METHODS^OF^DETERMINING^RELATIVE^UNPAIRED^GLYCAN^CONTENT^ CROSS^REFERENCE^TO^RELATED^APPLICATION^ [0001]^^^^ The^benefit^under^35^U.S.C.^§119(e)^of^U.S.^Provisional^Patent^Application^No.^63/576,448,^ filed^on^April^20,^2023,^is^hereby^claimed,^and^the^disclosure^thereof^is^hereby^incorporated^by^ reference^herein.^ BACKGROUND^ [0002]^^^^ Glycosylation^plays^a^role^in^multiple^cellular^functions,^including,^for^example,^protein^ folding,^quality^control,^molecular^trafficking^and^sorting,^and^cell^surface^receptor^interaction.^^The^ glycan^structure^of^a^protein^drug^affects^its^therapeutic^efficacy,^impacting^its^bioactivity,^ pharmacokinetics,^immunogenicity,^solubility,^and^in^vivo^clearance.^^Fc^glycoform^profiles^are^ product^quality^attributes^for^recombinant^antibodies,^since^they^directly^impact^the^clinical^efficacy^ and^pharmacokinetics^of^the^antibodies.^^See,^e.g.,^Reusch^and^Tejada,^Glycobiology^25(12):^1325^ 1334^(2015);^and^Boune^et^al.,^Antibodies^(Basel)^9(2):^22^(2020).^^^ [0003]^^^^ Given^their^importance,^the^glycan^structures^attached^to^recombinant^protein^drugs^are^ often^monitored^during^development^and^manufacturing^(Li^et^al.,^Front^Immunol^8:^1554^(2017).^^ Traditionally,^glycans^are^monitored^using^a^released^glycan^assay,^in^which^glycans^are^cleaved^from^ the^protein^backbone,^chromatographically^separated,^and^identified.^^Techniques^used^for^the^ analysis^of^immunoglobulin^glycosylation^are^reviewed^in^de^Haan^et^al.,^Glycobiology^30(4):^226^240^ (2020).^^The^glycan^population^of^IgG^therapeutics^is^innately^heterogenous^and^complex,^as^ glycosylation^requires^the^coordinated^functions^of^multiple^metabolic^enzymes^and^is^complicated^ by^the^linkage^of^sugar^isomers,^the^glycosylation^site^occupancy,^as^well^as^the^outer^arm^sugar^ addition,^which^can^include^one^or^more^of^fucose,^galactose,^bisecting^GlcNAc,^and^sialic^acid.^^ Further^diversification^of^the^antibody^glycan^population^is^due^to^the^asymmetry^of^the^two^N^ glycans^from^every^heavy^chain^and^the^involvement^of^random^pairing^of^two^different^heavy^chain^ glycans.^^^ [0004]^^^^ Even^when^successful,^the^current^methods^used^for^glycan^analysis^ignore^the^fact^that^ many^therapeutic^protein^molecules,^including^IgG^and^fusion^proteins^with^IgG^Fc^region,^contain^ two^glycosylated^Fc^chains.^^Because^the^current^commonly^used^methods^include^cleavage^of^the^ glycans^from^the^protein^backbone,^such^methods^fail^to^impart^knowledge^on^the^pairing^status^of^ the^glycan^species^within^individual^protein^molecules.^^For^example,^while^current^methods^may^be^ used^to^determine^the^overall^abundance^of^afucosylated^glycans^in^a^given^antibody^composition^ sample,^it^does^not^inform^the^level^of^antibodies^comprising^a^fully^afucosylated^pair^of^heavy^ chains^vs.^comprising^only^a^hemi^afucosylated^pair^of^heavy^chains.^^Such^information^on^pairing^ ^
status^of^the^protein^is^suggested^to^be^an^important^feature^of^an^antibody^composition^that^ influences^its^biological^activity^level^(see,^e.g.,^International^Patent^Application^Publication^No.^WO^ 2022/081824).^ [0005]^^^^ Glycan^pairing^analysis^is^challenging^due^to^the^size^of^the^molecules^and^the^highly^ heterogeneous^glycan^population.^^The^analysis^of^specifically^afucosylated^glycan^pairs^is^further^ complicated^by^the^low^abundance^of^afucosylated^and^high^mannose^glycans^relative^to^other^ glycans.^^Accordingly,^determining^the^abundance^of^afucosylated^glycan^pairs^and^teasing^out^the^ relative^abundance^of^fully^afucosylated^pairs^or^hemi^afucosylated^pairs^is^complex^and^time^ consuming.^^^ [0006]^^^^ Thus,^there^is^a^need^in^the^biopharmaceutical^industry^for^simple,^efficient^and^reliable^ methods^of^determining^unpaired^glycan^content^of^an^IgG^antibody^composition.^ SUMMARY^ [0007]^^^^ Presented^herein^are^data^demonstrating^the^feasibility^and^advantages^of^an^improved^ method^of^determining^the^relative^unpaired^glycan^content^of^an^IgG^antibody^composition.^^The^ method^includes^preparation^of^a^simplified^sample^and^the^analysis^thereof,^which,^ultimately,^ provides^for^an^easier,^faster,^and^more^accurate^way^of^determining^the^relative^unpaired^glycan^ content^of^an^IgG^antibody^composition.^^^ [0008]^^^^ Accordingly,^the^present^disclosure^provides^methods^of^determining^the^relative^unpaired^ glycan^content^of^an^IgG^antibody^composition.^^In^exemplary^embodiments,^the^method^comprises^ (a)^treating^the^IgG^antibody^composition^with^two^enzymes^to^form^a^mixture^of^Fab^fragments^and^ Fc^fragments,^wherein^one^enzyme^cleaves^an^antibody^heavy^chain^at^a^site^N^terminal^to^the^hinge^ region^disulfide^linkages^and^the^other^enzyme^cleaves^the^ɴ1,4^linkage^between^core^GlcNAc^ residues^to^form^Fc^fragments^each^comprising^a^pair^of^core^glycan^structures,^(b)^separating^the^ Fab^fragments^from^the^Fc^fragments,^and^(c)^quantifying^the^abundance^of^(i)^paired^afucosylated^Fc^ fragments,^(ii)^unpaired^afucosylated^Fc^fragments,^(iii)^paired^high^mannose^Fc^fragments,^and/or^ (iv)^unpaired^high^mannose^Fc^fragments,^to^determine^the^relative^unpaired^afucosylated^(AF)^ glycan^content^and^relative^unpaired^high^mannose^(HM)^glycan^content^of^the^IgG^antibody^ composition.^ [0009]^^^^ The^present^disclosure^also^provides^methods^of^preparing^an^IgG^antibody^composition^ having^an^ADCC^activity^within^a^target^range,^e.g.,^predetermined^target^range.^^In^exemplary^ embodiments,^the^method^comprises^determining^the^relative^unpaired^glycan^content^of^a^sample^ of^the^IgG^antibody^composition^in^accordance^with^any^one^of^the^presently^disclosed^methods^of^ determining^the^relative^unpaired^glycan^content.^^The^relative^unpaired^AF^glycan^content^and/or^ ^
relative^unpaired^HM^glycan^content^of^the^IgG^antibody^composition^may^be^compared^to^a^target^ range^of^unpaired^AF^glycan^content^and/or^relative^unpaired^HM^glycan^content.^^For^example,^the^ target^range^may^be^a^reference^level^of^a^specification,^or^of^a^reference^IgG^antibody^composition.^^ Also,^for^instance,^the^target^range^of^unpaired^AF^glycan^content^and/or^relative^unpaired^HM^ glycan^content^may^be^a^target^range^of^unpaired^AF^glycan^content^and/or^relative^unpaired^HM^ glycan^content^which^statistically^correlates^with^a^target^range^of^ADCC^activity^levels^of^a^reference^ IgG^antibody^composition.^ [0010]^^^^ Further^provided^are^methods^of^analyzing^an^IgG^antibody^composition.^^In^exemplary^ embodiments,^the^method^comprises^determining^the^relative^unpaired^glycan^content^of^a^sample^ of^the^IgG^antibody^composition^in^accordance^with^any^one^of^the^presently^disclosed^methods^of^ determining^the^relative^unpaired^glycan^content.^^In^various^aspects,^the^method^comprises^ comparing^the^relative^unpaired^AF^glycan^content^and/or^relative^unpaired^HM^glycan^content^of^ the^IgG^antibody^composition^to^the^relative^unpaired^AF^glycan^content^and/or^relative^unpaired^ HM^glycan^content^of^a^reference^product.^^The^method^may^be^performed^in^manufacturing,^for^ example^to^determine^if^a^lot^of^the^IgG^antibody^composition^meets^a^specification.^ [0011]^^^^ Additionally^provided^are^methods^of^monitoring^production^of^an^IgG^antibody^ composition.^^In^exemplary^embodiments,^the^method^comprises^determining^the^relative^unpaired^ glycan^content^of^an^IgG^antibody^composition^in^accordance^with^any^one^of^the^presently^disclosed^ methods^of^determining^the^relative^unpaired^glycan^content,^for^a^first^sample^obtained^at^a^first^ timepoint^and^for^a^second^sample^taken^at^a^second^timepoint^which^is^different^from^the^first^ timepoint.^^In^exemplary^aspects,^the^method^comprises^comparing^the^relative^unpaired^AF^glycan^ content^and/or^relative^unpaired^HM^glycan^content^of^the^first^sample^to^the^relative^unpaired^AF^ glycan^content^and/or^relative^unpaired^HM^glycan^content^of^the^second^sample.^ [0012]^^^^ Methods^of^producing^an^antibody^composition^are^also^provided^by^the^present^ disclosure.^^In^exemplary^embodiments,^the^method^comprises^determining^relative^unpaired^glycan^ content^of^the^antibody^composition^is^determined^in^accordance^with^any^one^of^the^presently^ disclosed^methods^of^determining^the^relative^unpaired^glycan^content,^wherein,^when^the^relative^ unpaired^glycan^content^is^determined^as^outside^a^predetermined^target^range,^the^method^further^ comprises^modifying^one^or^more^conditions^of^the^cell^culture^to^obtain^a^modified^cell^culture^and^ determining^the^relative^unpaired^glycan^content.^^The^method^in^various^instances^comprises^ repeating^steps^of^the^method^until^the^relative^unpaired^glycan^content^is^within^the^predetermined^ target^range.^ ^
[0013]^^^^ Additional^exemplary^embodiments^and^aspects^of^the^present^disclosure^are^provided^ below.^ BRIEF^DESCRIPTION^OF^THE^DRAWINGS^ [0014]^^^^ Figure^1A^is^an^illustration^of^exemplary^glycan^structures.^^ [0015]^^^^ Figure^1B^is^an^illustration^of^exemplary^glycan^groups.^ [0016]^^^^ Figure^2^is^a^diagram^of^the^salvage^pathway^and^the^de^novo^pathway^of^fucose^ metabolism.^^^ [0017]^^^^ Figure^3A^is^a^representative^glycan^map^chromatogram^(full^scale^view;^y^axis^max^~440.00^ EU))^obtained^through^HILIC.^^Figure^3B^is^a^representative^glycan^map^chromatogram^(expanded^ scale^view;^y^axis^max^~44.00^EU)^obtained^through^HILIC.^^^ [0018]^^^^ Figure^4A^is^a^series^of^drawings^exemplifying^antibodies^with^unpaired^or^paired^ afucosylated^glycans^and^unpaired^or^paired^high^mannose^glycans.^^Figure^4B^provides^exemplary^ flow^charts^useful^for^assigning^nomenclature^to^a^glycan^pair^comprising^canonical^glycans.^^As^ shown^in^the^exemplary^flow^chart^of^Box^1,^if^the^glycan^pair^comprises^zero^afucosylated^glycans^ (e.g.,^both^glycans^are^fucosylated),^then^the^glycan^pair^is^“paired^fucosylated”.^^If^both^glycans^of^ the^glycan^pair^are^afucosylated,^then^the^glycan^pair^is^“paired^afucosylated”.^^If^only^one^glycan^is^ afucosylated,^then^the^glycan^pair^is^“unpaired^afucosylated”.^^As^shown^in^the^exemplary^flow^chart^ of^Box^2,^if^the^glycan^pair^comprises^zero^high^mannose^glycans,^then^Box^1^is^used^for^determining^ glycan^pair^classification.^^If^both^glycans^comprise^a^high^mannose^and^are^afucosylated,^then^the^ glycan^pair^is^“paired^high^mannose”.^^If^only^one^glycan^of^the^glycan^pair^comprises^a^high^mannose^ and^is^afucosylated,^and^the^other^glycan^lacks^a^high^mannose^and^is^fucosylated,^then^the^glycan^ pair^is^“unpaired^high^mannose”.^^If^only^one^glycan^of^the^glycan^pair^comprises^a^high^mannose^and^ the^other^chain^is^afucosylated,^then^the^glycan^pair^is^“paired^afucosylated”.^^From^the^perspective^ of^high^mannose,^this^glycan^pair^is^“unpaired^high^mannose”.^^Additional^information^useful^for^ assigning^nomenclature^to^a^glycan^pair^is^provided^herein.^^See,^e.g.,^Example^1.^ [0019]^^^^ Figure^5^is^an^illustration^of^a^two^enzyme^digestion.^ [0020]^^^^ Figure^6A^shows^exemplary^chromatographic^peaks^obtained^from^a^95^min^HILIC^MS^ separation,^wherein^Fab^fragments^are^separated^from^glycosylated^Fc^fragments.^^The^inset^shows^a^ zoomed^view^of^the^glycosylated^Fc^fragment^peaks.^^Figure^6B^shows^a^further^zoomed^view^of^the^ glycosylated^Fc^fragment^peaks.^^The^glycopair^identity^for^each^peak^is^noted.^^Those^labeled^in^bold^ indicate^a^relevant^glycopair.^ ^
[0021]^^^^ Figure^7^shows^examples^of^an^unpaired^afucoslyated^Fc^fragment,^paired^afucosylated^Fc^ fragment,^unpaired^high^mannose^Fc^fragment,^and^paired^high^mannose^Fc^fragment,^following^a^ two^enzyme^digestion.^^^ [0022]^^^^ Figure^8A^is^an^illustration^of^the^simplified^chromatographic^separation^of^Fab^fragments^ from^Fc^fragments,^the^extraction^of^the^one^Fc^fragment^chromatographic^peak^for^mass^ spectroscopy,^and^mass^spectra^of^the^Fc^fragment^chromatographic^peak.^^Figure^8B^shows^ deconvoluted^mass^spectra^(left)^and^a^zoomed^view^thereof^(right).^^Deconvoluted^mass^spectra^ peaks^are^matched^to^a^database^for^peak^identification.^ [0023]^^^^ Each^of^Figures^9A^9C^is^a^deconvoluted^spectrum^of^samples^of^the^panel^described^in^ Example^2.^^Peaks^for^relevant^glycoform^species^are^shown.^ [0024]^^^^ Figure^10^is^a^schematic^of^a^cell^based^ADCC^assay.^^^ [0025]^^^^ Figure^11A^is^leverage^plot^of^relative^ADCC^activity^level^(%)^as^measured^by^the^cell^based^ ADCC^assay^plotted^as^a^function^of^measured^released^afucosylated^glycans^(%).^^The^best^fit^line^is^ the^solid^diagonal^line^in^the^middle^of^the^shaded^area.^^p<0.0001.^^^^^ [0026]^^^^ Figure^11B^is^leverage^plot^of^relative^ADCC^activity^level^(%)^as^measured^by^the^cell^based^ ADCC^assay^plotted^as^a^function^of^measured^released^high^mannose^glycans^(%).^^The^best^fit^line^is^ the^solid^diagonal^line^in^the^middle^of^the^shaded^area.^^p=0.2786.^^^^^ [0027]^^^^ Figure^11C^is^graph^of^the^Actual^ADCC^activity^level^(%)^as^measured^by^the^cell^based^ ADCC^assay^plotted^against^the^Predicted^ADCC^activity^level^(%)^as^calculated^using^Equation^8.^ [0028]^^^^ Figure^12A^is^leverage^plot^of^relative^ADCC^activity^level^(%)^as^measured^by^the^cell^based^ ADCC^assay^plotted^as^a^function^of^measured^unpaired^afucosylated^glycans^(%).^^The^best^fit^line^is^ the^solid^diagonal^line^in^the^middle^of^the^shaded^area.^^p<0.0001.^^^^^ [0029]^^^^ Figure^12B^is^leverage^plot^of^relative^ADCC^activity^level^(%)^as^measured^by^the^cell^based^ ADCC^assay^plotted^as^a^function^of^measured^unpaired^high^mannose^glycans^(%).^^The^best^fit^line^ is^the^solid^diagonal^line^in^the^middle^of^the^shaded^area.^^p=0.0012.^^^^^ [0030]^^^^ Figure^12C^is^graph^of^the^Actual^ADCC^activity^level^(%)^as^measured^by^the^cell^based^ ADCC^assay^plotted^against^the^Predicted^ADCC^activity^level^(%)^as^calculated^using^Equation^5.^^ DETAILED^DESCRIPTION^ [0031]^^^^ The^present^disclosure^provides^methods^of^determining^the^relative^unpaired^glycan^ content^of^an^IgG^antibody^composition.^^In^exemplary^embodiments,^the^method^comprises^ treating^the^IgG^antibody^composition^with^two^enzymes^to^form^a^mixture^of^Fab^fragments^and^Fc^ ^
fragments,^wherein^one^enzyme^cleaves^an^antibody^heavy^chain^at^a^site^N^terminal^to^the^hinge^ region^disulfide^linkages^and^the^other^enzyme^cleaves^the^ɴ1,4^linkage^between^core^GlcNAc^ residues^to^form^Fc^fragments^each^comprising^a^pair^of^core^glycan^structures,^(b)^separating^the^ Fab^fragments^from^the^Fc^fragments,^and^(c)^quantifying^the^abundance^of^(i)^paired^afucosylated^Fc^ fragments,^(ii)^unpaired^afucosylated^Fc^fragments,^(iii)^paired^high^mannose^Fc^fragments,^and/or^ (iv)^unpaired^high^mannose^Fc^fragments,^to^determine^the^relative^unpaired^afucosylated^(AF)^ glycan^content^and^relative^unpaired^high^mannose^(HM)^glycan^content.^^As^discussed^further^ below,^the^present^inventive^methods^may^be^used^for^preparing^an^IgG^composition^having^an^ ADCC^activity^within^a^target^range,^analyzing^an^IgG^antibody^composition,^monitoring^production^ of^an^IgG^antibody^composition,^and/or^producing^an^antibody^composition.^^^^^ [0032]^^^^ Glycosylation,^Glycans,^and^Methods^of^Glycan^Measurement^ [0033]^^^^ Many^secreted^proteins^undergo^post^translational^glycosylation,^a^process^by^which^sugar^ moieties^(e.g.,^glycans,^saccharides)^are^covalently^attached^to^specific^amino^acids^of^a^protein.^^In^ eukaryotic^cells,^two^types^of^glycosylation^reactions^occur:^^(1)^N^linked^glycosylation,^in^which^ glycans^are^attached^to^the^asparagine^of^the^recognition^sequence^Asn^X^Thr/Ser,^where^"X"^is^any^ amino^acid^except^proline,^and^(2)^O^linked^glycosylation^in^which^glycans^are^attached^to^serine^or^ threonine.^^Regardless^of^the^glycosylation^type^(N^linked^or^O^linked),^microheterogeneity^of^ protein^glycoforms^exists^due^to^the^large^range^of^glycan^structures^associated^with^each^site^(O^or^ N).^^^ [0034]^^^^ All^N^glycans^have^a^common^core^sugar^sequence:^^Manɲ1–6(Manɲ1–3)Manɴ1– 4GlcNAcɴ1–4GlcNAcɴ1^Asn^X^Ser/Thr^(Man
3GlcNAc
2Asn)^and^are^categorized^into^one^of^three^ types:^^(A)^a^high^mannose^(HM)^or^oligomannose^(OM)^type,^which^consists^of^two^N^ acetylglucosamine^(GalNAc)^moieties^and^at^least^5^(e.g.,^5,^6,^7,^8^or^9)^mannose^(Man)^residues,^(B)^ a^complex^type,^which^comprises^more^than^two^GlcNAc^moieties^and^any^number^of^other^sugar^ types,^or^(C)^a^hybrid^type,^which^comprises^a^Man^residue^on^one^side^of^the^branch^and^GlcNAc^at^ the^base^of^a^complex^branch.^^Figure^1A^(adapted^from^Stanley^et^al.,^Chapter^8:^N^Glycans,^ Essentials^of^Glycobiology,^2
nd^ed.,^Cold^Spring^Harbor^Laboratory^Press;^2009)^shows^the^three^types^ of^N^glycans.^^^ [0035]^^^^ N^linked^glycans^found^in^IgG^molecules^typically^comprise^one^or^more^monosaccharides^ of^galactose^(Gal),^N^^glucose^(Glc),^N^acetylglucoasamine^(GlcNAc),^glucoasamine^(GlcN),^mannose^ (Man),^fucose^(Fuc).^Exemplary^glycans,^their^identity^and^group^classifications^are^shown^in^Figure^ 1B.^ ^
[0036]^^^^ N^linked^glycosylation^begins^in^the^endoplasmic^reticulum^(ER),^where^a^complex^set^of^ reactions^result^in^the^attachment^of^a^core^glycan^structure^made^essentially^of^two^GlcNAc^ residues^and^three^Man^residues.^^The^glycan^complex^formed^in^the^ER^is^modified^by^action^of^ enzymes^in^the^Golgi^apparatus.^If^the^saccharide^is^relatively^inaccessible^to^the^enzymes,^it^typically^ stays^in^the^original^HM^form.^^If^enzymes^can^access^the^saccharide,^then^many^of^the^Man^residues^ are^cleaved^off^and^the^saccharide^is^further^modified,^resulting^in^the^complex^type^N^glycans^ structure.^For^example,^mannosidase^1^located^in^the^cis^Golgi,^can^cleave^or^hydrolyze^a^HM^glycan,^ while^fucosyltransferase^FUT^8,^located^in^the^medial^Golgi,^fucosylates^the^glycan^(Hanrue^Imai^^ Nishiya^(2007),^BMC^Biotechnology,^7:84).^^Fucose^metabolism^is^accomplished^through^the^salvage^ pathway^or^the^de^novo^pathway^as^shown^in^the^diagram^of^Figure^2.^^In^the^salvage^pathway,^free^ L^fucose^is^converted^to^GDP^fucose,^while^in^the^de^novo^pathway,^GDP^fucose^is^synthesized^via^ three^reactions^catalyzed^by^GMD^and^FX.^^GDP^fucose^is^then^transported^from^the^cytosol^to^the^ Golgi^lumen^by^GDP^Fuc^Transferase^and^transferred^to^acceptor^oligosaccharides^and^proteins.^^The^ other^reaction^product,^GDP,^is^converted^by^a^luminal^nucleotide^diphosphatase^to^guanosine^5^^ monophosphate^(GMP)^and^inorganic^phosphate^(Pi).^The^former^is^exported^to^the^cytosol^(via^an^ antiport^system^that^is^coupled^with^the^transport^of^GDP^fucose),^whereas^the^latter^is^postulated^ to^leave^the^Golgi^lumen^via^the^Golgi^anion^channel,^GOLAC.^^See,^e.g.,^Nordeen^et^al.^2000;^ Hirschberg^et^al.^2001.^^^ [0037]^^^^ Accordingly,^the^sugar^composition^and^the^structural^configuration^of^a^glycan^structure^ varies,^depending^on^the^glycosylation^machinery^in^the^ER^and^the^Golgi^apparatus,^the^accessibility^ of^the^machinery^enzymes^to^the^glycan^structure,^the^order^of^action^of^each^enzyme^and^the^stage^ at^which^the^protein^is^released^from^the^glycosylation^machinery,^among^other^factors.^^^ [0038]^^^^ Various^methods^may^be^used^for^assessing^glycans^present^in^a^glycoprotein^containing^ composition^or^for^determining,^detecting^or^measuring^a^glycoform^profile^(e.g.,^a^glycoprofile)^of^a^ particular^sample^comprising^glycoproteins.^^Suitable^methods^include,^but^are^not^limited^to,^ positive^ion^MALDI^TOF^analysis,^negative^ion^MALDI^TOF^analysis,^weak^anion^exchange^(WAX)^ chromatography,^normal^phase^chromatography^(NP^HPLC),^exoglycosidase^digestion,^Bio^Gel^P^4^ chromatography,^anion^exchange^chromatography^and^one^dimensional^n.m.r.^spectroscopy,^and^ combinations^thereof.^See,^e.g.,^Mattu^et^al.,^JBC^273:^2260^2272^(1998);^^Field^et^al.,^Biochem^J^ 299(Pt^1):^261^275^(1994);^Yoo^et^al.,^MAbs^2(3):^320^334^(2010)^Wuhrer^M.^et^al.,^Journal^of^ Chromatography^B,^2005,^Vol.825,^Issue^2,^pages^124^133;^Ruhaak^L.R.,^Anal^Bioanal^Chem,^2010,^ Vol.^397:3457^3481^and^Geoffrey,^R.^G.^et.^al.^Analytical^Biochemistry^1996,^Vol.^240,^pages^210^226.^^ Also,^a^suitable^method^of^assessing^glycans^present^in^a^glycoprotein^containing^composition^may^ comprise^enzymatic^cleavage^of^glycans^attached^to^the^glycoprotein.^^The^cleaved^or^released^ ^
glycans^are^subsequently^separated^by^Hydrophilic^Interaction^Liquid^Chromatography^(HILIC)^and^a^ chromatogram^with^several^peaks^is^produced.^^Each^peak^of^the^chromatogram^represents^a^ distribution^(amount^or^abundance)^of^a^different^glycan.^^Two^views^of^a^representative^HILIC^ chromatogram^comprising^peaks^for^different^glycans^are^provided^in^Figures^3A^and^3B.^^For^these^ purposes,^%^Peak^Area^=^Peak^Area/Total^Peak^Area^x^100%.^Accordingly,^the^level^of^a^particular^ glycan^(or^groups^of^glycans)^is^reported^as^a^%.^^For^example,^if^an^antibody^composition^is^ characterized^as^having^a^Man6^level^of^30%,^it^is^meant^that^30%^of^all^glycans^cleaved^from^the^ antibodies^of^the^composition^are^Man6.^^As^described^in^more^detail^herein,^it^is^noted^that^such^ methods^that^remove^glycans^from^the^glycoproteins^identify^a^distribution^of^glycan^content^for^a^ glycoprotein,^but^do^not^provide^information^relating^to^paired^glycans^and/or^unpaired^glycans.^ [0039]^^^^ The^present^disclosure^references^high^mannose^glycans^and^afucosylated^glycans^of^an^ antibody^composition^(see^Figure^1B^for^examples).^^As^used^herein,^the^term^“high^mannose^ glycans”^or^“HM^glycans”^encompasses^glycans^comprising^5,^6,^7,^8,^or^9^mannose^residues,^ abbreviated^as^Man5^or^M5,^Man6^or^M6,^Man7^or^M7,^Man8^or^M8,^and^Man9^or^M9,^respectively.^^ A^level^of^HM^glycans,^in^various^aspects,^is^obtained^by^summing^the^%^Man5,^the^%^Man6,^the^%^ Man7,^the^%^Man8,^and^the^%^Man9.^^As^used^herein,^the^term^"afucosylated^glycan"^or^“AF^glycan”^ refers^to^glycans^which^lack^a^core^fucose,^e.g.,^an^ɲ1,6^linked^fucose^on^the^GlcNAc^residue^involved^ in^the^amide^bond^with^the^Asn^of^the^N^glycosylation^site.^^Afucosylated^glycans^include,^but^are^not^ limited^to,^A1G0,^A1G1,^A2G0,^A2G1^(a^and^b),^A2G2,^A1G1M4^and^A1G1M5.^^It^is^noted^that^high^ mannose^glycans^also^lack^core^fucose^(and^thus^represent^a^subset^of^afucosylated^glycans),^but^ high^mannose^glycans^have^certain^characteristics^and^may^be^referred^to^as^a^separate^glycan^ group.^Accordingly,^unless^explicitly^stated^otherwise,^high^mannose^is^understood^to^represent^a^ separate^characteristic^and^may^be^classified^separately^from,^or^as^an^additional^characteristic^of^ afucosylated^glycans.^^See,^e.g.,^Reusch^and^Tejada,^Glycobiology^25(12):^1325^1334^(2015).^^A^level^ of^afucosylated^glycans,^in^various^aspects,^is^obtained^by^summing^the^%^A1G0,^the^%^A2G0,^the^%^ A2G1a,^the^%^A2G1b,^the^%^A2G2,^the^%^A1G1M5,^the^%^A1G1a.^^^ [0040]^^^^ The^level^(e.g.,^amount,^abundance)^of^glycans^(e.g.,^%^HM^glycans,^%^AF^glycans)^may^be^ determined^(e.g.,^measured)^by^any^of^the^various^methods^known^in^the^art^for^assessing^glycans^ present^in^a^glycoprotein^containing^composition^or^for^determining,^detecting^or^measuring^a^ glycoform^profile^(e.g.,^a^glycoprofile)^of^a^particular^sample^comprising^glycoproteins.^^The^level^ (e.g.,^amount,^abundance)^of^glycans^(e.g.,^%^HM^glycans,^%^AF^glycans)^of^an^antibody^composition^ may^be^determined^by^measuring^the^level^(e.g.,^amount,^abundance)^of^such^glycans^in^a^sample^of^ the^antibody^composition^though^a^chromatography^based^method,^e.g.,^HILIC,^and^the^level^(e.g.,^ amount,^abundance)^of^glycans^is^expressed^as^a^%,^as^described^herein.^^The^level^of^glycans^of^an^ ^
antibody^composition^may^be^expressed^as^a^%^of^all^glycans^cleaved^from^the^antibodies^of^the^ composition.^^The^level^(e.g.,^amount,^abundance)^of^glycans^(e.g.,^%^HM^glycans,^%^AF^glycans)^may^ be^determined^(e.g.,^measured)^by^measuring^the^level^of^such^glycans^in^a^sample^of^the^antibody^ composition.^^Samples^of^an^antibody^composition^may^be^taken^and^the^level^(e.g.,^amount,^ abundance)^of^glycans^(e.g.,^%^HM^glycans,^%^AF^glycans)^for^each^sample^may^be^determined^(e.g.,^ measured).^^The^%^HM^glycans^and/or^%^AF^glycans^may^be^determined.^^^ [0041]^^^^ Glycan^Pairing^and^Methods^of^Measuring^Unpaired^Glycans^of^an^Antibody^Composition^ [0042]^^^^ In^exemplary^instances^of^the^present^disclosure,^the^antibody^composition^comprises^ antibodies,^each^comprising^two^heavy^chains^and^two^light^chains.^^In^exemplary^aspects,^each^of^ the^antibodies^of^the^antibody^composition^comprises^a^fragment^crystallizable^(Fc)^region^and^two^ Fab^regions.^^The^Fc^region^of^the^antibody^comprises^two^identical^protein^fragments^derived^from^ constant^domains^of^the^antibody^heavy^chains,^and^each^protein^fragment^is^glycosylated,^e.g.,^ comprises^a^glycan.^^Accordingly,^in^exemplary^aspects,^the^Fc^region^of^the^antibody^comprises^a^ pair^of^glycans,^or^a^glycan^pair^(also^referred^to^herein^as^“glycopair”).^^In^various^aspects,^the^two^ glycans^of^the^glycan^pair^are^structurally^different^from^each^other.^^Alternatively,^the^glycans^of^the^ glycan^pair^are^structurally^identical.^^The^glycan^pair^may^be^classified^based^on^the^presence^or^ absence^of^glycans^of^the^glycan^pair^comprising^a^core^fucose,^as^well^as^the^presence^or^absence^of^ glycans^of^the^glycan^pair^comprising^high^mannose.^^Figure^4A^is^a^series^of^drawings^exemplifying^ an^unpaired^afucosylated^antibody,^a^paired^afucosylated^antibody,^an^unpaired^high^mannose^ antibody^and^a^paired^high^mannose^antibody.^^Figure^4B^provides^exemplary^flow^charts^useful^for^ assigning^nomenclature^to^a^glycan^pair^comprising^canonical^glycans.^^Exemplary^paired^high^ mannose^glycans^include^glycans^having^(a)^two^identical^high^mannose^glycans^(e.g.,^high^mannose^ of^identical^structure),^such^as^Man5,^Man6,^Man7,^Man8^or^Man9,^or^(b)^two^non^identical^high^ mannose^glycans^(e.g.,^high^mannose^glycans^having^different^structures)^but^each^high^mannose^ glycan^comprises^Man5,^Man6,^Man7,^Man8^or^Man9^(e.g.,^Man5^as^one^glycan^and^Man6,^Man7,^ Man8,^or^Man9^as^the^other^glycan,^or^Man6^as^one^glycan^and^Man7,^Man8,^or^Man9^as^the^other^ glycan,^or^Man7^as^one^glycan^and^Man8^or^Man9^as^the^other^glycan,^or^Man8^as^one^glycan^and^ Man9^as^the^other^glycan).^^^Exemplary^unpaired^high^mannose^glycans^include^glycans^having^Man5,^ Man6,^Man7,^Man8^or^Man9^as^one^glycan^and^a^fucosylated^or^afucosylated^glycan^as^the^other^ glycan.^^Exemplary^unpaired^afucosylated^glycan^pairs^include,^for^instance^A1G0,^A2G0,^A2G1a,^ A2G1b,^or^A2G2^as^one^glycan^and^a^fucosylated^glycan^as^the^other^glycan.^^Exemplary^paired^ afucosylated^glycans^include,^for^instance,^two^identical^afucosylated^glycans^(e.g.,^A1G0/A1G0)^or^ two^non^identical^afucosylated^glycans^(e.g.,^A1G0/A2G2).^^Paired^afucosylated^glycans^include,^for^ example,^(i)^A1G0^as^one^glycan^and^A2G0,^A2G1a,^A2G1b,^or^A2G2^or^(ii)^A2G0^on^one^chain^and^ ^
A2G1a,^A2G1b,^or^A2G2^on^the^other^Fc^chain^or^(iii)^A2G1a^on^one^Fc^chain^and^A2G1b^or^A2G2^on^ the^other^Fc^chain^or^(iv)^A2G1b^on^one^Fc^chain^and^A2G2^on^the^other^chain.^^Paired^afucosylated^ glycans^can^include^a^high^mannose^as^one^glycan^and^any^of^A1G0,^A2G0,^A2G1a,^A2G1b,^or^A2G2^ as^the^other,^e.g.,^M5/A1G0.^^From^the^high^mannose^perspective,^if^only^one^glycan^of^the^pair^ comprises^a^high^mannose,^the^paired^afucosylated^glycan^may^be^considered^as^an^“unpaired^high^ mannose”.^^^ [0043]^^^^ In^exemplary^instances,^the^glycan^pair^is^given^a^designation^based^on^the^ fucosylation/afucosylation^status^of^each^glycan^of^the^glycan^pair.^^In^exemplary^aspects,^the^glycan^ pair^is^given^a^designation^based^on^the^high^mannose^status^of^each^glycan^of^the^glycan^pair.^^ Suitable^designations^will^be^appreciated^in^the^context^of^the^glycan^structures^of^interest^in^the^ particular^scenario.^^In^various^aspects,^the^glycan^pair^is^given^two^designations,^wherein^a^first^ designation^is^based^on^the^fucosylation/afucosylation^status^of^each^glycan^of^the^glycan^pair^and^a^ second^designation^is^based^on^the^high^mannose^status^of^each^glycan^of^the^glycan^pair,^and,^ optionally,^a^summary^designation,^which^is^used^for^quantifying^the^abundance^of^Fc^fragments^ comprising^(i)^paired^afucosylated^glycans,^(ii)^unpaired^afucosylated^glycans,^(iii)^paired^high^ mannose^glycans,^and/or^(iv)^unpaired^high^mannose^glycans,^is^assigned.^^Table^A^below^provides^an^ exemplary^way^of^assigning^these^designations.^^In^various^instances,^a^Summary^Designation^is^ assigned^by^applying^three^rules^relating^to^the^First^Designation^(Designation^1)^and^Second^ Designation^(Designation^2),^as^follows:^^(1)^when^Designation^1^is^paired^fucosylated,^the^Summary^ Designation^is^paired^fucosylated^(regardless^of^Designation^2);^(2)^when^Designation^2^is^no^HM,^the^ Summary^Designation^is^the^same^as^Designation^1;^and^(3)^when^Designation^2^is^paired^HM^or^ unpaired^HM^and^Designation^1^is^not^paired^fucosylated,^the^Summary^Designation^is^the^same^as^ Designation^2^(e.g.,^Scenarios^4^and^10)^or^is^a^combination^of^Designation^1^and^Designation^2^(e.g.,^ Scenarios^5,^8,^and^9).^^^^^ ^ TABLE^A^

^
Fuc,^fucosylated;^AF,^afucosylated;^HM,^high^mannose.^^Designation^1^is^based^on^fucosylated/afucosylated^ status^of^each^glycan^on^each^Fc^chain.^^Designation^2^is^based^on^the^high^mannose^status^of^each^glycan^on^ each^Fc^chain.^^Summary^designation^is^based^on^3^rules:^^(1)^when^Designation^1^is^paired^fucosylated,^the^ Summary^Designation^is^paired^fucosylated^(regardless^of^Designation^2);^(2)^when^Designation^2^is^no^HM,^the^ Summary^Designation^is^the^same^as^Designation^1;^and^(3)^when^Designation^2^is^paired^HM^or^unpaired^HM^ and^Designation^1^is^not^paired^fucosylated,^the^Summary^Designation^is^the^same^as^Designation^2^(e.g.,^ Scenarios^4^and^10)^^or^is^a^combination^of^Designations^1^and^2^(e.g.,^Scenarios^5,^8^and^9).^^*Scenarios^5^and^ 9,^involving^a^fucosylated^glycan^comprising^a^high^mannose,^are^uncommon^occurrences.^ ^ [0044]^^^^ In^various^instances,^the^antibody,^or^its^Fc^region,^comprising^the^glycan^pair^may^be^ described^in^terms^of^its^glycan^pair^classification.^^Thus,^an^antibody^or^an^Fc^region^may^be^ described^as^“paired^afucosylated”,^“unpaired^afucosylated”,^“paired^fucosylated”,^“paired^high^ mannose”^or^“unpaired^high^mannose”^by^virtue^of^its^glycan^pair^classification.^^Similarly,^the^ antibody^or^its^Fc^region^may^be^described^as^comprising^paired^afucosylated^glycans^(or^paired^ afucosylated^glycan^pairs),^unpaired^afucosylated^glycans^(or^unpaired^afucosylated^glycan^pairs),^ paired^high^mannose^glycans^(paired^high^mannose^glycan^pairs)^and/or^unpaired^high^mannose^ glycans^(unpaired^high^mannose^glycan^pairs)^by^virtue^of^its^glycan^pair^classification.^^As^discussed^ below,^antibodies^may^be^cleaved^into^fragments.^^Fc^fragments^comprise^the^Fc^region^of^an^ antibody^and^may^be^described^as^“paired^afucosylated”,^“unpaired^afucosylated”,^“paired^ fucosylated”,^“paired^high^mannose”^or^“unpaired^high^mannose”^by^virtue^of^the^glycan^pair^ classification^of^the^Fc^region.^^^ [0045]^^^^ An^antibody^composition^may^be^characterized^in^terms^of^its^paired^glycan^content^and/or^ its^unpaired^glycan^content.^^For^example,^an^antibody^composition^may^be^characterized^in^terms^of^ ^
its^paired^afucosylated^glycan^content^and/or^unpaired^unfucosylated^glycan^content^and/or^its^ paired^high^mannose^content^and/or^unpaired^high^mannose^content.^^The^abundance^(e.g.,^paired^ afucosylated^glycan^content^and/or^unpaired^unfucosylated^glycan^content^and/or^its^paired^high^ mannose^content^and/or^unpaired^high^mannose^content)^as^described^herein^may^be^referred^to^as^ relative^or^absolute^abundance.^^In^exemplary^instances,^the^absolute^content^of^glycans^may^be^ expressed^in^units^measuring^levels^of^the^glycans^themselves,^for^example^in^terms^of^mass,^moles,^ mass^or^molar^units^per^volume^unit,^arbitrary^units,^area^under^the^curve,^or^intensities^of^mass^ spectral^peaks,^e.g.,^deconvoluted^mass^spectral^peaks^(e.g.,^as^may^be^determined^from^a^ chromatograph).^^In^exemplary^instances,^an^antibody^composition^is^characterized^in^terms^of^its^ relative^abundance^of^unpaired^glycans,^meaning^that^the^amount^of^unpaired^glycans^is^expressed^ as^an^amount^relative^to^the^sum^of^paired^glycans^and^unpaired^glycans^of^the^antibody^ composition.^^In^exemplary^aspects,^the^antibody^composition^is^characterized^in^terms^of^its^relative^ abundance^of^unpaired^afucosylated^glycans.^^In^exemplary^aspects,^the^antibody^composition^is^ characterized^in^terms^of^its^relative^abundance^of^unpaired^high^mannose^glycans.^^^ [0046]^^^^ ^^^The^term^“relative^abundance^of^unpaired^afucosylated^glycans”^which^is^synonymous^ with^“relative^unpaired^afucosylated^glycan^content”,^“relative^abundance^of^unpaired^afucosylated^ glycan^pairs”^and^“relative^%^unpaired^afucosylated^glycans”^is^calculated^as^dividing^the^percentage^ of^unpaired^afucosylated^glycans^by^the^sum^of^the^percentage^of^unpaired^afucosylated^glycans^and^ the^percentage^of^paired^afucosylated^glycans)^and^multiplying^by^100%.^^^ [0047]^^^^ The^term^“relative^abundance^of^unpaired^high^mannose^glycans”^which^is^synonymous^ with^“relative^unpaired^high^mannose^glycan^content”,^“relative^abundance^of^unpaired^high^ mannose^glycan^pairs”^and^“relative^%^unpaired^high^mannose^glycans”^is^calculated^as^the^ percentage^of^unpaired^high^mannose^glycans^divided^by^the^sum^of^the^percentage^of^unpaired^high^ mannose^glycans^and^the^percentage^of^paired^high^mannose^glycans)^multiplied^by^100%.^^^ [0048]^^^^ The^term^“relative^abundance^of^paired^afucosylated^glycans”^is^synonymous^with^“relative^ paired^afucosylated^glycan^content”^“relative^abundance^of^paired^afucosylated^glycan^pairs”^and^ “relative^%^paired^afucosylated^glycans”^is^calculated^as^dividing^the^percentage^of^paired^ afucosylated^glycans^by^the^sum^of^the^percentage^of^unpaired^afucosylated^glycans^and^the^ percentage^of^paired^afucosylated^glycans)^and^multiplying^by^100%.^^^ [0049]^^^^ The^term^“relative^abundance^of^paired^high^mannose^glycans”^is^synonymous^with^ “relative^paired^high^mannose^glycan^content”,^“relative^abundance^of^paired^high^mannose^glycan^ pairs”^and^“relative^%^paired^high^mannose^glycans”^is^calculated^as^dividing^the^percentage^of^ ^
paired^high^mannose^glycans^by^the^sum^of^the^percentage^of^unpaired^high^mannose^glycans^and^ the^percentage^of^paired^high^mannose^glycans)^and^multiplying^by^100%.^^ [0050]^^^^ In^various^aspects^of^the^present^disclosure,^the^sum^of^the^relative^%^unpaired^ afucosylated^glycans^and^the^relative^%^paired^afucosylated^glycans^equals^100%.^^Accordingly,^in^ various^aspects,^if^the^relative^%^paired^afucosylated^glycans^is^known,^the^relative^%^unpaired^ afucosylated^glycans^may^be^determined^(e.g.,^calculated)^by^subtracting^the^relative^%^paired^ afucosylated^glycans^from^100%.^^Also,^in^various^instances,^if^the^relative^%^unpaired^afucosylated^ glycans^is^known,^the^relative^%^paired^afucosylated^glycans^may^be^determined^(e.g.,^calculated)^by^ subtracting^the^relative^%^unpaired^afucosylated^glycans^from^100%.^^In^various^aspects^of^the^ present^disclosure,^the^sum^of^the^relative^%^unpaired^high^mannose^glycans^and^the^relative^%^ paired^high^mannose^glycans^equals^100%.^^Accordingly,^in^various^aspects,^if^the^relative^%^paired^ high^mannose^glycans^is^known,^the^relative^%^unpaired^high^mannose^glycans^may^be^determined^ (e.g.,^calculated)^by^subtracting^the^relative^%^paired^high^mannose^glycans^from^100%.^^Also,^in^ various^instances,^if^the^relative^%^unpaired^high^mannose^glycans^is^known,^the^relative^%^paired^ high^mannose^glycans^may^be^determined^(e.g.,^calculated)^by^subtracting^the^relative^%^unpaired^ high^mannose^glycans^from^100%.^^^^ [0051]^^^^ Methods^of^Determining^Relative^Unpaired^Glycan^Content^ [0052]^^^^ The^present^disclosure^provides^methods^of^determining^the^relative^unpaired^glycan^ content^of^an^IgG^antibody^composition.^^In^exemplary^embodiments,^the^method^comprises^(a)^ treating^the^IgG^antibody^composition^with^two^enzymes^to^form^a^mixture^of^Fab^fragments^and^Fc^ fragments,^wherein^one^enzyme^cleaves^an^antibody^heavy^chain^at^a^site^N^terminal^to^the^hinge^ region^disulfide^linkages^and^the^other^enzyme^cleaves^the^ɴ1,4^linkage^between^core^GlcNAc^ residues^to^form^Fc^fragments^each^comprising^a^pair^of^core^glycan^structures,^(b)^separating^the^ Fab^fragments^from^the^Fc^fragments,^and^(c)^quantifying^the^abundance^of^Fc^fragments^comprising^ (i)^paired^afucosylated^glycans,^(ii)^unpaired^afucosylated^glycans,^(iii)^paired^high^mannose^glycans,^ and/or^(iv)^unpaired^high^mannose^glycans,^to^determine^the^relative^unpaired^afucosylated^(AF)^ glycan^content^and^relative^unpaired^high^mannose^(HM)^glycan^content.^^In^(c),^the^abundance^of^(i)^ paired^afucosylated^Fc^fragments,^(ii)^unpaired^afucosylated^Fc^fragments,^(iii)^paired^high^mannose^ Fc^fragments,^and/or^(iv)^unpaired^high^mannose^Fc^fragments^is/are^quantified.^^In^exemplary^ aspects,^the^relative^abundance^of^(i)^paired^afucosylated^Fc^fragments,^(ii)^unpaired^afucosylated^Fc^ fragments,^(iii)^paired^high^mannose^Fc^fragments,^and/or^(iv)^unpaired^high^mannose^Fc^fragments^ is/are^quantified.^^In^exemplary^instances,^the^relative^abundance^of^unpaired^afucosylated^glycans^ and^the^relative^abundance^of^unpaired^high^mannose^glycans^is^determined.^^^In^exemplary^aspects^ the^one^enzyme^and^the^other^enzyme^are^incubated^simultaneously^or^concurrently^with^the^IgG^ ^
antibody^composition.^^For^example,^the^IgG^antibody^composition^may^be^incubated^with^the^one^ enzyme^(that^cleaves^an^antibody^heavy^chain^at^a^site^N^terminal),^and^the^other^enzyme^(that^ cleaves^the^ɴ1,4^linkage^between^core^GlcNAc^residues)^may^be^subsequently^added.^^^In^exemplary^ aspects,^the^one^enzyme^and^the^other^enzyme^are^incubated^sequentially.^^For^example,^the^ antibody^IgG^antibody^composition^may^be^incubated^with^the^one^enzyme^(that^cleaves^an^antibody^ heavy^chain^at^a^site^N^terminal),^and^after^that^incubation^is^over,^the^IgG^antibody^composition^ may^be^incubated^with^the^other^enzyme^(that^cleaves^the^ɴ1,4^linkage^between^core^GlcNAc^ residues).^ [0053]^^^^ In^various^aspects^of^the^presently^disclosed^method^of^determining^the^relative^unpaired^ glycan^content,^the^one^enzyme^which^cleaves^an^antibody^heavy^chain^at^a^site^N^terminal^to^the^ hinge^region^disulfide^linkages^is^a^cysteine^protease.^^In^various^instances,^the^enzyme^is^a^member^ of^the^IgdE^protease^family,^optionally,^an^IgdE^expressed^by^a^Streptococcus.^^In^various^instances,^ the^enzyme^is^structurally^identical^or^highly^similar^to^an^IgdE^protease^expressed^by^Streptococcus^ agalactiae.^^In^various^aspects,^the^enzyme^is^structurally^identical^or^highly^similar^to^an^enzyme^ expressed^by^a^Porphyromonas^anaerobe.^^In^various^instances,^the^enzyme^is^structurally^identical^ or^highly^similar^to^an^enzyme^expressed^by^a^Porphyromonas^gingivalis.^^In^various^instances,^the^ site^is^between^Thr^and^His^or^between^Lys^and^Thr^of^the^sequence^KTHTCPP^(SEQ^ID^NO:^1)^of^an^ IgG1^antibody^heavy^chain.^^In^exemplary^aspects,^the^method^comprises^treating^the^IgG^antibody^ composition^with^the^cysteine^protease^for^at^least^8^hours^or^at^least^12^hours,^e.g.,^at^least^9^hours,^ at^least^10^hours,^at^least^11^hours,^at^least^13^hours,^at^least^14^hours,^at^least^15^hours,^at^least^16^ hours,^at^least^17^hours,^at^least^18^hours,^at^least^19^hours,^at^least^20^hours,^at^least^21^hours,^at^ least^22^hours,^at^least^23^hours,^or^at^least^24^hours.^^^ [0054]^^^^ In^various^aspects,^the^other^enzyme^which^cleaves^the^ɴ1,4^linkage^between^core^GlcNAc^ residues^is^an^IgG^specific^enzyme.^^In^exemplary^aspects,^the^IgG^specific^enzyme^is^an^ endoglycosidase,^e.g.,^an^Endoglycosidase^D,^an^Endoglycosidase^F,^an^Endoglycosidase^F1,^an^ Endoglycosidase^F2,^an^Endoglycosidase^H,^or^an^Endo^S^or^EndoS2^endoglycosidase.^^In^exemplary^ instances,^the^endoglycosidase^is^an^Endoglycosidase^S.^^In^exemplary^aspects,^the^method^ comprises^treating^the^IgG^antibody^composition^with^the^IgG^specific^enzyme^for^less^than^1^hour,^ e.g.,^less^than^55^minutes,^less^than^50^minutes,^less^than^45^minutes,^less^than^40^minutes,^less^than^ 35^minutes,^less^than^30^minutes.^^In^various^aspects,^the^method^comprises^treating^the^IgG^ antibody^composition^with^the^IgG^specific^enzyme^for^about^30^minutes.^^In^various^aspects,^the^ other^enzyme^is^added^to^the^IgG^composition^during^its^the^incubation^with^the^enzyme^of^the^IgdE^ protease^family^(e.g.,^for^the^last^30^minutes^to^an^hour^of^the^incubation,^as^applicable).^^In^various^ ^
aspects,^the^other^enzyme^is^added^to^the^IgG^composition^after^the^incubation^with^the^enzyme^of^ the^IgdE^protease^family^has^been^completed.^^ [0055]^^^^ In^exemplary^aspects,^the^method^comprises^separating^the^Fab^fragments^from^the^Fc^ fragments^of^the^IgG^antibody^of^the^composition.^^Optionally,^the^method^comprises^separating^the^ Fab^fragments^from^the^Fc^fragments^by^a^chromatography.^^The^chromatography^may^be^any^ suitable^type^which^can^separate^the^Fab^fragments^from^the^Fc^fragments,^including,^but^not^limited^ to,^affinity^chromatography^(e.g.,^Protein^A^chromatography),^anion^or^cation^exchange^ chromatography,^liquid^chromatography^(LC,^e.g.,^reversed^phase^LC,^HILIC),^and^the^like.^^Optionally,^ the^chromatography^is^a^reversed^phase^liquid^chromatography.^^In^various^aspects,^spectral^peaks^ of^the^Fc^fragments^elute^as^one^chromatographic^peak^which^is^selected^for^analysis^of^the^mass^ spectrometry^data.^^In^exemplary^aspects,^the^method^comprises^performing^mass^spectroscopy^ data^analysis^of^the^Fc^fragments^to^obtain^one^or^more^mass^spectral^peaks.^^In^various^aspects,^the^ method^comprises^deconvoluting^the^mass^spectral^peaks^to^obtain^deconvoluted^mass^spectral^ peaks.^^Suitable^methods^of^deconvoluting^mass^spectral^peaks^are^known^in^the^art.^^See,^e.g.,^ Gadgil^et^al.,^J.^Am.^Soc.^Mass^Spectrom.^17,^867^(2006);^Murray^et^al.,^Pure^and^Applied^Chemistry,^ vol.^85,^no.^7,^2013,^pp.^1515^1609;^Marchetti^and^Mignerey,^Nuclear^Instruments^and^Methods^in^ Physics^Research^Section^A:^Accelerators,^Spectrometers,^Detectors^and^Associated^Equipment^ 324(1^2):^288^296^(1993);^Xu^et^al.,^Rapid^Commun^Mass^Spectrom.^2018^May^30;32(10):763^774.^^In^ exemplary^instances,^the^method^comprises^matching^the^molecular^weight^of^each^deconvoluted^ mass^spectral^peak^against^a^database^of^glycan^pairs^and^the^associated^molecular^weight^to^ identify^the^glycan^pair.^^In^exemplary^instances,^the^method^comprises^quantifying^the^abundance^ of^Fc^fragments^comprising^(i)^paired^afucosylated^glycans,^(ii)^unpaired^afucosylated^glycans,^(iii)^ paired^high^mannose^glycans,^and/or^(iv)^unpaired^high^mannose^glycans,^to^determine^the^relative^ unpaired^afucosylated^(AF)^glycan^content^and^relative^unpaired^high^mannose^(HM)^glycan^content.^^ In^various^aspects,^the^abundance^of^Fc^fragments^comprising^paired^afucosylated^glycans^and^the^ abundance^of^Fc^fragments^comprising^unpaired^afucosylated^glycans^are^summed^so^that^relative^ unpaired^afucosylated^glycans^may^be^determined.^^In^various^aspects,^the^abundance^of^Fc^ fragments^comprising^paired^high^mannose^glycans^and^the^abundance^of^Fc^fragments^comprising^ unpaired^high^mannose^glycans^are^summed^so^that^relative^unpaired^high^mannose^glycans^may^be^ determined.^^Suitable^methods^for^carrying^out^the^method^of^the^present^disclosure^is^described^in^ detail^in^Example^1.^^The^database^in^exemplary^aspects^comprises^the^glycan^pairs^and^their^ associated^theoretical^molecular^weights^of^Table^1^of^Example^1.^In^various^aspects,^the^separating^ and^quantifying^of^the^presently^disclosed^methods^occur^in^less^than^2^hours,^optionally,^less^than^ 95^minutes,^less^than^85^minutes,^less^than^75^minutes,^less^than^60^minutes,^less^than^45^minutes,^ ^
less^than^30^minutes^or^less^than^20^minutes^(optionally,^about^19^minutes,^about^18^minutes,^about^ 17^minutes,^about^16^minutes,^about^15^minutes,^about^14^minutes,^about^13^minutes,^about^12^ minutes,^about^11^minutes,^about^10^minutes,^about^9^minutes,^about^8^minutes,^about^7^minutes,^ about^6^minutes,^about^5^minutes).^^The^presently^disclosed^methods^in^exemplary^aspects^comprise^ separating^and^quantifying^in^less^than^30^minutes^or^less^than^20^minutes.^^In^various^aspects,^the^ separating^and^quantifying^of^the^presently^disclosed^methods^occur^in^about^5^minutes^to^about^25^ minutes,^about^5^minutes^to^about^20^minutes,^about^5^minutes^to^about^15^minutes,^about^5^ minutes^to^about^10^minutes,^about^10^minutes^to^about^25^minutes,^about^15^minutes^to^about^25^ minutes,^about^20^minutes^to^about^25^minutes).^^The^presently^disclosed^methods^of^determining^ the^relative^unpaired^glycan^content^advantageously^require^less^time^than^prior^methods.^^^ [0056]^^^^ Applications^of^Determining^Relative^Unpaired^Glycan^Content^ [0057]^^^^ The^present^inventive^methods^of^determining^relative^unpaired^glycans^are^useful^during^ the^development^and/or^production^of^an^antibody^composition.^^For^example,^the^method^of^ determining^relative^unpaired^glycans^may^be^used^for^analyzing^an^IgG^antibody^composition.^^ Accordingly,^methods^of^analyzing^an^IgG^antibody^composition^are^provided^herein.^^In^exemplary^ embodiments,^the^method^comprises^determining^the^relative^unpaired^glycan^content^of^a^sample^ of^the^IgG^antibody^composition^in^accordance^with^any^one^of^the^presently^disclosed^methods^of^ determining^the^relative^unpaired^glycan^content.^^In^various^aspects,^the^method^comprises^ comparing^the^relative^unpaired^AF^glycan^content^and/or^relative^unpaired^HM^glycan^content^of^ the^IgG^antibody^composition^to^the^relative^unpaired^AF^glycan^content^and/or^relative^unpaired^ HM^glycan^content^of^a^reference^product.^^The^method^may^be^performed^in^manufacturing,^for^ example^to^determine^if^a^lot^of^the^IgG^antibody^composition^meets^a^specification.^ [0058]^^^^ Additionally^provided^are^methods^of^monitoring^production^of^an^IgG^antibody^ composition.^^In^exemplary^embodiments,^the^method^comprises^determining^the^relative^unpaired^ glycan^content^of^an^IgG^antibody^composition^in^accordance^with^any^one^of^the^presently^disclosed^ methods^of^determining^the^relative^unpaired^glycan^content,^for^a^first^sample^obtained^at^a^first^ timepoint^and^for^a^second^sample^taken^at^a^second^timepoint^which^is^different^from^the^first^ timepoint.^^In^exemplary^aspects,^the^method^comprises^comparing^the^relative^unpaired^AF^glycan^ content^and/or^relative^unpaired^HM^glycan^content^of^the^first^sample^to^the^relative^unpaired^AF^ glycan^content^and/or^relative^unpaired^HM^glycan^content^of^the^second^sample.^ [0059]^^^^ ^^In^various^aspects,^each^of^the^first^sample^and^second^sample^is^a^sample^of^in^process^ material.^^In^various^instances,^the^first^sample^is^a^sample^of^in^process^material^and^the^second^ sample^is^a^sample^of^a^manufacturing^lot.^^Optionally,^the^first^sample^is^a^sample^obtained^before^ ^
one^or^more^conditions^of^the^cell^culture^are^modified^and^the^second^sample^is^a^sample^obtained^ after^the^one^or^more^conditions^of^the^cell^culture^are^modified.^^^ [0060]^^^^ In^various^aspects,^the^presently^disclosed^methods^are^useful^for^determining^whether^an^ IgG^antibody^composition^meets^the^statistical^criteria^which^includes^appropriate^acceptance^levels^ and/or^appropriate^rejection^levels.^^^ [0061]^^^^ In^various^aspects,^the^methods^of^analyzing^an^IgG^antibody^composition^and/or^methods^ of^monitoring^production^of^an^IgG^antibody^composition^occur^during^the^development^or^ production^of^an^antibody^composition.^^Accordingly,^methods^of^producing^an^antibody^ composition^are^also^provided^by^the^present^disclosure.^^In^exemplary^embodiments,^the^method^ comprises^(A)^determining^the^relative^unpaired^glycan^content^of^a^sample^of^an^IgG^antibody^ composition,^wherein^relative^unpaired^glycan^content^of^the^sample^of^the^antibody^composition^is^ determined^in^accordance^with^any^one^of^the^presently^disclosed^methods^of^determining^the^ relative^unpaired^glycan^content,^wherein^the^sample^is^a^sample^of^in^process^material,^wherein,^ when^the^relative^unpaired^glycan^content^is^determined^as^outside^a^predetermined^target^range,^ the^method^further^comprises^(B)^modifying^one^or^more^conditions^of^the^cell^culture^to^obtain^a^ modified^cell^culture^and^determining^the^relative^unpaired^glycan^content,^and^optionally,^repeating^ (A)^and^(B)^until^the^relative^unpaired^glycan^content^is^within^the^predetermined^target^range.^^In^ various^instances,^one^or^more^conditions^of^the^cell^culture^are^modified^to^primarily^change^the^ relative^unpaired^HM^glycan^content^to^achieve^the^target^range^of^relative^unpaired^glycan^content.^^ In^various^aspects,^one^or^more^conditions^of^the^cell^culture^are^modified^to^primarily^change^the^ relative^unpaired^AF^glycan^content^to^achieve^the^target^range^of^relative^unpaired^glycan^content.^^ Additional^embodiments^of^methods^of^producing^antibody^compositions^are^further^provided^ below.^^See,^e.g.,^Methods^of^Producing^Antibody^Compositions.^ [0062]^^^^ ADCC^and^Methods^of^Modifying^ADCC^Activity^Levels^ [0063]^^^^ The^data^presented^herein^support^that^the^relative^unpaired^glycan^content^of^an^antibody^ composition^is^related^to^the^ADCC^activity^level^for^the^antibody^composition^and^that^the^ADCC^ activity^level^of^the^antibody^composition^may^be^modified^by^modifying^the^relative^unpaired^glycan^ content^of^the^antibody^composition.^^Without^being^bound^to^a^particular^theory,^the^relative^ unpaired^afucosylated^glycan^content^and/or^the^relative^unpaired^high^mannose^glycan^content^of^ an^antibody^composition^is^related^to^the^ADCC^activity^level^of^the^antibody^composition,^and,^ changing^the^relative^unpaired^afucosylated^glycan^content^and/or^the^relative^unpaired^high^ mannose^glycan^content^of^the^antibody^composition^leads^to^changing^the^ADCC^activity^level^of^the^ antibody^composition.^^It^is^further^contemplated^that^relative^unpaired^glycan^content^(e.g.,^relative^ ^
unpaired^afucosylated^glycan^content^and/or^relative^unpaired^high^mannose^glycan^content)^has^ greater^leverage^on^ADCC^than^the^relative^paired^glycan^content^(e.g.,^relative^paired^afucosylated^ glycan^content^and/or^relative^paired^high^mannose^glycan^content),^so^that^a^percent^change^in^the^ relative^unpaired^glycan^content^will^have^a^greater^effect^on^ADCC^than^the^same^percent^change^in^ relative^paired^glycan^content.^^^Accordingly,^provided^herein^are^methods^of^modifying^the^ADCC^ level^of^an^antibody^composition.^^In^exemplary^embodiments,^the^method^comprises^modifying^the^ relative^unpaired^afucosylated^glycan^content^of^an^antibody^composition^and/or^the^relative^ unpaired^high^mannose^glycan^content^of^an^antibody^composition.^^^ [0064]^^^^ The^term^“ADCC”^or^“antibody^dependent^cell^mediated^cytotoxicity”^or^“antibody^ dependent^cellular^cytotoxicity”^refers^to^the^mechanism^by^which^an^effector^cell^of^the^immune^ system^(e.g.,^natural^killer^cells^(NK^cells),^macrophages,^neutrophils,^eosinophils)^actively^lyses^a^ target^cell,^whose^membrane^surface^antigens^have^been^bound^by^specific^antibodies.^^^ADCC^is^a^ part^of^the^adaptive^immune^response^and^occurs^when^antigen^specific^antibodies^bind^to^(1)^the^ membrane^surface^antigens^on^a^target^cell^through^its^antigen^binding^regions^and^(2)^to^Fc^ receptors^on^the^surface^of^the^effector^cells^through^its^Fc^region.^^Binding^of^the^Fc^region^of^the^ antibody^to^the^Fc^receptor^causes^the^effector^cells^to^release^cytotoxic^factors^that^lead^to^death^of^ the^target^cell^(e.g.,^through^cell^lysis^or^cellular^degranulation).^^^ [0065]^^^^ Fc^receptors^are^receptors^on^the^surfaces^of^B^lymphocytes,^follicular^dendritic^cells,^NK^ cells,^macrophages,^neutrophils,^eosinophils,^basophils,^platelets^and^mast^cells^that^bind^to^the^Fc^ region^of^an^antibody.^^Fc^receptors^are^grouped^into^different^classes^based^on^the^type^of^antibody^ that^they^bind.^^For^example,^an^Fcɶ^receptor^is^a^receptor^for^the^Fc^region^of^an^IgG^antibody,^an^ Fc^alpha^receptor^is^a^receptor^for^the^Fc^region^of^an^IgA^antibody,^and^an^Fc^epsilon^receptor^is^a^ receptor^for^the^Fc^region^of^an^IgE^antibody.^^^ [0066]^^^^ The^term^“FcɶR”^or^“Fc^gamma^receptor”^is^a^protein^belonging^to^the^IgG^superfamily^ involved^in^inducing^phagocytosis^of^opsonized^cells^or^microbes.^See,^e.g.,^Fridman^WH.^^Fc^ receptors^and^immunoglobulin^binding^factors.^FASEB^Journal.^5^(12):^2684–90^(1991).^Members^of^ the^Fc^gamma^receptor^family^include:^FcɶRI^(CD64),^FcɶRIIA^(CD32),^FcɶRIIB^(CD32),^FcɶRIIIA^ (CD16a),^and^FcɶRIIIB^(CD16b).^^The^sequences^of^FcɶRI,^FcɶRIIA,^FcɶRIIB,^FcɶRIIIA,^and^FcɶRIIIB^can^ be^found^in^many^sequence^databases,^for^example,^at^the^Uniprot^database^(www.uniprot.org)^ under^accession^numbers^P12314^(FCGR1_HUMAN),^P12318^(FCG2A_HUMAN),^P31994^ (FCG2B_HUMAN),^P08637^(FCG3A_HUMAN),^and^P08637^(FCG3A_HUMAN),^respectively.^^ [0067]^^^^ The^term^“ADCC^activity”^or^“ADCC^level”^refers^to^the^extent^to^which^ADCC^is^activated^or^ stimulated.^^Methods^of^measuring^or^determining^the^ADCC^level^of^an^antibody^composition,^ ^
including^commercially^available^assays^and^kits^for^measuring^or^determining^the^ADCC^level,^are^ well^known^in^the^art,^as^described,^Yamashita^et^al.,^Scientific^Reports^6:^article^number^19772^ (2016),^doi:10.1038/srep19772);^Kantakamalakul^et^al.,^“A^novel^EGFP^CEM^NKr^flow^cytometric^ method^for^measuring^antibody^dependent^cell^mediated^cytotoxicity^(ADCC)^activity^in^HIV^1^ infected^individuals”,^J^Immunol^Methods^315^(Issues^1^2):^1^10;^(2006);^Gomez^Roman^et^al.,^“A^ simplified^method^for^the^rapid^fluorometric^assessment^of^antibody^dependent^cell^mediated^ cytotoxicity”,^J^Immunol^Methods^308^(Issues^1^2):^53^67^(2006);^Schnueriger^et^al.,^Development^of^ a^quantitative,^cell^line^based^assay^to^measure^ADCC^activity^mediated^by^therapeutic^antibodies”,^ Molec^Immunology^38^(Issues^12^13):^1512^1517^(2011);^and^Mata^et^al.,^“Effects^of^ cryopreservation^on^effector^cells^for^antibody^dependent^cell^mediated^cytotoxicity^(ADCC)^and^ natural^killer^(NK)^cell^activity^in^
51Cr^release^and^CD107a^assays”,^J^Immunol^Methods^406:^1^9^ (2014);^all^herein^incorporated^by^reference^for^all^purposes.^^The^term^“ADCC^Assay”^or^“FcyR^ reporter^gene^assay”^refers^to^an^assay,^kit^or^method^useful^to^determine^the^ADCC^activity^of^an^ antibody.^^Exemplary^methods^of^measuring^or^determining^the^ADCC^activity^of^an^antibody^in^the^ methods^described^herein^include^the^ADCC^assay^described^in^the^Example^3^or^the^ADCC^Reporter^ Assay^commercially^available^from^Promega^(Catalog^No.^G7010^and^G7018).^^In^some^embodiments,^ ADCC^activity^is^measured^or^determined^using^a^calcein^release^assay^containing^one^or^more^of^the^ following:^a^FcɶRIIIa^(158V)^expressing^NK92(M1)^cells^as^effector^cells^and^HCC2218^cells^or^MT^3^ cells^as^target^cells^labeled^with^calcein^AM.^^An^illustration^of^an^exemplary^calcein^release^assay^is^ provided^as^Figure^10.^^In^exemplary^aspects^of^the^calcein^release^assay,^a^standard^curve^is^created^ using^various^concentrations^of^a^reference^antibody^(Figure^10B).^^^ [0068]^^^^ In^exemplary^aspects,^the^level^of^ADCC^of^an^antibody^composition^is^determined^by^a^ quantitative^cellbased^assay^which^measures^the^ability^of^the^antibodies^of^the^antibody^ composition^to^mediate^cell^cytotoxicity^in^a^dosedependent^manner^in^cells^expressing^the^antigen^ of^the^antibodies^and^engaging^FcɶRIIIA^receptors^on^effector^cells^through^the^Fc^domain^of^the^ antibodies.^^In^various^embodiments,^the^method^comprises^the^use^of^target^cells^harboring^ detectable^labels^that^are^released^when^the^target^cells^are^lysed^by^the^effector^cells.^^The^amount^ of^detectable^label^released^from^the^target^cells^is^a^measure^of^the^ADCC^activity^of^the^antibody^ composition.^^The^amount^of^detectable^label^released^from^the^target^cells,^in^some^aspects,^is^ compared^to^a^baseline.^^Also,^the^ADCC^level^may^be^reported^as^a^%^ADCC^relative^to^a^control^%^ ADCC.^^In^various^aspects,^the^%^ADCC^is^a^relative^%^ADCC,^which^optionally,^is^relative^to^a^control^ %^ADCC.^^In^various^aspects,^the^control^%^ADCC^is^the^%^ADCC^of^a^reference^antibody.^^In^ exemplary^instances,^the^control^%^ADCC^is^within^a^range^of^about^60%^to^about^130%.^^Optionally,^ the^%^ADCC^is^determined^by^the^assay^described^in^Example^3.^^^ ^
[0069]^^^^ In^exemplary^embodiments,^the^method^of^modifying^(increasing^or^decreasing)^the^ADCC^ level^of^an^antibody^composition^comprises^modifying^(increasing^or^decreasing)^the^relative^ unpaired^afucosylated^glycan^content^of^an^antibody^composition^and/or^the^relative^unpaired^high^ mannose^glycan^content^of^an^antibody^composition.^^In^exemplary^aspects,^the^presently^disclosed^ method^of^modifying^the^ADCC^level^of^an^antibody^composition^comprises^increasing^the^relative^ unpaired^afucosylated^glycan^content^to^increase^the^level^of^ADCC^activity.^^In^exemplary^instances,^ the^method^of^modifying^the^ADCC^level^of^an^antibody^composition^comprises^increasing^the^ relative^unpaired^high^mannose^glycan^content^to^increase^the^level^of^ADCC^activity.^^In^various^ aspects,^the^increase^in^ADCC^activity^level^provided^by^the^methods^of^the^disclosure^is^at^least^or^ about^a^1%^to^about^a^20%^increase^(e.g.,^at^least^or^about^a^1%^increase,^at^least^or^about^a^2%^ increase,^at^least^or^about^a^3%^increase,^at^least^or^about^a^4%^increase,^at^least^or^about^a^5%^ increase,^at^least^or^about^a^6%^increase,^at^least^or^about^a^7%^increase,^at^least^or^about^a^8%^ increase,^at^least^or^about^a^9%^increase,^at^least^or^about^a^10%^increase,^^at^least^or^about^a^11%^ increase,^at^least^or^about^a^12%^increase,^at^least^or^about^a^13%^increase,^at^least^or^about^a^14%^ increase,^at^least^or^about^a^15%^increase,^at^least^or^about^a^16%^increase,^at^least^or^about^a^17%^ increase,^at^least^or^about^a^18%^increase,^at^least^or^about^a^19%^increase,^at^least^or^about^a^20%^ increase)^relative^to^a^control.^^A^suitable^control^may^be^the^same^protein^or^antibody^composition^ without^the^increase^in^the^relative^unpaired^glycan^content.^^In^exemplary^embodiments,^the^ increase^in^ADCC^activity^level^provided^by^the^methods^of^the^disclosure^is^about^10%^to^about^ 100%,^optionally,^about^10%^to^about^90%,^about^10%^to^about^80%,^about^10%^to^about^70%,^ about^10%^to^about^70%,^about^10%^to^about^50%,^about^10%^to^about^40%,^about^10%^to^about^ 30%,^about^10%^to^about^20%,^about^10%^to^about^15%,^about^20%^to^about^100%,^about^30%^to^ about^100%,^about^40%^to^about^100%,^about^50%^to^about^100%,^about^60%^to^about^100%,^about^ 70%^to^about^100%,^about^80%^to^about^100%,^about^90%^to^about^100%,^or^about^95%^to^about^ 100%.^^The^increase^can^be^relative^to^the^control.^^In^exemplary^embodiments,^the^increase^in^ADCC^ activity^level^provided^by^the^methods^of^the^disclosure^is^over^100%,^e.g.,^200%,^300%,^400%,^500%,^ 600%,^700%,^800%,^900%^or^even^1000%^relative^a^control.^^In^exemplary^embodiments,^the^level^of^ ADCC^activity^increases^by^at^least^about^1.5^fold,^relative^a^control.^^A^suitable^control^may^be^an^ ADCC^activity^level^of^the^same^protein^or^antibody^composition^without^the^change^in^the^relative^ unpaired^glycan^content.^^In^exemplary^embodiments,^the^level^of^ADCC^activity^increases^by^at^least^ about^2^fold,^relative^a^control.^^In^exemplary^embodiments,^the^level^of^ADCC^activity^increases^by^ at^least^about^3^fold,^relative^a^control.^^In^exemplary^embodiments,^the^level^of^ADCC^activity^ increases^by^at^least^about^4^fold^or^about^5^fold,^relative^to^a^control.^^In^various^aspects,^the^ increase^in^the^level^of^ADCC^activity^of^the^antibody^composition^is^related^to^the^increase^in^ ^
relative^unpaired^glycan^content.^^For^instance,^the^increase^in^the^level^of^ADCC^activity^of^the^ antibody^composition^is^at^least^or^about^X%^per^~1%^increase^in^relative^unpaired^glycan^content,^ wherein^X%^is^at^least^or^about^a^1%^to^about^a^20%^increase^(e.g.,^at^least^or^about^a^1%^increase,^at^ least^or^about^a^2%^increase,^at^least^or^about^a^3%^increase,^at^least^or^about^a^4%^increase,^at^least^ or^about^a^5%^increase,^at^least^or^about^a^6%^increase,^at^least^or^about^a^7%^increase,^at^least^or^ about^a^8%^increase,^at^least^or^about^a^9%^increase,^at^least^or^about^a^10%^increase,^^at^least^or^ about^a^11%^increase,^at^least^or^about^a^12%^increase,^at^least^or^about^a^13%^increase,^at^least^or^ about^a^14%^increase,^at^least^or^about^a^15%^increase,^at^least^or^about^a^16%^increase,^at^least^or^ about^a^17%^increase,^at^least^or^about^a^18%^increase,^at^least^or^about^a^19%^increase,^at^least^or^ about^a^20%^increase).^^Also,^for^example,^X%^may^be^about^10%^to^about^100%,^optionally,^about^ 10%^to^about^90%,^about^10%^to^about^80%,^about^10%^to^about^70%,^about^10%^to^about^60%,^ about^10%^to^about^50%,^about^10%^to^about^40%,^about^10%^to^about^30%,^about^10%^to^about^ 20%,^about^10%^to^about^15%,^about^20%^to^about^100%,^about^30%^to^about^100%,^about^40%^to^ about^100%,^about^50%^to^about^100%,^about^60%^to^about^100%,^about^70%^to^about^100%,^about^ 80%^to^about^100%,^about^90%^to^about^100%,^or^about^95%^to^about^100%.^^^ [0070]^^^^ In^various^aspects,^the^method^of^modifying^the^ADCC^level^of^an^antibody^composition^ comprises^decreasing^the^relative^unpaired^afucosylated^glycan^content^to^decrease^the^level^of^ ADCC^activity.^^In^various^instances,^the^method^of^modifying^the^ADCC^level^of^an^antibody^ composition^comprises^decreasing^the^relative^unpaired^high^mannose^glycan^content^to^decrease^ the^level^of^ADCC^activity.^^In^various^aspects,^the^decrease^in^the^ADCC^activity^level^provided^by^the^ methods^of^the^disclosure^is^at^least^or^about^a^1%^to^about^a^20%^decrease^(e.g.,^at^least^or^about^a^ 1%^decrease,^at^least^or^about^a^2%^decrease,^at^least^or^about^a^3%^decrease,^at^least^or^about^a^4%^ decrease,^at^least^or^about^a^5%^decrease,^at^least^or^about^a^6%^decrease,^at^least^or^about^a^7%^ decrease,^at^least^or^about^a^8%^increase,^at^least^or^about^a^9%^increase,^at^least^or^about^a^10%^ increase,^^at^least^or^about^a^11%^increase,^at^least^or^about^a^12%^increase,^at^least^or^about^a^13%^ increase,^at^least^or^about^a^14%^increase,^at^least^or^about^a^15%^increase,^at^least^or^about^a^16%^ increase,^at^least^or^about^a^17%^increase,^at^least^or^about^a^18%^increase,^at^least^or^about^a^19%^ increase,^at^least^or^about^a^20%^increase)^relative^a^control.^^A^suitable^control^may^be^the^same^ protein^or^antibody^composition^without^the^change^in^the^relative^unpaired^glycan^and^overall^ glycan^composition^content.^^In^exemplary^embodiments,^the^decrease^in^the^ADCC^activity^level^ provided^by^the^methods^of^the^disclosure^is^about^10%^to^about^100%,^optionally,^about^10%^to^ about^90%,^about^10%^to^about^80%,^about^10%^to^about^70%,^about^10%^to^about^60%,^about^10%^ to^about^50%,^about^10%^to^about^40%,^about^10%^to^about^30%,^about^10%^to^about^20%,^about^ 10%^to^about^15%,^about^20%^to^about^100%,^about^30%^to^about^100%,^about^40%^to^about^100%,^ ^
about^50%^to^about^100%,^about^60%^to^about^100%,^about^70%^to^about^100%,^about^80%^to^about^ 100%,^about^90%^to^about^100%,^or^about^95%^to^about^100%.^^The^decrease^can^be^relative^to^a^ control.^^In^exemplary^embodiments,^the^decrease^in^the^ADCC^activity^level^provided^by^the^ methods^of^the^disclosure^is^over^100%,^e.g.,^200%,^300%,^400%,^500%,^600%,^700%,^800%,^900%^or^ even^1000%^relative^a^control.^^In^exemplary^embodiments,^the^level^of^ADCC^activity^decreases^by^ at^least^about^1.5^fold,^relative^a^control.^^A^suitable^control^may^be^the^ADCC^activity^level^of^the^ same^protein^or^antibody^composition^without^the^change^in^the^glycan^content.^^^In^exemplary^ embodiments,^the^level^of^ADCC^activity^decreases^by^at^least^about^2^fold,^relative^a^control.^^In^ exemplary^embodiments,^the^level^of^ADCC^activity^decreases^by^at^least^about^3^fold,^relative^a^ control.^^In^exemplary^embodiments,^the^level^of^ADCC^activity^decreases^by^at^least^about^4^fold^or^ about^5^fold,^relative^to^a^control.^^In^various^aspects,^the^decrease^in^the^level^of^ADCC^activity^of^ the^antibody^composition^is^related^to^the^decrease^in^relative^unpaired^glycan^content.^^For^ instance,^the^decrease^in^the^level^of^ADCC^activity^of^the^antibody^composition^is^at^least^or^about^ X%^per^~1%^decrease^in^relative^unpaired^glycan^content,^wherein^X%^is^at^least^or^about^a^1%^to^ about^a^20%^decrease^(e.g.,^at^least^or^about^a^1%^decrease,^at^least^or^about^a^2%^decrease,^at^least^ or^about^a^3%^decrease,^at^least^or^about^a^4%^decrease,^at^least^or^about^a^5%^decrease,^at^least^or^ about^a^6%^decrease,^at^least^or^about^a^7%^decrease,^at^least^or^about^a^8%^increase,^at^least^or^ about^a^9%^increase,^at^least^or^about^a^10%^increase,^^at^least^or^about^a^11%^increase,^at^least^or^ about^a^12%^increase,^at^least^or^about^a^13%^increase,^at^least^or^about^a^14%^increase,^at^least^or^ about^a^15%^increase,^at^least^or^about^a^16%^increase,^at^least^or^about^a^17%^increase,^at^least^or^ about^a^18%^increase,^at^least^or^about^a^19%^increase,^at^least^or^about^a^20%^increase).^^Also,^for^ example,^X%^may^be^about^10%^to^about^100%,^optionally,^about^10%^to^about^90%,^about^10%^to^ about^80%,^about^10%^to^about^70%,^about^10%^to^about^60%,^about^10%^to^about^50%,^about^10%^ to^about^40%,^about^10%^to^about^30%,^about^10%^to^about^20%,^about^10%^to^about^15%,^about^ 20%^to^about^100%,^about^30%^to^about^100%,^about^40%^to^about^100%,^about^50%^to^about^ 100%,^about^60%^to^about^100%,^about^70%^to^about^100%,^about^80%^to^about^100%,^about^90%^ to^about^100%,^or^about^95%^to^about^100%.^^^^ [0071]^^^^ In^exemplary^aspects,^the^modification^(increase^or^decrease)^effected^by^the^presently^ disclosed^methods^are^relative^to^a^“control”.^^In^exemplary^aspects,^the^control^is^the^level^of^ADCC^ activity^when^the^steps^of^the^method^are^not^carried^out.^^In^exemplary^aspects,^the^control^is^the^ level^of^ADCC^activity^when^the^relative^unpaired^afucosylated^glycan^content^and/or^the^relative^ unpaired^high^mannose^glycan^content^is^not^modified^(increased^or^decreased).^^For^example,^a^ suitable^control^may^be^the^ADCC^activity^level^of^the^same^protein^or^antibody^composition^but^ without^the^increase^in^the^relative^unpaired^glycan^content^(e.g.,^relative^unpaired^afucosylated^ ^
glycan^content^and/or^the^relative^unpaired^high^mannose^glycan^content),^or^a^suitable^control^may^ be^the^ADCC^activity^level^of^the^same^protein^or^antibody^composition^but^without^the^decrease^in^ the^relative^unpaired^glycan^content^(e.g.,^relative^unpaired^afucosylated^glycan^content^and/or^the^ relative^unpaired^high^mannose^glycan^content).^^In^exemplary^instances,^the^control^may^be^the^ ADCC^activity^level^of^the^same^protein^or^antibody^composition^produced^under^the^same^cell^ culture^conditions^with^exception^of^those^conditions^that^were^modified^to^cause^a^change^in^the^ relative^unpaired^glycan^content.^^In^exemplary^aspects,^the^control^may^be^the^ADCC^activity^level^of^ the^same^protein^or^antibody^composition^produced^under^a^first^set^of^cell^culture^conditions^which^ lead^to^an^ADCC^activity^level^which^is^outside^of^a^target^range^of^ADCC^activity^level.^^In^various^ aspects,^the^control^may^be^the^ADCC^activity^level^of^the^same^protein^or^antibody^composition^ produced^under^a^first^set^of^cell^culture^conditions^which^lead^to^a^relative^unpaired^afucosylated^ glycan^content^and/or^the^relative^unpaired^high^mannose^glycan^content^which^is/are^outside^of^a^ target^range.^ [0072]^^^^ The^term^“target^range”^refers^to^a^range^of^values^based^on^a^reference^standard^or^ reference^product.^^For^instance,^the^target^range^of^ADCC^activity^level^may^be^the^range^of^ADCC^ activity^level^exhibited^by^a^reference^product.^^Also,^for^example,^the^target^range^of^relative^ unpaired^glycan^content^(e.g.,^relative^unpaired^afucosylated^glycan^content^and/or^the^relative^ unpaired^high^mannose^glycan^content)^may^be^the^range^of^relative^unpaired^glycan^content^(e.g.,^ relative^unpaired^afucosylated^glycan^content^and/or^the^relative^unpaired^high^mannose^glycan^ content)^for^a^reference^product.^^In^various^aspects,^the^target^range^is^a^predetermined^target^ range,^meaning^that^the^target^range^was^determined^or^ascertained^at^a^prior^timepoint.^^^ [0073]^^^^ ^Methods^of^Modifying^Unpaired^Glycan^Content^ [0074]^^^^ In^exemplary^embodiments,^the^method^of^modifying^(increasing^or^decreasing)^the^ADCC^ level^of^an^antibody^composition^comprises^modifying^(increasing^or^decreasing)^the^unpaired^ afucosylated^glycan^content^of^an^antibody^composition^and/or^the^unpaired^high^mannose^glycan^ content^of^an^antibody^composition.^^In^exemplary^aspects,^the^presently^disclosed^method^of^ modifying^the^ADCC^level^of^an^antibody^composition^comprises^increasing^the^unpaired^ afucosylated^glycan^content^and/or^the^unpaired^high^mannose^content^to^increase^the^level^of^ ADCC^activity.^^In^various^aspects,^the^method^of^modifying^the^ADCC^level^of^an^antibody^ composition^comprises^increasing^the^unpaired^afucosylated^glycan^content^and/or^the^unpaired^ high^mannose^content^by^at^least^about^1%,^at^least^about^2%,^at^least^about^3%,^at^least^about^4%,^ at^least^about^5%,^or^more.^^In^various^aspects,^the^method^of^modifying^the^ADCC^level^of^an^ antibody^composition^comprises^increasing^the^unpaired^afucosylated^glycan^content^and/or^the^ unpaired^high^mannose^content^by^more^than^5%^or^more^than^10%,^e.g.,^by^about^5%,^about^6%,^ ^
about^7%,^about^8%,^about^9%,^about^10%,^about^11%,^about^12%,^about^13%,^about^14%,^about^ 15%,^about^16%,^about^17%,^about^18%,^about^19%,^or^about^20%.^^In^various^aspects,^the^method^ comprises^increasing^the^unpaired^afucosylated^glycan^content^and/or^the^unpaired^high^mannose^ content^by^at^least^about^25%,^at^least^about^30%,^at^least^about^40%,^at^least^about^50%,^at^least^ about^60%,^at^least^about^70%,^at^least^about^80%,^at^least^about^90%,^or^more.^ [0075]^^^^ In^exemplary^aspects,^the^presently^disclosed^method^of^modifying^the^ADCC^level^of^an^ antibody^composition^comprises^decreasing^the^unpaired^afucosylated^glycan^content^to^decrease^ the^level^of^ADCC^activity.^^In^various^aspects,^the^method^of^modifying^the^ADCC^level^of^an^ antibody^composition^comprises^decreasing^the^unpaired^afucosylated^glycan^content^and/or^the^ unpaired^high^mannose^content^by^at^least^about^1%,^at^least^about^2%,^at^least^about^3%,^at^least^ about^4%,^at^least^about^5%,^or^more.^^In^various^aspects,^the^method^of^modifying^the^ADCC^level^of^ an^antibody^composition^comprises^decreasing^the^unpaired^afucosylated^glycan^content^and/or^the^ unpaired^high^mannose^content^by^more^than^5%^or^more^than^10%,^e.g.,^by^about^5%,^about^6%,^ about^7%,^about^8%,^about^9%,^about^10%,^about^11%,^about^12%,^about^13%,^about^14%,^about^ 15%,^about^16%,^about^17%,^about^18%,^about^19%,^or^about^20%.^^In^various^aspects,^the^method^ comprises^decreasing^the^unpaired^afucosylated^glycan^content^and/or^the^unpaired^high^mannose^ content^by^at^least^about^25%,^at^least^about^30%,^at^least^about^40%,^at^least^about^50%,^at^least^ about^60%,^at^least^about^70%,^at^least^about^80%,^at^least^about^90%,^or^more.^ [0076]^^^^ In^exemplary^aspects,^the^increase^or^decrease^in^the^unpaired^afucosylated^glycan^content^ and/or^unpaired^high^mannose^glycan^content^is/are^relative^to^a^“control”.^^In^exemplary^aspects,^ the^control^is^the^unpaired^glycan^content^of^a^control^protein^or^antibody^composition^produced^ under^the^same^cell^culture^conditions^with^exception^of^those^conditions^that^lead^to^increased^or^ decreased^unpaired^glycan^content.^^In^exemplary^aspects,^the^control^may^be^the^unpaired^glycan^ content^of^the^same^protein^or^antibody^composition^produced^under^a^first^set^of^cell^culture^ conditions^which^lead^to^an^ADCC^activity^level^which^is^outside^of^a^target^range^of^ADCC^activity^ level.^^In^various^aspects,^the^control^may^be^the^unpaired^glycan^content^of^the^same^protein^or^ antibody^composition^produced^under^a^first^set^of^cell^culture^conditions^which^lead^to^an^unpaired^ afucosylated^glycan^content^and/or^the^unpaired^high^mannose^glycan^content^which^is/are^outside^ of^a^target^range.^^^ [0077]^^^^ Without^being^bound^to^a^particular^theory,^conditions^which^lead^to^a^modified^(increased^ or^decreased)^afucosylated^glycan^content^and/or^high^mannose^content^may^lead^to^increased^or^ decreased^unpaired^afucosylated^glycan^content^and/or^unpaired^high^mannose^content^of^an^ antibody^composition.^^In^various^instances,^the^unpaired^afucosylated^glycan^content^and/or^ unpaired^high^mannose^content^is^increased^or^decreased^by^following^the^teachings^of^any^one^of^ ^
International^Patent^Application^Publication^Nos.^WO2013/114164,^WO2013/114245,^ WO2013/114167,^WO2015128793,^or^WO2016/089919,^WO2018/170099,^WO2019/191150,^each^ of^which^is^incorporated^herein^by^reference.^^In^various^instances,^the^unpaired^afucosylated^glycan^ content^and/or^unpaired^high^mannose^content^is^increased^or^decreased^by^selecting^a^clone^that^ produces^antibody^or^antibody^protein^product^comprising^a^level^of^unpaired^afucosylated^glycan^ content^and/or^unpaired^high^mannose^content^within^a^target^range.^ [0078]^^^^ Methods^of^Producing^Antibody^Compositions^ [0079]^^^^ Simple^and^efficient^methods^to^predict^the^level^of^effector^function^(e.g.,^ADCC)^a^ particular^antibody^composition^will^exhibit^based^on^a^given^glycoform^profile^for^that^antibody^ composition^are^described^herein.^^The^data^provided^herein^support^that^the^ADCC^activity^level^for^ the^antibody^composition^may^be^predicted^based^on^the^relative^unpaired^glycan^content^of^an^ antibody^composition.^^Without^being^bound^to^a^particular^theory,^the^unpaired^afucosylated^ glycan^content^and/or^the^relative^unpaired^high^mannose^glycan^content^of^an^antibody^ composition^is^predictive^of^the^ADCC^activity^level^of^the^antibody^composition.^^Such^predicted^ ADCC^levels^are^useful^during^antibody^production,^when,^it^is^necessary^for^the^antibody^to^have^an^ ADCC^activity^level^within^a^target^range.^^For^instance,^by^monitoring^the^unpaired^afucosylated^ glycan^content^and/or^the^unpaired^high^mannose^glycan^content^of^an^antibody^composition,^it^may^ be^predicted^whether^the^antibody^composition^will^exhibit^an^ADCC^activity^level^within^a^target^ range.^^If^a^target^range^of^ADCC^activity^levels^for^an^antibody^composition^is^known,^the^target^ range^of^relative^unpaired^afucosylated^glycan^content^and/or^the^relative^unpaired^high^mannose^ glycan^content^may^be^determined.^^Selection^of^the^antibody^composition^for^continued^processing,^ e.g.,^downstream^processing,^may^occur^when^the^unpaired^afucosylated^glycan^content^and/or^the^ unpaired^high^mannose^glycan,^or^the^ADCC^activity^level,^as^calculated^based^on^the^unpaired^ afucosylated^glycan^content^and/or^the^unpaired^high^mannose^glycan^content,^is/are^within^a^target^ range.^In^exemplary^aspects,^the^target^range^is^based^on^a^target^range^of^ADCC^activity^levels^for^a^ reference^antibody^and^a^model^which^correlates^ADCC^activity^level^of^the^antibody^composition^to^ afucosylated^glycan^content^and/or^high^mannose^glycan^content^of^the^antibody^composition,^ optionally,^a^model^which^correlates^ADCC^activity^level^of^the^antibody^composition^to^unpaired^ afucosylated^glycan^content^and/or^unpaired^high^mannose^glycan^content^of^the^antibody^ composition.^^^ [0080]^^^^ Accordingly,^the^present^disclosure^provides^methods^of^producing^an^antibody^ composition.^^In^exemplary^embodiments,^the^method^comprises^(i)^determining^the^relative^ unpaired^afucosylated^glycan^content^and/or^the^relative^unpaired^high^mannose^glycan^content^of^a^ sample^of^the^antibody^composition;^and^(ii)^selecting^the^antibody^composition^for^downstream^ ^
processing^based^on^the^relative^unpaired^afucosylated^glycan^content^and/or^relative^unpaired^high^ mannose^glycan^content^determined^in^(i).^^In^exemplary^embodiments,^the^method^of^producing^an^ antibody^composition^comprises^(i)^determining^the^unpaired^afucosylated^glycan^content^of^an^ antibody^composition^and/or^the^unpaired^high^mannose^glycan^content^of^an^antibody^ composition;^(ii)^determining^the^ADCC^level^of^the^antibody^composition^based^on^the^unpaired^ afucosylated^glycan^content^and/or^the^unpaired^high^mannose^glycan^content^determined^in^(i);^ and^(iii)^selecting^the^antibody^composition^for^downstream^processing^when^the^ADCC^level^of^the^ antibody^composition^determined^in^(ii)^is^within^a^target^ADCC^range.^^In^exemplary^embodiments,^ the^method^of^producing^an^antibody^composition^comprises^(i)^determining^the^unpaired^ afucosylated^glycan^content^and/or^the^unpaired^high^mannose^glycan^content^of^a^sample^of^the^ antibody^composition^taken^from^a^cell^culture^comprising^glycosylation^competent^cells^expressing^ an^antibody^of^the^antibody^composition;^(ii)^optionally,^modifying^the^cell^culture^to^modulate^the^ unpaired^afucosylated^glycan^content^and/or^the^unpaired^high^mannose^glycan^content^and^ determining^the^unpaired^afucosylated^glycan^content^and/or^the^unpaired^high^mannose^glycan^ content^of^a^sample^of^the^antibody^composition^taken^from^the^modified^cell^culture;^and^(iii)^ selecting^the^antibody^composition^for^downstream^processing^based^on^the^unpaired^afucosylated^ glycan^content^and/or^unpaired^high^mannose^glycan^content.^ [0081]^^^^ Also^provided^herein^are^methods^of^preparing^an^IgG^antibody^composition^having^an^ ADCC^activity^level^within^a^target^range.^^In^exemplary^embodiments,^the^method^comprises^ determining^the^relative^unpaired^glycan^content^of^a^sample^of^the^IgG^antibody^composition^in^ accordance^with^any^one^of^the^presently^disclosed^methods^of^determining^the^relative^unpaired^ glycan^content.^^The^relative^unpaired^AF^glycan^content^and/or^relative^unpaired^HM^glycan^content^ of^the^IgG^antibody^composition^may^be^compared^to^a^target^range^of^unpaired^AF^glycan^content^ and/or^relative^unpaired^HM^glycan^content.^^For^example,^the^target^range^may^be^a^reference^level^ of^a^specification,^or^of^a^reference^IgG^antibody^composition.^^Also,^for^instance,^the^target^range^of^ unpaired^AF^glycan^content^and/or^relative^unpaired^HM^glycan^content^may^be^a^predetermined^ target^range^of^unpaired^AF^glycan^content^and/or^relative^unpaired^HM^glycan^content^which^ statistically^correlates^with^a^predetermined^target^range^of^ADCC^activity^levels^of^a^reference^IgG^ antibody^composition.^ [0082]^^^^ Methods^of^producing^an^antibody^composition^are^also^provided^by^the^present^ disclosure.^^In^exemplary^embodiments,^the^method^comprises^determining^relative^unpaired^glycan^ content^of^the^antibody^composition^is^determined^in^accordance^with^any^one^of^the^presently^ disclosed^methods^of^determining^the^relative^unpaired^glycan^content,^wherein,^when^the^relative^ unpaired^glycan^content^is^determined^as^outside^a^predetermined^target^range,^the^method^further^ ^
comprises^modifying^one^or^more^conditions^of^the^cell^culture^to^obtain^a^modified^cell^culture^and^ determining^the^relative^unpaired^glycan^content.^^The^method^in^various^instance^comprises^ repeating^steps^of^the^method^until^the^relative^unpaired^glycan^content^is^within^the^predetermined^ target^range.^ [0083]^^^^ In^various^aspects,^the^sample^is^taken^from^a^cell^culture^comprising^glycosylation^ competent^cells^expressing^an^antibody^of^the^antibody^composition.^^Optionally,^the^method^ further^comprises^modifying^one^or^more^conditions^of^the^cell^culture^to^modify^the^relative^ unpaired^afucosylated^glycan^content^and/or^the^relative^unpaired^high^mannose^glycan^content^of^ the^antibody^composition^and^determining^the^relative^unpaired^afucosylated^glycan^content^and/or^ the^relative^unpaired^high^mannose^glycan^content^of^a^sample^of^the^antibody^composition^taken^ from^the^modified^cell^culture.^^In^exemplary^aspects,^the^method^comprises^repeating^the^modifying^ until^the^relative^unpaired^afucosylated^glycan^content^and/or^relative^unpaired^high^mannose^ glycan^content^is^within^a^target^range.^^In^various^aspects,^the^relative^unpaired^afucosylated^glycan^ content^and/or^the^relative^unpaired^high^mannose^glycan^content^is/are^determined^in^real^time^ with^respect^to^production^of^the^antibody^composition.^^As^used^herein^“real^time”^refers^to^ determinations^that^are^made^while^a^production^process^is^ongoing,^without^interruption^to^the^ process.^^It^will^be^appreciated^that^production^of^therapeutic^proteins^involves^living^cells^and^ sensitive^materials^that^cannot^be^put^on^hold^indefinitely^while^assays^and^determinations^are^ performed.^^If^numerical^examples^of^interest,^a^“real^time”^determination^can^be^a^determination^ that^is^made^within^6^hours,^5^hours,^4^hours,^3^hours,^2^hours,^1^hour,^50^minutes,^40^minutes,^30^ minutes,^20^minute,^10^minutes,^5^minutes,^1^minute,^30^seconds,^20^seconds,^10^seconds,^5^ seconds,^1^seconds,^or^0.1^seconds^from^the^time^a^measurement^is^made^(while^the^production^ process^is^ongoing).^^^In^various^instances,^the^method^comprises^selecting^the^antibody^composition^ for^downstream^processing^when^the^relative^unpaired^afucosylated^glycan^content^and/or^relative^ unpaired^high^mannose^glycan^content^is/are^in^a^target^range.^^In^various^aspects,^the^relative^ unpaired^afucosylated^glycan^content^and/or^relative^unpaired^high^mannose^glycan^content^ correlate^with^the^ADCC^activity^level^of^the^antibody^composition.^^Optionally,^the^method^further^ comprises^determining^the^ADCC^activity^level^of^the^antibody^composition^based^on^the^relative^ unpaired^afucosylated^glycan^content^and/or^relative^unpaired^high^mannose^glycan^content^ determined^in^(i).^^Optionally,^the^method^comprises^selecting^the^antibody^composition^for^ downstream^processing^when^the^ADCC^activity^level^is^in^a^target^range.^ [0084]^^^^ In^various^aspects,^the^method^of^producing^an^antibody^composition^comprises^modifying^ the^ADCC^level^of^an^antibody^composition^according^to^a^method^of^modifying^the^ADCC^level^of^the^ present^disclosure.^^^ ^
[0085]^^^^ In^various^instances,^the^method^of^producing^an^antibody^composition^comprises^ determining^the^relative^unpaired^glycan^content^of^an^antibody^composition^according^to^any^of^the^ presently^disclosed^methods^of^determining^the^relative^unpaired^glycan^content^of^an^antibody^ composition.^^^ [0086]^^^^ Downstream^Processing^ [0087]^^^^ The^relative^%^unpaired^high^mannose^glycans^and/or^relative^%^unpaired^afucosylated^ glycans^are^determined^(e.g.,^measured)^to^better^inform^as^to^the^%^antibody^dependent^cell^ mediated^cytotoxicity^(ADCC)^of^the^antibody^composition.^^The^determining^(e.g.,^measuring)^may^ occur^at^any^stage^of^manufacture.^^In^particular,^measurements^may^be^taken^pre^^or^post^harvest,^ preceding^or^during^any^stage^of^downstream^processing.^^Example^downstream^processing^includes^ any^chromatography^unit^operation,^including^capture^chromatography,^intermediate^ chromatography,^and/or^polish^chromatography^unit^operations;^virus^inactivation^and^ neutralization;^virus^filtration;^and/or^final^formulation.^^The^relative^%^unpaired^high^mannose^ glycans,^and/or^relative^%^unpaired^afucosylated^glycans^in^various^aspects^is^determined^(e.g.,^ measured)^in^real^time,^near^real^time,^and/or^after^the^fact.^^Monitoring^and^measurements^can^be^ done^using^known^techniques^and^commercially^available^equipment.^^^ [0088]^^^^ In^various^aspects^of^the^present^disclosure,^the^determining^(e.g.,^measuring)^the^%^ unpaired^high^mannose^glycans^and/or^%^unpaired^afucosylated^glycans^is^carried^out^before^a^ harvest.^^As^used^herein^the^term^“harvest”^refers^to^cell^culture^media^containing^the^recombinant^ protein^of^interest^being^collected^and^separated^at^least^from^the^cells^of^the^cell^culture.^^The^ harvest^can^be^performed^continuously.^^The^harvest^in^some^aspects^is^performed^using^ centrifugation^and^can^further^comprise^precipitation,^filtration,^and^the^like.^^In^various^aspects,^the^ determining^is^carried^out^before^harvest.^^In^various^aspects,^the^determining^is^carried^out^before^ chromatography,^optionally,^Protein^A^chromatography.^^In^some^aspects,^the^determining^(e.g.,^ measuring)^the^relative^%^unpaired^high^mannose^glycans^and/or^relative^%^unpaired^afucosylated^ glycans^is^carried^out^at^least^3^days,^at^least^4^days,^or^at^least^5^days^before^harvest.^^Optionally,^ determining^(e.g.,^measuring)^the^relative^%^unpaired^high^mannose^glycans^and/or^relative^%^ unpaired^afucosylated^glycans^is^carried^out^in^real^time^with^regard^to^antibody^production.^^^ [0089]^^^^ In^various^aspects^of^the^present^disclosure,^determining^(e.g.,^measuring)^the^relative^%^ unpaired^high^mannose^glycans,^and/or^relative^%^unpaired^afucosylated^glycans^is^carried^out^after^ a^harvest.^^In^various^aspects,^the^determining^is^carried^out^after^chromatography,^optionally,^ Protein^A^chromatography.^^In^various^aspects,^the^determining^is^carried^out^after^harvest^and^after^ chromatography,^e.g.,^a^Protein^A^chromatography.^^^ ^
[0090]^^^^ With^regard^to^the^presently^disclosed^methods,^the^antibody^composition^in^various^ aspects^is^selected^or^chosen^for^further^processing,^e.g.,^for^downstream^processing,^and^the^ selection^is^based^on^a^particular^parameter,^e.g.,^%^ADCC,^relative^%^unpaired^high^mannose^ glycans,^and/or^relative^%^unpaired^afucosylated^glycans.^^In^various^instances,^the^presently^ disclosed^methods^comprise^using^the^antibody^composition^in^further^processing,^e.g.,^downstream^ processing,^based^on^a^particular^parameter,^e.g.,^based^on^the^%^ADCC,^relative^%^unpaired^high^ mannose^glycans,^and/or^relative^%^unpaired^afucosylated^glycans.^^In^various^instances,^the^ presently^disclosed^methods^comprise^carrying^out^further^processing,^e.g.,^downstream^processing,^ with^the^antibody^composition,^based^on^a^particular^parameter,^e.g.,^based^on^the^%^ADCC,^relative^ %^unpaired^high^mannose^glycans,^and/or^relative^%^unpaired^afucosylated^glycans.^^^ [0091]^^^^ In^exemplary^instances^the^downstream^processing^comprises^or^consists^of^any^processing^ which^occurs^after^(or^downstream^of)^the^processing^at^which^the^relative^%^unpaired^high^ mannose^glycans,^and/or^relative^%^unpaired^afucosylated^glycans^are^determined^(e.g.,^measured).^^ For^example,^if^the^relative^%^unpaired^high^mannose^glycans,^and/or^relative^%^unpaired^ afucosylated^glycans^were^determined^(e.g.,^measured)^at^harvest,^then^the^downstream^processing^ is^any^processing^which^occurs^after^(or^downstream^of)^the^harvest,^which^in^various^aspects^ comprise(s):^dilution,^filling,^filtration,^formulation,^chromatography,^viral^filtration,^viral^ inactivation,^or^a^combination^thereof.^^Also,^for^example,^if^the^relative^%^unpaired^high^mannose^ glycans,^and/or^relative^%^unpaired^afucosylated^glycans^were^determined^(e.g.,^measured)^after^ chromatography,^e.g.,^Protein^A^chromatography,^then^the^downstream^processing^comprises^or^ consists^of^any^processing^which^occurs^after^(or^downstream^of)^the^chromatography,^and^the^ downstream^processing^in^various^aspects^comprise(s):^a^dilution,^a^filling,^a^filtration,^a^formulation,^ further^chromatography,^a^viral^filtration,^a^viral^inactivation,^or^a^combination^thereof.^^In^ exemplary^instances^the^further^chromatography^is^an^ion^exchange^chromatography^(e.g.,^cation^ exchange^chromatography^or^anion^exchange^chromatography).^ [0092]^^^^ Stages/types^of^chromatography^used^during^downstream^processing^include^capture^or^ affinity^chromatography^which^is^used^to^separate^the^recombinant^product^from^other^proteins,^ aggregates,^DNA,^viruses^and^other^such^impurities.^^In^exemplary^instances,^initial^chromatography^ is^carried^out^with^Protein^A^(e.g.,^Protein^A^attached^to^a^resin).^^Intermediate^and^polish^ chromatography^in^various^aspects^further^purify^the^recombinant^protein,^removing^bulk^ contaminants,^adventitious^viruses,^trace^impurities,^aggregates,^isoforms,^etc.^^The^chromatography^ can^either^be^performed^in^bind^and^elute^mode,^where^the^recombinant^protein^of^interest^is^ bound^to^the^chromatography^medium^and^the^impurities^flow^through,^or^in^flow^through^mode,^ where^the^impurities^are^bound^and^the^recombinant^protein^flows^through.^^Examples^of^such^ ^
chromatography^methods^include^ion^exchange^chromatography^(IEX),^such^as^anion^exchange^ chromatography^(AEX)^and^cation^exchange^chromatography^(CEX);^hydrophobic^interaction^ chromatography^(HIC);^mixed^modal^or^multimodal^chromatography^(MM),^hydroxyapatite^ chromatography^(HA);^reverse^phase^chromatography^and^gel^filtration.^^^^^ [0093]^^^^ In^various^aspects,^the^downstream^processing^comprises^viral^inactivation.^^Enveloped^ viruses^have^a^capsid^enclosed^by^a^lipoprotein^membrane^or^“envelope”^and^are^therefore^ susceptible^to^inactivation.^^The^virus^inactivation^in^various^instances^includes^heat^ inactivation/pasteurization,^pH^inactivation,^UV^and^gamma^ray^irradiation,^use^of^high^intensity^ broad^spectrum^white^light,^addition^of^chemical^inactivating^agents,^surfactants,^and^ solvent/detergent^treatments.^^^ [0094]^^^^ In^various^aspects,^the^downstream^processing^comprises^virus^filtration.^^In^various^ aspects,^the^virus^filtration^comprises^removing^non^enveloped^viruses.^^In^various^aspects,^the^virus^ filtration^comprises^the^use^of^micro^^or^nano^filters.^ [0095]^^^^ In^various^aspects,^the^downstream^processing^comprises^formulation,^which^may^be^ performed^in^one^or^more^steps.^^Following^completion^of^the^chromatography,^the^purified^ recombinant^proteins^are^in^various^aspects^buffer^exchanged^into^a^formulation^buffer.^In^ exemplary^aspects,^the^buffer^exchange^is^performed^using^ultrafiltration^and^diafiltration^(UF/DF).^^ In^exemplary^aspects,^the^recombinant^protein^is^buffer^exchanged^into^a^desired^formulation^buffer^ using^diafiltration^and^concentrated^to^a^desired^final^formulation^concentration^using^ultrafiltration.^^ Additional^stability^enhancing^excipients^in^various^aspects^are^added^following^a^UF/DF^formulation.^ [0096]^^^^ Additional^Steps^ [0097]^^^^ The^methods^disclosed^herein,^in^various^aspects,^comprise^additional^steps.^^For^example,^ in^some^aspects,^the^methods^comprise^one^or^more^upstream^steps^or^downstream^steps^involved^ in^producing,^purifying,^and^formulating^an^antibody^composition.^^Optionally,^the^downstream^steps^ are^any^one^of^those^downstream^processing^steps^described^herein^or^known^in^the^art.^^See,^e.g.,^ Downstream^Processing.^In^exemplary^embodiments,^the^method^comprises^steps^for^generating^ host^cells^that^express^a^recombinant^glycosylated^protein^(e.g.,^antibody).^^The^host^cells,^in^some^ aspects,^are^prokaryotic^host^cells,^e.g.,^E.^coli^or^Bacillus^subtilis,^or^the^host^cells,^in^some^aspects,^ are^eukaryotic^host^cells,^e.g.,^yeast^cells,^filamentous^fungi^cells,^protozoa^cells,^insect^cells,^or^ mammalian^cells^(e.g.,^CHO^cells).^^Such^host^cells^are^described^in^the^art.^^See,^e.g.,^Kunert^et^al.,^ Appl.^Microbiol^Biotechnol.^100:^3451^61^(2016)^and^herein^under^“Cells.”^^For^example,^the^ methods^comprise,^in^some^instances,^introducing^into^host^cells^a^vector^comprising^a^nucleic^acid^ ^
comprising^a^nucleotide^sequence^encoding^the^recombinant^glycosylated^protein,^or^a^polypeptide^ chain^thereof.^^^ [0098]^^^^ In^exemplary^aspects,^the^methods^comprise^maintaining^cells,^e.g.,^glycosylation^ competent^cells^in^a^cell^culture.^^Accordingly,^the^methods^may^comprise^carrying^out^any^one^or^ more^steps^described^herein^in^Maintaining^Cells^In^A^Cell^Culture.^ [0099]^^^^ In^exemplary^embodiments,^the^methods^disclosed^herein^comprise^steps^for^isolating^ and/or^purifying^the^antibodies^from^the^culture.^^In^exemplary^aspects,^the^method^comprises^one^ or^more^chromatography^steps^including,^but^not^limited^to,^e.g.,^affinity^chromatography^(e.g.,^ protein^A^affinity^chromatography),^ion^exchange^chromatography,^and/or^hydrophobic^interaction^ chromatography.^^In^exemplary^aspects,^the^method^comprises^steps^for^producing^crystalline^ biomolecules^from^a^solution^comprising^the^recombinant^glycosylated^proteins.^^^ [00100]^^^^ The^methods^of^the^disclosure,^in^various^aspects,^comprise^one^or^more^steps^for^ preparing^a^composition,^including,^in^some^aspects,^a^pharmaceutical^composition,^comprising^the^ purified^recombinant^glycosylated^protein.^^Such^compositions^are^discussed^herein.^ [00101]^^^^ Maintaining^Cells^In^A^Cell^Culture^^ [00102]^^^^ With^regard^to^the^methods^of^producing^an^antibody^composition^of^the^present^ disclosure,^the^antibody^composition^may^be^produced^by^maintaining^cells^in^a^cell^culture.^^The^cell^ culture^may^be^maintained^according^to^any^set^of^conditions^suitable^for^production^of^a^ recombinant^glycosylated^protein.^^For^example,^in^some^aspects,^the^cell^culture^is^maintained^at^a^ particular^pH,^temperature,^cell^density,^culture^volume,^dissolved^oxygen^level,^pressure,^ osmolality,^and^the^like.^^In^exemplary^aspects,^the^cell^culture^prior^to^inoculation^is^shaken^(e.g.,^at^ 70^rpm)^at^5%^CO
2^under^standard^humidified^conditions^in^a^CO
2^incubator.^^In^exemplary^aspects,^ the^cell^culture^is^inoculated^with^a^seeding^density^of^about^10
6^cells/mL^in^1.5^L^medium.^^^ [00103]^^^^ In^exemplary^aspects,^the^methods^of^the^disclosure^comprise^maintaining^the^ glycosylation^competent^cells^in^a^cell^culture^medium^at^a^pH^of^about^6.85^to^about^7.05,^e.g.,^in^ various^aspects,^about^6.85,^about^6.86,^about^6.87,^about^6.88,^about^6.89,^about^6.90,^about^6.91,^ about^6.92,^about^6.93,^about^6.94,^about^6.95,^about^6.96,^about^6.97,^about^6.98,^about^6.99,^ about^7.00,^about^7.01,^about^7.02,^about^7.03,^about^7.04,^or^about^7.05.^^^ [00104]^^^^ In^exemplary^aspects,^the^methods^comprise^maintaining^the^cell^culture^at^a^ temperature^between^30ºC^and^40ºC.^^In^exemplary^embodiments,^the^temperature^is^between^ about^32ºC^to^about^38ºC^or^between^about^35ºC^to^about^38ºC.^^^ ^
[00105]^^^^ In^exemplary^aspects,^the^methods^comprise^maintaining^the^osmolality^between^about^ 200^mOsm/kg^to^about^500^mOsm/kg.^^In^exemplary^aspects,^the^method^comprises^maintaining^the^ osmolality^between^about^225^mOsm/kg^to^about^400^mOsm/kg^or^about^225^mOsm/kg^to^about^ 375^mOsm/kg.^^In^exemplary^aspects,^the^method^comprises^maintaining^the^osmolality^between^ about^225^mOsm/kg^to^about^350^mOsm/kg.^^In^various^aspects,^osmolality^(mOsm/kg)^is^ maintained^at^about^200,^225,^about^250,^about^275,^about^300,^about^325,^about^350,^about^375,^ about^400,^about^425,^about^450,^about^475,^or^about^500.^ [00106]^^^^ In^exemplary^aspects,^the^methods^comprise^maintaining^dissolved^the^oxygen^(DO)^level^ of^the^cell^culture^at^about^20%^to^about^60%^oxygen^saturation^during^the^initial^cell^culture^period.^^ In^exemplary^instances,^the^method^comprises^maintaining^DO^level^of^the^cell^culture^at^about^30%^ to^about^50%^(e.g.,^about^35%^to^about^45%)^oxygen^saturation^during^the^initial^cell^culture^period.^^ In^exemplary^instances,^the^method^comprises^maintaining^DO^level^of^the^cell^culture^at^about^20%,^ about^25%,^about^30%,^about^35%,^about^40%,^about^45%,^about^50%,^about^55%,^or^about^60%^ oxygen^saturation^during^the^initial^cell^culture^period.^^In^exemplary^aspects,^the^DO^level^is^about^ 35^mm^Hg^to^about^85^mmHg^or^about^40^mm^Hg^to^about^80^mmHg^or^about^45^mm^Hg^to^about^ 75^mm^Hg.^ [00107]^^^^ The^cell^culture^is^maintained^in^any^one^or^more^culture^medium.^^In^exemplary^aspects,^ the^cell^culture^is^maintained^in^a^medium^suitable^for^cell^growth^and/or^is^provided^with^one^or^ more^feeding^media^according^to^any^suitable^feeding^schedule.^^In^exemplary^aspects,^the^method^ comprises^maintaining^the^cell^culture^in^a^medium^comprising^glucose,^fucose,^lactate,^ammonia,^ glutamine,^and/or^glutamate.^^In^exemplary^aspects,^the^method^comprises^maintaining^the^cell^ culture^in^a^medium^comprising^manganese^at^a^concentration^less^than^or^about^1^μM^during^the^ initial^cell^culture^period.^^In^exemplary^aspects,^the^method^comprises^maintaining^the^cell^culture^ in^a^medium^comprising^about^0.25^μM^to^about^1^μM^manganese.^^In^exemplary^aspects,^the^ method^comprises^maintaining^the^cell^culture^in^a^medium^comprising^negligible^amounts^of^ manganese.^^In^exemplary^aspects,^the^method^comprises^maintaining^the^cell^culture^in^a^medium^ comprising^copper^at^a^concentration^less^than^or^about^50^ppb^during^the^initial^cell^culture^period.^^ In^exemplary^aspects,^the^method^comprises^maintaining^the^cell^culture^in^a^medium^comprising^ copper^at^a^concentration^less^than^or^about^40^ppb^during^the^initial^cell^culture^period.^^In^ exemplary^aspects,^the^method^comprises^maintaining^the^cell^culture^in^a^medium^comprising^ copper^at^a^concentration^less^than^or^about^30^ppb^during^the^initial^cell^culture^period.^^In^ exemplary^aspects,^the^method^comprises^maintaining^the^cell^culture^in^a^medium^comprising^ copper^at^a^concentration^less^than^or^about^20^ppb^during^the^initial^cell^culture^period.^^In^ exemplary^aspects,^the^medium^comprises^copper^at^a^concentration^greater^than^or^about^5^ppb^or^ ^
greater^than^or^about^10^ppb.^^In^exemplary^aspects,^the^cell^culture^medium^comprises^mannose.^^ In^exemplary^aspects,^the^cell^culture^medium^does^not^comprise^mannose.^ [00108]^^^^ In^exemplary^embodiments,^the^type^of^cell^culture^is^a^fed^batch^culture^or^a^continuous^ perfusion^culture.^^However,^the^methods^of^the^disclosure^are^advantageously^not^limited^to^any^ particular^type^of^cell^culture.^^^ [00109]^^^^ The^cells^maintained^in^cell^culture^may^be^glycosylation^competent^cells.^^In^exemplary^ aspects,^the^glycosylation^competent^cells^are^eukaryotic^cells,^including,^but^not^limited^to,^yeast^ cells,^filamentous^fungi^cells,^protozoa^cells,^algae^cells,^insect^cells,^or^mammalian^cells.^^Such^host^ cells^are^described^in^the^art.^^See,^e.g.,^Kunert^et^al.,^Appl.^Microbiol^Biotechnol.^100:^3451^61^ (2016).^^In^exemplary^aspects,^the^eukaryotic^cells^are^mammalian^cells.^^In^exemplary^aspects,^the^ mammalian^cells^are^non^human^mammalian^cells.^^In^some^aspects,^the^cells^are^Chinese^Hamster^ Ovary^(CHO)^cells^and^derivatives^thereof^(e.g.,^CHO^K1,^CHO^pro^3),^mouse^myeloma^cells^(e.g.,^NS0,^ GS^NS0,^Sp2/0),^cells^engineered^to^be^deficient^in^dihydrofolatereductase^(DHFR)^activity^(e.g.,^ DUKX^X11,^DG44),^human^embryonic^kidney^293^(HEK293)^cells^or^derivatives^thereof^(e.g.,^ HEK293T,^HEK293^EBNA),^green^African^monkey^kidney^cells^(e.g.,^COS^cells,^VERO^cells),^human^ cervical^cancer^cells^(e.g.,^HeLa),^human^bone^osteosarcoma^epithelial^cells^U2^OS,^adenocarcinomic^ human^alveolar^basal^epithelial^cells^A549,^human^fibrosarcoma^cells^HT1080,^mouse^brain^tumor^ cells^CAD,^embryonic^carcinoma^cells^P19,^mouse^embryo^fibroblast^cells^NIH^3T3,^mouse^ fibroblast^cells^L929,^mouse^neuroblastoma^cells^N2a,^human^breast^cancer^cells^MCF^7,^ retinoblastoma^cells^Y79,^human^retinoblastoma^cells^SO^Rb50,^human^liver^cancer^cells^Hep^G2,^ mouse^B^myeloma^cells^J558L,^or^baby^hamster^kidney^(BHK)^cells^(Gaillet^et^al.^2007;^Khan,^Adv^ Pharm^Bull^3(2):^257^263^(2013)).^^^ [00110]^^^^ Cells^that^are^not^glycosylation^competent^can^also^be^transformed^into^glycosylation^ competent^cells,^e.g.^by^transfecting^them^with^genes^encoding^relevant^enzymes^necessary^for^ glycosylation.^^Exemplary^enzymes^include^but^are^not^limited^to^oligosaccharyltransferases,^ glycosidases,^glucosidase^I,^glucosidease^II,^calnexin/calreticulin,^glycosyltransferases,^mannosidases,^ GlcNAc^transferases,^galactosyltransferases,^and^sialyltransferases.^ [00111]^^^^ In^exemplary^embodiments,^the^glycosylation^competent^cells^are^not^genetically^ modified^to^alter^the^activity^of^an^enzyme^of^the^de^novo^pathway^or^the^salvage^pathway.^^These^ two^pathways^of^fucose^metabolism^are^shown^in^Figure^2.^^In^exemplary^embodiments,^the^ glycosylation^competent^cells^are^not^genetically^modified^to^alter^the^activity^of^any^one^or^more^of:^^ a^fucosyl^transferase^(FUT,^e.g.,FUT1,^FUT2,^FUT3,^FUT4,^FUT5,^FUT6,^FUT7,^FUT8,^FUT9),^a^fucose^ kinase,^a^GDP^fucose^pyrophosphorylase,^GDP^D^mannose^4,6^dehydratase^(GMD),^and^GDP^keto^6^ ^
deoxymannose^3,5^epimerase,^4^reductase^(FX).^^In^exemplary^embodiments,^the^glycosylation^ competent^cells^are^not^genetically^modified^to^knock^out^a^gene^encoding^FX.^ [00112]^^^^ In^exemplary^embodiments,^the^glycosylation^competent^cells^are^not^genetically^ modified^to^alter^the^activity^ɴ(1,4)^N^acetylglucosaminyltransferase^III^(GNTIII)^or^GDP^6^deoxy^D^ lyxo^4^hexulose^reductase^(RMD).^^In^exemplary^aspects,^the^glycosylation^competent^cells^are^not^ genetically^modified^to^overexpress^GNTIII^or^RMD.^^^ [00113]^^^^ Antibodies^and^Fragments^Thereof^ [00114]^^^^ As^used^herein,^the^term^“antibody”^refers^to^a^protein^having^a^conventional^ immunoglobulin^format,^comprising^heavy^and^light^chains,^and^comprising^variable^and^constant^ regions.^^For^example,^an^antibody^may^be^an^IgG^which^is^a^“Y^shaped”^structure^of^two^identical^ pairs^of^polypeptide^chains,^each^pair^having^one^“light”^(typically^having^a^molecular^weight^of^ about^25^kDa)^and^one^“heavy”^chain^(typically^having^a^molecular^weight^of^about^50^70^kDa)..^^An^ antibody^has^a^variable^region^and^a^constant^region.^In^IgG^formats,^the^variable^region^is^generally^ about^100^110^or^more^amino^acids,^comprises^three^complementarity^determining^regions^(CDRs),^ is^primarily^responsible^for^antigen^recognition,^and^substantially^varies^among^other^antibodies^that^ bind^to^different^antigens.^^See,^e.g.,^Janeway^et^al.,^“Structure^of^the^Antibody^Molecule^and^the^ Immunoglobulin^Genes”,^Immunobiology:^^The^Immune^System^in^Health^and^Disease,^4th^ed.^ Elsevier^Science^Ltd./Garland^Publishing,^(1999).^^^ [00115]^^^^ Briefly,^in^an^antibody^scaffold,^the^CDRs^are^embedded^within^a^framework^in^the^heavy^ and^light^chain^variable^region^where^they^constitute^the^regions^largely^responsible^for^antigen^ binding^and^recognition.^A^variable^region^comprises^at^least^three^heavy^or^light^chain^CDRs^(Kabat^ et^al.,^1991,^Sequences^of^Proteins^of^Immunological^Interest,^Public^Health^Service^N.I.H.,^Bethesda,^ Md.;^see^also^Chothia^and^Lesk,^1987,^J.^Mol.^Biol.^196:901^917;^Chothia^et^al.,^1989,^Nature^342:^ 877^883),^within^a^framework^region^(designated^framework^regions^1^4,^FR1,^FR2,^FR3,^and^FR4,^by^ Kabat^et^al.,^1991;^see^also^Chothia^and^Lesk,^1987,^supra).^^ [00116]^^^^ Human^light^chains^are^classified^as^kappa^and^lambda^light^chains.^Heavy^chains^are^ classified^as^mu,^delta,^gamma,^alpha,^or^epsilon,^and^define^the^antibody's^isotype^as^IgM,^IgD,^IgG,^ IgA,^and^IgE,^respectively.^IgG^has^several^subclasses,^including,^but^not^limited^to^IgG1,^IgG2,^IgG3,^ and^IgG4.^IgM^has^subclasses,^including,^but^not^limited^to,^IgM1^and^IgM2.^The^light^chain^constant^ region^can^be,^for^example,^a^kappa^^or^lambda^type^light^chain^constant^region,^e.g.,^a^human^ kappa^^or^lambda^type^light^chain^constant^region.^The^heavy^chain^constant^region^can^be,^for^ example,^an^alpha^,^delta^,^epsilon^,^gamma^,^or^mu^type^heavy^chain^constant^regions,^e.g.,^a^ human^alpha^,^delta^,^epsilon^,^gamma^,^or^mu^type^heavy^chain^constant^region.^^In^various^aspects,^ ^
the^antibody^is^an^IgG,^including^any^one^of^IgG1,^IgG2,^IgG3^or^IgG4.^^In^various^instances,^the^ antibody^is^an^IgG1.^ [00117]^^^^ In^various^aspects,^the^antibody^can^be^a^monoclonal^antibody^or^a^polyclonal^antibody.^^ In^exemplary^instances,^the^antibody^is^a^mammalian^antibody,^e.g.,^a^mouse^antibody,^rat^antibody,^ rabbit^antibody,^goat^antibody,^horse^antibody,^chicken^antibody,^hamster^antibody,^pig^antibody,^ human^antibody,^and^the^like.^^In^certain^aspects,^the^recombinant^glycosylated^protein^is^a^ monoclonal^human^antibody.^^^^^ [00118]^^^^ In^various^aspects,^the^antibody^is^a^chimeric^antibody^or^a^humanized^antibody.^^The^ term^"chimeric^antibody"^is^used^herein^to^refer^to^an^antibody^containing^constant^domains^from^ one^species^and^the^variable^domains^from^a^second,^or^more^generally,^containing^stretches^of^ amino^acid^sequence^from^at^least^two^species.^^The^term^"humanized"^when^used^in^relation^to^ antibodies^refers^to^antibodies^having^at^least^CDR^regions^from^a^non^human^source^which^are^ engineered^to^have^a^structure^and^immunological^function^more^similar^to^true^human^antibodies^ than^the^original^source^antibodies.^^For^example,^humanizing^can^involve^grafting^CDR^from^a^non^ human^antibody,^such^as^a^mouse^antibody,^into^a^human^antibody.^^Humanizing^also^can^involve^ select^amino^acid^substitutions^to^make^a^non^human^sequence^look^more^like^a^human^sequence.^^^ [00119]^^^^ An^antibody,^in^various^aspects,^is^cleaved^into^fragments^by^enzymes,^such^as,^e.g.,^ papain^and^pepsin.^^Papain^cleaves^an^antibody^to^produce^two^Fab^fragments^and^a^single^Fc^ fragment.^^Pepsin^cleaves^an^antibody^to^produce^a^F(ab’)2^fragment^and^a^pFc’^fragment.^^As^ described^herein,^the^methods^of^determining^unpaired^glycan^content^comprises^a^two^enzyme^ digestion,^one^of^which^is^used^to^produce^a^mixture^of^Fab^fragments^and^Fc^fragments.^^^ [00120]^^^^ Advantageously,^the^methods^are^not^limited^to^the^antigen^specificity^of^the^antibody.^^ Accordingly,^the^antibody^has^any^binding^specificity^for^virtually^any^antigen.^^In^exemplary^aspects,^ the^antibody^binds^to^a^hormone,^growth^factor,^cytokine,^a^cell^surface^receptor,^or^any^ligand^ thereof.^^In^exemplary^aspects,^the^antibody^binds^to^a^protein^expressed^on^the^cell^surface^of^an^ immune^cell.^^In^exemplary^aspects,^the^antibody^binds^to^a^cluster^of^differentiation^molecule^ selected^from^the^group^consisting^of:^CD1a,^CD1b,^CD1c,^CD1d,^CD2,^CD3,^CD4,^CD5,^CD6,^CD7,^CD8,^ CD9,^CD10,^CD11A,^CD11B,^CD11C,^CDw12,^CD13,^CD14,^CD15,^CD15s,^CD16,^CDw17,^CD18,^CD19,^ CD20,^CD21,^CD22,^CD23,^CD24,^CD25,^CD26,^CD27,^CD28,^CD29,^CD30,^CD31,CD32,^CD33,^CD34,^ CD35,^CD36,^CD37,^CD38,^CD39,^CD40,^CD41,^CD42a,^CD42b,^CD42c,^CD42d,^CD43,^CD44,^CD45,^ CD45RO,^CD45RA,^CD45RB,^CD46,^CD47,^CD48,^CD49a,^CD49b,^CD49c,^CD49d,^CD49e,^CD49f,^CD50,^ CD51,^CD52,^CD53,^CD54,^CD55,^CD56,^CD57,^CD58,^CD59,^CDw60,^CD61,^CD62E,^CD62L,^CD62P,^ CD63,^CD64,^CD65,^CD66a,^CD66b,^CD66c,^CD66d,^CD66e,^CD66f,^CD68,^CD69,^CD70,^CD71,^CD72,^ ^
CD73,^CD74,^CD75,^CD76,^CD79ɲ,^CD79ɴ,^CD80,^CD81,^CD82,^CD83,^CDw84,^CD85,^CD86,^CD87,^ CD88,^CD89,^CD90,^CD91,^CDw92,^CD93,^CD94,^CD95,^CD96,^CD97,^CD98,^CD99,^CD100,^CD101,^ CD102,^CD103,^CD104,^CD105,^CD106,^CD107a,^CD107b,^CDw108,^CD109,^CD114,^CD^115,^CD116,^ CD117,^CD118,^CD119,^CD120a,^CD120b,^CD121a,^CDw121b,^CD122,^CD123,^CD124,^CD125,^CD126,^ CD127,^CDw128,^CD129,^CD130,^CDw131,^CD132,^CD134,^CD135,^CDw136,^CDw137,^CD138,^CD139,^ CD140a,^CD140b,^CD141,^CD142,^CD143,^CD144,^CD145,^CD146,^CD147,^CD148,^CD150,^CD151,^ CD152,^CD153,^CD154,^CD155,^CD156,^CD157,^CD158a,^CD158b,^CD161,^CD162,^CD163,^CD164,^ CD165,^CD166,^and^CD182.^^^ [00121]^^^^ In^exemplary^aspects,^the^antibody^is^one^of^those^described^in^U.S.^Patent^No.7947809^ and^U.S.^Patent^Application^Publication^No.^20090041784^(glucagon^receptor),^U.S.^Patent^No.^ 7939070,^U.S.^Patent^No.^7833527,^U.S.^Patent^No.^7767206,^and^U.S.^Patent^No.^7786284^(IL^17^ receptor^A),^U.S.^Patent^No.^7872106^and^U.S.^Patent^No.^7592429^(Sclerostin),^U.S.^Patent^No.^ 7871611,^U.S.^Patent^No.^7815907,^U.S.^Patent^No.^7037498,^U.S.^Patent^No.^7700742,^and^U.S.^ Patent^Application^Publication^No.^20100255538^(IGF^1^receptor),^U.S.^Patent^No.^7868140^(B7RP1),^ U.S.^Patent^No.^7807159^and^U.S.^Patent^Application^Publication^No.^20110091455^(myostatin),^U.S.^ Patent^No.^7736644,^U.S.^Patent^No.^7628986,^U.S.^Patent^No.^7524496,^and^U.S.^Patent^Application^ Publication^No.^20100111979^(deletion^mutants^of^epidermal^growth^factor^receptor),^U.S.^Patent^ No.^7728110^(SARS^coronavirus),^U.S.^Patent^No.^7718776^and^U.S.^Patent^Application^Publication^ No.^20100209435^(OPGL),^U.S.^Patent^No.^7658924^and^U.S.^Patent^No.^7521053^(Angiopoietin^2),^ U.S.^Patent^No.^7601818,^U.S.^Patent^No.^7795413,^U.S.^Patent^Application^Publication^No.^ 20090155274,^U.S.^Patent^Application^Publication^No.^20110040076^(NGF),^U.S.^Patent^No.^7579186^ (TGF^ɴ^type^II^receptor),^U.S.^Patent^No.^7541438^(connective^tissue^growth^factor),^U.S.^Patent^No.^ 7438910^(IL1^R1),^U.S.^Patent^No.^7423128^(properdin),^U.S.^Patent^No.^7411057,^U.S.^Patent^No.^ 7824679,^U.S.^Patent^No.^7109003,^U.S.^Patent^No.^6682736,^U.S.^Patent^No.^7132281,^and^U.S.^ Patent^No.^7807797^(CTLA^4),^U.S.^Patent^No.^7084257,^U.S.^Patent^No.^7790859,^U.S.^Patent^No.^ 7335743,^U.S.^Patent^No.^7084257,^and^U.S.^Patent^Application^Publication^No.^20110045537^ (interferon^gamma),^U.S.^Patent^No.^7932372^(MAdCAM),^U.S.^Patent^No.^7906625,^U.S.^Patent^ Application^Publication^No.^20080292639,^and^U.S.^Patent^Application^Publicaiton^No.^20110044986^ (amyloid),^U.S.^Patent^No.^7815907^and^U.S.^Patent^No.^7700742^(insulin^like^growth^factor^I),^U.S.^ Patent^No.^7566772^and^U.S.^Patent^No.^7964193^(interleukin^1ɴ),^U.S.^Patent^No.^7563442,^U.S.^ Patent^No.^7288251,^U.S.^Patent^No.^7338660,^U.S.^Patent^No.^7626012,^U.S.^Patent^No.^7618633,^ and^U.S.^Patent^Application^Publication^No.^20100098694^(CD40),^U.S.^Patent^No.^7498420^(c^Met),^ U.S.^Patent^No.^7326414,^U.S.^Patent^No.^7592430,^and^U.S.^Patent^No.^7728113^(M^CSF),^U.S.^Patent^ No.^6924360,^U.S.^Patent^No.^7067131,^and^U.S.^Patent^No.^7090844^(MUC18),^U.S.^Patent^No.^ ^
6235883,^U.S.^Patent^No.^7807798,^and^U.S.^Patent^Application^Publication^No.^20100305307^ (epidermal^growth^factor^receptor),^U.S.^Patent^No.^6716587,^U.S.^Patent^No.^7872113,^U.S.^Patent^ No.^7465450,^U.S.^Patent^No.^7186809,^U.S.^Patent^No.^7317090,^and^U.S.^Patent^No.^7638606^ (interleukin^4^receptor),^U.S.^Patent^Application^Publication^No.^20110135657^(BETA^KLOTHO),^U.S.^ Patent^No.^7887799^and^U.S.^Patent^No.^7879323^(fibroblast^growth^factor^like^polypeptides),^U.S.^ Patent^No.^7867494^(IgE),^U.S.^Patent^Application^Publication^No.^20100254975^(ALPHA^4^BETA^7),^ U.S.^Patent^Application^Publication^No.^20100197005^and^U.S.^Patent^No.^7537762^(ACTIVIN^ RECEPTOR^LIKE^KINASE^1),^U.S.^Patent^No.^7585500^and^U.S.^Patent^Application^Publication^No.^ 20100047253^(IL^13),^U.S.^Patent^Application^Publication^No.^20090263383^and^U.S.^Patent^No.^ 7449555^(CD148),^U.S.^Patent^Application^Publication^No.^20090234106^(ACTIVIN^A),^U.S.^Patent^ Application^Publication^No.^20090226447^(angiopoietin^1^and^angiopoietin^2),^U.S.^Patent^ Application^Publication^No.^20090191212^(Angiopoietin^2),^U.S.^Patent^Application^Publication^No.^ 20090155164^(C^FMS),^U.S.^Patent^No.^7537762^(activin^receptor^like^kinase^1),^U.S.^Patent^No.^ 7371381^(galanin),^U.S.^Patent^Application^Publication^No.^20070196376^(INSULIN^LIKE^GROWTH^ FACTORS),^U.S.^Patent^No.^7267960^and^U.S.^Patent^No.^7741115^(LDCAM),^US7265212^(CD45RB),^ U.S.^Patent^No.^7709611,^U.S.^Patent^Application^Publication^No.^20060127393^and^U.S.^Patent^ Application^Publication^No.^20100040619^(DKK1),^U.S.^Patent^No.^7807795,^U.S.^Patent^Application^ Publication^No.^20030103978^and^U.S.^Patent^No.^7923008^(osteoprotegerin),^U.S.^Patent^ Application^Publication^No.^20090208489^(OV064),^U.S.^Patent^Application^Publication^No.^ 20080286284^(PSMA),^U.S.^Patent^No.^7888482,^U.S.^Patent^Application^Publication^No.^ 20110165171,^and^U.S.^Patent^Application^Publication^No.^20110059063^(PAR2),^U.S.^Patent^ Application^Publication^No.^20110150888^(HEPCIDIN),^U.S.^Patent^No.^7939640^(B7L^1),^U.S.^Patent^ No.^7915391^(c^Kit),^U.S.^Patent^No.^7807796,^U.S.^Patent^No.^7193058,^and^U.S.^Patent^No.^7427669^ (ULBP),^U.S.^Patent^No.^7786271,^U.S.^Patent^No.^7304144,^and^U.S.^Patent^Application^Publication^ No.^20090238823^(TSLP),^U.S.^Patent^No.^7767793^(SIGIRR),^U.S.^Patent^No.^7705130^(HER^3),^U.S.^ Patent^No.^7704501^(ataxin^1^like^polypeptide),^U.S.^Patent^No.^7695948^and^U.S.^Patent^No.^ 7199224^(TNF^ɲ^converting^enzyme),^U.S.^Patent^Application^Publication^No.^20090234106^(ACTIVIN^ A),^U.S.^Patent^Application^Publication^No.^20090214559^and^U.S.^Patent^No.^7438910^(IL1^R1),^U.S.^ Patent^No.^7579186^(TGF^ɴ^type^II^receptor),^U.S.^Patent^No.^7569387^(TNF^receptor^like^molecules),^ U.S.^Patent^No.^7541438,^(connective^tissue^growth^factor),^U.S.^Patent^No.^7521048^(TRAIL^ receptor^2),^U.S.^Patent^No.^6319499,^U.S.^Patent^No.^7081523,^and^U.S.^Patent^Application^ Publication^No.^20080182976^(erythropoietin^receptor),^U.S.^Patent^Application^Publication^No.^ 20080166352^and^U.S.^Patent^No.^7435796^(B7RP1),^U.S.^Patent^No.^7423128^(properdin),^U.S.^ Patent^No.^7422742^and^U.S.^Patent^No.^7141653^(interleukin^5),^U.S.^Patent^No.^6740522^and^U.S.^ ^
Patent^No.^7411050^(RANKL),^U.S.^Patent^No.^7378091^(carbonic^anhydrase^IX^(CA^IX)^tumor^ antigen),^U.S.^Patent^No.^7318925and^U.S.^Patent^No.^7288253^(parathyroid^hormone),^U.S.^Patent^ No.^7285269^(TNF),^U.S.^Patent^No.^6692740^and^U.S.^Patent^No.^7270817^(ACPL),^U.S.^Patent^No.^ 7202343^(monocyte^chemo^attractant^protein^1),^U.S.^Patent^No.^7144731^(SCF),^U.S.^Patent^No.^ 6355779^and^U.S.^Patent^No.^7138500^(4^1BB),^U.S.^Patent^No.^7135174^(PDGFD),^U.S.^Patent^No.^ 6630143^and^U.S.^Patent^No.^7045128^(Flt^3^ligand),^U.S.^Patent^No.^6849450^(metalloproteinase^ inhibitor),^U.S.^Patent^No.^6596852^(LERK^5),^U.S.^Patent^No.^6232447^(LERK^6),^U.S.^Patent^No.^ 6500429^(brain^derived^neurotrophic^factor),^U.S.^Patent^No.^6184359^(epithelium^derived^T^cell^ factor),^U.S.^Patent^No.^6143874^(neurotrophic^factor^NNT^1),^U.S.^Patent^Application^Publication^ No.^20110027287^(PROPROTEIN^CONVERTASE^SUBTILISIN^KEXIN^TYPE^9^(PCSK9)),^U.S.^Patent^ Application^Publication^No.^20110014201^(IL^18^RECEPTOR),^and^U.S.^Patent^Application^Publication^ No.^20090155164^(C^FMS).^^The^above^patents^and^published^patent^applications^are^incorporated^ herein^by^reference^in^their^entirety^for^purposes^of^their^disclosure^of^variable^domain^ polypeptides,^variable^domain^encoding^nucleic^acids,^host^cells,^vectors,^methods^of^making^ polypeptides^encoding^said^variable^domains,^pharmaceutical^compositions,^and^methods^of^treating^ diseases^associated^with^the^respective^target^of^the^variable^domain^containing^antigen^binding^ protein^or^antibody.^ [00122]^^^^ In^exemplary^embodiments,^the^antibody^is^one^of^Muromonab^CD3^(product^marketed^ with^the^brand^name^Orthoclone^Okt3®),^Abciximab^(product^marketed^with^the^brand^name^ Reopro®.),^Rituximab^(product^marketed^with^the^brand^name^MabThera®,^Rituxan®),^Basiliximab^ (product^marketed^with^the^brand^name^Simulect®),^Daclizumab^(product^marketed^with^the^brand^ name^Zenapax®),^Palivizumab^(product^marketed^with^the^brand^name^Synagis®),^Infliximab^(product^ marketed^with^the^brand^name^Remicade®),^Trastuzumab^(product^marketed^with^the^brand^name^ Herceptin®),^Alemtuzumab^(product^marketed^with^the^brand^name^MabCampath®,^Campath^1H®),^ Adalimumab^(product^marketed^with^the^brand^name^Humira®),^Tositumomab^I131^(product^ marketed^with^the^brand^name^Bexxar®),^Efalizumab^(product^marketed^with^the^brand^name^ Raptiva®),^Cetuximab^(product^marketed^with^the^brand^name^Erbitux®),^Ibritumomab^tiuxetan^ (product^marketed^with^the^brand^name^Zevalin®),^Omalizumab^(product^marketed^with^the^brand^ name^Xolair®),^ocrelizumab^(product^marketed^with^the^brand^name^Ocrevus®),^Bevacizumab^ (product^marketed^with^the^brand^name^Avastin®),^Natalizumab^(product^marketed^with^the^brand^ name^Tysabri®),^Ranibizumab^(product^marketed^with^the^brand^name^Lucentis®),^Panitumumab^ (product^marketed^with^the^brand^name^Vectibix®),^Eculizumab^(product^marketed^with^the^brand^ name^Soliris®),^Certolizumab^pegol^(product^marketed^with^the^brand^name^Cimzia®),^Golimumab^ (product^marketed^with^the^brand^name^Simponi®),^Canakinumab^(product^marketed^with^the^brand^ ^
name^Ilaris®),^Catumaxomab^(product^marketed^with^the^brand^name^Removab®),^Ustekinumab^ (product^marketed^with^the^brand^name^Stelara®),^Tocilizumab^(product^marketed^with^the^brand^ name^RoActemra®,^Actemra®),^Ofatumumab^(product^marketed^with^the^brand^name^Arzerra®),^ Denosumab^(product^marketed^with^the^brand^name^Prolia®),^Belimumab^(product^marketed^with^ the^brand^name^Benlysta®),^Raxibacumab,^Ipilimumab^(product^marketed^with^the^brand^name^ Yervoy®),^and^Pertuzumab^(product^marketed^with^the^brand^name^Perjeta^antibodies,^such^as^ daclizumab.^^ [00123]^^^^ In^exemplary^aspects,^the^antibody^binds^to^a^tumor^associated^antigen^and^is^an^anti^ cancer^antibody.^Examples^of^suitable^anti^cancer^antibodies^include,^but^are^not^limited^to,^anti^ BAFF^antibodies^such^as^belimumab;^anti^CD20^antibodies^such^as^rituximab;^anti^CD22^antibodies^ such^as^epratuzumab;^anti^CD25^antibodies^such^as^daclizumab;^anti^CD30^antibodies^such^as^ iratumumab,^anti^CD33^antibodies^such^as^gemtuzumab,^anti^CD52^antibodies^such^as^ alemtuzumab;^anti^CD152^antibodies^such^as^ipilimumab;^anti^EGFR^antibodies^such^as^cetuximab;^ anti^HER2^antibodies^such^as^trastuzumab^and^pertuzumab;^anti^IL6^antibodies,^such^as^siltuximab;^ and^anti^VEGF^antibodies^such^as^bevacizumab;^anti^IL6^receptor^antibodies^such^as^tocilizumab.^^^ [00124]^^^^ Antibody^Compositions^ [00125]^^^^ The^presently^disclosed^methods^relate^to^antibody^compositions.^^In^various^aspects,^the^ composition^comprises^only^one^type^of^antibody.^^In^various^instances,^the^composition^comprises^ antibodies^wherein^each^antibody^of^the^composition^comprises^the^same^or^essentially^the^amino^ acid^sequence.^^In^various^aspects,^the^composition^comprises^antibodies^wherein^each^antibody^of^ the^composition^comprises^an^amino^acid^sequence^which^is^at^least^90%^identical^to^the^amino^acid^ sequences^of^all^other^antibodies^of^the^composition.^^In^various^aspects,^the^composition^comprises^ antibodies^wherein^each^antibody^of^the^composition^comprises^an^amino^acid^sequence^which^is^at^ least^95%,^at^least^96%,^at^least^97%,^at^least^98%,^or^at^least^99%^identical^to^the^amino^acid^ sequences^of^all^other^antibodies^of^the^composition.^^In^various^aspects,^the^composition^comprises^ antibodies^wherein^each^antibody^of^the^composition^comprises^an^amino^acid^sequence^which^is^ the^same^or^essentially^the^same^(e.g.,^at^least^90%^or^at^least^95%,^at^least^96%,^at^least^97%,^at^ least^98%,^or^at^least^99%^identical^to^the^amino^acid^sequences^of^all^other^antibodies^of^the^ composition)^but^the^glycan^pair^classifications^of^the^antibodies^of^the^composition^may^differ^from^ each^other.^^ [00126]^^^^ In^various^aspects,^the^composition^comprises^only^one^type^of^antibody.^^In^various^ instances,^the^composition^comprises^antibodies^wherein^each^antibody^of^the^antibody^composition^ comprises^the^same^or^essentially^the^amino^acid^sequence.^^In^various^aspects,^the^antibody^ ^
composition^comprises^antibodies^wherein^each^antibody^of^the^antibody^composition^comprises^an^ amino^acid^sequence^which^is^at^least^90%^identical^to^the^amino^acid^sequences^of^all^other^ antibodies^of^the^antibody^composition.^^In^various^aspects,^the^antibody^composition^comprises^ antibodies^wherein^each^antibody^of^the^antibody^composition^comprises^an^amino^acid^sequence^ which^is^at^least^95%,^at^least^96%,^at^least^97%,^at^least^98%,^or^at^least^99%^identical^to^the^amino^ acid^sequences^of^all^other^antibodies^of^the^antibody^composition.^^In^various^aspects,^the^antibody^ composition^comprises^antibodies^wherein^each^antibody^of^the^antibody^composition^comprises^an^ amino^acid^sequence^which^is^the^same^or^essentially^the^same^(e.g.,^at^least^90%^or^at^least^95%,^at^ least^96%,^at^least^97%,^at^least^98%,^or^at^least^99%^identical^to^the^amino^acid^sequences^of^all^ other^antibodies^of^the^antibody^composition)^but^the^glycan^pair^classifications^of^the^antibodies^of^ the^antibody^composition^may^differ^from^each^other.^^In^exemplary^aspects,^the^antibody^ composition^comprises^a^heterogeneous^mixture^of^antibodies^having^different^glycan^pair^ classifications.^^In^various^instances,^the^antibody^composition^may^be^characterized^in^terms^of^its^ unpaired^AF^glycan^content,^unpaired^HM^glycan^content,^paired^AF^glycan^content,^and/or^its^paired^ HM^glycan^content.^^In^various^instances,^the^antibody^composition^may^be^characterized^in^terms^of^ its^relative^abundance^of^unpaired^AF^glycans^and/or^relative^abundance^of^unpaired^HM^glycans.^^^ [00127]^^^^ The^following^examples^are^given^merely^to^illustrate^the^present^invention^and^not^in^any^ way^to^limit^its^scope.^ EXAMPLES^ EXAMPLE^1^ [00128]^^^^ This^example^describes^an^exemplary^simplified^method^of^determining^paired^glycan^ content^and^unpaired^glycan^content^for^an^antibody^composition.^^ [00129]^^^^ In^a^previous^study,^the^paired^afucosylated^and^high^mannose^glycan^content^and^ unpaired^afucosylated^and^high^mannose^glycan^content^were^determined^for^two^different^ antibody^compositions,^and^statistical^relationships^correlating^unpaired^glycan^content^of^the^ antibody^composition^with^ADCC^activity^and^FcɶRIIIa^binding^were^established.^^In^this^previous^ study,^the^method^used^to^measure^the^levels^of^unpaired^or^paired^glycopairs^comprised^three^ steps:^^an^antibody^cleavage^step,^a^chromatographic^separation^step,^and^a^mass^spectrometry^ (MS)^based^detection^step.^^Briefly,^samples^of^an^antibody^composition^comprising^Antibody^A^or^ Antibody^B^were^treated^with^the^IgdE^(FabALACTICA®^or^FL^from^Genovis^Inc.,^Cambridge,^MA)^ enzyme^which^cleaves^the^heavy^chain^of^antibodies^above^the^hinge^to^create^Fab^and^Fc^antibody^ fragments.^^The^FL^enzyme^cleaves^between^the^second^and^third^amino^acid^of^the^IgG1^heavy^chain^ sequence^KTHTCPP^(SEQ^ID^NO:^1).^^The^resulting^Fab^fragments^and^glycosylated^Fc^fragments^were^ ^
subsequently^separated^and^characterized^by^hydrophilic^interaction^liquid^chromatography^(HILIC)^ with^water/organic^with^ion^pairing^reagent^as^solvents^and^MS^based^detection.^^The^HILIC^ separation^was^carried^out^on^a^Waters^Acquity^UPLC^Glycoprotein^Amide^column^over^a^course^of^ 95^min^with^mobile^phase^composition^20:80^(v/v)^of^water^and^acetonitrile^with^0.1%^TFA.^At^1^min,^ a^linear^gradient^of^0.44^ml/min^was^applied^for^the^next^74^min^for^separating^glycopairs^based^on^ their^interaction^with^the^stationary^phase.^MS^detection^was^carried^out^using^an^Agilent^6545XT^ QToF^MS^with^an^Agilent^Jet^Stream^(AJS)^electrospray^ionization^source^(ESI)^with^settings^of:^ capillary^voltage^^^4500^V;^drying^gas^^^11^mL/min;^nebulizer^pressure^^^25^psi^and^gas^temperature^^^ 340^°C^using^a^mass^range^of^1000^3000^m/z.^Deconvolution^was^performed^using^Bioconfirm^ B.09.00^(Agilent)^using^S/N^of^30^and^output^mass^range^set^to^40^60^kDa.^^^ [00130]^^^^ Though^the^above^method^successfully^measured^the^content^of^unpaired^and^paired^ afucosylated^and^high^mannose^glycans^to^establish^statistically^significant^correlations^with^ biological^functions,^a^subsequent^study^was^conducted^to^improve^the^efficiency^of^the^method^for^ determining^the^levels^of^unpaired^and^paired^afucosylated^and^high^mannose^glycans.^Glycan^ pairing^analysis^is^complicated^due^to^the^high^degree^of^heterogeneity^of^the^glycopair^population,^ and^the^HILIC^separation^aims^to^reduce^the^complexity^through^the^introduction^of^a^high^ resolution^separation^prior^to^MS^analysis.^The^glycopairs^are^separated^into^a^glycopairing^map^of^ approximately^15^chromatographic^peaks^(Figure^6A).^However,^the^HILIC^MS^glycan^pairing^analysis^ remains^complicated^for^several^reasons.^First,^the^paired^fucosylated^(Fc,^A2G0F/A2G0F,^Fc,^ A2G0F/A2G1F^and^Fc,^A2G1F/A2G1F)^glycopairs,^which^do^not^significantly^impact^ADCC,^are^the^ dominant^species^in^the^analysis,^eluting^across^the^entire^chromatographic^region^of^the^ glycosylated^Fc^fragment^chromatogram^(Figure^6A^6B).^Second,^many^of^the^important^ chromatographic^peaks^are^poorly^resolved:^the^key^afucosylated^and^high^mannose^glycopairs^co^ elute^with^and/or^poorly^separate^from^the^dominant^paired^fucoslyated^glycopairs.^As^a^result,^it^is^ difficult^to^establish^the^correct^window^to^extract^the^mass^spectra^for^each^chromatographic^peak,^ and^a^flawed^extraction^window^can^artificially^inflate^or^deflate^the^peak^areas^of^the^key^ afucosylated^and^high^mannose^glycopairs^reducing^the^accuracy^of^the^analysis.^Third,^structural^ isomers^and^glycopairs^with^partially^reduced^disulfide^bonds^(Rd,^+2Da)^lead^to^multiple^ chromatographic^peaks^for^species^categorized^as^the^same^glycopair.^Finally,^key^afucosylated^and^ high^mannose^glycopairs^(shown^in^bold^in^Figure^6B)^have^extremely^low^abundances,^and^often^ represent^less^than^5%^of^the^population.^^Taken^together,^these^challenges^complicate^the^data^ analysis^and^quantitation^and^require^the^careful^consideration^of^a^highly^skilled^analyst,^and^ ultimately^increase^the^time^required^for^these^overall^method.^^^ ^
[00131]^^^^ A^new^method^was^designed^to^include^the^preparation,^separation,^measurement,^and^ analysis^of^a^simplified^sample^comprising^the^components^essential^for^quantitation^of^afucosylated^ and^high^mannose^glycans^(paired^or^unpaired)^without^an^abundance^of^non^essential^components.^ For^example,^in^the^previous^method,^every^type^of^glycopair^including^the^high^mannose,^and^all^ fucosylated^and^afucosylated^glycopairs^were^identified^and^quantified,^and^this^degree^of^resolution^ was^unnecessary^for^the^purposes^of^determining^levels^of^unpaired^and^paired^afucosylated^and^ high^mannose^glycopairs.^^Further,^the^separation^prior^to^the^MS^based^detection^was^designed^so^ the^glycosylated^Fc^fragments^eluted^under^one^chromatographic^peak^in^the^total^ion^ chromatogram^which^was^chromatographically^separated^from^the^Fab^region.^In^the^previous^HILIC^ MS^method,^poor^chromatographic^resolution^of^key^afucosylated^and^high^mannose^glycopairs^from^ the^abundant^fucosylated^glycopairs^lead^to^difficulties^determining^the^correct^mass^spectral^ extraction^windows^for^accurate^quantitation.^To^simplify^analysis^in^the^new^method^all^ glycosylated^Fc^fragments^intentionally^co^elute^under^one^chromatographic^peak^in^the^total^ion^ chromatogram.^The^mass^spectra^are^extracted^from^the^entire^chromatographic^peak^of^the^Fc^ fragment^and^deconvoluted.^The^deconvoluted^mass^spectral^peaks^in^the^one^deconvoluted^mass^ spectrum^are^leveraged^for^identification^and^quantitation^of^the^glycopairs.^Using^this^approach,^the^ Fc^fragments^are^separated^by^their^differences^(delta)^in^molecular^weights^(MW).^By^intentionally^ removing^the^chromatographic^separation^of^the^glycosylated^Fc^fragments^the^data^analysis^is^ simplified,^and^subsequently^quantitative^accuracy^and^sample^to^sample^comparability^is^improved.^^ Additionally,^the^sample^through^put^is^dramatically^increased.^An^automated^identification^system^ for^faster^identification^and^quantitation^of^the^paired^and^unpaired^afucosylated^and^high^mannose^ glycopairs^was^additionally^included^in^the^new^method.^^ [00132]^^^^ The^new^method^comprised^a^two^enzyme^digestion^of^the^antibody^composition^before^ chromatographic^separation^and^a^MS^based^detection.^This^two^enzyme^digestion^is^depicted^in^ Figure^5.^Briefly,^samples^of^the^antibody^composition^were^treated^with^the^enzyme^IgdE^ (FabALACTICA®^or^FL^from^Genovis^Inc.,^Cambridge,^MA)^which^cleaves^the^heavy^chain^of^IgG1^ antibodies^above^the^hinge^to^create^Fab^and^Fc^antibody^fragments.^The^FL^enzyme^cleaves^ between^the^second^and^third^amino^acid^of^the^IgG1^heavy^chain^sequence^KTHTCPP^(SEQ^ID^NO:^ 1).^The^digestion^was^carried^out^overnight^at^37^°C.^^A^second^digestion^with^EndoS^(IgGZERO®^from^ Genovis^Inc.,),^an^IgG^specific^endoglycosidase^S^enzyme,^was^carried^out^for^30^min^at^37°C.^EndoS^is^ an^IgG^specific^endoglycosidase^acting^on^complex^type^N^glycans^at^the^Fc^glycosylation^site^of^IgGs.^ The^enzyme^hydrolyzes^the^ɴ1,4^linkage^between^the^core^GlcNAc^residues^in^the^N^glycans,^leaving^ the^innermost^GlcNAc^intact^on^the^Fc^with^or^without^a^fucose.^^EndoS^has^limited^activity^on^high^ ^
mannose^and^hybrid^glycans,^this^is^leveraged^to^identify^and^quantify^the^high^mannose^glycopairs^ and^subsequently^the^%^relative^abundance^of^paired^and^unpaired^high^mannose.^ [00133]^^^^ This^two^enzyme^digestion^yielded^a^simplified^population^of^glycopairs^which^could^be^ categorized^into^one^of^two^afucosylated^glycopair^classes:^^(1)^paired^afucosylated^(both^heavy^ chains^of^the^Fc^fragment^were^afucosylated)^and^(2)^unpaired^afucosylated^(only^one^heavy^chain^of^ the^Fc^fragment^was^afucosylated,^while^the^other^heavy^chain^was^fucosylated)^(Figure^7).^^Based^on^ the^assumption^that^both^afucosylated^and^high^mannose^glycans^are^highly^potent,^pairs^containing^ a^combination^of^high^mannose^and^afucosylated^glycans^were^classed^as^afucosylated^paired,^ unpaired^high^mannose,^as^appropriate^and^according^to^Table^A.^^^ [00134]^^^^ ^EndoS^has^limited^activity^on^high^mannose^and^hybrid^glycans,^this^was^leveraged^to^ identify^high^mannose^glycopairs^and^quantify^the^high^mannose^glycan^pairing.^The^population^of^ individual^Fc^high^mannose^glycopairs^could^be^categorized^into^one^of^two^high^mannose^classes:^ (1)^paired^high^mannose^(both^heavy^chains^of^the^Fc^fragment^contained^high^mannose^glycans)^and^ (2)^unpaired^high^mannose^(only^one^heavy^chain^of^the^Fc^fragment^contained^high^mannose,^while^ the^other^heavy^chain^was^fucosylated^or^afucosylated)^(Figure^7).^^A^summary^designation^was^given^ according^to^Table^A^and^following^the^three^rules^described^herein.^ [00135]^^^^ Following^the^two^enzyme^digestion,^resulting^Fc^and^Fab^fragments^were^ chromatographically^separated^by^using^a^rapid^reversed^phase^liquid^chromatography^(RP^LC)^ separation^with^MS^detection.^This^separation^yielded^one^chromatographic^peak^comprising^the^Fc^ fragments^that^was^well^seperated^from^the^Fab^fragments^and^treatment^enzymes.^A^diagram^of^ the^chromatographic^separation^and^MS^analysis^steps^are^shown^in^Figures^8A^8B.^^^ [00136]^^^^ The^equipment^used^to^perform^steps^of^the^method^has^the^following^capabilities:^

^ [00137]^^^^ The^autosampler^temperature^was^held^at^4^C^and^an^aliquot^was^injected^and^the^ separation^was^carried^out^using^a^2.1^ID^x^50^mm^RP^analytical^column^at^60^°C.^Mobile^Phase^A^was^ ^
0.1%^(v/v)^trifluoroacetic^acid^in^water^while^Mobile^Phase^B^was^0.1%^(v/v)^trifluoroacetic^acid^in^ acetonitrile.^The^mobile^phase^gradient^example^is^provided^below:^

^ [00138]^^^^ ^MS^acquisition^was^performed^using^an^Agilent^6545^XT^Q^TOF^with^an^AJS^ESI^source^in^ positive^ion^mode.^The^source^conditions^were:^gas^temperature^–^325^°C,^drying^and^sheath^gas^ flow^–^12^L/min,^sheath^gas^temperature^–^300^°C,^nebulizer^–^40^psi,^capillary^voltage^–^5000^V,^ fragmentor^voltage^–^280^V,^and^skimmer^–^120^V.^Spectra^were^acquired^with^a^m/z^range^of^1200^ 3200.^^^ [00139]^^^^ The^chromatographic^peak^that^contained^the^Fc^fragments,^was^then^selected^for^data^ analysis,^the^mass^spectra^of^the^one^chromatographic^peak^that^contained^the^Fc^fragments^was^ extracted^and^deconvoluted^(Figures^8A^8B).^The^deconvolution^was^performed^using^Byos^(Protein^ Metrics^Inc,^Cupertino,^CA)^the^input^m/z^range^was^1300^3200^m/z^while^the^output^range^was^30^ 70^kDa.^The^intensity^of^each^deconvoluted^mass^spectral^peak^of^the^antibody^composition^was^ employed^for^quantitation.^ [00140]^^^^ The^EndoS^treated^glycopairs^were^identified^by^matching^the^molecular^weights^(MW)^of^ the^deconvoluted^mass^spectral^peaks^of^the^antibody^composition^against^a^custom^built^database^ of^theoretical^MWs^(Table^1).^The^glycan^MWs^used^in^this^approach^is^the^glycan^MW^associated^ with^the^N^linkage^(native^glycan^MW^–^H
2O).^The^database^comprised^of^the^theoretical^MWs^of^ two^EndoS^treated^glycans^(glycan^pairs)^whereby^complex^glycans^were^hydrolyzed^to^either^GlcNac^ or^GlucNac^+^Fuc^while^high^mannose^and^hybrid^glycans^remained^intact.^The^database^comprised^ of^the^theoretical^MWs^of^approximately^40^individual^EndoS^glycan^pairs^classed^as^(1)^afucosylated^ paired^and^(2)^afucosylated^unpaired^and^(3)^high^mannose^paired^and^(4)^high^mannose^unpaired^ (Table^1).^Glycan^pairs^that^have^been^hydroylzed^by^EndoS^are^italicized^in^the^subsequent^text.^ ^
[00141]^^^^ To^facilitate^the^automated^identification^of^the^EndoS^treated^glycopairs^(defined^as^the^ intact^Fc^and^two^glycan^moieties)^the^amino^acid^sequence^of^the^FL^treated^Fc^was^first^loaded^into^ the^protein^analysis^software^to^generate^a^theoretical^MW^of^the^Fc,^additionally^the^software^was^ instructed^to^consider^that^all^possible^disulfide^bonds^remain^intact^and^both^C^terminal^lysines^ were^clipped.^Second,^the^database^of^the^theoretical^MWs^of^EndoS^glycan^pairs^was^loaded^into^ the^protein^analysis^software^as^a^list^of^custom^modifications.^Following^this^user^input,^the^protein^ analysis^software^then^automatically^determined^a^list^of^theoretical^MWs^for^the^EndoS^glycopairs^ (seen^in^Table^1).^The^software^automatically^identified^the^deconvoluted^mass^spectral^peaks^if^the^ MW^matched^with^a^theoretical^MW^in^the^custom^built^database^of^EndoS^glycopairs.^ ^ TABLE^1^

^
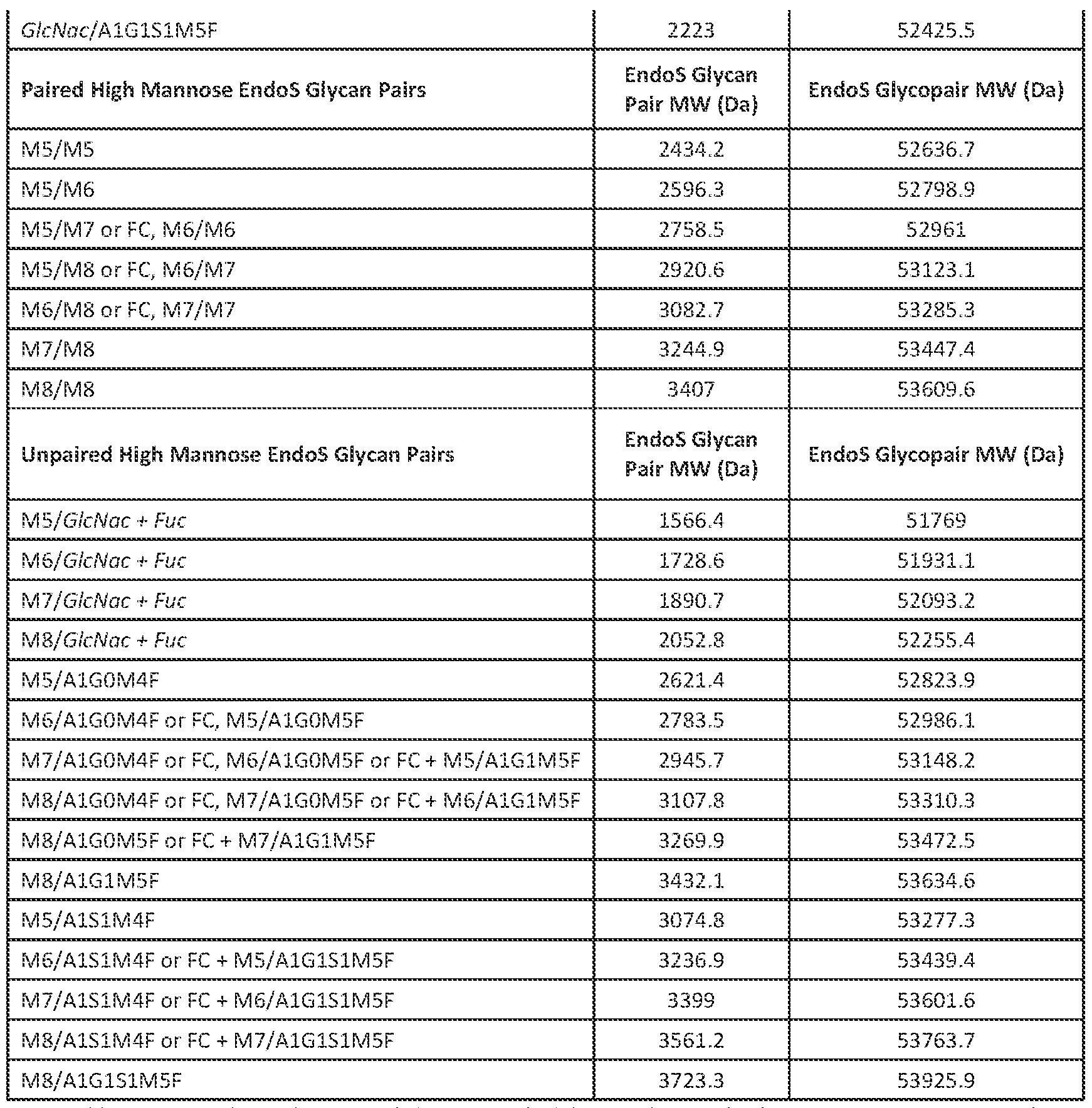
Note:^Additional^MWs^for^EndoS^treated^glycopairs^which^have^only^a^single^glycoslyation^or^a^bisecting^GlcNac^ are^included^in^the^database^but^not^listed^above.^^ ^ [00142]^^^^ The^most^abundant^EndoS^glycopair^is^Fc,^GlucNac^+^Fucose/GlucNac^+^Fucose^which^ contains^a^fucosylated^glycan^on^each^heavy^chain^and^is^therefore^not^appliciable^to^the^ determination^of^afucoslyated^or^high^mannose^glycan^pairing.^In^the^previous^study^using^HILIC^MS,^ the^highly^abundant^and^not^applicable^paired^fucosylated^glycopairs^elute^across^the^entire^region^ of^the^chromatogram^where^the^glycosylated^Fc^elutes^often^masking^the^presence^of^the^species^of^ interest^(Figure^6B).^In^the^current^study,^the^EndoS^treatment^simplifies^the^multitude^of^highly^ abundant^paired^fucoslyated^glycopairs^into^a^single^species,^Fc,^GlucNac^+^Fucose/GlucNac^+^Fucose.^ This^single^deconvoluted^mass^peak^is^well^seperated,^by^the^delta^molecular^weight,^from^the^ species^of^interest^which^faciliates^streamlined^data^interpretation^(Figure^8B).^^ ^
[00143]^^^^ The^abundance^of^the^individual^EndoS^glycopairs^was^determined^from^the^^relative^ intensities^or^areas^of^the^identified^deconvoluted^mass^spectral^peaks^as^follows:^ %^Glycopair^Intensity^=^Deconvoluted^Peak^Intensity/Total^Deconvoluted^Peak^Intensity^×^100^ [Equation^1]^ [00144]^^^^ The^abundence^of^paired^afucosyated^glycopairs^was^determined^by^summing^the^ individual^glycopairs^categorized^as^paired^afucosylated^glycopairs,^similarly^the^abundence^of^ unpaired^afucosyated^glycopairs^was^determined^by^summing^the^individual^glycopairs^categorized^ as^unpaired^afucosylated^glycopairs.^To^calculate^the^%^relative^abundence^of^the^Unpaired^ Afucosylated^Glycan^Content^of^the^antibody^composition^Equation^2^was^employed:^ %^Unpaired^Afucosylated^Glycan^Content^=^(%^Unpaired^Afucoslyated^Glycopairs/%^Total^Afucoslyated^ Glycopairs^(Unpaired+Paired))^x^100^ [Equation^2]^ [00145]^^^^ The^abundence^of^paired^high^mannose^glycopairs^was^determined^by^summing^the^ individual^glycopairs^categorized^as^paired^high^mannose^glycopairs,^similarly^the^abundence^of^ unpaired^high^mannose^glycopairs^was^determined^by^summing^the^individual^glycopairs^categorized^ as^unpaired^high^mannose^glycopairs.^To^calculate^the^%^relative^abundence^of^the^Unpaired^High^ Mannose^Glycan^content^of^the^antibody^composition^Equation^3^was^employed:^ %^Unpaired^High^Mannose^Glycan^Content^^=^(%^Unpaired^High^Mannose^Glycopairs/Total^High^Mannose^ Glycopairs^(Unpaired+Paired))^x^100^ [Equation^3]^ [00146]^^^^ In^the^previous^study,^a^95^min^HILIC^MS^analysis^was^employed^to^separate^a^highly^ complex^population^of^glycopairs^for^the^determination^of^the^levels^of^afucoslyated^and^high^ mannose^glycan^pairing.^This^approach^remains^challanging^as^the^highly^abundant^and^not^ applicable^paired^fucosylated^glycopairs^elute^across^the^entire^glycoslyated^Fc^chromatographic^ region^often^masking^the^presence^of^the^glycopairs^of^interest.^Key^high^mannose^and^afucosylated^ glycopairs^co^elute^with^and/or^elute^as^sholders^of^the^highly^abundant^paired^fucosylated^ glycopairs^making^accurate^quantitation^challanging.^This^example^demonstrates^the^feasibility^of^a^ streamlined^method^where^the^glycopair^population^is^simplified^prior^to^analysis^with^EndoS.^A^ rapid^17^min^chromatographic^seperation^prior^to^MS^detection^simplifes^analysis^by^yeilding^one,^ well^resolved,^chromatographic^peak^of^Fc^fragments^for^further^quantitation.^The^quantitation^of^ the^EndoS^glycopairs^is^achieved^using^the^deconvoluted^mass^spectral^peaks^where^the^highly^ abundent^paired^fucosylated^species^is^a^single^deconvoluted^mass^spectral^peak^well^seperared,^by^ MW,^from^the^key^afucosylated^and^high^mannose^glycopairs.^The^custom^built^EndoS^glycan^pair^ ^
MW^database^facilates^automated^indentification^and^quantation^of^the^critical^afucosylated^and^ high^mannose^glycopairs.^This^new^method^affords^a^rapid^analysis^and^increased^quantiative^ accuracy^when^determining^%^relative^abundance^of^the^unpaired^and^paired^afucosylated^glycan^ content^and^%^relative^abundance^of^the^unpaired^and^paired^high^mannose^glycan^content^of^an^ antibody^composition.^^^^ EXAMPLE^2^ [00147]^^^^ This^example^describes^an^exemplary^application^of^the^simplified^method^of^determining^ the^%^relative^paired^and^unpaired^afucosylated^and^high^mannose^glycan^content^for^an^antibody^ composition.^^ [00148]^^^^ The^new^method^described^in^Example^1^was^carried^out^on^a^panel^of^antibody^ composition^samples,^wherein^individual^samples^of^the^panel^comprised^an^IgG1^monoclonal^ antibody^produced^by^a^particular^clone^or^a^sample^of^a^reference^product^(RP)^lot.^^Deconvoluted^ mass^spectra^for^selected^samples^are^shown^in^Figures^9A^9C.^^Data^for^paired^afucosylated^ glycopairs^and^unpaired^afucosylated^glycopairs^are^provided^in^Tables^2A^and^2B,^respectively.^^Data^ for^paired^high^mannose^glycopairs^and^unpaired^high^mannose^glycopairs^are^provided^in^Tables^2C^ and^2D,^respectively.^^^ TABLE^2A^

^
*Note:^Only^the^paired^afucosylated^glycopairs^detected^in^1^or^more^samples^are^listed.^ND^=^Not^detected^for^ sample.^Italicized^glycans^have^been^hydrolyzed^by^EndoS^ TABLE^2B^
*Note:^Only^the^paired^afucosylated^glycopairs^detected^in^1^or^more^samples^are^listed.^ND^=^Not^detected^for^ sample.^Italicized^glycans^have^been^hydrolyzed^by^EndoS^ TABLE^2C^
*Note:^Only^the^paired^afucosylated^glycopairs^detected^in^1^or^more^samples^are^listed.^ND^=^Not^detected^for^ sample.^Italicized^glycans^have^been^hydrolyzed^by^EndoS^ TABLE^2D^
*Note:^Only^the^paired^afucosylated^glycopairs^detected^in^1^or^more^samples^are^listed.^ND^=^Not^detected^for^ sample.^Italicized^glycans^have^been^hydrolyzed^by^EndoS^ ^ [00149]^^^^ The^relative^unpaired^and^paired^afucosylated^glycan^content^and^the^relative^unpaired^ and^paired^high^mannose^glycan^content^were^calculated^as^described^in^Example^1^and^the^data^is^ provided^below^in^Table^3.^^^ TABLE^3^


^ [00150]^^^^ Based^on^these^data,^the^RP^range^of^unpaired^afucosylated^glycan^content^was^85.4^–^ 88.4^and^the^range^of^unpaired^high^mannose^glycan^content^was^67.8^–^81.2.^^The^clones^having^the^ best^unpaired^glycan^profiles^are^Clones^203,^116,^113,^126^and^236,^with^clones^116^and^126^having^ an^unpaired^afucosylated^glycan^content^within^the^RP^range^and^clone^203^having^both^unpaired^ afucosylated^glycan^content^and^unpaired^high^mannose^glycan^within^the^RP^range.^^^ EXAMPLE^3^ [00151]^^^^ This^example^describes^an^exemplary^method^for^measuring^ADCC^activity^levels.^^ [00152]^^^^ ADCC^activity^levels^(expressed^as^a^%^relative^value)^for^a^panel^of^samples^of^an^IgG1^ monoclonal^antibody^composition^are^determined^using^a^quantitative^cell^based^assay^that^ measures^the^ability^of^the^antibody^to^mediate^cell^cytotoxicity^in^a^dose^dependent^manner^of^ target^cells^stably^expressing^antigens^while^engaging^FcɶRIIIA^(158V)^receptors^on^NK92^M1^effector^ cells^via^the^antibody^Fc^domain.^^These^events^lead^to^the^activation^of^the^effector^cells^and^ destruction^of^the^target^cells^via^exocytosis^of^the^cytolytic^granule^complex^perforin/granzyme.^^A^ schematic^of^the^ADCC^assay^is^provided^in^Figure^10.^^Briefly,^target^cells^are^labeled^with^calcein^ acetoxymethyl^(calcein^AM),^which^readily^enters^the^cells^and^is^subsequently^cleaved^by^ intercellular^esterases^and^trapped^within^the^cells.^When^target^cells^are^lysed,^fluorescent^calcein^is^ released^into^the^medium.^^The^level^of^calcein^released^from^lysed^target^cells^is^determined^by^ measuring^the^fluorescence^of^the^reaction^supernatant^in^an^Envision^(Perkin^Elmer)^fluorescence^ plate^reader.^^Each^assay^is^performed^in^triplicate^and^the^mean^and^standard^deviation^are^ recorded.^^The^data^are^fitted^to^the^mean^fluorescence^values^using^a^constrained^4^parameter^fit^ ^
using^SoftMaxPro^software^and^reported^as^percentage^ADCC^activity^relative^to^a^reference^ standard^as^calculated^by^the^EC50^standard/EC50^sample^ratio.^^ EXAMPLE^4^ [00153]^^^^ HILIC^is^a^quantitative^analysis^of^the^N^linked^glycan^distribution^of^the^antibody^and^ comprises^three^steps:^(1)^release^and^label^N^linked^glycans^from^reference^and^test^samples^using^ PNGase^F^and^a^fluorophore^that^can^specifically^derivatize^free^glycan,^(2)^load^samples^within^the^ validated^linear^range^onto^a^HILIC^column,^the^labeled^N^linked^glycans^are^separated^using^a^ gradient^of^decreasing^organic^solvent,^and^(3)^monitor^elution^of^glycan^species^with^fluorescence^ detector.^^^ [00154]^^^^ The^standard^and^test^samples^are^prepared^by^carrying^out^the^following^steps:^(1)^dilute^ samples^and^controls^with^water,^(2)^add^PNGase^F^and^incubate^the^samples^and^controls^to^release^ N^linked^glycans,^(3)^mix^with^fluorophore^labeling^solution^using^a^fluorophore^such^as^2^ aminobenzoic^acid.^Vortex^and^incubate^the^samples^and^controls,^(4)^centrifuge^down^to^pellet^ protein^and^remove^supernatant,^and^(5)^dry^and^reconstitute^labeled^glycans^in^the^injection^ solution.^^^ [00155]^^^^ The^solutions^used^in^this^assay^are^a^Mobile^Phase^A^(100^mM^ammonium^formate,^ target^pH^3.0)^and^a^Mobile^Phase^B^(acetonitrile).^^The^equipment^used^to^perform^steps^of^the^ method^has^the^following^capabilities:^ ^ ^ ^ ^
^

^ [00156]^^^^ The^instrument^settings^for^HPLC^using^a^hydrophilic^interaction^analytical^BEH^Glycan^1.7^ ʅm^column^(2.1^mm^ID^X^150^mm)^and^2^aminobenzoic^acid^fluorophore^labeling^method^are^ provided^below:^
^ [00157]^^^^ The^mobile^phase^gradient^example^is^provided^below:^
^ ^ ^ ^
^ [00158]^^^^ Reports^of^the^results^comprise^the^following^format:^
*Calculation^formulas^depend^on^presence^of^individual^high^mannose^and^afucosylated^ glycans^ EXAMPLE^5^ [00159]^^^^ This^example^describes^the^relationships^of^ADCC^activity^to^%^released^high^mannose^ glycans,^%^released^afucosylated^glycans,^%^unpaired^high^mannose^glycans^and^%^unpaired^ afucosylated^glycans^for^an^antibody^composition.^ [00160]^^^^ The^ADCC^activity^level^of^a^panel^of^antibody^compositions^was^determined^by^a^cell^ based^ADCC^assay.^^The^%^released^high^mannose^glycans^and^%^released^afucosylated^glycans^were^ determined^by^the^method^described^in^Example^4.^The^ADCC^activity^levels,^%^released^high^ mannose^glycans,^and^%^released^afucosylated^glycans^of^each^antibody^composition^of^the^panel^ are^provided^in^Table^4.^^^ TABLE^4^

^ ^
[00161]^^^^ These^data^were^analyzed^using^the^JMP^suite^of^computer^programs^for^statistical^ analysis^(SAS^Institute,^Cary,^NC).^^Figure^11A^is^an^ADCC^leverage^plot^for^%^released^afucosylated^ glycans^and^Figure^11B^is^a^leverage^plot^for^%^released^high^mannose^glycans.^^The^best^fit^line^is^the^ solid^diagonal^line^in^the^middle^of^the^shaded^area.^^As^shown^in^Figures^11A^and^11B,^released^ afucosylated^glycans^demonstrated^a^statistically^significant^relationship^with^the^ADCC^activity^level^ of^the^antibody^composition^(p<0.0001),^whereas^released^high^mannose^glycans^did^not^(p=0.2786).^^^^ [00162]^^^^ The^relationship^between^%^ADCC^and^the^%^released^afucosylated^glycans^and^%^ released^high^mannose^glycans^may^be^described^by^Equation^4:^ Predicted^%^ADCC^=^^5.8^+^(21.7*^%^Released^Afucosylated^Glycans)^+^(4.3^*^%^Released^High^Mannose^ Glycans)^ [Equation^4]^ [00163]^^^^ Plugging^the^measured^values^for^%^released^afucosylated^glycans^and^%^released^high^ mannose^glycans^into^Equation^4,^a^predicted^%^ADCC^value^was^calculated^for^each^sample.^^The^ actual^%^ADCC^(as^measured^in^the^cell^based^assay)^was^plotted^against^the^predicted^%^ADCC^(as^ calculated^by^Equation^4)^and^the^plot^is^provided^as^Figure^11C.^^Statistical^parameters,^including^ Root^Mean^Square^Error^(RMSE),^r
2,^and^p^value,^are^shown^in^Figure^11C.^^These^results^suggested^ that^Equation^4^predicted^the^actual^(measured)^ADCC^with^accuracy^and^underlines^the^statistically^ significant^correlation^between^%^released^afucosylated^glycans^and^ADCC^(p<0.0001).^^Higher^levels^ of^afucosylated^glycans^result^in^higher^ADCC^activity.^^The^effect^of^high^mannose^on^ADCC^was^ weak^and^not^statistically^significant.^ [00164]^^^^ As^the^glycans^are^released^from^the^Fc^of^the^antibody^composition^in^the^approach^ outlined^in^Example^4^no^glycan^pairing^information^is^captured^and^the^leverage^of^both^the^ unpaired^and^paired^afucosylated^glycans^and^the^unpaired^and^paired^high^mannose^glycans^on^ ADCC^activity^cannot^be^determined.^In^the^previous^HILIC^MS^method^outlined^in^Example^1,^an^ increased^level^of^unpaired^afucosylated^glycans^was^shown^to^have^an^increased^leverage^on^ADCC,^ likely^because^the^percentage^of^afucosylated^glycans^is^spread^over^a^wider^distribution^of^ molecules^of^the^antibody^composition.^Paired^high^mannose^glycans^has^been^shown^to^have^a^very^ low^leverage^on^ADCC^activity,^however^the^structural^reason^for^this^is^currently^unknown.^ [00165]^^^^ ^The^RP^LC^MS^approach^outlined^in^Example^1^to^measure^the^levels^of^paired^and^ unpaired^afucosylated^and^high^mannose^glycan^content^offers^a^powerful,^but^simplified^approach^ to^determine^the^relationship^between^ADCC^activity^and^glycan^pairing.^The^overall^distribution^of^ total^afucosylated^and^total^high^mannose^glycans^determined^by^the^approaches^described^in^ Example^1^and^Example^4^should^provide^similar^results.^However,^the^RP^LC^MS^method^was^ ^
developed^as^a^targeted^workflow^focused^on^the^analysis^of^the^unpaired^afucosylated^and^high^ mannose^glycan^content,^the^full^composition^data^are^likely^less^accurate^compared^to^the^well^ established^released^glycan^assay^described^in^Example^4.^The^optimal^use^of^the^glycan^pairing^data^ on^the^afucosylated^and^high^mannose^subgroups^is^in^application^in^a^form^of^a^correction^factor^to^ the^released^afucosylated^and^high^mannose^glycan^content^determined^using^Example^4^to^account^ for^the^contribution^of^glycan^pairing.^This^correction^can^be^used^to^determine^the^relationship^ between^ADCC^activity^and^unpaired^afucosylated^glycans^and^unpaired^high^mannose^glycans^for^an^ antibody^composition.^^ [00166]^^^^ The^paired^and^unpaired^afucosylated^glycan^content^and^paired^and^unpaired^high^ mannose^glycan^content^of^the^same^antibody^composition^were^determined^following^the^ procedure^outlined^in^Example^1.^^The^results^are^provided^in^Table^3.^^ [00167]^^^^ The^fraction^of^paired^afucosylated^glycans^was^used^to^apply^a^correction^to^the^released^ afucosylated^glycan^data^in^Table^4^to^account^for^the^portion^of^the^antibody^composition^where^ the^afucosylated^glycans^are^paired^(paired^afucosylated^fraction).^Using^Equation^5,^the^%^Unpaired^ Afucosylated^Glycans^of^the^entire^antibody^composition^can^be^determined.^^ %^Unpaired^Afucosylated^Glycans^^=^%^released^afucosylated^glycans^(1^^paired^afucosylated^fraction)/(1+^ paired^afucosylated^fraction)^x^100^ [Equation^5]^ [00168]^^^^ The^relative^paired^high^mannose^glycan^content^was^used^to^apply^a^correction^to^the^ released^high^mannose^glycan^data^in^Table^4^to^account^for^the^portion^of^the^antibody^composition^ where^the^high^mannose^glycans^are^paired^(paired^high^mannose^fraction).^Using^Equation^6,^the^%^ Unpaired^High^Mannose^Glycans^of^the^entire^antibody^composition^can^be^determined.^^ %^Unpaired^High^Mannose^Glycans^^=^%^released^high^mannose^glycans^(1^^paired^high^mannose^fraction)/(1+^ paired^high^mannose^fraction)^x^100^ [Equation^6]^ [00169]^^^^ The^ADCC^activity^levels,^the^%^Unpaired^Afucosylated^Glycans^and^the^%^Unpaired^High^ Mannose^Glycans^were^calculated^as^described^in^Equation^5^and^Equation^6^and^the^data^is^ provided^below^in^Table^5.^ TABLE^5^

^
^ [00170]^^^^ The^data^in^Table^5^was^then^analyzed^using^the^JMP^suite^of^computer^programs^for^ statistical^analysis.^^Figure^12A^is^an^ADCC^leverage^plot^for^%^unpaired^afucosylated^glycans^and^ Figure^12B^is^a^leverage^plot^for^%^unpaired^high^mannose^glycans.^^The^best^fit^line^is^the^solid^ diagonal^line^in^the^middle^of^the^shaded^area.^^As^shown^in^Figures^12A^and^12B,^both^%^unpaired^ afucosylated^glycans^and^%^unpaired^high^mannose^glycans^demonstrated^a^statistically^significant^ relationship^with^ADCC^activity^levels^(p<0.0001^for^%^unpaired^afucosylated^glycans^and^p=0.0012^ for^%^unpaired^high^mannose^glycans).^^These^results^support^that^the^%^unpaired^high^mannose^ glycans^do^significantly^contribute^to^ADCC^activity.^^^ [00171]^^^^ Taken^together,^these^data^support^that^the^pairing^status^matters.^^The^overall^%^ released^high^mannose^glycan^content^did^not^appear^to^have^a^statistically^significant^relationship^ with^the^ADCC^activity^level,^whereas^the^%^unpaired^high^mannose^glycan^content^was^ demonstrated^to^correlate^with^ADCC^activity^statistically^significantly.^ [00172]^^^^ The^relationship^between^%^ADCC^and^the^%^unpaired^afucosylated^glycans^and^%^ unpaired^high^mannose^glycans^may^be^described^by^Equation^7:^ Predicted^%^ADCC^=^^27.5^+^(29.6*^%^Unpaired^Afucosylated^Glycans)^+^(14.5^*^%^Unpaired^High^Mannose^ Glycans)^ [Equation^7]^ [00173]^^^^ Plugging^the^measured^values^for^%^unpaired^high^mannose^glycans^and^%^unpaired^ afucosylated^glycans^into^Equation^7,^a^predicted^%^ADCC^value^was^calculated^for^each^sample.^^The^ actual^%^ADCC^(as^measured^in^the^cell^based^assay)^was^plotted^against^the^predicted^%^ADCC^(as^ ^
calculated^by^Equation^7)^and^the^plot^is^provided^as^Figure^12C.^^Statistical^parameters,^including^ Root^Mean^Square^Error^(RMSE),^r
2,^and^p^value,^are^shown^in^Figure^12C.^^^ [00174]^^^^ These^results^suggested^that^Equation^7^predicted^the^actual^(measured)^ADCC^with^ accuracy^and^underlines^the^statistically^significant^direct^correlation^between^%^unpaired^ afucosylated^glycans,^%^unpaired^high^mannose^glycans,^and^ADCC^activity^level^(p<0.0001).^^Higher^ levels^of^unpaired^afucosylated^glycan^content^and^unpaired^high^mannoses^content^result^in^higher^ ADCC^activity.^^The^leverage^of^unpaired^afucosylated^glycans^was^stronger^than^the^leverage^of^ unpaired^high^mannose^glycans^on^ADCC^activity^(29.6^and^14.5,^respectively).^^Determining^ unpaired^afucosylated^glycans^and^unpaired^high^mannose^glycans^can^predict^ADCC^levels^of^an^ antibody^composition^and^thus^predict^product^quality,^e.g.,^quality^of^the^antibody^composition.^ [00175]^^^^ A^similar^approach^can^be^used^to^correct^the^released^afucosylated^and^high^mannose^ glycan^content^to^determine^the^paired^afucosylated^and^paired^high^mannose^glycan^content^ respectively.^For^the^current^antibody^composition,^as^the^abundance^of^the^paired^glycan^species^ are^much^lower,^as^seen^in^Table^3,^a^lower^impact^to^ADCC^activity^is^expected.^Nonetheless,^this^ approach^can^be^employed^to^further^understand^the^relationship^between^the^paired^glycan^ content^and^ADCC^activity.^It^is^expected^that^paired^afucosylated^glycans^will^have^a^statistically^ significant^relationship^with^ADCC.^As^discussed^above^it^is^expected^that^the^paired^high^mannose^ glycan^content^will^have^a^very^low^leverage^on^ADCC^activity.^ [00176]^^^^ These^data^support^that^glycan^pairing^analysis^is^important^for^understanding^the^ structure^function^relationship.^ ^ ^ ^
EXAMPLE^6^ [00177]^^^^ This^example^describes^a^method^of^determining^the^product^quality^during^manufacture^ of^an^antibody^composition.^^^ [00178]^^^^ Samples^of^a^cell^culture^comprising^Clone^203^producing^an^antibody^are^collected^at^ different^pre^harvest^times^post^inoculation^and^post^harvest^prior^to^Protein^A^chromatography.^^ The^new^method^described^in^Example^1^is^carried^out^on^the^obtained^samples.^^Data^for^paired^ high^mannose^glycan^content^unpaired^high^mannose^glycan^pair^content,^paired^afucosylated^ glycan^content^and^unpaired^afucosylated^glycan^pair^content^for^each^sample^are^recorded.^^The^ range^of^unpaired^high^mannose^glycan^content^and^unpaired^afucosylated^glycan^content^is^pre^ determined.^^The^samples^that^are^deemed^to^be^outside^this^range^are^marked,^and^trouble^ shooting^on^the^corresponding^manufacture^days^are^monitored.^^It^is^discovered^that^a^cell^culture^ parameter^shifts^on^this^day^and^efforts^to^prevent^the^shift^are^taken.^^The^process^of^collecting^ samples^at^different^times^pre^harvest^and^post^harvest^as^well^as^determining^the^unpaired^high^ mannose^glycan^content^and^unpaired^afucosylated^glycan^content^of^the^samples^are^repeated.^^ The^samples^are^deemed^as^within^the^pre^determined^range.^ EXAMPLE^7^ [00179]^^^^ This^example^describes^a^method^of^determining^the^product^quality^of^different^ manufacture^lots^of^an^antibody^composition.^^^ [00180]^^^^ Samples^of^Lots^1^10^of^an^IgG1^monoclonal^antibody^are^obtained^following^storage^for^ 12,^24^and^36^months^at^4^°C.^^The^new^method^described^in^Example^1^is^carried^out^on^the^ obtained^samples.^^Data^for^paired^high^mannose^glycan^species,^unpaired^high^mannose^glycan^pair^ content,^paired^afucosylated^glycan^content^and^unpaired^afucosylated^glycan^pair^content^for^each^ sample^are^recorded.^^Using^Equation^7^and^the^data^for^%^unpaired^high^mannose^glycan^molecules^ and^%^unpaired^afucosylated^glycan^molecule,^predicted^ADCC^activity^levels^for^each^lot^are^ determined.^^Lots^that^are^within^the^pre^determined^ADCC^range^are^determined^as^acceptable^for^ administration^to^a^patient,^whereas^lots^that^are^outside^the^range^are^discarded.^^^ EXAMPLE^8^ [00181]^^^^ This^example^describes^a^simplified^method^of^determining^paired^glycan^content^and^ unpaired^glycan^content^for^a^large^scale^preparation^of^the^IgG^described^in^previous^Examples^1^7.^^ [00182]^^^^ Three^batches^of^an^IgG^were^made^in^large^scale^bioreactors^and^samples^from^each^ batch^were^obtained^and^used^in^the^two^enzyme^digestion^as^essentially^described^in^Example^1.^^ Briefly,^the^samples^were^treated^overnight^with^the^enzyme^IgdE^(FabALACTICA®^or^FL^from^Genovis^ ^
Inc.,^Cambridge,^MA),^and^a^second^digestion^was^carried^out^with^EndoS^(IgGZERO®^from^Genovis^ Inc.,)^for^30^min.^^Following^the^two^enzyme^digestion,^resulting^Fc^and^Fab^fragments^were^ chromatographically^separated^by^using^a^rapid^reversed^phase^liquid^chromatography^(RP^LC)^ separation^with^MS^detection,^as^essentially^described^in^Example^1.^^This^separation^yielded^one^ chromatographic^peak^comprising^the^Fc^fragments^that^was^well^seperated^from^the^Fab^fragments^ and^treatment^enzymes.^^The^chromatographic^peak^that^contained^the^Fc^fragments,^was^then^ selected^for^data^analysis,^the^mass^spectra^of^the^one^chromatographic^peak^that^contained^the^Fc^ fragments^was^extracted^and^deconvoluted,^as^essentially^described^in^Example^1.^^The^EndoS^ treated^glycopairs^were^identified^by^matching^the^molecular^weights^(MW)^of^the^deconvoluted^ mass^spectral^peaks^of^the^antibody^composition^against^a^custom^built^database^of^theoretical^ MWs^(Table^1),^as^essentially^described^in^Example^1.^^The^data^for^unpaired^afucosylated^glycopairs,^ paired^afucosylated^glycopairs,^unpaired^high^mannose^glycopairs^and^paired^afucosylated^glycopairs^ are^provided^in^Table^6.^^^^ TABLE^6^
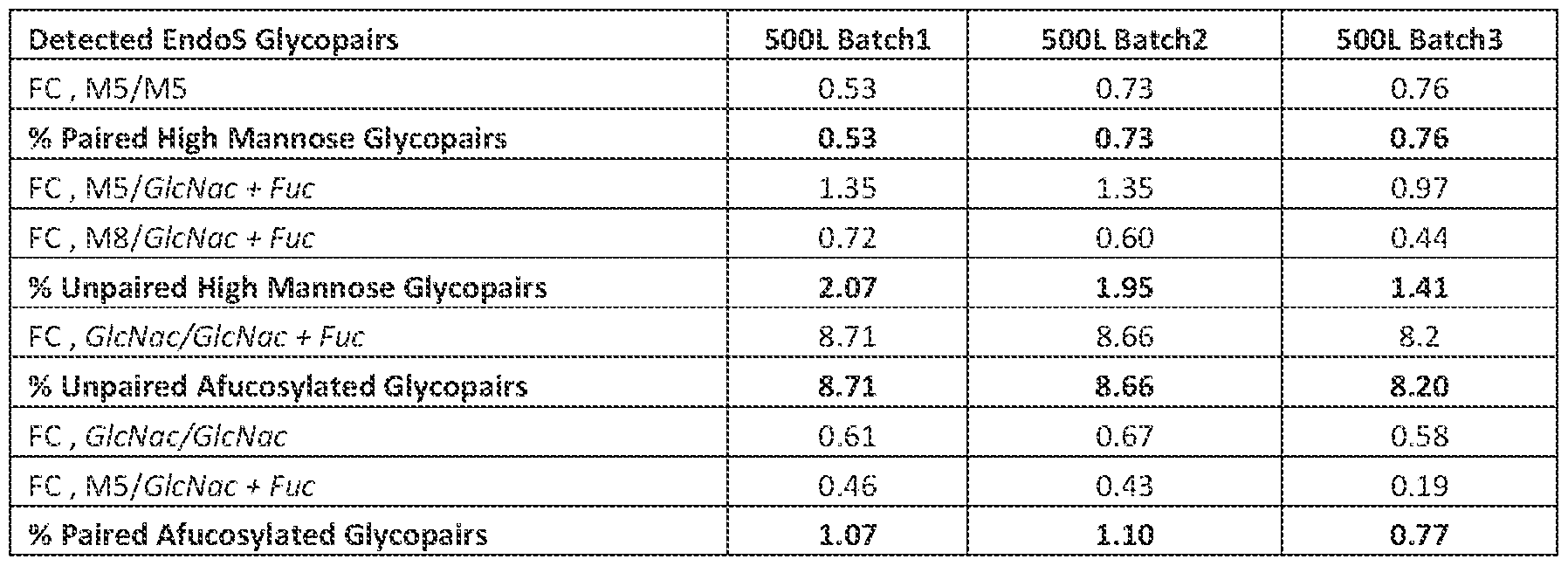
[00183]^^^^ The^relative^unpaired^and^paired^afucosylated^glycan^content^and^the^relative^unpaired^ and^paired^high^mannose^glycan^content^were^calculated^as^described^in^Example^1^and^the^data^is^ provided^below^in^Table^7.^^^ ^ ^ ^
TABLE^7^ ^

^ [00184]^^^^ This^example^demonstrated^that^the^new,^simplified^method^of^determining^the^relative^ unpaired^glycan^content^can^be^carried^out^with^a^large^scale^preparation^of^an^IgG^antibody^ composition.^^^ ^ EXAMPLE^9^ [00185]^^^^ This^example^describes^a^simplified^method^of^determining^paired^glycan^content^and^ unpaired^glycan^content^for^an^antibody^composition^comprising^an^IgG^different^from^the^IgG^ described^in^Examples^1^8.^^The^IgG^of^this^example^targeted^an^antigen^different^from^the^target^of^ the^IgG^of^Examples^1^8.^ [00186]^^^^ Samples^of^an^antibody^composition^comprising^an^IgG^different^from^the^IgG^described^in^ Examples^1^8^were^used^in^a^two^enzyme^digestion^as^essentially^described^in^Example^1.^^Briefly,^ the^samples^were^treated^overnight^with^the^enzyme^IgdE^(FabALACTICA®^or^FL^from^Genovis^Inc.,^ Cambridge,^MA),^and^a^second^digestion^was^carried^out^with^EndoS^(IgGZERO®^from^Genovis^Inc.,)^ for^30^min.^^Following^the^two^enzyme^digestion,^resulting^Fc^and^Fab^fragments^were^ chromatographically^separated^by^using^a^rapid^reversed^phase^liquid^chromatography^(RP^LC)^ separation^with^MS^detection,^as^essentially^described^in^Example^1.^^This^separation^yielded^one^ chromatographic^peak^comprising^the^Fc^fragments^that^was^well^seperated^from^the^Fab^fragments^ and^treatment^enzymes.^^The^chromatographic^peak^that^contained^the^Fc^fragments,^was^then^ selected^for^data^analysis,^the^mass^spectra^of^the^one^chromatographic^peak^that^contained^the^Fc^ fragments^was^extracted^and^deconvoluted,^as^essentially^described^in^Example^1.^^The^EndoS^ treated^glycopairs^were^identified^by^matching^the^molecular^weights^(MW)^of^the^deconvoluted^ mass^spectral^peaks^of^the^antibody^composition^against^a^custom^built^database^of^theoretical^ MWs^(Table^1),^as^essentially^described^in^Example^1.^^The^data^for^unpaired^afucosylated^glycopairs,^ paired^afucosylated^glycopairs,^unpaired^high^mannose^glycopairs^and^paired^afucosylated^glycopairs^ are^provided^in^Table^8.^^^^ TABLE^8^ ^

[00187]^^^^ The^relative^unpaired^and^paired^afucosylated^glycan^content^and^the^relative^unpaired^ and^paired^high^mannose^glycan^content^were^calculated^as^described^in^Example^1^and^the^data^is^ provided^below^in^Table^9.^^ TABLE^9^

^ [00188]^^^^ This^example^demonstrated^that^the^new,^simplified^method^of^determining^the^relative^ unpaired^glycan^content^can^be^used^with^IgG^antibody^compositions^other^than^the^ones^described^ in^Examples^1^8.^^^^ EXAMPLE^10^ [00189]^^^^ This^example^describes^a^simplified^method^of^determining^paired^glycan^content^and^ unpaired^glycan^content^for^an^antibody^composition^comprising^an^IgG^of^the^same^isotype^and^ antigen^specificity^as^the^IgG^of^Examples^1^7^but^different^allotype^(e.g.,^comprising^a^modified^Fc^ region^sequence,^relative^to^the^IgG^of^Examples^1^7).^^^ [00190]^^^^ Samples^of^an^antibody^composition^comprising^an^IgG^of^a^different^allotype^(relative^to^ the^IgG^described^in^Examples^1^7)^were^used^in^a^two^enzyme^digestion^as^essentially^described^in^ Example^1.^^Briefly,^the^samples^were^treated^overnight^with^the^enzyme^IgdE^(FabALACTICA®^or^FL^ from^Genovis^Inc.,^Cambridge,^MA),^and^a^second^digestion^was^carried^out^with^EndoS^(IgGZERO®^ from^Genovis^Inc.,)^for^30^min.^^Following^the^two^enzyme^digestion,^resulting^Fc^and^Fab^fragments^ were^chromatographically^separated^by^using^a^rapid^reversed^phase^liquid^chromatography^(RP^LC)^ separation^with^MS^detection,^as^essentially^described^in^Example^1.^^This^separation^yielded^one^ ^
chromatographic^peak^comprising^the^Fc^fragments^that^was^well^seperated^from^the^Fab^fragments^ and^treatment^enzymes.^^The^chromatographic^peak^that^contained^the^Fc^fragments,^was^then^ selected^for^data^analysis,^the^mass^spectra^of^the^one^chromatographic^peak^that^contained^the^Fc^ fragments^was^extracted^and^deconvoluted,^as^essentially^described^in^Example^1.^^The^EndoS^ treated^glycopairs^were^identified^by^matching^the^molecular^weights^(MW)^of^the^deconvoluted^ mass^spectral^peaks^of^the^antibody^composition^against^a^custom^built^database^of^theoretical^ MWs,^as^essentially^described^in^Example^1,^but^the^custom^built^database^used^in^this^example^is^ described^in^Table^10.^^The^data^for^unpaired^afucosylated^glycopairs,^paired^afucosylated^glycopairs,^ unpaired^high^mannose^glycopairs^and^paired^afucosylated^glycopairs^are^provided^in^Table^11.^^^^ TABLE^10^

^
^ ^ ^ ^
TABLE^11^
^ [00191]^^^^ The^relative^unpaired^and^paired^afucosylated^glycan^content^and^the^relative^unpaired^ and^paired^high^mannose^glycan^content^were^calculated^as^described^in^Example^1^and^the^data^is^ provided^below^in^Table^12.^ TABLE^12^

^ EXAMPLE^11^ [00192]^^^^ This^example^describes^a^simplified^method^of^determining^paired^glycan^content^and^ unpaired^glycan^content^for^^a^panel^of^antibody^composition^samples,^wherein^each^individual^ sample^of^the^panel^comprised^an^IgG1^monoclonal^antibody^produced^by^a^particular^clone^or^was^a^ sample^of^a^reference^product^(RP)^lot.^ [00193]^^^^ Samples^of^the^panel^were^used^in^a^two^enzyme^digestion^as^essentially^described^in^ Example^1.^^Briefly,^the^samples^were^treated^overnight^with^the^enzyme^IgdE^(FabALACTICA®^or^FL^ from^Genovis^Inc.,^Cambridge,^MA),^and^a^second^digestion^was^carried^out^with^EndoS^(IgGZERO®^ from^Genovis^Inc.,)^for^30^min.^^Following^the^two^enzyme^digestion,^resulting^Fc^and^Fab^fragments^ were^chromatographically^separated^by^using^a^rapid^reversed^phase^liquid^chromatography^(RP^LC)^ separation^with^MS^detection,^as^essentially^described^in^Example^1.^^This^separation^yielded^one^ chromatographic^peak^comprising^the^Fc^fragments^that^was^well^separated^from^the^Fab^fragments^ and^treatment^enzymes.^^The^chromatographic^peak^that^contained^the^Fc^fragments,^was^then^ ^
selected^for^data^analysis,^the^mass^spectra^of^the^one^chromatographic^peak^that^contained^the^Fc^ fragments^was^extracted^and^deconvoluted,^as^essentially^described^in^Example^1.^^The^EndoS^ treated^glycopairs^were^identified^by^matching^the^molecular^weights^(MW)^of^the^deconvoluted^ mass^spectral^peaks^of^the^antibody^composition^against^a^custom^built^database^of^theoretical^ MWs^(Table^10),^as^essentially^described^in^Example^1.^^The^data^for^unpaired^afucosylated^ glycopairs,^paired^afucosylated^glycopairs,^unpaired^high^mannose^glycopairs^and^paired^ afucosylated^glycopairs^are^provided^in^Table^13.^^^^ TABLE^13^

[00194]^^^^ The^relative^unpaired^and^paired^afucosylated^glycan^content^and^the^relative^unpaired^ and^paired^high^mannose^glycan^content^were^calculated^as^described^in^Example^1^and^the^data^is^ provided^below^in^Table^14.^ ^ ^ ^
TABLE^14^ ^
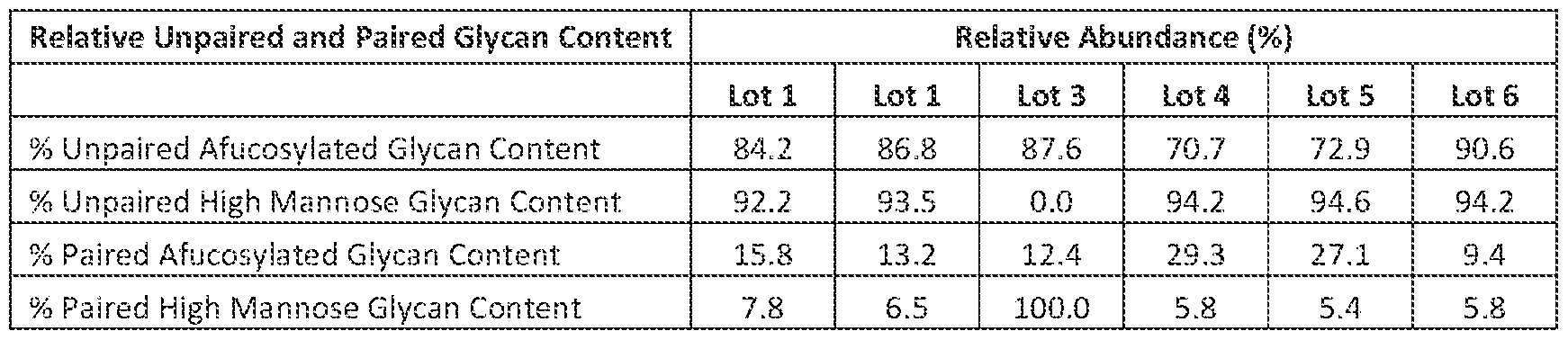
^ EXAMPLE^12^ [00195]^^^^ This^example^describes^a^simplified^method^of^determining^paired^high^mannose^glycan^ content^and^unpaired^high^mannose^glycan^content^for^an^antibody^composition^comprising^a^ completely^afucosylated^IgG.^^^ [00196]^^^^ Samples^of^an^antibody^composition^comprising^completely^afucosylated^IgG^were^used^in^ a^two^enzyme^digestion^as^essentially^described^in^Example^1.^^Briefly,^the^samples^were^treated^ overnight^with^the^enzyme^IgdE^(FabALACTICA®^or^FL^from^Genovis^Inc.,^Cambridge,^MA),^and^a^ second^digestion^was^carried^out^with^EndoS^(IgGZERO®^from^Genovis^Inc.,)^for^30^min.^^Following^ the^two^enzyme^digestion,^resulting^Fc^and^Fab^fragments^were^chromatographically^separated^by^ using^a^rapid^reversed^phase^liquid^chromatography^(RP^LC)^separation^with^MS^detection,^as^ essentially^described^in^Example^1.^^This^separation^yielded^one^chromatographic^peak^comprising^ the^Fc^fragments^that^was^well^seperated^from^the^Fab^fragments^and^treatment^enzymes.^^The^ chromatographic^peak^that^contained^the^Fc^fragments,^was^then^selected^for^data^analysis,^the^ mass^spectra^of^the^one^chromatographic^peak^that^contained^the^Fc^fragments^was^extracted^and^ deconvoluted,^as^essentially^described^in^Example^1.^^The^EndoS^treated^glycopairs^were^identified^ by^matching^the^molecular^weights^(MW)^of^the^deconvoluted^mass^spectral^peaks^of^the^antibody^ composition^against^a^custom^built^database^of^theoretical^MWs^(Table^15),^as^essentially^described^ in^Example^1.^^The^data^for^unpaired^high^mannose^glycopairs^and^paired^afucosylated^glycopairs^are^ provided^in^Table^16.^^^^ ^ ^ ^
TABLE^15^ ^

^ TABLE^16^
[00197]^^^^ The^relative^unpaired^and^paired^high^mannose^glycan^content^were^calculated^as^ described^in^Example^1^and^the^data^is^provided^below^in^Table^17.^ ^ ^ ^
TABLE^17^

^ ^ ^ [00198]^^^^ All^references,^including^publications,^patent^applications,^and^patents,^cited^herein^are^ hereby^incorporated^by^reference^to^the^same^extent^as^if^each^reference^were^individually^and^ specifically^indicated^to^be^incorporated^by^reference^and^were^set^forth^in^its^entirety^herein.^ [00199]^^^^ The^use^of^the^terms^“a”^and^“an”^and^“the”^and^similar^referents^in^the^context^of^ describing^the^disclosure^(especially^in^the^context^of^the^following^claims)^are^to^be^construed^to^ cover^both^the^singular^and^the^plural,^unless^otherwise^indicated^herein^or^clearly^contradicted^by^ context.^The^terms^“comprising,”^“having,”^“including,”^and^“containing”^are^to^be^construed^as^ open^ended^terms^including^the^indicated^component(s)^but^not^excluding^other^elements^(i.e.,^ meaning^“including,^but^not^limited^to,”)^unless^otherwise^noted.^^ [00200]^^^^ Recitation^of^ranges^of^values^herein^are^merely^intended^to^serve^as^a^shorthand^method^ of^referring^individually^to^each^separate^value^falling^within^the^range^and^each^endpoint,^unless^ otherwise^indicated^herein,^and^each^separate^value^and^endpoint^is^incorporated^into^the^ specification^as^if^it^were^individually^recited^herein.^^ [00201]^^^^ All^methods^described^herein^can^be^performed^in^any^suitable^order^unless^otherwise^ indicated^herein^or^otherwise^clearly^contradicted^by^context.^The^use^of^any^and^all^examples,^or^ exemplary^language^(e.g.,^“such^as”)^provided^herein,^is^intended^merely^to^better^illuminate^the^ disclosure^and^does^not^pose^a^limitation^on^the^scope^of^the^disclosure^unless^otherwise^claimed.^ No^language^in^the^specification^should^be^construed^as^indicating^any^non^claimed^element^as^ essential^to^the^practice^of^the^disclosure.^ [00202]^^^^ Preferred^embodiments^of^this^disclosure^are^described^herein,^including^the^best^mode^ known^to^the^inventors^for^carrying^out^the^disclosure.^^Variations^of^those^preferred^embodiments^ may^become^apparent^to^those^of^ordinary^skill^in^the^art^upon^reading^the^foregoing^description.^ The^inventors^expect^skilled^artisans^to^employ^such^variations^as^appropriate,^and^the^inventors^ intend^for^the^disclosure^to^be^practiced^otherwise^than^as^specifically^described^herein.^^ Accordingly,^this^disclosure^includes^all^modifications^and^equivalents^of^the^subject^matter^recited^ in^the^claims^appended^hereto^as^permitted^by^applicable^law.^^Moreover,^any^combination^of^the^ ^
above^described^elements^in^all^possible^variations^thereof^is^encompassed^by^the^disclosure^unless^ otherwise^indicated^herein^or^otherwise^clearly^contradicted^by^context.^ ^ ^ ^





































