WO2024223264A1 - Propylene-based copolymer compositon - Google Patents
Propylene-based copolymer compositon Download PDFInfo
- Publication number
- WO2024223264A1 WO2024223264A1 PCT/EP2024/059302 EP2024059302W WO2024223264A1 WO 2024223264 A1 WO2024223264 A1 WO 2024223264A1 EP 2024059302 W EP2024059302 W EP 2024059302W WO 2024223264 A1 WO2024223264 A1 WO 2024223264A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- measured
- ranges
- polyolefin composition
- nmr
- ethylene
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L23/10—Homopolymers or copolymers of propene
- C08L23/14—Copolymers of propene
- C08L23/142—Copolymers of propene at least partially crystalline copolymers of propene with other olefins
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29C—SHAPING OR JOINING OF PLASTICS; SHAPING OF MATERIAL IN A PLASTIC STATE, NOT OTHERWISE PROVIDED FOR; AFTER-TREATMENT OF THE SHAPED PRODUCTS, e.g. REPAIRING
- B29C43/00—Compression moulding, i.e. applying external pressure to flow the moulding material; Apparatus therefor
- B29C43/003—Compression moulding, i.e. applying external pressure to flow the moulding material; Apparatus therefor characterised by the choice of material
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L23/16—Ethene-propene or ethene-propene-diene copolymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2205/00—Polymer mixtures characterised by other features
- C08L2205/02—Polymer mixtures characterised by other features containing two or more polymers of the same C08L -group
- C08L2205/025—Polymer mixtures characterised by other features containing two or more polymers of the same C08L -group containing two or more polymers of the same hierarchy C08L, and differing only in parameters such as density, comonomer content, molecular weight, structure
Definitions
- the present disclosure relates to a composition comprising propylene ethylene copolymers.
- the composition having an high molecular weight and high abrasion resistant.
- the composition is particularly fit for the production of molding objects.
- Abrasion resistance is an important characteristics of articles made from polymeric materials, in particular for polypropylene, for many applications. For example in automotive industry wherein there is the need of durable plastic products as exterior and interior parts especially if they are part of moving parts such as gears.
- a polyolefin composition comprising:
- an object of the present disclosure is a polyolefin composition
- a polyolefin composition comprising:
- a polyolefin composition comprising:
- copolymer has to be intended as a bipolymer containing two monomers, propylene and a ethylene.
- the polyolefin composition of the present disclosure is endowed with the intrinsic viscosity, measured in tetrahydronaphthalene at 135 °C, of the of the fraction insoluble in xylene at 25°C ranges from 6.5 dl/g to 11.3 dl/g; preferably ranges from 6.8 dl/g to 10.3 dl/g; more preferably ranges from 7.2 dl/g to 9.5 dl/g.
- the polyolefin composition of the present disclosure is endowed with the content of ethylene derived units, measured by NMR, of the fraction insoluble in xylene at 25 °C is comprised between 8.3 wt% and 17.3 wt%; preferably is comprised between 10.5 wt% and 15.5 wt%; more preferably is comprised between 12.2 wt% and 14.6 wt%.
- the polyolefin composition of the present disclosure shows a low value of abrasion resistance measured according to ISO 15527 : 2007.
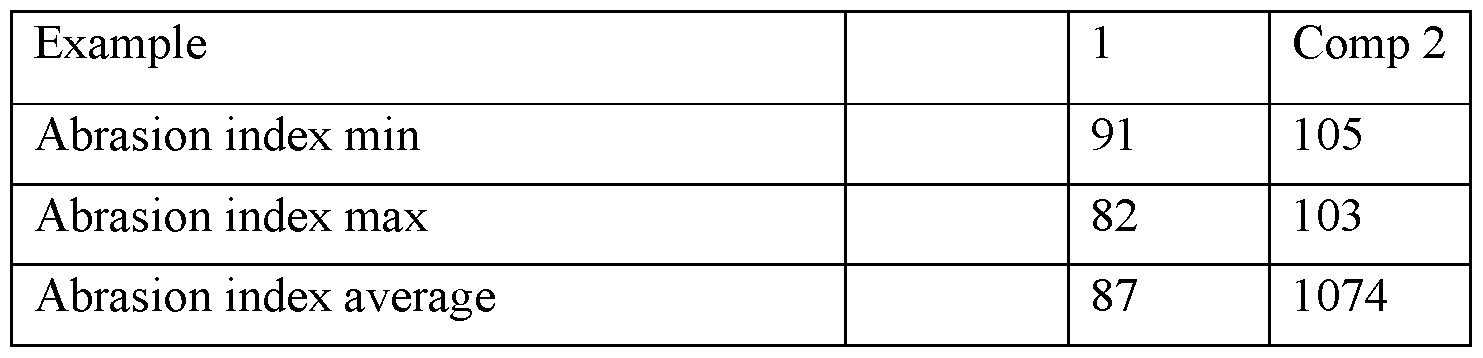
- the value of average abrasion index measured according to ISO 15527 : 2007, on compression molded plaque can be lower than 150 preferably lower than 100; more preferably lower than 93.
- Preferably the higher value of average abrasion index measured according to ISO 15527 : 2007 is 10.
- the propylene copolymer of the present disclosure shows a charpy impact test at 23°C ranging from 40 kJ/m2 to 70.0 kJ/m2; preferably from 50.5 kJ/m2 to 65.0 kJ/m2.
- the propylene copolymer of the present disclosure shows a haze value measured on 1 mm plaque comprised between 37.0 % and 67.4 %; preferably comprised between 47.0 % and 60.4 %.
- the polyolefin composition of the present disclosure if particularly fit for the production of molded articles that can be for example an injection molded article, a blow molded article or a compression molded article.
- the molded article of the present disclosure is a compression molded article.
- the low value of abrasion index renders the molded article of the present disclosure particularly fits for producing automotive articles especially articles subjected to movement.
- composition of the present disclosure can also be prepared by blending component A) and B).
- Components (A) and (B) can be also prepared in a continuous sequential polymerization process, wherein component A) is prepared in the first reactor and component (B) is prepared in the second reactor in the presence of component A) according to the known techniques and operating in gas phase, or in liquid phase in the presence or not of inert diluent, or by mixed liquidgas techniques.
- the polymerization of A) and B) can be carried out in the presence of Ziegler-Natta catalysts.
- An essential component of said catalysts is a solid catalyst component comprising a titanium compound having at least one titanium-halogen bond, and an electron-donor compound, both supported on a magnesium halide in active form.
- Another essential component (co-catalyst) is an organoaluminium compound, such as an aluminium alkyl compound.
- An external donor is optionally added.
- the catalysts generally used in the process of the invention are capable of producing polypropylene with a value of xylene insolubility at ambient temperature greater than 90%, preferably greater than 95%.
- Catalysts having the above mentioned characteristics are well known in the patent literature; particularly advantageous are the catalysts described in US patent 4,399,054 and European patent 45977. Other examples can be found in US patent 4,472,524.
- the solid catalyst components used in said catalysts comprise, as electron-donors (internal donors), compounds selected from the group consisting of ethers, ketones, lactones, compounds containing N, P and/or S atoms, and esters of mono- and dicarboxylic acids.
- Particularly suitable electron-donor compounds are esters of succinic acid (succinates)
- succinates succinates
- radicals R1 and R2 are a C1-C20 linear or branched alkyl, alkenyl, cycloalkyl, aryl, arylalkyl or alkylaryl group, optionally containing heteroatoms; and the radicals R3 and R4 equal to, or different from, each other, are Cl- C20 alkyl, C3-C20 cycloalkyl, C5-C20 aryl, arylalkyl or alkylaryl group with the proviso that at least one of them is a branched alkyl; said compounds being, with respect to the two asymmetric carbon atoms identified in the structure of formula (I), stereoisomers of the type (S,R) or (R,S) [0023] R1 and R2 are preferably C1-C8 alkyl, cycloalkyl, aryl, arylalkyl and alkylaryl groups.
- R1 and R2 are selected from primary alkyls and in particular branched primary alkyls.
- suitable R1 and R2 groups are methyl, ethyl, n-propyl, n-butyl, isobutyl, neopentyl, 2-ethylhexyl.
- Particularly preferred are ethyl, isobutyl, and neopentyl.
- R3 and/or R4 radicals are secondary alkyls like isopropyl, sec-butyl, 2-pentyl, 3 -pentyl or cycloakyls like cyclohexyl, cyclopentyl, cyclohexylmethyl.
- Examples of the above-mentioned compounds are the (S,R) (S,R) forms pure or in mixture, optionally in racemic form, of diethyl 2,3-bis(trimethylsilyl)succinate, diethyl 2,3-bis(2- ethylbutyl)succinate, diethyl 2,3 -dibenzylsuccinate, diethyl 2,3-diisopropylsuccinate, diisobutyl
- Particularly suitable electron-donor compounds are esters of phtalic acid and 1,3- diethers of formula: [0027] wherein R 1 and R n are the same or different and are Ci-Cis alkyl, C3-C18 cycloalkyl or C7-C18 aryl radicals; R 111 and R IV are the same or different and are C1-C4 alkyl radicals; or are the 1,3 -di ethers in which the carbon atom in position 2 belongs to a cyclic or polycyclic structure made up of 5, 6, or 7 carbon atoms, or of 5-n or 6-n' carbon atoms, and respectively n nitrogen atoms and n' heteroatoms selected from the group consisting of N, O, S and Si, where n is 1 or 2 and n' is 1, 2, or 3, said structure containing two or three unsaturations (cyclopolyenic structure), and optionally being condensed with other cyclic structures, or substituted with one or more substituents selected from
- diethers are 2-methyl-2-isopropyl-l,3- dimethoxypropane, 2,2-diisobutyl-l ,3-dimethoxypropane, 2-isopropyl-2-cyclopentyl-l ,3- dimethoxypropane, 2-isopropyl-2-isoamyl-l,3-dimethoxypropane, 9,9-bis (methoxymethyl) fluorene.
- Suitable electron-donor compounds are phthalic acid esters, such as diisobutyl, dioctyl, diphenyl and benzylbutyl phthalate.
- a MgC12»nROH adduct (in particular in the form of spheroidal particles) wherein n is generally from 1 to 3 and ROH is ethanol, butanol or isobutanol, is reacted with an excess of TiC14 containing the electron-donor compound.
- the reaction temperature is generally from 80 to 120° C.
- the solid is then isolated and reacted once more with TiC14, in the presence or absence of the electron-donor compound, after which it is separated and washed with aliquots of a hydrocarbon until all chlorine ions have disappeared.
- the titanium compound expressed as Ti, is generally present in an amount from 0.5 to 10% by weight.
- the quantity of electron- donor compound which remains fixed on the solid catalyst component generally is 5 to 20% by moles with respect to the magnesium dihalide.
- the titanium compounds which can be used for the preparation of the solid catalyst component, are the halides and the halogen alcoholates of titanium. Titanium tetrachloride is the preferred compound.
- the Al-alkyl compounds used as co-catalysts comprise the Al-trialkyls, such as Al- triethyl, Al-triisobutyl, Al-tri-n-butyl, and linear or cyclic Al-alkyl compounds containing two or more Al atoms bonded to each other by way of O or N atoms, or SO4 or SO3 groups.
- Al-trialkyls such as Al- triethyl, Al-triisobutyl, Al-tri-n-butyl, and linear or cyclic Al-alkyl compounds containing two or more Al atoms bonded to each other by way of O or N atoms, or SO4 or SO3 groups.
- the Al-alkyl compound is generally used in such a quantity that the Al/Ti ratio be from 1 to 1000.
- the electron-donor compounds that can be used as external donors include aromatic acid esters such as alkyl benzoates, and in particular silicon compounds containing at least one Si- OR bond, where R is a hydrocarbon radical.
- silicon compounds are (tert-butyl)2Si(OCH3)2, (cyclohexyl)(methyl)Si (OCH3)2, (cyclopentyl)2Si(OCH3)2 and (phenyl)2Si(OCH3)2 and (1,1,2- trimethylpropyl)Si(OCH3)3.
- 1,3 -diethers having the formulae described above can also be used advantageously. If the internal donor is one of these diethers, the external donors can be omitted.
- the component A) are preferably prepared by using catalysts containing a phthalate as internal donor and (cyclopentyl)2Si(OCH3)2 as outside donor, or the said 1,3-diethers as internal donors.
- a further The Ziegler-Natta catalysts that can be used to produce a propylene polymer of the present invention is a solid catalyst component comprising a magnesium halide, a titanium compound having at least a Ti-halogen bond as above described and at least two electron donor compounds selected from succinates and the other being selected from 1,3 diethers.
- a solid catalyst component comprising a magnesium halide, a titanium compound having at least a Ti-halogen bond as above described and at least two electron donor compounds selected from succinates and the other being selected from 1,3 diethers.
- weighting 6 ⁇ 1 mg is heated to 220 ⁇ 1° C at a rate of 20 °C/min and kept at 220 ⁇ 1° C for 2 minutes in nitrogen stream and it is thereafter cooled at a rate of 20° C/min to 40 ⁇ 2° C, thereby kept at this temperature for 2 min to crystallize the sample. Then, the sample is again fused at a temperature rise rate of 20° C/min up to 220° C ⁇ 1. The melting scan is recorded, a thermogram is obtained, and, from this, melting temperatures and crystallization temperatures are read.
- Xylene Solubles at 25°C have been determined according to ISO 16152: 2005; with solution volume of 250 ml, precipitation at 25°C for 20 minutes, 10 of which with the solution in agitation (magnetic stirrer), and drying at 70°C.
- the sample is dissolved by tetrahydronaphthalene at 135 °C and then it is poured into the capillary viscometer.
- the viscometer tube (Ubbelohde type) is surrounded by a cylindrical glass jacket; this setup allows temperature control with a circulating thermostated liquid.
- the downward passage of the meniscus is timed by a photoelectric device.
- the passage of the meniscus in front of the upper lamp starts the counter which has a quartz crystal oscillator.
- the meniscus stops the counter as it passes the lower lamp and the efflux time is registered: this is converted into a value of intrinsic viscosity through
- 13C NMR spectra were acquired on a Bruker AV-600 spectrometer equipped with cry oprobe, operating at 160.91 MHz in the Fourier transform mode at 120 °C.
- P% mol is the molar percentage of propylene content
- MWE and MWP are the molecular weights of ethylene and propylene, respectively.
- the tacticity of Propylene sequences was calculated as mm content from the ratio of the PPP mmTPP (28.90-29.65 ppm) and the whole TPP (29.80-28.37 ppm).
- C2tot is the amount of ethylene in the whole composition
- C2A and C2B are the amount of component A and B
- test specimens 80 x 10 x 4 mm were obtained according to the method ISO 1873-2:2007.
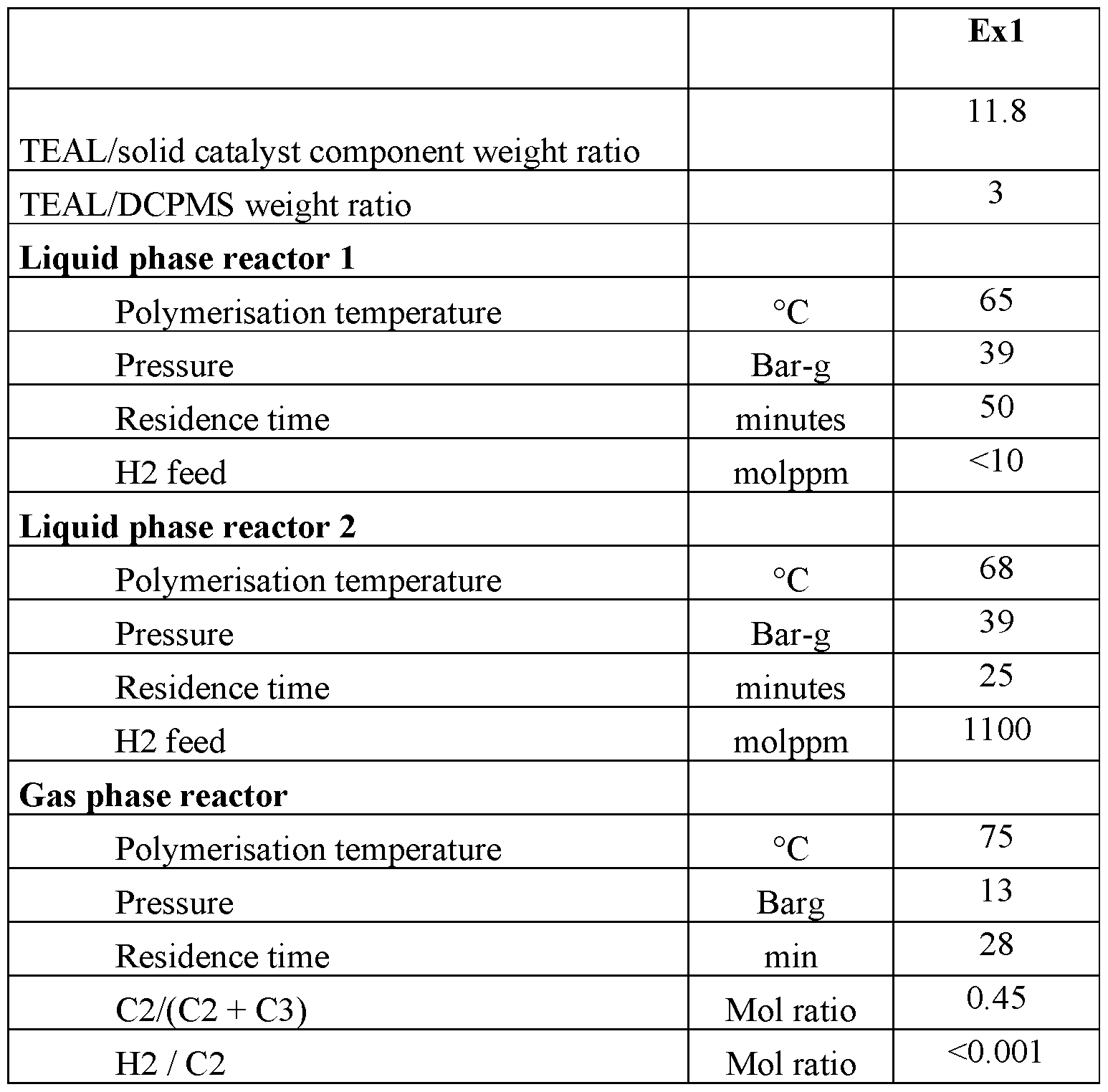
- the solid catalyst used in the following examples was prepared according to the Example 10 of the International Patent Application WO 00/63261.
- Triethylaluminium (TEAL) was used as co-catalyst and dicyclopentyldimethoxysilane as external donor, with the weight ratios indicated in Table 1.
- the polymerization run is conducted in continuous mode in a series of three reactors equipped with devices to transfer the product from one reactor to the one immediately next to it.
- the first two reactors are liquid phase reactors, and the third is a fluid bed gas phase reactor.
- Component (A) is prepared in the first and second reactor, the first and second reactor have the same polymeriziton parameters, while component (B) is prepared in the third.
- Hydrogen is used as molecular weight regulator.
- the gas phase (propylene, ethylene and hydrogen) is continuously analyzed via gaschromatography.
- Table 2 reports the features of the compositons of exmaple 1
- Abrasion test according to ISO 15527:2007, has been measure on compression molded plaque at 250°C, produced with polymers of example 1 and comparative example 2. The results are reported on table 3
- Comparative example 2 Is LP UHM 5000 an ultra-high molecular weight polyethylene designed to have an high abrasion resistance.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Mechanical Engineering (AREA)
- Crystallography & Structural Chemistry (AREA)
- Compositions Of Macromolecular Compounds (AREA)
Abstract
Description
Claims
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202480027655.XA CN121039224A (en) | 2023-04-26 | 2024-04-05 | propylene-based copolymer compositions |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23170149 | 2023-04-26 | ||
| EP23170149.1 | 2023-04-26 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024223264A1 true WO2024223264A1 (en) | 2024-10-31 |
Family
ID=86226764
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/059302 Pending WO2024223264A1 (en) | 2023-04-26 | 2024-04-05 | Propylene-based copolymer compositon |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN121039224A (en) |
| WO (1) | WO2024223264A1 (en) |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0045977A2 (en) | 1980-08-13 | 1982-02-17 | Montedison S.p.A. | Components and catalysts for the polymerization of olefins |
| US4399054A (en) | 1978-08-22 | 1983-08-16 | Montedison S.P.A. | Catalyst components and catalysts for the polymerization of alpha-olefins |
| US4472524A (en) | 1982-02-12 | 1984-09-18 | Montedison S.P.A. | Components and catalysts for the polymerization of olefins |
| EP0361493A1 (en) | 1988-09-30 | 1990-04-04 | Himont Incorporated | Diethers usable in the preparation of Ziegler-Natta catalysts and their preparation |
| EP0728769A1 (en) | 1995-02-21 | 1996-08-28 | Montell North America Inc. | Components and catalysts for the polymerization of olefins |
| WO2000063261A1 (en) | 1999-04-15 | 2000-10-26 | Basell Technology Company B.V. | Components and catalysts for the polymerization of olefins |
| WO2012049204A1 (en) * | 2010-10-14 | 2012-04-19 | Basell Poliolefine Italia S.R.L. | Automotive interior element |
| WO2021089473A1 (en) * | 2019-11-06 | 2021-05-14 | Borealis Ag | Heterophasic propylene ethylene copolymer composition with desirable balance of mechanical properties |
-
2024
- 2024-04-05 CN CN202480027655.XA patent/CN121039224A/en active Pending
- 2024-04-05 WO PCT/EP2024/059302 patent/WO2024223264A1/en active Pending
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4399054A (en) | 1978-08-22 | 1983-08-16 | Montedison S.P.A. | Catalyst components and catalysts for the polymerization of alpha-olefins |
| EP0045977A2 (en) | 1980-08-13 | 1982-02-17 | Montedison S.p.A. | Components and catalysts for the polymerization of olefins |
| US4472524A (en) | 1982-02-12 | 1984-09-18 | Montedison S.P.A. | Components and catalysts for the polymerization of olefins |
| EP0361493A1 (en) | 1988-09-30 | 1990-04-04 | Himont Incorporated | Diethers usable in the preparation of Ziegler-Natta catalysts and their preparation |
| EP0728769A1 (en) | 1995-02-21 | 1996-08-28 | Montell North America Inc. | Components and catalysts for the polymerization of olefins |
| WO2000063261A1 (en) | 1999-04-15 | 2000-10-26 | Basell Technology Company B.V. | Components and catalysts for the polymerization of olefins |
| WO2012049204A1 (en) * | 2010-10-14 | 2012-04-19 | Basell Poliolefine Italia S.R.L. | Automotive interior element |
| WO2021089473A1 (en) * | 2019-11-06 | 2021-05-14 | Borealis Ag | Heterophasic propylene ethylene copolymer composition with desirable balance of mechanical properties |
Non-Patent Citations (1)
| Title |
|---|
| C. J. CARMANR. A. HARRINGTONC. E. WILKES: "Monomer Sequence Distribution in Ethylene-Propylene Rubber Measured by 13C NMR. 3. Use of Reaction Probability Mode", MACROMOLECULES, vol. 10, 1977, pages 536 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN121039224A (en) | 2025-11-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3523370A1 (en) | Propylene-based composition for pipes | |
| US11174379B2 (en) | Compositions obtained from recycled polyolefins | |
| US10323141B2 (en) | Propylene polymer compositions | |
| WO2018202396A1 (en) | Propylene based polymer composition | |
| WO2013050507A1 (en) | Propylene polymer compositions | |
| WO2024223264A1 (en) | Propylene-based copolymer compositon | |
| WO2024099626A1 (en) | Propylene homopolymer | |
| WO2024099628A1 (en) | Propylene homopolymer | |
| WO2024223249A1 (en) | Propylene-based copolymer compositon | |
| EP2825593B1 (en) | Propylene polymer compositions | |
| EP4615889A1 (en) | Molded article comprising propylene homopolymer | |
| WO2023198643A1 (en) | Propylene-based copolymer compositon | |
| EP4536748A1 (en) | Propylene-based copolymer compositon | |
| EP4615888A1 (en) | Molded article comprising a propylene homopolymer composition | |
| EP4508137A1 (en) | Propylene-based copolymer composition | |
| WO2023237341A1 (en) | Propylene-based copolymer compositon | |
| EP4683952A1 (en) | Polypropylene resin compositon | |
| WO2024193834A1 (en) | Polypropylene resin compositon | |
| EP4683953A1 (en) | Polypropylene resin compositon | |
| WO2024193835A1 (en) | Polypropylene resin compositon | |
| WO2026021780A1 (en) | Propylene homopolymer composition | |
| WO2025219023A1 (en) | Propylene-based copolymer composition for pipes |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24717670 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2024717670 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2024717670 Country of ref document: EP Effective date: 20251126 |
|
| ENP | Entry into the national phase |
Ref document number: 2024717670 Country of ref document: EP Effective date: 20251126 |
|
| ENP | Entry into the national phase |
Ref document number: 2024717670 Country of ref document: EP Effective date: 20251126 |