WO2024077328A1 - Aqueous coating composition - Google Patents
Aqueous coating composition Download PDFInfo
- Publication number
- WO2024077328A1 WO2024077328A1 PCT/AU2023/050891 AU2023050891W WO2024077328A1 WO 2024077328 A1 WO2024077328 A1 WO 2024077328A1 AU 2023050891 W AU2023050891 W AU 2023050891W WO 2024077328 A1 WO2024077328 A1 WO 2024077328A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- coating composition
- basic salt
- epoxy
- aqueous polymer
- silane compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D125/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an aromatic carbocyclic ring; Coating compositions based on derivatives of such polymers
- C09D125/02—Homopolymers or copolymers of hydrocarbons
- C09D125/04—Homopolymers or copolymers of styrene
- C09D125/08—Copolymers of styrene
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D125/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an aromatic carbocyclic ring; Coating compositions based on derivatives of such polymers
- C09D125/02—Homopolymers or copolymers of hydrocarbons
- C09D125/04—Homopolymers or copolymers of styrene
- C09D125/08—Copolymers of styrene
- C09D125/14—Copolymers of styrene with unsaturated esters
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J3/00—Processes of treating or compounding macromolecular substances
- C08J3/24—Crosslinking, e.g. vulcanising, of macromolecules
- C08J3/26—Crosslinking, e.g. vulcanising, of macromolecules of latex
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/18—Oxygen-containing compounds, e.g. metal carbonyls
- C08K3/24—Acids; Salts thereof
- C08K3/26—Carbonates; Bicarbonates
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/32—Phosphorus-containing compounds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D131/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an acyloxy radical of a saturated carboxylic acid, of carbonic acid, or of a haloformic acid; Coating compositions based on derivatives of such polymers
- C09D131/02—Homopolymers or copolymers of esters of monocarboxylic acids
- C09D131/04—Homopolymers or copolymers of vinyl acetate
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D133/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Coating compositions based on derivatives of such polymers
- C09D133/04—Homopolymers or copolymers of esters
- C09D133/06—Homopolymers or copolymers of esters of esters containing only carbon, hydrogen and oxygen, the oxygen atom being present only as part of the carboxyl radical
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D133/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Coating compositions based on derivatives of such polymers
- C09D133/04—Homopolymers or copolymers of esters
- C09D133/06—Homopolymers or copolymers of esters of esters containing only carbon, hydrogen and oxygen, the oxygen atom being present only as part of the carboxyl radical
- C09D133/08—Homopolymers or copolymers of acrylic acid esters
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D167/00—Coating compositions based on polyesters obtained by reactions forming a carboxylic ester link in the main chain; Coating compositions based on derivatives of such polymers
- C09D167/08—Polyesters modified with higher fatty oils or their acids, or with natural resins or resin acids
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/02—Emulsion paints including aerosols
- C09D5/024—Emulsion paints including aerosols characterised by the additives
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/16—Antifouling paints; Underwater paints
- C09D5/1656—Antifouling paints; Underwater paints characterised by the film-forming substance
- C09D5/1662—Synthetic film-forming substance
- C09D5/1668—Vinyl-type polymers
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
- C09D7/60—Additives non-macromolecular
- C09D7/63—Additives non-macromolecular organic
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2325/00—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an aromatic carbocyclic ring; Derivatives of such polymers
- C08J2325/02—Homopolymers or copolymers of hydrocarbons
- C08J2325/04—Homopolymers or copolymers of styrene
- C08J2325/08—Copolymers of styrene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2331/00—Characterised by the use of copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an acyloxy radical of a saturated carboxylic acid, or carbonic acid, or of a haloformic acid
- C08J2331/02—Characterised by the use of omopolymers or copolymers of esters of monocarboxylic acids
- C08J2331/04—Homopolymers or copolymers of vinyl acetate
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2333/00—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Derivatives of such polymers
- C08J2333/04—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Derivatives of such polymers esters
- C08J2333/06—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Derivatives of such polymers esters of esters containing only carbon, hydrogen, and oxygen, the oxygen atom being present only as part of the carboxyl radical
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2367/00—Characterised by the use of polyesters obtained by reactions forming a carboxylic ester link in the main chain; Derivatives of such polymers
- C08J2367/08—Polyesters modified with higher fatty oils or their acids, or with resins or resin acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/18—Oxygen-containing compounds, e.g. metal carbonyls
- C08K3/24—Acids; Salts thereof
- C08K3/26—Carbonates; Bicarbonates
- C08K2003/262—Alkali metal carbonates
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/32—Phosphorus-containing compounds
- C08K2003/321—Phosphates
- C08K2003/324—Alkali metal phosphate
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/09—Carboxylic acids; Metal salts thereof; Anhydrides thereof
- C08K5/098—Metal salts of carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/54—Silicon-containing compounds
- C08K5/541—Silicon-containing compounds containing oxygen
- C08K5/5435—Silicon-containing compounds containing oxygen containing oxygen in a ring
Definitions
- the present invention relates in general to aqueous coating compositions.
- the aqueous coating compositions are well suited to being formulated for use as a water-based or aqueous decorative paint and the invention will be described with particular reference to that application.
- the aqueous coating compositions can be used in any application that calls for a substrate to be coated with a composition that forms a polymeric fdm.
- Aqueous coating compositions typically comprise an aqueous polymer dispersion, commonly referred to in the art as a latex.
- the latex component functions as a binder in such compositions, for example in water based decorative paint compositions.
- An aqueous polymer dispersion or latex is also known in the art simply as a binder.
- Decorative paints are those which are typically applied to architectural features and the like, and are most often used in households for application to both interior and exterior surfaces.
- the term "decorative paints” is intended to classify paints that do not need to be subjected to high temperatures (i.e. higher than ambient temperatures) after their application to afford a polymeric film having the requisite physical and mechanical properties fortheir intended application. Accordingly, such paints are to be distinguished from those paints, such as industrial coatings, that typically require high temperatures to enable the applied paint to form a polymeric film having the requisite physical and mechanical properties.
- Decorative paints are generally used to provide both aesthetic appeal and/or a degree of protection to a substrate.
- the aesthetic appeal and protection are provided by the polymeric film which forms on the substrate when the paint dries.
- stain resistance is an important parameter by which the quality of a polymeric film derived from a decorative paint is often measured. Assessment of such a parameter in effect provides an indication of how a paint film may perform under conditions which decorative paint films are typically exposed. For example, substrates coated with decorative paints are often exposed to domestic staining materials including food, oil or grease and beverages such as coffee and red wine.
- Paint films might also be susceptible to staining from the substrate upon which they have been coated, especially in the case of timber substrates containing tannins.
- the tannin material can "print through" the paint film, discolouring the paint after it has dried. That problem is particularly evident with water based latex paints.
- Various factors can influence the prevalence of substrate staining and special water based latex paints have been developed to overcome that problem.
- the present invention is not particularly concerned with that type of staining but rather staining on the outermost surface of a paint film. Paints that perform well for reducing substrate staining do not necessarily exhibit good resistance to domestic stains and vice versa.
- staining materials can be conveniently categorised into hydrophilic and hydrophobic materials.
- oil, grease, crayon, pastel and lipstick are considered in the art as hydrophobic staining materials
- red wine, coffee and waterborne drawing markers are considered as hydrophilic staining materials.
- stain resistance is, unless otherwise stated, intended to be a reference to both hydrophilic and hydrophobic stain resistance.
- Polymeric films formed from conventional organic solvent based paints such as those which employ alkyd resins as binders, are renowned for their excellent stain resistance. Such performance of those paint films is believed to be primarily derived from the paint's ability to form a highly crosslinked polymeric film upon drying.
- those paints present numerous disadvantages in that they have a high volatile organic content (VOC), require long recoat times and require the use of organic solvents during clean up.
- VOC volatile organic content
- aqueous based coating compositions that can, for example, be applied in the field of decorative paints and provide for polymeric films that exhibit improved stain resistance.
- the present invention provides an aqueous coating composition
- aqueous coating composition comprising: epoxy functionalised silane compound; aqueous polymer dispersion having functionality capable of reacting with the epoxy functionality of the epoxy functionalised silane compound; and non-surfactant basic salt in an amount ranging from 0.25 wt.% to 5 wt.%.
- the present invention also provides a kit for use in producing a coating composition, the kit comprising: a first part comprising epoxy functionalised silane compound; and a second part comprising non-surfactant basic salt and aqueous polymer dispersion having functionality capable of reacting with the epoxy functionality of the epoxy functionalised silane compound; wherein the first and second parts are combined to produce the coating composition in which the non-surfactant basic salt is present in an amount ranging from 0.25 wt.% to 5 wt.%.
- the present invention further provides a method of producing a coating composition, the method comprising combining together: epoxy functionalised silane compound; aqueous polymer dispersion having functionality capable of reacting with the epoxy functionality of the epoxy functionalised silane compound; and non-surfactant basic salt; to thereby produce the coating composition in which the non-surfactant basic salt is present in an amount ranging from 0.25 wt.% to 5 wt.%.
- coating compositions comprising (i) epoxy functionalised silane, (ii) aqueous polymer dispersion having functionality capable of reacting with the epoxy functionality of the epoxy functionalised silane compound, and (iii) non-surfactant basic salt in an amount ranging from 0.25 wt.% to 5 wt.% produce a polymeric film that exhibits improved stain resistance relative to a polymeric film formed from the same composition absent the defined amount of non-surfactant basic salt.
- the non-surfactant basic salt is outside the specified range the so formed polymer film has practically no improvement in stain resistance properties or presents even worse stain resistance properties.
- the coating compositions in accordance with the invention are particularly suitable for use in decorative aqueous paint formulations.
- Polymeric films formed from those decorative paint formulations exhibit corresponding improved stain resistance.
- the presence of the non-surfactant basic salt in the specified amount promotes enhanced reactivity between the epoxy functionalised silane compound and the aqueous polymer dispersion that in turn forms a cross-linked network.
- the non-surfactant basic salt may also promote enhanced self-reactivity of the epoxy functionalised silane compound that in turn can also promote the formation of a cross-linked network.
- the overall enhanced cross-linked network of a polymeric fdm derived from the coating composition is believed to impart improved stain resistance to the polymeric film.
- the specified amount used seems to provide a unique balance of crosslinking that has been found optimum for imparting stain resistance to the so formed polymeric film.
- aqueous coating compositions in accordance with the invention can advantageously be used in any application that calls for a substrate to be coated with a composition that forms a polymeric film.
- the aqueous coating compositions are well suited to being formulated for use as a water-based (aqueous) decorative paint and the invention will be described with particular reference toward that application. Such emphasis is not intended to be a limitation on the scope of application for the invention.
- the present invention provides for an aqueous coating composition.
- compositions in accordance with the invention are of a type suitable for application to a substrate. After application to the substrate, water within the composition evaporates and/or is absorbed into the substrate to leave behind a polymeric film on the substrate surface. Those skilled in the art will appreciate that formation of the polymeric film will typically be the intended ultimate product of the coating composition.
- aqueous coating compositions in accordance with the invention can advantageously be used in any application that calls for a substrate to be coated with a composition that forms a polymeric film. While the aqueous coating compositions are described herein with particular reference to being formulated for use as a water-based (aqueous) decorative paint, such emphasis is not intended to be a limitation on the scope of application for the invention.
- the aqueous coating composition comprises an epoxy functionalised silane compound.
- an epoxy functionalised silane means the compound has covalently bound thereto one or more epoxy functional groups (i.e. epoxy functionality).
- the epoxy functionalised silane compound may have a molecular structure comprising the moiety of formula (I): where R' is selected from optionally substituted: alkyl, aryl and -Si(0R')(R")(R-CHCH20), R" and R" are each independently selected from optionally substituted: alkyl, aryl, oxyalkyl and oxyaryl, and R is selected from optionally substituted: alkyl and aryl.
- the epoxy functionalised silane compound may be monomeric, oligomeric or polymeric.
- the epoxy functionalised silane compound is monomeric.
- the epoxy functionalised silane compound is oligomeric.
- the epoxy functionalised silane compound is polymeric.
- formula (I) may be represented as formula (IA): where R is selected from optionally substituted: alkyl, aryl, R" and R'" are each independently selected from optionally substituted: alkyl, aryl, oxyalkyl and oxyaryl, R is selected from optionally substituted: alkyl and aryl, and n is an integer ranging from 2 to about 100, or from 2 to about 70, or from 2 to about 50, or from 2 to about 30, or from 2 to about 15, or from 2 to about 10.
- n in formula (IA) will typically range from 2 to 10, or from 2 to 8, or from 2 to 5.
- n in formula (IA) will typically range from 11 to about 100, or from 11 to about 70, or from 11 to about 50, or from 11 to about 30, or from 11 to about 20.
- R, R', R" or R' include an alkyl or aryl group it may be independently selected from optionally substituted: Ci-io alkyl and C6-12 aryl.
- R in formulae (I) or (IA) is -(CH2)n-O-(CH2)n-, where n is an integer ranging from 1 to 10, or form 1 to 5.
- Epoxy functionalised silane compounds suitable for use in accordance with the invention are commercially available.
- the functional silane compound will generally be present in an amount ranging from about 0.5 wt. % to about 10 wt. %, or from about 0.5 wt. % to about 5 wt. %, or from about 1 wt. % to about 5 wt. %, or from about Iwt. % to about 3 wt. %, or from about 1.5 wt. % to about 2 wt. %.
- an important feature of the epoxy functionalised silane compound is its epoxy functionality. Through that functionality, the silane compound can take on a role of a cross-linking agent to facilitate cross-linking of the aqueous polymer dispersion.
- the aqueous polymer dispersion has functionality capable of reacting with the epoxy functionality of the epoxy functionalised silane compound.
- aqueous polymer dispersion having "functionality" is intended to mean it comprises a functional group.
- that functionality or functional group is one that is to be capable of reacting with epoxy functionality of the epoxy functionalised silane compound as described herein.
- functionality of the aqueous polymer dispersion provided it is capable of reaction with the epoxy functionality of the epoxy functionalised silane compound.
- Aqueous polymer dispersions are essentially dispersions of polymer particles throughout an aqueous medium. Such dispersion are often referred to as a latex and they are commonly used as (and called) binders in water-based coating compositions such as decorative paint formulations.
- the main aim of coating compositions is typically the formation of a polymeric film on the surface of a substrate on which the coating composition has been applied.
- a latex based composition comprises discrete, normally spherical, particles of high molecular weight polymer.
- the polymer particles overcome their coulombic and/or steric repulsions and begin to flatten and coalesce.
- coalesce or “coalescence” it is meant that the polymer particles flow together to form a continuous film in which individual particles have lost their identity.
- Polymer particle coalescence plays an important role in the polymeric film formation process of aqueous coating compositions. Those skilled in the art are well familiar with producing aqueous polymer dispersions and their subsequent formulation into coating compositions to produce polymeric film forming coating compositions.
- aqueous polymer dispersions used in accordance with the invention are intended to be film forming (i.e. a film forming aqueous polymer dispersion).
- the present invention can advantageously be performed using a diverse range of aqueous polymer dispersions.
- aqueous polymer dispersions include, but are not limited to, acrylic (e.g. styrene -acrylic copolymer, vinyl acetate -acrylic copolymer, urethane -acrylic copolymer and alkyd-acrylic copolymer) and vinyl acetate-ethylene copolymer aqueous polymer dispersions.
- reference to an acrylic aqueous polymer dispersion embraces all aqueous polymer dispersions that comprise polymer formed using (meth)acrylic acid monomer and/or (meth)acrylic ester (e.g. (meth)acrylates) monomer. Accordingly, the reference to an acrylic aqueous polymer dispersion herein embraces acrylic copolymer aqueous polymer dispersions such as styrene-acrylic copolymer, vinyl acetate-acrylic copolymer, urethane -acrylic copolymer and alkyd-acrylic copolymer aqueous polymer dispersions.
- aqueous polymer dispersions might therefore also be described to include, but are not limited to, acrylic and vinyl acetate-ethylene copolymer aqueous polymer dispersions.
- the aqueous polymer dispersion has functionality capable of reaction with the epoxy functionality of the epoxy functionalised silane compound.
- the aqueous polymer dispersion can react with the epoxy silane compound to facilitate formation of a cross-linked polymer network.
- the epoxy silane compound may be described as a cross-linking agent that promotes cross-linking of polymer that forms the aqueous polymer dispersion to create a cross-linked polymer network in the polymeric fdm derived from the coating composition.
- Suitable functionality of the aqueous polymer dispersion include, but are not limited to, carboxylic acid, phosphoric acid, sulphuric acid, sulfonic acid, salts thereof, amino and hydroxyl.
- aqueous polymer dispersions can be easily tailored as part of a conventional means of their production.
- monomer for example, monomer
- Rule 26 that is polymerised to produce the aqueous polymer dispersion can be selected so as to present the required functionality as either a pendant and/or terminal functional groups of the so formed polymer chains.
- Monomers carrying the required functionality that are polymerised to produce the aqueous polymer dispersions will typically be used in an amount of about 1 wt. % to about 20 wt. %, 1 wt. % to about 15 wt. %, 1 wt. % to about 10 wt. %, or form about 1 wt.% to about 5 wt. %, relative to the total amount of monomer polymerised.
- polymer that forms the aqueous polymer dispersion will have a molecular weight in the range of about 10,000 to about 1,000,000, or from about 50, 000 to about 500,000.
- molecular weight is intended to be a weight average molecular weight (Mw) as measured by gel permeation chromatography.
- aqueous polymer dispersion used in accordance with the invention can advantageously be prepared using conventional techniques, apparatus and reagents well known to those skilled in the art.
- the dispersed polymer particles that make up the aqueous polymer dispersion will generally be formed through the polymerisation of ethylenically unsaturated monomers.
- Such monomers will generally be selected from those which (i) can provide the required functionality, and (ii) are capable of undergoing free radical polymerisation in an aqueous environment.
- the dispersed polymer particles will comprise at least two different polymerised ethylenically unsaturated monomers in the form of a copolymer.
- a wide range of ethylenically unsaturated monomers may be used to form the functional aqueous polymer dispersion.
- the monomers will generally be capable of being polymerised with other monomers.
- the factors which determine copolymerisability of various monomers are well documented in the art. For example, see: Greenlee, R.Z., in Polymer Handbook 3 rd Edition (Brandup, J., and Immergut. E.H. Eds) Wiley: New York, 1989 p 11/53.
- Suitable ethylenically unsaturated monomers include those of general formula (II):
- V is selected from hydrogen, R 2 , -CO 2 H, -CO2R 2 , -COR 2 , -CSR 2 , -CSOR 2 , -COSR 2 , - CONH2, -CONHR 2 , -CONR 2 2, -OR 2 , -SR 2 , -O2CR 2 , -SCOR 2 , and -OCSR 2 ;
- R 2 is selected from optionally substituted Ci-Cis alkyl, optionally substituted C2- Ci8 alkenyl, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted carbocyclyl, optionally substituted heterocyclyl, optionally substituted aralkyl, optionally substituted heteroarylalkyl, optionally substituted alkaryl, optionally substituted alkylheteroaryl and polymer chains, wherein the optional substituents are independently selected from alkyleneoxidyl (epoxy), hydroxy, alkoxy, acyl, acyl
- Suitable mono-ethylenically unsaturated monomers include, but are not limited to, the following: methyl methacrylate, ethyl methacrylate, propyl methacrylate (all isomers), butyl methacrylate (all isomers), 2-ethylhexyl methacrylate, isobomyl methacrylate, methacrylic acid, benzyl methacrylate, phenyl methacrylate, methacrylonitrile, alpha-methylstyrene, methyl acrylate, ethyl acrylate, propyl acrylate (all isomers), butyl acrylate (all isomers), 2-ethylhexyl acrylate, isobomyl acrylate, acrylic acid, benzyl acrylate, phenyl acrylate, acrylonitrile, styrene, functional methacrylates, acrylates and styrenes selected from glycidyl
- Rule 26 methacrylate, triethyleneglycol methacrylate, itaconic anhydride, itaconic acid, glycidyl acrylate, 2-hydroxyethyl acrylate, hydroxypropyl acrylate (all isomers), hydroxybutyl acrylate (all isomers), N,N-dimethylaminoethyl acrylate, N,N -diethylaminoethyl acrylate, triethyleneglycol acrylate, methacrylamide, N-methylacrylamide, N,N -dimethylacrylamide, N- tert-butyhnethacrylamide, N-n-butylmethacrylamide, N-methylolmethacrylamide, N- ethylolmethacrylamide, N-tert-butylacrylamide, N-n-butylacrylamide, N-methylolacrylamide, N-ethylolacrylamide, vinyl benzoic acid (all isomers), diethyl acrylate
- the aqueous polymer dispersion will generally be present in an amount ranging from about 10 wt. % to about 85 wt. %.
- the aqueous coating composition also comprises non-surfactant basic salt in an amount ranging from 0.25 wt.% to 5 wt.%.
- non-surfactant basic salt By being a "non-surfactant" basic salt is intended to mean the basic salt does not comprise a fatty organic component that imparts surfactant like properties to the salt. Those skilled in the art will be able to readily characterise basic salts as being surfactant or non-surfactant in character.
- a non-surfactant basic salt will typically be a basic salt that has less than 10, or less than 8, or less than 6, or less than 4, including 0, carbon atoms.
- the non-surfactant basic salt may therefore also be expressed as a C0-9 basic salt, a C0-7 basic salt, a Co-5 basic salt, or a Co-3 basic salt.
- a “basic salt” is defined as being a reaction product derived from a weak acid and (i) a strong base or (ii) a weak base.
- a weak acid is an acid with a pKa value above 1
- a strong base is a base with a pKb value below 1
- a weak base is a base having a pKb value higher than the pKa value of the weak acid.
- a basic salt is only derived from a weak acid and a weak base if the Kb of the weak base is larger than the Ka of the weak acid.
- the presence of the non-surfactant basic salt in an amount ranging from 0.25 wt.% to 5 wt.% promotes at least enhanced reactivity between the epoxy silane compound and the aqueous polymer dispersion, which in turn forms a more extensive cross-linked network.
- the non-surfactant basic salt may also promote enhanced self -reactivity of the epoxy silane compound, which in turn can also promote the formation of a more extensive cross-linked network.
- the overall enhanced cross-linked network of a polymeric fdm derived from the coating composition is believed to impart improved stain resistance to the polymeric film.
- surfactant-like basic salts and also non-surfactant basic salts used in an amount less than 0.25 wt % or greater than 5 wt. % were not found to impart the improved stain resistance demonstrated by the present invention.
- the specified amount used seems to provide a unique balance of crosslinking that has been found optimum for imparting stain resistance to the so formed polymeric film.
- the present invention can advantageously be performed using a diverse range of non-surfactant basic salts, examples of which would be well known to those skilled in the art.
- non-surfactant basic salts include, but are not limited to, benzoate, phosphate dibasic, acetate and carbonate salts.
- the non-surfactant basic salts used in accordance with the invention will of course comprise a counter ion(s). There is no particular limitation on the type of counter cation that may be present.
- Suitable cations include, but are not limited to, sodium, potassium and lithium.
- the non-surfactant basic salt is present in an amount of at least about 0.3 wt. %, or at least about 0.35 wt. %, or at least about 0.4 wt. %, or at least about 0.45 wt. %, or at least about 0.5 wt. %, or at least about 0.75 wt.%, or at least about 1 wt.%.
- the non-surfactant basic salt may be present in an amount ranging from at least 0.25 wt. % to about 4 wt. %, or from at least 0.25 wt. % to about 3 wt. %, or from at least 0.25 wt. % to about 2 wt. %, or from about 0.3 wt. % to about 1.5 wt. %.
- the lower limit of those ranges may also include the lower limits of any one of those outlined in the paragraph directly above.
- the wt. % of the epoxy silane compound, aqueous polymer dispersion, and non-surfactant basic salt described herein is intended to be that present in the aqueous coating composition comprising all three components and formulated for application to a substrate.
- the rate of reaction between constituent components of the aqueous coating compositions in accordance with the invention i.e. the functional silane compound, the functional aqueous polymer dispersion and the non-surfactant basic salt
- the rate of reaction between constituent components of the aqueous coating compositions in accordance with the invention will vary depending upon a number of factors such as the specific composition of each constituent component and temperature.
- aqueous coating composition Upon combining the constituent components it will generally be desirable to apply the aqueous coating composition to a substrate in a timeframe before the cross-linked polymer network forms to such an extent that the composition begins to gel.
- a timeframe is commonly known in the art as the pot life of the composition.
- the pot life can vary greatly depending on the constituent components used to make the composition.
- the pot life may be about 5 days, or about 10 days, or about 15 days, or about 20 days, or possibly longer such as about 3 weeks, 5 weeks, 2 months, 4 months, 6 months, 8
- aqueous coating compositions in accordance with the invention may be desirable to present the constituent components in a multipart form, for example a two-part form.
- the epoxy silane compound can react with the aqueous polymer dispersion in the absence or presence of the non-surfactant basic salt at commercially undesirable rate, it may be desirable to present the epoxy silane compound within the kit as a first isolated part that can be added to a second part comprising the aqueous polymer dispersion.
- the non-surfactant basic salt may be provided in the first and/or second part.
- the present invention therefore provides a kit for use in producing a coating composition, the kit comprising: a first part comprising epoxy silane compound; and a second part comprising nonsurfactant basic salt and aqueous polymer dispersion having functionality capable of reacting with the epoxy functionality of the epoxy silane compound; wherein when the first and second parts are combined to produce the coating composition the non-surfactant basic salt is present in an amount ranging from 0.25 wt.% to 5 wt.%.
- the present invention also provides a kit for use in producing a coating composition, the kit comprising: a first part comprising epoxy silane compound and non-surfactant basic salt; and a second part comprising aqueous polymer dispersion having functionality capable of reacting with the epoxy functionality of the epoxy silane compound; wherein when the first and second parts are combined to produce the coating composition the non-surfactant basic salt is present in an amount ranging from 0.25 wt.% to 5 wt.%.
- the present invention further provides a kit for use in producing a coating composition, the kit comprising: a first part comprising epoxy silane compound; and a second part comprising aqueous polymer dispersion having functionality capable of reacting with the epoxy functionality of the epoxy silane compound; wherein the first and/or second part further comprises a non-surfactant basic salt and when the first and second parts are combined to produce the coating composition the non-surfactant basic salt is present in an amount ranging from 0.25 wt.% to 5 wt.%.
- crosslinking in the polymeric film produced from the coating composition is believed to be important at least in terms of deriving the improved stain resistance.
- other crosslinking agents for example isocyanate compounds
- Such other crosslinking agents can also adversely affect (reduce) the pot life of the compositions, for example by making the pot life shorter than the desired recoat time of the coating composition.
- Other crosslinking agents, for example isocyanate compounds may also give rise to micro-foaming during the curing stage that can compromise the surface finish of the final coating.
- the coating composition does not include another crosslinking agent.
- the coating composition does not include an isocyanate compound.
- the present invention also provides a method of producing a coating composition, the method comprising combining together: epoxy silane compound; aqueous polymer dispersion having functionality capable of reacting with the epoxy functionality of the epoxy silane compound; and non-surfactant basic salt; to thereby produce the coating composition in which the nonsurfactant basic salt is present in an amount ranging from 0.25 wt.% to 5 wt.%.
- composition comprising the aqueous polymer dispersion and the non-
- composition comprising the aqueous polymer dispersion and the non-surfactant basic salt is prepared by combining the non-surfactant basic salt with preformed aqueous polymer dispersion.
- references herein to "preformed" aqueous polymer dispersion is intended to mean aqueous polymer dispersion that has first been prepared and into which is added the non-surfactant basic salt.
- non-surfactant basic salt has not been added during the process of preparing the aqueous polymer dispersion.
- a composition comprising the epoxy silane compound and the nonsurfactant basic salt is combined with the aqueous polymer dispersion.
- Polymeric film produced from the aqueous coating compositions in accordance with the invention advantageously exhibits improved stain resistance, relative to the same composition absent the required non-surfactant basic salt.
- the degree of stain resistance improvement achieved will vary depending on the composition formulation, with optimal performance dependent on a number of factors. Those skilled in the art will appreciate the need to systematically vary multiple components to achieve an optimal balance of performance across relevant different measures, along with cost. Those skilled in the art will also appreciate that Design of Experiments (DoE) can be readily applied to such multidimensional variable to obtain an optimised composition formulation.
- DoE Design of Experiments
- aqueous coating compositions in accordance with the invention may also comprise one or more other constituent components (additives) commonly used in the art.
- additives include, but are not limited to, thickeners, pigments, extenders, matting agents, rheology modifiers, wet edge extenders, bases, dispersants, opaque polymers, coalescing agents, antimicrobial agents, UV absorbers, and tinting agents.
- the coating composition further comprises one or more additives selected from thickeners, pigments, extenders, matting agents, rheology modifiers, wet edge extenders, bases, dispersants, opaque polymers, coalescing agents, antimicrobial agents, UV absorbers, and
- the amount of additives used in the aqueous coating compositions are well known to those skilled in the art.
- the amount of pigment used commonly expressed in terms of the pigment volume concentration (PVC) will generally range in an amount from about 10% to about 90%.
- the PVC may be determined according to the following equation:
- alkyl used either alone or in compound words denotes straight chain, branched or cyclic alkyl, for example C1-20 alkyl, e.g. C1-10 or C1-6.
- straight chain and branched alkyl include methyl, ethyl, w-propyl. isopropyl, w-butyl. scc-butyl.
- cyclic alkyl examples include mono- or polycyclic alkyl groups such as cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl, cyclodecyl and the like. Where an alkyl group is referred to generally as "propyl", butyl” etc, it will be understood that this can refer to any of straight, branched and cyclic isomers where appropriate. An alkyl group may be optionally substituted by one or more optional substituents as herein defined.
- alkenyl denotes groups formed from straight chain, branched or cyclic hydrocarbon residues containing at least one carbon to carbon double bond including ethylenically mono-, di- or polyunsaturated alkyl or cycloalkyl groups as previously defined, for example C2-20 alkenyl (e.g. C2-10 or C2-6).
- alkenyl examples include vinyl, allyl, 1- m ethyl vinyl, butenyl, iso-butenyl, 3-methyl-2-butenyl, 1 -pentenyl, cyclopentenyl, 1 -methyl - cyclopentenyl, 1-hexenyl, 3-hexenyl, cyclohexenyl, 1-heptenyl, 3-heptenyl, 1-octenyl, cyclooctenyl, 1-nonenyl, 2-nonenyl, 3-nonenyl, 1-decenyl, 3-decenyl, 1,3-butadienyl, 1,4- pentadienyl, 1,3-cyclopentadienyl, 1,3-hexadienyl, 1,4-hexadienyl, 1,3 -cyclohexadienyl, 1,4- cyclohexadienyl, 1,3 -cycloheptadien
- alkynyl denotes groups formed from straight chain, branched or cyclic hydrocarbon residues containing at least one carbon-carbon triple bond including ethylenically mono-, di- or polyunsaturated alkyl or cycloalkyl groups as previously defined. Unless the number of carbon atoms is specified the term preferably refers to C2-20 alkynyl (e.g. C2-10 or C2-6). Examples include ethynyl, 1-propynyl, 2-propynyl, and butynyl isomers, and pentynyl isomers. An alkynyl group may be optionally substituted by one or more optional substituents as herein defined.
- halogen denotes fluorine, chlorine, bromine or iodine (fluoro, chloro, bromo or iodo). Preferred halogens are chlorine, bromine or iodine.
- aryl denotes any of single, polynuclear, conjugated and fused residues of aromatic hydrocarbon ring systems (e.g. Ce-18 aryl).
- aryl include phenyl, biphenyl, terphenyl, quaterphenyl, naphthyl, tetrahydronaphthyl, anthracenyl, dihydroanthracenyl, benzanthracenyl, dibenzanthracenyl, phenanthrenyl, fluorenyl, pyrenyl, idenyl, azulenyl, chrysenyl.
- aryl include phenyl and naphthyl.
- An aryl group may or may not be optionally substituted by one or more optional substituents as herein defined.
- arylene is intended to denote the divalent form of aryl.
- carbocyclyl includes any of non-aromatic monocyclic, polycyclic, fused or conjugated hydrocarbon residues, for example C3-20 (e.g. C3-10 or C3-8).
- the rings may be saturated, e.g. cycloalkyl, or may possess one or more double bonds (cycloalkenyl) and/or one or more triple bonds (cycloalkynyl).
- Particularly preferred carbocyclyl moieties are 5-6- membered or 9-10 membered ring systems.
- Suitable examples include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl, cyclodecyl, cyclopentenyl,
- heterocyclyl when used alone or in compound words includes any of monocyclic, polycyclic, fused or conjugated hydrocarbon residues, for example C3-20 (e.g. C3-10 or C3-8) wherein one or more carbon atoms are replaced by a heteroatom so as to provide a non-aromatic residue.
- Suitable heteroatoms include O, N, S, P and Se, particularly O, N and S. Where two or more carbon atoms are replaced, this may be by two or more of the same heteroatom or by different heteroatoms.
- the heterocyclyl group may be saturated or partially unsaturated, i.e. possess one or more double bonds. Particularly preferred heterocyclyl are 5-6 and 9-10 membered heterocyclyl.
- heterocyclyl groups may include azridinyl, oxiranyl, thiiranyl, azetidinyl, oxetanyl, thietanyl, 2H-pyrrolyl, pyrrolidinyl, pyrrolinyl, piperidyl, piperazinyl, morpholinyl, indolinyl, imidazolidinyl, imidazolinyl, pyrazolidinyl, thiomorpholinyl, dioxanyl, tetrahydrofuranyl, tetrahydropyranyl, tetrahydropyrrolyl, tetrahydrothiophenyl, pyrazolinyl, dioxalanyl, thiazolidinyl, isoxazolidinyl, dihydropyranyl, oxazinyl, thiazinyl, thiomorpholinyl, oxathianyl, dithi
- heteroaryl includes any of monocyclic, polycyclic, fused or conjugated hydrocarbon residues, wherein one or more carbon atoms are replaced by a heteroatom so as to provide an aromatic residue.
- Preferred heteroaryl have 3-20 ring atoms, e.g. 3-10.
- Particularly preferred heteroaryl are 5-6 and 9-10 membered bicyclic ring systems.
- Suitable heteroatoms include, O, N, S, P and Se, particularly O, N and S. Where two or more carbon atoms are replaced, this may be by two or more of the same heteroatom or by different heteroatoms.
- heteroaryl groups may include pyridyl, pyrrolyl, thienyl, imidazolyl, furanyl, benzothienyl, isobenzothienyl, benzofuranyl, isobenzofuranyl, indolyl, isoindolyl, pyrazolyl, pyrazinyl, pyrimidinyl, pyridazinyl, indolizinyl, quinolyl, isoquinolyl, phthalazinyl, 1,5-naphthyridinyl, quinozalinyl, quinazolinyl, quinolinyl, oxazolyl, thiazolyl, isothiazolyl, isoxazolyl, triazolyl, oxadialzolyl, oxatriazolyl, triazinyl, and furazanyl.
- a heteroaryl group may be optionally substituted by one or more optional substituents as
- Preferred acyl includes C(O)-R e , wherein R e is hydrogen or an alkyl, alkenyl, alkynyl, aryl, heteroaryl, carbocyclyl, or heterocyclyl residue.
- R e is hydrogen or an alkyl, alkenyl, alkynyl, aryl, heteroaryl, carbocyclyl, or heterocyclyl residue.
- Examples of acyl include formyl, straight chain or branched alkanoyl (e.g.
- C1-20 such as acetyl, propanoyl, butanoyl, 2-methylpropanoyl, pentanoyl, 2,2-dimethylpropanoyl, hexanoyl, heptanoyl, octanoyl, nonanoyl, decanoyl, undecanoyl, dodecanoyl, tridecanoyl, tetradecanoyl, pentadecanoyl, hexadecanoyl, heptadecanoyl, octadecanoyl, nonadecanoyl and icosanoyl; cycloalkylcarbonyl such as cyclopropylcarbonyl cyclobutylcarbonyl, cyclopentylcarbonyl and cyclohexylcarbonyl; aroyl such as benzoyl, toluoyl and naphthoyl; aralkanoyl
- phenylacetyl phenylpropanoyl, phenylbutanoyl, phenylisobutylyl, phenylpentanoyl and phenylhexanoyl
- naphthylalkanoyl e.g. naphthylacetyl, naphthylpropanoyl and naphthylbutanoyl]
- aralkenoyl such as phenylalkenoyl (e.g.
- phenylpropenoyl e.g., phenylbutenoyl, phenylmethacryloyl, phenylpentenoyl and phenylhexenoyl and naphthylalkenoyl (e.g.
- aryloxyalkanoyl such as phenoxyacetyl and phenoxypropionyl
- arylthiocarbamoyl such as phenylthiocarbamoyl
- arylglyoxyloyl such as phenylglyoxyloyl and naphthylglyoxyloyl
- arylsulfonyl such as phenylsulfonyl and napthylsulfonyl
- heterocycliccarbonyl heterocyclicalkanoyl such as thienylacetyl, thienylpropanoyl, thienylbutanoyl, thienylpentanoyl, thienylhexanoyl, thiazolylacetyl, thiadiazolylacetyl and tetrazolylacetyl
- sulfoxide refers to a group -S(O)R f wherein R f is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, heterocyclyl, carbocyclyl, and aralkyl.
- R f is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, heterocyclyl, carbocyclyl, and aralkyl.
- R f is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, heterocyclyl, carbocyclyl, and aralkyl.
- R f is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, heterocyclyl, carbocyclyl, and aralkyl.
- R f include Ci-2oalkyl, phenyl and benzyl.
- sulfonyl refers to a group S(O)2-R f , wherein R f is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, heterocyclyl, carbocyclyl and aralkyl.
- R f is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, heterocyclyl, carbocyclyl and aralkyl.
- R f is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, heterocyclyl, carbocyclyl and aralkyl.
- R f is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, heterocyclyl, carbocyclyl and aralkyl.
- R f include Ci-2oalkyl, phenyl and benzyl.
- sulfonamide refers to a group S(O)NR f R f wherein each R f is independently selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, heterocyclyl, carbocyclyl, and aralkyl.
- R f is independently selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, heterocyclyl, carbocyclyl, and aralkyl.
- preferred R f include Ci-2oalkyl, phenyl and benzyl.
- at least one R f is hydrogen.
- both R f are hydrogen.
- amino is used here in its broadest sense as understood in the art and includes groups of the formula NR a R b wherein R a and R b may be independently selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, carbocyclyl, heteroaryl, heterocyclyl, arylalkyl, and acyl. R a and R b , together with the nitrogen to which they are attached, may also form a monocyclic, or polycyclic ring system e.g. a 3-10 membered ring, particularly, 5-6 and 9-10 membered systems. Examples of “amino” include NH2, NHalkyl (e.g.
- Ci-2oalkyl NHaryl (e.g. NHphenyl), NHaralkyl (e.g. NHbenzyl), NHacyl (e.g. NHC(0)Ci-2oalkyl, NHC(O)phenyl), Nalkylalkyl (wherein each alkyl, for example C1-20, may be the same or different) and 5 or 6 membered rings, optionally containing one or more same or different heteroatoms (e.g. O, N and S).
- NHaryl e.g. NHphenyl
- NHaralkyl e.g. NHbenzyl
- NHacyl e.g. NHC(0)Ci-2oalkyl, NHC(O)phenyl
- Nalkylalkyl wherein each alkyl, for example C1-20, may be the same or different
- 5 or 6 membered rings optionally containing one or more same or different heteroatoms (e.g. O, N and S).
- amido is used here in its broadest sense as understood in the art and includes groups having the formula C(O)NR a R b , wherein R a and R b are as defined as above.
- amido include C(O)NH2, C(O)NHalkyl (e.g. Ci-2oalkyl), C(O)NHaryl (e.g. C(O)NHphenyl), C(O)NHaralkyl (e.g. C(O)NHbenzyl), C(O)NHacyl (e.g.
- each alkyl for example C 1-20, may be the same or different
- 5 or 6 membered rings optionally containing one or more same or different heteroatoms (e.g. O, N and S).
- carboxy ester is used here in its broadest sense as understood in the art and includes groups having the formula CC>2R g , wherein R g may be selected from groups including alkyl, alkenyl, alkynyl, aryl, carbocyclyl, heteroaryl, heterocyclyl, aralkyl, and acyl.
- R g may be selected from groups including alkyl, alkenyl, alkynyl, aryl, carbocyclyl, heteroaryl, heterocyclyl, aralkyl, and acyl.
- Examples of carboxy ester include CChCi ⁇ oalkyl, CCharyl (e.g.. CChphenyl), CCharalkyl (e.g. CO2 benzyl).
- a group may or may not be substituted or fused (so as to form a condensed polycyclic group) with one, two, three or more of organic and inorganic groups, including those selected from: alkyl, alkenyl, alkynyl, carbocyclyl, aryl, heterocyclyl, heteroaryl, acyl, aralkyl, alkaryl, alkheterocyclyl, alkheteroaryl, alkcarbocyclyl, halo, haloalkyl, haloalkenyl, haloalkynyl, haloaryl, halocarbocyclyl, haloheterocyclyl, haloheteroaryl, haloacyl, haloaryalkyl, hydroxy, hydroxyalkyl, hydroxyalkenyl, hydroxyalkynyl, hydroxycarbocyclyl, hydroxyaryl, hydroxyaryl, hydroxy
- Rule 26 nitroheteroayl, nitrocarbocyclyl, nitroacyl, nitroaralkyl, amino (NH2), alkylamino, dialkylamino, alkenylamino, alkynylamino, arylamino, diarylamino, aralkylamino, diaralkylamino, acylamino, diacylamino, heterocyclamino, heteroarylamino, carboxy, carboxyester, amido, alkylsulphonyloxy, arylsulphenyloxy, alkylsulphenyl, arylsulphenyl, thio, alkylthio, alkenylthio, alkynylthio, arylthio, aralkylthio, carbocyclylthio, heterocyclylthio, heteroarylthio, acylthio, sulfoxide, sulfonyl, sulfonamide
- Optional substitution may also be taken to refer to where a -CH2- group in a chain or ring is replaced by a group selected from -O-, -S-, -NR a -, -C(O)- (i.e. carbonyl), -C(O)O- (i.e. ester), and -C(O)NR a - (i.e. amide), where R a is as defined herein.
- Preferred optional substituents include alkyl, (e.g. C1-6 alkyl such as methyl, ethyl, propyl, butyl, cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl), hydroxyalkyl (e.g. hydroxymethyl, hydroxyethyl, hydroxypropyl), alkoxyalkyl (e.g. methoxymethyl, methoxyethyl, methoxypropyl, ethoxymethyl, ethoxyethyl, ethoxypropyl etc) alkoxy (e.g.
- alkyl e.g. C1-6 alkyl such as methyl, ethyl, propyl, butyl, cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl
- hydroxyalkyl e.g. hydroxymethyl, hydroxyethyl, hydroxypropyl
- C1-6 alkoxy such as methoxy, ethoxy, propoxy, butoxy, cyclopropoxy, cyclobutoxy
- halo trifluoromethyl, trichloromethyl, tribromomethyl, hydroxy, phenyl (which itself may be further substituted e.g., by C1-6 alkyl, halo, hydroxy, hydroxyCi-6 alkyl, C1-6 alkoxy, haloCi-ealkyl, cyano, nitro
- Ci-6 alkyl such as methylamino, ethylamino, propylamino etc), dialkylamino (e.g. Ci-6 alkyl, such as dimethylamino, diethylamino, dipropylamino), acylamino (e.g. NHC(O)CH3), phenylamino (wherein phenyl itself may be further substituted e.g., by Ci-6 alkyl, halo, hydroxy, hydroxyCi-6 alkyl, Ci-6 alkoxy, haloCi-6 alkyl, cyano, nitro OC(O)Ci-6 alkyl, and amino), nitro, formyl, -C(O)-alkyl (e.g.
- Ci-6 alkyl such as acetyl
- O-C(O)-alkyl e.g. Ci-6alkyl, such as acetyloxy
- benzoyl wherein the phenyl group itself may be further substituted e.g., by Ci-6 alkyl, halo, hydroxy hydroxyCi-6 alkyl, Ci-6 alkoxy, haloCi-6 alkyl, cyano, nitro OC(O)Ci-6alkyl, and amino
- C1-6 alkyl such as methyl ester, ethyl ester, propyl ester, butyl ester
- CC phenyl wherein phenyl itself may be further substituted e.g., by C1-6 alkyl, halo, hydroxy, hydroxyl C1-6 alkyl, Ci- 6 alkoxy, halo C1-6 alkyl, cyano, nitro OC(O)Ci-6 alkyl, and amino
- CONH2 CONHphenyl (wherein phenyl itself may be further substituted e.g., by C1-6 alkyl, halo, hydroxy, hydroxyl C1-6 alkyl, Ci -6 alkoxy, halo C1-6 alkyl, cyano, nitro OC(O)Ci-6 alkyl, and amino)
- CONHbenzyl wherein benzyl itself may be further substituted e.g., by C1-6 alkyl, halo, hydroxy hydroxyl C1-6 alkyl, Ci
- C1-6 alkyl such as methyl ester, ethyl ester, propyl ester, butyl amide) CONHdialkyl (e.g. C1-6 alkyl) aminoalkyl (e.g., HN C1-6 alkyl-, Ci-6alkylHN-Ci-6 alkyl- and (Ci-6alkyl)2N-Ci-6 alkyl-), thioalkyl (e.g., HS C1-6 alkyl-), carboxyalkyl (e.g., HO2CC1-6 alkyl-), carboxyesteralkyl (e.g., Ci- 6 alkylChCCi-6 alkyl-), amidoalkyl (e.g., H2N(O)CC 1-6 alkyl-, H(Ci-6 alkyl)N(O)CCi-6 alkyl-), formylalkyl (e.g., OHCCusalkyl-), acylalkyl (e.g., Ci-6 alky
- heteroatom refers to any atom other than a carbon atom which may be a member of a cyclic organic group.
- heteroatoms include nitrogen, oxygen, sulfur, phosphorous, boron, silicon, selenium and tellurium, more particularly nitrogen, oxygen and sulfur.
- thio [group] refers to a particular group when linked by sulfur, for example, the terms “thioalkyl”, “thioalkenyl”, thioalkynyl” and “thioaryl”, respectively, denote alkyl, alkenyl, alkynyl and aryl groups as hereinbefore defined when linked by sulfur.
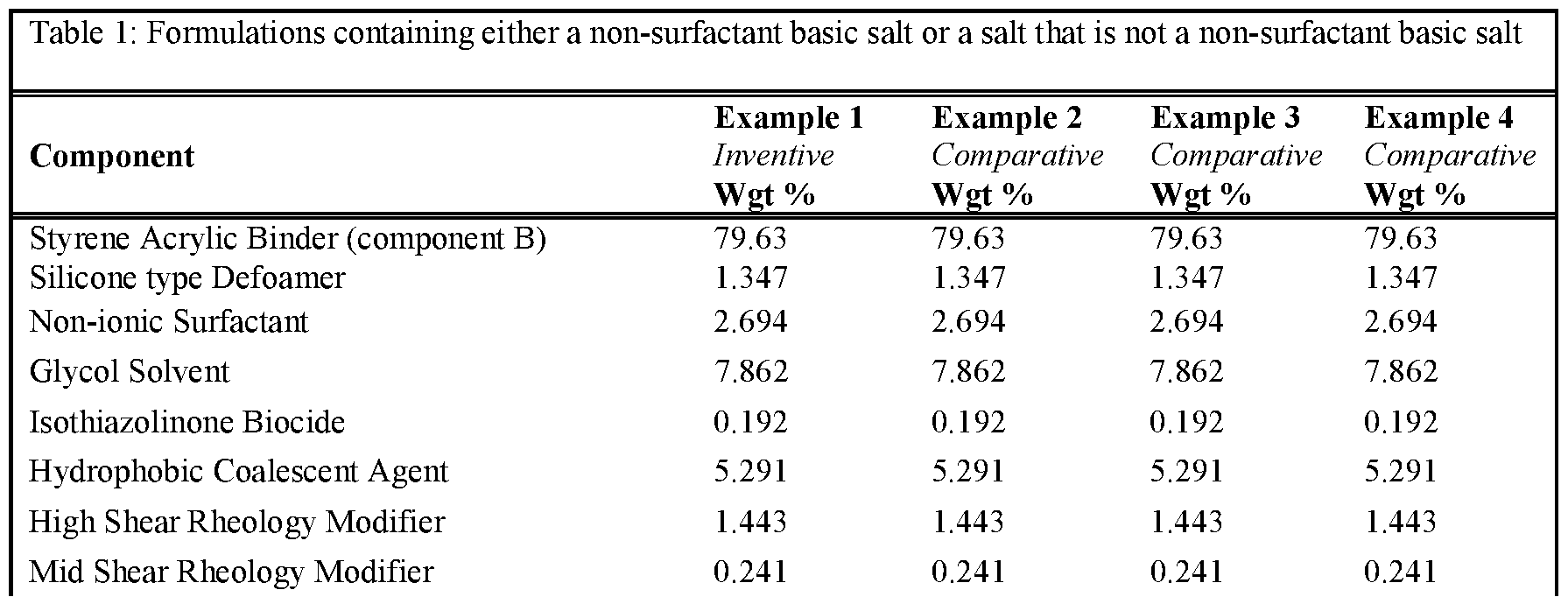
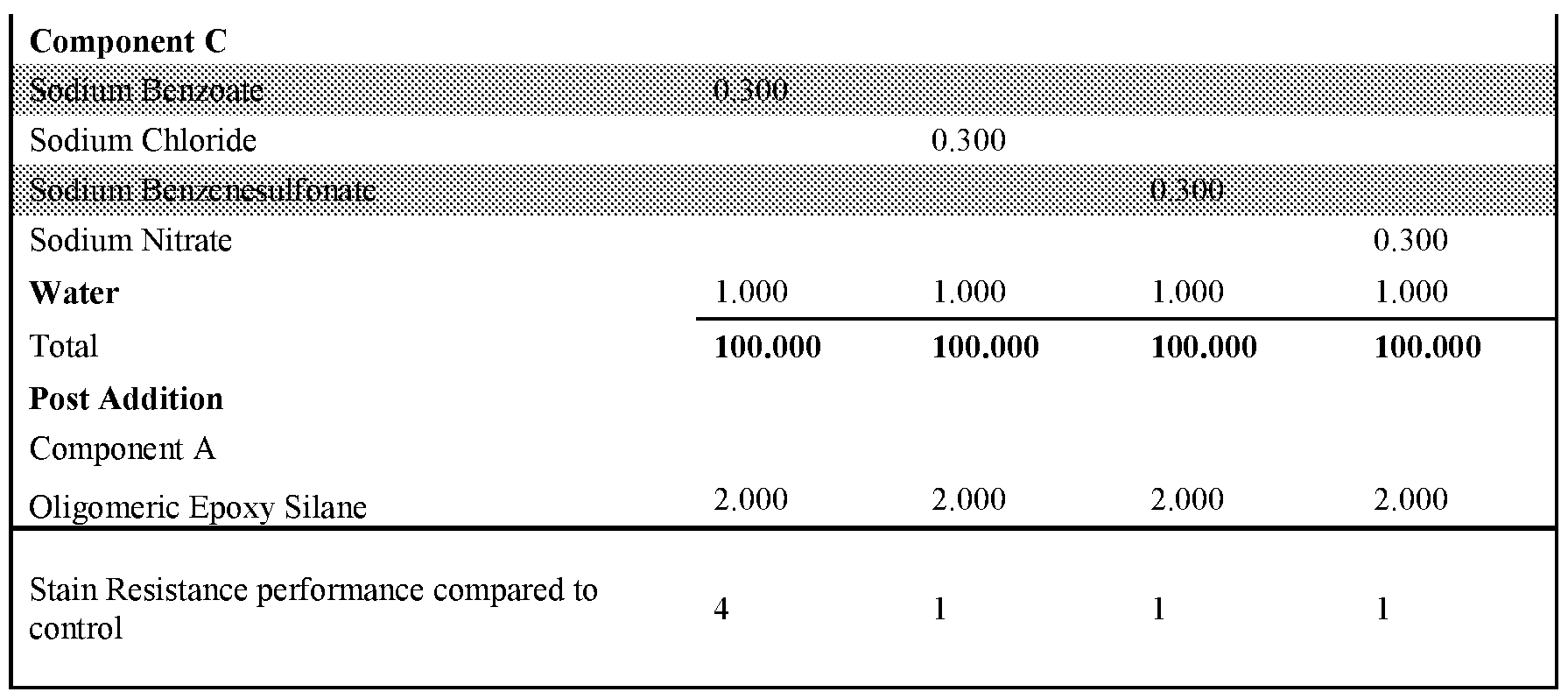
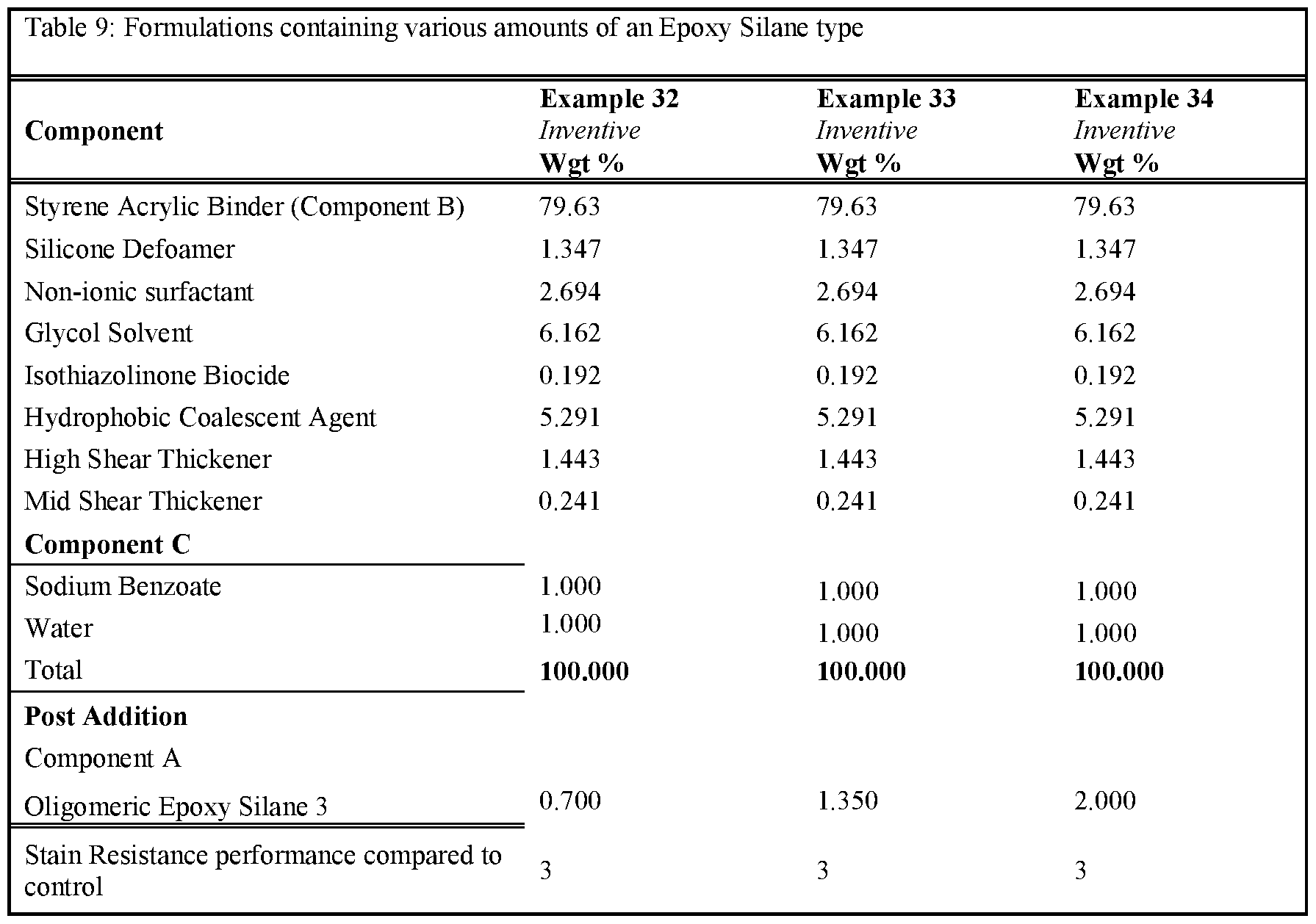
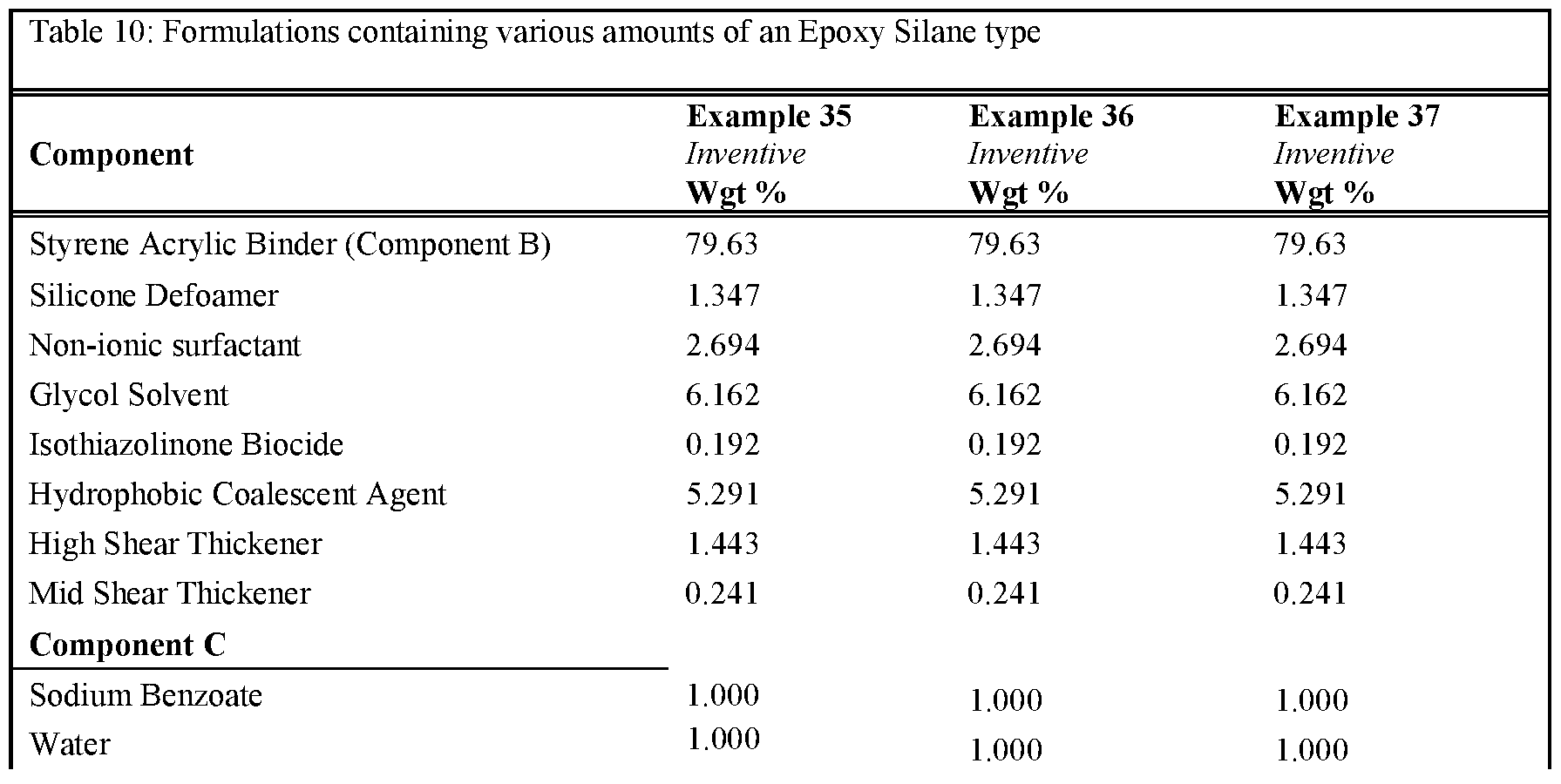
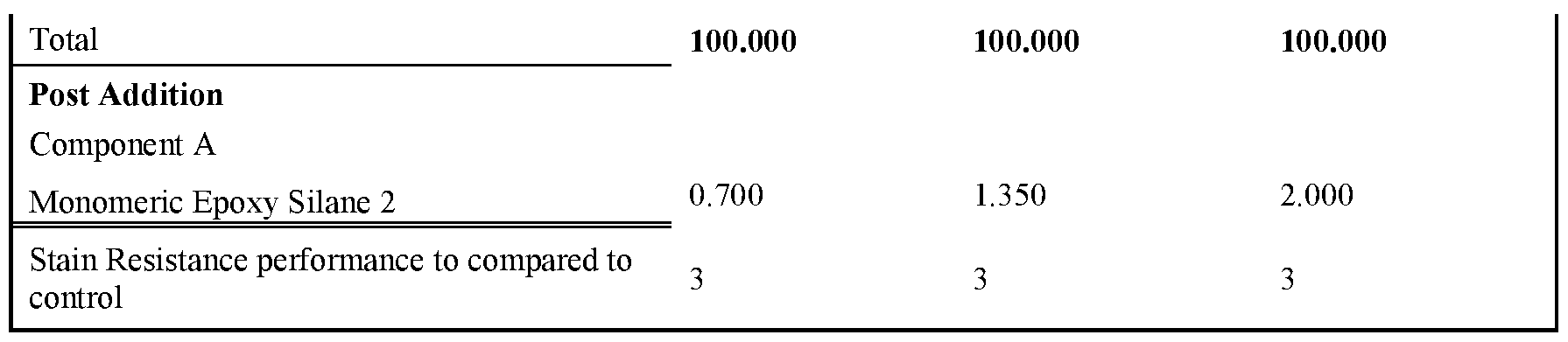
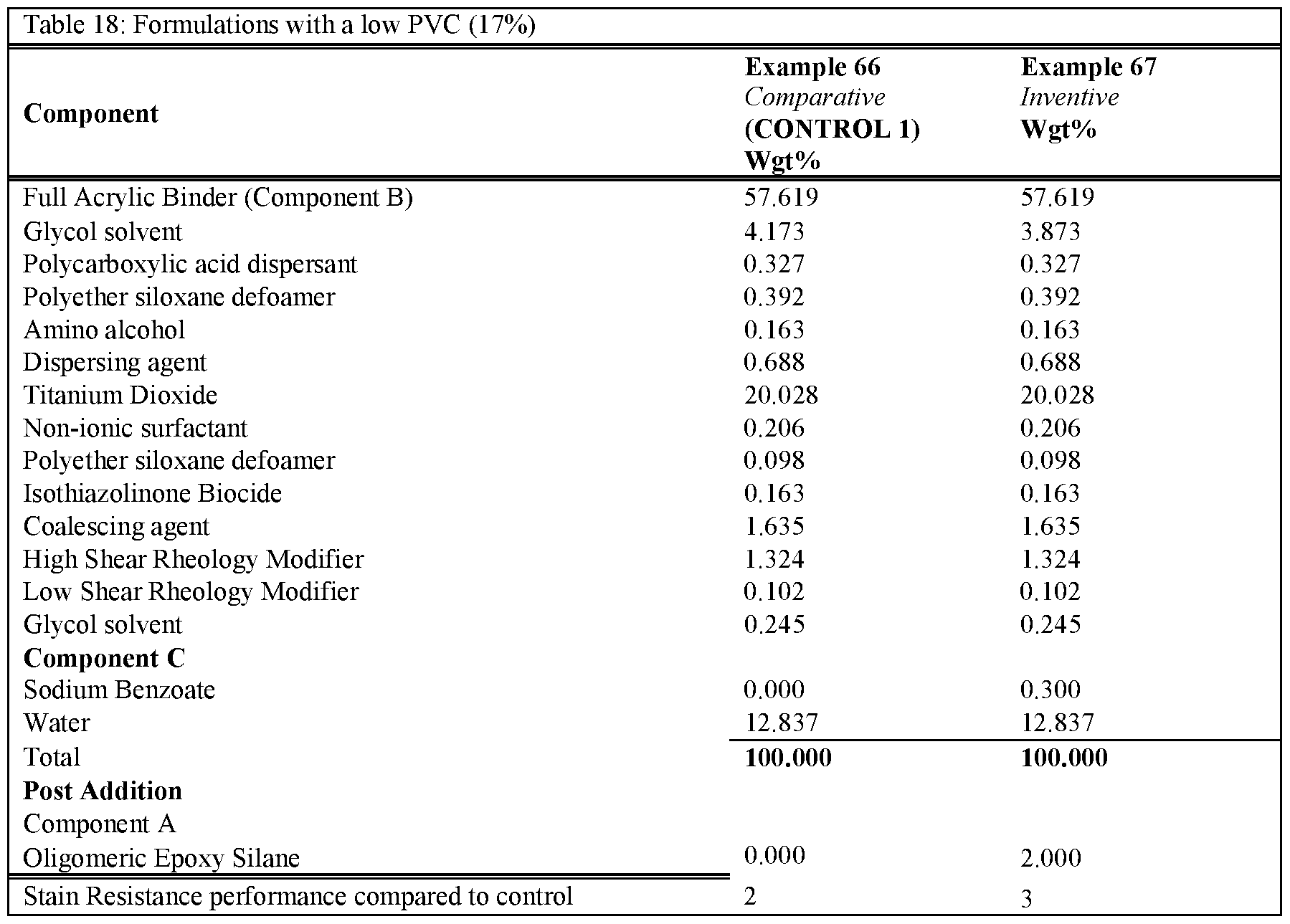
- the formulations were prepared according to the described procedure.
- an epon lined can of 250mL the Binder (component B - the functional aqueous polymer dispersion), Silicone type Defoamer, Non-ionic Surfactant, Glycol Solvent, Isothiazolinone Biocide, Hydrophobic Coalescent Agent, High, Shear Rheology Modifier and Mid Shear Rheology Modifier were combined under stirring by a high-quality paint stirrer. The final composition was left under stirring for approximately 30 minutes. Next, component C was combined with an aliquot of water until fully dissolved. Once the water became transparent, the mixture was added to the composition under stirring for another 5 minutes to fully incorporate. Finally, component A was added to the composition and stirred for l-2min by hand prior to application.
- Stain resistance ability was tested according to Australian Standard AS 1580. 108.8. Test samples were cast on a ceramic tile by rolling or using a drawdown bar. The films on the test panels were cured for 7 days at room temperature before stains were applied. The test panels were placed horizontally on a flat surface and within the test areas, staining materials (blue food dye, pink food dye, lipstick, 10% coffee solution, red wine, white vinegar,) were applied on the
- the examples presented below are rated according to a simple system to avoid any subjectiveness when testing their performance.
- the stain resistance performance of the samples is compared to a control sample which contains an oligomeric epoxy silane but does not contain the non-surfactant basic salt.
- each sample is compared to the control and given a rating ranging from 1 to 4. Where 1 indicates the washability performance is worse than the control.
- a rating of 2 indicates the performance is comparable to the control. When a rating of 3 is given, it indicates better performance than the control, finally a rating 4 indicates the formulation has an excellent performance with much better performance than the control.
- the control is a formulation that does not contain any non-surfactant basic salt but does contain component A (such as example 48).
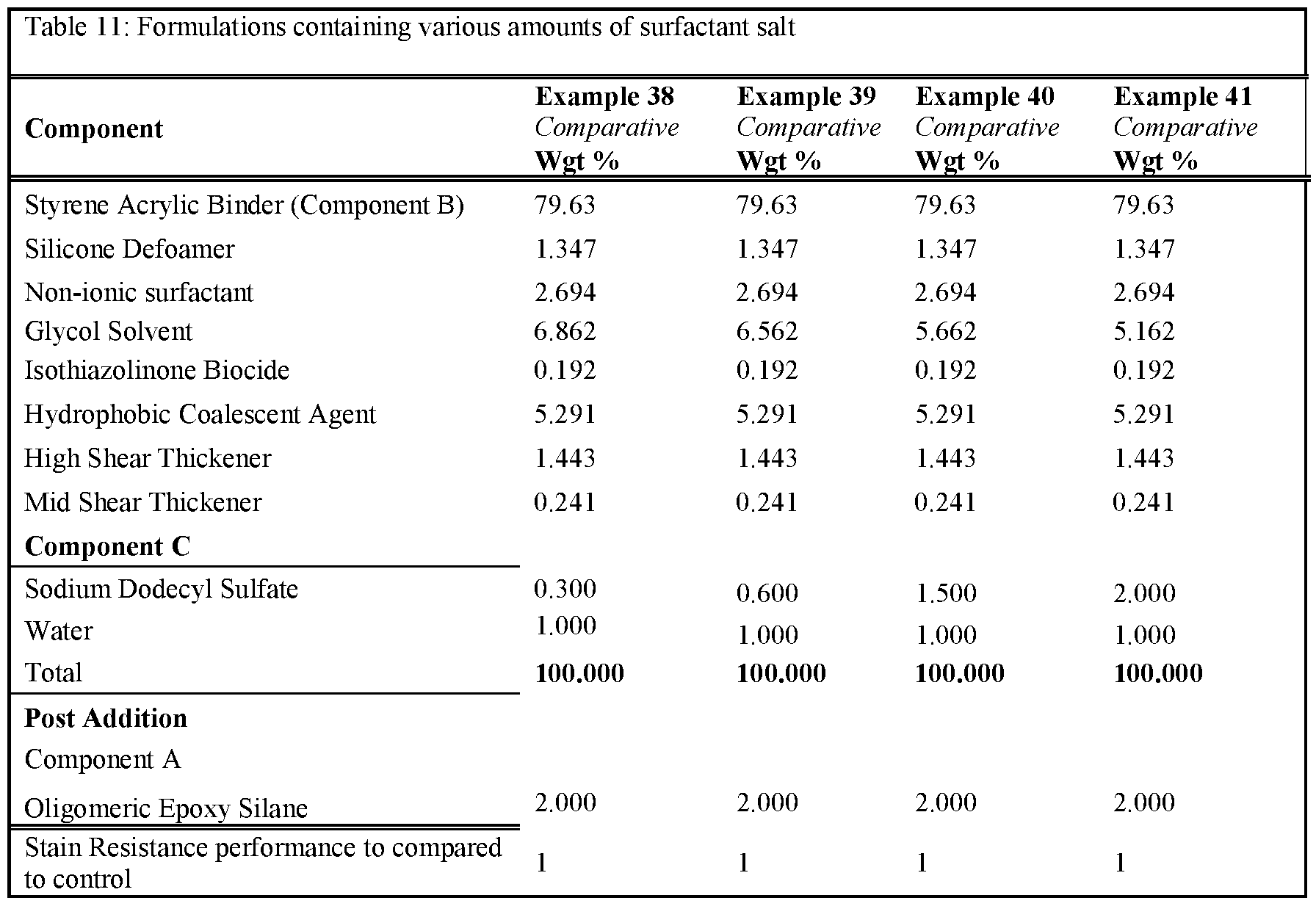
- Tables 1 and 2 present the results for formulations created either with a non-surfactant basic salt or a salt that is not a non-surfactant-basic salt.
- the formulations were prepared as per the examples 1-9.
- Non-surfactant basic salts included in these examples are sodium benzoate, sodium carbonate, potassium benzoate, sodium phosphate dibasic and sodium acetate. All other salts used in those examples can be considered not to be non-surfactant basic salts.
- the use of 0.3 wt% of a non-surfactant basic salt delivers an improvement in stain resistance relative to the control, whereas the same level of a salt which is not a non-surfactant basic salt leads to no change or deterioration in stain resistance.
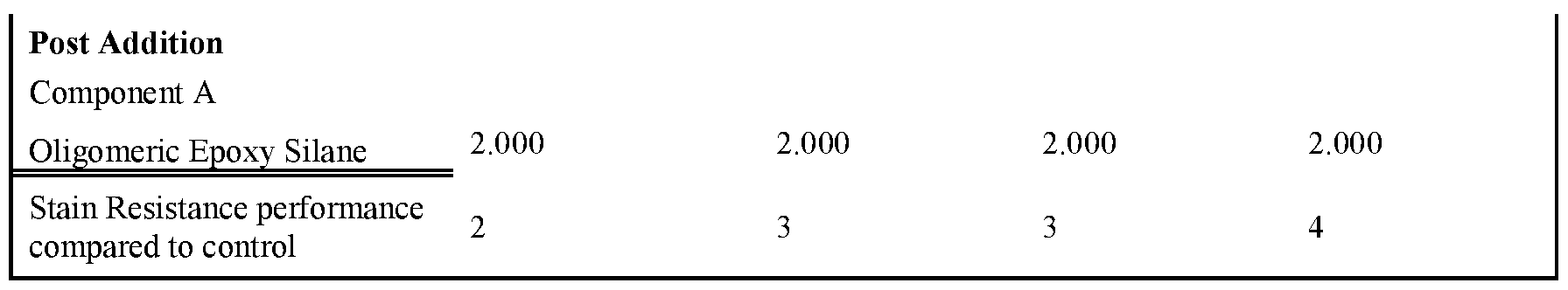
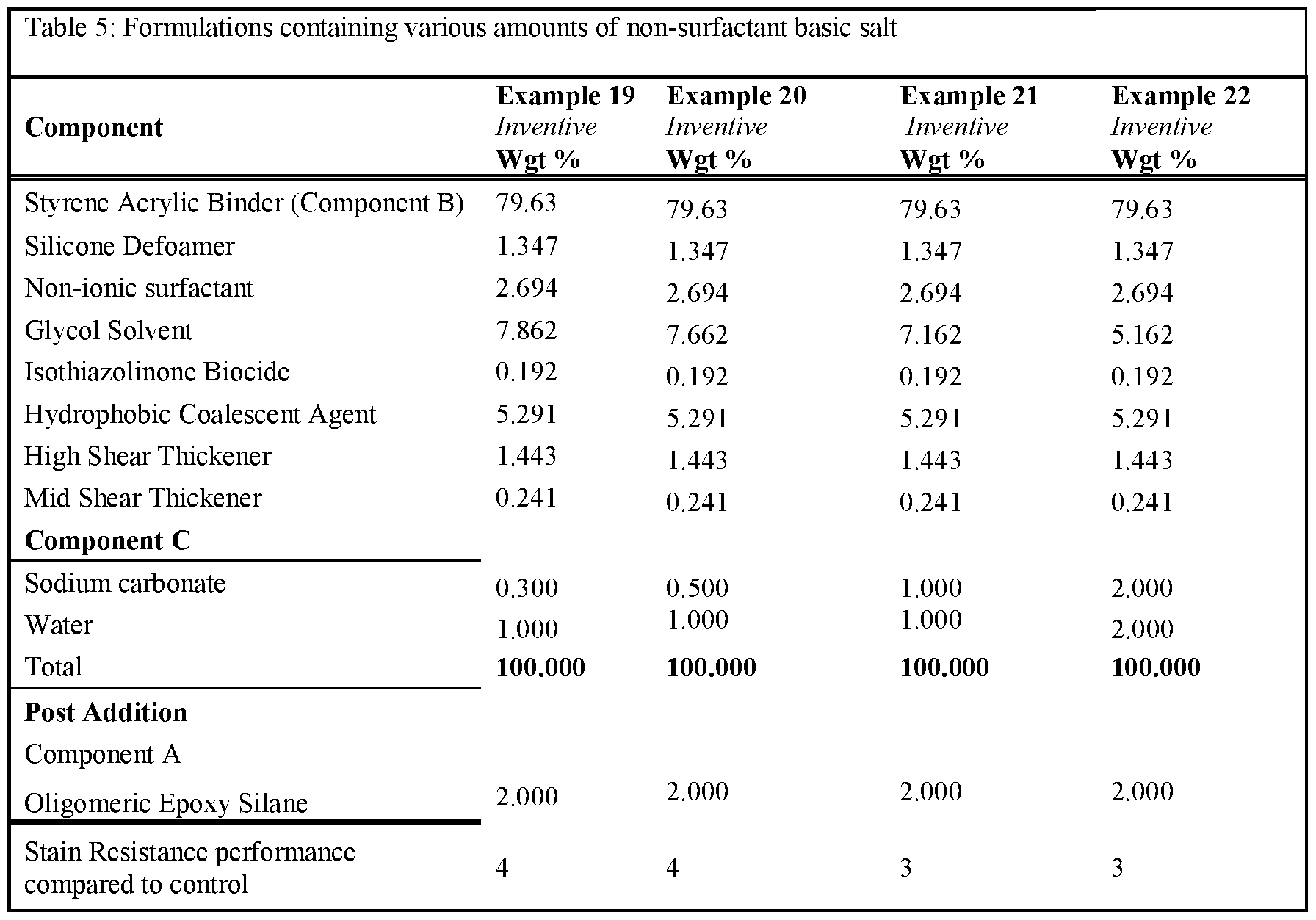
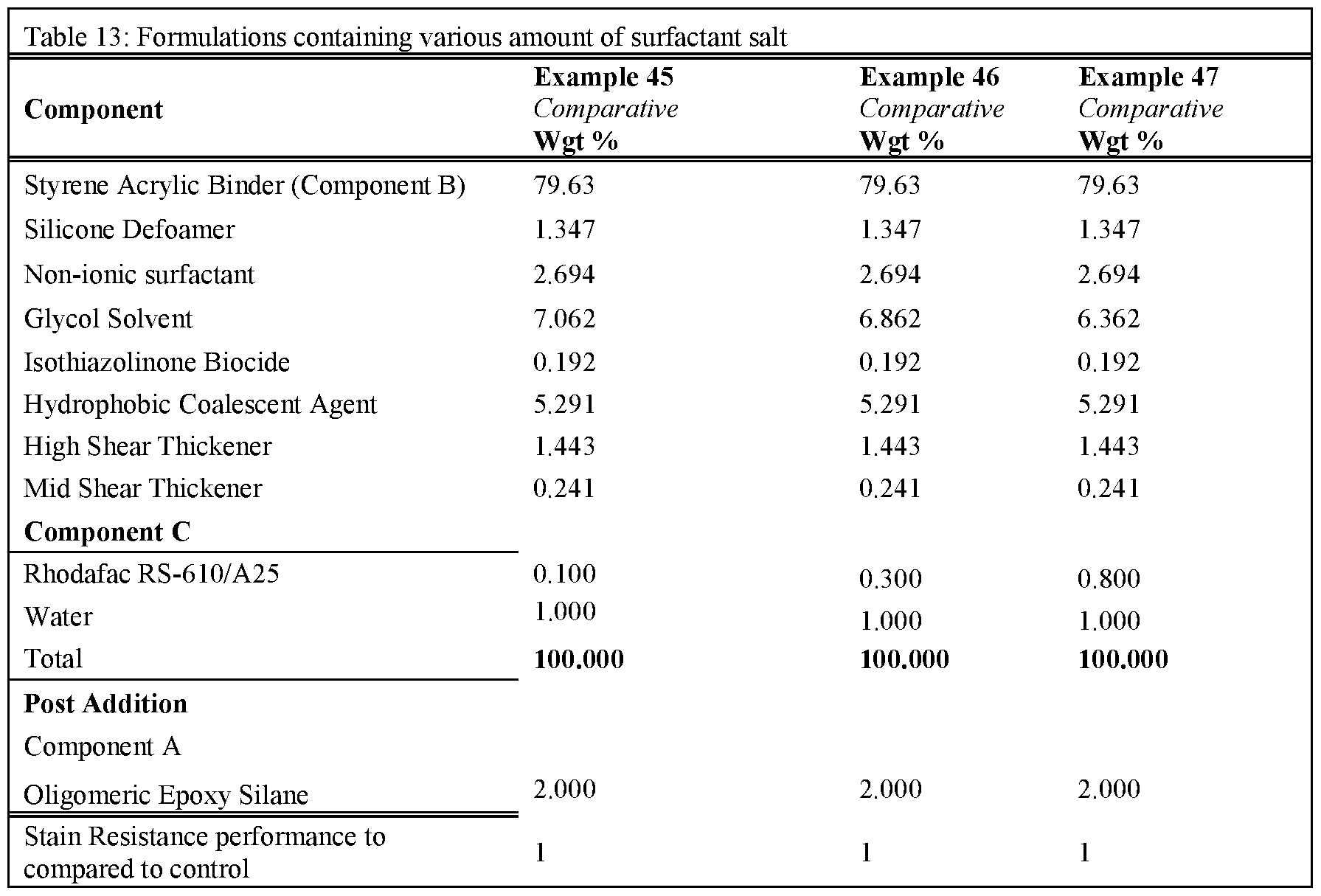
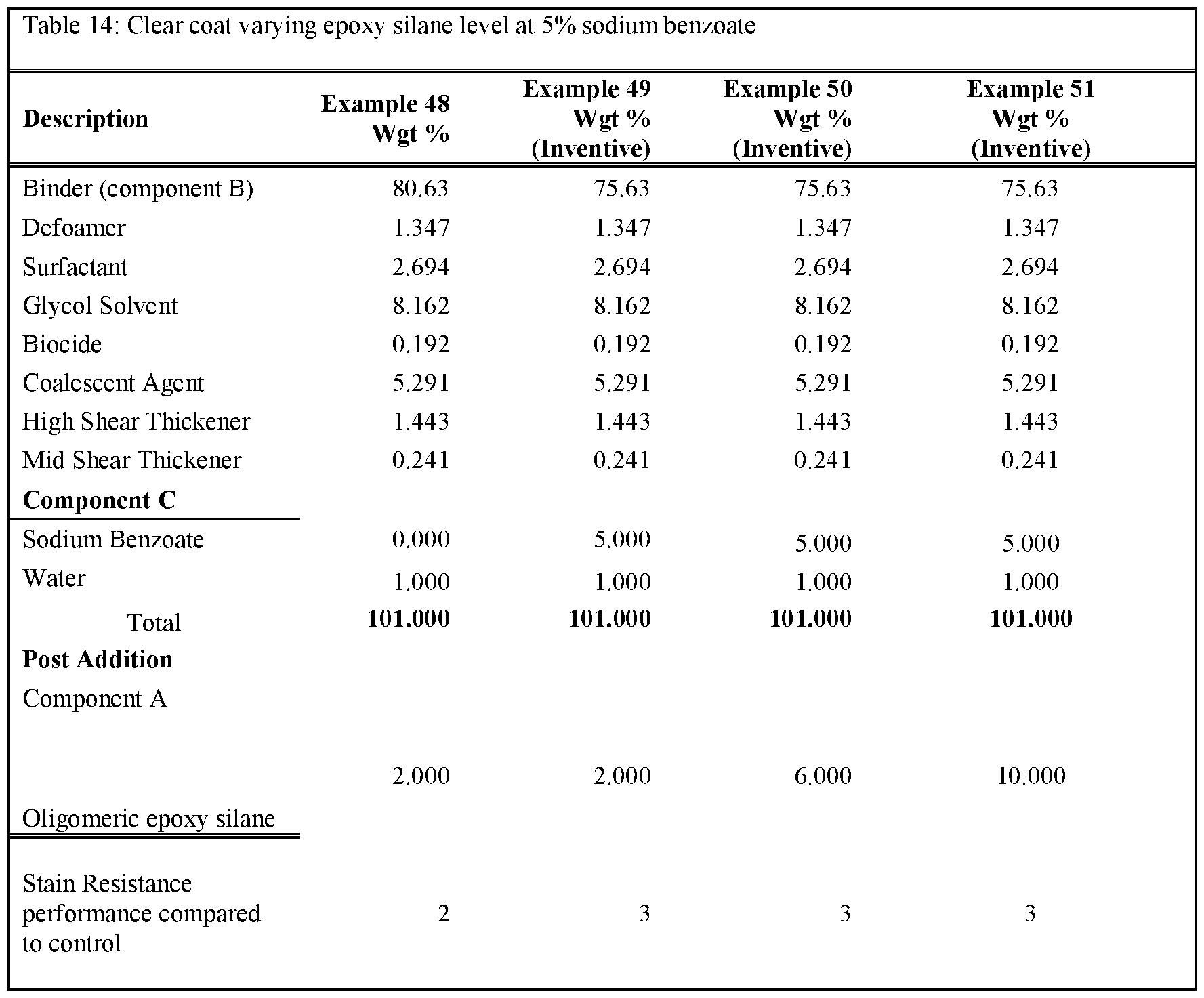
- Tables 3-5 present the results of formulations in which the amount of non-surfactant basic salt used in the formula was varied from 0.1 wgt% up to 3wgt%.
- the formulations were prepared as per examples 10-22. Note that for all these formulations the amount of component A was kept constant at 2 wgt%. As can be seen, the inclusion of >0.25% of a non-surfactant basic salt
- Rule 26 delivers an improvement in stain resistance relative to the control, whereas a level of ⁇ 0.25% gave no change in stain resistance.
- Tables 6-10 provides formulations in which the type and quantity of component A was varied. The formulations tested were made as per examples 23-37. Note that for these examples the amount and type of non-surfactant basic salt was kept consistent throughout all examples. As can be seen, the inclusion of an epoxy silane, along with >0.25% a non-surfactant basic salt, delivers an improvement in stain resistance relative to the control.
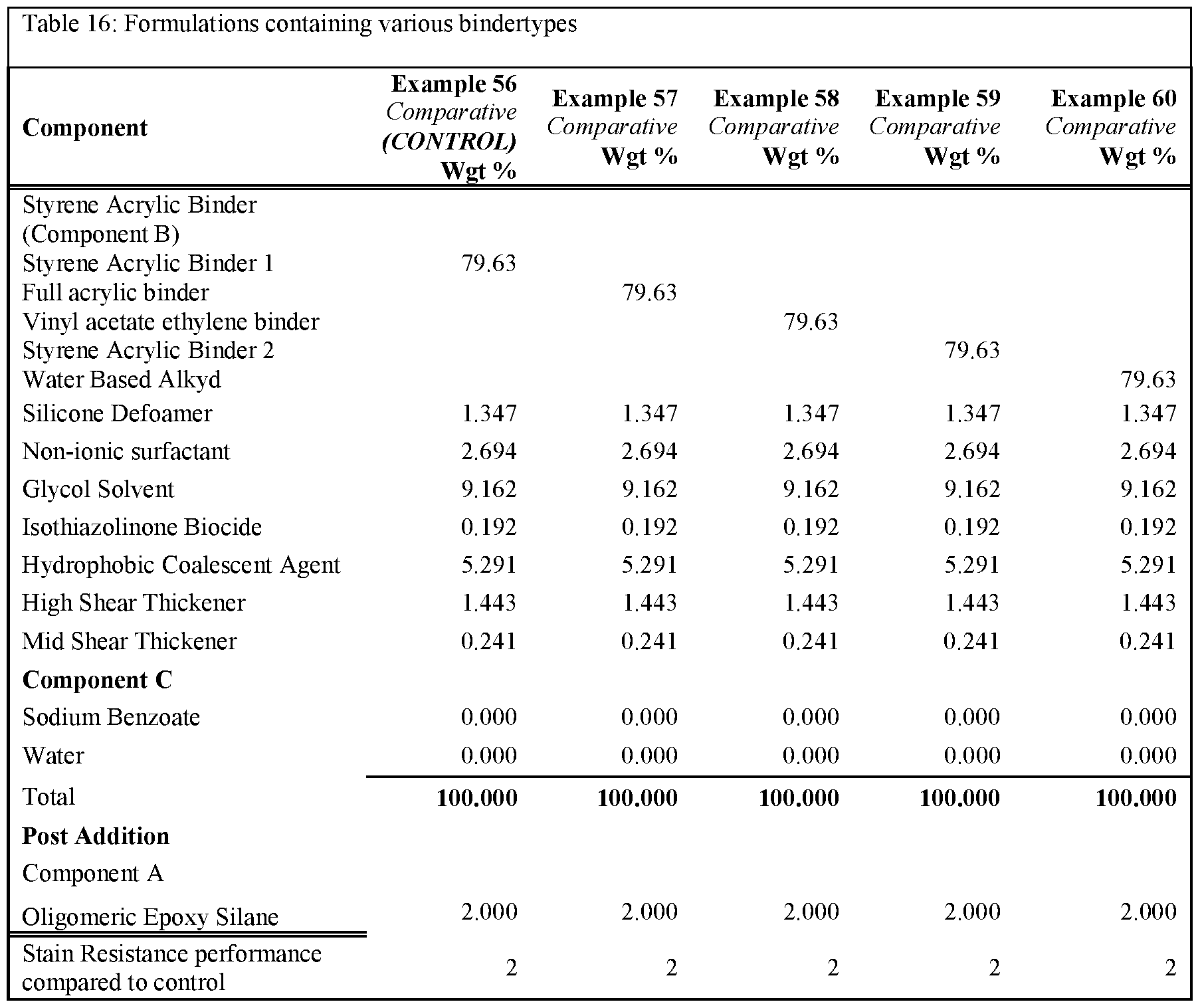
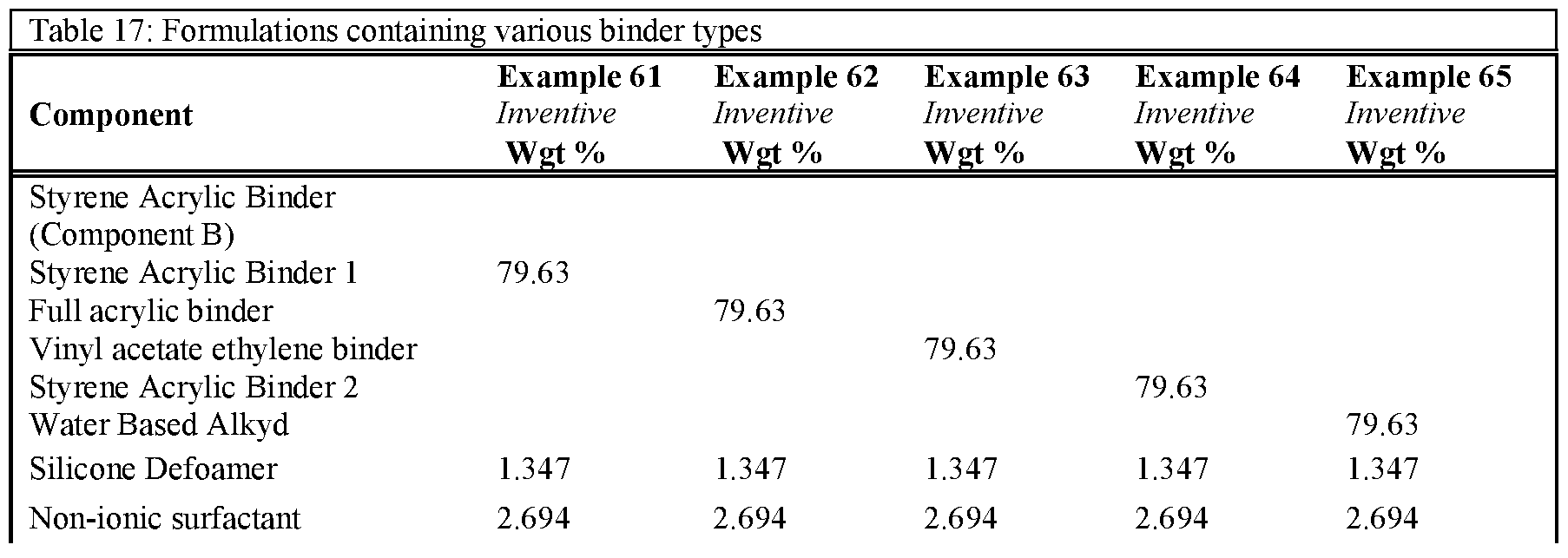
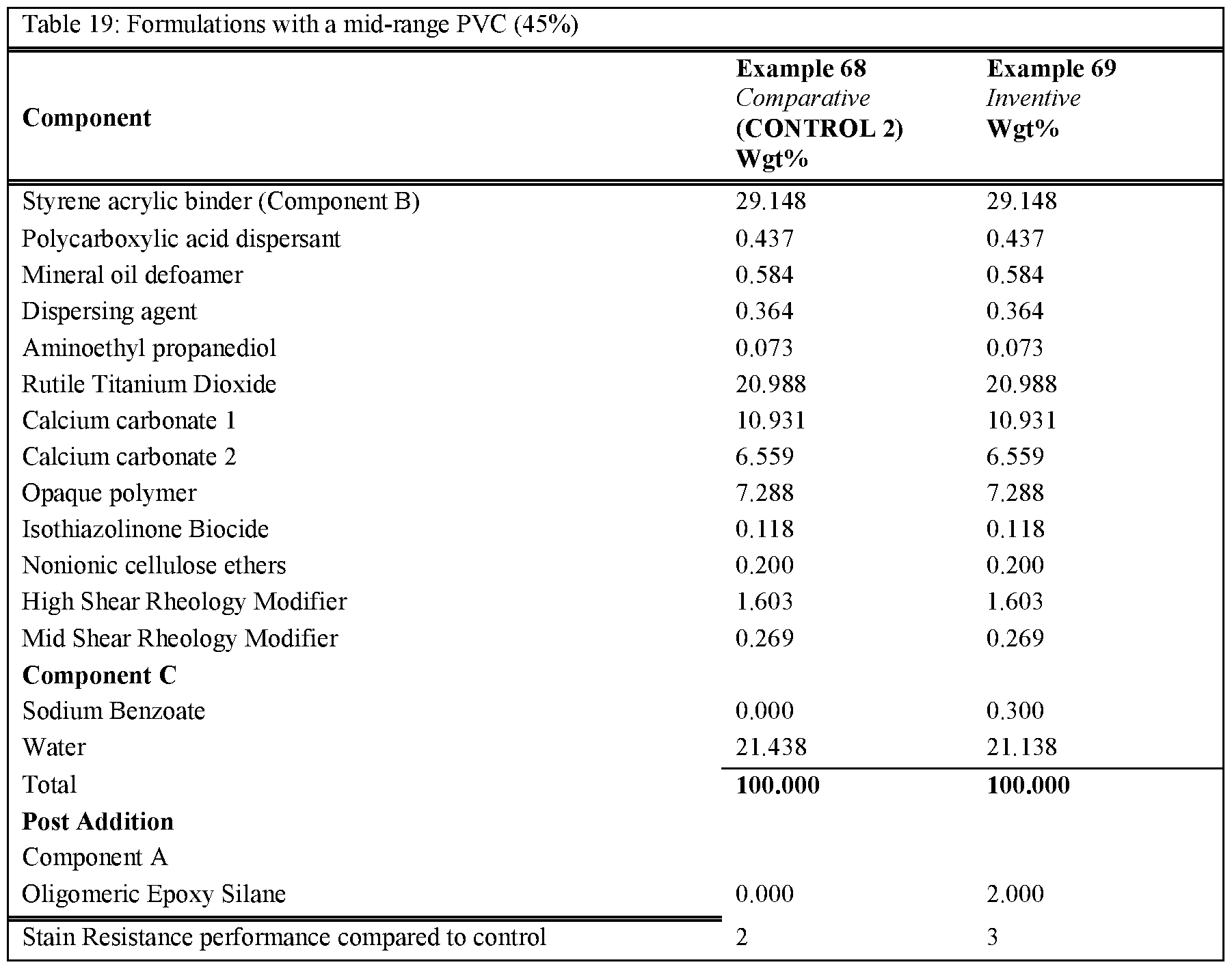
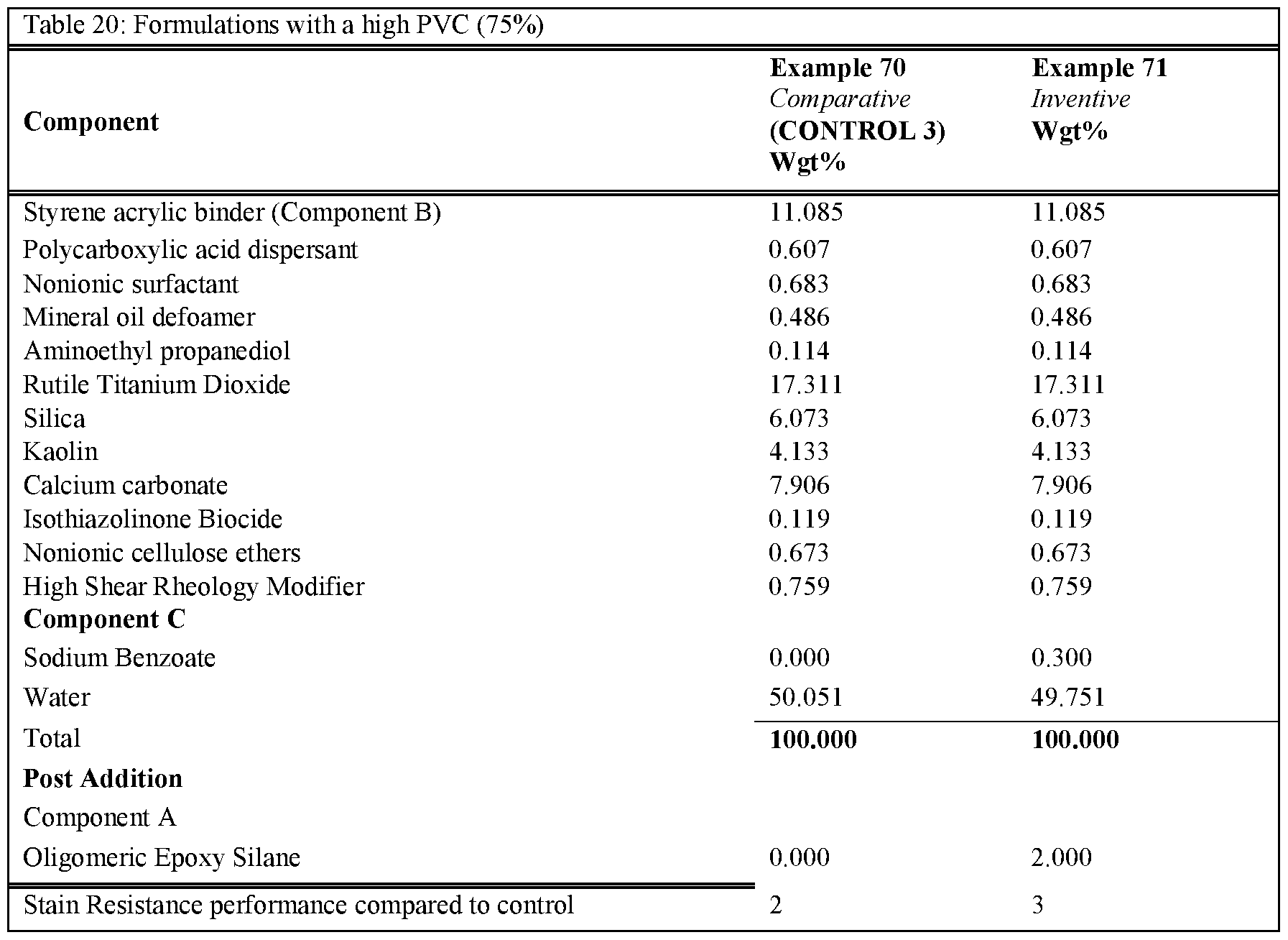
- Table 16 and Table 17 show formulations utilising different binder systems but with and without Component C added. These formulations demonstrate that the improved stain resistance is only present when component C is added above 0.25% in a range of binder systems.
- Table 16 Table 17 and Table 18 demonstrate formulations at various PVC values, with Component A and Component C added. These formulations show that the improved stain resistance is present when both Component A and C are added.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Wood Science & Technology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Polymers & Plastics (AREA)
- Dispersion Chemistry (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Paints Or Removers (AREA)
Abstract
Description
Claims
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2023360028A AU2023360028A1 (en) | 2022-10-12 | 2023-09-14 | Aqueous coating composition |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2022902993A AU2022902993A0 (en) | 2022-10-12 | Aqueous coating composition | |
| AU2022902993 | 2022-10-12 | ||
| AU2023901442 | 2023-05-12 | ||
| AU2023901442A AU2023901442A0 (en) | 2023-05-12 | Aqueous coating composition |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024077328A1 true WO2024077328A1 (en) | 2024-04-18 |
Family
ID=90668351
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/AU2023/050891 Ceased WO2024077328A1 (en) | 2022-10-12 | 2023-09-14 | Aqueous coating composition |
Country Status (2)
| Country | Link |
|---|---|
| AU (1) | AU2023360028A1 (en) |
| WO (1) | WO2024077328A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025230884A1 (en) * | 2024-04-29 | 2025-11-06 | Covestro Llc | Polymer dispersions for scrub and stain resistant wall paint |
| EP4660258A1 (en) * | 2024-06-05 | 2025-12-10 | Covestro LLC | Polymer dispersions for scrub and stain resistant wall paint |
Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5714532A (en) * | 1995-04-12 | 1998-02-03 | Osi Specialties, Inc. | Composition of epoxysilane emulsion additives in water based reactive polymer dispersions and methods of preparation |
| US6627682B1 (en) * | 1995-04-12 | 2003-09-30 | Crompton Corporation | Epoxy compositions |
| US20070179268A1 (en) * | 2006-01-27 | 2007-08-02 | Alain Lejeune | Low VOC epoxy silane oligomer and compositions containing same |
| US20110151265A1 (en) * | 2008-08-15 | 2011-06-23 | Valspar Sourcing Inc. | Self-etching cementitious substrate coating composition |
| US8338531B2 (en) * | 2005-04-07 | 2012-12-25 | Momentive Perofrmance Materials Inc. | Epoxy silane oligomer and coating composition containing same |
| US20170335116A1 (en) * | 2014-12-22 | 2017-11-23 | Dow Global Technologies Llc | Aqueous emulsion paint with improved stain removal and anticlogging properties |
| US20190144688A1 (en) * | 2016-05-10 | 2019-05-16 | Hoden Seimitsu Kako Kenkyusyo Co., Ltd. | Aqueous anti-rust surface treatment composition and surface-coated metal member |
| US20200181420A1 (en) * | 2017-07-25 | 2020-06-11 | Jotun A/S | Aqueous primer |
| US20210324114A1 (en) * | 2018-10-24 | 2021-10-21 | Dow Global Technologies Llc | Aqueous dispersion and aqueous coating composition |
| WO2022048487A1 (en) * | 2020-09-01 | 2022-03-10 | Guangdong Huarun Paints Co., Ltd. | A coating composition, an article made thereof and a process for preparing the same |
| US20220243049A1 (en) * | 2021-01-29 | 2022-08-04 | Nan Ya Plastics Corporation | Polymeric composition, aqueous acrylic resin having high solvent resistance, and method for manufacturing the same |
-
2023
- 2023-09-14 AU AU2023360028A patent/AU2023360028A1/en active Pending
- 2023-09-14 WO PCT/AU2023/050891 patent/WO2024077328A1/en not_active Ceased
Patent Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5714532A (en) * | 1995-04-12 | 1998-02-03 | Osi Specialties, Inc. | Composition of epoxysilane emulsion additives in water based reactive polymer dispersions and methods of preparation |
| US6627682B1 (en) * | 1995-04-12 | 2003-09-30 | Crompton Corporation | Epoxy compositions |
| US8338531B2 (en) * | 2005-04-07 | 2012-12-25 | Momentive Perofrmance Materials Inc. | Epoxy silane oligomer and coating composition containing same |
| US20070179268A1 (en) * | 2006-01-27 | 2007-08-02 | Alain Lejeune | Low VOC epoxy silane oligomer and compositions containing same |
| US20110151265A1 (en) * | 2008-08-15 | 2011-06-23 | Valspar Sourcing Inc. | Self-etching cementitious substrate coating composition |
| US20170335116A1 (en) * | 2014-12-22 | 2017-11-23 | Dow Global Technologies Llc | Aqueous emulsion paint with improved stain removal and anticlogging properties |
| US20190144688A1 (en) * | 2016-05-10 | 2019-05-16 | Hoden Seimitsu Kako Kenkyusyo Co., Ltd. | Aqueous anti-rust surface treatment composition and surface-coated metal member |
| US20200181420A1 (en) * | 2017-07-25 | 2020-06-11 | Jotun A/S | Aqueous primer |
| US20210324114A1 (en) * | 2018-10-24 | 2021-10-21 | Dow Global Technologies Llc | Aqueous dispersion and aqueous coating composition |
| WO2022048487A1 (en) * | 2020-09-01 | 2022-03-10 | Guangdong Huarun Paints Co., Ltd. | A coating composition, an article made thereof and a process for preparing the same |
| US20220243049A1 (en) * | 2021-01-29 | 2022-08-04 | Nan Ya Plastics Corporation | Polymeric composition, aqueous acrylic resin having high solvent resistance, and method for manufacturing the same |
Non-Patent Citations (1)
| Title |
|---|
| CECILIA DEYA: "Silane as adhesion promoter in damages areas", PROGRESS IN ORGANIC COATINGS, vol. 90, 2016, pages 28 - 33, XP029342012, DOI: 10.1016/j.porgcoat.2015.09.001 * |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025230884A1 (en) * | 2024-04-29 | 2025-11-06 | Covestro Llc | Polymer dispersions for scrub and stain resistant wall paint |
| EP4660258A1 (en) * | 2024-06-05 | 2025-12-10 | Covestro LLC | Polymer dispersions for scrub and stain resistant wall paint |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2023360028A1 (en) | 2025-04-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2007312956B2 (en) | Vesiculated polymer particles | |
| AU2017383105B2 (en) | Aqueous polymer composition | |
| US9255198B2 (en) | Water swellable polymer materials comprising particulate core and water swellable R.A.F.T polymer shell | |
| US20120128743A1 (en) | Polymer particles | |
| AU2011305043B2 (en) | Continuous flow polymerisation process | |
| CA2837897C (en) | Encapsulated polymer particles | |
| AU2023360028A1 (en) | Aqueous coating composition | |
| US20220177659A1 (en) | Non-core shell polymer particles | |
| US20170298026A1 (en) | All purpose raft agent | |
| AU2016275557A1 (en) | Versatile RAFT agent | |
| WO2010091465A1 (en) | Polymer particles | |
| AU2024281782A1 (en) | Opacified coating composition | |
| AU2016314031A1 (en) | Amphiphilic raft agent | |
| NZ618219B2 (en) | Polymer encapsulated non-polymeric particulate material | |
| WO2009009554A1 (en) | Azo colourant |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 23875944 Country of ref document: EP Kind code of ref document: A1 |
|
| DPE1 | Request for preliminary examination filed after expiration of 19th month from priority date (pct application filed from 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 819762 Country of ref document: NZ Ref document number: AU2023360028 Country of ref document: AU |
|
| WWP | Wipo information: published in national office |
Ref document number: 819762 Country of ref document: NZ |
|
| ENP | Entry into the national phase |
Ref document number: 2023360028 Country of ref document: AU Date of ref document: 20230914 Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 23875944 Country of ref document: EP Kind code of ref document: A1 |