INHIBITING α
vβ
8 INTEGRIN RELATED APPLICATION [0001] This application claims the benefit of priority to U.S. Provisional Patent Application No.63/484,763, filed February 14, 2023, the content of which is incorporated herein by reference in its entirety. TECHNICAL FIELD [0002] This disclosure relates to novel chemical compounds and methods useful for inhibiting α
vβ
8 integrin. BACKGROUND [0003] In the tumor microenvironment α
vβ
8 is expressed on immune cells, mainly antigen presenting cells (APCs) and regulatory T cells (Tregs) as well as on the tumor cells and cancer associated fibroblast. The major function of αvβ8 is activation of growth factor TGF-β1 and 3. The TGF-βs are biosynthesized and stored in tissues as latent forms. The TGF-β homodimer is kept latent by association with its dimeric prodomain (pro-TGF-β). The prodomain-derived homodimer prevents TGF-β from binding TGF-β receptor and is called latency-associated peptide (LAP). The latent TGF-β complex is stored in the extracellular matrix or on the cell surface for subsequent, integrin-dependent activation. The integrin αvβ8 heterodimer binds arginine–glycine–aspartic acid motifs (RGD domain) of the latent- TGF-β-1 and 3 to liberate the active TGF-β cytokine from latent complex. [0004] TGF-β is a pleiotropic cytokine mediating multiple biological process including development and homeostasis. TGF-β is a key player in cell growth, differentiation, and apoptosis. It regulates extracellular matrix (ECM) production contributing to tissue repair processes. In the immune system TGF-β is necessary for the development of distinct immune cell types, as well to promote immunosuppression. Homeostatic role of TGF-β on immunity is critical to prevent excessive inflammatory responses and essential for maintaining tolerance to self- antigens to prevent autoimmunity. As TGF-β is ubiquitously expressed it activity needs to be tightly regulated and when exacerbated lead to disease states. Dysregulation of TGF-β signaling is involved in multiple disorders, especially cancer and fibrosis. [0005] In cancer, the TGF-β pathway has been implicated in many human neoplastic diseases, including solid and hematopoietic tumors. As a potent inhibitor of cell proliferation,
TGF-β acts as a tumor suppressor; however, in tumor cells, TGF-β loses its anti-proliferative response and promotes cancer progression. The TGF-β-promoted tumorigenesis is mainly driven by downregulation of anti-tumor immunity. The immunosuppressive effect leads to tumor immune tolerance. Additionally, TGF-β facilitates epithelial to mesenchymal transition (EMT) and angiogenesis to increase tumor invasiveness. The integrin avb8 expression in cancers correlates with TGF-β activity. It modulates inflammatory phenotype of the APCs and Tregs, the main cell types fundamental for T/NK cell driven anti-tumor activity. The α
vβ
8 integrin locally activates TGF-β to regulate cross-talk between APCs and effector cells to skew immunity from inflammation to tolerance. [0006] Inhibition of the integrin αvβ8-driven TGF-β activation is proposed to reverse tumor tolerance and enhance anti-tumor T/NK cell responses. In agreement with TGF-β blockage, avb8 inhibition can enhance outcomes in checkpoint inhibitors regimens or reverse checkpoint inhibitor resistance. [0007] Ubiquitous expression and multifunctionality of TGF-β limits application of strategies based on TGF-β systemic blockage, as those approaches result is unwanted side effects. Blockage of TGF-β activity by antagonizing the integrin αvβ8 increases the safety and has therapeutic advantage over global TGF-β inhibition. It allows for tissue localized and isoform selective TGF- β blockage in specific immunological milieu. Therefore, there remains a need for therapeutic compounds for inhibiting α
vβ
8 integrin. SUMMARY OF THE INVENTION [0008] This disclosure relates to novel chemical compounds and methods useful for inhibiting αvβ8 integrin. [0009] In one aspect, the invention features a compound of Formula (I), or a pharmaceutically acceptable salt thereof:
wherein:
h is optionally substituted with 1 to 6 R
4; each of R
1a, R
1b, R
1c, R
1d, R
1e, and R
1f is independently H, C
1-4alkyl, halogen, C
1- 4alkyloxy, OH, C
1-4alkyl-OH, C
1-4alkyl-C
1-4alkyloxy, C
1-4alkyloxy-C
1-4alkyloxy, CF
3, CHF
2, CH
2F, CN, NO
2, NR
aR
b or C
1-4alkyl-NR
aR
b, R
2 is H, C1-4alkyl or C3-5cycloalkyl, each of C1-4alkyl and C3-5cycloalkyl is optionally substituted with 1 to 4 R
5; R
3a is C1-4alkyloxy, C3-5cycloalkoxy, CF3, CHF2, CH2F, OCF3, OCHF2 or OCH2F; R
3b is H, halogen, CF
3 or CN; R
3c is H, F, CN, or C
1-4alkyl; R
3d is H, C
1-4alkyl, C
3-5cycloalkyl, or 4-6-membered heterocycloalkyl, each of which is optionally substituted with 1 to 4 R
6; R
3e is H or F; each R
4 is independently H, C1-4alkyl, halogen, CF3, CHF2 or CH2F, cyclopropyl, or two geminal R
4 groups together can form a spiro-cyclopropyl; each R
5 is independently H, C
1-4alkyl, C
3-5cycloalkyl, C
1-4alkyloxy, C
3-5cycloalkoxy, F, CF
3, CHF
2, CH
2F, OCF
3, OCHF
2 or OCH
2F; each R
6 is independently H, C1-4alkyl, C2-4alkenyl, C3-5cycloalkyl, C1-4alkyloxy, C3- 5cycloalkoxy, F, CF3, CHF2, CH2F, OCF3, OCHF2, OCH2F, OH, 5-6-membered heteroaryl, or NRaRb; each R
7 is independently C
1-4alkyl or F; each of R
a and R
b is independently hydrogen, C
1-4alkyl, C
3-5cycloalkyl, or R
a and R
b, together with the nitrogen atom to which they are attached, form a saturated or unsaturated heterocyclic ring containing from three to seven ring atoms, which ring may optionally contain
additional one or two heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur and may be optionally substituted by from one to three groups which may be the same or different selected from the group consisting of F, C1-4alkyl, phenyl and benzyl; and n is 0, 1 or 2; and m is 0, 1 or 2. [0010] In one aspect, the invention features a compound of Formula (I), or a pharmaceutically acceptable salt thereof:
wherein:
h is optionally substituted with 1 to 6 R
4; each of R
1a, R
1b, R
1c, R
1d, R
1e, and R
1f is independently H, C1-4alkyl, halogen, C1-
4alkyloxy, OH, C
1-4alkyl-OH, C
1-4alkyl-C
1-4alkyloxy, C
1-4alkyloxy-C
1-4alkyloxy, CF
3, CHF
2, CH
2F, CN, NO
2, NR
aR
b or C
1-4alkyl-NR
aR
b, R
2 is H, C
1-4alkyl or C
3-5cycloalkyl, each of C
1-4alkyl and C
3-5cycloalkyl is optionally substituted with 1 to 4 R
5; R
3a is cyano, halogen, C1-4alkyl, C1-4alkyloxy, C3-5cycloalkoxy, CF3, CHF2, CH2F, OCF3, OCHF2 or OCH2F; R
3b is H, halogen, CF
3 or CN; R
3c is H, F, CN, or C
1-4alkyl;
R
3d is H, C
1-4alkyl, C
3-6cycloalkyl, or 4-6-membered heterocycloalkyl, each of which is optionally substituted with 1 to 4 R
6; R
3e is H or F; each R
4 is independently H, C1-4alkyl, halogen, CF3, CHF2 or CH2F, cyclopropyl, or two geminal R
4 groups together can form a spiro-cyclopropyl; each R
5 is independently H, C
1-4alkyl, C
3-5cycloalkyl, C
1-4alkyloxy, C
3-5cycloalkoxy, F, CF
3, CHF
2, CH
2F, OCF
3, OCHF
2 or OCH
2F; each R
6 is independently H, C
1-4alkyl, C
2-4alkenyl, C
3-5cycloalkyl, C
1-4alkyloxy, C
3- 5cycloalkoxy, F, CF3, CHF2, CH2F, OCF3, OCHF2, OCH2F, OH, 5-6-membered heteroaryl, or NRaRb; each R
7 is independently C1-4alkyl or F; each of R
a and R
b is independently hydrogen, C
1-4alkyl, C
3-5cycloalkyl, or R
a and R
b, together with the nitrogen atom to which they are attached, form a saturated or unsaturated heterocyclic ring containing from three to seven ring atoms, which ring may optionally contain additional one or two heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur and may be optionally substituted by from one to three groups which may be the same or different selected from the group consisting of F, C1-4alkyl, phenyl and benzyl; and n is 0, 1 or 2; and m is 0, 1 or 2. [0011] In embodiments, m is 0 and each of R
1a, R
1c, R
1d, R
1e and R
1f is independently H; and R
1b is H or methoxy. [0012] In embodiments, L is
optionally substituted with 1 to 2 R
4; and each R
4 is independently H, methyl, F, CF3, CHF2 or CH2F. [0013] In embodiments, R
2 is C
1-4alkyl. [0014] In embodiments, R
3c is H, and R
3e is H. [0015] In embodiments, R
3a is OMe. [0016] In embodiments, R
3b is F. [0017] In embodiments, R
3d is C1-4alkyl.
[0018] In embodiments,
each of R
1a, R
1b, R
1c, R
1d, R
1e, and R
1f is independently H, R
2 is C
1-4alkyl or cyclopropyl, and each of C
1-4alkyl is optionally substituted with 1 to 4 R
5; R
3a is OMe; R
3b is F; R
3c is H or F; R
3d is C
1-4alkyl, C
3-5cycloalkyl, or 4-6-membered heterocycloalkyl, each of which is optionally substituted with 1 to 4 R
6; R
3e is H; each R
4 is independently F or CH3; each R
5 is independently cyclopropyl, C1-4alkyloxy, F, CF3, CHF2, CH2F, OCF3, OCHF2 or OCH
2F; n is 0, 1 or 2; and m is 0. [0019] In embodiments, the compound has a structure according to Formula (III),
pharmaceutically acceptable salt thereof. In embodiments, the carbon marked by the asterisk has the (R)-configuration. In embodiments, the carbon marked by the asterisk has the (S)-configuration.
[0020] In embodiments, the Q ring i
[0021] In embodiments, each of R
1c, R
1d, and R
1e is independently H. [0022] In embodiments, each R
1a is independently H. [0023] In embodiments, each R
1b is independently H. [0024] In embodiments, each R
1b is independently OMe. [0025] In embodiments, the Q ring i
[0026] In embodiments, each of R
1c and R
1d is independently H. [0027] In embodiments, each of R
1a, R
1b and R
1f is independently H. [0028] In embodiments,
[0029] In embodiments,
[0030] In embodiments, R
2 is C1-4alkyl. [0031] In embodiments, R
2 is methyl. [0032] In embodiments, R
2 is ethyl. [0033] In embodiments, R
2 is cyclopropyl. [0034] In embodiments, R
3a is C1-4alkyl. [0035] In embodiments, R
3a is ethyl. [0036] In embodiments, R
3a is methyl. [0037] In embodiments, R
3a is CN. [0038] In embodiments, R
3a is halogen. [0039] In embodiments, R
3a is Cl. [0040] In embodiments, R
3a is F.
[0041] In embodiments, R
3a is C
1-4alkyl optionally substituted with one or more halogen. [0042] In embodiments, R
3a is C
1-4alkyl optionally substituted with one or more F. [0043] In embodiments, R
3a is methyl optionally substituted with one or more F. [0044] In embodiments, R
3a is C1-4alkyloxy. [0045] In embodiments, R
3a is OMe, OEt, OCF3, OCHF2 or OCH2F. [0046] In embodiments, R
3a is OMe. [0047] In embodiments, R
3b is F. [0048] In embodiments, R
3c is H. [0049] In embodiments, R
3d is C1-4alkyl. [0050] In embodiments, R
3d is C3-5cycloalkyl. [0051] In embodiments, R
3d is oxetanyl, tetrahydrofuranyl, tetrahydro-2H-pyranyl, morpholinyl or piperazinyl-C
1-4alkyl. [0052] In embodiments, R
3d is C
6 cyckoalkyl optionally substituted with 1 to 4 R
6. [0053] In embodiments, R
3d is C
3-6 cyckoalkyl optionally substituted with 1 to 4 R
6. [0054] In embodiments, R
3d is iso-propyl. [0055] In embodiments, R
3e is H. [0056] In embodiments, each R
4 is independently methyl. [0057] In embodiments, each R
4 is independently F. [0058] In embodiments, each R
4 is independently CF
3, CHF
2 or CH
2F. [0059] In embodiments, each R
4 is independently H. [0060] In embodiments, each R
5 is independently C1-4alkyl. [0061] In embodiments, each R
5 is independently C3-5cycloalkyl. [0062] In embodiments, each R
5 is independently C1-4alkyloxy. [0063] In embodiments, each R
5 is independently C
3-5cycloalkoxy. [0064] In embodiments, each R
5 is independently F, CF
3, CHF
2, CH
2F, OCF
3, OCHF
2 or OCH
2F.
[0065] In embodiments, each R
5 is independently H. [0066] In embodiments, each R
6 is independently H. [0067] In embodiments, each R
6 is independently C1-4alkyl. [0068] In embodiments, each R
6 is independently C2-4alkenyl. [0069] In embodiments, each R
6 is independently C3-5cycloalkyl. [0070] In embodiments, each R
6 is independently C
1-4alkyloxy. [0071] In embodiments, each R
6 is independently C
3-5cycloalkoxy. [0072] In embodiments, each R
6 is independently F, CF
3, CHF
2, CH
2F, OCF
3, OCHF
2, OCH2F or OH. [0073] In embodiments, each R
6 is independently a 5-6-membered heteroaryl. [0074] In embodiments, each R
6 is independently H. [0075] In embodiments, each R
7 is independently methyl or F. [0076] In embodiments, n is 0. [0077] In embodiments, n is 1. [0078] In embodiments, n is 2. [0079] In embodiments, m is 0. [0080] In embodiments, a compound has a structure according to Formula (II),
pharmaceutically acceptable salt thereof.
[0081] In embodiments, a compound has a structure according to Formula (IIA),
pharmaceutically acceptable salt thereof. [0082] In embodiments, a compound has a structure according to Formula (IIB),
pharmaceutically acceptable salt thereof. [0083] In embodiments, a compound has a structure according to Formula (III),
pharmaceutically acceptable salt thereof. [0084] In embodiments, a compound has a structure according to Formula (IIIA),
pharmaceutically acceptable salt thereof.
[0085] In embodiments, a compound has a structure according to Formula (IIIB),
pharmaceutically acceptable salt thereof. [0086] In embodiments, a compound has a structure according to Formula (IV),
pharmaceutically acceptable salt thereof. [0087] In embodiments, a compound has a structure according to Formula (IVA),
pharmaceutically acceptable salt thereof. [0088] In embodiments, a compound has a structure according to Formula (IVB),
pharmaceutically acceptable salt thereof.
[0089] In embodiments, a compound has a structure according to Formula (V),
pharmaceutically acceptable salt thereof. [0090] In embodiments, a compound has a structure according to Formula (VA),
pharmaceutically acceptable salt thereof. [0091] In embodiments, a compound has a structure according to Formula (VB),
pharmaceutically acceptable salt thereof. [0092] In embodiments, a compound has a structure according to Formula (VI),
pharmaceutically acceptable salt thereof. [0093] In embodiments, the carbon marked by the asterisk has the (R)-configuration. [0094] In embodiments, the carbon marked by the asterisk has the (S)-configuration. [0095] In embodiments, wherein R
2 is C
1-4alkyl optionally substituted with 1 or 2 R
5. [0096] In embodiments, R
2 is –CH
3, -CH
2CH
3, -CH(CH
3)
2, -C(CH
3)
3, -CH2CH(CH3)2, -CH2CF3, -(CH2)2CF3, -CHF2, -CH2CHF2, -(CH2)2CHF2,
-(CH
2)
2OCH
3, -(CH
2)
2O-(cyclopropyl), -(CH
2)
2OCF
3, -(CH
2)
2C(OCH
3)(CH
3)CH
3, or cyclopropyl. [0097] In embodiments, R
2 is –CH3. [0098] In embodiments, R
3c is H. [0099] In embodiments, R
3c is F. [0100] In embodiments, R
3b is F. [0101] In embodiments, R
3d is isopropyl. [0102] In embodiments, L is unsubstituted
. [0103] In embodiments, L is
substituted by 1 or 2 R
5. [0104] In embodiments, each R
5 is independently selected from -CH3, -CH2F, and –F. [0105] In embodiments, L is unsubstituted
. [0106] In embodiments, the compound is selected from any compound described in Table 1, or a pharmaceutically acceptable salt thereof. [0107] In embodiments, the compound is selected from the group consisting of:
or a pharmaceutically acceptable salt thereof. [0108] In embodiments, a compound of Formula (I) is
pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 1A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 1B, or a pharmaceutically acceptable salt thereof. [0109] In embodiments, a compound of Formula (I) is
salt thereof. In embodiments, a compound is Compound 7A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 7B, or a pharmaceutically acceptable salt thereof.
[0110] In embodiments, a compound of Formula (I) is
salt thereof. In embodiments, a compound is Compound 8A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 8B, or a pharmaceutically acceptable salt thereof. [0111] In embodiments, a compound of Formula (I) is
pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 18A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 18B, or a pharmaceutically acceptable salt thereof. [0112] In embodiments, a compound of Formula (I) is
pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 28A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 28B, or a pharmaceutically acceptable salt thereof.
[0113] In embodiments, a compound of Formula (I) is
pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 30A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 30B, or a pharmaceutically acceptable salt thereof. [0114] In embodiments, a compound of Formula (I) is
pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 100A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 100B, or a pharmaceutically acceptable salt thereof. [0115] In embodiments, a compound of Formula (I) is
pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 101A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 101B, or a pharmaceutically acceptable salt thereof.
[0116] In embodiments, a compound of Formula (I) is
pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 102A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 102B, or a pharmaceutically acceptable salt thereof. [0117] In embodiments, a compound of Formula (I) is
pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 103A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 103B, or a pharmaceutically acceptable salt thereof. [0118] In embodiments, a compound of Formula (I) is
pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 110A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 110B, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 110C, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 110D, or a pharmaceutically acceptable salt thereof.
[0119] In embodiments, a compound of Formula (I) is
pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 112A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 112B, or a pharmaceutically acceptable salt thereof. [0120] In embodiments, a compound of Formula (I) is
salt thereof. In embodiments, a compound is Compound 113A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 113B, or a pharmaceutically acceptable salt thereof. [0121] In embodiments, a compound of Formula (I) is
pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 114A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 114B, or a pharmaceutically acceptable salt thereof.
[0122] In embodiments, a compound of Formula (I) is
pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 117A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 117B, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 117C, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 117D, or a pharmaceutically acceptable salt thereof. [0123] In embodiments, a compound of Formula (I) is
pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 127A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 127B, or a pharmaceutically acceptable salt thereof. [0124] In embodiments, a compound of Formula (I) is
pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 128A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 128B, or a pharmaceutically acceptable salt thereof.
[0125] In embodiments, a compound of Formula (I) is
pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 131A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 131B, or a pharmaceutically acceptable salt thereof. [0126] In embodiments, a compound of Formula (I) is

pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 135A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 135B, or a pharmaceutically acceptable salt thereof. [0127] In another aspect, the invention features a method of inhibiting avb8 integrin in a patient, the method comprising administering to the patient in need thereof a therapeutically effective amount of any compound described herein, or a pharmaceutically acceptable salt thereof. In embodiments, the method is for treating a solid tumor in a patient in need thereof. [0128] In another aspect, the invention features a method of treating solid tumor in a patient, the method comprising administering to the patient in need thereof (a) a therapeutically effective amount of any compound described herein, or a pharmaceutically acceptable salt thereof, and (b) a therapeutically effective amount of a second active agent. [0129] In embodiments, the solid tumor is selected from: anal cancer, bile duct cancer, bladder cancer, breast cancer, cervical cancer, colorectal cancer, endometrial cancer, esophageal cancer, fallopian tube cancer, gastric cancer, glioma liver cancer, lung cancer, melanoma, nasopharyngeal carcinoma, neuroblastoma, osteosarcoma, ovarian cancer, pancreatic cancer,
primary peritoneal carcinoma, prostate cancer, renal cell carcinoma, skin cancer, squamous cell carcinoma of the head and neck (SCCHN), testicular cancer, urothelial carcinoma, and uterine cancer. [0130] In embodiments, the solid tumor is selected from: breast cancer, squamous cell carcinoma of the head and neck (SCCHN), renal cell carcinoma, ovarian cancer, gastric cancer, esophageal cancer, lung cancer, pancreatic cancer, bile duct cancer, endometrial cancer, melanoma, and urothelial carcinoma. [0131] In embodiments, the second active agent is an immune checkpoint inhibitor (e.g., an anti-PD-1 or an anti-PD-L1 therapy). In embodiments, an immune checkpoint inhibitor is selected from: nivolumab, pembrolizumab, cemiplimab, dostarlimab, atezolizumab, avelumab, and durvalumab. DETAILED DESCRIPTION OF INVENTION Definitions [0132] For convenience, before further description of the present invention, certain terms employed in the specification, examples and appended claims are collected here. These definitions should be read in light of the remainder of the disclosure and understood as by a person of skill in the art. Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by a person of ordinary skill in the art. [0133] In order for the present invention to be more readily understood, certain terms and phrases are defined below and throughout the specification. [0134] The articles “a” and “an” are used herein to refer to one or to more than one (i.e., to at least one) of the grammatical object of the article. By way of example, “an element” means one element or more than one element. [0135] The phrase “and/or,” as used herein in the specification and in the claims, should be understood to mean “either or both” of the elements so conjoined, i.e., elements that are conjunctively present in some cases and disjunctively present in other cases. Multiple elements listed with “and/or” should be construed in the same fashion, i.e., “one or more” of the elements so conjoined. Other elements may optionally be present other than the elements specifically identified by the “and/or” clause, whether related or unrelated to those elements specifically identified. Thus, as a non-limiting example, a reference to “A and/or B”, when used in
conjunction with open-ended language such as “comprising” can refer, in one embodiment, to A only (optionally including elements other than B); in another embodiment, to B only (optionally including elements other than A); in yet another embodiment, to both A and B (optionally including other elements); etc. [0136] As used herein in the specification and in the claims, “or” should be understood to have the same meaning as “and/or” as defined above. For example, when separating items in a list, “or” or “and/or” shall be interpreted as being inclusive, i.e., the inclusion of at least one, but also including more than one, of a number or list of elements, and, optionally, additional unlisted items. Only terms clearly indicated to the contrary, such as “only one of” or “exactly one of,” or, when used in the claims, “consisting of,” will refer to the inclusion of exactly one element of a number or list of elements. In general, the term “or” as used herein shall only be interpreted as indicating exclusive alternatives (i.e., “one or the other but not both”) when preceded by terms of exclusivity, such as “either,” “one of,” “only one of,” or “exactly one of.” “Consisting essentially of,” when used in the claims, shall have its ordinary meaning as used in the field of patent law. [0137] As used herein in the specification and in the claims, the phrase “at least one,” in reference to a list of one or more elements, should be understood to mean at least one element selected from any one or more of the elements in the list of elements, but not necessarily including at least one of each and every element specifically listed within the list of elements and not excluding any combinations of elements in the list of elements. This definition also allows that elements may optionally be present other than the elements specifically identified within the list of elements to which the phrase “at least one” refers, whether related or unrelated to those elements specifically identified. Thus, as a non-limiting example, “at least one of A and B” (or, equivalently, “at least one of A or B,” or, equivalently “at least one of A and/or B”) can refer, in one embodiment, to at least one, optionally including more than one, A, with no B present (and optionally including elements other than B); in another embodiment, to at least one, optionally including more than one, B, with no A present (and optionally including elements other than A); in yet another embodiment, to at least one, optionally including more than one, A, and at least one, optionally including more than one, B (and optionally including other elements); etc. [0138] It should also be understood that, unless clearly indicated to the contrary, in any methods claimed herein that include more than one step or act, the order of the steps or acts of the method is not necessarily limited to the order in which the steps or acts of the method are recited.
[0139] In the claims, as well as in the specification above, all transitional phrases such as “comprising,” “including,” “carrying,” “having,” “containing,” “involving,” “holding,” “composed of,” and the like are to be understood to be open-ended, i.e., to mean including but not limited to. Only the transitional phrases “consisting of” and “consisting essentially of” shall be closed or semi-closed transitional phrases, respectively, as set forth in the United States Patent Office Manual of Patent Examining Procedures, Section 2111.03. [0140] Certain compounds contained in compositions of the present invention may exist in particular geometric or stereoisomeric forms. In addition, polymers of the present invention may also be optically active. The present invention contemplates all such compounds, including cis- and trans-isomers, R- and S-enantiomers, diastereomers, (d)-isomers, (l)-isomers, the racemic mixtures thereof, and other mixtures thereof, as falling within the scope of the invention. Additional asymmetric carbon atoms may be present in a substituent such as an alkyl group. All such isomers, as well as mixtures thereof, are intended to be included in this invention. [0141] If, for instance, a particular enantiomer of compound of the present invention is desired, it may be prepared by asymmetric synthesis, or by derivation with a chiral auxiliary, where the resulting diastereomeric mixture is separated and the auxiliary group cleaved to provide the pure desired enantiomers. Alternatively, where the molecule contains a basic functional group, such as amino, or an acidic functional group, such as carboxyl, diastereomeric salts are formed with an appropriate optically-active acid or base, followed by resolution of the diastereomers thus formed by fractional crystallization or chromatographic means well known in the art, and subsequent recovery of the pure enantiomers. [0142] Structures depicted herein are also meant to include compounds that differ only in the presence of one or more isotopically enriched atoms. For example, compounds produced by the replacement of a hydrogen with deuterium or tritium, or of a carbon with a
13C- or
14C-enriched carbon are within the scope of this invention. [0143] The terms “αvβ8”, “avB8”, “avb8”, “alpha-v beta-8” and “alpha v beta 8” and the like as used herein all refer to αvβ8. [0144] The phrase “pharmaceutically acceptable excipient” or “pharmaceutically acceptable carrier” as used herein means a pharmaceutically acceptable material, composition or vehicle, such as a liquid or solid filler, diluent, excipient, solvent or encapsulating material, involved in carrying or transporting the subject chemical from one organ or portion of the body, to another organ or portion of the body. Each carrier must be “acceptable” in the sense of being compatible
with the other ingredients of the formulation, not injurious to the patient, and substantially non- pyrogenic. Some examples of materials which can serve as pharmaceutically acceptable carriers include: (1) sugars, such as lactose, glucose, and sucrose; (2) starches, such as corn starch and potato starch; (3) cellulose, and its derivatives, such as sodium carboxymethyl cellulose, ethyl cellulose, and cellulose acetate; (4) powdered tragacanth; (5) malt; (6) gelatin; (7) talc; (8) excipients, such as cocoa butter; (9) oils, such as peanut oil, cottonseed oil, safflower oil, sesame oil, olive oil, corn oil, and soybean oil; (10) glycols, such as propylene glycol; (11) polyols, such as glycerin, sorbitol, mannitol, and polyethylene glycol; (12) esters, such as ethyl oleate and ethyl laurate; (13) agar; (14) buffering agents, such as magnesium hydroxide and aluminum hydroxide; (15) alginic acid; (16) pyrogen-free water; (17) isotonic saline; (18) Ringer’s solution; (19) ethyl alcohol; (20) phosphate buffer solutions; and (21) other non-toxic compatible substances employed in pharmaceutical formulations. In certain embodiments, pharmaceutical compositions of the present invention are non-pyrogenic, i.e., do not induce significant temperature elevations when administered to a patient. [0145] The term “pharmaceutically acceptable salts” refers to the relatively non-toxic, inorganic and organic acid addition salts of the compound(s). These salts can be prepared in situ during the final isolation and purification of the compound(s), or by separately reacting a purified compound(s) in its free base form with a suitable organic or inorganic acid, and isolating the salt thus formed. Representative salts include the hydrobromide, hydrochloride, sulfate, bisulfate, phosphate, nitrate, acetate, valerate, oleate, palmitate, stearate, laurate, benzoate, lactate, phosphate, tosylate, citrate, maleate, fumarate, succinate, tartrate, naphthylate, mesylate, glucoheptonate, lactobionate, and laurylsulphonate salts, and the like. (See, for example, Berge et al. (1977) “Pharmaceutical Salts”, J. Pharm. Sci.66:1-19.) [0146] In other cases, the compounds useful in the methods of the present invention may contain one or more acidic functional groups and, thus, are capable of forming pharmaceutically acceptable salts with pharmaceutically acceptable bases. The term “pharmaceutically acceptable salts” in these instances refers to the relatively non-toxic inorganic and organic base addition salts of a compound(s). These salts can likewise be prepared in situ during the final isolation and purification of the compound(s), or by separately reacting the purified compound(s) in its free acid form with a suitable base, such as the hydroxide, carbonate, or bicarbonate of a pharmaceutically acceptable metal cation, with ammonia, or with a pharmaceutically acceptable organic primary, secondary, or tertiary amine. Representative alkali or alkaline earth salts include the lithium, sodium, potassium, calcium, magnesium, and aluminum salts, and the like.
Representative organic amines useful for the formation of base addition salts include ethylamine, diethylamine, ethylenediamine, ethanolamine, diethanolamine, piperazine, and the like (see, for example, Berge et al., supra). [0147] A “therapeutically effective amount” (or “effective amount”) of a compound with respect to use in treatment, refers to an amount of the compound in a preparation which, when administered as part of a desired dosage regimen (to a mammal, preferably a human) alleviates a symptom, ameliorates a condition, or slows the onset of disease conditions according to clinically acceptable standards for the disorder or condition to be treated or the cosmetic purpose, e.g., at a reasonable benefit/risk ratio applicable to any medical treatment. [0148] The term “prophylactic or therapeutic” treatment is art-recognized and includes administration to the host of one or more of the subject compositions. If it is administered prior to clinical manifestation of the unwanted condition (e.g., disease or other unwanted state of the host animal) then the treatment is prophylactic, (i.e., it protects the host against developing the unwanted condition), whereas if it is administered after manifestation of the unwanted condition, the treatment is therapeutic, (i.e., it is intended to diminish, ameliorate, or stabilize the existing unwanted condition or side effects thereof). [0149] The term “patient” refers to a mammal in need of a particular treatment. In certain embodiments, a patient is a primate, canine, feline, or equine. In certain embodiments, a patient is a human. [0150] Whenever a term (e.g., alkyl or aryl) or either of their prefix roots (e.g., alk- or ar-) appear in a name of a substituent the name is to be interpreted as including those limitations provided herein. For example, affixing the suffix “-ene” to a group indicates the group is a divalent moiety, e.g., arylene is the divalent moiety of aryl, heteroarylene is the divalent moiety of heteroaryl, and heterocycloalkylene is the divalent moiety of heterocycloalkyl. Similarly, affixing the suffix “-oxy” to a group indicates the group is attached to the parent molecular structure through an oxygen atom (-O-) such as “alkyloxy,” “alkoxy” or “cycloalkoxy” as used herein. [0151] An aliphatic chain comprises the classes of alkyl, alkenyl and alkynyl defined below. A straight aliphatic chain is limited to unbranched carbon chain moieties. As used herein, the term “aliphatic group” refers to a straight chain, branched-chain, or cyclic aliphatic hydrocarbon group and includes saturated and unsaturated aliphatic groups, such as an alkyl group, an alkenyl group, or an alkynyl group.
[0152] “Alkyl” refers to a fully saturated cyclic or acyclic, branched or unbranched carbon chain moiety having the number of carbon atoms specified, or 1 up to 30 carbon atoms if no specification is made. For example, alkyl of 1 to 8 carbon atoms refers to moieties such as methyl, ethyl, propyl, butyl, pentyl, hexyl, heptyl, and octyl, and those moieties which are positional isomers of these moieties. Alkyl of 10 to 30 carbon atoms includes decyl, undecyl, dodecyl, tridecyl, tetradecyl, pentadecyl, hexadecyl, heptadecyl, octadecyl, nonadecyl, eicosyl, heneicosyl, docosyl, tricosyl and tetracosyl. In certain embodiments, a straight chain or branched chain alkyl has 30 or fewer carbon atoms in its backbone (e.g., C
1-C
30 for straight chains, C
3-C
30 for branched chains), and more preferably 20 or fewer. Alkyl goups may be substituted or unsubstituted. As used herein, “Me” and –CH3 both refer to methyl. [0153] As used herein, the term “alkylene” refers to an alkyl group having the specified number of carbons, for example from 2 to 12 carbon atoms, that contains two points of attachment to the rest of the compound on its longest carbon chain. Non-limiting examples of alkylene groups include methylene -(CH
2)-, ethylene -(CH
2CH
2)-, n-propylene -(CH
2CH
2CH
2)-, isopropylene -(CH2CH(CH3))-, and the like. Alkylene groups can be cyclic or acyclic, branched or unbranched carbon chain moiety, and may be optionally substituted with one or more substituents. [0154] "Cycloalkyl" means mono- or bicyclic or bridged or spirocyclic, or polycyclic saturated carbocyclic rings, each having from 3 to 12 carbon atoms. Likewise, preferred cycloalkyls have from 3-10 carbon atoms in their ring structure, and more preferably have 3-6 carbons in the ring structure. Cycloalkyl groups may be substituted or unsubstituted. Exemplary cycloalkyl groups include cyclopropyl (C3), cyclobutyl (C4), cyclopentyl (C5), cyclopentenyl (C5), cyclohexyl (C6), cycloheptyl(C7), and cyclooctyl (C8). [0155] Unless the number of carbons is otherwise specified, “lower alkyl,” as used herein, means an alkyl group, as defined above, but having from one to ten carbons, more preferably from one to six carbon atoms in its backbone structure such as methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, and tert-butyl. Likewise, “lower alkenyl” and “lower alkynyl” have similar chain lengths. Throughout the application, preferred alkyl groups are lower alkyls. In certain embodiments, a substituent designated herein as alkyl is a lower alkyl. [0156] The term “aryl” as used herein includes 3- to 12-membered substituted or unsubstituted single-ring aromatic groups in which each atom of the ring is carbon (i.e., carbocyclic aryl) or where one or more atoms are heteroatoms (i.e., heteroaryl). Preferably, aryl
groups include 5- to 12-membered rings, more preferably 6- to 10-membered rings The term “aryl” also includes polycyclic ring systems having two or more cyclic rings in which two or more carbons are common to two adjoining rings wherein at least one of the rings is aromatic, e.g., the other cyclic rings can be cycloalkyls, cycloalkenyls, cycloalkynyls, aryls, heteroaryls, and/or heterocyclyls. Carbocyclic aryl groups include benzene, naphthalene, phenanthrene, phenol, aniline, and the like. Heteroaryl groups include substituted or unsubstituted aromatic 3- to 12-membered ring structures, more preferably 5- to 12-membered rings, more preferably 5- to 10- membered rings, whose ring structures include one to four heteroatoms. Heteroaryl groups include, for example, pyrrole, furan, thiophene, imidazole, oxazole, thiazole, triazole, pyrazole, pyridine, pyrazine, pyridazine and pyrimidine, and the like. Aryl and heteroaryl can be monocyclic, bicyclic, or polycyclic. [0157] The term “halo”, “halide”, or “halogen” as used herein means halogen and includes, for example, and without being limited thereto, fluoro, chloro, bromo, iodo and the like, in both radioactive and non-radioactive forms. In a preferred embodiment, halo is selected from the group consisting of fluoro, chloro and bromo. [0158] The terms “heterocyclyl” or “heterocyclic group” refer to 3- to 12-membered ring structures, more preferably 5- to 12-membered rings, more preferably 5- to 10-membered rings, whose ring structures include one to four heteroatoms. Heterocycles can be monocyclic, bicyclic, spirocyclic, or polycyclic. Heterocyclyl groups include, for example, thiophene, thianthrene, furan, pyran, isobenzofuran, chromene, xanthene, phenoxathiin, pyrrole, imidazole, pyrazole, isothiazole, isoxazole, pyridine, pyrazine, pyrimidine, pyridazine, indolizine, isoindole, indole, indazole, purine, quinolizine, isoquinoline, quinoline, phthalazine, naphthyridine, quinoxaline, quinazoline, cinnoline, pteridine, carbazole, carboline, phenanthridine, acridine, pyrimidine, phenanthroline, phenazine, phenarsazine, phenothiazine, furazan, phenoxazine, pyrrolidine, oxolane, thiolane, oxazole, piperidine, piperazine, morpholine, lactones, lactams such as azetidinones and pyrrolidinones, sultams, sultones, and the like. The heterocyclic ring can be substituted at one or more positions with such substituents as described above, as for example, halogen, alkyl, aralkyl, alkenyl, alkynyl, cycloalkyl, hydroxyl, amino, nitro, sulfhydryl, imino, amido, phosphate, phosphonate, phosphinate, carbonyl, carboxyl, silyl, sulfamoyl, sulfinyl, ether, alkylthio, sulfonyl, ketone, aldehyde, ester, a heterocyclyl, an aromatic or heteroaromatic moiety, -CF
3, -CN, and the like. [0159] The term “heterocycloalkyl” as used herein, is a non-aromatic heterocyclyl wherein at least one atom is a heteroatom such as, but not limited to, nitrogen, oxygen, sulfur, or
phosphorus, and the remaining atoms are carbon. Examples of heterocycloalkyl groups are pyrrolidinyl, tetrahydrofuranyl, dihydrofuranyl, tetrahydrothienyl, tetrahydropyranyl, dihydropyranyl, tetrahydrothiopyranyl, piperidino, morpholino, thiomorpholino, thioxanyl, piperazinyl, azetidinyl, oxetanyl, thietanyl, homopiperidinyl, oxepanyl, thiepanyl, oxazepinyl, diazepinyl, thiazepinyl, 1,2,3,6-tetrahydropyridinyl, 2-pyrrolinyl, 3-pyrrolinyl, indolinyl, 2H- pyranyl, 4H-pyranyl, dioxanyl, 1,3-dioxolanyl, pyrazolinyl, dithianyl, dithiolanyl, dihydropyranyl, dihydrothienyl, dihydrofuranyl, pyrazolidinyl, imidazolinyl, imidazolidinyl, 3- azabicyclo[3.1.0]hexanyl, 3-azabicyclo[4.1.0]heptanyl, 3H-indolyl and quinolizinyl. The heterocycloalkyl group can be substituted or unsubstituted as recited, e.g., for heterocyclyls as described herein. [0160] The term “carbonyl” is art-recognized and includes such moieties as can be represented by the formula:

[0161] wherein X’ is a bond or represents an oxygen or a sulfur, and R
15 represents a hydrogen, an alkyl, an alkenyl, -(CH
2)
m-R
10 or a pharmaceutically acceptable salt, R
16 represents a hydrogen, an alkyl, an alkenyl or -(CH
2)
m-R
10, where m and R
10 are as defined above. Where X’ is an oxygen and R15 or R16 is not hydrogen, the formula represents an “ester.” Where X’ is an oxygen, and R15 is as defined above, the moiety is referred to herein as a carboxyl group, and particularly when R15 is a hydrogen, the formula represents a “carboxylic acid”. Where X’ is an oxygen, and R
16 is a hydrogen, the formula represents a “formate.” On the other hand, where X’ is a bond, and R
15 is not hydrogen, the above formula represents a “ketone” group. Where X’ is a bond, and R
15 is a hydrogen, the above formula represents an “aldehyde” group. [0162] As used herein, the term “substituted” is contemplated to include all permissible substituents of organic compounds. In a broad aspect, the permissible substituents include acyclic and cyclic, branched and unbranched, carbocyclic and heterocyclic, aromatic and nonaromatic substituents of organic compounds. Illustrative substituents include, for example, those described herein above, and for example substituted with one or more substituents selected from alkyl, cycloalkyl, heterocyclylakyl, halogen, OH, OMe, C(H)F
2, C(F)H
2, CF
3, C(H)
2CF
3, SF
5, CHFCH2amine, CH2amine, and CN. The permissible substituents can be one or more and the same or different for appropriate organic compounds. For purposes of this invention, the heteroatoms such as nitrogen may have hydrogen substituents and/or any permissible substituents
of organic compounds described herein which satisfy the valences of the heteroatoms. This invention is not intended to be limited in any manner by the permissible substituents of organic compounds. It will be understood that “substitution” or “substituted with” includes the implicit proviso that such substitution is in accordance with permitted valence of the substituted atom and the substituent, and that the substitution results in a stable compound, e.g., which does not spontaneously undergo transformation such as by rearrangement, cyclization, elimination, etc. [0163] As used herein, the term “nitro” means -NO
2; the term “halogen” designates - F, -Cl, -Br, or -I; the term “hydroxyl” means -OH; and the term “cyano” means –CN;. [0164] As used herein, the definition of each expression, e.g., alkyl, m, n, etc., when it occurs more than once in any structure, is intended to be independent of its definition elsewhere in the same structure. [0165] The term “prodrug” as used herein encompasses compounds that, under physiological conditions, are converted into therapeutically active agents. A common method for making a prodrug is to include selected moieties that are hydrolyzed under physiological conditions to reveal the desired molecule. In other embodiments, the prodrug is converted by an enzymatic activity of the host animal. Accordingly, prodrugs include compounds that are transformed in vivo to yield a disclosed compound or any other pharmaceutically acceptable form of the compound. In embodiments, a prodrug may be inactive when administered to a subject but may be converted in vivo to an active compound, for example, by hydrolysis. See, e.g., Bundgard, H., Design of Prodrugs (1985), pp.7-9, 21-24 (Elsevier, Amsterdam). A discussion of prodrugs is provided in Higuchi, T., et al., “Pro-drugs as Novel Delivery Systems,” A.C.S. Symposium Series, Vol.14, and in Bioreversible Carriers in Drug Design, ed. Edward B. Roche, American Pharmaceutical Association and Pergamon Press, 1987, both of which are incorporated in full by reference herein. Prodrugs can typically be prepared using well known methods, such as those described in Burger's Medicinal Chemistry and Drug Discovery, 172-178, 949-982 (Manfred E. Wolff ed., 5th ed., 1995), and Design of Prodrugs (H. Bundgaard ed., Elselvier, New York, 1985). The term “prodrug” is also meant to include any covalently bonded carriers, which release the active compound in vivo when such prodrug is administered to a subject. [0166] Prodrugs of compounds described herein may be prepared by modifying functional groups present in the active compound in such a way that the modifications are cleaved, either in routine manipulation or in vivo, to provide a compound described herein (i.e., the parent active compound). Prodrugs include compounds wherein a hydroxy, amino or mercapto group is bonded
to any group that, when the prodrug of the active compound is administered to a subject, cleaves to form a free hydroxy, free amino or free mercapto group, respectively. Examples of prodrugs include, but are not limited to, acetate, formate and benzoate derivatives of an alcohol or acetamide, formamide and benzamide derivatives of an amine functional group in the active compound and the like. Other examples of prodrugs include compounds that comprise —NO, — NO
2, —ONO, or —ONO
2 moieties. [0167] For purposes of this invention, the chemical elements are identified in accordance with the Periodic Table of the Elements, CAS version, Handbook of Chemistry and Physics, 67th Ed., 1986-87, inside cover. Exemplary Compounds of the Invention [0168] This disclosure relates to novel chemical compounds and methods useful for inhibiting α
vβ
8 integrin. [0169] Exemplary formulas and compounds are described herein. Also provided herein are exemplary embodiments of structural features which may be present in any formula described herein. Any exemplary embodiment of a structural feature may occur in combination with any other exemplary structural feature described herein. Further, and unless otherwise indicated herein, any description of a formula or compound also includes any pharmaceutically acceptable forms of the compound, including but not limited to any pharmaceutically acceptable salts, hydrates, solvates, isomers, polymorphs, prodrugs, and isotopically labeled derivatives of disclosed formulas and compounds. [0170] In embodiments, a compound described herein is a selective inhibitor of αvβ8 integrin. In embodiments, a compound described herein selectively inhibits αvβ8 integrin over, e.g., αvβ6 integrin (e.g., a selectivity of at least about 10×, 50×, 100×, or 1000× as measured according to an assay (e.g., fluorescence polarization assay)). [0171] Certain exemplary formulas, compounds, and structural features are described herein. Any structural features and embodiments described herein can be used in any combination with any other structural feature(s) and embodiments(s) described herein.
Formula (I) [0172] In one aspect, the invention features a compound of Formula (I), or a pharmaceutically acceptable salt thereof:

wherein:
h is optionally substituted with 1 to 6 R
4; each of R
1a, R
1b, R
1c, R
1d, R
1e, and R
1f is independently H, C
1-4alkyl, halogen, C
1- 4alkyloxy, OH, C
1-4alkyl-OH, C
1-4alkyl-C
1-4alkyloxy, C
1-4alkyloxy-C
1-4alkyloxy, CF
3, CHF
2, CH2F, CN, NO2, NRaRb or C1-4alkyl-NRaRb, R
2 is H, C1-4alkyl or C3-5cycloalkyl, each of C1-4alkyl and C3-5cycloalkyl is optionally substituted with 1 to 4 R
5; R
3a is C
1-4alkyloxy, C
3-5cycloalkoxy, CF
3, CHF
2, CH
2F, OCF
3, OCHF
2 or OCH
2F; R
3b is H, halogen, CF
3 or CN; R
3c is H, F, CN, or C
1-4alkyl; R
3d is H, C1-4alkyl, C3-5cycloalkyl, or 4-6-membered heterocycloalkyl, each of which is optionally substituted with 1 to 4 R
6; R
3e is H or F; each R
4 is independently H, C1-4alkyl, halogen, CF3, CHF2 or CH2F, cyclopropyl, or two geminal R
4 groups together can form a spiro-cyclopropyl;
each R
5 is independently H, C
1-4alkyl, C
3-5cycloalkyl, C
1-4alkyloxy, C
3-5cycloalkoxy, F, CF
3, CHF
2, CH
2F, OCF
3, OCHF
2 or OCH
2F; each R
6 is independently H, C1-4alkyl, C2-4alkenyl, C3-5cycloalkyl, C1-4alkyloxy, C3- 5cycloalkoxy, F, CF3, CHF2, CH2F, OCF3, OCHF2, OCH2F, OH, 5-6-membered heteroaryl, or NRaRb; each R
7 is independently C
1-4alkyl or F; each of R
a and R
b is independently hydrogen, C
1-4alkyl, C
3-5cycloalkyl, or R
a and R
b, together with the nitrogen atom to which they are attached, form a saturated or unsaturated heterocyclic ring containing from three to seven ring atoms, which ring may optionally contain additional one or two heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur and may be optionally substituted by from one to three groups which may be the same or different selected from the group consisting of F, C
1-
4alkyl, phenyl and benzyl; and n is 0, 1 or 2; and m is 0, 1 or 2. [0173] In one aspect, the invention features a compound of Formula (I), or a pharmaceutically acceptable salt thereof:
wherein:
to 6 R
4;
each of R
1a, R
1b, R
1c, R
1d, R
1e, and R
1f is independently H, C
1-4alkyl, halogen, C
1- 4alkyloxy, OH, C
1-4alkyl-OH, C
1-4alkyl-C
1-4alkyloxy, C
1-4alkyloxy-C
1-4alkyloxy, CF
3, CHF
2, CH2F, CN, NO2, NRaRb or C1-4alkyl-NRaRb, R
2 is H, C1-4alkyl or C3-5cycloalkyl, each of C1-4alkyl and C3-5cycloalkyl is optionally substituted with 1 to 4 R
5; R
3a is cyano, halogen, C
1-4alkyl, C
1-4alkyloxy, C
3-5cycloalkoxy, CF
3, CHF
2, CH
2F, OCF
3, OCHF
2 or OCH
2F; R
3b is H, halogen, CF3 or CN; R
3c is H, F, CN, or C1-4alkyl; R
3d is H, C1-4alkyl, C3-6cycloalkyl, or 4-6-membered heterocycloalkyl, each of which is optionally substituted with 1 to 4 R
6; R
3e is H or F; each R
4 is independently H, C
1-4alkyl, halogen, CF
3, CHF
2 or CH
2F, cyclopropyl, or two geminal R
4 groups together can form a spiro-cyclopropyl; each R
5 is independently H, C1-4alkyl, C3-5cycloalkyl, C1-4alkyloxy, C3-5cycloalkoxy, F, CF3, CHF2, CH2F, OCF3, OCHF2 or OCH2F; each R
6 is independently H, C1-4alkyl, C2-4alkenyl, C3-5cycloalkyl, C1-4alkyloxy, C3-
5cycloalkoxy, F, CF
3, CHF
2, CH
2F, OCF
3, OCHF
2, OCH
2F, OH, 5-6-membered heteroaryl, or NR
aR
b; each R
7 is independently C
1-4alkyl or F; each of Ra and Rb is independently hydrogen, C1-4alkyl, C3-5cycloalkyl, or Ra and Rb, together with the nitrogen atom to which they are attached, form a saturated or unsaturated heterocyclic ring containing from three to seven ring atoms, which ring may optionally contain additional one or two heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur and may be optionally substituted by from one to three groups which may be the same or different selected from the group consisting of F, C
1-
4alkyl, phenyl and benzyl; and n is 0, 1 or 2; and m is 0, 1 or 2.
[0174] In one aspect, the invention features a compound of Formula (I), or a pharmaceutically acceptable salt thereof:
wherein:
h is optionally substituted with 1 to 6 R
4; each of R
1a, R
1b, R
1c, R
1d, R
1e, and R
1f is independently H, C
1-4alkyl, halogen, C
1- 4alkyloxy, OH, C
1-4alkyl-OH, C
1-4alkyl-C
1-4alkyloxy, C
1-4alkyloxy-C
1-4alkyloxy, CF
3, CHF
2, CH
2F, CN, NO
2, NR
aR
b or C
1-4alkyl-NR
aR
b, R
2 is H, C1-4alkyl or C3-5cycloalkyl, each of C1-4alkyl and C3-5cycloalkyl is optionally substituted with 1 to 4 R
5; R
3a is cyano, halogen, or C1-4alkyl; R
3b is H, halogen, CF
3 or CN; R
3c is H, F, CN, or C
1-4alkyl; R
3d is H, C
1-4alkyl, C
3-6cycloalkyl, or 4-6-membered heterocycloalkyl, each of which is optionally substituted with 1 to 4 R
6; R
3e is H or F; each R
4 is independently H, C1-4alkyl, halogen, CF3, CHF2 or CH2F, cyclopropyl, or two geminal R
4 groups together can form a spiro-cyclopropyl;
each R
5 is independently H, C
1-4alkyl, C
3-5cycloalkyl, C
1-4alkyloxy, C
3-5cycloalkoxy, F, CF
3, CHF
2, CH
2F, OCF
3, OCHF
2 or OCH
2F; each R
6 is independently H, C1-4alkyl, C2-4alkenyl, C3-5cycloalkyl, C1-4alkyloxy, C3- 5cycloalkoxy, F, CF3, CHF2, CH2F, OCF3, OCHF2, OCH2F, OH, 5-6-membered heteroaryl, or NRaRb; each R
7 is independently C
1-4alkyl or F; each of R
a and R
b is independently hydrogen, C
1-4alkyl, C
3-5cycloalkyl, or R
a and R
b, together with the nitrogen atom to which they are attached, form a saturated or unsaturated heterocyclic ring containing from three to seven ring atoms, which ring may optionally contain additional one or two heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur and may be optionally substituted by from one to three groups which may be the same or different selected from the group consisting of F, C
1-
4alkyl, phenyl and benzyl; and n is 0, 1 or 2; and m is 0, 1 or 2. [0175] In embodiments, the Q ring i
, wherein R
1c1 and R
1c2 are each independently selected from R
1c; R
1d1 and R
1d2 are each independently selected from R
1d; and R1e1 and R1e2 are each independently selected from R1e. [0176] In embodiments, each of R1c1, R1c2, R1d1, R1d2, R1e1 and R1e2 are each independently H. [0177] In embodiments, the Q ring i
. [0178] In embodiments, each of R
1c, R
1d, and R
1e is independently H. [0179] In embodiments, each R
1a is independently H. [0180] In embodiments, each R
1b is independently H. [0181] In embodiments, each R
1b is independently OMe.
[0182] In embodiments, the Q ring i
. [0183] In embodiments, the Q ring i
[0184]
are each independently selected from R
1c; and R
1d1 and R
1d2 are each independently selected from R
1d. [0185] In embodiments, each of R1c1, R1c2, R1d1, and R1d2 are each independently H. [0186] In embodiments, the Q ring i
. [0187] In embodiments, the Q ring i
. [0188] In embodiments, each of R
1c and R
1d is independently H. [0189] In embodiments, each of R
1a, R
1b and R
1f is independently H. [0190] In embodiments,
. [0191] In embodiments,
. [0192] In embodiments,
. [0193] In embodiments,
.
[0194] In embodiments, L is , wherein R4a, R4b, R4c and R4d are each independently an R
4.
[0195] In embodiments,
wherein R4a is C1-4alkyl, and R4b, R4c and R
4d are each independently H. [0196] In embodiments, L is
, wherein R
4a is methyl, and R
4b, R
4c and R
4d are each independently H.
[0197] In embodiments, L is , wherein R4a and R4b are each independently C
1-4alkyl, and R
4c and R
4d are each independently H. [0198] In embodiments, L is
, wherein R
4a and R
4b are each independently methyl, and R
4c and R
4d are each independently H. [0199] In embodiments, L i
, wherein R
4c is halogen, and R
4a, R
4b and R
4d are each independently H. [0200] In embodiments, L i
, wherein R
4c is F, and R
4a, R
4b and R
4d are each independently H. [0201] In embodiments, L i
, wherein R
4a and R
4b are each independently H, and R
4c and R
4d are each independently halogen. [0202] In embodiments, L is
, wherein R
4a and R
4b are each independently H, and R
4c and R
4d are each independently F. [0203] In embodiments,
[0204] In embodiments,
. [0205] In embodiments,
. [0206] In embodiments,
. [0207] In embodiments,
. [0208] In embodiments,
. [0209] In embodiments, R
2 is C
1-4alkyl. [0210] In embodiments, R
2 is methyl. [0211] In embodiments, R
2 is ethyl. [0212] In embodiments, R
2 is cyclopropyl. [0213] In embodiments, R
3a is C1-4alkyloxy. [0214] In embodiments, R
3a is OMe, OEt, OCF3, OCHF2 or OCH2F. [0215] In embodiments, R
3a is OMe. [0216] In embodiments, R
3a is -CN. [0217] In embodiments, R
3a is halogen. [0218] In embodiments, R
3a is Cl. [0219] In embodiments, R
3a is C1-4alkyl. [0220] In embodiments, R
3a is methyl. [0221] In embodiments, R
3a is ethyl. [0222] In embodiments, R
3b is F. [0223] In embodiments, R
3c is H. [0224] In embodiments, R
3d is C
1-4alkyl. [0225] In embodiments, R
3d is C3-5cycloalkyl.
[0226] In embodiments, R
3d is oxetanyl, tetrahydrofuranyl, tetrahydro-2H-pyranyl, morpholinyl or piperazinyl-C
1-4alkyl. [0227] In embodiments, R
3d is iso-propyl. [0228] In embodiments, R
3e is H. [0229] In embodiments, each R
4 is independently methyl. [0230] In embodiments, each R
4 is independently F. [0231] In embodiments, each R
4 is independently CF
3, CHF
2 or CH
2F. [0232] In embodiments, each R
4 is independently H. [0233] In embodiments, each R
5 is independently C1-4alkyl. [0234] In embodiments, each R
5 is independently C3-5cycloalkyl. [0235] In embodiments, each R
5 is independently C1-4alkyloxy. [0236] In embodiments, each R
5 is independently C
3-5cycloalkoxy. [0237] In embodiments, each R
5 is independently F, CF
3, CHF
2, CH
2F, OCF
3, OCHF
2 or OCH
2F. [0238] In embodiments, each R
5 is independently H. [0239] In embodiments, each R
6 is independently C1-4alkyl, C2-4alkenyl, C3-5cycloalkyl, C1- 4alkyloxy, C3-5cycloalkoxy, F, CF3, CHF2, CH2F, OCF3, OCHF2, OCH2F, OH, 5-6-membered heteroaryl, or NR
aR
b. [0240] In embodiments, each R
6 is independently H. [0241] In embodiments, each R
6 is independently C
1-4alkyl. [0242] In embodiments, each R
6 is independently C2-4alkenyl. [0243] In embodiments, each R
6 is independently C3-5cycloalkyl. [0244] In embodiments, each R
6 is independently C1-4alkyloxy. [0245] In embodiments, each R
6 is independently C
3-5cycloalkoxy. [0246] In embodiments, each R
6 is independently F, CF
3, CHF
2, CH
2F, OCF
3, OCHF
2, OCH
2F or OH. [0247] In embodiments, each R
6 is independently a 5-6-membered heteroaryl.
[0248] In embodiments, each R
6 is independently H. [0249] In embodiments, each R
7 is independently methyl or F. [0250] In embodiments, n is 0. [0251] In embodiments, n is 1. [0252] In embodiments, n is 2. [0253] In embodiments, m is 0. [0254] In embodiments, m is 1. [0255] In embodiments, m is 2. [0256] In embodiments, R
2 is independently C1-4alkyl (e.g., methyl, ethyl, or cyclopropyl); R
3a is independently C1-4alkyloxy (e.g., OMe or OEt), OCF3, OCHF2, or OCH2F; R
3b is independently F; R
3c is independently H; R
3d is independently C
1-4alkyl (e.g., isopropyl), C
3-5cycloalkyl, oxetanyl, tetrahydrofuranyl, tetrahydro-2H-pyranyl, morpholinyl or piperazinyl-C1-4alkyl; R
3e is independently H; each R
4 is independently H, CH3, F, CF3, CHF2, or CH2F; each R
5 is independently, H, C
1-4alkyl, C
3-5cycloalkyl, C
1-4alkyloxy, F, CF
3, CHF
2, CH
2F, OCF
3, OCHF
2 or OCH
2F; each R
7 is independently CH
3 or F; n is 0, 1, or 2; and/or m is 0. [0257] In embodiments, R
3a is OMe, R
3b is F, and/or R
3d is iso-propyl. [0258] In embodiments, R
3a is OMe and R
3b is F. [0259] In embodiments, R
3a is OMe and R
3d is iso-propyl. [0260] In embodiments, R
3b is F and R
3d is iso-propyl. [0261] In embodiments, R
3a is OMe, R
3b is F, and R
3d is iso-propyl.
[0262] In embodiments ; then each of R
1c 1d 1e
, R , and R is independently H; each R
1a is independently H; and/or each R
1b is independently H or OMe. [0263] In embodiments ; then
1c 1d
each of R and R is independently H; and/or each of R
1a, R
1b and R
1f is independently H. Formulas (II)-(VI) [0264] In embodiments, a compound has a structure according to Formula (II),
pharmaceutically acceptable salt thereof, wherein L, R
1b, R
2, R
3a, R
3b, R
3c, R
3d, and R
3e are according to any embodiments described herein. [0265] In embodiments, a compound has a structure according to Formula (IIA),
pharmaceutically acceptable salt thereof, wherein L, R
1b, R
2, R
3c, and R
3d are according to any embodiments described herein.
[0266] In embodiments, a compound has a structure according to Formula (IIB),
pharmaceutically acceptable salt thereof, wherein L, R
1b, R
2, R
3b, and R
3c are according to any embodiments described herein. [0267] In embodiments, a compound has a structure according to Formula (III),
pharmaceutically acceptable salt thereof, wherein L, R
1b, R
2, R
3a, R
3b, R
3c, R
3d, and R
3e are according to any embodiments described herein. [0268] In embodiments, a compound has a structure according to Formula (IIIA),
pharmaceutically acceptable salt thereof, wherein L, R
1b, R
2, R
3c, and R
3d are according to any embodiments described herein. [0269] In embodiments, a compound has a structure according to Formula (IIIB),
pharmaceutically acceptable salt thereof, wherein L, R
1b, R
2, R
3b, and R
3c are according to any embodiments described herein.
[0270] In embodiments, a compound has a structure according to Formula (IV),
pharmaceutically acceptable salt thereof, wherein L, R
2, R
3a, R
3b, R
3c, R
3d, and R
3e are according to any embodiments described herein. [0271] In embodiments, a compound has a structure according to Formula (IVA),
pharmaceutically acceptable salt thereof, wherein L, R
2, R
3c, and R
3d are according to any embodiments described herein. [0272] In embodiments, a compound has a structure according to Formula (IVB),
thereof, wherein L, R
2, R
3b, and R
3c are according to any embodiments described herein. [0273] In embodiments, a compound has a structure according to Formula (V),
pharmaceutically acceptable salt thereof, wherein L, R
2, R
3a, R
3b, R
3c, R
3d, and R
3e are according to any embodiments described herein.
[0274] In embodiments, a compound has a structure according to Formula (VA),
pharmaceutically acceptable salt thereof, wherein L, R
2, R
3c, and R
3d are according to any embodiments described herein. [0275] In embodiments, a compound has a structure according to Formula (VB),
thereof, wherein L, R
2, R
3b, and R
3c are according to any embodiments described herein. [0276] In embodiments, a compound has a structure according to Formula (VI),
pharmaceutically acceptable salt thereof, wherein R
2, R
3a, R
3b and R
3d are according to any embodiments described herein. [0277] In embodiments, a compound has a structure according to Formula (VI),
pharmaceutically acceptable salt thereof, wherein R
2 is methyl, R
3a is methoxy and R
3b and R
3d are according to any embodiments described herein.
[0278] In embodiments, a compound has a structure according to Formula (VI),
pharmaceutically acceptable salt thereof, wherein R
2 is methyl, R
3a is methoxy, R
3b is F and R
3d is according to any embodiments described herein. [0279] In embodiments, the carbon marked by the asterisk has the (R)-configuration. [0280] In embodiments, the carbon marked by the asterisk has the (S)-configuration. [0281] In embodiments, wherein R
2 is C1-4alkyl optionally substituted with 1 or 2 R
5. [0282] In embodiments, R
2 is –CH
3, -CH
2CH
3, -CH(CH
3)
2, -C(CH
3)
3, -CH
2CH(CH
3)
2, -CH
2CF
3, -(CH
2)
2CF
3, -CHF
2, -CH
2CHF
2, -(CH
2)
2CHF
2, -(CH
2)
2OCH
3, -(CH
2)
2O-(cyclopropyl), -(CH
2)
2OCF
3, -(CH
2)
2C(OCH
3)(CH
3)CH
3, or cyclopropyl. [0283] In embodiments, R
2 is –CH3. [0284] In embodiments, R
3c is H. [0285] In embodiments, R
3c is F. [0286] In embodiments, R
3b is F. [0287] In embodiments, R
3d is isopropyl. [0288] In embodiments, L is unsubstituted
. [0289] In embodiments, L is
substituted by 1 or 2 R
5. [0290] In embodiments, each R
5 is independently selected from -CH3, -CH2F, and –F. [0291] In embodiments, L is unsubstituted
. [0292] In embodiments, the compound is selected from any compound described in Table 1, or a pharmaceutically acceptable salt thereof.
Additional Exemplary Embodiments [0293] Still further exemplary embodiments of variables in the exemplary Formulas (I)-(VI) which may be present in any combination as valency permits are described herein. [0294] In embodiments, L is
optionally substituted with 1 to 6 R
4. In embodiments, L is unsubstituted. In embodiments, L is substituted with 1 to 6 R
4. In embodiments, L is substituted with 1, 2, or 3 R
4. In embodiments, L is substituted with 1 R
4. In embodiments, L is substituted with 2 R
4. In embodiments, L is substituted with 3 R
4. In embodiments, L is substituted with 4 R
4. In embodiments, L is substituted with 5 R
4. In embodiments, L is substituted with 6 R
4. [0295] In embodiments, L is
optionally substituted with 1 to 6 R
4. In embodiments, L is unsubstituted. In embodiments, L is substituted with 1 to 6 R
4. In embodiments, L is substituted with 1, 2, or 3 R
4. In embodiments, L is substituted with 1 R
4. In embodiments, L is substituted with 2 R
4. In embodiments, L is substituted with 3 R
4. In embodiments, L is substituted with 4 R
4. In embodiments, L is substituted with 5 R
4. In embodiments, L is substituted with 6 R
4. [0296] In embodiments, R
2 is H. [0297] In embodiments, R
2 is C1-4alkyl optionally substituted with 1 to 4 R
5. In embodiments, R
2 is unsubstituted C
1-4alkyl. In embodiments, R
2 is C
1-4alkyl substituted with 1 to 4 R
5. In embodiments, R
2 is C
1-4alkyl substituted with 1 R
5. In embodiments, R
2 is C
1-4alkyl substituted with 2 R
5. In embodiments, R
2 is C
1-4alkyl substituted with 3 R
5. In embodiments, R
2 is C1-4alkyl substituted with 4 R
5. [0298] In embodiments, R
2 is C3-5cycloalkyl optionally substituted with 1 to 4 R
5. In embodiments, R
2 is unsubstituted C3-5cycloalkyl. In embodiments, R
2 is C3-5cycloalkyl substituted with 1 to 4 R
5. In embodiments, R
2 is C
3-5cycloalkyl substituted with 1 R
5. In embodiments, R
2 is C
3-5cycloalkyl substituted with 2 R
5. In embodiments, R
2 is C
3-5cycloalkyl substituted with 3 R
5. In embodiments, R
2 is C
3-5cycloalkyl substituted with 4 R
5. [0299] In embodiments, R
2 is C1-4alkyl (e.g., methyl, ethyl, or cyclopropyl). [0300] In embodiments, R
3a is -CN. [0301] In embodiments, R
3a is halogen. [0302] In embodiments, R
3a is Cl.
[0303] In embodiments, R
3a is C
1-4alkyl. [0304] In embodiments, R
3a is methyl. [0305] In embodiments, R
3a is ethyl. [0306] In embodiments, R
3a is C1-4alkyloxy. [0307] In embodiments, R
3a is C3-5cycloalkoxy. [0308] In embodiments, R
3a is CF
3. [0309] In embodiments, R
3a is CHF
2. [0310] In embodiments, R
3a is CH
2F. [0311] In embodiments, R
3a is OCF3. [0312] In embodiments, R
3a is OCHF2. [0313] In embodiments, R
3a is OCH2F. [0314] In embodiments, R
3a is C
1-4alkyloxy (e.g., OMe or OEt), OCF
3, OCHF
2, or OCH
2F. In embodiments, R
3a is OMe. [0315] In embodiments, R
3b is H. [0316] In embodiments, R
3b is halogen (e.g., F). [0317] In embodiments, R
3b is CF3. [0318] In embodiments, R
3b is CN. [0319] In embodiments, R
3c is H. [0320] In embodiments, R
3c is F. [0321] In embodiments, R
3c is CN. [0322] In embodiments, R
3c is C1-4alkyl. [0323] In embodiments, R
3d is H. [0324] In embodiments, R
3d is C1-4alkyl optionally substituted with 1 to 4 R
6. In embodiments, R
3d is unsubstituted C
1-4alkyl. In embodiments, R
3d is C
1-4alkyl substituted with 1 to 4 R
6. In embodiments, R
3d is C
1-4alkyl substituted with 1 R
6. In embodiments, R
3d is C
1-4alkyl substituted with 2 R
6. In embodiments, R
3d is C
1-4alkyl substituted with 3 R
6. In embodiments, R
3d is C1-4alkyl substituted with 4 R
6.
[0325] In embodiments, R
3d is C
3-5cycloalkyl optionally substituted with 1 to 4 R
6. In embodiments, R
3d is unsubstituted C
3-5cycloalkyl. In embodiments, R
3d is C
3-5cycloalkyl substituted with 1 to 4 R
6. In embodiments, R
3d is C3-5cycloalkyl substituted with 1 R
6. In embodiments, R
3d is C3-5cycloalkyl substituted with 2 R
6. In embodiments, R
3d is C3-5cycloalkyl substituted with 3 R
6. In embodiments, R
3d is C3-5cycloalkyl substituted with 4 R
6. [0326] In embodiments, R
3d is C
3-6cycloalkyl optionally substituted with 1 to 4 R
6. In embodiments, R
3d is unsubstituted C
3-6cycloalkyl. In embodiments, R
3d is C
3-6cycloalkyl substituted with 1 to 4 R
6. In embodiments, R
3d is C
3-6cycloalkyl substituted with 1 R
6. In embodiments, R
3d is C3-6cycloalkyl substituted with 2 R
6. In embodiments, R
3d is C3-6cycloalkyl substituted with 3 R
6. In embodiments, R
3d is C3-6cycloalkyl substituted with 4 R
6. [0327] In embodiments, R
3d is 4-6-membered heterocycloalkyl optionally substituted with 1 to 4 R
6. In embodiments, R
3d is unsubstituted 4-6-membered heterocycloalkyl. In embodiments, R
3d is 4-6-membered heterocycloalkyl substituted with 1 to 4 R
6. In embodiments, R
3d is 4-6- membered heterocycloalkyl substituted with 1 R
6. In embodiments, R
3d is 4-6-membered heterocycloalkyl substituted with 2 R
6. In embodiments, R
3d is 4-6-membered heterocycloalkyl substituted with 3 R
6. In embodiments, R
3d is 4-6-membered heterocycloalkyl substituted with 4 R
6. [0328] In embodiments, R
3d is C
1-4alkyl (e.g., isopropyl), C
3-6cycloalkyl, oxetanyl, tetrahydrofuranyl, tetrahydro-2H-pyranyl, morpholinyl or piperazinyl-C
1-4alkyl. [0329] In embodiments, R
3e is H. [0330] In embodiments, R
3e is F. [0331] In embodiments, R
4 is independently H. [0332] In embodiments, R
4 is independently C1-4alkyl. [0333] In embodiments, R
4 is independently halogen. [0334] In embodiments, R
4 is independently CF
3. [0335] In embodiments, R
4 is independently CHF
2. [0336] In embodiments, R
4 is independently CH2F. [0337] In embodiments, R
4 is independently cyclopropyl. [0338] In embodiments, two geminal R
4 groups together form a spiro-cyclopropyl.
[0339] In embodiments, each R
4 is independently H, CH
3, F, CF
3, CHF
2, or CH
2F. [0340] In embodiments, R
5 is independently H. [0341] In embodiments, R
5 is independently C1-4alkyl. [0342] In embodiments, R
5 is independently C3-5cycloalkyl. [0343] In embodiments, R
5 is independently C1-4alkyloxy. [0344] In embodiments, R
5 is independently C
3-5cycloalkoxy. [0345] In embodiments, R
5 is independently F. [0346] In embodiments, R
5 is independently CF
3. [0347] In embodiments, R
5 is independently CHF2. [0348] In embodiments, R
5 is independently CH2F. [0349] In embodiments, R
5 is independently OCF3. [0350] In embodiments, R
5 is independently OCHF
2. [0351] In embodiments, R
5 is independently OCH
2F. [0352] In embodiments, each R
5 is independently, H, C
1-4alkyl, C
3-5cycloalkyl, C
1-4alkyloxy, F, CF3, CHF2, CH2F, OCF3, OCHF2 or OCH2F. [0353] In embodiments, R
6 is independently H. [0354] In embodiments, R
6 is independently C1-4alkyl. [0355] In embodiments, R
6 is independently C
2-4alkenyl. [0356] In embodiments, R
6 is independently C
3-5cycloalkyl. [0357] In embodiments, R
6 is independently C
1-4alkyloxy. [0358] In embodiments, R
6 is independently C3-5cycloalkoxy. [0359] In embodiments, R
6 is independently F. [0360] In embodiments, R
6 is independently CF3. [0361] In embodiments, R
6 is independently CHF
2. [0362] In embodiments, R
6 is independently CH
2F. [0363] In embodiments, R
6 is independently OCF
3.
[0364] In embodiments, R
6 is independently OCHF
2. [0365] In embodiments, R
6 is independently OCH
2F. [0366] In embodiments, R
6 is independently OH. [0367] In embodiments, R
6 is independently 5-6-membered heteroaryl. [0368] In embodiments, R
6 is independently NRaRb. [0369] In embodiments, R
7 is independently C
1-4alkyl. [0370] In embodiments, R
7 is F. [0371] In embodiments, R
7 is CH
3 or F. [0372] In embodiments, Ra is independently hydrogen. [0373] In embodiments, Ra is independently C1-4alkyl. [0374] In embodiments, Ra is independently C3-5cycloalkyl. [0375] In embodiments, R
b is independently hydrogen. [0376] In embodiments, R
b is independently C
1-4alkyl. [0377] In embodiments, R
b is independently C
3-5cycloalkyl. [0378] In embodiments, Ra and Rb, together with the nitrogen atom to which they are attached, form a saturated or unsaturated heterocyclic ring containing from three to seven ring atoms, wherein said ring may optionally contain additional one or two heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur and may be optionally substituted by from one to three groups which may be the same or different selected from the group consisting of F, C
1-
4alkyl, phenyl and benzyl. [0379] In embodiments, Ra and Rb, together with the nitrogen atom to which they are attached, form a saturated heterocyclic ring. In embodiments, said heterocyclic ring is unsubstituted. In embodiments, said heterocyclic ring is substituted by 1, 2 or 3 groups independently selected from the group consisting of F, C
1-
4alkyl, phenyl and benzyl. In embodiments, said heterocyclic ring does not contain additional one or two heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur. In embodiments, said heterocyclic ring contains additional one or two heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur.
[0380] In embodiments, R
a and R
b, together with the nitrogen atom to which they are attached, form an unsaturated heterocyclic ring. In embodiments, said heterocyclic ring is unsubstituted. In embodiments, said heterocyclic ring is substituted by 1, 2 or 3 groups independently selected from the group consisting of F, C1-4alkyl, phenyl and benzyl. In embodiments, said heterocyclic ring does not contain additional one or two heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur. In embodiments, said heterocyclic ring contains additional one or two heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur. [0381] In embodiments, n is 0. [0382] In embodiments, n is 1. [0383] In embodiments, n is 2. [0384] In embodiments, m is 0. [0385] In embodiments, m is 1. [0386] In embodiments, m is 2. [0387] In embodiments, the Q ring i
[0388] In embodiments, the Q ring i
. [0389] In embodiments, R
1a is independently H. In embodiments, R
1a is independently C1-
4alkyl. In embodiments, R
1a is independently halogen. In embodiments, R
1a is independently C
1- 4alkyloxy. In embodiments, R
1a is independently OH. In embodiments, R
1a is independently C
1- 4alkyl-OH. In embodiments, R
1a is independently C
1-4alkyl-C
1-4alkyloxy. In embodiments, R
1a is independently C1-4alkyloxy-C1-4alkyloxy. In embodiments, R
1a is independently CF3. In embodiments, R
1a is independently CHF2. In embodiments, R
1a is independently CH2F. In embodiments, R
1a is independently CN. In embodiments, R
1a is independently NO2. In embodiments, R
1a is independently NR
aR
b. In embodiments, R
1a is independently C
1-4alkyl- NR
aR
b.
[0390] In embodiments, R
1b is independently H. In embodiments, R
1b is independently C
1- 4alkyl. In embodiments, R
1b is independently halogen. In embodiments, R
1b is independently C
1- 4alkyloxy. In embodiments, R
1b is independently OH. In embodiments, R
1b is independently C1- 4alkyl-OH. In embodiments, R
1b is independently C1-4alkyl-C1-4alkyloxy. In embodiments, R
1b is independently C1-4alkyloxy-C1-4alkyloxy. In embodiments, R
1b is independently CF3. In embodiments, R
1b is independently CHF
2. In embodiments, R
1b is independently CH
2F. In embodiments, R
1b is independently CN. In embodiments, R
1b is independently NO
2. In embodiments, R
1b is independently NR
aR
b. In embodiments, R
1b is independently C
1-4alkyl- NRaRb. [0391] In embodiments, R
1c is independently H. In embodiments, R
1c is independently C1- 4alkyl. In embodiments, R
1c is independently halogen. In embodiments, R
1c is independently C1-
4alkyloxy. In embodiments, R
1c is independently OH. In embodiments, R
1c is independently C
1- 4alkyl-OH. In embodiments, R
1c is independently C
1-4alkyl-C
1-4alkyloxy. In embodiments, R
1c is independently C
1-4alkyloxy-C
1-4alkyloxy. In embodiments, R
1c is independently CF
3. In embodiments, R
1c is independently CHF2. In embodiments, R
1c is independently CH2F. In embodiments, R
1c is independently CN. In embodiments, R
1c is independently NO2. In embodiments, R
1c is independently NRaRb. In embodiments, R
1c is independently C1-4alkyl- NR
aR
b. [0392] In embodiments, R
1d is independently H. In embodiments, R
1d is independently C
1- 4alkyl. In embodiments, R
1d is independently halogen. In embodiments, R
1d is independently C
1- 4alkyloxy. In embodiments, R
1d is independently OH. In embodiments, R
1d is independently C1- 4alkyl-OH. In embodiments, R
1d is independently C1-4alkyl-C1-4alkyloxy. In embodiments, R
1d is independently C1-4alkyloxy-C1-4alkyloxy. In embodiments, R
1d is independently CF3. In embodiments, R
1d is independently CHF
2. In embodiments, R
1d is independently CH
2F. In embodiments, R
1d is independently CN. In embodiments, R
1d is independently NO
2. In embodiments, R
1d is independently NR
aR
b. In embodiments, R
1d is independently C
1-4alkyl- NRaRb. [0393] In embodiments, R
1e is independently H. In embodiments, R
1e is independently C1- 4alkyl. In embodiments, R
1e is independently halogen. In embodiments, R
1e is independently C1-
4alkyloxy. In embodiments, R
1e is independently OH. In embodiments, R
1e is independently C
1- 4alkyl-OH. In embodiments, R
1e is independently C
1-4alkyl-C
1-4alkyloxy. In embodiments, R
1e is independently C
1-4alkyloxy-C
1-4alkyloxy. In embodiments, R
1e is independently CF
3. In embodiments, R
1e is independently CHF2. In embodiments, R
1e is independently CH2F. In
embodiments, R
1e is independently CN. In embodiments, R
1e is independently NO
2. In embodiments, R
1e is independently NR
aR
b. In embodiments, R
1e is independently C
1-4alkyl- NRaRb. [0394] In embodiments, R
1f is independently H. In embodiments, R
1f is independently C1- 4alkyl. In embodiments, R
1f is independently halogen. In embodiments, R
1f is independently C1-
4alkyloxy. In embodiments, R
1f is independently OH. In embodiments, R
1f is independently C
1- 4alkyl-OH. In embodiments, R
1f is independently C
1-4alkyl-C
1-4alkyloxy. In embodiments, R
1f is independently C
1-4alkyloxy-C
1-4alkyloxy. In embodiments, R
1f is independently CF
3. In embodiments, R
1f is independently CHF2. In embodiments, R
1f is independently CH2F. In embodiments, R
1f is independently CN. In embodiments, R
1f is independently NO2. In embodiments, R
1f is independently NRaRb. In embodiments, R
1f is independently C1-4alkyl- NR
aR
b. [0395] In embodiments, each of R
1c, R
1d, and R
1e is independently H; each R
1a is independently H; and/or each R
1b is is independently H or OMe. [0396] In embodiments, each of R
1a, R
1b, R
1c, R
1d, and R
1e is H. [0397] In embodiments, each of R
1a, R
1b, R
1d, and R
1e is H. [0398] In embodiments, each of R
1c and R
1d is H; and/or each of each R
1a, R
1b, and R
1f is independently H. [0399] In embodiments, each of R
1a, R
1b, R
1c, R
1d, and R
1f is H. [0400] In embodiments, each of R
1a, R
1c, R
1d, and R
1f is H. Exemplary Compounds [0401] Exemplary compounds include those described in Table 1 herein and pharmaceutically acceptable salts thereof. [0402] In embodiments, a compound may be used as a mixture of stereoisomers (e.g., a mixture of diastereomers or a mixture of enantiomers). In embodiments, a stereochemically enriched composition comprising a compound described herein (e.g., a composition of a compound is substantially free of any other stereoisomer of that compound). [0403] Where the absolute stereochemistry of a stereocenter is not indicated, it is understood that embodiments of the compound encompass (R)- and (S)- configurations in the alternative.
Likewise, such compounds may be used in methods described herein as compositions comprising stereochemical mixtures or as stereochemically-enriched compositions (e.g., compositions of a compound substantially free of any other stereoisomer of that compound). [0404] In embodiments, a compound is

(Compound 1A/1B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 1A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 1B, or a pharmaceutically acceptable salt thereof. [0405] In embodiments, a compound i
(Compound 2A/2B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 2A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 2B, or a pharmaceutically acceptable salt thereof. [0406] In embodiments, a compound i
(Compound 3A/3B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 3A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 3B, or a pharmaceutically acceptable salt thereof.
[0407] In embodiments, a compound i
(Compound 4A/4B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 4A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 4B, or a pharmaceutically acceptable salt thereof. [0408] In embodiments, a compound i
(Compound 5A/5B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 5A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 5B, or a pharmaceutically acceptable salt thereof. [0409] In embodiments, a compound i
(Compound 6A/6B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 6A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 6B, or a pharmaceutically acceptable salt thereof. [0410] In embodiments, a compound i
(Compound 7A/7B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 7A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 7B, or a pharmaceutically acceptable salt thereof.
[0411] In embodiments, a compound
(Compound 8A/8B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 8A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 8B, or a pharmaceutically acceptable salt thereof. [0412] In embodiments, a compound
(Compound 9A/9B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 9A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 9B, or a pharmaceutically acceptable salt thereof. [0413] In embodiments, a compound i
(Compound 10A/10B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 10A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 10B, or a pharmaceutically acceptable salt thereof. [0414] In embodiments, a compound i
(Compound 11A/11B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is
Compound 11A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 11B, or a pharmaceutically acceptable salt thereof. [0415] In embodiments, a compound
(Compound 12A/12B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 12A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 12B, or a pharmaceutically acceptable salt thereof. [0416] In embodiments, a compound
(Compound 13A/13B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 13A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 13B, or a pharmaceutically acceptable salt thereof. [0417] In embodiments, a compound
(Compound 14A/14B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 14A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 14B, or a pharmaceutically acceptable salt thereof. [0418] In embodiments, a compound
(Compound 15A/15B), or a pharmaceutically acceptable salt thereof. In embodiments, a
compound is Compound 15A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 15B, or a pharmaceutically acceptable salt thereof. [0419] In embodiments, a compound
(Compound 16A/16B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 16A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 16B, or a pharmaceutically acceptable salt thereof. [0420] In embodiments, a compound
(Compound 17A/17B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 17A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 17B, or a pharmaceutically acceptable salt thereof. [0421] In embodiments, a compound
(Compound 18A/18B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 18A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 18B, or a pharmaceutically acceptable salt thereof.
[0422] In embodiments, a compound i
(Compound 18C/18D), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 18C, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 18D, or a pharmaceutically acceptable salt thereof. [0423] In embodiments, a compound is
(Compound 19A/19B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 19A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 19B, or a pharmaceutically acceptable salt thereof. [0424] In embodiments, a compound
(Compound 19C/19D), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 19C, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 19D, or a pharmaceutically acceptable salt thereof. [0425] In embodiments, a compound
(Compound 20A/20B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 20A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 20B, or a pharmaceutically acceptable salt thereof.
[0426] In embodiments, a compound
(Compound 21A/21B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 21A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 21B, or a pharmaceutically acceptable salt thereof. [0427] In embodiments, a compound i
(Compound 22A/22B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 22A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 22B, or a pharmaceutically acceptable salt thereof. [0428] In embodiments, a compound i
(Compound 23A/23B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 23A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 23B, or a pharmaceutically acceptable salt thereof. [0429] In embodiments, a compound
(Compound 24A/24B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 24A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 24B, or a pharmaceutically acceptable salt thereof.
[0430] In embodiments, a compound i
(Compound 25A/25B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 25A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 25B, or a pharmaceutically acceptable salt thereof. [0431] In embodiments, a compound
(Compound 25C/25D), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 25C, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 25D, or a pharmaceutically acceptable salt thereof.
(Compound 26A/26B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 26A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 26B, or a pharmaceutically acceptable salt thereof.
(Compound 26C/26D), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 26C, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 26D, or a pharmaceutically acceptable salt thereof.
[0434] In embodiments, a compound
(Compound 27A/27B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 27A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 27B, or a pharmaceutically acceptable salt thereof. [0435] In embodiments, a compound
(Compound 28A/28B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 28A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 28B, or a pharmaceutically acceptable salt thereof. [0436] In embodiments, a compound i
(Compound 29A/29B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 29A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 29B, or a pharmaceutically acceptable salt thereof. [0437] In embodiments, a compound i
(Compound 30A/30B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 30A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 30B, or a pharmaceutically acceptable salt thereof.
(Compound 31A/31B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 31A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 31B, or a pharmaceutically acceptable salt thereof. [0439] In embodiments, a compound i
(Compound 32A/32B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 32A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 32B, or a pharmaceutically acceptable salt thereof. [0440] In embodiments, a compound i
(Compound 33A/33B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 33A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 33B, or a pharmaceutically acceptable salt thereof. [0441] In embodiments, a compound
(Compound 34A/34B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 34A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 34B, or a pharmaceutically acceptable salt thereof.
[0442] In embodiments, a compound
(Compound 35A/35B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 35A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 35B, or a pharmaceutically acceptable salt thereof. [0443] In embodiments, a compound
(Compound 100A/100B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 100A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 100B, or a pharmaceutically acceptable salt thereof. [0444] In embodiments, a compound
(Compound 101A/101B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 101A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 101B, or a pharmaceutically acceptable salt thereof. [0445] In embodiments, a compound
(Compound 102A/102B), or a pharmaceutically acceptable salt thereof. In embodiments, a
compound is Compound 102A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 102B, or a pharmaceutically acceptable salt thereof. [0446] In embodiments, a compound i
(Compound 103A/103B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 103A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 103B, or a pharmaceutically acceptable salt thereof. [0447]
(Compound 104A/104B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 104A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 104B, or a pharmaceutically acceptable salt thereof. [0448] In embodiments, a compound
(Compound 105A/105B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 105A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 105B, or a pharmaceutically acceptable salt thereof.
[0449] In embodiments, a compound
(Compound 106A/106B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 106A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 106B, or a pharmaceutically acceptable salt thereof. [0450] In embodiments, a compound
(Compound 107A/107B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 107A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 107B, or a pharmaceutically acceptable salt thereof. [0451] In embodiments, a compound i
(Compound 108A/108B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 108A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 108B, or a pharmaceutically acceptable salt thereof.
(Compound 109A/109B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 109A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 109B, or a pharmaceutically acceptable salt thereof. [0453] In embodiments, a compound
(Compound 110A/110B/110C/110D), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 110A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 110B, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 110C, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 110D, or a pharmaceutically acceptable salt thereof. [0454] In embodiments, a compound i
(Compound 111A/111B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 111A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 111B, or a pharmaceutically acceptable salt thereof. [0455] In embodiments, a compound
(Compound 112A/112B), or a pharmaceutically acceptable salt thereof. In embodiments, a
compound is Compound 112A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 112B, or a pharmaceutically acceptable salt thereof. [0456] In embodiments, a compound
(Compound 113A/113B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 113A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 113B, or a pharmaceutically acceptable salt thereof. [0457] In embodiments, a compound
(Compound 114A/114B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 114A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 114B, or a pharmaceutically acceptable salt thereof. [0458] In embodiments, a compound
(Compound 115A/115B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 115A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 115B, or a pharmaceutically acceptable salt thereof.
[0459] In embodiments, a compound is
(Compound 116A/116B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 116A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 116B, or a pharmaceutically acceptable salt thereof. [0460] In embodiments, a compound i
(Compound 117A/117B/117C/117D), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 117A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 117B, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 117C, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 117D, or a pharmaceutically acceptable salt thereof. [0461] In embodiments, a compound i
(Compound 118A/118B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 118A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 118B, or a pharmaceutically acceptable salt thereof.
[0462] In embodiments, a compound i
(Compound 119A/119B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 119A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 119B, or a pharmaceutically acceptable salt thereof. [0463]
(Compound 120A/120B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 120A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 120B, or a pharmaceutically acceptable salt thereof. [0464] In embodiments, a compound i
(Compound 121A/121B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 121A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 121B, or a pharmaceutically acceptable salt thereof. [0465] In embodiments, a compound i
(Compound 122A/122B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 122A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 122B, or a pharmaceutically acceptable salt thereof.
[0466] In embodiments, a compound i
(Compound 123A/123B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 123A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 123B, or a pharmaceutically acceptable salt thereof. [0467] In embodiments, a compound
(Compound 124A/124B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 124A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 124B, or a pharmaceutically acceptable salt thereof. [0468] In embodiments, a compound
(Compound 125A/125B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 125A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 125B, or a pharmaceutically acceptable salt thereof. [0469] In embodiments, a compound is
(Compound 126A/126B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 126A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 126B, or a pharmaceutically acceptable salt thereof.
[0470] In embodiments, a compound i
(Compound 127A/127B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 127A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 127B, or a pharmaceutically acceptable salt thereof. [0471] In embodiments, a compound i
(Compound 128A/128B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 128A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 128B, or a pharmaceutically acceptable salt thereof. [0472] In embodiments, a compound
(Compound 129A/129B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 129A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 129B, or a pharmaceutically acceptable salt thereof. [0473] In embodiments, a compound i
(Compound 130A/130B), or a pharmaceutically acceptable salt thereof. In embodiments, a
compound is Compound 130A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 130B, or a pharmaceutically acceptable salt thereof. [0474] In embodiments, a compound i
(Compound 131A/131B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 131A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 131B, or a pharmaceutically acceptable salt thereof. [0475] In embodiments, a compound
(Compound 132A/132B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 132A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 132B, or a pharmaceutically acceptable salt thereof. [0476] In embodiments, a compound
(Compound 133A/133B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 133A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 133B, or a pharmaceutically acceptable salt thereof. [0477] In embodiments, a compound
(Compound 134A/134B), or a pharmaceutically acceptable salt thereof. In embodiments, a
compound is Compound 134A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 134B, or a pharmaceutically acceptable salt thereof.
[0478] In embodiments, a compound
(Compound 135A/135B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 135A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 135B, or a pharmaceutically acceptable salt thereof. [0479] In embodiments, a compound i
(Compound 136A/136B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 136A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 136B, or a pharmaceutically acceptable salt thereof. [0480] In embodiments, a compound i
(Compound 137A/137B), or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 137A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 137B, or a pharmaceutically acceptable salt thereof. [0481] In embodiments, a compound i
(Compound 138A/138B), or a pharmaceutically acceptable salt thereof. In embodiments, a
compound is Compound 138A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 138B, or a pharmaceutically acceptable salt thereof. [0482] In embodiments, a compound is
pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 139A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is 139B, or a pharmaceutically acceptable salt thereof. [0483] In embodiments, a compound i

(Compound 140A/140B) , or a pharmaceutically acceptable salt thereof. In embodiments, a compound is Compound 140A, or a pharmaceutically acceptable salt thereof. In embodiments, a compound is 140B, or a pharmaceutically acceptable salt thereof. Deuterated Compounds [0484] Compounds described herein can comprise atoms that exhibit their natural isotopic abundances, or one or more of the atoms may be artificially enriched in a particular isotope having the same atomic number, but an atomic mass or mass number different from the atomic mass or mass number predominately found in nature. The term “isotopologue” refers to a species that has the same chemical structure and formula as a specific compound provided herein, with the exception of the positions of isotopic substitution and/or level of isotopic enrichment at one or more positions, e.g., hydrogen vs. deuterium. The present invention is meant to include all suitable isotopic variations of the compounds of the compounds described herein. For example, different isotopic forms of hydrogen (H) include protium (
1H), deuterium (
2H), and tritium (
3H), as well as compositions enriched in isotopologues of any compound described herein.
[0485] In embodiments, one or more of the hydrogens of the compounds described herein is replaced by a deuterium. When a position is designated as “H” or “hydrogen”, the position is understood to have hydrogen at its natural abundance isotopic composition. When a position is designated as “
2H” or “deuterium”, the position is understood to have deuterium at an abundance that is at least 3340 times greater than the natural abundance of deuterium, which is 0.015% (i.e., the term “
2H” or “deuterium” indicates at least 50.1% incorporation of deuterium). Accordingly, the invention also features compositions enriched in deuterated compounds. [0486] In embodiments, compositions of any compound provided herein may have an isotopic enrichment factor for each deuterium present at a site designated as a potential site of deuteration on the compound of at least 3500 (52.5% deuterium incorporation), at least 4000 (60% deuterium incorporation), at least 4500 (67.5% deuterium incorporation), at least 5000 (75% deuterium), at least 5500 (82.5% deuterium incorporation), at least 6000 (90% deuterium incorporation), at least 6333.3 (95% deuterium incorporation), at least 6466.7 (97% deuterium incorporation), at least 6600 (99% deuterium incorporation), or at least 6633.3 (99.5% deuterium incorporation). Exemplary Pharmaceutical Compositions [0487] Compounds described herein (e.g., compounds of any one of Formulas (I)-(VI)) or pharmaceutically acceptable salts thereof can be formulated in various pharmaceutical compositions. A compound described herein (e.g., a compound of Formula (I) (including compounds of Formulas (II)-(VI) and any compound of Table 1 as provided herein), as well as pharmaceutically acceptable salts thereof, may be the active pharmaceutical ingredient (API) combined with one or more other ingredients to form a drug substance pharmaceutical composition. The drug substance (DS) pharmaceutical composition can comprise the API (e.g., a compound of Formula (I) or pharmaceutically acceptable salt thereof) and one or more pharmaceutically acceptable carriers, diluents, and/or excipients. The carrier(s), diluent(s) or excipient(s) can be selected to be compatible with the other ingredients of the formulation and appropriately safe and effective for an intended therapy. A desired weight concentration of a compound described herein (e.g., a compound of Formula (I)) or a pharmaceutically acceptable salt thereof as the active pharmaceutical ingredient (API) can be combined with the other inactive ingredients to form a drug substance (DS) in a formulation batch. Pharmaceutically acceptable compositions can be formulated for administration by an appropriate route, for example by the
oral delivery (including as a capsule or tablet) in unit dosage forms. Such compositions may be prepared by bringing into association the active pharmaceutical ingredient (API) comprising a compound of Formula (I) with the carrier(s) or excipient(s). [0488] In certain embodiments, the invention provides a pharmaceutical composition formulated for oral delivery of an ^
4 ^
7 integrin integrin inhibitor, the composition comprising the ^4 ^7 integrin inhibitor compound described herein (e.g., a compound of any one of Formulas (I)- (VI) such as any compound of Table 1, or a pharmaceutically acceptable salt thereof) as an API and a pharmaceutically acceptable carrier formulated for oral therapeutic administration of the ^
4 ^
7 integrin inhibitor compound. [0489] In certain embodiments, the invention provides a pharmaceutical composition comprising a compound of Formula (I), or a pharmaceutically acceptable salt thereof as the active pharmaceutical ingredient (API). [0490] In certain embodiments, the invention provides a pharmaceutical composition comprising a compound of Formula (II), or a pharmaceutically acceptable salt thereof as the active pharmaceutical ingredient (API). [0491] In certain embodiments, the invention provides a pharmaceutical composition comprising a compound of Formula (IIA), or a pharmaceutically acceptable salt thereof as the active pharmaceutical ingredient (API). [0492] In certain embodiments, the invention provides a pharmaceutical composition comprising a compound of Formula (IIB), or a pharmaceutically acceptable salt thereof as the active pharmaceutical ingredient (API). [0493] In certain embodiments, the invention provides a pharmaceutical composition comprising a compound of Formula (III), or a pharmaceutically acceptable salt thereof as the active pharmaceutical ingredient (API). [0494] In certain embodiments, the invention provides a pharmaceutical composition comprising a compound of Formula (IIIA), or a pharmaceutically acceptable salt thereof as the active pharmaceutical ingredient (API). [0495] In certain embodiments, the invention provides a pharmaceutical composition comprising a compound of Formula (IIIB), or a pharmaceutically acceptable salt thereof as the active pharmaceutical ingredient (API).
[0496] In certain embodiments, the invention provides a pharmaceutical composition comprising a compound of Formula (IV), or a pharmaceutically acceptable salt thereof as the active pharmaceutical ingredient (API). [0497] In certain embodiments, the invention provides a pharmaceutical composition comprising a compound of Formula (IVA), or a pharmaceutically acceptable salt thereof as the active pharmaceutical ingredient (API). [0498] In certain embodiments, the invention provides a pharmaceutical composition comprising a compound of Formula (IVB), or a pharmaceutically acceptable salt thereof as the active pharmaceutical ingredient (API). [0499] In certain embodiments, the invention provides a pharmaceutical composition comprising a compound of Formula (V), or a pharmaceutically acceptable salt thereof as the active pharmaceutical ingredient (API). [0500] In certain embodiments, the invention provides a pharmaceutical composition comprising a compound of Formula (VA), or a pharmaceutically acceptable salt thereof as the active pharmaceutical ingredient (API). [0501] In certain embodiments, the invention provides a pharmaceutical composition comprising a compound of Formula (VB), or a pharmaceutically acceptable salt thereof as the active pharmaceutical ingredient (API). [0502] In certain embodiments, the invention provides a pharmaceutical composition comprising a compound of Formula (VI), or a pharmaceutically acceptable salt thereof as the active pharmaceutical ingredient (API). [0503] In certain embodiments, the invention provides a pharmaceutical composition comprising a compound of Table 1, or a pharmaceutically acceptable salt thereof as the active pharmaceutical ingredient (API). [0504] Pharmaceutically acceptable compositions comprising a compound described herein (e.g., a compound of Formula (I)) or a pharmaceutically acceptable salt thereof can be prepared by various procedures. For example, the compounds of Formula (I) can be formulated with suitable excipients, diluents, or carriers, and formed into tablets, or capsules, and other suitable dosage forms. [0505] Pharmaceutical compositions can be provided in unit dose forms containing a predetermined amount of API comprising a compound described herein (e.g., a compound of
Formula (I)) or a pharmaceutically acceptable salt thereof per unit dose. Such a unit may contain, a desired amount of a compound (e.g., a compound of the Formula (I)) or pharmaceutically acceptable salt thereof, depending on the condition being treated, the route of administration and the age, weight and condition of the patient. Such unit doses may therefore be administered at a desired dose interval. The concentration of active compound in the drug composition will depend on various applicable parameters and considerations such as the absorption, inactivation and excretion rates of the drug as well as other factors known to those of skill in the art. It is to be noted that dosage values will also vary with the severity of the condition to be alleviated. It is to be further understood that for any particular subject, specific dosage regimens should be adjusted over time according to the individual need and the professional judgment of the person administering or supervising the administration of the compositions, and that the concentration ranges set forth herein are exemplary only and are not intended to limit the scope or practice of the claimed composition. The active ingredient can be administered at once, or can be divided into a number of smaller doses to be administered at varying intervals of time. [0506] In certain embodiments, the mode of administration of the active compound is oral. Oral compositions will generally include an inert diluent or an edible carrier. They can be enclosed in gelatin capsules or compressed into tablets. For the purpose of oral therapeutic administration, the active compound can be incorporated with excipients and used in the form of tablets, troches or capsules. Pharmaceutically compatible binding agents, and/or adjuvant materials can be included as part of the composition. Pharmaceutical compositions comprising a compound described herein (e.g., a compound of Formula (I)) or a pharmaceutically acceptable salt thereof formulated for oral delivery can be prepared in a unit dosage form, such as a capsule at a desired dosage strength (e.g., of the compound of Formula (I) or a pharmaceutically acceptable salt thereof). For oral administration in liquid form, the oral drug components can be combined with any oral, non-toxic, pharmaceutically acceptable inert carrier such as ethanol, glycerol, water and the like. For oral administration in the form of a tablet or capsule, a compound described herein (e.g., a compound of Formula (I)) or a pharmaceutically acceptable salt thereof can be combined with an oral, non-toxic, pharmaceutically acceptable, inert carrier. Other examples of excipients, diluents, and carriers that are suitable for such formulations include the following: fillers and extenders such as starch, and sugars; and binding agents such as cellulose derivatives. Moreover, when desired or necessary, suitable binders, lubricants, disintegrating agents and coloring agents can also be incorporated into the mixture. Suitable
binders include starch, natural sugars, natural and synthetic gums, and the like. Lubricants and/or glidants can be used in these dosage forms. [0507] The tablets, pills, capsules, troches and the like can contain any of the following ingredients, or compounds of a similar nature: a binder such as microcrystalline cellulose, gum tragacanth or gelatin; an excipient such as starch or lactose, a disintegrating agent such as alginic acid, Primogel or corn starch; a lubricant such as magnesium stearate or Sterotes; a glidant such as colloidal silicon dioxide; a sweetening agent such as sucrose or saccharin; or a flavoring agent such as peppermint, methyl salicylate, or orange flavoring. When the dosage unit form is a capsule, it can contain, in addition to material of the above type, a liquid carrier such as a fatty oil. In addition, unit dosage forms can contain various other materials that modify the physical form of the dosage unit, for example, coatings of sugar, or other enteric agents. [0508] The compound can be administered as a component of an elixir, suspension, syrup, wafer, or the like. A syrup can contain, in addition to the active compound(s), sucrose or sweetener as a sweetening agent and certain preservatives, dyes and colorings and flavors. [0509] The compounds can be formulated as solutions appropriate for parenteral administration, for example, by intramuscular, subcutaneous or intravenous routes. For example, a compound described herein (e.g., a compound of Formula (I)) or a pharmaceutically acceptable salt thereof can be dissolved in a suitable buffer. A pharmaceutical composition comprising a desired concentration of a compound described herein (e.g., a compound of Formula (I)) or a pharmaceutically acceptable salt thereof can be formulated as an injectable drug solution in (useful, e.g., in preclinical animal studies). Exemplary Therapeutic Methods [0510] Compounds described herein can be useful for the treatment of various diseases and disorders that benefit from antagonizing the integrin α
vβ
8. For example, inhibiting the integrin α
vβ
8-driven TGFβ activation is proposed to reverse tumor tolerance and enhance anti-tumor T/NK cell responses. For example, αvβ8 inhibition can enhance outcomes in checkpoint inhibitors regimens or reverse checkpoint inhibitor resistance. In embodiments, a compound described herein can modulate an anti-tumor immune response (e.g., in checkpoint-inhibitor resistant tumors). [0511] In embodiments, the invention features a method of inhibiting α
vβ
8 integrin in a patient, said method comprising administering to a patient in need thereof a therapeutically
effective amount of a compound described herein (e.g., a compound of any one of Formulas (I)- (VI) such as any compound of Table 1) or a pharmaceutically acceptable salt thereof. [0512] In embodiments, the invention features a method of treating a cancer (e.g., a solid tumor) in a patient, said method comprising administering to a patient in need thereof a therapeutically effective amount of a compound described herein (e.g., a compound of any one of Formulas (I)-(VI) such as any compound of Table 1) or a pharmaceutically acceptable salt thereof. In embodiments, a method further comprises administration of a therapeutically effective amount of a second active agent. Solid Tumors [0513] In embodiments, administration of a compound described herein (e.g., a compound of any one of Formulas (I)-(VI) such as any compound of Table 1) or a pharmaceutically acceptable salt thereof can be useful for treating a solid tumor in a patient in need thereof, optionally in combination with one or more additional therapies (e.g., a second active agent). [0514] In embodiments, a solid tumor is resistant to one or more previous lines of therapy (e.g., a solid tumor is a treatment-resistant tumor). In embodiments, a solid tumor is resistant to immune checkpoint therapy. [0515] In embodiments, a cancer is a solid tumor such as fibrosarcoma, myxosarcoma, liposarcoma, chondrosarcoma, osteogenic sarcoma, chordoma, angiosarcoma, endotheliosarcoma, lymphangiosarcoma, lymphangioendotheliosarcoma, synovioma, mesothelioma, Ewing’s tumor, leiomyosarcoma, rhabdomyosarcoma, osteosarcoma, colon cancer, colorectal cancer, kidney cancer, pancreatic cancer, bone cancer, breast cancer, ovarian cancer, prostate cancer, esophageal cancer, stomach cancer, oral cancer, nasal cancer, throat cancer, squamous cell carcinoma, basal cell carcinoma, adenocarcinoma, sweat gland carcinoma, sebaceous gland carcinoma, papillary carcinoma, papillary adenocarcinomas, cystadenocarcinoma, medullary carcinoma, bronchogenic carcinoma, renal cell carcinoma, hepatoma, bile duct carcinoma, choriocarcinoma, seminoma, embryonal carcinoma, Wilms tumor, cervical cancer, uterine cancer, testicular cancer, non small cell lung cancer (NSCLC), small cell lung carcinoma, bladder carcinoma, lung cancer, epithelial carcinoma, skin cancer, melanoma, neuroblastoma (NB), or retinoblastoma. In embodiments, a solid tumor is an advanced stage solid tumor (e.g., a locally advanced solid tumor). In embodiments, a solid tumor is a metastatic solid tumor.
[0516] In embodiments, a cancer (e.g., solid tumor) is anal cancer, bile duct cancer (cholangiocarcinoma), bladder cancer, breast cancer, cervical cancer, colorectal cancer, endometrial cancer, esophageal cancer, fallopian tube cancer, gastric cancer, glioma liver cancer, lung cancer, melanoma, nasopharyngeal carcinoma, neuroblastoma, osteosarcoma, ovarian cancer, pancreatic cancer, primary peritoneal carcinoma, prostate cancer, renal cell carcinoma, skin cancer, squamous cell carcinoma of the head and neck (SCCHN), testicular cancer, urothelial carcinoma, or uterine cancer. [0517] In embodiments, a cancer (e.g., solid tumor) is breast cancer, squamous cell carcinoma of the head and neck (SCCHN), renal cell carcinoma, ovarian cancer, gastric cancer, esophageal cancer, lung cancer, pancreatic cancer, bile duct cancer, endometrial cancer, melanoma, or urothelial carcinoma. [0518] In embodiments, a cancer (e.g., a solid tumor) is advanced (e.g., locally advanced). [0519] In embodiments, a cancer (e.g., a solid tumor) is metastatic. [0520] In embodiments, a cancer (e.g., solid tumor) is anal cancer. [0521] In embodiments, a cancer (e.g., solid tumor) is bile duct cancer (cholangiocarcinoma). [0522] In embodiments, a cancer (e.g., solid tumor) is bladder cancer. [0523] In embodiments, a cancer (e.g., solid tumor) is breast cancer. [0524] In embodiments, a cancer (e.g., solid tumor) is cervical cancer. [0525] In embodiments, a cancer (e.g., solid tumor) is colorectal cancer. [0526] In embodiments, a cancer (e.g., solid tumor) is endometrial cancer. [0527] In embodiments, a cancer (e.g., solid tumor) is esophageal cancer. In embodiments, an esophageal cancer is adenocarcinoma. In embodiments, an esophageal cancer is squamous cell carcinoma. [0528] In embodiments, a cancer (e.g., solid tumor) is fallopian tube cancer. [0529] In embodiments, a cancer (e.g., solid tumor) is gastric cancer. [0530] In embodiments, a cancer (e.g., solid tumor) is glioma. [0531] In embodiments, a cancer (e.g., solid tumor) is liver cancer. In embodiments, a liver cancer is hepatocellular carcinoma.
[0532] In embodiments, a cancer (e.g., solid tumor) is lung cancer. In embodiments, a lung cancer is squamous cell carcinoma of the lung. In embodiments, a lung cancer is non small cell lung cancer (NSCLC). [0533] In embodiments, a cancer (e.g., solid tumor) is melanoma. [0534] In embodiments, a cancer (e.g., solid tumor) is nasopharyngeal carcinoma. [0535] In embodiments, a cancer (e.g., solid tumor) is neuroblastoma. [0536] In embodiments, a cancer (e.g., solid tumor) is osteosarcoma. [0537] In embodiments, a cancer (e.g., solid tumor) is ovarian cancer. [0538] In embodiments, a cancer (e.g., solid tumor) is pancreatic cancer. [0539] In embodiments, a cancer (e.g., solid tumor) is primary peritoneal carcinoma. [0540] In embodiments, a cancer (e.g., solid tumor) is prostate cancer. [0541] In embodiments, a cancer (e.g., solid tumor) is renal cell carcinoma (RCC). In embodiments, a renal cell carcinoma is clear cell renal cell carcinoma (ccRCC). In embodiments, a renal cell carcinoma is papillary renal cell carcinoma (PRCC). [0542] In embodiments, a cancer (e.g., solid tumor) is skin cancer. In embodiments, a skin cancer is cutaneous squamous cell carcinoma (CSCC). In embodiments, a skin cancer is basal cell carcinoma (BCC). [0543] In embodiments, a cancer (e.g., solid tumor) is squamous cell carcinoma of the head and neck (SCCHN). [0544] In embodiments, a cancer (e.g., solid tumor) is testicular cancer. [0545] In embodiments, a cancer (e.g., solid tumor) is urothelial carcinoma. [0546] In embodiments, a cancer (e.g., solid tumor) is uterine cancer. [0547] In embodiments, a patient has a cancer (e.g., a solid tumor such as those described herein) that is therapy resistant. In embodiments, a therapy resistant cancer (e.g., a solid tumor) is checkpoint resistant. In embodiments, a therapy resistant cancer (e.g., a solid tumor) is resistant to an anti-PD-1 or anti-PD-L1 therapy (collectively, a PD-(L)1 resistant cancer).
Exemplary Combination Therapies and Second Active Agents [0548] In embodiments, a compound described herein (e.g., a compound of any one of Formulas (I)-(VI) such as any compound of Table 1) or a pharmaceutically acceptable salt thereof, may be used in combination therapies. In embodiments, administration of a compound or pharmaceutically acceptable salt described herein enhances response to one or more additional therapies (e.g., a second active agent). For example, administration of a compound or pharmaceutically acceptable salt described herein can improve response to additional therapy (e.g., a second active agent). [0549] For example, a compound or pharmaceutically acceptable salt described herein may be administered in combination with one or more additional therapies (e.g., a second active agent) in the treatment of certain diseases and disorders. [0550] In embodiments, a compound or pharmaceutically acceptable salt described herein may be administered in combination with an immunotherapy. In embodiments, an immunotherapy is a cancer immunotherapy. In embodiments, a cancer immunotherapy is an immune checkpoint therapy (e.g., therapy comprising administration of an immune checkpoint inihbitor). In embodiments, a cancer immunotherapy is a cellular immunotherapy such as adoptive T cell transfer therapy (e.g., Chimeric Antigen Receptor (CAR) T cell therapy, CAR natural killer (NK) cell therapy, tumor infiltrating lymphocyte (TIL) therapy, or endogenous T cell (ETC) therapy). In embodiments, a cancer immunotherapy is a cancer vaccine. In embodiments, a cancer immunotherapy is monoclonal antibody therapy (e.g., an antibody useful for immune checkpoint therapy such as those described herein). In embodiments, a cancer immunotherapy is cytokine therapy (e.g., interferon or interleukin therapy). [0551] In embodiments, a compound or pharmaceutically acceptable salt described herein is administered with a second active agent to a patient in need thereof for the treatment of a solid tumor (e.g., as described herein). [0552] In embodiments, a second active agent is an immune checkpoint inhibitor. In embodiments, an immune checkpoint inhibitor targets PD-1 (e.g., inhibition via anti-PD-1, anti- PD-L1, or anti-PD-L2 therapies), CTLA-4, TIM-3, TIGIT, LAGs (e.g., LAG-3), CEACAM (e.g., CEACAM-1, -3 and/or -5), VISTA, BTLA, LAIR1, CD160, 2B4, CD80, CD86, B7-H3 (CD276), B7-H4 (VTCN1), HVEM (TNFRSF14 or CD270), KIR, A2aR, MHC class I, MHC class II, GALS, adenosine, TGFR (e.g., TGFR beta), B7-H1, B7-H4 (VTCN1), OX-40, CD137, CD40, IDO, or CSF-1/CSF-1R.
[0553] In embodiments, a checkpoint inhibitor is a small molecule, a nucleic acid, a polypeptide (e.g., an antibody), a carbohydrate, a lipid, a metal, a toxin, or a binding agent. In embodiments, a checkpoint inhibitor is an antibody, an antibody conjugate, or an antigen-binding fragment thereof. [0554] In embodiments, an immune checkpoint inhibitor is an agent that inhibits PD-1, TIM- 3, CTLA-4, LAG-3, TIGIT, IDO or CSF-1/CSF-1R. [0555] In embodiments, an immune checkpoint inhibitor is selected from: pembrolizumab (Keytruda®), nivolumab (Opdivo®), cemiplimab (Libtayo®), dostarlimab (Jemperli®), atezolizumab (Tecentriq®), avelumab (Bavencio®), durvalumab (Imfinzi®), ipilimumab (Yervoy®), and relatlimab, as well as biosimilars thereof. [0556] In embodiments, a second active agent is an anti-PD-1 therapy or an anti-PD-L1 therapy (collectively referred to as anti-PD(L)-1 therapy). [0557] In embodiments, an anti-PD(L)-1 therapy is selected from the group consisting of: pembrolizumab, nivolumab, cemiplimab, dostarlimab, atezolizumab, avelumab, durvalumab, PDR001, Cemiplimab, BGB-A317, LY3300054, BI 754091, IBI308, INCSHR-1210, JNJ-63723283, JS-001, MEDI0680 (AMP-514), MGA-012, PF-06801591, CX-072, FAZ053, and PD-L1 millamolecule, as well as biosimilars thereof. [0558] In embodiments, an anti-PD(L)-1 therapy is selected from atezolizumab, avelumab, BGB-A317, BI 754091, CX-072, durvalumab, FAZ053, IBI308, INCSHR-1210, JNJ-63723283, JS-001, MEDI-0680, MGA-012, nivolumab, PDR001, pembrolizumab, PF-06801591, cemiplimab, dostarlimab, any of the antibodies disclosed in WO2014/179664, as well as biosimilarsthereof. In embodiments, an anti-PD(L)-1 therapy is selected from the group consisting of BGB-A317, BI 754091, CX-072, FAZ053, IBI308, INCSHR-1210, JNJ-63723283, JS-001, LY3300054, MEDI-0680, MGA-012, nivolumab, PD-L1 millamolecule, PDR001, pembrolizumab, PF-06801591, cemiplimab, and dostarlimab, as well as biosimilars thereof. [0559] In embodiments, an anti-PD(L)-1 therapy is selected from pembrolizumab, nivolumab, atezolizumab, durvalumab, avelumab, dostarlimab, PDR-001, tislelizumab (BGB- A317), cemiplimab (REGN2810), LY-3300054, JNJ-63723283, MGA012, BI-754091, IBI-308, camrelizumab (HR-301210), BCD-100, JS-001, CX-072, BGB-A333, AMP-514 (MEDI-0680), AGEN-2034, CS1001, Sym-021, SHR-1316, PF-06801591, LZM009, KN-035, AB122, genolimzumab (CBT-501), FAZ-053, CK-301, AK 104, GLS-010, JTX-4014, SHR-1210, AMP- 224, AUN-P12, CA-170, BMS-986189, and any of the PD-1 antibodies disclosed in
WO2014/179664. In embodiments, an anti-PD(L)-1 therapy is selected from durvalumab, atezolizumab, avelumab, BGB-A333, SHR-1316, FAZ-053, CK-301, and PD-L1 millamolecule, or derivatives thereof. [0560] In embodiments, an anti-PD(L)-1 therapy is selected from nivolumab, pembrolizumab, cemiplimab, dostarlimab, atezolizumab, avelumab, and durvalumab. In embodiments, an anti-PD(L)-1 therapy is selected from nivolumab, pembrolizumab, cemiplimab, and dostarlimab. In embodiments, an anti-PD(L)-1 therapy is selected from atezolizumab, avelumab, and durvalumab. [0561] In embodiments, a second active agent is an anti-CTLA-4 therapy. In embodiments, an anti-CTLA-4 therapy is ipilimumab. [0562] In embodiments, a second active agent is an anti-LAG-3 therapy. In embodiments, an anti-LAG-3 therapy is selected from: LAG525 (IMP701), REGN3767 (R3767), BI 754,091, tebotelimab (MGD013), eftilagimod alpha (IMP321), TSR-033, and FS118. [0563] In embodiments, a second active agent is an anti-TIM-3 therapy. In embodiments, an anti-TIM-3 therapy is selected from: MBG453, Sym023, and TSR-022. [0564] In embodiments, a second active agent is an anti-CSF-1/R therapy. In embodiments, an anti-CSF-1/R therapy is selected from: lacnotuzumab (MCS110), LY3022855, SNDX-6352, emactuzumab (RG7155), and pexidartinib (PLX3397). [0565] In embodiments, a second active agent is an anti-TIGIT therapy. In embodiments, an anti-TIGIT therapy is selected from: BMS-986207, ociperlimab, tiragolumab, vibostolimab, domvanalimab, EOS448, COM902, and AGEN307.In embodiments, a second active agent is an anti-CEACAM therapy. In embodiments, an anti-TIGIT therapy is selected from: CM24 and NEO-201.
Examples General Schemes General Scheme 1:
wherein Q, L, R
2, R
7, m, R
3e, R
3d, R
3c, R
3b, R
3a are defined as in Formula (I) and B(OR)
2 is a boronic acid or boronic ester [0566] A general method of preparing compounds of Formula (I) is outlined in General Scheme 1. Treatment of a pyrrolidine (or pyrrolidine-HCl salt) 2-a, an aryl boronic acid or ester (i.e., R=H or alkyl, respectively) 2-b, oxoacetic acid and 4A molecular sieves in a solvent (i.e, DCM/hexafluoroisopropanol) at an elevated temperature (i.e., 60 °C) provides diastereomeric compounds of Formula (I) which could be separated by supercritical fluid chromatography (SFC) or prep-HPLC. General Scheme 2:
wherein Q, L, R
2, R
7, m, R
3e, R
3d, R
3c, R
3b and R
3a are defined as in Formula (I) and R is an alkyl group [0567] Alternatively, compounds of Formula (I) can be prepared as outlined in General Scheme 2. Treatment of a 2-bromo-2-arylacetate 2-c (i.e., R= Et or t-Bu) and a pyrrolidine (or
pyrrolidine-HCl salt) 2-a in a solvent (i.e., acetonitrile) in the presence of base (i.e., DIEA) at ambient to slightly elevated temperatures (i.e., 25-50 °C) yields an amino ester 2-d. Hydrolysis of 2-d in basic conditions (i.e., LiOH/MeOH for R=CH3) or under acidic conditions (i.e., formic acid for R=t-Bu) yields diastereomeric compounds of Formula (I), which could be separated by supercritical fluid chromatography (SFC) or prep-HPLC. Abbreviations Abbreviation Chemical Name 4A MS Molecular sieves, 4A, powder 9-BBN 9-Borabicyclo[3.3.1]nonane dimer AcOH acetic acid AgOTf silver trifluoromethanesulfonate AIBN 2,2-Azobis(2-methylpropionitrile) B(O-iPr)
3 triisopropyl borate BBr3 tribromoborane BH
3 ^THF borane tetrahydrofuran complex Cs2CO3 cesium carbonate CuI copper(I) iodide DBU 1,8-diazabicyclo[5.4.0]undec-7-ene DCM dichloromethane DIEA N,N-diisopropylethylamine DMF dimethyl formamide DMSO dimethyl sulfoxide EtOAc ethyl acetate EtOH ethanol H2 hydrogen H
2O
2 hydrogen peroxide H2SO4 sulfuric acid HCl hydrochloric acid HFIP 1,1,1,3,3,3-hexafluoropropan-2-ol i-PrMgBr isopropylmagnesium bromide i-PrMgCl ^LiCl isopropylmagnesium chloride-lithium chloride complex K
2CO
3 potassium carbonate K3PO4 potassium phosphate KF potassium fluoride KI potassium iodide KOAc potassium acetate LDA lithium diisopropyl amide LiHMDS lithium bis(trimethylsilyl)azanide Lindlar catalyst palladiumoncalciumcarbonate LiOH lithium hydroxide MeCN acetonitrile MeI iodomethane
MeOH methanol Mg magnesium MTBE tert-butyl methyl Ether Na2SO3 disodium sulfite Na
2SO
4 sodium sulfate NaH sodium hydride NaHCO
3 sodium bicarbonate NaI sodium iodide sodium acetate sodium hydroxide N-bromosuccinimide ammonium hydroxide ammonium chloride ammoniumhydrogencarbonate N-iodosuccinimide
tricyclohexylphosphine Pd(dba)
2 bis(dibenzylideneacetone)palladium Pd(dppf)Cl2 1,1-bis(diphenylphosphino)ferrocene]-dichloropalladium(II) Pd(OAc)
2 palladium (II) acetate Pd(t-Bu3P)2 bis(tri-tert-butylphosphine)palladium(0) Pd/C palladium on activated charcoal Pd
2(dba)
3 Tris(dibenzylideneacetone)dipalladium(0) PPh
3 triphenylphosphane prep-HPLC preparative high performance liquid chromatography Phos pentaphenyl(di-tert-butylphosphino)ferrocene S
electfluor 1-(chloromethyl)-4-fluoro-1,4- d
iazoniabicyclo[2.2.2]octane;ditetrafluoroborate SFC supercritical fluid chromatography SiO2 silicon dioxide TBAB tetrabutylazanium bromide TBAF tetrabutylazanium fluoride t-BuOK potassium tert-butoxide TEA triethylamine TFA trifluoroacetic acid TFAA trifluoroacetic anhydride THF tetrahydrofuran TMSCF3 trimethyl(trifluoromethyl)silane TMSCl chloro(trimethyl)silane ZnCl2 zinc chloride Analytical Methods, Materials, and Instrumentation
[0568] Unless otherwise noted, reagents and solvents were used as received from commercial suppliers. Default methods, materials and instrumentation are listed below. HNMR instrumental information: [0569] Proton nuclear magnetic resonance (NMR) spectra were obtained on either Bruker or Varian spectrometers at 400 MHz. Spectra are given in ppm (d) and coupling constants, J, are reported in Hertz. Tetramethylsilane (TMS) was used as an internal standard. LCMS instrumental information and analysis method: [0570] Mass spectra were collected using an Agilent 6120 Single Quad Mass Spectrometer (ESI). Purity and low resolution mass spectral data were measured using Agilent 1260 Infinity II high-performance liquid chromatography (HPLC) system with Photo Diode Array Detector, Agilent 1260 Infinity II Evaporative Light Scattering Detector (ELSD) and Agilent 6120 Mass Spectrometer. Data was acquired using Chemstation C.01.10 and purity characterized by UV wavelength 220 nm, ELSD and ESI. [0571] Method A: Column: Kinetex C182.1 x 50 mm, 5 um; Flow rate 1.0 mL/min; Mobile phase A: water with 0.04% TFA, Mobile phase B: acetonitrile with 0.02% trifluoroacetic acid; Gradient: 5% B from 0-0.40 min, then a gradient of 5-95% B over 2.60 min, then hold on 95% B for 1.00 min, then 95-5% B over 0.01 min. [0572] Method B: Column: Xbridge C182.1 x 50 mm column (5 um particles); Flow rate 0.8 mL/min; Mobile phase A: water with 10 mM NH
4HCO
3; Mobile phase B: acetonitrile; Gradient: 5% B from 0-0.40 min, then a gradient of 5-95% B from 0.40-3.40 min, then hold 95% B for 0.45 min, then 95-5% B over 0.01 min. SFC instrumental and analytical methods information: [0573] Instrument: Waters UPCC with PDA detector [0574] Conditions: Mobile phase A: CO
2; Mobile phase B: MeOH with 0.1% isopropylamine (or EtOH with 0.1% isopropylamine, or isopropanol with 0.1% isopropylamine). [0575] Columns: Daicel CHIRALPAK AD-3, 50 × 4.6 mm I.D., 3 um; Daicel CHIRALCEL OD-3, 50 × 4.6 mm I.D., 3 um; Daicel CHIRALCEL OJ-3, 50 × 4.6 mm I.D., 3 um; Daicel CHIRALPAK IG-3, 50 × 4.6 mm I.D., 3 um; Daicel CHIRALPAK AS-3, 50 × 4.6 mm I.D., 3 um; Daicel CHIRALPAK IC-3, 50 x 4.6 mm I.D., 3 um; Phenomenex Lux Cellulose-2, 50 × 4.6 mm I.D., 3 um; Regis (S,S)-Whelk-O 1, 50 × 4.6 mm I.D., 3.5 um
[0576] Column temperature: 35°C [0577] Gradient: 5% B from 0.0-0.2 min, then a gradient of 5-50% B from 0.2-1.2 min, then hold 50% B for 1.0 min, then a gradient of 50-5% B from 2.2-2.6 min, then hold 50% B for 0.4 min. [0578] Flow rate: 3.4 mL/min [0579] Automated back pressure regulator (ABPR): 1800 psi [0580] Alternatively, products may be analyzed by chiral SFC using an SFC-80 (Thar, Waters) instrument, detection wavelength 214 nm, using the “Chiral SFC B” method: column: AD 4.6*100mm, 5 µm (Daicel), column temperature: 40 °C, mobile phase: CO2/methanol (0.2% methanol ammonia), isocratic elution as in text, flow rate: 4 g/min, back pressure: 120 bar. Preparative HPLC Method [0581] Instruments: Gilson 281 semi-preparative HPLC system [0582] Conditions: Mobile Phase A: water with 0.2% formic acid (or water with 0.1% TFA, or water with 0.05% HCl, or water with 10 mM NH
4HCO
3 or water with 0.04% ammonium hydroxide); Mobile Phase B: acetonitrile [0583] Columns: Phenomenex Luna C18100 x 30 mm x 5um, Phenomenex Gemini C18100 x 30 mm x 5 um; Waters Xbridge BEH C18100 x 30 mm x 10 um [0584] Column temperature: ambient [0585] LC gradient: gradient of 5% to 50% B over 10 min, then hold 50% B for 2 min, then a gradient of 50-100% Bover 0.1 min, then hold at 100% for 2 min, then a gradient of 100% to 5% over 0.1 min, then hold at 5% for 2 min [0586] LC flow rate: 25 mL/min [0587] UV wavelength: 220 nm and 254 nm [0588] Alternatively, crude samples may be dissolved in MeOH and purified by prep HPLC using a Gilson 215 instrument, detection wavelength 214 nm, using one of the following methods noted in the procedures:
[0589] “Prep-HPLC A”: column: XBridge C18, 21.2 x 250 mm, 10 µm; Mobile phase A: water (10 mM ammonium hydrogen carbonate), Mobile phase B: acetonitrile; gradient elution as in text; flow rate: 20 mL/min. [0590] “Prep-HPLC B”: column: XBridge C18, 21.2 x 250 mm, 10 µm; Mobile phase A: water (10 mM formic acid), Mobile phase B: acetonitrile; gradient elution as in text; flow rate: 20 mL/min. [0591] Preparative Chiral HPLC method [0592] Instruments: Gilson-281 semi preparative HPLC system; UV: Gilson-156 UV [0593] Conditions: Mobile Phase A: n-heptane; Mobile Phase B: ethanol with 0.1% ammonium hydroxide (or 2-propanol with 0.1% ammonium hydroxide, or ethanol with 0.1% TFA, or 2-propanol with 0.1% TFA). [0594] Columns: Daicel CHIRALPAK AD, 10 um, 30 mm x 250 mm; Daicel CHIRALPAK IH, 10 um, 30 mm x 250 mm; Daicel CHIRALCEL OD, 10 um, 30 mm x 250 mm; Daicel CHIRALCEL OJ, 10 um, 30 mm x 250 mm; Daicel CHIRALPAK IC, 10 um, 30 mm x 250 mm; Daicel CHIRALPAK IG, 10 um, 30 mm x 250 mm; Phenomenex Lux Cellulose-2, 10 um, 30 mm x 250 mm; Regis (S,S) Whelk-O1, 10 um, 30 mm x 250 mm [0595] Column temperature: ambient [0596] LC gradient: A:B = 80:20 or other ratio of isocratic elution mode [0597] LC flow rate: 25 mL/min binary pump [0598] UV wavelength: 220 nm and 254 nm [0599] Preparative Chiral SFC methods [0600] Instruments: SFC: Waters 80Q preparative SFC; UV: Waters 2489 UV [0601] Conditions: Mobile Phase A: CO2; Mobile Phase B: methanol with 0.1% ammonium hydroxide (or ethanol with 0.1% ammonium hydroxide, or 2-propanol with 0.1% ammonium hydroxide, or MeCN/EtOH/ammonium hydroxide 50:50:0.1) [0602] Columns: Daicel CHIRALPAK AD, 10 um, 30 mm x 250 mm; Daicel CHIRALPAK IH, 10 um, 30 mm x 250 mm; Daicel CHIRALCEL OD, 10 um, 30 mm x 250 mm; Daicel CHIRALCEL OJ, 10 um, 30 mm x 250 mm; Daicel CHIRALPAK IC, 10 um, 30 mm x 250 mm; Daicel CHIRALPAK IG, 10 um, 30 mm x 250 mm; Phenomenex Lux Cellulose-2, 10 um, 30 mm x 250 mm; Regis (S, S) Whelk-O1, 10 um, 30 mm x 250 mm;
[0603] Column temperature: 40 °C [0604] LC gradient: A:B = 80:20 or other ratios of isocratic elution mode [0605] LC flow rate: 70 g/min binary pump [0606] UV wavelength: 220 nm [0607] System back pressure: 100 bar [0608] Alternatively, racemic products could be separated to individual enantiomers by chiral Prep SFC using an SFC-80 (Thar, Waters) instrument, detection wavelength 214 nm using the “Prep chiral SFC B” method: column: AD 20*250mm, 10 µm (Daicel), column temperature: 35 °C, mobile phase: CO2/methanol (0.2% methanol ammonia)= 60/40, flow rate: 80 g/min, back pressure: 100 bar. Left-Side Intermediates Preparation of tert-butyl 7-(5-oxopentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate Step 1: tert-butyl(pent-4-en-1-yloxy)diphenylsilane

[0609] To a solution of pent-4-en-1-ol (30 g, 348.30 mmol, 35.46 mL) in DCM (300 mL) was added imidazole (47.42 g, 696.61 mmol), tert-butylchlorodiphenylsilane (107.00 g, 389.29 mmol) at 0 °C. The mixture was stirred at 25 °C for 16 h. The reaction mixture was quenched by water (200 mL), extracted with DCM (100 mL x 2). The combined organic layers were washed with brine (100 mL), dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO2, petroleum ether/ethyl acetate = 1/0 to 1/1). tert-butyl(pent-4-en-1-yloxy)diphenylsilane (100 g, 246.51 mmol, 70.77% yield) was obtained as colorless oil.
1H NMR (400MHz, CHLOROFORM-d) δ = 7.71-7.65 (m, 4H), 7.44-7.35 (m, 6H), 5.81 (tdd, J =6.7, 10.3, 17.0 Hz, 1H), 5.07-4.93 (m, 2H), 3.69 (t, J =6.3 Hz, 2H), 2.16 (q, J =7.1 Hz, 2H), 1.72-1.63 (m, 2H), 1.06 (s, 9H). Step 2: tert-butyl 7-(5-((tert-butyldiphenylsilyl)oxy)pentyl)-3,4-dihydro-1,8-naphthyridine- 1(2H)-carboxylate
[0610] To a solution of tert-butyl(pent-4-en-1-yloxy)diphenylsilane (18.11 g, 55.82 mmol) in THF (60 mL) was added 9-BBN (0.5 M in THF, 163.73 mL), the reaction was stirred at 30 °C for 12 h. This resulting solution was added into a mixture of tert-butyl 7-chloro-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate (10 g, 37.21 mmol), PCy3 (1.04 g, 3.72 mmol, 1.21 mL), Pd(dba)
2 (2.14 g, 3.72 mmol) and K
2CO
3 (10.29 g, 74.42 mmol) in THF (300 mL) and water (15 mL), the reaction was stirred at 70 °C for 16 h. After the reaction, water (1000 mL) was added, extracted with ethyl acetate (800 mL x 2), the organic layer was combined and concentrated to give the crude product. The residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate = 100/1 to 3/1). tert-butyl 7-(5-((tert-butyldiphenylsilyl)oxy)pentyl)-3,4- dihydro-1,8-naphthyridine-1(2H)-carboxylate (160 g, 188.97 mmol, 84.6% yield, 66% purity) was obtained as brown oil. LCMS (ESI) m/z = 559.3 (M+1). Step 3: tert-butyl 7-(5-hydroxypentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0611] To a solution of tert-butyl 7-(5-((tert-butyldiphenylsilyl)oxy)pentyl)-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate (11 g, 19.68 mmol) in THF (400 mL) was added TBAF (1 M in THF, 78.74 mL), the mixture was stirred at 50 °C for 16 h. Water (500 mL) was added, the mixture was extracted with ethyl acetate (300 mL x 2). The combined organic layers were washed with (500 mL), dried over Na
2SO
4, concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (ISCO®; 330 g SepaFlash® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 300 mL/min). tert- butyl 7-(5-hydroxypentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (3.6 g, 56.18 mmol, 57.08% yield) was obtained as a yellow solid.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.46 (d, J = 7.6 Hz, 1H), 6.96 (d, J = 7.8 Hz, 1H), 3.77-3.70 (m, 2H), 3.54 (t, J = 6.6 Hz, 2H), 2.80-2.65 (m, 4H), 1.95-1.88 (m, 2H), 1.76-1.68 (m, 2H), 1.60-1.53 (m, 2H), 1.51 (s, 9H), 1.46-1.38 (m, 2H). Step 4: tert-butyl 7-(5-oxopentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0612] To a solution of oxalyl dichloride (11.88 g, 93.63 mmol, 8.20 mL) in DCM (80 mL) was added DMSO (7.32 g, 93.63 mmol, 7.32 mL) at -70 °C, after stirred at -70 °C for 10 min, a solution of compound 5 (10 g, 31.21 mmol) in DCM (20 mL) was added dropwise at -70 °C. The mixture was stirred at -70 °C for 1.5 h, then TEA (18.95 g, 187.25 mmol, 26.06 mL) was added and the resulting mixture stirred at -70 °C for 20 min. The reaction mixture was quenched by water (100 mL) at 0 °C, extracted with ethyl acetate (50 mL x 2). The combined organic layers were washed with brine (100 mL), dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO
2, Petroleum ether/Ethyl acetate = 100/1 to 0/100). tert-butyl 7-(5-oxopentyl)-3,4-dihydro-1,8-naphthyridine- 1(2H)-carboxylate (6.5 g, 20.41 mmol, 65.41% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 9.81-9.76 (m, 1H), 7.30 (br d, J = 7.5 Hz, 1H), 6.81 (br d, J = 7.6 Hz, 1H), 3.76 (br t, J = 5.9 Hz, 2H), 2.76-2.71 (m, 4H), 2.51-2.46 (m, 2H), 1.96-1.89 (m, 2H), 1.82-1.70 (m, 4H), 1.52 (s, 9H). LCMS (ESI) m/z = 319.2 (M+1). Preparation of (R)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)-N-(2,2,2- trifluoroethyl)pyrrolidin-3-amine Step 1: tert-butyl (R)-3-(2,2,2-trifluoroacetamido)pyrrolidine-1-carboxylate

[0613] To a solution of tert-butyl (R)-3-aminopyrrolidine-1-carboxylate (1 g, 5.37 mmol) in EtOH (10 mL) was added TEA (815.08 mg, 8.06 mmol, 1.12 mL) and TFAA (1.53 g, 10.74 mmol, 1.48 mL), the mixture was stirred at 50 °C for 12 h. The solvent was distilled under vacuum and the resulting residue was diluted with water (40 mL) and extracted with ethyl acetate (20 mL x 2). The combined organic layers were washed with brine (20 mL), dried over Na2SO4, filtered and concentrated to give crude product. The residue was purified by column chromatography (SiO
2, Petroleum ether/Ethyl acetate = 100/1 to 1/1). tert-butyl (R)-3-(2,2,2- trifluoroacetamido)pyrrolidine-1-carboxylate (0.5 g, 1.77 mmol, 33.0% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 227 (M-56+1). Step 2: tert-butyl (R)-3-((2,2,2-trifluoroethyl)amino)pyrrolidine-1-carboxylate
[0614] To a solution of tert-butyl (R)-3-(2,2,2-trifluoroacetamido)pyrrolidine-1-carboxylate (3.1 g, 10.98 mmol) in THF (15 mL) was added BH3 ^THF (1 M in THF, 32.95 mL) at 0 °C, the mixture was stirred at 70 °C for 12 h. The reaction mixture was quenched with MeOH (10 mL) at 0 °C and the mixture was concentrated to give crude product. The residue was purified by column chromatography (SiO
2, Petroleum ether/Ethyl acetate=100/1 to 3/1). tert-butyl (R)-3- ((2,2,2-trifluoroethyl)amino)pyrrolidine-1-carboxylate (1.6 g, 5.96 mmol, 54.3% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 3.63-3.34 (m, 4H), 3.32- 3.02 (m, 3H), 2.13-1.99 (m, 1H), 1.72 (br d, J = 5.8 Hz, 1H), 1.46 (s, 9H). LCMS (ESI) m/z = 213 (M-56+1). Step 3: tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(2,2,2- trifluoroethyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0615] To a solution of tert-butyl (R)-3-((2,2,2-trifluoroethyl)amino)pyrrolidine-1- carboxylate (842.55 mg, 3.14 mmol) and sodium triacetoxyborohydride (832.03 mg, 3.93 mmol) in DCM (10 mL) was added a solution of tert-butyl 7-(5-oxopentyl)-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate (0.5 g, 1.57 mmol) in DCM (3 mL), the reaction was stirred at 25 °C for 3 h. Water (5 mL) was added, adjusted to pH = 8 with aqueous sodium bicarbonate, the resulting mixture was extracted with DCM (10 mL x 2). The combined organic layers were washed with water (20 mL) and brine (20 mL x 2), dried over Na2SO4, concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO
2, Petroleum ether/Ethyl acetate = 10/1 to 5/1). tert-butyl (R)-7-(5-((1-(tert- butoxycarbonyl)pyrrolidin-3-yl)(2,2,2-trifluoroethyl)amino)pentyl)-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate (0.8 g, crude) was obtained as a yellow oil. LCMS (ESI) m/z = 571.3 (M+1).
Step 4: (R)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)-N-(2,2,2- trifluoroethyl)pyrrolidin-3-amine

[0616] To a solution of tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(2,2,2- trifluoroethyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (400 mg, 700.91 umol) in MeOH (3 mL) was added HCl/MeOH (4 M, 6 mL) and the mixture was stirred at 25 °C for 12 h. The solvent was removed under reduced pressure leaving (R)-N-(5-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)-N-(2,2,2-trifluoroethyl)pyrrolidin-3-amine (220 mg, crude) as a yellow oil which was used directly in the next step. LCMS (ESI) m/z = 371.2 (M+1). Preparation of (R)-N-methyl-N-(5-((S)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine Step 1: phenyl 2-allyl-1,8-naphthyridine-1(2H)-carboxylate

[0617] To a solution of 1,8-naphthyridine (15 g, 115.25 mmol) in acetonitrile (150 mL) was added phenyl chloroformate (27.07 g, 172.88 mmol, 21.65 mL) and silver trifluoromethanesulfonate (2.96 g, 11.53 mmol) at 20 °C. The mixture was stirred for 0.5 h at 20 °C. To the mixture was added allyltrimethylsilane (19.75 g, 172.88 mmol, 27.59 mL) at 0 °C and the mixture was stirred for 16 h at 20 °C. The mixture was diluted with MTBE (300 mL) and filtered. The filtrate was washed with aqueous NaHCO3 (200 mL) and brine (200 mL). The organic layers were dried over Na2SO4, filtered and the filtrate was concentrated to give product. The residue was purified by flash silica gel chromatography (ISCO ®; 330 g SepaFlash ® Silica Flash Column, Eluent of 0~60% Ethyl acetate/Petroleum ether gradient @ 200 mL/min). phenyl 2-allyl-1,8-naphthyridine-1(2H)-carboxylate (40 g, 136.83 mmol, 59.36% yield) was obtained as a yellow solid. LCMS (ESI) m/z = 293.2 (M+1). Step 2: phenyl (E)-2-(5-hydroxypent-2-en-1-yl)-1,8-naphthyridine-1(2H)-carboxylate

[0618] (1,3-Bis(2,4,6-trimethylphenyl)-2- imidazolidinylidene)dichloro(phenylmethylene)(tricyclohexylphosphine)ruthenium (1.45 g, 1.71 mmol) was added to a solution of phenyl 2-allyl-1,8-naphthyridine-1(2H)-carboxylate (10 g, 34.21 mmol) and but-3-en-1-ol (12.33 g, 171.04 mmol, 14.72 mL) in DCM (200 mL) at 25 °C. The mixture was stirred for 16 h at 40 °C and was concentrated to give a crude residue which was purified by flash silica gel chromatography (ISCO ®; 100 g SepaFlash ® Silica Flash Column, Eluent of 0~50% Ethyl acetate/Petroleum ether gradient @ 100 mL/min). Phenyl (E)-2-(5- hydroxypent-2-en-1-yl)-1,8-naphthyridine-1(2H)-carboxylate (10 g, 29.73 mmol, 86.90% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 337.2 (M+1). Step 3: Mixture of phenyl 2-(5-hydroxypentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate and methyl 2-(5-hydroxypentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate

[0619] To a suspension of Pd/C (3 g, 10 wt% loading) in MeOH (300 mL) was added phenyl (E)-2-(5-hydroxypent-2-en-1-yl)-1,8-naphthyridine-1(2H)-carboxylate (9.5 g, 28.24 mmol) under argon atmosphere. The suspension was degassed and purged with hydrogen three times. The mixture was stirred under hydrogen (40 Psi) at 50 °C for 16 h. The reaction mixture was filtered through a celite pad and the filtrate was concentrated in vacuum. A mixture of phenyl 2-(5- hydroxypentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate and methyl 2-(5- hydroxypentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (8 g of mixture, crude) was obtained as a yellow oil. LCMS (ESI) m/z = 341.2 & 279.2 (M+1). Step 4: methyl 2-(5-hydroxypentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0620] To a solution of a mixture of phenyl 2-(5-hydroxypentyl)-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate and methyl 2-(5-hydroxypentyl)-3,4-dihydro-1,8-naphthyridine- 1(2H)-carboxylate (8 g, 23.50 mmol) in MeOH (80 mL) was added sodium methoxide (2.54 g,
47.00 mmol) and the mixture was stirred at 25 °C for 16 h. The reaction was concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO ®; 80 g SepaFlash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 120 mL/min). methyl 2-(5-hydroxypentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (4.8 g, 17.24 mmol, 73.38% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 279.2 (M+1). Step 5: methyl 2-(5-bromopentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0621] To a solution of methyl 2-(5-hydroxypentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (2.3 g, 8.26 mmol) in THF (30 mL) was added triphenylphosphine (3.22 g, 12.28 mmol) and tetrabromomethane (4.12 g, 12.41 mmol) at 0 °C. The mixture was stirred at 25 °C for 16 h. The reaction was concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO ®; 40 g SepaFlash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 100 mL/min). methyl 2-(5-bromopentyl)-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate (4.9 g, crude) was obtained as a yellow oil. LCMS (ESI) m/z = 341.1 (M+1). Step 6: methyl 2-(5-(((R)-1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)pentyl)-3,4- dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0622] To a solution of methyl 2-(5-bromopentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (4.9 g, 8.33 mmol, 58% purity), tert-butyl (R)-3-(methylamino)pyrrolidine-1- carboxylate (1.7 g, 8.49 mmol) in toluene (40 mL) was added a solution of NaOH (3.33 g, 83.28 mmol) in water (40 mL) and tetrabutylammonium bromide (TBAB) (268.48 mg, 832.84 umol). The mixture was stirred at 100 °C for 16 h. The reaction was quenched by ice water (100 mL) slowly, extracted with ethyl acetate (50 mL x 2). The combined organic phase was washed with brine (100 mL), dried over anhydrous Na
2SO
4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO ®; 40 g SepaFlash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 100 mL/min). methyl 2-(5-(((R)-1- (tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-
1(2H)-carboxylate (2 g, 4.34 mmol, 24.8% yield over 2 steps) was obtained as a yellow oil. LCMS (ESI) m/z = 461.3 (M+1) Step 7: tert-butyl (3R)-3-(methyl(5-(1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidine-1-carboxylate

[0623] To a solution of methyl 2-(5-(((R)-1-(tert-butoxycarbonyl)pyrrolidin-3- yl)(methyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.9 g, 4.12 mmol) in dioxane (20 mL) was added aqueous NaOH (3 M, 20 mL) and the mixture was stirred at 80 °C for 16 h. The reaction was quenched by ice water (50 mL) slowly, extracted with ethyl acetate (20 mL x 2). The combined organic phase was washed with brine (50 mL), dried over anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by prep-HPLC (column: Phenomenex luna C18250 x 70 mm x 15 um; mobile phase: A water (TFA 0.1%), B acetonitrile, 8%-35%, 24 min; Flow Rate (25 mL/min). tert-butyl (3R)-3-(methyl(5-(1,2,3,4-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidine-1-carboxylate (2.1 g, 3.33 mmol, 80.73% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 403.3 (M+1). [0624] Racemic tert-butyl (3R)-3-(methyl(5-(1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidine-1-carboxylate (2.1 g, 5.22 mmol) was separated by SFC (column: Regis (S, S) Whelk-O1 (250 mm x 25 mm x 10 um); mobile phase: [0.1% NH3water IPA]; B%: 45%-45%, 5 min) to give two peaks: [0625] Peak 1, arbitrarily assigned as tert-butyl (R)-3-(methyl(5-((S)-1,2,3,4-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidine-1-carboxylate (800 mg, 1.99 mmol, 76.19% yield) was obtained as a yellow oil. SFC: R
t = 2.512 min, 98.18%.
1H NMR (400 MHz, METHANOL- d
4) δ = 7.67 (dd, J = 6.6, 15.0 Hz, 2H), 6.76 (t, J = 6.8 Hz, 1H), 3.99-3.78 (m, 2H), 3.63-3.56 (m, 2H), 3.49-3.38 (m, 2H), 3.19-3.09 (m, 2H), 2.94-2.78 (m, 5H), 2.39 (br s, 1H), 2.20-2.00 (m, 2H), 1.83-1.46 (m, 17H). [0626] Peak 2, arbitrarily assigned as tert-butyl (R)-3-(methyl(5-((R)-1,2,3,4-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidine-1-carboxylate (680 mg, 1.69 mmol, 64.76% yield) was obtained as a yellow oil. SFC: R
t =2.693 min, 98.52%.
1H NMR (400 MHz, METHANOL- d
4) δ = 7.70 (dd, J = 1.7, 5.2 Hz, 1H), 7.27 (d, J = 6.6 Hz, 1H), 6.52 (dd, J = 5.4, 7.1 Hz, 1H), 3.57-3.41 (m, 2H), 3.25 (br s, 1H), 3.19-3.09 (m, 2H), 2.79-2.72 (m, 2H), 2.57 (br d, J = 7.5 Hz,
2H), 2.37 (br s, 3H), 2.18 (br d, J = 6.3 Hz, 1H), 2.01-1.94 (m, 1H), 1.83 (br d, J = 10.8 Hz, 1H), 1.63-1.37 (m, 19H). Step 8: (R)-N-methyl-N-(5-((S)-1,2,3,4-tetrahydro-1,8-naphthyridin-2-yl)pentyl)pyrrolidin- 3-amine
[0627] A solution of tert-butyl (R)-3-(methyl(5-((S)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidine-1-carboxylate (800 mg, 1.99 mmol) in HCl/MeOH (4 M, 20 mL) was stirred at 50 °C for 16 h then was concentrated in vacuum. (R)-N-methyl-N-(5-((S)-1,2,3,4- tetrahydro-1,8-naphthyridin-2-yl)pentyl)pyrrolidin-3-amine (700 mg, crude, HCl) was obtained as a yellow oil. LCMS (ESI) m/z = 303.2 (M+1). Preparation of (R)-N-methyl-N-(5-((R)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine

[0628] A solution of tert-butyl (R)-3-(methyl(5-((R)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidine-1-carboxylate (680 mg, 1.69 mmol) in HCl/MeOH (4 M, 20 mL) was stirred at 50 °C for 16 h then was concentrated in vacuum. (R)-N-methyl-N-(5-((R)-1,2,3,4- tetrahydro-1,8-naphthyridin-2-yl)pentyl)pyrrolidin-3-amine (600 mg, crude, HCl) was obtained as a yellow oil. LCMS (ESI) m/z = 303.2 (M+1). Preparation of (R)-N-(2,2-difluoroethyl)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine Step 1: tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)amino)pentyl)-3,4- dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0629] To a solution of tert-butyl 7-(5-oxopentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (1 g, 3.14 mmol) and tert-butyl (R)-3-aminopyrrolidine-1-carboxylate (877.41 mg,
4.71 mmol, 797.65 uL) in DCM (20 mL) was added sodium triacetoxyborohydride (1.33 g, 6.28 mmol) at 0 °C. The resulting mixture was stirred at 25 °C for 16 h. The reaction was quenched by ice water (20 mL) slowly, adjusted to pH = 10, extracted with ethyl acetate (10 mL x 2). The combined organic phase was washed with brine (20 mL), dried over anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO ®; 10 g SepaFlash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether to 0~100% MeOH/Ethyl acetate gradient @ 80 mL/min). tert-butyl (R)-7-(5-((1-(tert- butoxycarbonyl)pyrrolidin-3-yl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.8 g, crude) was obtained as a yellow oil. LCMS (ESI) m/z = 489.4 (M+1). Step 2: tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(2,2- difluoroethyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0630] To a solution of tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3- yl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.25 g, 2.56 mmol) and 2,2- difluoroethyl trifluoromethanesulfonate (1.64 g, 7.67 mmol) in DMF (10 mL) was added TEA (1.04 g, 10.23 mmol, 1.42 mL). The mixture was stirred at 80 °C for 16 h. The reaction was quenched by ice water (20 mL) slowly, extracted with ethyl acetate (15 mL x 2). The combined organic phase was washed with brine (20 mL), dried over anhydrous Na
2SO
4, filtered and concentrated in vacuum. The residue was purified by prep-HPLC (column: Agela DuraShell C18 250 x 70 mm x 10 um; mobile phase: A water (TFA 0.1%), B acetonitrile 15%-45%, 20 min; Flow Rate 130 mL/min). tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(2,2- difluoroethyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1 g, 1.81 mmol, 70.7% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 553.4 (M+1). Step 3: (R)-N-(2,2-difluoroethyl)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine

[0631] A solution of tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(2,2- difluoroethyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (300 mg, 542.79 umol) in HCl/MeOH (4 M, 10 mL) was stirred at 50 °C for 3 h. The reaction was concentrated in vacuum. (R)-N-(2,2-difluoroethyl)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine (345 mg, crude) was obtained as a yellow oil. LCMS (ESI) m/z = 353.3 (M+1). Preparation of (R)-N-(3-methoxy-3-methylbutyl)-N-(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)pyrrolidin-3-amine Step 1: 3-methoxy-3-methylbutanal
[0632] To a solution of oxalyl chloride (32.22 g, 253.86 mmol, 22.22 mL) in DCM (100 mL) was added DMSO (19.84 g, 253.86 mmol, 19.84 mL) at -78
oC, the mixture was stirred at -78
oC for 0.5 h and then added dropwise a solution of 3-methoxy-3-methylbutan-1-ol (10 g, 84.62 mmol, 10.80 mL) in DCM (10 mL) at -78
oC. After stirred at -78
oC for 50 min, TEA (51.38 g, 507.73 mmol, 70.67 mL) was added and the resulting mixture was stirred for 0.5 h at -78
oC and 20 min at 25
oC. water (40 mL) was added, the mixture was extracted with DCM (30 mL x 3). The combined organic layers were washed with brine (30 mL), dried over Na2SO4, concentrated under reduced pressure to give a residue. 3-methoxy-3-methylbutanal (15.8 g, crude) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 9.82 (t, J = 3.00 Hz, 1H), 3.21-3.25 (m, 3H), 2.51 (d, J = 3.00 Hz, 2H), 1.29 (s, 6H). Step 2: tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(3-methoxy-3- methylbutyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate
[0633] To a solution of tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3- yl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (880.37 mg, 1.80 mmol) and sodium triacetoxyborohydride (1.15 g, 5.40 mmol) in DCM (14 mL) was added 3-methoxy-3-
methylbutanal (837.08 mg, 7.21 mmol) at 25
oC. The mixture was stirred at 25
oC for 16 h. The reaction mixture was quenched by adding water (20 mL), adjusted to pH = 8 with saturated aqueous NaHCO3, and the resulting mixture was extracted with ethyl acetate (20 mL x 3). The combined organic layers were washed with brine (20 mL), dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by MPLC (Biotage®; 20 g SepaFlash® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum to 0~30% MeOH/Ethyl acetate gradient @ 150 mL/min). tert-butyl (R)-7-(5-((1-(tert- butoxycarbonyl)pyrrolidin-3-yl)(3-methoxy-3-methylbutyl)amino)pentyl)-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate (0.42 g, 713.29 umol, 39.59% yield) was obtained as a yellow oil.
1H NMR (400 MHz, METHANOL-d4) δ = 7.47 (d, J = 7.6 Hz, 1H), 6.97 (d, J = 7.7 Hz, 1H), 3.77-3.71 (m, 2H), 3.63-3.45 (m, 3H), 3.21-3.16 (m, 4H), 3.13-3.04 (m, 1H), 2.80-2.52 (m, 9H), 2.11 (br d, J = 5.3 Hz, 1H), 1.95-1.88 (m, 3H), 1.77-1.64 (m, 5H), 1.51 (s, 8H), 1.46 (s, 12H), 1.42-1.31 (m, 2H), 1.17 (s, 4H). LCMS (ESI) m/z = 589.4 (M+1). Step 3: (R)-N-(3-methoxy-3-methylbutyl)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine
[0634] A solution of tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(3-methoxy- 3-methylbutyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (0.23 g, 390.61 umol) in formic acid (18.77 mg, 390.61 umol, 6 mL) was stirred at 25
oC for 12 h. The mixture was concentrated. (R)-N-(3-methoxy-3-methylbutyl)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin- 2-yl)pentyl)pyrrolidin-3-amine (0.275 g, crude) was obtained as brown oil. LCMS (ESI) m/z = 389.3 (M+1). Preparation of (R)-N-methyl-N-(6-((S)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)hexyl)pyrrolidin-3-amine Step 1: phenyl (E)-2-(6-hydroxyhex-2-en-1-yl)-1,8-naphthyridine-1(2H)-carboxylate

[0635] (1,3-Bis(2,4,6-trimethylphenyl)-2- imidazolidinylidene)dichloro(phenylmethylene)(tricyclohexylphosphine)ruthenium (1.45 g, 1.71 mmol) was added to a solution of phenyl 2-allyl-1,8-naphthyridine-1(2H)-carboxylate (10 g, 34.21 mmol) and pent-4-en-1-ol (14.73 g, 171.04 mmol) in DCM (50 mL) at 25 °C under nitrogen. The mixture was stirred for 16 h at 40 °C. The mixture was concentrated to give crude residue. The residue was purified by flash silica gel chromatography (ISCO ®; 40 g SepaFlash ® Silica Flash Column, Eluent of 0~100% THF/Petroleum ether gradient @ 100 mL/min). phenyl (E)-2-(6-hydroxyhex-2-en-1-yl)-1,8-naphthyridine-1(2H)-carboxylate (2 g, 5.71 mmol, 16.7% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 351.2 (M+1). Step 2: phenyl 2-(6-hydroxyhexyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0636] To a stirred mixture of Pd/C (3 g, 10 wt% loading) in MeOH (350 mL) was added phenyl (E)-2-(6-hydroxyhex-2-en-1-yl)-1,8-naphthyridine-1(2H)-carboxylate (12.5 g, 35.67 mmol) under argon atmosphere. The suspension was degassed and purged with hydrogen for 3 times. The mixture was stirred under hydrogen (50 psi) at 50 °C for 16 h. The reaction mixture was filtered through celite pad and rinsed by MeOH (100 mL). The filtrate was concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO ®; 120 g SepaFlash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 150 mL/min). phenyl 2-(6-hydroxyhexyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (8.2 g, 23.13 mmol, 64.9% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 355.2 (M+1). Step 3: phenyl 2-(6-bromohexyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0637] To a solution of phenyl 2-(6-hydroxyhexyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (8.2 g, 23.13 mmol) in DCM (85 mL) was added triphenylphosphine (9.04 g, 34.47 mmol) and tetrabromomethane (11.51 g, 34.70 mmol) at 0 °C. The resulting mixture was stirred at 25 °C for 16 h. The reaction was concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO ®; 120 g SepaFlash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 200 mL/min). phenyl 2-(6-bromohexyl)-3,4-dihydro-
1,8-naphthyridine-1(2H)-carboxylate (6.3 g, 15.10 mmol, 65.3% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 417.1, 419.1 (M+1) Step 4: phenyl 2-(6-(((R)-1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)hexyl)-3,4- dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0638] To a solution of phenyl 2-(6-bromohexyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (5.8 g, 13.90 mmol) and tert-butyl (R)-3-(methylamino)pyrrolidine-1-carboxylate (2.78 g, 13.90 mmol) in DMF (60 mL) was added DIEA (7.75 g, 58.74 mmol, 10.44 mL). The mixture was stirred at 80 °C for 16 h. The reaction was quenched by ice water (100 mL) slowly, extracted with ethyl acetate (50 mL x 2). The combined organic phase was washed with brine (50 mL), dried over anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO®; 120 g SepaFlash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 180 mL/min). phenyl 2-(6-(((R)-1- (tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)hexyl)-3,4-dihydro-1,8-naphthyridine- 1(2H)-carboxylate (6.3 g, 10.68 mmol, 76.9% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 537.3 (M+1). Step 5: tert-butyl (3R)-3-(methyl(6-(1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)hexyl)amino)pyrrolidine-1-carboxylate

[0639] To a solution of phenyl 2-(6-(((R)-1-(tert-butoxycarbonyl)pyrrolidin-3- yl)(methyl)amino)hexyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (3.8 g, 7.08 mmol) in THF (40 mL) was added t-BuOK (1.19 g, 10.62 mmol) at 0 °C. The resulting mixture was stirred at 25 °C for 16 h. The reaction was quenched by ice water (20 mL) slowly, extracted with ethyl acetate (10 mL x 2). The combined organic phase was washed with brine (20 mL), dried over anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by prep-HPLC (column: Welch Xtimate C18250 x 70 mm x 10 um; mobile phase: A water (NH
4HCO
310 mmol/L), B acetonitrile 45%-75%, 20 min; Flow Rate (25 mL/min). tert-butyl (3R)-3-(methyl(6-
(1,2,3,4-tetrahydro-1,8-naphthyridin-2-yl)hexyl)amino)pyrrolidine-1-carboxylate (1.3 g, 3.15 mmol, 44.5% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 417.3 (M+1) [0640] tert-butyl (3R)-3-(methyl(6-(1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)hexyl)amino)pyrrolidine-1-carboxylate (1.1 g, 2.64 mmol) was separated by SFC (column: Regis (S,S) Whelk-O1 (250 mm x 25 mm x 10 um); mobile phase: [0.1% NH3water IPA]; B%: 45%-45%, 4 min) to give two peaks: [0641] Peak 1, arbitrarily assigned as tert-butyl (R)-3-(methyl(6-((S)-1,2,3,4-tetrahydro-1,8- naphthyridin-2-yl)hexyl)amino)pyrrolidine-1-carboxylate (500 mg, 1.20 mmol, 45.5% yield) was obtained as a white solid. SFC: Rt = 2.514 min, 98.8%.
1H NMR (400 MHz, METHANOL-d4) δ = 7.73-7.65 (m, 1H), 7.26-7.18 (m, 1H), 6.49 (dd, J = 5.1, 7.1 Hz, 1H), 3.54-3.42 (m, 2H), 3.41- 3.36 (m, 1H), 3.28-3.20 (m, 1H), 3.11-2.96 (m, 2H), 2.76-2.71 (m, 2H), 2.50-2.39 (m, 2H), 2.26 (s, 3H), 2.12 (dq, J = 5.1, 6.2 Hz, 1H), 2.01-1.93 (m, 1H), 1.84-1.71 (m, 1H), 1.65-1.48 (m, 7H), 1.43-1.35 (m, 4H). [0642] Peak 2, arbitrarily assigned as tert-butyl (R)-3-(methyl(6-((R)-1,2,3,4-tetrahydro-1,8- naphthyridin-2-yl)hexyl)amino)pyrrolidine-1-carboxylate (520 mg, 1.25 mmol, 47.3% yield) was obtained as a white solid. SFC: Rt = 2.720 min, 98.84%.
1H NMR (400 MHz, METHANOL-d4) δ = 7.69 (dd, J = 1.6, 5.2 Hz, 1H), 7.26-7.18 (m, 1H), 6.49 (dd, J = 5.2, 7.2 Hz, 1H), 3.55-3.42 (m, 2H), 3.41-3.35 (m, 1H), 3.28-3.20 (m, 1H), 3.11-2.93 (m, 2H), 2.74 (dd, J = 6.0, 6.9 Hz, 2H), 2.50-2.39 (m, 2H), 2.26 (s, 3H), 2.17-2.07 (m, 1H), 2.02-1.91 (m, 1H), 1.82-1.71 (m, 1H), 1.63- 1.47 (m, 6H), 1.42-1.33 (m, 4H). Step 6: (R)-N-methyl-N-(6-((S)-1,2,3,4-tetrahydro-1,8-naphthyridin-2-yl)hexyl)pyrrolidin-3- amine

[0643] The solution of tert-butyl (R)-3-(methyl(6-((S)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)hexyl)amino)pyrrolidine-1-carboxylate (500 mg, 1.20 mmol) in HCl/MeOH (4 M, 9.62 mL) was stirred at 50 °C for 16 h. The reaction was concentrated in vacuum and used without further purification. (R)-N-methyl-N-(6-((S)-1,2,3,4-tetrahydro-1,8-naphthyridin-2-yl)hexyl)pyrrolidin- 3-amine (600 mg, crude, HCl salt) was obtained as a yellow oil. LCMS (ESI) m/z = 317.3 (M+1)
Preparation of (R)-N-methyl-N-(6-((R)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)hexyl)pyrrolidin-3-amine

[0644] A solution tert-butyl (R)-3-(methyl(6-((R)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)hexyl)amino)pyrrolidine-1-carboxylate (500 mg, 1.20 mmol) in HCl/MeOH (4 M, 9.62 mL) was stirred at 50 °C for 3 h. The reaction was concentrated in vacuum. (R)-N-methyl-N-(6-((R)- 1,2,3,4-tetrahydro-1,8-naphthyridin-2-yl)hexyl)pyrrolidin-3-amine (645 mg, crude, HCl salt) was obtained as a yellow oil. LCMS (ESI) m/z = 317.3 (M+1). Preparation of (R)-N-methyl-N-(4-((S)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)butyl)pyrrolidin-3-amine Step 1: (but-3-yn-1-yloxy)(tert-butyl)diphenylsilane

[0645] To a solution of but-3-yn-1-ol (48.3 g, 689.12 mmol, 52.16 mL) in DCM (1.45 L) was added imidazole (56.30 g, 826.94 mmol) and tert-butylchlorodiphenylsilane (189.41 g, 689.12 mmol, 177.02 mL) at 25 °C. The mixture was stirred at 25 °C for 2 h. The reaction mixture (three parallel reactions) was quenched by water (4.0 L) and extracted with ethyl acetate (1.0 L x 2). The combined organic layers were washed with brine (3.0 L x 1), dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO
2, petroleum ether/ethyl acetate = 1/0 to 100/1). (but-3-yn-1-yloxy)(tert- butyl)diphenylsilane (600 g, 1.94 mol, 94.1% yield) was obtained as colorless oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.70 (dd, J = 1.5, 7.8 Hz, 4H), 7.48-7.37 (m, 6H), 3.81 (t, J = 7.1 Hz, 2H), 2.47 (dt, J = 2.6, 7.1 Hz, 2H), 1.96 (t, J = 2.6 Hz, 1H), 1.08 (s, 9H). Step 2: ethyl 2-(4-((tert-butyldiphenylsilyl)oxy)but-1-yn-1-yl)-1,8-naphthyridine-1(2H)- carboxylate

[0646] To a solution of (but-3-yn-1-yloxy)(tert-butyl)diphenylsilane (231.11 g, 749.16 mmol) in THF (1.0 L) was added i-PrMgCl ^LiCl (1.3 M, 500.92 mL) at 20 °C and the solution was stirred at this temperature for 0.5 h to give a gray solution. Then a solution of ZnCl2 (109.96 g, 806.78 mmol) in THF (500 mL) was added dropwise to the former gray solution and the resulting mixture was stirred for 2 h. During this time, the solution becomes heterogeneous with the formation of a pale grey precipitate. Next, a solution of 1,8-naphthyridine (75 g, 576.27 mmol) in THF (600 mL) was added dropwise to the organozinc mixture over 0.035 h, and the reaction was stirred for a further 0.5 h at 20 °C. During this time, this mixture thickened with the formation of small clumps of pale-yellow matter. After 0.085 h, a pale-yellow suspension was formed that darkened over 0.5 h. Next was added ethyl chloroformate (81.30 g, 749.16 mmol, 71.32 mL) dropwise at 20 °C and the resulting pale yellow mixture was stirred for 18 h. The reaction mixture (three parallel reactions) was quenched by saturated NH
4Cl (3.5 L) and extracted with ethyl acetate (1.0 L x 2). The combined organic layers were washed with brine (2.0 L x 3), dried over Na
2SO
4, filtered and concentrated under reduced pressure to give a residue. The crude product was triturated with MTBE (1.0 L) at 25
oC for 12 h. ethyl 2-(4-((tert- butyldiphenylsilyl)oxy)but-1-yn-1-yl)-1,8-naphthyridine-1(2H)-carboxylate (240 g, 469.95 mmol, 81.6% yield) was obtained as a white solid.
1H NMR (400 MHz, CHLOROFORM-d) δ = 8.36 (dd, J = 2.0, 4.8 Hz, 1H), 7.62 (ddd, J = 1.8, 3.3, 6.4 Hz, 4H), 7.44-7.37 (m, 7H), 7.09-7.01 (m, 1H), 6.49-6.43 (m, 1H), 6.11-6.04 (m, 1H), 5.88-5.80 (m, 1H), 4.38-4.29 (m, 2H), 3.68-3.60 (m, 2H), 2.40-2.31 (m, 2H), 1.36-1.31 (m, 3H), 1.01-1.00 (m, 9H). Step 3: ethyl 2-(4-((tert-butyldiphenylsilyl)oxy)butyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate
[0647] To a suspension of Pd/C (7 g, 10 wt% loading) in EtOH (50 mL) was added a solution of ethyl 2-(4-((tert-butyldiphenylsilyl)oxy)but-1-yn-1-yl)-1,8-naphthyridine-1(2H)- carboxylate (25 g, 48.95 mmol) in THF (50 mL) and EtOH (450 mL) under argon at 20
oC. The suspension was degassed under vacuum and purged with argon for three times. The mixture was stirred under hydrogen (40 psi) at 50 °C for 10 h. The reaction mixture was filtered and concentrated under reduced pressure to give a residue. The residue was used for next step and without purificationethyl 2-(4-((tert-butyldiphenylsilyl)oxy)butyl)-3,4-dihydro-1,8-naphthyridine- 1(2H)-carboxylate (25 g, crude) was obtained as a yellow oil.
1H NMR (400 MHz,
CHLOROFORM-d) δ = 8.34 (dd, J = 1.5, 4.6 Hz, 1H), 7.67-7.61 (m, 4H), 7.43-7.35 (m, 7H), 7.00 (dd, J = 4.8, 7.4 Hz, 1H), 4.58 (br t, J = 5.8 Hz, 1H), 4.32-4.21 (m, 2H), 3.62 (dt, J = 3.0, 6.4 Hz, 2H), 2.77-2.67 (m, 2H), 2.23-2.12 (m, 1H), 1.73-1.68 (m, 2H), 1.59-1.53 (m, 3H), 1.46-1.42 (m, 1H), 1.47-1.40 (m, 1H), 1.32-1.27 (m, 3H), 1.03 (s, 9H). LCMS (ESI) m/z = 517.4 (M+1). Step 4: ethyl 2-(4-hydroxybutyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate
[0648] To a solution of ethyl 2-(4-((tert-butyldiphenylsilyl)oxy)butyl)-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate (50 g, 96.76 mmol) in THF (700 mL) was added TBAF (1 M, 101.60 mL) at 25 °C. The mixture was stirred at 25 °C for 12 h. The reaction mixture (three parallel reactions) was quenched by aqueous HCl (1.0 L, 1 M) and extracted with petroleum ether (500 mL). The aqueous phase was added aqueous NaHCO
3 and adjusted the pH of the reaction solution to 6-7 and extracted with ethyl acetate (500 mL x 3), the combined organic layers were dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue. The crude product was triturated with ethyl acetate at 25 °C for 0.5 h. ethyl 2-(4-hydroxybutyl)-3,4- dihydro-1,8-naphthyridine-1(2H)-carboxylate (63.75 g, 229.03 mmol, 78.9% yield) was obtained as a white solid. LCMS (ESI) m/z = 279.3 (M+1) Step 6: ethyl 2-(4-bromobutyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate
[0649] To a solution of ethyl (S)-2-(4-hydroxybutyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (10 g, 35.93 mmol) in THF (130 mL) was added triphenylphosphine (14.04 g, 53.53 mmol) and tetrabromomethane (17.87 g, 53.89 mmol). The mixture was stirred at 25 °C for 16 h. Water (100 mL) was added, the mixture was extracted with ethyl acetate (80 mL x 2). The combined organic layers were washed with water (40 mL) and brine (50 mL), dried over Na
2SO
4, concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate = 100/1 to 0/1). ethyl 2-(4- bromobutyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (9.8 g, 28.72 mmol, 79.9% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 8.36-8.32 (m, 1H), 7.43 (dd, J = 0.8, 7.4 Hz, 1H), 7.03-6.99 (m, 1H), 4.65-4.57 (m, 1H), 4.33-4.22 (m, 2H), 3.42-
3.31 (m, 2H), 2.76-2.70 (m, 2H), 2.27-2.16 (m, 1H), 1.92-1.80 (m, 2H), 1.75-1.67 (m, 1H), 1.61- 1.47 (m, 3H), 1.45-1.37 (m, 1H), 1.33-1.27 (m, 3H). Step 7: ethyl 2-(4-(((R)-1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)butyl)-3,4- dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0650] To a solution of ethyl 2-(4-bromobutyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (4 g, 11.72 mmol), tert-butyl (R)-3-(methylamino)pyrrolidine-1-carboxylate (2.35 g, 11.72 mmol) in DMF (40 mL) was added DIEA (6.41 g, 49.58 mmol, 8.64 mL). The mixture was stirred at 80 °C for 16 h. Water (80 mL) was added, the mixture was extracted with ethyl acetate (80 mL x 2). The combined organic layers were washed with water (40 mL x 2) and brine (40 mL x 2), dried over Na2SO4, concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate = 100/1 to 0/1). ethyl 2-(4-(((R)-1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)butyl)-3,4-dihydro- 1,8-naphthyridine-1(2H)-carboxylate (3.64 g, 7.90 mmol, 67.4% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 461.3 (M+1). Step 8: tert-butyl (3R)-3-(methyl(4-(1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)butyl)amino)pyrrolidine-1-carboxylate

[0651] To a solution of ethyl 2-(4-(((R)-1-(tert-butoxycarbonyl)pyrrolidin-3- yl)(methyl)amino)butyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (3.64 g, 7.90 mmol) in dioxane (20 mL) was added aqueous NaOH (6 M, 20 mL) and the mixture was stirred at 100 °C for 32 h. Water (5 mL) was added, the mixture was extracted with ethyl acetate (10 mL x 3). The combined organic layers were dried over Na2SO4, concentrated under reduced pressure to give a residue. tert-butyl (3R)-3-(methyl(4-(1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)butyl)amino)pyrrolidine-1-carboxylate (2.8 g, crude) was obtained as a yellow oil. LCMS (ESI) m/z = 389.3 (M+1). [0652] tert-butyl (3R)-3-(methyl(4-(1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)butyl)amino)pyrrolidine-1-carboxylate (2.8 g, 7.21 mmol) was separated by SFC (column:
Regis (S,S) Whelk-O1 (250 mm x 25 mm, 10 um); mobile phase: [0.1% NH
3water IPA]; B%: 40%-40%, 6 min). [0653] Peak 1, arbitrarily assigned as tert-butyl (R)-3-(methyl(4-((S)-1,2,3,4-tetrahydro-1,8- naphthyridin-2-yl)butyl)amino)pyrrolidine-1-carboxylate (1.1 g, 2.83 mmol, 78.6% yield) was obtained as a yellow oil. SFC: Rt = 2.438 min, 98.66%. [0654] Peak 2, arbitrarily assigned as tert-butyl (R)-3-(methyl(4-((R)-1,2,3,4-tetrahydro-1,8- naphthyridin-2-yl)butyl)amino)pyrrolidine-1-carboxylate (880 mg, 2.26 mmol, 62.9% yield) was obtained as a yellow oil. SFC: R
t = 2.640 min, 98.46%. Step 9: (R)-N-methyl-N-(4-((S)-1,2,3,4-tetrahydro-1,8-naphthyridin-2-yl)butyl)pyrrolidin-3- amine
[0655] To a solution of tert-butyl (R)-3-(methyl(4-((S)-1,2,3,4-tetrahydro-1,8-naphthyridin- 2-yl)butyl)amino)pyrrolidine-1-carboxylate (300 mg, 772.11 umol) in DCM (4 mL) was added TFA (2 mL), the mixture was stirred at 25 °C for 16 h. The mixture was concentrated to give crude residue. (R)-N-methyl-N-(4-((S)-1,2,3,4-tetrahydro-1,8-naphthyridin-2-yl)butyl)pyrrolidin- 3-amine (370 mg, crude) was obtained as brown oil. LCMS (ESI) m/z = 289.3 (M+1). Procedure for (R)-N-methyl-N-(4-((R)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)butyl)pyrrolidin-3-amine

[0656] To a solution of tert-butyl (R)-3-(methyl(4-((R)-1,2,3,4-tetrahydro-1,8-naphthyridin- 2-yl)butyl)amino)pyrrolidine-1-carboxylate (300 mg, 772.11 umol) in DCM (4 mL) was added TFA (2 mL), the mixture was stirred at 25 °C for 16 h. The mixture was concentrated to give crude residue. (R)-N-methyl-N-(4-((R)-1,2,3,4-tetrahydro-1,8-naphthyridin-2-yl)butyl)pyrrolidin- 3-amine (278 mg, crude) was obtained as brown oil. LCMS (ESI) m/z = 289.3 (M+1). Preparation of (R)-N-isopropyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine Step 1: tert-butyl (R)-3-(isopropylamino)pyrrolidine-1-carboxylate
[0657] To a solution of tert-butyl (R)-3-aminopyrrolidine-1-carboxylate (5 g, 26.85 mmol, 4.55 mL), acetone (14.75 g, 253.91 mmol, 18.67 mL) and acetic acid (2.52 g, 41.92 mmol, 2.40 mL) in MeOH (90 mL) was added sodium cyanoborohydride (3.37 g, 53.69 mmol) at 25
oC, the reaction was stirred at 25
oC for 16 h. The reaction mixture was quenched by water (50 mL), extracted with ethyl acetate (100 mL x 7). The combined organic layers were dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by MPLC (Biotage®; 120 g SepaFlash® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum and 0~27% MeOH/Ethyl acetate gradient @ 150 mL/min). tert-butyl (R)-3- (isopropylamino)pyrrolidine-1-carboxylate (5.4 g, 23.65 mmol, 88.10% yield) was obtained as a light yellow solid.
1H NMR (400 MHz, METHANOL-d4) δ = 3.75-3.60 (m, 2H), 3.47 (br s, 1H), 3.36-3.28 (m, 1H), 3.16 (dd, J = 5.9, 10.8 Hz, 2H), 2.22 (br s, 1H), 1.87 (br d, J = 4.8 Hz, 1H), 1.41 (s, 9H), 1.21-1.18 (m, 6H). Step 2: tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3- yl)(isopropyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate
[0658] To a solution of tert-butyl (R)-3-(isopropylamino)pyrrolidine-1-carboxylate (1.43 g, 6.28 mmol) and sodium triacetoxyborohydride (2.00 g, 9.42 mmol) in DCM (20 mL) was added a solution of tert-butyl 7-(5-oxopentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1 g, 3.14 mmol) in DCM (10 mL) dropwise at 25
oC. The mixture was stirred at 25
oC for 16 h. The reaction mixture was quenched by adding water (30 mL), adjusted to pH = 8 with saturated aqueous NaHCO
3 (10 mL), and extracted with ethyl acetate (50 mL x 2). The combined organic layers were washed with brine (30 mL), dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by MPLC (Biotage®; 20 g SepaFlash® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether and 0~29% MeOH/Ethyl acetate gradient @ 120 mL/min). tert-butyl (R)-7-(5-((1-(tert-
butoxycarbonyl)pyrrolidin-3-yl)(isopropyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (0.45 g, 847.87 umol, 27.00% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.30 (d, 1H), 6.80 (d, J = 7.6 Hz, 1H), 3.77-3.73 (m, 2H), 3.58- 3.48 (m, 1H), 3.47-3.39 (m, 1H), 3.35-3.14 (m, 2H), 3.07-2.92 (m, 2H), 2.75-2.67 (m, 4H), 2.38 (br t, J = 7.4 Hz, 2H), 1.92 (quin, J = 6.3 Hz, 3H), 1.80-1.75 (m, 1H), 1.68 (br s, 1H), 1.52 (s, 9H), 1.49-1.40 (m, 12H), 1.38-1.29 (m, 2H), 1.00 (br d, J = 5.4 Hz, 6H). Step 3: (R)-N-isopropyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)pyrrolidin-3- amine
[0659] A solution of tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3- yl)(isopropyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (0.4 g, 753.66 umol) in HCl/MeOH (4 M, 4 mL) was stirred at 25
oC for 12 h. The mixture was concentrated in vacuum. (R)-N-isopropyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)pyrrolidin-3- amine (0.4 g, crude, HCl salt) was obtained as a yellow oil.
1H NMR (400 MHz, METHANOL- d
4) δ = 7.59 (d, J = 7.3 Hz, 1H), 6.65 (d, J = 7.3 Hz, 1H), 4.40-4.29 (m, 1H), 3.91-3.60 (m, 4H), 3.51 (t, J = 5.6 Hz, 2H), 3.44-3.32 (m, 3H), 3.25-3.13 (m, 1H), 2.82 (br t, J = 6.1 Hz, 2H), 2.76 (br t, J = 7.6 Hz, 2H), 2.67-2.54 (m, 1H), 2.53-2.38 (m, 1H), 2.00-1.86 (m, 4H), 1.81 (td, J = 7.7 Hz, 14.9 Hz, 2H), 1.57-1.38 (m, 8H). LCMS (ESI) m/z = 331.4 (M+1). Preparation of (R)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)-N-(3,3,3- trifluoropropyl)pyrrolidin-3-amine Step 1: tert-butyl (R)-3-((3,3,3-trifluoropropyl)amino)pyrrolidine-1-carboxylate
[0660] To a solution of tert-butyl (R)-3-aminopyrrolidine-1-carboxylate (0.95 g, 5.10 mmol) and 1,1,1-trifluoro-3-iodopropane (1.71 g, 7.65 mmol, 897.14 uL) in DMF (10 mL) was added DIEA (1.98 g, 15.30 mmol, 2.67 mL). The mixture was stirred at 80 °C for 16 h. The reaction was quenched by ice water (15 mL) slowly, extracted with ethyl acetate (10 mL x 3). The
combined organic phase was washed with brine (15 mL), dried over anhydrous Na
2SO
4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO ®; 20 g SepaFlash ® Silica Flash Column, Eluent of 10~100% Ethyl acetate/Petroleum ether gradient @ 80 mL/min). tert-butyl (R)-3-((3,3,3-trifluoropropyl)amino)pyrrolidine-1-carboxylate (700 mg, 2.48 mmol, 48.61% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 227.1 (M+1). Step 2: tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(3,3,3- trifluoropropyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0661] To a solution of tert-butyl 7-(5-oxopentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate tert-butyl 7-(5-oxopentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (806.45 mg, 2.53 mmol) and tert-butyl (R)-3-((3,3,3-trifluoropropyl)amino)pyrrolidine-1-carboxylate (650 mg, 2.30 mmol) in DCM (10 mL) was added sodium triacetoxyborohydride (975.99 mg, 4.61 mmol) at 0 °C. The resulting mixture was stirred at 25 °C for 16 h. The reaction was quenched by ice water (20 mL) slowly, adjusted to pH = 10, extracted with ethyl acetate (10 mL x 2). The combined organic phase was washed with brine (15 mL), dried over anhydrous Na
2SO
4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO ®; 12 g SepaFlash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether to 0~100% MeOH/Ethyl acetate gradient @ 80 mL/min). tert-butyl (R)- 7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(3,3,3-trifluoropropyl)amino)pentyl)-3,4-dihydro- 1,8-naphthyridine-1(2H)-carboxylate (1.3 g, 2.22 mmol, 96.56% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 585.4 (M+1). Step 3: (R)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)-N-(3,3,3- trifluoropropyl)pyrrolidin-3-amine

[0662] A solution of tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(3,3,3- trifluoropropyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.3 g, 2.22 mmol) in HCl/MeOH (4 M, 20 mL) was stirred at 50 °C for 3 h. The reaction was concentrated in vacuum. (R)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)-N-(3,3,3- trifluoropropyl)pyrrolidin-3-amine (1.6 g, crude, HCl salt) was obtained as a yellow oil. LCMS (ESI) m/z = 385.3 (M+1). Preparation of (R)-N-methyl-N-(2-methyl-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)hexan-2-yl)pyrrolidin-3-amine Step 1: tert-butyl (R)-3-(methyl(2-methylbut-3-yn-2-yl)amino)pyrrolidine-1-carboxylate

[0663] To a stirred mixture of tert-butyl (R)-3-(methylamino)pyrrolidine-1-carboxylate (3 g, 14.98 mmol), 3-chloro-3-methylbut-1-yne (2.00 g, 19.47 mmol, 2.19 mL), TEA (1.97 g, 19.47 mmol, 2.71 mL) in THF (30 mL) was added CuI (285.28 mg, 1.50 mmol) under nitrogen and the mixture was stirred for 1 h at 25 °C under nitrogen. The mixture was quenched with water (50 mL), extracted with ethyl acetate (30 mL x 3), washed with brine (30 mL x 2), dried with anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO ®; 40 g SepaFlash ® Silica Flash Column, Eluent of 0~30% Ethyl acetate/Petroleum ether gradient @ 120 mL/min). tert-butyl (R)-3-(methyl(2-methylbut-3-yn-2- yl)amino)pyrrolidine-1-carboxylate (3.3 g, 12.39 mmol, 82.70% yield) was obtained as a yellow solid.
1H NMR (400 MHz, CHLOROFORM-d) δ = 3.80 - 3.94 (m, 1 H), 3.37 - 3.55 (m, 2H), 3.12 - 3.24 (m, 2 H), 2.28 - 2.35 (m, 3 H), 2.26 (s, 1 H), 1.95 (td, J = 7.6, 3.4 Hz, 2 H), 1.44 (s, 9 H), 1.42 ppm (s, 6 H). Step 2: tert-butyl 7-(bromoethynyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate
[0664] NBS (344.51 mg, 1.94 mmol) was added portion wise to a solution of tert-butyl 7- ethynyl-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (0.5 g, 1.94 mmol), DBU (294.67 mg, 1.94 mmol, 291.76 uL) in acetonitrile (5 mL) at 20 °C. The mixture was stirred for 1 h at 20 °C.
The reaction mixture was diluted with water (50 mL) and extracted with ethyl acetate (50 mL x 3). The combined organic layers were washed with brine (50 mL), dried over Na
2SO
4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (ISCO ®; 25 g SepaFlash ® Silica Flash Column, Eluent of 0~50% Ethyl acetate/Petroleum ether gradient @ 100 mL/min). tert-butyl 7-(bromoethynyl)-3,4- dihydro-1,8-naphthyridine-1(2H)-carboxylate (0.62 g, 1.84 mmol, 94.99% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.33 (d, J = 7.6 Hz, 1H), 7.09 (d, J = 7.6 Hz, 1H), 3.82 - 3.71 (m, 2H), 2.75 (t, J = 6.5 Hz, 2H), 1.93 (quin, J = 6.3 Hz, 2H), 1.54 (s, 9H). Step 3: tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)-5- methylhexa-1,3-diyn-1-yl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate
[0665] A mixture of tert-butyl 7-(bromoethynyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (0.65 g, 1.93 mmol), tert-butyl (R)-3-(methyl(2-methylbut-3-yn-2- yl)amino)pyrrolidine-1-carboxylate (564.81 mg, 2.12 mmol), CuI (36.71 mg, 192.76 umol), K
2CO
3 (532.82 mg, 3.86 mmol) in MeOH (10 mL) was stirred for 16 h at 25 °C under air. The reaction mixture was diluted with water (50 mL) and extracted with ethyl acetate (50 mL x 3). The combined organic layers were washed with brine (50 mL), dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (ISCO ®; 25 g SepaFlash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 100 mL/min). tert-butyl (R)-7-(5-((1-(tert- butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)-5-methylhexa-1,3-diyn-1-yl)-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate (0.62 g, 1.19 mmol, 61.54% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.32 (d, J = 7.6 Hz, 1H), 7.13 (br d, J = 6.6 Hz, 1H), 3.89 - 3.81 (m, 1H), 3.79 - 3.73 (m, 2H), 3.54 - 3.43 (m, 2H), 3.25 - 3.15 (m, 2H), 2.76 (t, J = 6.4 Hz, 2H), 2.35 (s, 3H), 2.02 - 1.90 (m, 4H), 1.54 (s, 9H), 1.50 - 1.44 (m, 15H) Step 4: tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)-5- methylhexyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0666] To a mixture of tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3- yl)(methyl)amino)-5-methylhexa-1,3-diyn-1-yl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (330 mg, 631.36 umol) in ethyl acetate (10 mL) was added Lindlar catalyst (165.00 mg, 39.95 umol, 5% purity) at 25 °C under argon. The mixture was stirred at 25 °C for 8 h under hydrogen (15 Psi). The mixture was filtered and concentrated in vacuum. The residue was purified by prep-HPLC (column: Waters Xbridge BEH C18100 x 30 mm x 10 um; mobile phase: A water (NH4HCO310 mmol/L), B acetonitrile, 50%-80%, 10 min; Flow Rate (25 ml/min). tert- butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)-5-methylhexyl)-3,4- dihydro-1,8-naphthyridine-1(2H)-carboxylate (130 mg, 244.94 umol, 38.8% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 531.4 (M+1) Step 5: (R)-N-methyl-N-(2-methyl-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2- yl)pyrrolidin-3-amine

[0667] A solution of tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3- yl)(methyl)amino)-5-methylhexyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (130 mg, 244.94 umol) in HCl/MeOH (4 M, 10 mL) was stirred at 50 °C for 3 h. The reaction was concentrated in vacuum. (R)-N-methyl-N-(2-methyl-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)hexan-2-yl)pyrrolidin-3-amine (130 mg, crude) was obtained as a yellow oil. LCMS (ESI) m/z = 331.3 (M+1). Preparation of (R)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)-N-(2- (trifluoromethoxy)ethyl)pyrrolidin-3-amine Step 1: tert-butyl (R)-3-((2-((tert-butyldimethylsilyl)oxy)ethyl)amino)pyrrolidine-1- carboxylate
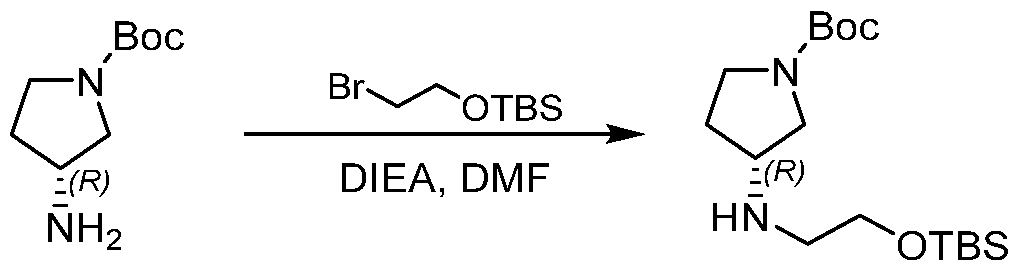
[0668] A mixture of (2-bromoethoxy)(tert-butyl)dimethylsilane (20 g, 83.60 mmol), tert- butyl (R)-3-aminopyrrolidine-1-carboxylate (10 g, 53.69 mmol, 9.09 mL), DIEA (13.88 g, 107.38 mmol, 18.70 mL) in DMF (100 mL) was stirred for 16 h at 50 °C. The reaction mixture was diluted with water (150 mL) and extracted with ethyl acetate (100 mL x 3). The combined organic layers were washed with brine (150 mL), dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (ISCO®; 80 g SepaFlash® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 200 mL/min). tert-butyl (R)-3-((2-((tert- butyldimethylsilyl)oxy)ethyl)amino)pyrrolidine-1-carboxylate (10 g, 29.02 mmol, 54.06% yield) was obtained as a yellow oil. Step 2: tert-butyl (R)-3-(((benzyloxy)carbonyl)(2-((tert- butyldimethylsilyl)oxy)ethyl)amino)pyrrolidine-1-carboxylate

[0669] To a solution of tert-butyl (R)-3-((2-((tert- butyldimethylsilyl)oxy)ethyl)amino)pyrrolidine-1-carboxylate (11 g, 31.92 mmol) in DCM (100 mL) was added TEA (6.46 g, 63.85 mmol, 8.89 mL) and benzyl chloroformate (7.08 g, 41.50 mmol, 5.90 mL) at 0 °C. The mixture was stirred at 20 °C for 16 h. The reaction was quenched by ice water (200 mL) slowly, extracted with DCM (100 mL x 2). The combined organic phase was washed with brine (200 mL), dried over anhydrous Na
2SO
4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO®; 120 g SepaFlash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 200 mL/min). tert-butyl (R)-3-(((benzyloxy)carbonyl)(2-((tert- butyldimethylsilyl)oxy)ethyl)amino)pyrrolidine-1-carboxylate (10 g, 20.89 mmol, 65.44% yield) was obtained as colorless oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.41-7.30 (m, 5H), 5.15 (s, 2H), 4.73-4.41 (m, 1H), 3.83-3.45 (m, 4H), 3.40-3.17 (m, 4H), 2.13-2.06 (m, 1H), 2.04 (br d, J = 7.6 Hz, 1H), 1.46 (s, 9H), 0.87 (s, 9H), 0.01 (br s, 6H). Step 3: tert-butyl (R)-3-(((benzyloxy)carbonyl)(2-hydroxyethyl)amino)pyrrolidine-1- carboxylate


[0670] To a solution of tert-butyl (R)-3-(((benzyloxy)carbonyl)(2-((tert- butyldimethylsilyl)oxy)ethyl)amino)pyrrolidine-1-carboxylate (8.8 g, 18.38 mmol) in THF (100 mL) was added TBAF (1 M in THF, 22.06 mL) and the mixture was stirred at 20 °C for 2 h. The reaction was quenched by ice water (100 mL) slowly, extracted with ethyl acetate (50 mL x 2). The combined organic phase was washed with brine (100 mL), dried over anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO ®; 120 g SepaFlash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 200 mL/min). tert-butyl (R)-3-(((benzyloxy)carbonyl)(2- hydroxyethyl)amino)pyrrolidine-1-carboxylate (4.77 g, 13.09 mmol, 67.42% yield) was obtained as colorless oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.43-7.29 (m, 5H), 5.16 (s, 2H), 4.64-4.54 (m, 1H), 3.74 (br t, J = 5.1 Hz, 2H), 3.66-3.36 (m, 4H), 3.32-3.15 (m, 2H), 2.11-2.02 (m, 2H), 1.46 (s, 9H). Step 4: tert-butyl (R)-3-(((benzyloxy)carbonyl)(2- (trifluoromethoxy)ethyl)amino)pyrrolidine-1-carboxylate
[0671] A solution of silver trifluoromethanesulfonate (2.12 g, 8.23 mmol), Selectfluor (1.46 g, 4.12 mmol), KF (637.66 mg, 10.98 mmol), tert-butyl (R)-3-(((benzyloxy)carbonyl)(2- hydroxyethyl)amino)pyrrolidine-1-carboxylate (1 g, 2.74 mmol) were added successively in a nitrogen-filled glovebox. Then ethyl acetate (14 mL), 2-fluoropyridine (532.83 mg, 5.49 mmol) and TMSCF
3 (1.17 g, 8.23 mmol) were added successively under nitrogen atmosphere. The mixture was stirred at 25 °C under nitrogen for 48 h. The reaction was filtered and concentrated in vacuum. The residue was purified by prep-HPLC (column: Phenomenex luna C18250 x 70 mm x 15 um; mobile phase: A water (TFA 0.1%), B acetonitrile 65%-85%, 27 min; Flow Rate 25 mL/min). tert-butyl (R)-3-(((benzyloxy)carbonyl)(2-(trifluoromethoxy)ethyl)amino)pyrrolidine- 1-carboxylate (600 mg, 1.39 mmol, 16.86% yield) was obtained as colorless oil.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.41-7.29 (m, 5H), 5.17 (s, 2H), 4.54-4.37 (m, 1H), 4.12 (br s, 2H),
3.68-3.44 (m, 4H), 3.30-3.21 (m, 2H), 2.27-1.96 (m, 2H), 1.45 (s, 9H). LCMS (ESI) m/z = 377.1 (M+1). Step 5: tert-butyl (R)-3-((2-(trifluoromethoxy)ethyl)amino)pyrrolidine-1-carboxylate

[0672] To a suspension of Pd/C (200 mg, 10 wt% loading) in MeOH (20 mL) was added tert- butyl (R)-3-(((benzyloxy)carbonyl)(2-(trifluoromethoxy)ethyl)amino)pyrrolidine-1-carboxylate (740 mg, 1.71 mmol) under nitrogen atmosphere. The suspension was degassed and purged with hydrogen for 3 times. The mixture was stirred under hydrogen (15 Psi) at 20 °C for 3 h. The reaction mixture was filtered through a Celite pad and the filtrate was concentrated in vacuum. tert-butyl (R)-3-((2-(trifluoromethoxy)ethyl)amino)pyrrolidine-1-carboxylate (560 mg, crude) was obtained as a yellow oil. LCMS (ESI) m/z = 299.2 (M+1). Step 6: tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(2- (trifluoromethoxy)ethyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0673] To a solution of tert-butyl 7-(5-oxopentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (578.50 mg, 1.94 mmol), sodium triacetoxyborohydride (865.31 mg, 4.08 mmol) in DCM (10 mL) was added tert-butyl (R)-3-((2-(trifluoromethoxy)ethyl)amino)pyrrolidine-1- carboxylate (650 mg, 2.04 mmol) at 0 °C and the mixture was stirred at 20 °C for 4 h. The reaction was quenched by ice water (30 mL) slowly, adjusted to pH = 8 by aqueous sodium hydroxide, extracted with DCM (20 mL x 2). The combined organic phase was washed with brine (40 mL), dried over anhydrous Na
2SO
4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO ®; 12 g SepaFlash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 80 mL/min). tert-butyl (R)-7-(5-((1- (tert-butoxycarbonyl)pyrrolidin-3-yl)(2-(trifluoromethoxy)ethyl)amino)pentyl)-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate (600 mg, 998.82 umol, 48.93% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.29 (d, J = 7.6 Hz, 1H), 6.80 (d, J = 7.6 Hz,
1H), 3.94 (br s, 2H), 3.80-3.71 (m, 2H), 3.65-3.43 (m, 3H), 3.34-3.21 (m, 2H), 3.03 (br d, J = 8.1 Hz, 1H), 2.81 (br s, 2H), 2.75-2.66 (m, 4H), 2.53 (br d, J = 6.5 Hz, 2H), 2.03-1.88 (m, 4H), 1.83- 1.67 (m, 4H), 1.52 (s, 9H), 1.46 (s, 9H), 1.35 (br d, J = 8.1 Hz, 2H). LCMS (ESI) m/z = 601.3 (M+1). Step 7: (R)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)-N-(2- (trifluoromethoxy)ethyl)pyrrolidin-3-amine

[0674] A solution of tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(2- (trifluoromethoxy)ethyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (550 mg, 915.58 umol) in HCl/MeOH (4 M, 15 mL) was stirred at 40 °C for 16 h. The reaction was concentrated in vacuum. (R)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)-N-(2- (trifluoromethoxy)ethyl)pyrrolidin-3-amine (500 mg, crude) was obtained as a yellow oil. LCMS (ESI) m/z = 401.3 (M+1). Preparation of (S)-N-(3,3-difluoropropyl)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine Step 1: 2-(3,3-difluoropropyl)isoindoline-1,3-dione

[0675] To a solution of 3-(1,3-dioxoisoindolin-2-yl)propanal (9 g, 44.29 mmol) in DCM (90 mL) was added diethylaminosulfur trifluoride (14.28 g, 88.59 mmol, 11.70 mL) at -78 °C, the mixture was stirred at 25 °C for 12 h. Water (50 mL) was added, the mixture was extracted with DCM (120 mL x 2). The combined organic layers were washed with water (50 mL x 2) and brine (50 mL x 2), dried over Na
2SO
4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate = 100/1 to 0/1). 2-(3,3-difluoropropyl)isoindoline-1,3-dione (5.6 g, 24.87 mmol, 18.71% yield) was obtained as a yellow solid.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.95-7.69 (m, 4H), 6.12-5.78 (m, 1H), 3.89 (t, J = 6.8 Hz, 2H), 2.33-2.20 (m, 2H).
Step 2: 3,3-difluoropropan-1-amine hydrochloride
[0676] To a solution of 2-(3,3-difluoropropyl)isoindoline-1,3-dione (5.7 g, 25.31 mmol) in EtOH (120 mL) was added hydrazine hydrate (3.26 g, 63.82 mmol, 3.17 mL, 98% purity), the mixture was stirred at 80 °C for 2 h. The reaction mixture was filtered and the filtrate was adjusted to pH = 6 with aqueous HCl (3M) at 0 °C. The solution was concentrated to remove EtOH and the resulting solution was lyophilized to give the crude product which was used for next step directly without purification. 3,3-difluoropropan-1-amine hydrochloride (3.6 g, crude, HCl salt) was obtained as a yellow solid.
1H NMR (400 MHz, DMSO-d
6) δ = 8.40 (br s, 2H), 6.39-6.11 (m, 1H), 2.95-2.85 (m, 2H), 2.30-2.15 (m, 2H). Step 3: tert-butyl 7-(5-((3,3-difluoropropyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine- 1(2H)-carboxylate

[0677] A solution of 3,3-difluoropropan-1-amine hydrochloride (1.5 g, 11.40 mmol, HCl) in THF (10 mL) was free-based by basic resin and filtered. Then sodium triacetoxyborohydride (7.25 g, 34.21 mmol) was added, a solution of tert-butyl 7-(5-oxopentyl)-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate (1.09 g, 3.42 mmol) in DCM (30 mL) was added at 0 °C, then the reaction was stirred at 25 °C for 2 h. The mixture was diluted with water (10 mL), adjusted to pH = 7 with saturated aqueous NaOH at 0 °C. The mixture was concentrated and then diluted with water (20 mL), extracted with ethyl acetate (10 mL x 2). The combined organic layers were washed with water (20 mL) and brine (20 mL), dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO
2, Petroleum ether/Ethyl acetate = 100/1 to 0/1). tert-butyl 7-(5-((3,3- difluoropropyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (730 mg, 1.84 mmol, 16.11% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 398.3 (M+1).
Step 4: tert-butyl 7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(3,3- difluoropropyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate
[0678] To a solution of tert-butyl 7-(5-((3,3-difluoropropyl)amino)pentyl)-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate (730 mg, 1.84 mmol) in DCM (25 mL) was added tert-butyl 3- oxopyrrolidine-1-carboxylate (1.82 g, 9.85 mmol) and sodium triacetoxyborohydride (1.17 g, 5.51 mmol). The mixture was stirred at 25 °C for 16 h. The reaction mixture was quenched by water (30 ml), and adjusted to pH = 9 by aqueous sodium hydroxide. The resulting mixture was extracted with ethyl acetate (15 mL x 2). The combined organic layers were washed with brine (30 mL), dried over Na
2SO
4, filtered and concentrated under reduced pressure to give a residue. The crude product was purified by prep-HPLC (column: Welch Xtimate C18250 x 70 mm x 10 um; mobile phase: A water (NH4HCO310 mmol/L), B acetonitrile 55%-85%, 20 min; Flow Rate (25 mL/min) to give tert-butyl 7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(3,3- difluoropropyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (480 mg, 846.98 umol, 46.12% yield) as a yellow oil which was separated by SFC (column: DAICEL CHIRALPAK IG (250 mm x 30 mm, 10 um); mobile phase: [0.1% NH3water MeOH]; B%: 25%- 25%, 7 min.) to give two peaks. [0679] Peak 1, arbitrarily assigned as tert-butyl (S)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin- 3-yl)(3,3-difluoropropyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (240 mg, 423.49 umol, 82.76% yield) obtained as a white solid. SFC: Rt = 4.641 min, 100.00%.
1H NMR (400 MHz, METHANOL-d4) δ = 7.47 (d, J = 7.8 Hz, 1H), 6.97 (d, J = 7.6 Hz, 1H), 5.93 (tt, J = 4.4, 56.9 Hz, 1H), 3.79-3.70 (m, 2H), 3.58-3.46 (m, 2H), 3.37-3.33 (m, 1H), 3.30-3.17 (m, 2H), 3.13-2.97 (m, 1H), 2.82-2.66 (m, 6H), 2.52 (br t, J = 7.4 Hz, 2H), 2.07-1.86 (m, 5H), 1.81- 1.66 (m, 3H), 1.51 (s, 9H), 1.45 (s, 9H), 1.38-1.29 (m, 2H). LCMS (ESI) m/z = 567 (M+1). [0680] Peak 2, arbitrarily assigned as tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin- 3-yl)(3,3-difluoropropyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (270 mg, 476.42 umol, 93.10% yield) obtained as a white solid. SFC: Rt =4.050 min, 100.00%.
1H NMR (400 MHz, METHANOL-d4) δ = 7.47 (d, J = 7.6 Hz, 1H), 6.97 (d, J = 7.6 Hz, 1H), 5.92 (tt, J = 4.5, 56.9 Hz, 1H), 3.80-3.72 (m, 2H), 3.55-3.44 (m, 2H), 3.35-3.35 (m, 1H), 3.30-3.17 (m,
2H), 3.10-2.98 (m, 1H), 2.82-2.68 (m, 6H), 2.56-2.46 (m, 2H), 2.11-1.79 (m, 6H), 1.78-1.65 (m, 3H), 1.51 (s, 9H), 1.45 (s, 9H), 1.38-1.29 (m, 2H). LCMS (ESI) m/z = 567 (M+1). Step 5: (S)-N-(3,3-difluoropropyl)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine

[0681] A solution of tert-butyl (S)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(3,3- difluoropropyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (240.00 mg, 423.49 umol) in HCl/MeOH (4 M, 5 mL) was stirred at 25 °C for 16 h. The mixture was concentrated to give product to give (S)-N-(3,3-difluoropropyl)-N-(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)pyrrolidin-3-amine (250 mg, crude) as a yellow oil. LCMS (ESI) m/z = 367.3 (M+1). Preparation of (R)-N-(3,3-difluoropropyl)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine

[0682] A solution of tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(3,3- difluoropropyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (270.00 mg, 476.42 umol) in HCl/MeOH (4 M, 10 mL) was stirred at 25 °C for 16 h. The mixture was concentrated to give (R)-N-(3,3-difluoropropyl)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine (150 mg, crude) as a yellow oil. LCMS (ESI) m/z = 367.3 (M+1). Preparation of (S)-N-((R)-1,1-difluoro-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2- yl)-N-methylpyrrolidin-3-amine Step 1: tert-butyl (S,E)-7-(5-((tert-butylsulfinyl)imino)pentyl)-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate

[0683] To a solution of tert-butyl 7-(5-oxopentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (2 g, 6.28 mmol), (S)-2-methylpropane-2-sulfinamide (913.55 mg, 7.54 mmol) in THF (40 mL) was added tetraisopropoxytitanium (3.57 g, 12.56 mmol, 3.71 mL) and the mixture was stirred at 80 °C for 16 h. The reaction was quenched by ice water (100 mL) slowly, extracted with ethyl acetate (40 mL x 2). The combined organic phase was washed with brine (100 mL), dried over anhydrous Na
2SO
4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO ®; 40 g SepaFlash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 150 mL/min). tert-butyl (S,E)-7-(5- ((tert-butylsulfinyl)imino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.62 g, 3.84 mmol, 61.2% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 8.07 (t, J = 4.7 Hz, 1H), 7.30 (br d, J = 7.9 Hz, 1H), 6.80 (br d, J = 7.5 Hz, 1H), 3.78 - 3.72 (m, 2H), 2.78 - 2.71 (m, 4H), 2.56 (dt, J = 4.9, 7.3 Hz, 2H), 1.97 - 1.90 (m, 2H), 1.87 - 1.80 (m, 2H), 1.76 - 1.69 (m, 2H), 1.52 (s, 9H), 1.20 (s, 9H). Step 2: tert-butyl 7-((R)-5-(((S)-tert-butylsulfinyl)amino)-6,6-difluoro-6- (phenylsulfonyl)hexyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0684] To a solution of tert-butyl (S,E)-7-(5-((tert-butylsulfinyl)imino)pentyl)-3,4-dihydro- 1,8-naphthyridine-1(2H)-carboxylate (1.62 g, 3.84 mmol), ((difluoromethyl)sulfonyl)benzene (723.99 mg, 3.77 mmol, 536.29 uL) in THF (40 mL) was added LiHMDS (1 M, 4.52 mL) dropwise at -78 °C under nitrogen and the mixture was stirred at -78 °C for 0.25 h. The reaction was quenched with brine (80 mL) slowly, extracted with ethyl acetate (40 mL x 2). The combined organic phase was washed with brine (100 mL), dried over anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO ®; 40 g SepaFlash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 80 mL/min). tert-butyl 7-((R)-5-(((S)-tert-butylsulfinyl)amino)-6,6-difluoro-6-
(phenylsulfonyl)hexyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.64 g, 2.67 mmol, 70.9% yield) was obtained as a yellow oil. SFC: R
t = 1.224 min, 94.28%.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.97 (d, J = 7.5 Hz, 2H), 7.81 - 7.76 (m, 1H), 7.67 - 7.62 (m, 2H), 7.30 (br d, J = 0.6 Hz, 1H), 6.85 (br d, J = 6.7 Hz, 1H), 3.77 (br t, J = 5.8 Hz, 2H), 3.56 - 3.47 (m, 1H), 2.82 - 2.70 (m, 4H), 2.29 - 2.13 (m, 1H), 1.99 - 1.69 (m, 7H), 1.54 (s, 9H), 1.27 (s, 9H). Step 3: tert-butyl 7-((R)-5-(((S)-tert-butylsulfinyl)(methyl)amino)-6,6-difluoro-6- (phenylsulfonyl)hexyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0685] To a solution of tert-butyl 7-((R)-5-(((S)-tert-butylsulfinyl)amino)-6,6-difluoro-6- (phenylsulfonyl)hexyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.64 g, 2.67 mmol) in THF (30 mL) was added NaH (160.30 mg, 4.01 mmol, 60% purity) at 0 °C and the mixture was stirred for 0.5 h. Then MeI (758.51 mg, 5.34 mmol, 332.68 uL) was added to the solution dropwise at 0 °C. The mixture was stirred at 20 °C for 15.5 h. The reaction was quenched by ice water (100 mL) slowly, extracted with ethyl acetate (50 mL x 2). The combined organic phase was washed with brine (100 mL), dried over anhydrous Na
2SO
4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO ®; 40 g SepaFlash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 100 mL/min). tert-butyl 7-((R)-5-(((S)-tert-butylsulfinyl)(methyl)amino)-6,6-difluoro-6- (phenylsulfonyl)hexyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.4 g, 2.23 mmol, 83.5% yield) was obtained as a yellow oil. SFC: R
t = 2.139 min, 93.36%.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.97 (d, J = 7.8 Hz, 2H), 7.81 - 7.74 (m, 1H), 7.67 - 7.60 (m, 2H), 7.30 (br d, J = 7.6 Hz, 1H), 6.85 (br d, J = 7.6 Hz, 1H), 4.40 - 4.23 (m, 1H), 3.76 (br dd, J = 4.1, 6.5 Hz, 2H), 2.81 - 2.71 (m, 4H), 2.59 (s, 3H), 2.02 - 1.69 (m, 8H), 1.53 (s, 9H), 1.26 - 1.21 (m, 9H). Step 4: tert-butyl 7-((R)-5-(((S)-tert-butylsulfinyl)(methyl)amino)-6,6-difluorohexyl)-3,4- dihydro-1,8-naphthyridine-1(2H)-carboxylate
[0686] To a solution of tert-butyl 7-((R)-5-(((S)-tert-butylsulfinyl)(methyl)amino)-6,6- difluoro-6-(phenylsulfonyl)hexyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.3 g, 2.07 mmol) in DMF (40 mL) was added 20 mL HOAc/NaOAc (1:1, 8 M) buffer solution, Mg (1.71 g, 70.36 mmol) in portions and the mixture was stirred at 20 °C for 48 h. The reaction was quenched by ice water (100 mL) slowly, extracted with ethyl acetate (40 mL x 2). The combined organic phase was washed with brine (100 mL), dried over anhydrous Na
2SO
4, filtered and concentrated in vacuum. tert-butyl 7-((R)-5-(((S)-tert-butylsulfinyl)(methyl)amino)-6,6- difluorohexyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.3 g, crude) was obtained as a yellow oil. LCMS (ESI) m/z = 488.3 (M+1). Step 5: (R)-1,1-difluoro-N-methyl-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2- amine
[0687] To a solution of tert-butyl 7-((R)-5-(((S)-tert-butylsulfinyl)(methyl)amino)-6,6- difluorohexyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.5 g, 3.08 mmol) in HCl/MeOH (4 M, 20 mL) and stirred at 20 °C for 16 h. The reaction was concentrated in vacuum. The residue was purified by prep-HPLC (column: Phenomenex luna C18250 x 50 mm x 10 um; mobile phase: A water (TFA 0.1%), B acetonitrile, 1%-30%, 10 min; Flow Rate (25 ml/min). (R)-1,1-difluoro-N-methyl-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2-amine (840 mg, 2.11 mmol, 68.72% yield, TFA) was obtained as a yellow oil. SFC: R
t = 1.325 min, 94.28%.
1H NMR (400 MHz, METHANOL-d4) δ = 7.58 (d, J = 7.4 Hz, 1H), 6.61 (d, J = 7.3 Hz, 1H), 6.45 - 6.16 (m, 1H), 3.72 - 3.62 (m, 1H), 3.52 - 3.48 (m, 2H), 2.84 - 2.80 (m, 5H), 2.77 - 2.71 (m, 2H), 1.95 (td, J = 6.0, 11.9 Hz, 2H), 1.91 - 1.82 (m, 2H), 1.77 (td, J = 7.6, 15.6 Hz, 2H), 1.61 - 1.53 (m, 2H). LCMS (ESI) m/z = 284.2 (M+1) Step 6: tert-butyl 3-(((R)-1,1-difluoro-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2- yl)(methyl)amino)pyrrolidine-1-carboxylate

[0688] To a solution of (R)-1,1-difluoro-N-methyl-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)hexan-2-amine (800 mg, 2.82 mmol), sodium triacetoxyborohydride (1.20 g, 5.65 mmol) in DCM (10 mL) was added a solution of tert-butyl 3-oxopyrrolidine-1-carboxylate (1.05 g, 5.65 mmol) in DCM (1 mL) dropwise at 0 °C and the mixture was stirred at 20 °C for 16 h. The reaction was quenched by ice water (30 mL) slowly, adjusted to pH = 8, extracted with DCM (20 mL x 2). The combined organic phase was washed with brine (80 mL), dried over anhydrous Na
2SO
4, filtered and concentrated in vacuum. tert-butyl 3-(((R)-1,1-difluoro-6-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)hexan-2-yl)(methyl)amino)pyrrolidine-1-carboxylate was separated by SFC (column: DAICEL CHIRALCEL OJ (250 mm x 50 mm x 10 um); mobile phase: [0.1% NH3water EtOH]; B%: 30%-30%, 5 min). LCMS (ESI) m/z = 453.3 (M+1). [0689] Peak 1, arbitrarily assigned as tert-butyl (S)-3-(((R)-1,1-difluoro-6-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)hexan-2-yl)(methyl)amino)pyrrolidine-1-carboxylate (190 mg, 419.82 umol, 29.74% yield) was obtained as a yellow oil. SFC: R
t = 1.020 min, 100%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.15 (br d, J = 7.3 Hz, 1H), 6.38 (d, J = 7.4 Hz, 1H), 6.03 - 5.68 (m, 1H), 3.50 - 3.36 (m, 5H), 3.28 - 3.18 (m, 1H), 3.02 (dt, J = 4.4, 9.1 Hz, 1H), 2.88 - 2.75 (m, 1H), 2.70 (t, J = 6.2 Hz, 2H), 2.54 (dt, J = 2.5, 7.6 Hz, 2H), 2.33 (s, 3H), 2.09 - 1.98 (m, 1H), 1.93 - 1.84 (m, 2H), 1.81 - 1.54 (m, 5H), 1.50 (br s, 1H), 1.46 (s, 9H), 1.42 - 1.34 (m, 1H). [0690] Peak 2, arbitrarily assigned as tert-butyl (R)-3-(((R)-1,1-difluoro-6-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)hexan-2-yl)(methyl)amino)pyrrolidine-1-carboxylate (240 mg, 530.29 umol, 37.57% yield) was obtained as a yellow oil. SFC: R
t = 1.109 min, 98.42%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.11 (d, J = 7.3 Hz, 1H), 6.36 (d, J = 7.3 Hz, 1H), 6.02 - 5.70 (m, 1H), 3.54 (dd, J = 7.6, 9.7 Hz, 1H), 3.50 - 3.43 (m, 1H), 3.41 - 3.36 (m, 3H), 3.27 - 3.18 (m, 1H), 2.98 - 2.82 (m, 2H), 2.69 (t, J = 6.3 Hz, 2H), 2.59 - 2.47 (m, 2H), 2.31 (s, 3H), 2.04 (br dd, J = 5.1, 7.6 Hz, 1H), 1.92 - 1.83 (m, 2H), 1.76 - 1.51 (m, 5H), 1.46 (s, 11H). Step 7: (S)-N-((R)-1,1-difluoro-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2-yl)-N- methylpyrrolidin-3-amine

[0691] A solution of tert-butyl (S)-3-(((R)-1,1-difluoro-6-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)hexan-2-yl)(methyl)amino)pyrrolidine-1-carboxylate (170 mg, 375.62 umol) in HCl/MeOH (4 M, 10 mL) was stirred at 20 °C for 16 h. The reaction was concentrated in vacuum to afford (S)-N-((R)-1,1-difluoro-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2- yl)-N-methylpyrrolidin-3-amine (230 mg, crude, HCl) as a yellow oil. LCMS (ESI) m/z = 353.3 (M+1). Preparation of (R)-N-((R)-1,1-difluoro-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2- yl)-N-methylpyrrolidin-3-amine

[0692] A solution of tert-butyl (R)-3-(((R)-1,1-difluoro-6-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)hexan-2-yl)(methyl)amino)pyrrolidine-1-carboxylate (210 mg, 464.01 umol) in HCl/MeOH (4 M, 10 mL) was stirred at 20 °C for 16 h. The reaction was concentrated in vacuum. (R)-N-((R)-1,1-difluoro-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2-yl)-N- methylpyrrolidin-3-amine (230 mg, crude, HCl) was obtained as a yellow oil. LCMS (ESI) m/z = 353.3 (M+1). Preparation of (2S,3S)-N,2-dimethyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine Step 1: rel-tert-butyl 7-(5-(((2R,3R)-1-(tert-butoxycarbonyl)-2-methylpyrrolidin-3- yl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0693] To a mixture of sodium triacetoxyborohydride (1.59 g, 7.49 mmol) and rel-1,1- dimethylethyl (2R,3R)-3-amino-2-methyl-1-pyrrolidinecarboxylate (0.75, 3.74 mmol) in DCM (20 mL) was added a solution of tert-butyl 7-(5-oxopentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (1.19 g, 3.74 mmol) in DCM (10 mL) dropwise at 0 °C, and then the mixture was
stirred at 20 °C for 2 h. The mixture was added ice-water (10 mL) and stirred for 5 min. Then the mixture was adjusted to pH = 7~8 by aqueous NaOH (2 M). The reaction mixture was partitioned between water (15 mL) and DCM (20 mL). The organic phase was separated, washed with DCM (15 mL x 3). The combined organic phase was washed with brine (15 mL), dried with anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (Biotage®; 20 g SepaFlash® Silica Flash Column, Eluent of 0~50% Ethyl acetate/MeOH @ 150 mL/min). rel-tert-butyl 7-(5-(((2R,3R)-1-(tert-butoxycarbonyl)-2- methylpyrrolidin-3-yl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.24 g, 2.47 mmol, 65.87% yield) was obtained as a yellow oil. LCMS: (ESI) m/z = 503.4 (M+1). Step 2: tert-butyl 7-(5-(((2S,3S)-1-(tert-butoxycarbonyl)-2-methylpyrrolidin-3- yl)(methyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate and tert-butyl 7-(5-(((2R,3R)-1-(tert-butoxycarbonyl)-2-methylpyrrolidin-3-yl)(methyl)amino)pentyl)-3,4- dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0694] A mixture of rel-tert-butyl 7-(5-(((2R,3R)-1-(tert-butoxycarbonyl)-2- methylpyrrolidin-3-yl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.24 g, 2.47 mmol), aqueous formaldehyde (886.91 mg, 10.93 mmol, 813.68 uL, 37% purity) and sodium triacetoxyborohydride (1.57 g, 7.40 mmol) in DCM (20 mL) was stirred at 20 °C for 1 h. The mixture was diluted with iced water (20 mL), adjusted to pH = 7 with saturated NaOH aqueous at 0
oC. The aqueous phase was extracted with DCM (20 mL x 3). The combined organic phase was washed with brine (20 mL), dried with anhydrous Na
2SO
4, filtered and concentrated in vacuum. rel-tert-butyl 7-(5-(((2R,3R)-1-(tert-butoxycarbonyl)-2- methylpyrrolidin-3-yl)(methyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.2 g, 2.32 mmol, 94.15% yield) was obtained as a yellow oil and was separated by SFC (column: DAICEL CHIRALPAK IG (250 mm x 50 mm x 10 um); mobile phase: [Heptane- EtOH, 1:1]; B%: 18% - 18%, 5 min). LCMS: (ESI) m/z = 517.4 (M+1). [0695] Peak 1, arbitrarily assigned as tert-butyl 7-(5-(((2S,3S)-1-(tert-butoxycarbonyl)-2- methylpyrrolidin-3-yl)(methyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (0.65 g, crude) was obtained as a yellow oil. SFC: R
t = 3.727 min, 91.10%.
1H NMR (400 MHz, METHANOL-d4) δ = 7.37 (d, J = 7.8 Hz, 1H), 6.86 (d, J = 7.6 Hz, 1H), 3.87 (td, J = 6.3, 12.4 Hz, 1H), 3.69 - 3.58 (m, 2H), 3.36 - 3.25 (m, 1H), 3.19 - 3.06 (m, 1H), 2.76 - 2.48 (m, 5H), 2.41 - 2.27 (m, 1H), 2.27 - 2.16 (m, 1H), 2.11 (s, 3H), 2.01 - 1.88 (m, 1H), 1.81 (quin, J = 6.3 Hz, 2H),
1.72 - 1.59 (m, 3H), 1.41 (s, 11H), 1.36 (d, J = 4.5 Hz, 9H), 1.27 (br dd, J = 5.3, 8.8 Hz, 2H), 0.94 (dd, J = 3.8, 6.3 Hz, 3H). [0696] Peak 2, arbitrarily assigned as tert-butyl 7-(5-(((2R,3R)-1-(tert-butoxycarbonyl)-2- methylpyrrolidin-3-yl)(methyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (0.4 g, 766.30 umol, 65.99% yield, 98.99% purity) was obtained as a yellow oil. SFC: Rt =4.420 min, 100.00%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.47 (d, J = 7.6 Hz, 1H), 6.96 (d, J = 7.8 Hz, 1H), 3.97 (td, J = 6.3, 12.5 Hz, 1H), 3.79 - 3.68 (m, 2H), 3.46 - 3.34 (m, 1H), 3.30 - 3.17 (m, 1H), 2.82 - 2.61 (m, 5H), 2.51 - 2.37 (m, 1H), 2.36 - 2.25 (m, 1H), 2.21 (s, 3H), 2.09 - 2.00 (m, 1H), 1.97 - 1.86 (m, 2H), 1.81 - 1.63 (m, 3H), 1.63 - 1.50 (m, 11H), 1.46 (d, J = 4.6 Hz, 9H), 1.43 - 1.29 (m, 2H), 1.04 (dd, J = 3.9, 6.4 Hz, 3H). Step 3: (2S,3S)-N,2-dimethyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine

[0697] To a solution of tert-butyl 7-(5-(((2S,3S)-1-(tert-butoxycarbonyl)-2-methylpyrrolidin- 3-yl)(methyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (600 mg, 1.16 mmol) in HCl/MeOH (4 M, 10 mL) and the mixture was stirred at 30 °C for 24 h. The reaction was concentrated in vacuum. (2S,3S)-N,2-dimethyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine (450 mg, crude, 2 HCl) was obtained as a yellow oil. LCMS: (ESI) m/z = 317.3 (M+1). Preparation of (2R,3R)-N,2-dimethyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine

[0698] A solution of tert-butyl 7-(5-(((2R,3R)-1-(tert-butoxycarbonyl)-2-methylpyrrolidin-3- yl)(methyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (400 mg, 774.12 umol) in HCl/MeOH (4 M, 10 mL) was stirred at 30 °C for 12 h. The reaction was concentrated in vacuum. (2R,3R)-N,2-dimethyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine (450 mg, crude, HCl) was obtained as a yellow oil. LCMS: (ESI) m/z = 317.3 (M+1).
Preparation of (R)-N-ethyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine Step 1: tert-butyl (R)-3-(pent-4-en-1-ylamino)pyrrolidine-1-carboxylate

[0699] A mixture of tert-butyl (R)-3-aminopyrrolidine-1-carboxylate (1 g, 5.4 mmol), 5- bromopent-1-ene (794 mg, 5.4 mmol) and DIEA (1.39 g, 10.8 mmol) in acetonitrile (16 mL) was refluxed for 16 h. The mixture was concentrated and purified by silica gel chromatography (pet ether: ethyl acetate = 0%~50%) to give the desired product tert-butyl (R)-3-(pent-4-en-1- ylamino)pyrrolidine-1-carboxylate as orange oil (700 mg). Yield 51% (ESI 255.1 (M+H)
+). Step 2: tert-butyl (R)-3-(ethyl(pent-4-en-1-yl)amino)pyrrolidine-1-carboxylate
[0700] A solution of tert-butyl (R)-3-(pent-4-en-1-ylamino)pyrrolidine-1-carboxylate (350 mg, 1.4 mmol), iodoethane (437 mg, 2.8 mmol) and Cs
2CO
3 (910 mg, 2.8 mmol) in 10 mL of DMF was stirred at 60 oC for 16 h. The mixture was cooled to room temperature, quenched with water and extracted with ethyl acetate (20 mL X 3). The combined organic solvents were concentrated and purified by silica gel chromatography (pet ether: ethyl acetate = 0%~20%) to give the desired product tert-butyl (R)-3-(ethyl(pent-4-en-1-yl)amino)pyrrolidine-1-carboxylate as orange oil (300 mg). Yield 77% (ESI 283.1 (M+H) +). Step 3: tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(ethyl)amino)pentyl)- 3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate
[0701] A solution of tert-butyl (R)-3-(ethyl(pent-4-en-1-yl)amino)pyrrolidine-1-carboxylate (1.6 g, 5.7 mmol) was added to 9-BBN (22.8 mL, 11.4 mmol, 0.5 N in THF) under nitrogen atmosphere. The mixture was stirred at 60
oC for 2 h and cooled to room temperature. tert-butyl 7-chloro-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.76 g, 5 mmol), Pd(OAc)2 (128 mg, 0.57 mmol), PCy3 (203 mg, 0.57 mmol) and KOH (280 mg, 5 mmol) were added. The mixture was stirred at 70 oC for 3 h, concentrated and purified by silica gel chromatography (MeOH:
ethyl acetate = 0%~25%) to give the desired product tert-butyl (R)-7-(5-((1-(tert- butoxycarbonyl)pyrrolidin-3-yl)(ethyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate as orange oil (2.2 g). Yield 85% (ESI 517.1 (M+H) +). Step 4: (R)-N-ethyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)pyrrolidin-3- amine

[0702] TFA (2 mL) was added to a soluion of tert-butyl (R)-7-(5-((1-(tert- butoxycarbonyl)pyrrolidin-3-yl)(ethyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (2.2 g, 4.3 mmol) in 8 mL of DCM. The mixture was stirred at room temperature for 4 h and concentrated. The residue was neutralized with solid Na2CO3 and purified by silica gel chromatography (MeOH: DCM = 0%~20%) to give the desired product (R)-N-ethyl-N-(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)pyrrolidin-3-amine as orange oil (1.1 g). Yield 82% (ESI 317.1 (M+H)
+). Preparation of (R)-N-(5-(4-methoxy-5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)-N- methylpyrrolidin-3-amine Step 1: tert-butyl (R)-3-(methyl(pent-4-en-1-yl)amino)pyrrolidine-1-carboxylate
[0703] A mixture of tert-butyl (R)-3-(methylamino)pyrrolidine-1-carboxylate (1 g, 5 mmol), 5-bromopent-1-ene (894 mg, 6 mmol) and DIEA (1.29 g, 10 mmol) in acetonitrile (16 mL) was refluxed for 16 h. The mixture was concentrated and purified by silica gel chromatography (pet ether: ethyl acetate = 0%~20%) to give the desired product tert-butyl (R)-3-(methyl(pent-4-en-1- yl)amino)pyrrolidine-1-carboxylate as orange oil (1.2 g). Yield 91% (ESI 269.3 (M+H) +). Step 2: tert-butyl (R)-3-((5-(4-methoxy-1,8-naphthyridin-2- yl)pentyl)(methyl)amino)pyrrolidine-1-carboxylate

[0704] A solution of tert-butyl (R)-3-(methyl(pent-4-en-1-yl)amino)pyrrolidine-1- carboxylate (1.22 g, 4.56 mmol) was added to 9-BBN (18.2 mL, 9.1 mmol, 0.5 N in THF) under nitrogen atmosphere. The mixture was stirred at 60 °C for 2 h and cooled to room temperature.2- chloro-4-methoxy-1,8-naphthyridine (740 mg, 3.8 mmol), Pd(OAc)2 (85 mg, 0.38 mmol), PCy3 (213 mg, 0.76 mmol) and KOH (255 mg, 4.56 mmol) were added. The mixture was stirred at 70 °C for 3 h, concentrated and purified by silica gel chromatography (MeOH: ethyl acetate = 0%~25%) to give the desired product tert-butyl (R)-3-((5-(4-methoxy-1,8-naphthyridin-2- yl)pentyl)(methyl)amino)pyrrolidine-1-carboxylate as orange oil (1.25 g). Yield 76% (ESI 429.1 (M+H) +). Step 3: tert-butyl (R)-3-((5-(4-methoxy-5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)(methyl)amino)pyrrolidine-1-carboxylate

[0705] A mixture of tert-butyl (R)-3-((5-(4-methoxy-1,8-naphthyridin-2- yl)pentyl)(methyl)amino)pyrrolidine-1-carboxylate (1.25 g, 2.9 mmol) and Pd/C (250 mg, 10 wt% loading) in 25 mL of MeOH was stirred at 55 °C under hydrogen atmosphere for 24 h. The mixture was filtered, concentrated and purified by silica gel chromatography (MeOH: DCM = 0%~25%) to give the desired product tert-butyl (R)-3-((5-(4-methoxy-5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)(methyl)amino)pyrrolidine-1-carboxylate as orange oil (1.08 g). Yield 76% (ESI 433.2 (M+H) +). Step 4: (R)-N-(5-(4-methoxy-5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)-N- methylpyrrolidin-3-amine

[0706] TFA (2 mL) was added to a soluion of tert-butyl (R)-3-((5-(4-methoxy-5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)(methyl)amino)pyrrolidine-1-carboxylate (1.08 g, 2.5 mmol) in 8 mL of DCM. The mixture was stirred at room temperature for 4 h and concentrated. The residue was neutralized with solid Na2CO3 and purified by silica gel chromatography (MeOH: DCM = 0%~20%) to give the desired product (R)-N-(5-(4-methoxy-5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)-N-methylpyrrolidin-3-amine as orange oil (765 mg). Yield 92% (ESI 333.1 (M+H)
+). Preparation of (R)-N-methyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine Step 1: tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)pentyl)- 3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0707] To a solution of tert-butyl (R)-3-(methyl(pent-4-en-1-yl)amino)pyrrolidine-1- carboxylate (1.1 g, 4.1 mmol) in THF (dry, 5 mL) under argon, was added 9-BBN (0.5 M in THF, 16.4 mL, 8.2 mmol). The reaction mixture was stirred at 50°C for 2 h, then cooled to ambient temperature, added 2-chloro-4-methoxy-1,8-naphthyridine (1.32 g, 4.9 mmol), Pd(OAc)2 (46 mg, 0.205 mmol), PCy3 (115 mg, 0.41 mmol) and KOH (681 mg, 12.3 mmol). The reaction mixture was stirred at 80°C for 15 h. Solvent was removed in vacuo, and the residue was purified by silica gel column (DCM:MeOH 30:1) to give the desired product tert-butyl (R)-7-(5-((1-(tert- butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate as a pale yellow oil (1.38 g, 67% yield ). MS (ESI +, m/z): 503[M+H]+. Step 2: (R)-N-methyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)pyrrolidin-3- amine

[0708] A mixture of tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3- yl)(methyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.38 g, 2.7 mmol) and TFA (20 mL) in DCM (20 mL) was stirred at RT for 16 h. The mixture was concentrated in vacuo to give the desired product (R)-N-methyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine as a yellow oil (1.34 g, 92% yield). MS (ESI +, m/z): 302 [M+H]
+.
Preparation of (R)-N-(2-methoxyethyl)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine Step 1: tert-butyl (R)-3-((2-methoxyethyl)(pent-4-en-1-yl)amino)pyrrolidine-1-carboxylate
[0709] A solution of tert-butyl (R)-3-(pent-4-en-1-ylamino)pyrrolidine-1-carboxylate (2.4 g, 9.4 mmol), 1-bromo-2-methoxyethane (2.6 g, 18.8 mmol) and Cs2CO3 (4.2 g, 18.8 mmol) in 50 mL of DMF was stirred at 100 °C for 16 h. The mixture was cooled to room temperature, quenched with water and extracted with ethyl acetate (20 mL x 3). The combined organic solvents were concentrated and purified by silica gel chromatography (pet ether: ethyl acetate = 0%~20%) to give the desired product tert-butyl (R)-3-((2-methoxyethyl)(pent-4-en-1- yl)amino)pyrrolidine-1-carboxylate as orange oil (2.7 g). Yield 91% (ESI 313.1 (M+H)
+). Step 2: tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(2- methoxyethyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0710] A solution of tert-butyl (R)-3-((2-methoxyethyl)(pent-4-en-1-yl)amino)pyrrolidine-1- carboxylate (1.5 g, 4.8 mmol) was added to 9-BBN (19.2 mL, 9.6 mmol, 0.5 N in THF) under nitrogen atmosphere. The mixture was stirred at 60°C for 2 h and cooled to room temperature, then tert-butyl 7-chloro-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.28 g, 4.8 mmol), Pd(OAc)2 (108 mg, 0.48 mmol), PCy3 (134 mg, 0.48 mmol) and KOH (269 mg, 4.8 mmol) were added. The mixture was stirred at 70°C for 3 h, concentrated and purified by silica gel chromatography (MeOH: DCM = 0%~25%) to give the desired product tert-butyl (R)-7-(5-((1- (tert-butoxycarbonyl)pyrrolidin-3-yl)(2-methoxyethyl)amino)pentyl)-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate as orange oil (2.2 g). Yield 83% (ESI 547.1 (M+H) +). Step 3: (R)-N-(2-methoxyethyl)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine

[0711] TFA (2 mL) was added to a soluion of tert-butyl (R)-7-(5-((1-(tert- butoxycarbonyl)pyrrolidin-3-yl)(2-methoxyethyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine- 1(2H)-carboxylate (2.2 g, 4.0 mmol) in 8 mL of DCM. The mixture was stirred at room temperature for 4 h and concentrated. The residue was neutralized with solid Na2CO3 and purified by silica gel chromatography (MeOH: DCM = 0%~20%) to give the desired product (R)- N-(2-methoxyethyl)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)pyrrolidin-3-amine as a pale yellow oil (1.26 g). Yield 90% (ESI 347.1 (M+H)
+). Preparation of (R)-N-cyclopropyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine Step 1: tert-butyl (R)-3-(cyclopropyl(pent-4-en-1-yl)amino)pyrrolidine-1-carboxylate
[0712] To a solution of tert-butyl (R)-3-(pent-4-en-1-ylamino)pyrrolidine-1-carboxylate (1.8 g, 7 mmol), (1-ethoxycyclopropoxy)trimethylsilane (1700 mg, 10 mmol) in MeOH (40 mL) was added sodium cyanoborohydride (640 mg, 10 mmol) and acetic acid (0.9 mL). The mixture was stirred at reflux for 10 h. The volatiles were removed under reduced pressure. The residue was treated with water (20 mL) and extracted with ethyl acetate (3 x 30 mL). The combined extracts were washed with brine (3 x 10 mL), dried over anhydrous Na
2SO
4, filtered, and concentrated and purified by silica gel chromatography (pet ether: ethyl acetate = 0%~20%) to give the desired product tert-butyl (R)-3-(cyclopropyl(pent-4-en-1-yl)amino)pyrrolidine-1-carboxylate as orange oil (1.4 g). Yield 58% (ESI 175.1 (M+H) +). Step 2: tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3- yl)(cyclopropyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0713] A solution of ert-butyl (R)-3-(cyclopropyl(pent-4-en-1-yl)amino)pyrrolidine-1- carboxylate (1.46 g, 5 mmol) was added to 9-BBN (20 mL, 10 mmol, 0.5 N in THF) under nitrogen atmosphere. The mixture was stirred at 60 °C for 2 h and cooled to room temperature. tert-butyl 7-chloro-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1.76 g, 5 mmol), Pd(OAc)2 (128 mg, 0.57 mmol), PCy3 (203 mg, 0.57 mmol) and KOH (280 mg, 5 mmol) were added. The mixture was stirred at 70 °C for 3 h, concentrated and purified by silica gel chromatography (MeOH: ethyl acetate = 0%~25%) to give the desired product tert-butyl (R)-7-(5-((1-(tert- butoxycarbonyl)pyrrolidin-3-yl)(cyclopropyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine- 1(2H)-carboxylate as orange oil (0.9 g). Yield 34% (ESI 529.1 (M+H) +). Step 3: (R)-N-cyclopropyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)pyrrolidin- 3-amine
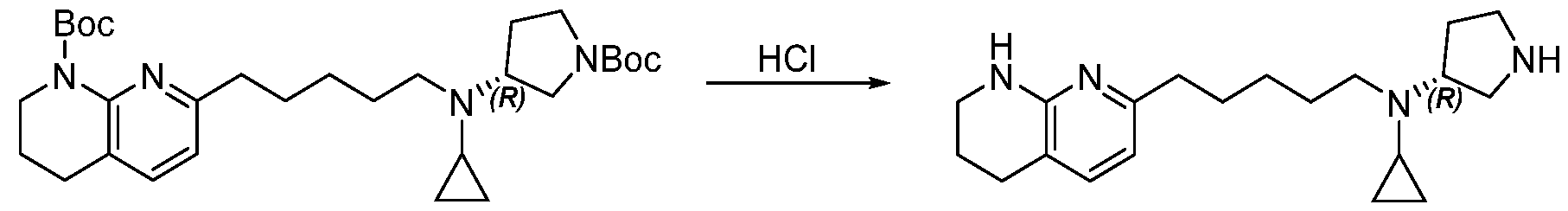
[0714] HCl/dioxane (3 mL, 4N in dioxane) was added to a soluion of tert-butyl (R)-7-(5-((1- (tert-butoxycarbonyl)pyrrolidin-3-yl)(cyclopropyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine- 1(2H)-carboxylate (0.9 g, 1.7 mmol) in 6 mL of MeOH. The mixture was stirred at room temperature for 4 h and concentrated to give the desired product (R)-N-cyclopropyl-N-(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)pyrrolidin-3-amine as white solid (0.6 g). Yield 100% (ESI 329.1 (M+H)
+). Preparation of (R)-N-methyl-N-(2-(2-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)ethoxy)ethyl)pyrrolidin-3-amine Step 1: ethyl 2-(1,8-naphthyridin-2-yl)acetate
[0715] To a solution of 2-methyl-1,8-naphthyridine (5.0 g, 34.7 mmol) in THF (50 mL) at 0°C, was added a solution of LiHMDS in THF (1M, 40 mL, 40 mmol) dropwise. The mixture was stirred at 0°C for 30 min, then added a solution of diethyl carbonate (4.7 g, 40 mmol) in THF (10 mL) dropwise. The reaction was stirred at ambient temperature overnight, quenched with water and extracted with ethyl acetate (50 mL x 3). The combined organic solvents were concentrated and purified by silica gel chromatography (pet ether: ethyl acetate = 0%~50%) to
give the desired product ethyl 2-(1,8-naphthyridin-2-yl)acetate as a pale yellow solid (5.7 g, 26.4 mmol). Yield 76% (ESI 217 (M+H)
+). Step 2: tert-butyl 7-(2-ethoxy-2-oxoethyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate
[0716] A mixture of ethyl 2-(1,8-naphthyridin-2-yl)acetate as a pale yellow solid (5.7 g, 26.4 mmol) and Pd/C (570 mg, 10 wt% loading) in MeOH (50 mL) was stirred at room temperature under hydrogen overnight. The mixture was filtered and the filtrate was concentrated in vacuo. The residue was dissolved in dioxane (50 mL), then added Boc2O (17.3 g, 79.2 mmol). The reaction mixture was stirred at 80 ℃ for 16 hrs. The mixture was concentrated and the residue was purified by silica gel column chromatography (pet ether:ethyl acetate 1:1) to give tert-butyl 7-(2-ethoxy-2-oxoethyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate as a light yellow oil (4.1 g, 12.8 mmol).Yield 48% (ESI 321 (M+H) +). Step 3: tert-butyl 7-(2-hydroxyethyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0717] To a solution of tert-butyl 7-(2-ethoxy-2-oxoethyl)-3,4-dihydro-1,8-naphthyridine- 1(2H)-carboxylate (1.6 g, 4.99 mmol) in THF (20 mL) and EtOH (4 mL) at 0 ℃, was added a solution of lithium borohydride in THF (1M, 10 mL, 10 mmol) dropwise. The reaction was stirred at ambient temperature for 15 h. The solution was quenched with water (20 mL), and extracted with ethyl acetate (50 mL x 3). The combined organic solvents were concentrated to give the desired product tert-butyl 7-(2-hydroxyethyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate as a pale yellow oil (1.3 g, 4.67 mmol).Yield 94% (ESI 279 (M+H) +). Step 4: tert-butyl 7-(2-(allyloxy)ethyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0718] To a solution of tert-butyl 7-(2-hydroxyethyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (1.3 g, 4.67 mmol) in n-heptane (20 mL) was added sodium hydroxide 50% solution in water (10 mL, 62.4 mmol), tetrabutylammonium bromide (74 mg, 0.23 mmol) and 3-
bromoprop-1-ene (1.13 g, 9.34 mmol). The mixture was heated at 80 °C for 2 h. Then the reaction mixture was cooled to ambient temperature, ethyl acetate (100 mL) and water (100 mL) were added. The organic phase was separated and the aqueous phase was extracted with ethyl acetate (50 mL x2). The combined organic layer was dried over Na2SO4, filtered. The solvent was removed in vacuo and the residue was purified by silica gel column (pet ether: ethyl acetate 2:1) to give the desired product tert-butyl 7-(2-(allyloxy)ethyl)-3,4-dihydro-1,8-naphthyridine- 1(2H)-carboxylate as a colorless oil (890 mg, 2.80 mmol). Yield 60% (ESI 319 (M+H) +). Step 5: tert-butyl 7-(2-(2-oxoethoxy)ethyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate
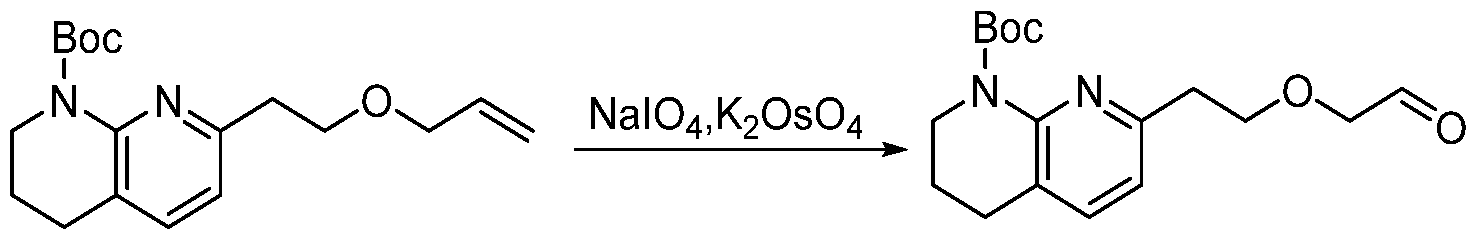
[0719] To a solution of tert-butyl 7-(2-(allyloxy)ethyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (890 mg, 2.80 mmol) in THF (30 mL) and water (10 mL) was added sodium periodate (1.5 g, 7.0 mmol) and potassium osmate (11 mg, 0.03 mmol). The reaction was stirred at ambient temperature for 2 h, diluted with water (10 mL) and ethyl acetate (20 mL). The organic phase was separated and the aqueous phase was extracted with ethyl acetate (20 mL x2). The combined organic layer was dried over Na2SO4, filtered and concentrated in vacuo to give the desired product tert-butyl 7-(2-(2-oxoethoxy)ethyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate as a colorless oil (780 mg, 2.44 mmol). Yield 87% (ESI 321 (M+H) +). Step 6: tert-butyl (R)-7-(2-(2-((1-(tert-butoxycarbonyl)pyrrolidin-3- yl)(methyl)amino)ethoxy)ethyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0720] Sodium cyanoborohydride (461 mg, 7.32 mmol) was added to a stirred mixture of tert-butyl 7-(2-(2-oxoethoxy)ethyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (780 mg, 2.44 mmol) and tert-butyl (R)-3-(methylamino)pyrrolidine-1-carboxylate (488 mg, 2.44 mmol) in MeOH (20 mL) at room temperature. The resulting mixture was stirred at room temperature for 15 hrs, then diluted with water (20 mL) and extracted with DCM (3 × 20 mL). The combined organic solvents were concentrated and purified by silica gel chromatography (pet ether: ethyl acetate = 0%~50%) to give the desired product tert-butyl (R)-7-(2-(2-((1-(tert-
butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)ethoxy)ethyl)-3,4-dihydro-1,8-naphthyridine- 1(2H)-carboxylate as a pale yellow oil (145 mg, 0.29 mmol). Yield 12% (ESI 505 (M+H) +). Step 7: (R)-N-methyl-N-(2-(2-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)ethoxy)ethyl)pyrrolidin-3-amine

[0721] HCl/dioxane (3 mL, 4N in dioxane) was added to a soluion of tert-butyl (R)-7-(2-(2- ((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)ethoxy)ethyl)-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate (210 mg, 0.42 mmol) in 6 mL of MeOH. The mixture was stirred at room temperature for 4 h and concentrated to give the desired product (R)-N-methyl-N- (2-(2-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)ethoxy)ethyl)pyrrolidin-3-amine as a white solid (125 mg, 0.41 mmol). Yield 100% (ESI 305 (M+H) +). Preparation of (R)-N-(2-cyclopropoxyethyl)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine Step 1: tert-butyl (R)-3-((2-cyclopropoxyethyl)amino)pyrrolidine-1-carboxylate
[0722] A solution of tert-butyl (R)-3-aminopyrrolidine-1-carboxylate (1.96 g, 10.6 mmol), 2- cyclopropoxyethyl 4-methylbenzenesulfonate (900 mg, 3.52 mmol) and K
2CO
3 (1.5 g, 10.6 mmol) in 15 mL of DMF was stirred at 80 °C for 16 h. The mixture was cooled to room temperature, quenched with water and extracted with ethyl acetate (50 mL x 3). The combined organic solvents were concentrated and purified by silica gel chromatography (pet ether: ethyl acetate = 0%~50%) to give the desired product tert-butyl (R)-3-((2- cyclopropoxyethyl)amino)pyrrolidine-1-carboxylate as orange oil (900 mg). Yield 89% (ESI 271.1 (M+H)
+). Step 2: tert-butyl (R)-3-((2-cyclopropoxyethyl)(pent-4-en-1-yl)amino)pyrrolidine-1- carboxylate
[0723] A mixture of tert-butyl (R)-3-((2-cyclopropoxyethyl)amino)pyrrolidine-1-carboxylate (900 mg, 3.34 mmol), 5-bromopent-1-ene (2.47 g, 16.7 mmol) and DIEA (2.15 g, 16.7 mmol) in acetonitrile (12 mL) was refluxed for 48 h. The mixture was cooled to roomt temperature, quenched with water and extracted with ethyl acetate (100 mL x 2). The combined organic solvents were concentrated and purified by silica gel chromatography (pet ether: ethyl acetate = 0%~30%) to give the desired product tert-butyl (R)-3-((2-cyclopropoxyethyl)(pent-4-en-1- yl)amino)pyrrolidine-1-carboxylate as orange oil (750 mg). Yield 67% (ESI 339.1 (M+H) +). Step 3: tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(2- cyclopropoxyethyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0724] A solution of tert-butyl (R)-3-((2-cyclopropoxyethyl)(pent-4-en-1- yl)amino)pyrrolidine-1-carboxylate (760 mg, 2.25 mmol) was added to 9-BBN (9 mL, 4.5 mmol, 0.5 N in THF) under nitrogen atmosphere. The mixture was stirred at 50 °C for 2 h and cooled to room temperature. tert-butyl 7-chloro-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (603 g, 2.25 mmol), Pd(OAc)
2 (31 mg, 0.26 mmol), PCy
3 (95 mg, 0.15 mmol) and KOH (190 mg, 3.4 mmol) were added. The mixture was stirred at 70°C for 16 h, concentrated and purified by silica gel chromatography (MeOH: ethyl acetate = 0%~25%) to give the desired product tert-butyl (R)- 7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(2-cyclopropoxyethyl)amino)pentyl)-3,4-dihydro- 1,8-naphthyridine-1(2H)-carboxylate as orange oil (560 mg). Yield 39% (ESI 573.1 (M+H) +). Step 4: (R)-N-(2-cyclopropoxyethyl)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine
[0725] TFA (3 mL) was added to a soluion of tert-butyl (R)-7-(5-((1-(tert- butoxycarbonyl)pyrrolidin-3-yl)(2-cyclopropoxyethyl)amino)pentyl)-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate (370 mg, 0.65 mmol) in 3 mL of DCM. The mixture was stirred at room temperature for 3 h and concentrated to give the desired product (R)-N-(2- cyclopropoxyethyl)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)pyrrolidin-3-amine as orange oil (310 mg). Yield 100% (ESI 373.1 (M+H) +). Preparation of (R)-N-isobutyl-N-(2-(2-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)ethoxy)ethyl)pyrrolidin-3-amine dihydrochloride Step 1: tert-butyl (R)-7-(2-(2-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)amino)ethoxy)ethyl)- 3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0726] To a solution of tert-butyl 7-(2-(2-oxoethoxy)ethyl)-3,4-dihydro-1,8-naphthyridine- 1(2H)-carboxylate (1.95 g, 6.09 mmol) and tert-butyl (R)-3-aminopyrrolidine-1-carboxylate (1.25 g, 6.70 mmol) in DCM (40 ml), was added sodium triacetoxyborohydride (2.58 g, 12.18 mmol). The reaction was stirred at ambient temperature for 15 h under nitrogen. The reaction was quenched with saturated aqueous NaHCO3 and extracted with DCM (3 x 20 mL). The combined organic layer was dried over Na2SO4, filtered, concentrated in vacuo, and the residue was purified by silica gel column (DCM: MeOH 20 : 1) to give the desired product tert-butyl (R)-7-(2-(2-((1- (tert-butoxycarbonyl)pyrrolidin-3-yl)amino)ethoxy)ethyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (1.31 g, 2.67 mmol) as a light yellow oil. Yield 44% (ESI 491 [M+H] +). Step 2: tert-butyl (R)-7-(2-(2-((1-(tert-butoxycarbonyl)pyrrolidin-3- yl)(isobutyl)amino)ethoxy)ethyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0727] To a solution of tert-butyl (R)-7-(2-(2-((1-(tert-butoxycarbonyl)pyrrolidin-3- yl)amino)ethoxy)ethyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (600 mg, 1.22 mmol) and isobutyraldehyde (176 mg, 2.44 mmol) in DCM (20 ml), was added sodium triacetoxyborohydride (517 mg, 2.44 mmol). The reaction was stirred at ambient temperature for 15 h under nitrogen. The reaction was quenched with saturated aqueous NaHCO
3 and extracted with DCM (3 x 20 mL). The combined organic layer was dried over Na2SO4, filtered, concentrated in vacuo, and the residue was purified by silica gel column (DCM: MeOH 20 : 1) to give the desired product tert-butyl (R)-7-(2-(2-((1-(tert-butoxycarbonyl)pyrrolidin-3- yl)(isobutyl)amino)ethoxy)ethyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (520 mg, 0.95 mmol) as a light yellow oil. Yield 78% (ESI 547 [M+H] +). Step 3: (R)-N-isobutyl-N-(2-(2-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)ethoxy)ethyl)pyrrolidin-3-amine dihydrochloride

[0728] Tert-butyl (R)-7-(2-(2-((1-(tert-butoxycarbonyl)pyrrolidin-3- yl)(isobutyl)amino)ethoxy)ethyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (630 mg, 1.15 mmol) was treated with HCl in 1,4-dioxane (4M, 20 mL) at ambient temperature overnight. Solvent was removed in vacuo to afford the desired product (R)-N-isobutyl-N-(2-(2-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)ethoxy)ethyl)pyrrolidin-3-amine dihydrochloride (434 mg, 1.03 mmol) as a yellow oil. Yield 90% (ESI 347 (M+H) +). Preparation of (3R)-N-methyl-N-(6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2- yl)pyrrolidin-3-amine dihydrochloride Step 1: tert-butyl (3R)-3-(hex-5-en-2-yl(methyl)amino)pyrrolidine-1-carboxylate

[0729] To a solution of hex-5-en-2-one (539 mg, 5.5 mmol) and tert-butyl (R)-3- (methylamino)pyrrolidine-1-carboxylate (1.0 g, 5.00 mmol) in DCM (40 ml), was added sodium triacetoxyborohydride (2.12 g, 10.0 mmol). The reaction was stirred at ambient temperature for 15 h under nitrogen. The reaction was quenched with saturated aqueous NaHCO3 and extracted with DCM (3 x 20 mL). The combined organic layer was dried over Na2SO4, filtered, concentrated in vacuo, and the residue was purified by silica gel column (DCM: MeOH 20 : 1) to give the desired product tert-butyl (3R)-3-(hex-5-en-2-yl(methyl)amino)pyrrolidine-1-carboxylate (340 mg, 1.21 mmol) as a light yellow oil. Yield 24% (ESI 283 [M+H] +). Step 2: tert-butyl 7-(5-(((R)-1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)hexyl)- 3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0730] To a solution of tert-butyl (3R)-3-(hex-5-en-2-yl(methyl)amino)pyrrolidine-1- carboxylate (650 mg, 2.3 mmol) in THF (dry, 5 mL) under argon, was added 9-BBN (0.5 M in THF, 9.2 mL, 4.6 mmol). The reaction mixture was stirred at 50°C for 2 h, then cooled to ambient temperature, added 2-chloro-4-methoxy-1,8-naphthyridine (618 mg, 2.3 mmol), Pd(OAc)
2 (27 mg, 0.12 mmol), PCy
3 (67 mg, 0.24 mmol) and KOH (386 mg, 6.9 mmol). The reaction mixture was stirred at 80°C for 15 h. Solvent was removed in vacuo, and the residue was purified by silica gel column (DCM:MeOH 30:1) to give the desired product tert-butyl 7-(5-(((R)- 1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)hexyl)-3,4-dihydro-1,8-naphthyridine- 1(2H)-carboxylate as a pale yellow oil (880 mg, 74% yield ). MS (ESI +, m/z): 517 [M+H]+ Step 3: (3R)-N-methyl-N-(6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2- yl)pyrrolidin-3-amine dihydrochloride

[0731] Tert-butyl 7-(5-(((R)-1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)hexyl)- 3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (350 mg, 0.68 mmol) was treated with HCl in 1,4-dioxane (4M, 10 mL) at ambient temperature overnight. Solvent was removed in vacuo to afford the desired product (3R)-N-methyl-N-(6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan- 2-yl)pyrrolidin-3-amine dihydrochloride (253 mg, 0.65 mmol) as a yellow oil. Yield 96% (ESI 317 (M+H) +).
Preparation of (S)-N-methyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine Step 1: tert-butyl (S)-3-(methyl(pent-4-en-1-yl)amino)pyrrolidine-1-carboxylate

[0732] A solution of tert-butyl (S)-3-(methylamino)pyrrolidine-1-carboxylate (1.5 g, 7.5 mmol), DIEA (2.91 g, 22.5 mmol) and 5-bromopent-1-ene (1.34 g, 9.0 mmol) in MeCN (20 mL) was stirred under reflux for 15 h. The reaction mixture was concentrated in vacuo and the residue was purified by silica gel column (pet ether: ethyl acetate 3:1) to give the desired product tert- butyl (S)-3-(methyl(pent-4-en-1-yl)amino)pyrrolidine-1-carboxylate as a pale yellow oil (1.9 g, 94 % yield ). MS (ESI +, m/z): 269 [M+H]+. Step 2: tert-butyl (S)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)pentyl)- 3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0733] To a solution of tert-butyl (S)-3-(methyl(pent-4-en-1-yl)amino)pyrrolidine-1- carboxylate (1.9 g, 7.1 mmol) in THF (dry, 10 mL) under argon, was added 9-BBN (0.5 M in THF, 28.4 mL, 14.2 mmol). The reaction mixture was stirred at 50°C for 2 h, then cooled to ambient temperature, added 2-chloro-4-methoxy-1,8-naphthyridine (2.28 g, 8.5 mmol), Pd(OAc)2 (80 mg, 0.35 mmol), PCy
3 (199 mg, 0.71 mmol) and KOH (1.18 g, 21.3 mmol). The reaction mixture was stirred at 80°C for 15 h. Solvent was removed in vacuo, and the residue was purified by silica gel column (DCM:MeOH 30:1) to give the desired product tert-butyl (S)-7-(5- ((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine- 1(2H)-carboxylate as a pale yellow oil (2.4 g, 67% yield ). MS (ESI +, m/z): 503[M+H]+ Step 3: (S)-N-methyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)pyrrolidin-3- amine

[0734] A mixture of tert-butyl (S)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3- yl)(methyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (2.4 g, 4.8 mmol) and TFA (20 mL) in DCM (20 mL) was stirred at RT for 16 h. The mixture was concentrated in vacuo to give the desired product (S)-N-methyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine as a yellow oil (1.35 g, 93% yield). MS (ESI +, m/z): 302 [M+H]+. Preparation of tert-butyl (R)-7-(5-((tert-butoxycarbonyl)(pyrrolidin-3-yl)amino)pentyl)-3,4- dihydro-1,8-naphthyridine-1(2H)-carboxylate Step 1: (R)-1-benzyl-N-(pent-4-en-1-yl)pyrrolidin-3-amine

[0735] A solution of (R)-1-benzylpyrrolidin-3-amine (1.0 g, 5.7 mmol), DIEA (2.20 g, 17.0 mmol) and 5-bromopent-1-ene (849 mg, 5.7 mmol) in MeCN (20 mL) was stirred under reflux for 15 h. The reaction mixture was concentrated in vacuo and the residue was purified by silica gel column (pet ether: ethyl acetate 1:1) to give the desired product (R)-1-benzyl-N-(pent-4-en-1- yl)pyrrolidin-3-amine as a pale yellow oil (900 mg, 65 % yield ). MS (ESI +, m/z): 245 [M+H]+. Step 2: tert-butyl (R)-(1-benzylpyrrolidin-3-yl)(pent-4-en-1-yl)carbamate

[0736] To a solution of (R)-1-benzyl-N-(pent-4-en-1-yl)pyrrolidin-3-amine (900 mg, 3.69 mmol) and DIEA (952 mg, 7.38 mmol) in DCM (30 mL) were added Boc2O (1.6 g, 7.38 mmol). The reaction mixture was stirred at 25°C for 15 h. The solvent was removed in vacuo, and the residue was purified by silica gel column (pet ether: ethyl acetate 2:1) to give the desired product tert-butyl (R)-(1-benzylpyrrolidin-3-yl)(pent-4-en-1-yl)carbamate as a colorless oil (855 mg). Yield 67% (ESI 345 (M+H) +). Step 3: tert-butyl (R)-7-(5-((1-benzylpyrrolidin-3-yl)(tert-butoxycarbonyl)amino)pentyl)- 3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0737] To a solution of tert-butyl (R)-(1-benzylpyrrolidin-3-yl)(pent-4-en-1-yl)carbamate (855 mg, 2.48 mmol) in THF (dry, 10 mL) under argon, was added 9-BBN (0.5 M in THF, 10 mL, 5 mmol). The reaction mixture was stirred at 50°C for 2 h, then cooled to ambient temperature, added 2-chloro-4-methoxy-1,8-naphthyridine (667 mg, 2.48 mmol), Pd(OAc)2 (27 mg, 0.12 mmol), PCy3 (70 mg, 0.25 mmol) and KOH (417 mg, 7.44 mmol). The reaction mixture was stirred at 80°C for 15 h. Solvent was removed in vacuo, and the residue was purified by silica gel column (DCM:MeOH 30:1) to give the desired product tert-butyl (R)-7-(5-((1- benzylpyrrolidin-3-yl)(tert-butoxycarbonyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate as a pale yellow oil (750 mg, 52% yield ). MS (ESI +, m/z): 579[M+H]+. Step 4: tert-butyl (R)-7-(5-((tert-butoxycarbonyl)(pyrrolidin-3-yl)amino)pentyl)-3,4- dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0738] A mixture of tert-butyl (R)-7-(5-((1-benzylpyrrolidin-3-yl)(tert- butoxycarbonyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (750 mg, 1.30 mmol) and Pd/C (10 wt% loading, 75 mg) were stirred under hydrogen balloon at 25°C for 15 h. The mixture was filtered and concentrated in vacuo to give the desired product tert-butyl (R)-7- (5-((tert-butoxycarbonyl)(pyrrolidin-3-yl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (510 mg, 1.04 mmol) as a colorless oil. Yield 80% (ESI 489 (M+H) +). Preparation of 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((S)-3-(methyl(4-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)butyl)amino)pyrrolidin-1-yl)acetic acid Step 1: But-3-enoxy-tert-butyl-diphenyl-silane

[0739] A mixture of but-3-en-1-ol (20 g, 277.37 mmol, 23.87 mL) in DMF (200 mL) was added imidazole (37.77 g, 554.74 mmol), tert-butylchlorodiphenylsilane (99.11 g, 360.58 mmol,
92.63 mL) dropwise at 0 °C. The mixture was stirred at 20 °C for 16 h. The reaction was quenched by ice water slowly, extracted with ethyl acetate (200 mL x 2). The combined organic phase was washed with brine (300 mL), dried over anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate = 100/1 to 50/1). But-3-enoxy-tert-butyl-diphenyl-silane (85 g, 273.75 mmol, 98.69% yield) was obtained as a colorless oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.72 (dd, J = 1.5, 7.8 Hz, 4H), 7.50-7.37 (m, 6H), 5.88 (tdd, J = 6.9, 10.3, 17.1 Hz, 1H), 5.16-5.00 (m, 2H), 3.76 (t, J = 6.7 Hz, 2H), 2.37 (q, J = 6.8 Hz, 2H), 1.10 (s, 10H). Step 2: tert-butyl 7-(4-((tert-butyldiphenylsilyl)oxy)butyl)-3,4-dihydro-1,8-naphthyridine- 1(2H)-carboxylate
[0740] A mixture of (but-3-en-1-yloxy)(tert-butyl)diphenylsilane (3 g, 9.66 mmol) in THF (30 mL) was added 9-BBN (0.5 M in THF, 38.65 mL) in one portion at 0 °C under N2 and stirred at 50 °C for 2 h. Then the mixture was added to a solution of tert-butyl 7-chloro-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate (2.60 g, 9.66 mmo), Pd(dppf)Cl
2.CH
2Cl
2 (789.01 mg, 966.17 umol), Cs
2CO
3 (6.30 g, 19.32 mmol) in DMF (26 mL) and stirred at 100 °C for 16 h. The reaction was quenched by ice water slowly, extracted with ethyl acetate (100 mL x 2). The combined organic phase was washed with brine (200 mL), dried over anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate=100/1 to 1/1). tert-butyl 7-(4-((tert-butyldiphenylsilyl)oxy)butyl)- 3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (5.6 g, crude) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.70-7.63 (m, 4H), 7.46-7.34 (m, 6H), 7.29 (s, 1H), 6.79 (d, J = 7.7 Hz, 1H), 4.38 (br s, 1H), 4.05 (s, 1H), 3.78-3.73 (m, 2H), 3.69 (t, J = 6.5 Hz, 2H), 2.77-2.67 (m, 4H), 1.96-1.76 (m, 8H), 1.72-1.60 (m, 7H), 1.50 (s, 9H), 1.05 (s, 9H). LCMS (ESI) m/z = 545.3 (M+1). Step 3: tert-butyl 7-(4-hydroxybutyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate
[0741] To a solution of tert-butyl 7-(4-((tert-butyldiphenylsilyl)oxy)butyl)-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate (31 g, 56.90 mmol) in THF (300 mL) was added TBAF (1 M in THF, 170.71 mL). The mixture was stirred at 50 °C for 16 h. The reaction was concentrated in vacuum. The residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate = 100/1 to 0/1). tert-butyl 7-(4-hydroxybutyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (10 g, 32.64 mmol, 57.36% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.30 (d, J = 7.5 Hz, 1H), 6.82 (d, J = 7.5 Hz, 1H), 3.78-3.73 (m, 2H), 3.65 (br d, J = 5.0 Hz, 2H), 2.79 (t, J = 7.3 Hz, 2H), 2.73 (t, J = 6.6 Hz, 2H), 1.97-1.84 (m, 4H), 1.65 (br d, J = 7.7 Hz, 2H), 1.53 (s, 9H). Step 4: tert-butyl 7-(4-oxobutyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0742] To a solution of oxalyl chloride (4.97 g, 39.16 mmol, 3.43 mL) in DCM (80 mL) was added DMSO (4.59 g, 58.75 mmol, 4.59 mL) at -70 °C, after stirred at -70 °C for 10 min, then a solution of tert-butyl 7-(4-hydroxybutyl)-3,4-dihydro-2H-1,8-naphthyridine-1-carboxylate (6 g, 19.58 mmol) in DCM (15 mL) was added at -70 °C, after stirred at -70 °C for 0.5 h, Et3N (11.89 g, 117.49 mmol, 16.35 mL) was added and the resulting mixture was stirred at 25 °C for 20 min. Then water (70 mL) was added, the mixture was extracted with dichloromethane (50 mL x 2). The combined organic layers were washed with water (100 mL x 2) and brine (20 mL x 2), dried over Na2SO4, concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (ISCO®; 120 g SepaFlash® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum gradient @ 150 mL/min). tert-butyl 7-(4-oxobutyl)-3,4- dihydro-2H-1,8-naphthyridine-1-carboxylate (5 g, 16.43 mmol, 83.89% yield) was obtained as yellow oil. LCMS (ESI) m/z = 305.2 (M+1). Step 5: (S)-tert-butyl 7-(4-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)butyl)- 3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0743] A mixture of tert-butyl (3S)-3-(methylamino)pyrrolidine-1-carboxylate (500 mg, 2.50 mmol), tert-butyl 7-(4-oxobutyl)-3,4-dihydro-2H-1,8-naphthyridine-1-carboxylate (759.90 mg,
2.50 mmol), sodium triacetyloxyboranuide (1.32 g, 6.24 mmol) in DCM (15 mL) was degassed and purged with N
2 for 3 times, and then the mixture was stirred at 25 °C for 2 h under N
2 atmosphere. The mixture was quenched with H2O (10 mL), extracted with ethyl acetate (10 mL x 3), washed with brine (10 mL x 2), dried with anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO®; 20 g SepaFlash® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 100 mL/min) to give tert-butyl 7-[4-[[(3S)-1-tert-butoxycarbonylpyrrolidin-3-yl]-methyl-amino]butyl]-3,4- dihydro-2H-1,8-naphthyridine-1-carboxylate (1 g, 2.05 mmol, 81.97% yield) as yellow oil. LCMS (ESI) m/z = 489.4 (M+1). Step 6: (S)-N-methyl-N-(4-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)butyl)pyrrolidin-3- amine

[0744] A mixture of tert-butyl 7-[4-[[(3S)-1-tert-butoxycarbonylpyrrolidin-3-yl]-methyl- amino]butyl]-3,4-dihydro-2H-1,8-naphthyridine-1-carboxylate (1 g, 2.05 mmol) in HCl/MeOH (4 M, 15 mL) was stirred at 25 °C for 16 h. Then the reaction mixture was concentrated in vacuum to give (S)-N-methyl-N-(4-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)butyl)pyrrolidin-3-amine (0.4 g, 1.39 mmol, 67.77% yield) as yellow oil. The crude product was used the next step directly without further purification. LCMS (ESI) m/z = 289.2 (M+1). Preparation of 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(4-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)butyl)amino)pyrrolidin-1-yl)acetic acid Step 1: (R)-tert-butyl 7-(4-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)butyl)- 3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0745] To a mixture of tert-butyl (3R)-3-(methylamino)pyrrolidine-1-carboxylate (384.92 mg, 1.92 mmol) and sodium triacetyloxyboranuide (626.67 mg, 2.96 mmol) in DCM (10 mL) was added a solution of tert-butyl 7-(4-oxobutyl)-3,4-dihydro-2H-1,8-naphthyridine-1-carboxylate (450 mg, 1.48 mmol) in DCM (3 mL) dropwise at 0 °C under N
2. The mixture was stirred at 20 °C for 5 h. The reaction was quenched by ice water (15 mL) slowly, adjusted to pH = 9,
extracted with DCM (30 mL x 3). The combined organic phase was washed with brine (40 mL), dried over anhydrous Na
2SO
4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO®; 20 g SepaFlash® Silica Flash Column, Eluent of 0~95% Ethyl acetate/Methanol gradient @ 80 mL/min). tert-butyl 7-[4-[[(3R)-1-tert- butoxycarbonylpyrrolidin-3-yl]-methyl-amino] butyl]-3, 4-dihydro-2H-1, 8-naphthyridine-1- carboxylate (0.6 g, 1.23 mmol, 83.05% yield) was obtained as yellow oil. LCMS (ESI) m/z = 489.3 (M+1). Step 2: (R)-N-methyl-N-(4-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)butyl)pyrrolidin-3- amine

[0746] To a solution of tert-butyl 7-[4-[[(3R)-1-tert-butoxycarbonylpyrrolidin-3-yl]-methyl- amino] butyl]-3,4-dihydro-2H-1,8-naphthyridine-1-carboxylate (0.6 g, 1.23 mmol) in MeOH (3 mL) was added HCl/MeOH (4 M, 15 mL). The mixture was stirred at 20 °C for 12 h. The reaction mixture was concentrated in vacuo. (R)-N-methyl-N-(4-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)butyl)pyrrolidin-3-amine (400 mg, crude, HCl salt) was obtained as yellow oil. LCMS (ESI) m/z = 289.2 (M+1). Preparation of (R)-N-(tert-butyl)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine Step 1: tert-butyl (R)-3-(tert-butylamino)pyrrolidine-1-carboxylate

[0747] A mixture of tert-butyl (S)-3-((methylsulfonyl)oxy)pyrrolidine-1-carboxylate (3 g, 11.3 mmol), 2-methylpropan-2-amine (8.3 g, 113 mmol) in DMA(50 mL) was stirred at 100 °C for 16 hours. The mixture was cooled to room temperature, quenched with water and extracted with EtOAc (30 mL X 3). The combined organic solvents were concentrated and purified by silica gel chromatography (pet ether: EtOAc = 0%~50%) to give the desired product tert-butyl
(R)-3-(tert-butylamino)pyrrolidine-1-carboxylate as orange oil (2.4 g). Yield 88% (ESI 243.1 (M+H) +). Step 2: tert-butyl (R)-3-(tert-butyl(pent-4-en-1-yl)amino)pyrrolidine-1-carboxylate

[0748] A solution of tert-butyl (R)-3-(tert-butylamino)pyrrolidine-1-carboxylate (2.4 g, 9.4 mmol), 1-bromo-2-methoxyethane (2.2 g, 15 mmol) and DIPEA (2.58 g, 20 mmol) in 40 mL of DMA was stirred at 100 °C for 72 hours. The mixture was cooled to room temperature, quenched with water and extracted with EtOAc (20 mL X 3). The combined organic solvents were concentrated and purified by silica gel chromatography (pet ether: EtOAc = 0%~20%) to give the desired product tert-butyl (R)-3-(tert-butyl(pent-4-en-1-yl)amino)pyrrolidine-1-carboxylate as orange oil (350 mg). Yield 11% (ESI 311.1 (M+H) +). Step 3: tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(tert- butyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0749] A solution of tert-butyl (R)-3-(tert-butyl(pent-4-en-1-yl)amino)pyrrolidine-1- carboxylate (380 mg, 1.23 mmol) was added to 9-BBN (4.9 mL, 2.46 mmol, 0.5 N in THF) under N2 atmosphere. The mixture was stirred at 60 °C for 1.5 h and cooled to room temperature. tert- butyl 7-chloro-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (328 mg, 1.23 mmol), Pd(OAc)
2 (16 mg, 0.07 mmol), PCy
3 (42 mg, 0.14 mmol) and KOH (82 mg, 1.47 mmol) were added. The mixture was stirred at 70 °C for 16 hours, concentrated and purified by silica gel chromatography (EtOAc: PE = 0%~70%) to give the desired product tert-butyl (R)-7-(5-((1-(tert- butoxycarbonyl)pyrrolidin-3-yl)(tert-butyl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate as orange oil (416 mg). Yield 62% (ESI 545.1 (M+H) +). Step 4: (R)-N-(tert-butyl)-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)pyrrolidin- 3-amine

[0750] tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(tert-butyl)amino)pentyl)- 3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (416 mg, 0.76 mmol) was added to a solution of HCl/dioxane (2 mL, 4N). The mixture was stirred at room temperature for 16 h and concentrated to give the desired product (R)-N-(tert-butyl)-N-(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)pyrrolidin-3-amine as white solid (260 mg), which was used directly to next step. Yield 100% (ESI 345.1 (M+H) +). Preparation of (R)-N-(2,2-difluoro-5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)-N- methylpyrrolidin-3-amine Step 1: 2,2-difluoropent-4-en-1-ol

[0751] NaBH4 (2.1 g, 56 mmol) was added to a solution of ethyl 2,2-difluoropent-4-enoate 4.6 g, 28 mmol) in 38 mL of MeOH at 0 °C. The mixture was stirred at room temperature for 30 min. The mixture was quenched with water and extracted with DCM (30 mL x 3). The combined organic solvents were concentrated to give the desired product 2,2-difluoropent-4-en-1-ol as orange oil (2.3 g). Yield 67% (ESI 123.1 (M+H) +). Step 2: 2,2-difluoropent-4-en-1-yl trifluoromethanesulfonate

[0752] Pyridine (1.5 g, 18.9 mmol) and Tf2O (5.3 g, 18.9 mmol) was added to a stirred solution of 2,2-difluoropent-4-en-1-ol (2.3 g, 18.9 mmol) in 30 mL of DCM at 0 °C. The mixture was stirred at 0 °C for 1 h. The mixture was quenched with water and extracted with DCM (30 mL x 3). The combined organic solvents were concentrated to give the desired product 2,2- difluoropent-4-en-1-yl trifluoromethanesulfonate (1.9 g). Yield 38% (ESI 255.1 (M+H) +). Step 3: tert-butyl (R)-3-((2,2-difluoropent-4-en-1-yl)(methyl)amino)pyrrolidine-1- carboxylate

[0753] A solution of 2,2-difluoropent-4-en-1-yl trifluoromethanesulfonate (700 mg, 2.74 mmol), tert-butyl (R)-3-(methylamino)pyrrolidine-1-carboxylate (1.1 g, 5.5 mmol) in 8 mL of CH3CN was stirred 80 °C under MW condition for 3 hours. The mixture was concentrated and purified by silica gel chromatography (pet ether: EtOAc = 0%~30%) to give the desired product tert-butyl (R)-3-((2,2-difluoropent-4-en-1-yl)(methyl)amino)pyrrolidine-1-carboxylate (350 mg). Yield 41% (ESI 351.1 (M+H) +). Step 4: tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)-4,4- difluoropentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0754] A solution of tert-butyl (R)-3-((2,2-difluoropent-4-en-1- yl)(methyl)amino)pyrrolidine-1-carboxylate (380 mg, 1.25 mmol) was added to 9-BBN (5 mL, 2.5 mmol, 0.5 N in THF) under N2 atmosphere. The mixture was stirred at 60 °C for 1.5 h and cooled to room temperature. tert-butyl 7-chloro-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (355 mg, 1.25 mmol), Pd(OAc)2 (28 mg, 0.125 mmol), PCy3 (28 mg, 0.125 mmol) and KOH (75 mg, 1.85 mmol) were added. The mixture was stirred at 70 °C for 16 hours, concentrated and purified by silica gel chromatography (EtOAc: PE = 0%~70%) to give the desired product tert- butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)-4,4-difluoropentyl)-3,4- dihydro-1,8-naphthyridine-1(2H)-carboxylate as orange oil (359 mg). Yield 53% (ESI 539.1 (M+H) +). Step 5: (R)-N-(2,2-difluoro-5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)-N- methylpyrrolidin-3-amine
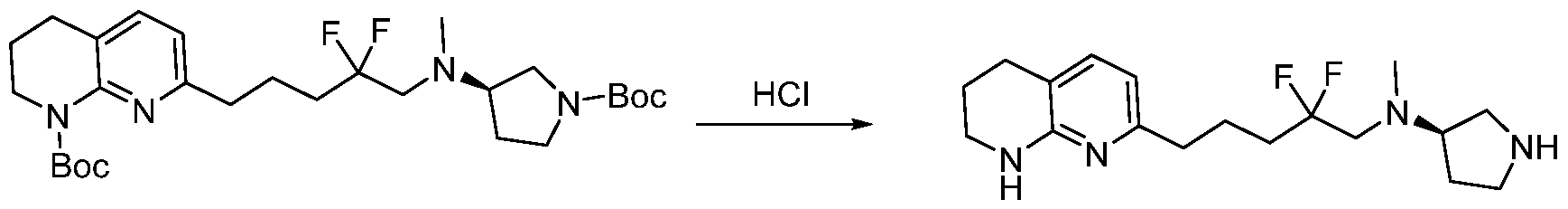
[0755] HCl/dioxane (1.63 mL, 6.4 mmol, 4N) was added to a solution of tert-butyl (R)-7-(5- ((1-(tert-butoxycarbonyl)pyrrolidin-3-yl)(methyl)amino)-4,4-difluoropentyl)-3,4-dihydro-1,8- naphthyridine-1(2H)-carboxylate (360 mg, 0.65 mmol) in 1 mL of DCM. The mixture was stirred
at room temperature for 16 hours and concentrated to give the desired product (R)-N-(2,2- difluoro-5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)-N-methylpyrrolidin-3-amine as white solid (240 mg), which was used directly to next step. Yield 100% (ESI 339.1 (M+H) +). Preparation of benzyl (R)-pyrrolidin-3-yl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)carbamate Step 1: (R)-tert-butyl 7-(5-(((benzyloxy)carbonyl)(1-(tert-butoxycarbonyl)pyrrolidin-3- yl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate

[0756] To a solution of tert-butyl (R)-7-(5-((1-(tert-butoxycarbonyl)pyrrolidin-3- yl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (1 g, 2.05 mmol) in DCM (10 mL) was added benzyl chloroformate (872.76 mg, 5.12 mmol, 727.30 uL) and TEA (621.22 mg, 6.14 mmol, 854.50 uL) at 0 °C. The mixture was stirred at 25 °C for 2 h. The reaction mixture was quenched by addition H
2O (80 mL), and extracted with ethyl acetate (20 mL x 3). The combined organic layers were washed with brine (50 mL), dried over Na
2SO
4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (ISCO®; 20 g SepaFlash® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 90 mL/min). (R)-tert-butyl 7-(5-(((benzyloxy)carbonyl)(1- (tert-butoxycarbonyl)pyrrolidin-3-yl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (1 g, 1.52 mmol, 67.69% yield, 94.9% purity) was obtained as yellow oil. LCMS (ESI) m/z = 623.4 (M+1). Step 2: benzyl (R)-pyrrolidin-3-yl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)carbamate

[0757] A solution of (R)-tert-butyl 7-(5-(((benzyloxy)carbonyl)(1-(tert- butoxycarbonyl)pyrrolidin-3-yl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (0.4 g, 642.27 umol) in HCl/MeOH (20 mL) was stirred at 50 °C for 3 h. The reaction mixture was concentrated under reduced pressure to remove solvent. benzyl (R)-pyrrolidin-3-yl(5-
(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)carbamate (0.2 g, 473.30 umol, 73.69% yield) was obtained as yellow oil. LCMS (ESI) m/z = 423.3 (M+1). Right-Side Intermediates Preparation of (2,5-difluoro-3-isopropyl-6-methoxyphenyl)boronic acid Step 1: 1-bromo-2,5-difluoro-4-methoxybenzene

[0758] A suspension of 4-bromo-2,5-difluorophenol (10 g, 47.85 mmol), K2CO3 (19.84 g, 143.55 mmol) and MeI (20.38 g, 143.55 mmol, 8.94 mL) in DMF (250 mL) was stirred for 2 h at 60 °C. The mixture was cooled to 25 °C and quenched with water (600 mL). The mixture was extracted with ethyl acetate (200 mL x 2). The combined organic phase was washed with brine (200 mL x 2), dried over anhydrous Na
2SO
4, filtered and concentrated in vacuum. The residue was purified by MPLC (Biotage ®; 80 g SepaFlash ® Silica Flash Column, Eluent of 0~3% ethyl acetate/petroleum ether gradient @ 150 mL/min) to give 1-bromo-2,5-difluoro-4- methoxybenzene (10.4 g, 46.63 mmol, 97.5% yield) as a white solid.
1H NMR (400MHz, CHLOROFORM-d) δ = 7.27 (dd, J =6.6, 10.1 Hz, 1H), 6.78 (dd, J =7.4, 9.6 Hz, 1H), 3.88 (s, 3H). Step 2: 1,4-difluoro-2-methoxy-5-(prop-1-en-2-yl)benzene
[0759] A solution of 1-bromo-2,5-difluoro-4-methoxybenzene (8.2 g, 36.77 mmol), 4,4,5,5- tetramethyl-2-(prop-1-en-2-yl)-1,3,2-dioxaborolane (15.45 g, 91.92 mmol), K
3PO
4 (23.41 g, 110.31 mmol), and Pd(OAc)
2 (825.49 mg, 3.68 mmol) in dioxane (160 mL) and water (16 mL) was stirred at 100 °C for 3 h under nitrogen atmosphere. The mixture was cooled to 25 °C and
partitioned between ethyl acetate (150 mL) and water (200 mL). The aqueous phase was extracted with ethyl acetate (150 mL x 3). The combined organic phase was washed with brine (150 mL), dried with anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by MPLC (Biotage ®; 120 g SepaFlash ® Silica Flash Column, Eluent of 0~1% ethyl acetate/petroleum ether gradient @ 150 mL/min) to give 1,4-difluoro-2-methoxy-5-(prop-1-en-2- yl)benzene (2.4 g, 13.03 mmol, 28.4% yield) as light yellow oil.
1H NMR (400MHz, CHLOROFORM-d) δ = 7.04 (dd, J = 7.2, 12.1 Hz, 1H), 6.68 (dd, J = 7.2, 11.8 Hz, 1H), 5.24 (d, J = 0.7 Hz, 1H), 5.21 (d, J = 1.1 Hz, 1H), 3.88 (s, 3H), 2.11 (d, J = 0.7 Hz, 3H) Step 3: 1,4-difluoro-2-isopropyl-5-methoxybenzene
[0760] A suspension of 1,4-difluoro-2-methoxy-5-(prop-1-en-2-yl)benzene (2.2 g, 11.94 mmol) and Pd/C (0.5 g, 10 wt% loading) in MeOH (100 mL) was stirred at 25 °C for 3 h under hydrogen (40 Psi). The mixture was filtered and the filtrate was concentrated under reduced pressure. The mixture was purified by MPLC (Biotage ®; 40 g SepaFlash ® Silica Flash Column, Eluent of 0~1% ethyl acetate/petroleum ether gradient @ 120 mL/min) to give 1,4- difluoro-2-isopropyl-5-methoxybenzene (1.9 g, 10.20 mmol, 78.4% yield) as colorless oil.
1H NMR (400MHz, CHLOROFORM-d) δ = 6.95 (dd, J = 7.1, 12.2 Hz, 1H), 6.66 (dd, J = 7.3, 11.3 Hz, 1H), 3.86 (s, 3H), 3.16 (spt, J = 6.9 Hz, 1H), 1.22 (d, J = 7.0 Hz, 6H) Step 4: (2,5-difluoro-3-isopropyl-6-methoxyphenyl)boronic acid

[0761] To a solution of 1,4-difluoro-2-isopropyl-5-methoxybenzene (500 mg, 2.69 mmol) in THF (10 mL) was added LDA (2 M, 1.75 mL) at -78 °C under nitrogen atmosphere. The reaction was stirred at -78 °C for 0.5 h. Trimethyl borate (837.12 mg, 8.06 mmol, 909.91 uL) in THF (2 mL) was added into the reaction. The reaction was stirred at -78 °C for 2 h. The reaction mixture was quenched with saturated NH4Cl (20 mL) and extracted with ethyl acetate (20 mL x 2). The combined organic layers were washed with brine (20 mL), dried over Na2SO4, filtered
and concentrated under reduced pressure to give a residue. The residue was purified by MPLC (Biotage ®; 20 g SepaFlash ® Silica Flash Column, Eluent of 0~30% ethyl acetate/petroleum ether gradient @ 120 mL/min) to give (2,5-difluoro-3-isopropyl-6-methoxyphenyl)boronic acid (380 mg, 1.65 mmol, 61.5% yield) as a white solid. Preparation of tert-butyl 2-bromo-2-(5-(1,1-difluoroethyl)-3-fluoro-2- methoxyphenyl)acetate Step 1: 1-(3-bromo-5-fluoro-4-hydroxyphenyl)ethan-1-one

[0762] To a solution of 1-(3-fluoro-4-hydroxyphenyl)ethan-1-one (25 g, 162.19 mmol) in DMF (250 mL) was added NBS (30 g, 168.55 mmol,) in portions. The mixture was stirred at 25 °C for 16 h. The reaction mixture was quenched by water (1 L), extracted with ethyl acetate (1 L x 3). The combined organic layers were washed with brine (1 L), dried over Na
2SO
4, filtered and concentrated under reduced pressure to give 1-(3-bromo-5-fluoro-4- hydroxyphenyl)ethan-1-one (151.19 g, crude) as red oil. Step 2: 1-(3-bromo-5-fluoro-4-methoxyphenyl)ethan-1-one
[0763] To a solution of 1-(3-bromo-5-fluoro-4-hydroxyphenyl)ethan-1-one (37.5 g, 160.92 mmol) in DMF (450 mL) was added K2CO3 (55.60 g, 402.30 mmol) at 0 °C. The mixture was stirred at 0 °C for 0.5 h. Iodomethane (45.68 g, 321.84 mmol, 20.04 mL) was slow dropwise added into the reaction at 0~5 °C. The mixture was stirred at 25 °C for 15.5 h. The reaction mixture was quenched by water (2 L), extracted with ethyl acetate (1 L x 3). The combined organic layers were washed with brine (1 L), dried over Na
2SO
4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO
2, petroleum ether/ethyl acetate = 100/1 to 10/1) to give 1-(3-bromo-5-fluoro-4- methoxyphenyl)ethan-1-one (130 g, 526.19 mmol, 81.1% yield) as a yellow oil.
1H NMR
(400MHz, CHLOROFORM-d) δ = 7.92 (t, J = 1.7 Hz, 1H), 7.65 (dd, J = 2.1, 11.8 Hz, 1H), 4.07 (d, J = 2.8 Hz, 3H), 2.55 (s, 3H). Step 3: 1-bromo-5-(1,1-difluoroethyl)-3-fluoro-2-methoxybenzene
[0764] 1-(3-bromo-5-fluoro-4-methoxyphenyl)ethan-1-one (2 g, 8.10 mmol) was added dropwise to (bis-(2-methoxyethyl)amino)sulfur trifluoride (10.10 g, 45.65 mmol, 10 mL) at 0°C. The mixture was stirred at 60 °C for 16 h. The reactions were poured into ice saturated NaHCO3 (400 mL) slowly and extracted with ethyl acetate (100 mL x 3). The combined organic phase was washed with brine (100 mL), dried with anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by column (Biotage ®; 80 g SepaFlash® Silica Flash Column, Eluent of petroleum ether gradient @ 120 mL/min) to give 1-bromo-5-(1,1-difluoroethyl)-3- fluoro-2-methoxybenzene (5.3 g, 19.70 mmol, 81.1% yield) as colorless oil.
1H NMR (400MHz, CHLOROFORM-d) δ = 7.48 (s, 1H), 7.22 (dd, J = 1.6, 11.3 Hz, 1H), 4.00 (d, J = 1.8 Hz, 3H), 1.90 (t, J = 18.1 Hz, 3H). Step 4: tert-butyl 2-(5-(1,1-difluoroethyl)-3-fluoro-2-methoxyphenyl)acetate

[0765] A suspension of bromo-(2-tert-butoxy-2-oxo-ethyl)zinc (1 M in THF, 27.88 mL), 1- bromo-5-(1,1-difluoroethyl)-3-fluoro-2-methoxybenzene (3 g, 11.15 mmol) and palladium tritert- butylphosphane (455.86 mg, 892.00 umol) in THF (60 mL) was stirred at 70 °C for 2 h under nitrogen atmosphere. The mixture was quenched with water (50 mL) and filtered. The filtrate was extracted with ethyl acetate (80 mL x 3). The combined organic phase was washed with brine (80 mL), dried with anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by MPLC (Biotage ®; 20 g SepaFlash® Silica Flash Column, Eluent of Petroleum ether gradient @ 100 mL/min) to give tert-butyl 2-(5-(1,1-difluoroethyl)-3-fluoro-2- methoxyphenyl)acetate (2.6 g, 8.54 mmol, 76.6% yield) as light yellow oil.
1H NMR (400MHz, CHLOROFORM-d) δ = 7.20-7.14 (m, 1H), 7.12 (s, 1H), 3.96 (d, J = 2.3 Hz, 3H), 3.59 (s, 2H), 1.90 (t, J = 18.1 Hz, 3H), 1.47 (s, 9H).
Step 5: tert-butyl 2-bromo-2-(5-(1,1-difluoroethyl)-3-fluoro-2-methoxyphenyl)acetate
[0766] To a solution of tert-butyl 2-(5-(1,1-difluoroethyl)-3-fluoro-2-methoxyphenyl)acetate (1.5 g, 4.93 mmol) in THF (30 mL) was added LiHMDS (1 M in THF, 7.39 mL) dropwise at - 70 °C under nitrogen atmosphere. The mixture was stirred at -70 °C for 0.5 h. Then TMSCl (803.29 mg, 7.39 mmol, 938.42 uL) was added. After stirred at -70 °C for 0.5 h, a solution of NBS (2.63 g, 14.79 mmol) in THF (30 mL) was added dropwise. The reaction was stirred at - 70 °C for 1 h. Then the reaction was quenched with aqueous NH
4Cl (20 mL) and extracted with ethyl acetate (20 mL x 2). The combined organic phase was washed with brine (30 mL), dried with anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by MPLC (Biotage ®; 20 g SepaFlash® Silica Flash Column, Eluent of Petroleum ether gradient @ 100 mL/min) to give tert-butyl 2-bromo-2-(5-(1,1-difluoroethyl)-3-fluoro-2- methoxyphenyl)acetate (1.5 g, 3.91 mmol, 79.4% yield) as a yellow oil.
1H NMR (400MHz, CHLOROFORM-d) δ = 7.54 (s, 1H), 7.27-7.21 (m, 1H), 5.71 (s, 1H), 4.05 (d, J = 2.4 Hz, 3H), 1.91 (t, J = 18.1 Hz, 3H), 1.48 (s, 9H). Preparation of (3-fluoro-2-methoxy-5-(1-methylcyclopropyl)phenyl)boronic acid Step 1: 1-bromo-3-fluoro-2-methoxy-5-(prop-1-en-2-yl)benzene
[0767] To a solution of methyltriphenylphosphonium bromide (11.86 g, 33.19 mmol) and t- BuOK (4.66 g, 41.49 mmol) in THF (40 mL), the mixture was stirred at 25 °C for 0.5 h. Then 1- (3-bromo-5-fluoro-4-methoxyphenyl)ethan-1-one (4.1 g, 16.60 mmol) in THF (5 mL) was added dropwise in the mixture at 0 °C, the reaction was stirred at 25 °C for 12 h. Water (10 mL) was added, the mixture was extracted with ethyl acetate (10 mL x 2). The combined organic layers were washed with water (20 mL x 2) and brine (20 mL x 2), dried over Na
2SO
4, concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO
2, Petroleum ether/Ethyl acetate = 1/0 to 0/1). 1-bromo-3-fluoro-2-methoxy-5-(prop-1-en- 2-yl)benzene (2.16 g, 8.81 mmol, 53.1% yield) was obtained as a yellow oil.
1H NMR (400 MHz,
CHLOROFORM-d) δ = 7.44-7.39 (m, 1H), 7.16 (dd, J = 2.2, 12.4 Hz, 1H), 5.35-5.30 (m, 1H), 5.12 (s, 1H), 3.96 (d, J = 1.5 Hz, 3H), 2.10 (s, 3H) Step 2: 1-bromo-3-fluoro-2-methoxy-5-(1-methylcyclopropyl)benzene
[0768] To a solution of diethylzinc (1 M, 32.64 mL) in DCM (16 mL) was added dropwise a solution of TFA (3.72 g, 32.64 mmol, 2.42 mL) in DCM (15 mL) at 0 °C under nitrogen, after stirred for 0.5 h, a solution of diiodomethane (8.74 g, 32.64 mmol, 2.63 mL) in DCM (15 mL) was added at 0 °C under nitrogen, after additional 0.5 h of stirring, a solution of 1-bromo-3- fluoro-2-methoxy-5-(prop-1-en-2-yl)benzene (1.6 g, 6.53 mmol) in DCM (15 mL) was added at 0 °C, and the resulting mixture was stirred at 25 °C for 2 h. The reaction mixture was quenched by addition ice water (60 mL) and extracted with DCM (30 mL x 3). The combined organic layers were washed with brine (60 mL), dried over Na
2SO
4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate = 100/1 to 1/1). 1-bromo-3-fluoro-2-methoxy-5-(1- methylcyclopropyl)benzene (1.5 g, 5.79 mmol, 88.7% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.20-7.18 (m, 1H), 6.93 (dd, J = 2.1, 12.2 Hz, 1H), 3.92 (s, 3H), 2.41-2.35 (m, 2H), 1.37 (s, 3H), 0.87-0.81 (m, 2H), 0.78-0.73 (m, 2H). Step 3: (3-fluoro-2-methoxy-5-(1-methylcyclopropyl)phenyl)boronic acid
[0769] To a mixture of 1-bromo-3-fluoro-2-methoxy-5-(1-methylcyclopropyl)benzene (400 mg, 1.54 mmol) and triisopropyl borate (435.49 mg, 2.32 mmol, 532.39 uL) in THF (10 mL) was added n-butyllithium (2.5 M, 926.23 uL) at -78 °C under nitrogen. The resulting mixture was stirred for 1 h at -78 °C and 1 h at 25 °C. Water (10 mL) was added, the mixture was extracted with ethyl acetate (10 mL x 2). The combined organic layers were washed with water (8 mL) and brine (5 mL), dried over Na2SO4, concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO
2, Petroleum ether/Ethyl acetate = 100/1 to
1/1). (3-fluoro-2-methoxy-5-(1-methylcyclopropyl)phenyl)boronic acid (140 mg, 624.90 umol, 40.5% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 225.1 (M+1). Preparation of (3,4-difluoro-5-isopropyl-2-methoxyphenyl)boronic acid Step 1: 2,3-difluoro-1-methoxy-4-(prop-1-en-2-yl)benzene
[0770] A solution of 1-bromo-2,3-difluoro-4-methoxybenzene (25 g, 112.10 mmol), 4,4,5,5- tetramethyl-2-(prop-1-en-2-yl)-1,3,2-dioxaborolane (47.09 g, 280.25 mmol), K3PO4 (71.39 g, 336.30 mmol) and Pd(OAc)2 (2.52 g, 11.21 mmol) in dioxane (500 mL) and water (50 mL) was stirred at 100 °C for 10 h under nitrogen atmosphere. The mixture was cooled to 25 °C and partitioned between MTBE (300 mL) and water (500 mL). The aqueous phase was further extracted with MTBE (150 mL x 2). The combined organic phase was washed with brine (200 mL), dried with anhydrous Na
2SO
4, filtered and concentrated in vacuum. The residue was purified by MPLC (Biotage ®, 220 g SepaFlash® Silica Flash Column, Eluent of petroleum ether gradient @ 150 mL/min). 2,3-difluoro-1-methoxy-4-(prop-1-en-2-yl)benzene (14.4 g, 78.18 mmol, 66.4% yield) was obtained as a yellow oil.
1H NMR (400MHz, CHLOROFORM-d) δ = 6.99 (dt, J = 2.4, 8.4 Hz, 1H), 6.70 (dt, J = 2.0, 8.2 Hz, 1H), 5.24 (br d, J = 0.6 Hz, 1H), 5.22 (br d, J = 1.3 Hz, 1H), 3.91 (s, 3H), 2.13 (d, J = 0.7 Hz, 3H) Step 2: 2,3-difluoro-1-isopropyl-4-methoxybenzene
[0771] A suspension of 2,3-difluoro-1-methoxy-4-(prop-1-en-2-yl)benzene (7.2 g, 39.09 mmol) and Pd/C (2 g, 10 wt% loading) in MeOH (150 mL) was stirred at 25 °C for 3 h under hydrogen (40 Psi). The mixture was filtered and the filtrate was concentrated under reduced pressure. The mixture was purified by MPLC (Biotage ®; 40 g SepaFlash® Silica Flash Column, Eluent of petroleum ether gradient @ 120 mL/min) to give 2,3-difluoro-1-isopropyl-4- methoxybenzene (6.3 g, 33.83 mmol, 86.6% yield) as colorless oil.
1H NMR (400MHz,
CHLOROFORM-d) δ = 6.89 (dt, J = 2.3, 8.3 Hz, 1H), 6.69 (dt, J = 2.0, 8.2 Hz, 1H), 3.89 (s, 3H), 3.25 - 3.11 (m, 1H), 1.25 (d, J = 7.0 Hz, 6H) Step 3: 1-bromo-3,4-difluoro-5-isopropyl-2-methoxybenzene
[0772] A solution of 2,3-difluoro-1-isopropyl-4-methoxybenzene (3 g, 16.11 mmol) and NBS (3.44 g, 19.33 mmol) in acetic acid (40 mL) was stirred at 80 °C for 16 h. The reaction was quenched by water (20 mL) and adjusted pH = 7 by adding saturated NaHCO3. The aqueous layer was extracted with ethyl acetate (100 mL x 2). The combined organic phase was washed with brine (100 mL), dried over anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by MPLC (Biotage ®; 80 g SepaFlash ® Silica Flash Column, Eluent of Petroleum ether gradient @ 100 mL/min) to give 1-bromo-3,4-difluoro-5-isopropyl-2- methoxybenzene (1.3 g, 4.90 mmol, 30.4% yield) as colorless oil.
1H NMR (400MHz, CHLOROFORM-d) δ = 7.16 (dd, J = 2.5, 7.1 Hz, 1H), 3.97 (d, J = 1.4 Hz, 3H), 3.19 (spt, J = 6.9 Hz, 1H), 1.25 (d, J = 6.9 Hz, 6H) Step 4: (3,4-difluoro-5-isopropyl-2-methoxyphenyl)boronic acid
[0773] To a solution of 1-bromo-3,4-difluoro-5-isopropyl-2-methoxybenzene (1.3 g, 4.90 mmol) and triisopropyl borate (1.38 g, 7.36 mmol, 1.69 mL) in THF (30 mL) was added n- butyllithium (2.5 M, 2.94 mL) at -70 °C under nitrogen atmosphere. The mixture was stirred at - 70 °C for 2 h. The reaction was quenched with saturated NH4Cl (50 mL) and extracted with ethyl acetate (20 mL x 3). The combined organic phase was washed with brine (20 mL), dried with anhydrous Na
2SO
4, filtered and concentrated in vacuum. The residue was purified by silica gel chromatography (petroleum ether/ethyl acetate = 3/1) to give (3,4-difluoro-5-isopropyl-2- methoxyphenyl)boronic acid (0.91 g, 3.96 mmol, 80.7% yield) as light yellow oil.
1H NMR
(400MHz, CHLOROFORM-d) δ = 7.45 (dd, J = 2.1, 8.4 Hz, 1H), 5.82 (br s, 2H), 4.07 (d, J = 2.6 Hz, 3H), 3.19 (spt, J = 7.0 Hz, 1H), 1.27 (d, J = 7.0 Hz, 6H). Preparation of tert-butyl 2-bromo-2-(3-fluoro-2-methoxy-5-(trifluoromethyl)phenyl)acetate Step 1: 2-bromo-6-fluoro-4-(trifluoromethyl)phenol
[0774] To a solution of 2-fluoro-4-(trifluoromethyl)phenol (1.9 g, 10.55 mmol) in acetic acid (20 mL) was added Br2 (2.19 g, 13.73 mmol, 707.90 uL) at 25 °C. The mixture was stirred at 100 °C for 12 h under nitrogen atmosphere. The reaction mixture was quenched by NaHCO3 (200 mL) and extracted with ethyl acetate (100 mL x 2). The combined organic layers were washed with brine (100 mL), dried over Na
2SO
4, filtered and concentrated under reduced pressure to give 2-bromo-6-fluoro-4-(trifluoromethyl)phenol (2.93 g, crude) as a yellow oil.
1H NMR (400 MHz, METHANOL-d) δ = 7.61 (s, 1H), 7.43 (dd, J = 2.1, 10.5 Hz, 1H). Step 2: 1-bromo-3-fluoro-2-methoxy-5-(trifluoromethyl)benzene
[0775] To a solution of 2-bromo-6-fluoro-4-(trifluoromethyl)phenol (2.73 g, 10.54 mmol) in DMF (30 mL) was added K
2CO
3 (3.64 g, 26.35 mmol) at 25 °C. Iodomethane (2.99 g, 21.08 mmol, 1.31 mL) was added dropwise slowly into the reaction. The mixture was stirred at 25 °C for 16 h. The reaction mixture was quenched with water (60 mL), extracted with ethyl acetate (20 ml x 3). The combined organic layers were washed with brine (60 ml), dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO
2, petroleum ether/ethyl acetate = 0/1) to give 1-bromo-3-fluoro-2- methoxy-5-(trifluoromethyl)benzene (1.1 g, 4.03 mmol, 38.2% yield over 2 steps) as a yellow oil.
1H NMR (400 MHz, METHANOL-d) δ = 7.93-7.18 (m, 2H), 3.92 (br d, J = 7.6 Hz, 3H). Step 3: tert-butyl 2-(3-fluoro-2-methoxy-5-(trifluoromethyl)phenyl)acetate
[0776] To a solution of 1-bromo-3-fluoro-2-methoxy-5-(trifluoromethyl)benzene (1 g, 3.66 mmol) in THF (10 mL) was added bis(tri-tert-butylphosphine)palladium(0) (374.37 mg, 732.54 umol) and (2-(tert-butoxy)-2-oxoethyl)zinc(II) bromide (1 M in THF, 20 mL) at 25 °C. The mixture was stirred at 70 °C for 16 h under nitrogen atmosphere. The reaction mixture was quenched by water (20 mL), the mixture was filtered extracted, to give filtrate and extracted filtrate with ethyl acetate (10 mL x 2). The combined organic layers were washed with brine (10 mL), dried over Na
2SO
4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO
2, petroleum ether/ethyl acetate = 1/0 to 1/1) to give tert-butyl 2-(3-fluoro-2-methoxy-5-(trifluoromethyl)phenyl)acetate (0.9 g, 2.92 mmol, 79.7% yield) as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.27 (br s, 2H), 4.01 (d, J = 2.6 Hz, 3H), 3.61 (s, 2H), 1.48 (s, 9H). Step 4: tert-butyl 2-bromo-2-(3-fluoro-2-methoxy-5-(trifluoromethyl)phenyl)acetate
[0777] A mixture of tert-butyl 2-(3-fluoro-2-methoxy-5-(trifluoromethyl)phenyl)acetate (0.8 g, 2.60 mmol) in THF (5 mL) was added LDA (2 M, 2 mL) at -78 °C. The reaction was stirred at -78 °C for 0.5 h. TMSCl (422.90 mg, 3.89 mmol, 494.05 uL) was added into the reaction. The reaction was stirred at -78 °C for 0.5 h. A solution of NBS (923.77 mg, 5.19 mmol) in THF (5 mL) was added into the reaction. The reaction was stirred at -78 °C for 1 h. The reaction mixture was quenched by aqueous NH4Cl (20 mL) and extracted with ethyl acetate (20 mL x 2). The combined organic layers were washed with brine (20 mL), dried over Na
2SO
4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO
2, petroleum ether/ethyl acetate = 1/0 to 1/1) to give tert-butyl 2-bromo-2- (3-fluoro-2-methoxy-5-(trifluoromethyl)phenyl)acetate (710 mg, 1.83 mmol, 70.6% yield) as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.70 (s, 1H), 7.35 (dd, J = 1.9, 11.5 Hz, 1H), 5.68 (s, 1H), 4.09 (d, J = 3.0 Hz, 3H), 1.48 (s, 9H).
Preparation of (3-fluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4-yl)phenyl)boronic acid Step 1: 1-bromo-3-fluoro-5-iodo-2-methoxybenzene
[0778] To a solution of 1-bromo-3-fluoro-2-methoxybenzene (9 g, 43.90 mmol) and NIS (14.81 g, 65.85 mmol) in acetic acid (90 mL) was added H
2SO
4 (9 mL) dropwise at 25 °C. The mixture was stirred for 2 h at 25 °C. The reaction mixture was quenched with water (50 mL) and extracted with ethyl acetate (50 mL x 3). The combined organic layers were washed with brine (50 mL), dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (ISCO ®; 80 g Sepa Flash ® Silica Flash Column, Eluent of 0~20% Ethyl acetate/Petroleum ether gradient @ 100 mL/min) to give 1-bromo-3-fluoro-5-iodo-2-methoxybenzene (28 g, 84.61 mmol, 96.38% yield) as a yellow oil. Step 2: 4-(3-bromo-5-fluoro-4-methoxyphenyl)tetrahydro-2H-pyran-4-ol

[0779] i-PrMgBr (2 M, 11.33 mL) was added dropwise to a solution of 1-bromo-3-fluoro-5- iodo-2-methoxybenzene (5 g, 15.11 mmol) in THF (100 mL) at 0 °C. The mixture was stirred for 1 h at 0 °C and then cooled to -78 °C. Then compound 3 (2.27 g, 22.66 mmol, 2.08 mL) was added dropwise to the mixture and the resulting mixture was stirred for 1 h at -78 °C and 1 h at 25 °C. The reaction mixture was quenched with aqueous NH4Cl (50 mL) and extracted with ethyl acetate (100 mL x 2). The combined organic layers were washed with brine (100 mL), dried over Na
2SO
4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (ISCO ®; 40 g Sepa Flash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 150 mL/min) to give 4-(3- bromo-5-fluoro-4-methoxyphenyl)tetrahydro-2H-pyran-4-ol (3 g, 9.83 mmol, 65.07% yield) as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.43 (t, J = 1.8 Hz, 1H), 7.18 (dd, J = 2.2, 12.2 Hz, 1H), 3.96-3.91 (m, 3H), 3.90-3.81 (m, 4H), 2.13-2.03 (m, 2H), 1.66-1.57 (m, 2H). Step 3: 4-(3-bromo-5-fluoro-4-methoxyphenyl)-4-methyltetrahydro-2H-pyran
[0780] Titanium (IV) chloride (1.86 g, 9.83 mmol) was added to DCM (50 mL) at 0 °C. The mixture was cooled to -40 °C and treated with dimethylzinc (1 M, 9.83 mL) at -40 °C. The resulting mixture was stirred for 0.5 h at -40 °C, then a solution of 4-(3-bromo-5-fluoro-4- methoxyphenyl)tetrahydro-2H-pyran-4-ol (1.5 g, 4.92 mmol) in DCM (10 mL) was added dropwise to the mixture. The resulting mixture was stirred for 0.5 h at -40 °C and 2 h at 25 °C. The reaction mixture was added dropwise to ice water (200 mL) at 0 °C, then extracted with ethyl acetate (150 mL x 3). The combined organic layers were washed with aqueous NaHCO
3 (100 mL), brine (50 mL), dried over Na
2SO
4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (ISCO ®; 25 g Sepa Flash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/Petroleum ether gradient @ 100 mL/min) to give 4-(3-bromo-5-fluoro-4-methoxyphenyl)-4-methyltetrahydro-2H-pyran (1 g, 3.30 mmol, 67.10% yield) as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.26-7.24 (m, 1H), 7.01 (dd, J = 2.3, 12.8 Hz, 1H), 3.97-3.94 (m, 3H), 3.80-3.63 (m, 4H), 2.04-1.95 (m, 2H), 1.76-1.67 (m, 2H), 1.27 (s, 3H). Step 4: (3-fluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4-yl)phenyl)boronic acid

[0781] n-Butyllithium (2.5 M, 990.00 uL) was added dropwise to a solution of triisopropyl borate (930.95 mg, 4.95 mmol, 1.14 mL) and 4-(3-bromo-5-fluoro-4-methoxyphenyl)-4- methyltetrahydro-2H-pyran (0.5 g, 1.65 mmol) in THF (10 mL) at -78 °C under nitrogen. The resulting mixture was stirred for 1 h at -78 °C and 1 h at 20 °C. The reaction mixture was diluted with water (50 mL) and then adjusted to pH = 5 with 1 N HCl, the resulting mixture was extracted with ethyl acetate (50 mL x 3). The combined organic layers were washed with brine (50 mL), dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (ISCO ®; 20 g Sepa Flash ® Silica Flash Column, Eluent of 0~100% THF/Petroleum ether gradient @ 100 mL/min). (3-fluoro-2- methoxy-5-(4-methyltetrahydro-2H-pyran-4-yl)phenyl)boronic acid (0.3 g, 1.12 mmol, 67.82% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 233.1 (M+1).
Preparation of (S)-(3-fluoro-2-methoxy-5-(1-methoxypropan-2-yl)phenyl)boronic acid Step 1: 2-(3-bromo-5-fluoro-4-methoxyphenyl)propan-1-ol

[0782] To a solution of 1-bromo-3-fluoro-2-methoxy-5-(prop-1-en-2-yl)benzene (10 g, 40.80 mmol) in THF (50 mL) was added BH3 ^THF (1 M, 48.96 mL) at 0 °C, after stirred at 25 °C for 2 h, aqueous NaOH (6 M, 17.00 mL) and water
2 (23.13 g, 204.01 mmol, 19.60 mL, 30% purity) was added below 10 °C, the reaction was stirred at 25 °C for 2 h. The mixture was quenched with Na
2SO
3 solution (100 mL), then extracted with ethyl acetate (100 mL). The organic layer was concentrated and the residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate = 5/1) to afford racemic 2-(3-bromo-5-fluoro-4-methoxyphenyl)propan-1-ol (8 g, 30.41 mmol, 74.5% yield) as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.13 (s, 1H), 6.88 (d, J = 13.6 Hz, 1H), 3.59-3.62 (m, 2H), 2.78-2.83 (m, 1H), 1.17 (d, J = 6.8 Hz, 3H). [0783] 2-(3-bromo-5-fluoro-4-methoxyphenyl)propan-1-ol (5 g, 19.00 mmol) was separated by SFC (column: DAICEL CHIRALPAK AD (250 mm x 50 mm x 10 um); mobile phase: [0.1% NH3water EtOH]; B%: 20%-20%, 5 min) to give two peaks: [0784] Peak 1, arbitrarily assigned as (S)-2-(3-bromo-5-fluoro-4-methoxyphenyl)propan-1- ol (2 g, 7.60 mmol, 80.0% yield) was obtained as colorless oil. SFC: R
t = 1.971 min, 49.48% [0785] Peak 2, arbitrarily assigned as (R)-2-(3-bromo-5-fluoro-4-methoxyphenyl)propan-1- ol (2 g, 7.60 mmol, 80.0% yield) was obtained as colorless oil. SFC: R
t = 2.449 min, 50.52% Step 2: (S)-1-bromo-3-fluoro-2-methoxy-5-(1-methoxypropan-2-yl)benzene
[0786] To a solution of NaH (136.82 mg, 3.42 mmol, 60% purity) in DMF (6 mL) was added a solution of (S)-2-(3-bromo-5-fluoro-4-methoxyphenyl)propan-1-ol (0.6 g, 2.28 mmol) in DMF (2 mL) at 0 °C, after stirred at 0 °C for 1 h, iodomethane (485.53 mg, 3.42 mmol, 212.95 uL) was
added, the resulting mixture was stirred at 25 °C for 2 h. The reaction mixture was quenched by ice water (10 mL), extracted with ethyl acetate (10 mL x 2). The combined organic layers were washed with brine (10 mL), dried over Na2SO4, filtered and concentrated, the residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate = 5/1). (S)-1-bromo-3- fluoro-2-methoxy-5-(1-methoxypropan-2-yl)benzene (0.5 g, 1.80 mmol, 79.1% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 245.1, 247.0 (M-32+1). Step 3: (S)-(3-fluoro-2-methoxy-5-(1-methoxypropan-2-yl)phenyl)boronic acid

[0787] To a solution of (S)-1-bromo-3-fluoro-2-methoxy-5-(1-methoxypropan-2-yl)benzene (450 mg, 1.62 mmol) and 2-(5,5-dimethyl-1,3,2-dioxaborinan-2-yl)-5,5-dimethyl-1,3,2- dioxaborinane (1.10 g, 4.87 mmol) in dioxane (6 mL) was added PCy
3 (68.30 mg, 243.57 umol), Pd(dba)
2 (140.05 mg, 243.57 umol) and KOAc (318.72 mg, 3.25 mmol), the reaction was stirred at 90 °C for 16 h under nitrogen. The mixture was filtered and concentrated, the residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate = 5/1 to 1/1), (S)-(3- fluoro-2-methoxy-5-(1-methoxypropan-2-yl)phenyl)boronic acid (400 mg, crude) was obtained as a yellow oil. Preparation of (R)-(3-fluoro-2-methoxy-5-(1-methoxypropan-2-yl)phenyl)boronic acid Step 1: (R)-1-bromo-3-fluoro-2-methoxy-5-(1-methoxypropan-2-yl)benzene

[0788] To a solution of NaH (228.03 mg, 5.70 mmol, 60% purity) in DMF (10 mL) was added dropwise a solution of (R)-2-(3-bromo-5-fluoro-4-methoxyphenyl)propan-1-ol (1 g, 3.80 mmol) in DMF (2 mL) at 0 °C, after stirred for 0.25 h, iodomethane (809.22 mg, 5.70 mmol, 354.92 uL) was added to the mixture. The resulting mixture was stirred at 25 °C for 12 h. The reaction was quenched by ice water (10 mL), extracted with ethyl acetate (10 mL x 2). The combined organic phase was washed with brine (20 mL), dried over anhydrous Na2SO4, filtered
and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO®; 4 g SepaFlash® Silica Flash Column, Eluent of 0~100% Ethyl acetate/petroleum ether gradient @ 100 mL/min). (R)-1-bromo-3-fluoro-2-methoxy-5-(1-methoxypropan-2-yl)benzene (1.1 g, crude) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.19 (t, J = 1.6 Hz, 1H), 6.94 (dd, J = 2.0, 11.9 Hz, 1H), 3.93 (d, J = 0.9 Hz, 3H), 3.48-3.37 (m, 2H), 3.33 (s, 3H), 3.00-2.88 (m, 1H), 1.23 (d, J = 7.0 Hz, 3H). Step 2: (R)-2-(3-fluoro-2-methoxy-5-(1-methoxypropan-2-yl)phenyl)-4,4,5,5-tetramethyl- 1,3,2-dioxaborolane
[0789] To a solution of (R)-1-bromo-3-fluoro-2-methoxy-5-(1-methoxypropan-2-yl)benzene (400 mg, 1.44 mmol) in dioxane (8 mL) was added KOAc (283.31 mg, 2.89 mmol), bis(pinacolato)diboron (733.06 mg, 2.89 mmol), Pd(dba)
2 (165.99 mg, 288.67 umol) and PCy
3 (80.95 mg, 288.67 umol). The mixture was stirred at 90 °C for 16 h. The reaction was filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO®; 10 g SepaFlash® Silica Flash Column, Eluent of 0~100% ethyl acetate/petroleum ether gradient @ 100 mL/min). (R)-2-(3-fluoro-2-methoxy-5-(1-methoxypropan-2-yl)phenyl)-4,4,5,5- tetramethyl-1,3,2-dioxaborolane (300 mg, 1.24 mmol, 85.9% yield) was obtained as a yellow solid. Preparation of (S)-2-(3-(5,5-dimethyl-1,3,2-dioxaborinan-2-yl)-5-fluoro-4- methoxyphenyl)propan-1-ol

[0790] To a solution of (S)-2-(3-bromo-5-fluoro-4-methoxyphenyl)propan-1-ol (0.6 g, 2.28 mmol) and 5,5,5',5'-tetramethyl-2,2'-bi(1,3,2-dioxaborinane) (1.55 g, 6.84 mmol) in dioxane (5 mL) was added Pd(dba)2 (131.10 mg, 228.00 umol), PCy3 (95.91 mg, 342.00 umol) and KOAc (447.53 mg, 4.56 mmol), the reaction was stirred at 90 °C for 12 h under nitrogen. The reaction mixture was filtered and the filtrate was concentrated to give crude product. The residue was purified by column chromatography (SiO
2, Petroleum ether/ethyl acetate = 100/1 to 0/1). (S)-2- (3-(5,5-dimethyl-1,3,2-dioxaborinan-2-yl)-5-fluoro-4-methoxyphenyl)propan-1-ol (1.6 g, crude) was obtained as a yellow oil. LCMS (ESI) m/z = 211.1 (M-86+1). Preparation of (R)-2-(3-(5,5-dimethyl-1,3,2-dioxaborinan-2-yl)-5-fluoro-4- methoxyphenyl)propan-1-ol
[0791] To a solution of (R)-2-(3-bromo-5-fluoro-4-methoxyphenyl)propan-1-ol (1.55 g, 6.84 mmol) and compound 2 (1.55 g, 6.84 mmol) in dioxane (5 mL) was added Pd(dba)
2 (131.13 mg, 228.05 umol), PCy
3 (95.93 mg, 342.07 umol) and KOAc (447.62 mg, 4.56 mmol), the reaction was stirred at 90 °C for 12 h. The reaction mixture was filtered and the filtrate was concentrated to give product. The residue was purified by column chromatography (SiO2, Petroleum ether/ethyl acetate = 100/1 to 0/1). (R)-2-(3-(5,5-dimethyl-1,3,2-dioxaborinan-2-yl)-5-fluoro-4- methoxyphenyl)propan-1-ol (1.2 g, crude) was obtained as a yellow oil. LCMS (ESI) m/z = 211.1 (M-86+1). Preparation of tert-butyl 2-bromo-2-(5-(cyclopropyldifluoromethyl)-3-fluoro-2- methoxyphenyl)acetate Step 1: 1-bromo-3-fluoro-2-methoxybenzene
[0792] To a solution of 2-bromo-6-fluorophenol (16.5 g, 86.39 mmol) in acetonitrile (165 mL) was added K
2CO
3 (23.88 g, 172.78 mmol) and iodomethane (18.39 g, 129.58 mmol, 8.07 mL), the reaction was stirred at 25 °C for 12 h. Water (300 mL) was added, the mixture was extracted with ethyl acetate (200 mL x 2). The combined organic layers were washed with water mL (200 mL x 2) and brine (100 mL x 2), dried over Na2SO4, concentrated under reduced pressure to give a residue. The crude product was used for next step directly without purification. 1-bromo-3-fluoro-2-methoxybenzene (53 g, crude) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.20-7.18 (m, 1H), 7.00-6.90 (m, 1H), 6.80-6.70 (m, 1H), 3.84- 3.81 (m, 3H). Step 2: 1-bromo-3-fluoro-5-iodo-2-methoxybenzene
[0793] To a solution of 1-bromo-3-fluoro-2-methoxybenzene (7 g, 34.14 mmol), NIS (11.52 g, 51.21 mmol) in acetic acid (80 mL) was added H2SO4 (4 mL) dropwise at 25 °C and the mixture was stirred at 25 °C for 12 h. Water (500 mL) was added, the mixture was extracted with ethyl acetate (300 mL x 2). The combined organic layers were washed with Na2SO3 (100 mL x 2) and brine (100 mL), dried over Na2SO4, concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO
2, Petroleum ether/Ethyl acetate = 10/1 to 0/1). 1-bromo-3-fluoro-5-iodo-2-methoxybenzene (22 g, 66.48 mmol, 48.7% yield) was obtained as a colorless oil. Step 3: (3-bromo-5-fluoro-4-methoxyphenyl)(cyclopropyl)methanone
[0794] To a solution of 1-bromo-3-fluoro-5-iodo-2-methoxybenzene (15 g, 45.33 mmol) in THF (150 mL) was added isopropylmagnesium chloride (1.3 M, 41.84 mL) at 0 °C, after stirred at 0 °C for 0.5 h, a solution of N-methoxy-N-methylcyclopropanecarboxamide (14.64 g, 113.32 mmol) in THF (10 mL) was added at 0 °C, then the reaction was stirred at 0 °C for 1.5 h. The mixture was poured into water (50 mL), extracted with ethyl acetate (80 mL x 2), the organic layer was concentrated to give the crude product, the residue was purified by column
chromatography (SiO
2, Petroleum ether/Ethyl acetate = 10/1). (3-bromo-5-fluoro-4- methoxyphenyl)(cyclopropyl)methanone (7 g, 25.63 mmol, 56.6% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 273.0, 275.0 (M+1). Step 4: 1-bromo-5-(cyclopropyldifluoromethyl)-3-fluoro-2-methoxybenzene
[0795] To a solution of (3-bromo-5-fluoro-4-methoxyphenyl)(cyclopropyl)methanone (5 g, 18.31 mmol) in (bis-(2-methoxyethyl)amino)sulfur trifluoride (30.30 g, 136.95 mmol, 30.00 mL) was added EtOH (84.34 mg, 1.83 mmol) at 0 °C, the reaction was stirred at 60 °C for 72 h. The mixture was added dropwise to a solution of water (30 mL) and NaHCO3 (30 mL) solution below 0 °C, then the mixture was extracted with ethyl acetate (50 mL x 2), the organic layer was concentrated, the residue was purified by column chromatography (SiO
2, Petroleum ether/Ethyl acetate = 1/0 to 20/1). 1-bromo-5-(cyclopropyldifluoromethyl)-3-fluoro-2-methoxybenzene (0.5 g, 1.69 mmol, 4.6% yield) was obtained as a yellow oil. Step 5: tert-butyl 2-(5-(cyclopropyldifluoromethyl)-3-fluoro-2-methoxyphenyl)acetate

[0796] To a solution of 1-bromo-5-(cyclopropyldifluoromethyl)-3-fluoro-2-methoxybenzene (0.5 g, 1.69 mmol) and bis(tri-tert-butylphosphine)palladium(0) (173.18 mg, 338.87 umol) in THF (2 mL) was added bromo-(2-tert-butoxy-2-oxo-ethyl)zinc (1 M in THF, 10.17 mL), the reaction was stirred at 70 °C for 2 h. Water (10 mL) was added, the mixture was extracted with ethyl acetate (10 mL x 2). The combined organic layers were washed with water (20 mL x 2) and brine (20 mL x 2), dried over Na2SO4, concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate = 10/1). tert-butyl 2-(5-(cyclopropyldifluoromethyl)-3-fluoro-2-methoxyphenyl)acetate (0.5 g, 1.51 mmol, 89.3% yield) was obtained as a yellow oil.
Step 6: tert-butyl 2-bromo-2-(5-(cyclopropyldifluoromethyl)-3-fluoro-2- methoxyphenyl)acetate

[0797] To a solution of tert-butyl 2-(5-(cyclopropyldifluoromethyl)-3-fluoro-2- methoxyphenyl)acetate (0.5 g, 1.51 mmol) in THF (11 mL) was added LiHMDS (1 M, 2.27 mL) at -78 °C, after stirred at -78 °C for 0.5 h, TMSCl (246.66 mg, 2.27 mmol, 288.15 uL) was added and the resulting mixture was stirred at -78 °C for 0.5 h. Then a solution of NBS (808.18 mg, 4.54 mmol) in THF (4 mL) was added at -78 °C and the mixture was stirred at 25 °C for 2 h. After the reaction, the mixture was poured into ice water (10 mL), extracted with ethyl acetate (10 mL x 2), the organic layer was concentrated to give the crude product. The residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate = 10/1). tert-butyl 2- bromo-2-(5-(cyclopropyldifluoromethyl)-3-fluoro-2-methoxyphenyl)acetate (0.6 g, 1.47 mmol, 96.8% yield) was obtained as a yellow oil. Preparation of 2-(3-fluoro-2-methoxy-5-(2,2,2-trifluoroethyl)phenyl)-4,4,5,5-tetramethyl- 1,3,2-dioxaborolane Step 1: 3-bromo-5-fluoro-4-hydroxybenzaldehyde

[0798] To a solution of 3-fluoro-4-hydroxybenzaldehyde (23.65 g, 168.79 mmol) in DMF (235 mL) was added NBS (31.54 g, 177.23 mmol) at 25 °C. The mixture was stirred at 100 °C for 16 h under nitrogen atmosphere. The reaction mixture was quenched with water (720 mL) and extracted with ethyl acetate (300 mL x 2). The combined organic layers were washed with brine (200 mL), dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue. 3-bromo-5-fluoro-4-hydroxybenzaldehyde (54.32 g, crude) was obtained as a brown oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 9.78 (d, J = 2.25 Hz, 1H), 7.83 (t, J = 1.63 Hz, 1H), 7.56 (dd, J = 9.88, 1.88 Hz, 1H). Step 2: 3-bromo-5-fluoro-4-methoxybenzaldehyde
[0799] To a solution of 3-bromo-5-fluoro-4-hydroxybenzaldehyde (37.5 g, 160.92 mmol) in DMF (450 mL) was added K
2CO
3 (55.60 g, 402.30 mmol) at 0 °C. The mixture was stirred at 0 °C for 0.5 h. Iodomethane (45.68 g, 321.84 mmol, 20.04 mL) was slowly dropwise added into the reaction at 0~5
oC. The mixture was stirred at 25 °C for 15.5 h. The reaction mixture was quenched with water (2 L) and extracted with ethyl acetate (1 L x 3). The combined organic layers were washed with brine (1 L), dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO
2, petroleum ether/ethyl acetate = 100/1 to 10/1). 3-bromo-5-fluoro-4-methoxybenzaldehyde (130 g, 526.19 mmol, 81.8% yield over 2 steps) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.92 (t, J = 1.7 Hz, 1H), 7.65 (dd, J = 2.1, 11.8 Hz, 1H), 4.07 (d, J = 2.8 Hz, 3H), 2.55 (s, 3H). Step 3: 1-bromo-5-(2,2-difluorovinyl)-3-fluoro-2-methoxybenzene

[0800] A solution of triphenylphosphine (13.51 g, 51.49 mmol), KI (5.70 g, 34.33 mmol), 3- bromo-5-fluoro-4-methoxybenzaldehyde (4 g, 17.16 mmol) in acetonitrile (30 mL) was stirred for 0.5 h, then methyl 2,2-difluoro-2-(fluorosulfonyl)acetate (6.60 g, 34.33 mmol, 4.37 mL) was added slowly over a period 1 h. The resulting mixture was stirred for 3 h at 70°C under nitrogen. The reaction was quenched by ice water (20 mL) slowly, extracted with ethyl acetate (10 mL x 2). The combined organic phase was washed with brine (20 mL), dried over anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO®; 80 g Sepa Flash® Silica Flash Column, Eluent of 0~10% Ethyl acetate/Petroleum gradient @ 100 mL/min). 1-bromo-5-(2,2-difluorovinyl)-3-fluoro-2-methoxybenzene (2.66 g, 9.96 mmol, 58.03% yield) was obtained as white oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.27 (s, 1H), 7.07 (dd, J = 2.1, 12.0 Hz, 1H), 5.24-5.11 (m, 1H), 3.96 (d, J = 1.5 Hz, 3H). Step 4: 1-bromo-3-fluoro-2-methoxy-5-(2,2,2-trifluoroethyl)benzene
[0801] To a solution of 1-bromo-5-(2,2-difluorovinyl)-3-fluoro-2-methoxybenzene (2.4 g, 8.99 mmol) in water (1.2 mL) and DMA (24 mL) was added KF (1.57 g, 26.96 mmol, 631.61 uL) and 18-crown-6 (1.19 g, 4.49 mmol). The mixture was stirred at 130 °C for 16 h. The reaction was quenched by ice water (30 mL) slowly, extracted with Ethyl acetate/Petroleum = 3:1 (20 mL x 2). The combined organic phase was washed with brine (30 mL), dried over anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO ®; 12 g SepaFlash ® Silica Flash Column, Eluent of 0~20% anhydrous THF/Petroleum ether gradient @ 80 mL/min). 1-bromo-3-fluoro-2-methoxy-5-(2,2,2- trifluoroethyl)benzene (1.36 g, 4.74 mmol, 52.72% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.29-7.27 (m, 1H), 7.03 (br d, J = 11.3 Hz, 1H), 3.98 (d, J = 1.6 Hz, 3H), 3.30 (q, J = 10.5 Hz, 2H). Step 5: 2-(3-fluoro-2-methoxy-5-(2,2,2-trifluoroethyl)phenyl)-4,4,5,5-tetramethyl-1,3,2- dioxaborolane
[0802] To a solution of 1-bromo-3-fluoro-2-methoxy-5-(2,2,2-trifluoroethyl)benzene (500 mg, 1.74 mmol) in dioxane (5 mL) was added bis(pinacolato)diboron (884.66 mg, 3.48 mmol) and Pd(dba)2 (200.32 mg, 348.37 umol) and PCy3 (97.69 mg, 348.37 umol) and KOAc (341.90 mg, 3.48 mmol). The mixture was stirred at 80 °C for 16 h. The reaction was quenched by ice water (10 mL) slowly, extracted with ethyl acetate (5 mL x 2). The combined organic phase was washed with brine (10 mL), dried over anhydrous Na
2SO
4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO ®; 12 g SepaFlash ® Silica Flash Column, Eluent of 0~20% Tetrahydrofuran/Petroleum gradient @ 80 mL/min). 2-(3- fluoro-2-methoxy-5-(2,2,2-trifluoroethyl)phenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane (1.2 g,
crude) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.76 (d, J = 16.0 Hz, 1H), 7.12 (s, 1H), 3.94-3.91 (m, 2H), 3.36-3.27 (m, 2H), 1.40-1.34 (m, 12H). Preparation of (3-fluoro-5-(3-hydroxyoxetan-3-yl)-2-methoxyphenyl)boronic acid Step 1: 3-(3-bromo-5-fluoro-4-methoxyphenyl)oxetan-3-ol

[0803] i-PrMgCl ^LiCl (1.3 M, 17.43 mL) was added dropwise to a solution of 1-bromo-3- fluoro-5-iodo-2-methoxybenzene (5 g, 15.11 mmol) in THF (60 mL) at 0 °C. The mixture was stirred for 1 h at 0 °C and then cooled to -78 °C. Then oxetan-3-one (2.18 g, 30.22 mmol) was added to the reaction mixture and the mixture was stirred for 1 h at -78 °C and 1 h at 25 °C. The reaction mixture was quenched with aqueous NH4Cl (50 mL) and extracted with ethyl acetate (50 mL x 3). The combined organic layers were washed with brine (50 mL), dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (ISCO®; 40 g SepaFlash® Silica Flash Column, Eluent of 0~50% Ethyl acetate/Petroleum ether gradient @ 100 mL/min). 3-(3-bromo-5-fluoro-4- methoxyphenyl)oxetan-3-ol (3 g, 10.83 mmol, 71.66% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.60 (t, J = 1.7 Hz, 1H), 7.34 (dd, J = 2.1, 11.8 Hz, 1H), 4.94-4.75 (m, 4H), 3.97 (d, J = 1.3 Hz, 3H). Step 2: (3-fluoro-5-(3-hydroxyoxetan-3-yl)-2-methoxyphenyl)boronic acid
[0804] To the mixture of 3-(3-bromo-5-fluoro-4-methoxyphenyl)oxetan-3-ol (1 g, 3.61 mmol), 5,5,5',5'-tetramethyl-2,2'-bi(1,3,2-dioxaborinane) (2.45 g, 10.83 mmol) in dioxane (50 mL) was added Pd(dba)2 (294.72 mg, 360.90 umol) and KOAc (708.39 mg, 7.22 mmol). The mixture was stirred at 90 °C for 16 h under nitrogen. The mixture was quenched with water (50 mL), extracted with ethyl acetate (50 mL x 3). The combined organic layers were washed with brine (50 mL x 2), dried with anhydrous Na
2SO
4, filtered and concentrated in vacuum. The residue was purified by prep-HPLC (column: Phenomenex Luna 80 x 30 mm x 3 um; mobile
phase: A water (TFA 0.1%), B acetonitrile 0%-30%, 8 min; Flow Rate 25 mL/min) to give 220 mg racemic product. (3-fluoro-5-(3-hydroxyoxetan-3-yl)-2-methoxyphenyl)boronic acid (220 mg, 909.06 umol, 25.19% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d), δ ppm 7.82 (d, J = 1.34 Hz, 1H), 7.47 (dd, J = 13.20, 2.08 Hz, 1H), 4.97 (d, J = 5.75 Hz, 4H), 4.09 (d, J = 2.69 Hz, 3H). LCMS: (ESI) m/z = 225.2 (M-18+1). Preparation of (2-ethoxy-3-fluoro-5-isopropylphenyl)boronic acid Step 1: 1-(3-bromo-4-ethoxy-5-fluorophenyl)ethan-1-one
[0805] To a solution of 1-(3-bromo-5-fluoro-4-hydroxyphenyl)ethan-1-one (10 g, 42.91 mmol) in DMF (50 mL) was added K2CO3 (11.86 g, 85.82 mmol) and iodoethane (13.39 g, 85.82 mmol, 6.86 mL), the mixture was stirred at 25 °C for 16 h. Water (150 mL) was added, the mixture was extracted with ethyl acetate (50 mL x 2). The combined organic layers were washed with water (50 mL x 2) and brine (20 mL x 2), dried over Na
2SO
4, concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (ISCO ®; 200 g Sepa Flash ® Silica Flash Column, Eluent of 0~20% ethyl acetate/petroleum ether gradient @ 120 mL/min). 1-(3-bromo-4-ethoxy-5-fluorophenyl)ethan-1-one (7.46 g, 28.57 mmol, 66.6% yield) was obtained as colorless oil. LCMS (ESI) m/z = 261.0, 263.0 (M+1). Step 2: 1-bromo-2-ethoxy-3-fluoro-5-(prop-1-en-2-yl)benzene

[0806] To a solution of 1-(3-bromo-4-ethoxy-5-fluorophenyl)ethan-1-one (7.46 g, 28.57 mmol) in THF (100 mL) was added methyltriphenylphosphonium bromide (20.41 g, 57.15 mmol) and t-BuOK (6.41 g, 57.15 mmol), the mixture was stirred at 25 °C for 16 h. Water (100 mL) was added, the mixture was extracted with ethyl acetate (100 mL x 2). The combined organic layers were washed with water (80 mL x 2) and brine (20 mL x 2), dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (ISCO ®; 80 g Sepa Flash ® Silica Flash Column, Eluent of
0~20% Ethyl acetate/petroleum ether gradient @ 120 mL/min). 1-bromo-2-ethoxy-3-fluoro-5- (prop-1-en-2-yl)benzene (6 g, 23.16 mmol, 81.1% yield) was obtained as brown oil.1H NMR (400MHz, CHLOROFORM-d) δ = 7.42 (t, J = 1.9 Hz, 1H), 7.15 (dd, J = 2.1, 12.3 Hz, 1H), 5.35- 5.30 (m, 1H), 5.11 (s, 1H), 4.18 (dq, J = 0.6, 7.0 Hz, 2H), 2.10 (s, 3H), 1.44 (t, J = 7.1 Hz, 3H). Step 3: 1-bromo-2-ethoxy-3-fluoro-5-isopropylbenzene

[0807] To a solution of 1-bromo-2-ethoxy-3-fluoro-5-(prop-1-en-2-yl)benzene (5.4 g, 20.84 mmol) in THF (80 mL) was added Pd/C (2.5 g, 5 wt% loading) under hydrogen (15 psi), the mixture was stirred at 25 °C for 16 h. The reaction mixture was filtered and concentrated in vacuum to get a residue. 1-bromo-2-ethoxy-3-fluoro-5-isopropylbenzene (4.8 g, crude) was obtained as brown oil.
1H NMR (400MHz, CHLOROFORM-d) δ = 7.17 (s, 1H), 6.95-6.87 (m, 1H), 4.17-4.08 (m, 2H), 2.83 (td, J = 6.8, 13.6 Hz, 1H), 1.47-1.39 (m, 3H), 1.22 (d, J = 6.9 Hz, 6H). Step 4: (2-ethoxy-3-fluoro-5-isopropylphenyl)boronic acid
[0808] n-Butyllithium (2.5 M, 3.45 mL) was added dropwise to a solution of 1-bromo-2- ethoxy-3-fluoro-5-isopropylbenzene (1.5 g, 5.74 mmol) and triisopropyl borate (1.62 g, 8.62 mmol, 1.98 mL) in THF (35 mL) at -78 °C under nitrogen. The resulting mixture was stirred for 1 h at -78 °C and 1 h at 25 °C. Water (30 mL) was added, the mixture was extracted with ethyl acetate (30 mL x 2). The combined organic layers were washed with brine (10 mL x 2), dried over Na
2SO
4, filtered and concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (ISCO ®; 80 g Sepa Flash ® Silica Flash Column, Eluent of 0~100% Ethyl acetate/petroleum ether gradient @ 120 mL/min). (2-ethoxy-3-fluoro-5- isopropylphenyl)boronic acid (1.03 g, 4.56 mmol, 79.3% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 227.1 (M+1).
Preparation of 2-(2-(difluoromethoxy)-3-fluoro-5-isopropylphenyl)-5,5-dimethyl-1,3,2- dioxaborinane Step 1: 1-bromo-3-fluoro-5-isopropyl-2-methoxybenzene
[0809] To a solution of 1-bromo-3-fluoro-2-methoxy-5-(prop-1-en-2-yl)benzene (8.1 g, 33.05 mmol) in THF (100 mL) was added Pd/C (4 g, 10 wt% loading). The suspension was degassed under vacuum, purged with hydrogen several times, and was stirred under hydrogen (15 psi) at 25 °C for 12 h. The reaction mixture was filtered and the filtrate was concentrated to a residue which was purified by column chromatography (SiO
2, Petroleum ether/ethyl acetate = 1/0 to 0/1). 1-bromo-3-fluoro-5-isopropyl-2-methoxybenzene (34 g, 137.59 mmol, 83.27% yield) was obtained as colorless oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.18 (s, 1H), 6.92 (dd, J = 1.8, 12.0 Hz, 1H), 3.93 (s, 3H), 2.84 (td, J = 6.9, 13.8 Hz, 1H), 1.23 (s, 3H), 1.21 (s, 3H). Step 2: 2-bromo-6-fluoro-4-isopropylphenol
[0810] To a solution of 1-bromo-3-fluoro-5-isopropyl-2-methoxybenzene (3.9 g, 15.78 mmol) in DCM (40 mL) was added boron tribromide (11.86 g, 47.35 mmol, 4.56 mL) at 0 °C, the reaction was stirred at 25 °C for 2 h. After the reaction, the mixture was poured into ice water (70 mL) and stirred for 0.5 h, then the mixture was extracted with dichloromethane (50 mL x 2), the organic layer was combined and concentrated. The residue was purified by column chromatography (SiO
2, Petroleum ether/Ethyl acetate = 100/1 to 0/1). 2-bromo-6-fluoro-4- isopropylphenol (6 g, 25.74 mmol, 81.6% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.12 (t, J = 1.6 Hz, 1H), 6.95-6.90 (m, 1H), 2.82 (td, J = 6.9, 13.9 Hz, 1H), 1.21 (d, J = 6.9 Hz, 6H). Step 3: 1-bromo-2-(difluoromethoxy)-3-fluoro-5-isopropylbenzene
[0811] To a solution of 2-bromo-6-fluoro-4-isopropylphenol (1 g, 4.29 mmol) in DMF (10 mL) was added sodium chlorodifluoroacetate (3.27 g, 21.45 mmol), Cs2CO3 (4.19 g, 12.87 mmol) and water (2 mL), the mixture was stirred at 100 °C for 12 h. Water (20 mL) was added, the mixture was extracted with ethyl acetate (10 mL x 2). The combined organic layers were washed with water (20 mL x 2) and brine (20 mL x 2), dried over Na
2SO
4, concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate = 100/1 to 0/1). 1-bromo-2-(difluoromethoxy)-3-fluoro-5- isopropylbenzene (640 mg, 2.26 mmol, 52.7% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.26 (br s, 1H), 7.00 (dd, J = 1.8, 11.0 Hz, 1H), 6.76-6.36 (m, 1H), 2.89 (td, J = 6.9, 13.9 Hz, 1H), 1.27-1.22 (m, 6H). Step 4: 2-(2-(difluoromethoxy)-3-fluoro-5-isopropylphenyl)-5,5-dimethyl-1,3,2- dioxaborinane
[0812] To a solution of 1-bromo-2-(difluoromethoxy)-3-fluoro-5-isopropylbenzene (590 mg, 2.08 mmol) in dioxane (15 mL) was added 5,5,5',5'-tetramethyl-2,2'-bi(1,3,2-dioxaborinane) (1.41 g, 6.25 mmol), Pd(dba)2 (119.84 mg, 208.42 umol), PCy3 (87.67 mg, 312.63 umol) and KOAc (613.64 mg, 6.25 mmol), the mixture was stirred at 90 °C for 12 h. The reaction mixture was filtered and the filtrate was concentrated in vacuum to get a residue. The residue was purified by column chromatography (SiO
2, Petroleum ether/Ethyl acetate = 100/1 to 0/1).2-(2- (difluoromethoxy)-3-fluoro-5-isopropylphenyl)-5,5-dimethyl-1,3,2-dioxaborinane(540 mg, 1.71 mm ol, 82.0% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.35-7.27 (m, 2H), 7.08 (br d, J = 11.4 Hz, 1H), 6.74-6.32 (m, 1H), 3.80 (d, J = 1.5 Hz, 4H), 1.24 (dd, J = 1.5, 6.9 Hz, 6H), 1.06 (d, J = 1.6 Hz, 6H).
Preparation of (3-fluoro-5-isopropyl-2-methoxyphenyl)boronic acid
[0813] n-Butyllithium (2.5 M, 6.07 mL) was added dropwise to a solution of 1-bromo-3- fluoro-5-isopropyl-2-methoxybenzene (2.5 g, 10.12 mmol) and triisopropyl borate (2.85 g, 15.18 mmol, 3.49 mL) in THF (50 mL) at -78 °C under nitrogen. The resulting mixture was stirred for 1 h at -78 °C and 1 h at 20 °C. The reaction mixture was quenched with ice water (100 mL), adjusted to pH = 5 with aqueous HCl (1M), and the resulting mixture was extracted with ethyl acetate (100 mL x 2). The combined organic layers were washed with brine (100 mL), dried over Na
2SO
4, filtered and concentrated under reduced pressure to give a residue which was purified by flash silica gel chromatography (ISCO®; 25 g SepaFlash® Silica Flash Column, Eluent of 0~50% Acetone/Petroleum ether gradient @ 100 mL/min) to afford (3-fluoro-5-isopropyl-2- methoxyphenyl)boronic acid (1.8 g, 8.49 mmol, 83.91% yield) as a yellow oil.
1H NMR (400 MHz, METHANOL-d4) δ = 7.00 (dd, J = 1.9, 13.4 Hz, 1H), 6.89 (s, 1H), 3.94-3.80 (m, 3H), 2.86 (td, J = 6.9, 13.8 Hz, 1H), 1.22 (d, J = 6.9 Hz, 6H). LCMS (ESI) m/z = 213.1 (M+1). Preparation of (3-fluoro-5-isopropyl-2-(trifluoromethoxy)phenyl)boronic acid Step 1: 1-bromo-2-(bromodifluoromethoxy)-3-fluoro-5-isopropylbenzene
[0814] To a solution of 2-bromo-6-fluoro-4-isopropylphenol (3 g, 12.87 mmol) in DMF (20 mL) was added dibromo(difluoro)methane (10.80 g, 51.49 mmol, 4.76 mL) and Cs2CO3 (8.39 g, 25.74 mmol). The mixture was stirred at 100 °C for 12 h. Water (50 mL) was added, the mixture was extracted with ethyl acetate (20 mL x 2). The combined organic layers were washed with water mL (20 mL x 2) and brine (20 mL x 2), dried over Na
2SO
4, concentrated, the residue was purified by column chromatography (SiO
2, Petroleum ether/Ethyl acetate = 100/1 to 0/1). 1- bromo-2-(bromodifluoromethoxy)-3-fluoro-5-isopropylbenzene (1.2 g, 3.32 mmol, 25.76% yield) was obtained as a yellow oil.
Step 2: 1-bromo-3-fluoro-5-isopropyl-2-(trifluoromethoxy)benzene
[0815] To a solution of 1-bromo-2-(bromodifluoromethoxy)-3-fluoro-5-isopropylbenzene (1 g, 2.76 mmol) in DCE (10 mL) was added silver tetrafluoroborate (2.15 g, 11.05 mmol), the reaction was stirred at 60 °C for 12 h. The reaction mixture was filtered and the filtrate was concentrated, the residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate = 100/1 to 0/1). 1-bromo-3-fluoro-5-isopropyl-2-(trifluoromethoxy)benzene (700 mg, 2.33 mmol, 84.2% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.16-7.24 (m, 1 H) 6.88-7.01 (m, 1 H) 2.73-2.92 (m, 1 H) 1.08-1.23 (m, 6 H). Step 3: (3-fluoro-5-isopropyl-2-(trifluoromethoxy)phenyl)boronic acid
[0816] To a solution of 1-bromo-3-fluoro-5-isopropyl-2-(trifluoromethoxy)benzene (1.2 g, 3.99 mmol) and 5,5,5',5'-tetramethyl-2,2'-bi(1,3,2-dioxaborinane) (2.70 g, 11.96 mmol) in dioxane (20 mL) was added PCy
3 (111.77 mg, 398.57 umol), Pd(dba)
2 (229.18 mg, 398.57 umol) and KOAc (1.56 g, 15.94 mmol), the reaction was stirred at 90 °C for 12 h under nitrogen. The mixture was filtered and the filtrate was concentrated. The residue was purified by prep-HPLC (column: Phenomenex Luna C1875 x 30 mm x 3 um; mobile phase: A water (TFA 0.1%), B acetonitrile 30%-60%, 7 min; Flow Rate 25 mL/min. (3-fluoro-5-isopropyl-2- (trifluoromethoxy)phenyl)boronic acid (500 mg, 1.88 mmol, 47.2% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 290.0 (M+23). Preparation of methyl 2-bromo-2-(3-fluoro-5-isopropyl-2-(trifluoromethyl)phenyl)acetate Step 1: dimethyl 2-(5-bromo-3-fluoro-2-(trifluoromethyl)phenyl)malonate
[0817] To a solution of 5-bromo-1,3-difluoro-2-(trifluoromethyl)benzene (5 g, 19.16 mmol) and dimethyl malonate (2.53 g, 19.16 mmol, 2.20 mL) in DMF (100 mL) was added Cs
2CO
3 (6.24 g, 19.16 mmol), the reaction was stirred at 25 °C for 12 h. Water (100 mL) was added, the mixture was extracted with ethyl acetate (100 mL x 2). The combined organic layers were washed with water (50 mL x 2) and brine (50 mL x 2), dried over Na2SO4, concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO
2, Petroleum ether/Ethyl acetate = 5/1), dimethyl 2-(5-bromo-3-fluoro-2- (trifluoromethyl)phenyl)malonate (6.6 g, 17.69 mmol, 46.2 % yield) was obtained as a white solid. LCMS (ESI) m/z = 373.0, 375.0 (M+1). Step 2: methyl 2-(5-bromo-3-fluoro-2-(trifluoromethyl)phenyl)acetate
[0818] To a solution of dimethyl 2-(5-bromo-3-fluoro-2-(trifluoromethyl)phenyl)malonate (3.3 g, 8.84 mmol) in DMSO (30 mL) and water (4 mL) was added LiCl (937.43 mg, 22.11 mmol), the reaction was stirred at 100 °C for 24 h. Water (50 mL) was added, the mixture was extracted with ethyl acetate (50 mL x 2). The combined organic layers were washed with water (20 mL x 2) and brine (20 mL x 2), dried over Na
2SO
4, concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO
2, Petroleum ether/Ethyl acetate = 10/1). methyl 2-(5-bromo-3-fluoro-2-(trifluoromethyl)phenyl)acetate (2.5 g, 7.94 mmol, 44.9% yield) was obtained as colorless oil.
1H NMR (400 MHz, CHLOROFORM- d) δ = 7.34 (d, J = 10.51 Hz, 1H), 7.28 (s, 1H), 3.80-3.84 (m, 2H), 3.73 (s, 3H). Step 3: methyl 2-(3-fluoro-5-(prop-1-en-2-yl)-2-(trifluoromethyl)phenyl)acetate
[0819] To a solution of methyl 2-(5-bromo-3-fluoro-2-(trifluoromethyl)phenyl)acetate (2.4 g, 7.62 mmol) and 4,4,5,5-tetramethyl-2-(prop-1-en-2-yl)-1,3,2-dioxaborolane (3.84 g, 22.85 mmol) in dioxane (30 mL) and water (0.3 mL) was added Cs2CO3 (7.45 g, 22.85 mmol) and Pd(dppf)Cl2.CH2Cl2 (622.08 mg, 761.76 umol), the reaction was stirred at 90 °C for 12 h. Water (50 mL) was added, the mixture was extracted with ethyl acetate (10 mL x 2). The combined organic layers were washed with water (20 mL x 2) and brine (20 mL x 2), dried over Na
2SO
4, concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO
2, Petroleum ether/Ethyl acetate = 4/1), methyl 2-(3-fluoro-5-(prop-1-en-2- yl)-2-(trifluoromethyl)phenyl)acetate (1.6 g, 5.79 mmol, 76 % yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.20 (br d, J = 12.63 Hz, 1H), 7.15 (s, 1H), 5.47 (s, 1H), 5.25 (s, 1H), 3.86 (d, J = 2.13 Hz, 2H), 3.73 (s, 3H), 2.14 (s, 3H). LCMS (ESI) m/z = 257.1 (M+1). Step 4: methyl 2-(3-fluoro-5-isopropyl-2-(trifluoromethyl)phenyl)acetate
[0820] To a solution of methyl 2-(3-fluoro-5-(prop-1-en-2-yl)-2- (trifluoromethyl)phenyl)acetate (1.6 g, 5.79 mmol) in THF (30 mL) was added Pd/C (0.8 g, 5 wt% loading), the reaction was stirred at 25 °C for 12 h under hydrogen (15 psi). After the reaction, the mixture was filtered and concentrated to give the crude product. methyl 2-(3-fluoro- 5-isopropyl-2-(trifluoromethyl)phenyl)acetate (1.2 g, 4.31 mmol, crude) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 6.99 (br d, J = 12.4 Hz, 1H), 6.92 (s, 1H), 3.82 (br s, 2H), 3.71 (s, 3H), 2.96-2.86 (m, 1H), 1.25 (d, J = 7.0 Hz, 6H). LCMS (ESI) m/z = 259.1 (M+1). Step 5: methyl 2-bromo-2-(3-fluoro-5-isopropyl-2-(trifluoromethyl)phenyl)acetate

[0821] To a solution of methyl 2-(3-fluoro-5-isopropyl-2-(trifluoromethyl)phenyl)acetate (200 mg, 718.80 umol) in THF (5 mL) was added LiHMDS (1 M, 1.08 mL) with stirring at - 78 °C, the reaction was stirred at -78 °C for 0.67 h. Then TMSCl (117.14 mg, 1.08 mmol, 136.84
uL) was added at -78 °C, the mixture was stirred at -78 °C for 0.33 h. Then a solution of NBS (383.80 mg, 2.16 mmol) in THF (2 mL) was added at -78 °C, the resulting mixture was stirred at 25 °C for 0.5 h. The mixture was quenched by water (10 mL), exacted by ethyl acetate (10 mL x 3). The combined organic layers were washed with brine (20 mL), dried over Na2SO4, concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO
2, Petroleum ether/Ethyl acetate = 100/1 to 0/1). methyl 2-bromo-2-(3- fluoro-5-isopropyl-2-(trifluoromethyl)phenyl)acetate (150 mg, 420.01 umol, 58.4 % yield) was obtained as a yellow oil. Preparation of (3-fluoro-2-methoxy-5-(3-methyloxetan-3-yl)phenyl)boronic acid Step 1: 4,5,6,7-tetrachloro-1,3-dioxoisoindolin-2-yl 3-methyloxetane-3-carboxylate
[0822] N,N′-Diisopropylcarbodiimide (1.79 g, 14.21 mmol, 2.20 mL) was added dropwise to a solution of 3-methyloxetane-3-carboxylic acid (1.5 g, 12.92 mmol), 4,5,6,7-tetrachloro-2- hydroxyisoindoline-1,3-dione (4.08 g, 13.56 mmol) and N,N-dimethylpyridin-4-amine (157.82 mg, 1.29 mmol) in DCM (100 mL) at 20 °C. The mixture was stirred for 1 h at 20 °C. The mixture was filtered and the filtrate was concentrated. 4,5,6,7-tetrachloro-1,3-dioxoisoindolin-2- yl 3-methyloxetane-3-carboxylate (5.15 g, 12.91 mmol, 100.00% yield) was obtained as a yellow solid. Step 2: 3-(3-bromo-5-fluoro-4-methoxyphenyl)-3-methyloxetane

[0823] Step 2a: i-PrMgBr (2 M, 12.03 mL) was added to a solution of 1-bromo-3-fluoro-5- iodo-2-methoxybenzene (7.96 g, 24.06 mmol) in THF (36 mL) at 0 °C under nitrogen. The mixture was stirred for 0.25 h at 0 °C and then treated with zinc dibromide (5.42 g, 24.06 mmol) at 0 °C. The resulting mixture was stirred for 0.5 h at 0 °C to afford the zinc reagent.
[0824] Step 2b: 4,5,6,7-tetrachloro-1,3-dioxoisoindolin-2-yl 3-methyloxetane-3-carboxylate (3.2 g, 8.02 mmol) was dissolved in 1,3-dimethyl-2-imidazolidinone (48 mL) under nitrogen. Then nickel (II) (E)-2, 2, 6, 6-tetramethyl-5-oxohept-3-en-3-olate (682.05 mg, 1.60 mmol) and zinc dibromide (1.81 g, 8.02 mmol) was added at 0 °C and the mixture was stirred for 0.25 h. Then the prepared zinc reagent from Step 2a was added at 0 °C and the resulting mixture was stirred for 2 h at 0 °C and 16 h at 20 °C. The reaction mixture was quenched by aqueous NH
4Cl (200 mL) at 0 °C, extracted with ethyl acetate (100 mL x 2). The combined organic layers were washed with brine (200 mL), dried over Na
2SO
4, filtered and concentrated under reduced pressure to give a residue. The residue was purified twice by column chromatography (SiO2, Petroleum ether/MTBE = 100/1 to 0/100). 3-(3-bromo-5-fluoro-4-methoxyphenyl)-3- methyloxetane (900 mg, 3.27 mmol, 18.00% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.19 (s, 1H), 6.98-6.93 (m, 1H), 4.87 (d, J = 5.6 Hz, 2H), 4.62 (d, J = 5.6 Hz, 2H), 3.96 (s, 3H), 1.70 (s, 3H). Step 3: (3-fluoro-2-methoxy-5-(3-methyloxetan-3-yl)phenyl)boronic acid
[0825] To the mixture of 3-(3-bromo-5-fluoro-4-methoxyphenyl)-3-methyloxetane (500 mg, 1.82 mmol) and 5,5,5',5'-tetramethyl-2,2'-bi(1,3,2-dioxaborinane) (1.23 g, 5.45 mmol) in dioxane (5 mL) was added Pd(dppf)Cl2.CH2Cl2 (148.42 mg, 181.74 umol) and KOAc (356.73 mg, 3.63 mmol) under nitrogen. The mixture was stirred at 90 °C for 16 h under nitrogen. The reaction mixture was concentrated under reduced pressure to remove solvent. The residue was purified by column chromatography (SiO
2, Petroleum ether/MTBE = 100/1 to 0/1). The mixture was further purified by pre-HPLC (column: Waters Xbridge BEH C18100 x 30 mm x 10 um; mobile phase: A water (NH4HCO310 mmol/L), B acetonitrile 10%-40%, 8 min; Flow Rate (25 mL/min). (3- fluoro-2-methoxy-5-(3-methyloxetan-3-yl)phenyl)boronic acid (240 mg, 778.84 umol, 28.57% yield) was obtained as a yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.42 (d, J = 1.9 Hz, 1H), 7.10-7.05 (m, 1H), 5.86 (s, 2H), 4.94 (d, J = 5.6 Hz, 2H), 4.63 (d, J = 5.6 Hz, 2H), 4.06 (d, J = 2.5 Hz, 3H), 1.72 (s, 3H).
Preparation of ethyl 2-bromo-2-(3-fluoro-2-methoxy-5-(2-methoxypropan-2- yl)phenyl)acetate Step 1: methyl 3-bromo-5-fluoro-4-methoxybenzoate
[0826] A solution of methyl 3-fluoro-4-methoxybenzoate (5 g, 27.17 mmol) in conc. H
2SO
4 (30 mL) was stirred at 0 °C for 10 min, then NBS (5.3 g, 29.89 mmol) was added and the reaction stirred at ambient temperature for 1 h. The reaction mixture was quenched with ice, and extracted with ethyl acetate (3x 30 mL). The combined organic layer was concentrated in vacuo, and the residue was purified by silica gel column (PE: EA 10:1) to give the desired product methyl 3- bromo-5-fluoro-4-methoxybenzoate as a yellow oil (4.5 g). Yield 63.2% (ESI 263.0 (M+H)
+). Step 2: 2-(3-bromo-5-fluoro-4-methoxyphenyl)propan-2-ol
[0827] To a solution of methyl 3-bromo-5-fluoro-4-methoxybenzoate (2 g, 7.63 mmol) in THF (15 mL) was added MeMgBr (1M in THF, 15 mL,15 mmol) at 0 °C and stirred at ambient temperature for 4 h. The reaction mixture was quenched with water (30 mL), and extracted with ethyl acetate (3x 30 mL). The combined organic layer was concentrated in vacuo, and the residue was purified by silica gel column (PE: EA 10:1) to give the desired product 2-(3-bromo-5-fluoro- 4-methoxyphenyl)propan-2-ol as a yellow oil (1.5 g). Yield 75.0% (ESI 244.0 (M-OH) +). Step 3: 1-bromo-3-fluoro-2-methoxy-5-(2-methoxypropan-2-yl)benzene

[0828] To a solution of 2-(3-bromo-5-fluoro-4-methoxyphenyl)propan-2-ol (1.5 g, 5.72 mmol) in THF (15 mL) was added NaH (344 mg, 8.59 mmol) and stirred at 0 °C for 10 min, then added MeI (1.2 g, 8.59 mmol) and stirred at ambient temperature for 1 h. The mixture was
quenched with aqueous NH4Cl (30 mL) and extracted with ethyl acetate (3x 30mL). The combined organic layer was concentrated in vacuo, and the residue was purified by silica gel column (PE: EA 10:1) to give the desired product 1-bromo-3-fluoro-2-methoxy-5-(2- methoxypropan-2-yl)benzene as a yellow oil (1.2 g). Yield 75.9% (ESI 244.0 (M-33) +). Step 4: ethyl 2-(3-fluoro-2-methoxy-5-(2-methoxypropan-2-yl)phenyl)acetate
[0829] To a mixture of 1-bromo-3-fluoro-2-methoxy-5-(2-methoxypropan-2-yl)benzene (690 mg, 2.5 mmol), Pd
2(dba)
3 (92 mg, 0.125 mmol) and Q-Phos (115 mg, 0.125 mmol) in THF (5 mL) was added (2-ethoxy-2-oxoethyl)zinc(II) bromide solution 1 M in THF (6.5 mL, 6.5 mmol) at ambient temperature under argon. The reaction was stirred at 50°C for 2 h. The mixture was quenched with NaHCO3 solution, the solid was removed by filtration. The filtrate was concentrated in vacuo, and the residue was purified by silica gel column (pet ether: ethyl acetate 5:1) to give the desired product ethyl 2-(3-fluoro-2-methoxy-5-(2-methoxypropan-2- yl)phenyl)acetate as a pale red oil (610 mg). Yield 85.9% (ESI 307.0 (M+23) +). Step 5: ethyl 2-bromo-2-(3-fluoro-2-methoxy-5-(2-methoxypropan-2-yl)phenyl)acetate

[0830] To a solution of ethyl 2-(3-fluoro-2-methoxy-5-(2-methoxypropan-2- yl)phenyl)acetate (1.53 g, 5.0 mmol) in THF (15 mL) at -78°C, was added lithium diisopropylamide solution 2.0 M in THF/hexanes (5.0 mL, 10.0 mmol) dropwise. The reaction was stirred at -78°C for 30 min, then chlorotrimethylsilane (1.1 g, 10.0 mmol) was added and the reaction was stirred at -78°C for another 30 min. Then a solution of NBS (1.8 g, 10 mmol) in THF (10 mL) was added and the reaction was stirred at -78°C for 30min. The reaction was quenched with water (10 mL) and extracted with ethyl acetate(2x 20mL). The combined organic phase was washed with saturated NaHCO3 solution, brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether: ethyl acetate 5:1)
to give the desired product ethyl 2-bromo-2-(3-fluoro-2-methoxy-5-(2-methoxypropan-2- yl)phenyl)acetate as a yellow oil (1.4 g). Yield 77.3% (ESI 385.0 (M+23) +). Preparation of ethyl 2-bromo-2-(5-(tert-butyl)-3-fluoro-2-methoxyphenyl)acetate Step 1: 1-bromo-5-(tert-butyl)-3-fluoro-2-methoxybenzene

[0831] To a solution of TiCl4 (2.1 g, 11.1 mmol) in DCM (20 mL) at -50°C, was added a solution of dimethyl zinc (0.5 M in THF, 22.2 mL, 11.1 mmol) dropwise. The reaction mixture was stirred at -50°C for 15 min, then added a solution of 1-(3-bromo-5-fluoro-4- methoxyphenyl)ethan-1-one (1.25 g, 5.1 mmol) in anhydrous DCM (10 mL) dropwise. The reaction mixture was warmed to ambient temperature slowly, then stirred at ambient temperature overnight. The reaction was quenched with water (20 mL) and extracted with DCM (2 x 20 mL). The combined organic layer was dried over Na2SO4, filtered, concentrated in vacuo, and the residue was purified by silica gel column (pet ether: ethyl acetate 100 : 1) to give the desired product 1-bromo-5-(tert-butyl)-3-fluoro-2-methoxybenzene (1.06 g, 4.1 mmol) as a light red oil. Yield 80% (ESI 261/263[M+H] +). Step 2: ethyl 2-(5-(tert-butyl)-3-fluoro-2-methoxyphenyl)acetate

[0832] To a mixture of 1-bromo-5-(tert-butyl)-3-fluoro-2-methoxybenzene (1.26 g, 4.83 mmol), Q-Phos (171 mg, 0.24 mmol) and Pd2(dba)3 (249 mg, 0.24 mmol) in THF (10 mL) was added (2-ethoxy-2-oxoethyl)zinc(II) bromide (1 M in THF, 14.4 mL, 14.4 mmol) at RT under argon. The reaction was stirred at 50°C under nitrogen for 1 h. The reaction was quenched with saturated aqueous NaHCO3 and extracted with ethyl acetate (2 x 20 mL). The combined organic layer was dried over Na2SO4, filtered, concentrated in vacuo, and the residue was purified by silica gel column (pet ether: ethyl acetate 20:1) to give the desired product ethyl 2-(5-(tert-butyl)-
3-fluoro-2-methoxyphenyl)acetate (1.19 g, 4.44 mmol) as a light red oil. Yield 92% (ESI 269 [M+H] +). Step 3: ethyl 2-bromo-2-(5-(tert-butyl)-3-fluoro-2-methoxyphenyl)acetate

[0833] To a solution of ethyl 2-(5-(tert-butyl)-3-fluoro-2-methoxyphenyl)acetate (1.19 g, 4.44 mmol) in THF (20 mL) at -78°C, was added lithium diisopropylamide solution 2.0 M in THF/hexanes (5.6 mL, 11.2 mmol) dropwise. The reaction was stirred at -78°C for 30 min, then chlorotrimethylsilane (1.20 g, 11.1 mmol) was added and the reaction was stirred at -78°C for another 30 min. Then a solution of NBS (1.98 g, 11.1 mmol) in THF (10 mL) was added and the reaction was stirred at -78°C for 30 min. The reaction was quenched with water (20 mL), then extracted with ethyl acetate (50 mL × 3). The combined organic phase was washed with saturated NaHCO3 solution, brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether/ethyl acetate = 0%~10%) to give the desired product ethyl 2-bromo-2-(5-(tert-butyl)-3-fluoro-2-methoxyphenyl)acetate as a brown oil (1.28 g, 83% yield). ESI 347/349 (M+H) +. Preparation of ethyl 2-bromo-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetate Step 1: ethyl 2-(3-fluoro-2-methoxy-5-(prop-1-en-2-yl)phenyl)acetate

[0834] To a mixture of 1-bromo-3-fluoro-2-methoxy-5-(prop-1-en-2-yl)benzene (10 g, 40.8 mmol), Q-Phos(1.45g, 2.04 mmol) and Pd
2(dba)
3 (2.11 g, 2.04 mmol) in THF (50 mL) was added (2-ethoxy-2-oxoethyl)zinc(II) bromide (1 M in THF, 122 mL, 122 mmol) at RT under argon. The reaction was stirred at 50°C under nitrogen for 1 h. The reaction was quenched with aqueous NaHCO3 and extracted with ethyl acetate (2 x 100 mL). The combined organic layer
was dried over Na2SO4, filtered, concentrated in vacuo, and the residue was purified by silica gel column (pet ether: ethyl acetate 20:1) to give the desired product ethyl 2-(3-fluoro-2-methoxy-5- (prop-1-en-2-yl)phenyl)acetate (7.9 g, 31.1 mmol) as a light red oil. Yield 76% (ESI 253[M+H] +). Step 2: ethyl 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetate

[0835] A mixture of ethyl 2-(3-fluoro-2-methoxy-5-(prop-1-en-2-yl)phenyl)acetate (7.9 g, 31.1 mmol), and Pd/C (0.79 g, 10 wt% loading) in ethyl acetate (100 mL) was stirred at 25 °C under hydrogen for 16 h. Then the solid was removed by filtration and the filtrate was concentrated in vacuo to give the desired product ethyl 2-(2-cyano-5-fluoro-4- isopropylphenyl)acetate (7.3 g, 28.7 mmol) as a light red oil. Yield 92% (ESI 255[M+H] +). Step 3: ethyl 2-bromo-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetate

[0836] To a solution of ethyl 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetate (7.3 g, 28.7 mmol) in THF (200 mL) at -78°C, was added lithium diisopropylamide solution 2.0 M in THF/hexanes (35.9 mL, 71.8 mmol) dropwise. The reaction was stirred at -78°C for 30 min, then chlorotrimethylsilane (7.76 g, 71.8 mmol) was added and the reaction was stirred at -78°C for another 30 min. Then a solution of NBS (12.8 g, 71.8 mmol) in THF (100 mL) was added and the reaction was stirred at -78°C for 30 min. The reaction was quenched with water (100 mL), then extracted with ethyl acetate (150 mL × 3). The combined organic phase was washed with saturated NaHCO3 solution, brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether/ethyl acetate = 0%~10%) to give the desired product ethyl 2-bromo-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetate as a brown oil (8.46 g, 89% yield) ESI 333/335 (M+H) +.
Preparation of (2-ethyl-3-fluoro-5-isopropylphenyl)boronic acid Step 1: 2-(3-bromo-5-fluoro-phenyl) propan-2-ol

[0837] To a solution of methyl 3-bromo-5-fluoro-benzoate (35 g, 150.19 mmol) in THF (530 mL) was added bromo(methyl)magnesium (3 M, 150.19 mL) dropwise at 0 °C under N2. The mixture was stirred at 25 °C for 2 h. Three batches reactions were carried out together. The reaction mixtures were quenched with saturated aqueous HCl (1000 mL), extracted with ethyl acetate (1000 mL x 2). The combined organic layers were washed with brine (500 mL), dried over Na
2SO
4, filtered and concentrated to give crude product. The residue was purified by flash silica gel chromatography (Biotage®; 330 g SepaFlash® Silica Flash Column, Eluent of 0~20% Ethyl acetate/Petroleum ether gradient @ 200 mL/min). 2-(3-bromo-5-fluoro-phenyl) propan-2- ol (103 g, 441.91 mmol, 98.08% yield) was obtained as yellow oil.
1H NMR (400 MHz, CHLOROFORM-d) δ = 7.41 (t, J = 1.3 Hz, 1H), 7.16-7.08 (m, 2H), 1.54 (s, 6H). Step 2: 1-bromo-3-fluoro-5-isopropyl-benzene
[0838] A mixture of 2-(3-bromo-5-fluoro-phenyl) propan-2-ol (34.3 g, 147.16 mmol), Et3SiH (34.22 g, 294.32 mmol, 47.01 mL) and TFA (105.71 g, 927.12 mmol, 68.64 mL) in DCM (1.2 L) was degassed and purged with N
2 for 3 times, and then the mixture was stirred at 30 °C for 17 h under N
2 atmosphere. The reaction mixture was poured into water (500 mL) slowly and the pH of the mixture was adjusted to 8 with aqueous NaOH (2M), the resulting mixture was extracted with dichloromethane (500 mL x 3), the combined organic layers were dried over Na2SO4, concentrated under reduced pressure to give a residue. The residue was purified by flash silica gel chromatography (Biotage®; 330 g SepaFlash® Silica Flash Column, Eluent of 0~10% Ethyl acetate/Petroleum ether gradient @ 200 mL/min) to give 1-bromo-3-fluoro-5-isopropyl-benzene (79 g, 363.92 mmol, 82.43% yield) as white oil.
1H NMR (400 MHz, CHLOROFORM-d) δ =
7.08 (s, 1H), 6.98 (td, J = 1.8, 8.1 Hz, 1H), 6.79 (br d, J = 9.8 Hz, 1H), 2.79 (spt, J = 6.9 Hz, 1H), 1.15 (d, J = 6.9 Hz, 6H). Step 3: 1-(2-bromo-6-fluoro-4-isopropylphenyl) ethanol
[0839] To a solution of 1-bromo-3-fluoro-5-isopropyl-benzene (4 g, 18.43 mmol) in THF (40 mL) was added LDA (2 M, 13.82 mL) at -70 °C under N
2, the resulting mixture was stirred for 0.5 h at -70 °C. Then acetaldehyde (2.03 g, 46.07 mmol, 2.59 mL) was added to the mixture. The resulting mixture was stirred at -70 °C for 1.5 h. The reaction was quenched by aqueous NH4Cl (60 mL) slowly, extracted with ethyl acetate (30 mL x 2). The combined organic layers were washed with brine (60 mL), dried over anhydrous Na
2SO
4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO®; 100 g SepaFlash® Silica Flash Column, Eluent of 0~50% Ethyl acetate/Petroleum gradient @ 80 mL/min). 1-(2-bromo-6-fluoro-4-isopropyl-phenyl) ethanol (2.06 g, 39.44 mmol, 42.81% yield) was obtained as yellow oil.
1H NMR (400 MHz, METHANOL-d4) δ = 7.26 (s, 1H), 6.98 (dd, J = 1.5, 12.4 Hz, 1H), 5.33 (dq, J = 1.0, 6.8 Hz, 1H), 2.87 (td, J = 6.9, 13.8 Hz, 1H), 1.53 (dd, J = 1.0, 6.7 Hz, 3H), 1.24-1.21 (m, 6H). Step 4: 1-bromo-2-ethyl-3-fluoro-5-isopropylbenzene
[0840] To a solution of 1-(2-bromo-6-fluoro-4-isopropyl-phenyl) ethanol (1.8 g, 6.89 mmol) in DCM (20 mL) was added Et3SiH (1.60 g, 13.79 mmol, 2.20 mL) and TFA (4.72 g, 41.36 mmol, 3.06 mL). The mixture was stirred at 40 °C for 16 h. The reaction was quenched by ice water (40 mL) slowly, extracted with ethyl acetate (40 mL x 2). The combined organic phase was washed with brine (40 mL), dried over anhydrous Na
2SO
4, filtered and concentrated in vacuo. The residue was purified by flash silica gel chromatography (ISCO®; 40 g SepaFlash® Silica Flash Column, Eluent of 0~30% Ethyl acetate/Petroleum gradient @ 80 mL/min) to give 1- bromo-2-ethyl-3-fluoro-5-isopropyl-benzene (0.5 g, 4.08 mmol, 29.59% yield) as yellow oil.
1H
NMR (400 MHz, METHANOL-d
4) δ = 7.24 (s, 1H), 7.03-6.80 (m, 1H), 2.94-2.71 (m, 3H), 1.22 (d, J = 6.8 Hz, 6H), 1.14 (t, J = 7.5 Hz, 3H). Step 5: (2-ethyl-3-fluoro-5-isopropylphenyl)boronic acid

[0841] To a solution of 1-bromo-2-ethyl-3-fluoro-5-isopropyl-benzene (500 mg, 2.04 mmol) in THF (10 mL) was added n-BuLi (2.5 M, 1.22 mL) at -78 °C under N2. The mixture was stirred for 0.5 h, then trimethyl borate (635.86 mg, 6.12 mmol, 691.16 uL) was added dropwise to the mixture. The mixture was stirred at -78 °C for 0.5 h. The reaction was quenched by ice water (10 mL) slowly, adjusted to pH = 5 with 6 N HCl. The resulting mixture was extracted with ethyl acetate (5 mL x 2). The combined organic phase was washed with brine (10 mL), dried over anhydrous Na2SO4, filtered and concentrated in vacuum. The residue was purified by flash silica gel chromatography (ISCO®; 12 g SepaFlash® Silica Flash Column, Eluent of 0~30% Ethyl acetate/Petroleum gradient @ 60 mL/min). (2-ethyl-3-fluoro-5-isopropyl-phenyl)boronic acid (350 mg, 1.67 mmol, 81.69% yield) was obtained as yellow oil.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.13-6.78 (m, 2H), 2.86 (qd, J = 7.0, 13.9 Hz, 1H), 2.72 (q, J = 7.1 Hz, 1H), 2.62 (s, 1H), 1.23 (d, J = 7.0 Hz, 6H), 1.18-1.08 (m, 3H). Preparation of ethyl 2-bromo-2-(3,4-difluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran- 4-yl)phenyl)acetate Step 1: 6-bromo-2,3-difluorophenol
[0842] To a mixture of 2,3-difluorophenol (10.0 g, 76.9 mmol) and isopropylamine (5.07 g, 84.6 mmol) in DCM (50 mL) was added NBS (13.69 g, 76.9 mmol) portionwise at -30°C. The mixture was stirred at that temperature for 30 min and then allowed to reach rt. The solvent was diluted with aq. HCl (1N, 50 mL) and extracted with DCM (3x 50 mL). The combined organic layer was dried over sodium sulfate, filtered, concentrated in vacuo and the residue was purified
by silica gel chromatography (EtOAc/pet ether =5%~30%) to give the desired product 6-bromo- 2,3-difluorophenol (6.1 g) as a colorless oil. Yield 38% (ESI 209 [M+H] +). Step 2: 1-bromo-3,4-difluoro-5-iodo-2-methoxybenzene
[0843] A mixture of 6-bromo-2,3-difluorophenol (6.0 g, 28.9 mmol), KI (7.18 g, 43.3 mmol) and I2 (10.99 g, 43.3 mmol) in H2O (30 mL) was stirred at 0°C for 10 min, then added a solution of NaOH (2.3g, 57.7 mmol) in H
2O (30mL) at 0°C. The mixture was stirred at room temperature for 8 hours, then diluted with aq. HCl (1N) and extracted with EtOAc. The combined organic layer was removed under vacuum, the residue was diluted with DMF (60 mL), then MeI (6.12 g, 43.1 mmol) and K2CO3 (11.0 g, 86.2 mmol) was added. The mixture was stirred at room temperature for 16 hours, then diluted with aq. HCl (1N) and extracted with EtOAc. The combined organic layer was removed under vacuum and the residue and purified by silica gel chromatography (EtOAc/pet ether = 5%~30%) to give the desired product 1-bromo-3,4-difluoro- 5-iodo-2-methoxybenzene (6.8 g) as a colorless oil. Yield 67% (ESI 349 [M+H] +). Step 3: 4-(5-bromo-2,3-difluoro-4-methoxyphenyl)tetrahydro-2H-pyran-4-ol

[0844] To a solution of 1-bromo-3,4-difluoro-5-iodo-2-methoxybenzene (6.8 g, 19.5 mmol) in THF (40 mL) at -78°C, was added i-PrMgCl solution 2.0 M in THF (19 mL, 38 mmol) dropwise. The reaction was stirred at -78°C for 30 min, then tetrahydro-4H-pyran-4-one (3.9 g, 39 mmol) in THF (20 mL) was added and the reaction was stirred at -78°C for another 30 min. The reaction was quenched with H2O (60 mL) and extracted with EtOAc (60 mL × 3). The combined organic phase was washed with brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether/EtOAc = 0%~30%) to give the desired product 4-(5-bromo-2,3-difluoro-4-methoxyphenyl)tetrahydro-2H-pyran-4-ol (1.6 g) as a pale yellow oil. Yield 26% (ESI 323 [M+H] +). Step 4: 4-(5-bromo-2,3-difluoro-4-methoxyphenyl)-4-methyltetrahydro-2H-pyran

[0845] To a solution of TiCl4 (2.19 g, 11.6 mmol) in DCM (30 mL) at -55°C, was added Me2Zn (1M in hexanes, 11.6 mL,11.6 mmol) dropwise. The reaction was stirred at -78°C for 1 hour, then 4-(5-bromo-2,3-difluoro-4-methoxyphenyl)tetrahydro-2H-pyran-4-ol (1.5 g, 4.65 mmol) in THF (10 mL) was added at -78 °C. The reaction was stirred at room temperature under N
2 for 3 hours. Then the reaction was stirred at 55°C under N
2 for 1 hour. The reaction was quenched with H2O (60 mL) and extracted with EtOAc (60 mL × 3). The combined organic phase was washed with brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether/EtOAc = 0%~30%) to give the desired product 4-(5- bromo-2,3-difluoro-4-methoxyphenyl)-4-methyltetrahydro-2H-pyran (360mg) as a pale yellow oil. Yield 22% (ESI 321 [M+H]+). Step 5: ethyl 2-(3,4-difluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4- yl)phenyl)acetate

[0846] To a mixture of 4-(5-bromo-2,3-difluoro-4-methoxyphenyl)-4-methyltetrahydro-2H- pyran (360 mg, 1.12 mmol), Q-phos (43 mg, 0.23 mmol) and Pd2(dba)3 (55 mg, 0.23 mmol) in THF (10 mL), was added (2-ethoxy-2-oxoethyl)zinc(II) bromide (1 M in THF, 4.8 mL). The reaction mixture was stirred at 50 °C under N2 for 2 hours. The reaction was quenched with NaHCO3 (aq) and extracted with EtOAc (2 x 20 mL). The combined organic layer was concentrated in vacuo and the residue was purified by silica gel column (pet ether: EtOAc 2 : 1) to give the desired product ethyl 2-(3,4-difluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4- yl)phenyl)acetate (260 mg) as a red oil. Yield 70% (ESI 329 [M+H] +). Step 6: ethyl 2-bromo-2-(3,4-difluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4- yl)phenyl)acetate

[0847] To a solution of ethyl 2-(3,4-difluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4- yl)phenyl)acetate (260 mg, 0.79 mmol) in THF (20 mL) at -78°C, was added lithium diisopropylamide solution 2.0 M in THF/hexanes (0.8 mL, 1.6 mmol) dropwise. The reaction was stirred at -78°C for 30 min, then chlorotrimethylsilane (172 mg, 1.58 mmol) was added and the reaction was stirred at -78°C for another 30 min. Then a solution of NBS (283 mg, 1.58 mmol) in THF (10 mL) was added and the reaction was stirred at -78°C for 30 min. The reaction was quenched with H2O (10 mL), then extracted with EtOAc (20 mL × 3). The combined organic phase was washed with sat. NaHCO3 solution, brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (EtOAc/pet ether = 0%~30%) to give the desired product ethyl 2-bromo-2-(3,4-difluoro-2-methoxy-5-(4- methyltetrahydro-2H-pyran-4-yl)phenyl)acetate (276 mg) as a pale brown oil. Yield 85% ( ESI 407/409 [M+H]). Preparation of ethyl 2-bromo-2-(5-cyclopropyl-3,4-difluoro-2-methoxyphenyl)acetate Step 1: 1-bromo-3,4-difluoro-2-methoxybenzene

[0848] To a mixture of 6-bromo-2,3-difluorophenol (624 mg, 3.0 mmol), K2CO3 (1.24g, 9.0 mmol) and CH
3I (639 mg, 4.5 mmol) in DMF (8 mL) was stirred at RT under Ar atmophere overnight. The reaction mixture was poured into water (20 mL) and extracted with EtOAc (3 × 40 mL). The combined organic phase was washed with bine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether: EtOAc 10:1~5:1) to give the desired product 1-bromo-3,4-difluoro-2-methoxybenzene (600 mg). Yield 90% (ESI N/A). Step 2: ethyl 2-(3,4-difluoro-2-methoxyphenyl)acetate

[0849] To a mixture of 1-bromo-3,4-difluoro-2-methoxybenzene (490 mg, 2.2 mmol), Pd2(dba)3 (101 mg, 0.05 mmol) and Q-phos (80 mg, 0.05 mmol) in THF (5 mL) was added (2- tert-butoxy-2-oxoethyl)zinc(II) bromide solution 1 M in THF (7.0 mL, 7.0 mmol) at rt under Ar. The reaction was stirred at 60°C for 2 hours. The mixture was quenched with NaHCO3 solution, the solid was removed by filtration. The filtrate was concentrated in vacuo, and the residue was purified by silica gel column (pet ether: EtOAc 20:1~10:1) to give the desired product ethyl 2- (3,4-difluoro-2-methoxyphenyl)acetate as a pale red oil (237 mg). Yield 47% (ESI 231 (M + H) +). Step 3: ethyl 2-(5-bromo-3,4-difluoro-2-methoxyphenyl)acetate

[0850] To a mixture of ethyl 2-(3,4-difluoro-2-methoxyphenyl)acetate (115 mg, 0.5 mmol) in HOAc (0.5 mL) /conc. H2SO4 (0.15 mL) was added NBS (98 mg, 0.55 mmol) at 0°C, the mixture was warmed to rt and stirred for 10 hours. The mixture was poured onto ice, adjusted PH to 6~7 with 4N NaOH solution, then extracted with EtOAc (2x 20 mL). The organic phase was concentrated in vacuo, and the residue was purified by silica gel column (EtOAc :pet ether 35%) to give the desired product ethyl 2-(5-bromo-3,4-difluoro-2-methoxyphenyl)acetate as a yellow oil (107 mg). Yield 70%. (ESI 309.0 (M+H) +). Step 4: ethyl 2-(5-cyclopropyl-3,4-difluoro-2-methoxyphenyl)acetate
[0851] A mixture of ethyl 2-(5-bromo-3,4-difluoro-2-methoxyphenyl)acetate (100 mg, 0.33 mmol), cyclopropylboronic acid (42 mg, 0.49 mmol), Pd(OAc)
2 (10 mg, 0.05 mmol), PCy
3 (14 mg, 0.05 mmol) and K3PO4(14 mg, 0.05 mmol) in toluene/H2O (5 mL/ 1mL) was stirred at 80°C for 6 h. Solvent was removed in vacuo, and the residue was purified by silica gel column (pet ether: EtOAc 2:1) to give the desired product ethyl 2-(5-cyclopropyl-3,4-difluoro-2- methoxyphenyl)acetate as a yollew oil (75 mg). Yield 86 % (ESI 271(M + H) +). Step 5: ethyl 2-bromo-2-(5-cyclopropyl-3,4-difluoro-2-methoxyphenyl)acetate

[0852] To a solution of ethyl 2-(5-cyclopropyl-3,4-difluoro-2-methoxyphenyl)acetate (120 mg, 0.44 mmol) in THF (5 mL) at -78°C, was added lithium diisopropylamide solution 2.0 M in THF/hexanes (0.6 mL, 1.2 mmol) dropwise. The reaction was stirred at -78°C for 30 min, then chlorotrimethylsilane (120 mg, 1.2 mmol) was added and the reaction was stirred at -78°C for another 30 min. Then a solution of NBS (198 mg, 1.2 mmol) in THF (2 mL) was added and the reaction was stirred at -78°C for 1 hour. The reaction was quenched with NH4Cl solution and extracted with DCM (2x 20 mL). The organic phase was concentrated in vacuo, and the residue was purified by silica gel column (EtOAc/pet ether = 0%~30%) to give the desired product ethyl 2-bromo-2-(5-cyclopropyl-3,4-difluoro-2-methoxyphenyl)acetate (126 mg) as a pale brown oil. Yield 82% ( ESI 349 [M+H]) Preparation of ethyl 2-bromo-2-(5-(2,2-dimethyltetrahydrofuran-3-yl)-3-fluoro-2- methoxyphenyl)acetate Step 1: methyl 2-(3-fluoro-4-hydroxyphenyl)acetate

[0853] To a solution of 2-(3-fluoro-4-hydroxyphenyl)acetic acid (5.0 g, 29.39 mmol) in 50 mL of MeOH was added conc. H2SO4 (1 mL). The mixture was stirred at 60 °C for 16 h. The mixture was concentrated and dissolved in 50 mL of EtOAc. The mixture was washed with aq. NaHCO
3 solution, water, dried over anhydrous Na
2SO
4, filtered and concentrated to give the
desired product methyl 2-(3-fluoro-4-hydroxyphenyl)acetate (4.5 g) as a colorless oil. Yield 83% (ESI 185 (M+H) +). Step 2: methyl 2-(3-bromo-5-fluoro-4-hydroxyphenyl)acetate
[0854] To a solution of methyl 2-(3-fluoro-4-hydroxyphenyl)acetate (4.5 g, 24.43 mmol) in THF (45 mL) were added NBS (5.2 g, 29.22 mmol) and K2CO3 (3.54 g, 25.64 mmol). The mixture was stirred at rt overnight. The reaction mixture was diluted with H2O (50 mL) and extracted with EtOAc (3x 100 mL). The combined organic layer was washed with brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether: EtOAc 5:1) to give the desired product methyl 2-(3-bromo-5-fluoro-4- hydroxyphenyl)acetate as a pale yellow oil (6.05 g). Yield 94% (ESI 263 (M+H) +). Step 3: methyl 2-(3-bromo-5-fluoro-4-methoxyphenyl)acetate

[0855] To a solution of methyl 2-(3-bromo-5-fluoro-4-hydroxyphenyl)acetate (6.1 g, 23.1 mmol) in ACN (40 mL) were added MeI (4.9 g, 34.7 mmol) and K2CO3 (6.4 g, 46.2 mmol). The mixture was stirred at rt overnight. The reaction mixture was diluted with H2O (50 mL) and extracted with EtOAc (3x 100 mL). The combined organic layer was washed with brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether: EtOAc 5:1) to give the desired product methyl 2-(3-bromo-5-fluoro-4- methoxyphenyl)acetate as a white solid (5.1 g). Yield 80% (ESI 277 (M+H) +). Step 4: methyl 2-(3-bromo-5-fluoro-4-methoxyphenyl)-4-((tert- butyldimethylsilyl)oxy)butanoate

[0856] To a solution of methyl 2-(3-bromo-5-fluoro-4-methoxyphenyl)acetate (3 g, 10.83 mmol) in THF (30 mL) at -78°C, was added lithium diisopropylamide solution 2.0 M in THF/hexanes (8.1 mL, 16.2 mmol) dropwise. The reaction was stirred at -78°C for 30 min, then (2-bromoethoxy)(tert-butyl)dimethylsilane (5.1 g, 21.6 mmol) was added and the reaction was stirred at -78°C for 20 min. Then the reaction was stirred at RT for 18 hours. The reaction was quenched with H
2O (50 mL) and extracted with EtOAc (100 mL × 3). The combined organic phase was washed with sat. NaHCO
3 solution, brine, dried over Na
2SO
4, filtered and concentrated in vacuo. The residue was purified by silica gel column (EtOAc /pet ether = 10%~20%) to give the desired product methyl 2-(3-bromo-5-fluoro-4-methoxyphenyl)-4-((tert- butyldimethylsilyl)oxy)butanoate as a pale yellow oil (3.1 g, 94% yield) ESI 435 (M+H) +. Step 5: 3-(3-bromo-5-fluoro-4-methoxyphenyl)-5-((tert-butyldimethylsilyl)oxy)-2- methylpentan-2-ol

[0857] MeMgBr (3N in THF, 8.5 mL, 25.5 mmol) was added dropwise to a solution of methyl 2-(3-bromo-5-fluoro-4-methoxyphenyl)-4-((tert-butyldimethylsilyl)oxy)butanoate (3.1 g, 7.6 mmol) in THF (30 mL) at 0 °C. The mixture was stirred at room temperature for 18 hours, then quenched with aq. H2SO4 (1M) and extraced with EtOAc (50 mL x 2). The combined organic solvents were concentrated and the residue was purified by silica gel chromatography (EtOAc:pet ether = 10%~20%) to give the desired product 3-(3-bromo-5-fluoro-4- methoxyphenyl)-5-((tert-butyldimethylsilyl)oxy)-2-methylpentan-2-ol (1.1 g) as a colorless oil. Yield 33% (ESI 435 (M+H) +). Step 6: 3-(3-bromo-5-fluoro-4-methoxyphenyl)-2,2-dimethyltetrahydrofuran

[0858] 3-(3-bromo-5-fluoro-4-methoxyphenyl)-5-((tert-butyldimethylsilyl)oxy)-2- methylpentan-2-ol (472 mg, 1.5 mmol) was treated with TBAF (1M in THF, 10 mL) at rt for 4
hours, then added MsCl (422 mg, 3.7 mmol) and TEA (522 mg, 5.2 mmol). The reaction mixture was stirred at rt for 18 hours. The solvent was concentrated and the residue was purified by silica gel chromatography (EtOAc:pet ether = 10%~30%) to give the desired product 3-(3-bromo-5- fluoro-4-methoxyphenyl)-2,2-dimethyltetrahydrofuran (110 mg) as a colorless oil. Yield 33% (ESI 303 (M+H) +). Step 7: ethyl 2-(5-(2,2-dimethyltetrahydrofuran-3-yl)-3-fluoro-2-methoxyphenyl)acetate
[0859] To a mixture of 3-(3-bromo-5-fluoro-4-methoxyphenyl)-2,2-dimethyltetrahydrofuran (443 mg, 1.5 mmol), Q-phos(53 mg, 0.075 mmol) and Pd
2(dba)
3 (69 mg, 0.075 mmol) in THF (10 mL), was added (2-ethoxy-2-oxoethyl)zinc(II) bromide (1 M in THF, 4.4 mL). The reaction mixture was stirred at 50 °C under N2 for 1 hour. The reaction was quenched with NaHCO3 (aq) and extracted with EtOAc (2 x 20 mL). The combined organic layer was concentrated in vacuo and the residue was purified by silica gel column (pet ether: EtOAc 5 : 1) to give the desired product ethyl 2-(5-(2,2-dimethyltetrahydrofuran-3-yl)-3-fluoro-2-methoxyphenyl)acetate (415 mg, 1.3 mmol) as a red oil. Yield 87% (ESI 311 [M+H] +). Step 8: ethyl 2-bromo-2-(5-(2,2-dimethyltetrahydrofuran-3-yl)-3-fluoro-2- methoxyphenyl)acetate

[0860] To a solution of ethyl 2-(5-(2,2-dimethyltetrahydrofuran-3-yl)-3-fluoro-2- methoxyphenyl)acetate (415 mg, 1.3 mmol) in THF (10 mL) at -78°C, was added lithium diisopropylamide solution 2.0 M in THF/hexanes (1.5 mL, 3.0 mmol) dropwise. The reaction was stirred at -78°C for 30 min, then chlorotrimethylsilane (319 mg, 2.94 mmol) was added and the reaction was stirred at -78°C for another 30 min. Then a solution of NBS (523 mg, 2.94 mmol) in THF (10 mL) was added and the reaction was stirred at -78°C for 30 min. The reaction was quenched with H
2O (10 mL) and extracted with EtOAc (20 mL × 3). The combined organic phase
was washed with sat. NaHCO
3 solution, brine, dried over Na
2SO
4, filtered and concentrated in vacuo. The residue was purified by silica gel column (EtOAc/pet ether = 0%~20%) to give the desired product ethyl 2-bromo-2-(5-(2,2-dimethyltetrahydrofuran-3-yl)-3-fluoro-2- methoxyphenyl)acetate as a pale brown oil (465 mg, 92% yield). ESI 389/341 (M+H) +. Preparation of ethyl 2-bromo-2-(2-fluoro-3-isopropyl-6-methoxyphenyl)acetate Step 1: ethyl 2-(3-bromo-2-fluoro-6-methoxyphenyl)acetate

[0861] To a solution of 1-bromo-2-fluoro-4-methoxybenzene (3.0 g, 14.6 mmol) in THF (50 mL) at -78°C, was added lithium diisopropylamide solution 2.0 M in THF/hexanes (11 mL, 22.0 mmol) dropwise. The reaction was stirred at -78°C for 1 hour, then CuI (285 mg, 1.5 mmol) was added and the reaction was stirred at -78°C for another 1 hour. Then a solution of ethyl 2- bromoacetate (3.7 g, 22.0 mmol) in THF (20 mL) was added. The reaction was warmed to 0 °C slowly and stirred at 0 °C for 2 hours. The reaction was quenched with H2O (20 mL), then extracted with EtOAc (50 mL × 3). The combined organic phase was washed with Sat. NaHCO3 solution, brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether/EtOAc = 0%~10%) to give the desired product ethyl 2-(3-bromo- 2-fluoro-6-methoxyphenyl)acetate as a colorless oil (1.85 g, 44% yield) ESI 291/293 (M+H) +. Step 2: ethyl 2-(2-fluoro-6-methoxy-3-(prop-1-en-2-yl)phenyl)acetate

[0862] A mixture of ethyl 2-(3-bromo-2-fluoro-6-methoxyphenyl)acetate (1.85 g, 6.36 mmol), 4,4,5,5-tetramethyl-2-(prop-1-en-2-yl)-1,3,2-dioxaborolane (1.60 g, 9.52 mmol), Pd(dppf)Cl2 (462 mg, 0.64 mmol) and K2CO3 (2.62 g, 19.0 mmol) in 1,4-dioxane (40 mL) and H2O (8 mL) was stirred at 80°C overnight under Ar. The reaction mixture was poured into water (20 mL), then extracted with EtOAc (2 × 50 mL). The combined organic phase was washed with bine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether: EtOAc 10:1~5:1) to give the desired product ethyl 2-(2-fluoro-6-methoxy-
3-(prop-1-en-2-yl)phenyl)acetate as a pale yellow oil (650 mg, 2.58 mmol). Yield 41% (ESI 253 (M+H) +). Step 3: ethyl 2-(2-fluoro-3-isopropyl-6-methoxyphenyl)acetate

[0863] A mixture of ethyl 2-(2-fluoro-6-methoxy-3-(prop-1-en-2-yl)phenyl)acetate (780 mg, 3.10 mmol) and Pd/C (78 mg, 10%) in EtOH (30 mL) was stirred at 25 °C under H2 for 16 hours. Then the solid was removed by filtration and the filtrate was concentrated in vacuo to give the desired product ethyl 2-(2-fluoro-3-isopropyl-6-methoxyphenyl)acetate (750 mg, 2.95 mmol) as a light yellow oil. Yield 95% (ESI 255[M+H] +). Step 4: ethyl 2-bromo-2-(2-fluoro-3-isopropyl-6-methoxyphenyl)acetate

[0864] To a solution of ethyl ethyl 2-(2-fluoro-3-isopropyl-6-methoxyphenyl)acetate (750 mg, 2.95 mmol) in THF (20 mL) at -78°C, was added lithium diisopropylamide solution 2.0 M in THF/hexanes (3.6 mL, 7.2 mmol) dropwise. The reaction was stirred at -78°C for 30 min, then chlorotrimethylsilane (0.78 g, 7.18 mmol) was added and the reaction was stirred at -78°C for another 30 min. Then a solution of NBS (1.28 g, 7.18 mmol) in THF (10 mL) was added and the reaction was stirred at -78°C for 30 min. The reaction was quenched with H2O (10 mL), then extracted with EtOAc (20 mL × 3). The combined organic phase was washed with Sat. NaHCO3 solution, brine, dried over Na
2SO
4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether/EtOAc = 0%~10%) to give the desired product ethyl 2-bromo-2- (2-fluoro-3-isopropyl-6-methoxyphenyl)acetate as a pale brown oil (809 mg, 82% yield) ESI 333/335 (M+H) +. Preparation of ethyl 2-bromo-2-(3-fluoro-2-methoxy-5-((R)-tetrahydrofuran-2- yl)phenyl)acetate Step 1: ethyl 2-(3-fluoro-2-methoxyphenyl)acetate
[0865] (2-ethoxy-2-oxoethyl)zinc(II) bromide (300 mL, 150 mmol, 0.5 N in THF) was added to a solution of 1-bromo-3-fluoro-2-methoxybenzene (12.5 g, 60 mmol), Pd2(dba)3 (1.65 g, 1.8 mmol) and Q-phos (1.25 g, 1.8 mmol) in 25 mL of THF under argon atmosphere. The mixture was stirred at 60 °C for 2 hours, concentrated and purified by silica gel column (EtOAc:pet ether = 0%~10%) to give the desired product ethyl 2-(3-fluoro-2-methoxyphenyl)acetate as orange oil (7.1 g). Yield 56% (ESI 213.3 (M+H) +). Step 2: ethyl 2-(5-bromo-3-fluoro-2-methoxyphenyl)acetate

[0866] NBS (6.6 g, 36.9 mmol) was added to a solution of ethyl 2-(3-fluoro-2- methoxyphenyl)acetate (7.1 g, 33.5 mmol) in HOAc (55 mL) and concentrated H2SO4 (9 mL) at 0 °C. The mixture was stirred at 0 °C for 5 hours, quenched with water (20 mL) and extracted with EtOAc (20 mL x3). The combined organic solvents were concentrated and purified by silica gel column (EtOAc:pet ether = 0%~10%) to give the desired product ethyl 2-(5-bromo-3-fluoro- 2-methoxyphenyl)acetate as orange oil (5.5 g). Yield 57% (ESI 291.0 (M+H) +). Step 3: ethyl 2-(5-(2,5-dihydrofuran-2-yl)-3-fluoro-2-methoxyphenyl)acetate

[0867] A mixture of ethyl 2-(5-bromo-3-fluoro-2-methoxyphenyl)acetate (2.9 g, 10 mmol), 2,3-dihydrofuran (3.5 g, 50 mmol), Pd(OAc)2 (224 mg, 1 mmol), PPh3 (262 mg, 1 mmol) and K2CO3 (2.76 g, 20 mmol) in 40 mL of DMF was stirred at 90 °C for 1 h under nitrogen atmosphere. The mixture was quenched with water (20 mL) and extracted with EtOAc (20 mL x3). The combined organic solvents were concentrated and purified by silica gel column
(EtOAc:pet ether = 0%~10%) to give the desired product ethyl 2-(5-(2,5-dihydrofuran-2-yl)-3- fluoro-2-methoxyphenyl)acetate as orange oil (2.7 g). Yield 53% (ESI 281.0 (M+H)
+). Step 4: ethyl 2-(3-fluoro-2-methoxy-5-(tetrahydrofuran-2-yl)phenyl)acetate
[0868] A mixture of ethyl 2-(5-(2,5-dihydrofuran-2-yl)-3-fluoro-2-methoxyphenyl)acetate (2.7 g, 9.6 mmol) and Pd/C (300 mg, 10% on activated carbon) in 40 mL of MeOH was stirred at room temperature under H2 atmosphere for 24 hours. The mixture was filtered, concentrated and purified by by Prep-HPLC A (30-60% MeCN) to give 1.9 g of racemic ethyl 2-(3-fluoro-2- methoxy-5-(tetrahydrofuran-2-yl)phenyl)acetate. Yield 70% (ESI 283.1 (M+H) +). The racemate was separated by Prep Chiral SFC B to give 800 mg of each isomer. The first eluting isomer was arbitrarily assigned as ethyl (R)-2-(3-fluoro-2-methoxy-5-(tetrahydrofuran-2-yl)phenyl)acetate and used in the next step. Step 5: ethyl 2-bromo-2-(3-fluoro-2-methoxy-5-((R)-tetrahydrofuran-2-yl)phenyl)acetate

[0869] LDA (1.2 mL, 2.3 mmol, 2N in THF) was added to a mixture of ethyl (R)-2-(3- fluoro-2-methoxy-5-(tetrahydrofuran-2-yl)phenyl)acetate (300 mg, 1.06 mmol) in 10 mL of THF at -78 °C. After stirring for 30 min, TMSCl (250 mg, 2.3 mmol) was added at -78 °C. After stirring for additional 30 min, NBS (409 mg, 2.3 mmol). The mixture was stirred for 1 h at - 78 °C, quenched with water (10 mL) and extracted with EtOAc (15 mL x 3). The combined organic solvents were concentrated and purified by silica gel chromatography (EtOAc: PE = 0%~25%) to give the desired product ethyl 2-bromo-2-(3-fluoro-2-methoxy-5-((R)- tetrahydrofuran-2-yl)phenyl)acetate (210 mg). Yield 55% (ESI 361.2 (M+H) +).
Preparation of ethyl 2-(5-(cyclopropylmethyl)-3-fluoro-2-methoxyphenyl)acetate and ethyl 2-(5-(but-3-en-1-yl)-3-fluoro-2-methoxyphenyl)acetate

[0870] A mixture of ethyl 2-(5-bromo-3-fluoro-2-methoxyphenyl)acetate (873 mg, 3.0 mmol), Potassium cyclopyropylmethyltrifluoroborate (583 mg, 3.6 mmol), Pd2(dba)3 (138 mg, 0.15 mmol), RuPhos (140 mg, 0.3 mmol) and K
2CO
3 (1.24 g, 9.0 mmol) in toluene (40 mL) and H
2O (8 mL) was stirred at 100°C overnight under Ar. The reaction mixture was poured into water (20 mL), then extracted with EtOAc (2 × 50 mL). The combined organic phase was washed with bine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether: EtOAc 10:1~5:1) to give a mixture of ethyl 2-(5-(cyclopropylmethyl)-3- fluoro-2-methoxyphenyl)acetate and ethyl 2-(5-(but-3-en-1-yl)-3-fluoro-2-methoxyphenyl)acetate as a pale yellow oil (154 mg, ratio ~1:1, ESI 267 (M+H) +). Preparation of ethyl 2-bromo-2-(5-cyclopropyl-3-fluoro-2-methoxyphenyl)acetate Step 1: ethyl 2-(5-cyclopropyl-3-fluoro-2-methoxyphenyl)acetate
[0871] A mixture of ethyl 2-(5-bromo-3-fluoro-2-methoxyphenyl)acetate (1.2 g, 4.1 mmol), cyclopropylboronic acid (860 mg, 10 mmol), Pd(OAc)
2 (90 mg, 0.4 mmol), PCy
3 (112 mg, 0.4 mmol) and K3PO4 (1.7 g, 8.2 mmol) in 25 mL of toluene was stirred at 100 °C for 8 hours. The mixture was concentrated and purified by silica gel chromatography (pet ether: EtOAc = 0%~20%) to give the desired product ethyl 2-(5-cyclopropyl-3-fluoro-2-methoxyphenyl)acetate (510 mg). Yield 49% (ESI 253.3 (M+H) +). Step 2: ethyl 2-bromo-2-(5-cyclopropyl-3-fluoro-2-methoxyphenyl)acetate

[0872] LDA (2.3 mL, 4.6 mmol, 2N in THF) was added to a mixture of ethyl 2-(5- cyclopropyl-3-fluoro-2-methoxyphenyl)acetate (500 mg, 2.1 mmol) in 3 mL of THF at -78 °C. After stirring for 30 min, TMSCl (500 mg, 4.6 mmol) was added at -78 °C. After stirring for additional 30 min, NBS (819 mg, 4.6 mmol). The mixture was stirred for 1 h at -78 °C, quenched with water and extracted with EtOAc (15 mL X 3). The combined organic solvents were concentrated and purified silica gel chromatography (EtOAc: PE = 0%~25%) to give the desired product ethyl 2-bromo-2-(5-cyclopropyl-3-fluoro-2-methoxyphenyl)acetate as orange oil (520 mg). Yield 75% (ESI 331.2 (M+H) +). Preparation of ethyl 2-bromo-2-(5-isopropyl-2-methoxy-3-(trifluoromethyl)phenyl)acetate Step 1: 1-bromo-2-methoxy-3-(trifluoromethyl)benzene
[0873] MeI (1.28 g, 9 mmol) was added to a mixture of 2-bromo-6-(trifluoromethyl)phenol (1.45 g, 6 mmol) and K
2CO
3 (1.66 g, 12 mmol) in 5 mL of DMF and 15 mL of THF. The mixture was stirred at room temperature for 16 hours, quenched with water and extracted with EtOAc (15 mL x3). The combined organic solvents were concentrated and purified by silica gel chromatography (EtOAc:pet ether = 0%~5%) to give the desired product 1-bromo-2-methoxy-3- (trifluoromethyl)benzene as colorless oil (1.31 g). Yield 86% (ESI 255.3 (M+H) +). Step 2: ethyl 2-(2-methoxy-3-(trifluoromethyl)phenyl)acetate
[0874] (2-ethoxy-2-oxoethyl)zinc(II) bromide (15 mL, 15 mmol, 1 N in THF) was added to a solution of 1-bromo-2-methoxy-3-(trifluoromethyl)benzene (1.28 g, 5 mmol), Pd2(dba)3 (73 mg, 0.08 mmol) and Q-phos (57 mg, 0.08 mmol) in 15 mL of THF under argon atmosphere. The
mixture was stirred at 60 °C for 2 hours, concentrated and purified by silica gel column (EtOAc:pet ether = 0%~10%) to give the desired product ethyl 2-(2-methoxy-3- (trifluoromethyl)phenyl)acetate as orange oil (1.21 g). Yield 92% (ESI 263.3 (M+H)
+). Step 3: ethyl 2-(5-bromo-2-methoxy-3-(trifluoromethyl)phenyl)acetate
[0875] NBS (427 mg, 2.4 mmol) was added to a solution of ethyl 2-(2-methoxy-3- (trifluoromethyl)phenyl)acetate (524 mg, 2 mmol) in HOAc (4 mL) and concentrated H2SO4 (2 mL) at 0 °C. The mixture was stirred at room temperature for 4 hours, quenched with water ( 10 mL) and extracted with EtOAc (10 mLx3). The combined organic solvents were concentrated and purified by silica gel column (EtOAc:pet ether = 0%~10%) to give the desired product ethyl 2-(5- bromo-2-methoxy-3-(trifluoromethyl)phenyl)acetate as orange oil (286 mg). Yield 42% (ESI 340.9 (M+H)
+). Step 4: ethyl 2-(2-methoxy-5-(prop-1-en-2-yl)-3-(trifluoromethyl)phenyl)acetate
[0876] A mixture of 4,4,5,5-tetramethyl-2-(prop-1-en-2-yl)-1,3,2-dioxaborolane (1 g, 6 mmol), ethyl 2-(5-bromo-2-methoxy-3-(trifluoromethyl)phenyl)acetate (1 g, 3 mmol), K
2CO
3 (830 mg, 6 mmol) and Pd(dppf)Cl2 (110 mg, 0.15 mmol) in 15 mL of dioxane and 3 mL of H2O was stirred at 90 °C for 2 hours. The mixture was concentrated and silica gel chromatography (EtOAc:pet ether = 0%~25%) to give the desired product ethyl 2-(2-methoxy-5-(prop-1-en-2-yl)- 3-(trifluoromethyl)phenyl)acetate as orange oil (861 mg). Yield 95% (ESI 303.2 (M+H) +). Step 5: ethyl 2-(5-isopropyl-2-methoxy-3-(trifluoromethyl)phenyl)acetate

[0877] A mixture of ethyl 2-(2-methoxy-5-(prop-1-en-2-yl)-3- (trifluoromethyl)phenyl)acetate (861 mg, 2.85 mmol) and Pd/C (170 mg, 10% on activated carbon) in 12 mL of MeOH was stirred at 55 °C under H2 atmosphere for 24 hours. The mixture was filtered, concentrated and purified by silica gel chromatography (EtOAc:pet ether = 0%~25%) to give the desired product ethyl 2-(5-isopropyl-2-methoxy-3- (trifluoromethyl)phenyl)acetate as orange oil (850 mg). Yield 98% (ESI 305.2 (M+H) +). Step 6: ethyl 2-bromo-2-(5-isopropyl-2-methoxy-3-(trifluoromethyl)phenyl)acetate

[0878] LDA (1.6 mL, 3.2 mmol, 2N in THF) was added to a mixture of ethyl 2-(5-isopropyl- 2-methoxy-3-(trifluoromethyl)phenyl)acetate (487 mg, 1.6 mmol) in 5 mL of THF at -78 °C. After stirring for 30 min, TMSCl (348 mg, 3.2 mmol) was added at -78 °C. After stirring for additional 30 min, NBS (570 mg, 3.2 mmol). The mixture was stirred for 1 h at -78 °C, quenched with water and extracted with EtOAc (15 mL x 3). The combined organic solvents were concentrated and purified silica gel chromatography (EtOAc:pet ether = 0%~25%) to give the desired product ethyl 2-bromo-2-(5-isopropyl-2-methoxy-3-(trifluoromethyl)phenyl)acetate (586 mg). Yield 83% (ESI 384.9 (M+H) +). Preparation of ethyl 2-bromo-2-(3-chloro-5-isopropyl-2-methoxyphenyl)acetate Step 1: ethyl 2-(3-chloro-2-methoxyphenyl)acetate
[0879] (2-ethoxy-2-oxoethyl)zinc(II) bromide (27 mL, 27 mmol, 1 N in THF) was added to a solution of 1-bromo-3-chloro-2-methoxybenzene (2 g, 9 mmol), Pd
2(dba)
3 (412 mg, 0.45 mmol) and Q-phos (320 mg, 0.45 mmol) in 5 mL of THF under argon atmosphere. The mixture was stirred at 60 °C for 16 hours, concentrated and purified by silica gel column (EtOAc:pet ether = 0%~10%) to give the desired product ethyl 2-(3-chloro-2-methoxyphenyl)acetate as orange oil (1.5 g). Yield 73% (ESI 229.1 (M+H) +). Step 2: ethyl 2-(5-bromo-3-chloro-2-methoxyphenyl)acetate
[0880] NBS (85 mg, 0.48 mmol) was added to a solution of ethyl 2-(3-chloro-2- methoxyphenyl)acetate (100 mg, 0.44 mmol) in HOAc (3 mL) and concentrated H
2SO
4 (0.5 mL) at 0 °C. The mixture was stirred at 0 °C for 4 hours, quenched water (10 mL) and extracted with EtOAc (10 mLx3). The combined organic solvents were concentrated and purified by silica gel column (EtOAc:pet ether = 0%~20%) to give the desired product ethyl 2-(5-bromo-3-chloro-2- methoxyphenyl)acetate as orange oil (110 mg). Yield 82% (ESI 307 (M+H) +). Step 3: ethyl 2-(3-chloro-2-methoxy-5-(prop-1-en-2-yl)phenyl)acetate
[0881] A mixture of 4,4,5,5-tetramethyl-2-(prop-1-en-2-yl)-1,3,2-dioxaborolane (1 g, 6 mmol), ethyl 2-(5-bromo-3-chloro-2-methoxyphenyl)acetate (913 mg, 3 mmol), K
2CO
3 (826 mg, 6 mmol) and Pd(dppf)Cl
2 (110 mg, 0.15 mmol) in 15 mL of dioxane and 3 mL of H
2O was stirred at 80 °C for 2 hours. The mixture was concentrated and purified by silica gel chromatography (EtOAc:pet ether = 0%~20%) to give the desired product ethyl 2-(3-chloro-2-methoxy-5-(prop-1- en-2-yl)phenyl)acetate as orange oil (780 mg). Yield 97% (ESI 269.2 (M+H) +). Step 4: ethyl 2-(3-chloro-5-isopropyl-2-methoxyphenyl)acetate
[0882] A mixture of ethyl 2-(3-chloro-2-methoxy-5-(prop-1-en-2-yl)phenyl)acetate (780 mg, 2.9 mmol) and Pd/C (78 mg, 10% on activated carbon) in 5 mL of MeOH was stirred at room temperature under H2 atmosphere for 16 hours. The mixture was filtered, concentrated and purified by silica gel chromatography (EtOAc:pet ether = 0%~25%) to give the desired product ethyl 2-(3-chloro-5-isopropyl-2-methoxyphenyl)acetate as orange oil (720 mg). Yield 92% (ESI 271.2 (M+H) +). Step 5: ethyl 2-bromo-2-(3-chloro-5-isopropyl-2-methoxyphenyl)acetate

[0883] LDA (5.4 mL, 2.7 mmol, 2N in THF) was added to a mixture of ethyl 2-(3-chloro-5- isopropyl-2-methoxyphenyl)acetate (660 mg, 2.44 mmol) in 3 mL of THF at -78 °C. After stirring for 30 min, TMSCl (583 mg, 5.4 mmol) was added at -78 °C. After stirring for additional 30 min, NBS (961 mg, 5.4 mmol) was added to the reaction solution. The mixture was stirred at - 78 °C for 1 h, quenched with water (10 mL) and extracted with EtOAc (15 mL x 3). The combined organic solvents were concentrated and purified silica gel chromatography (EtOAc: PE = 0%~20%) to give the desired product ethyl 2-bromo-2-(3-chloro-5-isopropyl-2- methoxyphenyl)acetate (610 mg). Yield 72% (ESI 349.0 (M+H) +). Preparation of ethyl 2-bromo-2-(3-fluoro-5-isobutyl-2-methoxyphenyl)acetate Step 1: ethyl 2-(5-bromo-3-fluoro-2-methoxyphenyl)acetate

[0884] NBS (1.45 g, 8.17 mmol) was added to a solution of ethyl 2-(3-fluoro-2- methoxyphenyl)acetate (1.7 g, 8 mmol) in HOAc (30 mL) and concentrated H2SO4 (6 mL) at 0 °C. The mixture was stirred at room temperature for 16 hours, quenched with water (30 mL) and extracted with EtOAc (30 mL x3). The combined organic solvents were concentrated and purified by silica gel column (EtOAc:pet ether = 0%~20%) to give the desired product ethyl 2-(5- bromo-3-fluoro-2-methoxyphenyl)acetateas orange oil (1.8 g). Yield 78% (ESI 291 (M+H) +). Step 2: ethyl 2-(3-fluoro-2-methoxy-5-(2-methylprop-1-en-1-yl)phenyl)acetate
[0885] A mixture of 4,4,5,5-tetramethyl-2-(2-methylprop-1-en-1-yl)-1,3,2-dioxaborolane (1.5 g, 8.6 mmol), ethyl 2-(5-bromo-3-fluoro-2-methoxyphenyl)acetate (1 g, 3.4 mmol), K
2CO
3 (940 mg, 6.8 mmol) and Pd(dppf)Cl
2 (124 mg, 0.17 mmol) in 10 mL of dioxane and 2 mL of H
2O was stirred at 80 °C for 3 hours. The mixture was concentrated and purified by silica gel chromatography (EtOAc:pet ether = 0%~20%) to give the desired product ethyl 2-(3-fluoro-2- methoxy-5-(2-methylprop-1-en-1-yl)phenyl)acetate (1.1 g). Yield 99% (ESI 267.2 (M+H) +). Step 3: ethyl 2-(3-fluoro-5-isobutyl-2-methoxyphenyl)acetate
[0886] A mixture of ethyl 2-(3-fluoro-2-methoxy-5-(2-methylprop-1-en-1-yl)phenyl)acetate (1.1 g, 4.1 mmol) and Pd/C (110 mg, 10% on activated carbon) in 10 mL of MeOH was stirred at room temperature under H2 atmosphere for 16 hours. The mixture was filtered, concentrated and purified by silica gel chromatography (EtOAc:pet ether = 0%~25%) to give the desired product ethyl 2-(3-fluoro-5-isobutyl-2-methoxyphenyl)acetate as orange oil (950 mg). Yield 86% (ESI 269.2 (M+H) +). Step 4: ethyl 2-bromo-2-(3-fluoro-5-isobutyl-2-methoxyphenyl)acetate

[0887] LDA (3.9 mL, 7.8 mmol, 2N in THF) was added to a mixture of ethyl 2-(3-fluoro-5- isobutyl-2-methoxyphenyl)acetate (950 mg, 3.5 mmol) in 4 mL of THF at -78 °C. After stirring for 30 min, TMSCl (836 mg, 7.8 mmol) was added at -78 °C. After stirring for additional 30 min, NBS (1.4 g, 7.8 mmol) was added to the reaction solution. The mixture was stirred for 1 h at - 78 °C, quenched with water (10 mL) and extracted with EtOAc (15 mL x 3). The combined organic solvents were concentrated and purified by silica gel chromatography (EtOAc:pet ether = 0%~20%) to give the desired product ethyl 2-bromo-2-(3-fluoro-5-isobutyl-2- methoxyphenyl)acetate (810 mg). Yield 67% (ESI 347.0 (M+H) +). Preparation of ethyl 2-bromo-2-(3-fluoro-5-isopentyl-2-methoxyphenyl)acetate Step 1: ethyl 2-(3-fluoro-5-isopentyl-2-methoxyphenyl)acetate

[0888] A mixture of isopentylboronic acid (719 mg, 6.2 mmol), ethyl 2-(5-bromo-3-fluoro-2- methoxyphenyl)acetate (900 mg, 3.1 mmol), K3PO4 (1.34 g, 6.2 mmol), PCy3 (84 mg, 0.3 mmol) and Pd(OAc)
2 (67 mg, 0.3 mmol) in 10 mL of toluene and 2 mL of H2O was stirred at 80 °C for 8 hours. The mixture was concentrated and purified by silica gel chromatography (EtOAc:pet ether = 0%~20%) to give the desired product ethyl 2-(3-fluoro-5-isopentyl-2- methoxyphenyl)acetate (450 mg). Yield 51% (ESI 283.2 (M+H) +). Step 2: ethyl 2-bromo-2-(3-fluoro-5-isopentyl-2-methoxyphenyl)acetate
[0889] LDA (1.25 mL, 2.5 mmol, 2N in THF) was added to a solution of ethyl 2-(3-fluoro-5- isopentyl-2-methoxyphenyl)acetate (320 mg, 1.13 mmol) in 4 mL of THF at -78 °C. After stirring for 30 min, TMSCl (286 mg, 2.61 mmol) was added at -78 °C. After stirring for additional 30 min, NBS (465 mg, 2.61 mmol) was added to the reaction solution. The mixture was stirred for 1 h at -78 °C, quenched with water (10 mL) and extracted with EtOAc (15 mL x 3). The combined organic solvents were concentrated and purified by silica gel chromatography (EtOAc: PE = 0%~20%) to give the desired product ethyl 2-bromo-2-(3-fluoro-5-isopentyl-2- methoxyphenyl)acetate (405 mg). Yield 95% (ESI 361.0 (M+H) +). Preparation of ethyl 2-bromo-2-(3-fluoro-5-(2-hydroxypropan-2-yl)-2- methoxyphenyl)acetate Step 1: ethyl 2-(3-fluoro-5-(2-hydroxypropan-2-yl)-2-methoxyphenyl)acetate

[0890] To a mixture of 2-(3-bromo-5-fluoro-4-methoxyphenyl)propan-2-ol (2.0 g, 7.6 mmol), Pd2(dba)3 (276 mg, 0.38 mmol) and Q-phos (345 mg, 0.38 mmol) in THF (20 mL) was added (2-tert-butoxy-2-oxoethyl)zinc(II) bromide solution 1 M in THF (19.5 mL, 19.5 mmol) at rt under Ar. The reaction was stirred at 50°C for 2 hours. The mixture was quenched with NaHCO3 solution, the solid was removed by filtration. The filtrate was concentrated in vacuo, and the residue was purified by silica gel column (pet ether: EtOAc 3:1) to give the desired product ethyl 2-(3-fluoro-2-methoxy-5-(2-methoxypropan-2-yl)phenyl)acetate as a pale red oil (1.7 g, 6.29 mmol). Yield 83% (ESI 271.0 (M+H) +). Step 2: ethyl 2-bromo-2-(3-fluoro-5-(2-hydroxypropan-2-yl)-2-methoxyphenyl)acetate
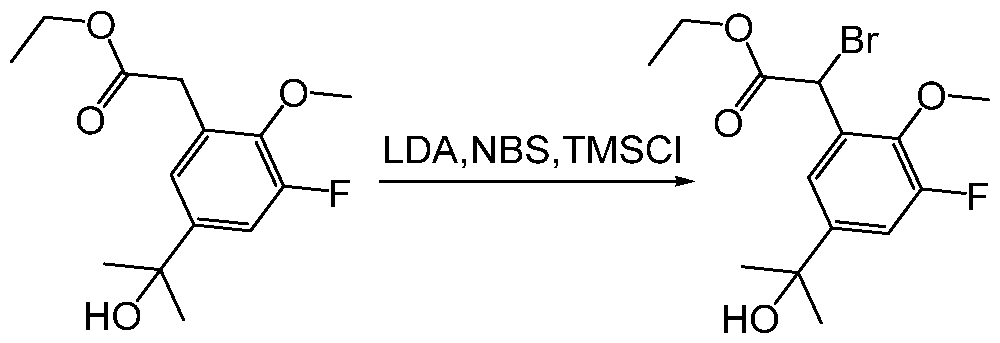
[0891] To a solution of ethyl 2-(3-fluoro-5-(2-hydroxypropan-2-yl)-2-methoxyphenyl)acetate (500 mg, 1.85 mmol) in THF (10 mL) at -78°C, was added lithium diisopropylamide solution 2.0 M in THF/hexanes (1.9 mL, 3.8 mmol) dropwise. The reaction was stirred at -78°C for 30 min, then chlorotrimethylsilane (413 mg, 3.8 mmol) was added and the reaction was stirred at - 78°C for another 30 min. Then a solution of NBS (676 mg, 3.8 mmol) in THF (10 mL) was added and the reaction was stirred at -78°C for 30min. The reaction was quenched with H2O (10 mL) and extracted with EtOAc(2x 20mL). The combined organic phase was washed with sat. NaHCO3 solution, brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether: EtOAc 3:1) to give the desired product ethyl 2-bromo-2- (3-fluoro-5-(2-hydroxypropan-2-yl)-2-methoxyphenyl)acetate as a yellow oil (606 mg). Yield 94% (ESI 349 (M+H) +). Preparation of ethyl 2-bromo-2-(3-fluoro-2-methoxy-5-(1- methoxycyclohexyl)phenyl)acetate Step 1: 1-bromo-3-fluoro-5-iodo-2-methoxybenzene

[0892] To a solution of 2-bromo-6-fluorophenol (1 g, 5.24 mmol), I2 (1.90 g, 7.5 mmol) and KI (1.25 g, 7.5 mmol) in H2O (20 mL) at 0 °C, was added a solution of NaOH (0.4 g, 10 mmol) in H2O (5 mL) dropwise. The reaction was warmed to rt, and stirred at rt for 1.5 hours. The reaction was quenched with sat. NH
4Cl to PH~7, then extracted with EtOAc (20 mLx 3). The combined organic phase was washed with sat. sodium thiosulfate solution, brine, dried over Na2SO4, filtered and concentrated in vacuo to give the crude product 2-bromo-6-fluoro-4- iodophenol. This crude product was dissolved in DMF (20 mL), added K2CO3 (1.04 g, 7.5 mmol) and MeI (1.07 g, 7.5 mmol). The reaction mixture was stirred at rt overnight. The reaction was quenched with H2O (20 mL) and extracted with EtOAc(2x 20mL). The combined organic phase was washed with brine, dried over Na
2SO
4, filtered and concentrated in vacuo. The residue was
purified by silica gel column (pet ether: EtOAc 5:1) to give the desired product 1-bromo-3- fluoro-5-iodo-2-methoxybenzene as a yellow solid (1.2 g). Yield 69% (ESI 331 (M+H) +). Step 2: 1-(3-bromo-5-fluoro-4-methoxyphenyl)cyclohexan-1-ol

[0893] To a solution of 1-bromo-3-fluoro-5-iodo-2-methoxybenzene (500 mg, 1.51 mmol) in THF (dry, 20 mL) under Ar at 0°C, was added a solution of iso-propylmagnesium chloride in THF (1.0 M, 1.8 mL, 1.8 mmol). The reaction was stirred at 0°C for 1 hour, then a solution of tetrahydro-4H-pyran-4-one (180 mg, 1.8 mmol) in THF (5 mL) was added dropwise. After stirring at 0°C for 10 min, the cooling bath was removed and the reaction mixture was stirred at rt overnight. The reaction mixture was quenched with MeOH (10 mL), concentrated in vacuo and the residue was purified by silica gel column (20% ethyl acetate in PE) to give the desired product 4-(3-bromo-5-fluoro-4-methoxyphenyl)tetrahydro-2H-pyran-4-ol as a pale yellow oil (360 mg). Yield 78 % (ESI 305 (M+H) +). Step 3: 1-(3-bromo-5-fluoro-4-methoxyphenyl)cyclohexan-1-ol

[0894] To a solution of 4-(3-bromo-5-fluoro-4-methoxyphenyl)tetrahydro-2H-pyran-4-ol (1.0 g, 3.30 mmol) in THF (20 mL) was added NaH (60%, 200 mg, 5.0 mmol) and stirred at 0 °C for 10 min, then added MeI (710 mg, 5.0 mmol) and stirred at rt for 1 hour. The mixture was quenched with aq. NH4Cl (30 mL) and extracted with EtOAc (3x 30mL). The combined organic layer was concentrated in vacuo, and the residue was purified by silica gel column (PE: EA 10:1) to give the desired product 1-bromo-3-fluoro-2-methoxy-5-(1-methoxycyclohexyl)benzene as a yellow oil (750 mg, 2.37 mmol). Yield 72% (ESI 317 (M+H) +). Step 4: ethyl 2-(3-fluoro-2-methoxy-5-(1-methoxycyclohexyl)phenyl)acetate

[0895] To a mixture of 1-bromo-3-fluoro-2-methoxy-5-(1-methoxycyclohexyl)benzene (750 mg, 2.37 mmol), Pd2(dba)3 (92 mg, 0.125 mmol) and Q-phos (115 mg, 0.125 mmol) in THF (5 mL), was added (2-tert-butoxy-2-oxoethyl)zinc(II) bromide solution 1 M in THF (6.5 mL, 6.5 mmol) at rt under Ar. The reaction was stirred at 50°C for 2 hours. The mixture was quenched with NaHCO3 solution, the solid was removed by filtration. The filtrate was concentrated in vacuo, and the residue was purified by silica gel column (pet ether: EtOAc 5:1) to give the desired product ethyl 2-(3-fluoro-2-methoxy-5-(1-methoxycyclohexyl)phenyl)acetate as a pale red oil (510 mg, 1.57 mmol). Yield 66% (ESI 325 (M+H) +). Step 5: ethyl 2-bromo-2-(3-fluoro-2-methoxy-5-(1-methoxycyclohexyl)phenyl)acetate

[0896] To a solution of ethyl 2-(3-fluoro-2-methoxy-5-(1-methoxycyclohexyl)phenyl)acetate (510 mg, 1.57 mmol) in THF (15 mL) at -78°C, was added lithium diisopropylamide solution 2.0 M in THF/hexanes (1.5 mL, 3.0 mmol) dropwise. The reaction was stirred at -78°C for 30 min, then chlorotrimethylsilane (0.34 g, 3.1 mmol) was added and the reaction was stirred at - 78°C for another 30 min. Then a solution of NBS (0.56 g, 3.1 mmol) in THF (5 mL) was added and the reaction was stirred at -78°C for 30min. The reaction was quenched with H2O (10 mL) and extracted with EtOAc(2x 20mL). The combined organic phase was washed with sat. NaHCO3 solution, brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether: EtOAc 5:1) to give the desired product ethyl 2-bromo-2- (3-fluoro-2-methoxy-5-(1-methoxycyclohexyl)phenyl)acetate as a yellow oil (480 mg). Yield 76% (ESI 403 (M+H) +). Preparation of ethyl 2-bromo-2-(3-fluoro-2-methoxy-5-(2-methyltetrahydro-2H-pyran-2- yl)phenyl)acetate Step 1: ethyl 5-(3-bromo-5-fluoro-4-methoxyphenyl)-5-hydroxyhexanoate

[0897] To a solution of 1-bromo-3-fluoro-5-iodo-2-methoxybenzene (2.0 g, 6.04 mmol) in THF (dry, 40 mL) under Ar at 0°C, was added a solution of iso-propylmagnesium chloride in THF (1.0 M, 7.2 mL, 7.2 mmol). The reaction was stirred at 0°C for 1 hour, then a solution of ethyl 5-oxohexanoate (1.14 g, 7.2 mmol) in THF (5 mL) was added dropwise. After stirring at 0°C for 10 min, the cooling bath was removed and the reaction mixture was stirred at rt overnight. The reaction mixture was quenched with MeOH (10 mL), concentrated in vacuo and the residue was purified by silica gel column (30% ethyl acetate in PE) to give the desired product ethyl 5-(3-bromo-5-fluoro-4-methoxyphenyl)-5-hydroxyhexanoate as a pale yellow oil (932 mg, 2.57 mmol). Yield 42 % (ESI 363 (M+H) +). Step 2: 5-(3-bromo-5-fluoro-4-methoxyphenyl)hexane-1,5-diol

[0898] To a solution of ethyl 5-(3-bromo-5-fluoro-4-methoxyphenyl)-5-hydroxyhexanoate (932 mg, 2.57 mmol) at 0°C, was added a solution of LAH in THF (1.0 M, 3.8 mL, 3.8 mmol). The reaction was stirred at 0°C for 2 hours, then quenched with HCl (1N, 10 mL) and extracted with EtOAc(2x 20mL). The combined organic phase was washed with sat. NaHCO3 solution, brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether: EtOAc 1:1) to give the desired product 5-(3-bromo-5-fluoro-4- methoxyphenyl)hexane-1,5-diol as a pale yellow oil (530 mg). Yield 64 % (ESI 321 (M+H) +). Step 3: 2-(3-bromo-5-fluoro-4-methoxyphenyl)-2-methyltetrahydro-2H-pyran

[0899] To a solution of 5-(3-bromo-5-fluoro-4-methoxyphenyl)hexane-1,5-diol (430 mg, 1.34 mmol) and Et3N (271 mg, 2.68 mmol) in THF (10 mL) at 0 °C, was added MsCl (230 mg,
2.01mmol). The reaction mixture was stirred at rt for 2 hours, then added H2O (10 mL), and extracted with EtOAc (3x 20 mL). The combined organic layer was washed with brine, dried over Na2SO4, filtered, and concentrated in vacuo to give the crude product 5-(3-bromo-5-fluoro-4- methoxyphenyl)-5-hydroxyhexyl methanesulfonate. This crude product was dissolved in THF (10 mL), in n-heptane (10 mL), and added aq. NaOH (50%, 4 mL). The reaction mixture was stirred at 80 °C for 2 hours, then cooled to rt, added H
2O (20 mL), and extracted with EtOAc (3x 20 mL). The combined organic layer was washed with brine, dried over Na2SO4, filtered, and concentrated in vacuo. The residue was purified by silica gel column (pet ether: EtOAc = 0%~20%) to give the desired product 2-(3-bromo-5-fluoro-4-methoxyphenyl)-2- methyltetrahydro-2H-pyran as a yellow oil (260 mg). Yield 64% (ESI 303 (M+H) +). Step 4: ethyl 2-(3-fluoro-2-methoxy-5-(2-methyltetrahydro-2H-pyran-2-yl)phenyl)acetate

[0900] To a mixture of 2-(3-bromo-5-fluoro-4-methoxyphenyl)-2-methyltetrahydro-2H- pyran (298 mg, 0.98 mmol), Pd2(dba)3 (56 mg, 0.05 mmol) and Q-phos (38 mg, 0.05 mmol) in THF (3 mL) was added (2-ethoxy-2-oxoethyl)zinc(II) bromide solution 1 M in THF (5.0 mL, 5.0 mmol) at rt under Ar. The reaction was stirred at 60°C for 2 hours.The mixture was quenched with NaHCO3 solution, the solid was removed by filtration. The filtrate was concentrated in vacuo, and the residue was purified by silica gel column (pet ether: EtOAc 5:1) to give the desired product ethyl 2-(3-fluoro-2-methoxy-5-(2-methyltetrahydro-2H-pyran-2- yl)phenyl)acetate as a pale red oil (250 mg, 0.81 mmol). Yield 82% (ESI 311 (M+H) +). Step 5: ethyl 2-bromo-2-(3-fluoro-2-methoxy-5-(2-methyltetrahydro-2H-pyran-2- yl)phenyl)acetate

[0901] To a solution of ethyl 2-(3-fluoro-2-methoxy-5-(2-methyltetrahydro-2H-pyran-2- yl)phenyl)acetate (250 mg, 0.81 mmol) in THF (10 mL) at -78°C, was added lithium diisopropylamide solution 2.0 M in THF/hexanes (0.8 mL, 1.6 mmol) dropwise. The reaction
was stirred at -78°C for 30 min, then chlorotrimethylsilane (174 mg, 1.6 mmol) was added and the reaction was stirred at -78°C for another 30 min. Then a solution of NBS (285 mg, 1.6 mmol) in THF (5 mL) was added and the reaction was stirred at -78°C for 30min. The reaction was quenched with H2O (10 mL) and extracted with EtOAc(2x 20 mL). The combined organic phase was washed with sat. NaHCO3 solution, brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether: EtOAc 5:1) to give the desired product ethyl 2-bromo-2-(3-fluoro-2-methoxy-5-(2-methyltetrahydro-2H-pyran-2- yl)phenyl)acetate as a yellow oil (308 mg, 0.79 mmol). Yield 98% (ESI 389 (M+H)
+). Preparation of ethyl 2-bromo-2-(3-fluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4- yl)phenyl)acetate Step 1: 1-bromo-3-fluoro-5-iodo-2-methoxybenzene
[0902] To a solution of 2-bromo-6-fluorophenol (1 g, 5.24 mmol), I2 (1.90 g, 7.5 mmol) and KI (1.25 g, 7.5 mmol) in H2O (20 mL) at 0 °C, was added a solution of NaOH (0.4 g, 10 mmol) in H
2O (5 mL) dropwise. The reaction was warmed to rt, and stirred at rt for 1.5 hours. The reaction was quenched with sat. NH
4Cl to pH~7, then extracted with EtOAc (20 mLx 3). The combined organic phase was washed with sat. sodium thiosulfate solution, brine, dried over Na2SO4, filtered and concentrated in vacuo to give the crude product 2-bromo-6-fluoro-4- iodophenol. This crude product was dissolved in DMF (20 mL), added K2CO3 (1.04 g, 7.5 mmol) and MeI (1.07 g, 7.5 mmol). The reaction mixture was stirred at rt overnight. The reaction was quenched with H2O (20 mL) and extracted with EtOAc(2x 20mL). The combined organic phase was washed with brine, dried over Na
2SO
4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether: EtOAc 5:1) to give the desired product 1-bromo-3- fluoro-5-iodo-2-methoxybenzene as a yellow solid (1.2 g). Yield 69% (ESI 331 (M+H) +). Step 2: 4-(3-bromo-5-fluoro-4-methoxyphenyl)tetrahydro-2H-pyran-4-ol

[0903] To a solution of 1-bromo-3-fluoro-5-iodo-2-methoxybenzene (500 mg, 1.51 mmol) in THF (dry, 20 mL) under Ar at 0°C, was added a solution of iso-propylmagnesium chloride in THF (1.0 M, 1.8 mL, 1.8 mmol). The reaction was stirred at 0°C for 1 hour, then a solution of tetrahydro-4H-pyran-4-one (180 mg, 1.8 mmol) in THF (5 mL) was added dropwise. After stirring at 0°C for 10 min, the cooling bath was removed and the reaction mixture was stirred at rt overnight. The reaction mixture was quenched with MeOH (10 mL), concentrated in vacuo and the residue was purified by silica gel column (pet ether: EtOAc 4:1) to give the desired product 4- (3-bromo-5-fluoro-4-methoxyphenyl)tetrahydro-2H-pyran-4-ol as a pale yellow oil (360 mg). Yield 78 % (ESI 305 (M+H) +). Step 3: 4-(3-bromo-5-fluoro-4-methoxyphenyl)-4-methyltetrahydro-2H-pyran
[0904] To a solution of TiCl
4 (3.60 g, 18.8 mmol) in DCM (30 mL) at -55°C, was added Me2Zn (1M in hexanes, 19 mL,19 mmol) dropwise. The reaction was stirred at -78°C for 1 hour, then 4-(3-bromo-5-fluoro-4-methoxyphenyl)tetrahydro-2H-pyran-4-ol (2.28 g, 7.47 mmol) in THF (10 mL) was added at -78 °C. The reaction was stirred at room temperature under N2 for 3 hours. Then the reaction was stirred at 55°C under N2 overnight. The reaction was quenched with H
2O (60 mL) and extracted with EtOAc (60 mL × 3). The combined organic phase was washed with brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether/EtOAc = 0%~30%) to give the desired product 4-(3-bromo-5-fluoro- 4-methoxyphenyl)-4-methyltetrahydro-2H-pyran (1.1 g) as a pale yellow oil. Yield 49% (ESI 303 [M+H]+). Step 4: ethyl 2-(3-fluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4-yl)phenyl)acetate
[0905] To a mixture of 4-(3-bromo-5-fluoro-4-methoxyphenyl)-4-methyltetrahydro-2H- pyran (660 mg, 2.18 mmol), Q-phos (80 mg, 0.11 mmol) and Pd2(dba)3 (100 mg, 0.11 mmol) in THF (10 mL), was added (2-ethoxy-2-oxoethyl)zinc(II) bromide (1 M in THF, 8.8 mL). The
reaction mixture was stirred at 50 °C under N
2 for 2 hours. The reaction was quenched with NaHCO
3 (aq) and extracted with EtOAc (2 x 20 mL). The combined organic layer was concentrated in vacuo and the residue was purified by silica gel column (pet ether: EtOAc 2 : 1) to give the desired product ethyl 2-(3-fluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4- yl)phenyl)acetate (550 mg) as a red oil. Yield 81% (ESI 311 [M+H] +). Step 5: ethyl 2-bromo-2-(3-fluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4- yl)phenyl)acetate

[0906] To a solution of ethyl 2-(3-fluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4- yl)phenyl)acetate (550 mg, 1.77 mmol) in THF (20 mL) at -78°C, was added lithium diisopropylamide solution 2.0 M in THF/hexanes (2.2 mL, 4.4 mmol) dropwise. The reaction was stirred at -78°C for 30 min, then chlorotrimethylsilane (483 mg, 4.44 mmol) was added and the reaction was stirred at -78°C for another 30 min. Then a solution of NBS (790 mg, 4.44 mmol) in THF (10 mL) was added and the reaction was stirred at -78°C for 30 min. The reaction was quenched with H2O (10 mL), then extracted with EtOAc (20 mL × 3). The combined organic phase was washed with sat. NaHCO3 solution, brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (EtOAc/pet ether = 0%~30%) to give the desired product ethyl 2-bromo-2-(3-fluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4- yl)phenyl)acetate (590 mg) as a pale brown oil. Yield 86% ( ESI 389/391 [M+H]) Preparation of ethyl 2-bromo-2-(5-ethyl-3-fluoro-2-methoxyphenyl)acetate

Step 1: 1-(3-bromo-5-fluoro-4-hydroxyphenyl)ethan-1-one
[0907] To a solution of 1-(3-fluoro-4-hydroxyphenyl)ethan-1-one (10.0 g, 64.93 mmol) in DMF (100 mL) was added NBS (13.9 g, 77.92 mmol) in portions. The reaction solution was stirred at rt for 16 hours. Water (500 mL) was added, the mixture was extracted with EtOAc (2x 250 mL). The combined organic phase was washed with water (2x 300 mL) and brine (300 mL), dried over Na2SO4, concentrated in vacuo, and the residue was purified by silica gel column (pet ether: EtOAc 4:1) to give the desired product 1-(3-bromo-5-fluoro-4-hydroxyphenyl)ethan-1-one as a gray solid (12.1 g). Yield 80% (ESI 233.0 (M+H) +). Step 2: 1-(3-bromo-5-fluoro-4-methoxyphenyl)ethan-1-one

[0908] A mixture of 1-(3-bromo-5-fluoro-4-hydroxyphenyl)ethan-1-one (12.1 g, 52.16 mmol), Methyl iodide (11.1 g, 78.24 mmol) and K2CO3 (14.4 g, 104.32 mmol) in DMF (100 mL) were stirred at rt for 16 hours. Water (500 mL) was added, the mixture was extracted with EtOAc (2x 200 mL). The combined organic phase was washed with water (2x 300 mL) and brine (300 mL), dried over Na2SO4, concentrated in vacuo, and the residue was purified by silica gel column (pet ether: EtOAc 20:1) to give the desired product 1-(3-bromo-5-fluoro-4- methoxyphenyl)ethan-1-one as a yellow oil (10.4 g). Yield 81% (ESI 247.0 (M+H) +).
1H NMR (400 MHz, CDCl
3) δ 7.93 – 7.92 (m, 1H), 7.67 – 7.63 (m, 1H), 4.07 (d, J = 7.0 Hz, 3H), 2.55 (s, 3H). Step 3: 1-bromo-5-ethyl-3-fluoro-2-methoxybenzene
[0909] A solution of 1-(3-bromo-5-fluoro-4-methoxyphenyl)ethan-1-one (4.92 g, 20 mmol) in DCM (50 mL) was cooled to 0°C, added TFA (13.68 g, 120 mmol) and stirred at 0°C for 10 min, then Et3SiH (13.92 g, 120 mmol) was added and the reaction mixture was stirred at 0°C for
30min. The mixture was quenched with sat.NaHCO3 solution (100 mL), then extracted with DCM (3 × 50mL). The combined organic phase was dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column (pet ether: EtOAc 20:1~10:1) to give the desired product 1-bromo-5-ethyl-3-fluoro-2-methoxybenzene as a colorless oil (4.35 g). Yield 94% (ESI 233 (M+H) +). Step 4: ethyl 2-(5-ethyl-3-fluoro-2-methoxyphenyl)acetate

[0910] To a mixture of 1-bromo-5-ethyl-3-fluoro-2-methoxybenzene (4.35 g, 18.67 mmol), Pd2(dba)3 (512 mg, 0.56 mmol) and Q-phos (409 mg, 0.56 mmol) in THF (50 mL) was added (2- ethoxy-2-oxoethyl)zinc(II) bromide solution 1 M in THF (93.3 mL, 93.3 mmol) at RT under Ar. The reaction was stirred at 60°C for 2 hours. Then the reaction mixture was poured into sat. NaHCO3 solution (100 mL), extracted with EtOAc (3 × 50 mL). The combined organic layers were dried over Na2SO4, filtered and concentrated under vacuum. The residue was purified by silica gel column (pet ether/EtOAc = 0%~10%) to give the desired product product ethyl 2-(5- ethyl-3-fluoro-2-methoxyphenyl)acetate as a yellow oil (3.92 g, 87% yield). ESI 241 (M+H) +. Step 5: ethyl 2-bromo-2-(5-ethyl-3-fluoro-2-methoxyphenyl)acetate

[0911] To a solution of ethyl 2-(5-ethyl-3-fluoro-2-methoxyphenyl)acetate (3.92 g, 16.33 mmol) in THF (30 mL) at -78°C, was added lithium diisopropylamide solution 2.0 M in THF/hexanes (16.3 mL, 32.6 mmol) dropwise. The reaction was stirred at -78°C for 30 min, then chlorotrimethylsilane (3.56 g, 32.67 mmol) was added and the reaction was stirred at -78°C for another 30 min. Then a solution of NBS (5.82 g, 32.67 mmol) in THF (10 mL) was added and the reaction was stirred at -78°C for 30min. The reaction was quenched with H2O (10 mL), then extracted with EtOAc (50 mL × 3). The combined organic phase was washed with Sat. NaHCO3 solution, brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified
by silica gel column (pet ether/EtOAc = 0%~10%) to give the desired product as a brown oil (3.88 g, 75% yield). ESI 319 (M+H) +. Final Compounds General Example of a Petasis Reaction Preparation of 2-(3-fluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4-yl)phenyl)-2-((R)- 3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 1A and 1B)

[0912] A mixture of (R)-N-methyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine (230.43 mg, 559.52 umol, 3HCl), (3-fluoro-2-methoxy-5-(4- methyltetrahydro-2H-pyran-4-yl)phenyl)boronic acid (0.15 g, 559.52 umol), oxoacetic acid (103.01 mg, 1.12 mmol), 4A MS (0.2 g) in DCM (6 mL) and HFIP (2 mL) was stirred for 2 h at 60 °C. The mixture was filtered over a celite pad and the filtrate was concentrated. The crude residue was purified by prep-HPLC (column: Phenomenex Luna C1875 x 30 mm x 3 um; mobile phase: A water (0.2% Formic acid); B acetonitrile 10%-50%, 8 min; Flow Rate 25 mL/min) to afford 130 mg of 2-(3-fluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4-yl)phenyl)-2-((R)-3- (methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid which was separated by SFC (column: Regis (S,S) Whelk-O1 (250 mm x 25 mm x 10 um); mobile phase: [MeCN/EtOH (0.1%NH3water)]; B%: 60%-60%, 15 min) to give two peaks: [0913] Compound 1A, arbitrarily assigned as (S)-2-(3-fluoro-2-methoxy-5-(4- methyltetrahydro-2H-pyran-4-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (34.0 mg, 58.34 umol, 10.43% yield, 100% purity) was obtained as a yellow solid. SFC: Rt = 1.364 min, 99.6%.
1H NMR (400 MHz, METHANOL-d4) δ = 7.38 (d, J = 1.1 Hz, 1H), 7.21-7.11 (m, 2H), 6.38 (d, J = 7.3 Hz, 1H), 4.61 (s, 1H), 3.97 (d, J = 1.5 Hz, 3H), 3.80-3.68 (m, 2H), 3.65-3.55 (m, 2H), 3.44-3.33 (m, 3H), 3.30-
3.24 (m, 1H), 2.97-2.74 (m, 4H), 2.73-2.64 (m, 2H), 2.63-2.48 (m, 3H), 2.48-2.35 (m, 3H), 2.16- 2.03 (m, 3H), 1.95-1.82 (m, 3H), 1.77-1.54 (m, 6H), 1.40 (quin, J = 7.1 Hz, 2H), 1.25 (s, 3H). LCMS (ESI) m/z = 583.2 (M+1). [0914] Compound 1B, arbitrarily assigned as (R)-2-(3-fluoro-2-methoxy-5-(4- methyltetrahydro-2H-pyran-4-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (formic acid salt, 60.9 mg, 104.51 umol, 18.68% yield, 100% purity) was obtained as a yellow solid. SFC: R
t = 2.483 min, 99.14%.
1H NMR (400 MHz, METHANOL-d
4) δ = 8.39 (br s, 1H), 7.46 (d, J = 7.3 Hz, 1H), 7.17 (dd, J = 2.3, 13.9 Hz, 1H), 7.07 (s, 1H), 6.51 (d, J = 7.3 Hz, 1H), 4.89 (s, 1H), 3.94 (d, J = 2.0 Hz, 3H), 3.80-3.71 (m, 2H), 3.68-3.50 (m, 5H), 3.44-3.36 (m, 2H), 3.23-3.11 (m, 1H), 3.02-2.88 (m, 1H), 2.79-2.71 (m, 3H), 2.69-2.57 (m, 4H), 2.49-2.36 (m, 2H), 2.12-2.01 (m, 4H), 1.91-1.71 (m, 8H), 1.65 (br dd, J = 6.3, 12.2 Hz, 2H), 1.28 (s, 3H). LCMS (ESI) m/z = 583.2 (M+1). The following compounds were prepared according to the Petasis Example procedure substituting the appropriate reagents and purified using the conditions shown: 2-(5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin- 2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 2A and 2B)

[0915] The crude racemate was purified by prep-HPLC (column: Phenomenex Luna 80 x 30 mm x 3 um; mobile phase: [water (TFA 0.1%), B acetonitrile 10%-40%, 8 min; Flow Rate 25 mL/min), and then separated by SFC (column: Regis (S,S) Whelk-O1 (250 mm x 25mm x 10 um); mobile phase: [0.1% NH3water EtOH]; B%: 60%-60%, 30 min) to give two peaks: [0916] Compound 2A, arbitrarily assigned as (S)-2-(5-isopropyl-2-methoxyphenyl)-2-((R)- 3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (45.7 mg, 89.84 umol, 42.31% yield) was obtained as white-solid. SFC: Rt = 1.204 min, 100%. 1H NMR (400MHz, METHANOL-d4) δ = 7.40 (d, J = 2.1 Hz, 1H), 7.26 (dd, J = 2.2, 8.5 Hz, 1H), 7.13 (d, J = 7.3 Hz, 1H), 6.99 (d, J = 8.6 Hz, 1H), 6.35 (d, J = 7.4 Hz, 1H), 4.90 (s, 1H), 3.91-3.81 (m, 3H), 3.40-3.34 (m, 2H), 3.30-3.22 (m, 3H), 3.19-3.02 (m, 2H), 2.86 (td, J = 6.9, 13.8 Hz, 1H), 2.69 (t, J = 6.3 Hz, 2H), 2.57-2.37 (m, 4H), 2.26 (s, 3H), 2.20-2.08 (m, 1H), 1.95-
1.82 (m, 3H), 1.70-1.59 (m, 2H), 1.58-1.45 (m, 2H), 1.34 (quin, J = 7.4 Hz, 2H), 1.22 (dd, J = 1.8, 6.9 Hz, 6H). LCMS (ESI) m/z = 509.2 (M+1) [0917] Compound 2B, arbitrarily assigned as (R)-2-(5-isopropyl-2-methoxyphenyl)-2-((R)- 3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (44.2 mg, 86.89 umol, 40.93% yield) was obtained as a yellow oil. SFC: Rt = 2.055 min, 100%. 1H NMR (400MHz, METHANOL-d4) δ = 7.40 (d, J = 2.0 Hz, 1H), 7.24 (dd, J = 2.2, 8.5 Hz, 1H), 7.13 (d, J = 7.4 Hz, 1H), 6.98 (d, J = 8.6 Hz, 1H), 6.35 (d, J = 7.3 Hz, 1H), 4.90 (s, 1H), 3.86 (s, 3H), 3.41-3.33 (m, 4H), 3.29-3.23 (m, 1H), 3.02-2.91 (m, 2H), 2.90-2.80 (m, 1H), 2.70 (t, J = 6.2 Hz, 2H), 2.57-2.39 (m, 4H), 2.30 (s, 3H), 2.13 (dt, J = 7.4, 13.2 Hz, 1H), 1.97 (td, J = 7.1, 14.1 Hz, 1H), 1.87 (td, J = 6.0, 11.7 Hz, 2H), 1.64 (td, J = 7.6, 15.3 Hz, 2H), 1.57-1.47 (m, 2H), 1.33 (td, J = 7.5, 15.0 Hz, 2H), 1.22 (dd, J = 2.0, 6.9 Hz, 6H). LCMS (ESI) m/z = 509.2 (M+1). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-((5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)(2,2,2-trifluoroethyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 3A and 3B)

[0918] The residue was purified by prep-HPLC (column: Phenomenex Luna C18250 x 50 mm x 10 um; mobile phase: A water (TFA 0.1%), B acetonitrile 20%-50%, 10 min; Flow Rate 25 mL/min). The residue was purified by SFC (column: Regis (S,S) Whelk-O1 (250 mm x 25 mm x 10 um); mobile phase: [0.1% NH3water EtOH]; B%: 50%-50%, 10 min). [0919] Compound 3A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-((5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)(2,2,2- trifluoroethyl)amino)pyrrolidin-1-yl)acetic acid (14.9 mg, 25.10 umol, 8.4% yield, 100% purity) was obtained as white solid. SFC: R
t = 1.491 min, 100.0%.
1H NMR (400 MHz, METHANOL-d) δ = 7.25-7.21 (m, 1H), 7.17 (d, J = 7.3 Hz, 1H), 7.09 (dd, J = 2.0, 12.9 Hz, 1H), 6.37 (d, J = 7.3 Hz, 1H), 4.79 (s, 1H), 3.98 (d, J = 1.8 Hz, 3H), 3.75 (quin, J = 8.0 Hz, 1H), 3.39-3.35 (m, 2H), 3.27-3.14 (m, 4H), 3.13-3.01 (m, 2H), 2.92-2.84 (m, 1H), 2.76-2.62 (m, 4H), 2.56-2.49 (m, 2H),
2.15-2.06 (m, 1H), 1.93-1.84 (m, 3H), 1.69-1.61 (m, 2H), 1.49 (qd, J = 6.9, 13.4 Hz, 2H), 1.40- 1.28 (m, 2H), 1.23 (d, J = 6.8 Hz, 6H). LCMS (ESI) m/z = 595.2 (M+1). [0920] Compound 3B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-((5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)(2,2,2- trifluoroethyl)amino)pyrrolidin-1-yl)acetic acid (29.3 mg, 49.14 umol, 16.6% yield, 99.7% purity) was obtained as white solid. SFC: R
t = 1.703 min, 99.8%.
1H NMR (400 MHz, METHANOL-d) δ = 7.22-7.15 (m, 2H), 7.09 (dd, J = 2.1, 12.9 Hz, 1H), 6.38 (d, J = 7.4 Hz, 1H), 4.84-4.83 (m, 1H), 3.97 (d, J = 1.6 Hz, 3H), 3.77-3.66 (m, 1H), 3.42-3.36 (m, 4H), 3.25-3.19 (m, 2H), 3.06-2.86 (m, 3H), 2.75-2.63 (m, 4H), 2.56-2.49 (m, 2H), 2.14-2.04 (m, 1H), 1.97-1.84 (m, 3H), 1.71-1.62 (m, 2H), 1.53-1.46 (m, 2H), 1.40-1.33 (m, 2H), 1.25-1.20 (m, 1H), 1.23 (d, J = 6.9 Hz, 5H). LCMS (ESI) m/z = 595.2 (M+1) 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(5-((S)-1,2,3,4-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 4A and 4B)

[0921] The residue was purified by prep-HPLC (column: Phenomenex Luna C18250 x 50 mm x 10 um; mobile phase: A water (TFA 0.1%), B acetonitrile, 5%-35%, 10 min; Flow Rate (25 mL/min). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(5-((S)-1,2,3,4-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (300 mg, 569.60 umol, 80.51% yield) was obtained as a yellow oil and was separated by SFC (column: Regis (S,S) Whelk-O1 (250 mm x 25 mm x 10 um); mobile phase: [0.1% NH
3water EtOH]; B%: 50%-50%, 7 min) to give two peaks: [0922] Compound 4A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(5-((S)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (133.5 mg, 253.47 umol, 89.00% yield, 100% purity), was obtained as a yellow solid. SFC: Rt = 1.929 min, 100%.
1H NMR (400 MHz, METHANOL- d
4) δ = 7.70-7.60 (m, 2H), 7.14 (s, 1H), 7.06 (dd, J = 2.1, 12.8 Hz, 1H), 6.75 (t, J = 6.8 Hz, 1H), 4.64 (s, 1H), 3.94 (d, J = 1.5 Hz, 4H), 3.71-3.54 (m, 2H), 3.37-3.32 (m, 1H), 3.21-3.09 (m, 1H),
3.00-2.79 (m, 8H), 2.64-2.53 (m, 1H), 2.34-2.23 (m, 1H), 2.14-2.01 (m, 2H), 1.95-1.83 (m, 1H), 1.80-1.62 (m, 5H), 1.59-1.42 (m, 3H), 1.21 (d, J = 6.9 Hz, 6H). LCMS (ESI) m/z = 527.2 (M+1). [0923] Compound 4B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(5-((S)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (82.3 mg, 156.26 umol, 54.87% yield), was obtained as a white solid. SFC: R
t =2.163 min, 98.98%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.69 (d, J = 4.8 Hz, 1H), 7.29-7.22 (m, 2H), 7.03 (dd, J = 1.8, 12.8 Hz, 1H), 6.51 (dd, J = 5.4, 7.0 Hz, 1H), 4.67 (s, 1H), 3.96 (s, 3H), 3.46-3.39 (m, 2H), 3.28-3.23 (m, 1H), 3.11-2.92 (m, 2H), 2.87 (td, J = 6.8, 13.8 Hz, 1H), 2.81-2.71 (m, 4H), 2.71-2.61 (m, 1H), 2.46 (s, 3H), 2.21-2.10 (m, 1H), 2.07-1.92 (m, 2H), 1.68-1.37 (m, 9H), 1.22 (d, J = 6.8 Hz, 6H). LCMS (ESI) m/z = 527.2 (M+1). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(5-((R)-1,2,3,4-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 5A and 5B)

[0924] The residue was purified by prep-HPLC (column: Phenomenex luna C18250 x 50 mm x 10 um; mobile phase: A water (TFA 0.1%), B acetonitrile, 5%-35%, 10 min; Flow Rate (25 mL/min). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(5-((R)-1,2,3,4-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (300 mg, 569.60 umol, 60.38% yield) was obtained as a yellow oil and 100 mg was separated by SFC (column: Regis (S,S) Whelk-O1 (250 mm x 25 mm x 10 um); mobile phase: [0.1% NH
3water EtOH]; B%: 50%-50%, 25 min) to give two peaks: [0925] Compound 5A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(5-((R)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (33.8 mg, 62.80 umol, 22.05% yield, 97.85% purity) was obtained as a white solid. SFC: Rt = 1.771 min, 100%.
1H NMR (400 MHz, METHANOL- d
4) δ = 7.64 (br d, J = 5.1 Hz, 1H), 7.36 (br d, J = 7.0 Hz, 1H), 7.25 (s, 1H), 7.04 (dd, J = 1.9, 12.9 Hz, 1H), 6.60-6.51 (m, 1H), 4.63 (s, 1H), 3.96 (d, J = 1.4 Hz, 3H), 3.57-3.40 (m, 3H), 3.00-
2.63 (m, 8H), 2.50 (s, 3H), 2.22-2.10 (m, 1H), 2.06-1.92 (m, 2H), 1.72-1.55 (m, 6H), 1.53-1.40 (m, 3H), 1.22 (d, J = 6.9 Hz, 6H). LCMS (ESI) m/z = 527.2 (M+1). [0926] Compound 5B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(5-((R)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (63.9 mg, 118.58 umol, 41.64% yield, 97.74% purity) was obtained as a white solid. SFC, R
t =2.089 min, 100%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.69 (d, J = 4.0 Hz, 1H), 7.25 (br s, 2H), 7.02 (dd, J = 1.9, 12.8 Hz, 1H), 6.50 (dd, J = 5.3, 7.2 Hz, 1H), 4.66 (s, 1H), 3.95 (d, J = 1.4 Hz, 3H), 3.46-3.36 (m, 2H), 3.29-3.23 (m, 1H), 3.16-3.03 (m, 1H), 2.98-2.82 (m, 2H), 2.80-2.60 (m, 5H), 2.45 (s, 3H), 2.21-2.11 (m, 1H), 2.06-1.93 (m, 2H), 1.64-1.37 (m, 9H), 1.22 (dd, J = 1.3, 6.9 Hz, 6H). LCMS (ESI) m/z = 527.2 (M+1). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-((3-methoxy-3-methylbutyl)(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 6A and 6B)
[0927] The crude product was purified by prep-HPLC (column: Waters Xbridge BEH C18 100 x 30 mm x 10 um; mobile phase: A water (NH
4HCO
310 mmol/L), B acetonitrile 30%-60%, 8 min; Flow Rate (25 mL/min) to give two peaks: [0928] Compound 6A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-((3-methoxy-3-methylbutyl)(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (0.0458 g, 74.15 umol, 7.68% yield, 99.221% purity) was obtained as a yellow solid. SFC: R
t = 1.103 min, 100.0%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.23 (s, 1H), 7.15 (d, J = 7.4 Hz, 1H), 7.01 (dd, J = 2.0, 12.9 Hz, 1H), 6.36 (d, J = 7.3 Hz, 1H), 4.63 (s, 1H), 3.95 (d, J = 1.4 Hz, 3H), 3.65-3.58 (m, 1H), 3.39-3.36 (m, 2H), 3.30-3.24 (m, 1H), 3.14 (s, 3H), 3.02-2.93 (m, 1H), 2.92-2.79 (m, 5H), 2.70 (br t, J = 6.2 Hz, 4H),
2.53 (dt, J = 3.8, 7.6 Hz, 2H), 2.20-2.07 (m, 1H), 2.03-1.92 (m, 1H), 1.87 (quin, J = 5.9 Hz, 2H), 1.75-1.63 (m, 4H), 1.60-1.51 (m, 2H), 1.36 (quin, J = 7.1 Hz, 2H), 1.22 (dd, J = 1.6, 6.9 Hz, 6H), 1.15 (d, J = 1.3 Hz, 6H). LCMS (ESI) m/z = 613.3 (M+1), Rt = 1.889 min, 99.221% [0929] Compound 6B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-((3-methoxy-3-methylbutyl)(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (0.0124 g, 19.67 umol, 2.04% yield, 97.231% purity) was obtained as a yellow solid. SFC: R
t = 1.087 min, 100.0%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.24 (s, 1H), 7.16 (d, J = 7.3 Hz, 1H), 7.04 (dd, J = 2.1, 12.9 Hz, 1H), 6.37 (d, J = 7.4 Hz, 1H), 4.67 (s, 1H), 3.98-3.94 (m, 3H), 3.68-3.58 (m, 1H), 3.39-3.35 (m, 2H), 3.24 (br dd, J = 11.3, 14.6 Hz, 1H), 3.16 (s, 3H), 2.98 (br dd, J = 7.5, 11.4 Hz, 3H), 2.90-2.84 (m, 1H), 2.83-2.75 (m, 2H), 2.74-2.68 (m, 3H), 2.66-2.58 (m, 1H), 2.54 (t, J = 7.7 Hz, 2H), 2.15-2.06 (m, 1H), 1.94-1.84 (m, 3H), 1.73-1.64 (m, 4H), 1.61-1.48 (m, 2H), 1.41-1.32 (m, 2H), 1.22 (d, J = 6.9 Hz, 6H), 1.16 (s, 6H). LCMS (ESI) m/z = 613.3 (M+1), R
t = 1.900 min, 97.231%. 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(6-((S)-1,2,3,4-tetrahydro-1,8- naphthyridin-2-yl)hexyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 7A and 7B)
[0930] The residue was purified by prep-HPLC (column: C18-1150 x 30 mm x 5 um; mobile phase: A water (TFA 0.1%), B acetonitrile 1%-40%, 8 min; Flow Rate 25 mL/min) to afford 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(6-((S)-1,2,3,4-tetrahydro-1,8- naphthyridin-2-yl)hexyl)amino)pyrrolidin-1-yl)acetic acid (199 mg, 368.03 umol, 65.0% yield) as a yellow oil which was separated by SFC (column: DAICEL CHIRALPAK IG (250 mm x 30 mm x 10 um); mobile phase: [0.1% NH
3water IPA]; B%: 55%-55%, 20 min) to give two peaks: [0931] Compound 7A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(6-((S)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)hexyl)amino)pyrrolidin-1-yl)acetic acid (23.9 mg, 44.20 umol, 12.0% yield) was obtained as a white solid. SFC: R
t = 1.296 min, 100%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.68 (dd, J = 1.5, 5.3 Hz, 1H), 7.27-7.23 (m, 2H), 7.05 (dd, J = 2.1, 12.9 Hz, 1H), 6.50 (dd, J = 5.3, 7.1 Hz, 1H), 4.72 (s, 1H), 3.97 (d, J = 1.6 Hz, 3H), 3.45-3.38 (m, 2H), 3.33 (br s, 1H), 3.10-2.97 (m, 3H), 2.87 (td, J = 6.9, 13.8 Hz, 1H), 2.77-2.67 (m, 3H), 2.63-2.54 (m, 1H), 2.43 (s, 3H), 2.21-2.11 (m,
1H), 2.00-1.92 (m, 2H), 1.64-1.54 (m, 5H), 1.48-1.34 (m, 6H), 1.22 (d, J = 6.9 Hz, 6H). LCMS (ESI) m/z = 541.3 (M+1). [0932] Compound 7B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(6-((S)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)hexyl)amino)pyrrolidin-1-yl)acetic acid (40.9 mg, 75.64 umol, 20.6% yield) was obtained as a white solid. SFC: R
t =2.082 min, 100%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.71-7.66 (m, 1H), 7.25 (br s, 2H), 7.03 (dd, J = 2.0, 12.9 Hz, 1H), 6.50 (dd, J = 5.3, 7.1 Hz, 1H), 4.66 (s, 1H), 3.96 (d, J = 1.4 Hz, 3H), 3.43 (br s, 2H), 3.41 (br d, J = 5.9 Hz, 1H), 3.36 (br s, 1H), 3.14 (br s, 1H), 2.96-2.83 (m, 2H), 2.80-2.67 (m, 5H), 2.48 (s, 3H), 2.21-2.13 (m, 1H), 2.05-1.94 (m, 2H), 1.67-1.50 (m, 5H), 1.48-1.36 (m, 6H), 1.22 (dd, J = 1.1, 6.9 Hz, 6H). LCMS (ESI) m/z = 541.3 (M+1). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(6-((R)-1,2,3,4-tetrahydro-1,8- naphthyridin-2-yl)hexyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 8A and 8B)

[0933] The residue was purified by prep-HPLC (column: C18-1150 x 30 mm x 5 um; mobile phase: A water (TFA 0.1%), B acetonitrile 1%-40%, 8 min; Flow Rate 25 mL/min) to afford 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(6-((R)-1,2,3,4-tetrahydro-1,8- naphthyridin-2-yl)hexyl)amino)pyrrolidin-1-yl)acetic acid (285 mg, 527.08 umol, 74.5% yield) as a yellow oil which was separated by SFC (column: DAICEL CHIRALPAK AD (250 mm x 30 mm x 10 um); mobile phase: [0.1%NH
3water EtOH]; B%: 38%-38%, 7 min) to give two peaks: [0934] Compound 8A, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(6-((R)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)hexyl)amino)pyrrolidin-1-yl)acetic acid (30.4 mg, 56.22 umol, 10.7% yield) was obtained as a white solid. SFC: R
t = 3.352 min, 100%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.69 (dd, J = 1.6, 5.3 Hz, 1H), 7.27-7.23 (m, 2H), 7.06 (dd, J = 2.1, 12.9 Hz, 1H), 6.50 (dd, J = 5.3, 7.2 Hz, 1H), 4.72 (s, 1H), 3.97 (d, J = 1.6 Hz, 3H), 3.43-3.38 (m, 2H), 3.09-2.98 (m, 3H), 2.91-2.84 (m, 1H), 2.76-2.72 (m, 2H), 2.68-2.55 (m, 2H), 2.42 (s, 3H), 2.20-2.11 (m, 1H), 2.01-1.93 (m, 2H),
1.60-1.51 (m, 5H), 1.50-1.39 (m, 6H), 1.29 (br s, 1H), 1.23 (d, J = 6.9 Hz, 7H). LCMS (ESI) m/z = 541.3 (M+1). [0935] Compound 8B, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(6-((R)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)hexyl)amino)pyrrolidin-1-yl)acetic acid (75 mg, 138.71 umol, 26.3% yield) was obtained as a white solid. SFC: R
t = 3.810 min, 100%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.67-7.59 (m, 2H), 7.10-7.04 (m, 2H), 6.74 (t, J = 6.8 Hz, 1H), 4.81 (br s, 1H), 3.93 (d, J = 1.8 Hz, 3H), 3.65- 3.56 (m, 2H), 3.36-3.32 (m, 1H), 3.25-3.20 (m, 2H), 3.06-2.95 (m, 2H), 2.92-2.81 (m, 3H), 2.92- 2.78 (m, 1H), 2.69 (s, 3H), 2.63 (br d, J = 8.9 Hz, 1H), 2.13-2.03 (m, 2H), 1.78-1.61 (m, 6H), 1.47 (br s, 5H), 1.32-1.27 (m, 1H), 1.23 (dd, J = 1.9, 6.9 Hz, 6H). LCMS (ESI) m/z = 541.3 (M+1). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(4-((S)-1,2,3,4-tetrahydro-1,8- naphthyridin-2-yl)butyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 9A and 9B)

[0936] The residue was purified by prep-HPLC (column: Phenomenex Luna C18150 x 30 mm x 5 um; mobile phase: A water (TFA 0.1%), B acetonitrile 5%-35%, 8 min; Flow Rate 25 mL/min). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(4-((S)-1,2,3,4-tetrahydro- 1,8-naphthyridin-2-yl)butyl)amino)pyrrolidin-1-yl)acetic acid (300 mg, 585.19 umol, 84.4% yield) was obtained as a yellow oil and was separated by SFC (column: DAICEL CHIRALPAK AD (250 mm x 30 mm x 10 um); mobile phase: [0.1% NH
3water EtOH]; B%: 40%-40%, 9 min). [0937] Compound 9A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(4-((S)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)butyl)amino)pyrrolidin-1-yl)acetic acid (47.3 mg, 92.26 umol, 26.3% yield, 100% purity) was obtained as white solid. SFC: Rt = 3.463 min, 100.0%. LCMS (ESI) m/z = 513.3 (M+1).
1H NMR (400 MHz, METHANOL-d) δ = 7.70 (br t, J = 7.5 Hz, 2H), 7.19-7.09 (m, 2H), 6.84-6.77 (m, 1H), 4.95 (br s, 1H), 4.16-4.04 (m, 1H), 3.96 (s, 3H), 3.77-3.59 (m, 2H), 3.32 (br s, 2H), 3.20- 3.04 (m, 2H), 3.02-2.81 (m, 7H), 2.49-2.34 (m, 1H), 2.26-2.12 (m, 1H), 2.12-2.02 (m, 1H), 1.95- 1.41 (m, 7H), 1.26-1.18 (m, 6H).
[0938] Compound 9B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(4-((S)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)butyl)amino)pyrrolidin-1-yl)acetic acid (74.4 mg, 145.13 umol, 41.3% yield, 100% purity) was obtained as white solid. SFC: Rt = 3.976 min, 100.0%.
1H NMR (400 MHz, METHANOL-d) δ = 7.73-7.68 (m, 1H), 7.29-7.20 (m, 2H), 7.10-7.01 (m, 1H), 6.51 (dd, J = 5.2, 7.2 Hz, 1H), 4.71 (s, 1H), 3.96 (d, J = 1.5 Hz, 3H), 3.49-3.37 (m, 2H), 3.30-3.29 (m, 2H), 3.24-3.10 (m, 1H), 2.98 (br dd, J = 5.5, 11.4 Hz, 1H), 2.91-2.79 (m, 2H), 2.78-2.61 (m, 4H), 2.49-2.34 (m, 3H), 2.22-1.92 (m, 3H), 1.65-1.43 (m, 6H), 1.25-1.19 (m, 6H). LCMS (ESI) m/z = 513.3 (M+1). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(4-((R)-1,2,3,4-tetrahydro-1,8- naphthyridin-2-yl)butyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 10A and 10B)

[0939] The residue was purified by prep-HPLC (column: C18-1150 x 30 mm x 5 um; mobile phase: A water (TFA 0.1%), B acetonitrile 1%-40%, 8 min; Flow Rate 25 mL/min). 2-(3- fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(4-((R)-1,2,3,4-tetrahydro-1,8- naphthyridin-2-yl)butyl)amino)pyrrolidin-1-yl)acetic acid (300 mg, 585.19 umol, 84.4% yield) was obtained as a yellow oil which was separated by SFC (column: DAICEL CHIRALPAK AD (250 mm x 30 mm, 10 um); mobile phase: [0.1% NH3water EtOH]; B%: 33%-33%, 7 min). [0940] Compound 10A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(4-((R)-1,2,3,4-tetrahydro-1,8-naphthyridin-2- yl)butyl)amino)pyrrolidin-1-yl)acetic acid (49 mg, 95.58 umol, 49.0% yield, 100% purity) was obtained as white solid. SFC: R
t = 1.238 min, 97.2%.
1H NMR (400 MHz, METHANOL-d) δ = 7.72-7.68 (m, 1H), 7.27-7.22 (m, 2H), 7.06 (dd, J = 2.0, 12.9 Hz, 1H), 6.53-6.48 (m, 1H), 4.76 (s, 1H), 3.99-3.94 (m, 3H), 3.47-3.36 (m, 2H), 3.30-3.29 (m, 1H), 3.17-2.99 (m, 3H), 2.88 (quind, J = 6.8, 13.8 Hz, 1H), 2.77-2.76 (m, 1H), 2.77-2.72 (m, 1H), 2.71-2.53 (m, 2H), 2.40 (s, 3H), 2.23- 2.10 (m, 1H), 2.02-1.91 (m, 2H), 1.65-1.42 (m, 7H), 1.26-1.20 (m, 6H). LCMS (ESI) m/z = 513.3 (M+1) [0941] Compound 10B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(4-((R)-1,2,3,4-tetrahydro-1,8-naphthyridin-2-
yl)butyl)amino)pyrrolidin-1-yl)acetic acid (47.2 mg, 92.07 umol, 47.2% yield, 100% purity) was obtained as white solid. SFC: R
t = 1.354 min, 98.3%.
1H NMR (400 MHz, METHANOL-d) δ = 7.71 (d, J = 3.8 Hz, 1H), 7.25 (br s, 2H), 7.04 (dd, J = 1.9, 12.9 Hz, 1H), 6.51 (dd, J = 5.3, 7.1 Hz, 1H), 4.69 (s, 1H), 3.96 (d, J = 1.4 Hz, 3H), 3.46-3.37 (m, 2H), 3.30-3.27 (m, 1H), 3.23-3.08 (m, 1H), 2.95 (br dd, J = 5.4, 11.5 Hz, 1H), 2.91-2.78 (m, 2H), 2.77-2.62 (m, 4H), 2.46 (s, 3H), 2.23- 2.12 (m, 1H), 2.09-1.93 (m, 2H), 1.69-1.53 (m, 5H), 1.52-1.42 (m, 2H), 1.22 (dd, J = 1.7, 6.9 Hz, 6H). LCMS (ESI) m/z = 513.3 (M+1). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(isopropyl(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 11A and 11B)

[0942] The crude product which was purified by prep-HPLC (column: Phenomenex Luna C1875 x 30 mm x 3 um; mobile phase: A water (Formic acid 0.2%), B acetonitrile 1%-40%, 8 min); Flow Rate (25 mL/min). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(isopropyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (0.1 g, 180.27 umol, 24.78% yield) was obtained as a white solid and was separated by SFC (column: Regis (S,S) Whelk-O1 (250 x 25 mm, 10 um); mobile phase: [0.1% NH3water EtOH]; B%: 60%-60%, 20 min) to give two peaks: [0943] Compound 11A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(isopropyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (20.40 mg, 35.93 umol, 39.87% yield, 97.715% purity), was obtained as a white powder. SFC: Rt = 0.705 min, 100.0%.
1H NMR (400 MHz, METHANOL-d4) δ = 7.23 (s, 1H), 7.14 (d, J = 7.1 Hz, 1H), 7.05 (dd, J = 2.1, 12.8 Hz, 1H), 6.36 (d, J = 7.3 Hz, 1H), 4.70 (s, 1H), 3.96 (d, J = 1.6 Hz, 3H), 3.81-3.72 (m, 1H), 3.38-3.35 (m, 2H), 3.24-3.12 (m, 2H), 3.04-2.79 (m, 4H), 2.74-2.60 (m, 4H), 2.52 (t, J = 7.6 Hz, 2H), 2.08-1.99 (m, 1H), 1.95-1.84 (m, 3H), 1.69-1.61 (m, 2H), 1.57-1.46 (m, 2H), 1.38-1.31 (m, 2H), 1.22 (d, J = 6.9 Hz, 6H), 1.08 (br d, J = 6.5 Hz, 6H). LCMS (ESI) m/z = 555.3 (M+1).
[0944] Compound 11B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(isopropyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (30.5 mg, 54.30 umol, 60.24% yield, 98.762% purity) was obtained as a white powder. SFC: Rt = 1.166 min, 100.0%.
1H NMR (400 MHz, METHANOL-d4) δ = 7.25 (s, 1H), 7.14 (d, J = 7.4 Hz, 1H), 7.00 (dd, J = 2.0, 12.9 Hz, 1H), 6.35 (d, J = 7.3 Hz, 1H), 4.60 (s, 1H), 3.94 (d, J = 1.5 Hz, 3H), 3.79-3.71 (m, 1H), 3.39-3.36 (m, 2H), 3.28 (br d, J = 4.9 Hz, 1H), 3.05-2.90 (m, 1H), 2.90-2.78 (m, 3H), 2.78-2.64 (m, 5H), 2.50 (t, J = 7.5 Hz, 2H), 2.15-2.05 (m, 1H), 2.04-1.94 (m, 1H), 1.87 (td, J = 6.0, 11.7 Hz, 2H), 1.67-1.51 (m, 4H), 1.36-1.29 (m, 2H), 1.21 (dd, J = 2.8, 6.9 Hz, 6H), 1.16-1.11 (m, 6H). LCMS (ESI) m/z = 555.4 (M+1). 2-(2,5-difluoro-3-isopropyl-6-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 12A and 12B)

[0945] The residue was purified by prep-HPLC (column: Phenomenex Luna C1875 x 30 mm x 3 um; mobile phase: A water (Formic acid 0.2%), B acetonitrile, 10%-40%, 8 min; Flow Rate (25 ml/min) to give 2-(2,5-difluoro-3-isopropyl-6-methoxyphenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (150 mg, 282.15 umol, 51.0% yield) as a white solid which was further purified by SFC (column: Regis (S,S) Whelk-O1 (250 mm x 50 mm x 10 um); mobile phase: [0.1% NH3water MeOH]; B%: 50%- 50%, 13 min). [0946] Compound 12A, arbitrarily assigned as (S)-2-(2,5-difluoro-3-isopropyl-6- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (14.3 mg, 26.25 umol, 9.3% yield) was obtained as a white solid. SFC: Rt = 1.712 min, 99.99%.
1H NMR (400 MHz, METHANOL-d4) δ = 7.16 (d, J = 1H), 7.09 (dd, J = 7.0, 12.6 Hz, 1H), 6.37 (d, J = 7.3 Hz, 1H), 4.82 (s, 1H), 3.94 (d, J = 2.0 Hz, 3H), 3.39 - 3.33 (m, 3H), 3.22 - 3.05 (m, 3H), 3.02 - 2.94 (m, 1H), 2.86 - 2.73 (m, 2H), 2.70 (t, J = 6.3 Hz, 2H), 2.65 - 2.57 (m, 1H), 2.57 - 2.51 (m, 2H), 2.44 (s, 3H), 2.11 - 2.02 (m,
1H), 1.91 - 1.84 (m, 3H), 1.72 - 1.64 (m, 2H), 1.64 - 1.56 (m, 2H), 1.38 (quin, J = 7.3 Hz, 2H), 1.23 (d, J = 7.0 Hz, 6H). LCMS (ESI) m/z = 545.3 (M+1). [0947] Compound 12B, arbitrarily assigned as (R)-2-(2,5-difluoro-3-isopropyl-6- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (39.2 mg, 71.97 umol, 25.5% yield) was obtained as a white solid. SFC: R
t = 1.929 min, 99.3%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.19 (d, J = 7.3 Hz, 1H), 7.06 (dd, J = 7.1, 12.6 Hz, 1H), 6.39 (d, J = 7.3 Hz, 1H), 4.89 - 4.87 (m, 1H), 3.92 (d, J = 2.1 Hz, 3H), 3.39 - 3.33 (m, 3H), 3.17 (td, J = 6.8, 13.6 Hz, 2H), 3.06 - 2.90 (m, 2H), 2.71 (br t, J = 6.3 Hz, 3H), 2.67 - 2.51 (m, 4H), 2.47 (s, 3H), 2.02 - 1.84 (m, 4H), 1.79 - 1.67 (m, 2H), 1.67 - 1.57 (m, 2H), 1.48 - 1.38 (m, 2H), 1.22 (d, J = 6.9 Hz, 6H). LCMS (ESI) m/z = 545.3 (M+1). 2-(3-fluoro-2-methoxy-5-(1-methylcyclopropyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 13A and 13B)

[0948] Crude 2-(3-fluoro-2-methoxy-5-(1-methylcyclopropyl)phenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was purified and separated by prep-HPLC (column: Waters Xbridge BEH C18100 x 30 mm x 10 um; mobile phase: A water (NH4HCO310 mmol/L), B acetonitrile 15%-40%, 8 min; Flow Rate 25 mL/min) to give two peaks. [0949] Compound 13A, arbitrarily assigned as (S)-2-(3-fluoro-2-methoxy-5-(1- methylcyclopropyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (63.4 mg, 117.69 umol, 44.0% yield, 100% purity) was obtained as white solid. SFC: Rt = 1.132 min, 100.0%.
1H NMR (400 MHz, METHANOL-d) δ = 7.24 (s, 1H), 7.16 (d, J = 7.3 Hz, 1H), 7.01 (dd, J = 2.1, 13.1 Hz, 1H), 6.38-6.34 (m, 1H), 4.63 (s, 1H), 3.94 (d, J = 1.2 Hz, 3H), 3.44-3.35 (m, 3H), 3.27-3.19 (m, 1H), 3.02-2.89 (m, 2H), 2.81- 2.60 (m, 5H), 2.58-2.50 (m, 2H), 2.48-2.42 (m, 3H), 2.18-1.94 (m, 2H), 1.87 (quin, J = 5.9 Hz,
2H), 1.72-1.54 (m, 4H), 1.41-1.32 (m, 5H), 0.86-0.81 (m, 2H), 0.76-0.71 (m, 2H). LCMS (ESI) m/z = 539.3 (M+1), R
t = 1.765 min, 100.0% [0950] Compound 13B, arbitrarily assigned as (R)-2-(3-fluoro-2-methoxy-5-(1- methylcyclopropyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (11.7 mg, 21.59 umol, 8.1% yield, 99.4% purity) was obtained as white solid. SFC: R
t = 1.118 min, 96.4%.
1H NMR (400 MHz, METHANOL-d) δ = 7.29-7.22 (m, 1H), 7.19-7.12 (m, 1H), 7.03 (dd, J = 2.0, 13.1 Hz, 1H), 6.37 (d, J = 7.4 Hz, 1H), 4.63 (s, 1H), 3.95 (s, 3H), 3.42-3.35 (m, 3H), 3.30-3.15 (m, 3H), 2.95 (br dd, J = 7.7, 10.9 Hz, 3H), 2.70 (br t, J = 6.2 Hz, 2H), 2.54 (br t, J = 7.7 Hz, 2H), 2.39 (s, 3H), 2.17-2.07 (m, 1H), 1.97- 1.81 (m, 3H), 1.72-1.50 (m, 4H), 1.44-1.31 (m, 5H), 0.89-0.81 (m, 2H), 0.77-0.70 (m, 2H). LCMS (ESI) m/z = 539.3 (M+1), Rt = 1.775 min, 99.426% 2-((R)-3-((2,2-difluoroethyl)(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid (Compounds 14A and 14B)

[0951] Crude 2-((R)-3-((2,2-difluoroethyl)(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid was purified by prep-HPLC (column: Phenomenex Luna C18250 x 50 mm x 10 um; mobile phase: A water (TFA 0.1%), B acetonitrile 10%-50%, 10 min; Flow Rate 25 mL/min) to give two peaks: [0952] Compound 14A, arbitrarily assigned as (S)-2-((R)-3-((2,2-difluoroethyl)(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)acetic acid (173.5 mg, 251.19 umol, 59.6% yield) was obtained as a white solid. SFC: Rt = 1.218 min, 96.3%.
1H NMR (400 MHz, METHANOL-d4) δ = 7.59-7.54 (m, 1H), 7.24 (dd, J = 2.0, 13.1 Hz, 1H), 7.16 (s, 1H), 6.59 (d, J = 7.4 Hz, 1H), 5.86 (tt, J = 4.0, 55.9 Hz, 1H), 5.36-5.27 (m, 1H), 3.99 (d, J = 2.1 Hz, 3H), 3.72 (quin, J = 7.2 Hz, 1H), 3.50-3.47 (m, 2H), 3.35
(br d, J = 7.2 Hz, 3H), 3.15 (td, J = 7.7, 11.5 Hz, 1H), 3.03-2.88 (m, 3H), 2.81 (t, J = 6.1 Hz, 2H), 2.73-2.65 (m, 4H), 2.22-2.10 (m, 1H), 2.10-2.01 (m, 1H), 1.94 (quin, J = 6.0 Hz, 2H), 1.73 (quin, J = 7.7 Hz, 2H), 1.61-1.51 (m, 2H), 1.48-1.39 (m, 2H), 1.25 (d, J = 6.9 Hz, 6H). LCMS (ESI) m/z = 577.3 (M+1), Rt = 2.166 min, 100% [0953] Compound 14B, arbitrarily assigned as (R)-2-((R)-3-((2,2-difluoroethyl)(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)acetic acid (52 mg, 73.97 umol, 17.9% yield) was obtained as a white solid. SFC: R
t =1.073 min, 100%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.56 (d, J = 7.4 Hz, 1H), 7.27- 7.19 (m, 1H), 7.17 (s, 1H), 6.59 (d, J = 7.4 Hz, 1H), 5.84 (tt, J = 3.8, 56.0 Hz, 1H), 5.30 (s, 1H), 4.00 (d, J = 2.1 Hz, 3H), 3.77 (td, J = 7.7, 15.7 Hz, 1H), 3.50-3.47 (m, 2H), 3.44-3.32 (m, 3H), 3.21 (br s, 1H), 3.02-2.87 (m, 3H), 2.81 (t, J = 6.1 Hz, 2H), 2.68 (td, J = 7.4, 19.1 Hz, 4H), 2.20 (dtd, J = 3.5, 6.8, 13.2 Hz, 1H), 2.04-1.91 (m, 3H), 1.78-1.64 (m, 2H), 1.61-1.48 (m, 2H), 1.47- 1.37 (m, 2H), 1.25 (dd, J = 1.0, 6.9 Hz, 6H). LCMS (ESI) m/z = 577.2 (M+1), R
t = 2.212 min, 98.252%. 2-(3,4-difluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 15A and 15B)
[0954] Crude 2-(3,4-difluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was purified by prep- HPLC (column: Waters Xbridge BEH C18100 x 30 mm x 10 um; mobile phase: A water (NH4HCO310 mmol/L), B acetonitrile 30%-50%, 8min; Flow Rate (25 ml/min) to give two peaks: [0955] Compound 15A, arbitrarily assigned as (S)-2-(3,4-difluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (55.5 mg, 101.90 umol, 62.50% yield, 100% purity) was obtained as a white solid. SFC: R
t = 1.220 min, 100%.
1H NMR (400MHz,
CHLOROFORM-d) δ = 7.27 (dd, J = 7.7, 1.9 Hz, 1H), 7.17 (d, J = 7.3 Hz, 1H), 6.37 (d, J = 7.4 Hz, 1H), 4.53 (s, 1H), 3.98 (d, J = 1.3 Hz, 3H), 3.41-3.48 (m, 1H), 3.36 - 3.39 (m, 2H), 3.17 (dt, J = 13.8, 6.9 Hz, 2H), 2.81 - 2.92 (m, 3H), 2.70 (br t, J = 6.2 Hz, 3H), 2.52 - 2.62 (m, 3H), 2.50 (s, 3H), 2.07 - 2.16 (m, 1H), 1.94 - 2.03 (m, 1H), 1.85 - 1.90 (m, 2H), 1.59 - 1.70 (m, 4H), 1.35 - 1.42 (m, 2H), 1.24 ppm (dd, J = 6.9, 4.8 Hz, 6H). LCMS (ESI) m/z = 545.3 (M+1), Rt = 2.537 min, 100% [0956] Compound 15B, arbitrarily assigned as (R)-2-(3,4-difluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (29.6 mg, 53.62 umol, 32.89% yield, 98.66% purity) was obtained as a white solid. SFC: Rt =1.102 min, 100%.
1H NMR (400MHz, CHLOROFORM- d) δ = 7.30 (dd, J = 7.8, 2.0 Hz, 1H), 7.18 (d, J = 7.3 Hz, 1H), 6.38 (d, J = 7.4 Hz, 1H), 4.52 (s, 1H), 3.99 (d, J = 1.6 Hz, 3H), 3.40 - 3.46 (m, 1H), 3.36 - 3.39 (m, 2H), 3.14 - 3.28 (m, 2H), 2.76 - 2.91 (m, 4H), 2.70 (br t, J = 6.2 Hz, 2H), 2.59 - 2.66 (m, 1H), 2.53 - 2.57 (m, 2H), 2.45 (s, 3H), 2.06 - 2.15 (m, 1H), 1.88 (dq, J = 11.4, 5.7 Hz, 3H), 1.69 (br t, J = 7.5 Hz, 2H), 1.53 - 1.65 (m, 2H), 1.41 (quin, J = 7.3 Hz, 2H), 1.25 ppm (dd, J = 8.6, 7.1 Hz, 6H). LCMS (ESI) m/z = 545.3 (M+1), Rt = 2.596 min, 98.66% 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-((5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)(3,3,3-trifluoropropyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 16A and 16B)

[0957] The residue was purified by prep-HPLC (column: Phenomenex luna C18250 x 50 mm x 10 um; mobile phase: A water (TFA 0.1%), B acetonitrile 10%-40%, 8 min; Flow Rate 25 mL/min). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-((5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)(3,3,3-trifluoropropyl)amino)pyrrolidin-1-yl)acetic acid (459 mg, 754.06 umol, 79.94% yield) was obtained as a yellow oil and was further separated by SFC (column: DAICEL CHIRALPAK IG (250 mm x 30 mm, 10 um); mobile phase: [0.1% NH3water EtOH]; B%: 50%-50%, 8 min) to give two peaks:
[0958] Compound 16A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-((5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)(3,3,3- trifluoropropyl)amino)pyrrolidin-1-yl)acetic acid (89.2 mg, 146.54 umol, 19.43% yield) was obtained as a white solid. SFC: Rt = 1.369 min, 99.7%.
1H NMR (400 MHz, METHANOL-d4) δ = 7.27-7.21 (m, 1H), 7.17 (d, J = 7.4 Hz, 1H), 7.09 (br d, J = 12.6 Hz, 1H), 6.37 (d, J = 7.3 Hz, 1H), 4.82 (br d, J = 1.8 Hz, 1H), 3.98 (s, 3H), 3.56 (br d, J = 6.1 Hz, 1H), 3.39-3.35 (m, 3H), 3.13 (br s, 1H), 3.08-2.98 (m, 1H), 2.92-2.85 (m, 1H), 2.83-2.74 (m, 2H), 2.70 (br t, J = 6.1 Hz, 2H), 2.54 (br d, J = 7.6 Hz, 2H), 2.47 (br dd, J = 6.9, 13.2 Hz, 1H), 2.38-2.28 (m, 2H), 2.13-2.04 (m, 1H), 2.12-1.82 (m, 3H), 1.65 (br dd, J = 4.2, 7.4 Hz, 2H), 1.51-1.42 (m, 2H), 1.40-1.27 (m, 4H), 1.23 (d, J = 6.7 Hz, 6H). LCMS (ESI) m/z = 609.2 (M+1). [0959] Compound 16B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-((5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)(3,3,3- trifluoropropyl)amino)pyrrolidin-1-yl)acetic acid (251 mg, 412.35 umol, 54.68% yield) was obtained as a white solid. SFC: R
t =1.542 min, 99.7%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.54 (d, J = 7.3 Hz, 1H), 7.25-7.14 (m, 2H), 6.58 (d, J = 7.4 Hz, 1H), 5.17 (s, 1H), 3.98 (d, J = 1.9 Hz, 3H), 3.57 (br d, J = 3.8 Hz, 2H), 3.50-3.34 (m, 4H), 3.08-2.98 (m, 1H), 2.97-2.84 (m, 3H), 2.80 (br t, J = 6.2 Hz, 2H), 2.74-2.68 (m, 2H), 2.67-2.54 (m, 2H), 2.45-2.30 (m, 2H), 2.15-2.04 (m, 2H), 1.92 (quin, J = 5.9 Hz, 2H), 1.82-1.71 (m, 2H), 1.69-1.51 (m, 4H), 1.25 (d, J = 7.0 Hz, 6H). LCMS (ESI) m/z = 609.2 (M+1). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(2-methyl-6-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)hexan-2-yl)amino)pyrrolidin-1-yl)acetic acid (Compounds 17A and 17B)

[0960] The residue was purified by prep-HPLC (column: Phenomenex Luna 80 x 30 mm x 3 um; mobile phase: A water (TFA 0.1%), B acetonitrile, 1%-35%, 8 min; Flow Rate (25 ml/min). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(2-methyl-6-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)hexan-2-yl)amino)pyrrolidin-1-yl)acetic acid (110 mg, 198.29 umol, 52.55% yield) was obtained as colorless oil and was further separated by SFC (column: DAICEL
CHIRALPAK AD (250 mm x 30 mm, 10 um); mobile phase: [0.1% NH
3water EtOH]; B%: 42%- 42%, 12 min). [0961] Compound 17A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(2-methyl-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2- yl)amino)pyrrolidin-1-yl)acetic acid (8 mg, 14.30 umol, 14.42% yield, 99.15% purity), was obtained as a white solid. SFC: R
t = 4.851 min, 100%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.27 (s, 1H), 7.11 (d, J = 7.4 Hz, 1H), 7.09 - 7.03 (m, 1H), 6.35 (d, J = 7.4 Hz, 1H), 4.70 (br s, 1H), 4.05 - 3.96 (m, 4H), 3.36 (br d, J = 5.6 Hz, 4H), 2.87 (qd, J = 6.8, 13.7 Hz, 3H), 2.69 (t, J = 6.3 Hz, 2H), 2.50 (t, J = 7.4 Hz, 2H), 2.34 (br s, 3H), 2.05 - 1.98 (m, 1H), 1.93 - 1.84 (m, 3H), 1.65 - 1.58 (m, 2H), 1.52 - 1.44 (m, 2H), 1.29 (br d, J = 3.3 Hz, 2H), 1.23 (dd, J = 1.1, 6.9 Hz, 6H), 1.06 (br d, J = 6.1 Hz, 6H). LCMS (ESI) m/z = 555.3 (M+1) [0962] Compound 17B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(2-methyl-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2- yl)amino)pyrrolidin-1-yl)acetic acid (29.9 mg, 53.58 umol, 54.04% yield, 99.4% purity), was obtained as a yellow solid. SFC: Rt =6.073 min, 97.66%.
1H NMR (400 MHz, METHANOL-d4) δ = 7.27 (s, 1H), 7.13 (d, J = 7.3 Hz, 1H), 7.05 (dd, J = 1.6, 12.8 Hz, 1H), 6.36 (d, J = 7.3 Hz, 1H), 4.67 (br s, 1H), 4.11 - 4.03 (m, 1H), 3.97 (d, J = 1.4 Hz, 3H), 3.39 - 3.35 (m, 2H), 3.30 (br s, 2H), 2.94 - 2.76 (m, 3H), 2.69 (t, J = 6.3 Hz, 2H), 2.52 (t, J = 7.6 Hz, 2H), 2.39 (br s, 3H), 2.09 - 1.96 (m, 2H), 1.90 - 1.83 (m, 2H), 1.62 (td, J = 7.5, 14.9 Hz, 2H), 1.50 (br dd, J = 5.0, 10.9 Hz, 2H), 1.32 - 1.28 (m, 2H), 1.23 (d, J = 7.0 Hz, 6H), 1.08 (br d, J = 6.6 Hz, 6H). LCMS (ESI) m/z = 555.3 (M+1) 2-(3-fluoro-2-methoxy-5-((S)-1-methoxypropan-2-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 18A and 18B)

[0963] The residue was purified by prep-HPLC (column: A water (TFA 0.1%), B acetonitrile 5%-35%, 8 min; Flow Rate 25 mL/min). 2-(3-fluoro-2-methoxy-5-((S)-1-methoxypropan-2- yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-
yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (150 mg, 269.44 umol, 27.1% yield) was obtained as a white solid and was separated by SFC (column: Regis (S,S) Whelk-O1 (250 mm x 25 mm, 10 um); mobile phase: [acetonitrile/EtOH = 1:1, (0.1% NH3water)]; B%: 60%-60%, 15 min) to give two peaks: [0964] Compound 18A, arbitrarily assigned as (S)-2-(3-fluoro-2-methoxy-5-((S)-1- methoxypropan-2-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (17.94 mg, 31.61 umol, 98.1% purity) which was obtained as white solid. SFC: R
t = 1.074 min, 100%.
1H NMR (400 MHz, METHANOL-d) δ = 7.15 (s, 1 H), 7.07 (d, J = 7.27 Hz, 1 H), 6.95 (dd, J = 12.81, 2.09 Hz, 1 H), 6.27 (d, J = 7.27 Hz, 1 H), 4.54 (s, 1 H), 3.86 (d, J = 1.67 Hz, 3 H), 3.24-3.39 (m, 6 H), 3.19 (s, 5 H), 2.86 (br dd, J = 13.65, 6.97 Hz, 4 H), 2.60 (t, J = 6.26 Hz, 2 H), 2.43 (br d, J = 7.87 Hz, 2 H), 2.30 (s, 3 H), 1.95- 2.08 (m, 1 H), 1.73-1.87 (m, 3 H), 1.36-1.65 (m, 4 H), 1.24-1.34 (m, 2 H), 1.12 (d, J = 7.03 Hz, 3 H). LCMS (ESI) m/z = 557.3 (M+1). [0965] Compound 18B, arbitrarily assigned as (R)-2-(3-fluoro-2-methoxy-5-((S)-1- methoxypropan-2-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (60.64 mg, 106.16 umol, 97.5% purity) which was obtained as white solid. SFC: Rt = 2.142 min, 99.78%.
1H NMR (400 MHz, METHANOL-d) δ ppm 7.44 (d, J = 7.39 Hz, 1 H), 6.94-7.07 (m, 2 H), 6.47 (d, J = 7.39 Hz, 1 H), 4.73 (s, 1 H), 3.72-3.88 (m, 4 H), 3.30-3.42 (m, 4 H), 3.24 (s, 1 H), 3.11-3.20 (m, 4 H), 2.95-3.05 (m, 1 H), 2.83-2.92 (m, 1 H), 2.67-2.82 (m, 6 H), 2.47-2.66 (m, 3 H), 1.98-2.23 (m, 2 H), 1.78-1.90 (m, 2 H), 1.60-1.75 (m, 4 H), 1.35-1.50 (m, 2 H), 1.12 (d, J = 7.03 Hz, 3 H). LCMS (ESI) m/z = 557.3 (M+1) 2-(3-fluoro-2-methoxy-5-((R)-1-methoxypropan-2-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 18C and 18D)
[0966] Crude 2-(3-fluoro-2-methoxy-5-((R)-1-methoxypropan-2-yl)phenyl)-2-((R)-3- (methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was
purified by prep-HPLC (column: Waters Xbridge BEH C18100 x 30 mm x 10 um; mobile phase: A: water (NH
4HCO
310 mmol/L), B acetonitrile 30%-60%, 8 min; Flow Rate (25 mL/min). [0967] Compound 18C, arbitrarily assigned as (S)-2-(3-fluoro-2-methoxy-5-((R)-1- methoxypropan-2-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (47.9 mg, 86.04 umol, 41.65% yield, 100% purity) was obtained as a white solid. SFC: R
t = 1.150 min, 97.86%.
1H NMR (400 MHz, METHANOL- d) δ = 7.27-7.25 (m, 1H), 7.18 (br d, J = 7.5 Hz, 1H), 7.06 (dd, J = 2.0, 12.8 Hz, 1H), 6.38 (d, J = 7.3 Hz, 1H), 4.64 (s, 1H), 3.99-3.93 (m, 3H), 3.50-3.33 (m, 9H), 3.00-2.88 (m, 4H), 2.75-2.64 (m, 3H), 2.60-2.51 (m, 3H), 2.47 (s, 3H), 2.17-2.08 (m, 1H), 1.98-1.82 (m, 3H), 1.74-1.52 (m, 4H), 1.46-1.34 (m, 2H), 1.25-1.18 (m, 3H). LCMS (ESI) m/z = 557.3 (M+1), Rt = 1.700 min, 100% [0968] Compound 18D, arbitrarily assigned as (R)-2-(3-fluoro-2-methoxy-5-((R)-1- methoxypropan-2-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (69.1 mg, 120.47 umol, 58.3% yield, 97.060% purity) was obtained as a white solid. SFC: R
t = 1.168 min, 100%.
1H NMR (400 MHz, METHANOL-d) δ = 7.21 (s, 1H), 7.17 (d, J = 7.4 Hz, 1H), 7.03 (dd, J = 2.1, 12.8 Hz, 1H), 6.37 (d, J = 7.3 Hz, 1H), 4.64 (s, 1H), 3.95 (d, J = 1.5 Hz, 3H), 3.50-3.33 (m, 6H), 3.28 (s, 3H), 3.02-2.86 (m, 3H), 2.85-2.73 (m, 1H), 2.72-2.63 (m, 4H), 2.54 (dt, J = 2.6, 7.7 Hz, 2H), 2.47 (s, 3H), 2.18-2.06 (m, 1H), 2.05-1.92 (m, 1H), 1.90-1.83 (m, 2H), 1.72-1.53 (m, 4H), 1.43-1.33 (m, 2H), 1.21 (d, J = 7.0 Hz, 3H). LCMS (ESI) m/z = 557.3 (M+1), R
t = 1.691 min, 97.060% 2-(3-fluoro-5-((S)-1-hydroxypropan-2-yl)-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 19A and 19B)
[0969] Crude 2-(3-fluoro-5-((S)-1-hydroxypropan-2-yl)-2-methoxyphenyl)-2-((R)-3- (methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was purified by prep-HPLC (column: Phenomenex luna C18250 x 50 mm x 10 um; mobile phase: [water (TFA 0.1%), B acetonitrile 1%-40%, 10 min; Flow Rate 25 mL/min) and then separated
by SFC (column: Regis (S,S) Whelk-O1250 mm x 25 mm, 10 um); mobile phase: [MeCN/EtOH = 1/1, (0.1% NH
3water)]; B%: 55%-55%, 15 min) to give two peaks: [0970] Compound 19A, arbitrarily assigned as (S)-2-(3-fluoro-5-((S)-1-hydroxypropan-2- yl)-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (85.7 mg, 1.11 mmol, 28.6% yield) was obtained as white solid. SFC: R
t = 1.651 min, 100.0%.
1H NMR (400 MHz, METHANOL-d) δ = 7.23 (s, 1H), 7.17 (d, J = 7.3 Hz, 1H), 7.05 (dd, J = 2.0, 12.8 Hz, 1H), 6.38 (d, J = 7.3 Hz, 1H), 4.65 (s, 1H), 3.96 (d, J = 1.5 Hz, 3H), 3.63-3.54 (m, 2H), 3.43-3.33 (m, 4H), 3.04-2.89 (m, 3H), 2.89-2.81 (m, 1H), 2.70 (br t, J = 6.3 Hz, 3H), 2.55 (br t, J = 7.8 Hz, 3H), 2.40 (s, 3H), 2.12 (dt, J = 5.9, 12.9 Hz, 1H), 1.95-1.85 (m, 3H), 1.73-1.51 (m, 4H), 1.44-1.36 (m, 2H), 1.23 (d, J = 7.0 Hz, 3H). LCMS (ESI) m/z = 543.3 (M+1). [0971] Compound 19B, arbitrarily assigned as (R)-2-(3-fluoro-5-((S)-1-hydroxypropan-2- yl)-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (64.3 mg, 118.49 umol, 21.4% yield) was obtained as white solid. SFC: Rt = 2.846 min, 98.7%.
1H NMR (400 MHz, METHANOL-d) δ = 7.22 (s, 1H), 7.16 (d, J = 7.3 Hz, 1H), 7.03 (dd, J = 1.8, 12.8 Hz, 1H), 6.37 (d, J = 7.3 Hz, 1H), 4.64 (s, 1H), 3.95 (d, J = 1.4 Hz, 3H), 3.65-3.53 (m, 2H), 3.42-3.35 (m, 3H), 3.23 (dt, J = 4.9, 9.0 Hz, 1H), 3.04-2.92 (m, 2H), 2.88-2.80 (m, 1H), 2.79-2.63 (m, 5H), 2.56-2.50 (m, 2H), 2.45 (s, 3H), 2.17- 2.07 (m, 1H), 2.03-1.94 (m, 1H), 1.91-1.84 (m, 2H), 1.72-1.57 (m, 4H), 1.42-1.34 (m, 2H), 1.22 (d, J = 7.0 Hz, 3H). LCMS (ESI) m/z = 543.3 (M+1). 2-(3-fluoro-5-((R)-1-hydroxypropan-2-yl)-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 19C and 19D)

[0972] Crude 2-(3-fluoro-5-((R)-1-hydroxypropan-2-yl)-2-methoxyphenyl)-2-((R)-3- (methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was purified by prep-HPLC (column: Phenomenex Luna C18250 x 50 mm x 10 um; mobile phase: A water (TFA 0.1%), B acetonitrile 1%-40%, 10 min; Flow Rate 25 mL/min). The residue was
separated by SFC (column: Regis (S,S) Whelk-O1 (250 mm x 25 mm, 10 um); mobile phase: [MeCN/EtOH = 1:1, (0.1% NH
3water)]; B%: 55%-55%, 15 min) to give two peaks: [0973] Compound 19C, arbitrarily assigned as (S)-2-(3-fluoro-5-((R)-1-hydroxypropan-2- yl)-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (800 mg, 1.47 mmol, 6.9% yield) was obtained as a white solid. SFC: R
t = 1.676 min, 100.0%.
1H NMR (400 MHz, METHANOL-d) δ = 7.25 (s, 1H), 7.17 (d, J = 7.4 Hz, 1H), 7.04 (dd, J = 2.0, 12.8 Hz, 1H), 6.37 (d, J = 7.3 Hz, 1H), 4.62 (s, 1H), 3.96 (d, J = 1.6 Hz, 3H), 3.65-3.53 (m, 2H), 3.41-3.35 (m, 3H), 3.30-3.24 (m, 1H), 3.00-2.81 (m, 4H), 2.70 (br t, J = 6.2 Hz, 3H), 2.60-2.51 (m, 3H), 2.40 (s, 3H), 2.17-2.07 (m, 1H), 1.95-1.84 (m, 3H), 1.71-1.53 (m, 4H), 1.39 (quin, J = 7.3 Hz, 2H), 1.26-1.20 (m, 3H). LCMS (ESI) m/z = 543.3 (M+1). [0974] Compound 19D, arbitrarily assigned as (S)-2-(3-fluoro-5-((R)-1-hydroxypropan-2- yl)-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (167.6 mg, 308.84 umol, 21.0% yield) was obtained as a white solid. SFC: Rt = 2.754 min, 98.1%.
1H NMR (400 MHz, METHANOL-d) δ = 7.54 (d, J = 7.3 Hz, 1H), 7.12-7.06 (m, 2H), 6.57 (d, J = 7.3 Hz, 1H), 4.83 (s, 1H), 3.95-3.92 (m, 3H), 3.91-3.84 (m, 1H), 3.59 (d, J = 6.9 Hz, 2H), 3.50-3.45 (m, 2H), 3.39-3.32 (m, 2H), 3.28-3.19 (m, 1H), 3.15-3.05 (m, 1H), 2.93-2.84 (m, 2H), 2.83-2.78 (m, 5H), 2.75-2.60 (m, 3H), 2.34-2.22 (m, 1H), 2.21-2.10 (m, 1H), 1.97-1.90 (m, 2H), 1.84-1.72 (m, 4H), 1.55-1.46 (m, 2H), 1.23 (d, J = 7.0 Hz, 3H). LCMS (ESI) m/z = 543.3 (M+1). 2-(3-fluoro-2-methoxy-5-(2,2,2-trifluoroethyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 20A and 20B)
[0975] The residue was purified by prep-HPLC (column: C18250 x 50 mm x 10 um; mobile phase: A water (NH
4HCO
310 mmol/L), B acetonitrile 25%-45%, 10 min; Flow Rate (25 mL/min). 2-(3-fluoro-2-methoxy-5-(2,2,2-trifluoroethyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (100 mg, 176.48 umol,
19.66% yield) was obtained as a yellow oil and was separated by SFC (column: DAICEL CHIRALPAK AD (250 mm x 30 mm x 10 um); mobile phase: [0.1% NH
3water EtOH]; B%: 20%-20%, 22 min) to give two peaks: [0976] Compound 20A, arbitrarily assigned as (S)-2-(3-fluoro-2-methoxy-5-(2,2,2- trifluoroethyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (26.4 mg, 46.59 umol, 52.80% yield) was obtained as a white solid. SFC: R
t = 1.062 min, 100%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.36 (s, 1H), 7.18 (d, J = 7.4 Hz, 1H), 7.13 (br d, J = 12.4 Hz, 1H), 6.38 (d, J = 7.3 Hz, 1H), 4.53 (s, 1H), 3.98 (d, J = 1.6 Hz, 3H), 3.49-3.41 (m, 3H), 3.38-3.35 (m, 2H), 3.24 (br dd, J = 3.5, 7.9 Hz, 1H), 2.84 (br dd, J = 7.1, 10.9 Hz, 3H), 2.77-2.61 (m, 4H), 2.59-2.53 (m, 2H), 2.48 (s, 3H), 2.14-2.05 (m, 1H), 1.93-1.82 (m, 3H), 1.73-1.54 (m, 4H), 1.47-1.36 (m, 2H). LCMS (ESI) m/z = 567.2 (M+1). [0977] Compound 20B, arbitrarily assigned as (R)-2-(3-fluoro-2-methoxy-5-(2,2,2- trifluoroethyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (32.5 mg, 57.36 umol, 65.00% yield) was obtained as a white solid. SFC: Rt = 1.214 min, 99.5%.
1H NMR (400 MHz, METHANOL-d4) δ = 7.33 (s, 1H), 7.16 (d, J = 7.3 Hz, 1H), 7.18-7.14 (m, 1H), 7.14-7.08 (m, 1H), 6.37 (d, J = 7.3 Hz, 1H), 4.56 (s, 1H), 3.97 (d, J = 1.8 Hz, 3H), 3.49-3.40 (m, 3H), 3.39-3.36 (m, 2H), 3.19-3.10 (m, 1H), 2.93-2.76 (m, 3H), 2.70 (br t, J = 6.2 Hz, 3H), 2.62-2.52 (m, 3H), 2.51 (s, 3H), 2.17-2.06 (m, 1H), 2.04-1.92 (m, 1H), 1.87 (quin, J = 5.9 Hz, 2H), 1.71-1.58 (m, 4H), 1.43-1.33 (m, 2H). LCMS (ESI) m/z = 567.2 (M+1). 2-(3-fluoro-5-(3-hydroxyoxetan-3-yl)-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 21A and 21B)
[0978] The residue was purified by prep-HPLC (column: Waters Xbridge BEH C18100 x 30 mm x 10 um; mobile phase: A water (NH
4HCO
310 mmol/L), B acetonitrile 20%-40%, 8 min; Flow Rate (25 mL/min). 2-(3-fluoro-5-(3-hydroxyoxetan-3-yl)-2-methoxyphenyl)-2-((R)-3-
(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (100 mg, 179.64 umol, 31.05% yield) was obtained as a yellow oil and was separated by SFC (column: Regis (S,S) Whelk-O1 (250 mm x 30 mm, 5 um); mobile phase: [0.1% NH3water MeOH]; B%: 50%-50%, 22 min) to give two peaks: [0979] Compound 21A, arbitrarily assigned as (S)-2-(3-fluoro-5-(3-hydroxyoxetan-3-yl)-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (5.8 mg, 10.28 umol, 14.30% yield, 98.64% purity) was obtained as a white solid. SFC: R
t = 1.034 min, 98.64%.
1H NMR (400 MHz, METHANOL- d4) δ 7.68 (s, 1H), 7.37 (s, 1H), 7.19 (s, 1H), 6.39 (s, 1H), 4.81 (br d, J = 5.25 Hz, 4H), 4.62 (br s, 1H), 3.99 (s, 3H), 3.42-3.47 (m, 1H), 3.38 (br d, J = 5.75 Hz, 2H), 2.87-2.92 (m, 2H), 2.79-2.85 (m, 1H), 2.70 (br t, J = 6.32 Hz, 3H), 2.60-2.65 (m, 1H), 2.55 (br t, J = 7.75 Hz, 2H), 2.46 (br s, 3H), 2.07-2.13 (m, 1H), 1.84-1.91 (m, 3H), 1.66-1.72 (m, 2H), 1.55-1.63 (m, 2H), 1.37-1.43 (m, 2H), 1.29-1.32 (m, 1H). LCMS (ESI) m/z = 557.2 (M+1). [0980] Compound 21B, arbitrarily assigned as (R)-2-(3-fluoro-5-(3-hydroxyoxetan-3-yl)-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (15.1 mg, 26.98 umol, 25.04% yield, 99.48% purity) was obtained as a white solid. SFC: Rt = 1.553 min, 99.48%.
1H NMR (400 MHz, METHANOL- d4) δ 7.63 (s, 1H), 7.33-7.43 (m, 1H), 7.17 (d, J = 7.25 Hz, 1H), 6.37 (d, J = 7.25 Hz, 1H), 4.83 (br d, J = 3.25 Hz, 3H), 4.76 (d, J = 6.50 Hz, 1H), 4.66 (s, 1H), 3.98 (d, J = 1.63 Hz, 3H), 3.45 (br s, 1H), 3.36-3.39 (m, 2H), 3.19-3.27 (m, 1H), 2.94-3.03 (m, 1H), 2.76-2.93 (m, 2H), 2.70 (br t, J = 6.25 Hz, 3H), 2.52-2.59 (m, 2H), 2.52 (s, 3H), 2.08-2.17 (m, 1H), 1.98-2.06 (m, 1H), 1.85-1.91 (m, 2H), 1.58-1.71 (m, 4H), 1.35-1.42 (m, 2H), 1.29 (br d, J = 4.13 Hz, 1H). LCMS (ESI) m/z = 557.2 (M+1). 2-(2-ethoxy-3-fluoro-5-isopropylphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 22A and 22B)
[0981] Crude 2-(2-ethoxy-3-fluoro-5-isopropylphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was purified by prep-
HPLC (column: Waters Xbridge BEH C18100 x 30 mm x 10 um; mobile phase: A water (NH
4HCO
310 mmol/L), B acetonitrile 35%-55%, 8 min; Flow Rate (25 mL/min) to give two peaks: [0982] Compound 22A, arbitrarily assigned as (S)-2-(2-ethoxy-3-fluoro-5-isopropylphenyl)- 2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (88.5 mg, 161.41 umol, 36.37% yield, 98.62% purity) was obtained as a white solid. SFC: R
t = 1.661 min, 98.62%.
1H NMR (400 MHz, METHANOL-d
4), δ = 7.24 (s, 1H), 7.16 (d, J = 7.25 Hz, 1H), 7.00 (dd, J = 12.69, 1.94 Hz, 1H), 6.36 (d, J = 7.25 Hz, 1H), 4.66-4.72 (m, 1H), 4.14- 4.24 (m, 2H), 3.36-3.40 (m, 3H), 3.21-3.26 (m, 1H), 2.96 (br d, J = 4.38 Hz, 2H), 2.85 (dt, J = 13.79, 6.93 Hz, 2H),, 2.69-2.74 (m, 3H) 2.60-2.66 (m, 1H), 2.53 (br dd, J = 8.00, 5.75 Hz, 2H) 2.45 (s, 3H), 2.12 (br dd, J = 13.32, 5.32 Hz, 1H), 1.99 (br dd, J = 13.26, 5.88 Hz, 1H), 1.86-1.91 (m, 2H), 1.64-1.70 (m, 2H), 1.54-1.61 (m, 2H), 1.41 (t, J = 7.07 Hz, 3H), 1.32-1.39 (m, 2H), 1.21 (dd, J = 6.94, 1.94 Hz, 6H). LCMS: (ESI) m/z = 541.4 (M+1), R
t = 2.591 min, 100.0% [0983] Compound 22B, arbitrarily assigned as (R)-2-(2-ethoxy-3-fluoro-5- isopropylphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (28.1 mg, 51.97 umol, 46.83% yield, 100% purity) was obtained as a white solid. SFC: R
t = 0.977 min, 100.0%.
1H NMR (400 MHz, METHANOL- d4), δ 7.27 (s, 1H), 7.17 (br d, J = 7.25 Hz, 1H), 7.02 (dd, J = 12.76, 1.63 Hz, 1H), 6.37 (d, J = 7.25 Hz, 1H), 4.86 (s, 9H), 4.67 (s, 1H), 4.17-4.25 (m, 2H), 3.36-3.40 (m, 3H), 2.83-3.01 (m, 5H), 2.70 (br t, J = 6.07 Hz, 3H), 2.54 (br t, J = 7.69 Hz, 3H), 2.39 (s, 3H), 2.12 (br dd, J = 12.19, 5.19 Hz, 1H), 1.85-1.92 (m, 3H), 1.65-1.70 (m, 2H), 1.54-1.61 (m, 2H), 1.41-1.45 (m, 3H), 1.38 (br d, J = 7.38 Hz, 2H), 1.22 (d, J = 6.88 Hz, 6H). LCMS: (ESI) m/z = 541.4 (M+1), R
t = 2.669 min, 100.0%. 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-((5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)(2-(trifluoromethoxy)ethyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 23A and 23B)
[0984] The residue was purified by prep-HPLC (column: Phenomenex Luna 80 x 30 mm x 3 um; mobile phase: A water (TFA 0.1%), B acetonitrile 20%-40%, 8 min; Flow Rate 25 mL/min). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-((5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)(2-(trifluoromethoxy)ethyl)amino)pyrrolidin-1-yl)acetic acid (170 mg, 272.13 umol, 59.45% yield) was obtained as a yellow oil and then was separated by SFC (column: Regis (S,S) Whelk-O1 (250 mm x 30 mm x 5 um);mobile phase: [0.1%NH
3water EtOH]; B%: 50%-50%, 11 min). Compound 23A (peak 1) was further purified by prep-HPLC (column: C18-1150 x 30 mm x 5 um; mobile phase: A water (TFA 0.1%), B acetonitrile 25%-70%, 8 min; Flow Rate 25 mL/min). Compound 23B (peak 2) was further purified by prep-HPLC (column: C18-1150 x 30 mm x 5 um; mobile phase: A water (TFA 0.1%), B acetonitrile 5%-50%, 8 min; Flow Rate (25 mL/min). [0985] Compound 23A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-((5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)(2- (trifluoromethoxy)ethyl)amino)pyrrolidin-1-yl)acetic acid (15.8 mg, 25.29 umol, 22.57% yield) was obtained as a white solid. SFC: Rt = 1.489 min, 100%.
1H NMR (400 MHz, METHANOL- d4) δ = 7.56 (d, J = 7.4 Hz, 1H), 7.24-7.14 (m, 2H), 6.59 (d, J = 7.3 Hz, 1H), 5.11 (s, 1H), 4.15 (br t, J = 5.3 Hz, 2H), 3.99 (d, J = 2.0 Hz, 3H), 3.83 (td, J = 7.3, 14.3 Hz, 1H), 3.61 (br s, 1H), 3.52-3.42 (m, 3H), 3.21-3.09 (m, 3H), 3.05 (br s, 1H), 2.91 (td, J = 6.9, 13.8 Hz, 1H), 2.85-2.69 (m, 6H), 2.24 (br dd, J = 7.6, 12.8 Hz, 1H), 2.05-1.91 (m, 3H), 1.77-1.63 (m, 3H), 1.60-1.45 (m, 3H), 1.24 (d, J = 6.9 Hz, 6H). LCMS (ESI) m/z = 625.3 (M+1). [0986] Compound 23B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-((5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)(2- (trifluoromethoxy)ethyl)amino)pyrrolidin-1-yl)acetic acid (37.7 mg, 60.35 umol, 53.86% yield) was obtained as a white solid. SFC: R
t =1.711 min, 99.6%.
1H NMR (400 MHz, METHANOL- d
4) δ = 7.57 (d, J = 7.4 Hz, 1H), 7.19 (dd, J = 1.9, 13.0 Hz, 1H), 7.12 (s, 1H), 6.59 (d, J = 7.3 Hz, 1H), 5.17 (s, 1H), 4.17 (t, J = 5.2 Hz, 2H), 3.97 (d, J = 2.0 Hz, 3H), 3.81 (quin, J = 6.7 Hz, 1H), 3.51-3.37 (m, 4H), 3.18 (br s, 1H), 3.12 (br d, J = 5.1 Hz, 1H), 3.04-2.74 (m, 7H), 2.74-2.66 (m, 2H), 2.25-2.15 (m, 1H), 2.13-2.03 (m, 1H), 1.94 (td, J = 6.1, 11.6 Hz, 2H), 1.79-1.69 (m, 2H), 1.62 (qd, J = 6.7, 14.0 Hz, 2H), 1.51-1.40 (m, 2H), 1.24 (d, J = 6.9 Hz, 6H). LCMS (ESI) m/z = 625.3 (M+1).
2-(2-(difluoromethoxy)-3-fluoro-5-isopropylphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 24A and 24B)

[0987] The residue of 2-(2-(difluoromethoxy)-3-fluoro-5-isopropylphenyl)-2-((R)-3- (methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was purified by prep-HPLC (column: Phenomenex Luna C1875 x 30 mm x 3 um; mobile phase: A water (TFA 0.1%), B acetonitrile 20%-45%, 7 min; Flow Rate 25 mL/min) to give two peaks, Compound 24A (peak 1) was further purified by prep-HPLC (column: Phenomenex Luna C18 75 x 30 mm x 3 um; mobile phase: A water (FA 0.2%), B acetonitrile 10%-50%, 8 min; Flow Rate 25 mL/min). [0988] Compound 24A, arbitrarily assigned as (S)-2-(2-(difluoromethoxy)-3-fluoro-5- isopropylphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (15.1 mg, 26.84 umol, 7.5% yield, 100% purity) was obtained as white-solid. SFC: R
t = 1.142 min, 98.5%.
1H NMR (400 MHz, METHANOL-d) δ = 7.45 (d, J = 7.3 Hz, 1H), 7.17-7.09 (m, 1H), 7.02 (s, 1H), 6.70 (s, 1H), 6.50 (d, J = 7.3 Hz, 1H), 4.93 (s, 1H), 3.89-3.72 (m, 1H), 3.71-3.66 (m, 1H), 3.58 (br d, J = 11.8 Hz, 1H), 3.39 (br t, J = 5.4 Hz, 2H), 3.18-3.03 (m, 2H), 2.93 (quind, J = 7.0, 13.8 Hz, 1H), 2.81-2.72 (m, 6H), 2.60 (dt, J = 4.1, 12.5 Hz, 1H), 2.36-2.25 (m, 2H), 2.12-2.02 (m, 2H), 1.94-1.83 (m, 4H), 1.78 (dt, J = 6.0, 11.9 Hz, 2H), 1.72-1.58 (m, 2H), 1.25 (dd, J = 2.6, 6.9 Hz, 6H). LCMS (ESI) m/z = 563.3 (M+1), R
t = 1.866 min, 100%. [0989] Compound 24B, arbitrarily assigned as (R)-2-(2-(difluoromethoxy)-3-fluoro-5- isopropylphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (34.1 mg, 57.20 umol, 15.9% yield, 94.4% purity) was obtained as white-solid. SFC: Rt = 1.138 min, 99.1%.
1H NMR (400 MHz, METHANOL-d) δ = 7.56 (d, J = 7.3 Hz, 1H), 7.30 (s, 1H), 7.21 (dd, J = 1.4, 11.8 Hz, 1H), 7.04-6.65 (m, 1H), 6.60 (d, J = 7.4 Hz, 1H), 4.69 (s, 1H), 4.04 (br s, 1H), 3.58 (br d, J = 10.9 Hz, 1H), 3.48 (br t, J = 5.5 Hz, 2H), 3.27 (br d, J = 3.1 Hz, 1H), 3.17 (br s, 1H), 3.03-2.95 (m, 1H), 2.95-2.84 (m, 5H), 2.81 (br t, J = 6.1 Hz, 2H), 2.73 (br t, J = 7.7 Hz, 2H), 2.58 (q, J = 8.6 Hz, 1H), 2.36-2.26 (m, 1H),
2.15-2.04 (m, 1H), 1.94 (quin, J = 5.8 Hz, 2H), 1.78 (br d, J = 7.6 Hz, 4H), 1.50 (quin, J = 7.2 Hz, 2H), 1.24 (d, J = 6.9 Hz, 6H).LCMS (ESI) m/z = 563.3 (M+1), R
t = 1.895 min, 94.380%. 2-((S)-3-((3,3-difluoropropyl)(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid (Compounds 25A and 25B)

[0990] The crude product was purified by prep-HPLC (column: C18-1150 x 30 mm x 5 um; mobile phase: A water (TFA 0.1%), B acetonitrile 5%-50%, 8 min; Flow Rate 25 mL/min) to give 2-((S)-3-((3,3-difluoropropyl)(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid (0.12 g, 203.14 umol, 59.51% yield, 100% purity) as a yellow oil. which was separated by SFC (column: Regis (S,S) Whelk-O1 (250 mm x 30 mm, 5um); mobile phase: [0.1% NH3water EtOH]; B%: 50%-50%, 13 min) to give two peaks: [0991] Compound 25A, arbitrarily assigned as (S)-2-((S)-3-((3,3-difluoropropyl)(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)acetic acid (51.5 mg, 86.83 umol, 61.06% yield, 99.60% purity), obtained as a white solid. SFC: Rt = 1.561 min, 100.00%.
1H NMR (400 MHz, METHANOL-d4) δ = 7.21 (s, 1H), 7.19-7.14 (m, J = 7.3 Hz, 1H), 7.09 (dd, J = 1.8, 12.9 Hz, 1H), 6.39-6.35 (m, J = 7.3 Hz, 1H), 5.91 (tt, J = 4.5, 56.9 Hz, 1H), 4.86-4.85 (m, 1H), 3.99-3.96 (m, 3H), 3.56 (quin, J = 7.4 Hz, 1H), 3.42-3.32 (m, 4H), 3.05-2.95 (m, 2H), 2.88 (td, J = 6.9, 13.8 Hz, 1H), 2.75-2.66 (m, 4H), 2.59-2.42 (m, 4H), 2.12-1.86 (m, 6H), 1.70-1.57 (m, 2H), 1.48 (quin, J = 7.3 Hz, 2H), 1.34 (td, J = 7.0, 13.9 Hz, 2H), 1.23 (d, J = 6.8 Hz, 6H). LCMS (ESI) m/z = 591.2 (M+1). [0992] Compound 25B, arbitrarily assigned as (R)-2-((S)-3-((3,3-difluoropropyl)(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)acetic acid (21.0 mg, 35.35 umol, 58.01% yield, 99.44% purity), obtained as a white solid. SFC: R
t =1.809 min, 99.4%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.24 (s, 1H), 7.15 (d, J = 7.4 Hz, 1H), 7.09 (dd, J = 1.4, 12.9 Hz, 1H), 6.36 (d, J = 7.3 Hz, 1H), 6.05-5.73 (m,
1H), 4.80 (s, 1H), 3.98 (s, 3H), 3.58 (quin, J = 8.0 Hz, 1H), 3.40-3.35 (m, 2H), 3.25 (br d, J = 12.9 Hz, 2H), 3.15-3.02 (m, 2H), 2.88 (td, J = 6.8, 13.6 Hz, 1H), 2.72-2.62 (m, 4H), 2.57-2.41 (m, 4H), 2.03 (br d, J = 3.3 Hz, 6H), 1.69-1.60 (m, 2H), 1.47 (qd, J = 7.2, 14.4 Hz, 2H), 1.39-1.31 (m, 2H), 1.23 (d, J = 6.9 Hz, 6H). LCMS (ESI) m/z = 591 (M+1). 2-((R)-3-((3,3-difluoropropyl)(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid (Compounds 25C and 25D)

[0993] The crude product was purified by prep-HPLC (column: C18–1150 x 30 mm x 5 um; mobile phase: A water (formic acid 0.2%), B acetonitrile 5%-50%, 8 min); Flow Rate (25 mL/min) to give 2-((R)-3-((3,3-difluoropropyl)(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid (90 mg, 170.88 umol, 62.57% yield, 100.00% purity) as a yellow oil that was further separated by SFC (column: Regis (S,S) Whelk-O1 (250 mm x 30 mm x 5 um); mobile phase: [0.1% NH3water EtOH]; B%: 50%-50%, 15 min). [0994] Compound 25C, arbitrarily assigned as (S)-2-((R)-3-((3,3-difluoropropyl)(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)acetic acid (15.1 mg, 25.56 umol, 55.93% yield, 100% purity) was obtained as a white solid. SFC: R
t = 1.644 min, 99.92%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.24 (s, 1H), 7.15 (d, J = 7.3 Hz, 1H), 7.09 (dd, J = 1.8, 12.9 Hz, 1H), 6.36 (d, J = 7.3 Hz, 1H), 6.07-5.74 (m, 1H), 4.79 (s, 1H), 3.98 (d, J = 1.5 Hz, 3H), 3.63-3.52 (m, 1H), 3.39-3.35 (m, 2H), 3.28-3.17 (m, 2H), 3.15-3.02 (m, 2H), 2.88 (td, J = 6.9, 13.9 Hz, 1H), 2.73-2.61 (m, 4H), 2.58-2.41 (m, 4H), 2.08-1.82 (m, 6H), 1.65 (quin, J = 7.5 Hz, 2H), 1.54-1.42 (m, 2H), 1.39-1.30 (m, 2H), 1.23 (d, J = 6.9 Hz, 6H). LCMS (ESI) m/z = 591.2 (M+1). [0995] Compound 25D, arbitrarily assigned as (R)-2-((R)-3-((3,3-difluoropropyl)(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl- 2-methoxyphenyl)acetic acid (37.2 mg, 62.84 umol, 58.92% yield, 99.78% purity) was obtained as a white solid. SFC: Rt =1.944 min, 100.00%.
1H NMR (400 MHz, METHANOL-d4) δ = 7.21
(s, 1H), 7.17 (d, J = 7.3 Hz, 1H), 7.09 (dd, J = 1.6, 12.9 Hz, 1H), 6.37 (d, J = 7.4 Hz, 1H), 6.08- 5.76 (m, 1H), 4.86-4.85 (m, 1H), 3.98 (d, J = 1.0 Hz, 3H), 3.56 (quin, J = 7.4 Hz, 1H), 3.41-3.32 (m, 4H), 3.04-2.94 (m, 2H), 2.88 (td, J = 6.9, 13.8 Hz, 1H), 2.73-2.65 (m, 4H), 2.62-2.47 (m, 4H), 2.08-1.85 (m, 6H), 1.72-1.61 (m, 2H), 1.52-1.44 (m, 2H), 1.39-1.31 (m, 2H), 1.23 (d, J = 6.9 Hz, 6H). LCMS (ESI) m/z = 591.2 (M+1). 2-((R)-3-(((R)-1,1-difluoro-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2- yl)(methyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid (Compounds 26A and 26B)

[0996] The residue was purified by prep-HPLC (column: Phenomenex C1880 x 40 mm x 3 um; mobile phase: A water (NH4HCO310 mmol/L), B acetonitrile, 35%-55%, 8 min; Flow Rate (25 ml/min). 2-((R)-3-(((R)-1,1-difluoro-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2- yl)(methyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid (70 mg, 121.38 umol, 47.21% yield) was obtained as a yellow oil and was separated by SFC (column: DAICEL CHIRALPAK AD (250 mm x 30 mm, 10 um); mobile phase: [0.1% NH
3water EtOH]; B%: 27%-27%, 15 min). [0997] Compound 26A, arbitrarily assigned as (S)-2-((R)-3-(((R)-1,1-difluoro-6-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)hexan-2-yl)(methyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5- isopropyl-2-methoxyphenyl)acetic acid (23.3 mg, 40.21 umol, 66.26% yield, 99.53% purity) was obtained as a white solid. SFC: R
t = 1.067 min, 100%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.23 (s, 1H), 7.16 - 7.09 (m, 2H), 6.36 (d, J = 7.4 Hz, 1H), 5.99 - 5.69 (m, 1H), 4.87 (br s, 1H), 3.99 (d, J = 1.8 Hz, 3H), 3.74 (t, J = 7.6 Hz, 1H), 3.54 (br dd, J = 6.8, 10.9 Hz, 1H), 3.39 - 3.33 (m, 3H), 3.11 - 2.98 (m, 2H), 2.94 - 2.83 (m, 2H), 2.70 (t, J = 6.3 Hz, 2H), 2.58 - 2.49 (m, 2H), 2.35 (s, 3H), 2.11 - 1.98 (m, 2H), 1.92 - 1.84 (m, 2H), 1.73 - 1.57 (m, 3H), 1.53 - 1.45 (m, 2H), 1.40 - 1.33 (m, 1H), 1.23 (d, J = 6.9 Hz, 6H). LCMS (ESI) m/z = 577.2 (M+1). [0998] Compound 26B, arbitrarily assigned as (R)-2-((R)-3-(((R)-1,1-difluoro-6-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)hexan-2-yl)(methyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5- isopropyl-2-methoxyphenyl)acetic acid (21.8 mg, 37.80 umol, 62.29% yield, 100% purity) was
obtained as a white solid. SFC: R
t =1.302 min, 100%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.24 (s, 1H), 7.15 - 7.08 (m, 2H), 6.35 (d, J = 7.3 Hz, 1H), 5.99 - 5.68 (m, 1H), 4.88 (brs, 1H), 3.99 (d, J = 1.6 Hz, 3H), 3.73 (quin, J = 7.8 Hz, 1H), 3.40 - 3.35 (m, 2H), 3.30 - 3.23 (m, 2H), 3.16 - 3.04 (m, 2H), 2.98 - 2.85 (m, 2H), 2.69 (t, J = 6.3 Hz, 2H), 2.59 - 2.46 (m, 2H), 2.34 (s, 3H), 2.10 - 1.94 (m, 2H), 1.91 - 1.83 (m, 2H), 1.73 - 1.56 (m, 3H), 1.54 - 1.44 (m, 2H), 1.41 - 1.33 (m, 1H), 1.23 (dd, J = 1.2, 6.9 Hz, 6H). LCMS (ESI) m/z = 577.2 (M+1). 2-((S)-3-(((R)-1,1-difluoro-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2- yl)(methyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid (Compounds 26C and 26D)

[0999] Crude 2-((S)-3-(((R)-1,1-difluoro-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan- 2-yl)(methyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid was purified by prep-HPLC (column: Phenomenex C1880 x 40 mm x 3 um; mobile phase: A water (NH4HCO310 mmol/L), B acetonitrile, 40%-60%, 8 min; Flow Rate (25 ml/min), then was separated by SFC (column: Regis (S,S) Whelk-O1 (250 mm x 30 mm, 5 um); mobile phase: [0.1% NH
3water EtOH]; B%: 50%-50%, 10 min). [1000] Compound 26C was arbitrarily assigned as (S)-2-((S)-3-(((R)-1,1-difluoro-6- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2-yl)(methyl)amino)pyrrolidin-1-yl)-2-(3-fluoro- 5-isopropyl-2-methoxyphenyl)acetic acid (4.8 mg, 8.32 umol, 3.67% yield, 100% purity) was obtained as a white solid. SFC: Rt = 2.306 min, 100%.
1H NMR (400 MHz, METHANOL-d4) δ = 7.25 (s, 1H), 7.17 (d, J = 7.4 Hz, 1H), 7.09 (dd, J = 1.9, 12.9 Hz, 1H), 6.37 (d, J = 7.4 Hz, 1H), 6.00 - 5.68 (m, 1H), 4.80 (br s, 1H), 3.98 (d, J = 1.6 Hz, 3H), 3.71 (br t, J = 8.3 Hz, 1H), 3.40 - 3.36 (m, 2H), 3.29 - 3.15 (m, 2H), 3.11 - 2.98 (m, 2H), 2.93 - 2.78 (m, 2H), 2.70 (t, J = 6.2 Hz, 2H), 2.55 (br t, J = 6.7 Hz, 2H), 2.35 (s, 3H), 2.10 - 2.02 (m, 1H), 1.98 - 1.82 (m, 3H), 1.72 - 1.53 (m, 3H), 1.46 (br d, J = 3.1 Hz, 3H), 1.23 (d, J = 6.9 Hz, 6H). LCMS (ESI) m/z = 577.2 (M+1) [1001] Compound 26D was arbitrarily assigned as (R)-2-((S)-3-(((R)-1,1-difluoro-6- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2-yl)(methyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-
5-isopropyl-2-methoxyphenyl)acetic acid (16.7 mg, 28.29 umol, 12.76% yield, 97.68% purity) was obtained as a white solid. SFC: R
t = 1.682 min, 98.82%.
1H NMR (400 MHz, METHANOL- d4) δ = 7.24 (s, 1H), 7.19 - 7.10 (m, 2H), 6.39 (d, J = 7.4 Hz, 1H), 6.01 - 5.69 (m, 1H), 4.91 (s, 1H), 4.01 (s, 3H), 3.76 (quin, J = 7.6 Hz, 1H), 3.54 (br dd, J = 3.1, 10.4 Hz, 1H), 3.41 - 3.36 (m, 3H), 3.13 - 3.02 (m, 2H), 2.95 - 2.84 (m, 2H), 2.72 (t, J = 6.3 Hz, 2H), 2.55 (t, J = 7.5 Hz, 2H), 2.38 (s, 3H), 2.10 - 1.98 (m, 2H), 1.89 (quin, J = 5.9 Hz, 2H), 1.74 - 1.59 (m, 3H), 1.57 - 1.42 (m, 3H), 1.25 (d, J = 7.0 Hz, 6H). LCMS (ESI) m/z = 577.2 (M+1) 2-(3-fluoro-5-isopropyl-2-(trifluoromethoxy)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 27A and 27B)

[1002] Crude 2-(3-fluoro-5-isopropyl-2-(trifluoromethoxy)phenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was purified by prep-HPLC (column: Phenomenex Luna C1875 x 30 mm x 3 um; mobile phase: A water (FA 0.2%), B acetonitrile 10%-45%, 8 min); Flow Rate (25 mL/min) to give two peaks: [1003] Compound 27A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- (trifluoromethoxy)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (8.2 mg, 13.97 umol, 2.97% yield, 98.9% purity) was obtained as white-solid. SFC: Rt = 1.124 min, 99.7%.
1H NMR (400 MHz, METHANOL-d) δ = 7.45 (s, 1 H), 7.19 (d, J = 7.45 Hz, 1 H), 7.10 (br d, J = 11.40 Hz, 1 H), 6.38 (d, J = 7.45 Hz, 1 H), 4.38 (s, 1 H), 3.52-3.62 (m, 1 H), 3.38 (t, J = 5.48 Hz, 2 H), 2.97-3.10 (m, 2 H), 2.79-2.96 (m, 3 H), 2.68-2.74 (m, 2 H), 2.47-2.67 (m, 6 H), 2.41 (q, J = 8.11 Hz, 1 H), 2.07-2.17 (m, 1 H), 1.92- 2.05 (m, 1 H), 1.87 (quin, J = 5.86 Hz, 2 H), 1.59-1.76 (m, 4 H), 1.33-1.48 (m, 2 H), 1.24 (dd, J = 6.80, 1.75 Hz, 6 H). LCMS (ESI) m/z = 581.3 (M+1). [1004] Compound 27B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- (trifluoromethoxy)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (9.6 mg, 16.05 umol, 3.42% yield, 97.1% purity) was obtained as a yellow oil. SFC: R
t = 1.099 min, 100%.
1H NMR (400 MHz, METHANOL-d) δ = 8.44 (s, 1 H), 7.37-7.56 (m, 2 H), 7.14 (dd, J = 11.51, 1.64 Hz, 1 H), 6.49 (d, J = 7.23 Hz, 1 H), 4.18 (s, 1 H), 3.89-4.00 (m, 1 H), 3.78 (br d, J = 11.84 Hz, 1 H), 3.36 (q, J = 5.85 Hz, 4 H), 3.04-
3.17 (m, 1 H), 2.87-2.96 (m, 1 H), 2.83 (s, 3 H), 2.66-2.79 (m, 4 H), 2.45-2.66 (m, 2 H), 1.92- 2.20 (m, 4 H), 1.79-1.91 (m, 4 H), 1.56-1.76 (m, 2 H), 1.22 (dd, J = 6.80, 2.85 Hz, 6 H). LCMS (ESI) m/z = 581.3 (M+1). 2-(3-fluoro-2-methoxy-5-(3-methyloxetan-3-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compound 28A and 28B)

[1005] Crude 2-(3-fluoro-2-methoxy-5-(3-methyloxetan-3-yl)phenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was further purified by pre-HPLC (column: Waters Xbridge Prep OBD C18150 x 40 mm x 10 um; mobile phase: A water (NH4HCO310 mmol/L), B acetonitrile 25%-45%, 8 min; Flow Rate (25 mL/min) to give two peaks: [1006] Compound 28A, arbitrarily assigned as (S)-2-(3-fluoro-2-methoxy-5-(3- methyloxetan-3-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (52.9 mg, 95.37 umol, 45.78% yield, 100% purity) was obtained as white solid. SFC: Rt = 3.812 min, 88.34%.
1H NMR (400 MHz, METHANOL- d4) δ = 7.23 (s, 1H), 7.16 (d, J = 7.3 Hz, 1H), 7.07-7.01 (m, 1H), 6.37 (d, J = 7.3 Hz, 1H), 4.92 (d, J = 5.8 Hz, 1H), 4.89 (d, J = 5.6 Hz, 1H), 4.60 (d, J = 5.8 Hz, 3H), 3.96 (d, J = 1.6 Hz, 3H), 3.47- 3.40 (m, 1H), 3.39-3.36 (m, 2H), 3.19 (br d, J = 4.1 Hz, 1H), 2.97-2.86 (m, 2H), 2.84-2.75 (m, 1H), 2.70 (t, J = 6.1 Hz, 3H), 2.64 (br d, J = 8.8 Hz, 1H), 2.56-2.51 (m, 2H), 2.49 (s, 3H), 2.17- 2.08 (m, 1H), 2.03-1.94 (m, 1H), 1.90-1.85 (m, 2H), 1.70-1.65 (m, 5H), 1.63-1.57 (m, 2H), 1.41- 1.34 (m, 2H). LCMS (ESI) m/z = 555.3 (M+1), Rt = 1.670 min. [1007] Compound 28B, arbitrarily assigned as (R)-2-(3-fluoro-2-methoxy-5-(3- methyloxetan-3-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (27.2 mg, 47.96 umol, 23.02% yield, 97.801% purity) was obtained as white solid. SFC: R
t = 1.551 min, 98.62%.
1H NMR (400 MHz, METHANOL- d
4) δ = 7.27 (s, 1H), 7.18 (d, J = 7.4 Hz, 1H), 7.09-7.04 (m, 1H), 6.38 (d, J = 7.4 Hz, 1H), 4.93- 4.89 (m, 2H), 4.62-4.58 (m, 3H), 3.97 (d, J = 1.6 Hz, 3H), 3.46-3.39 (m, 1H), 3.38-3.36 (m, 2H),
3.29-3.23 (m, 1H), 2.94-2.76 (m, 4H), 2.70 (t, J = 6.2 Hz, 2H), 2.66-2.59 (m, 1H), 2.57-2.52 (m, 2H), 2.44 (s, 3H), 2.15-2.08 (m, 1H), 1.92-1.85 (m, 3H), 1.71 (br d, J = 1.6 Hz, 1H), 1.67 (s, 3H), 1.66-1.51 (m, 3H), 1.44-1.37 (m, 2H). LCMS (ESI) m/z = 555.3 (M+1), Rt = 1.671 min. 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((2S,3S)-2-methyl-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 29A and 29B)

[1008] The crude product was purified by prep-HPLC (column: Phenomenex Luna C1880 x 30 mm x 3 um; mobile phase: A water (TFA 0.1%), B acetonitrile 20%-50%, 8 min; Flow Rate 25 ml/min). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((2S,3S)-2-methyl-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (0.2 g, 369.88 umol, 68.59% yield) was obtained as a yellow oil and was separated SFC (column: DAICEL CHIRALPAK IG (250 mm x 30 mm x 10 um); mobile phase: [0.1% NH3water EtOH]; B%: 50%- 50%, 15 min). [1009] Compound 29A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((2S,3S)-2-methyl-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (52.3 mg, 96.72 umol, 52.30% yield, 100% purity) was obtained as a yellow solid. SFC: Rt = 1.667 min, 100%.
1H NMR (400 MHz, METHANOL- d4) δ = 7.30 (s, 1H), 7.17 - 7.01 (m, 2H), 6.35 (d, J = 7.3 Hz, 1H), 4.95 (br s, 1H), 4.01 (d, J = 1.5 Hz, 3H), 3.71 (br d, J = 4.3 Hz, 1H), 3.42 - 3.33 (m, 3H), 3.25 - 3.17 (m, 1H), 3.12 - 3.07 (m, 1H), 2.88 (td, J = 6.8, 13.8 Hz, 1H), 2.69 (t, J = 6.3 Hz, 2H), 2.51 (t, J = 7.6 Hz, 2H), 2.40 (br s, 2H), 2.23 (s, 3H), 2.20 - 2.11 (m, 1H), 1.98 - 1.80 (m, 3H), 1.64 (quin, J = 7.5 Hz, 2H), 1.58 - 1.45 (m, 2H), 1.36 - 1.29 (m, 5H), 1.22 (d, J = 7.0 Hz, 6H). LCMS: (ESI) m/z = 541.3 (M+1). [1010] Compound 29B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((2S,3S)-2-methyl-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (25.5 mg, 44.57 umol, 24.10% yield, 94.51% purity) was obtained as a white solid. SFC: R
t =1.923 min, 92.72%
1H NMR (400 MHz, METHANOL-
d
4) δ = 7.31 (s, 1H), 7.18 - 7.04 (m, 2H), 6.35 (d, J = 7.3 Hz, 1H), 4.99 (s, 1H), 4.00 (d, J = 1.6 Hz, 3H), 3.59 - 3.42 (m, 1H), 3.42 - 3.32 (m, 3H), 3.15 - 2.96 (m, 2H), 2.89 (td, J = 6.9, 13.8 Hz, 1H), 2.70 (t, J = 6.3 Hz, 2H), 2.50 (t, J = 7.6 Hz, 2H), 2.36 (br t, J = 7.4 Hz, 2H), 2.20 (s, 3H), 2.16 - 2.10 (m, 1H), 1.92 - 1.83 (m, 3H), 1.68 - 1.59 (m, 2H), 1.55 - 1.44 (m, 2H), 1.34 - 1.28 (m, 2H), 1.23 (dd, J = 1.4, 6.9 Hz, 6H), 1.18 (br d, J = 6.9 Hz, 3H). LCMS: (ESI) m/z = 541.3 (M+1). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((2R,3R)-2-methyl-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 30A and 30B)

[1011] The crude product was purified by prep-HPLC (column: Phenomenex Luna 80 x 30 mm x 3 um; mobile phase: A water (TFA 0.1%), B acetonitrile 20%-50%, 8 min; Flow Rate 25 ml/min). 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((2R,3R)-2-methyl-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (120 mg, 221.93 umol, 49.4% yield) was obtained as a yellow oil and was separated by SFC (column: DAICEL CHIRALCEL OD (250 mm x 30 mm x 10 um); mobile phase: [0.1% NH
3water EtOH]; B%: 37%-37%, 10 min). [1012] Compound 30A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((2R,3R)-2-methyl-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (15.7 mg, 29.04 umol, 26.17% yield, 100.00% purity) was obtained as a yellow solid. SFC: R
t = 1.191 min, 99.62%.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.31 (s, 1H), 7.20 - 7.05 (m, 2H), 6.36 (d, J = 7.4 Hz, 1H), 4.99 (s, 1H), 4.00 (d, J = 1.6 Hz, 3H), 3.64 - 3.44 (m, 1H), 3.42 - 3.35 (m, 2H), 3.35 - 3.32 (m, 1H), 3.19 - 2.98 (m, 2H), 2.89 (td, J = 6.9, 13.8 Hz, 1H), 2.70 (t, J = 6.2 Hz, 2H), 2.51 (br t, J = 7.6 Hz, 2H), 2.37 (br t, J = 7.1 Hz, 2H), 2.20 (s, 3H), 2.17 - 2.09 (m, 1H), 1.95 - 1.83 (m, 3H), 1.67 - 1.58 (m, 2H), 1.57 - 1.42 (m, 2H), 1.35 - 1.28 (m, 2H), 1.23 (dd, J = 1.5, 6.9 Hz, 6H), 1.18 (br d, J = 6.6 Hz, 3H). LCMS: (ESI) m/z = 541.2 (M+1). [1013] Compound 30B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((2R,3R)-2-methyl-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-
yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (39.1 mg, 72.31 umol, 65.17% yield, 100.00% purity) was obtained as a white solid. SFC: R
t =1.340 min, 100.00%.
1H NMR (400 MHz, METHANOL- d4) δ = 7.30 (s, 1H), 7.18 - 7.04 (m, 2H), 6.35 (d, J = 7.3 Hz, 1H), 4.95 (br s, 1H), 4.01 (d, J = 1.5 Hz, 3H), 3.69 (br d, J = 5.6 Hz, 1H), 3.40 - 3.35 (m, 2H), 3.32 (br s, 1H), 3.24 - 3.16 (m, 1H), 3.13 - 3.06 (m, 1H), 2.88 (td, J = 6.9, 13.8 Hz, 1H), 2.69 (t, J = 6.3 Hz, 2H), 2.51 (t, J = 7.6 Hz, 2H), 2.40 (br t, J = 6.9 Hz, 2H), 2.23 (s, 3H), 2.16 (br d, J = 4.6 Hz, 1H), 1.96 - 1.80 (m, 3H), 1.64 (br t, J = 7.6 Hz, 2H), 1.58 - 1.46 (m, 2H), 1.40 - 1.29 (m, 5H), 1.23 (d, J = 6.9 Hz, 6H). LCMS: (ESI) m/z = 541.3 (M+1) 2-(3-fluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4-yl)phenyl)-2-((R)-3-methyl-3- (methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 31A and 31B)

[1014] Crude 2-(3-fluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4-yl)phenyl)-2-((R)-3- methyl-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1- yl)acetic acid was purified by prep-HPLC (column: Waters Xbridge BEH C18100 x 30 mm x 10 um; mobile phase: A water (NH
4HCO
310 mmol/L), B acetonitrile, 30%-60%, 8 min; Flow Rate (25 mL/min) to give two peaks: [1015] Compound 31A, arbitrarily assigned as (S)-2-(3-fluoro-2-methoxy-5-(4- methyltetrahydro-2H-pyran-4-yl)phenyl)-2-((R)-3-methyl-3-(methyl(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (19.2 mg, 30.89 umol, 9.9% yield, 96% purity) was obtained as white solid. SFC: R
t = 0.870 min, 100.0%.
1H NMR (400 MHz, METHANOL-d) δ = 7.38-7.34 (m, 1H), 7.18-7.09 (m, 2H), 6.37 (d, J = 7.0 Hz, 1H), 4.64-4.41 (m, 1H), 3.95 (s, 3H), 3.79-3.70 (m, 2H), 3.67-3.56 (m, 2H), 3.37 (br t, J = 5.4 Hz, 2H), 3.08-2.98 (m, 1H), 2.96-2.82 (m, 1H), 2.80-2.80 (m, 1H), 2.76-2.63 (m, 3H), 2.57-2.47 (m, 6H), 2.26 (td, J = 7.0, 14.0 Hz, 1H), 2.08-2.01 (m, 2H), 1.89-1.84 (m, 3H), 1.75-1.63 (m, 7H), 1.42-1.35 (m, 2H), 1.26 (d, J = 7.9 Hz, 6H). LCMS (ESI) m/z = 597.3 (M+1), R
t = 1.754 min, 96.336% [1016] Compound 31B, arbitrarily assigned as (R)-2-(3-fluoro-2-methoxy-5-(4- methyltetrahydro-2H-pyran-4-yl)phenyl)-2-((R)-3-methyl-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-
naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (27.9 mg, 45.82 umol, 14.7% yield, 98% purity) was obtained as white solid. SFC, R
t = 0.816 min, 100%.
1H NMR (400 MHz, METHANOL-d) δ = 7.38 (s, 1H), 7.19-7.15 (m, 1H), 7.14-7.09 (m, 1H), 6.39 (d, J = 7.2 Hz, 1H), 4.52-4.47 (m, 1H), 3.96 (s, 3H), 3.78-3.71 (m, 2H), 3.60 (dt, J = 3.4, 7.6 Hz, 2H), 3.39-3.33 (m, 3H), 2.90-2.77 (m, 2H), 2.70 (br t, J = 6.2 Hz, 3H), 2.61-2.52 (m, 6H), 2.15-2.09 (m, 1H), 2.09- 2.02 (m, 2H), 1.87 (td, J = 5.6, 11.5 Hz, 3H), 1.80-1.58 (m, 7H), 1.48-1.41 (m, 2H), 1.26 (d, J = 7.9 Hz, 6H). LCMS (ESI) m/z = 597.3 (M+1), R
t = 1.758 min, 98.333% 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((S)-3-(methyl(4-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)butyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 32A and 32B)
[1017] Crude 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((S)-3-(methyl(4-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)butyl)amino)pyrrolidin-1-yl)acetic acid was purified by prep- HPLC (column: Phenomenex luna C18100 x 40 mm x 3 um; mobile phase: A water with 0.1% TFA; B acetonitrile 1%-45%, 8 min; Flow Rate 25 mL/min) to give two peaks: [1018] Compound 32A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((S)-3-(methyl(4-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)butyl)amino)pyrrolidin-1-yl)acetic acid (11.1 mg, 21.65 umol, 10.57% yield) was obtained as a yellow solid.
1H NMR (400 MHz, METHANOL-d
4) δ ppm 7.26 (s, 1 H), 7.12 (d, J = 7.25 Hz, 1 H), 6.97-7.06 (m, 1 H), 6.35 (d, J = 7.25 Hz, 1 H), 4.62 (br s, 1 H), 3.94 (d, J = 1.38 Hz, 3 H), 3.35-3.39 (m, 3 H), 3.21-3.26 (m, 1 H), 2.96-3.12 (m, 1 H), 2.86 (dt, J = 13.88, 7.07 Hz, 2 H), 2.75 (br d, J = 8.00 Hz, 1 H), 2.69 (t, J = 6.32 Hz, 2 H), 2.56-2.66 (m, 2 H), 2.53 (t, J = 7.32 Hz, 2 H), 2.39 (s, 3 H), 2.08-2.16 (m, 1 H), 1.92-2.01 (m, 1 H), 1.88 (dd, J = 11.51, 5.50 Hz, 2 H), 1.54-1.66 (m, 4 H), 1.29 (s, 1 H), 1.21 (dd, J = 6.88, 1.75 Hz, 6 H). LCMS (ESI) m/z = 513.2 (M+1), R
t = 1.691 min, 96.41%. SFC: R
t = 0.593 min, 100.0%. [1019] Compound 32B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((S)-3-(methyl(4-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)butyl)amino)pyrrolidin-1-yl)acetic acid (5.6 mg, 10.92 umol, 12.44% yield) was obtained as a
yellow solid.
1H NMR (400 MHz, METHANOL-d
4) δ ppm 7.22 (br s, 1 H), 7.14 (d, J = 7.38 Hz, 1 H), 7.05 (dd, J = 12.95, 1.56 Hz, 1 H), 6.37 (d, J = 7.25 Hz, 1 H), 4.71 (br s, 1 H), 3.95 (s, 3 H), 3.36-3.39 (m, 3 H), 3.26 (br dd, = 6.82, 3.06 Hz, 2 H), 2.95-3.04 (m, 2 H), 2.81-2.92 (m, 1 H), 2.65-2.73 (m, 3 H), 2.54 (br t, J = 7.32 Hz, 3 H), 2.38 (br s, 3 H), 2.06-2.14 (m, 1 H), 1.92 (s, 1 H), 1.87 (dt, J = 11.69, 6.03 Hz, 2 H), 1.60-1.69 (m, 2 H), 1.54-1.60 (m, 2 H), 1.22 (d, J = 6.88 Hz, 6 H). LCMS (ESI) m/z = 513.2 (M+1), R
t = 1.764 min, 98.36%. SFC: R
t = 1.422 min, 100.0%. 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(4-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)butyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 33A and 33B)
[1020] Crude 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(4-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)butyl)amino)pyrrolidin-1-yl)acetic acid was purified by prep- HPLC (column: Phenomenex Luna 80 x 30 mm x 3 um; mobile phase: A water with 0.1% TFA; B acetonitrile 10%-40%, 8 min; Flow Rate 25 mL/min) and then separated by SFC (column: DAICEL CHIRALPAK IG (250 mm x 30 mm, 10 um); mobile phase: EtOH with 0.1% ammonium hydroxide; B%: 50%-50%, 18 min) to give two peaks: [1021] Compound 33A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl (4-(5, 6, 7, 8-tetrahydro-1, 8-naphthyridin-2-yl) butyl) amino) pyrrolidin-1-yl) acetic acid (42 mg, 81.93 umol, 17.87% yield) was obtained as a light yellow solid.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.24 (s, 1H), 7.14 (d, J = 7.3 Hz, 1H), 7.06 (dd, J = 2.0, 12.9 Hz, 1H), 6.37 (d, J = 7.4 Hz, 1H), 4.73 (s, 1H), 3.96 (d, J = 1.6 Hz, 3H), 3.40-3.35 (m, 3H), 3.29-3.23 (m, 1H), 3.05 (br dd, J = 7.3, 11.6 Hz, 3H), 2.87 (td, J = 6.8, 13.8 Hz, 1H), 2.71-2.61 (m, 3H), 2.54 (t, J = 7.4 Hz, 3H), 2.37 (s, 3H), 2.21-2.10 (m, 1H), 1.97-1.90 (m, 1H), 1.90-1.84 (m, 2H), 1.69-1.62 (m, 2H), 1.60-1.51 (m, 2H), 1.30 (br s, 1H), 1.22 (d, J = 6.9 Hz, 6H). LCMS (ESI) m/z = 513.2 (M+1), Rt = 1.722 min, 97.05%. SFC: Rt = 1.767 min, 100.00%. [1022] Compound 33B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl (4-(5, 6, 7, 8-tetrahydro-1, 8-naphthyridin-2-yl) butyl) amino)
pyrrolidin-1-yl) acetic acid (82.5 mg, 160.93 umol, 35.11% yield) was obtained as a light yellow solid.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.25 (s, 1H), 7.14 (d, J = 7.4 Hz, 1H), 7.03 (dd, J = 1.9, 12.8 Hz, 1H), 6.36 (d, J = 7.4 Hz, 1H), 4.66 (s, 1H), 3.95 (d, J = 1.3 Hz, 3H), 3.39-3.35 (m, 3H), 3.26 (br s, 1H), 3.17-3.02 (m, 1H), 2.92 (br dd, J = 5.4, 11.8 Hz, 1H), 2.88-2.82 (m, 1H), 2.81-2.74 (m, 1H), 2.72-2.61 (m, 4H), 2.54 (t, J = 7.3 Hz, 2H), 2.42 (s, 3H), 2.19-2.10 (m, 1H), 2.05-1.96 (m, 1H), 1.87 (quin, J = 5.9 Hz, 2H), 1.69-1.55 (m, 4H), 1.21 (dd, J = 1.9, 6.9 Hz, 6H). LCMS (ESI) m/z = 513.2 (M+1), R
t = 1.708 min, 98.42%. SFC: R
t = 2.143 min, 100.00%. 2-(2-ethyl-3-fluoro-5-isopropylphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 34A and 34B)
[1023] Crude 2-(2-ethyl-3-fluoro-5-isopropylphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was purified by prep-HPLC (column: Phenomenex Luna 250 x 50 mm x 10 um; mobile phase: A water with 0.1% TFA; B acetonitrile 5%-40%, 10 min; Flow Rate 25 mL/min) and separated by SFC (column: (s,s) WHELK-O1 (250 mm x 30 mm, 5 um); mobile phase: EtOH with 0.1% ammonium hydroxide; B%: 50%-50%,14 min) to give two peaks: [1024] Compound 34A, arbitrarily assigned as (S)-2-(2-ethyl-3-fluoro-5-isopropylphenyl)- 2-((R)-3-(methyl (5-(5, 6, 7, 8-tetrahydro-1, 8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1- yl)acetic acid (100.3 mg, 191.15 umol, 50.15% yield) was obtained as a light yellow solid.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.34 (s, 1H), 7.21 (d, J = 7.3 Hz, 1H), 6.86 (dd, J = 1.6, 11.4 Hz, 1H), 6.39 (d, J = 7.3 Hz, 1H), 4.24 (br s, 1H), 3.55 (br s, 1H), 3.39-3.33 (m, 3H), 3.01- 2.94 (m, 1H), 2.90-2.75 (m, 4H), 2.70 (br t, J = 6.1 Hz, 4H), 2.62-2.50 (m, 6H), 2.12-2.04 (m, 1H), 1.97-1.90 (m, 1H), 1.89-1.83 (m, 2H), 1.77-1.68 (m, 3H), 1.64-1.58 (m, 1H), 1.50-1.42 (m, 2H), 1.22 (d, J = 6.9 Hz, 6H), 1.20-1.16 (m, 3H). LCMS (ESI) m/z = 536.4 (M+1), Rt = 1.921 min, 97.36%. SFC: Rt = 1.670 min, 100.00%. [1025] Compound 34B, arbitrarily assigned as (R)-2-(2-ethyl-3-fluoro-5-isopropylphenyl)- 2-((R)-3-(methyl (5-(5, 6, 7, 8-tetrahydro-1, 8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-
yl)acetic acid (160 mg, 304.93 umol, 80.00% yield) was obtained as a light yellow solid.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.36 (d, J = 1.4 Hz, 1H), 7.15 (d, J = 7.3 Hz, 1H), 6.86 (dd, J = 1.6, 11.4 Hz, 1H), 6.36 (d, J = 7.4 Hz, 1H), 4.35 (s, 1H), 3.53-3.45 (m, 1H), 3.40-3.35 (m, 2H), 3.21-3.12 (m, 1H), 2.96-2.74 (m, 7H), 2.70 (t, J = 6.3 Hz, 2H), 2.64-2.59 (m, 1H), 2.57 (s, 3H), 2.56-2.51 (m, 2H), 2.21-2.12 (m, 1H), 2.09-2.00 (m, 1H), 1.90-1.84 (m, 2H), 1.71-1.59 (m, 4H), 1.41-1.33 (m, 2H), 1.23-1.19 (m, 9H). LCMS (ESI) m/z = 536.4 (M+1), R
t = 1.892 min, 100.00%. SFC: R
t = 2.236 min, 99.38%. Preparation of 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-((5-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 35A and 35B)
[1026] Step 1: 2-((R)-3-(((benzyloxy)carbonyl)(5-(5,6,7,8-tetrahydro-1,8-naphthyridin- 2-yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid
To a solution of benzyl (R)-pyrrolidin-3-yl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)carbamate (150 mg, 354.98 umol) in DCM (4 mL) and HFIP (0.8 mL) was added (3- fluoro-5-isopropyl-2-methoxyphenyl)boronic acid (127.95 mg, 603.46 umol), oxoacetic acid (78.84 mg, 1.06 mmol) and 4A MS (0.4 g). The mixture was stirred at 60 °C for 3 h, then was filtered and the filtrate was concentrated under vacuum. 2-((R)-3-(((benzyloxy)carbonyl)(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl- 2-methoxyphenyl)acetic acid (0.25 g, 386.52 umol, 81.87% yield) was obtained as yellow oil. LCMS (ESI) m/z = 647.4 (M+1). Step 2: 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-((5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid

[1027] To a solution of 2-((R)-3-(((benzyloxy)carbonyl)(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid (200 mg, 309.22 umol) in MeOH (10 mL) was added Pd/C (0.1 g, 10% purity) under argon. The mixture was stirred at 25 °C for 4 h under H2 (15 psi). The reaction mixture was filtered with celite pad and the filtrate was concentrated in vacuum to get a residue. The mixture was further purification by pre-HPLC (column: Phenomenex Luna 80 x 30 mm x 3 um; mobile phase: A water (TFA 0.1%), B MeCN 5%-40%, 8 min; Flow Rate 25 mL/min). The mixture was then separated by SFC (column: REGIS(S, S) WHELK-O1 (250 mm x 25 mm, 10 um); mobile phase: [ACN/EtOH = 1:1, with 0.1% ammonium hydroxide]; B%: 60%-60%, 8 min). [1028] Compound 35A, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-((5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin- 1-yl)acetic acid (17 mg, 33.16 umol, 13.60% yield) was obtained as white solid.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.26 (s, 1H), 7.14 (d, J = 7.3 Hz, 1H), 6.95 (dd, J = 1.9, 12.8 Hz, 1H), 6.38 (d, J = 7.3 Hz, 1H), 4.42 (s, 1H), 3.92 (d, J = 1.1 Hz, 3H), 3.59 (br s, 1H), 3.39-3.36 (m, 3H), 3.28-3.23 (m, 1H), 2.92-2.79 (m, 4H), 2.70 (t, J = 6.3 Hz, 2H), 2.60-2.50 (m, 3H), 2.21- 2.14 (m, 1H), 1.91-1.85 (m, 2H), 1.73-1.64 (m, 4H), 1.49-1.41 (m, 2H), 1.33-1.27 (m, 1H), 1.21 (d, J = 6.9 Hz, 6H). LCMS (ESI) m/z = 513.2 (M+1), R
t = 1.785 min, 100.0%. SFC: R
t = 1.024 min, 100.0%. [1029] Compound 35B, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-((5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin- 1-yl)acetic acid (107.1 mg, 208.91 umol, 85.68% yield) was obtained as white solid.
1H NMR (400 MHz, METHANOL-d4) δ = 7.55 (d, J = 7.3 Hz, 1H), 7.13-7.04 (m, 2H), 6.58 (d, J = 7.4 Hz, 1H), 4.78 (s, 1H), 3.93 (d, J = 1.4 Hz, 3H), 3.81-3.75 (m, 1H), 3.49-3.45 (m, 2H), 3.15 (br d, J = 12.4 Hz, 1H), 3.05-2.97 (m, 2H), 2.93-2.85 (m, 2H), 2.82-2.78 (m, 2H), 2.73-2.65 (m, 3H), 2.39- 2.29 (m, 1H), 2.08-2.00 (m, 1H), 1.94 (br t, J = 5.6 Hz, 2H), 1.79-1.68 (m, 4H), 1.55-1.45 (m, 2H), 1.30 (d, J = 6.4 Hz, 1H), 1.22 (dd, J = 1.5, 6.9 Hz, 6H). LCMS (ESI) m/z = 513.3 (M+1), Rt = 1.787 min, 100.0%. SFC: Rt = 2.016 min, 98.3%.
General Example of Alkylation and Hydrolysis Preparation of 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 100A and 100B)
Step 1: Ethyl 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetate
[1030] A mixture of (R)-N-methyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine (302 mg, 1.0 mmol), ethyl 2-bromo-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)acetate (333 mg, 1.0 mmol) and DIEA (390 mg, 3.0 mmol) in acetonitrile (5 mL) was stirred at 60 °C for 15 h. Solvent was removed in vacuo, and the residue was purified by silica gel column (DCM: MeOH 15:1) to give the desired product ethyl 2-(3-fluoro-5-isopropyl- 2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetate as a yellow oil (410 mg). Yield 74 % (ESI 555 (M+H) +). Step 2: 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid

[1031] Ethyl 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetate (410 mg, 0.74 mmol) was treated with lithium hydroxide monohydrate (311 mg, 7.4 mmol) in MeOH (2.5 mL) and water
(0.5 mL) at room temperature for 2 h. Solvent was removed in vacuo, and the residue was purified by Prep-HPLC A (30-60% acetonitrile) to give diastereomeric products Compound 100A (110 mg) and Compound 100B (109 mg) as white solids. [1032] Compound 100A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.25 (s, 1H), 7.17 (d, J=7.2 Hz, 1H), 7.01 (dd, J=12.8, 2.0 Hz, 1H), 6.37 (d, J=7.6 Hz, 1H), 4.62 (s, 1H), 3.95 (s, 3H), 3.42-3.38 (m, 3H), 3.22-3.18 (m, 1H), 3.02-2.51 (m, 10H), 2.47 (s, 3H), 2.20-1.85 (m, 4H), 1.70– 1.58 (m, 4H), 1.42–1.18 (m, 8H). LC/MS ESI 527 (M+H)
+. [1033] Compound 100B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.28 (s, 1H), 7.16 (d, J=7.2 Hz, 1H), 7.02 (dd, J=12.8, 2.0 Hz, 1H), 6.37 (d, J=7.6 Hz, 1H), 4.62 (s, 1H), 3.95 (s, 3H), 3.42-3.38 (m, 3H), 3.22-3.18 (m, 1H), 3.02-2.51 (m, 10H), 2.48 (s, 3H), 2.20-1.85 (m, 4H), 1.70– 1.58 (m, 4H), 1.42–1.18 (m, 8H). LC/MS ESI 527 (M+H)
+. The following compounds were prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified using the conditions shown: Preparation of 2-(3-fluoro-2-methoxy-5-(2-methoxypropan-2-yl)phenyl)-2-((R)-3- (isobutyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 101A and 101B)

[1034] 2-(3-fluoro-2-methoxy-5-(2-methoxypropan-2-yl)phenyl)-2-((R)-3-(isobutyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate
reagents and purified by Prep-HPLC A (33-65% acetonitrile) to give diastereomers Compound 101A (57 mg) and Compound 101B (48 mg) as white solids. [1035] Compound 101A, arbitrarily assigned as (S)-2-(3-fluoro-2-methoxy-5-(2- methoxypropan-2-yl)phenyl)-2-((R)-3-(isobutyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d4) δ 7.33 – 7.16 (m, 3H), 6.43 (d, J = 7.4 Hz, 1H), 3.98 (d, J = 2.0 Hz, 3H), 3.80 – 3.64 (m, 1H), 3.42 – 3.36 (m, 3H), 3.29-3.22 (m, 2H), 3.08 (s, 3H), 2.87 – 2.44 (m, 9H), 2.10 – 1.37 (m, 18H), 0.93 (d, J = 6.8 Hz, 6H). LC/MS ESI 599.4 (M+H)
+. [1036] Compound 101B, arbitrarily assigned as (R)-2-(3-fluoro-2-methoxy-5-(2- methoxypropan-2-yl)phenyl)-2-((R)-3-(isobutyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d4) δ 7.41 (d, J = 1.9 Hz, 1H), 7.32 – 7.17 (m, 2H), 6.42 (d, J = 7.2 Hz, 1H), 4.77 – 4.60 (m, 1H), 3.99 (d, J = 1.9 Hz, 3H), 3.85 – 3.72 (m, 1H), 3.41 – 3.33 (m, 1H), 3.05 (s, 3H), 3.00 – 2.85 (m, 2H), 2.76 – 2.36 (m, 7H), 2.13 – 1.35 (m, 18H), 0.93 (d, J = 6.5 Hz, 6H). LC/MS ESI 599.4 (M+H)
+. Preparation of 2-(5-(tert-butyl)-3-fluoro-2-methoxyphenyl)-2-((R)-3-(ethyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 102A and 102B)
[1037] 2-(5-(tert-butyl)-3-fluoro-2-methoxyphenyl)-2-((R)-3-(ethyl(5-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (33-65% acetonitrile) to give diastereomeric products Compound 102A (83 mg) and Compound 102B (51 mg) as white solids. [1038] Compound 102A, arbitrarily assigned as (S)-2-(5-(tert-butyl)-3-fluoro-2- methoxyphenyl)-2-((R)-3-(ethyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d4) δ 7.44 – 7.38 (m, 1H), 7.23 – 7.06 (m, 2H), 6.36 (d, J = 7.3 Hz, 1H), 4.57 (s, 1H), 3.93 (s, 3H), 3.68 – 3.58 (m,
1H), 3.43 – 3.34 (m, 2H), 3.26 – 3.16 (m, 1H), 3.04 – 2.74 (m, 6H), 2.73 – 2.67 (m, 2H), 2.63 – 2.46 (m, 3H), 1.91 – 1.81 (m, 2H), 1.73 – 1.47 (m, 4H), 1.41 – 1.31 (m, 2H), 1.28 (s, 9H), 1.15 (t, J = 7.2 Hz, 3H). LC/MS ESI 555.4 (M+H)
+. [1039] Compound 102B, arbitrarily assigned as (R)-2-(5-(tert-butyl)-3-fluoro-2- methoxyphenyl)-2-((R)-3-(ethyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d
4) δ 7.43 – 7.38 (m, 1H), 7.21 – 7.11 (m, 2H), 6.37 (d, J = 7.3 Hz, 1H), 4.60 (s, 1H), 3.95 (d, J = 1.6 Hz, 3H), 3.75 – 3.62 (m, 1H), 3.40 – 3.34 (m, 2H), 3.00 – 2.75 (m, 6H), 2.74 – 2.64 (m, 3H), 2.60 – 2.48 (m, 2H), 2.18 – 2.02 (m, 1H), 1.97 – 1.80 (m, 3H), 1.77 – 1.30 (m, 8H), 1.28 (s, 9H), 1.13 (t, J = 7.1 Hz, 3H). LC/MS ESI 555.4 (M+H)
+. Preparation of 2-(5-(tert-butyl)-3-fluoro-2-methoxyphenyl)-2-((R)-3-((5-(4-methoxy-5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)(methyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 103A and 103B)
[1040] 2-(5-(tert-butyl)-3-fluoro-2-methoxyphenyl)-2-((R)-3-((5-(4-methoxy-5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)(methyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% acetonitrile) to give the diastereoisomeric products Compound 103A (45 mg) and Compound 103B (44 mg) as white solids. [1041] Compound 103A, arbitrarily assigned as (S)-2-(5-(tert-butyl)-3-fluoro-2- methoxyphenyl)-2-((R)-3-((5-(4-methoxy-5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)(methyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d
4) δ 7.29 (d, J = 1.8 Hz, 1H), 7.15 (dd, J = 13.8, 2.4 Hz, 1H), 6.34 (s, 1H), 4.73 (s, 1H), 3.93 (d, J = 1.7 Hz, 3H), 3.89 (s, 3H), 3.48 – 3.37 (m, 1H), 3.23 – 3.10 (m, 2H), 3.08 – 2.94 (m, 1H), 2.88 – 2.53 (m, 8H), 2.49 (s, 3H), 2.12 – 1.95 (m, 2H), 1.88 – 1.79 (m, 2H), 1.77 – 1.60 (m, 4H), 1.53 – 1.42 (m, 2H), 1.29 (s, 9H). LC/MS ESI 571.0 (M+H)
+.
[1042] Compound 103B, arbitrarily assigned as (R)-2-(5-(tert-butyl)-3-fluoro-2- methoxyphenyl)-2-((R)-3-((5-(4-methoxy-5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)(methyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d4) δ 7.41 (dd, J = 2.4, 1.2 Hz, 1H), 7.16 (dd, J = 13.7, 2.4 Hz, 1H), 6.36 (s, 1H), 4.54 (s, 1H), 3.95 (d, J = 1.7 Hz, 3H), 3.59 – 3.41 (m, 2H), 3.09 – 2.95 (m, 1H), 2.89 – 2.78 (m, 2H), 2.74 – 2.53 (m, 7H), 2.45 (s, 3H), 2.14 – 2.02 (m, 1H), 2.00 – 1.67 (m, 7H), 1.64 – 1.46 (m, 4H), 1.28 (s, 9H). LC/MS ESI 5571.0 (M+H)
+. Preparation of 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-((5-(4-methoxy-5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)(methyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 104A and 104B)
[1043] 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-((5-(4-methoxy-5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)(methyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% acetonitrile) to give diastereomeric products Compound 104A (84 mg) and Compound 104B (89 mg) as white solids. [1044] Compound 104A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-((5-(4-methoxy-5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)(methyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d
4) δ 7.19 – 7.14 (m, 1H), 6.99 (dd, J = 12.9, 2.1 Hz, 1H), 6.30 (s, 1H), 4.66 (s, 1H), 3.93 (d, J = 1.5 Hz, 3H), 3.87 (s, 3H), 3.43 – 3.35 (m, 1H), 3.18 – 3.09 (m, 1H), 3.08 – 2.99 (m, 1H), 2.95 – 2.78 (m, 3H), 2.69 – 2.51 (m, 6H), 2.46 (s, 3H), 2.12 – 1.38 (m, 11H), 1.24 – 1.17 (m, 6H). LC/MS ESI 557.0 (M+H)
+. [1045] Compound 104B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-((5-(4-methoxy-5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)(methyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d
4) δ 7.28 – 7.23 (m, 1H), 7.04 – 6.97 (m, 1H), 6.32 (s, 1H), 4.54 (s, 1H), 3.94 (d, J = 1.5 Hz, 3H), 3.87 (s,
3H), 3.43 – 3.34 (m, 2H), 2.90 – 2.68 (m, 5H), 2.66 – 2.52 (m, 5H), 2.40 (s, 3H), 2.13 – 1.45 (m, 10H), 1.23 – 1.18 (m, 6H). LC/MS ESI 557.0 (M+H)
+. Preparation of 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-((2-methoxyethyl)(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 105A and 105B)
[1046] 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-((2-methoxyethyl)(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% acetonitrile) to give the diastereomers products Compound 105A (56 mg) and Compound 105B (87 mg) as white solids. [1047] Compound 105A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-((2-methoxyethyl)(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d
4) δ 7.23 – 7.14 (m, 2H), 7.05 (dd, J = 12.9, 2.2 Hz, 1H), 6.37 (d, J = 7.3 Hz, 1H), 4.78 (s, 1H), 3.96 (d, J = 1.6 Hz, 3H), 3.69 – 3.59 (m, 1H), 3.49 – 3.42 (m, 2H), 3.41 – 3.34 (m, 3H), 3.32 (s, 3H), 3.21 – 2.99 (m, 2H), 2.95 – 2.43 (m, 10H), 2.16 – 1.94 (m, 2H), 1.91 – 1.83 (m, 2H), 1.71 – 1.60 (m, 2H), 1.57 – 1.44 (m, 1H), 1.43 – 1.29 (m, 2H), 1.26 – 1.18 (m, 6H). LC/MS ESI 571.3 (M+H)
+. [1048] Compound 105B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-((2-methoxyethyl)(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d4) δ 7.29 – 7.22 (m, 1H), 7.15 (d, J = 7.3 Hz, 1H), 7.06 (dd, J = 13.0, 2.2 Hz, 1H), 6.35 (d, J = 7.3 Hz, 1H), 4.73 (s, 1H), 3.96 (d, J = 1.7 Hz, 3H), 3.64 (p, J = 7.6 Hz, 1H), 3.47 – 3.40 (m, 2H), 3.39 – 3.33 (m, 2H), 3.27 – 3.08 (m, 1H), 3.08 – 2.94 (m, 2H), 2.91 – 2.45 (m, 9H), 2.15 – 2.02 (m, 1H), 1.98 – 1.81 (m, 3H), 1.72 – 1.57 (m, 2H), 1.58 – 1.41 (m, 2H), 1.34 (h, J = 7.7, 6.9 Hz, 2H), 1.25 – 1.20 (m, 6H). LC/MS ESI 571.3 (M+H)
+.
Preparation of 2-((R)-3-(cyclopropyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid (Compounds 106A and 106B)
[1049] 2-((R)-3-(cyclopropyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% acetonitrile) , then Prep Chiral SFC B to give the diastereomers products Compound 106A (40 mg) and Compound 106B (21 mg) as white solids. [1050] Compound 106A, arbitrarily assigned as (R)-2-((R)-3-(cyclopropyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)acetic acid.
1H NMR (400 MHz, Methanol-d4) δ 7.26 (d, J = 2.2 Hz, 1H), 7.19 – 7.07 (m, 2H), 6.36 (d, J = 7.3 Hz, 1H), 4.86 (s, 1H), 4.00 (d, J = 1.9 Hz, 3H), 3.71 – 3.58 (m, 1H), 3.42 – 3.26 (m, 5H), 3.20 – 3.07 (m, 2H), 2.95 – 2.81 (m, 1H), 2.75 – 2.58 (m, 4H), 2.57 – 2.48 (m, 2H), 2.19 – 1.99 (m, 2H), 1.93 – 1.81 (m, 3H), 1.72 – 1.50 (m, 4H), 1.36 – 1.28 (m, 2H), 1.24 (d, J = 7.0 Hz, 6H), 0.61 – 0.32 (m, 4H). Chiral SFC B (25% MeOH): ee 100%, Rt = 1.42 min. LC/MS ESI 553.0 (M+H)
+. [1051] Compound 106B, arbitrarily assigned as (S)-2-((R)-3-(cyclopropyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)acetic acid.
1H NMR (400 MHz, Methanol-d4) δ 7.23 (d, J = 2.1 Hz, 1H), 7.15 (d, J = 7.3 Hz, 1H), 7.08 (dd, J = 12.9, 2.2 Hz, 1H), 6.36 (d, J = 7.3 Hz, 1H), 4.86 (s, 1H), 3.97 (d, J = 1.7 Hz, 3H), 3.68 – 3.58 (m, 1H), 3.54 – 3.43 (m, 1H), 3.40 – 3.23 (m, 4H), 3.17 – 2.97 (m, 2H), 2.92 – 2.82 (m, 1H), 2.75 – 2.58 (m, 4H), 2.56 – 2.46 (m, 2H), 2.14 – 1.99 (m, 2H), 1.91 – 1.80 (m, 3H), 1.71 – 1.51 (m, 4H), 1.36 – 1.25 (m, 2H), 1.25 – 1.18 (m, 6H), 0.61 – 0.31 (m, 4H). Chiral SFC B (25% MeOH): ee 95%, Rt = 3.17 min. LC/MS ESI 553.0 (M+H)
+.
Preparation of 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(2-(2-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)ethoxy)ethyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 107A and 107B)
[1052] 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(2-(2-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)ethoxy)ethyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% acetonitrile) to give the diastereomers products Compound 107A (53 mg) and Compound 107B (59 mg) as white solids. [1053] Compound 107A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(2-(2-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)ethoxy)ethyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d
4) δ 7.27 (s, 1H), 7.16 (d, J = 7.6 Hz, 1H), 7.04 (dd, J = 12.9, 2.1 Hz, 1H), 6.42 (d, J = 7.3 Hz, 1H), 4.67 (s, 1H), 3.97 (d, J = 1.9 Hz, 3H), 3.71 – 3.58 (m, 4H), 3.40 – 3.22 (m, 5H), 2.95 – 2.71 (m, 9H), 2.37 (s, 3H), 2.10 – 1.85 (m, 4H), 1.24 (d, J = 7.0 Hz, 6H). LC/MS ESI 529.0 (M+H)
+. [1054] Compound 107B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(2-(2-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)ethoxy)ethyl)amino)pyrrolidin-1-yl)acetic acid .
1H NMR (400 MHz, Methanol-d
4) δ 7.26 (s, 1H), 7.15 (d, J = 7.6 Hz, 1H), 7.04 (dd, J = 12.9, 2.1 Hz, 1H), 6.42 (d, J = 7.3 Hz, 1H), 4.73 (s, 1H), 3.98 (d, J = 1.9 Hz, 3H), 3.71 – 3.58 (m, 4H), 3.40 – 3.22 (m, 5H), 2.95 – 2.71 (m, 9H), 2.37 (s, 3H), 2.10 – 1.85 (m, 4H), 1.24 (d, J = 7.0 Hz, 6H). LC/MS ESI 529.0 (M+H)
+.
Preparation of 2-((R)-3-((2-cyclopropoxyethyl)(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid (Compounds 108A and 108B)
[1055] 2-((R)-3-((2-cyclopropoxyethyl)(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% acetonitrile) to give diastereomers products Compound 108A (50 mg) and Compound 108B (32 mg) as white solids. [1056] Compound 108A, arbitrarily assigned as (S)-2-((R)-3-((2-cyclopropoxyethyl)(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl- 2-methoxyphenyl)acetic acid.
1H NMR (400 MHz, Methanol-d4) δ 7.23 – 7.15 (m, 2H), 7.06 (dd, J = 12.9, 2.2 Hz, 1H), 6.37 (d, J = 7.4 Hz, 1H), 4.79 (s, 1H), 3.96 (d, J = 1.7 Hz, 3H), 3.66 – 3.54 (m, 3H), 3.40 – 3.34 (m, 3H), 3.29 – 2.98 (m, 3H), 2.94 – 2.44 (m, 10H), 2.14 – 1.81 (m, 4H), 1.72 – 1.57 (m, 2H), 1.51 (d, J = 7.2 Hz, 0H), 1.40 – 1.28 (m, 2H), 1.25 – 1.19 (m, 6H), 0.54 – 0.39 (m, 4H). LC/MS ESI 597.5 (M+H)
+. [1057] Compound 108B, arbitrarily assigned as (R)-2-((R)-3-((2-cyclopropoxyethyl)(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl- 2-methoxyphenyl)acetic acid.
1H NMR (400 MHz, Methanol-d4) δ 7.26 – 7.17 (m, 2H), 7.08 (dd, J = 12.9, 2.1 Hz, 1H), 6.39 (d, J = 7.3 Hz, 1H), 4.74 (s, 1H), 3.97 (d, J = 1.7 Hz, 3H), 3.73 – 3.62 (m, 1H), 3.60 – 3.53 (m, 2H), 3.45 – 3.34 (m, 3H), 3.09 – 2.49 (m, 12H), 1.98 – 1.81 (m, 3H), 1.75 – 1.33 (m, 6H), 1.25 – 1.17 (m, 6H), 0.52 – 0.39 (m, 4H). LC/MS ESI 597.4 (M+H)
+.
Preparation of (R)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(isobutyl(2-(2- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)ethoxy)ethyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 109A and 109B)

[1058] 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(isobutyl(2-(2-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)ethoxy)ethyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC B (30-60% acetonitrile) to give diastereomers products Compound 109A (45 mg) and Compound 109B (50 mg) as white solids. [1059] Compound 109A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(isobutyl(2-(2-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)ethoxy)ethyl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.25 (s, 1H), 7.18-7.09 (m, 2H), 6.41 (d, J=7.2 Hz, 1H), 4.82 (s, 1H), 4.00 (s, 3H), 3.72-3.38 (m, 9H), 3.02- 2.65 (m, 9H), 2.27 (d, J=7.2 Hz, 2H), 2.05-1.65 (m, 5H), 1.24–1.20 (m, 6H), 0.92–0.85 (m, 6H). LC/MS ESI 571 (M+H)
+. [1060] Compound 109B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(isobutyl(2-(2-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)ethoxy)ethyl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.51 (d, J=7.2 Hz, 1H), 7.27 (s, 1H), 7.16 (dd, J=13.2, 2.0 Hz, 1H), 6.61 (d, J=7.2 Hz, 1H), 4.95 (s, 1H), 4.00 (s, 3H), 3.72-3.38 (m, 9H), 3.02-2.65 (m, 9H), 2.27 (d, J=7.2 Hz, 2H), 2.05-1.65 (m, 5H), 1.24–1.20 (m, 6H), 0.92–0.85 (m, 6H). LC/MS ESI 571 (M+H)
+.
Preparation of 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((3R)-3-(methyl(6-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)hexan-2-yl)amino)pyrrolidin-1-yl)acetic acid (Compound 110A, Compound 110B/110C and Compound 110D)

[1061] 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((3R)-3-(methyl(6-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)hexan-2-yl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% acetonitrile) to give diastereomers products Compound 110A (12 mg), Compound 110B/110C (15 mg) and Compound 110D (17 mg) as white solids. [1062] Compound 110A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl((S)-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2- yl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.28 (s, 1H), 7.14 (d, J=7.2 Hz, 1H), 7.00 (dd, J=12.8, 2.0 Hz, 1H), 6.36 (d, J=7.2 Hz, 1H), 4.58 (s, 1H), 3.95 (s, 3H), 3.62- 3.38 (m, 3H), 3.25-2.69 (m, 8H), 2.52 (t, J=7.6 Hz, 2H), 2.38 (s, 3H), 2.18-1.85 (m, 4H), 1.68– 1.15 (m, 15H). LC/MS ESI 541 (M+H)
+. [1063] Compound 110B/110C, arbitrarily assigned as the diastereomeric mixture of (R)- and (S)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl((R)-6-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)hexan-2-yl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.28 (s, 1H), 7.18-7.00 (m, 2H), 6.39-6.36 (m, 1H), 4.70-4.65 (m, 1H), 3.97-3.95 (m, 3H), 3.62- 3.38 (m, 3H), 3.25-2.69 (m, 8H), 2.58-2.32 (m, 5H), 2.18-1.85 (m, 4H), 1.68–1.15 (m, 15H). LC/MS ESI 541 (M+H)
+. [1064] Compound 110D, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl((S)-6-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)hexan-2- yl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.25-7.20 (m, 2H), 7.06 (dd, J=12.8, 2.0 Hz, 1H), 6.40 (d, J=7.2 Hz, 1H), 4.68 (s, 1H), 3.95 (s, 3H), 3.62-3.38 (m, 3H), 3.25- 2.69 (m, 8H), 2.52 (t, J=7.6 Hz, 2H), 2.38 (s, 3H), 2.18-1.85 (m, 4H), 1.68–1.15 (m, 15H). LC/MS ESI 541 (M+H)
+.
Preparation of 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((S)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 111A and 111B)

[1065] 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((S)-3-(methyl(5-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% acetonitrile) to give diastereomers products Compound 111A (69 mg) and Compound 111B (68 mg) as white solids. [1066] Compound 111A, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((S)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.23 (s, 1H), 7.15 (d, J=7.2 Hz, 1H), 7.00 (dd, J=12.8, 2.0 Hz, 1H), 6.36 (d, J=7.6 Hz, 1H), 4.60 (s, 1H), 3.95 (s, 3H), 3.42-3.38 (m, 3H), 3.22-3.18 (m, 1H), 3.02-2.51 (m, 10H), 2.47 (s, 3H), 2.20-1.85 (m, 4H), 1.70– 1.58 (m, 4H), 1.42–1.18 (m, 8H). LC/MS ESI 527 (M+H)
+. [1067] Compound 111B, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((S)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.28 (s, 1H), 7.16 (d, J=7.2 Hz, 1H), 7.02 (dd, J=12.8, 2.0 Hz, 1H), 6.37 (d, J=7.6 Hz, 1H), 4.62 (s, 1H), 3.95 (s, 3H), 3.42-3.38 (m, 3H), 3.22-3.18 (m, 1H), 3.02-2.51 (m, 10H), 2.48 (s, 3H), 2.20-1.85 (m, 4H), 1.70– 1.58 (m, 4H), 1.42–1.18 (m, 8H). LC/MS ESI 527 (M+H)
+.
Preparation of 2-((R)-3-(ethyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid (Compounds 112A and 112B)

[1068] 2-((R)-3-(ethyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin- 1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% acetonitrile) to give diastereomers products Compound 112A (60 mg) and Compound 112B (43 mg) as white solids. [1069] Compound 112A, arbitrarily assigned as (S)-2-((R)-3-(ethyl(5-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.22-7.19 (m, 2H), 7.00 (dd, J=12.8, 2.0 Hz, 1H), 6.39 (d, J=7.6 Hz, 1H), 4.61 (s, 1H), 3.95 (s, 3H), 3.75-3.73 (m, 1H), 3.42-3.28 (m, 3H), 3.18-2.51 (m, 12H), 2.20-1.55 (m, 8H), 1.48–1.18 (m, 11H). LC/MS ESI 541 (M+H)
+. [1070] Compound 112B, arbitrarily assigned as (R)-2-((R)-3-(ethyl(5-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)acetic acid1H NMR (400 MHz, MeOD) δ 7.26-7.19 (m, 2H), 7.04 (dd, J=12.8, 2.0 Hz, 1H), 6.39 (d, J=7.6 Hz, 1H), 4.59 (s, 1H), 3.95 (s, 3H), 3.75-3.73 (m, 1H), 3.42-3.28 (m, 3H), 3.18-2.51 (m, 12H), 2.20-1.55 (m, 8H), 1.48–1.18 (m, 11H).. LC/MS ESI 541 (M+H)
+.
Preparation of 2-(3-fluoro-2-methoxy-5-(2-methoxypropan-2-yl)phenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 113A and 113B)

[1071] 2-(3-fluoro-2-methoxy-5-(2-methoxypropan-2-yl)phenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% acetonitrile) to give diastereomers products Compound 113A (36 mg) and Compound 113B (41 mg) as white solids. [1072] Compound 113A, arbitrarily assigned as (S)-2-(3-fluoro-2-methoxy-5-(2- methoxypropan-2-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.48 (d, J=0.8 Hz, 1H), 7.15-7.10 (m, 2H), 6.36 (d, J=7.6 Hz, 1H), 4.47 (s, 1H), 3.98 (s, 3H), 3.40-3.35 (m, 3H), 3.06 (s, 3H), 2.98-2.95 (m, 2H), 2.72-2.48 (m, 8H), 2.38 (s, 3H), 2.05-1.80 (m, 4H), 1.70-1.35 (m, 12H). LC/MS ESI 557 (M+H)
+. [1073] Compound 113B, arbitrarily assigned as (R)-2-(3-fluoro-2-methoxy-5-(2- methoxypropan-2-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.47 (d, J=0.8 Hz, 1H), 7.16-7.13 (m, 2H), 6.36 (d, J=7.6 Hz, 1H), 4.53 (s, 1H), 3.98 (s, 3H), 3.40-3.35 (m, 3H), 3.06 (s, 3H), 2.98-2.48 (m, 10H), 2.34 (s, 3H), 2.05-1.80 (m, 4H), 1.70-1.35 (m, 12H). LC/MS ESI 557 (M+H)
+.
Preparation of 2-(5-(tert-butyl)-3-fluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 114A and 114B)

[1074] 2-(5-(tert-butyl)-3-fluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% acetonitrile) to give diastereomers products Compound 114A (41 mg) and Compound 114B (47 mg) as white solids. [1075] Compound 114A, arbitrarily assigned as (S)-2-(5-(tert-butyl)-3-fluoro-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.30-7.26 (m, 2H), 7.18 (dd, J=12.8, 2.4 Hz, 1H), 6.42 (d, J=7.6 Hz, 1H), 4.76 (s, 1H), 3.95 (s, 3H), 3.42-3.28 (m, 3H), 3.18-2.51 (m, 13H), 2.20-1.42 (m, 10H), 1.31 (s, 9H). LC/MS ESI 541 (M+H)
+. [1076] Compound 114B, arbitrarily assigned as (R)-2-(5-(tert-butyl)-3-fluoro-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.41-7.30 (m, 2H), 7.19 (dd, J=12.8, 2.4 Hz, 1H), 6.45 (d, J=7.6 Hz, 1H), 4.60 (s, 1H), 3.98 (s, 3H), 3.42-3.28 (m, 3H), 3.18-2.51 (m, 13H), 2.20-1.42 (m, 10H), 1.31 (s, 9H). LC/MS ESI 541 (M+H)
+. Preparation of 2-(5-cyclopropyl-3,4-difluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 115A and 115B)
[1077] 2-(5-cyclopropyl-3,4-difluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A to give the diastereomers products Compound 115A (6 mg) and Compound 115B (3 mg) as white solids. [1078] Compound 115A, arbitrarily assigned as (S)-2-(5-cyclopropyl-3,4-difluoro-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, MeOD) δ 7.17 (d, J = 7.3 Hz, 1H), 6.96 (dd, J = 8.0, 1.9 Hz, 1H), 6.37 (d, J = 7.3 Hz, 1H), 4.47 (s, 1H), 3.99 (d, J = 1.6 Hz, 3H), 3.48 – 3.34 (m, 3H), 3.15-3.10 (m, 1H), 2.85 – 2.50 (m, 12H), 2.18 – 1.85 (m, 5H), 1.75 – 1.58 (m, 4H), 1.41 – 1.32 (m, 2H), 1.01 – 0.92 (m, 2H), 0.78 – 0.70 (m, 2H). LC/MS ESI 543.3 (M+H)
+. [1079] Compound 115B, arbitrarily assigned as (R)-2-(5-cyclopropyl-3,4-difluoro-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, MeOD) δ 7.22 (d, J = 7.3 Hz, 1H), 6.98 (dd, J = 8.0, 1.9 Hz, 1H), 6.40 (d, J = 7.3 Hz, 1H), 4.46 (s, 1H), 3.99 (d, J = 1.6 Hz, 3H), 3.48 – 3.34 (m, 3H), 2.95 – 2.50 (m, 12H), 2.18 – 1.85 (m, 5H), 1.75 – 1.58 (m, 4H), 1.41 – 1.32 (m, 2H), 1.01 – 0.92 (m, 2H), 0.78 – 0.70 (m, 2H). LC/MS ESI 543.3 (M+H)
+. Preparation of 2-(3,4-difluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4-yl)phenyl)-2- ((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1- yl)acetic acid (Compounds 116A and 116B)
[1080] 2-(3,4-difluoro-2-methoxy-5-(4-methyltetrahydro-2H-pyran-4-yl)phenyl)-2-((R)-3- (methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A to give the diastereomers products Compound 116A (87 mg) and Compound 116B (111 mg) as white solids.
[1081] Compound 116A, arbitrarily assigned as (S)-2-(3,4-difluoro-2-methoxy-5-(4- methyltetrahydro-2H-pyran-4-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, MeOD) δ 7.31 (dd, J = 8.5, 1.9 Hz, 1H), 7.15 (d, J = 7.3 Hz, 1H), 6.36 (d, J = 7.3 Hz, 1H), 4.48 (s, 1H), 3.99 (d, J = 1.6 Hz, 3H), 3.88 – 3.74 (m, 8H), 3.10-3.08 (m, 1H), 3.19 – 2.55 (m, 9H), 2.49 (s, 3H), 2.32 – 2.00 (m, 3H), 1.99 – 1.49 (m, 10H), 1.36 (s, 3H). LC/MS ESI 601.3 (M+H)
+. [1082] Compound 116B, arbitrarily assigned as (R)-2-(3,4-difluoro-2-methoxy-5-(4- methyltetrahydro-2H-pyran-4-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, MeOD) δ 7.35 (d, J = 8.3 Hz, 1H), 7.20 (d, J = 6.9 Hz, 1H), 6.38 (d, J = 7.2 Hz, 1H), 4.43 (s, 1H), 4.00 (s, 3H), 3.84 – 3.13 (m, 8H), 2.97-2.95 (m, 1H), 2.84 – 2.55 (m, 9H), 2.49 (s, 3H), 2.29 – 2.01 (m, 3H), 1.99 – 1.39 (m, 10H), 1.36 (s, 3H). LC/MS ESI 601.3 (M+H)
+. Preparation of 2-(5-(2,2-dimethyltetrahydrofuran-3-yl)-3-fluoro-2-methoxyphenyl)-2-((R)- 3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 117A/117B and 117C/117D)
[1083] 2-(5-(2,2-dimethyltetrahydrofuran-3-yl)-3-fluoro-2-methoxyphenyl)-2-((R)-3- (methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A to give diastereomeric products Compound 117A/117B (136 mg) and Compound 117C/117D (113 mg) as white solids. [1084] Compound 117A/117B, arbitrarily assigned as the diastereomeric mixture (2S)-2-(5- (2,2-dimethyltetrahydrofuran-3-yl)-3-fluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d4) δ 7.28-7.03 (m, 2H), 7.07 (d, J = 13.2 Hz, 1H), 6.39 (d, J = 7.2 Hz, 1H), 4.65 (s, 1H), 4.10 – 3.86 (m, 5H), 3.51 – 3.29 (m, 3H), 3.25 – 2.79 (m, 5H), 2.77 – 2.22 (m, 11H), 2.21 – 1.82 (m, 4H), 1.73 – 1.57 (m, 4H), 1.46 – 1.31 (m, 5H), 0.85 (d, J = 7.6 Hz, 3H). LC/MS ESI 583.3 (M+H)
+.
[1085] Compound 117C/117D, arbitrarily assigned as the diastereomeric mixture (2R)-2-(5- (2,2-dimethyltetrahydrofuran-3-yl)-3-fluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d4) δ 7.31 (d, J = 6.4 Hz, 1H), 7.21 (d, J = 7.6 Hz, 1H), 7.08 (d, J = 12.8 Hz, 1H), 6.40 (d, J = 7.2 Hz, 1H), 4.57 (s, 1H), 4.08 – 3.88 (m, 5H), 3.52 – 3.33 (m, 4H), 3.25 – 2.76 (m, 4H), 2.75 – 2.50 (m, 11H), 2.47 – 1.79 (m, 4H), 1.78 – 1.52 (m, 4H), 1.50 – 1.28 (m, 5H), 0.83 (s, 3H). LC/MS ESI 583.3 (M+H)
+. Preparation of 2-((R)-3-(tert-butyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid (Compounds 118A and 118B)
[1086] 2-((R)-3-(tert-butyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (33-65% MeCN) to give diastereomeric products Compound 118A (52 mg) and Compound 118B (61 mg) as white solids. [1087] Compound 118A, arbitrarily assigned as (S)-2-((R)-3-(tert-butyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)acetic acid.
1H NMR (400 MHz, Methanol-d
4) δ 7.25 (t, J = 1.5 Hz, 1H), 7.15 (d, J = 7.3 Hz, 1H), 7.02 (dd, J = 12.8, 2.3 Hz, 1H), 6.37 (d, J = 7.3 Hz, 1H), 4.65 (s, 1H), 4.17 – 4.05 (m, 1H), 3.94 (d, J = 1.6 Hz, 3H), 3.41 – 3.34 (m, 2H), 3.28 – 3.18 (m, 1H), 3.14 – 2.96 (m, 1H), 2.90 – 2.63 (m, 7H), 2.59 – 2.47 (m, 2H), 2.12 – 1.81 (m, 4H), 1.72 – 1.60 (m, 2H), 1.52 (s, 2H), 1.41 – 1.28 (m, 2H), 1.21 (dd, J = 6.9, 1.2 Hz, 6H), 1.16 (s, 9H). LC/MS ESI 569.4 (M+H)
+. [1088] Compound 118B, arbitrarily assigned as (R)-2-((R)-3-(tert-butyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)acetic acid.
1H NMR (400 MHz, Methanol-d
4) δ 7.23 (d, J = 1.9 Hz, 1H), 7.15 (d,
J = 7.4 Hz, 1H), 7.10 – 7.00 (m, 1H), 6.37 (d, J = 7.4 Hz, 1H), 4.74 (s, 1H), 4.18 – 4.04 (m, 1H), 3.95 (d, J = 1.6 Hz, 3H), 3.40 – 3.33 (m, 2H), 3.27 – 3.15 (m, 1H), 3.04 – 2.61 (m, 7H), 2.58 – 2.48 (m, 2H), 2.09 – 1.79 (m, 4H), 1.74 – 1.62 (m, 2H), 1.58 – 1.41 (m, 2H), 1.41 – 1.28 (m, 3H), 1.21 (dd, J = 7.0, 1.2 Hz, 6H), 1.15 (s, 9H). LC/MS ESI 569.3 (M+H)
+. Preparation of 2-((R)-3-((2,2-difluoro-5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)(methyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid (Compounds 119A and 119B)
[1089] 2-((R)-3-((2,2-difluoro-5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)(methyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (33-65% MeCN) to give diastereomeric products Compound 119A (68 mg) and Compound 118B (68 mg) as white solids. [1090] Compound 119A,arbitrarily assigned as (S)-2-((R)-3-((2,2-difluoro-5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)(methyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl- 2-methoxyphenyl)acetic acid.
1H NMR (400 MHz, Methanol-d4) δ 7.25 – 7.16 (m, 2H), 7.14 – 7.07 (m, 1H), 6.40 (d, J = 7.6 Hz, 1H), 4.94 (d, J = 3.7 Hz, 1H), 3.97 (d, J = 1.9 Hz, 3H), 3.49 – 3.34 (m, 5H), 3.22 – 3.12 (m, 1H), 3.03 – 2.68 (m, 6H), 2.59 (t, J = 7.5 Hz, 2H), 2.35 (s, 3H), 2.11 – 1.79 (m, 8H), 1.22 (dd, J = 7.0, 1.4 Hz, 6H). LC/MS ESI 563.4 (M+H)
+. [1091] Compound 119B,arbitrarily assigned as (R)-2-((R)-3-((2,2-difluoro-5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)(methyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-5-isopropyl- 2-methoxyphenyl)acetic acid.
1H NMR (400 MHz, Methanol-d4) δ 7.32 – 7.22 (m, 2H), 7.10 (dd, J = 13.0, 2.2 Hz, 1H), 6.44 (d, J = 7.3 Hz, 1H), 4.78 (s, 1H), 3.98 (d, J = 1.7 Hz, 3H), 3.59 – 3.46 (m, 1H), 3.42 – 3.33 (m, 3H), 3.19 – 2.94 (m, 3H), 2.91 – 2.58 (m, 7H), 2.34 (s, 3H), 2.18 – 2.03 (m, 2H), 1.99 – 1.72 (m, 6H), 1.23 (dd, J = 6.9, 1.2 Hz, 6H). LC/MS ESI 563.4 (M+H)
+.
Preparation of 2-(2-fluoro-3-isopropyl-6-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 120A and 120B)

[1092] 2-(2-fluoro-3-isopropyl-6-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (33-65% MeCN) to give diastereomeric products Compound 120A (93 mg) and Compound 118B (99 mg) as white solids. [1093] Compound 120A, arbitrarily assigned as (R)-2-(2-fluoro-3-isopropyl-6- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.47 (d, J=7.2 Hz, 1H), 7.29 (t, J=8.8 Hz, 1H), 6.87 (d, J=8.4 Hz, 1H), 6.54 (d, J=7.2 Hz, 1H), 4.88 (s, 1H), 4.05 (d, J=10.0 Hz, 1H), 3.87 (s, 3H), 3.55-3.38 (m, 3H), 3.25-2.70 (m, 10H), 2.54 (s, 3H), 2.20-1.58 (m, 10H), 1.26–1.20 (m, 6H). LC/MS ESI 527 (M+H)
+. [1094] Compound 120B, arbitrarily assigned as (S)-2-(2-fluoro-3-isopropyl-6- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.47 (d, J=7.2 Hz, 1H), 7.29 (t, J=8.8 Hz, 1H), 6.87 (d, J=8.4 Hz, 1H), 6.51 (d, J=7.2 Hz, 1H), 5.18 (s, 1H), 3.87- 3.55 (m, 6H), 3.38-2.70 (m, 9H), 2.64 (s, 3H), 2.30-1.58 (m, 12H), 1.26–1.20 (m, 6H). LC/MS ESI 527 (M+H)
+.
Preparation of 2-(3-fluoro-2-methoxy-5-((R)-tetrahydrofuran-2-yl)phenyl)-2-((R)-3- (methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 121A and 121B)
[1095] 2-(3-fluoro-2-methoxy-5-((R)-tetrahydrofuran-2-yl)phenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% MeCN) to give diastereomeric products Compound 121A (33 mg) and Compound 121B (43 mg) as white solids. [1096] Compound 121A, arbitrarily assigned as (R)-2-(3-fluoro-2-methoxy-5-((R)- tetrahydrofuran-2-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d4) δ 7.24 (d, J = 7.5 Hz, 2H), 7.12 (d, J = 12.5 Hz, 1H), 6.40 (d, J = 7.1 Hz, 1H), 4.80 (t, J = 7.2 Hz, 1H), 4.75 – 4.65 (m, 1H), 4.11 – 4.00 (m, 1H), 3.95 (d, J = 1.8 Hz, 3H), 3.92 – 3.79 (m, 1H), 3.53 – 3.43 (m, 1H), 3.37 (t, J = 5.6 Hz, 2H), 3.26 – 2.94 (m, 3H), 2.85 – 2.66 (m, 4H), 2.67 – 2.45 (m, 6H), 2.37 – 2.24 (m, 1H), 2.01 (q, J = 7.1 Hz, 4H), 1.92 – 1.83 (m, 2H), 1.81 – 1.58 (m, 5H), 1.53 – 1.37 (m, 2H). LC/MS ESI 555.3 (M+H)
+. [1097] Compound 121B, arbitrarily assigned as (S)-2-(3-fluoro-2-methoxy-5-((R)- tetrahydrofuran-2-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d
4) δ 7.35 (s, 1H), 7.28 (s, 1H), 7.11 (dd, J = 12.6, 2.1 Hz, 1H), 6.53 – 6.33 (m, 1H), 4.80 (t, J = 7.1 Hz, 1H), 4.59 – 4.49 (m, 1H), 4.06 – 3.93 (m, 4H), 3.91 – 3.82 (m, 1H), 3.66 – 3.45 (m, 2H), 3.40 – 3.33 (m, 2H), 3.22 – 2.98 (m, 1H), 2.91 – 2.45 (m, 11H), 2.36 – 2.24 (m, 1H), 2.19 – 1.41 (m, 14H). LC/MS ESI 555.3 (M+H)
+.
Preparation of 2-(5-cyclopropyl-3-fluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 122A and 122B)
[1098] 2-(5-cyclopropyl-3-fluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% MeCN) to give diastereomeric products Compound 122A (36 mg) and Compound 122B (24 mg) as white solids. [1099] Compound 122A, arbitrarily assigned as (S)-2-(5-cyclopropyl-3-fluoro-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d4) δ 7.15 (d, J = 7.3 Hz, 1H), 7.12 – 7.05 (m, 1H), 6.82 (dd, J = 12.8, 2.2 Hz, 1H), 6.36 (d, J = 7.3 Hz, 1H), 4.57 (s, 1H), 3.92 (d, J = 1.5 Hz, 3H), 3.43 – 3.34 (m, 3H), 3.23 – 3.11 (m, 1H), 2.95 (s, 1H), 2.90 – 2.82 (m, 1H), 2.79 – 2.57 (m, 5H), 2.56 – 2.50 (m, 2H), 2.45 (s, 3H), 2.16 – 1.78 (m, 5H), 1.71 – 1.52 (m, 4H), 1.43 – 1.27 (m, 2H), 0.98 – 0.89 (m, 2H), 0.69 – 0.59 (m, 2H). LC/MS ESI 525.3 (M+H)
+. [1100] Compound 122B, arbitrarily assigned as (R)-2-(5-cyclopropyl-3-fluoro-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d
4) δ 7.16 (d, J = 7.2 Hz, 1H), 7.13 – 7.08 (m, 1H), 6.84 (dd, J = 12.8, 2.2 Hz, 1H), 6.36 (d, J = 7.4 Hz, 1H), 4.57 (s, 1H), 3.93 (d, J = 1.5 Hz, 3H), 3.36 (t, J = 5.6 Hz, 3H), 3.27 – 3.17 (m, 1H), 2.97 – 2.82 (m, 3H), 2.76 – 2.63 (m, 3H), 2.57 – 2.48 (m, 3H), 2.39 (s, 3H), 2.16 – 2.04 (m, 1H), 1.95 – 1.79 (m, 4H), 1.73 – 1.48 (m, 4H), 1.43 – 1.26 (m, 3H), 0.99 – 0.89 (m, 2H), 0.70 – 0.61 (m, 2H). LC/MS ESI 525.3 (M+H)
+.
Preparation of 2-(5-isopropyl-2-methoxy-3-(trifluoromethyl)phenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 123A and 123B)
[1101] 2-(5-isopropyl-2-methoxy-3-(trifluoromethyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% MeCN) to give diastereomeric products Compound 123A (173 mg) and Compound 123B (151 mg) as white solids. [1102] Compound 123A, arbitrarily assigned as (S)-2-(5-isopropyl-2-methoxy-3- (trifluoromethyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d
4) δ 7.79 (d, J = 2.3 Hz, 1H), 7.42 (d, J = 2.3 Hz, 1H), 7.17 (dd, J = 7.3, 1.1 Hz, 1H), 6.37 (d, J = 7.3 Hz, 1H), 4.52 (s, 1H), 3.50 (s, 1H), 3.41 – 3.33 (m, 2H), 3.18 – 3.08 (m, 1H), 2.98 – 2.86 (m, 2H), 2.84 – 2.65 (m, 5H), 2.61 – 2.42 (m, 6H), 2.18 – 2.05 (m, 1H), 2.04 – 1.93 (m, 1H), 1.91 – 1.81 (m, 2H), 1.75 – 1.55 (m, 4H), 1.44 – 1.32 (m, 2H), 1.24 (dd, J = 6.9, 3.4 Hz, 6H). LC/MS ESI 577.3 (M+H)
+. [1103] Compound 123B, arbitrarily assigned as (R)-2-(5-isopropyl-2-methoxy-3- (trifluoromethyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d
4) δ 7.84 (d, J = 2.3 Hz, 1H), 7.42 (d, J = 2.3 Hz, 1H), 7.20 (d, J = 7.3 Hz, 1H), 6.38 (d, J = 7.3 Hz, 1H), 4.43 (s, 1H), 3.60 – 3.46 (m, 1H), 3.41 – 3.33 (m, 2H), 3.07 – 2.84 (m, 2H), 2.82 – 2.63 (m, 5H), 2.63 – 2.46 (m, 6H), 2.13 – 1.98 (m, 1H), 1.96 – 1.79 (m, 3H), 1.77 – 1.53 (m, 4H), 1.48 – 1.36 (m, 2H), 1.24 (dd, J = 6.8, 2.3 Hz, 6H). LC/MS ESI 577.3 (M+H)
+.
Preparation of 2-(3-chloro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 124A and 124B)
[1104] 2-(3-chloro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% MeCN) to give diastereomeric products Compound 124A (82 mg) and Compound 124B (85 mg) as white solids. [1105] Compound 124A, arbitrarily assigned as (S)-2-(3-chloro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid
1H NMR (400 MHz, Methanol-d4) δ 7.44 – 7.29 (m, 1H), 7.24 – 7.11 (m, 2H), 6.44 – 6.31 (m, 1H), 4.67 – 4.47 (m, 1H), 3.92 (s, 3H), 3.47 – 3.33 (m, 3H), 3.24 – 3.10 (m, 1H), 2.96 – 2.47 (m, 13H), 2.16 – 1.80 (m, 4H), 1.74 – 1.50 (m, 6H), 1.44 – 1.31 (m, 2H), 0.92 (t, J = 7.3 Hz, 3H). LC/MS ESI 543.3 (M+H)
+. [1106] Compound 124B, arbitrarily assigned as (R)-2-(3-chloro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid
1H NMR (400 MHz, Methanol-d4) δ 7.37 (d, J = 2.3 Hz, 1H), 7.22 (d, J = 2.5 Hz, 2H), 6.39 (dd, J = 7.3, 3.0 Hz, 1H), 4.52 (s, 1H), 3.53 – 3.42 (m, 1H), 3.41 – 3.33 (m, 3H), 2.97 – 2.41 (m, 13H), 2.13 – 1.99 (m, 1H), 1.96 – 1.77 (m, 3H), 1.76 – 1.50 (m, 6H), 1.52 – 1.37 (m, 2H), 1.24 – 1.17 (m, 1H), 0.92 (t, J = 7.4 Hz, 3H). LC/MS ESI 577.3 (M+H)
+.
Preparation of 2-(3-fluoro-5-isobutyl-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 125A and 125B)
[1107] 2-(3-fluoro-5-isobutyl-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% MeCN) to give diastereomeric products Compound 125A (76 mg) and Compound 125B (74 mg) as white solids. [1108] Compound 125A, arbitrarily assigned as (S)-2-(3-fluoro-5-isobutyl-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d
4) δ 7.18 (d, J = 7.3 Hz, 1H), 7.10 (s, 1H), 6.93 (dd, J = 12.5, 2.2 Hz, 1H), 6.37 (d, J = 7.3 Hz, 1H), 4.67 (s, 1H), 3.94 (d, J = 1.6 Hz, 3H), 3.45 – 3.34 (m, 3H), 3.27 – 3.18 (m, 1H), 2.91 (d, J = 67.0 Hz, 3H), 2.74 – 2.39 (m, 11H), 2.05 – 1.93 (m, 1H), 1.92 – 1.76 (m, 3H), 1.68 (q, J = 7.3 Hz, 4H), 1.46 – 1.30 (m, 2H), 0.89 (dd, J = 6.6, 1.3 Hz, 6H). LC/MS ESI 541.3 (M+H)
+. [1109] Compound 125B, arbitrarily assigned as (R)-2-(3-fluoro-5-isobutyl-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d
4) δ 7.21 (d, J = 7.3 Hz, 1H), 7.16 (d, J = 1.7 Hz, 1H), 6.95 (dd, J = 12.6, 2.0 Hz, 1H), 6.39 (d, J = 7.3 Hz, 1H), 4.60 (s, 1H), 3.96 (d, J = 1.6 Hz, 3H), 3.49 – 3.33 (m, 4H), 2.96 – 2.76 (m, 4H), 2.74 – 2.51 (m, 5H), 2.50 – 2.38 (m, 5H), 2.17 – 2.05 (m, 1H), 1.97 – 1.37 (m, 11H), 0.88 (dd, J = 6.6, 2.4 Hz, 6H). LC/MS ESI 541.3 (M+H)
+.
Preparation of 2-(3-fluoro-5-isopentyl-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 126A and 126B)
[1110] 2-(3-fluoro-5-isopentyl-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% MeCN) to give diastereomeric products Compound 126A (76 mg) and Compound 126B (83 mg) as white solids. [1111] Compound 126A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopentyl-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d
4) δ 7.20 – 7.08 (m, 2H), 7.00 – 6.88 (m, 1H), 6.37 (d, J = 7.4 Hz, 1H), 4.66 (d, J = 2.5 Hz, 1H), 3.94 (dd, J = 3.2, 1.5 Hz, 3H), 3.46 – 3.34 (m, 3H), 3.25 – 3.17 (m, 1H), 3.03 – 2.89 (m, 2H), 2.86 – 2.74 (m, 1H), 2.75 – 2.49 (m, 8H), 2.47 (s, 3H), 2.16 – 2.05 (m, 1H), 2.04 – 1.92 (m, 1H), 1.92 – 1.82 (m, 2H), 1.74 – 1.30 (m, 9H), 0.97 – 0.81 (m, 6H). LC/MS ESI 555.4 (M+H)
+. [1112] Compound 126B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopentyl-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, Methanol-d
4) δ 7.23 – 7.13 (m, 2H), 7.02 – 6.91 (m, 1H), 6.38 (d, J = 7.3 Hz, 1H), 4.60 (s, 1H), 3.95 (d, J = 1.6 Hz, 3H), 3.47 – 3.32 (m, 4H), 2.99 – 2.49 (m, 11H), 2.42 (s, 3H), 2.17 – 2.02 (m, 1H), 2.00 – 1.80 (m, 3H), 1.76 – 1.34 (m, 9H), 0.95 – 0.79 (m, 6H). LC/MS ESI 555.4 (M+H)
+.
Preparation of 2-(3-fluoro-5-(2-hydroxypropan-2-yl)-2-methoxyphenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 127A and 127B)
[1113] 2-(3-fluoro-5-(2-hydroxypropan-2-yl)-2-methoxyphenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% MeCN) to give diastereomeric products Compound 127A (44 mg) and Compound 127B (44 mg) as white solids. [1114] Compound 127A, arbitrarily assigned as (S)-2-(3-fluoro-5-(2-hydroxypropan-2-yl)- 2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, MeOD) δ 7.41-7.20 (m, 3H), 6.39 (d, J=7.6 Hz, 1H), 4.66 (s, 1H), 3.97 (s, 3H), 3.48-3.35 (m, 4H), 3.18-2.48 (m, 11H), 2.18- 1.35 (m, 16H). LC/MS ESI 543 (M+H)
+. [1115] Compound 127B, arbitrarily assigned as (R)-2-(3-fluoro-5-(2-hydroxypropan-2-yl)- 2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, MeOD) δ 7.47-7.27 (m, 3H), 6.41 (d, J=7.6 Hz, 1H), 4.62 (s, 1H), 3.97 (s, 3H), 3.48-3.35 (m, 4H), 3.05-2.42 (m, 11H), 2.18- 1.35 (m, 16H). LC/MS ESI 543 (M+H)
+. Preparation of 2-(3-fluoro-2-methoxy-5-(1-methoxycyclohexyl)phenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 128A and 128B)
[1116] 2-(3-fluoro-2-methoxy-5-(1-methoxycyclohexyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% MeCN) to give diastereomeric products Compound 128A (52 mg) and Compound 128B (59 mg) as white solids. [1117] Compound 128A, arbitrarily assigned as (S)-2-(3-fluoro-2-methoxy-5-(1- methoxycyclohexyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, MeOD) δ 7.42 (s, 1H), 7.25- 7.10 (m, 2H), 6.38 (d, J=7.6 Hz, 1H), 4.67 (s, 1H), 3.98 (s, 3H), 3.45-3.20 (m, 4H), 2.98-2.48 (m, 15H), 2.15-1.32 (m, 20H). LC/MS ESI 597 (M+H)
+. [1118] Compound 128B, arbitrarily assigned as (R)-2-(3-fluoro-2-methoxy-5-(1- methoxycyclohexyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, MeOD) δ 7.45 (s, 1H), 7.22- 7.10 (m, 2H), 6.40 (d, J=7.6 Hz, 1H), 4.62 (s, 1H), 3.99 (s, 3H), 3.45-3.20 (m, 4H), 2.98-2.48 (m, 15H), 2.15-1.32 (m, 20H). LC/MS LC/MS ESI 597 (M+H)
+. Preparation of 2-(3-fluoro-2-methoxy-5-(2-methyltetrahydro-2H-pyran-2-yl)phenyl)-2-((R)- 3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 129A and 129B)
[1119] 2-(3-fluoro-2-methoxy-5-(2-methyltetrahydro-2H-pyran-2-yl)phenyl)-2-((R)-3- (methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% MeCN) to give diastereomeric products Compound 129A (40 mg) and Compound 129B (50 mg) as white solids. [1120] Compound 129A, arbitrarily assigned as (2S)-2-(3-fluoro-2-methoxy-5-(2- methyltetrahydro-2H-pyran-2-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, MeOD) δ 7.40
(s, 1H), 7.19-7.14 (m, 2H), 6.38 (d, J=7.6 Hz, 1H), 4.62 (s, 1H), 3.98 (s, 3H), 3.75-3.73 (m, 1H), 3.45-3.20 (m, 5H), 2.98-2.48 (m, 12H), 2.25-1.32 (m, 19H). LC/MS ESI 583 (M+H) +. [1121] Compound 129B, arbitrarily assigned as (2R)-2-(3-fluoro-2-methoxy-5-(2- methyltetrahydro-2H-pyran-2-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, MeOD) δ 7.42 (s, 1H), 7.21-7.16 (m, 2H), 6.39 (d, J=7.6 Hz, 1H), 4.63 (s, 1H), 3.99 (s, 3H), 3.75-3.73 (m, 1H), 3.45-3.20 (m, 5H), 2.98-2.48 (m, 12H), 2.25-1.32 (m, 19H). LC/MS ESI 583 (M+H) +. Preparation of 2-(5-ethyl-3-fluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 130A and 130B)

[1122] 2-(3-fluoro-2-methoxy-5-(2-methyltetrahydro-2H-pyran-2-yl)phenyl)-2-((R)-3- (methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% MeCN) to give diastereomeric products Compound 130A (62 mg) and Compound 130B (52 mg) as white solids. [1123] Compound 130A, arbitrarily assigned as (S)-2-(5-ethyl-3-fluoro-2-methoxyphenyl)- 2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, MeOD) δ 7.19-7.17 (m, 2H), 6.98 (dd, J=12.8, 1.6 Hz, 1H), 6.38 (d, J=7.6 Hz, 1H), 4.63 (s, 1H), 3.95 (s, 3H), 3.45-3.32 (m, 3H), 3.22-3.18 (m, 1H), 2.98-2.52 (m, 11H), 2.47 (s, 3H), 2.20-1.18 (m, 13H). LC/MS ESI 513 (M+H)
+. [1124] Compound 130B, arbitrarily assigned as (R)-2-(5-ethyl-3-fluoro-2-methoxyphenyl)- 2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, MeOD) δ 7.23-7.19 (m, 2H), 7.00 (dd, J=12.8, 1.6 Hz, 1H), 6.39 (d, J=7.6 Hz, 1H), 4.61 (s, 1H), 3.96 (s, 3H), 3.45-3.32 (m, 4H), 2.98-2.52 (m, 11H), 2.44 (s, 3H), 2.20-1.18 (m, 13H). LC/MS ESI 513 (M+H)
+.
Preparation of 2-((R)-3-((2,2-difluoro-5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)(methyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-2-methoxy-5-(4-methyltetrahydro-2H- pyran-4-yl)phenyl)acetic acid (Compounds 131A and 131B)
[1125] 2-((R)-3-((2,2-difluoro-5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)(methyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-2-methoxy-5-(4-methyltetrahydro-2H- pyran-4-yl)phenyl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by Prep-HPLC A (30-60% MeCN) to give diastereomeric products Compound 131A (71 mg) and Compound 131B (79 mg) as white solids. [1126] Compound 131A, arbitrarily assigned as (S)-2-((R)-3-((2,2-difluoro-5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)(methyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-2-methoxy- 5-(4-methyltetrahydro-2H-pyran-4-yl)phenyl)acetic acid.
1H NMR (400 MHz, Methanol-d4) δ 7.33-7.20 (m, 3H), 6.40 (d, J = 7.6 Hz, 1H), 4.93 (s, 1H), 4.01 (d, J = 1.9 Hz, 3H), 3.78-3,60 (m, 4H), 3.40 – 3.34 (m, 4H), 3.22 –2.58 (m, 8H), 2.37 (s, 3H), 2.11 – 1.77 (m, 13H), 1.28 (s, 3H). LC/MS ESI 619 (M+H)
+. [1127] Compound 131B, arbitrarily assigned as (R)-2-((R)-3-((2,2-difluoro-5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)(methyl)amino)pyrrolidin-1-yl)-2-(3-fluoro-2-methoxy- 5-(4-methyltetrahydro-2H-pyran-4-yl)phenyl)acetic acid.
1H NMR (400 MHz, Methanol-d4) δ 7.41 (s, 1H), 7.28-7.20 (m, 2H), 6.43 (d, J = 7.6 Hz, 1H), 4.72 (s, 1H), 4.01 (d, J = 1.9 Hz, 3H), 3.78-3,60 (m, 4H), 3.40 – 3.34 (m, 4H), 3.22 –2.58 (m, 8H), 2.35 (s, 3H), 2.11 – 1.77 (m, 13H), 1.28 (s, 3H). LC/MS ESI 619 (M+H)
+.
Preparation of 2-(3-fluoro-5-isopropyl-2-(trifluoromethyl)phenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 132A and 132B)
[1128] 2-(3-fluoro-5-isopropyl-2-(trifluoromethyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid was prepared according to the General Example of Alkylation and Hydrolysis, substituting the appropriate reagents and purified by prep-HPLC (Waters Xbridge BEH C 18100 x 30 mm x 10 um; mobile phase: [water (NH
4HCO
310 mmol/L), B acetonitrile 30%-60%, 8 min; Flow Rate (25 mL/min) to give diastereomeric products Compound 132A and 132B. [1129] Compound 132A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- (trifluoromethyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (51.1 mg, 89.32 umol, 35.7% yield, 98.7% purity) was obtained as white solid.
1H NMR (400 MHz, METHANOL-d) δ = 7.64 (s, 1H), 7.18 (d, J = 7.3 Hz, 1H), 7.08 (d, J = 13.1 Hz, 1H), 6.37 (d, J = 7.4 Hz, 1H), 4.47 (s, 1H), 3.62-3.56 (m, 1H), 3.38 (d, J = 5.5 Hz, 2H), 3.08 (dt, J = 4.4, 8.6 Hz, 1H), 3.03-2.92 (m, 2H), 2.91-2.85 (m, 1H), 2.79 (br dd, J = 2.9, 11.0 Hz, 1H), 2.732.69 (m, 2H), 2.64 (s, 4H), 2.58-2.52 (m, 2H), 2.49-2.43 (m, 1H), 2.20-2.11 (m, 1H), 2.06-1.98 (m, 1H), 1.87 (dd, J = 5.3, 6.2 Hz, 2H), 1.71-1.63 (m, 4H), 1.44-1.37 (m, 2H), 1.25 (dd, J = 2.6, 6.9 Hz, 6H). SFC: Rt = 1.147 min, 100.0%. LCMS (ESI) m/z = 565.3 (M+1), Rt = 1.946 min, 98.662%. [1130] Compound 132B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- (trifluoromethyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (62.2 mg, 109.49 umol, 43.7% yield, 99.4% purity) was obtained as white solid.
1H NMR (400 MHz, METHANOL-d) δ = 7.75 (s, 1H), 7.22 (d, J = 7.3 Hz, 1H), 7.06 (d, J = 13.3 Hz, 1H), 6.40 (d, J = 7.4 Hz, 1H), 4.33 (s, 1H), 3.64-3.56 (m, 1H), 3.37 (dd, J = 4.4, 6.5 Hz, 2H), 3.25-3.15 (m, 1H), 2.94 (td, J = 6.9, 13.9 Hz, 1H), 2.80-2.74 (m, 1H), 2.73-2.60 (m, 8H), 2.59-2.51 (m, 2H), 2.41-2.33 (m, 1H), 2.09-2.00 (m, 1H), 1.93-1.83 (m, 3H), 1.79-1.69 (m, 3H), 1.66-1.58 (m, 1H), 1.48 (td, J = 7.4, 14.7 Hz, 2H), 1.24 (dd, J = 1.2, 6.9 Hz, 6H). SFC: R
t = 1.123 min, 100%. LCMS (ESI) m/z = 565.3 (M+1), R
t = 2.012 min, 99.397%.
Preparation of 2-(5-(cyclopropyldifluoromethyl)-3-fluoro-2-methoxyphenyl)-2-((R)-3- (methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 133A and 133B)
Step 1: tert-butyl 2-(5-(cyclopropyldifluoromethyl)-3-fluoro-2-methoxyphenyl)-2-((R)-3- (methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetate

[1131] To a solution of tert-butyl 2-bromo-2-(5-(cyclopropyldifluoromethyl)-3-fluoro-2- methoxyphenyl)acetate (0.4 g, 977.43 umol) and (R)-N-methyl-N-(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)pyrrolidin-3-amine (402.54 mg, 977.43 umol, 3 HCl) in acetonitrile (5 mL) was added DIEA (757.95 mg, 5.86 mmol, 1.02 mL), the reaction was stirred at 25 °C for 16 h. Water (10 mL) was added, the mixture was extracted with ethyl acetate (10 mL x 2). The combined organic layers were washed with water (20 mL x 2) and brine (20 mL x 2), dried over Na2SO4, concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate = 1/1 to DCM/MeOH = 2/1). tert- butyl 2-(5-(cyclopropyldifluoromethyl)-3-fluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetate (340 mg, 55.15% yield) was obtained as a yellow oil. LCMS (ESI) m/z = 631.4 (M+1) Step 2: 2-(5-(cyclopropyldifluoromethyl)-3-fluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid

[1132] To a solution of tert-butyl 2-(5-(cyclopropyldifluoromethyl)-3-fluoro-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-
yl)pentyl)amino)pyrrolidin-1-yl)acetate (0.24 g, 380.48 umol) in acetonitrile (4 mL) was added TMSI (152.26 mg, 760.96 umol, 103.58 uL), the reaction was stirred at 25 °C for 2 h. The pH of the solution was adjusted to about 9 with a lithium hydroxide solution (40 mg lithium hydroxide in 1mL THF and 1 mL water), then the mixture was concentrated, and purified by prep-HPLC (column: Phenomenex C1875 x 30 mm x 3um; mobile phase: [A water (NH4HCO310 mmol/L), B acetonitrile 30%-70%, 8 min; Flow Rate (25 mL/min) to give two peaks: [1133] Compound 133A, arbitrarily assigned as (S)-2-(5-(cyclopropyldifluoromethyl)-3- fluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (25.9 mg, 45.07 umol, 23.69% yield) was obtained as white solid.
1H NMR (400 MHz, METHANOL-d) δ ppm 7.60 (s, 1 H), 7.27 (dd, J = 11.95, 1.64 Hz, 1 H), 7.17 (d, J = 7.23 Hz, 1 H), 6.37 (d, J = 7.23 Hz, 1 H), 4.55 (s, 1 H), 4.00 (d, J = 1.53 Hz, 3 H), 3.44-3.52 (m, 1 H), 3.35-3.41 (m, 2 H), 3.09-3.19 (m, 1 H), 2.82-2.99 (m, 2 H), 2.66- 2.80 (m, 4 H), 2.46-2.61 (m, 6 H), 2.07-2.17 (m, 1 H), 1.94-2.05 (m, 1 H), 1.87 (quin, J = 5.92 Hz, 2 H), 1.52-1.74 (m, 5 H), 1.32-1.45 (m, 2 H), 0.63-0.74 (m, 4 H). SFC: R
t = 1.182 min, 100%. LCMS (ESI) m/z = 575.3 (M+1), Rtp1 = 1.848 min, 99.536%. [1134] Compound 133B, arbitrarily assigned as (R)-2-(5-(cyclopropyldifluoromethyl)-3- fluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (24 mg, 41.76 umol, 21.95% yield) was obtained as a yellow oil.
1H NMR (400 MHz, METHANOL-d) δ = 7.64 (s, 1 H), 7.24-7.32 (m, 1 H), 7.17 (d, J = 7.23 Hz, 1 H), 6.38 (d, J = 7.45 Hz, 1 H), 4.51 (s, 1 H), 4.01 (d, J = 1.53 Hz, 3 H), 3.41-3.52 (m, 1 H), 3.34-3.39 (m, 2 H), 3.12-3.21 (m, 1 H), 2.59-2.91 (m, 7 H), 2.54 (br t, J = 7.78 Hz, 2 H), 2.48 (s, 3 H), 2.03-2.13 (m, 1 H), 1.87 (quin, J = 6.03 Hz, 3 H), 1.50-1.76 (m, 5 H), 1.34-1.47 (m, 2 H), 0.63-0.70 (m, 4 H). SFC: Rt = 1.159 min, 100%. LCMS (ESI) m/z = 575.2 (M+1), Rtp1 = 1.857 min, 98.541%. Preparation of 2-(3-fluoro-2-methoxy-5-(trifluoromethyl)phenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 134A and 134B)
Step 1: tert-butyl 2-(3-fluoro-2-methoxy-5-(trifluoromethyl)phenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetate
[1135] To a solution of tert-butyl 2-bromo-2-(3-fluoro-2-methoxy-5- (trifluoromethyl)phenyl)acetate (0.25 g, 645.72 umol) and (R)-N-methyl-N-(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)pyrrolidin-3-amine (195.30 mg, 645.72 umol) in acetonitrile (4 mL) was added DIEA (333.82 mg, 2.58 mmol, 449.89 uL), the reaction was stirred at 25 °C for 16 h. The mixture was concentrated, the residue was purified by column chromatography (SiO
2, Petroleum ether/Ethyl acetate = 1/1 to 0/1). tert-butyl 2-(3-fluoro-2- methoxy-5-(trifluoromethyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetate (300 mg, crude) was obtained as a yellow oil. LCMS (ESI) m/z = 609 (M+1). Step 2: 2-(3-fluoro-2-methoxy-5-(trifluoromethyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid
[1136] A solution of tert-butyl 2-(3-fluoro-2-methoxy-5-(trifluoromethyl)phenyl)-2-((R)-3- (methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetate (350 mg, 574.99 umol) in formic acid (27.62 mg, 574.99 umol, 5 mL) was stirred at 25 °C for 16 h. The mixture was concentrated to give crude residue which was purified and separated by prep- HPLC (column: Waters Xbridge BEH C18100 x 30 mm x 10 um; mobile phase: A water (NH
4HCO
310 mmol/L), B acetonitrile 15%-65%, 8 min; Flow Rate 25 mL/min. [1137] Compound 134A, arbitrarily assigned as (S)-2-(3-fluoro-2-methoxy-5- (trifluoromethyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (26.7 mg, 47.26 umol, 16.4% yield, 97.8% purity) was obtained as white-solid.
1H NMR (400 MHz, METHANOL-d) δ = 7.72 (s, 1 H), 7.43 (dd, J = 11.68, 1.91 Hz, 1 H), 7.19 (d, J = 7.27 Hz, 1 H), 6.38 (d, J = 7.27 Hz, 1 H), 4.55 (s, 1 H) 3.54 (br
s, 1 H), 3.34-3.41 (m, 2 H), 3.07-3.17 (m, 1 H), 2.95-3.05 (m, 1 H), 2.77-2.92 (m, 2 H), 2.71-2.72 (m, 1 H), 2.71 (t, J = 6.20 Hz, 2 H), 2.51-2.59 (m, 2 H), 2.39-2.50 (m, 1 H), 2.07-2.23 (m, 1 H), 1.94-2.05 (m, 1 H), 1.82-1.93 (m, 2 H), 1.58-1.77 (m, 4 H), 1.34-1.47 (m, 2 H). SFC: Rt = 1.094 min, 95.24%. LCMS (ESI) m/z = 553 (M+1), Rt = 2.616 min, 97.824%. [1138] Compound 134B, arbitrarily assigned as (R)-2-(3-fluoro-2-methoxy-5- (trifluoromethyl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (21.8 mg, 37.25 umol, 13.0% yield, 94.4% purity) was obtained as white-solid.
1H NMR (400 MHz, METHANOL-d) δ = 7.79 (s, 1 H), 7.42 (br d, J = 11.56 Hz, 1 H), 7.20 (d, J = 7.27 Hz, 1 H), 6.39 (d, J = 7.27 Hz, 1 H), 4.44 (br s, 1 H), 4.04 (d, J = 2.03 Hz, 3 H), 3.56 (br s, 1 H), 3.33-3.43 (m, 2 H), 3.24-3.29 (m, 1 H), 3.03 (br s, 1 H), 2.65- 2.84 (m, 5 H), 2.42-2.62 (m, 6 H), 2.07 (td, J = 7.93, 2.98 Hz, 1 H), 1.83-1.96 (m, 3 H), 1.57-1.80 (m, 4 H), 1.39-1.52 (m, 2 H). SFC: R
t = 1.097min, 96.28%. LCMS (ESI) m/z = 553 (M+1), R
t = 2.691 min, 94.420%. Preparation of 2-(5-(1,1-difluoroethyl)-3-fluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compound 135A and 135B)
Step 1: tert-butyl 2-(5-(1,1-difluoroethyl)-3-fluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetate
[1139] To a mixture of tert-butyl 2-bromo-2-(5-(1,1-difluoroethyl)-3-fluoro-2- methoxyphenyl)acetate (43.41 mg, 143.53 umol) in acetonitrile (1 mL) was added DIEA (67.45 mg, 521.92 umol, 90.91 uL). Then the solution of (R)-N-methyl-N-(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)pyrrolidin-3-amine (50 mg, 130.48 umol) in acetonitrile (1 mL) and NaI (1.96 mg, 13.05 umol) was added at 25 °C under nitrogen. The mixture was stirred at 50 °C for 2
h. The mixture was quenched with water (10 mL), extracted with ethyl acetate (10 mL x 3), washed with brine (10 mL), dried with anhydrous Na
2SO
4, filtered and concentrated in vacuum. The residue was used the next step directly without further purification tert-butyl 2-(5-(1,1- difluoroethyl)-3-fluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetate (200 mg, crude) was obtained as a yellow oil. LCMS (ESI) m/z = 605.3 (M+1). Step 2: 2-(5-(1,1-difluoroethyl)-3-fluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid
[1140] A mixture of tert-butyl 2-(5-(1,1-difluoroethyl)-3-fluoro-2-methoxyphenyl)-2-((R)-3- (methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetate (140 mg, 231.50 umol) in formic acid (11.12 mg, 231.50 umol) was stirred at 25 °C for 16 h. The reaction mixture was concentrated in vacuum. The residue was purified by prep-HPLC (column: Waters Xbridge BEH C18100 x 30 mm x 10 um; mobile phase: A water (NH
4HCO
310 mmol/L), B acetonitrile 25%-45%, 8 min; Flow Rate (25 mL/min) to give two peaks: [1141] Compound 135A, arbitrarily assigned as (S)-2-(5-(1,1-difluoroethyl)-3-fluoro-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (30.1 mg, 84.86 umol, 30.1% yield) was obtained as a white solid.
1H NMR (400 MHz, METHANOL-d4) δ = 7.58 (s, 1 H), 7.27 (br d, J = 11.26 Hz, 1 H), 7.17 (d, J = 7.25 Hz, 1 H), 6.37 (d, J = 7.25 Hz, 1 H), 4.55 (s, 1 H), 3.99 (d, J = 1.13 Hz, 3 H), 3.49 (br s, 1 H), 3.36-3.39 (m, 2 H), 3.11-3.17 (m, 1 H), 2.79-3.00 (m, 3 H), 2.68-2.78 (m, 4 H), 2.56 (s, 3 H), 2.47-2.54 (m, 2 H), 2.08-2.15 (m, 1 H), 1.96-2.02 (m, 1 H), 1.84-1.94 (m, 5 H), 1.58-1.72 (m, 4 H), 1.35-1.45 (m, 2 H). SFC: Rt = 2.362 min, 97.38%. LCMS (ESI) m/z = 549.3 (M+1), Rt = 2.442 min, 98.702%. [1142] Compound 135B, arbitrarily assigned as (R)-2-(5-(1,1-difluoroethyl)-3-fluoro-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (31.7 mg, 87.78 umol, 31.7% yield) was obtained as a white solid.
1H NMR (400 MHz, METHANOL-d
4) δ = 7.61 (s, 1 H), 7.28 (dd, J = 12.19, 2.06 Hz, 1 H), 7.19 (d, J = 7.25 Hz, 1 H), 6.39 (d, J = 7.38 Hz, 1 H), 4.52 (s, 1 H), 4.01 (d, J = 2.00
Hz, 3 H), 3.47-3.53 (m, 1 H), 3.35-3.38 (m, 2 H), 3.28 (br s, 1 H), 2.92 (br s, 1 H), 2.78 (br dd, J = 11.01, 7.38 Hz, 2 H), 2.63-2.73 (m, 4 H), 2.54-2.58 (m, 2 H), 2.52 (s, 3 H), 2.06-2.14 (m, 1 H), 1.85-1.94 (m, 6 H), 1.59-1.73 (m, 4 H), 1.39-1.47 (m, 2 H). SFC: Rt = 1.687 min, 97.92%. LCMS (ESI) m/z = 549.3 (M+1), Rt = 2.484 min, 96.097%. Preparation of 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(isobutyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 136A and 136B)
Step 1: tert-butyl 7-(5-((tert-butoxycarbonyl)((3R)-1-(2-ethoxy-1-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-oxoethyl)pyrrolidin-3-yl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine- 1(2H)-carboxylate

[1143] A mixture of tert-butyl (R)-7-(5-((tert-butoxycarbonyl)(pyrrolidin-3- yl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxylate (250 mg, 0.51 mmol), ethyl 2-bromo-2-(3-fluoro-5-isopropyl-2-methoxyphenyl)acetate (170 mg, 0.51 mmol) and DIEA (197 mg, 1.53 mmol) in acetonitrile (5 mL) was stirred at 60 °C for 15 h. Solvent was removed in vacuo, and the residue was purified by silica gel column (DCM: MeOH 50:1) to give the desired product tert-butyl 7-(5-((tert-butoxycarbonyl)((3R)-1-(2-ethoxy-1-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-oxoethyl)pyrrolidin-3-yl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate as a yellow oil (230 mg, 0.31 mmol). Yield 61 % (ESI 741 (M+H) +). Step 2: ethyl 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-((5-(5,6,7,8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetate dihydrochloride

[1144] Tert-butyl 7-(5-((tert-butoxycarbonyl)((3R)-1-(2-ethoxy-1-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-oxoethyl)pyrrolidin-3-yl)amino)pentyl)-3,4-dihydro-1,8-naphthyridine-1(2H)- carboxylate (230 mg, 0.31 mmol) was treated with HCl in 1,4-dioxane (4M, 10 mL) at ambient temperature overnight. Solvent was removed in vacuo to afford the desired product ethyl 2-(3- fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-((5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetate dihydrochloride (168 mg, 0.27 mmol) as a yellow oil. Yield 88% (ESI 541 (M+H) +). Step 3: ethyl 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(isobutyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetate

[1145] A mixture of ethyl 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-((5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetate dihydrochloride (168 mg, 0.27 mmol), 1-bromo-2-methylpropane (37 mg, 0.27 mmol) and DIEA (139 mg, 1.08 mmol) in acetonitrile (5 mL) was stirred at 60 °C for 15 h. Solvent was removed in vacuo, and the residue was purified by silica gel column (DCM: MeOH 50:1) to give the desired product ethyl 2-(3- fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(isobutyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin- 2-yl)pentyl)amino)pyrrolidin-1-yl)acetate as a yellow oil (38 mg, 0.06 mmol). Yield 24 % (ESI 597 (M+H) +). Step 4: 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(isobutyl(5-(5,6,7,8-tetrahydro- 1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid

[1146] Ethyl 2-(3-fluoro-5-isopropyl-2-methoxyphenyl)-2-((R)-3-(isobutyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetate (38 mg, 0.06 mmol) was treated with lithium hydroxide monohydrate (25 mg, 0.6 mmol) in MeOH (2.5 mL) and water (0.5 mL) at room temperature for 2 h. Solvent was removed in vacuo, and the residue was purified by Prep-HPLC A (30-60% acetonitrile) to give diastereomers products Compound 204A (5 mg) and Compound 204B (5 mg) as white solids. [1147] Compound 136A, arbitrarily assigned as (S)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(isobutyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.24 (s, 1H), 7.15 (d, J=7.2 Hz, 1H), 7.07 (dd, J=12.8, 2.0 Hz, 1H), 6.36 (d, J=7.6 Hz, 1H), 4.77 (s, 1H), 3.98 (s, 3H), 3.65-3.18 (m, 5H), 3.02-2.30 (m, 10H), 2.20-1.22 (m, 18H), 0.92–0.88 (m, 6H). LC/MS ESI 569 (M+H)
+. [1148] Compound 136B, arbitrarily assigned as (R)-2-(3-fluoro-5-isopropyl-2- methoxyphenyl)-2-((R)-3-(isobutyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.25 (s, 1H), 7.15 (d, J=7.2 Hz, 1H), 7.07 (dd, J=12.8, 2.0 Hz, 1H), 6.36 (d, J=7.6 Hz, 1H), 4.80 (s, 1H), 3.98 (s, 3H), 3.65-3.18 (m, 5H), 3.02-2.30 (m, 10H), 2.20-1.22 (m, 18H), 0.92–0.88 (m, 6H). LC/MS ESI 569 (M+H)
+. Preparation of 2-(5-(cyclopropylmethyl)-3-fluoro-2-methoxyphenyl)-2-((R)-3-(methyl(5- (5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 137A and 137B) and 2-(5-(but-3-en-1-yl)-3-fluoro-2-methoxyphenyl)-2-((R)-3- (methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 138A and 138B)

[1149] To a solution of ethyl 2-(5-(cyclopropylmethyl)-3-fluoro-2-methoxyphenyl)acetate and ethyl 2-(5-(but-3-en-1-yl)-3-fluoro-2-methoxyphenyl)acetate (ratio ~1:1, 200 mg, 0.75 mmol) in THF (10 mL) at -78°C, was added lithium diisopropylamide solution 2.0 M in THF/hexanes (0.75 mL, 1.5 mmol) dropwise. The reaction was stirred at -78°C for 30 min, then chlorotrimethylsilane (163 mg, 1.5 mmol) was added and the reaction was stirred at -78°C for another 30 min. Then a solution of NBS (267 mg, 1.5 mmol) in THF (4 mL) was added and the reaction was stirred at -78°C for 30 min. The reaction was quenched with H2O (10 mL), then extracted with EtOAc (20 mL × 3). The combined organic phase was washed with Sat. NaHCO3 solution, brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was dissolved in acetonitrile (5 mL), added (R)-N-methyl-N-(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)pyrrolidin-3-amine (302 mg, 1.0 mmol) and DIPEA (390 mg, 3.0 mmol). The reaction was stirred at 60 °C for 15 hours. Solvent was removed in vacuo, and the residue was diluted with MeOH (4 mL), then added LiOH aqueous solution (2 mL, 2 N). The mixture was stirred at 60°C for 4 hours. The mixture was concentrated in vacuo, and neutralized with 1 N HCl aqueous solution and the residue was purified by Prep-HPLC A (30-60% MeCN), then Prep Chiral SFC B to give the diastereomers products Compound 137A (25 mg), Compound 137B (19 mg), Compound 138A (18 mg) and Compound 138B (22 mg) as white solids. [1150] Compound 137A, arbitrarily assigned as (S)-2-(5-(cyclopropylmethyl)-3-fluoro-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.
1H NMR (400 MHz, MeOD) δ 7.24 (s, 1H), 7.18- 7.03 (m, 2H), 6.37 (d, J=7.2 Hz, 1H), 4.63 (s, 1H), 3.96 (s, 3H), 3.40-3.36 (m, 3H), 3.25-2.45 (m, 14H), 2.20-1.28 (m, 11H), 0.98-0.95 (m, 1H), 0.52–0.48 (m, 2H), 0.22–0.18 (m, 2H). LC/MS ESI 539 (M+H)
+. [1151] Compound 137B, arbitrarily assigned as (R)-2-(5-(cyclopropylmethyl)-3-fluoro-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.27 (s, 1H), 7.18- 7.05 (m, 2H), 6.37 (d, J=7.2 Hz, 1H), 4.59 (s, 1H), 3.97 (s, 3H), 3.40-3.36 (m, 3H), 3.25-2.45 (m, 14H), 2.20-1.28 (m, 11H), 0.98-0.95 (m, 1H), 0.52–0.48 (m, 2H), 0.22–0.18 (m, 2H). LC/MS ESI 539 (M+H)
+. [1152] Compound 138A, arbitrarily assigned as (S)-2-(5-(but-3-en-1-yl)-3-fluoro-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.19-7.17 (m, 2H), 7.00 (dd, J=12.4, 2.0 Hz), 6.38 (d, J=7.2 Hz, 1H), 5.85-5.83 (m, 1H), 5.05-4.95 (m, 2H), 4.66 (s,
1H), 3.96 (s, 3H), 3.40-3.36 (m, 3H), 3.25-2.35 (m, 17H), 2.20-1.38 (m, 10H). LC/MS ESI 539 (M+H)+. [1153] Compound 138B, arbitrarily assigned as (R)-2-(5-(but-3-en-1-yl)-3-fluoro-2- methoxyphenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid.1H NMR (400 MHz, MeOD) δ 7.23-7.17 (m, 2H), 6.99 (dd, J=12.4, 2.0 Hz), 6.38 (d, J=7.2 Hz, 1H), 5.85-5.83 (m, 1H), 5.05-4.95 (m, 2H), 4.58 (s, 1H), 3.96 (s, 3H), 3.40-3.36 (m, 3H), 3.25-2.35 (m, 17H), 2.20-1.38 (m, 10H). LC/MS ESI 539 (M+H)+. 2-(2-ethyl-3-fluoro-5-(tetrahydro-2H-pyran-4-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8- tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compounds 139A and 139B)

[1154] Compound 139A, arbitrarily assigned as (S)-2-(2-ethyl-3-fluoro-5-(tetrahydro-2H- pyran-4-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid. LC/MS ESI 567.3(M+H)
+.
1H NMR (400 MHz, MeOD) δ 7.42 (s, 1H), 7.13 (d, J = 7.6 Hz, 1H), 6.84 (dd, J = 11.2, 1.2 Hz, 1H), 6.35 (d, J = 7.2 Hz, 1H), 4.21 (s, 1H), 4.01 – 3.99 (m, 2H), 3.54 – 3.35 (m, 5H), 3.00 – 2.92 (m, 2H), 2.76 – 2.67 (m, 8H), 2.53 – 2.49 (m, 6H), 2.12 – 2.08 (m, 1H), 1.97 – 1.85 (m, 3H), 1.75 – 1.58 (m, 8H), 1.36 – 1.32 (m, 2H), 1.21 – 1.17 (m, 3H). [1155] Compound 139B, arbitrarily assigned as (R)-2-(2-ethyl-3-fluoro-5-(tetrahydro-2H- pyran-4-yl)phenyl)-2-((R)-3-(methyl(5-(5,6,7,8-tetrahydro-1,8-naphthyridin-2- yl)pentyl)amino)pyrrolidin-1-yl)acetic acid. LC/MS ESI 567.3 (M+H)
+.
1H NMR (400 MHz, MeOD) δ 7.37 (s, 1H), 7.20 (d, J = 7.2 Hz, 1H), 6.86 (dd, J = 11.6, 1.2 Hz, 1H), 6.39 (d, J = 7.6 Hz, 1H), 4.18 (s, 1H), 4.02 – 3.99 (m, 2H), 3.55 – 3.34 (m, 5H), 3.13 – 2.95 (m, 2H), 2.80 – 2.68 (m, 7H), 2.55 – 2.46 (m, 6H), 2.06 – 2.03 (m, 1H), 1.92 – 1.84 (m, 3H), 1.75 – 1.60 (m, 8H), 1.46 – 1.28 (m, 3H), 1.20 – 1.16 (m, 3H).
2-(2-cyano-3-fluoro-5-isopropy lphenyl)-2-((R)-3-(methyl (5-(5,6,7, 8-tetrahydro-1,8- naphthyridin-2-yl)pentyl)amino)pyrrolidin-1-yl)acetic acid (Compound 140A and 140B)
[1156] Compound 140A, arbitrarily assigned (S)-2-(2-cyano-3-fluoro-5-isopropy lphenyl)- 2-((R)-3-(methyl (5-(5,6,7, 8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1- yl)acetic acid. LC/MS ESI 522.3 (M+H)
+.
1H NMR (400 MHz, MeOD) δ 7.52 (s, 1H), 7.19 – 7.05 (m, 2H), 6.38 (d, J = 7.3 Hz, 1H), 4.33 (s, 1H), 3.62-3.58 (m, 1H), 3.42 – 3.35 (m, 2H), 3.14 – 2.89 (m, 3H), 2.88 – 2.78 (m, 2H), 2.76 – 2.65 (m, 3H), 2.62 (s, 3H), 2.58-2.42 (m, 3H), 2.19 – 2.07 (m, 1H), 2.05 – 1.83 (m, 3H), 1.72 – 1.56 (m, 4H), 1.42-1.38 (m, 2H), 1.24 (dd, J = 6.9, 1.4 Hz, 6H). [1157] Compound 140B, arbitrarily assigned (R)-2-(2-cyano-3-fluoro-5-isopropy lphenyl)- 2-((R)-3-(methyl (5-(5,6,7, 8-tetrahydro-1,8-naphthyridin-2-yl)pentyl)amino)pyrrolidin-1- yl)acetic acid. LC/MS ESI 522.3 (M+H)
+.
1H NMR (400 MHz, MeOD) δ 7.54 (s, 1H), 7.23-7.14 (m, 2H), 6.39 (d, J = 7.3 Hz, 1H), 4.12 (s, 1H), 3.59 (s, 1H), 3.43 – 3.35 (m, 2H), 3.23 – 2.92 (m, 3H), 2.87 – 2.63 (m, 5H), 2.59 (s, 3H), 2.51-2.45 (m, 3H), 2.13 – 2.02 (m, 1H), 1.89-1.80 (m, 3H), 1.77-1.62 (m, 4H), 1.45-1.43 (m, 2H), 1.25 (d, J = 6.9 Hz, 6H). Assay Conditions [1158] Fluorescence Polarization (FP) assays were used to measure compound activity through binding competition with a small molecule ligand fluorescently labeled with TAMRA dye and having a Kd of 0.7 nM. In the assay, 1 nM of integrin avb8 was incubated with the test compound in 2 mM manganese chloride, 0.1 mM calcium chloride, 20 mM HEPES buffer at pH 7.3, 150 mM sodium chloride, 0.01% Triton X-100, 2% DMSO, and 3 nM of the fluorescein- labeled peptide. The assays were run in 384-well plates. The integrin protein was pre-incubated with the test compounds for 15 minutes at 22 ⁰C before the small molecule ligand was added.
After the small molecule ligand was added, the assay was incubated at 22 ⁰C for 1 hour and fluorescence polarization was measured. IC
50 values were determined by nonlinear regression, 4- parameter curve fitting. [1159] IC50 values of Tables 1 and 2 as determined by the fluorescence polarization assay are categorized as: A: < 5 nM; B: 5-500 nM; and C: >500 nM. [1160] Compounds described herein can be selective for avb8 over other integrins such as avb6. The avb6/avb8 selectivity values of Tables 1 and 2 are the ratios of the avb6 IC50 value divided by the avb8 IC50 value as determined by the fluorescence polarization assay. These values are categorized as shown below: A: >1000; B: 100-1000; C: 10<selectivity<100; and D: <10. Table 1. Potency Data Table
Table 2. Comparative Potency Data Table
[1161] From the foregoing description, one skilled in the art can easily ascertain the essential characteristics of this invention, and without departing from the spirit and scope thereof, can make various changes and modifications of the invention to adapt it to various usages and conditions.
[1162] All references, patents or applications, U.S. or foreign, cited in the application are hereby incorporated by reference as if written herein in their entireties. Where any inconsistencies arise, material literally disclosed herein controls. Additional Embodiments [1163] In some embodiments, a compound or use of the present invention can be selected from one or more of the enumerated emboidments provided below. 1. A compound of Formula (I) or a pharmaceutically acceptable salt thereof:
wherein:
to 6 R
4; each of R
1a, R
1b, R
1c, R
1d, R
1e, and R
1f is independently H, C
1-4alkyl, halogen, C
1- 4alkyloxy, OH, C
1-4alkyl-OH, C
1-4alkyl-C
1-4alkyloxy, C
1-4alkyloxy-C
1-4alkyloxy, CF
3, CHF
2, CH
2F, CN, NO
2, NR
aR
b or C
1-4alkyl-NR
aR
b, R
2 is H, C1-4alkyl or C3-5cycloalkyl, each of C1-4alkyl and C3-5cycloalkyl is optionally substituted with 1 to 4 R
5; R
3a is C1-4alkyloxy, C3-5cycloalkoxy, CF3, CHF2, CH2F, OCF3, OCHF2 or OCH2F; R
3b is H, halogen, CF
3 or CN; R
3c is H, F, CN, or C
1-4alkyl;
R
3d is H, C
1-4alkyl, C
3-5cycloalkyl, or 4-6-membered heterocycloalkyl, each of which is optionally substituted with 1 to 4 R
6; R
3e is H or F; each R
4 is independently H, C1-4alkyl, halogen, CF3, CHF2 or CH2F, cyclopropyl, or two geminal R
4 groups together can form a spiro-cyclopropyl; each R
5 is independently H, C
1-4alkyl, C
3-5cycloalkyl, C
1-4alkyloxy, C
3-5cycloalkoxy, F, CF
3, CHF
2, CH
2F, OCF
3, OCHF
2 or OCH
2F; each R
6 is independently H, C
1-4alkyl, C
2-4alkenyl, C
3-5cycloalkyl, C
1-4alkyloxy, C
3- 5cycloalkoxy, F, CF3, CHF2, CH2F, OCF3, OCHF2, OCH2F, OH, 5-6-membered heteroaryl, or NRaRb; each R
7 is independently C1-4alkyl or F; each of R
a and R
b is independently hydrogen, C
1-4alkyl, C
3-5cycloalkyl, or R
a and R
b, together with the nitrogen atom to which they are attached, form a saturated or unsaturated heterocyclic ring containing from three to seven ring atoms, which ring may optionally contain additional one or two heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur and may be optionally substituted by from one to three groups which may be the same or different selected from the group consisting of F, C1-4alkyl, phenyl and benzyl; and n is 0, 1 or 2; and m is 0, 1 or 2. 2. The compound of Embodiment 1, wherein the Q ring i
. 3. The compound of Embodiment 1 or 2, wherein each of R
1c, R
1d, and R
1e is independently H. 4. The compound of any one of Embodiments 1-3, wherein each R
1a is independently H. 5. The compound of any one of Embodiments 1-4, wherein each R
1b is independently H. 6. The compound of any one of Embodiments 1-4, wherein each R
1b is independently OMe.
7. The compound of Embodiment 1, wherein the Q ring i
8. The compound of Embodiment 7, wherein each of R
1c and R
1d is independently H. 9. The compound of Embodiment 7 or 8, wherein each of R
1a, R
1b and R
1f is independently H. 10. The compound of any one of Embodiments 1-9, wherein L is
. 11. The compound of any one of Embodiments 1-9, wherein L is
. 12. The compound of any one of Embodiments 1-11, wherein R
2 is C1-4alkyl. 13. The compound of any one of Embodiments 1-11, wherein R
2 is methyl. 14. The compound of any one of Embodiments 1-11, wherein R
2 is ethyl. 15. The compound of any one of Embodiments 1-11, wherein R
2 is cyclopropyl. 16. The compound of any one of Embodiments 1-15, wherein R
3a is C1-4alkyloxy. 17. The compound of any one of Embodiments 1-15, wherein R
3a is OMe, OEt, OCF3, OCHF2 or OCH2F. 18. The compound of any one of Embodiments 1-15, wherein R
3a is OMe. 19. The compound of any one of Embodiments 1-18, wherein R
3b is F. 20. The compound of any one of Embodiments 1-19, wherein R
3c is H. 21. The compound of any one of Embodiments 1-20, wherein R
3d is C1-4alkyl. 22. The compound of any one of Embodiments 1-20, wherein R
3d is C3-6cycloalkyl. 23. The compound of any one of Embodiments 1-20, wherein R
3d is oxetanyl, tetrahydrofuranyl, tetrahydro-2H-pyranyl, morpholinyl or piperazinyl-C
1-4alkyl.
24. The compound of any one of Embodiments 1-20, wherein R
3d is iso-propyl. 25. The compound of any one of Embodiments 1-24, wherein R
3e is H. 26. The compound of any one of Embodiments 1-25, wherein each R
4 is independently methyl. 27. The compound of any one of Embodiments 1-25, wherein each R
4 is independently F. 28. The compound of any one of Embodiments 1-25, wherein each R
4 is independently CF
3, CHF
2 or CH
2F. 29. The compound of any one of Embodiments 1-25, wherein each R
4 is independently H. 30. The compound of any one of Embodiments 1-29, wherein each R
5 is independently C1- 4alkyl. 31. The compound of any one of Embodiments 1-29, wherein each R
5 is independently C3- 5cycloalkyl. 32. The compound of any one of Embodiments 1-29, wherein each R
5 is independently C
1- 4alkyloxy. 33. The compound of any one of Embodiments 1-29, wherein each R
5 is independently C3- 5cycloalkoxy. 34. The compound of any one of Embodiments 1-29, wherein each R
5 is independently F, CF
3, CHF
2, CH
2F, OCF
3, OCHF
2 or OCH
2F. 35. The compound of any one of Embodiments 1-29, wherein each R
5 is independently H. 36. The compound of any one of Embodiments 1-35, wherein each R
6 is independently C
1- 4alkyl. 37. The compound of any one of Embodiments 1-35, wherein each R
6 is independently C2- 4alkenyl. 38. The compound of any one of Embodiments 1-35, wherein each R
6 is independently C
3- 5cycloalkyl.
39. The compound of any one of Embodiments 1-35, wherein each R
6 is independently C
1- 4alkyloxy. 40. The compound of any one of Embodiments 1-35, wherein each R
6 is independently C3- 5cycloalkoxy. 41. The compound of any one of Embodiments 1-35, wherein each R
6 is independently F, CF
3, CHF
2, CH
2F, OCF
3, OCHF
2, OCH
2F or OH. 42. The compound of any one of Embodiments 1-35, wherein each R
6 is independently a 5-6- membered heteroaryl. 43. The compound of any one of Embodiments 1-35, wherein each R
6 is independently H. 44. The compound of any one of Embodiments 1-43, wherein each R
7 is independently methyl or F. 45. The compound of any one of Embodiments 1-44, wherein n is 0. 46. The compound of any one of Embodiments 1-44, wherein n is 1. 47. The compound of any one of Embodiments 1-44, wherein n is 2. 48. The compound of any one of Embodiments 1-47, wherein m is 0. 49. The compound of Embodiment 1, having a structure according to Formula (II),
pharmaceutically acceptable salt thereof.
50. The compound of Embodiment 49, having a structure according to Formula (IIA),
pharmaceutically acceptable salt thereof. 51. The compound of Embodiment 49, having a structure according to Formula (IIB),
pharmaceutically acceptable salt thereof. 52. The compound of Embodiment 1, having a structure according to Formula (III),
pharmaceutically acceptable salt thereof. 53. The compound of Embodiment 52, having a structure according to Formula (IIIA),
pharmaceutically acceptable salt thereof.
54. The compound of Embodiment 52, having a structure according to Formula (IIIB),
pharmaceutically acceptable salt thereof. 55. The compound of Embodiment 1, having a structure according to Formula (IV),
pharmaceutically acceptable salt thereof. 56. The compound of Embodiment 55, having a structure according to Formula (IVA),
pharmaceutically acceptable salt thereof.
57. The compound of Embodiment 55, having a structure according to Formula (IVB),
pharmaceutically acceptable salt thereof. 58. The compound of Embodiment 1, having a structure according to Formula (V),
pharmaceutically acceptable salt thereof. 59. The compound of Embodiment 58, having a structure according to Formula (VA),
pharmaceutically acceptable salt thereof. 60. The compound of Embodiment 58, having a structure according to Formula (VB),
pharmaceutically acceptable salt thereof.
61. The compound of any one of Embodiments 49-60, wherein the carbon marked by the asterisk has the (R)-configuration. 62. The compound of any one of Embodiments 49-60, wherein the carbon marked by the asterisk has the (S)-configuration. 63. The compound of any one of Embodiments 49-62, wherein R
2 is C1-4alkyl optionally substituted with 1 or 2 R
5. 64. The compound of Embodiment 63, wherein R
2 is –CH
3, -CH
2CH
3, -CH(CH
3)
2, -C(CH
3)
3, -CH
2CH(CH
3)
2, -CH
2CF
3, -(CH
2)
2CF
3, -CHF
2, -CH
2CHF
2, -(CH
2)
2CHF
2, -(CH2)2OCH3, -(CH2)2O-(cyclopropyl), -(CH2)2OCF3, -(CH2)2C(OCH3)(CH3)CH3, or cyclopropyl. 65. The compound of Embodiment 64, wherein R
2 is –CH3. 66. The compound of any one of Embodiments 49-65, wherein R
3c is H. 67. The compound of any one of Embodiments 49-65, wherein R
3c is F. 68. The compound of any one of Embodiments 49-50, 52-53, 55-56, and 58-59 wherein R
3d is isopropyl. 69. The compound of any one of Embodiments 51, 54, 57, and 60 wherein R
3b is F. 70. The compound of any one of Embodiments 49-69, wherein L is unsubstituted
. 71. The compound of any one of Embodiments 49-69, wherein L is
substituted by 1 or 2 R
5. 72. The compound of Embodiment 71, wherein each R
5 is independently selected from -CH3, -CH2F, and –F. 73. The compound of any one of Embodiments 49-69, wherein L is unsubstituted
.
74. The compound of Embodiment 1 or a pharmaceutically acceptable salt thereof, wherein said compound is selected from Table 1. 75. The compound of Embodiment 1, selected from:
or a pharmaceutically acceptable salt thereof. 76. The compound of Embodiment 75, wherein the compound is
pharmaceutically acceptable salt thereof. 77. The compound of Embodiment 75, wherein the compound is
pharmaceutically acceptable salt thereof.
78. The compound of Embodiment 75, wherein the compound is
pharmaceutically acceptable salt thereof. 79. The compound of Embodiment 75, wherein the compound is
pharmaceutically acceptable salt thereof. 80. The compound of Embodiment 75, wherein the compound is
pharmaceutically acceptable salt thereof. 81. The compound of Embodiment 75, wherein the compound is
pharmaceutically acceptable salt thereof.
82. The compound of Embodiment 75, wherein the compound is
pharmaceutically acceptable salt thereof. 83. The compound of Embodiment 75, wherein the compound is
pharmaceutically acceptable salt thereof. 84. The compound of Embodiment 75, wherein the compound is
pharmaceutically acceptable salt thereof. 85. The compound of Embodiment 75, wherein the compound is
pharmaceutically acceptable salt thereof.
86. The compound of Embodiment 75, wherein the compound is
pharmaceutically acceptable salt thereof. 87. The compound of Embodiment 75, wherein the compound is
pharmaceutically acceptable salt thereof. 88. The compound of Embodiment 75, wherein the compound is
pharmaceutically acceptable salt thereof. 89. The compound of Embodiment 75, wherein the compound is
pharmaceutically acceptable salt thereof.
90. The compound of Embodiment 75, wherein the compound is
pharmaceutically acceptable salt thereof. 91. The compound of Embodiment 75, wherein the compound is
acceptable salt thereof. 92.
acceptable salt thereof. 93. The compound of Embodiment 75, wherein the compound is
pharmaceutically acceptable salt thereof.
94. The compound of Embodiment 75, wherein the compound is
pharmaceutically acceptable salt thereof. 95. A pharmaceutical composition, comprising a compound of any one of Embodiments 1- 94, or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient. 96. A method of inhibiting α
vβ
8 integrin in a patient, the method comprising administering to the patient in need thereof a therapeutically effective amount of a compound of any one of Embodiments 1-94, or a pharmaceutically acceptable salt thereof. 97. The method of Embodiment 96, wherein the method is for treating a solid tumor in a patient in need thereof. 98. A method of treating solid tumor in a patient, the method comprising administering to the patient in need thereof (a) a therapeutically effective amount of a compound of any one of Embodiments 1-94, or a pharmaceutically acceptable salt thereof, and (b) a therapeutically effective amount of a second active agent. 99. The method of Embodiment 98, wherein the solid tumor is selected from: anal cancer, bile duct cancer, bladder cancer, breast cancer, cervical cancer, colorectal cancer, endometrial cancer, esophageal cancer, fallopian tube cancer, gastric cancer, glioma liver cancer, lung cancer, melanoma, nasopharyngeal carcinoma, neuroblastoma, osteosarcoma, ovarian cancer, pancreatic cancer, primary peritoneal carcinoma, prostate cancer, renal cell carcinoma, skin cancer, squamous cell carcinoma of the head and neck (SCCHN), testicular cancer, urothelial carcinoma, and uterine cancer. 100. The method of Embodiment 99, wherein the solid tumor is selected from: breast cancer, squamous cell carcinoma of the head and neck (SCCHN), renal cell
carcinoma, ovarian cancer, gastric cancer, esophageal cancer, lung cancer, pancreatic cancer, bile duct cancer, endometrial cancer, melanoma, and urothelial carcinoma. 101. The method of any one of Embodiments 98-100, wherein the second active agent is an immune checkpoint inhibitor. 102. The method of Embodiment 101, wherein the immune checkpoint inhibitor is an anti-PD-1 or an anti-PD-L1 therapy. 103. The method of Embodiment 102, wherein the immune checkpoint inhibitor is selected from nivolumab, pembrolizumab, cemiplimab, dostarlimab, atezolizumab, avelumab, and durvalumab.