WO2023208928A1 - Dosage of psma-ligands for fluorescence based detection of cancerous tissue - Google Patents
Dosage of psma-ligands for fluorescence based detection of cancerous tissue Download PDFInfo
- Publication number
- WO2023208928A1 WO2023208928A1 PCT/EP2023/060799 EP2023060799W WO2023208928A1 WO 2023208928 A1 WO2023208928 A1 WO 2023208928A1 EP 2023060799 W EP2023060799 W EP 2023060799W WO 2023208928 A1 WO2023208928 A1 WO 2023208928A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- labeling
- cancerous tissue
- compound
- subject
- dose
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/001—Preparation for luminescence or biological staining
- A61K49/0013—Luminescence
- A61K49/0017—Fluorescence in vivo
- A61K49/0019—Fluorescence in vivo characterised by the fluorescent group, e.g. oligomeric, polymeric or dendritic molecules
- A61K49/0021—Fluorescence in vivo characterised by the fluorescent group, e.g. oligomeric, polymeric or dendritic molecules the fluorescent group being a small organic molecule
- A61K49/0032—Methine dyes, e.g. cyanine dyes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/001—Preparation for luminescence or biological staining
- A61K49/0013—Luminescence
- A61K49/0017—Fluorescence in vivo
- A61K49/005—Fluorescence in vivo characterised by the carrier molecule carrying the fluorescent agent
- A61K49/0056—Peptides, proteins, polyamino acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K51/00—Preparations containing radioactive substances for use in therapy or testing in vivo
- A61K51/02—Preparations containing radioactive substances for use in therapy or testing in vivo characterised by the carrier, i.e. characterised by the agent or material covalently linked or complexing the radioactive nucleus
- A61K51/04—Organic compounds
- A61K51/0402—Organic compounds carboxylic acid carriers, fatty acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K51/00—Preparations containing radioactive substances for use in therapy or testing in vivo
- A61K51/02—Preparations containing radioactive substances for use in therapy or testing in vivo characterised by the carrier, i.e. characterised by the agent or material covalently linked or complexing the radioactive nucleus
- A61K51/04—Organic compounds
- A61K51/08—Peptides, e.g. proteins, carriers being peptides, polyamino acids, proteins
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/531—Production of immunochemical test materials
- G01N33/532—Production of labelled immunochemicals
- G01N33/533—Production of labelled immunochemicals with fluorescent label
-
- G01N33/57555—
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/58—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving labelled substances
- G01N33/582—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving labelled substances with fluorescent label
Definitions
- the present invention relates to a use of a labeling compound having a chemical structure of formula (I): (A)-xi-(B) -X2-(C), wherein (A) is at least one motif specifically binding to cell membranes of cancer cells; (B) at least one chelator moiety; (C) a dye moiety; xi is a spacer covalently connecting (A) and (B); X2 is a spacer or a chemical single bond connecting (B) and (C), or a pharmaceutically acceptable salt thereof, as a labeling agent for detecting cancerous tissue in a subject, said use comprising administration of a labeling dose of said labeling compound to said subject; and to compounds for use, and methods related thereto.
- a labeling compound having a chemical structure of formula (I): (A)-xi-(B) -X2-(C), wherein (A) is at least one motif specifically binding to cell membranes of cancer cells; (B) at least one chelator moiety; (C)
- Prostate cancer is the leading cancer in the US and European population. At least 1-2 million men in the western hemisphere suffer from prostate cancer and it is estimated that the disease will affect one in six men between the ages of 55 and 85. There are more than 300,000 new cases of prostate cancer diagnosed each year in the USA. The mortality from the disease is second only to lung cancer.
- imaging methods with high resolution of the anatomy such as computed tomography (CT), magnetic resonance (MR) imaging and ultrasound, predominate for clinical imaging of prostate cancer.
- An estimated annual $ 2 billion is currently spent worldwide on surgical, radiation, drug therapy and minimally invasive treatments.
- CT computed tomography
- MR magnetic resonance
- tumors may express unique proteins associated with their malignant phenotype or may over-express normal constituent proteins in greater number than normal cells.
- the expression of distinct proteins on the surface of tumor cells offers the opportunity to diagnose and characterize disease by probing the phenotypic identity and biochemical composition and activity of the tumor.
- Radioactive molecules that selectively bind to specific tumor cell surface proteins provide an attractive route for imaging and treating tumors under non-invasive conditions.
- a promising new series of low molecular weight imaging agents targets the prostate-specific membrane antigen (PSMA) (Mease R.C. et al. Clin Cancer Res. 2008, 14, 3036-3043; Foss, C.A.; et al.
- PSMA prostate-specific membrane antigen
- PSMA is a trans-membrane, 750 amino acid type II glycoprotein that has abundant and restricted expression on the surface of PCa, particularly in androgen-independent, advanced and metastatic disease (Schulke, N.; et al. Proc Natl Acad Sci U S A 2003, 100, 12590-12595). The latter is important since almost all PCa become androgen independent over the time. PSMA possesses the criteria of a promising target for therapy (Schulke, N.; et al. Proc. Natl. Acad. Sci. U S A 2003, 100, 12590-12595).
- the PSMA gene is located on the short arm of chromosome 11 and functions both as a folate hydrolase and neuropeptidase.
- GCPII glutamate carboxypeptidase II
- brain PSMA glutamate carboxypeptidase II
- NAAG N-acetylaspartylglutamate
- NAA N-acetylaspartate
- the present invention relates to a use of a labeling compound having a chemical structure of formula (I): (A)-XI-(B)-X2-(C), wherein (A) is at least one motif specifically binding to cell membranes of cancer cells; (B) at least one chelator moiety; (C) a dye moiety; xi is a spacer covalently connecting (A) and (B); X2 is a spacer or a chemical single bond connecting (B) and (C), or a pharmaceutically acceptable salt thereof, as a labeling agent for detecting cancerous tissue in a subject, said use comprising administration of a labeling dose of said labeling compound to said subject.
- a labeling compound having a chemical structure of formula (I): (A)-XI-(B)-X2-(C), wherein (A) is at least one motif specifically binding to cell membranes of cancer cells; (B) at least one chelator moiety; (C) a dye moiety; xi is a spacer co
- the terms “have”, “comprise” or “include” or any arbitrary grammatical variations thereof are used in a non-exclusive way. Thus, these terms may both refer to a situation in which, besides the feature introduced by these terms, no further features are present in the entity described in this context and to a situation in which one or more further features are present.
- the expressions “A has B”, “A comprises B” and “A includes B” may both refer to a situation in which, besides B, no other element is present in A (i.e. a situation in which A solely and exclusively consists of B) and to a situation in which, besides B, one or more further elements are present in entity A, such as element C, elements C and D or even further elements.
- the expressions “comprising a” and “comprising an” preferably refer to “comprising one or more", i.e. are equivalent to "comprising at least one”.
- standard conditions if not otherwise noted, relates to IUPAC standard ambient temperature and pressure (SATP) conditions, i.e. preferably, a temperature of 25°C and an absolute pressure of 100 kPa; also preferably, standard conditions include a pH of 7.
- SATP standard ambient temperature and pressure
- the term “about” relates to the indicated value with the commonly accepted technical precision in the relevant field, preferably relates to the indicated value ⁇ 20%, more preferably ⁇ 10%, most preferably ⁇ 5%.
- the term “essentially” indicates that deviations having influence on the indicated result or use are absent, i.e. potential deviations do not cause the indicated result to deviate by more than ⁇ 20%, more preferably ⁇ 10%, most preferably ⁇ 5%.
- compositions defined using the phrase “consisting essentially of’ encompasses any known acceptable additive, excipient, diluent, carrier, and the like.
- a composition consisting essentially of a set of components will comprise less than 5% by weight, more preferably less than 3% by weight, even more preferably less than 1%, most preferably less than 0.1% by weight of non-specified component(s).
- the use of the labeling compound comprises at least administration of a labeling dose of said labeling compound.
- the use may, however, in addition comprise further steps before, concomitant to, and/or after said administration deemed appropriate by the skilled person.
- the use preferably, additionally comprises at least one step as specified herein, in particular a step of a use and/or a step of a method as described herein.
- the use comprises intraoperative identification of cancerous tissue.
- the labeling compound has a structure of formula (I) (A)-XI-(B)-X2-(C), or is a pharmaceutically acceptable salt thereof, wherein
- (A) is at least one motif specifically binding to cell membranes of cancer cells
- (C) a dye moiety
- xi is a spacer covalently connecting (A) and (B);
- X2 is a spacer or a chemical single bond connecting (B) and (C). and wherein xi comprises a group -AA-, wherein AA is an amino acid sequence of 4 to 8 (preferably 5 to 7, more preferably 6) naturally occurring amino acids and wherein at least two amino acids are histidine
- the compound is PSMA-914 (“Glu-urea-Lys-(HE)3-HBED-CC-PEG2-
- IRDye800CW has the following chemical structure, including pharmaceutically
- the labeling compound has a structure of formula (I): (A)-XI-(B)-X2-(C), including pharmaceutically acceptable salts thereof, wherein
- (A) is at least one motif specifically binding to cell membranes of cancer cells
- (C) a dye moiety
- xi is a spacer covalently connecting (A) and (B);
- X2 is a spacer or a chemical single bond connecting (B) and (C); wherein xi comprises a group -AA-, wherein AA is an amino acid sequence of 4 to 8 (preferably 5 to 7, more preferably 6) naturally occurring amino acids and wherein at least two amino acids are histidine.
- any remaining positive or negative charge or charges are compensated by pharmaceutically acceptable negatively or positively charged counterion or counterions.
- pharmaceutically acceptable salt may be understood in the broadest sense as any charged form of the labeling compound of the present description.
- the compound may, exemplarily, comprise one or more charged residue(s) selected from the group consisting of but not limited to carboxylate anion residue(s), primary ammonium cation(s), secondary ammonium cation residue(s), tertiary ammonium cation residue(s), primary phosphate anion residue(s), secondary phosphate anion residue(s), sulfate anion residue(s), sulfite anion residue(s) and an alkoxide residue(s).
- carboxylate anion residue(s) selected from the group consisting of but not limited to carboxylate anion residue(s), primary ammonium cation(s), secondary ammonium cation residue(s), tertiary ammonium cation residue(s), primary phosphate anion residue(s), secondary phosphate anion residue(s), sulfate anion residue(s), sulfite anion residue(s) and an alkoxide residue(s).
- the counterions may be any ions known to be pharmaceutically acceptable in the art such as, e.g., acetate, fatty acid carboxylate, chloride, sodium ions, potassium ion, magnesium ion, calcium ion, aluminum ion, lithium ion, ammonium, phosphate, hydroxyl, proton and fluoride ion.
- the term “motif’ may be understood in the broadest sense as a molecular structure pattern that enables specific binding to cell membranes of cancer cells.
- the compound or pharmaceutically acceptable salt thereof comprises a moiety (A) with at least one motif.
- moiety (A) can represent one or more motifs.
- Exemplarily moiety (A) represents a chemical structure with one, two or three motifs. However, preferably (A) represents one motif.
- the motif comprises at least one naturally occurring amino acid moiety, more preferably at least two naturally occurring amino acid moieties.
- the terms “moiety”, “residue” and “rest” in the context of a chemical structure may be understood interchangeably in the broadest sense as a part of a molecule tightly bound to the other parts of the molecule, in particular via a covalent bond.
- conjuggated to” and “bound to” as used herein may be understood interchangeably.
- the motif comprises at least one non-proteinogenic amide bond, more preferably at least two non-proteinogenic amide bonds. More preferably, the motif comprises at least one naturally occurring amino acid moiety conjugated via a non-proteinogenic amide bond, even more preferably at least two naturally occurring amino acid moieties conjugated via non- proteinogenic amide bonds.
- the motif specifically binding to cell membranes of cancer cells comprises not more than 20 amino acid moieties, more preferably not more than ten amino acid moieties, even more preferably not more than five amino acid moieties, even more preferably not more than four amino acid moieties, in particular not more than three amino acid moieties.
- the motif further comprises at least one urea moiety, more preferably at least one urea moiety covalently bound to two amino acids via amide bond formation.
- the at least one motif specifically binding to cell membranes of cancer cells (A) is covalently linked with the at least one chelator moiety (B) via spacer xi.
- the spacer xi is of not more than 5 nm in length, preferably of not more than 2 nm in length, in particular of not more than 1 nm in length.
- the motif may preferably be conjugated to the chelator moiety (B) via the epsilon amino group of a lysine moiety.
- chelator moiety may be understood in the broadest sense as any moiety that is able to form a complex with a radiometal under suitable conditions.
- chelator moiety chelant
- chelating moiety chelating moiety
- complexing moiety may be understood interchangeably.
- the chelator moiety is preferably an organic moiety.
- Complexing by chelation preferably involves the formation or presence of two or more separate coordinate bonds between a polydentate (multiple bonded) ligand and a single central radiometal.
- IUPAC International Union of Pure and Applied Chemistry
- Chelator moieties as used herein will typically bear at least two heteroatoms enabling an interaction with a radiometal.
- the chelator moiety will have at least three, in particular at least four heteroatoms enabling an interaction with a metal, in particular a radiometal.
- the chelator moiety may comprise a chelated metal, preferably a metal ion, more preferably a radiometal ion, all a specified herein below. More preferably, the labeling agent is provided and/or admininstered without chelated metal.
- the labeling compound or pharmaceutically acceptable salt thereof comprises a moiety (B) with at least one chelator moiety.
- moiety (B) can represent one or more chelators.
- Exemplarily moiety (B) represents a chemical structure with one, two or three chelators. However, preferably (B) represents one chelator.
- Moiety (B) is connected to moiety (A) via spacer xi as outlined herein.
- the compound or pharmaceutically acceptable salt thereof comprises a dye moiety (C), which may also be referred to as a "label”.
- Moiety (C) is connected to (B) via x 2 , which represents a spacer or a chemical single bond.
- the moiety (C) has preferably the formula wherein
- X 2 and X 3 are independently selected from the group consisting of O, S, Se, N(R 5 ), and C(R 6 R 7 ), preferably both are C(CH3) 2 ;
- Y is a linker connecting the two moieties of (C) and permitting electron delocalization between said moieties, wherein Y optionally comprises a group (L-)cZ 0 ; a and b are independently selected from the group consisting of 1, 2, and 3; each R 1 and each R 2 is independently selected from the group consisting of (L-) C Z, (L-) C Z° and H; and two adjacent R 1 and/or two adjacent R 2 can also form an aromatic ring, which is optionally substituted with one or more (L-) C Z or (L-) C Z°;
- R 3 , R 4 , R 5 , R 6 , R 7 , R 9 are independently selected from the group consisting of (L-)cZ, (L-) C Z°, and H; each c is independently 0, or 1 ; each L is independently T 1 , -OT 1 -, -ST 1 -, -Cf jT 1 -, -QOjOT 1 -, -(DC(O)T 1 -, -Cf jNHT 1 -, - NH OjT 1 , or a Ci -io alkylene group, which is optionally interrupted and/or terminated by one or more of -O-, -S-, -C(O)-, -C(O)O-, -OC(O)-, -C(O)NH-, -NHC(O)O-, and T 1 ;
- T 1 is phenyl, naphthyl, indenyl, indanyl, tetralinyl, decalinyl, adamantyl, C3-7 cycloalkyl, 3 to 7 membered heterocyclyl, or 7 to 11 membered heterobicyclyl, wherein T 1 is optionally substituted with one or more substituents selected from the group consisting of halogen, CN, C(O)R 8 , COOR 8 , OR 8 , C(O)N(R 8 R 8a ), S(O) 2 N(R 8 R 8a ), S(O)N(R 8 R 8a ), S(O) 2 R 8 , N(R 8 )S(O) 2 N(R 8a R 8b ), SR 8 , N(R 8 R 8a ), NO 2 ; OC(O)R 8 , N(R 8 )C(O)R 8a , N(R 8 )S(O) 2 R 8a
- R 8 , R 8a , R 8b are independently selected from the group consisting of H, or Ci-6 alkyl, wherein Ci-6 alkyl is optionally substituted with one or more halogen, which are the same or different;
- Z° is a chemical bond connecting (C) to x 2 or to (B) in case x 2 is a chemical single bond; provided that one of R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 9 is (L-) C Z° or that Y comprises (L-) C Z°.
- substituents means one, two or three, preferably one or two substituents and more preferably one substituent. Generally these substituents can be the same or different.
- Alkyl means a straight-chain or branched hydrocarbon chain. Each hydrogen of an alkyl carbon may be replaced by a substituent as further specified herein.
- alkyl As used throughout the present application, the terms “alkyl”, “alkyl residue” and “alkyl group” and “alkyl moiety” may be understood as a straight-chain or branched saturated hydrocarbon chain. “Straight-chain” may be also designated as “unbranched” or “linear”. Preferably, the alkyl is a straight chain.
- alkylene means a straight-chain or branched saturated hydrocarbon chain wherein two moieties of a molecule are linked by the alkylene residue. “Straight-chain” may be also designated as “unbranched” or “linear”. Each hydrogen of an alkylene carbon may or may not be replaced by a substituent (i.e., may be substituted or unsubstituted) as further specified herein).
- Ci-4 alkyl means an alkyl chain having 1 - 4 carbon atoms, e.g. if present at the end of a molecule: methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, or e.g. - CH 2 -, -CH 2 -CH 2 -, -CH(CH 3 )-, -CH 2 -CH 2 -CH 2 -, -CH(C 2 H 5 )-, -C(CH 3 ) 2 -, when two moieties of a molecule are linked by the alkyl group.
- CM alkyl means an alkyl chain having 1 - 6 carbon atoms, e.g. if present at the end of a molecule: C alkyl, methyl, ethyl, n-propyl, isopropyl, n- butyl, isobutyl, sec-butyl; tert-butyl, n-pentyl, n-hexyl, or e.g.
- CM alkyl carbon when two moieties of a molecule are linked by the alkyl group.
- Each hydrogen of a CM alkyl carbon may be replaced by a substituent as further specified herein.
- C1-8 alkylene residue means an alkylene chain having 1 - 8 carbon atoms, e.g. -CH2-, -CH2-CH2-, -CH(CH 3 )-, -CH2-CH2-CH2-, -CH(C 2 H 5 )-, -C(CH 3 ) 2 -, -CH 2 -C(CH 3 )2-, - C(CH 2 -CH 3 )2-, -CH(CH 2 -CH 3 )-, -CH2-CH(CH 3 )(CH 2 -CH3)-,
- C4-8 alkylene and “Ce alkylene” are defined accordingly.
- C3-7 alkylene and “C5 (C4) alkylene” are defined accordingly.
- C1-10 alkylene group means a bivalent straight-chain or branched hydrocarbon chain having 1 to 10 carbon atoms.
- Each hydrogen of an alkyl carbon may be replaced by a substituent as further specified herein. Examples are methylene (-CH2-) -CH2- CH 2 -, -CH(CH 3 )-, -CH2-CH2-CH2-, -CH(C 2 H 5 )-, -C(CH 3 ) 2 -.
- Each hydrogen of a Ci-io alkylene group carbon may be replaced by a substituent as further specified herein.
- “C1-10 alkylene residue” means an alkylene chain having 1 - 10 carbon atoms when two moieties of a molecule are linked by the alkylene group.
- the C1-10 alkylene residue in the context of residue f of the spacer y is a straight-chain, i.e., unbranched, C1-10 alkylene residue, in which optionally one or more hydrogen(s) are substituted and/or in which optionally one or more -CEE- moieties may be replaced by -O- or -NH-.
- one or more -CH2- moieties may optionally be replaced by” means that the indicated number of CEE groups can be replaced by an atom or group specified herein.
- one or more hydrogens as specified herein can be replaced by a substituent.
- Ci-10 alkylene group “optionally interrupted and/or terminated” means that the alkylene chain is interrupted between two carbon atoms by an atom or a chemical group as specified herein or the alkylene group is terminated by said atom or group following the carbon at least at one end of the alkylene chain or the alkylene chain is both, interrupted and terminated or the alkylene chain is neither interrupted nor terminated.
- a C3 alkylene group which is optionally interrupted and/or terminated with one or more X may have the sequence C-C-C, C-C-C-X, X-C-C-C, X-C-C-C-X, C-X-C-C, C-C-X-C, C-X-C-X-C, X-C-C-X-C, X-C-X-C-X-C, X-C-X-C-C-X, X-C-X-C-C-X, X-C-X-C-C-X.
- carbocycle refers to a partly or fully saturated or aromatic carbocyclic mono-, bi- or tricyclic fused or unfused ring system. This includes phenyl and C3-7 cycloalkyl rings. Preferred carbocycles having 5, 6 or 7 carbon atoms are cyclopentene, cyclohexene, phenyl, cycloheptane, especially cyclohexane.
- C3-7 cycloalkyl or “C3 -7 cycloalkyl ring” means a cyclic alkyl chain having 3 - 7 carbon atoms, e.g. cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cyclohexenyl, cycloheptyl.
- cycloalkyl refers to cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, or cycloheptyl.
- Each hydrogen of a cycloalkyl carbon may be replaced by a substituent as further specified herein.
- the term “C3-5 cycloalkyl” or “C3-5 cycloalkyl ring” is defined accordingly.
- Halogen means fluoro, chloro, bromo or iodo. It is generally preferred that halogen is fluoro or chloro.
- aromatic ring means a carbocyclic or heterocyclic aromatic ring. Examples are benzene, naphthalene, 5 to 6 membered aromatic heterocycle and 9 to 11 membered aromatic heterobicyclyl.
- Examples for a 3 to 7 membered heterocycle are aziridine, azetidine, oxetane, thietane, furan, thiophene, pyrrole, pyrroline, imidazole, imidazoline, pyrazole, pyrazoline, oxazole, oxazoline, isoxazole, isoxazoline, thiazole, thiazoline, isothiazole, isothiazoline, thiadiazole, thiadiazoline, tetrahydrofiiran, tetrahydrothiophene, pyrrolidine, imidazolidine, pyrazolidine, oxazolidine, isoxazolidine, thiazolidine, isothiazolidine, thiadiazolidine, sulfolane, pyran, dihydropyran, tetrahydropyran, imidazolidine, pyridine, pyridazine, pyrazin
- heterocycles examples include furan, thiophene, pyrrole, imidazole, pyrazole, oxazole, isoxazole, thiazole, isothiazole, thiadiazole, triazole, tetrazole, pyridine, pyrimidine, pyridazine, pyrazine, triazine.
- heterocycles are furan, thiophene, pyrrole, imidazole, pyrazole, oxazole, isoxazole, thiazole, isothiazole, thiadiazole, triazole, tetrazole.
- Examples for a 7 to 11 membered heterobicycle are indole, indoline, benzofuran, benzothiophene, benzoxazole, benzisoxazole, benzothiazole, benzisothiazole, benzimidazole, benzimidazoline, quinoline, quinazoline, dihydroquinazoline, quinoline, dihydroquinoline, tetrahydroquinoline, decahydroquinoline, isoquinoline, decahydroisoquinoline, tetrahydroisoquinoline, dihydroisoquinoline, benzazepine, purine or pteridine.
- 7 to 11 membered heterobicycle also includes spiro structures of two rings like 6-oxa-2-azaspiro[3,4]octane, 2-oxa-6-azaspiro[3.3]heptan-6-yl or 2,6- diazaspiro[3.3]heptan-6-yl or bridged heterocycles like 8-aza-bicyclo[3.2.1]octane or 2,5- diazabicyclo[2.2.2]octan-2-yl or 3,8-diazabicyclo[3.2.1]octane.
- Examples for a 9 to 11 membered aromatic heterobicycle are indole, indoline, benzofuran, benzothiophene, benzoxazole, benzisoxazole, benzothiazole, benzisothiazole, benzimidazole, benzimidazoline, quinoline, quinazoline, dihydroquinazoline, dihydroquinoline, tetrahydroquinoline, isoquinoline, tetrahydroisoquinoline, dihydroisoquinoline, benzazepine, purine or pteridine.
- the terms “9 to 10 membered aromatic heterobicyclyl” or “9 to 10 membered aromatic heterobicycle” are defined accordingly.
- Preferred compounds of formula (I) are those compounds in which one or more of the residues contained therein have the meanings given below, with all combinations of preferred substituent definitions being a subject of the present invention.
- the present invention also includes all tautomeric and stereoisomeric forms and mixtures thereof in all ratios, and their pharmaceutically acceptable salts.
- tautomerism like e.g. keto-enol tautomerism
- the individual forms like e.g. the keto and enol form, are comprised separately and together as mixtures in any ratio.
- stereoisomers like e.g. enantiomers, cis/trans isomers, conformers and the like.
- each pure form separately and any mixture of at least two of the pure forms in any ratio is comprised by formula (I) and is a subject of the present invention.
- Isotopic labeled compounds of formula (I) are also within the scope of the present invention.
- Methods for isotope labeling are known in the art.
- Preferred isotopes are those of the elements H, C, N, O and S.
- Solvates of compounds of formula (I) are also within the scope of the present invention.
- the compounds according to formula (I) contain one or more acidic or basic groups, the invention also comprises their corresponding pharmaceutically or toxicologically acceptable salts, in particular their pharmaceutically utilizable salts.
- the compounds of the formula (I) which contain acidic groups can be used according to the invention, for example, as alkali metal salts, alkaline earth metal salts or as ammonium salts.
- salts include sodium salts, potassium salts, calcium salts, magnesium salts or salts with ammonia or organic amines such as, for example, ethylamine, ethanolamine, triethanolamine or amino acids.
- Compounds of the formula (I) which contain one or more basic groups, i.e. groups which can be protonated, can be present and can be used according to the invention in the form of their addition salts with inorganic or organic acids.
- acids examples include hydrogen chloride, hydrogen bromide, phosphoric acid, sulfuric acid, nitric acid, methanesulfonic acid, p-toluenesulfonic acid, naphthalenedisulfonic acids, oxalic acid, acetic acid, tartaric acid, lactic acid, salicylic acid, benzoic acid, formic acid, propionic acid, pivalic acid, diethylacetic acid, malonic acid, succinic acid, pimelic acid, fumaric acid, maleic acid, malic acid, sulfaminic acid, phenylpropionic acid, gluconic acid, ascorbic acid, isonicotinic acid, citric acid, adipic acid, and other acids known to the person skilled in the art.
- the invention also includes, in addition to the salt forms mentioned, inner salts or betaines (zwitterions).
- the respective salts according to the formula (I) can be obtained by customary methods which are known to the person skilled in the art like, for example by contacting these with an organic or inorganic acid or base in a solvent or dispersant, or by anion exchange or cation exchange with other salts.
- the present invention also includes all salts of the compounds of the formula (I) which, owing to low physiological compatibility, are not directly suitable for use in pharmaceuticals but which can be used, for example, as intermediates for chemical reactions or for the preparation of pharmaceutically acceptable salts.
- the compounds or pharmaceutically acceptable salts thereof of the present invention may show one or more charged functional groups, wherein each negative charge is preferably compensated by a positive charge so that the molecule is neutral. Intramolecular compensation is possible as well as by other ions in form of inner and outer salts. Suitable counterions are those cited above. Accordingly, in the formulas of the present invention any remaining positive or negative charge or charges are preferably compensated by pharmaceutically acceptable negatively or positively charged counterion or counterions. Likewise any protonated form may be partly or fully deprotonated with appropriate counterion(s).
- a counterion as used herein may be any pharmaceutically acceptable ion that is suitable for neutralizing the charge of a residue or the compound of the present invention. It will be understood that a single counterion does not necessarily has the same valency as a charged residue or a charged compound of the present invention. Also two or more counterions may be used to neutralize a compound bearing a charge of higher charge valency than +1 or -1. Likewise, the other way round, also a single counterion bearing a charge of higher valency than +1 or -1 may be used to neutralize more than one compound. Preferably, the counterion is wellsoluble in aqueous liquids.
- a pharmaceutically acceptable negatively charged counterion X’ in the context of the present invention may have any charge valency. Therefore, X’ may exemplarily have a charge of -1, - 2, -3 or -4, preferably of -1 or -2. Charge may also optionally depend on ion strength and pH, respectively.
- X ⁇ may be any pharmaceutically acceptable negatively charged ion. Preferably, the ion is such well-soluble in aqueous liquids.
- X ⁇ may be selected from the group consisting of a halide anion (e.g., F’ or Cl’), acetate, phosphate, hydrogen phosphate, and a pharmaceutically acceptable carboxylate (e.g., a fatty acid carboxylate).
- the counterion typically depends on the surrounding liquids such as those comprised in the buffer the compound is dissolved in and the body fluids after injection in vivo. In vivo, extracellularly, one of the main, but not sole negatively charged counterions is Cl’.
- the terms “dye moiety”, “label” and “stain” may be understood interchangeably in the broadest sense as any moiety with the above formula that provides a visible stain.
- the dye moiety may be a fluorescent dye moiety and/or a chromatic moiety, particularly preferably the dye moiety is a fluorescent dye moiety.
- a fluorescent dye moiety as used herein may be understood in the broadest sense as any dye moiety enabling fluorescence detection.
- fluorescence detection is in a range of from 400 to 1000 nm, i.e. in the visible spectrum and in the Near Infrared (NIR) spectrum, in particular in a range of from 400 to 800 nm, i.e. in the visible spectrum.
- NIR Near Infrared
- the fluorescence signal emitted by the fluorescence dye moiety is well-distinguishable from the autofluorescence of cancer and surrounding tissue.
- Numerous fluorescent dye moieties are known in the art, and will be readily apparent to one of ordinary skill.
- the dye enables a fluorescence detection by radiation with a wavelength in a range of from with a wavelength of from 400 to 1000 nm, i.e. in the visible spectrum and in the Near Infrared (NIR) spectrum, preferably of from 600 nm to 850, preferably 700 nm to 780 nm and/or allows for a detection of fluorescence by eye and/or at a wavelength in the range of from 780 nm to 850 nm.
- NIR Near Infrared
- the dye moiety may also be chromatic, i.e., provoke a colour perception when illuminated by any light.
- chromatic effect may be provoked by absorbing light of one or more particular wavelength range(s) in the visible range (i.e., in range(s) from approximately 400 nm to approximately 800 nm) and/or by emitting light of one or more particular wavelength range(s) in the visible range.
- the colour is different from the neoplasia and the surrounding tissue intended to be examined. Therefore, a dye moiety, when not intended for fluorescence detection, is preferably not red or brown, but rather preferably blue or green.
- the chromatic dye moiety in the context of the present invention is a small-molecule dye, i.e., a dye moiety having a molecular weight (MW) of not more than 1000 Da, preferably not more than 750 Da, in particular nor more than 500 Da.
- MW molecular weight
- the dye moiety (C) is covalently linked with the at least one motif specifically binding to cell membranes of cancer cells (A) and the at least one chelator moiety (B) via (A)-XI-(B)-X2-(C).
- Such covalent conjugation to a chelator moiety (B) may be the formation of a covalent bond directly between the dye moiety (C) and the chelator moiety (B) or may be covalent linkage via a spacer, preferably via spacer.
- a spacer is of not more than 5 nm in length, preferably of not more than 2 nm in length, in particular of not more than 1 nm in length.
- the molecular distance between the motif specifically binding to cell membranes of cancer cells (A) and the dye moiety (C) is preferably not longer than 20 nm, more preferably not longer than 10 nm, in particular not longer than 5 nm.
- This may, depending on the chemical properties of the fluorescence dye moiety/moieties in the compound of the present invention and the presence of fluorophore(s) and/or quenchers on the surface of the target cells, i.e., the cell membranes of the respective cancer cells, also enable to observe effects such as fluorescence energy transfer (FRET) and/or fluorescence quenching upon binding of the compound according to the present invention to said cell membranes.
- FRET fluorescence energy transfer
- the presence of the fluorescence dye moiety/moieties also enables to conduct further examination methods based on fluorescence such as, e.g., fluorescence recovery after photobleaching (FRAP), fluorescence loss in photobleaching (FLIP). These methods may provide information on the mobility of the compound or salt thereof bound to or associated with the cell membranes of a cancer cell.
- fluorescence recovery after photobleaching FRAP
- fluorescence loss in photobleaching FLIP
- the compound or pharmaceutically acceptable salt thereof according to the present invention has a molecular weight (MW) of not more than 10 kDa, more preferably not more than 5 kDa, even more preferably not more than 3.5 kDa, even more preferably not more than 3 kDa, in particular not more than 2.5 kDa.
- MW molecular weight
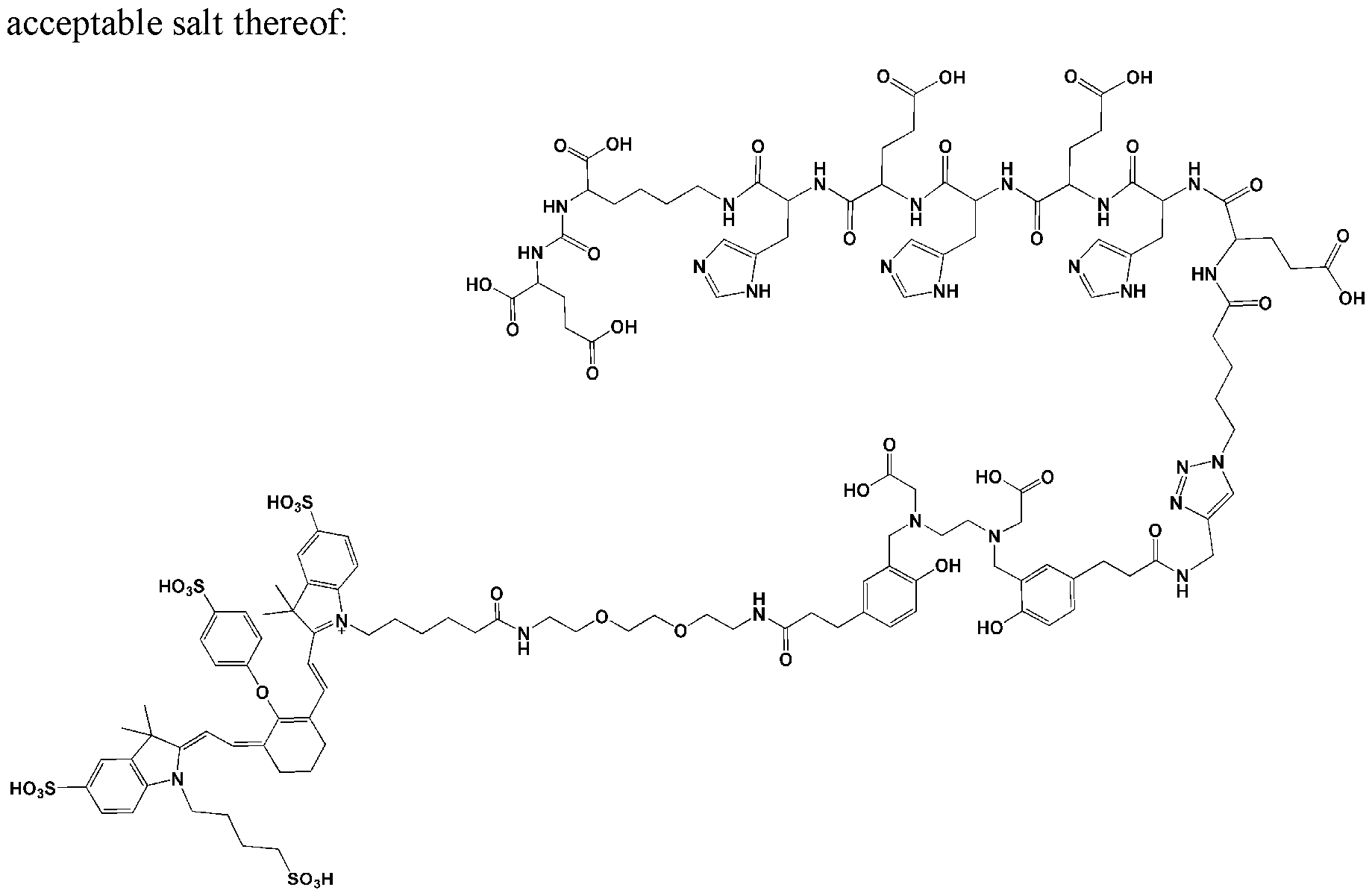
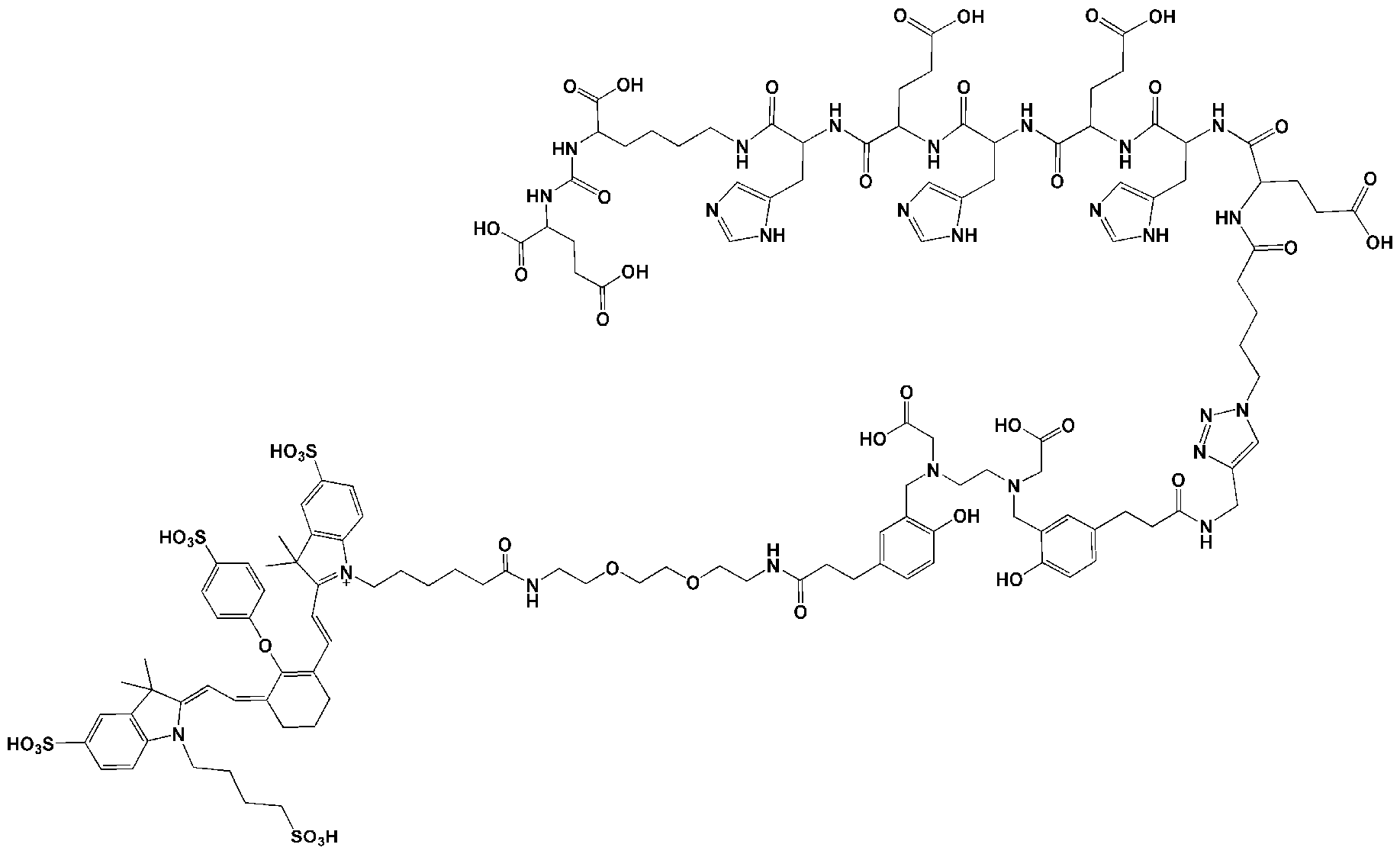
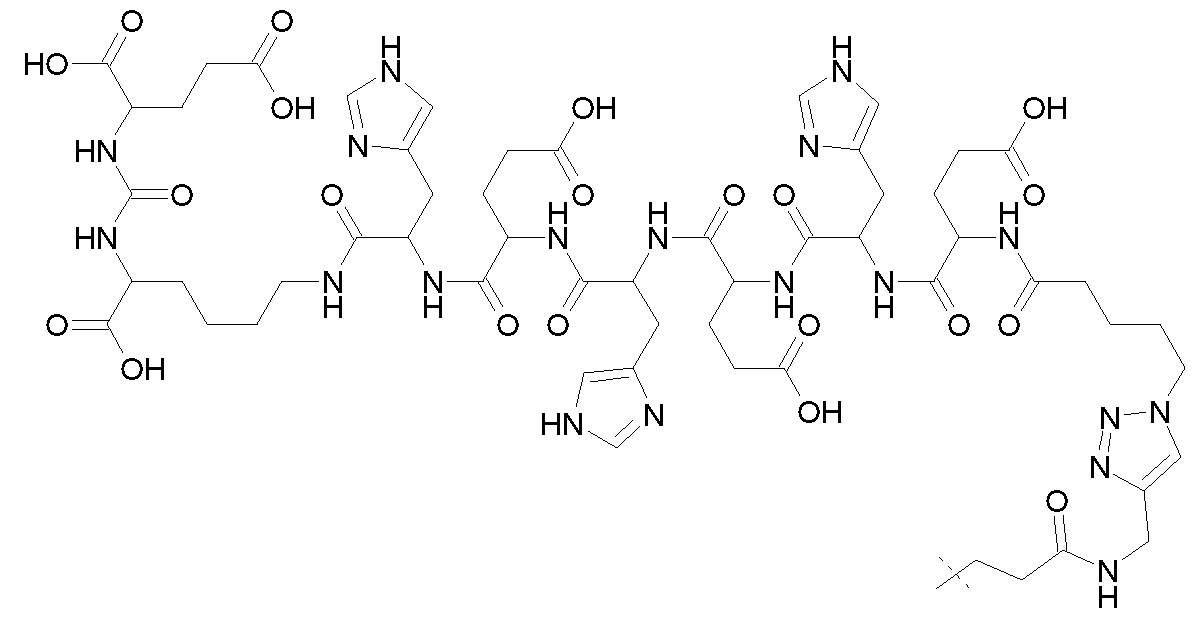
- the motif specifically binding to cell membranes of cancer cells (A) is a PSMA binding motif having the following structure: wherein Z 1 , Z 2 and Z 3 are each independently from another selected from the group consisting of -C(O)OR la , -SO 2 R la , -SO 3 R la , -SO 4 R la , -PO 2 R la , -PO 3 R la , and -PO 4 R la R 2a , wherein R la and R 2a are independently from another H or a Ci- 4 -alkyl residue (preferably the same and more preferably H); wherein a’ represents a -[CH2] 0 - residue, wherein o is an integer from 1 to 4, preferably wherein o is 3 or 4, in particular wherein o is 4.
- b’ represents a residue selected from the group consisting of -NH-, -C(O)- and -O-, in particular wherein b’ is -NH-; and wherein the wavy line indicates the conjugation site to the chelator moiety (B), conjugated via a spacer molecule xi.
- Z 1 , Z 2 , Z 3 are the same and more preferably, Z 1 , Z 2 , Z 3 are -C(O)OR la .
- the motif specifically binding to cell membranes of cancer cells is a PSMA binding motif having the following structure: wherein the wavy line indicates the conjugation site to the chelator moiety (B), conjugated via the spacer molecule xi.
- the spacers xi and X2 may be spacers with not more than 5 nm in length, preferably of not more than 2 nm in length, in particular of not more than 1 nm in length.
- the spacer xi comprises a group -AA-, wherein AA is an amino acid sequence of 4 to 8 (preferably 5 to 7, more preferably 6) naturally occurring amino acids and wherein at least two amino acids are histidine.
- naturally occurring amino acids refers to an amino acid occurring in a composition of matter occuring in nature, preferably selected from the group consisting of Ala (Alanine), Arg (Arginine), Asn (Asparagine), Asp (Aspartic acid), Cys (Cysteine), Gin (Glutamine), Glu (Glutamic acid), Gly (Glycine), His (Histidine), He (Isoleucine), Leu (Leucine), Lys (Lysine), Met (Methionine), Phe (Phenylalanine), Pro (Proline), Ser (Serine), Thr (Threonine), Trp (Tryptophan), Tyr (Tyrosine), and Vai (Valine).
- the naturally occurring amino acids may be present in racemic, D- or L-form, preferably in L-form.
- the naturally occurring amino acid is a proteinogenic amino acid.
- AA comprises three histidine amino acids.
- AA consists of histidine and glutamic acid.
- AA is represented by the formula -His-Glu-His-Glu-His-Glu-, wherein at least one of said amino acid residues is a D-amino acid, or is represented by the amino acid sequence HEHEHE, wherein all amino acids are L-amino acids (SEQ ID NO: 1).
- the compound or pharmaceutically acceptable salt thereof may comprise the aforesaid amino acid sequence bound to the rest of the molecule via C- and N-terminus and preferably is derived from the following free polypeptide sequence: EHEHEH (SEQ ID NO:2).
- the C-terminus or the N-terminus, preferably the C-terminus, of AA forms an amide bond with (A), preferably with histidine comprised in AA.
- the (A)-xi comprises the following partial structure with histidine as part of AA and xi bound to (A): wherein the dashed line indicates the attachment to the rest of AA comprised in xi.
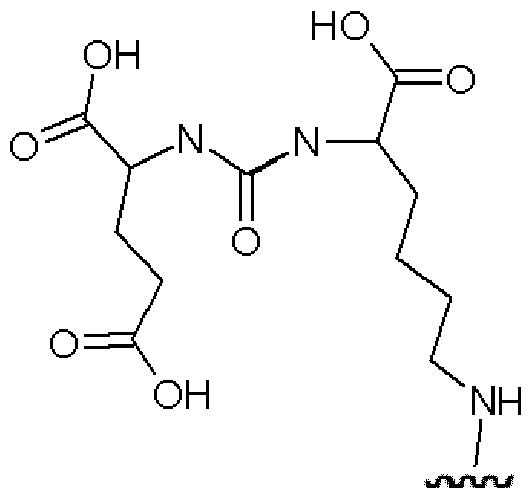
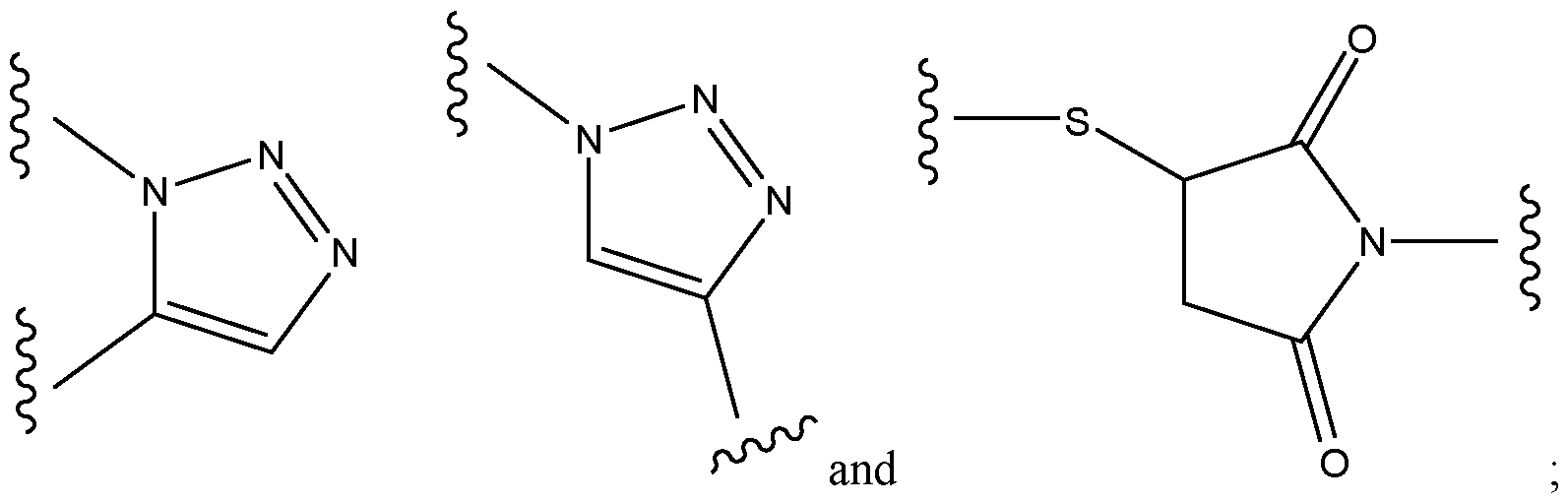
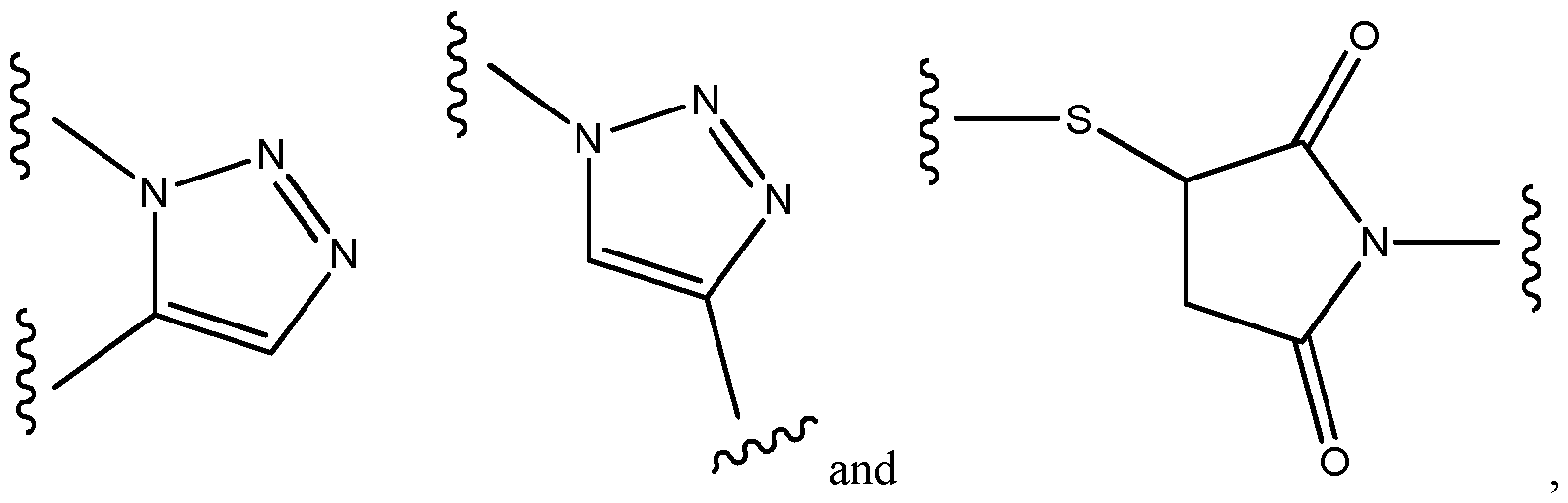
- the spacer xi bears the following structure: -AA-[b”-e-b”’]n-b””-d 1 -, wherein b” is -C(O)- or -N(H)-, preferably forming an amide bond with AA; wherein e represents a residue selected from the group consisting of an Ci-8-alkylene wherein one or more -CH2- moieties may optionally be replaced by one or more -O-, -S-, -C(O)NH- - C(O)N(CI-6 alkyl), -C(O)O-, succinimide, triazole.
- b’ is selected from the group consisting of -NH-, and -C(O)-; wherein b”” is selected from the group consisting of -C(O)-, and -NH-; and wherein b’” and b”” or a terminus of AA, preferably the N-terminus of AA, and b”” together form an amide group; wherein d 1 is -[CH2] P -, wherein p is 1 or 2, in particular 2; and wherein n is 0 or 1, preferably 0.
- b” is C(O).
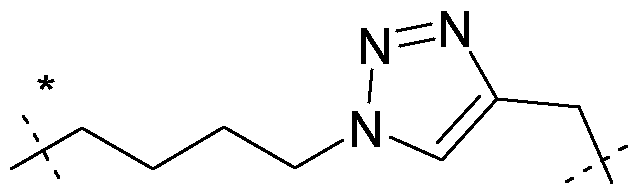
- e is without any replacement, or with one, two or three replacements, preferably one replacement, especially with triazole, preferably wherein e is a residue selected from the group consisting of an unsubstituted Cns-alkylene, Ci-8 alkylene, where one -CH2- is replaced by triazole, C4-8 alkylene (especially Ce alkylene), where one -CH2- is replaced by triazole, -CH2- (O-CH2-CH 2 )2-CH 2 -, -(CH 2 )2-(O-CH2-CH 2 )2-, -(CH 2 )3-O-CH2-CH2-O-CH 2 -, -CH 2 -O-(CH 2 )6-, -(CH 2 )2-O-(CH 2 ) 5 -, -(CH 2 )3-O-(CH 2 )4-, -(CH 2 )4-O-(CH 2 )3-, -(CH 2 ) 5 -O-(CH 2
- e is represented by the structure wherein the dashed line with the star represents attachment to b“ and the dashed line without star represents attachment to b‘“.
- b’ is -NH-.
- b’ is -C(O)-.
- n is 1.
- the spacer xi bears the following structure: -AA-[C(O)-e-NH] n -C(O)-(CH 2 )p- wherein e is C4-8 alkylene, where one -CH2- is replaced by triazole, in particular C , alkylene, where one -CH2- is replaced by triazole; wherein n is 0 or 1, in particular 1; and wherein p is 1 or 2, in particular 2.
- the spacer xi bears the following structure:
- (A)-xi is represented by the following structure:
- X2 is a spacer or a chemical single bond connecting (B) and (C).
- X2 is a spacer.
- the spacer X2 preferably is rather hydrophilic.
- the spacer X2 bears the following structure:
- d 2 is -[CH2]r-, wherein r is 1 or 2, in particular 2; and wherein e is selected from the group consisting of -C(O)-NH-, -NH-C(O)-, -C(O)-O-, -O-C(O)-, -NH-C(O)-NH-, -NH-C(S)- NH-, wherein one of the wavy lines indicates the conjugation site to d 2 and the other wavy line indicates the conjugation site to f, in particular wherein e is -C(O)-NH-;
- e is -C(O)NH-.
- each f independently represents a residue selected from the group consisting of an Ci-io-alkylene wherein one or more -CH2- moieties may optionally be replaced by -O- or -NH- , and wherein f is unsubstituted or substituted with one or more groups independently selected from the group consisting of -NH2, -COOH and R 3a , wherein R 3a is selected from the group consisting of -(CH2)2-COOH, -(CH2)4-NH2, -(CH2)4- N + (CH 3 ) 3 + X , -CH 2 -COOH,-CH 2 -SH, -CH 2 -SO 3 H, and wherein X' is a pharmaceutically acceptable negatively charged counterion; preferably wherein f is selected from the group consisting of -CH2-(O-CH2-CH2)2-CH2-, - (CH 2 )2-(O-CH2-CH 2 )2-, -(CH 2 )2-(CH2-CH2-O
- the spacer x 2 bears one of the following structures:
- t is 1 or 2, in particular 2; wherein u is an integer from 1 to 10, preferably from 1 to 3, in particular 2; wherein v is an integer from 0 to 3, in particular 2; wherein w is an integer from 0 to 2, in particular 0;
- the spacer y bears one of the following structures: -(CH2)2-C(O)-NH-(CH2)2-(O-CH2-CH 2 )2-e”-
- n’ is an integer from 1 to 3; wherein n” is an integer from 0 to 2; wherein X' is a pharmaceutically acceptable negatively charged counterion; and wherein each e” is independently selected from the group consisting of a chemical bond, -NH- C(O)-, -C(O)-NH-, -C(O)-O- and -O-C(O)-, -NH-C(O)-NH-, -NH-C(S)-NH-, -C(O)-N(CH 3 )-, -N(CH 3 )-C(O)-, -NH-C(S)-, -C(S)-NH-, wherein one of the wavy lines indicates the conjugation site to f and the other wavy line indicates the conjugation site to the at least one dye moiety (C), in particular wherein e” is - -
- X2 is represented by formula -(CH2)2-C(O)-NH-(CH2)2-(O-CH2-CH2)2-e”-.
- the labeling as spcified herein may comprise a (further) step of administration of a radioactively labeled complex of the compound of formula (I), preferably at least two days before administration of the compound of formula (I). It is, however, also envisaged to use a non-radioactively labeled complex of the compound of formula (I).
- At least one radiometal preferably selected from the group consisting of 89 Zr, 44 Sc, i n In, 90 Y, 67 Ga, 68 Ga, 177 Lu, " m Tc, 82 Rb, 64 Cu, 67 Cu, 153 Gd, 155 Gd, 157 Gd, 213 Bi, 225 Ac and 59 Fe, in particular 68 Ga.
- non-radioactively labeled complex comprises
- At least one non-radioactive metal ion wherein said metal is preferably selected from the group consisting of Zr, Sc, In, Y, Ga, Lu, Tc, Rb, Cu, Gd, Bi, Ac and Fe, in particular is Ga.
- the complex preferably comprises one or more radiometal(s), preferably one radiometal.
- a “radiometal” as used in the context of the present invention may be understood in the broadest sense as any radioactive metal or radioactive metal ion, i.e., a metal or metal ion that emits radioactive emission. It may be a metal or metal ion that is typically radioactive or a radioactive isotope of a metal that also has non-radioactive isotopes.
- a radiometal may be a radioactive isotope of gallium (Ga) (e.g., 67 Ga, 68 Ga), copper (e.g., 64 Cu, 67 Cu), iron (e.g., 59 Fe), zirconium (e.g., 89 Zr), scandium (e.g., 44 Sc), indium (e.g., H 1 In), yttrium (e.g., 90 Y) rubidium (e.g., 82 Rb), cobalt (e.g., 60 Co), lutetium (e.g., 177 Lu), gadolinium (e.g., 153 Gd, 155 Gd, 157 Gd), bismuth (e.g., 213 Bi), strontium (e.g., 90 Sr), actinium (e.g., 225 Ac), or technetium (e.g., 99m Tc).
- Ga gallium
- copper e.g., 64 Cu, 67 Cu
- the radiometal is a radioactive isotope of 67 Ga, 68 Ga, i n In, 90 Y, 177 Lu, 64 Cu, 67 Cu, 153 Gd, 155 Gd, 157 Gd, 89 Zr, 44 Sc, " m Tc, 213 Bi, 225 Ac, 59 Fe or 82 Rb, more preferably 68 Ga, 64 Cu, 89 Zr, 44 Sc, or 82 Rb, even more preferably 68 Ga or 64 Cu, in particular 68 Ga.
- chelator moieties suitable for complexing each of the aforementioned radiometals and may select the chelator moiety accordingly.
- a chelator moiety suitable for complexing " m Tc or 82 Rb may or may not differ from that suitable for complexing 68 Ga or 64 Cu.
- the radiometal may be obtained commercially of, may be obtained from nature or may be obtained from a cyclotron.
- the radiometal is obtained from a gallium-68 generator or a cyclotron, in particular from a gallium-68 generator or a cyclotron that is localized near the site where the composition according to the present invention is mixed and where optionally also the diagnosis in vivo and/or in vitro is performed.
- the radiometal 68 Ga is obtained from a gallium-68 generator, thus, a device used to extract the positronemitting isotope 68 Ga of gallium from a source of decaying germanium-68.
- the parent isotope 68 Ge is known to have a half-life of 271 days and may thus be shipped to the site where the gallium-68 generator is located.
- the radiometal is such that has a half-life of no longer than four days, more preferably of no longer than one day, even more preferably no longer than 12 h, even more preferably, not more than 6 h, even more preferably not more than 3 h, even more preferably not more than 2.5 h, even more preferably not more than 120 min, even more preferably not more than 100 min, even more preferably not more than 80 min, in particular not more than 70 min.
- the radiometal may be obtained from any source suitable for this purpose.
- the radiometal may be obtained and isolated from nature or artificially be generated such as, e.g., 68 Ga from a gallium-68-generator. The person skilled in the art will know how to obtain the respective radiometal.
- the non-radioactive metal may be any metal ion not emitting radiactivity deemed appropriate by the skilled person, selected according to the intended use.
- the non-radioactive metal is at least one non-radioactive isotope of a radiometal as specified herein above.
- the chelator moiety (B) is a chelator suitable to complex 68 Ga, " m Tc or 82 Rb, in particular suitable to complex 68 Ga in aqueous environment.
- Gallium-68 ( 68 Ga) has a half-life of approximately 68 minutes and is thus rather inconvenient for longer transports. Therefore, it may typically be generated nearby the site where it is complexed with the compound of the present invention or a pharmaceutically acceptable salt thereof and administered to a patient in vivo and/or a sample in vitro.
- the chelator moiety (B) is a 68 Ga-chelator moiety, preferably a 68 Ga-chelator moiety.
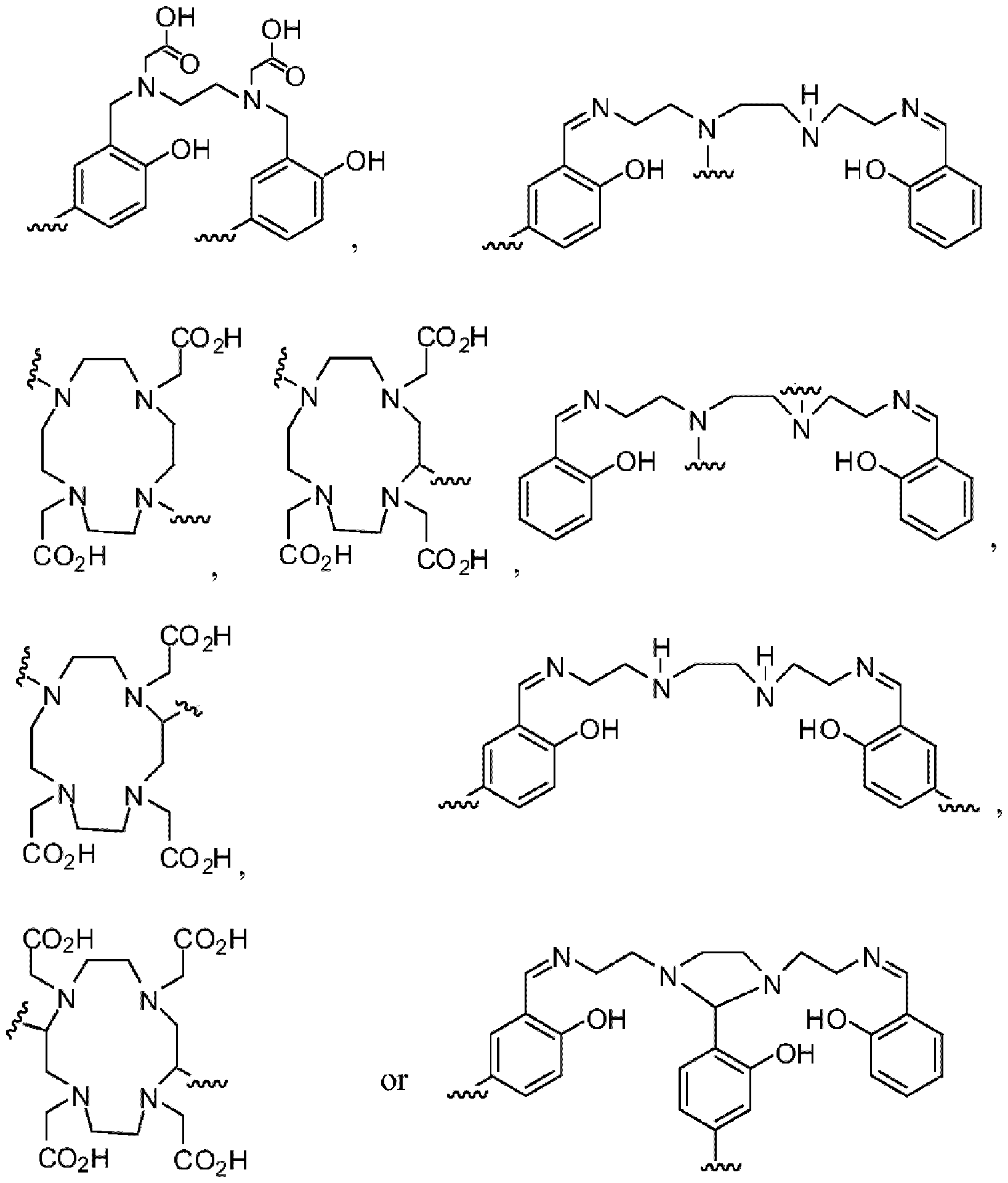
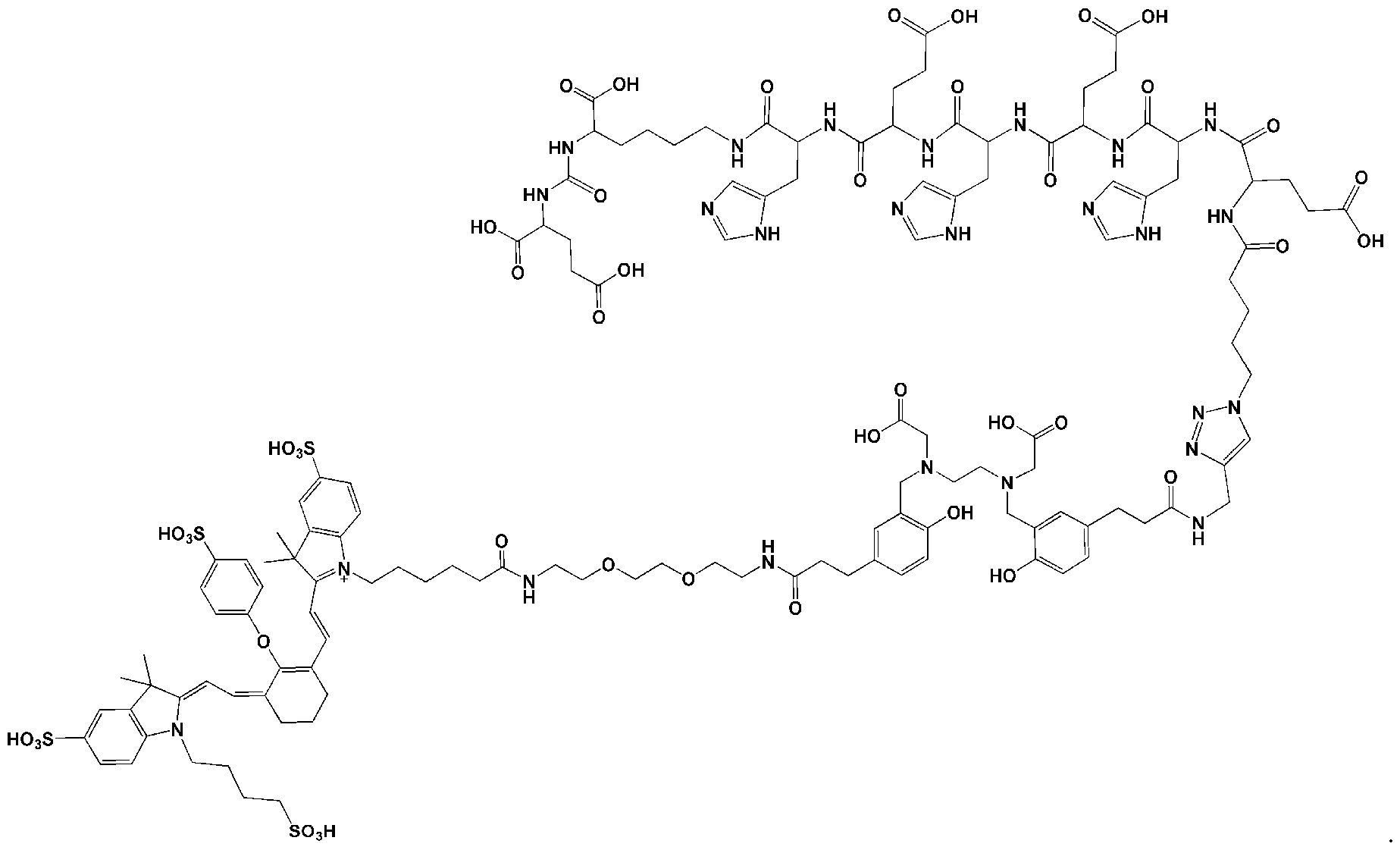
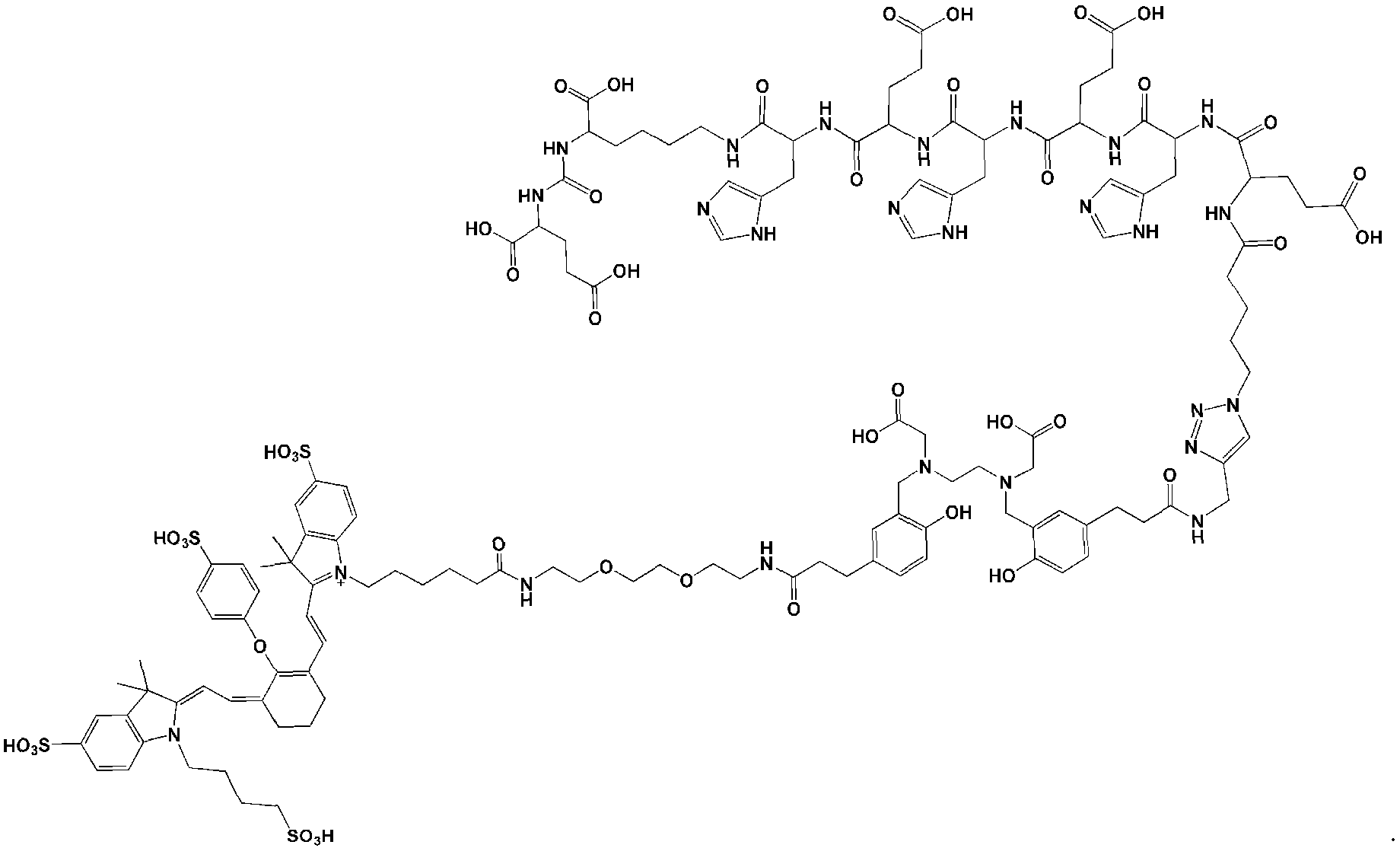
- the chelator moiety (B) is preferably selected from the following group:
- one of the wavy lines indicates the conjugation site to the at least one motif specifically binding to cell membranes of cancer cells (A) via spacer xi as defined herein, and the other wavy line indicates the conjugation site to the dye moiety (C), preferably via the spacer X2 as defined herein.
- the chelator moiety is When this chelator is used, b”” preferably is -C(O)-, d 1 and d 2 preferably are each -(CH2)2-, and e’ preferably is -C(O)-NH or -C(O)-O-, especially -C(O)NH-. Therefore, the chelator moiety is conjugated with two -C(O)-(CH2)2- # moieties, wherein these are conjugated at the position indicated by “#” with the binding sites of the aforementioned chelator moiety indicated by the wavy lines. Then, this residue is also designated as “HBED-CC”.
- (A)-XI-(B)-X2- is represented by the following formula:
- 68 Ga-chelator moieties may also serve as structures complexing one or more other radiometal(s) such as, e.g., 64 Cu, in particular in aqueous environment of approximate neutral pH.
- neither the motif specifically binding to cell membranes of cancer cells (A) nor the complexed radiometal nor the chelator moiety (B) quench the intensity of the fluorescence signal obtainable from the dye moiety (C) at its emission maximum in an aqueous environment of approximately neutral pH (i.e., pH 6-8, in particular 6.5-7.5) by more than 50%.
- the dye moiety (C) is suitable for emit light in an aqueous environment of approximately neutral pH, i.e., pH 6-8, in particular 6.5-7.5, in particular pH 7.0-7.5.
- the dye moiety (C) is a fluorescent dye moiety having an emission maximum in the range from 400 nm to 1000 nm, as described above.
- the dye moiety (C) has preferably the formula wherein
- X 2 and X 3 are independently selected from the group consisting of O, S, Se, N(R 5 ), and C(R 6 R 7 ), preferably both are C(CHs)2;
- Y is a linker connecting the two moieties of (C) and permitting electron delocalization between said moieties, wherein Y optionally comprises a group (L-)cZ 0 ; a and b are independently selected from the group consisting of 1, 2, and 3; each R 1 and each R 2 is independently (L-) C Z, (L-) C Z° or H; and two adjacent R 1 and/or two adjacent R 2 can also form an aromatic ring, which is optionally substituted with one or more (L-)cZ or (L-)cZ°;
- R 3 , R 4 , R 5 , R 6 , R 7 , R 9 are independently selected from the group consisting of (L-)cZ, (L-) C Z°, and H; each c is independently 0, or 1 ; each L is independently NHC O) 1 , or a Ci -io alkylene group, which is optionally interrupted and/or terminated by one or more of -O-, -S-, -C(O)-, -C(O)O-, -OC(O)-, -C(O)NH-, -NHC(O)O-, and T 1 ;
- T 1 is phenyl, naphthyl, indenyl, indanyl, tetralinyl, decalinyl, adamantyl, C3-7 cycloalkyl, 3 to 7 membered heterocyclyl, or 7 to 11 membered heterobicyclyl, wherein T 1 is optionally substituted with one or more substituents selected from the group consisting of halogen, CN, C(O)R 8 , COOR 8 , OR 8 , C(O)N(R 8 R 8a ), S(O) 2 N(R 8 R 8a ), S(O)N(R 8 R 8a ), S(O) 2 R 8 , N(R 8 )S(O) 2 N(R 8a R 8b ), SR 8 , N(R 8 R 8a ), NO 2 ; OC(O)R 8 , N(R 8 )C(O)R 8a , N(R 8 )S(O) 2 R 8a
- R 8 , R 8a , R 8b are independently selected from the group consisting of H, or C1-6 alkyl, wherein C1-6 alkyl is optionally substituted with one or more halogen, which are the same or different;
- Z° is a chemical bond connecting (C) to x 2 or to (B) in case x 2 is a chemical single bond; provided that one of R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 9 is (L-) C Z° or that Y comprises (L-) C Z°.
- one of the residues R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 serve as connecting group, atom or bond (L-) C Z° of dye moiety (C) to x 2 or (B).
- R 3 or R 4 represent (L-) C Z°. More preferably, R 3 is (L-) C Z°.
- X 1 and X 4 are the same and preferably C(R 6 ), more preferably CH.
- R 9 is H.
- Y does not comprise (L-) C Z° and preferably Y is wherein g is 1, 2, 3, or 4 (preferably 2 or 3, more preferably 3) and each R 9a is (L-) C Z, or H; and two R 9a can also form a carbocyclic ring having 5, 6, or 7 carbon atoms or a 4 to 7 membered heterocylic ring; each c is independently 0, or 1 ; each L is independently NHC O) 1 , or a Ci -io alkylene group, which is optionally interrupted and/or terminated by one or more of -O-, -S-, -C(O)-, -C(O)O-, -OC(O)-, -C(O)NH-, -NHC(O)O-, and T 1 ;
- T 1 is phenyl, naphthyl, indenyl, indanyl, tetralinyl, decalinyl, adamantyl, C3-7 cycloalkyl, 4 to 7 membered heterocyclyl, or 7 to 11 membered heterobicyclyl, wherein T 1 is optionally substituted with one or more substituents selected from the group consisting of halogen, CN, C(O)R 8 , COOR 8 , OR 8 , C(O)N(R 8 R 8a ), S(O) 2 N(R 8 R 8a ), S(O)N(R 8 R 8a ), S(O) 2 R 8 , N(R 8 )S(O) 2 N(R 8a R 8b ), SR 8 , N(R 8 R 8a ), NO 2 ; OC(O)R 8 , N(R 8 )C(O)R 8a , N(R 8 )S(O) 2 R 8a
- R 8 , R 8a , R 8b are independently selected from the group consisting of H, or C1-6 alkyl, wherein C1-6 alkyl is optionally substituted with one or more halogen, which are the same or different; each Z is independently H, halogen, CN, C(O)R 8 , C(O)OR 8 , C(O)O’ OR 8 , C(O)N(R 8 R 8a ), S(O) 2 OR 8 , S(O) 2 O’, S(O) 2 N(R 8 R 8a ), S(O)N(R 8 R 8a ), S(O) 2 R 8 , S(O)R 8 , N(R 8 )S(O) 2 N(R 8a R 8b ), SR 8 , N(R 8 R 8a ), NO 2 ; P(O)(OR 8 ) 2 , P(O)(OR 8 )O’, OC(O)R 8 , N(R 8 )C(O)R 8
- a and b are the same and 2, preferably wherein two adjacent R 1 and two adjacent R 2 form a phenyl ring.
- (L-) C Z is C1-10 alkylene, preferably C3-7 alkylene, more preferably C4 alkylene, c is 1 and Z is SCh'.
- the dye moiety (C) is a fluorescent dye moiety selected from the group consisting of the following structures: wherein X' is a pharmaceutically acceptable negatively charged counterion; wherein Y + is a pharmaceutically acceptable positively charged counterion; and wherein the wavy line indicates the conjugation site to the rest of the compound of the present invention.
- the dye is IRDye800CW.
- a pharmaceutically acceptable negatively charged counterion X' may be understood in the broadest sense as laid out above.
- Y + may have any valency. Therefore, Y + may exemplarily have a charge of +1, +2, +3 or +4, preferably of +1 or +2. Y + may be any pharmaceutically acceptable positively charged ion. Preferably, the ion is such well-soluble in aqueous liquids.
- Y + may be selected from the group consisting of a cation of an alkali metal (e.g., Na + , K + , Li + ), a cation of an alkaline earth metal (e.g., Mg 2+ , Ca 2+ ), Al 3+ , NH4 + , H + and a cation of an organically bound amine.
- the counterion typically depends on the surrounding liquids such as those comprised in the buffer the compound is dissolved in and the body fluids after injection in vivo. In vivo, extracellularly, one of the main, but not sole positively charged counterions is Na + .
- the wavy line indicates the conjugation site to the spacer X2. More preferably, the wavy line indicates the conjugation site to e”.
- the compound of the present invention has the following chemical structure:
- the compound of formula (I) may be obtained from rational chemical synthesis.
- Exemplary routes for the preparation of compounds or pharmaceutically acceptable salts thereof of the present invention are described in US 2015/0110715 Al.
- labeling agent is understood by the skilled person.
- the term relates to a compound labeling cancerous tissue, more preferably, specifically labeling cancerous tissue, as specified herein below.
- specific labeling of a cancerous tissue preferably relates to a labeling which enables differentiation between cancerous tissue and non-cancerous tissue, such as e.g. cancer-adjacent tissue.
- said differentiation is enabled in vitro and/or in vivo, more preferably in vivo.
- the labeling enables differentiation between adjacent cancerous and non-cancerous tissues, e.g. in an operation area.
- specific labeling does not necessarily have to enable differentiation between cancerous tissue and any non- cancerous tissue in a subject; it, preferably, is sufficient if cancerous tissue can be differentiated from non-cancerous tissue in the vicinity of cancerous tissue.
- the differentiation may preferably be between cancerous tissue and non-cancerous tissue in the abdominal cavity, more preferably within at most 10 cm, more preferably at most 5 cm, even more preferably at most 2 cm from a cancerous tissue.
- it preferably is not required that differentiation is enabled from easily identifiable intact structures in the subject, e.g. the kidney, the liver, and the like.
- the differentiation is preferably made by visual inspection, e.g.
- specific labeling is labeling of cancerous tissue, preferably of cancer cells, more intensely by a factor of at least 2, more preferably at least 5, even more preferably at least 10, still more preferably at least 25, more preferably at least 100, compared to non-cancerous tissue.
- the labeling agent preferably has an affinity for cancerous tissue, preferably for cancer cells, being at least 5fold, more preferably at least lOfold, even more preferably at least lOOfold, still more preferably at least 250fold, most preferably at least lOOOfold higher than for non-cancerous tissue.
- the labeling agent specifically labels PSMA expressing cancerous tissue, preferably PSMA expressing cancer cells, all as specified herein below;
- the dissociation constant (Ka) of the labeling agent in its binding to PSMA is preferably at most 10' 6 mol/L, more preferably at most 10' 7 mol/1, most preferably at most 10' 8 mol/L; also preferably, the Ka of the labeling agent in its binding to any non-PSMA cell constituent preferably is at least 10' 6 mol/L, more preferably at least 10' 5 mol/1, most preferably at least 10' 4 mol/L.
- the dissociation constant is determined by a method known to the skilled person, e.g.
- the labeling agent may be the compound having the chemical structure of formula (I) or a metal chelate thereof, in particular a radiometal chelate thereof.
- the labeling agent is a compound having the chemical structure of formula (I) or a pharmaceutically acceptable salt thereof, i.e. preferably is said compound not comprising a chelated metal.
- detecting will preferably be based on the physical properties of the dye moiety in case the labeling agent is not a metal chelate or is administered as a non-metal chelate; in case the labeling agent is a metal chelate or is administered as a metal chelate, detecting may also be based on physical properties of the chelated metal, exclusively or in addition, preferably in addition.
- the labeling agent may e.g. be a chelate of a radiometal and detecting may be based on fluorescence of the dye moiety, e.g. in areas easily accessible, and may be assisted by detection of radioactivity from the chelated radiometal to detect small numbers of residual cancer cells and/or cancer cells in areas which are not or not easily accessible.
- cancer relates to a disease of an animal, including man, characterized by uncontrolled growth by a group of body cells (“cancer cells”). This uncontrolled growth may be accompanied formation of a cell mass of cancer cells (tumor), by intrusion into and destruction of surrounding tissue (infiltration) and possibly spread of cancer cells to other locations in the body (metastasis).
- cancer is also included by the term cancer is a recurrence of a cancer (relapse).
- the cancer is a solid cancer, including a primary tumor, a metastasis, and a relapse thereof.
- the cancer may, however, also be a nonsolid cancer.
- the cancer is a prostate-specific membrane antigen (PSMA) expressing cancer, preferably a PSMA overexpressing cancer.
- PSMA prostate-specific membrane antigen
- the cancer is a cancer comprising PSMA-expressing neovasculature.
- the cancer preferably is selected from the list consisting of prostate cancer, breast cancer, kidney cancer, bladder cancer, ovarian cancer, or non-small cell lung cancer (NSCLC), more preferably is prostate cancer, still more preferably PSMA overexpressing prostate cancer.
- cancerous tissue relates to any tissue known or suspected to comprise cancer cells as specified herein above.
- the fraction of cancer cells in the cancerous tissue is at least 10%, preferably at least 20%, more preferably at least 30%, even more preferably at least 40%, most preferably at least 50%.
- the cancerous tissue preferably is a primary tumor, an infiltration, a metastasis, a relapse, or a lymph node, or a sample thereof.
- detecting is in principle understood by the skilled person and preferably relates to establishing the presence of cancerous tissue, preferably of cancer cells, in a subject and/or of cancer cells in a sample.
- determining is identifying cancerous tissue in a subject, preferably in a surgical operation zone.
- detecting is differentiating between cancerous tissue and non-cancerous tissue.
- the detecting may be a step aiding a surgeon in deciding which tissue or parts thereof shall be excised and which shall not.
- labeling by the labeling compound may be, but preferably is not, the only basis for said decision.
- the decision takes into account further parameters such as tissue morphology, tissue density, proximity to the primary tumor, location in relation to afferent or efferent blood and/or lymph vessels, vascularization, and the like.
- the detecting may comprise additional steps and/or activities, such as illuminating tissue suspected to be cancerous tissue with radiation of a wavelength absorbed and/or causing radiation from the labeling compound, preferably causing fluorescence.
- the labeling compound is e.g.
- PSMA- 914 determining preferably further comprises illuminating tissue suspected to be cancerous tissue by radiation with a wavelength of from 600 nm to 850, preferably 700 nm to 780 nm, and/or detection of radiation, by eye and/or at a wavelength in the range of from 780 nm to 850 nm, preferably comprises illuminating tissue suspected to be cancerous tissue by radiation with a wavelength of about 774 nm, and/or detection of radiation of a wavelength of about 789 nm, in particular in case the dye moiety is IRDye800CW, and/or the labeling compound is PSMA- 914.
- the selection of additional steps will depend on a variety of parameters, including in particular the dye moiety used in the labeling compound, the mode of administration of the labeling compound, the type of determining (e.g. in vivo or in vitro), and the like.
- subject relates to a vertebrate animal, preferably a mammal, in particular a livestock, companion, or laboratory animal. Most preferably, subject is a human. Preferably, the subject has been diagnosed to suffer from cancer, is suspected to suffer from cancer, or is known to be at risk of suffering from cancer, in particular a hereditary form of cancer. Preferably, the subject is a male, more preferably a male suffering from prostate cancer, even more preferably with a Gleason score of at least 5, more preferably at least 6, even more preferably at least 7, most preferably at least 8. Preferably, the cancer was diagnosed to warrant, preferably to require, surgical removal, preferably by partial or complete prostatectomy.
- labeling dose relates to a dose of the labeling compound enabling labeling cancerous tissue, preferably, specifically labeling cancerous tissue as specified herein above, more preferably enabling differentiation between cancerous tissue and non-cancerous tissue as specified herein above.
- the labeling dose is at least 200 pg, preferably at least 500 pg, more preferably at least 1 mg of labeling compound per subject.
- the labeling dose is of from 0.2 mg to 100 mg, more preferably is of from 0.5 to 25 mg, still more preferably is of from 1 mg to 10 mg, most preferably is of from 1 mg to 5 mg, of said labeling compound per subject.
- the labeling dose may e.g.
- the aforesaid labeling doses preferably are labeling doses for human subjects and, preferably, for PSMA-914.
- the labeling dose is administered independent of the subject's body mass, i.e. is determined without taking into account the subject's body mass.
- the labeling dose preferably is a unit dose for all subjects, preferably for all adult subjects also preferably for subjects in a body mass range of from 50 kg to 200 kg.
- the labeling dose may, however, also be adjusted to a subject's body mass; in such case, the dose preferably is of from 3 pg/kg body mass to 300 pg/kg body mass, more preferably of from 5 pg/kg body mass to 100 pg/kg body mass, even more preferably of from 7,5 pg/kg body mass to 50 pg/kg body mass, still more preferably about 15 pg/kg body mass.
- the dose is a single dose, i.e. exactly one dose, preferably administered within a time frame of at least twelve hours, preferably at least one day, more preferably at least two days; thus, in the aforesaid time frame, preferably no further dose of labeling compound is administered.
- said labeling dose is administered to the patient at of from 0.25 h to 48 h before determining, preferably before start of surgery. More preferably, said labeling dose is administered to the patient at of from 0.5 h to 24 h before determining, still more preferably of from 0.75 h to 12 h before determining, even more preferably at about 1 h before determining, most preferably 1 h before determining, preferably before start of surgery.
- the labeling further comprises fine-tuning the labeling dose.
- the labeling may comprise a further step of administration of a radioactively labeled complex of the labeling compound, preferably at least two days before administration of the labeling compound, and further comprises increasing the labeling dose of the labeling compound in case the signal of said radioactively labeled complex of the labeling compound in tumor or relapse tissue is found to be weak compared to a reference.
- the radioactively labeled complex comprises
- a radiometal preferably selected from the group consisting of 89 Zr, 44 Sc, i n In, 90 Y, 67 Ga, 68 Ga, 177 Lu, " m Tc, 82 Rb, 64 Cu, 67 Cu, 153 Gd, 155 Gd, 157 Gd, 213 Bi, 225 Ac and 59 Fe, in particular 68 Ga.
- the dose of the labeling compound is at least 20fold, preferably at least 30fold, the dose of the dose of the radioactively labeled complex of the labeling compound preferably in case the signal of said radioactively labeled complex in tumor or relapse tissue is found to be above or within the average of a reference.
- the signal of said radioactively labeled complex is determined on a primary tumor or part thereof; however, other methods of determination may be used, e.g. detectability of metastatic lymph nodes in a PET scan.
- the radioactively labeled complex of the labeling compound is preferably administered at a dose of from 2 pg per subject to 50 pg per subject, preferably of from 10 pg per subject to 30 pg per subject.
- the dose of the radioactively labeled derivative is preferably adjusted or re-adjusted based on its specific activity, preferably such that a pre-determined dose of radioactivity is administered to the subject.
- the dose of the labeling compound preferably is at least 20fold, preferably at least 30fold, the dose of the dose of the radioactively labeled complex of the labeling compound, preferably in case the signal of said radioactively labeled complex of the labeling compound in tumor or relapse tissue is found to be above or equal to a reference.
- the dose is least 50fold, preferably at least 75fold the dose of the dose of the radioactively labeled complex of the labeling compound, preferably in case the signal of said radioactively labeled complex of the labeling compound in tumor or relapse tissue is found to be below a reference.
- the dose is at most 30000fold, preferably at most lOOOOfold, more preferably at most lOOOfold the dose of the dose of the radioactively labeled complex of the labeling compound.
- the aforesaid ratios are determined on a molar basis.
- the term “reference” refers to a signal or a value representing said signal, i.e. data correlating with a labeling intensity of a cancerous tissue with a radioactively labeled complex of the labeling compound.
- the reference may e.g. be a value of a radioactivity in a tissue, a blackening of a photographic film, and intensity value in a scintigram or a PET scan, or the like.
- Suitable references are known to the skilled person and may in particular be derived from a subject or a population of subjects known to have had specifically labeled cancerous tissue at a standard dose as specified herein above, or may be the average of a population of subjects; thus, the reference may in particular be obtained from a multitude of primary and/ or relapse prostate cancer samples. How to calculate a suitable reference value, in an embodiment, the average or median, is well known in the art.
- the population of subjects referred to before shall comprise a plurality of subjects, preferably at least 5, more preferably at least 10, even more preferably at least 50, more preferably at least 100, subjects.
- the subject to be diagnosed by the method of the present invention and the subjects of the plurality of subjects used to derive a reference from are preferably of the same species.
- the skilled person knows howto establish a reference value, which may be a cut-off vale, an upper limit of normal, a median or average, a reference range, or the like.
- the labeling compound in particular PSMA-914
- the labeling compound can be administered at a dose which enables specific in vivo labeling of cancerous tissue, in particular prostate cancer tissue and, thereby, enables reliable excision of cancer tissue e.g. in surgery.
- cancerous tissue comprising a low fraction of cancer cells, such as metastases and in particular lymph nodes, can be specifically labeled, thus making conservative surgery possible while ensuring complete removal of cancer cells.
- the present invention further relates to a labeling compound for use for in vivo labeling of cancerous tissue in a subject, said labeling comprising administration of a labeling dose of the labeling compound to said subject; and to a labeling compound for use in a method of diagnosing of cancerous tissue in a subject, wherein said method comprises administration of a labeling dose of the labeling compound to said subject.
- diagnosis refers to assessing the probability according to which a subject is suffering from a disease or condition or is at risk of developing a disease or condition. Accordingly, the method provides an aid for diagnosis since it might be necessary to further strengthen or confirm said diagnosis by, e.g., a medical practitioner. In particular, as will be understood by those skilled in the art, such an assessment, although preferred to be, may usually not be correct for 100% of the subjects to be diagnosed. The term, preferably, requires that a statistically significant portion of subjects can be identified as suffering from the disease.
- Whether a portion is statistically significant can be determined without further ado by the person skilled in the art using various well known statistic evaluation tools, e.g., determination of confidence intervals, p-value determination, Student's t-test, Mann- Whitney test, etc.. Details are found in Dowdy and Wearden, Statistics for Research, John Wiley & Sons, New York 1983.
- Preferred confidence intervals are at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%.
- the p-values are, in an embodiment, 0.2, 0.1, 0.05. It will be understood, moreover, that the methods of the present invention essentially provide an aid for diagnosis and may be included into or supplemented by other diagnostic measures.
- Diagnosing according to the present invention includes monitoring, confirmation, and classification of the relevant disease or its symptoms.
- Monitoring relates to keeping track of an already diagnosed disease, or a complication, e.g. to analyze the progression or regression of the disease, the influence of a particular treatment on the progression of disease or complications arising during the disease period or after successful treatment of the disease.
- Confirmation relates to the strengthening or substantiating a diagnosis already established using other indicators or markers.
- Classification relates to allocating the diagnosis according to the strength or kind of symptoms into different classes, e.g. the stages for prostate carcinomas.
- diagnosing cancer comprises identification of cancerous tissue, preferably of cancer cells, preferably in vivo in a subject, more preferably comprises intraoperative identification of cancerous tissue.
- diagnosing comprises identification of primary tumor, relapse, metastases and/or lymph nodes comprising cancer cells, more preferably comprises identification of metastases and/or lymph nodes comprising cancer cells, preferably is intraoperative identification of metastases and/or lymph nodes comprising cancer cells.
- the present invention also relates to a method for labeling cancerous tissue in a subject, comprising administering a labeling dose of the labeling compound to said subject; to a method for identifying cancerous tissue in a subject comprising (a) labeling cancerous tissue by administering a labeling dose of the labeling compound to said subject; and (b) identifying labeled cancerous tissue in situ; as well as to a method for removing cancerous tissue from a subject, said method comprising (a) labeling cancerous tissue by administering a labeling dose of the labeling compound to said subject; (b) identifying labeled cancerous tissue in situ; and (c) removing said cancerous tissue.
- the present invention also relates to a labeling compound accordig to formula (I) for use in a method of removing a cancerous tissue from a subject, said method comprising
- the method of removing a cancerous tissue is an in vivo method and may comprise further steps not explicitly mentioned, e.g. diagnosing said cancer tissue, preferably according to a method as specified herein, and/or additional curative and/or preventive method steps, such as administration of radiation, chemotherapy, immunotherapy, and the like before, concomitant to, and/or after said removing of cancerous tissue.
- additional curative and/or preventive method steps such as administration of radiation, chemotherapy, immunotherapy, and the like before, concomitant to, and/or after said removing of cancerous tissue.
- the steps of (a) labeling cancerous tissue and (b) identifying labeled cancerous tissue have been described herein above.
- labeled cancerous tissue may further be identified in vitro, e.g. in histopathological analysis of excised cancerous tissue.
- the method further comprises step (c) removing said cancerous tissue.
- step (c) comprises or is surgical removal of cancerous tissue.
- the present invention also relates to a use of a labeling compound having the structure of formula (I) as specified herein above in the manufacture of a diagnostic composition for labeling cancerous tissue, preferably in vivo labeling cancerous tissue, wherein said diagnostic composition comprises a labeling dose of said labeling compound.
- the present invention also relates to a diagnostic composition
- a diagnostic composition comprising of from 0.2 mg to 100 mg, preferably is of from 0.5 to 25 mg, more preferably is of from 1 mg to 10 mg, most preferably is of from 1 mg to 5 mg of a labeling compound having the structure of formula (I) as specified herein above.
- diagnostic composition is, in principle, known to the skilled person. As referred to herein, the term relates to any composition of matter comprising at least the specified labeling compound as diagnostically active compound(s) and one or more excipient.
- the diagnostically active compound(s) can be present in liquid or dry, e.g. lyophilized, form. It will be appreciated that the form and character of the pharmaceutically acceptable excipient, e.g. carrier or diluent, is dictated by the amount of active ingredient with which it is to be combined, the route of administration, and other well-known variables.
- the excipient(s) must be acceptable in the sense of being compatible with the other ingredients of the formulation and being not deleterious to the recipient thereof.
- the excipient employed may include a solid, a gel, or a liquid.
- solid carriers are lactose, terra alba, sucrose, talc, gelatin, agar, pectin, acacia, magnesium stearate, stearic acid and the like.
- liquid carriers are phosphate buffered saline solution, physiological saline, Ringer's solutions, dextrose solution, and Hank's solution, syrup, oil, water, emulsions, various types of wetting agents, and the like.
- the carrier or diluent may include time delay material well known to the art, such as glyceryl mono-stearate or glyceryl distearate alone or with a wax.
- the excipient may in particular be water, an aqueous buffer (e.g., a Hepes, Tris, or phosphate buffer, a phosphate buffer), a pharmaceutically acceptable organic solvent (e.g., dimethyl sulfoxide (DMSO), ethanol, vegetable oil, paraffin oil) or a combination of two or more thereof.
- aqueous buffer e.g., a Hepes, Tris, or phosphate buffer, a phosphate buffer
- a pharmaceutically acceptable organic solvent e.g., dimethyl sulfoxide (DMSO), ethanol, vegetable oil, paraffin oil
- the pharmaceutically acceptable carrier may contain one or more detergents, one or more foaming agents (e.g., sodium lauryl sulfate (SLS)/ sodium doceyl sulfate (SDS)), one or more coloring agents (e.g., TiCh, food coloring), one or more vitamins, one or more salts (e.g., sodium, potassium, calcium, zinc salts), one or more humectants (e.g., sorbitol, glycerol, mannitol, propylenglycol, polydextrose), one or more enzymes, one or more preserving agents (e.g., benzoic acid, methylparabene), one or more texturing agents (e.g., carboxymethyl cellulose (CMC), polyethylene glycol (PEG), sorbitol), one or more emulsifiers, one or more bulking agents, one or more glacing agents, one or more separating agents, one or more antioxidants, one or more herbal and plant extracts, one or more
- the diagnostic composition will typically be formed by contacting the aforementioned component with another by any way known in the art.
- the diagnostic composition is formed, thus, the above-referenced components are contacted with another prior to being administered to a patient.
- the excipient(s) is/are preferably selected so as not to affect the biological activity of the combination.
- the diagnostic composition is, preferably, administered systemically, preferably orally or parenterally, e.g. by intravenous administration, or is administered topically, preferably intra- tumorally; in case of cancer diagnostics, topical administration may be intratumoral or peritumoral, and/or topical at a site of tumor excision. Administration may, however, also be into a blood vessel, typically an artery, afferent to an intended site of effect, such as a tumor. However, depending on the nature of the formulation and the desired therapeutic application, the diagnostic composition may be administered by other routes as well.
- Embodiment 1 Use of a labeling compound as a labeling agent for detecting cancerous tissue in a subject, said use comprising administration of a labeling dose of said labeling compound to said subject.
- Embodiment 2 The use of embodiment 1, wherein said labeling dose is adjusted to label cancerous tissue, preferably specifically label cancerous tissue.
- Embodiment 3 The use of embodiment 1 or 2, wherein said labeling dose is at least 200 pg, preferably at least 500 pg, of labeling compound per subject.
- Embodiment 4 The use of any one of embodiments 1 to 3, wherein said labeling dose is at least 1 mg of said labeling compound per subject.
- Embodiment 5 The use of any one of embodiments 1 to 4, wherein said labeling dose is of from 0.2 mg to 100 mg, preferably is of from 0.5 to 25 mg, more preferably is of from 1 mg to 10 mg, most preferably is of from 1 mg to 5 mg, of said labeling compound per subject.
- Embodiment 6 The use of any one of embodiments 1 to 5, wherein said labeling dose is administered independent of the subject's body mass.
- Embodiment 7 The use of any one of embodiments 1 to 6, wherein said labeling dose is exactly one dose of said labeling compound administered within a time frame of at least twelve hours, preferably at least one day, more preferably at least two days.
- Embodiment 8 The use of any one of embodiments 1 to 7, wherein said cancerous tissue is tissue comprising prostate cancer cells.
- Embodiment 9 The use of any one of embodiments 1 to 8, wherein said cancerous tissue is a primary tumor, a relapse, an infiltrate, a metastasis and/or a lymph node comprising cancer cells.
- Embodiment 10 The use of any one of embodiments 1 to 9, wherein said cancerous tissue is an infiltrate, a metastasis and/or a lymph node comprising cancer cells.
- Embodiment 11 The use of any one of embodiments 1 to 10, wherein said labeling further comprises administration of a radioactively labeled derivative of the labeling compound at least two days before administration of said labeling compound, and further comprises increasing the labeling dose of labeling compound in case the signal of said radioactively labeled complex of the labeling compound in tumor or relapse tissue is found to be weak compared to a reference.
- Embodiment 12 The use of any one of embodiments 1 to 11, wherein said dose of the labeling compound is at least 20fold, preferably at least 30fold, the dose of the dose of the radioactively labeled complex of the labeling compound, preferably in case the signal of said radioactively labeled complex of the labeling compound in tumor or relapse tissue is found to be above or equal to a reference.
- Embodiment 13 The use of any one of embodiments 1 to 11, wherein said dose is least
- Embodiment 14 The use of any one of embodiments 1 to 13, wherein said dose is at most
- Embodiment 15 The use of any one of embodiments 1 to 14, wherein said reference is obtained from a multitude of primary and/ or relapse prostate cancer samples.
- Embodiment 16 The use of any one of embodiments 1 to 15, wherein said use comprises intraoperative identification of cancerous tissue.
- Embodiment 17 A labeling compound as specified in any one of embodiments 1 to 44 for use for in vivo labeling of cancerous tissue in a subject, said labeling comprising administration of a labeling dose of the labeling compound to said subject.
- Embodiment 18 A labeling compound as specified in any one of embodiments 1 to 44 for use in a method of diagnosing of cancerous tissue in a subject, wherein said method comprises administration of a labeling dose of the labeling compound to said subject.
- Embodiment 19 The labeling compound for use of embodiment 18, wherein said diagnosing is in vivo diagnosing.
- Embodiment 20 The labeling compound for use of embodiment 18 or 19, wherein said diagnosing is intraoperative identification of cancerous tissue.
- Embodiment 21 The labeling compound for use of any one of embodiments 18 to 20, wherein said diagnosing is identification of primary tumor, relapse, metastases and/or lymph nodes comprising cancer cells.
- Embodiment 22 The labeling compound for use of any one of embodiments 18 to 21, wherein said diagnosing is identification of metastases and/or lymph nodes comprising cancer cells, preferably is intraoperative identification of metastases and/or lymph nodes comprising cancer cells.
- Embodiment 23 A labeling compound as specified in any one of embodiments 1 to 44 for use in a method of removing a cancerous tissue from a subject, said method comprising
- Embodiment 24 The labeling compound for use of embodiment 23, wherein said identifying in step (b) comprises irradiating tissue assumed to comprise or be cancerous tissue with light with a wavelength absorbed by said labeling compound, preferably with a wavelength of from 600 nm to 780 nm.
- Embodiment 25 The labeling compound for use of embodiment 23 or 24, wherein said identifying in step (b) further comprises detecting fluorescent light emitted by said labeling compound, thereby identifying labeled cancerous tissue.
- Embodiment 26 The labeling compound for use of any one of embodiments 23 to 25, wherein said removing in step (c) comprises surgical removal of labeled cancerous tissue, preferably all labeled cancerous tissue identified in step (b).
- Embodiment 27 The labeling compound for use of any one of embodiments 23 to 26, wherein no further doses of the labeling compound are administered.
- Embodiment 28 The labeling compound for use of any one of embodiments 23 to 27, wherein step (a) is performed within a time frame of from 0.25 h to 48 h, more preferably of from 0.5 h to 24 h, still more preferably of from 0.75 h to 12 h, even more preferably at about 1 h, most preferably 1 h, before identifying labeled cancerous tissue in step (b).
- Embodiment 29 The labeling compound for use of any one of embodiments 17 to 28, further comprising at least one further feature of any one of embodiments 1 to 16.
- Embodiment 30 A method for labeling cancerous tissue in a subject, comprising administering a labeling dose of labeling compound as specified in any one of embodiments 1 to 44 to said subject.
- Embodiment 31 A method for identifying cancerous tissue in a subject comprising
- Embodiment 32 A method for removing cancerous tissue from a subject, said method comprising
- Embodiment 33 The method of any one of embodiments 30 to 32, further comprising at least one further feature of any one of embodiments 1 to 29.
- Embodiment 34 Use of a labeling compound as specified in any one of embodiments 1 to
- diagnostic composition for labeling cancerous tissue, preferably in vivo labeling cancerous tissue, wherein said diagnostic composition comprises a labeling dose of said labeling compound.
- Embodiment 35 The use of embodiment 34, further comprising at least one further feature of any one of embodiments 1 to 33.
- Embodiment 36 A diagnostic composition comprising of from 0.2 mg to 100 mg, preferably is of from 0.5 to 25 mg, more preferably is of from 1 mg to 10 mg, most preferably is of from 1 mg to 5 mg of a labeling compound as specified in any one of embodiments 1 to 44.
- Embodiment 37 The subject matter of any of the preceding embodiments, wherein said labeling compound has a structure of formula (I)
- (A) is at least one motif specifically binding to cell membranes of cancer cells
- (C) a dye moiety
- xi is a spacer covalently connecting (A) and (B);
- X2 is a spacer or a chemical single bond connecting (B) and (C). and wherein xi comprises a group -AA-, wherein AA is an amino acid sequence of 4 to 8 (preferably 5 to 7, more preferably 6) naturally occurring amino acids and wherein at least two amino acids are histidine.
- Embodiment 38 The subject matter of the preceding embodiment 37, wherein said dye moiety has the formula wherein
- X 2 and X 3 are independently selected from the group consisting of O, S, Se, N(R 5 ), and C(R 6 R 7 ), preferably are both C(CH3)2;
- Y is a linker connecting the two moieties of (C) and permitting electron delocalization between said moieties, wherein Y optionally comprises a group (L-)cZ 0 ; a and b are independently selected from the group consisting of 1, 2, and 3; each R 1 and each R 2 is independently (L-) C Z, (L-) C Z° or H; and two adjacent R 1 and/or two adjacent R 2 can also form an aromatic ring, which is optionally substituted with one or more (L-)cZ or (L-)cZ°;
- R 3 , R 4 , R 5 , R 6 , R 7 , R 9 are independently selected from the group consisting of (L-)cZ, (L-) C Z°, and H; each c is independently 0, or 1 ; each L is independently NH OjT 1 , or a Ci -io alkylene group, which is optionally interrupted and/or terminated by one or more of -O-, -S-, -C(O)-, -C(O)O-, -OC(O)-, -C(O)NH-, -NHC(O)O-, and T 1 ;
- T 1 is phenyl, naphthyl, indenyl, indanyl, tetralinyl, decalinyl, adamantyl, C3-7 cycloalkyl, 3 to 7 membered heterocyclyl, or 7 to 11 membered heterobicyclyl, wherein T 1 is optionally substituted with one or more substituents selected from the group consisting of halogen, CN, C(O)R 8 , COOR 8 , OR 8 , C(O)N(R 8 R 8a ), S(O) 2 N(R 8 R 8a ), S(O)N(R 8 R 8a ), S(O) 2 R 8 , N(R 8 )S(O) 2 N(R 8a R 8b ), SR 8 , N(R 8 R 8a ), NO 2 ; OC(O)R 8 , N(R 8 )C(O)R 8a , N(R 8 )S(O) 2 R 8a
- R 8 , R 8a , R 8b are independently selected from the group consisting of H, or Ci-6 alkyl, wherein Ci-6 alkyl is optionally substituted with one or more halogen, which are the same or different;
- Z° is a chemical bond connecting (C) to x 2 or to (B) in case x 2 is a chemical single bond; provided that one of R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 9 is (L-) C Z° or that Y comprises (L-) C Z°.
- Embodiment 39 The subject matter of the preceding embodiment 37, wherein said dye moiety (C) is a fluorescent dye moiety selected from the group consisting of the following structures: wherein X' is a pharmaceutically acceptable negatively charged counterion; wherein Y + is a pharmaceutically acceptable positively charged counterion; and wherein the wavy line indicates the conjugation site to the rest of the compound of formula (I).
- said dye moiety (C) is a fluorescent dye moiety selected from the group consisting of the following structures: wherein X' is a pharmaceutically acceptable negatively charged counterion; wherein Y + is a pharmaceutically acceptable positively charged counterion; and wherein the wavy line indicates the conjugation site to the rest of the compound of formula (I).
- Embodiment 40 The subject matter of any one of the preceding embodiment 37 to 39, wherein the chelator moiety (B) is selected from the following group: wherein in each of the structures one of the wavy lines indicates the conjugation site to the at least one motif specifically binding to cell membranes of cancer cells (A) via spacer xi as defined herein, and the other wavy line indicates the conjugation site to the dye moiety (C), preferably via the spacer X2 Emodiment 41: The subject matter of the preceding embodiment 40, wherein the chelator moiety (B) is selected from the following group: wherein in each of the structures one of the wavy lines indicates the conjugation site to the at least one motif specifically binding to cell membranes of cancer cells (A) via spacer xi as defined herein, and the other wavy line indicates the conjugation site to the dye moiety (C), preferably via the spacer X2 Emodiment 41: The subject matter of the preceding embodiment 40, wherein the chelator moiety (B) is
- Embodiment 42 The subject matter of any one of the preceding embodiments 37 to 41, wherein (A) is a PSMA binding motif having the following structure:
- Embodiment 43 The subject matter of any one of the preceding embodiment 37 to 42, wherein
- Embodiment 44 The subject matter of any one of the preceding embodiments, wherein the compound of formula (I) has the following chemical structure:
- Fig. 1 Preoperative 68 Ga-PSMA-914 PET/CT imaging: (A) MIP of a 76-year old patient with high-risk prostate carcinoma (Gleason score 9, initial PSA level 99.2 ng/ml) at 1 h p.i. of 195 MBq 68 Ga-PSMA-914 with corresponding axial slices of the extensive primary tumor (B) and mesorectal lymph node metastases (C) using predefined PET window settings (inverted grey scale, SUV range 0 to 5 g/ml)
- Fig. 2 Intraoperative fluorescence guidance: PSMA-914 was administered 1 h prior to surgery and resection of primary tumor (A) and lymph node metastases (B) was performed under fluorescence guidance in situ. The fluorescence signal was additionally confirmed ex situ with an exemplary lymph node shown in (C); fluorescence detection was performed with an IMAGE S system (Excitation wavelength 805 nm).
- Fig. 3 Postoperative fluorescence detection and histopathology: Fluorescence detection of the primary tumor (A) and lymph node metastases (C) was performed ex situ (excitation wavelength 800 nm, 10 gm sections) and correlated with corresponding PSMA stainings (B and D).
- Preoperative 68 Ga-PSMA-914 PET/CT imaging revealed high tracer uptake in the primary tumors at 1 h (mean SUVmax 21.6 ⁇ 19.5 g/ml; range 6.1 - 49.0 g/ml), slightly increasing at 2 h (mean SUVmax 26.9 ⁇ 25.8 g/ml; range 6.6 - 63.3 g/ml). Lymph node metastases were additionally found on PET imaging in three patients. Intraoperatively, the fluorescence signal of PSMA-914 specifically visualized primary tumor tissue and lymph node metastases with a high contrast to surrounding healthy structures and resections were performed under fluorescence-guidance.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Immunology (AREA)
- Chemical & Material Sciences (AREA)
- Biomedical Technology (AREA)
- General Health & Medical Sciences (AREA)
- Urology & Nephrology (AREA)
- Hematology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Physics & Mathematics (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- Molecular Biology (AREA)
- Optics & Photonics (AREA)
- Cell Biology (AREA)
- Microbiology (AREA)
- Biotechnology (AREA)
- Pharmacology & Pharmacy (AREA)
- Food Science & Technology (AREA)
- Analytical Chemistry (AREA)
- Biochemistry (AREA)
- General Physics & Mathematics (AREA)
- Pathology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The present invention relates to a use of a labeling compound having a chemical structure of formula (I): (A)-x1-(B)-x2-(C), wherein (A) is at least one motif specifically binding to cell membranes of cancer cells; (B) at least one chelator moiety; (C) a dye moiety; x1 is a spacer covalently connecting (A) and (B); x2 is a spacer or a chemical single bond connecting (B) and (C), or a pharmaceutically acceptable salt thereof, as a labeling agent for detecting cancerous tissue in a subject, said use comprising administration of a labeling dose of said labeling compound to said subject; and to compounds for use, and methods related thereto.
Description
Dosage of PSMA-ligands for fluorescence based detection of cancerous tissue
The present invention relates to a use of a labeling compound having a chemical structure of formula (I): (A)-xi-(B) -X2-(C), wherein (A) is at least one motif specifically binding to cell membranes of cancer cells; (B) at least one chelator moiety; (C) a dye moiety; xi is a spacer covalently connecting (A) and (B); X2 is a spacer or a chemical single bond connecting (B) and (C), or a pharmaceutically acceptable salt thereof, as a labeling agent for detecting cancerous tissue in a subject, said use comprising administration of a labeling dose of said labeling compound to said subject; and to compounds for use, and methods related thereto.
Prostate cancer (PCa) is the leading cancer in the US and European population. At least 1-2 million men in the western hemisphere suffer from prostate cancer and it is estimated that the disease will affect one in six men between the ages of 55 and 85. There are more than 300,000 new cases of prostate cancer diagnosed each year in the USA. The mortality from the disease is second only to lung cancer. Currently, imaging methods with high resolution of the anatomy, such as computed tomography (CT), magnetic resonance (MR) imaging and ultrasound, predominate for clinical imaging of prostate cancer. An estimated annual $ 2 billion is currently spent worldwide on surgical, radiation, drug therapy and minimally invasive treatments. However, there is presently no effective therapy for relapsing, metastatic, androgenindependent prostate cancer.
It is well known that tumors may express unique proteins associated with their malignant phenotype or may over-express normal constituent proteins in greater number than normal cells. The expression of distinct proteins on the surface of tumor cells offers the opportunity to diagnose and characterize disease by probing the phenotypic identity and biochemical composition and activity of the tumor. Radioactive molecules that selectively bind to specific tumor cell surface proteins provide an attractive route for imaging and treating tumors under non-invasive conditions. A promising new series of low molecular weight imaging agents targets the prostate-specific membrane antigen (PSMA) (Mease R.C. et al. Clin Cancer Res. 2008, 14, 3036-3043; Foss, C.A.; et al. Clin Cancer Res 2005, 11 , 4022-4028; Pomper, M.G.; et al. Mol Imaging 2002, 1 , 96-101 ; Zhou, J.; et al. Nat Rev Drug Discov 2005, 4, 015-1026;
WO 2013/022797). In WO 2015/055318 new imaging agents with improved tumor targeting properties and pharmacokinetics were described. These compounds comprise a motif specifically binding to cell membranes of cancer cells, wherein said motif comprises a prostatespecific membrane antigen (PSMA), that is the above mentioned glutamate-urea-lysine motif.
PSMA is a trans-membrane, 750 amino acid type II glycoprotein that has abundant and restricted expression on the surface of PCa, particularly in androgen-independent, advanced and metastatic disease (Schulke, N.; et al. Proc Natl Acad Sci U S A 2003, 100, 12590-12595). The latter is important since almost all PCa become androgen independent over the time. PSMA possesses the criteria of a promising target for therapy (Schulke, N.; et al. Proc. Natl. Acad. Sci. U S A 2003, 100, 12590-12595). The PSMA gene is located on the short arm of chromosome 11 and functions both as a folate hydrolase and neuropeptidase. It has neuropeptidase function that is equivalent to glutamate carboxypeptidase II (GCPII), which is referred to as the "brain PSMA", and may modulate glutamatergic transmission by cleaving /V-acetylaspartylglutamate (NAAG) to N-acetylaspartate (NAA) and glutamate (Nan, F.; et al. J Med Chem 2000, 43, 772- 774). There are up to 106 PSMA molecules per cancer cell, further suggesting it as an ideal target for imaging and therapy with radionuclide-based techniques (Tasch, J.; et al. Crit Rev Immunol 2001 , 21 , 249-261).
For treatment of localized prostate cancer, radical prostatectomy with lymph node dissection is an established curative strategy. However, the precise intraoperative localization and delineation of tumor margins and metastases remain challenging.
Thus there is still a need for improved PSMA ligands which provide advantageous options for the detection, treatment, and management of PSMA-expressing cancers, in particular prostate cancer. The technical problem underlying the present invention can be seen as the provision of means and methods for complying with the aforementioned needs. The technical problem is solved by the embodiments characterized in the claims and herein below.
In accordance, the present invention relates to a use of a labeling compound having a chemical structure of formula (I): (A)-XI-(B)-X2-(C), wherein (A) is at least one motif specifically binding to cell membranes of cancer cells; (B) at least one chelator moiety; (C) a dye moiety; xi is a spacer covalently connecting (A) and (B); X2 is a spacer or a chemical single bond connecting (B) and (C), or a pharmaceutically acceptable salt thereof, as a labeling agent for detecting
cancerous tissue in a subject, said use comprising administration of a labeling dose of said labeling compound to said subject.
As used in the following, the terms “have”, “comprise” or “include” or any arbitrary grammatical variations thereof are used in a non-exclusive way. Thus, these terms may both refer to a situation in which, besides the feature introduced by these terms, no further features are present in the entity described in this context and to a situation in which one or more further features are present. As an example, the expressions “A has B”, “A comprises B” and “A includes B” may both refer to a situation in which, besides B, no other element is present in A (i.e. a situation in which A solely and exclusively consists of B) and to a situation in which, besides B, one or more further elements are present in entity A, such as element C, elements C and D or even further elements. Also, as is understood by the skilled person, the expressions "comprising a" and "comprising an" preferably refer to "comprising one or more", i.e. are equivalent to "comprising at least one".
Further, as used in the following, the terms "preferably", "more preferably", "most preferably", "particularly", "more particularly", "specifically", "more specifically" or similar terms are used in conjunction with optional features, without restricting further possibilities. Thus, features introduced by these terms are optional features and are not intended to restrict the scope of the claims in any way. The invention may, as the skilled person will recognize, be performed by using alternative features. Similarly, features introduced by "in an embodiment" or similar expressions are intended to be optional features, without any restriction regarding further embodiments of the invention, without any restrictions regarding the scope of the invention and without any restriction regarding the possibility of combining the features introduced in such way with other optional or non-optional features of the invention.
As used herein, the term "standard conditions", if not otherwise noted, relates to IUPAC standard ambient temperature and pressure (SATP) conditions, i.e. preferably, a temperature of 25°C and an absolute pressure of 100 kPa; also preferably, standard conditions include a pH of 7. Moreover, if not otherwise indicated, the term "about" relates to the indicated value with the commonly accepted technical precision in the relevant field, preferably relates to the indicated value ± 20%, more preferably ± 10%, most preferably ± 5%. Further, the term "essentially" indicates that deviations having influence on the indicated result or use are absent, i.e. potential deviations do not cause the indicated result to deviate by more than ± 20%, more preferably ±
10%, most preferably ± 5%. Thus, “consisting essentially of’ means including the components specified but excluding other components except for materials present as impurities, unavoidable materials present as a result of processes used to provide the components, and components added for a purpose other than achieving the technical effect of the invention. For example, a composition defined using the phrase “consisting essentially of’ encompasses any known acceptable additive, excipient, diluent, carrier, and the like. Preferably, a composition consisting essentially of a set of components will comprise less than 5% by weight, more preferably less than 3% by weight, even more preferably less than 1%, most preferably less than 0.1% by weight of non-specified component(s).
The use of the labeling compound, as referred to herein, comprises at least administration of a labeling dose of said labeling compound. The use may, however, in addition comprise further steps before, concomitant to, and/or after said administration deemed appropriate by the skilled person. The use, preferably, additionally comprises at least one step as specified herein, in particular a step of a use and/or a step of a method as described herein. Preferably, the use comprises intraoperative identification of cancerous tissue.
As referred to herein, the labeling compound has a structure of formula (I) (A)-XI-(B)-X2-(C), or is a pharmaceutically acceptable salt thereof, wherein
(A) is at least one motif specifically binding to cell membranes of cancer cells;
(B) at least one chelator moiety ;
(C) a dye moiety; xi is a spacer covalently connecting (A) and (B);
X2 is a spacer or a chemical single bond connecting (B) and (C). and wherein xi comprises a group -AA-, wherein AA is an amino acid sequence of 4 to 8 (preferably 5 to 7, more preferably 6) naturally occurring amino acids and wherein at least two amino acids are histidine
Such compounds are described e.g. in WO2019/092242, the contents of which is herewith incorporated by reference. Exemplary routes for the preparation of compounds or pharmaceutically acceptable salts thereof of the present description are described in US 2015/0110715 Al.
Preferably, the compound is PSMA-914 (“Glu-urea-Lys-(HE)3-HBED-CC-PEG2-
An exemplary synthesis of this compound is described in the experimental procedures of
WO20 19/092242 which contents is herewith incorporated by reference.
Thus, the labeling compound has a structure of formula (I): (A)-XI-(B)-X2-(C), including pharmaceutically acceptable salts thereof, wherein
(A) is at least one motif specifically binding to cell membranes of cancer cells;
(B) at least one chelator moiety;
(C) a dye moiety; xi is a spacer covalently connecting (A) and (B);
X2 is a spacer or a chemical single bond connecting (B) and (C); wherein xi comprises a group -AA-, wherein AA is an amino acid sequence of 4 to 8 (preferably 5 to 7, more preferably 6) naturally occurring amino acids and wherein at least two amino acids are histidine.
Preferably any remaining positive or negative charge or charges are compensated by pharmaceutically acceptable negatively or positively charged counterion or counterions.
As used herein, the term “pharmaceutically acceptable salt” may be understood in the broadest sense as any charged form of the labeling compound of the present description. Depending on the chemical structure of the compound and on the environment it is dissolved in, the compound may, exemplarily, comprise one or more charged residue(s) selected from the group consisting of but not limited to carboxylate anion residue(s), primary ammonium cation(s), secondary ammonium cation residue(s), tertiary ammonium cation residue(s), primary phosphate anion residue(s), secondary phosphate anion residue(s), sulfate anion residue(s), sulfite anion residue(s) and an alkoxide residue(s). The counterions may be any ions known to be pharmaceutically acceptable in the art such as, e.g., acetate, fatty acid carboxylate, chloride, sodium ions, potassium ion, magnesium ion, calcium ion, aluminum ion, lithium ion, ammonium, phosphate, hydroxyl, proton and fluoride ion.
As used throughout the present description, the term “motif’ may be understood in the broadest sense as a molecular structure pattern that enables specific binding to cell membranes of cancer cells.
The moiety A:
The compound or pharmaceutically acceptable salt thereof comprises a moiety (A) with at least one motif. Accordingly moiety (A) can represent one or more motifs. Exemplarily moiety (A) represents a chemical structure with one, two or three motifs. However, preferably (A) represents one motif. Preferably, the motif comprises at least one naturally occurring amino acid moiety, more preferably at least two naturally occurring amino acid moieties. As used throughout the present description, the terms “moiety”, “residue” and “rest” in the context of a chemical structure may be understood interchangeably in the broadest sense as a part of a molecule tightly bound to the other parts of the molecule, in particular via a covalent bond. Further, the terms “conjugated to” and “bound to” as used herein may be understood interchangeably.
Preferably, the motif comprises at least one non-proteinogenic amide bond, more preferably at least two non-proteinogenic amide bonds. More preferably, the motif comprises at least one naturally occurring amino acid moiety conjugated via a non-proteinogenic amide bond, even more preferably at least two naturally occurring amino acid moieties conjugated via non-
proteinogenic amide bonds. Preferably, the motif specifically binding to cell membranes of cancer cells comprises not more than 20 amino acid moieties, more preferably not more than ten amino acid moieties, even more preferably not more than five amino acid moieties, even more preferably not more than four amino acid moieties, in particular not more than three amino acid moieties. Preferably, the motif further comprises at least one urea moiety, more preferably at least one urea moiety covalently bound to two amino acids via amide bond formation. The at least one motif specifically binding to cell membranes of cancer cells (A) is covalently linked with the at least one chelator moiety (B) via spacer xi. In this context, preferably, the spacer xi is of not more than 5 nm in length, preferably of not more than 2 nm in length, in particular of not more than 1 nm in length. The motif may preferably be conjugated to the chelator moiety (B) via the epsilon amino group of a lysine moiety.
The term “chelator moiety”, as used herein, may be understood in the broadest sense as any moiety that is able to form a complex with a radiometal under suitable conditions. Herein, the terms “chelator moiety”, “chelant”, “chelating moiety”, “sequestering moiety” and “complexing moiety” may be understood interchangeably. The chelator moiety is preferably an organic moiety. Complexing by chelation preferably involves the formation or presence of two or more separate coordinate bonds between a polydentate (multiple bonded) ligand and a single central radiometal. The common definition of the International Union of Pure and Applied Chemistry (IUPAC) on chelation, interpreted in its broadest sense, may also be noted. Chelator moieties as used herein will typically bear at least two heteroatoms enabling an interaction with a radiometal. Preferably, the chelator moiety will have at least three, in particular at least four heteroatoms enabling an interaction with a metal, in particular a radiometal. The chelator moiety may comprise a chelated metal, preferably a metal ion, more preferably a radiometal ion, all a specified herein below. More preferably, the labeling agent is provided and/or admininstered without chelated metal.
The labeling compound or pharmaceutically acceptable salt thereof comprises a moiety (B) with at least one chelator moiety. Accordingly moiety (B) can represent one or more chelators. Exemplarily moiety (B) represents a chemical structure with one, two or three chelators. However, preferably (B) represents one chelator. Moiety (B) is connected to moiety (A) via spacer xi as outlined herein.
The compound or pharmaceutically acceptable salt thereof comprises a dye moiety (C), which may also be referred to as a "label". Moiety (C) is connected to (B) via x2, which represents a spacer or a chemical single bond.
X1 and X4 are independently selected from the group consisting of -N=, -N(R5)=, and -C(R6)=;
X2 and X3 are independently selected from the group consisting of O, S, Se, N(R5), and C(R6R7), preferably both are C(CH3)2;
Y is a linker connecting the two moieties of (C) and permitting electron delocalization between said moieties, wherein Y optionally comprises a group (L-)cZ0; a and b are independently selected from the group consisting of 1, 2, and 3; each R1 and each R2 is independently selected from the group consisting of (L-)CZ, (L-)CZ° and H; and two adjacent R1 and/or two adjacent R2 can also form an aromatic ring, which is optionally substituted with one or more (L-)CZ or (L-)CZ°;
R3, R4, R5, R6, R7, R9 are independently selected from the group consisting of (L-)cZ, (L-)CZ°, and H; each c is independently 0, or 1 ; each L is independently T1, -OT1-, -ST1-, -Cf jT1-, -QOjOT1-, -(DC(O)T1-, -Cf jNHT1-, - NH OjT1, or a Ci -io alkylene group, which is optionally interrupted and/or terminated by one or more of -O-, -S-, -C(O)-, -C(O)O-, -OC(O)-, -C(O)NH-, -NHC(O)O-, and T1;
T1 is phenyl, naphthyl, indenyl, indanyl, tetralinyl, decalinyl, adamantyl, C3-7 cycloalkyl, 3 to 7 membered heterocyclyl, or 7 to 11 membered heterobicyclyl, wherein T1 is optionally substituted with one or more substituents selected from the group consisting of halogen, CN, C(O)R8, COOR8, OR8, C(O)N(R8R8a), S(O)2N(R8R8a), S(O)N(R8R8a), S(O)2R8, N(R8)S(O)2N(R8aR8b), SR8, N(R8R8a), NO2; OC(O)R8, N(R8)C(O)R8a, N(R8)S(O)2R8a,
N(R8)S(O)R8a, N(R8)C(O)N(R8aR8b), N(R8)C(O)OR8a, OC(O)N(R8R8a), oxo (=0), where the ring is at least partially saturated, or Ci-6 alkyl, wherein Ci-6 alkyl is optionally substituted with one or more halogen, which are the same or different; each Z is independently H, halogen, CN, C(O)R8, C(O)OR8, C(O)O’ OR8, C(O)N(R8R8a), S(O)2OR8, S(O)2O’, S(O)2N(R8R8a), S(O)N(R8R8a), S(O)2R8, S(O)R8, N(R8)S(O)2N(R8aR8b), SR8, N(R8R8a), NO2; P(O)(OR8)2, P(O)(OR8)O’, OC(O)R8, N(R8)C(O)R8a, N(R8)S(O)2R8a, N(R8)S(O)R8a, N(R8)C(O)N(R8aR8b), N(R8)C(O)OR8a, or OC(O)N(R8R8a);
R8, R8a, R8b are independently selected from the group consisting of H, or Ci-6 alkyl, wherein Ci-6 alkyl is optionally substituted with one or more halogen, which are the same or different; Z° is a chemical bond connecting (C) to x2 or to (B) in case x2 is a chemical single bond; provided that one of R1, R2, R3, R4, R5, R6, R7, R9 is (L-)CZ° or that Y comprises (L-)CZ°.
The term “optionally substituted” means unsubstituted or substituted. Generally -but not limited to-, “one or more substituents” means one, two or three, preferably one or two substituents and more preferably one substituent. Generally these substituents can be the same or different.
“Alkyl” means a straight-chain or branched hydrocarbon chain. Each hydrogen of an alkyl carbon may be replaced by a substituent as further specified herein.
As used throughout the present application, the terms “alkyl”, “alkyl residue” and “alkyl group” and “alkyl moiety” may be understood as a straight-chain or branched saturated hydrocarbon chain. “Straight-chain” may be also designated as “unbranched” or “linear”. Preferably, the alkyl is a straight chain.
As used throughout the present description, the term “alkylene” means a straight-chain or branched saturated hydrocarbon chain wherein two moieties of a molecule are linked by the alkylene residue. “Straight-chain” may be also designated as “unbranched” or “linear”. Each hydrogen of an alkylene carbon may or may not be replaced by a substituent (i.e., may be substituted or unsubstituted) as further specified herein).
“Ci-4 alkyl” means an alkyl chain having 1 - 4 carbon atoms, e.g. if present at the end of a molecule: methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, or e.g. - CH2-, -CH2-CH2-, -CH(CH3)-, -CH2-CH2-CH2-, -CH(C2H5)-, -C(CH3)2-, when two moieties of
a molecule are linked by the alkyl group. Each hydrogen of a C alkyl carbon may be replaced by a substituent as further specified herein. “CM alkyl” means an alkyl chain having 1 - 6 carbon atoms, e.g. if present at the end of a molecule: C alkyl, methyl, ethyl, n-propyl, isopropyl, n- butyl, isobutyl, sec-butyl; tert-butyl, n-pentyl, n-hexyl, or e.g. -CH2-, -CH2-CH2-, -CH(CH3)-, - CH2-CH2-CH2-, -CH(C2HS)-, -C(CH3)2-, when two moieties of a molecule are linked by the alkyl group. Each hydrogen of a CM alkyl carbon may be replaced by a substituent as further specified herein.
“C1-8 alkylene residue” means an alkylene chain having 1 - 8 carbon atoms, e.g. -CH2-, -CH2-CH2-, -CH(CH3)-, -CH2-CH2-CH2-, -CH(C2H5)-, -C(CH3)2-, -CH2-C(CH3)2-, - C(CH2-CH3)2-, -CH(CH2-CH3)-, -CH2-CH(CH3)(CH2-CH3)-,
-CH(CH3)(CH2-CH3)-, -(CH2)4-, -(CH2)5-, -(CH2)6-, -(CH2)7-, -(CH2)8-, etc., when two moieties of a molecule are linked by the alkylene group. The terms “C4-8 alkylene” and “Ce alkylene” are defined accordingly. The terms “C3-7 alkylene” and “C5 (C4) alkylene” are defined accordingly. The term “C1-10 alkylene group” means a bivalent straight-chain or branched hydrocarbon chain having 1 to 10 carbon atoms. Each hydrogen of an alkyl carbon may be replaced by a substituent as further specified herein. Examples are methylene (-CH2-) -CH2- CH2-, -CH(CH3)-, -CH2-CH2-CH2-, -CH(C2H5)-, -C(CH3)2-. Each hydrogen of a Ci-io alkylene group carbon may be replaced by a substituent as further specified herein. Accordingly, “C1-10 alkylene residue” means an alkylene chain having 1 - 10 carbon atoms when two moieties of a molecule are linked by the alkylene group. Preferably, but not necessarily, the C1-10 alkylene residue in the context of residue f of the spacer y is a straight-chain, i.e., unbranched, C1-10 alkylene residue, in which optionally one or more hydrogen(s) are substituted and/or in which optionally one or more -CEE- moieties may be replaced by -O- or -NH-.
The expression “one or more -CH2- moieties may optionally be replaced by” means that the indicated number of CEE groups can be replaced by an atom or group specified herein. In addition one or more hydrogens as specified herein can be replaced by a substituent.
Ci-10 alkylene group “optionally interrupted and/or terminated” means that the alkylene chain is interrupted between two carbon atoms by an atom or a chemical group as specified herein or the alkylene group is terminated by said atom or group following the carbon at least at one end of the alkylene chain or the alkylene chain is both, interrupted and terminated or the alkylene chain is neither interrupted nor terminated. As an example without being bound to that example
a C3 alkylene group which is optionally interrupted and/or terminated with one or more X may have the sequence C-C-C, C-C-C-X, X-C-C-C, X-C-C-C-X, C-X-C-C, C-C-X-C, C-X-C-X-C, X-C-C-X-C, X-C-X-C-X-C, X-C-X-C-C-X, X-C-X-C-X-C-X.
The term “carbocycle” refers to a partly or fully saturated or aromatic carbocyclic mono-, bi- or tricyclic fused or unfused ring system. This includes phenyl and C3-7 cycloalkyl rings. Preferred carbocycles having 5, 6 or 7 carbon atoms are cyclopentene, cyclohexene, phenyl, cycloheptane, especially cyclohexane.
“C3-7 cycloalkyl” or “C3 -7 cycloalkyl ring” means a cyclic alkyl chain having 3 - 7 carbon atoms, e.g. cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cyclohexenyl, cycloheptyl. Preferably, cycloalkyl refers to cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, or cycloheptyl. Each hydrogen of a cycloalkyl carbon may be replaced by a substituent as further specified herein. The term “C3-5 cycloalkyl” or “C3-5 cycloalkyl ring” is defined accordingly.
“Halogen” means fluoro, chloro, bromo or iodo. It is generally preferred that halogen is fluoro or chloro.
Within the meaning of the present description the term “aromatic ring” means a carbocyclic or heterocyclic aromatic ring. Examples are benzene, naphthalene, 5 to 6 membered aromatic heterocycle and 9 to 11 membered aromatic heterobicyclyl.
“3 to 7 membered heterocyclyl” or “3 to 7 membered heterocycle” means a ring with 3, 4, 5, 6 or 7 ring atoms that may contain up to the maximum number of double bonds (aromatic or nonaromatic ring which is fully, partially or un-saturated) wherein at least one ring atom up to 4 ring atoms are replaced by a heteroatom selected from the group consisting of sulfur (including -S(O)-, -S(O)2-), oxygen and nitrogen (including =N(O)-) and wherein the ring is linked to the rest of the molecule via a carbon or nitrogen atom. Examples for a 3 to 7 membered heterocycle are aziridine, azetidine, oxetane, thietane, furan, thiophene, pyrrole, pyrroline, imidazole, imidazoline, pyrazole, pyrazoline, oxazole, oxazoline, isoxazole, isoxazoline, thiazole, thiazoline, isothiazole, isothiazoline, thiadiazole, thiadiazoline, tetrahydrofiiran, tetrahydrothiophene, pyrrolidine, imidazolidine, pyrazolidine, oxazolidine, isoxazolidine, thiazolidine, isothiazolidine, thiadiazolidine, sulfolane, pyran, dihydropyran, tetrahydropyran, imidazolidine, pyridine, pyridazine, pyrazine, pyrimidine, piperazine, piperidine, morpholine,
tetrazole, triazole, triazolidine, tetrazolidine, diazepane, azepine or homopiperazine. The term “4 to 7 membered heterocyclyl” or “4 to 7 membered heterocycle” is defined accordingly. The term “5 to 6 membered heterocyclyl” or “5 to 6 membered heterocycle” is defined accordingly.
“5 to 6 membered aromatic heterocyclyl” or “5 to 6 membered aromatic heterocycle” means a heterocycle derived from cyclopentadienyl or benzene, where at least one carbon atom is replaced by a heteroatom selected from the group consisting of sulfur (including -S(O)-, -S(O)2- ), oxygen and nitrogen (including =N(O)-). Examples for such heterocycles are furan, thiophene, pyrrole, imidazole, pyrazole, oxazole, isoxazole, thiazole, isothiazole, thiadiazole, triazole, tetrazole, pyridine, pyrimidine, pyridazine, pyrazine, triazine.
“5 membered aromatic heterocyclyl” or “5 membered aromatic heterocycle” means a heterocycle derived from cyclopentadienyl, where at least one carbon atom is replaced by a heteroatom selected from the group consisting of sulfur (including -S(O)-, -S(O)2-), oxygen and nitrogen (including =N(O)-). Examples for such heterocycles are furan, thiophene, pyrrole, imidazole, pyrazole, oxazole, isoxazole, thiazole, isothiazole, thiadiazole, triazole, tetrazole.
"7 to 11 membered heterobicyclyl" or "7 to 11 membered heterobicycle" means a heterocyclic system of two rings with 7 to 11 ring atoms, where at least one ring atom is shared by both rings and that may contain up to the maximum number of double bonds (aromatic or non-aromatic ring which is fully, partially or un-saturated) wherein at least one ring atom up to 6 ring atoms are replaced by a heteroatom selected from the group consisting of sulfur (including -S(O)-, - S(O)2-), oxygen and nitrogen (including =N(O)-) and wherein the ring is linked to the rest of the molecule via a carbon or nitrogen atom. Examples for a 7 to 11 membered heterobicycle are indole, indoline, benzofuran, benzothiophene, benzoxazole, benzisoxazole, benzothiazole, benzisothiazole, benzimidazole, benzimidazoline, quinoline, quinazoline, dihydroquinazoline, quinoline, dihydroquinoline, tetrahydroquinoline, decahydroquinoline, isoquinoline, decahydroisoquinoline, tetrahydroisoquinoline, dihydroisoquinoline, benzazepine, purine or pteridine. The term 7 to 11 membered heterobicycle also includes spiro structures of two rings like 6-oxa-2-azaspiro[3,4]octane, 2-oxa-6-azaspiro[3.3]heptan-6-yl or 2,6- diazaspiro[3.3]heptan-6-yl or bridged heterocycles like 8-aza-bicyclo[3.2.1]octane or 2,5- diazabicyclo[2.2.2]octan-2-yl or 3,8-diazabicyclo[3.2.1]octane.
“9 to 11 membered aromatic heterobicyclyl” or “9 to 11 membered aromatic heterobicycle” means a heterocyclic system of two rings, wherein at least one ring is aromatic and wherein the heterocyclic ring system has 9 to 11 ring atoms, where two ring atoms are shared by both rings and that may contain up to the maximum number of double bonds (fully or partially aromatic) wherein at least one ring atom up to 6 ring atoms are replaced by a heteroatom selected from the group consisting of sulfur (including -S(O)-, -S(O)2-), oxygen and nitrogen (including =N(O)-) and wherein the ring is linked to the rest of the molecule via a carbon or nitrogen atom. Examples for a 9 to 11 membered aromatic heterobicycle are indole, indoline, benzofuran, benzothiophene, benzoxazole, benzisoxazole, benzothiazole, benzisothiazole, benzimidazole, benzimidazoline, quinoline, quinazoline, dihydroquinazoline, dihydroquinoline, tetrahydroquinoline, isoquinoline, tetrahydroisoquinoline, dihydroisoquinoline, benzazepine, purine or pteridine. The terms “9 to 10 membered aromatic heterobicyclyl” or “9 to 10 membered aromatic heterobicycle” are defined accordingly.
Preferred compounds of formula (I) are those compounds in which one or more of the residues contained therein have the meanings given below, with all combinations of preferred substituent definitions being a subject of the present invention. With respect to all preferred compounds of the formula (I) the present invention also includes all tautomeric and stereoisomeric forms and mixtures thereof in all ratios, and their pharmaceutically acceptable salts.
Where tautomerism, like e.g. keto-enol tautomerism, of compounds of formula (I) may occur, the individual forms, like e.g. the keto and enol form, are comprised separately and together as mixtures in any ratio. Same applies for stereoisomers, like e.g. enantiomers, cis/trans isomers, conformers and the like.
Especially, when enantiomeric or diastereomeric forms are given in a compound according to formula (I) each pure form separately and any mixture of at least two of the pure forms in any ratio is comprised by formula (I) and is a subject of the present invention.
Isotopic labeled compounds of formula (I) are also within the scope of the present invention. Methods for isotope labeling are known in the art. Preferred isotopes are those of the elements H, C, N, O and S. Solvates of compounds of formula (I) are also within the scope of the present invention.
In case the compounds according to formula (I) contain one or more acidic or basic groups, the invention also comprises their corresponding pharmaceutically or toxicologically acceptable salts, in particular their pharmaceutically utilizable salts. Thus, the compounds of the formula (I) which contain acidic groups can be used according to the invention, for example, as alkali metal salts, alkaline earth metal salts or as ammonium salts. More precise examples of such salts include sodium salts, potassium salts, calcium salts, magnesium salts or salts with ammonia or organic amines such as, for example, ethylamine, ethanolamine, triethanolamine or amino acids. Compounds of the formula (I) which contain one or more basic groups, i.e. groups which can be protonated, can be present and can be used according to the invention in the form of their addition salts with inorganic or organic acids. Examples for suitable acids include hydrogen chloride, hydrogen bromide, phosphoric acid, sulfuric acid, nitric acid, methanesulfonic acid, p-toluenesulfonic acid, naphthalenedisulfonic acids, oxalic acid, acetic acid, tartaric acid, lactic acid, salicylic acid, benzoic acid, formic acid, propionic acid, pivalic acid, diethylacetic acid, malonic acid, succinic acid, pimelic acid, fumaric acid, maleic acid, malic acid, sulfaminic acid, phenylpropionic acid, gluconic acid, ascorbic acid, isonicotinic acid, citric acid, adipic acid, and other acids known to the person skilled in the art. If the compounds of the formula (I) simultaneously contain acidic and basic groups in the molecule, the invention also includes, in addition to the salt forms mentioned, inner salts or betaines (zwitterions). The respective salts according to the formula (I) can be obtained by customary methods which are known to the person skilled in the art like, for example by contacting these with an organic or inorganic acid or base in a solvent or dispersant, or by anion exchange or cation exchange with other salts. The present invention also includes all salts of the compounds of the formula (I) which, owing to low physiological compatibility, are not directly suitable for use in pharmaceuticals but which can be used, for example, as intermediates for chemical reactions or for the preparation of pharmaceutically acceptable salts.
It is to be understood that the compounds or pharmaceutically acceptable salts thereof of the present invention may show one or more charged functional groups, wherein each negative charge is preferably compensated by a positive charge so that the molecule is neutral. Intramolecular compensation is possible as well as by other ions in form of inner and outer salts. Suitable counterions are those cited above. Accordingly, in the formulas of the present invention any remaining positive or negative charge or charges are preferably compensated by pharmaceutically acceptable negatively or positively charged counterion or counterions.
Likewise any protonated form may be partly or fully deprotonated with appropriate counterion(s).
A counterion as used herein may be any pharmaceutically acceptable ion that is suitable for neutralizing the charge of a residue or the compound of the present invention. It will be understood that a single counterion does not necessarily has the same valency as a charged residue or a charged compound of the present invention. Also two or more counterions may be used to neutralize a compound bearing a charge of higher charge valency than +1 or -1. Likewise, the other way round, also a single counterion bearing a charge of higher valency than +1 or -1 may be used to neutralize more than one compound. Preferably, the counterion is wellsoluble in aqueous liquids.
A pharmaceutically acceptable negatively charged counterion X’ in the context of the present invention may have any charge valency. Therefore, X’ may exemplarily have a charge of -1, - 2, -3 or -4, preferably of -1 or -2. Charge may also optionally depend on ion strength and pH, respectively. X~ may be any pharmaceutically acceptable negatively charged ion. Preferably, the ion is such well-soluble in aqueous liquids. Exemplarily, X~ may be selected from the group consisting of a halide anion (e.g., F’ or Cl’), acetate, phosphate, hydrogen phosphate, and a pharmaceutically acceptable carboxylate (e.g., a fatty acid carboxylate). Further, it will be understood that the counterion typically depends on the surrounding liquids such as those comprised in the buffer the compound is dissolved in and the body fluids after injection in vivo. In vivo, extracellularly, one of the main, but not sole negatively charged counterions is Cl’.
As used in the context of the present invention, the terms “dye moiety”, “label” and “stain” may be understood interchangeably in the broadest sense as any moiety with the above formula that provides a visible stain. Preferably, the dye moiety may be a fluorescent dye moiety and/or a chromatic moiety, particularly preferably the dye moiety is a fluorescent dye moiety.
A fluorescent dye moiety as used herein may be understood in the broadest sense as any dye moiety enabling fluorescence detection. Preferably, such fluorescence detection is in a range of from 400 to 1000 nm, i.e. in the visible spectrum and in the Near Infrared (NIR) spectrum, in particular in a range of from 400 to 800 nm, i.e. in the visible spectrum. Preferably, the fluorescence signal emitted by the fluorescence dye moiety is well-distinguishable from the autofluorescence of cancer and surrounding tissue. Numerous fluorescent dye moieties are
known in the art, and will be readily apparent to one of ordinary skill. Many fluorescent dyes are commercially available with activated groups used to react with protein sidechains or other compounds such as precursor compounds for the preparation of a compound of the present invention. Preferably, the dye enables a fluorescence detection by radiation with a wavelength in a range of from with a wavelength of from 400 to 1000 nm, i.e. in the visible spectrum and in the Near Infrared (NIR) spectrum, preferably of from 600 nm to 850, preferably 700 nm to 780 nm and/or allows for a detection of fluorescence by eye and/or at a wavelength in the range of from 780 nm to 850 nm.
Additionally or alternatively, the dye moiety may also be chromatic, i.e., provoke a colour perception when illuminated by any light. Such chromatic effect may be provoked by absorbing light of one or more particular wavelength range(s) in the visible range (i.e., in range(s) from approximately 400 nm to approximately 800 nm) and/or by emitting light of one or more particular wavelength range(s) in the visible range. Preferably, the colour is different from the neoplasia and the surrounding tissue intended to be examined. Therefore, a dye moiety, when not intended for fluorescence detection, is preferably not red or brown, but rather preferably blue or green. When the dye moiety is intended for fluorescence detection, the difference in colour will typically play a minor role as long as the fluorescence is detectable over the autofluorescence background. Preferably, the chromatic dye moiety in the context of the present invention is a small-molecule dye, i.e., a dye moiety having a molecular weight (MW) of not more than 1000 Da, preferably not more than 750 Da, in particular nor more than 500 Da.
The dye moiety (C) is covalently linked with the at least one motif specifically binding to cell membranes of cancer cells (A) and the at least one chelator moiety (B) via (A)-XI-(B)-X2-(C). Such covalent conjugation to a chelator moiety (B) may be the formation of a covalent bond directly between the dye moiety (C) and the chelator moiety (B) or may be covalent linkage via a spacer, preferably via spacer. Preferably, a spacer is of not more than 5 nm in length, preferably of not more than 2 nm in length, in particular of not more than 1 nm in length.
Accordingly, the molecular distance between the motif specifically binding to cell membranes of cancer cells (A) and the dye moiety (C) is preferably not longer than 20 nm, more preferably not longer than 10 nm, in particular not longer than 5 nm.
This may, depending on the chemical properties of the fluorescence dye moiety/moieties in the compound of the present invention and the presence of fluorophore(s) and/or quenchers on the surface of the target cells, i.e., the cell membranes of the respective cancer cells, also enable to observe effects such as fluorescence energy transfer (FRET) and/or fluorescence quenching upon binding of the compound according to the present invention to said cell membranes. Additionally or alternatively, the presence of the fluorescence dye moiety/moieties also enables to conduct further examination methods based on fluorescence such as, e.g., fluorescence recovery after photobleaching (FRAP), fluorescence loss in photobleaching (FLIP). These methods may provide information on the mobility of the compound or salt thereof bound to or associated with the cell membranes of a cancer cell.
Preferably, the compound or pharmaceutically acceptable salt thereof according to the present invention has a molecular weight (MW) of not more than 10 kDa, more preferably not more than 5 kDa, even more preferably not more than 3.5 kDa, even more preferably not more than 3 kDa, in particular not more than 2.5 kDa.
Preferably, the motif specifically binding to cell membranes of cancer cells (A) is a PSMA binding motif having the following structure:
wherein Z1, Z2 and Z3 are each independently from another selected from the group consisting of -C(O)ORla, -SO2Rla, -SO3Rla, -SO4Rla, -PO2Rla, -PO3Rla, and -PO4RlaR2a, wherein Rla and R2a are independently from another H or a Ci-4-alkyl residue (preferably the same and more preferably H);
wherein a’ represents a -[CH2]0- residue, wherein o is an integer from 1 to 4, preferably wherein o is 3 or 4, in particular wherein o is 4. wherein b’ represents a residue selected from the group consisting of -NH-, -C(O)- and -O-, in particular wherein b’ is -NH-; and wherein the wavy line indicates the conjugation site to the chelator moiety (B), conjugated via a spacer molecule xi.
Preferably, Z1, Z2, Z3 are the same and more preferably, Z1, Z2, Z3 are -C(O)ORla.
In a particularly preferred embodiment, the motif specifically binding to cell membranes of cancer cells (A) is a PSMA binding motif having the following structure:
wherein the wavy line indicates the conjugation site to the chelator moiety (B), conjugated via the spacer molecule xi.
The spacers xi and X2 may be spacers with not more than 5 nm in length, preferably of not more than 2 nm in length, in particular of not more than 1 nm in length.
The spacer xi comprises a group -AA-, wherein AA is an amino acid sequence of 4 to 8 (preferably 5 to 7, more preferably 6) naturally occurring amino acids and wherein at least two amino acids are histidine.
The term “naturally occurring amino acids” refers to an amino acid occurring in a composition of matter occuring in nature, preferably selected from the group consisting of Ala (Alanine), Arg (Arginine), Asn (Asparagine), Asp (Aspartic acid), Cys (Cysteine), Gin (Glutamine), Glu (Glutamic acid), Gly (Glycine), His (Histidine), He (Isoleucine), Leu (Leucine), Lys (Lysine), Met (Methionine), Phe (Phenylalanine), Pro (Proline), Ser (Serine), Thr (Threonine), Trp
(Tryptophan), Tyr (Tyrosine), and Vai (Valine). The naturally occurring amino acids may be present in racemic, D- or L-form, preferably in L-form. Preferably, the naturally occurring amino acid is a proteinogenic amino acid.
Preferably, AA comprises three histidine amino acids. Preferably, AA consists of histidine and glutamic acid. Preferably, AA is represented by the formula -His-Glu-His-Glu-His-Glu-, wherein at least one of said amino acid residues is a D-amino acid, or is represented by the amino acid sequence HEHEHE, wherein all amino acids are L-amino acids (SEQ ID NO: 1). Accordingly the compound or pharmaceutically acceptable salt thereof may comprise the aforesaid amino acid sequence bound to the rest of the molecule via C- and N-terminus and preferably is derived from the following free polypeptide sequence: EHEHEH (SEQ ID NO:2).
Preferably, the C-terminus or the N-terminus, preferably the C-terminus, of AA forms an amide bond with (A), preferably with histidine comprised in AA.
Accordingly, in a preferred embodiment the (A)-xi comprises the following partial structure with histidine as part of AA and xi bound to (A):
wherein the dashed line indicates the attachment to the rest of AA comprised in xi.
In a preferred embodiment, the spacer xi bears the following structure: -AA-[b”-e-b”’]n-b””-d1-, wherein b” is -C(O)- or -N(H)-, preferably forming an amide bond with AA; wherein e represents a residue selected from the group consisting of an Ci-8-alkylene wherein one or more -CH2- moieties may optionally be replaced by one or more -O-, -S-, -C(O)NH- - C(O)N(CI-6 alkyl), -C(O)O-, succinimide, triazole.
wherein b’” is selected from the group consisting of -NH-, and -C(O)-; wherein b”” is selected from the group consisting of -C(O)-, and -NH-; and wherein b’” and b”” or a terminus of AA, preferably the N-terminus of AA, and b”” together form an amide group; wherein d1 is -[CH2]P-, wherein p is 1 or 2, in particular 2; and wherein n is 0 or 1, preferably 0.
Preferably, b” is C(O).
Preferably e is without any replacement, or with one, two or three replacements, preferably one replacement, especially with triazole, preferably wherein e is a residue selected from the group consisting of an unsubstituted Cns-alkylene, Ci-8 alkylene, where one -CH2- is replaced by triazole, C4-8 alkylene (especially Ce alkylene), where one -CH2- is replaced by triazole, -CH2- (O-CH2-CH2)2-CH2-, -(CH2)2-(O-CH2-CH2)2-, -(CH2)3-O-CH2-CH2-O-CH2-, -CH2-O-(CH2)6-, -(CH2)2-O-(CH2)5-, -(CH2)3-O-(CH2)4-, -(CH2)4-O-(CH2)3-, -(CH2)5-O-(CH2)2-, -(CH2)6-O- CH2-, -CH2-(O-CH2-CH2)2-, -(CH2)2-O-CH2-CH2-O-CH2-, -CH2-O-(CH2)5-, -(CH2)2-O- (CH2)4-, -(CH2)3-O-(CH2)3-, -(CH2)4-O-(CH2)2-, -(CH2)5-O-CH2-, -CH2-O-CH2-CH2-O-CH2-, -CH2-O-(CH2)4-, -(CH2)2-O-(CH2)3-, -(CH2)3-O-(CH2)2-, -(CH2)4-O-CH2-, -CH2-O-(CH2)3-, - (CH2)2-O-(CH2)2-, -(CH2)3-O-CH2-, -CH2-O-(CH2)2-, -(CH2)2-O-CH2-, and -CH2-O-CH2-, in particular a residue selected from the group consisting of a butylene residue, a pentylene residue, a hexylene residue, or C4-8 alkylene, where one -CH2- is replaced by triazole;
In particular e is represented by the structure
wherein the dashed line with the star represents attachment to b“ and the dashed line without star represents attachment to b‘“. Preferably, b’” is -NH-. Preferably b’” is -C(O)-. Preferably n is 1. Preferably, b’” and b”” or -in case n=0- a terminus of AA, preferably the N-terminus of AA, and b”” together form an amide group.
In a more preferred embodiment, the spacer xi bears the following structure:
-AA-[C(O)-e-NH]n-C(O)-(CH2)p- wherein e is C4-8 alkylene, where one -CH2- is replaced by triazole, in particular C , alkylene, where one -CH2- is replaced by triazole; wherein n is 0 or 1, in particular 1; and wherein p is 1 or 2, in particular 2.
In formula (I) X2 is a spacer or a chemical single bond connecting (B) and (C). Preferably, X2 is a spacer. The spacer X2 preferably is rather hydrophilic.
In a preferred embodiment, the spacer X2 bears the following structure:
-d2-e-[f-e’]m- wherein d2 is -[CH2]r-, wherein r is 1 or 2, in particular 2; and wherein e is selected from the group consisting of -C(O)-NH-, -NH-C(O)-, -C(O)-O-, -O-C(O)-, -NH-C(O)-NH-, -NH-C(S)- NH-,
wherein one of the wavy lines indicates the conjugation site to d2 and the other wavy line indicates the conjugation site to f, in particular wherein e is -C(O)-NH-;
Preferably e is -C(O)NH-.
Preferably each f independently represents a residue selected from the group consisting of an Ci-io-alkylene wherein one or more -CH2- moieties may optionally be replaced by -O- or -NH- , and wherein f is unsubstituted or substituted with one or more groups independently selected from the group consisting of -NH2, -COOH and R3a, wherein R3a is selected from the group consisting of -(CH2)2-COOH, -(CH2)4-NH2, -(CH2)4- N+(CH3)3 + X , -CH2-COOH,-CH2-SH, -CH2-SO3H, and
wherein X' is a pharmaceutically acceptable negatively charged counterion; preferably wherein f is selected from the group consisting of -CH2-(O-CH2-CH2)2-CH2-, - (CH2)2-(O-CH2-CH2)2-, -(CH2)2-(CH2-CH2-O)2-(CH2)2-, -(CH2)3-(CH2-CH2-O)2-CH2-, -(CH2- CH2-O)3-CH2-, -(CH2)2-(CH2-CH2-O)2-CH2-, -(CH2)2-(CH2-CH2-NH)2-(CH2)2-, -(CH2)3- (CH2-CH2-NH)2-CH2-, -(CH2-CH2-NH)3-CH2-, -(CH2)2-(CH2-CH2-NH)2-CH2-, -(CH2)3-O- CH2-CH2-O-CH2-, -CH2-O-(CH2)6-, -(CH2)2-O-(CH2)5-, -(CH2)3-O-(CH2)4-, -(CH2)4-O- (CH2)3-, -(CH2)5-O-(CH2)2-, -(CH2)6-O-CH2-, -CH2-(O-CH2-CH2)2-, -(CH2)2-O-CH2-CH2-O- CH2-, -CH2-O-(CH2)5-, -(CH2)2-O-(CH2)4-, -(CH2)3-O-(CH2)3-, -(CH2)4-O-(CH2)2-, -(CH2)5-O- CH2-, -CH2-O-CH2-CH2-O-CH2-, -CH2-O-(CH2)4-, -(CH2)2-O-(CH2)3-, -(CH2)3-O-(CH2)2-, - (CH2)4-O-CH2-, -CH2-O-(CH2)3-, -(CH2)2-O-(CH2)2-, -(CH2)3-O-CH2-, -CH2-O-(CH2)2-, - (CH2)2-O-CH2-, -CH2-O-CH2-, -(CH2)3-(O-CH2-CH2)2-CH2-, -(CH2)2-(O-CH2-CH2)2-(CH2)2, - CH2-(O-CH2-CH2)2-(CH2)3, -CH2-(O-CH2-CH2)3-, -(CH2)2-(O-CH2-CH2)2-CH2-, -CH2-(O-
CH2-CH2)2-(CH2)2-, and -(CH2)3-(O-CH2-CH2)2-, CH2-(NH-CH2-CH2)2-CH2-, -(CH2)2-(NH- CH2-CH2)2-, -(CH2)3-NH-CH2-CH2-NH-CH2-, -CH2-NH-(CH2)6-, -(CH2)2-NH-(CH2)5-, - (CH2)3-NH-(CH2)4-, -(CH2)4-NH-(CH2)3-, -(CH2)5-NH-(CH2)2-, -(CH2)6-NH-CH2-, -CH2- (NH-CH2-CH2)2-, -(CH2)2-NH-CH2-CH2-NH-CH2-, -CH2-NH-(CH2)5-, -(CH2)2-NH-(CH2)4-, - (CH2)3-NH-(CH2)3-, -(CH2)4-NH-(CH2)2-, -(CH2)5-NH-CH2-, -CH2-NH-CH2-CH2-NH-CH2-, - CH2-NH-(CH2)4-, -(CH2)2-NH-(CH2)3-, -(CH2)3-NH-(CH2)2-, -(CH2)4-NH-CH2-, -CH2-NH- (CH2)3-, -(CH2)2-NH-(CH2)2-, -(CH2)3-NH-CH2-, -CH2-NH-(CH2)2-, -(CH2)2-NH-CH2-, -CH2- NH-CH2-, -(CH2)3-(NH-CH2-CH2)2-CH2-, -(CH2)2-(NH-CH2-CH2)2-(CH2)2, -CH2-(NH-CH2- CH2)2-(CH2)3, -CH2-(NH-CH2-CH2)3-, -(CH2)2-(NH-CH2-CH2)2-CH2-, -CH2-(NH-CH2-CH2)2- (CH2)2-, -(CH2)3-(NH-CH2-CH2)2-, -CH2-O-(CH2)8-, -(CH2)2-O-(CH2)7-, -(CH2)3-O-(CH2)6-, - (CH2)4-O-(CH2)5-, -(CH2)5-O-(CH2)4-, -(CH2)6-O-(CH2)3-, -(CH2)7-O-(CH2)2-, -(CH2)8-O- CH2-, -CH2-O-(CH2)7-, -(CH2)2-O-(CH2)6-, -(CH2)3-O-(CH2)5-, -(CH2)4-O-(CH2)4-, -(CH2)5-O- (CH2)3-, -(CH2)6-O-(CH2)2-, -(CH2)7-O-CH2-, -CH2-NH-(CH2)8-, -(CH2)2-NH-(CH2)7-, - (CH2)3-NH-(CH2)6-, -(CH2)4-NH-(CH2)5-, -(CH2)5-NH-(CH2)4-, -(CH2)6-NH-(CH2)3-, -(CH2)7- NH-(CH2)2-, -(CH2)8-NH-CH2-, -CH2-NH-(CH2)7-, -(CH2)2-NH-(CH2)6-, -(CH2)3-NH-(CH2)5- , -(CH2)4-NH-(CH2)4-, -(CH2)5-NH-(CH2)3-, -(CH2)6-NH-(CH2)2-, -(CH2)7-NH-CH2-, - CH(NH2)-CH2-, -CH2-CH(NH2)-, -CH(COOH)-CH2-, -CH2-CH(COOH)-, and -CH(R3)-, in particular f is a residue selected from the group consisting of -(CH2)2-(O-CH2-CH2)2-, -CH2- (O-CH2-CH2)2-CH2-, and -(CH2)3-O-CH2-CH2-O-CH2-, especially -(CH2)2-(O-CH2-CH2)2-; wherein each e” is independently selected from the group consisting of a chemical bond, -NH-
C(O)-, -C(O)-NH-, -C(O)-O- and -O-C(O)-, -NH-C(O)-NH-, -NH-C(S)-NH-, -C(O)-N(CH3)-,
-N(CH3)-C(O)-, -NH-C(S)-, -C(S)-NH-,
wherein one of the wavy lines indicates the conjugation site to f and the other wavy line indicates the conjugation site to the at least one dye moiety (C), in particular wherein e” is - NH-C(O)-; and
wherein m indicates an integer from 0 to 8, preferably 0 to 4, even more preferably 0 to 2, even more preferably 0 or 1 and in particular 1.
In a more preferred embodiment, the spacer x2 bears one of the following structures:
-(CH2)t-C(O)-NH-(CH2)u-(O-CH2-CH2)v-(CH2)w-e”-, or -(CH2)t-C(O)-NH-(CH2-CH2-O)v-CH2-e”- wherein t is 1 or 2, in particular 2; wherein u is an integer from 1 to 10, preferably from 1 to 3, in particular 2; wherein v is an integer from 0 to 3, in particular 2; wherein w is an integer from 0 to 2, in particular 0;
In an even more preferred embodiment, the spacer y bears one of the following structures: -(CH2)2-C(O)-NH-(CH2)2-(O-CH2-CH2)2-e”-
-(CH2)2-C(O)-NH-(CH2)2-(O-CH2-CH2)2-NH-C(O)-CH2-(O-CH2-CH2)n’-O-CH2-e”-,
-(CH2)2-C(O)-NH-(CH2)2-(O-CH2-CH2)2-NH-[C(O)-CH((CH2)2COOH)-NH]n ”-C(O)-
CH((CH2)2COOH)-e”-,
-(CH2)2-C(O)-NH-(CH2)2-(O-CH2-CH2)2-NH-[C(O)-CH((CH2)4NH2)-NH]n ”-C(O)-
CH((CH2)4NH2)-e”-, or
-(CH2)2-C(O)-NH-(CH2)2-(O-CH2-CH2)2-NH-[C(O)-CH((CH2)4N+(CH3)3)-NH]n ”-C(O)-
CH((CH2)4N+(CH3)3)-e”- + X , wherein n’ is an integer from 1 to 3; wherein n” is an integer from 0 to 2; wherein X' is a pharmaceutically acceptable negatively charged counterion; and wherein each e” is independently selected from the group consisting of a chemical bond, -NH- C(O)-, -C(O)-NH-, -C(O)-O- and -O-C(O)-, -NH-C(O)-NH-, -NH-C(S)-NH-, -C(O)-N(CH3)-, -N(CH3)-C(O)-, -NH-C(S)-, -C(S)-NH-,
wherein one of the wavy lines indicates the conjugation site to f and the other wavy line indicates the conjugation site to the at least one dye moiety (C), in particular wherein e” is - NH-C(O)-.
In particular X2 is represented by formula -(CH2)2-C(O)-NH-(CH2)2-(O-CH2-CH2)2-e”-.
The labeling as spcified herein may comprise a (further) step of administration of a radioactively labeled complex of the compound of formula (I), preferably at least two days before administration of the compound of formula (I). It is, however, also envisaged to use a non-radioactively labeled complex of the compound of formula (I).
The radioactively labeled complex comprises
(a) the compound having a chemical structure of formula (I) as defined above and below, or a pharmaceutically acceptable salt thereof; and
(b) at least one radiometal, preferably selected from the group consisting of 89Zr, 44Sc, i nIn, 90Y, 67Ga, 68Ga,177Lu, "mTc, 82Rb, 64Cu, 67Cu, 153Gd, 155Gd, 157Gd, 213Bi, 225 Ac and 59Fe, in particular 68Ga.
In accordance, the non-radioactively labeled complex comprises
(a) the compound having a chemical structure of formula (I) as defined above and below, or a pharmaceutically acceptable salt thereof; and
(b) at least one non-radioactive metal ion, wherein said metal is preferably selected from the group consisting of Zr, Sc, In, Y, Ga, Lu, Tc, Rb, Cu, Gd, Bi, Ac and Fe, in particular is Ga.
The complex preferably comprises one or more radiometal(s), preferably one radiometal.
A “radiometal” as used in the context of the present invention may be understood in the broadest sense as any radioactive metal or radioactive metal ion, i.e., a metal or metal ion that emits
radioactive emission. It may be a metal or metal ion that is typically radioactive or a radioactive isotope of a metal that also has non-radioactive isotopes. Exemplarily, a radiometal may be a radioactive isotope of gallium (Ga) (e.g., 67Ga, 68Ga), copper (e.g., 64Cu, 67Cu), iron (e.g., 59Fe), zirconium (e.g., 89Zr), scandium (e.g., 44Sc), indium (e.g., H 1In), yttrium (e.g., 90Y) rubidium (e.g., 82Rb), cobalt (e.g., 60Co), lutetium (e.g., 177Lu), gadolinium (e.g., 153Gd, 155Gd, 157Gd), bismuth (e.g., 213Bi), strontium (e.g., 90Sr), actinium (e.g., 225 Ac), or technetium (e.g., 99mTc). Preferably, the radiometal is a radioactive isotope of 67Ga, 68Ga, i nIn, 90Y, 177Lu, 64Cu, 67Cu, 153Gd, 155Gd, 157Gd, 89Zr, 44Sc, "mTc, 213Bi, 225 Ac, 59Fe or 82Rb, more preferably 68Ga, 64Cu, 89Zr, 44Sc, or 82Rb, even more preferably 68Ga or 64Cu, in particular 68Ga. The person skilled in the art will know several examples for chelator moieties suitable for complexing each of the aforementioned radiometals and may select the chelator moiety accordingly. Exemplarily, a chelator moiety suitable for complexing "mTc or 82Rb, may or may not differ from that suitable for complexing 68Ga or 64Cu.
The radiometal may be obtained commercially of, may be obtained from nature or may be obtained from a cyclotron. Preferably, the radiometal is obtained from a gallium-68 generator or a cyclotron, in particular from a gallium-68 generator or a cyclotron that is localized near the site where the composition according to the present invention is mixed and where optionally also the diagnosis in vivo and/or in vitro is performed. Particularly preferably, the radiometal 68Ga is obtained from a gallium-68 generator, thus, a device used to extract the positronemitting isotope 68Ga of gallium from a source of decaying germanium-68. The parent isotope 68Ge is known to have a half-life of 271 days and may thus be shipped to the site where the gallium-68 generator is located.
Preferably, the radiometal is such that has a half-life of no longer than four days, more preferably of no longer than one day, even more preferably no longer than 12 h, even more preferably, not more than 6 h, even more preferably not more than 3 h, even more preferably not more than 2.5 h, even more preferably not more than 120 min, even more preferably not more than 100 min, even more preferably not more than 80 min, in particular not more than 70 min.
The radiometal may be obtained from any source suitable for this purpose. The radiometal may be obtained and isolated from nature or artificially be generated such as, e.g., 68Ga from a
gallium-68-generator. The person skilled in the art will know how to obtain the respective radiometal.
The non-radioactive metal may be any metal ion not emitting radiactivity deemed appropriate by the skilled person, selected according to the intended use. Preferably, the non-radioactive metal is at least one non-radioactive isotope of a radiometal as specified herein above.
In particular, the chelator moiety (B) is a chelator suitable to complex 68Ga, "mTc or 82Rb, in particular suitable to complex 68Ga in aqueous environment.
Gallium-68 (68Ga) has a half-life of approximately 68 minutes and is thus rather inconvenient for longer transports. Therefore, it may typically be generated nearby the site where it is complexed with the compound of the present invention or a pharmaceutically acceptable salt thereof and administered to a patient in vivo and/or a sample in vitro.
Therefore, in a preferred embodiment, the chelator moiety (B) is a 68Ga-chelator moiety, preferably a 68Ga-chelator moiety.
The chelator moiety (B) is preferably selected from the following group:
wherein in each of the structures one of the wavy lines indicates the conjugation site to the at least one motif specifically binding to cell membranes of cancer cells (A) via spacer xi as defined herein, and the other wavy line indicates the conjugation site to the dye moiety (C), preferably via the spacer X2 as defined herein.
In particular, the chelator moiety is
When this chelator is used, b”” preferably is -C(O)-, d1 and d2 preferably are each -(CH2)2-, and e’ preferably is -C(O)-NH or -C(O)-O-, especially -C(O)NH-. Therefore, the chelator moiety is conjugated with two -C(O)-(CH2)2-# moieties, wherein these are conjugated at the position indicated by “#” with the binding sites of the aforementioned chelator moiety indicated by the wavy lines. Then, this residue is also designated as “HBED-CC”.
It will be noted that several of these “68Ga-chelator moieties” exemplarily listed above, may also serve as structures complexing one or more other radiometal(s) such as, e.g., 64Cu, in particular in aqueous environment of approximate neutral pH.
Preferably, neither the motif specifically binding to cell membranes of cancer cells (A) nor the complexed radiometal nor the chelator moiety (B) quench the intensity of the fluorescence
signal obtainable from the dye moiety (C) at its emission maximum in an aqueous environment of approximately neutral pH (i.e., pH 6-8, in particular 6.5-7.5) by more than 50%.
Preferably, the dye moiety (C) is suitable for emit light in an aqueous environment of approximately neutral pH, i.e., pH 6-8, in particular 6.5-7.5, in particular pH 7.0-7.5.
In a preferred embodiment, the dye moiety (C) is a fluorescent dye moiety having an emission maximum in the range from 400 nm to 1000 nm, as described above.
X1 and X4 are independently selected from the group consisting of -N=, -N(R5)=, and -C(R6)=;
X2 and X3 are independently selected from the group consisting of O, S, Se, N(R5), and C(R6R7), preferably both are C(CHs)2;
Y is a linker connecting the two moieties of (C) and permitting electron delocalization between said moieties, wherein Y optionally comprises a group (L-)cZ0; a and b are independently selected from the group consisting of 1, 2, and 3; each R1 and each R2 is independently (L-)CZ, (L-)CZ° or H; and two adjacent R1 and/or two adjacent R2 can also form an aromatic ring, which is optionally substituted with one or more (L-)cZ or (L-)cZ°;
R3, R4, R5, R6, R7, R9 are independently selected from the group consisting of (L-)cZ, (L-)CZ°, and H; each c is independently 0, or 1 ;
each L is independently
NHC O) 1, or a Ci -io alkylene group, which is optionally interrupted and/or terminated by one or more of -O-, -S-, -C(O)-, -C(O)O-, -OC(O)-, -C(O)NH-, -NHC(O)O-, and T1;
T1 is phenyl, naphthyl, indenyl, indanyl, tetralinyl, decalinyl, adamantyl, C3-7 cycloalkyl, 3 to 7 membered heterocyclyl, or 7 to 11 membered heterobicyclyl, wherein T1 is optionally substituted with one or more substituents selected from the group consisting of halogen, CN, C(O)R8, COOR8, OR8, C(O)N(R8R8a), S(O)2N(R8R8a), S(O)N(R8R8a), S(O)2R8, N(R8)S(O)2N(R8aR8b), SR8, N(R8R8a), NO2; OC(O)R8, N(R8)C(O)R8a, N(R8)S(O)2R8a, N(R8)S(O)R8a, N(R8)C(O)N(R8aR8b), N(R8)C(O)OR8a, OC(O)N(R8R8a), oxo (=0), where the ring is at least partially saturated, or C1-6 alkyl, wherein C1-6 alkyl is optionally substituted with one or more halogen, which are the same or different; each Z is independently H, halogen, CN, C(O)R8, C(O)OR8, C(O)O’ OR8, C(O)N(R8R8a), S(O)2OR8, S(O)2O’, S(O)2N(R8R8a), S(O)N(R8R8a), S(O)2R8, S(O)R8, N(R8)S(O)2N(R8aR8b), SR8, N(R8R8a), NO2; P(O)(OR8)2, P(O)(OR8)O’, OC(O)R8, N(R8)C(O)R8a, N(R8)S(O)2R8a, N(R8)S(O)R8a, N(R8)C(O)N(R8aR8b), N(R8)C(O)OR8a, or OC(O)N(R8R8a);
R8, R8a, R8b are independently selected from the group consisting of H, or C1-6 alkyl, wherein C1-6 alkyl is optionally substituted with one or more halogen, which are the same or different; Z° is a chemical bond connecting (C) to x2 or to (B) in case x2 is a chemical single bond; provided that one of R1, R2, R3, R4, R5, R6, R7, R9 is (L-)CZ° or that Y comprises (L-)CZ°.
Accordingly, one of the residues R1, R2, R3, R4, R5, R6, R7 serve as connecting group, atom or bond (L-)CZ° of dye moiety (C) to x2 or (B). Preferably R3 or R4 represent (L-)CZ°. More preferably, R3 is (L-)CZ°.
Preferably, in formula (C) X1 and X4 are the same and preferably C(R6), more preferably CH.
Preferably, in formula (C) X2 and X3 are the same and preferably C(R6R7), more preferably R6 and R7 are the same and even more preferably L-Z with L = C1-10 alkylene, and even more preferably L = CH2, and Z = H.
Preferably, R9 is H.
Preferably, in formula (C) Y does not comprise (L-)CZ° and preferably Y is
wherein g is 1, 2, 3, or 4 (preferably 2 or 3, more preferably 3) and each R9a is (L-)CZ, or H; and two R9a can also form a carbocyclic ring having 5, 6, or 7 carbon atoms or a 4 to 7 membered heterocylic ring; each c is independently 0, or 1 ; each L is independently
NHC O) 1, or a Ci -io alkylene group, which is optionally interrupted and/or terminated by one or more of -O-, -S-, -C(O)-, -C(O)O-, -OC(O)-, -C(O)NH-, -NHC(O)O-, and T1;
T1 is phenyl, naphthyl, indenyl, indanyl, tetralinyl, decalinyl, adamantyl, C3-7 cycloalkyl, 4 to 7 membered heterocyclyl, or 7 to 11 membered heterobicyclyl, wherein T1 is optionally substituted with one or more substituents selected from the group consisting of halogen, CN, C(O)R8, COOR8, OR8, C(O)N(R8R8a), S(O)2N(R8R8a), S(O)N(R8R8a), S(O)2R8, N(R8)S(O)2N(R8aR8b), SR8, N(R8R8a), NO2; OC(O)R8, N(R8)C(O)R8a, N(R8)S(O)2R8a, N(R8)S(O)R8a, N(R8)C(O)N(R8aR8b), N(R8)C(O)OR8a, OC(O)N(R8R8a), oxo (=0), where the ring is at least partially saturated, or C1-6 alkyl, wherein C1-6 alkyl is optionally substituted with one or more halogen, which are the same or different;
R8, R8a, R8b are independently selected from the group consisting of H, or C1-6 alkyl, wherein C1-6 alkyl is optionally substituted with one or more halogen, which are the same or different; each Z is independently H, halogen, CN, C(O)R8, C(O)OR8, C(O)O’ OR8, C(O)N(R8R8a), S(O)2OR8, S(O)2O’, S(O)2N(R8R8a), S(O)N(R8R8a), S(O)2R8, S(O)R8, N(R8)S(O)2N(R8aR8b), SR8, N(R8R8a), NO2; P(O)(OR8)2, P(O)(OR8)O’, OC(O)R8, N(R8)C(O)R8a, N(R8)S(O)2R8a, N(R8)S(O)R8a, N(R8)C(O)N(R8aR8b), N(R8)C(O)OR8a, or OC(O)N(R8R8a).
Preferably, in formula (C) a and b are the same and preferably 1, more preferably with R1 and R2 = so3;
Preferably, in formula (C) a and b are the same and 2, preferably wherein two adjacent R1 and two adjacent R2 form a phenyl ring.
Preferably, in formula (C) one of R3 and R4 is (L-)cZzo and the other is (L-)CZ with L- = Cnio alkylene and Z = H or SOs' and c preferably = 1.
Preferably, (L-)CZ° is Cnio alkylene (c=l, Z° a chemical bond), preferably C3-7 alkylene, more preferably C5 alkylene, connecting (C) to X2 or to (B) in case X2 is a chemical single bond.
Preferably, (L-)CZ is C1-10 alkylene, preferably C3-7 alkylene, more preferably C4 alkylene, c is 1 and Z is SCh'.
In a highly preferred embodiment, the dye moiety (C) is a fluorescent dye moiety selected from the group consisting of the following structures:
wherein X' is a pharmaceutically acceptable negatively charged counterion; wherein Y+ is a pharmaceutically acceptable positively charged counterion; and
wherein the wavy line indicates the conjugation site to the rest of the compound of the present invention.
Preferably the dye is IRDye800CW.
A pharmaceutically acceptable negatively charged counterion X' may be understood in the broadest sense as laid out above.
Likewise, also a pharmaceutically acceptable positively charged counterion Y+ may have any valency. Therefore, Y+ may exemplarily have a charge of +1, +2, +3 or +4, preferably of +1 or +2. Y+ may be any pharmaceutically acceptable positively charged ion. Preferably, the ion is such well-soluble in aqueous liquids. Exemplarily, Y+ may be selected from the group consisting of a cation of an alkali metal (e.g., Na+, K+, Li+), a cation of an alkaline earth metal (e.g., Mg2+, Ca2+), Al3+, NH4+, H+ and a cation of an organically bound amine. Further, it will be understood that the counterion typically depends on the surrounding liquids such as those comprised in the buffer the compound is dissolved in and the body fluids after injection in vivo. In vivo, extracellularly, one of the main, but not sole positively charged counterions is Na+.
Preferably, the wavy line indicates the conjugation site to the spacer X2. More preferably, the wavy line indicates the conjugation site to e”.
Accordingly, in a highly preferred embodiment, the compound of the present invention has the following chemical structure:
The compound of formula (I) may be obtained from rational chemical synthesis. Exemplary routes for the preparation of compounds or pharmaceutically acceptable salts thereof of the present invention are described in US 2015/0110715 Al.
The term "labeling agent" is understood by the skilled person. Preferably, the term relates to a compound labeling cancerous tissue, more preferably, specifically labeling cancerous tissue, as specified herein below. The term "specific labeling" of a cancerous tissue preferably relates to a labeling which enables differentiation between cancerous tissue and non-cancerous tissue, such as e.g. cancer-adjacent tissue. Preferably, said differentiation is enabled in vitro and/or in vivo, more preferably in vivo. Thus, preferably, the labeling enables differentiation between adjacent cancerous and non-cancerous tissues, e.g. in an operation area. Thus, specific labeling does not necessarily have to enable differentiation between cancerous tissue and any non- cancerous tissue in a subject; it, preferably, is sufficient if cancerous tissue can be differentiated from non-cancerous tissue in the vicinity of cancerous tissue. Thus, the differentiation may preferably be between cancerous tissue and non-cancerous tissue in the abdominal cavity, more preferably within at most 10 cm, more preferably at most 5 cm, even more preferably at most 2 cm from a cancerous tissue. Also, it preferably is not required that differentiation is enabled from easily identifiable intact structures in the subject, e.g. the kidney, the liver, and the like. The differentiation is preferably made by visual inspection, e.g. by a medical practitioner, or may be assisted or made by an optical device adapted to detect the label comprised in the
labeling compound. Preferably, specific labeling is labeling of cancerous tissue, preferably of cancer cells, more intensely by a factor of at least 2, more preferably at least 5, even more preferably at least 10, still more preferably at least 25, more preferably at least 100, compared to non-cancerous tissue. Thus, the labeling agent preferably has an affinity for cancerous tissue, preferably for cancer cells, being at least 5fold, more preferably at least lOfold, even more preferably at least lOOfold, still more preferably at least 250fold, most preferably at least lOOOfold higher than for non-cancerous tissue. Thus, preferably, the labeling agent specifically labels PSMA expressing cancerous tissue, preferably PSMA expressing cancer cells, all as specified herein below; in such case, the dissociation constant (Ka) of the labeling agent in its binding to PSMA is preferably at most 10'6 mol/L, more preferably at most 10'7 mol/1, most preferably at most 10'8 mol/L; also preferably, the Ka of the labeling agent in its binding to any non-PSMA cell constituent preferably is at least 10'6 mol/L, more preferably at least 10'5 mol/1, most preferably at least 10'4 mol/L. Preferably, the dissociation constant is determined by a method known to the skilled person, e.g. a surface plasmon resonance based method or by competition experiments with a PSMA ligand having a known affinity, e.g. in cell-basd inhibition experiments. The labeling agent may be the compound having the chemical structure of formula (I) or a metal chelate thereof, in particular a radiometal chelate thereof. Preferably, the labeling agent is a compound having the chemical structure of formula (I) or a pharmaceutically acceptable salt thereof, i.e. preferably is said compound not comprising a chelated metal. As the skilled person understands, detecting will preferably be based on the physical properties of the dye moiety in case the labeling agent is not a metal chelate or is administered as a non-metal chelate; in case the labeling agent is a metal chelate or is administered as a metal chelate, detecting may also be based on physical properties of the chelated metal, exclusively or in addition, preferably in addition. Thus, the labeling agent may e.g. be a chelate of a radiometal and detecting may be based on fluorescence of the dye moiety, e.g. in areas easily accessible, and may be assisted by detection of radioactivity from the chelated radiometal to detect small numbers of residual cancer cells and/or cancer cells in areas which are not or not easily accessible.
The term "cancer", as used herein, relates to a disease of an animal, including man, characterized by uncontrolled growth by a group of body cells (“cancer cells”). This uncontrolled growth may be accompanied formation of a cell mass of cancer cells (tumor), by intrusion into and destruction of surrounding tissue (infiltration) and possibly spread of cancer cells to other locations in the body (metastasis). Preferably, also included by the term cancer is
a recurrence of a cancer (relapse). Thus, preferably, the cancer is a solid cancer, including a primary tumor, a metastasis, and a relapse thereof. The cancer may, however, also be a nonsolid cancer. Preferably, the cancer is a prostate-specific membrane antigen (PSMA) expressing cancer, preferably a PSMA overexpressing cancer. Also preferably, the cancer is a cancer comprising PSMA-expressing neovasculature. Thus, the cancer preferably is selected from the list consisting of prostate cancer, breast cancer, kidney cancer, bladder cancer, ovarian cancer, or non-small cell lung cancer (NSCLC), more preferably is prostate cancer, still more preferably PSMA overexpressing prostate cancer.
The term "cancerous tissue", as used herein, relates to any tissue known or suspected to comprise cancer cells as specified herein above. Preferably, the fraction of cancer cells in the cancerous tissue is at least 10%, preferably at least 20%, more preferably at least 30%, even more preferably at least 40%, most preferably at least 50%. Thus, the cancerous tissue preferably is a primary tumor, an infiltration, a metastasis, a relapse, or a lymph node, or a sample thereof.
The term "detecting" is in principle understood by the skilled person and preferably relates to establishing the presence of cancerous tissue, preferably of cancer cells, in a subject and/or of cancer cells in a sample. Preferably, determining is identifying cancerous tissue in a subject, preferably in a surgical operation zone. Also preferably, detecting is differentiating between cancerous tissue and non-cancerous tissue. Thus, the detecting may be a step aiding a surgeon in deciding which tissue or parts thereof shall be excised and which shall not. As the skilled person will understand, labeling by the labeling compound may be, but preferably is not, the only basis for said decision. Preferably, the decision takes into account further parameters such as tissue morphology, tissue density, proximity to the primary tumor, location in relation to afferent or efferent blood and/or lymph vessels, vascularization, and the like.
As will be understood in light of the description herein, the detecting may comprise additional steps and/or activities, such as illuminating tissue suspected to be cancerous tissue with radiation of a wavelength absorbed and/or causing radiation from the labeling compound, preferably causing fluorescence. Thus, preferably, in case the labeling compound is e.g. PSMA- 914, determining preferably further comprises illuminating tissue suspected to be cancerous tissue by radiation with a wavelength of from 600 nm to 850, preferably 700 nm to 780 nm, and/or detection of radiation, by eye and/or at a wavelength in the range of from 780 nm to 850
nm, preferably comprises illuminating tissue suspected to be cancerous tissue by radiation with a wavelength of about 774 nm, and/or detection of radiation of a wavelength of about 789 nm, in particular in case the dye moiety is IRDye800CW, and/or the labeling compound is PSMA- 914. As the skilled person understands, the selection of additional steps will depend on a variety of parameters, including in particular the dye moiety used in the labeling compound, the mode of administration of the labeling compound, the type of determining (e.g. in vivo or in vitro), and the like.
The term "subject", as referred to herein, relates to a vertebrate animal, preferably a mammal, in particular a livestock, companion, or laboratory animal. Most preferably, subject is a human. Preferably, the subject has been diagnosed to suffer from cancer, is suspected to suffer from cancer, or is known to be at risk of suffering from cancer, in particular a hereditary form of cancer. Preferably, the subject is a male, more preferably a male suffering from prostate cancer, even more preferably with a Gleason score of at least 5, more preferably at least 6, even more preferably at least 7, most preferably at least 8. Preferably, the cancer was diagnosed to warrant, preferably to require, surgical removal, preferably by partial or complete prostatectomy.
In view of the above, the term "labeling dose" relates to a dose of the labeling compound enabling labeling cancerous tissue, preferably, specifically labeling cancerous tissue as specified herein above, more preferably enabling differentiation between cancerous tissue and non-cancerous tissue as specified herein above. Preferably, the labeling dose is at least 200 pg, preferably at least 500 pg, more preferably at least 1 mg of labeling compound per subject. Preferably, the labeling dose is of from 0.2 mg to 100 mg, more preferably is of from 0.5 to 25 mg, still more preferably is of from 1 mg to 10 mg, most preferably is of from 1 mg to 5 mg, of said labeling compound per subject. Thus, the labeling dose may e.g. be about 0.2 mg, about 0.3 mg, about 0.4 mg, about 0.5 mg, about 0.6 mg, about 0.7 mg, about 0.8 mg, about 0.9 mg, about 1.0 mg, about 1.1 mg, about 1.2 mg, about 1.3 mg, about 1.4 mg, or about 1.5 mg, per subject. The aforesaid labeling doses preferably are labeling doses for human subjects and, preferably, for PSMA-914. Preferably, the labeling dose is administered independent of the subject's body mass, i.e. is determined without taking into account the subject's body mass. Thus, the labeling dose preferably is a unit dose for all subjects, preferably for all adult subjects also preferably for subjects in a body mass range of from 50 kg to 200 kg. The labeling dose may, however, also be adjusted to a subject's body mass; in such case, the dose preferably is of from 3 pg/kg body mass to 300 pg/kg body mass, more preferably of from 5 pg/kg body mass
to 100 pg/kg body mass, even more preferably of from 7,5 pg/kg body mass to 50 pg/kg body mass, still more preferably about 15 pg/kg body mass. Also preferably, the dose is a single dose, i.e. exactly one dose, preferably administered within a time frame of at least twelve hours, preferably at least one day, more preferably at least two days; thus, in the aforesaid time frame, preferably no further dose of labeling compound is administered. Preferably, said labeling dose is administered to the patient at of from 0.25 h to 48 h before determining, preferably before start of surgery. More preferably, said labeling dose is administered to the patient at of from 0.5 h to 24 h before determining, still more preferably of from 0.75 h to 12 h before determining, even more preferably at about 1 h before determining, most preferably 1 h before determining, preferably before start of surgery.
Preferably, the labeling further comprises fine-tuning the labeling dose. Thus, the labeling may comprise a further step of administration of a radioactively labeled complex of the labeling compound, preferably at least two days before administration of the labeling compound, and further comprises increasing the labeling dose of the labeling compound in case the signal of said radioactively labeled complex of the labeling compound in tumor or relapse tissue is found to be weak compared to a reference.
As specified herein above, the radioactively labeled complex comprises
(a) the compound having a chemical structure of formula (I) as defined above and below, or a pharmaceutically acceptable salt thereof; and
(b) a radiometal, preferably selected from the group consisting of 89Zr, 44Sc, i nIn, 90Y, 67Ga, 68Ga,177Lu, "mTc, 82Rb, 64Cu, 67Cu, 153Gd, 155Gd, 157Gd, 213Bi, 225 Ac and 59Fe, in particular 68Ga.
Preferably, the dose of the labeling compound is at least 20fold, preferably at least 30fold, the dose of the dose of the radioactively labeled complex of the labeling compound preferably in case the signal of said radioactively labeled complex in tumor or relapse tissue is found to be above or within the average of a reference. Preferably, the signal of said radioactively labeled complex is determined on a primary tumor or part thereof; however, other methods of determination may be used, e.g. detectability of metastatic lymph nodes in a PET scan. In accordance with the above, the radioactively labeled complex of the labeling compound is preferably administered at a dose of from 2 pg per subject to 50 pg per subject, preferably of from 10 pg per subject to 30 pg per subject. The skilled person is aware that the dose of the radioactively labeled derivative is preferably adjusted or re-adjusted based on its specific
activity, preferably such that a pre-determined dose of radioactivity is administered to the subject. Thus, the dose of the labeling compound preferably is at least 20fold, preferably at least 30fold, the dose of the dose of the radioactively labeled complex of the labeling compound, preferably in case the signal of said radioactively labeled complex of the labeling compound in tumor or relapse tissue is found to be above or equal to a reference. Also preferably, the dose is least 50fold, preferably at least 75fold the dose of the dose of the radioactively labeled complex of the labeling compound, preferably in case the signal of said radioactively labeled complex of the labeling compound in tumor or relapse tissue is found to be below a reference. Also preferably, the dose is at most 30000fold, preferably at most lOOOOfold, more preferably at most lOOOfold the dose of the dose of the radioactively labeled complex of the labeling compound. Preferably, in particular in case the labeling compound is admininstered as a non-metal-complexed form while the fine-tuning is adjusted using a radioactively labeled complex of the labeling compound, the aforesaid ratios are determined on a molar basis.
The term “reference” refers to a signal or a value representing said signal, i.e. data correlating with a labeling intensity of a cancerous tissue with a radioactively labeled complex of the labeling compound. Thus, the reference may e.g. be a value of a radioactivity in a tissue, a blackening of a photographic film, and intensity value in a scintigram or a PET scan, or the like. Suitable references are known to the skilled person and may in particular be derived from a subject or a population of subjects known to have had specifically labeled cancerous tissue at a standard dose as specified herein above, or may be the average of a population of subjects; thus, the reference may in particular be obtained from a multitude of primary and/ or relapse prostate cancer samples. How to calculate a suitable reference value, in an embodiment, the average or median, is well known in the art. The population of subjects referred to before shall comprise a plurality of subjects, preferably at least 5, more preferably at least 10, even more preferably at least 50, more preferably at least 100, subjects. It is to be understood that the subject to be diagnosed by the method of the present invention and the subjects of the plurality of subjects used to derive a reference from are preferably of the same species. The skilled person knows howto establish a reference value, which may be a cut-off vale, an upper limit of normal, a median or average, a reference range, or the like.
Advantageously, it was found in the work underlying the present invention that the labeling compound, in particular PSMA-914, can be administered at a dose which enables specific in
vivo labeling of cancerous tissue, in particular prostate cancer tissue and, thereby, enables reliable excision of cancer tissue e.g. in surgery. Surprisingly, it was found that also cancerous tissue comprising a low fraction of cancer cells, such as metastases and in particular lymph nodes, can be specifically labeled, thus making conservative surgery possible while ensuring complete removal of cancer cells.
The definitions made above apply mutatis mutandis to the following. Additional definitions and explanations made further below also apply for all embodiments described in this specification mutatis mutandis.
The present invention further relates to a labeling compound for use for in vivo labeling of cancerous tissue in a subject, said labeling comprising administration of a labeling dose of the labeling compound to said subject; and to a labeling compound for use in a method of diagnosing of cancerous tissue in a subject, wherein said method comprises administration of a labeling dose of the labeling compound to said subject.
The term “diagnosing”, as used herein, refers to assessing the probability according to which a subject is suffering from a disease or condition or is at risk of developing a disease or condition. Accordingly, the method provides an aid for diagnosis since it might be necessary to further strengthen or confirm said diagnosis by, e.g., a medical practitioner. In particular, as will be understood by those skilled in the art, such an assessment, although preferred to be, may usually not be correct for 100% of the subjects to be diagnosed. The term, preferably, requires that a statistically significant portion of subjects can be identified as suffering from the disease. Whether a portion is statistically significant can be determined without further ado by the person skilled in the art using various well known statistic evaluation tools, e.g., determination of confidence intervals, p-value determination, Student's t-test, Mann- Whitney test, etc.. Details are found in Dowdy and Wearden, Statistics for Research, John Wiley & Sons, New York 1983. Preferred confidence intervals are at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%. The p-values are, in an embodiment, 0.2, 0.1, 0.05. It will be understood, moreover, that the methods of the present invention essentially provide an aid for diagnosis and may be included into or supplemented by other diagnostic measures. Diagnosing according to the present invention includes monitoring, confirmation, and classification of the relevant disease or its symptoms. Monitoring relates to keeping track of an already diagnosed disease, or a complication, e.g. to analyze the progression or regression of the disease, the influence of
a particular treatment on the progression of disease or complications arising during the disease period or after successful treatment of the disease. Confirmation relates to the strengthening or substantiating a diagnosis already established using other indicators or markers. Classification relates to allocating the diagnosis according to the strength or kind of symptoms into different classes, e.g. the stages for prostate carcinomas. Preferably, diagnosing cancer comprises identification of cancerous tissue, preferably of cancer cells, preferably in vivo in a subject, more preferably comprises intraoperative identification of cancerous tissue. Also preferably, diagnosing comprises identification of primary tumor, relapse, metastases and/or lymph nodes comprising cancer cells, more preferably comprises identification of metastases and/or lymph nodes comprising cancer cells, preferably is intraoperative identification of metastases and/or lymph nodes comprising cancer cells.
The present invention also relates to a method for labeling cancerous tissue in a subject, comprising administering a labeling dose of the labeling compound to said subject; to a method for identifying cancerous tissue in a subject comprising (a) labeling cancerous tissue by administering a labeling dose of the labeling compound to said subject; and (b) identifying labeled cancerous tissue in situ; as well as to a method for removing cancerous tissue from a subject, said method comprising (a) labeling cancerous tissue by administering a labeling dose of the labeling compound to said subject; (b) identifying labeled cancerous tissue in situ; and (c) removing said cancerous tissue.
The present invention also relates to a labeling compound accordig to formula (I) for use in a method of removing a cancerous tissue from a subject, said method comprising
(a) labeling cancerous tissue by administering a labeling dose of the labeling compound to said subject;
(b) identifying labeled cancerous tissue in situ; and
(c) removing said cancerous tissue.
The method of removing a cancerous tissue is an in vivo method and may comprise further steps not explicitly mentioned, e.g. diagnosing said cancer tissue, preferably according to a method as specified herein, and/or additional curative and/or preventive method steps, such as administration of radiation, chemotherapy, immunotherapy, and the like before, concomitant to, and/or after said removing of cancerous tissue.
The steps of (a) labeling cancerous tissue and (b) identifying labeled cancerous tissue have been described herein above. As the skilled person understands, labeled cancerous tissue may further be identified in vitro, e.g. in histopathological analysis of excised cancerous tissue. The method further comprises step (c) removing said cancerous tissue. This step may be accomplished in principle by any method deemed appropriate by the skilled person. As used herein, the expression "removing cancerous tissue" includes any and all methods leading to elimination of cancerous tissue from the subject. Said removing may be essentially concomitant to step (b), e.g. in surgical removal, e.g. excision, of cancerous tissue. The removing may, however, occur or be completed substantially later than step (b), e.g. in case the cancerous tissue is treated with label-guided irradiation, administration of chemotherapeutic agents, or the like. Preferably, step (c) comprises or is surgical removal of cancerous tissue.
The present invention also relates to a use of a labeling compound having the structure of formula (I) as specified herein above in the manufacture of a diagnostic composition for labeling cancerous tissue, preferably in vivo labeling cancerous tissue, wherein said diagnostic composition comprises a labeling dose of said labeling compound.
The present invention also relates to a diagnostic composition comprising of from 0.2 mg to 100 mg, preferably is of from 0.5 to 25 mg, more preferably is of from 1 mg to 10 mg, most preferably is of from 1 mg to 5 mg of a labeling compound having the structure of formula (I) as specified herein above.
The term "diagnostic composition" is, in principle, known to the skilled person. As referred to herein, the term relates to any composition of matter comprising at least the specified labeling compound as diagnostically active compound(s) and one or more excipient. The diagnostically active compound(s) can be present in liquid or dry, e.g. lyophilized, form. It will be appreciated that the form and character of the pharmaceutically acceptable excipient, e.g. carrier or diluent, is dictated by the amount of active ingredient with which it is to be combined, the route of administration, and other well-known variables. The excipient(s) must be acceptable in the sense of being compatible with the other ingredients of the formulation and being not deleterious to the recipient thereof. The excipient employed may include a solid, a gel, or a liquid. Exemplary of solid carriers are lactose, terra alba, sucrose, talc, gelatin, agar, pectin, acacia, magnesium stearate, stearic acid and the like. Exemplary of liquid carriers are phosphate buffered saline solution, physiological saline, Ringer's solutions, dextrose solution, and Hank's
solution, syrup, oil, water, emulsions, various types of wetting agents, and the like. Similarly, the carrier or diluent may include time delay material well known to the art, such as glyceryl mono-stearate or glyceryl distearate alone or with a wax. Said suitable carriers comprise those mentioned above and others well known in the art, see, e.g., Remington's Pharmaceutical Sciences, Mack Publishing Company, Easton, Pennsylvania. Thus, the excipient may in particular be water, an aqueous buffer (e.g., a Hepes, Tris, or phosphate buffer, a phosphate buffer), a pharmaceutically acceptable organic solvent (e.g., dimethyl sulfoxide (DMSO), ethanol, vegetable oil, paraffin oil) or a combination of two or more thereof.
Furthermore, the pharmaceutically acceptable carrier may contain one or more detergents, one or more foaming agents (e.g., sodium lauryl sulfate (SLS)/ sodium doceyl sulfate (SDS)), one or more coloring agents (e.g., TiCh, food coloring), one or more vitamins, one or more salts (e.g., sodium, potassium, calcium, zinc salts), one or more humectants (e.g., sorbitol, glycerol, mannitol, propylenglycol, polydextrose), one or more enzymes, one or more preserving agents (e.g., benzoic acid, methylparabene), one or more texturing agents (e.g., carboxymethyl cellulose (CMC), polyethylene glycol (PEG), sorbitol), one or more emulsifiers, one or more bulking agents, one or more glacing agents, one or more separating agents, one or more antioxidants, one or more herbal and plant extracts, one or more stabilizing agents, one or more polymers (e.g., hydroxypropyl methacrylamide (HPMA), polyethylene imine (PEI), carboxymethyl cellulose (CMC), polyethylene glycol (PEG)), one or more uptake mediators (e.g., polyethylene imine (PEI), dimethyl sulfoxide (DMSO), a cell-penetrating peptide (CPP), a protein transduction domain (PTD), an antimicrobial peptide, etc.) one or more antibodies, one or more sweeteners (e.g., acesulfame, an acesulfame salt (e.g., acesulfame potassium (acesulfame K), aspartame, cyclamate, saccharin, a saccharin salt (e.g., saccharin sodium (saccharin Na)), alitame, neotame, sucralose, dulcin, salt of aspartame-acesulfame, sorbitol, stevia, glycerol, inulin, mannitol, isomalt, maltitol, malto-oligosaccharide, lactitol, xylitol, glucin, neohesperidin dihydrochalcone, P-4000, brazzein, curculin, erythritol, glycyrrhizin, hydrogenated starch hydrolysates, luo han guo, mabinlin, miraculin, monatin, monellin, osladin, pentadin, tagatose, thaumatin), one or more counterstain dyes (e.g., fluorescein, fluorescein derivatives, Cy dyes, an Alexa Fluor dyes, rhodamine, quantum dots, etc.), one or more homeopathic ingredients one or more gustatory substances and/or one or more fragrances. The diagnostic composition will typically be formed by contacting the aforementioned component with another by any way known in the art. Preferably, the diagnostic composition is formed, thus, the above-referenced components are contacted with another prior to being
administered to a patient. The excipient(s) is/are preferably selected so as not to affect the biological activity of the combination.
The diagnostic composition is, preferably, administered systemically, preferably orally or parenterally, e.g. by intravenous administration, or is administered topically, preferably intra- tumorally; in case of cancer diagnostics, topical administration may be intratumoral or peritumoral, and/or topical at a site of tumor excision. Administration may, however, also be into a blood vessel, typically an artery, afferent to an intended site of effect, such as a tumor. However, depending on the nature of the formulation and the desired therapeutic application, the diagnostic composition may be administered by other routes as well.
In view of the above, the following embodiments are particularly envisaged:
Embodiment 1 : Use of a labeling compound as a labeling agent for detecting cancerous tissue in a subject, said use comprising administration of a labeling dose of said labeling compound to said subject.
Embodiment 2: The use of embodiment 1, wherein said labeling dose is adjusted to label cancerous tissue, preferably specifically label cancerous tissue.
Embodiment 3: The use of embodiment 1 or 2, wherein said labeling dose is at least 200 pg, preferably at least 500 pg, of labeling compound per subject.
Embodiment 4: The use of any one of embodiments 1 to 3, wherein said labeling dose is at least 1 mg of said labeling compound per subject.
Embodiment 5: The use of any one of embodiments 1 to 4, wherein said labeling dose is of from 0.2 mg to 100 mg, preferably is of from 0.5 to 25 mg, more preferably is of from 1 mg to 10 mg, most preferably is of from 1 mg to 5 mg, of said labeling compound per subject.
Embodiment 6: The use of any one of embodiments 1 to 5, wherein said labeling dose is administered independent of the subject's body mass.
Embodiment 7: The use of any one of embodiments 1 to 6, wherein said labeling dose is exactly one dose of said labeling compound administered within a time frame of at least twelve hours, preferably at least one day, more preferably at least two days.
Embodiment 8: The use of any one of embodiments 1 to 7, wherein said cancerous tissue is tissue comprising prostate cancer cells.
Embodiment 9: The use of any one of embodiments 1 to 8, wherein said cancerous tissue is a primary tumor, a relapse, an infiltrate, a metastasis and/or a lymph node comprising cancer cells.
Embodiment 10: The use of any one of embodiments 1 to 9, wherein said cancerous tissue is an infiltrate, a metastasis and/or a lymph node comprising cancer cells.
Embodiment 11 : The use of any one of embodiments 1 to 10, wherein said labeling further comprises administration of a radioactively labeled derivative of the labeling compound at least two days before administration of said labeling compound, and further comprises increasing the labeling dose of labeling compound in case the signal of said radioactively labeled complex of the labeling compound in tumor or relapse tissue is found to be weak compared to a reference. Embodiment 12: The use of any one of embodiments 1 to 11, wherein said dose of the labeling compound is at least 20fold, preferably at least 30fold, the dose of the dose of the radioactively labeled complex of the labeling compound, preferably in case the signal of said radioactively labeled complex of the labeling compound in tumor or relapse tissue is found to be above or equal to a reference.
Embodiment 13: The use of any one of embodiments 1 to 11, wherein said dose is least
50fold, preferably at least 75fold the dose of the dose of the radioactively labeled complex of the labeling compound, preferably in case the signal of said radioactively labeled complex of the labeling compound in tumor or relapse tissue is found to be below a reference.
Embodiment 14: The use of any one of embodiments 1 to 13, wherein said dose is at most
30000fold, preferably at most lOOOOfold, more preferably at most lOOOfold the dose of the dose of the radioactively labeled complex of the labeling compound.
Embodiment 15: The use of any one of embodiments 1 to 14, wherein said reference is obtained from a multitude of primary and/ or relapse prostate cancer samples.
Embodiment 16: The use of any one of embodiments 1 to 15, wherein said use comprises intraoperative identification of cancerous tissue.
Embodiment 17: A labeling compound as specified in any one of embodiments 1 to 44 for use for in vivo labeling of cancerous tissue in a subject, said labeling comprising administration of a labeling dose of the labeling compound to said subject.
Embodiment 18: A labeling compound as specified in any one of embodiments 1 to 44 for use in a method of diagnosing of cancerous tissue in a subject, wherein said method comprises administration of a labeling dose of the labeling compound to said subject.
Embodiment 19: The labeling compound for use of embodiment 18, wherein said diagnosing is in vivo diagnosing.
Embodiment 20: The labeling compound for use of embodiment 18 or 19, wherein said diagnosing is intraoperative identification of cancerous tissue.
Embodiment 21 : The labeling compound for use of any one of embodiments 18 to 20, wherein said diagnosing is identification of primary tumor, relapse, metastases and/or lymph nodes comprising cancer cells.
Embodiment 22: The labeling compound for use of any one of embodiments 18 to 21, wherein said diagnosing is identification of metastases and/or lymph nodes comprising cancer cells, preferably is intraoperative identification of metastases and/or lymph nodes comprising cancer cells.
Embodiment 23 : A labeling compound as specified in any one of embodiments 1 to 44 for use in a method of removing a cancerous tissue from a subject, said method comprising
(a) labeling cancerous tissue by administering a labeling dose of the labeling compound to said subject;
(b) identifying labeled cancerous tissue in situ; and
(c) removing said cancerous tissue.
Embodiment 24: The labeling compound for use of embodiment 23, wherein said identifying in step (b) comprises irradiating tissue assumed to comprise or be cancerous tissue with light with a wavelength absorbed by said labeling compound, preferably with a wavelength of from 600 nm to 780 nm.
Embodiment 25: The labeling compound for use of embodiment 23 or 24, wherein said identifying in step (b) further comprises detecting fluorescent light emitted by said labeling compound, thereby identifying labeled cancerous tissue.
Embodiment 26: The labeling compound for use of any one of embodiments 23 to 25, wherein said removing in step (c) comprises surgical removal of labeled cancerous tissue, preferably all labeled cancerous tissue identified in step (b).
Embodiment 27: The labeling compound for use of any one of embodiments 23 to 26, wherein no further doses of the labeling compound are administered.
Embodiment 28: The labeling compound for use of any one of embodiments 23 to 27, wherein step (a) is performed within a time frame of from 0.25 h to 48 h, more preferably of from 0.5 h to 24 h, still more preferably of from 0.75 h to 12 h, even more preferably at about 1 h, most preferably 1 h, before identifying labeled cancerous tissue in step (b).
Embodiment 29: The labeling compound for use of any one of embodiments 17 to 28, further comprising at least one further feature of any one of embodiments 1 to 16.
Embodiment 30: A method for labeling cancerous tissue in a subject, comprising administering a labeling dose of labeling compound as specified in any one of embodiments 1 to 44 to said subject.
Embodiment 31 : A method for identifying cancerous tissue in a subject comprising
(a) labeling cancerous tissue by administering a labeling dose of a labeling compound as specified in any one of embodiments 1 to 44 to said subject; and
(b) identifying labeled cancerous tissue in situ.
Embodiment 32: A method for removing cancerous tissue from a subject, said method comprising
(a) labeling cancerous tissue by administering a labeling dose of a labeling compound as specified in any one of embodiments 1 to 44 to said subject;
(b) identifying labeled cancerous tissue in situ; and
(c) removing said cancerous tissue.
Embodiment 33: The method of any one of embodiments 30 to 32, further comprising at least one further feature of any one of embodiments 1 to 29.
Embodiment 34: Use of a labeling compound as specified in any one of embodiments 1 to
44 in the manufacture of a diagnostic composition for labeling cancerous tissue, preferably in vivo labeling cancerous tissue, wherein said diagnostic composition comprises a labeling dose of said labeling compound.
Embodiment 35 : The use of embodiment 34, further comprising at least one further feature of any one of embodiments 1 to 33.
Embodiment 36: A diagnostic composition comprising of from 0.2 mg to 100 mg, preferably is of from 0.5 to 25 mg, more preferably is of from 1 mg to 10 mg, most preferably is of from 1 mg to 5 mg of a labeling compound as specified in any one of embodiments 1 to 44.
Embodiment 37: The subject matter of any of the preceding embodiments, wherein said labeling compound has a structure of formula (I)
(A)-xi-(B) -X2-(C), or is a pharmaceutically acceptable salt or therof wherein
(A) is at least one motif specifically binding to cell membranes of cancer cells;
(B) at least one chelator moiety ;
(C) a dye moiety; xi is a spacer covalently connecting (A) and (B);
X2 is a spacer or a chemical single bond connecting (B) and (C).
and wherein xi comprises a group -AA-, wherein AA is an amino acid sequence of 4 to 8 (preferably 5 to 7, more preferably 6) naturally occurring amino acids and wherein at least two amino acids are histidine.
Embodiment 38: The subject matter of the preceding embodiment 37, wherein said dye moiety has the formula
wherein
X1 and X4 are independently selected from the group consisting of -N=, -N(R5)=, and -C(R6)=;
X2 and X3 are independently selected from the group consisting of O, S, Se, N(R5), and C(R6R7), preferably are both C(CH3)2;
Y is a linker connecting the two moieties of (C) and permitting electron delocalization between said moieties, wherein Y optionally comprises a group (L-)cZ0; a and b are independently selected from the group consisting of 1, 2, and 3; each R1 and each R2 is independently (L-)CZ, (L-)CZ° or H; and two adjacent R1 and/or two adjacent R2 can also form an aromatic ring, which is optionally substituted with one or more (L-)cZ or (L-)cZ°;
R3, R4, R5, R6, R7, R9 are independently selected from the group consisting of (L-)cZ, (L-)CZ°, and H; each c is independently 0, or 1 ; each L is independently
NH OjT1, or a Ci -io alkylene group, which is optionally interrupted and/or terminated by one or more of -O-, -S-, -C(O)-, -C(O)O-, -OC(O)-, -C(O)NH-, -NHC(O)O-, and T1;
T1 is phenyl, naphthyl, indenyl, indanyl, tetralinyl, decalinyl, adamantyl, C3-7 cycloalkyl, 3 to 7 membered heterocyclyl, or 7 to 11 membered heterobicyclyl, wherein T1 is optionally substituted with one or more substituents selected from the group consisting of halogen, CN, C(O)R8, COOR8, OR8, C(O)N(R8R8a), S(O)2N(R8R8a), S(O)N(R8R8a), S(O)2R8, N(R8)S(O)2N(R8aR8b), SR8, N(R8R8a), NO2; OC(O)R8, N(R8)C(O)R8a, N(R8)S(O)2R8a,
N(R8)S(O)R8a, N(R8)C(O)N(R8aR8b), N(R8)C(O)OR8a, OC(O)N(R8R8a), oxo (=0), where the ring is at least partially saturated, or Ci-6 alkyl, wherein Ci-6 alkyl is optionally substituted with one or more halogen, which are the same or different; each Z is independently H, halogen, CN, C(O)R8, C(O)OR8, C(O)O’ OR8, C(O)N(R8R8a), S(O)2OR8, S(O)2O’, S(O)2N(R8R8a), S(O)N(R8R8a), S(O)2R8, S(O)R8, N(R8)S(O)2N(R8aR8b), SR8, N(R8R8a), NO2; P(O)(OR8)2, P(O)(OR8)O’, OC(O)R8, N(R8)C(O)R8a, N(R8)S(O)2R8a, N(R8)S(O)R8a, N(R8)C(O)N(R8aR8b), N(R8)C(O)OR8a, or OC(O)N(R8R8a);
R8, R8a, R8b are independently selected from the group consisting of H, or Ci-6 alkyl, wherein Ci-6 alkyl is optionally substituted with one or more halogen, which are the same or different; Z° is a chemical bond connecting (C) to x2 or to (B) in case x2 is a chemical single bond; provided that one of R1, R2, R3, R4, R5, R6, R7, R9 is (L-)CZ° or that Y comprises (L-)CZ°.
Embodiment 39: The subject matter of the preceding embodiment 37, wherein said dye moiety (C) is a fluorescent dye moiety selected from the group consisting of the following structures:
wherein X' is a pharmaceutically acceptable negatively charged counterion; wherein Y+ is a pharmaceutically acceptable positively charged counterion; and wherein the wavy line indicates the conjugation site to the rest of the compound of formula (I).
Embodiment 40: The subject matter of any one of the preceding embodiment 37 to 39, wherein the chelator moiety (B) is selected from the following group:
wherein in each of the structures one of the wavy lines indicates the conjugation site to the at least one motif specifically binding to cell membranes of cancer cells (A) via spacer xi as defined herein, and the other wavy line indicates the conjugation site to the dye moiety (C), preferably via the spacer X2
Emodiment 41: The subject matter of the preceding embodiment 40, wherein the chelator moiety (B) is
Embodiment 42: The subject matter of any one of the preceding embodiments 37 to 41, wherein (A) is a PSMA binding motif having the following structure:
Embodiment 43 : The subject matter of any one of the preceding embodiment 37 to 42, wherein
(A)-xi is represented by the following structure:
Embodiment 44: The subject matter of any one of the preceding embodiments, wherein the compound of formula (I) has the following chemical structure:
All references cited in this specification are herewith incorporated by reference with respect to their entire disclosure content and the disclosure content specifically mentioned in this specification.
Figure Legends
Fig. 1 : Preoperative 68Ga-PSMA-914 PET/CT imaging: (A) MIP of a 76-year old patient with high-risk prostate carcinoma (Gleason score 9, initial PSA level 99.2 ng/ml) at 1 h p.i. of 195 MBq 68Ga-PSMA-914 with corresponding axial slices of the extensive primary tumor (B) and mesorectal lymph node metastases (C) using predefined PET window settings (inverted grey scale, SUV range 0 to 5 g/ml)
Fig. 2: Intraoperative fluorescence guidance: PSMA-914 was administered 1 h prior to surgery and resection of primary tumor (A) and lymph node metastases (B) was performed under fluorescence guidance in situ. The fluorescence signal was additionally confirmed ex situ with an exemplary lymph node shown in (C); fluorescence detection was performed with an IMAGE S system (Excitation wavelength 805 nm).
Fig. 3: Postoperative fluorescence detection and histopathology: Fluorescence detection of the primary tumor (A) and lymph node metastases (C) was performed ex situ (excitation
wavelength 800 nm, 10 gm sections) and correlated with corresponding PSMA stainings (B and D).
The following Examples shall merely illustrate the invention. They shall not be construed, whatsoever, to limit the scope of the invention.
Example 1 : Materials and Methods
After giving written informed consent, 4 patients with high-risk prostate cancer (Gleason Score >8) underwent preoperative PET/CT imaging 1 h and 2 h after intravenous injection of approximately 200 MBq 68Ga-PSMA-914 (68Ga-Glu-urea-Lys-(HE)3-HBED-CC- IRDye800CW) on a compassionate-use basis. One to six days after PET/CT, fluorescence- guided surgery was performed with PSMA-914 administration 1 h prior to surgery. The dose was 1 mg unlabeled PSMA-914 for all patients. Verification of intraoperative findings was done by postoperative ex situ fluorescence analysis and histopathology.
Example 2: Results
Preoperative 68Ga-PSMA-914 PET/CT imaging revealed high tracer uptake in the primary tumors at 1 h (mean SUVmax 21.6 ± 19.5 g/ml; range 6.1 - 49.0 g/ml), slightly increasing at 2 h (mean SUVmax 26.9 ± 25.8 g/ml; range 6.6 - 63.3 g/ml). Lymph node metastases were additionally found on PET imaging in three patients. Intraoperatively, the fluorescence signal of PSMA-914 specifically visualized primary tumor tissue and lymph node metastases with a high contrast to surrounding healthy structures and resections were performed under fluorescence-guidance. The PSMA-specific enrichment of the hybrid molecule in malignant tissue was additionally confirmed by ex situ fluorescence detection and histopathology. Conclusion: This first clinical translation demonstrates a successful application highlighting the potential of PSMA-targeting hybrid molecules. The novel P SM A- 11 -derived hybrid molecule PSMA-914 supports the precise pre- and intraoperative detection of PSMA-expressing tumor manifestations.
References
Mease R.C. et al. Clin Cancer Res. 2008, 14, 3036-3043;
Foss, C. A.; et al. Clin Cancer Res 2005, 11 , 4022-4028;
Nan, F.; et al. J Med Chem 2000, 43, 772-774
Pomper, M.G.; et al. Mol Imaging 2002, 1 , 96-101;
Schulke, N.; et al. Proc Natl Acad Sci U S A 2003, 100, 12590-12595
Tasch, J.; et al. Crit Rev Immunol 2001 , 21 , 249-261
US 2015/0110715 Al
WO 2013/022797
WO 2015/055318
WO20 19/092242
Zhou, J.; et al. Nat Rev Drug Discov 2005, 4, 015-1026;
Claims
1. Use of a labeling compound having a chemical structure of formula (I):
(A)-XI-(B)-X2-(C), wherein (A) is at least one motif specifically binding to cell membranes of cancer cells;
(B) is at least one chelator moiety;
(C) is a dye moiety; xi is a spacer covalently connecting (A) and (B);
X2 is a spacer or a chemical single bond connecting (B) and (C), or a pharmaceutically acceptable salt thereof, as a labeling agent for detecting cancerous tissue in a subject, said use comprising administration of a labeling dose of said labeling compound to said subject.
2. The use of any claim 1, wherein said labeling dose is of from 0.2 mg to 100 mg, preferably is of from 0.5 to 25 mg, more preferably is of from 1 mg to 10 mg, most preferably is of from 1 mg to 5 mg, of said labeling compound per subject.
3. The use of claim 1 or 2, wherein said labeling dose is at least 1 mg of said labeling compound per subject.
4. The use of any one of claims 1 to 3, wherein said labeling dose is administered independent of the subject's body mass.
5. The use of any one of claims 1 to 4, wherein said labeling dose is exactly one dose of said labeling compound administered within a time frame of at least twelve hours, preferably at least one day, more preferably at least two days.
6. The use of any one of claims 1 to 5, wherein said cancerous tissue is tissue comprising prostate cancer cells.
7. The use of any one of claims 1 to 6, wherein said cancerous tissue is a primary tumor, a relapse, an infiltrate, a metastasis and/or a lymph node comprising cancer cells, preferably is an infiltrate, a metastasis and/or a lymph node comprising cancer cells.
The use of any one of claims 1 to 7, wherein said labeling further comprises administration of a radioactively labeled complex of the labeling compound at least two days before administration of said labeling compound, and further comprises increasing the labeling dose of labeling compound in case the signal of said radioactively labeled complex of the labeling compound in tumor or relapse tissue is found to be weak compared to a reference. The use of any one of claims 1 to 8, wherein said use comprises intraoperative identification of cancerous tissue. A labeling compound as specified in claim 1 for use for in vivo labeling of cancerous tissue in a subject, said labeling comprising administration of a labeling dose of the labeling compound to said subject. A labeling compound as specified in claim 1 for use in a method of removing a cancerous tissue from a subject, said method comprising
(a) labeling cancerous tissue by administering a labeling dose of the labeling compound to said subject;
(b) identifying labeled cancerous tissue in situ; and
(c) removing said cancerous tissue. A method for identifying cancerous tissue in a subject comprising
(a) labeling cancerous tissue by administering a labeling dose of a labeling compound as specified in claim 1 to said subject; and
(b) identifying labeled cancerous tissue in situ. A diagnostic composition comprising of from 0.2 mg to 100 mg, preferably is of from 0.5 to 25 mg, more preferably is of from 1 mg to 10 mg, most preferably is of from 1 mg to 5 mg of a labeling compound as specified in claim 1. The subject matter of any of the preceding claims, wherein the dye moiety (C) is a fluorescent dye moiety selected from the group consisting of the following structures:
wherein X' is a pharmaceutically acceptable negatively charged counterion; wherein Y+ is a pharmaceutically acceptable positively charged counterion; and wherein the wavy line indicates the conjugation site to the rest of the compound of formula (I). The subject matter of any of the preceding claims, wherein the compound of formula (I) has the following chemical structure:
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP22170044 | 2022-04-26 | ||
| EP22170044.6 | 2022-04-26 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2023208928A1 true WO2023208928A1 (en) | 2023-11-02 |
Family
ID=81387165
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2023/060799 Ceased WO2023208928A1 (en) | 2022-04-26 | 2023-04-25 | Dosage of psma-ligands for fluorescence based detection of cancerous tissue |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2023208928A1 (en) |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013022797A1 (en) | 2011-08-05 | 2013-02-14 | Molecular Insight Pharmaceuticals | Radiolabeled prostate specific membrane antigen inhibitors |
| WO2015055318A1 (en) | 2013-10-18 | 2015-04-23 | Deutsches Krebsforschungszentrum | Labeled inhibitors of prostate specific membrane antigen (psma), their use as imaging agents and pharmaceutical agents for the treatment of prostate cancer |
| US20150110715A1 (en) | 2013-10-17 | 2015-04-23 | Ruprecht-Karls-Universitat Heidelberg | Double-Labeled Probe for Molecular Imaging and Use Thereof |
| WO2019092242A1 (en) | 2017-11-13 | 2019-05-16 | Deutsches Krebsforschungszentrum | A double-labeled probe for molecular imaging and use thereof |
-
2023
- 2023-04-25 WO PCT/EP2023/060799 patent/WO2023208928A1/en not_active Ceased
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013022797A1 (en) | 2011-08-05 | 2013-02-14 | Molecular Insight Pharmaceuticals | Radiolabeled prostate specific membrane antigen inhibitors |
| US20150110715A1 (en) | 2013-10-17 | 2015-04-23 | Ruprecht-Karls-Universitat Heidelberg | Double-Labeled Probe for Molecular Imaging and Use Thereof |
| WO2015055318A1 (en) | 2013-10-18 | 2015-04-23 | Deutsches Krebsforschungszentrum | Labeled inhibitors of prostate specific membrane antigen (psma), their use as imaging agents and pharmaceutical agents for the treatment of prostate cancer |
| WO2019092242A1 (en) | 2017-11-13 | 2019-05-16 | Deutsches Krebsforschungszentrum | A double-labeled probe for molecular imaging and use thereof |
Non-Patent Citations (11)
| Title |
|---|
| "Remington's Pharmaceutical Sciences", MACK PUBLISHING COMPANY |
| DOWDYWEARDEN: "Statistics for Research", 1983, JOHN WILEY & SONS |
| EDER ANN-CHRISTIN ET AL: "The PSMA-11-derived hybrid molecule PSMA-914 specifically identifies prostate cancer by preoperative PET/CT and intraoperative fluorescence imaging", EUROPEAN JOURNAL OF NUCLEAR MEDICINE AND MOLECULAR IMAGING, SPRINGER BERLIN HEIDELBERG, BERLIN/HEIDELBERG, vol. 48, no. 6, 23 January 2021 (2021-01-23), pages 2057 - 2058, XP037450345, ISSN: 1619-7070, [retrieved on 20210123], DOI: 10.1007/S00259-020-05184-0 * |
| FOSS, C.A. ET AL., CLIN CANCER RES, vol. 11, 2005, pages 4022 - 4028 |
| MEASE R.C. ET AL., CLIN CANCER RES., vol. 14, 2008, pages 3036 - 3043 |
| NAN, F. ET AL., J MED CHEM, vol. 43, 2000, pages 772 - 774 |
| POMPER, M.G. ET AL., MOL IMAGING, vol. 1, 2002, pages 96 - 101 |
| SCHULKE, N. ET AL., PROC NATL ACAD SCI U S A, vol. 100, 2003, pages 12590 - 12595 |
| SCHULKE, N. ET AL., PROC. NATL. ACAD. SCI. USA, vol. 100, 2003, pages 12590 - 12595 |
| TASCH, J. ET AL., CRIT REV IMMUNOL, vol. 21, 2001, pages 249 - 261 |
| ZHOU, J. ET AL., NAT REV DRUG DISCOV, vol. 4, 2005, pages 015 - 1026 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11020493B2 (en) | Double-labeled probe for molecular imaging and use thereof | |
| JP2023111939A (en) | Near-infrared fluorescence contrast bioimaging agent and method of use thereof | |
| AU2018373675B2 (en) | A double-labeled probe for molecular imaging and use thereof | |
| AU2018363807B2 (en) | A double-labeled probe for molecular imaging and use thereof | |
| US20200237936A1 (en) | Fibroblast activation protein (fap)-targeted imaging and therapy | |
| EP3554526B1 (en) | Luteinizing hormone-releasing hormone receptor (lhrh-r) conjugates and uses thereof | |
| CA3035542A1 (en) | Psma-targeted nir dyes and their uses | |
| EA037512B1 (en) | IMPROVED PROSTATIC SPECIFIC MEMBRANE ANTIGEN (PSMA) INHIBITORS LABELED WITH 18F AND THEIR USE AS VISUALIZING AGENTS IN PROSTATE CANCER | |
| EP1343758A1 (en) | Substituted bis-indole derivatives useful as contrast agents, pharmaceutical compositions containing them and intermediates for producing them | |
| EP3976575A1 (en) | Ph responsive compositions and uses thereof | |
| JP2019512511A (en) | Drugs and methods for targeting carbonic anhydrase IX | |
| WO2023208928A1 (en) | Dosage of psma-ligands for fluorescence based detection of cancerous tissue | |
| WO2023222679A1 (en) | Prostate specific membrane antigen (psma) ligands | |
| US20250360236A1 (en) | Prostate specific membrane antigen (psma) ligands | |
| TWI704922B (en) | Bladder cancer photodynamic therapeutic agents with off-on magnetic resonance imaging enhancement | |
| CA3016191A1 (en) | Ca ix-target nir dyes and their uses |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 23722507 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 23722507 Country of ref document: EP Kind code of ref document: A1 |