WO2024220930A2 - Mapt-modulating compositions and methods of use thereof - Google Patents
Mapt-modulating compositions and methods of use thereof Download PDFInfo
- Publication number
- WO2024220930A2 WO2024220930A2 PCT/US2024/025577 US2024025577W WO2024220930A2 WO 2024220930 A2 WO2024220930 A2 WO 2024220930A2 US 2024025577 W US2024025577 W US 2024025577W WO 2024220930 A2 WO2024220930 A2 WO 2024220930A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- modified oligonucleotide
- certain embodiments
- positions
- ligands
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/7125—Nucleic acids or oligonucleotides having modified internucleoside linkage, i.e. other than 3'-5' phosphodiesters
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/713—Double-stranded nucleic acids or oligonucleotides
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/14—Type of nucleic acid interfering nucleic acids [NA]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/31—Chemical structure of the backbone
- C12N2310/312—Phosphonates
- C12N2310/3125—Methylphosphonates
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/31—Chemical structure of the backbone
- C12N2310/315—Phosphorothioates
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/32—Chemical structure of the sugar
- C12N2310/321—2'-O-R Modification
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/32—Chemical structure of the sugar
- C12N2310/322—2'-R Modification
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/34—Spatial arrangement of the modifications
- C12N2310/343—Spatial arrangement of the modifications having patterns, e.g. ==--==--==--
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/34—Spatial arrangement of the modifications
- C12N2310/346—Spatial arrangement of the modifications having a combination of backbone and sugar modifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/35—Nature of the modification
- C12N2310/351—Conjugate
- C12N2310/3513—Protein; Peptide
Definitions
- the present disclosure provides compounds, compositions, and methods for modulating the expression or activity of microtubule associated protein tau (MAPT).
- the compounds, compositions, and methods can be used to reduce the expression of MAPT mRNA in a cell or animal.
- the compounds, compositions, and methods can be used to reduce the amount of MAPT protein in a cell or animal.
- the animal has a CNS related disease, disorder or condition.
- the disease, disorder or condition is a neurodegenerative disease, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, or Dravet’s Syndrome.
- a neurodegenerative disease including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, or Dravet’s Syndrome.
- Certain compounds, compositions and methods provided herein are directed to reducing a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP- 17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment in an animal.
- a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP- 17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment in an animal.
- the compounds and compositions provided herein are potent and tolerable and inhibit MAPT expression, which can be used to treat, prevent, ameliorate, or slow progression of a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- the compounds and compositions comprise one or more features that are effective for increasing potency.
- the compounds and compositions comprise one or more features that are effective for increasing tolerability. In certain embodiments, compounds and compositions comprise one or more features that are effective for targeting the compound or composition to a cell or tissue. In certain embodiments, the compounds and compositions are more potent, have greater duration of action or have greater therapeutic value than compounds publicly disclosed.
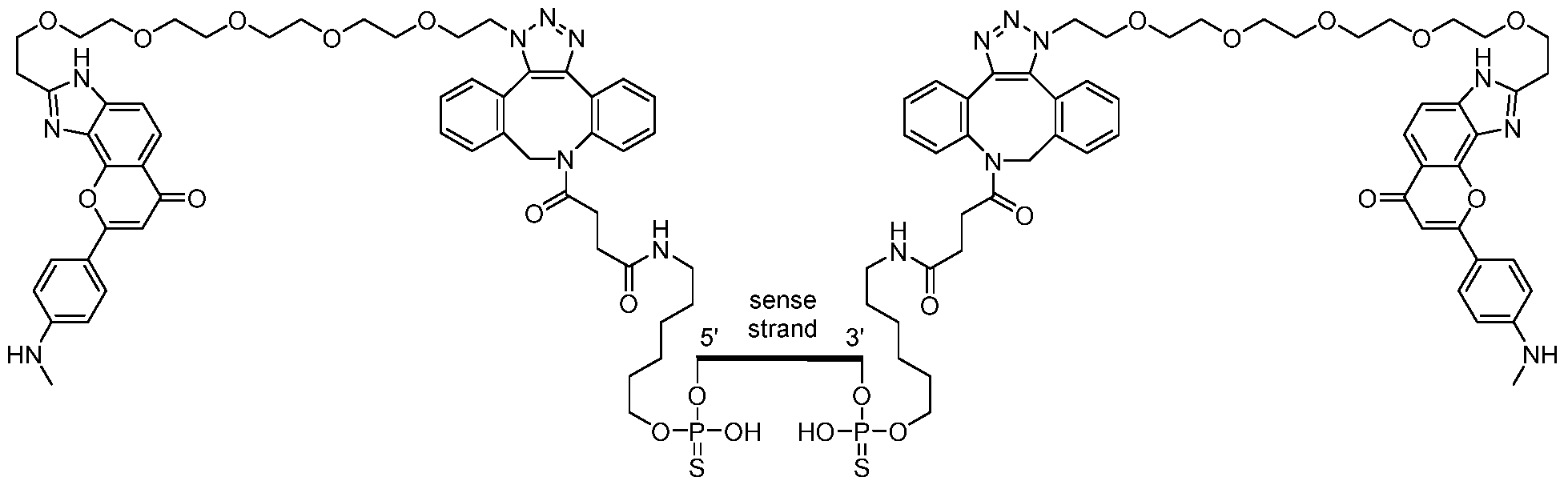
- FIG. 1 shows exemplary compounds of the present disclosure comprising one or more ligands.
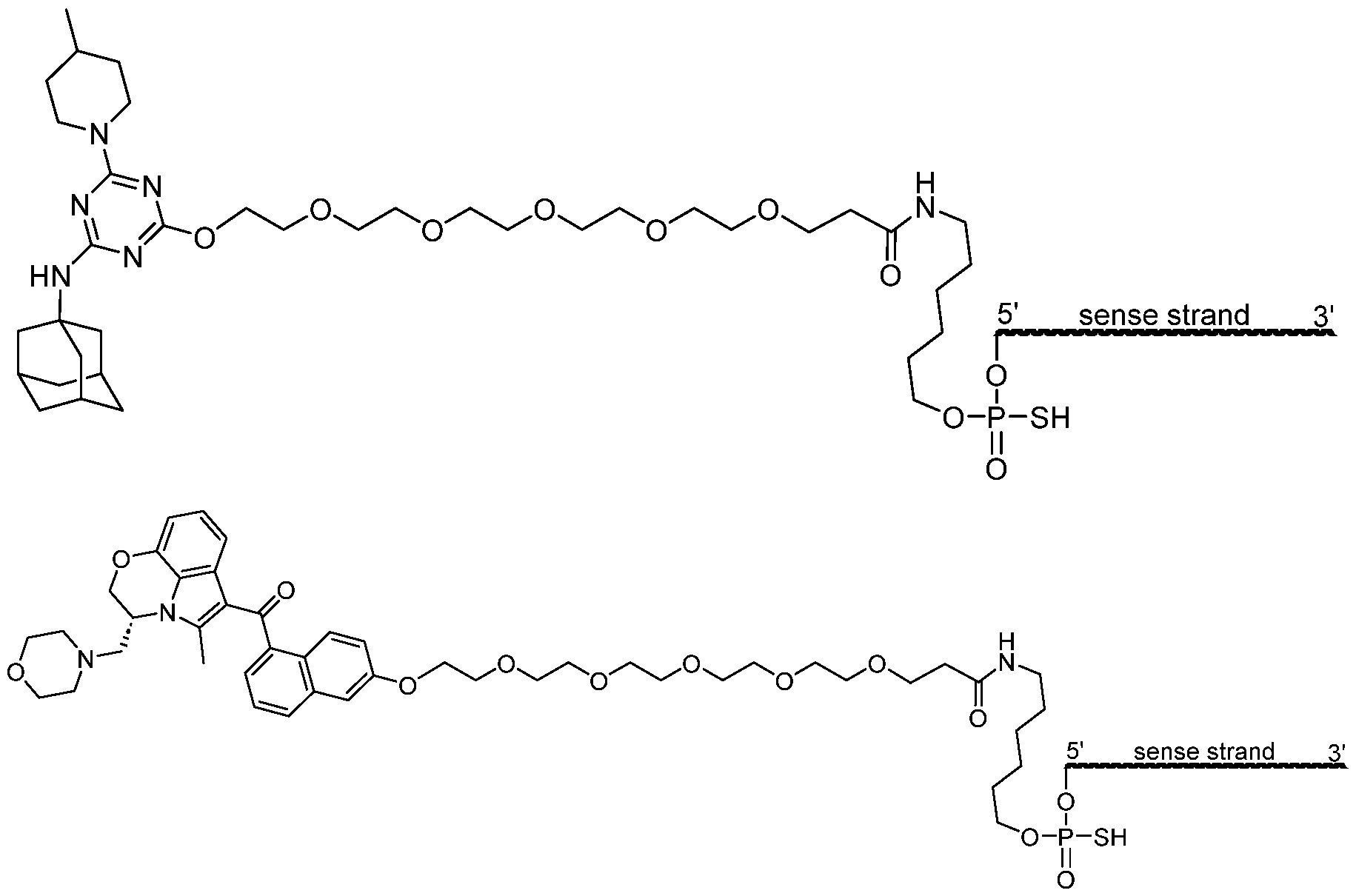
- FIG. 2 shows exemplary compounds of the present disclosure comprising one or more Tropomyosin Receptor Kinase B (TrkB) ligands.
- TrkB Tropomyosin Receptor Kinase B
- FIG. 3 shows exemplary compounds of the present disclosure comprising one or more cannabinoid receptor type 1 (CB 1 ) ligands.
- FIG. 4 shows exemplary compounds of the present disclosure comprising one or more ⁇ 4 ⁇ 1/7 integrin ligands.
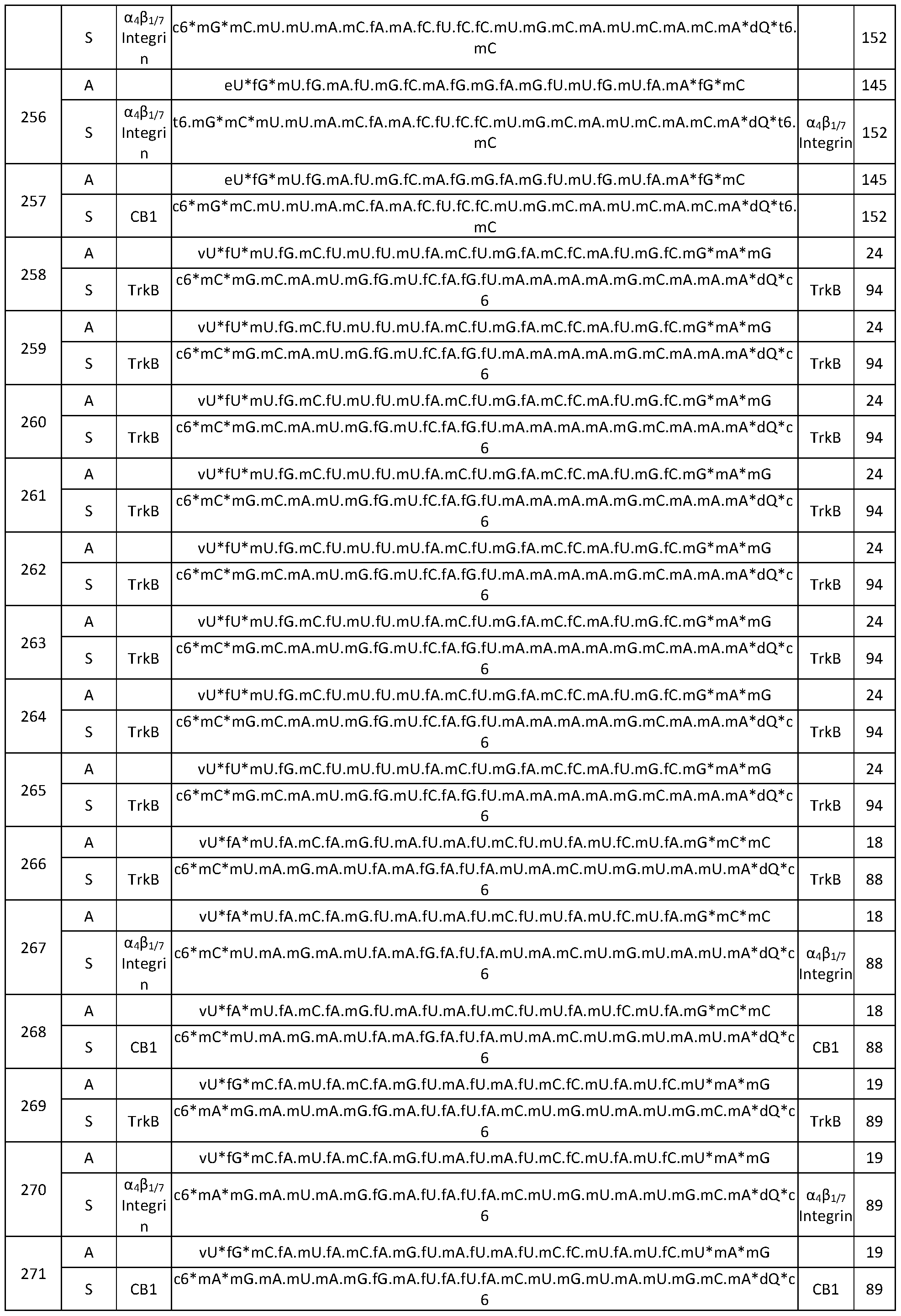
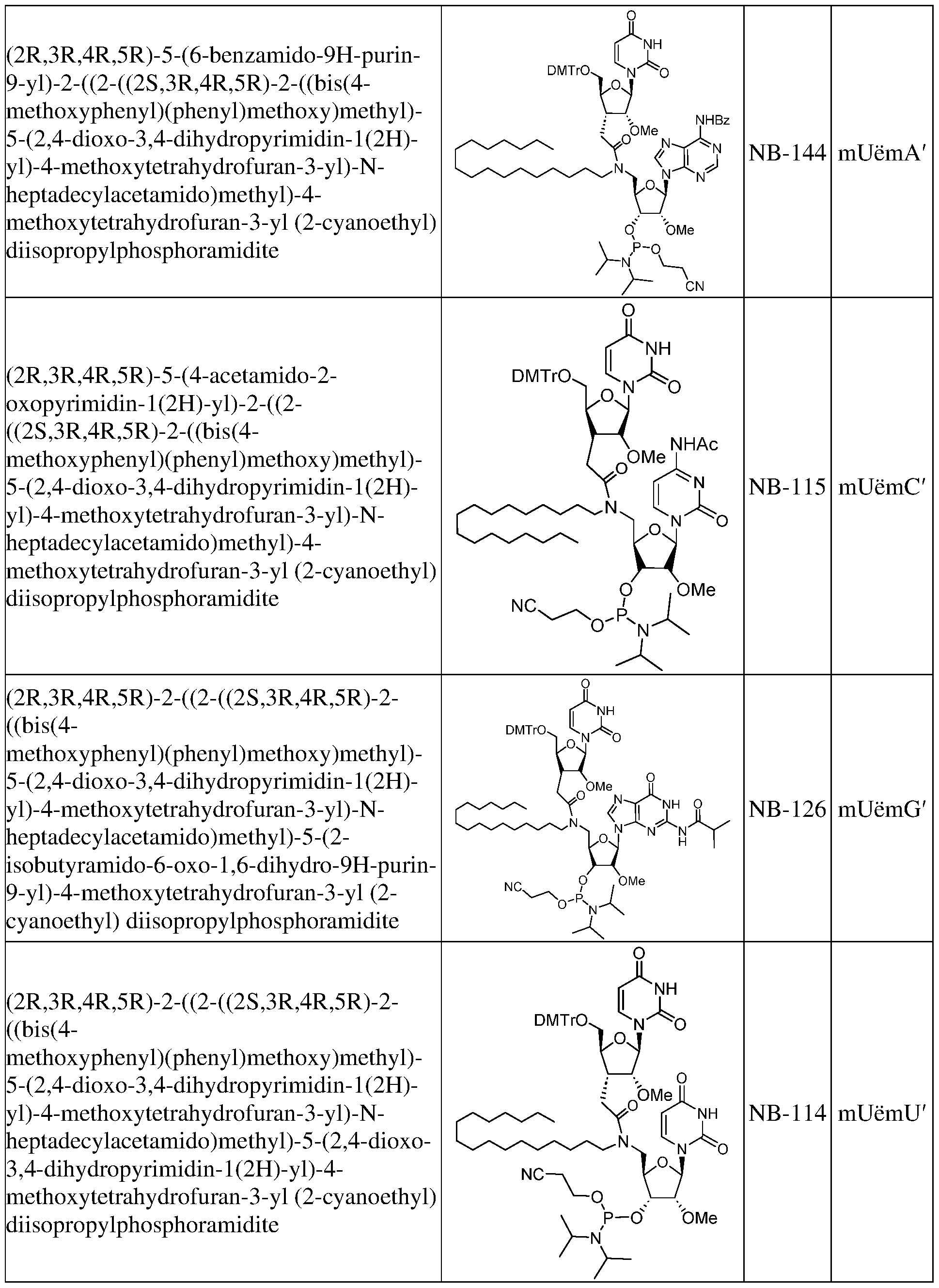
- Oligomeric compounds referenced by Compound Number or Ref ID NO indicate a combination of nucleobase sequence, chemical modification, and motif.
- the use of the singular includes the plural unless specifically stated otherwise.
- the articles “a” and “an” are used herein to refer to one or to more than one (i.e., to at least one) of the grammatical object of the article.
- an element means one element or more than one element, e.g., a plurality of elements.
- the use of “or” means “and/or” unless stated otherwise.
- MMT Microtubule Associated Protein Tau
- SEQ ID NO: 1 GenBank Accession No. NM_001377265.1
- SEQ ID NO: 2 nucleotide sequence of MAPT
- “MAPT mRNA” means an mRNA encoding a MAPT protein.
- MAPT may be referred to in either upper or lower case.
- MAPT specific inhibitor refers to any agent capable of specifically inhibiting MAPT RNA and/or MAPT protein expression or activity at the molecular level.
- MAPT specific inhibitors include nucleic acids (including oligonucleotide compounds), peptides, antibodies, small molecules, and other agents capable of inhibiting the expression of MAPT RNA and/or MAPT protein.
- “2’-O-methoxyethyl” or “2’-MOE” means a 2’-O(CH 2 ) 2 -OCH 3 modification.
- a 2’-O- methoxyethyl modified sugar is a modified sugar with 2’-O(CH 2 ) 2 -OCH 3 in the place of the 2’-OH group of a ribosyl ring.
- “5’ start site” means the nucleotide of the target nucleic acid or region which is aligned to the 3’-most nucleoside of an antisense oligonucleotide.
- 3’ stop site means the nucleotide of the target nucleic acid or region which is aligned to the 5’-most nucleoside of an antisense oligonucleotide.
- “About” means within ⁇ 10% of a value.
- administering refers to routes of introducing a compound or composition provided herein to an individual to perform its intended function.
- routes of administration include, but are not limited to, intrathecal (IT) administration, intracerebroventricular (ICV) administration, parenteral administration, such as subcutaneous administration, intravenous administration, intramuscular administration, intraarterial administration, intraperitoneal administration, or intracranial administration, e.g.
- “Ameliorate” refers to an improvement or lessening of at least one indicator, sign, or symptom of an associated disease, disorder, or condition. In certain embodiments, amelioration includes a delay or slowing in the progression or severity of one or more indicators of a condition or disease. The progression or severity of indicators may be determined by subjective or objective measures, which are known to those skilled in the art.
- “Animal” refers to a human or non-human animal, including, but not limited to, mice, rats, rabbits, dogs, cats, pigs, and non-human primates, including, but not limited to, monkeys and chimpanzees.
- Antisense oligonucleotide or “antisense strand” means an oligonucleotide which includes a region that is complementary to a target nucleic acid, e.g., a MAPT RNA or a region thereof.
- “Complementarity” in reference to an oligonucleotide means the nucleobase sequence of such oligonucleotide or one or more regions thereof that is complementary to the nucleobase sequence of another oligonucleotide or nucleic acid or one or more regions thereof when the two nucleobase sequences are aligned in opposing directions.
- Complementary nucleobases are limited to the following pairs: adenine (A) and thymine ( ⁇ ), adenine (A) and uracil (U), and cytosine (C) and guanine (G) unless otherwise specified.
- Complementary oligonucleotides and/or nucleic acids need not have nucleobase complementarity at each nucleoside and may include one or more nucleobase mismatches.
- “fully complementary” or “100% complementary” in reference to oligonucleotides means that such oligonucleotides have nucleobase matches at each nucleoside without any nucleobase mismatches.
- composition or “pharmaceutical composition” means a mixture of substances suitable for administering to an individual.
- a composition may comprise one or more compounds or salt thereof and a sterile aqueous solution.
- Co-administration means administration of two or more compounds in any manner in which the pharmacological effects of both are manifest in the patient. Co-administration does not require both compounds to be administered in a single pharmaceutical composition, in the same dosage form, by the same route of administration, or at the same time. The effects of both compounds need not manifest themselves at the same time. The effects need only be overlapping for a period of time and need not be coextensive. Co-administration includes parallel or sequential administration of the one or more compounds.
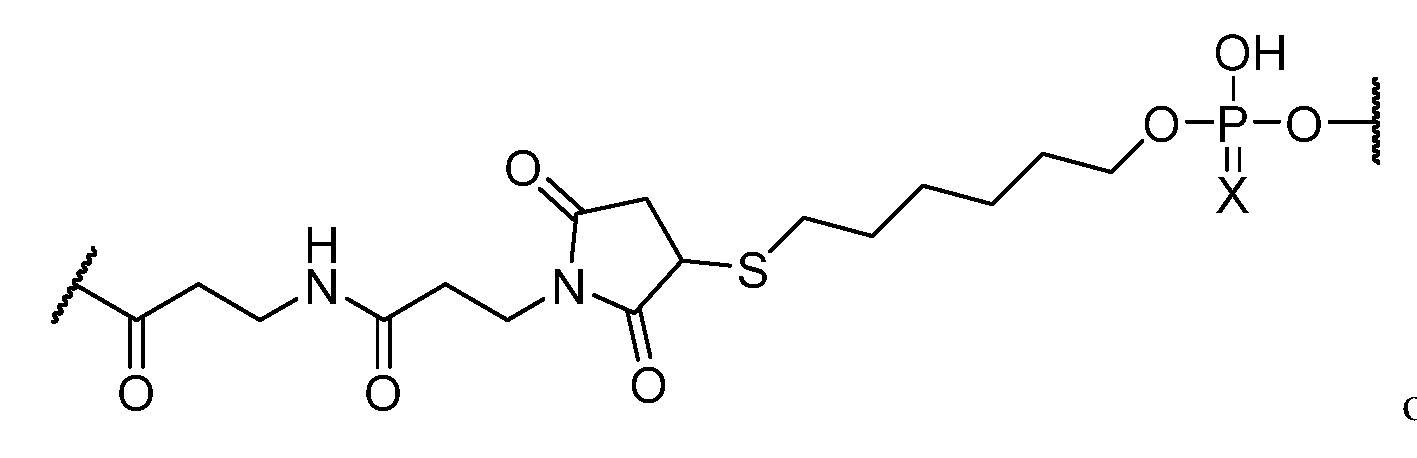
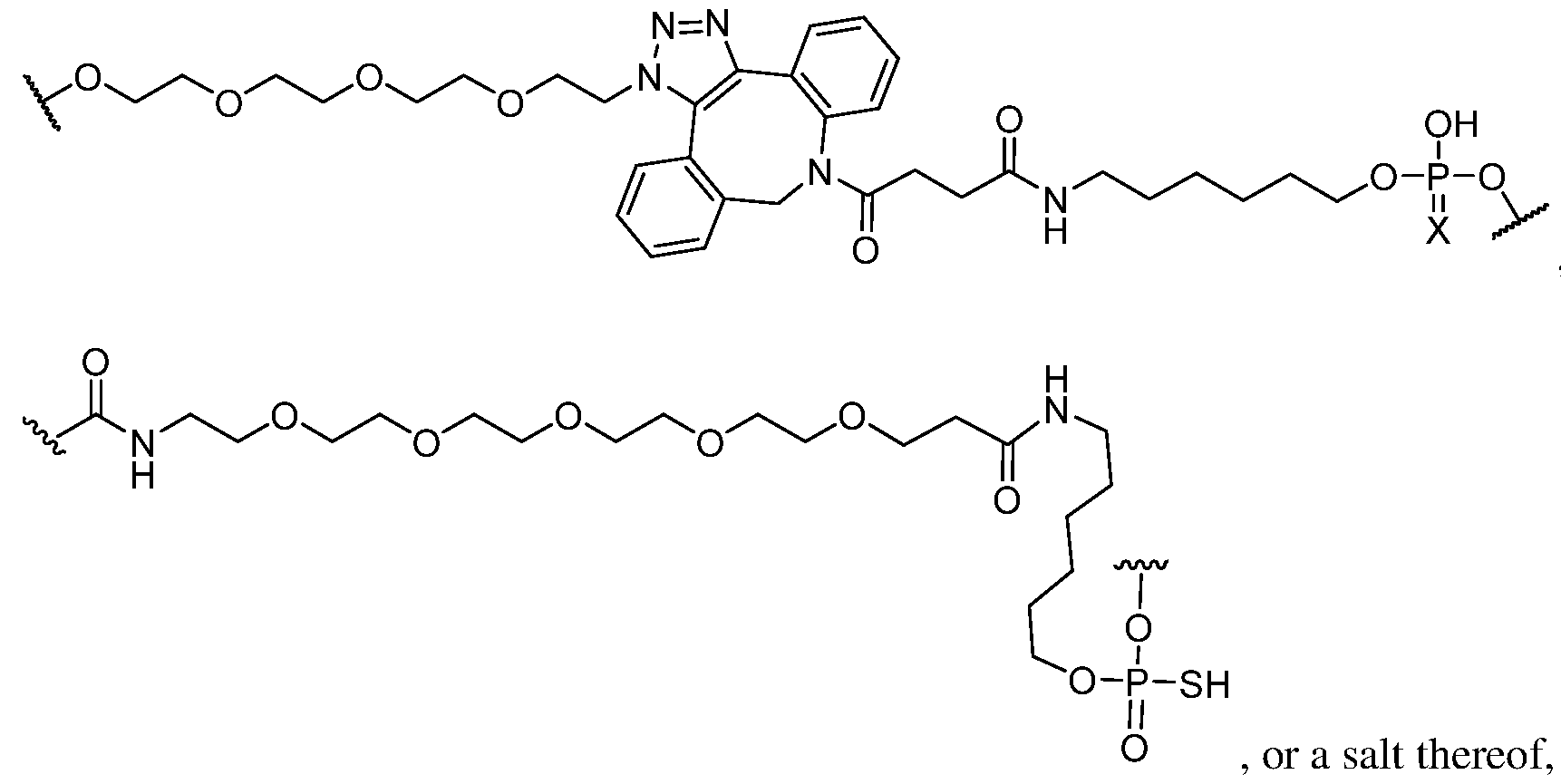
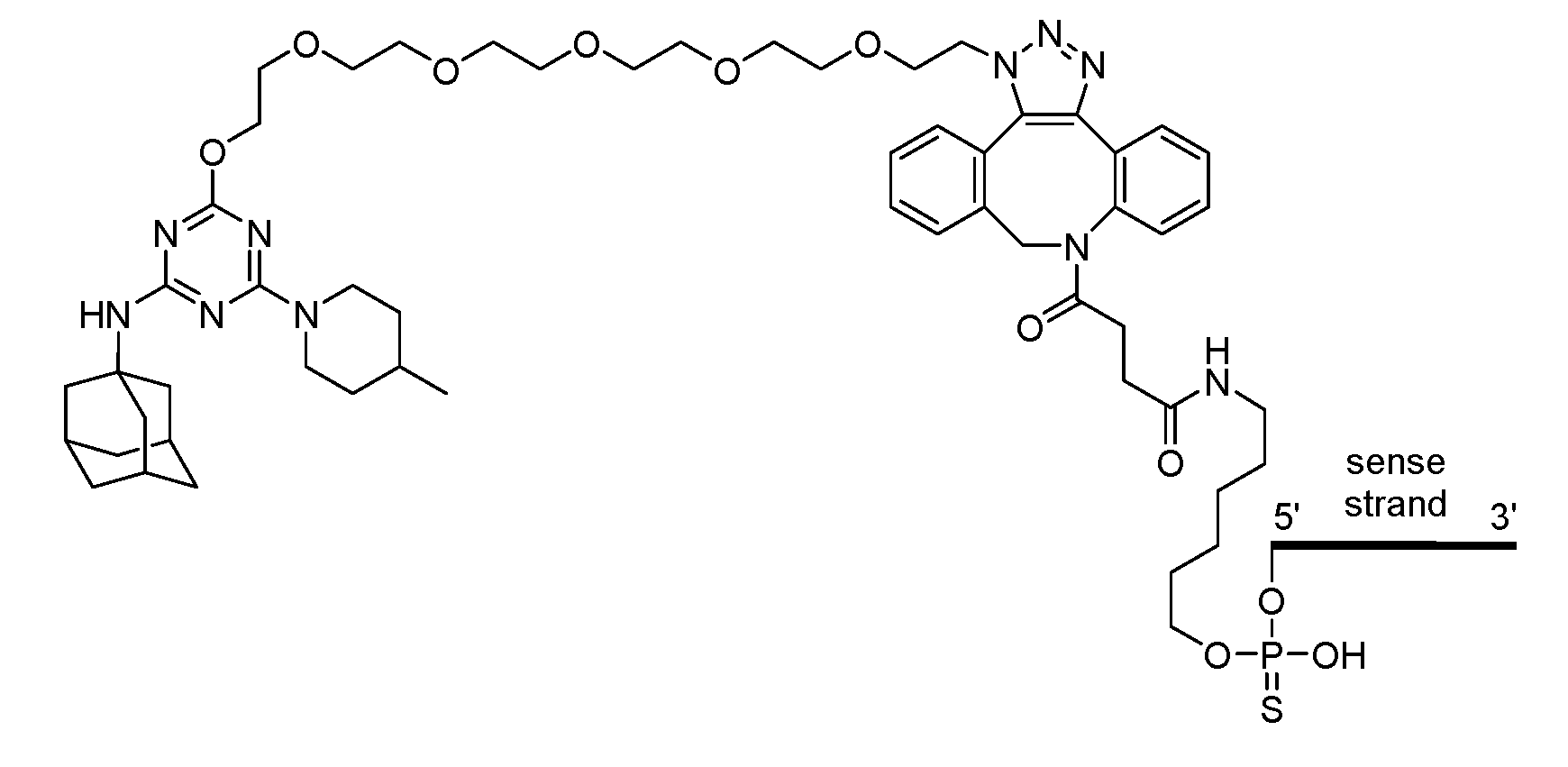
- Conjugate group means a group of atoms that is attached to an oligonucleotide.
- a conjugate group is optionally attached to an oligonucleotide through a conjugate linker.
- a conjugate group may, for example, alter the distribution, targeting, or half-life of a compound into which it is incorporated.
- Conjugate groups include lipids (or lipophilic moieties), ligands, and other targeting moieties.
- Conjugate linker means a group of atoms comprising at least one bond that connects a linked moiety to an oligonucleotide.
- Identity in reference to an oligonucleotide means the nucleobase sequence of such oligonucleotide or one or more regions thereof that matches the nucleobase sequence of another oligonucleotide or nucleic acid or one or more regions thereof. Identity of an oligonucleotide to another oligonucleotide or nucleic acid need not require each nucleobase to match and may include one or more different nucleobases. By contrast, “fully identical” or “100% identity” in reference to oligonucleotides means that such oligonucleotides have the same nucleobase at each relative position over its length as the other oligonucleotide or nucleic acid.
- “Individual” means a human or non-human animal selected for treatment or therapy. “Inhibiting the expression or activity” with reference to a target nucleic acid or protein means to reduce or block the expression or activity of such target relative to the expression or activity in an untreated or control sample and does not necessarily indicate a total elimination of expression or activity.
- the term “internucleoside linkage” is the covalent linkage between adjacent nucleosides in an oligonucleotide.
- “modified internucleoside linkage” means any internucleoside linkage other than a phosphodiester internucleoside linkage.
- “Phosphorothioate internucleoside linkage” is a modified internucleoside linkage in which one of the non-bridging oxygen atoms of a phosphodiester internucleoside linkage is replaced with a sulfur atom.
- Representative internucleoside linkages having a chiral center include but are not limited to alkylphosphonates and phosphorothioates.
- Modified oligonucleotides comprising internucleoside linkages having a chiral center can be prepared as populations of modified oligonucleotides comprising stereorandom internucleoside linkages, or as populations of modified oligonucleotides comprising phosphorothioate linkages in particular stereochemical configurations as further described below.
- chiral internucleoside linkages of modified oligonucleotides described herein can be stereorandom or in a particular stereochemical configuration.
- the compounds of the present disclosure may also contain unnatural proportions of atomic isotopes at one or more of the atoms that constitute such compounds.
- the compounds may be radiolabeled with radioactive isotopes, such as for example tritium ( 3 H), iodine-125 ( 125 I), or carbon-14 ( 14 C). All isotopic variations of the compounds of the present disclosure, whether radioactive or not, are encompassed within the scope of the present disclosure.
- isotopic variant refers to a therapeutic agent (e.g., a compound and/or modified oligonucleotide disclosed herein) that contains an unnatural proportion of an isotope at one or more of the atoms that constitute such a therapeutic agent.
- a therapeutic agent e.g., a compound and/or modified oligonucleotide disclosed herein
- an “isotopic variant” of a therapeutic agent contains unnatural proportions of one or more isotopes, including, but not limited to, hydrogen (H), deuterium ( 2 H), tritium ( 3 H), carbon-11 ( 11 C), carbon-12 ( 12 C), carbon-13 ( 13 C), carbon-14 ( 14 C), nitrogen-13 ( 13 N), nitrogen-14 ( 14 N), nitrogen-15 ( 15 N), oxygen-14 ( 14 O), oxygen-15 ( 15 O), oxygen-16 ( 16 O), oxygen-17 ( 17 O), oxygen-18 ( 18 O), fluorine-17 ( 17 F), fluorine-18 ( 18 F), phosphorus-31 ( 31 P), phosphorus-32 ( 32 P), phosphorus-33 ( 33 P), sulfur-32 ( 32 S), sulfur-33 ( 33 S), sulfur-34 ( 34 S), sulfur-35 ( 35 S), sulfur-36 ( 36 S), chlorine-35 ( 35 Cl), chlorine-36 ( 36 Cl), chlorine-37 ( 37 Cl), bromine-79 ( 79 Br), bromine-81 ( 81 Br), iodine 123 (
- an “isotopic variant” of a therapeutic agent contains unnatural proportions of one or more isotopes, including, but not limited to, hydrogen (H), deuterium ( 2 H), tritium ( 3 H), carbon-11 ( 11 C), carbon-12 ( 12 C), carbon-13 ( 13 C), carbon-14 ( 14 C), nitrogen-13 ( 13 N), nitrogen-14 ( 14 N), nitrogen-15 ( 15 N), oxygen-14 ( 14 O), oxygen-15 ( 15 O), oxygen-16 ( 16 O), oxygen-17 ( 17 O), oxygen-18 ( 18 O), fluorine-17 ( 17 F), fluorine-18 ( 18 F), phosphorus-31 ( 31 P), phosphorus-32 ( 32 P), phosphorus-33 ( 33 P), sulfur-32 ( 32 S), sulfur-33 ( 33 S), sulfur-34 ( 34 S), sulfur-35 ( 35 S), sulfur-36 ( 36 S), chlorine-35 ( 35 Cl), chlorine-36 ( 36 Cl), chlorine-37 ( 37 Cl), bromine-79 ( 79 Br), bromine-81 ( 81 Br), iodine 123 (
- any hydrogen can be 2 H, for example, or any carbon can be 13 C, for example, or any nitrogen can be 15 N, for example, or any oxygen can be 18 O, for example, where feasible according to the judgment of one of skill.
- an “isotopic variant” of a therapeutic agent contains unnatural proportions of deuterium (D).
- Lipid or “lipophilic moiety” refers to an aliphatic, cyclic (such as alicyclic), or polycyclic (such as polyalicyclic) compound, such as a steroid (e.g., sterol) or a linear or branched aliphatic hydrocarbon.
- a steroid e.g., sterol
- a linear or branched aliphatic hydrocarbon such as a steroid (e.g., sterol) or a linear or branched aliphatic hydrocarbon.
- lipid includes cholesterol, retinoic acid, cholic acid, adamantane acetic acid, 1-pyrene butyric acid, dihydrotestosterone, 1,3-bis- O(hexadecyl)glycerol, geranyloxyhexyanol, hexadecylglycerol, borneol, menthol, 1,3- propanediol, heptadecyl group, palmitic acid, myristic acid, O3-(oleoyl)lithocholic acid, O3- (oleoyl)cholenic acid, ibuprofen, naproxen, dimethoxytrityl, or phenoxazine.
- lipid includes a saturated or unsaturated C 4 -C 30 hydrocarbon chain (e.g., C 4 -C 30 alkyl or alkenyl).
- the lipophilic moiety contains a saturated or unsaturated C 5 -C 20 hydrocarbon chain (e.g., a linear C 5 -C 20 alkyl or alkenyl).
- the lipophilic moiety contains a saturated or unsaturated C 14 -C 20 hydrocarbon chain (e.g., a linear C 14 -C 20 alkyl or alkenyl).
- the lipophilic moiety contains a saturated or unsaturated C 6 -C 18 hydrocarbon chain (e.g., a linear C 6 -C 18 alkyl or alkenyl). In certain embodiments, the lipophilic moiety contains a saturated or unsaturated C16 hydrocarbon chain (e.g., a linear C 16 alkyl or alkenyl). In certain embodiments, the lipophilic moiety contains a saturated or unsaturated C 17 hydrocarbon chain (e.g., a linear C 17 alkyl or alkenyl). In certain embodiments, the lipophilic moiety contains a saturated or unsaturated C18 hydrocarbon chain (e.g., a linear C 18 alkyl or alkenyl).
- the lipophilic moiety contains a saturated or unsaturated C 22 hydrocarbon chain (e.g., a linear C 22 alkyl or alkenyl).
- “Mismatch” or “non-complementary” means a nucleobase of a first oligonucleotide or nucleic acid that is not complementary to the corresponding nucleobase of a second oligonucleotide or nucleic acid when the first oligonucleotide/nucleic acid and second oligonucleotide/nucleic acid are aligned in an antiparallel orientation.
- nucleobases including, but not limited to, a universal nucleobase, inosine, and hypoxanthine, are capable of hybridizing with at least one nucleobase but are still mismatched or non- complementary with respect to the nucleobase to which they are hybridized.
- a nucleobase of a first oligonucleotide/nucleic acid that is not capable of hybridizing to the corresponding nucleobase of a second oligonucleotide/nucleic acid when the first and second oligonucleotides are aligned in an antiparallel orientation is a mismatch or non-complementary nucleobase.
- Modified oligonucleotide means an oligonucleotide, wherein at least one sugar, nucleobase, or internucleoside linkage is modified. “Modulating” refers to changing or adjusting a feature in a cell, tissue, organ or organism. For example, modulating MAPT RNA can mean to increase or decrease the level of MAPT RNA and/or MAPT protein in a cell, tissue, organ or organism. A “modulator” effects the change in the cell, tissue, organ or organism. For example, a MAPT compound can be a modulator that decreases the amount of MAPT RNA and/or MAPT protein in a cell, tissue, organ or organism.
- “Motif” means the pattern of unmodified and modified sugar moieties, nucleobases, and/or internucleoside linkages, in an oligonucleotide.
- Nucleic acid refers to molecules composed of monomeric nucleotides. A nucleic acid includes, but is not limited to, ribonucleic acids (RNA), deoxyribonucleic acids (DNA), single-stranded nucleic acids, and double-stranded nucleic acids.
- “Nucleobase” means a heterocyclic moiety capable of pairing with a base of another nucleic acid.
- nucleobase sequence means the order of contiguous nucleobases in a nucleic acid or oligonucleotide independent of any sugar or internucleoside linkage.
- Nucleoside means a compound comprising a nucleobase and a sugar moiety. The nucleobase and sugar moiety are each, independently, unmodified or modified.
- Modified nucleoside means a nucleoside comprising a modified nucleobase and/or a modified sugar moiety. Modified nucleosides include abasic nucleosides, which lack a nucleobase.
- Oligonucleotides means a compound comprising one or more oligonucleotides and optionally one or more additional features, such as a conjugate group or terminal group.
- oligomeric compounds include single-stranded and double- stranded compounds, such as, oligonucleotides, antisense oligonucleotides, interfering RNA compounds (RNAi compounds), microRNA targeting oligonucleotides, occupancy-based compounds (e.g., mRNA processing or translation blocking compounds and splicing compounds).
- RNAi compounds interfering RNA compounds
- microRNA targeting oligonucleotides e.g., occupancy-based compounds (e.g., mRNA processing or translation blocking compounds and splicing compounds).
- RNAi compounds include double-stranded compounds (e.g., short-interfering RNA (siRNA) and double-stranded RNA (dsRNA)) and single-stranded compounds (e.g., single-stranded siRNA (ssRNA), single-stranded RNAi (ssRNAi), short hairpin RNA (shRNA) and microRNA mimics) which work at least in part through the RNA-induced silencing complex (RISC) pathway resulting in sequence specific degradation and/or sequestration of a target nucleic acid through a process known as RNA interference (RNAi).
- siRNA single-stranded siRNA
- ssRNAi single-stranded RNAi
- shRNA short hairpin RNA
- microRNA mimics RNA-induced silencing complex
- RNAi compound is meant to be equivalent to other terms used to describe nucleic acid compounds that are capable of mediating sequence-specific RNA interference, for example, interfering RNA (iRNA), iRNA agent, RNAi agent, short interfering oligonucleotide, short interfering nucleic acid, short interfering modified oligonucleotide, chemically modified siRNA, and others. Additionally, the term “RNAi” is meant to be equivalent to other terms used to describe sequence-specific RNA interference. “Oligomeric duplex” means a duplex formed by two oligomeric compounds having complementary nucleobase sequences.
- Each oligomeric compound of an oligomeric duplex may be referred to as a “duplexed oligomeric compound.”
- the oligonucleotides of each oligomeric compound of an oligomeric duplex may include non-complementary overhanging nucleosides.
- duplexed oligomeric compound and “modified oligonucleotide” are used interchangeably.
- oligomeric duplex and “compound” are used interchangeably.
- “Oligonucleotide” means a polymer of linked nucleosides, each of which can be modified or unmodified, independent from one another.
- Parent administration means administration through injection or infusion.
- Parenteral administration includes subcutaneous administration, intravenous administration, intramuscular administration, intraarterial administration, intraperitoneal administration, or intracranial administration, e.g. intrathecal or intracerebroventricular administration.
- “Pharmaceutically acceptable carrier or diluent” means any substance suitable for use in administering to an individual.
- a pharmaceutically acceptable carrier or diluent aids the administration of a compound to and absorption by an individual and can be included in the compositions of the present disclosure without causing a significant adverse toxicological effect on the patient.
- Non-limiting examples of pharmaceutically acceptable excipients include water, NaCl, normal saline solutions, and the like.
- a pharmaceutically acceptable carrier can be a sterile aqueous solution, such as PBS or water-for-injection.
- a pharmaceutically acceptable salt means or refers to physiologically and pharmaceutically acceptable salts of compounds, such as oligomeric compounds or oligonucleotides, i.e., salts that retain the desired biological activity of the parent compound and do not impart undesired toxicological effects thereto.
- Tropomyosin Receptor Kinase B” or “TrkB,” as may be used interchangeably herein, means the receptor for brain-derived neurotrophic factor (BDNF) protein encoded by the NTRK2 gene.
- BDNF brain-derived neurotrophic factor
- TrkB is also known as tyrosine receptor kinase B, BDNF/NT-3 growth factors receptor and neurotrophic tyrosine kinase, receptor, type 2.
- a pharmaceutically acceptable salt is any salt of a compound provided herein which retains its biological properties and which is not toxic or otherwise undesirable for pharmaceutical use.
- the pharmaceutically acceptable salts of the therapeutic agents disclosed herein include salts that are prepared with relatively nontoxic acids or bases, depending on the particular substituents found on the compounds or modified oligonucleotides described herein. When compounds of the present disclosure contain relatively acidic functionalities, base addition salts can be obtained by contacting the neutral form of such compounds with a sufficient amount of the desired base, either neat or in a suitable inert solvent.
- acid addition salts can be obtained by contacting the neutral form of such compounds with a sufficient amount of the desired acid, either neat or in a suitable inert solvent.
- the compounds of the present disclosure may exist as salts, such as with pharmaceutically acceptable acids.
- Such salts may be derived from a variety of organic and inorganic counter-ions well known in the art.
- Such salts include, but are not limited to: (1) acid addition salts formed with organic or inorganic acids such as hydrochloric, hydrobromic, sulfuric, nitric, phosphoric, sulfamic, acetic, trifluoroacetic, trichloroacetic, propionic, hexanoic, cyclopentylpropionic, glycolic, glutaric, pyruvic, lactic, malonic, succinic, sorbic, ascorbic, malic, maleic, fumaric, tartaric, citric, benzoic, 3-(4-hydroxybenzoyl)benzoic, picric, cinnamic, mandelic, phthalic, lauric, methanesulfonic, ethanesulfonic, 1,2-ethane- disulfonic, 2-hydroxyethanesulfonic, benzenesulfonic, 4-chlorobenzenesulfonic, 2- naphthalenesulfonic, 4-to
- Pharmaceutically acceptable salts further include, by way of example only and without limitation, sodium, potassium, calcium, magnesium, ammonium, tetraalkylammonium, and the like, and when the compound contains a basic functionality, salts of non-toxic organic or inorganic acids, such as hydrohalides, e.g.
- the pharmaceutically acceptable salt of the compounds and modified oligonucleotides disclosed herein is a sodium or a potassium salt. In some embodiments, the pharmaceutically acceptable salt of the compounds and modified oligonucleotides disclosed herein is a sodium salt.
- the neutral forms of the compounds are preferably regenerated by contacting the salt with a base or acid and isolating the parent compound in the conventional manner. The parent form of the compound may differ from the various salt forms in certain physical properties, such as solubility in polar solvents.
- compounds of the present disclosure contain both basic and acidic functionalities that allow the compounds to be converted into either base or acid addition salts.
- the neutral forms of the compounds may be regenerated by contacting the salt with a base or acid and isolating the parent compound in a conventional manner.
- the parent form of the compounds differs from the various salt forms in certain physical properties, such as solubility in polar solvents, but, unless specifically indicated, the salts disclosed herein are equivalent to the parent form of the compound for the purposes of the present disclosure.
- “Pharmaceutical agent” means a compound that provides a therapeutic benefit when administered to an individual.
- “Phosphorothioate linkage” means a modified phosphate linkage in which one of the non-bridging oxygen atoms is replaced with a sulfur atom.
- “Portion” means a defined number of contiguous (i.e., linked) nucleobases of a nucleic acid. In certain embodiments, a portion is a defined number of contiguous nucleobases of a target nucleic acid. In certain embodiments, a portion is a defined number of contiguous nucleobases of an oligonucleotide. “Prevent” refers to delaying or forestalling the onset, development or progression of a disease, disorder, or condition for a period of time.
- RNA interference compound or “RNAi compound” means a compound that acts, at least in part, through an RNA-induced silencing complex (RISC) pathway or Ago2, but not through RNase ⁇ , to modulate a target nucleic acid and/or protein encoded by a target nucleic acid.
- RNAi compounds include, but are not limited to double-stranded siRNA, single- stranded siRNA, and microRNA, including microRNA mimics.
- Sense oligonucleotide” or “sense strand” means the strand of a double-stranded compound that includes a region that is substantially complementary to a region of the antisense strand of the compound.
- Specifically inhibit with reference to a target nucleic acid or protein means to reduce or block expression or activity of the target nucleic acid or protein while minimizing or eliminating effects on non-target nucleic acids or proteins.
- “Subunit” with reference to an oligonucleotide means a nucleotide, nucleoside, nucleobase or sugar or a modified nucleotide, nucleoside, nucleobase or sugar as provided herein.
- Target nucleic acid “target RNA,” and “nucleic acid target” all mean a nucleic acid capable of being targeted by compounds described herein.
- “Target region” means a portion of a target nucleic acid to which one or more compounds is targeted.
- Targeting moiety means a conjugate group that provides an enhanced affinity for a selected target, e.g., molecule, cell or cell type, compartment, e.g., a cellular or organ compartment, tissue, organ or region of the body, as, e.g., compared to a compound absent such a moiety.
- Terminal group means a chemical group or group of atoms that is covalently linked to a terminus of an oligonucleotide.
- “Therapeutically effective amount” or “effective amount” means an amount of a compound, pharmaceutical agent, or composition that provides a therapeutic benefit to an individual.
- a “therapeutically effective amount” or “effective amount” is an amount sufficient for a compound to accomplish a stated purpose relative to the absence of the compound (e.g., achieve the effect for which it is administered, treat, prevent or ameliorate a disease or reduce one or more symptoms of a disease or condition).
- An example of a “therapeutically effective amount” or “effective amount” is an amount sufficient to contribute to the treatment, prevention, amelioration, or reduction of a symptom or symptoms of a disease.
- a “reduction” of a symptom or symptoms means decreasing of the severity or frequency of the symptom(s), or elimination of the symptom(s).
- a “prophylactically effective amount” of a drug is an amount of a drug that, when administered to a subject, will have the intended prophylactic effect, e.g., preventing or delaying the onset (or reoccurrence) of an injury, disease, pathology or condition, or reducing the likelihood of the onset (or reoccurrence) of an injury, disease, pathology, or condition, or their symptoms.
- a therapeutically effective amount will show an increase or decrease of at least 5%, 10%, 15%, 20%, 25%, 40%, 50%, 60%, 75%, 80%, 90%, or at least 100%.
- Therapeutic efficacy can also be expressed as “-fold” increase or decrease.
- a therapeutically effective amount can have at least a 1.2- fold, 1.5-fold, 2-fold, 5-fold, or more effect over a control.
- treating refers to any indicia of success in the therapy or amelioration of an injury, disease, pathology or condition, including any objective or subjective parameter such as abatement; remission; diminishing of symptoms or making the injury, pathology or condition more tolerable to the patient; slowing in the rate of degeneration or decline; making the final point of degeneration less debilitating; improving a patient's physical or mental well-being.
- the treatment or amelioration of symptoms can be based on objective or subjective parameters, including the results of a physical examination.
- the term “treating” and conjugations thereof, may include prevention of an injury, pathology, condition, or disease.
- treating is preventing.
- treating does not include preventing.
- Treating” or “treatment” as used herein also broadly includes any approach for obtaining beneficial or desired results in a subject's condition, including clinical results.
- Beneficial or desired clinical results can include, but are not limited to, alleviation or amelioration of one or more symptoms or conditions, diminishment of the extent of a disease, stabilizing (i.e., not worsening) the state of disease, prevention of a disease’s transmission or spread, delay or slowing of disease progression, amelioration or palliation of the disease state, diminishment of the reoccurrence of disease, and remission, whether partial or total and whether detectable or undetectable.
- treatment as used herein includes any cure, amelioration, or prevention of a disease.
- Treatment may prevent the disease from occurring; inhibit the disease’s spread; relieve the disease’s symptoms, fully or partially remove the disease’s underlying cause, shorten a disease’s duration, or do a combination of these things.
- Treating” and “treatment” as used herein include prophylactic treatment.
- Treatment methods include administering to a subject a therapeutically effective amount of a compound described herein.
- the administering step may consist of a single administration or may include a series of administrations.
- the length of the treatment period depends on a variety of factors, such as the severity of the condition, the age of the patient, the concentration of the compound, the activity of the compositions used in the treatment, or a combination thereof.
- the effective dosage of an agent used for the treatment or prophylaxis may increase or decrease over the course of a particular treatment or prophylaxis regime. In some instances, chronic administration may be required.
- the compositions are administered to the subject in an amount and for a duration sufficient to treat the patient. “Treat” refers to administering a compound or pharmaceutical composition to an animal in order to effect an alteration or improvement of a disease, disorder, or condition in the animal.
- Certain compounds of the present disclosure possess asymmetric carbon atoms (optical or chiral centers) or double bonds; the enantiomers, racemates, diastereomers, tautomers, geometric isomers, stereoisometric forms that may be defined, in terms of absolute stereochemistry, as (R)- or (S)- or, as (D)- or (L)-for amino acids, and individual isomers are encompassed within the scope of the present disclosure.
- the compounds of the present disclosure do not include those that are known in art to be too unstable to synthesize and/or isolate.
- the present disclosure is meant to include compounds in racemic and optically pure forms.
- Optically active (R)- and (S)-, or (D)- and (L)-isomers may be prepared using chiral synthons or chiral reagents or resolved using conventional techniques.
- the compounds described herein contain olefinic bonds or other centers of geometric asymmetry, and unless specified otherwise, it is intended that the compounds include both E and Z geometric isomers.
- the term “isomers” refers to compounds having the same number and kind of atoms, and hence the same molecular weight, but differing in respect to the structural arrangement or configuration of the atoms.
- tautomer refers to one of two or more structural isomers which exist in equilibrium and which are readily converted from one isomeric form to another.
- chirally enriched population means a plurality of molecules of identical molecular formula, wherein the number or percentage of molecules within the population that contain a particular stereochemical configuration at a particular chiral center is greater than the number or percentage of molecules expected to contain the same particular stereochemical configuration at the same particular chiral center within the population if the particular chiral center were stereorandom. Chirally enriched populations of molecules having multiple chiral centers within each molecule may contain one or more stereorandom chiral centers.
- the molecules are modified oligonucleotides. In certain embodiments, the molecules are compounds comprising modified oligonucleotides.
- structures depicted herein are also meant to include compounds which differ only in the presence of one or more isotopically enriched atoms.
- compounds having the present structures except for the replacement of a hydrogen by a deuterium or tritium, or the replacement of a carbon by 13 C- or 14 C-enriched carbon are within the scope of this disclosure.
- “stereorandom chiral center” in the context of a population of molecules of identical molecular formula means a chiral center having a random stereochemical configuration.
- the number of molecules having the (S) configuration of the stereorandom chiral center may be but is not necessarily the same as the number of molecules having the (R) configuration of the stereorandom chiral center.
- the stereochemical configuration of a chiral center is considered random when it is the results of a synthetic method that is not designed to control the stereochemical configuration.
- a stereorandom chiral center is a stereorandom phosphorothioate internucleoside linkage.
- MAPT expression is inhibited.
- MAPT translation is inhibited.
- MAPT activity is inhibited.
- MAPT expression, translation, or activity is reduced by at least 10% relative to the expression, translation, or activity in an untreated or control sample.
- MAPT expression, translation, or activity is reduced by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%, 10-50%, 25- 50%, 25-75%, 50-75%, 50-99%, or 75-99% relative to the expression, translation, or activity in an untreated or control sample.
- MAPT expression, translation, or activity is reduced as measured by any suitable assay, including but not limited to, an immunoassay, a hybridization-based assay, or a sequencing-based assay (e.g., RNA-Seq).
- the disclosure relates to compounds targeted to a MAPT nucleic acid.
- the MAPT nucleic acid has the sequence set forth in GenBank Accession No. NM_001377265.1 (incorporated herein as SEQ ID NO: 1), and nucleotides 2624000 to 2761000 of NT_010783.14 (incorporated herein as SEQ ID NO: 2).
- the compound is an oligomeric compound.
- the compound is single-stranded. In certain embodiments, the compound is double-stranded. Certain embodiments provide a compound comprising a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209- .
- a modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any
- Certain embodiments provide a compound comprising a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprising a modified oligonucleotide having a nucleobase sequence selected from the group consisting of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- the modified oligonucleotide is at least 80%, at least 85%, at least 90%, or at least 95% complementary to SEQ ID NO: 1 or 2.
- the modified oligonucleotide comprises at least one modification selected from a modified internucleoside linkage, a modified sugar, and a modified nucleobase.
- the compound is double-stranded.
- Certain embodiments provide a compound comprising a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201- 206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a region of complementarity to the first modified oligonucleotide.
- the compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence provided in Tables 2 and 3, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a region of complementarity to the first modified oligonucleotide.
- a first modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a second modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- Certain embodiments provide a compound comprising a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11- 73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a region of complementarity to the first modified oligonucleotide.
- a first modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- Certain embodiments provide a compound comprising a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 and SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide 19 to 23 linked nucleosides in length having a region of complementarity to the first modified oligonucleotide.
- Certain embodiments provide a compound comprising a first modified oligonucleotide comprising a 5′-phosphonate modification, where the first modified oligonucleotide is at least 80% complementary to a region of SEQ ID NO: 1 or 2, and a second modified oligonucleotide comprising one or more ligands described herein (e.g., one or more Tropomyosin receptor B (TrkB) ligands, one or more cannabinoid receptor type 1 (CB 1 ) ligands, or one or more ⁇ 4 ⁇ 1/7 integrin ligands).
- ligands described herein e.g., one or more Tropomyosin receptor B (TrkB) ligands, one or more cannabinoid receptor type 1 (CB 1 ) ligands, or one or more ⁇ 4 ⁇ 1/7 integrin ligands.
- the first modified oligonucleotide comprises a 5′-terminal nucleoside comprising the 5′-phosphonate modification.
- the 5′-phosphonate modification is a 5′- vinylphosphonate modification or a 5′-ethylenephosphonate modification.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequences of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- the compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11- 73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 and a second modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- the modified oligonucleotide or first modified oligonucleotide of any preceding compound has at least 80%, at least 85%, at least 90%, or at least 95% complementarity or identity to SEQ ID NO: 1 or 2 over its length.
- the modified oligonucleotide or first modified oligonucleotide has at least 1, at least 2, at least 3 mismatches to a region of SEQ ID NO: 1 or 2.
- the region of complementarity between the first modified oligonucleotide or first strand and the second modified oligonucleotide or second strand is 14 to 30 linked nucleosides in length.
- the region of complementarity between the first modified oligonucleotide or first strand and the second modified oligonucleotide or second strand is 14 to 23 linked nucleosides in length. In certain embodiments, the region of complementarity between the first modified oligonucleotide or first strand and the second modified oligonucleotide or second strand is 19 to 23 linked nucleosides in length. In certain embodiments, the region of complementarity between the first modified oligonucleotide or first strand and the second modified oligonucleotide or second strand is 21 to 23 linked nucleosides in length.
- the first modified oligonucleotide is fully complementary to the second modified oligonucleotide.
- the modified oligonucleotide or first modified oligonucleotide of any preceding compound comprises at least one modification selected from a modified internucleoside linkage, a modified sugar, and a modified nucleobase.
- the second modified oligonucleotide of any preceding compound comprises at least one modification selected from the group consisting of a modified internucleoside linkage, a modified sugar, and a modified nucleobase.
- the modified internucleoside linkage is a phosphorothioate internucleoside linkage or a methylphosphonate internucleoside linkage. In certain embodiments, the phosphorothioate internucleoside linkage or methylphosphonate internucleoside linkage is at the 3’ terminus of the first or second modified oligonucleotide or at the 5’ terminus of the first modified oligonucleotide.
- the modified sugar comprises a modification selected from the group consisting of a halogen, an alkoxy group and a bicyclic sugar. In certain embodiments, the modified sugar comprises a 2’-F modification.
- the modified sugar comprises a 2’-OMe modification.
- each nucleoside of the first modified oligonucleotide comprises a modified sugar.
- each nucleoside of the second modified oligonucleotide comprises a modified sugar.
- the modified sugar comprises a modification selected from the group consisting of a halogen, an alkoxy group and a bicyclic sugar or a combination thereof.
- the modified sugar comprises a modification selected from the group consisting of 2’-MOE, 2’-F, and 2’-OMe or a combination thereof.
- the first modified oligonucleotide comprises no more than ten 2’-F sugar modifications.
- the second modified oligonucleotide comprises no more than five 2’-F sugar modifications.
- the compound of any preceding embodiment comprises a conjugate group.
- the conjugate group is attached to the 5’ end of the modified oligonucleotide.
- the conjugate group is a targeting moiety.
- the targeting moiety comprises one or more ligands.
- the targeting moiety comprises one or more ligands selected from one or more Tropomyosin receptor B (TrkB) ligands, one or more cannabinoid receptor type 1 (CB 1 ) ligands, and one or more ⁇ 4 ⁇ 1/7 integrin ligands.
- the targeting moiety comprises one or more TrkB ligands. In certain embodiments, the targeting moiety comprises one or more CB 1 ligands. In certain embodiments, the targeting moiety comprises one or more ⁇ 4 ⁇ 1/7 integrin ligands.
- the modified oligonucleotide is the second modified oligonucleotide or sense oligonucleotide. In certain embodiments, the one or more TrkB ligands is attached at the 5’ or 3’ end of the oligonucleotide or both the 5’ and 3’ ends of the oligonucleotide.
- the one or more CB 1 ligands is attached at the 5’ or 3’ end of the oligonucleotide or both the 5’ and 3’ ends of the oligonucleotide. In certain embodiments, the one or more ⁇ 4 ⁇ 1/7 integrin ligands is attached at the 5’ or 3’ end of the oligonucleotide or both the 5’ and 3’ ends of the oligonucleotide.
- R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , R 15 , R 16 , R 17 , R 18 , R 19 , R 20 , R 21 , and R 22 are each independently optionally substituted unsaturated or partially unsaturated alkyl.
- R 7 , R 8 , R 9 , and R 10 are each independently alkenyl.
- R 7 , R 8 , R 9 , and R 10 are each independently alkynyl.
- R 2 is OR 7 .
- R 3 is OR 11 .
- R 7 and R 11 are each independently hydrogen, optionally substituted alkyl or optionally substituted alkenyl. In certain embodiments, one or both R 7 and R 11 are each independently hydrogen. In certain embodiments, one or both R 7 and R 11 are each independently optionally substituted alkyl. In certain embodiments, one or both R 7 and R 11 are each independently optionally substituted unsaturated or partially unsaturated alkyl. In certain embodiments, one or both R 7 and R 11 are each independently alkenyl. In certain embodiments, R 7 is optionally substituted alkyl and R 11 is hydrogen. In certain embodiments, R 7 is hydrogen and R 11 is optionally substituted alkyl. In certain embodiments, R 7 is alkenyl and R 11 is hydrogen.
- R 7 is hydrogen and R 11 is optionally substituted alkenyl.
- the TrkB ligand of a modified oligonucleotide is selected from the following Formulae or a salt, solvate, or hydrate thereof: Formula (II-C), wherein: R 1 is the modified oligonucleotide; L 1 , L 2 , L 3 , L 4 , and R 1 are as described herein.
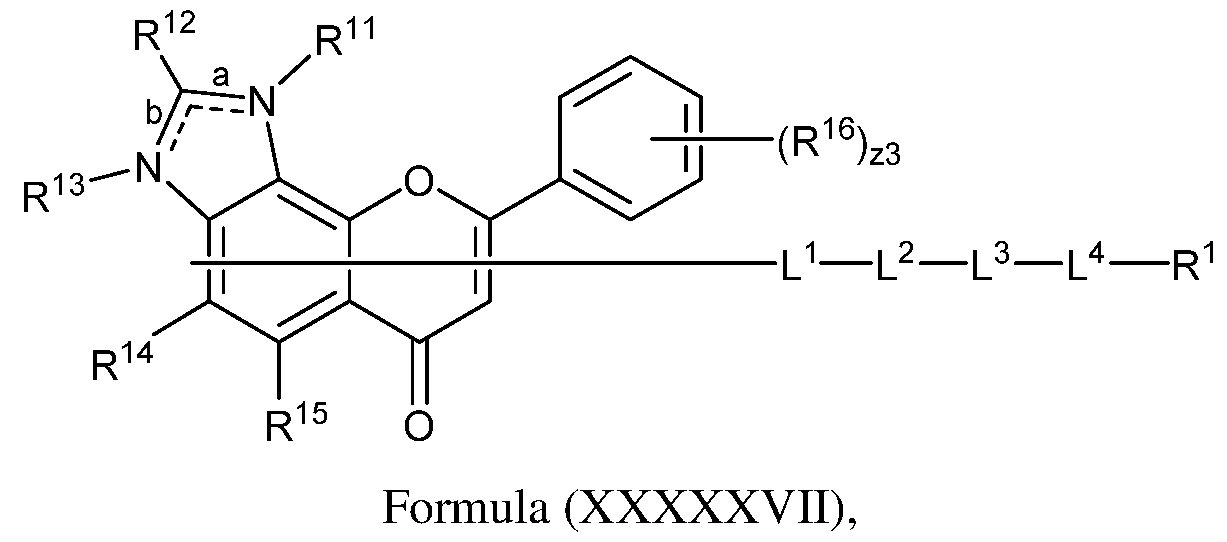
- the TrkB ligand of a modified oligonucleotide is of the Formula (XXXXXVII) or a salt, solvate, or hydrate thereof: Formula (XXXXVII), wherein: L 1 , L 2 , L 3 , L 4 , and R 1 are as described herein; R 11 and R 13 are each independently absent, hydrogen, or optionally substituted alkyl; R 12 , R 14 , and R 15 are each independently hydrogen, optionally substituted alkyl, optionally substituted heteroalkyl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl, optionally substituted aryl, or optionally substituted heteroaryl; R 16 is hydrogen, halogen, –CN, –N3, –SOn16R 1A , –SOv16NR 16B R 16C , ⁇ NHNR 16B R 16C , ⁇ ONR 16B R 16C , ⁇ NHC(O)NHNR 16B
- the TrkB ligand of a modified oligonucleotide is of the Formula (XXXXIX) or a salt, solvate, or hydrate thereof: Formula (XXXXIX), wherein: L 1 , L 2 , L 3 , L 4 , and R 1 are as described herein; R 17 , R 18 , and R 19 are each independently hydrogen, optionally substituted alkyl, optionally substituted heteroalkyl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl, optionally substituted aryl, or optionally substituted heteroaryl; z4 is 0, 1, or 2; and z5 is 0, 1, 2, or 3.
- the TrkB ligand of a modified oligonucleotide is of the Formula (XXXXX) or a salt, solvate, or hydrate thereof: Formula (XXXXX), wherein: L 1 , L 2 , L 3 , L 4 , and R 1 are as described herein; R 20 is hydrogen, halogen, –CN, –N 3 , –SO n20 R 1A , –SO v20 NR 20B R 20C , ⁇ NHNR 20B R 20C , ⁇ ONR 20B R 20C , ⁇ NHC(O)NHNR 20B R 20C , ⁇ NHC(O)NR 20B R 20C , –N(O)m20, –NR 20B R 20C , –C(O)R 20D , –C(O)OR 20D , –C(O)NR 20B R 20C , –OR 20A , -NR 20B SO2R
- the CB 1 ligand of a modified oligonucleotide is of the Formula (XXXXXI) or a salt, solvate, or hydrate thereof: , Formula (XXXXXI) wherein: L 1 , L 2 , L 3 , L 4 , and R 1 are as described herein;
- X 1 is NR 10 or CR 11 R 12 ;
- R 10 , R 11 , and R 12 are each independently hydrogen, optionally substituted alkyl, optionally substituted heteroalkyl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl, optionally substituted aryl, or optionally substituted heteroaryl;
- R 19 is hydrogen, –SOn19R 19A , –SOv19NR 19B R 19C , ⁇ NHNR 19B R 19C , ⁇ ONR 19B R 19C , ⁇ NHC(O)NHNR 19B R 19C , ⁇ NHC(O)NR 19B R
- the ⁇ 4 ⁇ 1/7 integrin ligand of a modified oligonucleotide is of the Formula (XXXXXXII) or a salt, solvate, or hydrate thereof: .
- Formula (XXXXXII) wherein L 1 , L 2 , L 3 , L 4 , and R 1 are as described herein; R 2 is H, polyethylene glycol (PEG), optionally substituted heteroalkyl, or optionally substituted heteroaryl; and R 3 , and R 4 are each independently H, halogen, optionally substituted alkyl, or optionally substituted -O-alkyl.
- the ⁇ 4 ⁇ 1/7 integrin ligand of a modified oligonucleotide is of the Formula (XXXXXXIII) or a salt, solvate, or hydrate thereof: .
- Formula (XXXXXIII) wherein L 1 , L 2 , L 3 , L 4 , and R 1 are as described herein; R 2 , R 3 , R 4 , and R 5 are each independently H, halogen, optionally substituted alkyl, optionally substituted -O-alkyl, cycloalkyl, or absent; R 8 is optionally substituted C 1 -C 5 alkyl, optionally substituted C 1 -C 5 alkylene-(C 3 - C 6 )-cycloalkyl, or optionally substituted (C 1 -C 4 )-alkylene-(C 1 -C 4 )-alkoxy; and R 6 , and R 7 are each independently H, halogen, alkyl, or optionally
- R 2 is H, -CONHR 4 , -CH 2 OR 4 , -(CH 2 ) 2 OR 4 , -CH 2 NHCOR 4 , or -OR 4 ;
- R 3 is H, optionally substituted alkyl, or optionally substituted cycloalkyl;
- R 4 is H, polyethylene glycol, optionally substituted alkyl, optionally substituted heteroalkyl, optionally substituted cycloalkyl, or optionally substituted heteroaryl;
- R 5 is -OH or absent;
- X is H, optionally substituted CH 2 , optionally substituted NH, or cycloalkyl.
- the ⁇ 4 ⁇ 1/7 integrin ligand of a modified oligonucleotide is of the Formula (XXXXXXXIII) or a salt, solvate, or hydrate thereof: .
- Formula (XXXXXXXIII) wherein L 1 , L 2 , L 3 , L 4 , and R 1 are as described herein; R 2 is H, -CONHR 3 , -CH 2 OR 3 , -(CH 2 ) 2 OR 3 , -CH 2 NHCOR 3 , or -OR 3 ; each instance of R 3 is independently H, polyethylene glycol, optionally substituted alkyl, optionally substituted heteroalkyl, optionally substituted cycloalkyl, or optionally substituted heteroaryl; and X is H or halogen.
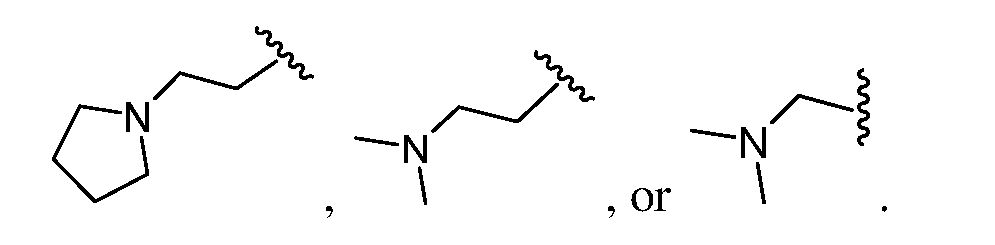
- L 1 , L 2 , L 3 , and L 4 are each independently absent, a bond, an optionally substituted alkyl linker, an optionally substituted polyethylene glycol (PEG) linker, an optionally substituted heteroalkyl linker, or an optionally substituted heteroaryl linker.
- L 1 is an optionally substituted heteroaryl linker.
- L 1 is an optionally substituted unsaturated heteroaryl, an optionally substituted heteroaryl or an optionally substituted saturated or partially unsaturated heterocycloalkyl linker.
- L 1 comprises the structure: .
- L 1 is an optionally substituted heteroalkyl linker.
- the optionally substituted heteroalkyl linker is an optionally substituted heteroalkyl or optionally substituted C 1 -C 10 alkyl chain in which one or more carbon atoms are replaced with O, N, or S.
- L 1 comprises the structure: or .
- L 1 comprises the structure: or –N(CH 3 )–.
- L 2 is an optionally substituted PEG linker.
- the PEG linker is five PEG units in length.
- the PEG linker is four PEG units in length.
- the PEG linker is three PEG units in length.
- L 2 is an optionally substituted alkyl linker.
- the heteroalkyl linker comprises two substituents joined together to form an optionally substituted carbocyclyl ring.
- L 4 comprises the structure: or a salt thereof, wherein X is O or S.
- L 4 comprises the structure: or a salt thereof, wherein X is O or S.
- L 1 – L 2 –L 3 –L 4 comprises the structure: ,
- TrkB ligand of a modified oligonucleotide is selected from the following Formulae or a salt, solvate, or hydrate thereof:
- R is the modified oligonucleotide; and X is S or O.
- the CB 1 ligand of a modified oligonucleotide is selected from the following Formulae or a salt, solvate, or hydrate thereof:
- Formula (XXXXXXXVII) Formula (XXXXXXXVIII) wherein: R is the modified oligonucleotide; and X is S or O.
- the ⁇ 4 ⁇ 1/7 integrin ligand of a modified oligonucleotide is selected from the following Formulae or a salt, solvate, or hydrate thereof: Formula (XXXXXXX)
- R is the modified oligonucleotide; and X is S or O.
- the compound of any preceding embodiment comprises a lipid.
- the lipid is attached to an internucleoside linkage of the modified oligonucleotide.
- the modified oligonucleotide comprises one or more lipids.
- the one or more lipids are attached to one or more internucleoside linkages of the modified oligonucleotide.
- the modified oligonucleotide is the second modified oligonucleotide or sense oligonucleotide.
- the compound of any preceding embodiment comprises one or more substituted or unsubstituted alkyl or alkenyl.
- the substituted or unsubstituted alkyl or alkenyl is attached to an internucleoside linkage of a modified oligonucleotide.
- the modified oligonucleotide comprises one or more substituted or unsubstituted alkyl or alkenyl.
- the one or more substituted or unsubstituted alkyl or alkenyl are attached to one or more internucleoside linkages of the modified oligonucleotide.
- the modified oligonucleotide is the second modified oligonucleotide or sense oligonucleotide.
- the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 4 -C 30 hydrocarbon chain.
- the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 5 -C 20 hydrocarbon chain.
- the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 14 -C 20 hydrocarbon chain.
- the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 16 hydrocarbon chain. In certain embodiments, the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 17 hydrocarbon chain. In certain embodiments, the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 18 hydrocarbon chain. In certain embodiments, the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 22 hydrocarbon chain.
- a substituted or unsubstituted alkyl or alkenyl is attached to an internucleoside linkage of a modified oligonucleotide (e.g., a second modified oligonucleotide or sense oligonucleotide).
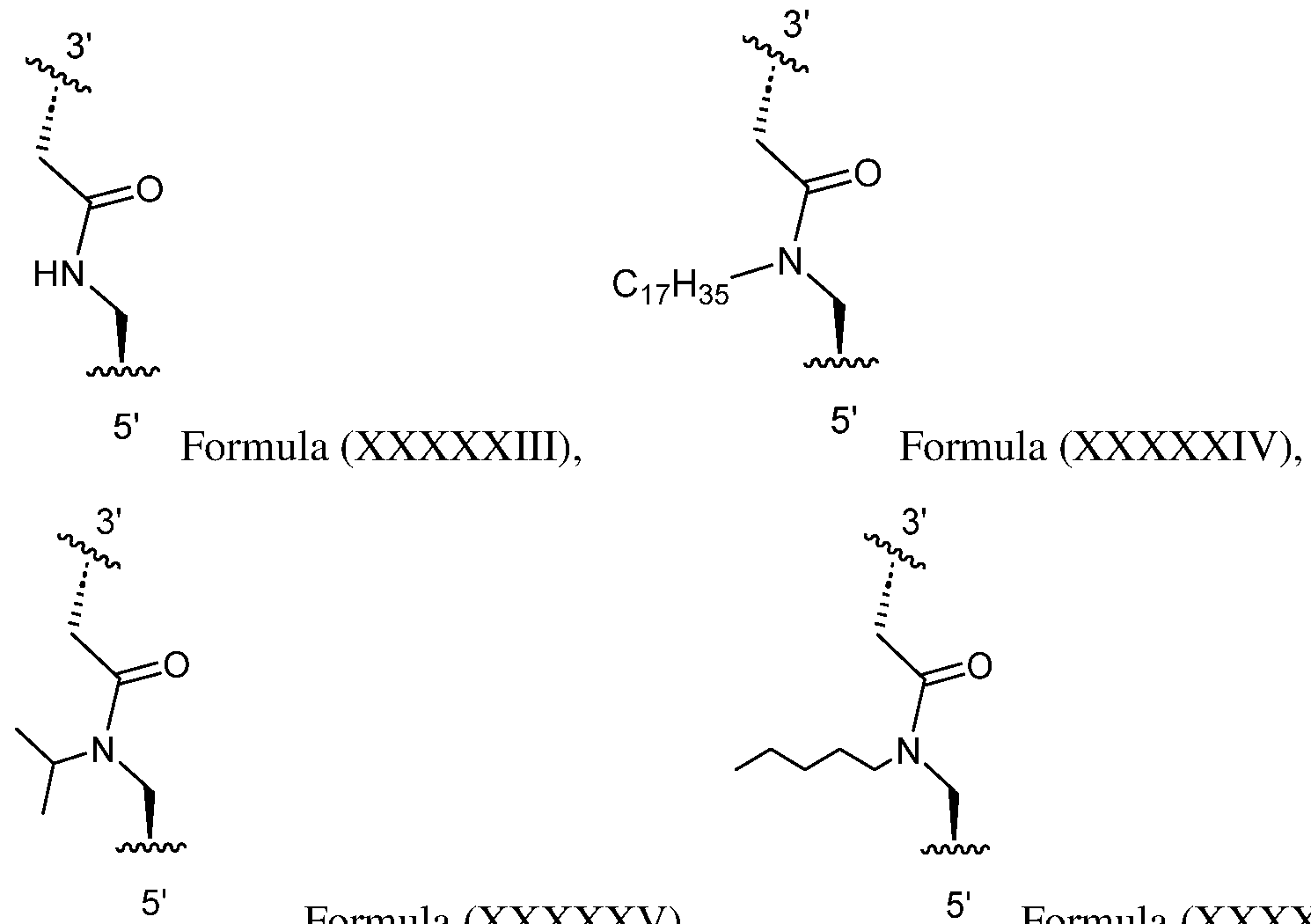
- the internucleoside linkage is between nucleosides that are within 10 positions (e.g., within 8 positions, within 6 positions, within 5 positions, within 4 positions, within 3 positions, within 2 positions) from a terminal end (e.g., the 5′ and/or 3′ end) of the modified oligonucleotide.
- the internucleoside linkage is between nucleosides that are within 5 positions from the 5′ end of the modified oligonucleotide. In certain embodiments, the internucleoside linkage is between nucleosides that are within 5 positions from the 3′ end of the modified oligonucleotide. In certain embodiments, the internucleoside linkage is between positions 1 and 2, positions 2 and 3, positions 3 and 4, positions 4 and 5, positions 5 and 6, positions 6 and 7, positions 7 and 8, positions 8 and 9, positions 9 and 10, positions 10 and 11, positions 11 and 12, positions 12 and 13, or positions 13 and 14 from the 5′ end of the modified oligonucleotide.
- the internucleoside linkage is between positions 1 and 2, positions 2 and 3, positions 3 and 4, positions 4 and 5, positions 5 and 6, positions 6 and 7, or positions 7 and 8 from the 5′ end of the modified oligonucleotide. In certain embodiments, the internucleoside linkage is between positions 2 and 3 from the 5′ end of the modified oligonucleotide. In certain embodiments, the internucleoside linkage is between positions 1 and 2, positions 2 and 3, positions 3 and 4, positions 4 and 5, positions 5 and 6, positions 6 and 7, positions 7 and 8, positions 8 and 9, positions 9 and 10, positions 10 and 11, positions 11 and 12, positions 12 and 13, or positions 13 and 14 from the 3′ end of the modified oligonucleotide.
- the internucleoside linkage is between positions 1 and 2, positions 2 and 3, positions 3 and 4, positions 4 and 5, positions 5 and 6, positions 6 and 7, or positions 7 and 8 from the 3′ end of the modified oligonucleotide. In certain embodiments, the internucleoside linkage is between positions 2 and 3 from the 3′ end of the modified oligonucleotide. In certain embodiments, the internucleoside linkage of the modified oligonucleotide is selected from any one of Formulae XXXXIII-XXXXXVI. In certain embodiments, the modified oligonucleotide comprises any one of Formulae XXXV-XXXXVI.
- the modified oligonucleotide comprises Formula (XXXVI), or a salt, solvate, or hydrate thereof: Formula (XXXVI), wherein: R C is –H, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted heteroalkyl, substituted or unsubstituted aryl, or substituted or unsubstituted heteroaryl; each R 2 is independently –H, halogen, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, –OR 6 , -N(R 6 ), or -SR 6 ; each R 3 is independently –H, halogen, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubsti
- the modified oligonucleotide comprises Formula (XXXVII), or a salt, solvate, or hydrate thereof: Formula (XXXVII), wherein: each R 2 is independently –H, halogen, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, –OR 6 , -N(R 6 ), or -SR 6 ; each R 3 is independently –H, halogen, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, –OR 7 , -N(R 7 ), or -SR 7 ; R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; each R 6 is independently hydrogen, substituted or unsubstit
- the modified oligonucleotide comprises Formula (XXXVIII), or a salt, solvate, or hydrate thereof: Formula (XXXVIII), wherein: each R 2 is independently –H, halogen, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, –OR 6 , -N(R 6 ), or -SR 6 ; each R 3 is independently –H, halogen, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, –OR 7 , -N(R 7 ), or -SR 7 ; R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; each R 6 is independently hydrogen, substituted or unsubstit
- the modified oligonucleotide comprises Formula (XXXXII), or a salt, solvate, or hydrate thereof: mA ⁇ mA Formula (XXXXII) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXIII), or a salt, solvate, or hydrate thereof: mA ⁇ mU Formula (XXXXIII) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXIV), or a salt, solvate, or hydrate thereof: mA ⁇ mG Formula (XXXXIV) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXV), or a salt, solvate, or hydrate thereof: mA ⁇ mC Formula (XXXXV) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXVI), or a salt, solvate, or hydrate thereof: mU ⁇ mA Formula (XXXXVI) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXVII), or a salt, solvate, or hydrate thereof: mU ⁇ mG Formula (XXXXVII) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXVIII), or a salt, solvate, or hydrate thereof: mU ⁇ fC Formula (XXXXVIII) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXIX), or a salt, solvate, or hydrate thereof: fG ⁇ mU Formula (XXXXIX) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXX), or a salt, solvate, or hydrate thereof: mG ⁇ fG Formula (XXXX) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXXI), or a salt, solvate, or hydrate thereof: mG ⁇ mC Formula (XXXXI) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXXII), or a salt, solvate, or hydrate thereof:
- the modified oligonucleotide comprises an internucleoside linkage of one of the following Formulae: Formula (XXXXXV), Formula (XXXXXVI).
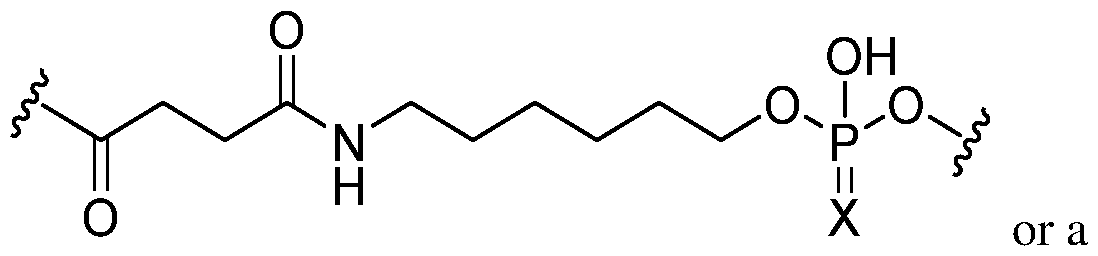
- the compound of any preceding embodiment comprises a 5′- phosphonate modification.
- a modified oligonucleotide comprises one or more sugars having a phosphonate modification at the 5′ position.
- the modified oligonucleotide comprises a 5′-phosphonate modification. In certain embodiments, the modified oligonucleotide comprises a 5′-terminal nucleoside (e.g., 5′ terminus) comprising the 5′-phosphonate modification. In certain embodiments, the 5′-phosphonate modification is a 5′-vinylphosphonate modification or a 5′- ethylenephosphonate modification. In certain embodiments, the 5′-phosphonate modification is a 5′-vinylphosphonate modification. In certain embodiments, the 5′-phosphonate modification is a 5′-ethylenephosphonate modification.
- the modified oligonucleotide is the first modified oligonucleotide or antisense oligonucleotide.
- Certain embodiments provide a compound comprising: a first modified oligonucleotide comprising a 5′-phosphonate modification, where the first modified oligonucleotide is at least 80% complementary to a region of SEQ ID NO: 1 or 2; and a second modified oligonucleotide comprising one or more ligands.
- the first modified oligonucleotide comprises a 5′-terminal nucleoside comprising the 5′-phosphonate modification.
- the 5′- phosphonate modification is a 5′-vinylphosphonate modification or a 5′-ethylenephosphonate modification. In some embodiments, the 5′-phosphonate modification is a 5′- vinylphosphonate modification. In some embodiments, the 5′-phosphonate modification is a 5′-ethylenephosphonate modification. In some embodiments, the second modified oligonucleotide comprises one or more ligands selected from one or more TrkB ligands, one or more CB 1 ligands, and one or more ⁇ 4 ⁇ 1/7 integrin ligands.
- the one or more TrkB ligands, the one or more CB 1 ligands, or the one or more ⁇ 4 ⁇ 1/7 integrin ligands are attached to the 5’ end of the second modified oligonucleotide. In some embodiments, the one or more TrkB ligands, the one or more CB 1 ligands, or the one or more ⁇ 4 ⁇ 1/7 integrin ligands are attached to the 3’ end of the second modified oligonucleotide.
- the one or more TrkB ligands, the one or more CB 1 ligands, or the one or more ⁇ 4 ⁇ 1/7 integrin ligands are attached to the 5’ end and the 3’ end of the second modified oligonucleotide.
- the one or more TrkB ligands are selected from any one of Formulae I-XXXIV, XXXXVII, XXXXIX-XXXXXX, XXXXXV-XXXXXVII, and XXXXXIX-XXXXXXIII;
- the one or more CB 1 ligands are selected from any one of Formulae XXXXXI and XXXXXIV-XXXXXXXVIII;
- the one or more ⁇ 4 ⁇ 1/7 integrin ligands are selected from any one of Formulae XXXXXII-XXXXXIV and XXXXXXIX-XXXXXXXIV.
- the second modified oligonucleotide comprises one or more TrkB ligands.
- the one or more TrkB ligands are selected from any one of Formulae I-XXXIV, XXXXVII, XXXXIX-XXXXXX, XXXXXV- XXXXXVII, and XXXXXIX-XXXXXXIII.
- the one or more TrkB ligands are selected from any one of Formulae XIX-XXXI and XXXXXV.
- the second modified oligonucleotide comprises one TrkB ligand.
- the second modified oligonucleotide comprises two TrkB ligands. In some embodiments, the second modified oligonucleotide comprises at least two TrkB ligands. In some embodiments, the at least two TrkB ligands are the same. In some embodiments, the at least two TrkB ligands are different. In some embodiments, the second modified oligonucleotide comprises one or more CB 1 ligands. In some embodiments, the one or more CB 1 ligands are selected from any one of Formulae XXXXXI and XXXXXIV-XXXXXXXVIII.
- the one or more CB 1 ligands are selected from any one of Formulae XXXXXXIV- XXXXXXVI.
- the second modified oligonucleotide comprises one CB 1 ligand. In some embodiments, the second modified oligonucleotide comprises two CB 1 ligands. In some embodiments, the second modified oligonucleotide comprises at least two CB 1 ligands. In some embodiments, the at least two CB 1 ligands are the same. In some embodiments, the at least two CB 1 ligands are different.
- the second modified oligonucleotide comprises one or more ⁇ 4 ⁇ 1/7 integrin ligands.
- the one or more ⁇ 4 ⁇ 1/7 integrin ligands are selected from any one of Formulae XXXXXII-XXXXXIV and XXXXXXIX- XXXXXXXIV.
- the one or more ⁇ 4 ⁇ 1/7 integrin ligands are selected from any one of Formulae XXXXXXI, XXXXXXXII, and XXXXXXIV.
- the second modified oligonucleotide comprises one ⁇ 4 ⁇ 1/7 integrin ligand. In some embodiments, the second modified oligonucleotide comprises two ⁇ 4 ⁇ 1/7 integrin ligands. In some embodiments, the second modified oligonucleotide comprises at least two ⁇ 4 ⁇ 1/7 integrin ligands. In some embodiments, the at least two ⁇ 4 ⁇ 1/7 integrin ligands are the same. In some embodiments, the at least two ⁇ 4 ⁇ 1/7 integrin ligands are different. In some embodiments, the second modified oligonucleotide comprises one or more lipids.
- the second modified oligonucleotide comprises one or more substituted or unsubstituted alkyl or alkenyl.
- the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 4 -C 30 hydrocarbon chain.
- the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 5 -C 20 hydrocarbon chain.
- the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 14 -C 20 hydrocarbon chain.
- the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C16 hydrocarbon chain, a saturated or unsaturated C 17 hydrocarbon chain, a saturated or unsaturated C 18 hydrocarbon chain, or a saturated or unsaturated C 22 hydrocarbon chain.
- the one or more substituted or unsubstituted alkyl or alkenyl are attached to an internucleoside linkage of the second modified oligonucleotide.
- the internucleoside linkage is between nucleosides that are within 10 positions (e.g., within 8 positions, within 6 positions, within 5 positions, within 4 positions, within 3 positions, within 2 positions) from a terminal end (e.g., the 5′ and/or 3′ end) of the second modified oligonucleotide. In certain embodiments, the internucleoside linkage is between nucleosides that are within 5 positions from the 5′ end of the second modified oligonucleotide. In certain embodiments, the internucleoside linkage is between nucleosides that are within 5 positions from the 3′ end of the second modified oligonucleotide.
- the internucleoside linkage is between positions 1 and 2, positions 2 and 3, positions 3 and 4, positions 4 and 5, positions 5 and 6, positions 6 and 7, positions 7 and 8, positions 8 and 9, positions 9 and 10, positions 10 and 11, positions 11 and 12, positions 12 and 13, or positions 13 and 14 from the 5′ end of the second modified oligonucleotide. In certain embodiments, the internucleoside linkage is between positions 1 and 2, positions 2 and 3, positions 3 and 4, positions 4 and 5, positions 5 and 6, positions 6 and 7, or positions 7 and 8 from the 5′ end of the second modified oligonucleotide. In certain embodiments, the internucleoside linkage is between positions 2 and 3 from the 5′ end of the second modified oligonucleotide.
- the internucleoside linkage is between positions 1 and 2, positions 2 and 3, positions 3 and 4, positions 4 and 5, positions 5 and 6, positions 6 and 7, positions 7 and 8, positions 8 and 9, positions 9 and 10, positions 10 and 11, positions 11 and 12, positions 12 and 13, or positions 13 and 14 from the 3′ end of the second modified oligonucleotide. In certain embodiments, the internucleoside linkage is between positions 1 and 2, positions 2 and 3, positions 3 and 4, positions 4 and 5, positions 5 and 6, positions 6 and 7, or positions 7 and 8 from the 3′ end of the second modified oligonucleotide. In certain embodiments, the internucleoside linkage is between positions 2 and 3 from the 3′ end of the second modified oligonucleotide.
- the internucleoside linkage of the second modified oligonucleotide is selected from any one of Formulae XXXXIII-XXXXVI.
- the second modified oligonucleotide comprises any one of Formulae XXXV- XXXXVI.
- the first modified oligonucleotide is 14 to 30 linked nucleosides in length.
- the second modified oligonucleotide is 14 to 30 linked nucleosides in length having a region of complementarity to the first modified oligonucleotide.
- the first modified oligonucleotide has a nucleobase sequence comprising at least 14 contiguous nucleobases of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200, and 209-217.
- the second modified oligonucleotide has a nucleobase sequence comprising at least 14 contiguous nucleobases of any one of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171- 172, 174, 176 and 201-206.
- the first modified oligonucleotide is selected from any one of the IA Ref ID NOs in Table 3.
- the second modified oligonucleotide is selected from any one of the IS Ref ID NOs in Table 3.
- Certain embodiments provide a compound comprising a first modified oligonucleotide selected from the group consisting of any one of Ref ID NOs listed in Table 3 and a second modified oligonucleotide 14 to 21 linked nucleosides in length fully complementary to the first modified oligonucleotide.
- Certain embodiments provide a compound comprising a first modified oligonucleotide selected from the group consisting of any one of the IA Ref ID NOs listed in Table 3 and a second modified oligonucleotide selected from the group consisting of any one of the IS Ref ID NOs listed in Table 3.
- the second modified oligonucleotide comprises any one or more TrkB ligands provided herein.
- the TrkB ligand is selected from Formulae I-XXXIV, XXXXVII, XXXXIX-XXXXXX, XXXXXV-XXXXXVII, and XXXXXIX-XXXXXXIII, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide.
- the TrkB ligand is selected from Formulae XIX-XXXI and XXXXXV, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide.
- the modified oligonucleotide is attached to the TrkB ligand through a phosphodiester group. In certain embodiments, the modified oligonucleotide is attached to the TrkB ligand through a phosphorothioate group. In certain embodiments, provided herein is a compound selected from the group consisting of any one of the compounds listed in Table 3. Certain embodiments provide a compound comprising a first modified oligonucleotide selected from the group consisting of any one of the IA Ref ID NOs listed in Table 3 and a second modified oligonucleotide selected from the group consisting of any one of the IS Ref ID NOs listed in Table 3.
- the second modified oligonucleotide comprises any one or more CB 1 ligands provided herein.
- the CB 1 ligand is selected from Formulae XXXXXI and XXXXXIV- XXXXXXVIII, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide.
- the CB 1 ligand is selected from Formulae XXXXXIV-XXXXXXXVI, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide.
- the modified oligonucleotide is attached to the CB 1 ligand through a phosphodiester group. In certain embodiments, the modified oligonucleotide is attached to the CB 1 ligand through a phosphorothioate group. In certain embodiments, provided herein is a compound selected from the group consisting of any one of the compounds listed in Table 3. Certain embodiments provide a compound comprising a first modified oligonucleotide selected from the group consisting of any one of the IA Ref ID NOs listed in Table 3 and a second modified oligonucleotide selected from the group consisting of any one of the IS Ref ID NOs listed in Table 3.
- the second modified oligonucleotide comprises any one or more ⁇ 4 ⁇ 1/7 integrin ligands provided herein.
- the ⁇ 4 ⁇ 1/7 integrin ligand is selected from Formulae XXXXXII-XXXXXIV and XXXXXXIX-XXXXXXXIV, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide.
- the ⁇ 4 ⁇ 1/7 integrin ligand is selected from Formulae XXXXXXXI, XXXXXXII, and XXXXXXXIV, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide.
- the modified oligonucleotide is attached to the ⁇ 4 ⁇ 1/7 integrin ligand through a phosphodiester group.
- the modified oligonucleotide is attached to the ⁇ 4 ⁇ 1/7 integrin ligand through a phosphorothioate group.
- provided herein is a compound selected from the group consisting of any one of the compounds listed in Table 3.
- the pharmaceutically acceptable salt of the modified oligonucleotides provided herein is a sodium salt or a potassium salt.
- the pharmaceutically acceptable salt of the compounds provided herein is a sodium salt or a potassium salt.
- provided herein is a population of modified oligonucleotides, wherein all of the phosphorothioate internucleoside linkages of the modified oligonucleotide are stereorandom.
- provided herein is a population of compounds, wherein all of the phosphorothioate internucleoside linkages of the modified oligonucleotide are stereorandom.
- the compound of any foregoing embodiment is in a pharmaceutically acceptable salt form.
- the pharmaceutically acceptable salt is a sodium salt.
- the pharmaceutically acceptable salt is a potassium salt.
- Certain embodiments provide a composition comprising the compound of any one of the foregoing embodiments and a pharmaceutically acceptable carrier.
- Certain embodiments provide a composition comprising a compound of any preceding embodiment, for use in therapy.
- Certain embodiments provide a method of treating, preventing, or ameliorating a disease, disorder or condition associated with MAPT in an individual comprising administering to the individual a compound targeted to MAPT, thereby treating, preventing, or ameliorating the disease.
- the compound or composition of any foregoing embodiment is administered to an individual.
- the disease, disorder or condition is a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- administering the compound inhibits or reduces or improves a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- a compound or composition comprising a compound of any preceding embodiment is administered to an individual in a therapeutically effective amount.
- a compound or composition comprising a compound of any preceding embodiment is administered to an individual at a dosage level sufficient to deliver about 1 to 100 mg/kg of body weight of the individual. In certain embodiments, a compound or composition comprising a compound of any preceding embodiment is administered to an individual at a fixed dose of about 25 mg to about 1,000 mg. In certain embodiments, the compound or composition is administered to the individual one or more times in a day up to the dosage level or fixed dose. In certain embodiments, a compound or composition comprising a compound of any preceding embodiment is administered to an individual daily, weekly, monthly, quarterly or yearly.
- a compound or composition comprising a compound of any preceding embodiment is administered to an individual about once per quarter (i.e., once every three months) to about once per year. In certain embodiments, a compound or composition comprising a compound of any preceding embodiment is administered to an individual about once per quarter, about once every six months or about once per year. Certain embodiments provide a method of inhibiting expression of MAPT in a cell comprising contacting the cell with a compound targeted to MAPT, thereby inhibiting expression of MAPT in the cell. In certain embodiments, the cell is in the liver of an individual.
- the individual has, or is at risk of having, a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- Certain embodiments provide a method of reducing or inhibiting a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment in an individual, comprising administering a compound targeted to MAPT to the individual, thereby reducing or inhibiting a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive
- the individual has, or is at risk of having, a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- the compound is a compound targeted to MAPT.
- the compound is any of the foregoing compounds.
- the compound or composition is administered parenterally.
- the compound or composition is administered by intrathecal (IT) administration.
- the disease, disorder or condition is a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- the compound is a compound targeted to MAPT.
- the compound is any of the foregoing compounds.
- Certain embodiments provide use of a compound targeted to MAPT in the manufacture of a medicament for treating, preventing, or ameliorating a disease, disorder or condition associated with MAPT.
- the disease, disorder or condition is a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairmentvv.
- the compound is a compound targeted to MAPT.
- the compound is any of the foregoing compounds. In certain embodiments, any one of the compounds shown in FIGs. 1-4, Table A, Table 2, and Table 3 can be used in any aspect of the disclosure. In some embodiments of a compound, composition, method, or use of the disclosure, the compound is any one of the compounds shown in FIGs. 1-4, Table A, Table 2, and Table 3. In certain embodiments, any one of the compounds shown in FIGs. 1-4 can be used in any aspect of the disclosure. In some embodiments of a compound, composition, method, or use of the disclosure, the compound is any one of the compounds shown in FIGs. 1-4. In certain embodiments, any one of the compounds in Table 2 can be used in any aspect of the disclosure.
- the compound is any one of the compounds in Table 2. In certain embodiments, any one of the compounds in Table 3 can be used in any aspect of the disclosure. In some embodiments of a compound, composition, method, or use of the disclosure, the compound is any one of the compounds in Table 3. In certain embodiments, any one of the compounds in Table A can be used in any aspect of the disclosure. In some embodiments of a compound, composition, method, or use of the disclosure, the compound is any one of the compounds in Table A. In some embodiments of any of the aspects of the disclosure, the compound is selected from any one of compounds 1-285 in Table A.
- Table A shown below, provides the following for each of compounds 1-285: antisense (“A”) and sense (“S”) strand nucleobase sequences (SEQ ID NO) shown with strand modifications (as defined in Table 1), and, where a sense strand comprises a ligand, the type of ligand (e.g., a TrkB ligand, a CB 1 ligand, or an ⁇ 4 ⁇ 1/7 integrin ligand as described herein) at the 5’ end (“5’-Ligand”) and/or the 3’ end (“3’-Ligand”) of the sense strand.
- the 5’-Ligand and/or the 3’-Ligand are attached to the sense strand via a linker.
- the linker comprises polyethylene glycol (PEG). In some embodiments, the linker comprises a PEG chain of one, two, three, four, five, six, seven, or eight PEG units in length. In certain embodiments, the linker comprises a PEG chain one PEG unit in length. In certain embodiments, the linker comprises a PEG chain two PEG units in length. In certain embodiments, the linker comprises a PEG chain three PEG units in length. In certain embodiments, the linker comprises a PEG chain four PEG units in length. In certain embodiments, the linker comprises a PEG chain five PEG units in length. In certain embodiments, the linker comprises a PEG chain six PEG units in length.
- PEG polyethylene glycol
- the linker comprises a PEG chain seven PEG units in length. In certain embodiments, the linker comprises a PEG chain eight PEG units in length. In some embodiments, the linker comprises an optionally substituted heteroaryl or optionally substituted heterocyclyl group. In certain embodiments, the linker comprises PEG and an optionally substituted heteroaryl or optionally substituted heterocyclyl group. Table A. Exemplary Compounds
- the disclosure relates to methods of inhibiting MAPT expression, which can be useful for treating, preventing, or ameliorating a disease associated with MAPT in an individual, by administration of a compound that targets MAPT.
- the compound can be a MAPT specific inhibitor.
- the compound can be an antisense oligonucleotide, an oligomeric compound, or an oligonucleotide targeted to MAPT (e.g., a compound of any one of the compounds shown in FIGs. 1-4, Table A, Table 2, and Table 3).
- the disclosure relates to treating, preventing, or ameliorating a disease, disorder or condition associated with MAPT.
- diseases, disorders or conditions associated with MAPT treatable, preventable, and/or ameliorable with the methods provided herein include a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- Certain compounds provided herein are directed to compounds and compositions that reduce a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP- 17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment in an animal.
- a method of treating, preventing, or ameliorating a disease associated with MAPT in an individual comprises administering to the individual a compound comprising a MAPT specific inhibitor, thereby treating, preventing, or ameliorating the disease.
- the individual is identified as having, or at risk of having, a disease associated with MAPT.
- the disease is a CNS related disease.
- the compound comprises an antisense oligonucleotide targeted to MAPT. In certain embodiments, the compound comprises an oligonucleotide targeted to MAPT.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides) in length having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides
- a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) and having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequences of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209- 217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- the compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 and a second modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 81- 143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- the compound can be single-stranded or double-stranded.
- a single-stranded compound can be 14 to 30, 14 to 23, 14 to 20, 16 to 20, or 14 to 16, linked nucleosides in length. In certain embodiments, a single-stranded compound can be 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, or 30, linked nucleosides in length. In certain embodiments, a double-stranded compound can comprise two oligonucleotides of the same or different lengths, as described elsewhere herein. In any of the foregoing embodiments, the compound can be an antisense oligonucleotide or oligomeric compound.
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a region of complementarity to the first modified oligonucleotide.
- a first modified oligonucleotide e.
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194- 197, 199-200 and 209-217 or SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a region of complementarity to the first modified oligonucleotide.
- a first modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a second modified oligonucleotide e
- a compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 and SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide 19 to 23 linked nucleosides in length having a region of complementarity to the first modified oligonucleotide.
- the compound is administered to the individual parenterally.
- the compound is administered to the individual by intrathecal (IT) administration.
- administering the compound improves, preserves, or prevents a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment in an animal.
- a method of treating, preventing, or ameliorating a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment in an animal comprises administering to the individual a compound comprising a MAPT specific inhibitor, thereby treating, preventing, or ameliorating a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dr
- the compound comprises an antisense oligonucleotide targeted to MAPT. In certain embodiments, the compound comprises an oligonucleotide targeted to MAPT. In certain embodiments, a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) and having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequences of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159- 168, 171-172, 174, 176 and 201-206.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171- 172, 174, 176 and 201-206.
- the compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199- 200 and 209-217 and a second modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 81-143, 149- 155, 157, 159-168, 171-172, 174, 176 and 201-206.
- the compound can be single-stranded or double-stranded.
- the compound can be an antisense oligonucleotide or oligomeric compound.
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or SEQ ID NOs: 81- 143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length)
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201- 206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a region of complementarity to the first modified oligonucleotide.
- a first modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a second modified oligonucleotide
- a compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 and ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide 19 to 23 linked nucleosides in length having a region of complementarity to the first modified oligonucleotide.
- administering the compound improves, preserves, or prevents a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment in an animal.
- the individual is identified as having, or at risk of having, a disease associated with MAPT.
- a method of inhibiting expression of MAPT in an individual having, or at risk of having, a disease associated with MAPT comprises administering to the individual a compound comprising a MAPT specific inhibitor, thereby inhibiting expression of MAPT in the individual. In certain embodiments, administering the compound inhibits expression of MAPT in the liver. In certain embodiments, the disease is a CNS related disease.
- the individual has, or is at risk of having, a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- the compound comprises an antisense oligonucleotide targeted to MAPT.
- the compound comprises an oligonucleotide targeted to MAPT.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188,
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) and having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequences of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159- 168, 171-172, 174, 176 and 201-206.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171- 172, 174, 176 and 201-206.
- the compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199- 200 and 209-217 and a second modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 81-143, 149- 155, 157, 159-168, 171-172, 174, 176 and 201-206.
- the compound can be single-stranded or double-stranded.
- the compound can be an antisense oligonucleotide or oligomeric compound.
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or SEQ ID NOs: 81- 143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length)
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201- 206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a region of complementarity to the first modified oligonucleotide.
- a first modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a second modified oligonucleotide
- a compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 and SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide 19 to 23 linked nucleosides in length having a region of complementarity to the first modified oligonucleotide.
- the compound is administered to the individual parenterally.
- the compound is administered to the individual by intrathecal (IT) administration.
- administering the compound improves, preserves, or prevents a CNS related disease, disorder or condition or a symptom thereof, a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- IT intrathecal
- administering the compound improves, preserves, or prevents a CNS related disease, disorder or condition or a symptom thereof, a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD
- a method of inhibiting expression of MAPT in a cell comprises contacting the cell with a compound comprising a MAPT specific inhibitor, thereby inhibiting expression of MAPT in the cell.
- the cell is a hepatocyte. In certain embodiments, the cell is in the liver.
- the cell is in the liver of an individual who has, or is at risk of having, a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- the compound comprises an antisense oligonucleotide targeted to MAPT.
- the compound comprises an oligonucleotide targeted to MAPT.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188,
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) and having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequences of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- the compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11- 73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 and a second modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- the compound can be single-stranded or double-stranded.
- the compound can be an antisense oligonucleotide or oligomeric compound.
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201- 206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a region of complementarity to the first modified oligonucleotide.
- a first modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 and sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide 19 to 23 linked nucleosides in length having a region of complementarity to the first modified oligonucleotide.
- the individual has, or is at risk of having, a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- the compound comprises an antisense oligonucleotide targeted to MAPT.
- the compound comprises an oligonucleotide targeted to MAPT.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188,
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) and having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequences of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- the compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11- 73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 and a second modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- the compound can be single-stranded or double-stranded.
- the compound can be an antisense oligonucleotide or oligomeric compound.
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a region of complementarity to the first modified oligonucleotide.
- a first modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a second modified oligonucleotide e.
- a compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 and SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide 19 to 23 linked nucleosides in length having a region of complementarity to the first modified oligonucleotide.
- the compound is administered to the individual parenterally.
- the compound is administered to the individual by intrathecal (IT) administration.
- the individual is identified as having, or at risk of having, a disease associated with MAPT.
- Certain embodiments are drawn to a compound comprising a MAPT specific inhibitor for use in treating a disease, disorder or condition associated with MAPT.
- the disease, disorder or condition is a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- the compound comprises an antisense oligonucleotide targeted to MAPT. In certain embodiments, the compound comprises an oligonucleotide targeted to MAPT.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188,
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) and having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequences of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- the compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11- 73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 and a second modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- the compound can be single-stranded or double-stranded.
- the compound can be an antisense oligonucleotide or oligomeric compound.
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a region of complementarity to the first modified oligonucleotide.
- a first modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a second modified oligonucleotide e.
- a compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 and SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide 19 to 23 linked nucleosides in length having a region of complementarity to the first modified oligonucleotide.
- the compound is administered to the individual parenterally.
- the compound is administered to the individual by intrathecal (IT) administration.
- IT intrathecal
- Certain embodiments are drawn to a compound comprising a MAPT specific inhibitor for use in reducing or inhibiting a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- the compound comprises an antisense oligonucleotide targeted to MAPT.
- the compound comprises an oligonucleotide targeted to MAPT.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) and having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a region of complementarity to the first modified oligonucleotide.
- a first modified oligonucleotide e.
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201- 206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a region of complementarity to the first modified oligonucleotide.
- a first modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a second modified oligonucleotide
- a compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 and SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide 19 to 23 linked nucleosides in length having a region of complementarity to the first modified oligonucleotide.
- Certain embodiments are drawn to the use of a compound comprising a MAPT specific inhibitor for the manufacture or preparation of a medicament for treating a disease associated with MAPT. Certain embodiments are drawn to the use of a compound comprising a MAPT specific inhibitor for the preparation of a medicament for treating a disease, disorder or condition associated with MAPT. In certain embodiments, the disease, disorder or condition is a CNS related disease, disorder or condition or a symptom thereof.
- the disease, disorder or condition is a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP- 17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- the compound comprises an antisense oligonucleotide targeted to MAPT. In certain embodiments, the compound comprises an oligonucleotide targeted to MAPT.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188,
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) and having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194- 197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequences of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190- 192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- the compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11- 73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 and a second modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- the compound can be single-stranded or double-stranded.
- the compound can be an antisense oligonucleotide or oligomeric compound.
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a region of complementarity to the first modified oligonucleotide.
- a first modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a second modified oligonucleotide e.
- a compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 and SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide 19 to 23 linked nucleosides in length having a region of complementarity to the first modified oligonucleotide.
- Certain embodiments are drawn to the use of a compound comprising a MAPT specific inhibitor for the manufacture or preparation of a medicament for reducing or inhibiting a CNS related disease, disorder or condition or a symptom thereof in an individual having, or at risk of having, a CNS related disease, disorder or condition or a symptom thereof associated with MAPT.
- the CNS related disease, disorder or condition is a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- Certain embodiments are drawn to use of a compound comprising a MAPT specific inhibitor for the preparation of a medicament for treating a disease, disorder or condition associated with MAPT.
- the disease, disorder or condition is a CNS related disease, disorder or condition or a symptom thereof or a neurodegenerative disease or a symptom thereof, including a tauopathy, Alzheimer’s disease, frontotemporal dementia (FTD), FTDP-17, progressive supranuclear palsy (PSP), chronic traumatic encephalopathy (CTE), corticobasal ganglionic degeneration (CBD), epilepsy, Dravet’s Syndrome or cognitive impairment.
- the compound comprises an antisense oligonucleotide targeted to MAPT.
- the compound comprises an oligonucleotide targeted to MAPT.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequences of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) and having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequences of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a compound comprises a modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- a compound comprises a modified oligonucleotide selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- the compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 11- 73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 and a second modified oligonucleotide having a nucleobase sequence selected from the group consisting of the nucleobase sequence of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- the compound can be single-stranded or double-stranded.
- the compound can be an antisense oligonucleotide or oligomeric compound.
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23 contiguous nucleobases of any of the nucleobase sequence of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having
- a compound comprises a first modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a nucleobase sequence comprising the nucleobase sequence of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 or SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide (e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length) having a region of complementarity to the first modified oligonucleotide.
- a first modified oligonucleotide e.g., of 14 to 30, for example, 14 to 23, linked nucleosides in length
- a second modified oligonucleotide e.
- a compound comprises a first modified oligonucleotide having a nucleobase sequence selected from the group consisting of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217 and SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206, and a second modified oligonucleotide 19 to 23 linked nucleosides in length having a region of complementarity to the first modified oligonucleotide.
- the compound can be an oligomeric compound.
- the compound can be single-stranded or double-stranded. In any of the foregoing methods or uses, the compound can be targeted to MAPT. In certain embodiments, the compound comprises or consists of a modified oligonucleotide. In certain embodiments, the compound comprises one or more modified oligonucleotides. In certain embodiments, the compound comprises a first modified oligonucleotide and a second modified oligonucleotide.
- a modified oligonucleotide is 8 to 80 linked nucleosides in length, 10 to 30 linked nucleosides in length, 14 to 30 linked nucleosides in length, 14 to 23 linked nucleosides in length, or 19 to 23 linked nucleosides in length.
- a modified oligonucleotide is at least 80%, at least 85%, at least 90%, at least 95% or 100% complementary to any of the nucleobase sequences recited in SEQ ID NOs: 1 and 3 over its length.
- a modified oligonucleotide comprises at least one modified internucleoside linkage, at least one modified sugar and/or at least one modified nucleobase.
- the modified internucleoside linkage is a phosphorothioate internucleoside linkage.
- the modified sugar is a bicyclic sugar, 2’-MOE, 2’-F, or 2’-OMe.
- the modified nucleobase is a 5-methylcytosine.
- each modified oligonucleotide is independently 12 to 30, 14 to 30, 14 to 25, 14 to 24, 14 to 23, 16 to 23, 17 to 23, 18 to 23, 19 to 23, 19 to 22, or 19 to 20 linked nucleosides in length.
- a modified oligonucleotide has at least 1, at least 2, at least 3 mismatches to a region of SEQ ID NO: 1 and 2.
- the compound comprises a first and second modified oligonucleotide, wherein there is a region of complementarity between a first modified oligonucleotide and a second modified oligonucleotide.
- the region of complementarity between the first oligonucleotide and the second oligonucleotide is 14 to 23, 19 to 23, or 21 to 23 linked nucleosides in length.
- the first modified oligonucleotide is fully complementary to the second modified oligonucleotide.
- the first modified oligonucleotide comprises at least one modification selected from a modified internucleoside linkage, a modified sugar, and a modified nucleobase.
- the second modified oligonucleotide comprises at least one modification selected from the group consisting of a modified internucleoside linkage, a modified sugar, and a modified nucleobase.
- the modified internucleoside linkage is a phosphorothioate internucleoside linkage or a methylphosphonate internucleoside linkage.
- the modified internucleoside linkage is at the 3’ terminus of the first or second modified oligonucleotide or at the 5’ terminus of the first or second modified oligonucleotide.
- the first or second modified oligonucleotide comprises one or more modified sugars.
- each nucleoside of the first or second modified oligonucleotide comprises a modified sugar.
- the modified sugar comprises a modification selected from the group consisting of a halogen, an alkoxy group and a bicyclic sugar.
- the modified sugar comprises a modification selected from group consisting of 2’-MOE, 2’-F, and 2’-OMe or a combination thereof.
- the first or second modified oligonucleotide comprises no more than ten 2’-F sugar modifications. In certain embodiments, the first or second modified oligonucleotide comprises no more than five 2’-F sugar modifications.
- a compound comprises a conjugate group. In certain embodiments, the conjugate group is attached to the 5’ end of a modified oligonucleotide. In certain embodiments, the conjugate group is attached to the 3’ end of a modified oligonucleotide.
- a conjugate group is attached to the 5’ end of a modified oligonucleotide, and a conjugate group is attached to the 3’ end of the modified oligonucleotide.
- the conjugate group is a targeting moiety.
- the targeting moiety comprises one or more ligands.
- the targeting moiety comprises one or more TrkB ligands.
- the one or more TrkB ligands is attached at the 5’ or 3’ end of the oligonucleotide or both the 5’ and 3’ ends of the oligonucleotide.
- the TrkB ligand is selected from Formulae I- XXXIV, XXXXVII, XXXXIX-XXXXXX, XXXXXV-XXXXXVII, and XXXXXIX-XXXXXXIII, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide.
- the modified oligonucleotide is attached to the TrkB ligand through a phosphodiester group.
- the modified oligonucleotide is attached to the TrkB ligand through a phosphorothioate group.
- the conjugate group comprises one or more lipids (e.g., one or more substituted or unsubstituted alkyl or alkenyl).
- the modified oligonucleotide comprises one or more ligands and one or more lipids.
- the modified oligonucleotide is the second modified oligonucleotide.
- the one or more lipids are attached to an internucleoside linkage of the modified oligonucleotide.
- the internucleoside linkage of the modified oligonucleotide is selected from any one of Formulae XXXXIII-XXXXVI, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide.
- the modified oligonucleotide comprises any one of Formulae XXXV-XXXXVI.
- the modified oligonucleotide comprises one or more TrkB ligands and one or more lipids (e.g., one or more substituted or unsubstituted alkyl or alkenyl).
- the modified oligonucleotide is the second modified oligonucleotide.
- the one or more TrkB ligands is attached to the 5’ end of the modified oligonucleotide.
- the one or more TrkB ligands is attached to the 3’ end of the modified oligonucleotide.
- the one or more TrkB ligands is attached to the 5’ end and the 3’ end of the modified oligonucleotide.
- the one or more lipids are attached to an internucleoside linkage of the modified oligonucleotide.
- the one or more TrkB ligands is selected from any one of Formulae I-XXXIV, XXXXVII, XXXXXIX-XXXXX, XXXXXV- XXXXXVII, and XXXXXIX-XXXXXXXIII, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide, and the internucleoside linkage of the modified oligonucleotide is selected from any one of Formulae XXXXIII-XXXXXVI, or a salt, solvate, or hydrate thereof.
- the one or more TrkB ligands is selected from any one of Formulae I-XXXIV, XXXXVII, XXXXIX-XXXXXX, XXXXXV- XXXXVII, and XXXXXIX-XXXXXXIII, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide, and the modified oligonucleotide also comprises any one of Formulae XXV-XXXXVI, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide.
- a compound comprises a conjugate group.
- the conjugate group is attached to the 5’ end of a modified oligonucleotide.
- the conjugate group is attached to the 3’ end of a modified oligonucleotide.
- a conjugate group is attached to the 5’ end of a modified oligonucleotide, and a conjugate group is attached to the 3’ end of the modified oligonucleotide.
- the conjugate group is a targeting moiety.
- the targeting moiety comprises one or more ligands.
- the targeting moiety comprises one or more CB 1 ligands.
- the one or more CB 1 ligands is attached at the 5’ or 3’ end of the oligonucleotide or both the 5’ and 3’ ends of the oligonucleotide.
- the CB 1 ligand is selected from Formulae XXXXXI and XXXXXXIV- XXXXXXVIII, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide.
- the modified oligonucleotide is attached to the CB 1 ligand through a phosphodiester group.
- the modified oligonucleotide is attached to the CB 1 ligand through a phosphorothioate group.
- the conjugate group comprises one or more lipids (e.g., one or more substituted or unsubstituted alkyl or alkenyl).
- the modified oligonucleotide comprises one or more ligands and one or more lipids (e.g., one or more substituted or unsubstituted alkyl or alkenyl).
- the modified oligonucleotide is the second modified oligonucleotide.
- the one or more lipids are attached to an internucleoside linkage of the modified oligonucleotide.
- the internucleoside linkage of the modified oligonucleotide is selected from any one of Formulae XXXXIII-XXXXVI, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide.
- the modified oligonucleotide comprises any one of Formulae XXXV-XXXXVI.
- the modified oligonucleotide comprises one or more CB 1 ligands, and one or more lipids.
- the modified oligonucleotide is the second modified oligonucleotide.
- the one or more CB 1 ligands is attached to the 5’ end of the modified oligonucleotide.
- the one or more CB 1 ligands is attached to the 3’ end of the modified oligonucleotide.
- the one or more CB 1 ligands is attached to the 5’ end and the 3’ end of the modified oligonucleotide.
- the one or more lipids are attached to an internucleoside linkage of the modified oligonucleotide.
- the one or more CB 1 ligands is selected from any one of Formulae XXXXXI and XXXXXXIV-XXXXXXVIII, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide, and the internucleoside linkage of the modified oligonucleotide is selected from any one of Formulae XXXXIII-XXXXXVI, or a salt, solvate, or hydrate thereof.
- the one or more CB 1 ligands is selected from any one of Formulae XXXXXI and XXXXXXIV-XXXXXXVIII, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide, and the modified oligonucleotide also comprises any one of Formulae XXXV-XXXXVI, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide.
- a compound comprises a conjugate group.
- the conjugate group is attached to the 5’ end of a modified oligonucleotide. In certain embodiments, the conjugate group is attached to the 3’ end of a modified oligonucleotide. In certain embodiments, a conjugate group is attached to the 5’ end of a modified oligonucleotide, and a conjugate group is attached to the 3’ end of the modified oligonucleotide. In certain embodiments, the conjugate group is a targeting moiety. In certain embodiments, the targeting moiety comprises one or more ligands. In certain embodiments, the targeting moiety comprises one or more ⁇ 4 ⁇ 1/7 integrin ligands.
- the one or more ⁇ 4 ⁇ 1/7 integrin ligands is attached at the 5’ or 3’ end of the oligonucleotide or both the 5’ and 3’ ends of the oligonucleotide.
- the ⁇ 4 ⁇ 1/7 integrin ligand is selected from Formulae XXXXXII- XXXXXIV and XXXXXXIX-XXXXXXXIV, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide.
- the modified oligonucleotide is attached to the ⁇ 4 ⁇ 1/7 integrin ligand through a phosphodiester group. In certain embodiments, the modified oligonucleotide is attached to the ⁇ 4 ⁇ 1/7 integrin ligand through a phosphorothioate group.
- the conjugate group comprises one or more lipids (e.g., one or more substituted or unsubstituted alkyl or alkenyl). In certain embodiments, the modified oligonucleotide comprises one or more ligands and one or more lipids (e.g., one or more substituted or unsubstituted alkyl or alkenyl).
- the modified oligonucleotide is the second modified oligonucleotide.
- the one or more lipids e.g., one or more substituted or unsubstituted alkyl or alkenyl
- the internucleoside linkage of the modified oligonucleotide is selected from any one of Formulae XXXXIII-XXXXVI, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide.
- the modified oligonucleotide comprises any one of Formulae XXXV-XXXXVI. In certain embodiments, the modified oligonucleotide comprises one or more ⁇ 4 ⁇ 1/7 integrin ligands and one or more lipids. In certain embodiments, the modified oligonucleotide is the second modified oligonucleotide. In certain embodiments, the one or more ⁇ 4 ⁇ 1/7 integrin ligands is attached to the 5’ end of the modified oligonucleotide. In certain embodiments, the one or more ⁇ 4 ⁇ 1/7 integrin ligands is attached to the 3’ end of the modified oligonucleotide.
- the one or more ⁇ 4 ⁇ 1/7 integrin ligands is attached to the 5’ end and the 3’ end of the modified oligonucleotide.
- the one or more lipids are attached to an internucleoside linkage of the modified oligonucleotide.
- the one or more ⁇ 4 ⁇ 1/7 integrin ligands is selected from any one of Formulae XXXXXII-XXXXXIV and XXXXXXIX-XXXXXXXIV, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide, and the internucleoside linkage of the modified oligonucleotide is selected from any one of Formulae XXXXIII-XXXXXVI, or a salt, solvate, or hydrate thereof.
- the one or more ⁇ 4 ⁇ 1/7 integrin ligands is selected from any one of Formulae XXXXXII-XXXXXIV and XXXXXXIX-XXXXXXXIV, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide, and the modified oligonucleotide also comprises any one of Formulae XXXV-XXXXVI, or a salt, solvate, or hydrate thereof, wherein R is the modified oligonucleotide.
- a compound comprises one or more substituted or unsubstituted alkyl or alkenyl.
- the substituted or unsubstituted alkyl or alkenyl is attached to an internucleoside linkage of a modified oligonucleotide.
- the modified oligonucleotide comprises one or more substituted or unsubstituted alkyl or alkenyl.
- the one or more substituted or unsubstituted alkyl or alkenyl are attached to one or more internucleoside linkages of the modified oligonucleotide.
- the modified oligonucleotide is the second modified oligonucleotide or sense oligonucleotide.
- the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 4 -C 30 hydrocarbon chain.
- the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 5 -C 20 hydrocarbon chain.
- the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 14 -C 20 hydrocarbon chain.
- the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 16 hydrocarbon chain. In certain embodiments, the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C17 hydrocarbon chain. In certain embodiments, the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 18 hydrocarbon chain. In certain embodiments, the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 22 hydrocarbon chain.
- a substituted or unsubstituted alkyl or alkenyl is attached to an internucleoside linkage of a modified oligonucleotide (e.g., a second modified oligonucleotide or sense oligonucleotide).
- a substituted or unsubstituted alkyl or alkenyl is attached to an internucleoside linkage of a modified oligonucleotide (e.g., a second modified oligonucleotide or sense oligonucleotide).
- the internucleoside linkage is between nucleosides that are within 10 positions (e.g., within 8 positions, within 6 positions, within 5 positions, within 4 positions, within 3 positions, within 2 positions) from a terminal end (e.g., the 5′ and/or 3′ end) of the modified oligonucleotide. In certain embodiments, the internucleoside linkage is between nucleosides that are within 5 positions from the 5′ end of the modified oligonucleotide. In certain embodiments, the internucleoside linkage is between nucleosides that are within 5 positions from the 3′ end of the modified oligonucleotide.
- the internucleoside linkage is between positions 1 and 2, positions 2 and 3, positions 3 and 4, positions 4 and 5, positions 5 and 6, positions 6 and 7, positions 7 and 8, positions 8 and 9, positions 9 and 10, positions 10 and 11, positions 11 and 12, positions 12 and 13, or positions 13 and 14 from the 5′ end of the modified oligonucleotide. In certain embodiments, the internucleoside linkage is between positions 1 and 2, positions 2 and 3, positions 3 and 4, positions 4 and 5, positions 5 and 6, positions 6 and 7, or positions 7 and 8 from the 5′ end of the modified oligonucleotide. In certain embodiments, the internucleoside linkage is between positions 2 and 3 from the 5′ end of the modified oligonucleotide.
- the internucleoside linkage is between positions 1 and 2, positions 2 and 3, positions 3 and 4, positions 4 and 5, positions 5 and 6, positions 6 and 7, positions 7 and 8, positions 8 and 9, positions 9 and 10, positions 10 and 11, positions 11 and 12, positions 12 and 13, or positions 13 and 14 from the 3′ end of the modified oligonucleotide. In certain embodiments, the internucleoside linkage is between positions 1 and 2, positions 2 and 3, positions 3 and 4, positions 4 and 5, positions 5 and 6, positions 6 and 7, or positions 7 and 8 from the 3′ end of the modified oligonucleotide. In certain embodiments, the internucleoside linkage is between positions 2 and 3 from the 3′ end of the modified oligonucleotide.
- the internucleoside linkage of the modified oligonucleotide is selected from any one of Formulae XXXXIII-XXXXVI.
- the modified oligonucleotide comprises any one of Formulae XXXV-XXXXVI.
- a compound comprises a 5′-phosphonate modification.
- a modified oligonucleotide comprises one or more sugars having a phosphonate modification at the 5′ position.
- the modified oligonucleotide comprises a 5′-phosphonate modification.
- the modified oligonucleotide comprises a 5′-terminal nucleoside (e.g., 5′ terminus) comprising the 5′-phosphonate modification.
- the 5′-phosphonate modification is a 5′-vinylphosphonate modification or a 5′- ethylenephosphonate modification.
- the 5′-phosphonate modification is a 5′-vinylphosphonate modification.
- the 5′-phosphonate modification is a 5′-ethylenephosphonate modification.
- the modified oligonucleotide is the first modified oligonucleotide or antisense oligonucleotide.
- the compound comprises a first modified oligonucleotide selected from the group consisting of any one of the IA Ref ID NOs in Table 3 and a second modified oligonucleotide 14 to 23 linked nucleosides in length fully complementary to the first modified oligonucleotide.
- the compound comprises a first modified oligonucleotide selected from any one of the IA Ref ID NOs in Table 3 and a second modified oligonucleotide selected from any one of the IS Ref ID NOs in Table 3.
- the compound is in a pharmaceutically acceptable salt form.
- the pharmaceutically acceptable salt is a sodium salt.
- the pharmaceutically acceptable salt is a potassium salt.
- a composition comprises the compound of any one of the foregoing embodiments and a pharmaceutically acceptable carrier.
- a compound or composition comprising a compound of any preceding embodiment is administered to an individual in a therapeutically effective amount.
- a compound or composition comprising a compound of any preceding embodiment is administered to an individual at a dosage level sufficient to deliver about 1 to 100 mg/kg of body weight of the individual.
- a compound or composition comprising a compound of any preceding embodiment is administered to an individual at a fixed dose of about 25 mg to about 1,000 mg.
- the composition is administered to the individual one or more times in a day up to the dosage level or fixed dose.
- a compound or composition comprising a compound of any preceding embodiment is administered to an individual daily, weekly, monthly, quarterly or yearly.
- a compound or composition comprising a compound of any preceding embodiment is administered to an individual about once per quarter (i.e., once every three months) to about once per year.
- a compound or composition comprising a compound of any preceding embodiment is administered to an individual about once per quarter, about once every six months or about once per year.
- the oligomeric compound comprises a nucleobase sequence complementary to that of a target nucleic acid.
- the disclosure relates to a compound that comprises or consists of a modified oligonucleotide.
- the modified oligonucleotide has a nucleobase sequence complementary to that of a target nucleic acid.
- the disclosure relates to a compound that comprises or consists of an antisense oligonucleotide.
- the antisense oligonucleotide has a nucleobase sequence complementary to that of a target nucleic acid.
- the disclosure relates to a compound that is a single-stranded compound.
- the single-stranded compound comprises or consists of an oligomeric compound.
- such an oligomeric compound comprises or consists of an oligonucleotide and optionally a conjugate group.
- the oligonucleotide is a modified oligonucleotide.
- the oligonucleotide is an antisense oligonucleotide.
- the oligonucleotide or modified oligonucleotide of a single-stranded compound comprises a self-complementary nucleobase sequence.
- the disclosure relates to a compound that is a double-stranded compound.
- the double-stranded compound comprises or consists of an oligomeric compound.
- the double-stranded compound comprises a first oligonucleotide and a second oligonucleotide.
- the first oligonucleotide has a region complementarity to a target nucleic acid and the second oligonucleotide has a region complementarity to the first modified oligonucleotide.
- the double-stranded compound comprises a modified oligonucleotide.
- the modified oligonucleotide has a region complementarity to a target nucleic acid.
- the double-stranded compound comprises a first modified oligonucleotide and a second modified oligonucleotide.
- the first modified oligonucleotide has a region complementarity to a target nucleic acid and the second modified oligonucleotide has a region complementarity to the first modified oligonucleotide.
- an oligonucleotide or modified oligonucleotide of a double-stranded compound is an RNA oligonucleotide.
- the thymine nucleobase in the modified oligonucleotide is replaced by a uracil nucleobase.
- a compound described herein comprises a conjugate group.
- the first oligonucleotide or first modified oligonucleotide of a double-stranded compound comprises a conjugate group.
- the second oligonucleotide or second modified oligonucleotide of a double-stranded compound comprises a conjugate group.
- a first oligonucleotide or first modified oligonucleotide and a second oligonucleotide or second modified oligonucleotide of a double-stranded compound each comprises a conjugate group.
- a compound is 14-30 linked nucleosides in length.
- the first oligonucleotide or first modified oligonucleotide of a double-stranded compound is 14-30 linked nucleosides in length.
- the second oligonucleotide or second modified oligonucleotide is 14-30 linked nucleosides in length.
- the oligonucleotides or modified oligonucleotides of a double-stranded compound are blunt ended at one or both ends of the compound.
- the oligonucleotides or modified oligonucleotides of a double-stranded compound include non- complementary overhanging nucleosides at one or both ends of the compound.
- a compound has a nucleobase sequence comprising at least 14 contiguous nucleobases of any of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194- 197, 199-200 and 209-217.
- one of the oligonucleotides or modified oligonucleotides of a double-stranded compound has a nucleobase sequence comprising at least 14 contiguous nucleobases of any of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- single-stranded and double-stranded compounds include, but are not limited to, oligonucleotides, antisense oligonucleotides, siRNAs, microRNA targeting oligonucleotides, occupancy-based compounds (e.g., mRNA processing or translation blocking compounds and splicing compounds), and single-stranded RNAi compounds (e.g. small hairpin RNAs (shRNAs), single stranded siRNAs (ssRNAs) and microRNA mimics).
- a compound described herein has a nucleobase sequence that, when written in the 5’ to 3’ direction, comprises the reverse complement of the target region of a target nucleic acid to which it is targeted.
- a compound described herein comprises an oligonucleotide 12 to 30 linked subunits in length. In certain embodiments, a compound described herein comprises an oligonucleotide 12 to 23 linked subunits in length. In certain embodiments, compound described herein comprises an oligonucleotide 14 to 30 linked subunits in length. In certain embodiments, compound described herein comprises an oligonucleotide 14 to 23 linked subunits in length. In certain embodiments, a compound described herein comprises an oligonucleotide 15 to 30 linked subunits in length. In certain embodiments, a compound described herein comprises an oligonucleotide 15 to 23 linked subunits in length.
- a compound described herein comprises an oligonucleotide 16 to 30 linked subunits in length. In certain embodiments, a compound described herein comprises an oligonucleotide 16 to 23 linked subunits in length. In certain embodiments, a compound described herein comprises an oligonucleotide 17 to 30 linked subunits in length. In certain embodiments, a compound described herein comprises an oligonucleotide 17 to 23 linked subunits in length. In certain embodiments, a compound described herein comprises an oligonucleotide 18 to 30 linked subunits in length. In certain embodiments, a compound described herein comprises an oligonucleotide 18 to 23 linked subunits in length.
- a compound described herein comprises an oligonucleotide 19 to 30 linked subunits in length. In certain embodiments, a compound described herein comprises an oligonucleotide 19 to 23 linked subunits in length.
- such oligonucleotides are 12 to 30 linked subunits, 12 to 23 linked subunits, 14 to 30 linked subunits, 14 to 23 linked subunits, 15 to 30 linked subunits, 15 to 23 linked subunits, 16 to 30 linked subunits, 16 to 23 linked subunits, 17 to 30 linked subunits, 17 to 23 linked subunits, 18 to 30 linked subunits, 18 to 23 linked subunits, 19 to 30 linked subunits or 19 to 23 linked subunits, respectively.
- a compound described herein comprises an oligonucleotide 14 linked subunits in length. In certain embodiments, a compound described herein comprises an oligonucleotide 16 linked subunits in length. In certain embodiments, a compound described herein comprises an oligonucleotide 17 linked subunits in length. In certain embodiments, compound described herein comprises an oligonucleotide 18 linked subunits in length. In certain embodiments, a compound described herein comprises an oligonucleotide 19 linked subunits in length. In certain embodiments, a compound described herein comprises an oligonucleotide 20 linked subunits in length.
- a compound described herein comprises an oligonucleotide 21 linked subunits in length. In certain embodiments, a compound described herein comprises an oligonucleotide 22 linked subunits in length. In certain embodiments, a compound described herein comprises an oligonucleotide 23 linked subunits in length. In other embodiments, a compound described herein comprises an oligonucleotide 8 to 80, 12 to 50, 13 to 30, 13 to 50, 14 to 30, 14 to 50, 15 to 30, 15 to 50, 16 to 30, 16 to 50, 17 to 30, 17 to 50, 18 to 23, 18 to 24, 18 to 25, 18 to 50, 19 to 23, 19 to 30, 19 to 50, 20 to 23 or 20 to 30 linked subunits.
- the compound described herein comprises an oligonucleotide 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, or 30 linked subunits in length, or a range defined by any two of the above values.
- the compound may further comprise an additional moiety, such as a conjugate group or delivery moiety.
- such compounds are oligomeric compounds, and the additional moiety is attached to an oligonucleotide.
- a conjugate group is attached to a nucleoside of an oligonucleotide.
- compounds may be shortened or truncated.
- one or more subunits may be deleted from the 5’ end (5’ truncation), or alternatively from the 3’ end (3’ truncation) of an oligonucleotide.
- compounds may be lengthened.
- one or more subunits may be attached to the 3′ end or 5′ end of an oligonucleotide.
- At least one subunit e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, or more subunits
- oligonucleotide e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, or more subunits
- At least one subunit e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, or more subunits
- at least one or more subunits may be attached to the 3′ end or 5′ end of an oligonucleotide of a double-stranded compound creating a 3′ and/or 5′ end overhang.
- At least one subunit e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, or more subunits
- at least one subunit is attached to the 5′ end of both oligonucleotides of a double-stranded compound.
- At least one subunit e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, or more subunit
- at least one subunit is attached to the 3′ end of both oligonucleotides of a double-stranded compound.
- subunits are attached to both oligonucleotides of a double-stranded compound at the same end (e.g., that subunits are attached to the 3′ end of one of the oligonucleotides and subunits are attached to the 5′ end of the other oligonucleotide).
- the number of subunits attached to each oligonucleotide may be the same or may be different.
- the number of subunits attached to each oligonucleotide is the same. In certain embodiments, when subunits are attached to both oligonucleotides of a double-stranded compound at the same end, the number of subunits attached to each oligonucleotide is different. This scenario, where subunits are attached to both oligonucleotides of a double-stranded compound at the same end, may occur at one or both ends of a double-stranded compound. In certain embodiments, the subunits attached to the 3′ and/or 5′ end are modified.
- compounds described herein are oligonucleotides. In certain embodiments, compounds described herein are modified oligonucleotides. In certain embodiments, compounds described herein are antisense oligonucleotides. In certain embodiments, compounds described herein are oligomeric compounds. In certain embodiments, compounds described herein are RNAi compounds. In certain embodiments, compounds described herein are siRNA compounds. In certain embodiments, a compound described herein can comprise any of the oligonucleotide sequences targeted to MAPT described herein. In certain embodiments, the compound can be double-stranded.
- the compound comprises an oligonucleotide comprising at least an 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22 or 23 contiguous nucleobase portion of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- the compound comprises an oligonucleotide comprising at least an 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22 or 23 contiguous nucleobase portion of any one of SEQ ID NOs: 11-73, 144-147, 177-188, 190-192, 194-197, 199-200 and 209-217.
- the compound comprises a second oligonucleotide.
- the compound comprises an oligonucleotide comprising at least an 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22 or 23 contiguous nucleobase portion of any one of SEQ ID NOs: 81-143, 149-155, 157, 159-168, 171-172, 174, 176 and 201-206.
- the compound comprises ribonucleotides in which the oligonucleotide has uracil (U) in place of thymine ( ⁇ ) for any of the sequences provided here.
- the compound comprises deoxyribonucleotides in which the oligonucleotide has thymine ( ⁇ ) in place of uracil (U) for any of the sequences provided here.
- compounds described herein comprise or consist of modified oligonucleotides.
- compounds described herein comprise or consist of antisense oligonucleotides.
- compounds comprise or consist of oligomeric compounds.
- compounds described herein are capable of hybridizing to a target nucleic acid.
- compounds described herein selectively affect one or more target nucleic acid.
- Such compounds comprise a nucleobase sequence that hybridizes to one or more target nucleic acid, resulting in one or more desired activity and does not hybridize to one or more non-target nucleic acid or does not hybridize to one or more non-target nucleic acid in such a way that results in a significant undesired activity.
- hybridization of a compound described herein to a target nucleic acid results in recruitment of one or more proteins that cause the cleavage of the target nucleic acid.
- certain compounds described herein or a portion of the compound is loaded into an RNA-induced silencing complex (RISC), ultimately resulting in cleavage of the target nucleic acid.
- RISC RNA-induced silencing complex
- RNAi compounds may be double-stranded (siRNA) or single-stranded (ssRNA).
- hybridization of compounds described herein to a target nucleic acid does not result in recruitment of a protein that cleaves that target nucleic acid.
- hybridization of the compound to the target nucleic acid results in the alteration of splicing of the target nucleic acid.
- hybridization of the compound to a target nucleic acid results in inhibition of a binding interaction between the target nucleic acid and a protein or other nucleic acid.
- hybridization of the compound to the target nucleic acid results in the alteration of RNA processing. In certain such embodiments, hybridization of the compound to a target nucleic acid results in alteration of translation of the target nucleic acid. Activities resulting from the hybridization of a compound to a target nucleic acid may be observed directly or indirectly. In certain embodiments, observation or detection of an activity involves observation or detection of a change in an amount of a target nucleic acid or protein encoded by such target nucleic acid, a change in the ratio of splice variants of a nucleic acid or protein, and/or a phenotypic change in a cell or animal.
- the disclosure relates to compounds that comprise or consist of oligonucleotides.
- Oligonucleotides consist of linked nucleosides.
- oligonucleotides may be unmodified RNA or DNA or may be modified.
- the oligonucleotides are modified oligonucleotides.
- the modified oligonucleotides comprise at least one modified sugar, modified nucleobase or modified internucleoside linkage relative to an unmodified RNA or DNA.
- an oligonucleotide has a modified nucleoside.
- a modified nucleoside may comprise a modified sugar, a modified nucleobase or both a modified sugar and a modified nucleobase.
- Modified oligonucleotides may also include end modifications, e.g., 5’-end modifications and 3’-end modifications.
- Sugar Modifications and Motifs In certain embodiments, a modified sugar is a substituted furanosyl sugar or non- bicyclic modified sugar. In certain embodiments, a modified sugar is a bicyclic or tricyclic modified sugar. In certain embodiments, a modified sugar is a sugar surrogate. A sugar surrogate may comprise one or more substitutions described herein. In certain embodiments, a modified sugar is a substituted furanosyl or non-bicyclic modified sugar.
- the furanosyl sugar is a ribosyl sugar.
- the furanosyl sugar comprises one or more substituent groups, including, but not limited to, substituent groups at the 2’, 3’, 4’, and 5’ positions.
- substituents at the 2’ position include, but are not limited to, F and OCH 3 (“OMe”, “O-methyl” or “methoxy”).
- substituent groups at the 2’ position suitable for non-bicyclic modified sugars include, but are not limited to, halo, allyl, amino, azido, SH, CN, OCN, CF 3 , OCF 3 , F, Cl, Br, SCH 3 , SOCH 3 , SO 2 CH 3 , ⁇ 2 , ⁇ 2 , ⁇ 3 , and ⁇ H 2 .
- substituent groups at the 2’ position include, but are not limited to, O-(C 1 -C 10 ) alkoxy, alkoxyalkyl, O-alkyl, S-alkyl, N-alkyl, O- alkenyl, S-alkenyl, N-alkenyl, O-alkynyl, S-alkynyl, N-alkynyl, O-alkyl-O-alkyl, alkynyl, wherein the alkyl, alkenyl and alkynyl can be substituted or unsubstitutedC 1 to C 10 alkyl or C 2 to C 10 alkenyl and alkynyl.
- substituent groups at the 2’ position include, but are not limited to, alkaryl, aralkyl, O-alkaryl, and O-aralkyl.
- these 2’ substituent groups can be further substituted with one or more substituent groups independently selected from hydroxyl, alkoxy, carboxy, benzyl, phenyl, nitro ( ⁇ 2 ), thiol, thioalkoxy, thioalkyl, halogen, alkyl, aryl, alkenyl, and alkynyl.
- substituent groups at the 2’ position include, but are not limited to, O[(CH 2 )nO]mCH 3 , O(CH 2 )nOCH 3 , O(CH 2 )nCH 3 , O(CH2)nONH 2 , O(CH 2 )nNH 2 , O(CH 2 ) n SCH 3 , and O(CH 2 ) n ON[(CH 2 ) n CH 3 )] 2 , where n and m are independently from 1 to about 10.
- substituent groups at the 4’ position suitable for non-bicyclic modified sugars include, but are not limited to, alkoxy (e.g., methoxy), alkyl, and those described in Manoharan et al., WO 2015/106128.
- substituent groups at the 5’ position suitable for non-bicyclic modified sugars include, but are not limited to, methyl (“Me” or “CH 3 ”) (R or S), vinyl, and methoxy.
- the 5' modification is a 5'-monophosphate ((HO)2(O)P-O-5'); 5'-diphosphate ((HO)2(O)P-O- P(HO)(O)-O-5'); 5'-triphosphate ((HO) 2 (O)P-O-(HO)(O)P-O-P(HO)(O)-O-5'); 5'-guanosine cap (7-methylated or non-methylated) (7m-G-O-5'-(HO)(O)P-O-(HO)(O)P-O-P(HO)(O)-O- 5'); 5'adenosine cap (Appp), and any modified or unmodified nucleotide cap structure (N-O- 5'(HO)
- one or more sugars comprise a 5′- phosphonate modification.
- the 5′-phosphonate modification is a 5′- vinylphosphonate modification or a 5′-ethylenephosphonate modification.
- one or more sugars comprise a 5′-vinylphosphonate modification.
- one or more sugars comprise a 5′-ethylenephosphonate modification.
- the 5′ modification is at the terminus of an oligonucleotide. In certain embodiments the 5′ modification is at the terminus of an antisense oligonucleotide.
- substituents described herein for the 2’, 4’ and 5’ position can be added to other specific positions on the sugar. In certain embodiments, such substituents may be added to the 3’ position of the sugar on the 3’ terminal nucleoside or the 5’ position of the 5’ terminal nucleoside.
- a non-bicyclic modified sugar may comprise more than one non-bridging sugar substituent.
- non-bicyclic modified sugars substituents include, but are not limited to, 5’-Me-2’-F, 5’-Me-2’-OMe (including both R and S isomers).
- modified sugar substituents include those described in Migawa et al., WO 2008/101157 and Rajeev et al., US2013/0203836.
- a modified sugar is a bicyclic sugar.
- a bicyclic sugar is a modified sugar comprising two rings, wherein the second ring is formed via a bridge connecting two of the atoms in the first ring thereby forming a bicyclic structure.
- a bicyclic sugar comprises a bridging substituent that bridges two atoms of the furanosyl ring to form a second ring. In certain embodiments, a bicyclic sugar does not comprise a furanosyl moiety.
- a “bicyclic nucleoside” (“BNA”) is a nucleoside having a bicyclic sugar.
- the bicyclic sugar comprises a bridge between the 4’ and 2’ furanose ring atoms.
- the bicyclic sugar comprises a bridge between the 5’ and 3’ furanose ring atoms.
- the furanose ring is a ribose ring.
- 4’ to 2’ bridging substituents include, but are not limited to, 4'-CH 2 -2', 4'-(CH 2 ) 2 -2', 4'- (CH 2 ) 3 -2', 4'-CH 2 -O-2' (“LNA”), 4'-CH 2 -S-2', 4'- (CH 2 )2-O-2' (“ENA”), 4'-CH(CH 3 )-O-2' (“constrained ethyl” or “cEt” when in the S configuration), 4’-CH2-O-CH 2 -2’, 4’-CH 2 -N(R)-2’, 4'- CH(CH 2 OCH 3 )-O-2' (“constrained MOE” or “cMOE”) and analogs thereof (e.g., U.S.
- Patent No. 7,399,845) 4'-C(CH 3 )(CH 3 )- O-2' and analogs thereof (e.g., U.S. Patent No. 8,278,283), 4'-CH 2 -N(OCH 3 )-2' and analogs thereof (e.g., U.S. Patent No. 8,278,425), 4'-CH 2 -O-N(CH 3 )-2' (e.g., U.S. Patent Publication No. 2004/0171570), 4'-CH 2 -N(R)-O-2', wherein R is ⁇ , C 1 -C 12 alkyl, or a protecting group (e.g., U.S. Patent No.
- bicyclic nucleosides can be prepared having one or more stereochemical sugar configurations including for example ⁇ -L-ribofuranose and ⁇ -D-ribofuranose (see e.g., WO 99/14226). Specified bicyclic nucleosides herein are in the ⁇ -D configuration, unless otherwise specified.
- a modified sugar is a sugar surrogate.
- a sugar surrogate has the oxygen atom replaced, e.g., with a sulfur, carbon or nitrogen atom.
- the sugar surrogate may also comprise bridging and/or non-bridging substituents as described herein.
- sugar surrogates comprise rings having other than 5 atoms.
- the sugar surrogate comprises a cyclobutyl moiety in place of the pentofuranosyl sugar.
- the sugar surrogate comprises a six membered ring in place of the pentofuranosyl sugar.
- the sugar surrogate comprises a tetrahydropyran (“THP”) in place of the pentofuranosyl sugar.
- the sugar surrogate comprises a morpholino in place of the pentofuranosyl sugar.
- sugar surrogates comprise acyclic moieties.
- the sugar surrogate is an unlocked nucleic acid (“UNA”).
- UNA is unlocked acyclic nucleic acid, wherein any of the bonds of the sugar has been removed, forming an unlocked "sugar” residue.
- UNA also encompasses a monomer where the bonds between C1’-C4’ have been removed (i.e. the covalent carbon-oxygen-carbon bond between the C1’ and C4’ carbons).
- the C2’-C3’ bond i.e. the covalent carbon-carbon bond between the C2’ and C3’ carbons
- sugar surrogates comprise peptide nucleic acid (“PNA”), acyclic butyl nucleic acid (see, e.g., Kumar et al., Org. Biomol. Chem., 2013, 11, 5853-5865), and nucleosides and oligonucleotides described in Manoharan et al., US2013/130378, the entire contents of which is hereby incorporated herein by reference.
- PNA peptide nucleic acid
- acyclic butyl nucleic acid see, e.g., Kumar et al., Org. Biomol. Chem., 2013, 11, 5853-5865
- nucleosides and oligonucleotides described in Manoharan et al., US2013/130378, the entire contents of which is hereby incorporated herein by reference.
- the disclosure relates to compounds comprising at least one oligonucleotide wherein the nucleosides of such oligonucleotide comprise one or more types of modified sugars and/or unmodified sugars arranged along the oligonucleotide or region thereof in a defined pattern or “sugar motif”.
- such sugar motifs include, but are not limited to, any of the patterns of sugar modifications described herein.
- an oligonucleotide comprises a gapmer sugar motif.
- a gapmer oligonucleotide comprises or consists of a region having two external “wing” regions and a central or internal “gap” region.
- the gap and wing regions form a contiguous sequence of nucleosides, wherein the majority of nucleoside sugars of each of the wings differ from the majority of nucleoside sugars of the gap.
- the wing regions comprise a majority of modified sugars and the gap comprises a majority of unmodified sugars.
- the nucleosides of the gap are deoxynucleosides.
- one or both oligonucleotides of a double-stranded compound comprise a triplet sugar motif.
- An oligonucleotide with a triplet sugar motif comprises three identical sugar modifications on three consecutive nucleosides.
- the triplet is at or near the cleavage site of the oligonucleotide.
- an oligonucleotide of a double-stranded compound may contain more than one triplet sugar motif.
- the identical sugar modification of the triplet sugar motif is a 2’-F modification.
- one or both oligonucleotides of a double-stranded compound comprise a quadruplet sugar motif.
- An oligonucleotide with a quadruplet sugar motif comprises four identical sugar modifications on four consecutive nucleosides.
- the quadruplet is at or near the cleavage site.
- an oligonucleotide of a double-stranded compound may contain more than one quadruplet sugar motif.
- the identical sugar modification of the quadruplet sugar motif is a 2’-F modification.
- the cleavage site of the antisense oligonucleotide is typically around the 10, 11, and 12 positions from the 5’-end.
- the quadruplet sugar motif is at the 8, 9, 10, 11 positions; the 9, 10, 11, 12 positions; the 10, 11, 12, 13 positions; the 11, 12, 13, 14 positions; or the 12, 13, 14, 15 positions of the sense oligonucleotide, counting from the first nucleoside of the 5’-end of the sense oligonucleotide, or, the count starting from the first paired nucleotide within the duplex region from the 5’-end of the sense oligonucleotide.
- the quadruplet sugar motif is at the 8, 9, 10, 11 positions; the 9, 10, 11, 12 positions; the 10, 11, 12, 13 positions; the 11, 12, 13, 14 positions; or the 12, 13, 14, 15 positions of the antisense oligonucleotide, counting from the first nucleoside of the 5’-end of the antisense oligonucleotide, or, the count starting from the first paired nucleotide within the duplex region from the 5’- end of the antisense oligonucleotide.
- the cleavage site may change according to the length of the duplex region of the double- stranded compound and may change the position of the quadruplet accordingly.
- an oligonucleotide comprises an alternating sugar motif.
- one or both oligonucleotides of a double-stranded compound comprise an alternating sugar motif.
- An oligonucleotide with an alternating sugar motif comprises at least two different sugar modifications wherein one or more consecutive nucleosides comprising a first sugar modification alternates with one or more consecutive nucleosides comprising a second sugar modification and one or more consecutive nucleosides comprising a third sugar modification, etc.
- the alternating motif can be “ABABABABABAB...,” “AABBAABBAABB...,” “AABAABAABAAB...,” “AAABAAABAAAB...,” “AAABBBAAABBB...,” or “ABCABCABCABC...” etc.
- the alternating sugar motif is repeated for at least 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23 contiguous nucleobases along an oligonucleotide.
- the alternating sugar motif is comprised of two different sugar modifications.
- the alternating sugar motif comprises 2’-OMe and 2’-F sugar modifications.
- each nucleoside of an oligonucleotide is independently modified with one or more sugar modifications provided herein.
- each oligonucleotide of a double-stranded compound independently has one or more sugar motifs provided herein.
- an oligonucleotide containing a sugar motif is fully modified in that each nucleoside other than the nucleosides comprising the sugar motif comprises a sugar modification.
- Nucleobase Modifications and Motifs In certain embodiments, compounds described herein comprise modified oligonucleotides.
- modified oligonucleotides comprise one or more nucleosides comprising a modified nucleobase.
- modified oligonucleotides comprise one or more nucleosides that do not comprise a nucleobase, referred to as an abasic nucleoside.
- modified nucleobases are selected from: 5-substituted pyrimidines, 6-azapyrimidines, alkyl or alkynyl substituted pyrimidines, alkyl substituted purines, and ⁇ -2, N-6 and O-6 substituted purines.
- modified nucleobases are selected from: 2-aminopropyladenine, 5- hydroxymethyl cytosine, 5- methylcytosine, xanthine, hypoxanthine, 2-aminoadenine, 6-N-methylguanine, 6-N- methyladenine, 2-propyladenine, 2-thiouracil, 2-thiothymine and 2-thiocytosine, 5-propynyl (C ⁇ C-CH 3 ) uracil, 5-propynylcytosine, 6-azouracil, 6-azocytosine, 6-azothymine, 5- ribosyluracil (pseudouracil), 4-thiouracil, 8-halo, 8-amino, 8-thiol, 8-thioalkyl, 8-hydroxyl, 8- aza and other 8-substituted purines, 5-halo, particularly, 5-bromo, 5-trifluoromethyl, 5- halouracil, and 5-halocytosine
- nucleobases include tricyclic pyrimidines, such as 1,3-diazaphenoxazine-2-one, 1,3-diazaphenothiazine-2- one, and 9-(2-aminoethoxy)-1,3-diazaphenoxazine-2-one (G-clamp).
- Modified nucleobases may also include those in which the purine or pyrimidine base is replaced with other heterocycles, for example, 7-deaza-adenine, 7-deazaguanosine, 2-aminopyridine and 2- pyridone.
- Further nucleobases include those disclosed in U.S.
- Patent 3,687,808 Modified Nucleosides in Biochemistry, Biotechnology and Medicine, Herdewijn, ⁇ . ed. Wiley-VCH, 2008; The Concise Encyclopedia Of Polymer Science And Engineering, pages 858-859; Kroschwitz, J.L., Ed., John Wiley & Sons, 1990, 858-859; Englisch et al., Angewandte Chemie, International Edition, 1991, 30, 613; Sanghvi, Y.S., Chapter 15, dsRNA Research and Applications, pages 289-302; Antisense Research and Applications, Crooke, S.T.
- compounds described herein comprise oligonucleotides.
- oligonucleotides comprise modified and/or unmodified nucleobases arranged along the oligonucleotide or region thereof in a defined pattern or motif.
- each nucleobase is modified.
- none of the nucleobases are modified.
- each purine or each pyrimidine is modified.
- each adenine is modified.
- each guanine is modified.
- each thymine is modified.
- each uracil is modified.
- each cytosine is modified.
- some or all of the cytosine nucleobases in a modified oligonucleotide are 5-methylcytosines.
- modified oligonucleotides comprise a block of modified nucleobases. In certain such embodiments, the block is at the 3’-end of the oligonucleotide.
- the block is within 3 nucleosides of the 3’-end of the oligonucleotide. In certain embodiments, the block is at the 5’-end of the oligonucleotide. In certain embodiments, the block is within 3 nucleosides of the 5’-end of the oligonucleotide.
- Internucleoside Linkage Modifications and Motifs A 3' to 5' phosphodiester linkage is the naturally occurring internucleoside linkage of RNA and DNA. In certain embodiments, compounds described herein have one or more modified, i.e. non-naturally occurring, internucleoside linkages.
- internucleoside linkages may impart desirable properties such as, for example, enhanced cellular uptake, enhanced affinity for target nucleic acids, and increased stability in the presence of nucleases.
- Methods of preparation of phosphorous- containing and non-phosphorous-containing internucleoside linkages are well known to those skilled in the art.
- Further neutral internucleoside linkages include nonionic linkages comprising siloxane (dialkylsiloxane), carboxylate ester, carboxamide, sulfide, sulfonate ester and amides (See, for example: Carbohydrate Modifications in Antisense Research; Y.S. Sanghvi and P.D. Cook, Eds., ACS Symposium Series 580; Chapters 3 and 4, 40-65).
- Further neutral internucleoside linkages include nonionic linkages comprising mixed ⁇ , O, S and CH 2 component parts.
- compounds provided herein comprise at least one modified internucleoside linkage.
- a modified internucleoside linkage may be placed at any position of an oligonucleotide.
- a modified internucleoside linkage may be placed within the sense oligonucleotide, antisense oligonucleotide, or both oligonucleotides of the double-stranded compound.
- the internucleoside linkage modification may occur on every nucleoside of an oligonucleotide.
- internucleoside linkage modifications may occur in an alternating pattern along an oligonucleotide.
- a double-stranded compound comprises 6-8 modified internucleoside linkages.
- the 6-8 modified internucleoside linkages are phosphorothioate internucleoside linkages or alkylphosphonate internucleoside linkages.
- the sense oligonucleotide comprises at least two modified internucleoside linkages at either or both the 5’-end and the 3’-end.
- the modified internucleoside linkages are phosphorothioate internucleoside linkages or alkylphosphonate internucleoside linkages.
- the antisense oligonucleotide comprises at least two modified internucleoside linkages at either or both the 5’-end and the 3’-end.
- the modified internucleoside linkages are phosphorothioate internucleoside linkages or alkylphosphonate internucleoside linkages.
- a double-stranded compound comprises an overhang region.
- a double-stranded compound comprises a phosphorothioate or alkylphosphonate internucleoside linkage modification in the overhang region.
- a double-stranded compound comprises a phosphorothioate or alkylphosphonate internucleotide linkage linking the overhang nucleotide with a paired nucleotide that is next to the overhang nucleotide.
- a phosphorothioate or alkylphosphonate internucleotide linkage linking the overhang nucleotide with a paired nucleotide that is next to the overhang nucleotide.
- modified oligonucleotides comprise one or more internucleoside linkages having chiral centers. Representative chiral internucleoside linkages include, but are not limited to, alkylphosphonates and phosphorothioates.
- Modified oligonucleotides comprising internucleoside linkages having chiral centers can be prepared as populations of modified oligonucleotides comprising stereorandom internucleoside linkages, or as populations of modified oligonucleotides comprising phosphorothioate linkages in particular stereochemical configurations.
- populations of modified oligonucleotides comprise phosphorothioate internucleoside linkages wherein all of the phosphorothioate internucleoside linkages are stereorandom.
- Such modified oligonucleotides can be generated using synthetic methods that result in random selection of the stereochemical configuration of each phosphorothioate linkage.
- each individual phosphorothioate of each individual oligonucleotide molecule has a defined stereoconfiguration.
- populations of modified oligonucleotides are enriched for modified oligonucleotides comprising one or more particular phosphorothioate internucleoside linkages in a particular, independently selected stereochemical configuration.
- the particular configuration of the particular phosphorothioate linkage is present in at least 65% of the molecules in the population. In certain embodiments, the particular configuration of the particular phosphorothioate linkage is present in at least 70% of the molecules in the population.
- the particular configuration of the particular phosphorothioate linkage is present in at least 80% of the molecules in the population. In certain embodiments, the particular configuration of the particular phosphorothioate linkage is present in at least 90% of the molecules in the population. In certain embodiments, the particular configuration of the particular phosphorothioate linkage is present in at least 99% of the molecules in the population.
- Such enriched populations of modified oligonucleotides can be generated using synthetic methods known in the art, e.g., methods described in Oka et al., JACS 125, 8307 (2003), Wan et al. Nuc. Acid. Res. 42, 13456 (2014), and WO 2017/015555.
- a population of modified oligonucleotides is enriched for modified oligonucleotides having at least one indicated phosphorothioate in the (Sp) configuration. In certain embodiments, a population of modified oligonucleotides is enriched for modified oligonucleotides having at least one phosphorothioate in the (Rp) configuration.
- Conjugate Groups In certain embodiments, the compounds described herein comprise or consist of one or more oligonucleotides and, optionally, one or more conjugate groups. Conjugate groups may be attached to either or both ends of an oligonucleotide and/or at any internal position.
- a conjugate group is attached at the 3’ end of an oligonucleotide. In certain embodiments, a conjugate group is attached at the 5’ end of an oligonucleotide. In certain embodiments, oligonucleotides are covalently attached to one or more conjugate groups. In certain embodiments, conjugate groups are terminal groups attached to either or both ends of an oligonucleotide. In certain such embodiments, terminal groups are attached at the 3’ end of an oligonucleotide. In certain such embodiments, terminal groups are attached at the 5’ end of an oligonucleotide.
- terminal groups include, but are not limited to, capping groups, phosphate moieties, protecting groups, modified or unmodified nucleosides, and two or more nucleosides that are independently modified or unmodified, such as an overhang.
- conjugate groups modify one or more properties of the attached oligonucleotide, including, but not limited to, pharmacodynamics, pharmacokinetics, stability, activity, half-life, binding, absorption, tissue distribution, cellular distribution, cellular uptake, charge and clearance.
- conjugate groups enhance the affinity of a compound for a selected target, e.g., molecule, cell or cell type, compartment, e.g., a cellular or organ compartment, tissue, organ or region of the body, as, e.g., compared to a compound absent such a conjugate group.
- conjugate groups impart a new property on the attached oligonucleotide, e.g., fluorophores or reporter groups that enable detection of the oligonucleotide.
- conjugate groups include, but are not limited to, intercalators, reporter molecules, polyamines, polyamides, peptides, carbohydrates, vitamin moieties, polyethylene glycols, thioethers, polyethers, cholesterols, thiocholesterols, cholic acid moieties, folate, lipids, phospholipids, biotin, phenazine, phenanthridine, anthraquinone, adamantane, acridine, fluoresceins, rhodamines, coumarins, fluorophores, and dyes.
- conjugate groups include an active drug substance, for example, aspirin, warfarin, phenylbutazone, ibuprofen, suprofen, fen-bufen, ketoprofen, (S)- (+)-pranoprofen, carprofen, dansylsarcosine, 2,3,5-triiodobenzoic acid, fingolimod, flufenamic acid, folinic acid, a benzothiadiazide, chlorothiazide, a diazepine, indo-methicin, a barbiturate, a cephalosporin, a sulfa drug, an antidiabetic, an antibacterial, or an antibiotic.
- active drug substance for example, aspirin, warfarin, phenylbutazone, ibuprofen, suprofen, fen-bufen, ketoprofen, (S)- (+)-pranoprofen, carprofen, dans
- conjugate groups are targeting moieties.
- a targeting moiety includes, but is not limited to, a lectin, glycoprotein, lipid, protein, peptide, peptide mimetic, receptor ligand, antibody, thyrotropin, melanotropin, surfactant protein A, carbohydrate, carbohydrate derivative, modified carbohydrate, carbohydrate cluster, polysaccharide, modified polysaccharide, or polysaccharide derivative, mucin carbohydrate, multivalent lactose, multivalent galactose, N-acetyl-galactosamine (GalNAc), N-acetylglucosamine multivalent mannose, multivalent fucose, glycosylated polyaminoacids, multivalent galactose, transferrin, bisphosphonate, polyglutamate, polyaspartate, a lipid, cholesterol, a steroid, bile acid, folate, vitamin ⁇ 12, vitamin A, bio
- conjugate groups may include, but are not limited to, the conjugate groups described in the following references such as cholesterol (e.g., Letsinger et al., Proc. Natl. Acid. Sci. USA, 1989, 86: 6553-6556), cholic acid (e.g., Manoharan et al., Biorg. Med. Chem. Let., 1994, 4:1053-1060), thioether, e.g., hexyl-S-tritylthiol (e.g., Manoharan et al., ⁇ nn. NY. Acad. Sci., 1992, 660:306-309; Manoharan et al., Biorg. Med. Chem.
- cholesterol e.g., Letsinger et al., Proc. Natl. Acid. Sci. USA, 1989, 86: 6553-6556
- cholic acid e.g., Manoharan et al., Biorg. Med. Chem.
- thiocholesterol e.g., Oberhauser et al., Nucl. Acids Res., 1992, 20:533-538
- aliphatic chains e.g., do-decan-diol or undecyl residues
- phospholipids e.g., di-hexadecyl-rac- glycerol or triethyl-ammonium 1,2-di-O-hexadecyl-rac-glycero-3-H-phosphonate (e.g, Manoharan et al., Tetrahedron Lett., 1995, 36:3651-365
- tocopherol e.g., Nishina et al., Molecular Therapy Nucleic Acids, 2015, 4, e220 and Nishina et al., Molecular Therapy, 2008, 16:734-740
- GalNAc and other carbohydrates (e.g., Maier et al., Bioconjugate Chemistry, 2003, 14, 18-29; Rensen et al., J. Med. Chem.
- Conjugate groups may be attached to oligonucleotides through conjugate linkers.
- a conjugate linker comprises a chain structure, such as a hydrocarbyl chain, or an oligomer of repeating units or combination of such repeating units.
- a conjugate linker comprises one or more groups selected from alkyl, amino, ⁇ x ⁇ , amide, disulfide, polyethylene glycol, ether, thioether, and hydroxylamino. In certain embodiments, a conjugate linker comprises at least one phosphorus group. In certain embodiments, a conjugate linker comprises at least one phosphate group. In certain embodiments, a conjugate linker includes at least one neutral linking group.
- conjugate linkers include, but are not limited to, pyrrolidine, 8-amino-3,6- dioxaoctanoic acid (ADO), succinimidyl 4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC) and 6-aminohexanoic acid ( ⁇ or AHA).
- ADO 8-amino-3,6- dioxaoctanoic acid
- SMCC succinimidyl 4-(N-maleimidomethyl) cyclohexane-1-carboxylate
- ⁇ or AHA 6-aminohexanoic acid
- conjugate linkers include, but are not limited to, substituted or unsubstituted C 1 -C 10 alkyl, substituted or unsubstituted C 2 - C 10 alkenyl, or substituted or unsubstituted C 2 -C 10 alkynyl, wherein a nonlimiting list of preferred substituent groups includes hydroxyl, amino, alkoxy, carboxy, benzyl, phenyl, nitro, thiol, thioalkoxy, halogen, alkyl, aryl, alkenyl, and alkynyl.
- conjugate linkers comprise 1-10 linker-nucleosides.
- linker- nucleosides may be modified or unmodified nucleosides. It is typically desirable for linker- nucleosides to be cleaved from the compound after it reaches a target tissue. Accordingly, linker-nucleosides herein can be linked to one another and to the remainder of the compound through cleavable bonds. Herein, linker-nucleosides are not considered to be part of the oligonucleotide.
- a compound comprises an oligonucleotide consisting of a specified number or range of linked nucleosides and/or a specified percent complementarity to a reference nucleic acid and the compound also comprises a conjugate group comprising a conjugate linker comprising linker-nucleosides
- those linker-nucleosides are not counted toward the length of the oligonucleotide and are not used in determining the percent complementarity of the oligonucleotide for the reference nucleic acid.
- conjugate groups and conjugate linkers as well as other modifications include, without limitation, those described in the following references: US 5,994,517; US 6,300,319; US 6,660,720; US 6,906,182; US 7,262,177; US 7,491,805; US 8,106,022; US 7,723,509; US 9,127,276; US 2006/0148740; US 2011/0123520; WO2013/033230; WO2012/037254, Biessen et al., J. Med. Chem. 1995, 38, 1846-1852; Lee et al., Bioorganic & Medicinal Chemistry 2011,19, 2494-2500; Rensen et al., J. Biol. Chem.
- a compound provided herein comprises a conjugate group.
- an oligonucleotide provided herein comprises a conjugate group.
- the conjugate group is a targeting moiety.
- the targeting moiety comprises one or more TrkB ligands.
- R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , R 15 , R 16 , R 17 , R 18 , R 19 , R 20 , R 21 , and R 22 are each independently optionally substituted unsaturated or partially unsaturated alkyl.
- R 7 , R 8 , R 9 , and R 10 are each independently alkenyl.
- R 7 , R 8 , R 9 , and R 10 are each independently alkynyl.
- R 2 is OR 7 .
- R 3 is OR 11 .
- R 7 and R 11 are each independently hydrogen, optionally substituted alkyl or optionally substituted alkenyl. In certain embodiments, one or both R 7 and R 11 are each independently hydrogen. In certain embodiments, one or both R 7 and R 11 are each independently optionally substituted alkyl. In certain embodiments, one or both R 7 and R 11 are each independently optionally substituted unsaturated or partially unsaturated alkyl. In certain embodiments, one or both R 7 and R 11 are each independently alkenyl. In certain embodiments, R 7 is optionally substituted alkyl and R 11 is hydrogen. In certain embodiments, R 7 is hydrogen and R 11 is optionally substituted alkyl. In certain embodiments, R 7 is alkenyl and R 11 is hydrogen.
- R 7 is hydrogen and R 11 is optionally substituted alkenyl.
- the TrkB ligand of a modified oligonucleotide is selected from the following Formulae or a salt, solvate, or hydrate thereof: Formula (II-A),
- R 1 is the modified oligonucleotide
- L 1 , L 2 , L 3 , L 4 , and R 1 are as described herein.
- the TrkB ligand of a modified oligonucleotide is of the Formula (XXXXVII) or a salt, solvate, or hydrate thereof: wherein: L 1 , L 2 , L 3 , L 4 , and R 1 are as described herein;
- R 11 and R 13 are each independently absent, hydrogen, or optionally substituted alkyl;
- R 12 , R 14 , and R 15 are each independently hydrogen, optionally substituted alkyl, optionally substituted heteroalkyl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl, optionally substituted aryl, or optionally substituted heteroaryl;
- R 16 is hydrogen, halogen, –CN, –N3, –SOn16R 1A , –SOv16NR 16
- the TrkB ligand of a modified oligonucleotide is of the Formula (XXXXIX) or a salt, solvate, or hydrate thereof: Formula (XXXXIX), wherein: L 1 , L 2 , L 3 , L 4 , and R 1 are as described herein; R 17 , R 18 , and R 19 are each independently hydrogen, optionally substituted alkyl, optionally substituted heteroalkyl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl, optionally substituted aryl, or optionally substituted heteroaryl; z4 is 0, 1, or 2; and z5 is 0, 1, 2, or 3.
- the TrkB ligand of a modified oligonucleotide is of the Formula (XXXXX) or a salt, solvate, or hydrate thereof: Formula (XXXXX), wherein: L 1 , L 2 , L 3 , L 4 , and R 1 are as described herein; R 20 is hydrogen, halogen, –CN, –N3, –SOn20R 1A , –SOv20NR 20B R 20C , ⁇ NHNR 20B R 20C , ⁇ ONR 20B R 20C , ⁇ NHC(O)NHNR 20B R 20C , ⁇ NHC(O)NR 20B R 20C , –N(O)m20, –NR 20B R 20C , –C(O)R 20D , –C(O)OR 20D , –C(O)NR 20B R 20C , –OR 20A , -NR 20B SO2R 20A ,
- the CB 1 ligand of a modified oligonucleotide is of the Formula (XXXXXI) or a salt, solvate, or hydrate thereof: , Formula (XXXXXI) wherein: L 1 , L 2 , L 3 , L 4 , and R 1 are as described herein;
- X 1 is NR 10 or CR 11 R 12 ;
- R 10 , R 11 , and R 12 are each independently hydrogen, optionally substituted alkyl, optionally substituted heteroalkyl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl, optionally substituted aryl, or optionally substituted heteroaryl;
- R 19 is hydrogen, –SOn19R 19A , –SOv19NR 19B R 19C , ⁇ NHNR 19B R 19C , ⁇ ONR 19B R 19C , ⁇ NHC(O)NHNR 19B R 19C , ⁇ NHC(O)NR 19B R
- the ⁇ 4 ⁇ 1/7 integrin ligand of a modified oligonucleotide is of the Formula (XXXXXXII) or a salt, solvate, or hydrate thereof: .
- Formula (XXXXXII) wherein L 1 , L 2 , L 3 , L 4 , and R 1 are as described herein; R 2 is H, polyethylene glycol (PEG), optionally substituted heteroalkyl, or optionally substituted heteroaryl; and R 3 , and R 4 are each independently H, halogen, optionally substituted alkyl, or optionally substituted -O-alkyl.
- the ⁇ 4 ⁇ 1/7 integrin ligand of a modified oligonucleotide is of the Formula (XXXXXXIII) or a salt, solvate, or hydrate thereof: .
- Formula (XXXXXIII) wherein L 1 , L 2 , L 3 , L 4 , and R 1 are as described herein; R 2 , R 3 , R 4 , and R 5 are each independently H, halogen, optionally substituted alkyl, optionally substituted -O-alkyl, cycloalkyl, or absent; R 8 is optionally substituted C 1 -C 5 alkyl, optionally substituted C 1 -C 5 alkylene-(C 3 - C 6 )-cycloalkyl, or optionally substituted (C 1 -C 4 )-alkylene-(C 1 -C 4 )-alkoxy; and R 6 , and R 7 are each independently H, halogen, alkyl, or optionally
- R 2 is H, -CONHR 4 , -CH 2 OR 4 , -(CH 2 ) 2 OR 4 , -CH 2 NHCOR 4 , or -OR 4 ;
- R 3 is H, optionally substituted alkyl, or optionally substituted cycloalkyl;
- R 4 is H, polyethylene glycol, optionally substituted alkyl, optionally substituted heteroalkyl, optionally substituted cycloalkyl, or optionally substituted heteroaryl;
- R 5 is -OH or absent;
- X is H, optionally substituted CH 2 , optionally substituted NH, or cycloalkyl.
- the ⁇ 4 ⁇ 1/7 integrin ligand of a modified oligonucleotide is of the Formula (XXXXXXXIII) or a salt, solvate, or hydrate thereof: .
- Formula (XXXXXXXIII) wherein L 1 , L 2 , L 3 , L 4 , and R 1 are as described herein; R 2 is H, -CONHR 3 , -CH 2 OR 3 , -(CH 2 ) 2 OR 3 , -CH 2 NHCOR 3 , or -OR 3 ; each instance of R 3 is independently H, polyethylene glycol, optionally substituted alkyl, optionally substituted heteroalkyl, optionally substituted cycloalkyl, or optionally substituted heteroaryl; and X is H or halogen.
- L 1 , L 2 , L 3 , and L 4 are each independently absent, a bond, an optionally substituted alkyl linker, an optionally substituted polyethylene glycol (PEG) linker, an optionally substituted heteroalkyl linker, or an optionally substituted heteroaryl linker.
- L 1 is an optionally substituted heteroaryl linker.
- L 1 is an optionally substituted unsaturated heteroaryl, an optionally substituted heteroaryl or an optionally substituted saturated or partially unsaturated heterocycloalkyl linker.
- L 1 comprises the structure: .
- L 1 is an optionally substituted heteroalkyl linker.
- the optionally substituted heteroalkyl linker is an optionally substituted heteroalkyl or optionally substituted C 1 -C 1 0 alkyl chain in which one or more carbon atoms are replaced with O, N, or S.
- L 1 comprises the structure: o .
- L 1 comprises the structure: –N(CH 3 )–.
- L 2 is an optionally substituted PEG linker.
- the PEG linker is five PEG units in length.
- the PEG linker is four PEG units in length.
- the PEG linker is three PEG units in length.
- L 2 is an optionally substituted alkyl linker.
- the heteroalkyl linker comprises two substituents joined together to form an optionally substituted carbocyclyl ring.
- L 4 comprises the structure: salt thereof, wherein X is O or S.
- L 4 comprises the structure: or a salt thereof, wherein X is O or S.
- L 1 – L 2 –L 3 –L 4 comprises the structure: ,
- TrkB ligand of a modified oligonucleotide is selected from the following Formulae or a salt, solvate, or hydrate thereof: Formula (IV),
- the CB 1 ligand of a modified oligonucleotide is selected from the following Formulae or a salt, solvate, or hydrate thereof: Formula (XXXXXXXVI)
- the ⁇ 4 ⁇ 1/7 integrin ligand of a modified oligonucleotide is selected from the following Formulae or a salt, solvate, or hydrate thereof: Formula (XXXXXXXIX)
- a compound provided herein comprises a conjugate group.
- an oligonucleotide provided herein comprises a conjugate group.
- the conjugate group is a lipid.
- an internucleoside linkage of a modified oligonucleotide provided herein comprises one or more lipids.
- R C is –H, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted heteroalkyl, substituted or unsubstituted aryl, or substituted or unsubstituted heteroaryl; each R 2 is independently –H, halogen, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, –OR 6 , -N(R 6 ), or -SR 6 ; each R 3 is independently –H, halogen, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, –OR 7 , -N(R 7 ), or -SR 7 ; R 4 and
- the modified oligonucleotide comprises Formula (XXXXII), or a salt, solvate, or hydrate thereof: mA ⁇ mA Formula (XXXXII) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXIII), or a salt, solvate, or hydrate thereof: mA ⁇ mU Formula (XXXXIII) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXIV), or a salt, solvate, or hydrate thereof: mA ⁇ mG Formula (XXXXIV) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXV), or a salt, solvate, or hydrate thereof: mA ⁇ mC Formula (XXXXV) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXVII), or a salt, solvate, or hydrate thereof: mU ⁇ mG Formula (XXXXVII) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXVIII), or a salt, solvate, or hydrate thereof: mU ⁇ fC Formula (XXXXVIII) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXX), or a salt, solvate, or hydrate thereof: mG ⁇ fG Formula (XXXX) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXXI), or a salt, solvate, or hydrate thereof: mG ⁇ mC Formula (XXXXI) wherein: R 4 and R 5 are independently an oligonucleotide, or R 4 and R 5 are joined together to form a single oligonucleotide; and each X is independently O or S.
- the modified oligonucleotide comprises Formula (XXXXXII), or a salt, solvate, or hydrate thereof:
- the modified oligonucleotide comprises an internucleoside linkage of one of the following Formulae: Formula (XXXXXV), Formula (XXXXXVI).
- the compound of any preceding embodiment comprises one or more lipid conjugate groups.
- the one or more lipid conjugate groups are attached to one or more internucleoside linkages of the modified oligonucleotide.
- the one or more lipid conjugate groups are attached to the 5’ or 3’ end of the modified oligonucleotide. In certain embodiments, the one or more lipid conjugate groups are attached to an internucleoside linkage and the 5’ or 3’ end of the modified oligonucleotide. In certain embodiments, the one or more lipid conjugate groups are attached to an internucleoside linkage and both the 5’ and 3’ ends of the modified oligonucleotide.
- the one or more ligands are attached to the 5’ or 3’ end of the modified oligonucleotide or both the 5’ and 3’ ends of the modified oligonucleotide.
- the one or more conjugate groups comprise at least one ligand (e.g., at least one TrkB ligand, at least one CB 1 ligand, at least one ⁇ 4 ⁇ 1/7 integrin ligand) attached to the 5’ or 3’ end of the modified oligonucleotide or both the 5’ and 3’ ends of the modified oligonucleotide and at least one lipid.
- at least one ligand e.g., at least one TrkB ligand, at least one CB 1 ligand, at least one ⁇ 4 ⁇ 1/7 integrin ligand
- the one or more conjugate groups comprise at least one ligand (e.g., at least one TrkB ligand, at least one CB 1 ligand, at least one ⁇ 4 ⁇ 1/7 integrin ligand) attached to the 5’ or 3’ end of the modified oligonucleotide or both the 5’ and 3’ ends of the modified oligonucleotide and one or more lipid conjugate groups attached to one or more internucleoside linkages of the modified oligonucleotide.
- ligand e.g., at least one TrkB ligand, at least one CB 1 ligand, at least one ⁇ 4 ⁇ 1/7 integrin ligand
- the modified oligonucleotide comprises a ligand (e.g., a TrkB ligand, a CB 1 ligand, an ⁇ 4 ⁇ 1/7 integrin ligand) and a lipid.
- the modified oligonucleotide comprises one or more ligands (e.g., one or more TrkB ligands, one or more CB 1 ligands, one or more ⁇ 4 ⁇ 1/7 integrin ligands) and one or more lipids.
- the modified oligonucleotide is the second modified oligonucleotide or sense oligonucleotide.
- the compound of any preceding embodiment comprises one or more substituted or unsubstituted alkyl or alkenyl.
- the substituted or unsubstituted alkyl or alkenyl is attached to an internucleoside linkage of a modified oligonucleotide.
- the modified oligonucleotide comprises one or more substituted or unsubstituted alkyl or alkenyl.
- the one or more substituted or unsubstituted alkyl or alkenyl are attached to one or more internucleoside linkages of the modified oligonucleotide.
- the modified oligonucleotide is the second modified oligonucleotide or sense oligonucleotide.
- the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 4 -C 30 hydrocarbon chain.
- the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 5 -C 20 hydrocarbon chain.
- the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 14 -C 20 hydrocarbon chain.
- the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 16 hydrocarbon chain. In certain embodiments, the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 17 hydrocarbon chain. In certain embodiments, the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 18 hydrocarbon chain. In certain embodiments, the one or more substituted or unsubstituted alkyl or alkenyl comprise a saturated or unsaturated C 22 hydrocarbon chain.
- a substituted or unsubstituted alkyl or alkenyl is attached to an internucleoside linkage of a modified oligonucleotide (e.g., a second modified oligonucleotide or sense oligonucleotide).
- a substituted or unsubstituted alkyl or alkenyl is attached to an internucleoside linkage of a modified oligonucleotide (e.g., a second modified oligonucleotide or sense oligonucleotide).
- the internucleoside linkage is between nucleosides that are within 10 positions (e.g., within 8 positions, within 6 positions, within 5 positions, within 4 positions, within 3 positions, within 2 positions) from a terminal end (e.g., the 5′ and/or 3′ end) of the modified oligonucleotide. In certain embodiments, the internucleoside linkage is between nucleosides that are within 5 positions from the 5′ end of the modified oligonucleotide. In certain embodiments, the internucleoside linkage is between nucleosides that are within 5 positions from the 3′ end of the modified oligonucleotide.
- the internucleoside linkage is between positions 1 and 2, positions 2 and 3, positions 3 and 4, positions 4 and 5, positions 5 and 6, positions 6 and 7, positions 7 and 8, positions 8 and 9, positions 9 and 10, positions 10 and 11, positions 11 and 12, positions 12 and 13, or positions 13 and 14 from the 5′ end of the modified oligonucleotide. In certain embodiments, the internucleoside linkage is between positions 1 and 2, positions 2 and 3, positions 3 and 4, positions 4 and 5, positions 5 and 6, positions 6 and 7, or positions 7 and 8 from the 5′ end of the modified oligonucleotide. In certain embodiments, the internucleoside linkage is between positions 2 and 3 from the 5′ end of the modified oligonucleotide.
- the internucleoside linkage is between positions 1 and 2, positions 2 and 3, positions 3 and 4, positions 4 and 5, positions 5 and 6, positions 6 and 7, positions 7 and 8, positions 8 and 9, positions 9 and 10, positions 10 and 11, positions 11 and 12, positions 12 and 13, or positions 13 and 14 from the 3′ end of the modified oligonucleotide. In certain embodiments, the internucleoside linkage is between positions 1 and 2, positions 2 and 3, positions 3 and 4, positions 4 and 5, positions 5 and 6, positions 6 and 7, or positions 7 and 8 from the 3′ end of the modified oligonucleotide. In certain embodiments, the internucleoside linkage is between positions 2 and 3 from the 3′ end of the modified oligonucleotide.
- the internucleoside linkage of the modified oligonucleotide is selected from any one of Formulae XXXXIII-XXXXVI.
- the modified oligonucleotide comprises any one of Formulae XXXV-XXXXVI.
- Target Nucleic Acids and Target Regions compounds described herein comprise or consist of an oligonucleotide comprising a region that is complementary to a target nucleic acid.
- the target nucleic acid is an endogenous RNA molecule.
- the target nucleic acid encodes a protein.
- the target nucleic acid is non-coding.
- the target nucleic acid is selected from an mRNA and a pre-mRNA, including intronic, exonic and untranslated regions.
- the target RNA is an mRNA.
- the target nucleic acid is a pre-mRNA.
- the target region is entirely within an exon.
- the target region is entirely within an intron.
- the target region spans an intron/exon junction.
- the target region is at least 50% within an intron.
- compounds disclosed herein hybridize with a MAPT nucleic acid. The most common mechanism of hybridization involves hydrogen bonding between complementary nucleobases of the nucleic acid molecules.
- Hybridization can occur under varying conditions. Hybridization conditions are sequence-dependent and are determined by the nature and composition of the nucleic acid molecules to be hybridized. Methods of determining whether a sequence hybridizes specifically to a target nucleic acid are well known in the art. In certain embodiments, the compounds provided herein specifically hybridize with a MAPT nucleic acid. Nucleotide sequences that encode MAPT include, without limitation, the following: GenBank Accession No. NM_001377265.1 (incorporated herein as SEQ ID NO: 1), and nucleotides 2624000 to 2761000 of NT_010783.14 (incorporated herein as SEQ ID NO: 2).
- Complementarity Oligonucleotides provided herein may have a defined percent complementarity to a particular nucleic acid, target region, oligonucleotide, or portion thereof. Non-complementary nucleobases may be tolerated provided that the oligonucleotide remains able to specifically hybridize to the nucleic acid, oligonucleotide, or portion thereof.
- the oligonucleotides provided herein, or a specified portion thereof are at least, or are up to 70%, 80%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% complementary to a target nucleic acid, a target region, an oligonucleotide or specified portion thereof.
- the oligonucleotides provided herein, or a specified portion thereof are 70% to 75%, 75% to 80%, 80% to 85%, 85% to 90%, 90% to 95%, 95% to 100%, or any number in between these ranges, complementary to a target nucleic acid, a target region, an oligonucleotide or specified portion thereof. Percent complementarity of an oligonucleotide with a target nucleic acid, a target region, an oligonucleotide or specified portion thereof can be determined using routine methods.
- an oligonucleotide in which 18 of 20 nucleobases of the oligonucleotide are complementary to a target region, and would therefore specifically hybridize would represent 90 percent complementarity.
- the remaining non-complementary nucleobases may be clustered or interspersed with complementary nucleobases and need not be contiguous to each other or to complementary nucleobases.
- an oligonucleotide which is 18 nucleobases in length having four non-complementary nucleobases which are flanked by two regions of complete complementarity with the target nucleic acid would have 77.8% overall complementarity with the target nucleic acid.
- Percent complementarity of an oligonucleotide with a region of a target nucleic acid, a target region, an oligonucleotide or specified portion thereof can be determined routinely using BLAST programs (basic local alignment search tools) known in the art.
- oligonucleotides described herein, or specified portions thereof are fully complementary (i.e. 100% complementary) to a target nucleic acid, a target region, an oligonucleotide or specified portion thereof.
- an oligonucleotide may be fully complementary to a target nucleic acid, a target region, an oligonucleotide, or specified portion thereof.
- each nucleobase of an oligonucleotide is complementary to the corresponding nucleobase of a target nucleic acid, a target region, an oligonucleotide, or a specified portion thereof.
- a 20 nucleobase oligonucleotide is fully complementary to a target sequence that is 400 nucleobases long, so long as there is a corresponding 20 nucleobase portion of the target nucleic acid that is fully complementary to the compound.
- “Fully complementary” can also be used in reference to a specified portion of the first and/or the second nucleic acid.
- a 20 nucleobase portion of a 30 nucleobase oligonucleotide can be “fully complementary” to a 20 nucleobase region of a target sequence that is 400 nucleobases long.
- the 20 nucleobase portion of the 30 nucleobase compound is fully complementary to the target sequence if the target sequence has a corresponding 20 nucleobase portion wherein each nucleobase is complementary to the 20 nucleobase portion of the compound.
- the entire 30 nucleobase compound may or may not be fully complementary to the target sequence, depending on whether the remaining 10 nucleobases of the compound are also complementary to the target sequence.
- oligonucleotides described herein comprise one or more mismatched nucleobases relative to a target nucleic acid, a target region, an oligonucleotide or a specified portion thereof.
- oligonucleotides described herein that are, or are up to 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23 nucleobases in length comprise no more than 4, no more than 3, no more than 2, or no more than 1 non- complementary nucleobase(s) relative to a target nucleic acid, or specified portion thereof.
- oligonucleotides described herein that are, or are up to 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, or 30 nucleobases in length comprise no more than 6, no more than 5, no more than 4, no more than 3, no more than 2, or no more than 1 non-complementary nucleobase(s) relative to a target nucleic acid, a target region, an oligonucleotide, or specified portion thereof.
- the mismatch is at position 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14 from the 5’-end of the oligonucleotide.
- the mismatch is at position 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12, 13 or 14 from the 3’-end of the oligonucleotide.
- the mismatch forms a wobble base pair with a corresponding nucleobase on the target nucleic acid.
- the mismatch forms a wobble base pair selected from hypoxanthine (nucleobase of inosine) and uracil (I:U base pair); guanine and uracil (G:U base pair); hypoxanthine and adenine (I:A base pair); and hypoxanthine and cytosine (I:C base pair).
- a mismatched nucleobase on an oligonucleotide comprises hypoxanthine, guanine, or uracil.
- oligonucleotides described herein may be complementary to a portion of a nucleic acid.
- portion refers to a defined number of contiguous nucleobases within a region of a nucleic acid.
- a “portion” can also refer to a defined number of contiguous nucleobases of an oligonucleotide.
- the oligonucleotides are complementary to at least an 8 nucleobase portion of a nucleic acid.
- the oligonucleotides are complementary to at least a 9 nucleobase portion of a nucleic acid. In certain embodiments, the oligonucleotides are complementary to at least a 10 nucleobase portion of a nucleic acid. In certain embodiments, the oligonucleotides are complementary to at least an 11 nucleobase portion of a nucleic acid. In certain embodiments, the oligonucleotides are complementary to at least a 12 nucleobase portion of a nucleic acid. In certain embodiments, the oligonucleotides are complementary to at least a 13 nucleobase portion of a nucleic acid.
- the oligonucleotides are complementary to at least a 14 nucleobase portion of a nucleic acid. In certain embodiments, the oligonucleotides are complementary to at least a 15 nucleobase portion of a nucleic acid. In certain embodiments, the oligonucleotides are complementary to at least a 16 nucleobase portion of a nucleic acid. Also contemplated are oligonucleotides that are complementary to at least a 9, 10, 17, 18, 19, 20, 21, 22, 23 or more nucleobase portion of a nucleic acid, or a range defined by any two of these values. In certain embodiments, the oligonucleotide is an antisense oligonucleotide.
- a portion of the antisense oligonucleotide is compared to an equal length portion of the target nucleic acid. In certain embodiments, an 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, or 25 nucleobase portion is compared to an equal length portion of the target nucleic acid.
- the oligonucleotide is a sense oligonucleotide. In certain embodiments, a portion of the sense oligonucleotide is compared to an equal length portion of an antisense oligonucleotide.
- an 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, or 25 nucleobase portion of a sense oligonucleotide is compared to an equal length portion of an antisense oligonucleotide.
- Identity The oligonucleotides provided herein may also have a defined percent identity to a particular nucleic acid, target region, oligonucleotide, or specified portion thereof. As used herein, an oligonucleotide is identical to a sequence disclosed herein if it has the same nucleobase pairing ability.
- RNA which contains thymidine in place of uracil in a disclosed RNA sequence would be considered identical to the RNA sequence since both uracil and thymidine pair with adenine.
- Shortened and lengthened versions of the compounds described herein as well as compounds having non-identical bases relative to the compounds provided herein also are contemplated.
- the non-identical bases may be adjacent to each other or dispersed throughout the compound. Percent identity of an oligonucleotide is calculated according to the number of bases that have identical base pairing relative to the sequence to which it is being compared.
- oligonucleotides described herein, or portions thereof are, or are at least, 70%, 75%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% or 100% identical to one or more of the nucleic acids, oligonucleotides, or a portion thereof, disclosed herein. In certain embodiments, oligonucleotides described herein are about 70%, 75%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identical, or any percentage between such values, to a particular nucleic acid or oligonucleotide, or portion thereof.
- an oligonucleotide may have one or more mismatched nucleobases.
- the mismatch is at position 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14 from the 5’-end of the oligonucleotide.
- the mismatch is at position 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12, 13 or 14 from the 3’-end of the oligonucleotide.
- a portion of the oligonucleotide is compared to an equal length portion of the target nucleic acid.
- an 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, or 25 nucleobase portion is compared to an equal length portion of the target nucleic acid.
- the oligonucleotide is a sense oligonucleotide. In certain embodiments, a portion of the sense oligonucleotide is compared to an equal length portion of the target nucleic acid. In certain embodiments, an 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, or 25 nucleobase portion is compared to an equal length portion of the target nucleic acid.
- compositions and Formulations Compounds described herein may be admixed with pharmaceutically acceptable active or inert substances for the preparation of pharmaceutical compositions or formulations. Compositions and methods for the formulation of pharmaceutical compositions are dependent upon a number of criteria, including, but not limited to, route of administration, extent of disease, or dose to be administered. Certain embodiments provide pharmaceutical compositions comprising one or more compounds or a salt thereof. In certain embodiments, the compounds are antisense oligonucleotides. In certain embodiments, the compounds are oligomeric compounds. In certain embodiments, the compounds comprise or consist of one or more modified oligonucleotides. In certain such embodiments, the pharmaceutical composition comprises one or more compound and a suitable pharmaceutically acceptable diluent or carrier.
- a pharmaceutical composition comprises one or more compound and a sterile saline solution. In certain embodiments, such pharmaceutical composition consists of one compound and a sterile saline solution. In certain embodiments, the sterile saline is pharmaceutical grade saline. In certain embodiments, a pharmaceutical composition comprises one or more compound and sterile water. In certain embodiments, a pharmaceutical composition consists of one compound and sterile water. In certain embodiments, the sterile water is pharmaceutical grade water. In certain embodiments, a pharmaceutical composition comprises one or more compounds and phosphate-buffered saline (PBS). In certain embodiments, a pharmaceutical composition consists of one compound and sterile PBS.
- PBS phosphate-buffered saline
- the sterile PBS is pharmaceutical grade PBS.
- a compound described herein targeted to MAPT can be utilized in pharmaceutical compositions by combining the compound with a suitable pharmaceutically acceptable diluent or carrier.
- a pharmaceutically acceptable diluent is water, such as sterile water suitable for injection.
- employed in the methods described herein is a pharmaceutical composition comprising a compound targeted to MAPT and a pharmaceutically acceptable diluent.
- the pharmaceutically acceptable diluent is water.
- the compound comprises or consists of one or more modified oligonucleotide provided herein.
- compositions comprising compounds provided herein encompass any pharmaceutically acceptable salts, esters, or salts of such esters, or any other oligonucleotide which, upon administration to an animal, including a human, is capable of providing (directly or indirectly) the biologically active metabolite or residue thereof.
- the compounds are antisense oligonucleotides.
- the compounds are oligomeric compounds.
- the compound comprises or consists of one or more modified oligonucleotide. Accordingly, for example, the disclosure is also drawn to pharmaceutically acceptable salts of compounds, prodrugs, pharmaceutically acceptable salts of such prodrugs, and other bioequivalents.
- Suitable pharmaceutically acceptable salts include, but are not limited to, sodium and potassium salts.
- a prodrug can include the incorporation of additional nucleosides at one or both ends of a compound which are cleaved by endogenous nucleases within the body, to form the active compound.
- the compounds or compositions further comprise a pharmaceutically acceptable carrier or diluent.
- RNA Ribonucleic acid
- DNA DNA having a modified sugar (2’-OH for the natural 2’-H of DNA) or as an RNA having a modified base (methylated uracil for natural uracil of RNA).
- nucleic acid sequences provided herein are intended to encompass nucleic acids containing any combination of natural or modified RNA and/or DNA, including, but not limited to, such nucleic acids having modified nucleobases.
- Each of the references recited in the present application is incorporated herein by reference in its entirety.
- Example 1 Preparation of N-((3s,5s,7s)-adamantan-1-yl)-4-((17-azido-3,6,9,12,15- pentaoxaheptadecyl)oxy)-6-(4-methylpiperidin-1-yl)-1,3,5-triazin-2-amine (BA-120) STEP 1: 2-((17-azido-3,6,9,12,15-pentaoxaheptadecyl)oxy)-4,6-dichloro-1,3,5-triazine To a suspension of 2,4,6-trichloro-1,3,5-triazine (1.0 g, 5.42 mmol) and NaHCO 3 (911 mg, 10.84 mmol) in acetone (8 mL) at 0°C was added 17-azido-3,6,9,12,15- pentaoxaheptadecan-1-ol (1.67 g, 5.42 mmol
- STEP 2 N-((3s,5s,7s)-adamantan-1-yl)-4-((17-azido-3,6,9,12,15-pentaoxaheptadecyl)oxy)- 6-chloro-1,3,5-triazin-2-amine
- 2-((17-azido-3,6,9,12,15-pentaoxaheptadecyl)oxy)-4,6-dichloro- 1,3,5-triazine 500 mg, 1.1 mmol
- THF 9.5 mL
- DIEA 0.29 mL, 1.65 mmol
- STEP 3 N-((3s,5s,7s)-adamantan-1-yl)-4-((17-azido-3,6,9,12,15-pentaoxaheptadecyl)oxy)- 6-(4-methylpiperidin-1-yl)-1,3,5-triazin-2-amine
- N-((3s,5s,7s)-adamantan-1-yl)-4-((17-azido-3,6,9,12,15- pentaoxaheptadecyl)oxy)-6-chloro-1,3,5-triazin-2-amine 50 mg, 0.088 mmol
- 4-methylpiperidine (16 mL, 0.13 mmol
- DIEA 18 mL, 0.10 mmol
- reaction mixture was stirred at room temperature for 16 h then quenched by addition of water (10 ml) and extracted with DCM (3x10 ml). The organic layer was washed with a saturated solution of NaHCO 3 (10 ml) and brine (10 ml). The combined organic extracts were dried over anhydrous Na 2 SO 4 and concentrated to obtain the title compound as a brown oil (0.11 g, quant.).
- reaction mixture was stirred at room temperature under inert atmosphere for 2 hours and then diluted with saturated ammonium chloride solution (15 ml) and extracted with ethyl acetate (3 x 15 ml). The combined organic extracts were washed with brine (10 mL), dried over anhydrous Na 2 SO 4 , filtered, concentrated and purified by column chromatography using 0-15 % MeOH in DCM to provide the title product as a clear oil (0.12 mg, 55%).
- reaction mixture was stirred at 0°C for 2 hours and then diluted with saturated aqueous sodium bicarbonate (10 ml) and extracted with DCM (3 x 20 ml). The combined organic extracts were washed with brine (10 ml), dried over anhydrous Na 2 SO 4 , filtered and concentrated under reduced pressure to obtain the title compound as a brown oil (0.11 g, 83%).
- reaction mixture was heated at 65°C for 2 hours and then diluted with water (15 ml) and extracted with ethyl acetate (3 x 15 ml). The combined organic extracts were washed with brine (10 ml), dried over anhydrous Na 2 SO 4, filtered, concentrated and purified by silica gel column chromatography using a gradient 0-15% MeOH in DCM to provide the title compound as a clear oil (45 mg, 44%).
- reaction was neutralized and quenched with glacial acetic acid and then extracted with ethyl acetate (3 x 100 mL). The combined organic extracts were washed with brine (3 x 100 mL), dried over anhydrous Na 2 SO 4 , filtered, concentrated and purified by reversed-phase flash chromatography (column, C 1 8 silica gel; mobile phase, acetonitrile in water, 10 % to 100 % gradient in 20 min; detector, UV 254 nm.) to afford the title compound (484 mg, 26 %) as a light yellow solid.
- the reaction mixture was diluted with ethyl acetate (50 mL) and washed with water (2 x 100 mL). The organic layer was washed with sat. NaHCO 3 (50 mL) and brine (50 mL), dried over Na 2 SO 4 , filtered, concentrated and purified and by silica gel chromatography (0 to 100% ethyl acetate in hexane, followed by 0 to 40% methanol in ethyl acetate, 12g Claricep column) to afford the title compound (231 mg, 90%) as a clear oil.
- STEP 4 3-hydroxy-1-(6-((2,2,3,3-tetramethyl-4,7,10,13,16,19-hexaoxa-3-silahenicosan-21- yl)oxy)naphthalen-1-yl)butan-1-one
- a solution of 1-(6-((2,2,3,3-tetramethyl-4,7,10,13,16,19-hexaoxa-3-silahenicosan-21- yl)oxy) naphthalen-1-yl)ethan-1-one (6.5 g, 11 mmol) in THF (260 mL) was treated with LiHMDS (15.9 mL, 0.796 mmol) for 10 min at -40°C under nitrogen atmosphere followed by the addition of CH 3 CHO (4.2 mL, 44 mmol) in THF dropwise in portions at -40 °C.
- reaction mixture was stirred at room temperature under inert atmosphere for 3 hours, then diluted with water (20 ml) and extracted with DCM (3 x 20 ml). The combined organic extracts were washed with brine (10 ml), dried over anhydrous Na 2 SO 4 , concentrated and purified by silica gel column chromatography using a gradient 50-100% EtOAc in hexane to afford the title compound (0.20 g, 57%) as a yellow oil.
- reaction mixture was concentrated under reduced pressure, treated with citric acid solution (10%) and extracted with DCM (3 x 20 ml). The combined organic extracts were washed with brine (5 ml), dried over anhydrous Na 2 SO 4 and concentrated to afford the title compound (0.19 g, 97%) as a white solid.
- Example 7 Preparation of (S)-2-(1-(4-(2-(4-(3-(4-(17-azido-3-oxo-6,9,12,15-tetraoxa-2- azaheptadecyl)phenyl) ureido) phenyl)acetyl)morpholine-3-carbonyl)piperidin-4- yl)acetic acid (BA-215)
- STEP 1 methyl (S)-2-(1-(4-(2-(4-(3-(4-(((tert-butoxycarbonyl)amino)methyl)phenyl)ureido) phenyl)acetyl) morpholine-3-carbonyl)piperidin-4-yl)acetate
- 2-(4-(3-(4-(((tert- butoxycarbonyl)amino)methyl)phenyl)ureido)phenyl)acetic acid (0.15 g, 0.36
- reaction mixture was stirred at room temperature under inert atmosphere for 3 hours and then diluted with water (20 ml) and extracted with DCM (3 x 20 ml). The combined organic extracts were washed with brine (15 ml), dried over anhydrous Na 2 SO 4 , filtered, concentrated and purified by silica gel column chromatography using a gradient 0- 20% MeOH in DCM to afford the title compound (0.11 g, 48%) as a yellow oil.
- Example 12 Preparation of N1-(2-(2-(2-(2-azidoethoxy)ethoxy)ethoxy)ethyl)-N3,N5- bis(2-hydroxyethyl)benzene-1,3,5-tricarboxamide (BA-144)
- STEP 1 tert-butyl (2-((2-oxoazepan-3-yl)carbamoyl)phenyl)carbamate
- 2-((tert-butoxycarbonyl)amino)benzoic acid (2.01 g, 8.4 mmol) and 3-aminoazepan-2-one (1.19 g, 9.27 mmol, 1.1 equiv.) in DMF (10 mL) was added DIPEA (3.0 mL, 16.9 mmol, 2 equiv.) and HATU (4.8 g, 12.6 mmol, 1.5 equiv.).
- the reaction mixture was stirred at 20 °C for 18h during which time an off-white precipitate formed.
- the mixture was poured into water (200 mL) and sonicated with swirling for 5-10 min.
- the precipitate was isolated by filtration, washed with water and air-drying to afford the title product (340 mg, 33.5%) as a gray powder.
- STEP 5 5-(1-azido-3,6,9,12-tetraoxapentadecan-15-amido)-N-(2-((2-oxoazepan-3- yl)carbamoyl)phenyl)benzo[b]thiophene-2-carboxamide
- To a mixture of 5-amino-N-(2-((2-oxoazepan-3- yl)carbamoyl)phenyl)benzo[b]thiophene-2-carboxamide (0.16 g, 0.4 mmol, 1.5 eq) in dry DMA (2 mL) was added DIPEA (0.13 mL, 0.77 mmol, 3 eq) and 2,5-dioxopyrrolidin-1-yl 1- azido-3,6,9,12-tetraoxapentadecan-15-oate (0.1 g, 0.26 mmol).
- Example 15 Preparation of N-(2-(2-(2-(2-azidoethoxy)ethoxy)ethoxy)ethyl)-4-(7,8- dihydroxy-4-oxochroman-2-yl)benzamide (BA-167)
- a suspension of 2,5-dioxopyrrolidin-1-yl 4-(7,8-dihydroxy-4-oxochroman-2- yl)benzoate (665 mg, 1.684 mmol, 1 eq) and 2-(2-(2-(2-azidoethoxy)ethoxy)ethan-1- amine (440 mg, 2 mmol, 1.2 eq) and DIPEA (0.5 ml, 3.7 mmol, 2.2 eq) in THF (20 mL) was stirred for 3h at 60°C.
- Example 17 Preparation of (Z)-1-(3-((3-((1H-pyrrol-2-yl)methylene)-2-oxoindolin-6- yl)amino)-4-methylphenyl)-3-(3-(2-(2-(2-(2-azidoethoxy)ethoxy)ethoxy)-2-fluoro- 5-(trifluoromethyl)phenyl)urea (BA-173)
- STEP 1 methyl 3-(benzyloxy)-2-fluoro-5-(trifluoromethyl)benzoate
- To a solution of 3-(benzyloxy)-2-fluoro-5-(trifluoromethyl)benzoic acid (4.9 g, 15.593 mmol, 1 eq) in methanol (49.00 mL, 1210.243 mmol, 77.61 eq) was added two drop of H 2 SO 4 (305.85 mg, 3.119 mmol, 0.2 eq) at room temperature under nitrogen atmosphere.
- reaction was quenched by the addition of water (5 mL) and extracted with ethyl acetate (3 x 50 mL). The combined organic extracts were washed with brine (3 x 50 mL), dried over anhydrous Na 2 SO 4 , filtered, concentrated and purified by silica gel column chromatography, eluted with EA/PE ( ⁇ 30%) to afford the title compound (740 mg) as a yellow oil.
- the filtrate was concentrated and purified by reversed-phase flash chromatography: column, C 1 8 silica gel; mobile phase, ACN in water, 10% to 95% gradient in 30 min; detector, UV 254 nm, to afford the title compound (1 g, 70.9%) as a yellow oil.
- Example 18 Preparation of 2-(17-azido-3,6,9,12,15-pentaoxaheptadecyl)-8-(4- (methylamino)phenyl)chromeno[7,8-d]imidazol-6(3H)-one (BA-183)
- STEP 1 methyl 4-(benzyl(methyl)amino)benzoate
- bromomethylbenzene (12.42 g, 72.64 mmol, 8.63 mL, 1.2 eq) and K 2 CO 3 (25.10 g, 181.61 mmol, 3 eq) in DMF (100 mL) was stirred at 80°C for 2 hours.
- reaction mixture was stirred at 25 °C for 8 hr and then quenched by addition of water (30mL) and partitioned between ethyl acetate (50 mL) and brine (20mL). The aqueous phase was further extracted with ethyl acetate (2 ⁇ 40 mL) and the combined organic extracts were washed with brine (2 ⁇ 50 mL), dried over Na 2 SO 4 , filtered and purified by flash silica gel chromatography (ISCO®; 20 g SepaFlash® Silica Flash Column, Eluent of 0 ⁇ 30% Ethyl acetate/Petroleum ether gradient @ 50 mL/min) to afford the title compound (960 mg,69.0%) as a yellow oil.
- ISCO® 20 g SepaFlash® Silica Flash Column, Eluent of 0 ⁇ 30% Ethyl acetate/Petroleum ether gradient @ 50 mL/min
- STEP 7 1-phenyl-2,5,8,11,14,17-hexaoxaicosan-20-oic acid
- a solution of tert-butyl 1-phenyl-2,5,8,11,14,17-hexaoxaicosan-20-oate (640 mg, 1.40 mmol, 1 eq) in HCl/dioxane (3 mL) was stirred at 25°C for 0.5 hr.
- the reaction mixture was concentrated under reduced pressure to afford the title compound (600 mg, crude) as a yellow oil which was used for next step without further purification.
- STEP 12 2-(17-azido-3,6,9,12,15-pentaoxaheptadecyl)-8-(4- (methylamino)phenyl)chromeno [7,8-d]imidazol-6(3H)-one 2-(17-chloro-3,6,9,12,15-pentaoxaheptadecyl)-8-(4-(methylamino)phenyl)chromeno [7,8-d]imidazol-6(3H)-one (57 mg, 0.099 mmol, 1 eq), and sodium azide (32 mg, 0.497 mmol, 5 eq) in dry DMF (1 mL) were heated at 70°C for 12 h.
- Example 19 Preparation of N-(17-azido-3,6,9,12,15-pentaoxaheptadecyl)-4-(8-hydroxy- 4-methoxyquinolin-2-yl)benzamide (BA-225) STEP 1: methyl 4-(8-hydroxy-4-methoxyquinolin-2-yl)benzoate To a solution of 1-(2-amino-3-hydroxyphenyl)ethan-1-one (2.15g, 14.2 mmol) in methanol (30 mL), was added methyl 4-formylbenzoate (7.0 g, 43 mmol) followed by the sulfuric acid (650 mL, 12 mmol) and the mixture heated at reflux for 48 hours.
- HATU (1.03g, 2.72 mmol) and N 3 -PEG 5 -NH 2 (686 mg, 2.24 mmol) were added at RT, followed by the addition of DIPEA (0.9 mL, 4.8 mmol) and stirred for 30 minutes.
- the reaction mixture was diluted with water and extracted with DCM.
- the combined organic extracts were dried over Na 2 SO 4 , filtered, concentrated and purified by flash column chromatography (column: Biotage sfar silica HCD Duo 5, 20 micron, 50g) eluted with MeOH:DCM (0 to 20%, v/v, 10CV) to afford the title compound (770 mg, 83%) as a brown solid.
- Example 21 Preparation of 8-(4-((17-azido-3,6,9,12,15- pentaoxaheptadecyl)(methyl)amino)phenyl)chromeno[7,8-d]imidazol-6(3H)-one (BA- 169) STEPS 1-4 STEP 1: 1-phenyl-2,5,8,11,14,17-hexaoxanonadecan-19-yl 4-methylbenzenesulfonate To a solution of 2-[2-[2-[2-[2-[2-(2- benzyloxyethoxy)ethoxy]ethoxy]ethoxy]ethanol (5 g, 13.42 mmol, 1 eq) in DCM (100 mL) was added TEA (2.72 g, 26.85 mmol, 3.74 mL, 2 eq), 4-toluenesulfonyl chloride (2.82 g, 14.77 mmol, 1.1 eq) and D
- STEP 2 ethyl 4-((1-phenyl-2,5,8,11,14,17-hexaoxanonadecan-19-yl)amino)benzoate
- 1-phenyl-2,5,8,11,14,17-hexaoxanonadecan-19-yl 4- methylbenzenesulfonate (3.25 g, 6.17 mmol, 1 eq) and ethyl 4-aminobenzoate (1.02 g, 6.17 mmol, 1.29 mL, 1 eq) in DMF (10 mL) was added K 2 CO 3 (2.56 g, 18.51 mmol, 3 eq) and KI (1.02 g, 6.17 mmol, 1 eq).
- STEP 3 ethyl 4-(methyl(1-phenyl-2,5,8,11,14,17-hexaoxanonadecan-19-yl)amino)benzoate
- ethyl 4-((1-phenyl-2,5,8,11,14,17-hexaoxanonadecan-19- yl)amino)benzoate (1 g, 1.92 mmol, 1 eq) in THF (10 mL) was added NaH (115.46 mg, 2.89 mmol, 60% purity, 1.5 eq) at 0°C under nitrogen atmosphere.
- STEP 7 1-(4-amino-2-(benzyloxy)-3-nitrophenyl)ethan-1-one
- a solution of 1-(2-(benzyloxy)-4-fluoro-3-nitrophenyl)ethan-1-one (11.5 g, 39.76 mmol, 1 eq) in acetonitrile (100 mL) was added NH 3 ⁇ H 2 O (83.60 g, 596.35 mmol, 91.87 mL, 25% purity, 15 eq) dropwise at 25°C.
- the mixture was heated at 50°C for 2 hours and then concentrated to afford the title compound (8.5 g, crude) as a yellow oil which was used for next step without further purification.
- STEP 11 N-(3-hydroxy-4-(3-(4-(methyl(1-phenyl-2,5,8,11,14,17-hexaoxanonadecan-19- yl)amino)phenyl)-3-oxopropanoyl)-2-nitrophenyl)acetamide
- 3-acetamido-6-acetyl-2-nitrophenyl 4-(methyl(1-phenyl- 2,5,8,11,14,17-hexaoxanonadecan-19-yl)amino)benzoate (1 g, 1.38 mmol, 1 eq) in pyridine (15 mL) was added KOH (386.52 mg, 6.89 mmol, 5 eq) and the mixture was stirred at 60°C for 2 hr.
- STEP 12 7-amino-2-(4-((17-hydroxy-3,6,9,12,15- pentaoxaheptadecyl)(methyl)amino)phenyl)-8-nitro-4H-chromen-4-one
- N-(3-hydroxy-4-(3-(4-(methyl(1-phenyl-2,5,8,11,14,17- hexaoxanonadecan-19-yl)amino)phenyl)-3-oxopropanoyl)-2-nitrophenyl)acetamide (1.3 g, 1.79 mmol, 1 eq) in acetic acid (20 mL) was added H 2 SO 4 (175.68 mg, 1.79 mmol, 95.48 ⁇ L, 1 eq).
- STEP 13 7,8-diamino-2-(4-((17-hydroxy-3,6,9,12,15-pentaoxaheptadecyl) (methyl)amino)phenyl)-4H-chromen-4-one
- 7-amino-2-(4-((17-hydroxy-3,6,9,12,15-pentaoxaheptadecyl) (methyl)amino)phenyl)-8-nitro-4H-chromen-4-one (1 g, 1.74 mmol, 1 eq) in methanol (30 mL) was added Pd/C (10%, 0.5 g) under nitrogen atmosphere. The suspension was degassed and purged with hydrogen (x 3).
- STEP 15 8-(4-((17-chloro-3,6,9,12,15-pentaoxaheptadecyl)(methyl)amino) phenyl)chromeno[7,8-d]imidazol-6(3H)-one
- 8-(4-((17-hydroxy-3,6,9,12,15-pentaoxaheptadecyl) (methyl)amino)phenyl)chromeno[7,8-d]imidazol-6(3H)-one 450 mg, 809.91 ⁇ mol, 1 eq) in DCM (20 mL) was added SOCl 2 (8.10 mmol, 587.53 ⁇ L, 10 eq).
- Example 22 Preparation of 8-(4-((17-azido-3,6,9,12,15- pentaoxaheptadecyl)(methyl)amino)phenyl)-2-methylchromeno[7,8-d]imidazol-6(3H)- one (BA-170 and tautomer BA-201)
- STEP 1 8-(4-((17-azido-3,6,9,12,15-pentaoxaheptadecyl)(methyl)amino)phenyl)-2- methylchromeno[7,8-d]imidazol-6(3H)-one
- 7,8-diamino-2-(4-((17-azido-3,6,9,12,15- pentaoxaheptadecyl)(methyl) amino)phenyl)-4H-chromen-4-one 700 mg, 1.28 mmol, 1 eq) in acetonitrile
- STEP 3 8-(4-((17-azido-3,6,9,12,15-pentaoxaheptadecyl)(methyl)amino)phenyl)-2- methylchromeno[7,8-d]imidazol-6(3H)-one 8-(4-((17-chloro-3,6,9,12,15-pentaoxaheptadecyl)(methyl)amino)phenyl)-2- methylchromeno[7,8-d]imidazol-6(3H)-one (310 mg, 0.527 mmol, 1 eq), and sodium azide (171 mg, 2.63 mmol, 5 eq) in dry DMF (2mL) was heated at 70°C for 12 hours.
- EXAMPLE 23 N-(18-azido-3,6,9,12,15-pentaoxaoctadecyl)-4-(7,8-bis(allyloxy)-4-oxo- 4H-chromen-2-yl)benzamide (BA-203) To a solution of N-(18-azido-3,6,9,12,15-pentaoxaoctadecyl)-4-(7,8-dihydroxy-4- oxo-4H-chromen-2-yl)benzamide (BA-118, 200 mg, 0.34 mmol, 1eq) in DMF (5mL), was added K 2 CO 3 (94mg, 0.68 mmol, 2eq) followed by allyl bromide (35.2uL, 0.408 mmol, 1.2eq) at room temperature and the mixture was then heated to 80°C for 3 hours.
- one Ligand is conjugated to the 3' end of an oligonucleotide.
- Example 24 General Procedure I Type A - Ligand Conjugated to 5’ end of Sense Strand STEP 1: 5'-DBCO Functionalized Sense Strand Sodium Phosphate buffer (10% V/V, 1M, pH7) and acetonitrile (20%-50% V/V) were added to an aqueous solution of 5’-amine functionalized sense strand. A solution of DBCO- NHS (1.5-3 eq) in DMSO or acetonitrile was then added and the reaction monitored by LCMS and HPLC.
- Example 25 General Procedure I Type B - Ligand Conjugated to 5’ end of Sense Strand STEP 1: 5'-DBCO Functionalized Sense Strand Sodium Phosphate buffer (10% V/V 1M, pH7) is added to an aqueous solution of 5’- (C6-SS-C6)-mC functionalized sense strand. Tris(2-carboxyethyl)phosphine hydrochloride (TCEP) (25 eq) in water (pH7) is added and the reaction monitored by HPLC and LCMS. Upon completion, excess TCEP is removed by molecular weight cut-off with sodium phosphate buffer (100 mM, pH7, 3x).
- TCEP Tris(2-carboxyethyl)phosphine hydrochloride
- Example 26 General Procedure II Type A - Bis-homo-3',5'-Ligand Conjugated Sense Strand STEP 1: 3’,5’-bis-DBCO modified sense strand Sodium Phosphate buffer (10% V/V 1M, pH7) and acetonitrile (20%-50% V/V) were added to an aqueous solution of 3’,5’ amine functionalized sense strand. A solution of DBCO-NHS (3 eq) in DMSO or CH 3 CN was then added and the reaction monitored by LCMS and HPLC.
- Example 27 General Procedure II Type B - Bis-homo-5',3'- Ligand Conjugated Sense Strand STEP 1: 3’,5’-bis-DBCO modified sense strand Sodium Phosphate buffer (10% V/V 1M, pH7) is added to an aqueous solution of 5’, 3’-Bis (C6-SS-C6)-mC functionalized sense strand. Tris(2-carboxyethyl)phosphine hydrochloride (TCEP) (25 eq) in water (pH7) is added and the reaction monitored by HPLC and LCMS. Upon completion, excess TCEP is removed by MWCO with sodium phosphate buffer (100 mM, pH7, 3x).
- TCEP Tris(2-carboxyethyl)phosphine hydrochloride
- Example 28 General Procedure II Type C - Bis-homo-5',3'- Ligand Conjugated Sense Strand STEP 1: 5'-DBCO / 3'-(C6-SS-C6)-mC Functionalized Sense Strand Sodium Phosphate buffer (10% V/V 1M, pH7) and acetonitrile (20% -50% V/V) were added to an aqueous solution of 5’-amine functionalized sense strand.
- Tris(2-carboxyethyl)phosphine hydrochloride (TCEP) 25 eq) in water (pH7) was added and the reaction was monitored by HPLC and LCMS. Upon completion, excess TCEP was removed by MWCO with sodium phosphate buffer (100 mM, pH7, 3x).
- a solution of DBCO-MAL (3 eq) in DMSO was added and the reaction was monitored by LCMS and HPLC. Upon completion, any solids were removed via centrifugation and the solution was purified by reverse phase HPLC, dried by lyophilization and the dried bis-DBCO modified sense strand reconstituted in RNase free water for step 3.
- STEP 3 Bis-homo-5',3'- Ligand Conjugated Sense Strand
- a solution of 5’-, 3’-Bis-DBCO functionalized sense strand (1 eq) was added a solution of Ligand A-N 3 (3 eq) in DMSO or THF and the reaction was monitored by HPLC and LCMS.
- the bis-homo-5’-, 3’conjugated sense strand was purified by reverse phase HPLC or molecular weight cut-off with Amicon® Ultra-15 Centrifugal filter (3K, 5 times). The product was confirmed by HPLC and LCMS.
- Example 29 General Procedure II Type D - Bis-homo-5',3'- Ligand Conjugated Sense Strand STEP 1: 5'-(C6-SS-C6)-mC / 3'-DBCO Functionalized Sense Strand Sodium Phosphate buffer (10% V/V 1M, pH7) and acetonitrile (20% -50% V/V) were added to an aqueous solution of 5’-amine functionalized sense strand. A solution of DBCO- NHS (1.5-3 eq) in DMSO or acetonitrile was then added and the reaction was monitored by LCMS and HPLC. Upon completion, any precipitate was removed via centrifugation and the aqueous solution purified by reverse phase HPLC.
- DBCO- NHS 1.5-3 eq
- STEP 3 Bis-homo-5',3'- Ligand Conjugated Sense Strand
- a solution of 5’-, 3’-Bis-DBCO functionalized sense strand (1 eq) was added a solution of Ligand A-N 3 (3 eq) in DMSO or THF and the reaction was monitored by HPLC and LCMS.
- the bis-homo-5’-, 3’conjugated sense strand was purified by reverse phase HPLC or molecular weight cut-off with Amicon® Ultra-15 Centrifugal filter (3K, 5 times). The product was confirmed by HPLC and LCMS.
- Example 30 General Procedure III Type A - Bis-hetero-3',5'- Ligand Conjugated Sense Strand STEP 1: 5’-conjugated, 3’-(C6-SS-C6)-mC functionalized sense strand A solution of Ligand A—N 3 (2 eq) in DMSO was added to an aqueous solution of 5’- DBCO modified sense strand (1 eq, see above for preparation) and the reaction was monitored by HPLC and LCMS. Upon completion, the 5’-conjugated sense strand was purified by reverse phase HPLC or molecular weight cut-off with Amicon ® Ultra-15 Centrifugal filter (3K, 5 times).
- STEP 2 The 5’-conjugated, 3’-DBCO modified sense strand Sodium phosphate buffer (10% V/V 1M, pH7) was added to a solution of 5’- conjugated, 3’-(C6-SS-C6)-mC functionalized sense strand (1 eq) in water. Tris(2- carboxyethyl)phosphine hydrochloride (TCEP, 25 eq) in water (pH7) was added and the reaction was monitored by HPLC and LCMS. Upon completion, excess TCEP was removed by MWCO with sodium phosphate buffer (100 mM, pH7, 3x). A solution of DBCO-MAL (3 eq) in DMSO was added and the reaction monitored by HPLC and LCMS.
- Tris(2- carboxyethyl)phosphine hydrochloride (TCEP, 25 eq) in water (pH7) was added and the reaction was monitored by HPLC and LCMS.
- TCEP Tris(2- carboxyeth
- STEP 2 The 5’-DBCO, 3’-conjugated sense strand Sodium phosphate buffer (10% V/V 1M, pH7) was added to a solution of 5’- conjugated, 3’-(C6-SS-C6)-mC functionalized sense strand (1 eq) in water. Tris(2- carboxyethyl)phosphine hydrochloride (TCEP, 25 eq) in water (pH7) was added and the reaction was monitored by HPLC and LCMS. Upon completion, excess TCEP was removed by MWCO with sodium phosphate buffer (100 mM, pH7, 3x). A solution of DBCO-MAL (3 eq) in DMSO was added and the reaction monitored by HPLC and LCMS.
- Tris(2- carboxyethyl)phosphine hydrochloride (TCEP, 25 eq) in water (pH7) was added and the reaction was monitored by HPLC and LCMS.
- TCEP Tris(2- carboxyethy
- Example 32 General Procedure IV Type A - Ligand Conjugated to 3’ end of Sense Strand STEP 1: 3’- DBCO modified sense strand Sodium Phosphate buffer (10% V/V 1M, pH7) was added to an aqueous solution of 3’-(C6-SS-C6)-mC functionalized sense strand. Tris(2-carboxyethyl)phosphine hydrochloride (TCEP, 25 eq) in water (pH7) was added and the reaction monitored by HPLC and LCMS.
- TCEP Tris(2-carboxyethyl)phosphine hydrochloride
- Example 33 General Procedure IV Type B - Ligand Conjugated to 3’ end of Sense Strand STEP 1: 3'- DBCO Functionalized Sense Strand Sodium Phosphate buffer (10% V/V, 1M, pH7) and acetonitrile (20%-50% V/V) are added to an aqueous solution of 3’-amine functionalized sense strand. A solution of DBCO- NHS (1.5-3 eq) in DMSO or acetonitrile is then added and the reaction monitored by LCMS and HPLC. Upon completion, any precipitate is removed via centrifugation, the aqueous solution purified by reverse phase HPLC, dried by lyophilization and the dried DBCO modified sense strand reconstituted in RNase free water.
- 3'- DBCO Functionalized Sense Strand Sodium Phosphate buffer (10% V/V, 1M, pH7) and acetonitrile (20%-50% V/V) are added to an aqueous solution of 3’-amine functionalized sense strand.
- Example 33-A General Procedure V STEP 1: 5’-conjugated sense strand A solution of Ligand A—N3 (2 eq) in DMSO was added to an aqueous solution of 5’- DBCO modified sense strand (1 eq), and the reaction was monitored by HPLC and LCMS. Upon completion, the 5’-conjugated sense strand was purified by reverse phase HPLC and desalted with molecular weight cut-off with Amicon ® Ultra-15 Centrifugal filter (3K, 5 times). Compounds RD3171-RD3188 were prepared using General Procedure V.
- Example 34 Bis-5'-, 3'- conjugated sense strand RD3937 5' BA-120 / 3' BA-120 FXXXXXXIV+IS1525+FXXXXXXV SEQ: RD3181 / 5'EP BA-120 was conjugated to an oligo sense strand according to general procedure II type C. The product was prepared with 90% purity and confirmed by HPLC. LCMS: m/z: 9418.9 (calc. 9420.6g/mol)
- Example 35 Mono 5' conjugated sense strand RD3666 5' BA-120 FXXXXXXIV+IS1402 SEQ: RD3181 / 5'VP BA-120 was conjugated to an oligo sense strand according to general procedure I type A. The product was prepared with 92% purity, confirmed by HPLC. LCMS: m/z 8342.7 (calc. 8344.2 g/mol)
- Example 36 Mono 5' conjugated sense strand RD3913 5' BA-168 FXXXXXXVI+IS1402 SEQ: RD3181 / 5'EP BA-168 was conjugated to an oligo sense strand according to general procedure I type A. The product was prepared with 92% purity, confirmed by HPLC. LCMS: m/z: 8620.5 (calc. 8622.2 g/mol)
- Example 37 Mono 5' conjugated sense strand RD3995 5' BA-177 FXXXXXXVII+IS1402 SEQ: RD3181 / 5'EP BA-177 was conjugated to an oligo sense strand according to general procedure I type A. The product was prepared with 97% purity, confirmed by HPLC.
- Example 39 Bis 5'-, 3'- conjugated sense strand RD6054 5' BA-236 / 3' BA-236 FXXXXXXVIII+IS2460+LK0181 5'VP BA-236 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 93% purity and confirmed by HPLC. LCMS: m/z: 9401.6 (calc.
- Example 40 Bis 5'-, 3'- conjugated sense strand RD6039 5' BA-236 / 3' BA-236 FXXXXXXVIII+IS2458+FXXXXXXVIII BA-236 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 95% purity and confirmed by HPLC. LCMS: m/z: 9045.3 (calc.
- Example 42 Bis 5'-, 3'- conjugated sense strand RD6049 5' BA-236 / 3' BA-236 FXXXXXXVIII+IS2459+FXXXXXXVIII BA-236 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 92% purity and confirmed by HPLC. LCMS: m/z: 9082.3 (calc. 9084.0g/mol)
- Example 43 Mono 5' conjugated sense strand RD4518 5' BA-236 FXXXXXXVIII+IS1640 SEQ: RD3953 / 5'EP BA-120 was conjugated to an oligo sense strand according to general procedure type I type A. The product was prepared with 95% purity, confirmed by HPLC. LCMS: m/z: 8641.4 (calc. 8639.7 g/mol)
- Example 44 Mono 5' conjugated sense strand RD3941 5' BA-128 / 3' BA-128 FXXXXXIX+IS1525+FXXXXXXXIV SEQ: RD3181 / 5'EP BA-128 was conjugated to an oligo sense strand according to general procedure II type C. The product was prepared with 96% purity, confirmed by HPLC. LCMS: m/z: 9482.4 (calc.
- Example 45 Bis-5'-, 3'- conjugated sense strand RD6037 5' BA-128 / 3' BA-128 FXXXXXXIX+IS2458+FXXXXXXIX BA-128 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 97% purity and confirmed by HPLC. LCMS: m/z: 9429.1 (calc.
- Example 46 Bis-5'-, 3'- conjugated sense strand RD6047 5' BA-128 / 3' BA-128 FXXXXXXIX+IS2459+FXXXXXXIX BA-128 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 90% purity and confirmed by HPLC. LCMS: m/z: 9466.1 (calc.
- Example 47 Bis-5'-, 3'- conjugated sense strand RD6042 5' BA-128 / 3' BA-128 FXXXXXXIX+IS1664+FXXXXXXIX BA-128 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 98% purity and confirmed by HPLC. LCMS: m/z: 9785.3 (calc. 9485.8 g/mol) AND
- Example 48 Bis-5'-, 3'- conjugated sense strand RD6052 5' BA-128 / 3' BA-128 FXXXXXXIX+IS2460+FXXXXXIX BA-128 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 90% purity and confirmed by HPLC. LCMS: m/z: 9785.3 (calc. 9787.0 g/mol)
- Example 49 Mono 5' conjugated sense strand RD3667 5' BA-128 FXXXXXXIX+IS1402 SEQ: RD3181 / 5'VP BA-128 was conjugated to an oligo sense strand according to general procedure I type A. The product was ma prepared de with 97% purity, confirmed by HPLC. LCMS: m/z: 8374.4 (calc. 8375.9 g/mol)
- Example 50 Mono 5' conjugated sense strand RD3971 5' BA-171 FXXXXXX+IS1402 SEQ: RD3181 / 5'EP BA-171 was conjugated to an oligo sense strand according to general procedure I type A. The product was prepared with 98% purity, confirmed by HPLC.
- Example 52 Bis 5'-, 3'-conjugated sense strand RD4235 5' BA-171 /3' BA-171 FXXXXXX+IS1525+FXXXXXXXX SEQ: RD3181 / 5'EP BA-171 was conjugated to an oligo sense strand according to general procedure II type C. The product was prepared with 89% purity, confirmed by HPLC. LCMS: m/z: 9664.9 (calc. 9666.9 g/mol)
- Example 53 Bis 5'-, 3'- conjugated sense strand RD4420 5' BA-171 /3' BA-171 FXXXXXXX+IS1641+FXXXXXXXX SEQ: RD3953 / 5'EP BA-171 was conjugated to an oligo sense strand according to general procedure II type B. The product was prepared with 98% purity, confirmed by HPLC. LCMS: m/z: 9794.2 (calc. 9795.9 g/mol)
- Example 54 Mono 5' conjugated sense strand RD4336 5' BA-210 FXXXXXXXI+IS1402 SEQ: RD3181 / 5'EP BA-210 was conjugated to an oligo sense strand according to general procedure I type A. The prepared was made with 96% purity, confirmed by HPLC. LCMS: m/z: 8503.5 (calc. 8505.3 g/mol)
- Example 56 Bis 5'-, 3'- conjugated sense strand RD6027 5' BA-135 /3' BA-135 FXXXXXV+IS2006+FXXXXXV SEQ: RD3181 / 5'VP BA-135 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 87% purity, confirmed by HPLC. LCMS: m/z 9249.5, (calc.
- Example 57 Bis 5'-, 3'- conjugated sense strand RD6028 5' BA-136 /3' BA-136 FXXXXXVI+IS2006+FXXXXXVI SEQ: RD3181 / 5'VP BA-136 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 97% purity, confirmed by HPLC. LCMS: m/z 9075.2, (calc.
- Example 58 Bis 5'-, 3'- conjugated sense strand RD6029 5' BA-137 /3' BA-137 FXXXXXVII+IS2006+FXXXXXVII SEQ: RD3181 / 5'VP BA-137 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 92% purity, confirmed by HPLC. LCMS: m/z 9169.2, (calc. 9171.0 g/mol)
- Example 59 Bis 5'-, 3'- conjugated sense strand RD6030 5' BA-144 /3' BA-144 FXXXXXIX+IS2006+FXXXXXIX SEQ: RD3181 / 5'VP BA-144 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 94% purity, confirmed by HPLC. LCMS: m/z 9567.7, (calc. 9569.5 g/mol)
- Example 60 Bis 5'-, 3'- conjugated sense strand RD6031 5' BA-173 /3' BA-173 FXXIV+IS2006+FXXIV SEQ: RD3181 / 5'VP BA-173 was conjugated to an oligo sense strand according to general procedure II type A. The product was made with 81% purity, confirmed by HPLC. LCMS: m/z 9681.5, (calc. 9683.4 g/mol)
- Example 61 Bis 5'-, 3'- conjugated sense strand RD6032 5' BA-183 /3' BA-183 FXXXXXXI+IS2006+FXXXXXXI SEQ: RD3181 / 5'VP BA-183 was conjugated to an oligo sense strand according to general procedure II type A. The product was made with 86% purity, confirmed by HPLC. LCMS: m/z9337.4, (calc.
- Example 62 Bis 5'-, 3'- conjugated sense strand RD6034 5' BA-216 /3' BA-216 FXXXXXXII+IS2006+FXXXXXXII SEQ: RD3181 / 5'VP BA-216 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 94% purity, confirmed by HPLC. LCMS: m/z 9373.3, (calc.
- Example 63 Bis 5'-, 3'- conjugated sense strand RD6036 5' BA-198 /3' BA-198 FXVI+IS2458+FXVI SEQ: RD3175 / 5'VP BA-198 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 98% purity, confirmed by HPLC. LCMS: m/z 9329.4, (calc.
- Example 64 Bis 5'-, 3'- conjugated sense strand RD6041 5' BA-198 /3' BA-198 FXVI+ IS1664+FXVI SEQ: RD3176/ 5'VP BA-198 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 93% purity, confirmed by HPLC. LCMS: m/z 9384.4, (calc.
- Example 65 Bis 5'-, 3'- conjugated sense strand RD6046 5' BA-198 /3' BA-198 FXVI+ IS2459+FXVI SEQ: RD3186/ 5'VP BA-198 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 90% purity, confirmed by HPLC. LCMS: m/z 9366.4, (calc.
- Example 66 Bis 5'-, 3'- conjugated sense strand RD6051 5' BA-198 /3' BA-198 FXVI+ IS2460+FXVI SEQ: RD3959/ 5'VP BA-198 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 94% purity, confirmed by HPLC. LCMS: m/z 9685.6, (calc. 9687.3 g/mol)
- Example 67 Bis 5'-, 3'- conjugated sense strand RD6035 5' BA-225 /3' BA-225 FXXXIII+IS2006+FXXXIII BA-225 was conjugated to an oligo sense strand according to general procedure II type A.
- Example 69 Mono 5'- conjugated sense strand RD4310 5' BA-197 FXV+IS1402 BA-197 was conjugated to an oligo sense strand according to general procedure I type A. The product was prepared with 95% purity, confirmed by HPLC. LCMS: m/z 8310.4, (calc. 8312.0 g/mol)
- Example 70 Mono 3'- conjugated sense strand RD4157 3' BA-118 IS1599+FIV BA-118 was conjugated to an oligo sense strand according to general procedure IV type A. The product was prepared with 86% purity, confirmed by HPLC. LCMS: m/z 8652.1, (calc. 8653.4 g/mol)
- Example 71 Mono 3'- conjugated sense strand RD4269 3' BA-129 IS1599+FXXXXXXIII BA-129 was conjugated to an oligo sense strand according to general procedure IV type A. The product was prepared with 97% purity, confirmed by HPLC. LCMS: m/z 8582.1, (calc. 8583.8 g/mol)
- Example 72 Bis 5'-, 3'- conjugated sense strand RD5470 5' BA-198 /3' BA-198 FXVI+IS2178+FXVI SEQ: RD3953/ 5'VP IA1603 BA-198 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 95% purity, confirmed by HPLC.
- Example 74 Bis 5'-, 3'- conjugated sense strand RD5947 5' BA-198 /3' BA-198 FXVI+IS1681+FXVI SEQ: RD3953/ 5'VP IA1603 BA-198 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 94% purity, confirmed by HPLC. LCMS: m/z 9215.3, (calc.
- Example 75 Bis 5'-, 3'- conjugated sense strand RD5948 5' BA-198 /3' BA-198 FXVI+IS1681+FXVI SEQ: RD3953/ 5'EP IA1299 BA-198 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 94% purity, confirmed by HPLC. LCMS: m/z 9215.3, (calc.
- Example 76 Bis 5'-, 3'- conjugated sense strand RD5954 5' BA-198 /3' BA-198 FXVI+IS1688+FXVI SEQ: RD3953/ 5'VP IA1603 BA-198 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 93% purity, confirmed by HPLC. LCMS: m/z 9414.7, (calc.
- Example 77 Bis 5'-, 3'- conjugated sense strand RD5955 5' BA-198 /3' BA-198 FXVI+IS1688+FXVI SEQ: RD3953/ 5'EP IA1299 BA-198 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 93% purity, confirmed by HPLC. LCMS: m/z 9414.7, (calc.
- Example 78 Bis 5'-, 3'- conjugated sense strand RD5956 5' BA-198 /3' BA-198 FXVI+IS2422+FXVI SEQ: RD3953/ 5'VP IA1250 BA-198 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 87% purity, confirmed by HPLC. LCMS: m/z 9774.1, (calc.
- Example 79 Bis 5'-, 3'- conjugated sense strand RD5967 5' BA-198 /3' BA-198 FXVI+IS2424+FXVI SEQ: RD3953/ 5'VP IA1250 BA-198 was conjugated to an oligo sense strand according to general procedure II type A. The product was prepared with 87% purity, confirmed by HPLC. LCMS: m/z 9774.1, (calc. 9775.7 g/mol)
- Example 80 Synthesis of ligand-conjugated oligonucleotides using amide linkers Oligonucleotides conjugated to ligands using amide linkers may be synthesized using the following methods: The synthetic methods above may be used to synthesize, for example, the following oligonucleotides comprising ligands at the 5' and/or 3' ends:
- Example 81 General Procedures for Synthesis of Oligonucleotides Strands were synthesized on solid phase using an oligonucleotide synthesizer Oligopilot100 (Cytiva Life Sciences). Solid support (CPG, 80-90 ⁇ mol/g, 500A, from LGC- Biosearch Technologies, Petaluma, CA) was loaded to 150-300 ⁇ mol scales. RNA and 2' modified RNA phosphoramidites were purchased from Hongene Biotech (Union City, CA).
- 2'-O-methyl phosphoramidites used were: 5'-O-(4,4'-Dimethoxytrityl)-N 6 -benzoyl-2'-O-methyl-adenosine-3'-O-[(2-cyanoethyl)-(N,N- diisopropyl)]-phosphoramidite 5'-O-(4,4'-Dimethoxytrityl)-N 4 -acetyl-2'-O-methyl-cytidine-3'-O-[(2-cyanoethyl)-(N,N- diisopropyl)]-phosphoramidite 5'-O-(4,4'-Dimethoxytrityl)-N 2 -isobutyryl-2'-O-methyl-guanosine-3'-O-[(2-cyanoethyl)-(N,N- diisopropyl)]-phosphoramidite 5'-O-(4,4'-Dimethoxytrityl
- 2'-Fluoro phosphoramidites used were: 5'-O-(4,4'-Dimethoxytrityl)-N 6 -benzoyl-2'-fluoroadenosine-3'-O-[(2-cyanoethyl)-(N,N- diisopropyl)]-phosphoramidite 5'-O-(4,4'-Dimethoxytrityl)-N 4 -acetyl-2'-fluorocytidine-3'-O-[(2-cyanoethyl)-(N,N- diisopropyl)]-phosphoramidite 5'-O-(4,4'-Dimethoxytrityl)-N 2 -isobutyryl-2'-fluoroguanosine-3'-O-[(2-cyanoethyl)-(N,N- diisopropyl)]-phosphoramidite 5'-O-(4,4'-Dimethoxytrityl)-2'-
- Coupling times were 6 minutes carried out at 3.0 equivalents for each step.
- the support bound oligonucleotide Prior to coupling the support bound oligonucleotide is treated with a solution of Dichloroacetic Acid in Dichloromethane (3% Deblock, Sigma Aldrich) and washed with Anhydrous Acetonitrile. Cleavage and deprotection of support bound oligomer After completion of the solid phase synthesis, the support was treated with AMA solution, a 1:1 volume solution of NH 4 OH:CH 3 NH 2 (Fisher Scientific, Spectrum Chemicals), for 20 minutes at 65°C. The solution was then evaporated. Prior to purification, in-process analysis is performed on analytical HPLC and LCMS to determine crude purity, identify the target mass and monitor the deprotection for completion.
- Buffer solution mixtures are 100 mM TEAA, 5% ACN at pH of 7.0 (buffer A) and 1:1 acetonitrile:methanol (buffer B). Gradient was set at 5-30% or 30-60% Buffer B over 5 minutes at 70°C with a flowrate of 1.0 mL/minute. The minimum spec of the purified pool is 85%. Desalting Once a pool has been established, the oligos are desalted using Pall Minimate EVO System (Product ID: OAPMPUNV). Cassette used is the Pall Minimate TFF capsule with 3k Omega membrane (Product ID: OA003C12). Retentate is collected for lyophilization or annealing directly.
- RNA antisense strands were prepared according to procedures well known to those of skill in the art. The concentrations of both sense strand and antisense strands were determined by Nanodrop. The double-stranded siRNA was prepared by mixing equimolar amounts of sense stand and antisense strand. The annealing process was monitored by RP-HPLC, non- denaturing method. After annealing, no more that 5% of antisense strand was in the duplex mixture. Duplex concentration was determined by measuring the solution absorbance on Nanodrop.
- Example 83 General procedure for preparation of Carbon chain containing dinucleotide amidites STEP 1: N,N-Diisopropylethylamine (DIPEA, 3 eq) is added to a stirred solution of optionally base protected 3-(5-["A/C/G/U”]-5-(DMTrO-methyl)-2-[F/OMe”]- tetrahydrofuran-3-yl)acetic acid (1 eq), optionally base protected 1-["A/C/G/U”]-2-[F/OMe”]- 5-((heptadecylamino)methyl) tetrahydrofuran-3-ol (1.15 eq) and HATU (1.5 eq) in DMF.
- DIPEA N,N-Diisopropylethylamine
- RNA Isolation was performed according to the RNeasy Micro Kit (Qiagen Cat #74004) instructions. Following RNA isolation, a 96-well plate was placed on ice while the qRT-PCR reaction was prepared.
- RNA 2 ⁇ l was added to the reaction mixture containing 5 ⁇ l TaqMan Fast Virus 1-Step Master Mix (Thermo Fisher #44444432), 1 ⁇ l human MAPT TaqMan Gene Expression Assay (Thermo Fisher: Hs00213484_m1, FAM), 1 ⁇ l mouse GAPDH TaqMan Gene Expression Assay (Thermo Fisher: Mm99999915_g1, VIC) and 11 ⁇ l RT-PCR grade nuclease-free water in a MicroAmp Optical 96-well plate (0.2 mL).
- RNA Isolation was performed according to the RNeasy Micro Kit (Qiagen Cat #74004) instructions. Following RNA isolation, a 96-well plate was placed on ice while the qRT-PCR reaction was prepared.
- RNA 2 ⁇ l was added to the reaction mixture containing 5 ⁇ l TaqMan Fast Virus 1-Step Master Mix (Thermo Fisher #44444432), 1 ⁇ l cyno MAPT TaqMan Gene Expression Assay (Thermo Fisher: Mf00902189_m1, FAM), 1 ⁇ l cyno ARL1 TaqMan Gene Expression Assay (Thermo Fisher: Mf02795431_m1, VIC) and 11 ⁇ l RT- PCR grade nuclease-free water in a MicroAmp Optical 96-well plate (0.2 mL).
- qPCR was performed using a QuantStudio3 qPCR machine with the following cycles: 50°C for 1 minute, 95°C for 20 seconds, 40 cycles at 95°C for 15 seconds, and 60°C for 1 minute. Results are presented in Table 5 below as percent inhibition of MAPT RNA, relative to control (pre-dosed level from the same animal). Due to intrathecal injection variation, certain animals may have received a lower dose and therefore may show lower inhibition as a result. The amount of test compound present in CSF was measured at 1h, 6h, and 24h post dose. Table 6 shows the CSF compound concentration ( ⁇ g/mL) for the two animals dosed with RD4845. Table 5 Percent MAPT RNA Inhibition in Brain and Spinal Cord
- any particular embodiment of this disclosure that falls within the prior art may be explicitly excluded from any one or more of the claims. Because such embodiments are deemed to be known to one of ordinary skill in the art, they may be excluded even if the exclusion is not set forth explicitly herein. Any particular embodiment of the disclosure can be excluded from any claim, for any reason, whether or not related to the existence of prior art.
- the citation of any publication is for its disclosure prior to the filing date and should not be construed as an admission that the present disclosure is not entitled to antedate such publication by virtue of prior disclosure. Further, the dates of publication provided could be different from the actual publication dates that may need to be independently confirmed.
- a pronoun in a gender e.g., masculine, feminine, neuter, other, etc.
- the pronoun shall be construed as gender neutral (e.g., construed to refer to all genders equally) regardless of the implied gender unless the context clearly indicates or requires otherwise.
- words used in the singular include the plural, and words used in the plural includes the singular, unless the context clearly indicates or requires otherwise. Claims or descriptions that include “or” between one or more members of a group are considered satisfied if one, more than one, or all of the group members are present in, employed in, or otherwise relevant to a given product or process unless indicated to the contrary or otherwise evident from the context.
- the disclosure includes embodiments in which exactly one member of the group is present in, employed in, or otherwise relevant to a given product or process.
- the disclosure includes embodiments in which more than one, or all of the group members are present in, employed in, or otherwise relevant to a given product or process.
- the disclosure encompasses all variations, combinations, and permutations in which one or more limitations, elements, clauses, and descriptive terms from one or more of the listed claims is introduced into another claim.
- any claim that is dependent on another claim can be modified to include one or more limitations found in any other claim that is dependent on the same base claim.
- elements are presented as lists (e.g., in Markush group format), each subgroup of the elements is also disclosed, and any element(s) can be removed from the group.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Biomedical Technology (AREA)
- General Health & Medical Sciences (AREA)
- Biochemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Veterinary Medicine (AREA)
- Organic Chemistry (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Epidemiology (AREA)
- Biotechnology (AREA)
- General Engineering & Computer Science (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Biophysics (AREA)
- Plant Pathology (AREA)
- Microbiology (AREA)
- Physics & Mathematics (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Steroid Compounds (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Hospice & Palliative Care (AREA)
- Psychiatry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020257038056A KR20260007584A (en) | 2023-04-20 | 2024-04-19 | MAPT-modulating composition and method of use thereof |
| AU2024258561A AU2024258561A1 (en) | 2023-04-20 | 2024-04-19 | Mapt-modulating compositions and methods of use thereof |
| CN202480033384.9A CN121285630A (en) | 2023-04-20 | 2024-04-19 | MAPT modulating compositions and methods of use thereof |
| IL323920A IL323920A (en) | 2023-04-20 | 2025-10-13 | MAPT modulators and methods of using them |
| MX2025012458A MX2025012458A (en) | 2023-04-20 | 2025-10-17 | Mapt-modulating compositions and methods of use thereof |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363460874P | 2023-04-20 | 2023-04-20 | |
| US63/460,874 | 2023-04-20 | ||
| US202363520070P | 2023-08-16 | 2023-08-16 | |
| US63/520,070 | 2023-08-16 | ||
| US202463619022P | 2024-01-09 | 2024-01-09 | |
| US63/619,022 | 2024-01-09 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| WO2024220930A2 true WO2024220930A2 (en) | 2024-10-24 |
| WO2024220930A3 WO2024220930A3 (en) | 2024-12-26 |
| WO2024220930A8 WO2024220930A8 (en) | 2025-11-06 |
Family
ID=91277045
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/025577 Pending WO2024220930A2 (en) | 2023-04-20 | 2024-04-19 | Mapt-modulating compositions and methods of use thereof |
Country Status (7)
| Country | Link |
|---|---|
| KR (1) | KR20260007584A (en) |
| CN (1) | CN121285630A (en) |
| AU (1) | AU2024258561A1 (en) |
| IL (1) | IL323920A (en) |
| MX (1) | MX2025012458A (en) |
| TW (1) | TW202448484A (en) |
| WO (1) | WO2024220930A2 (en) |
Citations (142)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3687808A (en) | 1969-08-14 | 1972-08-29 | Univ Leland Stanford Junior | Synthetic polynucleotides |
| US4751219A (en) | 1985-02-05 | 1988-06-14 | Nederlandse Centrale Organisatie Voor Toegepast-Natuur-Wetenschappelijk Onderzoek | Synthetic glycolipides, a process for the preparation thereof and several uses for these synthetic glycolipides |
| US4845205A (en) | 1985-01-08 | 1989-07-04 | Institut Pasteur | 2,N6 -disubstituted and 2,N6 -trisubstituted adenosine-3'-phosphoramidites |
| US4981957A (en) | 1984-07-19 | 1991-01-01 | Centre National De La Recherche Scientifique | Oligonucleotides with modified phosphate and modified carbohydrate moieties at the respective chain termini |
| US5118800A (en) | 1983-12-20 | 1992-06-02 | California Institute Of Technology | Oligonucleotides possessing a primary amino group in the terminal nucleotide |
| US5130302A (en) | 1989-12-20 | 1992-07-14 | Boron Bilogicals, Inc. | Boronated nucleoside, nucleotide and oligonucleotide compounds, compositions and methods for using same |
| US5134066A (en) | 1989-08-29 | 1992-07-28 | Monsanto Company | Improved probes using nucleosides containing 3-dezauracil analogs |
| US5166315A (en) | 1989-12-20 | 1992-11-24 | Anti-Gene Development Group | Sequence-specific binding polymers for duplex nucleic acids |
| US5175273A (en) | 1988-07-01 | 1992-12-29 | Genentech, Inc. | Nucleic acid intercalating agents |
| US5185444A (en) | 1985-03-15 | 1993-02-09 | Anti-Gene Deveopment Group | Uncharged morpolino-based polymers having phosphorous containing chiral intersubunit linkages |
| US5319080A (en) | 1991-10-17 | 1994-06-07 | Ciba-Geigy Corporation | Bicyclic nucleosides, oligonucleotides, process for their preparation and intermediates |
| US5359044A (en) | 1991-12-13 | 1994-10-25 | Isis Pharmaceuticals | Cyclobutyl oligonucleotide surrogates |
| US5367066A (en) | 1984-10-16 | 1994-11-22 | Chiron Corporation | Oligonucleotides with selectably cleavable and/or abasic sites |
| US5432272A (en) | 1990-10-09 | 1995-07-11 | Benner; Steven A. | Method for incorporating into a DNA or RNA oligonucleotide using nucleotides bearing heterocyclic bases |
| US5434257A (en) | 1992-06-01 | 1995-07-18 | Gilead Sciences, Inc. | Binding compentent oligomers containing unsaturated 3',5' and 2',5' linkages |
| US5446137A (en) | 1993-12-09 | 1995-08-29 | Syntex (U.S.A.) Inc. | Oligonucleotides containing 4'-substituted nucleotides |
| US5457191A (en) | 1990-01-11 | 1995-10-10 | Isis Pharmaceuticals, Inc. | 3-deazapurines |
| US5457187A (en) | 1993-12-08 | 1995-10-10 | Board Of Regents University Of Nebraska | Oligonucleotides containing 5-fluorouracil |
| US5459255A (en) | 1990-01-11 | 1995-10-17 | Isis Pharmaceuticals, Inc. | N-2 substituted purines |
| US5466786A (en) | 1989-10-24 | 1995-11-14 | Gilead Sciences | 2'modified nucleoside and nucleotide compounds |
| US5484908A (en) | 1991-11-26 | 1996-01-16 | Gilead Sciences, Inc. | Oligonucleotides containing 5-propynyl pyrimidines |
| US5502177A (en) | 1993-09-17 | 1996-03-26 | Gilead Sciences, Inc. | Pyrimidine derivatives for labeled binding partners |
| US5514785A (en) | 1990-05-11 | 1996-05-07 | Becton Dickinson And Company | Solid supports for nucleic acid hybridization assays |
| US5519134A (en) | 1994-01-11 | 1996-05-21 | Isis Pharmaceuticals, Inc. | Pyrrolidine-containing monomers and oligomers |
| US5525711A (en) | 1994-05-18 | 1996-06-11 | The United States Of America As Represented By The Secretary Of The Department Of Health And Human Services | Pteridine nucleotide analogs as fluorescent DNA probes |
| US5552540A (en) | 1987-06-24 | 1996-09-03 | Howard Florey Institute Of Experimental Physiology And Medicine | Nucleoside derivatives |
| US5567811A (en) | 1990-05-03 | 1996-10-22 | Amersham International Plc | Phosphoramidite derivatives, their preparation and the use thereof in the incorporation of reporter groups on synthetic oligonucleotides |
| US5576427A (en) | 1993-03-30 | 1996-11-19 | Sterling Winthrop, Inc. | Acyclic nucleoside analogs and oligonucleotide sequences containing them |
| US5587470A (en) | 1990-01-11 | 1996-12-24 | Isis Pharmaceuticals, Inc. | 3-deazapurines |
| US5591722A (en) | 1989-09-15 | 1997-01-07 | Southern Research Institute | 2'-deoxy-4'-thioribonucleosides and their antiviral activity |
| US5594121A (en) | 1991-11-07 | 1997-01-14 | Gilead Sciences, Inc. | Enhanced triple-helix and double-helix formation with oligomers containing modified purines |
| US5596091A (en) | 1994-03-18 | 1997-01-21 | The Regents Of The University Of California | Antisense oligonucleotides comprising 5-aminoalkyl pyrimidine nucleotides |
| US5597909A (en) | 1994-08-25 | 1997-01-28 | Chiron Corporation | Polynucleotide reagents containing modified deoxyribose moieties, and associated methods of synthesis and use |
| US5610300A (en) | 1992-07-01 | 1997-03-11 | Ciba-Geigy Corporation | Carbocyclic nucleosides containing bicyclic rings, oligonucleotides therefrom, process for their preparation, their use and intermediates |
| US5614617A (en) | 1990-07-27 | 1997-03-25 | Isis Pharmaceuticals, Inc. | Nuclease resistant, pyrimidine modified oligonucleotides that detect and modulate gene expression |
| US5627053A (en) | 1994-03-29 | 1997-05-06 | Ribozyme Pharmaceuticals, Inc. | 2'deoxy-2'-alkylnucleotide containing nucleic acid |
| WO1997020563A1 (en) | 1995-11-22 | 1997-06-12 | The Johns-Hopkins University | Ligands to enhance cellular uptake of biomolecules |
| US5639873A (en) | 1992-02-05 | 1997-06-17 | Centre National De La Recherche Scientifique (Cnrs) | Oligothionucleotides |
| US5646265A (en) | 1990-01-11 | 1997-07-08 | Isis Pharmceuticals, Inc. | Process for the preparation of 2'-O-alkyl purine phosphoramidites |
| US5645985A (en) | 1991-11-26 | 1997-07-08 | Gilead Sciences, Inc. | Enhanced triple-helix and double-helix formation with oligomers containing modified pyrimidines |
| US5658873A (en) | 1993-04-10 | 1997-08-19 | Degussa Aktiengesellschaft | Coated sodium percarbonate particles, a process for their production and detergent, cleaning and bleaching compositions containing them |
| US5670633A (en) | 1990-01-11 | 1997-09-23 | Isis Pharmaceuticals, Inc. | Sugar modified oligonucleotides that detect and modulate gene expression |
| US5681941A (en) | 1990-01-11 | 1997-10-28 | Isis Pharmaceuticals, Inc. | Substituted purines and oligonucleotide cross-linking |
| WO1997046098A1 (en) | 1996-06-06 | 1997-12-11 | Neorx Corporation | Cluster clearing agents |
| WO1998013381A1 (en) | 1996-09-26 | 1998-04-02 | Ajinomoto Co., Inc. | Modified physiologically active proteins and medicinal compositions containing the same |
| US5830653A (en) | 1991-11-26 | 1998-11-03 | Gilead Sciences, Inc. | Methods of using oligomers containing modified pyrimidines |
| WO1999014226A2 (en) | 1997-09-12 | 1999-03-25 | Exiqon A/S | Bi- and tri-cyclic nucleoside, nucleotide and oligonucleotide analogues |
| US5948903A (en) | 1991-01-11 | 1999-09-07 | Isis Pharmaceuticals, Inc. | Synthesis of 3-deazapurines |
| US6015886A (en) | 1993-05-24 | 2000-01-18 | Chemgenes Corporation | Oligonucleotide phosphate esters |
| US6147200A (en) | 1999-08-19 | 2000-11-14 | Isis Pharmaceuticals, Inc. | 2'-O-acetamido modified monomers and oligomers |
| US6166197A (en) | 1995-03-06 | 2000-12-26 | Isis Pharmaceuticals, Inc. | Oligomeric compounds having pyrimidine nucleotide (S) with 2'and 5 substitutions |
| US6222025B1 (en) | 1995-03-06 | 2001-04-24 | Isis Pharmaceuticals, Inc. | Process for the synthesis of 2′-O-substituted pyrimidines and oligomeric compounds therefrom |
| US6235887B1 (en) | 1991-11-26 | 2001-05-22 | Isis Pharmaceuticals, Inc. | Enhanced triple-helix and double-helix formation directed by oligonucleotides containing modified pyrimidines |
| US6268490B1 (en) | 1997-03-07 | 2001-07-31 | Takeshi Imanishi | Bicyclonucleoside and oligonucleotide analogues |
| US6300319B1 (en) | 1998-06-16 | 2001-10-09 | Isis Pharmaceuticals, Inc. | Targeted oligonucleotide conjugates |
| US6383812B1 (en) | 1999-05-28 | 2002-05-07 | Academia Sinica | Anti liver disease drug R-YEEE and method of synthesizing branched galactose-terminal glycoproteins |
| WO2002043771A2 (en) | 2000-12-01 | 2002-06-06 | Cell Works Inc. | Conjugates of glycosylated/galactosylated peptide |
| US6525191B1 (en) | 1999-05-11 | 2003-02-25 | Kanda S. Ramasamy | Conformationally constrained L-nucleosides |
| US6528640B1 (en) | 1997-11-05 | 2003-03-04 | Ribozyme Pharmaceuticals, Incorporated | Synthetic ribonucleic acids with RNAse activity |
| US20030077829A1 (en) | 2001-04-30 | 2003-04-24 | Protiva Biotherapeutics Inc.. | Lipid-based formulations |
| US20030119724A1 (en) | 1995-11-22 | 2003-06-26 | Ts`O Paul O.P. | Ligands to enhance cellular uptake of biomolecules |
| US20030158403A1 (en) | 2001-07-03 | 2003-08-21 | Isis Pharmaceuticals, Inc. | Nuclease resistant chimeric oligonucleotides |
| US6617438B1 (en) | 1997-11-05 | 2003-09-09 | Sirna Therapeutics, Inc. | Oligoribonucleotides with enzymatic activity |
| US20030175906A1 (en) | 2001-07-03 | 2003-09-18 | Muthiah Manoharan | Nuclease resistant chimeric oligonucleotides |
| US6639062B2 (en) | 1997-02-14 | 2003-10-28 | Isis Pharmaceuticals, Inc. | Aminooxy-modified nucleosidic compounds and oligomeric compounds prepared therefrom |
| US6670461B1 (en) | 1997-09-12 | 2003-12-30 | Exiqon A/S | Oligonucleotide analogues |
| WO2004024757A2 (en) | 2002-09-11 | 2004-03-25 | Santaris Pharma A/S | Modified pna molecules |
| US6770748B2 (en) | 1997-03-07 | 2004-08-03 | Takeshi Imanishi | Bicyclonucleoside and oligonucleotide analogue |
| US20040171570A1 (en) | 2002-11-05 | 2004-09-02 | Charles Allerson | Polycyclic sugar surrogate-containing oligomeric compounds and compositions for use in gene modulation |
| WO2004101619A1 (en) | 2003-05-15 | 2004-11-25 | Shionogi Co., Ltd. | Rational design and synthesis of functional glycopeptide |
| US6908903B1 (en) | 1994-12-07 | 2005-06-21 | Aletheon Pharmaceuticals, Inc. | Cluster clearing agents |
| US20050164235A1 (en) | 2003-04-17 | 2005-07-28 | Muthiah Manoharan | Modified iRNA agents |
| US6998484B2 (en) | 2000-10-04 | 2006-02-14 | Santaris Pharma A/S | Synthesis of purine locked nucleic acid analogues |
| US7045610B2 (en) | 1998-04-03 | 2006-05-16 | Epoch Biosciences, Inc. | Modified oligonucleotides for mismatch discrimination |
| US7053207B2 (en) | 1999-05-04 | 2006-05-30 | Exiqon A/S | L-ribo-LNA analogues |
| US20060148740A1 (en) | 2005-01-05 | 2006-07-06 | Prosensa B.V. | Mannose-6-phosphate receptor mediated gene transfer into muscle cells |
| US7084125B2 (en) | 1999-03-18 | 2006-08-01 | Exiqon A/S | Xylo-LNA analogues |
| US20080039618A1 (en) | 2002-11-05 | 2008-02-14 | Charles Allerson | Polycyclic sugar surrogate-containing oligomeric compounds and compositions for use in gene modulation |
| US20080108801A1 (en) | 2003-04-17 | 2008-05-08 | Muthiah Manoharan | Lipophilic Conjugated iRNA Agents |
| US7399845B2 (en) | 2006-01-27 | 2008-07-15 | Isis Pharmaceuticals, Inc. | 6-modified bicyclic nucleic acid analogs |
| WO2008098788A2 (en) | 2007-02-16 | 2008-08-21 | Ktb Tumorforschungsgesellschaft Mbh | Receptor and antigen targeted prodrug |
| WO2008101157A1 (en) | 2007-02-15 | 2008-08-21 | Isis Pharmaceuticals, Inc. | 5'-substituted-2'-f modified nucleosides and oligomeric compounds prepared therefrom |
| US20080206869A1 (en) | 2005-01-24 | 2008-08-28 | Avaris Ab | Nucleic Acid Complex |
| US7427672B2 (en) | 2003-08-28 | 2008-09-23 | Takeshi Imanishi | Artificial nucleic acids of n-o bond crosslinkage type |
| US7491805B2 (en) | 2001-05-18 | 2009-02-17 | Sirna Therapeutics, Inc. | Conjugates and compositions for cellular delivery |
| US7495088B1 (en) | 1989-12-04 | 2009-02-24 | Enzo Life Sciences, Inc. | Modified nucleotide compounds |
| WO2009073809A2 (en) | 2007-12-04 | 2009-06-11 | Alnylam Pharmaceuticals, Inc. | Carbohydrate conjugates as delivery agents for oligonucleotides |
| US7569686B1 (en) | 2006-01-27 | 2009-08-04 | Isis Pharmaceuticals, Inc. | Compounds and methods for synthesis of bicyclic nucleic acid analogs |
| US20090203132A1 (en) | 2004-09-09 | 2009-08-13 | Swayze Eric E | Pyrrolidinyl groups for attaching conjugates to oligomeric compounds |
| US7582744B2 (en) | 2004-08-10 | 2009-09-01 | Alnylam Pharmaceuticals, Inc. | Chemically modified oligonucleotides |
| WO2009126933A2 (en) | 2008-04-11 | 2009-10-15 | Alnylam Pharmaceuticals, Inc. | Site-specific delivery of nucleic acids by combining targeting ligands with endosomolytic components |
| WO2009134487A2 (en) | 2008-01-31 | 2009-11-05 | Alnylam Pharmaceuticals, Inc. | Optimized methods for delivery of dsrna targeting the pcsk9 gene |
| WO2010054406A1 (en) | 2008-11-10 | 2010-05-14 | Alnylam Pharmaceuticals, Inc. | Novel lipids and compositions for the delivery of therapeutics |
| US7723509B2 (en) | 2003-04-17 | 2010-05-25 | Alnylam Pharmaceuticals | IRNA agents with biocleavable tethers |
| WO2010088537A2 (en) | 2009-01-29 | 2010-08-05 | Alnylam Pharmaceuticals, Inc. | Improved lipid formulation |
| WO2010129709A1 (en) | 2009-05-05 | 2010-11-11 | Alnylam Pharmaceuticals, Inc. | Lipid compositions |
| WO2010144740A1 (en) | 2009-06-10 | 2010-12-16 | Alnylam Pharmaceuticals, Inc. | Improved lipid formulation |
| WO2010148013A2 (en) | 2009-06-15 | 2010-12-23 | Alnylam Pharmaceuticals, Inc. | Lipid formulated dsrna targeting the pcsk9 gene |
| US7875733B2 (en) | 2003-09-18 | 2011-01-25 | Isis Pharmaceuticals, Inc. | Oligomeric compounds comprising 4′-thionucleosides for use in gene modulation |
| WO2011038356A2 (en) | 2009-09-25 | 2011-03-31 | Johns Hopkins University | Novel liver-targeting agents and their synthesis |
| US20110097265A1 (en) | 2009-10-26 | 2011-04-28 | Institute Of Nuclear Energy Research Atomic Energy Council, Executive Yuan | Quantification method for remaining liver function and novel liver receptor imaging agent |
| US20110097264A1 (en) | 2009-10-26 | 2011-04-28 | Institute Of Nuclear Energy Research Atomic Energy Council, Executive Yuan | Radiolabeling method using multivalent glycoligands as hepatic receptor imaging agent |
| WO2011100131A2 (en) | 2010-01-28 | 2011-08-18 | Alnylam Pharmacuticals, Inc. | Monomers and oligonucleotides comprising cycloaddition adduct(s) |
| US20110207799A1 (en) | 2010-02-24 | 2011-08-25 | Roche Madison Inc. | Compositions for Targeted Delivery of siRNA |
| WO2011120053A1 (en) | 2010-03-26 | 2011-09-29 | Mersana Therapeutics, Inc. | Modified polymers for delivery of polynucleotides, method of manufacture, and methods of use thereof |
| US8030467B2 (en) | 2006-05-11 | 2011-10-04 | Isis Pharmaceuticals, Inc. | 5′-modified bicyclic nucleic acid analogs |
| US20110313020A1 (en) | 2008-12-03 | 2011-12-22 | Marina Biotech, Inc. | UsiRNA Complexes |
| WO2011163121A1 (en) | 2010-06-21 | 2011-12-29 | Alnylam Pharmaceuticals, Inc. | Multifunctional copolymers for nucleic acid delivery |
| US8088904B2 (en) | 2007-08-15 | 2012-01-03 | Isis Pharmaceuticals, Inc. | Tetrahydropyran nucleic acid analogs |
| US20120035115A1 (en) | 2008-09-23 | 2012-02-09 | Alnylam Pharmaceuticals, Inc. | Chemical modifications of monomers and oligonucleotides with cycloaddition |
| US8137695B2 (en) | 2006-08-18 | 2012-03-20 | Arrowhead Madison Inc. | Polyconjugates for in vivo delivery of polynucleotides |
| WO2012037254A1 (en) | 2010-09-15 | 2012-03-22 | Alnylam Pharmaceuticals, Inc. | MODIFIED iRNA AGENTS |
| WO2012068187A1 (en) | 2010-11-19 | 2012-05-24 | Merck Sharp & Dohme Corp. | Poly(amide) polymers for the delivery of oligonucleotides |
| WO2012083185A2 (en) | 2010-12-17 | 2012-06-21 | Arrowhead Research Corporations | Peptide-based in vivo sirna delivery system |
| WO2012083046A2 (en) | 2010-12-17 | 2012-06-21 | Arrowhead Research Corporation | Galactose cluster-pharmacokinetic modulator targeting moiety for sirna |
| WO2012089602A1 (en) | 2010-12-29 | 2012-07-05 | F. Hoffmann-La Roche Ag | Small molecule conjugates for intracellular delivery of biologically active compounds |
| US20120230938A1 (en) | 2006-08-18 | 2012-09-13 | Arrowhead Madison Inc. | Polyconjugates for In Vivo Delivery of Polynucleotides |
| US8278425B2 (en) | 2007-05-30 | 2012-10-02 | Isis Pharmaceuticals, Inc. | N-substituted-aminomethylene bridged bicyclic nucleic acid analogs |
| US8278426B2 (en) | 2007-06-08 | 2012-10-02 | Isis Pharmaceuticals, Inc. | Carbocyclic bicyclic nucleic acid analogs |
| US8278283B2 (en) | 2007-07-05 | 2012-10-02 | Isis Pharmaceuticals, Inc. | 6-disubstituted or unsaturated bicyclic nucleic acid analogs |
| US8314227B2 (en) | 2007-05-22 | 2012-11-20 | Marina Biotech, Inc. | Hydroxymethyl substituted RNA oligonucleotides and RNA complexes |
| WO2012177947A2 (en) | 2011-06-21 | 2012-12-27 | Alnylam Pharmaceuticals, Inc. | Compositions and methods for inhibition of expression of apolipoprotein c-iii (apoc3) genes |
| US20130004427A1 (en) | 2009-12-11 | 2013-01-03 | The Regents Of The University Of Michigan | Targeted dendrimer-drug conjugates |
| US20130011922A1 (en) | 2007-03-02 | 2013-01-10 | F/K/A Mdrna, Inc. | Nucleic acid compounds for inhibiting gene expression and uses thereof |
| WO2013033230A1 (en) | 2011-08-29 | 2013-03-07 | Isis Pharmaceuticals, Inc. | Oligomer-conjugate complexes and their use |
| WO2013036868A1 (en) | 2011-09-07 | 2013-03-14 | Marina Biotech Inc. | Synthesis and uses of nucleic acid compounds with conformationally restricted monomers |
| US20130109817A1 (en) | 2010-03-26 | 2013-05-02 | Mersana Therapeutics, Inc. | Modified Polymers for Delivery of Polynucleotides, Method of Manufacture, and Methods of Use Thereof |
| US20130121954A1 (en) | 2011-08-26 | 2013-05-16 | Arrowhead Madison Inc. | Poly(vinyl ester) Polymers for In Vivo Nucleic Acid Delivery |
| WO2013075035A1 (en) | 2011-11-18 | 2013-05-23 | Alnylam Pharmaceuticals | Rnai agents, compositions and methods of use thereof for treating transthyretin (ttr) associated diseases |
| US20130130378A1 (en) | 2010-04-22 | 2013-05-23 | Alnylam Pharmaceuticals, Inc. | Oligonucleotides comprising acyclic and abasic nucleosides and analogs |
| US20130190383A1 (en) | 2010-04-26 | 2013-07-25 | Marina Biotech, Inc. | Nucleic acid compounds with conformationally restricted monomers and uses thereof |
| US20130203836A1 (en) | 2010-04-01 | 2013-08-08 | Isis Pharmaceuticals, Inc. | 2' and 5' modified monomers and oligonucleotides |
| US8541548B2 (en) | 1999-06-07 | 2013-09-24 | Arrowhead Madison Inc. | Compounds and methods for reversible modification of biologically active molecules |
| WO2013166121A1 (en) | 2012-05-02 | 2013-11-07 | Merck Sharp & Dohme Corp. | Novel tetragalnac containing conjugates and methods for delivery of oligonucleotides |
| WO2013165816A2 (en) | 2012-05-02 | 2013-11-07 | Merck Sharp & Dohme Corp. | SHORT INTERFERING NUCLEIC ACID (siNA) COMPOSITIONS |
| US8790919B2 (en) | 2004-03-15 | 2014-07-29 | Isis Pharmaceuticals, Inc. | Compositions and methods for optimizing cleavage of RNA by RNase H |
| US8828957B2 (en) | 2003-12-11 | 2014-09-09 | Microvax, Llc | Methods for generating immunity to antigen |
| WO2014179620A1 (en) | 2013-05-01 | 2014-11-06 | Isis Pharmaceuticals, Inc. | Conjugated antisense compounds and their use |
| WO2014179445A1 (en) | 2013-05-01 | 2014-11-06 | Regulus Therapeutics Inc. | Compounds and methods for enhanced cellular uptake |
| WO2015106128A2 (en) | 2014-01-09 | 2015-07-16 | Alnylam Pharmaceuticals, Inc. | MODIFIED RNAi AGENTS |
| WO2017015555A1 (en) | 2015-07-22 | 2017-01-26 | Wave Life Sciences Ltd. | Oligonucleotide compositions and methods thereof |
| US10668170B2 (en) | 2011-11-18 | 2020-06-02 | Alnylam Pharmaceuticals, Inc. | Modified RNAi agents |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009045536A2 (en) * | 2007-10-05 | 2009-04-09 | The University Of North Carolina At Chapel Hill | Receptor targeted oligonucleotides |
| TW202246503A (en) * | 2013-07-19 | 2022-12-01 | 美商百健Ma公司 | Compositions for modulating tau expression |
| JOP20200228A1 (en) * | 2015-12-21 | 2017-06-16 | Novartis Ag | Compositions and methods for decreasing tau expression |
| CA3171246A1 (en) * | 2020-03-18 | 2021-09-23 | University Of Massachusetts | Oligonucleotides for mapt modulation |
| WO2021202511A2 (en) * | 2020-03-30 | 2021-10-07 | Alnylam Pharmaceuticals, Inc. | MICROTUBULE ASSOCIATED PROTEIN TAU (MAPT) iRNA AGENT COMPOSITIONS AND METHODS OF USE THEREOF |
| MX2024003519A (en) * | 2021-09-24 | 2024-04-01 | Alnylam Pharmaceuticals Inc | Microtubule associated protein tau (mapt) irna agent compositions and methods of use thereof. |
| WO2023154896A2 (en) * | 2022-02-11 | 2023-08-17 | Adarx Pharmaceuticals, Inc. | Trkb ligand conjugated compounds and uses thereof |
| CN119136797A (en) * | 2022-03-01 | 2024-12-13 | 阿达尔克斯制药有限公司 | CB1 ligand-conjugated compounds and uses thereof |
| EP4504196A1 (en) * | 2022-04-04 | 2025-02-12 | Adarx Pharmaceuticals, Inc. | Alpha4beta1/7 integrin ligand conjugated compounds and uses thereof |
-
2024
- 2024-04-19 WO PCT/US2024/025577 patent/WO2024220930A2/en active Pending
- 2024-04-19 TW TW113114826A patent/TW202448484A/en unknown
- 2024-04-19 KR KR1020257038056A patent/KR20260007584A/en active Pending
- 2024-04-19 CN CN202480033384.9A patent/CN121285630A/en active Pending
- 2024-04-19 AU AU2024258561A patent/AU2024258561A1/en active Pending
-
2025
- 2025-10-13 IL IL323920A patent/IL323920A/en unknown
- 2025-10-17 MX MX2025012458A patent/MX2025012458A/en unknown
Patent Citations (191)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3687808A (en) | 1969-08-14 | 1972-08-29 | Univ Leland Stanford Junior | Synthetic polynucleotides |
| US5118800A (en) | 1983-12-20 | 1992-06-02 | California Institute Of Technology | Oligonucleotides possessing a primary amino group in the terminal nucleotide |
| US4981957A (en) | 1984-07-19 | 1991-01-01 | Centre National De La Recherche Scientifique | Oligonucleotides with modified phosphate and modified carbohydrate moieties at the respective chain termini |
| US5367066A (en) | 1984-10-16 | 1994-11-22 | Chiron Corporation | Oligonucleotides with selectably cleavable and/or abasic sites |
| US4845205A (en) | 1985-01-08 | 1989-07-04 | Institut Pasteur | 2,N6 -disubstituted and 2,N6 -trisubstituted adenosine-3'-phosphoramidites |
| US4751219A (en) | 1985-02-05 | 1988-06-14 | Nederlandse Centrale Organisatie Voor Toegepast-Natuur-Wetenschappelijk Onderzoek | Synthetic glycolipides, a process for the preparation thereof and several uses for these synthetic glycolipides |
| US5185444A (en) | 1985-03-15 | 1993-02-09 | Anti-Gene Deveopment Group | Uncharged morpolino-based polymers having phosphorous containing chiral intersubunit linkages |
| US5552540A (en) | 1987-06-24 | 1996-09-03 | Howard Florey Institute Of Experimental Physiology And Medicine | Nucleoside derivatives |
| US5175273A (en) | 1988-07-01 | 1992-12-29 | Genentech, Inc. | Nucleic acid intercalating agents |
| US5134066A (en) | 1989-08-29 | 1992-07-28 | Monsanto Company | Improved probes using nucleosides containing 3-dezauracil analogs |
| US5591722A (en) | 1989-09-15 | 1997-01-07 | Southern Research Institute | 2'-deoxy-4'-thioribonucleosides and their antiviral activity |
| US5466786A (en) | 1989-10-24 | 1995-11-14 | Gilead Sciences | 2'modified nucleoside and nucleotide compounds |
| US5466786B1 (en) | 1989-10-24 | 1998-04-07 | Gilead Sciences | 2' Modified nucleoside and nucleotide compounds |
| US7495088B1 (en) | 1989-12-04 | 2009-02-24 | Enzo Life Sciences, Inc. | Modified nucleotide compounds |
| US5130302A (en) | 1989-12-20 | 1992-07-14 | Boron Bilogicals, Inc. | Boronated nucleoside, nucleotide and oligonucleotide compounds, compositions and methods for using same |
| US5166315A (en) | 1989-12-20 | 1992-11-24 | Anti-Gene Development Group | Sequence-specific binding polymers for duplex nucleic acids |
| US6166199A (en) | 1990-01-11 | 2000-12-26 | Isis Pharmaceuticals, Inc | N-2 substituted purines |
| US5670633A (en) | 1990-01-11 | 1997-09-23 | Isis Pharmaceuticals, Inc. | Sugar modified oligonucleotides that detect and modulate gene expression |
| US5646265A (en) | 1990-01-11 | 1997-07-08 | Isis Pharmceuticals, Inc. | Process for the preparation of 2'-O-alkyl purine phosphoramidites |
| US5459255A (en) | 1990-01-11 | 1995-10-17 | Isis Pharmaceuticals, Inc. | N-2 substituted purines |
| US5808027A (en) | 1990-01-11 | 1998-09-15 | Isis Pharmaceuticals, Inc. | N-2 substituted purines in oligonucleotides |
| US5457191A (en) | 1990-01-11 | 1995-10-10 | Isis Pharmaceuticals, Inc. | 3-deazapurines |
| US5750692A (en) | 1990-01-11 | 1998-05-12 | Isis Pharmaceuticals, Inc. | Synthesis of 3-deazapurines |
| US5587470A (en) | 1990-01-11 | 1996-12-24 | Isis Pharmaceuticals, Inc. | 3-deazapurines |
| US5587469A (en) | 1990-01-11 | 1996-12-24 | Isis Pharmaceuticals, Inc. | Oligonucleotides containing N-2 substituted purines |
| US5681941A (en) | 1990-01-11 | 1997-10-28 | Isis Pharmaceuticals, Inc. | Substituted purines and oligonucleotide cross-linking |
| US5567811A (en) | 1990-05-03 | 1996-10-22 | Amersham International Plc | Phosphoramidite derivatives, their preparation and the use thereof in the incorporation of reporter groups on synthetic oligonucleotides |
| US5514785A (en) | 1990-05-11 | 1996-05-07 | Becton Dickinson And Company | Solid supports for nucleic acid hybridization assays |
| US5614617A (en) | 1990-07-27 | 1997-03-25 | Isis Pharmaceuticals, Inc. | Nuclease resistant, pyrimidine modified oligonucleotides that detect and modulate gene expression |
| US5432272A (en) | 1990-10-09 | 1995-07-11 | Benner; Steven A. | Method for incorporating into a DNA or RNA oligonucleotide using nucleotides bearing heterocyclic bases |
| US5948903A (en) | 1991-01-11 | 1999-09-07 | Isis Pharmaceuticals, Inc. | Synthesis of 3-deazapurines |
| US5319080A (en) | 1991-10-17 | 1994-06-07 | Ciba-Geigy Corporation | Bicyclic nucleosides, oligonucleotides, process for their preparation and intermediates |
| US5393878A (en) | 1991-10-17 | 1995-02-28 | Ciba-Geigy Corporation | Bicyclic nucleosides, oligonucleotides, process for their preparation and intermediates |
| US5594121A (en) | 1991-11-07 | 1997-01-14 | Gilead Sciences, Inc. | Enhanced triple-helix and double-helix formation with oligomers containing modified purines |
| US6380368B1 (en) | 1991-11-26 | 2002-04-30 | Isis Pharmaceuticals, Inc. | Enhanced triple-helix and double-helix formation with oligomers containing modified pyrimidines |
| US5645985A (en) | 1991-11-26 | 1997-07-08 | Gilead Sciences, Inc. | Enhanced triple-helix and double-helix formation with oligomers containing modified pyrimidines |
| US5484908A (en) | 1991-11-26 | 1996-01-16 | Gilead Sciences, Inc. | Oligonucleotides containing 5-propynyl pyrimidines |
| US6235887B1 (en) | 1991-11-26 | 2001-05-22 | Isis Pharmaceuticals, Inc. | Enhanced triple-helix and double-helix formation directed by oligonucleotides containing modified pyrimidines |
| US5830653A (en) | 1991-11-26 | 1998-11-03 | Gilead Sciences, Inc. | Methods of using oligomers containing modified pyrimidines |
| US5359044A (en) | 1991-12-13 | 1994-10-25 | Isis Pharmaceuticals | Cyclobutyl oligonucleotide surrogates |
| US5639873A (en) | 1992-02-05 | 1997-06-17 | Centre National De La Recherche Scientifique (Cnrs) | Oligothionucleotides |
| US5434257A (en) | 1992-06-01 | 1995-07-18 | Gilead Sciences, Inc. | Binding compentent oligomers containing unsaturated 3',5' and 2',5' linkages |
| US5610300A (en) | 1992-07-01 | 1997-03-11 | Ciba-Geigy Corporation | Carbocyclic nucleosides containing bicyclic rings, oligonucleotides therefrom, process for their preparation, their use and intermediates |
| US5700920A (en) | 1992-07-01 | 1997-12-23 | Novartis Corporation | Carbocyclic nucleosides containing bicyclic rings, oligonucleotides therefrom, process for their preparation, their use and intermediates |
| US5576427A (en) | 1993-03-30 | 1996-11-19 | Sterling Winthrop, Inc. | Acyclic nucleoside analogs and oligonucleotide sequences containing them |
| US5658873A (en) | 1993-04-10 | 1997-08-19 | Degussa Aktiengesellschaft | Coated sodium percarbonate particles, a process for their production and detergent, cleaning and bleaching compositions containing them |
| US6015886A (en) | 1993-05-24 | 2000-01-18 | Chemgenes Corporation | Oligonucleotide phosphate esters |
| US6005096A (en) | 1993-09-17 | 1999-12-21 | Gilead Sciences, Inc. | Pyrimidine derivatives |
| US5502177A (en) | 1993-09-17 | 1996-03-26 | Gilead Sciences, Inc. | Pyrimidine derivatives for labeled binding partners |
| US5763588A (en) | 1993-09-17 | 1998-06-09 | Gilead Sciences, Inc. | Pyrimidine derivatives for labeled binding partners |
| US5457187A (en) | 1993-12-08 | 1995-10-10 | Board Of Regents University Of Nebraska | Oligonucleotides containing 5-fluorouracil |
| US5446137A (en) | 1993-12-09 | 1995-08-29 | Syntex (U.S.A.) Inc. | Oligonucleotides containing 4'-substituted nucleotides |
| US5446137B1 (en) | 1993-12-09 | 1998-10-06 | Behringwerke Ag | Oligonucleotides containing 4'-substituted nucleotides |
| US5519134A (en) | 1994-01-11 | 1996-05-21 | Isis Pharmaceuticals, Inc. | Pyrrolidine-containing monomers and oligomers |
| US5811534A (en) | 1994-02-01 | 1998-09-22 | Isis Pharmaceuticals, Inc. | Substituted purines and oligonucleotide cross-linking |
| US5596091A (en) | 1994-03-18 | 1997-01-21 | The Regents Of The University Of California | Antisense oligonucleotides comprising 5-aminoalkyl pyrimidine nucleotides |
| US5627053A (en) | 1994-03-29 | 1997-05-06 | Ribozyme Pharmaceuticals, Inc. | 2'deoxy-2'-alkylnucleotide containing nucleic acid |
| US5525711A (en) | 1994-05-18 | 1996-06-11 | The United States Of America As Represented By The Secretary Of The Department Of Health And Human Services | Pteridine nucleotide analogs as fluorescent DNA probes |
| US5597909A (en) | 1994-08-25 | 1997-01-28 | Chiron Corporation | Polynucleotide reagents containing modified deoxyribose moieties, and associated methods of synthesis and use |
| US6908903B1 (en) | 1994-12-07 | 2005-06-21 | Aletheon Pharmaceuticals, Inc. | Cluster clearing agents |
| US6166197A (en) | 1995-03-06 | 2000-12-26 | Isis Pharmaceuticals, Inc. | Oligomeric compounds having pyrimidine nucleotide (S) with 2'and 5 substitutions |
| US6222025B1 (en) | 1995-03-06 | 2001-04-24 | Isis Pharmaceuticals, Inc. | Process for the synthesis of 2′-O-substituted pyrimidines and oligomeric compounds therefrom |
| US20060183886A1 (en) | 1995-11-22 | 2006-08-17 | Cell Works Therapeutics, Inc., A Delaware Corporation | Ligands to enhance cellular uptake of biomolecules |
| US20030119724A1 (en) | 1995-11-22 | 2003-06-26 | Ts`O Paul O.P. | Ligands to enhance cellular uptake of biomolecules |
| WO1997020563A1 (en) | 1995-11-22 | 1997-06-12 | The Johns-Hopkins University | Ligands to enhance cellular uptake of biomolecules |
| US5994517A (en) | 1995-11-22 | 1999-11-30 | Paul O. P. Ts'o | Ligands to enhance cellular uptake of biomolecules |
| WO1997046098A1 (en) | 1996-06-06 | 1997-12-11 | Neorx Corporation | Cluster clearing agents |
| US6620916B1 (en) | 1996-09-26 | 2003-09-16 | Ajinomoto Co., Inc. | Modified physiologically active proteins and medicinal compositions containing the same |
| WO1998013381A1 (en) | 1996-09-26 | 1998-04-02 | Ajinomoto Co., Inc. | Modified physiologically active proteins and medicinal compositions containing the same |
| US6639062B2 (en) | 1997-02-14 | 2003-10-28 | Isis Pharmaceuticals, Inc. | Aminooxy-modified nucleosidic compounds and oligomeric compounds prepared therefrom |
| US6268490B1 (en) | 1997-03-07 | 2001-07-31 | Takeshi Imanishi | Bicyclonucleoside and oligonucleotide analogues |
| US6770748B2 (en) | 1997-03-07 | 2004-08-03 | Takeshi Imanishi | Bicyclonucleoside and oligonucleotide analogue |
| WO1999014226A2 (en) | 1997-09-12 | 1999-03-25 | Exiqon A/S | Bi- and tri-cyclic nucleoside, nucleotide and oligonucleotide analogues |
| US7034133B2 (en) | 1997-09-12 | 2006-04-25 | Exiqon A/S | Oligonucleotide analogues |
| US6670461B1 (en) | 1997-09-12 | 2003-12-30 | Exiqon A/S | Oligonucleotide analogues |
| US6794499B2 (en) | 1997-09-12 | 2004-09-21 | Exiqon A/S | Oligonucleotide analogues |
| US6528640B1 (en) | 1997-11-05 | 2003-03-04 | Ribozyme Pharmaceuticals, Incorporated | Synthetic ribonucleic acids with RNAse activity |
| US6617438B1 (en) | 1997-11-05 | 2003-09-09 | Sirna Therapeutics, Inc. | Oligoribonucleotides with enzymatic activity |
| US7045610B2 (en) | 1998-04-03 | 2006-05-16 | Epoch Biosciences, Inc. | Modified oligonucleotides for mismatch discrimination |
| US6525031B2 (en) | 1998-06-16 | 2003-02-25 | Isis Pharmaceuticals, Inc. | Targeted Oligonucleotide conjugates |
| US6300319B1 (en) | 1998-06-16 | 2001-10-09 | Isis Pharmaceuticals, Inc. | Targeted oligonucleotide conjugates |
| US6660720B2 (en) | 1998-06-16 | 2003-12-09 | Isis Pharmaceuticals, Inc. | Targeted oligonucleotide conjugates |
| US7084125B2 (en) | 1999-03-18 | 2006-08-01 | Exiqon A/S | Xylo-LNA analogues |
| US7053207B2 (en) | 1999-05-04 | 2006-05-30 | Exiqon A/S | L-ribo-LNA analogues |
| US6525191B1 (en) | 1999-05-11 | 2003-02-25 | Kanda S. Ramasamy | Conformationally constrained L-nucleosides |
| US6383812B1 (en) | 1999-05-28 | 2002-05-07 | Academia Sinica | Anti liver disease drug R-YEEE and method of synthesizing branched galactose-terminal glycoproteins |
| US8541548B2 (en) | 1999-06-07 | 2013-09-24 | Arrowhead Madison Inc. | Compounds and methods for reversible modification of biologically active molecules |
| US6147200A (en) | 1999-08-19 | 2000-11-14 | Isis Pharmaceuticals, Inc. | 2'-O-acetamido modified monomers and oligomers |
| US6998484B2 (en) | 2000-10-04 | 2006-02-14 | Santaris Pharma A/S | Synthesis of purine locked nucleic acid analogues |
| US7262177B2 (en) | 2000-12-01 | 2007-08-28 | Cell Works Therapeutics, Inc. | Conjugates of glycosylated/galactosylated peptide, bifunctional linker, and nucleotidic monomers/polymers, and related compositions and methods of use |
| US6906182B2 (en) | 2000-12-01 | 2005-06-14 | Cell Works Therapeutics, Inc. | Conjugates of glycosylated/galactosylated peptide, bifunctional linker, and nucleotidic monomers/polymers, and related compositions and method of use |
| WO2002043771A2 (en) | 2000-12-01 | 2002-06-06 | Cell Works Inc. | Conjugates of glycosylated/galactosylated peptide |
| US20030077829A1 (en) | 2001-04-30 | 2003-04-24 | Protiva Biotherapeutics Inc.. | Lipid-based formulations |
| US7491805B2 (en) | 2001-05-18 | 2009-02-17 | Sirna Therapeutics, Inc. | Conjugates and compositions for cellular delivery |
| US20030158403A1 (en) | 2001-07-03 | 2003-08-21 | Isis Pharmaceuticals, Inc. | Nuclease resistant chimeric oligonucleotides |
| US20030175906A1 (en) | 2001-07-03 | 2003-09-18 | Muthiah Manoharan | Nuclease resistant chimeric oligonucleotides |
| WO2004024757A2 (en) | 2002-09-11 | 2004-03-25 | Santaris Pharma A/S | Modified pna molecules |
| US20080039618A1 (en) | 2002-11-05 | 2008-02-14 | Charles Allerson | Polycyclic sugar surrogate-containing oligomeric compounds and compositions for use in gene modulation |
| US20040171570A1 (en) | 2002-11-05 | 2004-09-02 | Charles Allerson | Polycyclic sugar surrogate-containing oligomeric compounds and compositions for use in gene modulation |
| US20080108801A1 (en) | 2003-04-17 | 2008-05-08 | Muthiah Manoharan | Lipophilic Conjugated iRNA Agents |
| US8344125B2 (en) | 2003-04-17 | 2013-01-01 | Alnylam Pharmaceuticals, Inc. | Modified iRNA agents |
| US7851615B2 (en) | 2003-04-17 | 2010-12-14 | Alnylam Pharmaceuticals, Inc. | Lipophilic conjugated iRNA agents |
| US7723509B2 (en) | 2003-04-17 | 2010-05-25 | Alnylam Pharmaceuticals | IRNA agents with biocleavable tethers |
| US20050164235A1 (en) | 2003-04-17 | 2005-07-28 | Muthiah Manoharan | Modified iRNA agents |
| WO2004101619A1 (en) | 2003-05-15 | 2004-11-25 | Shionogi Co., Ltd. | Rational design and synthesis of functional glycopeptide |
| US7427672B2 (en) | 2003-08-28 | 2008-09-23 | Takeshi Imanishi | Artificial nucleic acids of n-o bond crosslinkage type |
| US7939677B2 (en) | 2003-09-18 | 2011-05-10 | Isis Pharmaceuticals, Inc. | Oligomeric compounds comprising 4′-thionucleosides for use in gene modulation |
| US7875733B2 (en) | 2003-09-18 | 2011-01-25 | Isis Pharmaceuticals, Inc. | Oligomeric compounds comprising 4′-thionucleosides for use in gene modulation |
| US8828957B2 (en) | 2003-12-11 | 2014-09-09 | Microvax, Llc | Methods for generating immunity to antigen |
| US8790919B2 (en) | 2004-03-15 | 2014-07-29 | Isis Pharmaceuticals, Inc. | Compositions and methods for optimizing cleavage of RNA by RNase H |
| US7582744B2 (en) | 2004-08-10 | 2009-09-01 | Alnylam Pharmaceuticals, Inc. | Chemically modified oligonucleotides |
| US8404862B2 (en) | 2004-08-10 | 2013-03-26 | Alnylam Pharmaceuticals, Inc. | Ligand-conjugated monomers |
| US20090203132A1 (en) | 2004-09-09 | 2009-08-13 | Swayze Eric E | Pyrrolidinyl groups for attaching conjugates to oligomeric compounds |
| US20060148740A1 (en) | 2005-01-05 | 2006-07-06 | Prosensa B.V. | Mannose-6-phosphate receptor mediated gene transfer into muscle cells |
| US20080206869A1 (en) | 2005-01-24 | 2008-08-28 | Avaris Ab | Nucleic Acid Complex |
| US7741457B2 (en) | 2006-01-27 | 2010-06-22 | Isis Pharmaceuticals, Inc. | 6-modified bicyclic nucleic acid analogs |
| US20090012281A1 (en) | 2006-01-27 | 2009-01-08 | Isis Pharmaceuticals, Inc. | 6-modified bicyclic nucleic acid analogs |
| US7399845B2 (en) | 2006-01-27 | 2008-07-15 | Isis Pharmaceuticals, Inc. | 6-modified bicyclic nucleic acid analogs |
| US8022193B2 (en) | 2006-01-27 | 2011-09-20 | Isis Pharmaceuticals, Inc. | 6-modified bicyclic nucleic acid analogs |
| US7569686B1 (en) | 2006-01-27 | 2009-08-04 | Isis Pharmaceuticals, Inc. | Compounds and methods for synthesis of bicyclic nucleic acid analogs |
| US8030467B2 (en) | 2006-05-11 | 2011-10-04 | Isis Pharmaceuticals, Inc. | 5′-modified bicyclic nucleic acid analogs |
| US8137695B2 (en) | 2006-08-18 | 2012-03-20 | Arrowhead Madison Inc. | Polyconjugates for in vivo delivery of polynucleotides |
| US20120230938A1 (en) | 2006-08-18 | 2012-09-13 | Arrowhead Madison Inc. | Polyconjugates for In Vivo Delivery of Polynucleotides |
| WO2008101157A1 (en) | 2007-02-15 | 2008-08-21 | Isis Pharmaceuticals, Inc. | 5'-substituted-2'-f modified nucleosides and oligomeric compounds prepared therefrom |
| WO2008098788A2 (en) | 2007-02-16 | 2008-08-21 | Ktb Tumorforschungsgesellschaft Mbh | Receptor and antigen targeted prodrug |
| US20130011922A1 (en) | 2007-03-02 | 2013-01-10 | F/K/A Mdrna, Inc. | Nucleic acid compounds for inhibiting gene expression and uses thereof |
| US8314227B2 (en) | 2007-05-22 | 2012-11-20 | Marina Biotech, Inc. | Hydroxymethyl substituted RNA oligonucleotides and RNA complexes |
| US20130096289A1 (en) | 2007-05-22 | 2013-04-18 | Marina Biotech, Inc. | Hydroxymethyl substituted rna oligonucleotides and rna complexes |
| US8278425B2 (en) | 2007-05-30 | 2012-10-02 | Isis Pharmaceuticals, Inc. | N-substituted-aminomethylene bridged bicyclic nucleic acid analogs |
| US8278426B2 (en) | 2007-06-08 | 2012-10-02 | Isis Pharmaceuticals, Inc. | Carbocyclic bicyclic nucleic acid analogs |
| US8278283B2 (en) | 2007-07-05 | 2012-10-02 | Isis Pharmaceuticals, Inc. | 6-disubstituted or unsaturated bicyclic nucleic acid analogs |
| US8088904B2 (en) | 2007-08-15 | 2012-01-03 | Isis Pharmaceuticals, Inc. | Tetrahydropyran nucleic acid analogs |
| US8440803B2 (en) | 2007-08-15 | 2013-05-14 | Isis Pharmaceuticals, Inc. | Tetrahydropyran nucleic acid analogs |
| US9005906B2 (en) | 2007-08-15 | 2015-04-14 | Isis Pharmaceuticals, Inc. | Tetrahydropyran nucleic acid analogs |
| US8106022B2 (en) | 2007-12-04 | 2012-01-31 | Alnylam Pharmaceuticals, Inc. | Carbohydrate conjugates as delivery agents for oligonucleotides |
| US8450467B2 (en) | 2007-12-04 | 2013-05-28 | Alnylam Pharmaceuticals, Inc. | Carbohydrate conjugates as delivery agents for oligonucleotides |
| WO2009082607A2 (en) | 2007-12-04 | 2009-07-02 | Alnylam Pharmaceuticals, Inc. | Targeting lipids |
| WO2009073809A2 (en) | 2007-12-04 | 2009-06-11 | Alnylam Pharmaceuticals, Inc. | Carbohydrate conjugates as delivery agents for oligonucleotides |
| US20120136042A1 (en) | 2007-12-04 | 2012-05-31 | Alnylam Pharmaceuticals, Inc | Carbohydrate conjugates as delivery agents for oligonucleotides |
| WO2009134487A2 (en) | 2008-01-31 | 2009-11-05 | Alnylam Pharmaceuticals, Inc. | Optimized methods for delivery of dsrna targeting the pcsk9 gene |
| WO2009126933A2 (en) | 2008-04-11 | 2009-10-15 | Alnylam Pharmaceuticals, Inc. | Site-specific delivery of nucleic acids by combining targeting ligands with endosomolytic components |
| US20110123520A1 (en) | 2008-04-11 | 2011-05-26 | Alnylam Pharmaceuticals, Inc. | Site-specific delivery of nucleic acids by combining targeting ligands with endosomolytic components |
| US20120035115A1 (en) | 2008-09-23 | 2012-02-09 | Alnylam Pharmaceuticals, Inc. | Chemical modifications of monomers and oligonucleotides with cycloaddition |
| WO2010054406A1 (en) | 2008-11-10 | 2010-05-14 | Alnylam Pharmaceuticals, Inc. | Novel lipids and compositions for the delivery of therapeutics |
| US20120095075A1 (en) | 2008-11-10 | 2012-04-19 | Alnylam Pharmaceuticals, Inc. | Novel lipids and compositions for the delivery of therapeutics |
| US20110313020A1 (en) | 2008-12-03 | 2011-12-22 | Marina Biotech, Inc. | UsiRNA Complexes |
| US20120101148A1 (en) | 2009-01-29 | 2012-04-26 | Alnylam Pharmaceuticals, Inc. | lipid formulation |
| WO2010088537A2 (en) | 2009-01-29 | 2010-08-05 | Alnylam Pharmaceuticals, Inc. | Improved lipid formulation |
| WO2010129709A1 (en) | 2009-05-05 | 2010-11-11 | Alnylam Pharmaceuticals, Inc. | Lipid compositions |
| WO2010144740A1 (en) | 2009-06-10 | 2010-12-16 | Alnylam Pharmaceuticals, Inc. | Improved lipid formulation |
| US8158601B2 (en) | 2009-06-10 | 2012-04-17 | Alnylam Pharmaceuticals, Inc. | Lipid formulation |
| WO2010148013A2 (en) | 2009-06-15 | 2010-12-23 | Alnylam Pharmaceuticals, Inc. | Lipid formulated dsrna targeting the pcsk9 gene |
| US8552163B2 (en) | 2009-09-25 | 2013-10-08 | Johns Hopkins University | Liver-targeting agents and their synthesis |
| WO2011038356A2 (en) | 2009-09-25 | 2011-03-31 | Johns Hopkins University | Novel liver-targeting agents and their synthesis |
| US20110097264A1 (en) | 2009-10-26 | 2011-04-28 | Institute Of Nuclear Energy Research Atomic Energy Council, Executive Yuan | Radiolabeling method using multivalent glycoligands as hepatic receptor imaging agent |
| US20110097265A1 (en) | 2009-10-26 | 2011-04-28 | Institute Of Nuclear Energy Research Atomic Energy Council, Executive Yuan | Quantification method for remaining liver function and novel liver receptor imaging agent |
| US8435491B2 (en) | 2009-10-26 | 2013-05-07 | Institute Of Nuclear Energy Research Atomic Energy Council, Executive Yuan | Quantification method for remaining liver function and novel liver receptor imaging agent |
| US20130004427A1 (en) | 2009-12-11 | 2013-01-03 | The Regents Of The University Of Michigan | Targeted dendrimer-drug conjugates |
| WO2011100131A2 (en) | 2010-01-28 | 2011-08-18 | Alnylam Pharmacuticals, Inc. | Monomers and oligonucleotides comprising cycloaddition adduct(s) |
| US20110207799A1 (en) | 2010-02-24 | 2011-08-25 | Roche Madison Inc. | Compositions for Targeted Delivery of siRNA |
| US8313772B2 (en) | 2010-02-24 | 2012-11-20 | Arrowhead Madison Inc. | Compositions for targeted delivery of siRNA |
| US20130109817A1 (en) | 2010-03-26 | 2013-05-02 | Mersana Therapeutics, Inc. | Modified Polymers for Delivery of Polynucleotides, Method of Manufacture, and Methods of Use Thereof |
| WO2011120053A1 (en) | 2010-03-26 | 2011-09-29 | Mersana Therapeutics, Inc. | Modified polymers for delivery of polynucleotides, method of manufacture, and methods of use thereof |
| US8349308B2 (en) | 2010-03-26 | 2013-01-08 | Mersana Therapeutics, Inc. | Modified polymers for delivery of polynucleotides, method of manufacture, and methods of use thereof |
| US20130203836A1 (en) | 2010-04-01 | 2013-08-08 | Isis Pharmaceuticals, Inc. | 2' and 5' modified monomers and oligonucleotides |
| US20130130378A1 (en) | 2010-04-22 | 2013-05-23 | Alnylam Pharmaceuticals, Inc. | Oligonucleotides comprising acyclic and abasic nucleosides and analogs |
| US20130190383A1 (en) | 2010-04-26 | 2013-07-25 | Marina Biotech, Inc. | Nucleic acid compounds with conformationally restricted monomers and uses thereof |
| WO2011163121A1 (en) | 2010-06-21 | 2011-12-29 | Alnylam Pharmaceuticals, Inc. | Multifunctional copolymers for nucleic acid delivery |
| WO2012037254A1 (en) | 2010-09-15 | 2012-03-22 | Alnylam Pharmaceuticals, Inc. | MODIFIED iRNA AGENTS |
| WO2012068187A1 (en) | 2010-11-19 | 2012-05-24 | Merck Sharp & Dohme Corp. | Poly(amide) polymers for the delivery of oligonucleotides |
| WO2012083185A2 (en) | 2010-12-17 | 2012-06-21 | Arrowhead Research Corporations | Peptide-based in vivo sirna delivery system |
| WO2012083046A2 (en) | 2010-12-17 | 2012-06-21 | Arrowhead Research Corporation | Galactose cluster-pharmacokinetic modulator targeting moiety for sirna |
| US20120165393A1 (en) | 2010-12-17 | 2012-06-28 | Arrowhead Madison Inc. | Peptide-Based In Vivo siRNA Delivery System |
| US8501930B2 (en) | 2010-12-17 | 2013-08-06 | Arrowhead Madison Inc. | Peptide-based in vivo siRNA delivery system |
| WO2012089602A1 (en) | 2010-12-29 | 2012-07-05 | F. Hoffmann-La Roche Ag | Small molecule conjugates for intracellular delivery of biologically active compounds |
| WO2012089352A1 (en) | 2010-12-29 | 2012-07-05 | F. Hoffmann-La Roche Ag | Small molecule conjugates for intracellular delivery of nucleic acids |
| WO2012177947A2 (en) | 2011-06-21 | 2012-12-27 | Alnylam Pharmaceuticals, Inc. | Compositions and methods for inhibition of expression of apolipoprotein c-iii (apoc3) genes |
| US20130121954A1 (en) | 2011-08-26 | 2013-05-16 | Arrowhead Madison Inc. | Poly(vinyl ester) Polymers for In Vivo Nucleic Acid Delivery |
| WO2013033230A1 (en) | 2011-08-29 | 2013-03-07 | Isis Pharmaceuticals, Inc. | Oligomer-conjugate complexes and their use |
| WO2013036868A1 (en) | 2011-09-07 | 2013-03-14 | Marina Biotech Inc. | Synthesis and uses of nucleic acid compounds with conformationally restricted monomers |
| US10668170B2 (en) | 2011-11-18 | 2020-06-02 | Alnylam Pharmaceuticals, Inc. | Modified RNAi agents |
| WO2013075035A1 (en) | 2011-11-18 | 2013-05-23 | Alnylam Pharmaceuticals | Rnai agents, compositions and methods of use thereof for treating transthyretin (ttr) associated diseases |
| WO2013166121A1 (en) | 2012-05-02 | 2013-11-07 | Merck Sharp & Dohme Corp. | Novel tetragalnac containing conjugates and methods for delivery of oligonucleotides |
| WO2013165816A2 (en) | 2012-05-02 | 2013-11-07 | Merck Sharp & Dohme Corp. | SHORT INTERFERING NUCLEIC ACID (siNA) COMPOSITIONS |
| WO2014179445A1 (en) | 2013-05-01 | 2014-11-06 | Regulus Therapeutics Inc. | Compounds and methods for enhanced cellular uptake |
| US9127276B2 (en) | 2013-05-01 | 2015-09-08 | Isis Pharmaceuticals, Inc. | Conjugated antisense compounds and their use |
| US9181549B2 (en) | 2013-05-01 | 2015-11-10 | Isis Pharmaceuticals, Inc. | Conjugated antisense compounds and their use |
| WO2014179620A1 (en) | 2013-05-01 | 2014-11-06 | Isis Pharmaceuticals, Inc. | Conjugated antisense compounds and their use |
| US10844379B2 (en) | 2013-05-01 | 2020-11-24 | Ionis Pharmaceuticals, Inc. | Conjugated antisense compounds and their use |
| WO2015106128A2 (en) | 2014-01-09 | 2015-07-16 | Alnylam Pharmaceuticals, Inc. | MODIFIED RNAi AGENTS |
| WO2017015555A1 (en) | 2015-07-22 | 2017-01-26 | Wave Life Sciences Ltd. | Oligonucleotide compositions and methods thereof |
Non-Patent Citations (66)
| Title |
|---|
| "Biotechnology and Medicine", 2008, WILEY-VCH, article "Modified Nucleosides in Biochemistry" |
| "GenBank", Database accession no. NM_001377265.1 |
| BERGE ET AL.: "Pharmaceutical Salts", JOURNAL OF PHARMACEUTICAL SCIENCE, vol. 66, 1977, pages 1 - 19, XP002675560, DOI: 10.1002/jps.2600660104 |
| BIESSEN ET AL., FASEB, vol. 14, 2000, pages 1784 - 1792 |
| BIESSEN ET AL., J MED CHEM, vol. 38, 1995, pages 1846 - 1852 |
| BIESSEN ET AL., J. MED. CHEM., vol. 38, 1995, pages 1846 - 1852 |
| CHATTOPADHYAYA, J. ORG. CHEM., vol. 74, 2009, pages 118 - 134 |
| CHIBA: "Identified a morpholinyl-4-piperidinylacetic acid derivative as a potent oral active VLA-4 antagonist", BIOORG. & MED. CHEM. LETT., vol. 15, no. 1, pages 41 - 45, XP025313466, DOI: 10.1016/j.bmcl.2004.10.041 |
| CONNOLLY ET AL., J BIOL CHEM, vol. 257, 1982, pages 939 - 945 |
| CROOKE ET AL., J. PHARMACOL. EXP. THER., vol. 277, 1996, pages 923 - 937 |
| DUFF ET AL., METHODS ENZYMOL, vol. 313, 2000, pages 297 - 321 |
| ENGLISCH ET AL., ANGEWANDTE CHEMIE, vol. 30, 1991, pages 613 |
| GONG: "Synthesis and Biological Evaluation of Novel Pyridazinone-Based α4 Integrin Receptor Antagonists", J. MED. CHEM, vol. 49, no. 11, 2006, pages 3402 - 3411 |
| J. MED. CHEM., vol. 61, no. 22, 2018, pages 10276 - 10298 |
| JAYAPRAKASH ET AL., ORG LETT, vol. 12, 2010, pages 5410 - 5413 |
| KABANOV ET AL., FEBS LETT., vol. 259, 1990, pages 327 - 330 |
| KATO ET AL., GLYCOHIOL, vol. 11, 2001, pages 821 - 829 |
| KHOREV ET AL., BIOORG MED CHEM, vol. 16, 2008, pages 5216 - 5231 |
| KIM ET AL., TETRAHEDRON LETT, vol. 38, 1997, pages 3487 - 3490 |
| KORNILOVA ET AL., ANALYT BIOCHEM, vol. 425, 2012, pages 43 - 46 |
| KUMAR ET AL., ORG. BIOMOL. CHEM., vol. 11, 2013, pages 5853 - 5865 |
| LEE ET AL., BIOCHEM, vol. 23, 1984, pages 4255 - 4261 |
| LEE ET AL., BIOORG MED CHEM LETT, vol. 16, no. 19, 2006, pages 5132 - 5135 |
| LEE ET AL., BIOORG MED CHEM, vol. 19, 2011, pages 2494 - 2500 |
| LEE ET AL., BIOORGANIC & MEDICINAL CHEMISTRY, vol. 19, 2011, pages 2494 - 2500 |
| LEE ET AL., GLYCOCONJUGATE J, vol. 4, 1987, pages 317 - 328 |
| LEE ET AL., J ORG CHEM, vol. 77, 2012, pages 7564 - 7571 |
| LEE ET AL., METHODS ENZYMOL, vol. 362, 2003, pages 38 - 43 |
| LEE, CARHOHYDR RES, vol. 67, 1978, pages 509 - 514 |
| LETSINGER ET AL., PROC. NATL. ACID. SCI. USA, vol. 86, 1989, pages 6553 - 6556 |
| MAIER ET AL., BIOCONJUG CHEM, vol. 14, 2003, pages 18 - 29 |
| MAIER ET AL., BIOCONJUGATE CHEMISTRY, vol. 14, 2003, pages 18 - 29 |
| MAIERHOFER ET AL., BIOORG MED CHEM, vol. 15, 2007, pages 7661 - 7676 |
| MANOHARAN ET AL., ANN. NY. ACAD. SCI., vol. 660, 1992, pages 306 - 309 |
| MANOHARAN ET AL., BIORG. MED. CHEM. LET., vol. 3, 1993, pages 2765 - 2770 |
| MANOHARAN ET AL., BIORG. MED. CHEM. LET., vol. 4, 1994, pages 1053 - 1060 |
| MANOHARAN ET AL., NUCLEOSIDES & NUCLEOTIDES, vol. 14, 1995, pages 969 - 973 |
| MANOHARAN ET AL., TETRAHEDRON LETT., vol. 36, 1995, pages 3651 - 3654 |
| MANOHARAN, ANTISENSE NUCLEIC ACID DRUG DEV, vol. 12, 2002, pages 103 - 128 |
| MERWIN ET AL., BIOCONJUG CHEM, vol. 5, 1994, pages 612 - 620 |
| MISHRA ET AL., BIOCHIM. BIOPHYS. ACTA, vol. 1264, 1995, pages 229 - 237 |
| NISHINA ET AL., MOLECULAR THERAPY NUCLEIC ACIDS, vol. 4, 2015, pages e220 |
| NISHINA ET AL., MOLECULAR THERAPY, vol. 16, 2008, pages 734 - 166,442-443 |
| OBERHAUSER ET AL., NUCL. ACIDS RES., vol. 20, 1992, pages 533 - 538 |
| OKA ET AL., JACS, vol. 125, 2003, pages 8307 - 65 |
| PAVIA ET AL., INT J PEP PROTEIN RES, vol. 22, 1983, pages 539 - 548 |
| PUJOL ET AL., ANGEW CHEMIE INT ED ENGL, vol. 51, 2012, pages 7445 - 7448 |
| RAJUR ET AL., BIOCONJUG CHEM, vol. 8, 1997, pages 935 - 940 |
| RENSEN ET AL., ARTERIOSCLER THROMH VASE BIOL, vol. 26, 2006, pages 169 - 175 |
| RENSEN ET AL., J BIOL CHEM, vol. 276, 2001, pages 37577 - 37584 |
| RENSEN ET AL., J MED CHEM, vol. 47, 2004, pages 5798 - 5808 |
| RENSEN ET AL., J. BIOL. CHEM., vol. 276, 2001, pages 37577 - 37584 |
| RENSEN ET AL., J. MED. CHEM., vol. 47, 2004, pages 5798 - 5808 |
| SAISON-BEHMOARAS ET AL., EMBO J, vol. 10, 1991, pages 1111 - 1118 |
| SATO ET AL., JAM CHEM SOC, vol. 126, 2004, pages 14013 - 14022 |
| SHEA ET AL., NUCL. ACIDS RES., vol. 18, 1990, pages 3777 - 3783 |
| SLIEDREGT ET AL., J MED CHEM, vol. 42, 1999, pages 609 - 618 |
| SLIEDREGT ET AL., J. MED. CHEM., vol. 42, 1999, pages 609 - 618 |
| SVINARCHUK ET AL., BIOCHIMIE, vol. 75, 1993, pages 49 - 54 |
| TOLOMELLI: "Dehydro-β-proline Containing aa(3i Integrin Antagonists: Stereochemical Recognition in Ligand-Receptor Interplay", ACS MED. CHEM. LETT., vol. 6, no. 6, 2015, pages 701 - 706 |
| TOMIYA ET AL., BIOORG MED CHEM, vol. 21, 2013, pages 5275 - 5281 |
| TOYOKUNI ET AL., TETRAHEDRON LETT, vol. 31, 1990, pages 2673 - 2676 |
| VALENTIJN ET AL., TETRAHEDRON, vol. 53, 1997, pages 759 - 770 |
| VAN ROSSENBERG ET AL., GENE THER, vol. 11, 2004, pages 457 - 464 |
| WAN ET AL., NUC. ACID. RES., vol. 42, 2014, pages 13456 |
| WESTERLIND ET AL., GLYCOCONJ J, vol. 21, 2004, pages 227 - 241 |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2024258561A1 (en) | 2025-10-30 |
| WO2024220930A3 (en) | 2024-12-26 |
| MX2025012458A (en) | 2026-01-07 |
| KR20260007584A (en) | 2026-01-14 |
| IL323920A (en) | 2025-12-01 |
| WO2024220930A8 (en) | 2025-11-06 |
| CN121285630A (en) | 2026-01-06 |
| TW202448484A (en) | 2024-12-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| TW202146650A (en) | Oligonucleotide compositions and methods thereof | |
| EP3359523A2 (en) | Oligonucleotide compositions and methods thereof | |
| WO2019182037A1 (en) | Antisense oligonucleotide having reduced toxicity | |
| CN119256083A (en) | Compositions for regulating complement factor B and methods of use thereof | |
| CN111902537A (en) | Modulators of DNM2 expression | |
| KR20240099244A (en) | Angiotensinogen regulating composition | |
| TW202333750A (en) | Rnai oligonucleotide conjugates | |
| CN121294438A (en) | Advanced RNA Targeting (ARNATAR) | |
| CN119137272A (en) | Angiotensinogen modulating compositions and methods of use thereof | |
| WO2021054370A1 (en) | Nucleic acid complex | |
| WO2024220930A2 (en) | Mapt-modulating compositions and methods of use thereof | |
| WO2024249328A2 (en) | Sod1-modulating compositions and methods of use thereof | |
| WO2012165616A1 (en) | OLIGONUCLEOTIDE DERIVATIVE, OLIGONUCLEOTIDE DERIVATIVE-CONTAINING PHARMACEUTICAL COMPOSITION FOR TREATMENT AND PHARMACEUTICAL COMPOSITION FOR DIAGNOSIS, AND OLIGONUCLEOTIDE DERIVATIVE FOR REGULATION OF miRNA FUNCTION | |
| KR20260019519A (en) | SOD1-modulating composition and method of use thereof | |
| US12454693B1 (en) | Nucleic acids for inhibiting expression of transferrin receptor 2 | |
| TW202506137A (en) | Lrrk2-modulating compositions and methods of use thereof | |
| TW202521694A (en) | Coagulation factor xi-modulating compositions and methods of use thereof | |
| CN118786133A (en) | Angiotensinogen regulating compositions and methods of use thereof | |
| CN118922540A9 (en) | Prekallikrein regulating composition and its usage | |
| HK40115145A (en) | Angiotensinogen-modulating compositions and methods of use thereof | |
| TW202335674A (en) | Lipid conjugation for targeting astrocytes of the central nervous system | |
| HK40034188A (en) | Single-stranded nucleic acid molecule, and production method therefor |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24729118 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: AU2024258561 Country of ref document: AU Ref document number: 825984 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 323920 Country of ref document: IL |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112025021275 Country of ref document: BR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2025/012458 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: P2025-03342 Country of ref document: AE |
|
| ENP | Entry into the national phase |
Ref document number: 2024258561 Country of ref document: AU Date of ref document: 20240419 Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 323920 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: KR1020257038056 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2024729118 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2024729118 Country of ref document: EP Effective date: 20251120 |
|
| ENP | Entry into the national phase |
Ref document number: 2024729118 Country of ref document: EP Effective date: 20251120 |
|
| ENP | Entry into the national phase |
Ref document number: 2024729118 Country of ref document: EP Effective date: 20251120 |
|
| ENP | Entry into the national phase |
Ref document number: 2024729118 Country of ref document: EP Effective date: 20251120 |
|
| WWP | Wipo information: published in national office |
Ref document number: MX/A/2025/012458 Country of ref document: MX |