WO2023046182A1 - Application of pyrido[1,2-a]pyrimidone compound - Google Patents
Application of pyrido[1,2-a]pyrimidone compound Download PDFInfo
- Publication number
- WO2023046182A1 WO2023046182A1 PCT/CN2022/121443 CN2022121443W WO2023046182A1 WO 2023046182 A1 WO2023046182 A1 WO 2023046182A1 CN 2022121443 W CN2022121443 W CN 2022121443W WO 2023046182 A1 WO2023046182 A1 WO 2023046182A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- twenty
- tumor
- independently
- substance
- optionally substituted
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/519—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D519/00—Heterocyclic compounds containing more than one system of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring system not provided for in groups C07D453/00 or C07D455/00
Definitions
- the invention relates to the application of a pyrido[1,2-a]pyrimidinone compound.
- Insulinomas are very rare neuroendocrine tumors characterized clinically by hyperinsulinemia leading to hypoglycemic syndrome. Although insulinomas are less malignant, long-term hypoglycemia caused by insulinomas can cause irreversible damage to vital organs of the human body, seriously affecting the quality of life of patients and even threatening their lives. For example, the symptoms of hypoglycemia caused by insulinoma include: slow thinking, abnormal speech behavior, limb paralysis, and finally coma and death. Controlling hypoglycemic syndrome and inhibiting tumor growth are the goals of treating insulinoma.
- Limited-stage neuroendocrine tumors can be surgically resected. However, about half of neuroendocrine tumors are diagnosed as locally advanced or have distant metastases. Radical surgical resection is difficult, and drug therapy is required to delay tumor progression and prolong patient survival.
- the technical problem to be solved by the present invention is that the existing drug structure for treating pancreatic neuroendocrine tumors is relatively single, and cannot well improve the hypoglycemia symptoms of patients. Therefore, the present invention provides an application of pyrido[1,2-a]pyrimidinone compounds. The compound was able to inhibit the growth of pancreatic neuroendocrine tumors and increase blood sugar levels.
- the present invention provides a kind of application of substance X in the preparation of medicine, and said substance X is the compound as shown in formula I, its pharmaceutically acceptable salt, its solvate or its pharmaceutically acceptable salt A solvate; the drug is a drug for treating pancreatic neuroendocrine tumors;
- E is a C 1-6 alkyl group optionally substituted by R 3 , a C 3-10 cycloalkyl group optionally substituted by R 3 or a C 3-10 heterocycloalkyl group optionally substituted by R 3 ;
- Q is a single bond or -C(R 3 )(R 3 )-;
- A is N or C (R 3 );
- n 1 0, 1, 2 or 3;
- R 1 , R 2 and R 3 are independently H, F, Cl, Br, I, CN, -OR a , -N(R b )(R c ), C 1-3 alkane optionally substituted by R d base,
- n is independently 1, 2, 3, 4, 5 or 6;
- D 2 is independently -C(R a )(R a )-;
- R a , R b and R c are independently H, C 1-6 alkyl optionally substituted by R d or C 3-6 cycloalkyl optionally substituted by R d ;
- R e is H, C 1-6 alkyl optionally substituted by R d , C 1-6 alkoxy optionally substituted by R d , C 3-6 cycloalkyl optionally substituted by R d , or any A C 3-6 cycloalkoxy group substituted by R d ;
- Rd is independently 1, 2 or 3;
- Rd is independently F, Cl, Br, I, CN, -OH, -CHO, -COOH, CH3- , CF3- , CH3O- or CH3CH2O- ;
- any two R 1 , between R a and R a in the same D 2 , between two D 2 , or between R a and a D 2 are jointly connected to the same carbon atom or oxygen
- One or two 3, 4, 5 or 6-membered carbocyclic rings or oxygen heterocyclic rings are formed on the atoms, and the number of oxygen atoms is 1 or 2.
- E is C 1-6 alkyl substituted by R 3 or C 3-6 cycloalkyl substituted by R 3 , and the number of R 3 is 1, 2 or 3 independently.
- E is C 1-6 alkyl or C 3-6 cycloalkyl.
- G 17 is N or C (R 3 );
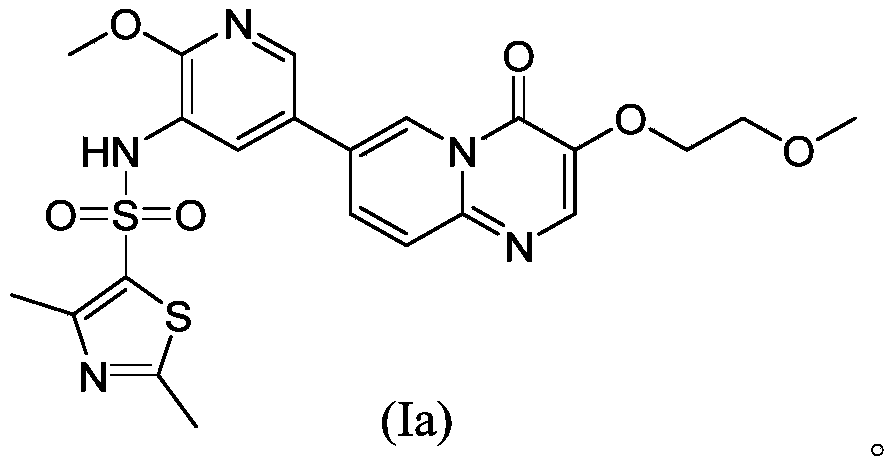
- said substance X can be a compound represented by formula (Ia), its pharmaceutically acceptable salt, its solvate or its pharmaceutically acceptable salt solvate;
- the XRPD pattern of the compound represented by formula (Ia) can be as shown in Figure 1 , Figure 2 , Figure 3 , Figure 4 , Figure 5 or Figure 6 .
- said pancreatic neuroendocrine tumor may be insulinoma, amine precursor uptake and decarboxylation cell tumor, vasoactive intestinal peptide tumor, glucagon tumor, pancreatic polypeptide tumor or gastrinoma.
- said drug is a drug used for treating pancreatic neuroendocrine tumors and increasing blood sugar concentration.
- the present invention provides a method of treating pancreatic neuroendocrine tumors comprising administering to a patient in need thereof a therapeutically effective amount of substance X as described herein.
- the pancreatic neuroendocrine tumor may be an insulinoma, an amine precursor uptake and decarboxylation cell tumor, a vasoactive intestinal peptide tumor, a glucagon tumor, a pancreatic polypeptide tumor, or a gastrinoma.
- the administration regimen of substance X (including administration route, administration dose, administration interval, etc.) can be adjusted by those skilled in the art as needed to provide the optimal therapeutic effect.
- the substance X can be administered by any suitable route known in the art, including oral, injection (eg intravenous, intramuscular, subcutaneous) and the like.
- the substance X is administered orally.
- the substance X can be administered according to the weight of the patient, a non-limiting example range can be 0.01-1 mg/kg (referring to a single dose), such as 0.01 mg/kg, 0.05 mg/kg, 0.1 mg/kg, 0.15 mg/kg kg, 0.2mg/kg, 0.25mg/kg, 0.3mg/kg, 0.35mg/kg, 0.4mg/kg, 0.45mg/kg, 0.5mg/kg, 0.55mg/kg, 0.6mg/kg, 0.7mg/kg kg, 0.8mg/kg, 0.9mg/kg or 1mg/kg.
- the dose of Compound I is 0.05-0.2 mg/kg, such as 0.05 mg/kg, 0.1 mg/kg or 0.2 mg/kg.
- the above doses of the substance X may be administered according to the frequency of QD (once a day), QOD (one day apart) or QW (once a week).
- the substance X is administered on a QD frequency.
- the substance X is administered orally according to the dosage and frequency mentioned above.
- the substance X is administered orally at 0.2 mg/kg, QD.
- the substance X is administered orally at 0.2 mg/kg, QD, and the substance X is a compound represented by formula (Ia).
- the substance X may also be administered to the patient in fixed doses, ie a fixed or predetermined amount of dose is given to the patient.
- a non-limiting example of a fixed dose (referred to as a single dose) may range from 0.01 to 50 mg, such as 0.01 mg, 0.1 mg, 0.5 mg, 1 mg, 2 mg, 3 mg, 4 mg, 5 mg, 6 mg, 7 mg, 8 mg, 9 mg, 10 mg, 11mg, 12mg, 13mg, 14mg, 15mg, 16mg, 17mg, 18mg, 19mg, 20mg, 21mg, 22mg, 23mg, 24mg, 25mg, 26mg, 27mg, 28mg, 29mg, 30mg, 31mg, 32mg, 33mg, 34mg, 35mg, 36mg, 37mg, 38mg, 39mg, 40mg, 41mg, 42mg, 43mg, 44mg, 45mg, 46mg, 47m
- the above-mentioned fixed doses of the substance X may be administered at a frequency of QD (once a day), QOD (once a day) or QW (once a week).
- the substance X is administered on a QD frequency.
- pharmaceutically acceptable salt refers to a salt obtained by reacting a compound with a pharmaceutically acceptable (relatively non-toxic, safe, and suitable for use by patients) acid or base.
- base addition salts can be obtained by contacting the free form of the compound with a sufficient amount of a pharmaceutically acceptable base in a suitable inert solvent.
- Pharmaceutically acceptable base addition salts include, but are not limited to, sodium salts, potassium salts, calcium salts, aluminum salts, magnesium salts, bismuth salts, ammonium salts, and the like.
- acid addition salts can be obtained by contacting the free form of the compound with a sufficient amount of a pharmaceutically acceptable acid in a suitable inert solvent.
- Pharmaceutically acceptable acid addition salts include, but are not limited to, hydrochlorides, sulfates, methanesulfonates, and the like. See Handbook of Pharmaceutical Salts: Properties, Selection, and Use (P. Heinrich Stahl, 2002) for details.
- solvate refers to a substance formed when a compound is combined with a solvent (including but not limited to: water, methanol, ethanol, etc.). Solvates are divided into stoichiometric solvates and non-stoichiometric solvates.
- solvate of a pharmaceutically acceptable salt refers to a compound with a pharmaceutically acceptable (relatively non-toxic, safe, and suitable for patient use) acid or base, solvent (including but not limited to: water, methanol, ethanol etc.), wherein the pharmaceutically acceptable salt has the same meaning as the term “pharmaceutically acceptable salt” above, and the solvent is stoichiometric or non-stoichiometric.
- solvent including but not limited to: water, methanol, ethanol etc.
- Solvates of pharmaceutically acceptable salts include, but are not limited to, hydrochloride monohydrate.
- the reagents and raw materials used in the present invention are all commercially available.
- crystal forms I, II, III, IV, V and VI used in the present invention are prepared according to the method described in CN201680072859.0.
- the positive progress effect of the present invention lies in that the compound can inhibit the growth of pancreatic neuroendocrine tumors and improve the blood sugar level.
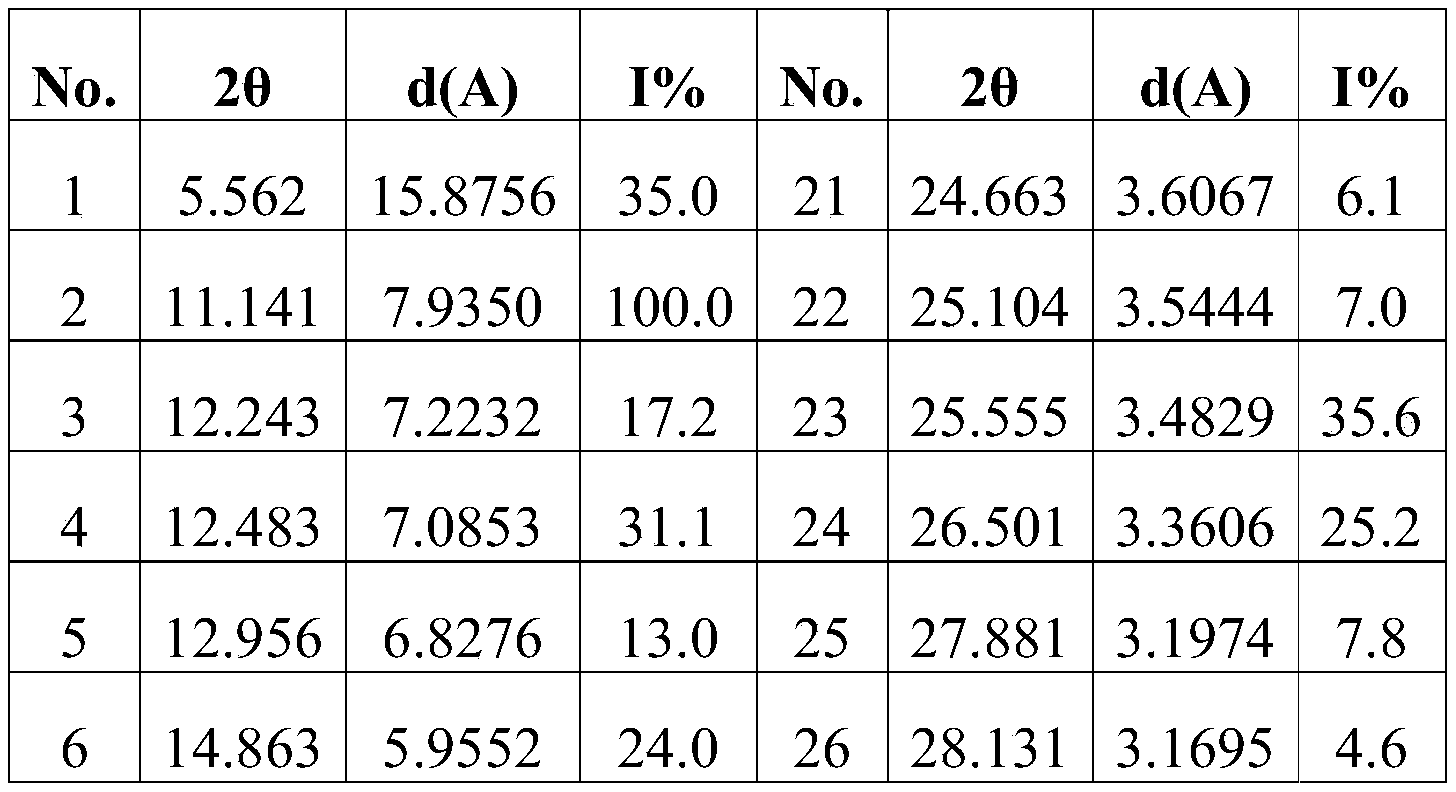
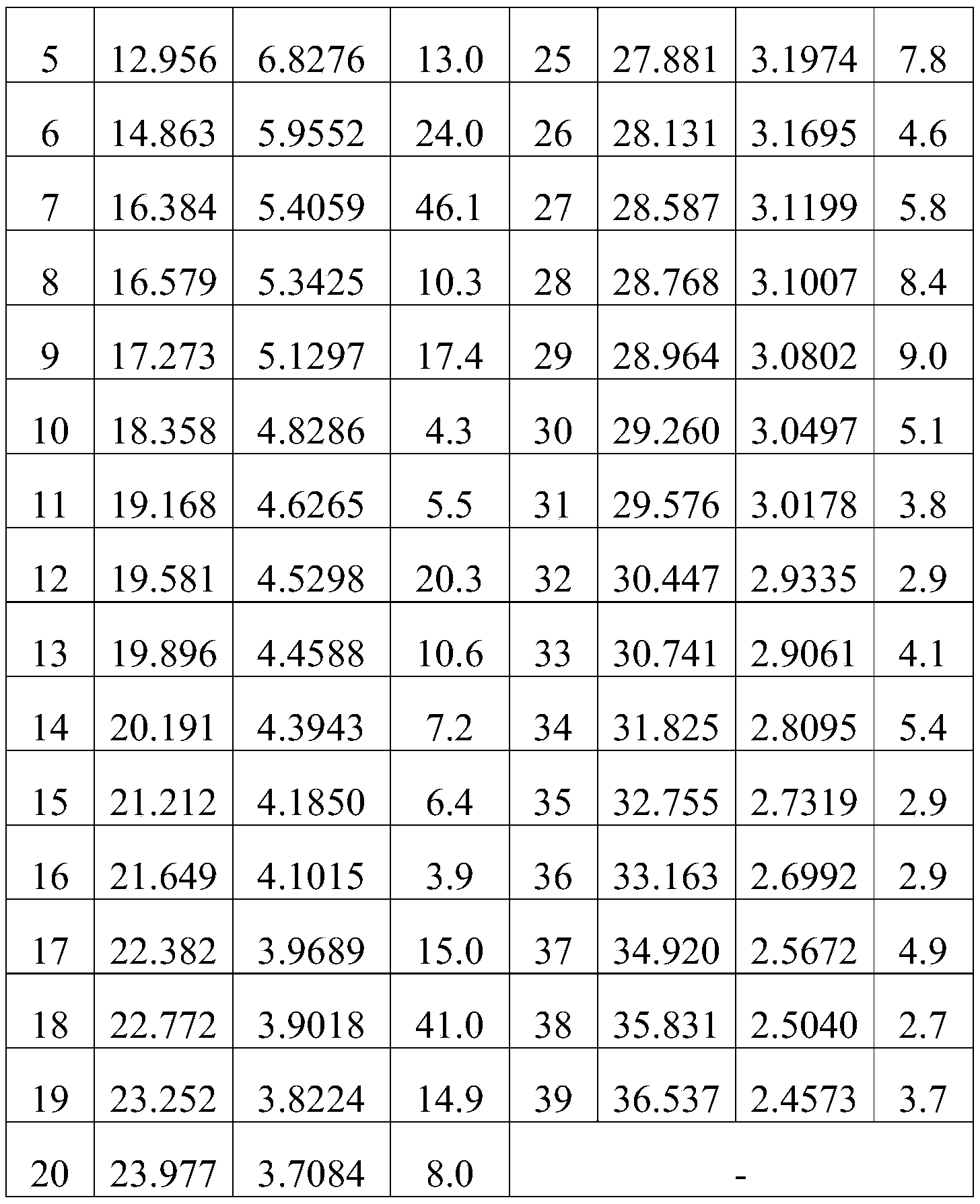
- Figure 1 is the Cu-K ⁇ radiation XRPD spectrum of Form I.
- Fig. 2 is the Cu-K ⁇ radiation XRPD spectrum of the II crystal form.
- Fig. 3 is the Cu-K ⁇ radiation XRPD spectrum of the III crystal form.
- Fig. 4 is the Cu-K ⁇ radiation XRPD spectrum of the IV crystal form.
- Fig. 5 is the Cu-K ⁇ radiation XRPD spectrum of V crystal form.
- Fig. 6 is the Cu-K ⁇ radiation XRPD spectrum of VI crystal form.
- Compound (Ia) in the following examples refers to Provided by Shanghai Jiatan Pharmaceutical Technology Co., Ltd., prepared according to Example 176 of CN 105461712 A.
- the XRPD pattern analysis data of compound (Ia) are shown in Table 1, Table 2, Table 3, Table 4, Table 5 or Table 6; the XRPD pattern of compound (Ia) is shown in Figure 1, Figure 2, Figure 3, Figure 4, Figure 5 or Figure 6.
- DMSO purchased from Sigma
- Matrigel purchased from Corning
- fetal bovine serum purchased from GIBCO
- PRMI1640 purchased from GIBCO
- trypsin purchased from GIBCO
- INS-1 cells purchased from Shanghai Fuxiang Biotechnology Co., Ltd.
- Experimental model rat-derived insulinoma INS-1 cell line subcutaneously allografted female BALB/c nude mouse model
- Cell culture In vitro monolayer culture of rat insulinoma INS-1 cells, the culture conditions are RPMI1640 medium plus 10% fetal bovine serum, 100 U/mL penicillin and 100 ⁇ g/mL streptomycin, 37 ° C, 5% CO 2. Cultivate under the condition of 95% relative humidity, passage with trypsin twice a week, when the cells are in the logarithmic growth phase, digest the cells for inoculation.
- mice BALB/c nude mice, female, 7-8 weeks (the age of mice at the time of tumor cell inoculation), weighing 19-23 g, provided by Jiangsu Jicui Yaokang Biotechnology Co., Ltd.
- Tumor inoculation Experimental mice were subcutaneously inoculated with 10 7 /mouse INS-1 cells, the cells were resuspended in 1:1 PBS and Matrigel, and the tumor growth was observed regularly. The day of tumor cell inoculation was defined as day 0. The initial blood glucose of 5 animals was randomly measured before inoculation. Afterwards, the blood glucose of 5 animals was randomly measured every week to monitor the blood glucose changes during the growth of insulinoma. After the blood glucose of some animals was observed to drop to ⁇ 4mM, the blood glucose at the tip of the tail of all animals was taken to measure the blood glucose. Grouped according to the blood sugar level, and started administration, the average blood sugar level of each group before administration was 5.36mM (the 23rd day).
- test compound (Ia) (0.2mg/kg) group, test compound (Ia) (0.1mg/kg) group, test compound (Ia) (0.05mg/kg) group and Everolimus (1mg/kg) group, 5 rats in each group, the test compound (Ia) and Everolimus were administered orally by gavage, once a day, see Table 7 for details. Three days after administration, the blood glucose curve was analyzed.
- Vehicle one 1% DMSO + 99% (1% methylcellulose aqueous solution), the percentages are volume percentages.
- the average tumor volume of mice in the vehicle control group was 2465.09mm 3
- the second group of compound (Ia), 0.2mg/kg treatment group, the third group of compound ( Ia), 0.1mg/kg treatment group, the fourth compound (Ia), 0.05mg/kg treatment group and the fifth Everolimus, 1mg/kg treatment group mean tumor volumes were 1902.62mm 3 , 2113.97mm 3 , 2233.47mm 3 and 2162.24mm 3 .
- mice The tumor volumes of mice in each treatment group and control group are shown in Table 10-11 (tumor volume growth curves of mice in each treatment group and control group).
- TGI tumor growth rate
- compound (Ia) can significantly increase the blood glucose and inhibit tumor growth in mice.
- compound (Ia) compared with Everolimus, compound (Ia) has a stronger effect on improving blood sugar level and inhibiting tumor growth in mice.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Diabetes (AREA)
- Emergency Medicine (AREA)
- Endocrinology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Epidemiology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
本申请要求申请日为2021年9月27日的中国专利申请2021111383820的优先权。本申请引用上述中国专利申请的全文。This application claims the priority of Chinese patent application 2021111383820 with a filing date of September 27, 2021. This application cites the full text of the above-mentioned Chinese patent application.
本发明涉及一种吡啶并[1,2-a]嘧啶酮类化合物的应用。The invention relates to the application of a pyrido[1,2-a]pyrimidinone compound.
胰岛素瘤是非常罕见的神经内分泌肿瘤,其临床表现为胰岛素过多导致低血糖综合征。尽管胰岛素瘤的恶性程度较低,但由胰岛素瘤引起的长期低血糖对人体重要器官可造成不可逆损伤,严重影响了患者的生活质量乃至威胁生命。如胰岛素瘤致低血糖症状有:思维迟钝、言语行为异常、肢体瘫痪,最后昏迷、死亡。控制低血糖综合征以及抑制肿瘤生长是治疗胰岛素瘤的目标。Insulinomas are very rare neuroendocrine tumors characterized clinically by hyperinsulinemia leading to hypoglycemic syndrome. Although insulinomas are less malignant, long-term hypoglycemia caused by insulinomas can cause irreversible damage to vital organs of the human body, seriously affecting the quality of life of patients and even threatening their lives. For example, the symptoms of hypoglycemia caused by insulinoma include: slow thinking, abnormal speech behavior, limb paralysis, and finally coma and death. Controlling hypoglycemic syndrome and inhibiting tumor growth are the goals of treating insulinoma.
局限期神经内分泌肿瘤可以手术切除,但是,约半数神经内分泌肿瘤诊断时已为局部晚期或出现远处转移,根治性手术切除困难,需要药物治疗以延缓肿瘤进展,延长患者的生存期。Limited-stage neuroendocrine tumors can be surgically resected. However, about half of neuroendocrine tumors are diagnosed as locally advanced or have distant metastases. Radical surgical resection is difficult, and drug therapy is required to delay tumor progression and prolong patient survival.
对于胰岛素瘤来说,绝大多数的患者病灶小。据报道,24%的胰岛素瘤患者的病灶小于1cm,42%的患者病灶在1-2cm之间,30%的患者病灶在2-3cm之间,仅有4%患者的病灶大于3cm(参考文献:Surgical approach to insulinomas.Assessing the need for preoperative localization)。病灶过小无疑增加了局部切除手术的难度以及加大了对手术设备的要求。手术后并发症不可忽视,包括糖尿病(参考文献:Incidence and management of postoperative hyperglycemia in patients undergoing insulinoma resection;Overtly manifested diabetes mellitus after resection of insulinoma.Intern.),胰瘘(参考文献: Diagnosis and management of insulinoma;Risk factors,predictors and prevention of pancreatic fistula formation after pancreatoduodenectomy.)等。For insulinomas, most patients have small lesions. It has been reported that 24% of insulinoma patients had lesions smaller than 1 cm, 42% had lesions between 1-2 cm, 30% had lesions between 2-3 cm, and only 4% had lesions larger than 3 cm (Ref. : Surgical approach to insulinomas. Assessing the need for preoperative localization). Too small lesions will undoubtedly increase the difficulty of local resection and increase the requirements for surgical equipment. Postoperative complications should not be ignored, including diabetes (reference: Incidence and management of postoperative hyperglycemia in patients undergoing insulinoma resection; Overtly manifested diabetes mellitus after resection of insulinoma. Intern.), pancreatic fistula (reference: Diagnosis and management of insulinoma; Risk factors, predictors and prevention of pancreatic fistula formation after pancreatoduodenectomy.), etc.
此外有些病灶位于胰头深部靠近胰管或胰尾末段深部,手术后胰管损伤和瘘管形成的风险较高(参考文献:Beta-islet cell tumors of the pancreas:results of a study on 1,067cases)。对于无法手术的患者,如拒绝手术者、高龄患者、一般情况较差者、已多次腹部手术者或因其他原因术后并发症风险增加者(参考文献:Diagnosis and management of insulinoma),治疗手段欠缺。In addition, some lesions are located deep in the head of the pancreas close to the pancreatic duct or in the deep end of the tail of the pancreas, and the risk of pancreatic duct injury and fistula formation after surgery is high (reference: Beta-islet cell tumors of the pancreas: results of a study on 1,067 cases). For inoperable patients, such as those who refuse surgery, those who are elderly, those who are in poor general condition, those who have had multiple abdominal surgeries, or those who are at increased risk of postoperative complications for other reasons (reference: Diagnosis and management of insulinoma), treatment options lacking.
FDA在2011年5月批准依维莫司(Everolimus)用来治疗胰腺来源进展性神经内分泌肿瘤。依维莫司在多个临床实验中被报道具有缓解胰岛素瘤导致的低血糖效应,然而仍有报道表明依维莫司无法改善胰岛素瘤患者低血糖的症状(参考文献Management of Intractable Hypoglycemia With Yttirum-90 Radioembolization in a Patient With Malignant Insulinoma)。The FDA approved Everolimus in May 2011 for the treatment of progressive neuroendocrine tumors of pancreatic origin. Everolimus has been reported to alleviate the hypoglycemia effect caused by insulinoma in many clinical trials, but there are still reports that Everolimus cannot improve the symptoms of hypoglycemia in patients with insulinoma (reference Management of Intractable Hypoglycemia With Yttirum- 90 Radioembolization in a Patient With Malignant Insulinoma).
综上所述,开发出控糖效果好和抑癌作用强的药物来治疗胰腺神经内分泌肿瘤和控制血糖综合征是目前亟需解决的问题。In summary, it is an urgent problem to develop drugs with good glucose control effect and strong tumor suppressor effect to treat pancreatic neuroendocrine tumors and control blood sugar syndrome.
发明内容Contents of the invention
本发明所要解决的技术问题是现有的用于治疗胰腺神经内分泌肿瘤的药物结构较为单一,且无法很好地改善患者的低血糖症状。为此,本发明提供了一种吡啶并[1,2-a]嘧啶酮类化合物的应用。该化合物能够抑制胰腺神经内分泌肿瘤的生长,提高血糖水平。The technical problem to be solved by the present invention is that the existing drug structure for treating pancreatic neuroendocrine tumors is relatively single, and cannot well improve the hypoglycemia symptoms of patients. Therefore, the present invention provides an application of pyrido[1,2-a]pyrimidinone compounds. The compound was able to inhibit the growth of pancreatic neuroendocrine tumors and increase blood sugar levels.
本发明提供了一种物质X在制备药物中的应用,所述的物质X为如式I所示的化合物、其药学上可接受的盐、其溶剂合物或其药学上可接受的盐的溶剂合物;所述的药物为用于治疗胰腺神经内分泌肿瘤的药物;The present invention provides a kind of application of substance X in the preparation of medicine, and said substance X is the compound as shown in formula I, its pharmaceutically acceptable salt, its solvate or its pharmaceutically acceptable salt A solvate; the drug is a drug for treating pancreatic neuroendocrine tumors;
其中,E为任选被R 3取代的C 1-6烷基、任选被R 3取代的C 3-10环烃基或任选被R 3取代的C 3-10杂环烃基;所述C 3-10杂环烃基中的“杂”表示杂原子或杂原子团,独立地为-C(=O)N(R a)-、-N(R a)-、-C(=NR a)-、-S(=O) 2N(R a)-、-S(=O)N(R a)-、-O-、-S-、-C(=O)O-、-C(=O)-、-C(=S)-、-S(=O)-、-S(=O) 2-或-N(R a)C(=O)N(R a)-; Wherein, E is a C 1-6 alkyl group optionally substituted by R 3 , a C 3-10 cycloalkyl group optionally substituted by R 3 or a C 3-10 heterocycloalkyl group optionally substituted by R 3 ; the C "Hetero" in 3-10 heterocyclic hydrocarbon group means heteroatom or heteroatom group, independently -C(=O)N(R a )-, -N(R a )-, -C(=NR a )- , -S(=O) 2 N(R a )-, -S(=O)N(R a )-, -O-, -S-, -C(=O)O-, -C(=O )-, -C(=S)-, -S(=O)-, -S(=O) 2 - or -N(R a )C(=O)N(R a )-;
L为-C(R 3)(R 3)-、-C(=O)N(R a)-、-N(R a)-、-C(=NR a)-、-S(=O) 2N(R a)-、-S(=O)N(R a)-、-O-、-S-、-C(=O)O-、-C(=O)-、-C(=S)-、-S(=O)-、-S(=O) 2-或-N(R a)C(=O)N(R a)-; L is -C(R 3 )(R 3 )-, -C(=O)N(R a )-, -N(R a )-, -C(=NR a )-, -S(=O) 2 N(R a )-, -S(=O)N(R a )-, -O-, -S-, -C(=O)O-, -C(=O)-, -C(= S)-, -S(=O)-, -S(=O) 2 - or -N(R a )C(=O)N(R a )-;
Q为单键或-C(R 3)(R 3)-; Q is a single bond or -C(R 3 )(R 3 )-;
A为N或C(R 3); A is N or C (R 3 );
X、Y和Z中的0或1个为N,其余为C(R 3); 0 or 1 of X, Y and Z is N, and the rest are C (R 3 );
m 1为0、1、2或3; m 1 is 0, 1, 2 or 3;
R 1、R 2和R 3独立地为H、F、Cl、Br、I、CN、-OR a、-N(R b)(R c)、任选被R d取代的C 1-3烷基、 R 1 , R 2 and R 3 are independently H, F, Cl, Br, I, CN, -OR a , -N(R b )(R c ), C 1-3 alkane optionally substituted by R d base,
D 1独立地为单键、-C(R e)(R e)-、-C(=O)N(R a)-、-N(R a)-、-C(=NR a)-、-S(=O) 2N(R a)-、-S(=O)N(R a)-、-O-、-S-、-C(=O)O-、-C(=O)-、-C(=S)-、-S(=O)-、-S(=O) 2-或-N(R a)C(=O)N(R a)-; D 1 is independently a single bond, -C(R e )(R e )-, -C(=O)N(R a )-, -N(R a )-, -C(=NR a )-, -S(=O) 2 N(R a )-, -S(=O)N(R a )-, -O-, -S-, -C(=O)O-, -C(=O) -, -C(=S)-, -S(=O)-, -S(=O) 2 - or -N(R a )C(=O)N(R a )-;
n独立地为1、2、3、4、5或6;n is independently 1, 2, 3, 4, 5 or 6;
D 2独立地为-C(R a)(R a)-; D 2 is independently -C(R a )(R a )-;
R a、R b和R c独立地为H、任选被R d取代的C 1-6烷基或任选被R d取代的C 3-6环烷基; R a , R b and R c are independently H, C 1-6 alkyl optionally substituted by R d or C 3-6 cycloalkyl optionally substituted by R d ;
R e为H、任选被R d取代的C 1-6烷基、任选被R d取代的C 1-6烷氧基、任选被R d取代的C 3-6环烷基或任选被R d取代的C 3-6环烷氧基; R e is H, C 1-6 alkyl optionally substituted by R d , C 1-6 alkoxy optionally substituted by R d , C 3-6 cycloalkyl optionally substituted by R d , or any A C 3-6 cycloalkoxy group substituted by R d ;
R d的数目独立地为1、2或3;R d独立地为F、Cl、Br、I、CN、-OH、-CHO、-COOH、CH 3-、CF 3-、CH 3O-或CH 3CH 2O-; The number of Rd is independently 1, 2 or 3; Rd is independently F, Cl, Br, I, CN, -OH, -CHO, -COOH, CH3- , CF3- , CH3O- or CH3CH2O- ;
任选地,任意两个R 1之间、同一个D 2中的R a与R a之间、两个D 2之间、或R a与一个D 2之间共同连接到同一碳原子或氧原子上形成一个或两个3、4、5或6元碳环或氧杂环,其中氧原子的数目为1或2。 Optionally, between any two R 1 , between R a and R a in the same D 2 , between two D 2 , or between R a and a D 2 are jointly connected to the same carbon atom or oxygen One or two 3, 4, 5 or 6-membered carbocyclic rings or oxygen heterocyclic rings are formed on the atoms, and the number of oxygen atoms is 1 or 2.
在所述的应用中,所述的如式I所示的化合物中某些基团的定义可如下所述,其余基团的定义如其他任一方案所述:In the described application, the definition of certain groups in the described compound shown in formula I can be as follows, and the definition of all the other groups is as described in any other scheme:
E为被R 3取代的C 1-6烷基或被R 3取代的C 3-6环烷基,R 3的数目独立地为1、2或3。 E is C 1-6 alkyl substituted by R 3 or C 3-6 cycloalkyl substituted by R 3 , and the number of R 3 is 1, 2 or 3 independently.
在所述的应用中,所述的如式I所示的化合物中某些基团的定义可如下所述,其余基团的定义如其他任一方案所述:In the described application, the definition of certain groups in the described compound shown in formula I can be as follows, and the definition of all the other groups is as described in any other scheme:
E为C 1-6烷基或C 3-6环烷基。 E is C 1-6 alkyl or C 3-6 cycloalkyl.
在所述的应用中,所述的如式I所示的化合物中某些基团的定义可如下所述,其余基团的定义如其他任一方案所述:In the described application, the definition of certain groups in the described compound shown in formula I can be as follows, and the definition of all the other groups is as described in any other scheme:
E为 E is
其中,in,
G 1~5中的0、1、2或3个为N,其余为C(R 3); 0, 1, 2 or 3 of G 1 to 5 are N, and the rest are C (R 3 );
G 6为-C(R 3)(R 3)-、-C(=O)N(R 3)-、-N(R 3)-、-C(=NR 3)-、-S(=O) 2N(R 3)-、- S(=O)N(R 3)-、-O-、-S-、-C(=O)O-、-C(=O)-、-C(=S)-、-S(=O)-、-S(=O) 2-或-N(R 3)C(=O)N(R 3)-; G 6 is -C(R 3 )(R 3 )-, -C(=O)N(R 3 )-, -N(R 3 )-, -C(=NR 3 )-, -S(=O ) 2 N(R 3 )-, -S(=O)N(R 3 )-, -O-, -S-, -C(=O)O-, -C(=O)-, -C( =S)-, -S(=O)-, -S(=O) 2 - or -N(R 3 )C(=O)N(R 3 )-;
G 7~9中的0、1或2个为N,其余为C(R 3); 0, 1 or 2 of G 7-9 are N, and the rest are C (R 3 );
G 10~16中的0、1、2、3或4个为N,其余为C(R 3); 0, 1, 2, 3 or 4 of G 10-16 are N, and the rest are C (R 3 );
G 17为N或者C(R 3); G 17 is N or C (R 3 );
G 18~22中的0、1、2或3个独立地为-C(=O)N(R 3)-、-N(R 3)-、-C(=NR 3)-、-S(=O) 2N(R 3)-、-S(=O)N(R 3)-、-O-、-S-、-C(=O)O-、-C(=O)-、-C(=S)-、-S(=O)-、-S(=O) 2-或-N(R 3)C(=O)N(R 3)-,其余为-C(R 3)(R 3)-。 0, 1, 2 or 3 of G 18-22 are independently -C(=O)N(R 3 )-, -N(R 3 )-, -C(=NR 3 )-, -S( =O) 2 N(R 3 )-, -S(=O)N(R 3 )-, -O-, -S-, -C(=O)O-, -C(=O)-, - C(=S)-, -S(=O)-, -S(=O) 2 - or -N(R 3 )C(=O)N(R 3 )-, the rest are -C(R 3 ) (R 3 )-.
在所述的应用中,所述的物质X可为如式(Ia)所示的化合物、其药学上可接受的盐、其溶剂合物或其药学上可接受的盐的溶剂合物;In said application, said substance X can be a compound represented by formula (Ia), its pharmaceutically acceptable salt, its solvate or its pharmaceutically acceptable salt solvate;
在所述的应用中,所述的如式(Ia)所示的化合物的XRPD图谱解析数据可如表1(Ⅰ晶型)、表2(II晶型)、表3(III晶型)、表4(IV晶型)、表5(V晶型)或表6(VI晶型)所示:In the application, the XRPD spectrum analysis data of the compound represented by formula (Ia) can be shown in Table 1 (crystal form I), table 2 (crystal form II), table 3 (crystal form III), Shown in Table 4 (Form IV), Table 5 (Form V) or Table 6 (Form VI):
表1Table 1
表2Table 2
表3table 3
表4Table 4
表5table 5
表6Table 6
在所述的应用中,所述的如式(Ia)所示的化合物的XRPD图谱可如图1、图2、图3、图4、图5或图6所示。In the application, the XRPD pattern of the compound represented by formula (Ia) can be as shown in Figure 1 , Figure 2 , Figure 3 , Figure 4 , Figure 5 or Figure 6 .
在所述的应用中,所述的胰腺神经内分泌肿瘤可为胰岛素瘤、胺前体摄取和脱羧细胞瘤、血管活性肠肽瘤、胰高血糖素瘤、胰多肽瘤或胃泌素瘤。In said application, said pancreatic neuroendocrine tumor may be insulinoma, amine precursor uptake and decarboxylation cell tumor, vasoactive intestinal peptide tumor, glucagon tumor, pancreatic polypeptide tumor or gastrinoma.
在所述的应用中,所述的药物为用于治疗胰腺神经内分泌肿瘤并提高血糖浓度的药物。In said application, said drug is a drug used for treating pancreatic neuroendocrine tumors and increasing blood sugar concentration.
另一方面,本发明提供一种治疗胰腺神经内分泌肿瘤的方法,其包括向 有此需要的患者给予治疗有效量的如本文所述的物质X。In another aspect, the present invention provides a method of treating pancreatic neuroendocrine tumors comprising administering to a patient in need thereof a therapeutically effective amount of substance X as described herein.
在所述的方法中,所述的胰腺神经内分泌肿瘤可为胰岛素瘤、胺前体摄取和脱羧细胞瘤、血管活性肠肽瘤、胰高血糖素瘤、胰多肽瘤或胃泌素瘤。In the method, the pancreatic neuroendocrine tumor may be an insulinoma, an amine precursor uptake and decarboxylation cell tumor, a vasoactive intestinal peptide tumor, a glucagon tumor, a pancreatic polypeptide tumor, or a gastrinoma.
在所述的应用和方法中:In said uses and methods:
物质X的施用方案(包括施用途径、施用剂量、施用间隔等)可以由本领域技术人员根据需要进行调整,以提供最优的治疗效果。The administration regimen of substance X (including administration route, administration dose, administration interval, etc.) can be adjusted by those skilled in the art as needed to provide the optimal therapeutic effect.
所述物质X可以采用本领域中任何合适的途径施用,包括口服、注射(例如静脉、肌肉、皮下)等。The substance X can be administered by any suitable route known in the art, including oral, injection (eg intravenous, intramuscular, subcutaneous) and the like.
在一些实施方案中,所述物质X经口服施用。In some embodiments, the substance X is administered orally.
所述物质X可根据患者的体重来施用,非限制性实例范围可以为0.01-1mg/kg(指单次剂量),例如0.01mg/kg、0.05mg/kg、0.1mg/kg、0.15mg/kg、0.2mg/kg、0.25mg/kg、0.3mg/kg、0.35mg/kg、0.4mg/kg、0.45mg/kg、0.5mg/kg、0.55mg/kg、0.6mg/kg、0.7mg/kg、0.8mg/kg、0.9mg/kg或1mg/kg。The substance X can be administered according to the weight of the patient, a non-limiting example range can be 0.01-1 mg/kg (referring to a single dose), such as 0.01 mg/kg, 0.05 mg/kg, 0.1 mg/kg, 0.15 mg/kg kg, 0.2mg/kg, 0.25mg/kg, 0.3mg/kg, 0.35mg/kg, 0.4mg/kg, 0.45mg/kg, 0.5mg/kg, 0.55mg/kg, 0.6mg/kg, 0.7mg/kg kg, 0.8mg/kg, 0.9mg/kg or 1mg/kg.
在一些实施方案中,化合物I的剂量为0.05-0.2mg/kg,例如0.05mg/kg、0.1mg/kg或0.2mg/kg。In some embodiments, the dose of Compound I is 0.05-0.2 mg/kg, such as 0.05 mg/kg, 0.1 mg/kg or 0.2 mg/kg.
所述物质X的上述剂量可以按照QD(一天一次)、QOD(间隔一天)或QW(每周一次)的频次施用。The above doses of the substance X may be administered according to the frequency of QD (once a day), QOD (one day apart) or QW (once a week).
在一些实施方案中,所述物质X按照QD频次施用。In some embodiments, the substance X is administered on a QD frequency.
在一些实施方案中,所述物质X经口服按照上述剂量、频次施用。In some embodiments, the substance X is administered orally according to the dosage and frequency mentioned above.
在一些实施方案中,所述物质X经口服,按照0.2mg/kg,QD施用。In some embodiments, the substance X is administered orally at 0.2 mg/kg, QD.
在一些实施方案中,所述物质X经口服,按照0.2mg/kg,QD施用,所述的物质X为如式(Ia)所示的化合物。In some embodiments, the substance X is administered orally at 0.2 mg/kg, QD, and the substance X is a compound represented by formula (Ia).
所述物质X也可以固定剂量施用于患者,即给予患者固定或预定量的剂量。固定剂量(指单次剂量)的非限制性实例范围可以为0.01-50mg,例如0.01mg、0.1mg、0.5mg、1mg、2mg、3mg、4mg、5mg、6mg、7mg、8mg、9mg、10mg、11mg、12mg、13mg、14mg、15mg、16mg、17mg、 18mg、19mg、20mg、21mg、22mg、23mg、24mg、25mg、26mg、27mg、28mg、29mg、30mg、31mg、32mg、33mg、34mg、35mg、36mg、37mg、38mg、39mg、40mg、41mg、42mg、43mg、44mg、45mg、46mg、47mg、48mg、49mg或50mg。The substance X may also be administered to the patient in fixed doses, ie a fixed or predetermined amount of dose is given to the patient. A non-limiting example of a fixed dose (referred to as a single dose) may range from 0.01 to 50 mg, such as 0.01 mg, 0.1 mg, 0.5 mg, 1 mg, 2 mg, 3 mg, 4 mg, 5 mg, 6 mg, 7 mg, 8 mg, 9 mg, 10 mg, 11mg, 12mg, 13mg, 14mg, 15mg, 16mg, 17mg, 18mg, 19mg, 20mg, 21mg, 22mg, 23mg, 24mg, 25mg, 26mg, 27mg, 28mg, 29mg, 30mg, 31mg, 32mg, 33mg, 34mg, 35mg, 36mg, 37mg, 38mg, 39mg, 40mg, 41mg, 42mg, 43mg, 44mg, 45mg, 46mg, 47mg, 48mg, 49mg or 50mg.
所述物质X的上述固定剂量可以QD(一天一次)、QOD(间隔一天)或QW(每周一次)的频次施用。The above-mentioned fixed doses of the substance X may be administered at a frequency of QD (once a day), QOD (once a day) or QW (once a week).
在一些实施方案中,所述物质X按照QD频次施用。In some embodiments, the substance X is administered on a QD frequency.
术语“药学上可接受的盐”是指化合物与药学上可接受的(相对无毒、安全、适合于患者使用)酸或碱反应得到的盐。当化合物中含有相对酸性的官能团时,可以通过在合适的惰性溶剂中用足量的药学上可接受的碱与化合物的游离形式接触的方式获得碱加成盐。药学上可接受的碱加成盐包括但不限于钠盐、钾盐、钙盐、铝盐、镁盐、铋盐、铵盐等。当化合物中含有相对碱性的官能团时,可以通过在合适的惰性溶剂中用足量的药学上可接受的酸与化合物的游离形式接触的方式获得酸加成盐。药学上可接受的酸加成盐包括但不限于盐酸盐、硫酸盐、甲磺酸盐等。具体参见Handbook of Pharmaceutical Salts:Properties,Selection,and Use(P.Heinrich Stahl,2002)。The term "pharmaceutically acceptable salt" refers to a salt obtained by reacting a compound with a pharmaceutically acceptable (relatively non-toxic, safe, and suitable for use by patients) acid or base. When the compound contains relatively acidic functional groups, base addition salts can be obtained by contacting the free form of the compound with a sufficient amount of a pharmaceutically acceptable base in a suitable inert solvent. Pharmaceutically acceptable base addition salts include, but are not limited to, sodium salts, potassium salts, calcium salts, aluminum salts, magnesium salts, bismuth salts, ammonium salts, and the like. When the compound contains relatively basic functional groups, acid addition salts can be obtained by contacting the free form of the compound with a sufficient amount of a pharmaceutically acceptable acid in a suitable inert solvent. Pharmaceutically acceptable acid addition salts include, but are not limited to, hydrochlorides, sulfates, methanesulfonates, and the like. See Handbook of Pharmaceutical Salts: Properties, Selection, and Use (P. Heinrich Stahl, 2002) for details.
术语“溶剂合物”是指化合物与溶剂(包括但不限于:水、甲醇、乙醇等)结合后形成的物质。溶剂合物分为化学计量类溶剂合物和非化学计量类溶剂合物。The term "solvate" refers to a substance formed when a compound is combined with a solvent (including but not limited to: water, methanol, ethanol, etc.). Solvates are divided into stoichiometric solvates and non-stoichiometric solvates.
术语“药学上可接受的盐的溶剂合物”是指化合物与药学上可接受的(相对无毒、安全、适合于患者使用)酸或碱、溶剂(包括但不限于:水、甲醇、乙醇等)结合形成的物质,其中,药学上可接受的盐与上文术语“药学上可接受的盐”的含义相同,溶剂为化学计量的或非化学计量的。药学上可接受的盐的溶剂合物包括但不限于盐酸盐一水合物。The term "solvate of a pharmaceutically acceptable salt" refers to a compound with a pharmaceutically acceptable (relatively non-toxic, safe, and suitable for patient use) acid or base, solvent (including but not limited to: water, methanol, ethanol etc.), wherein the pharmaceutically acceptable salt has the same meaning as the term "pharmaceutically acceptable salt" above, and the solvent is stoichiometric or non-stoichiometric. Solvates of pharmaceutically acceptable salts include, but are not limited to, hydrochloride monohydrate.
在不违背本领域常识的基础上,上述各优选条件,可任意组合,即得本发明各较佳实例。On the basis of not violating common knowledge in the field, the above-mentioned preferred conditions can be combined arbitrarily to obtain preferred examples of the present invention.
本发明所用试剂和原料均市售可得。The reagents and raw materials used in the present invention are all commercially available.
本发明所使用的晶型I晶型、Ⅱ晶型、Ⅲ晶型、Ⅳ晶型、Ⅴ晶型、Ⅵ晶型按照CN201680072859.0记载的方法制备。The crystal forms I, II, III, IV, V and VI used in the present invention are prepared according to the method described in CN201680072859.0.
本发明的积极进步效果在于:该化合物能够抑制胰腺神经内分泌肿瘤的生长,提高血糖水平。The positive progress effect of the present invention lies in that the compound can inhibit the growth of pancreatic neuroendocrine tumors and improve the blood sugar level.
图1为I晶型的Cu-Kα辐射的XRPD谱图。Figure 1 is the Cu-Kα radiation XRPD spectrum of Form I.
图2为Ⅱ晶型的Cu-Kα辐射的XRPD谱图。Fig. 2 is the Cu-Kα radiation XRPD spectrum of the II crystal form.
图3为Ⅲ晶型的Cu-Kα辐射的XRPD谱图。Fig. 3 is the Cu-Kα radiation XRPD spectrum of the III crystal form.
图4为Ⅳ晶型的Cu-Kα辐射的XRPD谱图。Fig. 4 is the Cu-Kα radiation XRPD spectrum of the IV crystal form.
图5为Ⅴ晶型的Cu-Kα辐射的XRPD谱图。Fig. 5 is the Cu-Kα radiation XRPD spectrum of V crystal form.
图6为Ⅵ晶型的Cu-Kα辐射的XRPD谱图。Fig. 6 is the Cu-Kα radiation XRPD spectrum of VI crystal form.
下面通过实施例的方式进一步说明本发明,但并不因此将本发明限制在所述的实施例范围之中。下列实施例中未注明具体条件的实验方法,按照常规方法和条件,或按照商品说明书选择。The present invention is further illustrated below by means of examples, but the present invention is not limited to the scope of the examples. For the experimental methods that do not specify specific conditions in the following examples, select according to conventional methods and conditions, or according to the product instructions.
下述实施例中的化合物(Ⅰa)是指 由上海嘉坦医药科技有限公司提供,按照CN 105461712 A实施例176制备得到。 Compound (Ia) in the following examples refers to Provided by Shanghai Jiatan Pharmaceutical Technology Co., Ltd., prepared according to Example 176 of CN 105461712 A.
化合物(Ⅰa)的XRPD图谱解析数据如表1、表2、表3、表4、表5或表6所示;化合物(Ⅰa)的XRPD图谱如图1、图2、图3、图4、图5或图 6所示。The XRPD pattern analysis data of compound (Ia) are shown in Table 1, Table 2, Table 3, Table 4, Table 5 or Table 6; the XRPD pattern of compound (Ia) is shown in Figure 1, Figure 2, Figure 3, Figure 4, Figure 5 or Figure 6.
实施例1:Example 1:
研究目的:评价测试化合物(Ⅰa)在大鼠来源的胰岛素瘤INS-1皮下异种移植BALB/c雌性裸小鼠低血糖模型中的药效学研究Purpose of the study: To evaluate the pharmacodynamics of the test compound (Ⅰa) in the hypoglycemia model of rat-derived insulinoma INS-1 subcutaneous xenograft BALB/c female nude mice
实验材料:DMSO(购于Sigma)、基质胶(购于Corning)、胎牛血清(购于GIBCO)、PRMI1640(购于GIBCO)、胰酶(购于GIBCO)、INS-1细胞(购于上海富祥生物科技有限公司);Experimental materials: DMSO (purchased from Sigma), Matrigel (purchased from Corning), fetal bovine serum (purchased from GIBCO), PRMI1640 (purchased from GIBCO), trypsin (purchased from GIBCO), INS-1 cells (purchased from Shanghai Fuxiang Biotechnology Co., Ltd.);
实验模型:大鼠源胰岛素瘤INS-1细胞株皮下同种移植雌性BALB/c裸小鼠模型;Experimental model: rat-derived insulinoma INS-1 cell line subcutaneously allografted female BALB/c nude mouse model;
实验方法:experimental method:
细胞培养:大鼠胰岛素瘤INS-1细胞体外单层培养,培养条件为RPMI1640培养基中加10%胎牛血清,100U/mL的青霉素和100μg/mL的链霉素,37℃,5%CO 2,95%相对湿度条件下培养,一周两次用胰酶消化传代,当细胞处于对数生长期时,消化细胞用于接种。 Cell culture: In vitro monolayer culture of rat insulinoma INS-1 cells, the culture conditions are RPMI1640 medium plus 10% fetal bovine serum, 100 U/mL penicillin and 100 μg/mL streptomycin, 37 ° C, 5% CO 2. Cultivate under the condition of 95% relative humidity, passage with trypsin twice a week, when the cells are in the logarithmic growth phase, digest the cells for inoculation.
实验动物:BALB/c裸小鼠,雌性,7-8周(肿瘤细胞接种时的小鼠周龄),体重19-23g,由江苏集萃药康生物科技有限公司提供。Experimental animals: BALB/c nude mice, female, 7-8 weeks (the age of mice at the time of tumor cell inoculation), weighing 19-23 g, provided by Jiangsu Jicui Yaokang Biotechnology Co., Ltd.
肿瘤接种:实验小鼠于皮下接种10
7/鼠的INS-1细胞,细胞重悬在1:1的PBS与基质胶中,定期观察肿瘤生长情况。肿瘤细胞接种当天定义为第0天。接种前随机测定5只动物初始血糖。之后,每周随机测定5只动物的血糖监测胰岛素瘤生长过程中的血糖变化。待观察到部分动物血糖下降至<4mM,取所有动物尾尖血糖测定血糖。根据血糖值分组,并开始给药,给药前每组平均血糖值为5.36mM(第23天)。
Tumor inoculation: Experimental mice were subcutaneously inoculated with 10 7 /mouse INS-1 cells, the cells were resuspended in 1:1 PBS and Matrigel, and the tumor growth was observed regularly. The day of tumor cell inoculation was defined as
实验分组:试验分为溶媒对照组、测试化合物(Ⅰa)(0.2mg/kg)组、测试化合物(Ⅰa)(0.1mg/kg)组、测试化合物(Ⅰa)(0.05mg/kg)组与Everolimus(1mg/kg)组,每组5只,测试化合物(Ⅰa)和Everolimus口服灌胃给药,每天给药一次,具体见表7。给药3天后,分析血糖曲线。Experimental grouping: The test was divided into vehicle control group, test compound (Ia) (0.2mg/kg) group, test compound (Ia) (0.1mg/kg) group, test compound (Ia) (0.05mg/kg) group and Everolimus (1mg/kg) group, 5 rats in each group, the test compound (Ia) and Everolimus were administered orally by gavage, once a day, see Table 7 for details. Three days after administration, the blood glucose curve was analyzed.
表7Table 7
备注:Remark:
溶媒一:1%DMSO+99%(1%甲基纤维素水溶液),所述百分数为体积百分数。Vehicle one: 1% DMSO + 99% (1% methylcellulose aqueous solution), the percentages are volume percentages.
实验结果:Experimental results:
1)肿瘤接种后第26天(给药后第3天),溶媒对照组小鼠平均血糖为3.90mmol/L,第二组化合物(Ⅰa),0.2mg/kg治疗组,第三组化合物(Ⅰa),0.1mg/kg治疗组和第四组化合物(Ⅰa),0.05mg/kg治疗组平均血糖分别为9.28mmol/L、7.68mmol/L和6.32mmol/L。相较对照组,化合物(Ⅰa)高、中、低剂量组都表现明显地升糖作用。并且相对于对照组,化合物(Ⅰa)高、中、低剂量组在统计学上存在显著性差异(p<0.001、p=0.0011和p=0.0366)。第五组Everolimus,1mg/kg治疗组平均血糖为5.02mmol/L,相较对照组统计学上无显著性差异(p=0.5165)。各治疗组和对照组小鼠血糖情况见表8和表 9(各治疗组和对照组小鼠血糖情况,表中各数字的单位为mmol/L)。1) On the 26th day after tumor inoculation (the 3rd day after administration), the average blood glucose of mice in the vehicle control group was 3.90mmol/L, the second group of compound (Ia), 0.2mg/kg treatment group, the third group of compound ( Ⅰa), the 0.1mg/kg treatment group and the fourth compound (Ⅰa), 0.05mg/kg treatment group mean blood glucose were 9.28mmol/L, 7.68mmol/L and 6.32mmol/L respectively. Compared with the control group, the high, middle and low dose groups of compound (Ia) all showed obvious blood sugar-increasing effect. And compared with the control group, there are statistically significant differences in the compound (Ia) high, middle and low dose groups (p<0.001, p=0.0011 and p=0.0366). The fifth group Everolimus, 1mg/kg treatment group average blood sugar was 5.02mmol/L, compared with the control group, there was no statistically significant difference (p=0.5165). See Table 8 and Table 9 for the blood glucose status of the mice in each treatment group and control group (the blood glucose status of the mice in each treatment group and control group, the unit of each number in the table is mmol/L).
表8各治疗组和对照组小鼠血糖情况Table 8 Blood glucose situation of mice in each treatment group and control group
表9各治疗组和对照组小鼠血糖情况Table 9 Blood glucose situation of mice in each treatment group and control group
2)肿瘤接种后第26天(给药后第3天),溶媒对照组小鼠平均肿瘤体积为2465.09mm 3,第二组化合物(Ⅰa),0.2mg/kg治疗组,第三组化合物(Ⅰa),0.1mg/kg治疗组,第四组化合物(Ⅰa),0.05mg/kg治疗组和第五组Everolimus,1mg/kg治疗组平均肿瘤体积分别为1902.62mm 3,2113.97mm 3,2233.47mm 3和2162.24mm 3。相较对照组,仅第二组化合物(Ⅰa),0.2mg/kg治疗组对肿瘤有显著抑制作用,TGI为82%,并且统计学上有显著性差异(p<0.05)。Everolimus组与溶媒对照组相比P=0.86,没有抑瘤作用。 2) On the 26th day after tumor inoculation (the 3rd day after administration), the average tumor volume of mice in the vehicle control group was 2465.09mm 3 , the second group of compound (Ia), 0.2mg/kg treatment group, the third group of compound ( Ⅰa), 0.1mg/kg treatment group, the fourth compound (Ⅰa), 0.05mg/kg treatment group and the fifth Everolimus, 1mg/kg treatment group mean tumor volumes were 1902.62mm 3 , 2113.97mm 3 , 2233.47mm 3 and 2162.24mm 3 . Compared with the control group, only the second group of compound (Ia), the 0.2mg/kg treatment group had a significant inhibitory effect on the tumor, with a TGI of 82%, and there was a statistically significant difference (p<0.05). Compared with the vehicle control group, Everolimus group had P=0.86, showing no tumor-inhibiting effect.
各治疗组和对照组小鼠肿瘤体积情况见表10-11(各治疗组和对照组小鼠肿瘤体积增长曲线)。The tumor volumes of mice in each treatment group and control group are shown in Table 10-11 (tumor volume growth curves of mice in each treatment group and control group).
表10各治疗组和对照组小鼠肿瘤体积增长曲线Table 10 Tumor volume growth curves of mice in each treatment group and control group
表11各治疗组和对照组小鼠肿瘤体积增长曲线Table 11 Tumor volume growth curves of mice in each treatment group and control group
实验结论:给药后3天后,相较对照组(血糖为3.90mmol/L),化合物(Ⅰa)高(0.2mg/kg)、中(0.1mg/kg)、低(0.05mg/kg)剂量组都表现明显地升糖作用,平均血糖分别为9.28mmol/L、7.68mmol/L和6.32mmol/L。而Everolimus,1mg/kg治疗组平均血糖为5.02mmol/L。Experimental conclusion: 3 days after administration, compared with the control group (blood sugar is 3.90mmol/L), compound (Ia) high (0.2mg/kg), medium (0.1mg/kg), low (0.05mg/kg) dose Both groups showed obvious blood sugar-raising effects, with average blood sugar levels of 9.28mmol/L, 7.68mmol/L and 6.32mmol/L, respectively. The Everolimus, 1mg/kg treatment group average blood sugar was 5.02mmol/L.
给药后3天后,化合物(Ⅰa)高(0.2mg/kg)和中(0.1mg/kg)剂量组的相对肿瘤增殖率(TGI)分别为82%和64%。而Everolimus,1mg/kg治疗组的TGI为33%。Three days after administration, the relative tumor growth rate (TGI) of compound (Ia) high (0.2 mg/kg) and medium (0.1 mg/kg) dose groups were 82% and 64%, respectively. And Everolimus, the TGI of 1mg/kg treatment group was 33%.
在胰岛素瘤INS-1异种移植小鼠低血糖模型中,化合物(Ⅰa)可显著提升小鼠的血糖和抑制肿瘤生长。此外相对于依维莫司(Everolimus),化合物(Ⅰa)提高小鼠的血糖能力和抑制肿瘤生长的效果更强。In the hypoglycemia model of insulinoma INS-1 xenograft mice, compound (Ia) can significantly increase the blood glucose and inhibit tumor growth in mice. In addition, compared with Everolimus, compound (Ia) has a stronger effect on improving blood sugar level and inhibiting tumor growth in mice.
Claims (12)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111138382 | 2021-09-27 | ||
| CN202111138382.0 | 2021-09-27 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2023046182A1 true WO2023046182A1 (en) | 2023-03-30 |
Family
ID=85720143
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2022/121443 Ceased WO2023046182A1 (en) | 2021-09-27 | 2022-09-26 | Application of pyrido[1,2-a]pyrimidone compound |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN115869320A (en) |
| WO (1) | WO2023046182A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP4282866A4 (en) * | 2021-01-25 | 2024-07-03 | Guangzhou Joyo Pharmatech Co., Ltd | USE OF PYRIDO[1,2-A PYRIMIDINONE ANALOGUES |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008144463A1 (en) * | 2007-05-18 | 2008-11-27 | Smithkline Beecham Corporation | Quinoline derivatives as pi3 kinase inhibitors |
| CN105461711A (en) * | 2014-06-17 | 2016-04-06 | 南京明德新药研发股份有限公司 | Pyrido[1,2-a]pyrimidone analogs as PI3K inhibitors |
| CN105461712A (en) * | 2014-06-17 | 2016-04-06 | 南京明德新药研发股份有限公司 | Pyrido[1,2-a]pyrimidone analogs as mTOR/PI3K inhibitors |
-
2022
- 2022-09-26 WO PCT/CN2022/121443 patent/WO2023046182A1/en not_active Ceased
- 2022-09-26 CN CN202211177234.4A patent/CN115869320A/en active Pending
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008144463A1 (en) * | 2007-05-18 | 2008-11-27 | Smithkline Beecham Corporation | Quinoline derivatives as pi3 kinase inhibitors |
| CN105461711A (en) * | 2014-06-17 | 2016-04-06 | 南京明德新药研发股份有限公司 | Pyrido[1,2-a]pyrimidone analogs as PI3K inhibitors |
| CN105461712A (en) * | 2014-06-17 | 2016-04-06 | 南京明德新药研发股份有限公司 | Pyrido[1,2-a]pyrimidone analogs as mTOR/PI3K inhibitors |
Non-Patent Citations (1)
| Title |
|---|
| HAN XU, WEN-HUI LOU: "Advances in Research on Genotyping and the Molecular Mechanism of Pancreatic Neuroendocrine Neoplasias", MEDICAL JOURNAL OF PEKING UNION MEDICAL COLLEGE HOSPITAL, vol. 11, no. 4, 31 July 2020 (2020-07-31), pages 377 - 382, XP093054273, ISSN: 1674-9081, DOI: 10.3969/j.issn.1674-9081.2020.04.004 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP4282866A4 (en) * | 2021-01-25 | 2024-07-03 | Guangzhou Joyo Pharmatech Co., Ltd | USE OF PYRIDO[1,2-A PYRIMIDINONE ANALOGUES |
Also Published As
| Publication number | Publication date |
|---|---|
| CN115869320A (en) | 2023-03-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2023179764A5 (en) | ||
| US20090105298A1 (en) | Pharmaceutical composition for therapy of interstitial cystitis | |
| ES2655455T3 (en) | Compositions for the treatment of hypertension and / or fibrosis | |
| WO2023046182A1 (en) | Application of pyrido[1,2-a]pyrimidone compound | |
| CN101631558A (en) | Exenatide and dalargin-based drugs for the treatment of diabetes, uses and modes of treatment thereof | |
| KR20160003652A (en) | Methods and compositions for gamma-glutamyl cycle modulation | |
| EP4349335A1 (en) | Use of 5-nitro-8-hydroxyquinoline | |
| WO2004002473A1 (en) | Drug composition for blood sugar control | |
| JP2549480B2 (en) | Urinary disorder improving agent | |
| CN111803489A (en) | Application of michelia lactone and derivatives thereof in treatment of pituitary adenoma | |
| TW200539857A (en) | Methods for treating multiple sclerosis and pharmaceutical compositions therefor | |
| CN116925015A (en) | Active substances for inhibiting premature ovarian failure and preparation methods thereof | |
| Foldes et al. | Use of mytolon chloride with pentothal sodium and nitrous oxide-oxygen for abdominal surgery. | |
| US20040132803A1 (en) | Agent for the treatment of overactive bladder | |
| JP2015134732A (en) | Vascular hyperpermeability inhibitor | |
| WO2019164010A1 (en) | Antitumor agent for bladder cancer and method for treating bladder cancer | |
| Asai | Use of the laryngeal mask for exchange of orotracheal tubes | |
| JP3007097B2 (en) | Hypnotic composition | |
| CN119033787A (en) | Application of afatinib in preparation of medicines for treating pulmonary fibrosis | |
| JPS5973522A (en) | Remedy for anoxia of cerebral neurocyte | |
| WO2023125536A1 (en) | Compound for inhibiting vascular endothelial growth and use thereof | |
| CN121102192A (en) | Application of ephedrine rupestonic acid ester in treating tumor | |
| JP2006213645A (en) | A pharmaceutical comprising a combination of geranylgeranylacetone and an anticancer agent | |
| CN120420312A (en) | Application of benzyl phenyl ether compound | |
| WO2023035200A1 (en) | Application of pentafluorite in preparation of drug for treating endometrial cancer |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22872225 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 32PN | Ep: public notification in the ep bulletin as address of the adressee cannot be established |
Free format text: NOTING OF LOSS OF RIGHTS PURSUANT TO RULE 112(1) EPC (EPO FORM 1205A DATED 02.08.2024) |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 22872225 Country of ref document: EP Kind code of ref document: A1 |