WO2024149821A1 - Treatment of cancer using a hla-a2/wt1 x cd3 bispecific antibody and venetoclax/azacitidine - Google Patents
Treatment of cancer using a hla-a2/wt1 x cd3 bispecific antibody and venetoclax/azacitidine Download PDFInfo
- Publication number
- WO2024149821A1 WO2024149821A1 PCT/EP2024/050523 EP2024050523W WO2024149821A1 WO 2024149821 A1 WO2024149821 A1 WO 2024149821A1 EP 2024050523 W EP2024050523 W EP 2024050523W WO 2024149821 A1 WO2024149821 A1 WO 2024149821A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- hla
- seq
- bispecific antibody
- antigen binding
- heavy chain
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
- C07K16/2809—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily against the T-cell receptor (TcR)-CD3 complex
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/63—Compounds containing para-N-benzenesulfonyl-N-groups, e.g. sulfanilamide, p-nitrobenzenesulfonyl hydrazide
- A61K31/635—Compounds containing para-N-benzenesulfonyl-N-groups, e.g. sulfanilamide, p-nitrobenzenesulfonyl hydrazide having a heterocyclic ring, e.g. sulfadiazine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7042—Compounds having saccharide radicals and heterocyclic rings
- A61K31/7052—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides
- A61K31/706—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
- C07K16/2833—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily against MHC-molecules, e.g. HLA-molecules
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
- A61K39/39533—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals
- A61K39/39558—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals against tumor tissues, cells, antigens
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/46—Hybrid immunoglobulins
- C07K16/468—Immunoglobulins having two or more different antigen binding sites, e.g. multifunctional antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/31—Immunoglobulins specific features characterized by aspects of specificity or valency multispecific
Definitions
- the present invention relates to the treatment of cancer, in particular to the treatment of cancer using a HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine.
- T-cell activating bispecific antibodies are a promising class of cancer therapeutics, designed to engage cytotoxic T cells against tumor cells.
- the simultaneous binding of such an antibody to CD3 on T-cells and to an antigen expressed on the tumor cells will force a temporary interaction between tumor cell and T cell, causing activation of the T-cell and subsequent lysis of the tumor cell.
- WT1 Wilms tumor 1, Wilms tumor protein
- WT1 is an oncogenic transcription factor involved in cell proliferation, differentiation, as well as apoptosis and organ development, whose expression in normal adult tissue is rare (Hinrichs and Restifo, Nat Biotechnol (2013) 31, 999-1008).
- WT1 is, however, reported to be overexpressed in several types of haematological maligancies and a wide range of solid tumors (Van Driessche et al., Oncologist (2012) 17, 250-259).
- WT1 is a nuclear protein, localized intracellularly.
- Intracellular protein can be degraded in the proteasome, processed and presented on the cell surface by major histocompatibility complex (MHC) I as T cell epitopes, and recognized by T cell receptors (TCR).
- MHC major histocompatibility complex
- TCR T cell receptors
- WT1 -derived peptides are presented in the context of HLA-A2 on the cell surface and can trigger T cell recognition.
- T-cell activating bispecific antibodies targeting HLA-A2/WT1 have been described in WO 2019/122052. Such T-cell activating bispecific antibodies may be useful, e.g., in the treatment of acute myeloid leukemia (AML).
- a HLA-A2/WT1 x CD3 bispecific antibody (“WT1-TCB”) is currently under investigation in a phase I clinical trial in patients with relap sed/refractory (r/r) AML (Augsberger et al., Blood (2021) doi: 10.1182/blood.2020010477; NCT04580121).
- r/r relap sed/refractory
- the present inventors have found that combination of HLA-A2/WT1 targeted T-cell activating bispecific antibodies with venetoclax/azacitidine leads to enhanced activity as compared to HLA- A2/WT1 targeted T-cell activated bispecific antibody alone.
- the inventors have surprisingly found that tumor cell lysis in vitro, as well as antitumor efficacy in vivo, induced by HLA-A2/WT1 x CD3 bispecific antibody was enhanced by the addition of venetoclax/azacitidine.
- the present invention provides a HLA-A2/WT1 x CD3 bispecific antibody for use in the treatment of a cancer in an individual, wherein the treatment comprises administration of the HLA-A2/WT1 x CD3 bispecific antibody in combination with venetocl ax/ azaciti dine .
- the invention provides the use of a HLA-A2/WT1 x CD3 bispecific antibody in the manufacture of a medicament for the treatment of cancer in an individual, wherein the treatment comprises administration of the HLA-A2/WT1 x CD3 bispecific antibody in combination with venetoclax/azacitidine.
- the invention provides a method for treating cancer in an individual comprising administering to the individual a HLA-A2/WT1 x CD3 bispecific antibody and venetocl ax/ azaciti dine .
- the invention also provides a kit comprising a first medicament comprising a HLA- A2/WT1 x CD3 bispecific antibody, and a second medicament comprising venetoclax and/or a third medicament comprising azacitidine, and optionally further comprising a package insert comprising instructions for administration of the first medicament in combination with the second and/or the third medicament for treating cancer in an individual.
- the HLA-A2/WT1 x CD3 bispecific antibodies, methods, uses or kits described above and herein, may incorporate, singly or in combination, any of the features described in the following (unless the context dictates otherwise).
- the HLA-A2/WT1 x CD3 bispecific antibody herein is a bispecific antibody that specifically binds to CD3 and to HLA-A2/WT1, particularly HLA-A2/WT1 RMF.
- Particularly useful HLA- A2/WT1 x CD3 bispecific antibodies for use in the present invention are described e.g. in PCT publication no. WO 2019/122052 (incorporated herein by reference in its entirety).
- bispecific means that the antibody is able to specifically bind to at least two distinct antigenic determinants.
- a bispecific antibody comprises two antigen binding sites, each of which is specific for a different antigenic determinant.
- the bispecific antibody is capable of simultaneously binding two antigenic determinants, particularly two antigenic determinants expressed on two distinct cells.
- antigenic determinant is synonymous with “antigen” and “epitope”, and refers to a site (e.g. a contiguous stretch of amino acids or a conformational configuration made up of different regions of non-contiguous amino acids) on a polypeptide macromolecule to which an antigen binding moiety binds, forming an antigen binding moiety-antigen complex.
- Useful antigenic determinants can be found, for example, on the surfaces of tumor cells, on the surfaces of virus-infected cells, on the surfaces of other diseased cells, on the surface of immune cells, free in blood serum, and/or in the extracellular matrix (ECM).
- ECM extracellular matrix
- an antigen binding moiety refers to a polypeptide molecule that specifically binds to an antigenic determinant.
- an antigen binding moiety is able to direct the entity to which it is attached (e.g. a second antigen binding moiety) to a target site, for example to a specific type of tumor cell bearing the antigenic determinant.
- an antigen binding moiety is able to activate signaling through its target antigen, for example a T cell receptor complex antigen.
- Antigen binding moieties include antibodies and fragments thereof as further defined herein. Particular antigen binding moieties include an antigen binding domain of an antibody, comprising an antibody heavy chain variable region and an antibody light chain variable region.
- the antigen binding moieties may comprise antibody constant regions as further defined herein and known in the art.
- Useful heavy chain constant regions include any of the five isotypes: a, 5, a, y, or p.
- Useful light chain constant regions include any of the two isotypes: K and .
- ELISA enzyme-linked immunosorbent assay
- SPR surface plasmon resonance
- an antigen binding moiety that binds to the antigen, or an antibody comprising that antigen binding moiety has a dissociation constant (KD) of ⁇ 1 pM, ⁇ 100 nM, ⁇ 10 nM, ⁇ 1 nM, ⁇ 0.1 nM, ⁇ 0.01 nM, or ⁇ 0.001 nM (e.g. 10' 8 M or less, e.g. from 10' 8 M to 10' 13 M, e.g., from 10' 9 M to 10’ 13 M).
- KD dissociation constant
- Binding affinity refers to intrinsic binding affinity which reflects a 1 : 1 interaction between members of a binding pair (e.g., an antigen binding moiety and an antigen, or a receptor and its ligand).
- the affinity of a molecule X for its partner Y can generally be represented by the dissociation constant (KD), which is the ratio of dissociation and association rate constants (koir and k on , respectively).
- affinities may comprise different rate constants, as long as the ratio of the rate constants remains the same.
- Affinity can be measured by well established methods known in the art, including those described herein.
- a particular method for measuring affinity is Surface Plasmon Resonance (SPR).
- CD3 refers to any native CD3 from any vertebrate source, including mammals such as primates (e.g. humans), non-human primates (e.g. cynomolgus monkeys) and rodents (e.g. mice and rats), unless otherwise indicated.
- the term encompasses “full-length,” unprocessed CD3 as well as any form of CD3 that results from processing in the cell.
- the term also encompasses naturally occurring variants of CD3, e.g., splice variants or allelic variants.
- CD3 is human CD3, particularly the epsilon subunit of human CD3 (CD3s).
- the amino acid sequence of human CD3s is shown in UniProt (www.uniprot.org) accession no.
- WT1 also known as “Wilms tumor 1” or “Wilms tumor protein”, refers to any native WT1 from any vertebrate source, including mammals such as primates (e.g. humans), non-human primates (e.g. cynomolgus monkeys) and rodents (e.g. mice and rats), unless otherwise indicated.
- the term encompasses “full-length,” unprocessed WT1 as well as any form of WT1 that results from processing in the cell.
- the term also encompasses naturally occurring variants of WT1, e.g., splice variants or allelic variants.
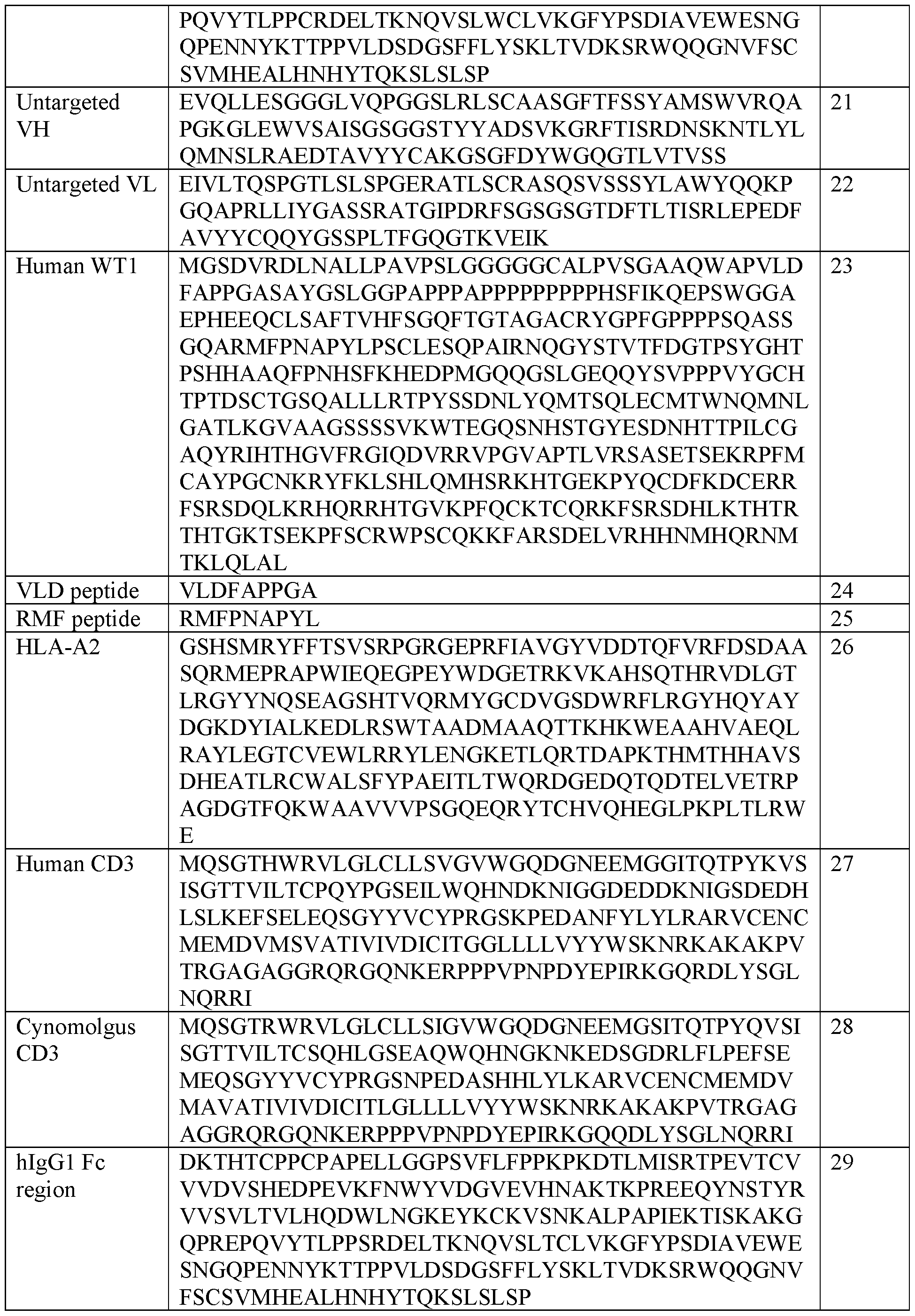
- WT1 is human WT1, particularly the protein of SEQ ID NO: 23. Human WT1 is described in UniProt (www.uniprot.org) accession no. P19544 (entry version 215), and an amino acid sequence of human WT1 is also shown in SEQ ID NO: 23.
- VLD VLD peptide
- WTIVLD WT1 derived peptide having the amino acid sequence VLDFAPPGA (SEQ ID NO: 24; position 37-45 of the WT1 protein of SEQ ID NO: 23).
- RMF RMF peptide
- WTIRMF WT1 derived peptide having the amino acid sequence RMFRNAPYL (SEQ ID NO: 25; position 126-134 of the WT1 protein of SEQ ID NO: 23).
- HLA-A2 refers to a human leukocyte antigen serotype in the HL A- A serotype group.
- the HLA-A2 protein encoded by the respective HL A gene constitutes the a chain of the respective class I MHC (major histocompatibility complex) protein, which further comprises a P2 microglobulin subunit.
- a specific HLA-A2 protein is HLA-A201 (also referred to as HLA-A0201, HLA-A02.01, or HLA- A*02:01).
- the HLA-A2 protein described herein is HLA-A201.
- An exemplary sequence of human HLA-A2 is given in SEQ ID NO: 26.
- HLA-A2/WT1 refers to a complex of a HLA-A2 molecule and a WT1 derived peptide (also referred to herein as a “WT1 peptide”), specifically the RMF or VLD peptide (“HLA-A2/WT1RMF” and “HLA-A2/WT1 VLD”, respectively).
- the bispecific antibody used in the present invention specifically may bind to either the HLA-A2/WT1RMF or the HLA-A2/WT1VLD complex.
- the terms “first”, “second” or “third” with respect to Fab molecules etc. are used for convenience of distinguishing when there is more than one of each type of moiety. Use of these terms is not intended to confer a specific order or orientation of the bispecific antibody unless explicitly so stated.
- antibody herein is used in the broadest sense and encompasses various antibody structures, including but not limited to monoclonal antibodies, polyclonal antibodies, multispecific antibodies (e.g. bispecific antibodies), and antibody fragments so long as they exhibit the desired antigen-binding activity.
- full length antibody “intact antibody,” and “whole antibody” are used herein interchangeably to refer to an antibody having a structure substantially similar to a native antibody structure.
- antibody fragment refers to a molecule other than an intact antibody that comprises a portion of an intact antibody that binds the antigen to which the intact antibody binds.
- antibody fragments include but are not limited to Fv, Fab, Fab', Fab’-SH, F(ab')2, diabodies, linear antibodies, single-chain antibody molecules (e.g. scFv), and single-domain antibodies.
- scFv single-chain antibody molecules
- Diabodies are antibody fragments with two antigen-binding sites that may be bivalent or bispecific.
- Single-domain antibodies are antibody fragments comprising all or a portion of the heavy chain variable domain or all or a portion of the light chain variable domain of an antibody.
- a single-domain antibody is a human single-domain antibody (Domantis, Inc., Waltham, MA; see e.g. U.S. Patent No. 6,248,516 Bl).
- Antibody fragments can be made by various techniques, including but not limited to proteolytic digestion of an intact antibody as well as production by recombinant host cells (e.g. E. coli or phage), as described herein.
- variable region refers to the domain of an antibody heavy or light chain that is involved in binding the antibody to antigen.
- the variable domains of the heavy chain and light chain (VH and VL, respectively) of a native antibody generally have similar structures, with each domain comprising four conserved framework regions (FRs) and three hypervariable regions (HVRs). See, e.g., Kindt et al., Kuby Immunology, 6 th ed., W.H. Freeman and Co., page 91 (2007).
- a single VH or VL domain may be sufficient to confer antigen-binding specificity.
- Kabat numbering refers to the numbering system set forth by Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, MD (1991).
- amino acid positions of all constant regions and domains of the heavy and light chain are numbered according to the Kabat numbering system described in Kabat, et al., Sequences of Proteins of Immunological Interest, 5th ed., Public Health Service, National Institutes of Health, Bethesda, MD (1991), referred to as “numbering according to Kabat” or “Kabat numbering” herein.
- Kabat numbering system see pages 647-660 of Kabat, et al., Sequences of Proteins of Immunological Interest, 5th ed., Public Health Service, National Institutes of Health, Bethesda, MD (1991)
- CL light chain constant domain
- Kabat EU index numbering system see pages 661-723
- CHI heavy chain constant domains
- hypervariable region refers to each of the regions of an antibody variable domain which are hypervariable in sequence and which determine antigen binding specificity, for example “complementarity determining regions” (“CDRs”).
- CDRs complementarity determining regions
- antibodies comprise six CDRs; three in the VH (HCDR1, HCDR2, HCDR3), and three in the VL (LCDR1, LCDR2, LCDR3).
- Exemplary CDRs herein include:
- FR Framework or "FR” refers to variable domain residues other than hypervariable region (HVR) residues.
- the FR of a variable domain generally consists of four FR domains: FR1, FR2, FR3, and FR4. Accordingly, the HVR and FR sequences generally appear in the following order in VH (or VL) : FR1 -H 1 (L 1 )-FR2-H2(L2)-FR3 -H3 (L3 )-FR4.
- the “class” of an antibody or immunoglobulin refers to the type of constant domain or constant region possessed by its heavy chain.
- the heavy chain constant domains that correspond to the different classes of immunoglobulins are called a, 5, a, y, and p, respectively.
- a “Fab molecule” refers to a protein consisting of the VH and CHI domain of the heavy chain (the “Fab heavy chain”) and the VL and CL domain of the light chain (the “Fab light chain”) of an immunoglobulin.
- crossover Fab molecule also termed “Crossfab” is meant a Fab molecule wherein the variable domains or the constant domains of the Fab heavy and light chain are exchanged (i.e. replaced by each other), i.e. the crossover Fab molecule comprises a peptide chain composed of the light chain variable domain VL and the heavy chain constant domain 1 CHI (VL-CH1, in N- to C-terminal direction), and a peptide chain composed of the heavy chain variable domain VH and the light chain constant domain CL (VH-CL, in N- to C-terminal direction).
- the peptide chain comprising the heavy chain constant domain 1 CHI is referred to herein as the “heavy chain” of the (crossover) Fab molecule.
- the peptide chain comprising the heavy chain variable domain VH is referred to herein as the “heavy chain” of the (crossover) Fab molecule.
- a “conventional” Fab molecule is meant a Fab molecule in its natural format, i.e. comprising a heavy chain composed of the heavy chain variable and constant domains (VH- CH1, in N- to C-terminal direction), and a light chain composed of the light chain variable and constant domains (VL-CL, in N- to C-terminal direction).
- immunoglobulin molecule refers to a protein having the structure of a naturally occurring antibody.
- immunoglobulins of the IgG class are heterotetrameric glycoproteins of about 150,000 daltons, composed of two light chains and two heavy chains that are disulfide-bonded. From N- to C-terminus, each heavy chain has a variable domain (VH), also called a variable heavy domain or a heavy chain variable region, followed by three constant domains (CHI, CH2, and CH3), also called a heavy chain constant region.
- each light chain has a variable domain (VL), also called a variable light domain or a light chain variable region, followed by a constant light (CL) domain, also called a light chain constant region.
- VL variable domain
- CL constant light
- the heavy chain of an immunoglobulin may be assigned to one of five types, called a (IgA), 5 (IgD), 8 (IgE), y (IgG), or p (IgM), some of which may be further divided into subtypes, e.g. yi (IgGi), 72 (IgG?), 73 (IgGi), 74 (IgG4), ai (IgAi) and a? (IgA?).
- the light chain of an immunoglobulin may be assigned to one of two types, called kappa (K) and lambda (X), based on the amino acid sequence of its constant domain.
- K kappa
- X lambda
- An immunoglobulin essentially consists of two Fab molecules and an Fc domain, linked via the immunoglobulin hinge region.
- Fc domain or “Fc region” herein is used to define a C-terminal region of an immunoglobulin heavy chain that contains at least a portion of the constant region.
- the term includes native sequence Fc regions and variant Fc regions.
- the boundaries of the Fc region of an IgG heavy chain might vary slightly, the human IgG heavy chain Fc region is usually defined to extend from Cys226, or from Pro230, to the carboxyl-terminus of the heavy chain.
- antibodies produced by host cells may undergo post-translational cleavage of one or more, particularly one or two, amino acids from the C-terminus of the heavy chain.
- an antibody produced by a host cell by expression of a specific nucleic acid molecule encoding a full- length heavy chain may include the full-length heavy chain, or it may include a cleaved variant of the full-length heavy chain.
- This may be the case where the final two C-terminal amino acids of the heavy chain are glycine (G446) and lysine (K447, numbering according to Kabat EU index). Therefore, the C-terminal lysine (Lys447), or the C-terminal glycine (Gly446) and lysine (K447), of the Fc region may or may not be present.
- a “subunit” of an Fc domain as used herein refers to one of the two polypeptides forming the dimeric Fc domain, i.e. a polypeptide comprising C-terminal constant regions of an immunoglobulin heavy chain, capable of stable self-association.
- a subunit of an IgG Fc domain comprises an IgG CH2 and an IgG CH3 constant domain.
- a “modification promoting the association of the first and the second subunit of the Fc domain” is a manipulation of the peptide backbone or the post-translational modifications of an Fc domain subunit that reduces or prevents the association of a polypeptide comprising the Fc domain subunit with an identical polypeptide to form a homodimer.
- a modification promoting association as used herein particularly includes separate modifications made to each of the two Fc domain subunits desired to associate (i.e. the first and the second subunit of the Fc domain), wherein the modifications are complementary to each other so as to promote association of the two Fc domain subunits.
- a modification promoting association may alter the structure or charge of one or both of the Fc domain subunits so as to make their association sterically or electrostatically favorable, respectively.
- (hetero)dimerization occurs between a polypeptide comprising the first Fc domain subunit and a polypeptide comprising the second Fc domain subunit, which might be non-identical in the sense that further components fused to each of the subunits (e.g. antigen binding moieties) are not the same.
- the modification promoting association comprises an amino acid mutation in the Fc domain, specifically an amino acid substitution.
- the modification promoting association comprises a separate amino acid mutation, specifically an amino acid substitution, in each of the two subunits of the Fc domain.
- effector functions refers to those biological activities attributable to the Fc region of an antibody, which vary with the antibody isotype.
- antibody effector functions include: Clq binding and complement dependent cytotoxicity (CDC), Fc receptor binding, antibody-dependent cell-mediated cytotoxicity (ADCC), antibody-dependent cellular phagocytosis (ADCP), cytokine secretion, immune complex-mediated antigen uptake by antigen presenting cells, down regulation of cell surface receptors (e.g. B cell receptor), and B cell activation.
- Percent (%) amino acid sequence identity with respect to a reference polypeptide sequence is defined as the percentage of amino acid residues in a candidate sequence that are identical with the amino acid residues in the reference polypeptide sequence, after aligning the sequences and introducing gaps, if necessary, to achieve the maximum percent sequence identity, and not considering any conservative substitutions as part of the sequence identity. Alignment for purposes of determining percent amino acid sequence identity can be achieved in various ways that are within the skill in the art, for instance, using publicly available computer software such as BLAST, BLAST-2, Clustal W, Megalign (DNASTAR) software or the FASTA program package.
- % amino acid sequence identity values are generated using the ggsearch program of the FASTA package version 36.3.8c or later with a BLOSUM50 comparison matrix.
- the FASTA program package was authored by W. R. Pearson and D. J. Lipman (1988), “Improved Tools for Biological Sequence Analysis”, PNAS 85:2444-2448; W. R. Pearson (1996) “Effective protein sequence comparison” Meth. Enzymol. 266:227- 258; and Pearson et. al.
- Genomics 46:24-36 is publicly available from http://fasta.bioch.virginia.edu/fasta_www2/fasta_down.shtml.
- an “activating Fc receptor” is an Fc receptor that following engagement by an Fc domain of an antibody elicits signaling events that stimulate the receptor-bearing cell to perform effector functions.
- Human activating Fc receptors include FcyRIIIa (CD16a), FcyRI (CD64), FcyRIIa (CD32), and FcaRI (CD89).
- Reduced binding for example reduced binding to an Fc receptor, refers to a decrease in affinity for the respective interaction, as measured for example by SPR.
- the term includes also reduction of the affinity to zero (or below the detection limit of the analytic method), i.e. complete abolishment of the interaction.
- increased binding refers to an increase in binding affinity for the respective interaction.
- fused is meant that the components (e.g. a Fab molecule and an Fc domain subunit) are linked by peptide bonds, either directly or via one or more peptide linkers.
- the HLA-A2/WT1 x CD3 bispecific antibody comprises a first antigen binding moiety that specifically binds to CD3, and a second antigen binding moiety that specifically binds to HLA- A2/WT1, particularly HLA-A2/WT1RMF.
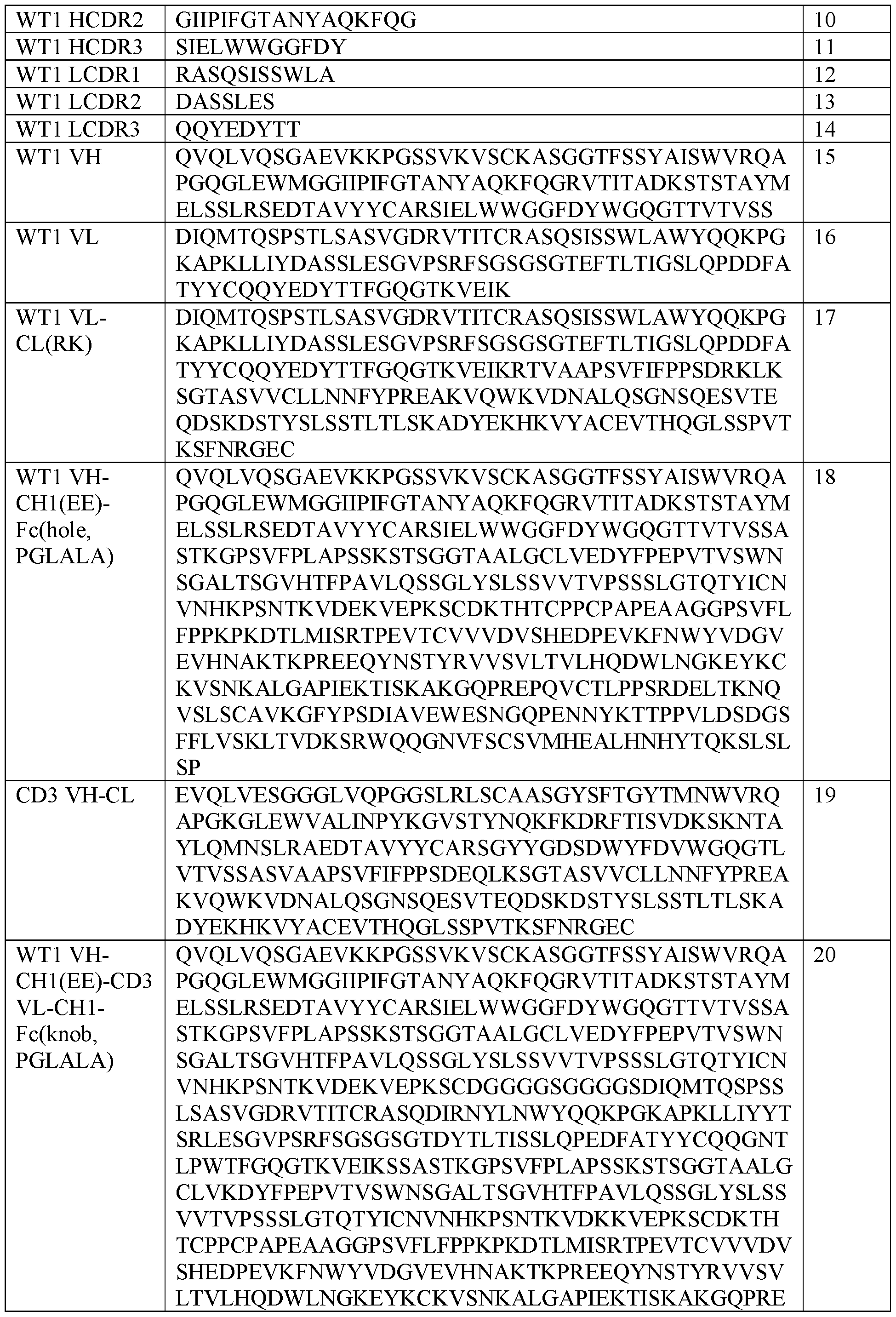
- the first antigen binding moiety comprises a heavy chain variable region comprising the heavy chain CDR (HCDR) 1 of SEQ ID NO: 1, the HCDR2 of SEQ ID NO: 2, and the HCDR3 of SEQ ID NO: 3; and a light chain variable region comprising the light chain CDR (LCDR) 1 of SEQ ID NO: 4, the LCDR2 of SEQ ID NO: 5 and the LCDR3 of SEQ ID NO: 6.
- the second antigen binding moiety comprises a heavy chain variable region comprising the heavy chain CDR (HCDR) 1 of SEQ ID NO: 9, the HCDR2 of SEQ ID NO: 10, and the HCDR3 of SEQ ID NO: 11; and a light chain variable region comprising the light chain CDR (LCDR) 1 of SEQ ID NO: 12, the LCDR2 of SEQ ID NO: 13 and the LCDR3 of SEQ ID NO: 14.
- the HLA-A2/WT1 x CD3 bispecific antibody comprises
- a first antigen binding moiety that specifically binds to CD3 and comprises a heavy chain variable region comprising the heavy chain CDR (HCDR) 1 of SEQ ID NO: 1, the HCDR2 of SEQ ID NO: 2, and the HCDR3 of SEQ ID NO: 3; and a light chain variable region comprising the light chain CDR (LCDR) 1 of SEQ ID NO: 4, the LCDR2 of SEQ ID NO: 5 and the LCDR3 of SEQ ID NO: 6; and
- a second antigen binding moiety that specifically binds to HLA-A2/WT1 and comprises a heavy chain variable region comprising the heavy chain CDR (HCDR) 1 of SEQ ID NO: 9, the HCDR2 of SEQ ID NO: 10, and the HCDR3 of SEQ ID NO: 11; and a light chain variable region comprising the light chain CDR (LCDR) 1 of SEQ ID NO: 12, the LCDR2 of SEQ ID NO: 13 and the LCDR3 of SEQ ID NO: 14.
- the first antigen binding moiety comprises a heavy chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 7 and a light chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 8.
- the first antigen binding moiety comprises the heavy chain variable region sequence of SEQ ID NO: 7 and the light chain variable region sequence of SEQ ID NO: 8.
- the second antigen binding moiety comprises a heavy chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 15 and a light chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 16.
- the second antigen binding moiety comprises the heavy chain variable region sequence of SEQ ID NO: 15 and the light chain variable region sequence of SEQ ID NO: 16.
- the first and/or the second antigen binding moiety is a Fab molecule.
- the first antigen binding moiety is a crossover Fab molecule wherein either the variable or the constant regions of the Fab light chain and the Fab heavy chain are exchanged.
- the second antigen binding moiety preferably is a conventional Fab molecule.
- the first and the second antigen binding moiety of the bi specific antibody are both Fab molecules, and in one of the antigen binding moieties (particularly the first antigen binding moiety) the variable domains VL and VH of the Fab light chain and the Fab heavy chain are replaced by each other, i) in the constant domain CL of the first antigen binding moiety the amino acid at position 124 is substituted by a positively charged amino acid (numbering according to Kabat), and wherein in the constant domain CHI of the first antigen binding moiety the amino acid at position 147 or the amino acid at position 213 is substituted by a negatively charged amino acid (numbering according to Kabat EU index); or ii) in the constant domain CL of the second antigen binding moiety the amino acid at position 124 is substituted by a positively charged amino acid (numbering according to Kabat), and wherein in the constant domain CHI of the second antigen binding moiety the amino acid at position 147 or the amino acid at position 213 is substituted by a negatively charged amino acid
- the bispecific antibody does not comprise both modifications mentioned under i) and ii).
- the constant domains CL and CHI of the antigen binding moiety having the VH/VL exchange are not replaced by each other (i.e. remain unexchanged).
- the amino acid at position 124 is substituted independently by lysine (K), arginine (R) or histidine (H) (numbering according to Kabat), and in the constant domain CHI of the first antigen binding moiety the amino acid at position 147 or the amino acid at position 213 is substituted independently by glutamic acid (E), or aspartic acid (D) (numbering according to Kabat EU index); or ii) in the constant domain CL of the second antigen binding moiety the amino acid at position 124 is substituted independently by lysine (K), arginine (R) or histidine (H) (numbering according to Kabat), and in the constant domain CHI of the second antigen binding moiety the amino acid at position 147 or the amino acid at position 213 is substituted independently by glutamic acid (E), or aspartic acid (D) (numbering according to Kabat EU index).
- the amino acid at position 124 is substituted independently by lysine (K), arginine (R) or histidine (H) (numbering according to Kabat), and in the constant domain CHI of the second antigen binding moiety the amino acid at position 147 or the amino acid atposition 213 is substituted independently by glutamic acid (E), or aspartic acid (D) (numbering according to Kabat EU index).
- the amino acid at position 124 is substituted independently by lysine (K), arginine (R) or histidine (H) (numbering according to Kabat), and in the constant domain CHI of the second antigen binding moiety the amino acid at position 147 is substituted independently by glutamic acid (E), or aspartic acid (D) (numbering according to Kabat EU index).
- the amino acid at position 124 is substituted independently by lysine (K), arginine (R) or histidine (H) (numbering according to Kabat) and the amino acid at position 123 is substituted independently by lysine (K), arginine (R) or histidine (H) (numbering according to Kabat), and in the constant domain CHI of the second antigen binding moiety the amino acid at position 147 is substituted independently by glutamic acid (E), or aspartic acid (D) (numbering according to Kabat EU index) and the amino acid at position 213 is substituted independently by glutamic acid (E), or aspartic acid (D) (numbering according to Kabat EU index).
- the amino acid at position 124 is substituted by lysine (K) (numbering according to Kabat) and the amino acid at position 123 is substituted by lysine (K) (numbering according to Kabat), and in the constant domain CHI of the second antigen binding moiety the amino acid at position 147 is substituted by glutamic acid (E) (numbering according to Kabat EU index) and the amino acid at position 213 is substituted by glutamic acid (E) (numbering according to Kabat EU index).
- the amino acid at position 124 is substituted by lysine (K) (numbering according to Kabat) and the amino acid at position 123 is substituted by arginine (R) (numbering according to Kabat), and in the constant domain CHI of the second antigen binding moiety the amino acid at position 147 is substituted by glutamic acid (E) (numbering according to Kabat EU index) and the amino acid at position 213 is substituted by glutamic acid (E) (numbering according to Kabat EU index).
- the constant domain CL of the second antigen binding moiety is of kappa isotype.
- first and the second antigen binding moiety are fused to each other, optionally via a peptide linker.
- the first and the second antigen binding moiety are each a Fab molecule and either (i) the second antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N- terminus of the Fab heavy chain of the first antigen binding moiety, or (ii) the first antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N-terminus of the Fab heavy chain of the second antigen binding moiety.
- the HLA-A2/WT1 x CD3 bispecific antibody provides monovalent binding to CD3.
- the HLA-A2/WT1 x CD3 bispecific antibody comprises a single antigen binding moiety that specifically binds to CD3, and two antigen binding moi eties that specifically bind to HLA-A2/WT1.
- the HLA-A2/WT1 x CD3 bispecific antibody comprises a third antigen binding moiety, particularly a Fab molecule, more particularly a conventional Fab molecule, that specifically binds to HLA-A2/WT1.
- the third antigen binding moiety may incorporate, singly or in combination, all of the features described hereinabove in relation to the second antigen binding moiety (e.g. the CDR sequences, variable region sequences, and/or amino acid substitutions in the constant regions).
- the third antigen moiety is identical to the first antigen binding moiety (e.g. is also a conventional Fab molecule and comprises the same amino acid sequences).
- the HLA-A2/WT1 x CD3 bispecific antibody further comprises an Fc domain composed of a first and a second subunit.
- the Fc domain is an IgG Fc domain.
- the Fc domain is an IgGi Fc domain.
- the Fc domain is an IgG4 Fc domain.
- the Fc domain is an IgG4 Fc domain comprising an amino acid substitution at position S228 (Kabat EU index numbering), particularly the amino acid substitution S228P. This amino acid substitution reduces in vivo Fab arm exchange of IgG4 antibodies (see Stubenrauch et al., Drug Metabolism and Disposition 38, 84-91 (2010)).
- the Fc domain is a human Fc domain.
- the Fc domain is a human IgGi Fc domain.
- An exemplary sequence of a human IgGi Fc region is given in SEQ ID NO: 29.
- the first, the second and, where present, the third antigen binding moiety are each a Fab molecule, (a) either (i) the second antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N-terminus of the Fab heavy chain of the first antigen binding moiety and the first antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N- terminus of the first subunit of the Fc domain, or (ii) the first antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N-terminus of the Fab heavy chain of the second antigen binding moiety and the second antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N-terminus of the first subunit of the Fc domain; and (b) the third antigen binding moiety, where present, is fused at the C-terminus of the Fab heavy chain to the N-terminus of the second subunit of the Fc domain.
- the Fc domain comprises a modification promoting the association of the first and the second subunit of the Fc domain.
- the site of most extensive protein-protein interaction between the two subunits of a human IgG Fc domain is in the CH3 domain.
- said modification is in the CH3 domain of the Fc domain.
- said modification promoting the association of the first and the second subunit of the Fc domain is a so-called “knob-into-hole” modification, comprising a “knob” modification in one of the two subunits of the Fc domain and a “hole” modification in the other one of the two subunits of the Fc domain.
- the knob-into-hole technology is described e.g. in US 5,731,168; US 7,695,936; Ridgway et al., Prot Eng 9, 617-621 (1996) and Carter, J Immunol Meth 248, 7-15 (2001).
- the method involves introducing a protuberance (“knob”) at the interface of a first polypeptide and a corresponding cavity (“hole”) in the interface of a second polypeptide, such that the protuberance can be positioned in the cavity so as to promote heterodimer formation and hinder homodimer formation.
- Protuberances are constructed by replacing small amino acid side chains from the interface of the first polypeptide with larger side chains (e.g. tyrosine or tryptophan).
- Compensatory cavities of identical or similar size to the protuberances are created in the interface of the second polypeptide by replacing large amino acid side chains with smaller ones (e.g. alanine or threonine).
- an amino acid residue in the CH3 domain of the first subunit of the Fc domain is replaced with an amino acid residue having a larger side chain volume, thereby generating a protuberance within the CH3 domain of the first subunit which is positionable in a cavity within the CH3 domain of the second subunit, and an amino acid residue in the CH3 domain of the second subunit of the Fc domain is replaced with an amino acid residue having a smaller side chain volume, thereby generating a cavity within the CH3 domain of the second subunit within which the protuberance within the CH3 domain of the first subunit is positionable.
- said amino acid residue having a larger side chain volume is selected from the group consisting of arginine (R), phenylalanine (F), tyrosine (Y), and tryptophan (W).
- said amino acid residue having a smaller side chain volume is selected from the group consisting of alanine (A), serine (S), threonine (T), and valine (V).
- the protuberance and cavity can be made by altering the nucleic acid encoding the polypeptides, e.g. by site-specific mutagenesis, or by peptide synthesis.
- the threonine residue at position 366 is replaced with a tryptophan residue (T366W)
- the tyrosine residue at position 407 is replaced with a valine residue (Y407V) and optionally the threonine residue at position 366 is replaced with a serine residue (T366S) and the leucine residue at position 368 is replaced with an alanine residue (L368A) (numbering according to Kabat EU index).
- the serine residue at position 354 is replaced with a cysteine residue (S354C) or the glutamic acid residue at position 356 is replaced with a cysteine residue (E356C) (particularly the serine residue at position 354 is replaced with a cysteine residue), and in the second subunit of the Fc domain additionally the tyrosine residue at position 349 is replaced by a cysteine residue (Y349C) (numbering according to Kabat EU index).
- the first subunit of the Fc domain comprises the amino acid substitutions S354C and T366W
- the second subunit of the Fc domain comprises the amino acid substitutions Y349C, T366S, L368A and Y407V (numbering according to Kabat EU index).
- the Fc domain comprises one or more amino acid substitution that reduces binding to an Fc receptor and/or effector function.
- the Fc receptor is an Fey receptor. In one aspect the Fc receptor is a human Fc receptor. In one aspect the Fc receptor is an activating Fc receptor. In a specific aspect the Fc receptor is an activating human Fey receptor, more specifically human Fey RUIa, FcyRI or FcyRIIa, most specifically human FcyRIIIa.
- the effector function is one or more selected from the group of complement dependent cytotoxicity (CDC), antibody-dependent cell-mediated cytotoxicity (ADCC), antibody-dependent cellular phagocytosis (ADCP), and cytokine secretion. In a particular aspect, the effector function is ADCC.
- the same one or more amino acid substitution is present in each of the two subunits of the Fc domain.
- the one or more amino acid substitution reduces the binding affinity of the Fc domain to an Fc receptor.
- the one or more amino acid substitution reduces the binding affinity of the Fc domain to an Fc receptor by at least 2-fold, at least 5-fold, or at least 10-fold.
- the Fc domain comprises an amino acid substitution at a position selected from the group of E233, L234, L235, N297, P331 and P329 (numberings according to Kabat EU index). In a more specific aspect, the Fc domain comprises an amino acid substitution at a position selected from the group of L234, L235 and P329 (numberings according to Kabat EU index). In some aspects, the Fc domain comprises the amino acid substitutions L234A and L235A (numberings according to Kabat EU index). In one such aspect, the Fc domain is an IgGi Fc domain, particularly a human IgGi Fc domain. In one aspect, the Fc domain comprises an amino acid substitution at position P329.
- the amino acid substitution is P329A or P329G, particularly P329G (numberings according to Kabat EU index).
- the Fc domain comprises an amino acid substitution at position P329 and a further amino acid substitution at a position selected from E233, L234, L235, N297 and P331 (numberings according to Kabat EU index).
- the further amino acid substitution is E233P, L234A, L235A, L235E, N297A, N297D or P331S.
- the Fc domain comprises amino acid substitutions at positions P329, L234 and L235 (numberings according to Kabat EU index).
- the Fc domain comprises the amino acid mutations L234A, L235A and P329G (“P329G LALA”, “PGLALA” or “LALAPG”).
- P329G LALA amino acid mutations L234A, L235A and P329G
- P329G LALA amino acid substitutions L234A, L235A and P329G (Kabat EU index numbering), i.e.
- the leucine residue at position 234 is replaced with an alanine residue (L234A)
- the leucine residue at position 235 is replaced with an alanine residue (L235A)
- the proline residue at position 329 is replaced by a glycine residue (P329G) (numbering according to Kabat EU index).
- the Fc domain is an IgGi Fc domain, particularly a human IgGi Fc domain.
- the HLA-A2/WT1 x CD3 bispecific antibody comprises (i) a first antigen binding moiety that specifically binds to CD3, comprising a heavy chain variable region comprising the heavy chain CDR (HCDR) 1 of SEQ ID NO: 1, the HCDR2 of SEQ ID NO: 2, and the HCDR3 of SEQ ID NO: 3; and a light chain variable region comprising the light chain CDR (LCDR) 1 of SEQ ID NO: 4, the LCDR2 of SEQ ID NO: 5 and the LCDR3 of SEQ ID NO: 6, wherein the first antigen binding moiety is a crossover Fab molecule wherein either the variable or the constant regions, particularly the variable regions, of the Fab light chain and the Fab heavy chain are exchanged;
- a second and a third antigen binding moiety that specifically bind to HLA-A2/WT 1 , comprising a heavy chain variable region comprising the heavy chain CDR (HCDR) 1 of SEQ ID NO: 9, the HCDR2 of SEQ ID NO: 10, and the HCDR3 of SEQ ID NO: 11; and a light chain variable region comprising the light chain CDR (LCDR) 1 of SEQ ID NO: 12, the LCDR2 of SEQ ID NO: 13 and the LCDR3 of SEQ ID NO: 14, wherein the second and third antigen binding moiety are each a Fab molecule, particularly a conventional Fab molecule;
- an Fc domain composed of a first and a second subunit, wherein the second antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N-terminus of the Fab heavy chain of the first antigen binding moiety, and the first antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N-terminus of the first subunit of the Fc domain, and wherein the third antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N-terminus of the second subunit of the Fc domain.
- the first antigen binding moiety comprises a heavy chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 7 and a light chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 8.
- the first antigen binding moiety comprises the heavy chain variable region sequence of SEQ ID NO: 7 and the light chain variable region sequence of SEQ ID NO: 8.
- the second and third antigen binding moiety comprise a heavy chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 15 and a light chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 16.
- the second and third antigen binding moieties comprise the heavy chain variable region of SEQ ID NO: 15 and the light chain variable region of SEQ ID NO: 16.
- the Fc domain according to the above aspects may incorporate, singly or in combination, all of the features described hereinabove in relation to Fc domains.
- the antigen binding moieties and the Fc region are fused to each other by peptide linkers, particularly by peptide linkers as in SEQ ID NO: 18 and SEQ ID NO: 20.
- the amino acid at position 124 is substituted by lysine (K) (numbering according to Kabat) and the amino acid at position 123 is substituted by lysine (K) or arginine (R), particularly by arginine (R) (numbering according to Kabat), and in the constant domain CHI of the second and the third Fab molecule under (ii) the amino acid at position 147 is substituted by glutamic acid (E) (numbering according to Kabat EU index) and the amino acid at position 213 is substituted by glutamic acid (E) (numbering according to Kabat EU index).
- the HLA-A2/WT1 x CD3 bispecific antibody comprises a polypeptide (particularly two polypeptides) comprising a sequence that is at least 80%, 85%, 90%, 95%, 96%, 97%, 98%, or 99% identical to the sequence of SEQ ID NO: 17, a polypeptide comprising a sequence that is at least 80%, 85%, 90%, 95%, 96%, 97%, 98%, or 99% identical to the sequence of SEQ ID NO: 18, a polypeptide comprising a sequence that is at least 80%, 85%, 90%, 95%, 96%, 97%, 98%, or 99% identical to the sequence of SEQ ID NO: 19, and a polypeptide comprising a sequence that is at least 80%, 85%, 90%, 95%, 96%, 97%, 98%, or 99% identical to the sequence of SEQ ID NO: 20.
- the HLA-A2/WT1 x CD3 bispecific antibody comprises a polypeptide (particularly two polypeptides) comprising the sequence of SEQ ID NO: 17, a polypeptide comprising the sequence of SEQ ID NO: 18, a polypeptide comprising the sequence of SEQ ID NO: 19, and a polypeptide comprising the sequence of SEQ ID NO: 20.
- the HLA-A2/WT1 x CD3 bispecific antibody herein is used in combination with venetoclax and/or azacitidine, preferably venetoclax and (i.e. plus) azacitidine.
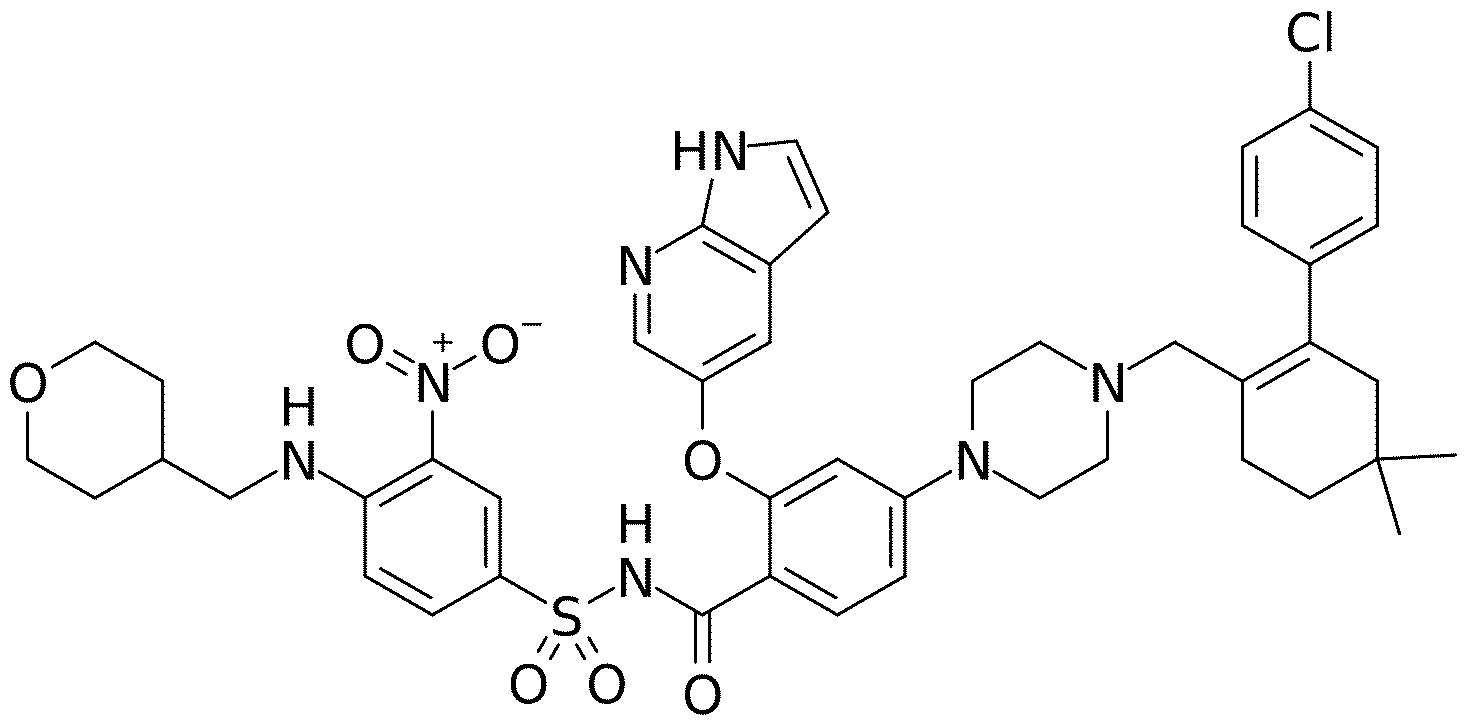
- Venetoclax refers to the compound with the chemical (IUPAC) name 4-[4-[[2-(4-chlorophenyl)-4,4-dimethylcyclohex- 1 -en- 1 -yl]methyl]piperazin- 1 -y 1 ] -N- [3 - nitro-4-(oxan-4-ylmethylamino)phenyl]sulfonyl-2-(17/-pyrrolo[2,3-Z>]pyridin-5-yloxy)benzamide, and the following chemical structure:
- Venetoclax is administered orally, and is available as tablets of 10, 50 or 100 mg venetoclax. It is an inhibitor of the anti-apoptotic B-cell lymphoma-2 (BCL-2) protein. It is, for example, used in the treatment of chronic lymphocytic leukemia (CLL), small lymphocytic leukemia (SLL) or acute myeloid leukemia (AML). In the treatment of AML, it may be used in combination with azacitidine.
- the venetoclax daily dose for AML treatment is typically between 100 mg and 600 mg (between 100 mg and 400 mg when used in combination with azacitidine).
- a particular AML dosing schedule is described hereinbelow.
- azacitidine or “5 -azacitidine” (abbreviated as “Aza” or “5-Aza”) refers to the compound with the chemical (IUPAC) name 4-amino-l-[(2A, 3A,45, 5A)-3,4-dihydroxy-5- (hydroxymethyl)oxolan-2-yl]-l,3,5-triazin-2-one, and the following chemical structure:
- Azacitidine is a cytidine analogue and acts as a nucleid acid synthesis inhibitor (hypomethylation agent).
- Azacitidine is marketed under the tradename VIDAZA® or ONUREG®. It may be administered subcutaneously, intravenously or orally (tablets of 200 mg or 200 mg azacitidine being available). It is used in the treatment of myelodysplastic syndrome. It is also used in the treatment of acute myeloid leukemia (AML). In the treatment of AML, it may be used in combination with venetoclax.
- An azacitidine daily dose for AML treatment is typically 300 mg (orally, once daily).
- an azacitidine daily dose for AML treatment is 75 mg/m2 (intravenously or subcutaneously).
- Azacitidine is typically administered in treatment cycles of 28 days, consisting of a treatment period of 14 days followed by a treatment free period of 14 days.
- nucleic acid synthesis inhibitors that will be known to the skilled practitioner (e.g. decitabine (also known as 5-aza-2'-deoxycytidine; CAS Registry Number 2353-33-5; sold under the trade name DACOGEN®), or cytarabine (also known as cytosine arabinoside (ara-C); CAS Registry Number 147-94-4; sold under the trade name CYTOSAR-U®)) are also contemplated for use in the present invention.
- decitabine also known as 5-aza-2'-deoxycytidine
- CAS Registry Number 2353-33-5 sold under the trade name DACOGEN®
- cytarabine also known as cytosine arabinoside (ara-C); CAS Registry Number 147-94-4; sold under the trade name CYTOSAR-U®
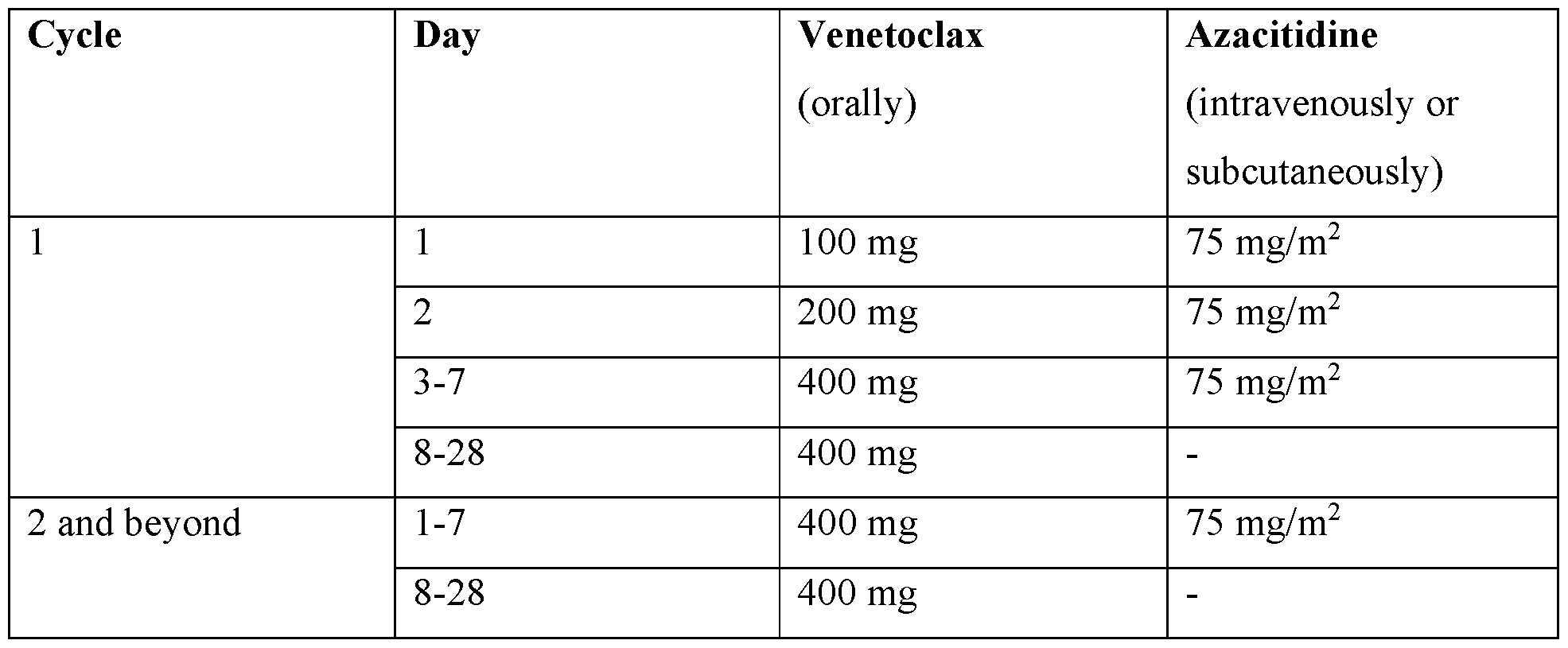
- a dosing schedule for the combination comprises a first 28-day dosing cycle with 100 mg venetoclax on day 1, 200 mg venetoclax on day 2, and 400 mg venetoclax on day 3 to 28, plus 75 mg/m 2 azacitidine on day 1-7, followed by 28- day dosing cycles with 400 mg venetoclax daily plus 75 mg/m 2 azacitidine on day 1-7 of each cycle.
- Such dosing schedule is illustrated in Table 1 below.
- Venetoclax/azazitidine is meant venetoclax and/or azazitidine, preferably venetoclax and (i.e. plus) azazitidine.
- cancer refers to the physiological condition in mammals that is typically characterized by unregulated cell proliferation.
- examples of cancer include but are not limited to, carcinoma, lymphoma, blastoma, sarcoma and leukemia. More non-limiting examples of cancers include haematological cancer such as leukemia, bladder cancer, brain cancer, head and neck cancer, pancreatic cancer, biliary cancer, thyroid cancer, lung cancer, breast cancer, ovarian cancer, uterine cancer, cervical cancer, endometrial cancer, esophageal cancer, colon cancer, colorectal cancer, rectal cancer, gastric cancer, prostate cancer, skin cancer, squamous cell carcinoma, sarcoma, bone cancer, and kidney cancer.
- haematological cancer such as leukemia, bladder cancer, brain cancer, head and neck cancer, pancreatic cancer, biliary cancer, thyroid cancer, lung cancer, breast cancer, ovarian cancer, uterine cancer, cervical cancer, endometrial cancer, esophageal cancer, colon cancer, colorectal
- cell proliferation disorders include, but are not limited to neoplasms located in the: abdomen, bone, breast, digestive system, liver, pancreas, peritoneum, endocrine glands (adrenal, parathyroid, pituitary, testicles, ovary, thymus, thyroid), eye, head and neck, nervous system (central and peripheral), lymphatic system, pelvic, skin, soft tissue, spleen, thoracic region, and urogenital system. Also included are pre-cancerous conditions or lesions and cancer metastases.
- the cancer is a haematological cancer.
- haematological cancers include leukemia (e.g. acute lymphocytic leukemia (ALL), acute myeloid leukemia (AML), chronic lymphcytic leukemia (CLL) chronic myeloid leukemia (CML), hairy cell leukemia (HCL)), lymphoma (e.g. Non-Hodgkin lymphoma (NHL), Hodgkin lymphoma), myeloma (e.g. multiple myeloma (MM)), myelodysplastic syndrome (MDS) and myeloproliferative diseases.
- ALL acute lymphocytic leukemia
- AML acute myeloid leukemia
- CLL chronic lymphcytic leukemia
- HCL hairy cell leukemia
- NHL Non-Hodgkin lymphoma
- NHL Non-Hodgkin lymphoma
- myeloma e.g. multiple myeloma (MM
- the cancer is chosen from the group consisting of haematological cancer (such as leukemia), kidney cancer, bladder cancer, skin cancer, lung cancer, colorectal cancer, breast cancer, brain cancer, head and neck cancer and prostate cancer.
- haematological cancer such as leukemia
- kidney cancer bladder cancer
- skin cancer skin cancer
- lung cancer colorectal cancer
- breast cancer brain cancer
- head and neck cancer prostate cancer.
- the cancer is a haematological cancer, particularly leukemia, most particularly acute lymphocytic leukemia (ALL) or acute myeloid leukemia (AML).
- ALL acute lymphocytic leukemia
- AML acute myeloid leukemia
- the cancer is acute myeloid leukemia (AML).
- the cancer is myelodysplastic syndrome (MDS).
- MDS myelodysplastic syndrome
- the cancer is a WT1 -positive cancer.
- WT1 -positive cancer or “WT1- expressing cancer” is meant a cancer characterized by expression or overexpression of WT1 in cancer cells.
- the expression of WT1 may be determined for example by quantitative real-time PCR (measuring WT1 mRNA levels), immunohistochemistry (IHC) or western blot assays.
- the cancer expresses WT1.
- the cancer expresses WT1 in at least 20%, preferably at least 50% or at least 80% of tumor cells as determined by immunohistochemistry (IHC) using an antibody specific for WT1.
- a “patient”, “subject” or “individual” herein is any single human subject eligible for treatment who is experiencing or has experienced one or more signs, symptoms, or other indicators of cancer.
- the patient has cancer or has been diagnosed with cancer.
- the patient may have been previously treated with a HLA-A2/WT1 x CD3 bispecific antibody or another drug, or not so treated.
- the patient has not been previously treated with a HLA-A2/WT1 x CD3 bispecific antibody.
- the patient may have been treated with a therapy comprising one or more drugs other than a HLA-A2/WT1 x CD3 bispecific antibody before the HLA-A2/WT1 x CD3 bispecific antibody therapy is commenced.
- the patient carries a HLA- A2 allele, in particular the HLA-A*02:01 allele.
- treatment refers to clinical intervention in an attempt to alter the natural course of a disease in the individual being treated, and can be performed either for prophylaxis or during the course of clinical pathology. Desirable effects of treatment include, but are not limited to, preventing occurrence or recurrence of disease, alleviation of symptoms, diminishment of any direct or indirect pathological consequences of the disease, preventing metastasis, decreasing the rate of disease progression, amelioration or palliation of the disease state, and remission or improved prognosis.
- the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine are administered in an effective amount.
- an “effective amount” of an agent refers to an amount effective, at dosages and for periods of time necessary, to achieve the desired therapeutic or prophylactic result.
- administration of the HLA-A2/WT1 x CD3 bispecific antibody results in activation of T cells, particularly cytotoxic T cells, particularly at the site of the cancer.
- Said activation may comprise proliferation of T cells, differentiation of T cells, cytokine secretion by T cells, cytotoxic effector molecule release from T cells, cytotoxic activity of T cells, and expression of activation markers by T cells.
- the administration of the HLA-A2/WT1 x CD3 bispecific antibody results in an increase of T cell, particularly cytotoxic T cell, numbers at the site of the cancer.
- the treatment with or administration of the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine results in increased activation of T cells, particularly cytotoxic T cells, particularly at the site of the cancer, as compared to treatment with or administration of the HLA-A2/WT1 x CD3 bispecific antibody alone.
- the activation comprises cytotoxic activity (specifically lysis of cancer cells) of T cells and/or cytokine (specifically IL-2, TNF-a, and/or interferon-y) secretion by T cells.
- the treatment with or administration of the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine results in reduced cancer growth and/or increased survival time, as compared to treatment with or administration of the HLA-A2/WT1 x CD3 bispecific antibody alone.
- the treatment with or administration of the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine may result in a response in the individual.
- the response may be a complete response.
- the response may be a sustained response after cessation of the treatment.
- the response may be a complete response that is sustained after cessation of the treatment.
- the response may be a partial response.
- the response may be a partial response that is sustained after cessation of the treatment.
- the treatment with or administration of the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine may improve the response as compared to treatment with or administration of the HLA-A2/WT1 x CD3 bispecific antibody alone (i.e. without venetocl ax/ azaciti dine) .
- the treatment or administration of the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine may increase response rates in a patient population, as compared to a corresponding patient population treated with the HLA-A2/WT1 x CD3 bispecific antibody alone (i.e. without venetoclax/azacitidine).
- the combination therapy of the invention comprises administration of a HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine.
- “combination” encompasses combinations of a HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine according to the invention wherein the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine are in the same or in different containers, in the same or in different pharmaceutical formulations, administered together or separately, administered simultaneously or sequentially, in any order, and administered by the same or by different routes, provided that the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine can simultaneously exert their biological effects in the body.
- the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine may be administered in any suitable manner known in the art.
- the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine are administered sequentially (at different times).
- the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine are administered concurrently (at the same time).
- the HLA-A2/WT1 x CD3 bispecific antibody is in a separate composition from venetoclax/azacitidine (wherein venetoclax and azacitidine may each be in separate compositions as well).
- the HLA-A2/WT1 x CD3 bispecific antibody is in the same composition as venetoclax/azacitidine.
- the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine can be administered by any suitable route, and may be administered by the same route of administration or by different routes of administration.
- the HLA-A2/WT1 x CD3 bispecific antibody is administered intravenously, intramuscularly, subcutaneously, topically, orally, transdermally, intraperitoneally, intraorbitally, by implantation, by inhalation, intrathecally, intraventricularly, or intranasally.
- the HLA-A2/WT1 x CD3 bispecific antibody is administrered intravenously.
- venetoclax/azacitidine is administered intravenously, intramuscularly, subcutaneously, topically, orally, transdermally, intraperitoneally, intraorbitally, by implantation, by inhalation, intrathecally, intraventricularly, or intranasally.
- venetoclax/azacitidine in particular venetoclax
- venetoclax/azacitidine is administered orally.
- venetoclax/azacitidine in particular azacitidine
- venetoclax/azacitidine is administered intravenously or subcutaneously.
- venetoclax is administered orally and azacitidine is administered intravenously or subcutaneously.
- An effective amount of the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine may be administered for prevention or treatment of disease.
- the appropriate route of administration and dosage of the HLA-A2/WT1 x CD3 bispecific antibody and/or venetoclax/azacitidine may be determined based on the type of disease to be treated, the type of the HLA-A2/WT1 x CD3 bispecific antibody, the severity and course of the disease, the clinical condition of the individual, the individual’s clinical history and response to the treatment, and the discretion of the attending physician. Dosing can be by any suitable route, e.g. by injections, such as intravenous or subcutaneous injections, depending in part on whether the administration is brief or chronic.
- Various dosing schedules including but not limited to single or multiple administrations over various time-points, bolus administration, and pulse infusion are contemplated herein.
- the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine are suitably administered to the patient at one time or over a series of treatments.
- a typical dosing schedule for venetoclax/azacitidine is described hereinabove.
- Combinations of the invention can be used either alone or together with other agents in a therapy.
- a combination of the invention may be co-administered with at least one additional therapeutic agent.
- an additional therapeutic agent is an anti-cancer agent, e.g. a chemotherapeutic agent, an inhibitor of tumor cell proliferation, or an activator of tumor cell apoptosis.
- Combinations of the invention can also be combined with radiation therapy.
- a kit as provided herein typically comprises one or more container and a label or package insert on or associated with the container.
- Suitable containers include, for example, bottles, vials, syringes, IV solution bags, etc.
- the containers may be formed from a variety of materials such as glass or plastic.
- the container holds a composition which is by itself or combined with another composition effective for treating, preventing and/or diagnosing the condition and may have a sterile access port (for example the container may be an intravenous solution bag or a vial having a stopper pierceable by a hypodermic injection needle).
- At least one active agent in the composition is a HLA-A2/WT1 x CD3 bispecific antibody to be used in the combinations of the invention.
- Another active agent is venetoclax/azacitidine to be used in the combinations of the invention, which may be in the same composition and container like the bispecific antibody, or may be provided in a different composition and container.
- the label or package insert indicates that the composition(s) is/are used for treating the condition of choice, such as cancer.
- the invention provides a kit intended for the treatment of cancer, comprising in the same or in separate containers (a) a HLA-A2/WT1 x CD3 bispecific antibody, and (b) venetoclax/azacitidine, and optionally further comprising (c) a package insert comprising printed instructions directing the use of the combined treatment as a method for treating cancer.
- the kit may comprise (a) a first container with a composition contained therein, wherein the composition comprises a HLA-A2/WT1 x CD3 bispecific antibody; (b) a second and/or a third container with a composition contained therein, wherein the composition comprises venetoclax/azacitidine; and optionally (c) a fourth container with a composition contained therein, wherein the composition comprises a further cytotoxic or otherwise therapeutic agent.
- the kit in these aspects of the invention may further comprise a package insert indicating that the compositions can be used to treat cancer.
- the kit may further comprise a third (or fourth) container comprising a pharmaceutically-acceptable buffer, such as bacteriostatic water for injection (BWFI), phosphate-buffered saline, Ringer's solution and dextrose solution. It may further include other materials desirable from a commercial and user standpoint, including other buffers, diluents, filters, needles, and syringes.
- a pharmaceutically-acceptable buffer such as bacteriostatic water for injection (BWFI), phosphate-buffered saline, Ringer's solution and dextrose solution.
- FIG. 1 Schematic illustration of the HLA-A2/WT1 -targeted T-cell bispecific (TCB) antibody molecule used in the Examples (“WT1-TCB”).
- the molecule comprises a single antigen binding moiety for CD3, two antigen binding sites for HLA-A2/WT1, and an Fc domain.
- Statistical test Two-way ANOVA with Tukey’s multiple comparisons test. *p ⁇ 0.05, **** ? ⁇ 0.0001.
- (B) T-cell expansion after 6 days of coculture calculated as fold-change compared to the start of the coculture (n 3). Bars represent mean ⁇ SEM.
- (B) T-cell expansion in after 6 days of coculture with OCI-AML3 cells calculated as fold-change compared to the start of the coculture (n 9). Bars represent mean ⁇ SEM.
- B Detailed representation of the data from (A) for all patients. Of note, one patient showed overall a low specific lysis that was further decreased upon Ven-Aza pre-treatment.
- FIG. 7 Schematic overview of the study design of Example 2.
- AZA azacytidine
- d day
- NSG NOD.Cg-Prkdcscid I12rgtmlWjl/SzJ
- Ven Venetoclax.
- FIG. 8 In vivo tumor growth curves (on therapy day 36).
- FIG. 9 In vivo Kaplan-Meier curve.
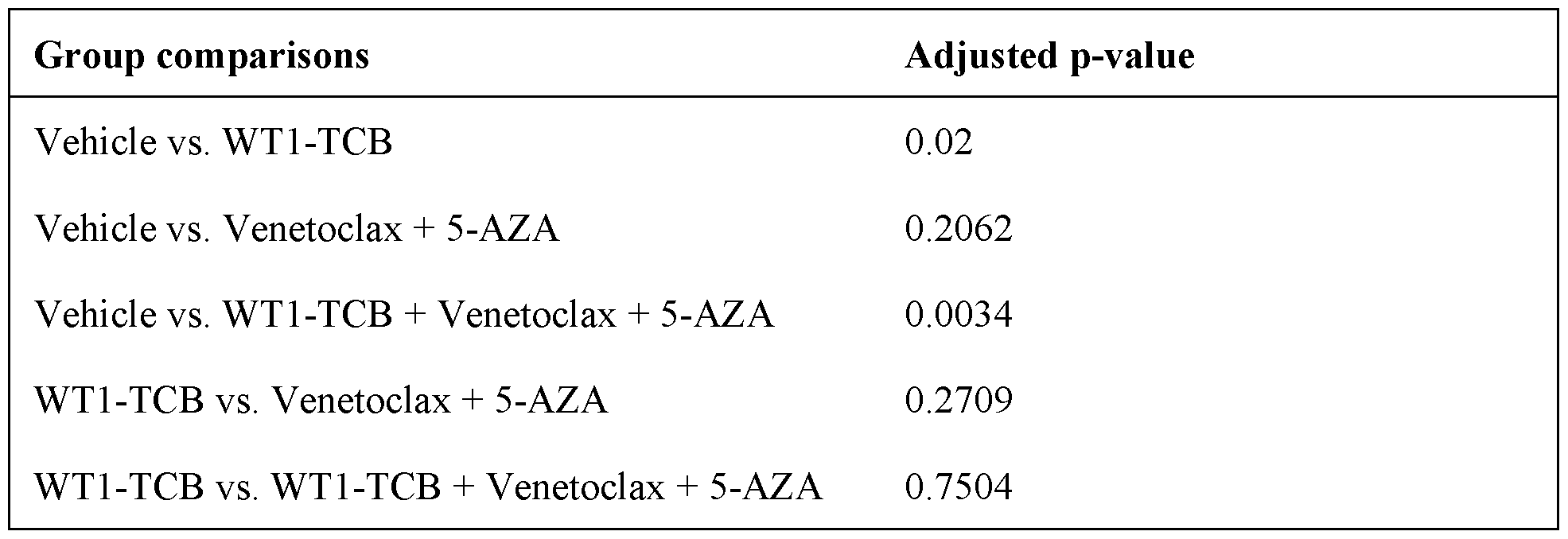
- P-values on Study Day 36 were calculated using the log-rank test adjusted for multiple testing and are shown in Table 4.
- 5-AZA 5-azacytidine
- IP intraperitoneally
- IV intravenously
- PO orally.
- Luminescence was induced by using the Bright-Glo Luciferase Assay System (Promega, Fitchburg, WI) and measured on an Infinite M1000 Plate Reader (Tecan, Mannedorf, Switzerland).
- Lysis of AML cell lines by the combination of WT1-TCB and Ven/ Aza was analyzed in vitro.
- LOxlO 5 SKM-1 cells or OCI-AML cells (both HLA-A*O2 + /WT1 + ) were cocultured with 5.0xl0 4 healthy donor T cells (negatively isolated using Human T cell isolation kit; Stemcell, Vancouver, Canada) in a 96-well plate in the presence of 25 nM Venetoclax and 1 pM Azacytidine (both Selleckchem, Houston, TX). Azacytidine was replenished after 24 and 48 h and Venetoclax after 72 h assuming full degradation.
- WT1-TCB was added at 0.01 pg/mL (SKM-1) or 1 pg/mL (OCL AML3) at the start of the experiment and replenished after 72 h assuming full consumption (parallel dosing), or was only added after 72 h (sequential dosing).
- DMSO and the non-targeted control antibody construct (DP47-TCB) were included as controls, as well as an untreated control without WT1-TCB, Venetoclax and Azacytidine. Cocultures were analyzed after six days for overall lysis of AML cell lines and expansion of T cells.
- pAML Primary AML cells from HLA-A*02 + patients were collected at initial diagnosis after written informed consent in accordance with the Declaration of Helsinki and approval granted by the Institutional Review Board of the Ludwig-Maximilian-Universitat (Munich, Germany).
- pAML cells were pre-cultivated in a twelve well-plate at a concentration of 1.5xl0 6 cells/mL for 2-3 days on a feeder layer of 1.2xl0 5 murine MS5 cells (irradiated with 60 Gy) per well and subsequently enriched by depletion of T cells [using CD3 MicroBeads, human (Miltenyi Biotec)].
- WT1-TCB mediated lysis of pAML cells was analyzed after in vitro-pretreatment of pAML cells with Ven/ Aza.
- pAML cells were cultivated on irradiated MS5 cells and were left untreated or were pretreated for seven days with 1 pM Azacytidine (daily addition on days 1-5) and 300 nM Ven (added on day 5).
- T cells and pAML cells were subsequently cocultivated for 3 days at an E:T ratio of 1 : 1 with various concentrations of WT1-TCB (0.01-10 pg/mL) and specific lysis of AML cells was calculated relative to corresponding controls with DP47-TCB based on viable AML cell counts.
- pAML cells were co-cultured with healthy donor T cells for 7 days at an E:T ratio of 1 :2 on a layer of MS5 cells in the presence of 25 nM Ven, 1 pM Aza.
- Azacytidine was replenished after 24 and 48 h and Venetoclax after 72 h assuming full degradation.
- WT1-TCB was added at 1 pg/mL at the start of the experiment and replenished after 72 h assuming full consumption DMSO and the non-targeted control antibody construct (DP47-TCB) were included as controls, as well as an untreated control without WT1-TCB, Venetoclax and Azacytidine. Overall lysis was assessed by flow cytometry on day 6 and calculated relative to the untreated control.
- Target antigen expression on AML cell lines was measured in response to stimulation with Ven/ Aza.
- WT1-TCB induced TCR downstream signaling in cocultures with SKM-1 and OCI- AML3 cells, as measured by luminescence induced in NF AT Luciferase Reporter Jurkat cells.
- WT1-TCB mediated lysis in combination with Ven/ Aza was furthermore tested in vitro in cocultures of HD T cells and OCI-AML3 cells (Figure 4) for six days in the presence of 1 pg/mL WT1-TCB, 25 nM Ven and 1 pM Aza.
- WT1-TCB mediated lysis by HD T cells was furthermore analyzed in combination with pAML cells.
- a reduction in cell lysis by Ven-Aza pretreatment was only observed for one patient sample whereas no differences were observed for the other two patient samples between with and without Ven-Aza pretreatment (Figure 5B).
- mice Humanized HLA-A*02-positive mice were injected with SKM-1 tumor cells. After tumors reached an average tumor size of 198 mm 3 , mice were treated with WT1-TCB (0.05 mg/kg), Venetoclax (50 mg/kg; Sigma Aldrich) plus 5-AZA (1 mg/kg; Sigma Aldrich), or the combination of these compounds.
- mice humanized using an HLA-A*02 + (CD34 + ) hematopoietic stem cell donor were purchased from Jackson Laboratories (Bar Harbor, ME). Mice were aged 16-26 weeks at the start of the experiments and were maintained under specific-pathogen-free conditions, with 12: 12 h light/dark daily cycles, in accordance to the required local and federal guidelines and laws (GVSolas; Felasa; TierschG). The experimental study protocol was reviewed and approved by local government authorities (approval number ROB-55.2-2532. Vet_02-20-170). After arrival, animals were maintained for 1 week to get accustomed to the new environment and for observation. Continuous health monitoring was carried out daily.
- mice Humanized NSG mice were injected subcutaneously (SC) with 10 x 10 6 SKM-1 tumor cells (Catalog No. ACC547; DSMZ) on Study Day 0. Mice were randomized in 4 different study groups (Table 2). Treatment of humanized mice with the test compounds started 14 days after tumor-cell injection, when tumors reached an average size of 198 mm 3 , and lasted 4 weeks. Mice were dosed with compounds according to their body weight with a volume of 10 mL/kg, as described in the following.
- mice received vehicle as control (for vehicle treatment, see Table 2).
- the second group received WT1-TCB intravenously at 0.05 mg/kg once weekly.
- the third group received histidine buffer intravenously once weekly and at the following 5 days Venetoclax at 50 mg/kg orally plus 5-AZA at 1 mg/kg intraperitoneally for the first week. In the following 3 weeks, this treatment group received only vehicle plus Venetoclax.
- the fourth treatment group received WT1- TCB plus Venetoclax plus 5-AZA at the same dosing schedule as described for Groups 2 and 3. Table 2. Study groups.
- Tumor volume was measured by caliper every second day. Tumor growth curves were plotted in GraphPad Prism (version 8.4.2; San Diego, CA). P-values for group comparisons were calculated using the non-parametric Dunn’s test. Pairwise group comparison of the survival data was performed using the log-rank test, which places more weight on later survival events and is adjusted for multiple testing.
- 5-AZA 5-azacytidine
- 5-AZA 5-azacytidine
- the aim of this study was to examine the in vivo antitumor activity of WT1-TCB in combination with Venetoclax plus 5-AZA, a scheme used currently as standard of care for acute myeloid leukemia.
- WT1-TCB was used at a dose of 0.05 mg/kg to provide a therapeutic window for the drug combination effect.
- Tumor growth and survival analysis showed significant antitumor activity for WT1-TCB in combination with Venetoclax plus 5-AZA when compared with vehicle on Study Day 36.
- an improved efficacy was observed when compared with WT1-TCB monotherapy at 0.05 mg/kg.
Landscapes
- Health & Medical Sciences (AREA)
- Immunology (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Organic Chemistry (AREA)
- Molecular Biology (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Epidemiology (AREA)
- Peptides Or Proteins (AREA)
Abstract
The present invention relates to the treatment of cancer, in particular to the treatment of cancer using a HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine.
Description
Treatment of cancer using a HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine
Field of the Invention
The present invention relates to the treatment of cancer, in particular to the treatment of cancer using a HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine.
Background
T-cell activating bispecific antibodies are a promising class of cancer therapeutics, designed to engage cytotoxic T cells against tumor cells. The simultaneous binding of such an antibody to CD3 on T-cells and to an antigen expressed on the tumor cells will force a temporary interaction between tumor cell and T cell, causing activation of the T-cell and subsequent lysis of the tumor cell.
WT1 (Wilms tumor 1, Wilms tumor protein) is an oncogenic transcription factor involved in cell proliferation, differentiation, as well as apoptosis and organ development, whose expression in normal adult tissue is rare (Hinrichs and Restifo, Nat Biotechnol (2013) 31, 999-1008). WT1 is, however, reported to be overexpressed in several types of haematological maligancies and a wide range of solid tumors (Van Driessche et al., Oncologist (2012) 17, 250-259). WT1 is a nuclear protein, localized intracellularly. Intracellular protein can be degraded in the proteasome, processed and presented on the cell surface by major histocompatibility complex (MHC) I as T cell epitopes, and recognized by T cell receptors (TCR). As such, WT1 -derived peptides are presented in the context of HLA-A2 on the cell surface and can trigger T cell recognition.
T-cell activating bispecific antibodies targeting HLA-A2/WT1 have been described in WO 2019/122052. Such T-cell activating bispecific antibodies may be useful, e.g., in the treatment of acute myeloid leukemia (AML). A HLA-A2/WT1 x CD3 bispecific antibody (“WT1-TCB”) is currently under investigation in a phase I clinical trial in patients with relap sed/refractory (r/r) AML (Augsberger et al., Blood (2021) doi: 10.1182/blood.2020010477; NCT04580121).
In order to maximize the therapeutic benefit of HLA-A2/WT1 -targeting T-cell activating antibodies, e.g. in AML, it would thus be desirable to identify combination treatments involving such T-cell activating antibodies and other therapeutic agents.
Description of the Invention
The present inventors have found that combination of HLA-A2/WT1 targeted T-cell activating bispecific antibodies with venetoclax/azacitidine leads to enhanced activity as compared to HLA- A2/WT1 targeted T-cell activated bispecific antibody alone.
In particular, the inventors have surprisingly found that tumor cell lysis in vitro, as well as antitumor efficacy in vivo, induced by HLA-A2/WT1 x CD3 bispecific antibody was enhanced by the addition of venetoclax/azacitidine.
Accordingly, in a first aspect, the present invention provides a HLA-A2/WT1 x CD3 bispecific antibody for use in the treatment of a cancer in an individual, wherein the treatment comprises administration of the HLA-A2/WT1 x CD3 bispecific antibody in combination with venetocl ax/ azaciti dine .
In a further aspect, the invention provides the use of a HLA-A2/WT1 x CD3 bispecific antibody in the manufacture of a medicament for the treatment of cancer in an individual, wherein the treatment comprises administration of the HLA-A2/WT1 x CD3 bispecific antibody in combination with venetoclax/azacitidine.
In still a further aspect, the invention provides a method for treating cancer in an individual comprising administering to the individual a HLA-A2/WT1 x CD3 bispecific antibody and venetocl ax/ azaciti dine .
In one aspect, the invention also provides a kit comprising a first medicament comprising a HLA- A2/WT1 x CD3 bispecific antibody, and a second medicament comprising venetoclax and/or a third medicament comprising azacitidine, and optionally further comprising a package insert comprising instructions for administration of the first medicament in combination with the second and/or the third medicament for treating cancer in an individual.
The HLA-A2/WT1 x CD3 bispecific antibodies, methods, uses or kits described above and herein, may incorporate, singly or in combination, any of the features described in the following (unless the context dictates otherwise).
The HLA-A2/WT1 x CD3 bispecific antibody herein is a bispecific antibody that specifically binds to CD3 and to HLA-A2/WT1, particularly HLA-A2/WT1 RMF. Particularly useful HLA- A2/WT1 x CD3 bispecific antibodies for use in the present invention are described e.g. in PCT publication no. WO 2019/122052 (incorporated herein by reference in its entirety).
The term “bispecific” means that the antibody is able to specifically bind to at least two distinct antigenic determinants. Typically, a bispecific antibody comprises two antigen binding sites, each of which is specific for a different antigenic determinant. In certain aspects, the bispecific antibody is capable of simultaneously binding two antigenic determinants, particularly two antigenic determinants expressed on two distinct cells.
As used herein, the term "antigenic determinant" is synonymous with "antigen" and "epitope", and refers to a site (e.g. a contiguous stretch of amino acids or a conformational configuration made up of different regions of non-contiguous amino acids) on a polypeptide macromolecule to which an antigen binding moiety binds, forming an antigen binding moiety-antigen complex. Useful antigenic determinants can be found, for example, on the surfaces of tumor cells, on the surfaces of virus-infected cells, on the surfaces of other diseased cells, on the surface of immune cells, free in blood serum, and/or in the extracellular matrix (ECM).
As used herein, the term "antigen binding moiety" refers to a polypeptide molecule that specifically binds to an antigenic determinant. In one aspect, an antigen binding moiety is able to direct the entity to which it is attached (e.g. a second antigen binding moiety) to a target site, for example to a specific type of tumor cell bearing the antigenic determinant. In another aspect an antigen binding moiety is able to activate signaling through its target antigen, for example a T cell receptor complex antigen. Antigen binding moieties include antibodies and fragments thereof as further defined herein. Particular antigen binding moieties include an antigen binding domain of an antibody, comprising an antibody heavy chain variable region and an antibody light chain variable region. In certain aspects, the antigen binding moieties may comprise antibody constant regions as further defined herein and known in the art. Useful heavy chain constant regions include any of the five isotypes: a, 5, a, y, or p. Useful light chain constant regions include any of the two isotypes: K and .
By "specific binding" is meant that the binding is selective for the antigen and can be discriminated from unwanted or non-specific interactions. The ability of an antigen binding moiety to bind to a specific antigenic determinant can be measured either through an enzyme-linked immunosorbent
assay (ELISA) or other techniques familiar to one of skill in the art, e.g. surface plasmon resonance (SPR) technique (analyzed e.g. on a BIAcore instrument) (Liljeblad et al., Glyco J 17, 323-329 (2000)), and traditional binding assays (Heeley, Endocr Res 28, 217-229 (2002)). In one aspect, the extent of binding of an antigen binding moiety to an unrelated protein is less than about 10% of the binding of the antigen binding moiety to the antigen as measured, e.g., by SPR. In certain aspects, an antigen binding moiety that binds to the antigen, or an antibody comprising that antigen binding moiety, has a dissociation constant (KD) of < 1 pM, < 100 nM, < 10 nM, < 1 nM, < 0.1 nM, < 0.01 nM, or < 0.001 nM (e.g. 10'8 M or less, e.g. from 10'8 M to 10'13 M, e.g., from 10'9 M to 10’13 M).
“Affinity” refers to the strength of the sum total of non-covalent interactions between a single binding site of a molecule (e.g., a receptor) and its binding partner (e.g., a ligand). Unless indicated otherwise, as used herein, “binding affinity” refers to intrinsic binding affinity which reflects a 1 : 1 interaction between members of a binding pair (e.g., an antigen binding moiety and an antigen, or a receptor and its ligand). The affinity of a molecule X for its partner Y can generally be represented by the dissociation constant (KD), which is the ratio of dissociation and association rate constants (koir and kon, respectively). Thus, equivalent affinities may comprise different rate constants, as long as the ratio of the rate constants remains the same. Affinity can be measured by well established methods known in the art, including those described herein. A particular method for measuring affinity is Surface Plasmon Resonance (SPR).
“CD3” refers to any native CD3 from any vertebrate source, including mammals such as primates (e.g. humans), non-human primates (e.g. cynomolgus monkeys) and rodents (e.g. mice and rats), unless otherwise indicated. The term encompasses “full-length,” unprocessed CD3 as well as any form of CD3 that results from processing in the cell. The term also encompasses naturally occurring variants of CD3, e.g., splice variants or allelic variants. In one aspect, CD3 is human CD3, particularly the epsilon subunit of human CD3 (CD3s). The amino acid sequence of human CD3s is shown in UniProt (www.uniprot.org) accession no. P07766 (version 144), or NCBI (www.ncbi.nlm.nih.gov/) RefSeq NP_000724.1. See also SEQ ID NO: 27. The amino acid sequence of cynomolgus [Macaca fascicularis] CD3s is shown in NCBI GenBank no. BAB71849.1. See also SEQ ID NO: 28.
“WT1”, also known as “Wilms tumor 1” or “Wilms tumor protein”, refers to any native WT1 from any vertebrate source, including mammals such as primates (e.g. humans), non-human primates (e.g. cynomolgus monkeys) and rodents (e.g. mice and rats), unless otherwise indicated. The term
encompasses “full-length,” unprocessed WT1 as well as any form of WT1 that results from processing in the cell. The term also encompasses naturally occurring variants of WT1, e.g., splice variants or allelic variants. In one aspect, WT1 is human WT1, particularly the protein of SEQ ID NO: 23. Human WT1 is described in UniProt (www.uniprot.org) accession no. P19544 (entry version 215), and an amino acid sequence of human WT1 is also shown in SEQ ID NO: 23.
By “VLD”, “VLD peptide” or “WTIVLD” is meant the WT1 derived peptide having the amino acid sequence VLDFAPPGA (SEQ ID NO: 24; position 37-45 of the WT1 protein of SEQ ID NO: 23).
By “RMF”, “RMF peptide” or “WTIRMF” is meant the WT1 derived peptide having the amino acid sequence RMFRNAPYL (SEQ ID NO: 25; position 126-134 of the WT1 protein of SEQ ID NO: 23).
“HLA-A2”, “HLA-A*02”, “HLA-A02”, or “HLA-A*2” (used interchangeably) refers to a human leukocyte antigen serotype in the HL A- A serotype group. The HLA-A2 protein (encoded by the respective HL A gene) constitutes the a chain of the respective class I MHC (major histocompatibility complex) protein, which further comprises a P2 microglobulin subunit. A specific HLA-A2 protein is HLA-A201 (also referred to as HLA-A0201, HLA-A02.01, or HLA- A*02:01). In specific aspects, the HLA-A2 protein described herein is HLA-A201. An exemplary sequence of human HLA-A2 is given in SEQ ID NO: 26.
“HLA-A2/WT1” refers to a complex of a HLA-A2 molecule and a WT1 derived peptide (also referred to herein as a “WT1 peptide”), specifically the RMF or VLD peptide (“HLA-A2/WT1RMF” and “HLA-A2/WT1 VLD”, respectively). The bispecific antibody used in the present invention specifically may bind to either the HLA-A2/WT1RMF or the HLA-A2/WT1VLD complex.
As used herein, the terms “first”, “second” or “third” with respect to Fab molecules etc., are used for convenience of distinguishing when there is more than one of each type of moiety. Use of these terms is not intended to confer a specific order or orientation of the bispecific antibody unless explicitly so stated.
The term “valent” as used herein denotes the presence of a specified number of antigen binding sites in an antibody. As such, the term “monovalent binding to an antigen” denotes the presence of one (and not more than one) antigen binding site specific for the antigen in the antibody.
The term "antibody" herein is used in the broadest sense and encompasses various antibody structures, including but not limited to monoclonal antibodies, polyclonal antibodies, multispecific antibodies (e.g. bispecific antibodies), and antibody fragments so long as they exhibit the desired antigen-binding activity.
The terms “full length antibody,” “intact antibody,” and “whole antibody” are used herein interchangeably to refer to an antibody having a structure substantially similar to a native antibody structure.
An "antibody fragment" refers to a molecule other than an intact antibody that comprises a portion of an intact antibody that binds the antigen to which the intact antibody binds. Examples of antibody fragments include but are not limited to Fv, Fab, Fab', Fab’-SH, F(ab')2, diabodies, linear antibodies, single-chain antibody molecules (e.g. scFv), and single-domain antibodies. For a review of certain antibody fragments, see Hudson et al., Nat Med 9, 129-134 (2003). For a review of scFv fragments, see e.g. Pliickthun, in The Pharmacology of Monoclonal Antibodies, vol. 113, Rosenburg and Moore eds., Springer- Verlag, New York, pp. 269-315 (1994); see also WO 93/16185; and U.S. Patent Nos. 5,571,894 and 5,587,458. For discussion of Fab and F(ab')2 fragments comprising salvage receptor binding epitope residues and having increased in vivo halflife, see U.S. Patent No. 5,869,046. Diabodies are antibody fragments with two antigen-binding sites that may be bivalent or bispecific. See, for example, EP 404,097; WO 1993/01161; Hudson et al., Nat Med 9, 129-134 (2003); and Hollinger et al., Proc Natl Acad Sci USA 90, 6444-6448 (1993). Triabodies and tetrabodies are also described in Hudson et al., Nat Med 9, 129-134 (2003). Single-domain antibodies are antibody fragments comprising all or a portion of the heavy chain variable domain or all or a portion of the light chain variable domain of an antibody. In certain aspects, a single-domain antibody is a human single-domain antibody (Domantis, Inc., Waltham, MA; see e.g. U.S. Patent No. 6,248,516 Bl). Antibody fragments can be made by various techniques, including but not limited to proteolytic digestion of an intact antibody as well as production by recombinant host cells (e.g. E. coli or phage), as described herein.
The term “variable region” or “variable domain” refers to the domain of an antibody heavy or light chain that is involved in binding the antibody to antigen. The variable domains of the heavy chain and light chain (VH and VL, respectively) of a native antibody generally have similar structures, with each domain comprising four conserved framework regions (FRs) and three hypervariable regions (HVRs). See, e.g., Kindt et al., Kuby Immunology, 6th ed., W.H. Freeman and Co., page 91 (2007). A single VH or VL domain may be sufficient to confer antigen-binding specificity. As
used herein in connection with variable region sequences, "Kabat numbering" refers to the numbering system set forth by Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, MD (1991).
As used herein, the amino acid positions of all constant regions and domains of the heavy and light chain are numbered according to the Kabat numbering system described in Kabat, et al., Sequences of Proteins of Immunological Interest, 5th ed., Public Health Service, National Institutes of Health, Bethesda, MD (1991), referred to as “numbering according to Kabat” or “Kabat numbering” herein. Specifically the Kabat numbering system (see pages 647-660 of Kabat, et al., Sequences of Proteins of Immunological Interest, 5th ed., Public Health Service, National Institutes of Health, Bethesda, MD (1991)) is used for the light chain constant domain CL of kappa and lambda isotype and the Kabat EU index numbering system (see pages 661-723) is used for the heavy chain constant domains (CHI, Hinge, CH2 and CH3), which is herein further clarified by referring to “numbering according to Kabat EU index” in this case.
The term “hypervariable region” or “HVR”, as used herein, refers to each of the regions of an antibody variable domain which are hypervariable in sequence and which determine antigen binding specificity, for example “complementarity determining regions” (“CDRs”). Generally, antibodies comprise six CDRs; three in the VH (HCDR1, HCDR2, HCDR3), and three in the VL (LCDR1, LCDR2, LCDR3). Exemplary CDRs herein include:
(a) hypervariable loops occurring at amino acid residues 26-32 (LI), 50-52 (L2), 91-96 (L3), 26-32 (Hl), 53-55 (H2), and 96-101 (H3) (Chothia and Lesk, J. Mol. Biol. 196:901-917 (1987));
(b) CDRs occurring at amino acid residues 24-34 (LI), 50-56 (L2), 89-97 (L3), 31-35b (Hl), 50-65 (H2), and 95-102 (H3) (Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, MD (1991)); and
(c) antigen contacts occurring at amino acid residues 27c-36 (LI), 46-55 (L2), 89-96 (L3), 30-35b (Hl), 47-58 (H2), and 93-101 (H3) (MacCallum et al. J. Mol. Biol. 262: 732-745 (1996)). Unless otherwise indicated, the CDRs are determined according to Kabat et al., supra. One of skill in the art will understand that the CDR designations can also be determined according to Chothia, supra, McCallum, supra, or any other scientifically accepted nomenclature system.
"Framework" or "FR" refers to variable domain residues other than hypervariable region (HVR) residues. The FR of a variable domain generally consists of four FR domains: FR1, FR2, FR3, and
FR4. Accordingly, the HVR and FR sequences generally appear in the following order in VH (or VL) : FR1 -H 1 (L 1 )-FR2-H2(L2)-FR3 -H3 (L3 )-FR4.
The “class” of an antibody or immunoglobulin refers to the type of constant domain or constant region possessed by its heavy chain. There are five major classes of antibodies: IgA, IgD, IgE, IgG, and IgM, and several of these may be further divided into subclasses (isotypes), e.g., IgGi, IgG2, IgGs, IgG4, IgAi, and IgA2. The heavy chain constant domains that correspond to the different classes of immunoglobulins are called a, 5, a, y, and p, respectively.
A “Fab molecule” refers to a protein consisting of the VH and CHI domain of the heavy chain (the “Fab heavy chain”) and the VL and CL domain of the light chain (the “Fab light chain”) of an immunoglobulin.
By a “crossover” Fab molecule (also termed “Crossfab”) is meant a Fab molecule wherein the variable domains or the constant domains of the Fab heavy and light chain are exchanged (i.e. replaced by each other), i.e. the crossover Fab molecule comprises a peptide chain composed of the light chain variable domain VL and the heavy chain constant domain 1 CHI (VL-CH1, in N- to C-terminal direction), and a peptide chain composed of the heavy chain variable domain VH and the light chain constant domain CL (VH-CL, in N- to C-terminal direction). For clarity, in a crossover Fab molecule wherein the variable domains of the Fab light chain and the Fab heavy chain are exchanged, the peptide chain comprising the heavy chain constant domain 1 CHI is referred to herein as the “heavy chain” of the (crossover) Fab molecule. Conversely, in a crossover Fab molecule wherein the constant domains of the Fab light chain and the Fab heavy chain are exchanged, the peptide chain comprising the heavy chain variable domain VH is referred to herein as the “heavy chain” of the (crossover) Fab molecule.
In contrast thereto, by a “conventional” Fab molecule is meant a Fab molecule in its natural format, i.e. comprising a heavy chain composed of the heavy chain variable and constant domains (VH- CH1, in N- to C-terminal direction), and a light chain composed of the light chain variable and constant domains (VL-CL, in N- to C-terminal direction).
The term “immunoglobulin molecule” refers to a protein having the structure of a naturally occurring antibody. For example, immunoglobulins of the IgG class are heterotetrameric glycoproteins of about 150,000 daltons, composed of two light chains and two heavy chains that are disulfide-bonded. From N- to C-terminus, each heavy chain has a variable domain (VH), also
called a variable heavy domain or a heavy chain variable region, followed by three constant domains (CHI, CH2, and CH3), also called a heavy chain constant region. Similarly, from N- to C -terminus, each light chain has a variable domain (VL), also called a variable light domain or a light chain variable region, followed by a constant light (CL) domain, also called a light chain constant region. The heavy chain of an immunoglobulin may be assigned to one of five types, called a (IgA), 5 (IgD), 8 (IgE), y (IgG), or p (IgM), some of which may be further divided into subtypes, e.g. yi (IgGi), 72 (IgG?), 73 (IgGi), 74 (IgG4), ai (IgAi) and a? (IgA?). The light chain of an immunoglobulin may be assigned to one of two types, called kappa (K) and lambda (X), based on the amino acid sequence of its constant domain. An immunoglobulin essentially consists of two Fab molecules and an Fc domain, linked via the immunoglobulin hinge region.
The term “Fc domain” or “Fc region” herein is used to define a C-terminal region of an immunoglobulin heavy chain that contains at least a portion of the constant region. The term includes native sequence Fc regions and variant Fc regions. Although the boundaries of the Fc region of an IgG heavy chain might vary slightly, the human IgG heavy chain Fc region is usually defined to extend from Cys226, or from Pro230, to the carboxyl-terminus of the heavy chain. However, antibodies produced by host cells may undergo post-translational cleavage of one or more, particularly one or two, amino acids from the C-terminus of the heavy chain. Therefore an antibody produced by a host cell by expression of a specific nucleic acid molecule encoding a full- length heavy chain may include the full-length heavy chain, or it may include a cleaved variant of the full-length heavy chain. This may be the case where the final two C-terminal amino acids of the heavy chain are glycine (G446) and lysine (K447, numbering according to Kabat EU index). Therefore, the C-terminal lysine (Lys447), or the C-terminal glycine (Gly446) and lysine (K447), of the Fc region may or may not be present. Unless otherwise specified herein, numbering of amino acid residues in the Fc region or constant region is according to the EU numbering system, also called the EU index, as described in Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, MD, 1991 (see also above). A “subunit” of an Fc domain as used herein refers to one of the two polypeptides forming the dimeric Fc domain, i.e. a polypeptide comprising C-terminal constant regions of an immunoglobulin heavy chain, capable of stable self-association. For example, a subunit of an IgG Fc domain comprises an IgG CH2 and an IgG CH3 constant domain.
A “modification promoting the association of the first and the second subunit of the Fc domain” is a manipulation of the peptide backbone or the post-translational modifications of an Fc domain
subunit that reduces or prevents the association of a polypeptide comprising the Fc domain subunit with an identical polypeptide to form a homodimer. A modification promoting association as used herein particularly includes separate modifications made to each of the two Fc domain subunits desired to associate (i.e. the first and the second subunit of the Fc domain), wherein the modifications are complementary to each other so as to promote association of the two Fc domain subunits. For example, a modification promoting association may alter the structure or charge of one or both of the Fc domain subunits so as to make their association sterically or electrostatically favorable, respectively. Thus, (hetero)dimerization occurs between a polypeptide comprising the first Fc domain subunit and a polypeptide comprising the second Fc domain subunit, which might be non-identical in the sense that further components fused to each of the subunits (e.g. antigen binding moieties) are not the same. In some aspects the modification promoting association comprises an amino acid mutation in the Fc domain, specifically an amino acid substitution. In a particular aspect, the modification promoting association comprises a separate amino acid mutation, specifically an amino acid substitution, in each of the two subunits of the Fc domain.
The term “effector functions” refers to those biological activities attributable to the Fc region of an antibody, which vary with the antibody isotype. Examples of antibody effector functions include: Clq binding and complement dependent cytotoxicity (CDC), Fc receptor binding, antibody-dependent cell-mediated cytotoxicity (ADCC), antibody-dependent cellular phagocytosis (ADCP), cytokine secretion, immune complex-mediated antigen uptake by antigen presenting cells, down regulation of cell surface receptors (e.g. B cell receptor), and B cell activation.
“Percent (%) amino acid sequence identity" with respect to a reference polypeptide sequence is defined as the percentage of amino acid residues in a candidate sequence that are identical with the amino acid residues in the reference polypeptide sequence, after aligning the sequences and introducing gaps, if necessary, to achieve the maximum percent sequence identity, and not considering any conservative substitutions as part of the sequence identity. Alignment for purposes of determining percent amino acid sequence identity can be achieved in various ways that are within the skill in the art, for instance, using publicly available computer software such as BLAST, BLAST-2, Clustal W, Megalign (DNASTAR) software or the FASTA program package. Those skilled in the art can determine appropriate parameters for aligning sequences, including any algorithms needed to achieve maximal alignment over the full length of the sequences being compared. For purposes herein, however, % amino acid sequence identity values are generated
using the ggsearch program of the FASTA package version 36.3.8c or later with a BLOSUM50 comparison matrix. The FASTA program package was authored by W. R. Pearson and D. J. Lipman (1988), “Improved Tools for Biological Sequence Analysis”, PNAS 85:2444-2448; W. R. Pearson (1996) “Effective protein sequence comparison” Meth. Enzymol. 266:227- 258; and Pearson et. al. (1997) Genomics 46:24-36, and is publicly available from http://fasta.bioch.virginia.edu/fasta_www2/fasta_down.shtml. Alternatively, a public server accessible at http://fasta.bioch.virginia.edu/fasta_www2/index.cgi can be used to compare the sequences, using the ggsearch (global protein: protein) program and default options (BLOSUM50; open: -10; ext: -2; Ktup = 2) to ensure a global, rather than local, alignment is performed. Percent amino acid identity is given in the output alignment header.
An “activating Fc receptor” is an Fc receptor that following engagement by an Fc domain of an antibody elicits signaling events that stimulate the receptor-bearing cell to perform effector functions. Human activating Fc receptors include FcyRIIIa (CD16a), FcyRI (CD64), FcyRIIa (CD32), and FcaRI (CD89).
“Reduced binding”, for example reduced binding to an Fc receptor, refers to a decrease in affinity for the respective interaction, as measured for example by SPR. For clarity, the term includes also reduction of the affinity to zero (or below the detection limit of the analytic method), i.e. complete abolishment of the interaction. Conversely, “increased binding” refers to an increase in binding affinity for the respective interaction.
By “fused” is meant that the components (e.g. a Fab molecule and an Fc domain subunit) are linked by peptide bonds, either directly or via one or more peptide linkers.
The HLA-A2/WT1 x CD3 bispecific antibody comprises a first antigen binding moiety that specifically binds to CD3, and a second antigen binding moiety that specifically binds to HLA- A2/WT1, particularly HLA-A2/WT1RMF.
In one aspect, the first antigen binding moiety comprises a heavy chain variable region comprising the heavy chain CDR (HCDR) 1 of SEQ ID NO: 1, the HCDR2 of SEQ ID NO: 2, and the HCDR3 of SEQ ID NO: 3; and a light chain variable region comprising the light chain CDR (LCDR) 1 of SEQ ID NO: 4, the LCDR2 of SEQ ID NO: 5 and the LCDR3 of SEQ ID NO: 6.
In one aspect, the second antigen binding moiety comprises a heavy chain variable region comprising the heavy chain CDR (HCDR) 1 of SEQ ID NO: 9, the HCDR2 of SEQ ID NO: 10,
and the HCDR3 of SEQ ID NO: 11; and a light chain variable region comprising the light chain CDR (LCDR) 1 of SEQ ID NO: 12, the LCDR2 of SEQ ID NO: 13 and the LCDR3 of SEQ ID NO: 14.
In a particular aspect, the HLA-A2/WT1 x CD3 bispecific antibody comprises
(i) a first antigen binding moiety that specifically binds to CD3 and comprises a heavy chain variable region comprising the heavy chain CDR (HCDR) 1 of SEQ ID NO: 1, the HCDR2 of SEQ ID NO: 2, and the HCDR3 of SEQ ID NO: 3; and a light chain variable region comprising the light chain CDR (LCDR) 1 of SEQ ID NO: 4, the LCDR2 of SEQ ID NO: 5 and the LCDR3 of SEQ ID NO: 6; and
(ii) a second antigen binding moiety that specifically binds to HLA-A2/WT1 and comprises a heavy chain variable region comprising the heavy chain CDR (HCDR) 1 of SEQ ID NO: 9, the HCDR2 of SEQ ID NO: 10, and the HCDR3 of SEQ ID NO: 11; and a light chain variable region comprising the light chain CDR (LCDR) 1 of SEQ ID NO: 12, the LCDR2 of SEQ ID NO: 13 and the LCDR3 of SEQ ID NO: 14.
In one aspect, the first antigen binding moiety comprises a heavy chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 7 and a light chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 8.
In one aspect, the first antigen binding moiety comprises the heavy chain variable region sequence of SEQ ID NO: 7 and the light chain variable region sequence of SEQ ID NO: 8.
In one aspect, the second antigen binding moiety comprises a heavy chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 15 and a light chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 16.
In one aspect, the second antigen binding moiety comprises the heavy chain variable region sequence of SEQ ID NO: 15 and the light chain variable region sequence of SEQ ID NO: 16.
In some aspects, the first and/or the second antigen binding moiety is a Fab molecule. In some aspects, the first antigen binding moiety is a crossover Fab molecule wherein either the variable
or the constant regions of the Fab light chain and the Fab heavy chain are exchanged. In such aspects, the second antigen binding moiety preferably is a conventional Fab molecule.
In some aspects wherein the first and the second antigen binding moiety of the bi specific antibody are both Fab molecules, and in one of the antigen binding moieties (particularly the first antigen binding moiety) the variable domains VL and VH of the Fab light chain and the Fab heavy chain are replaced by each other, i) in the constant domain CL of the first antigen binding moiety the amino acid at position 124 is substituted by a positively charged amino acid (numbering according to Kabat), and wherein in the constant domain CHI of the first antigen binding moiety the amino acid at position 147 or the amino acid at position 213 is substituted by a negatively charged amino acid (numbering according to Kabat EU index); or ii) in the constant domain CL of the second antigen binding moiety the amino acid at position 124 is substituted by a positively charged amino acid (numbering according to Kabat), and wherein in the constant domain CHI of the second antigen binding moiety the amino acid at position 147 or the amino acid at position 213 is substituted by a negatively charged amino acid (numbering according to Kabat EU index).
The bispecific antibody does not comprise both modifications mentioned under i) and ii). The constant domains CL and CHI of the antigen binding moiety having the VH/VL exchange are not replaced by each other (i.e. remain unexchanged).
In a more specific aspect, i) in the constant domain CL of the first antigen binding moiety the amino acid at position 124 is substituted independently by lysine (K), arginine (R) or histidine (H) (numbering according to Kabat), and in the constant domain CHI of the first antigen binding moiety the amino acid at position 147 or the amino acid at position 213 is substituted independently by glutamic acid (E), or aspartic acid (D) (numbering according to Kabat EU index); or ii) in the constant domain CL of the second antigen binding moiety the amino acid at position 124 is substituted independently by lysine (K), arginine (R) or histidine (H) (numbering according to Kabat), and in the constant domain CHI of the second antigen binding moiety the amino acid at
position 147 or the amino acid at position 213 is substituted independently by glutamic acid (E), or aspartic acid (D) (numbering according to Kabat EU index).
In one such aspect, in the constant domain CL of the second antigen binding moiety the amino acid at position 124 is substituted independently by lysine (K), arginine (R) or histidine (H) (numbering according to Kabat), and in the constant domain CHI of the second antigen binding moiety the amino acid at position 147 or the amino acid atposition 213 is substituted independently by glutamic acid (E), or aspartic acid (D) (numbering according to Kabat EU index).
In a further aspect, in the constant domain CL of the second antigen binding moiety the amino acid at position 124 is substituted independently by lysine (K), arginine (R) or histidine (H) (numbering according to Kabat), and in the constant domain CHI of the second antigen binding moiety the amino acid at position 147 is substituted independently by glutamic acid (E), or aspartic acid (D) (numbering according to Kabat EU index).
In preferred aspects, in the constant domain CL of the second antigen binding moiety the amino acid at position 124 is substituted independently by lysine (K), arginine (R) or histidine (H) (numbering according to Kabat) and the amino acid at position 123 is substituted independently by lysine (K), arginine (R) or histidine (H) (numbering according to Kabat), and in the constant domain CHI of the second antigen binding moiety the amino acid at position 147 is substituted independently by glutamic acid (E), or aspartic acid (D) (numbering according to Kabat EU index) and the amino acid at position 213 is substituted independently by glutamic acid (E), or aspartic acid (D) (numbering according to Kabat EU index).
In one aspect, in the constant domain CL of the second antigen binding moiety the amino acid at position 124 is substituted by lysine (K) (numbering according to Kabat) and the amino acid at position 123 is substituted by lysine (K) (numbering according to Kabat), and in the constant domain CHI of the second antigen binding moiety the amino acid at position 147 is substituted by glutamic acid (E) (numbering according to Kabat EU index) and the amino acid at position 213 is substituted by glutamic acid (E) (numbering according to Kabat EU index).
In one aspect, in the constant domain CL of the second antigen binding moiety the amino acid at position 124 is substituted by lysine (K) (numbering according to Kabat) and the amino acid at position 123 is substituted by arginine (R) (numbering according to Kabat), and in the constant domain CHI of the second antigen binding moiety the amino acid at position 147 is substituted by
glutamic acid (E) (numbering according to Kabat EU index) and the amino acid at position 213 is substituted by glutamic acid (E) (numbering according to Kabat EU index).
In particular aspects, if amino acid substitutions according to the above aspects are made in the constant domain CL and the constant domain CHI of the second antigen binding moiety, the constant domain CL of the second antigen binding moiety is of kappa isotype.
In some aspects, the first and the second antigen binding moiety are fused to each other, optionally via a peptide linker.
In some aspects, the first and the second antigen binding moiety are each a Fab molecule and either (i) the second antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N- terminus of the Fab heavy chain of the first antigen binding moiety, or (ii) the first antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N-terminus of the Fab heavy chain of the second antigen binding moiety.
In some aspects, the HLA-A2/WT1 x CD3 bispecific antibody provides monovalent binding to CD3.
In particular aspects, the HLA-A2/WT1 x CD3 bispecific antibody comprises a single antigen binding moiety that specifically binds to CD3, and two antigen binding moi eties that specifically bind to HLA-A2/WT1. Thus, in some aspects, the HLA-A2/WT1 x CD3 bispecific antibody comprises a third antigen binding moiety, particularly a Fab molecule, more particularly a conventional Fab molecule, that specifically binds to HLA-A2/WT1. The third antigen binding moiety may incorporate, singly or in combination, all of the features described hereinabove in relation to the second antigen binding moiety (e.g. the CDR sequences, variable region sequences, and/or amino acid substitutions in the constant regions). In some aspects, the third antigen moiety is identical to the first antigen binding moiety (e.g. is also a conventional Fab molecule and comprises the same amino acid sequences).
In particular aspects, the HLA-A2/WT1 x CD3 bispecific antibody further comprises an Fc domain composed of a first and a second subunit. In one aspect, the Fc domain is an IgG Fc domain. In a particular aspect, the Fc domain is an IgGi Fc domain. In another aspect the Fc domain is an IgG4 Fc domain. In a more specific aspect, the Fc domain is an IgG4 Fc domain comprising an amino acid substitution at position S228 (Kabat EU index numbering), particularly the amino acid substitution S228P. This amino acid substitution reduces in vivo Fab arm exchange of IgG4
antibodies (see Stubenrauch et al., Drug Metabolism and Disposition 38, 84-91 (2010)). In a further particular aspect, the Fc domain is a human Fc domain. In a particularly preferred aspect, the Fc domain is a human IgGi Fc domain. An exemplary sequence of a human IgGi Fc region is given in SEQ ID NO: 29.
In some aspects wherein the first, the second and, where present, the third antigen binding moiety are each a Fab molecule, (a) either (i) the second antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N-terminus of the Fab heavy chain of the first antigen binding moiety and the first antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N- terminus of the first subunit of the Fc domain, or (ii) the first antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N-terminus of the Fab heavy chain of the second antigen binding moiety and the second antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N-terminus of the first subunit of the Fc domain; and (b) the third antigen binding moiety, where present, is fused at the C-terminus of the Fab heavy chain to the N-terminus of the second subunit of the Fc domain.
In particular aspects, the Fc domain comprises a modification promoting the association of the first and the second subunit of the Fc domain. The site of most extensive protein-protein interaction between the two subunits of a human IgG Fc domain is in the CH3 domain. Thus, in one aspect said modification is in the CH3 domain of the Fc domain.
In a specific aspect said modification promoting the association of the first and the second subunit of the Fc domain is a so-called “knob-into-hole” modification, comprising a “knob” modification in one of the two subunits of the Fc domain and a “hole” modification in the other one of the two subunits of the Fc domain. The knob-into-hole technology is described e.g. in US 5,731,168; US 7,695,936; Ridgway et al., Prot Eng 9, 617-621 (1996) and Carter, J Immunol Meth 248, 7-15 (2001). Generally, the method involves introducing a protuberance (“knob”) at the interface of a first polypeptide and a corresponding cavity (“hole”) in the interface of a second polypeptide, such that the protuberance can be positioned in the cavity so as to promote heterodimer formation and hinder homodimer formation. Protuberances are constructed by replacing small amino acid side chains from the interface of the first polypeptide with larger side chains (e.g. tyrosine or tryptophan). Compensatory cavities of identical or similar size to the protuberances are created in the interface of the second polypeptide by replacing large amino acid side chains with smaller ones (e.g. alanine or threonine).
Accordingly, in some aspects, an amino acid residue in the CH3 domain of the first subunit of the Fc domain is replaced with an amino acid residue having a larger side chain volume, thereby generating a protuberance within the CH3 domain of the first subunit which is positionable in a cavity within the CH3 domain of the second subunit, and an amino acid residue in the CH3 domain of the second subunit of the Fc domain is replaced with an amino acid residue having a smaller side chain volume, thereby generating a cavity within the CH3 domain of the second subunit within which the protuberance within the CH3 domain of the first subunit is positionable. Preferably said amino acid residue having a larger side chain volume is selected from the group consisting of arginine (R), phenylalanine (F), tyrosine (Y), and tryptophan (W). Preferably said amino acid residue having a smaller side chain volume is selected from the group consisting of alanine (A), serine (S), threonine (T), and valine (V). The protuberance and cavity can be made by altering the nucleic acid encoding the polypeptides, e.g. by site-specific mutagenesis, or by peptide synthesis.
In a specific such aspect, in the first subunit of the Fc domain the threonine residue at position 366 is replaced with a tryptophan residue (T366W), and in the second subunit of the Fc domain the tyrosine residue at position 407 is replaced with a valine residue (Y407V) and optionally the threonine residue at position 366 is replaced with a serine residue (T366S) and the leucine residue at position 368 is replaced with an alanine residue (L368A) (numbering according to Kabat EU index). In a further aspect, in the first subunit of the Fc domain additionally the serine residue at position 354 is replaced with a cysteine residue (S354C) or the glutamic acid residue at position 356 is replaced with a cysteine residue (E356C) (particularly the serine residue at position 354 is replaced with a cysteine residue), and in the second subunit of the Fc domain additionally the tyrosine residue at position 349 is replaced by a cysteine residue (Y349C) (numbering according to Kabat EU index). In a preferred aspect, the first subunit of the Fc domain comprises the amino acid substitutions S354C and T366W, and the second subunit of the Fc domain comprises the amino acid substitutions Y349C, T366S, L368A and Y407V (numbering according to Kabat EU index).
In some aspects, the Fc domain comprises one or more amino acid substitution that reduces binding to an Fc receptor and/or effector function.
In a particular aspect the Fc receptor is an Fey receptor. In one aspect the Fc receptor is a human Fc receptor. In one aspect the Fc receptor is an activating Fc receptor. In a specific aspect the Fc receptor is an activating human Fey receptor, more specifically human Fey RUIa, FcyRI or FcyRIIa, most specifically human FcyRIIIa. In one aspect the effector function is one or more selected from
the group of complement dependent cytotoxicity (CDC), antibody-dependent cell-mediated cytotoxicity (ADCC), antibody-dependent cellular phagocytosis (ADCP), and cytokine secretion. In a particular aspect, the effector function is ADCC.
Typically, the same one or more amino acid substitution is present in each of the two subunits of the Fc domain. In one aspect, the one or more amino acid substitution reduces the binding affinity of the Fc domain to an Fc receptor. In one aspect, the one or more amino acid substitution reduces the binding affinity of the Fc domain to an Fc receptor by at least 2-fold, at least 5-fold, or at least 10-fold.
In one aspect, the Fc domain comprises an amino acid substitution at a position selected from the group of E233, L234, L235, N297, P331 and P329 (numberings according to Kabat EU index). In a more specific aspect, the Fc domain comprises an amino acid substitution at a position selected from the group of L234, L235 and P329 (numberings according to Kabat EU index). In some aspects, the Fc domain comprises the amino acid substitutions L234A and L235A (numberings according to Kabat EU index). In one such aspect, the Fc domain is an IgGi Fc domain, particularly a human IgGi Fc domain. In one aspect, the Fc domain comprises an amino acid substitution at position P329. In a more specific aspect, the amino acid substitution is P329A or P329G, particularly P329G (numberings according to Kabat EU index). In one aspect, the Fc domain comprises an amino acid substitution at position P329 and a further amino acid substitution at a position selected from E233, L234, L235, N297 and P331 (numberings according to Kabat EU index). In a more specific aspect, the further amino acid substitution is E233P, L234A, L235A, L235E, N297A, N297D or P331S. In particular aspects, the Fc domain comprises amino acid substitutions at positions P329, L234 and L235 (numberings according to Kabat EU index). In more particular aspects, the Fc domain comprises the amino acid mutations L234A, L235A and P329G (“P329G LALA”, “PGLALA” or “LALAPG”). Specifically, in preferred aspects, each subunit of the Fc domain comprises the amino acid substitutions L234A, L235A and P329G (Kabat EU index numbering), i.e. in each of the first and the second subunit of the Fc domain the leucine residue at position 234 is replaced with an alanine residue (L234A), the leucine residue at position 235 is replaced with an alanine residue (L235A) and the proline residue at position 329 is replaced by a glycine residue (P329G) (numbering according to Kabat EU index). In one such aspect, the Fc domain is an IgGi Fc domain, particularly a human IgGi Fc domain.
In preferred aspects, the HLA-A2/WT1 x CD3 bispecific antibody comprises
(i) a first antigen binding moiety that specifically binds to CD3, comprising a heavy chain variable region comprising the heavy chain CDR (HCDR) 1 of SEQ ID NO: 1, the HCDR2 of SEQ ID NO: 2, and the HCDR3 of SEQ ID NO: 3; and a light chain variable region comprising the light chain CDR (LCDR) 1 of SEQ ID NO: 4, the LCDR2 of SEQ ID NO: 5 and the LCDR3 of SEQ ID NO: 6, wherein the first antigen binding moiety is a crossover Fab molecule wherein either the variable or the constant regions, particularly the variable regions, of the Fab light chain and the Fab heavy chain are exchanged;
(ii) a second and a third antigen binding moiety that specifically bind to HLA-A2/WT 1 , comprising a heavy chain variable region comprising the heavy chain CDR (HCDR) 1 of SEQ ID NO: 9, the HCDR2 of SEQ ID NO: 10, and the HCDR3 of SEQ ID NO: 11; and a light chain variable region comprising the light chain CDR (LCDR) 1 of SEQ ID NO: 12, the LCDR2 of SEQ ID NO: 13 and the LCDR3 of SEQ ID NO: 14, wherein the second and third antigen binding moiety are each a Fab molecule, particularly a conventional Fab molecule;
(iii) an Fc domain composed of a first and a second subunit, wherein the second antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N-terminus of the Fab heavy chain of the first antigen binding moiety, and the first antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N-terminus of the first subunit of the Fc domain, and wherein the third antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N-terminus of the second subunit of the Fc domain.
In one aspect, the first antigen binding moiety comprises a heavy chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 7 and a light chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 8.
In one aspect, the first antigen binding moiety comprises the heavy chain variable region sequence of SEQ ID NO: 7 and the light chain variable region sequence of SEQ ID NO: 8.
In one aspect, the second and third antigen binding moiety comprise a heavy chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 15 and a light chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 16.
In one aspect, the second and third antigen binding moieties comprise the heavy chain variable region of SEQ ID NO: 15 and the light chain variable region of SEQ ID NO: 16.
The Fc domain according to the above aspects may incorporate, singly or in combination, all of the features described hereinabove in relation to Fc domains.
In one aspect, the antigen binding moieties and the Fc region are fused to each other by peptide linkers, particularly by peptide linkers as in SEQ ID NO: 18 and SEQ ID NO: 20.
In one aspect, in the constant domain CL of the second and the third Fab molecule under (ii) the amino acid at position 124 is substituted by lysine (K) (numbering according to Kabat) and the amino acid at position 123 is substituted by lysine (K) or arginine (R), particularly by arginine (R) (numbering according to Kabat), and in the constant domain CHI of the second and the third Fab molecule under (ii) the amino acid at position 147 is substituted by glutamic acid (E) (numbering according to Kabat EU index) and the amino acid at position 213 is substituted by glutamic acid (E) (numbering according to Kabat EU index).
In one aspect, the HLA-A2/WT1 x CD3 bispecific antibody comprises a polypeptide (particularly two polypeptides) comprising a sequence that is at least 80%, 85%, 90%, 95%, 96%, 97%, 98%, or 99% identical to the sequence of SEQ ID NO: 17, a polypeptide comprising a sequence that is at least 80%, 85%, 90%, 95%, 96%, 97%, 98%, or 99% identical to the sequence of SEQ ID NO: 18, a polypeptide comprising a sequence that is at least 80%, 85%, 90%, 95%, 96%, 97%, 98%, or 99% identical to the sequence of SEQ ID NO: 19, and a polypeptide comprising a sequence that is at least 80%, 85%, 90%, 95%, 96%, 97%, 98%, or 99% identical to the sequence of SEQ ID NO: 20.
In one aspect, the HLA-A2/WT1 x CD3 bispecific antibody comprises a polypeptide (particularly two polypeptides) comprising the sequence of SEQ ID NO: 17, a polypeptide comprising the sequence of SEQ ID NO: 18, a polypeptide comprising the sequence of SEQ ID NO: 19, and a polypeptide comprising the sequence of SEQ ID NO: 20.
The HLA-A2/WT1 x CD3 bispecific antibody herein is used in combination with venetoclax and/or azacitidine, preferably venetoclax and (i.e. plus) azacitidine.
The term “venetoclax” (abbreviated as “Ven”) refers to the compound with the chemical (IUPAC) name 4-[4-[[2-(4-chlorophenyl)-4,4-dimethylcyclohex- 1 -en- 1 -yl]methyl]piperazin- 1 -y 1 ] -N- [3 -
nitro-4-(oxan-4-ylmethylamino)phenyl]sulfonyl-2-(17/-pyrrolo[2,3-Z>]pyridin-5-yloxy)benzamide, and the following chemical structure:
CAS registry number 1257044-40-8. The empirical formula for venetoclax is C45H50CIN7O7S and the gram molecular weight is 868.4. Venetoclax is marketed under the tradename VENCLEXTA® and VENCLYXTO®. Venetoclax is administered orally, and is available as tablets of 10, 50 or 100 mg venetoclax. It is an inhibitor of the anti-apoptotic B-cell lymphoma-2 (BCL-2) protein. It is, for example, used in the treatment of chronic lymphocytic leukemia (CLL), small lymphocytic leukemia (SLL) or acute myeloid leukemia (AML). In the treatment of AML, it may be used in combination with azacitidine. The venetoclax daily dose for AML treatment is typically between 100 mg and 600 mg (between 100 mg and 400 mg when used in combination with azacitidine). A particular AML dosing schedule (including dose-titration) is described hereinbelow.
The term “azacitidine” or “5 -azacitidine” (abbreviated as “Aza” or “5-Aza”) refers to the compound with the chemical (IUPAC) name 4-amino-l-[(2A, 3A,45, 5A)-3,4-dihydroxy-5- (hydroxymethyl)oxolan-2-yl]-l,3,5-triazin-2-one, and the following chemical structure:
CAS registry number 320-67-2. The empirical formula for azacitidine is C8H12N4O5 and the gram molecular weight is 244.2. Azacitidine is a cytidine analogue and acts as a nucleid acid synthesis
inhibitor (hypomethylation agent). Azacitidine is marketed under the tradename VIDAZA® or ONUREG®. It may be administered subcutaneously, intravenously or orally (tablets of 200 mg or 200 mg azacitidine being available). It is used in the treatment of myelodysplastic syndrome. It is also used in the treatment of acute myeloid leukemia (AML). In the treatment of AML, it may be used in combination with venetoclax. An azacitidine daily dose for AML treatment is typically 300 mg (orally, once daily). Alternatively, an azacitidine daily dose for AML treatment is 75 mg/m2 (intravenously or subcutaneously). Azacitidine is typically administered in treatment cycles of 28 days, consisting of a treatment period of 14 days followed by a treatment free period of 14 days.
Other nucleic acid synthesis inhibitors that will be known to the skilled practitioner (e.g. decitabine (also known as 5-aza-2'-deoxycytidine; CAS Registry Number 2353-33-5; sold under the trade name DACOGEN®), or cytarabine (also known as cytosine arabinoside (ara-C); CAS Registry Number 147-94-4; sold under the trade name CYTOSAR-U®)) are also contemplated for use in the present invention.
The combination of venetoclax plus azacitidine is the standard of care treatment for patients with AML, specifically patients with newly diagnosed AML who are not candidates for intensive chemotherapy. A dosing schedule (including dose titration of venetoclax) for the combination comprises a first 28-day dosing cycle with 100 mg venetoclax on day 1, 200 mg venetoclax on day 2, and 400 mg venetoclax on day 3 to 28, plus 75 mg/m2 azacitidine on day 1-7, followed by 28- day dosing cycles with 400 mg venetoclax daily plus 75 mg/m2 azacitidine on day 1-7 of each cycle. Such dosing schedule is illustrated in Table 1 below.
Table 1. Dosing schedule for AML for venetoclax in combination with azacitidine.
Unless expressly stated otherwise, by “venetoclax/azazitidine” is meant venetoclax and/or azazitidine, preferably venetoclax and (i.e. plus) azazitidine.
The term “cancer” refers to the physiological condition in mammals that is typically characterized by unregulated cell proliferation. Examples of cancer include but are not limited to, carcinoma, lymphoma, blastoma, sarcoma and leukemia. More non-limiting examples of cancers include haematological cancer such as leukemia, bladder cancer, brain cancer, head and neck cancer, pancreatic cancer, biliary cancer, thyroid cancer, lung cancer, breast cancer, ovarian cancer, uterine cancer, cervical cancer, endometrial cancer, esophageal cancer, colon cancer, colorectal cancer, rectal cancer, gastric cancer, prostate cancer, skin cancer, squamous cell carcinoma, sarcoma, bone cancer, and kidney cancer. Other cell proliferation disorders include, but are not limited to neoplasms located in the: abdomen, bone, breast, digestive system, liver, pancreas, peritoneum, endocrine glands (adrenal, parathyroid, pituitary, testicles, ovary, thymus, thyroid), eye, head and neck, nervous system (central and peripheral), lymphatic system, pelvic, skin, soft tissue, spleen, thoracic region, and urogenital system. Also included are pre-cancerous conditions or lesions and cancer metastases.
In some aspects of the HLA-A2/WT1 x CD3 bispecific antibodies, methods, uses and kits of the invention, the cancer is a haematological cancer. Non-limiting examples of haematological cancers include leukemia (e.g. acute lymphocytic leukemia (ALL), acute myeloid leukemia (AML), chronic lymphcytic leukemia (CLL) chronic myeloid leukemia (CML), hairy cell leukemia (HCL)), lymphoma (e.g. Non-Hodgkin lymphoma (NHL), Hodgkin lymphoma), myeloma (e.g. multiple myeloma (MM)), myelodysplastic syndrome (MDS) and myeloproliferative diseases.
In certain aspects the cancer is chosen from the group consisting of haematological cancer (such as leukemia), kidney cancer, bladder cancer, skin cancer, lung cancer, colorectal cancer, breast cancer, brain cancer, head and neck cancer and prostate cancer.
In particular aspects, the cancer is a haematological cancer, particularly leukemia, most particularly acute lymphocytic leukemia (ALL) or acute myeloid leukemia (AML).
In preferred aspects the cancer is acute myeloid leukemia (AML).
In further particular aspects, the cancer is myelodysplastic syndrome (MDS).
In some aspects, the cancer is a WT1 -positive cancer. By “WT1 -positive cancer” or “WT1- expressing cancer” is meant a cancer characterized by expression or overexpression of WT1 in cancer cells. The expression of WT1 may be determined for example by quantitative real-time PCR (measuring WT1 mRNA levels), immunohistochemistry (IHC) or western blot assays. In one aspect, the cancer expresses WT1. In one aspect, the cancer expresses WT1 in at least 20%, preferably at least 50% or at least 80% of tumor cells as determined by immunohistochemistry (IHC) using an antibody specific for WT1.
A “patient”, “subject” or “individual” herein is any single human subject eligible for treatment who is experiencing or has experienced one or more signs, symptoms, or other indicators of cancer. In some aspects, the patient has cancer or has been diagnosed with cancer. The patient may have been previously treated with a HLA-A2/WT1 x CD3 bispecific antibody or another drug, or not so treated. In particular aspects, the patient has not been previously treated with a HLA-A2/WT1 x CD3 bispecific antibody. The patient may have been treated with a therapy comprising one or more drugs other than a HLA-A2/WT1 x CD3 bispecific antibody before the HLA-A2/WT1 x CD3 bispecific antibody therapy is commenced. In particular aspects, the patient carries a HLA- A2 allele, in particular the HLA-A*02:01 allele.
As used herein, “treatment” (and grammatical variations thereof such as “treat” or “treating”) refers to clinical intervention in an attempt to alter the natural course of a disease in the individual being treated, and can be performed either for prophylaxis or during the course of clinical pathology. Desirable effects of treatment include, but are not limited to, preventing occurrence or recurrence of disease, alleviation of symptoms, diminishment of any direct or indirect pathological consequences of the disease, preventing metastasis, decreasing the rate of disease progression, amelioration or palliation of the disease state, and remission or improved prognosis.
The HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine are administered in an effective amount.
An "effective amount" of an agent, e.g. a pharmaceutical composition, refers to an amount effective, at dosages and for periods of time necessary, to achieve the desired therapeutic or prophylactic result.
In some aspects, administration of the HLA-A2/WT1 x CD3 bispecific antibody results in activation of T cells, particularly cytotoxic T cells, particularly at the site of the cancer. Said
activation may comprise proliferation of T cells, differentiation of T cells, cytokine secretion by T cells, cytotoxic effector molecule release from T cells, cytotoxic activity of T cells, and expression of activation markers by T cells. In one aspect, the administration of the HLA-A2/WT1 x CD3 bispecific antibody results in an increase of T cell, particularly cytotoxic T cell, numbers at the site of the cancer.
In some aspects of the HLA-A2/WT1 x CD3 bi specific antibodies, methods, uses or kits described above and herein, the treatment with or administration of the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine results in increased activation of T cells, particularly cytotoxic T cells, particularly at the site of the cancer, as compared to treatment with or administration of the HLA-A2/WT1 x CD3 bispecific antibody alone. In particular aspects, the activation comprises cytotoxic activity (specifically lysis of cancer cells) of T cells and/or cytokine (specifically IL-2, TNF-a, and/or interferon-y) secretion by T cells.
In some aspects of the HLA-A2/WT1 x CD3 bi specific antibodies, methods, uses or kits described above and herein, the treatment with or administration of the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine results in reduced cancer growth and/or increased survival time, as compared to treatment with or administration of the HLA-A2/WT1 x CD3 bispecific antibody alone.
In some aspects of the HLA-A2/WT1 x CD3 bi specific antibodies, methods, uses or kits described above and herein, the treatment with or administration of the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine may result in a response in the individual. In some aspects, the response may be a complete response. In some aspects, the response may be a sustained response after cessation of the treatment. In some aspects, the response may be a complete response that is sustained after cessation of the treatment. In other aspects, the response may be a partial response. In some aspects, the response may be a partial response that is sustained after cessation of the treatment. In some aspects, the treatment with or administration of the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine may improve the response as compared to treatment with or administration of the HLA-A2/WT1 x CD3 bispecific antibody alone (i.e. without venetocl ax/ azaciti dine) .
In some aspects, the treatment or administration of the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine may increase response rates in a patient population, as compared to a
corresponding patient population treated with the HLA-A2/WT1 x CD3 bispecific antibody alone (i.e. without venetoclax/azacitidine).
The combination therapy of the invention comprises administration of a HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine.
As used herein, “combination” (and grammatical variations thereof such as “combine” or “combining”) encompasses combinations of a HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine according to the invention wherein the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine are in the same or in different containers, in the same or in different pharmaceutical formulations, administered together or separately, administered simultaneously or sequentially, in any order, and administered by the same or by different routes, provided that the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine can simultaneously exert their biological effects in the body. For example “combining” HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine according to the invention may mean first administering the HLA-A2/WT1 x CD3 bispecific antibody in a particular pharmaceutical formulation, followed by administration of venetoclax/azacitidine in one or more other pharmaceutical formulation, or vice versa.
The HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine may be administered in any suitable manner known in the art. In one aspect, the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine are administered sequentially (at different times). In another aspect, the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine are administered concurrently (at the same time). In some aspects, the HLA-A2/WT1 x CD3 bispecific antibody is in a separate composition from venetoclax/azacitidine (wherein venetoclax and azacitidine may each be in separate compositions as well). In some aspects, the HLA-A2/WT1 x CD3 bispecific antibody is in the same composition as venetoclax/azacitidine.
The HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine can be administered by any suitable route, and may be administered by the same route of administration or by different routes of administration. In some aspects, the HLA-A2/WT1 x CD3 bispecific antibody is administered intravenously, intramuscularly, subcutaneously, topically, orally, transdermally, intraperitoneally, intraorbitally, by implantation, by inhalation, intrathecally, intraventricularly, or intranasally. In a particular aspect, the HLA-A2/WT1 x CD3 bispecific antibody is administrered intravenously. In some aspects, venetoclax/azacitidine is administered intravenously,
intramuscularly, subcutaneously, topically, orally, transdermally, intraperitoneally, intraorbitally, by implantation, by inhalation, intrathecally, intraventricularly, or intranasally. In a particular aspect, venetoclax/azacitidine, in particular venetoclax, is administered orally. In another particular aspect, venetoclax/azacitidine, in particular azacitidine, is administered intravenously or subcutaneously. In a particular aspect, venetoclax is administered orally and azacitidine is administered intravenously or subcutaneously. An effective amount of the HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine may be administered for prevention or treatment of disease. The appropriate route of administration and dosage of the HLA-A2/WT1 x CD3 bispecific antibody and/or venetoclax/azacitidine may be determined based on the type of disease to be treated, the type of the HLA-A2/WT1 x CD3 bispecific antibody, the severity and course of the disease, the clinical condition of the individual, the individual’s clinical history and response to the treatment, and the discretion of the attending physician. Dosing can be by any suitable route, e.g. by injections, such as intravenous or subcutaneous injections, depending in part on whether the administration is brief or chronic. Various dosing schedules including but not limited to single or multiple administrations over various time-points, bolus administration, and pulse infusion are contemplated herein. The HLA-A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine are suitably administered to the patient at one time or over a series of treatments. A typical dosing schedule for venetoclax/azacitidine is described hereinabove.
Combinations of the invention can be used either alone or together with other agents in a therapy. For instance, a combination of the invention may be co-administered with at least one additional therapeutic agent. In certain aspects, an additional therapeutic agent is an anti-cancer agent, e.g. a chemotherapeutic agent, an inhibitor of tumor cell proliferation, or an activator of tumor cell apoptosis. Combinations of the invention can also be combined with radiation therapy.
A kit as provided herein typically comprises one or more container and a label or package insert on or associated with the container. Suitable containers include, for example, bottles, vials, syringes, IV solution bags, etc. The containers may be formed from a variety of materials such as glass or plastic. The container holds a composition which is by itself or combined with another composition effective for treating, preventing and/or diagnosing the condition and may have a sterile access port (for example the container may be an intravenous solution bag or a vial having a stopper pierceable by a hypodermic injection needle). At least one active agent in the composition is a HLA-A2/WT1 x CD3 bispecific antibody to be used in the combinations of the invention. Another active agent is venetoclax/azacitidine to be used in the combinations of the
invention, which may be in the same composition and container like the bispecific antibody, or may be provided in a different composition and container. The label or package insert indicates that the composition(s) is/are used for treating the condition of choice, such as cancer.
In one aspect the invention provides a kit intended for the treatment of cancer, comprising in the same or in separate containers (a) a HLA-A2/WT1 x CD3 bispecific antibody, and (b) venetoclax/azacitidine, and optionally further comprising (c) a package insert comprising printed instructions directing the use of the combined treatment as a method for treating cancer. Moreover, the kit may comprise (a) a first container with a composition contained therein, wherein the composition comprises a HLA-A2/WT1 x CD3 bispecific antibody; (b) a second and/or a third container with a composition contained therein, wherein the composition comprises venetoclax/azacitidine; and optionally (c) a fourth container with a composition contained therein, wherein the composition comprises a further cytotoxic or otherwise therapeutic agent. The kit in these aspects of the invention may further comprise a package insert indicating that the compositions can be used to treat cancer. Alternatively, or additionally, the kit may further comprise a third (or fourth) container comprising a pharmaceutically-acceptable buffer, such as bacteriostatic water for injection (BWFI), phosphate-buffered saline, Ringer's solution and dextrose solution. It may further include other materials desirable from a commercial and user standpoint, including other buffers, diluents, filters, needles, and syringes.
Figure 1. Schematic illustration of the HLA-A2/WT1 -targeted T-cell bispecific (TCB) antibody molecule used in the Examples (“WT1-TCB”). The molecule comprises a single antigen binding moiety for CD3, two antigen binding sites for HLA-A2/WT1, and an Fc domain.
Figure 2. Induction of TCR downstream signaling in Jurkat-NFAT reporter cells by DP47-TCB and WT1-TCB in cocultures with SKM-1 and OCI-AML3 cells pretreated for three days with Ven/ Aza (n=3). Bars represent mean±SEM. Statistical test: Two-way ANOVA with Tukey’s multiple comparisons test. *p<0.05, **** ?<0.0001.
Figure 3. (A) WT1-TCB mediated cytotoxicity of SKM1 cells after 6 days of coculture with healthy donor (HD) T cells by WT1-TCB and Ven/ Aza. Aza was replenished after 24 and 48 h and Ven after 72 h assuming full degradation. 0.01 pg/mL WT1-TCB was added at the start of the experiment and replenished after 72 h assuming full consumption (parallel dosing), or was only added after 72 h (sequential dosing) (n=3). (B) T-cell expansion after 6 days of coculture calculated as fold-change compared to the start of the coculture (n=3). Bars represent mean±SEM.
Figure 4. (A) WT1-TCB mediated cytotoxicity of OCI-AML3 cells after 6 days of coculture with healthy donor (HD) T cells by WT1-TCB and Ven/ Aza. Aza was replenished after 24 and 48 h and Ven after 72 h assuming full degradation. 1 pg/mL WT1-TCB was added at the start of the experiment and replenished after 72 h assuming full consumption (n=9). (B) T-cell expansion in after 6 days of coculture with OCI-AML3 cells calculated as fold-change compared to the start of the coculture (n=9). Bars represent mean±SEM. (C) Cytokine secretion in cocultures of OCI- AML3 cells, healthy donor T cells and WT1-TCB ± Ven/Aza after 6 days of coculture (n=9). Statistical testing: (A+B) One-way ANOVA and Tukey’s test; (C) Paired t-test
Figure 5. (A) WT1-TCB mediated cytotoxicity by HD T cells of primary AML cells pre-treated with Ven-Aza for 7 days or left untreated (n=3). (B) Detailed representation of the data from (A) for all patients. Of note, one patient showed overall a low specific lysis that was further decreased upon Ven-Aza pre-treatment.
Figure 6. WT1-TCB mediated cytotoxicity of primary AML cells after 6 days of coculture with healthy donor T cells and Ven/Aza. Aza was replenished after 24 and 48 h and Ven after 72
RECTIFIED SHEET (RULE 91) ISA/EP
h assuming full degradation. 1 pg/mL WT1-TCB was added at the start of the experiment and replenished after 72 h assuming full consumption (n=4). Bars represent mean ± SEM.
Figure 7. Schematic overview of the study design of Example 2. AZA = azacytidine; d = day; NSG = NOD.Cg-Prkdcscid I12rgtmlWjl/SzJ; Ven = Venetoclax.
Figure 8. In vivo tumor growth curves (on therapy day 36). (A) Efficacy. Tumor growth curve in SKM-1 tumor-bearing humanized NSG mice treated IV with vehicle once weekly; WT1-TCB alone at 0.05 mg/kg IV once weekly; Venetoclax 50 mg/kg PO + 5-AZA 1 mg/kg IP 5 times a week; or WT1-TCB in combination with Venetoclax + 5-AZA (only for 1 week) at the described dosing schedules (Table 2). Dashed perpendicular lines: Vehicle or WT1-TCB according to treatment group. Dotted perpendicular lines: 1st week = Venetoclax 50 mg/kg + 5-AZA 1 mg/kg; 2nd-4th week Venetoclax 50 mg/kg. For P-values on Study Day 36, see Table 3. (B) Tumor load. Tumor growth in individual mice of each group, as indicated. 5-AZA = 5-azacytidine; IP = intraperitoneally; IV = intravenously; PO = orally.
Figure 9. In vivo Kaplan-Meier curve. Kaplan-Meier curve of humanized mice bearing SKM-1 tumors treated IV with vehicle once weekly; WT1-TCB alone at 0.05 mg/kg IV once weekly; Venetoclax 50 mg/kg PO + 5-AZA 1 mg/kg IP 5 times a week; or WT1-TCB in combination with Venetoclax + 5-AZA (only for 1 week) at the previously described dosing schedules (Table 2). P-values on Study Day 36 were calculated using the log-rank test adjusted for multiple testing and are shown in Table 4. 5-AZA = 5-azacytidine; IP = intraperitoneally; IV = intravenously; PO = orally.
Examples
The following are examples of methods and compositions of the invention. It is understood that various other aspects may be practiced, given the general description provided above.
Example 1 - Combination of WT1-TCB with Venetoclax/Azazitidine (Ven/Aza)
Experimental set-up
Target antigen expression in response to Ven/ Aza was indirectly evaluated by analyzing TCR- downstream signaling. SKM-1 and 0CI-AML3 cells (both HLA-A*O2+/WT1+) were cultured for 72 h in the presence of 1 pM Aza (daily treatment) and 25 nM Ven. Cells were subsequently cocultured at a ratio of 1 : 1 with a Jurkat cell line that has stably integrated a NFAT-dependent Luciferase Reporter system (Signosis, Santa Clara, CA) for 6 h, together with 1 pg/mL WT1-TCB (SEQ ID NOs 9-16 (HLA-A2/WT1 CDRs and V-regions), 1-8 (CD3 CDRs and V-regions) and 17-20 (full heavy and light chains), molecular structure as in Figure 1), the DP47-TCB control construct (an untargeted TCB of similar structure, binding only CD3 but no tumor antigen, having SEQ ID NOs 21-22 as non-binding V-regions), as well as no TCB. Luminescence was induced by using the Bright-Glo Luciferase Assay System (Promega, Fitchburg, WI) and measured on an Infinite M1000 Plate Reader (Tecan, Mannedorf, Switzerland).
Lysis of AML cell lines by the combination of WT1-TCB and Ven/ Aza was analyzed in vitro. LOxlO5 SKM-1 cells or OCI-AML cells (both HLA-A*O2+/WT1+) were cocultured with 5.0xl04 healthy donor T cells (negatively isolated using Human T cell isolation kit; Stemcell, Vancouver, Canada) in a 96-well plate in the presence of 25 nM Venetoclax and 1 pM Azacytidine (both Selleckchem, Houston, TX). Azacytidine was replenished after 24 and 48 h and Venetoclax after 72 h assuming full degradation. WT1-TCB was added at 0.01 pg/mL (SKM-1) or 1 pg/mL (OCL AML3) at the start of the experiment and replenished after 72 h assuming full consumption (parallel dosing), or was only added after 72 h (sequential dosing). DMSO and the non-targeted control antibody construct (DP47-TCB) were included as controls, as well as an untreated control without WT1-TCB, Venetoclax and Azacytidine. Cocultures were analyzed after six days for overall lysis of AML cell lines and expansion of T cells. Overall lysis of AML cell lines by WT1- TCB was calculated relative to an untreated control based on viable AML cell counts as determined by flow cytometry. Proliferation of T cells after 96 h was calculated relative to Day 0 based on viable T cell counts. Cell culture supernatants from cytotoxicity assays with OCI-AML3 cells were analyzed on day 6 for cytokine secretion using LEGENDplex Human CD8/NK panel (Biolegend, San Diego, CA) according to the manufacturer’s instruction.
Primary AML (pAML) cells from HLA-A*02+ patients were collected at initial diagnosis after written informed consent in accordance with the Declaration of Helsinki and approval granted by the Institutional Review Board of the Ludwig-Maximilian-Universitat (Munich, Germany). pAML cells were pre-cultivated in a twelve well-plate at a concentration of 1.5xl06 cells/mL for 2-3 days
on a feeder layer of 1.2xl05 murine MS5 cells (irradiated with 60 Gy) per well and subsequently enriched by depletion of T cells [using CD3 MicroBeads, human (Miltenyi Biotec)].
WT1-TCB mediated lysis of pAML cells was analyzed after in vitro-pretreatment of pAML cells with Ven/ Aza. To this end, pAML cells were cultivated on irradiated MS5 cells and were left untreated or were pretreated for seven days with 1 pM Azacytidine (daily addition on days 1-5) and 300 nM Ven (added on day 5). T cells and pAML cells were subsequently cocultivated for 3 days at an E:T ratio of 1 : 1 with various concentrations of WT1-TCB (0.01-10 pg/mL) and specific lysis of AML cells was calculated relative to corresponding controls with DP47-TCB based on viable AML cell counts.
Alternatively, pAML cells were co-cultured with healthy donor T cells for 7 days at an E:T ratio of 1 :2 on a layer of MS5 cells in the presence of 25 nM Ven, 1 pM Aza. Azacytidine was replenished after 24 and 48 h and Venetoclax after 72 h assuming full degradation. WT1-TCB was added at 1 pg/mL at the start of the experiment and replenished after 72 h assuming full consumption DMSO and the non-targeted control antibody construct (DP47-TCB) were included as controls, as well as an untreated control without WT1-TCB, Venetoclax and Azacytidine. Overall lysis was assessed by flow cytometry on day 6 and calculated relative to the untreated control.
Results
Target antigen expression on AML cell lines was measured in response to stimulation with Ven/ Aza. WT1-TCB induced TCR downstream signaling in cocultures with SKM-1 and OCI- AML3 cells, as measured by luminescence induced in NF AT Luciferase Reporter Jurkat cells. Prestimulation of AML cells with Ven and Aza significantly increased TCR downstream signaling by WT1-TCB (SKM-1 : 5632.0±134.2 vs. 7952.0±241.8; <0.0001; OCI-AML3: 439.7±19.9 vs. 733.0±46.3; /?=0.0146; mean±SEM; both n=3). Only minimal luminescence was observed in cocultures if the untargeted DP47-TCB was used (Figure 2).
In cocultures of SKM-1 cells and healthy donor (HD) T cells a mean overall lysis of 75.2±9.2% (±SEM; n=3) was observed after six days in the presence of WT1-TCB and was increased to 95.5±0.8% by parallel administration of Ven/ Aza. If SKM-1 cells were precultured for three days with Ven/ Aza and sequentially treated for further three days with WT1-TCB a mean overall lysis of 96.4±1.7% was observed. Venetoclax and Azacytidine induced a mean overall lysis of 85.6- 87.4% in combinations with the untargeted DP47-TCB. No lysis was observed by DP47-TCB single treatment (Figure 3A). Analysis of T-cell expansion on day 6 of coculture revealed a
7.5±2.1-fold (n=3) increase by WT1-TCB compared to the start of the coculture, whereas a 3.8±1.6-fold change was observed upon parallel administration of Ven/ Aza. Sequential culturing with Ven/ Aza followed by WT1-TCB lead to a 0.78±0.25-fold change in T-cell counts, most likely due to a lack of T-cell stimulation within the first three days of culture and a shorter stimulation with WT1-TCB for only three days compared to six days (Figure 3B).
WT1-TCB mediated lysis in combination with Ven/ Aza was furthermore tested in vitro in cocultures of HD T cells and OCI-AML3 cells (Figure 4) for six days in the presence of 1 pg/mL WT1-TCB, 25 nM Ven and 1 pM Aza. Ven/ Aza mediated cytotoxicity of 0CI-AML3 cells was significantly increased in combination with the WT1-TCB from 29.5±7.6% to 69.8±7.8% (p=0.0004; ±SEM; n=9) (Figure 4A). Moreover, WT1-TCB led to a significant increase in T-cell proliferation in the presence of Ven/Aza (mean fold expansion compared to day 0: 2.7±0.4) (Figure 4B). This was comparable to monotherapy with WT1-TCB (2.3±0.2), whereas treatments with the untargeted DP47-TCB or only Ven/Aza did not lead to a T-cell expansion (0.8±0.1 and 0.9±0.1, respectively) (Figure 4B). Furthermore, lower levels of secreted proinflammatory cytokines IFNy (p<0.0001), IL-6 (p=0.004), and TNF (p=0.056), as well as secretion of Granzyme B (p=0.125) and IL-2 (p=0.004) were observed in the Ven/Aza and WT1-TCB combination treatment compared to WT1-TCB monotherapy (Figure 4C).
WT1-TCB mediated lysis by HD T cells was furthermore analyzed in combination with pAML cells. Pretreatment of pAML cells with Ven- Aza resulted at a concentration of 1 pg/mL WT1- TCB in a mean specific lysis of 61.1±29.3% compared to 67.0±15.9% (±SEM; n=3) with untreated pAML cells (Figure 5A). Of note, a reduction in cell lysis by Ven-Aza pretreatment was only observed for one patient sample whereas no differences were observed for the other two patient samples between with and without Ven-Aza pretreatment (Figure 5B).
WT1-TCB mediated lysis of pAML cells by HD T cells was furthermore analyzed upon parallel treatment with Ven/Aza for seven days. While monotherapies with Ven/Aza or WT1-TCB already achieved 40-50% lysis, the combinatorial approach of WT1-TCB and Ven/Aza enhanced cytotoxicity up to 61.2±11.2% (n=4; Figure 6).
Example 2 - Combination study of WT1-TCB with Venetoclax/Azacytidine (Ven/Aza) in
SKM-1 tumor-bearing humanized NSG mice
The purpose of this study was to identify whether combination with standard of care treatment for acute myeloid leukemia (venetoclax, an inhibitor of B-cell lymphoma-2 (BCL-2), plus 5-azacytidine [5-AZA], a cytidine analog causing hypomethylation at low doses and cell death at high doses) further increases antitumor efficacy of WT1-TCB.
Experimental set-up
Humanized HLA-A*02-positive mice were injected with SKM-1 tumor cells. After tumors reached an average tumor size of 198 mm3, mice were treated with WT1-TCB (0.05 mg/kg), Venetoclax (50 mg/kg; Sigma Aldrich) plus 5-AZA (1 mg/kg; Sigma Aldrich), or the combination of these compounds.
A schematic overview of the experimental design is presented in Figure 7.
NSG mice humanized using an HLA-A*02+ (CD34+) hematopoietic stem cell donor were purchased from Jackson Laboratories (Bar Harbor, ME). Mice were aged 16-26 weeks at the start of the experiments and were maintained under specific-pathogen-free conditions, with 12: 12 h light/dark daily cycles, in accordance to the required local and federal guidelines and laws (GVSolas; Felasa; TierschG). The experimental study protocol was reviewed and approved by local government authorities (approval number ROB-55.2-2532. Vet_02-20-170). After arrival, animals were maintained for 1 week to get accustomed to the new environment and for observation. Continuous health monitoring was carried out daily.
Humanized NSG mice were injected subcutaneously (SC) with 10 x 106 SKM-1 tumor cells (Catalog No. ACC547; DSMZ) on Study Day 0. Mice were randomized in 4 different study groups (Table 2). Treatment of humanized mice with the test compounds started 14 days after tumor-cell injection, when tumors reached an average size of 198 mm3, and lasted 4 weeks. Mice were dosed with compounds according to their body weight with a volume of 10 mL/kg, as described in the following.
The first group of mice received vehicle as control (for vehicle treatment, see Table 2). The second group received WT1-TCB intravenously at 0.05 mg/kg once weekly. The third group received histidine buffer intravenously once weekly and at the following 5 days Venetoclax at 50 mg/kg orally plus 5-AZA at 1 mg/kg intraperitoneally for the first week. In the following 3 weeks, this treatment group received only vehicle plus Venetoclax. The fourth treatment group received WT1- TCB plus Venetoclax plus 5-AZA at the same dosing schedule as described for Groups 2 and 3.
Table 2. Study groups.
Iq7d = once weekly; 5-AZA = azacytidine; 5q7d = 5 times a week; IP = intraperitoneal; IV = intravenous; PO = oral.
Tumor volume was measured by caliper every second day. Tumor growth curves were plotted in GraphPad Prism (version 8.4.2; San Diego, CA). P-values for group comparisons were calculated using the non-parametric Dunn’s test. Pairwise group comparison of the survival data was performed using the log-rank test, which places more weight on later survival events and is adjusted for multiple testing.
Results
The in vivo antitumor activity of WT1-TCB in combination with Venetoclax plus 5-AZA, which constitutes the current standard of care for acute myeloid leukemia, was assessed by treating SKM- 1 tumor-bearing humanized NSG mice for 4 weeks with WT1-TCB at 0.05 mg/kg as monotherapy or in combination with Venetoclax plus 5-AZA.
The results depicted in Figure 8 and Figure 9 show significant antitumor activity of WT1-TCB in combination with Venetoclax plus 5-AZA in terms of tumor growth (p = 0.0023; Table 3) and median survival (p = 0.0034; Table 4) as compared with vehicle on Study Day 36. Furthermore, the combination treatment resulted in improved efficacy compared with that achieved by WT1-
TCB monotherapy at 0.05 mg/kg. The median survival was reached on Study Day 26 upon WT1- TCB monotherapy and was increased to 32 days upon treatment with WT1-TCB in combination with Venetoclax + 5-AZA (Figure 9).
5-AZA = 5-azacytidine.
Dunn's test (non-parametric, all scenarios) based on data depicted in Figure 8A.
5-AZA = 5-azacytidine.
Log-rank test (adjusted for multiple testing), based on data depicted in Figure 9.
Summary and conclusions
The aim of this study was to examine the in vivo antitumor activity of WT1-TCB in combination with Venetoclax plus 5-AZA, a scheme used currently as standard of care for acute myeloid leukemia. For this purpose, WT1-TCB was used at a dose of 0.05 mg/kg to provide a therapeutic window for the drug combination effect.
Tumor growth and survival analysis showed significant antitumor activity for WT1-TCB in combination with Venetoclax plus 5-AZA when compared with vehicle on Study Day 36. Furthermore, an improved efficacy was observed when compared with WT1-TCB monotherapy at 0.05 mg/kg.
* * *
Although the foregoing invention has been described in some detail by way of illustration and example for purposes of clarity of understanding, the descriptions and examples should not be construed as limiting the scope of the invention. The disclosures of all patent and scientific literature cited herein are expressly incorporated in their entirety by reference.
Claims
1. A HLA-A2/WT1 x CD3 bispecific antibody for use in the treatment of a cancer in an individual, wherein the treatment comprises administration of the HLA-A2/WT1 x CD3 bispecific antibody in combination with venetoclax/azacitidine.
2. Use of a HLA-A2/WT1 x CD3 bispecific antibody in the manufacture of a medicament for the treatment of cancer in an individual, wherein the treatment comprises administration of the HLA- A2/WT1 x CD3 bispecific antibody in combination with venetoclax/azacitidine.
3. A method for treating cancer in an individual comprising administering to the individual a HLA- A2/WT1 x CD3 bispecific antibody and venetoclax/azacitidine.
4. A kit comprising a first medicament comprising a HLA-A2/WT1 x CD3 bispecific antibody and a second medicament comprising venetoclax and/or a third medicament comprising azacitidine, and optionally further comprising a package insert comprising instructions for administration of the first medicament in combination with the second and/or third medicament for treating cancer in an individual.
5. The HLA-A2/WT1 x CD3 bispecific antibody for use, the use, the method or the kit of any one of the preceding claims, wherein the HLA-A2/WT1 x CD3 bispecific antibody comprises
(i) a first antigen binding moiety that specifically binds to CD3 and comprises a heavy chain variable region comprising the heavy chain CDR (HCDR) 1 of SEQ ID NO: 1, the HCDR2 of SEQ ID NO: 2, and the HCDR3 of SEQ ID NO: 3; and a light chain variable region comprising the light chain CDR (LCDR) 1 of SEQ ID NO: 4, the LCDR2 of SEQ ID NO: 5 and the LCDR3 of SEQ ID NO: 6; and
(ii) a second antigen binding moiety that specifically binds to HLA-A2/WT1 and comprises a heavy chain variable region comprising the heavy chain CDR (HCDR) 1 of SEQ ID NO: 9, the HCDR2 of SEQ ID NO: 10, and the HCDR3 of SEQ ID NO: 11; and a light chain variable region comprising the light chain CDR (LCDR) 1 of SEQ ID NO: 12, the LCDR2 of SEQ ID NO: 13 and the LCDR3 of SEQ ID NO: 14.
6. The HLA-A2/WT1 x CD3 bispecific antibody for use, the use, the method or the kit of any one of the preceding claims, wherein the HLA-A2/WT1 x CD3 bispecific antibody comprises a third antigen binding moiety that specifically binds to HLA-A2/WT1 and/or an Fc domain composed of a first and a second subunit.
7. The HLA-A2/WT1 x CD3 bispecific antibody for use, the use, the method or the kit of any one of the preceding claims, wherein the HLA-A2/WT1 x CD3 bispecific antibody comprises
(i) a first antigen binding moiety that specifically binds to CD3, comprising a heavy chain variable region comprising the heavy chain CDR (HCDR) 1 of SEQ ID NO: 1, the HCDR2 of SEQ ID NO: 2, and the HCDR3 of SEQ ID NO: 3; and a light chain variable region comprising the light chain CDR (LCDR) 1 of SEQ ID NO: 4, the LCDR2 of SEQ ID NO: 5 and the LCDR3 of SEQ ID NO: 6, wherein the first antigen binding moiety is a crossover Fab molecule wherein either the variable or the constant regions of the Fab light chain and the Fab heavy chain are exchanged;
(ii) a second and a third antigen binding moiety that specifically bind to HLA-A2/WT 1 , comprising a heavy chain variable region comprising the heavy chain CDR (HCDR) 1 of SEQ ID NO: 9, the HCDR2 of SEQ ID NO: 10, and the HCDR3 of SEQ ID NO: 11; and a light chain variable region comprising the light chain CDR (LCDR) 1 of SEQ ID NO: 12, the LCDR2 of SEQ ID NO: 13 and the LCDR3 of SEQ ID NO: 14, wherein the second and third antigen binding moiety are each a Fab molecule, particularly a conventional Fab molecule;
(iii) an Fc domain composed of a first and a second subunit, wherein the second antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N-terminus of the Fab heavy chain of the first antigen binding moiety, and the first antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N-terminus of the first subunit of the Fc domain, and wherein the third antigen binding moiety is fused at the C-terminus of the Fab heavy chain to the N-terminus of the second subunit of the Fc domain.
8. The HLA-A2/WT1 x CD3 bispecific antibody for use, the use, the method or the kit of any one of the preceding claims, wherein the first antigen binding moiety of the HLA-A2/WT1 x CD3 bispecific antibody comprises a heavy chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 7 and a light chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 8, and/or the second and (where present) third antigen binding moiety of the HLA-A2/WT1 x CD3 bispecific antibody comprise a heavy chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 15 and a light chain variable region sequence that is at least about 95%, 96%, 97%, 98%, 99% or 100% identical to the amino acid sequence of SEQ ID NO: 16.
9. The HLA-A2/WT1 x CD3 bispecific antibody for use, the use, the method or the kit of any one of the preceding claims, wherein the first antigen binding moiety of the HLA-A2/WT1 x CD3
bispecific antibody is a crossover Fab molecule wherein the variable regions of the Fab light chain and the Fab heavy chain are exchanged, and wherein the second and (where present) third antigen binding moiety of the HLA-A2/WT1 x CD3 bispecific antibody is a conventional Fab molecule wherein in the constant domain CL the amino acid at position 124 is substituted independently by lysine (K), arginine (R) or histidine (H) (numbering according to Kabat) and the amino acid at position 123 is substituted independently by lysine (K), arginine (R) or histidine (H) (numbering according to Kabat) and in the constant domain CHI the amino acid at position 147 is substituted independently by glutamic acid (E), or aspartic acid (D) (numbering according to Kabat EU index) and the amino acid at position 213 is substituted independently by glutamic acid (E), or aspartic acid (D) (numbering according to Kabat EU index).
10. The HLA-A2/WT1 x CD3 bispecific antibody for use, the use, the method or the kit of any one of the preceding claims, wherein the Fc domain of the HLA-A2/WT1 x CD3 bispecific antibody comprises a modification promoting the association of the first and the second subunit of the Fc domain, and/or the Fc domain comprises one or more amino acid substitution that reduces binding to an Fc receptor and/or effector function.
11. The HLA-A2/WT1 x CD3 bispecific antibody for use, the use, the method or the kit of any one of the preceding claims, wherein the cancer is a WT1 -positive cancer.
12. The HLA-A2/WT1 x CD3 bispecific antibody for use, the use, the method or the kit of any one of the preceding claims, wherein the cancer is acute myeloid leukemia (AML).
13. The invention as described hereinbefore.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23151497.7 | 2023-01-13 | ||
| EP23151497 | 2023-01-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024149821A1 true WO2024149821A1 (en) | 2024-07-18 |
Family
ID=85018576
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/050523 Ceased WO2024149821A1 (en) | 2023-01-13 | 2024-01-11 | Treatment of cancer using a hla-a2/wt1 x cd3 bispecific antibody and venetoclax/azacitidine |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2024149821A1 (en) |
Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0404097A2 (en) | 1989-06-22 | 1990-12-27 | BEHRINGWERKE Aktiengesellschaft | Bispecific and oligospecific, mono- and oligovalent receptors, production and applications thereof |
| WO1993001161A1 (en) | 1991-07-11 | 1993-01-21 | Pfizer Limited | Process for preparing sertraline intermediates |
| WO1993016185A2 (en) | 1992-02-06 | 1993-08-19 | Creative Biomolecules, Inc. | Biosynthetic binding protein for cancer marker |
| US5571894A (en) | 1991-02-05 | 1996-11-05 | Ciba-Geigy Corporation | Recombinant antibodies specific for a growth factor receptor |
| US5587458A (en) | 1991-10-07 | 1996-12-24 | Aronex Pharmaceuticals, Inc. | Anti-erbB-2 antibodies, combinations thereof, and therapeutic and diagnostic uses thereof |
| US5731168A (en) | 1995-03-01 | 1998-03-24 | Genentech, Inc. | Method for making heteromultimeric polypeptides |
| US5869046A (en) | 1995-04-14 | 1999-02-09 | Genentech, Inc. | Altered polypeptides with increased half-life |
| US6248516B1 (en) | 1988-11-11 | 2001-06-19 | Medical Research Council | Single domain ligands, receptors comprising said ligands methods for their production, and use of said ligands and receptors |
| WO2019122052A2 (en) | 2017-12-21 | 2019-06-27 | F. Hoffmann-La Roche Ag | Antibodies binding to hla-a2/wt1 |
| WO2021089513A1 (en) * | 2019-11-05 | 2021-05-14 | F. Hoffmann-La Roche Ag | Treatment of cancer using a hla-a2/wt1 x cd3 bispecific antibody and lenalidomide |
| WO2022246244A1 (en) * | 2021-05-21 | 2022-11-24 | Aptevo Research And Development Llc | Dosing regimens for protein therapeutics |
-
2024
- 2024-01-11 WO PCT/EP2024/050523 patent/WO2024149821A1/en not_active Ceased
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6248516B1 (en) | 1988-11-11 | 2001-06-19 | Medical Research Council | Single domain ligands, receptors comprising said ligands methods for their production, and use of said ligands and receptors |
| EP0404097A2 (en) | 1989-06-22 | 1990-12-27 | BEHRINGWERKE Aktiengesellschaft | Bispecific and oligospecific, mono- and oligovalent receptors, production and applications thereof |
| US5571894A (en) | 1991-02-05 | 1996-11-05 | Ciba-Geigy Corporation | Recombinant antibodies specific for a growth factor receptor |
| WO1993001161A1 (en) | 1991-07-11 | 1993-01-21 | Pfizer Limited | Process for preparing sertraline intermediates |
| US5587458A (en) | 1991-10-07 | 1996-12-24 | Aronex Pharmaceuticals, Inc. | Anti-erbB-2 antibodies, combinations thereof, and therapeutic and diagnostic uses thereof |
| WO1993016185A2 (en) | 1992-02-06 | 1993-08-19 | Creative Biomolecules, Inc. | Biosynthetic binding protein for cancer marker |
| US5731168A (en) | 1995-03-01 | 1998-03-24 | Genentech, Inc. | Method for making heteromultimeric polypeptides |
| US7695936B2 (en) | 1995-03-01 | 2010-04-13 | Genentech, Inc. | Knobs and holes heteromeric polypeptides |
| US5869046A (en) | 1995-04-14 | 1999-02-09 | Genentech, Inc. | Altered polypeptides with increased half-life |
| WO2019122052A2 (en) | 2017-12-21 | 2019-06-27 | F. Hoffmann-La Roche Ag | Antibodies binding to hla-a2/wt1 |
| WO2021089513A1 (en) * | 2019-11-05 | 2021-05-14 | F. Hoffmann-La Roche Ag | Treatment of cancer using a hla-a2/wt1 x cd3 bispecific antibody and lenalidomide |
| WO2022246244A1 (en) * | 2021-05-21 | 2022-11-24 | Aptevo Research And Development Llc | Dosing regimens for protein therapeutics |
Non-Patent Citations (24)
| Title |
|---|
| "NCBI GenBank", Database accession no. BAB71849.1 |
| "UniProt", Database accession no. P19544 |
| AUGSBERGER CHRISTIAN ET AL: "Targeting intracellular WT1 in AML with a novel RMF-peptide-MHC-specific T-cell bispecific antibody", BLOOD, vol. 138, no. 25, 23 December 2021 (2021-12-23), US, pages 2655 - 2669, XP093020501, ISSN: 0006-4971, DOI: 10.1182/blood.2020010477 * |
| AUGSBERGER ET AL., BLOOD, 2021 |
| CARTER, J IMMUNOL METH, vol. 248, 2001, pages 7 - 15 |
| CHOTHIALESK, J. MOL. BIOL., vol. 196, 1987, pages 901 - 917 |
| HEELEY, ENDOCR RES, vol. 28, 2002, pages 217 - 229 |
| HINRICHSRESTIFO, NAT BIOTECHNOL, vol. 31, 2013, pages 999 - 1008 |
| HOLLINGER ET AL., PROC NATL ACAD SCI USA, vol. 90, 1993, pages 6444 - 6448 |
| HUDSON ET AL., NAT MED, vol. 9, 2003, pages 129 - 134 |
| HUTCHINGS MARTIN ET AL: "Dose Escalation of HLA-A2-WT1 CD3 T-Cell Bispecic Antibody in a Phase I Study in Patients with Relapsed/Refractory Acute Myeloid Leuk", 2 November 2023 (2023-11-02), XP093146814, Retrieved from the Internet <URL:https://doi.org/10.1182/blood-2023-173302> [retrieved on 20240328] * |
| KABAT ET AL.: "Sequences of Proteins of Immunological Interest,", 1991, PUBLIC HEALTH SERVICE, NATIONAL INSTITUTES OF HEALTH |
| KINDT ET AL.: "Kuby Immunology", 2007, W.H. FREEMAN AND CO, pages: 91 |
| LECLERCQ GABRIELLE ET AL: "Src/lck inhibitor dasatinib reversibly switches off cytokine release and T cell cytotoxicity following stimulation with T cell bispecific antibodies", JOURNAL FOR IMMUNOTHERAPY OF CANCER, vol. 9, no. 7, 29 July 2021 (2021-07-29), pages e002582, XP055883398, Retrieved from the Internet <URL:http://dx.doi.org/10.1136/jitc-2021-002582> DOI: 10.1136/jitc-2021-002582 * |
| LILJEBLAD ET AL., GLYCO J, vol. 17, 2000, pages 323 - 329 |
| MACCALLUM ET AL., J. MOL. BIOL., vol. 262, 1996, pages 732 - 745 |
| no. 1257044-40-8 |
| PEARSON, GENOMICS, vol. 46, 1997, pages 24 - 36 |
| PLUCKTHUN: "The Pharmacology of Monoclonal Antibodies", vol. 113, 1994, SPRINGER-VERLAG, pages: 269 - 315 |
| RIDGWAY ET AL., PROT ENG, vol. 9, 1996, pages 617 - 621 |
| STUBENRAUCH ET AL., DRUG METABOLISM AND DISPOSITION, vol. 38, 2010, pages 84 - 91 |
| VAN DRIESSCHE ET AL., ONCOLOGIST, vol. 17, 2012, pages 250 - 259 |
| W. R. PEARSON: "Effective protein sequence comparison", METH. ENZYMOL, vol. 266, 1996, pages 227 - 258 |
| W. R. PEARSOND. J. LIPMAN: "Improved Tools for Biological Sequence Analysis", PNAS, vol. 85, 1988, pages 2444 - 2448 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20220275093A1 (en) | Treatment of cancer using a hla-a2/wt1 x cd3 bispecific antibody and lenalidomide | |
| TW202221021A (en) | Improved antigen binding receptors | |
| US20230416412A1 (en) | Prevention or mitigation of t-cell engaging agent-related adverse effects | |
| US20220088195A1 (en) | Prevention or mitigation of T-cell bispecific antibody-related adverse effects | |
| US20220168418A1 (en) | Prevention or mitigation of t-cell engaging agent-related adverse effects | |
| CN111212660A (en) | Use of CEA CD3 bispecific antibodies and PD-1 axis binding antagonists in dosage regimens for the treatment of cancer | |
| US20240287193A1 (en) | Treatment of cancer using a hla-a2/wt1 x cd3 bispecific antibody and a 4-1bb (cd137) agonist | |
| US20240316193A1 (en) | Treatment of cancer using a hla-a2/mage-a4 x cd3 bispecific antibody and a 4-1bb (cd137) agonist | |
| WO2024188966A1 (en) | Treatment of cancer using an anti-hla-g/anti-cd3 bispecific antibody and a 4‑1bb (cd137) agonist | |
| WO2024149821A1 (en) | Treatment of cancer using a hla-a2/wt1 x cd3 bispecific antibody and venetoclax/azacitidine | |
| HK40112386A (en) | Treatment of cancer using a hla-a2/wt1 x cd3 bispecific antibody and a 4-1bb (cd137) agonist | |
| HK40071202A (en) | Treatment of cancer using a hla-a2/wt1 x cd3 bispecific antibody and lenalidomide | |
| WO2022090439A1 (en) | TREATMENT OF CANCER USING A CEA CD3 BISPECIFIC ANTIBODY AND A TGFβ SIGNALING INHIBITOR | |
| HK40031518A (en) | Use of a cea cd3 bispecific antibody and a pd-1 axis binding antagonist in a dosage regime to treat cancer |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24700881 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 24700881 Country of ref document: EP Kind code of ref document: A1 |