WO2024213656A1 - Imidazo[1,2-a]pyrazine derivatives - Google Patents
Imidazo[1,2-a]pyrazine derivatives Download PDFInfo
- Publication number
- WO2024213656A1 WO2024213656A1 PCT/EP2024/059875 EP2024059875W WO2024213656A1 WO 2024213656 A1 WO2024213656 A1 WO 2024213656A1 EP 2024059875 W EP2024059875 W EP 2024059875W WO 2024213656 A1 WO2024213656 A1 WO 2024213656A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- 6alkyl
- 6alkoxy
- formula
- halogen
- compounds
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/90—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having two or more relevant hetero rings, condensed among themselves or with a common carbocyclic ring system
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/08—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having one or more single bonds to nitrogen atoms
- A01N47/10—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof
- A01N47/18—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof containing a —O—CO—N< group, or a thio analogue thereof, directly attached to a heterocyclic or cycloaliphatic ring
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01P—BIOCIDAL, PEST REPELLANT, PEST ATTRACTANT OR PLANT GROWTH REGULATORY ACTIVITY OF CHEMICAL COMPOUNDS OR PREPARATIONS
- A01P3/00—Fungicides
Definitions
- Imidazo[1,2-a]pyrazine derivatives The present invention relates to microbiocidal imidazo[1,2-a]pyrazine derivatives, e.g. as active ingredients, which have microbiocidal activity, in particular fungicidal activity, more particularly activity against oomycetes.
- the invention also relates to preparation of these imidazo[1,2-a]pyrazine derivatives, to intermediates useful in the preparation of these imidazo[1,2-a]pyrazine derivatives, to the preparation of these intermediates, to agrochemical compositions which comprise at least one of the imidazo[1,2-a]pyrazine derivatives, to preparation of these compositions and to the use of the imidazo[1,2-a]pyrazine derivatives or compositions in agriculture or horticulture for combating, controlling or preventing infestation of plants, harvested food crops, seeds or non-living materials by phytopathogenic microorganisms, in particular fungi, more particularly oomycetes.
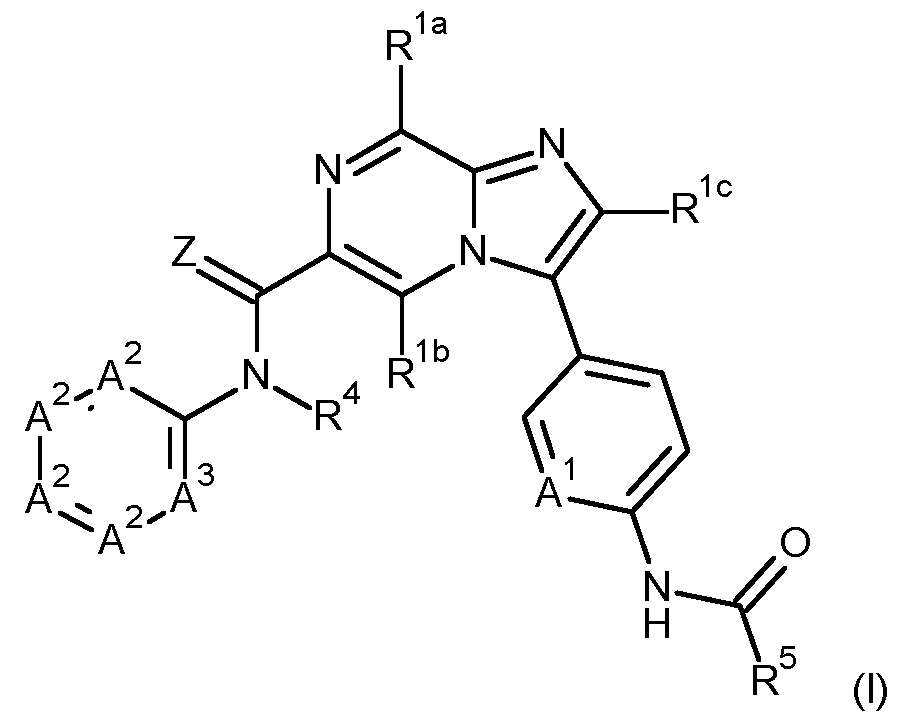
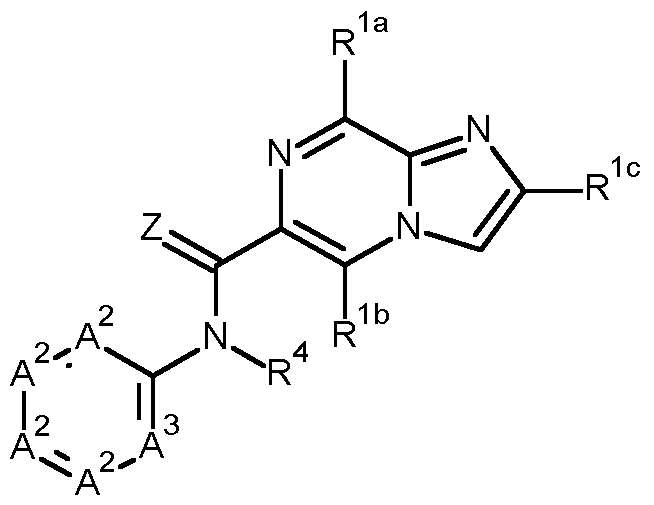
- the present invention provides compounds of formula (I) wherein Z is O or S, and preferably Z is O;
- a 1 is CH or N, and preferably N;
- R 1a is selected from hydroxy, halogen, CN, C1-6alkyl, C3-6cycloalkyl, C1-6alkoxy-C1-6alkyl, C3-6cycloalkyl- C1-4alkyl, C1-6alkylsulfanyl, C1-6alkylsulfinyl, C1-6alkylsulfonyl, C1-6alkoxy, amino, and NHC(O)C1-6alkyl;
- R 1b and R 1c are independently selected from hydrogen, hydroxy, halogen, CN, C1-6alkyl, C3-6cycloalkyl, C1-6alkoxy-C1-6alkyl, C3-6cycloalkyl
- the present invention provides an agrochemical composition comprising a compound of formula (I), and more particularly an agrochemical composition comprising a fungicidally effective amount of a compound of formula (I).
- Said composition can further comprise at least one compound selected among an additional active ingredient, an appropriate formulation inert, a carrier, an adjuvant, and any mixtures thereof.
- Compounds of formula (I) may be used to control phytopathogenic microorganisms.
- a compound of formula (I), or a composition comprising a compound of formula (I) according to the invention may be applied directly to the phytopathogen, to the locus of a phytopathogen, in particular to a plant susceptible to attack by phytopathogens, or to a propagation material of a plant.
- the present invention provides the use of a compound of formula (I), or a composition comprising a compound of formula (I), as described herein to combat, prevent or control a phytopathogen.
- the present invention provides a method of combating, preventing or controlling phytopathogens, comprising applying a compound of formula (I), or a composition comprising a compound of formula (I), as described herein to said phytopathogen, to the locus of said phytopathogen, in particular to a plant susceptible to attack by a phytopathogen, or to a propagation material of a plant.
- Compounds of formula (I) are particularly effective in combating, preventing or controlling phytopathogenic fungi, in particular oomycetes.
- the present invention provides the use of a compound of formula (I), or a composition comprising a compound of formula (I), as described herein to control phytopathogenic fungi, in particular oomycetes.
- the present invention provides a method of combating, preventing or controlling phytopathogenic disease, such as phytopathogenic fungi, comprising applying a compound of formula (I), or a composition comprising a compound of formula (I), as described herein to said phytopathogenic fungi, or to the locus of said phytopathogenic fungi, in particular to a plant susceptible to attack by phytopathogenic fungi, in particular oomycetes, or to a propagation material of a plant.
- a group is indicated as being substituted, e.g. alkyl, this includes those groups that are part of other groups, e.g. the alkyl in alkylthio.
- halogen refers to fluorine (fluoro or F), chlorine (chloro or Cl), bromine (bromo or Br) or iodine (iodo or I), preferably fluorine, chlorine or bromine.
- amino refers to a -NH2 group.
- Alkyl as used herein- in isolation or as part of a chemical group – represents straight-chain or branched hydrocarbons, preferably with 1 to 6 carbon atoms, for example methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, s-butyl, t-butyl, pentyl, 1- methylbutyl, 2-methylbutyl, 3-methylbutyl, 1,2- dimethylpropyl, 1,1 -dimethylpropyl, 2,2- dimethylpropyl, 1 -ethylpropyl, hexyl, 1 -methylpentyl, 2- methylpentyl, 3-methylpentyl, 4- methylpentyl, 1,2-dimethylpropyl, 1,3-dimethylbutyl, 1,4-dimethylbutyl, 2,3-dimethylbutyl, 1,1- dimethylbutyl, 2,2-dimethylbut
- Alkyl groups with 1 to 4 carbon atoms are preferred, for example methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, s-butyl or t-butyl.
- Alkenyl in isolation or as part of a chemical group - represents straight-chain or branched hydrocarbons, preferably with 2 to 6 carbon atoms and at least one double bond, for example vinyl, 2- propenyl, 2-butenyl, 3-butenyl, 1- methyl-2-propenyl, 2-methyl-2-propenyl, 2-pentenyl, 3-pentenyl, 4- pentenyl, 1-methyl-2-butenyl, 2- methyl-2-butenyl, 3-methyl-2-butenyl, 1-methyl-3-butenyl, 2-methyl-3- butenyl, 3-methyl-3-butenyl, 1,1 - dimethyl-2-propenyl, 1,2-dimethyl-2-propenyl, 1 -ethyl-2-propenyl, 2- hexenyl, 3-hexenyl, 4- hexenyl, 5-hexenyl, 1 -methyl-2-pentenyl, 2-methyl-2-pentenyl, 2-methyl-2-penten
- Alkenyl groups with 2 to 4 carbon atoms are preferred, for example 2-propenyl, 2-butenyl or 1-methyl-2-propenyl.
- the term "Alkynyl" - in isolation or as part of a chemical group - represents straight-chain or branched hydrocarbons, preferably with 2 to 6 carbon atoms and at least one triple bond, for example 2-propynyl, 2-butynyl, 3-butynyl, 1-methyl-2- propynyl, 2-pentynyl, 3-pentynyl, 4-pentynyl, 1-methyl-3-butynyl, 2- methyl-3-butynyl, 1-methyl-2- butynyl, 1,1 -dimethyl-2-propynyl, 1 -ethyl-2-propynyl, 2-hexynyl, 3- hexynyl, 4-hexynyl, 5-hexynyl, 1- methyl-2-pentyn
- Alkynyls with 2 to 4 carbon atoms are preferred, for example ethynyl, 2- propynyl or 2-butynyl-2-propenyl.
- haloalkyl refers to an alkyl radical as generally defined above substituted by one or more of the same or different halogen atoms, for examples fluoromethyl, fluoroethyl, difluoromethyl, trifluoromethyl, or 2,2,2-trifluoroethyl.
- cyanoalkyl refers to an alkyl radical as generally defined above substituted by one or more cyano groups.
- cycloalkyl in isolation or as part of a chemical group - represents saturated or partially unsaturated mono-, bi- or tricyclic hydrocarbons, preferably with 3 to 10 carbon atoms, for example cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, bicyclo[2.2.1]heptyl, bicyclo[2.2.2]octyl or adamantyl. Cycloalkyls with 3, 4, 5, 6 or 7 carbon atoms are preferred, for example cyclopropyl or cyclobutyl.
- halocycloalkyl refers to a cycloalkyl ring as defined above substituted by one or more of the same or different halogen atoms.
- cyanocycloalkyl refers to a cycloalkyl radical as generally defined above substituted by one or more cyano groups.
- alkoxy refers to a radical of the formula -ORa wherein Ra is an alkyl radical as generally defined above. Examples of alkoxy include, but are not limited to methoxy, ethoxy, propoxy, iso-propoxy, and tert-butoxy.
- alkoxyalkyl refers to an alkyl radical (as mentioned above) substituted with said alkoxy group. Examples are methoxymethyl, methoxyethyl, ethoxymethyl and propoxymethyl.
- alkylsulfanyl refers to a radical of the formula -SRa wherein Ra is an alkyl radical as generally defined above.
- alkylsulfinyl refers to a radical of the formula -S(O)Ra wherein Ra is an alkyl radical as generally defined above.
- alkylsulfonyl refers to a radical of the formula -S(O)2Ra wherein Ra is an alkyl radical as generally defined above.
- alkylcarbonyl refers to a radical of the formula RaC(O)- wherein Ra is an alkyl radical as generally defined above.
- alkoxycarbonyl refers to a radical of the formula RaOC(O)-, wherein Ra is an alkyl radical as generally defined above.
- alkylamino refers to a radical of the formula RaNH- wherein Ra is an alkyl radical as generally defined above.
- cycloalkylamino refers to a radical of the formula RaNH- wherein Ra is a cycloalkyl radical as generally defined above.
- alkoxyamino refers to a radical of the formula R a NH-, wherein R a is an alkoxy radical as generally defined above.
- alkylaminocarbonyl refers to a radical of the formula RaNHC(O)- wherein Ra is an alkyl radical as generally defined above.
- - Hydroxyl or hydroxy stands for a –OH group.
- the term "effective amount” refers to the amount of the compound, a salt, or N-oxide thereof, which, upon single or multiple applications provides the desired effect. An effective amount is readily determined by the skilled person in the art, by the use of known techniques and by observing results obtained under analogous circumstances.
- Compounds of formula (I) which have at least one basic centre can form, for example, acid addition salts, for example with strong inorganic acids such as mineral acids, for example perchloric acid, sulfuric acid, nitric acid, nitrous acid, a phosphorus acid or a hydrohalic acid, with strong organic carboxylic acids, such as C1-4alkanecarboxylic acids which are unsubstituted or substituted, for example by halogen, for example acetic acid, such as saturated or unsaturated dicarboxylic acids, for example oxalic acid, malonic acid, succinic acid, maleic acid, fumaric acid or phthalic acid, such as hydroxycarboxylic acids, for example ascorbic acid, lactic acid, malic acid, tartaric
- Compounds of formula (I) which have at least one acidic group can form, for example, salts with bases, for example mineral salts such as alkali metal or alkaline earth metal salts, for example sodium, potassium or magnesium salts, or salts with ammonia or an organic amine, such as morpholine, piperidine, pyrrolidine, a mono-, di- or tri-lower-alkylamine, for example ethyl-, diethyl-, triethyl- or dimethylpropylamine, or a mono-, di- or trihydroxy-lower-alkylamine, for example mono-, di- or triethanolamine.
- bases for example mineral salts such as alkali metal or alkaline earth metal salts, for example sodium, potassium or magnesium salts
- salts with ammonia or an organic amine such as morpholine, piperidine, pyrrolidine, a mono-, di- or tri-lower-alkylamine, for example ethyl-, die
- the compounds of formula (I) according to the invention are in free form, in oxidized form as an N-oxide, in covalently hydrated form, or in salt form, e.g., an agronomically usable or agrochemically acceptable salt form.
- N-oxides are oxidized forms of tertiary amines or oxidized forms of nitrogen containing heteroaromatic compounds. They are described for instance in the book “Heterocyclic N-oxides” by A. Albini and S. Pietra, CRC Press, Boca Raton 1991.
- the compounds of formula (I) according to the invention also include hydrates, which may be formed during salt formation.
- the compounds of formula (I) according to the invention also include hydrates which may be formed during the salt formation.
- R 1a is selected from C1-6alkyl, C3-6cycloalkyl, C1-6alkoxy-C1-6alkyl, C3-6cycloalkyl-C1-4alkyl, and C 1-6 alkoxy, and preferably C 1-6 alkyl.
- R 1b and R 1c are independently selected from hydrogen, C1-6alkyl, C3-6cycloalkyl, C1-6alkoxy-C1- 6alkyl, C3-6cycloalkyl-C1-4alkyl, and C1-6alkoxy; and preferably hydrogen.
- R 1a is selected from hydroxy, halogen, CN, C1-6alkyl, C3-6cycloalkyl, C1-6alkoxy-C1- 6 alkyl, C 3-6 cycloalkyl-C 1-4 alkyl, C 1-6 alkylsulfanyl, C 1-6 alkylsulfinyl, C 1-6 alkylsulfonyl, C 1-6 alkoxy, amino, and NHC(O)C1-6alkyl; and R 1b and R 1c are hydrogen.
- R 2 are independently selected from hydrogen, hydroxy, halogen, CN, C1-6alkyl, C1-6alkoxy, C1- 6alkoxy-C1-6alkyl, and C1-6alkoxy-C1-6alkoxy, C1-6alkoxycarbonyl, C1-6alkylaminocarbonyl, diC1- 6alkylaminocarbonyl, and C1-6alkylcarbonyl, wherein each of the C1-6alkyl, C1-6alkoxy, C1-6alkoxy-C1- 6alkyl, C1-6alkoxy-C1-6alkoxy, C1-6alkoxycarbonyl, C1-6alkylaminocarbonyl, diC1-6alkylaminocarbonyl, and C 1-6 alkylcarbonyl groups is optionally substituted with one to three substituents independently selected from halogen, hydroxy, and CN; preferably R 2 are independently selected from hydrogen, halogen, CN,
- R 3 is selected from hydrogen, hydroxy, halogen, CN, C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkoxy-C 1-6 alkyl, C1-6alkoxy-C1-6alkoxy, C3-6cycloalkyl, C3-6cycloalkyl-C1-6alkyl, amino, C1-6alkylamino, diC1-6-alkylamino, and C3-6cycloalkylamino, wherein each of the C1-6alkyl, C1-6alkoxy, C1-6alkoxy-C1-6alkyl, C1-6alkoxy-C1- 6alkoxy, C3-6cycloalkyl, C3-6cycloalkyl-C1-6alkyl, amino, C1-6alkylamino, diC1-6-alkylamino and C3- 6cycloalkylamino groups is optionally substituted with one to three substituents independently selected from halogen, hydroxy, and
- R 3 can be hydrogen.
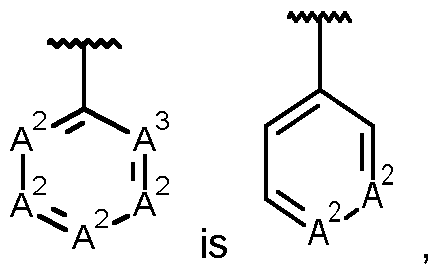
- a compound of formula (I) according to the present invention wherein four A 2 are CR 2 and A 3 is N.
- a compound of formula (I) according to the present invention wherein , and preferably the three A 2 are CR 2 and A 3 is CR 3 .
- a compound of formula (I) according to the present invention wherein preferably the three A 2 are CR 2 and A 3 is CR 3 .
- R 2 are independently selected from hydrogen, hydroxy, halogen, CN, C1-6alkyl, C1-6alkoxy, C1-6alkoxy-C1-6alkyl, C1-6alkoxy-C1-6alkoxy, C3-6cycloalkyl, C3-6cycloalkyl-C1-6alkyl, C1-6alkoxycarbonyl, C1-6alkylaminocarbonyl, diC1-6alkylaminocarbonyl, and C1-6alkylcarbonyl, wherein each of the C1-6alkyl, C1-6alkoxy, C1-6alkoxy-C1-6alkyl, C1-6alkoxy-C1-6alkoxy, C3-6cycloalkyl, C3-6cycloalkyl-C1-6alkyl, C1- 6alkoxycarbonyl, C1-6alkyla
- R 4 is selected from C1-6alkyl, C1-6alkoxy-C1-6alkyl, C3-6cycloalkyl, C3-6cycloalkyl-C1-4alkyl, C2- 6alkenyl, C2-6alkynyl, and C1-6alkoxy, wherein each of the C1-6alkyl, C1-6alkoxy-C1-6alkyl, C3-6cycloalkyl, C3-6cycloalkyl-C1-4alkyl, C2-6alkenyl, C2-6alkynyl, and C1-6alkoxy groups is optionally substituted with one to three substituents independently selected from halogen and CN; preferably and R 4 is selected from C1-6alkyl, C1-6alkoxy-C1-6alkyl, C3-6cycloalkyl, C3-6cycloalkyl-C1-4alkyl, and C1-6alkoxy, wherein each of the C1-6alkyl, C1-6alk
- a compound of formula (I) according to the present invention, wherein A 3 is CR 3 and wherein R 3 and R 4 taken together form a ring, preferably a 5-8-membered heterocycle, preferably a 6-membered heterocycle, and more preferably one of the rings W1, W2 or W3 as described in the compounds of the formula (I) below:
- the carbon and/or the nitrogen atoms forming said ring (W1, W2 or W3) can be substituted, especially by a R 3’ group, wherein R 3’ is selected from hydrogen, C1-6alkyl, and C3-6cycloalkyl, wherein each of the C1-6alkyl and C3-6cycloalkyl groups is optionally substituted with one to three substituents independently selected from halogen and CN.
- the compounds of the formula (I-W3) can be as follows:
- the compounds of the formula (I-W1), (I-W2) and (I-W3) can be as described below:
- the carbon and/or the nitrogen atoms forming said ring (W1, W2 or W3) can be substituted, especially by a R 3’ group, wherein R 3’ is selected from hydrogen, C1-6alkyl, and C3-6cycloalkyl, wherein each of the C1-6alkyl and C3-6cycloalkyl groups is optionally substituted with one to three substituents independently selected from halogen and CN.
- the compounds of the formula (I-W3) can be as follows:
- R 5 is selected from C1-6alkyl, C1-6alkoxy, C3-6cycloalkyl, C3-6cycloalkyl-C1-6alkyl, and C1- 6alkoxyC1-6alkyl, wherein each of said groups is optionally substituted with one to three substituents independently selected from halogen and CN.
- a compound of formula (I) wherein Z is O; A 1 is N; R 1a is selected from C1-6alkyl, C3-6cycloalkyl, C1-6alkoxy-C1-6alkyl, C3-6cycloalkyl-C1-4alkyl, and C1-6alkoxy, and preferably C1-6alkyl; R 1b is selected from hydrogen, C1-6alkyl, C3-6cycloalkyl, C1-6alkoxy-C1-6alkyl, C3-6cycloalkyl-C1-4alkyl, and C1-6alkoxy, and preferably hydrogen; R 1c is selected from hydrogen, C1-6alkyl, C3-6cycloalkyl, C1-6alkoxy-C1-6alkyl, C3-6cycloalkyl-C1-4alkyl, and C1-6alkoxy, and preferably hydrogen; the four A 2 are CR 2 ; with R 2 being independently selected from hydrogen, hydroxy, halogen, CN, C1- 6alkyl, C1-6
- R 2 is as defined in the present invention; preferably R 2 are independently selected from hydrogen, hydroxy, halogen, CN, C1-6alkyl, C1-6alkoxy, C1-6alkoxy-C1-6alkyl, C1-6alkoxy-C1-6alkoxy, C3-6cycloalkyl, C3-6cycloalkyl-C1-6alkyl, C1-6alkoxycarbonyl, C1-6alkylaminocarbonyl, diC1-6alkylaminocarbonyl, and C1-6alkylcarbonyl, wherein each of the C1-6alkyl, C1-6alkoxy, C1-6alkoxy-C1-6alkyl, C1-6alkoxy-C1-6alkoxy, C3-6cycloalkyl, C3-6cycloalkyl-C1-6alkyl, C1- 6alkoxycarbonyl, C1-6alkylaminocarbonyl, diC1-6alkylaminocarbonyl, and C1-6alkylcarbonyl groups is optionally substituted with one to three substitu
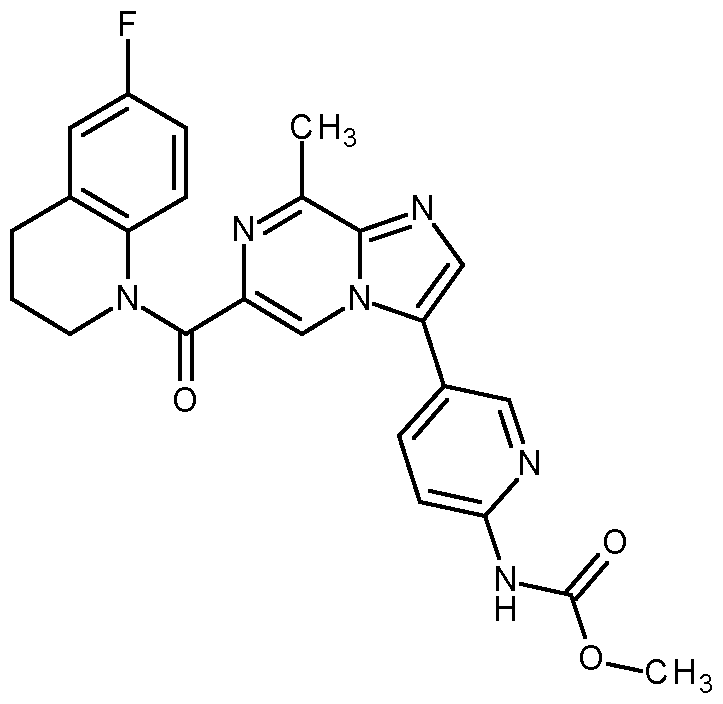
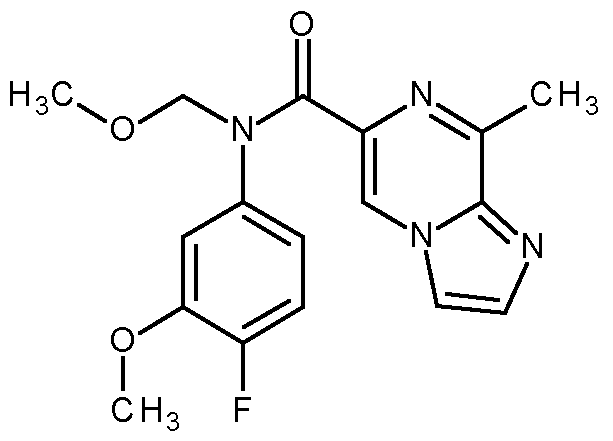
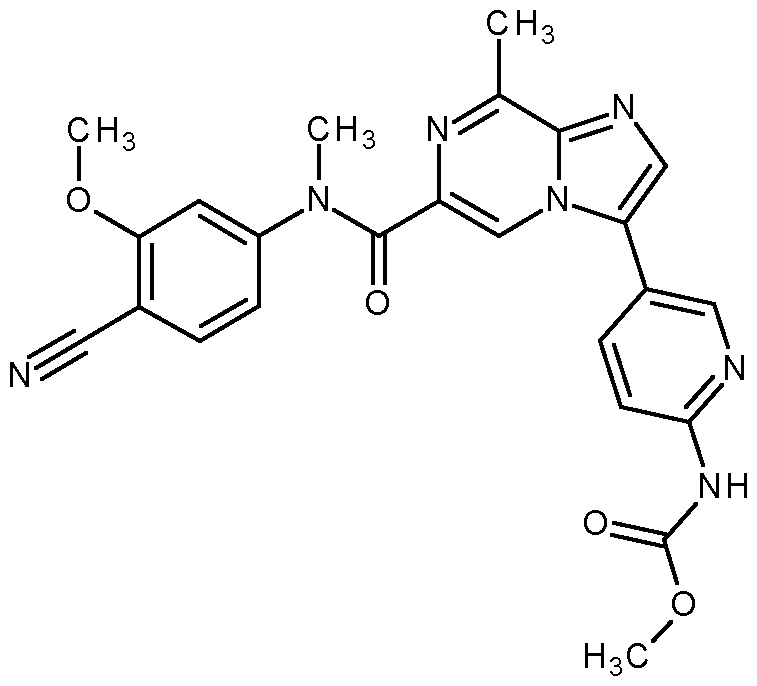
- the compound according to the present invention is selected from: methyl N-[5-[6-[2-cyanoethyl-(4-fluoro-3-methoxy-phenyl)carbamoyl]-8-methyl-imidazo[1,2-a]pyrazin-3- yl]-2-pyridyl]carbamate; methyl N-[5-[6-[cyanomethyl-(4-fluoro-3-methoxy-phenyl)carbamoyl]-8-methyl-imidazo[1,2-a]pyrazin-3- yl]-2-pyridyl]carbamate; methyl N-[5-[6-[ethyl-(4-fluoro-3-methoxy-phenyl)carbamoyl]-8-methyl-imidazo[1,2-a]pyrazin-3-yl]-2- pyridyl]carbamate; methyl N-[5-[6-[6-(6-fluoro-3,4-fluor
- the method according to the present invention has advantageous properties for protecting plants against pathogenic, such as phytopathogenic, especially fungi such as oomycetes, attack or infestation, which result in a disease and damage to the plant; particularly in instance of plants, the present invention can control, limit or prevent pathogenic damage on plant, parts of plant, plant propagation material and/or plant grown.
- Tables 1.1 to 1.297 below illustrate specific compounds of the invention.
- Table 1.1 provides compounds E1.1 to E1.418 of formula (Ia) wherein R 1a is CH3, R 2 is H, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z. More particularly, table Z also includes compounds wherein A 3 and R 4 taken together form a ring.
- Table 1.3 provides 418 compounds E3.1 to E3.418 of formula (Ia) wherein R 1a is CH3, R 2 is H, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.4 provides 418 compounds E4.1 to E4.418 of formula (Ia) wherein R 1a is CH3, R 2 is H, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.5 provides 418 compounds E5.1 to E5.418 of formula (Ia) wherein R 1a is CH3, R 2 is H, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.6 provides 418 compounds E6.1 to E6.418 of formula (Ia) wherein R 1a is CH3, R 2 is CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.7 provides 418 compounds E7.1 to E7.418 of formula (Ia) wherein R 1a is CH3, R 2 is CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.8 provides 418 compounds E8.1 to E8.418 of formula (Ia) wherein R 1a is CH3, R 2 is CH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.9 provides 418 compounds E9.1 to E9.418 of formula (Ia) wherein R 1a is CH3, R 2 is CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.10 provides 418 compounds E10.1 to E10.418 of formula (Ia) wherein R 1a is CH 3 , R 2 is CH 3 , R5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.11 provides 418 compounds E11.1 to E11.418 of formula (Ia) wherein R 1a is CH3, R 2 is CH2CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.12 provides 418 compounds E12.1 to E12.418 of formula (Ia) wherein R 1a is CH3, R 2 is CH2CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.13 provides 418 compounds E13.1 to E13.418 of formula (Ia) wherein R 1a is CH3, R 2 is CH2CH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.14 provides 418 compounds E14.1 to E14.418 of formula (Ia) wherein R 1a is CH 3 , R 2 is CH 2 CH 3 , R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.15 provides 418 compounds E15.1 to E15.418 of formula (Ia) wherein R 1a is CH3, R 2 is CH2CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.16 provides 418 compounds E16.1 to E16.418 of formula (Ia) wherein R 1a is CH3, R 2 is F, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.17 provides 418 compounds E17.1 to E17.418 of formula (Ia) wherein R 1a is CH3, R 2 is F, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.18 provides 418 compounds E18.1 to E18.418 of formula (Ia) wherein R 1a is CH 3 , R 2 is F, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.19 provides 418 compounds E19.1 to E19.418 of formula (Ia) wherein R 1a is CH3, R 2 is F, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.20 provides 418 compounds E20.1 to E20.418 of formula (Ia) wherein R 1a is CH3, R 2 is F, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.21 provides 418 compounds E21.1 to E21.418 of formula (Ia) wherein R 1a is CH3, R 2 is Cl, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.22 provides 418 compounds E22.1 to E22.418 of formula (Ia) wherein R 1a is CH 3 , R 2 is Cl, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.23 provides 418 compounds E23.1 to E23.418 of formula (Ia) wherein R 1a is CH3, R 2 is Cl, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.24 provides 418 compounds E24.1 to E24.418 of formula (Ia) wherein R 1a is CH3, R 2 is Cl, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.25 provides 418 compounds E25.1 to E25.418 of formula (Ia) wherein R 1a is CH3, R 2 is Cl, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.26 provides 418 compounds E26.1 to E26.418 of formula (Ia) wherein R 1a is CH 3 , R 2 is Br, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.27 provides 418 compounds E27.1 to E27.418 of formula (Ia) wherein R 1a is CH3, R 2 is Br, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.28 provides 418 compounds E28.1 to E28.418 of formula (Ia) wherein R 1a is CH3, R 2 is Br, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.29 provides 418 compounds E29.1 to E29.418 of formula (Ia) wherein R 1a is CH3, R 2 is Br, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.30 provides 418 compounds E30.1 to E30.418 of formula (Ia) wherein R 1a is CH3, R 2 is Br, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.31 provides 418 compounds E31.1 to E31.418 of formula (Ia) wherein R 1a is CH 3 , R 2 is CN, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.32 provides 418 compounds E32.1 to E32.418 of formula (Ia) wherein R 1a is CH3, R 2 is CN, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.33 provides 418 compounds E33.1 to E33.418 of formula (Ia) wherein R 1a is CH3, R 2 is CN, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.34 provides 418 compounds E34.1 to E34.418 of formula (Ia) wherein R 1a is CH3, R 2 is CN, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.35 provides 418 compounds E35.1 to E35.418 of formula (Ia) wherein R 1a is CH 3 , R 2 is CN, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.36 provides 418 compounds E36.1 to E36.418 of formula (Ia) wherein R 1a is CH3, R 2 is OCH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.37 provides 418 compounds E37.1 to E37.418 of formula (Ia) wherein R 1a is CH3, R 2 is OCH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.38 provides 418 compounds E38.1 to E38.418 of formula (Ia) wherein R 1a is CH3, R 2 is OCH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.39 provides 418 compounds E39.1 to E39.418 of formula (Ia) wherein R 1a is CH 3 , R 2 is OCH 3 , R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.40 provides 418 compounds E40.1 to E40.418 of formula (Ia) wherein R 1a is CH3, R 2 is OCH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.41 provides 418 compounds E41.1 to E41.418 of formula (Ia) wherein R 1a is CH3, R 2 is OCH2CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.42 provides 418 compounds E42.1 to E42.418 of formula (Ia) wherein CH3, R 2 is OCH2CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.43 provides 418 compounds E43.1 to E43.418 of formula (Ia) wherein R 1a is CH 3 , R 2 is OCH2CH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.44 provides 418 compounds E44.1 to E44.418 of formula (Ia) wherein CH3, R 2 is OCH2CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.45 provides 418 compounds E45.1 to E45.418 of formula (Ia) wherein R 1a is CH3, R 2 is OCH2CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.46 provides 418 compounds E46.1 to E46.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is H, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.47 provides 418 compounds E47.1 to E47.418 of formula (Ia) wherein R 1a is CH 2 CH 3 , R 2 is H, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.48 provides 418 compounds E48.1 to E48.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is H, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.49 provides 418 compounds E49.1 to E49.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is H, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.50 provides 418 compounds E50.1 to E50.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is H, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.51 provides 418 compounds E51.1 to E51.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.52 provides 418 compounds E52.1 to E52.418 of formula (Ia) wherein R 1a is CH 2 CH 3 , R 2 is CH 3 , R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.53 provides 418 compounds E53.1 to E53.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is CH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.54 provides 418 compounds E54.1 to E54.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.55 provides 418 compounds E55.1 to E55.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.56 provides 418 compounds E56.1 to E56.418 of formula (Ia) wherein R 1a is CH 2 CH 3 , R 2 is CH2CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.57 provides 418 compounds E57.1 to E57.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is CH2CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.58 provides 418 compounds E58.1 to E58.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is CH2CH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.59 provides 418 compounds E59.1 to E59.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is CH2CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.60 provides 418 compounds E60.1 to E60.418 of formula (Ia) wherein R 1a is CH 2 CH 3 , R 2 is CH2CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.61 provides 418 compounds E61.1 to E61.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is F, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.62 provides 418 compounds E62.1 to E62.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is F, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.63 provides 418 compounds E63.1 to E63.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is F, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.64 provides 418 compounds E64.1 to E64.418 of formula (Ia) wherein R 1a is CH 2 CH 3 , R 2 is F, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.65 provides 418 compounds E65.1 to E65.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is F, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.66 provides 418 compounds E66.1 to E66.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is Cl, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.67 provides 418 compounds E67.1 to E67.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is Cl, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.68 provides 418 compounds E68.1 to E68.418 of formula (Ia) wherein R 1a is CH 2 CH 3 , R 2 is Cl, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.69 provides 418 compounds E69.1 to E69.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is Cl, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.70 provides 418 compounds E70.1 to E70.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is Cl, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.71 provides 418 compounds E71.1 to E71.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is Br, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.72 provides 418 compounds E72.1 to E72.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is Br, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.73 provides 418 compounds E73.1 to E73.418 of formula (Ia) wherein R 1a is CH 2 CH 3 , R 2 is Br, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.74 provides 418 compounds E74.1 to E74.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is Br, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.75 provides 418 compounds E75.1 to E75.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is Br, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.76 provides 418 compounds E76.1 to E76.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is CN, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.77 provides 418 compounds E77.1 to E77.418 of formula (Ia) wherein R 1a is CH 2 CH 3 , R 2 is CN, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.78 provides 418 compounds E78.1 to E78.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is CN, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.79 provides 418 compounds E79.1 to E79.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is CN, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.80 provides 418 compounds E80.1 to E80.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is CN, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.81 provides 418 compounds E81.1 to E81.418 of formula (Ia) wherein R 1a is CH 2 CH 3 , R 2 is OCH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.82 provides 418 compounds E82.1 to E82.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is OCH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.83 provides 418 compounds E83.1 to E83.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is OCH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.84 provides 418 compounds E84.1 to E84.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is OCH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.85 provides 418 compounds E85.1 to E85.418 of formula (Ia) wherein R 1a is CH 2 CH 3 , R 2 is OCH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.86 provides 418 compounds E86.1 to E86.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is OCH2CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.87 provides 418 compounds E87.1 to E87.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is OCH2CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.88 provides 418 compounds E88.1 to E88.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is OCH2CH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.89 provides 418 compounds E89.1 to E89.418 of formula (Ia) wherein R 1a is CH 2 CH 3 , R 2 is OCH2CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.90 provides 418 compounds E90.1 to E90.418 of formula (Ia) wherein R 1a is CH2CH3, R 2 is OCH2CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.91 provides 418 compounds E91.1 to E91.418 of formula (Ia) wherein R 1a is CH2OCH3, R 2 is H, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.92 provides 418 compounds E92.1 to E92.418 of formula (Ia) wherein R 1a is CH2OCH3, R 2 is H, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.93 provides 418 compounds E93.1 to E93.418 of formula (Ia) wherein R 1a is CH2OCH3, R 2 is H, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.94 provides 418 compounds E94.1 to E94.418 of formula (Ia) wherein R 1a is CH 2 OCH 3 , R 2 is H, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.95 provides 418 compounds E95.1 to E95.418 of formula (Ia) wherein R 1a is CH2OCH3, R 2 is H, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.96 provides 418 compounds E96.1 to E96.418 of formula (Ia) wherein R 1a is CH2OCH3, R 2 is CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.97 provides 418 compounds E97.1 to E97.418 of formula (Ia) wherein R 1a is CH2OCH3, R 2 is CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.98 provides 418 compounds E98.1 to E98.418 of formula (Ia) wherein R 1a is CH 2 OCH 3 , R 2 is CH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.99 provides 418 compounds E99.1 to E99.418 of formula (Ia) wherein R 1a is CH2OCH3, R 2 is CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.100 provides 418 compounds E100.1 to E100.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.101 provides 418 compounds E101.1 to E101.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is CH2CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.102 provides 418 compounds E102.1 to E102.418 of formula (Ia) wherein R 1a is CH 2 OCH 3 , R2 is CH2CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.103 provides 418 compounds E103.1 to E103.418 of formula (Ia) wherein R 1a is CH2OCH3, R 2 is CH2CH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.104 provides 418 compounds E104.1 to E104.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is CH2CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.105 provides 418 compounds E105.1 to E105.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is CH2CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.106 provides 418 compounds E106.1 to E106.418 of formula (Ia) wherein R 1a is CH 2 OCH 3 , R2 is F, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.107 provides 418 compounds E107.1 to E107.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is F, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.108 provides 418 compounds E108.1 to E108.418 of formula (Ia) wherein R 1a is CH2OCH3, R 2 is F, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.109 provides 418 compounds E109.1 to E109.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is F, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.110 provides 418 compounds E110.1 to E110.418 of formula (Ia) wherein R 1a is CH 2 OCH 3 , R2 is F, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.111 provides 418 compounds E111.1 to E111.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is Cl, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.112 provides 418 compounds E112.1 to E112.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is Cl, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.113 provides 418 compounds E113.1 to E113.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is Cl, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.114 provides 418 compounds E114.1 to E114.418 of formula (Ia) wherein R 1a is CH2OCH3, R 2 is Cl, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.115 provides 418 compounds E115.1 to E115.418 of formula (Ia) wherein R 1a is CH 2 OCH 3 , R2 is Cl, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.116 provides 418 compounds E116.1 to E116.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is Br, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.117 provides 418 compounds E117.1 to E117.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is Br, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.118 provides 418 compounds E118.1 to E118.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is Br, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.119 provides 418 compounds E119.1 to E119.418 of formula (Ia) wherein R 1a is CH 2 OCH 3 , R 2 is Br, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.120 provides 418 compounds E120.1 to E120.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is Br, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.121 provides 418 compounds E121.1 to E121.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is CN, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.122 provides 418 compounds E122.1 to E122.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is CN, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.123 provides 418 compounds E123.1 to E123.418 of formula (Ia) wherein R 1a is CH 2 OCH 3 , R2 is CN, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.124 provides 418 compounds E124.1 to E124.418 of formula (Ia) wherein R 1a is CH2OCH3, R 2 is CN, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.125 provides 418 compounds E125.1 to E125.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is CN, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.126 provides 418 compounds E126.1 to E126.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is OCH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.127 provides 418 compounds E127.1 to E127.418 of formula (Ia) wherein R 1a is CH 2 OCH 3 , R2 is OCH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.128 provides 418 compounds E128.1 to E128.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is OCH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.129 provides 418 compounds E129.1 to E129.418 of formula (Ia) wherein R 1a is CH2OCH3, R 2 is OCH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.130 provides 418 compounds E130.1 to E130.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is OCH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.131 provides 418 compounds E131.1 to E131.418 of formula (Ia) wherein R 1a is CH 2 OCH 3 , R2 is OCH2CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.132 provides 418 compounds E132.1 to E132.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is OCH2CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.133 provides 418 compounds E133.1 to E133.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is OCH2CH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.134 provides 418 compounds E134.1 to E134.418 of formula (Ia) wherein R 1a is CH2OCH3, R2 is OCH2CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.135 provides 418 compounds E135.1 to E135.418 of formula (Ia) wherein R 1a is CH2OCH3, R 2 is OCH2CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.136 provides 418 compounds E136.1 to E136.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is H, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.137 provides 418 compounds E137.1 to E137.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is H, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.138 provides 418 compounds E138.1 to E138.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is H, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.139 provides 418 compounds E139.1 to E139.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is H, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.140 provides 418 compounds E140.1 to E140.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.141 provides 418 compounds E141.1 to E141.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.142 provides 418 compounds E142.1 to E142.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.143 provides 418 compounds E143.1 to E143.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.144 provides 418 compounds E144.1 to E144.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is CH2CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.145 provides 418 compounds E145.1 to E145.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is CH2CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.146 provides 418 compounds E146.1 to E146.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is CH2CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.147 provides 418 compounds E147.1 to E147.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is CH2CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.148 provides 418 compounds E148.1 to E148.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is F, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.149 provides 418 compounds E149.1 to E149.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is F, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.150 provides 418 compounds E150.1 to E150.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is F, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.151 provides 418 compounds E151.1 to E151.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is F, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.152 provides 418 compounds E152.1 to E152.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is Cl, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.153 provides 418 compounds E153.1 to E153.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is Cl, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.154 provides 418 compounds E154.1 to E154.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is Cl, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.155 provides 418 compounds E155.1 to E155.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is Cl, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.156 provides 418 compounds E156.1 to E156.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is Br, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.157 provides 418 compounds E157.1 to E157.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is Br, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.158 provides 418 compounds E158.1 to E158.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is Br, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.159 provides 418 compounds E159.1 to E159.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is Br, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.160 provides 418 compounds E160.1 to E160.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is CN, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.161 provides 418 compounds E161.1 to E161.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is CN, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.162 provides 418 compounds E162.1 to E162.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is CN, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.163 provides 418 compounds E163.1 to E163.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is CN, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.164 provides 418 compounds E164.1 to E164.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is OCH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.165 provides 418 compounds E165.1 to E165.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is OCH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.166 provides 418 compounds E166.1 to E166.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is OCH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.167 provides 418 compounds E167.1 to E167.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is OCH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.168 provides 418 compounds E168.1 to E168.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is OCH2CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.169 provides 418 compounds E169.1 to E169.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is OCH2CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.170 provides 418 compounds E170.1 to E170.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is OCH2CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.171 provides 418 compounds E171.1 to E171.418 of formula (Ia) wherein R 1a is cyclopropyl, R 2 is OCH2CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.171 provides 418 compounds E172.1 to E172.418 of formula (Ia) wherein R 1a is is H, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.173 provides 418 compounds E173.1 to E173.418 of formula (Ia) wherein R 1a is , R 2 is H, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.174 provides 418 compounds E174.1 to E174.418 of formula (Ia) wherein R 1a is , R 2 is H, R 5 is CH 2 OCH 3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.175 provides 418 compounds E175.1 to E175.418 of formula (Ia) wherein R 1a is , R 2 is H, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.176 provides 418 compounds E176.1 to E176.418 of formula (Ia) wherein R 1a is , R 2 is CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.177 provides 418 compounds E177.1 to E177.418 of formula (Ia) wherein R 1a is , R 2 is CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.178 provides 418 compounds E178.1 to E178.418 of formula (Ia) wherein R 1a is , R 2 is CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.179 provides 418 compounds E179.1 to E179.418 of formula (Ia) wherein R 1a is , R 2 is CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.180 provides 418 compounds E180.1 to E180.418 of formula (Ia) wherein R 1a is , R 2 is CH2CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.181 provides 418 compounds E181.1 to E181.418 of formula (Ia) wherein R 1a is , R 2 is CH2CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.182 provides 418 compounds E182.1 to E182.418 of formula (Ia) wherein R 1a is , R 2 is CH2CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.183 provides 418 compounds E183.1 to E183.418 of formula (Ia) wherein R 1a is , R 2 is CH2CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.184 provides 418 compounds E184.1 to E184.418 of formula (Ia) wherein R 1a is , R 2 is F, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.185 provides 418 compounds E185.1 to E185.418 of formula (Ia) wherein R 1a is , R 2 is F, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.186 provides 418 compounds E186.1 to E186.418 of formula (Ia) wherein R 1a is , R 2 is F, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.187 provides 418 compounds E187.1 to E187.418 of formula (Ia) wherein R 1a is , R 2 is F, R 5 is NHCH 2 CH 3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.188 provides 418 compounds E188.1 to E188.418 of formula (Ia) wherein R 1a is , R 2 is Cl, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.189 provides 418 compounds E189.1 to E189.418 of formula (Ia) wherein R 1a is , R 2 is Cl, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.190 provides 418 compounds E190.1 to E190.418 of formula (Ia) wherein R 1a is , R 2 is Cl, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.191 provides 418 compounds E191.1 to E191.418 of formula (Ia) wherein R 1a is , R 2 is Cl, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.192 provides 418 compounds E192.1 to E192.418 of formula (Ia) wherein R 1a is , R 2 is Br, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.193 provides 418 compounds E193.1 to E193.418 of formula (Ia) wherein R 1a is , R 2 is Br, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.194 provides 418 compounds E194.1 to E194.418 of formula (Ia) wherein R 1a is , R 2 is Br, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.195 provides 418 compounds E195.1 to E195.418 of formula (Ia) wherein R 1a is , R 2 is Br, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.196 provides 418 compounds E196.1 to E196.418 of formula (Ia) wherein R 1a is , R 2 is CN, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.197 provides 418 compounds E197.1 to E197.418 of formula (Ia) wherein R 1a is , R 2 is CN, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.198 provides 418 compounds E198.1 to E198.418 of formula (Ia) wherein R 1a is , R 2 is CN, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.199 provides 418 compounds E199.1 to E199.418 of formula (Ia) wherein R 1a is , R 2 is CN, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.200 provides 418 compounds E200.1 to E200.418 of formula (Ia) wherein R 1a is , R 2 is OCH 3 , R 5 is CH 3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.201 provides 418 compounds E201.1 to E201.418 of formula (Ia) wherein R 1a is , R 2 is OCH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.202 provides 418 compounds E202.1 to E202.418 of formula (Ia) wherein R 1a is , R 2 is OCH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.203 provides 418 compounds E203.1 to E203.418 of formula (Ia) wherein R 1a is , R 2 is OCH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.204 provides 418 compounds E204.1 to E204.418 of formula (Ia) wherein R 1a is , R 2 is OCH2CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.205 provides 418 compounds E205.1 to E205.418 of formula (Ia) wherein R 1a is , R 2 is OCH2CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.206 provides 418 compounds E206.1 to E206.418 of formula (Ia) wherein R 1a is , R 2 is OCH2CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.207 provides 418 compounds E207.1 to E207.418 of formula (Ia) wherein R 1a is , R 2 is OCH2CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.208 provides 418 compounds E208.1 to E208.418 of formula (Ia) wherein R 1a is NHAc, R 2 is H, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.209 provides 418 compounds E209.1 to E209.418 of formula (Ia) wherein R 1a is NHAc, R 2 is H, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.210 provides 418 compounds E210.1 to E210.418 of formula (Ia) wherein R 1a is NHAc, R 2 is H, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.211 provides 418 compounds E211.1 to E211.418 of formula (Ia) wherein R 1a is NHAc, R 2 is H, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.212 provides 418 compounds E212.1 to E212.418 of formula (Ia) wherein R 1a is NHAc, R 2 is H, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.213 provides 418 compounds E213.1 to E213.418 of formula (Ia) wherein R 1a is NHAc, R 2 is CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.214 provides 418 compounds E214.1 to E214.418 of formula (Ia) wherein R 1a is NHAc, R 2 is CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.215 provides 418 compounds E215.1 to E215.418 of formula (Ia) wherein R 1a is NHAc, R 2 is CH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.216 provides 418 compounds E216.1 to E216.418 of formula (Ia) wherein R 1a is NHAc, R 2 is CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.217 provides 418 compounds E217.1 to E217.418 of formula (Ia) wherein R 1a is NHAc, R 2 is CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.218 provides 418 compounds E218.1 to E218.418 of formula (Ia) wherein R 1a is NHAc, R 2 is CH2CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.219 provides 418 compounds E219.1 to E219.418 of formula (Ia) wherein R 1a is NHAc, R 2 is CH2CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.220 provides 418 compounds E220.1 to E220.418 of formula (Ia) wherein R 1a is NHAc, R 2 is CH2CH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.221 provides 418 compounds E221.1 to E221.418 of formula (Ia) wherein R 1a is NHAc, R 2 is CH2CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.222 provides 418 compounds E222.1 to E222.418 of formula (Ia) wherein R 1a is NHAc, R 2 is CH2CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.223 provides 418 compounds E223.1 to E223.418 of formula (Ia) wherein R 1a is NHAc, R 2 is F, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.224 provides 418 compounds E224.1 to E224.418 of formula (Ia) wherein R 1a is NHAc, R 2 is F, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.225 provides 418 compounds E225.1 to E225.418 of formula (Ia) wherein R 1a is NHAc, R 2 is F, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.226 provides 418 compounds E226.1 to E226.418 of formula (Ia) wherein R 1a is NHAc, R 2 is F, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.227 provides 418 compounds E227.1 to E227.418 of formula (Ia) wherein R 1a is NHAc, R 2 is F, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.228 provides 418 compounds E228.1 to E228.418 of formula (Ia) wherein R 1a is NHAc, R 2 is Cl, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.229 provides 418 compounds E229.1 to E229.418 of formula (Ia) wherein R 1a is NHAc, R 2 is Cl, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.230 provides 418 compounds E230.1 to E230.418 of formula (Ia) wherein R 1a is NHAc, R 2 is Cl, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.231 provides 418 compounds E231.1 to E231.418 of formula (Ia) wherein R 1a is NHAc, R 2 is Cl, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.232 provides 418 compounds E232.1 to E232.418 of formula (Ia) wherein R 1a is NHAc, R 2 is Cl, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.233 provides 418 compounds E233.1 to E233.418 of formula (Ia) wherein R 1a is NHAc, R 2 is Br, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.234 provides 418 compounds E234.1 to E234.418 of formula (Ia) wherein R 1a is NHAc, R 2 is Br, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.235 provides 418 compounds E235.1 to E235.418 of formula (Ia) wherein R 1a is NHAc, R 2 is Br, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.236 provides 418 compounds E236.1 to E236.418 of formula (Ia) wherein R 1a is NHAc, R 2 is Br, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.237 provides 418 compounds E237.1 to E237.418 of formula (Ia) wherein R 1a is NHAc, R 2 is Br, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.238 provides 418 compounds E238.1 to E238.418 of formula (Ia) wherein R 1a is NHAc, R 2 is CN, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.239 provides 418 compounds E239.1 to E239.418 of formula (Ia) wherein R 1a is NHAc, R 2 is CN, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.240 provides 418 compounds E240.1 to E240.418 of formula (Ia) wherein R 1a is NHAc, R 2 is CN, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.241 provides 418 compounds E241.1 to E241.418 of formula (Ia) wherein R 1a is NHAc, R 2 is CN, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.242 provides 418 compounds E242.1 to E242.418 of formula (Ia) wherein R 1a is NHAc, R 2 is CN, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.243 provides 418 compounds E243.1 to E243.418 of formula (Ia) wherein R 1a is NHAc, R 2 is OCH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.244 provides 418 compounds E244.1 to E244.418 of formula (Ia) wherein R 1a is NHAc, R 2 is OCH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.245 provides 418 compounds E245.1 to E245.418 of formula (Ia) wherein R 1a is NHAc, R 2 is OCH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.246 provides 418 compounds E246.1 to E246.418 of formula (Ia) wherein R 1a is NHAc, R 2 is OCH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.247 provides 418 compounds E247.1 to E247.418 of formula (Ia) wherein R 1a is NHAc, R 2 is OCH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.248 provides 418 compounds E248.1 to E248.418 of formula (Ia) wherein R 1a is NHAc, R 2 is OCH2CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.249 provides 418 compounds E249.1 to E249.418 of formula (Ia) wherein R 1a is NHAc, R 2 is OCH2CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.250 provides 418 compounds E250.1 to E250.418 of formula (Ia) wherein R 1a is NHAc, R 2 is OCH2CH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.251 provides 418 compounds E251.1 to E251.418 of formula (Ia) wherein R 1a is NHAc, R 2 is OCH2CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.252 provides 418 compounds E252.1 to E252.418 of formula (Ia) wherein R 1a is NHAc, R 2 is OCH2CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.253 provides 418 compounds E253.1 to E253.418 of formula (Ia) wherein R 1a is CN, R 2 is H, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.254 provides 418 compounds E254.1 to E254.418 of formula (Ia) wherein R 1a is CN, R 2 is H, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.255 provides 418 compounds E255.1 to E255.418 of formula (Ia) wherein R 1a is CN, R 2 is H, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.256 provides 418 compounds E256.1 to E256.418 of formula (Ia) wherein R 1a is CN, R 2 is H, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.257 provides 418 compounds E257.1 to E257.418 of formula (Ia) wherein R 1a is CN, R 2 is H, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.258 provides 418 compounds E258.1 to E258.418 of formula (Ia) wherein R 1a is CN, R 2 is CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.259 provides 418 compounds E259.1 to E259.418 of formula (Ia) wherein R 1a is CN, R 2 is CH 3 , R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.260 provides 418 compounds E260.1 to E260.418 of formula (Ia) wherein R 1a is CN, R 2 is CH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.261 provides 418 compounds E261.1 to E261.418 of formula (Ia) wherein R 1a is CN, R 2 is CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.262 provides 418 compounds E262.1 to E262.418 of formula (Ia) wherein R 1a is CN, R 2 is CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.263 provides 418 compounds E263.1 to E263.418 of formula (Ia) wherein R 1a is CN, R 2 is CH2CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.264 provides 418 compounds E264.1 to E264.418 of formula (Ia) wherein R 1a is CN, R 2 is CH2CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.265 provides 418 compounds E265.1 to E265.418 of formula (Ia) wherein R 1a is CN, R 2 is CH2CH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.266 provides 418 compounds E266.1 to E266.418 of formula (Ia) wherein R 1a is CN, R 2 is CH2CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.267 provides 418 compounds E267.1 to E267.418 of formula (Ia) wherein R 1a is CN, R 2 is CH2CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.268 provides 418 compounds E268.1 to E268.418 of formula (Ia) wherein R 1a is CN, R 2 is F, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.269 provides 418 compounds E269.1 to E269.418 of formula (Ia) wherein R 1a is CN, R 2 is F, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.270 provides 418 compounds E270.1 to E270.418 of formula (Ia) wherein R 1a is CN, R 2 is F, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.271 provides 418 compounds E271.1 to E271.418 of formula (Ia) wherein R 1a is CN, R 2 is F, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.272 provides 418 compounds E272.1 to E272.418 of formula (Ia) wherein R 1a is CN, R 2 is F, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.273 provides 418 compounds E273.1 to E273.418 of formula (Ia) wherein R 1a is CN, R 2 is Cl, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.274 provides 418 compounds E274.1 to E274.418 of formula (Ia) wherein R 1a is CN, R 2 is Cl, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.275 provides 418 compounds E275.1 to E275.418 of formula (Ia) wherein R 1a is CN, R 2 is Cl, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.276 provides 418 compounds E276.1 to E276.418 of formula (Ia) wherein R 1a is CN, R 2 is Cl, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.277 provides 418 compounds E277.1 to E277.418 of formula (Ia) wherein R 1a is CN, R 2 is Cl, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.278 provides 418 compounds E278.1 to E278.418 of formula (Ia) wherein R 1a is CN, R 2 is Br, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.279 provides 418 compounds E279.1 to E279.418 of formula (Ia) wherein R 1a is CN, R 2 is Br, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.280 provides 418 compounds E280.1 to E280.418 of formula (Ia) wherein R 1a is CN, R 2 is Br, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.281 provides 418 compounds E281.1 to E281.418 of formula (Ia) wherein R 1a is CN, R 2 is Br, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.282 provides 418 compounds E282.1 to E282.418 of formula (Ia) wherein R 1a is CN, R 2 is Br, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.283 provides 418 compounds E283.1 to E283.418 of formula (Ia) wherein R 1a is CN, R 2 is CN, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.284 provides 418 compounds E284.1 to E284.418 of formula (Ia) wherein R 1a is CN, R 2 is CN, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.285 provides 418 compounds E285.1 to E285.418 of formula (Ia) wherein R 1a is CN, R 2 is CN, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.286 provides 418 compounds E286.1 to E286.418 of formula (Ia) wherein R 1a is CN, R 2 is CN, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.287 provides 418 compounds E287.1 to E287.418 of formula (Ia) wherein R 1a is CN, R 2 is CN, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.288 provides 418 compounds E288.1 to E288.418 of formula (Ia) wherein R 1a is CN, R 2 is OCH 3 , R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.289 provides 418 compounds E289.1 to E289.418 of formula (Ia) wherein R 1a is CN, R 2 is OCH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.290 provides 418 compounds E290.1 to E290.418 of formula (Ia) wherein R 1a is CN, R 2 is OCH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.291 provides 418 compounds E291.1 to E291.418 of formula (Ia) wherein R 1a is CN, R 2 is OCH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.292 provides 418 compounds E292.1 to E292.418 of formula (Ia) wherein R 1a is CN, R 2 is OCH 3 , R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.293 provides 418 compounds E293.1 to E293.418 of formula (Ia) wherein R 1a is CN, R 2 is OCH2CH3, R 5 is CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.294 provides 418 compounds E294.1 to E294.418 of formula (Ia) wherein R 1a is CN, R 2 is OCH2CH3, R 5 is OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.295 provides 418 compounds E295.1 to E295.418 of formula (Ia) wherein R 1a is CN, R 2 is OCH2CH3, R 5 is cyclopropyl and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.296 provides 418 compounds E296.1 to E296.418 of formula (Ia) wherein R 1a is CN, R 2 is OCH2CH3, R 5 is CH2OCH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Table 1.297 provides 418 compounds E297.1 to E297.418 of formula (Ia) wherein R 1a is CN, R 2 is OCH2CH3, R 5 is NHCH2CH3 and A 1 , A 2a , A 2b , A 3 , R 4 are as defined in table Z.
- Compounds according to the invention may possess any number of benefits including, inter alia, advantageous levels of biological activity for protecting plants against diseases that are caused by fungi or superior properties for use as agrochemical active ingredients (for example, greater biological activity, an advantageous spectrum of activity, an increased safety profile, improved physico-chemical properties, or increased biodegradability).
- Compounds according to the invention have particularly advantageous levels of biological activity for protecting plants against oomycetes such as Phytophthora, Plasmopara and Pythium.
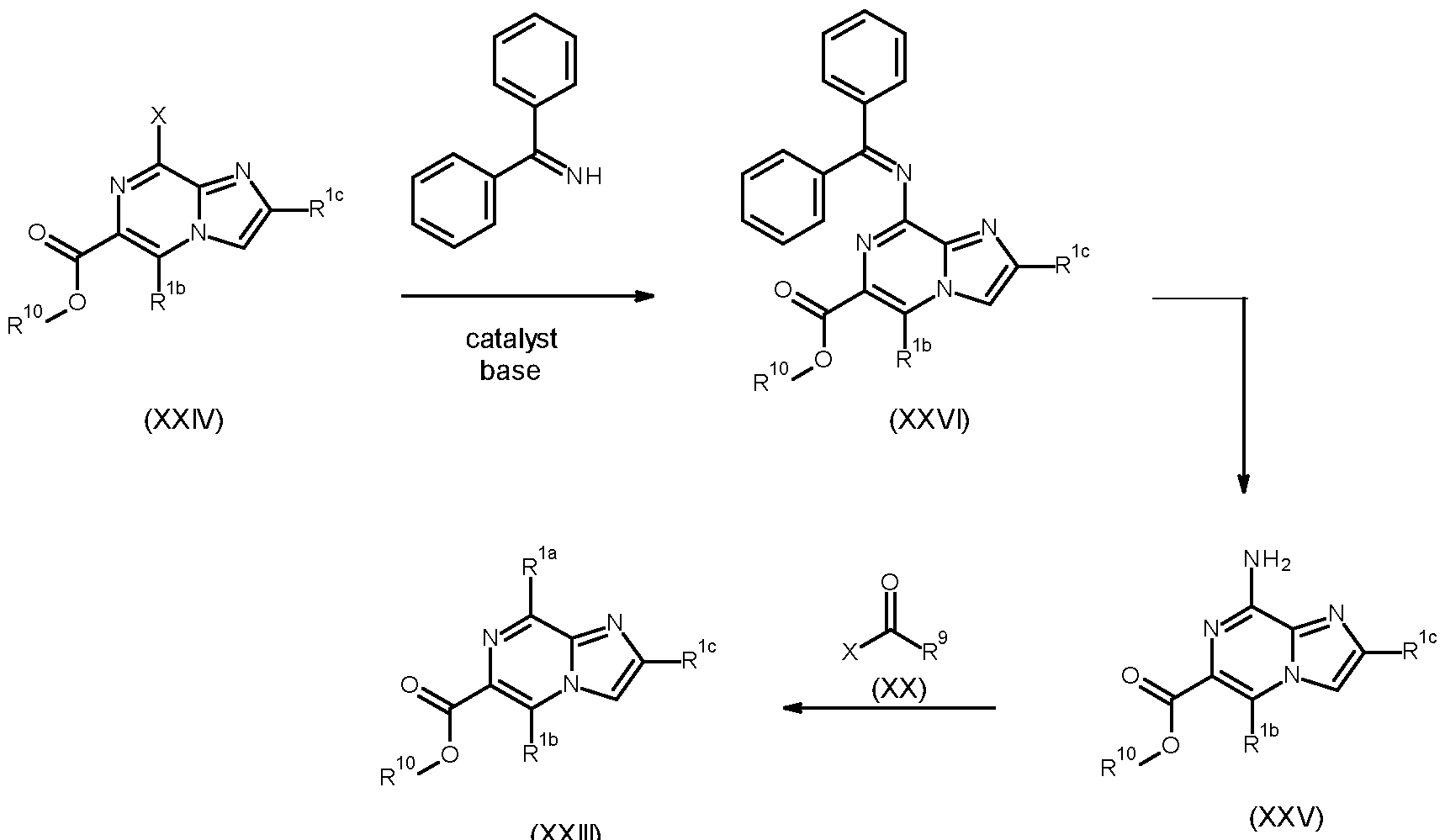
- Compounds of formula (I), wherein Z is O, can be made as shown in the following schemes 1 to 18, in which, unless otherwise stated, the definition of each variable is as defined in the present invention.
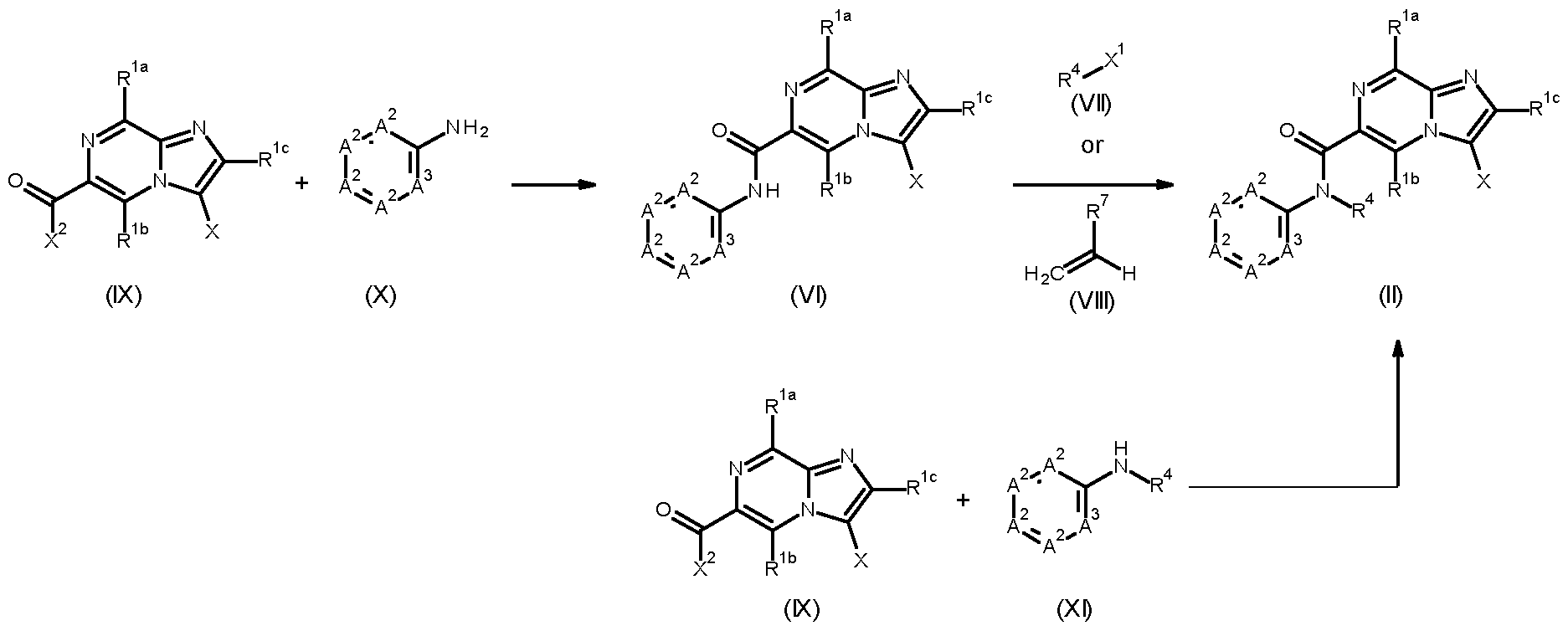
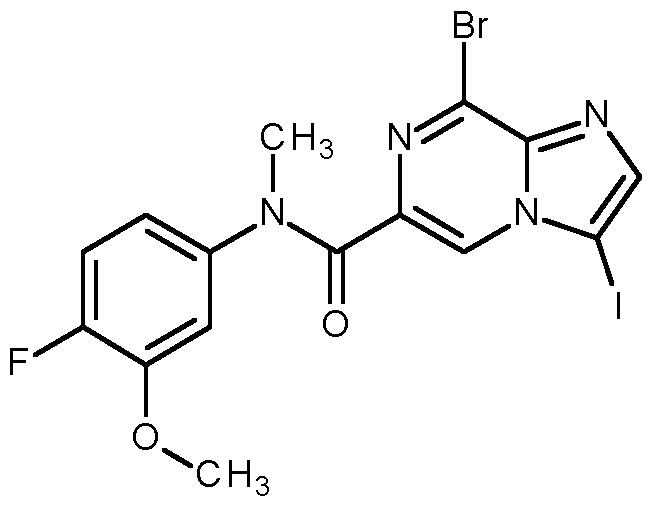
- Compounds of formula (I) can be prepared via Suzuki cross coupling of compounds of formula (II), wherein X is Cl, Br or I, and a compound of formula (III), wherein either R 6 is independently from each other hydrogen, C 1-6 alkyl or wherein two R 6 together can form a C 3-8 cycloalkyl, in the presence of a base, such as Cs2CO3, K2CO3 or NaOtBu, and a suitable palladium catalyst, such as tetrakistriphenylphosphinepalladium, [1,1-bis(diphenylphosphino)ferrocene]dichloropalladium(II), bis(diphenylphosphine)palladium(II) chloride, palladium dichloride or palladium acetate, in a suitable solvent, such as dimethylformamide, dioxane, tetrahydrofuran, ethanol or water.
- a base such as Cs2CO3, K2CO3 or Na
- a compound of formula (VIII), wherein R 7 is an electron-withdrawing group such as a cyano or an ester, can alternatively be used to functionalize the amide nitrogen, optionally in the presence of a base such as triethylamine or 1,8-diazabicyclo[5.4.0]undec-7-ene.
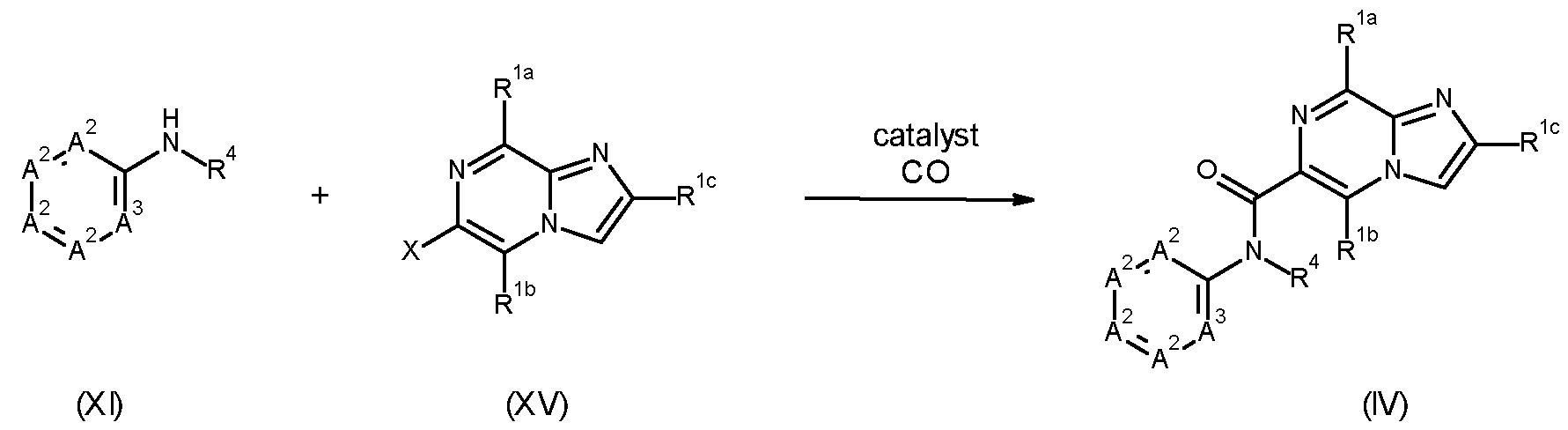
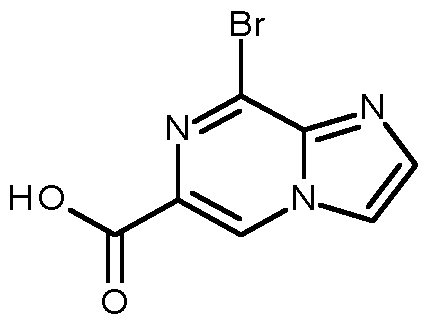
- Compounds of formula (VI), wherein X is Cl, Br or I can be prepared by a peptide-coupling reaction between a compound of formula (IX), wherein X is Cl, Br or I, and X 2 is OH, and amines of formula (X), and by activating the carboxylic acid function of the compounds of formula (IX), a process that usually takes place by converting the -OH of the carboxylic acid into a good leaving group, such as a chloride group, for example by using (COCl)2 or SOCl2, prior to treatment with amines of formula (X), preferably in a suitable solvent (e.g., N-methylpyrrolidone, acetonitrile, dimethylacetamide, dichloromethane or tetrahydrofuran), preferably at temperatures between 25°C and 60°C, and optionally in the presence of a base such as triethylamine or N,N- diisopropylethylamine; or alternatively under conditions described in
- compounds of formula (XI) can be obtained from amines of formula (X) by a reductive amination with aldehydes of formula (XII), wherein R 8 is selected from C1-5alkyl, C1-6alkoxy-C1-5alkyl, C3- 6cycloalkyl-C1-3alkyl, C1-5alkenyl, C1-5alkynyl, C1-5alkoxy, C1-6alkylsulfanyl-C1-5alkyl, C1-6alkylsulfinyl-C1- 5alkyl, C1-6alkylsulfonyl-C1-5alkyl, C1-6alkoxycarbonyl-C1-5alkyl, C1-6alkylaminocarbonyl-C1-5alkyl, diC1- 6alkylaminocarbonyl-C1-5alkyl., e.g.
- compounds of formula (II), wherein X is Cl, Br or I can be prepared from the reaction of a compound of formula (IV) and a halogenating agent, such as N-chlorosuccinimide, N-bromosuccinimide, N-iodosuccinimide or bromine in a suitable solvent, such as dichloromethane, chloroform, tetrahydrofuran or acetonitrile.
- a halogenating agent such as N-chlorosuccinimide, N-bromosuccinimide, N-iodosuccinimide or bromine
- a suitable solvent such as dichloromethane, chloroform, tetrahydrofuran or acetonitrile.
- Compounds of formula (IV) can be obtained by an amide-coupling transformation of amines of formula (XI) with compounds of formula (XIII), wherein X 2 is OH, and by activating the carboxylic acid function of the compounds of formula (XIII), a process that usually takes place by converting the -OH of the carboxylic acid into a good leaving group, such as a chloride group, for example by using (COCl)2 or SOCl2, prior to treatment with amines of formula (XI), preferably in a suitable solvent (e.g., N-methylpyrrolidone, acetonitrile, dimethylacetamide, dichloromethane or tetrahydrofuran), preferably at temperatures between 25 °C and 60 °C, and optionally in the presence of a base such as triethylamine or N,N-diisopropylethylamine; or alternatively under conditions described in the literature for an amide coupling such as 1-propanephosphonic acid cycl
- compounds of formula (IV) can be obtained by alkylation of compounds of formula (XIV) with compounds of formula (VII), wherein X 1 is a good leaving group such as Cl, Br, I, triflate, tosyl or mesyl, in the presence of a base such as Cs2CO3, K2CO3, NaH or NaOtBu.
- compounds of formula (IV) can also be synthesized by reacting compounds of formula (XIV) with compounds of formula (VIII), wherein R 7 is an electron-withdrawing group such as cyano or ester, optionally in the presence of a base such as triethylamine or 1,8-diazabicyclo[5.4.0]undec-7- ene.
- compounds of formula (IV) can be obtained from compounds of formula (XVI), wherein X is Cl, Br or I, by a cross-coupling transformation with a compound of formula (XVII), wherein either R 6 is independently from each other hydrogen, C1-6alkyl or wherein two R 6 together can form a C3-8cycloalkyl (Suzuki cross-coupling), or a compound of formula (XVIII) (Stille coupling), or by a palladium catalyzed cyanation reaction.
- Suzuki cross-couplings can be achieved by using a suitable palladium catalyst, such as tetrakistriphenylphosphinepalladium, [1,1-bis(diphenylphosphino)ferrocene]dichloropalladium(II), bis(diphenylphosphine)palladium(II) chloride, palladium dichloride or palladium acetate, in the presence of a base, such as Cs2CO3, K2CO3 or NaOtBu, in a suitable solvent, such as dimethylformamide, dioxane, tetrahydrofuran, ethanol or water.
- a suitable palladium catalyst such as tetrakistriphenylphosphinepalladium, [1,1-bis(diphenylphosphino)ferrocene]dichloropalladium(II), bis(diphenylphosphine)palladium(II) chloride, palladium dichloride or palladium
- Stille couplings can be achieved a suitable palladium catalyst such as tetrakistriphenylphosphinepalladium, tris(dibenzylideneacetone)dipalladium, palladium dichloride or palladium acetate in the presence of a base, such as Cs2CO3 or sodium acetate, in a suitable solvent, such as dimethylformamide, dioxane or acetonitrile.
- a suitable palladium catalyst such as tetrakistriphenylphosphinepalladium, tris(dibenzylideneacetone)dipalladium, palladium dichloride or palladium acetate in the presence of a base, such as Cs2CO3 or sodium acetate, in a suitable solvent, such as dimethylformamide, dioxane or acetonitrile.
- Palladium-catalyzed cyanation can be achieved by using a suitable catalyst such as tris(dibenzylidenaceton)dipalladium(0) or palladium acetate, [1,1-bis(diphenylphosphino)ferrocene]dichloropalladium(II), optionally with suitable ligands such as 5-diphenylphosphanyl-9,9-dimethylxanthen-4-yl)-diphenyl-phosphane (Xantphos), 2- Dicyclohexylphosphin-2′,4′,6′-triisopropylbiphenyl (XPhos), and a cyanating reagent such as zinc cyanide, potassium cyanide or potassium hexacyanoferrate(II) trihydrate optionally in the presence of a base such as potassium acetate or cesium carbonate, in a suitable solvent such as dimethyl formamide, dioxane, tetrahydrofuran with or without the
- Amines of formula (XIX) can be prepared by treatment of compounds of formula (XXI) with an acid, typically hydrochloric acid, or with sodium acetate and hydroxyamine hydrochloride in a protic solvent such as methanol.
- Compounds of formula (XXI) can be synthesized by reaction of compounds of formula (XVI), wherein X is Cl, Br or I, with diphenylmethanimine (CAS number 1013-88-3) in the presence of a catalyst, such as palladium acetate or tris(dibenzylideneacetone)dipalladium, optionally a ligand such as 1,1′-[1,1′-binaphthalene]-2,2′- diylbis[1,1-diphenylphosphine] or (9,9-dimethyl-9H-xanthene-4,5-diyl)bis(diphenylphosphane), and in the presence of a base such as cesium carbonate or sodium tert-butoxide.
- compounds of formula (IV) may be obtained directly by reacting compounds of formula (XVI) with compounds of formula (XXII), wherein R 9 is C1-6alkyl, by a palladium catalyzed coupling reaction (Buchwald-Hartwig reaction).
- This reaction can be achieved by using a suitable catalyst such as Xantphos-Pd-G2 (CAS 1375325-77-1), XantPhos Pd G3 (Cas 1445085-97-1) or [1,1′- Bis(diphenylphosphino)ferrocen]dichlorpalladium(II), in the presence of a base such as cesium carbonate, sodium tert-butoxide or potassium phosphate in a suitable solvent such as dioxane, tetrahydrofuran or toluene.
- a suitable catalyst such as Xantphos-Pd-G2 (CAS 1375325-77-1), XantPhos Pd G3 (Cas 1445085-97-1) or [1,1′- Bis(diphenylphosphino)ferrocen]dichlorpalladium(II)
- compounds of formula (XXIII), wherein R 10 is C1-6alkyl can be obtained from compounds of formula (XXV), wherein R 10 is C1-6alkyl, by reaction with a compound of formula (XX), wherein X is Cl or Br and R 9 is C1-6alkyl, in the presence of a base such as pyridine or diisopropylethylamine.
- Amines of formula (XXV), wherein R 10 is C1-6alkyl can be prepared by treatment of compounds of formula (XXVI), wherein R 10 is C1-6alkyl, with an acid, typically hydrochloric acid, or with sodium acetate and hydroxyamine hydrochloride in a protic solvent such as methanol.
- Scheme 10 Compounds of formula (XV), wherein X is Cl, Br or I, can be prepared by cross-coupling of hydrazones of formula (XXVII), wherein X is Cl, Br or I, with a compound of formula (XXVIII), wherein either R 6 is independently from each other hydrogen, C1-6alkyl or wherein two R 6 together can form a C3-8cycloalkyl and R 11 is selected from C1-5alkyl, C1-6alkoxy-C1-5alkyl and C3-6cycloalkyl-C1-3alkyl, in the presence of a base, such as Cs2CO3, K2CO3 or 1,8-diazabicyclo[5.4.0]undec-7-ene.
- a base such as Cs2CO3, K2CO3 or 1,8-diazabicyclo[5.4.0]undec-7-ene.
- a compound of formula (XV), wherein X is Cl, Br or I can be obtained by reacting hydrazone of formula (XXVII), wherein X is Cl, Br or I, with an alkoxide of formula (XXIX), wherein R 12 is C1-5alkyl, in a protic solvent such as methanol or tert-butanol.
- Hydrazones of formula (XXVII), wherein X is Cl, Br or I can be prepared by condensation of p-toluenesulfonhydrazide (CAS 1576-35-8) with aldehydes of formula (XXX), wherein X is Cl, Br or I.
- Aldehydes of formula (XXX), wherein X is Cl, Br or I are commercially available or, alternatively, can be prepared by the condensation of a compound of formula (XXXI), wherein X is Cl, Br or I, with a compound of formula (XXXII), wherein X is Cl, Br or I, or its corresponding acetal of formula (XXXIII), wherein X is Cl, Br or I and either R 13 is independently from each other C 1-6 alkyl or wherein two R 13 together can form a C 3-8 cycloalkyl, in a solvent, such as water, ethanol, acetone or acetonitrile.
- a solvent such as water, ethanol, acetone or acetonitrile.
- the outcome of the reaction can be improved by using a base, such as sodium bicarbonate or potassium carbonate, or by using an acid, such as p-toluenesulfonic acid or hydrogen bromide.
- a base such as sodium bicarbonate or potassium carbonate
- an acid such as p-toluenesulfonic acid or hydrogen bromide.
- Compounds of formula (XXXIV), wherein X is Cl, Br or I, and X 2 is OH, can be prepared by hydrolysis of the ester function of compounds of formula (XXIV), wherein X is Cl, Br or I, and R 10 is C1-6alkyl, which in turn can be synthesized by condensation of compounds of formula (XXXV), wherein X is Cl, Br or I, and R 10 is C1-6alkyl, with a compound of formula (XXXII), wherein X is Cl, Br or I, or its corresponding acetal of formula (XXXIII), wherein X is Cl, Br or I and either R 13 is independently from each other C1-6alkyl or wherein two R 13 together can form a C3- 8 cycloalkyl, using the conditions described above.
- Compounds of formula (I) can alternatively be prepared by alkylation of compounds of formula (XXXVII) with compounds of formula (VII), wherein X 1 is a good leaving group such as Cl, Br, I, triflate, tosyl or mesyl, in the presence of a base such as Cs2CO3, K2CO3, NaH or NaOtBu.
- compounds of formula (I) can also be synthesized by reacting compounds of formula (XXXVII) with compounds of formula (VIII), wherein R 7 is an electron-withdrawing group such as cyano or ester, optionally in the presence of a base such as triethylamine or 1,8- diazabicyclo[5.4.0]undec-7-ene.
- Compounds of formula (XXXVIII), wherein R10 is C1-6alkyl can be prepared by direct arylation of compounds of formula (XXIII), wherein R 10 is C1-6alkyl, with compounds of formula (V), wherein X is Cl, Br or I, in the presence of a catalyst such as palladium diacetate, a ligand such as triphenylphosphine or (dicyclohexylphosphino)biphenyl, and a base such as cesium carbonate or potassium carbonate.
- a catalyst such as palladium diacetate
- a ligand such as triphenylphosphine or (dicyclohexylphosphino)biphenyl
- a base such as cesium carbonate or potassium carbonate.
- compounds of formula (XXXVIII), wherein R 10 is C1-6alkyl can result from a Suzuki cross-coupling reaction between compounds of formula (XXXIX), wherein X is Cl, Br or I, and R 10 is C1-6alkyl, and compounds of formula (III), wherein either R 6 is independently from each other hydrogen, C1-6alkyl or wherein two R 6 together can form a C3-8cycloalkyl, in the presence of a base, such as Cs2CO3, K2CO3 or NaOtBu, and a suitable palladium catalyst, such as tetrakistriphenylphosphinepalladium, [1,1-bis(diphenylphosphino)ferrocene]dichloropalladium(II), bis(diphenylphosphine)palladium(II) chloride, palladium dichloride or palladium acetate, in a suitable solvent, such as dimethylformamide, dioxane, tetrahydr
- compounds of formula (I) can be synthesized by reacting compounds of formula (XL), wherein X is Cl, Br or I, with amines of formula (XI) and carbon monoxide in the presence of a catalyst such as [1,1-bis(diphenylphosphino)ferrocene]dichloropalladium(II), and, optionally, a base such as triethylamine.
- a catalyst such as [1,1-bis(diphenylphosphino)ferrocene]dichloropalladium(II)
- a base such as triethylamine.
- Compounds of formula (XL), wherein X is Cl, Br or I can be prepared from compounds of formula (XLI), wherein either X is independently from each other Cl, Br or I, through a Suzuki cross- coupling with compounds of formula (III), wherein either R 6 is independently from each other hydrogen, C1-6alkyl or wherein two R 6 together can form a C3-8cycloalkyl, in the presence of a base, such as Cs2CO3, K2CO3 or NaOtBu, and a suitable palladium catalyst, such as tetrakistriphenylphosphinepalladium, [1,1-bis(diphenylphosphino)ferrocene]dichloropalladium(II), bis(diphenylphosphine)palladium(II) chloride, palladium dichloride or palladium acetate, in a suitable solvent, such as dimethylformamide, dioxane, tetrahydrofuran, ethanol or water.
- Scheme 15 compounds of formula (I) can be prepared by the reaction of a compound of formula (XLII) with compounds of formula (XLIII), wherein X 2 is OH, in a peptide-coupling transformation, using the conditions described above.
- Compounds of formula (XLII) can be prepared via Suzuki cross coupling of a compound of formula (II), wherein X is Cl, Br or I, and a compound of formula (XLIV), wherein either R 6 is independently from each other hydrogen, C1-6alkyl or wherein two R 6 together can form a C3- 8cycloalkyl, in the presence of a base, such as Cs2CO3, K2CO3 or NaOtBu, and a suitable palladium catalyst, such as tetrakistriphenylphosphinepalladium, [1,1- bis(diphenylphosphino)ferrocene]dichloropalladium(II), bis(diphenylphosphine)palladium(II) chloride, palladium dichloride or palladium acetate, in a suitable solvent, such as dimethylformamide, dioxane, tetrahydrofuran, ethanol or water.
- a base such as Cs2CO3, K
- compounds of formula (Ib), wherein Z is S can be prepared by the reaction of a compound of formula (XLIIb) with compounds of formula (XLIII), wherein X 2 is OH, in a peptide-coupling transformation, using the conditions described above.
- Compounds of formula (XLIIb) can be prepared by the reaction of a compound of formula (XLII), with phosphorus pentasulfide or Lawesson’s reagent (CAS: 19172-47-5) in a suitable solvent such as toluene, xylene or dichloromethane. This transformation is depicted in Scheme 18.
- Non-exhaustive examples include oxidation reactions, reduction reactions, hydrolysis reactions, coupling reactions, aromatic nucleophilic or electrophilic substitution reactions, nucleophilic substitution reactions, nucleophilic addition reactions, olefination reactions, oxime formation, alkylation and halogenation reactions.
- a compound according to the present invention can be converted in a manner known per se into another compound according to the present invention by replacing one or more substituents of the starting compound according to the present invention in the customary manner by (an)other substituent(s) according to the invention.
- Salts of the compounds according to the present invention can be prepared in a manner known per se.
- acid addition salts of the compounds according to the present invention are obtained by treatment with a suitable acid or a suitable ion exchanger reagent and salts with bases are obtained by treatment with a suitable base or with a suitable ion exchanger reagent.
- Salts of compounds the compounds according to the present invention can be converted in the customary manner into the free compounds, acid addition salts, for example, by treatment with a suitable basic compound or with a suitable ion exchanger reagent and salts with bases, for example, by treatment with a suitable acid or with a suitable ion exchanger reagent.
- Salts of the compounds according to the present invention can be converted in a manner known per se into other salts of the compounds according to the present invention, acid addition salts, for example, into other acid addition salts, for example by treatment of a salt of inorganic acid such as hydrochloride with a suitable metal salt such as a sodium, barium or silver salt, of an acid, for example with silver acetate, in a suitable solvent in which an inorganic salt which forms, for example silver chloride, is insoluble and thus precipitates from the reaction mixture.
- a salt of inorganic acid such as hydrochloride
- a suitable metal salt such as a sodium, barium or silver salt
- an acid for example with silver acetate
- a suitable solvent in which an inorganic salt which forms, for example silver chloride, is insoluble and thus precipitates from the reaction mixture.
- the compounds according to the present invention, which have salt-forming properties can be obtained in free form or in the form of salts.
- the compounds according to the present invention and, where appropriate, the tautomers thereof, in each case in free form or in salt form, can be present in the form of one of the stereoisomers which are possible or as a mixture of these, for example in the form of pure stereoisomers, such as antipodes and/or diastereomers, or as stereoisomer mixtures, such as enantiomer mixtures, for example racemates, diastereomer mixtures or racemate mixtures, depending on the number, absolute and relative configuration of asymmetric carbon atoms which occur in the molecule and/or depending on the configuration of non-aromatic double bonds which occur in the molecule; the invention relates to the pure stereoisomers and also to all stereoisomer mixtures which are possible and is to be understood in each case in this sense hereinabove and hereinbelow, even when stereochemical details are not mentioned specifically in each case.
- Diastereomer mixtures or racemate mixtures of the compounds according to the present invention in free form or in salt form, which can be obtained depending on which starting materials and procedures have been chosen can be separated in a known manner into the pure diastereomers or racemates on the basis of the physicochemical differences of the components, for example by fractional crystallization, distillation and/or chromatography.
- Enantiomer mixtures such as racemates, which can be obtained in a similar manner can be resolved into the optical antipodes by known methods, for example by recrystallization from an optically active solvent, by chromatography on chiral adsorbents, for example high-performance liquid chromatography (HPLC) on acetyl cellulose, with the aid of suitable microorganisms, by cleavage with specific, immobilized enzymes, via the formation of inclusion compounds, for example using chiral crown ethers, where only one enantiomer is complexed, or by conversion into diastereomeric salts, for example by reacting a basic end-product racemate with an optically active acid, such as a carboxylic acid, for example camphor, tartaric or malic acid, or sulfonic acid, for example camphorsulfonic acid, and separating the diastereomer mixture which can be obtained in this manner, for example by fractional crystallization based on their differing solubilities, to give the di
- N-oxides can be prepared by reacting a compound according to the present invention with a suitable oxidizing agent, for example the H2O2/urea adduct in the presence of an acid anhydride, e.g. trifluoroacetic anhydride.
- a suitable oxidizing agent for example the H2O2/urea adduct in the presence of an acid anhydride, e.g. trifluoroacetic anhydride.
- stereoisomer for example enantiomer or diastereomer, or stereoisomer mixture, for example enantiomer mixture or diastereomer mixture
- the individual components have a different biological activity.
- the compounds according to the present invention and, where appropriate, the tautomers thereof, in each case in free form or in salt form, can, if appropriate, also be obtained in the form of hydrates and/or include other solvents, for example those which may have been used for the crystallization of compounds which are present in solid form.
- the following Examples illustrate, but do not limit, the invention.
- the present invention also provides intermediates useful for the preparation of compounds according to the present invention. The below intermediates form a further aspect of the invention.
- R 1a is selected from hydroxy, halogen, CN, C1-6alkyl, C3-6cycloalkyl, C1-6alkoxy-C1-6alkyl, C3-6cycloalkyl- C1-4alkyl, C1-6alkylsulfanyl, C1-6alkylsulfinyl, C1-6alkylsulfonyl, C1-6alkoxy, amino, and NHC(O)C1-6alkyl; and preferably R 1a is selected from C1-6alkyl, C3-6cycloalkyl, C1-6alkoxy-C1-6alkyl, C3-6cycloalkyl-C1- 4alkyl, and C1-6alkoxy; R 1b and R 1c are independently selected from hydrogen, hydroxy, halogen, CN, C 1-6 alkyl, C 3-6 cycloalkyl, C1-6alkoxy-C1-6alkyl, C3-6cycloalkyl-C1-4alkyl, C1-6alkylsulfanyl, C
- R 1b and R 1c are independently selected from hydrogen, hydroxy, halogen, CN, C 1-6 alkyl, C 3-6 cycloalkyl, C1-6alkoxy-C1-6alkyl, C3-6cycloalkyl-C1-4alkyl, C1-6alkylsulfanyl, C1-6alkylsulfinyl, C1-6alkylsulfonyl, C1- 6alkoxy, amino, and NHC(O)C1-6alkyl; and preferably R 1b and R 1c are hydrogen;
- a 2 are independently CR 2 or N, with the proviso that no more than three A 2 are N, preferably no more than two A 2 are N, preferably no more than one A 2 is N, and more preferably the four A 2 are CR 2 ;
- R 2 are independently selected from hydrogen, hydroxy, halogen, CN, C1-6alkyl, C1-6alkoxy
- the compounds of formula (I) as defined in the present invention can be used in the agricultural sector and related fields of use e.g. as active ingredients for controlling plant pathogens or on non-living materials for control of spoilage microorganisms or organisms potentially harmful to man.
- the novel compounds are distinguished by excellent activity at low rates of application, by being well tolerated by plants and by being environmentally safe. They have very useful curative, preventive and systemic properties and may be used for protecting numerous cultivated plants.
- the compounds of formula (I) as defined in the present invention can be used to inhibit or destroy the pathogens that occur on plants or parts of plants (fruit, blossoms, leaves, stems, tubers, roots) of different crops of useful plants, while at the same time protecting also those parts of the plants that grow later e.g.
- fungicide as used herein means a compound that controls, modifies, or prevents the growth of fungi.
- fungicidally effective amount means the quantity of such a compound or combination of such compounds that is capable of producing an effect on the growth of fungi. Controlling or modifying effects include all deviation from natural development, such as killing, retardation and the like, and prevention includes barrier or other defensive formation in or on a plant to prevent fungal infection.
- compounds of formula (I) as defined in the present invention as dressing agents for the treatment of plant propagation material, e.g., seed, such as fruits, tubers or grains, or plant cuttings (for example rice), for the protection against fungal infections as well as against phytopathogenic fungi occurring in the soil.
- plant propagation material e.g., seed, such as fruits, tubers or grains, or plant cuttings (for example rice)
- the propagation material can be treated with a composition comprising a compound of formula (I) as defined in the present invention before planting: seed, for example, can be dressed before being sown.
- the compounds of formula (I) as defined in the present invention can also be applied to grains (coating), either by impregnating the seeds in a liquid formulation or by coating them with a solid formulation.
- the composition can also be applied to the planting site when the propagation material is being planted, for example, to the seed furrow during sowing.
- the invention relates also to such methods of treating plant propagation material and to the plant propagation material so treated.
- the compounds of formula (I) as defined in the present invention can be used for controlling fungi in related areas, for example in the protection of technical materials, including wood and wood related technical products, in food storage, in hygiene management.
- the invention could be used to protect non-living materials from fungal attack, e.g. lumber, wall boards and paint.
- Compounds of formula (I) as defined in the present invention and fungicidal compositions containing them may be used to control plant diseases caused by a broad spectrum of fungal plant pathogens.
- fungi and fungal vectors of disease are for example: Absidia corymbifera, Alternaria spp, Aphanomyces spp, Ascochyta spp, Aspergillus spp. including A. flavus, A. fumigatus, A. nidulans, A. niger, A. terrus, Aureobasidium spp. including A.
- Botryosphaeria spp. including B. dothidea, B. obtusa, Botrytis spp. contributing B. cinerea, Candida spp. including C. albicans, C. glabrata, C. krusei, C. lusitaniae, C. parapsilosis, C. tropicalis, Cephaloascus fragrans, Ceratocystis spp, Cercospora spp. including C.
- capsulatum Laetisaria fuciformis, Leptographium lindbergi, Leveillula taurica, Lophodermium seditiosum, Microdochium nivale, Microsporum spp, Monilinia spp, Mucor spp, Mycosphaerella spp. including M. graminicola, M. pomi, Oncobasidium theobromaeon, Ophiostoma piceae, Paracoccidioides spp, Penicillium spp. including P. digitatum, P. italicum, Petriellidium spp, Peronosclerospora spp. Including P. maydis, P.
- leucotricha Polymyxa graminis, Polymyxa betae, Pseudocercosporella herpotrichoides, Pseudomonas spp, Pseudoperonospora spp. including P. cubensis, P. humuli, Pseudopeziza tracheiphila, Puccinia Spp. including P. hordei, P. recondita, P. striiformis, P. triticina, Pyrenopeziza spp, Pyrenophora spp, Pyricularia spp. including P. oryzae, Pythium spp. including P.
- compounds of formula (I) as defined in the present invention and fungicidal compositions containing them may be used to control plant diseases caused by a broad spectrum of fungal plant pathogens in the Basidiomycete, Ascomycete, Oomycete and/or Deuteromycete, Blasocladiomycete, Chrytidiomycete, Glomeromycete and/or Mucoromycete classes. More particularly, the compounds of formula (I) as defined in the present invention may be used to conrol oomycetes.
- pathogens may include: Oomycetes, including Phytophthora diseases such as those caused by Phytophthora capsici, Phytophthora infestans, Phytophthora sojae, Phytophthora fragariae, Phytophthora nicotianae, Phytophthora cinnamomi, Phytophthora citricola, Phytophthora citrophthora and Phytophthora erythroseptica; Pythium diseases such as those caused by Pythium aphanidermatum, Pythium arrhenomanes, Pythium graminicola, Pythium irregulare, Pythium sylvaticum and Pythium ultimum; diseases caused by Peronosporales such as Peronospora destructor, Peronospora parasitica, Plasmopara viticola, Plasmopara halstedii, Pseudoperonospora cubens
- Ascomycetes including blotch, spot, blast or blight diseases and/or rots for example those caused by Pleosporales such as Stemphylium solani, Stagonospora tainanensis, Spilocaea oleaginea, Setosphaeria turcica, Pyrenochaeta lycoperisici, Pleospora herbarum, Phoma destructiva, Phaeosphaeria herpotrichoides, Phaeocryptocus gaeumannii, Ophiosphaerella graminicola, Ophiobolus graminis, Leptosphaeria maculans, Hendersonia creberrima, Helminthosporium triticirepentis, Setosphaeria turcica, Drechslera glycines, Didymella bryoniae, Cycloconium oleagineum, Corynespora cassiicola, Cochliobolus sativus, Bi
- Gerlachia nivale Gibberella fujikuroi
- Gibberella zeae Gibberella zeae
- Gliocladium spp. Myrothecium verrucaria
- Nectria ramulariae Trichoderma viride
- Trichothecium roseum Trichothecium roseum
- Verticillium theobromae Myrothecium verrucaria
- Basidiomycetes including smuts for example those caused by Ustilaginales such as Ustilaginoidea virens, Ustilago nuda, Ustilago tritici, Ustilago zeae, rusts for example those caused by Pucciniales such as Cerotelium fici, Chrysomyxa arctostaphyli, Coleosporium ipomoeae, Hemileia vastatrix, Puccinia arachidis, Puccinia cacabata, Puccinia graminis, Puccinia recondita, Puccinia sorghi, Puccinia hordei, Puccinia striiformis f.sp.
- Ustilaginales such as Ustilaginoidea virens, Ustilago nuda, Ustilago tritici, Ustilago zeae
- rusts for example those caused by Pucciniales such as Cerotelium fici, Chr
- Puccinia striiformis f.sp. Secalis Pucciniastrum coryli, or Uredinales such as Cronartium ribicola, Gymnosporangium juniperi-viginianae, Melampsora medusae, Phakopsora pachyrhizi, Phragmidium mucronatum, Physopella ampelosidis, Tranzschelia discolor and Uromyces viciae-fabae; and other rots and diseases such as those caused by Cryptococcus spp., Exobasidium vexans, Marasmiellus inoderma, Mycena spp., Sphacelotheca reiliana, Typhula ishikariensis, Urocystis agropyri, Itersonilia perplexans, Corticium invisum, Laetisaria fuciformis, Waitea circinata, Rhizoctonia solani, Tha
- Blastocladiomycetes such as Physoderma maydis. Mucoromycetes, such as Choanephora cucurbitarum.; Mucor spp.; Rhizopus arrhizus. As well as diseases caused by other species and genera closely related to those listed above.
- the compounds and compositions comprising compounds of formula (I) as defined in the present invention may also have activity against bacteria such as Erwinia amylovora, Erwinia caratovora, Xanthomonas campestris, Pseudomonas syringae, Strptomyces scabies and other related species as well as certain protozoa.
- target crops and/or useful plants to be protected typically comprise perennial and annual crops, such as berry plants for example blackberries, blueberries, cranberries, raspberries and strawberries; cereals for example barley, maize (corn), millet, oats, rice, rye, sorghum triticale and wheat; fibre plants for example cotton, flax, hemp, jute and sisal; field crops for example sugar and fodder beet, coffee, hops, mustard, oilseed rape (canola), poppy, sugar cane, sunflower, tea and tobacco; fruit trees for example apple, apricot, avocado, banana, cherry, citrus, nectarine, peach, pear and plum; grasses for example Bermuda grass, bluegrass, bentgrass, centipede grass, fescue, ryegrass, St.
- perennial and annual crops such as berry plants for example blackberries, blueberries, cranberries, raspberries and strawberries
- cereals for example barley, maize (corn), millet, oats
- Augustine grass and Zoysia grass herbs such as basil, borage, chives, coriander, lavender, lovage, mint, oregano, parsley, rosemary, sage and thyme; legumes for example beans, lentils, peas and soya beans; nuts for example almond, cashew, ground nut, hazelnut, peanut, pecan, pistachio and walnut; palms for example oil palm; ornamentals for example flowers, shrubs and trees; other trees, for example cacao, coconut, olive and rubber; vegetables for example asparagus, aubergine, broccoli, cabbage, carrot, cucumber, garlic, lettuce, marrow, melon, okra, onion, pepper, potato, pumpkin, rhubarb, spinach and tomato; and vines for example grapes.
- herbs such as basil, borage, chives, coriander, lavender, lovage, mint, oregano, parsley, rosemary, sage and thyme
- legumes for example beans, lentils, peas and soya beans
- the useful plants and / or target crops in accordance with the invention include conventional as well as genetically enhanced or engineered varieties such as, for example, insect resistant (e.g. Bt. and VIP varieties) as well as disease resistant, herbicide tolerant (e.g. glyphosate- and glufosinate-resistant maize varieties commercially available under the trade names RoundupReady® and LibertyLink®) and nematode tolerant varieties.
- suitable genetically enhanced or engineered crop varieties include the Stoneville 5599BR cotton and Stoneville 4892BR cotton varieties.
- useful plants and/or “target crops” is to be understood as including also useful plants that have been rendered tolerant to herbicides like bromoxynil or classes of herbicides (such as, for example, HPPD inhibitors, ALS inhibitors, for example primisulfuron, prosulfuron and trifloxysulfuron, EPSPS (5- enol-pyrovyl-shikimate-3-phosphate-synthase) inhibitors, GS (glutamine synthetase) inhibitors or PPO (protoporphyrinogen-oxidase) inhibitors) as a result of conventional methods of breeding or genetic engineering.
- herbicides like bromoxynil or classes of herbicides
- EPSPS 5- enol-pyrovyl-shikimate-3-phosphate-synthase
- GS glutamine synthetase
- PPO protoporphyrinogen-oxidase
- An example of a crop that has been rendered tolerant to imidazolinones, e.g. imazamox, by conventional methods of breeding (mutagenesis) is Clearfield® summer rape (Canola).
- crops that have been rendered tolerant to herbicides or classes of herbicides by genetic engineering methods include glyphosate- and glufosinate-resistant maize varieties commercially available under the trade names RoundupReady® , Herculex I® and LibertyLink®.
- the term "useful plants" and/or “target crops” is to be understood as including those which naturally are or have been rendered resistant to harmful insects.
- toxins which can be expressed include ⁇ -endotoxins, vegetative insecticidal proteins (Vip), insecticidal proteins of bacteria colonising nematodes, and toxins produced by scorpions, arachnids, wasps and fungi.
- An example of a crop that has been modified to express the Bacillus thuringiensis toxin is the Bt maize KnockOut ⁇ (Syngenta Seeds).
- VipCot ⁇ Surgera Seeds
- Crops or seed material thereof can also be resistant to multiple types of pests (so-called stacked transgenic events when created by genetic modification).
- a plant can have the ability to express an insecticidal protein while at the same time being herbicide tolerant, for example Herculex I ⁇ (Dow AgroSciences, Pioneer Hi-Bred International).
- useful plants and/or “target crops” is to be understood as including also useful plants which have been so transformed by the use of recombinant DNA techniques that they are capable of synthesising antipathogenic substances having a selective action, such as, for example, the so-called "pathogenesis-related proteins" (PRPs, see e.g. EP-A-0392225).
- PRPs pathogenesis-related proteins
- Examples of such antipathogenic substances and transgenic plants capable of synthesising such antipathogenic substances are known, for example, from EP-A-0392225, WO 95/33818, and EP-A-0353191.
- the methods of producing such transgenic plants are generally known to the person skilled in the art and are described, for example, in the publications mentioned above.
- Toxins that can be expressed by transgenic plants include, for example, insecticidal proteins from Bacillus cereus or Bacillus popilliae; or insecticidal proteins from Bacillus thuringiensis, such as ⁇ - endotoxins, e.g. Cry1Ab, Cry1Ac, Cry1F, Cry1Fa2, Cry2Ab, Cry3A, Cry3Bb1 or Cry9C, or vegetative insecticidal proteins (Vip), e.g. Vip1, Vip2, Vip3 or Vip3A; or insecticidal proteins of bacteria colonising nematodes, for example Photorhabdus spp.
- insecticidal proteins from Bacillus cereus or Bacillus popilliae such as ⁇ - endotoxins, e.g. Cry1Ab, Cry1Ac, Cry1F, Cry1Fa2, Cry2Ab, Cry3A, Cry3Bb1 or Cry9C, or vegetative
- Xenorhabdus spp. such as Photorhabdus luminescens, Xenorhabdus nematophilus
- toxins produced by animals such as scorpion toxins, arachnid toxins, wasp toxins and other insect-specific neurotoxins
- toxins produced by fungi such as Streptomycetes toxins, plant lectins, such as pea lectins, barley lectins or snowdrop lectins
- agglutinins proteinase inhibitors, such as trypsin inhibitors, serine protease inhibitors, patatin, cystatin, papain inhibitors
- ribosome- inactivating proteins (RIP) such as ricin, maize-RIP, abrin, luffin, saporin or bryodin
- steroid metabolism enzymes such as 3-hydroxysteroidoxidase, ecdysteroid-UDP-glycosyl-transferase, cholesterol oxidases, ecd
- ⁇ -endotoxins for example Cry1Ab, Cry1Ac, Cry1F, Cry1Fa2, Cry2Ab, Cry3A, Cry3Bb1 or Cry9C, or vegetative insecticidal proteins (Vip), for example Vip1, Vip2, Vip3 or Vip3A
- Vip vegetative insecticidal proteins
- Hybrid toxins are produced recombinantly by a new combination of different domains of those proteins (see, for example, WO 02/15701).
- Truncated toxins for example a truncated Cry1Ab, are known.
- modified toxins one or more amino acids of the naturally occurring toxin are replaced.
- preferably non-naturally present protease recognition sequences are inserted into the toxin, such as, for example, in the case of Cry3A055, a cathepsin-G- recognition sequence is inserted into a Cry3A toxin (see WO03/018810).
- More examples of such toxins or transgenic plants capable of synthesising such toxins are disclosed, for example, in EP-A-0374753, WO93/07278, WO95/34656, EP-A-0427529, EP-A-451878 and WO03/052073.
- transgenic plants are generally known to the person skilled in the art and are described, for example, in the publications mentioned above.
- CryI-type deoxyribonucleic acids and their preparation are known, for example, from WO 95/34656, EP-A-0367 474, EP-A-0401979 and WO 90/13651.
- the toxin contained in the transgenic plants imparts to the plants tolerance to harmful insects.
- Such insects can occur in any taxonomic group of insects, but are especially commonly found in the beetles (Coleoptera), two-winged insects (Diptera) and butterflies (Lepidoptera).
- Transgenic plants containing one or more genes that code for an insecticidal resistance and express one or more toxins are known and some of them are commercially available. Examples of such plants are: YieldGard ⁇ (maize variety that expresses a Cry1Ab toxin); YieldGard Rootworm ⁇ (maize variety that expresses a Cry3Bb1 toxin); YieldGard Plus ⁇ (maize variety that expresses a Cry1Ab and a Cry3Bb1 toxin); Starlink ⁇ (maize variety that expresses a Cry9C toxin); Herculex I ⁇ (maize variety that expresses a Cry1Fa2 toxin and the enzyme phosphinothricine N-acetyltransferase (PAT) to achieve tolerance to the herbicide glufosinate ammonium); NuCOTN 33B ⁇ (cotton variety that expresses a Cry1Ac toxin); Bollgard I
- transgenic crops are: 1. Bt11 Maize from Syngenta Seeds SAS, Chemin de l'Hobit 27, F-31 790 St. Sauveur, France, registration number C/FR/96/05/10. Genetically modified Zea mays which has been rendered resistant to attack by the European corn borer (Ostrinia nubilalis and Sesamia nonagrioides) by transgenic expression of a truncated Cry1Ab toxin. Bt11 maize also transgenically expresses the enzyme PAT to achieve tolerance to the herbicide glufosinate ammonium. 2. Bt176 Maize from Syngenta Seeds SAS, Chemin de l'Hobit 27, F-31790 St.
- This toxin is Cry3A055 modified by insertion of a cathepsin-G- protease recognition sequence.
- the preparation of such transgenic maize plants is described in WO 03/018810.
- MON 863 Maize from Monsanto Europe S.A. 270-272 Avenue de Tervuren, B-1150 Brussels, Belgium, registration number C/DE/02/9.
- MON 863 expresses a Cry3Bb1 toxin and has resistance to certain Coleoptera insects. 5.
- NK603 ⁇ MON 810 Maize transgenically expresses the protein CP4 EPSPS, obtained from Agrobacterium sp. strain CP4, which imparts tolerance to the herbicide Roundup® (contains glyphosate), and also a Cry1Ab toxin obtained from Bacillus thuringiensis subsp. kurstaki which brings about tolerance to certain Lepidoptera, include the European corn borer.
- locus as used herein means fields in or on which plants are growing, or where seeds of cultivated plants are sown, or where seed will be placed into the soil. It includes soil, seeds, and seedlings, as well as established vegetation.
- plants refers to all physical parts of a plant, including seeds, seedlings, saplings, roots, tubers, stems, stalks, foliage, and fruits.
- plant propagation material is understood to denote generative parts of the plant, such as seeds, which can be used for the multiplication of the latter, and vegetative material, such as cuttings or tubers, for example potatoes.
- seeds in the strict sense
- Germinated plants and young plants which are to be transplanted after germination or after emergence from the soil, may also be mentioned. These young plants may be protected before transplantation by a total or partial treatment by immersion.
- plant propagation material is understood to denote seeds.
- Pesticidal agents referred to herein using their common name are known, for example, from "The Pesticide Manual", 19th Ed., British Crop Protection Council 2021.
- the compounds of formula (I) as defined in the present invention may be used in unmodified form or, preferably, together with the adjuvants conventionally employed in the art of formulation. To this end they may be conveniently formulated in known manner to emulsifiable concentrates, coatable pastes, directly sprayable or dilutable solutions or suspensions, dilute emulsions, wettable powders, soluble powders, dusts, granulates, and also encapsulations e.g. in polymeric substances.
- compositions may also contain further adjuvants such as stabilizers, antifoams, viscosity regulators, binders or tackifiers as well as fertilizers, micronutrient donors or other formulations for obtaining special effects.
- Suitable carriers and/or adjuvants e.g. for agricultural use, can be solid or liquid and are substances useful in formulation technology, e.g. natural or regenerated mineral substances, solvents, dispersants, wetting agents, tackifiers, thickeners, binders or fertilizers.
- Suspension concentrates are aqueous formulations in which finely divided solid particles of the active compound are suspended.
- Such formulations include anti-settling agents and dispersing agents and may further include a wetting agent to enhance activity as well an anti-foam and a crystal growth inhibitor.
- these concentrates are diluted in water and normally applied as a spray to the area to be treated.
- the amount of active ingredient may range from 0.5% to 95% of the concentrate.
- Wettable powders are in the form of finely divided particles which disperse readily in water or other liquid carriers. The particles contain the active ingredient retained in a solid matrix.
- Typical solid matrices include fuller’s earth, kaolin clays, silicas and other readily wet organic or inorganic solids.
- Wettable powders normally contain from 5% to 95% of the active ingredient plus a small amount of wetting, dispersing or emulsifying agent.
- Emulsifiable concentrates are homogeneous liquid compositions dispersible in water or other liquid and may consist entirely of the active compound with a liquid or solid emulsifying agent, or may also contain a liquid carrier, such as xylene, heavy aromatic naphthas, isophorone and other non-volatile organic solvents. In use, these concentrates are dispersed in water or other liquid and normally applied as a spray to the area to be treated.
- the amount of active ingredient may range from 0.5% to 95% of the concentrate.
- Granular formulations include both extrudates and relatively coarse particles and are usually applied without dilution to the area in which treatment is required.
- Typical carriers for granular formulations include sand, fuller’s earth, attapulgite clay, bentonite clays, montmorillonite clay, vermiculite, perlite, calcium carbonate, brick, pumice, pyrophyllite, kaolin, dolomite, plaster, wood flour, ground corn cobs, ground peanut hulls, sugars, sodium chloride, sodium sulphate, sodium silicate, sodium borate, magnesia, mica, iron oxide, zinc oxide, titanium oxide, antimony oxide, cryolite, gypsum, diatomaceous earth, calcium sulphate and other organic or inorganic materials which absorb or which can be coated with the active compound.
- Granular formulations normally contain 5% to 25% of active ingredients which may include surface-active agents such as heavy aromatic naphthas, kerosene and other petroleum fractions, or vegetable oils; and/or stickers such as dextrins, glue or synthetic resins.
- Dusts are free-flowing admixtures of the active ingredient with finely divided solids such as talc, clays, flours and other organic and inorganic solids which act as dispersants and carriers.
- Microcapsules are typically droplets or granules of the active ingredient enclosed in an inert porous shell which allows escape of the enclosed material to the surroundings at controlled rates. Encapsulated droplets are typically 1 to 50 microns in diameter.
- the enclosed liquid typically constitutes 50 to 95% of the weight of the capsule and may include solvent in addition to the active compound.
- Encapsulated granules are generally porous granules with porous membranes sealing the granule pore openings, retaining the active species in liquid form inside the granule pores.
- Granules typically range from 1 millimetre to 1 centimetre and preferably 1 to 2 millimetres in diameter.
- Granules are formed by extrusion, agglomeration or prilling, or are naturally occurring. Examples of such materials are vermiculite, sintered clay, kaolin, attapulgite clay, sawdust and granular carbon.
- Shell or membrane materials include natural and synthetic rubbers, cellulosic materials, styrene-butadiene copolymers, polyacrylonitriles, polyacrylates, polyesters, polyamides, polyureas, polyurethanes and starch xanthates.
- Other useful formulations for agrochemical applications include simple solutions of the active ingredient in a solvent in which it is completely soluble at the desired concentration, such as acetone, alkylated naphthalenes, xylene and other organic solvents.
- Pressurised sprayers wherein the active ingredient is dispersed in finely-divided form as a result of vaporisation of a low boiling dispersant solvent carrier, may also be used.
- Liquid carriers that can be employed include, for example, water, toluene, xylene, petroleum naphtha, crop oil, acetone, methyl ethyl ketone, cyclohexanone, acetic anhydride, acetonitrile, acetophenone, amyl acetate, 2-butanone, chlorobenzene, cyclohexane, cyclohexanol, alkyl acetates, diacetonalcohol, 1,2-dichloropropane, diethanolamine, p-diethylbenzene, diethylene glycol, diethylene glycol abietate, diethylene glycol butyl ether, diethylene glycol ethyl ether, diethylene glycol methyl ether, N,N-dimethyl formamide, dimethyl sulfoxide,
- Water is generally the carrier of choice for the dilution of concentrates.
- suitable solid carriers include, for example, talc, titanium dioxide, pyrophyllite clay, silica, attapulgite clay, kieselguhr, chalk, diatomaxeous earth, lime, calcium carbonate, bentonite clay, fuller’s earth, cotton seed hulls, wheat flour, soybean flour, pumice, wood flour, walnut shell flour and lignin.
- a broad range of surface-active agents are advantageously employed in both said liquid and solid compositions, especially those designed to be diluted with carrier before application. These agents, when used, normally comprise from 0.1% to 15% by weight of the formulation.
- Typical surface active agents include salts of alkyl sulfates, such as diethanolammonium lauryl sulphate; alkylarylsulfonate salts, such as calcium dodecylbenzenesulfonate; alkylphenol-alkylene oxide addition products, such as nonylphenol-C.sub.18 ethoxylate; alcohol-alkylene oxide addition products, such as tridecyl alcohol-C.sub.
- soaps such as sodium stearate
- alkylnaphthalenesulfonate salts such as sodium dibutylnaphthalenesulfonate
- dialkyl esters of sulfosuccinate salts such as sodium di(2-ethylhexyl) sulfosuccinate
- sorbitol esters such as sorbitol oleate
- quaternary amines such as lauryl trimethylammonium chloride
- polyethylene glycol esters of fatty acids such as polyethylene glycol stearate
- block copolymers of ethylene oxide and propylene oxide and salts of mono and dialkyl phosphate esters.
- compositions of the invention include crystallisation inhibitors, viscosity modifiers, suspending agents, spray droplet modifiers, pigments, antioxidants, foaming agents, anti- foaming agents, light-blocking agents, compatibilizing agents, antifoam agents, sequestering agents, neutralising agents and buffers, corrosion inhibitors, dyes, odorants, spreading agents, penetration aids, micronutrients, emollients, lubricants and sticking agents.
- biocidally active ingredients or compositions may be combined with the compositions of the invention and used in the methods of the invention and applied simultaneously or sequentially with the compositions of the invention.
- these further active ingredients may be formulated together with the compositions of the invention or mixed in, for example, the spray tank.
- These further biocidally active ingredients may be fungicides, herbicides, insecticides, bactericides, acaricides, nematicides, plant growth regulators, and/or biologicals.
- TX means “one compound selected from the compounds defined in the Tables 1.1 to 1.297 and Table A): (7E,9Z)-dodeca-7,9-dien-1-yl acetate + TX, (9Z,11E)-tetradeca-9,11-dien-1-yl acetate + TX, (9Z,12E)- tetradeca-9,12-dien-1-yl acetate + TX, (E)-6-methylhept-2-en-4-ol + TX, (E)-dec-5-en-1-yl acetate with (E)-dec-5-en-1-ol + TX, (E)-tridec-4-en-1-yl acetate + TX, (E,Z)-tetradeca-4,10-dien-1-yl acetate + TX, (Z)-dodec-7-en
- Israelensis + TX Bacillus thuringiensis subsp. Japonensis + TX, Bacillus thuringiensis subsp. Kurstaki + TX, Bacillus thuringiensis subsp. Tenebrionis + TX, Bacillus thuringiensis subspec.
- lecontei NPV + TX nickel bis(dimethyldithiocarbamate) + TX, niclosamide + TX, niclosamide-olamine + TX, nicofluprole + TX, nitenpyram + TX, nithiazine + TX, nitrapyrin + TX, octadeca-2,13-dien-1-yl acetate + TX, octadeca-3,13-dien-1-yl acetate + TX, octhilinone + TX, omethoate + TX, orfralure + TX, Orius spp.
- TX trifenmorph + TX, trifluenfuronate + TX, triflumezopyrim + TX, trimedlure + TX, trimedlure A + TX, trimedlure B1 + TX, trimedlure B2 + TX, trimedlure C + TX, trimethacarb + TX, triphenyltin acetate + TX, triphenyltin hydroxide + TX, trunc-call + TX, tyclopyrazoflor + TX, Typhlodromus occidentalis + TX, uredepa + TX, Verticillium lecanii + TX, Verticillium spp.
- acridum + TX Metarhizium anisopliae var. anisopliae + TX, metarylpicoxamid + TX, metconazole + TX, metepa + TX, methacrifos + TX, methanesulfonyl fluoride + TX, methasulfocarb + TX, methiotepa + TX, methocrotophos + TX, methoprene + TX, methoquin-butyl + TX, methothrin + TX, methoxychlor + TX, methyl (Z)-2-(5- cyclohexyl-2-methyl-phenoxy)-3-methoxy-prop-2-enoate + TX, methyl (Z)-2-(5-cyclopentyl-2-methyl- phenoxy)-3-methoxy-prop-2-enoate (these compounds may be prepared from the methods described in WO2020/193387) + TX,
- Bacillus subtilis strain AQ178 + TX Bacillus subtilis strain AQ153 + TX, Bacillus subtilis strain AQ743 + TX, Bacillus subtilis strain QST 713 (CEASE®, Serenade®, Rhapsody®) + TX, Bacillus subtilis strain QST 714 (JAZZ®) + TX, Bacillus subtilis strain QST3002 + TX, Bacillus subtilis strain QST3004 + TX, Bacillus subtilis var.
- amyloliquefaciens strain FZB24 (Taegro®, Rhizopro®) + TX, Bacillus thuringiensis aizawai GC 91 (Agree®) + TX, Bacillus thuringiensis Cry 2Ae + TX, Bacillus thuringiensis Cry1Ab + TX, Bacillus thuringiensis israelensis (BMP123®, Aquabac®, VectoBac®) + TX, Bacillus thuringiensis kurstaki (Javelin®, Deliver®, CryMax®, Bonide®, Scutella WP®, Turilav WP ®, Astuto®, Dipel WP®, Biobit®, Foray®) + TX, Bacillus thuringiensis kurstaki BMP 123 (Baritone®) + TX, Bacillus thuringiensis kurstaki HD-1 (Bioprotec-CAF / 3P®) + TX
- TX Botrytis cineria + TX, Bradyrhizobium japonicum (TerraMax®) + TX, Brevibacillus brevis + TX, Burkholderia cepacia (Deny®, Intercept®, Blue Circle®) + TX, Burkholderia gladii + TX, Burkholderia gladioli + TX, Burkholderia spp.
- TX Canadian thistle fungus (CBH Canadian Bioherbicide®) + TX, Candida butyri + TX, Candida famata + TX, Candida fructus + TX, Candida glabrata + TX, Candida guilliermondii + TX, Candida melibiosica + TX, Candida oleophila strain O + TX, Candida parapsilosis + TX, Candida pelliculosa + TX, Candida pulcherrima + TX, Candida reuêtii + TX, Candida saitoana (Bio-Coat®, Biocure®) + TX, Candida sake + TX, Candida spp.
- TX Cladosporium tenuissimum + TX, Clonostachys rosea (EndoFine®) + TX, Colletotrichum acutatum + TX, Coniothyrium minitans (Cotans WG®) + TX, Coniothyrium spp.
- TX Filobasidium floriforme + TX, Fusarium acuminatum + TX, Fusarium chlamydosporum + TX, Fusarium oxysporum (Fusaclean®, Biofox C®) + TX, Fusarium proliferatum + TX, Fusarium spp. + TX, Galactomyces geotrichum + TX, Gliocladium catenulatum (Primastop®, Prestop®) + TX, Gliocladium roseum + TX, Gliocladium spp.
- TX Lagenidium giganteum (Laginex®) + TX, Lecanicillium lecanii (formerly known as Verticillium lecanii (Mycotal®) conidia of strain KV01 (e.g. Vertalec® by Koppert/Arysta) + TX, Lecanicillium longisporum (Vertiblast®) + TX, Lecanicillium muscarium (Vertikil®) + TX, Lymantria Dispar nucleopolyhedrosis virus (Disparvirus®) + TX, Marinococcus halophilus + TX, Meira geulakonigii + TX, Metarhizium anisopliae (Destruxin WP®) + TX, Metarhizium anisopliae (Met52®) + TX, Metschnikowia fruticola (Shemer®) + TX, Metschnikowia pulcherrima + TX, Microdochium dimerum (Antibot®) + TX
- NRRL 305408 + TX, Mycorrhizae spp. (AMykor®, Root Maximizer®) + TX, Myrothecium verrucaria strain AARC-0255 (DiTera®, BROS PLUS®) + TX, Ophiostoma piliferum strain D97 (Sylvanex®) + TX, Paecilomyces farinosus + TX, Paecilomyces lilacinus strain 251 (MeloCon WG®) + TX, Paecilomyces linacinus (Biostat WP®) + TX, Paenibacillus polymyxa + TX, Pantoea agglomerans (BlightBan C9-1®) + TX, Pantoea spp.
- TX Pasteuria nishizawae in particular strain Pn1 (CLARIVA from Syngenta/ChemChina); + TX, Pasteuria spp. (Econem®) + TX, Penicillium aurantiogriseum + TX, Penicillium billai (Jumpstart®, TagTeam®) + TX, Penicillium brevicompactum + TX, Penicillium frequentans + TX, Penicillium griseofulvum + TX, Penicillium purpurogenum + TX, Penicillium spp.
- TX Penicillium viridicatum + TX, Phlebiopsis gigantean (Rotstop®) + TX, phosphate solubilizing bacteria (Phosphomeal®) + TX, Phytophthora cryptogea + TX, Phytophthora palmivora (Devine®) + TX, Pichia anomala + TX, Pichia guilliermondii + TX, Pichia membranaefaciens + TX, Pichia onychis + TX, Pichia stipites + TX, Pseudomonas aeruginosa + TX, Pseudomonas aureofasciens (Spot-Less Biofungicide®) + TX, Pseudomonas cepacia + TX, Pseudomonas chlororaphis (AtEze®) + TX, Pseudomonas corrugate + TX, P
- TX Pseudomonas syringae (Bio-Save®) + TX, Pseudomonas viridiflava + TX, Pseudozyma flocculosa strain PF-A22 UL (Sporodex L®) + TX, Puccinia canaliculata + TX, Puccinia thlaspeos (Wood Warrior®) + TX, Pythium paroecandrum + TX, Pythium oligandrum (Polygandron®, Polyversum®) + TX, Pythium periplocum + TX, Rhanella aquatilis + TX, Rhanella spp.
- Rhizobia Distal®, Vault®
- Rhizoctonia + TX Rhodococcus globerulus strain AQ719 + TX, Rhodosporidium diobovatum + TX, Rhodosporidium toruloides + TX, Rhodotorula glutinis + TX, Rhodotorula graminis + TX, Rhodotorula mucilagnosa + TX, Rhodotorula rubra + TX, Rhodotorula spp.
- Trichoderma asperellum T34 Biocontrol®
- TX Trichoderma atroviride
- Trichoderma gamsii TX
- Trichoderma hamatum TH 382 + TX Trichoderma harzianum rifai (Mycostar®) + TX
- Trichoderma harzianum T-22 Trianum- P®, PlantShield HC®, RootShield®, Trianum-G® + TX, Trichoderma harzianum T-39 (Trichodex®) + TX, Trichoderma inhamatum + TX, Trichoderma koningii + TX, Trichoderma lignorum + TX, Trichoderma longibrachiatum + TX, Trichoderma polysporum (Binab T®) + TX, Trichoderma spp.
- LC 52 (Sentinel®) + TX, Trichoderma taxi + TX, Trichoderma virens (formerly Gliocladium virens GL-21) (SoilGuard®) + TX, Trichoderma virens + TX, Trichoderma viride + TX, Trichoderma viride strain ICC 080 (Remedier®) + TX, Trichosporon pullulans + TX, Trichosporon spp. + TX, Trichothecium roseum + TX, Trichothecium spp.
- TX maydis + TX, various bacteria and supplementary micronutrients (Natural II®) + TX, various fungi (Millennium Microbes®) + TX, Verticillium chlamydosporium + TX, Vip3Aa20 (VIPtera®) + TX, Virgibaclillus marismortui + TX, Xanthomonas campestris pv.
- TX Bombus terrestris (Beeline®, Tripol®) + TX, Bombus terrestris (Natupol Beehive®) + TX, Cephalonomia stephanoderis + TX, Chilocorus nigritus + TX, Chrysoperla carnea (Chrysoline®, Chrysopa®) + TX, Chrysoperla rufilabris + TX, Cirrospilus ingenuus + TX, Cirrospilus quadristriatus + TX, Citrostichus phyllocnistoides + TX, Closterocerus chamaeleon + TX, Closterocerus spp.
- TX Coccidoxenoides perminutus (Planopar®) + TX, Coccophagus cowperi + TX, Coccophagus lycimnia + TX, Cotesia flavipes + TX, Cotesia plutellae + TX, Cryptolaemus montrouzieri (Cryptobug®, Cryptoline®) + TX, Cybocephalus nipponicus + TX, Dacnusa sibirica (Minusa®, DacDigline®, Minex®) + TX, Delphastus catalinae (Delphastus®) + TX, Delphastus pusillus + TX, Diachasmimorpha krausii + TX, Diachasmimorpha longicaudata + TX, Diaparsis jucunda + TX, Diaphorencyrtus aligarhensis + TX, Diglyphus isaea (Diminex®, Miglyphus,
- TX Steinernematid spp. (Guardian Nematodes®) + TX, Stethorus punctillum (Stethorus®) + TX, Tamarixia radiate + TX, Tetrastichus setifer + TX, Thripobius semiluteus + TX, Torymus sinensis + TX, Trichogramma brassicae (Tricholine b®) + TX, Trichogramma brassicae (Tricho-Strip®) + TX, Trichogramma evanescens + TX, Trichogramma minutum + TX, Trichogramma ostriniae + TX, Trichogramma platneri + TX, Trichogramma pretiosum + TX, Xanthopimpla stemmator + TX; abscisic acid + TX, Aminomite® + TX, BioGain® + TX, bioSea® + TX, CAS Number: 2643947-26-4 + TX, Chondroster
- NCAIM (P) B001389) (WO 2013/034938) from Certis USA LLC + TX
- Bacillus pumilus in particular strain BU F-33, having NRRL Accession No. 50185 (CARTISSA® from BASF, EPA Reg. No.71840-19) + TX
- Bacillus subtilis CX-9060 from Certis USA LLC
- Bacillus sp. in particular strain D747 (available as DOUBLE NICKEL® from Kumiai Chemical Industry Co., Ltd.), having Accession No. FERM BP-8234, U.S.
- Patent No.7,094,592 + TX Bacillus subtilis strain BU1814, (VELONDIS® PLUS, VELONDIS® FLEX and VELONDIS® EXTRA from BASF SE) + TX, Bacillus subtilis var. amyloliquefaciens strain FZB24 having Accession No. DSM 10271 (available from Novozymes as TAEGRO® or TAEGRO® ECO (EPA Registration No.70127-5)) + TX, Bacillus subtilis, in particular strain QST713/AQ713 (having NRRL Accession No. B-21661 and described in U.S.
- Patent No.6,060,051 available as SERENADE® OPTI or SERENADE® ASO from Bayer CropScience LP, US
- TX Paenibacillus polymyxa
- strain AC-1 e.g. TOPSEED® from Green Biotech Company Ltd.
- TX Paenibacillus sp. strain having Accession No. NRRL B-50972 or Accession No. NRRL B-67129, WO 2016/154297 + TX, Pantoea agglomerans, in particular strain E325 (Accession No.
- NRRL B-21856 (available as BLOOMTIME BIOLOGICALTM FD BIOPESTICIDE from Northwest Agri Products) + TX, Pseudomonas proradix (e.g. PRORADIX® from Sourcon Padena) + TX; Aureobasidium pullulans, in particular blastospores of strain DSM14940, blastospores of strain DSM 14941 or mixtures of blastospores of strains DSM14940 and DSM14941 (e.g., BOTECTOR® and BLOSSOM PROTECT® from bio-ferm, CH) + TX, Pseudozyma aphidis (as disclosed in WO2011/151819 by Yissum Research Development Company of the Hebrew University of Jerusalem) + TX, Saccharomyces cerevisiae, in particular strains CNCM No.1-3936, CNCM No.1-3937, CNCM No.1-3938 or CNCM No.1-3939 (WO 2010/086790) from
- DSM 23117 (available as RHIZOVITAL® from ABiTEP, DE) + TX
- Bacillus amyloliquefaciens in particular strain D747 (available as Double NickelTM from Kumiai Chemical Industry Co., Ltd., having accession number FERM BP-8234, US Patent No.7,094,592) + TX
- Bacillus licheniformis FMCH001 and Bacillus subtilis FMCH002 (QUARTZO® (WG) and PRESENCE® (WP) from FMC Corporation) + TX
- Bacillus licheniformis in particular strain SB3086, having Accession No.
- ATCC 55406, WO 2003/000051 (available as ECOGUARD® Biofungicide and GREEN RELEAFTM from Novozymes) + TX, Bacillus methylotrophicus strain BAC-9912 (from Chinese Academy of Sciences’ Institute of Applied Ecology) + TX, Bacillus mycoides, isolate, having Accession No. B-30890 (available as BMJ TGAI® or WG and LifeGardTM from Certis USA LLC) + TX, Bacillus pumilus, in particular strain GB34 (available as Yield Shield® from Bayer AG, DE) + TX, Bacillus pumilus, in particular strain QST2808 (available as SONATA® from Bayer CropScience LP, US, having Accession No.
- Patent No.5,061,495 + TX Bacillus subtilis strain Y1336 (available as BIOBAC® WP from Bion- Tech, Taiwan, registered as a biological fungicide in Taiwan under Registration Nos.4764, 5454, 5096 and 5277) + TX, Bacillus subtilis var. amyloliquefaciens strain FZB24 having Accession No. DSM 10271 (available from Novozymes as TAEGRO® or TAEGRO® ECO (EPA Registration No.70127-5)) + TX, Bacillus subtilis Y1336 (available as BIOBAC® WP from Bion-Tech, Taiwan, registered as a biological fungicide in Taiwan under Registration Nos.
- NRRL B-50897, WO 2017/019448 e.g., HOWLERTM and ZIO® from AgBiome Innovations, US
- TX Pseudomonas chlororaphis
- strain MA342 e.g. CEDOMON®, CERALL®, and CEDRESS® by Bioagri and Koppert
- TX Pseudomonas fluorescens strain A506 (e.g. BLIGHTBAN® A506 by NuFarm) + TX
- Pseudomonas proradix e.g.
- PRORADIX® from Sourcon Padena + TX
- Streptomyces griseoviridis strain K61 also known as Streptomyces galbus strain K61
- DSM 7206 Streptomyces galbus strain K61
- MYCOSTOP® from Verdera, PREFENCE® from BioWorks, cf.
- BIOKUPRUMTM by AgriLife + TX
- Chaetomium globosum available as RIVADIOM® by Rivale
- TX Cladosporium cladosporioides
- strain H39 having Accession No. CBS122244, US 2010/0291039 (by Stichting Moowgrass Onderzoek) + TX
- Coniothyrium minitans in particular strain CON/M/91-8 (Accession No. DSM9660, e.g.
- strain ICC 080 having Accession No. IMI 392151 (e.g., BIO-TAMTM from Isagro USA, Inc. or BIODERMA® by Agrobiosol de Mexico, S.A. de C.V.) + TX, Penicillium vermiculatum + TX, Phlebiopsis gigantea strain VRA 1992 (ROTSTOP® C from danstar Ferment) + TX, Pseudozyma flocculosa, strain PF-A22 UL (available as SPORODEX® L by Plant Products Co., CA) + TX, Saccharomyces cerevisiae strain LAS117 cell walls (CEREVISANE® from Lesaffre, ROMEO® from BASF SE) + TX, Saccharomyces cerevisiae strains CNCM No.1-3936, CNCM No.1-3937, CNCM No.1-3938, CNCM No.1-3939 (WO 2010/086790) from Lesaffre et Compag
- T- Gro from Andermatt Biocontrol + TX
- Trichoderma atroviride strain 77B T77 from Andermatt Biocontrol
- Trichoderma atroviride strain ATCC 20476 IMI 206040
- Trichoderma atroviride strain LC52 e.g. Tenet by Agrimm Technologies Limited
- Trichoderma atroviride strain LU132 e.g. Sentinel from Agrimm Technologies Limited
- TX Trichoderma atroviride strain NMI no. V08/002388 + TX
- Trichoderma atroviride strain NMI no. V08/002389 + TX Trichoderma atroviride strain NMI no.
- Patent No.8,431,120 (from Bi-PA)) + TX, Trichoderma atroviride,strain CNCM 1-1237 (e.g. Esquive® WP from Agrauxine, FR) + TX, Trichoderma fertile (e.g. product TrichoPlus from BASF) + TX, Trichoderma gamsii (formerly T. viride) + TX, Trichoderma gamsii (formerly T. viride) strain ICC 080 (IMI CC 392151 CABI) (available as BIODERMA® by AGROBIOSOL DE MEXICO, S.A.
- Trichoderma gamsii strain ICC080 IMI CC 392151 CABI, e.g. BioDerma by AGROBIOSOL DE MEXICO, S.A. DE C.V.
- + TX Trichoderma harmatum + TX
- Trichoderma harmatum having Accession No. ATCC 28012 + TX, Trichoderma harzianum + TX, Trichoderma harzianum rifai T39 (e.g.
- Trichodex® from Makhteshim, US + TX, Trichoderma harzianum strain Cepa SimbT5 (from Simbiose Agro), + TX, Trichoderma harzianum strain DB 103 (available as T-GRO® 7456 by Dagutat Biolab) + TX, Trichoderma harzianum strain ITEM 908 (e.g. Trianum-P from Koppert) + TX, Trichoderma harzianum strain T-22 (e.g. Trianum-P from Andermatt Biocontrol or Koppert) + TX, Trichoderma harzianum strain TH35 (e.g.
- Trichoderma polysporum strain IMI 206039 e.g. Binab TF WP by BINAB Bio- Innovation AB, Sweden
- TX Trichoderma stromaticum having Accession No. Ts3550 (e.g. Tricovab by CEPLAC, Brazil) + TX
- Trichoderma virens also known as Gliocladium virens
- strain GL-21 e.g. SoilGard by Certis, US
- Trichoderma virens strain G-41 formerly known as Gliocladium virens (Accession No.
- ATCC 20906 (e.g., ROOTSHIELD® PLUS WP and TURFSHIELD® PLUS WP from BioWorks, US) + TX, Trichoderma viride in particular strain B35 (Pietr et al., 1993, Zesz. Nauk. A R w Szczecinie 161: 125-137) + TX, Trichoderma viride strain TV1(e.g. Trianum-P by Koppert) + TX, Ulocladium oudemansii strain U3, having Accession No.
- NM 99/06216 e.g., BOTRY-ZEN® by Botry-Zen Ltd, New Zealand and BOTRYSTOP® from BioWorks, Inc.
- TX Verticillium albo-atrum (formerly V. dahliae) strain WCS850 having Accession No.
- WCS850 deposited at the Central Bureau for Fungi Cultures (e.g., DUTCH TRIG® by Tree Care Innovations) + TX, Verticillium chlamydosporium + TX; a mixture of Azotobacter vinelandii and Clostridium pasteurianum (available as INVIGORATE® from Agrinos) + TX, a mixture of Bacillus licheniformis FMCH001 and Bacillus subtilis FMCH002 (available as QUARTZO® (WG), PRESENCE® (WP) from FMC Corporation) + TX, Azorhizobium caulinodans, in particular strain ZB-SK-5 + TX, Azospirillum brasilense (e.g., VIGOR® from KALO, Inc.) + TX, Azospirillum lipoferum (e.g., VERTEX-IFTM from TerraMax, Inc.) + TX, Azotobacter chroococcum, in particular strain H
- NRRL B-5015 + TX
- Bacillus amyloliquefaciens in particular strain FZB42 e.g. RHIZOVITAL® from ABiTEP, DE
- Bacillus amyloliquefaciens in particular strain IN937a + TX Bacillus amyloliquefaciens pm414 (LOLI-PEPTA® from Biofilm Crop Protection) + TX
- Bacillus amyloliquefaciens SB3281 ATCC # PTA-7542, WO 2017/205258
- Bacillus amyloliquefaciens TJ1000 available as QUIKROOTS® from Novozymes
- Bacillus cereus family member EE128 NRRL No.
- YIELD SHIELD® from Bayer Crop Science, DE
- + TX Bacillus pumilus in particular strain QST2808 (Accession No. NRRL No. B-30087) + TX, Bacillus siamensis in particular strain KCTC 13613T + TX, Bacillus subtilis in particular strain AQ30002 (Accession No. NRRL No. B-50421 and described in U.S. Patent Application No.13/330,576) + TX, Bacillus subtilis in particular strain AQ30004 (NRRL No. B-50455 and described in U.S. Patent Application No. 13/330,576) + TX, Bacillus subtilis in particular strain MBI 600 (e.g.
- BIOBOOST® from Brett Young Seeds + TX, Lactobacillus sp. (e.g. LACTOPLANT® from LactoPAFI) + TX, Mesorhizobium cicer (e.g., NODULATOR from BASF SE) + TX, Paenibacillus polymyxa in particular strain AC-1 (e.g. TOPSEED® from Green Biotech Company Ltd.) + TX, Pseudomonas aeruginosa in particular strain PN1 + TX, Pseudomonas proradix (e.g.
- PRORADIX® from Sourcon Padena + TX, Rhizobium leguminosarium biovar viciae (e.g., NODULATOR from BASF SE) + TX, Rhizobium leguminosarum in particular bv. viceae strain Z25 (Accession No. CECT 4585) + TX, Serratia marcescens in particular strain SRM (Accession No. MTCC 8708), + TX, Sinorhizobium meliloti strain NRG-185-1 (NITRAGIN® GOLD from Bayer CropScience) + TX, Thiobacillus sp. (e.g.
- Trichoderma atroviride strain SC1 (described in WO2009/116106) + TX, Trichoderma harzianum strain 1295-22 + TX, Trichoderma harzianum strain ITEM 908 + TX, Trichoderma harzianum strain T-22 (e.g. Trianum-P from Andermatt Biocontrol or Koppert) + TX, Trichoderma harzianum strain TSTh20, + TX, Trichoderma virens strain GI-3 + TX, Trichoderma virens strain GL-21 (e.g.
- aizawai in particular serotype H-7 (e.g. FLORBAC® WG from Valent BioSciences, US) + TX, Bacillus thuringiensis subsp. aizawai, in particular strain ABTS-1857 (SD-1372, e.g. XENTARI® from Valent BioSciences) + TX, Bacillus thuringiensis subsp. israelensis (serotype H-14) strain AM65-52 (Accession No. ATCC 1276) (e.g. VECTOBAC® by Valent BioSciences, US) + TX, Bacillus thuringiensis subsp.
- serotype H-7 e.g. FLORBAC® WG from Valent BioSciences, US
- TX Bacillus thuringiensis subsp. aizawai, in particular strain ABTS-1857 (SD-1372, e.g. XENTARI® from Valent BioSciences) + TX
- israeltaki strain ABTS 351 + TX Bacillus thuringiensis subsp. kurstaki strain BMP 123 (from Becker Microbial Products, IL, BARITONE from Bayer CropScience) + TX, Bacillus thuringiensis subsp. kurstaki strain EG 2348 (LEPINOX from Certis, US) + TX, Bacillus thuringiensis subsp. kurstaki strain EG 7841 (CRYMAX from Certis, US) + TX, Bacillus thuringiensis subsp. kurstaki strain HD-1 (e.g. DIPEL® ES from Valent BioSciences, US) + TX, Bacillus thuringiensis subsp.
- BMP 123 from Becker Microbial Products, IL, BARITONE from Bayer CropScience
- TX Bacillus thuringiensis subsp. kurstaki strain EG 2348 (LEPINOX from Certis, US) + TX
- israeltaki strain PB 54 + TX Bacillus thuringiensis subsp. kurstaki strain SA 11 (JAVELIN from Certis, US) + TX, Bacillus thuringiensis subsp. kurstaki strain SA 12 (THURICIDE from Certis, US) + TX, Bacillus thuringiensis subsp. tenebrionis strain NB 176 (SD-5428, e.g. NOVODOR® FC from BioFa DE) + TX, Bacillus thuringiensis var. Colmeri (e.g. TIANBAOBTC by Changzhou Jianghai Chemical Factory) + TX, Bacillus thuringiensis var.
- SD-5428 e.g. NOVODOR® FC from BioFa DE
- Bacillus thuringiensis var. Colmeri e.g. TIANBAOBTC by Changzhou Jianghai Chemical Factory
- MBI206 TGAI and ZELTO® from Marrone Bio Innovations + TX
- Chromobacterium subtsugae in particular strain PRAA4-1T e.g. MBI-203, e.g. GRANDEVO® from Marrone Bio Innovations
- TX Chromobacterium subtsugae in particular strain PRAA4-1T
- MBI-203 e.g. GRANDEVO® from Marrone Bio Innovations
- TX Lecanicillium muscarium Ve6 (MYCOTAL from Koppert) + TX
- Paenibacillus popilliae (formerly Bacillus popilliae, e.g. MILKY SPORE POWDERTM or MILKY SPORE GRANULARTM from St. Gabriel Laboratories) + TX
- Serratia entomophila e.g.
- ATCC74250 e.g. BOTANIGUARD® ES and MYCONTROL-O® from Laverlam International Corporation
- TX Metarhizium anisopliae 3213-1 (deposited under NRRL accession number 67074 disclosed in WO 2017/066094, Pioneer Hi-Bred International) + TX, Metarhizium robertsii 15013-1 (deposited under NRRL accession number 67073) + TX, Metarhizium robertsii 23013-3 (deposited under NRRL accession number 67075) + TX, Paecilomyces lilacinus strain 251 (MELOCON from Certis, US) + TX; Cydia pomonella (codling moth) granulosis virus (GV) + TX, Helicoverpa armigera (cotton bollworm) nuclear polyhedrosis virus (NPV) + TX, of Adoxophyes orana (summer fruit tortrix) granul
- Burkholderia cepacia (formerly known as Pseudomonas cepacia) + TX, Gigaspora spp. + TX, Glomus spp. + TX, Laccaria spp. + TX, LactoBacillus buchneri + TX, Paraglomus spp. + TX, Pisolithus tinctorus + TX, Pseudomonas spp. + TX, Rhizobium spp. in particular Rhizobium trifolii + TX, Rhizopogon spp. + TX, Scleroderma spp. + TX, Streptomyces spp. + TX, Suillus spp.
- compositions of the invention may also be applied with one or more systemically acquired resistance inducers (“SAR” inducer).
- SAR inducers are known and described in, for example, United States Patent No. US 6,919,298 and include, for example, salicylates and the commercial SAR inducer acibenzolar-S-methyl.
- the compounds of formula (I) as defined in the present invention are normally used in the form of compositions and can be applied to the crop area or plant to be treated, simultaneously or in succession with further compounds. These further compounds can be e.g. fertilizers or micronutrient donors or other preparations, which influence the growth of plants.
- the compounds of formula (I) as defined in the present invention may be used in the form of (fungicidal) compositions for controlling or protecting against phytopathogenic microorganisms, comprising as active ingredient at least one compound of formula (I) as defined in the present invention or of at least one preferred individual compound as above-defined, in free form or in agrochemically usable salt form, and at least one of the above-mentioned adjuvants.
- the invention therefore provides a composition, preferably a fungicidal composition, comprising at least one compound of formula (I) as defined in the present invention, an agriculturally acceptable carrier and optionally an adjuvant.
- An agricultural acceptable carrier is for example a carrier that is suitable for agricultural use.
- Agricultural carriers are well known in the art.
- said composition may comprise at least one or more pesticidally active compounds, for example an additional fungicidal active ingredient in addition to the compound of formula (I) as defined in the present invention.
- a further aspect of invention is related to a method of controlling or preventing an infestation of plants, e.g. useful plants such as crop plants, propagation material thereof, e.g. seeds, harvested crops, e.g.
- Controlling or preventing means reducing infestation by insects or by phytopathogenic or spoilage microorganisms or organisms potentially harmful to man, especially fungal organisms, to such a level that an improvement is demonstrated.
- a preferred method of controlling or preventing an infestation of crop plants by phytopathogenic microorganisms, especially fungal organisms, which comprises the application of a compound of formula (I) as defined in the present invention, or an agrochemical composition which contains at least one of said compounds, is foliar application.
- the frequency of application and the rate of application will depend on the risk of infestation by the corresponding pathogen or insect.
- the compounds of formula (I) as defined in the present invention can also penetrate the plant through the roots via the soil (systemic action) by drenching the locus of the plant with a liquid formulation, or by applying the compounds in solid form to the soil, e.g. in granular form (soil application). In crops of water rice such granulates can be applied to the flooded rice field.
- the compounds of formula (I) as defined in any the present invention may also be applied to seeds (coating) by impregnating the seeds or tubers either with a liquid formulation of the fungicide or coating them with a solid formulation.
- a formulation e.g. a composition containing the compound of formula (I) as defined in the present invention, and, if desired, a solid or liquid adjuvant or monomers for encapsulating the compound of formula (I) as defined in the present invention, may be prepared in a known manner, typically by intimately mixing and/or grinding the compound with extenders, for example solvents, solid carriers and, optionally, surface active compounds (surfactants).
- compositions that is the methods of controlling pathogens of the abovementioned type, such as spraying, atomizing, dusting, brushing on, dressing, scattering or pouring - which are to be selected to suit the intended aims of the prevailing circumstances - and the use of the compositions for controlling pathogens of the abovementioned type are other subjects of the invention.
- Typical rates of concentration are between 0.1 and 1000 ppm, preferably between 0.1 and 500 ppm, of active ingredient.
- the rate of application per hectare is preferably 1g to 2000 g of active ingredient per hectare, more preferably 10 to 1000 g/ha, most preferably 10 to 600 g/ha.
- a composition comprising a compound of formula (I) as defined in the present invention according to the present invention is applied either preventative, meaning prior to disease development or curative, meaning after disease development.
- compositions of the invention may be employed in any conventional form, for example in the form of a twin pack, a powder for dry seed treatment (DS), an emulsion for seed treatment (ES), a flowable concentrate for seed treatment (FS), a solution for seed treatment (LS), a water dispersible powder for seed treatment (WS), a capsule suspension for seed treatment (CF), a gel for seed treatment (GF), an emulsion concentrate (EC), a suspension concentrate (SC), a suspo-emulsion (SE), a capsule suspension (CS), a water dispersible granule (WG), an emulsifiable granule (EG), an emulsion, water in oil (EO), an emulsion, oil in water (EW), a micro-emulsion (ME), an oil dispersion (OD), an oil miscible flowable (OF), an oil miscible liquid (OL), a soluble concentrate (SL), an ultra-low volume suspension (SU), an ultra-low volume liquid (UL), a technical concentrate (TK
- compositions may be produced in conventional manner, e.g. by mixing the active ingredients with appropriate formulation inerts (diluents, solvents, fillers and optionally other formulating ingredients such as surfactants, biocides, anti-freeze, stickers, thickeners and compounds that provide adjuvancy effects).
- appropriate formulation inerts diiluents, solvents, fillers and optionally other formulating ingredients such as surfactants, biocides, anti-freeze, stickers, thickeners and compounds that provide adjuvancy effects.
- conventional slow release formulations may be employed where long lasting efficacy is intended.
- Particularly formulations to be applied in spraying forms such as water dispersible concentrates (e.g. EC, SC, DC, OD, SE, EW, EO and the like), wettable powders and granules, may contain surfactants such as wetting and dispersing agents and other compounds that provide adjuvancy effects, e.g.
- a seed dressing formulation is applied in a manner known per se to the seeds employing the combination of the invention and a diluent in suitable seed dressing formulation form, e.g. as an aqueous suspension or in a dry powder form having good adherence to the seeds.
- suitable seed dressing formulation form e.g. as an aqueous suspension or in a dry powder form having good adherence to the seeds.
- seed dressing formulations are known in the art.
- Seed dressing formulations may contain the single active ingredients or the combination of active ingredients in encapsulated form, e.g.
- the formulations include from 0.01 to 90% by weight of active agent, from 0 to 20% agriculturally acceptable surfactant and 10 to 99.99% solid or liquid formulation inerts and adjuvant(s), the active agent consisting of at least the compound of formula (I) as defined in the present invention together with component (B) and (C), and optionally other active agents, particularly microbiocides or conservatives or the like.
- Concentrated forms of compositions generally contain in between about 2 and 80%, preferably between about 5 and 70% by weight of active agent.
- Application forms of formulation may for example contain from 0.01 to 20% by weight, preferably from 0.01 to 5% by weight of active agent.
- Wettable powders a) b) c) active ingredient [compound of formula (I)] 25 % 50 % 75 % sodium lignosulfonate 5 % 5 % - sodium lauryl sulfate 3 % - 5 % sodium diisobutylnaphthalenesulfonate - 6 % 10 % phenol polyethylene glycol ether - 2 % - (7-8 mol of ethylene oxide) highly dispersed silicic acid 5 % 10 % 10 % Kaolin 62 % 27 % -
- the active ingredient is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording wettable powders that can be diluted with water to give suspensions of the desired concentration.
- Powders for dry seed treatment a) b) c) active ingredient [compound of formula (I)] 25 % 50 % 75 % light mineral oil 5 % 5 % 5 % highly dispersed silicic acid 5 % 5 % - Kaolin 65 % 40 % - Talcum - 20%
- active ingredient is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording powders that can be used directly for seed treatment.
- Emulsifiable concentrate active ingredient [compound of formula (I)] 10 % octylphenol polyethylene glycol ether 3 % (4-5 mol of ethylene oxide) calcium dodecylbenzenesulfonate 3 % castor oil polyglycol ether (35 mol of ethylene oxide) 4 % Cyclohexanone 30 % xylene mixture 50 % Emulsions of any required dilution, which can be used in plant protection, can be obtained from this concentrate by dilution with water.
- Coated granules Active ingredient [compound of formula (I)] 8 % polyethylene glycol (mol. wt.200) 3 % Kaolin 89 % The finely ground active ingredient is uniformly applied, in a mixer, to the kaolin moistened with polyethylene glycol. Non-dusty coated granules are obtained in this manner.
- Suspension concentrate active ingredient [compound of formula (I)] 40 % propylene glycol 10 % nonylphenol polyethylene glycol ether (15 mol of ethylene oxide) 6 % Sodium lignosulfonate 10 % carboxymethylcellulose 1 % silicone oil (in the form of a 75 % emulsion in water) 1 % Water 32 %
- the finely ground active ingredient is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water. Using such dilutions, living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
- Flowable concentrate for seed treatment active ingredient [compound of formula (I)] 40 % propylene glycol 5 % copolymer butanol PO/EO 2 % tristyrenephenole with 10-20 moles EO 2 % 1,2-benzisothiazolin-3-one (in the form of a 20% solution in water) 0.5 % monoazo-pigment calcium salt 5 % Silicone oil (in the form of a 75 % emulsion in water) 0.2 % Water 45.3 % The finely ground active ingredient is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- LC/MS means Liquid Chromatography Mass Spectrometry and the description of the apparatus and the method is as follows: Method A: Equipment: Shimadzu LCMS 2020 Mass Spectrometer; Column: HALO C182.7 ⁇ m, 3.0 mm ⁇ 30 mm; Mobile Phase: MeCN (with either 0.05% HCOOH or 0.05% TFA) - Water (with either 0.05% HCOOH or 0.05% TFA); Gradient: MeCN from 5% to 95% over 1.4 min, hold 0.6 min, total run time is 2.5 min; Flow rate: 1.8 mL/min; Column temperature: 50 o C; Wavelength: 214 and 254 nm PDA.
- Method B Spectra were recorded on a Mass Spectrometer from Waters (Acquity QDa Mass Spectrometer) equipped with an electrospray source (Polarity: Positive and Negative Polarity Switch), Capillary: 0.8 kV, Cone range: 25 V, Extractor: V (No extractor voltage for QDa detector) Source Temperature: 120°C, Desolvation Temperature: 600°C, Cone Gas Flow: 50 L/h, Desolvation Gas Flow: 1000 L/h, Mass range: 110 to 850 Da) and an Acquity UPLC from Waters: Quaternary solvent manager, heated column compartment , diode-array detector.
- Method C Spectra were recorded on a ACQUITY Mass Spectrometer from Waters Corporations (SQD or SQDII Single quadrupole mass spectrometer) equipped with an electrospray source (Polarity: positive or negative ions, Capillary: 3.0 kV, Cone: 30V, Extractor: 3.00 V, Source Temperature: 150°C, Desolvation Temperature: 400°C, Cone Gas Flow: 60 L/hr, Desolvation Gas Flow: 700 L/hr, Mass range: 140 to 800 Da) and an ACQUITY UPLC from Waters Corporations with solvent degasser, binary pump, heated column compartment and diode-array detector.
- an electrospray source Polarity: positive or negative ions, Capillary: 3.0 kV, Cone: 30V, Extractor: 3.00 V, Source Temperature: 150°C, Desolvation Temperature: 400°C, Cone Gas Flow: 60 L/hr, Desolvation Gas Flow: 700 L/h
- Method D Spectra were recorded on a Mass Spectrometer from Agilent (Single quad mass spectrometer) equipped with a Multimode- Electron Spray and APCI (Polarity: positive and negative ions), Capillary: 4.00 kV, Corona Current 4.0 ⁇ A, Charging Voltage, 2.00 kV, Nitrogen Gas Flow: 12.0 L/min, Nebulizer Pressure: 40 psig, Mass range: 100 to 1000 m/z), dry gas temperature 250 °C, Vaporizer temperature 200 °C and an UPLC from Waters: quaternary pump, heated column compartment, Variable wave length detector.
- Agilent Single quad mass spectrometer
- APCI Multimode- Electron Spray and APCI
- reaction mixture was stirred at room temperature for 2 hours.
- the reaction mixture was concentrated under reduce pressure and the residue was quenched with 10% NaOH, followed with a saturated solution of Na2S2O3 and extracted with chloroform.
- the combined organic layers were dried over Na2SO4, filtered, and concentrated under reduced pressure to afford 6-bromo-3-iodo-8-methyl- imidazo[1,2-a]pyrazine.
- the reaction mixture was degased with argon and then stirred in the microwave at 80 °C for 2 hours. After cooling down to room temperature, the reaction mixture was filtered through a Celite pad and washed with methanol. The filtrate was washed with water, then brine, dried over Na 2 SO 4 , filtered, and concentrated under reduced pressure. The crude residue was purified over a silica gel cartridge (cyclohexane/ethyl acetate) to afford methyl N-[5-(6-bromo-8-methyl-imidazo[1,2-a]pyrazin-3-yl)-2-pyridyl]carbamate.
- reaction mixture was stirred at 60 °C for 16 hours.
- the reaction mixture was cooled down to room temperature and then dissolved with water and extracted with ethyl acetate.
- the combined organic layers were dried over Na2SO4, filtered, and concentrated under reduced pressure.
- the crude residue was purified over a silica gel cartridge (cyclohexane/ethyl acetate) to afford 3-(4-fluoro-3- methoxy-anilino)propanenitrile.
- reaction mixture was stirred at 110 °C for 6 hours.
- the reaction mixture was filtered through celite and the filtrate was concentrated under reduce pressure.
- the residue was purified by reverse phase chromatography (C18 column, eluting with 5% to 90% MeCN/H2O) to afford methyl N-[5-[6-[2-cyanoethyl-(4-fluoro-3-methoxy- phenyl)carbamoyl]-8-methyl-imidazo[1,2-a]pyrazin-3-yl]-2-pyridyl]carbamate.
- Example 2 Preparation of methyl N-[5-[6-(6-fluoro-3,4-dihydro-2H-quinoline-1-carbonyl)-8- methyl-imidazo[1,2-a]pyrazin-3-yl]-2-pyridyl]carbamate (Compound 4) (Compound 4) Step A: Preparation of 6-bromo-8-methyl-imidazo[1,2-a]pyrazine To a solution of 5-bromo-3-methyl-pyrazin-2-amine (10.00 g, 53.19 mmol 1.00 equiv.) in ethanol (212 mL) was added a chloroacetaldehyde solution 50% in water (20.27 mL, 159.6 mmol, 3.00 equiv.).
- reaction mixture was stirred at 78 °C for 16 hours.
- the reaction mixture was quenched with a saturated NaHCO3 solution, extracted with ethyl acetate and the combined organic layers were washed with brine, dried over Na2SO4, filtered, and concentrated under reduced pressure to afford 6-bromo-8-methyl- imidazo[1,2-a]pyrazine.
- Step B Preparation of (6-fluoro-3,4-dihydro-2H-quinolin-1-yl)-(8-methylimidazo[1,2-a]pyrazin-6- yl)methanone
- 6-bromo-8-methyl-imidazo[1,2-a]pyrazine (1.00 g, 4.72 mmol, 1.00 equiv.)
- 6-fluoro-1,2,3,4- tetrahydroquinoline (0.720 g, 4.763 mmol, 1.01 equiv.)
- 4,5-bis(diphenylphosphino)-9,9- dimethylxanthene 0.278 g, 0.471 mmol, 0.10 equiv.
- bis(benzonitrile)palladium chloride (0.190 g, 0.471 mmol, 0.10 equiv.) and triethylamine (0.991 mL, 7.07 mmol, 1.50 equiv.) in toluene (10
- Step C Preparation of (3-bromo-8-methyl-imidazo[1,2-a]pyrazin-6-yl)-(6-fluoro-3,4-dihydro-2H-quinolin- 1-yl)methanone
- 6-fluoro-3,4-dihydro-2H-quinolin-1-yl)-(8-methylimidazo[1,2-a]pyrazin-6- yl)methanone (0.300 g, 0.966 mmol, 1.00 equiv.) in acetonitrile (3.86 mL) was added N- bromosuccinimide (0.172 g, 0.966 mmol, 1.00 equiv.) at room temperature and the reaction mixture was stirred for 1 hour.
- reaction mixture was quenched with a saturated aqueous solution of Na2S2O3 and extracted with ethyl acetate. The combined organic layers were washed with brine, dried over Na2SO4, filtered, and concentrated under reduced pressure to afford (3-bromo-8-methyl-imidazo[1,2- a]pyrazin-6-yl)-(6-fluoro-3,4-dihydro-2H-quinolin-1-yl)methanone.
- Example 3 preparation of methyl N-[5-[6-[(4-fluoro-3-methoxy-phenyl)-methyl-carbamoyl]-8- methyl-imidazo[1,2-a]pyrazin-3-yl]-2-pyridyl]carbamate (compound 7)
- Compound 7) Step A: Preparation of methyl 5-amino-6-bromo-pyrazine-2-carboxylate To an ice-cooled solution of methyl 5-aminopyrazine-2-carboxylate (10.0 g, 62.0 mmol, 1.00 equiv.) in acetonitrile (100 mL) was added N-bromosuccinimide (12.1 g, 68.2 mmol, 1.10 equiv.).
- Step B Preparation of methyl 5-amino-6-methyl-pyrazine-2-carboxylate
- Step C Preparation of ethyl 8-methylimidazo[1,2-a]pyrazine-6-carboxylate (Compound X-1) To a stirred solution of methyl 5-amino-6-methyl-pyrazine-2-carboxylate (1.10 g, 5.92 mmol, 1.00 equiv.) in ethanol (25.0 mL) was added 2-chloroacetaldehyde (2.32 g, 11.8 mmol, 2.00 equiv.).
- the reaction mixture was stirred at 100 oC for 16 hours.
- the reaction mixture was concentrated under reduced pressure, then diluted with ethyl acetate and washed with a saturated solution of NaHCO3.
- the combined organic layers were washed with brine, dried over Na 2 SO 4 , filtered, and concentrated under reduced pressure.
- the crude residue was purified over a silica gel cartridge (cyclohexane/ethyl acetate) to afford a mixture (1:1) of ethyl 8-methylimidazo[1,2-a]pyrazine-6-carboxylate and methyl 8- methylimidazo[1,2-a]pyrazine-6-carboxylate as brown solid.
- Step D Preparation of N-(4-fluoro-3-methoxy-phenyl)-8-methyl-imidazo[1,2-a]pyrazine-6-carboxamide
- ethyl 8-methylimidazo[1,2-a]pyrazine-6-carboxylate Compound X-1 (2.20 g, 9.65 mmol, 1.00 equiv.)
- 4-fluoro-3-methoxy-aniline (1.63 g, 11.6 mmol, 1.20 equiv.) in dry toluene (50.0 mL) was added trimethylaluminium (9.65 mL, 19.3 mmol, 2.00 equiv.) in toluene.
- the resulting reaction mixture was stirred at 80 oC for 4 hours under nitrogen atmosphere.
- the reaction mixture was cooled to 0 oC, quenched with aqueous Rochelle's salt and diluted with ethyl acetate.
- the reaction mixture was filtered through a celite pad and washed with ethyl acetate.
- the combined organic layers were washed with brine, dried over Na2SO4, filtered, and concentrated under reduced pressure.
- the crude residue was purified over a silica gel cartridge (cyclohexane/ethyl acetate) to afford N-(4-fluoro-3-methoxy- phenyl)-8-methyl-imidazo[1,2-a]pyrazine-6-carboxamide as a brown solid.
- Step E Preparation of N-(4-fluoro-3-methoxy-phenyl)-N,8-dimethyl-imidazo[1,2-a]pyrazine-6- carboxamide
- N-(4-fluoro-3-methoxy-phenyl)-8-methyl-imidazo[1,2-a]pyrazine-6- carboxamide (1.00 g, 3.16 mmol, 1.00 equiv.) in dry THF (20.0 mL) was added sodium hydride 60% in oil (0.380 g, 9.49 mmol, 3.00 equiv.).
- Step F Preparation of N-(4-fluoro-3-methoxy-phenyl)-3-iodo-N,8-dimethyl-imidazo[1,2-a]pyrazine-6- carboxamide
- N-(4-fluoro-3-methoxy-phenyl)-N,8-dimethyl-imidazo[1,2-a]pyrazine-6- carboxamide 30.0 mg, 0.0859 mmol, 1.00 equiv.
- DMF 2 mL
- N-iodosuccinimide 21.3 mg, 0.0945 mmol, 1.10 equiv.
- reaction mixture was quenched with water and extracted with ethyl acetate.
- the combined organic layers were washed with brine, dried over Na2SO4, filtered, and concentrated under reduced pressure.
- the crude residue was purified over a silica gel cartridge (cyclohexane/ethyl acetate) to afford N-(4-fluoro-3-methoxy-phenyl)- 3-iodo-N,8-dimethyl-imidazo[1,2-a]pyrazine-6-carboxamide as an off-white solid.
- Step G Preparation of methyl N-[5-[6-[(4-fluoro-3-methoxy-phenyl)-methyl-carbamoyl]-8-methyl- imidazo[1,2-a]pyrazin-3-yl]-2-pyridyl]carbamate (Compound 7)
- N-(4-fluoro-3-methoxy-phenyl)-3-iodo-N,8-dimethyl-imidazo[1,2-a]pyrazine-6- carboxamide 400 mg, 0.818 mmol, 1.00 equiv.
- methyl N-[5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan- 2-yl)-2-pyridyl]carbamate 0.273 g, 0.981 mmol, 1.20 equiv.
- 1,4-dioxane/ water (3:1) was added cesium carbonate (0.533 g, 1.
- Example 4 preparation of methyl N-[5-[6-[(4-fluoro-3-methoxy-phenyl)- (methoxymethyl)carbamoyl]-8-methyl-imidazo[1,2-a]pyrazin-3-yl]-2-pyridyl]carbamate (compound 8) (Compound 8) Step A: Preparation of N-(4-fluoro-3-methoxy-phenyl)-N-(methoxymethyl)-8-methyl-imidazo[1,2- a]pyrazine-6-carboxamide To an ice-cooled solution of N-(4-fluoro-3-methoxy-phenyl)-8-methyl-imidazo[1,2-a]pyrazine-6- carboxamide (1.20 g, 3.80 mmol, 1.00 equiv.) in DMF (50.0 mL) was added sodium hydride 60 % in oil (497 mg, 11.4 mmol, 3.00 equiv.).
- chloro(methoxy)methane (611 mg, 7.59 mmol, 2.00 equiv.) was added dropwise and the reaction mixture was stirred at room temperature for 2 hours.
- the reaction mixture was cooled to 0 °C, quenched with NH4Cl solution and extracted with ethyl acetate. The combined organic layers were washed with brine, dried over Na 2 SO 4 , filtered, and concentrated under reduced pressure.
- Step B Preparation of N-(4-fluoro-3-methoxy-phenyl)-3-iodo-N-(methoxymethyl)-8-methyl-imidazo[1,2- a]pyrazine-6-carboxamide
- N-(4-fluoro-3-methoxy-phenyl)-N-(methoxymethyl)-8-methyl-imidazo[1,2- a]pyrazine-6-carboxamide (0.500 g, 1.31 mmol, 1.00 equiv.) in DMF (10 mL) was added N- iodosuccinimide (0.676 g, 3.01 mmol, 2.30 equiv.) at room temperature and stirred for 16 hours.
- Step C Preparation of methyl N-[5-[6-[(4-fluoro-3-methoxy-phenyl)-(methoxymethyl)carbamoyl]-8- methyl-imidazo[1,2-a]pyrazin-3-yl]-2-pyridyl]carbamate (Compound 8)
- N-(4-fluoro-3-methoxy-phenyl)-3-iodo-N-(methoxymethyl)-8-methyl-imidazo[1,2- a]pyrazine-6-carboxamide (0.400 g, 0.766 mmol, 1.00 equiv.)
- methyl N-[5-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)-2-pyridyl]carbamate 255 mg, 0.919 mmol, 1.20 equiv.
- cesium carbonate (499 mg, 1.53 mmol
- Example 5 Preparation of 3-[6-(cyclopropanecarbonylamino)-3-pyridyl]-N-(4-fluoro-3-methoxy- phenyl)-N,8-dimethyl-imidazo[1,2-a]pyrazine-6-carboxamide (Compound 19) (Compound 19) Step A: Preparation of ethyl 3-bromo-8-methyl-imidazo[1,2-a]pyrazine-6-carboxylate To a solution of ethyl 8-methylimidazo[1,2-a]pyrazine-6-carboxylate (Compound X-1) (5.40 g, 26.3 mmol) in dimethylformamide (60.0 mL) was added N-bromosuccinimide (5.62 g, 31.6 mmol) in portions at room temperature.
- Step E Preparation of 3-[6-(cyclopropanecarbonylamino)-3-pyridyl]-N-(4-fluoro-3-methoxy-phenyl)- N,8-dimethyl-imidazo[1,2-a]pyrazine-6-carboxamide (Compound 19) Under an argon atmosphere, N-[5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-2- pyridyl]cyclopropanecarboxamide (0.052 mmol), XPhos Pd G3, CAS 1445085-55-1, (3.455 mg, 0.004 mmol) were placed in a vial.
- the mixture was heated at 140 °C in a microwave reactor for 2 hours. After completion of the reaction, the resulting mixture was concentrated under vacuum.
- the crude product was purified by flash chromatography using a dichloromethane/methanol gradient to obtain ethyl 8-bromoimidazo[1,2-a]pyrazine-6-carboxylate as a yellow solid.
- Step B Preparation of 8-bromoimidazo[1,2-a]pyrazine-6-carboxylic acid (Compound X-4) (Compound X-4)
- ethyl 8-bromoimidazo[1,2-a]pyrazine-6-carboxylate (1.00 g, 3.70 mmol)
- tetrahydrofuran (10.0 mL)
- water (2.50 mL)
- lithium hydroxide (0.177 g, 7.41 mmol
- Step D Preparation of 8-bromo-N-(4-fluoro-3-methoxy-phenyl)-3-iodo-N-methyl-imidazo[1,2- a]pyrazine-6-carboxamide
- 8-bromo-N-(4-fluoro-3-methoxy-phenyl)-N-methyl-imidazo[1,2-a]pyrazine-6- carboxamide Compound X-5 (0.200 g, 0.527 mmol) in acetonitrile (2.0 mL) was added N- Iodosuccinimide (0.119 g, 0.527 mmol) dropwise at 0 °C.
- Step F Preparation of methyl N-[5-[8-bromo-6-[(4-fluoro-3-methoxy-phenyl)-methyl- carbamoyl]imidazo[1,2-a]pyrazin-3-yl]-2-pyridyl]carbamate (Compound 25) To a solution of 8-bromo-N-(4-fluoro-3-methoxy-phenyl)-3-iodo-N-methyl-imidazo[1,2-a]pyrazine-6- carboxamide (0.120 g, 0.238 mmol) in dioxane (10.0 mL) and water (2.00 mL) was added methyl N-[5- (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-2-pyridyl]carbamate (0.0991 g, 0.356 mmol), bis(diphenylphosphino)ferrocene-palladium(II)dich
- Example 7 Preparation of methyl N-[5-[8-cyano-6-[(4-fluoro-3-methoxy-phenyl)-methyl- carbamoyl]imidazo[1,2-a]pyrazin-3-yl]-2-pyridyl]carbamate (Compound 23) Step A: Preparation of 8-cyano-N-(4-fluoro-3-methoxy-phenyl)-N-methyl-imidazo[1,2-a]pyrazine-6- carboxamide To a solution of 8-bromo-N-(4-fluoro-3-methoxy-phenyl)-N-methyl-imidazo[1,2-a]pyrazine-6- carboxamide (Compound X-5) (0.200 g, 0.527 mmol) in dimethylformamide (4.00 mL) was added zinc dicyanide (0.124 g, 1.05 mmol), (5-diphenylphosphanyl-9,9-dimethyl-xanthen-4-
- Step B Preparation of 3-bromo-8-cyano-N-(4-fluoro-3-methoxy-phenyl)-N-methyl-imidazo[1,2- a]pyrazine-6-carboxamide
- 8-cyano-N-(4-fluoro-3-methoxy-phenyl)-N-methyl-imidazo[1,2-a]pyrazine-6- carboxamide (0.150 g, 0.461 mmol) in acetonitrile (3.0 mL) was added 1-bromopyrrolidine-2,5-dione (0.0821 g, 0.461 mmol) dropwise at 0 °C.
- Example 8 Preparation of methyl N-[5-[6-[cyanomethyl-(4-fluoro-3-methoxy-phenyl)carbamoyl]- 8-methyl-imidazo[1,2-a]pyrazin-3-yl]-2-pyridyl]carbamate (Compound 2) (Compound 2) Step A: Preparation of 2-(4-fluoro-3-methoxy-anilino)acetonitrile To a solution of 4-fluoro-3-methoxy-aniline (0.500 g, 3.188 mmol) in acetonitrile (3.18 mL) was added sodium iodide (0.503 g, 3.188 mmol) and 2-chloroacetonitrile (0.225 mL, 3.379 mmol).
- the mixture was stirred at 70 °C for 3 hours. Then, the reaction was cooled to room temperature and water was added. The aqueous layer was extracted with ethyl acetate and the combined organic layers were washed with brine, dried over sodium sulfate, filtered and concentrated under reduced pressure. The crude material was used as such without further purification in the following step.
- the vial was flushed with nitrogen, then the vial was pressurized with nitrogen, then flushed with carbon monoxide twice and finally pressurized with 10 bar of carbon monoxide.
- the pressurized vial was heated at 110 °C for 7 hours. Then, the mixture was filtered through celite pad, concentrated under reduced pressure.
- the crude material was purified by flash chromatography using a cyclohexane/ethyl acetate gradient to afford N-(cyanomethyl)-N-(4-fluoro- 3-methoxy-phenyl)-8-methyl-imidazo[1,2-a]pyrazine-6-carboxamide.
- Example 9 Preparation of methyl N-[4-[6-[(4-fluoro-3-methoxy-phenyl)-methyl-carbamoyl]-8- methyl-imidazo[1,2-a]pyrazin-3-yl]phenyl]carbamate (Compound 36) (Compound 36) To a mixture of 3-bromo-N-(4-fluoro-3-methoxy-phenyl)-N,8-dimethyl-imidazo[1,2-a]pyrazine-6- carboxamide (Compound X-3) (0.200 g, 0.509 mmol) and methyl N-[4-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)phenyl]carbamate, CAS 844500-75-0 (0.169 g, 0.610 mmol) in mixture of dioxane/H2O (4:1, 5.0 mL) was added 1,1'-bis(diphenylphosphino)fer
- Step B Preparation of 8-acetamido-N-ethyl-N-(4-fluoro-3-methoxy-phenyl)imidazo[1,2-a]pyrazine-6- carboxamide
- 8-bromo-N-ethyl-N-(4-fluoro-3-methoxy-phenyl)imidazo[1,2-a]pyrazine-6-carboxamide (0.400 g, 1.02 mmol) in dioxane (5.00 mL) was added acetamide (0.120 g, 2.03 mmol), Xantphos-Pd- G2 (CAS 1375325-77-1, 0.0904 g, 0.102 mmol) and cesium carbonate (0.497 g, 1.53 mmol).
- Example 11 Preparation of methyl N-[5-[6-[ethyl-(4-fluoro-3-methoxy-phenyl)carbamoyl]-8- methyl-imidazo[1,2-a]pyrazin-3-yl]-2-pyridyl]carbamate (Compound 3) (Compound 3) Step A: Preparation of N-ethyl-N-(4-fluoro-3-methoxy-phenyl)-8-methyl-imidazo[1,2-a]pyrazine-6- carboxamide A vial was charged with 6-bromo-8-methyl-imidazo[1,2-a]pyrazine (CAS 1159815-50-5) (0.600 g, 2.83 mmol), N-ethyl-4-fluoro-3-methoxy-aniline (0.484 g, 2.86 mmol), bis(benzonitrile)palladium chloride (0.114 g, 0.283 mmol), 4,5-bis(diphenylphos
- the vial was flushed with nitrogen, then the vial was pressurized with nitrogen, then flushed with carbon monoxide twice and finally pressurized with 10 bar of carbon monoxide.
- the pressurized vial was heated at 110 °C for 6 hours. Then, the mixture was filtered through celite pad, concentrated under reduced pressure.
- the crude material was purified by flash chromatography using a cyclohexane/ethyl acetate gradient to afford N-ethyl-N-(4-fluoro-3- methoxy-phenyl)-8-methyl-imidazo[1,2-a]pyrazine-6-carboxamide as a brown solid.
- Example 13 Preparation of methyl N-[5-[6-[(4-cyano-3-methoxy-phenyl)-methyl-carbamoyl]-8- methyl-imidazo[1,2-a]pyrazin-3-yl]-2-pyridyl]carbamate (Compound 34) (Compound 34) To a solution of 3-bromo-N-(4-cyano-3-methoxy-phenyl)-N,8-dimethyl-imidazo[1,2-a]pyrazine-6- carboxamide (Compound X-6) (0.400 g, 0.999 mmol) in dioxane (4.0 mL) and water (1.0 mL) was added methyl N-[5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-2-pyridyl]carbamate (0.278 g, 0.999 mmol), bis(diphenylphosphino)ferrocene-palladium(
- Step A Preparation of ethyl 2,8-dimethylimidazo[1,2-a]pyrazine-6-carboxylate
- the vial was sealed and pressurized to 2.5 MPa with carbon monoxide.
- the reaction mixture was stirred at 100 °C for 8 hours. Then the vessel was cooled to room temperature and pressure was released. The reaction mixture was concentrated and the residue was purified by flash chromatography using a methanol/dichloromethane gradient to afford ethyl 2,8-dimethylimidazo[1,2-a]pyrazine-6- carboxylate as a brown solid.
- Step B Preparation of ethyl 3-bromo-2,8-dimethyl-imidazo[1,2-a]pyrazine-6-carboxylate
- ethyl 2,8-dimethylimidazo[1,2-a]pyrazine-6-carboxylate (0.800 g, 3.65 mmol) in acetonitrile (10.0 mL) was added 1-bromopyrrolidine-2,5-dione (0.779 g, 4.38 mmol) dropwise at room temperature. The reaction was stirred at room temperature for 2 hours. Then the resulting mixture was diluted with water.
- Step D Preparation of 3-bromo-N-(4-fluoro-3-methoxy-phenyl)-N,2,8-trimethyl-imidazo[1,2-a]pyrazine- 6-carboxamide
- 3-bromo-2,8-dimethyl-imidazo[1,2-a]pyrazine-6-carboxylic acid (0.260 g, 0.963 mmol) in dimethylformamide (5.0 mL) was added 1-methylimidazole (0.237 g, 2.89 mmol) and [chloro(dimethylamine)methylene]-dimethyl-ammonium hexafluorophosphate (CAS 94790-35-9, 0.405 g, 1.44 mmol) at room temperature.
- Example 15 Preparation of methyl N-[5-[6-[(4-fluoro-3-methyl-phenyl)-methyl-carbamoyl]-8- methyl-imidazo[1,2-a]pyrazin-3-yl]-2-pyridyl]carbamate (Compound 5) (Compound 5) Step A: Preparation of N-(4-fluoro-3-methyl-phenyl)-N,8-dimethyl-imidazo[1,2-a]pyrazine-6- carboxamide A vial was charged with 6-bromo-8-methyl-imidazo[1,2-a]pyrazine (CAS 1159815-50-5) (0.700 g, 3.30 mmol), 4-fluoro-N,3-dimethylaniline (0.464 g, 3.33 mmol), bis(benzonitrile)palladium chloride (0.133 g, 0.330 mmol), 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene
- the vial was flushed with nitrogen, then the vial was pressurized with nitrogen, then flushed with carbon monoxide twice and finally pressurized with 10 bar of carbon monoxide.
- the pressurized vial was heated at 110 °C for 6 hours. Then, the mixture was filtered through celite pad, concentrated under reduced pressure.
- the crude material was purified by flash chromatography using a cyclohexane/ethyl acetate gradient to afford N-(4-fluoro-3-methyl-phenyl)-N,8- dimethyl-imidazo[1,2-a]pyrazine-6-carboxamide.
- Example 18 Preparation of methyl N-[5-[6-[(4-fluoro-3-methoxy-phenyl)-methyl-carbamoyl]-2- methyl-imidazo[1,2-a]pyrazin-3-yl]-2-pyridyl]carbamate (Compound 37) (Compound 37) Step A: Preparation of ethyl 3-bromo-2-methyl-imidazo[1,2-a]pyrazine-6-carboxylate To a solution of ethyl 2-methylimidazo[1,2-a]pyrazine-6-carboxylate (CAS 1610666-95-9) (0.800 g, 3.90 mmol) in acetonitrile (10.0 mL) was added 1-bromopyrrolidine-2,5-dione (0.833 g, 4.68 mmol) portionwise at room temperature.
- Step B Preparation of 3-bromo-2-methyl-imidazo[1,2-a]pyrazine-6-carboxylic acid
- ethyl 3-bromo-2-methyl-imidazo[1,2-a]pyrazine-6-carboxylate (0.600 g, 2.11 mmol) in tetrahydrofuran/H2O (1:1, 10.0 mL) was added lithium hydroxide (0.101 g, 4.22 mmol). The mixture was stirred at room temperature for 3 hours. Then, the organic solvent was removed under reduced pressure.
- the leaf disks are inoculated with a spore suspension of the fungus 1 day after application.
- the inoculated leaf disks are incubated at 16 °C and 75% rh under a light regime of 24 h darkness followed by 12 h light / 12 h darkness in a climate cabinet and the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf disks (5 - 7 days after application).
- the inoculated leaf disks are incubated at 19 °C and 80% rh under a light regime of 12 h light / 12 h darkness in a climate cabinet and the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf disks (6 - 8 days after application).
- the following compounds gave at least 80% control of Plasmopara viticola at 200 ppm when compared to untreated control under the same conditions, which showed extensive disease development: 1 , 2 , 3 , 4 , 6 , 7 , 8 , 15 , 16 , 19 , 23 , 24 , 25 , 29 , 30 , 31 , 34 , 35 , 36 Pythium ultimum / liquid culture (seedling damping off) Mycelia fragments and oospores of a newly grown liquid culture of the fungus are directly mixed into nutrient broth (PDB potato dextrose broth).
- PDB potato dextrose broth Pythium ultimum / liquid culture
- test compound After placing a (DMSO) solution of test compound into a microtiter plate (96-well format), the nutrient broth containing the fungal mycelia/spore mixture is added. The test plates are incubated at 24 °C and the inhibition of growth is determined photometrically 2-3 days after application.
- the following compounds gave at least 80% control of Pythium ultimum at 20 ppm when compared to untreated control under the same conditions, which showed extensive disease development: 1 , 2 , 3 , 6 , 7 , 8 , 14 , 15 , 16 , 17 , 19 , 26 , 29 , 31 , 34 , 35 , 36
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Environmental Sciences (AREA)
- Engineering & Computer Science (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Organic Chemistry (AREA)
- Agronomy & Crop Science (AREA)
- Health & Medical Sciences (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Microbiology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Mycology (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2024251684A AU2024251684A1 (en) | 2023-04-13 | 2024-04-11 | Imidazo[1,2-a]pyrazine derivatives |
| KR1020257037541A KR20250170124A (en) | 2023-04-13 | 2024-04-11 | imidazo[1,2-a]pyrazine derivatives |
| CN202480022192.8A CN121013850A (en) | 2023-04-13 | 2024-04-11 | Imidazolo[1,2-a]pyrazine derivatives |
| IL323645A IL323645A (en) | 2023-04-13 | 2025-09-28 | Imidazo[1,2-a]pyrazine derivatives |
| MX2025011849A MX2025011849A (en) | 2023-04-13 | 2025-10-03 | Imidazo[1,2-a]pyrazine derivatives |
| CONC2025/0015496A CO2025015496A2 (en) | 2023-04-13 | 2025-11-06 | Imidazo[1,2-a]pyrazine derivatives |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23167802.0 | 2023-04-13 | ||
| EP23167802 | 2023-04-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024213656A1 true WO2024213656A1 (en) | 2024-10-17 |
Family
ID=86007292
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/059875 Ceased WO2024213656A1 (en) | 2023-04-13 | 2024-04-11 | Imidazo[1,2-a]pyrazine derivatives |
Country Status (9)
| Country | Link |
|---|---|
| KR (1) | KR20250170124A (en) |
| CN (1) | CN121013850A (en) |
| AR (1) | AR132351A1 (en) |
| AU (1) | AU2024251684A1 (en) |
| CO (1) | CO2025015496A2 (en) |
| IL (1) | IL323645A (en) |
| MX (1) | MX2025011849A (en) |
| TW (1) | TW202446260A (en) |
| WO (1) | WO2024213656A1 (en) |
Citations (68)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2016A (en) | 1841-03-26 | Mode of constructing fireplaces and chimney-staoks ii | ||
| EP0353191A2 (en) | 1988-07-29 | 1990-01-31 | Ciba-Geigy Ag | DNA sequences encoding polypeptides having beta-1,3-glucanase activity |
| EP0367474A1 (en) | 1988-11-01 | 1990-05-09 | Mycogen Corporation | Novel bacillus thuringiensis isolate denoted b.t. ps81gg, active against lepidopteran pests, and a gene encoding a lepidopteran-active toxin |
| EP0374753A2 (en) | 1988-12-19 | 1990-06-27 | American Cyanamid Company | Insecticidal toxines, genes coding therefor, antibodies binding them, transgenic plant cells and plants expressing these toxines |
| EP0392225A2 (en) | 1989-03-24 | 1990-10-17 | Ciba-Geigy Ag | Disease-resistant transgenic plants |
| WO1990013651A1 (en) | 1989-05-09 | 1990-11-15 | Imperial Chemical Industries Plc | Bacterial genes |
| EP0401979A2 (en) | 1989-05-18 | 1990-12-12 | Mycogen Corporation | Novel bacillus thuringiensis isolates active against lepidopteran pests, and genes encoding novel lepidopteran-active toxins |
| EP0427529A1 (en) | 1989-11-07 | 1991-05-15 | Pioneer Hi-Bred International, Inc. | Larvicidal lectins and plant insect resistance based thereon |
| EP0451878A1 (en) | 1985-01-18 | 1991-10-16 | Plant Genetic Systems, N.V. | Modifying plants by genetic engineering to combat or control insects |
| US5061495A (en) | 1988-03-07 | 1991-10-29 | Agricultural Genetics Company Limited | Antibiotic derived from b. subtilis |
| WO1993007278A1 (en) | 1991-10-04 | 1993-04-15 | Ciba-Geigy Ag | Synthetic dna sequence having enhanced insecticidal activity in maize |
| WO1995033818A2 (en) | 1994-06-08 | 1995-12-14 | Ciba-Geigy Ag | Genes for the synthesis of antipathogenic substances |
| WO1995034656A1 (en) | 1994-06-10 | 1995-12-21 | Ciba-Geigy Ag | Novel bacillus thuringiensis genes coding toxins active against lepidopteran pests |
| WO1997033890A1 (en) | 1996-03-11 | 1997-09-18 | Novartis Ag | Pyrimidin-4-one derivatives as pesticide |
| JPH11253151A (en) | 1997-11-13 | 1999-09-21 | Kumiai Chem Ind Co Ltd | Rice seedling raising disease control agent |
| WO2000015615A1 (en) | 1998-09-15 | 2000-03-23 | Syngenta Participations Ag | Pyridine ketones useful as herbicides |
| US6060051A (en) | 1997-05-09 | 2000-05-09 | Agraquest, Inc. | Strain of bacillus for controlling plant diseases and corn rootworm |
| US6245551B1 (en) | 1999-03-30 | 2001-06-12 | Agraquest, Inc. | Strain of Bacillus pumilus for controlling plant diseases caused by fungi |
| WO2002015701A2 (en) | 2000-08-25 | 2002-02-28 | Syngenta Participations Ag | Bacillus thuringiensis crystal protein hybrids |
| WO2002048146A2 (en) * | 2000-12-13 | 2002-06-20 | Basf Aktiengesellschaft | Use of substituted imidazoazines, novel imidazoazines, methods for the production thereof, and agents containing these compounds |
| WO2003000051A2 (en) | 2001-06-22 | 2003-01-03 | Drahos David J | Novel biofungicide |
| WO2003018810A2 (en) | 2001-08-31 | 2003-03-06 | Syngenta Participations Ag | Modified cry3a toxins and nucleic acid sequences coding therefor |
| WO2003052073A2 (en) | 2001-12-17 | 2003-06-26 | Syngenta Participations Ag | Novel corn event |
| US6919298B2 (en) | 2002-04-04 | 2005-07-19 | Valent Biosciences Corporation | Enhanced herbicide composition |
| US7094592B2 (en) | 2001-11-26 | 2006-08-22 | Kumiai Chemical Industry Co., Ltd. | Bacillus sp. D747 strain, plant disease controlling agents and insect pest controlling agents using the same and control method using the agents |
| US7579183B1 (en) | 2006-12-01 | 2009-08-25 | The United States Of America As Represented By The Secretary Of Agriculture | Saprophytic yeast, Pichia anomala |
| WO2009116106A1 (en) | 2008-03-21 | 2009-09-24 | Trentino Sviluppo S.P.A. | Trichoderma atroviride sc1 for biocontrol of fungal diseases in plants |
| WO2010086790A1 (en) | 2009-01-27 | 2010-08-05 | Lesaffre Et Compagnie | Saccharomyces cerevisiae strains with phytosanitary capabilities |
| US20100291039A1 (en) | 2007-12-14 | 2010-11-18 | Kohl Jurgen Anton | Novel micro-organisms controlling plant pathogens |
| WO2011106491A2 (en) | 2010-02-25 | 2011-09-01 | Marrone Bio Innovations, Inc. | Isolated bacterial strain of the genus burkholderia and pesticidal metabolites therefrom |
| WO2011138281A2 (en) | 2010-05-06 | 2011-11-10 | Bayer Cropscience Ag | Process for the preparation of dithiine tetracarboxydiimides |
| WO2011151819A2 (en) | 2010-06-01 | 2011-12-08 | Yissum Research Development Company Of The Hebrew University Of Jerusalem Ltd. | Pseudozyma aphidis as a biocontrol agent against various plant pathogens |
| WO2013032693A2 (en) | 2011-08-27 | 2013-03-07 | Marrone Bio Innovations, Inc. | Isolated bacterial strain of the genus burkholderia and pesticidal metabolites therefrom-formulations and uses |
| WO2013034938A2 (en) | 2011-09-08 | 2013-03-14 | Szegedi Tudományegyetem | A copper resistant, fengycin-producing bacillus mojavensis strain for controlling vegetable pathogens, its use and compositions containing it |
| WO2014006945A1 (en) | 2012-07-04 | 2014-01-09 | アグロカネショウ株式会社 | 2-aminonicotinic acid ester derivative and bactericide containing same as active ingredient |
| WO2014028521A1 (en) | 2012-08-14 | 2014-02-20 | Marrone Bio Innovations, Inc. | Bacillus sp. strain with antifungal, antibacterial and growth promotion activity |
| WO2014095675A1 (en) | 2012-12-19 | 2014-06-26 | Bayer Cropscience Ag | Difluoromethyl-nicotinic-indanyl carboxamides as fungicides |
| WO2015067800A1 (en) | 2013-11-11 | 2015-05-14 | Basf Se | Antifungal penicillium strains, fungicidal extrolites thereof, and their use |
| WO2015155075A1 (en) | 2014-04-11 | 2015-10-15 | Syngenta Participations Ag | Fungicidal n'-[2-methyl-6-[2-alkoxy-ethoxy]-3-pyridyl]-n-alkyl-formamidine derivatives for use in agriculture |
| WO2015197503A1 (en) | 2014-06-25 | 2015-12-30 | F. Hoffmann-La Roche Ag | Imidazo[1,2-a]pyrazin-1yl-benzamide compounds for treating spinal muscular atrophy |
| US9242996B2 (en) | 2013-11-01 | 2016-01-26 | Novartis Ag | Aminoheteroaryl benzamides as kinase inhibitors |
| WO2016020371A1 (en) | 2014-08-04 | 2016-02-11 | Basf Se | Antifungal paenibacillus strains, fusaricidin-type compounds, and their use |
| WO2016154297A1 (en) | 2015-03-26 | 2016-09-29 | Bayer Cropscience Lp | A novel paenibacillus strain, antifungal compounds, and methods for their use |
| WO2016156085A1 (en) | 2015-03-27 | 2016-10-06 | Syngenta Participations Ag | Microbiocidal heterobicyclic derivatives |
| WO2016156290A1 (en) | 2015-04-02 | 2016-10-06 | Bayer Cropscience Aktiengesellschaft | Novel 5-substituted imidazole derivatives |
| WO2016202742A1 (en) | 2015-06-15 | 2016-12-22 | Bayer Cropscience Aktiengesellschaft | Halogen-substituted phenoxyphenylamidines and the use thereof as fungicides |
| WO2017019448A1 (en) | 2015-07-24 | 2017-02-02 | AgBiome, Inc. | Modified biological control agents and their uses |
| WO2017025510A1 (en) | 2015-08-12 | 2017-02-16 | Syngenta Participations Ag | Microbiocidal heterobicyclic derivatives |
| WO2017029179A1 (en) | 2015-08-14 | 2017-02-23 | Bayer Cropscience Aktiengesellschaft | Triazole derivatives, intermediates thereof and their use as fungicides |
| WO2017055473A1 (en) | 2015-10-02 | 2017-04-06 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017055469A1 (en) | 2015-10-02 | 2017-04-06 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017066094A1 (en) | 2015-10-12 | 2017-04-20 | Pioneer Hi-Bred International, Inc. | Biologicals and their use in plants |
| WO2017093348A1 (en) | 2015-12-02 | 2017-06-08 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017118689A1 (en) | 2016-01-08 | 2017-07-13 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017153380A1 (en) | 2016-03-10 | 2017-09-14 | Syngenta Participations Ag | Microbiocidal quinoline (thio)carboxamide derivatives |
| WO2017205258A1 (en) | 2016-05-26 | 2017-11-30 | Novozymes Bioag A/S | Bacillus and lipochitooligosaccharide for improving plant growth |
| WO2017220485A1 (en) | 2016-06-21 | 2017-12-28 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2018065414A1 (en) | 2016-10-06 | 2018-04-12 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2018153707A1 (en) | 2017-02-22 | 2018-08-30 | Basf Se | Crystalline forms of a strobilurin type compound for combating phytopathogenic fungi |
| WO2018158365A1 (en) | 2017-03-03 | 2018-09-07 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2018202428A1 (en) | 2017-05-02 | 2018-11-08 | Basf Se | Fungicidal mixture comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles |
| WO2018228896A1 (en) | 2017-06-14 | 2018-12-20 | Syngenta Participations Ag | Fungicidal compositions |
| WO2019110427A1 (en) | 2017-12-04 | 2019-06-13 | Syngenta Participations Ag | Microbiocidal phenylamidine derivatives |
| WO2020056090A1 (en) | 2018-09-14 | 2020-03-19 | Fmc Corporation | Fungicidal halomethyl ketones and hydrates |
| WO2020079111A1 (en) | 2018-10-18 | 2020-04-23 | Syngenta Crop Protection Ag | Microbiocidal compounds |
| WO2020097012A1 (en) | 2018-11-06 | 2020-05-14 | Fmc Corporation | Substituted tolyl as fungicides |
| WO2020109391A1 (en) | 2018-11-28 | 2020-06-04 | Bayer Aktiengesellschaft | Pyridazine (thio)amides as fungicidal compounds |
| WO2020193387A1 (en) | 2019-03-22 | 2020-10-01 | Syngenta Crop Protection Ag | Fungicidal compounds |
-
2024
- 2024-04-09 AR ARP240100867A patent/AR132351A1/en unknown
- 2024-04-10 TW TW113113352A patent/TW202446260A/en unknown
- 2024-04-11 WO PCT/EP2024/059875 patent/WO2024213656A1/en not_active Ceased
- 2024-04-11 CN CN202480022192.8A patent/CN121013850A/en active Pending
- 2024-04-11 KR KR1020257037541A patent/KR20250170124A/en active Pending
- 2024-04-11 AU AU2024251684A patent/AU2024251684A1/en active Pending
-
2025
- 2025-09-28 IL IL323645A patent/IL323645A/en unknown
- 2025-10-03 MX MX2025011849A patent/MX2025011849A/en unknown
- 2025-11-06 CO CONC2025/0015496A patent/CO2025015496A2/en unknown
Patent Citations (69)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2016A (en) | 1841-03-26 | Mode of constructing fireplaces and chimney-staoks ii | ||
| EP0451878A1 (en) | 1985-01-18 | 1991-10-16 | Plant Genetic Systems, N.V. | Modifying plants by genetic engineering to combat or control insects |
| US5061495A (en) | 1988-03-07 | 1991-10-29 | Agricultural Genetics Company Limited | Antibiotic derived from b. subtilis |
| EP0353191A2 (en) | 1988-07-29 | 1990-01-31 | Ciba-Geigy Ag | DNA sequences encoding polypeptides having beta-1,3-glucanase activity |
| EP0367474A1 (en) | 1988-11-01 | 1990-05-09 | Mycogen Corporation | Novel bacillus thuringiensis isolate denoted b.t. ps81gg, active against lepidopteran pests, and a gene encoding a lepidopteran-active toxin |
| EP0374753A2 (en) | 1988-12-19 | 1990-06-27 | American Cyanamid Company | Insecticidal toxines, genes coding therefor, antibodies binding them, transgenic plant cells and plants expressing these toxines |
| EP0392225A2 (en) | 1989-03-24 | 1990-10-17 | Ciba-Geigy Ag | Disease-resistant transgenic plants |
| WO1990013651A1 (en) | 1989-05-09 | 1990-11-15 | Imperial Chemical Industries Plc | Bacterial genes |
| EP0401979A2 (en) | 1989-05-18 | 1990-12-12 | Mycogen Corporation | Novel bacillus thuringiensis isolates active against lepidopteran pests, and genes encoding novel lepidopteran-active toxins |
| EP0427529A1 (en) | 1989-11-07 | 1991-05-15 | Pioneer Hi-Bred International, Inc. | Larvicidal lectins and plant insect resistance based thereon |
| WO1993007278A1 (en) | 1991-10-04 | 1993-04-15 | Ciba-Geigy Ag | Synthetic dna sequence having enhanced insecticidal activity in maize |
| WO1995033818A2 (en) | 1994-06-08 | 1995-12-14 | Ciba-Geigy Ag | Genes for the synthesis of antipathogenic substances |
| WO1995034656A1 (en) | 1994-06-10 | 1995-12-21 | Ciba-Geigy Ag | Novel bacillus thuringiensis genes coding toxins active against lepidopteran pests |
| WO1997033890A1 (en) | 1996-03-11 | 1997-09-18 | Novartis Ag | Pyrimidin-4-one derivatives as pesticide |
| US6060051A (en) | 1997-05-09 | 2000-05-09 | Agraquest, Inc. | Strain of bacillus for controlling plant diseases and corn rootworm |
| JPH11253151A (en) | 1997-11-13 | 1999-09-21 | Kumiai Chem Ind Co Ltd | Rice seedling raising disease control agent |
| WO2000015615A1 (en) | 1998-09-15 | 2000-03-23 | Syngenta Participations Ag | Pyridine ketones useful as herbicides |
| US6245551B1 (en) | 1999-03-30 | 2001-06-12 | Agraquest, Inc. | Strain of Bacillus pumilus for controlling plant diseases caused by fungi |
| WO2002015701A2 (en) | 2000-08-25 | 2002-02-28 | Syngenta Participations Ag | Bacillus thuringiensis crystal protein hybrids |
| WO2002048146A2 (en) * | 2000-12-13 | 2002-06-20 | Basf Aktiengesellschaft | Use of substituted imidazoazines, novel imidazoazines, methods for the production thereof, and agents containing these compounds |
| WO2003000051A2 (en) | 2001-06-22 | 2003-01-03 | Drahos David J | Novel biofungicide |
| WO2003018810A2 (en) | 2001-08-31 | 2003-03-06 | Syngenta Participations Ag | Modified cry3a toxins and nucleic acid sequences coding therefor |
| US7094592B2 (en) | 2001-11-26 | 2006-08-22 | Kumiai Chemical Industry Co., Ltd. | Bacillus sp. D747 strain, plant disease controlling agents and insect pest controlling agents using the same and control method using the agents |
| WO2003052073A2 (en) | 2001-12-17 | 2003-06-26 | Syngenta Participations Ag | Novel corn event |
| US6919298B2 (en) | 2002-04-04 | 2005-07-19 | Valent Biosciences Corporation | Enhanced herbicide composition |
| US7579183B1 (en) | 2006-12-01 | 2009-08-25 | The United States Of America As Represented By The Secretary Of Agriculture | Saprophytic yeast, Pichia anomala |
| US20100291039A1 (en) | 2007-12-14 | 2010-11-18 | Kohl Jurgen Anton | Novel micro-organisms controlling plant pathogens |
| WO2009116106A1 (en) | 2008-03-21 | 2009-09-24 | Trentino Sviluppo S.P.A. | Trichoderma atroviride sc1 for biocontrol of fungal diseases in plants |
| US8431120B2 (en) | 2008-03-21 | 2013-04-30 | Trentino Sviluppo S.P.A. | Trichoderma atroviride SC1 for biocontrol of fungal diseases in plants |
| WO2010086790A1 (en) | 2009-01-27 | 2010-08-05 | Lesaffre Et Compagnie | Saccharomyces cerevisiae strains with phytosanitary capabilities |
| WO2011106491A2 (en) | 2010-02-25 | 2011-09-01 | Marrone Bio Innovations, Inc. | Isolated bacterial strain of the genus burkholderia and pesticidal metabolites therefrom |
| WO2011138281A2 (en) | 2010-05-06 | 2011-11-10 | Bayer Cropscience Ag | Process for the preparation of dithiine tetracarboxydiimides |
| WO2011151819A2 (en) | 2010-06-01 | 2011-12-08 | Yissum Research Development Company Of The Hebrew University Of Jerusalem Ltd. | Pseudozyma aphidis as a biocontrol agent against various plant pathogens |
| WO2013032693A2 (en) | 2011-08-27 | 2013-03-07 | Marrone Bio Innovations, Inc. | Isolated bacterial strain of the genus burkholderia and pesticidal metabolites therefrom-formulations and uses |
| WO2013034938A2 (en) | 2011-09-08 | 2013-03-14 | Szegedi Tudományegyetem | A copper resistant, fengycin-producing bacillus mojavensis strain for controlling vegetable pathogens, its use and compositions containing it |
| WO2014006945A1 (en) | 2012-07-04 | 2014-01-09 | アグロカネショウ株式会社 | 2-aminonicotinic acid ester derivative and bactericide containing same as active ingredient |
| WO2014028521A1 (en) | 2012-08-14 | 2014-02-20 | Marrone Bio Innovations, Inc. | Bacillus sp. strain with antifungal, antibacterial and growth promotion activity |
| WO2014095675A1 (en) | 2012-12-19 | 2014-06-26 | Bayer Cropscience Ag | Difluoromethyl-nicotinic-indanyl carboxamides as fungicides |
| US9242996B2 (en) | 2013-11-01 | 2016-01-26 | Novartis Ag | Aminoheteroaryl benzamides as kinase inhibitors |
| WO2015067800A1 (en) | 2013-11-11 | 2015-05-14 | Basf Se | Antifungal penicillium strains, fungicidal extrolites thereof, and their use |
| WO2015155075A1 (en) | 2014-04-11 | 2015-10-15 | Syngenta Participations Ag | Fungicidal n'-[2-methyl-6-[2-alkoxy-ethoxy]-3-pyridyl]-n-alkyl-formamidine derivatives for use in agriculture |
| WO2015197503A1 (en) | 2014-06-25 | 2015-12-30 | F. Hoffmann-La Roche Ag | Imidazo[1,2-a]pyrazin-1yl-benzamide compounds for treating spinal muscular atrophy |
| WO2016020371A1 (en) | 2014-08-04 | 2016-02-11 | Basf Se | Antifungal paenibacillus strains, fusaricidin-type compounds, and their use |
| WO2016154297A1 (en) | 2015-03-26 | 2016-09-29 | Bayer Cropscience Lp | A novel paenibacillus strain, antifungal compounds, and methods for their use |
| WO2016156085A1 (en) | 2015-03-27 | 2016-10-06 | Syngenta Participations Ag | Microbiocidal heterobicyclic derivatives |
| WO2016156290A1 (en) | 2015-04-02 | 2016-10-06 | Bayer Cropscience Aktiengesellschaft | Novel 5-substituted imidazole derivatives |
| WO2016202742A1 (en) | 2015-06-15 | 2016-12-22 | Bayer Cropscience Aktiengesellschaft | Halogen-substituted phenoxyphenylamidines and the use thereof as fungicides |
| WO2017019448A1 (en) | 2015-07-24 | 2017-02-02 | AgBiome, Inc. | Modified biological control agents and their uses |
| WO2017025510A1 (en) | 2015-08-12 | 2017-02-16 | Syngenta Participations Ag | Microbiocidal heterobicyclic derivatives |
| WO2017029179A1 (en) | 2015-08-14 | 2017-02-23 | Bayer Cropscience Aktiengesellschaft | Triazole derivatives, intermediates thereof and their use as fungicides |
| WO2017055469A1 (en) | 2015-10-02 | 2017-04-06 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017055473A1 (en) | 2015-10-02 | 2017-04-06 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017066094A1 (en) | 2015-10-12 | 2017-04-20 | Pioneer Hi-Bred International, Inc. | Biologicals and their use in plants |
| WO2017093348A1 (en) | 2015-12-02 | 2017-06-08 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017118689A1 (en) | 2016-01-08 | 2017-07-13 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017153380A1 (en) | 2016-03-10 | 2017-09-14 | Syngenta Participations Ag | Microbiocidal quinoline (thio)carboxamide derivatives |
| WO2017205258A1 (en) | 2016-05-26 | 2017-11-30 | Novozymes Bioag A/S | Bacillus and lipochitooligosaccharide for improving plant growth |
| WO2017220485A1 (en) | 2016-06-21 | 2017-12-28 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2018065414A1 (en) | 2016-10-06 | 2018-04-12 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2018153707A1 (en) | 2017-02-22 | 2018-08-30 | Basf Se | Crystalline forms of a strobilurin type compound for combating phytopathogenic fungi |
| WO2018158365A1 (en) | 2017-03-03 | 2018-09-07 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2018202428A1 (en) | 2017-05-02 | 2018-11-08 | Basf Se | Fungicidal mixture comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles |
| WO2018228896A1 (en) | 2017-06-14 | 2018-12-20 | Syngenta Participations Ag | Fungicidal compositions |
| WO2019110427A1 (en) | 2017-12-04 | 2019-06-13 | Syngenta Participations Ag | Microbiocidal phenylamidine derivatives |
| WO2020056090A1 (en) | 2018-09-14 | 2020-03-19 | Fmc Corporation | Fungicidal halomethyl ketones and hydrates |
| WO2020079111A1 (en) | 2018-10-18 | 2020-04-23 | Syngenta Crop Protection Ag | Microbiocidal compounds |
| WO2020097012A1 (en) | 2018-11-06 | 2020-05-14 | Fmc Corporation | Substituted tolyl as fungicides |
| WO2020109391A1 (en) | 2018-11-28 | 2020-06-04 | Bayer Aktiengesellschaft | Pyridazine (thio)amides as fungicidal compounds |
| WO2020193387A1 (en) | 2019-03-22 | 2020-10-01 | Syngenta Crop Protection Ag | Fungicidal compounds |
Non-Patent Citations (10)
| Title |
|---|
| "The Pesticide Manual", 2021, BRITISH CROP PROTECTION COUNCIL |
| CAN JOUR PLANT SCI, vol. 83, no. 3, 2003, pages 519 - 524 |
| CHEM. SOC. REV., vol. 38, 2009, pages 606 |
| CHEM. SOC. REV., vol. 40, 2011, pages 5084 |
| CROP PROTECTION, vol. 25, no. 2643947-26-4, 2006, pages 468 - 475 |
| J. MED. CHEM., vol. 32, no. 12, 1989, pages 2561 - 73 |
| JENSEN DF ET AL.: "Development of a biocontrol agent for plant disease control with special emphasis on the near commercial fungal antagonist Clonostachys rosea strain 'IK726", AUSTRALASIAN PLANT PATHOL., vol. 36, no. 2, 2007, pages 95 - 101 |
| no. 1610666-95-9 |
| ORG. LETT., vol. 22, 2020, pages 2271 |
| PIETR ET AL., ZESZ. NAUK. A R W SZCZECINIE, vol. 161, 1993, pages 125 - 137 |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2024251684A1 (en) | 2025-10-09 |
| CO2025015496A2 (en) | 2025-11-28 |
| CN121013850A (en) | 2025-11-25 |
| IL323645A (en) | 2025-11-01 |
| MX2025011849A (en) | 2025-11-03 |
| TW202446260A (en) | 2024-12-01 |
| KR20250170124A (en) | 2025-12-04 |
| AR132351A1 (en) | 2025-06-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2022364034B2 (en) | Imidazo[1,2-a]pyridine derivatives | |
| WO2021244951A9 (en) | Fungicidal compositions | |
| WO2024126407A1 (en) | Benzimidazole derivatives | |
| AU2022325463B2 (en) | Fungicide mixture | |
| EP4634178A1 (en) | Imidazo[1,2-a]pyridine derivatives | |
| WO2024213656A1 (en) | Imidazo[1,2-a]pyrazine derivatives | |
| WO2024213662A1 (en) | Pyrazolo[1,5-a]pyridine derivatives | |
| WO2024213653A1 (en) | Imidazo[1,2-a]pyridine derivatives | |
| WO2024213659A1 (en) | Imidazo[1,2-a]pyrazine derivatives | |
| AU2024251657A1 (en) | Imidazo bicyclic derivatives | |
| WO2024213663A1 (en) | Pyrazolo[1,5-a]pyridine derivatives | |
| AU2024300580A1 (en) | Benzimidazole derivatives | |
| WO2024213651A1 (en) | Imidazo[1,2-a]pyridine derivatives | |
| WO2024213650A1 (en) | Imidazo[1,2-a]pyridine derivatives | |
| WO2025257024A1 (en) | Fungicidal compositions | |
| WO2024213720A1 (en) | Fungicidal compositions | |
| WO2025257023A1 (en) | Fungicidal compositions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24718479 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: AU2024251684 Country of ref document: AU Ref document number: 825293 Country of ref document: NZ |
|
| WWP | Wipo information: published in national office |
Ref document number: 825293 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 323645 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2025/011849 Country of ref document: MX |
|
| ENP | Entry into the national phase |
Ref document number: 2024251684 Country of ref document: AU Date of ref document: 20240411 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2501006955 Country of ref document: TH |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112025022178 Country of ref document: BR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202517101737 Country of ref document: IN |
|
| WWP | Wipo information: published in national office |
Ref document number: MX/A/2025/011849 Country of ref document: MX |
|
| ENP | Entry into the national phase |
Ref document number: 1020257037541 Country of ref document: KR Free format text: ST27 STATUS EVENT CODE: A-0-1-A10-A15-NAP-PA0105 (AS PROVIDED BY THE NATIONAL OFFICE) |
|
| WWE | Wipo information: entry into national phase |
Ref document number: KR1020257037541 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202592941 Country of ref document: EA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2024718479 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2024718479 Country of ref document: EP Effective date: 20251113 |
|
| ENP | Entry into the national phase |
Ref document number: 2024718479 Country of ref document: EP Effective date: 20251113 |
|
| ENP | Entry into the national phase |
Ref document number: 2024718479 Country of ref document: EP Effective date: 20251113 |
|
| ENP | Entry into the national phase |
Ref document number: 2024718479 Country of ref document: EP Effective date: 20251113 |
|
| WWP | Wipo information: published in national office |
Ref document number: 202517101737 Country of ref document: IN |
|
| ENP | Entry into the national phase |
Ref document number: 2024718479 Country of ref document: EP Effective date: 20251113 |