WO2024226471A2 - Compositions and methods for treating stxbp1 disorders - Google Patents
Compositions and methods for treating stxbp1 disorders Download PDFInfo
- Publication number
- WO2024226471A2 WO2024226471A2 PCT/US2024/025787 US2024025787W WO2024226471A2 WO 2024226471 A2 WO2024226471 A2 WO 2024226471A2 US 2024025787 W US2024025787 W US 2024025787W WO 2024226471 A2 WO2024226471 A2 WO 2024226471A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- aso
- stxbp1
- seq
- another embodiment
- sugars
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/11—Antisense
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/31—Chemical structure of the backbone
- C12N2310/315—Phosphorothioates
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2320/00—Applications; Uses
- C12N2320/30—Special therapeutic applications
- C12N2320/33—Alteration of splicing
Definitions
- compositions comprising ASOs, and methods of using the ASOs and compositions for treating, preventing, or delaying the onset of an STXBP1 disorder or a disorder in which disease processes lower STXBP1 levels.
- STXBP1 Syntaxin binding protein 1
- STXBP1 is encoded by the STXBP1 gene (also referred to as Muncl8-1) and is involved in synaptic vesicle fusion with the neuronal cell membrane via its multiple interactions with the soluble N-ethylmaleimide sensitive factor adaptor protein receptors (SNARE) complex, and thus vital for neurotransmitter release (Rizo J et al. Annu. Rev. Biophys. 2015, 44, 339-67; Misura KM, et al Nature 2000, 404, 355-62; Dulubova 1, et al PNAS 2007. 104, 2697-702; Shen J. et al Cell 2006, 128. 183-95).
- SNARE soluble N-ethylmaleimide sensitive factor adaptor protein receptors
- the STXBP1 gene is located on chromosome 9q34. 1. Its association with disease was discovered in 2008 in studies of patients with Ohtahara Syndrome, a severe early onset epilepsy. In these studies, five patients with Ohtahara syndrome, a severe, early-onset epilepsy characterized by a suppression-burst pattern on EEG and severe psychomotor retardation, were described with a variety of mutations in the STXBP1 gene, including missense, frameshift, splice site, and nonsense mutations (Saitsu H. et al Nat Genet. 2008, 40(6), 782-8).
- Arg406His is the most common recurrent mutation. Missense mutations occur throughout the protein structure with a couple of minor hotspots (Xian J, et al, supra; Abramov D, et al J. Neurochem. 2021. 157, 165-78) and most missense mutations studied to date cause protein destabilization, aggregation, and degradation (Abramov D, et al, supra). Multiple mutations result in the formation of premature termination codons, which have been found early in the protein sequence (L36X) as well as quite late (W522X) (Stamberger H. et al, supra). Diseasecausing mutations associated with STXBP1 include missense, nonsense, frameshift, and splice-site mutations, as well as intragenic, whole gene, and multi-gene deletions.
- STXBP 1 -related disorders A recent estimate of the incidence for STXBP 1 -related disorders is approximately 1 :30,000 births (Lopez-Rivera JA, et al Brain 2020, 143, 1099-1105) and STXBP1 is the fifth most implicated gene associated with epileptic developmental disorders (Symonds JD et al Eur. J. Paediatr. Neurol. 2020, 24, 15-23), suggesting that these disorders are not as rare as once believed.
- STXBP 1 Autism Xian J, et al, supra.
- HPO Human Phenotype Ontology
- haploinsufficiency The primary mechanism of disorder formation is haploinsufficiency, as approximately 50%-60% of the reported mutations are either deletions, nonsense, frameshift, or splice site variants (Stamberger H, et al, supra,' Xian J, et al, supra). A subset of missense mutations studied appear to promote aggregation and may decrease wild type protein levels, thus potentially producing a dominant-negative effect (Guiberson NGL, et al Nature Comm. 2018, 9, 3986-4009). In animal models, haploinsufficiency has been demonstrated to recapitulate several patient phenotypes (Kovacevik J.
- Genotype-Phenotype relationships have been difficult to establish.
- 534 STXBP1 patients Xian J, et al, supra
- five genetic hotspots with recurrent variants were identified in more than 10 individuals but none were associated with a specific phenotypic feature, though there were several nominal associations.
- STXBP1 disorders including epilepsy, global delay, cognitive impairment (mild to profound), movement disorders, hypotonia and autism.
- ASOs Provided herein are ASOs, compositions containing the ASOs, and methods of treating or delaying an STXBP1 disorder using the ASOs.
- the ASOs provided herein are useful for modulating expression of STXBP1.
- the ASOs are useful for increasing expression of STXBP1 in a cell such as a neuronal cell.
- the ASOs hybridize with, bind to, or target an STXBP1 mRNA or pre-mRNA.
- the ASOs modulate splicing of a STXBP1 pre-mRNA.
- the ASOs induce exon skipping in STXBPl pre- mRNA.
- the ASOs hybridize with, bind to. or target a 5'- or 3'- untranslated region (“UTR”) in a STXBP1 mRNA or pre-mRNA.
- the STXBP1 mRNA or pre-mRNA is a wild type STXBP1 mRNA or pre-mRNA.
- the STXBP1 mRNA or pre-mRNA is a mutant STXBP1 mRNA or pre-mRNA, such as a mutant STXBP1 mRNA or pre-mRNA associated with an STXBP1 disorder.
- the ASOs provided herein are complementary to an STXBP1 mRNA or pre-mRNA. In another embodiment, the ASOs provided herein are reverse complementary’ to an STXBP1 mRNA or pre-mRNA.
- provided herein is a method of treating or delaying the onset of an STXBP1 disorder by administering to subject having an STXBP1 disorder an ASO provided herein.
- the STXBP1 disorder is epilepsy, global delay, cognitive impairment (mild to profound), a movement disorder, hypotonia or autism.
- the subject having an STXBP1 disorder is characterized by STXBP1 haploinsufficiency.
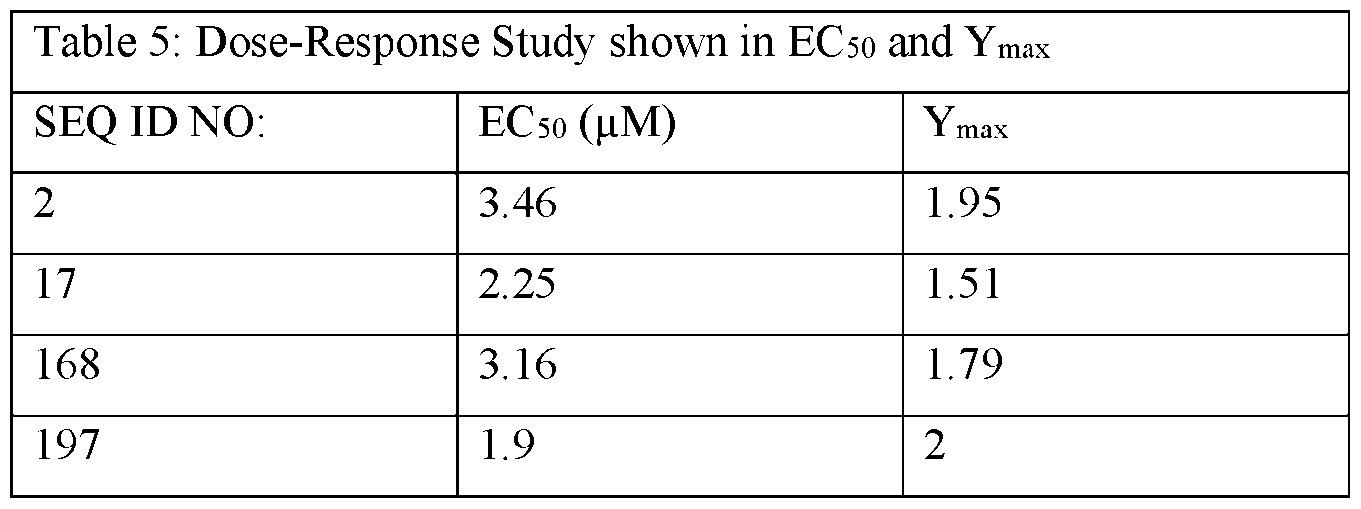
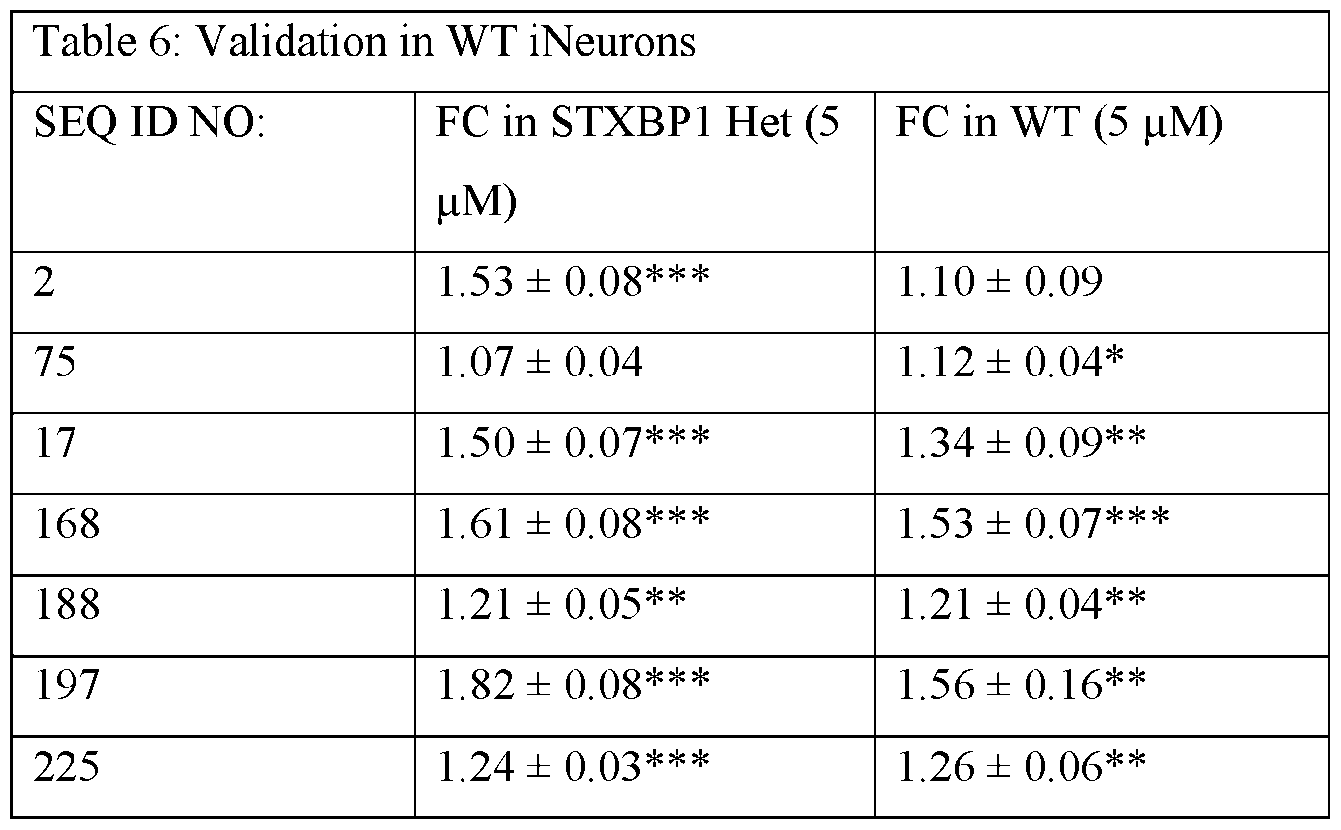
- FIGs. 1A-G show 6-point dose-response curves generated using STXBP1 Het iNeurons treated by gymnosis with ASOs provided herein.
- FIG. 1A SEQ ID NO: 2;
- FIG. IB SEQ ID NO: 17;
- FIG. 1C SEQ ID NO: 75;
- FIG. ID SEQ ID NO: 168;
- FIG. IE SEQ ID NO: 197;
- FIG. IF SEQ ID NO: 188;
- FIG. 1G SEQ ID NO: 225.
- FIGs. 2A-G show the effect of treatment with ASOs provided herein and controls for 17-days at a 5 pM dose on cell viability determined using a Cell-Titer Fluor Assay.
- FIG. 2A SEQ ID NO: 2;
- FIG. 2B SEQ ID NO: 75;
- FIG. 2C SEQ ID NO: 17;
- FIG. 2D SEQ ID NO: 168;
- FIG. 2E SEQ ID NO: 188;
- FIG. 2F SEQ ID NO: 197;
- FIG. 2G SEQ ID NO: 225
- FIG. 3 shows the immunostimulatory effects on cytokine production following treatment of PBMCs for 24 hours at 10 pM with ASOs provided herein and controls.
- FIGs. 4A-J show the results from orthogonal validation of ASOs provided herein and controls using ELISA using STXBP1 Het iNeurons along with each ASO’s NHP mismatches.
- FIG. 5 shows the results from an ELISA screen at 5 pM using non-HiBiT tagged STXBP1 Het iNeurons for ASOs provided herein and controls.
- FIG. 6 shows the level of synaptically-localized STXBP1 protein in STXBP1 +/- (HZ) Ngn2-induced neuron cultures, cultured without glia, after treatment with ASOs provided herein and controls.
- FIG. 7 shows the level of synaptically-localized STXBP1 protein in WT Ngn2- induced neuron cultures after treatment with ASOs provided herein and controls.
- FIG. 8 shows the level of synaptically-localized STXBP1 protein in STXBP1 +/- (HZ) Ngn2-induced neuron cultures, cultured with rat glia, after treatment with ASOs provided herein and controls.
- FIG. 9 shows the level of bulk STXBP1 protein in Ngn2-induced neuron cultures, cultured without glia, after treatment with ASOs provided herein and controls.
- subject is an animal, such as a mammal, including human, such as a patient.
- biological activity refers to the in vivo activities of an ASO or physiological responses that result upon in vivo administration of an ASO, composition or other mixture.
- Biological activity thus, encompasses therapeutic effects and pharmacokinetic behavior of such ASOs, compositions and mixtures.
- Biological activities can be observed in in vitro systems designed to test for such activities.
- the biological activity 7 of the ASO refers to modulating gene expression. Methods for detecting and/or quantifying changes in gene expression are known in the art, including but not limited to the methods disclosed in the Examples.
- oligonucleotide As used herein, when the word “oligonucleotide” is used it may be replaced by “antisense oligonucleotide” and vice versa unless otherwise indicated.
- the term “complementary” encompasses both forward complementary and reverse complementary sequences, as will be apparent to a skilled person from the context.
- an oligonucleotide is complementary, it is understood that it can also be reverse complementary.
- an oligonucleotide is complementary to’' a target sequence is used, this means that said oligonucleotide is reverse complementary to said target sequence as the sequence of the oligonucleotide is the reverse complement of the target sequence, unless otherwise stated.
- an antisense oligonucleotide is complementary to a target sequence
- the term “(reverse) complementarity” means a stretch of nucleic acids that can hybridize to another stretch of nucleic acids under physiological conditions.
- An antisense strand is generally said to be complementary' to a matching sense strand.
- an antisense oligonucleotide is complementary to its target.
- Hybridization conditions are defined herein. It is thus not absolutely required that all the bases in the region of complementarity 7 are capable of pairing with bases in the opposing strand. For instance, when designing an antisense oligonucleotide, one may want to incorporate for instance a residue that does not base pair with the base on the complementary strand.
- the ASOs provided herein have 1 or 2 mismatches with a target sequence, such as a target sequence within a STXBP1 mRNA or pre-mRNA.
- the term “binds to” can be replaced with “complementary to”, “hybridizes to”, “overlaps with” and/or “targets” when used in the context of an antisense oligonucleotide which is complementary to a part of a pre-mRNA as identified herein. In this disclosure, such terms are synonymous.
- “hybridizes” is used under physiological conditions in a cell. In one embodiment, the cell is a neuronal cell unless otherwise indicated.
- the ASOs provided herein may have one or more substitutions or modifications relative to ASOs made up of unmodified single stranded DNA or RNA oligonucleotides.
- a substitution replaces one chemical group, which might be hydrogen, by another chemical group.
- an RNA monomer is inherently 2'-substituted because it has a hydroxyl group at its 2'-position. A DNA monomer would therefore not be 2'-substituted, and an RNA monomer can be seen as a 2'-substituted DNA monomer.
- 2'-0-methyl RNA is a 2'-substituted monomer (-OMe substitutes -H) and a 2 -substituted RNA monomer (-OMe substitutes -OH) and a 2'-O-substituted RNA monomer (-Me substitutes -H), while 2'-F RNA is a 2'-substituted RNA monomer (-F substitutes -OH or -H) yet not a 2'-O-substituted RNA monomer (2'-0 is either no longer present, or is not substituted).
- 2'-F RNA where F is substituted for 2'-OH is a 2'-F-2'-deoxy RNA, which is also a 2'-F DNA.
- bicyclic nucleic acid may be replaced by conformationally restricted scaffold modification, locked scaffold modification, locked nucleotide, locked nucleoside, locked monomer, or Tm enhancing scaffold modification, or high-affinity modification and the like, as appropriate.
- sequence identity means a relationship between two or more nucleic acid (polynucleotide or nucleotide or oligonucleotide) sequences, as determined by comparing the sequences. In one embodiment, sequence identity is calculated based on the full length of two given SEQ ID NO or on part thereof. Part thereof means at least 50%, 60%, 70%, 80%, 90%, or 100% of both SEQ ID NO. As used herein, “identity” also means the degree of sequence relatedness between nucleic acid sequences, as the case may be, as determined by the match between strings of such sequences.
- Methods to determine identity are designed to give the largest match between the sequences tested. Methods to determine identity and similarity are codified in publicly available computer programs. Computer program methods to determine identity and similarity between two sequences include, e.g., the GCG program package (Devereux, J., et al., Nucleic Acids Research 12 (1): 387 (1984)), BestFit, BLASTN, and FASTA (Altschul, S. F. et al., J. Mol. Biol. 215:403-410 (1990)).
- the BLAST X program is publicly available from NCBI and other sources (BLAST Manual, Altschul, S., et al., NCBI NLM NIH Bethesda, MD 20894; Altschul, S., et al., J. Mol. Biol. 215:403-410 (1990)).
- the well-known Smith Waterman algorithm may also be used to determine identity.
- Hybridization conditions for a nucleic acid molecule may have low or medium or high stringency (southern blotting procedures).
- Low or medium or high stringency conditions means pre-hybridization and hybridization at 42 °C in 5x SSPE, 0.3% SDS, 200pg/ml sheared and denatured salmon sperm DNA, and either 25% or 35% or 50% formamide for low or medium or high stringencies respectively.
- the hybridization reaction is washed three times for 30 minutes each using 2x SSC, 0.2% SDS and either 55 °C or 65 °C, or 75 °C for low 7 or medium or high stringencies respectively.
- treatment means any manner in which one or more of the symptoms of a disease or disorder are ameliorated or otherwise beneficially altered. Treatment also encompasses any pharmaceutical use of the compositions herein, such as use for treating an STXBP1 disorder.
- amelioration of the symptoms of a particular disorder byadministration of a particular ASO or pharmaceutical composition refers to any lessening, whether permanent or temporary, lasting or transient that can be attributed to or associated with administration of the ASO or pharmaceutical composition.
- Certain ASOs provided herein possess asy mmetric atoms (optical centers) or double bonds; the racemates, diastereomers, tautomers, geometric isomers and individual isomers are encompassed within the scope of the present disclosure.
- the ASOs provided herein do not include those which are known in the art to be too unstable to synthesize and/or isolate.
- the ASOs provided herein may also contain unnatural proportions of atomic isotopes at one or more of the atoms that constitute such ASOs.
- the ASOs maybe radiolabeled with radioactive isotopes, such as for example tritium ( 3 H). iodine- 125 ( 125 I) or carbon- 14 ( 14 C). All isotopic variations of the ASOs provided herein, whether radioactive or not, are encompassed w ithin the scope of the present disclosure.
- an STXBP1 gene is well known in the art.
- the coding strand of the human STXBP1 gene spans -80,423 bp and is set forth in NCBI Accession No. NC 000009.12.
- Mutant STXBP1 genes are also w ell known in the art.
- the STXBP1 gene encodes two major splice variants: i) NM_003165 (isoform a), in which 19 consecutive exons are translated to generate a 68.7 kDa protein of 603 amino acids; and ii) NM_001032221 (isoform b), in which exon 19 is skipped, and a stop codon in exon 20 terminates translation to generate a 67.6 kDa protein of 594 amino acids.
- ASOs antisense oligonucleotides
- the ASOs provided herein are single stranded oligonucleotides.
- the ASOs provided herein contain fewer than 50 nucleotides.
- the ASOs provided herein comprise a base sequence that is complementary to a target sequence in an STXBP1 mRNA or pre-mRNA.
- the ASOs provided herein target or bind to a portion of a complementary nucleic acid molecule, such as a mRNA or pre-mRNA corresponding to the coding strand of the human STXBP1 gene set forth in NCBI Accession No. NC 000009 12.
- the ASOs provided herein target or bind to a portion of a complementary nucleic acid molecule, such as a mRNA or pre-mRNA corresponding to human STXBP1 gene splice variants NM_003165 or NM_001032221.
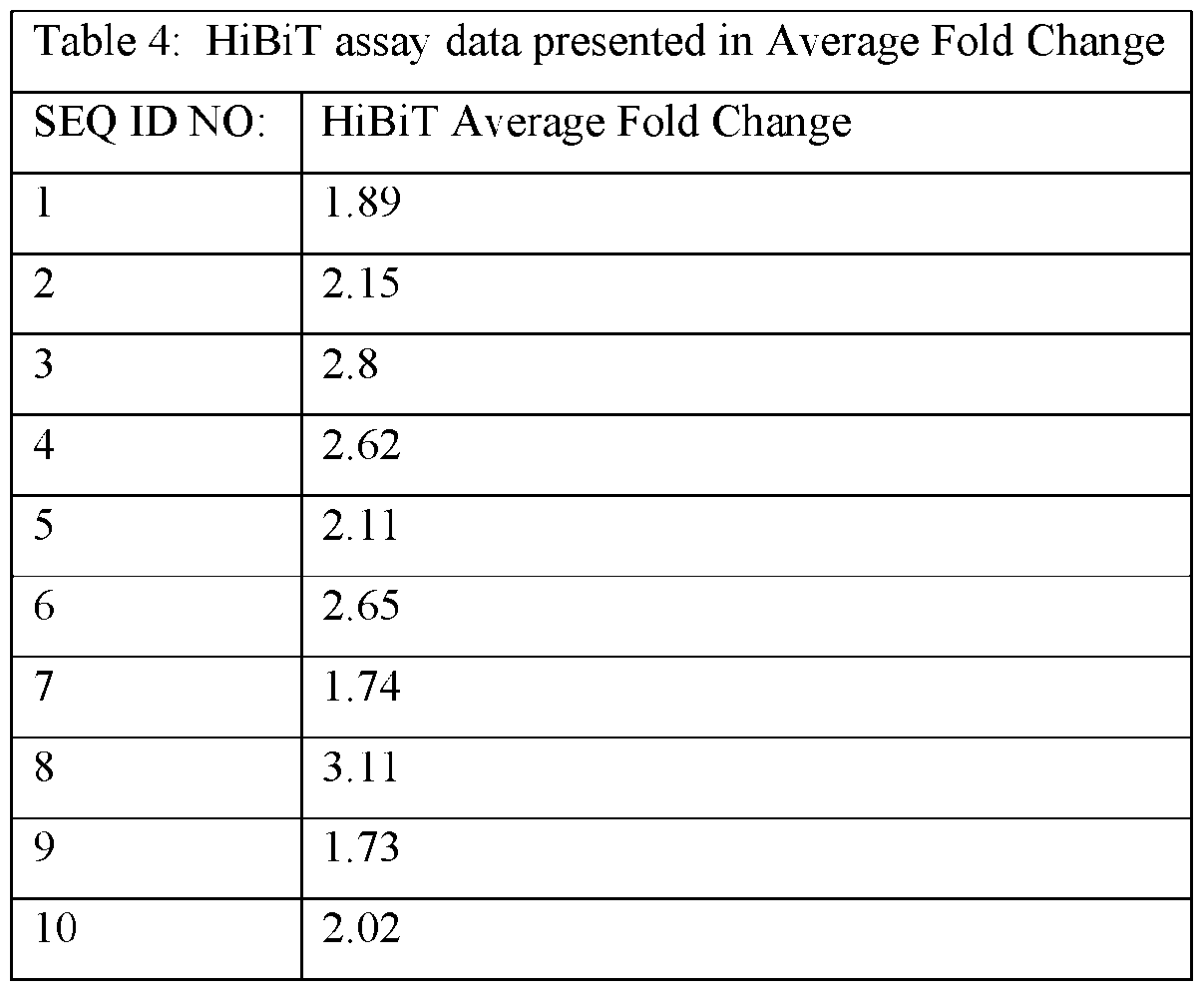
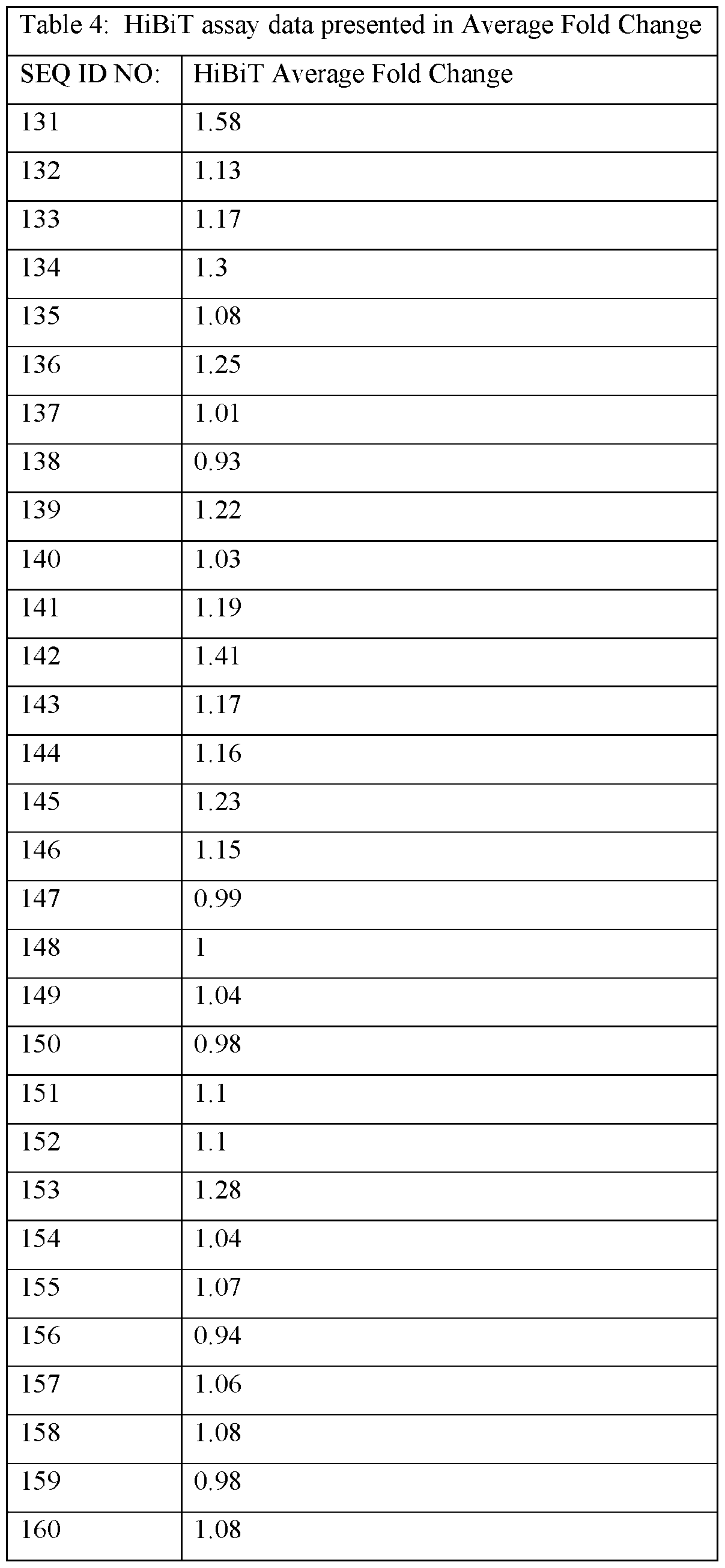
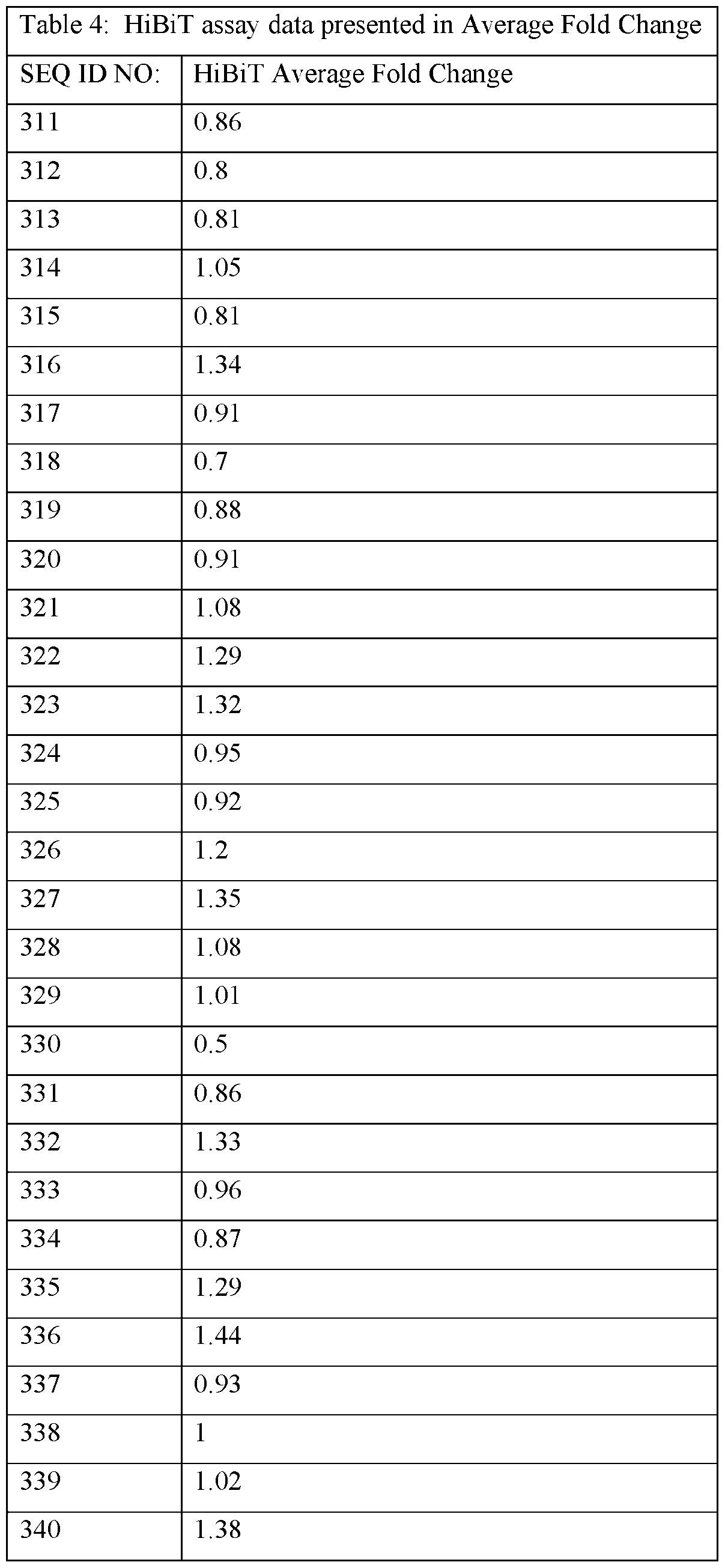
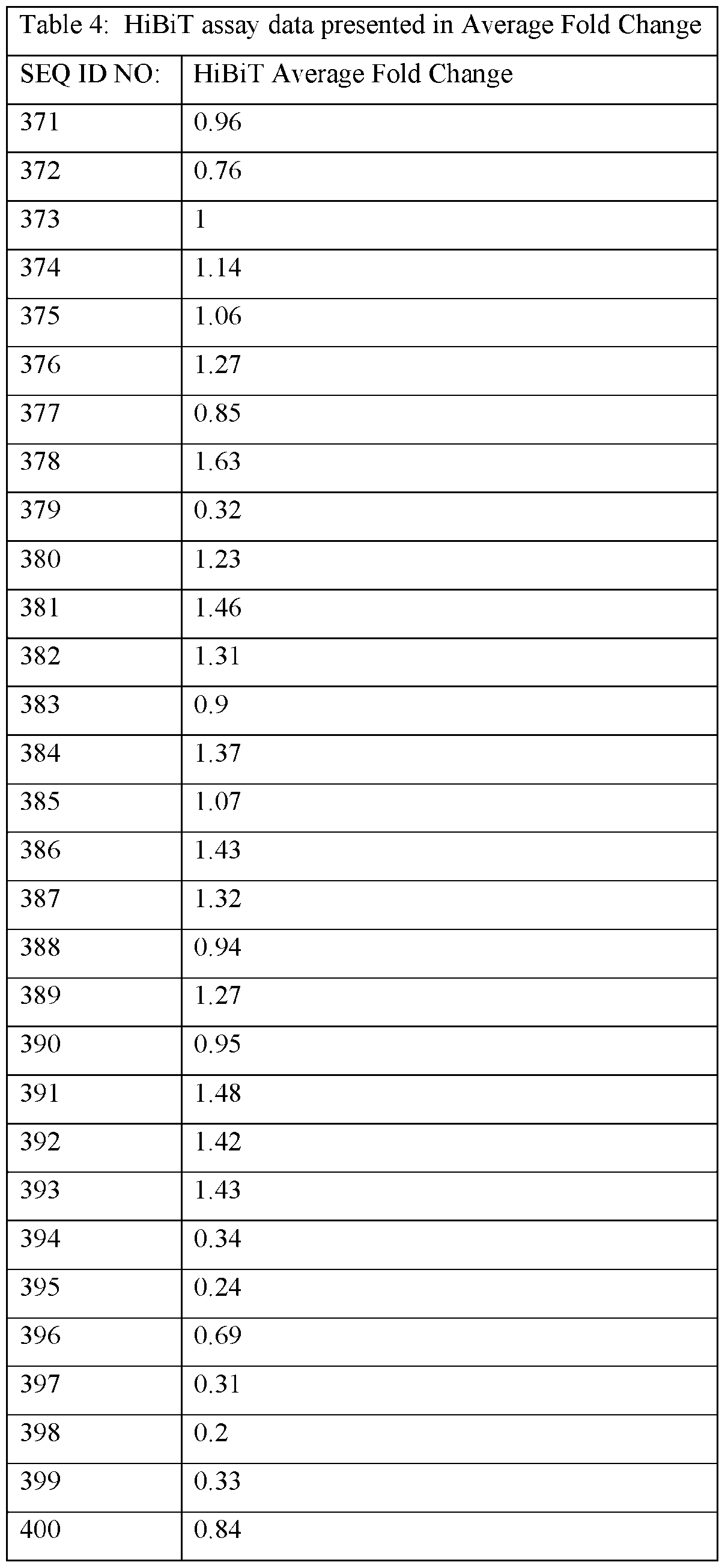
- the ASOs provided herein comprise a base sequence set forth in any one of SEQ ID NOs: 1-626.
- the base sequence consists of a sequence set forth in any one of SEQ ID NOs: 1-626.
- the ASO provided herein comprises a base sequence with at least 80%, 85%, 90% or 95% sequence identity to any one of SEQ ID NOs: 1-626 and when contacted with a cell increases expression of STXBP1 protein.
- the cell is a neuronal cell, optionally in vivo or in vitro, and the ASO provided herein increases a level of functional STXBP1 protein in the neuronal cell relative to a control cell not contacted with the ASO.
- the ASOs provided herein contain at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of one of the following sequences: CAGAGGCCAGCTGACTGC (SEQ ID NO: 1), TGTACTCACAGTCAGTG (SEQ ID NO: 2), AGGAGGCAGCTTCCCTG (SEQ ID NO: 3), ACTGACGCGCGGACTG (SEQ ID NO: 4), CTGCGCGAGTCTCCCG (SEQ ID NO: 5), TGCCAGCCAGGGCGTGCAGG (SEQ ID NO: 6), CGAGAATGCAGCGGCAACAG (SEQ ID NO: 7), AACAGTCCAGAAATTTCTCC (SEQ ID NO: 8), CCAAGCAATGTGCACGTCAC (SEQ ID NO: 9), CAAAGAGATGGAGGCTTCCA (SEQ ID NO: 10), CAGAGGCCAGCTGACTG (SEQ ID NO: 17), AGGAGGCAGCTTCCCT (SEQ ID NO: 75), CACGAGAATGCAGCGGCA (SEQ ID NO:
- SEQ ID NOs: 1-3, 17 and 75 target a splice site and/or an exon splicing enhancer (ESE) of a mutant STXBP1 pre-mRNA, and/or induce exon skipping or inclusion in a mutant STXBP1 pre-mRNA.
- ESE exon splicing enhancer
- SEQ ID NOs: 4 and 5 target a 5'-UTR of an STXBP1 mRNA.
- SEQ ID NOs: 6-10, 168, 188, 197 and 225 target a 3'-UTR of an STXBP1 mRNA.
- the ASOs provided herein are complementary or reverse complementary to their intended mRNA or pre-mRNA target.
- the terms complementarity and reverse complementarity are used herein to refer to a stretch of nucleic acids that can hybridize to another stretch of nucleic acids under physiological conditions in a cell. Such hybridization generally follows well-known A-T(U)/G-C base pairing.
- T also includes the corresponding ASO containing a U instead of the T.
- the ASOs provided herein are fully T containing ASOs.
- the ASOs provided herein are fully U containing ASOs.
- the ASOs provided herein contain a mixture of T and U bases.
- the ASOs provided herein are fully complementary or reverse complementary to their target. In another embodiment, the ASOs provided herein are less than fully complementary' or reverse complementary' to their target. In such embodiments, such “mismatches” are within the scope of this disclosure to the extent that the resulting ASO hybridizes to its target under physiological conditions in a cell. In certain embodiments, the degree of complementarity' or reverse complementarity of the ASO provided herein to its target is at least 85%, 90%, 95%, 96%, 97%, 98%, 99% or is 100%.
- the ASOs specifically disclosed herein, represented by SEQ ID NOs are 100% reverse complementary to their target.
- the ASO provided herein has a length of less than 50 nucleotides, or from 8-40, 10-33 or 15-25 nucleotides. In another embodiment, the ASO provided herein has a length of 12-30 nucleotides. In another embodiment, the ASO provided herein has a length of 16-25 nucleotides. In another embodiment, the ASO provided herein has a length of 16-20 nucleotides. In another embodiment, the ASO provided herein has a length of 16, 17, 18, 19 or 20 nucleotides.
- the ASO provided herein has one or more modified bases, one or more modified sugars, and/or one or more modified intemucleoside linkages. In another embodiment, the ASO provided herein has one or more modified bases. In another embodiment, the ASO provided herein has one or more modified sugars. In another embodiment, the ASO provided herein has one or more modified intemucleoside linkages.
- the ASO provided herein has one or more modified bases selected from hypoxanthine, pseudouracil, pseudocytosine, 1 -methylpseudouracil, orotic acid, agmatidine, lysidine, 2-thiouracil, 2-thiothymine, 5-halouracil, 5-halomethyluracil, 5- trifluoromethyluracil, 5-propynyluracil, 5 -methylcytosine, 5-propynylcytosine.
- modified bases selected from hypoxanthine, pseudouracil, pseudocytosine, 1 -methylpseudouracil, orotic acid, agmatidine, lysidine, 2-thiouracil, 2-thiothymine, 5-halouracil, 5-halomethyluracil, 5- trifluoromethyluracil, 5-propynyluracil, 5 -methylcytosine, 5-propynylcytosine.
- the ASO provided herein has one or more modified sugars selected from 2'-substituted RNA sugars including threose nucleic acid sugars, 2'-F sugars, 2'- O-substituted RNA sugars including 2'-OMe and 2'-M0E (2'-O-(2-methoxyethyl)) sugars, and a bicyclic (BNA) or tricyclic (TNA) nucleic acid sugars.
- BNA bicyclic
- TAA tricyclic
- the ASO provided herein has one or more BNAs and/or TNAs selected from a conformationally restricted nucleotide (CRN) monomer, a locked nucleic acid (LNA) monomer, a xylo-LNA monomer, an a-LNA monomer, an a-L-LNA monomer, a -D-LNA monomer, a 2'-amino- LNA monomer, a 2'-(alkylamino)-LNA monomer, a 2'-(acylamino)-LN A monomer, a 2'-N- substituted-2'-amino-LNA monomer, a 2'-thio-LNA monomer, a (2'-O,4'-C) constrained ethyl (cEt) BNA monomer, a (2'-O,4'-C) constrained methoxyethyl (cMOE) BNA monomer, a 2',4'-BNANC
- the ASO provided herein has more than one distinct modified sugar.
- the ASO provided herein has a combination of 2'-O- substituted RNA sugars and BNAs.
- the ASO provided herein has a combination of 2'-M0E and LNA sugars.
- the ASO provided herein has all 2'-M0E sugars. See, e.g.. Seth et al.. J. Org. Chem. 2010, 75, 1569-1581; Osawa et al., J. Org. Chem., 2015, 80 (21), pp 10474-10481; WO 2014/145356; WO 2014/126229; Yamamoto et al. Org. Biomol.
- the ASO provided herein has a modified intemucleoside linkage, such as a phosphorothioate or phosphoroamidate. In another embodiment, the ASO provided herein has a phosphorothioate intemucleoside linkage. In another embodiment, the ASO provided herein has at least 50%, 60%, 70%, 80%, 90% or all phosphorothioate intemucleoside linkages.
- the ASO provided herein is a phosphorodiamidate morpholino oligomer (PMO), or is incorporated into a conjugate, such as a pepti deconjugated PMO (PPMO) or an ASO-antibody conjugate.
- PMO phosphorodiamidate morpholino oligomer
- PPMO pepti deconjugated PMO
- ASO-antibody conjugate See, e.g., WO 2022/192749.
- the ASO provided herein has a combination of 2 -MOE and LNA sugars and all phosphorothioate intemucleoside linkages. In another embodiment, the ASO provided herein has all 2'-MOE sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CTGCGCGAGTCTCCCG (SEQ ID NO: 1), and optionally has all 2'-MOE sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CTGCGCGAGTCTCCCG (SEQ ID NO: 1), and optionally has all 2'-MOE sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CTGCGCGAGTCTCCCG (SEQ ID NO: 1), and optionally has all 2'-MOE sugars and all phosphorothioate intemucleoside linkages. In another embodiment, the ASO provided herein contains at least 10, 11, 12, 13. 14, 15 or 16 consecutive bases of CTGCGCGAGTCTCCCG (SEQ ID NO: 1), has all 2'- MOE sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CTGCGCGAGTCTCCCG (SEQ ID NO: 1), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10. 11. 12, 13, 14, 15 or 16 consecutive bases of CTGCGCGAGTCTCCCG (SEQ ID NO: 1), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12. 13, 14, 15 or 16 consecutive bases of TGTACTCACAGTCAGTG (SEQ ID NO: 2), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of TGTACTCACAGTCAGTG (SEQ ID NO: 2), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of TGTACTCACAGTCAGTG (SEQ ID NO: 2), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages. In another embodiment, the ASO provided herein contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of TGTACTCACAGTCAGTG (SEQ ID NO: 2), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages. In another embodiment, the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13.
- TGTACTCACAGTCAGTG SEQ ID NO: 2
- ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of TGTACTCACAGTCAGTG (SEQ ID NO: 2), has all 2'-MOE sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12. 13. 14, 15 or 16 consecutive bases of AGGAGGCAGCTTCCCTG (SEQ ID NO: 3), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of AGGAGGCAGCTTCCCTG (SEQ ID NO: 3), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of AGGAGGCAGCTTCCCTG (SEQ ID NO: 3), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages. In another embodiment, the ASO provided herein contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of AGGAGGCAGCTTCCCTG (SEQ ID NO: 3), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of AGGAGGCAGCTTCCCTG (SEQ ID NO: 3), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14. 15 or 16 consecutive bases of AGGAGGCAGCTTCCCTG (SEQ ID NO: 3), has all 2’-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of ACTGACGCGCGGACTG (SEQ ID NO: 4), and optionally has all 2’-MOE sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of ACTGACGCGCGGACTG (SEQ ID NO: 4), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 1 , 1 1, 12, 13, 14, 15 or 16 consecutive bases of ACTGACGCGCGGACTG (SEQ ID NO: 4), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of ACTGACGCGCGGACTG (SEQ ID NO: 4), has all 2'-MOE sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of ACTGACGCGCGGACTG (SEQ ID NO: 4), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of ACTGACGCGCGGACTG (SEQ ID NO: 4), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12. 13, 14, 15 or 16 consecutive bases of CTGCGCGAGTCTCCCG (SEQ ID NO: 5), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CTGCGCGAGTCTCCCG (SEQ ID NO: 5), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CTGCGCGAGTCTCCCG (SEQ ID NO: 5), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12, 13. 14, 15 or 16 consecutive bases of CTGCGCGAGTCTCCCG (SEQ ID NO: 5), has all 2'- MOE sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CTGCGCGAGTCTCCCG (SEQ ID NO: 5). has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CTGCGCGAGTCTCCCG (SEQ ID NO: 5), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12. 13. 14, 15 or 16 consecutive bases of TGCCAGCCAGGGCGTGCAGG (SEQ ID NO: 6), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of TGCCAGCCAGGGCGTGCAGG (SEQ ID NO: 6), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of TGCCAGCCAGGGCGTGCAGG (SEQ ID NO: 6), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of TGCCAGCCAGGGCGTGCAGG (SEQ ID NO: 6), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13. 14.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14. 15 or 16 consecutive bases of TGCCAGCCAGGGCGTGCAGG (SEQ ID NO: 6), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CGAGAATGCAGCGGCAACAG (SEQ ID NO: 7).
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CGAGAATGCAGCGGCAACAG (SEQ ID NO: 7). and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CGAGAATGCAGCGGCAACAG (SEQ ID NO: 7), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages. In another embodiment, the ASO provided herein contains at least 10. 11. 12, 13, 14, 15 or 16 consecutive bases of CGAGAATGCAGCGGCAACAG (SEQ ID NO: 7), has all 2'-MOE sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CGAGAATGCAGCGGCAACAG (SEQ ID NO: 7), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CGAGAATGCAGCGGCAACAG (SEQ ID NO: 7), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12. 13. 14, 15 or 16 consecutive bases of AAC AGTCC AGAAATTTCTCC (SEQ ID NO: 8), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of AAC AGTCC AGAAATTTCTCC (SEQ ID NO: 8), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of AACAGTCCAGAAATTTCTCC (SEQ ID NO: 8), and optionally has all 2'-MOE sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 1 1, 12, 13, 14, 15 or 16 consecutive bases of AACAGTCCAGAAATTTCTCC (SEQ ID NO: 8), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14.
- ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14. 15 or 16 consecutive bases of AACAGTCCAGAAATTTCTCC (SEQ ID NO: 8), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CCAAGCAATGTGCACGTCAC (SEQ ID NO: 9), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CCAAGCAATGTGCACGTCAC (SEQ ID NO: 9). and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CCAAGCAATGTGCACGTCAC (SEQ ID NO: 9), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CCAAGCAATGTGCACGTCAC (SEQ ID NO: 9). has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CCAAGCAATGTGCACGTCAC (SEQ ID NO: 9), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 1 1 , 12, 13, 14, 15 or 16 consecutive bases of CCAAGCAATGTGCACGTCAC (SEQ ID NO: 9), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12. 13. 14, 15 or 16 consecutive bases of C AAAGAGATGGAGGCTTCCA (SEQ ID NO: 10), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CAAAGAGATGGAGGCTTCCA (SEQ ID NO: 10), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CAAAGAGATGGAGGCTTCCA (SEQ ID NO: 10), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 1 1, 12, 13, 14, 15 or 16 consecutive bases of CAAAGAGATGGAGGCTTCCA (SEQ ID NO: 10), has all 2'-MOE sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CAAAGAGATGGAGGCTTCCA (SEQ ID NO: 10), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14. 15 or 16 consecutive bases of CAAAGAGATGGAGGCTTCCA (SEQ ID NO: 10), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CAGAGGCCAGCTGACTG (SEQ ID NO: 17), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CAGAGGCCAGCTGACTG (SEQ ID NO: 17), and optionally has all 2'-MOE sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CAGAGGCCAGCTGACTG (SEQ ID NO: 17), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages. In another embodiment, the ASO provided herein contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CAGAGGCCAGCTGACTG (SEQ ID NO: 17), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CAGAGGCCAGCTGACTG (SEQ ID NO: 17), has all 2'-MOE sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CAGAGGCCAGCTGACTG (SEQ ID NO: 17), has all 2'-MOE sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12. 13. 14. 15 or 16 consecutive bases of AGGAGGCAGCTTCCCT (SEQ ID NO: 75), and optionally has all 2’-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of AGGAGGCAGCTTCCCT (SEQ ID NO: 75), and optionally has all 2'-MOE sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of AGGAGGCAGCTTCCCT (SEQ ID NO: 75), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of AGGAGGCAGCTTCCCT (SEQ ID NO: 75), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12. 13. 14.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of AGGAGGCAGCTTCCCT (SEQ ID NO: 75), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CACGAGAATGCAGCGGCA (SEQ ID NO: 168), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CACGAGAATGCAGCGGCA (SEQ ID NO: 168), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CACGAGAATGCAGCGGCA (SEQ ID NO: 168). and optionally has all 2'-MOE sugars and all phosphorothioate intemucleoside linkages. In another embodiment, the ASO provided herein contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CACGAGAATGCAGCGGCA (SEQ ID NO: 168), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CACGAGAATGCAGCGGCA (SEQ ID NO: 168), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14. 15 or 16 consecutive bases of CACGAGAATGCAGCGGCA (SEQ ID NO: 168), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CCAGAAATTTCTCCTG (SEQ ID NO: 188), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CCAGAAATTTCTCCTG (SEQ ID NO: 188), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CCAGAAATTTCTCCTG (SEQ ID NO: 188), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages. In another embodiment, the ASO provided herein contains at least 10. 11. 12, 13, 14, 15 or 16 consecutive bases of CCAGAAATTTCTCCTG (SEQ ID NO: 188), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CCAGAAATTTCTCCTG (SEQ ID NO: 188), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CCAGAAATTTCTCCTG (SEQ ID NO: 188), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12. 13. 14. 15 or 16 consecutive bases of CAAGC AATGTGCACGTCACC (SEQ ID NO: 197). and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CAAGCAATGTGCACGTCACC (SEQ ID NO: 197), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CAAGCAATGTGCACGTCACC (SEQ ID NO: 197), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 1 1, 12, 13, 14, 15 or 16 consecutive bases of CAAGCAATGTGCACGTCACC (SEQ ID NO: 197), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of CAAGCAATGTGCACGTCACC (SEQ ID NO: 197), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of TTCC AAAGAGATGGAGGCTT (SEQ ID NO: 225), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of TTCC AAAGAGATGGAGGCTT (SEQ ID NO: 225), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of TTCCAAAGAGATGGAGGCTT (SEQ ID NO: 225), and optionally has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages. In another embodiment, the ASO provided herein contains at least 10. 11. 12, 13, 14, 15 or 16 consecutive bases of TTCCAAAGAGATGGAGGCTT (SEQ ID NO: 225), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-25 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of TTCCAAAGAGATGGAGGCTT (SEQ ID NO: 225), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- the ASO provided herein is 16-20 nucleotides in length, contains at least 10, 11, 12, 13, 14, 15 or 16 consecutive bases of TTCCAAAGAGATGGAGGCTT (SEQ ID NO: 225), has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages.
- an ASO comprising a base sequence with at least 80%, 85%, 90%, 95% or 100% sequence identity to a sequence set forth in any one of Tables 1-4.
- an ASO comprising at least 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19 or 20 consecutive bases of a sequence set forth in any one of Tables 1-4.
- an ASO comprising between 16-25 nucleotides and containing at least 8, 9, 10, 11, 12, 13, 14, 15, 17, 17, 18 19 or 20 consecutive bases of any one of SEQ ID Nos: 1-626.
- an ASO comprising a base sequence set forth in Table 1 or Table 2.
- an ASO comprising a base sequence selected from SEQ ID NOs: 2, 17, 75, 168. 188, 197 and 225.
- the ASO provided herein comprises one or more modified bases, one or more modified sugars, and/or one or more modified intemucleoside linkages.
- the ASO provided herein comprises one or more 2’-M0E sugars and one or more phosphorothioate intemucleoside linkages.
- at least 50%, 75%, 90% or all of the sugars in the ASO provided herein are 2’-M0E sugars and, independently, at least 50%, 75%, 90% or all of the intemucleoside linkages in the ASO provided herein are phosphorothioate intemucleoside linkages.
- the ASO provided herein comprises a base sequence selected from those in Table 1, optionally wherein the ASO has one or more modified bases, one or more modified sugars, and/or one or more modified intemucleoside linkages.
- the ASO provided herein has all 2 -MOE sugars and all phosphorothioate intemucleoside linkages, and has a base sequence selected from those in Table I:
- the ASO provided herein comprises a base sequence selected from those in Table 2, optionally wherein the ASO has one or more modified bases, one or more modified sugars, and/or one or more modified intemucleoside linkages.
- the ASO provided herein has all 2 -MOE sugars and all phosphorothioate intemucleoside linkages, and has a base sequence selected from those in Table 2:
- the ASO provided herein comprises a base sequence selected from those in Table 3, optionally wherein the ASO has one or more modified bases, one or more modified sugars, and/or one or more modified intemucleoside linkages.
- the ASO provided herein has all 2 -MOE sugars and all phosphorothioate intemucleoside linkages, and has a base sequence selected from those in Table 3:
- the ASO provided herein comprises a base sequence selected from SEQ ID NOs: 2, 17, 75, 168, 188, 197 and 225, optionally wherein the ASO has one or more modified bases, one or more modified sugars, and/or one or more modified intemucleoside linkages.
- the ASO provided herein has all 2'-M0E sugars and all phosphorothioate intemucleoside linkages, and has a base sequence selected from SEQ ID NOs: 2, 17, 75, 168, 188, 197 and 225.
- ASOs provided herein may be prepared according to standard methods well known in the art. See, e.g., Beaucage, et al. Tetrahedron 1992, 48(12), 2223; Reese Org. & Biomol. Chem. 2005, 3(21). 3851-3868.
- compositions provided herein contain therapeutically effective amounts of one or more of the ASOs provided herein and a pharmaceutically acceptable carrier, diluent or excipient.
- the ASOs can be formulated into suitable pharmaceutical preparations such as solutions, suspensions, in sterile solutions or suspensions for ophthalmic or parenteral administration, as well as transdermal patch preparation.
- suitable pharmaceutical preparations such as solutions, suspensions, in sterile solutions or suspensions for ophthalmic or parenteral administration, as well as transdermal patch preparation.
- the ASOs described above are formulated into pharmaceutical compositions using techniques and procedures well known in the art (see, e.g.. Ansel Introduction to Pharmaceutical Dosage Forms. Twelfth Edition 2021).
- compositions effective concentrations of one or more ASOs or pharmaceutically acceptable salts is (are) mixed with a suitable pharmaceutical carrier or vehicle.
- concentrations of the ASOs in the compositions are effective for delivery of an amount, upon administration, that treats, prevents, or ameliorates one or more of the symptoms and/or progression of a disease or disorder disclosed herein.
- the compositions are formulated for single dosage administration.
- the weight fraction of ASO is dissolved, suspended, dispersed or otherwise mixed in a selected vehicle at an effective concentration such that the treated condition is relieved or ameliorated.
- Pharmaceutical carriers or vehicles suitable for administration of the ASOs provided herein include any such carriers known to those skilled in the art to be suitable for the particular mode of administration.
- the ASOs may be formulated as the sole pharmaceutically active ingredient in the composition or may be combined with other active ingredients.
- Liposomal suspensions including tissue-targeted liposomes, may also be suitable as pharmaceutically acceptable carriers. These may be prepared according to methods known to those skilled in the art.
- liposome formulations may be prepared as known in the art. Briefly, liposomes such as multilamellar vesicles (MLVs) may be formed by drying down egg phosphatidyl choline and brain phosphatidyl serine (7:3 molar ratio) on the inside of a flask.
- MLVs multilamellar vesicles
- a solution of an ASO provided herein in phosphate buffered saline lacking divalent cations (PBS) is added and the flask shaken until the lipid film is dispersed.
- PBS phosphate buffered saline lacking divalent cations
- the active ASO is included in the pharmaceutically acceptable carrier in an amount sufficient to exert a therapeutically useful effect in the absence of undesirable side effects on the subject treated.
- the therapeutically effective concentration may be determined empirically by testing the ASOs in in vitro and in vivo systems described herein and then extrapolated therefrom for dosages for humans.
- the active ASO is administered in a method to achieve a therapeutically effective concentration of the drug.
- a companion diagnostic see, e.g., Olsen D and Jorgensen J T, Front. Oncol., 2014 May 16, 4: 105, doi: 10.3389/fonC.2014.00105 is used to determine the therapeutic concentration and safety profile of the active ASO in specific subjects or subject populations.
- the concentration of active ASO in the pharmaceutical composition will depend on tissue distribution, inactivation and excretion rates of the active ASO, the physicochemical characteristics of the ASO, the dosage schedule, and amount administered as well as other factors known to those of skill in the art. For example, the amount that is delivered is sufficient to ameliorate one or more of the symptoms of a disease or disorder disclosed herein.
- a therapeutically effective dosage should produce a serum concentration of active ingredient of from about 0.1 ng/mL to about 50-100 pg/mL.
- the pharmaceutical compositions provide a dosage of from about 0.001 mg to about 2000 mg of ASO per kilogram of body weight per day.
- Pharmaceutical dosage unit forms are prepared to provide from about 1 mg to about 1000 mg and in certain embodiments, from about 10 to about 500 mg of the essential active ingredient or a combination of essential ingredients per dosage unit form.
- the active ingredient may be administered at once, or may be divided into a number of smaller doses to be administered at intervals of time. It is understood that the precise dosage and duration of treatment is a function of the disease being treated and may be determined empirically using known testing protocols or by extrapolation from in vivo or in vitro test data. It is to be noted that concentrations and dosage values may also vary with the severity of the condition to be alleviated. It is to be further understood that for any particular subject, specific dosage regimens should be adjusted over time according to the individual need and the professional judgment of the person administering or supervising the administration of the compositions, and that the concentration ranges set forth herein are exemplary only and are not intended to limit the scope or practice of the claimed compositions.
- compositions are mixed with a suitable pharmaceutical carrier or vehicle for systemic, topical or local administration to form pharmaceutical compositions.
- ASOs are included in an amount effective for ameliorating one or more symptoms of, or for treating, retarding progression, or preventing.
- concentration of active ASO in the composition will depend on absorption, tissue distribution, inactivation, excretion rates of the active ASO, the dosage schedule, amount administered, particular formulation as well as other factors know n to those of skill in the art.
- compositions are intended to be administered by a suitable route, including but not limited to parenteral, subcutaneous, intravenous, intramuscular, intraperitoneal, intrathecal, intracerebroventricular, intraocular, mucosal, dermal, transdermal, buccal, rectal, topical or local.
- the compositions are in liquid, semi-liquid or solid form and are formulated in a manner suitable for each route of administration.
- intracerebroventricular refers to administration of a composition into the ventricular system of the brain, e.g., via injection, infusion, or implantation (for example, into a ventricle of the brain).
- intraocular refers to the administration of a composition to the eye region, e.g., via injection, infusion, or implantation (for example, into the eyeball) or topical/ophthalmic administration (for example, using a cream, ointment, gel or liquid drops).
- intrathecal refers to administration of a composition into the lumbar region, e.g., via injection, infusion, or implantation (for example, into the subarachnoid space of the spinal cord).
- Solutions or suspensions used for parenteral, intradermal, subcutaneous, or topical application can include any of the following components: a sterile diluent, such as water for injection, saline solution, fixed oil, polyethylene glycol, glycerin, propylene glycol, dimethyl acetamide or other synthetic solvent; antimicrobial agents, such as benzyl alcohol and methyl parabens; antioxidants, such as ascorbic acid and sodium bisulfite; chelating agents, such as ethylenediaminetetraacetic acid (EDTA); buffers, such as acetates, citrates and phosphates; and agents for the adjustment of tonicity such as sodium chloride or dextrose.
- a sterile diluent such as water for injection, saline solution, fixed oil, polyethylene glycol, glycerin, propylene glycol, dimethyl acetamide or other synthetic solvent
- antimicrobial agents such as benzyl alcohol and methyl parabens
- ASOs exhibit insufficient solubility
- methods for solubilizing ASOs may be used. Such methods are known to those of skill in this art, and include, but are not limited to, using cosolvents, such as dimethylsulfoxide (DMSO), using surfactants, such as TWEEN®, or dissolution in aqueous sodium bicarbonate.
- cosolvents such as dimethylsulfoxide (DMSO)
- surfactants such as TWEEN®
- the resulting mixture may be a solution, suspension, emulsion or the like.
- the form of the resulting mixture depends upon a number of factors, including the intended mode of administration and the solubility of the ASO in the selected carrier or vehicle.
- the effective concentration is sufficient for ameliorating the symptoms of the disease, disorder or condition treated and may be empirically determined.
- the pharmaceutical compositions are provided for administration to humans and animals in unit dosage forms, such as powders, granules, sterile parenteral solutions or suspensions, and oil water emulsions containing suitable quantities of the ASOs or pharmaceutically acceptable salts thereof.
- the pharmaceutically therapeutically active ASOs and salts thereof are formulated and administered in unit dosage forms or multiple dosage forms.
- Unit dose forms as used herein refer to physically discrete units suitable for human and animal subjects and packaged individually as is known in the art. Each unit dose contains a predetermined quantity of the therapeutically active ASO sufficient to produce the desired therapeutic effect, in association with the required pharmaceutical carrier, vehicle or diluent. Examples of unit dose forms include ampules and syringes and individually packaged tablets or capsules. Unit dose forms may be administered in fractions or multiples thereof.
- a multiple dose form is a plurality of identical unit dosage forms packaged in a single container to be administered in segregated unit dose form. Examples of multiple dose forms include vials, bottles of tablets or capsules or bottles of pints or gallons. Hence, multiple dose form is a multiple of unit doses which are not segregated in packaging.
- sustained-release preparations can also be prepared. Suitable examples of sustained- release preparations include semipermeable matrices of solid hydrophobic polymers containing the ASO provided herein, which matrices are in the form of shaped articles, e.g., films, or microcapsule.
- sustained-release matrices include iontophoresis patches, polyesters, hydrogels (for example, poly(2-hydroxyethyl-methaciylate), or poly(vinylalcohol)), polylactides, copolymers of L-glutamic acid and ethyl-L-glutamate, non- degradable ethylene-vinyl acetate, degradable lactic acid-glycolic acid copolymers such as the LUPRON DEPOTTM (injectable microspheres composed of lactic acid-glycolic acid copolymer and leuprolide acetate), and poly-D-(-)-3-hydroxybutyric acid.
- iontophoresis patches for example, polyesters, hydrogels (for example, poly(2-hydroxyethyl-methaciylate), or poly(vinylalcohol)), polylactides, copolymers of L-glutamic acid and ethyl-L-glutamate, non- degradable ethylene-
- stabilization may be achieved by modifying sulfhydry l residues, lyophilizing from acidic solutions, controlling moisture content, using appropriate additives, and developing specific polymer matrix compositions.
- compositions containing active ingredient in the range of 0.005% to 100% with the balance made up from non-toxic carrier may be prepared.
- a pharmaceutically acceptable non-toxic composition is formed by the incorporation of any of the normally employed excipients, such as, for example pharmaceutical grades of mannitol, lactose, starch, magnesium stearate, talcum, cellulose derivatives, sodium croscarmellose, glucose, sucrose, magnesium carbonate or sodium saccharin.
- compositions include solutions, suspensions, powders and sustained release formulations, such as, but not limited to, implants and microencapsulated delivery systems, and biodegradable, biocompatible polymers, such as collagen, ethylene vinyl acetate, polyanhydrides, polygly colic acid, polyorthoesters, polylactic acid and others. Methods for preparation of these compositions are know n to those skilled in the art.
- the contemplated compositions may contain about 0.001% 100% active ingredient, in certain embodiments, about 0.1 85% or about 75-95%.
- the active ASOs or pharmaceutically acceptable salts may be prepared with carriers that protect the ASO against rapid elimination from the body, such as time release formulations or coatings.
- compositions may include other active ASOs to obtain desired combinations of properties.
- the ASOs provided herein, or pharmaceutically acceptable salts thereof as described herein may also be advantageously administered for therapeutic or prophylactic purposes together with another pharmacological agent known in the general art to be of value in treating one or more of the diseases or medical conditions referred to hereinabove, such as diseases related to STXBP1 haploinsufficiency. It is to be understood that such combination therapy constitutes a further aspect of the compositions and methods of treatment provided herein.
- anhydrous pharmaceutical compositions and dosage forms containing an ASO provided herein.
- water e.g., 5%
- water and heat accelerate the decomposition of some ASOs.
- the effect of water on a formulation can be of great significance since moisture and/or humidity are commonly encountered during manufacture, handling, packaging, storage, shipment and use of formulations.
- Anhydrous pharmaceutical compositions and dosage forms provided herein can be prepared using anhydrous or low moisture containing ingredients and low moisture or low humidity conditions.
- Pharmaceutical compositions and dosage forms that comprise lactose and at least one active ingredient that comprises a primary or secondary’ amine are anhydrous if substantial contact w ith moisture and/or humidity during manufacturing, packaging, and/or storage is expected.
- anhydrous pharmaceutical composition should be prepared and stored such that its anhydrous nature is maintained. Accordingly, anhy drous compositions are packaged using materials known to prevent exposure to water such that they can be included in suitable formulary kits. Examples of suitable packaging include, but are not limited to, hermetically sealed foils, plastics, unit dose containers (e.g., vials), blister packs and strip packs.
- Parenteral administration generally characterized by injection, either intrathecally, intracerebroventricularly, intraocularly, subcutaneously, intramuscularly or intravenously is also contemplated herein.
- injectables can be prepared in conventional forms, either as liquid solutions or suspensions, solid forms suitable for solution or suspension in liquid prior to injection, or as emulsions.
- the suspension is a suspension of microparticles or nanoparticles.
- the emulsion is an emulsion of microparticles or nanoparticles.
- Suitable excipients are, for example, water, saline, dextrose, glycerol or ethanol.
- compositions to be administered may also contain minor amounts of non-toxic auxiliary substances such as wetting or emulsifying agents. pH buffering agents, stabilizers, solubility enhancers, and other such agents, such as for example, sodium acetate, sorbitan monolaurate. triethanolamine oleate and cyclodextrins.
- auxiliary substances such as wetting or emulsifying agents. pH buffering agents, stabilizers, solubility enhancers, and other such agents, such as for example, sodium acetate, sorbitan monolaurate. triethanolamine oleate and cyclodextrins.
- preservatives, stabilizers or excipients may be used in the composition.
- numerous well known and routinely employed preservatives, stabilizers and excipients useful for formulations for intrathecal or intracerebroventricular delivery are known in the art.
- an ASO provided herein is dispersed in a solid inner matrix, e.g., polymethylmethacrylate, poly butyl methacryl ate.
- plasticized or unplasticized polyvinylchloride plasticized nylon, plasticized polyethyleneterephthalate, natural rubber, polyisoprene, polyisobutylene, polybutadiene, polyethylene, ethylene-vinylacetate copolymers, silicone rubbers, polydimethylsiloxanes, silicone carbonate copolymers, hydrophilic polymers such as hydrogels of esters of acrylic and methacry lic acid, collagen, cross-linked polyvinylalcohol and cross-linked partially hydrolyzed polyvinyl acetate, that is surrounded by an outer polymeric membrane, e.g., polyethylene, polypropylene, ethylene/propylene copolymers, ethylene/ethyl acrylate copolymers, ethylene/vinylacetate copolymers, silicone rubbers, poly dimethyl siloxanes, neoprene rubber, chlorinated polyethylene, polyvinylchloride, vinylchloride copolymers with vinyl acetate, vinyli
- Parenteral administration of the compositions includes intrathecal, intracerebroventricular, intraocular, intravenous, subcutaneous and intramuscular administrations.
- Preparations for parenteral administration include sterile solutions ready for injection, sterile dry soluble products, such as lyophilized powders, ready to be combined with a solvent just prior to use, including hypodermic tablets, sterile suspensions ready for injection, sterile dry insoluble products ready to be combined with a vehicle just prior to use and sterile emulsions.
- the solutions may be either aqueous or nonaqueous.
- suitable carriers include physiological saline or phosphate buffered saline (PBS), and solutions containing thickening and solubilizing agents, such as glucose, polyethylene glycol, and polypropylene glycol and mixtures thereof.
- suitable carriers include physiological saline or phosphate buffered saline (PBS), and solutions containing thickening and solubilizing agents, such as glucose, polyethylene glycol, and polypropylene glycol and mixtures thereof.
- Pharmaceutically acceptable carriers used in parenteral preparations include aqueous vehicles, nonaqueous vehicles, antimicrobial agents, isotonic agents, buffers, antioxidants, local anesthetics, suspending and dispersing agents, emulsifying agents, sequestering or chelating agents and other pharmaceutically acceptable substances.
- aqueous vehicles include Sodium Chloride Injection, Ringers Injection, Isotonic Dextrose Injection, Sterile Water Injection, Dextrose and Lactated Ringers Injection.
- Nonaqueous parenteral vehicles include fixed oils of vegetable origin, cottonseed oil, com oil. sesame oil and peanut oil.
- Antimicrobial agents in bacteriostatic or fungistatic concentrations must be added to parenteral preparations packaged in multiple dose containers which include phenols or cresols, mercurials, benzy l alcohol, chlorobutanol, methyl and propyl p hy droxy benzoic acid esters, thimerosal, benzalkonium chloride and benzethonium chloride.
- Isotonic agents include sodium chloride and dextrose. Buffers include phosphate and citrate. Antioxidants include sodium bisulfate. Local anesthetics include procaine hydrochloride. Suspending and dispersing agents include sodium carboxymethylcelluose, hydroxypropyl methylcellulose and polyvinylpyrrolidone. Emulsifying agents include Polysorbate 80 (TWEEN® 80). A sequestering or chelating agent of metal ions include EDTA. Pharmaceutical carriers also include ethyl alcohol, polyethylene glycol and propylene glycol for water miscible vehicles and sodium hydroxide, hydrochloric acid, citric acid or lactic acid for pH adjustment.
- the concentration of the pharmaceutically active ASO is adjusted so that an injection provides an effective amount to produce the desired pharmacological effect.
- the exact dose depends on the age, weight and condition of the subject or animal as is known in the art.
- the unit dose parenteral preparations are packaged in an ampule, a vial or a syringe with a needle. All preparations for parenteral administration must be sterile, as is known and practiced in the art.
- intravenous or intraarterial infusion of a sterile aqueous solution containing an active ASO is an effective mode of administration.
- Another embodiment is a sterile aqueous or oily solution or suspension containing an active material injected as necessary to produce the desired pharmacological effect.
- Injectables are designed for local and systemic administration.
- a therapeutically effective dosage is formulated to contain a concentration of at least about 0.1% w/w up to about 90% w/w or more, such as more than 1% w/w of the active ASO to the treated tissue(s).
- the active ingredient may be administered at once, or may be divided into a number of smaller doses to be administered at intervals of time.
- the precise dosage and duration of treatment is a function of the tissue being treated and may be determined empirically using known testing protocols or by extrapolation from in vivo or in vitro test data. It is to be noted that concentrations and dosage values may also vary with the age of the individual treated. It is to be further understood that for any particular subject, specific dosage regimens should be adjusted over time according to the individual need and the professional judgment of the person administering or supervising the administration of the formulations, and that the concentration ranges set forth herein are exemplary only and are not intended to limit the scope or practice of the claimed formulations.
- the ASO may be suspended in micronized or other suitable form or may be derivatized to produce a more soluble active product or to produce a prodrug.
- the form of the resulting mixture depends upon a number of factors, including the intended mode of administration and the solubility of the ASO in the selected carrier or vehicle.
- the effective concentration is sufficient for ameliorating the symptoms of the condition and may be empirically determined.
- lyophilized powders which can be reconstituted for administration as solutions, emulsions and other mixtures. They may also be reconstituted and formulated as solids or gels.
- the sterile, lyophilized powder is prepared by dissolving an ASO provided herein, or a pharmaceutically acceptable salt thereof, in a suitable solvent.
- the solvent may contain an excipient which improves the stability or other pharmacological component of the powder or reconstituted solution, prepared from the powder. Excipients that may be used include, but are not limited to, dextrose, sorbitol, fructose, com syrup, xylitol, glycerin, glucose, sucrose or other suitable agent.
- the solvent may also contain a buffer, such as citrate, sodium or potassium phosphate or other such buffer known to those of skill in the art at, in one embodiment, about neutral pH.
- lyophilized powder can be stored under appropriate conditions, such as at about 4° C. to room temperature.
- Reconstitution of this lyophilized powder with water for injection provides a formulation for use in parenteral administration.
- about 1-50 mg, about 5- 35 mg, or about 9-30 mg of lyophilized powder is added per mL of sterile water or other suitable carrier.
- the precise amount depends upon the selected ASO. Such amount can be empirically determined.
- Topical mixtures are prepared as described for the local and systemic administration.
- the resulting mixture may be a solution, suspension, emulsion or the like and are formulated as creams, gels, ointments, emulsions, solutions, elixirs, lotions, suspensions, tinctures, pastes, foams, aerosols, irrigations, sprays, suppositories, bandages, dermal patches or any other formulations suitable for topical administration.
- the ASOs or pharmaceutically acceptable salts thereof may be formulated as aerosols for topical application, such as by inhalation (see, e.g., U.S. Pat. Nos. 4,044.126, 4,414,209. and 4,364,923. which describe aerosols for delivery of a steroid useful for treatment of inflammatory diseases, particularly asthma).
- These formulations for administration to the respiratory 7 tract can be in the form of an aerosol or solution for a nebulizer, or as a microfine powder for insufflation, alone or in combination with an inert carrier such as lactose. In such a case, the particles of the formulation will have diameters of less than 50 microns or less than 10 microns.
- the ASOs may be formulated for local or topical application, such as for topical application to the skin and mucous membranes, such as in the eye, in the form of gels, creams, and lotions and for application to the eye or for intracistemal or intraspinal application.
- Topical administration is contemplated for trans dermal delivery and also for administration to the eyes or mucosa, or for inhalation therapies.
- Nasal solutions of the active ASO alone or in combination with other pharmaceutically acceptable excipients can also be administered.
- solutions particularly those intended for ophthalmic use. may be formulated as 0.01%-10% isotonic solutions, pH about 5-7, with appropriate salts.
- Active ingredients provided herein can be administered by controlled release means or by delivery devices that are well known to those of ordinary skill in the art. Examples include, but are not limited to, those described in U.S. Pat. Nos. 3,845,770; 3,916,899; 3,536,809; 3,598,123; and U.S. Pat. Nos. 4,008,719, 5,674,533, 5,059,595, 5,591,767, 5,120,548, 5.073,543, 5,639.476, 5,354,556, 5,639,480.
- Such dosage forms can be used to provide slow or controlled-release of one or more active ingredients using, for example, hydroxypropylmethyl cellulose, other polymer matrices, gels, permeable membranes, osmotic systems, multilayer coatings, microparticles, liposomes, microspheres, or a combination thereof to provide the desired release profile in varying proportions.
- Suitable controlled- release formulations known to those of ordinary skill in the art, including those described herein, can be readily selected for use with the active ingredients provided herein.
- controlled-release pharmaceutical products have a common goal of improving drug therapy over that achieved by their non-controlled counterparts.
- the use of an optimally designed controlled-release preparation in medical treatment is characterized by a minimum of drug substance being employed to cure or control the condition in a minimum amount of time.
- advantages of controlled- release formulations include extended activity of the drug, reduced dosage frequency, and increased subject compliance.
- controlled-release formulations can be used to affect the time of onset of action or other characteristics, such as blood levels of the drug, and can thus affect the occurrence of side (e.g., adverse) effects.
- Controlled release of an active ingredient can be stimulated by various conditions including, but not limited to, pH, temperature, enzymes, water, or other physiological conditions or ASOs.
- the agent may be administered using intravenous infusion, an implantable osmotic pump, a transdermal patch, liposomes, or other modes of administration.
- a pump may be used (see, Sefton, CRC Crit. Ref.
- polymeric materials can be used.
- a controlled release system can be placed in proximity of the therapeutic target, i.e., thus requiring only a fraction of the systemic dose (see, e.g., Goodson, Medical Applications of Controlled Release, vol. 2, pp. 115-138 (1984).
- a controlled release device is introduced into a subject in proximity of the site of inappropriate immune activation or a tumor.
- the active ingredient can be dispersed in a solid inner matrix, e.g., polymethylmethacrylate, polybutylmethacrylate, plasticized or unplasticized polyvinylchloride, plasticized nylon, plasticized polyethyleneterephthalate, natural rubber, polyisoprene, polyisobutylene, polybutadiene, polyethylene, ethylene-vinylacetate copolymers, silicone rubbers, polydimethylsiloxanes, silicone carbonate copolymers, hydrophilic polymers such as hydrogels of esters of acrylic and methacrylic acid, collagen, cross-linked polyvinylalcohol and cross-linked partially hydrolyzed polyvinyl acetate, that is surrounded by an outer polymeric membrane, e.g., polyethylene, poly
- the ASOs provided herein, or pharmaceutically acceptable salts thereof, may also be formulated to be targeted to a particular tissue, receptor, or other area of the body of the subject to be treated, including liposome-, resealed erythrocyte-, and antibody-based delivery systems. Many such targeting methods are well known to those of skill in the art. All such targeting methods are contemplated herein for use in the instant compositions. For nonlimiting examples of targeting methods, see, e.g., U.S. Pat. Nos. 6,316.652. 6,274,552.
- the antibody-based delivery system is an antibody-drug conjugate ("ADC"), e.g.. as described in Hamilton G S, Biologicals, 2015 September, 43(5):318-32; Kim E G and Kim K M, Biomol. Ther. (Seoul), 2015 November, 23(6):493- 509; and Peters C and Brown S, Biosci. Rep., 2015 Jun. 12, 35(4) pii: e00225, each of which is incorporated herein by reference.
- ADC antibody-drug conjugate
- liposomal suspensions including tissue-targeted liposomes, such as tumor-targeted liposomes, may also be suitable as pharmaceutically acceptable carriers.
- tissue-targeted liposomes such as tumor-targeted liposomes
- liposome formulations may be prepared according to methods known to those skilled in the art.
- liposome formulations may be prepared as described in U.S. Pat. No. 4,522,811. Briefly, liposomes such as multilamellar vesicles (MLV's) may be formed by drying down egg phosphatidyl choline and brain phosphatidyl serine (7:3 molar ratio) on the inside of a flask.
- MLV's multilamellar vesicles
- a solution of an ASO provided herein in phosphate buffered saline lacking divalent cations (PBS) is added and the flask shaken until the lipid film is dispersed.
- PBS phosphate buffered saline lacking divalent cations
- ASOs and pharmaceutical compositions provided herein may be dosed in certain therapeutically or prophylactically effective amounts, certain time intervals, certain dosage forms, and certain dosage administration methods as described below.
- a therapeutically or prophylactically effective amount of the ASO is from about 0.005 to about 1,000 mg per day, from about 0.01 to about 500 mg per day, from about 0.01 to about 250 mg per day, from about 0.01 to about 100 mg per day. from about 0. 1 to about 100 mg per day. from about 0.5 to about 100 mg per day, from about 1 to about 100 mg per day, from about 0.01 to about 50 mg per day, from about 0.1 to about 50 mg per day, from about 0.5 to about 50 mg per day, from about 1 to about 50 mg per day, from about 0.02 to about 25 mg per day. from about 0.05 to about 10 mg per day, from about 0.05 to about 5 mg per day, from about 0. 1 to about 5 mg per day, or from about 0.5 to about 5 mg per day.
- the therapeutically or prophylactically effective amount is about 0.1, about 0.2, about 0.5, about 1, about 2, about 3, about 4, about 5, about 6, about 7, about 8, about 9, about 10, about 15, about 20, about 25, about 30, about 40, about 45, about 50. about 60. about 70. about 80, about 90, about 100. or about 150 mg per day.
- the recommended daily dose range of the ASO provided herein, or a derivative thereof, for the conditions described herein he within the range of from about 0.5 mg to about 50 mg per day, in one embodiment given as a single once-a-day dose, or in divided doses throughout a day.
- the dosage ranges from about 1 mg to about 50 mg per day.
- the dosage ranges from about 0.5 to about 5 mg per day.
- Specific doses per day include 0.1, 0.2, 0.5, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27. 28. 29. 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49 or 50 mg per day.
- the recommended starting dosage may be 0.5, 1, 2, 3, 4, 5, 10, 15, 20, 25 or 50 mg per day. In another embodiment, the recommended starting dosage may be 0.5, 1, 2, 3. 4, or 5 mg per day.
- the dose may be escalated to 15, 20, 25, 30, 35, 40, 45 and 50 mg/day.
- the ASO can be administered in an amount of about 25 mg/day. In a particular embodiment, the ASO can be administered in an amount of about 10 mg/day. In a particular embodiment, the ASO can be administered in an amount of about 5 mg/day. In a particular embodiment, the ASO can be administered in an amount of about 4 mg/day. In a particular embodiment, the ASO can be administered in an amount of about 3 mg/day.
- the therapeutically or prophylactically effective amount is from about 0.001 to about 100 mg/kg/day, from about 0.01 to about 50 mg/kg/day, from about 0.01 to about 25 mg/kg/day. from about 0.01 to about 10 mg/kg/day, from about 0.01 to about 9 mg/kg/day, 0.01 to about 8 mg/kg/day. from about 0.01 to about 7 mg/kg/day. from about 0.01 to about 6 mg/kg/day, from about 0.01 to about 5 mg/kg/day, from about 0.01 to about 4 mg/kg/day, from about 0.01 to about 3 mg/kg/day, from about 0.01 to about 2 mg/kg/day. from about 0.01 to about 1 mg/kg/day, or from about 0.01 to about 0.05 mg/kg/day.
- the administered dose can also be expressed in units other than mg/kg/day.
- doses for parenteral administration can be expressed as mg/m 2 /day.
- doses for parenteral administration can be expressed as mg/m 2 /day.
- One of ordinary skill in the art would readily know how to convert doses from mg/kg/day to mg/m 2 /day to given either the height or weight of a subject or both (see. www.fda.gov/cder/cancer/animalframe.htm).
- a dose of 1 mg/kg/day for a 65 kg human is approximately equal to 38 mg/m 2 /day.
- the amount of the ASO administered is sufficient to provide a plasma concentration of the ASO at steady state, ranging from about 0.001 to about 500 pM, about 0.002 to about 200 pM, about 0.005 to about 100 pM, about 0.01 to about 50 pM, from about 1 to about 50 pM, about 0.02 to about 25 pM, from about 0.05 to about 20 pM, from about 0. 1 to about 20 pM, from about 0.5 to about 20 pM, or from about 1 to about 20 pM.
- the amount of the ASO administered is sufficient to provide a plasma concentration of the ASO at steady state, ranging from about 5 to about 100 nM, about 5 to about 50 nM, about 10 to about 100 nM, about 10 to about 50 nM or from about 50 to about 100 nM.
- plasma concentration at steady state is the concentration reached after a period of administration of an ASO provided herein, or a derivative thereof. Once steady state is reached, there are minor peaks and troughs on the time dependent curve of the plasma concentration of the ASO.
- the amount of the ASO administered is sufficient to provide a maximum plasma concentration (peak concentration) of the ASO, ranging from about 0.001 to about 50 pM, about 0.002 to about 200 pM, about 0.005 to about 100 pM, about 0.01 to about 50 pM, from about 1 to about 50 pM, about 0.02 to about 25 pM, from about 0.05 to about 20 pM, from about 0. 1 to about 20 pM. from about 0.5 to about 20 pM, or from about 1 to about 20 pM.
- the amount of the ASO administered is sufficient to provide a minimum plasma concentration (trough concentration) of the ASO, ranging from about 0.001 to about 500 pM, about 0.002 to about 200 pM, about 0.005 to about 100 pM. about 0.01 to about 50 pM, from about 1 to about 50 pM. about 0.01 to about 25 pM, from about 0.01 to about 20 pM, from about 0.02 to about 20 pM, from about 0.02 to about 20 pM, or from about 0.01 to about 20 pM.
- the amount of the ASO administered is sufficient to provide an area under the curve (AUC) of the ASO. ranging from about 100 to about 100,000 ng*hr/mL, from about 1 ,000 to about 50,000 ng*hr/mL, from about 5,000 to about 25,000 ng*hr/mL, or from about 5,000 to about 10,000 ng*hr/mL.
- AUC area under the curve
- the methods provided herein encompass treating a patient regardless of subject's age, although some diseases or disorders are more common in certain age groups.
- the ASO provided herein, or a derivative thereof may be administered by oral, parenteral (e.g., intramuscular, intraperitoneal, intravenous, CIV, intracistemal injection or infusion, subcutaneous injection, or implant), inhalation, nasal, vaginal, rectal, sublingual, or topical (e.g.. transdermal or local) routes of administration.
- parenteral e.g., intramuscular, intraperitoneal, intravenous, CIV, intracistemal injection or infusion, subcutaneous injection, or implant
- inhalation nasal, vaginal, rectal, sublingual, or topical (e.g.. transdermal or local) routes of administration.
- the ASO provided herein, or a derivative thereof may be formulated, alone or together, in suitable dosage unit with pharmaceutically acceptable excipients, carriers, adjuvants and vehicles, appropriate for each route of administration.
- the ASO provided herein, or a derivative thereof is administered orally. In another embodiment, the ASO provided herein, or a derivative thereof, is administered parenterally. In yet another embodiment, the ASO provided herein, or a derivative thereof, is administered intravenously.
- the ASO provided herein, or a derivative thereof can be delivered as a single dose such as, e.g., a single bolus injection, or oral tablets or pills; or over time, such as, e.g., continuous infusion over time or divided bolus doses over time.
- the ASO can be administered repeatedly if necessary, for example, until the subject experiences stable disease or regression, or until the subject experiences disease progression or unacceptable toxicity.
- stable disease for solid tumors generally means that the perpendicular diameter of measurable lesions has not increased by 25% or more from the last measurement.
- Stable disease or lack thereof is determined by methods known in the art such as evaluation of patient symptoms, physical examination, visualization of the tumor that has been imaged using X-ray, CAT, PET, or MRI scan and other commonly accepted evaluation modalities.
- the ASO provided herein, or a derivative thereof can be administered once daily (QD), or divided into multiple daily doses such as twice daily (BID), three times daily (TID), and four times daily (QID).
- the administration can be continuous (i.e., daily for consecutive days or every day), intermittent, e.g., in cycles (i.e., including days, weeks, or months of rest without drug).
- the term "daily” is intended to mean that a therapeutic ASO, such as the ASO provided herein, or a derivative thereof, is administered once or more than once each day, for example, for a period of time.
- continuous is intended to mean that a therapeutic ASO, such as the ASO provided herein or a derivative thereof, is administered daily for an uninterrupted period of at least 10 days to 52 weeks.
- intermittent or “intermittently” as used herein is intended to mean stopping and starting at either regular or irregular intervals.
- intermittent administration of the ASO provided herein or a derivative thereof is administration for one to six days per week, administration in cycles (e.g., daily administration for two to eight consecutive weeks, then a rest period with no administration for up to one week), or administration on alternate days.
- cycling as used herein is intended to mean that a therapeutic ASO, such as the ASO provided herein or a derivative thereof, is administered daily or continuously but with a rest period. In some such embodiments, administration is once a day for two to six days, then a rest period with no administration for five to seven days.
- the frequency of administration is in the range of about a daily dose to about a monthly dose.
- administration is once a day. twice a day, three times a day, four times a day, once every other day, twice a week, once every week, once every two weeks, once every three weeks, or once every four weeks.
- the ASO provided herein, or a derivative thereof is administered once a day.
- the ASO provided herein, or a derivative thereof is administered twice a day.
- the ASO provided herein, or a derivative thereof is administered three times a day.
- the ASO provided herein, or a derivative thereof is administered four times a day.
- the ASO provided herein, or a derivative thereof is administered once per day from one day to six months, from one week to three months, from one w eek to four w eeks, from one week to three weeks, or from one week to two w eeks. In certain embodiments, the ASO provided herein, or a derivative thereof, is administered once per day for one week, two weeks, three weeks, or four weeks. In one embodiment, the ASO provided herein, or a derivative thereof, is administered once per day for 4 days. In one embodiment, the ASO provided herein, or a derivative thereof, is administered once per day for 5 days. In one embodiment, the ASO provided herein, or a derivative thereof, is administered once per day for 6 days.
- the ASO provided herein, or a derivative thereof is administered once per day for one week. In another embodiment, the ASO provided herein, or a derivative thereof, is administered once per day for two weeks. In yet another embodiment, the ASO provided herein, or a derivative thereof, is administered once per day for three weeks. In still another embodiment, the ASO provided herein, or a derivative thereof, is administered once per day for four weeks.
- the AS Os provided herein are useful for increasing expression of STXBP1.
- the ASOs provided herein increase the expression of STXBP1 protein when contacted with a cell such as a neuronal cell.
- an ASO provided herein increases a level of functional STXBP1 protein in a neuronal cell relative to a control cell not contacted with the ASO.
- an ASO provided herein increases the synaptic local expression of STXBP1 protein in a composition containing neuronal cells.
- the composition containing neuronal cells is in vivo.
- the ASOs provided herein modulate expression of STXBP1.
- the ASOs provided herein increase expression of STXBP1 in a neuronal cell.
- the neuronal cell is haploinsufficient for STXBP1 and/or heterozygous for a deleterious mutation in STXBP1.
- Also provided is a method of treating, preventing, or delaying the onset of an STXBP1 disorder using the ASOs provided herein.
- the methods provided herein result in increased levels of STXBP1 in a cell and therefore are useful in methods of treatment of diseases which are associated with STXBP1 defects (i.e., reduced STXBP1 levels and/or loss-of-function mutations of the STXBP1 gene).
- the methods provided herein can be performed in vitro, ex vivo or in vivo.
- the STXBP1 disorder is associated with STXBP1 haploinsufficiency.
- the STXBP1 disorder is encephalopathy.
- the STXBP1 disorder is STXBP1 encephalopathy.
- the STXBP1 disorder is epilepsy.
- the STXBP1 disorder is epileptic encephalopathy.
- the STXBP1 disorder is a severe early onset epileptic encephalopathy or a non-syndromic epilepsy selected from Ohtahara syndrome, West syndrome, Lennox-Gastaut syndrome, Dravet syndrome, early myoclonic encephalopathy, an unclassified early onset epileptic encephalopathy that is associated with STXBP1 haploinsufficiency, atypical Rett syndrome and a severe intellectual disability without epilepsy associated with STXBP1 haploinsufficiency.
- the STXBP1 disorder is epilepsy, global delay, cognitive impairment (mild to profound), a movement disorder, hypotonia or autism.
- the STXBP1 disorder is DEE4, Developmental and epileptic encephalopathy 4, Developmental and epileptic encephalopathy, type 4, Early- infantile epileptic encephalopathy 4, EIEE4, STXBP1 encephalopathy with epilepsy, STXBP1 epileptic encephalopathy, STXBP1 -related developmental and epileptic encephalopathy, STXBP1 -related early-onset encephalopathy, or STXBP1 -related epileptic encephalopathy.
- a method for treating, preventing or delaying the onset of a disorder caused by an STXBP 1 mutation using an ASO provided herein is provided.
- Many such mutations, many of which are missense mutations, are known (see, e.g., www. stxbp 1 disorders, org/what-is-stxbp 1 ).
- provided is a method for treating, preventing, or delaying the onset of a disease in which disease processes lower STXBP 1 levels using an ASO provided herein.
- the disease is amyotrophic lateral sclerosis (ALS).
- the STXBP 1 gene may not carry a mutation.
- the ASO provided herein, or a derivative thereof, can also be combined or used in combination w ith other therapeutic agents useful in the treatment and/or prevention of an STXBP 1 disorder.
- provided herein is a method of treating, preventing, or delaying an STXBP 1 disorder, comprising administering to a subject an ASO provided herein, or a derivative thereof; in combination with one or more second active agents.
- the term "in combination” includes the use of more than one therapy (e.g., one or more prophylactic and/or therapeutic agents). However, the use of the term “in combination” does not restrict the order in which therapies (e g., prophylactic and/or therapeutic agents) are administered to a subject with a disease or disorder.
- a first therapy e.g., a prophylactic or therapeutic agent such as an ASO provided herein, an ASO provided herein, e.g., the ASO provided herein, or a derivative thereof
- can be administered prior to e.g., 5 minutes, 15 minutes, 30 minutes, 45 minutes, 1 hour, 2 hours. 4 hours, 6 hours, 12 hours, 24 hours, 48 hours, 72 hours, 96 hours.
- a second therapy e.g., a prophylactic or therapeutic agent
- Triple therapy is also contemplated herein.
- Administration of the ASO provided herein, or a derivative thereof and one or more second active agents to a subject can occur simultaneously or sequentially by the same or different routes of administration.
- the suitability of a particular route of administration employed for a particular active agent will depend on the active agent itself (e.g.. whether it can be administered orally without decomposing prior to entering the blood stream) and the disease or disorder being treated.
- the route of administration of the ASO provided herein, or a derivative thereof is independent of the route of administration of a second therapy.
- the ASO provided herein, or a derivative thereof is administered intrathecally, intracerebroventricularly, intraocularly or intravenously.
- the ASO provided herein, or a derivative thereof is administered intrathecally, intracerebroventricularly, intraocularly or intravenously
- the second therapy can be administered orally, parenterally, intraperitoneally, intravenously, intraarterially, transdermally, sublingually, intramuscularly, rectally, transbuccally, intranasally, liposomally, via inhalation, vaginally, intraoccularly, via local delivery by catheter or stent, subcutaneously, intraadiposally. intraarticularly, intrathecally, intracerebroventricularly, or in a slow release dosage form.
- the ASO provided herein, or a derivative thereof, and a second therapy are administered by the same mode of administration, e.g., intrathecally, intracerebroventricularly, intraocularly or intravenously.
- the ASO provided herein, or a derivative thereof is administered by one mode of administration, e.g., intrathecally, intracerebroventricularly, intraocularly or intravenously, whereas the second agent is administered by another mode of administration, e.g., orally.
- the second active agent is administered intravenously or subcutaneously and once or twice daily in an amount of from about 1 to about 1000 mg, from about 5 to about 500 mg. from about 10 to about 350 mg.
- the specific amount of the second active agent will depend on the specific agent used, the type of disease being treated or managed, the severity and stage of disease, and the amount of the ASO provided herein, or a derivative thereof, and any optional additional active agents concurrently administered to the subject.
- Second active ingredients or agents can be used together with the ASO provided herein, or a derivative thereof, in the methods and compositions provided herein.
- Second active agents can be large molecules (e.g.. proteins) or small molecules (e.g., synthetic inorganic, organometallic, or organic molecules).
- large molecule active agents include, but are not limited to, hematopoietic growth factors, cytokines, and monoclonal and polyclonal antibodies.
- Typical large molecule active agents are biological molecules, such as naturally occurring or synthetic or recombinant proteins.
- the second active agent is stiripentol. cannabidiol, levetiracetam, a gene therapy, 4-phenylbutyrate, adrenocorticotropic hormone (ACTH), topiramate or prednisone.
- cannabidiol cannabidiol
- levetiracetam a gene therapy
- 4-phenylbutyrate 4-phenylbutyrate
- adrenocorticotropic hormone (ACTH) topiramate or prednisone.
- ASOs with varying lengths were designed to the STXBP1 mRNA sequence and then tested in STXBP1 heterozygous iPSC-derived neurons cell line model for resuce of the STXBP1 protein levels.
- An artificial intelligent (Al)-based platform developed by Deep Genomics, Inc. (Toronto, Canada) was used to design ASOs that target STXBP1 mRNA or pre-mRNA and predicted to rescue or increase STXBP1 expression using a variety of different mechanisms.
- the set of 390 ASOs identified in Table 3 were then selected for primary' screening in vitro using an STXBP1 haploinsufficient cell model with a HiBiT-Tag Luciferase System as set out below.
- iPSC-derived neurons obtained under licence from iPS Academia Japan (Kyoto, Japan) were used as a physiologically-relevant model. As iNeurons mature in culture, they form functional pre- and postsynaptic specializations, and by 3 weeks in vitro, they exhibit action potentials, voltage-gated Na+ and K+ currents and evoke excitatory postsynaptic currents (Zhang et al.. Neuron 78, no. 5 (June 2013): 785-98; Meijer et al., Cell Reports 27, no. 7 (May 2019): 2199-221 l.e6). Calcium imaging and multi-electrode array recordings have also shown spontaneous and synaptically-mediated action potentials that are blocked by glutamate receptor antagonist CNQX.
- the model used has a neurogenin-2 (NGN2) transgene under the control of an inducible promoter.
- NGN2 neurogenin-2
- iPSCs with this NGN2 inducible system robustly express neurogenin-2, which rapidly and irreversibly induces their differentiation into excitatory' iNeurons.
- CRISPR-Cas9 was used to edit cells with a guide RNA (5’-TGGTGGATCAGTTAAGCATG-3 ) (SEQ ID NO: 627) that cuts specifically and efficiently at position chr9: 127,653,738 (hg38).
- a guide RNA 5’-TGGTGGATCAGTTAAGCATG-3
- One successfully edited clone (1 A2) showed a 2 nucleotide deletion on one allele, leading to a disruption in the reading frame to make the STXBP1 Het-HiBiT iNeurons (i.e., STXBP1 heterozygous clones).
- STXBP1 Het-HiBiT iNeurons i.e., STXBP1 heterozygous clones.
- a HiBiT tag was introduced at aa527 of the STXBP1 protein of clone 1 A2 (524-531 loop).
- the resulting clone 1A2-E11 was tested for protein stability using the Jess WB system.
- STXBP1 Heterozygous HiBiT-tagged iNeurons showed approximately half the amount of STXBP1 protein when compared to untagged WT iNeurons, indicating that protein stability was not adversely affected by the HiBiT tag.
- STXBP1 Het-HiBiT iNeurons (clone 1A2-E11) were seeded into pre-coated PDL 96-well plates at a density of 25.000 cells/well. On the day after plating, media was removed and ASOs were added at a final concentration of 5 pM. After 10 days, a Cell-Titer Fluor assay was used to estimate the number of viable cells and a HiBiT assay was conducted.
- HiBiT values were normalized to CTF values using a linear regression. Fold change values were calculated relative to the average of non-targeting controls. Data from the HiBiT- tag assay are provided in Table 4 below showing the fold change over a non-targeting control.
- a subset of the ASOs from Table 4 were selected for further validation by ELISA (Example 2) based on reproducability and a fold-change threshold.
- ASOs were screened with ELISA at 5 pM. Briefly, non-HiBiT tagged STXBP1 Het iNeurons were seeded into pre-coated PDL 96-well plates at a density of 50,000 cells/well. On the day after plating, media was removed and ASOs were added at a final concentration of 5 pM. After 17 days, a Cell-Titer Fluor assay was used to estimate the number of viable cells and cells were lysed for ELISA. To avoid artifacts from normalization, concentration values from ELISA were not normalized to the CTF values. Rather, both sets of values were considered individually when evaluating the ASOs. ELISA fold changes were calculated relative to the average of non-targeting controls within the plate. Results are shown in FIGs.
- ASOs provided herein were evaluated with a 6-point, 1 :2 dilution series doseresponse study. Briefly, STXBP1 Het iNeurons were treated by gymnosis with ASOs at 0.3125 pM, 0.625 pM, 1.25 pM, 2.5 pM, 5 pM, and 10 pM. STXBP1 protein levels were measured using the Jess WB system. STXBP1 signals were normalized to a corresponding gamma-tubulin signal as a loading control. Fold change values were calculated relative to non-targeting control at the equivalent dose and displayed in FIGs. 1A-G. Each point represents a technical replicate. Analysis was performed using a median fit to a four- parameter logistic curve. ECso and Ymax. where calculable, are shown in Table 5 below.
- STXBP1 heterozygous (Het) and wild-type (WT) iNeurons were treated in parallel at a 5 pM dose. Data were collected from 2 independent biological replicates, each of which were conducted on different days, used a different batch of neurons and were treated with an independently synthesized lot of ASO compounds. For STXBP1 Het only, data from the 5 pM treatment dose from the doseresponse study (Example 3) were included as an additional third biological replicate.
- STXBP1 protein levels were measured using the Jess WB system.
- STXBP1 signals were normalized to a corresponding gamma-tubulin signal as a loading control.
- Fold change values were calculated relative to the respective STXBP1 Het or WT iNeurons treated with non-targeting control and displayed in the table below.
- a MaxWell high-density multi-electrode array (hd-MEA) system was used to evaluate the effect of ASOs provided herein on synaptic function. As neurons mature in vitro, synapse formation leads to synchronized bursts of action potentials, in which the burst frequency is partially regulated by presynaptic function. Sun and Sudhof (2021) Journal of Neuroscience Methods 349, 109041, previously demonstrated with calcium imaging that burst frequency in STXBP1 heterozygous neurons is reduced when compared to WT neurons.
- STXBP1 Het iNeurons and WT iNeurons were separately co-cultured with human primary' astrocytes at a ratio of 5: 1 (iNeuron: astrocyte). ASOs were added on Day 1 and Day 15 after plating. Following this protocol, unsynchronized action potential activity can typically be detected approximately two weeks after plating. By -17-20 days in vitro, action potentials begin to synchronize, however, burst frequency tends to be inconsistent and fluctuates within minutes. To avoid capturing inaccurate burst frequency data during this period, the burst frequency was measured at 28 and 35 days in vitro (DIV) once bursting activity had stabilized. Burst frequency at both time points is displayed in the table below as a percentage of WT.
- the burst frequency of STXBP1 Het iNeurons treated with nontargeting ASO control at DIV28 is 52.4% of WT, the estimated baseline when evaluating the ASOs provided herein for functional rescue.
- STXBP1 Het iNeurons treated with non-targeting ASO control a two-tailed t-test assuming unequal variance was used (* p ⁇ 0.05, ** p ⁇ 0.01. ***p ⁇ 0.001). The data are provided in Table 7 below.
- Cell viability was evaluated after a 17-day treatment at a 5 pM dose. Cell viability was determined from using a Cell-Titer Fluor Assay. To facilitate comparisons, raw CTF values (in relative fluorescence units) from 4 technical replicates for each of ASOs were replotted in FIGs. 2A-G alongside values from non-targeting ASO controls that were collected from the same plate as the targeted ASO. The cell viability was also calculated as a percentage of the non-targeting ASO controls and displayed in Table 8 below.
- PBMC peripheral blood mononuclear cell
- the ASOs provided herein were tested for STXBP1 RNA modulation, protein increase, synaptic localization, synaptic function, and in vitro network function in neuronal cell cultures. In addition to excitatory neurons that lack one copy of the gene which serve as the baseline, the ASOs provided herein will also be tested in inhibitory neurons and isogenic cell lines engineered with pathogenic patient mutations.
- WT Ngn2-induced neuron (BIONiOlO-C-13), cultured without glia, were treated with vehicle, 5 pM ASOs, and compared with controls: WT isogenic control cell line + vehicle, WT + lenti virus overexpressing STXBP1, or WT + non-targeting control ASO.
- Horizontal lines represent the median of the distribution.
- STXBP1 +/- (CRISPR engineered) Ngn2-induced neuron (BIONi010-C-13), cultured with rat glia, were treated with vehicle, 1 pM, 2.5 pM, 5 pM, or 10 pM ASOs, and compared with controls: HZ + lentivirus overexpressing STXBP1, or HZ + non-targeting control ASO.
- Viohn plots represent the distribution of data points (which are individual fields of view collected across two independent biological replicates). Horizontal lines represent the median of the distribution.
- STXBP1 +/- neurons when cultured with rat glia, and treated with ASOs having SEQ ID NOS: 17 or 197 have significantly elevated synaptic STXBP1 expression compared to treatment with vehicle.
- Experimental parameters Cell type: Ngn2-induced neurons cultured with rat glia (DIV28); Gymnosis; Treatment: ASOs at 5 or 10 pM; immunostaining: 20 days after ASO treatment
- ELISA analysis of bulk STXBP1 protein was performed using a RayBiotech kit (ELH-STXBP1-A, 0621222675), with the calibration curve generated from recombinant human Muncl8-1 protein (Abeam: ab267979, GR3337760-2).
- Each dot represents a cell culture well (technical replicate), and data was collected across three independent biological replicates. Horizontal lines represent the median of the distribution.
- the cynomolgus monkey will used for testing the tolerability of ASO provided herein administered through intrathecal injection.
- ASOs that have perfect homology between human and monkey, we further expect that STXBP1 protein will increase, in vivo. These studies will determine the ASOs tolerability- and pharmacodynamics in a wild type monkey.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Genetics & Genomics (AREA)
- Biomedical Technology (AREA)
- Chemical & Material Sciences (AREA)
- Molecular Biology (AREA)
- Organic Chemistry (AREA)
- Biotechnology (AREA)
- General Engineering & Computer Science (AREA)
- Zoology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Wood Science & Technology (AREA)
- Microbiology (AREA)
- Plant Pathology (AREA)
- Physics & Mathematics (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- Biophysics (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
Claims
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363497836P | 2023-04-24 | 2023-04-24 | |
| US63/497,836 | 2023-04-24 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2024226471A2 true WO2024226471A2 (en) | 2024-10-31 |
| WO2024226471A3 WO2024226471A3 (en) | 2024-12-12 |
Family
ID=91193468
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/025787 Pending WO2024226471A2 (en) | 2023-04-24 | 2024-04-23 | Compositions and methods for treating stxbp1 disorders |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2024226471A2 (en) |
Citations (63)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3536809A (en) | 1969-02-17 | 1970-10-27 | Alza Corp | Medication method |
| US3598123A (en) | 1969-04-01 | 1971-08-10 | Alza Corp | Bandage for administering drugs |
| US3845770A (en) | 1972-06-05 | 1974-11-05 | Alza Corp | Osmatic dispensing device for releasing beneficial agent |
| US3916899A (en) | 1973-04-25 | 1975-11-04 | Alza Corp | Osmotic dispensing device with maximum and minimum sizes for the passageway |
| US4008719A (en) | 1976-02-02 | 1977-02-22 | Alza Corporation | Osmotic system having laminar arrangement for programming delivery of active agent |
| US4044126A (en) | 1972-04-20 | 1977-08-23 | Allen & Hanburys Limited | Steroidal aerosol compositions and process for the preparation thereof |
| US4364923A (en) | 1972-04-20 | 1982-12-21 | Allen & Hanburs Limited | Chemical compounds |
| US4522811A (en) | 1982-07-08 | 1985-06-11 | Syntex (U.S.A.) Inc. | Serial injection of muramyldipeptides and liposomes enhances the anti-infective activity of muramyldipeptides |
| US5059595A (en) | 1989-03-22 | 1991-10-22 | Bioresearch, S.P.A. | Pharmaceutical compositions containing 5-methyltetrahydrofolic acid, 5-formyltetrahydrofolic acid and their pharmaceutically acceptable salts in controlled-release form active in the therapy of organic mental disturbances |
| US5073543A (en) | 1988-07-21 | 1991-12-17 | G. D. Searle & Co. | Controlled release formulations of trophic factors in ganglioside-lipsome vehicle |
| US5120548A (en) | 1989-11-07 | 1992-06-09 | Merck & Co., Inc. | Swelling modulated polymeric drug delivery device |
| US5354556A (en) | 1984-10-30 | 1994-10-11 | Elan Corporation, Plc | Controlled release powder and process for its preparation |
| US5591767A (en) | 1993-01-25 | 1997-01-07 | Pharmetrix Corporation | Liquid reservoir transdermal patch for the administration of ketorolac |
| US5639480A (en) | 1989-07-07 | 1997-06-17 | Sandoz Ltd. | Sustained release formulations of water soluble peptides |
| US5639476A (en) | 1992-01-27 | 1997-06-17 | Euro-Celtique, S.A. | Controlled release formulations coated with aqueous dispersions of acrylic polymers |
| US5674533A (en) | 1994-07-07 | 1997-10-07 | Recordati, S.A., Chemical And Pharmaceutical Company | Pharmaceutical composition for the controlled release of moguisteine in a liquid suspension |
| US5709874A (en) | 1993-04-14 | 1998-01-20 | Emory University | Device for local drug delivery and methods for using the same |
| US5733566A (en) | 1990-05-15 | 1998-03-31 | Alkermes Controlled Therapeutics Inc. Ii | Controlled release of antiparasitic agents in animals |
| US5739108A (en) | 1984-10-04 | 1998-04-14 | Monsanto Company | Prolonged release of biologically active polypeptides |
| US5759542A (en) | 1994-08-05 | 1998-06-02 | New England Deaconess Hospital Corporation | Compositions and methods for the delivery of drugs by platelets for the treatment of cardiovascular and other diseases |
| US5840674A (en) | 1990-11-01 | 1998-11-24 | Oregon Health Sciences University | Covalent microparticle-drug conjugates for biological targeting |
| US5891474A (en) | 1997-01-29 | 1999-04-06 | Poli Industria Chimica, S.P.A. | Time-specific controlled release dosage formulations and method of preparing same |
| US5900252A (en) | 1990-04-17 | 1999-05-04 | Eurand International S.P.A. | Method for targeted and controlled release of drugs in the intestinal tract and more particularly in the colon |
| US5922356A (en) | 1996-10-09 | 1999-07-13 | Sumitomo Pharmaceuticals Company, Limited | Sustained release formulation |
| US5972891A (en) | 1992-12-07 | 1999-10-26 | Takeda Chemical Industries, Ltd. | Sustained-release preparation |
| US5972366A (en) | 1994-11-28 | 1999-10-26 | The Unites States Of America As Represented By The Secretary Of The Army | Drug releasing surgical implant or dressing material |
| US5980945A (en) | 1996-01-16 | 1999-11-09 | Societe De Conseils De Recherches Et D'applications Scientifique S.A. | Sustained release drug formulations |
| US5985307A (en) | 1993-04-14 | 1999-11-16 | Emory University | Device and method for non-occlusive localized drug delivery |
| US5993855A (en) | 1995-09-18 | 1999-11-30 | Shiseido Company, Ltd. | Delayed drug-releasing microspheres |
| US6004534A (en) | 1993-07-23 | 1999-12-21 | Massachusetts Institute Of Technology | Targeted polymerized liposomes for improved drug delivery |
| US6039975A (en) | 1995-10-17 | 2000-03-21 | Hoffman-La Roche Inc. | Colon targeted delivery system |
| US6045830A (en) | 1995-09-04 | 2000-04-04 | Takeda Chemical Industries, Ltd. | Method of production of sustained-release preparation |
| US6048736A (en) | 1998-04-29 | 2000-04-11 | Kosak; Kenneth M. | Cyclodextrin polymers for carrying and releasing drugs |
| US6060082A (en) | 1997-04-18 | 2000-05-09 | Massachusetts Institute Of Technology | Polymerized liposomes targeted to M cells and useful for oral or mucosal drug delivery |
| US6071495A (en) | 1989-12-22 | 2000-06-06 | Imarx Pharmaceutical Corp. | Targeted gas and gaseous precursor-filled liposomes |
| US6087324A (en) | 1993-06-24 | 2000-07-11 | Takeda Chemical Industries, Ltd. | Sustained-release preparation |
| US6113943A (en) | 1996-10-31 | 2000-09-05 | Takeda Chemical Industries, Ltd. | Sustained-release preparation capable of releasing a physiologically active substance |
| US6120751A (en) | 1997-03-21 | 2000-09-19 | Imarx Pharmaceutical Corp. | Charged lipids and uses for the same |
| US6131570A (en) | 1998-06-30 | 2000-10-17 | Aradigm Corporation | Temperature controlling device for aerosol drug delivery |
| US6139865A (en) | 1996-10-01 | 2000-10-31 | Eurand America, Inc. | Taste-masked microcapsule compositions and methods of manufacture |
| US6197350B1 (en) | 1996-12-20 | 2001-03-06 | Takeda Chemical Industries, Ltd. | Method of producing a sustained-release preparation |
| US6248363B1 (en) | 1999-11-23 | 2001-06-19 | Lipocine, Inc. | Solid carriers for improved delivery of active ingredients in pharmaceutical compositions |
| US6253872B1 (en) | 1996-05-29 | 2001-07-03 | Gmundner Fertigteile Gesellschaft M.B.H & Co., Kg | Track soundproofing arrangement |
| US6264970B1 (en) | 1996-06-26 | 2001-07-24 | Takeda Chemical Industries, Ltd. | Sustained-release preparation |
| US6267981B1 (en) | 1995-06-27 | 2001-07-31 | Takeda Chemical Industries, Ltd. | Method of producing sustained-release preparation |
| US6271359B1 (en) | 1999-04-14 | 2001-08-07 | Musc Foundation For Research Development | Tissue-specific and pathogen-specific toxic agents and ribozymes |
| US6274552B1 (en) | 1993-03-18 | 2001-08-14 | Cytimmune Sciences, Inc. | Composition and method for delivery of biologically-active factors |
| US6316652B1 (en) | 1995-06-06 | 2001-11-13 | Kosta Steliou | Drug mitochondrial targeting agents |
| US6419961B1 (en) | 1996-08-29 | 2002-07-16 | Takeda Chemical Industries, Ltd. | Sustained release microcapsules of a bioactive substance and a biodegradable polymer |
| US6589548B1 (en) | 1998-05-16 | 2003-07-08 | Mogam Biotechnology Research Institute | Controlled drug delivery system using the conjugation of drug to biodegradable polyester |
| US6613358B2 (en) | 1998-03-18 | 2003-09-02 | Theodore W. Randolph | Sustained-release composition including amorphous polymer |
| US6740634B1 (en) | 1998-01-16 | 2004-05-25 | Takeda Chemical Industries, Ltd. | Sustained release compositions, process for producing the same and utilization thereof |
| WO2011097641A1 (en) | 2010-02-08 | 2011-08-11 | Isis Pharmaceuticals, Inc. | Methods and compositions useful in treatment of diseases or conditions related to repeat expansion |
| WO2013096899A2 (en) | 2011-12-23 | 2013-06-27 | Shire Human Genetic Therapies, Inc. | Stable formulations for cns delivery of arylsulfatase a |
| WO2014126229A1 (en) | 2013-02-18 | 2014-08-21 | 塩野義製薬株式会社 | Nucleoside and nucleotide, having nitrogen-containing hetercycle structure |
| WO2014145356A1 (en) | 2013-03-15 | 2014-09-18 | MiRagen Therapeutics, Inc. | Bridged bicyclic nucleosides |
| WO2015142910A1 (en) | 2014-03-17 | 2015-09-24 | Isis Pharmaceuticals, Inc. | Bicyclic carbocyclic nucleosides and oligomeric compounds prepared therefrom |
| WO2016017422A1 (en) | 2014-07-31 | 2016-02-04 | 国立大学法人大阪大学 | Crosslinking nucleoside and nucleotide |
| WO2020028832A1 (en) | 2018-08-02 | 2020-02-06 | Dyne Therapeutics, Inc. | Muscle targeting complexes and uses thereof for treating dystrophinopathies |
| WO2021142307A1 (en) | 2020-01-10 | 2021-07-15 | Dyne Therapeutics, Inc. | Muscle targeting complexes and uses thereof for treating dystrophinopathies |
| WO2022020107A1 (en) | 2020-07-23 | 2022-01-27 | Dyne Therapeutics, Inc. | Muscle targeting complexes and uses thereof for treating dystrophinopathies |
| WO2022140535A1 (en) | 2020-12-23 | 2022-06-30 | Sarepta Therapeutics, Inc. | Compositions comprising exon skipping oligonucleotide conjugates for treating muscular dystrophy |
| WO2022192749A2 (en) | 2021-03-12 | 2022-09-15 | Pepgen Inc. | Methods of treating duchenne muscular dystrophy using peptide-oligonucleotide conjugates |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP4104867A3 (en) * | 2015-12-14 | 2023-03-01 | Cold Spring Harbor Laboratory | Compositions and methods for treatment of central nervous system diseases |
| US20220228146A1 (en) * | 2019-05-06 | 2022-07-21 | The Children's Hospital Of Philadelphia | Micro-rna site blocking oligonucleotides for the treatment of epileptic encephalopathy and neurodevelopmental disorders |
| CA3233095A1 (en) * | 2021-09-23 | 2023-03-30 | Q-State Biosciences, Inc. | Therapeutics for haploinsufficiency conditions |
-
2024
- 2024-04-23 WO PCT/US2024/025787 patent/WO2024226471A2/en active Pending
Patent Citations (66)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3536809A (en) | 1969-02-17 | 1970-10-27 | Alza Corp | Medication method |
| US3598123A (en) | 1969-04-01 | 1971-08-10 | Alza Corp | Bandage for administering drugs |
| US4044126A (en) | 1972-04-20 | 1977-08-23 | Allen & Hanburys Limited | Steroidal aerosol compositions and process for the preparation thereof |
| US4364923A (en) | 1972-04-20 | 1982-12-21 | Allen & Hanburs Limited | Chemical compounds |
| US4414209A (en) | 1972-04-20 | 1983-11-08 | Allen & Hanburys Limited | Micronized aerosol steroids |
| US3845770A (en) | 1972-06-05 | 1974-11-05 | Alza Corp | Osmatic dispensing device for releasing beneficial agent |
| US3916899A (en) | 1973-04-25 | 1975-11-04 | Alza Corp | Osmotic dispensing device with maximum and minimum sizes for the passageway |
| US4008719A (en) | 1976-02-02 | 1977-02-22 | Alza Corporation | Osmotic system having laminar arrangement for programming delivery of active agent |
| US4522811A (en) | 1982-07-08 | 1985-06-11 | Syntex (U.S.A.) Inc. | Serial injection of muramyldipeptides and liposomes enhances the anti-infective activity of muramyldipeptides |
| US5739108A (en) | 1984-10-04 | 1998-04-14 | Monsanto Company | Prolonged release of biologically active polypeptides |
| US5354556A (en) | 1984-10-30 | 1994-10-11 | Elan Corporation, Plc | Controlled release powder and process for its preparation |
| US5073543A (en) | 1988-07-21 | 1991-12-17 | G. D. Searle & Co. | Controlled release formulations of trophic factors in ganglioside-lipsome vehicle |
| US5059595A (en) | 1989-03-22 | 1991-10-22 | Bioresearch, S.P.A. | Pharmaceutical compositions containing 5-methyltetrahydrofolic acid, 5-formyltetrahydrofolic acid and their pharmaceutically acceptable salts in controlled-release form active in the therapy of organic mental disturbances |
| US5639480A (en) | 1989-07-07 | 1997-06-17 | Sandoz Ltd. | Sustained release formulations of water soluble peptides |
| US5120548A (en) | 1989-11-07 | 1992-06-09 | Merck & Co., Inc. | Swelling modulated polymeric drug delivery device |
| US6071495A (en) | 1989-12-22 | 2000-06-06 | Imarx Pharmaceutical Corp. | Targeted gas and gaseous precursor-filled liposomes |
| US5900252A (en) | 1990-04-17 | 1999-05-04 | Eurand International S.P.A. | Method for targeted and controlled release of drugs in the intestinal tract and more particularly in the colon |
| US5733566A (en) | 1990-05-15 | 1998-03-31 | Alkermes Controlled Therapeutics Inc. Ii | Controlled release of antiparasitic agents in animals |
| US5840674A (en) | 1990-11-01 | 1998-11-24 | Oregon Health Sciences University | Covalent microparticle-drug conjugates for biological targeting |
| US5639476A (en) | 1992-01-27 | 1997-06-17 | Euro-Celtique, S.A. | Controlled release formulations coated with aqueous dispersions of acrylic polymers |
| US5972891A (en) | 1992-12-07 | 1999-10-26 | Takeda Chemical Industries, Ltd. | Sustained-release preparation |
| US5591767A (en) | 1993-01-25 | 1997-01-07 | Pharmetrix Corporation | Liquid reservoir transdermal patch for the administration of ketorolac |
| US6274552B1 (en) | 1993-03-18 | 2001-08-14 | Cytimmune Sciences, Inc. | Composition and method for delivery of biologically-active factors |
| US5709874A (en) | 1993-04-14 | 1998-01-20 | Emory University | Device for local drug delivery and methods for using the same |
| US5985307A (en) | 1993-04-14 | 1999-11-16 | Emory University | Device and method for non-occlusive localized drug delivery |
| US6376461B1 (en) | 1993-06-24 | 2002-04-23 | Takeda Chemical Industries, Ltd. | Sustained-release preparation |
| US6087324A (en) | 1993-06-24 | 2000-07-11 | Takeda Chemical Industries, Ltd. | Sustained-release preparation |
| US6004534A (en) | 1993-07-23 | 1999-12-21 | Massachusetts Institute Of Technology | Targeted polymerized liposomes for improved drug delivery |
| US5674533A (en) | 1994-07-07 | 1997-10-07 | Recordati, S.A., Chemical And Pharmaceutical Company | Pharmaceutical composition for the controlled release of moguisteine in a liquid suspension |
| US5759542A (en) | 1994-08-05 | 1998-06-02 | New England Deaconess Hospital Corporation | Compositions and methods for the delivery of drugs by platelets for the treatment of cardiovascular and other diseases |
| US5972366A (en) | 1994-11-28 | 1999-10-26 | The Unites States Of America As Represented By The Secretary Of The Army | Drug releasing surgical implant or dressing material |
| US6316652B1 (en) | 1995-06-06 | 2001-11-13 | Kosta Steliou | Drug mitochondrial targeting agents |
| US6267981B1 (en) | 1995-06-27 | 2001-07-31 | Takeda Chemical Industries, Ltd. | Method of producing sustained-release preparation |
| US6045830A (en) | 1995-09-04 | 2000-04-04 | Takeda Chemical Industries, Ltd. | Method of production of sustained-release preparation |
| US5993855A (en) | 1995-09-18 | 1999-11-30 | Shiseido Company, Ltd. | Delayed drug-releasing microspheres |
| US6039975A (en) | 1995-10-17 | 2000-03-21 | Hoffman-La Roche Inc. | Colon targeted delivery system |
| US5980945A (en) | 1996-01-16 | 1999-11-09 | Societe De Conseils De Recherches Et D'applications Scientifique S.A. | Sustained release drug formulations |
| US6253872B1 (en) | 1996-05-29 | 2001-07-03 | Gmundner Fertigteile Gesellschaft M.B.H & Co., Kg | Track soundproofing arrangement |
| US6264970B1 (en) | 1996-06-26 | 2001-07-24 | Takeda Chemical Industries, Ltd. | Sustained-release preparation |
| US6419961B1 (en) | 1996-08-29 | 2002-07-16 | Takeda Chemical Industries, Ltd. | Sustained release microcapsules of a bioactive substance and a biodegradable polymer |
| US6139865A (en) | 1996-10-01 | 2000-10-31 | Eurand America, Inc. | Taste-masked microcapsule compositions and methods of manufacture |
| US5922356A (en) | 1996-10-09 | 1999-07-13 | Sumitomo Pharmaceuticals Company, Limited | Sustained release formulation |
| US6699500B2 (en) | 1996-10-31 | 2004-03-02 | Takeda Chemical Industries, Ltd. | Sustained-release preparation capable of releasing a physiologically active substance |
| US6113943A (en) | 1996-10-31 | 2000-09-05 | Takeda Chemical Industries, Ltd. | Sustained-release preparation capable of releasing a physiologically active substance |
| US6197350B1 (en) | 1996-12-20 | 2001-03-06 | Takeda Chemical Industries, Ltd. | Method of producing a sustained-release preparation |
| US5891474A (en) | 1997-01-29 | 1999-04-06 | Poli Industria Chimica, S.P.A. | Time-specific controlled release dosage formulations and method of preparing same |
| US6120751A (en) | 1997-03-21 | 2000-09-19 | Imarx Pharmaceutical Corp. | Charged lipids and uses for the same |
| US6060082A (en) | 1997-04-18 | 2000-05-09 | Massachusetts Institute Of Technology | Polymerized liposomes targeted to M cells and useful for oral or mucosal drug delivery |
| US6740634B1 (en) | 1998-01-16 | 2004-05-25 | Takeda Chemical Industries, Ltd. | Sustained release compositions, process for producing the same and utilization thereof |
| US6613358B2 (en) | 1998-03-18 | 2003-09-02 | Theodore W. Randolph | Sustained-release composition including amorphous polymer |
| US6048736A (en) | 1998-04-29 | 2000-04-11 | Kosak; Kenneth M. | Cyclodextrin polymers for carrying and releasing drugs |
| US6589548B1 (en) | 1998-05-16 | 2003-07-08 | Mogam Biotechnology Research Institute | Controlled drug delivery system using the conjugation of drug to biodegradable polyester |
| US6131570A (en) | 1998-06-30 | 2000-10-17 | Aradigm Corporation | Temperature controlling device for aerosol drug delivery |
| US6271359B1 (en) | 1999-04-14 | 2001-08-07 | Musc Foundation For Research Development | Tissue-specific and pathogen-specific toxic agents and ribozymes |
| US6248363B1 (en) | 1999-11-23 | 2001-06-19 | Lipocine, Inc. | Solid carriers for improved delivery of active ingredients in pharmaceutical compositions |
| WO2011097641A1 (en) | 2010-02-08 | 2011-08-11 | Isis Pharmaceuticals, Inc. | Methods and compositions useful in treatment of diseases or conditions related to repeat expansion |
| WO2013096899A2 (en) | 2011-12-23 | 2013-06-27 | Shire Human Genetic Therapies, Inc. | Stable formulations for cns delivery of arylsulfatase a |
| WO2014126229A1 (en) | 2013-02-18 | 2014-08-21 | 塩野義製薬株式会社 | Nucleoside and nucleotide, having nitrogen-containing hetercycle structure |
| WO2014145356A1 (en) | 2013-03-15 | 2014-09-18 | MiRagen Therapeutics, Inc. | Bridged bicyclic nucleosides |
| WO2015142910A1 (en) | 2014-03-17 | 2015-09-24 | Isis Pharmaceuticals, Inc. | Bicyclic carbocyclic nucleosides and oligomeric compounds prepared therefrom |
| WO2016017422A1 (en) | 2014-07-31 | 2016-02-04 | 国立大学法人大阪大学 | Crosslinking nucleoside and nucleotide |
| WO2020028832A1 (en) | 2018-08-02 | 2020-02-06 | Dyne Therapeutics, Inc. | Muscle targeting complexes and uses thereof for treating dystrophinopathies |
| WO2021142307A1 (en) | 2020-01-10 | 2021-07-15 | Dyne Therapeutics, Inc. | Muscle targeting complexes and uses thereof for treating dystrophinopathies |
| WO2022020107A1 (en) | 2020-07-23 | 2022-01-27 | Dyne Therapeutics, Inc. | Muscle targeting complexes and uses thereof for treating dystrophinopathies |
| WO2022140535A1 (en) | 2020-12-23 | 2022-06-30 | Sarepta Therapeutics, Inc. | Compositions comprising exon skipping oligonucleotide conjugates for treating muscular dystrophy |
| WO2022192749A2 (en) | 2021-03-12 | 2022-09-15 | Pepgen Inc. | Methods of treating duchenne muscular dystrophy using peptide-oligonucleotide conjugates |
Non-Patent Citations (46)
| Title |
|---|
| "NCBI", Database accession no. NC_000009, 1 2 |
| "Response Evaluation Criteria in Solid Tumors (RECIST) Guidelines", JOURNAL OF THE NATIONAL CANCER INSTITUTE, vol. 92, no. 3, 2000, pages 205 - 216 |
| ABRAMOV D ET AL., J. NEUROCHEM., vol. 157, 2021, pages 165 - 78 |
| ALLEN AS ET AL., NATURE, vol. 501, 2013, pages 217 - 21 |
| ALTSCHUL, S. F. ET AL., J. MOL. BIOL., vol. 215, 1990, pages 403 - 410 |
| BEAUCAGE ET AL., TETRAHEDRON, vol. 48, no. 12, 1992, pages 2223 |
| BOLLI ET AL., CHEM BIOL., vol. 3, no. 3, March 1996 (1996-03-01), pages 197 - 206 |
| BUCHWALD ET AL., SURGERY, vol. 88, 1980, pages 507 |
| CARVILL GL ET AL., NEUROLOGY, vol. 82, 2014, pages 1245 - 53 |
| CHEN W ET AL., ELIFE, vol. 9, 2020, pages e48705 |
| DEPREZ L ET AL., NEUROLOGY, vol. 75, 2010, pages 1159 - 65 |
| DEVERE , J. ET AL., NUCLEIC ACIDS RESEARCH, vol. 12, no. 1, 1984, pages 387 |
| DULUBOVA 1 ET AL., PNAS, vol. 104, 2007, pages 2697 - 702 |
| GOODSON, MEDICAL APPLICATIONS OF CONTROLLED RELEASE, vol. 2, 1984, pages 115 - 138 |
| GRONE BP ET AL., PLOS ONE, vol. 11, 2016, pages e0151148 |
| GUIBERSON NGL ET AL., NATURE COMM, vol. 9, 2018, pages 3986 - 4009 |
| HAMDAN FF ET AL., EUR. J. HUM. GENET., vol. 19, 2011, pages 607 - 9 |
| HAMILTON G S, BIOLOGICALS, vol. 3, no. 5, 4 September 2015 (2015-09-04), pages 318 - 32 |
| HANESSIAN ET AL., J. ORG. CHEM., vol. 78, no. 18, 2013, pages 9064 - 9075 |
| JENS T. CARSTENSEN: "Drug Stability: Principles & Practice", 1995, MARCEL DEKKER, pages: 379 - 80 |
| KIM E GKIM K M, BIOMOL. THER. (SEOUL, vol. 23, no. 6, November 2015 (2015-11-01), pages 493 - 509 |
| KOVACEVIK J ET AL., BRAIN, vol. 141, 2018, pages 1350 - 1374 |
| LANGER, SCIENCE, vol. 249, 1990, pages 1527 - 1533 |
| LOPEZ-RIVERA JA ET AL., BRAIN, vol. 143, 2020, pages 1099 - 1105 |
| MEIJER ET AL., CELL REPORTS, vol. 27, no. 7, May 2019 (2019-05-01), pages 2199 - 2211 |
| MISURA KM ET AL., NATURE, vol. 404, 2000, pages 355 - 62 |
| MURRAY ET AL., NUCL. ACIDS RES., vol. 40, no. 13, 2012, pages 6135 - 6143 |
| NEEDLEMANWUNSCH, J. MOL. BIOL., vol. 48, 1970, pages 443 - 453 |
| NISHIDA ET AL., CHEM. COMMUN., vol. 46, 2010, pages 5283 |
| OLSEN DJORGENSEN J T, FRONT. ONCOL., vol. 4, 16 May 2014 (2014-05-16), pages 105 |
| OSAWA ET AL., J. ORG. CHEM., vol. 80, no. 21, 2015, pages 10474 - 10481 |
| OTSUKA M ET AL., EPILEPSIA, vol. 51, 2010, pages 2449 - 52 |
| PETERS CBROWN S, BIOSCI. REP., vol. 35, no. 4, 12 June 2015 (2015-06-12), pages e00225 |
| REESE, ORG. & BIOMOL. CHEM., vol. 3, no. 21, 2005, pages 3851 - 3868 |
| RIZO J ET AL., ANNU. REV. BIOPHYS., vol. 44, 2015, pages 339 - 67 |
| SAITSU H ET AL., NAT GENET, vol. 40, no. 6, 2008, pages 782 - 8 |
| SAUDEK ET AL., N. ENGL. J. MED., vol. 321, 1989, pages 574 |
| SEFTON, CRC CRIT. REF. BIOMED. ENG., vol. 14, 1987, pages 201 |
| SETH ET AL., J. ORG. CHEM., vol. 75, 2010, pages 1569 - 1581 |
| SHEN J. ET AL., CELL, vol. 128, 2006, pages 183 - 95 |
| STAMBERGER H ET AL., NEUROLOGY, vol. 86, 2016, pages 954 - 62 |
| SUNSUDHOF, JOURNAL OF NEUROSCIENCE METHODS, vol. 349, 2021, pages 109041 |
| SYMONDS JD ET AL., EUR. J. PAEDIATR. NEUROL., vol. 24, 2020, pages 15 - 23 |
| XIAN J ET AL., BRAIN, 2021, pages awab327, Retrieved from the Internet <URL:https://doi.org/10.1093/brain/awab327> |
| YAMAMOTO ET AL., ORG. BIOMOL. CHEM., vol. 13, 2015, pages 3757 |
| ZHANG ET AL., NEURON, vol. 78, no. 5, June 2013 (2013-06-01), pages 785 - 98 |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2024226471A3 (en) | 2024-12-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Lyu et al. | Microglial responses to brain injury and disease: functional diversity and new opportunities | |
| US9539307B2 (en) | Compositions and methods for treating amyotrophic lateral sclerosis | |
| WO2019246262A2 (en) | Methods of treating or inhibiting onset of huntington's disease | |
| US20200016189A1 (en) | Method for treating schizophrenia | |
| JP2025114630A (en) | Treatment and detection of hereditary neuropathies and related disorders | |
| US20220380777A1 (en) | Exon skipping of fc-epsilon-ri-beta and ms4a6a in combination for the treatment of allergic diseases | |
| RU2663100C2 (en) | Mi-rna and their use in methods and compositions for treatment and / or prevention of eye conditions | |
| JP2022537581A (en) | PPM1 inhibitors and methods of use thereof | |
| JP2025525099A (en) | Compounds for the Treatment of Conditions and Diseases | |
| EP3622958A1 (en) | Use of potassium ion channel inhibitor for treatment of depression and pharmaceutical composition | |
| US20140161769A1 (en) | Methods for treating inflammatory autoimmune disorders | |
| US20220372492A1 (en) | Compounds for modulating fc-epsilon-ri-beta expression and uses thereof | |
| WO2024226471A2 (en) | Compositions and methods for treating stxbp1 disorders | |
| US20110313016A1 (en) | Treatment of Intestinal Conditions | |
| Crosson et al. | IL-13 promotes sensory-sympathetic neurons crosstalk in asthma | |
| KR20250155076A (en) | Antisense oligomer formulations | |
| WO2008007131A2 (en) | Treatment for demyelinating disease | |
| US20250136980A1 (en) | Modulation of microrna-335-5p for the treatment of sodium channelopathies | |
| Orozco-Suárez et al. | Epilepsy: Experimental and Human Studies | |
| US20250283086A1 (en) | Compositions and methods for treatment of kidney disease | |
| Orozco-Suárez et al. | GABAergic Neurotransmission Abnormalities in Pharmacoresistant Epilepsy: Experimental and Human Studies | |
| TW202346585A (en) | Methods and compositions for treating epilepsy | |
| WO2025111653A1 (en) | Improved compositions and methods for treatment of monogenic neurodevelopmental disorders | |
| TW202515583A (en) | Oligonucleotides for modulating kcnt1 expression | |
| EP4642913A2 (en) | Splice-switching oligonucleotides for treating syngap1-associated disorders |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24727865 Country of ref document: EP Kind code of ref document: A2 |
|
| ENP | Entry into the national phase |
Ref document number: 2024727865 Country of ref document: EP Effective date: 20251124 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2024727865 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2024727865 Country of ref document: EP Effective date: 20251124 |
|
| ENP | Entry into the national phase |
Ref document number: 2024727865 Country of ref document: EP Effective date: 20251124 |
|
| ENP | Entry into the national phase |
Ref document number: 2024727865 Country of ref document: EP Effective date: 20251124 |