WO2022248870A1 - Combination therapies for treating cancer - Google Patents
Combination therapies for treating cancer Download PDFInfo
- Publication number
- WO2022248870A1 WO2022248870A1 PCT/GB2022/051348 GB2022051348W WO2022248870A1 WO 2022248870 A1 WO2022248870 A1 WO 2022248870A1 GB 2022051348 W GB2022051348 W GB 2022051348W WO 2022248870 A1 WO2022248870 A1 WO 2022248870A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- combination
- antigen binding
- binding protein
- bcma
- cancer
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
- C07K16/2809—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily against the T-cell receptor (TcR)-CD3 complex
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
- C07K16/283—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily against Fc-receptors, e.g. CD16, CD32, CD64
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2878—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the NGF-receptor/TNF-receptor superfamily, e.g. CD27, CD30, CD40, CD95
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/31—Immunoglobulins specific features characterized by aspects of specificity or valency multispecific
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/40—Immunoglobulins specific features characterized by post-translational modification
- C07K2317/41—Glycosylation, sialylation, or fucosylation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/55—Fusion polypeptide containing a fusion with a toxin, e.g. diphteria toxin
Definitions
- this disclosure provides methods and materials for using one or more antibody-drug conjugates (ADCs) and one or more T cell engagers for treating a mammal (e.g., a human) having cancer.
- ADCs antibody-drug conjugates
- T cell engagers for treating a mammal (e.g., a human) having cancer.
- the disclosure further provides methods and materials for using one or more antigen binding proteins (for example anti- B-cell maturation antigen (BCMA) antigen binding protein) and one or more T cell engagers for treating a subject having cancer.
- BCMA anti- B-cell maturation antigen binding protein
- this disclosure provides methods and materials for using one or more molecules where each molecule includes: (i) an anti-BCMA antigen binding protein or ADC having binding specificity for a BCMA polypeptide and one or more T cell engagers for treating a subject having cancer.
- a mammal e.g., a human such as a human having cancer
- a combination treatment disclosed herein comprising (a) an anti-BCMA antigen binding protein or ADC having binding specificity for a BCMA polypeptide and (b) one or more T cell engagers.
- combinations comprising an anti-BCMA antigen binding protein and a T cell engager.
- the T cell engager binds to CD3.
- the anti- BCMA antigen binding protein comprises an antibody.
- the antibody is a monoclonal antibody.
- the monoclonal antibody is an IgG1.
- the antibody is afucosylated.
- the antibody is fucosylated.
- the antibody is sialylated.
- the antibody is glucosylated.
- the antibody is glycosylated.
- the antibody is galactosylated.
- the anti-BCMA antigen binding protein is human, humanized or chimeric.
- the anti-BCMA antigen binding protein comprises a CDRH1 comprising the amino acid sequence set out in SEQ ID NO:1; a CDRH2 comprising the amino acid sequence set out in SEQ ID NO:2; a CDRH3 comprising the amino acid sequence set out in SEQ ID NO:3; a CDRL1 comprising the amino acid sequence set out in SEQ ID NO:4; a CDRL2 comprising the amino acid sequence set out in SEQ ID NO:5; and a CDRL3 comprising the amino acid sequence set out in SEQ ID NO:6.
- the anti-BCMA antigen binding protein comprises a heavy chain variable region (VH) comprising the amino acid sequence set out in SEQ ID NO:7; and a light chain variable region (VL) comprising the amino acid sequence set out in SEQ ID NO:8.
- the anti-BCMA antigen binding protein comprises a heavy chain (H) comprising the amino acid sequence set out in SEQ ID NO:9 and a light chain (L) comprising the amino acid sequence set out in SEQ ID NO:10.
- the anti-BCMA antigen binding protein is an immunoconjugate.
- the anti-BCMA antigen binding protein is an immunoconjugate comprising an antibody conjugated to a cytotoxin.
- the cytotoxin is MMAE or MMAF. In some cases, the cytotoxin is MMAF. In some embodiments, the cytotoxin is AFP, MMAF, MMAE, AEB, AEVB or auristatin E. In some embodiments, the cytotoxin is paclitaxel, docetaxel, CC-1065, SN-38, topotecan, morpholino-doxorubicin, rhizoxin, cyanomorpholino-doxorubicin, dolastatin-10, echinomycin, combretatstatin, calicheamicin, or netropsin.
- the cytotoxin is an auristatin, a maytansinoid, or calicheamicin.
- the cytotoxin is vincristine, vinblastine, vindesine, vinorelbine, VP-16, camptothecin, epothilone A, epothilone B, nocodazole, colchicines, colcimid, estramustine, cemadotin, discodermolide, maytansinol, maytansine, DM1, DM2, DM3, DM4 or eleutherobin.
- the anti-BCMA antigen binding protein is belantamab mafodotin.
- belantamab mafodotin is present in the combination at a dose of at least about 0.5 mg/kg, 0.95 mg/kg, 1.0 mg/kg, 1.25 mg/kg, 1.4 mg/kg, 1.7 mg/kg, 1.9 mg/kg, 2.5 mg/kg, or 3.4 mg/kg.
- the therapeutically effective dose of the anti-BCMA antigen binding protein is 0.95 mg/kg.
- the therapeutically effective dose of the anti-BCMA antigen binding protein is 1.0 mg/kg.
- the therapeutically effective dose of the anti-BCMA antigen binding protein is 1.4 mg/kg.
- the therapeutically effective dose of the anti-BCMA antigen binding protein is 1.9 mg/kg.
- the therapeutically effective dose of the anti-BCMA antigen binding protein is 1.92 mg/kg. In some embodiments, the therapeutically effective dose of the anti-BCMA antigen binding protein is 2.5 mg/kg. In some embodiments, the therapeutically effective dose of the anti-BCMA antigen binding protein is 3.4 mg/kg. In some embodiments, the therapeutically effective dose of the anti-BCMA antigen binding protein is administered to the subject every week. In some embodiments, the therapeutically effective dose of the anti-BCMA antigen binding protein is administered to the subject every 2 weeks. In some embodiments, the therapeutically effective dose of the anti- BCMA antigen binding protein is administered to the subject every 3 weeks.
- the therapeutically effective dose of the anti-BCMA antigen binding protein is administered to the subject every 4 weeks. In some embodiments, the therapeutically effective dose of the anti-BCMA antigen binding protein is administered to the subject every 5 weeks. In some embodiments, the therapeutically effective dose of the anti-BCMA antigen binding protein is administered to the subject every 6 weeks. In some embodiments, dosage of the therapeutically effective dose of the anti-BCMA antigen binding protein is step-down to a lower dose described herein following a first administration. In some embodiments, 3.4 mg/kg dosage of the therapeutically effective dose of the anti-BCMA antigen binding protein is step-down to a 1.9 mg/kg dose, 1.4 mg/kg or less.
- a 2.5 mg/kg dosage of the therapeutically effective dose of the anti-BCMA antigen binding protein is step-down to a 1.9 mg/kg dose, 1.4 mg/kg or less.
- the therapeutically effective dose of the anti-BCMA antigen binding protein is administered to the subject on day 1, day 8 and thereafter every 3-12 weeks.
- the T cell engager is a bispecific T cell engager.
- the T cell engager is selected from the group consisting of Cevostamab, Talquetamab, Teclistimab, PF-3135, TNB-383B, REGN5458, blinatumomab, and solitomab.
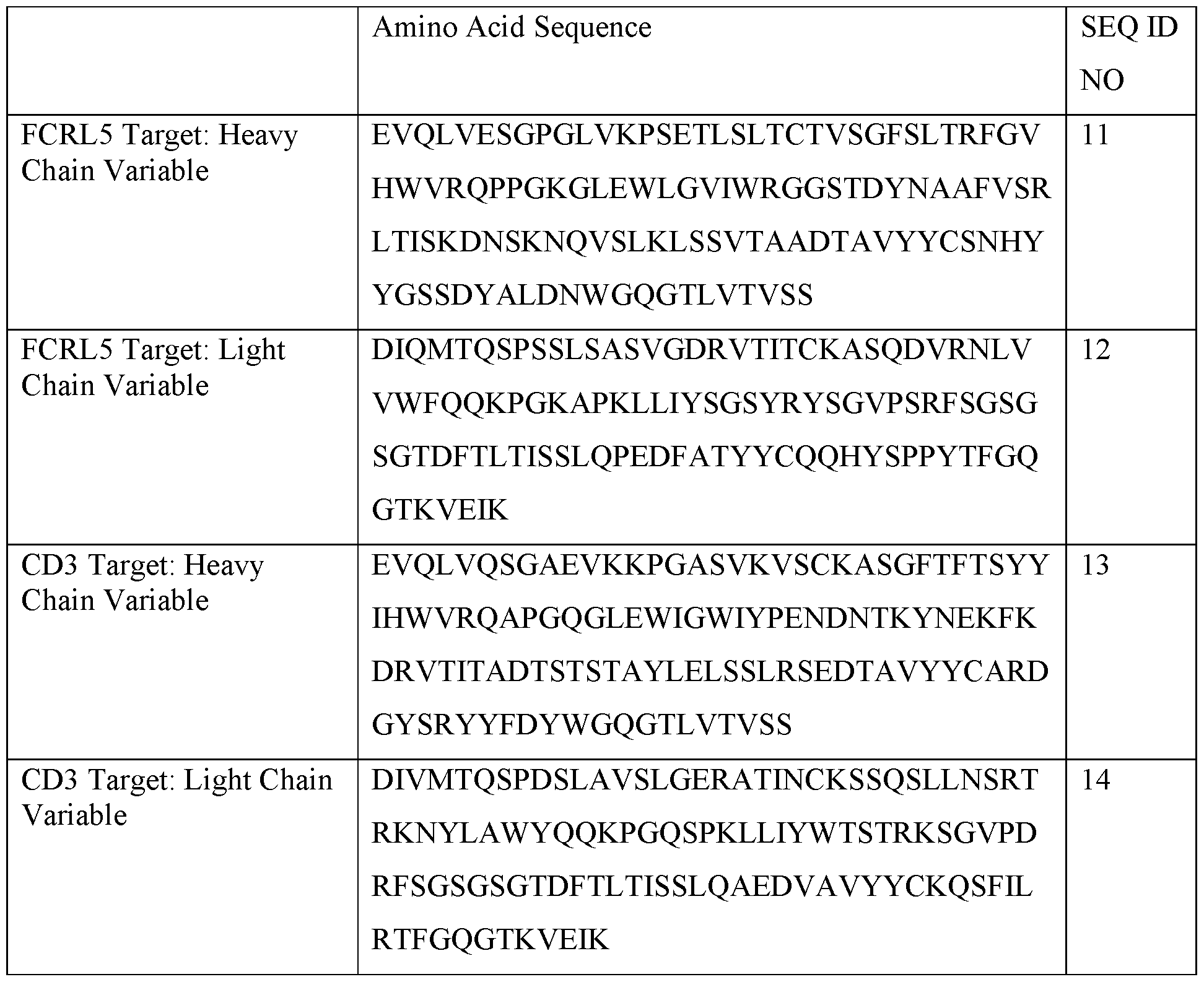
- the T cell engager is an anti-FcRH5 T cell engager. In some cases, the T cell engager is Cevostamab. In some embodiments, the T cell engager comprises the sequences set out in SEQ ID NO:11, 12, 13 and 14. In some cases, the combination comprises at least about 1.5 mg, 2mg, 3mg, 3.6mg, 10mg, 15mg, 20mg, 90 mg, or 132 mg Cevostamab. In some cases, the T cell engager is an anti-GPRC5D T cell engager. In some cases, the T cell engager is Talquetamab. In some cases, the T cell engager is an anti-BCMA T cell engager.
- the T cell engager is selected from the group consisting of Teclistimab, PF-3135, TNB-383B, and REGN5458. In some cases, the T cell engager is selected from the group consisting of CC- 93269, AMG701, JNJ-7957, and GBR 1342. In some cases, the T cell engager does not bind ICOS. In some cases, the T cell engager does not bind CD38. In some cases, the combination comprises a pharmaceutically acceptable carrier. In some cases, the combination further comprising an adjuvant. Disclosed herein are methods of treating cancer. In some cases, the method comprises treating cancer in a subject in need thereof comprising administering to the subject a therapeutically effective dose of a combination disclosed herein.
- the cancer is selected from the group consisting of multiple myeloma, chronic lymphocytic leukemia, Waldenstrom macroglobulinemia, and non-Hodgkin's lymphoma. In some cases, the cancer is multiple myeloma. In some cases, the cancer is relapsed and/or refractory multiple myeloma. In some cases, the subject has received at least one previous cancer treatment. In some cases, the therapeutically effective dose of the combination is administered to the subject at least about once every 1-60 days. In some cases, the therapeutically effective dose of the combination is administered to the subject at least about once every 21 days. In some cases, the therapeutically effective dose of the combination is administered to the subject at least about once every 8 days.
- administering the therapeutically effective dose of the combination reduces ocular toxicity as compared to administering a therapeutically effective amount of the anti-BCMA antigen binding protein alone.
- the anti-BCMA antigen binding protein is belantamab mafodotin.
- ocular toxicity is at least one of: changes in corneal epithelium, dry eyes, irritation, redness, blurred vision, dry eyes, photophobia, or changes in visual acuity.
- ocular toxicity is measured by at least one of the following methods: best corrected visual acuity, documentation of manifest refraction and the method used to obtain best corrected visual acuity, current glasses prescription (if applicable), intraocular pressure measurement, anterior segment (slit lamp) examination including fluorescein staining of the cornea and lens examination, dilated funduscopic examination, or an ocular surface disease index (OSDI).
- an anti-BCMA antigen binding protein disclosed herein is administer to a subject in a dose of at least about 0.5 mg/kg, 0.95 mg/kg, 1.25 mg/kg, 1.4 mg/kg, 1.7 mg/kg, 1.9 mg/kg, 1.92 mg/kg, 2.5 mg/kg, or 3.4 mg/kg.
- dosage of the therapeutically effective dose of the anti-BCMA antigen binding protein is step-down to a lower dose described herein following a first administration.
- 3.4 mg/kg dosage of the therapeutically effective dose of the anti-BCMA antigen binding protein is step-down to a 1.9 mg/kg dose, 1.4 mg/kg or less.
- a 2.5 mg/kg dosage of the therapeutically effective dose of the anti-BCMA antigen binding protein is step-down to a 1.9 mg/kg dose, 1.4 mg/kg or less.
- the therapeutically effective dose of the anti-BCMA antigen binding protein is administered to the subject on day 1, day 8 and thereafter every 3-12 weeks. Disclosed herein are the manufacture of a medicament for use.
- kits In some cases, a kit disclosed herein is for use in treatment of cancer. In some cases, a kit disclosed herein comprises a combination disclosed herein and instructions for use in the treatment of cancer. Disclosed herein are pre-filled syringes or autoinjector devices. In some cases, a pre- filled syringe or autoinjector device disclosed herein comprises a combination disclosed herein. Unless otherwise defined, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention pertains.
- the additional cancer therapeutic may be carfilzomib, daratumumab, isatuximab, ixazomib, marizomib, oprozomib, or a pharmaceutically acceptable salt thereof.
- an additional cancer therapeutic agent is a PD-1 inhibitor.
- the PD-1 inhibitor is selected from the group consisting of PDR001, Nivolumab, Pembrolizumab, Pidilizumab, MEDI0680, REGN2810, TSR-042, PF- 06801591, and AMP-224.
- the PD-1 inhibitor is Jemperli.
- an additional cancer therapeutic agent is a PD-L1 inhibitor.
- the PD-L1 inhibitor is selected from the group consisting of FAZ053, Atezolizumab, Avelumab, Durvalumab, and BMS-93655.
- an additional cancer therapeutic agent is a CTLA-4 inhibitor.
- the CTLA-4 inhibitor is Ipilimumab or Tremelimumab.
- an additional cancer therapeutic agent is a TIM-3 inhibitor.
- the TIM-3 inhibitor is MGB453 or TSR-022.
- an additional cancer therapeutic agent is a LAG-3 inhibitor.
- the LAG-3 inhibitor is selected from the group consisting of LAG525, BMS-986016, and TSR-033.
- an additional cancer therapeutic agent is an mTOR inhibitor.
- the mTOR inhibitor is RAD001 or rapamycin.
- the administration of the combinations of the disclosure may be advantageous over the individual therapeutic agents in that the combinations may provide one or more of the following improved properties when compared to the individual administration of a single therapeutic agent alone: i) a greater anticancer effect than the most active single agent, ii) synergistic or highly synergistic anticancer activity, iii) a dosing protocol that provides enhanced anticancer activity with reduced side effect profile, iv) a reduction in the toxic effect profile, v) an increase in the therapeutic window, or vi) an increase in the bioavailability of one or both of the therapeutic agents.
- a “pharmaceutical composition” contains a combination described herein, and one or more pharmaceutically acceptable carriers, diluents, or excipients.
- the carrier(s), diluent(s) or excipient(s) must be acceptable in the sense of being compatible with the other ingredients of the formulation, capable of pharmaceutical formulation, and not deleterious to the recipient thereof.
- each therapeutic agent in a combination is individually formulated into its own pharmaceutical composition and each of the pharmaceutical compositions are administered to treat cancer.
- each of the pharmaceutical compositions may have the same or different carriers, diluents or excipients.
- anti-BCMA antigen binding protein refers to antibodies and other protein constructs, such as domains, which are capable of binding to BCMA.
- the terms “BCMA binding protein” and “anti-BCMA antigen binding protein” are used interchangeably herein.
- the anti-BCMA antigen binding proteins described herein may bind to human BCMA having, including, for example, human BCMA containing the amino acid sequence of GenBank Accession Number Q02223.2, or genes encoding human BCMA having at least 90 percent homology or at least 90 percent identity thereto. Exemplary anti-BCMA antigen binding proteins and methods of making the same are disclosed in International Publication No. WO2012/163805 which is incorporated by reference herein in its entirety.
- Additional exemplary anti-BCMA antigen binding proteins include those described in WO2016/014789, WO2016/090320, WO2016/090327, WO2016/020332, WO2016/079177, WO2014/122143, WO2014/122144, WO2017/021450, WO2016/014565, WO2014/068079, WO2015/166649, WO2015/158671, WO2015/052536, WO2014/140248, WO2013/072415, WO2013/072406, WO2014/089335, US2017/165373, WO2013/154760, WO2018/201051 and WO2017/051068, each of which is incorporated by reference herein in its entirety.
- antibody refers to antibodies and other protein constructs, such as domains, which are capable of binding to the antigen.
- antibody is used herein in the broadest sense to refer to molecules with an immunoglobulin-like domain (for example IgG, IgM, IgA, IgD or IgE) and includes monoclonal, recombinant, polyclonal, chimeric, human, humanized, multispecific antibodies, including bispecific antibodies, and heteroconjugate antibodies; a single variable domain (e.g., a domain antibody (DAB)), antigen binding antibody fragments, Fab, F(ab’) 2 , Fv, disulphide linked Fv, single chain Fv, disulphide-linked scFv, diabodies, TANDABS, etc.
- DAB domain antibody
- a BCMA binding protein disclosed herein may be derived from rat, mouse, primate (e.g. cynomolgus, Old World monkey or Great Ape) or human.
- the BCMA binding protein may be a human, humanized or chimeric antibody.
- the BCMA binding protein may comprise a constant region, which may be of any isotype or subclass.
- the constant region may be of the IgG isotype, for example IgG1, IgG2, IgG3, IgG4 or variants thereof.
- the BCMA binding protein constant region may be IgG1.
- full refers to a heterotetrameric glycoprotein.
- An intact antibody is composed of two identical heavy chains (HCs) and two identical light chains (LCs) linked by covalent disulphide bonds. This H 2 L 2 structure folds to form three functional domains comprising two antigen-binding fragments, known as ‘Fab’ fragments, and a ‘Fc’ crystallisable fragment.
- the Fab fragment is composed of the variable domain at the amino-terminus, variable heavy (VH) or variable light (VL), and the constant domain at the carboxyl terminus, CH1 (heavy) and CL (light).
- the Fc fragment is composed of two domains formed by dimerization of paired CH2 and CH3 regions.
- the Fc may elicit effector functions by binding to receptors on immune cells or by binding C1q, the first component of the classical complement pathway.
- the five classes of antibodies IgM, IgA, IgG, IgE and IgD are defined by distinct heavy chain amino acid sequences, which are called ⁇ , ⁇ , ⁇ , ⁇ and ⁇ respectively, each heavy chain can pair with either a ⁇ or ⁇ light chain.
- the majority of antibodies in the serum belong to the IgG class, there are four isotypes of human IgG (IgG1, IgG2, IgG3 and IgG4), the sequences of which differ mainly in their hinge region.
- Fully human antibodies can be obtained using a variety of methods, for example using yeast-based libraries or transgenic animals (e.g. mice) that can produce repertoires of human antibodies. Yeast presenting human antibodies on their surface that bind to an antigen of interest can be selected using FACS (Fluorescence-Activated Cell Sorting) based methods or by capture on beads using labelled antigens. Transgenic animals that have been modified to express human immunoglobulin genes can be immunized with an antigen of interest and antigen-specific human antibodies isolated using B-cell sorting techniques. Human antibodies produced using these techniques can then be characterized for desired properties such as affinity, developability and selectivity. In some aspects, alternative antibody formats can be used.
- FACS Fluorescence-Activated Cell Sorting
- Alternative antibody formats include alternative scaffolds in which the one or more CDRs of the BCMA antibody can be arranged onto a suitable non-immunoglobulin protein scaffold or skeleton, such as an affibody, a SpA scaffold, an LDL receptor class A domain, an avimer (see, e.g., U.S. Patent Application Publication Nos.2005/0053973, 2005/0089932, 2005/0164301) or an EGF domain.
- domain refers to a folded polypeptide structure which retains its tertiary structure independent of the rest of the polypeptide.
- single variable domain refers to a folded polypeptide domain comprising sequences characteristic of antibody variable domains. It therefore includes complete antibody variable domains such as VH, VHH and VL and modified antibody variable domains, for example, in which one or more loops have been replaced by sequences which are not characteristic of antibody variable domains, or antibody variable domains which have been truncated or comprise N- or C-terminal extensions, as well as folded fragments of variable domains which retain at least the binding activity and specificity of the full-length domain.
- a single variable domain can bind an antigen or epitope independently of a different variable region or domain.
- a “domain antibody” or “DAB” may be considered the same as a “single variable domain”.
- a single variable domain may be a human single variable domain, but also includes single variable domains from other species such as rodent (for example, as disclosed in WO 00/29004 A1), nurse shark and Camelid VHH DABs.
- rodent for example, as disclosed in WO 00/29004 A1
- Camelid VHH are immunoglobulin single variable domain polypeptides that are derived from species including camel, llama, alpaca, dromedary, and guanaco, which produce heavy chain antibodies naturally devoid of light chains.
- VHH domains may be humanized according to standard techniques available in the art, and such domains are considered to be “single variable domains”.
- VH includes camelid VHH domains.
- An antigen binding fragment, BCMA binding protein fragment, functional fragment, biologically active fragment or an immunologically effective fragment may comprise partial heavy or light chain variable sequences. Fragments are at least 5, 6, 8 or 10 amino acids in length. Alternatively, the fragments are at least 15, at least 20, at least 50, at least 75, or at least 100 amino acids in length.
- An antigen binding fragment may be provided by means of arrangement of one or more CDRs on non-antibody protein scaffolds.
- Protein Scaffold as used herein includes but is not limited to an immunoglobulin (Ig) scaffold, for example an IgG scaffold, which may be a four chain or two chain antibody, or which may comprise only the Fc region of an antibody, or which may comprise one or more constant regions from an antibody, which constant regions may be of human or primate origin, or which may be an artificial chimera of human and primate constant regions.
- the protein scaffold may be an Ig scaffold, for example an IgG, or IgA scaffold.
- the IgG scaffold may comprise some or all the domains of an antibody (i.e. CH1, CH2, CH3, VH, VL).
- An antigen binding protein disclosed herein may comprise an IgG scaffold selected from IgG1, IgG2, IgG3, IgG4 or IgG4PE.
- the scaffold may be IgG1.
- the scaffold may consist of, or comprise, the Fc region of an antibody, or is a part thereof.
- the protein scaffold may be a derivative of a scaffold selected from the group consisting of CTLA-4, lipocalin, Protein A derived molecules such as Z-domain of Protein A (Affibody, SpA), A-domain (Avimer/Maxibody); heat shock proteins such as GroEl and GroES; transferrin (trans-body); ankyrin repeat protein (DARPin); peptide aptamer; C-type lectin domain (Tetranectin); human ⁇ -crystallin and human ubiquitin (affilins); PDZ domains; scorpion toxin kunitz type domains of human protease inhibitors; and fibronectin/adnectin; which has been subjected to protein engineering in order to obtain binding to an antigen other than the natural ligand.
- Protein A derived molecules such as Z-domain of Protein A (Affibody, SpA), A-domain (Avimer/Maxibody); heat shock proteins such as GroEl and GroES; transferrin (trans-body
- Antigen binding site refers to a site on an antigen binding protein which is capable of specifically binding to an antigen, this may be a single variable domain, or it may be paired VH/VL domains as can be found on a standard antibody. Single-chain Fv (ScFv) domains can also provide antigen-binding sites.
- the term multi-specific antigen binding protein refers to an antigen binding protein that comprises at least two different antigen binding sites. Each of these antigen-binding sites is capable of binding to a different epitope, which may be present on the same antigen or different antigens.
- the multi-specific antigen binding protein may have specificity for more than one antigen, for example two antigens, or three antigens, or four antigens.
- Symmetric formats combine multiple binding specificities in a single polypeptide chain or single HL pair including Fc-fusion proteins of fragment-based formats and formats whereby antibody fragments are fused to regular antibody molecules.
- Examples of symmetric formats may include DVD-Ig, TVD-Ig, CODV-Ig, (scFv)4-Fc, IgG-(scFv)2, Tetravalent DART-Fc, F(ab) 4 CrossMab, IgG-HC-scFv, IgG-LC-scFv, mAb-dAb etc.
- Asymmetric formats retain as closely as possible the native architecture of natural antibodies by forcing correct HL chain pairing and/or promoting H chain heterodimerization during the co-expression of three (if common heavy or light chains are used) or four polypeptide chains e.g. Triomab, asymmetric reengineering technology immunoglobulin (ART-Ig), CrossMab, Biclonics common light chain, ZW1 common light chain, DuoBody and knobs into holes (KiH), DuetMab, ⁇ body, Xmab, YBODY, HET-mAb, HET-Fab, DART-Fc, SEEDbody, mouse/rat chimeric IgG Bispecific formats also include an antibody fused to a non-Ig scaffold such as Affimabs, Fynomabs, Zybodies, and Anticalin-IgG fusions, ImmTAC.
- ART-Ig asymmetric reengineering technology immunoglobulin
- CrossMab Biclonics common
- an antigen binding protein described herein is a multi-specific antigen binding protein.
- chimeric antigen receptor refers to an engineered receptor which consists of an extracellular antigen binding domain (which is usually derived from a monoclonal antibody, or fragment thereof, e.g. a VH domain and a VL domain in the form of a scFv), optionally a spacer region, a transmembrane region, and one or more intracellular effector domains.
- CARs have also been referred to as chimeric T cell receptors or chimeric immunoreceptors (CIRs).
- transmembrane domain refers to the part of the CAR molecule which traverses the cell membrane.
- intracellular effector domain also referred to as the “signaling domain” as used herein refers to the domain in the CAR which is responsible for intracellular signaling following the binding of the antigen binding domain to the target.
- the intracellular effector domain is responsible for the activation of at least one of the normal effector functions of the immune cell in which the CAR is expressed.
- the effector function of a T cell can be a cytolytic activity or helper activity including the secretion of cytokines.
- Avidity also referred to as functional affinity, is the cumulative strength of binding at multiple interaction sites, e.g. the sum total of the strength of binding of two molecules (or more, e.g. in the case of a bispecific or multispecific molecule) to one another at multiple sites, e.g. taking into account the valency of the interaction.
- the equilibrium dissociation constant (KD) of an antigen binding protein disclosed herein – antigen interaction is 100 nM or less, 10 nM or less, 2 nM or less or 1 nM or less.
- the KD may be between 5 and 10 nM; or between 1 and 2 nM.
- the dissociation rate constant (kd) is 1x10 -3 s -1 or less, 1x10 -4 s -1 or less, 1x10 -5 s -1 or less, or 1x10 -6 s- 1 or less.
- the kd may be between 1x10 -5 s -1 and 1x10 -4 s -1 ; or between 1x10 -4 s -1 and 1x10 -3 s -1 .
- the association rate constant (ka) is 6.49x10 6 M- 1 s -1 , 4.65x10 6 M -1 s -1 , 3.27x10 6 M -1 s -1 , 8.28x10 6 M -1 s -1 , 1.47x10 7 M -1 s -1 , 1.10x10 7 M -1 s -1 , 5.90x10 6 M -1 s -1 .
- the term “derived” is intended to define not only the source in the sense of it being the physical origin for the material but also to define material which is structurally identical to the material but which does not originate from the reference source.
- the molecule such as a BCMA binding protein
- the molecule may be purified away from substances with which it would normally exist in nature.
- the BCMA binding protein can be purified to at least 95%, 96%, 97%, 98% or 99%, or greater with respect to a culture media containing the BCMA binding protein.
- the BCMA binding proteins and antibodies disclosed herein may be isolated BCMA binding proteins and antibodies. “CDRs” are defined as the complementarity determining region amino acid sequences of an antigen binding protein. These are the hypervariable regions of immunoglobulin heavy and light chains.
- CDRs There are three heavy chain and three light chain CDRs (or CDR regions) in the variable portion of an immunoglobulin.
- CDRs refers to all three heavy chain CDRs, all three light chain CDRs, all heavy and light chain CDRs, or at least two CDRs.
- amino acid residues in variable domain sequences and variable domain regions within full-length antigen binding sequences e.g. within an antibody heavy chain sequence or antibody light chain sequence, are numbered according to the Kabat numbering convention.
- CDR “CDRL1”, “CDRL2”, “CDRL3”, “CDRH1”, “CDRH2”, “CDRH3” used in the Examples follow the Kabat numbering convention.
- CDR sequences available to a skilled person include “AbM” (University of Bath) and “contact” (University College London) methods.
- the minimum overlapping region using at least two of the Kabat, Chothia, AbM and contact methods can be determined to provide the “minimum binding unit”.
- the minimum binding unit may be a sub- portion of a CDR.
- Table 1 below represents one definition using each numbering convention for each CDR or binding unit.
- the Kabat numbering scheme is used in Table 1 to number the variable domain amino acid sequence. It should be noted that some of the CDR definitions may vary depending on the individual publication used.
- a BCMA binding protein which comprises any one or a combination of the following CDRs: CDRH1 of SEQ ID NO:1, CDRH2 of SEQ ID NO:2, CDRH3 of SEQ ID NO:3, CDRL1 of SEQ ID NO:4, CDRL2 of SEQ ID NO:5, CDRL3 of SEQ ID NO:6.
- CDRs may be modified by at least one amino acid substitution, deletion or addition, wherein the variant antigen binding protein substantially retains the biological characteristics of the unmodified protein, such as binding to the antigen.
- Table 2 Exemplary CDR sequences for an anti-BCMA antigen binding protein.
- each of CDR H1, H2, H3, L1, L2, L3 may be modified alone or in combination with any other CDR, in any permutation or combination.
- a CDR is modified by the substitution, deletion or addition of up to 3 amino acids, for example 1 or 2 amino acids, for example 1 amino acid.
- the modification is a substitution, particularly a conservative substitution, for example as shown in Table 3 below.
- Table 3 Substitutions.
- the flanking residues that comprise the CDR as part of alternative definition(s) e.g. Kabat or Chothia may be substituted with a conservative amino acid residue.
- the VH or VL (or HC or LC) sequence disclosed herein may be a variant sequence with up to 10 amino acid substitutions, additions or deletions.
- the variant sequence may have up to 9, 8, 7, 6, 5, 4, 3, 2 or 1 amino acid substitution(s), addition(s) or deletion(s).
- the sequence variation may exclude one or more or all of the CDRs, for example the CDRs are the same as the VH or VL (or HC or LC) sequence and the variation is in the remaining portion of the VH or VL (or HC or LC) sequence, so that the CDR sequences are fixed and intact.
- the heavy chain variable region may have 75% or greater, 80% or greater, 85% or greater, 90% or greater, 95% or greater, 98% or greater, 99% or greater or 100% identity to an amino acid sequence described herein for an antibody; and the light chain variable region may have 75% or greater, 80% or greater, 85% or greater, 90% or greater, 95% or greater, 98% or greater, 99% or greater, or 100% identity to an amino acid sequence disclosed herein for an antibody.
- the heavy chain variable region of an antibody or amino acid sequence disclosed herein may be a variant which may contain 30, 25, 20, 15, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 amino acid substitutions, insertions or deletions.
- the light chain variable region of an antibody or amino acid sequence disclosed herein may be a variant which may contain 30, 25, 20, 15, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 amino acid substitutions, insertions or deletions.
- epitope refers to that portion of the antigen that makes contact with a particular binding domain of an antigen binding protein, also known as the paratope.
- An epitope may be linear or conformational/discontinuous.
- a conformational or discontinuous epitope comprises amino acid residues that are separated by other sequences, i.e. not in a continuous sequence in the antigen's primary sequence assembled by tertiary folding of the polypeptide chain.
- residues may be from different regions of the polypeptide chain, they are in close proximity in the three dimensional structure of the antigen.
- a conformational or discontinuous epitope may include residues from different peptide chains.
- Particular residues comprised within an epitope can be determined through computer modelling programs or via three-dimensional structures obtained through methods known in the art, such as X-ray crystallography.
- Epitope mapping can be carried out using various techniques known to persons skilled in the art as described in publications such as Methods in Molecular Biology ‘Epitope Mapping Protocols’, Mike Schutkowski and Ulrich Reineke (volume 524, 2009) and Johan Rockberg and Johan Nilvebrant (volume 1785, 2018).
- Exemplary methods include peptide based approaches such as pepscan whereby a series of overlapping peptides are screened for binding using techniques such as ELISA or by in vitro display of large libraries of peptides or protein mutants, e.g. on phage.
- Detailed epitope information can be determined by structural techniques including X-ray crystallography, solution nuclear magnetic resonance (NMR) spectroscopy and cryogenic-electron microscopy (cryo-EM).
- Mutagenesis such as alanine scanning, is an effective approach whereby loss of binding analysis is used for epitope mapping.
- Percent Identity is the “Identities” value, expressed as a percentage, that is calculated using a suitable algorithm or software, such as BLASTN, FASTA, DNASTAR Lasergene, GeneDoc, Bioedit, EMBOSS needle or EMBOSS infoalign, over the entire length of the query sequence after a pair-wise global sequence alignment has been performed using a suitable algorithm or software, such as BLASTN, FASTA, ClustalW, MUSCLE, MAFFT, EMBOSS Needle, T-Coffee, and DNASTAR Lasergene.
- a suitable algorithm or software such as BLASTN, FASTA, ClustalW, MUSCLE, MAFFT, EMBOSS Needle, T-Coffee, and DNASTAR Lasergene.
- a query nucleic acid sequence may be described by a nucleic acid sequence identified in one or more claims herein. “Percent identity” between a query amino acid sequence and a subject amino acid sequence is the “Identities” value, expressed as a percentage, that is calculated using a suitable algorithm or software, such as BLASTP, FASTA, DNASTAR Lasergene, GeneDoc, Bioedit, EMBOSS needle or EMBOSS infoalign, over the entire length of the query sequence after a pair-wise global sequence alignment has been performed using a suitable algorithm/software such as BLASTP, FASTA, ClustalW, MUSCLE, MAFFT, EMBOSS Needle, T-Coffee, and DNASTAR Lasergene.
- a suitable algorithm or software such as BLASTP, FASTA, ClustalW, MUSCLE, MAFFT, EMBOSS Needle, T-Coffee, and DNASTAR Lasergene.
- an anti-BCMA binding protein disclosed herein comprises a sequence that is at least about 50, 60, 70, 75, 80, 85, 90, 95, 96, 97, 98, or 99% identical to a sequence disclosed herein.
- the terms “peptide”, “polypeptide” and “protein” each refers to a molecule comprising two or more amino acid residues.
- a peptide may be monomeric or polymeric.
- Fc engineering methods can be applied to modify the functional or pharmacokinetics properties of an antibody. Effector function may be altered by making mutations in the Fc region that increase or decrease binding to C1q or Fc ⁇ receptors and modify CDC or ADCC activity respectively.
- FcR Fc receptors
- FcR Fc receptors
- Effector function can be assessed in a number of ways including, for example, evaluating ADCC effector function of antibody coated to target cells mediated by Natural Killer (NK) cells via Fc ⁇ RIII, or monocytes/macrophages via Fc ⁇ RI, or evaluating CDC effector function of antibody coated to target cells mediated by complement cascade via C1q.
- NK Natural Killer
- an antigen binding protein of the present invention can be assessed for ADCC effector function in a Natural Killer cell assay.
- Antigen binding proteins of the present invention may include any of the following mutations.
- Enhanced CDC Fc engineering can be used to enhance complement-based effector function. For example (with reference to IgG1), K326W/E333S; S267E/H268F/S324T; and IgG1/IgG3 cross subclass can increase C1q binding; E345R (Diebolder et al., Science 2014; 343: 1260-1293) and E345R/E430G/S440Y results in preformed IgG hexamers (Wang et al., Protein Cell.2018 Jan; 9(1): 63–73).
- G236A/S239D/I332E increases Fc ⁇ RIIa binding and increases Fc ⁇ RIIIa binding (Richards J et al., Mol. Cancer Ther.2008; 7: 2517-2527).
- Increased co-engagement Fc engineering can be used to increase co-engagement with FcRs.
- S267E/L328F increases Fc ⁇ RIIb binding
- N325S/L328F increases Fc ⁇ RIIa binding and decreases Fc ⁇ RIIIa binding (Wang et al.2018).
- An antigen binding protein of the present invention may comprise a heavy chain constant region with an altered glycosylation profile, such that the antigen binding protein has an enhanced effector function, e.g. enhanced ADCC, enhanced CDC, or both enhanced ADCC and CDC.
- an enhanced effector function e.g. enhanced ADCC, enhanced CDC, or both enhanced ADCC and CDC.
- suitable methodologies to produce antigen binding proteins with an altered glycosylation profile are described in WO2003011878, WO2006014679 and EP1229125, all of which can be applied to the antigen binding proteins of the present invention.
- the absence of the ⁇ 1,6 innermost fucose residues on the Fc glycan moiety on N297 of IgG1 antibodies enhances affinity for Fc ⁇ RIIIA.
- afucosylated or low fucosylated monoclonal antibodies may have increased therapeutic efficacy (Shields et al., J Biol Chem. 2002, 277(30): 26733-40 and Monnet et al., 2014, mAbs, 6:2, 422-436).
- Potelligent The present disclosure also provides a method of producing an antigen binding protein according to the invention comprising the steps of: a) culturing a recombinant host cell comprising an expression vector comprising the isolated nucleic acid as described herein, wherein the FUT8 gene encoding alpha-1,6- fucosyltransferase has been inactivated in the recombinant host cell; and b) recovering the antigen binding protein.
- antigen binding proteins can be performed, for example, using the POTELLIGENT technology system available from BioWa, Inc. (Princeton, NJ) in which CHOK1SV cells lacking a functional copy of the FUT8 gene produce monoclonal antibodies having enhanced ADCC activity that is increased relative to an identical monoclonal antibody produced in a cell with a functional FUT8 gene as described in US7214775, US6946292, WO0061739 and WO0231240, all of which are incorporated herein by reference. Those of ordinary skill in the art will also recognize other appropriate systems.
- the antigen binding protein is produced in a host cell in which the FUT8 gene has been inactivated.
- An antigen binding protein may have a half-life of at least 6 hours, at least 1 day, at least 2 days, at least 3 days, at least 4 days, at least 5 days, at least 7 days, or at least 9 days in vivo in humans, or in a murine animal model. Mutational changes to the Fc effector portion of the antibody can be used to change the affinity of the interaction between the FcRn and antibody to modulate antibody turnover. The half-life of the antibody can be extended in vivo. This could be beneficial to patient populations as maximal dose amounts and maximal dosing frequencies could be achieved as a result of maintaining in vivo IC 50 for longer periods of time.
- an antigen binding protein comprising a constant region may have reduced ADCC and/or complement activation or effector functionality.
- the constant domain may comprise a naturally disabled constant region of IgG2 or IgG4 isotype or a mutated IgG1 constant domain. Examples of suitable modifications are described in EP0307434.
- One way to achieve Fc disablement comprises the substitutions of alanine residues at positions 235 and 237 (EU index numbering) of the heavy chain constant region, i.e. L235A and G237A (commonly referred to as “LAGA” mutations).
- Another example comprises substitution with alanines at positions 234 and 235 (EU index numbering), i.e.
- L234A and L235A (commonly referred to as “LALA” mutations).
- the Fc effector function of an antigen binding protein disclosed herein has been disabled using the LAGA mutation. Additional alterations and mutations to decrease effector function include: (with reference to IgG1 unless otherwise noted): a glycosylated N297A or N297Q or N297G; L235E; IgG4:F234A/L235A; and chimeric IgG2/IgG4.
- IgG2 H268Q/V309L/A330S/P331S
- IgG2 V234A/G237A/P238S/H268A/V309L/A330S/P331S can reduce Fc ⁇ R and C1q binding (Wang et al.2018 and US 8,961,967).
- L234F/L235E/P331S a chimeric antibody created using the CH1 and hinge region from human IgG2 and the CH2 and CH3 regions from human IgG4; IgG2m4, based on the IgG2 isotype with four key amino acid residue changes derived from IgG4 (H268Q, V309L, A330S and P331S); IgG2 ⁇ which contains V234A/G237A /P238S/H268A/V309L/A330S/P331S substitutions to eliminate affinity for Fc ⁇ receptors and C1q complement protein; IgG2m4 (H268Q/V309L/A330S/P331S, changes to IgG4); IgG4 (S228P/L234A/L235A); huIgG1 L234A/L235A (AA); huIgG4 S228P/L234A/
- an antigen binding protein disclosed herein may comprise one or more modifications selected from a mutated constant domain such that the antibody has enhanced effector functions/ ADCC and/or complement activation. Examples of suitable modifications are described in Shields et al. J. Biol. Chem (2001) 276:6591-6604, Lazar et al. PNAS (2006) 103:4005-4010 and US6737056, WO2004063351 and WO2004029207.
- the antigen binding protein may comprise a constant domain with an altered glycosylation profile such that the antigen binding protein has enhanced effector functions/ ADCC and/or complement activation.
- Half-life Half-life refers to the time required for the serum concentration of an antigen binding protein to reach half of its original value.
- the serum half-life of proteins can be measured by pharmacokinetic studies according to the method described by Kim et al., 1994, Eur. J. of Immuno.24: 542-548. According to this method, radio-labelled protein is injected intravenously into mice and its plasma concentration is periodically measured as a function of time, for example, at about 3 minutes to about 72 hours after the injection.
- Antigen binding proteins of the present invention may have amino acid modifications that increase the affinity of the constant domain or fragment thereof for FcRn. Increasing the half-life (i.e., serum half-life) of therapeutic and diagnostic IgG antibodies and other bioactive molecules has many benefits including reducing the amount and/or frequency of dosing of these molecules.
- an antigen binding protein of the invention comprises all or a portion (an FcRn binding portion) of an IgG constant domain having one or more of the following amino acid modifications.
- M252Y/S254T/T256E (commonly referred to as “YTE” mutations) and M428L/N434S (commonly referred to as “LS” mutations) increase FcRn binding at pH 6.0 (Wang et al.2018).
- Half-life can also be enhanced by T250Q/M428L, V259I/V308F/M428L, N434A, and T307A/E380A/N434A mutations (with reference to IgG1 and Kabat numbering) (Monnet et al.).
- H433K and N434F mutations (commonly referred to as “HN” or “NHance” mutations) (with reference to IgG1) (WO2006/130834).
- WO00/42072 discloses a polypeptide comprising a variant Fc region with altered FcRn binding affinity, which polypeptide comprises an amino acid modification at any one or more of amino acid positions 238, 252, 253, 254, 255, 256, 265, 272, 286, 288, 303, 305, 307, 309, 311, 312, 317, 340, 356, 360, 362, 376, 378, 380, 386,388, 400, 413, 415, 424, 433, 434, 435, 436, 439, and 447 of the Fc region (EU index numbering).
- WO02/060919 discloses a modified IgG comprising an IgG constant domain comprising one or more amino acid modifications relative to a wild-type IgG constant domain, wherein the modified IgG has an increased half-life compared to the half-life of an IgG having the wild-type IgG constant domain, and wherein the one or more amino acid modifications are at one or more of positions 251, 253, 255, 285-290, 308-314, 385-389, and 428-435.
- Shields et al. 2001, J Biol Chem; 276:6591-604 used alanine scanning mutagenesis to alter residues in the Fc region of a human IgG1 antibody and then assessed the binding to human FcRn.
- Positions that effectively abrogated binding to FcRn when changed to alanine include I253, S254, H435, and Y436. Other positions showed a less pronounced reduction in binding as follows: E233-G236, R255, K288, L309, S415, and H433.
- the antigen binding protein of the invention comprises the E380A/N434A mutations and has increased binding to FcRn. Dall’Acqua et al.
- IgG1-human FcRn complex stability occurs when substituting residues located in a band across the Fc-FcRn interface (M252, S254, T256, H433, N434, and Y436) and to lesser extent substitutions of residues at the periphery, such as V308, L309, Q311, G385, Q386, P387, and N389.
- the variant with the highest affinity to human FcRn was obtained by combining the M252Y/S254T/T256E (“YTE”) and H433K/N434F/Y436H mutations and exhibited a 57-fold increase in affinity relative to the wild- type IgG1.
- the in vivo behavior of such a mutated human IgG1 exhibited a nearly 4-fold increase in serum half-life in cynomolgus monkey as compared to wild-type IgG1.
- the present invention therefore provides an antigen binding protein with optimized binding to FcRn.
- the antigen binding protein comprises at least one amino acid modification in the Fc region of said antigen binding protein, wherein said modification is at an amino acid position selected from the group consisting of 226, 227, 228, 230, 231, 233, 234, 239, 241, 243, 246, 250, 252, 256, 259, 264, 265, 267, 269, 270, 276, 284, 285, 288, 289, 290, 291, 292, 294, 297, 298, 299, 301, 302, 303, 305, 307, 308, 309, 311, 315, 317, 320, 322, 325, 327, 330, 332, 334, 335, 338, 340, 342, 343, 345, 347, 350, 352, 354, 355, 356, 359, 360, 361, 362, 369, 370, 371, 375, 378, 380, 382, 384, 385, 386, 387, 389, 390, 392, 393, 394, 3
- variants show increased serum persistence in hFcRn mice, as well as conserved enhanced ADCC (Monnet et al.)
- Exemplary variants include (with reference to IgG1 and Kabat numbering): P230T/V303A/K322R/N389T/F404L/N434S; P228R/N434S; Q311R/K334R/Q342E/N434Y; C226G/Q386R/N434Y; T307P/N389T/N434Y; P230S/N434S;P230T/V305A/T307A/A378V/L398P/N434S; P23OT/P387S/N434S; P230Q/E269D/N434S; N276S/A378V/N434S; T307A/N315D/A330V/382V/N389T/N434Y; T256N/A3
- an immunoconjugate (interchangeably referred to as an "antibody-drug conjugate", "ADC” or “antigen binding protein-drug conjugate”) comprising an antigen binding protein according to the invention conjugated to one or more drugs, such as a cytotoxic agent, such as a chemotherapeutic agent, an immunotherapeutic agent, a growth inhibitory agent, a toxin (e.g., a protein toxin, such as an enzymatically active toxin of bacterial, fungal, plant, or animal origin, or fragments thereof), an antiviral agent, a radioactive isotope (i.e., a radioconjugate), an antibiotic, or a small interfering RNA (siRNA).
- drugs such as a cytotoxic agent, such as a chemotherapeutic agent, an immunotherapeutic agent, a growth inhibitory agent, a toxin (e.g., a protein toxin, such as an enzymatically active toxin of bacterial, fun
- Immunoconjugates have been used for the local delivery of cytotoxic agents, i.e., drugs that kill or inhibit the growth or proliferation of cells, in the treatment of cancer (Lambert, J. (2005) Curr. Opinion in Pharmacology 5:543-549; Wu et al. (2005) Nature Biotechnology 23(9):1137-1146; Payne, G. (2003) i 3:207-212; Syrigos and Epenetos (1999) Anticancer Research 19:605-614; Niculescu-Duvaz and Springer (1997) Adv. Drug Deliv. Rev.26:151-172; U.S. Pat. No.4,975,278).
- cytotoxic agents i.e., drugs that kill or inhibit the growth or proliferation of cells
- Immunoconjugates allow for, inter alia, the targeted delivery of a drug moiety to a tumor, and intracellular accumulation therein, where systemic administration of unconjugated drugs may result in unacceptable levels of toxicity to normal cells(Tsuchikama and An, Protein and Cell, (2016) 9: 33–46).
- Immunoconjugates can enable selective delivery of a potent cytotoxic payload to target cancer cells, resulting in improved efficacy, reduced systemic toxicity, and preferable pharmacokinetics (PK)/pharmacodynamics (PD) and biodistribution compared to traditional chemotherapy (Tsuchikama and An 2018); Beck A. et al (2017) Nature Rev. Drug Disc.16: 315-337).
- an immunoconjugate comprises an antigen binding protein, such as an antibody, and a drug, such as toxin, such as a chemotherapeutic agent.
- Enzymatically active toxins and fragments thereof that can be used include diphtheria A chain, nonbinding active fragments of diphtheria toxin, exotoxin A chain (from Pseudomonas aeruginosa), ricin A chain, abrin A chain, modeccin A chain, alpha-sarcin, Aleurites fordii proteins, dianthin proteins, Phytolaca americana proteins (PAPI, PAPII, and PAP-S), momordica charantia inhibitor, curcin, crotin, sapaonaria officinalis inhibitor, gelonin, mitogellin, restrictocin, phenomycin, enomycin, and the tricothecenes.
- diphtheria A chain nonbinding active fragments of diphtheria toxin
- exotoxin A chain from Pseudomonas aeruginosa
- ricin A chain abrin A chain
- modeccin A chain alpha-
- a radioactive material such as a radionucleotide
- a radionucleotide may be used as the drug in an ADC.
- a variety of radionucleotides are available for the production of radioconjugated antibodies. Examples include 212Bi, 131I, 131In, 90Y, and 186Re.
- Antigen binding proteins (such as antibodies) of the present invention may also be conjugated to one or more toxins, including, but not limited to, a calicheamicin, a maytansinoid, a dolastatin, an aurostatin, a trichothecene, and CC1065, and a derivative of these toxins that have toxin activity.
- toxins including, but not limited to, a calicheamicin, a maytansinoid, a dolastatin, an aurostatin, a trichothecene, and CC1065, and a derivative of these toxins that have toxin activity.
- Suitable cytotoxic agents include, but are not limited to, an auristatin including dovaline-valine-dolaisoleunine-dolaproine-phenylalanine (MMAF) and monomethyl auristatin E (MMAE) as well as an ester form of MMAE, a DNA minor groove binding agent, a DNA minor groove alkylating agent, an enediyne, a lexitropsin, a duocarmycin, a taxane (such as paclitaxel and docetaxel), a puromycin, a dolastatin, a maytansinoid, and a vinca alkaloid.
- an auristatin including dovaline-valine-dolaisoleunine-dolaproine-phenylalanine (MMAF) and monomethyl auristatin E (MMAE) as well as an ester form of MMAE
- a DNA minor groove binding agent e.g., a DNA minor groove alky
- cytotoxic agents include topotecan, morpholino-doxorubicin, rhizoxin, cyanomorpholino-doxorubicin, dolastatin-10, echinomycin, combretatstatin, chalicheamicin, maytansine, DM-1, DM-4, and netropsin.
- Other suitable cytotoxic agents include anti-tubulin agents, such as an auristatin, a vinca alkaloid, a podophyllotoxin, a taxane, a baccatin derivative, a cryptophysin, a maytansinoid, a combretastatin, or a dolastatin.
- Antitubulin agents include dimethylvaline-valine-dolaisoleuine-dolaproine-phenylalanine-p-phenylened- iamine (AFP), MMAF, MMAE, auristatin E, vincristine, vinblastine, vindesine, vinorelbine, VP-16, camptothecin, paclitaxel, docetaxel, epothilone A, epothilone B, nocodazole, colchicines, colcimid, estramustine, cemadotin, discodermolide, maytansine, DM-1, DM-4, and eleutherobin.
- AFP dimethylvaline-valine-dolaisoleuine-dolaproine-phenylalanine-p-phenylened- iamine
- MMAF MMAF
- MMAE auristatin E
- vincristine vinblastine
- vindesine vinorelbine
- Antibody drug conjugates can be produced by conjugating the anti-tubulin agent monomethylauristatin E (MMAE) or monomethylauristatin F (MMAF) to an antigen binding protein (such as an antibody).
- the linker can consist of a thiol-reactive maleimide, a caproyl spacer, the dipeptide valine-citrulline, or p-aminobenzyloxycarbonyl, a self-immolative fragmenting group.
- MMAF a protease-resistant maleimidocaproyl linker can be used.
- the conjugation process leads to heterogeneity in drug- antibody attachment, varying in both the number of drugs bound to each antibody molecule (mole ratio [MR]), and the site of attachment.
- the overall average drug-to- antibody MR is approximately 4.
- Auristatins and Dolastatins in some embodiments, the immunoconjugate comprises an antigen binding protein (such as an antibody) conjugated to a dolastatin or a dolostatin peptidic analog or derivative, an auristatin (U.S. Pat. Nos.5,635,483; 5,780,588).
- Dolastatins and auristatins have been shown to interfere with microtubule dynamics, GTP hydrolysis, and nuclear and cellular division (Woyke et al. (2001) Antimicrob. Agents and Chemother.45(12):3580-3584) and have anticancer (U.S. Pat. No.5,663,149) and antifungal activity (Pettit et al. (1998) Antimicrob. Agents Chemother. 42:2961-2965).
- the dolastatin or auristatin (a pentapeptide derivative of dolastatin) drug moiety may be attached to the antibody through the N (amino) terminus or the C (carboxyl) terminus of the peptidic drug moiety (WO 02/088172).
- Exemplary auristatin embodiments include the N-terminus linked monomethylauristatin drug moieties DE and DF, disclosed in "Monomethylvaline Compounds Capable of Conjugation to Ligands," U.S. Patent No.7,498,298.
- MMAE monomethyl auristatin E.
- MMAF dovaline-valine- dolaisoleuine-dolaproine-phenylalanine.
- peptide-based drug moieties can be prepared by forming a peptide bond between two or more amino acids and/or peptide fragments.
- Such peptide bonds can be prepared, for example, according to the liquid phase synthesis method (see E. Schroder and K. Lubke, "The Peptides,” volume 1, pp 76-136, 1965, Academic Press) that is well known in the field of peptide chemistry.
- the auristatin/dolastatin drug moieties may be prepared according to the methods of: U.S. Pat. No.5,635,483; U.S. Pat. No.5,780,588; Pettit et al. (1989) J. Am. Chem. Soc.111:5463-5465; Pettit et al. (1998) Anti-Cancer Drug Design 13:243-277; Pettit, G. R., et al.
- Maytansine and Maytansinoids are mitototic inhibitors that act by inhibiting tubulin polymerization. Maytansine was first isolated from the east African shrub Maytenus serrata (U.S. Pat. No. 3,896,111). Subsequently, it was discovered that certain microbes also produce maytansinoids, such as maytansinol and C-3 maytansinol esters (U.S. Pat. No.4,151,042).
- Highly cytotoxic maytansinoid drugs can be prepared from ansamitocin precursors produced by fermentation of microorganisms such as Actinosynnema. Methods for isolating ansamitocins are described in U.S. Patent No.6,573,074. Synthetic maytansinol and derivatives and analogues thereof are disclosed, for example, in U.S. Pat.
- Antibody-maytansinoid conjugates are prepared by chemically linking an antigen binding protein (such as an antibody)to a maytansinoid molecule without significantly diminishing the biological activity of either the antibody or the maytansinoid molecule.
- an antigen binding protein such as an antibody
- Maytansinoids are maytansinol and maytansinol analogues modified in the aromatic ring or at other positions of the maytansinol molecule, such as various maytansinol esters.
- Methods for preparing maytansinoids for linkage with antibodies are disclosed, e.g., in U.S. Patent Nos.6,570,024 and 6,884,874.
- Calicheamicin The calicheamicin family of antibiotics is capable of producing double-stranded DNA breaks at sub-picomolar concentrations. For the preparation of conjugates of the calicheamicin family, see, e.g., U.S. Pat.
- Structural analogues of calicheamicin that may be used include, but are not limited to, ⁇ 1I, ⁇ 2I, ⁇ 3I, N-acetyl ⁇ 1I, PSAG and ⁇ I1 (Hinman et al., Cancer Research 53:3336-3342 (1993), Lode et al., Cancer Research 58:2925-2928 (1998) and the aforementioned U.S. patents).
- Another anti-tumor drug that the antibody can be conjugated to is QFA, which is an antifolate.
- cytotoxic agents such as antitumor agents, that can be conjugated to an antigen binding protein (such as an antibody) include BCNU, streptozoicin, vincristine and 5- fluorouracil, the family of agents known collectively LL-E33288 complex described in U.S. Pat. Nos.5,053,394 and 5,770,710, as well as esperamicins (U.S. Pat. No.5,877,296).
- Enzymatically active toxins and fragments thereof that can be used include diphtheria A chain, nonbinding active fragments of diphtheria toxin, exotoxin A chain (from Pseudomonas aeruginosa), ricin A chain, abrin A chain, modeccin A chain, alpha-sarcin, Aleurites fordii proteins, dianthin proteins, Phytolaca americana proteins (PAPI, PAPII, and PAP-S), momordica charantia inhibitor, curcin, crotin, sapaonaria officinalis inhibitor, gelonin, mitogellin, restrictocin, phenomycin, enomycin and the tricothecenes.
- diphtheria A chain nonbinding active fragments of diphtheria toxin
- exotoxin A chain from Pseudomonas aeruginosa
- ricin A chain abrin A chain
- modeccin A chain alpha-s
- the present invention further contemplates an immunoconjugate formed between an antigen binding protein (such as an antibody) and a compound with nucleolytic activity (e.g., a ribonuclease or a DNA endonuclease such as a deoxyribonuclease; DNase).
- an antigen binding protein such as an antibody
- a compound with nucleolytic activity e.g., a ribonuclease or a DNA endonuclease such as a deoxyribonuclease; DNase.
- the antigen binding protein such as an antibody
- the antigen binding protein may comprise a highly radioactive atom.
- a variety of radioactive isotopes are available for the production of radioconjugated antibodies.
- the conjugate When the conjugate is used for detection, it may comprise a radioactive atom for scintigraphic studies, for example tc99m or I123, or a spin label for nuclear magnetic resonance (NMR) imaging (also known as magnetic resonance imaging, mri), such as iodine-123 again, iodine-131, indium-111, fluorine-19, carbon- 13, nitrogen-15, oxygen-17, gadolinium, manganese or iron.
- NMR nuclear magnetic resonance
- mri nuclear magnetic resonance
- the radio- or other labels may be incorporated in the conjugate in known ways.
- the peptide may be biosynthesized or may be synthesized by chemical amino acid synthesis using suitable amino acid precursors involving, for example, fluorine-19 in place of hydrogen.
- Labels such as tc99m or I123, Re186, Re188 and In111 can be attached via a cysteine residue in the peptide.
- Yttrium-90 can be attached via a lysine residue.
- the IODOGEN method (Fraker et al. (1978) Biochem. Biophys. Res. Commun.80: 49-57) can be used to incorporate iodine-123. "Monoclonal Antibodies in Immunoscintigraphy" (Chatal, CRC Press 1989) describes other methods in detail.
- an anti-BCMA antigen binding protein disclosed herein is an immunoconjugate comprising an antigen binding protein according to the disclosure as herein described including, but not limited to, an antibody conjugated to one or more cytotoxic agents, such as a chemotherapeutic agent, a drug, a growth inhibitory agent, a toxin (e.g. , a protein toxin, an enzymatically active toxin of bacterial, fungal, plant, or animal origin, or fragments thereof), or a radioactive isotope (i.e. , a radioconjugate).
- cytotoxic agents such as a chemotherapeutic agent, a drug, a growth inhibitory agent, a toxin (e.g. , a protein toxin, an enzymatically active toxin of bacterial, fungal, plant, or animal origin, or fragments thereof), or a radioactive isotope (i.e. , a radioconjugate).
- the anti- BCMA antigen binding protein is conjugated to a toxin such as an auristatin, e.g., monomethyl auristatin E (MMAE) or monomethyl auristatin F (MMAF).
- auristatin e.g., monomethyl auristatin E (MMAE) or monomethyl auristatin F (MMAF).
- MMAE monomethyl auristatin E
- MMAF monomethyl auristatin F
- the anti- BCMA antigen binding protein is conjugated to AFP, MMAF, MMAE, AEB, AEVB or auristatin E.
- the anti- BCMA antigen binding protein is conjugated to paclitaxel, docetaxel, CC-1065, SN-38, topotecan, morpholino-doxorubicin, rhizoxin, cyanomorpholino-doxorubicin, dolastatin-10, echinomycin, combretatstatin, calicheamicin, or netropsin.
- the anti- BCMA antigen binding protein is conjugated to an auristatin, a maytansinoid, or calicheamicin.
- the anti- BCMA antigen binding protein is conjugated to AFP, MMAP, MMAE, AEB, AEVB, auristatin E, vincristine, vinblastine, vindesine, vinorelbine, VP-16, camptothecin, paclitaxel, docetaxel, epothilone A, epothilone B, nocodazole, colchicines, colcimid, estramustine, cemadotin, discodermolide, maytansinol, maytansine, DM1, DM2, DM3, DM4 or eleutherobin.
- an anti-BCMA antigen binding protein conjugated to a toxin can include a heavy chain having a V H CDR1 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in SEQ ID NO:1, a V H CDR2 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in SEQ ID NO:2, and a V H CDR3 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in SEQ ID NO:3.
- an anti-BCMA antigen binding protein conjugated to a toxin described herein can include a heavy chain variable region including the amino acid sequence set forth in SEQ ID NO:7.
- an anti-BCMA antigen binding protein conjugated to a toxin described herein can include a heavy chain comprising the amino acid sequence set forth in SEQ ID NO:9.
- an anti-BCMA antigen binding protein conjugated to a toxin can include a light chain having a V L CDR1 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in SEQ ID NO:4, a VL CDR2 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in SEQ ID NO:5, and a V L CDR3 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in SEQ ID NO:6.
- An anti-BCMA antigen binding protein conjugated to a toxin described herein can include a light chain variable region including the amino acid sequence set forth in SEQ ID NO:8.
- an anti-BCMA antigen binding protein conjugated to a toxin described herein can include a light chain comprising the amino acid sequence set forth in SEQ ID NO:10.
- an anti-BCMA antigen binding protein conjugated to a toxin can include a heavy chain having a V H CDR1 including the amino acid sequence set forth in SEQ ID NO:1, a V H CDR2 including the amino acid sequence set forth in SEQ ID NO:2, and a V H CDR3 including the amino acid sequence set forth in SEQ ID NO:3, and can include a light chain having a V L CDR1 including the amino acid sequence set forth in SEQ ID NO:4, a V L CDR2 including the amino acid sequence set forth in SEQ ID NO:5, and a V L CDR3 including the amino acid sequence set forth in SEQ ID NO:6.
- an anti- BCMA antigen binding protein disclosed herein comprises the light chain variable region of SEQ ID NO:20, 24 or 28. In some embodiments, an anti-BCMA antigen binding protein disclosed herein comprises the heavy chain region of SEQ ID NO:21, 25, 30, or 32. In some embodiments, an anti-BCMA antigen binding protein disclosed herein comprises the light chain region of SEQ ID NO:22, 26, 31 or 33.
- an anti-BCMA antigen binding protein disclosed herein comprises the heavy chain variable region of SEQ ID NO:19 and the light chain variable region of SEQ ID NO:20, the heavy chain variable region of SEQ ID NO:23 and the light chain variable region of SEQ ID NO:24, or the heavy chain variable region of SEQ ID NO:27 and the light chain variable region of SEQ ID NO:28.
- an anti- BCMA antigen binding protein disclosed herein comprises the heavy chain region of SEQ ID NO:21 and the light chain of SEQ ID NO:22, the heavy chain region of SEQ ID NO: 25 and the light chain of SEQ ID NO:26, the heavy chain region of SEQ ID NO:30 and the light chain of SEQ ID NO:31 or the heavy chain region of SEQ ID NO:32 and the light chain of SEQ ID NO:33.
- an anti-BCMA antigen binding protein disclosed herein is an scFV-fc comprising SEQ ID NO:29.
- the anti-BCMA antigen binding protein is an immunoconjugate having the following general structure: ABP-((Linker)n-Ctx)m wherein ABP is an antigen binding protein Linker is either absent or any a cleavable or non-cleavable linker Ctx is any cytotoxic agent described herein n is 0, 1, 2, or 3 and m is 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10.
- Exemplary linkers include 6- maleimidocaproyl (MC), maleimidopropanoyl (MP), valine-citrulline (val-cit), alanine- phenylalanine (ala-phe), p-aminobenzyloxycarbonyl (PAB), N-Succinimidyl 4-(2- pyridylthio)pentanoate (SPP), N-succinimidyl 4-(N- maleimidomethyl)cyclohexane-1 carboxylate (SMCC), and N-succinimidyl (4-iodo-acetyl) aminobenzoate (SIAB).
- MC 6- maleimidocaproyl
- MP maleimidopropanoyl
- val-cit valine-citrulline
- ala-phe p-aminobenzyloxycarbonyl
- PAB p-aminobenzyloxycarbonyl
- SPP N-Succinimi
- the anti-BCMA antigen binding protein is an immunoconjugate containing a monoclonal antibody linked to MMAE or MMAF.
- the anti-BCMA antigen binding protein is an immunoconjugate containing a monoclonal antibody linked to MMAE or MMAF by an MC linker as depicted in the following structures:
- the anti-BCMA antigen binding protein is the antibody belantamab. In another embodiment, the anti-BCMA antigen binding protein is the immunoconjugate belantamab mafodotin.
- the conjugated antibodies (antibody-drug conjugates or ADCs) of the present disclosure are powerful anti-cancer agents designed to allow specific targeting of highly potent cytotoxic agents to tumor cells while sparing healthy tissues. Despite the use of tumor- specific antibodies, the emerging clinical data with ADCs indicates that adverse events frequently occur before ADCs have reached their optimal therapeutic dose.
- cancers and in particular B-cell mediated or plasma cell mediated diseases or antibody mediated diseases or disorders include Multiple Myeloma (MM), chronic lymphocytic leukemia (CLL), Follicular Lymphoma (FL), Diffuse Large B-Cell Lymphoma (DLBCL), Non-secretory multiple myeloma, Smoldering multiple myeloma, Monoclonal gammopathy of undetermined significance (MGUS), Solitary plasmacytoma (Bone, Extramedullary), Lymphoplasmacytic lymphoma (LPL), Waldenström’s Macroglobulinemia, Plasma cell leukemia, Heavy chain disease, Systemic lupus erythematosus (SLE), POEMS syndrome / osteosclerotic myeloma, Type I and II cryoglobulinemia, Light chain deposition disease, Goodpasture’s syndrome, Idiopathic thrombocytopenic purpura (ITP), Acute glomerulonephritis,
- the disease or disorder is selected from the group consisting of Multiple Myeloma (MM), Non-Hodgkin’s Lymphoma B-cell leukemia (NHL), Follicular Lymphoma (FL), and Diffuse Large B-Cell Lymphoma (DLBCL).
- the disease is Multiple Myeloma or Non-Hodgkin’s Lymphoma B-cell leukemia (NHL).
- the disease is Multiple Myeloma.
- the cancer may be a hematopoietic (or hematologic or haematological or blood-related) cancer, for example, cancers derived from blood cells or immune cells, which may be referred to as "liquid tumors”.
- the cancer is a B-cell related cancer and particularly a BCMA-expressing cancer.
- the cancer is a leukemia such as chronic myelocytic leukemia, acute myelocytic leukemia, chronic lymphocytic leukemia and acute lymphocytic leukemia.
- the cancer is a lymphoma such as non-Hodgkin's lymphoma, Hodgkin's lymphoma; and the like.
- the cancer is a plasma cell malignancy such as multiple myeloma, and Waldenstrom's macroglobulinemia.
- a combination disclosed herein treats AL amyloidosis.
- the cancer is multiple myeloma.
- the cancer is relapsed and/or refractory multiple myeloma.
- the patient with relapsed and/or refractory multiple myeloma has been previously treated with at least one, at least two, at least three or at least four therapeutics to treat the multiple myeloma.
- Prior Treatment In some cases, a subject described herein may have had 0, 1, 2, 3, or 4 or more prior lines of treatment before being treated with the combinations described herein. In another embodiment, the subject may have relapsed and/or refractory multiple myeloma and have had 0, 1, 2, 3, or 4 or more prior lines of treatment before being treated with the combinations described herein.
- a cancer can be a metastatic cancer. In some cases, a cancer can be a chemo-resistant cancer. In some cases, a cancer can be a B cell cancer (e.g., leukemias and lymphomas).
- B cell cancer e.g., leukemias and lymphomas. Examples of cancers that can be treated as described herein include, without limitation, multiple myeloma (MM), chronic lymphocytic leukemia (CLL), chronic myelocytic leukemia, acute myelocytic leukemia, acute lymphocytic leukemia, follicular lymphoma (FL), diffuse large B- cell lymphoma (DLBCL), non-secretory multiple myeloma, smoldering multiple myeloma, monoclonal gammopathy of undetermined significance (MGUS), solitary plasmacytoma (e.g., solitary plasmacytoma of the bone and extramedullary solitary plasmacytoma), lympho
- a combination disclosed herein can be used to treat a disease or condition for which a BCMA antigen binding protein is indicated, for example a cancer.
- a treatment may comprise: (i) an anti-BCMA antigen binding protein or an ADC having binding specificity for a BCMA polypeptide and (ii) one or more T cell engagers.
- a mammal e.g., a human such as a human having cancer
- a combination disclosed herein targets the cytotoxic agent of the ADC to cells (e.g., cancer cells) expressing a BCMA polypeptide (e.g., expressing a BCMA polypeptide on the cell surface) and to stimulate (e.g., induce or enhance) an immune response against cells (e.g., cancer cells) expressing a cancer related antigen.
- a BCMA antigen binding protein and a T cell engager can be bound to the same cancer cell.
- an anti-BCMA antigen binding protein and a T cell engager can be bound to different cancer cells.
- an anti-BCMA antigen binding protein and a T cell engager can interact with the same or different cancer cells.
- combinations disclosed herein are for the treatment of a subject.
- the terms “individual”, “subject” and “patient” are used herein interchangeably.
- the subject is typically a human.
- the subject may also be a mammal, such as a mouse, rat or primate (e.g. a marmoset or monkey).
- the subject can be a non-human animal.
- the subject to be treated may be a farm animal for example, a cow or bull, sheep, pig, ox, goat or horse or may be a domestic animal such as a dog or cat.
- the animal may be any age, or a mature adult animal.
- treatment may be therapeutic, prophylactic or preventative.
- the subject may be one who is in need thereof.
- compositions described herein can be used for prophylactic or preventative treatment.
- the compositions described herein is administered to an individual in order to prevent or delay the onset of one or more aspects or symptoms of a disease.
- the subject can be asymptomatic.
- the subject may have a genetic predisposition to the disease.
- a prophylactically effective amount of a combination disclosed herein is administered to such an individual.
- a prophylactically effective amount is an amount which prevents or delays the onset of one or more aspects or symptoms of a disease described herein.
- a combination disclosed herein may also be used in methods of therapy.
- a combination disclosed herein may be used to ameliorate or reduce one or more aspects or symptoms of a disease described herein.
- a combination described herein is used in an effective amount for therapeutic, prophylactic or preventative treatment.
- a therapeutically effective amount of a combination described herein is an amount effective to ameliorate or reduce one or more aspects or symptoms of the disease.
- a combination disclosed herein may also have a generally beneficial effect on the subject's health, for example it can increase the subject's expected longevity.
- a combination described herein need not affect a complete cure or eradicate every symptom or manifestation of the disease to constitute a viable therapeutic treatment.
- drugs employed as therapeutic agents may reduce the severity of a given disease state but need not abolish every manifestation of the disease to be regarded as useful therapeutic agents.
- a prophylactically administered treatment need not be completely effective in preventing the onset of a disease in order to constitute a viable prophylactic agent. Simply reducing the impact of a disease (for example, by reducing the number or severity of its symptoms, or by increasing the effectiveness of another treatment, or by producing another beneficial effect), or reducing the likelihood that the disease will occur (for example by delaying the onset of the disease) or worsen in a subject, is sufficient.
- the materials and methods provided herein can be used to reduce or eliminate the number of cancer cells present within a mammal (e.g., a human) having cancer.
- a mammal in need thereof e.g., a mammal having cancer
- a mammal in need thereof can be administered an anti-BCMA antigen binding protein and a T cell engager to reduce the size (e.g., volume) of one or more tumors present within a mammal having cancer by, for example, at least about 10, 20, 30, 40, 50, 60, 70, 80, 90, 95, or more percent.
- the number of cancer cells present within a mammal being treated can be monitored. Any appropriate method can be used to determine whether or not the number of cancer cells present within a mammal is reduced. For example, imaging techniques can be used to assess the number of cancer cells present within a mammal.
- the materials and methods provided herein can be used to improve survival of a mammal (e.g., a human) having cancer.
- a mammal in need thereof e.g., a mammal having cancer
- the materials and methods described herein can be used to improve the survival of a mammal having cancer by, for example, by at least about 10, 20, 30, 40, 50, 60, 70, 80, 90, 95, or more percent.
- the materials and methods described herein can be used to improve the survival of a mammal having cancer by, for example, at least about 3 months (e.g., at least about 3 months, at least about 6 months, at least about 8 months, at least about 10 months, at least about 1 year, at least about 1.5 years, at least about 2 years, at least about 2.5 years, at least about 3 years, at least about 4 years, at least about 5 years, or more).
- the materials and methods provided herein can be used to treat of mammal (e.g., a human) having cancer such that the mammal can experience minimal, reduced, or no side effects.
- Examples of side effects that can be experienced by a mammal having cancer includes without limitation, one or more side effects selected from vision or eye changes such as findings on eye exam (keratopathy), decreased vision or blurred vision, nausea, low blood cell counts, fever, infusion-related reactions, tiredness, changes in kidney or liver function blood tests, thrombocytopenia, ocular toxicity (e.g., changes in corneal epithelium, dry eyes, irritation, redness, blurred vision, dry eyes, photophobia, and changes in visual acuity).
- a combination comprising an anti-BCMA antigen binding protein and a T cell engager for use in preventing and/or reducing ocular toxicity in a patient with cancers, such as multiple myeloma.
- ocular toxicity is prevented or reduced when compared to a patient treated with the anti-BCMA antigen binding protein alone (monotherapy).
- a combination disclosed herein for example an anti- BCMA antigen binding protein and a T cell engager, decreases/slows the decline from baseline of 1 line on Snellen Visual Acuity as compared to treatment with the anti-BCMA antigen binding protein alone.
- a combination disclosed herein, for example an anti-BCMA antigen binding protein and a T cell engager decreases/slows the decline from baseline of 2 or 3 lines on Snellen Visual Acuity as compared to treatment with the anti-BCMA antigen binding protein alone.
- a combination disclosed herein decreases/slows the decline from baseline by more than 3 lines on Snellen Visual Acuity as compared to treatment with the anti-BCMA antigen binding protein alone.
- a combination disclosed herein, for example an anti-BCMA antigen binding protein and a T cell engager decreases/slows the change from baseline on Snellen Visual Acuity as compared to treatment with the anti-BCMA antigen binding protein alone.
- a combination disclosed herein decreases/slows the decrease in logMAR (logarithm of the minimum angle of resolution) units from baseline as compared to treatment with the anti-BCMA antigen binding protein alone.
- a combination disclosed herein, for example an anti-BCMA antigen binding protein and a T cell engager decreases/slows or prevents the progression of mild superficial keratopathy, moderate superficial keratopathy, severe superficial keratopathy or corneal epithelial defect in a subject as compared to treatment with the anti-BCMA antigen binding protein alone.
- a combination disclosed herein prevents mild superficial keratopathy, moderate superficial keratopathy, severe superficial keratopathy or corneal epithelial defect in a subject as compared to treatment with the anti-BCMA antigen binding protein alone.
- Ocular toxicity refers to any unintended exposure of a therapeutic agent to ocular tissue. Ocular toxicity can include: changes in corneal epithelium, dry eyes, irritation, redness, blurred vision, dry eyes, photophobia, and/or changes in visual acuity. Ophthalmic examination may be conducted by an ophthalmologist or optometrist. An ophthalmic examination may include one or more of the following: 1.
- Best corrected visual acuity 2. Documentation of manifest refraction and the method used to obtain best corrected visual acuity, 3. Current glasses prescription (if applicable), 4. Intraocular pressure measurement, 5. Anterior segment (slit lamp) examination including fluorescein staining of the cornea and lens examination, 6. Dilated funduscopic examination, and/or 7. An ocular surface disease index (OSDI) which is visual function questionnaire that assess the impact of potential ocular change in vision on function and health-related quality of life. The above methods are known and practiced by those skilled in the art. The ophthalmic examination may occur before, during, and/or after treatment.
- OSDI ocular surface disease index
- T Cell Engager Disclosed herein are combinations comprising (i) a polypeptide comprising an anti- BCMA antigen binding protein or an ADC having binding specificity for a BCMA polypeptide and (ii) one or more T cell engagers.
- a T cell engager disclosed herein can be directed against a T cell and a tumor associated antigen. Any appropriate T cell engager(s) can be used to treat a mammal (e.g., a human) having cancer as described herein.
- a T cell engager can be a bispecific T cell engager (BiTE).
- a T cell engager described herein is a bispecific T cell engager that binds to CD3 and BCMA (CD3xBCMA).
- a T cell engager described herein is a bispecific T cell engager that binds to CD3 and GPRC5D (CD3xGPRC5D).
- a T cell engager described herein is a bispecific T cell engager that binds to CD3 and FcRH5 (CD3xFcRH5).
- a T cell engager can be a checkpoint-inhibitory T cell engager (CiTE).
- a T cell engager can be a simultaneous multiple interaction T cell engager (SMITE). In some cases, a T cell engager can be a trispecific killer engager (TriKE). In some embodiments, a T cell engager disclosed herein binds to BCMA. In some embodiments, a T cell engager disclosed herein binds to GPRC5D. In some embodiments, a T cell engager disclosed herein binds to FcRH5, In some embodiments, a T cell engager disclosed herein binds to CD38. In some embodiments, a T cell engager disclosed herein does not bind to BCMA. In some embodiments, a T cell engager disclosed herein does not bind to GPRC5D.
- SMITE simultaneous multiple interaction T cell engager
- TriKE trispecific killer engager
- a T cell engager disclosed herein does not bind to FcRH5. In some embodiments, a T cell engager disclosed herein does not bind to CD38. In some cases, a T cell engager disclosed herein binds human CD3. In some cases, CD3 is an activating T cell antigen.
- An “activating T cell antigen” as used herein can refer to an antigenic determinant expressed on the surface of a T lymphocyte, particularly a cytotoxic T lymphocyte, which is capable of inducing T cell activation upon interaction with an antigen binding molecule. Specifically, interaction of an antigen binding molecule with an activating T cell antigen may induce T cell activation by triggering the signaling cascade of the T cell receptor complex.
- a T cell engager disclosed herein is capable of inducing T cell activation.
- T cell activation can refer to one or more cellular response of a T lymphocyte, particularly a cytotoxic T lymphocyte, selected from: proliferation, differentiation, cytokine secretion, cytotoxic effector molecule release, cytotoxic activity, and expression of activation markers.
- a T cell engager disclosed herein is bispecific.
- a T cell engager disclosed herein binds at least one surface molecule that is expressed on human tumor cells.
- a T cell engager disclosed herein binds to BCMA, CD19, CD20, CD30, CD33, CD38, CD44, CD123, CD138, CEA, CLEC12A, CS-1, EGFR, EGFRvIII, EPCAM, DLL3, LGR5, MSLN, FOLR1, FOLR3, HER2, HM1.24, MCSP, PSMA or a combination thereof.
- T cell engagers that can be used to treat a mammal having cancer as described herein can comprise, without limitation, cevostamab, talquetamab, teclistimab, PF- 3135, TNB-383B, REGN5458, blinatumomab, solitomab, sibrotuzumab, fresolimumab, defactinib AZD4547 or a combination thereof.
- a combination disclosed herein comprises Teclistimab, PF-3135, TNB-383B, REGN5458 or a combination thereof.
- a combination disclosed herein comprises CC-93269, AMG701, JNJ-7957, GBR 1342 or a combination thereof.
- a combination disclosed herein comprises Talquetamab. In some cases, a combination disclosed herein comprises Cevostamab. In some cases, a composition disclosed herein comprises at least about 1 mg, 1.5 mg, 2mg, 3mg, 3.6mg, 10mg, 15mg, 20mg, 90 mg, or 132 mg Cevostamab.
- Cevostamab is a bispecific T-cell engager antibody composed of two single-chain variable fragments (scFv), one directed against the tumor-associated antigen Fc receptor-like protein 5 (FCRH5; CD307; FCRL5; IRTA2; BXMAS1) and one that is directed against the CD3 antigen found on T lymphocytes.
- scFv single-chain variable fragments
- the bispecific antibody binds to both the CD3 antigen on cytotoxic T lymphocytes (CTLs) and FCRH5 found on FCRH5-expressing tumor cells. In some cases, this activates and crosslinks CTLs with FCRH5-expressing tumor cells, which results in the CTL-mediated cell death of FCRH5-expressing tumor cells.
- CTLs cytotoxic T lymphocytes
- FCRH5 an immune receptor translocation- associated protein/Fc receptor homolog
- B-cell lineage marker is overexpressed on myeloma cells.
- the T cell engager comprises a scFv that is specific for FCRL5 and comprises a heavy chain variable region having at least 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% or 100% sequence identity to the amino acid sequence set out in SEQ ID NO:11.
- the T cell engager comprises a scFv that is specific for FCRL5 and comprises a light chain variable region having at least 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% or 100% sequence identity to the amino acid sequence set out in SEQ ID NO:12.
- the T cell engager comprises a scFv that is specific for FCRL5 and comprises a heavy chain variable region having the amino acid sequence set out in SEQ ID NO:11. In some embodiments, the T cell engager comprises a scFv that is specific for FCRL5 and comprises a light chain variable region having the amino acid sequence set out in SEQ ID NO:12. In some embodiments, the T cell engager comprises an activating T cell antigen binding scFv that is specific for CD3 and comprises a heavy chain variable region having at least 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% or 100% sequence identity to the amino acid sequence set out in SEQ ID NO:13.
- the T cell engager comprises an activating T cell antigen binding scFv that is specific for CD3 and comprises a light chain variable region having at least 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% or 100% sequence identity to the amino acid sequence set out in SEQ ID NO:14.
- the T cell engager comprises an activating T cell antigen binding scFv that is specific for CD3 and comprises a heavy chain variable region having the amino acid sequence set out in SEQ ID NO:13.
- the T cell engager comprises an activating T cell antigen binding scFv that is specific for CD3 and comprises a light chain variable region having the amino acid sequence set out in SEQ ID NO:14. In some embodiments, the T cell engager comprises the sequences out in SEQ ID NO:11, 12, 13 and 14. In some embodiments, the T cell engager comprises an amino acid sequence having at least 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% or 100% sequence identity to the heavy chain set out in SEQ ID NO:15 and 17.
- the T cell engager comprises an amino acid sequence having at least 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% or 100% sequence identity to the light chain set out in SEQ ID NO:16 and 18.
- the T cell engager comprises an amino acid sequence having a heavy chain set out in SEQ ID NO:15 and 17; and a light chain set out in SEQ ID NO:16 and 18.
- Table 4 Exemplarily T cell engager sequence.
- Talquetamab is a bispecific humanized monoclonal antibody against human CD3 and human G-protein coupled receptor family C group 5 member D (GPRC5D), a tumor-associated antigen.
- Talquetamab binds to both CD3 on T cells and GPRC5D expressed on certain tumor cells. In some cases, this results in the cross-linking of T cells and tumor cells, and induces a potent cytotoxic T-lymphocyte (CTL) response against GPRC5D-expressing tumor cells. In some cases, GPRC5D is overexpressed on certain tumors, such as multiple myeloma, while minimally expressed on normal, healthy cells, and plays a key role in tumor cell proliferation.
- Blinatumomab is a recombinant, single-chain, anti-CD19/anti-CD3 bispecific monoclonal antibody.
- Blinatumomab possesses two antigen-recognition sites, one for the CD3 complex , and one for CD19, a tumor-associated antigen overexpressed on the surface of B cells.
- Blinatumomab brings CD19-expressing tumor B-cells and cytotoxic T lymphocytes (CTLs) and helper T lymphocytes (HTLs) together that result in the CTL- and HTL-mediated cell death of CD19-expressing B-lymphocytes.
- CTLs cytotoxic T lymphocytes
- HTLs helper T lymphocytes
- Solitomab is a recombinant bispecific monoclonal antibody directed against both CD3 and epithelial cell adhesion molecule (EpCAM).
- Solitomab attaches to both CD3- expressing T lymphocytes and EpCAM-expressing tumor cells, thereby selectively cross-linking tumor and T lymphocytes. In some cases, this results in the recruitment of cytotoxic T lymphocytes (CTL) to T lymphocyte/tumor cell aggregates and the CTL-mediated death of EpCAM-expressing tumor cells.
- CTL cytotoxic T lymphocytes
- REGN5458 is a human bispecific T-cell engager antibody composed of two single-chain variable fragments (scFvs): one directed against BCMA and another directed against the CD3 antigen expressed on T lymphocytes.
- anti-BCMA/anti-CD3 REGN5458 binds to both CD3 on cytotoxic T lymphocytes (CTLs) and BCMA on BCMA- expressing tumor cells. In some cases, this activates and redirects CTLs to BCMA-expressing tumor cells, leading to CTL-mediated killing of BCMA-expressing tumor cells.
- TNB-383B is a bispecific antibody directed against the tumor associated antigen BCMA and against the CD3 antigen found on T lymphocytes. TNB-383B is composed of two aBCMA moieties in sequence on one arm, a single aCD3 arm, and a silenced IgG4 Fc.
- this bispecific antibody binds to both CD3 on cytotoxic T lymphocytes (CTLs) and BCMA found on BCMA-expressing tumor cells. In some cases, this activates and redirects CTLs to BCMA- expressing tumor cells, which results in the CTL-mediated cell death of BCMA-expressing tumor cells.
- CTLs cytotoxic T lymphocytes
- an anti-BCMA antigen binding protein comprising CDRH1 according to SEQ ID NO:1; CDRH2 according to SEQ ID NO:2; CDRH3 according to SEQ ID NO:3; CDRL1 according to SEQ ID NO:4; CDRL2 according to SEQ ID NO:5; and CDRL3 according to SEQ ID NO:6; and a T cell engager, for use in the treatment of cancer.
- an anti-BCMA antigen binding protein comprising heavy chain variable region (VH) according to SEQ ID NO:7; and a light chain variable region (VL) according to SEQ ID NO:8; and a T cell engager, for use in the treatment of cancer.
- combinations comprising a therapeutically effective dose of an anti-BCMA antigen binding protein comprising heavy chain (H) according to SEQ ID NO:9 and a light chain (L) according to SEQ ID NO:10; and a T cell engager, for use in the treating cancer.
- an anti-BCMA antigen binding protein disclosed herein comprises the heavy chain variable region of SEQ ID NO:19, 23 or 27.
- an anti-BCMA antigen binding protein disclosed herein comprises the heavy chain variable region of SEQ ID NO:19 and the light chain variable region of SEQ ID NO:20, the heavy chain variable region of SEQ ID NO:23 and the light chain variable region of SEQ ID NO:24, or the heavy chain variable region of SEQ ID NO:27 and the light chain variable region of SEQ ID NO:28.
- an anti- BCMA antigen binding protein disclosed herein comprises the heavy chain region of SEQ ID NO:21 and the light chain of SEQ ID NO:22, the heavy chain region of SEQ ID NO: 25 and the light chain of SEQ ID NO:26, the heavy chain region of SEQ ID NO:30 and the light chain of SEQ ID NO:31 or the heavy chain region of SEQ ID NO:32 and the light chain of SEQ ID NO:33.
- an anti-BCMA antigen binding protein disclosed herein is an scFV-fc comprising SEQ ID NO:29.
- combinations disclosed herein, when in a pharmaceutical preparation, is present in unit dose form. In some embodiments, the dosage regimen will be determined by a medical profession and/or clinical factors.
- dosages for any one patient depend upon many factors, including the patient's size, body surface area, age, the combination to be administered, sex, time and route of administration, general health, and other drugs being administered concurrently. Exemplary doses can vary according to the size and health of the individual being treated, as well as the condition being treated.
- Suitable doses of the anti-BCMA antigen binding proteins described herein may be calculated for patients according to their weight, for example suitable doses may be in the range of about 0.1 mg/kg to about 20 mg/kg, for example about 1 mg/kg to about 20 mg/kg, for example about 10 mg/kg to about 20 mg/kg or for example about 1 mg/kg to about 15 mg/kg, for example about 10 mg/kg to about 15 mg/kg.
- the therapeutically effective dose of the anti-BCMA antigen binding protein is in the range of about 0.03 mg/kg to about 4.6 mg/kg.
- the therapeutically effective dose of the anti-BCMA antigen binding protein is 0.03 mg/kg, 0.06 mg/kg, 0.12 mg/kg, 0.24 mg/kg, 0.48 mg/kg, 0.96 mg/kg, 1.92 mg/kg, 2.5 mg/kg, 3.4 mg/kg, or 4.6 mg/kg. In yet another embodiment, the therapeutically effective dose of the anti-BCMA antigen binding protein is 1.9 mg/kg, 2.5 mg/kg or 3.4 mg/kg. In some embodiments, the therapeutically effective dose of the anti-BCMA antigen binding protein is 0.95 mg/kg. In some embodiments, the therapeutically effective dose of the anti-BCMA antigen binding protein is 1.0 mg/kg.
- the therapeutically effective dose of the anti-BCMA antigen binding protein is 1.4 mg/kg. In some embodiments, the therapeutically effective dose of the anti-BCMA antigen binding protein is 1.9 mg/kg. In some embodiments, the therapeutically effective dose of the anti-BCMA antigen binding protein is 1.92 mg/kg. In some embodiments, the therapeutically effective dose of the anti-BCMA antigen binding protein is 2.5 mg/kg. In some embodiments, the therapeutically effective dose of the anti-BCMA antigen binding protein is 3.4 mg/kg. In some embodiments, the therapeutically effective dose of the anti-BCMA antigen binding protein is administered to the subject every week. In some embodiments, the therapeutically effective dose of the anti-BCMA antigen binding protein is administered to the subject every 2 weeks.
- the therapeutically effective dose of the anti-BCMA antigen binding protein is administered to the subject every 3 weeks. In some embodiments, the therapeutically effective dose of the anti- BCMA antigen binding protein is administered to the subject every 4 weeks. In some embodiments, the therapeutically effective dose of the anti-BCMA antigen binding protein is administered to the subject every 5 weeks. In some embodiments, the therapeutically effective dose of the anti-BCMA antigen binding protein is administered to the subject every 6 weeks. In some embodiments, dosage of the therapeutically effective dose of the anti-BCMA antigen binding protein is step-down to a lower dose described herein following a first administration.
- 3.4 mg/kg dosage of the therapeutically effective dose of the anti-BCMA antigen binding protein is step-down to a 1.9 mg/kg dose, 1.4 mg/kg or less.
- a 2.5 mg/kg dosage of the therapeutically effective dose of the anti-BCMA antigen binding protein is step-down to a 1.9 mg/kg dose, 1.4 mg/kg or less.
- the therapeutically effective dose of the anti-BCMA antigen binding protein is administered to the subject on day 1, day 8 and thereafter every 3-12 weeks.
- Suitable doses of a T cell engager described herein may be calculated for patients according to their weight, for example suitable doses may be in the range of about 0.1 mg/kg to about 30 mg/kg, for example about 5 mg/kg to about 20 mg/kg, or for example about 10 mg/kg to about 20 mg/kg.
- the therapeutically effective dose of the T cell engager is about 5 mg/kg, about 6 mg/kg, about 7 mg/kg, about 8 mg/kg, about 9 mg/kg, about 10 mg/kg, about 11 mg/kg, about 12 mg/kg, about 13 mg/kg, about 14 mg/kg, about 15 mg/kg, about 16 mg/kg, about 17 mg/kg, about 18 mg/kg, about 19 mg/kg, or about 20 mg/kg.
- a combination comprising belantamab mafodotin and a T cell engager for use in the treatment of cancer, wherein belantamab mafodotin is administered at least about 0.5 mg/kg, 0.95 mg/kg, 1 mg/kg, 1.4 mg/kg, 1.9 mg/kg, 1.92 mg/kg, 2.5 mg/kg, or 3.4 mg/kg and the T cell engager (for example Cevostamab) is administered at least about 2 mg, 3 mg, 3.6 mg, 10 mg, 15 mg, 20 mg, 50 mg, 100 mg, or 150 mg.
- a combination comprising belantamab mafodotin and a T cell engager for use in the treatment of cancer, wherein belantamab mafodotin is administered at 0.5 mg/kg, 0.95 mg/kg, 1 mg/kg, 1.4 mg/kg, 1.9 mg/kg, 1.92 mg/kg, 2.5 mg/kg, or 3.4 mg/kg on day 1 of a 21-day cycle and the T cell engager (for example Cevostamab) is administered at 2 mg, 3 mg, 3.6 mg, 10 mg, 15 mg, 20 mg, 50 mg, 100 mg, or 150 mg on day 1 of a 21-day cycle.
- T cell engager for example Cevostamab
- a combination comprising belantamab mafodotin and a T cell engager for use in the treatment of cancer, wherein belantamab mafodotin is administered at 0.5 mg/kg, 0.95 mg/kg, 1 mg/kg, 1.4 mg/kg, 1.9 mg/kg, 1.92 mg/kg, 2.5 mg/kg, or 3.4 mg/kg on day 1 of a 21-day cycle and the T cell engager (for example Cevostamab) is administered at 2 mg, 3 mg, 3.6 mg, 10 mg, 15 mg, 20 mg, 50 mg, 100 mg, or 150 mg on day 1 of a 8-day cycle.
- a combination comprising belantamab mafodotin and a T cell engager for use in the treatment of cancer, wherein belantamab mafodotin is administered at 0.5 mg/kg, 0.95 mg/kg, 1 mg/kg, 1.4 mg/kg, 1.9 mg/kg, 1.92 mg/kg, 2.5 mg/kg, or 3.4 mg/kg on day 1 of a 21-day cycle and the T cell engager (for example Cevostamab) is administered at 2 mg, 3 mg, 3.6 mg, 10 mg, 15 mg, 20 mg, 50 mg, 100 mg, or 150 mg on day 1 of a 15-day cycle.
- a combination comprising belantamab mafodotin and a T cell engager for use in the treatment of cancer, wherein belantamab mafodotin is administered at 0.5 mg/kg, 0.95 mg/kg, 1 mg/kg, 1.4 mg/kg, 1.9 mg/kg, 1.94, mg/kg, 2.5 mg/kg, or 3.4 mg/kg and half of the dose is administered on day 1 and half of the dose is administered on day 8 of a 21-day cycle; and a T cell engager (for example Cevostamab) is administered at 2 mg, 3 mg, 3.6 mg, 10 mg, 15 mg, 20 mg, 50 mg, 100 mg, or 150 mg.
- a T cell engager for example Cevostamab
- a combination comprising belantamab mafodotin and a T cell engager for use in the treatment of multiple myeloma, wherein belantamab mafodotin is administered at 0.5 mg/kg, 0.95 mg/kg, 1 mg/kg, 1.4 mg/kg, 1.9 mg/kg, 1.92 mg/kg, 2.5 mg/kg, or 3.4 mg/kg and the T cell engager (for example Cevostamab) is administered at 2 mg, 3 mg, 3.6 mg, 10 mg, 15 mg, 20 mg, 50 mg, 100 mg, or 150 mg.
- a combination comprising belantamab mafodotin and a T cell engager for use in the treatment of multiple myeloma, wherein belantamab mafodotin is administered at 0.5 mg/kg, 0.95 mg/kg, 1 mg/kg, 1.4 mg/kg, 1.9 mg/kg, 1.92 mg/kg, 2.5 mg/kg, or 3.4 mg/kg and half of the dose is administered on day 1 and half of the dose is administered on day 8 of a 21-day cycle; and the T cell engager (for example Cevostamab) is administered at 2 mg, 3 mg, 3.6 mg, 10 mg, 15 mg, 20 mg, 50 mg, 100 mg, or 150 mg on days 1-7 of a 21-day cycle.
- belantamab mafodotin is administered at 0.5 mg/kg, 0.95 mg/kg, 1 mg/kg, 1.4 mg/kg, 1.9 mg/kg, 1.92 mg/kg, 2.5 mg/kg, or 3.4 mg/kg and half of the dose is administered
- the subject has received at least one previous cancer treatment.
- the therapeutically effective dose of the combination is administered to the subject at least about once every 1-60 days.
- the therapeutically effective dose of the composition is administered to the subject at least about once every 21 days.
- the therapeutically effective dose of the composition is administered to the subject at least about once every 8 days.
- Route of Administration In some cases, (i) a polypeptide comprising an anti-BCMA antigen binding protein or an ADC having binding specificity for a BCMA polypeptide and (ii) one or more T cell engagers can be administered to a mammal at the same time (e.g., in a single composition).
- a polypeptide comprising an anti-BCMA antigen binding protein or an ADC having binding specificity for a BCMA polypeptide and (ii) one or more T cell engagers can be administered to a subject separately. When administered separately, this may occur simultaneously or sequentially in any order (by the same or by different routes of administration). Such sequential administration may be close in time or remote in time.
- the dose of a therapeutic agents of the combination or pharmaceutical composition thereof and the further therapeutically active agent(s) and the relative timings of administration will be selected in order to achieve the desired combined therapeutic effect.
- the dosage is administered a single time or multiple times, for example daily, weekly, biweekly, or monthly, hourly, or is administered upon recurrence, relapse or progression of a disease or condition being treated.
- administration of a dose may be by slow continuous infusion over a period of from about 2 to about 24 hours, such as from about 2 to about 12 hours, or from about 2 to about 6 hours.
- a pharmaceutical composition disclosed herein comprises the combination for parenteral, transdermal, intraluminal, intraarterial, intrathecal and/or intranasal administration or by direct injection into tissue.
- the pharmaceutical composition is administered to a patient via infusion or injection.
- compositions comprising a BCMA binding protein and a T cell engager for intravenous administration.
- pharmaceutical compositions comprising a BCMA binding protein and a T cell engager for subcutaneous administration.
- a pharmaceutical composition described herein is administered to a subject transarterially, subcutaneously, intradermally, intratumorally, intranodally, intramedullary, intramuscularly, by intravenous (i.v.) injection, by intravenous (i.v.) infusion, or intraperitoneally.
- the combination is administered to a subject by intradermal or subcutaneous injection.
- one or more therapeutic agents of a combination are administered intravenously.
- one or more therapeutic agents of a combination are administered intratumorally. In another embodiment, one or more therapeutic agents of a combination are administered orally. In another embodiment, one or more therapeutic agents of a combination are administered systemically, e.g., intravenously, and one or more other therapeutic agents of a combination of are administered intratumorally. In another embodiment, all therapeutic agents of a combination disclosed herein are administered systemically, e.g., intravenously. In an alternative embodiment, all therapeutic agents of the combination described herein are administered intratumorally. In any of the embodiments, e.g., in this paragraph, the therapeutic agents of the disclosure are administered as one or more pharmaceutical compositions.
- a pharmaceutical composition is prepared by per se known methods for the preparation of pharmaceutically acceptable compositions that are administered to subjects, such that an effective quantity of a BCMA binding protein + T cell engager is combined in a mixture with a pharmaceutically acceptable carrier.
- suitable carriers are described, for example, in Remington's Pharmaceutical Sciences (Remington's Pharmaceutical Sciences, 20 th ed., Mack Publishing Company, Easton, Pa., USA, 2000).
- the compositions may include, albeit not exclusively, solutions of the substances in association with one or more pharmaceutically acceptable carriers or diluents, and contained in buffered solutions with a suitable pH and iso-osmotic with the physiological fluids.
- a pharmaceutical composition disclosed herein is acidic.
- a pharmaceutical composition disclosed herein is basic.
- a pharmaceutical composition can have a pH of about 1, 1.5, 2, 2.5, 3, 3.5, 4, 4.5, 5, 5.5, 6, 6.5, 7, 7.5, 8, 8.5, 9, 9.5, 10, 10.5, 11, 11.5, 12, 12.5, 13, 13.5, or about 14.
- suitable pharmaceutically acceptable carriers include essentially chemically inert and nontoxic compositions that do not interfere with the effectiveness of the biological activity of the pharmaceutical composition.
- Suitable pharmaceutical carriers include, but are not limited to, water, saline solutions, glycerol solutions, N-(1(2,3- dioleyloxy)propyl)N,N,N-trimethylammonium chloride (DOTMA), diolesylphosphotidyl- ethanolamine (DOPE), and liposomes.
- DOTMA N-(1(2,3- dioleyloxy)propyl)N,N,N-trimethylammonium chloride
- DOPE diolesylphosphotidyl- ethanolamine
- liposomes examples include, but are not limited to, water, saline solutions, glycerol solutions, N-(1(2,3- dioleyloxy)propyl)N,N,N-trimethylammonium chloride (DOTMA), diolesylphosphotidyl- ethanolamine (DOPE), and liposomes.
- such compositions contain a therapeutically effective amount of a BCMA binding protein and T cell
- compositions may include, without limitation, lyophilized powders or aqueous or non-aqueous sterile injectable solutions or suspensions, which may further contain antioxidants, buffers, bacteriostats and solutes that render the compositions substantially compatible with the tissues or the blood of an intended recipient.
- Other components that may be present in such compositions include water, surfactants (such as Tween), alcohols, preservatives, polyols, glycerin and vegetable oils, for example.
- Extemporaneous injection solutions and suspensions may be prepared from sterile powders, granules, tablets, or concentrated solutions or suspensions.
- a pharmaceutical composition disclosed herein may be formulated into a variety of forms and administered by a number of different means.
- a pharmaceutical formulation can be administered orally, rectally, or parenterally, in formulations containing conventionally acceptable carriers, adjuvants, and vehicles as desired.
- parenteral as used herein includes subcutaneous, intravenous, intramuscular, or intrasternal injection and infusion techniques.
- Administration includes injection or infusion, including intra-arterial, intracardiac, intracerebroventricular, intradermal, intraduodenal, intramedullary, intramuscular, intraosseous, intraperitoneal, intrathecal, intravascular, intravenous, intravitreal, epidural and subcutaneous), inhalational, transdermal, transmucosal, sublingual, buccal and topical (including epicutaneous, dermal, enema, eye drops, ear drops, intranasal, vaginal) administration.
- a route of administration is via an injection such as an intramuscular, intravenous, subcutaneous, or intraperitoneal injection.
- Liquid formulations may include an oral formulation, an intravenous formulation, an intranasal formulation, an ocular formulation, an otic formulation, an aerosol, and the like.
- a combination of various formulations is administered.
- a composition is formulated for an extended release profile.
- Pharmaceutical compositions of the disclosure can be administered in combination with other therapeutics or treatments.
- a treatment for a subject can be a surgery, radiation, chemotherapy, a nutrition regime, a physical activity, an immunotherapy, a pharmaceutical composition, a cell transplantation, a blood fusion, or any combination thereof.
- the combination disclosed herein is administered to a mammal having cancer together with one or more additional agents/therapies used to treat cancer.
- additional agents/therapies used to treat cancer include, without limitation, surgery, radiation therapies, chemotherapies, targeted therapies (e.g., monoclonal antibody therapies), hormonal therapies, angiogenesis inhibitors, immunosuppressants, checkpoint blockade therapies (e.g., anti-PD-1 antibody therapy, anti-PD-L1 antibody therapy, and/or anti-CTLA4 antibody therapy), bone marrow transplants.
- a combination/formulation disclosed herein is stable.
- a "stable" formulation is one in which the combination therein essentially retains its physical and/or chemical stability and/or biological activity upon storage.
- Stability can be measured at a selected temperature for a selected time period.
- the formulation is stable at ambient temperature or at 40 ° C for at least 1 month and/or stable at 2- 8°C for at least 1 to 2 years.
- the formulation is stable following freezing (e.g. to -70 0 C) and thawing.
- a protein "retains its physical stability” in a formulation if it shows little to no change in aggregation, precipitation and/or denaturation as observed by visual examination of color and/or clarity, or as measured by UV light scattering (measures visible aggregates) or size exclusion chromatography (SEC). SEC measures soluble aggregates that are not necessarily a precursor for visible aggregates.
- a protein "retains its chemical stability” in a formulation if the chemical stability at a given time is such that the protein is considered to retain its biological activity. Chemically degraded species may be biologically active and chemically unstable. Chemical stability can be assessed by detecting and quantifying chemically altered forms of the protein. Chemical alteration may involve size modification (e.g.
- a BCMA binding protein "retains its biological activity" in a pharmaceutical formulation, if the biological activity of the BCMA binding protein at a given time is within about 10% (within the errors of the assay) of the biological activity exhibited at the time the pharmaceutical formulation was prepared as determined in an antigen binding assay, for example.
- a T cell engager "retains its biological activity" in a pharmaceutical formulation, if the biological activity of the T cell engager at a given time is within about 10% (within the errors of the assay) of the biological activity exhibited at the time the pharmaceutical formulation was prepared as determined in an antigen binding assay, for example.
- a buffer disclosed herein refers to a buffered solution that resists changes in pH by the action of its acid-base conjugate components.
- a buffer can be phosphate, citrate and other organic acids.
- a buffer is selected from the group consisting of sodium acetate, sodium carbonate, citrate, glycylglycine, histidine, glycine, lysine, arginine, sodium dihydrogen phosphate, disodium hydrogen phosphate, sodium phosphate, sodium citrate, sodium borate, tris(hydroxymethyl)-aminomethan, bicine, tricine, malic acid, succinate, maleic acid, fumaric acid, tartaric acid, aspartic acid or mixtures thereof.
- a composition disclosed herein can comprise antioxidants including ascorbic acid and/or methionine.
- a composition disclosed herein comprises a preservative.
- a preservative is a compound which can be included in a formulation to essentially reduce microbial including bacterial action therein, thus facilitating the production of a multi-use formulation, for example.
- potential preservatives include octadecyldimethylbenzyl ammonium chloride; hexamethonium chloride; benzalkonium chloride, benzethonium chloride; phenol, butyl or benzyl alcohol; alkyl parabens such as methyl or propyl paraben; catechol; resorcinol; cyclohexanol; 3-pentanol; and m-cresol); low molecular weight (less than about 10 residues) polypeptides; proteins, such as serum albumin, gelatin, or immunoglobulins; hydrophilic polymers such as polyvinylpyrrolidone; amino acids such as glycine, glutamine, asparagine, histidine, arginine, or
- sustained-release matrices include polyesters, hydrogels (for example, poly(2-hydroxyethyl-methacrylate), or poly(vinylalcohol)), polylactides, copolymers of L-glutamic acid and ⁇ ethyl-L-glutamate, non- degradable ethylene-vinyl acetate, degradable lactic acid-glycolic acid copolymers such as the LUPRON DEPOTTM (injectable microspheres composed of lactic acid-glycolic acid copolymer and leuprolide acetate), and poly-D-( ⁇ )-3-hydroxybutyric acid.
- polyesters for example, poly(2-hydroxyethyl-methacrylate), or poly(vinylalcohol)
- polylactides copolymers of L-glutamic acid and ⁇ ethyl-L-glutamate
- non- degradable ethylene-vinyl acetate non- degradable ethylene-vinyl acetate
- compositions comprising a BCMA binding protein and a T cell engager which is present in a concentration from 1 mg/ml to 500 mg/ml, and wherein the pharmaceutical composition has a pH from 2.0 to 10.0.
- the pharmaceutical composition may further comprise a buffer system, preservative(s), tonicity agent(s), chelating agent(s), stabilizers and surfactants.
- the pharmaceutical composition is an aqueous formulation, for example, formulation comprising water. Such formulation is typically a solution or a suspension.
- the pharmaceutical formulation is an aqueous solution.
- an aqueous formulation is a formulation comprising at least 50 %w/w water.
- an aqueous solution is defined as a solution comprising at least 50 %w/w water.
- the pharmaceutical composition is a stable liquid aqueous pharmaceutical formulation comprising a combination described herein.
- the pharmaceutical compositions may also comprise additional stabilizing agents, that may further enhance stability of a therapeutically active combination.
- Stabilizing agents of can include, but are not limited to, methionine and EDTA, which protect polypeptides against methionine oxidation, and a nonionic surfactant, which protects polypeptides against aggregation associated with freeze -thawing or mechanical shearing.
- the pharmaceutical composition may further comprise a surfactant.
- the surfactant may be selected from a detergent, ethoxylated castor oil, polyglycolyzed glycerides, acetylated monoglycerides, sorbitan fatty acid esters, polyoxypropylene-polyoxyethylene block polymers (eg.
- poloxamers such as PLURONIC F68, poloxamer 188 and 407, Triton X-100
- polyoxyethylene sorbitan fatty acid esters polyoxyethylene and polyethylene derivatives such as alkylated and alkoxylated derivatives (tweens, e.g., Tween-20, Tween-40, Tween-80 and Brij-35), monoglycerides or ethoxylated derivatives thereof, diglycerides or polyoxyethylene derivatives thereof, alcohols, glycerol, lectins and phospholipids (e.g., phosphatidyl serine, phosphatidyl choline, phosphatidyl ethanolamine, phosphatidyl inositol, diphosphatidyl glycerol and sphingomyelin), derivates of phospholipids (e.g., dipalmitoyl phosphatidic acid) and lysophospholipids (e.g.,
- Kits A kit-of-parts comprising a pharmaceutical composition together with instructions for use is further provided.
- the kit-of-parts may comprise reagents in predetermined amounts with instructions for use.
- kids comprising a BCMA binding protein and a T cell engager disclosed herein.
- a kit may include a plurality of syringes, ampules, foil packets, or blister packs, each containing a single unit dose of a kit component described herein.
- Containers of a kit may be airtight, waterproof (e.g., impermeable to changes in moisture or evaporation), and/or light-tight.
- a kit may include a device suitable for administration of the component, e.g., a syringe, inhalant, pipette, forceps, measured spoon, dropper (e.g., eye dropper), swab (e.g., a cotton swab or wooden swab), or any such delivery device.
- the device may be a medical implant device, e.g., packaged for surgical insertion.
- a kit disclosed herein may comprise one or more reagents or instruments which enable the method to be carried out.
- instructions for use may be provided in a kit.
- instructions may be present in the kit in a variety of forms, such as printed information on a suitable medium or substrate (e.g., a piece or pieces of paper on which the information is printed), in the packaging of the kit, in a package insert, etc.
- instructions for use can be provided on a computer readable medium (e.g., jump/thumb drive, CD, etc.), on which the information has been recorded or at a website address which may be used via the internet to access the information at a website.
- a pre-filled syringe or autoinjector device comprising a BCMA antigen binding protein, a T cell engager or a combination described herein.
- a combination stored in a container, pre-filled syringe, injector or autoinjector device contains a BCMA antigen binding protein and a T cell engager disclosed herein.
- Example 1 Treating Cancer A subject will be identified as having cancer. The subject will be administered a combination comprising (a) an anti-BCMA antigen binding protein or an anti-BCMA ADC and (b) a T cell engager.
- Example 2 Treating Cancer A subject will be identified as having cancer. The subject will be administered (a) an anti-BCMA antigen binding protein or an anti-BCMA ADC and (b) a T cell engager in separate compositions that will be co-administered.
- the subject having cancer will be co- administered a first composition including one or more ADCs having binding specificity for a BCMA polypeptide and a second composition including one or more T cell engagers.
- Example 3 Treating Cancer A subject will be identified as having cancer. The subject will be administered (a) an anti-BCMA antigen binding protein or an anti-BCMA ADC and (b) a T cell engager in separate compositions that will be separately administered.
- the subject having cancer will be separately administered a first composition including one or more ADCs having binding specificity for a BCMA polypeptide and a second composition including one or more T cell engagers.
- Example 4 Treating Cancer A subject will be identified as having cancer.
- Embodiments 1. A combination comprising: a. an anti-BCMA antigen binding protein; and b. a T cell engager that binds to CD3. 2. The combination of embodiment 1, wherein the anti-BCMA antigen binding protein comprises an antibody. 3. The combination of embodiment 2, wherein the antibody is a monoclonal antibody. 4.
- the anti-BCMA antigen binding protein comprises a CDRH1 comprising the amino acid sequence set out in SEQ ID NO:1; a CDRH2 comprising the amino acid sequence set out in SEQ ID NO:2; a CDRH3 comprising the amino acid sequence set out in SEQ ID NO:3; a CDRL1 comprising the amino acid sequence set out in SEQ ID NO:4; a CDRL2 comprising the amino acid sequence set out in SEQ ID NO:5; and a CDRL3 comprising the amino acid sequence set out in SEQ ID NO:6.
- the anti-BCMA antigen binding protein comprises a heavy chain variable region (VH) comprising the amino acid sequence set out in SEQ ID NO:7; and a light chain variable region (VL) comprising the amino acid sequence set out in SEQ ID NO:8.
- VH heavy chain variable region
- VL light chain variable region
- the anti-BCMA antigen binding protein comprises a heavy chain (H) comprising the amino acid sequence set out in SEQ ID NO:9 and a light chain (L) comprising the amino acid sequence set out in SEQ ID NO:10.
- the combination of any one of embodiments 1-9, wherein the anti-BCMA antigen binding protein is an immunoconjugate.
- cytotoxin is paclitaxel, docetaxel, CC-1065, SN-38, topotecan, morpholino-doxorubicin, rhizoxin, cyanomorpholino-doxorubicin, dolastatin-10, echinomycin, combretatstatin, calicheamicin, netropsin, an auristatin, a maytansinoid, a calicheamicin, AFP, MMAP, MMAE, AEB, AEVB, vincristine, vinblastine, vindesine, vinorelbine, VP-16, camptothecin, epothilone A, epothilone B, nocodazole, colchicines, colcimid, estramustine, cemadotin, discodermolide, maytansinol, maytansine, DM1, DM2, DM3, DM4 or eleutherobin.
- any one of embodiments 1-14, wherein the anti-BCMA antigen binding protein is belantamab mafodotin.
- the combination comprises at least about 0.5 mg/kg, 0.95 mg/kg, 1 mg/kg, 1.4 mg/kg, 1.9 mg/kg, 1.92 mg/kg, 2.5 mg/kg or about 3.4 mg/kg belantamab mafodotin.
- the T cell engager is a bispecific T cell engager. 18.
- T cell engager is an anti- GPRC5D T cell engager.
- the combination of embodiment 22, wherein the T cell engager is Talquetamab.
- 24. The combination of any one of embodiments 1-17, wherein the T cell engager is an anti- BCMA T cell engager.
- 25. The combination of any one of embodiments 1-17, wherein the T cell engager is selected from the group consisting of Teclistimab, PF-3135, TNB-383B, and REGN5458.
- T cell engager is selected from the group consisting of CC-93269, AMG701, JNJ-7957, and GBR 1342.
- ocular toxicity is at least one of: changes in corneal epithelium, dry eyes, irritation, redness, blurred vision, dry eyes, photophobia, or changes in visual acuity.
- 41. The method of any one of embodiments 38-40, wherein the ocular toxicity is measured by at least one of the following methods: best corrected visual acuity, documentation of manifest refraction and the method used to obtain best corrected visual acuity, current glasses prescription (if applicable), intraocular pressure measurement, anterior segment (slit lamp) examination including fluorescein staining of the cornea and lens examination, dilated funduscopic examination, or an ocular surface disease index (OSDI). 42.
- any one of embodiments 31-41 wherein the anti-BCMA antigen binding protein is administered to the subject in a dose of at least about 0.5 mg/kg, 0.95 mg/kg, 1.25 mg/kg, 1.4 mg/kg, 1.7 mg/kg, 1.9 mg/kg, 1.92 mg/kg, 2.5 mg/kg or 3.4 mg/kg.
- 43. A combination of any one of embodiments 1-30, for use in the manufacture of a medicament for treatment of cancer.
- 44. A combination of any one of embodiments 1-30, for use in treatment of cancer.
- a kit for use in treatment of cancer comprising: a. the combination of any one of embodiments 1-30; and b. instructions for use in the treatment of cancer.
- a pre-filled syringe or autoinjector device comprising the combination of any one of embodiments 1-30.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Immunology (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Peptides Or Proteins (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Medicinal Preparation (AREA)
- Medicines Containing Plant Substances (AREA)
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA3220227A CA3220227A1 (en) | 2021-05-28 | 2022-05-26 | Combination therapies for treating cancer |
| EP22740439.9A EP4347656A1 (en) | 2021-05-28 | 2022-05-26 | Combination therapies for treating cancer |
| BR112023024804A BR112023024804A2 (en) | 2021-05-28 | 2022-05-26 | COMBINATION THERAPIES TO TREAT CANCER |
| US18/562,868 US20250263495A1 (en) | 2021-05-28 | 2022-05-26 | Combination Therapies for Treating Cancer |
| JP2023573231A JP2024521187A (en) | 2021-05-28 | 2022-05-26 | Combination Therapy for the Treatment of Cancer |
| CN202280038099.7A CN117396513A (en) | 2021-05-28 | 2022-05-26 | Combination therapy for cancer |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202163194547P | 2021-05-28 | 2021-05-28 | |
| US63/194,547 | 2021-05-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022248870A1 true WO2022248870A1 (en) | 2022-12-01 |
Family
ID=82483166
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/GB2022/051348 Ceased WO2022248870A1 (en) | 2021-05-28 | 2022-05-26 | Combination therapies for treating cancer |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20250263495A1 (en) |
| EP (1) | EP4347656A1 (en) |
| JP (1) | JP2024521187A (en) |
| CN (1) | CN117396513A (en) |
| BR (1) | BR112023024804A2 (en) |
| CA (1) | CA3220227A1 (en) |
| WO (1) | WO2022248870A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024259279A3 (en) * | 2023-06-14 | 2025-05-15 | Interius Biotherapeutics, Inc. | Pseudotyped viral particles comprising engineered chimeric receptor proteins |
Citations (86)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3896111A (en) | 1973-02-20 | 1975-07-22 | Research Corp | Ansa macrolides |
| US4137230A (en) | 1977-11-14 | 1979-01-30 | Takeda Chemical Industries, Ltd. | Method for the production of maytansinoids |
| US4151042A (en) | 1977-03-31 | 1979-04-24 | Takeda Chemical Industries, Ltd. | Method for producing maytansinol and its derivatives |
| US4248870A (en) | 1978-10-27 | 1981-02-03 | Takeda Chemical Industries, Ltd. | Maytansinoids and use |
| US4256746A (en) | 1978-11-14 | 1981-03-17 | Takeda Chemical Industries | Dechloromaytansinoids, their pharmaceutical compositions and method of use |
| US4260608A (en) | 1978-11-14 | 1981-04-07 | Takeda Chemical Industries, Ltd. | Maytansinoids, pharmaceutical compositions thereof and methods of use thereof |
| US4265814A (en) | 1978-03-24 | 1981-05-05 | Takeda Chemical Industries | Matansinol 3-n-hexadecanoate |
| US4294757A (en) | 1979-01-31 | 1981-10-13 | Takeda Chemical Industries, Ltd | 20-O-Acylmaytansinoids |
| US4307016A (en) | 1978-03-24 | 1981-12-22 | Takeda Chemical Industries, Ltd. | Demethyl maytansinoids |
| US4308268A (en) | 1979-06-11 | 1981-12-29 | Takeda Chemical Industries, Ltd. | Maytansinoids, pharmaceutical compositions thereof and method of use thereof |
| US4308269A (en) | 1979-06-11 | 1981-12-29 | Takeda Chemical Industries, Ltd. | Maytansinoids, pharmaceutical compositions thereof and method of use thereof |
| US4309428A (en) | 1979-07-30 | 1982-01-05 | Takeda Chemical Industries, Ltd. | Maytansinoids |
| US4313946A (en) | 1981-01-27 | 1982-02-02 | The United States Of America As Represented By The Secretary Of Agriculture | Chemotherapeutically active maytansinoids from Trewia nudiflora |
| US4315929A (en) | 1981-01-27 | 1982-02-16 | The United States Of America As Represented By The Secretary Of Agriculture | Method of controlling the European corn borer with trewiasine |
| US4317821A (en) | 1979-06-08 | 1982-03-02 | Takeda Chemical Industries, Ltd. | Maytansinoids, their use and pharmaceutical compositions thereof |
| US4322348A (en) | 1979-06-05 | 1982-03-30 | Takeda Chemical Industries, Ltd. | Maytansinoids |
| US4331598A (en) | 1979-09-19 | 1982-05-25 | Takeda Chemical Industries, Ltd. | Maytansinoids |
| US4362663A (en) | 1979-09-21 | 1982-12-07 | Takeda Chemical Industries, Ltd. | Maytansinoid compound |
| US4364866A (en) | 1979-09-21 | 1982-12-21 | Takeda Chemical Industries, Ltd. | Maytansinoids |
| US4371533A (en) | 1980-10-08 | 1983-02-01 | Takeda Chemical Industries, Ltd. | 4,5-Deoxymaytansinoids, their use and pharmaceutical compositions thereof |
| US4424219A (en) | 1981-05-20 | 1984-01-03 | Takeda Chemical Industries, Ltd. | 9-Thiomaytansinoids and their pharmaceutical compositions and use |
| US4450254A (en) | 1980-11-03 | 1984-05-22 | Standard Oil Company | Impact improvement of high nitrile resins |
| US4703039A (en) | 1984-04-10 | 1987-10-27 | New England Deaconess Hospital Corporation | Method of producing biologically active molecules having extended life time |
| EP0307434A1 (en) | 1987-03-18 | 1989-03-22 | Medical Res Council | Altered antibodies. |
| US4975278A (en) | 1988-02-26 | 1990-12-04 | Bristol-Myers Company | Antibody-enzyme conjugates in combination with prodrugs for the delivery of cytotoxic agents to tumor cells |
| US5053394A (en) | 1988-09-21 | 1991-10-01 | American Cyanamid Company | Targeted forms of methyltrithio antitumor agents |
| WO1991014438A1 (en) | 1990-03-20 | 1991-10-03 | The Trustees Of Columbia University In The City Of New York | Chimeric antibodies with receptor binding ligands in place of their constant region |
| US5208020A (en) | 1989-10-25 | 1993-05-04 | Immunogen Inc. | Cytotoxic agents comprising maytansinoids and their therapeutic use |
| WO1993021232A1 (en) | 1992-04-10 | 1993-10-28 | Research Development Foundation | IMMUNOTOXINS DIRECTED AGAINST c-erbB-2 (HER-2/neu) RELATED SURFACE ANTIGENS |
| WO1996032478A1 (en) | 1995-04-14 | 1996-10-17 | Genentech, Inc. | Altered polypeptides with increased half-life |
| US5635483A (en) | 1992-12-03 | 1997-06-03 | Arizona Board Of Regents Acting On Behalf Of Arizona State University | Tumor inhibiting tetrapeptide bearing modified phenethyl amides |
| US5663149A (en) | 1994-12-13 | 1997-09-02 | Arizona Board Of Regents Acting On Behalf Of Arizona State University | Human cancer inhibitory pentapeptide heterocyclic and halophenyl amides |
| WO1997043316A1 (en) | 1996-05-10 | 1997-11-20 | Beth Israel Deaconess Medical Center, Inc. | Physiologically active molecules with extended half-lives and methods of using same |
| US5712374A (en) | 1995-06-07 | 1998-01-27 | American Cyanamid Company | Method for the preparation of substantiallly monomeric calicheamicin derivative/carrier conjugates |
| US5714586A (en) | 1995-06-07 | 1998-02-03 | American Cyanamid Company | Methods for the preparation of monomeric calicheamicin derivative/carrier conjugates |
| US5739116A (en) | 1994-06-03 | 1998-04-14 | American Cyanamid Company | Enediyne derivatives useful for the synthesis of conjugates of methyltrithio antitumor agents |
| US5747035A (en) | 1995-04-14 | 1998-05-05 | Genentech, Inc. | Polypeptides with increased half-life for use in treating disorders involving the LFA-1 receptor |
| US5770710A (en) | 1987-10-30 | 1998-06-23 | American Cyanamid Company | Antitumor and antibacterial substituted disulfide derivatives prepared from compounds possessing a methlytrithio group |
| US5770701A (en) | 1987-10-30 | 1998-06-23 | American Cyanamid Company | Process for preparing targeted forms of methyltrithio antitumor agents |
| US5780588A (en) | 1993-01-26 | 1998-07-14 | Arizona Board Of Regents | Elucidation and synthesis of selected pentapeptides |
| US5869046A (en) | 1995-04-14 | 1999-02-09 | Genentech, Inc. | Altered polypeptides with increased half-life |
| WO1999043713A1 (en) | 1998-02-25 | 1999-09-02 | Lexigen Pharmaceuticals Corporation | Enhancing the circulating half-life of antibody-based fusion proteins |
| WO2000029004A1 (en) | 1998-11-18 | 2000-05-25 | Peptor Ltd. | Small functional units of antibody heavy chain variable regions |
| WO2000042072A2 (en) | 1999-01-15 | 2000-07-20 | Genentech, Inc. | Polypeptide variants with altered effector function |
| WO2000061739A1 (en) | 1999-04-09 | 2000-10-19 | Kyowa Hakko Kogyo Co., Ltd. | Method for controlling the activity of immunologically functional molecule |
| WO2002031240A2 (en) | 2000-10-06 | 2002-04-18 | Milliken & Company | Face plate for spun-like textured yarn |
| EP1229125A1 (en) | 1999-10-19 | 2002-08-07 | Kyowa Hakko Kogyo Co., Ltd. | Process for producing polypeptide |
| WO2002060919A2 (en) | 2000-12-12 | 2002-08-08 | Medimmune, Inc. | Molecules with extended half-lives, compositions and uses thereof |
| WO2002088172A2 (en) | 2001-04-30 | 2002-11-07 | Seattle Genetics, Inc. | Pentapeptide compounds and uses related thereto |
| WO2003011878A2 (en) | 2001-08-03 | 2003-02-13 | Glycart Biotechnology Ag | Antibody glycosylation variants having increased antibody-dependent cellular cytotoxicity |
| US6570024B2 (en) | 2000-09-12 | 2003-05-27 | Smithkline Beecham Corporation | Process and intermediates |
| US6573074B2 (en) | 2000-04-12 | 2003-06-03 | Smithkline Beecham Plc | Methods for ansamitocin production |
| EP1391213A1 (en) | 2002-08-21 | 2004-02-25 | Boehringer Ingelheim International GmbH | Compositions and methods for treating cancer using maytansinoid CD44 antibody immunoconjugates and chemotherapeutic agents |
| WO2004029207A2 (en) | 2002-09-27 | 2004-04-08 | Xencor Inc. | Optimized fc variants and methods for their generation |
| US6737056B1 (en) | 1999-01-15 | 2004-05-18 | Genentech, Inc. | Polypeptide variants with altered effector function |
| WO2004063351A2 (en) | 2003-01-09 | 2004-07-29 | Macrogenics, Inc. | IDENTIFICATION AND ENGINEERING OF ANTIBODIES WITH VARIANT Fc REGIONS AND METHODS OF USING SAME |
| US20050053973A1 (en) | 2001-04-26 | 2005-03-10 | Avidia Research Institute | Novel proteins with targeted binding |
| US20050089932A1 (en) | 2001-04-26 | 2005-04-28 | Avidia Research Institute | Novel proteins with targeted binding |
| US20050164301A1 (en) | 2003-10-24 | 2005-07-28 | Avidia Research Institute | LDL receptor class A and EGF domain monomers and multimers |
| US6946292B2 (en) | 2000-10-06 | 2005-09-20 | Kyowa Hakko Kogyo Co., Ltd. | Cells producing antibody compositions with increased antibody dependent cytotoxic activity |
| WO2006014679A1 (en) | 2004-07-21 | 2006-02-09 | Glycofi, Inc. | Immunoglobulins comprising predominantly a glcnac2man3glcnac2 glycoform |
| WO2006130834A2 (en) | 2005-05-31 | 2006-12-07 | Board Of Regents, The University Of Texas System | IGGl ANTIBODIES WITH MUTATED FC PORTION FOR INCREASED BINDING TO FCRN RECEPTOR AND USES THEREOF |
| US7256257B2 (en) | 2001-04-30 | 2007-08-14 | Seattle Genetics, Inc. | Pentapeptide compounds and uses related thereto |
| US7498298B2 (en) | 2003-11-06 | 2009-03-03 | Seattle Genetics, Inc. | Monomethylvaline compounds capable of conjugation to ligands |
| WO2012163805A1 (en) | 2011-05-27 | 2012-12-06 | Glaxo Group Limited | Bcma (cd269/tnfrsf17) -binding proteins |
| WO2013072406A1 (en) | 2011-11-15 | 2013-05-23 | Amgen Research (Munich) Gmbh | Binding molecules for bcma and cd3 |
| WO2013154760A1 (en) | 2012-04-11 | 2013-10-17 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Chimeric antigen receptors targeting b-cell maturation antigen |
| US20130280280A1 (en) * | 2011-05-27 | 2013-10-24 | Glaxosmithkline | Antigen binding proteins |
| WO2014068079A1 (en) | 2012-11-01 | 2014-05-08 | Max-Delbrück-Centrum für Molekulare Medizin | An antibody that binds cd269 (bcma) suitable for use in the treatment of plasma cell diseases such as multiple myeloma and autoimmune diseases |
| WO2014089335A2 (en) | 2012-12-07 | 2014-06-12 | Amgen Inc. | Bcma antigen binding proteins |
| WO2014122143A1 (en) | 2013-02-05 | 2014-08-14 | Engmab Ag | Method for the selection of antibodies against bcma |
| WO2014140248A1 (en) | 2013-03-15 | 2014-09-18 | Amgen Research (Munich) Gmbh | Binding molecules for bcma and cd3 |
| US8961967B2 (en) | 2009-11-30 | 2015-02-24 | Janssen Biotech, Inc. | Antibody Fc mutants with ablated effector functions |
| WO2015052536A1 (en) | 2013-10-10 | 2015-04-16 | Ucl Business Plc | Molecule |
| WO2015158671A1 (en) | 2014-04-14 | 2015-10-22 | Cellectis | Bcma (cd269) specific chimeric antigen receptors for cancer immunotherapy |
| WO2015166649A1 (en) | 2014-04-28 | 2015-11-05 | 株式会社ブリヂストン | Bias tire, and method for manufacturing same |
| WO2016014789A2 (en) | 2014-07-24 | 2016-01-28 | Bluebird Bio, Inc. | Bcma chimeric antigen receptors |
| WO2016014565A2 (en) | 2014-07-21 | 2016-01-28 | Novartis Ag | Treatment of cancer using humanized anti-bcma chimeric antigen receptor |
| WO2016020332A1 (en) | 2014-08-04 | 2016-02-11 | Engmab Ag | Bispecific antibodies against cd3epsilon and bcma |
| WO2016079177A1 (en) | 2014-11-20 | 2016-05-26 | Engmab Ag | Bispecific antibodies against cd3epsilon and bcma |
| WO2016090327A2 (en) | 2014-12-05 | 2016-06-09 | Memorial Sloan-Kettering Cancer Center | Antibodies targeting b-cell maturation antigen and methods of use |
| WO2016090320A1 (en) | 2014-12-05 | 2016-06-09 | Memorial Sloan-Kettering Cancer Center | Chimeric antigen receptors targeting b-cell maturation antigen and uses thereof |
| WO2017021450A1 (en) | 2015-08-03 | 2017-02-09 | Engmab Ag | Monoclonal antibodies against bcma |
| WO2017051068A1 (en) | 2015-09-22 | 2017-03-30 | Nokia Technologies Oy | Photodetector with conductive channel made from two dimensional material and its manufacturing method |
| US20170165373A1 (en) | 2012-12-07 | 2017-06-15 | Amgen Inc. | Bcma antigen binding proteins |
| WO2018201051A1 (en) | 2017-04-28 | 2018-11-01 | Novartis Ag | Bcma-targeting agent, and combination therapy with a gamma secretase inhibitor |
-
2022
- 2022-05-26 WO PCT/GB2022/051348 patent/WO2022248870A1/en not_active Ceased
- 2022-05-26 JP JP2023573231A patent/JP2024521187A/en active Pending
- 2022-05-26 CN CN202280038099.7A patent/CN117396513A/en active Pending
- 2022-05-26 EP EP22740439.9A patent/EP4347656A1/en active Pending
- 2022-05-26 BR BR112023024804A patent/BR112023024804A2/en not_active Application Discontinuation
- 2022-05-26 CA CA3220227A patent/CA3220227A1/en active Pending
- 2022-05-26 US US18/562,868 patent/US20250263495A1/en active Pending
Patent Citations (97)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3896111A (en) | 1973-02-20 | 1975-07-22 | Research Corp | Ansa macrolides |
| US4151042A (en) | 1977-03-31 | 1979-04-24 | Takeda Chemical Industries, Ltd. | Method for producing maytansinol and its derivatives |
| US4137230A (en) | 1977-11-14 | 1979-01-30 | Takeda Chemical Industries, Ltd. | Method for the production of maytansinoids |
| US4265814A (en) | 1978-03-24 | 1981-05-05 | Takeda Chemical Industries | Matansinol 3-n-hexadecanoate |
| US4307016A (en) | 1978-03-24 | 1981-12-22 | Takeda Chemical Industries, Ltd. | Demethyl maytansinoids |
| US4361650A (en) | 1978-03-24 | 1982-11-30 | Takeda Chemical Industries, Ltd. | Fermentation process of preparing demethyl maytansinoids |
| US4248870A (en) | 1978-10-27 | 1981-02-03 | Takeda Chemical Industries, Ltd. | Maytansinoids and use |
| US4256746A (en) | 1978-11-14 | 1981-03-17 | Takeda Chemical Industries | Dechloromaytansinoids, their pharmaceutical compositions and method of use |
| US4260608A (en) | 1978-11-14 | 1981-04-07 | Takeda Chemical Industries, Ltd. | Maytansinoids, pharmaceutical compositions thereof and methods of use thereof |
| US4294757A (en) | 1979-01-31 | 1981-10-13 | Takeda Chemical Industries, Ltd | 20-O-Acylmaytansinoids |
| US4322348A (en) | 1979-06-05 | 1982-03-30 | Takeda Chemical Industries, Ltd. | Maytansinoids |
| US4317821A (en) | 1979-06-08 | 1982-03-02 | Takeda Chemical Industries, Ltd. | Maytansinoids, their use and pharmaceutical compositions thereof |
| US4308269A (en) | 1979-06-11 | 1981-12-29 | Takeda Chemical Industries, Ltd. | Maytansinoids, pharmaceutical compositions thereof and method of use thereof |
| US4308268A (en) | 1979-06-11 | 1981-12-29 | Takeda Chemical Industries, Ltd. | Maytansinoids, pharmaceutical compositions thereof and method of use thereof |
| US4309428A (en) | 1979-07-30 | 1982-01-05 | Takeda Chemical Industries, Ltd. | Maytansinoids |
| US4331598A (en) | 1979-09-19 | 1982-05-25 | Takeda Chemical Industries, Ltd. | Maytansinoids |
| US4362663A (en) | 1979-09-21 | 1982-12-07 | Takeda Chemical Industries, Ltd. | Maytansinoid compound |
| US4364866A (en) | 1979-09-21 | 1982-12-21 | Takeda Chemical Industries, Ltd. | Maytansinoids |
| US4371533A (en) | 1980-10-08 | 1983-02-01 | Takeda Chemical Industries, Ltd. | 4,5-Deoxymaytansinoids, their use and pharmaceutical compositions thereof |
| US4450254A (en) | 1980-11-03 | 1984-05-22 | Standard Oil Company | Impact improvement of high nitrile resins |
| US4313946A (en) | 1981-01-27 | 1982-02-02 | The United States Of America As Represented By The Secretary Of Agriculture | Chemotherapeutically active maytansinoids from Trewia nudiflora |
| US4315929A (en) | 1981-01-27 | 1982-02-16 | The United States Of America As Represented By The Secretary Of Agriculture | Method of controlling the European corn borer with trewiasine |
| US4424219A (en) | 1981-05-20 | 1984-01-03 | Takeda Chemical Industries, Ltd. | 9-Thiomaytansinoids and their pharmaceutical compositions and use |
| US4703039A (en) | 1984-04-10 | 1987-10-27 | New England Deaconess Hospital Corporation | Method of producing biologically active molecules having extended life time |
| EP0307434A1 (en) | 1987-03-18 | 1989-03-22 | Medical Res Council | Altered antibodies. |
| US5770701A (en) | 1987-10-30 | 1998-06-23 | American Cyanamid Company | Process for preparing targeted forms of methyltrithio antitumor agents |
| US5770710A (en) | 1987-10-30 | 1998-06-23 | American Cyanamid Company | Antitumor and antibacterial substituted disulfide derivatives prepared from compounds possessing a methlytrithio group |
| US4975278A (en) | 1988-02-26 | 1990-12-04 | Bristol-Myers Company | Antibody-enzyme conjugates in combination with prodrugs for the delivery of cytotoxic agents to tumor cells |
| US5053394A (en) | 1988-09-21 | 1991-10-01 | American Cyanamid Company | Targeted forms of methyltrithio antitumor agents |
| US5208020A (en) | 1989-10-25 | 1993-05-04 | Immunogen Inc. | Cytotoxic agents comprising maytansinoids and their therapeutic use |
| WO1991014438A1 (en) | 1990-03-20 | 1991-10-03 | The Trustees Of Columbia University In The City Of New York | Chimeric antibodies with receptor binding ligands in place of their constant region |
| WO1993021232A1 (en) | 1992-04-10 | 1993-10-28 | Research Development Foundation | IMMUNOTOXINS DIRECTED AGAINST c-erbB-2 (HER-2/neu) RELATED SURFACE ANTIGENS |
| US5635483A (en) | 1992-12-03 | 1997-06-03 | Arizona Board Of Regents Acting On Behalf Of Arizona State University | Tumor inhibiting tetrapeptide bearing modified phenethyl amides |
| US5780588A (en) | 1993-01-26 | 1998-07-14 | Arizona Board Of Regents | Elucidation and synthesis of selected pentapeptides |
| US5773001A (en) | 1994-06-03 | 1998-06-30 | American Cyanamid Company | Conjugates of methyltrithio antitumor agents and intermediates for their synthesis |
| US5739116A (en) | 1994-06-03 | 1998-04-14 | American Cyanamid Company | Enediyne derivatives useful for the synthesis of conjugates of methyltrithio antitumor agents |
| US5877296A (en) | 1994-06-03 | 1999-03-02 | American Cyanamid Company | Process for preparing conjugates of methyltrithio antitumor agents |
| US5767285A (en) | 1994-06-03 | 1998-06-16 | American Cyanamid Company | Linkers useful for the synthesis of conjugates of methyltrithio antitumor agents |
| US5663149A (en) | 1994-12-13 | 1997-09-02 | Arizona Board Of Regents Acting On Behalf Of Arizona State University | Human cancer inhibitory pentapeptide heterocyclic and halophenyl amides |
| US5747035A (en) | 1995-04-14 | 1998-05-05 | Genentech, Inc. | Polypeptides with increased half-life for use in treating disorders involving the LFA-1 receptor |
| US5869046A (en) | 1995-04-14 | 1999-02-09 | Genentech, Inc. | Altered polypeptides with increased half-life |
| WO1996032478A1 (en) | 1995-04-14 | 1996-10-17 | Genentech, Inc. | Altered polypeptides with increased half-life |
| US5714586A (en) | 1995-06-07 | 1998-02-03 | American Cyanamid Company | Methods for the preparation of monomeric calicheamicin derivative/carrier conjugates |
| US5712374A (en) | 1995-06-07 | 1998-01-27 | American Cyanamid Company | Method for the preparation of substantiallly monomeric calicheamicin derivative/carrier conjugates |
| WO1997043316A1 (en) | 1996-05-10 | 1997-11-20 | Beth Israel Deaconess Medical Center, Inc. | Physiologically active molecules with extended half-lives and methods of using same |
| WO1999043713A1 (en) | 1998-02-25 | 1999-09-02 | Lexigen Pharmaceuticals Corporation | Enhancing the circulating half-life of antibody-based fusion proteins |
| WO2000029004A1 (en) | 1998-11-18 | 2000-05-25 | Peptor Ltd. | Small functional units of antibody heavy chain variable regions |
| WO2000042072A2 (en) | 1999-01-15 | 2000-07-20 | Genentech, Inc. | Polypeptide variants with altered effector function |
| US6737056B1 (en) | 1999-01-15 | 2004-05-18 | Genentech, Inc. | Polypeptide variants with altered effector function |
| WO2000061739A1 (en) | 1999-04-09 | 2000-10-19 | Kyowa Hakko Kogyo Co., Ltd. | Method for controlling the activity of immunologically functional molecule |
| US7214775B2 (en) | 1999-04-09 | 2007-05-08 | Kyowa Hakko Kogyo Co., Ltd. | Method of modulating the activity of functional immune molecules |
| EP1229125A1 (en) | 1999-10-19 | 2002-08-07 | Kyowa Hakko Kogyo Co., Ltd. | Process for producing polypeptide |
| US6573074B2 (en) | 2000-04-12 | 2003-06-03 | Smithkline Beecham Plc | Methods for ansamitocin production |
| US6884874B2 (en) | 2000-09-12 | 2005-04-26 | Smithkline Beecham Corporation | Process and intermediates |
| US6570024B2 (en) | 2000-09-12 | 2003-05-27 | Smithkline Beecham Corporation | Process and intermediates |
| US6946292B2 (en) | 2000-10-06 | 2005-09-20 | Kyowa Hakko Kogyo Co., Ltd. | Cells producing antibody compositions with increased antibody dependent cytotoxic activity |
| WO2002031240A2 (en) | 2000-10-06 | 2002-04-18 | Milliken & Company | Face plate for spun-like textured yarn |
| WO2002060919A2 (en) | 2000-12-12 | 2002-08-08 | Medimmune, Inc. | Molecules with extended half-lives, compositions and uses thereof |
| US20050053973A1 (en) | 2001-04-26 | 2005-03-10 | Avidia Research Institute | Novel proteins with targeted binding |
| US20050089932A1 (en) | 2001-04-26 | 2005-04-28 | Avidia Research Institute | Novel proteins with targeted binding |
| US7256257B2 (en) | 2001-04-30 | 2007-08-14 | Seattle Genetics, Inc. | Pentapeptide compounds and uses related thereto |
| US6884869B2 (en) | 2001-04-30 | 2005-04-26 | Seattle Genetics, Inc. | Pentapeptide compounds and uses related thereto |
| WO2002088172A2 (en) | 2001-04-30 | 2002-11-07 | Seattle Genetics, Inc. | Pentapeptide compounds and uses related thereto |
| US7098308B2 (en) | 2001-04-30 | 2006-08-29 | Seattle Genetics, Inc. | Pentapeptide compounds and uses related thereto |
| US7423116B2 (en) | 2001-04-30 | 2008-09-09 | Seattle Genetics Inc. | Pentapeptide compounds and uses related thereto |
| WO2003011878A2 (en) | 2001-08-03 | 2003-02-13 | Glycart Biotechnology Ag | Antibody glycosylation variants having increased antibody-dependent cellular cytotoxicity |
| EP1391213A1 (en) | 2002-08-21 | 2004-02-25 | Boehringer Ingelheim International GmbH | Compositions and methods for treating cancer using maytansinoid CD44 antibody immunoconjugates and chemotherapeutic agents |
| WO2004029207A2 (en) | 2002-09-27 | 2004-04-08 | Xencor Inc. | Optimized fc variants and methods for their generation |
| WO2004063351A2 (en) | 2003-01-09 | 2004-07-29 | Macrogenics, Inc. | IDENTIFICATION AND ENGINEERING OF ANTIBODIES WITH VARIANT Fc REGIONS AND METHODS OF USING SAME |
| US20050164301A1 (en) | 2003-10-24 | 2005-07-28 | Avidia Research Institute | LDL receptor class A and EGF domain monomers and multimers |
| US7498298B2 (en) | 2003-11-06 | 2009-03-03 | Seattle Genetics, Inc. | Monomethylvaline compounds capable of conjugation to ligands |
| WO2006014679A1 (en) | 2004-07-21 | 2006-02-09 | Glycofi, Inc. | Immunoglobulins comprising predominantly a glcnac2man3glcnac2 glycoform |
| WO2006130834A2 (en) | 2005-05-31 | 2006-12-07 | Board Of Regents, The University Of Texas System | IGGl ANTIBODIES WITH MUTATED FC PORTION FOR INCREASED BINDING TO FCRN RECEPTOR AND USES THEREOF |
| US8961967B2 (en) | 2009-11-30 | 2015-02-24 | Janssen Biotech, Inc. | Antibody Fc mutants with ablated effector functions |
| US20130280280A1 (en) * | 2011-05-27 | 2013-10-24 | Glaxosmithkline | Antigen binding proteins |
| WO2012163805A1 (en) | 2011-05-27 | 2012-12-06 | Glaxo Group Limited | Bcma (cd269/tnfrsf17) -binding proteins |
| WO2013072406A1 (en) | 2011-11-15 | 2013-05-23 | Amgen Research (Munich) Gmbh | Binding molecules for bcma and cd3 |
| WO2013072415A1 (en) | 2011-11-15 | 2013-05-23 | Amgen Research (Munich) Gmbh | Binding molecules for bcma and cd3 |
| WO2013154760A1 (en) | 2012-04-11 | 2013-10-17 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Chimeric antigen receptors targeting b-cell maturation antigen |
| WO2014068079A1 (en) | 2012-11-01 | 2014-05-08 | Max-Delbrück-Centrum für Molekulare Medizin | An antibody that binds cd269 (bcma) suitable for use in the treatment of plasma cell diseases such as multiple myeloma and autoimmune diseases |
| US20170165373A1 (en) | 2012-12-07 | 2017-06-15 | Amgen Inc. | Bcma antigen binding proteins |
| WO2014089335A2 (en) | 2012-12-07 | 2014-06-12 | Amgen Inc. | Bcma antigen binding proteins |
| WO2014122143A1 (en) | 2013-02-05 | 2014-08-14 | Engmab Ag | Method for the selection of antibodies against bcma |
| WO2014122144A1 (en) | 2013-02-05 | 2014-08-14 | Engmab Ag | BISPECIFIC ANTIBODIES AGAINST CD3ε AND BCMA |
| WO2014140248A1 (en) | 2013-03-15 | 2014-09-18 | Amgen Research (Munich) Gmbh | Binding molecules for bcma and cd3 |
| WO2015052536A1 (en) | 2013-10-10 | 2015-04-16 | Ucl Business Plc | Molecule |
| WO2015158671A1 (en) | 2014-04-14 | 2015-10-22 | Cellectis | Bcma (cd269) specific chimeric antigen receptors for cancer immunotherapy |
| WO2015166649A1 (en) | 2014-04-28 | 2015-11-05 | 株式会社ブリヂストン | Bias tire, and method for manufacturing same |
| WO2016014565A2 (en) | 2014-07-21 | 2016-01-28 | Novartis Ag | Treatment of cancer using humanized anti-bcma chimeric antigen receptor |
| WO2016014789A2 (en) | 2014-07-24 | 2016-01-28 | Bluebird Bio, Inc. | Bcma chimeric antigen receptors |
| WO2016020332A1 (en) | 2014-08-04 | 2016-02-11 | Engmab Ag | Bispecific antibodies against cd3epsilon and bcma |
| WO2016079177A1 (en) | 2014-11-20 | 2016-05-26 | Engmab Ag | Bispecific antibodies against cd3epsilon and bcma |
| WO2016090327A2 (en) | 2014-12-05 | 2016-06-09 | Memorial Sloan-Kettering Cancer Center | Antibodies targeting b-cell maturation antigen and methods of use |
| WO2016090320A1 (en) | 2014-12-05 | 2016-06-09 | Memorial Sloan-Kettering Cancer Center | Chimeric antigen receptors targeting b-cell maturation antigen and uses thereof |
| WO2017021450A1 (en) | 2015-08-03 | 2017-02-09 | Engmab Ag | Monoclonal antibodies against bcma |
| WO2017051068A1 (en) | 2015-09-22 | 2017-03-30 | Nokia Technologies Oy | Photodetector with conductive channel made from two dimensional material and its manufacturing method |
| WO2018201051A1 (en) | 2017-04-28 | 2018-11-01 | Novartis Ag | Bcma-targeting agent, and combination therapy with a gamma secretase inhibitor |
Non-Patent Citations (54)
| Title |
|---|
| "GenBank", Database accession no. Q02223.2 |
| "Monoclonal Antibodies in Immunoscintigraphy", 1989, CHATAL, CRC PRESS |
| "Peptide and Protein Drug Delivery", 1991, MARCEL DEKKER, INC., pages: 247 - 301 |
| "Remington's Pharmaceutical Sciences", 2000, MACK PUBLISHING COMPANY |
| BARILÀ GREGORIO ET AL: "Drug Conjugated and Bispecific Antibodies for Multiple Myeloma: Improving Immunotherapies off the Shelf", PHARMACEUTICALS, vol. 14, no. 1, 1 January 2021 (2021-01-01), CH, pages 40, XP055962781, ISSN: 1424-8247, DOI: 10.3390/ph14010040 * |
| BECK A. ET AL., NATURE REV. DRUG DISC., vol. 16, 2017, pages 315 - 337 |
| BECNEL MELODY R. ET AL: "The role of belantamab mafodotin for patients with relapsed and/or refractory multiple myeloma", THERAPEUTIC ADVANCES IN HEMATOLOGY, vol. 11, 14 January 2020 (2020-01-14), GB, XP055962980, ISSN: 2040-6207, Retrieved from the Internet <URL:http://journals.sagepub.com/doi/full-xml/10.1177/2040620720979813> DOI: 10.1177/2040620720979813 * |
| CAS , no. 7491-09-0 |
| CHAPPEL ET AL., THE JOURNAL OF BIOLOGICAL CHEMISTRY, vol. 268, 1993, pages 25124 - 25131 |
| COHEN ADAM D. ET AL: "Serial treatment of relapsed/refractory multiple myeloma with different BCMA-targeting therapies", BLOOD ADVANCES, vol. 3, no. 16, 27 August 2019 (2019-08-27), pages 2487 - 2490, XP055962481, ISSN: 2473-9529, Retrieved from the Internet <URL:https://ashpublications.org/bloodadvances/article-pdf/3/16/2487/832111/advancesadv2019000466.pdf> DOI: 10.1182/bloodadvances.2019000466 * |
| DALL'ACQUA ET AL., J IMMUNOL., vol. 169, 2002, pages 5171 - 80 |
| DIEBOLDER ET AL., SCIENCE, vol. 343, 2014, pages 1260 - 1293 |
| DORONINA, NAT BIOTECHNOL, vol. 21, no. 7, 2003, pages 778 - 784 |
| E. SCHRODERK. LUBKE: "The Peptides", vol. 1, 1965, ACADEMIC PRESS, pages: 76 - 136 |
| FERNANDEZ DE LARREA CARLOS ET AL: "Defining an Optimal Dual-Targeted CAR T-cell Therapy Approach Simultaneously Targeting BCMA and GPRC5D to Prevent BCMA Escape Driven Relapse in Multiple Myeloma", BLOOD CANCER DISCOVERY, vol. 1, no. 2, 1 September 2020 (2020-09-01), pages 146 - 154, XP055847845, ISSN: 2643-3230, Retrieved from the Internet <URL:https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7575057/pdf/nihms-1627155.pdf> DOI: 10.1158/2643-3230.BCD-20-0020 * |
| FRAKER ET AL., BIOCHEM. BIOPHYS. RES. COMMUN., vol. 80, 1978, pages 49 - 57 |
| GREVYS ET AL., J IMMUNOL., vol. 194, no. 11, 1 June 2015 (2015-06-01), pages 5497 - 5508 |
| HINMAN ET AL., CANCER RES., vol. 53, 1993, pages 3336 - 3342 |
| HINMAN ET AL., CANCER RESEARCH, vol. 53, 1993, pages 3336 - 3342 |
| HOLLIGERHUDSON, NATURE BIOTECHNOLOGY, vol. 23, no. 9, 2005, pages 1137 - 1146 |
| J IMM METH, vol. 184, 1995, pages 29 - 38 |
| JONES, A., ADV. DRUG DELIVERY REV., vol. 10, 1993, pages 29 - 90 |
| KABAT ET AL., SEQUENCES OF PROTEINS OF IMMUNOLOGICAL INTEREST, 1987 |
| KIM ET AL., EUR. J. OF IMMUNO., vol. 24, 1994, pages 542 - 548 |
| KODANDARAM PILLARISETTI ET AL: "A T-cell redirecting bispecific G-protein coupled receptor class 5 member D x CD3 antibody to treat multiple myeloma", BLOOD, vol. 135, no. 15, 9 April 2020 (2020-04-09), US, pages 1232 - 1243, XP055743043, ISSN: 0006-4971, DOI: 10.1182/blood.2019003342 * |
| LAMBERT, J., CURR. OPINION IN PHARMACOLOGY, vol. 5, 2005, pages 543 - 549 |
| LAZAR ET AL., PNAS, vol. 103, 2006, pages 4005 - 4010 |
| LIU ET AL., PROC. NATL. ACAD. SCI. USA, vol. 93, 1996, pages 8618 - 8623 |
| LODE ET AL., CANCER RES., vol. 58, 1998, pages 2928 |
| LODE ET AL., CANCER RESEARCH, vol. 58, 1998, pages 2925 - 2928 |
| MANDLER ET AL., BIOCONJUGATE CHEM., vol. 13, 2002, pages 786 - 791 |
| MANDLER ET AL., BIOORGANIC & MED. CHEM. LETTERS, vol. 10, 2000, pages 1025 - 1028 |
| MANDLER ET AL., J. NAT. CANCER INST., vol. 92, no. 19, 2000, pages 1573 - 1581 |
| MIKE SCHUTKOWSKIULRICH REINEKE: "Epitope Mapping Protocols", METHODS IN MOLECULAR BIOLOGY, vol. 524, 2009 |
| MONNET ET AL., MABS, vol. 6, no. 2, 2014, pages 422 - 436 |
| NICULESCU-DUVAZSPRINGER, ADV. DRUG DELIV. REV., vol. 26, 1997, pages 151 - 172 |
| NOOKA AJAY K ET AL: "Belantamab mafodotin in combination with novel agents in relapsed/refractory multiple myeloma: DREAMM-5 study design", FUTURE ONCOLOGY, vol. 17, no. 16, 8 March 2021 (2021-03-08), GB, pages 1987 - 2003, XP055962494, ISSN: 1479-6694, DOI: 10.2217/fon-2020-1269 * |
| PETTIT ET AL., ANTI-CANCER DRUG DESIGN, vol. 13, 1998, pages 243 - 277 |
| PETTIT ET AL., ANTIMICROB. AGENTS CHEMOTHER., vol. 42, 1998, pages 2961 - 2965 |
| PETTIT ET AL., J. AM. CHEM. SOC., vol. 111, 1989, pages 5463 - 5465 |
| PETTIT ET AL., J. CHEM. SOC. PERKIN TRANS., vol. 15, 1996, pages 859 - 863 |
| PETTIT, G. R. ET AL., SYNTHESIS, 1996, pages 719 - 725 |
| RICHARDS J ET AL., MOL. CANCER THER., vol. 7, 2008, pages 2517 - 2527 |
| ROWLAND ET AL., CANCER IMMUNOL. IMMUNOTHER., vol. 21, 1986, pages 183 - 87 |
| SHIELDS ET AL., J BIOL CHEM, vol. 276, 2001, pages 6591 - 604 |
| SHIELDS ET AL., J BIOL CHEM., vol. 277, no. 30, 2002, pages 26733 - 40 |
| SHIELDS ET AL., J. BIOL. CHEM, vol. 276, 2001, pages 6591 - 6604 |
| SHIELDS ET AL., THE JOURNAL OF BIOLOGICAL CHEMISTRY, vol. 276, 2001, pages 6591 - 6604 |
| SYRIGOSEPENETOS, ANTICANCER RESEARCH, vol. 19, 1999, pages 605 - 614 |
| TAI YU-TZU ET AL: "Novel anti-B-cell maturation antigen antibody-drug conjugate (GSK2857916) selectively induces killing of multiple myeloma", BLOOD, AMERICAN SOCIETY OF HEMATOLOGY, US, vol. 123, no. 20, 15 May 2014 (2014-05-15), pages 3128 - 3138, XP009505860, ISSN: 0006-4971, DOI: 10.1182/BLOOD-2013-10-535088 * |
| TAM ET AL., ANTIBODIES, vol. 6, no. 3, 2017 |
| TSUCHIKAMAAN, PROTEIN AND CELL, vol. 9, 2018, pages 33 - 46 |
| WANG ET AL., PROTEIN CELL, vol. 9, no. 1, January 2018 (2018-01-01), pages 63 - 73 |
| WOYKE ET AL., ANTIMICROB. AGENTS AND CHEMOTHER., vol. 45, no. 12, 2001, pages 3580 - 3584 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024259279A3 (en) * | 2023-06-14 | 2025-05-15 | Interius Biotherapeutics, Inc. | Pseudotyped viral particles comprising engineered chimeric receptor proteins |
Also Published As
| Publication number | Publication date |
|---|---|
| CN117396513A (en) | 2024-01-12 |
| BR112023024804A2 (en) | 2024-02-15 |
| JP2024521187A (en) | 2024-05-28 |
| CA3220227A1 (en) | 2022-12-01 |
| US20250263495A1 (en) | 2025-08-21 |
| EP4347656A1 (en) | 2024-04-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR102514317B1 (en) | Novel B7-H3-binding molecules, antibody drug conjugates thereof and methods of use thereof | |
| CN106471009B (en) | Bispecific antibodies that bind to CD38 and CD3 | |
| EP2934585B1 (en) | Anti-ntb-a antibodies and related compositions and methods | |
| CA3219684A1 (en) | Anti-bcma antibodies, anti-cd3 antibodies and bi-specific antibodies binding to bcma and cd3 | |
| CN107250157A (en) | Antibodies Comprising Modified Heavy Chain Constant Regions | |
| CA2862319A1 (en) | Antibodies to integrin .alpha.v.beta.6 and use of same to treat cancer | |
| AU2016287647A1 (en) | Anti-NTB-A antibodies and related compositions and methods | |
| TW201902512A (en) | treatment method | |
| EP3880707A1 (en) | HUMANIZED ANTI-SIRPa ANTIBODIES | |
| CA3158579A1 (en) | Heavy chain antibodies binding to cd38 | |
| JP2024056791A (en) | Biopharmaceutical Compositions and Related Methods | |
| EP4347656A1 (en) | Combination therapies for treating cancer | |
| WO2023057893A1 (en) | Combination therapies for treating cancer | |
| RU2824761C2 (en) | Biopharmaceutical compositions and methods related thereto | |
| JP2025508606A (en) | Antibodies to human B7-H3 and variants thereof | |
| EA042568B1 (en) | NEW B7-H3 BINDING MOLECULES CONTAINING THEIR ANTIBODY-DRUG CONJUGATES AND METHODS FOR THEIR APPLICATION |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22740439 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 3220227 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202280038099.7 Country of ref document: CN Ref document number: 2023573231 Country of ref document: JP |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112023024804 Country of ref document: BR |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2022740439 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2022740439 Country of ref document: EP Effective date: 20240102 |
|
| ENP | Entry into the national phase |
Ref document number: 112023024804 Country of ref document: BR Kind code of ref document: A2 Effective date: 20231127 |
|
| WWP | Wipo information: published in national office |
Ref document number: 18562868 Country of ref document: US |