WO2023144071A1 - Laundry composition - Google Patents
Laundry composition Download PDFInfo
- Publication number
- WO2023144071A1 WO2023144071A1 PCT/EP2023/051512 EP2023051512W WO2023144071A1 WO 2023144071 A1 WO2023144071 A1 WO 2023144071A1 EP 2023051512 W EP2023051512 W EP 2023051512W WO 2023144071 A1 WO2023144071 A1 WO 2023144071A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- composition
- alkyl
- group
- composition according
- surfactant
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/38—Products with no well-defined composition, e.g. natural products
- C11D3/386—Preparations containing enzymes, e.g. protease or amylase
- C11D3/38645—Preparations containing enzymes, e.g. protease or amylase containing cellulase
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/40—Dyes ; Pigments
- C11D3/42—Brightening agents ; Blueing agents
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D2111/00—Cleaning compositions characterised by the objects to be cleaned; Cleaning compositions characterised by non-standard cleaning or washing processes
- C11D2111/10—Objects to be cleaned
- C11D2111/12—Soft surfaces, e.g. textile
Definitions
- the present invention relates to a solid laundry composition, particularly to a solid laundry detergent composition having good biodegradability.
- a copolymer of acrylic and maleic acid is a well-known antiredeposition agent used in a solid laundry detergent composition.
- Copolymers of acrylic acid and maleic acid are also known to act as a structurant in a solid laundry detergent composition, particularly in a spray-dried detergent particle.
- a disadvantage of adding polycarboxylate polymer in the detergent composition is that these polymers are not easily biodegradable.
- Laundry detergent formulators may also add whitening agents such as fluorescers and shading dyes to the composition for improving the whiteness of the garments and reducing the yellowing over multiple washes, especially on white fabrics.
- whitening agents such as fluorescers and shading dyes
- a disadvantage of adding fluorescers and shading dyes in the detergent composition is that these whitening agents are not readily biodegradable.
- W02009/154934 A1 discloses a laundry composition having fluorescer and cellulase enzyme.
- JP2003 105389 A discloses a detergent composition for control of bacteria on fabric to prevent malodour and discloses a detergent composition which includes 0.5 wt.% acrylic maleic acid copolymer (AA/MA ratio 70:30) and 0.2 wt.% cellulase enzyme.
- WO 2011/109320 A1 discloses a solid laundry detergent composition for providing good cleaning performance even at low temperature conditions and discloses a granular dry laundry detergent composition which includes 1.7 wt.% STPP, 0.3 wt.% sodium polyacrylate/maleate copolymer (70:30 ratio), 0.6 wt.% carboxymethylcellulose, 0.5 wt.% HEDP, 5 wt.% sodium percarbonate and 53ppm cellulase.
- EP 2377914 A1 discloses a solid fabric treatment detergent composition for providing good cold water cleaning performance and discloses a composition which includes 0.6 wt.% STPP, 0.3 wt.% sodium polyacrylate/maleate copolymer (70:30 ratio), 0.5 wt.% hydrophobically modified carboxymethylcellulose, 0.3 wt.% HEDP, 7 wt.% sodium percarbonate and 53ppm cellulase.
- WO 99/13040 A1 discloses a detergent composition for addressing the problem of bleach instability of sequestrants and providing improved encrustation sequestering and improved bleach performance.
- This document discloses a composition which includes 0.5 wt.% MA/AA (copolymer with 1 :4 maleic/acrylic acid), 1 wt.% sodium perborate bleach and 0.2 wt.% cellulase.
- JP 2007/070613 A discloses a granular laundry detergent composition which provides excellent softness and prevents redeposition to the fibre products while washing.
- This document discloses a granular laundry composition which includes 0.1 wt.% cellulase, 0.5 wt.% CMC and 0.5 wt.% acrylic maleic acid copolymer.
- the present inventors have surprisingly found that combining cellulase enzyme along with specific optical brightener provides for significant improvement in whiteness benefits.
- the composition was also found to provide good biodegradability. It was surprisingly found that a combination of cellulase enzyme and specific optical brightener provides for improved whiteness benefits even at low levels of the specific optical brightener in the solid laundry composition.
- a solid laundry composition comprising:
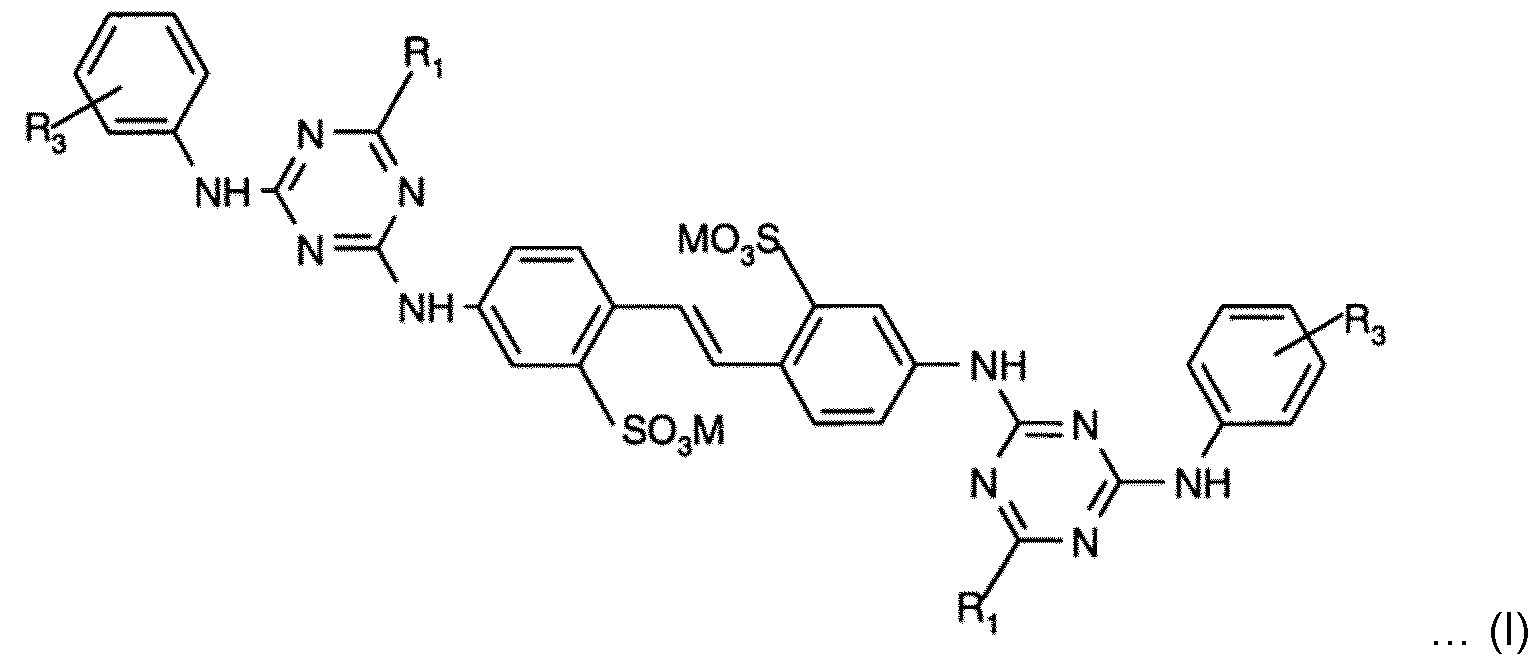
- an optical brightener represented by the general formula (I) wherein: Ri is same or different and is selected from the group consisting of -NH2, -NH(Ci to C4 alkyl), -N(Ci to C4 alkyl)2, -NH(C2 to C4 hydroxyalkyl), -N(C2 to C4 hydroxyalkyl)2, and - N(Ci to C4 alkyl)(C2 to C4 hydroxyalkyl);
- R3 is independently selected from the group consisting of hydrogen (-H), Ci to C4 alkyl group, Ci to C4 hydroxyalkyl group;
- M is independently selected from the group consisting of hydrogen, alkali metal atom, ammonium, and a cation formed from an amine.
- a method for laundering a textile surface with the solid laundry composition according to the first aspect of the present invention comprising the steps of: i) preparing an aqueous wash liquor by contacting the solid laundry detergent composition according to the first aspect with a liquid; ii) soaking said textile surface in the wash liquor for a predetermined period of time; and, iii) optionally rinsing the textile surface.
- a cellulase enzyme and an optical brightener of general formula (I) according to first aspect in a solid laundry composition to provide improved cleaning performance.
- a solid laundry composition having a cellulase enzyme and an optical brightener.
- solid laundry composition including a cellulase enzyme.
- cellulase refers to an enzyme that hydrolzes a cellulosic material. Such enzymes include those selected from endoglucanase (e.g., EC 3.2.1.4), cellobiohydrolase, betaglucosidase, or combinations thereof.
- the cellulase may for example be a mono-component or a mixture of endo-1,4-beta-glucanase also referred to as endoglucanase.
- Suitable cellulases include those from bacterial origin, fungal origin, or a combination thereof. Chemically modified or protein engineered mutants are also contemplated. Suitable cellulases include those from the genera Bacillus, Pseudomonas, Humicola, Myceliophthora, Fusarium, Thielavia, Trichoderma, and Acremonium. Exemplary cellulases include a fungal cellulase from Humicola insolens (US 4,435,307) or from Trichoderma, e.g., T. reesei or T. viride.
- Suitable cellulases are from Thielavia e.g., Thielavia terrestris as described in WO 96/29397 or the fungal cellulases produced from Myceliophthora thermophila and Fusarium oxysporum disclosed in US 5,648,263, US 5,691,178, US 5,776,757, WO 89/09259 and WO 91/17244. Also relevant are cellulases from Bacillus as described in WO 02/099091 and JP 2000210081. Suitable cellulases are alkaline or neutral cellulases, preferably those having colour care benefits.
- cellulases are described in EP 0495257, EP 0 531 372, WO 96/11262, WO 96/29397, WO 98/08940.
- Other examples are cellulase variants such as those described in WO 94/07998, EP 05/531315, US 5,457,046, US 5,686,593, US 5,763,254, WO 95/24471 , WO 98/12307.
- cellulases are endo-beta-1 ,4-glucanase enzyme having a sequence of at least 97% identity to the amino acid sequence of position 1 to position 773 of SEQ ID NO:2 of WO 2002/099091 or a family 44 xyloglucanase, which a xyloglucanase enzyme having a sequence of at least 60% identity to positions 40-559 of SEQ ID NO. 2 of WO 2001/062903.
- cellulases include Carezyme®, Carezyme® Premium, Celluzyme®, Celluclean®, Celluclast®, Endolase®, Renozyme®, Whitezyme®, Celluclean®, Classic, Cellusoft® (Novozymes A/S), Puradax® , Puradax HA, and Puradax EG (available from Genencor International Inc.) and KAC-500(B)TM (Kao Corporation).
- the cellulase is obtained from Humicola in particular Humicola insolens. In another embodiment, the cellulase is obtained from Bacillusin, in particular Bacillus akibai. In another embodiment, the cellulase is obtained from Paenibacillus, in particular Paenibacillus polmyxa. In yet another embodiment, the cellulase is obtained from Melanocarpus, in particular Melanocarpus albomyces.
- cellulase comprises the amino acid sequence of SEQ ID NO: 10, SEQ ID NO: 11, SEQ ID NO: 12 or SEQ ID NO: 13. More preferably the cellulase comprises an amino acid sequence having at least 60%, e.g., at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% sequence identity to the polypeptide of SEQ ID NO 10, SEQ ID NO: 10, SEQ ID NO: 11, SEQ ID NO: 12 or SEQ ID NO: 13.
- the cellulase comprises amino acid sequence which differ by up to 10 amino acids, e.g., 20 1 , 2, 3, 4, 5, 6, 7, 8, 9, or 10, from the cellulase comprising amino acid sequence of SEQ ID NO: 10, SEQ ID NO: 10, SEQ ID NO: 11 , SEQ ID NO: 12 or SEQ ID NO: 13.
- the cellulase enzyme is preferably formulated in a solid form, preferably a granular form.
- the granular form may be produced as disclosed in US 4,106,991 and US 4,661 ,452, and may optionally be coated by methods known in the art.
- waxy coating materials are poly(ethylene oxide) products (polyethyleneglycol, PEG) with mean molar weights of 1000 to 20000, ethoxylated nonylphenols having from 16 to 50 ethylene oxide units; ethoxylated fatty alcohols in which the alcohol contains from 12 to 20 carbon atoms and in which there are 15 to 80 ethylene oxide units; fatty alcohols; fatty acids; and mono- and di- and triglycerides of fatty acids.
- the cellulase may be formulated as a granule, preferably a co-granule which combines one or more additional enzymes.
- additional preferred enzymes includes proteases, amylases, lipases, cellulases, and/or nucleases (e.g., DNase, RNase).
- Each enzyme will then be present in more granules securing a more uniform distribution of enzymes in the detergent. This also reduces the physical segregation of different enzymes due to different particle sizes.
- Methods for producing multi-enzyme co-granulate for the detergent industry are known to a person skilled in the art.
- Another example of cellulase enzymes in the form of co-granulates are disclosed in WO 2013/188331 A1.
- the enzyme in the form of a granule having a core comprising cellulase enzyme and surrounded by one or more coating layers.
- the coating layers provide improved storage stability, reduce dust formation, or improve the color or appearance of the granule.
- the coating layers may include a salt, polyethylene glycol (PEG), methyl hydroxy propyl cellulose (MHPC), and polyvinyl alcohol (PVA).
- the cellulase enzyme may also be formulated in an encapsulate form.
- the cellulase may be encapsulated in a matrix, preferably a water-soluble or water dispersible matrix (e.g., water- soluble polymer particles), for example as described in WO 2016/023685.
- a water-soluble polymeric matrix is a matrix composition comprising polyvinyl alcohol.
- the enzyme may also be encapsulated in core-shell microcapsules, for example as described in WO 2015/144784.
- core-shell capsules can be prepared using a number of technologies known in the art, e.g., by interfacial polymerization using either a water-in-oil or an oil-in-water emulsion, where polymers are crosslinked at the surface of the droplets in the emulsion (the interface between water and oil), thus forming a wall/membrane around each droplet/capsule.
- the cellulase enzyme may also be present as a multienzyme co-granule which in addition to cellulase includes one or more additional preferred enzyme selected from the group consisting of lipases, per hydrolases, peroxidases, laccases, first-wash lipases, proteases, mannanase, reductases, oxidases, nuclease, DNase, amylase, or mixtures thereof.
- additional preferred enzyme selected from the group consisting of lipases, per hydrolases, peroxidases, laccases, first-wash lipases, proteases, mannanase, reductases, oxidases, nuclease, DNase, amylase, or mixtures thereof.
- the levels of cellulase enzyme in the composition ranges from 0.0001 wt.% to 0.5 wt.%.
- the amount of the cellulase present in the composition is at least 0.001 wt.%, still preferably at least 0.01 wt.%, still preferably at least 0.02 wt.%, most preferably at least 0.03 wt.%, but typically not more than 0.3 wt.%, still preferably not more than 0.25 wt.%, most preferably not more than 0.2 wt.%.
- Levels of enzyme present in the composition preferably relate to the level of enzyme as pure protein.
- a solid detergent composition which includes an optical brightener.
- Disclosed solid laundry composition includes an optical brightener which is preferably present in an amount ranging from 0.01 wt.% to 0.4 wt.%, preferably from 0.05 wt.% to 0.3 wt.%, still preferably from 0.05 wt.% to 0.2 wt.% in the composition.
- Optical brighteners are compounds which absorb ultraviolet light and reemit the energy in the form of visible light.
- the optical brighteners useful herein absorb light in the ultraviolet and violet region between a wavelength of 300nm and 400nm of the electromagnetic spectrum and re-emit light in the wavelength of 420nm and 450 nm.
- the light is generally emitted in the visible spectrum, preferably blue or green light.
- Optical brighteners may also be referred to as fluorescent whitening agents (FWAs), fluorescent brightening agents (FBAs), fluorescers, fluorescent agents, optical brightening agents (OBAs) and fluorescent dyes.
- Disclosed solid laundry composition includes an optical brightener represented by the general formula (I) where: Ri is same or different and is selected from the group consisting of -NH2, -NH(Ci to C4 alkyl), - N(Ci to C4 alkyl)2, -NH(C2 to C4 hydroxyalkyl), -N(C2 to C4 hydroxyalkyl)2, and -N(Ci to C4 alkyl)(C2 to C4 hydroxyalkyl);
- Ri is same or different and is selected from the group consisting of -NH2, -NH(Ci to C4 alkyl), - N(Ci to C4 alkyl)2, -NH(C2 to C4 hydroxyalkyl), -N(C2 to C4 hydroxyalkyl)2, and -N(Ci to C4 alkyl)(C2 to C4 hydroxyalkyl);
- R3 is independently selected from the group consisting of hydrogen (-H), Ci to C4 alkyl group and Ci to C4 hydroxyalkyl group;
- M is independently selected from the group consisting of hydrogen, alkali metal atom, ammonium, or a cation formed from an amine.
- R1 is selected from the group consisting of -NH2, -NH(Ci to C4 alkyl), -N(Ci to C4 alkyl)2, -NH(C2 to C4 hydroxyalkyl), -N(C2 to C4 hydroxyalkyl)2, and -N(Ci to C4 alkyl)(C2 to C4 hydroxyalkyl).
- the alkyl radical may be preferably branched or unbranched.
- Non-limiting examples of the alkyl radical includes methyl, ethyl, n-propyl, isopropyl, isobutyl.
- Non-limiting examples of the hydroxyalkyl radical includes hydroxymethyl, hydroxyethyl, hydroxypropyl and hydroxybutyl.
- each R1 group is independently selected from the group consisting of -N-mono (l-hydroxyethyl)amine, -N-mono (2-hydroxyethyl)amine, mono-(2-hydroxyethyl)-N-methylamino, di-(2-hydroxyethyl)amino, di-(2-hydroxypropyl)amino and N-(2-hydroxyethyl)-N-methylamino.
- the R1 are same or different.
- R3 is independently selected from the group consisting of hydrogen (H), Ci to C4 alkyl group, and Ci to C4 hydroxyalkyl group.
- the Ci to C4 alkyl group is a primary, secondary, or a tertiary alkyl group. More preferably the Ci to C4 alkyl group is a primary alkyl group.
- the Ci to C4 hydroxyalkyl group is a primary, secondary, or tertiary alkyl group.
- the alkyl radical may be preferably branched or unbranched.
- Non-limiting examples of the alkyl radical includes methyl, ethyl, n-propyl, isopropyl, isobutyl.
- Non-limiting examples of the hydroxyalkyl radical includes hydroxymethyl, hydroxyethyl, hydroxypropyl and hydroxybutyl.
- Preferably the R 3 are either same or different.
- M is independently selected from the group consisting of hydrogen, alkali metal atom, ammonium, or a cation formed from an amine, preferably selected from the group consisting of Na + , and K + . More preferably M represents sodium.
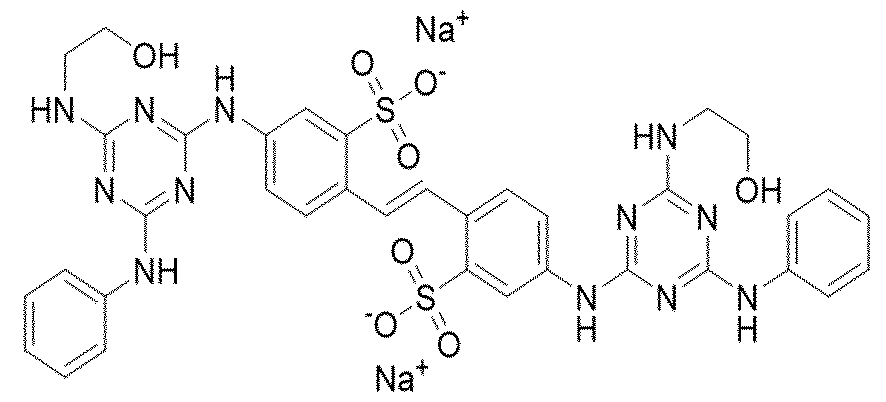
- optical brightener of the general formula (I) includes
- optical brightener of the general formula (I) includes Kayaphor B, Kayaphor AB, Mikephor BP, Lemonite RSJ (Supplier Khyati Chemical Pvt. Ltd), Megawhite DK (Supplier Meghmani Organics Ltd) and Deepwhite MDM powder (Supplier Deepak nitrite).
- the molecular weight of Disodium 4,4'-bis[(4-anilino-6-hydroxyethylamino-1 ,3,5- triazin-2-yl)amino]stilbene-2,2'-disulphonate is 872.84 and the molecular formula is Cse ⁇ N ⁇ I ⁇ C ⁇ .
- the composition of the present invention includes 0 wt.% Tinopal CBSX.
- the composition preferably includes 0 wt.% DASCC type optical brightener, 0 wt.% anilino- substituted bistriazinyl derivatives of 4,4'-diaminostilben-2,2'-disulphonic acid based on cyanuric chloride (CC), particularly 0 wt.% disodium 4,4'-bis[(4-anilino-6-morpholino-1,3,5-triazin-2- yl)amino] stilbene-2,2'-disulphonate.
- CC cyanuric chloride
- the fluorescent agents are commercially available and used in their alkali metal salt form, preferably the sodium salts.
- the optical brighteners may be added in particulate form or as a premix with a suitable solvent, for example nonionic surfactant, monoethanolamine, propane diol.
- the solid laundry composition of the present invention preferably includes from 0.01 wt.% to 0.4 wt.% optical brightener of the general formula (I).
- the amount of the optical brightener of general formula (I) in the composition ranges from 0.05 wt.% to 0.3 wt.%, still preferably from 0.05 wt.% to 0.2 wt.% in the composition.
- the amount of the optical brightener in the composition is at least 0.02 wt%, still preferably at least 0.025 wt%, still preferably at least 0.03 wt%, most preferably at least 0.05 wt%, but typically not more than 0.4wt.%, still preferably not more than 0.35 wt.%, still preferably not more than 0.3 wt.%, most preferably not more than 0. wt.%.
- the solid laundry composition according to the first aspect of the present invention may include a shading dye, preferably in an amount ranging 0.0005 wt.% to 0.005 wt.%, still preferably from 0.0005 wt.% to 0.004 wt.%, still more preferably from 0.0005 wt.% to 0.002 wt.%. More preferably the solid laundry composition according to the first aspect of the present invention is substantially free of the shading dye.
- Shading dyes deposit to fabric during the wash or rinse step of the washing process providing a visible hue to the fabric.
- the shading dyes used in the present invention are preferably blue or violet.
- the shading dye gives a blue or violet colour to a white cloth with a hue angle of 240 to 345, more preferably 260 to 320, most preferably 270 to 300.
- the white cloth used is bleached non-mercerised woven cotton sheeting.
- the shading dye preferably has a calculated logP (octanol-water partition coefficient) of between 0.5 and 3 more preferably between 1 .0 and 2.5.
- the shading dye preferably has a peak absorption wavelength of from 550nm to 650nm, preferably 570nm to 630nm.
- a combination of dyes which together have the visual effect on the human eye as a single dye having a peak absorption wavelength of from 550nm to 650nm, preferably 570nm to 630nm may also be suitable. This may be provided for example by mixing a red and a green-blue dye to yield a blue or a violent shade.
- Shading dyes are coloured organic molecules which are soluble in aqueous media that contain surfactants. Dyes are described in 'Industrial Dyes', Wiley VCH 2002, K. Hunger (editor).
- Dyes are listed in the Color Index International published by Society of Dyers and Colourists and the American Association of Textile Chemists and Colorists. Many examples of shading dyes are found in the classes of basic, solvent, acid, direct and disperse dyes. Dyes are preferably selected from the classes of basic, acid and direct dyes.
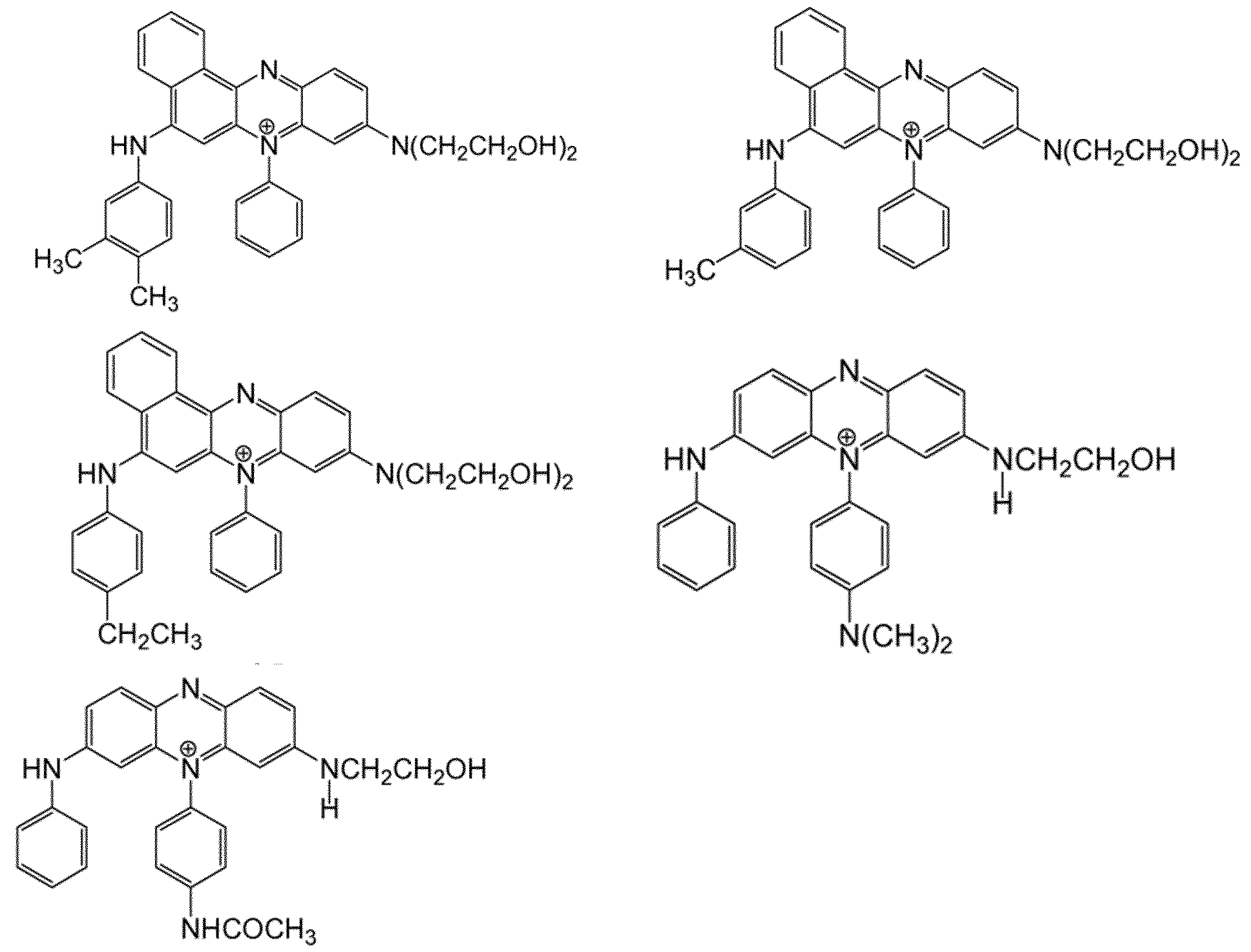
- the dye chromophore is preferably selected from the group comprising: mono- azo, triphenylmethane, naptholactam, azine and anthraquinone. Most preferably mono-azo, azine and anthraquinone.
- Preferred shading dyes are:
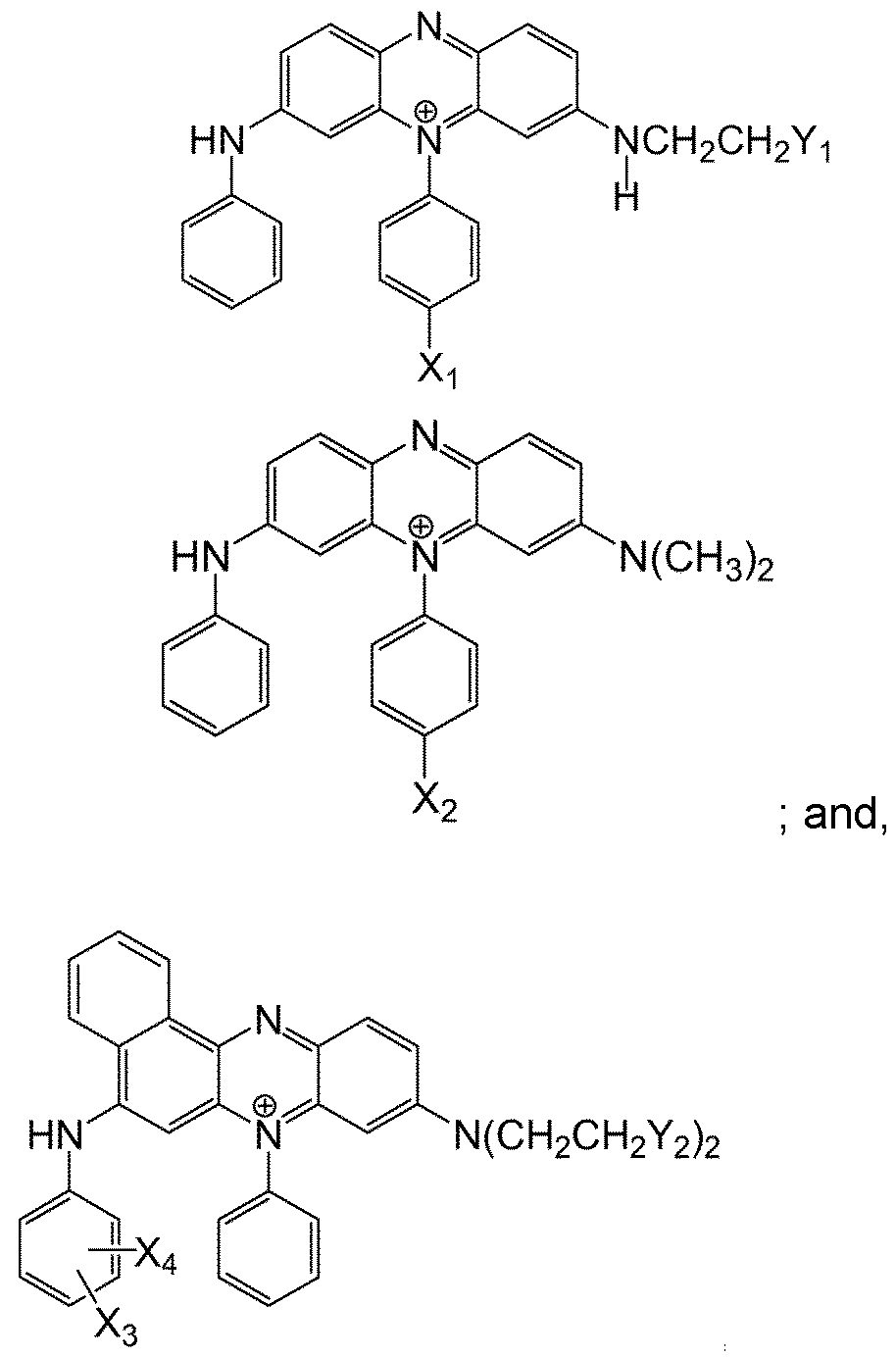
- a cationic azine dye preferably of the following form blue or violet cationic phenazine dye which is not covalently bound to a negatively charged substituent.
- a cationic azine dye as described in WO 2011/042372 A1 ,
- the cationic phenazine may be selected from: wherein:
- Xi is selected from: -F; -N(CH 3 ) 2 ; -N(CH 2 CH 3 ) 2 ; -N(H)COCH 3 ; and, - N(H)COCH 2 CH 3 ; preferably selected from -N(CH 3 ) 2 and -N(H)COCH 3
- X 2 is selected from: -F; -N(CH 3 ) 2 ; -N(CH 2 CH 3 ) 2 ; -N(H)COCH 3 ; and, - N(H)COCH 2 CH 3 ; preferably selected from -F, -N(CH 3 ) 2 and -N(H)COCH 3 .
- Xs is selected from: -H; -F; -CH3; -C2H5; -OCH3; and, -OC2 H5; preferably selected from -H; -F; - CH3 and preferably X3 is in the 4 position
- X4 is selected from: -H; -CH3; -C2H5 ; -OCH3 ; and, -OC2H5 ; preferably X4 is H.
- Y1 is selected from: -OH; -OCH2CH2OH; -CH(OH)CH2OH; and, preferably Y1 is -OH
- Y 2 is selected from: -OH; -OCH2CH2OH; -CH(OH)CH 2 OH; -OC(O)CH 3 ; and, C(O)OCH 3 .
- Y2 is -OH.
- the Y2 substituents are the same.
- the phenazine dye have a counter ion An- by virtue of being positively charged.
- the An- may be widely varied and preferably be an anion such as RCOO; BPI , CLOT, BF4; PFe' .RSOT, SO4 2 ; NOT, F; Cl; Br, or I' with R being hydrogen, optionally substituted alkyl or optionally substituted aryl.
- An- is selected from CH3SO3; CH3CO2; BF4; Cl; F; Br, and I'.
- these includes:
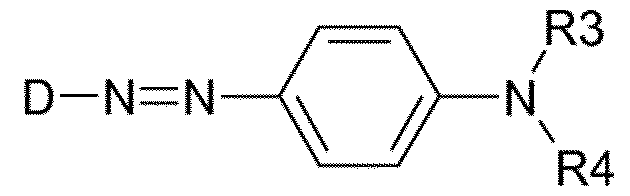
- cationic azo dyes preferably cationic mono-azo dyes.
- the cationic azo dye preferably bear the cationic groups on a chain.
- the dye may also a bear an anionic group.
- hydrophobic dyes as follows: (i) solvent violet 13 and disperse violet 27 and an anthraquinone structure of the following anthraquinone structure (I): wherein Ri, R4, R5, and Rs are independently selected from the groups consisting of -H, -OH, - NH2, NHCOCH3 and -NO2, such that a maximum of only one -NO2 group and a maximum of two -H are present as R1, R4, Rs, and Rs substituents; and R2, R3, Re, and R? is selected from -H, F, Br, Cl or -NO2, and - Oaryl.
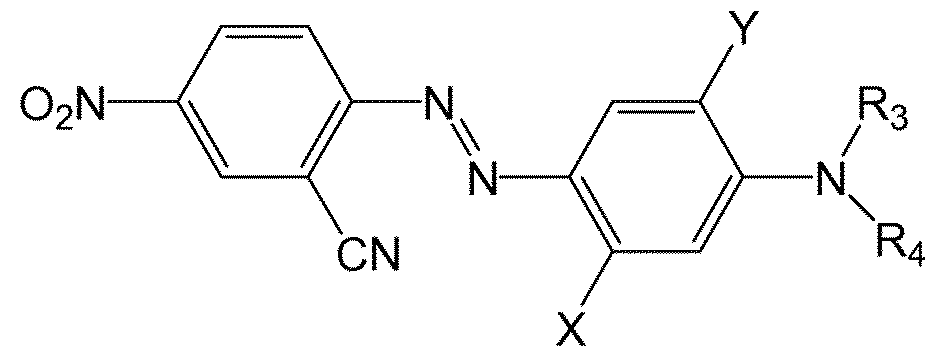
- mono-azo dye selected from a compound of the following formula: wherein R3 and R4 are optionally substituted C2 to C12 alkyl chains having optionally therein ether (-O-) or ester links, the chain being optionally substituted with -Cl, -Br, -CN, -NO2, and - SO2CH3; and, D denotes an aromatic or hetroaromatic group.
- the aromatic rings may be further substituted by preferably -Cl, -Br, -CN, CH3, C2H5, -NO2, -SO2CH3 and -NHCOR and R is selected form CH3, C2H5, and CH2CI.
- azo dyes are of the form:
- X and Y are selected from H, CH 3 , C 2 H 5 , -Cl, -Br, -CN, -NO 2 , -SO 2 CH 3 and -NHCOR and R is selected form CH 3 , C 2 Hs , and CH 2 CI.
- X is NHCOCH 3 or NHCOCH 2 CI.
- the dyes are reactive blue 19, reactive blue 163, reactive blue 182, reactive blue 96.
- Shading of white garments may be done with any colour depending on consumer preference.
- Blue and Violet are particularly preferred shades and consequently preferred dyes, or mixtures of dyes are ones that give a blue or violet shade on white fabrics.
- Suitable solvent and disperse dyes are available. However detailed toxicological studies have shown that a number of such dyes are possible carcinogens, for example disperse blue 1. Such dyes are not preferred. Most preferably the dye used in the invention is disperse violet 28.
- shading dyes are described in W008017570 A1 , WO11098356 A1 , WO09141172 A1 , WO09141173 A1 , and incorporated herein by reference. Shading dyes are discussed in W02005/003274, W02006/032327(Uni lever), WO 2006/032397(Unilever), W02006/045275(Unilever), WO 2006/027086(Unilever), WO 2008/017570(Unilever), WO 2008/141880 (Unilever), W02009/132870(Unilever), WO 2009/141 173 (Unilever), WO 2010/099997(Unilever), WO 2010/102861 (Unilever), WO 2010/148624(Unilever), W02008/087497 (P&G), WO2011/011799 (P&G), W02012/054820 (P&G), WO2013/142495 (P&G) and W02013/151970 (
- composition according to the present invention may further optionally include dye-clay conjugates, and other organic and inorganic pigments.
- a solid laundry composition having a mixture of 0.01 wt.% to 0.4 wt.% optical brightener according to the present invention and 0 to 0.005 wt.% shading dye.
- the composition may include a substituted polysaccharide.
- the substituted polysaccharide has a functional group present on the polysaccharide backbone, said functional group is selected from the group consisting of alkyl, carboxyalkyl, carboxylic acid, alkoxy or salts thereof.
- Polysaccharides are chemically modified at one or more of the available hydroxyl groups of the monomeric sugar units to introduce functional group suitably by oxidation, grafting, esterification and etherification in attempts to balance detergent performance and biodegradation.
- the polysaccharide is selected from the group consisting of cellulose, inulin, starch, dextrins and mixtures thereof.
- dextrins includes the group consisting of dextrins, maltodextrins and cyclodextrins.
- dextrins Preferably have an average degree of polymerization ranging from 3 to 200, still preferably 5 to 100 and most preferably 10 to 40.
- Dextrins are a group of low molecular weight carbohydrates obtainable by the hydrolysis of starch forming D-glucose units linked by a-(1 ,4) bonding starting with an a-(1 ,6) glycosidic bond.
- they have a dextrose equivalent between 1 and 30 %, preferably between 1 and 13% based on dextrose (glucose) with 100 %.
- Dextrose equivalent is a measure of the amount of reducing sugars present in a sugar product, expressed as a percentage on a dry basis relative to dextrose.
- the dextrose equivalent gives an indication of the average degree of polymerization (DP) for starch sugars, it is inversely related to the molecular weight.
- the degree of polymerization (DP) is an indicator of the degree of hydrolysis, unhydrolyzed starch has a DE of 0 while glucose has a DE value of 100.
- the dextrins are produced from starch using enzymes or by dry heating under acidic conditions (pyrolysis). Maltodextrins are composed of a-(1 ,4) bonding glucose only and have an average polytenization degree of between 10 and 20 and a dextrose equivalent of 3 to 20 %, preferably 10 to 20 %.
- the cyclodextrin can be any of the known cyclodextrins such as, unsubstituted cyclodextrins containing from six to twelve glucose monomers, especially, alpha-, beta-, and gamma- cyclodextrins, and/or their derivatives, and/or mixtures thereof.
- the alpha-, beta-, and gammacyclodextrins contain 6, 7, and 8 glucose monomer units, respectively, arranged in a donutshaped ring.
- cyclodextrin derivatives suitable for use in the present invention include methyl beta-cyclodextrin, hydroxy-ethyl beta-cyclodextrin, and hydroxypropyl betacyclodextrin of different degrees of substitution.
- Water-soluble cyclodextrin derivatives are preferred cyclodextrin derivatives.
- Preferably at least a major portion of the cyclodextrins is alpha-, beta- and/or gamma-cyclodextrins, more preferably alpha- and beta-cyclo-dextrins.
- An especially preferred cyclodextrin for use in the present invention is beta-cyclodextrin. It is also preferred to use mixtures of cyclodextrins.
- the levels of substituted polysaccharide in the composition ranges from 0.05 wt.% to 5 wt.%.
- the amount of the substituted polysaccharide present in the composition is at least 0.07 wt%, still preferably at least 0.08 wt%, still preferably at least 0.09 wt%, most preferably at least 0.1 wt%, but typically not more than 4 wt.%, still preferably not more than 3 wt.%, most preferably not more than 2 wt.%.
- the substituted polysaccharide is an alkyl substituted polysaccharide.
- Alkyl substituted polysaccharide includes an alkyl group which is typically linear or branched Ci to Cs alkyl group or arylalkyl group.
- Alkyl substituted polysaccharide are preferably manufactured by substitution of hydrogen atoms on the hydroxyl groups of the polysaccharide by alkyl groups and/or arylalkyl groups.
- the preferred alkyl group is a Ci to C4 alkyl group, examples being methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl or tert-butyl.
- the alkyl group may be substituted by an aromatic radical to form the arylalkyl group, such as with a phenyl radical, for example.
- arylalkyl group is benzyl. It is also possible to use mixed alkyl substituted polysaccharide, which contains more than one kind of alkyl, arylalkyl or functionally substituted alkyl group.
- alkyl substituted polysaccharide examples include methyl cellulose, ethyl cellulose, propyl cellulose or mixtures thereof.
- the substituted polysaccharide is an hydroxyalkyl substituted polysaccharide.
- the alkyl and/or arylalkyl groups of the alkyl polysaccharide could further be substituted by nonionic, anionic, cationic, or nonionic groups.
- the alkyl or arylalkyl group may be functionally substituted, by hydroxyl to form hydroxyalkyl substituted polysaccharide.
- hydroxyalkyl substituted polysaccharide includes hydroxyethyl-, hydroxypropyl-, hydroxypropyl methyl cellulose, ethylmethylcellulose cellulose, methylhydroxyethyl cellulose, methylhydroxybutyl cellulose, ethylhydroxydethyl cellulose or mixtures thereof.
- the substituted polysaccharide is a carboxyalkyl substituted polysaccharide.
- the alkyl and/or arylalkyl groups of the alkyl substituted polysaccharide could further be substituted by nonionic, anionic, cationic, or nonionic groups.
- the alkyl or arylalkyl group may be functionally substituted, by carboxyl or carboxylate group to form carboxyalkyl substituted polysaccharide.
- carboxylate groups are present, corresponding counterions are present as well, examples being alkali metal ions such as sodium or potassium, or ammonium ions.
- carboxyalkyl substituted polysaccharide includes carboxymethyl cellulose, carboxymethylhydroxyethylcellulose.
- carboxymethylcelluloses include sodium carboxymethylcellulose, crosslinked sodium carboxymethyl-cellulose or enzymatically hydrolyzed carboxymethylcellulose.
- a fructan polycarboxylic acid is any oligo-or polysaccharide containing a plurality of anhydrofructan units, and which has been converted into a polycarboxylic acid.
- the fructans on which a fructan polycarboxylic acid can be based can have a polydisperse chain length distribution and can have a straight or branched chain.
- the fructans contain chiefly p-2,1 bond, as in inulin, but they can also contain p -2, 6 bonds, as in levan.
- Suitable fructans can originate directly from a natural source, but they can also have undergone a modification.
- Suitable fructans have an average chain length (degree of polymerization, DP) of at least 2 up to about 1000.
- a fructan is used with a degree of polymerization of at least 3, more preferably at least 6, most preferably at least 10 up to about 60.
- the fructan polycarboxylic acid has a degree of substitution by carboxyl groups obtained by oxidation of at least 0.05 per original anhydrofructose unit and a degree of substitution by carboxyalkyl or carboxacyl groups of at least 0.1 per anhydrofructose unit.
- the fructan polycarboxylic acid is based on inulin.
- Inulin is a polysaccharide consisting of p-2,1 bound fructose units with an a-D-glucopyranose unit at the reducing end of the molecule.
- the principal difference between inulin different natural sources resides in the degree of polymerization.
- the degree of polymerization may vary from about 6 to higher than 20, more preferably 6 to 14, still preferably a fructan polycarboxylic acid having a degree of polymerization of 9 to 11.
- the derivatives of inulin which are used according to the invention are polycarboxylates.
- suitable polycarboxylate derivatives of inulin are dicarboxy inulin, obtained, for instance, by glycolytic oxidation of inulin, 6-carboxy inulin, obtained, for instance, by selective oxidation of the primary hydroxyl groups of inulin (TEMPO oxidation), carboxymethyl inulin, carboxyethyl inulin, obtained, for instance, by cyanoethylation followed by hydrolysis.
- Derivatives of inulin which are preferably used are dicarboxy inulin, carboxymethyl inulin and carboxyethyl inulin.
- fructan polycarboxylic acids containing carboxyl groups obtained by oxidation of carbon atoms forming part of the anhydrofructose units in the molecule as well as carboxyl groups obtained by attachment of carboxyalkyl or carboxyacyl groups to the anhydrofructose units.
- These twofold modified fructan polycarboxylic acids can be prepared by oxidizing a fructan in a known manner, followed by carboxyalkylation or carboxyacylation of the oxidation product in a known manner. It is also possible to change this order around and to have carboxyalkylation or carboxyacylation precede oxidation.
- carboxylic acid include both the free acid and metal or ammonium salts of the carboxylic acid.
- carboxyalkyl refers to a Ci to C4 alkyl group substituted by one or more carboxyl groups, such as carboxymethyl, carboxyethyl, dicarboxymethyl, 1 ,2- dicarboxyethyl or mixtures thereof.
- carboxyacyl refers to a Ci to C4 acyl group, in particular a Ci to C4 alkanoyl or alkenoyl group substituted by one or more carboxyl groups, such as carboxyacetyl, p-carboxypropionyl, p-carboxyacryloyl, y-carboxybutyryl, dicarboxyhydroxybutyryl, and mixtures thereof.
- carboxyalkyl and carboxyacyl groups a preference is expressed for carboxymethyl.
- the fructan polycarboxylic acid has an average, of at least 0.05 carboxyl groups per monosaccharide unit.
- the number of carboxyl groups per monosaccharide unit of the fructan polycarboxylic acid can be expressed in the total degree of substitution of carboxyl groups (DS) or the total degree of oxidation (DO).
- the fructan polycarboxylic acid includes between 0.5 and 3 carboxyl groups per monosaccharide unit. Most preferably the fructan polycarboxylic acid is a carboxymethyl inulin including 0.5 and 3 carboxyl groups per monosaccharide unit.
- the substituted polysaccharide is a carboxylic acid substituted polysaccharide.
- carboxylic acid substituted polysaccharide includes acrylic acid substituted polysaccharide, malic acid substituted polysaccharide, succinic acid substituted polysaccharide, most preferred are the acrylate substituted polysaccharide.
- Non-limiting example of the carboxylic acid substituted polysaccharide is acrylated starch.
- the substituted polysaccharide is selected from carboxyalkyl polysaccharide, carboxylic acid polysaccharide or mixtures thereof. Still more preferably the carboxyalkyl polysaccharide is selected from carboxymethyl cellulose, carboxymethyl inulin or mixtures thereof.
- the carboxylic acid polysaccharide is an acrylic acid substituted polysaccharide or salts thereof, still preferably an acrylic acid substituted starch or salts thereof.
- a solid laundry composition preferably having an acrylic acid maleic acid copolymer.
- maleic acid as used herein includes both maleic acid, maleic anhydride and/or salts thereof.
- acrylic acid as used herein includes acrylic acid, methacrylic acid and/or salts thereof.
- the acrylic maleic copolymer preferably includes from 10 weight percent to 90 weight percent acrylic acid and from 10 weight percent to 90 weight percent maleic acid and salts thereof. Still preferably from 60 weight percent to 90 weight percent acrylic acid and 10 weight percent to 40 weight percent maleic acid. In one preferred embodiment the copolymer of acrylic acid and maleic acid includes from 60 weight percent to 90 weight percent acrylic acid and 10 weight percent to 40 weight percent maleic acid.
- the copolymer of acrylic acid and maleic acid includes from 10 weight percent to 50 weight percent acrylic acid and 50 weight percent to 90 weight percent maleic acid. Still preferably from 10 weight percent to 40 weight percent acrylic acid and 60 weight percent to 90 weight percent maleic acid.
- the copolymer of acrylic acid and maleic acid or salt thereof has a weight ratio of acrylic acid segment to the maleic acid segment ranging from 1 : 1 to 1 :9. More preferably the weight ratio of acrylic acid segment to the maleic acid segment ranges from 1 :1 to 1 :8, still preferably from 1:1 to 1 :6, still more preferably from 1 :1 to 1 :5.
- the copolymer of acrylic acid and maleic acid or salt thereof preferably has a weight ratio of acrylic acid segment to the maleic acid segment ranging from 1 :1.5 to 1:9, still more preferably from 1:2 to 1 :9, furthermore preferably from 1 :2.5 to 1:9, still further preferably from 1 :3 to 1 :9, still furthermore preferably from 1 :3.5 to 1:9.
- copolymer of acrylic acid and maleic acid or salt thereof has a weight ratio of acrylic acid segment to the maleic acid segment ranging from 1:4 to 1:8, more preferably from 1 :5 to 1 :8, also preferably from 1:6 to 1 :8.
- the acrylic acid maleic acid copolymer has a weight average molecular weight ranging from 10,000 to 120,000, preferably 50,000 to 100,000, more preferably 60,000 to 100,000, and most preferably 60,000 to 90,000.
- the acrylic acid maleic acid copolymer should be partially or fully neutralized.
- the copolymer of acrylic acid and maleic acid has 70 weight percent acrylic acid and 30 weight percent maleic acid.
- a preferred example of acrylic acid maleic acid copolymer is Sokalan CP5 from BASF.
- the weight average molecular weight ranges from 500 to 7000 still preferably from 500 to 4000, still more preferably from 2000 to 4000.
- Water-soluble salts of the copolymer of acrylic acid and maleic acid is also preferred for the present invention.
- the salts include those selected from non-limiting examples selected from alkali metal, ammonium and substituted ammonium salts.
- those copolymer having a weight ratio of acrylic acid segment to the maleic acid segment ranging from 1 :1 to 1:9.
- the copolymer has a weight average molecular weight in the range from 500 to 7000 and a weight ratio of acrylic acid segment to the maleic acid segment from 1 :1 to 1 :9, more preferably 1 :1.5 to 1 :9; still preferably from a weight average molecular weight in the range from 500 to 4000 and a weight ratio of acrylic acid segment to the maleic acid segment from 1 :1 to 1:9, more preferably 1 :1.5 to 1:9; and still further preferably where the weight average molecular weight in the range from 2000 to 4000 and a weight ratio of acrylic acid segment to the maleic acid segment from 1 : 1 to 1 :9, more preferably from 1 : 1.5 to 1 :9.
- the solid laundry detergent composition includes a copolymer of acrylic acid and maleic acid with a weight ratio of acrylic acid segment to the maleic acid segment from 1 :1 to 1 :9 and another copolymer of acrylic acid and maleic acid with a weight ratio of acrylic acid segment to the maleic acid segment from 1.5:1 to 9:1.
- the solid detergent composition has higher levels of the copolymer of acrylic acid and maleic acid with a weight ratio of acrylic acid segment to the maleic acid segment from 1 :1 to 1:9.
- the solid laundry composition preferably includes from 0.02 wt.% to 0.5 wt.% acrylic acid maleic acid copolymer.
- the levels of acrylic acid maleic acid copolymer in the composition ranges from 0.03 wt.% to 0.5 wt.%.
- the amount of the acrylic acid maleic acid copolymer present in the composition is at least 0.022 wt%, still preferably at least 0.025 wt%, still preferably at least 0.028 wt%, most preferably at least 0.03 wt%, but typically not more than 0.45 wt.%, still preferably not more than 0.4 wt.%, still more preferably not more than 0.35 wt.%, most preferably not more than 0.3 wt.%.
- a solid laundry composition according to the present disclosure encompasses a variety of spray-dried or granulated forms including, for example powder, particulates; cast and extruded forms including, for example, solids, needles, pellets, blocks, bars, and tablets.
- the term “solid” refers to the state of the solid laundry composition under the expected conditions of storage and use of the solid laundry composition. In general, it is expected that the solid laundry composition will remain a solid when provided at a temperature of up to about 37°C and preferably greater than 50°C.
- the composition according to the present invention preferably has a pH from 7.0 to 13, preferably 7.0 to 12.5, still preferably 7.0 to 12, still further preferably from 8.5 to 9, when measured at 1 wt.% dilution in de-ionised water at 25°C.
- the composition may preferably include a buffer.
- the laundry composition of the present invention is in the solid form.
- the laundry composition according to the present invention may be made via a variety of conventional methods known in the art and those which includes but is not limited to the mixing of ingredients, including drymixing, wet-granulation, compaction such as agglomerating, extrusion, tabletting, or spraydrying of the various compounds comprised in the detergent component, or mixtures of these techniques, whereby the components herein also can be made by for example compaction, including extrusion and agglomerating, or spray-drying.
- the solid laundry composition may be made by any of the conventional processes, especially preferred is the technique of slurry making and spray drying or agglomeration.
- compositions herein can take a variety of physical solid forms including forms such as powder, granule, ribbon, noodle, paste, tablet, flake, needles, pastille and bar, and preferably the composition is in the form of powder, granules or a tablet, still preferably the composition is in the form of a powder.
- the laundry composition may be in the form of a unit dose formulation, delayed delivery formulation, detergent contained on or in a porous substrate or nonwoven sheet, and other suitable forms that may be apparent to one skilled in the art in view of the teachings herein.
- the composition according to the present invention may preferably be in a form selected from powder, unit dose or pouch form, tablet, bar, or flake.
- the solid laundry detergent composition according to the present invention is preferably free flowing.
- the composition is preferably a fully formulated detergent composition.
- the solid detergent composition includes but is not limited to powder, granular, particulate, agglomerates, noodles, flakes tablets, bar, sheet or other solid forms known in the art and combinations thereof.
- the composition is used for laundering fabrics using manual-washing method.
- the composition of the present invention is a solid laundry detergent composition in the form of a spray -dried powder or particulate free-flowing form.
- compositions preferably have a density of more than 350 grams/litre, more preferably more than 450 grams/litre or even more than 570 grams/litre. More preferably the solid laundry composition has a bulk density ranging from 350 grams/litre to 800 grams/litre.
- the solid laundry composition according to the first aspect of the present invention preferably includes a surfactant.
- the surfactant includes those selected from the group consisting of anionic surfactant, cationic surfactant, amphoteric surfactant, zwitterionic surfactant, nonionic surfactant or combinations thereof.
- anionic surfactant examples include those selected from the group consisting of alkyl sulfates, alkyl sulfonates, alkylaryl sulfonates, alkyl sulfosuccinates, alkyl ether sulfosuccinates, alkyl sulfosuccinamates, alkyl amidosulfosuccinates, alkyl carboxylates, alkyl amido ether carboxylates, alkyl succinates, fatty acyl sarcosinates, fatty acyl amino acids, fatty acyl taurates, fatty alkyl sulfoacetates, alkyl phosphates, and mixtures of two or more thereof.
- Non-limiting examples of the preferred anionic surfactant includes linear alkyl benzene sulphonate, primary
- Suitable sulphate surfactant includes Cs to C20 alkyl sulphate, preferably Cs to C alkyl sulphate, still preferably C12 to C14 alkyl sulphate, more preferably it is predominantly C12 alkyl sulphate.
- the alkyl sulphate surfactant may be linear or branched, substituted or un-substituted, and may be derived from petrochemical material or biomaterial.
- the alkyl sulphate surfactant is linear. Still preferably it is preferred that the alkyl sulphate surfactant is a C10 to C14 alkyl sulphate surfactant, most preferably a lauryl sulphate surfactant.
- R"OSOs'M + Conventional primary alkyl sulfate surfactant has the general formula: R"OSOs'M + wherein R" is typically a Cs to C20 alkyl group, which may be straight chain or branched chain, and M is a water-solubilizing cation.
- R" is a C10 to Cw alkyl group
- M is alkali metal, more specifically R" is C12 to C14 alkyl and M is sodium.
- anionic alkyl sulphate surfactant useful herein include: C10 to C20 primary, branched-chain and random alkyl sulfates (AS); or C10 to C secondary (2,3)-alkyl sulfates having following formulae: wherein M is hydrogen or a cation which provides charge neutrality, and all M units, can either be a hydrogen atom or a cation depending upon the form isolated or the relative pH of the system wherein the surfactant is used, with non-limiting examples of preferred cations including sodium, potassium, ammonium, and mixtures thereof, and x is an integer of at least about 7, preferably at least about 9, and y is an integer of at least 8, preferably at least about 9.
- a preferred sulphate surfactant is alkyl alkoxylated sulphate, preferably alkyl ethoxylated sulphate, preferably a Cs to C is alkyl alkoxylated sulphate, preferably a Cs to Cw alkyl ethoxylated sulphate, preferably the alkyl alkoxylated sulphate has an average degree of alkoxylation of from 0.5 to 20, preferably from 0.5 to 10, preferably the alkyl alkoxylated sulphate is a Cs to Cw alkyl ethoxylated sulphate having an average degree of ethoxylation of from 0.5 to 10, preferably from 0.5 to 5, more preferably from 0.5 to 3 and most preferably from 0.5 to 1.5.
- the alkyl alkoxylated sulphate surfactant may be linear or branched, substituted or un-substituted, and may be derived from petrochemical material or biomaterial.
- the sulphate surfactant is a linear or branched, substituted or unsubstituted Cs to Cw alkyl sulphate surfactant, Cs to Cw alkyl ether sulphate surfactant or mixtures thereof.
- Suitable sulphonate detersive surfactant include methyl ester sulphonate, alpha olefin sulphonates, alkyl benzene sulphonate, especially alkyl benzene sulphonate, preferably C10 to C14 alkyl benzene sulphonate.
- Suitable alkyl benzene sulphonate (LAS) is obtainable, preferably obtained, by sulphonating commercially available linear alkyl benzene (LAB); suitable LAB includes low 2-phenyl LAB, other suitable LAB includes high 2-phenyl LAB, such as those supplied by Sasol under the tradename Hyblene®.
- the sulphonate surfactant may be linear or branched, substituted or un-substituted, and may be derived from petrochemical material or biomaterial.
- the sulphonate surfactant is preferably a linear or branched, substituted or unsubstituted Cw to C14 alkyl benzene sulphonate.
- the sulphonate surfactant may also be selected from the modified alkylbenzene sulfonate (MLAS) as discussed in WO 99/05243, WO 99/05242 and WO 99/05244; methyl ester sulfonate (MES); and alpha-olefin sulfonate (AOS).
- MLAS modified alkylbenzene sulfonate
- MES methyl ester sulfonate
- AOS alpha-olefin sulfonate
- Anionic surfactants may exist in an acid form and the acid form may be neutralized to form a surfactant salt.
- Typical agents for neutralization include a metal counter ion base such as a hydroxide, e.g., NaOH or KOH.
- Further agents for neutralizing anionic surfactants include ammonia, amines, or alkanolamines. Suitable non-limiting examples include monoethanolamine, diethanolamine, triethanolamine, and other linear or branched alkanolamines known in the art, for example, 2-amino-1-propanol, 1-aminopropanol, monoisopropanolamine, or 1-amino-3-propanol.
- the anionic surfactant is a non-soap anionic surfactant.
- the term “soap” is used herein in its popular sense, i.e. , the alkali metal or alkanol ammonium salts of aliphatic, alkanes, or alkene monocarboxylic acids.
- the anionic surfactant includes 0 wt.% to 20 wt.% alkyl sulfates, preferably 0 wt.% to 15 wt.% alkyl sulfates, preferably 0 wt.% to 10 wt.% alkyl sulfates, preferably PAS.
- the anionic surfactant may also include from 0 wt.% to 10 wt.% MES, preferably 0 wt.% to 5 wt.% MES.
- the detergent composition of the present invention includes from 2 wt.% to 50 wt.% of anionic detersive surfactant, most preferably from 2 wt.% to 25 wt.% anionic surfactant.
- the detergent composition comprises at least 4 wt.%, still preferably at least 5 wt.%, still preferably at least 8 wt.%, still preferably at least 10 wt.%, still more preferably at least 12 wt.% of the anionic surfactant, most preferably at least 15 wt.% of the anionic surfactant, but typically not more than 45 wt.%, still preferably not more than 40 wt.%, still further preferably not more than 35 wt.%, still more preferably not more than 30 wt.% and most preferably not more than 25 wt.%, still more preferably not more than 20 wt.% of an anionic surfactant based on the weight of the detergent composition.
- Non-ionic surfactant is N-ionic surfactant
- Non-limiting examples of nonionic surfactants include: C12 to C alkyl ethoxylates,
- Non-limiting examples of cationic surfactants include: the quaternary ammonium surfactants, which can have up to 26 carbon atoms include: alkoxylate quaternary ammonium (AQA) surfactants, dimethyl hydroxyethyl quaternary ammonium, dimethyl hydroxyethyl lauryl ammonium chloride; polyamine cationic surfactants and cationic ester surfactants.
- AQA alkoxylate quaternary ammonium
- Non-limiting examples of zwitterionic or ampholytic surfactants include derivatives of secondary and tertiary amines, derivatives of heterocyclic secondary and tertiary amines, or derivatives of quaternary ammonium, quaternary phosphonium or tertiary sulfonium compounds.
- zwitterionic surfactants includes betaines, including alkyl dimethyl betaine and coco dimethyl aminopropyl betaine, Cs to C (for example from C12 to C ) amine oxides and sulfo and hydroxy betaines, such as N-alkyl-N, N-dimethylammino-1-propane sulfonate where the alkyl group can be Cs to Cw and in certain embodiments from C10 to C14.
- the solid laundry composition preferably has a combination of anionic surfactants.
- the anionic surfactant is selected from alkyl benzene sulphonate surfactant, alkyl sulphate surfactant, alkyl ether sulfate surfactant or mixtures thereof.
- the total amount of the alkyl sulphate surfactant and alkyl ether sulfate surfactant in the composition is lower than 50 wt.% of the total anionic surfactant present in the composition, still preferably the total amount of the alkyl sulphate surfactant and alkyl ether sulfate surfactant in the composition is lower than 50 wt.% of the total surfactant content present in the composition.
- the total amount of the alkyl sulphate surfactant and alkyl ether sulfate surfactant in the composition is higher than 50 wt.% of the total anionic surfactant present in the composition, still preferably the total amount of the alkyl sulphate surfactant and alkyl ether sulfate surfactant in the composition is higher than 50 wt.% of the total surfactant content present in the composition.
- the solid laundry composition may include a combination of anionic surfactant and nonionic surfactant.
- anionic surfactant is either or both alkyl sulphate surfactant and alkyl ether sulfate surfactant.
- the total amount of the alkyl sulphate surfactant, alkyl ether sulfate surfactant and nonionic surfactant in the composition is higher than 50 wt.% of the total surfactant content present in the composition.
- the detergent composition of the present invention preferably includes a carbonate builder.
- the composition may also include sodium bicarbonate, potassium carbonate, sodium sesquicarbonate or mixtures thereof.
- the detergent composition of the present invention includes from 0 wt.% to 35 wt.% carbonate builder, more preferably from 0 wt.% to 20 wt.%.
- the detergent composition comprises at least 3 wt.%, still preferably at least 5 wt.%, still preferably at least 8 wt.%, most preferably at least 10 wt.% of the carbonate builder, most preferably at least 12 wt.% of the carbonate builder but typically not more than 30 wt.%, still preferably not more than 28 wt.%, most preferably not more than 25 wt.%, most preferably not more than 22 wt.% of carbonate builder based on the weight of the laundry detergent composition.
- the carbonate builder may include 100 wt.% alkali carbonate builder, preferably sodium carbonate builder or may be a combination of alkali metal carbonate, alkali metal bicarbonate, alkali metal sesquicarbonate or combination thereof.
- the detergent composition of the present invention preferably includes a silicate salt.
- the silicate salt is preferably an alkali metal silicate salt which acts as an alkalinity agent and a builder in the solid detergent composition.
- Suitable silicates include the water-soluble sodium silicates with an SiC>2: Na2 ⁇ D ratio of from 1.0 to 2.8, with ratios of from 1.6 to 2.4 being preferred, and 2.0 ratio being most preferred.
- the silicates may be in the form of either the anhydrous salt or a hydrated salt.
- Sodium silicate with an SiC>2: Na2 ⁇ D ratio of 2.0 is the most preferred silicate.
- Silicates are preferably present in the solid laundry compositions in accordance with the invention at a level ranging from 0 wt.% to 17 wt.% by weight of the composition, more preferably from 0 wt.% to 15 wt.%, even more preferably from 0 wt.% to 13 wt.% in the solid laundry composition.
- the composition of the present invention preferably includes from 0 wt.% to 17 wt.%, still preferably from 0 wt.% to 15 wt.%, more preferably from 0 wt.% to 8 wt.% silicate salt, still preferably 0 wt.% to 5 wt.%, and still more preferably 0 wt.% to 1 wt.% silicate salt.
- the composition of the present invention is substantially free of silicate salt. By substantially free it is meant that there is no deliberately added carbonate salt in the composition.
- the solid laundry composition of the present invention preferably includes inorganics salts selected from the group consisting of calcite, dolomite, magnesium carbonate or mixtures thereof.
- the composition of the present invention preferably includes from 0 wt.% to 10 wt.%, still preferably from 0 wt.% to 6 wt.%, more preferably from 0 wt.% to 5 wt.% inorganics salts selected from calcite, dolomite, or mixtures thereof.
- the solid laundry composition of the present invention preferably includes soluble inorganic salts selected from the group consisting of alkali metal sulphate, alkali metal chloride, alkaline earth metal sulphate or mixtures thereof. More preferably the composition includes soluble inorganic salts selected from the group consisting of sodium sulphate, sodium chloride, magnesium sulphate or mixtures thereof.
- the composition of the present invention includes from 10 wt.% to 60 wt.%, still preferably from 15 wt.% to 50 wt.%, more preferably from 15 wt.% to 40 wt.% soluble inorganics salts.
- the solid laundry composition may preferably include a biodegradable chelating agent.
- the biodegradable chelating agent includes MGDA, GLDA, gluconate or mixtures thereof. More preferably the biodegradable chelating agent is gluconate.
- the composition of the present invention preferably includes from 0 wt.% to 10 wt.%, still preferably from 0 wt.% to 6 wt.%, more preferably from 0 wt.% to 5 wt.%, even more preferably 0 wt.% to 3 wt.% biodegradable chelating agents, more preferably gluconate.
- the solid laundry composition of the present invention may include a bleach.
- the bleach is preferably selected from percarbonate bleach. More preferably a bleach activator is present.
- An example of bleach activator includes TAED, other known bleach activators may also be used.
- Bleach system generally includes a bleach and a bleach activator.
- the solid laundry composition includes 0 wt.% to 6 wt.% bleach, still preferably from 0 wt.% to 5 wt.% bleach, still more preferably from 0 wt.% to 3 wt.% bleach.
- the total amount of the bleach and bleach activator present in the composition of the present invention ranges from 0 wt.% to 10 wt.%, more preferably 0 wt.% to 8 wt.%, still preferably from 0 wt.% to 5 wt.% and still more preferably from 0 wt.% to 3 wt.%.
- the solid laundry composition comprises 0 wt.% boron-based bleach.
- Example of the boron-based bleach includes sodium perborate.
- the solid laundry composition comprises 0 wt.% boron-based bleach activator.
- the solid laundry composition includes from 1 wt.% to 3.5 wt.%, still preferably 1 wt.% to 3 wt.% water.
- the solid laundry composition is either agglomerated or spray-dried.
- the solid laundry composition according to the present invention preferably has from 0 wt.% to 4 wt.% zeolite builder.
- the amount of zeolite builder is less than 3 wt.%, still preferably less than 2.5 wt.%, more preferably less than 2 wt.% by weight in the detergent composition and most preferably the detergent composition is substantially free of zeolite builder.
- the solid laundry detergent composition according to the present invention preferably has from 0 wt.% to 2 wt.% phosphorus containing chemicals.
- the phosphorous containing chemicals includes STPP, HEDP or mixtures thereof.
- the amount of phosphorus containing chemicals is less than 2 wt.%, still preferably less than 1.5 wt.%, more preferably less than 1 wt.% by weight in the solid laundry composition and most preferably the solid laundry composition is substantially free of phosphorus containing chemicals.
- the composition includes 0 wt.% to 2 wt.% STPP.
- the solid laundry composition is substantially free of STPP.
- the composition includes 0 wt.% to 2 wt.% HEDP.
- the solid laundry composition is substantially free of HEDP.
- the composition includes 0 wt.% STPP.
- the composition includes 0 wt.% HEDP.
- substantially free means that the indicated component is at the very minimum, not deliberately added to the composition to form part of it. It is meant to include compositions whereby the indicated material is present only as an impurity in one of the other materials deliberately included.
- the solid laundry composition of the present invention may preferably include one or more of the optional ingredients selected from the group consisting of cleaning and care ingredients.
- the optional ingredients include one or more adjunct cleaning additives selected from polymers, further enzymes, enzyme stabilizer, brightening agents, whitening agent, bleach, humectant, perfume, filler or carrier, an alkalinity system, a buffer, or combinations thereof.
- composition of the present invention may preferably include polymers which provide cleaning or care benefits.
- the cleaning polymer includes but is not limited to soil release polymer, antiredeposition polymers, cellulosic polymers, care polymers, amphiphilic alkoxylated grease cleaning polymers, clay soil cleaning polymers, soil suspending polymers or mixtures thereof.
- Suitable further carboxylate polymer may preferably include polyacrylic acid, polymethacrylic acid, polymaleic acid, copolymers of maleic acid with vinyl methyl ether or mixtures thereof.
- Anti-redeposition polymers are designed to suspend or disperse soil. Typically, antiredeposition polymers in addition to those already discussed above may preferably include those selected from polyethylene glycol polymers, polyethyleneimine polymers or mixtures thereof.
- Soil release polymers are designed to modify the surface of the fabric to facilitate the ease of removal of soil.
- Suitable soil release polymers are sold by Clariant under the TexCare® series of polymers, e.g. TexCare® SRN240, TexCare® SRN100, TexCare® SRN170, TexCare® SRN300, TexCare® SRN325, TexCare® SRA100 and TexCare® SRA300.
- Other suitable soil release polymers are sold by Rhodia under the Repel-o-Tex® series of polymers, e.g. Repel-o- Tex® SF2, Repel-o-Tex® SRP6 and Repel-o-Tex® Crystal.
- a preferred polymer is selected from the group consisting of polyester soil release polymer, both end-capped and non-end- capped sulphonated PET/POET polymers, both end-capped and non-end-capped unsulphonated PET/POET polymers or combinations thereof.
- the levels of these soil release polymer in the detergent composition ranges from 0 wt.% to 5 wt.%, at least 0.1 wt%, still preferably at least 0.25 wt.%, still preferably at least 0.3 wt.%, but typically not more than 5 wt%, still preferably not more than 3 wt%, still preferably not more than 1 wt.%, more preferably no more than 0.5 wt.%.
- Suitable care polymers include cellulosic polymers that are cationically modified or hydrophobically modified.
- a suitable commercially available dye lock polymer is Polyquart® FDI (Cognis).
- the adjunct particle includes from 0.01% to 10%, preferably from 0.05% to 0.5% by weight of care polymer.
- Suitable sequestering polymers are DEQUESTTM, organic phosphonate type sequestering polymers sold by Monsanto and alkanehydroxy phosphonates.
- the solid laundry composition is preferably substantially free of phosphate based sequestering polymers.
- substantially free it is meant herein that no phosphate based sequestering polymers is deliberately added.
- composition of the present invention preferably includes one or more further enzymes.
- Preferred examples of the further enzymes include those which provide cleaning performance and/or fabric care benefits.
- Suitable further enzymes include, but are not limited to, hemicellulases, peroxidases, proteases, xylanases, lipases, xyloglucanase, phospholipases, esterases, cutinases, pectinases, mannanases, pectate lyases, keratinases, reductases, oxidases, phenoloxidases, lipoxygenases, ligninases, pullulanases, tannases, pentosanases, malanases, G-glucanases, arabinosidases, hyaluronidase, chondroitinase, laccase, and amylases, or mixtures thereof.
- a typical combination is an enzyme cocktail that may comprise, for example, a protease and lipase in conjunction with one or more of amylase and mannanase.
- the further enzymes may be present at levels from about 0.00001% to about 2%, from about 0.0001% to about 1% or from 0.001% to about 0.5% enzyme protein by weight of the solid laundry composition.
- Suitable proteases include metalloproteases and serine proteases, including neutral or alkaline microbial serine proteases, such as subtilisins (EC 3.4.21.62).
- Suitable commercially available protease enzymes include those sold under the trade names Alcalase®, Savinase®, Primase®, Durazym®, Polarzyme®, Kannase®, Liquanase®, Liquanase Ultra®, Savinase Ultra®, Ovozyme®, Neutrase®, Everlase® and Esperase® by Novozymes A/S (Denmark), those sold under the tradename Maxatase®, Maxaca®l, Maxapem®, Properase®, Purafect®, Purafect Prime®, Purafect Ox®, FN3®, FN4®, 10 Excellase® and Purafect OXP® by Genencor International, those sold under the tradename Opticlean® and Optimase by Solvay Enzymes.
- Suitable alpha-amylases include those of bacterial or fungal origin. Suitable commercially available alpha-amylases include DURAMYL®, LIQUEZYME®, TERMAMYL®, TERMAMYL ULTRA®, NATALASE®, SUPRAMYL®, STAINZYME®, STAINZYME PLUS®, FUNGAMYL® and BAN® (Novozymes A/S, Bagsvaerd, Denmark), 15 KEMZYM® AT 9000 Biozym Biotech Trading GmbH Wehlistrasse 27b A-1200 Wien Austria, RAPIDASE®, PURASTAR®, ENZYSIZE®, OPTISIZE HT PLUS®, POWERASE® and PURASTAR OXAM® (Genencor International Inc., Palo Alto, California) and KAM® (Kao, 14-10 Nihonbashi Kayabacho, 1- chome, Chuo-ku Tokyo 103-8210, Japan). In one aspect, suitable amylases include
- such enzymes may be selected from the group consisting of: lipases, including "first cycle lipases".
- Preferred lipases would include those sold under the tradenames Lipex® and Lipolex®.
- Pectate lyases sold under the tradenames Pectawash®, Pectaway®, Xpect® and mannanases sold under the tradenames Mannaway® (all from Novozymes A/S, Bagsvaerd, Denmark), and Purabrite® (Genencor International Inc., Palo Alto, California).
- the enzyme-containing compositions described herein may optionally comprise from 0.001% to 10%, in some examples from about 0.005% to about 8%, and in other examples, from about 0.01 % to about 6%, by weight of the composition, of an enzyme stabilizing system.
- the enzyme stabilizing system can be any stabilizing system which is compatible with the detersive enzyme.
- Such stabilizing systems can, for example, comprise calcium ion, propylene glycol, short chain carboxylic acids, chlorine bleach scavengers and mixtures thereof, and are designed to address different stabilization problems depending on the type and physical form of the cleaning composition.
- a method for laundering a textile surface with the solid laundry composition according to the first aspect of the present invention comprising the steps of: iv) preparing an aqueous wash liquor by contacting the solid laundry composition according to the first aspect with a liquid; v) soaking said textile surface in the wash liquor for a predetermined period of time; and, vi) optionally rinsing the textile surface.
- compositions are preferably employed at concentrations of from about 200 ppm to about 15,000 ppm in solution.
- the liquid is water.
- Water temperature is preferably in the range from 5°C to 100°C.
- Machine laundry methods herein typically comprise treating soiled textile fabric with an aqueous wash solution in a washing machine having dissolved or dispensed therein an effective amount of the solid laundry composition in accordance with the invention.
- an effective amount of the solid laundry composition it is meant from 20 g to 300 g of product dissolved or dispersed in a wash solution of volume from 5 to 65 liters, as are typical product dosages and wash solution volumes commonly employed in conventional machine laundry methods.
- Hand-washing methods, and combined handwashing with semi-automatic washing machines are also included.
- a cellulase enzyme and an optical brightener represented by the general formula (I) according to the first aspect in a solid laundry composition to provide improved cleaning performance.
- the improved cleaning performance is as compared to a composition having cellulase enzyme and traditional (well known in laundry compositions) optical brightener selected from the group consisting of Tinopal CBSX and DASCC-type optical brightener.
- a cellulase enzyme and an optical brightener of the general formula (I) according to the first aspect in a solid laundry composition for exhibiting improved biodegradability.
- the improved biodegradability is as compared to a composition having cellulase enzyme and traditional (well known in laundry compositions) optical brightener selected from the group consisting of Tinopal CBSX and DASCC-type optical brightener.
- a spray-dried solid laundry composition in accordance with the present invention was prepared by spray drying an aqueous slurry in a counter current spray drier.
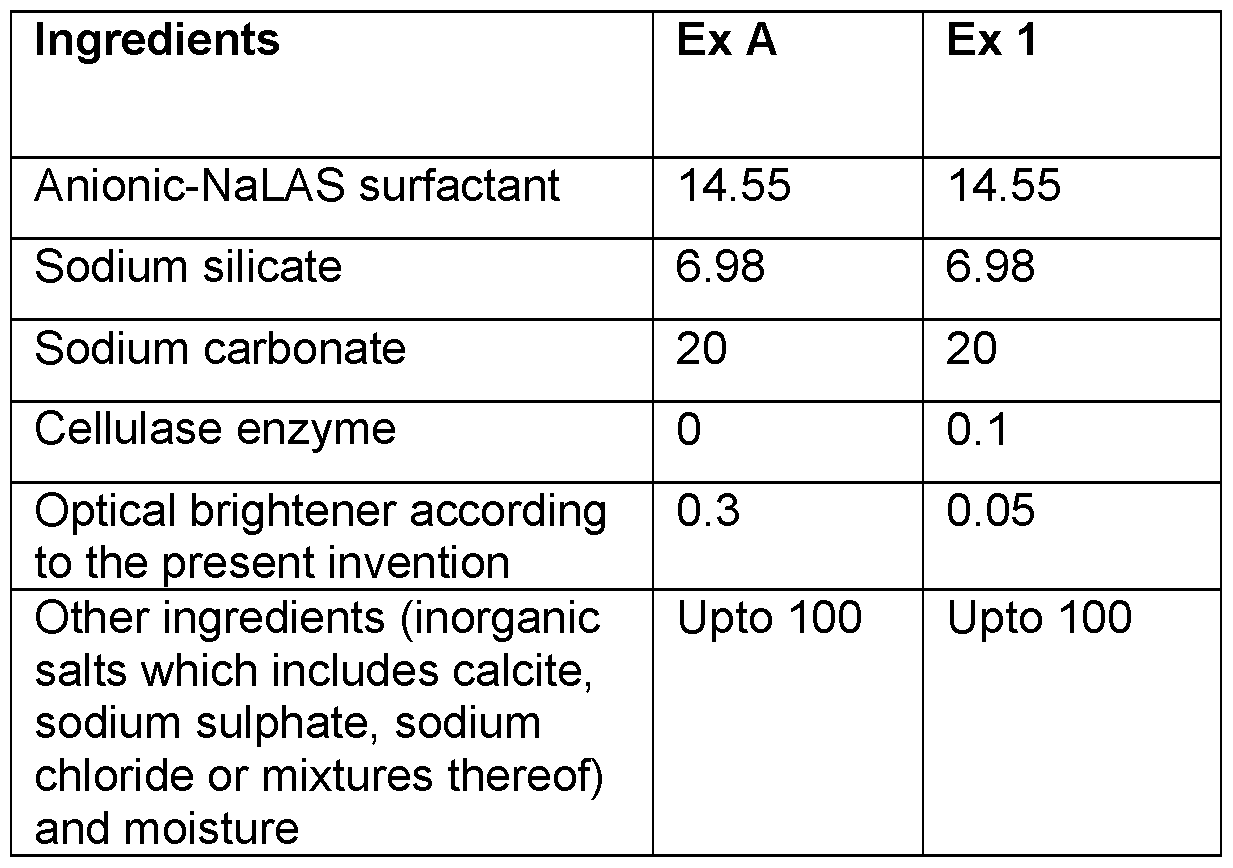
- the composition of a spray- dried solid laundry detergent composition according to the present invention (Ex 1) and a composition having higher levels is provided in table 1.
- Example 1 The composition in accordance to the present invention (Ex 1) which includes a combination of cellulase enzyme and low levels of optical brightener according to the present invention was found to provide a good cleaning performance and has good biodegradability.
- Example 2 The composition in accordance to the present invention which includes a combination of cellulase enzyme and low levels of optical brightener according to the present invention was found to provide a good cleaning performance and has good biodegradability.
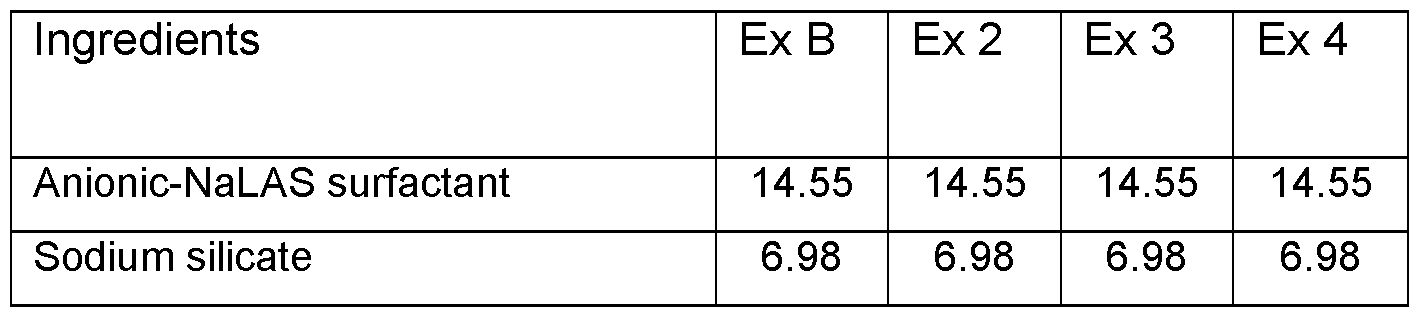
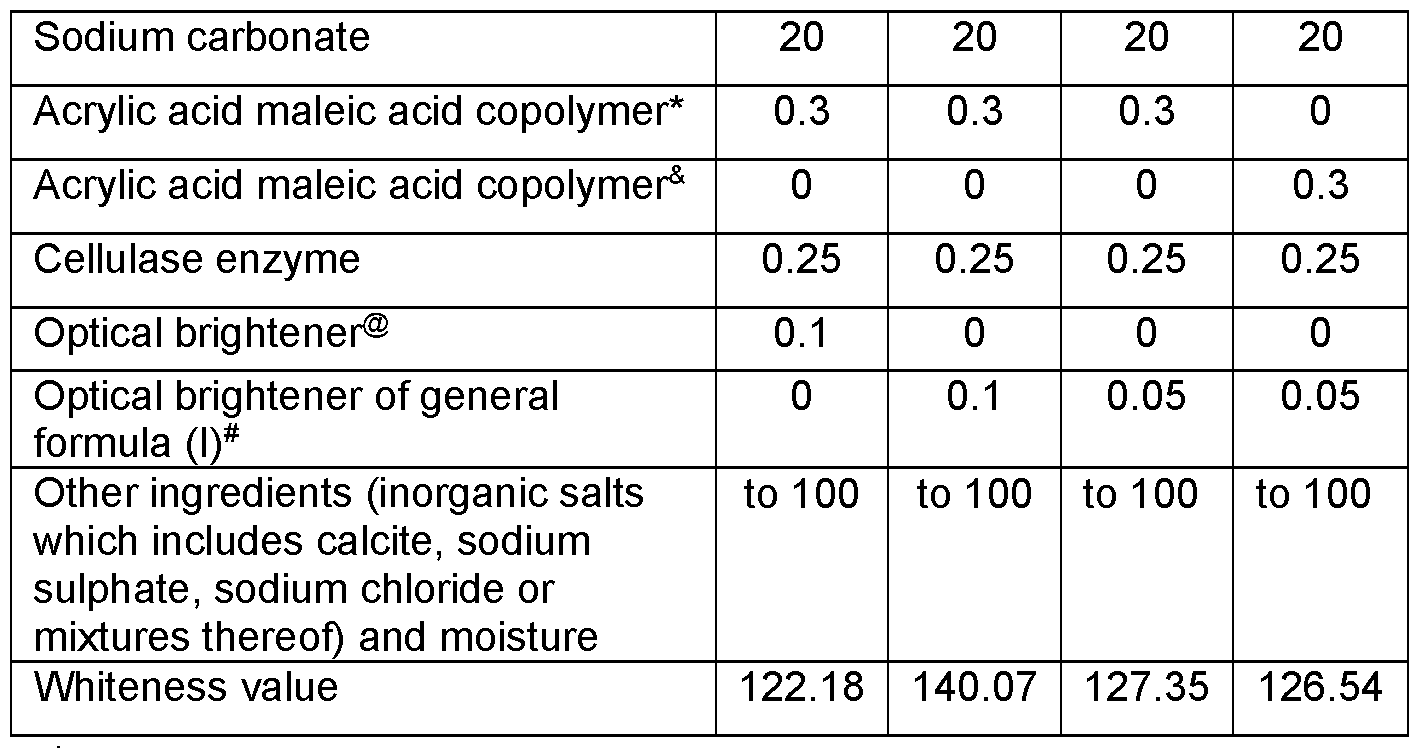
- Different spray-dried solid laundry composition as shown in table 2 were prepared by spray drying an aqueous slurry in a counter current spray drier.
- the acrylic acid maleic acid copolymer, cellulase enzyme, and optical brightener were added to the spray-dried powder to form the solid laundry compositions.
- each of the composition provided in table 2 was evaluated for their whiteness performance. For this, an amount of 4 gram/litre of the solid laundry composition was added to tergo-to-meter having 500 mL of water (water hardness 6FH Ca 2+ : Mg 2+ 2:1) and the device was run for 2 minutes at a speed of 100 rpm and a water temperature of 25°C. Next 2 types of standard commercially available soil strips were added to the wash liquor and the machine was run for another 2 minutes at a speed of 100 rpm and maintaining the water temperature of 25°C. These soil strips included an amount of 2 grams/Litre of SBL soil emulsion and an amount of 1.5 grams/Litre of Stanley clay soil.
- Acrylic acid maleic acid copolymer having weight ratio of acrylic acid to maleic acid of 50:50
- ® traditional optical brightener Tinopal® DMS (DASCC-type optical brightener) - Optical brightener with Structure A as shown hereinabove
- the data provided in table 2 shows that the comparative composition with DASCC type optical brightener (Ex B) and cellulase has a lower whiteness value as compared to the inventive composition (Ex 2) having cellulase and the optical brightener according to general formulae (I) at same levels.
- inventive composition with lower levels of optical brightener according to the present invention in combination with cellulase gives much higher whiteness performance as compared to comparative example (Ex B) having two times higher level of the traditional optical brightener and cellulase enzyme.
- the composition of the present invention provides better performance at much lower levels of the optical brightener and thus provides better biodegradability while maintaining good whiteness performance.
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Wood Science & Technology (AREA)
- Organic Chemistry (AREA)
- Detergent Compositions (AREA)
Abstract
Description
Claims
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23701698.5A EP4469551A1 (en) | 2022-01-28 | 2023-01-23 | Laundry composition |
| CN202380018847.XA CN118647704A (en) | 2022-01-28 | 2023-01-23 | Laundry composition |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP22154013.1 | 2022-01-28 | ||
| EP22154013 | 2022-01-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2023144071A1 true WO2023144071A1 (en) | 2023-08-03 |
Family
ID=80122654
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2023/051512 Ceased WO2023144071A1 (en) | 2022-01-28 | 2023-01-23 | Laundry composition |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP4469551A1 (en) |
| CN (1) | CN118647704A (en) |
| WO (1) | WO2023144071A1 (en) |
Citations (51)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4106991A (en) | 1976-07-07 | 1978-08-15 | Novo Industri A/S | Enzyme granulate composition and process for forming enzyme granulates |
| US4435307A (en) | 1980-04-30 | 1984-03-06 | Novo Industri A/S | Detergent cellulase |
| US4661452A (en) | 1984-05-29 | 1987-04-28 | Novo Industri A/S | Enzyme containing granulates useful as detergent additives |
| WO1989009259A1 (en) | 1988-03-24 | 1989-10-05 | Novo-Nordisk A/S | A cellulase preparation |
| EP0365103A2 (en) * | 1988-10-21 | 1990-04-25 | Colgate-Palmolive Company | Heavy duty particulate synthetic organic nonionic detergent compositions of improved cleaning action on laundry containing hard-to-remove soils |
| WO1991017244A1 (en) | 1990-05-09 | 1991-11-14 | Novo Nordisk A/S | An enzyme capable of degrading cellulose or hemicellulose |
| EP0495257A1 (en) | 1991-01-16 | 1992-07-22 | The Procter & Gamble Company | Compact detergent compositions with high activity cellulase |
| EP0531372A1 (en) | 1990-05-09 | 1993-03-17 | Novo Nordisk As | PREPARATION OF CELLULASE COMPRISING AN ENDOGLUCANASE ENZYME. |
| WO1994007998A1 (en) | 1992-10-06 | 1994-04-14 | Novo Nordisk A/S | Cellulase variants |
| WO1995024471A1 (en) | 1994-03-08 | 1995-09-14 | Novo Nordisk A/S | Novel alkaline cellulases |
| WO1996011262A1 (en) | 1994-10-06 | 1996-04-18 | Novo Nordisk A/S | An enzyme and enzyme preparation with endoglucanase activity |
| WO1996029397A1 (en) | 1995-03-17 | 1996-09-26 | Novo Nordisk A/S | Novel endoglucanases |
| US5648263A (en) | 1988-03-24 | 1997-07-15 | Novo Nordisk A/S | Methods for reducing the harshness of a cotton-containing fabric |
| WO1998008940A1 (en) | 1996-08-26 | 1998-03-05 | Novo Nordisk A/S | A novel endoglucanase |
| WO1998012307A1 (en) | 1996-09-17 | 1998-03-26 | Novo Nordisk A/S | Cellulase variants |
| US5780404A (en) * | 1996-02-26 | 1998-07-14 | The Procter & Gamble Company | Detergent compositions containing enduring perfume |
| WO1999005243A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Detergent compositions containing mixtures of crystallinity-disrupted surfactants |
| WO1999005244A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Improved alkyl aryl sulfonate surfactants |
| WO1999005242A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Improved alkylbenzenesulfonate surfactants |
| WO1999013040A1 (en) | 1997-09-11 | 1999-03-18 | The Procter & Gamble Company | Detergent compositions |
| JP2000210081A (en) | 1999-01-21 | 2000-08-02 | Kao Corp | Thermostable alkaline cellulase gene |
| WO2001062903A1 (en) | 2000-02-24 | 2001-08-30 | Novozymes A/S | Family 44 xyloglucanases |
| WO2002099091A2 (en) | 2001-06-06 | 2002-12-12 | Novozymes A/S | Endo-beta-1,4-glucanase from bacillus |
| JP2003105389A (en) | 2001-09-28 | 2003-04-09 | Lion Corp | Detergent composition and washing method |
| WO2005003274A1 (en) | 2003-06-18 | 2005-01-13 | Unilever Plc | Laundry treatment compositions |
| WO2006027086A1 (en) | 2004-09-11 | 2006-03-16 | Unilever Plc | Laundry treatment compositions |
| WO2006032397A1 (en) | 2004-09-23 | 2006-03-30 | Unilever Plc | Laundry treatment compositions |
| WO2006032327A1 (en) | 2004-09-23 | 2006-03-30 | Unilever Plc | Laundry treatment compositions |
| WO2006045275A2 (en) | 2004-10-25 | 2006-05-04 | Müller Weingarten AG | Drive system for a forming press |
| JP2007070613A (en) | 2005-08-08 | 2007-03-22 | Kao Corp | Detergent composition for clothing |
| WO2008017570A1 (en) | 2006-08-10 | 2008-02-14 | Unilever Plc | Shading composition |
| WO2008087497A1 (en) | 2007-01-19 | 2008-07-24 | The Procter & Gamble Company | Laundry care composition comprising a whitening agent for cellulosic substrates |
| WO2008141880A1 (en) | 2007-05-18 | 2008-11-27 | Unilever Plc | Triphenodioxazine dyes |
| WO2009132870A1 (en) | 2008-05-02 | 2009-11-05 | Unilever Plc | Reduced spotting granules |
| WO2009141172A1 (en) | 2008-05-20 | 2009-11-26 | Unilever Plc | Shading composition |
| WO2009154934A1 (en) | 2008-06-20 | 2009-12-23 | The Procter & Gamble Company | Laundry composition |
| WO2010099997A1 (en) | 2009-03-05 | 2010-09-10 | Unilever Plc | Dye radical initiators |
| WO2010102861A1 (en) | 2009-03-12 | 2010-09-16 | Unilever Plc | Dye-polymers formulations |
| WO2010148624A1 (en) | 2009-06-26 | 2010-12-29 | Unilever Plc | Dye polymers |
| WO2011011799A2 (en) | 2010-11-12 | 2011-01-27 | The Procter & Gamble Company | Thiophene azo dyes and laundry care compositions containing the same |
| WO2011042372A1 (en) | 2009-10-08 | 2011-04-14 | Unilever Plc | Shading composition |
| WO2011098356A1 (en) | 2010-02-12 | 2011-08-18 | Unilever Plc | Laundry treatment composition comprising bis-azo shading dyes |
| US20110212870A1 (en) * | 2010-03-01 | 2011-09-01 | Neil Joseph Lant | Solid Laundry Detergent Composition Comprising Guerbet Alcohol-Based Detersive Surfactant |
| EP2377914A1 (en) | 2010-04-19 | 2011-10-19 | The Procter & Gamble Company | Mildly alkaline, low-built, solid fabric treatment detergent composition comprising perhydrolase |
| WO2012054820A1 (en) | 2010-10-22 | 2012-04-26 | The Procter & Gamble Company | Detergent composition comprising bluing agent and rapidly water-soluble brightener |
| WO2013142495A1 (en) | 2012-03-19 | 2013-09-26 | Milliken & Company | Carboxylate dyes |
| WO2013151970A1 (en) | 2012-04-03 | 2013-10-10 | The Procter & Gamble Company | Laundry detergent composition comprising water-soluble phthalocyanine compound |
| WO2013188331A1 (en) | 2012-06-11 | 2013-12-19 | The Procter & Gamble Company | Detergent composition |
| WO2015144784A1 (en) | 2014-03-25 | 2015-10-01 | Novozymes A/S | Microencapsulation using small amines |
| WO2016023685A1 (en) | 2014-08-11 | 2016-02-18 | Novozymes A/S | Detergents and compositions with enzymatic polymer particles |
| WO2021058023A1 (en) | 2019-09-29 | 2021-04-01 | Novozymes A/S | Deoxyribonuclease uses in detergent composition |
-
2023
- 2023-01-23 WO PCT/EP2023/051512 patent/WO2023144071A1/en not_active Ceased
- 2023-01-23 CN CN202380018847.XA patent/CN118647704A/en active Pending
- 2023-01-23 EP EP23701698.5A patent/EP4469551A1/en active Pending
Patent Citations (58)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4106991A (en) | 1976-07-07 | 1978-08-15 | Novo Industri A/S | Enzyme granulate composition and process for forming enzyme granulates |
| US4435307A (en) | 1980-04-30 | 1984-03-06 | Novo Industri A/S | Detergent cellulase |
| US4661452A (en) | 1984-05-29 | 1987-04-28 | Novo Industri A/S | Enzyme containing granulates useful as detergent additives |
| US5691178A (en) | 1988-03-22 | 1997-11-25 | Novo Nordisk A/S | Fungal cellulase composition containing alkaline CMC-endoglucanase and essentially no cellobiohydrolase |
| US5648263A (en) | 1988-03-24 | 1997-07-15 | Novo Nordisk A/S | Methods for reducing the harshness of a cotton-containing fabric |
| WO1989009259A1 (en) | 1988-03-24 | 1989-10-05 | Novo-Nordisk A/S | A cellulase preparation |
| US5776757A (en) | 1988-03-24 | 1998-07-07 | Novo Nordisk A/S | Fungal cellulase composition containing alkaline CMC-endoglucanase and essentially no cellobiohydrolase and method of making thereof |
| EP0365103A2 (en) * | 1988-10-21 | 1990-04-25 | Colgate-Palmolive Company | Heavy duty particulate synthetic organic nonionic detergent compositions of improved cleaning action on laundry containing hard-to-remove soils |
| US5686593A (en) | 1990-05-09 | 1997-11-11 | Novo Nordisk A/S | Enzyme capable of degrading cellulose or hemicellulose |
| US5457046A (en) | 1990-05-09 | 1995-10-10 | Novo Nordisk A/S | Enzyme capable of degrading cellullose or hemicellulose |
| EP0531372A1 (en) | 1990-05-09 | 1993-03-17 | Novo Nordisk As | PREPARATION OF CELLULASE COMPRISING AN ENDOGLUCANASE ENZYME. |
| US5763254A (en) | 1990-05-09 | 1998-06-09 | Novo Nordisk A/S | Enzyme capable of degrading cellulose or hemicellulose |
| WO1991017244A1 (en) | 1990-05-09 | 1991-11-14 | Novo Nordisk A/S | An enzyme capable of degrading cellulose or hemicellulose |
| EP0495257A1 (en) | 1991-01-16 | 1992-07-22 | The Procter & Gamble Company | Compact detergent compositions with high activity cellulase |
| WO1994007998A1 (en) | 1992-10-06 | 1994-04-14 | Novo Nordisk A/S | Cellulase variants |
| WO1995024471A1 (en) | 1994-03-08 | 1995-09-14 | Novo Nordisk A/S | Novel alkaline cellulases |
| WO1996011262A1 (en) | 1994-10-06 | 1996-04-18 | Novo Nordisk A/S | An enzyme and enzyme preparation with endoglucanase activity |
| WO1996029397A1 (en) | 1995-03-17 | 1996-09-26 | Novo Nordisk A/S | Novel endoglucanases |
| US5780404A (en) * | 1996-02-26 | 1998-07-14 | The Procter & Gamble Company | Detergent compositions containing enduring perfume |
| WO1998008940A1 (en) | 1996-08-26 | 1998-03-05 | Novo Nordisk A/S | A novel endoglucanase |
| WO1998012307A1 (en) | 1996-09-17 | 1998-03-26 | Novo Nordisk A/S | Cellulase variants |
| WO1999005243A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Detergent compositions containing mixtures of crystallinity-disrupted surfactants |
| WO1999005244A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Improved alkyl aryl sulfonate surfactants |
| WO1999005242A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Improved alkylbenzenesulfonate surfactants |
| WO1999013040A1 (en) | 1997-09-11 | 1999-03-18 | The Procter & Gamble Company | Detergent compositions |
| JP2000210081A (en) | 1999-01-21 | 2000-08-02 | Kao Corp | Thermostable alkaline cellulase gene |
| WO2001062903A1 (en) | 2000-02-24 | 2001-08-30 | Novozymes A/S | Family 44 xyloglucanases |
| WO2002099091A2 (en) | 2001-06-06 | 2002-12-12 | Novozymes A/S | Endo-beta-1,4-glucanase from bacillus |
| JP2003105389A (en) | 2001-09-28 | 2003-04-09 | Lion Corp | Detergent composition and washing method |
| WO2005003274A1 (en) | 2003-06-18 | 2005-01-13 | Unilever Plc | Laundry treatment compositions |
| WO2006027086A1 (en) | 2004-09-11 | 2006-03-16 | Unilever Plc | Laundry treatment compositions |
| WO2006032397A1 (en) | 2004-09-23 | 2006-03-30 | Unilever Plc | Laundry treatment compositions |
| WO2006032327A1 (en) | 2004-09-23 | 2006-03-30 | Unilever Plc | Laundry treatment compositions |
| WO2006045275A2 (en) | 2004-10-25 | 2006-05-04 | Müller Weingarten AG | Drive system for a forming press |
| JP2007070613A (en) | 2005-08-08 | 2007-03-22 | Kao Corp | Detergent composition for clothing |
| WO2008017570A1 (en) | 2006-08-10 | 2008-02-14 | Unilever Plc | Shading composition |
| WO2008087497A1 (en) | 2007-01-19 | 2008-07-24 | The Procter & Gamble Company | Laundry care composition comprising a whitening agent for cellulosic substrates |
| WO2008141880A1 (en) | 2007-05-18 | 2008-11-27 | Unilever Plc | Triphenodioxazine dyes |
| WO2009132870A1 (en) | 2008-05-02 | 2009-11-05 | Unilever Plc | Reduced spotting granules |
| WO2009141172A1 (en) | 2008-05-20 | 2009-11-26 | Unilever Plc | Shading composition |
| WO2009141173A1 (en) | 2008-05-20 | 2009-11-26 | Unilever Plc | Shading composition |
| WO2009154934A1 (en) | 2008-06-20 | 2009-12-23 | The Procter & Gamble Company | Laundry composition |
| WO2010099997A1 (en) | 2009-03-05 | 2010-09-10 | Unilever Plc | Dye radical initiators |
| WO2010102861A1 (en) | 2009-03-12 | 2010-09-16 | Unilever Plc | Dye-polymers formulations |
| WO2010148624A1 (en) | 2009-06-26 | 2010-12-29 | Unilever Plc | Dye polymers |
| WO2011042372A1 (en) | 2009-10-08 | 2011-04-14 | Unilever Plc | Shading composition |
| WO2011098356A1 (en) | 2010-02-12 | 2011-08-18 | Unilever Plc | Laundry treatment composition comprising bis-azo shading dyes |
| WO2011109320A1 (en) | 2010-03-01 | 2011-09-09 | The Procter & Gamble Company | Solid laundry detergent composition comprising secondary alcohol - based detersive surfactant |
| US20110212870A1 (en) * | 2010-03-01 | 2011-09-01 | Neil Joseph Lant | Solid Laundry Detergent Composition Comprising Guerbet Alcohol-Based Detersive Surfactant |
| EP2377914A1 (en) | 2010-04-19 | 2011-10-19 | The Procter & Gamble Company | Mildly alkaline, low-built, solid fabric treatment detergent composition comprising perhydrolase |
| WO2012054820A1 (en) | 2010-10-22 | 2012-04-26 | The Procter & Gamble Company | Detergent composition comprising bluing agent and rapidly water-soluble brightener |
| WO2011011799A2 (en) | 2010-11-12 | 2011-01-27 | The Procter & Gamble Company | Thiophene azo dyes and laundry care compositions containing the same |
| WO2013142495A1 (en) | 2012-03-19 | 2013-09-26 | Milliken & Company | Carboxylate dyes |
| WO2013151970A1 (en) | 2012-04-03 | 2013-10-10 | The Procter & Gamble Company | Laundry detergent composition comprising water-soluble phthalocyanine compound |
| WO2013188331A1 (en) | 2012-06-11 | 2013-12-19 | The Procter & Gamble Company | Detergent composition |
| WO2015144784A1 (en) | 2014-03-25 | 2015-10-01 | Novozymes A/S | Microencapsulation using small amines |
| WO2016023685A1 (en) | 2014-08-11 | 2016-02-18 | Novozymes A/S | Detergents and compositions with enzymatic polymer particles |
| WO2021058023A1 (en) | 2019-09-29 | 2021-04-01 | Novozymes A/S | Deoxyribonuclease uses in detergent composition |
Non-Patent Citations (3)
| Title |
|---|
| "Industrial Dyes", 2002, WILEY VCH |
| CHEMSPIDER: "Tinopal 5BM | C38H38N12Na2O8S2 | ChemSpider", 1 January 2021 (2021-01-01), pages 1 - 3, XP093031733, Retrieved from the Internet <URL:http://www.chemspider.com/Chemical-Structure.4938712.html> [retrieved on 20230315] * |
| CHEMSPIDER: "Tinopal UNPA-GX | C40H44N12O10S2 | ChemSpider", 1 January 2021 (2021-01-01), pages 1 - 3, XP093031734, Retrieved from the Internet <URL:http://www.chemspider.com/Chemical-Structure.4812194.html> [retrieved on 20230315] * |
Also Published As
| Publication number | Publication date |
|---|---|
| EP4469551A1 (en) | 2024-12-04 |
| CN118647704A (en) | 2024-09-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN104350106B (en) | Comprise the laundry care composition of dyestuff | |
| CN104105787B (en) | Fabric care composition containing dyestuff | |
| CN103339245B (en) | Detergent composition comprising bluing agent and rapidly water-soluble brightener | |
| CN111718806B (en) | Leuco triphenylmethane colorants as bluing agents in laundry care compositions | |
| CN105764996B (en) | Laundry care compositions comprising carboxylate dyes | |
| CN102471738A (en) | Mild alkaline low built solid fabric treatment detergent compositions comprising phthalimido peroxycaproic acid | |
| CN105531326A (en) | Laundry care compositions containing thiophene azo carboxylate dyes | |
| CN117460814A (en) | laundry detergent powder | |
| EP4469551A1 (en) | Laundry composition | |
| EP4469545B1 (en) | Laundry composition | |
| EP4392511A1 (en) | Detergent composition | |
| EP3798290B1 (en) | Use of an anionically-modified cellulosic polymer as a dye transfer inhibitor during a textile laundering process | |
| WO2025045967A1 (en) | Solid laundry composition | |
| WO2025045923A1 (en) | Solid laundry composition | |
| WO2025045980A1 (en) | Solid laundry composition | |
| WO2025061599A1 (en) | Solid laundry composition | |
| WO2025045870A1 (en) | Solid laundry composition | |
| CN119343436A (en) | Free-flowing solid granular laundry detergent composition | |
| CN119317699A (en) | Free-flowing solid granular laundry detergent composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 23701698 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202427051587 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2401004815 Country of ref document: TH |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202380018847.X Country of ref document: CN |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112024015215 Country of ref document: BR |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2023701698 Country of ref document: EP Effective date: 20240828 |
|
| ENP | Entry into the national phase |
Ref document number: 112024015215 Country of ref document: BR Kind code of ref document: A2 Effective date: 20240725 |