WO2024038088A1 - Liquid formulations - Google Patents
Liquid formulations Download PDFInfo
- Publication number
- WO2024038088A1 WO2024038088A1 PCT/EP2023/072562 EP2023072562W WO2024038088A1 WO 2024038088 A1 WO2024038088 A1 WO 2024038088A1 EP 2023072562 W EP2023072562 W EP 2023072562W WO 2024038088 A1 WO2024038088 A1 WO 2024038088A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- conjugate
- sugar
- hyaluronan
- aqueous liquid
- liquid composition
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/56—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule
- A61K47/61—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule the organic macromolecular compound being a polysaccharide or a derivative thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K41/00—Medicinal preparations obtained by treating materials with wave energy or particle radiation ; Therapies using these preparations
- A61K41/10—Inactivation or decontamination of a medicinal preparation prior to administration to an animal or a person
- A61K41/17—Inactivation or decontamination of a medicinal preparation prior to administration to an animal or a person by ultraviolet [UV] or infrared [IR] light, X-rays or gamma rays
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2/00—Methods or apparatus for disinfecting or sterilising materials or objects other than foodstuffs or contact lenses; Accessories therefor
- A61L2/02—Methods or apparatus for disinfecting or sterilising materials or objects other than foodstuffs or contact lenses; Accessories therefor using physical phenomena
- A61L2/08—Radiation
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
- A61P27/12—Ophthalmic agents for cataracts
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P41/00—Drugs used in surgical methods, e.g. surgery adjuvants for preventing adhesion or for vitreum substitution
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A61L2103/05—
Definitions
- the present invention relates to aqueous liquid compositions comprising a conjugate of hyaluronan and a pharmaceutically active compound.
- the formulations are suitable for use in various methods of treatment in human or veterinary medicine, and for preparation of a medicament for use in human or veterinary medicine.
- the invention also relates to kits and methods for manufacturing the aqueous liquid compositions.
- Hyaluronan is an anionic, nonsulfated glycosaminoglycan distributed throughout connective, epithelial, and neural tissues in humans and other vertebrates.
- Hyaluronic acid is a polysaccharide built of disaccharide repeating residues of ⁇ -D-glucuronic acid and N-acetyl- ⁇ -D-glucosamine, where the linkage is (1 ⁇ 3) from the glucuronic acid to the glucosamine, and (1 ⁇ 4) from the glucosamine to the glucuronic acid.
- Hyaluronan refers to all physiological forms of hyaluronic acid, the most common being the sodium salt (sodium hyaluronate; NaHA).
- NaHA sodium hyaluronate
- hyaluronic acid is commonly used in the literature for referring to any of its forms: It is a very large molecule and can have a molecular weight of several or more million Daltons.
- Hyaluronan is present in most tissues in mammals in the extracellular matrix. In mammals, hyaluronan is found in higher amounts in the umbilical cord, and it is a constituent of the vitreous body and joint cartilage. Hyaluronan is an important constituent of the synovial fluid. It has high viscosity and provides lubrication to the joints. Hyaluronan and modified derivatives of hyaluronan are currently used in in vivo applications such as eye surgery, cosmetic injections and intraarticular injections to treat osteoarthritis. Osteoarthritis is a degenerative joint disease and a very common condition. Knee joints, hip joints and shoulder joints are often affected, and symptoms can be disabling to different degrees.
- NSAIDs non-steroidal anti-inflammatory drugs
- Some of these NSAIDs are known to give gastrointestinal problems after extended use, and such intestinal complications are far from uncommon.
- osteoarthritis local administration of the drug, e.g. by injection, would be desirable, but the duration of a small NSAID molecule is relatively short, and a more prolonged duration is necessary for efficient treatment.
- Many studies have been performed to investigate the efficiency of hyaluronan in arthritis treatment, see Lohmander et al. (Annals of the Rheumatic Diseases, 1996, 55, 424–431). However, the results of various studies have been contradictory and some reports indicate that injection of hyaluronan is not efficient, see Jorgensen et al.
- a composition of hyaluronan–drug conjugate which has an increased concentration of the conjugate in the composition.
- a composition of hyaluronan–drug conjugate which has an increased concentration of the conjugate in the composition.
- concentration of a conjugate in an aqueous composition affects the properties of the composition, which may impact its utility and use of use for the intended medical treatment.
- viscosity increases with concentration and a composition that is too viscous is unsuitable for administration by injection.
- the invention provides an aqueous liquid composition comprising a conjugate of hyaluronan and a pharmaceutically active compound, and a sugar or sugar alcohol, wherein the concentration of the sugar or the sugar alcohol in the composition is 10–100 mg/mL, and the concentration of the conjugate in the composition is 2–50 mg/mL.
- the invention also provides the aqueous liquid composition for use as a medicament.
- the invention also provides the aqueous liquid composition for use in the treatment or prevention of a joint disease, for example osteoarthritis.
- the invention also provides the aqueous liquid composition for use in cataract surgery or cancer therapy.
- the invention also provides a use of the aqueous liquid composition for the manufacture of a medicament for use in human or veterinary medicine.
- the invention also provides a method of treating or preventing a disease or disorder in a subject comprising administration of a therapeutically effective amount of the aqueous liquid composition, for example a method of treating or preventing a joint disease such as osteoarthritis, or a method of treating or preventing a cataract, or a method of treating or preventing a cancer.
- the invention further provides a method for manufacturing the aqueous liquid composition, comprising mixing the conjugate of hyaluronan and a pharmaceutically active compound with an aqueous solution of the sugar or the sugar alcohol.
- the invention further provides a kit comprising a conjugate of hyaluronan and a pharmaceutically active compound, and an aqueous sugar or sugar alcohol solution having a concentration of 20–100 mg/mL, wherein the ratio of the mass of conjugate to sugar or sugar alcohol is 1:50 to 5:1.
- the invention further provides a kit comprising a conjugate of hyaluronan and a pharmaceutically active compound, and a sugar or sugar alcohol, wherein the ratio of the mass of conjugate to sugar or sugar alcohol is 1:50 to 5:1.
- Figure 1 is a photograph of aqueous liquid compositions comprising 15 mg/mL of Conjugate 1 in 5% glucose (left), and 15 mg/mL Conjugate 1 in 0.9% NaCl (right).
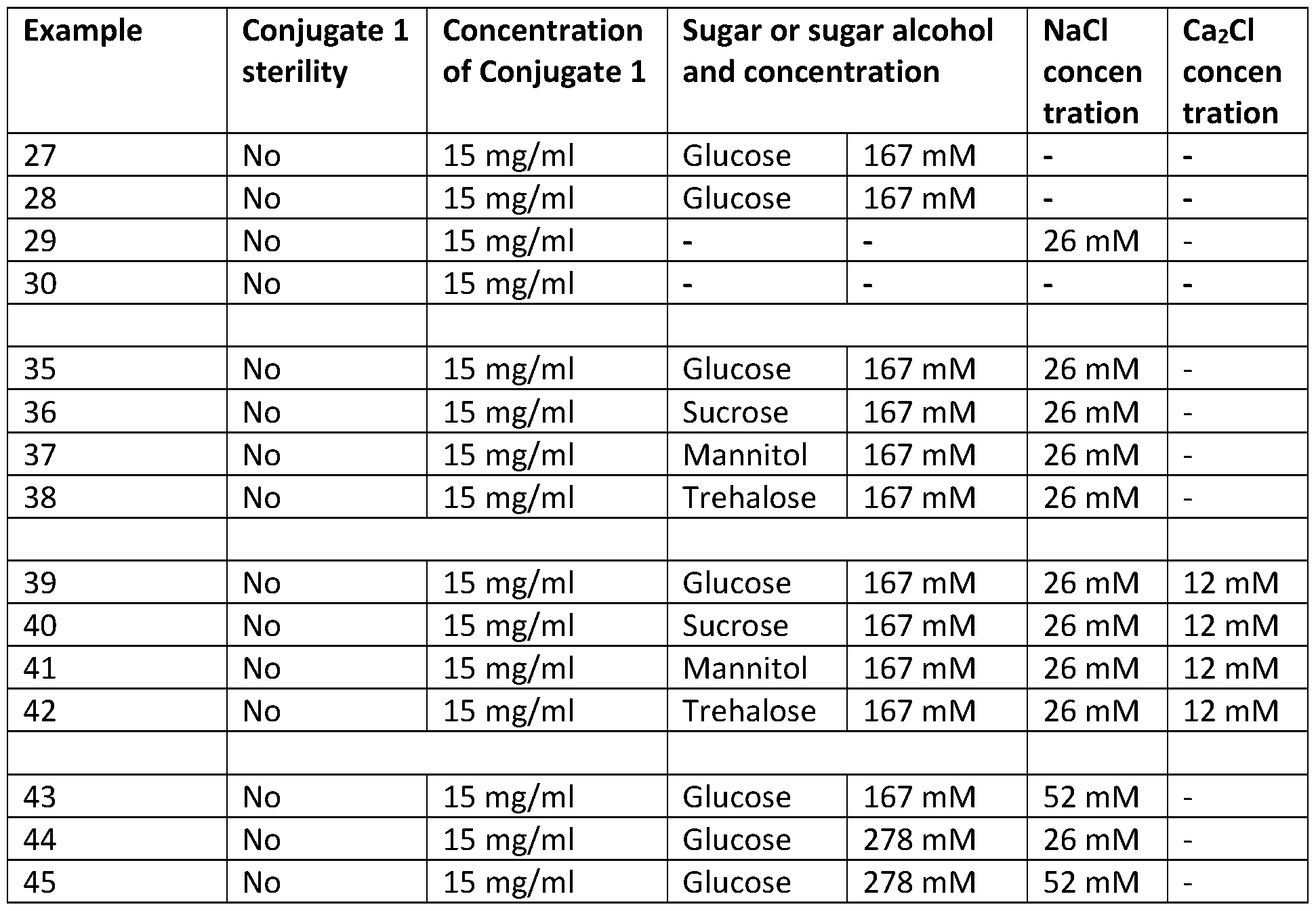
- Figure 2 shows a table of viscosity measurements of aqueous liquid compositions comprising 15 mg/mL Conjugate 1 and 3% glucose (167 mM) + 0.15% NaCl (26 mM) (Solution 27), 5% glucose (278 mM) (Solution 28), 0.9% NaCl (156 mM) (Solution 29), or MilliQ water (Solution 30).
- Figure 3 shows a table of viscosity measurements and the calculated zero shear rate viscosity of aqueous liquid compositions comprising 15 mg/mL sodium hyaluronate and 3% glucose (167 mM) + 0.15% NaCl (26 mM) (Solution 31), 5% glucose (278 mM) (Solution 32), 0.9% NaCl (156 mM) (Solution 33), or MilliQ water (Solution 34).
- Figure 4 shows a graph of viscosity measurements of aqueous liquid compositions comprising 15 mg/mL Conjugate 1 and 3% glucose (167 mM) + 0.15% NaCl (26 mM) (Solution 27), 5% glucose (278 mM) (Solution 28), 0.9% NaCl (156 mM) (Solution 29), or MilliQ water (Solution 30).
- Figure 5 shows a graph of viscosity measurements of aqueous liquid compositions comprising 15 mg/mL sodium hyaluronate and 3% glucose (167 mM) + 0.15% NaCl (26 mM) (Solution 31), 5% glucose (278 mM) (Solution 32), 0.9% NaCl (156 mM) (Solution 33), or MilliQ water (Solution 34).
- Figure 6 shows a table of viscosity measurements and the calculated zero shear rate viscosity of aqueous liquid compositions comprising 15 mg/mL Conjugate 1 with 3% glucose (167 mM) + 0.15% NaCl (26 mM) (Solution 35), 5.7% sucrose (167 mM) + 0.15% NaCl (26 mM) (Solution 36), 3% mannitol (167 mM) + 0.15% NaCl (26 mM) (Solution 37), or 5.7% trehalose (167 mM) + 0.15% NaCl (26 mM) (Solution 38).
- Figure 7 and Figure 8 show graphs (logarithmic scale y-axis and linear scale y-axis, respectively) of viscosity measurements of aqueous liquid compositions comprising 15 mg/mL Conjugate 1 with 3% glucose (167 mM) + 0.15% NaCl (26 mM) (Solution 35), 5.7% sucrose (167 mM) + 0.15% NaCl (26 mM) (Solution 36), 3% mannitol (167 mM) + 0.15% NaCl (26 mM) (Solution 37), or 5.7% trehalose (167 mM) + 0.15% NaCl (26 mM) (Solution 38).
- Figure 9 shows a table of viscosity measurements of aqueous liquid compositions comprising 15 mg/mL Conjugate 1 with 3% glucose (167 mM) + 0.15% NaCl (26 mM) + 0.13 % CaCl2 (12 mM) (Solution 39), 5.7% sucrose (167 mM) + 0.15% NaCl (26 mM) + 0.13 % CaCl2 (12 mM) (Solution 40), 3% mannitol (167 mM) + 0.15% NaCl (26 mM) + 0.13 % CaCl 2 (12 mM) (Solution 41), or 5.7% trehalose (167 mM) + 0.15% NaCl (26 mM) + 0.13 % CaCl 2 (12 mM) (Solution 42).
- Figure 10 shows a graph of viscosity measurements of aqueous liquid compositions comprising 15 mg/mL Conjugate 1 with 3% glucose (167 mM) + 0.15% NaCl (26 mM) + 0.13 % CaCl 2 (12 mM) (Solution 39), 5.7% sucrose (167 mM) + 0.15% NaCl (26 mM) + 0.13 % CaCl 2 (12 mM) (Solution 40), 3% mannitol (167 mM) + 0.15% NaCl (26 mM) + 0.13 % CaCl 2 (12 mM) (Solution 41), or 5.7% trehalose (167 mM) + 0.15% NaCl (26 mM) + 0.13 % CaCl 2 (12 mM) (Solution 42).
- Figure 11 shows a table of viscosity measurements and the calculated zero shear rate viscosity of aqueous liquid compositions comprising 15 mg/mL Conjugate 1 with 3% glucose (167 mM) + 0.3% NaCl (52 mM) (Solution 43), 5% glucose (278 mM) + 0.15% NaCl (26 mM) (Solution 44), or 5% glucose (278 mM) + 0.3% NaCl (52 mM) (Solution 45).
- Figure 12 and Figure 13 show graphs (logarithmic scale y-axis and linear scale y-axis, respectively) of viscosity measurements of aqueous liquid compositions comprising 15 mg/mL Conjugate 1 with 3% glucose (167 mM) + 0.3% NaCl (52 mM) (Solution 43), 5% glucose (278 mM) + 0.15% NaCl (26 mM) (Solution 44), or 5% glucose (278 mM) + 0.3% NaCl (52 mM) (Solution 45) or 3% glucose (167 mM) + 0.15% NaCl (26 mM) (Solution 35).
- the invention provides an aqueous liquid composition
- aqueous liquid composition comprising a conjugate of hyaluronan and a pharmaceutically active compound, and a sugar or sugar alcohol, wherein the concentration of the sugar or the sugar alcohol in the composition is 10–100 mg/mL, and the concentration of the conjugate in the composition is 2–50 mg/mL.
- the compositions of the present invention have been found to exhibit lower viscosity than aqueous liquid compositions having the same concentration of the conjugate in the composition wherein the composition does not comprise a sugar or sugar alcohol.
- Pharmaceutical compositions, particularly those administered parenterally are generally required to be isotonic with plasma, so as to avoid damage to tissue when administered.
- a solution that is commonly used to prepare liquid pharmaceutical formulations is isotonic saline, a 0.9 w/v% aqueous NaCl solution, also referred to as normal saline.
- Isotonic glucose (5 w/v% aqueous glucose) is also commonly used to prepare liquid pharmaceutical formulations.
- the inventors have found that, surprisingly, when an aqueous liquid composition of a conjugate of hyaluronan and a pharmaceutically active compound suitable for parenteral administration is prepared with some or all of the NaCl replaced so that the solution comprises a sugar or sugar alcohol, the viscosity of the formulation is significantly lower compared with that of a corresponding composition prepared with a standard concentration of NaCl solution (0.9 w/v%).
- aqueous liquid compositions comprising a sugar and hyaluronic acid that has not been conjugated to a drug have been found to be more viscous compared with that of a corresponding composition prepared with a standard concentration of NaCl solution (0.9 w/v%), i.e. compared to the corresponding compositions not comprising a sugar.
- the inventors have found that the more NaCl that is replaced with a sugar or sugar alcohol, the lower the viscosity achieved (i.e. that the presence of salt in the composition significantly increases viscosity), and that the lowest viscosities are achieved when all NaCl is replaced with glucose.
- a result of this surprising effect is that, by incorporating a sugar or a sugar alcohol into an aqueous liquid composition of a conjugate of hyaluronan and a pharmaceutically active compound in place of some or all of the NaCl that is commonly used in solutions suitable for parenteral administration, the inventors have been able to prepare more concentrated solutions of the conjugate while achieving an acceptable viscosity at the same time as retaining an acceptable tonicity.
- the viscosity is accordingly sufficiently low for the solution to be injectable and able to be handled without additional steps being needed to deal with high viscosity of the formulation.
- This is beneficial for uses of the compositions that require an increased dosage of the pharmaceutically active compound without increasing the volume of the composition.
- Hyaluronic acid is well known and widely used in medical applications.
- Conjugates can be prepared with various pharmaceutically active compounds by conventional chemical synthetic routes.
- Numerous conjugates of hyaluronic acid and a pharmaceutically active compound are known in the art.
- such conjugates are known from WO2007/126154, Dong et al. (Improved stability and tumor targeting of 5-fluorouracil by conjugation with hyaluronan, Journal of Applied Polymer Science, 130(2), 927–932), and WO2015/128787.
- the pharmaceutically active compound is linked to the hyaluronic acid by a linker group.
- linker groups have been proposed and certain linkers have advantages in certain situations and uses.
- a linker releases the pharmaceutically active compound from the hyaluronic acid when the conjugate is in a physiological environment.
- a linker comprises at least two atoms in its chain, with side groups as appropriate.
- the linker comprises a chain of 2 to 15 atoms length connecting the hyaluronic acid and the pharmaceutically active compound.
- a linker can be most conveniently attached to the hyaluronic acid polymer by attachment to an acid group or to acid groups, for example by formation of an ester or an amide group.
- Many pharmaceutically active compounds contain groups that can be used as attachment points for a linker. Examples of suitable attachment point groups are acid groups, alcohol groups and amine groups.
- Pharmaceutically active compounds that are of interest for attachment to hyaluronic acid include nonsteroidal anti-inflammatory drugs (NSAIDs).
- NSAIDs nonsteroidal anti-inflammatory drugs
- a prominent example of such a drug is diclofenac and diclofenac contains an acid group. That acid group can conveniently be used as the attachment point to the linker. That can be achieved, for example, by formation of an ester or an amide group.
- conjugates of hyaluronic acid and diclofenac are with structures of this type are known from, for example, WO2007/126154 and WO2015/128787.
- a conjugate of hyaluronic acid and a pharmaceutically active compound can comprise hyaluronic acid having free hemi-ester groups and a pharmaceutically active compound bound to the hyaluronan via reacted hemi-ester groups (becoming ester groups or amides), thereby forming a linker of chain length L of 2–9 atoms.
- some of the hemi-ester groups are free and others are bound to the pharmaceutically active compound.
- the hyaluronic acid conjugates may be manufactured by providing hyaluronic acid in solution, reacting the hyaluronic acid in solution with an anhydride reagent (for example succinic anhydride) to provide a hyaluronic acid hemi-ester with a chain of length L between the hyaluronic acid and the ester group, referred to herein as activated hyaluronic acid, and subsequently binding the hyaluronic acid hemi-ester to a pharmaceutically active compound.
- the hemi ester chain comprises a carbon backbone, optionally including one or two oxygen atoms in the backbone.
- the carbon backbone of the hemi ester chain can optionally include one or more branches of alkyl, aryl, oxy-alkyl or oxy-aryl.
- the activated intermediate includes free hemiester groups which may be in the form of salts, e.g. sodium salts, of the ester groups, wherein, in each of the above formulas, –COO – is –COONa.
- HSE hyaluronan-succinyl hemi-esters
- Anhydrides other than succinic anhydride, and esters formed therefrom, may also be used.
- glutaryl-hemi esters are employed.
- the degree of ester substitution can be influenced by changing the proportion of the anhydride reagent to the hyaluronan polymer, the reaction time, and the temperature. Typically, without raising the temperature above room temperature, an average degree of substitution (DS) of up to 3 mol hemi-succinate per mol hyaluronan repeating disaccharide unit can be obtained. In a specific embodiment, the average degree of substitution is 0.5–3 and, in a more specific embodiment, is 1–3 or 2–3 mol hemi-ester, e.g. hemi-succinate, per mol hyaluronan repeating disaccharide unit.
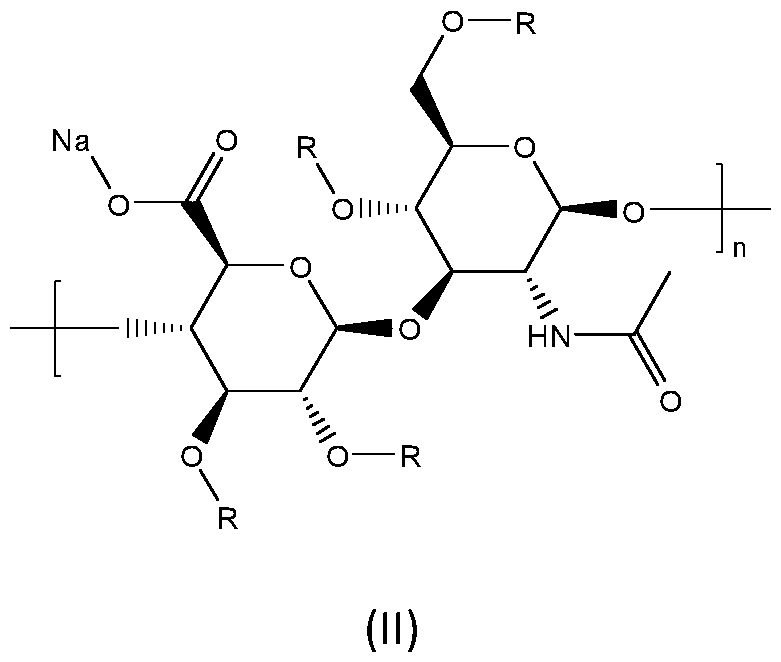
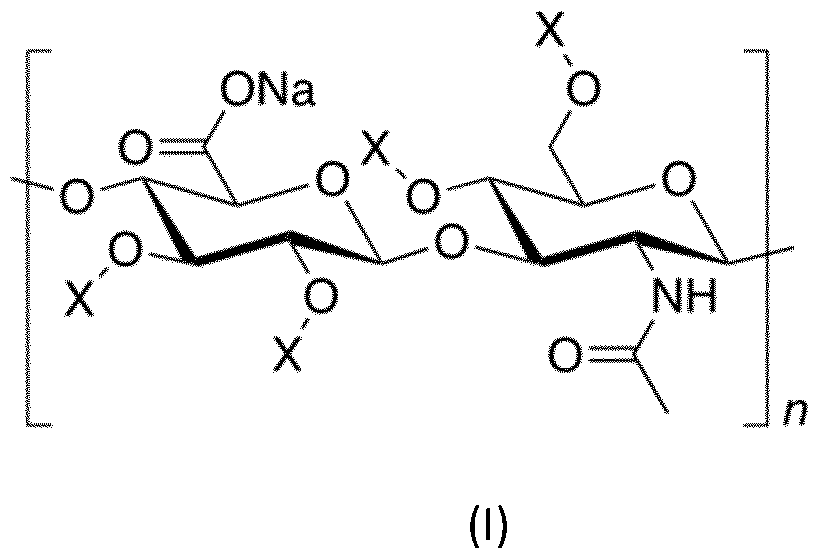
- Formula (I) shows a schematic representation of a conjugate of hyaluronan and a pharmaceutically active compound (which may also be referred to an HSE-drug conjugate) that can be used in the current invention: wherein X is H, –CO–CH2CH2–COONa, –CO–CH2CH2–CO–NH–CH2CH2–O–CH2CH2–O–DRUG, or –CO–CH 2 CH 2 –CO—NH–CH 2 CH 2 –O–CH 2 CH 2 –O–CO–CH 2 CH 2 –CO–DRUG, wherein DRUG represents the pharmaceutically active compound.
- the DRUG may be diclofenac, for example attached through its acid group.
- the drug (i.e. the pharmaceutically active compound) molecules can occupy all carboxyl groups exposed by the HSE, but in practice, higher substitutions can unfavorably change the properties of the polymer, particularly if a solution suitable for injection is desired.
- an average degree of substitution (DS) less than or equal to 0.3 mol drug per mol hyaluronan disaccharide repeating unit is favorable for the formulation of an injectable solution.
- an average substitution degree from 0.01–0.3, in particular 0.05–0.2, mol drug per mol hyaluronan disaccharide repeating unit may be employed.
- other substitution degrees might be preferred.
- the drug (i.e. the pharmaceutically active compound) in the conjugate is dexamethasone.
- the preparation of a suitable HA– dexamethasone conjugate is described in WO2015/128787.
- the pharmaceutically active compound for example the DRUG in the of Formula (I) above
- diclofenac for example diclofenac attached through its acid group.
- the pharmaceutically active compound i.e.
- the drug in the conjugate for example the DRUG in the of Formula (I) above is a non-steroidal anti- inflammatory drug (for example selected from the group consisting of diclofenac, ibuprofen, ketoprofen, bromfenac, aceclofenac, flunixin and carprofen), a steroid (for example selected from the group consisting of dexamethasone and prednisolone), an antibiotic (for example selected from the group consisting of metronidazole, azithromycin and levofloxacin), a plant alkaloid (for example podophyllotoxin), an antiviral (for example aciclovir), a chemotherapeutic agent (for example selected from the group consisting of paclitaxel, docetaxel, doxorubicin and daunorubicin), a retinoid (for example adapalene), an immunosuppressant (for example selected from the group consisting of cyclosporine and tacrolimus
- the pharmaceutically active compound i.e. the drug in the conjugate for example the DRUG in the of Formula (I) above
- the pharmaceutically active compound is a non-steroidal anti-inflammatory drug (for example selected from the group consisting of diclofenac, ibuprofen, ketoprofen, bromfenac and aceclofenac) or a steroid (for example selected from the group consisting of dexamethasone and prednisolone).
- the drug (i.e. the pharmaceutically active compound) in the conjugate is cisplatin.
- compositions that may be used include ibuprofen, ketoprofen, naproxen, bromfenac, aceclofenac, prednisolone, metronidazole, podophyllotoxin, paclitaxel, flunixin, carprofen, docetaxel, doxorubicin, daunorubicin, adapalene, azithromycin, levofloxacin, aciclovir, cyclosporine, tacrolimus, latanoprost, cromoglicic acid, levocabastine, nedocromil, olopatadine, bepotastine and morphine.
- the conjugate does not comprise sulphate groups. In another embodiment of the invention, the conjugate does not comprise sulphur containing groups. In another embodiment of the invention, the conjugate of the invention does not comprise sulphated sodium hyaluronate or sulphated hyaluronic acid groups (for example the compound does not comprise -OH groups that have been converted to sulphate groups, for example by esterification with sulphuric acid). It will be understood that in solution many of the carboxylate groups will be in their ionised form, the level typically depending on the pH of the solution. It will also be understood that the conjugates can be in the form of the sodium salt.
- a conjugate of hyaluronan and a pharmaceutically active compound should be understood to include a conjugate of hyaluronic acid in all physiological forms and a pharmaceutically active compound, including a conjugate of sodium hyaluronate (NaHA) and a pharmaceutically active compound.
- a conjugate of hyaluronic acid and a pharmaceutically active compound should (unless the context dictates otherwise) be understood to include a conjugate of hyaluronic acid in all physiological forms and a pharmaceutically active compound (i.e.
- hyaluronan is hyaluronic acid or sodium hyaluronate.
- the molecular weight of polymers such as hyaluronan (for example hyaluronic acid), and conjugates of hyaluronan is expressed as an average molecular weight, or as molecular mass distribution or molecular weight distribution, because polymers are made up of many molecular weights, or a distribution of chain lengths. The average or distribution can be defined in different ways, depending on the statistical method used.
- M n the number average molar mass
- M w the weight- (or mass-) average molar mass
- molecular weight or average molar mass when molecular weight or average molar mass are referred to herein, it may be Mn or Mw. Preferably it is Mw (for example Mw wherein AF4 is used as the method for the measurement of Mw; and even more preferably wherein AF4 combined with UV-FL- MALS-RI detectors is used to directly measure Mw using the light scattering and concentration data).
- Average molecular weight of the conjugates can be assessed by various methods known in the art. For example, average molecular weight of the conjugates can be measured by asymmetrical flow field-flow fractionation (AF4) (Kwon et al., Depolymerization study of sodium hyaluronate by flow field-flow fractionation/multiangle light scattering, Anal. Bioanal.
- AF4 asymmetrical flow field-flow fractionation
- AF4 has been found to be the most reliable method for the assessment of average molar mass (in particular for weight-average molar mass), and in particular AF4 combined with UV-FL-MALS-RI detectors used to directly obtain M w using the light scattering and concentration data.
- the hyaluronan has an average molecular weight of about 40,000 to 4,000,000 Da.
- the hyaluronan has a molecular weight of about 80,000 to 2,000,000 Da, for example about 100,000 to 2,000,000 Da.
- the hyaluronan has a molecular weight of about 100,000 to 1,500,000 Da, for example about 200,000 to 1,250,000 Da or about 250,000 to 1,000,000 Da.
- the hyaluronan has a molecular weight of about 500,000 to 1,250,000 Da. In an especially preferred embodiment, the hyaluronan has a molecular weight of about 500,000 to 1,000,000 Da. In another especially preferred embodiment, the hyaluronan has a molecular weight of about 200,000 to 750,000 Da. In another especially preferred embodiment, the hyaluronan has a molecular weight of about 200,000 to 400,000 Da.
- the hyaluronan has a molecular weight of about 16,000 to 2,400,000 Da, about 40,000 to 1,200,000 Da, or about 40,000 to 900,000 Da (for example, about 120,000 to 750,000 Da, about 150,000 to 600,000 Da, about 300,000 to 750,000 Da, about 500,000 to 1,000,000 Da, or about 300,000 to 500,000 or 300,000 to 420,000 Da).
- the hyaluronan has a molecular weight of about 200,000 – 500,000 Da (for example about 200,000 to 400,000 Da, about 250,000 to 400,000 Da, about 250,000 to 420,000 Da, about 250,000 to 420,000 Da, about 250,000 to 400,000 Da, or about 300,000 to 400,000 Da).
- the hyaluronan has a molecular weight of about 250,000 to 400,000 Da, or about 300,000 to 400,000 Da.
- M n or M w there is little difference between average molecular weight for hyaluronan, or conjugates of hyaluronan, when defined using M n or M w .
- the average molecular weights defined in this paragraph (and defined herein) are the Mw.
- the average molecular weights may be the M n .
- the average molecular weights are the M w and AF4 is used as the method for the measurement of the M w (for example, AF4 combined with UV-FL-MALS-RI detectors is used to directly measure M w using the light scattering and concentration data).
- the invention provides an aqueous liquid composition comprising a conjugate of hyaluronan and a pharmaceutically active compound and a sugar, wherein the conjugate is produced by providing hyaluronan in solution, reacting the hyaluronan in solution with an anhydride reagent to provide a hyaluronan hemi-ester having hemi-ester groups, and subsequently bonding the hyaluronan hemi-ester to the pharmaceutically active compound.
- the hyaluronan in solution is reacted with an anhydride reagent, for example succinic anhydride.
- a solution of the hyaluronan may be provided using a suitable solvent for solid sodium hyaluronate, for example formamide, with the addition of a tertiary amine, a pyridine or a substituted pyridine.
- the solvent is pyridine, optionally with the addition of 4-dimethyl-amino-pyridine (DMAP) or 2,6-dimethyl- 4-dimethylamino-pyridine.
- DMAP 4-dimethyl-amino-pyridine
- This procedure allows for dissolution of the solid sodium hyaluronate without extra steps such as ion exchange to the acid form, hyaluronic acid, that are typically used in the prior art.
- dimethyl formamide (DMF) is used as a solvent.
- DMF dimethyl formamide
- sodium hyaluronate is not soluble, and an ion exchange to the acid form of hyaluronan in water or transfer to an amine salt is required before dissolution in DMF, followed by evaporation to remove water, re-dissolution in DMF and then addition of reagents.
- the conjugate is produced by the addition of reagents directly after dissolution in the formamide solvent, thus giving a simpler and shorter procedure than those commonly employed in the prior art for the synthesis of the hemi-ester of Formula (II): in which R is H or the ester chain, for example, –CO–CH 2 –CH 2 –COO–Na in the case of succinic anhydride.
- the conjugate is produced by the addition of reagents directly after dissolution in the formamide solvent, thus giving a simpler and shorter procedure than those commonly employed in the prior art for the synthesis of the hemi-ester of Formula (I): in which X is H or the ester chain, for example, –CO–CH2–CH2–COO–Na in the case of succinic anhydride.
- the hemi-ester for example succinylated hyaluronan (HSE)
- HSE succinylated hyaluronan
- a desired pharmaceutically active agent can be provided with an amino functionality.
- the amino functionality is combined with a longer moiety in order to space the pharmaceutically active agent from the hyaluronan and to provide better access for the degrading enzymes in vivo.
- coupling of the amine-functionalized pharmaceutically active agent to the hyaluronan hemi-ester group may be performed in water-containing media, i.e., water or an aqueous solvent, for example in a DMF-water mixture or in suitable water-based buffers. This feature makes it possible to link molecules that are difficult to dissolve in aprotic solvents.
- the conjugate is produced by: providing hyaluronan in solution, reacting the hyaluronan in solution with an anhydride reagent to provide a hyaluronan hemi-ester having hemi-ester groups of the formula: –C(O)–(CH2)m–COO – , where m is 2–8, –C(O)–(CH2)p–O–(CH2)q–COO – , where p and q are both 1–4, or –C(O)–(CH2)r–O–(CH2)s–O–(CH2)t–COO – , where r and t are 1–2 and s is 2; and subsequently bonding the hyaluronan hemi-ester to a pharmaceutically active compound.
- the conjugate is produced by: providing hyaluronan in solution, reacting the hyaluronan in solution with an anhydride reagent to provide a hyaluronan hemi-ester having hemi-ester groups of the formula: –CO–CH 2 CH 2 –COO – , –CO–CH2CH2–CO–NH–CH2CH2–O–CH2CH2–O – , or –CO–CH2CH2–CO–NH–CH2CH2–O–CH2CH2–O–CO–CH2CH2–CO —; and subsequently bonding the hyaluronan hemi-ester to a pharmaceutically active compound.
- the conjugate of the invention is sterile (i.e. the conjugate of the invention has been sterilised).
- the conjugate of the invention is sterile and/or has been sterilised by ionising radiation, for example wherein the ionising radiation is beta, gamma or X-ray radiation, and preferably gamma radiation.
- the conjugate of the invention has been sterilised according to the following method: providing the conjugate of hyaluronan (for example sodium hyaluronate or hyaluronic acid) and a pharmaceutically active compound; and exposing the conjugate to ionising radiation, for example wherein the ionising radiation is beta or X-ray radiation, and preferably gamma radiation.
- the conjugate is exposed to the ionising radiation under an inert atmosphere (for example, wherein the inert atmosphere is an argon atmosphere or a nitrogen atmosphere).
- the dose of ionising radiation is 5-40 kGy, preferably 8-40 kGy or 20-35 kGy, for example 25 kGy or 32 kGy. In one embodiment, the dose of ionising radiation is around 25 kGy and is verified by the VDmax25 method in ISO 11137-2:2013).
- the conjugate of the invention is sterile and/or has been obtained by (or is obtainable by) the following method: providing a conjugate of hyaluronan (for example sodium hyaluronate or hyaluronic acid) and a pharmaceutically active compound; and exposing the conjugate to ionising radiation, for example wherein the ionising radiation is beta, gamma or X-ray radiation, and preferably gamma radiation.
- the conjugate is exposed to the ionising radiation under an inert atmosphere (for example, wherein the inert atmosphere is an argon atmosphere or a nitrogen atmosphere).
- the dose of ionising radiation is around 5-40 kGy, preferably around 8-40 kGy or around 20-35 kGy, for example around 25 kGy or around 32 kGy. In one embodiment, the dose of ionising radiation is around 25 kGy and is verified by the VDmax25 method in ISO 11137-2:2013).
- the conjugate of the invention is sterile and is characterised by a sterility assurance level of 10 -3 or better, 10 -4 or better, 10 -5 or better, 10 -6 or better; preferably 10 -6 or better.
- the sterility assurance level is the probability that a single unit that has been subjected to sterilization nevertheless remains nonsterile, i.e. the probability of any surviving microorganism following sterilisation.
- a SAL of 10 -3 means a 1 in 1,000 chance of a non-sterile unit.
- a SAL of 10 -6 means a 1 in 1,000,000 chance of a non-sterile unit.
- a SAL of 10 -6 is generally required for a medical material to be used in the body, whereas a SAL of 10 -3 may be acceptable for materials that are intended for intact skin contact only (Ph. Eur., 11 th edition, 5.1.1. monograph and ISO 11137-1:2006, -2:2013 and -3:2017 guidelines).
- the conjugate of the invention is sterile and/or has been subjected to a treatment such as dust removal, bacteria removal, and/or sterilization by, for example, filter filtration (for example membrane filter filtration).
- a treatment such as dust removal, bacteria removal, and/or sterilization by, for example, filter filtration (for example membrane filter filtration).
- filter filtration for example membrane filter filtration
- Such treatments including methods of sterilisation by filter filtration, are described in WO2020/101013 A1.
- the conjugate of the invention is sterile and/or has been subjected to a dust removal and sterilization step by filtration (for example membrane filter filtration).
- a membrane filter for example a commercially available membrane filter
- a sterilized container, sterilized injector, syringe barrel, or the like as appropriate.
- a membrane filter with a pore size of 0.05 ⁇ m to 1.0 ⁇ m can be used, for example 0.1 to 1.0 ⁇ m, and especially 0.22 ⁇ m.
- the viscosity of the aqueous liquid compositions of the present invention comprising a conjugate of hyaluronan and a pharmaceutically active compound, and a sugar or sugar alcohol, are significantly lower compared with that of a corresponding composition prepared with a standard concentration of NaCl solution (0.9 w/v%), the aqueous liquid compositions of the present invention have improved filterability and therefore can advantageously be sterilised by and/or undergo a treatment (e.g. dust removal, bacteria removal, and/or sterilization) by filter filtration.
- a treatment e.g. dust removal, bacteria removal, and/or sterilization
- the aqueous liquid composition may be subjected to filter filtration.
- the aqueous liquid composition may be subjected to filter filtration before or after the composition is sterilised.
- the filtering of the composition may be carried out by membrane filter filtration.
- a membrane filter for example a commercially available membrane filter
- a sterilized container, sterilized injector, syringe barrel, or the like can be used, optionally with a sterilized container, sterilized injector, syringe barrel, or the like, as appropriate.
- a membrane filter with a pore size of 0.05 ⁇ m to 20 ⁇ m can be used, for example 0.4 to 8.0 ⁇ m (for example, 0.4 ⁇ m, 0.5 ⁇ m, 1 ⁇ m, 2 ⁇ m, 3 ⁇ m, 4 ⁇ m, 5 ⁇ m, 6 ⁇ m, 7 ⁇ m or 8 ⁇ m), 0.5 to 6 ⁇ m or 1 to 5 ⁇ m, and especially 5 ⁇ m.
- An “aqueous liquid composition” in the context of the present invention includes any mixture resulting from admixture of or combination of the components defined to be in the composition with water, whether fully dissolved or not. In preferred embodiments, the components are fully dissolved.
- the aqueous liquid composition of the present invention comprises a sugar or sugar alcohol, preferably a sugar or mannitol, and more preferably a sugar.
- the aqueous liquid composition comprises a sugar or sugar alcohol in solution.
- the sugar is glucose, sucrose, fructose or trehalose and the sugar alcohol is mannitol, ethylene glycol, glycerol, sorbitol or xylitol.
- the sugar is glucose or sucrose.
- the sugar is glucose or trehalose.
- the sugar is glucose or fructose.
- the sugar is glucose, sucrose, or trehalose and the sugar alcohol is mannitol.
- the sugar is glucose. In one embodiment, the sugar is not sucrose.
- the aqueous liquid composition comprises a sugar.
- the sugar is selected from the group consisting of glucose, sucrose, fructose and trehalose.
- the sugar is glucose or sucrose.
- the sugar is glucose or trehalose.
- the sugar is glucose or fructose.
- the sugar is glucose. In one embodiment, the sugar is not sucrose.
- the concentration of the hyaluronan conjugate in the composition is 2- 50 mg/mL. In a preferred embodiment, the concentration of the hyaluronan conjugate in the composition is 10-40 mg/mL.
- the concentration of the hyaluronan conjugate in the composition is 12–30 mg/mL, for example 12–21 mg/mL. In a more preferred embodiment, the concentration of the hyaluronan conjugate in the composition is 15–21 mg/mL. In an especially preferred embodiment, the concentration of the hyaluronan conjugate in the composition is 21 mg/mL. In an embodiment, the concentration of the sugar or sugar alcohol in the composition is 10– 100 mg/mL. In a preferred embodiment, the concentration of the sugar or sugar alcohol in the composition is 20–100 mg/mL, for example 35–70 mg/mL. In a more preferred embodiment, the concentration of the sugar or sugar alcohol in the composition is 40–60 mg/mL.
- the concentration of the sugar or sugar alcohol in the composition is 45–55 mg/mL. In an especially preferred embodiment, the concentration of the sugar or sugar alcohol in the composition is 50 mg/mL.
- the aqueous liquid composition comprises a sugar, for example glucose.
- the concentration of the sugar or sugar alcohol in the composition is 20–60 mg/mL. In a preferred embodiment, the concentration of the sugar or sugar alcohol in the composition is 30–50 mg/mL. In a more preferred embodiment, the concentration of the sugar or sugar alcohol in the composition is 40–50 mg/mL. In an especially preferred embodiment, the concentration of the sugar or sugar alcohol in the composition is 50 mg/mL.
- the aqueous liquid composition comprises a sugar, for example glucose.
- the aqueous liquid composition may comprise additional components as well as the hyaluronan conjugate and the sugar or sugar alcohol.
- the aqueous liquid composition may contain NaCl or another salt (for example NaCl, KCl, CaCl 2 , NaBr, MgCl 2 , Choline chloride, NaHCO2, NaHPO4, or KH2PO4; preferably NaCl, KCl, CaCl2, NaHCO2, NaHPO4, or KH2PO4).
- the aqueous liquid composition comprises In an embodiment, the concentration of NaCl or other salt in the composition is 0.1–50 mg/mL.
- the concentration of NaCl or other salt in the composition is 0.5–3 mg/mL. In a more preferred embodiment, the concentration of NaCl or other salt in the composition is 1–2 mg/mL. In an especially preferred embodiment, the concentration of NaCl or other salt in the composition is 1.5 mg/mL. In such embodiments, preferably the aqueous liquid composition comprises NaCl.
- the concentration of the sugar or sugar alcohol in the composition is preferably 10–90 mg/mL. In a preferred embodiment, the concentration of the sugar or sugar alcohol in the composition is 15–60 mg/mL. In a more preferred embodiment, the concentration of the sugar or sugar alcohol in the composition is 20–50 mg/mL.
- the concentration of the sugar or sugar alcohol in the composition is 25–40 mg/mL. In an especially preferred embodiment, the concentration of the sugar or sugar alcohol in the composition is 30 mg/mL.
- the aqueous liquid composition comprises a sugar, for example glucose.
- the aqueous liquid composition comprises a sugar or sugar alcohol at a concentration of 10–90 mg/mL and NaCl or other salt at a concentration of 0.1–5 mg/mL.
- the aqueous liquid composition comprises a sugar or sugar alcohol at a concentration of 15–60 mg/mL and NaCl or other salt at a concentration of 0.5–3 mg/mL.
- the aqueous liquid composition comprises a sugar or sugar alcohol at a concentration of 20–50 mg/mL and NaCl or other salt at a concentration of 1–2 mg/mL.
- the aqueous liquid composition comprises a sugar or sugar alcohol at a concentration of 25–40 mg/mL and NaCl or other salt at a concentration of 1–1.5 mg/mL.
- the aqueous liquid composition comprises a sugar (for example glucose) and NaCl.
- the aqueous liquid composition comprises a sugar or sugar alcohol at a concentration of 10–90 mg/mL and NaCl or other salt at a concentration of 0.1–5 mg/mL, and the concentrations of the sugar or sugar alcohol and NaCl or other salt are such that the aqueous liquid composition is isotonic.
- the aqueous liquid composition comprises a sugar or sugar alcohol at a concentration of 15–60 mg/mL and NaCl or other salt at a concentration of 0.5–3 mg/mL, and the concentrations of the sugar or sugar alcohol and NaCl or other salt are such that the aqueous liquid composition is isotonic.
- the aqueous liquid composition comprises a sugar or sugar alcohol at a concentration of 20–50 mg/mL and NaCl or other salt at a concentration of 1–2 mg/mL, and the concentrations of the sugar or sugar alcohol and NaCl or other salt are such that the aqueous liquid composition is isotonic.
- the aqueous liquid composition comprises a sugar or sugar alcohol at a concentration of 25–40 mg/mL and NaCl or other salt at a concentration of 1–1.5 mg/mL, and the concentrations of the sugar or sugar alcohol and NaCl or other salt are such that the aqueous liquid composition is isotonic.
- the aqueous liquid composition comprises a sugar (for example glucose) and NaCl.
- the aqueous liquid composition comprises a sugar or sugar alcohol at a concentration of 30 mg/mL and NaCl or other salt at a concentration of 1.5 mg/mL, and the concentrations of the sugar or sugar alcohol and NaCl or other salt are such that the aqueous liquid composition is isotonic.
- the aqueous liquid composition comprises a sugar (for example glucose) at a concentration of 30 mg/mL and NaCl at a concentration of 1.5 mg/mL, and the concentrations of the sugar and NaCl are such that the aqueous liquid composition is isotonic.
- the aqueous liquid composition comprises salts and other compounds as well as or in place of NaCl.
- the composition may contain KCl, CaCl 2 , NaBr, MgCl 2 , Choline chloride, NaHCO 2 , NaHPO 4 , KH 2 PO 4 , citric acid buffer, phosphate buffered saline or Ringer ⁇ s solution, or combinations thereof (including all of them) (preferably KCl, CaCl 2 , NaHCO 2 , NaHPO 4 , KH 2 PO 4 , citric acid buffer, phosphate buffered saline or Ringer ⁇ s solution, or combinations thereof (including all of them)).
- the aqueous liquid composition is substantially free from KCl, CaCl 2 , NaBr, MgCl 2 , Choline chloride, NaHCO 2 , NaHPO 4 , KH 2 PO 4 , or combinations thereof (including all of them), for example the aqueous liquid composition is substantially free from CaCl 2 .
- the aqueous liquid composition is substantially free from salts other than NaCl and salts of hyaluronan (for example sodium hyaluronate).
- “substantially free from” is taken to mean free from any added component (e.g.
- the aqueous liquid composition of the invention consists essentially of, or consists of, a conjugate of hyaluronan and a pharmaceutically active compound, and a sugar or sugar alcohol.
- the aqueous liquid composition of the invention consists essentially of, or consists of, a conjugate of hyaluronan and a pharmaceutically active compound, and a sugar or sugar alcohol, and a NaCl or another salt.
- the aqueous liquid composition of the invention consists essentially of, or consists of, a conjugate of hyaluronan and a pharmaceutically active compound, and a sugar or sugar alcohol, and NaCl.
- the aqueous liquid composition of the invention consists essentially of, or consists of, a conjugate of hyaluronan and a pharmaceutically active compound, and a sugar (for example glucose) and NaCl.
- the liquid composition may additionally, or alternatively, include one or more further sugar and/or sugar alcohol (for example, one or more additional sugar and/or sugar alcohol selected from the group consisting of glucose, sucrose, fructose, trehalose, mannitol, ethylene glycol, glycerol, sorbitol or xylitol; preferably, one or more additional sugar selected from the group consisting of glucose, sucrose, fructose and trehalose).
- the aqueous liquid composition has a pH of 4–8, for example a pH of 5– 7.
- the composition has a pH of 6.5.
- the aqueous liquid composition is sterile. Sterility assurance level may be used to express sterility.
- the sterility assurance level is the probability that a single unit that has been subjected to sterilization nevertheless remains nonsterile, i.e. the probability of any surviving microorganism following sterilisation.
- a SAL of 10 -3 means a 1 in 1,000 chance of a non-sterile unit.
- a SAL of 10 -6 means a 1 in 1,000,000 chance of a non- sterile unit.
- a SAL of 10 -6 is generally required for a medical material to be used in the body, whereas a SAL of 10 -3 may be acceptable for materials that are intended for intact skin contact only (Ph. Eur., 11 th edition, 5.1.1. monograph and ISO 11137-1:2006, -2:2013 and - 3:2017 guidelines).
- the aqueous liquid composition is sterile and has a sterility assurance level (SAL) of 10 -3 or better, for example of 10 -3 or better, for example of 10 -5 or better, or for example of 10 -6 or better.
- the aqueous liquid composition is sterile and has a SAL of 10 -6 or better.
- the invention provides a method for manufacturing the aqueous liquid composition as described herein above, comprising mixing the conjugate of hyaluronan and a pharmaceutically active compound with an aqueous solution of the sugar or sugar alcohol.
- the invention also provides a method for manufacturing the aqueous liquid composition as described herein above, comprising mixing the conjugate of hyaluronan and a pharmaceutically active compound with an aqueous solution of the sugar or sugar alcohol and an aqueous solution of NaCl or another salt (for example NaCl, KCl, CaCl2, NaBr, MgCl2, Choline chloride, NaHCO 2 , NaHPO 4 , KH 2 PO 4 , or combinations thereof; preferably an aqueous solution of NaCl, KCl, CaCl 2 , NaHCO 2 , NaHPO 4 , KH 2 PO 4 , or combinations thereof; more preferably an aqueous solution of NaCl).
- NaCl or another salt for example NaCl, KCl, CaCl2, NaBr, MgCl2, Choline chloride, NaHCO 2 , NaHPO 4 , KH 2 PO 4 , or combinations thereof; preferably an aqueous solution of NaCl,
- the method comprises mixing the conjugate of hyaluronan and a pharmaceutically active compound with an aqueous solution of the sugar or sugar alcohol having a concentration of 10–100 mg/mL.
- the aqueous solution of the sugar or sugar alcohol has a concentration of 35–70 mg/mL.
- the aqueous solution of the sugar or sugar alcohol has a concentration of 40– 60 mg/mL.
- the aqueous solution of the sugar or sugar alcohol has a concentration of 45–55 mg/mL.
- the aqueous solution of the sugar or sugar alcohol has a concentration of 50 mg/mL.
- the method comprises mixing the conjugate of hyaluronan and a pharmaceutically active compound with an aqueous solution of the sugar or sugar alcohol at a mass ratio of conjugate to sugar or sugar alcohol of 1:50 to 5:1.
- the ratio of the mass of conjugate to sugar or sugar alcohol is 1:12 to 5:2.
- the ratio of the mass of conjugate to sugar or sugar alcohol is 6:25 to 3:2.
- the ratio of the mass of conjugate to sugar or sugar alcohol is 1:4 to 1:1.
- the ratio of the mass of conjugate to sugar or sugar alcohol is 1:3 to 1:1.5.
- the ratio of the mass of conjugate to sugar or sugar alcohol is 1:2.
- the method comprises mixing the conjugate of hyaluronan and a pharmaceutically active compound with an aqueous solution of the sugar or sugar alcohol, and an aqueous solution of NaCl or another salt having a concentration of 0.1–50 mg/mL.
- the aqueous solution of the NaCl or another salt has a concentration of 0.5–3 mg/mL.
- the aqueous solution of the NaCl or another salt has a concentration of 1–2 mg/mL.
- the aqueous solution of the NaCl or another salt has a concentration of 1.5 mg/mL.
- the aqueous solution of NaCl or another salt is a solution of NaCl.
- the sugar or sugar alcohol is glucose.
- the method comprises mixing the conjugate of hyaluronan and a pharmaceutically active compound with an aqueous solution of the sugar or sugar alcohol, and an aqueous solution of NaCl or another salt, at a mass ratio of sugar or sugar alcohol to NaCl or another salt of 2:1 to 900:1.
- the ratio of the mass of sugar or sugar alcohol to NaCl or another salt is 5:1 to 120:1.
- the ratio of the mass of sugar or sugar alcohol to NaCl or another salt is 10:1 to 50:1.
- the ratio of the mass of sugar or sugar alcohol to NaCl or another salt is 16:1 to 40:1. In a yet more preferred embodiment, the ratio of the mass of conjugate to sugar or sugar alcohol is 20:1. In such embodiments, preferably the aqueous solution of NaCl or another salt is a solution of NaCl. In such embodiments, preferably the sugar or sugar alcohol is glucose.
- conjugate of hyaluronan and a pharmaceutically active compound for use in the method may be in the form of a sterile composition, for example the conjugate is sterile and/or has been sterilised according to the methods described here, or, for example, the conjugate is sterile and/or has been obtained by (or is obtainable by) the sterilisation methods described herein.
- the conjugate for use in the method is sterile and is characterised by a sterility assurance level of 10 -3 or better, 10 -4 or better, 10 -5 or better, 10 -6 or better; preferably 10 -6 or better.
- the sugar or sugar alcohol for use in the method may be in the form of a sterile composition, for example in the form of a sterile glucose solution.
- sugar or sugar alcohol for use in the method may be in the form of a sterile composition, for example a sterile glucose solution (e.g. a glucose solution for injection), and for example wherein the composition has a sterility assurance level (SAL) of 10 -3 or better, for example of 10 -3 or better, for example of 10 -5 or better, or for example of 10 -6 or better.
- the aqueous liquid composition is sterile and has a SAL of 10 -6 or better.
- the NaCl or another salt for use in the method may be in the form of a sterile composition, for example a sterile solution (for example a sterile NaCl solution e.g. sodium chloride solution for injection (also referred to as saline solution for injection)), and for example wherein the composition has a sterility assurance level (SAL) of 10 -3 or better, for example of 10 -3 or better, for example of 10 -5 or better, or for example of 10 -6 or better.
- the aqueous liquid composition is sterile and has a SAL of 10 -6 or better.
- the method may further comprise a step of filter filtration of the aqueous liquid composition.
- the aqueous liquid composition is a sterile aqueous liquid composition.
- the filtering of the composition may be carried out by membrane filter filtration.
- a membrane filter for example a commercially available membrane filter
- a sterilized container, sterilized injector, syringe barrel, or the like can be used, optionally with a sterilized container, sterilized injector, syringe barrel, or the like, as appropriate.
- a membrane filter with a pore size of 0.05 ⁇ m to 20 ⁇ m can be used, for example 0.4 to 8.0 ⁇ m (for example, 0.4 ⁇ m, 0.5 ⁇ m, 1 ⁇ m, 2 ⁇ m, 3 ⁇ m, 4 ⁇ m, 5 ⁇ m, 6 ⁇ m, 7 ⁇ m or 8 ⁇ m), 0.5 to 6 ⁇ m or 1 to 5 ⁇ m, and especially 5 ⁇ m.
- the invention also provides an aqueous liquid composition of the invention for use as a medicament.
- a further aspect of the invention comprises the use of the aqueous liquid composition of the invention in human or veterinary medicine.
- the invention also provides a method of treating or preventing a disease or disorder in a subject comprising administration of a therapeutically effective amount of an aqueous liquid composition of the invention.
- the composition is administered by injection.
- the composition is administered by intra-articular injection.
- the disease or disorder is a joint disease, for example osteoarthritis (for example osteoarthritis of the knee).
- the disease or disorder is cataracts.
- the disease or disorder is a cancer.
- the invention also provides the use of the aqueous liquid composition for the manufacture of a medicament for use in human or veterinary medicine.
- the medicament is for use in the treatment of a joint disease, such as osteoarthritis (for example osteoarthritis of the knee).
- the medicament is for use in cataract surgery.
- the medicament is for use in cancer therapy.
- the pharmaceutically active compound is diclofenac and the aqueous liquid composition finds particular use in the treatment of osteoarthritis and other conditions of the joints.
- a composition of the invention may be made up into an injectable formulation and administered into a joint (for example the knee) by injection.
- the patient may be a human patient.
- compositions of the invention also find use in veterinary medicine, for example in the treatment of horses, such the treatment of osteoarthritis in horses (for example osteoarthritis of the knee).
- the invention also provides a kit comprising a conjugate of hyaluronan and a pharmaceutically active compound, and an aqueous sugar or sugar alcohol solution (preferably a sugar solution) having a concentration of 10–100 mg/mL, wherein the ratio of the mass of conjugate to sugar or sugar alcohol is 1:50 to 5:1.
- the kit may optionally contain NaCl or another salt (for example NaCl, KCl, CaCl2, NaBr, MgCl2, Choline chloride, NaHCO2, NaHPO4, or KH2PO4 , or for example NaCl, KCl, CaCl2, NaHCO2, NaHPO4, KH2PO4,), and preferably an aqueous solution of NaCl.
- the kit comprises an aqueous sugar or sugar alcohol solution having a concentration of 10–100 mg/mL.
- the kit comprises an aqueous sugar or sugar alcohol solution having a concentration of 20–60 mg/mL.
- the kit comprises an aqueous sugar or sugar alcohol solution having a concentration of 20–50 mg/mL.
- the ratio of the mass of conjugate to sugar or sugar alcohol in the kit is 1:12 to 5:2.
- the ratio of the mass of conjugate to sugar or sugar alcohol in the kit is 6:25 to 3:2.
- the ratio of the mass of conjugate to sugar or sugar alcohol in the kit is 1:4 to 1:1.
- the ratio of the mass of conjugate to sugar or sugar alcohol in the kit is 1:3 to 1:1.5.
- the ratio of the mass of conjugate to sugar or sugar alcohol in the kit is 1:2.
- the conjugate of the kit is sterile (i.e. the conjugate of the kit has been sterilised).
- the conjugate of the kit is sterile and/or has been sterilised by ionising radiation, for example wherein the ionising radiation is beta, gamma or X-ray radiation, and preferably gamma radiation, for example sterilised according to the following method: providing the conjugate of hyaluronan (for example sodium hyaluronate or hyaluronic acid) and a pharmaceutically active compound; and exposing the conjugate to ionising radiation, for example wherein the ionising radiation is beta, gamma or X-ray radiation, and preferably gamma radiation.
- ionising radiation for example sodium hyaluronate or hyaluronic acid
- the conjugate is exposed to the ionising radiation under an inert atmosphere (for example, wherein the inert atmosphere is an argon atmosphere or a nitrogen atmosphere).
- the dose of ionising radiation is around 5-40 kGy, preferably around 8-40 kGy or around 20-35 kGy, for example around 25 kGy or 32 around kGy. In one embodiment, the dose of ionising radiation is around 25 kGy and is verified by the VDmax25 method in ISO 11137-2:2013).
- the conjugate of the kit is sterile and/or has been obtained by (or is obtainable by) the following method: providing a conjugate of hyaluronan (for example sodium hyaluronate or hyaluronic acid) and a pharmaceutically active compound; and exposing the conjugate to ionising radiation, for example wherein the ionising radiation is beta, gamma or X-ray radiation, and preferably gamma radiation.
- the conjugate is exposed to the ionising radiation under an inert atmosphere (for example, wherein the inert atmosphere is an argon atmosphere or a nitrogen atmosphere).
- the dose of ionising radiation is around 5-40 kGy, preferably around 8-40 kGy or around 20-35 kGy, for example around 25 kGy or around 32 kGy. In one embodiment, the dose of ionising radiation is around 25 kGy and is verified by the VDmax25 method in ISO 11137-2:2013).
- the conjugate of the kit is sterile and is characterised by a sterility assurance level of 10 -3 or better, 10 -4 or better, 10 -5 or better, 10 -6 or better; preferably 10 -6 or better.
- the conjugate of the kit is sterile and/or has been subjected to a treatment such as dust removal, bacteria removal, and/or sterilization by, for example, filter filtration (for example membrane filter filtration).
- a treatment such as dust removal, bacteria removal, and/or sterilization by, for example, filter filtration (for example membrane filter filtration).
- filter filtration for example membrane filter filtration
- Such treatments including methods of sterilisation by filter filtration, are described in WO2020/101013 A1.
- the conjugate of the kit is sterile and/or has been subjected to a dust removal and sterilization step by filtration (for example membrane filter filtration).
- a membrane filter for example a commercially available membrane filter
- a sterilized container, sterilized injector, syringe barrel, or the like as appropriate.
- a membrane filter with a pore size of 0.05 ⁇ m to 1.0 ⁇ m can be used, for example 0.1 to 1.0 ⁇ m, and especially 0.22 ⁇ m.
- the aqueous sugar or sugar alcohol solution of the kit is in the form of a sterile solution, i.e. the aqueous sugar or sugar alcohol solution is a sterile aqueous sugar or sugar alcohol solution.
- the aqueous sugar or sugar alcohol solution of the kit is a sterile glucose solution (e.g. glucose for injection).
- the aqueous sugar or sugar alcohol solution of the kit is sterile and is characterised by a sterility assurance level of 10 -3 or better, 10 -4 or better, 10 -5 or better, 10 -6 or better; preferably 10 -6 or better.
- the kit may further comprise saline (NaCl) solution (for example sterile saline (NaCl) solution e.g. saline (NaCl) solution for injection) or water (for example sterile water, e.g. water for injection).
- the invention also provides a kit comprising a conjugate of hyaluronan and a pharmaceutically active compound, and a sugar or sugar alcohol (preferably a sugar), wherein the ratio of the mass of conjugate to sugar or sugar alcohol is 1:50 to 5:1.
- the ratio of the mass of conjugate to sugar or sugar alcohol in the kit is 1:12 to 5:2.
- the ratio of the mass of conjugate to sugar or sugar alcohol in the kit is 6:25 to 3:2.
- the ratio of the mass of conjugate to sugar or sugar alcohol in the kit is 1:4 to 1:1.
- the ratio of the mass of conjugate to sugar or sugar alcohol in the kit is 1:3 to 1:1.5.
- the ratio of the mass of conjugate to sugar or sugar alcohol in the kit is 1:2.
- the kit may optionally contain NaCl or another salt (for example NaCl, KCl, CaCl2, NaBr, MgCl2, Choline chloride, NaHCO2, NaHPO4, or KH2PO4 , or for example NaCl, KCl, CaCl 2 , NaHCO 2 , NaHPO 4 , KH 2 PO 4 ,), and preferably an aqueous solution of NaCl.
- the kit comprises a conjugate of hyaluronan and a pharmaceutically active compound in solid form, for example a powder, and a sugar or sugar alcohol in solid form, for example a powder, wherein the ratio of the mass of conjugate to sugar or sugar alcohol is 1:50 to 5:1. In an embodiment, the ratio of the mass of conjugate to sugar or sugar alcohol in the kit is 1:12 to 5:2. In a preferred embodiment, the ratio of the mass of conjugate to sugar or sugar alcohol in the kit is 6:25 to 3:2. In a more preferred embodiment, the ratio of the mass of conjugate to sugar or sugar alcohol in the kit is 1:4 to 1:1.
- the ratio of the mass of conjugate to sugar or sugar alcohol in the kit is 1:3 to 1:1.5. In an especially preferred embodiment, the ratio of the mass of conjugate to sugar or sugar alcohol in the kit is 1:2. In an embodiment, the conjugate of the kit is sterile (i.e. the conjugate of the kit has been sterilised).
- the conjugate of the kit is sterile and/or has been sterilised by ionising radiation, for example wherein the ionising radiation is beta, gamma or X-ray radiation, and preferably gamma radiation, for example sterilised according to the following method: providing the conjugate of hyaluronan (for example sodium hyaluronate or hyaluronic acid) and a pharmaceutically active compound; and exposing the conjugate to ionising radiation, for example wherein the ionising radiation is beta, gamma or X-ray radiation, and preferably gamma radiation.
- ionising radiation is beta, gamma or X-ray radiation, and preferably gamma radiation
- the conjugate is exposed to the ionising radiation under an inert atmosphere (for example, wherein the inert atmosphere is an argon atmosphere or a nitrogen atmosphere).
- the dose of ionising radiation is around 5-40 kGy, preferably around 8-40 kGy or around 20-35 kGy, for example around 25 kGy or around 32 kGy. In one embodiment, the dose of ionising radiation is around 25 kGy and is verified by the VDmax25 method in ISO 11137-2:2013).
- the conjugate of the kit is sterile and/or has been obtained by (or is obtainable by) the following method: providing a conjugate of hyaluronan (for example sodium hyaluronate or hyaluronic acid) and a pharmaceutically active compound; and exposing the conjugate to ionising radiation, for example wherein the ionising radiation is beta, gamma or X-ray radiation, and preferably gamma radiation.
- the conjugate is exposed to the ionising radiation under an inert atmosphere (for example, wherein the inert atmosphere is an argon atmosphere or a nitrogen atmosphere).
- the dose of ionising radiation is around 5-40 kGy, preferably around 8-40 kGy or around 20-35 kGy, for example around 25 kGy or around 32 kGy. In one embodiment, the dose of ionising radiation is around 25 kGy and is verified by the VDmax25 method in ISO 11137-2:2013).
- the conjugate of the kit is sterile and/or has been subjected to a treatment such as dust removal, bacteria removal, and/or sterilization by, for example, filter filtration (for example membrane filter filtration).
- filter filtration for example membrane filter filtration
- the conjugate of the kit is sterile and/or has been subjected to a dust removal and sterilization step by filtration (for example membrane filter filtration).
- a membrane filter for example a commercially available membrane filter
- a membrane filter can be used, optionally with a sterilized container, sterilized injector, syringe barrel, or the like, as appropriate.
- a membrane filter with a pore size of 0.05 ⁇ m to 1.0 ⁇ m can be used, for example 0.1 to 1.0 ⁇ m, and especially 0.22 ⁇ m.
- the conjugate of the kit is sterile and is characterised by a sterility assurance level of 10 -3 or better, 10 -4 or better, 10 -5 or better, 10 -6 or better; preferably 10 -6 or better.
- the sugar or sugar alcohol of the kit is in the form of a sterile composition.
- the sugar or sugar alcohol solution of the kit is a sterile glucose composition (e.g. glucose for injection).
- the sugar or sugar alcohol of the kit is in the form of a sterile composition and is characterised by a sterility assurance level of 10 -3 or better, 10 -4 or better, 10 -5 or better, 10 -6 or better; preferably 10 -6 or better.
- the kit may further comprise saline (NaCl) solution (for example sterile saline (NaCl) solution, e.g. saline (NaCl) solution for injection) or water (for example sterile water, e.g. water for injection).
- saline NaCl
- NaCl sterile saline
- water for example sterile water, e.g. water for injection
- Step 1 Synthesis of [2-(2,6-dichloro-phenylamino)-phenyl]-acetic acid 2-(2-tert- butoxycarbonylamino-ethoxy)-ethyl ester
- Diclofenac (50.0 g, 0.169 mol, 1.0 equiv.) and 2-[2-(BOC-amino)ethoxy]ethanol (69.5 g, 0.339 mol, 2.0 equiv.) were mixed in DCM (331 g) and the suspension was cooled to 1 o C.4- Dimethylaminopyridine (DMAP) (3.0 g, 0.025 mol, 0.15 equiv.) was added and the mixture was stirred at 1 o C for 10-20 min.
- DMAP Dimethylaminopyridine
- Ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC HCl) (40.5 g, 0.211 mol, 1.25 equiv.) was added over 5 h at 1 o C. The mixture was stirred for an additional 4 h at 1 o C before being warmed to 20 o C and stirred for 12 h. The mixture was quenched with water and the two phases were separated. The organic phase was washed twice with water and concentrated to dryness under vacuum. The residue was purified by column chromatography on silica gel (1.5 kg). Yield 72 g (88%) as a light yellow oil which solidified at ambient temperature.
- Step 2 Synthesis of 2-(2,6-dichloro-phenylamino)-phenyl]-acetic acid 2-(2-amino-ethoxy)- ethyl ester HCl salt (Compound 2) [2-(2,6-Dichloro-phenylamino)-phenyl]-acetic acid 2-(2-tert-butoxycarbonylamino-ethoxy)- ethyl ester (Compound 1, 65 g, 0.134 mol, 1.0 equiv.) from Step 1 was dissolved in DCM (665 g).
- Step 3 Synthesis of Hyaluronan-succinyl-ester (HSE) Sodium hyaluronate (NaHA) used in the synthesis was produced by bacterial fermentation (Streptococci) and had an intrinsic viscosity (I.V.) at 25 o C of 1.54 m 3 /kg.
- HSE Hyaluronan-succinyl-ester
- NaHA Sodium hyaluronate
- the weight-average molar mass (Mw) measured by AF4 ) was 667 kDa (the weight-average molar mass (Mw) of the sodium hyaluronate was measured using asymmetrical flow field-flow fractionation (AF4) combined with UV-FL-MALS-RI detectors and using the light scattering and concentration data, utilizing a first order fit to the scattering detectors 8 – 15 according to the Berry method, and a refractive index increment, dn/dc, of 0.167 mL/g).
- the sodium hyaluronate (200 g, 0.50 mol, 1.0 equiv.) was stirred in formamide (22.6 kg).
- the weight-average molar mass (M w ) measured by AF4 of the HSE was 579 kDa (the weight-average molar mass (M w ) of the HSE was measured using asymmetrical flow field-flow fractionation (AF4) combined with UV-FL-MALS-RI detectors and using the light scattering and concentration data, utilizing a first order fit to the scattering detectors 8 – 15 according to the Berry method, and a refractive index increment, dn/dc, of 0.167 mL/g).
- AF4 asymmetrical flow field-flow fractionation
- Step 4 Synthesis of Conjugate 1 (HSE-diclofenac) Hyaluronan-succinyl-ester (HSE) from Step 3, (220 g, 0.34 mol, 1.0 equiv.) was stirred in purified water (5.5 kg). Dimethyformamide (DMF) (15.6 kg) was added and the solution was stirred.
- Conjugate 1 HSE-diclofenac
- HSE Hyaluronan-succinyl-ester
- N-methylmorpholine (17.7 g) was added, followed by a solution of 2-(2,6-dichloro- phenylamino)-phenyl]-acetic acid 2-(2-amino-ethoxy)-ethyl ester HCl salt (Compound , 38.8 g, 0.085mol, 0.25 equiv.) in DMF (520 g).
- Hydroxybenzotriazole hydrate (HOBT) (1.16 g, 0.009 mol, 0.025 equiv.) was dissolved in DMF (160 g) and added to the reaction mixture.
- EDC HCl (16.2 g, 0.085 mol, 0.25 equiv.) was dissolved in DMF (260 g) and purified water (275 g) and added to the reaction. The mixture was stirred for 16 h. The reaction was quenched by adding a 25% NaCl aqueous solution (0.68kg). The crude product was precipitated by addition of ethanol. The solid was stirred in purified water (22 kg) for 16 h. The pH was adjusted to 5.5–6.0 by addition of 0.1 M NaOH.25% NaCl aqueous solution (0.6 kg) was added and the crude product was precipitated by addition of ethanol. The product was separated from the liquid and the solid was stirred in purified water (22 kg).
- the viscous solution was diluted with purified water (17.0 kg) and filtered through a filter cloth for clarity, and the filter was washed with purified water (5.0 kg).25% NaCl aqueous solution (1.2 kg) was added to the filtrate and the product was precipitated by addition of ethanol.
- the solid product was isolated by filtration and washed with ethanol and acetone. The wet cake was dried under vacuum at 33-37 o C. Yield 225 g (90%) as a white solid.6.3% w/w of diclofenac content based on an analytical UV-spectrophotometric method using a calibration curve from a diclofenac stock solution.
- the weight-average molar mass (M w ) measured by AF4 of Conjugate 1 was 611 kDa (the weight-average molar mass (M w ) of Conjugate 1 was measured using asymmetrical flow field-flow fractionation (AF4) combined with UV-FL-MALS-RI detectors and using the light scattering and concentration data, utilizing a first order fit to the scattering detectors 8 – 15 according to the Berry method, and a refractive index increment, dn/dc, of 0.167 mL/g).
- AF4 asymmetrical flow field-flow fractionation
- Example 2 Preparation and visual comparison of aqueous liquid compositions of hyaluronic acid–diclofenac conjugate and sodium hyaluronate Solutions (aqueous, 1 mL) of Conjugate 1 (5 mg/mL) and sodium hyaluronate (the sodium hyaluronate used in step 3 of Example 1) (5 mg/mL) were prepared with different co- solutes, and the viscosity and appearance were inspected visually by turning the vial upside down. The following solutions were prepared. Table 1 The solutions were put on a shake board for approximately 90 minutes and the solutions were visually inspected.
- Solutions 11, 12, 13, 14, 15 and 16 had more or less the same viscosity.
- the results of Example 2 show that aqueous liquid compositions of Conjugate 1 in a 1% NaCl or 1% KCl solution have a higher viscosity compared to sodium hyaluronate without conjugated drug under the same conditions. This effect does not appear to be because of the presence of chlorine ions.
- Conjugate 1 dissolved in a 1% choline chloride solution showed only marginally increased viscosity compared with dissolved Conjugate 1 in water without a co-solute (solution 15).
- Example 3 Preparation and visual comparison of clinically relevant aqueous liquid compositions of hyaluronic acid–diclofenac conjugate and compositions of sodium hyaluronate
- higher concentrations of Conjugate 1 and unconjugated sodium hyaluronate compositions were prepared.
- Stock solutions of Conjugate 1 and sodium hyaluronate at 15 mg/mL were prepared, and 0.16 mmol of the salt or glucose was added as a solid to the aqueous solution.
- Table 2 The mixtures were shaken on a shake board for approximately 2 hours and allowed to settle overnight. The solutions were then visually inspected.
- Solution 23 with Conjugate 1 was less viscous than solution 24 containing sodium hyaluronate.
- the results of Example 3 show that aqueous liquid compositions of Conjugate 1 in the presence of NaCl, KCl or NaBr (0.16 mmol) show significantly higher viscosity compared to the compositions of Conjugate 1 in a 5% glucose solution. Solutions 17 (NaCl), 19 (KCl) and 21 (NaBr) are more like gels at these concentrations, as shown in Figure 1.
- the inventors have found that preparing isotonic solutions of Conjugate 1 in 5% glucose solutions provides lower viscosity of the solution compared to the solutions in 0.9% saline.
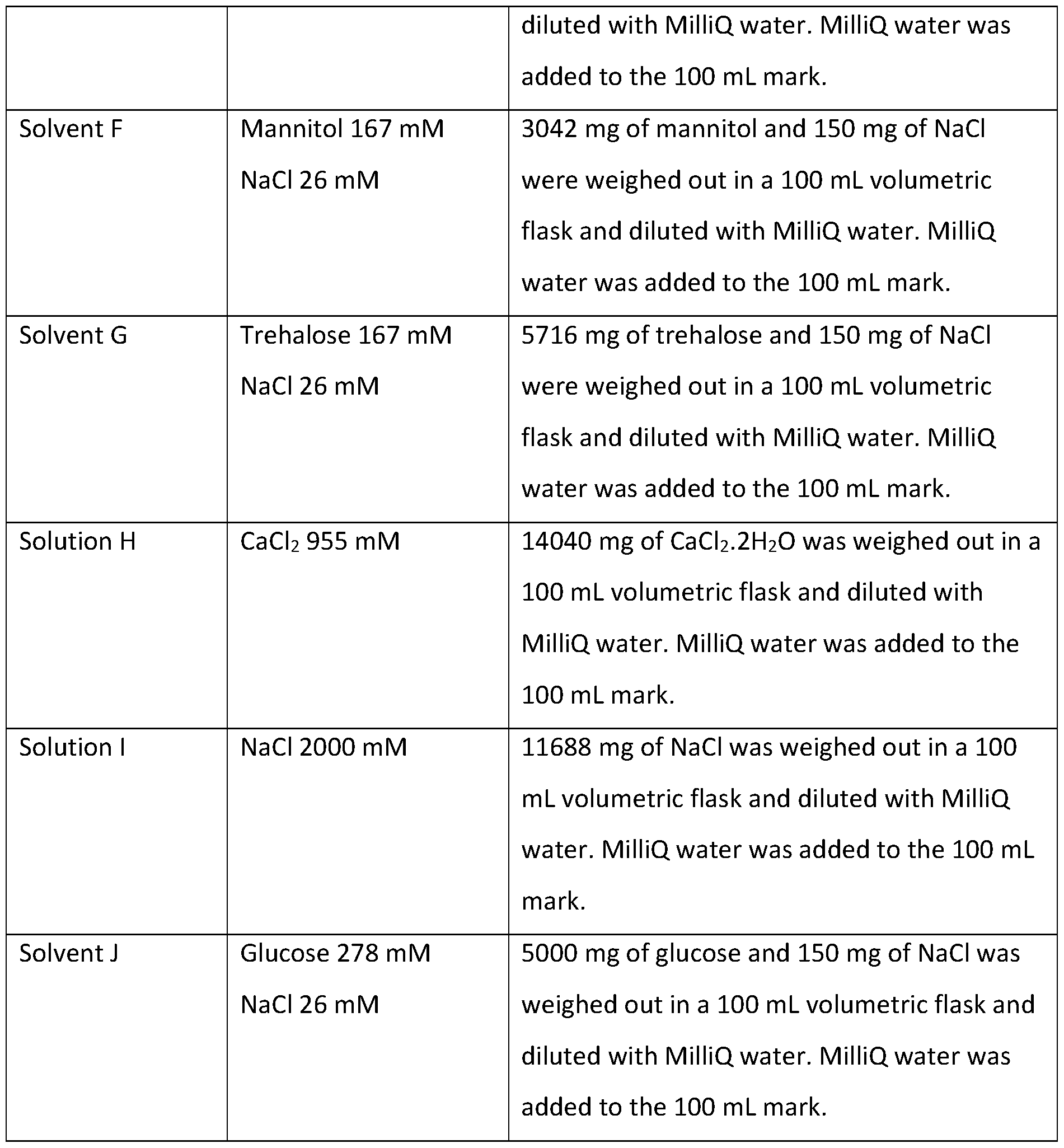
- Example 4 Solvents and solutions used for sample preparation The solvents and solutions shown in Table 3 were prepared for use in Examples 5 to 8. Table 3 Additional experiments were performed in order to further study the viscosity behaviour of the Conjugate 1 in solution with different additives. Table 4 provides a summary of the solutions preparing including Conjugate 1 in Experiments 5 to 8 (in Example 5, solutions comprising sodium hyaluronate were also prepared).
- Example 5 Determination of the viscosity of aqueous liquid compositions of Conjugate 1 or sodium hyaluronate with glucose, sodium chloride, glucose and sodium chloride, or water
- Four solutions of Conjugate 1, and four solutions of sodium hyaluronate (the sodium hyaluronate used was the starting material in step 3 of Example 1), with 3% glucose (167 mM) + 0.15% NaCl (26 mM), 5% glucose (278 mM), 0.9% NaCl (156 mM), or MilliQ water respectively, as shown in Table 5, were prepared according to the below procedure.
- FIG 4 viscosity measurements of aqueous liquid compositions comprising, in order of traces from top to bottom: 15 mg/mL Conjugate 1 and 0.9% NaCl (solution 29); 15 mg/mL Conjugate 1 and 3% glucose + 0.15% NaCl (solution 27); 15 mg/mL Conjugate 1 and 5% glucose (solution 28); 15 mg/mL Conjugate 1 and MilliQ water (solution 30).
- FIG 5 viscosity measurements of aqueous liquid compositions comprising, in order of traces from top to bottom: 15 mg/mL sodium hyaluronate and 5% glucose (solution 32); 15 mg/mL sodium hyaluronate and 3% glucose + 0.15% NaCl (solution 31); 15 mg/mL sodium hyaluronate and MilliQ water (solution 34); 15 mg/mL sodium hyaluronate and 0.9% NaCl (solution 33).
- Example 6 Determination of the viscosity of aqueous liquid compositions of Conjugate 1 solutions containing different sugars or sugar alcohols (167 mM) and sodium chloride (26 mM)
- 167 mM glucose
- 167 mM sucrose
- 167 mM sucrose
- 167 mM mannitol
- 0.15% NaCl 26 mM
- trehalose 167 mM + 0.15% NaCl (26 mM)
- Solutions 35 to 38 all exhibit shear thinning behavior, where the samples become less viscous as a function of shear rate. No notable difference in behavior over the measured shear rate was observed between the samples containing different sugars (35, 36 and 38) or sugar alcohol (37).
- the solution containing mannitol (Solution 37) exhibits the highest zero-shear viscosity (49075 mPa ⁇ s) and the solution containing glucose (Solution 35) has the lowest zero-shear viscosity (39880 mPa ⁇ s).
- Example 7 Determination of the viscosity of aqueous liquid compositions of Conjugate 1 containing different sugars or sugar alcohols (167 mM), sodium chloride (26 mM) and calcium chloride (12 mM)
- 167 mM glucose
- 26 mM sodium chloride
- calcium chloride 12 mM
- Shear banding happens when different kinds of laminar flow occur in the sample, thus changing the flow behavior. This can happen in aqueous “semi-diluted” or highly concentrated solutions where the compounds in the sample start interacting with each other, prohibiting normal laminar flow (see The Handbook of Rheology, 5th edition (2020), Thomas G. Mezger). Shear banding makes it impossible to determine the zero-shear viscosity. Comparison of Figure 10 (showing the graphed results for Solutions 39, 40, 41 and 42) and Figure 7 (showing the graphed results for Solutions 35, 36, 37 and 38) shows that addition of calcium chloride increases the initial viscosity (and most likely also the zero-shear viscosity, it had been possible to determine that).
- Example 8 Determination of the viscosity of aqueous liquid compositions of Conjugate 1 containing different concentrations of glucose and/or sodium chloride (167 mM and 52 mM, 278 mM and 26 mM and, 278 mM and 52 mM) Three solutions of Conjugate 1 with 3% glucose (167 mM) + 0.3% NaCl (52 mM), 5% glucose (278 mM) + 0.15% NaCl (26 mM), or 5% glucose (278 mM) + 0.3% NaCl (52 mM), respectively, as shown in Table 8, were prepared according to the below procedure.
- Sample 45 Approximately 60 mg of Conjugate 1 was weighed in a glass vial.3.95 mL of solvent component J (see Table 3 and Table 8) was added.0.051 mL (51 ⁇ L) of NaCl 2000 mM (solution I of Table 3) was added to a final concentration of NaCl 52 mM. The mixture was shaken vigorously for 15–30 seconds. The mixture was then shaken using a shake board for 24 hours (100–200 rpm). The mixture was then inverted 3–4 times, and then refrigerated for 24 hours. When mixture reached room temperature, it was inverted a couple of times and inspected. If the sample did not appear homogeneous, shaking and refrigeration steps were repeated until homogeneity was achieved.
- Solutions 44 and 45 contain a higher concentration of glucose compared to Solution 35 and 43, respectively. Comparing Solution 44 and 35, Solution 44 has a slightly higher viscosity over the entire measured shear rate range (see Figures 12 and 13) and an increased zero- shear viscosity from 47636 mPa ⁇ s compared to 39880 mPa ⁇ s for Solution 35 (see Figure 11 and Figure 6). A similar behavior is observed when comparing Solution 45 and 43 (which have a higher concentration of NaCl compared to Solutions 44 and 35). Solution 45 has a slightly higher zero-shear viscosity of 1.44 ⁇ 10 5 mPa ⁇ s compared to 1.29 ⁇ 10 5 mPa ⁇ s for Solution 43.
- Solutions 43 and 45 Comparing the graphs and zero-shear viscosity of Solutions 35, 43, 44 and 45, it can be observed that the raise of viscosity is more accentuated by increasing the concentration of NaCl than by increasing the concentration of glucose: Solutions 43 and 45 has a significantly higher viscosity than Solutions 44 and 35 (for example, the zero-shear viscosity of Solutions 43 and 45 are more than three times that of Solutions 44 and 35, respectively, as shown in Figure 11 and Figure 6).
- Example 9 Sterilisation, formulation and aseptic filling of Conjugate 1 and analysis of the formulation
- Example 9a Sterilisation of Conjugate 1 by irradiation (25 kGy) under argon Samples of Conjugate 1 prepared as described in Example 1 were sterilised by gamma ray irradiation as follows: Step 1: Conjugate 1 was packaged (as a powder) in sample sizes of 20.0 g ⁇ 0.1 g, 1.0 g ⁇ 0.1 g and 2 x 130 mg ⁇ 5 mg, each in three PE bags and an aluminum bag.
- each sample of Conjugate 1 was packaged in a primary polyethylene (PE) bag; the air was squeezed out of the primary PE bag and replaced with argon; the argon was squeezed out and the primary PE bag was closed with a zip-tie; the primary PE bag was placed in a secondary PE bag; the air was squeezed out of the secondary PE bag and replaced with argon; the argon was squeezed out and the secondary PE bag was closed by thermo-sealing (also referred to as heat sealing); the secondary PE bag was placed in a tertiary PE bag; the air was squeezed out of the tertiary PE bag and replaced with argon; the argon was squeezed out and the tertiary PE bag was closed by thermo-sealing; the tertiary PE bag was placed in an aluminium bag; the air was squeezed out of the aluminium bag and replaced with argon; and the argon was squeezed out and the aluminium bag was closed by thermo- sealing and labelled.
- PE polyethylene
- the four bags (20.0 g ⁇ 0.1 g, 1.0 g ⁇ 0.1 g and 2 x 130 mg ⁇ 5 mg) were then packaged in a carton box with dimensions 32.0 x 21.0 x 21.0 cm.
- the gross weight of the box was 0.5 kg, with an apparent density of 0.035 g/cm 3 .
- This process was repeated until five carton boxes each containing 4 bags of the packaged samples (20.0 g ⁇ 0.1 g, 1.0 g ⁇ 0.1 g and 2 x 130 mg ⁇ 5 mg) of Conjugate 1 were made. It is noted that the orientation of the 4 bags of the packaged samples within the box was not noted as it was not crucial to the method.
- the same orientation of the 4 bags of the packaged samples in each box was kept as similar as possible. (see ref. ISO 11137-39.2.1.3: Low-density products tend to be fairly homogeneous such that the orientation of individual products within the irradiation container is unlikely to have a significant effect on dose distribution when irradiating with gamma rays).
- the one box including the Conjugate 1 samples was put inside a larger box which contained dry ice. The measurements of the larger box was 46.0 x 46.0 x 57.0 cm and the total weight was 12.6 kg, with an apparent density of 0.104 g/cm 3 .
- Step 2 The irradiation position used gamma irradiations emitted by Cobalt60 ( 60 Co). A dose of 25 kGy was used. Cobalt60 was contained in stainless steel cylinders (“pencils”) placed on a rack and positioned into an irradiation bunker stored in a pool 6 meters deep. The irradiation plant (Gammatom Srl, Italy) uses a batch mode that uses totes. The pencils distribution into the source rack, as well as the exposure time based on the requested dose and the product density was managed by validated software. Step 3: Validation of the sterilization method was carried out according to ISO 11137- 2:2013, VDmax25 (single batch validation).
- a routine sterilisation dose of 25 kGy was shown to achieve a Sterility Assurance Level (SAL) of 10 -6 for Conjugate 1.
- SAL Sterility Assurance Level
- Example 9b - Formulation and aseptic filling of Conjugate 1 after irradiation A formulation solvent was prepared in a 2L polyethylene terephthalate glycol (PETG) media bottle by mixing 250 mL of 0.9% sterile sodium chloride solution with 350 mL of water for injection (WFI) to obtain 0.375% saline solution. The resulting 0.375% saline solution (600 mL) was then mixed with 900 mL of glucose 5% sterile solution to obtain a final 3% glucose, 0.15% sodium chloride mixture.
- PETG polyethylene terephthalate glycol
- WFI water for injection
- a solution of Conjugate 1 was prepared in a new 2L PETG media bottle by adding 19.6 g of sterile Conjugate 1 (irradiated as described in Example 9a, above: 25 kGy, 60 Co, under argon) in four portions to the 880 mL of the formulation solvent. The mixture was placed on a shaker and gently shaken for 2-3 hours at room temperature. The bottle was then transferred to a refrigerator and stored at 2-8 o C overnight. The next day, the mixture was shaken again for 1-3 hours at room temperature.
- Diclofenac content, degree of substitution, and impurities after formulation Methods Total content of diclofenac (mg/ml) by UV The concentration of diclofenac in a sample of Conjugate 1 formulated according to Example 9b and kept at 2-8 o C was measured to access the total amount of diclofenac in the sample at the following time points: at formulation and at 1 month, 3 months and 6 months after formulation. A spectrophotometric (UV) method was used and the absorbance in solution was measured to give a value of diclofenac concentration.
- UV spectrophotometric
- the quantitative determination of diclofenac was calculated with a six-point calibration using diclofenac as the reference, and the absorbance of the sample measured at 275 nm in duplicate.
- Content of free diclofenac and related impurities w/w% of diclofenac
- HPLC-UV A gradient reverse-phase HPLC method with UV detection was used to measure the content of free diclofenac and related impurities (w/w% of diclofenac) in a sample of Conjugate 1 formulated according to Example 9b and kept at 2-8 o C at the following time points: at formulation and at 1 month, 3 months and 6 months after formulation.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Epidemiology (AREA)
- Ophthalmology & Optometry (AREA)
- Physical Education & Sports Medicine (AREA)
- Rheumatology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Immunology (AREA)
- Surgery (AREA)
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Polysaccharides And Polysaccharide Derivatives (AREA)
Abstract
Description
Claims
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB2211970.5 | 2022-08-16 | ||
| GBGB2211970.5A GB202211970D0 (en) | 2022-08-16 | 2022-08-16 | Sterilisation method |
| GBGB2216467.7A GB202216467D0 (en) | 2022-11-04 | 2022-11-04 | Pharmaceutical formulation |
| GB2216467.7 | 2022-11-04 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024038088A1 true WO2024038088A1 (en) | 2024-02-22 |
Family
ID=87760278
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2023/072562 Ceased WO2024038088A1 (en) | 2022-08-16 | 2023-08-16 | Liquid formulations |
| PCT/EP2023/072561 Ceased WO2024038087A1 (en) | 2022-08-16 | 2023-08-16 | Method for preparing sterile compositions |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2023/072561 Ceased WO2024038087A1 (en) | 2022-08-16 | 2023-08-16 | Method for preparing sterile compositions |
Country Status (1)
| Country | Link |
|---|---|
| WO (2) | WO2024038088A1 (en) |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1996035720A1 (en) | 1995-05-10 | 1996-11-14 | Fidia Advanced Biopolymers S.R.L. | Heavy metal salts of succinic acid hemiesters with hyaluronic acid, or hyaluronic acid esters, a process for their preparation, and relative pharmaceutical compositions |
| WO2004035629A2 (en) * | 2002-10-18 | 2004-04-29 | Fidia Farmaceutici S.P.A. | Taxanes covalently bounded to hyaluronic acid or hyaluronic acid derivatives |
| WO2007126154A1 (en) | 2006-05-01 | 2007-11-08 | Seikagaku Corporation | Method of producing polysaccharide derivatives |
| WO2009130564A1 (en) * | 2008-04-22 | 2009-10-29 | Fidia Farmaceutici S.P.A. | Therapeutic use of new pharmaceutical preparations containing antitumoral drugs bound to hyaluronic acid in the treatment of neoplasias |
| WO2015128787A1 (en) | 2014-02-27 | 2015-09-03 | Pharmalink Ab | Hyaluronan conjugates with pharmaceutically active substances, methods and compositions |
| WO2017120193A1 (en) * | 2016-01-04 | 2017-07-13 | University Of Kansas | Drug delivery compositions and methods |
| WO2020101013A1 (en) | 2018-11-16 | 2020-05-22 | 生化学工業株式会社 | Pharmaceutical composition |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SE442820B (en) | 1984-06-08 | 1986-02-03 | Pharmacia Ab | GEL OF THE CROSS-BOND HYALURONIC ACID FOR USE AS A GLASS BODY SUBSTITUTE |
| US5621093A (en) | 1995-06-06 | 1997-04-15 | Anika Research, Inc. | Steam-sterilizing solid hyaluronic acid |
| US6383344B1 (en) | 2000-07-19 | 2002-05-07 | Genzyme Corporation | Molecular weight reduction of polymer using irradiation treatment |
| WO2009005790A2 (en) | 2007-06-29 | 2009-01-08 | Carbylan Biosurgery, Inc. | Sterile thiol-derivatized hyaluronic acid polymer compositions and uses thereof |
| IT201600117042A1 (en) * | 2016-11-18 | 2018-05-18 | Fidia Farm Spa | HYALURONIC ACIDS SULFATE FUNCTIONALIZED WITH DOPAMINE |
| IT202000008209A1 (en) * | 2020-04-17 | 2021-10-17 | Fidia Farm Spa | Process of synthesis of a hyaluronic acid-paclitaxel conjugate |
| AU2021269007A1 (en) * | 2020-05-04 | 2022-11-24 | Ascendis Pharma A/S | Hydrogel irradiation |
-
2023
- 2023-08-16 WO PCT/EP2023/072562 patent/WO2024038088A1/en not_active Ceased
- 2023-08-16 WO PCT/EP2023/072561 patent/WO2024038087A1/en not_active Ceased
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1996035720A1 (en) | 1995-05-10 | 1996-11-14 | Fidia Advanced Biopolymers S.R.L. | Heavy metal salts of succinic acid hemiesters with hyaluronic acid, or hyaluronic acid esters, a process for their preparation, and relative pharmaceutical compositions |
| WO2004035629A2 (en) * | 2002-10-18 | 2004-04-29 | Fidia Farmaceutici S.P.A. | Taxanes covalently bounded to hyaluronic acid or hyaluronic acid derivatives |
| WO2007126154A1 (en) | 2006-05-01 | 2007-11-08 | Seikagaku Corporation | Method of producing polysaccharide derivatives |
| WO2009130564A1 (en) * | 2008-04-22 | 2009-10-29 | Fidia Farmaceutici S.P.A. | Therapeutic use of new pharmaceutical preparations containing antitumoral drugs bound to hyaluronic acid in the treatment of neoplasias |
| WO2015128787A1 (en) | 2014-02-27 | 2015-09-03 | Pharmalink Ab | Hyaluronan conjugates with pharmaceutically active substances, methods and compositions |
| WO2017120193A1 (en) * | 2016-01-04 | 2017-07-13 | University Of Kansas | Drug delivery compositions and methods |
| WO2020101013A1 (en) | 2018-11-16 | 2020-05-22 | 生化学工業株式会社 | Pharmaceutical composition |
Non-Patent Citations (11)
| Title |
|---|
| "Japanese Pharmacopoeia", vol. 4, article "Sterility Test monograph", pages: 06 |
| "OECD Guidelines for the Testing of Chemicals", OECD PUBLISHING, article "Viscosity of Liquids" |
| "Ph. Eur.", article "Sterility monograph" |
| "USP", vol. 71, article "Sterility Tests monograph" |
| ARRICH ET AL., CMAJ, vol. 172, no. 8, 2005, pages 1039 - 1043 |
| COWMANMENDICHI: "Chemistry and Biology of Hyaluronan", 2004, ELSEVIER SCIENCE LTD., article "Methods for Determination of Hyaluronan Molecular Weight", pages: 41 - 69 |
| DONG ET AL.: "Improved stability and tumor targeting of 5-fluorouracil by conjugation with hyaluronan", JOURNAL OF APPLIED POLYMER SCIENCE, vol. 130, no. 2, pages 927 - 932, XP055188245, DOI: 10.1002/app.39247 |
| JORGENSEN ET AL., ANNALS OF THE RHEUMATIC DISEASES, vol. 69, 2010, pages 1097 - 1102 |
| KWON ET AL.: "Depolymerization study of sodium hyaluronate by flow field-flow fractionation/multiangle light scattering", ANAL. BIOANAL. CHEM., vol. 395, 2009, pages 519 - 525, XP019736650, DOI: 10.1007/s00216-009-2952-0 |
| LOHMANDER ET AL., ANNALS OF THE RHEUMATIC DISEASES, vol. 55, 1996, pages 424 - 431 |
| THOMAS G. MEZGER: "The Handbook of Rheology", 2020 |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2024038087A1 (en) | 2024-02-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11090328B2 (en) | Compositions and methods for treating joints | |
| Kaderli et al. | A novel biocompatible hyaluronic acid–chitosan hybrid hydrogel for osteoarthrosis therapy | |
| US20230190786A1 (en) | Continuous release compositions made from hyaluronic acid, and therapeutic applications of same | |
| US11642357B2 (en) | Hyaluronan conjugates with pharmaceutically active substances, methods and compositions | |
| US10420795B2 (en) | Pharmaceutical composition for relieving pain | |
| US20170340774A1 (en) | Polethylene glycol hydrogel injection | |
| WO2024038088A1 (en) | Liquid formulations | |
| US11744899B2 (en) | Pharmaceutical composition containing a hyaluronic acid derivative | |
| US20090239822A1 (en) | Process for the preparation of esters of diacerein with hyaluronic acid and pharmaceutical compositions containing such esters | |
| CN118217229A (en) | A pharmaceutical composition containing meloxicam and its preparation method and application | |
| US20240216573A1 (en) | Compositions comprising cross-linked hyaluronic acid and methods of use thereof | |
| CN117440838A (en) | Compositions including cross-linked hyaluronic acid and methods of use |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 23758288 Country of ref document: EP Kind code of ref document: A1 |
|
| WPC | Withdrawal of priority claims after completion of the technical preparations for international publication |
Ref document number: 2216467.7 Country of ref document: GB Date of ref document: 20250211 Free format text: WITHDRAWN AFTER TECHNICAL PREPARATION FINISHED Ref document number: 2211970.5 Country of ref document: GB Date of ref document: 20250211 Free format text: WITHDRAWN AFTER TECHNICAL PREPARATION FINISHED |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 32PN | Ep: public notification in the ep bulletin as address of the adressee cannot be established |
Free format text: NOTING OF LOSS OF RIGHTS PURSUANT TO RULE 112(1) EPC (EPO FORM 1205A DATED 23/07/2025) |