molecule comprising said isolated polypeptide and at least one additional moiety. In an embodiment, said polypeptide comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 47 - 122.
In an embodiment, the present disclosure refers to host cell comprising a vector composition according to the present disclosure encoding Helix-1 of an isolated polypeptide specific for IL-
13-Ra2 according to the present disclosure or a fusion molecule comprising said Helix-1 of an isolated polypeptide and at least one additional moiety, wherein said Helix-1 comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 1 - 18.
In an embodiment, the present disclosure refers to host cell comprising a vector composition according to the present disclosure encoding Helix-2 of an isolated polypeptide specific for IL- 13-Ra2 according to the present disclosure or a fusion molecule comprising said Helix-2 of an isolated polypeptide and at least one additional moiety, wherein said Helix-2 comprises an amino acid sequence selected from the group consisting of: SEQ ID NO 19 - 44.
In an embodiment, the present disclosure refers to a host cell comprising a vector composition according to the present disclosure encoding Helix-1 and Helix-2 of an isolated polypeptide specific for IL-13-Ra2 according to the present disclosure or a fusion molecule comprising said Helix- 1 and Helix-2 of an isolated polypeptide and at least one additional moiety, wherein said Helix-1 comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 1 - 18 and wherein said Helix-2 comprises an amino acid sequence selected from the group consisting of: SEQ ID NO 19 - 44.
In an embodiment, said host cell is able to express the polypeptide according to the present disclosure encoded by the vector composition. In a further embodiment, said host cell is an isolated cell. In a further embodiment, said isolated host cell is a mammalian cell. In an embodiment, said mammalian cell is a human cell. In another embodiment, said mammalian cell is a CHO cell.
Specificity
In an embodiment, the present disclosure pertains to an isolated polypeptide specific for IL- 13Ra2 disclosed in SEQ ID NO: 47 - 122.
In an embodiment, the isolated polypeptide according to the present disclosure is specific for IL-13Ra2. In an embodiment, the isolated polypeptide according to the present disclosure is specific for human IL-13Ra2 encoded by the amino acid sequence of SEQ ID NO: 123. In an embodiment, the isolated polypeptide according to the present disclosure is specific for a polypeptide comprising the amino acid sequence of SEQ ID NO: 123. In an embodiment, the polypeptide according to the present disclosure specifically binds to the extracellular region human IL-13Ra2.
In certain embodiment, the isolated polypeptide according to the present disclosure does not bind to IL-13Ra1. In an embodiment, the isolated polypeptide according to the present
disclosure does not bind to human and/or mouse IL-13Ra1. In certain embodiment, the isolated polypeptide according to the present disclosure is not cross-reactive to IL-13Ra1. In an embodiment, the isolated polypeptide according to the present disclosure does not bind to human and/or mouse IL-13Ra1.
In certain embodiment, the isolated polypeptide according to the present disclosure does not bind to IL-4Ra. In certain embodiment, the isolated polypeptide according to the present disclosure is not cross- reactive to IL-4Ra. In an embodiment, the isolated polypeptide according to the present disclosure does not bind to human and/or mouse IL-4Ra. In certain embodiment, the isolated polypeptide according to the present disclosure does not bind to IL- 13Ra1/IL-4Ra complex. In an embodiment, the isolated polypeptide according to the present disclosure does not bind to the human and/or mouse IL-13Ra1/IL-4Ra complex.
In an embodiment, said IL-13Ra1 , IL-4Ra or IL-13Ra1/IL-4Ra complex is expressed on cells.
In further embodiments, the present disclosure refers to an isolated polypeptide specific for IL- 13Ra2 according to the present disclosure, wherein said polypeptide binds to human IL-13Ra2 with an ECso concentration of 60 nM or less, 50 nM or less, 40 nM or less, 30 nM or less, 20 nM or less, 15 nM or less, 10 nM or less, 5 nM or less, 4 nM or less, 3 nM or less, 2 nM or less, 1 nM or less, 0.5 nM or less, 0.4 nM or less, 0.3 nM or less, 0.2 nM or less or 0.1 nM or less.
In an embodiment, the present disclosure pertains to an isolated polypeptide specific for IL- 13Ra2, wherein said polypeptide binds to cynomolgus IL-13Ra2 with an ECso concentration of 60 nM or less, 50 nM or less, 40 nM or less, 30 nM or less, 20 nM or less, 15 nM or less, 10 nM or less, 5 nM or less, 4 nM or less, 3 nM or less, 2 nM or less, 1 nM or less, 0.5 nM or less, 0.4 nM or less, 0.3 nM or less, 0.2 nM or less or 0.1 nM or less.
In embodiments, the present disclosure refers to an isolated polypeptide specific for IL-13Ra2, wherein said polypeptide binds to mouse IL-13Ra2 with an ECso concentration of 60 nM or less, 50 nM or less, 40 nM or less, 30 nM or less, 20 nM or less, 15 nM or less, 10 nM or less, 5 nM or less, 4 nM or less, 3 nM or less, 2 nM or less, 1 nM or less, 0.5 nM or less, 0.4 nM or less, 0.3 nM or less, 0.2 nM or less or 0.1 nM or less.
In an embodiment, the present disclosure refers to an isolated polypeptide specific for IL- 13Ra2, wherein said polypeptide binds to human, cynomolgus and mouse IL-13Ra2 with an ECso concentration of 10 nM or less. In an embodiment, said human IL-13Ra2 comprises the amino acid sequence of SEQ ID NO: 123. In an embodiment, said mouse IL-13Ra2 comprises the amino acid sequence of SEQ ID NO:125. In an embodiment, said cynomolgus IL-13Ra2
comprises the amino acid sequence of SEQ ID NO: 124. In embodiments, said human, cynomolgus or mouse IL-13Ra2 is expressed on cells. In embodiments, said human, cynomolgus or mouse IL-13Ra2 is expressed on engineered CHO cells expressing full-length human, cynomolgus, or mouse IL-13Ra2. In embodiments, said CHO cells are Flp-ln CHO cells. In certain embodiments, said human IL-13Ra2 is expressed on cancer cells. In certain embodiments, said human IL-13Ra2 is expressed on A-375 cells.
In certain embodiments, said EC50 concentration is determined as described herein in Example 3. In certain embodiments, said ECso concentration is determined by FACS described herein in Example 3. In further embodiments, the polypeptide specific for IL-13Ra2 according to the present invention does substantially not bind to IL-13Ra1 , IL-4Ra or to the IL-13Ra1/IL-4Ra complex. In an embodiment, said polypeptide according to the present disclosure does substantially not bind to IL-13Ra1 , IL-4Ra or to the IL-13Ra1/IL-4Ra complex at a polypeptide concentration of 1 pM or less. In an embodiment, said binding is determined as described in Example 3. In an embodiment, said binding is determined in ELISA. In an embodiment, said binding is determined by FACS. In an embodiment, said IL-13Ra1, IL-4Ra or IL-13Ra1/IL-4Ra complex is expressed on cells. In an embodiment, said IL-13Ra1 , IL-4Ra or IL-13Ra1/IL-4Ra complex is expressed on cancer cells. In an embodiment, said IL-13Ra1 , IL-4Ra or IL- 13Ra1/IL-4Ra complex is expressed on A- 549 cells.
Species cross-reactivity
In an embodiment, the isolated polypeptide according to the present disclosure is cross- reactive to cynomolgus monkey (cynomolgus) IL-13Ra2. In certain embodiment, the isolated polypeptide according to the present disclosure is cross-reactive to mouse IL-13Ra2. In certain embodiments, the isolated polypeptide according to the present disclosure is cross- reactive to cynomolgus and mouse IL-13Ra2. In certain embodiments, the isolated polypeptide according to the present disclosure is specific for human, cynomolgus and mouse IL-13Ra2. In certain embodiment, the present disclosure refers to an isolated polypeptide specific for human IL- 13Ra2, wherein said polypeptide cross-reactively binds to cynomolgus and mouse IL-13Ra2. In an embodiment, the isolated polypeptide according to the present disclosure specifically binds to the extracellular region of human, cynomolgus and mouse IL-13Ra2.
Internalization
In an embodiment, IL-13Ra2 is internalized from the cell surface of cells, such as cancer cells, expressing IL-13Ra2, upon binding of the polypeptide according to the present invention to
said cells. In an embodiment, the polypeptide according to the present invention is internalized from the surface of a cell, such as a cancer cell, expressing IL-13Ra2, upon binding to the cells.
IL-13 competition
In certain embodiments, the polypeptide of the present disclosure binds to the binding region of IL-13 on IL-13Ra2. Thus, in embodiments, the polypeptide of the present disclosure may compete with IL-13 for binding to IL-13Ra2. In certain embodiments, the disclosed polypeptides may interfere with IL-13 mediated signaling through IL-13Ra2.
In certain embodiments, the polypeptide of the present disclosure does not compete with IL- 13 for binding to IL-13Ra1. In certain embodiments, the polypeptide of the present disclosure does not compete with IL-13 for binding to IL-4Ra. In certain embodiments, the polypeptide of the present disclosure does not compete with I L-13 for binding to the IL13Ra1/IL-4Ra complex. In certain embodiments, the polypeptide according to the present disclosure does not interfere with IL-13 mediated signaling through IL-13Ra1 or the IL13Ra1/IL-4Ra complex. In an embodiment, said IL-13 is human IL-13.
Fusion molecules
The HTH polypeptides provided by the present disclosure may be fused, coupled or conjugated to one or more other moieties to generate fusion molecules in accordance with any of a variety of techniques, such as those employed in the production of immunoconjugates, which are known to the person skilled in the art. Such fusion molecules are generally achieved by covalent bonds between the HTH polypeptides of the present disclosure and the one or more moieties.
The fusion molecules are targeted only to cells expressing a target antigen for a HTH polypeptide according to the present invention. These targets may internalize in response to polypeptide binding. In certain embodiments, said target is IL-13Ra2. Accordingly, the present disclosure provides fusion molecules comprising an isolated polypeptide according to the present disclosure and at least one additional moiety. In an embodiment, said additional moiety is a selected from the group consisting of a therapeutic agent, a cytotoxic agent, a bacterial toxin (such as Pseudomonas exotoxin A and Diphtheria toxin, or plant toxins, such as ricin), a cytokine, an antibody or antibody fragment, a peptide, a polypeptide, a reporter enzyme, a detectable group, a moiety being capable of binding a metal ion, a tag suitable for detection and/or purification, a targeting ligands, a homo- or hetero-association domain, a moiety which
increases solubility of a protein, or a moiety which comprises an enzymatic cleavage site, a lipid, a liposome and virus-like-particles
A "therapeutic agent" as used herein may be any therapeutic agent including, but not limited to, genetic materials or agents, radionuclides, chemotherapeutic agents, and cytotoxic agents (See, e.g., U.S. Patent No. 6,949,245 to Sliwkowski), and antimicrobial peptides.
"Radionuclide" as described herein includes, but is not limited to 88Ga 225Ac, 210At, Ba, 77Br, 109Cd, 51Cr, 67Cu, 165Dy, 155Eu, 153Gd, 198Au, 166Ho, 113raln, 115111ln, 123l, 125l, 131l, 189lr, 191lr, 192lr, 194lr, 52Fe, 55Fe, 59Fe, 177Lu, 109Pd, 32P, 226Ra, 186Re, 188Re, 153Sm, 46Sc, 47Sc, 72Se, 75Se, 105Ag, 89Sr, 35S, 177Ta, 117mSn, 121Sn, 166Yb, 169Yb, 90Y, 212Bi, 213Bi 119Sb, 197Hg, 97Ru, 100Pd, 101mRh, and 212Pb.
"Chemotherapeutic agent" as used herein includes, but is not limited to, methotrexate, daunomycin, mitomycin C, cisplatin, vincristine, epirubicin, fluorouracil, verapamil, cyclophosphamide, cytosine arabinoside, aminopterin, bleomycin, mitomycin C, democolcine, etoposide, mithramycin, chlorambucil, melphalan, daunorubicin, doxorubicin, tamosifen, paclitaxel, vincristin, vinblastine, camptothecin, actinomycin D, and cytarabine.
"Cytotoxic agent" as used herein includes, but is not limited to, maytansinoids and maytansinoid analogs (such as DM1 , DM4), taxoids, CC- 1065 and CC- 1065 analogs, dolastatin and dolastatin analogs, ricin (or more particularly the ricin A chain), aclacinomycin, Diphtheria toxin, Monensin, Verrucarin A, Abrin, Tricothecenes, and Pseudomonas exotoxin A, taxol, cytochalasin B, gramicidin D, ethidium bromide, emetine, mitomycin, etoposide, tenoposide, anti-mitotic agents, such as the vinca alkaloids (e.g., vincristine and vinblastine), colchicin, anthracyclines, such as doxorubicin and daunorubicin, dihydroxy anthracin dione, mitoxantrone, mithramycin, actinomycin D, 1 -dehydrotestosterone, glucocorticoids, procaine, tetracaine, lidocaine, propranolol, and puromycin and analogs or homologs thereof, antimetabolites (e.g., methotrexate, 6-mercaptopurine, 6-thioguanine, cytarabine, and 5- fluorouracil decarbazine), alkylating agents (e.g., mechlorethamine, thioepa chlorambucil, melphalan, carmustine (BSNU), lomustine (CCNU), cyclothosphamide, busulfan, dibromomannitol, streptozotocin, mitomycin C, and cis-dichlorodiamine platinum (II) (DDP)), and antibiotics, including, but not limited to, dactinomycin (formerly actinomycin), bleomycin, mithramycin, calicheamicin, and anthramycin (AMO)) or Monomethyl auristatin E (MMAE) and Monomethyl auristatin F (MMAF)
"Detectable group" as used herein includes, but is not limited to, radiolabels, enzyme labels (e.g., horseradish peroxidase, alkaline phosphatase), gold beads, chemiluminescence labels, ligands (e.g., biotin, digoxin) and/or fluorescence labels (e.g., rhodamine, phycoerythrin,
fluorescein, fluorescent proteins), a fluorescent protein including, but not limited to, a green fluorescent protein or one of its many modified forms, a nucleic acid segment in accordance with known techniques, and energy absorbing and energy emitting agents. In certain embodiments, the polypeptide according to the present disclosure is fused to one or more Monomethyl auristatin E (MMAE) and/or Monomethyl auristatin F (MMAF) molecules (e.g., about 1 to about 10 MMAE or MMAF molecules per targeting polypeptide). In certain embodiments of the present disclosure, the HTH polypeptide conjugated to one or more maytansine molecules (such as DM-1 or DM-4) (e.g., about 1 to about 10 maytansine molecules per targeting polypeptide).
Enzymatically active toxins and fragments thereof which can be used are described above and include diphtheria A chain, nonbinding active fragments of diphtheria toxin, exotoxin A chain (from Pseiidomonas aeruginosa), ricin A chain, abrin A chain (from Corrybacterhim typhimuriae), modeccin A chain, alpha-sarcin, Aleurites fordii proteins, dianthin proteins, Phytolaca americana proteins (PAPI, PAPII, and PAP-S), momordica charantia inhibitor, curcin, crotin, sapaonaria officinalis inhibitor, gelonin, mitogellin, restrictocin, phenomycin, enomycin and the tricothecenes (see for example WO 93/21232)
A variety of radioactive isotopes or radionuclides are available for the production of radioconjugated fusion molecules as described above. In certain embodiments, the polypeptide specific for IL13-Ra2 according to the present disclosure is fused to one or more radionuclides.
In some embodiments, the fusion molecules according to the present disclosure may be made using a variety of bi-functional protein coupling agents such as N-succinimidyl-3-(2- pyridyldithiol)propionate (SPDP), succinimidyl-4-(N- maleimidomethyl) cyclohexane-l- carboxylate, iminothiolane (IT), bifunctional derivatives of imidoesters (such as dimethyl adipimidate HCL), active esters (such as disuccinimidyl suberate), aldehydes (such as glutareldehyde), bis-azido compounds (such as bis(p- azidobenzoyl)hexanediamine), bis- diazonium derivatives (such as bis-(p-diazoniumbenzoyl)- ethylenediamine), diisocyanates (such as tolyene 2,6-diisocyanate), and bis-active fluorine compounds (such as l,5-difluoro- 2,4-dinitrobenzene). Carbon- 14-labeled 1- isothiocyanatobenzyl-3-methyldiethylene triaminepentaacetic acid (MX-DTPA) is an exemplary chelating agent for conjugation of radionucleotide to the targeting peptide (see WO 94/11026). 1,4,7,10-Tetraazacyclododecane- 1 ,4,7,10-tetraacetic acid (DOTA (also known as tetraxetan)) is a further exemplary chelating agent for conjugation of radionucleotide to the polypeptides according to the present disclosure)
Accordingly, the present disclosure provides a fusion molecules comprising an isolated polypeptide specific for IL-13Ra2 according to the present disclosure and at least one or more radionuclides. In an embodiment, said radionuclide is contained within 1 ,4,7,10- tetraaza-
1.4.7.10-tetra(2-carbamoylmethyl)cyclododecane (TCMC) or 1 ,4,7,10- tetraazacyclododecane-
1.4.7.10-tetraacetic acid (DOTA). In an embodiment, said radionuclide is 177Lu. In an embodiment, said radionuclide is 255Ac. In an embodiment, said radionuclide is 213Bi.
In an embodiment, said polypeptide specific for IL-13Ra2 according to the present disclosure comprises an amino acid sequence selected from the group of SEQ ID NOs: 47 - 122.
In an embodiment, said polypeptide specific for IL-13Ra2 according to the present disclosure comprises an amino acid sequence selected from the group of: DELTQLKRELQKLYRQGVDSDELRALIRKLHALEQ (SEQ ID NO: 49), GDELTQLKRELQKLYRQGVDSDELRALIRKLHALEQG (SEQ ID NO: 68) GDELTQLKRELQKLYRQGVDSDELRALIRKLHALEQ (SEQ ID NO: 87), DELTQLKRELQKLYRQGVDSDELRALIRKLHALEQG (SEQ ID NO: 106), and GDELTQLKRELQKLYRQGVDSDELRALIRKLHALEQGGGGGSGGGGSGDELTQLKRELQK LYRQGVDSDELRALIRKLHALEQG (SEQ ID NO: 168)
Tethers
A fusion molecule according to the present disclosure may be made such that its individual components (e.g. toxin or antibody) are fused directly to each other or indirectly through a tether.
In certain embodiments, the components of a fusion molecule according to the present disclosure are genetically fused to each other. Such fusion can be achieved by a number of strategies, which include, but are not limited to polypeptide fusion between the N- and C- terminus of two polypeptides, fusion via disulfide bonds, and fusion via chemical cross-linking reagents.
Some suitable tethers include for example, but are not limited to peptide or polypeptide tethers such as glycine tethers, serine tethers, mixed glycine/serine tethers, glycine- and serine-rich tethers, tethers composed of largely polar polypeptide fragments or tethers comprising an amino acid sequence forming a random coil conformation.
A peptide or polypeptide tether may be any suitable amino acid sequence having a length between 1 and 500 amino acid residues, such as between 1 and 100, between 1 and 50, between 1 and 10, or between 1 and 5 amino acid residues.
The composition and length of a tether may be determined in accordance with methods well known in the art and may be tested for efficacy. Preferably, the tether is non-immunogenic. In an embodiment, the tether is a peptide tether. In an embodiment, the tether is a peptide tether comprising one or more amino acid residues, joined by peptide bonds that are known in the art. The peptide tether should have a length that is adequate to fuse two polypeptides (or components) in such a way that they assume the correct conformation relative to one another so that they retain or obtain the desired activity.
In an embodiment, a peptide or polypeptide tether according to the present disclosure is from 1 to 70 amino residues in length, 1 to 50 amino acid residues in length, 1 to 40 amino residues in length, 1 to 30 amino acid residues in length, 1 to 20 amino acid residues in length, 1 to 10 amino acid residue in length, 1 to 5 amino acid residues in length. In an embodiment, a peptide or polypeptide tether according to the present disclosure has a length of at least 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 25, 30, 35, 40, 45, 50, 60, 70 amino acids residues. In an embodiment, a peptide or polypeptide tether according to the present disclosure has a length of 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 25, 30, 35, 40, 45, 50, 60, or 70 amino acids residues.
The peptide or polypeptide tether may pre-dominantly comprise the following amino acid residues: Gly, Ser, Ala, or Thr. Suitable, non-immunogenic peptide tethers comprises glycineserine polymers for example, (GS)n (SEQ ID NO: 184), (G4S)n (SEQ ID NO: 185), (SG4)n (SEQ ID NO: 186), (GSGGS)n (SEQ ID NO: 187), (GGGS)n (SEQ ID NO: 188) or G4(SG4)n (SEQ ID NO: 189), wherein n is an integer between 1 and 10, typically between 2 and 4. A non- immunogenic peptide tether used herein may comprise glycine-alanine polymers, alanineserine polymers, and other flexible peptide tethers. A suitable peptide tether for fusing the polypeptide according to the present disclosure to the second polypeptide according to the present disclosure and/or to a moiety, such as an antibody or antibody fragment is a glycineserine polymer, such as (648)2 (SEQ ID NO: 190).
Peptide tethers can be also derived from immunoglobulin light or heavy chain constant domain, such as CLK or CL domains or the CH1 domain, but not all residues of such a constant domain, for example only the first 5 - 12 amino acid residues. In an embodiment, the peptide tether is not a immunoglobulin light or heavy chain constant domain. In an embodiment, the peptide linker is not a CLK, CL , CH1 , CH2 or CH3 domain. Exemplary peptide tethers which may be used in an antigen-binding molecule are derived from immunoglobulin light or heavy chain constant domain are QPKAAP (SEQ ID NO: 191) or ASTKGP (SEQ ID NO: 192). In general, peptide tethers can be derived from immunoglobulin heavy chains of any isotype, including for example Cy1 , Cy2, Cy3, Cy4, Ca1 , Ca2, C8, Cs, and Op.
A peptide tether may also comprise an immunoglobulin hinge (e.g. a human I gG 1 hinge or part thereof) or any peptide derived from such hinge. Preferably, where only a part or portion of an immunoglobulin hinge is used, the truncated hinge may not include one or more of its interchain cysteines. The presence of the interchain cysteines would allow for the formation of a dimeric peptide linker (or hinge region) by disulfide bridges.
It is understood that a peptide or polypeptide tethers as used herein is not limited to only one of the aforementioned and exemplified peptide tethers but my comprise any combination of two or more such tethers which are fused to each other. For instance, a peptide tether as used herein may be built from a glycine-serine polymer and an immunoglobulin hinge derived sequence in order to retain or obtain the desired activity.
Alternatively, a variety of non-proteinaceous polymers, including but not limited to polyethylene glycol (PEG), polypropylene glycol, polyoxyalkylenes, or copolymers of polyethylene glycol and polypropylene glycol, may be used as tethers.
The tether may be a cleavable tether facilitating release of the cytotoxic drug in the cell. For example, an acid-labile thether, peptidase-sensitive thether, dimethyl tether or disulfide- containing tether (Chari et al. (1992) Cancer Res. 52:127-131) may be used.
As further described herein, the total number of amino acid residues in a polypeptide of the present disclosure can be in the range of 25 to 50000, in the range of 25-10000, in the range of 25 to 5000, in the range of 25-1000, in the range of 25-500, in the range of 25-250, in the range of 25-100, in the range of 25-50, or in the range of 25-35, depending mainly on the length of the flexible linkers interconnecting the two a-helical peptides and the additional moieties which may be linked to the helix-turn-helix scaffold structure.
Particular fusions molecules
In an embodiment, the polypeptides of the present disclosure are fused to a poly-histidine tag. In other embodiment, the polypeptides of the present disclosure are fused to a FLAG tag. Yet in another embodiment, the polypeptides are fused to a FLAG and poly-histidine tag. In certain embodiments, said poly-histidine and/or FLAG tag is fused to the carboxyl- and/or aminoterminus of the polypeptide of the present disclosure. In another embodiment, the polypeptides of the present disclosure are fused to a maltose binding protein (MBP). Maltose binding protein may increase the solubility during expression in bacterial hosts. In certain embodiments, the maltose binding protein domain is fused to the amino-terminus of the polypeptide. In certain embodiments, the bacterial host used for expression is Escherichia coli. In another embodiment, an enzymatic cleavage side is present between the C-terminus of the maltose-
binding domain and the N-terminus of the polypeptides of the present disclosure. In certain embodiments, said enzymatic cleavage side is a FXa cleavage side.
Antibody - HTH polypeptide fusion molecules
The present disclosure provides a fusion molecule comprising an isolated polypeptide specific for IL-13Ra2 according to the present disclosure and an antibody or antibody fragment thereof.
In further embodiments, the polypeptides specific for IL-13Ra2 according to the present are fused to an antibody or an antibody fragment thereof. In certain embodiments, said antibody fragment is a Fab fragment. The antibody or antibody fragment thereof may have the same binding specificity or a different binding specificity as the polypeptides of the present disclosure.
The polypeptides specific for IL-13Ra2 according to the present disclosure may be fused to the antibody or an antibody fragment thereof, either by a direct linkage to the N- and/or C- terminus of the HTH polypeptides or by a peptide tether comprising one or more amino acid residues at the N- and/or C-terminus of the polypeptides. In an embodiment, the polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the C-terminus of the heavy chain of an antibody. In an embodiment, the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the N-terminus of the heavy chain of an antibody. In an embodiment, the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the C-terminus of the light chain of an antibody. In an embodiment, the HTH polypeptide according to the present disclosure is fused to the N- terminus of the light chain of an antibody. In an embodiment, the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the C-terminus of a Fab heavy chain. In an embodiment, the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the C-terminus of the CH1 domain of a Fab.
In an embodiment, the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the N-terminus of a Fab heavy chain. In an embodiment, the HTH polypeptide specific for IL- 13Ra2 according to the present disclosure is fused to the N-terminus of the VH domain of a Fab. In an embodiment, the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the C-terminus of a Fab light chain. In an embodiment, the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the C-terminus of the CL domain of a Fab. In an embodiment, the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the N-terminus of a Fab
light chain. In an embodiment, the HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the N-terminus of the VL domain of a Fab.
In an embodiment, the HTH polypeptide specific for IL-13Ra2 according to the present disclosure can be fused to an antibody or antibody fragment via a peptide tether comprising glycine-serine polymers, such as (GS)n (SEQ ID NO: 184), (G4S)n (SEQ ID NO: 185), (SG4)n (SEQ ID NO: 186), (GSGGS)n (SEQ ID NO: 187), (GGGS)n (SEQ ID NO: 188) or G4(SG4)n (SEQ ID NO: 189), wherein n is an integer between 1 and 10. A suitable peptide tether for fusing the polypeptide according to the present disclosure to an antibody or antibody fragment is a glycine-serine polymer, such as (648)2 (SEQ ID NO: 190).
The present disclosure also provides a novel approach of simultaneously engaging CD3 and the cancer specific antigen IL-13Ra2 via a fusion molecule having the following properties: (a) binding specificity for CD3; and (b) binding specificity for IL-13Ra2. This fusion molecule according to the present disclosure may be designed to provide a tumor-target-dependent activation of CD3 on T-cells, via IL-13Ra2 expressed on tumor cells. CD3 is a proven T-cell stimulating antigen with therapeutic relevance. The term "CD3” refers to an antigen which is expressed on T cells as part of the multi-molecular T cell receptor (TCR) and which consists of a homodimer or heterodimer formed from the association of two of four receptor chains: CD3-epsilon, CD3-delta, CD3-zeta, and CD3-gamma Human CD3 epsilon has the amino acid sequence as defined in UniProt: P07766 (SEQ ID NO: 193).
It is known that efficacy and safety of molecules with co-engagement of CD3 is mainly driven by the binding valency, the affinity of both specificities and the format used. The binding format should engage CD3 monovalently with moderate to low binding affinity to reduce the potential risk for side effects. Simultaneous binding of the bispecific fusion molecule according to the present disclosure to IL-13Ra2 expressed on cancer cells and to CD3 expressed on T-cells results in the formation of an immunological synapse wherein the cytotoxic T-cells are able to kill the cancer cell.
Thus, in an embodiment, the bispecific fusion molecule according to the present disclosure is capable of crosslinking a T-cell and an IL-13Ra2 expressing cell by simultaneous binding to an IL-13Ra2 expressing cell and a CD3 expressing T-cell. In an embodiment, such simultaneous binding results in lysis of the IL-13Ra2 expressing cell, particularly lysis of a tumor cell expressing IL-13Ra2. In one embodiment, such simultaneous binding results in activation of the T-cell. In an embodiment, the simultaneous binding results in a cellular response of a T-lymphocyte, particularly a cytotoxic T-lymphocyte, selected from the group of: proliferation, differentiation, cytokine secretion, cytotoxic effector molecule release, cytotoxic
activity, and expression of activation markers. In an embodiment, binding of the bispecific fusion molecule according to the present disclosure to CD3 without simultaneous binding to the target cell antigen does not result in T-cell activation.
In an embodiment, the bispecific fusion molecule according to the present disclosure is capable of re-directing cytotoxic activity of a T-cell to a IL-13Ra2 expressing cancer cell. Particularly, a T-cell according to any of the embodiments according to the present disclosure is a cytotoxic T-cell. In some embodiments, the T-cell is a CD4+ or a CD8+ T cell.
In certain embodiments, the present disclosure provides a bispecific fusion molecule comprising at least one HTH polypeptide specific for IL-13Ra2 according to the present disclosure and at least one antibody or antibody fragment specific for CD3.
In certain embodiments, the bispecific fusion molecule may comprise a second HTH polypeptide specific for IL-13Ra2 according to the present disclosure. In certain embodiments, the HTH polypeptides specific for IL-13Ra2 according to the present disclosure can be fused to the antibody Fab fragment as essentially described in Figure 6.
For example, a HTH polypeptides specific for IL-13Ra2 according to the present disclosure can be fused via a peptide tether to the C-terminus of the Fab heavy chain, the N-terminus of the Fab heavy chain, the C-terminus of the Fab light chain and/or the N-terminus of the Fab light chain. Preferably, the HTH polypeptide is fused to the C-terminus of the Fab heavy chain (Figure 6A). In certain embodiments, the HTH polypeptides specific for IL-13Ra2 according to the present disclosure is fused at its N-terminus or its C-terminus to the Fab. Preferably, the HTH polypeptide is fused at its C-terminus to Fab heavy chain. For example, the HTH polypeptides according to the present disclosure may be linked via a peptide tether between the C-terminus of the Fab heavy chain constant region (CH1) or the C-terminus of a Fab light chain constant region (CL) of the Fab. In some further embodiments, the peptide tether may be an unstructured (648)2 (SEQ ID NO: 190) tether. In this regard, in the bispecific fusion molecule according to the present disclosure, the Fab and/or the HTH polypeptide may be fused at its N-terminus and/or its C-terminus to another moiety. For example, when a first HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the heavy chain of a Fab, a second HTH polypeptide may be fused to the light chain of the Fab.
Alternatively, when a HTH polypeptide specific for IL-13Ra2 according to the present disclosure is fused to the heavy chain of a Fab, a second HTH polypeptide may be fused at its N-terminus and/or its C-terminus to the first HTH polypeptide fused to the Fab (Figure 6B). In an embodiment, said fusion may occur via an unstructured (648)2 (SEQ ID NO: 190) tether.
Accordingly, the present disclosure provides a bispecific fusion molecule comprising a polypeptide specific for IL-13Ra2 according to the present disclosure an antibody or antibody fragment specific for CD3.
In an embodiment, said IL-13Ra2 specific polypeptide comprised in the bispecific fusion molecule according to the present disclosure is fused to the C-terminus of the Fab heavy chain. In an embodiment, the Fab specific for CD3 comprises the variable heavy chain sequence of: EVQLVESGGGLVKPGGSLRLSCAASGFTFNTYAMNWVRQAPGKGLEWVGRIRSKYNNYAT YYADSVKDRFTISRDDSKNTLYLQMNSLKTEDTAVYYCTTHGNFGNSYVSWFAYWGQGTL VTVSS (SEQ ID NO: 180) and the variable light chain sequence of:
QTVVTQEPSLTVSPGGTVTLTCRSSTGAVTTSNYANWVQQKPGQAPRGLIGGTKFLAPGTP ARFSGSLLGGKAALTLLGAQPEDEAEYYCALWYSNLWVFGGGTKLTVLGQ (SEQ ID NO: 179).
In an embodiment, the Fab specific for CD3 comprises the heavy chain sequence of: EVQLVESGGGLVKPGGSLRLSCAASGFTFNTYAMNWVRQAPGKGLEWVGRIRSKYNNYAT YYADSVKDRFTISRDDSKNTLYLQMNSLKTEDTAVYYCTTHGNFGNSYVSWFAYWGQGTL VTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAV LQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKRVEPKSC (SEQ ID NO: 178) and the light chain sequence of:
QTVVTQEPSLTVSPGGTVTLTCRSSTGAVTTSNYANWVQQKPGQAPRGLIGGTKFLAPGTP ARFSGSLLGGKAALTLLGAQPEDEAEYYCALWYSNLWVFGGGTKLTVLGQPKAAPSVTLFP PSSEELQANKATLVCLISDFYPGAVTVAWKADSSPVKAGVETTTPSKQSNNKYAASSYLSLT PEQWKSHRSYSCQVTHEGSTVEKTVAPTECS (SEQ ID NO: 155).
In an embodiment, the IL-13Ra2 specific polypeptide comprised in the bispecific fusion molecule according to the present disclosure comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 47 - 122.
In an embodiment, the bispecific fusion molecule according to the present disclosure is composed of at least 2 polypeptides, wherein a. a first polypeptide comprises the light chain of the Fab specific for CD3, b. a second polypeptide comprises from its N-terminus to its C-terminus i. a heavy chain of the Fab specific for CD3 ii. a peptide tether and
iii. a polypeptide specific for IL-13Ra2 according to the present disclosure.
In an embodiment, the first polypeptide chain comprises the amino acid sequence of SEQ ID NO: 155. In an embodiment, the second polypeptide chain comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 150, 151 , 156 - 167, 171 , and 173.
In an embodiment, the second polypeptide chain comprises the amino acid sequence of EVQLVESGGGLVKPGGSLRLSCAASGFTFNTYAMNWVRQAPGKGLEWVGRIRSKYNNYAT YYADSVKDRFTISRDDSKNTLYLQMNSLKTEDTAVYYCTTHGNFGNSYVSWFAYWGQGTL VTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAV LQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKRVEPKSCEFGGGGSGGGGSG DELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQGGGGGSGGGGSGDELTQLKRELQKL YRQGVDSDELRALIRKLHMLEQG (SEQ ID NO: 150)
In another embodiment, the second polypeptide chain comprises an amino acid sequence of EVQLVESGGGLVKPGGSLRLSCAASGFTFNTYAMNWVRQAPGKGLEWVGRIRSKYNNYAT YYADSVKDRFTISRDDSKNTLYLQMNSLKTEDTAVYYCTTHGNFGNSYVSWFAYWGQGTL VTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAV LQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKRVEPKSCEFGGGGSGGGGSG DELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQGGGGGSGGGGSGDELTQLKRELQKL YRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 151)
In an embodiment, the bispecific fusion molecule according to the present disclosure bind to human IL-13Ra2 with an ECso concentration of 10 nM or less, such as 5 nM, 3 nM, 2 nM, 1 nM, 0.6 nM, 0.5 nM, 0.4 nM or 0.3 nM.
In an embodiment, said IL-13Ra2 is expressed on cells. In an embodiment, said IL-13Ra2 is human IL-13Ra2 expressed on cells. In an embodiment, said IL-13Ra2 is cynomolgus IL- 13Ra2 expressed on cells. In an embodiment, said IL-13Ra2 is mouse IL-13Ra2 expressed on cells. In an embodiment, said binding is measured in an FACS assay as described in Example 11. In certain embodiment, the bispecific fusion molecule according the present disclosure may be capable of co-stimulating T-cell responses in a functional T-cell activation assay as described in Example 12. In an embodiment, the bispecific fusion molecule according to the present disclosure may be capable to mediate killing of IL-13Ra2 expressing cancer cells. In an embodiment, the bispecific fusion molecule according to the present disclosure may be capable to induce T-cell mediated killing of cancer cells as described in Example 12. In an embodiment, the bispecific fusion molecule according to the present disclosure may be capable to induce T-cell mediated killing of A-375 cells in vitro as described in Example 12. In an embodiment, the bispecific fusion molecule according to the disclosure may be capable to
induce T-cell mediated killing of A-375 cells in vitro with an IC50 concentration of 10 nM or less, such as 5 nM, 3 nM, 2 nM, 1 nM, 0.6 nM, 0.5 nM, 0.4 nM or 0.3 nM.
Therapeutic methods
The isolated polypeptide specific for IL-13Ra2 or the fusion molecule according to the present disclosure may be used in therapeutic methods. The polypeptide or fusion molecule may be used for the treatment of a disease, such as cancer, an autoimmune disease or inflammatory disease. In an embodiment, the present disclosure provides a method for the treatment of a disease.
In an embodiment, the present disclosure provides an isolated polypeptide or a fusion molecule according to the present disclosure for the treatment of a disease. In an embodiment, the present disclosure provides an isolated polypeptide or a fusion molecule according to the present disclosure for use in the treatment of a disease. In an embodiment, the present disclosure provides an isolated polypeptide or a fusion molecule according to the present disclosure for use in the treatment of a disease in a subject in need thereof. In an embodiment, the present disclosure provides the use of an isolated polypeptide or a fusion molecule according to the present disclosure for the manufacture of a medicament. In an embodiment, the present disclosure provides an isolated polypeptide or a fusion molecule according to the present disclosure for use in medicine. In an embodiment, the present disclosure provides an isolated polypeptide or a fusion molecule according to the present disclosure for use as a medicament. In an embodiment, the present disclosure provides an isolated polypeptide or a fusion molecule according to the present disclosure for use in medicine. In an embodiment, the present disclosure provides an isolated polypeptide or a fusion molecule according to the present disclosure for use as a medicament for the treatment of a subject in need thereof. In an embodiment, the disease is associated with the undesired presence of IL13-Ra2, in particular human IL-13Ra2.
In an embodiment, the disease to be treated is a proliferative disease. In a particular embodiment, the disease is cancer. Non-limiting examples of cancers include bladder cancer, brain cancer (such as glioblastoma), head and neck cancer, pancreatic cancer, lung cancer, breast cancer, ovarian cancer, uterine cancer, cervical cancer, endometrial cancer, esophageal cancer, colon cancer, colorectal cancer, rectal cancer, gastric cancer, prostate cancer, blood cancer, sarcoma, skin cancer, squamous cell carcinoma, bone cancer, melanoma, renal cell carcinoma, and kidney cancer.
In an embodiment, the disease to be treated is an autoimmune or inflammatory disease. Nonlimiting examples an autoimmune or inflammatory disease include rheumatoid arthritis (RA),
psoriasis, psoriatic arthritis, systemic lupus erythematosus (SLE), lupus nephritis, type I diabetes, Grave's disease, Inflammatory bowel disease (IBD), Crohn's disease (CD), ulcerative colitis (UC), irritable bowel syndrome, multiple sclerosis (MS), autoimmune myocarditis, Kawasaki disease, coronary artery disease, chronic obstructive pulmonary disease (COPD), interstitial lung disease, autoimmune thyroiditis, scleroderma, systemic sclerosis, osteoarthritis, atoptic dermatitis, vitiligo, graft vs. host disease, Sjogren's syndrome, autoimmune nephritis, Goodpasture's syndrome, chronic inflammatory demyelinating polyneuropathy, ANCA-associated vasculitis, uveitis, scleroderma, bullous pemphigoid, Alzheimer's Disease, amyotrophic lateral sclerosis, Huntington's Chorea, cystic fibrosis, gout, age-related macular degeneration, allergy, asthma, antiphospholipid syndrome (APS), atherosclerosis, C3 glomerulopathy and IgA nephropathy, ischemia/reperfusion injury, peritonitis, sepsis and other autoimmune diseases that are a result of either acute or chronic inflammation.
In an embodiment, the present disclosure provides an isolated polypeptide or a fusion molecule specific for IL-13Ra2 according to the present disclosure for use in a method of treating a subject having a disease comprising administering to the subject a therapeutically effective amount of a polypeptide or a fusion molecule according to the present disclosure.
In an embodiment, the method further comprises administering to the subject a therapeutically effective amount of at least one additional therapeutic agent. The subject in need of treatment is typically a mammal, more specifically a human. For use in therapeutic methods, an isolated polypeptide or a fusion molecule according to the present disclosure would be formulated, dosed, and administered in a way consistent with good medical practice.
Pharmaceutical compositions
In an embodiment, the present disclosure provides a pharmaceutical composition comprising a polypeptide or a fusion molecule according to the present disclosure and a pharmaceutically acceptable carrier or excipient.
The pharmaceutical compositions may further comprise at least one other pharmaceutically active compound. The pharmaceutical composition according to the present disclosure can be used in the diagnosis, prevention and/or treatment of diseases associated with the undesired presence of IL-13Ra2, in particular human IL-13Ra2. In particular, the present disclosure provides a pharmaceutical composition comprising a polypeptide or a fusion molecule according to the present disclosure that is suitable for prophylactic, therapeutic and/or diagnostic use in a mammal, more particular in a human.
In general, a polypeptide or a fusion molecule according to the present disclosure may be formulated as a pharmaceutical composition comprising at least one polypeptide or fusion molecule according to the present disclosure and at least one pharmaceutically acceptable carrier or excipient, and optionally one or more further pharmaceutically active compounds. Such a formulation may be suitable for oral, parenteral, topical administration or for administration by inhalation. Accordingly, a pharmaceutical composition comprising at least one polypeptide or fusion molecule according to the present disclosure may be administered parenterally, such as intravenously, or intramuscularly, or subcutaneously. Alternatively, a polypeptide or fusion molecule according of the disclosure may be administered via a non- parenteral route, such as per-orally or topically. In a preferred embodiment, a pharmaceutical composition comprising a polypeptide or a fusion molecule according to the present disclosure is administered intravenously or subcutaneously.
In particular, a polypeptide or a fusion molecule according to the present disclosure may be used in combination with one or more pharmaceutically active compounds that are or can be used for the prevention and/or treatment of the diseases in which a target antigen of interest is involved, as a result of which a synergistic effect may or may not be obtained. Examples of such compounds, as well as routes, methods and pharmaceutical formulations or compositions for administering them will be clear to the clinician.
In an embodiment, the present disclosure provides a pharmaceutical composition comprising a polypeptide or a fusion molecule according to the present disclosure for use in the prevention and/or treatment of a disease associated with the undesired presence of IL-13Ra2. In an embodiment, the present disclosure provides a pharmaceutical composition comprising a polypeptide or a fusion molecule according to the present disclosure for the use as a medicament. In an embodiment, the present disclosure provides a pharmaceutical composition comprising a polypeptide or a fusion molecule according to the present disclosure for use in the prevention and/or treatment of an autoimmune disease and/or inflammatory disease and/or cancer.
In an embodiment, the present disclosure provides a method for the treatment of an autoimmune disease and/or inflammatory disease and/or cancer in a subject in need thereof using a pharmaceutical composition comprising a polypeptide or a fusion molecule according to the present disclosure.
Further provided is a method of producing a polypeptide or a fusion molecule according to the present disclosure in a form suitable for administration in vivo, the method comprising (a) obtaining a polypeptide or a fusion molecule according by a method disclosed herein, and (b) formulating said polypeptide or fusion molecule with at least one pharmaceutically acceptable
carrier or excipient, whereby a preparation of a polypeptide or fusion molecule is formulated for administration in vivo.
Pharmaceutical compositions according to the present disclosure comprise a therapeutically effective amount of one or more polypeptides or fusion molecules according to the present disclosure dissolved in a pharmaceutically acceptable carrier or excipient.
Diagnostic use
In an embodiment, the present disclosure provides the use of an isolated polypeptide specific for IL-13Ra2 according to the present disclosure for diagnosis tic purposes. In an embodiment, the present disclosure provides the use of an isolated polypeptide specific for IL-13Ra2 according to the present disclosure for the diagnosis of a disease. In an embodiment, the present disclosure provides the use of an isolated polypeptide specific for IL-13Ra2 according to the present disclosure for the detection of IL-13Ra2, in particular human IL-13Ra2. In an embodiment, the present disclosure provides a method for detecting IL-13Ra2 in a subject or a sample, comprising the step of contacting said subject or sample with an isolated polypeptide specific for IL-13Ra2 according to the present disclosure. In an embodiment, the present disclosure provides a method for diagnosing a disease in a subject, comprising the step of contacting said subject or sample with an isolated polypeptide specific for IL-13Ra2 according to the present disclosure.
WORKING EXAMPLES
Example 1 : Antigen generation and quality control
Synthetic peptides
Selected IL-13Ra2-specific HTH polypeptides of the present disclosure were chemically synthesized either with a biotin tag or fluorescence dye (PSL GmbH, Germany). Alternatively, peptides were conjugated to cytotoxic drugs (such as MMAE, MMAF, DM-1 or DM-4) (PSL GmbH, Germany). Peptides were purified and delivered as lyophilized material. The lyophilized peptides were stored at -80°C and reconstituted in an appropriate buffer when needed.
Recombinant proteins
Human IL-13 and mouse IL-13Ra2
Recombinant human IL-13 (hlL-13) or mouse IL-13Ra2 was produced in house (see Table 2). DNA encoding hlL-13 (Uniprot: P35225) or mouse IL-13Ra2 (Uniprot: 088786) was cloned in frame with an N-terminal VK leader sequence and a C-terminal AVI-6xHis-tag into a pMAX expression vector, which is a modified expression vectTable 1or based on pcDNA3.1 (Thermo Fisher).
HEK293-6E cells were transiently transfected one day post seeding with a commercially available transfection reagent according to the manufacturer’s instructions. The cells were cultured for 3 days and the conditioned cell culture supernatant was harvested by centrifugation followed by sterile filtration (0.22 pm). The conditioned cell culture supernatants were harvested by centrifugation followed by sterile filtration (0.22 pm). The antigen was purified by IMAC using Protino Ni-NTA columns (Macherey-Nagel). All chromatography steps were performed using AKTA chromatography systems (GE Healthcare). The samples were buffer-exchanged to D-PBS using PD10 columns (GE Healthcare). In some cases, a polishing preparative SEC step was performed in D-PBS using a Superdex 200 column (GE Healthcare). If required, biotinylation of the protein was performed by in vitro biotinylation using the BirA Kit (Avidity) followed by a preparative SEC using a Superdex 200 column (GE Healthcare). The quality of the samples was analyzed by denaturing, reducing or non-reducing SDS-PAGE, Streptavidin-Shift Assay, HP-SEC and DLS.
Cell lines with cell surface expression of IL-13Rcc2
CHO Flp-ln cells (Invitrogen) stably expressing full-length cynomolgus IL13Ra2 and CHO Flp- In cells transiently expressing full-length human IL-13Ra2 or mouse IL-13Ra were generated in-house. For the generation of the Flp-ln CHO cells, various vector constructs encoding the respective full-length receptor were gene synthesized in-house and transfection of cells was
performed according to the instructor’s manual (Thermofischer I Invitrogen). For fluorocytometric evaluation of receptor expression levels of transfected cells, cells were blocked in Superblock (Thermo Scientific) and subsequently incubated for 60 min on ice with a commercially available receptor-specific antibody diluted in Superblock to a final concentration of 5 pg/ml. After washing with PBS supplemented with 3% fetal calf serum (FCS) and 0.02% sodium azide, cells were incubated for 30 min on ice with a PE-conjugated anti- F(ab’)2 detection antibody (Jackson Immuno Research) diluted in Superblock. Cells were washed again and cells evaluated in a FACS Array (BD Biosciences).
Example 2: Isolation and characterization of IL-13Ra2 specific Helix-Turn-Helix (HTH) polypeptides
To isolate Helix-Turn-Helix (HTH) polypeptides binding to IL-13Ra2, the HTH polypeptide phage-display library described in WO 2017/149117 (MORPHOSYS AG) was used for selections against human IL-13Ra2. The library is based on a de novo generated helix-turn- helix scaffold structure, wherein two a-helical peptides form a conformational constrained coiled-coil structure. Each of the two a-helical peptides is diversified at six solvent exposed positions leading to a total number of 12 diversified positions.
Human IL-13Ra2 was used as a human Ig-Fc fusion protein (R&D Systems; Catalog Number: 7147-IR).
Selections were performed in solution using Protein G coupled magnetic beads (Dynabeads Protein G, Life Technologies) which are able to capture the antigen-phage complex by the human Fc-tag of the antigen. The polypeptide library was handled according to published standard protocols for phage display based peptide selections (Zwick, M. B., Menendez, A., Bonnycastle, L. L. C. and Scott, J.K. (2001). In C. F. Barbas, D. R. Burton, J.K. Scott and G. J. Silverman, (Eds.), Phage Display: A Laboratory Manual (pp.18.1-18.44). New York: Cold Spring Harbor Laboratory Press) with minor adjustments in terms of selection stringency and adaptation to phagemide vector system. The selections were performed over 3 subsequent enrichment rounds with monitoring of specific sequences by conventional sequencing. In short, all pannings were completed with various antigen concentrations (100 nM for round 1 , 50 nM for round 2, and 25 nM for round 3) under standard washing conditions. The target protein was incubated with pre-adsorbed phages. Washing of the coated magnetic Dynabeads was carried out with a magnetic particle separator and incubations were done by overhead rotation in low binding tubes. Subsequently, specifically bound phages were eluted using Glycine/HCl. E. coli TG1 F’ with an GD600nm of 0.6-0.8 was added to the phage eluates of each selection and was incubated in an incubator without shaking. After infection, bacteria were plated out evenly on
two large LB/Chloramphenicol/Glucose agar plates for each selection and incubated overnight at 37°C and Glycerol phage stocks were prepared. For the following panning rounds bacterial suspensions of each pool were collected and used to propagate phages for an additional panning round as described above. After each round of panning the phage titer was determined. The expected range goes from 1.0E+06 - 1.0E+07 phage/ml for the output titer.
After completion of the panning rounds, phage output pools were subcloned via PGR into an expression vector to facilitate the cytoplasmic expression of the polypeptides in E. coli. Expression of single clones resulted in the production of polypeptides that were N-terminally fused to the Maltose Binding Protein (MBP) and that included a Factor FXa protease cleavage site. In addition, all polypeptides were fused C-terminally to a Hiss-Flag tag (HHHHHHKDIRDYKDDDDK (SEQ ID NO: 181)). To check for target specificity, ELISA and FACS screenings with crude bacterial cell extracts containing the HTH polypeptides obtained from the panning outputs were carried out. For ELISA screenings, this was done by capturing the Fc-tagged hlL-13Ra2/Fc (R&D Systems; Catalog Number: 7147-IR) protein or unrelated Fc-tagged proteins (such as human Ox40/Fc (R&D Systems)) on the surface of a microtiter plate pre-coated with an anti-human Fc specific antibody (Jackson Immuno Research) and by adding E. coli lysates containing the polypeptides obtained from the panning outputs. For mouse IL-13Ra2, this was done by direct coating of the avi-His tagged protein on the surface of a microtiter plate. Bound polypeptides were detected by the encoded M BP-tag using anti- MBP detection (Abeam). To analyze polypeptide expression, anti-His capture (R&D Systems) and anti-MBP detection (Abeam) was applied.
ELISA screening experiments also included counter antigens human, mouse IL-13Ra1/Fc (R&D Systems; Catalog Number: 146-IR, Catalog Number: 491-IR/CF) and human, mouse IL4Ra/Fc (R&D Systems; Catalog Number: 7700-4R, Catalog Number: 530-MR) which helped to identify polypeptides truly specific for the IL-13Ra2 protein. As positive controls, mouse anti hlL-13Ra2 IgG (BioLegend), mouse anti hlL-13Ra1 IgG (R&D Systems), and mouse anti hlL- 4Ra IgG (R&D Systems) complexed with alkaline phosphate conjugated goat anti mouse IgG (Fab’)2 specific AP (Jackson) were used. FACS based cell screening experiments on the cancer cell line A-375 expressing hlL-13Ra2 but not hlL13Ra1/IL4Ra as well as cell binding studies on CHO cells engineered to stably express cynomolgus monkey IL-13Ra2 further confirmed cell binding and species cross-reactivity of the identified HTH polypeptides. Clones with desirable binding profiles were subjected to sequence analysis. Finally, 31 sequence unique clones specifically recognizing human, mouse, and cynomolgus IL-13Ra2 in ELISA and/or FACS with no binding to IL-13Ra1, IL-4Ra or to IL-13Ra1/IL-4Ra receptor complex were selected for recombinant production in E.coli in larger scale.
Large Scale Production and generation of M BP-free HTH polypeptides
For larger scale production, HTH polypeptides were expressed in E. coli cells as a N-terminally tagged maltose-binding protein (MBP) that includes a Factor FXa protease cleavage and a C- terminally tagged (His)e-Flag-fusion protein ("(His)6" disclosed as SEQ ID NO: 206). Protein expression was induced by the addition of IPTG and cultures were further cultivated for 20-23 h. Cells were harvested by centrifugation and the pellet was resuspended in lysis buffer (PBS buffer plus 2 mM MgCh, 20 ll/ml Benzonase (Roche) and 1 tablet/50ml complete, EDTA-free protease inhibitor cocktail tablets (Roche)). Cells were disrupted either by chemical lysis or high-pressure homogenization. The resulting suspension was centrifuged and the supernatant was sterile filtered for further purification steps. HTH polypeptide fusion molecules were purified by Dextrin-Sepharose affinity chromatography using a MBP-Trap column (GE- Healthcare) and optionally polished by cation exchange chromatography using a Hi-Trap SP FF column (GE LifeSciences).
Table 3: Amino acid sequences of bacterial produced IL-13Ra2 specific helix-turn-helix polypeptides including tags.
For generation of M BP-free HTH polypeptides, the purified M BP-fusion molecules were buffer- exchanged by PD10 columns (GE Healthcare) into FXa-digest buffer (20 mM Tris/HCI pH 8.0; 100 mM NaCI; 2 mM CaCh). The HTH polypeptides were released from the maltose-binding protein by addition of Factor Xa (1 :100 (w/w)) and incubation O/N in a rotary shaker at room temperature. The released peptides were further purified by IMAC using either 1 ml Protino IMAC columns or self-packed 1 ml columns filled with Profinity IMAC Ni-charged resin. All affinity chromatography steps were performed using the AKTA Avant 25 preparative chromatography system for low-throughput applications and using the Gilson ASPEC GX-274 solid phase extraction workstation for high-throughput applications. Buffer exchange to PBS was performed using PD 10 columns (GE Healthcare). Samples were sterile filtered and HTH polypeptide concentration was determined by UV-spectrophotometry. The purity and integrity of the samples were analyzed in denaturing, reducing or non-reducing SDS-PAGE, SEC- HPLC and CD-spectroscopy. Purified HTH polypeptides were re-characterized in more detail for ELISA and cell binding, affinity, and functional activity in a relevant in vitro assay.
Example 3: Characterization of IL-13Ra2 specific MBP-free HTH-polypeptides for ELISA and cell binding (FACS)
ELISA binding of 31 purified MBP-free HTH-polypeptides to human IL-13Ra2/Fc and mouse IL-13Ra2/avi-His as well as binding to human IL-13Ra1/Fc and human IL4Ra/Fc was analyzed. Cell binding of the HTH polypeptides to human, cynomolgus monkey, and mouse
IL-13Ra2 expressed on CHO cells was analyzed by FACS. In addition, cell binding to the parental CHO cells, the IL-13Ra2 positive but IL-13Ra1 I IL4Ra negative cancer cell line A- 375 (ATCC® CRL-1619™) and to the IL-13Ra2 negative but IL-13Ra1 I IL-4Ra positive cancer cell line A-549 (ATCC® CCL-185™) (for expression profile see LaPorte et al., Cell. 2008 Jan 25; 132(2): 259-72.) was tested.
Methods:
ELISA: Fc fusion proteins were captured at saturating concentration via an anti-human-Fc- capture (Jackson Immuno Research) specific antibody on Maxisorp plates. Avi-His fusion proteins were direct coated at saturating concentrations on Maxisorp plates. Bound M BP-free HTH polypeptide fusions were detected using a mouse anti-FLAG M2 conjugated to alkaline phosphatase (AP) secondary detection antibody (Sigma).
FACS: Cells were adjusted to 2x106 cells/ml in PBS/3% FCS/0.02% NaN3 (FACS buffer). FACS staining was performed in V-bottom 96-well microtiter and 1x 105 cells per well were mixed with purified HTH-polypeptides (diluted in FACS buffer) and incubated on ice for 1h. After 1 h incubation on ice cells were washed 4x with FACS buffer and taken up in 50 pl phycoerythrin-conjugated (PE) anti-FLAG M2 secondary antibody (Jackson Immuno Research), diluted 1 :100 in FACS buffer. After 30 min incubation on ice, cells were washed 4x with FACS buffer, resuspended in 100 pl FACS buffer and cell surface binding of IL-13Ra2 specific HTH polypeptides was measured via FL2 fluorescence intensity of cells in FACSArray (Becton Dickinson).
Results:
Results are summarized in Table 6.
9 sequence unique HTH polypeptides (Table 5) revealed reproducible strong cell binding to human, cynomolgus, and mouse IL-13Ra2 expressed on cells and no cell binding to the IL- 13Ra2 negative but IL-13Ra1/IL-4Ra positive cell line A-549. ELISA experiments with purified HTH polypeptides confirmed lack of binding to recombinant human IL13Ra1/Fc and human IL4Ra/Fc.
HTH-71 and HTH-125, both sharing preferred binding properties, were selected as a lead and back-up candidate, respectively. Noteworthy, both molecules share almost identical amino acid sequences (Table 1 and Figure 1) with the only difference in 3 amino acid positions located at variable positions Var4, Var5, and Var6 in Helix-2 indicating that these positions are less important for antigen binding.
This assumption was later confirmed in Ala-scanning experiments for HTH-71 and additional variants designed for HTH-71 (see Example 4). In sum, highly specific HTH-polypeptides specifically recognizing IL-13Ra2 were identified. These molecules can be further developed for diagnostic, imaging and therapeutic intervention of various types of cancer.
Table 5: Amino acid sequence of the core helix-turn-helix sequence of IL-13Ra2 specific HTHs identified in ELISA and FACS experiments.
Example 4: Optimization of HTH polypeptides
Lead HTH polypeptide HTH-71 was subjected to protein optimization.
Variants were designed to remove potential sites of oxidation and/or isomerization in the invariant positions of the HTH-scaffold structure of HTH-71. Isomerization and/or oxidation of HTH polypeptides can lead to product heterogeneity and loss of activity. In order to reduce the risk of oxidation, a M residue found in the invariant HTH-scaffold structure position X3 in Helix- 2 of HTH-71 was mutated to A. In addition, a D residue present in the Linker sequence Li was mutated to G to prevent potential isomerization. All variants were generated by site-directed
mutagenesis according to the manufacturer's recommendations. Sequencing of selected clones was performed using conventional techniques, which confirmed the presence of the selected mutation. Variant HTH polypeptides were produced in mammalian cells as Fab-HTH- HTH fusion molecules (bivalent HTH binding to IL-13Ra2, monovalent Fab binding to CD3) using glycine-serine tethers as described in Example 10 and assayed for binding to A-375 cells and for T-cell mediated killing of A-375 cells as described in Example 11 and 12.
Table 7: HTH scaffold amino acid sequence of generated variants of HTH-71 used in the Fab-HTH-HTH fusion molecules. As negative control, only one HTH (HTH-58) molecule was fused to the CD3-specific Fab heavy chain.
Results:
A summary of the binding and killing results is shown in Table 6. As expected, Fab-HTH-HTH fusion molecules comprising the negative control HTH polypeptide HTH-58 revealed no binding or killing of A-375 cells. Fab-HTH-HTH fusion molecules comprising a sequence optimized HTH-71 variant revealed similar binding to and killing of A-375 cells when compared to the parent Fab-HTH-HTH fusion. Accordingly, the removal of the potential isomerization site in the Linker sequence and the removal of the potential oxidation site in Helix-2 does not substantially affect binding and functional activity of the HTH polypeptides of the present disclosure. The absence or presence of a C-terminal G in the bispecific constructs BIS-602 and BIS-659, respectively, did not negatively affected binding and killing of the tested fusion molecules.
Table 10: Binding of Fab-HTH-HTH fusion molecules to A-375 cells as well as T-cell mediated killing of A-375 cells in the presence of human PBMCs.
Example 5: Identification of amino acid residues required for target binding - alanine scanning experiments.
To determine which of the variant positions of the identified HTH polypeptides are relevant for interaction with human IL-13Ra2, all variable positions Vari to Var6 on Helix-1 and Helix-2 of HTH-71 were separately mutated to Ala. Ala mutants were generated by site-directed mutagenesis according to the manufacturer's recommendations. Sequencing of selected clones was performed using conventional techniques, which confirmed the presence of the selected mutation. Mutants were generated and produced in mammalian cells as Fab-HTH- HTH fusion molecules as described in Example 4 and 10.
Produced Fab-HTH-HTH fusion molecules were tested for binding to IL-13Ra2 expressing cells as described in Example 11 as well as their capability to mediate redirected T-cell cytotoxicity as described in Example 12.
Results:
A summary of the sequences and the functional properties of the tested Ala-variants are summarized in Table 13. Amino acid residues of variant positions of HTH-71 , which were
involved or not involved in binding to IL-13Ra2 are shown in Table 14. Single Ala-mutants at positions Var2, Var3, Var4, Var5, and Var6 on Helix-1 as well as at positions Vari and Var2 on Helix-2 significantly abolished binding to and killing of A-375 cells. These findings indicate that Vari on Helix-1 as well as Var3, Var4, Var5 and Var6 on Helix-2 are not relevant for binding of the HTH-71 to IL-13Ra2.
Table 14: Overview of variable positions on Helix-1 and Helix-2 of HTH-71 being relevant and not relevant for binding to IL-13Ra2.
The aforementioned finding was partly confirmed by the back-up candidate HTH-125. HTH- 125 differs from HTH-71 in variable positions Var4, Var5 and Var6 on Helix-2 (see Table 15)
but has similar binding characteristics as HTH-71 (see Example 2) supporting that these variant positions are not relevant for binding to the IL-13Ra2 receptor.
Additional variants and further studies:
Additional variants of HTH-71 were designed to confirm the results of Example 4 and to further elucidate the aforementioned identified relevant positions of HTH-71 for binding to IL-13Ra2. For these studies, single MBP-free Hise/Flag tagged polypeptides ("Hise" disclosed as SEQ ID NO: 206) with monovalent binding to IL-13Ra2 were generated and recombinantly produced in bacteria and purified as described Example 1. Purified HTH polypeptides were tested for ELISA and cell binding to recombinant IL-13Ra2 proteins from different species as described in Example 1.
Results:
A summary of the variants and their binding characteristics are summarized in Table 16. In brief, the removal of a potential isomerization site (DS
DG) in the linker sequence of HTH- 71 and/or oxidation site (M A) in Helix-2 did not affected binding of HTH-71 to IL-13Ra2.
Conservative amino-acid mutations in Var6 of Helix-1 of HTH-71 had no effect on binding and conservative mutations in Var5 of Helix-1 revealed only minor effects on binding of HTH-71 on A-375 cells. In contrast, non-conservative amino acid mutations in Var3 and Var4 of Helix-1 resulted in loss of binding whereas a non-conservative amino acid mutation in positions Var4 of Helix- 1 of HTH-71 had only a minor effect on binding of HTH-71.
In sum, the generated variants for HTH-71 confirmed, that specific mutations in invariant positions of the HTH scaffold structure did not affected the binding and functional activity of HTH-71. Furthermore, conservative amino acid substitutions in variable positions of HTH-71 involved in binding to IL-13Ra2 did not altered binding or killing activity when compared to the parental molecule, whereas non-conservative substitutions in such positions resulted in loss- of-binding and killing activity.
Table 16: Summary of the sequences and functional properties of the tested variants of HTH-71
Example 6: IL-13Rot2 receptor internalization assays
The ability of identified IL-13Ra2 specific HTH polypeptides to deliver therapeutic agents into the cytoplasm of targeted cells expressing IL13Ra2 was assessed by analyzing their capacity to induce IL13Ra2 receptor internalization on target cells. IL-13Ra2 specific HTH polypeptides HTH-71 (SEQ ID NO: 66), HTH-125 (SEQ ID NO: 77), and HTH-82 (SEQ ID NO: 78), as well as one negative control HTH polypeptide were chemically synthesized by an external service provider (JPT, Germany). HTH polypeptides were either N-terminally biotinylated or alternatively directly conjugated to the fluorescence dye Atto488.
Method
To evaluate internalization of the HTH polypeptides, a FACS approach was employed by using fluorescently labeled HTH polypeptides. In these studies, CHO Flpln (Invitrogen) cells expressing the full-length cynomolgus IL13Ra2 or parental CHO cells as negative control cells were used. Cell bound biotinylated HTH polypeptides (tested at a final concentration of 100 nM) were incubated with Streptavidin-Alexa488 (Jackson) and the formed complex was incubated at 4°C and in parallel at 37°C for different time points to allow for receptor
internalization. After incubation, cells were immediately placed on ice. Cells were then transferred to a 384 well plate and washed with chilled FACS buffer containing NaNs. To quench the fluorescence of the HTH-polypeptide/Alexa488 complex which was not internalized, an anti-Alexa-488 IgG (Life Technologies/Thermo) was added and respective fluorescence levels were determined for quenched and non-quenched samples.
The amount of internalized fluorescence was determined based on the signal intensities of unquenched and quenched samples at individual time point using the following formula:
Internalization percentage = (1 -(Nx - Qx) I ((Nx - (Nx * QO / NO))) * 100
N1 = Fluorescence of unquenched sample at time point x
NO = Fluorescence of unquenched sample at time point zero
Q1 = Fluorescence of quenched sample at time point x
QO = Fluorescence of quenched sample at time point zero
Results:
Results from the internalization assay are shown in Figure 2 and are summarized in Table 17.
IL-13Ra2 specific HTH polypeptides were internalized in a time dependent manner, with HTH- 71 showing the fasted and highest rate of internalized fluorescence with up to 38% internalized fluorescence after 120 minutes.
In sum, the identified IL-13Ra2 specific HTH polypeptides of the present disclosure efficiently internalize into cells expressing IL-13Ra2 and as such are well suited to deliver therapeutic agents into cells expressing IL13Ra2.
Table 17: Internalized fluorescence of HTH-bio/Strep-A-488 complexes in CHO cells stably transfected with full-length IL13Ra2 protein.
Example 7: Design of HTH polypeptide drug conjugates (PDCs)
To confirm the ability of the identified IL-13Ra2 specific HTH polypeptides to deliver therapeutic agents into cancerous cells, IL-13Ra2 specific HTH polypeptide drug conjugates (PDCs) were generated and tested for toxin-mediated in vitro killing of A-375 cells and A-549 cells. As one exemplary cytotoxic agent, Monomethyl-Auristatin F (MMAF) was selected. Monomethyl auristatin F is an antimitotic agent, which inhibits cell division by blocking the polymerisation of tubulin. Chemical peptide synthesis of HTH-71 with N-terminal and C- terminal conjugation to MMAF using a PEG9 (1-amino-3,6-dioxaoctanoic acid) tether was performed by an external service provider (PSL GmbH, Germany). Fusion of two HTH- polypeptides to each other was done by a (648)2 (SEQ ID NO: 190) tether.
The following PDCs were generated:
PDC-1: MMAF - PEG9 - HTH-71 (one HTH polypeptide, one toxin)
PDC-2: MMAF - PEG9 - HTH-71 - (G4S)2 - HTH-71 (two HTH polypeptides, one toxin) ("(G4S)2" disclosed as SEQ ID NO: 190)
PDC-3: MMAF - PEG9 - HTH-71 - (G4S)2 - HTH-71 - PEG9 - MMAF (two HTH polypeptides, two toxins) ("(G4S)2" disclosed as SEQ ID NO: 190)
Cell binding
Cell binding studies of the PDCs was basically performed as described in Example 3 with the difference that cell bound PDCs were stained with a biotin-conjugated anti-MMAF secondary antibody (Epitope Diagnostics) with subsequent detection of the formed complex via Strep-PE (Jackson Immuno Research).
Results cell binding
Results of the cell binding studies are summarized in Table 19. PDCs retained cell binding to the A-375 and CHO cells expressing cynomolgus IL-13Ra2 in a dose dependent manner. No binding to the target negative cancer cell line A-549 and to parental CHO cells could be observed. PDC-2 and PDC-3 with bivalent binding to IL-13Ra2 revealed an approx. 5x stronger binding to the target positive CHO cells when compared to PDC-1 with monovalent binding to IL-13Ra2.
*very weak binding signal
Example 8: In vitro PDC growth assays
The ability of the IL-13Ra2 specific PDCs from Example 7 to inhibit growth of the cancer cell lines A-375 cells and A-549 cells was assessed in in vitro growth assays.
Method
On the day of the assay, cells were washed once with PBS, prior to being treated with Accutase (Life Technologies) at 37°C (until cells were detached) and suspended in the recommended culture medium. Cells were then counted and seeded in 96 well flat bottom plates (e.g. Costar or Corning) at densities of 200-400 cells/well in 120 pl of cell culture medium. PDCs were serially diluted in duplicate to a 5x stock solution in the appropriate cell culture medium and 30 pl/well of 5x serially diluted PDCs were added to the cells prior to incubation in a tissue culture incubator at 37°C with 5% CO2 for 5 days. Following this incubation period, relative cell viability was determined via the addition of 16 pl/well of PrestoBlue (Invitrogen). Plates were then incubated in a tissue culture incubator at 37°C with 5% CO2 for 2 hours . The resulting luminescence intensity was measured using an appropriate plate reader.
Results
Results are summarized in Table 20 and shown in Figure 3. All tested PDCs mediated potent and dose dependent cell killing of the IL-13Ra2 positive cancer cell line A-375 with IC50 values down to 12 pM. Killing of the IL-13Ra2 negative cancer cell line A-375 revealed an assay window of up to 40.000 fold for PDC-2.
In sum, the identified IL-13Ra2 specific HTH polypeptides of the present disclosure are well suited to deliver therapeutic agents into targeted cells expressing IL-13Ra2.
Example 9: In vitro PDC growth assays in presence of IL-13 (competition assays)
In order to determine the ability of IL-13 to compete with the IL13Ra2 specific polypeptides of the present disclosure for binding to IL13Ra2, the in vitro growth assay of Example 8 was performed in the presence of varying concentrations of human IL-13. The competition assays were basically performed as described in Example 6 with the difference that serially diluted PDC-1 and PDC-3 were pre-incubated with varying concentration of recombinant human IL- 13 (diluted in culture media) before being added to the cells.
Results:
Results are summarized in Table 21 and shown in Figure 4. Presence of human IL-13 clearly inhibited the anti-proliferative effects of the tested PDCs on A-375 cells in a dose dependent manner, indicating that the HTH polypeptides of the present invention compete with IL-13 for binding to IL-13Ra2. However, unlike natural IL-13, the HTH polypeptides does not bind to IL- 13Ra1 and as such does not interfere with IL-13 mediated signaling over the IL-13Ra1/IL-4Ra receptor complex.
Notably, despite the reported high affinity binding of human IL-13 to human IL-13Ra2, IL13 concentrations of up to 50 pM did not significantly altered the inhibitory activity of the tested PDC constructs. With IL-13 serum levels in tumor patients are in the range of ~ 1 pg/ml (-0.077 pM) [PMID 24833143], it can be deduced that the HTH polypeptides of the present disclosure won’t be displaced by the ~1000x fold lower serum concentration of IL-13 present in human cancer patients.
Example 10: Design and production of bispecific Fab-HTH polypeptide fusion molecules with specificity for CD3 and IL-13Roc2
Therapeutic delivery systems, which not only specifically recognize cancer cells but also enable the recruitment of immune cells, such as cytotoxic T-cells to their respective site of action were designed. This approach enables to kill such tumor cells by e.g. activated T-cells.
For generation of bispecific antibody-HTH polypeptide fusion molecules specifically recognizing CD3 and IL-13Ra2, several formats were designed. Two tested formats are depicted in Figure 6.
Construct 1 : a Fab-HTH format (Figure 6A) comprising one monoclonal Fab fragment specific for CD3 epsilon fused to one HTH polypeptide specific for IL-13Ra2. In this construct, the N- terminus of the HTH polypeptide is fused to the C-terminus of the Fab heavy chain via a glycine-serine tether.
Construct 2: a Fab-HTH-HTH format (Figure 6B) comprising one monoclonal Fab fragment specific for CD3 epsilon fused to two HTH-polypeptides specific for IL-13Ra2. In this construct, a first HTH polypeptide is fused with its N-terminus to the C-terminus of the Fab heavy chain via a peptide tether and a second HTH-polypeptide is fused via its N-terminus to the C-terminus of the first HTH polypeptide with a peptide tether.
For generation of the Fab-HTH or Fab-HTH-HTH fusion polypeptides, the DNA encoding the entire designed Fab-heavy chain constant region genetically fused to the HTH-polypeptide
were synthesized as double-stranded DNA fragments. All synthesized DNA fragments were cloned into suited mammalian human Fab expression vectors by replacing the parental Fab heavy chain constant region using standard molecular biology methods. For the Fab portion of the fusion molecules, the VH and VL domains of a CD3 specific monoclonal antibody disclosed in WO 2019/034580 were used. This CD3 specific antibody binds to the N-terminus of the epsilon chain of CD3.
For the HTH portion, the IL-13Ra2 specific HTH polypeptides, “HTH-71” (SEQ ID NO: 01) was used. As a negative control Fab, the VH and VL domains of the in-house negative isotype control antibody MQR03207 with specificity for hen-egg lysozyme were used. As a negative control HTH-polypeptide, “HTH-58” (SEQ ID NO: 149) comprising the HTH scaffold template sequence used for generation of the HTH-phage library disclosed in WO 2017/149117 was used.
Table 22: Overview of generated bispecific Fab-HTH - HTH polypeptide fusion molecules (Table discloses "(G4S)2" as SEQ ID NO: 190)
Table 23: Amino acid sequences of the generated bispecific antibody-HTH-polypeptide fusion molecules
For recombinant expression and purification, eukaryotic HKB11 or HEK 293 cells were transfected with pYMexlO eukaryotic expression vector DNA encoding both polypeptides chains of Fab-HTH polypeptide fusions. Cell culture supernatant was harvested on day 3 or day 6 post transfection and subjected to Capture select lgG-CH1 affinity chromatography (MabSelect SURE, GE Healthcare) for antibody-peptide purification. All samples were sterile filtered (0.2 pm pore size). Purity of Fab-HTH polypeptide fusions were analyzed under denaturing, reducing and non-reducing conditions using a Labchip System (Caliper GXII, Perkin Elmer) or on SDS-PAGE. Protein concentrations were determined by UV- spectrophotometry and HP-SEC was performed to analyze Fab-peptide fusion preparations in native state.
Production Results
Table 24 summarizes yields and final monomer content of 3 produced bispecific constructs. In general, the constructs could be generated by the described production and purification method with yields between 24 - 40 mg/L and final monomer content between 95 - 99 %.
Example 11: Binding of bispecific Fab-HTH polypeptide fusion molecules to IL13-Ra2 expressed on cells
For the assessment of IL-13Ra2 binding of the bispecific fusion molecules, A-375 (ATCC® CRL-1619™) cells, Flp-ln CHO cells engineered to express full-length cynomolgus IL-13Ra2 as well as HEK-293 cells engineered to express full-length mouse IL-13Ra2 were used. In
addition, the human CD3 positive T cell leukemia cell line, Jurkat (ATCC #TIB-152) was used to confirm binding of the Fab to CD3 (data not shown).
Method:
Cells were resuspended and counted in wash buffer (DPBS with calcium and magnesium (Gibco, #14040174) supplemented with 3% FBS and 0.02% sodium acid). 6x 104 cells per well were seeded in 384 well V-bottom plates (Greiner bio-one, #781280) and incubated with serially diluted constructs for 1 h on ice. Cells were washed 2 times in wash buffer. Bound constructs were detected using Alexa Fluor 647-conjugated detection antibody directed against human F(ab’)2 fragment (Jackson Immuno Research, #109-606-097). Staining was measured using IntelliCyt iQue flow cytometer and analyzed in or ForeCyt (version 4.1.5379, IntelliCyt) software. ECso values were calculated using 4-parameter non-linear regression analysis in Prism software (GraphPad Software Inc., version 5.04).
Results:
Results of the experiments are summarized in Table 25. As expected, bispecific Fab-HTH fusion molecules BIS-228, BIS-602 and BIS-659 with specificity for CD3 and IL-13Ra2 retained dose dependent binding to IL-13Ra2 positive cells, whereas the negative control fusion molecules BIS-648 and BIS-646 showed no binding to IL-13Ra2 positive cell. Fusion molecules BIS-779 with monovalent binding to IL-13Ra2 revealed weaker binding to IL-13Ra2 positive cells when compared to corresponding fusion molecules BIS-602 and BIS-659, with bivalent binding to IL-13Ra2. No apparent differences between BIS-602 and BIS-659 for binding to IL-13Ra2 positive cells was observable, confirming that the absence or presence of the C-terminal Glycine has no effect on target binding. All negative control constructs behaved as expected.
Table 25: Cell binding results of bispecific Fab-HTH polypeptide fusion molecules to IL13Ra2 expressed on cells
Example 12: T-cell mediated killing of cancer cells induced by bispecific Fab - HTH polypeptide fusion molecules
The produced bispecific Fab - HTH fusion molecules of Example 10 and 11 were analyzed for their potential to induce T-cell mediated killing of IL-13Ra2 expressing tumor cells in the presence of human T-cells or PBMCs.
Methods
Isolation of human PBMCs
Human whole blood from healthy donors was collected in Li-Heparin containing S-Monovette containers (Sarstedt). 20 mL blood were transferred to 50 ml conical tubes and diluted with an equal volume of PBS containing 2% fetal bovine serum (Sigma, #F7524) and 2 mM EDTA. Diluted blood was transferred to SepMate-50 tubes (StemCell Technologies, #86450) containing 15 ml Biocoll solution (Biochrom, #L6115) and centrifuged for 10 min at 1200 x g. Supernatant was transferred into a 50 ml conical tube, diluted to 45 ml with PBS containing 2% fetal bovine serum and 2 mM EDTA, and centrifuged for 8 min at 300 x g. The cell pellet was resuspended in 1 mL PBS containing 2 % fetal bovine serum and diluted to 20 ml, and
centrifuged for 10 min at 120 x g. The last step was repeated twice, the cell pellet was resuspended in assay medium, and diluted to 20 ml. Cells were counted using the CASY TT device (Beckmann Coulter).
Isolation of human T-cells
Human whole blood from healthy donors was collected in Li-Heparin containing S-Monovette containers (Sarstedt). 20 mL blood were transferred to 50 ml conical tubes, mixed with 1 ml of RosetteSep Human CD8+ Enrichment Cocktail (Stemcell Technologies, #15063) and incubated for 20 min at room temperature. Blood containing RosetteSep human CD8+ enrichment cocktail was diluted with an equal volume of PBS containing 2% fetal bovine serum (Sigma, #F7524) and 2 mM EDTA. Diluted blood was transferred to SepMate-50 tubes (StemCell Technologies, #15063) containing 15 ml of Lymphoprep density gradient medium (Stemcell Technologies, #07811) and centrifuged for 20 min at 1200 x g at room temperature. Supernatant was transferred into a 50 ml conical tube, diluted to 45 ml with PBS containing 2% fetal bovine serum and 2 mM EDTA and centrifuged for 5 min at 800x g. Supernatant was discarded, cell pellet resuspended in 1 ml PBS containing 2 % fetal bovine serum. Cell suspensions were pooled and transferred to a 50 ml tube and diluted to 30ml PBS containing 2 % fetal bovine serum. Cells were pelleted by centrifugation for 5 min at 800 x g. The cell pellet was resuspended in 2 ml of 1x Pharm Lyse Red Blood Cell lysing buffer (BD, #555899) and incubated at 4°C for 10 min. PBS containing 2 % fetal bovine serum was added to a final volume of 15 ml. Cells were pelleted for 10 min at 120 x g and the supernatant decanted. The cells were washed twice with PBS containing 2 % fetal bovine serum and counted (CASY TT device, Beckmann Coulter).
Assay Method
5,000 IL-13Ra2 expressing A-375 cells or IL-13Ra2 negative A- 549 cells were suspended in culture medium supplemented with 10% FCS, seeded in black 96 well assay plates (Corning) and incubated over night at 37°C and 5% CO2. CellToxGreen dye (Promega, #G8731), serially diluted bispecific Fab-HTH fusions molecules (final concentration: 0.00001 - 100 nM) and 100,000 purified T-cells or PBMCs, all diluted in assay medium comprising RPMI 1640 w/o Phenol red (Gibco, #32404-014), GlutaMAX and 10% fetal bovine serum, were added to the cells and incubated for 48 h at 37°C and 5% CO2. Cytotoxic activity was assessed by measuring incorporated CellToxGreen fluorescence at 485 nm excitation and 535 nm emission using a Tecan Infinite F500 device.
Results
The results of the experiments are shown in Figure 5 and summarized in Table 26.
Co-cultivation of T-cells or PBMCs with bispecific Fab-HTH polypeptide fusion molecules with specificity for CD3 and IL-13Ra2 induced killing of IL-13Ra2 positive A-375 target cells in a dose dependent manner. In the presence of IL-13Ra2 negative A-549 cells, none of the tested fusion molecules induced cytotoxic activity. As expected, the negative control Fab-HTH fusions BIS-127 and BIS-648 induced no killing activity using either cancer cell line. These results clearly demonstrates that the internalization properties of HTH polypeptides of the present disclosure are not dimetral for the killing activities of the Fab - HTH fusion, which requires the presence of the fusion molecule on the cell surface.
Accordingly, the HTH polypeptides of the present disclosure can be used in various approaches to efficiently target IL-13Ra2 expressing cells.
Claims
1. An isolated polypeptide specific for IL- 13 receptor alpha 2 (IL-13Ra2) comprising a helix- turn-helix scaffold structure of the formula Helix-1 - Li - Helix-2, wherein Li is a linker and wherein Helix-1 and Helix- 2 comprise a first and second a-helical peptide, wherein each of said a-helical peptides comprises the amino acid sequence
D - E - L - Vari -X1 - L - Var2 - Var3 - X2 - L - Var4 - X3 - L - Var5 - Var6 (Helix-1 disclosed as SEQ ID NO: 197; Helix-2 disclosed as SEQ ID NO: 204), wherein,
Vari is any natural occurring amino acid excluding G, P and C in Helix 1 and Helix 2,
Var2 is K, R, T or I in Helixl and I or K in Helix-2,
Var3 is R or Q in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
Var4 is Q or K in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
Var5 is F, Y or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
Var6 is K, R or H in Helix-1 and any natural occurring amino acid excluding G, P and C in Helix-2,
X1 is Q in Helix-1 and A in Helix-2,
X2 is E in Helix-1 and K in Helix-2, and
X3 is K in Helix-1 and M or A in Helix-2.
2. The isolated polypeptide according to claim 1 , wherein
Vari is A, T, H or K in Helix-1 and A, R, E or M in Helix-2,
Var3 is A, R, E or Q in Helix-2,
Var4 is A, H, D, I, or E in Helix-2,
Var5 is A, E, I, L, T, K or D in Helix-2, and
Var6 is A, Q, R or H in Helix-2
3. The isolated polypeptide according to claim 1 or claim 2, wherein said first and said second a-helical peptide form an antiparallel coiled-coiled structure.
4. The isolated polypeptide according to any one of the preceding claims, wherein the isolated polypeptide is cross-reactive to mouse and cynomolgus IL-13Ra2.
5. The isolated polypeptide according to any one of the preceding claims, wherein the isolated polypeptide competes with IL-13 for binding to IL-13Ra2.
6. The isolated polypeptide according to any one of the preceding claims, wherein the polypeptide does not bind to IL-13 receptor alpha 1 (IL-13Ra1), IL-4 receptor alpha (IL- 4Ra) or to the IL-13Ra1/ IL-4Ra complex.
7. The isolated polypeptide according to any one of the preceding claims, wherein the first a- helical peptide comprises an amino sequence selected from the group consisting of: DELTQLKRELQKLYR (SEQ ID NO: 1), DELAQLKRELQKLYR (SEQ ID NO: 2),
DELTQLKRELQKLYK (SEQ ID NO: 3), DELTQLKRELQKLFR (SEQ ID NO: 4),
DELHQLRRELQKLYR (SEQ ID NO: 5), DELKQLRRELQKLYH (SEQ ID NO: 6),
DELTQLRRELQKLHR (SEQ ID NO: 7), DELTQLTRELKKLYR (SEQ ID NO: 8), and
DELKQLIQELKKLYR (SEQ ID NO: 9).
8. The isolated polypeptide according to any one of the preceding claims, wherein the second a-helical peptide comprises the amino sequence selected from the group consisting of DELRALIRKLHMLEQ (SEQ ID NO: 19), DELRALIRKLHALEQ (SEQ ID NO: 20),
DELAALIRKLHMLEQ (SEQ ID NO: 21), DELRALIAKLHMLEQ (SEQ ID NO: 22),
DELRALIRKLAMLEQ (SEQ ID NO: 23), DELRALIRKLHMLAQ (SEQ ID NO: 24), DELRALIRKLHMLEA (SEQ ID NO: 25), DELRALIRKLDMLIR (SEQ ID NO: 26),
DELEALKRKLDMLLH (SEQ ID NO: 27), DELMALIEKLIMLTH (SEQ ID NO: 28),
DELRALIRKLHMLEH (SEQ ID NO: 29), DELRALIRKLEMLKR (SEQ ID NO: 30), and DELRALIQKLDMLDR (SEQ ID NO: 31).
9. The isolated polypeptide according to any one of the preceding claims, wherein the linker Li comprises 1 to 30 amino acid residues.
10. The isolated polypeptide according to any one of the preceding claims, wherein the linker Li comprises the amino acid sequence QGVDS (SEQ ID NO: 182) or QGVGS (SEQ ID NO: 183).
11 . The isolated polypeptide according to any one of the preceding claims, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 47 - 122.
12. An isolated polypeptide according to claims 1 - 10, wherein said polypeptide comprises an amino acid sequence selected from the group consisting of:
DELTQLKRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 47), DELTQLKRELQKLYRQGVDSDELRALIRKLHALEQ (SEQ ID NO: 49), DELTQLKRELQKLYRQGVDSDELRALIRKLDMLIR (SEQ ID NO: 58), DELTQLKRELQKLYRQGVDSDELEALKRKLDMLLH (SEQ ID NO: 59), DELHQLRRELQKLYRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 60), DELKQLRRELQKLYHQGVDSDELMALI EKLIM LTH (SEQ ID NO: 61), DELTQLRRELQKLHRQGVDSDELRALIRKLHMLEH (SEQ ID NO: 62), DELTQLRRELQKLHRQGVDSDELRALIRKLHMLEQ (SEQ ID NO: 63), DELTQLTRELKKLYRQGVDSDELRALIRKLEMLKR (SEQ ID NO: 64), and DELKQLIQELKKLYRQGVDSDELRALIQKLDMLDR (SEQ ID NO: 65).
13. The isolated polypeptide according to any one of the preceding claims, wherein a G is present at the N- and/or C-terminus of the helix-turn-helix scaffold structure.
14. The isolated polypeptide according to any one of the preceding claims, wherein said polypeptide is internalized from the surface of a cell expressing IL-13Ra2.
15. The isolated polypeptide according to claim 14, wherein said internalization occurs upon binding of said polypeptide to IL-13Ra2 present of the surface of said cell.
16. A fusion molecule comprising the isolated polypeptide specific for IL-13Ra2 according to any one of the preceding claims and at least one additional moiety.
17. The fusion molecule according to claim 16, wherein said at least one additional moiety is selected from the group consisting of a therapeutic agent, a cytotoxic agent, a bacterial toxin, a cytokine, an antibody or antibody fragment, a peptide, a polypeptide, a reporter enzyme, a detectable group, a moiety being capable of binding a metal ion, a tag suitable for detection and/or purification, a targeting ligands, a homo- or hetero-association domain, a moiety which increases solubility of a protein, or a moiety which comprises an enzymatic cleavage site, a lipid, a liposome and a virus-like-particle.
18. The fusion molecule according to claim 16 or claim 17, wherein said at least additional moiety is a radionuclide, a chemotherapeutic agent, a cytotoxic agent or a detectable group.
19. The fusion molecule according to claim 16 or claim 17, wherein said at least additional moiety is an antibody or antibody fragment.
20. The fusion molecule according to claim 19, wherein said at least additional moiety is an antibody or antibody fragment specific for CD3.
21. The fusion molecule according to claim 20, wherein said fusion molecule is capable of redirecting cytotoxic activity of a T-cell to an IL-13Ra2 expressing cell.
22. The fusion molecule according to claims 16 - 21 , wherein said fusion molecule is capable of mediating killing of IL-13Ra2 expressing cells.
23. The fusion molecule according to claim 16 - 20, wherein said at least additional moiety is fused to the isolated polypeptide specific for IL-13Ra2 via a peptide tether.
24. The fusion molecule according to claim 19, wherein said antibody or antibody fragment specific for CD3 comprises a VH comprising the amino acid sequence of SEQ ID NO: 180 and a VL comprising the amino acid sequence of SEQ ID NO: 179.
25. The isolated polypeptide according to claims 1 - 15 or the fusion molecule according to claims 16 - 24 for use in medicine.
26. A nucleic acid composition comprising a nucleic acid sequence or a plurality of nucleic acid sequences encoding the isolated polypeptide according to claims 1 - 15 or the fusion molecule according to claims 16 - 24.
27. A vector composition comprising a vector or a plurality of vectors comprising the nucleic acid composition of claim 26.
28. A host cell comprising the vector composition of claim 27 or the nucleic acid composition of claim 26.
29. A pharmaceutical composition comprising the isolated polypeptide according to claims 1 - 15 or the fusion molecule according claims 16 - 24 and a pharmaceutically acceptable carrier or excipient.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23174016.8 | 2023-05-17 | ||
| EP23174016 | 2023-05-17 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024236167A1 true WO2024236167A1 (en) | 2024-11-21 |
Family
ID=86387316
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/063669 Pending WO2024236167A1 (en) | 2023-05-17 | 2024-05-17 | Il-13 receptor alpha 2 binding polypeptides |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2024236167A1 (en) |
Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1993021232A1 (en) | 1992-04-10 | 1993-10-28 | Research Development Foundation | IMMUNOTOXINS DIRECTED AGAINST c-erbB-2 (HER-2/neu) RELATED SURFACE ANTIGENS |
| WO1994011026A2 (en) | 1992-11-13 | 1994-05-26 | Idec Pharmaceuticals Corporation | Therapeutic application of chimeric and radiolabeled antibodies to human b lymphocyte restricted differentiation antigen for treatment of b cell lymphoma |
| WO1996029417A1 (en) | 1995-03-15 | 1996-09-26 | The Government Of The United States Of America, Represented By The Secretary, Department Of Health And Human Services | Il-13 receptor specific chimeric proteins and uses thereof |
| US5641870A (en) | 1995-04-20 | 1997-06-24 | Genentech, Inc. | Low pH hydrophobic interaction chromatography for antibody purification |
| WO1999051643A1 (en) | 1998-04-03 | 1999-10-14 | The Penn State Research Foundation | Mutagenized il13-based chimeric molecules |
| WO2002018422A1 (en) | 2000-08-30 | 2002-03-07 | The Penn State Research Foundation | Amino acid substitution mutants of interleukin 13 |
| US6703199B1 (en) | 1997-06-12 | 2004-03-09 | Research Corporation Technologies, Inc. | Artificial antibody polypeptides |
| US6949245B1 (en) | 1999-06-25 | 2005-09-27 | Genentech, Inc. | Humanized anti-ErbB2 antibodies and treatment with anti-ErbB2 antibodies |
| WO2010121125A1 (en) | 2009-04-17 | 2010-10-21 | Wake Forest University Health Sciences | Il-13 receptor binding peptides |
| WO2013112871A1 (en) | 2012-01-27 | 2013-08-01 | The Board Of Trustees Of The Leland Stanford Junior University | Therapeutic il-13 polypeptides |
| WO2017149117A1 (en) | 2016-03-04 | 2017-09-08 | Morphosys Ag | Polypeptide library |
| WO2019034580A1 (en) | 2017-08-14 | 2019-02-21 | Morphosys Ag | Humanized antibodies for cd3 |
| WO2019086676A1 (en) | 2017-11-03 | 2019-05-09 | Consejo Superior De Investigaciones Científicas | IL13Rα2 PEPTIDE AND ITS USES |
-
2024
- 2024-05-17 WO PCT/EP2024/063669 patent/WO2024236167A1/en active Pending
Patent Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1993021232A1 (en) | 1992-04-10 | 1993-10-28 | Research Development Foundation | IMMUNOTOXINS DIRECTED AGAINST c-erbB-2 (HER-2/neu) RELATED SURFACE ANTIGENS |
| WO1994011026A2 (en) | 1992-11-13 | 1994-05-26 | Idec Pharmaceuticals Corporation | Therapeutic application of chimeric and radiolabeled antibodies to human b lymphocyte restricted differentiation antigen for treatment of b cell lymphoma |
| WO1996029417A1 (en) | 1995-03-15 | 1996-09-26 | The Government Of The United States Of America, Represented By The Secretary, Department Of Health And Human Services | Il-13 receptor specific chimeric proteins and uses thereof |
| US5641870A (en) | 1995-04-20 | 1997-06-24 | Genentech, Inc. | Low pH hydrophobic interaction chromatography for antibody purification |
| US6703199B1 (en) | 1997-06-12 | 2004-03-09 | Research Corporation Technologies, Inc. | Artificial antibody polypeptides |
| WO1999051643A1 (en) | 1998-04-03 | 1999-10-14 | The Penn State Research Foundation | Mutagenized il13-based chimeric molecules |
| US6949245B1 (en) | 1999-06-25 | 2005-09-27 | Genentech, Inc. | Humanized anti-ErbB2 antibodies and treatment with anti-ErbB2 antibodies |
| WO2002018422A1 (en) | 2000-08-30 | 2002-03-07 | The Penn State Research Foundation | Amino acid substitution mutants of interleukin 13 |
| WO2010121125A1 (en) | 2009-04-17 | 2010-10-21 | Wake Forest University Health Sciences | Il-13 receptor binding peptides |
| WO2013112871A1 (en) | 2012-01-27 | 2013-08-01 | The Board Of Trustees Of The Leland Stanford Junior University | Therapeutic il-13 polypeptides |
| WO2017149117A1 (en) | 2016-03-04 | 2017-09-08 | Morphosys Ag | Polypeptide library |
| WO2019034580A1 (en) | 2017-08-14 | 2019-02-21 | Morphosys Ag | Humanized antibodies for cd3 |
| WO2019086676A1 (en) | 2017-11-03 | 2019-05-09 | Consejo Superior De Investigaciones Científicas | IL13Rα2 PEPTIDE AND ITS USES |
Non-Patent Citations (27)
| Title |
|---|
| "Current Protocol in Molecular Biology", 1989, GREENE PUBLISHING ASSOC. |
| "Uniprot", Database accession no. P35225 |
| BIRD ET AL., SCIENCE, vol. 242, 1988, pages 423 - 426 |
| CHAKRAVARTI, A. ET AL., JOURNAL OF THE AMERICAN SOCIETY OF CLINICAL ONCOLOGY, vol. 20, 2002, pages 1063 - 1068 |
| CHARI ET AL., CANCER RES., vol. 52, 1992, pages 127 - 131 |
| CHOI, B.D., PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA, vol. 110, 2013, pages 270 - 275 |
| COHENPARRY, PROTEINS, vol. 7, 1990, pages 1 - 15 |
| HARBURY ET AL., SCIENCE, vol. 282, 1998, pages 1462 - 1467 |
| HOLLINGERHUDSON, NATURE BIOTECHNOLOGY, vol. 23, 2005, pages 1126 - 1136 |
| HUSTON ET AL., PROC. NATL. ACAD. SCI., vol. 85, 1988, pages 5879 - 5883 |
| KOHNHODGES, TRENDS BIOTECHNOL, vol. 16, 1998, pages 379 - 389 |
| KUNWAR S. ET AL., NEURO ONCOL, vol. 12, 2010, pages 871 - 81 |
| LAPORTE ET AL., CELL, vol. 132, no. 2, 28 January 2008 (2008-01-28), pages 259 - 72 |
| LUPASGRUBER, ADV PROTEIN CHEM, vol. 70, 2005, pages 79 - 112 |
| MASONARNDT, CHEM BIO CHEM, vol. 5, 2004, pages 170 - 176 |
| MCFARLANE ET AL., EUR J PHARMACOL, vol. 625, 2009, pages 101 - 107 |
| PARRY ET AL., J STRUCT BIOL, vol. 163, 2008, pages 258 - 269 |
| PURI RK ET AL., BLOOD, vol. 87, 1996, pages 4333 - 9 |
| SCHNEIDER ET AL., FOLD DES, vol. 3, 1998, pages R29 - R40 |
| STUPP, R. ET AL., N ENGL J MED, vol. 352, 2005, pages 987 - 996 |
| UEMATSU, M. ET AL., JOURNAL OF NEURO-ONCOLOGY, vol. 72, 2005, pages 231 - 238 |
| ULASOV, I. V. ET AL., BRITISH JOURNAL OF CANCER, vol. 100, 2009, pages 1154 - 1164 |
| VAN HOUDT, W. J. ET AL., J NEUROSURG, vol. 104, 2006, pages 583 - 592 |
| WARD ET AL., NATURE, vol. 341, 1989, pages 544 - 546 |
| ZAPATA ET AL., PROTEIN ENG., vol. 8, 1995, pages 1057 - 1062 |
| ZHENG, S. ET AL., JOURNAL OF GENE MEDICINE, vol. 9, 2007, pages 151 - 160 |
| ZWICK, M. B.MENENDEZ, A.BONNYCASTLE, L. L. C.SCOTT, J.K: "Phage Display: A Laboratory Manual", 2001, COLD SPRING HARBOR LABORATORY PRESS |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7779944B2 (en) | D domain-containing polypeptides and uses thereof | |
| CA2796010C (en) | Trail r2-specific multimeric scaffolds | |
| JP6385357B2 (en) | Heterologous dimer of antibody heavy chain constant region, CH3 domain mutant pair that induces high-efficiency formation, production method and use thereof | |
| KR102238326B1 (en) | Functional TNF receptor binding agents | |
| JP6708635B2 (en) | Bispecific antibodies to CD3ε and ROR1 | |
| JP7482488B2 (en) | A novel fusion protein specific for CD137 and PD-L1 | |
| AU2021364387A1 (en) | Fusions with cd8 antigen binding molecules for modulating immune cell function | |
| JP2017504328A (en) | Bispecific CD3 and CD19 antigen binding constructs | |
| JP2020511997A (en) | Tumor antigen presentation inducer construct and use thereof | |
| TW201201840A (en) | Antibodies specifically binding to human TSLPR and methods of use | |
| US11464803B2 (en) | D-domain containing polypeptides and uses thereof | |
| CN118076638A (en) | Novel anti-SIRPA antibodies | |
| KR20230098334A (en) | Polypeptide constructs that selectively bind to CLDN6 and CD3 | |
| CN113461820B (en) | anti-CD 3 humanized antibodies | |
| KR20230098335A (en) | Antigen binding domains with reduced clipping ratio | |
| KR20230104229A (en) | Polypeptide constructs that bind CD3 | |
| WO2024236167A1 (en) | Il-13 receptor alpha 2 binding polypeptides | |
| CN114144429A (en) | Bispecific antibodies against PD-1 and LAG-3 | |
| CA3254085A1 (en) | Klrb1 binding agents and methods of use thereof | |
| WO2022212836A1 (en) | Gpnmb antibodies and methods of use | |
| US20210253741A1 (en) | Bispecific antibodies | |
| HK40091554B (en) | Improved anti-oxmif antibodies with reduced aggregation potential and reduced hydrophobicity | |
| WO2022046997A1 (en) | Bama-binding agents and methods of use thereof | |
| CN120530142A (en) | Multispecific peptide complexes | |
| HK40091554A (en) | Improved anti-oxmif antibodies with reduced aggregation potential and reduced hydrophobicity |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24728164 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |