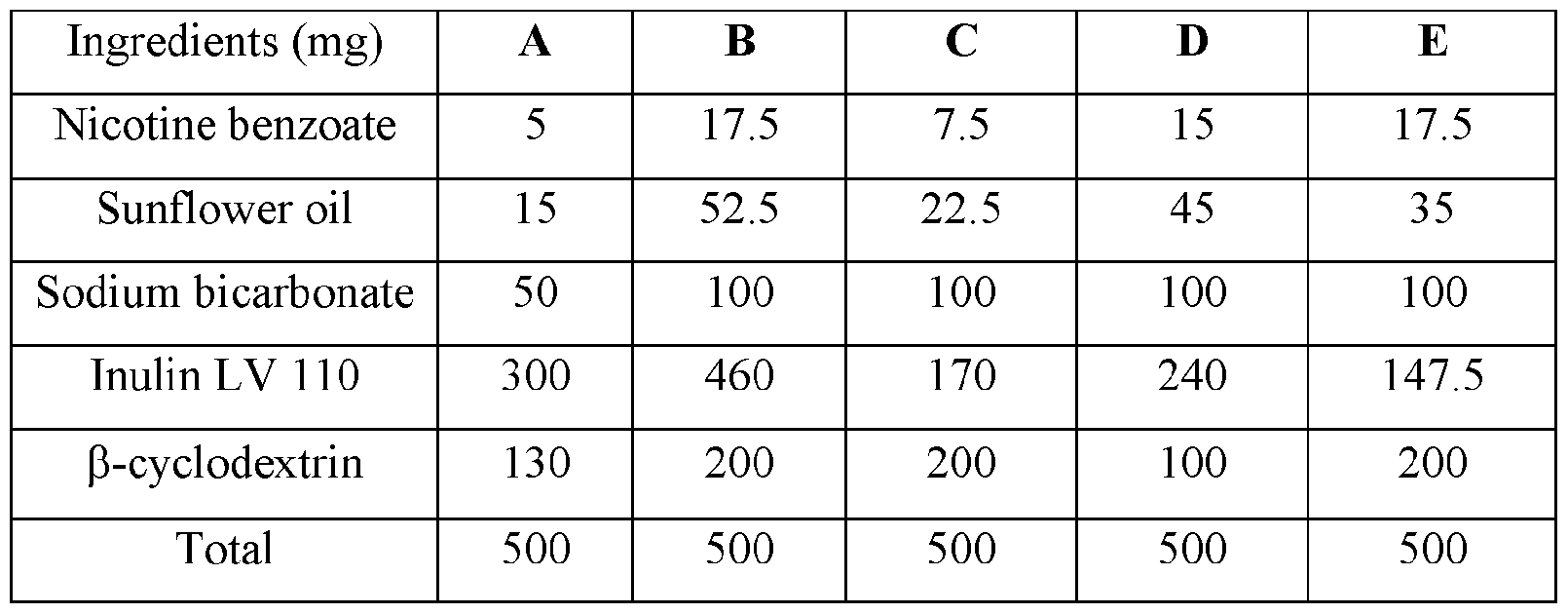
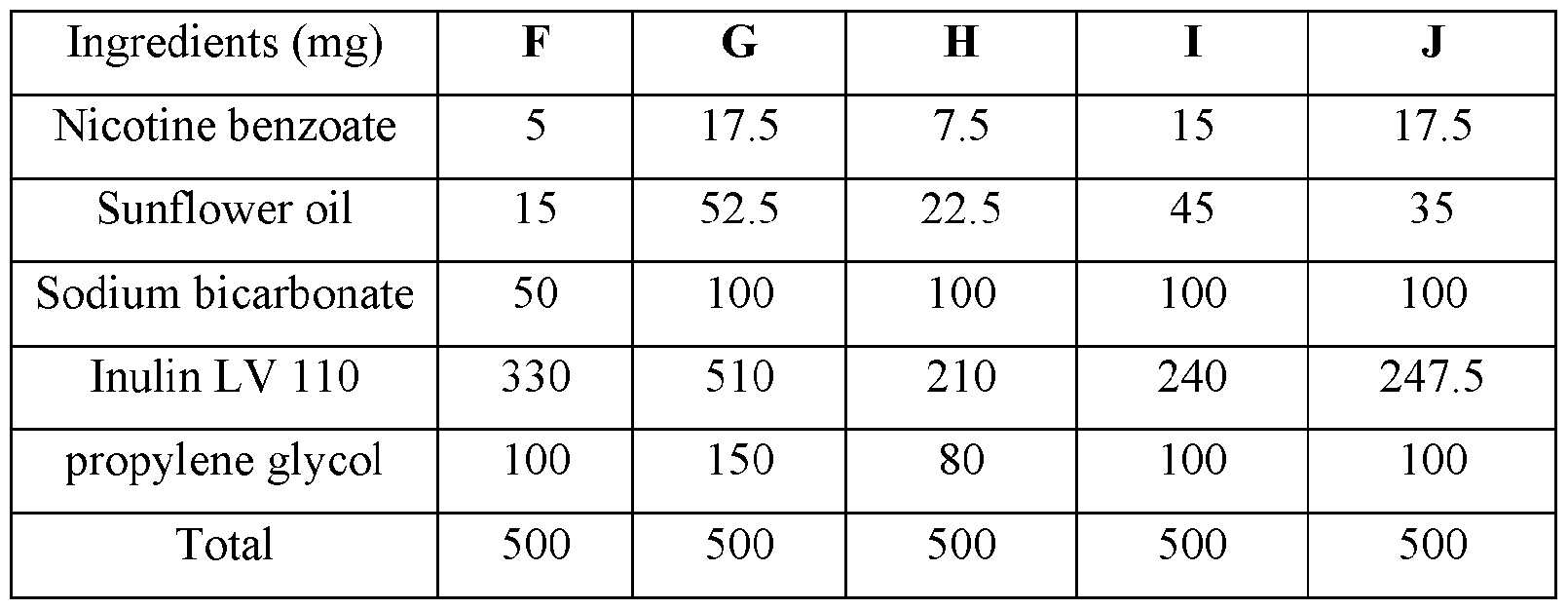
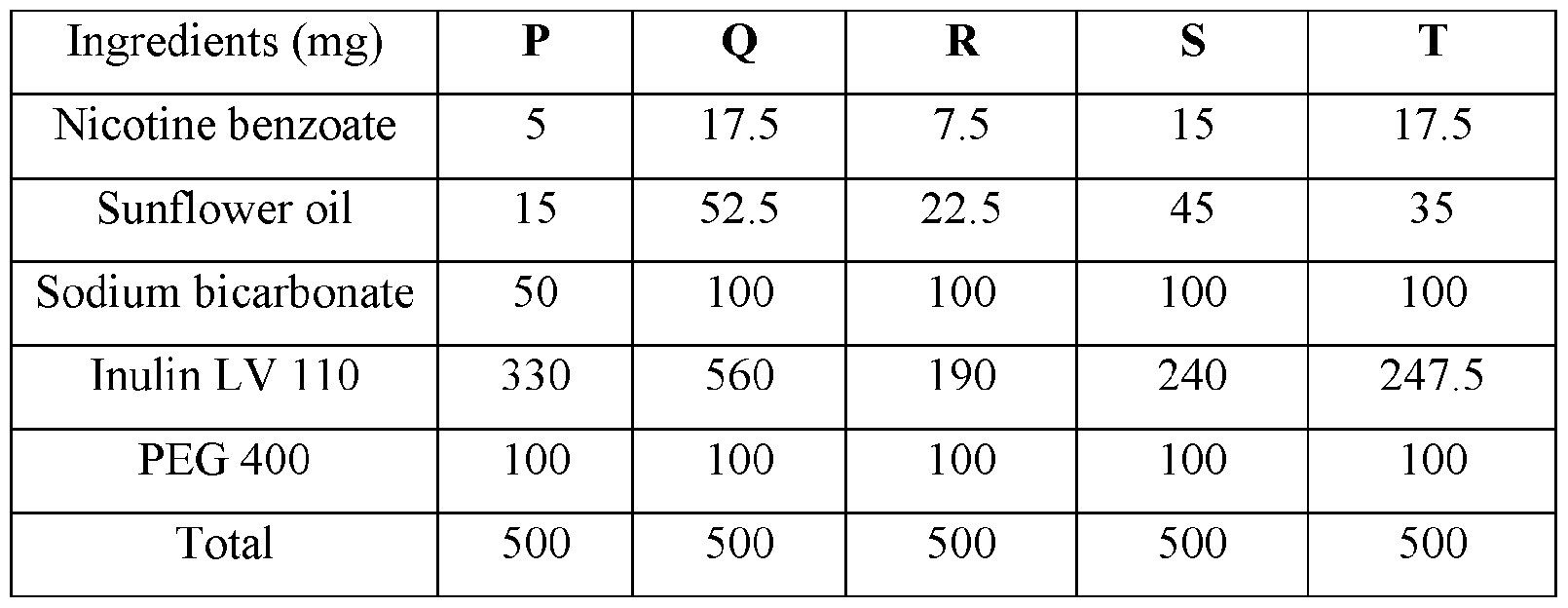
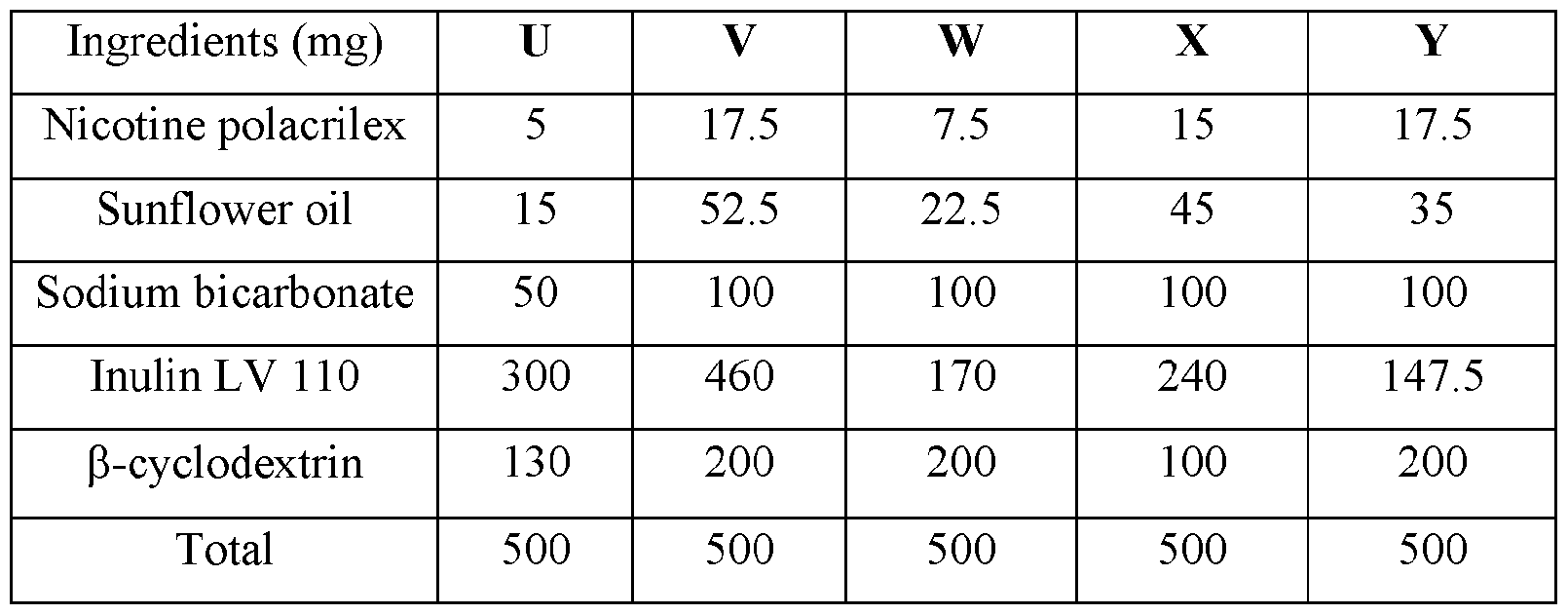
WO2023183228A1 - Compositions and methods for sublingual delivery of nicotine - Google Patents
Compositions and methods for sublingual delivery of nicotine Download PDFInfo
- Publication number
- WO2023183228A1 WO2023183228A1 PCT/US2023/015627 US2023015627W WO2023183228A1 WO 2023183228 A1 WO2023183228 A1 WO 2023183228A1 US 2023015627 W US2023015627 W US 2023015627W WO 2023183228 A1 WO2023183228 A1 WO 2023183228A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- nicotine
- weight
- fibers
- composition
- composition according
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A24—TOBACCO; CIGARS; CIGARETTES; SIMULATED SMOKING DEVICES; SMOKERS' REQUISITES
- A24B—MANUFACTURE OR PREPARATION OF TOBACCO FOR SMOKING OR CHEWING; TOBACCO; SNUFF
- A24B13/00—Tobacco for pipes, for cigars, e.g. cigar inserts, or for cigarettes; Chewing tobacco; Snuff
-
- A—HUMAN NECESSITIES
- A24—TOBACCO; CIGARS; CIGARETTES; SIMULATED SMOKING DEVICES; SMOKERS' REQUISITES
- A24B—MANUFACTURE OR PREPARATION OF TOBACCO FOR SMOKING OR CHEWING; TOBACCO; SNUFF
- A24B15/00—Chemical features or treatment of tobacco; Tobacco substitutes, e.g. in liquid form
- A24B15/10—Chemical features of tobacco products or tobacco substitutes
- A24B15/16—Chemical features of tobacco products or tobacco substitutes of tobacco substitutes
-
- A—HUMAN NECESSITIES
- A24—TOBACCO; CIGARS; CIGARETTES; SIMULATED SMOKING DEVICES; SMOKERS' REQUISITES
- A24B—MANUFACTURE OR PREPARATION OF TOBACCO FOR SMOKING OR CHEWING; TOBACCO; SNUFF
- A24B15/00—Chemical features or treatment of tobacco; Tobacco substitutes, e.g. in liquid form
- A24B15/18—Treatment of tobacco products or tobacco substitutes
- A24B15/28—Treatment of tobacco products or tobacco substitutes by chemical substances
- A24B15/30—Treatment of tobacco products or tobacco substitutes by chemical substances by organic substances
- A24B15/302—Treatment of tobacco products or tobacco substitutes by chemical substances by organic substances by natural substances obtained from animals or plants
- A24B15/303—Plant extracts other than tobacco
-
- A—HUMAN NECESSITIES
- A24—TOBACCO; CIGARS; CIGARETTES; SIMULATED SMOKING DEVICES; SMOKERS' REQUISITES
- A24B—MANUFACTURE OR PREPARATION OF TOBACCO FOR SMOKING OR CHEWING; TOBACCO; SNUFF
- A24B15/00—Chemical features or treatment of tobacco; Tobacco substitutes, e.g. in liquid form
- A24B15/18—Treatment of tobacco products or tobacco substitutes
- A24B15/28—Treatment of tobacco products or tobacco substitutes by chemical substances
- A24B15/30—Treatment of tobacco products or tobacco substitutes by chemical substances by organic substances
- A24B15/36—Treatment of tobacco products or tobacco substitutes by chemical substances by organic substances containing a heterocyclic ring
- A24B15/40—Treatment of tobacco products or tobacco substitutes by chemical substances by organic substances containing a heterocyclic ring having only oxygen or sulfur as hetero atoms
- A24B15/403—Treatment of tobacco products or tobacco substitutes by chemical substances by organic substances containing a heterocyclic ring having only oxygen or sulfur as hetero atoms having only oxygen as hetero atoms
-
- A—HUMAN NECESSITIES
- A24—TOBACCO; CIGARS; CIGARETTES; SIMULATED SMOKING DEVICES; SMOKERS' REQUISITES
- A24B—MANUFACTURE OR PREPARATION OF TOBACCO FOR SMOKING OR CHEWING; TOBACCO; SNUFF
- A24B15/00—Chemical features or treatment of tobacco; Tobacco substitutes, e.g. in liquid form
- A24B15/18—Treatment of tobacco products or tobacco substitutes
- A24B15/28—Treatment of tobacco products or tobacco substitutes by chemical substances
- A24B15/42—Treatment of tobacco products or tobacco substitutes by chemical substances by organic and inorganic substances
Definitions
- compositions and methods for oral delivery of nicotine and nicotine derivatives are disclosed herein.
- the nicotine is delivered in an oral packets, pouches, or sachets.
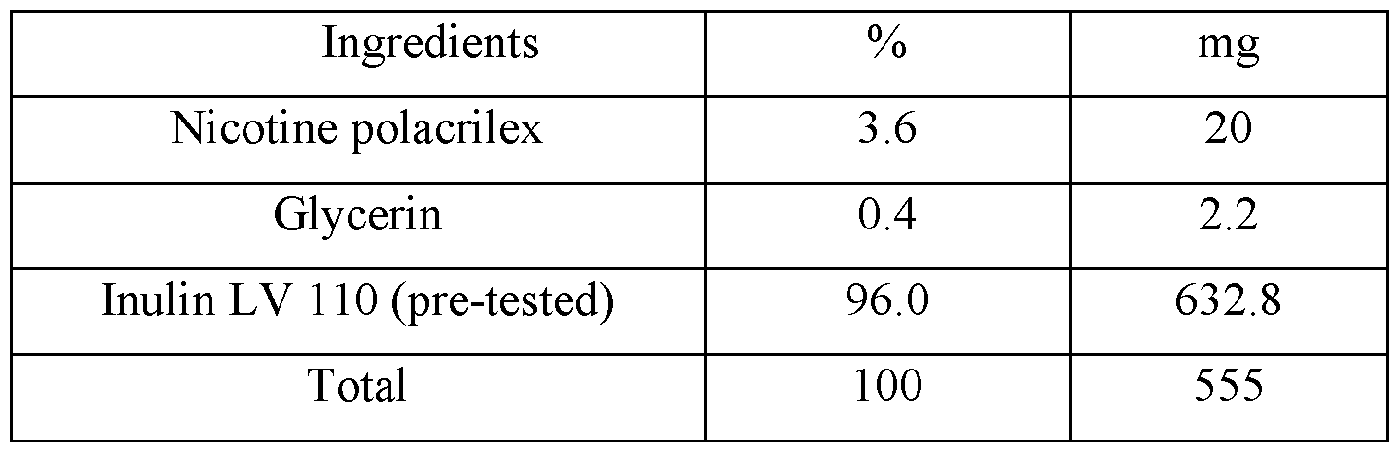
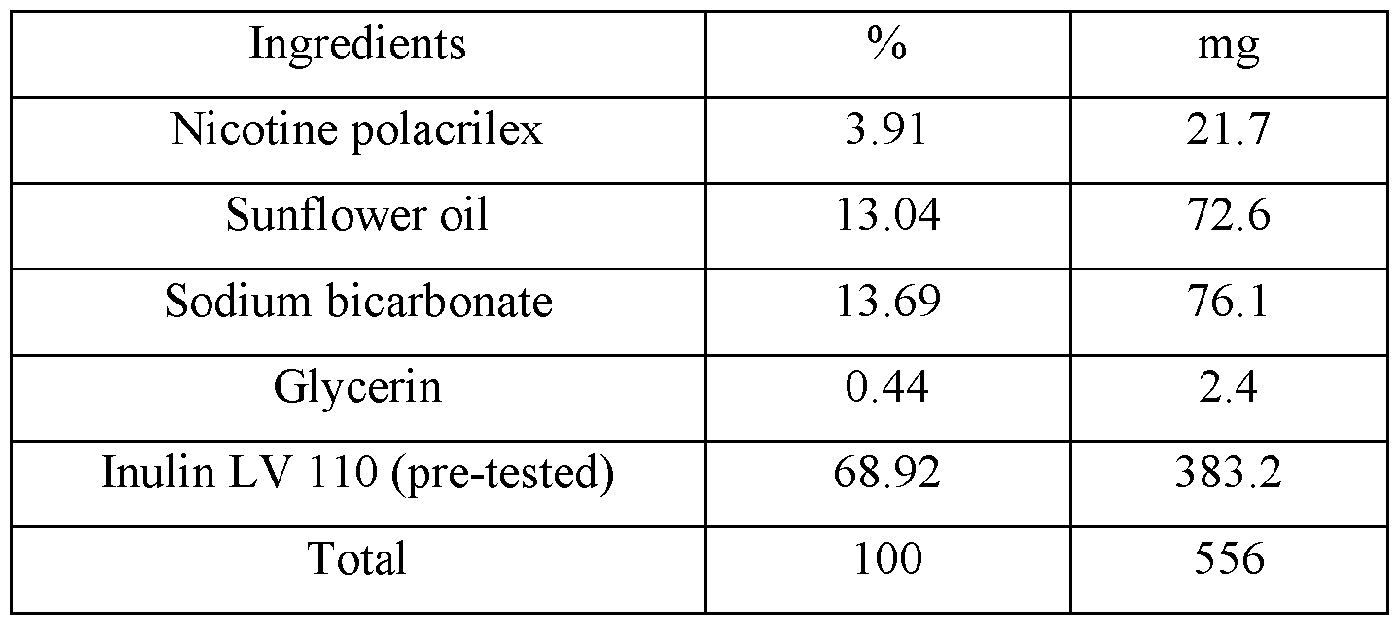
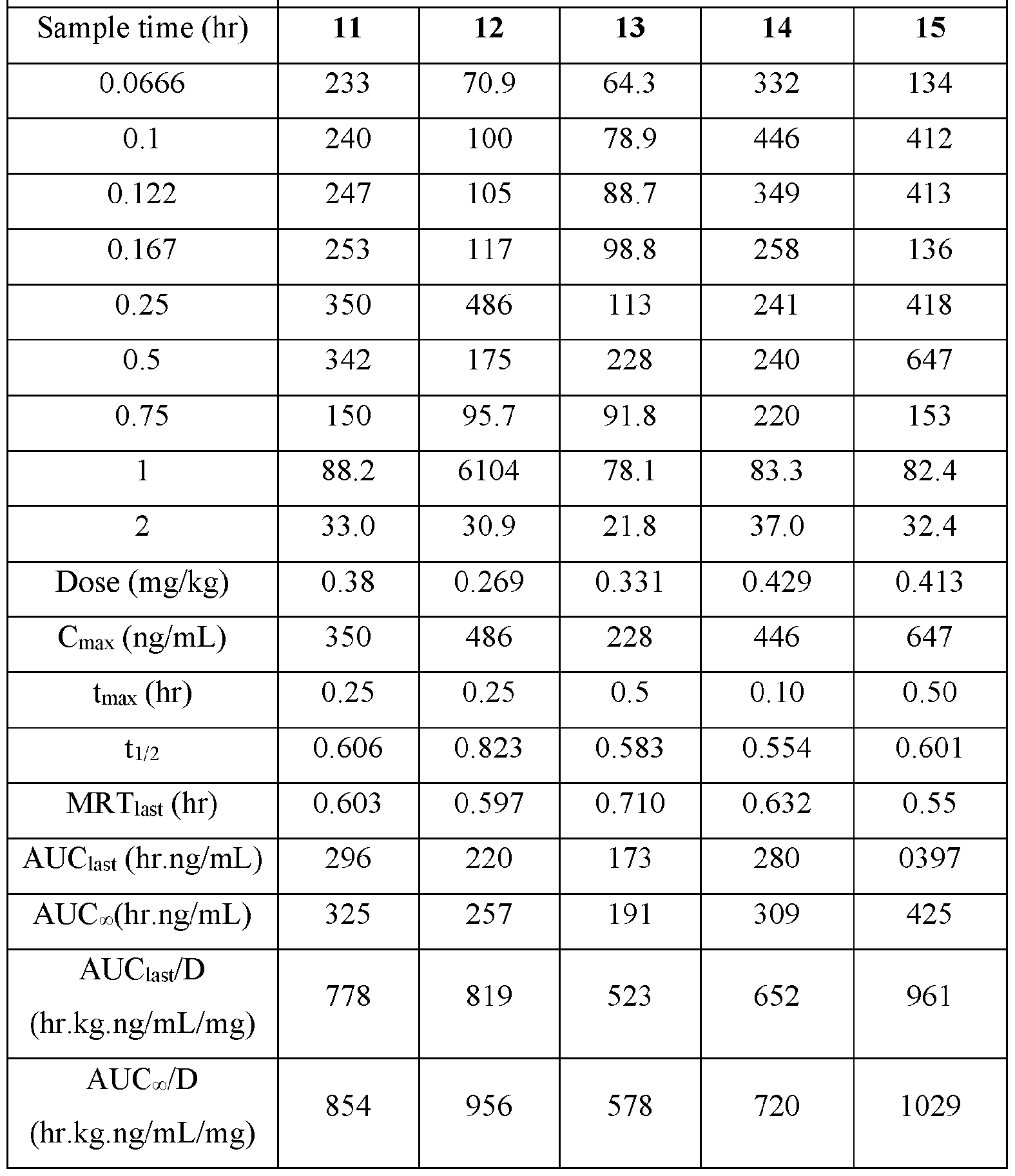
- Figure l is a plot of the individual plasma concentrations (ng/mL) for Nicotine versus time (hour) after buccal administration of the nicotine benzoate control composition disclosed in TABLE I in male Beagle dogs.
- Figure 2 is a plot of the mean plasma concentration (ng/mL) for Nicotine versus time (hour) after buccal administration of nicotine benzoate control composition disclosed in TABLE I in male Beagle dogs.
- Figure 3 is a plot of the individual plasma concentrations (ng/mL) for Nicotine versus time (hour) after buccal administration of the disclosed nicotine benzoate composition disclosed in Table II (4 mg) in male Beagle dogs.
- Figure 4 is a plot of the mean plasma concentration (ng/mL) for Nicotine versus time (hour) after buccal administration of the disclosed compound in Table II (4 mg) in male Beagle dogs.
- Figure 5 is a plot of the individual plasma concentrations (ng/mL) for Nicotine versus time (hour) after buccal administration of the nicotine polacrilex control composition disclosed in TABLE III (4 mg) in male Beagle dogs (Group 3).
- Figure 6 is a plot of the mean plasma concentration (ng/mL) for Nicotine versus time (hour) after buccal administration of the nicotine polacrilex control composition disclosed in TABLE III (4 mg) in Male Beagle dogs (Group 3)
- Figure 7 is a plot of the individual plasma poncentrations (ng/mL) for Nicotine versus time (hour) after buccal administration of the nicotine polarcilex composition disclosed in TABLE VI (4 mg) in male Beagle dogs (Group 4).
- Figure 8 is a plot of the mean plasma concentration (ng/mL) for Nicotine versus time (hour) after buccal administration of the nicotine polarcrilex composition disclosed in TABLE VI (4 mg) in male Beagle dogs (Group 4).
- Ranges may be expressed herein as from “about” one particular value, and/or to “about” another particular value. When such a range is expressed, another aspect includes from the one particular value and/or to the other particular value. Similarly, when values are expressed as approximations, by use of the antecedent “about,” it will be understood that the particular value forms another aspect. It will be further understood that the endpoints of each of the ranges are significant both in relation to the other endpoint, and independently of the other endpoint.
- any embodiment of any of the disclosed methods or compositions can consist of or consist essentially of - rather than comprise/include/contain/have - any of the described steps, elements, and/or features.
- the term “consisting of’ or “consisting essentially of’ can be substituted for any of the open-ended linking verbs recited above, in order to change the scope of a given claim from what it would otherwise be using the open- ended linking verb.
- any embodiment of any of the disclosed compounds or methods can consist of or consist essentially of - rather than comprise/include/contain/have - any of the described steps, elements, and/or features.
- the term “consisting of’ or “consisting essentially of’ can be substituted for any of the open-ended linking verbs recited above, in order to change the scope of a given claim from what it would otherwise be using the open- ended linking verb.
- the terms “sublingual” and “buccal” are used interchangeably.
- the definition of “sublingual” is administration of a drug under the tongue to be absorbed by the tissue therein.
- the definition of “buccal” is to administer a drug by placing it between your cheek and gum.
- the user can either place the disclosed compositions under the tongue of between the check and gum, whichever mode of delivery is more convenient. Therefore, the disclose compositions can be absorbed in any manner chosen by the user.
- a smokeless oral nicotine product can be provided to the user in a portioned or a nonportioned format. Portioned smokeless oral nicotine products can reduce or eliminate the handling of the tobacco by the user, which can offer significant advantages in terms of better hygiene, convenience and/or ease of use.
- compositions for sublingual delivery of nicotine are absorbed in the mucosa of the mouth and therefore avoid the side effect of direct contact of nicotine with the stomach, intestines and other digestive organs.
- base compositions for sublingual or buccal delivery of nicotine comprising: a) from about 1% to about 6% by weight of nicotine, a nicotine salt, nicotine in combination with a resin, or mixtures thereof; b) from about 3% to about 20% by weight of sunflower oil; c) from about 10% to about 20% by weight of sodium bicarbonate; and d) the balance one or more carriers.
- compositions do not comprise an organic or inorganic acid.
- compositions are free flowing solids containing less than 0.01% by weight moisture.
- compositions comprises: a) from about 1% to about 3.5% by weight of nicotine, a nicotine salt, or mixtures thereof; b) from about 3% to about 10.5% by weight of sunflower oil; c) from about 10% to about 20% by weight of sodium bicarbonate; and d) the balance one or more carriers.
- the base compositions comprise: a) from about 1% to about 3.5% by weight of nicotine, a nicotine salt, or mixtures thereof; b) from about 3% to about 10.5% by weight of sunflower oil; c) from about 10% to about 20% by weight of sodium bicarbonate; and d) the balance an admixture of microcrystalline cellulose and inulin.
- the disclosed base compositions can comprise from about 1% to about 6% by weight of nicotine, a nicotine salt or nicotine in combination with a resin.
- the base composition can comprise from about 1% to about 5% by weight of nicotine, a nicotine salt or nicotine in combination with a resin.
- the base composition can comprise from about 2% to about 6% by weight of nicotine, a nicotine salt or nicotine in combination with a resin.
- the base composition can comprise from about 2% to about 5% by weight of nicotine, a nicotine salt or nicotine in combination with a resin.
- the base composition can comprise from about 3% to about 5% by weight of nicotine, a nicotine salt or nicotine in combination with a resin.
- the amount of nicotine, a nicotine salt or nicotine in combination with a resin can be 1%, 2%, 3%, 4%, 5%, or 6% by weight or any fractional amounts, for example, 1.5%, 3,25%, and 5.75%.
- the disclosed base compositions can comprise from about 3% to about 20% by weight of sunflower oil. In one embodiment the base compositions can comprise from about 3% to about 15% by weight of sunflower oil. In another embodiment the base compositions can comprise from about 5% to about 17% by weight of sunflower oil. In a further embodiment the base compositions can comprise from about 7.5% to about 15% by weight of sunflower oil. In a still further embodiment the base compositions can comprise from about 5% to about 10% by weight of sunflower oil.
- the amount of sunflower oil can be 3%, 4%, 5%, 6%, 7%, 8%, 9%, 10%, 11%, 12%, 13%, 14%, 15%, 16%, 17%, 18%, 19%, or 20% by weight of sunflower oil or any fractional amounts, for example, 10.5%, 13.6%, and 17.5%.
- the ratio of nicotine, a nicotine salt or nicotine in combination with a resin to sunflower oil is from about 1 : 1 to about 1 :4.
- the ratio of nicotine, a nicotine salt or nicotine in combination with a resin to sunflower oil can be 1 :1, 1: 1.1, 1 :1.2, 1: 1.3, 1 :1.4, 1: 1.5, 1 :1.6, 1: 1.7, 1 :1.8, 1: 1.9, 1 :2, 1 :2.1, 1 :2.2, 1 :2.3, 1 :2.4, 1 :2.5, 1 :2.6, 1 :2.7, 1:2.8, 1 :2.9, or 1 :3.
- the disclosed base compositions can comprise from about 10% to about 20% by weight of sodium bicarbonate. In one embodiment the base compositions can comprise from about 10% to about 15% by weight of sodium bicarbonate. In another embodiment the base compositions can comprise from about 15% to about 20% by weight of sodium bicarbonate. In a further embodiment the base compositions can comprise from about 12.5% to about 17.5% by weight of sodium bicarbonate. In a still further embodiment the base compositions can comprise from about 14% to about 17% by weight of sodium bicarbonate.
- the amount of sodium bicarbonate can be 10%, 11%, 12%, 13%, 14%, 15%, 16%, 17%, 18%, 19%, or 20% by weight of sodium bicarbonate or any fractional amounts, for example, 10.5%, 13.6%, and 17.5%.
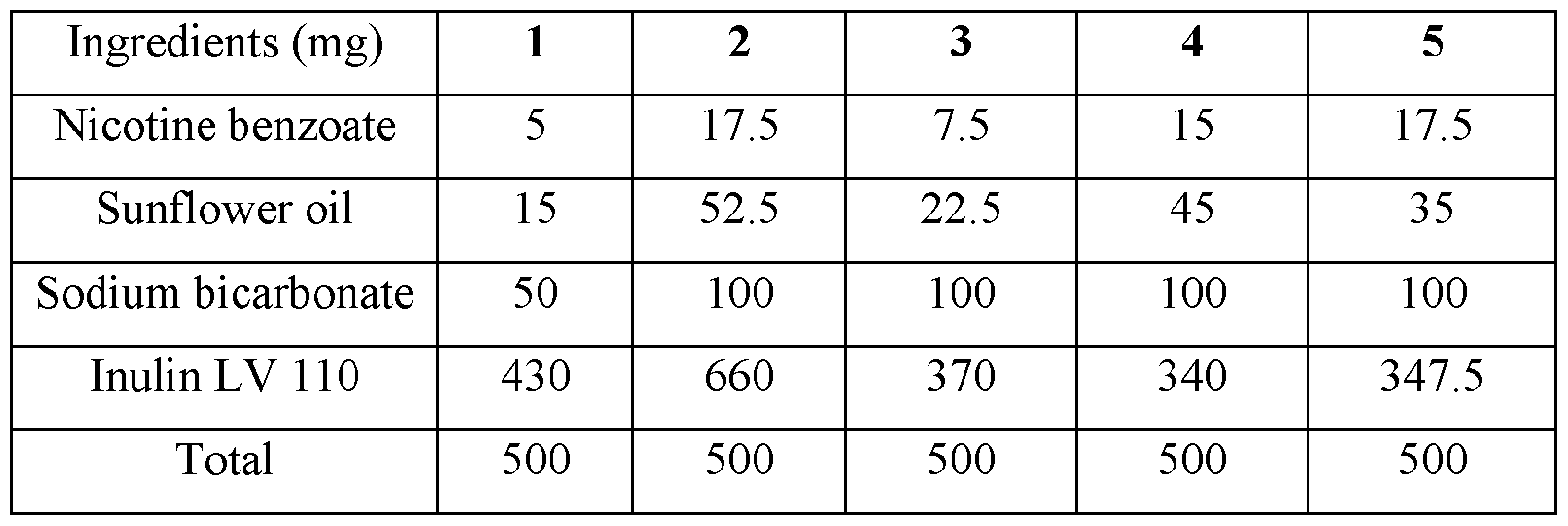
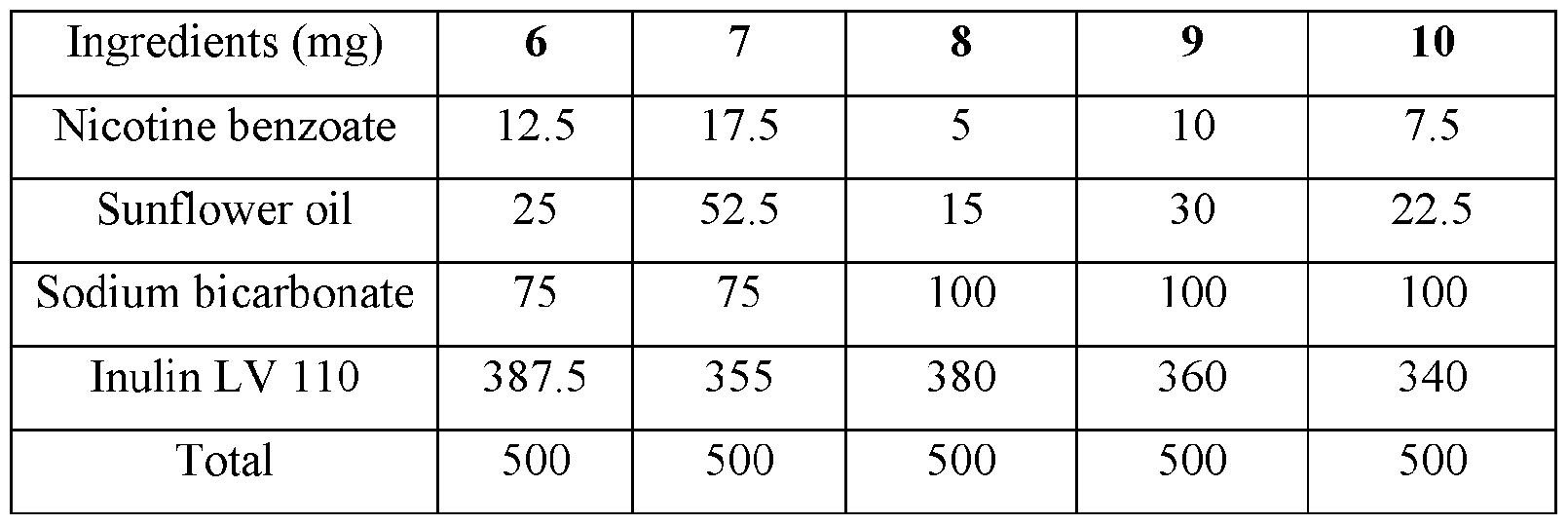
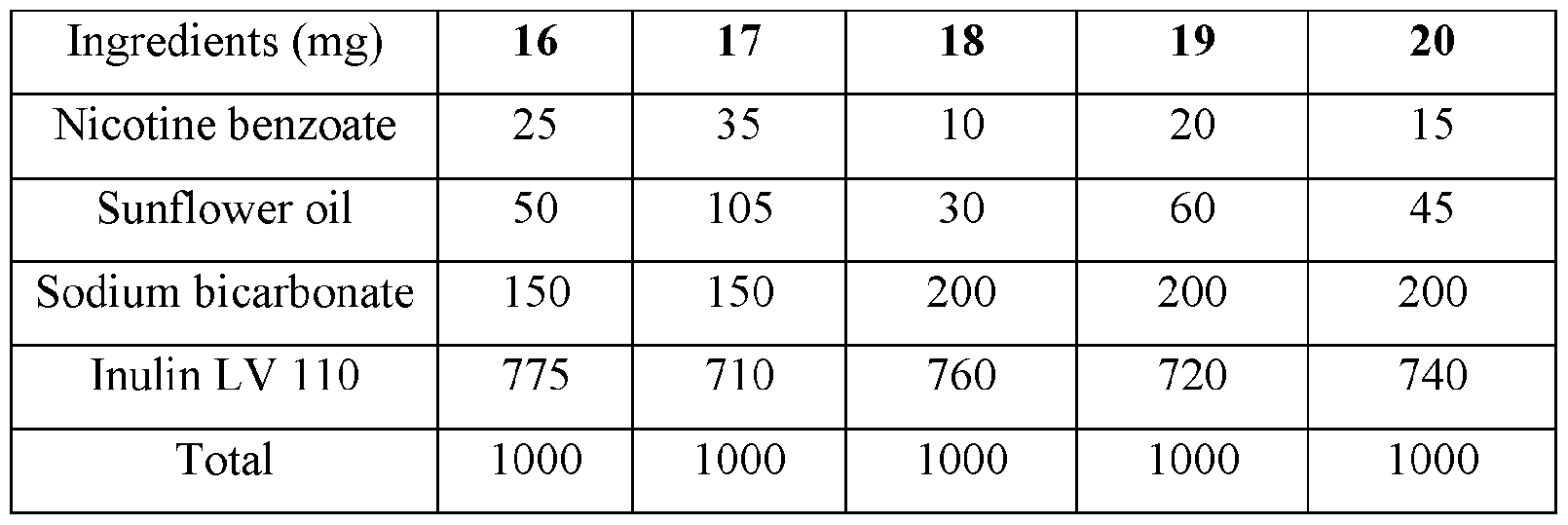
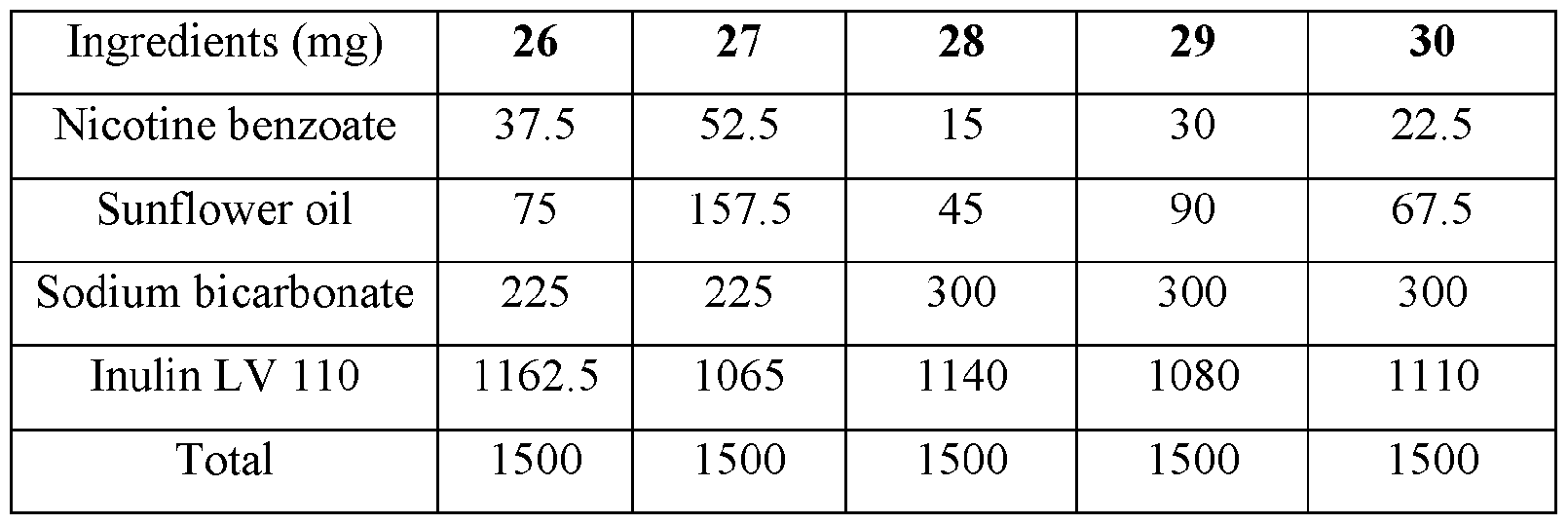
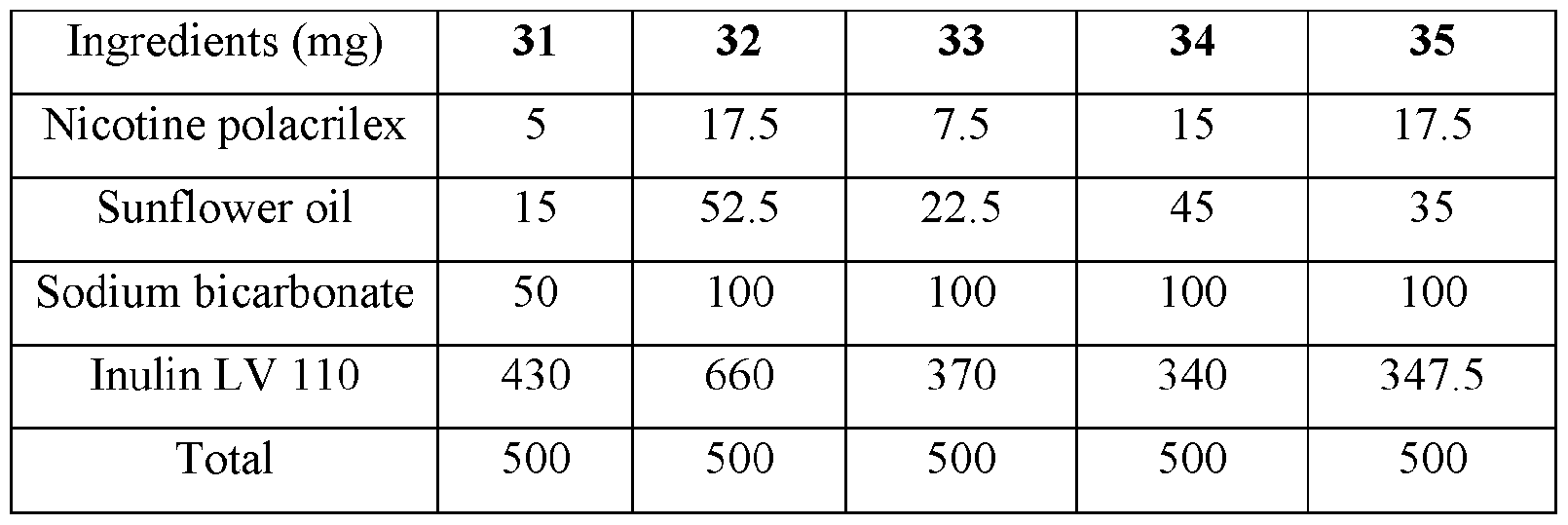
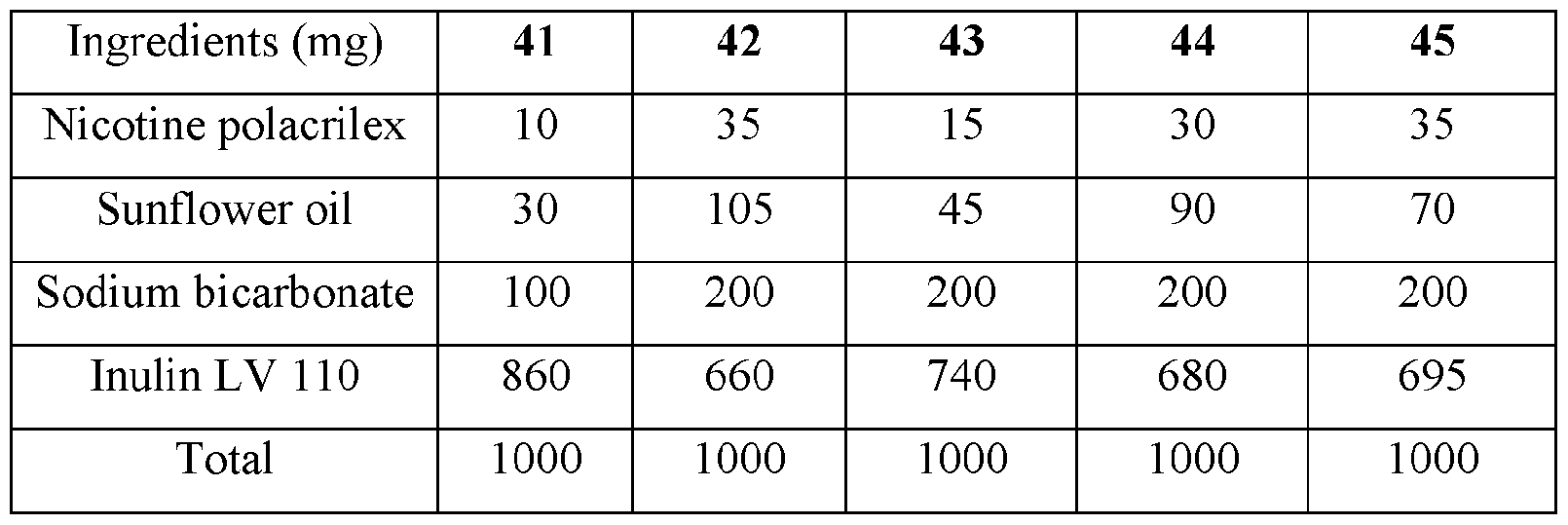
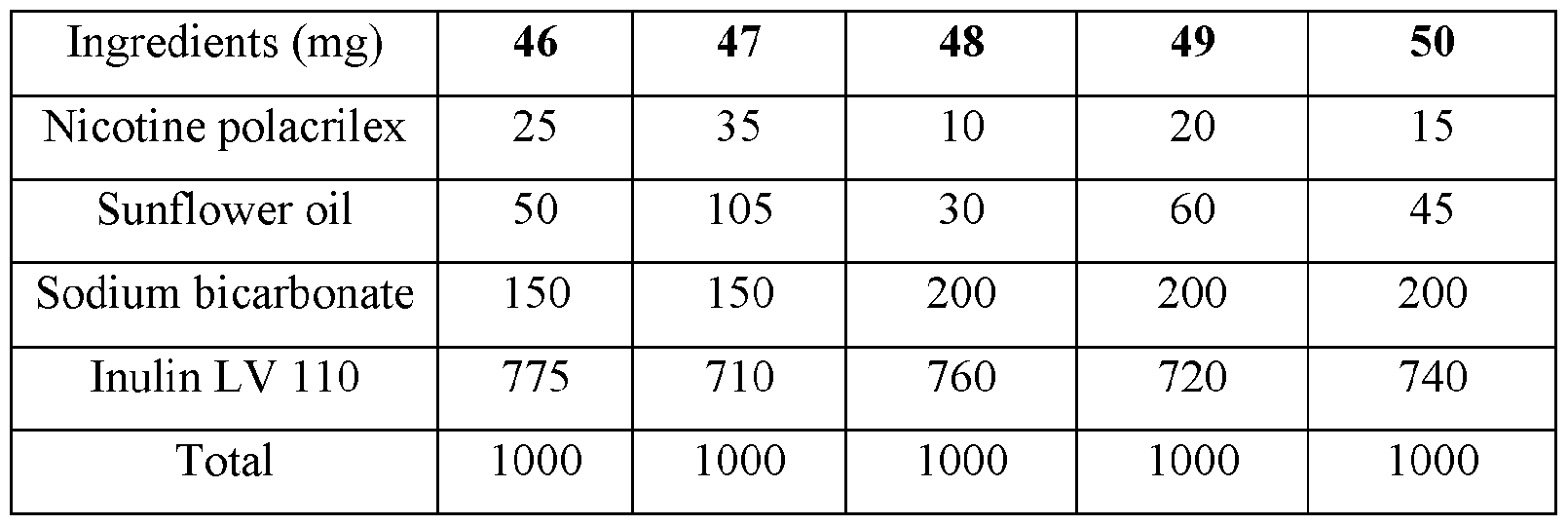
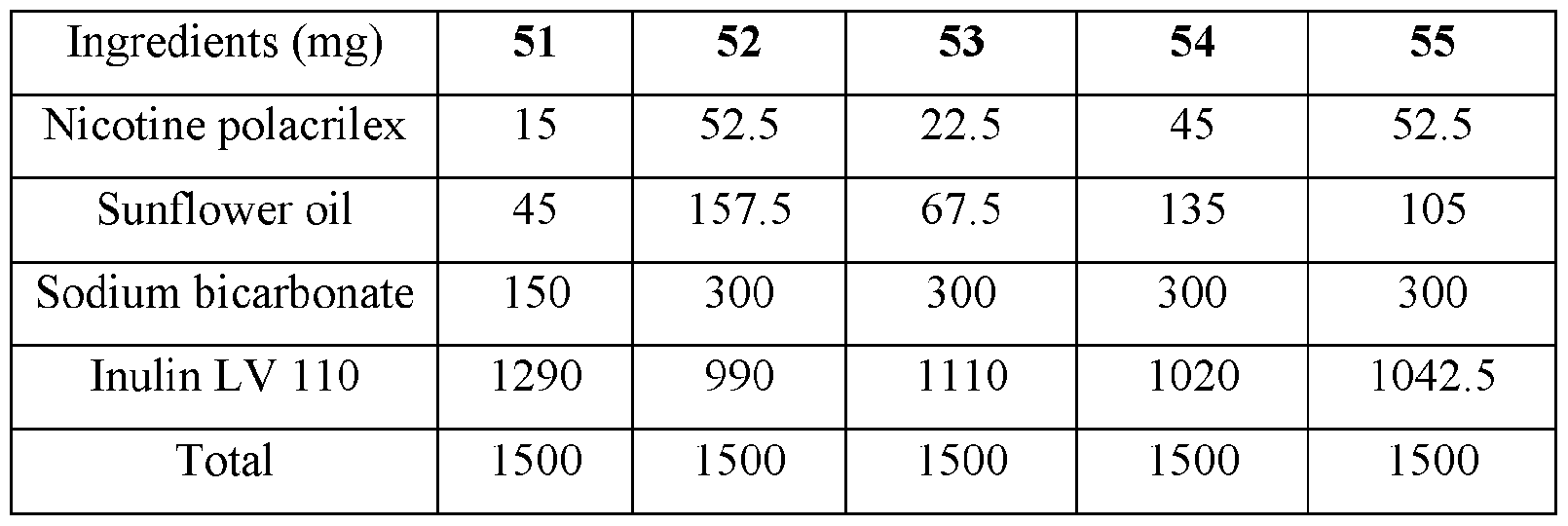
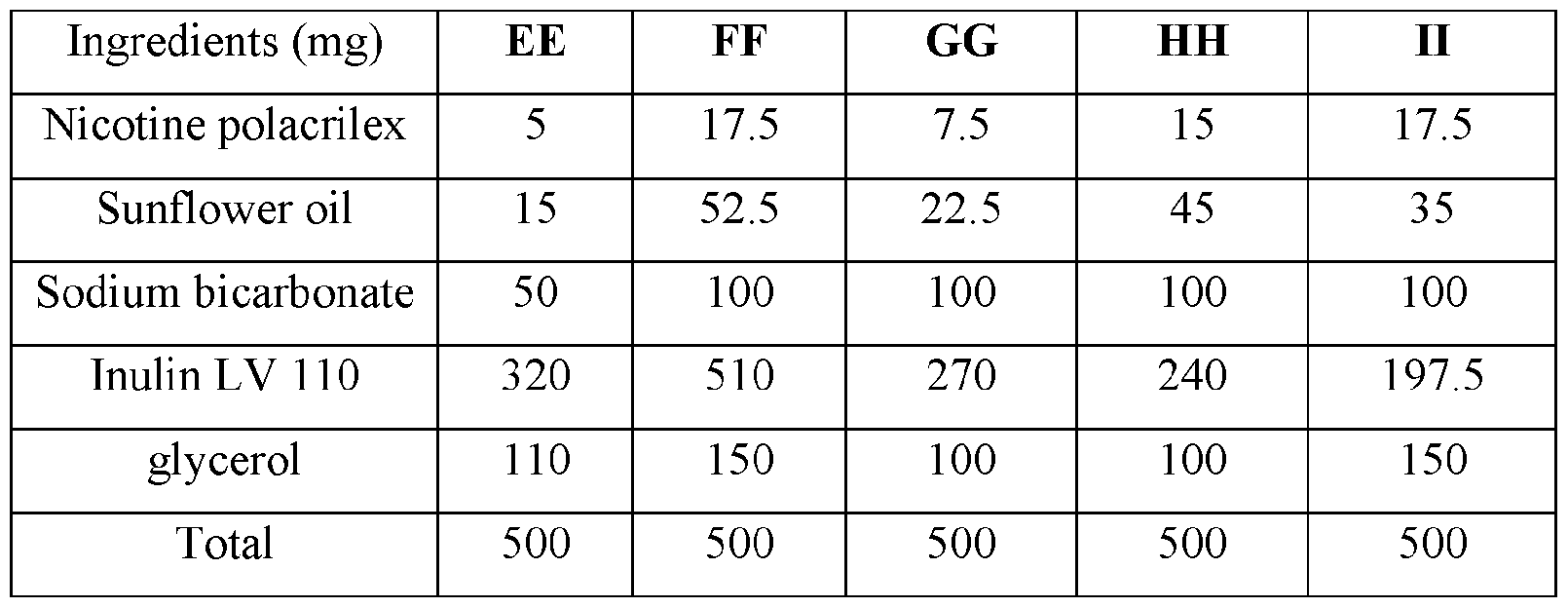
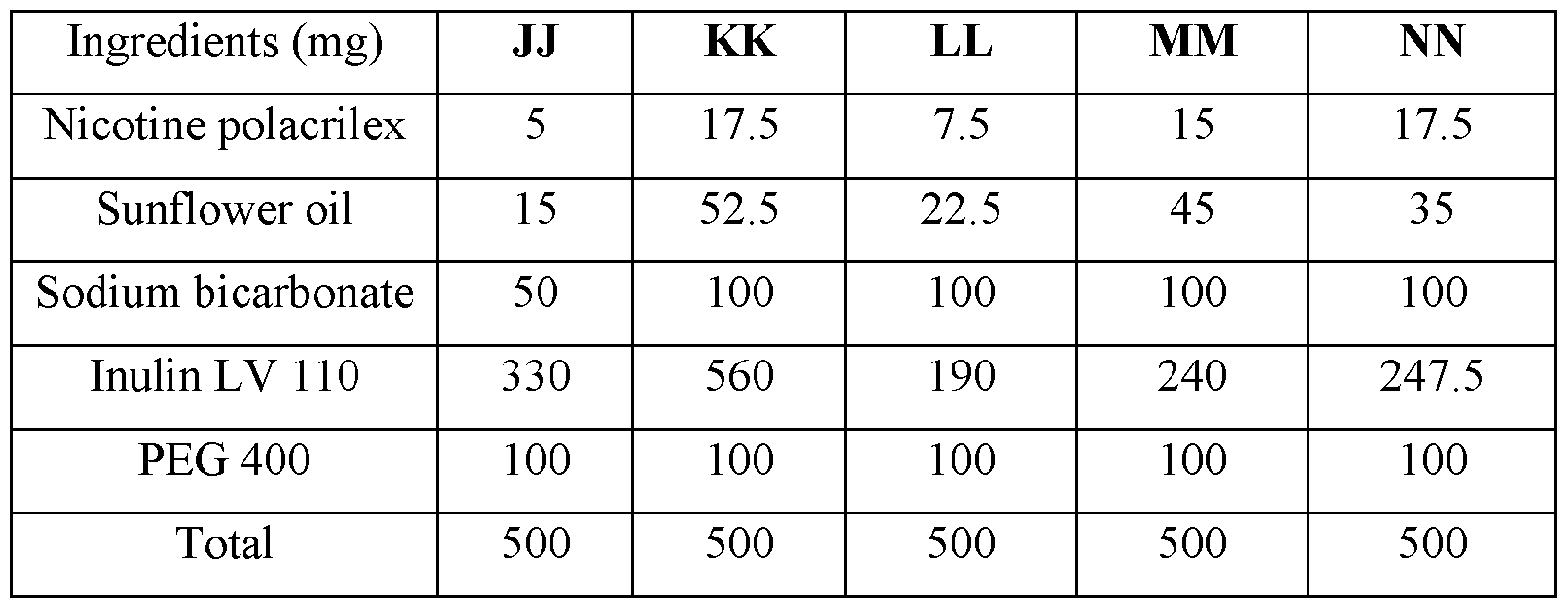
- the base compositions can comprise: a) from about 5 mg to about 50 mg by weight of nicotine, a nicotine salt, nicotine in combination with a resin, or mixtures thereof; b) from about 15 mg to about 160 mg by weight of sunflower oil; and c) from about 50 mg to about 300 mg by weight of sodium bicarbonate.
- the disclosed base compositions can comprise from 5 mg to about 50 mg by weight of nicotine, a nicotine salt, nicotine in combination with a resin, or mixtures thereof.
- the base compositions can comprise from 10 mg to about 50 mg by weight of nicotine, a nicotine salt, nicotine in combination with a resin, or mixtures thereof. In another embodiment the base compositions can comprise from 15 mg to about 40 mg by weight of nicotine, a nicotine salt, nicotine in combination with a resin, or mixtures thereof. In a further embodiment the base compositions can comprise from 10 mg to about 30 mg by weight of nicotine, a nicotine salt, nicotine in combination with a resin, or mixtures thereof. In a still further embodiment the base compositions can comprise from 15 mg to about 30 mg by weight of nicotine, a nicotine salt, nicotine in combination with a resin, or mixtures thereof.
- the base compositions can comprise from 10 mg to about 25 mg by weight of nicotine, a nicotine salt, nicotine in combination with a resin, or mixtures thereof.
- the base compositions can comprise, for example, 5 mg, 6 mg, 7 mg, 8 mg, 9 mg, 10 mg, 11 mg, 12 mg, 13 mg, 14 mg, 15 mg, 16 mg, 17 mg, 18 mg, 19 mg, 20 mg, 21 mg, 22 mg, 23 mg, 24 mg, 25 mg, 26 mg, 27 mg, 28 mg, 29 mg, 30 mg, 31 mg, 32 mg, 33 mg, 34 mg, 35 mg, 36 mg, 37 mg, 38 mg, 39 mg, 40 mg, 41 mg, 42 mg, 43 mg, 44 mg, 45 mg, 46 mg, 47 mg, 48 mg, 49 mg, or 50 mg by weight of nicotine, a nicotine salt, nicotine in combination with a resin, or mixtures thereof or any fractional amount, for example, 7.5 mg, 22.5 mg, and 34.6 mg.
- the disclosed base compositions can comprise from about 15 mg to about 160 mg by weight of sunflower oil. In one embodiment the base compositions can comprise from about 15 mg to about 160 mg by weight of sunflower oil. In another embodiment the base compositions can comprise from about 25 mg to about 120 mg by weight of sunflower oil. In a further embodiment the base compositions can comprise from about 40 mg to about 100 mg by weight of sunflower oil. In a still further embodiment the base compositions can comprise from about 50 mg to about 150 mg by weight of sunflower oil. In a yet further embodiment the base compositions can comprise from about 75 mg to about 120 mg by weight of sunflower oil.
- the disclosed base compositions can comprise, for example, 15 mg, 16 mg, 17 mg, 18 mg, 19 mg, 20 mg, 21 mg, 22 mg, 23 mg, 24 mg, 25 mg, 26 mg, 27 mg, 28 mg, 29 mg, 30 mg, 31 mg, 32 mg, 33 mg, 34 mg, 35 mg, 36 mg, 37 mg, 38 mg, 39 mg, 40 mg, 41 mg, 42 mg, 43 mg, 44 mg, 45 mg, 46 mg, 47 mg, 48 mg, 49 mg, 50 mg, 51 mg, 52 mg, 53 mg, 54 mg, 55 mg, 56 mg, 57 mg, 58 mg, 59 mg, 60 mg, 61 mg, 62 mg, 63 mg, 64 mg, 65 mg, 66 mg, 67 mg, 68 mg, 69 mg, 70 mg, 71 mg, 72 mg, 73 mg, 74 mg, 75 mg, 76 mg, 77 mg, 78 mg, 79 mg, 80 mg, 81 mg, 82 mg, 83 mg, 84 mg, 85 mg, 86 mg, 87 mg,
- the disclosed base compositions can comprise from about 50 mg to about 300 mg by weight of sodium bicarbonate. In one embodiment the base compositions comprise from about 50 mg to about 100 mg by weight of sodium bicarbonate. In another embodiment the base compositions comprise from about 75 mg to about 100 mg by weight of sodium bicarbonate. In a further embodiment the base compositions comprise from about 100 mg to about 200 mg by weight of sodium bicarbonate. In still further embodiment the base compositions comprise from about 150 mg to about 200 mg by weight of sodium bicarbonate. In a yet further embodiment the base compositions comprise from about 150 mg to about 300 mg by weight of sodium bicarbonate. In a yet another embodiment the base compositions comprise from about 225 mg to about 300 mg by weight of sodium bicarbonate.
- the disclosed base composition can comprise, for example, 50 mg, 51 mg, 52 mg, 53 mg, 54 mg, 55 mg, 56 mg, 57 mg, 58 mg, 59 mg, 60 mg, 61 mg, 62 mg, 63 mg, 64 mg, 65 mg, 66 mg, 67 mg, 68 mg, 69 mg, 70 mg, 71 mg, 72 mg, 73 mg, 74 mg, 75 mg, 76 mg, 77 mg, 78 mg, 79 mg, 80 mg, 81 mg, 82 mg, 83 mg, 84 mg, 85 mg, 86 mg, 87 mg, 88 mg, 89 mg, 90 mg, 90 mg, 91 mg, 92 mg, 93 mg, 94 mg, 95 mg, 96 mg, 97 mg, 98 mg, 99 mg, 100 mg, 101 mg, 102 mg, 103, mg, 104 mg, 105 mg, 106 mg, 107 mg, 108 mg, 109 mg, 110 mg, 111 mg, 112 mg, 113 mg, 114 mg,
- the disclosed nicotine compounds are chosen from nicotine, pharmacologically acceptable salts of nicotine, a nicotine complexes, and polymer resins of containing nicotine.
- nicotine salts includes nicotine benzoate, nicotine lactate, nicotine malate, nicotine ditartrate, nicotine salicylate, nicotine citrate and nicotine levulinate.
- nicotine in combination with a resin includes nicotine polacrilex and nicotine resinate.
- the nicotine salt is nicotine benzoate. In another nonlimiting example the nicotine salt is nicotine lactate. In a further non-limiting example the nicotine salt is nicotine malate. In a yet further non-limiting example the nicotine salt is nicotine ditartrate. In a still yet further non-limiting example the nicotine salt is nicotine salicylate. In a yet another non-limiting example the nicotine salt is nicotine citrate. In a still yet another non-limiting example the nicotine salt is nicotine levulinate.
- the disclosed carriers are polysaccharides.
- Non-limiting examples of poly saccharide carriers include inulin, galactogen, cellulose, chitin, pectin, psyllium, guar, hemicellulose, potato starch, and partially hydrolyzed polysaccharides.
- the carriers are sugar alcohols, for example, sorbitol, erythritol, xylitol, lactitol, maltitol, mannitol, hydrogenated starch hydrolysates, isomaltose, or any combination thereof.
- carrier component is based on a native or chemically modified agar, alginates, carrageenan gum, cellulose, chitosan, chitin, cyclodextrin, dextran, gellan gum, glycogen, glycosaminoglycan, gum karaya, inulin, pectin, polydextrose, xanthan gum, or any other starches, gums or other polysaccharide, including functionalized derivatives, dextrinized, hydrolyzed, oxidized, alkylated, hydroxyalkylated, acetylated, fractionated, and physically modified starches and mixtures thereof.
- glycerin and/or propylene glycol can be added as a carrier.
- the carrier can serve as a bulking agent.
- microcrystalline cellulose is utilized as a carrier in the base compositions and as a bulking agent in the pouches disclosed herein below.
- two or more carriers can be combined, for example, microcrystalline cellulose and inulin. This combination can be utilized in both the base composition, as well as in the pouches.
- dextrin is added as a bulking agent, however, dextrin can also serve as carrier for any flavors that the formulator wishes to add.
- ethylvanillin is a compound which provides vanilla flavoring. Ethylvanillin can be compounded with dextrin, microcrystalline cellulose or inulin and then admixed with the bulking agents or other carriers.
- one of the carriers is water soluble while others are not. This allows the formulator to control the release of the active base when the active base is delivered by way of a non-water soluble, but water permeable pouch as described herein below. This combining of carriers allows the delivery of nicotine either via a nicotine salt or by way of a polymer supported nicotine, for example, polacrilex.
- the disclosed compositions can comprise from about 80% to about 95% by weight of one or more carriers. In one embodiment the disclosed compositions can comprise from about 80% to about 90% by weight of one or more carriers. In another embodiment the disclosed compositions can comprise from about 85% to about 95% by weight of one or more carriers. In a further embodiment the disclosed compositions can comprise from about 85% to about 90% by weight of one or more carriers.
- compositions can comprise about 0.05% or less of an antioxidant.
- an antioxidant includes butylated hydroxytoluene (BHT), butylated hydroxyanisole (BHA), propyl gallate (PG), tert-butyl hydroquinone (TBHQ), and mixtures thereof.
- BHT butylated hydroxytoluene
- BHA butylated hydroxyanisole
- PG propyl gallate
- TBHQ tert-butyl hydroquinone
- kits for sublingual delivery of nicotine contain a base nicotine delivery system which comprises the active ingredients and a non-water soluble liquid permeable pouch into which the active ingredients and any necessary adjunct ingredients useful for delivery of the nicotine, nicotine salt of nicotine resin compositions.
- the kit comprises a pouch containing a disclosed composition, comprising:
- A) a liquid permeable pouch comprising a non-nicotine composition comprising: a) one or more delivery agents; and b) a bulking agent; and
- B) a base nicotine delivery composition comprising: a) nicotine, a nicotine salt, nicotine in combination with a resin, or mixtures thereof; b) sunflower oil; c) sodium bicarbonate; and d) the balance one or more carriers.
- kits contain one or more agents that control the release of nicotine into the mouth of the use. These agents are typically formulated after assembly of the base nicotine delivery compositions; however, the formulator can add a delivery control agent as part of a carrier system.
- the delivery agents are solubilizers, for example, lecithins, polyoxyethylene stearate, polyoxyethylene sorbitan fatty acid esters, fatty acid salts, mono and diacetyl tartaric acid esters of mono and diglycerides of edible fatty acids, citric acid esters of mono and diglycerides of edible fatty acids, saccharose esters of fatty acids, polyglycerol esters of fatty acids, polyglycerol esters of interesterified castor oil acid (E476), sodium stearoyl lactylate, sodium lauryl sulfate and sorbitan esters of fatty acids and polyoxy ethylated hydrogenated castor oil (for example, CREMOPHORTM), block copolymers of ethylene oxide and propylene oxide (for example, one or more PLURONICSTM or POLOXAMERSTM), polyoxyethylene fatty alcohol ethers, polyoxyethylene sorbitan fatty acid esters, sorbitan esters of
- the delivery agent is chosen from sodium stearoyl lactylate, sodium lauryl sulfate, glycerol, propylene glycol, b-cyclodextrin and propylene glycol 400 (PEG 400).
- Non-limiting examples of solubilizers includes glycerol, propylene glycol, b- cyclodextrin and propylene glycol 400 (PEG 400).
- kits comprise:
- A) a liquid permeable pouch comprising a non-nicotine composition comprising: a) one or more delivery agents; and b) a bulking agent; and
- B) a base nicotine delivery composition comprising: a) nicotine, a nicotine salt, nicotine in combination with a resin, or mixtures thereof; b) sunflower oil; c) sodium bicarbonate; and d) the balance one or more carriers.
- kits comprise:
- A) a liquid permeable pouch comprising a non-nicotine composition comprising: a) one or more delivery agents; and b) a bulking agent; and
- kits comprise:
- A) a liquid permeable pouch comprising a non-nicotine composition comprising: a) maltitol; and b) an admixture of microcrystalline cellulose and inulin; and
- B) a base nicotine delivery composition comprising: a) from about 1% to about 6% by weight of nicotine, a nicotine salt, nicotine in combination with a resin, or mixtures thereof; b) from about 3% to about 20% by weight of sunflower oil; c) from about 10% to about 20% by weight of sodium bicarbonate; and d) the balance: i) dextrose; and ii) a flavorant.
- Non-limiting examples of flavorants include apple, banana, cherry, cinnamon, grape, orange, pear, pineapple, raspberry, blueberry, strawberry, spearmint, peppermint, wintergreen, and vanilla.
- kit comprising:
- A) a liquid permeable pouch comprising a non-nicotine composition comprising: a) one or more delivery control agents; and b) a bulking agent; and
- B) a base nicotine delivery composition comprising: a) nicotine, a nicotine salt, nicotine in combination with a resin, or mixtures thereof; b) sunflower oil; c) sodium bicarbonate; and d) the balance one or more carriers.
- the kit comprises:
- A) a liquid permeable pouch comprising a non-nicotine composition comprising: a) one or more delivery control agents; and b) a bulking agent; and
- the kit comprises:
- A) a liquid permeable pouch comprising a non-nicotine composition comprising: a) one or more delivery control agents; and b) a bulking agent; and
- the disclosed base compositions can be prepared by the following general procedure. Nicotine, a nicotine salt, or nicotine in combination with a resin, is combined with sunflower oil in a vessel with adequate stirring.
- the amount of each ingredient varies depending upon the formulator’s choice of the ratio of the nicotine-containing ingredient and sunflower oil, i.e., the ratio of nicotine-containing ingredient to sunflower oil can be, as disclosed herein above, from about 1 : 1 to about 1 :3.
- the choice of ratio will also dictate the relative amounts of adjunct ingredients that are added.
- the nicotine-containing ingredient sunflower oil admixture is then slowly heated to from about 50 °C to about 75 °C, again predicated on the ratio of ingredients and the choice of excipients.
- antioxidants as well as other adjunct ingredients can be optionally added to the nicotine-containing compound/sunflower oil admixture during heating.
- the amount and ratio of any antioxidants added to the admixture varies depending upon the formulator’s choice. In one non-limiting embodiment, the amount of antioxidant is from about 0.01% to about 0.10% by weight of the admixture.
- the resulting admixture is then slowly added to a dry particulate substrate with sufficient mixing to form a homogenous dispersion.
- the quantity of the admixture that is added to the substrate is from about 5% to about 60% by weight.
- the final dispersion is then dehydrated by which ever means chosen by the formulator, for example, oven drying, lyophilization, convection drying, microwave radiation, etc. In one non-limiting embodiment, the dispersion is dried from about 45 to about 135 minutes. Depending upon many factors including the type of adjunct ingredients and the ratio of the nicotine-containing compound to sunflower oil, the time can be shortened or lengthened.
- an alkalizing agent is incorporated.
- sodium bicarbonate is used as the alkalizing agent.
- the amount of alkalizing agent is predicated on the amounts of other ingredients and the choice of substrate.
- the composition can comprise from about 1% to about 25% by weight of the alkalizing agent.
- adjunct ingredients can be added.
- Non-limiting examples include bulking agents which provide a mouthfeel that is compatible with the pouches, thereby providing the user with a feeling of “substance” inside the pouch.
- Bulking agents include microcrystalline cellulose and inulin.
- sweeteners for example, maltitol, and/or flavoring compounds are added to provide different oral sensations.
- composition can then be further compounded with other adjunct ingredients, at levels that vary depending upon the formulator’ s choice, such as bulking agents (e.g., microcrystalline cellulose), high potency sweeteners (e.g., maltitol) and/or flavoring compounds, and ultimately rendered in various different oral or intraoral form factors.
- adjunct ingredients e.g., bulking agents (e.g., microcrystalline cellulose), high potency sweeteners (e.g., maltitol) and/or flavoring compounds, and ultimately rendered in various different oral or intraoral form factors.
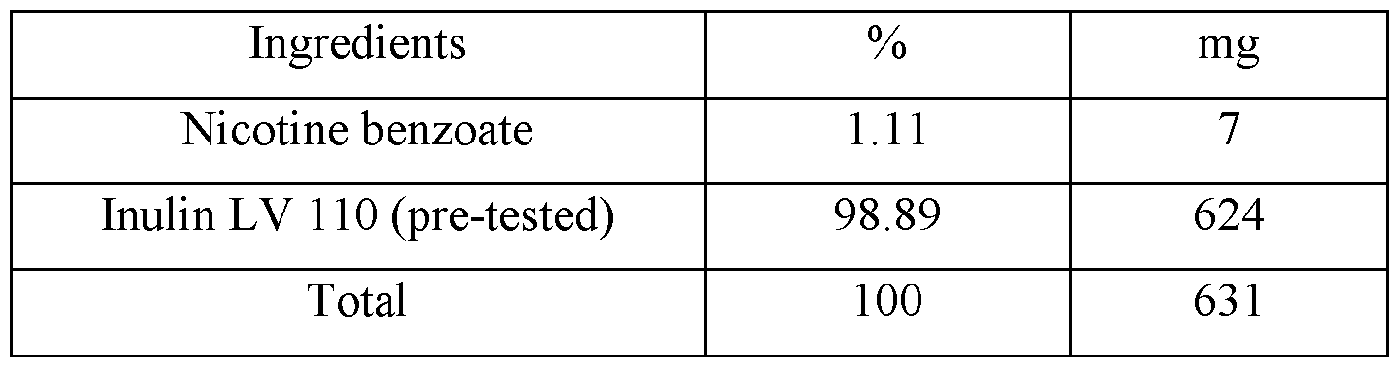
- nicotine benzoate (15 g) and sunflower oil (45 g) are combined in a stainless-steel reaction vessel with efficient stirring and heated to 50 °C until homogeneous.
- the nicotine benzoate sunflower oil admixture is then slowly metered into inulin (500 g) as a dry particulate substrate compound while mixing until homogeneously dispersed.
- Sodium bicarbonate (20 g) is added while mixing until homogenously dispersed.
- the admixture is then placed in a convection airflow dehydration chamber for 90 minutes to remove remaining moisture and effect a molecular association between the nicotine and the sunflower oil infused dry particulate.
- the resulting composition is then combined with microcrystalline cellulose (200 g), maltitol (175 g) and spearmint flavoring (100 g) and then charged to unit dose oral pouches.
- the properties of the pouch can influence the release of the nicotine, nicotine salt, or nicotine in combination with a resin from the pouch composition and thereby possibly influence the rate of uptake by the user.
- the disclosed pouches comprise water insoluble fiber which allows moisture, typically the user’s saliva, to enter the pouch and solubilize the water- soluble components.
- water-insoluble pouches which can comprise insoluble fiber, for example, wheat fibers, oat fibers, pea fibers, rice fiber, maize fibers, oat fibers, tomato fibers, barley fibers, rye fibers, sugar beet fibers, buckwheat fibers, potato fibers, cellulose fibers, apple fibers, cocoa fibers, cellulose fiber, powdered cellulose, bamboo fibers, bran fibers or combinations thereof.
- insoluble fiber for example, wheat fibers, oat fibers, pea fibers, rice fiber, maize fibers, oat fibers, tomato fibers, barley fibers, rye fibers, sugar beet fibers, buckwheat fibers, potato fibers, cellulose fibers, apple fibers, cocoa fibers, cellulose fiber, powdered cellulose, bamboo fibers, bran fibers or combinations thereof.
- the disclosed pouches comprise cellulose prepared by processing alpha-cellulose obtained as a pulp from strains of fibrous plant materials, such as wood pulp.
- the pouches can comprise wheat fibers, oat fibers, or combinations thereof.
- the amount of water-insoluble fiber can be reduced without compromising the mouthfeel during use. It is important that the pouch material does not cause swelling in use because this fact can counteract the dissolution of the water-soluble component, thereby preventing the user from experiencing any decrease in pouch content during use.
- the pouch composition can also provide for a desirable mouthfeel such as a soft and/or sticky texture.
- a desirable texture and mouthfeel can be obtained while still being able to store manufactured pouches together in abutment, for example, in cans and the like without sticking or clumping together to result in ruptures of the pouches when being removed.
- the desirable mouthfeel can in some embodiments also comprise a tingling sensation reminiscent of tobacco pouches, but without many of the undesirable effects associated therewith, for example, discoloring of tissue.
- kits comprise an active base composition and a pouch for delivery of nicotine sublingually to the user.
- kits comprise a water-permeable pouch, into which an active base composition and a delivery system is added.
- the disclosed pouches comprise:
- A) from about 5% to about 20% by weight of an active base composition comprising” a) from about 1% to about 6% by weight of nicotine, a nicotine salt, nicotine in combination with a resin, or mixtures thereof; b) from about 3% to about 20% by weight of sunflower oil; c) from about 10% to about 20% by weight of sodium bicarbonate; and
- the pouches comprise:
- A) from about 70 mg to about 510 mg of an active base composition comprising: a) from about 5 mg to about 50 mg by weight of nicotine benzoate; b) from about 15 mg to about 160 mg by weight of sunflower oil; c) from about 50 mg to about 300 mg by weight of sodium bicarbonate;
- the one or more carriers serves as the delivery control agent.
- a pouch comprising: a) from about 5 mg to about 50 mg by weight of nicotine benzoate; b) from about 15 mg to about 160 mg by weight of sunflower oil; c) from about 50 mg to about 300 mg by weight of sodium bicarbonate; and d) from about 300 mg to about 1300 mg by weight of a carrier chosen from inulin, galactogen, cellulose, chitin, pectin, psyllium, guar, hemicellulose, potato starch, or partially hydrolyzed polysaccharides.
- a pouch comprising: a) from about 5 mg to about 50 mg by weight of nicotine benzoate; b) from about 15 mg to about 160 mg by weight of sunflower oil; c) from about 50 mg to about 300 mg by weight of sodium bicarbonate; and d) from about 300 mg to about 1300 mg by weight of a carrier chosen from, sorbitol, erythritol, xylitol, lactitol, maltitol, mannitol, hydrogenated starch hydrolysates, isomaltose, or any combination thereof.
- a carrier chosen from, sorbitol, erythritol, xylitol, lactitol, maltitol, mannitol, hydrogenated starch hydrolysates, isomaltose, or any combination thereof.
- a pouch comprising: a) from about 5 mg to about 50 mg by weight of nicotine benzoate; b) from about 15 mg to about 160 mg by weight of sunflower oil; c) from about 50 mg to about 300 mg by weight of sodium bicarbonate; and d) from about 300 mg to about 1300 mg by weight of inulin.
- a pouch comprising: a) from about 5 mg to about 50 mg by weight of nicotine benzoate; b) from about 15 mg to about 160 mg by weight of sunflower oil; c) from about 50 mg to about 300 mg by weight of sodium bicarbonate; and d) from about 300 mg to about 1300 mg by weight of microcrystalline cellulose.
- a pouch comprising: a) from about 5 mg to about 50 mg by weight of nicotine benzoat; b) from about 15 mg to about 160 mg by weight of sunflower oil; c) from about 50 mg to about 300 mg by weight of sodium bicarbonate; and d) from about 300 mg to about 1300 mg by weight of an admixture of inulin and microcrystalline cellulose.
- the pouches comprise:
- A) from about 70 mg to about 510 mg of an active base composition comprising: a) from about 5 mg to about 50 mg by weight of nicotine polacrilex; b) from about 15 mg to about 160 mg by weight of sunflower oil; c) from about 50 mg to about 300 mg by weight of sodium bicarbonate;
- the one or more carriers serves as the delivery control agent.
- a pouch comprising: a) from about 5 mg to about 50 mg by weight of nicotine polacrilex; b) from about 15 mg to about 160 mg by weight of sunflower oil; c) from about 50 mg to about 300 mg by weight of sodium bicarbonate; and d) from about 300 mg to about 1300 mg by weight of a carrier chosen from inulin, galactogen, cellulose, chitin, pectin, psyllium, guar, hemicellulose, potato starch, or partially hydrolyzed polysaccharides.
- a pouch comprising: a) from about 5 mg to about 50 mg by weight of nicotine polacrilex; b) from about 15 mg to about 160 mg by weight of sunflower oil; c) from about 50 mg to about 300 mg by weight of sodium bicarbonate; and d) from about 300 mg to about 1300 mg by weight of a carrier chosen from, sorbitol, erythritol, xylitol, lactitol, maltitol, mannitol, hydrogenated starch hydrolysates, isomaltose, or any combination thereof.
- a carrier chosen from, sorbitol, erythritol, xylitol, lactitol, maltitol, mannitol, hydrogenated starch hydrolysates, isomaltose, or any combination thereof.
- a pouch comprising: a) from about 5 mg to about 50 mg by weight of nicotine polacrilex; b) from about 15 mg to about 160 mg by weight of sunflower oil; c) from about 50 mg to about 300 mg by weight of sodium bicarbonate; and d) from about 300 mg to about 1300 mg by weight of inulin.
- a pouch comprising: a) from about 5 mg to about 50 mg by weight of nicotine polacrilex; b) from about 15 mg to about 160 mg by weight of sunflower oil; c) from about 50 mg to about 300 mg by weight of sodium bicarbonate; and d) from about 300 mg to about 1300 mg by weight of microcrystalline cellulose.
- a pouch comprising: a) from about 5 mg to about 50 mg by weight of nicotine polacrilex; b) from about 15 mg to about 160 mg by weight of sunflower oil; c) from about 50 mg to about 300 mg by weight of sodium bicarbonate; and d) from about 300 mg to about 1300 mg by weight of an admixture of inulin and microcrystalline cellulose.
- the disclosed compositions can comprise from about 80% to about 95% by weight of one or more delivery control agents, carriers, solubilizers or mixtures thereof. In one embodiment the disclosed compositions can comprise from about 80% to about 90% by weight of one or more delivery control agents, carriers, solubilizers or mixtures thereof. In another embodiment the disclosed compositions can comprise from about 85% to about 95% by weight of one or more delivery control agents, carriers, solubilizers or mixtures thereof. In a further embodiment the disclosed compositions can comprise from about 85% to about 90% by weight of one or more delivery control agents, carriers, solubilizers or mixtures thereof
- the disclosed base compositions can be prepared by the following general procedure. Nicotine, a nicotine salt, or nicotine in combination with a resin, is combined with sunflower oil in a vessel with adequate stirring.
- the amount of each ingredient varies depending upon the formulator’s choice of the ratio of the nicotine-containing ingredient and sunflower oil, i.e., the ratio of nicotine-containing ingredient to sunflower oil is from about 1 : 1 to about 1:3.
- the choice of ratio will also dictate the relative amounts of adjunct ingredients that are added.
- the nicotine-containing ingredient sunflower oil admixture is then slowly heated to from about 50 °C to about 75 °C, again predicated on the ratio of ingredients and the choice of excipients.
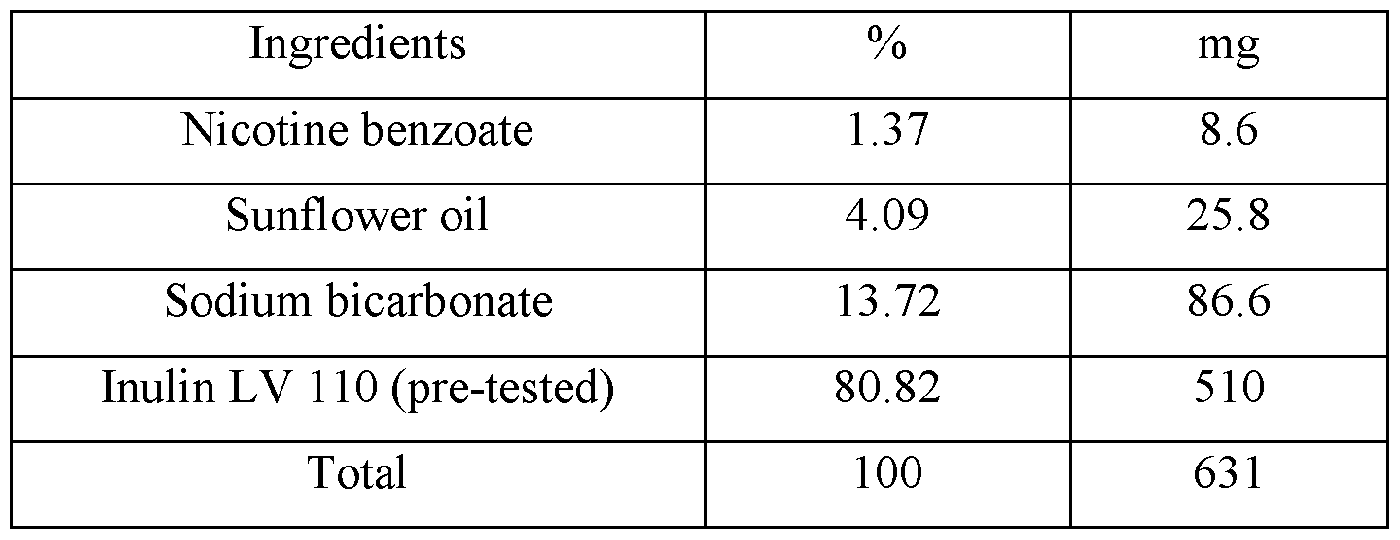
- nicotine benzoate (15 g) and sunflower oil (45 g) are combined in a stainless-steel reaction vessel with efficient stirring and heated to 50 °C until homogeneous.
- Inulin (500 g) is slowly metered in and stirring continued until all the inulin is dispersed.
- Sodium bicarbonate (150 g) is slowly added while raising the temperature to 60 °C.
- inulin (790 g) is added at a rate to maintain a homogeneous admixture.
- the admixture is then slowly cooled to 30 °C and placed in a vacuum oven for 5 hours to remove all of the remaining moisture.
- the resulting composition can then be combined with additional inulin or microcrystalline cellulose then charged to one or more pouches.
- compositions delivered by way of an insoluble plant or synthetic pouch are non-limiting examples of compositions delivered by way of an insoluble plant or synthetic pouch.
- compositions are more effective in increasing the plasma level of nicotine via oral delivery than providing nicotine alone in a carrier.
- the following animal study provides conclusive proof of this fact.
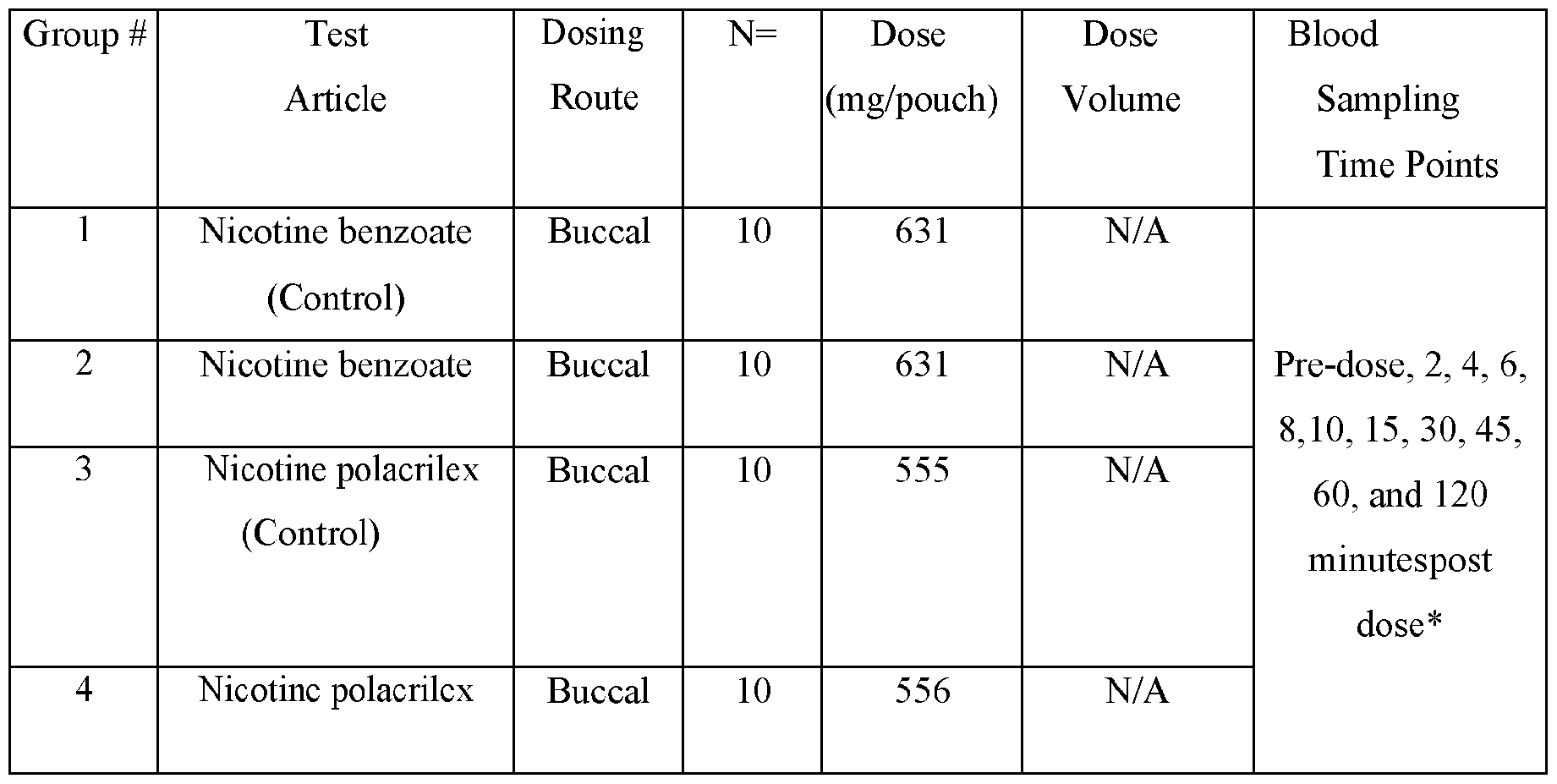
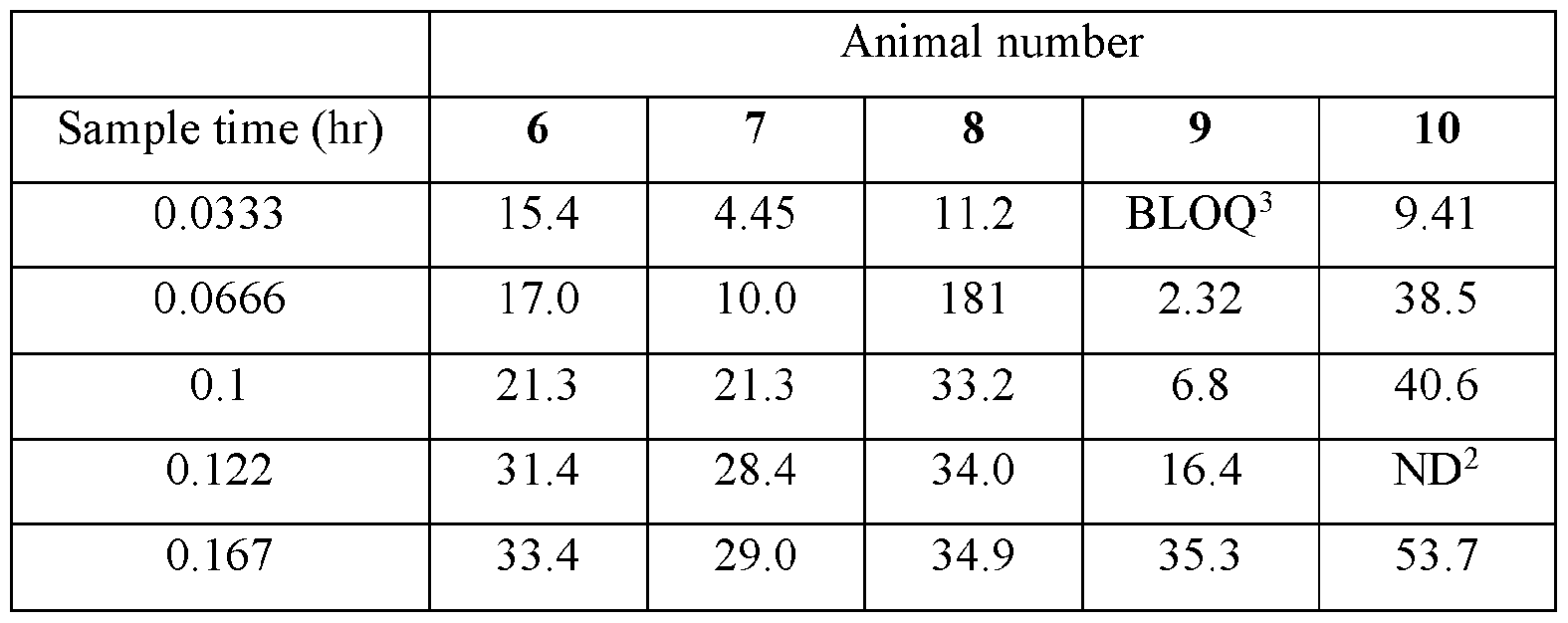
- the disclosed animal studies were conducted utilizing the disclosed compositions. Table I summarizes the Study design. Male Beagle dogs from Marshall Bioresources were utilized for this study. Animals were identified by ear tattoo and cage label. The study was not blinded. The animals were healthy at the start of the study. Body weights were recorded at each dosing time point. General health observations were recorded at each dosing and sample collection time point for the duration of the study.
- Nicotine 4 mg per pouch, was administered via buccal administration. Animals were anesthetized with propofol at a dose of 6 mg/kg, animals were then intubated and maintained in an anesthetic state using isoflurane at 1-5% and 2 L of oxygen flow.
- the pouch with test article was placed in the buccal space, rinsed with a small volume of water (0.5 - ImL). Every 5 minutes after placing the pouch the test article test article in the buccal space, the isoflurane mask was removed and the pouch gently squeezed. Special attention was taken to ensure saliva did not leak from the mouth. Following 30 minutes, the pouch test article was removed from the buccal space and the animal allowed to recover from anesthesia. All pouches were retained following dosing. Each pouch was placed in individual conical tube with the animal ID and pouch identification.
- Group 1 was administered the nicotine benzoate control group.
- Group 2 was administered the disclosed composition comprising nicotine benzoate.
- Group 3 was administered the nicotine polacrilex control.
- Group 4 was administered the disclose composition comprising nicotine polacrilex.
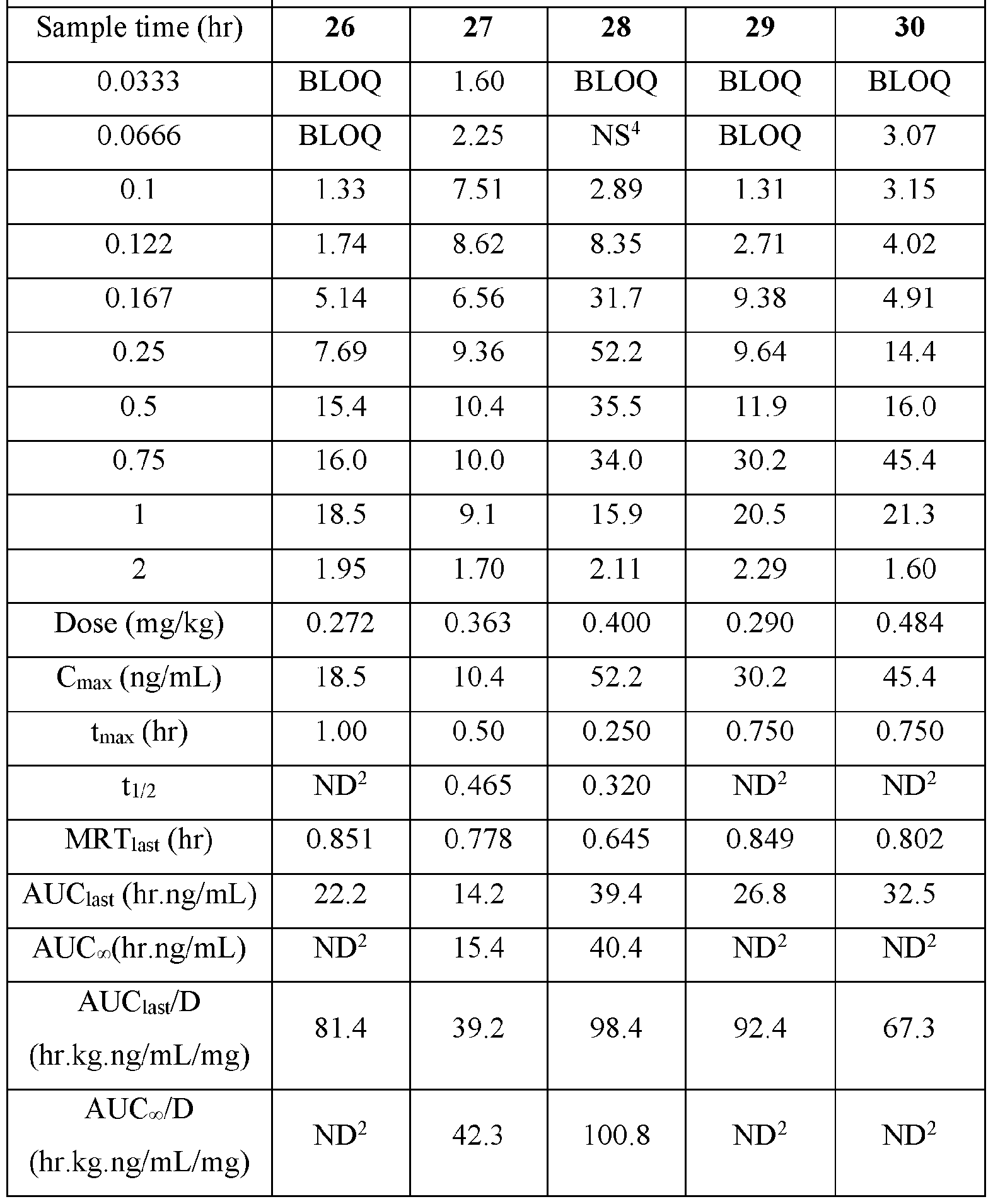
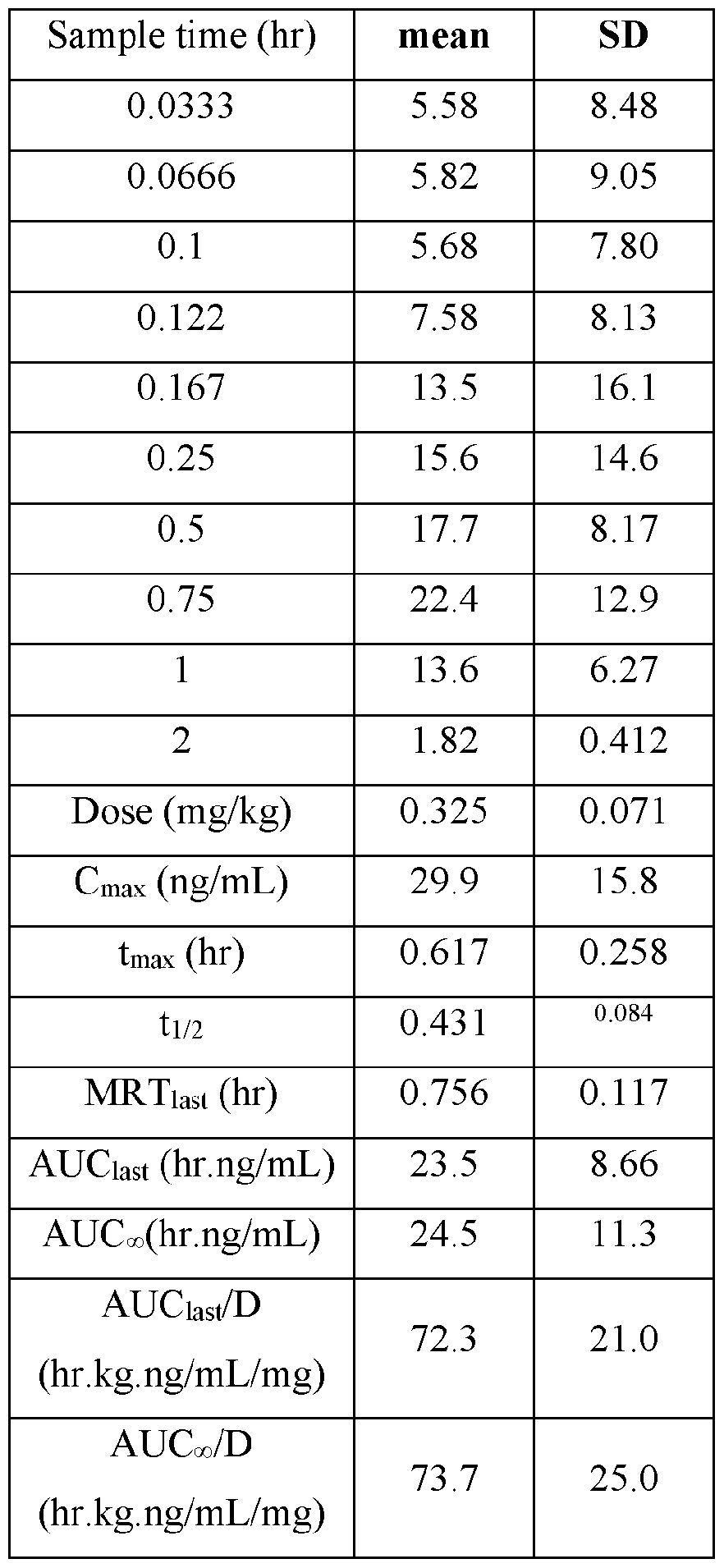
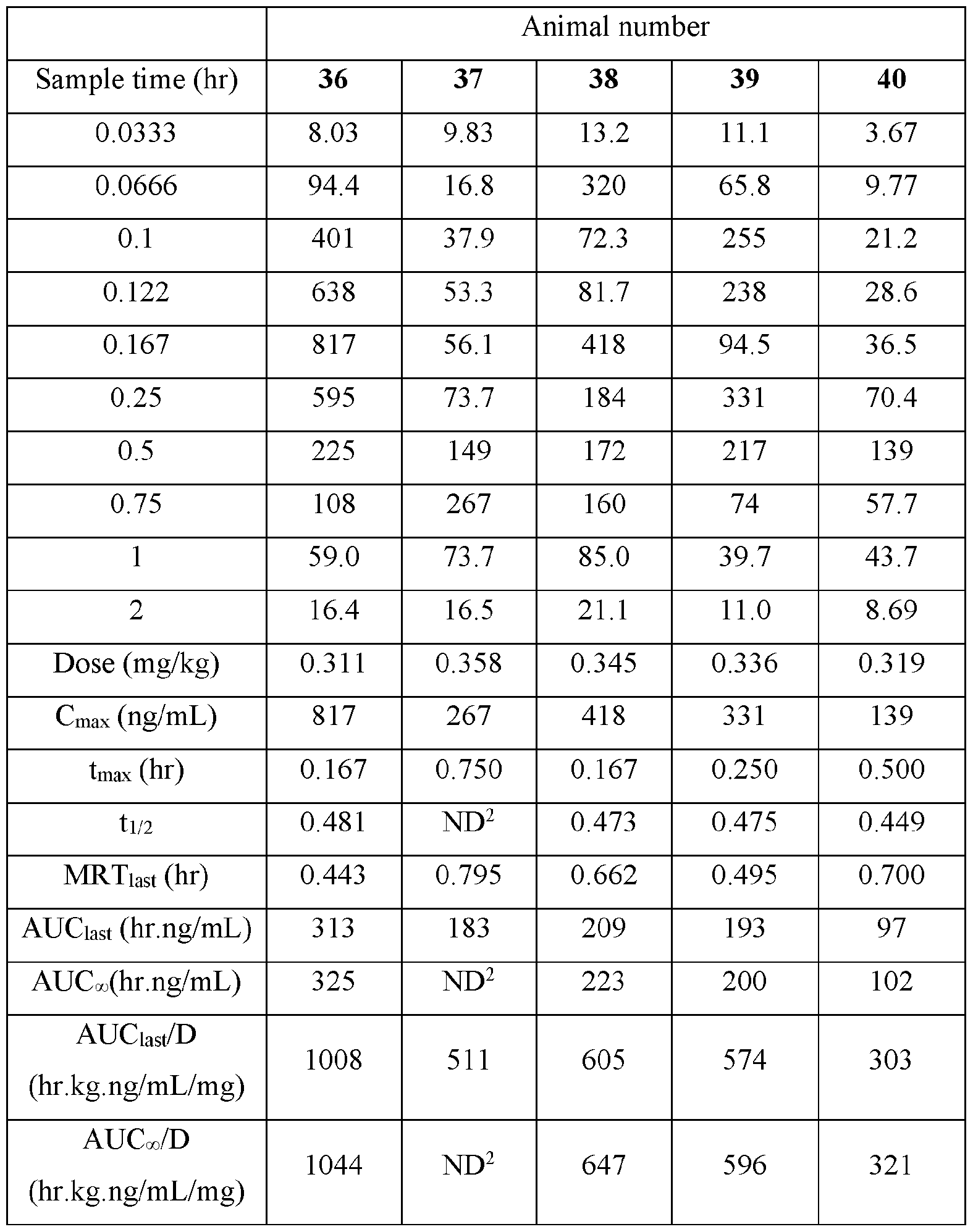
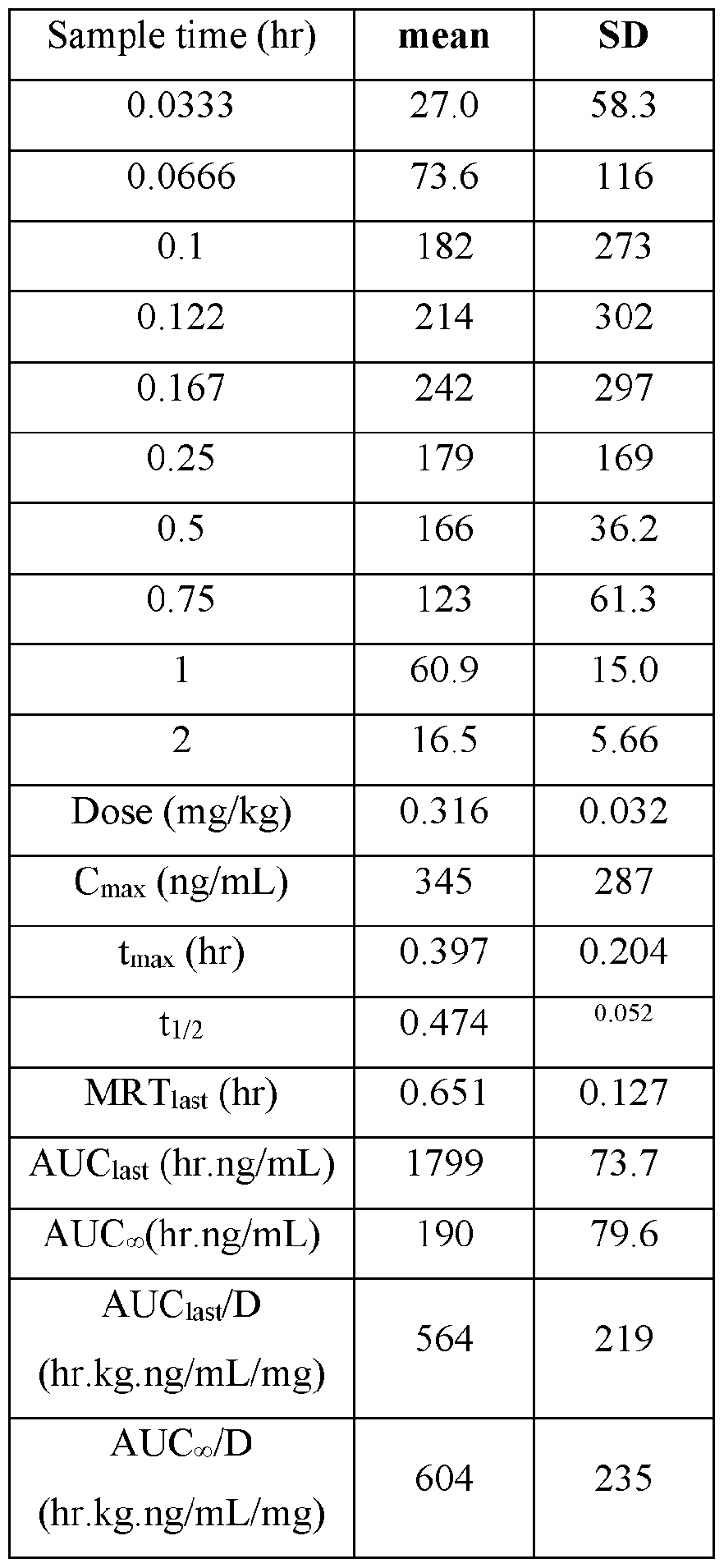
- the actual amounts based on results of potency testing was 3.12, 3.31, 3.48, and 3.79 mg per pouch, in Groups 1, 2,3, and 4, respectively TABLE V
- Each blood sample (approx. 2000 ⁇ L was collected from the jugular vein in a K2ETDA collection tube and gently inverted several time to mix. The samples were kept on ice until centrifugation at 4°C for 5 minutes at 3,000 x g. Approximately 1000 ⁇ L plasma was separated by centrifugation. The resulting plasma samples were stored at -80 C until bioanalysis was conducted.
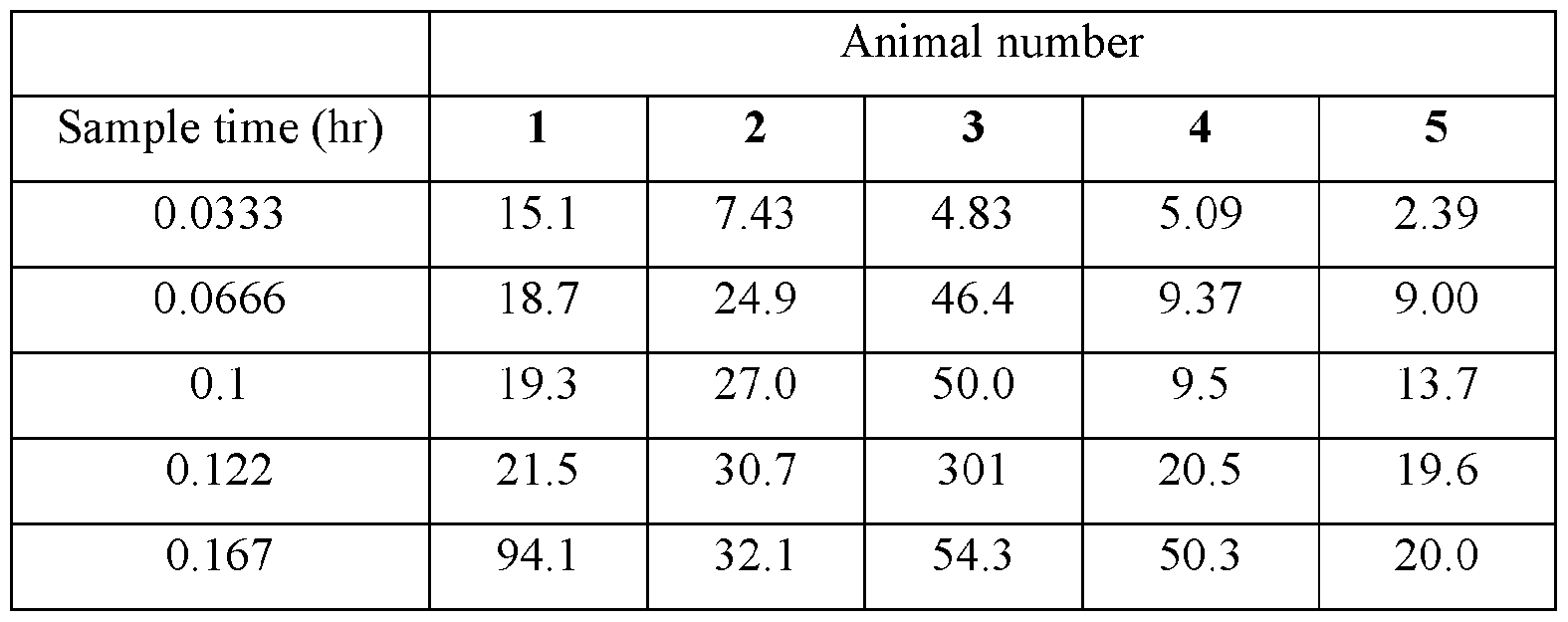
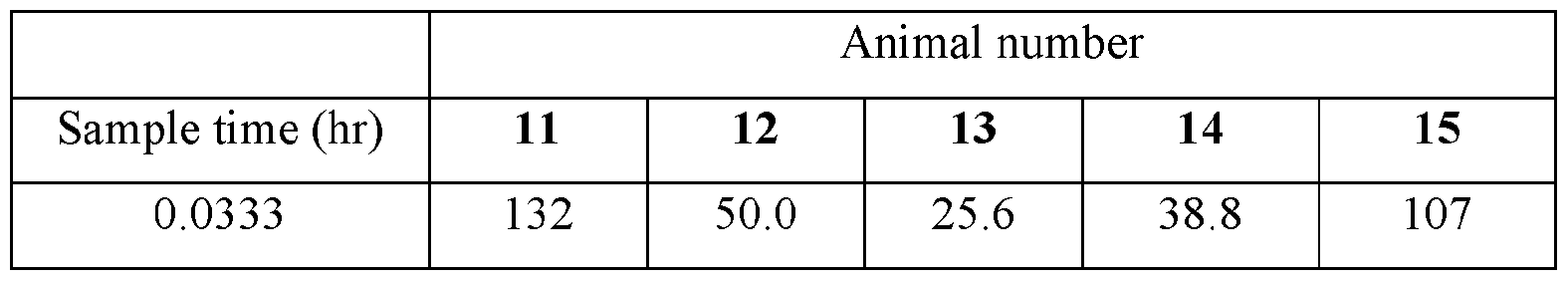
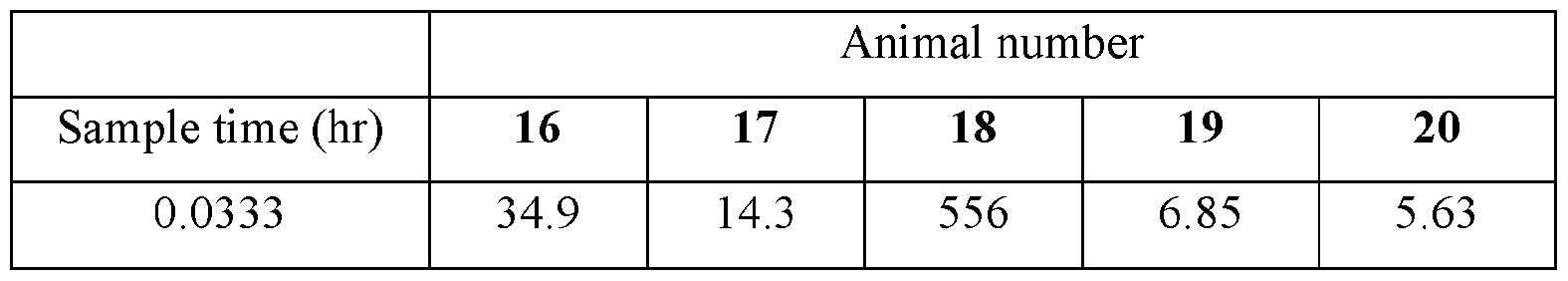
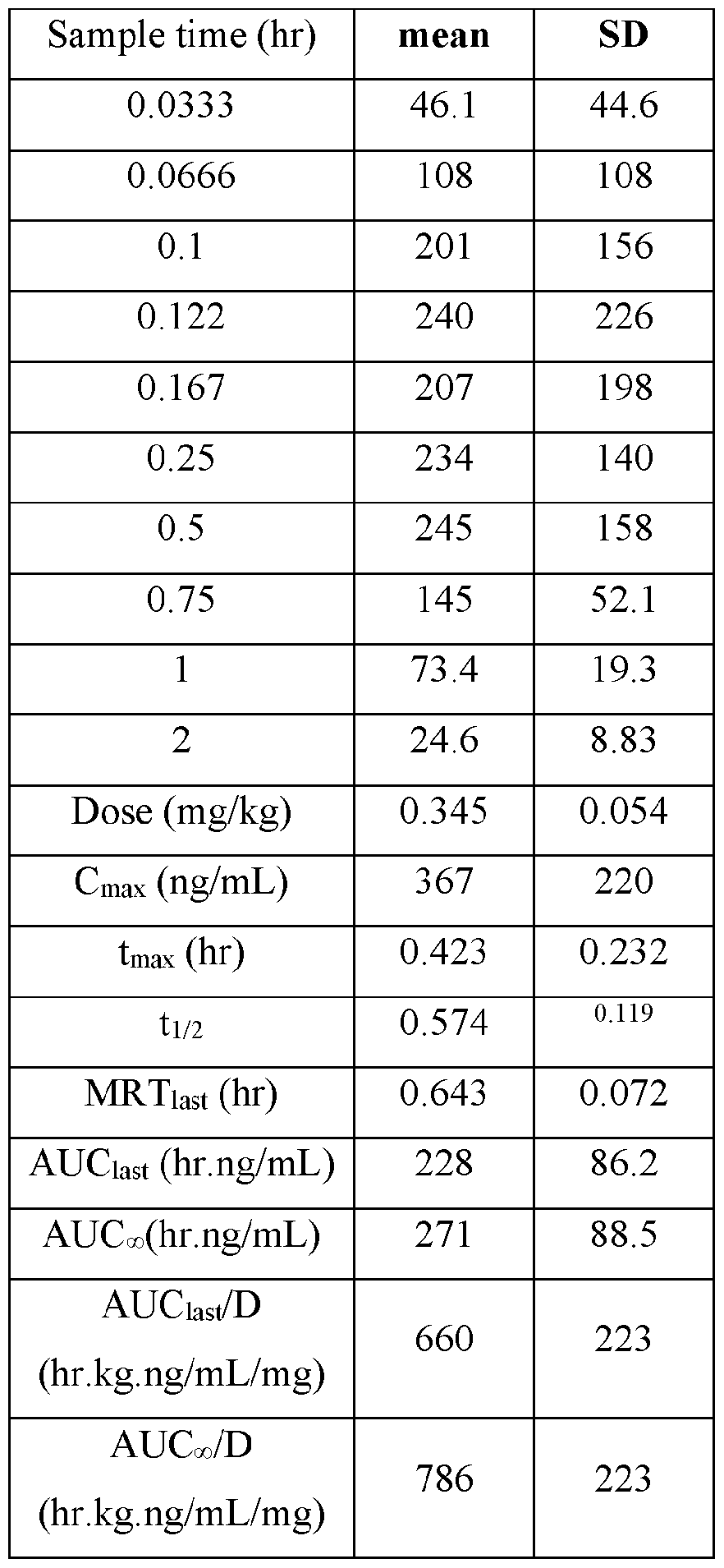
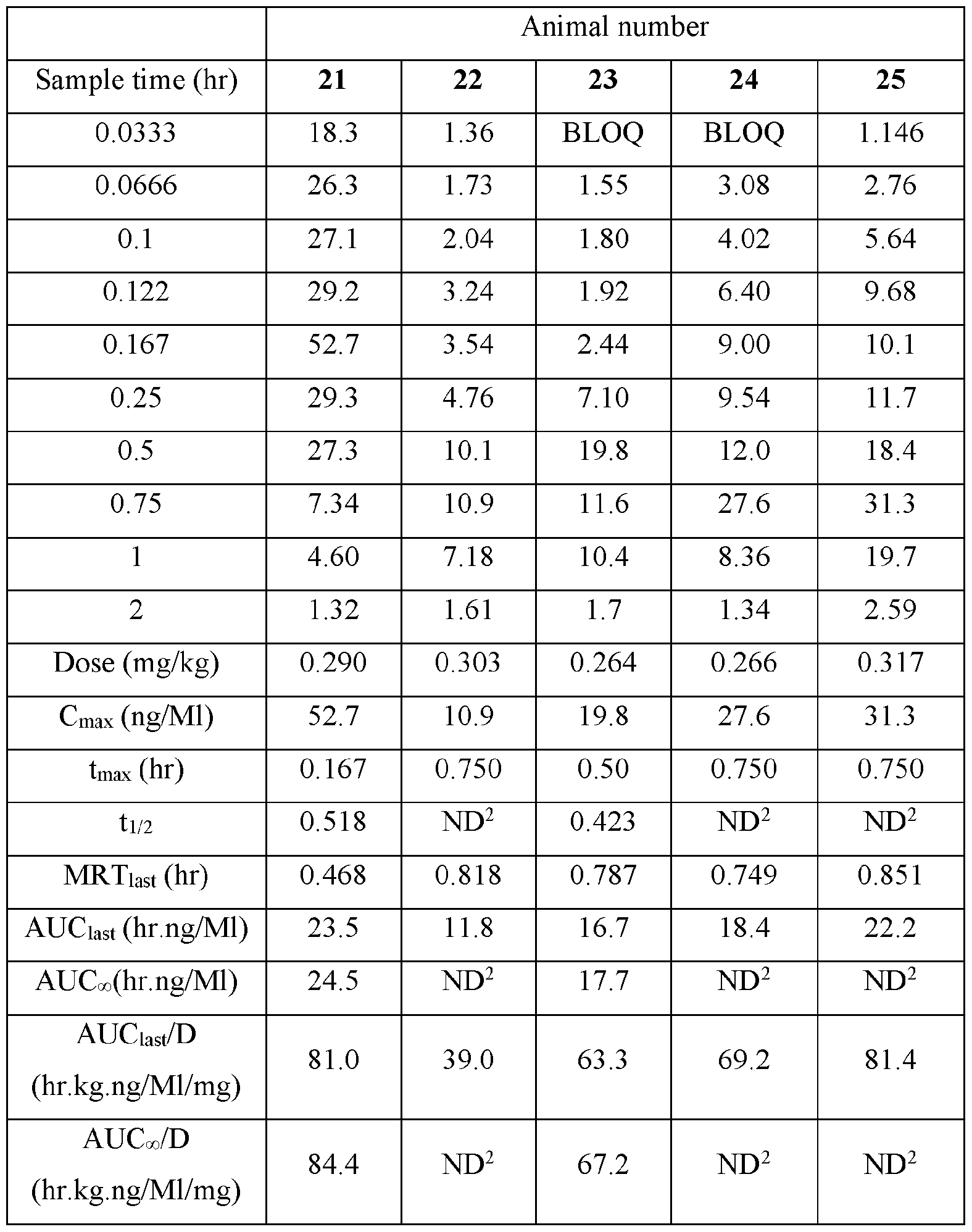
- Plasma samples were extracted by protein precipitation and analyzed using LC-MS/MS. Individual and Mean plasma concentrations and resulting pharmacokinetic parameters for nicotine are shown in Tables 4 - 7. All data are expressed as ng/mL of nicotine. Samples that were below the limit of quantification (1.0 ng/mL in plasma) were excluded from the calculation of mean values. Mean concentrations versus time data are plotted in Figures 1 - 8.
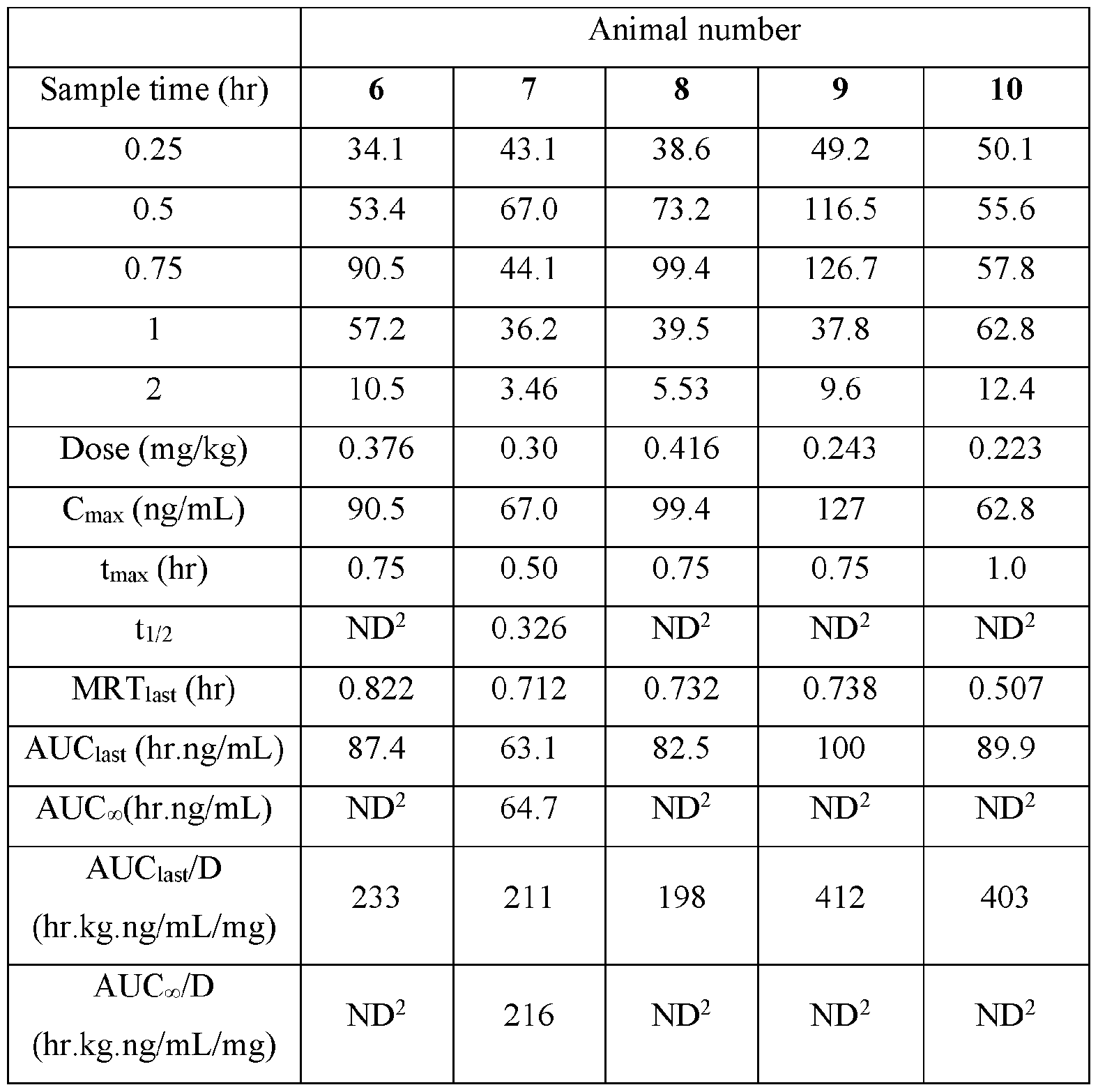
- Pharmacokinetic parameters were calculated from the time course of the plasma concentration. Pharmacokinetic parameters were determined with Phoenix WinNonlin (v8.0) software using a noncompartmental model. The maximum plasma concentration (Cmax) and the time to reach maximum plasma concentration (tmax) after dosing were observed from the data. The area under the time concentration curve (AUC) was calculated using the linear trapezoidal rule with calculation to the last quantifiable data point (AUC0-last), and with extrapolation to infinity ( AUC ⁇ ) if applicable. Plasma half-life (t 1/2) was calculated from 0.693/slope of the terminal elimination phase. Mean residence time, MRT, was calculated by dividing the area under the moment curve (AUMC) by the AUC. Any samples below the limit of quantitation (1.0 ng/mL plasma) were not used in the calculation of mean values
- AUC last /D (hr.kg. ng/mL/mg) and AUC ⁇ /D (hr.kg.ng/mL/mg) are dose normalized values. Not determined because the line defining the terminal elimination phase had an r 2 of ⁇ 0.85.
- AUC last /D (hr.kg ng/mL/mg) and AUC//D (hr.kg.ng/mL/mg) are dose normalized values.9.
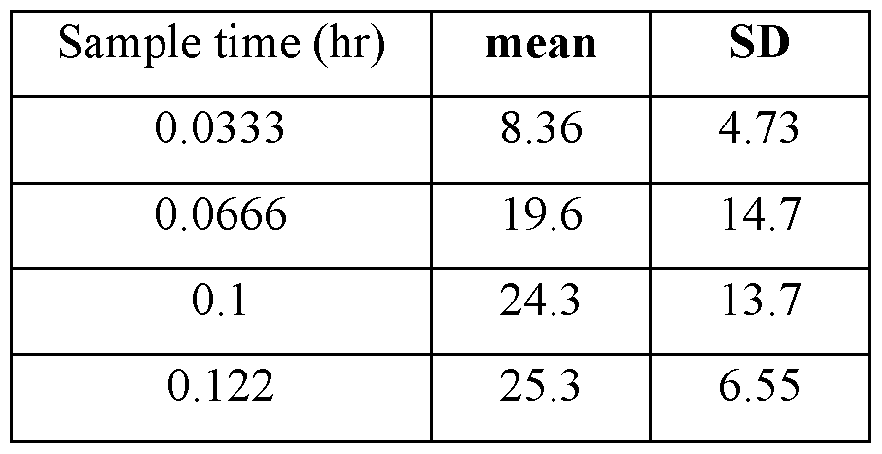
- TABLE VIII provides the mean and standard deviation for the results of Animals 1-10.
- Figure 1 is a plot of the individual plasma concentrations (ng/mL) for Nicotine versus time (hour) after buccal administration of the nicotine benzoate control composition disclosed in TABLE I in male Beagle dogs.
- Figure 1 is a plot of the mean plasma concentration (ng/mL) for Nicotine versus time (hour) after buccal administration of nicotine benzoate control composition disclosed in TABLE I in male Beagle dogs.
- AUC last /D (hr.kg. ng/mL/mg) and AUC ⁇ /D (hr.kg.ng/mL/mg) are dose normalized values.
- TABLE XI provides the mean and standard deviation for the results of Animals 11-21.
- Figure 3 depicts the individual plasma concentrations (ng/mL) for Nicotine versus time (hour) after buccal administration of the disclosed nicotine benzoate composition disclosed in Table II (4 mg) in male Beagle dogs.
- Figure 4 shows the mean plasma concentration (ng/mL) for Nicotine versus time (hour) after buccal administration of the disclosed compound in Table II (4 mg) in male Beagle dogs.
- AUC last /D (hr.kg.ng/Ml/mg) and AUC ⁇ /D (hr.kg.ng/Ml/mg) are dose normalized values. 2. Not determined because the line defining the terminal elimination phase had an r 2 of
- AUC last /D (hr.kg.ng/mL/mg) and AUC//D (hr.kg.ng/mL/mg) are dose normalized values.9.
- TABLE XIV provides the mean and standard deviation for the results of Animals 21- TABLE XIV
- Figure 5 shows the individual plasma poncentrations (ng/mL) for Nicotine versus time (hour) after buccal administration of the nicotine polacrilex control composition disclosed in TABLE III (4 mg) in male Beagle dogs (Group 3).
- Figure 6 displays the mean plasma concentration (ng/mL) for Nicotine versus time (hour) after buccal administration of the nicotine polacrilex control composition disclosed in TABLE III (4 mg) in Male Beagle dogs (Group 3) Pharmacokinetic Parameters and Plasma Concentrations (ng/mL) for Nicotine after Buccal Administration of pouches containing the nicotine polacrilex disclosed composition in TABLES XV and XVI (4 mg) in male Beagle dogs (Group 4) TABLE XV
- AUC last /D (hr.kg. ng/mL/mg) and AUC ⁇ /D (hr.kg.ng/mL/mg) are dose normalized values. . Not determined because the line defining the terminal elimination phase had an r 2 of ⁇ 0.85.
- TABLE XVII provides the mean and standard deviation for the results of Animals 31- 40.
- Figure 7 shows the individual plasma poncentrations (ng/mL) for Nicotine versus time (hour) after buccal administration of the nicotine polarcilex composition disclosed in TABLE VI (4 mg) in male Beagle dogs (Group 4).
- Figure 8 discloses the mean plasma concentration (ng/mL) for Nicotine versus time (hour) after buccal administration of the nicotine polarcrilex composition disclosed in TABLE VI (4 mg) in male Beagle dogs (Group 4).
- Standards were prepared in blank male Beagle dog plasma. Working solutions were prepared in 50:50 acetonitrile: water. Working solutions were then added to plasma to make calibration standards to final concentrations of 2000, 1000, 500, 250, 100, 50, 10, 5, 2 and 1 ng/mL. Standards were treated identically to the study samples.
- Plasma samples were manually extracted via precipitation with acetonitrile in a 96-well plate.
- MRM Multiple Reaction Monitoring
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Toxicology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Botany (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2023240953A AU2023240953B2 (en) | 2022-03-22 | 2023-03-20 | Compositions and methods for sublingual delivery of nicotine |
| EP23723799.5A EP4274437A4 (en) | 2022-03-22 | 2023-03-20 | Compositions and methods for sublingual delivery of nicotine |
| MX2023009660A MX2023009660A (en) | 2022-03-22 | 2023-03-20 | Compositions and methods for sublingual delivery of nicotine. |
| JP2023532287A JP7675819B2 (en) | 2022-03-22 | 2023-03-20 | Compositions and methods for sublingual delivery of nicotine |
| CA3196911A CA3196911C (en) | 2022-03-22 | 2023-03-20 | Compositions and methods for sublingual delivery of nicotine |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US17/700,646 US20230301341A1 (en) | 2022-03-22 | 2022-03-22 | Compositions and methods for sublingual delivery of nicotine |
| US17/700,628 US11700875B1 (en) | 2022-03-22 | 2022-03-22 | Compositions and methods for sublingual delivery of nicotine |
| US17/700,628 | 2022-03-22 | ||
| US17/700,646 | 2022-03-22 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2023183228A1 true WO2023183228A1 (en) | 2023-09-28 |
Family
ID=88102015
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2023/015627 Ceased WO2023183228A1 (en) | 2022-03-22 | 2023-03-20 | Compositions and methods for sublingual delivery of nicotine |
| PCT/US2023/015626 Ceased WO2023183227A1 (en) | 2022-03-22 | 2023-03-20 | Compositions and methods for sublingual delivery of nicotine |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2023/015626 Ceased WO2023183227A1 (en) | 2022-03-22 | 2023-03-20 | Compositions and methods for sublingual delivery of nicotine |
Country Status (1)
| Country | Link |
|---|---|
| WO (2) | WO2023183228A1 (en) |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20100170522A1 (en) * | 2008-12-19 | 2010-07-08 | U.S. Smokeless Tobacco Company | Tobacco Granules and Method of Producing Tobacco Granules |
| US20210204590A1 (en) * | 2019-12-09 | 2021-07-08 | Nicoventures Trading Limited | Pouched products |
| US20210345656A1 (en) * | 2017-12-08 | 2021-11-11 | Fertin Pharma A/S | High nicotine absorption |
| US20210378948A1 (en) * | 2020-06-08 | 2021-12-09 | Nicoventures Trading Limited | Effervescent oral composition |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018197454A1 (en) * | 2017-04-24 | 2018-11-01 | Swedish Match North Europe Ab | A flavoured moist oral pouched nicotine product comprising triglyceride |
| DK180339B1 (en) * | 2019-06-07 | 2020-12-18 | Ncp Nextgen As | Nicotine pouch composition and pouch comprising such |
-
2023
- 2023-03-20 WO PCT/US2023/015627 patent/WO2023183228A1/en not_active Ceased
- 2023-03-20 WO PCT/US2023/015626 patent/WO2023183227A1/en not_active Ceased
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20100170522A1 (en) * | 2008-12-19 | 2010-07-08 | U.S. Smokeless Tobacco Company | Tobacco Granules and Method of Producing Tobacco Granules |
| US20210345656A1 (en) * | 2017-12-08 | 2021-11-11 | Fertin Pharma A/S | High nicotine absorption |
| US20210204590A1 (en) * | 2019-12-09 | 2021-07-08 | Nicoventures Trading Limited | Pouched products |
| US20210378948A1 (en) * | 2020-06-08 | 2021-12-09 | Nicoventures Trading Limited | Effervescent oral composition |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2023183227A1 (en) | 2023-09-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3773494B1 (en) | Nicotine pouch composition and pouch comprising such | |
| DE69930964T2 (en) | COMPOSITIONS AND METHODS FOR MUCOSALE LEVY | |
| JP7223744B2 (en) | Starch-free soft chews for veterinary use | |
| CN115515639B (en) | New composition for oral or nasal use | |
| TWI259082B (en) | Stable ribavirin syrup formulations | |
| JP7759889B2 (en) | Novel compositions for oral or nasal use | |
| US20230301341A1 (en) | Compositions and methods for sublingual delivery of nicotine | |
| AU2002350081A1 (en) | Ribavirin syrup formulations | |
| AU2023240953B2 (en) | Compositions and methods for sublingual delivery of nicotine | |
| US11700875B1 (en) | Compositions and methods for sublingual delivery of nicotine | |
| WO2023183228A1 (en) | Compositions and methods for sublingual delivery of nicotine | |
| EP4271213A1 (en) | Compositions and methods for sublingual delivery of nicotine | |
| EP3380085A1 (en) | Oral preparations with omeprazole or pantoprazole | |
| US20250194662A1 (en) | Nicotine composition | |
| WO2022132018A1 (en) | A new powder composition | |
| HK1066719B (en) | Ribavirin syrup formulations |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 2023532287 Country of ref document: JP |
|
| ENP | Entry into the national phase |
Ref document number: 2023723799 Country of ref document: EP Effective date: 20230523 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2023/009660 Country of ref document: MX |
|
| ENP | Entry into the national phase |
Ref document number: 2023240953 Country of ref document: AU Date of ref document: 20230320 Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 2023723799 Country of ref document: EP |