WO2024036052A1 - Methods for preparing diphenylsilane bridged c1 symmetric catalysts and polymers made therefrom - Google Patents
Methods for preparing diphenylsilane bridged c1 symmetric catalysts and polymers made therefrom Download PDFInfo
- Publication number
- WO2024036052A1 WO2024036052A1 PCT/US2023/071323 US2023071323W WO2024036052A1 WO 2024036052 A1 WO2024036052 A1 WO 2024036052A1 US 2023071323 W US2023071323 W US 2023071323W WO 2024036052 A1 WO2024036052 A1 WO 2024036052A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- aryl
- substituted
- unsubstituted
- hydrocarbyl
- hydrogen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/60—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with refractory metals, iron group metals, platinum group metals, manganese, rhenium technetium or compounds thereof
- C08F4/62—Refractory metals or compounds thereof
- C08F4/64—Titanium, zirconium, hafnium or compounds thereof
- C08F4/659—Component covered by group C08F4/64 containing a transition metal-carbon bond
- C08F4/6592—Component covered by group C08F4/64 containing a transition metal-carbon bond containing at least one cyclopentadienyl ring, condensed or not, e.g. an indenyl or a fluorenyl ring
- C08F4/65922—Component covered by group C08F4/64 containing a transition metal-carbon bond containing at least one cyclopentadienyl ring, condensed or not, e.g. an indenyl or a fluorenyl ring containing at least two cyclopentadienyl rings, fused or not
- C08F4/65927—Component covered by group C08F4/64 containing a transition metal-carbon bond containing at least one cyclopentadienyl ring, condensed or not, e.g. an indenyl or a fluorenyl ring containing at least two cyclopentadienyl rings, fused or not two cyclopentadienyl rings being mutually bridged
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F17/00—Metallocenes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F10/00—Homopolymers and copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond
- C08F10/04—Monomers containing three or four carbon atoms
- C08F10/06—Propene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F110/00—Homopolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond
- C08F110/04—Monomers containing three or four carbon atoms
- C08F110/06—Propene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F210/00—Copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond
- C08F210/04—Monomers containing three or four carbon atoms
- C08F210/06—Propene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/60—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with refractory metals, iron group metals, platinum group metals, manganese, rhenium technetium or compounds thereof
- C08F4/62—Refractory metals or compounds thereof
- C08F4/64—Titanium, zirconium, hafnium or compounds thereof
- C08F4/659—Component covered by group C08F4/64 containing a transition metal-carbon bond
- C08F4/65912—Component covered by group C08F4/64 containing a transition metal-carbon bond in combination with an organoaluminium compound
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/60—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with refractory metals, iron group metals, platinum group metals, manganese, rhenium technetium or compounds thereof
- C08F4/62—Refractory metals or compounds thereof
- C08F4/64—Titanium, zirconium, hafnium or compounds thereof
- C08F4/659—Component covered by group C08F4/64 containing a transition metal-carbon bond
- C08F4/65916—Component covered by group C08F4/64 containing a transition metal-carbon bond supported on a carrier, e.g. silica, MgCl2, polymer
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F2420/00—Metallocene catalysts
- C08F2420/09—Cyclic bridge, i.e. Cp or analog where the bridging unit linking the two Cps or analogs is part of a cyclic group
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F2500/00—Characteristics or properties of obtained polyolefins; Use thereof
- C08F2500/12—Melt flow index or melt flow ratio
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F2500/00—Characteristics or properties of obtained polyolefins; Use thereof
- C08F2500/27—Amount of comonomer in wt% or mol%
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F2500/00—Characteristics or properties of obtained polyolefins; Use thereof
- C08F2500/34—Melting point [Tm]
Definitions
- Embodiments of the present invention generally relate to asymmetric metallocene catalyst compounds. More particularly, embodiments of the present invention relate to asymmetric, bridged metallocenes with substituted indacenyl ligands and propylene polymers made therefrom.
- a second nucleophile such as indenyl lithium or fluorenyl lithium
- references of interest include: CN 101235106B, which details synthesis of 1 -indenyl analogs of diphenylsilane; US 9,790,240 B2 describes a mixed C2 symmetric system of diphenylsilane derivatives; Izmer, V. et al. (2001 ) “ansa-Metallocenes with a Ph2Si Bridge: Molecular Structures of HfCl2[Ph2Si(n 5 -CsH4)] and HfCl2[Ph2Si(Ci3H9)(n 5 - CSH4)]2,” J. Chem. Soc., Dalton Trans., pp.
- EP 0754698A2 which describes Cp-fluorenyl analogs with diphenylsilane bridge
- US 2019/0161559A1 and W02018/151904A1 describe bis-Cp analogs with a diphenylsilane bridge
- US 2020/043758, WO2014/099303, US 9,266,91062; and KR 2020003044 describe bis-Cp analogs with a diphenylsilane bridge.
- Di-aryl silane bridged metallocene catalysts methods for making and propylene (co)polymers made therefrom are provided herein.
- the resulting silane bridged metallocene catalysts are very active in polymerization of olefins, especially ethylene and surprisingly propylene.
- a catalyst system comprising a metallocene compound described in the Formula (I): where M is a Group 4 metal; each of X1 and X2 is a univalent anionic ligand, orX1 and X2 are joined to form a metallocycle ring; R 1 is hydrogen, an unsubstituted C1-C40 hydrocarbyl, a C1-C40 substituted hydrocarbyl, an unsubstituted C4-C62 aryl, a substituted C4-C62 aryl, an unsubstituted C4-C62 heteroaryl, a substituted C4-C62 heteroaryl, -NR' 2 , -SR', -OR, -SiR' 3 , -OSiR' 3 , -PR' 2 , or-R"-SiR' 3 , where R" is C1-C10 alkyl and each R' is hydrogen, halogen, C1-C10 alkyl and each R' is
- J 1 and J 2 are independently hydrogen, an unsubstituted C1-C40 hydrocarbyl, a C1-C40 substituted hydrocarbyl, an unsubstituted C4-C62 aryl, a substituted C4-C62 aryl, an unsubstituted C4-C62 heteroaryl, a substituted C4-C62 heteroaryl, -NR' 2 , -SR', -OR, -SiR' 3 , -OSiR' 3 , -PR' 2 , or -R"-SiR' 3 , where R" is C1-C10 alkyl and each R' is hydrogen, halogen, C1-C10 alkyl, or C6-C10 aryl.
- J 1 and J 2 can be joined to form a 4, 5 or 6 membered ring.
- Each R 2 -R 6 can be hydrogen, an unsubstituted C1-C40 hydrocarbyl, a C1-C40 substituted hydrocarbyl, an unsubstituted C4-C62 aryl, a substituted C4-C62 aryl, an unsubstituted C4-C62 heteroaryl, a substituted C4-C62 heteroaryl, -NR'2, -SR', -OR, -SiR' 3 , -OSiR' 3 , -PR' 2 , or -R"-SiR' 3 , where R" is C1-C10 alkyl and each R' is hydrogen, halogen, C1-C10 alkyl, or Ce-C aryl.
- Each R 7 -R 16 can be hydrogen, an unsubstituted C1-C40 hydrocarbyl, a C1-C40 substituted hydrocarbyl, an unsubstituted C4-C62 aryl, a substituted C4-C62 aryl, an unsubstituted C4-C62 heteroaryl, a substituted C4-C62 heteroaryl, -NR' 2 , -SR', -OR, -SiR' 3 , -OSiR' 3 , -PR' 2 , or -R"-SiR' 3 , where R" is C1-C10 alkyl and each R' is hydrogen, halogen, C1-C10 alkyl, or C6-C10 aryl; optionally, R 11 and R 12 can be joined to form a silafluorene moiety.
- Each R 17 , R 18 , R 19 , and R 20 can be independently hydrogen, a halogen, an unsubstituted C1-C40 hydrocarbyl, a C1-C40 substituted hydrocarbyl, an unsubstituted C4-C62 aryl, a substituted C4-C62 aryl, an unsubstituted C4-C62 heteroaryl, a substituted C4-C62 heteroaryl, -NR' 2 , -SR', -OR, -SiR' 3 , -OSiR' 3 , -PR' 2 , or -R"-SiR' 3 , wherein R" is C1-C10 alkyl and each R' is hydrogen, halogen, C1-C10 alkyl, or C6-C10 aryl.
- Figure 1 shows the 1 H NMR spectrum of di-lithium salt prepared in a single step (“one pot”), according to one or more embodiments described herein.
- FIG. 2 shows the 1 H NMR spectrum of Catalyst I2 made in the experimental section below.
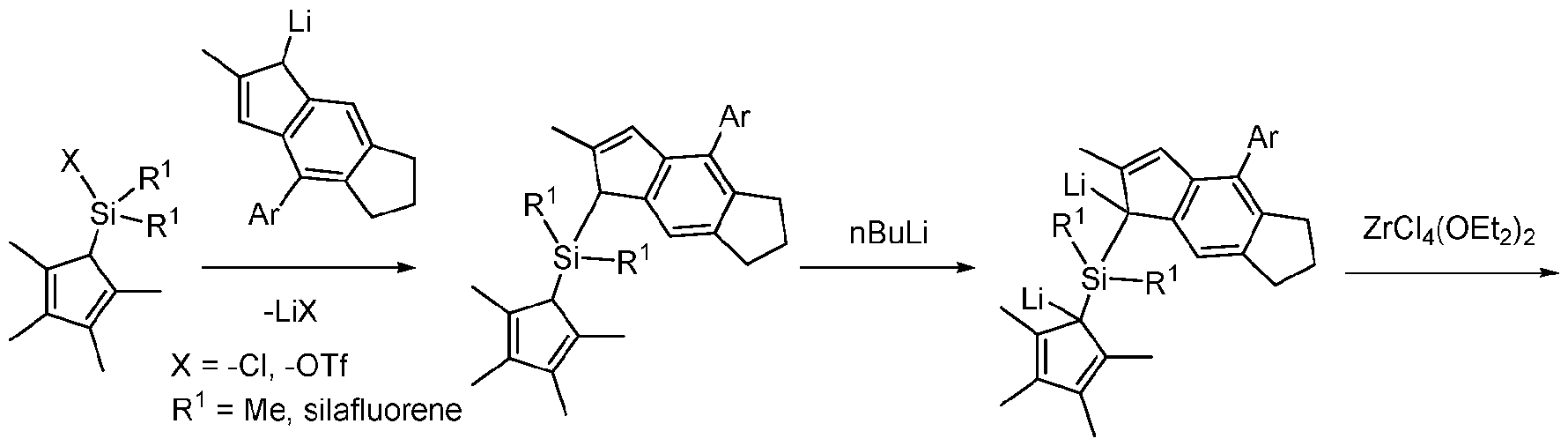
- a di-aryl silane bridged metallocene can be synthesized by substituting a dichloro cyclopentadienyl fragment with indenyl lithium.
- direct lithitation I substitution with aryl or alkyl lithium reagents provides a di-lithiated ligand that undergoes metalation with a Group IV metal precursor.
- the general synthesis method provided herein avoids and eliminates the electronic effects that promote deprotonation instead of nucleophilic substitution of the prior art.
- the general synthesis methods described herein also allow for significant diversification of aryl silane derivatives, and in particular di-aryl silane derivatives.
- the general synthesis for making the catalysts can also be applied towards mixed asymmetric di-aryl, or mono-aryl derivatives.
- the resulting silane bridged metallocene catalysts are very active in polymerization of olefins, especially ethylene and surprisingly propylene.
- diphenyl silane bridged metallocenes can be synthesized in just 3 steps. To do so, a dichloro cyclopentadienyl fragment first undergoes substitution with indenyl lithium. Next, direct lithitation I substitution with aryl or alkyl lithium reagents provides a di-lithiated ligand that undergoes transmetallation with at least one Group (IV) metal salt to provide a metallocene precursor containing a di-aryl bridge, as shown below:
- diphenylsilane bridge metallocenes provided herein can be supported or unsupported, and can be represented by the Formula (I):
- M is a Group 4 metal.
- Each of X1 and X2 can be a univalent anionic ligand, or X1 and X2 can be joined to form a metallocycle ring.
- R 1 can be hydrogen, an unsubstituted C1-C40 hydrocarbyl, a C1-C40 substituted hydrocarbyl, an unsubstituted C4-C62 aryl, a substituted C4-C62 aryl, an unsubstituted C4-C62 heteroaryl, a substituted C4-C62 heteroaryl, -NR' 2 , -SR', -OR, -SiR' 3 , -OSiR' 3 , -PR' 2 , or -R"-SiR' 3 , where R" is C1-C10 alkyl and each R' is hydrogen, halogen, C1-C10 alkyl, or Ce-C aryl.
- Each J 1 and J 2 can be hydrogen, an unsubstituted C1-C40 hydrocarbyl, a C1-C40 substituted hydrocarbyl, an unsubstituted C4-C62 aryl, a substituted C4-C62 aryl, an unsubstituted C4-C62 heteroaryl, a substituted C4-C62 heteroaryl, -NR'2, -SR', -OR, -SiR' 3 , -OSiR' 3 , -PR' 2 , or -R"-SiR' 3 , where R" is C1-C10 alkyl and each R' is hydrogen, halogen, C1-C10 alkyl, or C6-C10 aryl.
- J 1 and J 2 can be joined to form a 4, 5 or 6 membered ring.
- Each R 2 -R 6 can be hydrogen, an unsubstituted C1-C40 hydrocarbyl, a C1-C40 substituted hydrocarbyl, an unsubstituted C4-C62 aryl, a substituted C4-C62 aryl, an unsubstituted C4-C62 heteroaryl, a substituted C4-C62 heteroaryl, -NR' 2 , -SR', -OR, -SiR' 3 , -OSiR' 3 , -PR' 2 , or -R"-SiR' 3 , where R" is C1-C10 alkyl and each R' is hydrogen, halogen, C1-C10 alkyl, or Ce-Cio aryl.
- Each R 7 -R 16 can be hydrogen, an unsubstituted C1-C40 hydrocarbyl, a C1-C40 substituted hydrocarbyl, an unsubstituted C4-C62 aryl, a substituted C4-C62 aryl, an unsubstituted C4-C62 heteroaryl, a substituted C4-C62 heteroaryl, -NR' 2 , -SR', -OR, -SiR' 3 , -OSiR' 3 , -PR' 2 , or -R"-SiR' 3 , where R" is C1-C10 alkyl and each R' is hydrogen, halogen, C1-C10 alkyl, or C6-C10 aryl.
- R 11 and R 12 can be joined.
- Each R 17 , R 18 , R 19 , and R 20 can be independently hydrogen, a halogen, an unsubstituted C1-C40 hydrocarbyl, a C1-C40 substituted hydrocarbyl, an unsubstituted C4-C62 aryl, a substituted C4-C62 aryl, an unsubstituted C4-C62 heteroaryl, a substituted C4-C62 heteroaryl, -NR' 2 , -SR', -OR, -SiR' 3 , -OSiR' 3 , -PR' 2 , or -R"-SiR' 3 , wherein R" is C1-O10 alkyl and each R' is hydrogen, halogen, CI-C10 alkyl, or Ce-C aryl.
- indenyl can be considered a cyclopentadienyl fused with a benzene ring.
- the structure below is drawn and named as an anion.
- Non-limiting examples of indacenyls and cyclopenta[b]naphthalenyls include:
- the bridged metallocene compounds can be activated for polymerization catalysis in any manner sufficient to allow coordination or cationic polymerization. This can be achieved for coordination polymerization when one ligand can be abstracted and another will either allow insertion of the unsaturated monomers or will be similarly abstractable for replacement with a ligand that allows insertion of the unsaturated monomer (labile ligands), e.g., alkyl, silyl, or hydride.
- labile ligands e.g., alkyl, silyl, or hydride.
- the traditional activators of coordination polymerization art are suitable, for example, Lewis acids such as aluminoxane compounds, and ionizing, anion precursor compounds that abstract one so as to ionize the bridged metallocene metal center into a cation and provide a counterbalancing noncoordinating anion.
- Lewis acids such as aluminoxane compounds
- anion precursor compounds that abstract one so as to ionize the bridged metallocene metal center into a cation and provide a counterbalancing noncoordinating anion.
- suitable activators can include a cationic component.
- the cationic component can have the formula [R 1 R 2 R 3 AH] + , where A is nitrogen, R 1 and R 2 are together a -(CH2)a- group, where a is 3, 4, 5, or 6 and form, together with the nitrogen atom, a 4-, 5-, 6-, or 7-membered non-aromatic ring to which, via adjacent ring carbon atoms, optionally one or more aromatic or heteroaromatic rings can be fused, and R 3 is Ci, C2, C3, C4, or C5 alkyl, or N-methylpyrrolidinium or N-methylpiperidinium.
- the cationic component can have the formula [R n AH4_ n ] + , where A is nitrogen, n is 2 or 3, and all R are identical and are Ci to C3 alkyl groups, such as trimethylammonium, trimethylanilinium, triethylammonium, dimethylanilinium, and dimethylammonium.
- Suitable activators can also be or include an anionic component, [Y],
- the anionic component can be a non-coordinating anion (NCA), having the formula [B(R 4 ) 4 ]- , where R 4 is an aryl group or a substituted aryl group, of which the one or more substituents are identical or different and are selected from the group consisting of alkyl, aryl, a halogen atom, halogenated aryl, and haloalkylaryl groups.
- the substituents can be perhalogenated aryl groups, or perfluorinated aryl groups, including, perfluorophenyl, perfluoronaphthyl and perfluorobiphenyl.
- Suitable activators can also be or include a non-coordinating anion (NCA) represented by the formula: (Z)d + (Ad-), wherein Z is (L-H) or a reducible Lewis Acid, L is a Lewis base; H is hydrogen; (L-H)+ is a Bronsted acid; Ad- is a non-coordinating anion having the charge d-; and d is an integer from 1 to 3.
- NCA non-coordinating anion
- Suitable activators can also be or include a non-coordinating anion (NCA) represented by the formula: (Z)d+ (Ad-), wherein Ad- is a non-coordinating anion having the charge d-; d is an integer from 1 to 3, and Z is a reducible Lewis acid represented by the formula: (Ar3C+), where Ar is aryl or aryl substituted with a heteroatom, a Ci to C40 hydrocarbyl, or a substituted Ci to C40 hydrocarbyl.
- NCA non-coordinating anion
- Ad- is a non-coordinating anion having the charge d-
- d is an integer from 1 to 3
- Z is a reducible Lewis acid represented by the formula: (Ar3C+), where Ar is aryl or aryl substituted with a heteroatom, a Ci to C40 hydrocarbyl, or a substituted Ci to C40 hydrocarbyl.
- a ratio of the activator(s) to catalyst metal center can be at least 1 :100, or at least 1 :250, or at least 1 :1500.
- the activator can be N,N-dimethylanilinium-tetra(perfluorophenyl)borate, N,N-dimethylanilinium- tetra(perfluoronaphthyl)borate, N,N-dimethylanilinium- tetrakis(perfluorobiphenyl)borate, N,N-dimethylanilinium-tetrakis(3,5- bis(trifluoromethyl)phenyl)borate, triphenylcarbenium-tetra(perfluorophenyl)borate, triphenylcarbenium-tetra(perfluoronaphthyl)borate, triphenylcarbenium- tetrakis(perfluorobiphenyl)borate, or triphenylcarbenium-tetrakis(3,5- bis(trifluor
- the silane bridged metallocenes can be supported or unsupported. When supported, any suitable support material can be used. Suitable support materials can include, but are not limited to, AI2O3, ZrC>2, SiC>2, SiC /AhOs, SiC /TiC , silica clay, silicon oxide/clay, or mixtures thereof. Silica is preferred. Polymerization
- Any conventional solution, slurry, or gas phase polymerization process can be used to make an ethylene copolymer using the diphenylsilane bridged metallocene catalyst provided herein.
- a solution or gas phase process is used.
- any conventional solution or slurry or gas phase polymerization process can be used.
- a solution or slurry process is used.
- Solution polymerization is a bulk polymerization process, which refers to a polymerization process in which the monomers and/or comonomers being polymerized are used as a solvent or diluent using little or no inert solvent as a liquid or diluent. A small fraction of inert solvent might be used as a carrier for a catalyst and a scavenger.
- solution polymerization refers to a polymerization process in which the polymer is dissolved in a liquid polymerization medium, such as an inert solvent, monomer(s), or blends thereof.
- a solution polymerization is typically homogeneous, which refers to a polymerization process where the polymer product is dissolved in the polymerization medium. Such systems are preferably not turbid as described in Oliveira, J. et al (2000), Ind. Eng. Chem. Res., v.29, pg. 4627.
- a homogeneous polymerization process is typically a process where at least 90 wt% of the product is soluble in the reaction media.
- slurry polymerization means a polymerization process where a supported catalyst is employed and monomers are polymerized on the supported catalyst particles, and at least 95 wt% of polymer products derived from the supported catalyst are in granular form as solid particles (not dissolved in the diluent).
- the polymerization is carried out as a slurry (suspension) or solution polymerization
- an inert solvent or diluent can be used.
- Suitable diluents/solvents include non-coordinating, inert liquids.
- Examples include straight and branched-chain hydrocarbons, such as isobutane, butane, pentane, isopentane, hexanes, isohexane, heptane, octane, dodecane, and mixtures thereof; cyclic and alicyclic hydrocarbons, such as cyclohexane, cycloheptane, methylcyclohexane, methylcycloheptane, and mixtures thereof, such as can be found commercially (ISOPARTM); perhalogenated hydrocarbons, such as perfluorinated 04.1 o alkanes, chlorobenzene, and aromatic and alkylsubstituted aromatic compounds, such as benzene, toluene, mesitylene, and xylene.
- hydrocarbons such as isobutane, butane, pentane, isopentane, hexanes, isohexane, heptane,
- Suitable diluents/solvents also include liquid olefins which can act as monomers or co-monomers including ethylene, propylene, 1-butene, 1-hexene, 1-pentene, 3-methyl-1 -pentene, 4-methyl-1 -pentene, 1 -octene, 1 -decene, and mixtures thereof.
- aliphatic hydrocarbon solvents are used as the solvent, such as isobutane, butane, pentane, isopentane, hexanes, isohexane, heptane, octane, dodecane, and mixtures thereof; cyclic and alicyclic hydrocarbons, such as cyclohexane, cycloheptane, methylcyclohexane, methylcycloheptane, and mixtures thereof.
- the diluent/solvent is not aromatic, preferably aromatics are present in the diluent/solvent at less than 1 wt%, preferably less than 0.5 wt%, preferably less than 0 wt% based upon the weight of the diluents/solvents. It is also possible to use mineral spirit or a hydrogenated diesel oil fraction as a solvent. Toluene can also be used. The polymerization is preferably carried out in the liquid monomer(s). If inert solvents are used, the monomer(s) is (are) typically metered in gas or liquid form.
- the at least one other comonomer can include any one or more C4 to C20 olefins.
- the C4 to C20 comonomers can be linear, branched, or cyclic. Suitable C4 to C20 cyclic olefins can be strained or unstrained, monocyclic or polycyclic, and can optionally include heteroatoms and/or one or more functional groups.
- the reactor C2 concentration can range from 0.1 to 40.0 wt%; 1 to 40.0 wt%; 1 to 35 wt%; 2 to 20 wt%, 3 to 10 wt%; or 0.1 to 10 wt%.
- the reactor C4 to C20 comonomers concentration can range from 0.1 - 50 wt%; 1 to 35 wt%, 2 - 20 wt%, or 3 - 10 wt%.
- comonomers include butene, pentene, hexene, heptene, octene, nonene, decene, undecene, dodecene, norbornene, norbornadiene, dicyclopentadiene, cyclopentene, cycloheptene, cyclooctene, cyclooctadiene, cyclododecene, 7-oxanorbornene, 7-oxanorbornadiene, substituted derivatives thereof, and isomers thereof, preferably hexene, heptene, octene, nonene, decene, dodecene, cyclooctene, 1 ,5 -cyclooctadiene, l-hydroxy-4-cyclooctene, 1 -acetoxy-4-cyclooctene, 5-methylcyclopentene, cyclopentene, dicyclopen
- one or more dienes are added to the polymerization process.
- the diene can be present in the polymer produced herein at up to 10 wt%, preferably at 0.00001 to 8.0 wt%, preferably 0.002 to 8.0 wt%, even more preferably 0.003 to 8.0 wt%, based upon the total weight of the composition.
- 500 ppm or less of diene is added to the polymerization, preferably 400 ppm or less, preferably 300 ppm or less.
- at least 50 ppm of diene is added to the polymerization, or 100 ppm or more, or 150 ppm or more.
- Suitable diolefin comonomers include any hydrocarbon structure, preferably C4 to C30, having at least two unsaturated bonds, where at least one of the unsaturated bonds are readily incorporated into a polymer chain during chain growth. It is further preferred that the diolefin comonomers be selected from alpha, omega-diene monomers (i.e., di-vinyl monomers). More preferably, the diolefin comonomers are linear di-vinyl monomers, most preferably those containing from 4 to 30 carbon atoms.
- preferred dienes include butadiene, pentadiene, hexadiene, heptadiene, octadiene, nonadiene, decadiene, undecadiene, dodecadiene, tridecadiene, tetradecadiene, pentadecadiene, hexadecadiene, heptadecadiene, octadecadiene, nonadecadiene, icosadiene, heneicosadiene, docosadiene, tricosadiene, tetracosadiene, pentacosadiene, hexacosadiene, heptacosadiene, octacosadiene, nonacosadiene, triacontadiene, particularly preferred dienes include 1 ,6-heptadiene, 1 ,7-octadiene, 1 ,8-nonadiene, 1
- Preferred cyclic dienes include cyclopentadiene, 5-vinyl-2-norbornene, norbornadiene, 5-ethylidene-2-norbornene, divinylbenzene, and dicyclopentadiene or higher ring containing diolefins with or without substituents at various ring positions.
- the polymers produced from the diphenyl silane bridged metallocenes provided herein can be homopolymers of propylene or copolymers of propylene having from about 0.1 wt% to about 50 wt% based on the total amount of polymer (such as from 1 wt% to 20 wt%) of one or more of C2 or C4 to C20 olefin comonomer, based on a total amount of propylene copolymer, such as from about 0.5 wt% to about 18 wt%, such as from about 1 wt% to about 15 wt%, such as from about 3 wt% to about 10 wt%) of one or more of C2 or C4 to C20 olefin comonomer (such as ethylene or C4 to C12 alpha-olefin, such as ethylene, butene, hexene, octene, decene, dodecene,
- the propylene homopolymer or propylene copolymer produced herein may have some level of isotacticity and can be isotactic or highly isotactic.
- isotactic is defined as having at least 10% isotactic pentads according to analysis by 13 C NMR as described in US 2008/0045638 at paragraph [0613] et seq.
- highly isotactic is defined as having at least 60% isotactic pentads according to analysis by 13 C NMR.
- a propylene homopolymer having at least about 85% isotacticity, such as at least about 90% isotacticity can be produced herein.
- the propylene polymer produced can be atactic.
- Atactic polypropylene is defined to be less than 10% isotactic or syndiotactic pentads according to analysis by 13 C NMR.
- the distribution and the moments of molecular weight (Mw, Mn, Mz, Mw/Mn, etc.), the comonomer content, and the branching index (g 1 ) are determined by using a high temperature Gel Permeation Chromatography (Polymer Char GPC-IR) equipped with a multiple-channel band-filter based Infrared detector IR5 with a multiple-channel band filter based infrared detector ensemble IR5 with band region covering from about 2,700 cm -1 to about 3,000 cm -1 (representing saturated C-H stretching vibration), an 18-angle light scattering detector and a viscometer.
- Three Agilent PLgel 10-pm Mixed-B LS columns are used to provide polymer separation.
- Reagent grade 1 ,2,4-trichlorobenzene (TCB) (from Sigma-Aldrich) comprising -300 ppm antioxidant BHT can be used as the mobile phase at a nominal flow rate of -1 .0 mL/min and a nominal injection volume of -200 pL.
- the whole system including transfer lines, columns, and detectors can be contained in an oven maintained at ⁇ 145°C.
- a given amount of sample can be weighed and sealed in a standard vial with -10 pL flow marker (heptane) added thereto. After loading the vial in the autosampler, the oligomer or polymer may automatically be dissolved in the instrument with -8 mL added TCB solvent at ⁇ 160°C with continuous shaking.
- the sample solution concentration can be from -0.2 to -2.0 mg/ml, with lower concentrations used for higher molecular weight samples.
- the mass recovery can be calculated from the ratio of the integrated area of the concentration chromatography over elution volume and the injection mass which is equal to the pre-determined concentration multiplied by injection loop volume.
- the conventional molecular weight (IR MW) is determined by combining universal calibration relationship with the column calibration which is performed with a series of monodispersed polystyrene (PS) standards ranging from 700 to 10M gm/mole.
- PS monodispersed polystyrene
- the MW at each elution volume is calculated with following equation: where the variables with subscript “PS” stand for polystyrene while those without a subscript are for the test samples.
- a and K for other materials are as calculated as described in the published in literature (e.g., Sun, T. et al. (2001) Macromolecules, v.34, pg.
- Concentrations are expressed in g/cm 3 , molecular weight is expressed in g/mole, and intrinsic viscosity (hence K in the Mark-Houwink equation) is expressed in dL/g unless otherwise noted.
- the comonomer composition is determined by the ratio of the IR5 detector intensity corresponding to CH2 and CH3 channel calibrated with a series of PE and PP homo/copolymer standards whose nominal value are predetermined by NMR or FTIR. In particular, this provides the methyls per 1 ,000 total carbons (CH3/I OOOTC) as a function of molecular weight.
- the short-chain branch (SCB) content per 1 ,000TC (SCB/1000TC) is then computed as a function of molecular weight by applying a chainend correction to the CH3/I OOOTC function, assuming each chain to be linear and terminated by a methyl group at each end.
- the LS molecular weight (/W) at each point in the chromatogram is determined by analyzing the LS output using the Zimm model for static light scattering (Light Scattering from Polymer Solutions’, Huglin, M. B., Ed.; Academic Press, 1972.):
- AR(0) is the measured excess Rayleigh scattering intensity at scattering angle 0
- c is the polymer concentration determined from the IR5 analysis
- A2 is the second virial coefficient
- P(0) is the form factor for a monodisperse random coil
- K o is the optical constant for the system: where NA is Avogadro’s number, and (dn/dc) is the refractive index increment for the system.
- a high temperature Agilent (or Viscotek Corporation) viscometer which has four capillaries arranged in a Wheatstone bridge configuration with two pressure transducers, is used to determine specific viscosity.
- One transducer measures the total pressure drop across the detector, and the other, positioned between the two sides of the bridge, measures a differential pressure.
- the specific viscosity, q s for the solution flowing through the viscometer is calculated from their outputs.
- the intrinsic viscosity, [q] n s /c, where c is concentration and is determined from the IR5 broadband channel output.
- Kps is 0.000175.
- the branching index (g'vis) is calculated using the output of the GPC-IR5-LS- VIS method as follows.
- the average intrinsic viscosity, [n] av g, of the sample is calculated by: where the summations are over the chromatographic slices, i, between the integration limits.
- the ethylene-propylene random copolymers (RCP) produced herein can have: a) ethylene content of from 0.1 to 10 wt%, based on the total weight of the copolymer, such as 0.2-10 wt%; 0.5-10 wt%; 1-10 wt%; 2-10 wt%; or 3-8 wt%; b) an Mw of from about 5,000 to about 1 ,000,000 g/mol (such as from about 25,000 to about 750,000 g/mol, such as from about 50,000 to about 500,000 g/mol, such as from about 80,000 to about 300,000 g/mol, such as from about 100,000 to about 250,000 g/mol, or 5,000 to 500,000 g/mol, or from 5,000 to 350,000 or from 10,000 to 250,000) as determined by GPC-4D; c) a molecular weight distribution, MWD, (Mw/Mn) of greater than about 2, such as from about 2 to about 30, such as from
- ethylene-propylene elastomers and rubbers (EP) produced herein can have: a) ethylene content of from 0.1 to 50 wt%, based on the total weight of the copolymer, such as 1-35 wt%, 2-20 wt%, 3-10 wt%, 10-40 wt%; 10-25 wt%; 15-35 wt%, or 20-40 wt%; b) an Mw of from about 5,000 to about 1 ,000,000 g/mol (such as from about 25,000 to about 750,000 g/mol, such as from about 50,000 to about 500,000 g/mol, such as from about 80,000 to about 300,000 g/mol, or 5,000 to 500,000 g/mol, or from 5,000 to 350,000 or from 10,000 to 250,000, as determined by GPC-4D; c) a molecular weight distribution, MWD, (Mw/Mn) of greater than about 2, such as from about 2 to about 30, such as from
- propylene homopolymers (hPP) produced herein can have: a) an Mw of from about 5,000 to about 1 ,000,000 g/mol (such as from about 25,000 to about 750,000 g/mol, such as from about 50,000 to about 500,000 g/mol, such as from about 80,000 to about 300,000 g/mol, such as from about 80,000 to about 200,000 g/mol, or 5,000 to 500,000 g/mol, or from 5,000 to 350,000 or from 10,000 to 250,000) as determined by GPC-4D; b) a molecular weight distribution, MWD, (Mw/Mn) of greater than about 2, such as from about 2 to about 30, such as from about 3 to about 20, such as from about 4 to about 10 as determined by GPC-4D; c) a g’vis of greater than about 0.6, such as from about 0.7 to about 1 , such as from 0.8 to 0.95, such as from about 0.7 to about 0.90 as determined by GPC-4D
- Catalysts I2 and I3 were prepared using the following modified one pot technique:
- Figure 1 shows a representative spectrum of the in-situ substitution-lithiation for catalyst I2.
- Figure 2 shows the spectrum of the isolated catalyst 12, which confirms excellent purity. The spectrum demonstrates excellent purity of the isolated material.
- Catalysts Ci and C2 were prepared according to previously reported procedures, such as the ones described in US 9,266,910 and in WO2021/034459A1. [0073] Catalyst h was prepared according to the following Scheme 1 :
- ZrCl4(OEt2)2 (0.5 g, 1.31 mmol) was slowly added to a stirring suspension of dilithium- diphenyl(4-(4-tert-butylphenyl)-2-methyl-1 ,5,6,7- tetrahydro-s-indacenyl) (2, 3,4,5- tetramethylcyclopentadienyl)silane (1.05 g, 1.31 mmol, contains 1.2 equiv. BU2O and 0.6 equiv. THF) in diethyl ether ( ⁇ 30mL) at -30°C. The reaction mixture was allowed to warm up to room temperature and was stirred overnight. After 18 hours, solvent was removed in vacuo to give a bright yellow residue.
- An activator solution typically 1.1-1000 molar equivalents of methyl alumoxane (MAO) in toluene was then injected into the reaction vessel along with 500 microliters of toluene.
- Catalyst typically 0.50 mM in toluene, such as 20-40 nmol of catalyst
- another aliquot of toluene 500 microliters were then added to initiate the reaction. Equivalence is determined based on the mol equivalents relative to the moles of the transition metal in the catalyst complex.
- the reaction was then allowed to proceed until a pre-determined amount of pressure had been taken up by the reaction. At this point, the reaction was quenched by pressurizing the vessel with compressed air.
- the glass vial insert containing the polymer product and solvent was removed from the pressure cell and the inert atmosphere glove box, and the volatile components were removed using a Genevac HT-12 centrifuge and Genevac VC3000D vacuum evaporator operating at elevated temperature and reduced pressure.
- the vial was then weighed to determine the yield of the polymer product.
- the resultant polymer was analyzed by Rapid GPC (see below) to determine the molecular weight and by DSC (see below) to determine melting point.
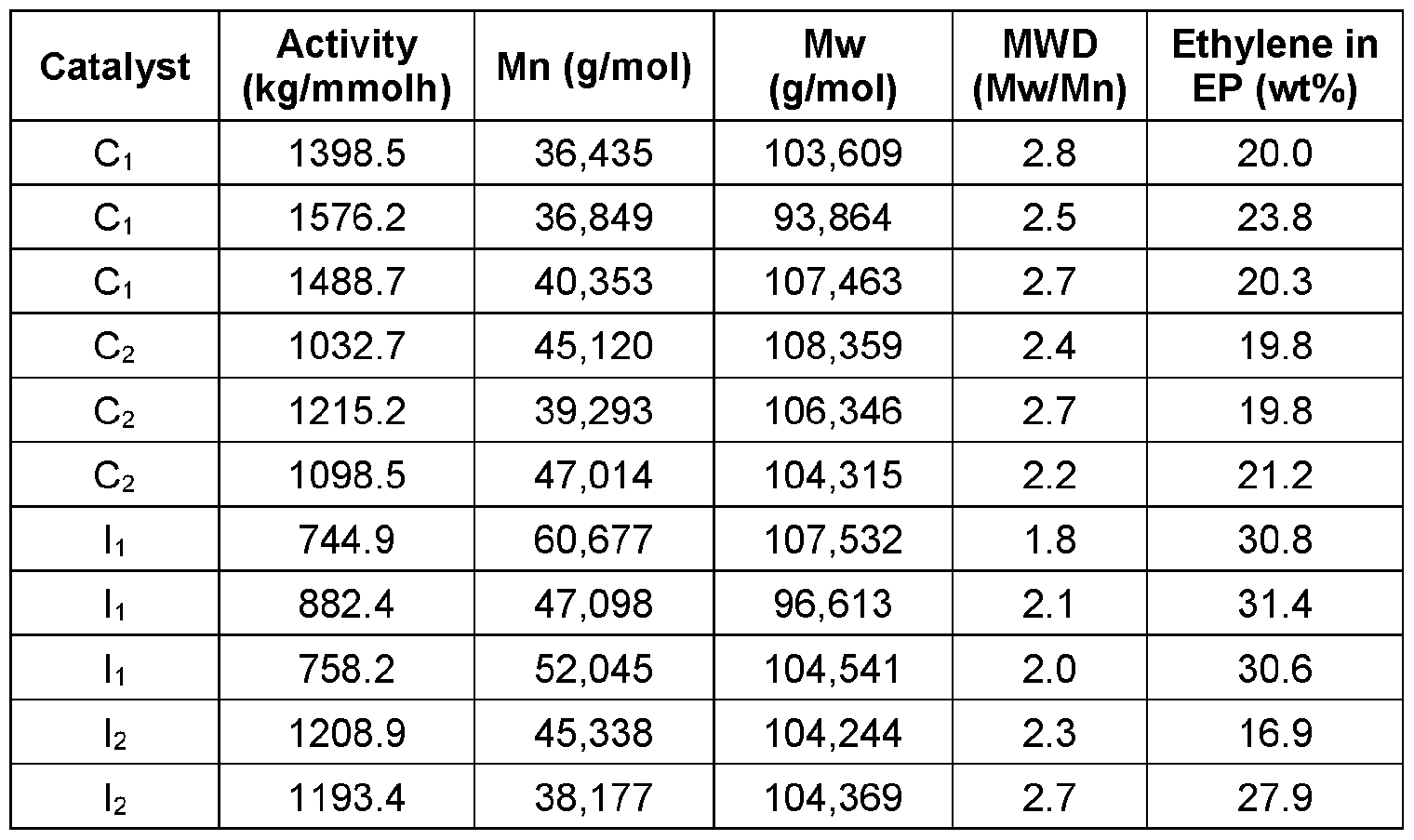
- Table 1 shows catalyst activities and polymerization results for homopolypropylene (hPP) polymerized using the control catalysts Ci, C2 and the inventive catalysts I1-I3.
- Table 2 shows the results for the EP copolymers polymerized using the same catalysts.
- Catalysts C1-C2 and I1-I3 were supported on conventional silica support and used to polymerize propylene under industrially relevant slurry and bulk slurry conditions.
- the slurry was filtered on a glass frit, the solid was washed with toluene (2 x 5 mL) and pentane (2 x 5 mL) and dried in vacuo to give a supported catalyst as orange/red free flowing solids.
- the resulting solids were suspended in mineral oil to make 5 wt% slurry which was used in lab reactor polymerizations.
- a 1 L autoclave reactor equipped with a mechanical stirrer was used for polymer preparation. Prior to the run, the reactor was placed under nitrogen purge while maintaining 90°C temperature for 30 minutes. Upon cooling back to ambient temperature, propylene feed (500 mL), scavenger (0.2 mL of 1 M TIBAL, triisobutylaluminum) and optionally hydrogen (charged from a 50 mL bomb at a desired pressure) were introduced to the reactor and were allowed to mix for 5 minutes. A desired amount of supported catalyst (typically 12.5 - 25.0 mg) was then introduced to the reactor by flushing the pre-determined amount of catalyst slurry (5 wt% in mineral oil) from a catalyst tube with 100 mL of liquid propylene.
- scavenger 0.2 mL of 1 M TIBAL, triisobutylaluminum
- optionally hydrogen charged from a 50 mL bomb at a desired pressure
- the reactor was kept for 5 minutes at room temperature (pre-poly stage), before raising the temperature to 70°C.
- the reaction was allowed to proceed at that temperature for a desired time period (typically 30 minutes). After the given time, the temperature was reduced to 25°C, the excess propylene was vented off and the polymer granules were collected, and dried under vacuum at 60°C overnight.
- Slurry propylene polymerizations were carried out under high-throughput conditions according to the following general procedure.
- a pre-weighed glass vial insert and disposable stirring paddle were fitted to each reaction vessel of the reactor, which contains 48 individual reaction vessels.
- the reactor was then closed and propylene (typically 1-4 mL) was introduced.
- Triisobutyl aluminum (TIBAL) is then introduced as a scavenger (typically 2.5 - 10 pmol) added as either toluene or isohexane solution.
- solvent typically the isohexane was added to bring the total reaction volume, including the subsequent additions, to 5 mL and the reactor vessels were heated to their set temperature (usually from about 50°C to about 110°C).
- ethylene typically 70 - 140 psi
- the contents of the vessel were stirred at 800 rpm.
- Supported catalyst typically 0.75 mg
- was introduced as toluene slurry typically at 3.0 mg I mL concentration
- Equivalence is determined based on the mol equivalents relative to the moles of the transition metal in the catalyst complex.
- the reaction was then allowed to proceed until a pre-determined amount of pressure had been taken up by the reaction or in the case of homopolymerizations it was allowed to proceed for 30 minutes. At this point, the reaction was quenched by pressurizing the vessel with either compressed air or carbon dioxide.
- the glass vial insert containing the polymer product and solvent was removed from the pressure cell and the inert atmosphere glove box, and the volatile components were removed using a Genevac HT-12 centrifuge and Genevac VC3000D vacuum evaporator operating at elevated temperature and reduced pressure.
- the vial was then weighed to determine the yield of the polymer product.
- the resultant polymer was analyzed by Rapid GPC (see below) to determine the molecular weight and by DSC (see below) to determine melting point.
- silane bridged metallocenes h, I2 and I3 showed excellent activity for making hPP and EP copolymers. It was also nothing short of surprising and unexpected that the silane bridged metallocenes h, I2 and I3 were able to produce higher melting point hPPs (>153°C) and ethylene-propylene copolymers with higher molecular weights (Mw), which allows for improved impact properties.
- Rapid GPC procedure To determine various molecular weight related values by GPC, high temperature size 5 exclusion chromatography was performed using an automated “Rapid GPC” system as generally described in US Patent Nos. 6,491 ,816; 6,491 ,823; 6,475,391 ; 6,461 ,515; 6,436,292; 6,406,632; 6,175,409; 6,454,947; 6,260,407; and 6,294,388, each of which is fully incorporated herein by reference for US purposes. This apparatus was a series of three 30 cm x 7.5 mm linear columns, each containing PLgel 10 pm, Mix B.
- the GPC system was calibrated using polystyrene standards ranging from 580 - 3,390,000g/mol. The system was operated at an eluent flow rate of 2.0 mL/minutes and an oven temperature of 165°C. 1 ,2,4-trichlorobenzene was used as the eluent. The polymer samples were dissolved in 1 ,2,4-trichlorobenzene at a concentration of 0.1 - 0.9 mg/mL. 250 uL of a polymer solution was injected into the system. The concentration of the polymer in the eluent was monitored using an evaporative light scattering detector (as shown by the examples in Table 3) or Polymer Char IR4 detector. The molecular weights presented are relative to linear polystyrene standards and are uncorrected.
- DSC Procedure DSC-1 : For the high throughput samples, the melting temperature (Tm) was measured using Differential Scanning Calorimetry (DSC) using commercially available equipment such as a TA Instruments TA-Q200 DSC. Typically, 5 to 10 mg of molded polymer or plasticized polymer was sealed in an aluminum pan and loaded into the instrument at about room temperature. Samples were pre-annealed at about 220°C for about 15 minutes and then allowed to cool to about room temperature overnight.
- DSC Differential Scanning Calorimetry
- the samples were then heated to about 220°C at a heating rate of about 100°C/minute, held at this temperature for at least about 5 minutes, and then cooled at a rate of about 50°C/m inute to a temperature typically at least about 50°C below the crystallization temperature. Melting points were collected during the heating period.
- DSC-2 Peak melting point, Tm, described for reactor batches (also referred to as melting point) and peak crystallization temperature, Tc, (also referred to as crystallization temperature) are determined using the following DSC procedure according to ASTM D3418-03.
- Differential scanning calorimetric (DSC-2) data can be obtained using a TA Instruments model DSC2500 machine. Samples weighing approximately 5 to 10 mg are sealed in an aluminum hermetic sample pan and loaded into the instrument at about room temperature. The DSC data are recorded by first gradually heating the sample to about 200°C at a rate of about 10°C/minute.
- the sample is kept at about 200°C for 5 minutes, then cooled to about -50°C at a rate of about 10°C/minute, followed by an isothermal for about 5 minutes and heating to about 200°C at about 10°C/minute, holding at about 200°C for about 5 minutes and then cooling down to about 25°C at a rate of about 10°C/minute. Both the first and second cycle thermal events were recorded. The melting and crystallization temperatures reported here were obtained during the second heating/cooling cycle unless otherwise noted. In the event of conflict between the DSC Procedure-1 and DSC procedure-2, DSC procedure-2 is used. [0099] Certain embodiments and features have been described using a set of numerical upper limits and a set of numerical lower limits.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Crystallography & Structural Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
- Transition And Organic Metals Composition Catalysts For Addition Polymerization (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2025507414A JP2025526711A (en) | 2022-08-09 | 2023-07-31 | Diphenylsilane-bridged C1 symmetric catalysts and polymers produced therefrom |
| CN202380058180.6A CN119654331A (en) | 2022-08-09 | 2023-07-31 | Method for preparing diphenylsilane-bridged C1-symmetric catalysts and polymers prepared therefrom |
| EP23758488.3A EP4568980A1 (en) | 2022-08-09 | 2023-07-31 | Methods for preparing diphenylsilane bridged c1 symmetric catalysts and polymers made therefrom |
| KR1020257007439A KR20250049337A (en) | 2022-08-09 | 2023-07-31 | Method for preparing diphenylsilane crosslinked C1 symmetric catalyst and polymer prepared therefrom |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202263370829P | 2022-08-09 | 2022-08-09 | |
| US63/370,829 | 2022-08-09 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024036052A1 true WO2024036052A1 (en) | 2024-02-15 |
Family
ID=87762593
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2023/071323 Ceased WO2024036052A1 (en) | 2022-08-09 | 2023-07-31 | Methods for preparing diphenylsilane bridged c1 symmetric catalysts and polymers made therefrom |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP4568980A1 (en) |
| JP (1) | JP2025526711A (en) |
| KR (1) | KR20250049337A (en) |
| CN (1) | CN119654331A (en) |
| WO (1) | WO2024036052A1 (en) |
Citations (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0754698A2 (en) | 1995-07-17 | 1997-01-22 | Mitsui Petrochemical Industries, Ltd. | An olefin polymerization catalyst and a process for preparing olefin polymer |
| WO2000024793A1 (en) | 1998-10-23 | 2000-05-04 | Exxon Chemical Patents Inc. | Bridged metallocenes for olefin copolymerization |
| EP1046642A2 (en) | 1999-04-22 | 2000-10-25 | TARGOR GmbH | Process for preparation of silyl-bridged fluorenyl-cyclopentadienyl ligands and silyl-bridged fluorenyl-cyclopentadienyl metallocenes |
| US6175409B1 (en) | 1999-04-02 | 2001-01-16 | Symyx Technologies, Inc. | Flow-injection analysis and variable-flow light-scattering methods and apparatus for characterizing polymers |
| WO2001005870A1 (en) | 1999-07-19 | 2001-01-25 | Bayer Aktiengesellschaft | Polycarbonates containing aspartic acid ester-functional silicones |
| US6260407B1 (en) | 1998-04-03 | 2001-07-17 | Symyx Technologies, Inc. | High-temperature characterization of polymers |
| US6294388B1 (en) | 1998-04-03 | 2001-09-25 | Symyx Technologies, Inc. | Indirect calibration of polymer characterization systems |
| WO2002002575A1 (en) | 2000-06-30 | 2002-01-10 | Exxonmobil Chemical Patents, Inc. | Metallocenes with a bridged 4-phenyl-indenyl-ligand for olefin polymerization |
| US6406632B1 (en) | 1998-04-03 | 2002-06-18 | Symyx Technologies, Inc. | Rapid characterization of polymers |
| US6436292B1 (en) | 1999-04-02 | 2002-08-20 | Symyx Technologies, Inc. | Parallel high-performance liquid chromatography with post-separation treatment |
| US6491816B2 (en) | 1999-04-02 | 2002-12-10 | Symyx Technologies, Inc. | Apparatus for parallel high-performance liquid chromatography with serial injection |
| US20080045638A1 (en) | 2002-08-12 | 2008-02-21 | Chapman Bryan R | Plasticized hetero-phase polyolefin blends |
| CN101235106A (en) | 2008-01-31 | 2008-08-06 | 河南省科学院化学研究所有限公司 | Preparation method of metallocene catalyst and its application in the production of ethylene-propylene rubber |
| WO2013134038A1 (en) | 2012-03-09 | 2013-09-12 | Exxonmobil Chemical Patents Inc. | Polyolefin adhesive compositions |
| WO2014099303A1 (en) | 2012-12-21 | 2014-06-26 | Exxonmobil Chemical Patents Inc. | Bridged metallocene compounds, catalyst systems and processes for polymerization therewith |
| US9266910B2 (en) | 2013-10-29 | 2016-02-23 | Exxonmobil Chemical Patents Inc. | Asymmetric polypropylene catalysts |
| US9790240B2 (en) | 2012-12-11 | 2017-10-17 | Lg Chem, Ltd. | Ligand compound, a preparation method thereof, a transition metal compound, and a preparation method thereof |
| WO2018151904A1 (en) | 2017-02-20 | 2018-08-23 | Exxonmobil Chemical Patents Inc. | Group 4 catalyst compounds and process for use thereof |
| US20190161559A1 (en) | 2017-11-28 | 2019-05-30 | Exxonmobil Chemical Patents Inc. | Catalyst Systems and Polymerization Processes for Using the Same |
| KR20200003044A (en) | 2017-05-04 | 2020-01-08 | 메조블라스트 인터내셔널 에스에이알엘 | Mesenchymal lineage precursors or stem cells with enhanced immunosuppression |
| US20200043758A1 (en) | 2018-07-31 | 2020-02-06 | Tainan Semiconductor Manufacturing Co., Ltd | Systems and methods for die container warehousing |
| WO2021034459A1 (en) | 2019-08-22 | 2021-02-25 | Exxonmobil Chemical Patents Inc. | Isotactic propylene homopolymers and copolymers produced with c1 symmetric metallocene catalysts |
| WO2021162748A1 (en) | 2020-02-11 | 2021-08-19 | Exxonmobil Chemical Patents Inc. | Polyethylene compositions obtained using transition metal bis(phenolate) catalyst complexes and homogeneous process for production thereof |
| EP3915997A1 (en) * | 2019-03-13 | 2021-12-01 | Lg Chem, Ltd. | Transition metal compound and catalyst composition comprising same |
-
2023
- 2023-07-31 JP JP2025507414A patent/JP2025526711A/en active Pending
- 2023-07-31 EP EP23758488.3A patent/EP4568980A1/en active Pending
- 2023-07-31 CN CN202380058180.6A patent/CN119654331A/en active Pending
- 2023-07-31 WO PCT/US2023/071323 patent/WO2024036052A1/en not_active Ceased
- 2023-07-31 KR KR1020257007439A patent/KR20250049337A/en active Pending
Patent Citations (28)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0754698A2 (en) | 1995-07-17 | 1997-01-22 | Mitsui Petrochemical Industries, Ltd. | An olefin polymerization catalyst and a process for preparing olefin polymer |
| US6454947B1 (en) | 1998-04-03 | 2002-09-24 | Symyx Technologies, Inc. | Overlaid injection for rapid characterizations of polymers |
| US6475391B2 (en) | 1998-04-03 | 2002-11-05 | Symyx Technologies, Inc. | Rapid characterization of polymers for combinatorial, analytical and process control applications |
| US6491823B1 (en) | 1998-04-03 | 2002-12-10 | Symyx Technologies, Inc. | Targeted separation protocols for rapid characterizations of polymers |
| US6461515B1 (en) | 1998-04-03 | 2002-10-08 | Symyx Technologies, Inc. | Parallel liquid chromatography for analyzing combinatorial libraries of non-biological polymers |
| US6260407B1 (en) | 1998-04-03 | 2001-07-17 | Symyx Technologies, Inc. | High-temperature characterization of polymers |
| US6294388B1 (en) | 1998-04-03 | 2001-09-25 | Symyx Technologies, Inc. | Indirect calibration of polymer characterization systems |
| US6406632B1 (en) | 1998-04-03 | 2002-06-18 | Symyx Technologies, Inc. | Rapid characterization of polymers |
| WO2000024793A1 (en) | 1998-10-23 | 2000-05-04 | Exxon Chemical Patents Inc. | Bridged metallocenes for olefin copolymerization |
| US6175409B1 (en) | 1999-04-02 | 2001-01-16 | Symyx Technologies, Inc. | Flow-injection analysis and variable-flow light-scattering methods and apparatus for characterizing polymers |
| US6436292B1 (en) | 1999-04-02 | 2002-08-20 | Symyx Technologies, Inc. | Parallel high-performance liquid chromatography with post-separation treatment |
| US6491816B2 (en) | 1999-04-02 | 2002-12-10 | Symyx Technologies, Inc. | Apparatus for parallel high-performance liquid chromatography with serial injection |
| EP1046642A2 (en) | 1999-04-22 | 2000-10-25 | TARGOR GmbH | Process for preparation of silyl-bridged fluorenyl-cyclopentadienyl ligands and silyl-bridged fluorenyl-cyclopentadienyl metallocenes |
| WO2001005870A1 (en) | 1999-07-19 | 2001-01-25 | Bayer Aktiengesellschaft | Polycarbonates containing aspartic acid ester-functional silicones |
| WO2002002575A1 (en) | 2000-06-30 | 2002-01-10 | Exxonmobil Chemical Patents, Inc. | Metallocenes with a bridged 4-phenyl-indenyl-ligand for olefin polymerization |
| US20080045638A1 (en) | 2002-08-12 | 2008-02-21 | Chapman Bryan R | Plasticized hetero-phase polyolefin blends |
| CN101235106A (en) | 2008-01-31 | 2008-08-06 | 河南省科学院化学研究所有限公司 | Preparation method of metallocene catalyst and its application in the production of ethylene-propylene rubber |
| WO2013134038A1 (en) | 2012-03-09 | 2013-09-12 | Exxonmobil Chemical Patents Inc. | Polyolefin adhesive compositions |
| US9790240B2 (en) | 2012-12-11 | 2017-10-17 | Lg Chem, Ltd. | Ligand compound, a preparation method thereof, a transition metal compound, and a preparation method thereof |
| WO2014099303A1 (en) | 2012-12-21 | 2014-06-26 | Exxonmobil Chemical Patents Inc. | Bridged metallocene compounds, catalyst systems and processes for polymerization therewith |
| US9266910B2 (en) | 2013-10-29 | 2016-02-23 | Exxonmobil Chemical Patents Inc. | Asymmetric polypropylene catalysts |
| WO2018151904A1 (en) | 2017-02-20 | 2018-08-23 | Exxonmobil Chemical Patents Inc. | Group 4 catalyst compounds and process for use thereof |
| KR20200003044A (en) | 2017-05-04 | 2020-01-08 | 메조블라스트 인터내셔널 에스에이알엘 | Mesenchymal lineage precursors or stem cells with enhanced immunosuppression |
| US20190161559A1 (en) | 2017-11-28 | 2019-05-30 | Exxonmobil Chemical Patents Inc. | Catalyst Systems and Polymerization Processes for Using the Same |
| US20200043758A1 (en) | 2018-07-31 | 2020-02-06 | Tainan Semiconductor Manufacturing Co., Ltd | Systems and methods for die container warehousing |
| EP3915997A1 (en) * | 2019-03-13 | 2021-12-01 | Lg Chem, Ltd. | Transition metal compound and catalyst composition comprising same |
| WO2021034459A1 (en) | 2019-08-22 | 2021-02-25 | Exxonmobil Chemical Patents Inc. | Isotactic propylene homopolymers and copolymers produced with c1 symmetric metallocene catalysts |
| WO2021162748A1 (en) | 2020-02-11 | 2021-08-19 | Exxonmobil Chemical Patents Inc. | Polyethylene compositions obtained using transition metal bis(phenolate) catalyst complexes and homogeneous process for production thereof |
Non-Patent Citations (5)
| Title |
|---|
| "Light Scattering from Polymer Solutions", 1972, ACADEMIC PRESS |
| HEUER, B. ET AL.: "Alternating Ethene/Propene Copolymers by Ci-Symmetric Metallocene/ MAO Catalysts", MACROMOLECULES, vol. 38, no. 8, 2005, pages 3054 - 3059, XP001227418, DOI: 10.1021/ma048009z |
| IZMER, V. ET AL.: "ansa-Metallocenes with a Ph2Si Bridge: Molecular Structures of HfCl2[Ph2Si(n5-CsH4)] and HfCl2[Ph2Si(Ci3H9)(n5-CSH4)]2", J. CHEM. SOC., DALTON TRANS., 2001, pages 1131 - 1136 |
| OLIVEIRA, J. ET AL., IND. ENG. CHEM. RES., vol. 29, 2000, pages 4627 |
| SUN, T. ET AL., MACROMOLECULES, vol. 34, 2001, pages 6812 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2025526711A (en) | 2025-08-15 |
| KR20250049337A (en) | 2025-04-11 |
| EP4568980A1 (en) | 2025-06-18 |
| CN119654331A (en) | 2025-03-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3022235B1 (en) | Metallocenes and catalyst compositions derived therefrom | |
| US9676882B2 (en) | Processes using staged hydrogen addition | |
| US20220315680A1 (en) | Isotactic Propylene Homopolymers and Copolymers Produced with C1 Symmetric Metallocene Catalysts | |
| US9644047B2 (en) | Metallocenes and catalyst compositions derived therefrom | |
| US9944665B2 (en) | Bridged bis(indenyl) transitional metal complexes, production, and use thereof | |
| US20200362071A1 (en) | Single Reactor Production of Polymers in Gas or Slurry Phase | |
| WO2016196331A1 (en) | Supported metallocene catalyst systems for polymerization | |
| SG195587A1 (en) | Catalyst system and process for olefin polymerization | |
| JP2016530360A (en) | Process using substituted metallocene catalysts and products derived therefrom | |
| SG178201A1 (en) | Catalyst system, process for olefin polymerization, and polymer compositions produced therefrom | |
| US20250002617A1 (en) | C1 symmetric metallocene catalysts tailored for production of vinyl-terminated polypropylene oligomers and macromonomers | |
| WO2018075245A1 (en) | Mixed catalyst systems and methods of using the same | |
| WO2017011073A1 (en) | Substituted bis indenyl metallocene catalyst compounds comprising-si-si-bridge | |
| EP4247825A1 (en) | Metallocene polypropylene prepared using aromatic solvent-free supports | |
| WO2024036052A1 (en) | Methods for preparing diphenylsilane bridged c1 symmetric catalysts and polymers made therefrom | |
| WO2018182860A1 (en) | Solution process to make ethylene copolymers | |
| EP3802644A1 (en) | Metallocenes with si-si bridges | |
| WO2015095188A1 (en) | Bridged bis(indenyl) transitional metal complexes, production, and use thereof | |
| US20240317905A1 (en) | Polymers of vinylcyclobutane and processes for making same | |
| WO2025117332A1 (en) | Methods for producing impact copolymers using dianionic complexes containing eight-membered chelate rings | |
| WO2026024926A1 (en) | Metallocene catalyst compounds for producing polyolefins | |
| WO2022093814A1 (en) | Non-aromatic hydrocarbon soluble olefin polymerization catalysts and use thereof | |
| WO2020214288A1 (en) | Amido-benzoquinone catalyst systems and processes thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 23758488 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2025507414 Country of ref document: JP Ref document number: 202380058180.6 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11202500318X Country of ref document: SG |
|
| WWP | Wipo information: published in national office |
Ref document number: 11202500318X Country of ref document: SG |
|
| ENP | Entry into the national phase |
Ref document number: 20257007439 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020257007439 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2023758488 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2023758488 Country of ref document: EP Effective date: 20250310 |
|
| WWP | Wipo information: published in national office |
Ref document number: 202380058180.6 Country of ref document: CN |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020257007439 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2023758488 Country of ref document: EP |