WO2024170723A1 - Method of pathogen control in soybean - Google Patents
Method of pathogen control in soybean Download PDFInfo
- Publication number
- WO2024170723A1 WO2024170723A1 PCT/EP2024/053952 EP2024053952W WO2024170723A1 WO 2024170723 A1 WO2024170723 A1 WO 2024170723A1 EP 2024053952 W EP2024053952 W EP 2024053952W WO 2024170723 A1 WO2024170723 A1 WO 2024170723A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- diaporthe
- spp
- compound
- formula
- soybean
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01P—BIOCIDAL, PEST REPELLANT, PEST ATTRACTANT OR PLANT GROWTH REGULATORY ACTIVITY OF CHEMICAL COMPOUNDS OR PREPARATIONS
- A01P3/00—Fungicides
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/56—1,2-Diazoles; Hydrogenated 1,2-diazoles
Definitions
- the present invention relates to a method of pathogen control in soybean crops.
- it relates to the use of a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, and to a method of using of said compound or composition.
- WO 2007/048556 discloses that certain heterocyclic amide derivatives have microbicidal activity, such as fungicidal activity.
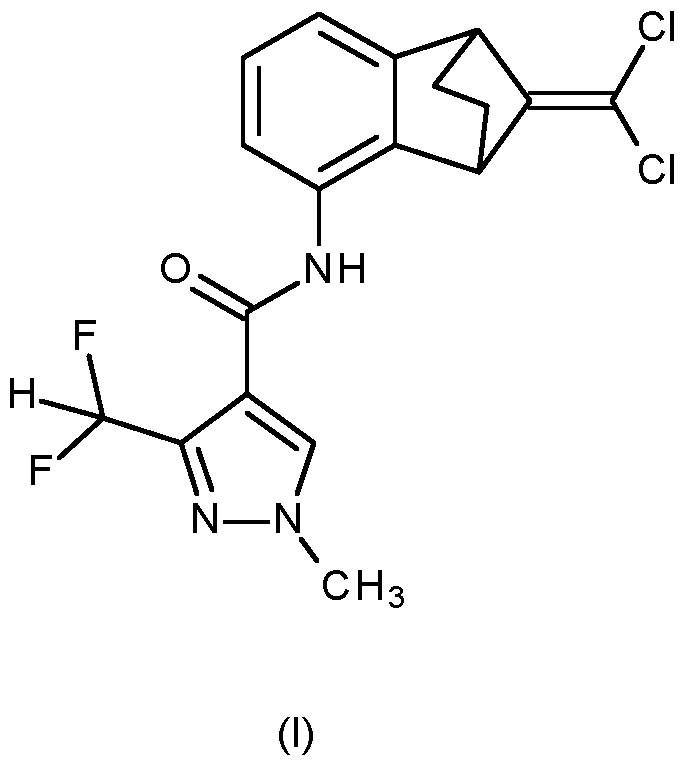
- a compound of formula (I) 3-difluoromethyl-1-methyl-1 H-pyrazole-4- carboxylic acid(9-dichloromethylene-1 ,2,3,4-tetrahydro-1 ,4-methano-naphthalen-5-yl)-amide is disclosed:
- the compound of formula (I) refers to benzovindiflupyr as active ingredient, with the following CAS number: 1072957-71-1.
- Symptoms appeared as rot at the junction of the pods, evolving into a partial or total necrosis of the pod, and causing rot, opening and sometimes abortion of the pods. Seeds from the pods were also compromised and appeared wrinkled and differed in colour in comparison with normal (healthy) seeds (for example compromised seeds appear either darker than or paler than typically healthy seeds).
- pod anamoly represents a new disease of Soybean
- the cause may be abiotic (e.g environmental), biotic (some form of phytopathogen) or even from a confluence of both.
- anthracnose a disease caused by the fungus Colleotricum truncatum
- the present invention stems from the finding that several different phytopathogenic fungi are present in the tissue from soybean plants affected by pod anomaly, as shown herein in the Examples.
- the present invention provides the use of a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean.
- a method of combating, preventing or controlling pod anomaly in soybean which comprises applying to the crop of soybean plants, the locus thereof, or propagation material thereof, a compound of formula (I) or a composition comprising a compound of formula (I).
- a method of combating, preventing or controlling seed decay in post harvest soybean which comprises applying to the crop of soybean plants, the locus thereof, or propagation material thereof, a compound of formula (I) or a composition comprising a compound of formula (I).
- a compound of formula (I) or a composition comprising a compound of formula (I) means reducing any undesired effect, such as pathogenic and more particularly phytopathogenic, especially fungi such as (but not limited to) Cercospora spp., Colletotrichum spp., Corynespora spp. or Diaporthe spp , infestation or attack of, and pathogenic damage to a plant or to a plant derived product to such a level that an improvement is demonstrated.
- pod anomaly refers to a phenotypic pathology of soybean pods, wherein the pod exhibits at least partial necrosis and/or seeds from affected pods develop abnormally. Pod anomaly has also been associated with the breakage of stems/stem blight, wherein black or brown streaks have been observed externally on stems and the inner marrow of stems exhibits necrosis. Where these symptoms also occur, the disease pathology is referred to herein as Soybean anomaly.
- the compound of formula (I) can occur in two different stereoisomers, which are described as the single enantiomers of formulae (l)i and (l) N :
- racemic compound of formula (I) means a racemic mixture of compounds of formula (l)i and (l)u.
- the compound of formula (I) may be prepared as described in WO 2007/048556.
- the compound of formula (I) can be applied in a fungicidally effective amount.
- the term "effective amount” refers to the amount of the compound, which, upon single or multiple applications provides the desired effect.
- An effective amount is readily determined by the skilled person in the art, by the use of known techniques and by observing results obtained under analogous circumstances. In determining the effective amount, a number of factors are considered including, but not limited to: the type of plant or derived product to be applied; the pathogen to be controlled & its lifecycle; the particular compound applied; the type of application; and other relevant circumstances.
- the compound of formula (I) according to the invention is suitable for use on any soybean plant, including those that have been genetically modified to be resistant to active ingredients such as herbicides, or to produce biologically active compounds that control infestation by plant pests.
- transgenic plants and plant cultivars obtained by genetic engineering methods are treated.
- plants of the plant cultivars which are in each case commercially available or in use are treated according to the invention.
- Plant cultivars are understood as meaning plants having novel properties ("traits") which have been obtained by conventional breeding, by mutagenesis or by recombinant DNA techniques. These can be cultivars, bio- or genotypes.
- the preferred transgenic plants or plant cultivars which are to be treated according to the invention include all plants which, by virtue of the genetic modification, received genetic material which imparts particularly advantageous, useful traits to these plants. Examples of such traits are better plant growth, increased tolerance to high or low temperatures, increased tolerance to drought or to water or soil salt content, increased flowering performance, easier harvesting, accelerated maturation, higher harvest yields, higher quality and/or a higher nutritional value of the harvested products, better storage stability and/or processability of the harvested products.
- the transgenic plants or plant cultivars are selected from Bt soybean plants.
- Traits that are emphasized in particular are the increased defence of the plants against insects, arachnids, nematodes and slugs and snails by virtue of toxins formed in the plants, in particular those formed in the plants by the genetic material from Bacillus thuringiensis (for example by the genes CrylA(a), CrylA(b), CrylA(c), CryllA, CrylllA, CrylllB2, Cry9c, Cry2Ab, Cry3Bb and CrylF and also combinations thereof) (referred to herein as "Bt plants”).
- Traits that are also particularly emphasized are the increased defence of the plants against fungi, bacteria and viruses by systemic acquired resistance (SAR), systemin, phytoalexins, elicitors and resistance genes and correspondingly expressed proteins and toxins.
- SAR systemic acquired resistance
- trasits that are furthermore particularly emphasized are the increased tolerance of the plants to certain herbicidally active compounds, for example imidazolinones, sulphonylureas, glyphosate or phosphinotricin (for example the "PAT" gene).
- herbicidally active compounds for example imidazolinones, sulphonylureas, glyphosate or phosphinotricin (for example the "PAT” gene).
- the genes which impart the desired traits in question can also be present in combination with one another in the transgenic plants.
- Examples of “Bt plants” are soybean varieties which are sold under the trade names YIELD GARD® or INTACTA®.
- herbicide-tolerant plants which may be mentioned are soybean varieties which are sold under the trade names Roundup Ready® (tolerance to glyphosate), Liberty Link® (tolerance to phosphinotricin), IMI® (tolerance to imidazolinones) and STS® (tolerance to sulphonylureas).
- Herbicide-resistant plants plants bred in a conventional manner for herbicide tolerance
- plants bred in a conventional manner for herbicide tolerance include the varieties sold under the name Clearfield® (for example maize).
- soybean plants carrying traits conferring resistance to 2.4D e.g. Enlist®
- glyphosate e.g. Roundup Ready®, Roundup Ready 2 Yield®
- sulfonylurea e.g. STS®
- glufosinate e.g. Liberty Link®, Ignite®
- Dicamba Monsanto
- HPPD tolerance e.g. isoxaflutole herbicide
- Double or triple stack in soybean plants of any of the traits described here are also of interest, including glyphosate and sulfonyl-urea tolerance (e.g.
- the genetic modified crops can be described in the following website https://www.isaaa.org/gmapprovaldatabase, incorporated by reference therein, and more particularly can be selected from:
- the use of a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean wherein the pod anomaly comprises symptoms of pod decay, abnormal seed phenotype and/or stem blight.
- the pod anomaly manifests at the R4 or R5 stage of the soybean plants reproductive cycle.
- the pod anomaly manifests at the R5 stage of the soybean plants reproductive cycle.
- the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp. and Diaporthe spp.
- the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp. Even more preferably, the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp., wherein the Diaporthe species comprise at least Diaporthe miriciae and Diaporthe longicolla, and Fusarium spp.
- the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., wherein the Colletotrichum species comprise at least Colletotrichum cliviicola and Colletotrichum truncatum, Corynespora spp., Diaporthe spp., wherein the Diaporthe species comprise at least Diaporthe miriciae and Diaporthe longicolla, and Fusarium spp.
- the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., wherein the Colletotrichum species comprise at least Colletotrichum cliviicola and Colletotrichum truncatum, Corynespora spp., wherein the Corynespora spp. species comprise at least Corynespora cassiicola., Diaporthe spp., wherein the Diaporthe species comprise at least Diaporthe miriciae and Diaporthe longicolla, and Fusarium spp.
- the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., wherein the Colletotrichum species comprise at least Colletotrichum cliviicola and Colletotrichum truncatum, Corynespora spp., wherein the Corynespora spp. species comprise at least Corynespora cassiicola., Diaporthe spp., wherein the Diaporthe species comprise at least Diaporthe miriciae and Diaporthe longicolla, and Fusarium spp., wherein the Fusarium spp. species comprise at least Fusarium fujikuroi and Fusarium incarnatum.
- a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp. and Diaporthe spp.
- the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp.
- a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp. and Diaporthe spp. wherein the Colletotrichum species comprise at least Colletotrichum cliviicola and Colletotrichum truncatum.
- a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp. and Diaporthe spp. wherein the Corynespora spp. species comprise at least Corynespora cassiicola.
- a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp. wherein the Fusarium spp. species comprise at least Fusarium fujikuroi and Fusarium incarnatum.
- a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp.
- the Diaporthe species comprise at least Diaporthe miriciae and Diaporthe longicolla (more preferably at least Diaporthe longicolla, Diaporthe miriciae, Diaporthe macadamiae and Diaporthe pyracanthae) and the Fusarium spp. species comprise at least Fusarium fujikuroi and Fusarium incarnatum.
- a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp.
- Diaporthe species comprise at least Diaporthe miriciae and Diaporthe longicolla (more preferably at least Diaporthe longicolla, Diaporthe miriciae, Diaporthe macadamiae and Diaporthe pyracanthae)
- the Colletotrichum species comprise at least Colletotrichum cliviicola and Colletotrichum truncatum
- the Fusarium spp. species comprise at least Fusarium fujikuroi and Fusarium incarnatum.
- a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp.
- Diaporthe species comprise at least Diaporthe miriciae and Diaporthe longicolla (more preferably at least Diaporthe longicolla, Diaporthe miriciae, Diaporthe macadamiae and Diaporthe pyracanthae),
- the Colletotrichum species comprise at least Colletotrichum cliviicola and Colletotrichum truncatum
- the Corynespora spp. species comprise at least Corynespora cassiicola and the Fusarium spp. species comprise at least Fusarium fujikuroi and Fusarium incarnatum.
- Diaporthe species also known as Phomopsis species, are phytopathogenic fungi which can more preferably comprise at least one of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
- Diaporthe miriciae is also known as Diaporthe ueckeri or Diaporthe ueckerae.
- the preferred species is Diaporthe miriciae.
- the Diaporthe species can comprise at least two of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
- the two preferred species are Diaporthe miriciae and Diaporthe longicolla.
- the Diaporthe species can comprise at least three of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
- the three preferred species are Diaporthe miriciae, Diaporthe longicolla, and Diaporthe masirevicii.
- the Diaporthe species can comprise at least four of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
- the four preferred species are Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
- the Diaporthe species can comprise at least five of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
- the five preferred species are Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, Diaporthe pyracanthae, and Diaporthe sojae.
- the Diaporthe species can comprise Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
- the Diaporthe species can comprise at least one of the species selected from Diaporthe miriciae, Diaporthe sojae, and Diaporthe macadamiae. More preferably, the Diaporthe species can comprise at least two of the species selected from Diaporthe miriciae, Diaporthe sojae, and Diaporthe macadamiae. Even more preferably, the Diaporthe species can comprise Diaporthe miriciae, Diaporthe sojae, and Diaporthe macadamiae.
- a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp.
- the soybean is a transgenic soybean plant (preferably a transgenic Bt soybean plant, even more preferably, the transgenic soybean plant comprises an event selected from the list consisting of event MON87701 , event MON87751 , event DAS- 81419-2, event DAS-44406-6/pDAB8264.44.06.1 , event DAS-14536-7/pDAB8291 .45.36.2, event DAS- 68416-4, event DP-305423-1 , event DP-356043-5, event FG72, event LL27, event LL55, event EE- GM3/FG72 (optionally stacked with event EE-GM1/LL27 or event EE-GM2/LL55), event MON87705, event MON87708, event MON87712, event MON87754, event MON87769, event MON89788, event SYHT0H2/SYN-000H2-5, event DAS-21606-3, event 8264.44.06.1 , event pDAB8291 .45.36.2
- a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of 1 to 15 % Corynespora spp, from 10 to 70% Diaporthe spp, from 5 to 35% Cercospora spp and from 10 to 60% Colletotrichum spp, (the skilled person would appreciate the total amount of phytopathogenic fungi is 100% and that the proportion of percentage values of each species can vary in the pod anomaly disease complex).
- the pod anomaly comprises phytopathogenic fungi selected from the group consisting of 1 to 15 % Corynespora spp, from 10 to 70% Diaporthe spp, from 5 to 35% Cercospora spp., from 10 to 60% Colletotrichum spp, and from 5 to 20% Fusarium spp. More preferably, the pod anomaly comprises phytopathogenic fungi selected from the group consisting of 1 to 5 % Corynespora spp, from 20 to 60% Diaporthe spp, from 5 to 20% Cercospora spp., from 10 to 30% Colletotrichum spp, and from 10 to 20% Fusarium spp.
- a compound of formula (I) or a composition comprising a compound of formula (I) to control seed decay in post harvest soybean, during the growing period of the soybean.
- a compound of formula (I) or a composition comprising a compound of formula (I) to control seed decay in post harvest soybean, wherein the seed comprises phytopathogenic fungi selected from the group consisting of Colletotrichum spp. and Diaporthe spp.
- composition comprising a compound of formula (I) to control seed decay in post harvest soybean
- the composition comprising a compound of formula (I) further comprises at least one fungicidally active compound. More preferably the at least one fungicidally active compound is selected from the list consisting of azoxystrobin, cyproconazole, difenoconazole, mancozeb, propiconazole, proth ioconazole and pydiflumetofen.
- compositions comprising a compound of formula (I) to control seed decay in post harvest soybean, wherein the composition comprising a compound of formula (I) further comprises two fungicidally active compounds (preferably the fungicidally active compounds are selected from the list consisting of azoxystrobin, cyproconazole, difenoconazole, mancozeb, propiconazole, prothioconazole and pydiflumetofen). More preferably, there is provided, the use of a composition comprising a compound of formula (I) to control seed decay in post harvest soybean, wherein the composition comprises a compound of formula (I), prothioconazole and mancozeb.
- the use of the compound of formula (I) according to the invention or a composition thereof is applied at a rate of from 5 to 200 g a.i./ha, preferably at a rate of from 10 to 100 g a.i./ha, more preferably at a rate of from 20 to 80 g a.i/ha, even more preferably from 25 to 75 g a.i/ha, even more preferably still from 50 to 75 g a.i/ha.
- a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean wherein the composition comprising a compound of formula (I) further comprises at least one fungicidally active compound. More preferably the at least one fungicidally active compound is selected from the list consisting of azoxystrobin, cyproconazole, difenoconazole, mancozeb, propiconazole, prothioconazole and pydiflumetofen.
- the azoxystrobin is applied at a rate of from 25 to 75 g a.i/ha.
- the cyproconazole is applied at a rate of from 25 to 75 g a.i/ha.
- the difenoconazole is applied at a rate of from 35 to 115 g a.i/ha.
- the mancozeb is applied at a rate of from 500 to 2,000 g a.i/ha.
- the propiconazole is applied at a rate of from 25 to 75 g a.i/ha.
- the prothioconazole is applied at a rate of from 50 to 150 g a.i/ha.
- the pydiflumetofen is applied at a rate of from 25 to 75 g a.i/ha.
- a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp.
- composition comprising a compound of formula (I) further comprises at least one fungicidally active compound (preferably at least one fungicidally active compound is selected from the list consisting of azoxystrobin, cyproconazole, difenoconazole, mancozeb, propiconazole, prothioconazole and pydiflumetofen).
- at least one fungicidally active compound is selected from the list consisting of azoxystrobin, cyproconazole, difenoconazole, mancozeb, propiconazole, prothioconazole and pydiflumetofen).
- compositions comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp.
- composition comprising a compound of formula (I) further comprises two fungicidally active compounds (preferably the fungicidally active compounds are selected from the list consisting of azoxystrobin, cyproconazole, difenoconazole, mancozeb, propiconazole, prothioconazole and pydiflumetofen).
- fungicidally active compounds are selected from the list consisting of azoxystrobin, cyproconazole, difenoconazole, mancozeb, propiconazole, prothioconazole and pydiflumetofen).
- composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp.
- composition comprising a compound of formula (I) further comprises prothioconazole and mancozeb (preferably, the compound of formula (I) is applied at a rate of from 5 to 200 g a.i./ha, more preferably at a rate of from 10 to 100 g a.i./ha, even more preferably at a rate of from 20 to 80 g a.i/ha, even more preferably still from 25 to 75 g a.i/ha, yet even more preferably still from 50 to 75 g a.i/ha).
- prothioconazole and mancozeb preferably, the compound of formula (I) is applied at a rate of from 5 to 200 g a.i./ha, more preferably at a rate of from 10 to 100 g a.i./ha, even more preferably at a rate of from 20 to 80 g a.i/ha, even more preferably still from 25 to 75 g a.i/ha, yet even more preferably still from 50 to 75 g
- a compound of formula (I) or a composition comprising a compound of formula (I) to control seed decay in post harvest soybean, wherein the composition comprising a compound of formula (I) further comprises at least one fungicidally active compound (preferably at least one fungicidally active compound is selected from the list consisting of azoxystrobin, cyproconazole, difenoconazole, mancozeb, propiconazole, prothioconazole and pydiflumetofen).
- the compound used according to the invention can be used in unmodified form, but it is generally formulated into compositions in various ways using formulation adjuvants, such as carriers, solvents and surface-active substances.
- the formulations can be in various physical forms, e.g. in the form of dusting powders, gels, wettable powders, water-dispersible granules, water-dispersible tablets, effervescent pellets, emulsifiable concentrates, microemulsifiable concentrates, oil-in-water emulsions, oil-flowables, aqueous dispersions, oily dispersions, suspo-emulsions, capsule suspensions, emulsifiable granules, soluble liquids, water-soluble concentrates (with water or a water-miscible organic solvent as carrier), impregnated polymer films or in other forms known e.g.
- Such formulations can either be used directly or diluted prior to use.
- the dilutions can be made, for example, with water, liquid fertilisers, micronutrients, biological organisms, oil or solvents.
- the formulations used according to the invention can be prepared e.g. by mixing the compound of formula (I) (‘active ingredient’) with the formulation adjuvants in order to obtain compositions in the form of finely divided solids, granules, solutions, dispersions or emulsions.
- the active ingredient can also be formulated with other adjuvants, such as finely divided solids, mineral oils, oils of vegetable or animal origin, modified oils of vegetable or animal origin, organic solvents, water, surface-active substances or combinations thereof.
- the active ingredient can also be contained in very fine microcapsules.
- Microcapsules contain the active ingredient in a porous carrier. This enables the active ingredient to be released into the environment in controlled amounts (e.g. slow-release).
- Microcapsules usually have a diameter of from 0.1 to 500 microns. They contain the active ingredient in an amount of about from 25 to 95 % by weight of the capsule weight.
- the active ingredient can be in the form of a monolithic solid, in the form of fine particles in solid or liquid dispersion or in the form of a suitable solution.
- the encapsulating membranes can comprise, for example, natural or synthetic rubbers, cellulose, styrene/butadiene copolymers, polyacrylonitrile, polyacrylate, polyesters, polyamides, polyureas, polyurethane or chemically modified polymers and starch xanthates or other polymers that are known to the person skilled in the art.
- very fine microcapsules can be formed in which the active ingredient is contained in the form of finely divided particles in a solid matrix of base substance, but the microcapsules are not themselves encapsulated.
- liquid carriers there may be used: water, toluene, xylene, petroleum ether, vegetable oils, acetone, methyl ethyl ketone, cyclohexanone, acid anhydrides, acetonitrile, acetophenone, amyl acetate, 2-butanone, butylene carbonate, chlorobenzene, cyclohexane, cyclohexanol, alkyl esters of acetic acid, diacetone alcohol, 1 ,2-dichloropropane, diethanolamine, p- diethylbenzene, diethylene glycol, diethylene glycol abietate, diethylene glycol butyl ether, diethylene glycol ethyl ether, diethylene glycol methyl ether, /V,/V-dimethylformamide, dimethyl sulfoxide, 1 ,4- dioxane,
- Suitable solid carriers are, for example, talc, titanium dioxide, pyrophyllite clay, silica, attapulgite clay, kieselguhr, limestone, calcium carbonate, bentonite, calcium montmorillonite, cottonseed husks, wheat flour, soybean flour, pumice, wood flour, ground walnut shells, lignin and similar substances.
- a large number of surface-active substances can advantageously be used in both solid and liquid formulations, especially in those formulations which can be diluted with a carrier prior to use.
- Surfaceactive substances may be anionic, cationic, non-ionic or polymeric and they can be used as emulsifiers, wetting agents or suspending agents or for other purposes.
- Typical surface-active substances include, for example, salts of alkyl sulfates, such as diethanolammonium lauryl sulfate; salts of alkylarylsulfonates, such as calcium dodecylbenzenesulfonate; alkylphenol/alkylene oxide addition products, such as nonylphenol ethoxylate; alcohol/alkylene oxide addition products, such as tridecylalcohol ethoxylate; soaps, such as sodium stearate; salts of alkylnaphthalenesulfonates, such as sodium dibutylnaphthalenesulfonate; dialkyl esters of sulfosuccinate salts, such as sodium di(2- ethylhexyl)sulfosuccinate; sorbitol esters, such as sorbitol oleate; quaternary amines, such as lauryltrimethylammonium chloride, polyethylene glycol esters of
- Further adjuvants that can be used in pesticidal formulations include crystallisation inhibitors, viscosity modifiers, suspending agents, dyes, anti-oxidants, foaming agents, light absorbers, mixing auxiliaries, antifoams, complexing agents, neutralising or pH-modifying substances and buffers, corrosion inhibitors, fragrances, wetting agents, take-up enhancers, micronutrients, plasticisers, glidants, lubricants, dispersants, thickeners, antifreezes, microbicides, and liquid and solid fertilisers.
- compositions used according to the invention can include an additive comprising an oil of vegetable or animal origin, a mineral oil, alkyl esters of such oils or mixtures of such oils and oil derivatives.
- the amount of oil additive in the composition according to the invention is generally from 0.01 to 10 %, based on the active ingredient to be applied.
- the oil additive can be added to a spray tank in the desired concentration after a spray mixture has been prepared.
- Preferred oil additives comprise mineral oils or an oil of vegetable origin, for example rapeseed oil, olive oil or sunflower oil, emulsified vegetable oil, alkyl esters of oils of vegetable origin, for example the methyl derivatives, or an oil of animal origin, such as fish oil or beef tallow.
- Preferred oil additives comprise alkyl esters of C8-C22 fatty acids, especially the methyl derivatives of C12-C18 fatty acids, for example the methyl esters of lauric acid, palmitic acid and oleic acid (methyl laurate, methyl palmitate and methyl oleate, respectively).
- Many oil derivatives are known from the Compendium of Herbicide Adjuvants, 10 th Edition, Southern Illinois University, 2010.
- compositions generally comprise from 0.1 to 99 % by weight, especially from 0.1 to 95 % by weight, of the compound of the present invention and from 1 to 99.9 % by weight of a formulation adjuvant which preferably includes from 0 to 25 % by weight of a surface-active substance.
- a formulation adjuvant which preferably includes from 0 to 25 % by weight of a surface-active substance.
- the spray volume of a composition comprising the compound of formula (I) may be applied from 1 to 2000 L/ha, preferably from 10 to 1000 L/ha, and more preferably from 20 to 200 L/ha. In a particular embodiment, the spray volume of a composition comprising the compound of formula (I) may be applied from 100 to 200 L/ha for ground application, or from 20 to 40 L/ha for aerial application.
- Preferred formulations can have the following compositions (weight %): Emulsifiable concentrates: active ingredient: 1 to 95 %, preferably 60 to 90 % surface-active agent: 1 to 30 %, preferably 5 to 20 % liquid carrier: 1 to 80 %, preferably 1 to 35 %
- Dusts active ingredient: 0.1 to 10 %, preferably 0.1 to 5 % solid carrier: 99.9 to 90 %, preferably 99.9 to 99 %
- Suspension concentrates active ingredient: 5 to 75 %, preferably 10 to 50 % water: 94 to 24 %, preferably 88 to 30 % surface-active agent: 1 to 40 %, preferably 2 to 30 %
- Wettable powders active ingredient: 0.5 to 90 %, preferably 1 to 80 % surface-active agent: 0.5 to 20 %, preferably 1 to 15 % solid carrier: 5 to 95 %, preferably 15 to 90 %
- Granules active ingredient: 0.1 to 30 %, preferably 0.1 to 15 % solid carrier: 99.5 to 70 %, preferably 97 to 85 %
- the combination is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording wettable powders that can be diluted with water to give suspensions of the desired concentration.
- the combination is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording powders that can be used directly for seed treatment.
- Emulsions of any required dilution which can be used in plant protection, can be obtained from this concentrate by dilution with water. Ready-for-use dusts are obtained by mixing the combination with the carrier and grinding the mixture in a suitable mill. Such powders can also be used for dry dressings for seed.
- the combination is mixed and ground with the adjuvants, and the mixture is moistened with water.
- the mixture is extruded and then dried in a stream of air.
- the finely ground combination is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
- the finely ground combination is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
- 28 parts of the compound of the invention are mixed with 2 parts of an aromatic solvent and 7 parts of toluene diisocyanate/polymethylene-polyphenylisocyanate-mixture (8:1).
- This mixture is emulsified in a mixture of 1 .2 parts of polyvinylalcohol, 0.05 parts of a defoamer and 51.6 parts of water until the desired particle size is achieved.
- a mixture of 2.8 parts 1 ,6-diaminohexane in 5.3 parts of water is added.
- the mixture is agitated until the polymerization reaction is completed.
- the obtained capsule suspension is stabilized by adding 0.25 parts of a thickener and 3 parts of a dispersing agent.
- the capsule suspension formulation contains 28% of the active ingredient.
- the medium capsule diameter is 8-15 microns.
- the resulting formulation is applied to seeds as an aqueous suspension in an apparatus suitable for that purpose.
- the amount of a compound according to the invention to be applied will depend on various factors, such as the subject of the treatment, such as, for example plants, soil or seeds; the type of treatment, such as, for example spraying, dusting or seed dressing; the purpose of the treatment, such as, for example prophylactic or therapeutic; or the application time.
- the compound of formula (I) (active ingredient) can be applied at a rate of 5 to 500 g active ingredient (a.i.)/ha, preferably at a rate of 5 to 200 g a.i./ha, and more preferably at a rate of 10 to 100 g a.i./ha, such as for example at a rate of 30, 40, 50 or 75 g a.i./ha.
- composition according to the invention When the composition according to the invention is used for treating seed, rates of 0.001 to 100 g of a compound of formula (I) per kg of seed, preferably from 0.001 to 50 g per kg of seed, are generally sufficient, and more preferably from 0.01 to 10 g per kg of seed, are generally sufficient.
- compositions and formulations of the present invention can include the compound of formula (I) as the sole active ingredient in the compositions or formulations, or can also be used in combination with other active ingredients, e.g. other fungicides, and/or insecticides, and/or acaricides, and/or nematocides, and/or molluscicides, and/or biologicals, and/or plant growth regulators.
- active ingredients e.g. other fungicides, and/or insecticides, and/or acaricides, and/or nematocides, and/or molluscicides, and/or biologicals, and/or plant growth regulators.
- active ingredients include other fungicides, and/or insecticides, and/or acaricides, and/or nematocides, and/or molluscicides, and/or biologicals, and/or plant growth regulators.
- the compound of the present invention may be used in combination with a further fungicide, as detailed in WO 2008/131901.
- the compound of formula (I) according to the present invention may be used in combination with a strobilurin fungicide (for example, azoxystrobin, picoxystrobin, trifloxystrobin), a triazole fungicide (for example, cyproconazole, difenoconazole, propiconazole, prothioconazole, tebuconazole, mefentrifluconazole), a multi-site fungicide (for example chlorothalonil, copper oxychloride, mancozeb, fluazinam), and/or an SDHI fungicide (e.g. bixafen, fluxapyroxad, pydiflumetofen, inpyrfluxam, izopyrazam, fluxapyroxad).
- a strobilurin fungicide for example, azoxystrobin
- locus means fields in or on which plants are growing, or where seeds of cultivated plants are sown, or where seed will be placed into the soil. It includes soil, seeds, and seedlings, as well as established vegetation.
- plants refers to all physical parts of a plant, including seeds, seedlings, saplings, roots, tubers, stems, stalks, foliage, and fruits.
- plant propagation material is understood to denote all the generative parts of the plant, such as seeds, which can be used for the multiplication of the latter including vegetative plant material such as cuttings.
- vegetative plant material such as cuttings.
- seeds in the strict sense
- roots in the strict sense
- fruits tubers
- bulbs bulbs
- rhizomes parts of plants.
- Germinated plants and young plants which are to be transplanted after germination or after emergence from the soil, may also be mentioned. These young plants may be protected before transplantation by a total or partial treatment by immersion.
- the plant propagation material can be treated with the compound of formula (I) or the composition of the invention before the material is sown or planted.
- the plant propagation material may be treated with the compound of formula (I) or the composition of the invention during sowing or planting. Additionally, the compound of formula (I) or the composition of the invention may be applied to the previously treated propagation material before or during its planting. The compound of formula (I) or the composition of the invention may be applied during the sowing of the seed. The compound of formula (I) or the composition may also be used to plant propagation material derived from plants grown in a green house and/or during transplantation. More preferably the plant propagation material is plant seeds.
- the seed treatment can occur to an unsown seed, and the term "unsown seed” is meant to include seed at any period between the harvest of the seed and the sowing of the seed in the ground for the purpose of germination and growth of the plant.
- Treatment to an unsown seed is not meant to include those practices in which the compound of formula (I) or the composition of the invention is applied to the soil but would include any application practice that would target the seed during the sowing/planting process.
- the treated plant propagation material of the present invention can be treated in the same manner as conventional plant propagation material.
- the treated propagation material can be stored, handled, sown and tilled in the same manner as any other pesticide treated material.
- Another object of the present invention relates to a use of the compound of formula (I) as defined in the present invention or the composition comprising a compound of formula (I) as defined in the present invention, to control Diaporthe species in soybean.
- Diaporthe species are phytophathogenic diseases as described in the present invention, and can more preferably comprise at least one of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
- the preferred species is Diaporthe miriciae.
- the Diaporthe species can comprise at least two of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
- the two preferred species are Diaporthe miriciae and Diaporthe longicolla.
- the Diaporthe species can comprise at least three of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
- the three preferred species are Diaporthe miriciae, Diaporthe longicolla, and Diaporthe masirevicii.
- the Diaporthe species can comprise at least four of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
- the four preferred species are Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
- the Diaporthe species can comprise at least five of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
- the five preferred species are Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, Diaporthe pyracanthae, and Diaporthe sojae.
- the Diaporthe species can comprise Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
- the Diaporthe species can comprise at least one of the species selected from Diaporthe miriciae, Diaporthe sojae, and Diaporthe macadamiae. More preferably, the Diaporthe species can comprise at least two of the species selected from Diaporthe miriciae, Diaporthe sojae, and Diaporthe macadamiae. Even more preferably, the Diaporthe species can comprise Diaporthe miriciae, Diaporthe sojae, and Diaporthe macadamiae.
- FIGURE 1 shows the EC50 in log scale (mg/L of active ingredient), wherein AI1 is the compound of formula (I) according to the present invention, and AI2 to AI5 are comparative active ingredients.
- EC50 is defined as the fungicide dose that inhibits growth by 50% when compared with a non-modified control.
- the compounds AI1 to AI5 belongs to the succinate dehydrogenase inhibitors (SDHI).
- AI1 refers benzovindiflupyr as active ingredient, commercialized by Syngenta AG under the name Elatus® Plus which is an EC100 formulation (emulsifiable concentrate with 100 g of active ingredient per liter);
- AI2 refers to izopyrazam as active ingredient, commercialized by Adama Ltd under the name Reflect® which is an EC125 formulation (emulsifiable concentrate with 125 g of active ingredient per liter);
- AI3 refers to bixafen as active ingredient, commercialized by Bayer CropScience AG under the name Thore® which is an EC125 formulation (emulsifiable concentrate with 125 g of active ingredient per liter);
- AI4 refers to Impyrfluxam as active ingredient, commercialized by Sumitomo Chemical under the name Excalia® which is a SC313 formulation (suspension concentrate with 313 g of active ingredient per liter);
- AI5 refers to fluxapyroxad as active ingredient, commercialized by BASF SE under the name Sercadis® which is a SC300 formulation (suspension concentrate with 300 g of active ingredient per liter.
- the isolates comprised at least Diaporthe miriciae, more particularly comprised at least Diaporthe miriciae and Diaporthe longicolla, more particularly comprised at least Diaporthe miriciae, Diaporthe longicolla and Diaporthe masirevicii, more particularly comprised at least Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae, more particularly comprised at least Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, Diaporthe pyracanthae, and Diaporthe sojae, and even more particularly comprised Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, Diaporthe pyracanthae, Diaporthe sojae, and even more particularly comprised Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, Diaporthe pyracanth
- Mycelial discs with 4-mm diameter from the edge of the colonies were transferred to Petri dishes containing fungicide-modified PDA media. Petri dishes with PDA without fungicide treatments were used as control. Serial dilutions from 0.01 , 0.05, 0.01 , 0.1 , 1 .0, 10.0 and 100 mg.L’ 1 of each Al (AI1 to AI5) were diluted in sterile distilled water and homogenized in the PDA culture medium.
- the isolates comprised at least Diaporthe miriciae, more particularly comprised at least Diaporthe miriciae and Diaporthe longicolla, more particularly comprised at least Diaporthe miriciae, Diaporthe longicolla and Diaporthe masirevicii, more particularly comprised at least Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae, more particularly comprised at least Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, Diaporthe pyracanthae, and Diaporthe sojae, and even more particularly comprised Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, Diaporthe pyracanthae, Diaporthe sojae, and even more particularly comprised Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, Diaporthe pyracanth
- ITS-rDNA internal transcribed spacer of the ribosomal DNA
- TEZ3 histone 3
- CHS1 chitin synthase
- GS glutamine synthetase
- TEF1 translation elongation factor 1
- ACT actin
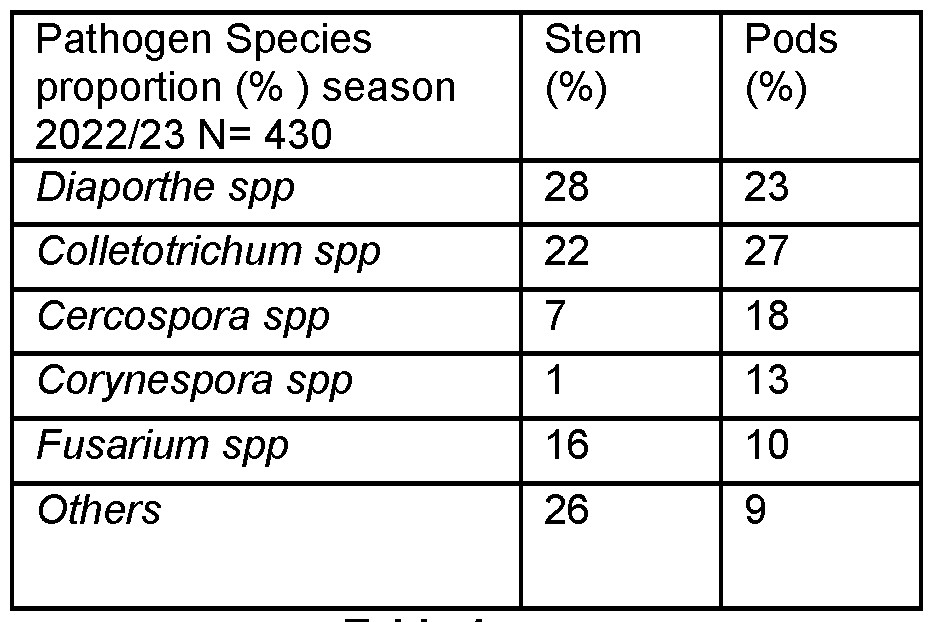
- Symptomatic soybean with ‘pod anomaly’ were sampled and isolations made from 254 pods and 176 stems from the 2022/2023 cropping season demonstrate a complex of fungal pathogens involved in the pods and are described below in Table 4.
- Table 5 below describes a further analysis of the data from Table 4 and from 47 isolates of Colletotrichum spp, two main species are indicated, Colletotrichum clivicola and Colletotrichum truncatum.
- Table 5 Table 6 below describes a further analysis of the data from Table 4 and from 66 isolates of Diaporthe spp a number of species are indicated, Diaporthe longicolla, Diaporthe Ueckerae, Diaporthe macadamiae and Diaporthe pyracanthae. These results confirm that the species responsible for stem canker are different from those associated with pod anomaly.
- Table 7 below describes a further analysis of the data from Table 4 and from 38 isolates of Fusarium spp a number of species are indicated, F. oxysporum spp complex, F. fujikuroi spp complex and F. incarnatum-equiseti spp complex.
- Symptomatic soybean with ‘pod anomaly’ were sampled and isolations made from 80 pods from the 2023/2024 cropping season demonstrate a complex of fungal pathogens involved in the pods and are described below in Table 8.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Wood Science & Technology (AREA)
- Environmental Sciences (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Engineering & Computer Science (AREA)
- Zoology (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Dentistry (AREA)
- Agronomy & Crop Science (AREA)
- Microbiology (AREA)
- Mycology (AREA)
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
The present invention relates to the use of a compound of formula (I), or a composition comprising a compound of formula (I), to control pod anomaly in soybean.
Description
METHOD OF PATHOGEN CONTROL IN SOYBEAN
The present invention relates to a method of pathogen control in soybean crops. In particular, it relates to the use of a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, and to a method of using of said compound or composition.
WO 2007/048556 discloses that certain heterocyclic amide derivatives have microbicidal activity, such as fungicidal activity. In particular, a compound of formula (I), 3-difluoromethyl-1-methyl-1 H-pyrazole-4- carboxylic acid(9-dichloromethylene-1 ,2,3,4-tetrahydro-1 ,4-methano-naphthalen-5-yl)-amide is disclosed:
The compound of formula (I) refers to benzovindiflupyr as active ingredient, with the following CAS number: 1072957-71-1.
In recent years, in particular since the 2019/2020 harvest new adverse effects have been reported in soybean pods in Brazil. These manifest in the abortion of pods and/or seeds at the stage of grain filling (typically from stage R5 of the soybean growth stages). In some instances pods are aborted from the plant, in others pods remain connected to the plants but the grain is unable to complete its development. As reported in the magazine “Cultivate Great Crops” (Grandes Culturas Cultivar October 2022 Issue 281 pp26-28, ISSN 1716-358x) and reported by Embrapa Soja in 2022, the problem occurred from the R5.4 development stage (51 % to 75% of grain formation) causing a 20% reduction in soybean productivity and a 30 % reduction in grain quality. Symptoms appeared as rot at the junction of the pods, evolving into a partial or total necrosis of the pod, and causing rot, opening and sometimes abortion of the pods. Seeds from the pods were also compromised and appeared wrinkled and differed in colour in comparison with normal (healthy) seeds (for example compromised seeds appear either darker than or paler than typically healthy seeds).
Given that pod anamoly represents a new disease of Soybean, very little is known with respect to its cause and the information is based primarily on symptomology. It has been postulated that the cause may be abiotic (e.g environmental), biotic (some form of phytopathogen) or even from a confluence of both. It had been proposed that anthracnose (a disease caused by the fungus Colleotricum truncatum) may have been linked to pod anomaly, however this is not the whole story as infection with this opportunistic pathogen does not result in green pods falling from the soy plants.
The present invention stems from the finding that several different phytopathogenic fungi are present in the tissue from soybean plants affected by pod anomaly, as shown herein in the Examples. The combination of these specific fungi is unexpected. It has now surprisingly been found that the compound of formula (I) or a composition comprising a compound of formula (I) is highly effective at combating, preventing or controlling pod anomaly in soybean, and is surprisingly more effective against this specific combination than other SDHI fungicides.
Accordingly, the present invention provides the use of a compound of formula (I)
or a composition comprising a compound of formula (I), to control pod anomaly in soybean.
In a second aspect of the invention there is provided the use of a compound of formula (I)
or a composition comprising a compound of formula (I), to control seed decay in post harvest soybean.
In a third aspect there is provided, a method of combating, preventing or controlling pod anomaly in soybean, which comprises applying to the crop of soybean plants, the locus thereof, or propagation material thereof, a compound of formula (I) or a composition comprising a compound of formula (I).
In a fourth aspect there is provided, a method of combating, preventing or controlling seed decay in post harvest soybean, which comprises applying to the crop of soybean plants, the locus thereof, or propagation material thereof, a compound of formula (I) or a composition comprising a compound of formula (I).
As used herein the term ’’combating”, “preventing” or “controlling”, and its inflections, within the context of the present invention, means reducing any undesired effect, such as pathogenic and more particularly phytopathogenic, especially fungi such as (but not limited to) Cercospora spp., Colletotrichum spp., Corynespora spp. or Diaporthe spp , infestation or attack of, and pathogenic damage to a plant or to a plant derived product to such a level that an improvement is demonstrated.
As used herein the term ‘pod anomaly’ refers to a phenotypic pathology of soybean pods, wherein the pod exhibits at least partial necrosis and/or seeds from affected pods develop abnormally. Pod anomaly has also been associated with the breakage of stems/stem blight, wherein black or brown streaks have been observed externally on stems and the inner marrow of stems exhibits necrosis. Where these symptoms also occur, the disease pathology is referred to herein as Soybean anomaly.
The compound of formula (I) can occur in two different stereoisomers, which are described as the single enantiomers of formulae (l)i and (l)N:
The present invention covers all such stereoisomers and mixtures thereof in any ratio. According to the invention “racemic compound of formula (I)” means a racemic mixture of compounds of formula (l)i and (l)u.
The compound of formula (I) may be prepared as described in WO 2007/048556.
The compound of formula (I) can be applied in a fungicidally effective amount. As used herein, the term "effective amount" refers to the amount of the compound, which, upon single or multiple applications provides the desired effect. An effective amount is readily determined by the skilled person in the art, by the use of known techniques and by observing results obtained under analogous circumstances. In determining the effective amount, a number of factors are considered including, but not limited to: the type of plant or derived product to be applied; the pathogen to be controlled & its lifecycle; the particular compound applied; the type of application; and other relevant circumstances.
The compound of formula (I) according to the invention is suitable for use on any soybean plant, including those that have been genetically modified to be resistant to active ingredients such as herbicides, or to produce biologically active compounds that control infestation by plant pests.
In a further preferred embodiment, transgenic plants and plant cultivars obtained by genetic engineering methods, if appropriate in combination with conventional methods (Genetically Modified Organisms), and parts thereof, are treated. Particularly preferably, plants of the plant cultivars which are in each case commercially available or in use are treated according to the invention. Plant cultivars are understood as meaning plants having novel properties ("traits") which have been obtained by conventional breeding, by mutagenesis or by recombinant DNA techniques. These can be cultivars, bio- or genotypes.
The preferred transgenic plants or plant cultivars (obtained by genetic engineering) which are to be treated according to the invention include all plants which, by virtue of the genetic modification, received genetic material which imparts particularly advantageous, useful traits to these plants. Examples of such traits are better plant growth, increased tolerance to high or low temperatures, increased tolerance to drought or to water or soil salt content, increased flowering performance, easier harvesting, accelerated maturation, higher harvest yields, higher quality and/or a higher nutritional value of the harvested products, better storage stability and/or processability of the harvested products. Preferably, the transgenic plants or plant cultivars are selected from Bt soybean plants.
Further and particularly emphasized examples of such traits are a better defence of the plants against animal and microbial pests, such as against insects, mites, phytopathogenic fungi, bacteria and/or viruses, and also increased tolerance of the plants to certain herbicidally active compounds.
Traits that are emphasized in particular are the increased defence of the plants against insects, arachnids, nematodes and slugs and snails by virtue of toxins formed in the plants, in particular those formed in the plants by the genetic material from Bacillus thuringiensis (for example by the genes CrylA(a), CrylA(b), CrylA(c), CryllA, CrylllA, CrylllB2, Cry9c, Cry2Ab, Cry3Bb and CrylF and also combinations thereof) (referred to herein as "Bt plants"). Traits that are also particularly emphasized are the increased defence of the plants against fungi, bacteria and viruses by systemic acquired resistance (SAR), systemin, phytoalexins, elicitors and resistance genes and correspondingly expressed proteins and toxins.
Traits that are furthermore particularly emphasized are the increased tolerance of the plants to certain herbicidally active compounds, for example imidazolinones, sulphonylureas, glyphosate or phosphinotricin (for example the "PAT" gene). The genes which impart the desired traits in question can also be present in combination with one another in the transgenic plants.
Examples of "Bt plants" are soybean varieties which are sold under the trade names YIELD GARD® or INTACTA®.
Examples of herbicide-tolerant plants which may be mentioned are soybean varieties which are sold under the trade names Roundup Ready® (tolerance to glyphosate), Liberty Link® (tolerance to phosphinotricin), IMI® (tolerance to imidazolinones) and STS® (tolerance to sulphonylureas).
Herbicide-resistant plants (plants bred in a conventional manner for herbicide tolerance) which may be mentioned include the varieties sold under the name Clearfield® (for example maize).
Of particular interest are soybean plants carrying traits conferring resistance to 2.4D (e.g. Enlist®), glyphosate (e.g. Roundup Ready®, Roundup Ready 2 Yield®), sulfonylurea (e.g. STS®), glufosinate (e.g. Liberty Link®, Ignite®), Dicamba (Monsanto) HPPD tolerance (e.g. isoxaflutole herbicide) (Bayer CropScience, Syngenta). Double or triple stack in soybean plants of any of the traits described here are also of interest, including glyphosate and sulfonyl-urea tolerance (e.g. Optimum GAT®, plants stacked with STS® and Roundup Ready® or Roundup Ready 2 Yield®), dicamba and glyphosate tolerance (Monsanto). Soybean Cyst Nematode resistance soybean (SCN® - Syngenta) and soybean with Aphid resistant trait (AMT® - Syngenta) are also of interest.
These statements also apply to plant cultivars having these genetic traits or genetic traits still to be developed, which plant cultivars will be developed and/or marketed in the future.
The genetic modified crops can be described in the following website https://www.isaaa.org/gmapprovaldatabase, incorporated by reference therein, and more particularly can be selected from:
In a preferred embodiment of the invention, there is provided, the use of a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises symptoms of pod decay, abnormal seed phenotype and/or stem blight. In another preferred embodiment the pod anomaly manifests at the R4 or R5 stage of the soybean plants reproductive cycle. Preferably the pod anomaly manifests at the R5 stage of the soybean plants reproductive cycle.
In another preferred embodiment the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp. and Diaporthe spp. More preferably, the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp. Even more preferably, the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp., wherein the Diaporthe species comprise at least Diaporthe miriciae and Diaporthe longicolla, and Fusarium spp. Even more preferably still, the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., wherein the Colletotrichum species comprise at least Colletotrichum cliviicola and Colletotrichum truncatum, Corynespora spp., Diaporthe spp., wherein the Diaporthe species comprise at least Diaporthe miriciae and Diaporthe longicolla, and Fusarium spp. Yet even more preferably still, the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., wherein the Colletotrichum species comprise at least Colletotrichum cliviicola and Colletotrichum truncatum, Corynespora spp., wherein the Corynespora spp. species comprise at least Corynespora cassiicola., Diaporthe spp., wherein the Diaporthe species comprise at least Diaporthe miriciae and Diaporthe longicolla, and Fusarium spp. Yet even more further preferably still, the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., wherein the Colletotrichum species comprise at least Colletotrichum cliviicola and Colletotrichum truncatum, Corynespora spp., wherein the Corynespora spp. species comprise at least Corynespora cassiicola., Diaporthe spp., wherein the Diaporthe species comprise at least Diaporthe miriciae and Diaporthe longicolla, and Fusarium spp., wherein the Fusarium spp. species comprise at least Fusarium fujikuroi and Fusarium incarnatum.
In one embodiment, there is provided the use of a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp. and Diaporthe spp. Preferably, the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp.
In a preferred embodiment, there is provided the use of a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp. and Diaporthe spp. wherein the Diaporthe species comprise at least Diaporthe miriciae and Diaporthe longicolla.
In another preferred embodiment, there is provided the use of a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp. and Diaporthe spp. wherein the Colletotrichum species comprise at least Colletotrichum cliviicola and Colletotrichum truncatum.
In another preferred embodiment, there is provided the use of a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp. and Diaporthe spp. wherein the Corynespora spp. species comprise at least Corynespora cassiicola.
In another preferred embodiment, there is provided the use of a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp. wherein the Fusarium spp. species comprise at least Fusarium fujikuroi and Fusarium incarnatum.
Preferably, there is provided the use of a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp. wherein the Diaporthe species comprise at least Diaporthe miriciae and Diaporthe longicolla (more preferably at least Diaporthe longicolla, Diaporthe miriciae, Diaporthe macadamiae and Diaporthe pyracanthae) and the Fusarium spp. species comprise at least Fusarium fujikuroi and Fusarium incarnatum.
Even more preferably, there is provided the use of a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp. wherein the Diaporthe species comprise at least Diaporthe miriciae and Diaporthe longicolla (more preferably at least Diaporthe longicolla, Diaporthe miriciae, Diaporthe macadamiae and Diaporthe pyracanthae), the Colletotrichum species comprise at least Colletotrichum cliviicola and Colletotrichum truncatum, and the Fusarium spp. species comprise at least Fusarium fujikuroi and Fusarium incarnatum.
Even more preferably still, there is provided the use of a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp. wherein the Diaporthe species comprise at least Diaporthe miriciae and Diaporthe longicolla (more preferably at least Diaporthe longicolla, Diaporthe miriciae, Diaporthe macadamiae and Diaporthe pyracanthae), the Colletotrichum species comprise at least Colletotrichum cliviicola and Colletotrichum truncatum, the Corynespora spp. species comprise at least Corynespora cassiicola and the Fusarium spp. species comprise at least Fusarium fujikuroi and Fusarium incarnatum.
In the present invention, Diaporthe species, also known as Phomopsis species, are phytopathogenic fungi which can more preferably comprise at least one of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe
pyracanthae. Diaporthe miriciae is also known as Diaporthe ueckeri or Diaporthe ueckerae. The preferred species is Diaporthe miriciae.
In a preferred embodiment, the Diaporthe species can comprise at least two of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae. The two preferred species are Diaporthe miriciae and Diaporthe longicolla.
In a further preferred embodiment, the Diaporthe species can comprise at least three of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae. The three preferred species are Diaporthe miriciae, Diaporthe longicolla, and Diaporthe masirevicii.
In a preferred embodiment, the Diaporthe species can comprise at least four of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae. The four preferred species are Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
In a preferred embodiment, the Diaporthe species can comprise at least five of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae. The five preferred species are Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, Diaporthe pyracanthae, and Diaporthe sojae.
In an even more preferred embodiment, the Diaporthe species can comprise Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
In another particular embodiment, the Diaporthe species can comprise at least one of the species selected from Diaporthe miriciae, Diaporthe sojae, and Diaporthe macadamiae. More preferably, the Diaporthe species can comprise at least two of the species selected from Diaporthe miriciae, Diaporthe sojae, and Diaporthe macadamiae. Even more preferably, the Diaporthe species can comprise Diaporthe miriciae, Diaporthe sojae, and Diaporthe macadamiae.
In one embodiment, there is provided the use of a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp. and wherein the soybean is a transgenic soybean plant (preferably a transgenic Bt soybean plant, even more preferably, the transgenic soybean plant comprises an event selected from the list consisting of event MON87701 , event MON87751 , event DAS- 81419-2, event DAS-44406-6/pDAB8264.44.06.1 , event DAS-14536-7/pDAB8291 .45.36.2, event DAS- 68416-4, event DP-305423-1 , event DP-356043-5, event FG72, event LL27, event LL55, event EE- GM3/FG72 (optionally stacked with event EE-GM1/LL27 or event EE-GM2/LL55), event MON87705,
event MON87708, event MON87712, event MON87754, event MON87769, event MON89788, event SYHT0H2/SYN-000H2-5, event DAS-21606-3, event 8264.44.06.1 , event pDAB8291 .45.36.2, event PDAB8264.42.32.1 , event A2704-12, event A5547-127, event BPS-CV127- 91 , event GU262, event BPS-CV127-9 and event GMB151.)
In a preferred embodiment of the invention there is provided, the use of a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of 1 to 15 % Corynespora spp, from 10 to 70% Diaporthe spp, from 5 to 35% Cercospora spp and from 10 to 60% Colletotrichum spp, (the skilled person would appreciate the total amount of phytopathogenic fungi is 100% and that the proportion of percentage values of each species can vary in the pod anomaly disease complex). Preferably, the pod anomaly comprises phytopathogenic fungi selected from the group consisting of 1 to 15 % Corynespora spp, from 10 to 70% Diaporthe spp, from 5 to 35% Cercospora spp., from 10 to 60% Colletotrichum spp, and from 5 to 20% Fusarium spp. More preferably, the pod anomaly comprises phytopathogenic fungi selected from the group consisting of 1 to 5 % Corynespora spp, from 20 to 60% Diaporthe spp, from 5 to 20% Cercospora spp., from 10 to 30% Colletotrichum spp, and from 10 to 20% Fusarium spp.
In another embodiment of the invention there is provided, the use of a compound of formula (I) or a composition comprising a compound of formula (I) to control seed decay in post harvest soybean.
Preferably, there is provided, the use of a compound of formula (I) or a composition comprising a compound of formula (I) to control seed decay in post harvest soybean, during the growing period of the soybean.
In another preferred embodiment, there is provided, the use of a compound of formula (I) or a composition comprising a compound of formula (I) to control seed decay in post harvest soybean, wherein the seed comprises phytopathogenic fungi selected from the group consisting of Colletotrichum spp. and Diaporthe spp.
In another preferred embodiment, there is provided, the use of a composition comprising a compound of formula (I) to control seed decay in post harvest soybean, wherein the composition comprising a compound of formula (I) further comprises at least one fungicidally active compound. More preferably the at least one fungicidally active compound is selected from the list consisting of azoxystrobin, cyproconazole, difenoconazole, mancozeb, propiconazole, proth ioconazole and pydiflumetofen.
In another preferred embodiment, there is provided, the use of a composition comprising a compound of formula (I) to control seed decay in post harvest soybean, wherein the composition comprising a compound of formula (I) further comprises two fungicidally active compounds (preferably the fungicidally active compounds are selected from the list consisting of azoxystrobin, cyproconazole, difenoconazole, mancozeb, propiconazole, prothioconazole and pydiflumetofen). More preferably, there is provided, the use of a composition comprising a compound of formula (I) to control seed decay in post harvest
soybean, wherein the composition comprises a compound of formula (I), prothioconazole and mancozeb.
Preferably, the use of the compound of formula (I) according to the invention or a composition thereof, is applied at a rate of from 5 to 200 g a.i./ha, preferably at a rate of from 10 to 100 g a.i./ha, more preferably at a rate of from 20 to 80 g a.i/ha, even more preferably from 25 to 75 g a.i/ha, even more preferably still from 50 to 75 g a.i/ha.
Preferably, there is provided the use of a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the composition comprising a compound of formula (I) further comprises at least one fungicidally active compound. More preferably the at least one fungicidally active compound is selected from the list consisting of azoxystrobin, cyproconazole, difenoconazole, mancozeb, propiconazole, prothioconazole and pydiflumetofen.
In one embodiment, where the composition comprises azoxystrobin, the azoxystrobin is applied at a rate of from 25 to 75 g a.i/ha.
In another embodiment, where the composition comprises cyproconazole, the cyproconazole is applied at a rate of from 25 to 75 g a.i/ha.
In another embodiment, where the composition comprises difenoconazole, the difenoconazole is applied at a rate of from 35 to 115 g a.i/ha.
In another embodiment, where the composition comprises mancozeb, the mancozeb is applied at a rate of from 500 to 2,000 g a.i/ha.
In another embodiment, where the composition comprises propiconazole, the propiconazole is applied at a rate of from 25 to 75 g a.i/ha.
In another embodiment, where the composition comprises prothioconazole, the prothioconazole is applied at a rate of from 50 to 150 g a.i/ha.
In another embodiment, where the composition comprises pydiflumetofen, the pydiflumetofen is applied at a rate of from 25 to 75 g a.i/ha.
In a preferred embodiment, there is provided the use of a compound of formula (I) or a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp. and wherein the composition comprising a compound of formula (I) further comprises at least one fungicidally active compound (preferably at least one fungicidally active compound is selected from the list consisting of azoxystrobin, cyproconazole, difenoconazole, mancozeb, propiconazole, prothioconazole and pydiflumetofen).
In another preferred embodiment, there is provided the use of a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp.,
Diaporthe spp. and Fusarium spp. and wherein the composition comprising a compound of formula (I) further comprises two fungicidally active compounds (preferably the fungicidally active compounds are selected from the list consisting of azoxystrobin, cyproconazole, difenoconazole, mancozeb, propiconazole, prothioconazole and pydiflumetofen).
In an even more preferred embodiment, there is provided the use of a composition comprising a compound of formula (I), to control pod anomaly in soybean, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp. and wherein the composition comprising a compound of formula (I) further comprises prothioconazole and mancozeb (preferably, the compound of formula (I) is applied at a rate of from 5 to 200 g a.i./ha, more preferably at a rate of from 10 to 100 g a.i./ha, even more preferably at a rate of from 20 to 80 g a.i/ha, even more preferably still from 25 to 75 g a.i/ha, yet even more preferably still from 50 to 75 g a.i/ha).
In another embodiment of the invention there is provided, the use of a compound of formula (I) or a composition comprising a compound of formula (I) to control seed decay in post harvest soybean, wherein the composition comprising a compound of formula (I) further comprises at least one fungicidally active compound (preferably at least one fungicidally active compound is selected from the list consisting of azoxystrobin, cyproconazole, difenoconazole, mancozeb, propiconazole, prothioconazole and pydiflumetofen).
The compound used according to the invention can be used in unmodified form, but it is generally formulated into compositions in various ways using formulation adjuvants, such as carriers, solvents and surface-active substances. The formulations can be in various physical forms, e.g. in the form of dusting powders, gels, wettable powders, water-dispersible granules, water-dispersible tablets, effervescent pellets, emulsifiable concentrates, microemulsifiable concentrates, oil-in-water emulsions, oil-flowables, aqueous dispersions, oily dispersions, suspo-emulsions, capsule suspensions, emulsifiable granules, soluble liquids, water-soluble concentrates (with water or a water-miscible organic solvent as carrier), impregnated polymer films or in other forms known e.g. from the Manual on Development and Use of FAO and WHO Specifications for Pesticides, United Nations, First Edition, Second Revision (2010). Such formulations can either be used directly or diluted prior to use. The dilutions can be made, for example, with water, liquid fertilisers, micronutrients, biological organisms, oil or solvents.
The formulations used according to the invention can be prepared e.g. by mixing the compound of formula (I) (‘active ingredient’) with the formulation adjuvants in order to obtain compositions in the form of finely divided solids, granules, solutions, dispersions or emulsions. The active ingredient can also be formulated with other adjuvants, such as finely divided solids, mineral oils, oils of vegetable or animal origin, modified oils of vegetable or animal origin, organic solvents, water, surface-active substances or combinations thereof.
The active ingredient can also be contained in very fine microcapsules. Microcapsules contain the active ingredient in a porous carrier. This enables the active ingredient to be released into the environment in
controlled amounts (e.g. slow-release). Microcapsules usually have a diameter of from 0.1 to 500 microns. They contain the active ingredient in an amount of about from 25 to 95 % by weight of the capsule weight. The active ingredient can be in the form of a monolithic solid, in the form of fine particles in solid or liquid dispersion or in the form of a suitable solution. The encapsulating membranes can comprise, for example, natural or synthetic rubbers, cellulose, styrene/butadiene copolymers, polyacrylonitrile, polyacrylate, polyesters, polyamides, polyureas, polyurethane or chemically modified polymers and starch xanthates or other polymers that are known to the person skilled in the art. Alternatively, very fine microcapsules can be formed in which the active ingredient is contained in the form of finely divided particles in a solid matrix of base substance, but the microcapsules are not themselves encapsulated.
The formulation adjuvants that are suitable for the preparation of the compositions according to the invention are known per se. As liquid carriers there may be used: water, toluene, xylene, petroleum ether, vegetable oils, acetone, methyl ethyl ketone, cyclohexanone, acid anhydrides, acetonitrile, acetophenone, amyl acetate, 2-butanone, butylene carbonate, chlorobenzene, cyclohexane, cyclohexanol, alkyl esters of acetic acid, diacetone alcohol, 1 ,2-dichloropropane, diethanolamine, p- diethylbenzene, diethylene glycol, diethylene glycol abietate, diethylene glycol butyl ether, diethylene glycol ethyl ether, diethylene glycol methyl ether, /V,/V-dimethylformamide, dimethyl sulfoxide, 1 ,4- dioxane, dipropylene glycol, dipropylene glycol methyl ether, dipropylene glycol dibenzoate, diproxitol, alkylpyrrolidone, ethyl acetate, 2-ethylhexanol, ethylene carbonate, 1 ,1 ,1 -trichloroethane, 2-heptanone, alpha-pinene, d-limonene, ethyl lactate, ethylene glycol, ethylene glycol butyl ether, ethylene glycol methyl ether, gamma-butyrolactone, glycerol, glycerol acetate, glycerol diacetate, glycerol triacetate, hexadecane, hexylene glycol, isoamyl acetate, isobornyl acetate, isooctane, isophorone, isopropylbenzene, isopropyl myristate, lactic acid, laurylamine, mesityl oxide, methoxypropanol, methyl isoamyl ketone, methyl isobutyl ketone, methyl laurate, methyl octanoate, methyl oleate, methylene chloride, m-xylene, n-hexane, n-octylamine, octadecanoic acid, octylamine acetate, oleic acid, oleylamine, o-xylene, phenol, polyethylene glycol, propionic acid, propyl lactate, propylene carbonate, propylene glycol, propylene glycol methyl ether, p-xylene, toluene, triethyl phosphate, triethylene glycol, xylenesulfonic acid, paraffin, mineral oil, trichloroethylene, perchloroethylene, ethyl acetate, amyl acetate, butyl acetate, propylene glycol methyl ether, diethylene glycol methyl ether, methanol, ethanol, isopropanol, and alcohols of higher molecular weight, such as amyl alcohol, tetrahydrofurfuryl alcohol, hexanol, octanol, ethylene glycol, propylene glycol, glycerol, A/-methyl-2-pyrrolidone and the like.
Suitable solid carriers are, for example, talc, titanium dioxide, pyrophyllite clay, silica, attapulgite clay, kieselguhr, limestone, calcium carbonate, bentonite, calcium montmorillonite, cottonseed husks, wheat flour, soybean flour, pumice, wood flour, ground walnut shells, lignin and similar substances.
A large number of surface-active substances can advantageously be used in both solid and liquid formulations, especially in those formulations which can be diluted with a carrier prior to use. Surfaceactive substances may be anionic, cationic, non-ionic or polymeric and they can be used as emulsifiers, wetting agents or suspending agents or for other purposes. Typical surface-active substances include, for example, salts of alkyl sulfates, such as diethanolammonium lauryl sulfate; salts of alkylarylsulfonates, such as calcium dodecylbenzenesulfonate; alkylphenol/alkylene oxide addition
products, such as nonylphenol ethoxylate; alcohol/alkylene oxide addition products, such as tridecylalcohol ethoxylate; soaps, such as sodium stearate; salts of alkylnaphthalenesulfonates, such as sodium dibutylnaphthalenesulfonate; dialkyl esters of sulfosuccinate salts, such as sodium di(2- ethylhexyl)sulfosuccinate; sorbitol esters, such as sorbitol oleate; quaternary amines, such as lauryltrimethylammonium chloride, polyethylene glycol esters of fatty acids, such as polyethylene glycol stearate; block copolymers of ethylene oxide and propylene oxide; and salts of mono- and dialkylphosphate esters; and also further substances described e.g. in McCutcheon's Detergents and Emulsifiers Annual, MC Publishing Corp., Ridgewood New Jersey (1981).
Further adjuvants that can be used in pesticidal formulations include crystallisation inhibitors, viscosity modifiers, suspending agents, dyes, anti-oxidants, foaming agents, light absorbers, mixing auxiliaries, antifoams, complexing agents, neutralising or pH-modifying substances and buffers, corrosion inhibitors, fragrances, wetting agents, take-up enhancers, micronutrients, plasticisers, glidants, lubricants, dispersants, thickeners, antifreezes, microbicides, and liquid and solid fertilisers.
The compositions used according to the invention can include an additive comprising an oil of vegetable or animal origin, a mineral oil, alkyl esters of such oils or mixtures of such oils and oil derivatives. The amount of oil additive in the composition according to the invention is generally from 0.01 to 10 %, based on the active ingredient to be applied. For example, the oil additive can be added to a spray tank in the desired concentration after a spray mixture has been prepared. Preferred oil additives comprise mineral oils or an oil of vegetable origin, for example rapeseed oil, olive oil or sunflower oil, emulsified vegetable oil, alkyl esters of oils of vegetable origin, for example the methyl derivatives, or an oil of animal origin, such as fish oil or beef tallow. Preferred oil additives comprise alkyl esters of C8-C22 fatty acids, especially the methyl derivatives of C12-C18 fatty acids, for example the methyl esters of lauric acid, palmitic acid and oleic acid (methyl laurate, methyl palmitate and methyl oleate, respectively). Many oil derivatives are known from the Compendium of Herbicide Adjuvants, 10th Edition, Southern Illinois University, 2010.
The compositions generally comprise from 0.1 to 99 % by weight, especially from 0.1 to 95 % by weight, of the compound of the present invention and from 1 to 99.9 % by weight of a formulation adjuvant which preferably includes from 0 to 25 % by weight of a surface-active substance. Whereas commercial products may preferably be formulated as concentrates, the end user will normally employ dilute formulations.
As a general guideline, the spray volume of a composition comprising the compound of formula (I) may be applied from 1 to 2000 L/ha, preferably from 10 to 1000 L/ha, and more preferably from 20 to 200 L/ha. In a particular embodiment, the spray volume of a composition comprising the compound of formula (I) may be applied from 100 to 200 L/ha for ground application, or from 20 to 40 L/ha for aerial application.
Preferred formulations can have the following compositions (weight %):
Emulsifiable concentrates: active ingredient: 1 to 95 %, preferably 60 to 90 % surface-active agent: 1 to 30 %, preferably 5 to 20 % liquid carrier: 1 to 80 %, preferably 1 to 35 %
Dusts: active ingredient: 0.1 to 10 %, preferably 0.1 to 5 % solid carrier: 99.9 to 90 %, preferably 99.9 to 99 %
Suspension concentrates: active ingredient: 5 to 75 %, preferably 10 to 50 % water: 94 to 24 %, preferably 88 to 30 % surface-active agent: 1 to 40 %, preferably 2 to 30 %
Wettable powders: active ingredient: 0.5 to 90 %, preferably 1 to 80 % surface-active agent: 0.5 to 20 %, preferably 1 to 15 % solid carrier: 5 to 95 %, preferably 15 to 90 %
Granules: active ingredient: 0.1 to 30 %, preferably 0.1 to 15 % solid carrier: 99.5 to 70 %, preferably 97 to 85 %
The combination is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording wettable powders that can be diluted with water to give suspensions of the desired concentration.
The combination is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording powders that can be used directly for seed treatment.
Emulsions of any required dilution, which can be used in plant protection, can be obtained from this concentrate by dilution with water.
Ready-for-use dusts are obtained by mixing the combination with the carrier and grinding the mixture in a suitable mill. Such powders can also be used for dry dressings for seed.
The combination is mixed and ground with the adjuvants, and the mixture is moistened with water.
The finely ground combination is uniformly applied, in a mixer, to the kaolin moistened with polyethylene glycol. Non-dusty coated granules are obtained in this manner.
Suspension concentrate
The finely ground combination is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water. Using such dilutions, living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
The finely ground combination is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water. Using such dilutions, living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
Slow Release Capsule Suspension
28 parts of the compound of the invention are mixed with 2 parts of an aromatic solvent and 7 parts of toluene diisocyanate/polymethylene-polyphenylisocyanate-mixture (8:1). This mixture is emulsified in a mixture of 1 .2 parts of polyvinylalcohol, 0.05 parts of a defoamer and 51.6 parts of water until the desired particle size is achieved. To this emulsion a mixture of 2.8 parts 1 ,6-diaminohexane in 5.3 parts of water is added. The mixture is agitated until the polymerization reaction is completed. The obtained capsule suspension is stabilized by adding 0.25 parts of a thickener and 3 parts of a dispersing agent. The capsule suspension formulation contains 28% of the active ingredient. The medium capsule diameter is 8-15 microns. The resulting formulation is applied to seeds as an aqueous suspension in an apparatus suitable for that purpose.
The amount of a compound according to the invention to be applied, will depend on various factors, such as the subject of the treatment, such as, for example plants, soil or seeds; the type of treatment,
such as, for example spraying, dusting or seed dressing; the purpose of the treatment, such as, for example prophylactic or therapeutic; or the application time.
The compound of formula (I) (active ingredient) can be applied at a rate of 5 to 500 g active ingredient (a.i.)/ha, preferably at a rate of 5 to 200 g a.i./ha, and more preferably at a rate of 10 to 100 g a.i./ha, such as for example at a rate of 30, 40, 50 or 75 g a.i./ha.
When the composition according to the invention is used for treating seed, rates of 0.001 to 100 g of a compound of formula (I) per kg of seed, preferably from 0.001 to 50 g per kg of seed, are generally sufficient, and more preferably from 0.01 to 10 g per kg of seed, are generally sufficient.
The compositions and formulations of the present invention can include the compound of formula (I) as the sole active ingredient in the compositions or formulations, or can also be used in combination with other active ingredients, e.g. other fungicides, and/or insecticides, and/or acaricides, and/or nematocides, and/or molluscicides, and/or biologicals, and/or plant growth regulators.
Normally, in the management of a crop a grower would use one or more other active ingredients in addition to the compound of the present invention. Examples of active ingredients include other fungicides, and/or insecticides, and/or acaricides, and/or nematocides, and/or molluscicides, and/or biologicals, and/or plant growth regulators.
For example, the compound of the present invention may be used in combination with a further fungicide, as detailed in WO 2008/131901. In particular, the compound of formula (I) according to the present invention may be used in combination with a strobilurin fungicide (for example, azoxystrobin, picoxystrobin, trifloxystrobin), a triazole fungicide (for example, cyproconazole, difenoconazole, propiconazole, prothioconazole, tebuconazole, mefentrifluconazole), a multi-site fungicide (for example chlorothalonil, copper oxychloride, mancozeb, fluazinam), and/or an SDHI fungicide (e.g. bixafen, fluxapyroxad, pydiflumetofen, inpyrfluxam, izopyrazam, fluxapyroxad).
In the present invention, the term “locus” as used herein means fields in or on which plants are growing, or where seeds of cultivated plants are sown, or where seed will be placed into the soil. It includes soil, seeds, and seedlings, as well as established vegetation.
The term “plants” refers to all physical parts of a plant, including seeds, seedlings, saplings, roots, tubers, stems, stalks, foliage, and fruits.
The term "plant propagation material" is understood to denote all the generative parts of the plant, such as seeds, which can be used for the multiplication of the latter including vegetative plant material such as cuttings. There may be mentioned, as plant propagation material, seeds (in the strict sense), roots, fruits, tubers, bulbs, rhizomes, parts of plants. Germinated plants and young plants which are to be transplanted after germination or after emergence from the soil, may also be mentioned. These young plants may be protected before transplantation by a total or partial treatment by immersion. The plant propagation material can be treated with the compound of formula (I) or the composition of the invention before the material is sown or planted. Alternatively, the plant propagation material may be treated with
the compound of formula (I) or the composition of the invention during sowing or planting. Additionally, the compound of formula (I) or the composition of the invention may be applied to the previously treated propagation material before or during its planting. The compound of formula (I) or the composition of the invention may be applied during the sowing of the seed. The compound of formula (I) or the composition may also be used to plant propagation material derived from plants grown in a green house and/or during transplantation. More preferably the plant propagation material is plant seeds. The seed treatment can occur to an unsown seed, and the term "unsown seed" is meant to include seed at any period between the harvest of the seed and the sowing of the seed in the ground for the purpose of germination and growth of the plant. Treatment to an unsown seed is not meant to include those practices in which the compound of formula (I) or the composition of the invention is applied to the soil but would include any application practice that would target the seed during the sowing/planting process. The treated plant propagation material of the present invention can be treated in the same manner as conventional plant propagation material. The treated propagation material can be stored, handled, sown and tilled in the same manner as any other pesticide treated material.
Another object of the present invention relates to a use of the compound of formula (I) as defined in the present invention or the composition comprising a compound of formula (I) as defined in the present invention, to control Diaporthe species in soybean.
Diaporthe species are phytophathogenic diseases as described in the present invention, and can more preferably comprise at least one of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae. The preferred species is Diaporthe miriciae.
In a preferred embodiment, the Diaporthe species can comprise at least two of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae. The two preferred species are Diaporthe miriciae and Diaporthe longicolla.
In a further preferred embodiment, the Diaporthe species can comprise at least three of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae. The three preferred species are Diaporthe miriciae, Diaporthe longicolla, and Diaporthe masirevicii.
In a preferred embodiment, the Diaporthe species can comprise at least four of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae. The four preferred species are Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
In a preferred embodiment, the Diaporthe species can comprise at least five of the species selected from Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae. The five preferred species are Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, Diaporthe pyracanthae, and Diaporthe sojae.
In an even more preferred embodiment, the Diaporthe species can comprise Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae.
In another particular embodiment, the Diaporthe species can comprise at least one of the species selected from Diaporthe miriciae, Diaporthe sojae, and Diaporthe macadamiae. More preferably, the Diaporthe species can comprise at least two of the species selected from Diaporthe miriciae, Diaporthe sojae, and Diaporthe macadamiae. Even more preferably, the Diaporthe species can comprise Diaporthe miriciae, Diaporthe sojae, and Diaporthe macadamiae.
Various aspects and embodiments of the present invention will now be illustrated in more detail by way of example and the following Figures in which:
FIGURE 1 shows the EC50 in log scale (mg/L of active ingredient), wherein AI1 is the compound of formula (I) according to the present invention, and AI2 to AI5 are comparative active ingredients.
According to the Fungicide Resistance Action Committee (FRAC), EC50 is defined as the fungicide dose that inhibits growth by 50% when compared with a non-modified control.
The compounds AI1 to AI5 belongs to the succinate dehydrogenase inhibitors (SDHI).
EXAMPLES
In Figure 1 :
AI1 refers benzovindiflupyr as active ingredient, commercialized by Syngenta AG under the name Elatus® Plus which is an EC100 formulation (emulsifiable concentrate with 100 g of active ingredient per liter);
AI2 refers to izopyrazam as active ingredient, commercialized by Adama Ltd under the name Reflect® which is an EC125 formulation (emulsifiable concentrate with 125 g of active ingredient per liter);
AI3 refers to bixafen as active ingredient, commercialized by Bayer CropScience AG under the name Thore® which is an EC125 formulation (emulsifiable concentrate with 125 g of active ingredient per liter);
AI4 refers to Impyrfluxam as active ingredient, commercialized by Sumitomo Chemical under the name Excalia® which is a SC313 formulation (suspension concentrate with 313 g of active ingredient per liter); and
AI5 refers to fluxapyroxad as active ingredient, commercialized by BASF SE under the name Sercadis® which is a SC300 formulation (suspension concentrate with 300 g of active ingredient per liter.
The preparation of the tests has been performed as follows:
Isolates of Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and/or Diaporthe pyracanthae, from soybean were collected at Mato Grosso (n = 3) and Mato Grosso do Sul (n = 1). More particularly, the isolates comprised at least Diaporthe miriciae, more particularly comprised at least Diaporthe miriciae and Diaporthe longicolla, more particularly comprised at least Diaporthe miriciae, Diaporthe longicolla and Diaporthe masirevicii, more particularly comprised at least Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae, more particularly comprised at least Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, Diaporthe pyracanthae, and Diaporthe sojae, and even more particularly comprised Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, Diaporthe pyracanthae, Diaporthe sojae, and Diaporthe macadamiae.
All the isolates collected in the cropping seasons of 2022 and were isolated from pods with symptoms of “pod anomaly”. Isolation was performed directly. Fragments of the fungus mycelium were removed from pods and were transferred to Petri dishes containing potato dextrose-agar (PDA) culture medium (4 g.L-1 potato extract, 20 g.L-1 dextrose, 15 g.L-1 agar, pH 5.6 ± 0.2). The isolates were cultivated in PDA culture medium at 22 °C under a photoperiod of 12 h/12 h for 7 days. Bipartite Petri dishes with 9 mm x 15 mm were used in this experiment. Each isolate was composed of 4 replicates. Mycelial discs with 4-mm diameter from the edge of the colonies were transferred to Petri dishes containing fungicide-modified PDA media. Petri dishes with PDA without fungicide treatments were used as control. Serial dilutions from 0.01 , 0.05, 0.01 , 0.1 , 1 .0, 10.0 and 100 mg.L’1 of each Al (AI1 to AI5) were diluted in sterile distilled water and homogenized in the PDA culture medium.
After, Petri dishes were incubated at 24 °C ± 2 °C and 12 h/12 h photoperiod for 5 days and the diameter of each colony was measured (orthogonal measurements) for each isolate to determine percentage of control by each fungicide compared to the growth on non-modified media following the methodology described by Ishii et al. 2009. The percentage control by the fungicide was calculated from the growth on modified media compared to growth on PDA (Mello et al., 2021). The EC50 graphical analysis were performed using TIBCO® Spotfire® Analyst software (version 10.3).
Table 1 The results in Table 1 clearly show that only a very low amount of the compound of formula (I) (0.38 mg/L) is surprisingly needed to control 50% of the pests, in comparison with the other SDHI active ingredients AI2 to AI5.
Similar tests were performed in 2023 in following the same protocol as described above, knowing that isolates of Diaporthe miriciae, Diaporthe sojae, Diaporthe macadamiae, Diaporthe longicolla, Diaporthe masirevicii, and/or Diaporthe pyracanthae, from soybean were collected at Rondonia (n = 3) and Mato Grosso (n = 16). More particularly, the isolates comprised at least Diaporthe miriciae, more particularly comprised at least Diaporthe miriciae and Diaporthe longicolla, more particularly comprised at least Diaporthe miriciae, Diaporthe longicolla and Diaporthe masirevicii, more particularly comprised at least Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, and Diaporthe pyracanthae, more particularly comprised at least Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, Diaporthe pyracanthae, and Diaporthe sojae, and even more particularly comprised Diaporthe miriciae, Diaporthe longicolla, Diaporthe masirevicii, Diaporthe pyracanthae, Diaporthe sojae, and Diaporthe macadamiae.
Table 2
The results in Table 2 clearly show that only a very low amount of the compound of formula (I) (1.8 mg/L) is surprisingly needed to control 50% of the pests, in comparison with the other SDHI active ingredients AI3 to AI5.
Further isolates were collected in the cropping seasons 2021/2022, 2022/2023 and 2023/2024 from pods with symptoms of “pod anomaly” across Brazil. The samples were morphologically diagnosed based on observation of fungal structures. Isolates were recovered from symptomatic soybean tissue by cutting a small piece of the lesioned plant tissue. This material was introduced into PDA media. Roughly 14 days after the isolation, fungi colonies were assessed according to their morphology. All the isolates were identified using molecular tools.
Aiming to investigate the relationship of these fungi with the pod anomaly symptoms, Koch's Postulate were carried out. After 21 days, under controlled conditions, the inoculated plants showed the first symptoms. Confirmation of pathogenicity occurred with the re-isolation of fungi recovered from soybean plants inoculated with the pathogens.
With the objective of demonstrating the effect of a compound of formula (I) to control Diaporthe spp. and Colletotrichum spp., the mycelial growth inhibition test was performed. To test the in vitro effectiveness of carboxamides, isolates were randomly selected. Among the carboxamides tested, the fungicide benzovindyflupyr showed the highest intrinsic activity for both fungi.
Molecular identification was carried out as follows:
Sanger sequencing was used for sequencing and analysis of fungal isolates. The sequenced genomic regions used in the analysis were the internal transcribed spacer of the ribosomal DNA (ITS-rDNA) and
the tubulin 2 (TUB2), the histone 3 (HIS3), the chitin synthase (CHS1), the glutamine synthetase (GS), the translation elongation factor 1 (TEF1), and the actin (ACT) genes.
Concatenated analysis of the sequences from the isolates, along with GenBank-NCBI deposited sequences, was used for the correct and precise identification of the species of each isolate. Symptomatic soybean with ‘pod anomaly’ were sampled and isolations made from 20 pods during the 2021/2022 cropping season demonstrate a complex of fungal pathogens involved in the pods and are described below in Table 3.
Table 3
Symptomatic soybean with ‘pod anomaly’ were sampled and isolations made from 254 pods and 176 stems from the 2022/2023 cropping season demonstrate a complex of fungal pathogens involved in the pods and are described below in Table 4.
Table 4
Table 5 below describes a further analysis of the data from Table 4 and from 47 isolates of Colletotrichum spp, two main species are indicated, Colletotrichum clivicola and Colletotrichum truncatum.
Table 5
Table 6 below describes a further analysis of the data from Table 4 and from 66 isolates of Diaporthe spp a number of species are indicated, Diaporthe longicolla, Diaporthe Ueckerae, Diaporthe macadamiae and Diaporthe pyracanthae. These results confirm that the species responsible for stem canker are different from those associated with pod anomaly.
Table 6
Table 7 below describes a further analysis of the data from Table 4 and from 38 isolates of Fusarium spp a number of species are indicated, F. oxysporum spp complex, F. fujikuroi spp complex and F. incarnatum-equiseti spp complex.
Table 7
Symptomatic soybean with ‘pod anomaly’ were sampled and isolations made from 80 pods from the 2023/2024 cropping season demonstrate a complex of fungal pathogens involved in the pods and are described below in Table 8.
Table 8 To assess the effect of a compound of formula (I) (benzovindiflupyr) on seed decay (caused by pod anomaly) in post harvest soybean the following procedure was followed:
After harvest grains/seeds of soybean were collected and stored in a large bag under ambient conditions (~25 °C) for a period of 5 months. For each method of treatment 100 g of grains/seeds were weighed
from the large bag in a randomized way at monthly intervals. To assess the impact of pod anomaly on the seeds, the seeds were classified and separated into good/healthy and infected/decayed seeds every month for each method of treatment and after each monthly evaluation those grains/seeds were returned to the same large bag. The results of this analysis are displayed in table 9 below.
Table 9
1Combined with Aureo® adjuvant at 400ml/ha
2Combined with Mess® adjuvant at 375ml/ha
3 Combined with Strides® adjuvant at 375 ml/ha
After 5 months in storage conditions the grains were further analysed to identify the fungal pathogens present. 25 seeds which showed symptoms of pod anomaly were put into an acrylic sterilized box containing two absorbent papers moistened with deionized water for humidity. They were placed in a randomized way approximately one cm from each other and kept in a temperature controlled environment (~ 20°C) and protected from light. After approximately 7 days, those pathogens in each seed were identified using the same methodology as described above. The results are summarized in table 10 below and were similar to the previous data obtained above from the stem and pods:
Table 10
Abbreviations used in Table 9:
AZT = Azoxystrobin
BIX = Bixafen
FXD = Fluxapyroxad
MZB = Mancozeb
PTZ = Prothioconazol
STL = Benzovindiflupyr
Claims
2. The use according to claim 1 , wherein the pod anomaly comprises symptoms of pod decay, abnormal seed phenotype and/or stem blight.
3. The use according to claim 1 or claim 2, wherein the pod anomaly manifests at the R4 or R5 stage of the soybean plants reproductive cycle.
4. The use according to any one of claims 1 to 3, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp. and Diaporthe spp.
5. The use according to any one of claims 1 to 4, wherein the pod anomaly comprises phytopathogenic fungi selected from the group consisting of Cercospora spp., Colletotrichum spp., Corynespora spp., Diaporthe spp. and Fusarium spp.
6. The use according to claim 4 or claim 5, wherein the Diaporthe species comprise at least Diaporthe miriciae and Diaporthe longicolla.
7. The use according to any one of claims 4 to 6, wherein the Colletotrichum species comprise at least Colletotrichum cliviicola and Colletotrichum truncatum.
8. The use according to any one of claims 4 to 7, wherein the Corynespora spp. species comprise at least Corynespora cassiicola.
9. The use according to claim 5, wherein the Fusarium spp. species comprise at least Fusarium fujikuroi and Fusarium incarnatum.
11. The use according to any one of claims 1 to 10, wherein the compound of formula (I) is applied to soybean at a rate of 5 to 200 g a.i./ha.
12. The use according to any one of claims 1 to 11 , wherein the soybean is a transgenic soybean plant.
13. The use according to any one of claims 1 to 12, wherein the composition comprising a compound of formula (I) further comprises at least one fungicidally active compound.
14. A method of combating, preventing or controlling pod anomaly in soybean, which comprises applying to the crop of soybean plants, the locus thereof, or propagation material thereof, a compound of formula (I) or a composition comprising a compound of formula (I).
15. A method of combating, preventing or controlling seed decay in post harvest soybean, which comprises applying to the crop of soybean plants, the locus thereof, or propagation material thereof, a compound of formula (I) or a composition comprising a compound of formula (I).
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23157237.1 | 2023-02-17 | ||
| EP23157237 | 2023-02-17 | ||
| EP23187961 | 2023-07-26 | ||
| EP23187961.0 | 2023-07-26 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024170723A1 true WO2024170723A1 (en) | 2024-08-22 |
Family
ID=89941133
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/053952 Ceased WO2024170723A1 (en) | 2023-02-17 | 2024-02-16 | Method of pathogen control in soybean |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2024170723A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN119061193A (en) * | 2024-11-05 | 2024-12-03 | 浙江省农业科学院 | A primer and probe combination, kit and application thereof for RPA-LFD of rice seedling pathogen-Fusarium fujikura species banding bacteria |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007048556A1 (en) | 2005-10-25 | 2007-05-03 | Syngenta Participations Ag | Heterocyclic amide derivatives useful as microbiocides |
| WO2008131901A1 (en) | 2007-04-25 | 2008-11-06 | Syngenta Participations Ag | Fungicidal compositions |
| US20210137119A1 (en) * | 2017-04-27 | 2021-05-13 | Upl Ltd | Fungicidal combinations |
-
2024
- 2024-02-16 WO PCT/EP2024/053952 patent/WO2024170723A1/en not_active Ceased
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007048556A1 (en) | 2005-10-25 | 2007-05-03 | Syngenta Participations Ag | Heterocyclic amide derivatives useful as microbiocides |
| WO2008131901A1 (en) | 2007-04-25 | 2008-11-06 | Syngenta Participations Ag | Fungicidal compositions |
| US20210137119A1 (en) * | 2017-04-27 | 2021-05-13 | Upl Ltd | Fungicidal combinations |
Non-Patent Citations (7)
| Title |
|---|
| "Compendium of Herbicide Adjuvants", 2010, SOUTHERN ILLINOIS UNIVERSITY |
| "McCutcheon's Detergents and Emulsifiers Annual", 1981, MC PUBLISHING CORP. |
| CLARK ADORA: "A18993 Fungicide", EPA PESTICIDE REGISTRATION, 28 August 2015 (2015-08-28), pages 1 - 31, XP093060395, Retrieved from the Internet <URL:https://www3.epa.gov/pesticides/chem_search/ppls/000100-01475-20150828.pdf> [retrieved on 20230703] * |
| KOEHLER ALYSSA: "Delaware Soybean Board Survey and Baseline Fungicide Sensitivities of Fungal Pathogens in Mid-Atlantic Soybean Production", 2019 FINAL REPORT:, 31 March 2020 (2020-03-31), pages 1 - 7, XP093060390, Retrieved from the Internet <URL:https://www.soybeanresearchdata.com/download.aspx?file=Progress5File&name=53109_2019_DSB_Report_Koehler.pdf> [retrieved on 20230703] * |
| SYNGENTA: "Elatus fungicide label", HTTPS://WWW3.EPA.GOV/, 17 December 2020 (2020-12-17), pages 1 - 46, XP093060590, Retrieved from the Internet <URL:https://www3.epa.gov/pesticides/chem_search/ppls/000100-01480-20201217.pdf> [retrieved on 20230704] * |
| SYNGENTA: "MITRION", REGISTRADO NO MINISTÉRIO DA AGRICULTURA, PECUÁRIA E ABASTECIMENTO - MAPA SOB NO 07621, 4 July 2023 (2023-07-04), pages 1 - 30, XP093150672, Retrieved from the Internet <URL:https://www.adapar.pr.gov.br/sites/adapar/arquivos_restritos/files/documento/2023-08/mitrion.pdf> [retrieved on 20240411] * |
| WISE KRISTEN: "Fungicide Efficacy for Foliar Soybean Diseases", CROP PROTECTION NETWORK, 12 March 2021 (2021-03-12), pages 1 - 3, XP093060517, Retrieved from the Internet <URL:https://crop-protection-network.s3.amazonaws.com/publications/fungicide-efficacy-for-control-of-soybean-foliar-diseases-filename-2021-03-12-182833.pdf> [retrieved on 20230704] * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN119061193A (en) * | 2024-11-05 | 2024-12-03 | 浙江省农业科学院 | A primer and probe combination, kit and application thereof for RPA-LFD of rice seedling pathogen-Fusarium fujikura species banding bacteria |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8440590B2 (en) | Method for controlling soybean rust | |
| JP2010520865A5 (en) | ||
| US20250089720A1 (en) | Fungicidal mixtures comprising combination containing phthalimide | |
| US20250302040A1 (en) | Fungicidal combinations of fluazinam and uses thereof | |
| JP2021193085A (en) | Pesticidal compositions | |
| US9049865B2 (en) | Use of fungicidal active substances for controlling mycoses on plants of the palm family | |
| US10798942B2 (en) | Abiotic stress tolerance | |
| US10798943B2 (en) | Use of 6-anilino purine derivatives to improve heat stress tolerance of rice seedlings | |
| WO2024170723A1 (en) | Method of pathogen control in soybean | |
| EP3451835B1 (en) | Insecticide mixtures comprising loline alkaloids | |
| JP2013505910A (en) | Method for reducing plant flower failure | |
| AU2015271898B2 (en) | A synergistic composition comprising insecticides and fungicides | |
| EP4616711A2 (en) | Fungicidal use | |
| WO2017004744A1 (en) | Use of penflufen | |
| JP3670729B2 (en) | Agricultural fungicide | |
| AU2017261561B2 (en) | A synergistic insecticidal composition comprising methomyl and fipronil | |
| EP4492981A1 (en) | Fungicidal mixture composition comprising sulphur | |
| WO2025073912A1 (en) | Compositions and methods for controlling infestation of plants by phytopathogenic fungi | |
| WO2025073941A1 (en) | Compositions and methods for controlling infestation of plants by phytopathogenic fungi | |
| WO2025238213A1 (en) | Method for treatment of root rot disease caused by plant pathogens in legume crops | |
| WO2025073919A1 (en) | Compositions and methods for controlling infestation of plants by phytopathogenic fungi |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24705166 Country of ref document: EP Kind code of ref document: A1 |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112025017239 Country of ref document: BR |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |