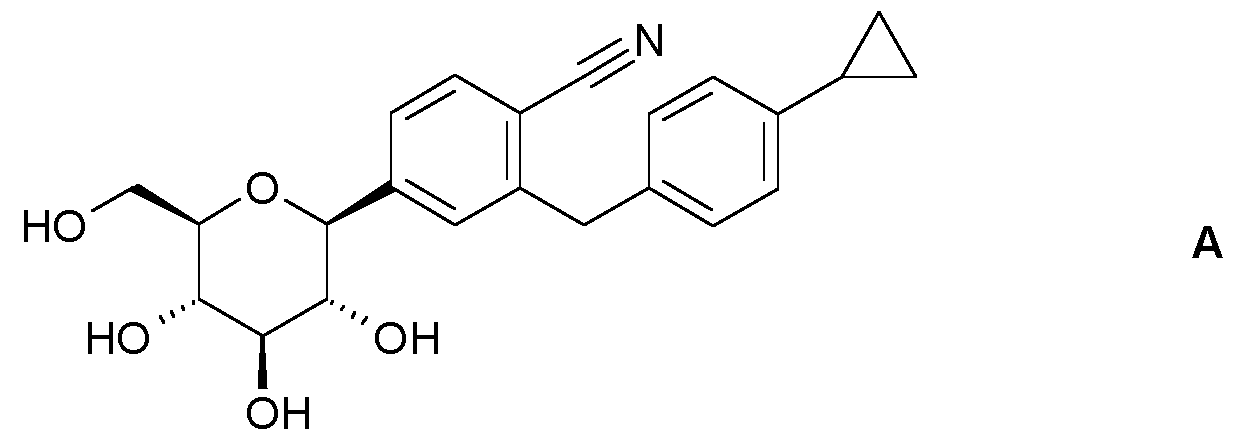
WO2024223388A1 - Crystalline complexes between velagliflozin and selected co-crystal forming agents, methods for their preparation and the use thereof for preparing medicaments - Google Patents
Crystalline complexes between velagliflozin and selected co-crystal forming agents, methods for their preparation and the use thereof for preparing medicaments Download PDFInfo
- Publication number
- WO2024223388A1 WO2024223388A1 PCT/EP2024/060490 EP2024060490W WO2024223388A1 WO 2024223388 A1 WO2024223388 A1 WO 2024223388A1 EP 2024060490 W EP2024060490 W EP 2024060490W WO 2024223388 A1 WO2024223388 A1 WO 2024223388A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- degrees
- ray powder
- powder diffraction
- diffraction pattern
- crystalline
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D309/00—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only ring hetero atom, not condensed with other rings
- C07D309/02—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only ring hetero atom, not condensed with other rings having no double bonds between ring members or between ring members and non-ring members
- C07D309/08—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only ring hetero atom, not condensed with other rings having no double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D309/10—Oxygen atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
Definitions
- the invention relates to the field of medicine, particularly veterinary medicine.
- the invention relates to crystalline complexes of velagliflozin and selected co-crystal forming agents, to methods for their preparation as well as to the use thereof for preparing medicaments.
- the compounds described in these two WO publications have a valuable inhibitory effect on the sodium-dependent glucose cotransporter SGLT, particularly SGLT-2.
- the methods of manufacture of velagliflozin as described therein do not yield crystalline forms nor do they yield crystalline complexes between velagliflozin and selected co-crystal forming agents.
- a certain pharmaceutical activity is of course the basic prerequisite to be fulfilled by a pharmaceutically active agent before same is approved as a medicament on the market.
- a pharmaceutically active agent has to comply with. These requirements are based on various parameters which are connected with the nature of the active substance itself. Without being restrictive, examples of these parameters are the stability of the active agent under various environmental conditions, its stability during production of the pharmaceutical formulation and the stability of the active agent in the final medicament compositions.
- the pharmaceutically active substance used for preparing the pharmaceutical compositions should be as pure as possible and its stability in long-term storage must be guaranteed under various environmental conditions. This is essential to prevent the use of pharmaceutical compositions which contain, in addition to the actual active substance, breakdown products thereof, for example. In such cases the content of active substance in the medicament might be less than that specified.
- Uniform distribution of the medicament in the formulation is a critical factor, particularly when the medicament has to be given in low doses.
- the particle size of the active substance can be reduced to a suitable level, e.g. by grinding. Since breakdown of the pharmaceutically active substance as a side effect of the grinding (or micronising) has to be avoided as far as possible, in spite of the hard conditions required during the process, it is essential that the active substance should be highly stable throughout the grinding process. Only if the active substance is sufficiently stable during the grinding process it is possible to produce a homogeneous pharmaceutical formulation which always contains the specified amount of active substance in a reproducible manner.
- Another problem which may arise in the grinding process for preparing the desired pharmaceutical formulation is the input of energy caused by this process and the stress on the surface of the crystals. This may in certain circumstances lead to polymorphous changes, to amorphization or to a change in the crystal lattice. Since the pharmaceutical quality of a pharmaceutical formulation requires that the active substance should always have the same crystalline morphology, the stability and properties of the crystalline active substance are subject to stringent requirements from this point of view as well.
- the stability of a pharmaceutically active substance is also important in pharmaceutical compositions for determining the shelf life of the particular medicament; the shelf life is the length of time during which the medicament can be administered without any risk. High stability of a medicament in the abovementioned pharmaceutical compositions under various storage conditions is therefore an additional advantage for both the patient and the manufacturer.
- compositions with a tendency to absorb moisture have to be protected from moisture during storage, e.g. by the addition of suitable drying agents or by storing the drug in an environment where it is protected from moisture.
- a pharmaceutically active substance should be at best slightly hygroscopic.
- WO 2014/016381 discloses crystalline complexes of velagliflozin with in particular L-proline, preferably as hydrate form.
- the preferred crystalline complex described therein is a monohydrate co-crystal and characterized by a molar ratio of velagliflozin and L-proline and crystal water of 1 : 1 : 1.
- WO 2023/006718 discloses the use of one or more SGLT-2 inhibitors or pharmaceutically acceptable forms thereof for the prophylaxis and/or treatment of one or more cardiac diseases in a non-human mammal / non- human mammal patient excluding a feline, in particular a canine / canine patient.
- the objective of the invention is to provide new, stable crystalline forms of velagliflozin, which meet important requirements imposed on pharmaceutically active substances as those mentioned above.
- the present invention relates to a crystalline complex between velagliflozin and a co-crystal forming agent.
- a second aspect of the present invention relates to a pharmaceutical composition or medicament comprising one or more crystalline complexes as defined hereinbefore and hereinafter and/or mixtures of crystalline forms as defined hereinbefore and hereinafter.
- a further aspect of the present invention relates to the herein defined crystalline complexes or the herein defined mixtures of crystalline forms for use as a medicament.
- the present invention relates to one or more crystalline complexes as defined hereinbefore or hereinafter and/or the pharmaceutical composition as defined hereinbefore or hereinafter and/or the medicament as defined hereinbefore or hereinafter for use in a method of treatment and/or prevention of diseases or conditions, which can be influenced by inhibiting sodium-dependent glucose cotransporter SGLT, preferably SGLT-2.
- a corresponding method of treatment and/or prevention of diseases or conditions which can be influenced by inhibiting sodium-dependent glucose cotransporter SGLT, preferably SGLT-2, as well as the corresponding use for the preparation of a medicament for the prevention and/or treatment of diseases or conditions, which can be influenced by inhibiting sodium-dependent glucose cotransporter SGLT, preferably SGLT-2, are also intended to be comprised by the present invention.
- the present invention relates to one or more crystalline complexes as defined hereinbefore or hereinafter and/or the pharmaceutical composition as defined hereinbefore or hereinafter and/or the medicament as defined hereinbefore or hereinafter for use in a method of inhibiting the sodium-dependent glucose cotransporter SGLT-2.
- the present invention relates to a method for making one or more crystalline complexes as defined hereinbefore and hereinafter, said method comprising the following steps: (a) preparing a solution of velagliflozin and the co-crystal forming agent in a solvent or a mixture of solvents;
- Figure 1 A shows an X-ray powder diffraction pattern of a representative batch of the crystalline complex of velagliflozin with L-proline (1:3), FORM 1000.
- Figure IB shows the determination of the melting point via DSC and of the weight loss via TG of a representative batch of the crystalline complex of velagliflozin with L-proline (1:3), FORM 1000.
- Figure 2A shows an X-ray powder diffraction pattern of a representative batch of the crystalline complex of velagliflozin with L-proline (1:1 - 1:1.3), FORM 1001.
- Figure 2B shows the determination of the melting point via DSC and of the weight loss via TG of a representative batch of the crystalline complex of velagliflozin with L-proline (1:1 - 1:1.3), FORM 1001.
- Figure 3 A shows an X-ray powder diffraction pattern of a representative batch of the crystalline complex of velagliflozin with L-proline (1:1.5), FORM 1002.
- Figure 3B shows the determination of the melting point via DSC and of the weight loss via TG of a representative batch of the crystalline complex of velagliflozin with L-proline (1:1.5), FORM 1002.
- Figure 4A shows an X-ray powder diffraction pattern of a representative batch of the crystalline complex of velagliflozin with L-proline (1:1), FORM 1003.
- Figure 4B shows the determination of the melting point via DSC and of the weight loss via TG of a representative batch of the crystalline complex of velagliflozin with L-proline (1:1), FORM 1003.
- Figure 5A shows an X-ray powder diffraction pattern of a representative batch of the crystalline complex of velagliflozin with L-proline (1:2), FORM 1004.
- Figure 5B shows the determination of the melting point via DSC and of the weight loss via TG of a representative batch of the crystalline complex of velagliflozin with L-proline (1:2), FORM 1004.
- Figure 6A shown an X-ray powder diffraction pattern of a representative batch of the crystalline complex of velagliflozin with L-proline (1:1), FORM 1005.
- Figure 7A shown an X-ray powder diffraction pattern of a representative batch of the crystalline complex of velagliflozin with L-proline (1:1.4 - 1:1.8), FORM 1006.
- Figure 7B shows the determination of the melting point via DSC and of the weight loss via TG of a representative batch of the crystalline complex of velagliflozin with L-proline (1:1.4 - 1:1.8), FORM 1006.
- Figure 8A shown an X-ray powder diffraction pattern of a representative batch of the crystalline complex of velagliflozin with L-proline (1:1), FORM 1007.
- Figure 8B shows the determination of the melting point via DSC of a representative batch of the crystalline complex of velagliflozin with L-proline (1:1), FORM 1007.
- Figure 9A shown an X-ray powder diffraction pattern of a representative batch of the crystalline complex of velagliflozin with L-proline (1:1) in tetrahydrofuran, FORM 1008.
- Figure 9B shows the determination of the melting point via DSC and of the weight loss via TG of a representative batch of the crystalline complex of velagliflozin with L-proline (1 : 1) in tetrahydrofuran, FORM 1008.
- Figure 10A shown an X-ray powder diffraction pattern of a representative batch of the crystalline complex of velagliflozin with theobromine (1:1), FORM 1009.
- Figure 10B shows the determination of the melting point via DSC and of the weight loss via TG of a representative batch of the crystalline complex of velagliflozin with theobromine (1:1), FORM 1009.
- the present invention relates to a crystalline complex between velagliflozin and a co-crystal forming agent selected from L-proline, theobromine, 3,5-dihydroxybenzoic acid, maltol, thymol, and caffeine.
- Preferred co-crystal forming agents are selected from L-proline and theobromine.
- the co-crystal forming agent is L-proline.
- the co-crystal forming agent is theobromine.
- the co-crystal forming agent is 3,5-dihydroxybenzoic acid.
- the co-crystal forming agent is maltol.
- the co-crystal forming agent is thymol.
- the co-crystal forming agent is caffeine.
- the molar ratio of velagliflozin and the co-crystal forming agent is in the range from about 1 : 1 to about 1 : 3.
- the crystalline complexes of velagliflozin and the co-crystal forming agent may be identified and distinguished from other crystalline forms by means of their characteristic X-ray powder diffraction (XRPD) patterns.
- XRPD characteristic X-ray powder diffraction
- the crystalline complex is a complex between velagliflozin and L-proline and it has a content of water of about 0 mol, i.e. it is a water-free / anhydrous co-crystal of velagliflozin with L-proline.
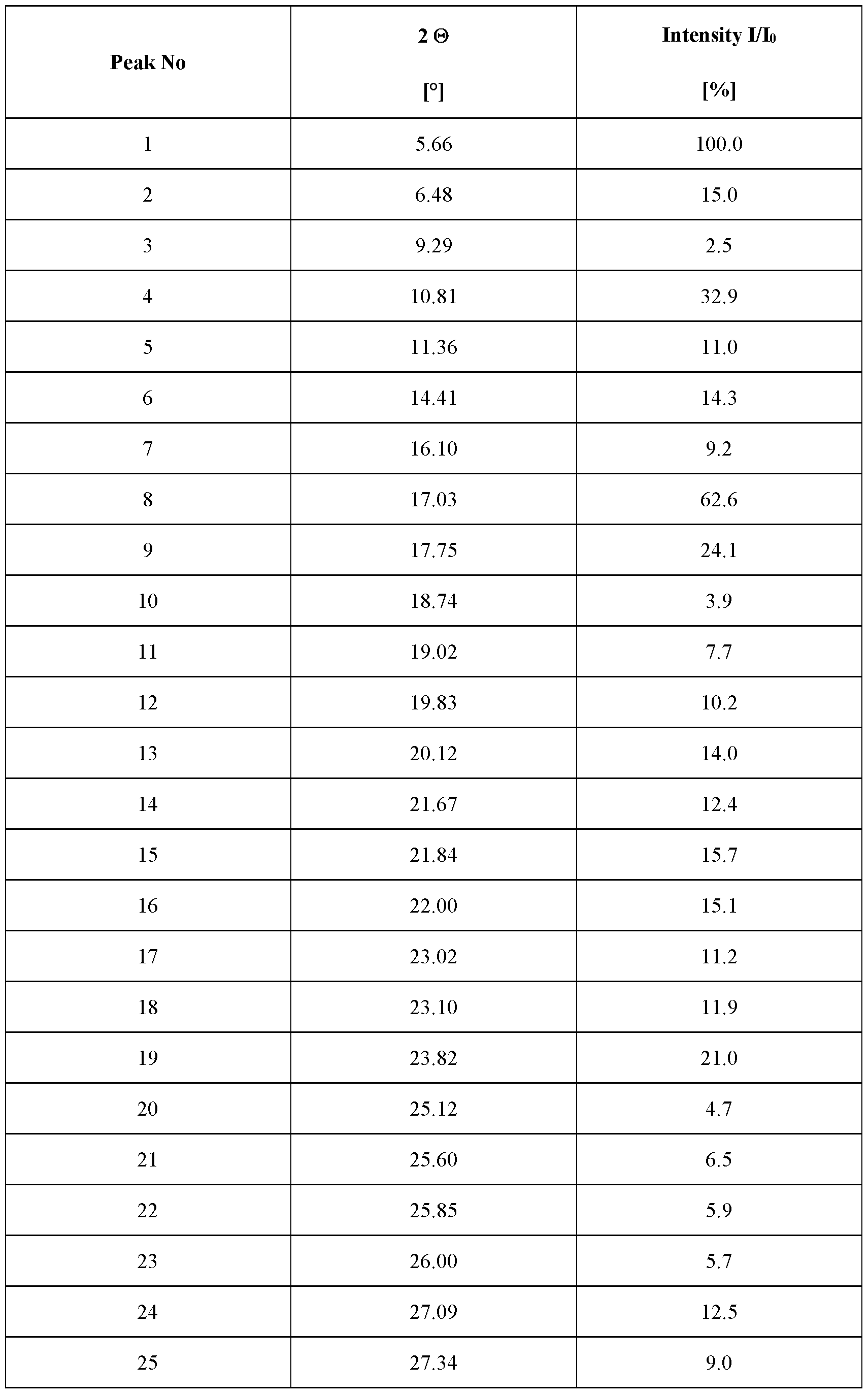
- the crystalline complex has a content of water of about 0 mol, characterized in that the crystalline complex is a complex (1:3) between velagliflozin and L-proline; and characterized by an X-ray powder diffraction pattern that comprises peaks at 5.77, 16.89, 18.95 and 20.39 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 17.40, 21.59, 23.69 and 25.19 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 5.10, 8.06, 9.64, 18.50, 19.36, 21.10, 23.23 and 26.06 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 11.58, 15.24, 17.88, 19.57, 22.13, 22.76, 24.64, 27.63 and 28.22 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- radiation comprises peaks at degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ) as contained in Table 1.
- Table 1 X-ray powder diffraction pattern of the crystalline complex of FORM 1000 (only peaks up to 30° in 2 ⁇ are listed): Even more specifically, said X-ray powder diffraction pattern, made using Cuk ⁇ 1
- said crystalline complex of velagliflozin with L-proline is characterized by a melting point of 195°C (determined via DSC; evaluated as onset-temperature; heating rate 10 K/min). The obtained DSC curve is shown in Figure IB.
- the crystalline complex is a complex of velagliflozin and L-proline with a molar ratio in the range from about 1 : 1 to about 1 : 1.3; and characterized by an X-ray powder diffraction pattern that comprises peaks at 4.00, 6.07, 7.30 and 20.36 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 18.29, 18.70, 19.90 and 22.09 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 14.59, 16.84, 17.24, 23.39, 24.49, 25.62, 27.10, and 27.45 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 16.15, 22.57, 24.28, 24.74, 29.45, 30.79, 34.97 and 36.18 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- radiation comprises peaks at degrees 2 ⁇ ( ⁇ 0. f degrees 2 ⁇ ) as contained in Table 2.
- Table 2 X-ray powder diffraction pattern of the crystalline complex of FORM 1001:
- said X-ray powder diffraction pattern made using Cuk ⁇ 1
- Said crystalline complex of velagliflozin with L-proline shows a weight loss by thermal gravimetry (TG). The observed weight loss indicates that the crystalline form contains water which may be bound by adsorption and/or may be part of the crystalline lattice, i.e the crystalline form may be present as a crystalline hydrate.
- said crystalline complex of velagliflozin with L-proline is characterized by a dehydration noted around 80°C (determined via DSC; evaluated as onset-temperature; heating rate 10 K/min). The obtained DSC curve is shown in Figure 2B.
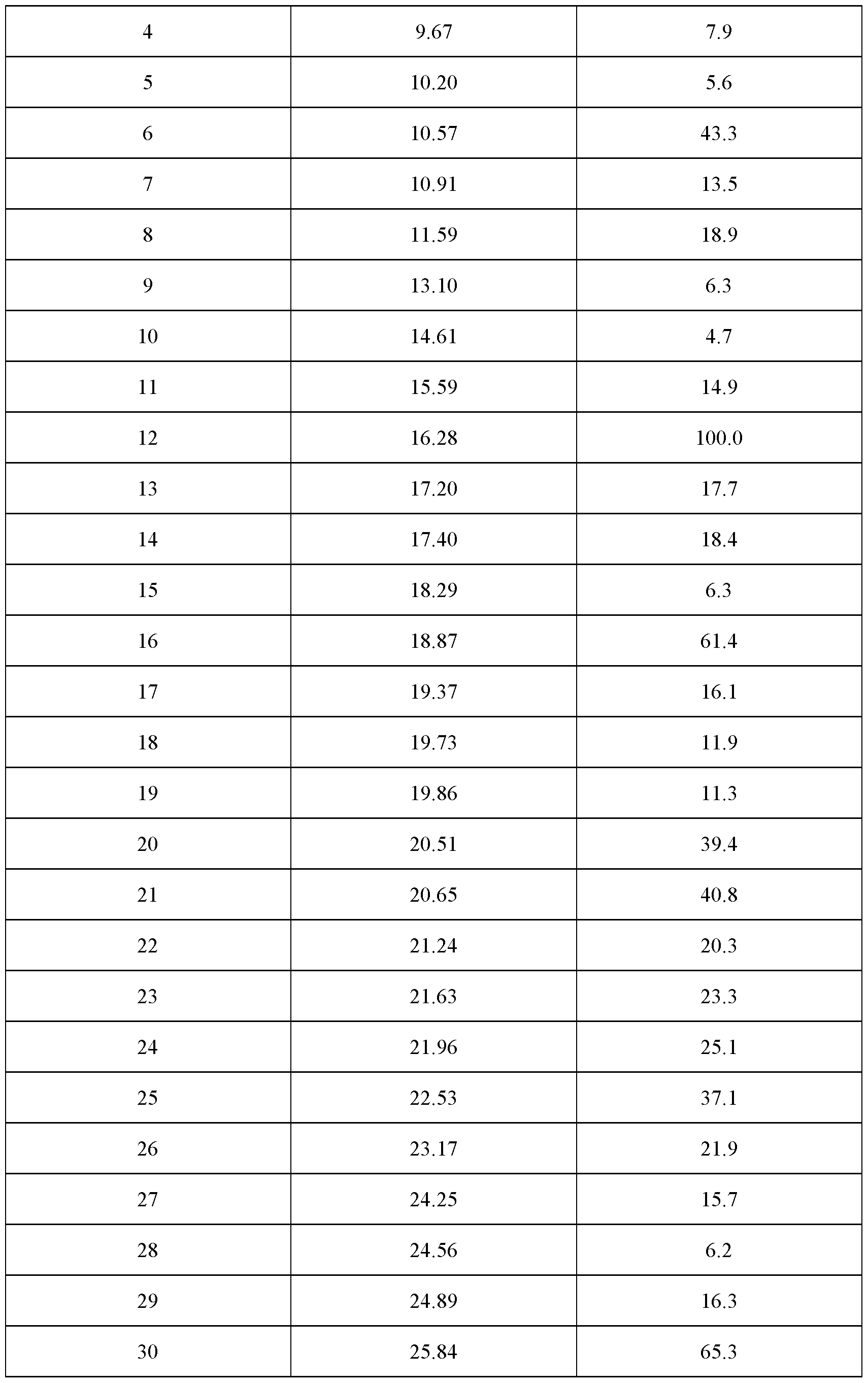
- the crystalline complex is a complex (1 : 1.5) between velagliflozin and L- proline; and characterized by an X-ray powder diffraction pattern that comprises peaks at 7.47, 16.28, 18.87 and 25.84 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 5.47, 10.57, 20.51 and 20.65 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 11.59, 17.20, 17.40, 21.24, 21.63, 21.96, 22.53 and 23.17 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 5.75, 10.91, 15.59, 19.37, 19.73, 19.86, 24.25 and 24.89 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- radiation comprises peaks at degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ) as contained in Table 3.
- Table 3 X-ray powder diffraction pattern of the crystalline complex of FORM 1002 (only peaks up to 30° in 2 ⁇ are listed): Even more specifically, said X-ray powder diffraction pattern, made using CuK ⁇ 1 radiation comprises peaks at degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ⁇ as shown in Figure 3A.
- Said crystalline complex of velagliflozin with L-proline shows a weight loss by thermal gravimetry (TG). The observed weight loss indicates that the crystalline form contains water which may be bound by adsorption and/or may be part of the crystalline lattice, i.e the crystalline form may be present as a crystalline hydrate.
- said crystalline complex of velagliflozin with L-proline is characterized by a dehydration noted around 50°C (determined via DSC; evaluated as onset-temperature; heating rate 10 K/min).
- the obtained DSC curve is shown in Figure 3B.
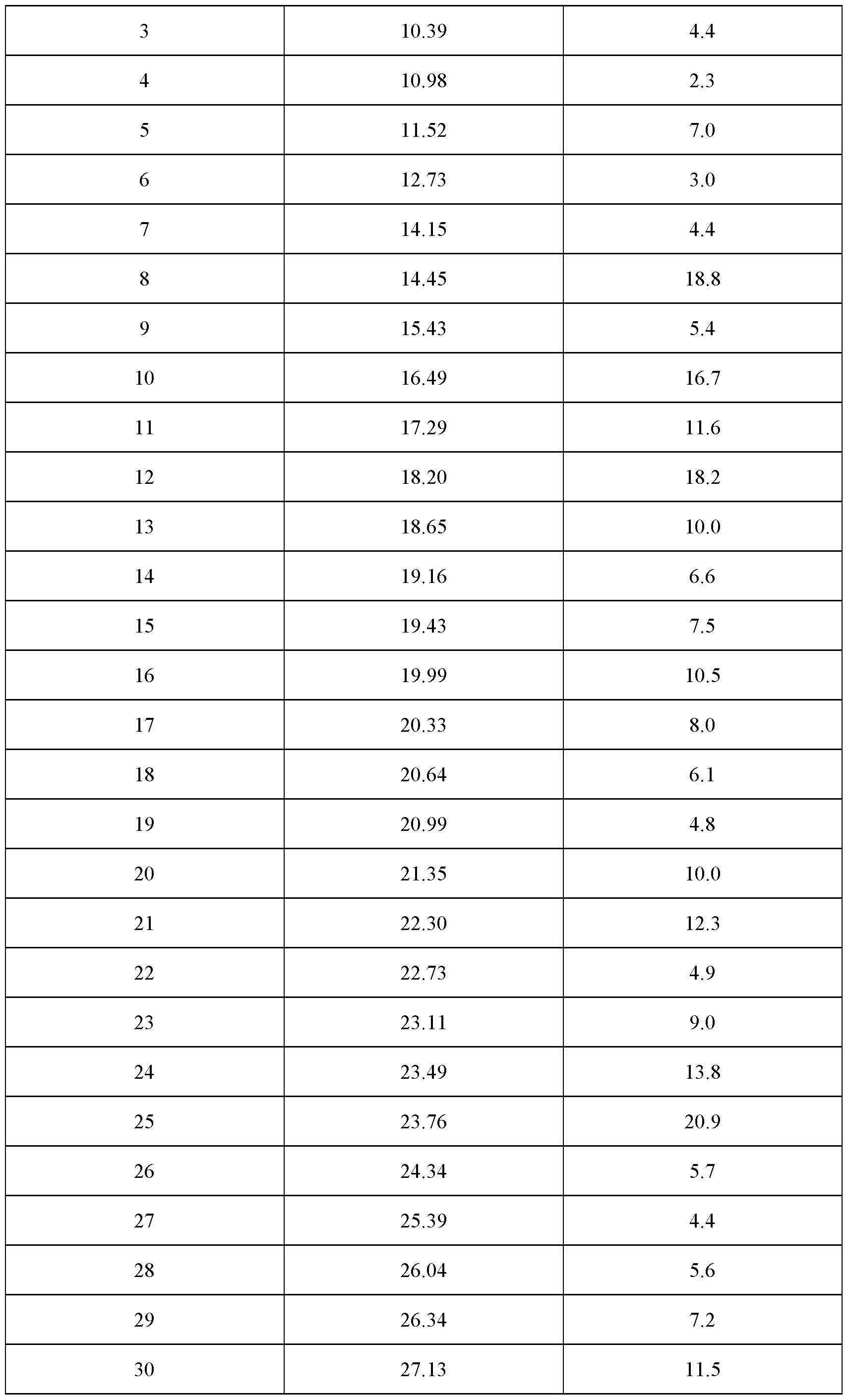
- the crystalline complex has a content of water of about 1.5 mol per mol of velagliflozin, characterized in that the crystalline complex is a complex (1 : 1) between velagliflozin and L- proline; and characterized by an X-ray powder diffraction pattern that comprises peaks at 5.49, 14.45, 18.20 and 23.76 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using uk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 16.49, 17.29, 22.30 and 23.49 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 11.52, 18.65, 19.43, 19.99, 20.33, 21.35, 23.11 and 26.34 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 6.36, 15.43, 19.16, 20.64, 20.99, 22.73, 24.34 and 26.04 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1 radiation.
- radiation comprises peaks at degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ) as contained in Table 4.
- Table 4 X-ray powder diffraction pattern of the crystalline complex of FORM 1003 (only peaks up to 30° in 2 ⁇ are listed): Even more specifically, said X-ray powder diffraction pattern, made using Cuk ⁇ 1 ] radiation comprises peaks at degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ) as shown in Figure 4A.
- Said crystalline complex of velagliflozin with L-proline shows a weight loss by thermal gravimetry (TG).
- the observed weight loss indicates that the crystalline form contains water which may be bound by adsorption and/or may be part of the crystalline lattice, i.e the crystalline form may be present as a crystalline hydrate.
- said crystalline complex of velagliflozin with L-proline is characterized by a dehydration noted around 50°C (determined via DSC; evaluated as onset-temperature; heating rate 10 K/min).
- the obtained DSC curve is shown in Figure 4B.
- the crystalline complex has a content of water of about 2 mol per mol of velagliflozin, characterized in that the crystalline complex is a complex (1 : 2) between velagliflozin and L- proline; and characterized by an X-ray powder diffraction pattern that comprises peaks at 5.94, 16.40, 17.95 and 20.28 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 18.49, 19.40, 19.72 and 23.17 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 16.12, 17.56, 20.59, 22.21, 22.41, 23.67, 26.59 and 27.38 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 8.77, 15.24, 19.09, 21.50, 22.82, 24.10, 24.83 and 25.26 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- radiation comprises peaks at degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ) as contained in Table 5.
- Table 5 X-ray powder diffraction pattern of the crystalline complex of FORM 1004 (only peaks up to 30° in 2 ⁇ are listed):
- radiation comprises peaks at degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ) as shown in Figure 5 A.
- Said crystalline complex of velagliflozin with L-proline shows a weight loss by thermal gravimetry (TG).
- the observed weight loss indicates that the crystalline form contains water which may be bound by adsorption and/or may be part of the crystalline lattice, i.e. the crystalline form may be present as a crystalline hydrate.
- said crystalline complex of velagliflozin with L-proline is characterized by a dehydration noted around 50°C (determined via DSC; evaluated as onset-temperature; heating rate 10 K/min). The obtained DSC curve is shown in Figure 5B.
- the crystalline complex has a content of water of about 0 mol, characterized in that the crystalline complex is a complex (1 : 1) between velagliflozin and L-proline; and characterized by an X-ray powder diffraction pattern that comprises peaks at 5.66, 10.81, 17.03 and 17.75 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 6.48, 21.84, 22.00 and 23.82 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cul ⁇ (/
- said X-ray powder diffraction pattern further comprises peaks at 11.36, 14.41, 20.12, 21.67, 23.02, 23.10, 27.09 and 28.03 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 16.10, 19.02, 19.83, 25.60, 25.85, 26.00, 27.34 and 28.47 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- radiation comprises peaks at degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ) as contained in Table 6.
- Table 6 X-ray powder diffraction pattern of the crystalline complex of FORM 1005 (only peaks up to 30° in 2 ⁇ are listed):
- radiation comprises peaks at degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ) as shown in Figure 6A.
- the crystalline complex has a content of water of about 0 mol, characterized in that the crystalline complex is a complex of velagliflozin and L-proline with a molar ratio in the range from about 1 : 1.4 to about 1 : 1.8; and characterized by an X-ray powder diffraction pattern that comprises peaks at 6.02, 16.64, 18.24 and 20.74 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 9.06, 15.49, 18.51 and 19.44 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cul ⁇ (/
- said X-ray powder diffraction pattern further comprises peaks at 17.79, 19.74, 22.70, 23.62, 24.06, 25.46, 27.03 and 27.88 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 12.06, 13.34, 20.21, 22.16, 23.04, 23.25, 24.48 and 27.31 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- radiation comprises peaks at degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ) as contained in Table 7.
- Table 7 X-ray powder diffraction pattern of the crystalline complex of FORM 1006 (only peaks up to 30° in 2 ⁇ are listed):
- radiation comprises peaks at degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ) as shown in Figure 7A.
- said crystalline complex of velagliflozin with L-proline is characterized by a melting point of 160°C (determined via DSC; evaluated as onset-temperature; heating rate 10 K/min). The obtained DSC curve is shown in Figure 7B.
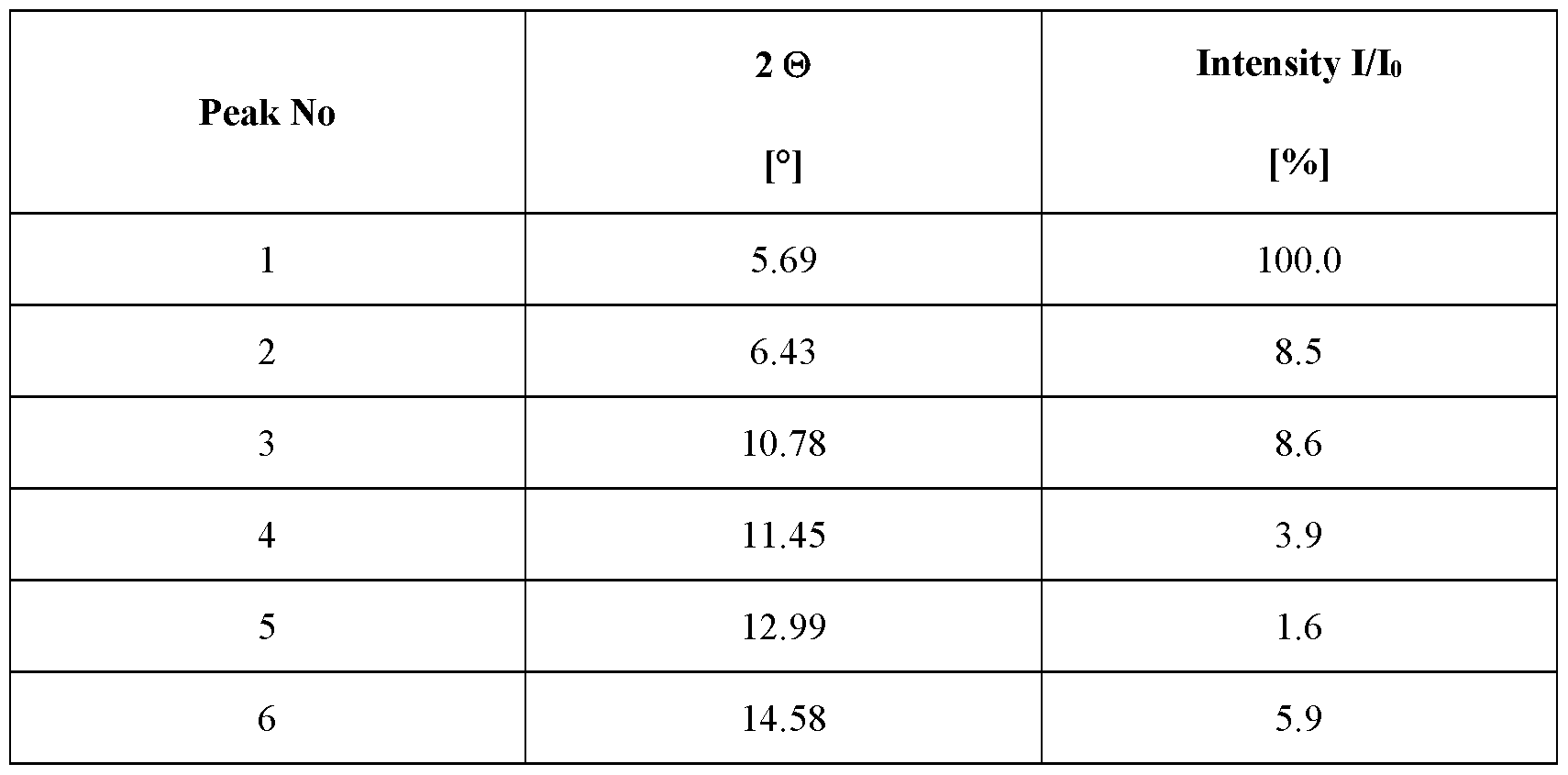
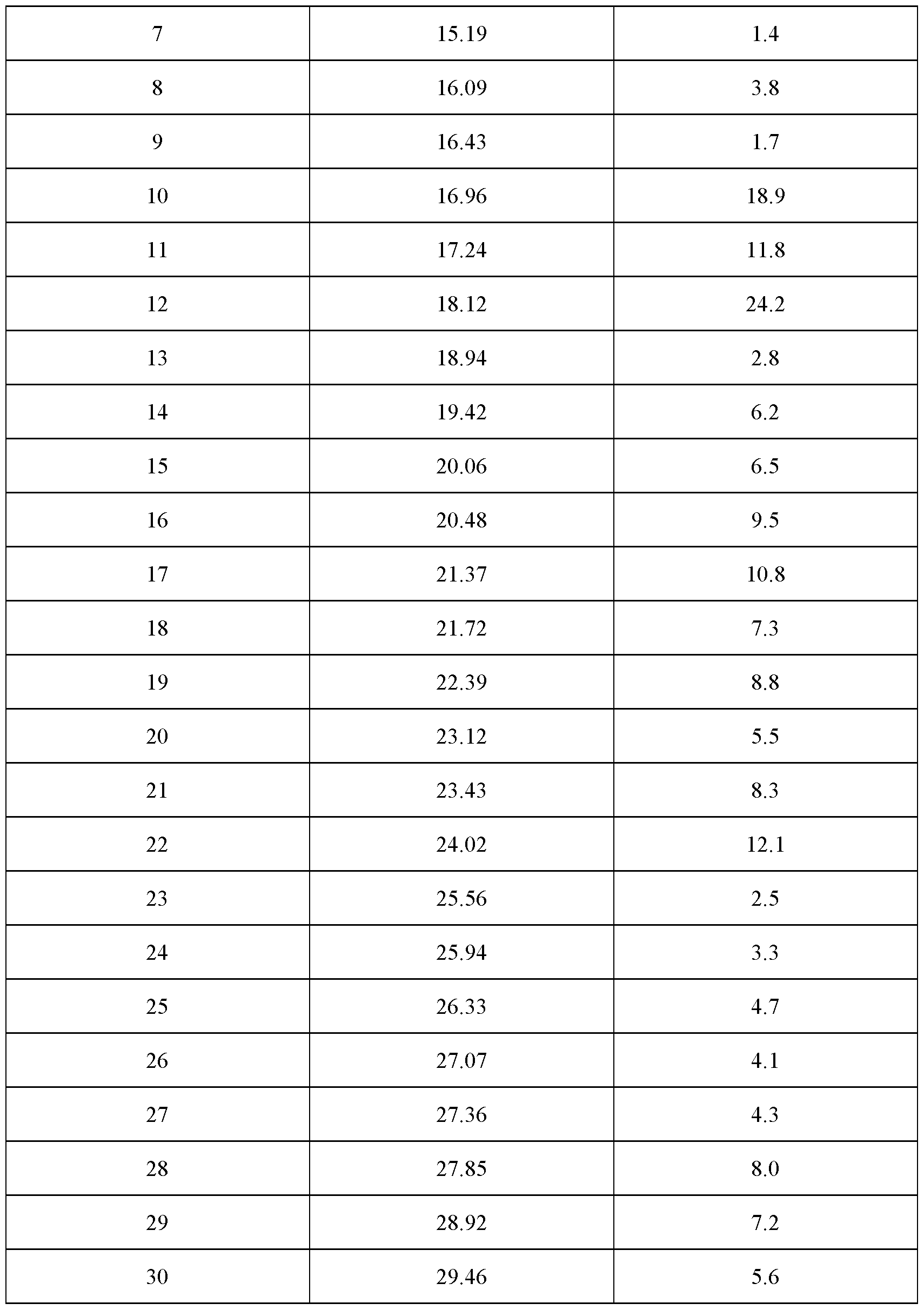
- the crystalline complex is in a hydrate form and is a complex (1 : 1) between velagliflozin and L-proline; and characterized by an X-ray powder diffraction pattern that comprises peaks at 8.69, 16.96, 18.12 and 24.02 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 10.78, 17.24, 20.48 and 21.39 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 6.43, 19.42, 20.06, 21.72, 22.39, 23.43, 27.85 and 28.92 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 11.45, 14.58, 16.09, 23.12, 26.33, 27.07, 27.36 and 29.46 degrees 2 > ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- radiation comprises peaks at degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ) as contained in Table 8.
- Table 8 X-ray powder diffraction pattern of the crystalline complex of FORM 1007 (only peaks up to 30° in 2 ⁇ are listed):
- radiation comprises peaks at degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ) as shown in Figure 8A.
- said crystalline complex of velagliflozin with L-proline is characterized by a dehydration noted around 60°C (determined via DSC; evaluated as onset-temperature; heating rate 10 K/min). The obtained DSC curve is shown in Figure 8B.
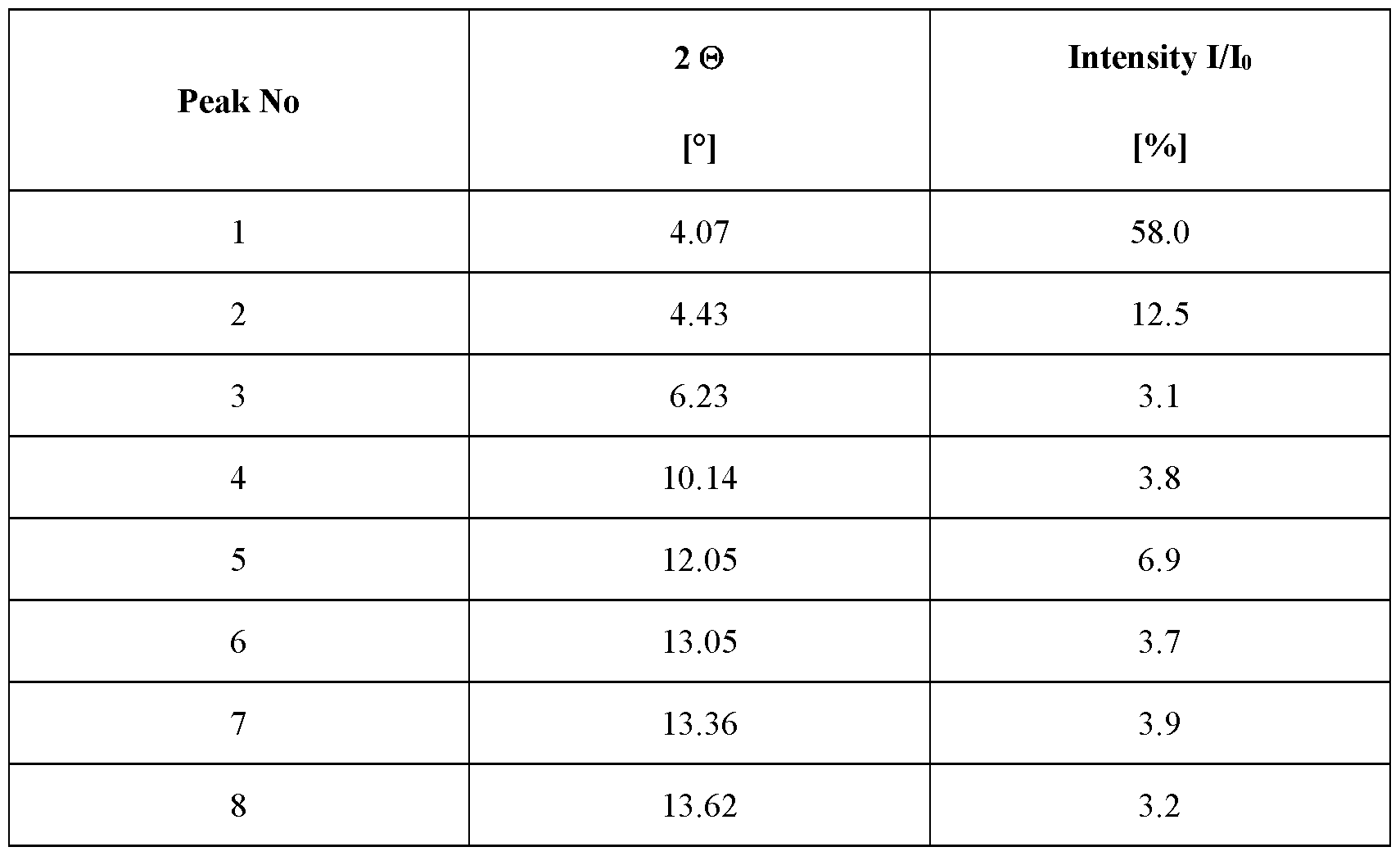
- the crystalline complex has a content of tetrahydrofuran of about 0.4 mol per mol of velagliflozin, characterized in that the crystalline complex is a complex (1 : 1) between velagliflozin and L-proline; and characterized by an X-ray powder diffraction pattern that comprises peaks at 4.07, 17.21, 20.30 and 23.50 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 16.09, 18.49, 21.42 and 25.71 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cul ⁇ (/
- said X-ray powder diffraction pattern further comprises peaks at 14.74, 18.87, 21.05, 22.00, 23.71, 24.68, 24.86, and 25.24 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 4.43, 12.05, 15.30, 16.67, 19.46, 22.63, 26.29 and 26.42 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said crystalline complex is characterized by an X-ray powder diffraction pattern, made using Cuk ⁇ 1
- Table 9 X-ray powder diffraction pattern of the crystalline complex of FORM 1008 (only peaks up to 30° in 2 ⁇ are listed):
- radiation comprises peaks at degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ) as shown in Figure 9A.
- said crystalline complex of velagliflozin with L-proline is characterized by a complex DSC profile with a dehydration / desolvation noted around 60°C (determined via DSC; evaluated as onset-temperature; heating rate 10 K/min). The obtained DSC curve is shown in Figure 9B.
- the crystalline complex is a complex between velagliflozin and theobromine and it has a content of water of about 0 mol, i.e. it is a water-free / anhydrous co-crystal of velagliflozin with theobromine.
- the crystalline complex is a complex (1 : 1) between velagliflozin and theobromine; and characterized by an X-ray powder diffraction pattern that comprises peaks at 18.22, 20.15, 22.56 and 25.36 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 14.19, 27.01, 34.11 and 38.63 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cul ⁇ (/
- said X-ray powder diffraction pattern further comprises peaks at 30.65, 30.95, 32.27, and 36.89 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- said X-ray powder diffraction pattern further comprises peaks at 15.23, 32.08, 36.06, 37.54, and 39.82 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cul ⁇ (/
- said crystalline complex is characterized by an X-ray powder diffraction pattern, made using Cuk ⁇ 1
- Table 10 X-ray powder diffraction pattern of the crystalline complex of FORM 1009:
- radiation comprises peaks at degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ) as shown in Figure 10A.
- said crystalline complex of velagliflozin with theobromine is characterized by a melting point of 120°C (determined via DSC; evaluated as onset-temperature; heating rate 10 K/min). The obtained DSC curve is shown in Figure 10B.
- the crystalline complexes according to the present invention have advantageous physicochemical properties which are beneficial in the preparation of a pharmaceutical composition.
- such crystalline complexes have a high physical and chemical stability under various environmental conditions and during the production of a medicament.
- the crystals can be obtained in a shape and particle size which are particularly suitable in a production method for solid pharmaceutical formulations.
- the crystals show a high mechanical stability that allows grinding of the crystals.
- the crystalline complexes do not show a high tendency to absorb moisture and are chemically stable, i.e., the crystalline complexes allow the production of a solid pharmaceutical formulation with a long shelf life.
- the crystalline complexes have a favorably high solubility over a wide pH-range which is advantageous in solid pharmaceutical formulations for oral administration.
- OED location-sensitive detector
- Cu-anode Cu-anode
- the above described 2 ⁇ values should be considered accurate to ⁇ 0.1 degrees 2 ⁇ , in particular ⁇ 0.05 degrees 2 ⁇ . That is to say, when assessing whether a given sample of crystals of velagliflozin is the crystalline form in accordance with the invention, a 2 ⁇ value which is experimentally observed for the sample should be considered identical with a characteristic value described above if it falls within ⁇ 0.1 degrees 2 ⁇ of the characteristic value, in particular if it falls within ⁇ 0.05 degrees 2 ⁇ of the characteristic value.
- the melting point is determined by DSC (Differential Scanning Calorimetry) using a DSC Q200 (TA- Instmment/Waters).
- the weight loss is determined by thermal gravimetry (TG) using a TGA Q500 (TA- Instruments/W aters) .
- a further aspect of the present invention relates to a method for making the one or more crystalline complexes of the present invention as defined hereinbefore and hereinafter, said method comprising the following steps:
- step (a) a solution of velagliflozin and the co-crystal forming agent in a solvent or a mixture of solvents is prepared.
- the solution is saturated or at least nearly saturated or even supersaturated with respect to the respective crystalline complex.
- velagliflozin may be dissolved in a solution comprising the co-crystal forming agent; or the co-crystal forming agent may be dissolved in a solution comprising velagliflozin.
- velagliflozin is dissolved in a solvent or mixture of solvents to yield a first solution; and the co-crystal forming agent is dissolved in a solvent or mixture of solvents to yield a second solution. Thereafter, said first solution and said second solution are combined to form the solution according to step (a).
- the molar ratio of the co-crystal forming agent and velagliflozin in the solution corresponds to the molar ratio of the co-crystal forming agent and velagliflozin in the respective crystalline complex to be obtained. Therefore, a preferred molar ratio is in the range from about 1 : 1 to about 1 : 3.
- Suitable solvents are preferably selected from the group consisting of Ci-4-alkanols, water, ethylacetate, acetonitrile, acetone, diethylether, tetrahydrofuran, and mixture of two or more of these solvents.
- More preferred solvents are selected from the group consisting of methanol, ethanol, isopropanol, water and mixture of two or more of these solvents, in particular mixtures of one or more of said organic solvents with water.
- Particularly preferred solvents are selected from the group consisting of ethanol, isopropanol, water and mixtures of ethanol and/or isopropanol with water.
- a preferred volume ratio of water : Ci-4-alkanol is in the range from about 99 : 1 to about 1 : 99; more preferably from about 50 : 1 to about 1 : 80; even more preferably from about 10 : 1 to about 1 : 60.
- the step (a) is carried out at about room temperature (about 20°C) or at an elevated temperature up to about the boiling point of the solvent or mixture of solvents used.
- the starting material of velagliflozin and/or of the co-crystal forming agent and/or of the solvent and mixtures of solvents contain an amount of H 2 O, which is at least the quantity required to form a hydrate of velagliflozin; in particular at least 1 mol, preferably at least 1.5 mol of water per mol of velagliflozin. Even more preferably the amount of water is at least 2 mol of water per mol of velagliflozin.
- velagliflozin as starting material or the co-crystal forming agent or said solvent or mixture of solvents, or said compounds and/or solvents in combination contain an amount of H 2 O as specified above.
- a water content of the solvent(s) is not mandatory.
- step (a) and/or in step (b) one or more antisolvents may be added, preferably during step (a) or at the beginning of step (b).
- Water is an example of a suitable antisolvent.
- the amount of antisolvent is preferably chosen to obtain a supersaturated or saturated solution with respect to the respective crystalline complex.
- step (b) the solution is stored for a time sufficient to obtain a precipitate, i.e. the respective crystalline complex.
- the temperature of the solution in step (b) is about the same as or lower than in step (a).
- the temperature of the solution is preferably lowered, preferably to a temperature in the range of 20°C to 0°C or even lower.
- Step (b) can be carried out with or without stirring.
- the crystallization may be induced by methods as known in the art, for example by mechanical means such as scratching or rubbing the contact surface of the reaction vessel for example with a glass rod.
- the (nearly) saturated or supersaturated solution may be inoculated with seed crystals.
- step (c) the solvent(s) can be removed from the precipitate by known methods as for example filtration, suction filtration, decantation or centrifugation.
- step (d) an excess of the solvent(s) is removed from the precipitate by methods known to the one skilled in the art as for example by reducing the partial pressure of the solvent(s), preferably in vacuum, and/or by heating above ca. 20°C, preferably in a temperature range below 100°C, even more preferably below 85°C.
- Velagliflozin may be synthesized by methods as specifically and/or generally described or cited in the international applications WO 2007/093610 and WO 2007/128749. Furthermore, the biological properties of velagliflozin may be investigated as it is described in the international applications WO 2007/093610 and WO 2007/128749.
- the one or more crystalline complexes in accordance with the invention are preferably employed as drug active substance in substantially pure form, that is to say, essentially free of other crystalline forms of velagliflozin. Nevertheless, the invention also embraces the crystalline complex in accordance with the invention in admixture with another crystalline form or forms of velagliflozin. Should the drug active substance be a mixture of crystalline forms, it is preferred that the substance comprises at least 50%-weight, even more preferably at least 90%-weight, most preferably at least 95%-weight of the crystalline complex as described herein.
- the one or more crystalline complexes as defined hereinbefore or hereinafter and/or the pharmaceutical composition as defined hereinbefore or hereinafter and/or the medicament as defined hereinbefore or hereinafter are suitable for the treatment and/or preventive treatment of conditions or diseases which may be affected by the inhibition of the SGLT activity, particularly the SGLT-2 activity.
- the one or more crystalline complexes as defined hereinbefore or hereinafter are also suitable for the preparation of pharmaceutical compositions for the treatment and/or preventive treatment of conditions or diseases which may be affected by the inhibition of the SGLT activity, particularly the SGLT-2 activity. Therefore, the one or more crystalline complexes according to the invention as defined hereinbefore or hereinafter are suitable for the treatment of diabetes, in particular feline diabetes and/or canine diabetes.
- the dosage required to achieve the corresponding activity for treatment and/or prevention usually depends on the patient, the nature and gravity of the illness or condition and the method and frequency of administration and is for the patient's doctor/veterinarian to decide.
- the dosage may be from 1 to 100 mg by intravenous route, and 1 to 1000 mg by oral route, in each case administered 1 to 4 times a day.
- the pharmaceutical compositions according to this invention preferably comprise the one or more crystalline complexes according to the invention together with one or more inert conventional carriers and/or diluents.
- Such pharmaceutical compositions may be formulated as conventional galenic preparations, such as plain or coated tablets, capsules, powders, suspensions or suppositories.
- H atoms of hydroxyl groups are not explicitly shown in every case in the structural formulae.
- the following example of synthesis serves to illustrate a method of preparing velagliflozin and its crystalline complex with different co-crystal forming agents. It is to be regarded only as a possible method described by way of example, without restricting the invention to its contents.
- the terms "room temperature” and “ambient temperature” are used interchangeably and denote temperatures of about 20°C.
- Velagliflozin is prepared as disclosed in W02014/016381 on pages 13 - 18.
- a crystalline complex (1 : 1) of velagliflozin with L-proline is prepared as disclosed in W02014/016381 on page 18.
- the resulting crystalline complex is a monohydrate co-crystal and characterized by a molar ratio of velagliflozin and L-proline and crystal water of 1 : 1 : 1.
- Step 1 10 mg of crystalline complex (1 : 1) of velagliflozin with L-proline is dissolved in 500 ⁇ l of methanol. The resulting solution is incubated at 40°C (evaporation under N 2 gas flow) to obtain a glassy material.
- Step 2 This material is mixed with 1 ml of ethyl acetate and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm filter and dried under vacuum at RT for Ih.
- the X-ray powder diffraction pattern as contained in Table 1 and as depicted in Figure 1 A and the DSC and TG diagram in Figure IB correspond to a batch with a melting point of approximately 195°C.
- Step 1 10 mg of crystalline complex (1 : 1) of velagliflozin with L-proline is dissolved in 500 ⁇ l of methanol. The resulting solution is incubated at 40°C (evaporation under N 2 gas flow) to obtain a glassy material.
- Step 2 This material is mixed with 1 ml of acetone and the resulting suspension is solubilized for 3h at RT. After slow evaporation at RT a white solid was obtained.
- Step 2 (alternative 2): Alternatively, the glassy material from step 1 is mixed with a mixture of acetone and ethanol (ratio 10: 110) and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm filter and dried under vacuum at RT for Ih.
- Step 2 (alternative 3): The glassy material from step 1 is mixed with 100 ⁇ l of methyl-THF and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm filter and dried under vacuum at RT for Ih.
- Complementary NMR analysis confirms the chemical integrity of the velagliflozin material, the presence of 1 mole eq. of proline and the presence of 0.6 mol acetone of residual solvent or in the absence of residual solvent.
- the X-ray powder diffraction pattern as contained in Table 2 and as depicted in Figure 2A and the DSC and TG diagram in Figure 2B correspond to a batch with a dehydration noted at around 80°C.
- Step 1 30 mg of crystalline complex (1 : 1) of velagliflozin with L-proline is dissolved in 500 ⁇ l of methanol. The resulting solution is incubated at 40°C (evaporation under N 2 gas flow) to obtain a glassy material.
- Step 2 This material is mixed with 1 ml of dimethylcarbonate and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm filter and dried under vacuum at RT for Ih.
- Step 2 (alternative 2): Alternatively, the glassy material from step 1 is mixed with a mixture of water and 2- propanol (ratio 12: 110) and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm filter and dried under vacuum at RT for Ih.
- Step 2 (alternative 3): The glassy material from step 1 is mixed 1 ml of acetonitrile and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm filter and dried under vacuum at RT for Ih.
- Step 1 (alternative 1): 40mg of crystalline complex (1 : 1) of velagliflozin with L-proline is suspended in 200 pl of Acetonitrile. The suspension is stirred 30 days at RT and then filtered using a 0.2 pm filter.
- Step 1 (alternative 2): 20mg of crystalline complex (1 : 1) of velagliflozin with L-proline is suspended in 100 pl of a mixture of water and dioxane (ratio 0.03:97 volume). The suspension is stirred 30 days at RT and then filtered using a 0.2 pm filter.
- Complementary NMR analysis confirms the chemical integrity of the velagliflozin material, the presence of 1.5 mole eq. of proline and the presence of trace of residual dioxane ( ⁇ 0.1 mol) or in the absence of residual solvent.
- Several batches of the crystalline complex according to the above preparation are obtained.
- the X-ray powder diffraction patterns coincide.
- the melting points are determined via DSC and evaluated as onset-temperature.
- DSC thermograms show a weak and broad endothermic event between RT and 90°C associated with the dehydration of crystalline material.
- An endothermic peak at ⁇ 150°C is associated with the melting transition of crystalline material obtained after dehydration. From these results it could be concluded that the crystalline complex was present in a hydrated form.
- the X-ray powder diffraction pattern as contained in Table 3 and as depicted in Figure 3 A and the DSC and TG diagram in Figure 3B correspond to a batch with a dehydration noted at around 50°C.
- Step 1 10 mg of crystalline complex (1 : 1) of velagliflozin with L-proline is dissolved in 500 ⁇ l of methanol. The resulting solution is incubated at 40°C (evaporation under N 2 gas flow) to obtain a glassy material.
- Step 2 This material is mixed with 100 ⁇ l of ethanol and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm filter and dried under vacuum at RT for Ih.
- Step 2 (alternative 2): Alternatively, the glassy material from step 1 is mixed with 1 ml of xylene and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm fdter and dried under vacuum at RT for Ih.
- Step 2 (alternative 3): The glassy material from step 1 is mixed with 300 ⁇ l of methanol and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm filter and dried under vacuum at RT for Ih.
- Complementary NMR analysis confirms the chemical integrity of the velagliflozin material, the presence of 1.0 mole eq. of proline and the presence of trace of residual dioxane ( ⁇ 0.1 mol) or in the absence of residual solvent.
- Several batches of the crystalline complex according to the above preparation are obtained.
- the X-ray powder diffraction patterns coincide.
- the melting points are determined via DSC and evaluated as onset-temperature.
- DSC thermograms show a weak and broad endothermic event between RT and 90°C associated with the dehydration of crystalline material. From these results it could be concluded that the crystalline complex was present in a hydrated form.
- the X-ray powder diffraction pattern as contained in Table 4 and as depicted in Figure 4A and the DSC and TG diagram in Figure 4B correspond to a batch with a dehydration noted at around 50°C.
- Step 1 10 mg of crystalline complex (1 : 1) of velagliflozin with L-proline is dissolved in 500 ⁇ l of methanol. The resulting solution is incubated at 40°C (evaporation under N2 gas flow) to obtain a glassy material.
- Step 2 This material is mixed with 1 ml of anisole and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm filter and dried under vacuum at RT for Ih.
- Step 2 (alternative 2): Alternatively, the glassy material from step 1 is mixed with 1 ml of xylene and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm filter and dried under vacuum at RT for Ih.
- the obtained solid is stored 7 days under room conditions.
- the X-ray powder diffraction pattern as contained in Table 5 and as depicted in Figure 5A and the DSC and TG diagram in Figure 5B correspond to a batch with a dehydration noted at around 50°C.
- Step 1 Step 1: 10 mg of crystalline complex (1 : 1) of velagliflozin with L-proline is dissolved in 500 of ⁇ l methanol. The resulting solution is incubated at 40°C (evaporation under N 2 gas flow) to obtain a glassy material.
- Step 2 This material is mixed with 400 of W ⁇ alter/Ethanol 2/20 (volume ratio) binary solvent and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm filter and dried under vacuum at RT for Ih.
- Step 3 Solid material is heated to 95°C.
- the X-ray powder diffraction patterns recorded at 95°C is associated with the Form 1005.
- Form 1005 being obtained from dehydration at 95°C of Form 1003, we conclude that the crystalline complex was present in an anhydrous form.
- Step 1 10 mg of crystalline complex (1 : 1) of velagliflozin with L-proline is dissolved in 500 of methanol. ⁇ l The resulting solution is incubated at 40°C (evaporation under N 2 gas flow) to obtain a glassy material.
- Step 2 This material is mixed with 1 ml of anisole and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm filter and dried under vacuum at RT for Ih.
- Step 2 (alternative 2): Alternatively, the glassy material from step 1 is mixed with 1 ml of xylene and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm fdter and dried under vacuum at RT for Ih.
- Step 2 (alternative 3): The glassy material from step 1 is mixed with 500 of cyclopro ⁇ plylmethylether and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm filter and dried under vacuum at RT for Ih.
- Figure 7A and the DSC and TG diagram in Figure 7B correspond to a batch with a melting point of approximately 160°C.
- Step 1 10 mg of crystalline complex (1 : 1) of velagliflozin with L-proline is dissolved in 500 of methanol. ⁇ l The resulting solution is incubated at 40°C (evaporation under N 2 gas flow) to obtain a glassy material.
- Step 2 This material is mixed with 100 of et ⁇ hlanol and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm filter and dried under vacuum at RT for Ih.
- Step 2 (alternative 2): Alternatively, the glassy material from step 1 is mixed with 1 ml of xylene and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm filter and dried under vacuum at RT for Ih.
- Step 2 (alternative 3): The glassy material from step 1 is mixed with 300 of methano ⁇ ll and the resulting suspension is solubilized for Ih at 60°C. The suspension is cooled by 0.01°C /min down to 5°C. The cooled suspension is filtered using a 0.2 pm filter and dried under vacuum at RT for Ih.
- Obtained solid is stored at least 30 days under room conditions.
- the X-ray powder diffraction pattern as contained in Table 8 and as depicted in Figure 8A and the DSC and TG diagram in Figure 8B correspond to a batch with a dehydration noted at around 60°C.
- Step 1 in 2 ml hermetic glass vial, 33mg of crystalline complex (1 : 1) of velagliflozin with L-proline is suspended in lOOpl of Water/Tetrahydrofuran 0.02/0.98 (volume ratio) binary mixture.
- Step 1 (alternative 2): in 2 ml hermetic glass vial, 56mg of crystalline complex (1 : 1) of velagliflozin with L- proline is suspended in lOOpl of Water/Tetrahydrofuran 0.15/0.85 (volume ratio) binary mixture.
- Step 2 After 30 days of storage, the suspension is mixed by vortex stirring. The cooled suspension is filtered using a 0.2 pm filter.
- Complementary NMR analysis confirms the chemical integrity of the velagliflozin material, the presence of 1 mole eq. of proline and the presence of 0.4 mole eq. of THF.
- Stepl In a 6 ml stainless steel milling tube, 2 stainless steel milling balls of 5 mm diameter are placed.
- Step2 11.3 mg of Theobromine and 25.3mg of crystalline complex (1 : 1) of velagliflozin with L-proline are added into the tube.
- Step 3 9 ⁇ l of Methanol are added into the tube, immediately after addition of solvent, the tube is hermetically closed.
- Step 4 Immediately after preparation, the tube is placed on the milling instrument (Retsch MM200) for 45 min milling at 20 Hz.
- the X-ray powder diffraction pattern as contained in Table 10 and as depicted in Figure 10A and the DSC and TG diagram in Figure 10B correspond to a batch with a melting point of 120°C.
- Crystalline complex between velagliflozin and a co-cry stal forming agent selected from L-proline, theobromine, 3,5-dihydroxybenzoic acid, maltol, thymol, and caffeine characterized in that the crystalline complex is a complex of velagliflozin and the co-crystal forming agent with a molar ratio in the range from about 1 : 1 to about 1 : 3.
- Crystalline complex according to any one of clauses 1 to 3, characterized in that the crystalline complex is a complex between velagliflozin and L-proline and in that it has a content of water of about 0 mol, i.e. it is a water- free / anhydrous co-crystal of velagliflozin with L-proline.
- Crystalline complex according to any one of clauses 1 to 3 and 9 having a content of water of about 0 mol, characterized in that the crystalline complex is a complex (1:3) between velagliflozin and L-proline; and characterized by an X-ray powder diffraction pattern that comprises peaks at 5.77, 16.89, 18.95 and 20.39 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- Crystalline complex according to clause 10 wherein the X-ray powder diffraction pattern further comprises peaks at 17.40, 21.59, 23.69 and 25.19 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 10-13 wherein the X-ray powder diffraction pattern, made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 10-14 characterized by an X-ray powder diffraction pattern, made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 1 to 3 in a hydrate form, characterized in that the crystalline complex is a complex of velagliflozin and L-proline with a molar ratio in the range from about 1 : 1 to about 1 : 1.3; and characterized by an X-ray powder diffraction pattern that comprises peaks at 4.00, 6.07, 7.30 and 20.36 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- Crystalline complex according to clause 16 wherein the X-ray powder diffraction pattern further comprises peaks at 18.29, 18.70, 19.90 and 22.09 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 1 to 3 in a hydrate form, characterized in that the crystalline complex is a complex (1 : 1.5) between velagliflozin and L-proline; and characterized by an X-ray powder diffraction pattern that comprises peaks at 7.47, 16.28, 18.87 and 25.84 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 22-25, characterized by an X-ray powder diffraction pattern, made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 28-31 characterized by an X-ray powder diffraction pattern, made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 28-32 characterized by an X-ray powder diffraction pattern, made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 34-37, characterized by an X-ray powder diffraction pattern, made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 34-38, characterized by an X-ray powder diffraction pattern, made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 1 to 3 and 9 having a content of water of about 0 mol, characterized in that the crystalline complex is a complex (1 : 1) between velagliflozin and L-proline; and characterized by an X-ray powder diffraction pattern that comprises peaks at 5.66, 10.81, 17.03 and 17.75 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- Crystalline complex according to clause 40 wherein the X-ray powder diffraction pattern further comprises peaks at 6.48, 21.84, 22.00 and 23.82 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 40-43, characterized by an X-ray powder diffraction pattern, made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 40-44 characterized by an X-ray powder diffraction pattern, made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 1 to 3 and 9 having a content of water of about 0 mol, characterized in that the crystalline complex is a complex of velagliflozin and L-proline with a molar ratio in the range from about 1 : 1.4 to about 1 : 1.8; and characterized by an X-ray powder diffraction pattern that comprises peaks at 6.02, 16.64, 18.24 and 20.74 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 46-50, characterized by an X-ray powder diffraction pattern, made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 1 to 3 in a hydrate form, characterized in that the crystalline complex is a complex (1 : 1) between velagliflozin and L-proline; and characterized by an X-ray powder diffraction pattern that comprises peaks at 8.69, 16.96, 18.12 and 24.02 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 52-55, characterized by an X-ray powder diffraction pattern, made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 1 to 3 characterized by a content of tetrahydrofuran of about 0.4 mol per mol of velagliflozin, characterized in that the crystalline complex is a complex (1 : 1) between velagliflozin and L-proline; and characterized by an X-ray powder diffraction pattern that comprises peaks at 4.07, 17.21, 20.30 and 23.50 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- Crystalline complex according to clause 58 wherein the X-ray powder diffraction pattern further comprises peaks at 16.09, 18.49, 21.42 and 25.71 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 58-61, the crystalline complex is characterized by an X- ray powder diffraction pattern, made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 1, 2 and 4, characterized in that the crystalline complex is a complex between velagliflozin and theobromine and in that it has a content of water of about 0 mol, i.e. it is a water-free / anhydrous co-crystal of velagliflozin with theobromine.
- Crystalline complex according to any one of clauses 1, 2, 4 and 64, characterized in that the crystalline complex is a complex (1 : 1) between velagliflozin and theobromine; and characterized by an X-ray powder diffraction pattern that comprises peaks at 18.22, 20.15, 22.56 and 25.36 degrees 2 ⁇ ( ⁇ 0.1 degrees 2 ⁇ ), wherein said X-ray powder diffraction pattern is made using Cuk ⁇ 1
- Crystalline complex according to any one of clauses 65-68 the crystalline complex is characterized by an X- ray powder diffraction pattern, made using Cuk ⁇ 1
- a mixture of crystalline forms comprising the crystalline complex according to any one of clauses 1 to 70 in admixture with another crystalline form or forms of velagliflozin.
- a pharmaceutical composition comprising one or more crystalline complexes in accordance with any one of clauses 1 to 70 and/or the mixture of crystalline forms according to any one of clauses 71 to 72.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Diabetes (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Hematology (AREA)
- Endocrinology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Obesity (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Emergency Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Steroid Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Epidemiology (AREA)
- Pyrrole Compounds (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202480026335.2A CN121039113A (en) | 2023-04-24 | 2024-04-18 | Crystallization complex of vildaggliflozin with selected cocrystallization agent, its preparation method and its use in drug preparation |
| AU2024264357A AU2024264357A1 (en) | 2023-04-24 | 2024-04-18 | Crystalline complexes between velagliflozin and selected co-crystal forming agents, methods for their preparation and the use thereof for preparing medicaments |
| KR1020257039431A KR20260004451A (en) | 2023-04-24 | 2024-04-18 | Crystalline complex of belagliplozine and selected co-crystal formers, process for preparing same and use thereof in the preparation of medicaments |
| MX2025012705A MX2025012705A (en) | 2023-04-24 | 2025-10-23 | Crystalline complexes between velagliflozin and selected co-crystal forming agents, methods for their preparation and the use thereof for preparing medicaments |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23169467.0 | 2023-04-24 | ||
| EP23169467 | 2023-04-24 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024223388A1 true WO2024223388A1 (en) | 2024-10-31 |
Family
ID=86185186
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/060490 Pending WO2024223388A1 (en) | 2023-04-24 | 2024-04-18 | Crystalline complexes between velagliflozin and selected co-crystal forming agents, methods for their preparation and the use thereof for preparing medicaments |
Country Status (5)
| Country | Link |
|---|---|
| KR (1) | KR20260004451A (en) |
| CN (1) | CN121039113A (en) |
| AU (1) | AU2024264357A1 (en) |
| MX (1) | MX2025012705A (en) |
| WO (1) | WO2024223388A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12397009B2 (en) | 2019-11-28 | 2025-08-26 | Boehringer Ingelheim Vetmedica Gmbh | Use of SGLT-2 inhibitors in the drying-off of non-human mammals |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2749339A (en) * | 1953-06-08 | 1956-06-05 | Wallace & Tiernan Inc | Mercurial diuretics and method of preparation thereof |
| WO2007093610A1 (en) | 2006-02-15 | 2007-08-23 | Boehringer Ingelheim International Gmbh | Glucopyranosyl-substituted benzonitrile derivatives, pharmaceutical compositions containing such compounds, their use and process for their manufacture |
| WO2007128749A1 (en) | 2006-05-03 | 2007-11-15 | Boehringer Ingelheim International Gmbh | Glucopyranosyl-substituted benzonitrile derivatives, pharmaceutical compositions containing such compounds, their use and process for their manufacture |
| WO2008153945A2 (en) * | 2007-06-06 | 2008-12-18 | University Of South Florida | Nutraceutical co-crystal compositions |
| WO2014016381A1 (en) | 2012-07-26 | 2014-01-30 | Boehringer Ingelheim Vetmedica Gmbh | CRYSTALLINE COMPLEX OF 1-CYANO-2-(4-CYCLOPROPYL-BENZYL)-4-(ß- D-GLUCOPYRANOS-1-YL)-BENZENE, METHODS FOR ITS PREPARATION AND THE USE THEREOF FOR PREPARING MEDICAMENTS |
| WO2023006718A1 (en) | 2021-07-28 | 2023-02-02 | Boehringer Ingelheim Vetmedica Gmbh | Use of sglt-2 inhibitors for the prevention and/or treatment of cardiac diseases in non-human mammals excluding felines, in particular canines |
-
2024
- 2024-04-18 WO PCT/EP2024/060490 patent/WO2024223388A1/en active Pending
- 2024-04-18 CN CN202480026335.2A patent/CN121039113A/en active Pending
- 2024-04-18 AU AU2024264357A patent/AU2024264357A1/en active Pending
- 2024-04-18 KR KR1020257039431A patent/KR20260004451A/en active Pending
-
2025
- 2025-10-23 MX MX2025012705A patent/MX2025012705A/en unknown
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2749339A (en) * | 1953-06-08 | 1956-06-05 | Wallace & Tiernan Inc | Mercurial diuretics and method of preparation thereof |
| WO2007093610A1 (en) | 2006-02-15 | 2007-08-23 | Boehringer Ingelheim International Gmbh | Glucopyranosyl-substituted benzonitrile derivatives, pharmaceutical compositions containing such compounds, their use and process for their manufacture |
| WO2007128749A1 (en) | 2006-05-03 | 2007-11-15 | Boehringer Ingelheim International Gmbh | Glucopyranosyl-substituted benzonitrile derivatives, pharmaceutical compositions containing such compounds, their use and process for their manufacture |
| WO2008153945A2 (en) * | 2007-06-06 | 2008-12-18 | University Of South Florida | Nutraceutical co-crystal compositions |
| WO2014016381A1 (en) | 2012-07-26 | 2014-01-30 | Boehringer Ingelheim Vetmedica Gmbh | CRYSTALLINE COMPLEX OF 1-CYANO-2-(4-CYCLOPROPYL-BENZYL)-4-(ß- D-GLUCOPYRANOS-1-YL)-BENZENE, METHODS FOR ITS PREPARATION AND THE USE THEREOF FOR PREPARING MEDICAMENTS |
| WO2023006718A1 (en) | 2021-07-28 | 2023-02-02 | Boehringer Ingelheim Vetmedica Gmbh | Use of sglt-2 inhibitors for the prevention and/or treatment of cardiac diseases in non-human mammals excluding felines, in particular canines |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12397009B2 (en) | 2019-11-28 | 2025-08-26 | Boehringer Ingelheim Vetmedica Gmbh | Use of SGLT-2 inhibitors in the drying-off of non-human mammals |
Also Published As
| Publication number | Publication date |
|---|---|
| MX2025012705A (en) | 2025-11-03 |
| AU2024264357A1 (en) | 2025-10-09 |
| KR20260004451A (en) | 2026-01-08 |
| CN121039113A (en) | 2025-11-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2877460B1 (en) | CRYSTALLINE COMPLEX OF 1-CYANO-2-(4-CYCLOPROPYL-BENZYL)-4-(ß-D-GLUCOPYRANOS-1-YL)-BENZENE, METHODS FOR ITS PREPARATION AND THE USE THEREOF FOR PREPARING MEDICAMENTS | |
| EP1926720B1 (en) | CRYSTALLINE FORMS OF 1-CHLORO-4-(ß-D-GLUCOPYRANOS-1-YL)-2-(4-ETHYNYL-BENZYL)-BENZENE, METHODS FOR ITS PREPARATION AND THE USE THEREOF FOR PREPARING MEDICAMENTS | |
| AU2006243859B8 (en) | Crystalline form of 1-chloro-4-(beta-D-glucopyranos-1-yl)-2-[4-((S)-tetrahydrofuran-3-yloxy)-benzyl]-benzene, a method for its preparation and the use thereof for preparing medicaments | |
| EP1888551B1 (en) | Crystalline forms of 1-chloro-4-(beta-d-glucopyranos-1-yl)-2-[4-((r)-tetrahydrofuran-3-yloxy)-benzyl]-benzene, a method for its preparation and the use thereof for preparing medicaments | |
| US8283326B2 (en) | Crystalline form of 4-(beta-D-glucopyranos-1-yl)-1-methyl-2-[4-((S)-tetrahydrofuran-3-yloxy)-benzyl]-benzene, a method for its preparation and the use thereof for preparing medicaments | |
| JP3317972B2 (en) | Crystalline N-acetylneuraminic acid derivative and method for producing the same | |
| AU2024264357A1 (en) | Crystalline complexes between velagliflozin and selected co-crystal forming agents, methods for their preparation and the use thereof for preparing medicaments | |
| US20170298087A1 (en) | L-proline complex of sodium-glucose cotransporter 2 inhibitor, monohydrate and crystal form thereof | |
| EP3508479A1 (en) | Crystal forms of valsartan disodium salt | |
| AU2022412842A1 (en) | Novel acid addition salt and crystalline form of (2r,3s)-2-(3-(4,5-dichloro-1h-benzo[d]imidazol-1-yl)propyl)piperidin-3-ol | |
| CN116813542A (en) | Milrinone drug polycrystalline |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24720795 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202517088485 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: AU2024264357 Country of ref document: AU |
|
| ENP | Entry into the national phase |
Ref document number: 2024264357 Country of ref document: AU Date of ref document: 20240418 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2025/012705 Country of ref document: MX |
|
| WWP | Wipo information: published in national office |
Ref document number: MX/A/2025/012705 Country of ref document: MX |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112025023124 Country of ref document: BR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202593036 Country of ref document: EA |
|
| ENP | Entry into the national phase |
Ref document number: 2024720795 Country of ref document: EP Effective date: 20251124 |
|
| ENP | Entry into the national phase |
Ref document number: 1020257039431 Country of ref document: KR Free format text: ST27 STATUS EVENT CODE: A-0-1-A10-A15-NAP-PA0105 (AS PROVIDED BY THE NATIONAL OFFICE) |
|
| WWE | Wipo information: entry into national phase |
Ref document number: KR1020257039431 Country of ref document: KR Ref document number: 2024720795 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWP | Wipo information: published in national office |
Ref document number: 202517088485 Country of ref document: IN |
|
| ENP | Entry into the national phase |
Ref document number: 2024720795 Country of ref document: EP Effective date: 20251124 |
|
| ENP | Entry into the national phase |
Ref document number: 2024720795 Country of ref document: EP Effective date: 20251124 |
|
| ENP | Entry into the national phase |
Ref document number: 2024720795 Country of ref document: EP Effective date: 20251124 |