WO2022232277A1 - COMPOSITIONS AND METHODS FOR TCR REPROGRAMMING USING FUSION PROTEINS AND TGFβR SWITCH - Google Patents
COMPOSITIONS AND METHODS FOR TCR REPROGRAMMING USING FUSION PROTEINS AND TGFβR SWITCH Download PDFInfo
- Publication number
- WO2022232277A1 WO2022232277A1 PCT/US2022/026542 US2022026542W WO2022232277A1 WO 2022232277 A1 WO2022232277 A1 WO 2022232277A1 US 2022026542 W US2022026542 W US 2022026542W WO 2022232277 A1 WO2022232277 A1 WO 2022232277A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- domain
- nucleic acid
- sequence
- tcr
- cell
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K40/00—Cellular immunotherapy
- A61K40/10—Cellular immunotherapy characterised by the cell type used
- A61K40/11—T-cells, e.g. tumour infiltrating lymphocytes [TIL] or regulatory T [Treg] cells; Lymphokine-activated killer [LAK] cells
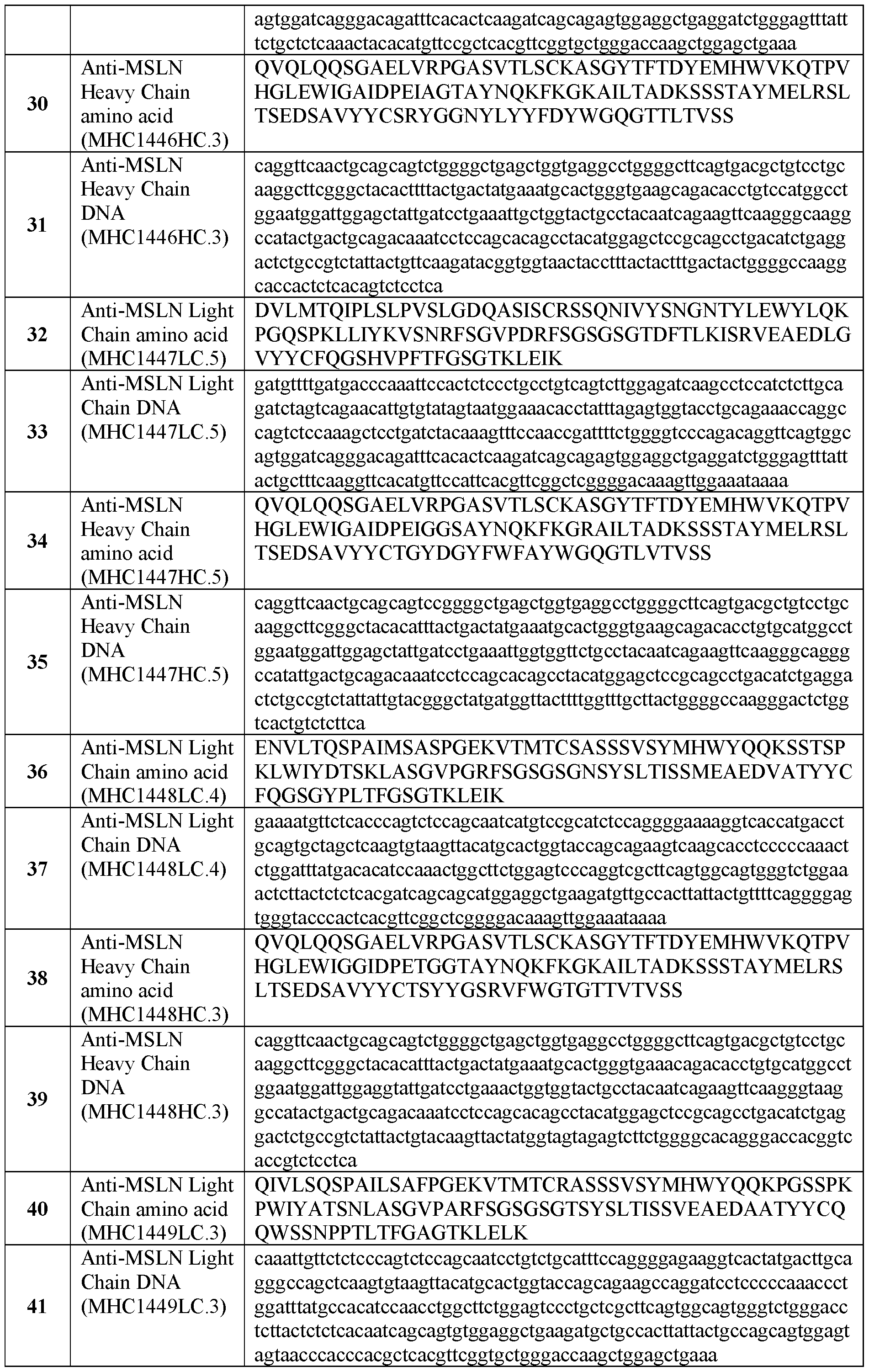
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K40/00—Cellular immunotherapy
- A61K40/30—Cellular immunotherapy characterised by the recombinant expression of specific molecules in the cells of the immune system
- A61K40/32—T-cell receptors [TCR]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K40/00—Cellular immunotherapy
- A61K40/40—Cellular immunotherapy characterised by antigens that are targeted or presented by cells of the immune system
- A61K40/41—Vertebrate antigens
- A61K40/42—Cancer antigens
- A61K40/4225—Growth factors
- A61K40/4229—Transforming growth factor [TGF]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K40/00—Cellular immunotherapy
- A61K40/40—Cellular immunotherapy characterised by antigens that are targeted or presented by cells of the immune system
- A61K40/41—Vertebrate antigens
- A61K40/42—Cancer antigens
- A61K40/4254—Adhesion molecules, e.g. NRCAM, EpCAM or cadherins
- A61K40/4255—Mesothelin [MSLN]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
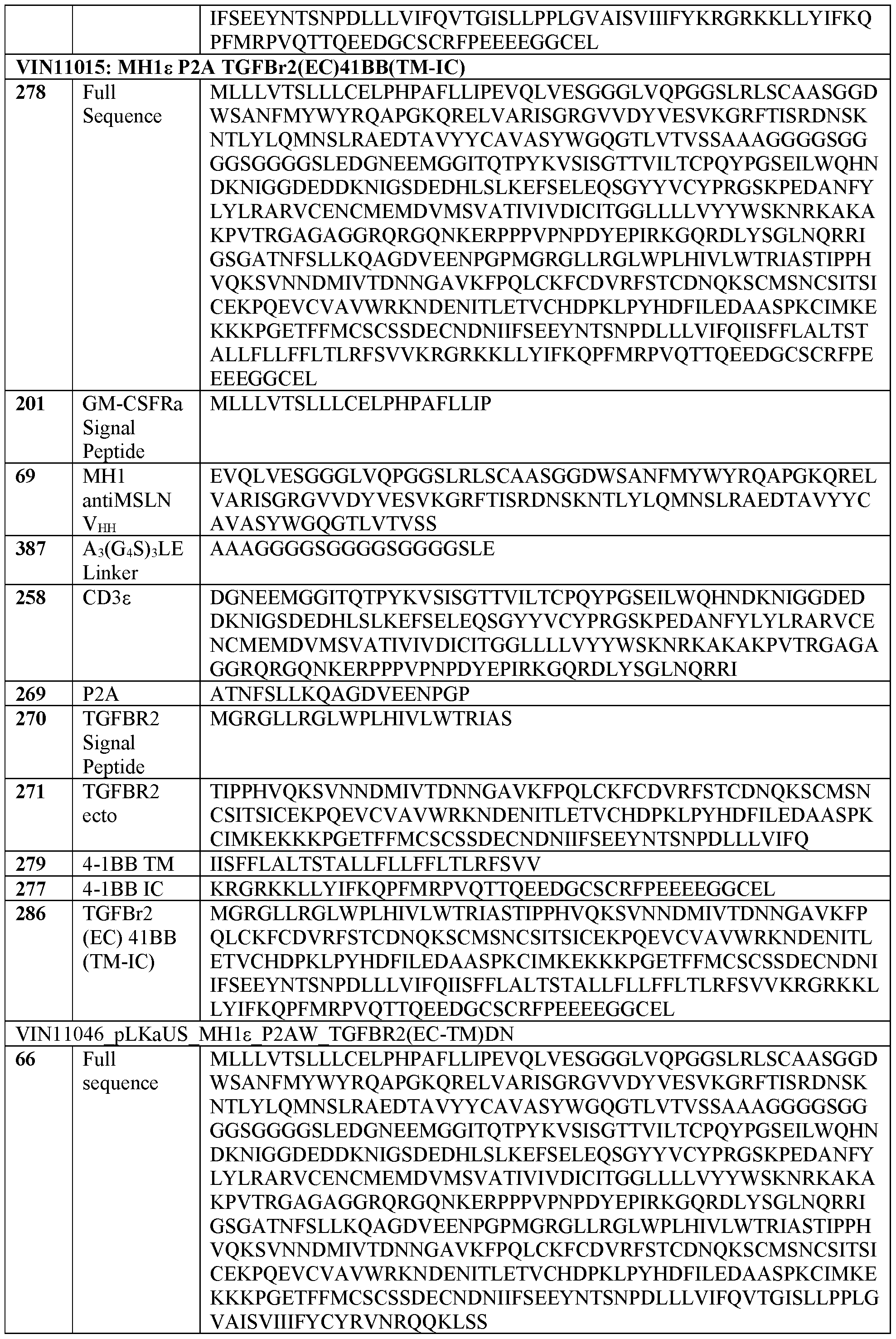
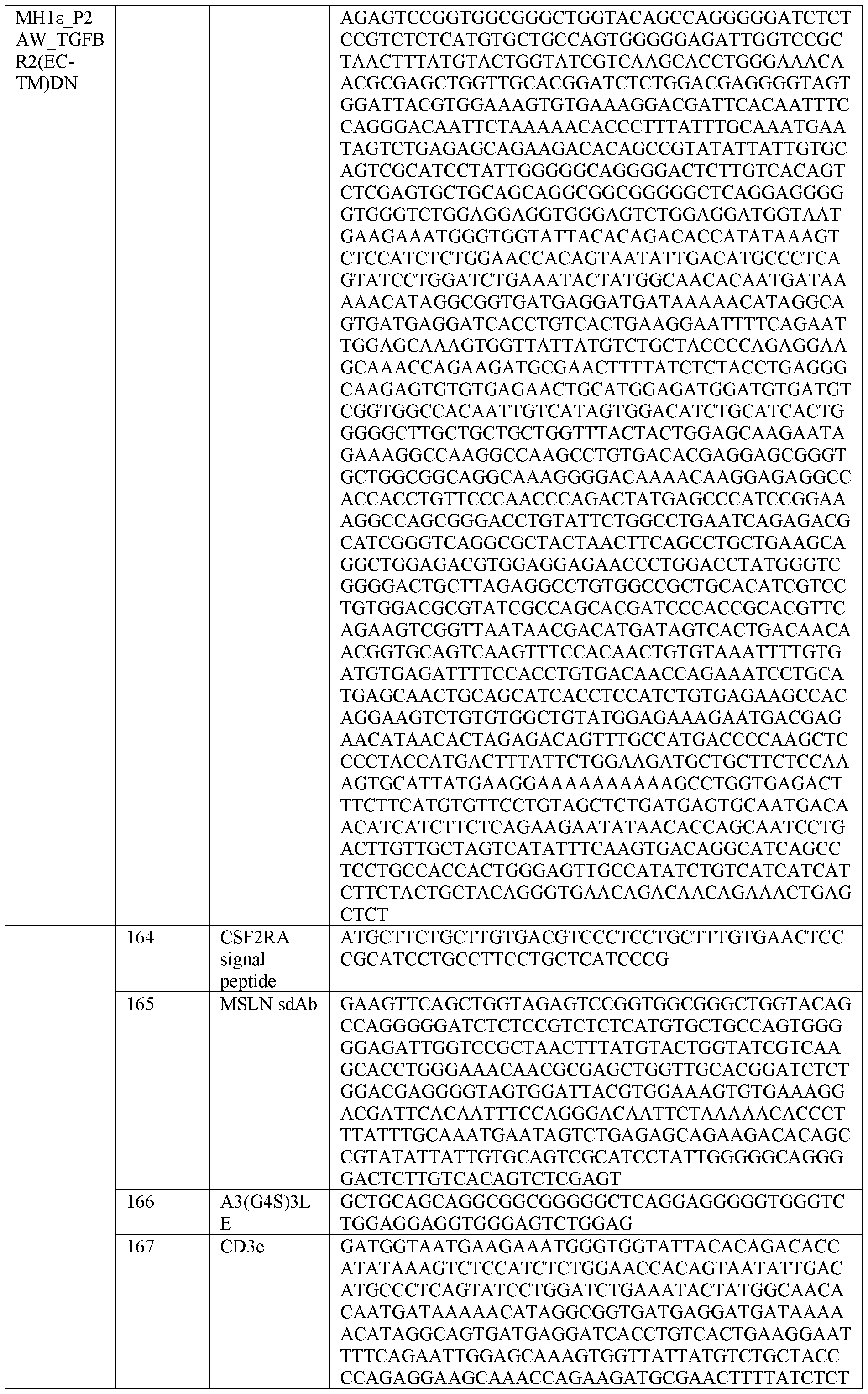
- A61P37/04—Immunostimulants
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
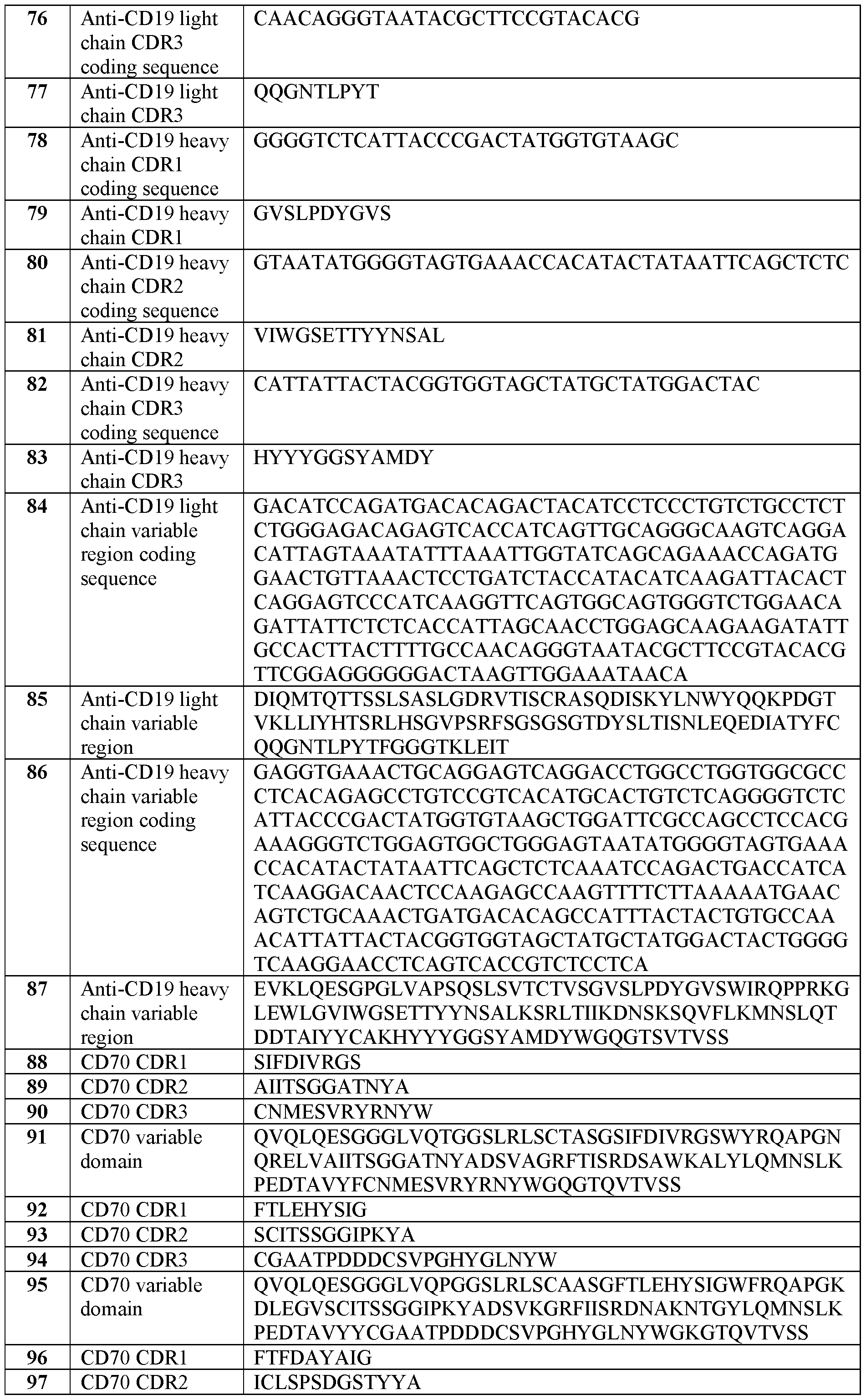
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
- C07K14/70503—Immunoglobulin superfamily
- C07K14/7051—T-cell receptor (TcR)-CD3 complex
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
- C07K14/71—Receptors; Cell surface antigens; Cell surface determinants for growth factors; for growth regulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2239/00—Indexing codes associated with cellular immunotherapy of group A61K40/00
- A61K2239/46—Indexing codes associated with cellular immunotherapy of group A61K40/00 characterised by the cancer treated
- A61K2239/54—Pancreas
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/50—Fusion polypeptide containing protease site
Definitions
- cancer cells use multiple mechanisms to render themselves invisible or hostile to the initiation and propagation of an immune attack by cancer immunotherapies.
- CAR chimeric antigen receptor
- CTL019 The clinical results with CD19-specific CAR T cells (called CTL019) have shown complete remissions in patients suffering from chronic lymphocytic leukemia (CLL) as well as in childhood acute lymphoblastic leukemia (ALL) (see, e.g., Kalos et al., Sci Transl Med 3:95ra73 (2011), Porter et al., NEJM 365:725-733 (2011), Grupp et al., NEJM 368:1509-1518 (2013)).
- An alternative approach is the use of T cell receptor (TCR) alpha and beta chains selected for a tumor-associated peptide antigen for genetically engineering autologous T cells.
- TCR T cell receptor
- TCR chains will form complete TCR complexes and provide the T cells with a TCR for a second defined specificity. Encouraging results were obtained with engineered autologous T cells expressing NY-ESO-1-specific TCR alpha and beta chains in patients with synovial carcinoma. [0004] Besides the ability for genetically modified T cells expressing a CAR or a second TCR to recognize and destroy respective target cells in vitro/ex vivo, successful patient therapy with engineered T cells may require the T cells to be capable of strong activation, expansion, persistence over time, effective tumor targeting, and, in case of relapsing disease, enabling a ‘memory’ response.
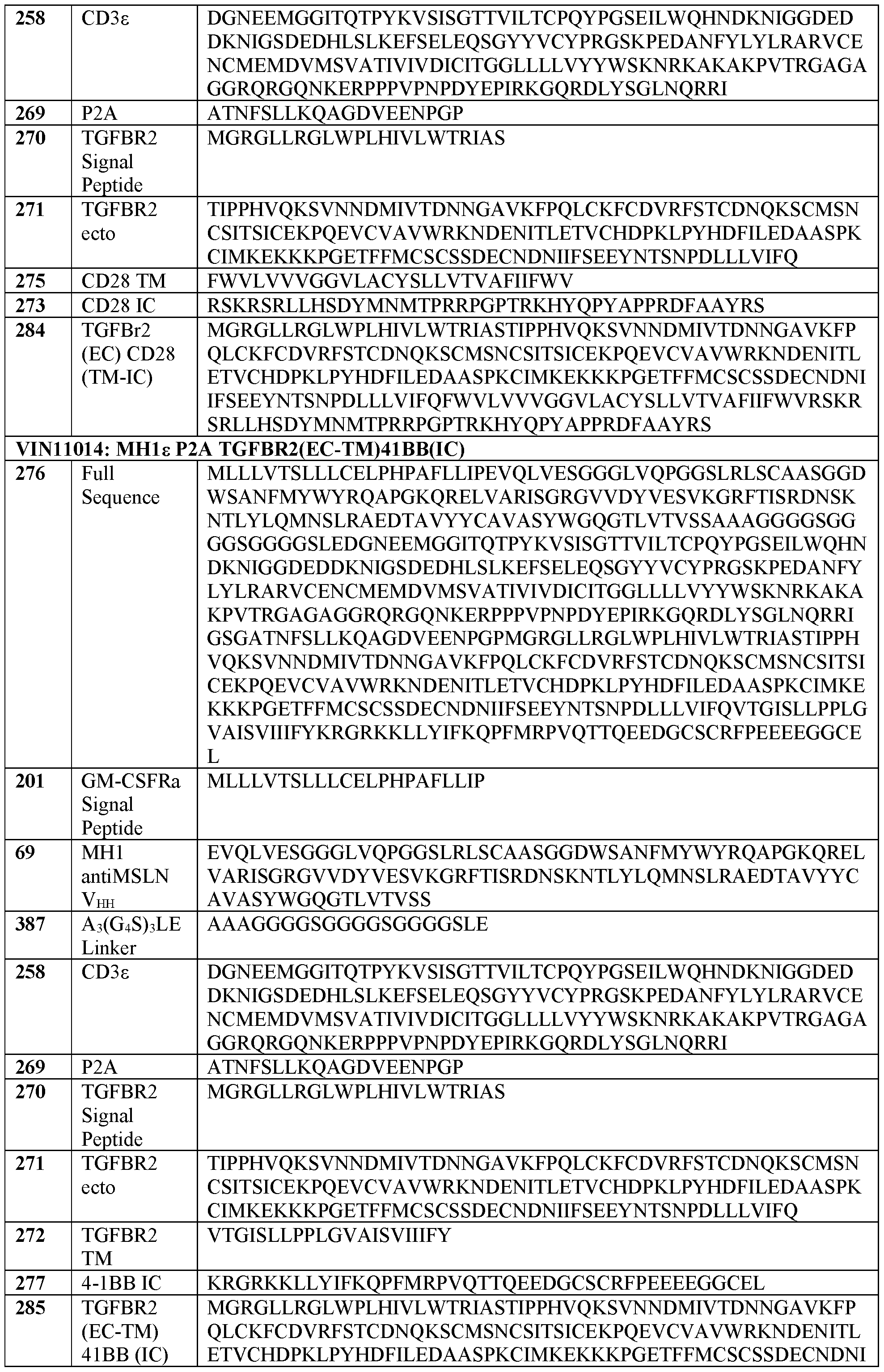
- recombinant nucleic acids comprising: a first nucleic acid sequence encoding a T cell receptor (TCR) fusion protein (TFP), wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain; and wherein the TCR subunit and the antigen binding domain are operatively linked, and a second nucleic acid sequence encoding a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof.
- TCR T cell receptor
- TFP T cell receptor fusion protein
- the second nucleic acid encodes a dominant negative TGFBr2 comprising a TGFBr2 extracellular domain or a functional fragment thereof. In some embodiments, the second nucleic acid encodes a switch polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof.
- the TCR subunit further comprises a TCR intracellular domain.
- the first nucleic acid sequence and the second nucleic acid sequence are operatively linked by a linker.
- the linker comprises a protease cleavage site.
- the protease cleavage site is a 2A cleavage site.
- the 2A cleavage site is a T2A cleavage site or a P2A cleavage site.
- the first nucleic acid sequence and the second nucleic acid sequence are present on different nucleic acid molecules.
- the TGFBr2 extracellular domain comprises a sequence with at least 80% sequence identity to SEQ ID NO:271. In some embodiments, the TGFBr2 extracellular domain comprises a sequence according to SEQ ID NO: 271.
- the dominant negative TGFBr2 comprises a TGFBr2 extracellular domain and a TGFBr2 transmembrane domain and lacks a functional kinase domain.
- the dominant negative TGFBr2 comprises a truncated TGFBr2, for example, a TGFBr2 with a truncated intracellular domain.
- the dominant negative TGFBr2 comprises a TGFBr2 extracellular domain, a TGFBr2 transmembrane domain, and a truncated portion of a TGFBr2 intracellular domain.
- the dominant negative TGFBr2 comprises a sequence with at least 80% sequence identity to SEQ ID NO: 68. In some embodiments, the dominant negative TGFBr2 comprises a sequence according to SEQ ID NO: 68.
- the switch polypeptide further comprises a switch intracellular domain. [0016] In some embodiments, the TGFBr2 extracellular domain is operably linked to the switch intracellular domain. [0017] In some embodiments, the switch intracellular domain comprises an intracellular domain of a costimulatory polypeptide.
- the costimulatory polypeptide is selected from the group consisting of CD28, 4-1BB, IL-15Ra, IL12R, IL18R, IL21R, OX40, CD2, CD27, CD5, ICAM-1, ICOS (CD278), GITR, CD30, CD40, BAFFR, HVEM, CD7, LIGHT, NKG2C, SLAMF7, NKp80, CD160, CD226, Fc ⁇ RI, Fc ⁇ RII, and Fc ⁇ RIII.
- the costimulatory polypeptide is CD28.
- the costimulatory polypeptide is 4-1BB.
- the costimulatory polypeptide is IL-15Ra.
- the switch intracellular domain comprises a sequence with at least 80% sequence identity to SEQ ID NO:273 or SEQ ID NO:277.
- the switch polypeptide further comprises a switch transmembrane domain.
- the TGFBr2 extracellular domain is operably linked to the switch intracellular domain via the switch transmembrane domain.
- the switch transmembrane domain is derived from a TGFBr2 transmembrane domain.
- the switch transmembrane domain comprises a sequence with at least 80% sequence identity to SEQ ID NO:272. [0027] In some embodiments, the switch transmembrane domain is derived from a transmembrane domain of the costimulatory polypeptide. [0028] In some embodiments, the switch transmembrane domain is derived from a transmembrane domain of CD28. [0029] In some embodiments, the switch transmembrane domain is derived from a transmembrane domain of 4-1BB. [0030] In some embodiments, the switch transmembrane domain is derived from a transmembrane domain of IL-15Ra.
- the switch transmembrane domain comprises a sequence with at least 80% sequence identity to SEQ ID NO:275 or SEQ ID NO:279.
- the switch polypeptide further comprises an additional intracellular domain.
- the additional intracellular domain is operably linked to C- terminus of the switch intracellular domain.
- the additional intracellular domain is derived from an intracellular domain of IL-15Ra.
- the additional intracellular domain comprises a sequence with at least 80% sequence identity to SEQ ID NO:372 or SEQ ID NO:383.
- the switch polypeptide comprises a transmembrane domain derived from a TGFBr2 transmembrane domain and an intracellular signaling domain of 4- 1BB. [0037] In some embodiments, the switch polypeptide comprises a transmembrane domain derived from a 4-1BB transmembrane domain and an intracellular signaling domain of 4- 1BB. [0038] In some embodiments, the switch polypeptide comprises a transmembrane domain derived from a TGFBr2 transmembrane domain and an intracellular signaling domain of CD28.
- the switch polypeptide comprises a transmembrane domain derived from a CD28 transmembrane domain and an intracellular signaling domain of CD28.
- the switch polypeptide comprises a sequence with at least 80% sequence identity to a sequence selected from SEQ ID NOs: 283, 284, 285, and 286.
- the TFP functionally interacts with an endogenous TCR complex when expressed in a T cell.
- the TCR intracellular domain comprises a stimulatory domain from an intracellular signaling domain of CD3 gamma, CD3 delta, or CD3 epsilon.
- the TCR intracellular domain comprises an intracellular domain from TCR alpha, TCR beta, TCR gamma, or TCR delta.
- the antigen binding domain is connected to the TCR extracellular domain by a linker sequence.
- the linker sequence is 120 amino acids in length or less.
- the linker sequence comprises (G 4 S) n , wherein G is glycine, S is serine, and n is an integer from 1 to 10 (SEQ ID NO: 390). [0047] In some embodiments, n is an integer from 1 to 4.
- At least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from the same TCR subunit. [0049] In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from TCR alpha. [0050] In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from TCR beta. [0051] In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from TCR gamma.
- At least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from TCR delta. [0053] In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 epsilon. [0054] In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 delta. [0055] In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 gamma.
- all three of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from the same TCR subunit. [0057] In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 epsilon. [0058] In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 delta. [0059] In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 gamma.
- the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain comprise the constant domain of TCR alpha.
- the constant domain of TCR alpha is murine.
- the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain comprise the constant domain of TCR beta.
- the constant domain of TCR beta is murine.
- the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain comprise the constant domain of TCR gamma.
- the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain comprise the constant domain of TCR delta.
- the antigen binding domain is a camelid antibody or binding fragment thereof.
- the antigen binding domain is a murine antibody or binding fragment thereof.
- the antigen binding domain is a human or humanized antibody or binding fragment thereof.
- the antigen binding domain is a single-chain variable fragment (scFv) or a single domain antibody (sdAb) domain.
- the antigen binding domain is a single domain antibody (sdAb).
- the sdAb is a VH or VHH.
- the antigen binding domain is selected from the group consisting of an anti-CD19 binding domain, an anti-B-cell maturation antigen (BCMA) binding domain, and an anti-mesothelin (MSLN) binding domain, an anti-CD20 binding domain, an anti-CD70 binding domain, anti-MUC16 binding domain, an anti-Nectin-4 binding domain, an anti-GPC3 binding domain, and an anti-TROP-2 binding domain.
- a T cell expressing the TFP inhibits tumor growth when expressed in a T cell.
- the recombinant nucleic acid as provided herein further comprises a leader sequence.
- the leader sequence comprises SEQ ID NO: 270.
- the recombinant nucleic acid is selected from the group consisting of a DNA and an RNA.
- the recombinant nucleic acid is an mRNA.
- the recombinant nucleic acid is a circRNA.
- the recombinant nucleic acid comprises a nucleotide analog.
- the nucleotide analog is selected from the group consisting of 2’-O-methyl, 2’-O-methoxyethyl (2’-O-MOE), 2’-O-aminopropyl, 2’-deoxy, T-deoxy-2’- fluoro, 2’-O-aminopropyl (2’-O-AP), 2'-O-dimethylaminoethyl (2’-O-DMAOE), 2’-O- dimethylaminopropyl (2’-O-DMAP), T-O-dimethylaminoethyloxyethyl (2’-O-DMAEOE), 2’-O-N-methylacetamido (2’-O-NMA) modified, a locked nucleic acid (LNA), an ethylene nucleic acid (ENA), a peptide nucleic acid (PNA), a 1’,5’- anhydrohexitol nucleic acid (HNA), a
- LNA locked nucleic
- the recombinant nucleic acid as provided herein further comprises a promoter.
- the recombinant nucleic acid is an in vitro transcribed nucleic acid.
- the recombinant nucleic acid further comprises a sequence encoding a poly(A) tail.
- the recombinant nucleic acid further comprises a 3’UTR sequence.
- the present disclosure provides a polypeptide encoded by the recombinant nucleic acid as provided herein.
- the present disclosure provides a vector comprising a recombinant nucleic acid as provided herein.
- the vector is a lentiviral vector.
- the present disclosure provides a cell comprising the recombinant nucleic acid as provided herein, the polypeptide as provided herein, or the vector as provided herein.
- the cell is a T cell.
- the T cell is a human T cell.
- the T cell is a CD8+ or CD4+ T cell.
- the T cell is a human ⁇ T cell.
- the T cell is a human ⁇ T cell.
- the cell is a human NKT cell.
- the cell is an allogeneic cell or an autologous cell.
- the present disclosure provides a modified T cell or a population of modified T cells comprising a dominant negative TGFBr2 receptor and a T cell receptor (TCR) fusion protein (TFP), wherein the TFP comprises (a) a TCR subunit comprising (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain; and (b) an antigen binding domain, wherein the TCR subunit and the antigen binding domain are operatively linked.
- TCR T cell receptor
- the modified T cell or population of modified T cells comprises a dominant negative TGFBr2 comprising a sequence with at least 80% sequence identity to SEQ ID NO: 68. In some embodiments, the modified T cell or population of modified T cells comprises a dominant negative TGFBr2 comprising a sequence according to SEQ ID NO: 68.
- the present disclosure provides a modified T cell or a population of modified T cells comprising a TGFBr2 switch receptor and a T cell receptor (TCR) fusion protein (TFP), wherein the TFP comprises (a) a TCR subunit comprising (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain; and (b) an antigen binding domain, wherein the TCR subunit and the antigen binding domain are operatively linked.
- the modified T cell or population of modified T cells comprises a switch receptor comprising an amino acid sequence with at least 80% sequence identity to any one of SEQ ID NOs: 283, 284, 285, or 286.
- the modified T cell or population of modified T cells comprises a switch receptor comprising an amino acid sequence selected from the group consisting of SEQ ID Nos: 283, 284, 285, and 286.
- anti-tumor efficacy of the cell is increased compared to a cell that does not comprise the second nucleic acid sequence encoding a polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a portion thereof.
- TGFBr2 transforming growth factor beta receptor II
- expression of an exhaustion marker of the cell is decreased compared to a cell that comprises the first nucleic acid sequence and does not comprise the second nucleic acid sequence.
- cytotoxicity of the cell is increased compared to a cell that comprises the first nucleic acid sequence and does not comprise the second nucleic acid sequence .
- the cytokine production of the cell is increased compared to a cell that comprises the first nucleic acid sequence and does not comprise the second nucleic acid sequence.
- the cytokine is selected from the group consisting of GM-CSF, IFNgamma, IL-2, and TNFalpha.
- pSMAD2/3 signaling is decreased compared to a cell that comprises the first nucleic acid sequence and does not comprise the second nucleic acid sequence.
- the cell comprises a population of cells.
- the present disclosure provides a pharmaceutical composition comprising the cell as provided herein and a pharmaceutically acceptable carrier.
- the present disclosure provides a method of increasing an activity or cytotoxicity of a cell expressing a recombinant nucleic acid comprising a sequence encoding a T cell receptor (TCR) fusion protein (TFP), the method comprising expressing a dominant negative TGFBr2 in the cell, wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain; and wherein the TCR subunit and the antigen binding domain are operatively linked.
- TCR T cell receptor
- TFP T cell receptor fusion protein
- the present disclosure provides a method of increasing an activity or cytotoxicity of a cell expressing a recombinant nucleic acid comprising a sequence encoding a T cell receptor (TCR) fusion protein (TFP), the method comprising expressing a switch polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof in the cell: wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain; and wherein the TCR subunit and the antigen binding domain are operatively linked.
- TCR T cell receptor
- TFP T cell receptor fusion protein
- the cell is the cell as provided herein.
- the present disclosure provides a method of treating a disease or a condition in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of the pharmaceutical composition as provided herein.
- the disease or the condition is a cancer or a disease or a condition associated with expression of CD19, B-cell maturation antigen (BCMA), mesothelin (MSLN), CD20, CD70, MUC16, Trop-2, Nectin-4, or GPC3.
- the cancer is a hematologic cancer selected from the group consisting of B-cell acute lymphoid leukemia (B-ALL), T cell acute lymphoid leukemia (T- ALL), acute lymphoblastic leukemia (ALL), chronic myelogenous leukemia (CML), chronic lymphocytic leukemia (CLL), B cell prolymphocytic leukemia, blastic plasmacytoid dendritic cell neoplasm, Burkitt’s lymphoma, diffuse large B cell lymphoma, follicular lymphoma, hairy cell leukemia, small cell-follicular lymphoma, large cell-follicular lymphoma, malignant lymphoproliferative conditions, MALT lymphoma, mantle cell lymphoma, Marginal zone lymphoma, multiple myeloma, myelodysplasia, myelodysplastic syndrome, non-Hodgkin’s lymphoma, plasmablastic
- the cancer is mesothelioma, renal cell carcinoma, stomach cancer, breast cancer, lung cancer, ovarian cancer, prostate cancer, colon cancer, cervical cancer, brain cancer, liver cancer, pancreatic cancer, thyroid cancer, bladder cancer, ureter cancer, kidney cancer, endometrial cancer, esophageal cancer, gastric cancer, thymic carcinoma or cholangiocarcinoma.
- the subject is a human. INCORPORATION BY REFERENCE
- FIG.1 is a series of graphs showing expansion of T cells expressing the TFP constructs shown over 10 days.
- FIG.2 is a series of flow plots showing VHH (anti-MSLN TFP) expression (transduction efficiency) in T cells expressing the TFP constructs shown in a representative donor. Also shown are graph with average % VHH and VHH MFI in T cells expressing the TFP constructs shown averaged across three donors.
- FIG.3 is a series of graphs showing the CD4:CD8 T cell ratio in T cells expressing the TFP constructs shown in three donors.
- FIG 4 is a series of flow plots showing TGF ⁇ RII and VHH expression in T cells expressing the TFP constructs shown in a representative donor. Also shown is a graph with average TGF ⁇ RII MFI in T cells expressing the TFP constructs shown averaged across three donors. [0117] FIG.5 is a series of graphs showing the memory phenotype of T cells expressing the TFP constructs shown in three donors.
- FIG.6 is a series of graphs showing cytokine expression (IFN ⁇ , GM-CSF, IL-2, and TNF- ⁇ ) by T cells expressing the TFP constructs shown when contacted with plate bound mesothelin in the presence or absence of 25 ng/ml TGF ⁇ , as described in Example 3.
- FIG.7 is a series of graphs showing cytokine expression (IL-2 and IFN ⁇ ) by T cells expressing the TFP constructs shown when contacted with plate bound mesothelin in the presence of TGF ⁇ at the concentrations shown, as described in Example 3.
- FIG.8 is a series of graphs showing cytokine expression (IFN ⁇ , GM-CSF, IL-2, and TNF- ⁇ ) by T cells expressing the TFP constructs shown when contacted with plate bound mesothelin and PD-L1 in the presence and absence of 25 ng/ml TGF ⁇ , as described in Example 3.
- FIG.9 is a schematic illustration of the protocol described in Example 4.
- FIG.10 is a series of graphs showing IL-2 expression by T cells expressing the TFP constructs shown when contacted with MSTO-MSLN, OVCAR3, or C30 target cells, in the presence or absence of 10 ng/ml TGF ⁇ , as described in Example 4.
- FIG.11 is a series of graphs showing cytokine expression (IFN ⁇ , GM-CSF, IL-2, and TNF- ⁇ ) by T cells expressing the TFP constructs shown when contacted with MSTO-MSLN in the presence of TGF ⁇ at the concentrations shown, as described in Example 4.
- FIG.12 is a schematic illustration of the protocol described in Example 5.
- FIG.13 is a series of flow plots showing expression of TGF ⁇ RII, PD-1, and VHH expression in T cells expressing the TFP constructs shown at 10 days of expansion.
- FIG.14 is a series of graphs showing pSMAD2/3 staining in CD4+ and CD8+ T cells incubated with 0, 0.1, 10, or 200 ng/ml TGF ⁇ , as described in Example 5.
- FIGs.15A and 15B are a series of graphs showing the level of pSMAD2/3 staining in T cells expressing the TFP constructs shown (VHH+) or untransduced cells from the same population (VHH-) incubated with 0, 0.1, 10, or 200 ng/ml TGF ⁇ , as described in Example 5.
- FIGs.16A and 16B show VHH expression and TGF ⁇ RII expression in T cells expressing the TFP constructs.
- FIG.16A provides a series of flow plots showing CD3 and VHH expression in cells expressing the TFP constructs from a representative donor and a bar graph with the %VHH+ cells, averaged across six donors.
- FIG.16B provides a series of flow plots showing TGF ⁇ RII and VHH expression in cells from the representative donor and a bar graph showing the TGF ⁇ RII MFI in VHH+ cells, averaged across six donors.
- FIG.17 shows the memory phenotype of CD4+ T cells expressing the TFP constructs. The flow plots provide the CD45RA and CCR7 staining of VHH+ CD4+ cells from a representative donor.
- FIG.18 shows the memory phenotype of CD8+ T cells expressing the TFP constructs.
- the flow plots provide the CD45RA and CCR7 staining of VHH+ CD8+ cells from a representative donor.
- a bar graph providing the frequencies of na ⁇ ve, TCM, TEM, and TEMRA cells in the CD8+ T cell compartment of cells expressing the TFP constructs, from six donors.
- FIG.19 shows the CD4:CD8 T cell ratio in T cells expressing the TFP constructs.
- FIG.20 is a series of bar graphs showing cytotoxicity of T cells expressing the TFP constructs against tumor cell lines C30 (ovarian cancer; no expression of MSLN), MSTO- MSLN (mesothelioma cancer; high expression of MSLN), and Suit-2 (pancreatic cancer, low expression of MSLN), at the indicated effector to target ratios (E:T).
- FIG.21A and FIG.21B are each a series of bar graphs showing cytokine production by T cells expressing the TFP constructs upon incubation with MSTO-MSLN (top row) or Suit-2 (bottom row) cell lines at decreasing E:T. Production of IFN ⁇ (left panels) and IL-2 (right panels) is shown in FIG.21A; and production of GM-CSF (left panels) and TNF- ⁇ (right panels) is shown in FIG.21B.
- FIG.22 is a representative set of histograms showing pSMAD2/3 staining in T cells expressing the indicated TFP constructs after incubation with TGF ⁇ , as described in Example 6.
- FIG.23 shows the pSMAD2/3 staining in T cells expressing the indicated TFP constructs after incubation with 0.01, 0.1, 1, 10, or 100 ng/mL TGF ⁇ .
- FIG.24A and FIG.24B are graphs showing cytokine expression by T cells expressing the TFP constructs shown, when contacted with MSTO-MSLN in the absence (0 ng/mL) or presence (1 ng/mL) of TGF ⁇ .
- FIG.24A shows the expression of IFN ⁇ (left panels), and IL-2 (right panels);
- FIG.24B shows the expression of GM-CSF (left panels) and TNF- ⁇ (right panels).
- FIG.25 shows the cytokine expression (IFN ⁇ , GM-CSF, IL-2, and TNF- ⁇ ) by T cells expressing the indicated TFP construct, when cultured with Suit-2, a TGF ⁇ secreting cell line.
- FIG.26 is a series of graphs showing cytokine expression (IFN ⁇ , GM-CSF, IL-2, and TNF- ⁇ ) by T cells expressing the indicated TFP construct, when cultured with Suit-2 cells and in the presence of increasing concentrations of TGF ⁇ .
- FIG.27A and FIG.27B are each a series of graphs showing the fold change in cytokine expression (GM-CSF, IFN ⁇ , IL-2, and TNF- ⁇ ) by T cells expressing the TFP constructs shown, when cultured with plate bound MSLN upon addition of 10 ng/mL TGF ⁇ .
- the data in FIG.27A and FIG 27B are from two different donors and show the fold change in cytokine levels comparing the 0 ng/mL to 10 ng/mL TGF ⁇ culture conditions.
- FIG.28A and FIG.28B are each a series of graphs showing cytokine expression (IFN ⁇ , GM-CSF, IL-2, and TNF- ⁇ ) by T cells expressing the TFP constructs shown, when cultured with plate bound MSLN in the presence the indicated increasing concentrations of TGF ⁇ .
- the data in FIG.28A and FIG 28B are from two different donors.
- FIG.29 shows the fold change in cell number (left panel) and VHH+ cell count (right panel) over time of T cells expressing the TFP constructs shown, upon repeated stimulation with Suit-2 cells.
- FIG.30 shows cytokine expression (IFN ⁇ , GM-CSF, IL-2, and TNF- ⁇ ) over time by T cells expressing the TFP constructs shown, upon repeated stimulation with Suit-2 cells.
- TCR T cell receptor
- the TFP can comprise (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain.
- the TCR subunit and the antigen binding domain can be operatively linked.
- the recombinant nucleic acid can comprise a second nucleic acid sequence encoding (i) a dominant negative form of transforming growth factor beta receptor II (TGFBr2) comprising a TGFBr2 extracellular domain or a functional fragment thereof, or (ii) a switch polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof.
- the first nucleic acid sequence and the second nucleic acid sequence can be present on the same or different nucleic acid molecules.
- the present disclosure further provides a vector comprising the recombinant nucleic acid, a cell comprising the recombinant nucleic acid or the vector described herein, or a pharmaceutical composition comprising the cell (e.g., modified cell).
- the present disclosure also provides a method of increasing an activity or cytotoxicity of a cell expressing a recombinant nucleic acid comprising a sequence encoding a T cell receptor (TCR) fusion protein (TFP).
- TCR T cell receptor
- TFP T cell receptor
- the method can comprise expressing a dominant negative and/or switch polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof in the cell.
- TGFBr2 transforming growth factor beta receptor II
- the present disclosure also provides a method of treating a disease such as cancer using the cell described herein.
- compositions and cells provided herein provide improved efficacy against tumors relative to modified T cells that lack a TGFBr2 switch or TGFBr2 dominant negative polypeptide or compositions thereof.
- the cells provided herein are capable of resisting and/or overcoming TGF ⁇ -mediated suppression and thus provide an effective therapy against solid tumors, given that TGF ⁇ is commonly found in the tumor microenvironment (TME) and is a potent suppressor of effector T cell function (Costanza et. al. IJC 145(6):1570-1584; 2019) (Flavell, et al. Nat Rev Immunol 10(8); 2010) (Gunderson et al, Nat Comm 11(1):1749; 2020).
- subject or “subjects” or “individuals” may include, but are not limited to, mammals such as humans or non-human mammals, e.g., domesticated, agricultural or wild, animals, as well as birds, and aquatic animals.
- “Patients” are subjects suffering from or at risk of developing a disease, disorder or condition or otherwise in need of the compositions and methods provided herein.
- the term “subject” is intended to include living organisms in which an immune response can be elicited (e.g., mammals, human).
- treating or “treatment” refers to any indicia of success in the treatment or amelioration of the disease or condition.
- Treating can include, for example, reducing, delaying or alleviating the severity of one or more symptoms of the disease or condition, or it can include reducing the frequency with which symptoms of a disease, defect, disorder, or adverse condition, and the like, are experienced by a patient.
- “treat or prevent” is sometimes used herein to refer to a method that results in some level of treatment or amelioration of the disease or condition, and contemplates a range of results directed to that end, including but not restricted to prevention of the condition entirely.
- “preventing” refers to the prevention of the disease or condition, e.g., tumor formation, in the patient.
- a “therapeutically effective amount” is the amount of a composition or an active component thereof sufficient to provide a beneficial effect or to otherwise reduce a detrimental non-beneficial event to the individual to whom the composition is administered.
- therapeutically effective dose herein is meant a dose that produces one or more desired or desirable (e.g., beneficial) effects for which it is administered, such administration occurring one or more times over a given period of time.
- the terms “dominant negative” or “dominant negative receptor” refer to a gene product, protein, or polypeptide that is designed to reduce the effect of a negative signal.
- a dominant negative polypeptide of the present disclosure reduces or eliminates the effect of a negative signal on a modified immune cell.
- a dominant negative receptor of the present disclosure may bind a signaling molecule (e.g. TGF ⁇ ) by way of an extracellular domain that associates with the molecule, and reduce or eliminate the effect of the molecule by way of a truncated, mutated, or otherwise disrupted signaling or kinase domain.
- a modified immune cell comprising a dominant negative receptor may bind to a signaling molecule (e.g., TGF ⁇ ), and reduce the effect the molecule would have on the cell in the absence of the dominant negative receptor.
- the term “fusion protein” relates to a protein which is made of polypeptide parts from different sources.
- fusion protein is used interchangeably with the term “switch polypeptide” or “switch-receptor” or “TGFBr2 switch” and the like.
- fusion proteins are proteins created through the joining of two or more genes (or, for example, cDNAs) that originally coded for separate proteins. Translation of this fusion gene (or, for example, fusion cDNA) results in a single polypeptide, for example, with functional properties derived from each of the original proteins.
- recombinant fusion proteins are created artificially by recombinant DNA technology for use in biological research or therapeutics. Further details to the exemplary production of the fusion protein of the present disclosure are described herein.
- the fusion protein further comprises an epitope tag.
- An epitope tag as described herein can be a peptide epitope tag or a protein epitope tag. Examples of a peptide epitope tag includes, but are not limited to, 6X His (also known as His-tag or hexahistidine tag (SEQ ID NO: 391)), FLAG (e.g., 3X FLAG), HA, Myc, and V5.
- a protein epitope tag examples include, but are not limited to, green fluorescent protein (GFP), glutathione-S-transferase (GST), ⁇ -galactosidase ( ⁇ -GAL), Luciferase, Maltose Binding Protein (MBP), Red Fluorescence Protein (RFP), and Vesicular Stomatitis Virus Glycoprotein (VSV-G).
- GFP green fluorescent protein
- GST glutathione-S-transferase
- ⁇ -GAL ⁇ -galactosidase
- Luciferase Maltose Binding Protein
- MBP Maltose Binding Protein
- RFP Red Fluorescence Protein
- VSV-G Vesicular Stomatitis Virus Glycoprotein
- the fusion protein further comprises a FLAG tag.
- the fusion protein further comprises a 3X FLAG tag.
- TGFBr2 switch refers to the TGFBr2 fusion proteins as described herein that receive an inhibitory signal by binding to, e.g., TGF-beta, and transform (e.g., “switch”) the signal via the co-stimulatory domain of the fusion protein into an activating signal.
- the TGFBr2 switch polypeptides provided herein lack a functional TGFBr2 kinase domain, and may function as a dominant negative receptor or exhibit functional characteristics of a dominant negative receptor.
- TGFBr2 or “transforming growth factor beta receptor II,” also known as transforming growth factor, beta receptor II, TGF beta receptor 2, TGFBR2, TGF ⁇ RII, TGFBRII, AAT3, FAA3, LDS1B, LDS2, LDS2B, MFS2, RIIC, TAAD2, TGFR-2, TGFbeta-RII, transforming growth factor beta receptor 2, TBR-ii, TBRII, refers to a protein that is a member of the serine/threonine protein kinase family and the TGFB receptor subfamily.
- TGFBr2 when used in the context of the functional or wild-type protein, refers to a transmembrane protein that has a protein kinase domain, forms a heterodimeric complex with another receptor protein (e.g., TGFBr1), and binds TGF-beta.
- TGFBr2 includes any of the recombinant or naturally-occurring forms of TGFBr2 or variants or homologs thereof that have or maintain TGFBr2 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring TGFBr2.
- TGFBr2 is substantially identical to the protein identified by the UniProt reference number P37173 or a variant or homolog having substantial identity thereto.
- TGFBr2 dominant negative refers to a TGFBr2 protein that lacks a functional kinase domain.
- the dominant negative TGFBr2 comprises a TGFBr2 extracellular domain that is capable of binding to or capturing TGF ⁇ ; and lacks a kinase domain.
- the dominant negative TGFBr2 comprises a truncated or mutated intracellular domain that is not capable of phosphorylation of the GS domain of TGFBr1.
- the dominant negative TGFBr2 is capable of binding to TGF ⁇ and complexing with TGFBr1, but is incapable of phosphorylating the GS domain of TGFBr1 such that the effect of TGF ⁇ on a cell expressing the dominant negative TGFBr2 is reduced or eliminated.
- Exemplary dominant negative forms of TGFBr2 are provided herein.
- GMCSFRa also known as CSF2RA, CD116, Cluster of Differentiation 116, CDw116, CSF2R, CSF2RAX, CSF2RAY, CSF2RX, CSF2RY, GM-CSF-R-alpha, GMCSFR, GMR, SMDP4, colony stimulating factor 2 receptor alpha subunit, alphaGMR, colony stimulating factor 2 receptor subunit alpha, GMR-alpha, GMCSFR-alpha, granulocyte-macrophage colony-stimulating factor receptor, as used herein, refers to a receptor for granulocyte-macrophage colony-stimulating factor, which stimulates the production of white blood cells.
- GM-CSF and its receptor play a role in earlier stages of development.
- GMCSFRa is associated with Surfactant metabolism dysfunction type 4.
- GMCSFRa includes any of the recombinant or naturally-occurring forms of GMCSFRa or variants or homologs thereof that have or maintain GMCSFRa activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring GMCSFRa.
- GMCSFRa is substantially identical to the protein identified by the UniProt reference number P15509 or a variant or homolog having substantial identity thereto.
- CD28 also known as Cluster of Differentiation 28, CD28, Tp44, and CD28 molecule, as used herein, refers to a protein expressed on T cells that provides co- stimulatory signals required for T cell activation and survival.
- CD28 is the receptor for CD80 (B7.1) and CD86 (B7.2) proteins.
- CD28 includes any of the recombinant or naturally-occurring forms of CD28 or variants or homologs thereof that have or maintain CD28 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD28.
- CD28 is substantially identical to the protein identified by the UniProt reference number P10747 or a variant or homolog having substantial identity thereto.
- 2A 2A self-cleaving peptide
- 2A peptide refers to a class of peptides, which can induce ribosomal skipping during translation of a protein in a cell. These peptides share a core sequence motif of DxExNPGP (SEQ ID NO: 392), and are found in a wide range of viral families. Exemplary members of 2A include, but are not limited to, P2A, E2A, F2A, and T2A. “T2A” refers to the 2A derived from thosea asigna virus, and the sequence is EGRGSLLTCGDVEENPGP (SEQ ID NO: 23).
- P2A refers to the 2A derived from porcine teschovirus-12A, and the sequence is ATNFSLLKQAGDVEENPGP (SEQ ID NO: 269).
- E2A refers to the 2A derived from quine rhinitis A virus, and the sequence is QCTNYALLKLAGDVESNPGP (SEQ ID NO: 280).
- F2A is derived from foot-and-mouth disease virus 18, and the sequence is VKQTLNFDLLKLAGDVESNPGP (SEQ ID NO: 281).
- adding the l linker “GSG” (Gly-Ser-Gly) on the N-terminal of a 2A peptide helps with efficiency.
- a “T cell receptor (TCR) fusion protein” or “TFP,” as used herein, includes a recombinant polypeptide derived from the various polypeptides comprising the TCR that is generally capable of i) binding to a surface antigen on target cells and ii) interacting with other polypeptide components of the intact TCR complex, typically when co-located in or on the surface of a T cell.
- stimulation refers to a primary response induced by binding of a stimulatory domain or stimulatory molecule (e.g., a TCR/CD3 complex) with its cognate ligand thereby mediating a signal transduction event, such as, but not limited to, signal transduction via the TCR/CD3 complex.
- a stimulatory domain or stimulatory molecule e.g., a TCR/CD3 complex
- signal transduction event such as, but not limited to, signal transduction via the TCR/CD3 complex.
- Stimulation can mediate altered expression of certain molecules, and/or reorganization of cytoskeletal structures, and the like.
- the term “stimulatory molecule” or “stimulatory domain,” as used herein, refers to a molecule or portion thereof expressed by a T cell that provides the primary cytoplasmic signaling sequence(s) that regulate primary activation of the TCR complex in a stimulatory way for at least some aspect of the T cell signaling pathway.
- the primary signal is initiated by, for instance, binding of a TCR/CD3 complex with an MHC molecule loaded with peptide, and which leads to mediation of a T cell response, including, but not limited to, proliferation, activation, differentiation, and the like.
- a primary cytoplasmic signaling sequence (also referred to as a “primary signaling domain”) that acts in a stimulatory manner may contain a signaling motif which is known as immunoreceptor tyrosine-based activation motif or “ITAM”.
- ITAM immunoreceptor tyrosine-based activation motif
- Examples of an ITAM containing primary cytoplasmic signaling sequence that is of particular use in the invention includes, but is not limited to, those derived from TCR zeta, FcR gamma, FcR beta, CD3 gamma, CD3 delta, CD3 epsilon, CD5, CD22, CD79a, CD79b, CD278 (also known as “ICOS”) and CD66d.
- T cells may recognize these complexes using their T cell receptors (TCRs).
- APCs process antigens and present them to T cells.
- MHC Major histocompatibility complex
- the complex may be on the surface of an antigen presenting cell, such as a dendritic cell or a B cell, or any other cell, including cancer cells, or it may be immobilized by, for example, coating on to a bead or plate.
- HLA human leukocyte antigen system
- MHC major histocompatibility complex
- HLA alleles A, B and C present peptides derived mainly from intracellular proteins, e.g., proteins expressed within the cell.
- T cells undergo a positive selection step to ensure recognition of self MHCs followed by a negative step to remove T cells that bind too strongly to MHC which present self-antigens.
- certain T cells and the TCRs they express will only recognize peptides presented by certain types of MHC molecules - i.e., those encoded by particular HLA alleles. This is known as HLA restriction.
- one HLA allele of interest is HLA-A*0201, which is expressed in the vast majority (>50%) of the Caucasian population. Accordingly, TCRs which bind WT1 peptides presented by MHC encoded by HLA-A*0201 (i.e.
- HLA- A*0201 restricted are advantageous since an immunotherapy making use of such TCRs will be suitable for treating a large proportion of the Caucasian population.
- other HLA- A alleles of interest are HLA-A*0101, HLA- A*2402, and HLA-A*0301.
- widely expressed HLA-B alleles of interest are HLA-B*3501, HLA-B*0702 and HLA-B*3502.
- intracellular signaling domain refers to an intracellular portion of a molecule.
- the intracellular signaling domain generates a signal that promotes an immune effector function of the TFP containing cell, e.g., a modified T-T cell.
- immune effector function e.g., in a modified T-T cell
- examples of immune effector function, e.g., in a modified T-T cell include, but are not limited to, cytolytic activity and T helper cell activity, including the secretion of cytokines.
- the intracellular signaling domain comprises a primary intracellular signaling domain. Exemplary primary intracellular signaling domains include, but are not limited to, those derived from the molecules responsible for primary stimulation, or antigen dependent simulation.
- the intracellular signaling domain comprises a costimulatory intracellular domain.
- a primary intracellular signaling domain comprises an ITAM (“immunoreceptor tyrosine-based activation motif”).
- ITAM immunoglobulin-based activation motif
- Examples of ITAM containing primary cytoplasmic signaling sequences include, but are not limited to, those derived from CD3 zeta, FcR gamma, FcR beta, CD3 gamma, CD3 delta, CD3 epsilon, CD5, CD22, CD79a, CD79b, and CD66d DAP10 and DAP12.
- costimulatory molecule refers to the cognate binding partner on a T cell that specifically binds with a costimulatory ligand, thereby mediating a costimulatory response by the T cell, such as, but not limited to, proliferation.
- costimulatory molecules are cell surface molecules other than antigen receptors or their ligands that are required for an efficient immune response.
- Exemplary costimulatory molecules include, but are not limited to, an MHC class 1 molecule, BTLA and a Toll ligand receptor, as well as OX40, CD2, CD27, CD28, CD5, ICAM-1, LFA-1 (CD11a/CD18), 4-1BB (CD137), IL-15Ra, IL12R, IL18R, IL21R, ICOS (CD278), GITR, CD30, CD40, BAFFR, HVEM, CD7, LIGHT, NKG2C, SLAMF7, NKp80, CD160, CD226, Fc ⁇ RI, Fc ⁇ RII, and Fc ⁇ RIII.
- OX40 CD2, CD27, CD28, CD5, ICAM-1, LFA-1 (CD11a/CD18), 4-1BB (CD137), IL-15Ra, IL12R, IL18R, IL21R, ICOS (CD278), GITR, CD30, CD40, BAFFR, HVEM, CD7,
- a costimulatory intracellular signaling domain is the intracellular portion of a costimulatory molecule.
- a costimulatory molecule is represented in the following protein families: TNF receptor proteins, Immunoglobulin-like proteins, cytokine receptors, integrins, signaling lymphocytic activation molecules (SLAM proteins), and activating NK cell receptors.
- Examples of such molecules include CD27, CD28, 4-1BB (CD137), OX40, GITR, CD30, CD40, ICOS, BAFFR, HVEM, lymphocyte function-associated antigen-1 (LFA-1), CD2, CD7, LIGHT, NKG2C, SLAMF7, NKp80, CD160, B7-H3, and a ligand that specifically binds with CD83, IL-15Ra, IL12R, IL18R, IL21R, CD27, CD5, ICAM-1, CD7, CD226, Fc ⁇ RI, Fc ⁇ RII, Fc ⁇ RIII, and the like.
- the intracellular signaling domain comprises the entire intracellular portion or the entire native intracellular signaling domain of the molecule from which it is derived, or a functional fragment thereof.
- 4-1BB refers to a member of the TNFR superfamily with an amino acid sequence provided as GenBank Acc. No. AAA62478.2, or the equivalent residues from a non-human species, e.g., mouse, rodent, monkey, ape and the like; and a “4- 1BB costimulatory domain,” as used herein, refers to amino acid residues 214-255 of GenBank Acc. No.
- 4-1BB also known as TNFRSF9, 4-1BB, CD137, Cluster of Differentiation 137, CDw137, ILA, tumor necrosis factor receptor superfamily member 9, and TNF receptor superfamily member 9, as used herein, includes any of the recombinant or naturally-occurring forms of 4-1BB or variants or homologs thereof that have or maintain 4-1BB activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring 4-1BB.
- 4-1BB is substantially identical to the protein identified by the UniProt reference number Q07011 or a variant or homolog having substantial identity thereto.
- the term “antibody,” as used herein, refers to a protein, or polypeptide sequences derived from an immunoglobulin molecule, which specifically binds to an antigen.
- Antibodies can be intact immunoglobulins of polyclonal or monoclonal origin, or fragments thereof and can be derived from natural or from recombinant sources.
- antibody fragment refers to at least one portion of an antibody, or recombinant variants thereof, that contains the antigen binding domain, i.e., an antigenic determining variable region of an intact antibody, that is sufficient to confer recognition and specific binding of the antibody fragment to a target, such as an antigen and its defined epitope.
- antibody fragments include, but are not limited to, Fab, Fab’, F(ab’) 2 , and Fv fragments, single-chain (sc)Fv (“scFv”) antibody fragments, linear antibodies, single domain antibodies such as sdAb (either VL or VH), camelid VHH domains, and multi-specific antibodies formed from antibody fragments.
- scFv refers to a fusion protein comprising at least one antibody fragment comprising a variable region of a light chain and at least one antibody fragment comprising a variable region of a heavy chain, wherein the light and heavy chain variable regions are contiguously linked via a short flexible polypeptide linker, and capable of being expressed as a single polypeptide chain, and wherein the scFv retains the specificity of the intact antibody from which it is derived.
- the term “Heavy chain variable region” or “VH” with regard to an antibody refers to the fragment of the heavy chain that contains three CDRs interposed between flanking stretches known as framework regions.
- a scFv may have the V L and V H variable regions in either order, e.g., with respect to the N-terminal and C-terminal ends of the polypeptide.
- the scFv may comprise VL-linker-VH or may comprise VH-linker-VL.
- the portion of the TFP composition of the disclosure comprising an antibody or antibody fragment thereof may exist in a variety of forms where the antigen binding domain is expressed as part of a contiguous polypeptide chain including, for example, a single domain antibody fragment (sdAb), or a single chain antibody (scFv) derived from a murine, humanized or human antibody (Harlow et al., 1999, In: Using Antibodies: A Laboratory Manual, Cold Spring Harbor Laboratory Press, N.Y.; Harlow et al., 1989, In: Antibodies: A Laboratory Manual, Cold Spring Harbor, N.Y.; Houston et al., 1988, Proc. Natl. Acad. Sci.
- sdAb single domain antibody fragment
- scFv single chain antibody
- the antigen binding domain of a TFP composition of the disclosure comprises an antibody fragment.
- the TFP comprises an antibody fragment that comprises a scFv or a sdAb.
- the term “recombinant antibody,” as used herein, refers to an antibody that is generated using recombinant DNA technology, such as, for example, an antibody expressed by a bacteriophage or yeast expression system.
- the term should also be construed to mean an antibody which has been generated by the synthesis of a DNA molecule encoding the antibody and which DNA molecule expresses an antibody protein, or an amino acid sequence specifying the antibody, wherein the DNA or amino acid sequence has been obtained using recombinant DNA or amino acid sequence technology which is available and well known in the art.
- antigen or “Ag,” as used herein, refers to a molecule that is capable of being bound specifically by an antibody, or otherwise provokes an immune response. In some embodiments, this immune response may involve either antibody production, or the activation of specific immunologically-competent cells, or both.
- antigens can be derived from recombinant or genomic DNA.
- any DNA which comprises a nucleotide sequences or a partial nucleotide sequence encoding a protein that elicits an immune response therefore encodes an “antigen” as that term is used herein.
- an antigen need not be encoded solely by a full length nucleotide sequence of a gene.
- an antigen need not be encoded by a “gene” at all. It is readily apparent that an antigen can be generated synthesized or can be derived from a biological sample, or might be macromolecule besides a polypeptide.
- a biological sample can include, but is not limited to a tissue sample, a tumor sample, a cell or a fluid with other biological components.
- CD3 or “Cluster of Differentiation 3,” as used herein, refers to a protein complex and T cell co-receptor that is involved in activating both the cytotoxic T cell and T helper cells. In some embodiments, it is composed of four distinct chains. For example, in some embodiments, the complex contains a CD3 ⁇ chain, a CD3 ⁇ chain, and two CD3 ⁇ chains in mammals.
- CD3 ⁇ includes any of the recombinant or naturally-occurring forms of CD3 ⁇ or variants or homologs thereof that have or maintain CD3 ⁇ activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD3 ⁇ .
- CD3 ⁇ is substantially identical to the protein identified by the UniProt reference number P07766 or a variant or homolog having substantial identity thereto.
- CD3 ⁇ includes any of the recombinant or naturally-occurring forms of CD3 ⁇ or variants or homologs thereof that have or maintain CD3 ⁇ activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD3 ⁇ .
- CD3 ⁇ is substantially identical to the protein identified by the UniProt reference number P04234 or a variant or homolog having substantial identity thereto.
- CD3 ⁇ includes any of the recombinant or naturally-occurring forms of CD3 ⁇ or variants or homologs thereof that have or maintain CD3 ⁇ activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD3 ⁇ .
- CD3 ⁇ is substantially identical to the protein identified by the UniProt reference number P09693 or a variant or homolog having substantial identity thereto.
- CD19 also known as B-lymphocyte antigen CD19, B4, CVID3, and CD19 molecule, refers to the Cluster of Differentiation 19 protein, which is an antigenic determinant detectable on B cell leukemia precursor cells, other malignant B cells and most cells of the normal B cell lineage.
- CD19 includes any of the recombinant or naturally-occurring forms of CD19 or variants or homologs thereof that have or maintain CD19 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD19.
- CD19 is substantially identical to the protein identified by the UniProt reference number P15391 or a variant or homolog having substantial identity thereto.
- BCMA refers to the B-cell maturation antigen, also known as tumor necrosis factor receptor superfamily member 17 (TNFRSF17), Cluster of Differentiation 269 protein (CD269), BCM, TNFRSF13A, tumor necrosis factor receptor superfamily member 17, and TNF receptor superfamily member 17, which is a protein that in humans is encoded by the TNFRSF17 gene.
- TNFRSF17 is a cell surface receptor of the TNF receptor superfamily which recognizes B-cell activating factor (BAFF) (see, e.g., Laabi et al., EMBO 11 (11): 3897–904 (1992).
- BCMA includes any of the recombinant or naturally-occurring forms of BCMA or variants or homologs thereof that have or maintain BCMA activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring BCMA.
- BCMA is substantially identical to the protein identified by the UniProt reference number Q02223 or a variant or homolog having substantial identity thereto.
- CD16 also known as Fc ⁇ RIII, refers to a cluster of differentiation molecule found on the surface of natural killer cells, neutrophil polymorphonuclear leukocytes, monocytes, and macrophages. CD16 has been identified as Fc receptors Fc ⁇ RIIIa (CD16a) and Fc ⁇ RIIIb (CD16b), which participate in signal transduction. In some embodiments, CD16 is a molecule of the immunoglobulin superfamily (IgSF) involved in antibody-dependent cellular cytotoxicity (ADCC).
- IgSF immunoglobulin superfamily
- CD16 includes any of the recombinant or naturally-occurring forms of CD16 or variants or homologs thereof that have or maintain CD16 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD16.
- CD16 is substantially identical to the protein identified by the UniProt reference number P08637 (CD16a) or a variant or homolog having substantial identity thereto or the protein identified by the UniProt reference number O7501 (CD16b) or a variant or homolog having substantial identity thereto.
- the term “NKG2D,” also known as KLRK1, CD314, D12S2489E, KLR, NKG2-D, NKG2D, natural killer group 2D, killer cell lectin-like receptor K1, and killer cell lectin like receptor K1 refers to a transmembrane protein belonging to the CD94/NKG2 family of C-type lectin-like receptors.
- NKG2D is expressed by NK cells, ⁇ T cells and CD8+ ⁇ T cells.
- NKG2D recognizes induced-self proteins from MIC and RAET1/ULBP families which appear on the surface of stressed, malignant transformed, and infected cells.
- NKG2D includes any of the recombinant or naturally-occurring forms of NKG2D or variants or homologs thereof that have or maintain NKG2D activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring NKG2D.
- NKG2D is substantially identical to the protein identified by the UniProt reference number P26718 or a variant or homolog having substantial identity thereto.
- mesothelin also known as MPF and SMRP, refers to a tumor differentiation antigen that is normally present on the mesothelial cells lining the pleura, peritoneum and pericardium. In some embodiments, mesothelin is over-expressed in several human tumors, including mesothelioma and ovarian and pancreatic adenocarcinoma.
- MSLN includes any of the recombinant or naturally-occurring forms of MSLN or variants or homologs thereof that have or maintain MSLN activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring MSLN.
- MSLN is substantially identical to the protein identified by the UniProt reference number Q13421 or a variant or homolog having substantial identity thereto.
- ROR1 includes any of the recombinant or naturally-occurring forms of ROR1 or variants or homologs thereof that have or maintain ROR1 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring ROR1.
- ROR1 is substantially identical to the protein identified by the UniProt reference number Q01973 or a variant or homolog having substantial identity thereto.
- MUC16 also known as mucin 16, cell-surface associated, ovarian cancer- related tumor marker CA125, CA-125 (cancer antigen 125, carcinoma antigen 125, or carbohydrate antigen 125), mucin 16, and CA125, refers to a membrane-tethered mucin that contains an extracellular domain at its amino terminus, a large tandem repeat domain, and a transmembrane domain with a short cytoplasmic domain.
- products of this gene have been used as a marker for different cancers, with higher expression levels associated with poorer outcomes.
- MUC16 includes any of the recombinant or naturally-occurring forms of MUC16 or variants or homologs thereof that have or maintain MUC16 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring MUC16.
- MUC16 is substantially identical to the protein identified by the UniProt reference number Q8WXI7 or a variant or homolog having substantial identity thereto.
- CD22 also known as cluster of differentiation-22, sialic acid binding Ig- like lectin 2, SIGLEC-2, SIGLEC2, CD22 molecule, T cell surface antigen leu-14, and B cell receptor CD22, refers to a protein that mediates B cell/B cell interactions, and is thought to be involved in, e.g., the localization of B cells in lymphoid tissues.
- CD22 is associated with diseases including, but not limited to, refractory hematologic cancer and hairy cell leukemia.
- CD22 includes any of the recombinant or naturally-occurring forms of CD22 or variants or homologs thereof that have or maintain CD22 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD22.
- CD22 is substantially identical to the protein identified by the UniProt reference number P20273 or a variant or homolog having substantial identity thereto.
- PD-1 is a cell surface receptor that belongs to the immunoglobulin superfamily and is expressed on T cells and pro-B cells. PD-1 binds two ligands, PD-L1 and PD-L2.
- PD-1 includes any of the recombinant or naturally-occurring forms of PD-1 or variants or homologs thereof that have or maintain PD-1 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring PD-1.
- PD-1 is substantially identical to the protein identified by the UniProt reference number Q15116 or a variant or homolog having substantial identity thereto.
- PD-L1 may play a major role in suppressing the adaptive arm of immune system during particular events such as, e.g., pregnancy, tissue allografts, autoimmune disease and other disease states such as, e.g., hepatitis.
- the adaptive immune system reacts to antigens that are associated with immune system activation by exogenous or endogenous danger signals.
- clonal expansion of antigen-specific CD8+ T cells and/or CD4+ helper cells is propagated.
- the binding of PD-L1 to the inhibitory checkpoint molecule PD-1 transmits an inhibitory signal based on interaction with phosphatases (SHP-1 or SHP-2) via Immunoreceptor Tyrosine-Based Switch Motif (ITSM) motif.
- SHP-1 or SHP-2 phosphatases
- IRS Immunoreceptor Tyrosine-Based Switch Motif
- PD-L1 includes any of the recombinant or naturally-occurring forms of PD-L1 or variants or homologs thereof that have or maintain PD-L1 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring PD-L1.
- PD-L1 is substantially identical to the protein identified by the UniProt reference number Q9NZQ7 or a variant or homolog having substantial identity thereto.
- CD79 ⁇ (Cluster of Differentiation 79 ⁇ ) and “CD79 ⁇ (Cluster of Differentiation 79 ⁇ )” genes encode proteins that make up the B lymphocyte antigen receptor, a multimeric complex that includes the antigen-specific component, surface immunoglobulin (Ig).
- Ig surface immunoglobulin
- Surface Ig non-covalently associates with two other proteins, Ig-alpha and Ig-beta (encoded by CD79 ⁇ and its paralog CD79 ⁇ , respectively) which are necessary for expression and function of the B-cell antigen receptor. Functional disruption of this complex can lead to, e.g., human B-cell chronic lymphocytic leukemias.
- CD79 ⁇ protein includes any of the recombinant or naturally-occurring forms of CD79 ⁇ protein or variants or homologs thereof that have or maintain CD79 ⁇ protein activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD79 ⁇ protein.
- CD79 ⁇ protein is substantially identical to the protein identified by the UniProt reference number P11912 or a variant or homolog having substantial identity thereto.
- CD79 ⁇ protein includes any of the recombinant or naturally-occurring forms of CD79 ⁇ protein or variants or homologs thereof that have or maintain CD79 ⁇ protein activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD79 ⁇ protein.
- CD79 ⁇ protein is substantially identical to the protein identified by the UniProt reference number P40259 or a variant or homolog having substantial identity thereto.
- B cell activating factor also known as tumor necrosis factor ligand superfamily member 13B, TNFSF13B, BLYS, CD257, DTL, TALL-1, TALL1, THANK, TNFSF20, ZTNF4, TNLG7A, tumor necrosis factor superfamily member 13b, and TNF superfamily member 13b, refers to a cytokine that belongs to the tumor necrosis factor (TNF) ligand family.
- TNF tumor necrosis factor
- This cytokine is a ligand for receptors TNFRSF13B/TACI, TNFRSF17/BCMA, and TNFRSF13C/BAFF-R.
- BAFF plays an important role in the proliferation and differentiation of B cells.
- BAFF includes any of the recombinant or naturally-occurring forms of BAFF or variants or homologs thereof that have or maintain BAFF activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring BAFF.
- BAFF is substantially identical to the protein identified by the UniProt reference number Q9Y275 or a variant or homolog having substantial identity thereto.
- CD70-CD27 pathway plays an important role in the generation and maintenance of T cell immunity, in particular, during antiviral responses.
- CD70 Upon CD27 binding, CD70 induces the proliferation of co-stimulated T-cells and enhances the generation of cytolytic T-cells.
- CD70 includes any of the recombinant or naturally-occurring forms of CD70 or variants or homologs thereof that have or maintain CD70 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD70.
- CD70 is substantially identical to the protein identified by the UniProt reference number P32970 or a variant or homolog having substantial identity thereto.
- the term “interleukin 15 receptor” or “IL-15R” refers to a type I cytokine receptor that IL-15 binds to and signals through.
- IL-15R is composed of three subunits: IL-15 receptor alpha chain (“IL-15R ⁇ ” or CD215), IL-2 receptor beta chain (“IL- 2R ⁇ ” or CD122) and IL-2 receptor gamma/the common gamma chain (“IL-2R ⁇ / ⁇ c” or CD132).
- IL-15R ⁇ IL-15 receptor alpha chain
- IL-2R ⁇ IL-2 receptor beta chain
- IL-2R ⁇ / ⁇ c the common gamma chain
- human IL-15R ⁇ precursor protein has a 30 amino acid signal peptide, a 175 amino acid extracellular domain, a 23 amino acid single membrane-spanning transmembrane stretch, and a 39 amino acid cytoplasmic (or intracellular) domain and contains N- and O-linked glycosylation sites.
- IL-15R ⁇ contains a Sushi domain (amino acid 31-95), which is essential for IL-15 binding.
- IL-15R ⁇ exists as a soluble form (sIL-15R ⁇ ).
- sIL-15R ⁇ is constitutively generated from the transmembrane receptor through a defined proteolytic cleavage, and this process can be enhanced by certain chemical agents, such as PMA.
- the human sIL-15R ⁇ about 42 kDa in size, may prolong the half-life of IL-15 or potentiate IL-15 signaling through IL-15 binding and IL- 2R ⁇ / ⁇ c heterodimer.
- IL-15R shares subunits with IL-2R that contain the cytoplasmic motifs required for signal transduction
- IL-15 signaling has separate biological effects in vivo apart from many biological activities overlapping with IL-2 signaling due to IL-15R ⁇ subunit that is unique to IL-15R, availability and concentration of IL-15, the kinetics and affinity of IL-15-IL-15R ⁇ binding.
- IL-15 binds to IL-15R ⁇ specifically with high affinity, which then associates with a complex composed of IL-2R ⁇ and IL-2R ⁇ / ⁇ c subunits, expressed on the same cell (“cis-presentation”) or on a different cell (“trans-presentation”).
- the interaction between IL-15 and IL-15R ⁇ is independent of the complex composed of IL-2R ⁇ and IL-2R ⁇ / ⁇ c subunits.
- IL-15 binding to the IL-2R ⁇ / ⁇ c heterodimeric receptor induces JAK1 activation that phosphorylates STAT3 via the beta chain, and JAK3 activation that phosphorylates STAT5 via the gamma chain.
- the IL- 15/IL-15R interaction modulates T-cell development and homeostasis in memory CD8+ T- cell.
- the IL-15/IL-15R interaction also modulates NK cell development, maintenance, expansion and activities.
- IL-15R ⁇ also known as CD215, IL-15 receptor subunit alpha, IL-15R-alpha, IL- 15RA, and Interleukin-15 receptor subunit alpha, as used herein, includes any of the recombinant or naturally-occurring forms of IL-15R ⁇ or variants or homologs thereof that have or maintain IL-15R ⁇ activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring IL-15R ⁇ .
- IL-15R ⁇ is substantially identical to the protein identified by the UniProt reference number Q13261 or a variant or homolog having substantial identity thereto.
- IL-2R ⁇ also known as CD122, IL-2 receptor subunit beta, IL-2R subunit beta, IL- 2RB, P70-75, IMD63, and Interleukin-2 receptor subunit beta, as used herein, includes any of the recombinant or naturally-occurring forms of IL-2R ⁇ or variants or homologs thereof that have or maintain IL-2R ⁇ activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring IL-2R ⁇ .
- IL-2R ⁇ is substantially identical to the protein identified by the UniProt reference number P14784 or a variant or homolog having substantial identity thereto.
- IL-2 receptor gamma/the common gamma chain also known as IL-2R ⁇ / ⁇ c, IL2RG, CIDX, IL-2RG, IMD4, P64, SCIDX, SCIDX1, interleukin 2 receptor subunit gamma, or CD132, as used herein, includes any of the recombinant or naturally-occurring forms of IL- 2R ⁇ / ⁇ c or variants or homologs thereof that have or maintain IL-2R ⁇ / ⁇ c activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity).
- the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring IL-2R ⁇ / ⁇ c.
- IL- 2R ⁇ / ⁇ c is substantially identical to the protein identified by the UniProt reference number P31785 or a variant or homolog having substantial identity thereto.
- IL-15R ⁇ cytoplasmic (or intracellular) domain comprises amino acids 229-267 of IL-15R ⁇ protein. In some embodiments, IL-15R ⁇ cytoplasmic (or intracellular) domain comprises a sequence of SEQ ID NO: 372. In some embodiments, IL- 15R ⁇ Sushi domain comprises amino acids 31-95 of IL-15R ⁇ protein. In some embodiments, IL-15R ⁇ Sushi domain comprises a sequence of SEQ ID NO: 382. In some embodiments, IL- 15R ⁇ comprises the transmembrane domain and the cytoplasmic (intracellular) domain of IL- 15R ⁇ protein. In some embodiments, IL-15R ⁇ comprises amino acids 96-267 of IL-15R ⁇ protein.
- IL-15R ⁇ comprises a sequence of SEQ ID NO: 383. In some embodiments, sIL-15R ⁇ comprises amino acids 21-205 of IL-15R ⁇ protein. In some embodiments, sIL-15R ⁇ comprises a sequence of SEQ ID NO: 379.
- SEQ ID NO: 372 (IL-15R ⁇ intracellular domain) KSRQTPPLASVEMEAMEALPVTWGTSSRDEDLENCSHHL [0207]
- SEQ ID NO: 379 (Soluble IL-15R ⁇ (sIL-15R ⁇ )) ITCPPPMSVEHADIWVKSYSLYSRERYICNSGFKRKAGTSSLTECVLNKATNVAHWT TPSLKCIRDPALVHQRPAPPSTVTTAGVTPQPESLSPSGKEPAASSPSSNNTAATTAAI VPGSQLMPSKSPSTGTTEISSHESSHGTPSQTTAKNWELTASASHQPPGVYPQGHSDT T [0208]
- SEQ ID NO: 382 (IL-15 Sushi domain) ITCPPPMSVEHADIWVKSYSLYSRERYICNSGFKRKAGTSSLTECVLNKATNVAHWT TPSLKCIR [0209]
- SEQ ID NO: 383 (IL-15R ⁇ region downstream of Sushi domain) DPALVHQRPAPP
- transfer vector refers to a composition of matter which comprises an isolated nucleic acid and which can be used to deliver the isolated nucleic acid to the interior of a cell.
- Numerous vectors are known in the art including, but not limited to, linear polynucleotides, polynucleotides associated with ionic or amphiphilic compounds, plasmids, and viruses.
- the term “transfer vector” includes an autonomously replicating plasmid or a virus.
- the term should also be construed to further include non-plasmid and non-viral compounds which facilitate transfer of nucleic acid into cells, such as, for example, a polylysine compound, liposome, and the like.
- viral transfer vectors examples include, but are not limited to, adenoviral vectors, adeno-associated virus vectors, retroviral vectors, lentiviral vectors, and the like.
- expression vector refers to a vector comprising a recombinant polynucleotide comprising expression control sequences operatively linked to a nucleotide sequence to be expressed.
- An expression vector comprises sufficient cis-acting elements for expression; other elements for expression can be supplied by the host cell or in an in vitro expression system.
- Expression vectors include all those known in the art, including cosmids, plasmids (e.g., naked or contained in liposomes) and viruses (e.g., lentiviruses, retroviruses, adenoviruses, and adeno-associated viruses) that incorporate the recombinant polynucleotide.
- viruses e.g., lentiviruses, retroviruses, adenoviruses, and adeno-associated viruses
- lentivirus refers to a genus of the Retroviridae family. Lentiviruses are unique among the retroviruses in being able to infect non-dividing cells; they can deliver a significant amount of genetic information into the DNA of the host cell, so they are one of the most efficient methods of a gene delivery vector.
- lentiviral vector refers to a vector derived from at least a portion of a lentivirus genome, including especially a self-inactivating lentiviral vector as provided in Milone et al., Mol. Ther.17(8): 1453-1464 (2009).
- Other examples of lentivirus vectors that may be used in the clinic include but are not limited to, e.g., the LENTIVECTOR TM gene delivery technology from Oxford BioMedica, the LENTIMAX TM vector system from Lentigen, and the like. Nonclinical types of lentiviral vectors are also available and would be known to one skilled in the art.
- homologous refers to the subunit sequence identity between two polymeric molecules, e.g., between two nucleic acid molecules, such as, two DNA molecules or two RNA molecules, or between two polypeptide molecules.
- two nucleic acid molecules such as, two DNA molecules or two RNA molecules
- polypeptide molecules between two polypeptide molecules.
- a subunit position in both of the two molecules is occupied by the same monomeric subunit; e.g., if a position in each of two DNA molecules is occupied by adenine, then they are homologous or identical at that position.
- the homology between two sequences is a direct function of the number of matching or homologous positions; e.g., if half (e.g., five positions in a polymer ten subunits in length) of the positions in two sequences are homologous, the two sequences are 50% homologous; if 90% of the positions (e.g., 9 of 10), are matched or homologous, the two sequences are 90% homologous.
- “Humanized” forms of non-human (e.g., murine) antibodies are chimeric immunoglobulins, immunoglobulin chains or fragments thereof (such as Fv, Fab, Fab’, F(ab’)2 or other antigen-binding subsequences of antibodies) which contain minimal sequence derived from non-human immunoglobulin.
- humanized antibodies and antibody fragments thereof are human immunoglobulins (recipient antibody or antibody fragment) in which residues from a complementary-determining region (CDR) of the recipient are replaced by residues from a CDR of a non-human species (donor antibody) such as mouse, rat or rabbit having the desired specificity, affinity, and capacity.
- Fv framework region (FR) residues of the human immunoglobulin are replaced by corresponding non-human residues.
- a humanized antibody/antibody fragment can comprise residues which are found neither in the recipient antibody nor in the imported CDR or framework sequences. These modifications can further refine and optimize antibody or antibody fragment performance.
- the humanized antibody or antibody fragment thereof will comprise substantially all of at least one, and typically two, variable domains, in which all or substantially all of the CDR regions correspond to those of a non-human immunoglobulin and all or a significant portion of the FR regions are those of a human immunoglobulin sequence.
- the humanized antibody or antibody fragment can also comprise at least a portion of an immunoglobulin constant region (Fc), typically that of a human immunoglobulin.
- “Human” or “fully human,” as used herein, refers to an immunoglobulin, such as an antibody or antibody fragment, where the whole molecule is of human origin or consists of an amino acid sequence identical to a human form of the antibody or immunoglobulin.
- the term “isolated,” as used herein, means altered or removed from the natural state.
- nucleic acid or a peptide naturally present in a living animal is not “isolated,” but the same nucleic acid or peptide partially or completely separated from the coexisting materials of its natural state is “isolated.”
- An isolated nucleic acid or protein can exist in substantially purified form, or can exist in a non-native environment such as, for example, a host cell.
- nucleic acid bases are used. “A” refers to adenosine, “C” refers to cytosine, “G” refers to guanosine, “T” refers to thymidine, and “U” refers to uridine.
- conservative sequence modifications refers to amino acid modifications that do not significantly affect or alter the binding characteristics of the antibody or antibody fragment containing the amino acid sequence. Such conservative modifications include amino acid substitutions, additions and deletions. Modifications can be introduced into an antibody or antibody fragment of the present disclosure by standard techniques known in the art, such as site-directed mutagenesis and PCR-mediated mutagenesis. Conservative amino acid substitutions are ones in which the amino acid residue is replaced with an amino acid residue having a similar side chain. Families of amino acid residues having similar side chains have been defined in the art.
- amino acids with basic side chains e.g., lysine, arginine, histidine
- acidic side chains e.g., aspartic acid, glutamic acid
- uncharged polar side chains e.g., glycine, asparagine, glutamine, serine, threonine, tyrosine, cysteine, tryptophan
- nonpolar side chains e.g., alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine
- beta-branched side chains e.g., threonine, valine, isoleucine
- aromatic side chains e.g., tyrosine, phenylalanine, tryptophan, histidine
- operably linked refers to functional linkage between a regulatory sequence and a heterologous nucleic acid sequence resulting in expression of the latter.
- a first nucleic acid sequence is operably linked with a second nucleic acid sequence when the first nucleic acid sequence is placed in a functional relationship with the second nucleic acid sequence.
- a promoter is operably linked to a coding sequence if the promoter affects the transcription or expression of the coding sequence.
- nucleotide refers to deoxyribonucleic acids (DNA) or ribonucleic acids (RNA) and polymers thereof in either single- or double-stranded form.
- RNA ribonucleic acids
- the term encompasses nucleic acids containing known analogues of natural nucleotides that have similar binding properties as the reference nucleic acid and are metabolized in a manner similar to naturally occurring nucleotides.
- nucleic acid sequence also implicitly encompasses conservatively modified variants thereof (e.g., degenerate codon substitutions), alleles, orthologs, SNPs, and complementary sequences as well as the sequence explicitly indicated.
- degenerate codon substitutions may be achieved by generating sequences in which the third position of one or more selected (or all) codons is substituted with mixed-base and/or deoxyinosine residues (Batzer et al., Nucleic Acid Res.19:5081 (1991); Ohtsuka et al., J. Biol. Chem.260:2605-2608 (1985); and Rossolini et al., Mol. Cell.
- peptide refers to a compound comprised of amino acid residues covalently linked by peptide bonds.
- a protein or peptide must contain at least two amino acids, and no limitation is placed on the maximum number of amino acids that can comprise a protein’s or peptide’s sequence.
- Polypeptides include any peptide or protein comprising two or more amino acids joined to each other by peptide bonds.
- polypeptides include, for example, biologically active fragments, substantially homologous polypeptides, oligopeptides, homodimers, heterodimers, variants of polypeptides, modified polypeptides, derivatives, analogs, fusion proteins, among others.
- a polypeptide includes a natural peptide, a recombinant peptide, or a combination thereof.
- promoter refers to a DNA sequence recognized by the transcription machinery of the cell, or introduced synthetic machinery, required to initiate the specific transcription of a polynucleotide sequence.
- promoter/regulatory sequence refers to a nucleic acid sequence which is required for expression of a gene product operably linked to the promoter/regulatory sequence. In some instances, this sequence may be the core promoter sequence and in other instances, this sequence may also include an enhancer sequence and other regulatory elements which are required for expression of the gene product.
- the promoter/regulatory sequence may, for example, be one which expresses the gene product in a tissue specific manner.
- the term “constitutive” promoter refers to a nucleotide sequence which, when operably linked with a polynucleotide which encodes or specifies a gene product, causes the gene product to be produced in a cell under most or all physiological conditions of the cell.
- the term “inducible” promoter refers to a nucleotide sequence which, when operably linked with a polynucleotide which encodes or specifies a gene product, causes the gene product to be produced in a cell substantially only when an inducer which corresponds to the promoter is present in the cell.
- tissue-specific promoter refers to a nucleotide sequence which, when operably linked with a polynucleotide encodes or specified by a gene, causes the gene product to be produced in a cell substantially only if the cell is a cell of the tissue type corresponding to the promoter.
- linker and “flexible polypeptide linker” as used in the context of a scFv refers to a peptide linker that consists of amino acids such as glycine and/or serine residues used alone or in combination, to link variable heavy and variable light chain regions together.
- the flexible polypeptide linker is a Gly/Ser linker and comprises the amino acid sequence (Gly-Gly-Gly-Ser) n , where n is a positive integer equal to or greater than 1 (SEQ ID NO: 393).
- n a positive integer equal to or greater than 1
- the flexible polypeptide linkers include, but are not limited to, (Gly4Ser)4 (SEQ ID NO: 394) or (Gly4Ser)3 (SEQ ID NO: 395).
- a 5’ cap (also termed an RNA cap, an RNA 7-methylguanosine cap or an RNA m7G cap) is a modified guanine nucleotide that has been added to the “front” or 5’ end of a eukaryotic messenger RNA shortly after the start of transcription.
- the 5’ cap consists of a terminal group which is linked to the first transcribed nucleotide. Its presence is critical for recognition by the ribosome and protection from RNases. Cap addition is coupled to transcription, and occurs co-transcriptionally, such that each influences the other.
- in vitro transcribed RNA refers to RNA, preferably mRNA, which has been synthesized in vitro. Generally, the in vitro transcribed RNA is generated from an in vitro transcription vector.
- the in vitro transcription vector comprises a template that is used to generate the in vitro transcribed RNA.
- a “poly(A)” refers to a series of adenosines attached by polyadenylation to the mRNA.
- the polyA is between 50 and 5000, preferably greater than 64, more preferably greater than 100, most preferably greater than 300 or 400 (SEQ ID NO: 399).
- Poly(A) sequences can be modified chemically or enzymatically to modulate mRNA functionality such as localization, stability or efficiency of translation.
- polyadenylation refers to the covalent linkage of a polyadenylyl moiety, or its modified variant, to a messenger RNA molecule.
- mRNA messenger RNA
- 3’ poly(A) tail is a long sequence of adenine nucleotides (often several hundred) added to the pre-mRNA through the action of an enzyme, polyadenylate polymerase.
- polyadenylate polymerase an enzyme that catalyzes the adenylation of adenine nucleotides
- the poly(A) tail is added onto transcripts that contain a specific sequence, the polyadenylation signal.
- the poly(A) tail and the protein bound to it aid in protecting mRNA from degradation by exonucleases. Polyadenylation is also important for transcription termination, export of the mRNA from the nucleus, and translation.
- Polyadenylation occurs in the nucleus immediately after transcription of DNA into RNA, but additionally can also occur later in the cytoplasm.
- the mRNA chain is cleaved through the action of an endonuclease complex associated with RNA polymerase.
- the cleavage site is usually characterized by the presence of the base sequence AAUAAA near the cleavage site.
- adenosine residues are added to the free 3’ end at the cleavage site.
- transient refers to expression of a non-integrated transgene for a period of hours, days or weeks, wherein the period of time of expression is less than the period of time for expression of the gene if integrated into the genome or contained within a stable plasmid replicon in the host cell.
- signal transduction pathway refers to the biochemical relationship between a variety of signal transduction molecules that play a role in the transmission of a signal from one portion of a cell to another portion of a cell.
- cell surface receptor includes molecules and complexes of molecules capable of receiving a signal and transmitting signal across the membrane of a cell.
- a “substantially purified” cell refers to a cell that is essentially free of other cell types.
- a substantially purified cell also refers to a cell which has been separated from other cell types with which it is normally associated in its naturally occurring state.
- a population of substantially purified cells refers to a homogenous population of cells. In other instances, this term refers simply to cell that have been separated from the cells with which they are naturally associated in their natural state.
- the cells are cultured in vitro. In other aspects, the cells are not cultured in vitro.
- nucleic acid refers to deoxyribonucleic acids (DNA) or ribonucleic acids (RNA) and polymers thereof in either single- or double-stranded form. Unless specifically limited, the term encompasses nucleic acids containing known analogues of natural nucleotides that have similar binding properties as the reference nucleic acid and are metabolized in a manner similar to naturally occurring nucleotides. Unless otherwise indicated, a particular nucleic acid sequence also implicitly encompasses conservatively modified variants thereof (e.g., degenerate codon substitutions), alleles, orthologs, SNPs, and complementary sequences as well as the sequence explicitly indicated.
- DNA deoxyribonucleic acids
- RNA ribonucleic acids
- degenerate codon substitutions may be achieved by generating sequences in which the third position of one or more selected (or all) codons is substituted with mixed-base and/or deoxyinosine residues (Batzer et al., Nucleic Acid Res.19:5081 (1991); Ohtsuka et al., J. Biol. Chem.260:2605-2608 (1985); and Rossolini et al., Mol. Cell. Probes 8:91-98 (1994)).
- the term “transfected” or “transformed” or “transduced,” as used herein, refers to a process by which exogenous nucleic acid is transferred or introduced into the host cell.
- a “transfected” or “transformed” or “transduced” cell is one which has been transfected, transformed or transduced with exogenous nucleic acid.
- the cell includes the primary subject cell and its progeny.
- the term “specifically binds,” as used herein, refers to an antibody, an antibody fragment or a specific ligand, which recognizes and binds a cognate binding partner (e.g., CD19) present in a sample, but which does not necessarily and substantially recognize or bind other molecules in the sample.
- tumor antigen or “hyperproliferative disorder antigen” or “antigen associated with a hyperproliferative disorder” refers to antigens that are common to specific hyperproliferative disorders.
- the hyperproliferative disorder antigens of the present disclosure are derived from, cancers including but not limited to primary or metastatic melanoma, thymoma, lymphoma, sarcoma, lung cancer, liver cancer, NHL, leukemias, uterine cancer, cervical cancer, bladder cancer, kidney cancer and adenocarcinomas such as breast cancer, prostate cancer, ovarian cancer, pancreatic cancer, and the like.
- anti-tumor effect refers to a biological effect which can be manifested by various means, including but not limited to, e.g., a decrease in tumor volume, a decrease in the number of tumor cells, a decrease in the number of metastases, an increase in life expectancy, decrease in tumor cell proliferation, decrease in tumor cell survival, or amelioration of various physiological symptoms associated with the cancerous condition.
- an “anti-tumor effect” can also be manifested by the ability of the peptides, polynucleotides, cells and antibodies of the present disclosure in prevention of the occurrence of tumor in the first place.
- autologous refers to any material derived from the same individual to whom it is later to be re-introduced into the individual.
- allogeneic or, alternatively, “allogenic,” as used herein, refers to any material derived from a different animal of the same species or different patient as the individual to whom the material is introduced. Two or more individuals are said to be allogeneic to one another when the genes at one or more loci are not identical. In some aspects, allogeneic material from individuals of the same species may be sufficiently unlike genetically to interact antigenically.
- xenogeneic refers to a graft derived from an animal of a different species.
- cancer refers to a disease characterized by the rapid and uncontrolled growth of aberrant cells. Cancer cells can spread locally or through the bloodstream and lymphatic system to other parts of the body. Examples of various cancers are described herein and include but are not limited to, breast cancer, prostate cancer, ovarian cancer, cervical cancer, skin cancer, pancreatic cancer, colorectal cancer, renal cancer, liver cancer, brain cancer, lymphoma, leukemia, lung cancer and the like.
- the term “encoding,” as used herein, refers to the inherent property of specific sequences of nucleotides in a polynucleotide, such as a gene, a cDNA, or an mRNA, to serve as templates for synthesis of other polymers and macromolecules in biological processes having either a defined sequence of nucleotides (e.g., rRNA, tRNA and mRNA) or a defined sequence of amino acids and the biological properties resulting therefrom.
- a gene, cDNA, or RNA encodes a protein if transcription and translation of mRNA corresponding to that gene produces the protein in a cell or other biological system.
- nucleotide sequence encoding an amino acid sequence includes all nucleotide sequences that are degenerate versions of each other and that encode the same amino acid sequence.
- nucleotide sequence that encodes a protein or an RNA may also include introns to the extent that the nucleotide sequence encoding the protein may in some versions contain one or more introns.
- the terms “effective amount” and “therapeutically effective amount,” as used herein, are used interchangeably herein, and refer to an amount of a compound, formulation, material, or composition, as described herein effective to achieve a particular biological or therapeutic result.
- the term “endogenous,” as used herein, refers to any material from or produced inside an organism, cell, tissue or system.
- the term “exogenous,” as used herein, refers to any material introduced from or produced outside an organism, cell, tissue or system.
- expression refers to the transcription and/or translation of a particular nucleotide sequence driven by a promoter.
- parenteral administration of an immunogenic composition includes, e.g., subcutaneous (s.c.), intravenous (i.v.), intramuscular (i.m.), or intrasternal injection, intratumoral, or infusion techniques.
- s.c. subcutaneous
- i.v. intravenous
- i.m. intramuscular
- intrasternal injection intratumoral, or infusion techniques.
- therapeutic means a treatment. A therapeutic effect is obtained by reduction, suppression, remission, or eradication of a disease state.
- prophylaxis as used herein, means the prevention of or protective treatment for a disease or disease state.
- a functional disruption refers to a physical or biochemical change to a specific (e.g., target) nucleic acid (e.g., gene, RNA transcript, of protein encoded thereby) that prevents its normal expression and/or behavior in the cell.
- a functional disruption refers to a modification of the gene via a gene editing method.
- a functional disruption prevents expression of a target gene (e.g., an endogenous gene).
- target gene e.g., an endogenous gene.
- the term “meganuclease” refers to an endonuclease that binds double- stranded DNA at a recognition sequence that is greater than 12 base pairs.
- the recognition sequence for a meganuclease of the present disclosure is 22 base pairs.
- a meganuclease may be an endonuclease that is derived from I-Crel and may refer to an engineered variant of I-Crel that has been modified relative to natural I-Crel with respect to, for example, DNA-binding specificity, DNA cleavage activity, DNA-binding affinity, or dimerization properties. Methods for producing such modified variants of I-Crel are known in the art (e.g., WO 2007/047859).
- a meganuclease binds to double-stranded DNA as a heterodimer or as a “single-chain meganuclease” in which a pair of DNA-binding domains are joined into a single polypeptide using a peptide linker.
- the term “homing endonuclease” is synonymous with the term “meganuclease.”
- meganucleases are substantially non-toxic when expressed in cells, particularly in human T cells, such that cells may be transfected and maintained at 37°C without observing deleterious effects on cell viability or significant reductions in meganuclease cleavage activity when measured using the methods described herein.
- single-chain meganuclease refers to a polypeptide comprising a pair of nuclease subunits joined by a linker.
- a single-chain meganuclease has the organization: N-terminal subunit - Linker - C-terminal subunit.
- the two meganuclease subunits may generally be non-identical in amino acid sequence and may recognize non-identical DNA sequences.
- single-chain meganucleases typically cleave pseudo-palindromic or non-palindromic recognition sequences.
- a single-chain meganuclease may be referred to as a “single-chain heterodimer” or “single-chain heterodimeric meganuclease” although it is not, in fact, dimeric.
- the term “meganuclease” can refer to a dimeric or single-chain meganuclease.
- TALEN refers to an endonuclease comprising a DNA- binding domain comprising 16-22 TAL domain repeats fused to any portion of the Fokl nuclease domain.
- the term “Compact TALEN” refers to an endonuclease comprising a DNA-binding domain with 16-22 TAL domain repeats fused in any orientation to any catalytically active portion of nuclease domain of the I-Tevl homing endonuclease.
- CRISPR refers to a caspase-based endonuclease comprising a caspase, such as Cas9, and a guide RNA that directs DNA cleavage of the caspase by hybridizing to a recognition site in the genomic DNA.
- the term “megaTAL” refers to a single-chain nuclease comprising a transcription activator-like effector (TALE) DNA binding domain with an engineered, sequence-specific homing endonuclease.
- TALE transcription activator-like effector
- T cell receptor and “T cell receptor complex” are used interchangeably to refer to a molecule found on the surface of T cells that is, in general, responsible for recognizing antigens.
- the TCR comprises a heterodimer consisting of a TCR alpha and TCR beta chain in 95% of T cells, whereas 5% of T cells have TCRs consisting of TCR gamma and TCR delta chains.
- the TCR further comprises one or more of CD3 ⁇ , CD3 ⁇ , and CD3 ⁇ .
- the TCR comprises CD3 ⁇ .
- the TCR comprises CD3 ⁇ .
- the TCR comprises CD3 ⁇ .
- the TCR comprises CD3 ⁇ .
- the TCR comprises CD3 ⁇ .
- the TCR comprises CD3 ⁇ .
- the constant domain of human TCR alpha has a sequence of SEQ ID NO: 142.
- the constant domain of human TCR alpha has an IgC domain having a sequence of SEQ ID NO: 143, a transmembrane domain having a sequence of SEQ ID NO: 144, and an intracellular domain having a sequence of SS (SEQ ID NO: 145).
- the constant domain of murine TCR alpha has a sequence of SEQ ID NO: 147.
- the constant domain of murine TCR alpha has a transmembrane domain having a sequence of SEQ ID NO: 144, and an intracellular domain having a sequence of SS (SEQ ID NO: 145).
- the constant domain of human TCR beta has a sequence of SEQ ID NO: 148.
- the constant domain of human TCR beta has an IgC domain having a sequence of SEQ ID NO: 149, a transmembrane domain having a sequence of SEQ ID NO: 150, and an intracellular domain having a sequence of SEQ ID NO: 151.
- the constant domain of murine TCR beta has a sequence of SEQ ID NO: 152.
- the constant domain of murine TCR beta has a transmembrane domain having a sequence of SEQ ID NO: 152, and an intracellular domain having a sequence of SEQ ID NO: 153.
- the constant domain of human TCR delta has a sequence of SEQ ID NO: 243.
- the constant domain of human TCR delta has an IgC domain having a sequence of SEQ ID NO: 265, a transmembrane domain having a sequence of SEQ ID NO: 158, and an intracellular domain having a sequence of L.
- the constant domain of human TCR gamma has a sequence of SEQ ID NO: 21.
- the constant domain of human TCR gamma has an IgC domain having a sequence of SEQ ID NO: 155, a transmembrane domain having a sequence of SEQ ID NO: 156, and an intracellular domain having a sequence of SEQ ID NO: 157.
- human CD3 epsilon has a sequence of SEQ ID NO: 258.
- human CD3 epsilon has an extracellular domain having a sequence of SEQ ID NO: 126, a transmembrane domain having a sequence of SEQ ID NO: 127, and an intracellular domain, e.g., an intracellular signaling domain, having a sequence of SEQ ID NO: 128.
- human CD3 delta has a sequence of SEQ ID NO: 136.
- human CD3 delta has an extracellular domain having a sequence of SEQ ID NO: 138, a transmembrane domain having a sequence of SEQ ID NO: 139, and an intracellular domain, e.g., an intracellular signaling domain, having a sequence of SEQ ID NO: 140.
- human CD3 gamma has a sequence of SEQ ID NO: 130.
- human CD3 gamma has an extracellular domain having a sequence of SEQ ID NO: 132, a transmembrane domain having a sequence of SEQ ID NO: 133, and an intracellular domain, e.g., an intracellular signaling domain, having a sequence of SEQ ID NO: 134.
- Ranges throughout this disclosure, various aspects of the present disclosure can be presented in a range format. It should be understood that the description in range format is merely for convenience and brevity and should not be construed as an inflexible limitation on the scope of the present disclosure. Accordingly, the description of a range should be considered to have specifically disclosed all the possible subranges as well as individual numerical values within that range. For example, description of a range such as from 1 to 6 should be considered to have specifically disclosed subranges such as from 1 to 3, from 1 to 4, from 1 to 5, from 2 to 4, from 2 to 6, from 3 to 6 etc., as well as individual numbers within that range, for example, 1, 2, 2.7, 3, 4, 5, 5.3, and 6.
- a range such as 95- 99% identity includes something with 95%, 96%, 97%, 98% or 99% identity, and includes subranges such as 96-99%, 96-98%, 96-97%, 97-99%, 97-98% and 98-99% identity. This applies regardless of the breadth of the range.
- compositions of matter and methods of use for the treatment of a disease such as cancer using recombinant nucleic acids comprising: a first nucleic acid sequence encoding a T cell receptor (TCR) fusion protein (TFP), wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain; and wherein the TCR subunit and the antigen binding domain are operatively linked, and a second nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof.
- TCR T cell receptor
- TFP T cell receptor fusion protein
- a “T cell receptor (TCR) fusion protein” or “TFP” includes a recombinant polypeptide derived from the various polypeptides comprising the TCR that is generally capable of i) binding to a surface antigen on target cells and ii) interacting with other polypeptide components of the intact TCR complex, typically when co-located in or on the surface of a T cell.
- TFPs provide substantial benefits as compared to Chimeric Antigen Receptors.
- CAR Chimeric Antigen Receptor
- a CAR refers to a recombinant polypeptide comprising an extracellular antigen binding domain in the form of, e.g., a single domain antibody or scFv, a transmembrane domain, and cytoplasmic signaling domains (also referred to herein as “intracellular signaling domains”) comprising a functional signaling domain derived from a stimulatory molecule as defined below.
- intracellular signaling domains also referred to herein as “intracellular signaling domains”
- the central intracellular signaling domain of a CAR is derived from the CD3 zeta chain that is normally found associated with the TCR complex.
- the CD3 zeta signaling domain can be fused with one or more functional signaling domains derived from at least one co-stimulatory molecule such as 4-1BB (i.e., CD137), CD27 and/or CD28.
- Dominant Negative TGFBR2 polypeptides [0268] Provided herein are recombinant nucleic acids comprising: a first nucleic acid sequence encoding a T cell receptor (TCR) fusion protein (TFP) wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain, wherein the TCR subunit and the antigen binding domain are operatively linked; and a second nucleic acid sequence encoding a dominant negative form of transforming growth factor beta receptor II (TGFBr2).
- TCR T cell receptor
- TFP T cell receptor fusion protein
- TFP comprises
- the first nucleic acid sequence and the second nucleic acid sequence are operatively linked by a linker.
- the linker comprises a protease cleavage site.
- the protease cleavage site is a 2A cleavage site.
- the cleavage site can be a self-cleaving peptide such as a T2A, P2A, E2A or F2A cleavage site.
- the 2A cleavage site is a T2A cleavage site or a P2A cleavage site.
- the dominant negative TGFBr2 comprises a TGFBr2 extracellular domain and does not comprise a functional kinase domain. In some embodiments, the dominant negative TGFBr2 comprises a truncated TGFBr2 intracellular domain. In some embodiments, the dominant negative TGFBr2 comprises or consists of a TGFBr2 extracellular domain and a TGFBR2 transmembrane domain. In some embodiments, the dominant negative TGFBr2 comprises a truncated or non-functional kinase domain.
- the dominant negative TGFBr2 comprises or consists of about 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 50, 75, 100, 150, or 200 amino acids of a TGFBr2 intracellular domain.
- the dominant negative TGFBr2 comprises no more than about 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids of SEQ ID NO: 294.
- the intracellular domain portion of the dominant negative TGFBr2 consists of SEQ ID NO: 389.
- the dominant negative TGFBr2 comprises a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to SEQ ID NO: 68.
- the dominant negative TGFBr2 comprises the sequence of SEQ ID NO: 68.
- the sequence of the dominant negative TGFBr2 extracellular domain is the sequence of SEQ ID NO:68.
- the dominant negative TGFBr2 polypeptide comprises an addition of amino acid residue(s) to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of a sequence of a dominant negative TGFBr2 polypeptide as described herein.
- the dominant negative TGFBr2 polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C- terminal ends of a sequence of a dominant negative TGFBr2 polypeptide as described herein.
- the dominant negative TGFBr2 polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N- terminal end, C-terminal end, or both N-terminal and C-terminal ends of a sequence of a dominant negative TGFBr2 polypeptide as described herein.
- the dominant negative TGFBr2 polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C- terminal end, or both N-terminal and C-terminal ends, of SEQ ID NO: 68.
- the dominant negative TGFBr2 polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N- terminal end, C-terminal end, or both N-terminal and C-terminal ends of SEQ ID NO: 68.
- the dominant negative TGFBr2 polypeptide comprises amino acid residue deletions from the N-terminal end, C-terminal end, or both N-terminal and C- terminal ends of a sequence of a dominant negative TGFBr2 polypeptide as described herein.
- the dominant negative TGFBr2 polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids deleted from the N-terminal or C-terminal end of a sequence of a dominant negative TGFBr2 polypeptide as described herein.
- the dominant negative TGFBr2 polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids deleted from the N-terminal or C-terminal end of a sequence of a dominant negative TGFBr2 polypeptide as described herein.
- the dominant negative TGFBr2 polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids deleted from the N-terminal or C-terminal end of SEQ ID NO: 68.
- the dominant negative TGFBr2 polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids deleted from the N-terminal or C-terminal end of SEQ ID NO: 68.
- the dominant negative TGFBr2 polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently deleted from both N-terminal and C-terminal ends of a sequence of a dominant negative TGFBr2 polypeptide as described herein.
- the dominant negative TGFBr2 polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently deleted from both N-terminal and C- terminal ends of a sequence of a dominant negative TGFBr2 polypeptide as described herein.
- the dominant negative TGFBr2 polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently deleted from both N-terminal and C-terminal ends of SEQ ID NO: 68.
- the dominant negative TGFBr2 polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently deleted from both N-terminal and C- terminal ends of SEQ ID NO: 68.
- the dominant negative TGFBr2 polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently mutated as compared to SEQ ID NO: 68.
- the dominant negative TGFBr2 polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids mutated as compared to SEQ ID NO: 68.
- Switch Polypeptides [0274] Provided herein are recombinant nucleic acids comprising: a first nucleic acid sequence encoding a T cell receptor (TCR) fusion protein (TFP) wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain; and wherein the TCR subunit and the antigen binding domain are operatively linked, and a second nucleic acid sequence encoding a switch polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof.
- TCR T cell receptor
- TFP T cell receptor fusion protein
- TGFBr2 transforming growth factor beta receptor II
- the first nucleic acid sequence and the second nucleic acid sequence are operatively linked by a linker.
- the linker comprises a protease cleavage site.
- the protease cleavage site is a 2A cleavage site.
- the cleavage site can be a self-cleaving peptide such as a T2A, P2A, E2A or F2A cleavage site.
- the 2A cleavage site is a T2A cleavage site or a P2A cleavage site.
- the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286. In some embodiments, the switch polypeptide comprises any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286.
- the sequence of the switch polypeptide is a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, or 99.9% sequence identity to any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286. In some embodiments, the sequence of the switch polypeptide is any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286. [0276] In some embodiments, the switch polypeptide comprises an addition of amino acid residue(s) to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of a sequence of a switch polypeptide as described herein.
- the switch polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of a sequence of a switch polypeptide as described herein.
- the switch polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of a sequence of a switch polypeptide as described herein.
- the switch polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286.
- the switch polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286.
- the switch polypeptide comprises amino acid residue deletions from the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of a sequence of a switch polypeptide as described herein.
- the switch polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids deleted from the N-terminal or C-terminal end of a sequence of a switch polypeptide as described herein.
- the switch polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids deleted from the N-terminal or C-terminal end of a sequence of a switch polypeptide as described herein.
- the switch polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids deleted from the N-terminal or C-terminal end of any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286.
- the switch polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids deleted from the N-terminal or C-terminal end of any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286.
- the switch polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently deleted from both N-terminal and C-terminal ends of a sequence of a switch polypeptide as described herein.
- the switch polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently deleted from both N-terminal and C-terminal ends of a sequence of a switch polypeptide as described herein.
- the switch polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently deleted from both N-terminal and C-terminal ends of any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286.
- the switch polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently deleted from both N-terminal and C-terminal ends of any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286.
- the TGFBr2 extracellular domain comprises a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to SEQ ID NO:271.
- the TGFBr2 extracellular domain comprises the sequence of SEQ ID NO:271.
- the sequence of the TGFBr2 extracellular domain is a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, or 99.9% sequence identity to SEQ ID NO:271.
- the sequence of the TGFBr2 extracellular domain is the sequence of SEQ ID NO:271.
- the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having an addition of amino acid residue(s) to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of the TGFBr2 extracellular domain as described herein.
- the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of the TGFBr2 extracellular domain as described herein.
- the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C- terminal ends of the sequence of the TGFBr2 extracellular domain as described herein.
- the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:271.
- the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:271.
- the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having a deletion of amino acid residue(s) from the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of the TGFBr2 extracellular domain as described herein.
- the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, or 130 amino acids deleted from the N-terminal or C-terminal end of the sequence of the TGFBr2 extracellular domain as described herein.
- the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, or 130 amino acids deleted from the N-terminal or C-terminal end of the sequence of the TGFBr2 extracellular domain as described herein.
- the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, or 130 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:271.
- the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, or 130 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:271.
- the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, or 130 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of the TGFBr2 extracellular domain as described herein.
- the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, or 130 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of the TGFBr2 extracellular domain as described herein.
- the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, or 130 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:271.
- the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, or 130 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:271.
- TGFBr2 extracellular domain TGFBR2 ecto: TIPPHVQKSVNNDMIVTDNNGAVKFPQLCKFCDVRFSTCDNQKSCMSNCSITSICEKP QEVCVAVWRKNDENITLETVCHDPKLPYHDFILEDAASPKCIMKEKKKPGETFFMCS CSSDECNDNIIFSEEYNTSNPDLLLVIFQ (SEQ ID NO:271) Switch Intracellular Domain
- the switch polypeptide further comprises a switch intracellular domain.
- the TGFBr2 extracellular domain is operably linked to the switch intracellular domain.
- the switch intracellular domain comprises an intracellular domain of a costimulatory polypeptide.
- the costimulatory polypeptide is selected from the group consisting of CD28, 4-1BB(CD137), IL-15Ra, IL12R, IL18R, IL21R, OX40, CD2, CD27, CD5, ICAM-1, ICOS (CD278), GITR, CD30, CD40, BAFFR, HVEM, CD7, LIGHT, NKG2C, SLAMF7, NKp80, CD160, CD226, Fc ⁇ RI, Fc ⁇ RII, and Fc ⁇ RIII.
- the costimulatory polypeptide is CD28.
- the costimulatory polypeptide is 4-1BB.
- the costimulatory polypeptide is IL-15Ra.
- the switch intracellular domain comprises a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to or SEQ ID NO:273 or SEQ ID NO:277. In some embodiments, the switch intracellular domain comprises the sequence of SEQ ID NO:273 or SEQ ID NO:277.
- the sequence of the switch intracellular domain is a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:273 or SEQ ID NO:277.
- the sequence of the switch intracellular domain is the sequence of SEQ ID NO:273 or SEQ ID NO:277.
- the switch polypeptide comprises an intracellular domain sequence having an addition of amino acid residue(s) to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the intracellular domain sequence as described herein.
- the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the intracellular domain sequence as described herein.
- the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the intracellular domain sequence as described herein.
- the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:273 or SEQ ID NO:277.
- the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:273 or SEQ ID NO:277.
- the switch polypeptide comprises an intracellular domain sequence having a deletion of amino acid residue(s) from the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the intracellular domain sequence as described herein.
- the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids deleted from the N-terminal or C-terminal end of the intracellular domain sequence as described herein.
- the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids deleted from the N-terminal or C-terminal end of the intracellular domain sequence as described herein. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids deleted from the N-terminal or C- terminal end of the sequence of SEQ ID NO:273 or SEQ ID NO:277.
- the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:273 or SEQ ID NO:277.
- the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids independently deleted from both N-terminal and C-terminal ends of the intracellular domain sequence as described herein.
- the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids independently deleted from both N-terminal and C-terminal ends of the intracellular domain sequence as described herein.
- the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:273 or SEQ ID NO:277.
- the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:273 or SEQ ID NO:277.
- the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a 4-IBB intracellular domain or a fragment thereof, a MyD88 intracellular domain or a fragment thereof, an ICOS intracellular domain or a fragment thereof, a CTLA4 intracellular domain or a fragment thereof, a CD28 intracellular domain or a fragment thereof, a CD200R intracellular domain or a fragment thereof, a BTLA intracellular domain or a fragment thereof, a TIM-3 intracellular domain or a fragment thereof, a TIGIT intracellular domain or a fragment thereof, a 4-1BB intracellular domain or a fragment thereof, an IL-10RA intracellular domain or a fragment thereof, an IL-7RA intracellular domain or a fragment thereof, a Fas intracellular domain or a fragment thereof, a TRAIL-R2 intracellular domain or a fragment thereof, a PD-1 intracellular domain or a fragment thereof, an IL-4RA intracellular domain or a fragment thereof, an
- an IL12R intracellular domain or a fragment thereof is an IL- 12 receptor subunit beta-1 intracellular domain or a fragment thereof. In some embodiments, an IL12R intracellular domain or a fragment thereof is an IL-12 receptor subunit beta-2 intracellular domain or a fragment thereof. In some embodiments, an IL18R intracellular domain or a fragment thereof is an interleukin-18 receptor 1 intracellular domain or a fragment thereof.
- the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human 4-IBB intracellular domain or a fragment thereof, a human MyD88 intracellular domain or a fragment thereof, a human ICOS intracellular domain or a fragment thereof, a human CTLA4 intracellular domain or a fragment thereof, a human CD28 intracellular domain or a fragment thereof, a human CD200R intracellular domain or a fragment thereof, a human BTLA intracellular domain or a fragment thereof, a human TIM-3 intracellular domain or a fragment thereof, a human TIGIT intracellular domain or a fragment thereof, a human 4-1BB intracellular domain or a fragment thereof, a human IL-10RA intracellular domain or a fragment thereof, a human IL-7RA intracellular domain or a fragment thereof, a Fas intracellular domain or a fragment thereof, a TRAIL-R2 intracellular domain or a fragment thereof, a human PD-1 intracellular domain or a fragment thereof, a Fas intracellular domain
- a human IL12R intracellular domain or a fragment thereof is a human IL-12 receptor subunit beta-1 intracellular domain or a fragment thereof. In some embodiments, a human IL12R intracellular domain or a fragment thereof is a human IL-12 receptor subunit beta-2 intracellular domain or a fragment thereof. In some embodiments, a human IL18R intracellular domain or a fragment thereof is a human interleukin-18 receptor 1 intracellular domain or a fragment thereof. [0290] In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a CD28 intracellular domain or a fragment thereof and a CTLA4 intracellular domain or a fragment thereof.
- the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human CD28 intracellular domain or a fragment thereof and a human CTLA4 intracellular domain or a fragment thereof.
- the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a Myd88 intracellular domain or a fragment thereof and a TGF ⁇ R2 intracellular domain or a fragment thereof.
- the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human Myd88 intracellular domain or a fragment thereof and a human TGF ⁇ R2 intracellular domain or a fragment thereof.
- the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a CD28 intracellular domain or a fragment thereof and a Fas intracellular domain or a fragment thereof. In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human CD28 intracellular domain or a fragment thereof and a human Fas intracellular domain or a fragment thereof. [0293] In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a 4-1BB intracellular domain or a fragment thereof and a Fas intracellular domain or a fragment thereof.
- the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human 4-1BB intracellular domain or a fragment thereof and a human Fas intracellular domain or a fragment thereof.
- the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a MyD88 intracellular domain or a fragment thereof and a Fas intracellular domain or a fragment thereof.
- the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human MyD88 intracellular domain or a fragment thereof and a human Fas intracellular domain or a fragment thereof.
- the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to an ICOS intracellular domain or a fragment thereof and a Fas intracellular domain or a fragment thereof. In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human ICOS intracellular domain or a fragment thereof and a human Fas intracellular domain or a fragment thereof. [0296] In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a CD28 intracellular domain or a fragment thereof and a TRAIL-R2 intracellular domain or a fragment thereof.
- the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human CD28 intracellular domain or a fragment thereof and a human TRAIL-R2 intracellular domain or a fragment thereof.
- the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a MyD88 intracellular domain or a fragment thereof and a PD-1 intracellular domain or a fragment thereof.
- the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human MyD88 intracellular domain or a fragment thereof and a human PD-1 intracellular domain or a fragment thereof.
- the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to any one selected from SEQ ID NOs:287, 277, 288, 289, 273, 290, 291, 292, 293, 294, 295, 296, 297, 298, 299, 313, 314, 315, 316, and a combination thereof.
- the switch polypeptide comprises any one sequence selected from the group consisting of SEQ ID NOs:287, 277, 288, 289, 273, 290, 291, 292, 293, 294, 295, 296, 297, 298, 299, 313, 314, 315, 316, and a combination thereof.
- the sequence of the switch intracellular domain is a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to any one selected from SEQ ID NOs:287, 277, 288, 289, 273, 290, 291, 292, 293, 294, 295, 296, 297, 298, 299, 313, 314, 315, 316, and a combination thereof.
- the sequence of the switch intracellular domain is any one sequence selected from the group consisting of SEQ ID NOs:287, 277, 288, 289, 273, 290, 291, 292, 293, 294, 295, 296, 297, 298, 299, 313, 314, 315, 316, and a combination thereof.
- the costimulatory polypeptide is IL-15Ra.
- the sequence of the switch intracellular domain is from the intracellular domain of IL-15Ra.
- the sequence of the switch intracellular domain comprises a sequence having at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to SEQ ID NO:372.
- the sequence of the switch intracellular domain comprises a sequence or portion thereof of SEQ ID NO:372.
- the sequence of the switch intracellular domain comprises a sequence having at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to SEQ ID NO:383.
- the sequence of the switch intracellular domain comprises a sequence or portion thereof of SEQ ID NO:383.
- the switch polypeptide comprises an intracellular domain sequence having an addition of amino acid residue(s) to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein.
- the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C- terminal ends of the sequence of an IL-15Ra intracellular domain as described herein.
- the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N- terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein.
- the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:372 or SEQ ID NO:383.
- the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N- terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:372 or SEQ ID NO:383.
- the switch polypeptide comprises an intracellular domain sequence having a deletion of amino acid residue(s) from the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein.
- the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids deleted from the N-terminal or C-terminal end of the sequence of an IL-15Ra intracellular domain as described herein.
- the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids deleted from the N-terminal or C-terminal end of the sequence of an IL-15Ra intracellular domain as described herein.
- the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of an IL- 15Ra intracellular domain as described herein.
- the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein.
- the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:372. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:372.
- the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:372. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:372.
- the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:383.
- the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids deleted from the N-terminal or C- terminal end of the sequence of SEQ ID NO:383.
- the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:383.
- the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids independently deleted from both N-terminal and C- terminal ends of the sequence of SEQ ID NO:383.
- the switch polypeptide and/or dominant negative polypeptide further comprises a transmembrane domain.
- the TGFBr2 extracellular domain of the switch polypeptide is operably linked to the switch intracellular domain via a switch transmembrane domain.
- the transmembrane domain is derived from a TGFBr2 transmembrane domain.
- the transmembrane domain comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:272.
- the transmembrane domain comprises the sequence of SEQ ID NO:272.
- the sequence of the transmembrane domain is a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:272.
- the sequence of the transmembrane domain is the sequence of SEQ ID NO:272.
- the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having an addition of amino acid residue(s) to the N- terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of a TGFBr2 transmembrane domain as described herein.
- the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of a TGFBr2 transmembrane domain as described herein.
- the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of a TGFBr2 transmembrane domain as described herein.
- the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:272.
- the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C- terminal ends of the sequence of SEQ ID NO:272.
- the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having a deletion of amino acid residue(s) from the N- terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of a TGFBr2 transmembrane domain as described herein.
- the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, or 18 amino acids deleted from the N-terminal or C-terminal end of the sequence of a TGFBr2 transmembrane domain as described herein.
- the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, or 18 amino acids deleted from the N-terminal or C-terminal end of the sequence of a TGFBr2 transmembrane domain as described herein. In some embodiments, the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, or 18 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:272.
- the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, or 18 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:272. In some embodiments, the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, or 18 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of a TGFBr2 transmembrane domain as described herein.
- the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, or 18 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of a TGFBr2 transmembrane domain as described herein. In some embodiments, the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, or 18 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:272.
- the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, or 18 amino acids independently deleted from both N- terminal and C-terminal ends of the sequence of SEQ ID NO:272.
- the switch transmembrane domain is derived from a transmembrane domain of the costimulatory polypeptide.
- the switch transmembrane domain is derived from a transmembrane domain of CD28.
- the switch transmembrane domain is derived from a transmembrane domain of 4-1BB.
- the switch transmembrane domain comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence to SEQ ID NO:275 or SEQ ID NO:279. In some embodiments, the switch transmembrane domain comprises the sequence of SEQ ID NO:275 or SEQ ID NO:279.
- the sequence of the switch transmembrane domain is a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence to SEQ ID NO:275 or SEQ ID NO:279. In some embodiments, the sequence of the switch transmembrane domain is the sequence of SEQ ID NO:275 or SEQ ID NO:279.
- the switch polypeptide comprises a transmembrane domain sequence having an addition of amino acid residue(s) to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of a CD28 transmembrane domain or a 4-1BB transmembrane domain as described herein.
- the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of a CD28 transmembrane domain or a 4-1BB transmembrane domain as described herein.
- the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of a CD28 transmembrane domain or a 4-1BB transmembrane domain as described herein.
- the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:275 or SEQ ID NO:279.
- the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:275 or SEQ ID NO:279.
- the switch polypeptide comprises a transmembrane domain sequence having a deletion of amino acid residue(s) from the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of a CD28 transmembrane domain or a 4-1BB transmembrane domain as described herein.
- the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 amino acids deleted from the N-terminal or C-terminal end of the sequence of a CD28 transmembrane domain or a 4-1BB transmembrane domain as described herein.
- the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 amino acids deleted from the N-terminal or C-terminal end of the sequence of a CD28 transmembrane domain or a 4- 1BB transmembrane domain as described herein.
- the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:275 or SEQ ID NO:279.
- the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:275 or SEQ ID NO:279.
- the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 amino acids independently deleted from both N- terminal and C-terminal ends of the sequence of a CD28 transmembrane domain or a 4-1BB transmembrane domain as described herein.
- the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of a CD28 transmembrane domain or a 4- 1BB transmembrane domain as described herein.
- the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:275 or SEQ ID NO:279.
- the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:275 or SEQ ID NO:279.
- the switch transmembrane domain is derived from a transmembrane domain of IL-15Ra.
- the switch transmembrane domain comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence to SEQ ID NO:300.
- the switch transmembrane domain comprises the sequence of SEQ ID NO:300.
- the sequence of the switch transmembrane domain is a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence to SEQ ID NO:300.
- the sequence of the switch transmembrane domain is the sequence of SEQ ID NO:300.
- the switch polypeptide comprises a transmembrane domain sequence having an addition of amino acid residue(s) to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra transmembrane domain as described herein.
- the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra transmembrane domain as described herein.
- the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra transmembrane domain as described herein.
- the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C- terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:300.
- the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N- terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:300.
- the switch polypeptide comprises a transmembrane domain sequence having a deletion of amino acid residue(s) from the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra transmembrane domain as described herein.
- the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 amino acids deleted from the N-terminal or C-terminal end of the sequence of an IL-15Ra transmembrane domain as described herein.
- the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 amino acids deleted from the N-terminal or C-terminal end of the sequence of an IL-15Ra transmembrane domain as described herein. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:300.
- the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:300. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of an IL-15Ra transmembrane domain as described herein.
- the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 amino acids independently deleted from both N- terminal and C-terminal ends of the sequence of an IL-15Ra transmembrane domain as described herein. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:300.
- the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:300.
- the switch transmembrane domain is derived from any one selected from the group consisting of an ICOS transmembrane domain or a fragment thereof, a PD-1 transmembrane domain or a fragment thereof, a CTLA4 transmembrane domain or a fragment thereof, a CD200R transmembrane domain or a fragment thereof, a BTLA transmembrane domain or a fragment thereof, a TIM-3 transmembrane domain or a fragment thereof, a TIGIT transmembrane domain or a fragment thereof, a CD28 transmembrane domain or a fragment thereof, a TGF ⁇ R2 transmembrane domain or a fragment thereof, a 4- IBB transmembrane domain or a fragment thereof, an IL-10RA transmembrane domain or a fragment thereof, an IL-7RA transmembrane domain or a fragment thereof, an IL-4RA transmembrane domain or a fragment thereof, a Fas
- an IL12R transmembrane domain or a fragment thereof is an IL-12 receptor subunit beta-1 transmembrane domain or a fragment thereof. In some embodiments, an IL12R transmembrane domain or a fragment thereof is an IL-12 receptor subunit beta-2 transmembrane domain or a fragment thereof.
- an IL18R transmembrane domain or a fragment thereof is an interleukin-18 receptor 1 transmembrane domain or a fragment thereof
- the switch transmembrane domain is derived from any one selected from the group consisting of a human ICOS transmembrane domain or a fragment thereof, a human PD-1 transmembrane domain or a fragment thereof, a human CTLA4 transmembrane domain or a fragment thereof, a human CD200R transmembrane domain or a fragment thereof, a human BTLA transmembrane domain or a fragment thereof, a human TIM-3 transmembrane domain or a fragment thereof, a human TIGIT transmembrane domain or a fragment thereof, a human CD28 transmembrane domain or a fragment thereof, a human TGF ⁇ R2 transmembrane domain or a fragment thereof, a human 4-IBB transmembrane domain or a fragment thereof, a human
- a human IL12R transmembrane domain or a fragment thereof is a human IL-12 receptor subunit beta-1 transmembrane domain or a fragment thereof. In some embodiments, a human IL12R transmembrane domain or a fragment thereof is a human IL-12 receptor subunit beta-2 transmembrane domain or a fragment thereof. In some embodiments, a human IL18R transmembrane domain or a fragment thereof is a human interleukin-18 receptor 1 trans-membrane domain or a fragment thereof.
- the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to any one selected from SEQ ID NOs:301, 279, 302, 303, 275, 304, 305, 306, 307, 272, 308, 309, 310, 311, 312, 317, 318, 319, 320, and a combination thereof.
- the switch polypeptide comprises any one sequence selected from the group consisting of SEQ ID NOs:301, 279, 302, 303, 275, 304, 305, 306, 307, 272, 308, 309, 310, 311, 312, 317, 318, 319, 320, and a combination thereof.
- the sequence of the switch intracellular domain is a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to any one selected from SEQ ID NOs:301, 279, 302, 303, 275, 304, 305, 306, 307, 272, 308, 309, 310, 311, 312, 317, 318, 319, 320, and a combination thereof.
- the sequence of the switch intracellular domain is any one sequence selected from the group consisting of SEQ ID NOs:301, 279, 302, 303, 275, 304, 305, 306, 307, 272, 308, 309, 310, 311, 312, 317, 318, 319, 320, and a combination thereof.
- the additional intracellular domain is operably linked to the C-terminus of the switch intracellular domain. In some embodiments, the additional intracellular domain is operably linked to the N-terminus of the switch intracellular domain. In some embodiments, the additional intracellular domain is operably linked to the C-terminus or the N-terminus of the switch intracellular domain. In some embodiments, the additional intracellular domain is operably linked to the C-terminus or the N-terminus of the switch intracellular domain via a linker. In some embodiments, the switch polypeptide may comprise two or more additional intracellular domains. In some embodiments, the intracellular domains are operably linked to the C-terminus and the N-terminus of the switch intracellular domain, independently.
- the additional intracellular domains are operably linked to the C-terminus, to the N-terminus, or independently to the C- terminus and the N-terminus of the switch intracellular domain via a linker.
- the switch polypeptide further comprises one or more additional intracellular domains.
- one or more additional intracellular domains are the same intracellular domain.
- one or more additional intracellular domains are different intracellular domains.
- the additional intracellular domain is derived from an intracellular domain of IL-15Ra.
- the additional intracellular domain comprises an intracellular domain of IL-15Ra or a fragment thereof.
- IL-15Ra or a fragment thereof comprises 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100, 101, 102, 103, 104, 105, 106, 107, 108, 109, 110, 111, 112, 113, 114
- IL-15Ra or a fragment thereof comprises a sequence having at least about 50%, 55%, 60%, 6%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99% or more sequence identity to a sequence encoding IL-15Ra.
- IL-15Ra or a fragment thereof comprises a sequence encoding IL-15R ⁇ having a truncation of at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100, or more amino acids at the N- or C-terminus or at both
- the additional intracellular domain comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:372 or SEQ ID NO:383. In some embodiments, the additional intracellular domain comprises the sequence of SEQ ID NO:372 or SEQ ID NO:383.
- the sequence of the additional intracellular domain is a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:372 or SEQ ID NO:383.
- the sequence of the additional intracellular domain is the sequence of SEQ ID NO:372 or SEQ ID NO:383.
- the additional intracellular domain comprises a sequence having an addition of amino acid residue(s) to the N-terminal end, C-terminal end, or both N- terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein.
- the additional intracellular domain comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein.
- the additional intracellular domain comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein.
- the additional intracellular domain comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:372 or SEQ ID NO:383.
- the additional intracellular domain comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C- terminal ends of the sequence of SEQ ID NO:372 or SEQ ID NO:383.
- the additional intracellular domain comprises a sequence having a deletion of amino acid residue(s) from the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein.
- the additional intracellular domain comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids deleted from the N-terminal or C- terminal end of the sequence of an IL-15Ra intracellular domain as described herein.
- the additional intracellular domain comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids deleted from the N-terminal or C-terminal end of the sequence of an IL-15Ra intracellular domain as described herein.
- the additional intracellular domain comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein.
- the additional intracellular domain comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids independently deleted from both N-terminal and C- terminal ends of the sequence of an IL-15Ra intracellular domain as described herein.
- the additional intracellular domain comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:372. In some embodiments, the additional intracellular domain comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:372.
- the additional intracellular domain comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids independently deleted from both N-terminal and C- terminal ends of the sequence of SEQ ID NO:372. In some embodiments, the additional intracellular domain comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:372.
- the additional intracellular domain comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:383.
- the additional intracellular domain comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:383.
- the additional intracellular domain comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:383.
- the additional intracellular domain comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:383.
- the switch polypeptide comprises a transmembrane domain derived from a TGFBr2 transmembrane domain and an intracellular signaling domain of 4- 1BB.
- the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:272 and a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:277.
- the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:272 operatively linked a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:277.
- the switch polypeptide comprises the sequence of SEQ ID NO:272 and the sequence of SEQ ID NO:277.
- the switch polypeptide comprises the sequence of SEQ ID NO:272 operatively linked to the sequence of SEQ ID NO:277. [0365] In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:285. In some embodiments, the switch polypeptide comprises the sequence of SEQ ID NO:285.
- the sequence of the switch polypeptide is a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:285.
- the sequence of the switch polypeptide is the sequence of SEQ ID NO:285.
- the switch polypeptide comprises a transmembrane domain derived from a 4-1BB transmembrane domain and an intracellular signaling domain of 4- 1BB.
- the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:279 and a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:277.
- the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:279 operatively linked a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:277.
- the switch polypeptide comprises the sequence of SEQ ID NO:279 and the sequence of SEQ ID NO:277.
- the switch polypeptide comprises the sequence of SEQ ID NO:279 operatively linked to the sequence of SEQ ID NO:277. [0368] In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:286. In some embodiments, the switch polypeptide comprises the sequence of SEQ ID NO:286.
- the sequence of the switch polypeptide is a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:286.
- the sequence of the switch polypeptide is the sequence of SEQ ID NO:286.
- the switch polypeptide comprises a transmembrane domain derived from a TGFBr2 transmembrane domain and an intracellular signaling domain of CD28.
- the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:272 and a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:273.
- the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:272 operatively linked a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:273.
- the switch polypeptide comprises the sequence of SEQ ID NO:272 and the sequence of SEQ ID NO:273.
- the switch polypeptide comprises the sequence of SEQ ID NO:272 operatively linked to the sequence of SEQ ID NO:273. [0371] In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:283. In some embodiments, the switch polypeptide comprises the sequence of SEQ ID NO:283.
- the sequence of the switch polypeptide is a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:283.
- the sequence of the switch polypeptide is the sequence of SEQ ID NO:283.
- the switch polypeptide comprises a transmembrane domain derived from a CD28 transmembrane domain and an intracellular signaling domain of CD28.
- the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:275 and a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:273.
- the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:275 operatively linked a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:273.
- the switch polypeptide comprises the sequence of SEQ ID NO:275 and the sequence of SEQ ID NO:273.
- the switch polypeptide comprises the sequence of SEQ ID NO:275 operatively linked to the sequence of SEQ ID NO:273. [0374] In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:284. In some embodiments, the switch polypeptide comprises the sequence of SEQ ID NO:284.
- sequence of the switch polypeptide is a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:284. In some embodiments, the sequence of the switch polypeptide is the sequence of SEQ ID NO:284.
- T-cell Receptor (TCR) Fusion Proteins TFPs
- TFP T-cell Receptor
- TFPs T-cell Receptor (TCR) Fusion Proteins
- the present disclosure encompasses recombinant nucleic acid constructs encoding TFPs, wherein the TFP comprises a binding domain, e.g., an antibody or antibody fragment, a ligand, or a ligand binding protein, wherein the sequence of the binding domain is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof.
- a binding domain e.g., an antibody or antibody fragment, a ligand, or a ligand binding protein
- the antibody or antibody fragment can comprise an antigen binding domain selected from a group consisting of an anti-CD19 binding domain, an anti-B-cell maturation antigen (BCMA) binding domain, an anti-mesothelin (MSLN) binding domain, an anti-CD20 binding domain, an anti-CD70 binding domain, an anti-CD79b binding domain, , an anti- PMSA binding domain, an anti-MUC16 binding domain, an anti-CD22 binding domain, an anti-PD-L1 binding domain, an anti BAFF receptor binding domain, an anti-Nectin-4 binding domain, an anti-TROP-2 binding domain, an anti-GPC3 binding domain, and anti-ROR-1 binding domain.
- an antigen binding domain selected from a group consisting of an anti-CD19 binding domain, an anti-B-cell maturation antigen (BCMA) binding domain, an anti-mesothelin (MSLN) binding domain, an anti-CD20 binding domain, an anti-CD70 binding domain, an anti-CD79b binding domain, , an
- the present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to a tumor associated antigen (a TAA) wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof.
- TAA tumor associated antigen
- the present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to CD19, e.g., human CD19, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof.
- the present disclosure encompasses recombinant nucleic acid, e.g., DNA, constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to mesothelin, e.g., human mesothelin, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof.
- TFP comprises an antibody fragment that binds specifically to mesothelin, e.g., human mesothelin, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof.
- the present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to MUC16, e.g., human MUC16, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof.
- the present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to CD20, e.g., human CD20, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof.
- the present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to CD70, e.g., human CD70, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof.
- the present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to CD79B, e.g., human CD79B, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof.
- the present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to HER2, e.g., human HER2, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof.
- the present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to PSMA, e.g., human PSMA, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof.
- the present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to BCMA, e.g., human BCMA, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof.
- the present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to ROR1, e.g., human ROR1, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof.
- the present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to CD22, e.g., human CD22, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof.
- the present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to GPC3, e.g., human GPC3, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof.
- the present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to Nectin-4, e.g., human Nectin-4, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof.
- the present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to Trop-2, e.g., human Trop-2, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof.
- the TFPs provided herein are able to associate with one or more endogenous (or alternatively, one or more exogenous, or a combination of endogenous and exogenous) TCR subunits in order to form a functional TCR complex.
- the TFP of the present disclosure comprises a target-specific binding element otherwise referred to as an antigen binding domain.
- the choice of moiety depends upon the type and number of target antigen that define the surface of a target cell.
- the antigen binding domain may be chosen to recognize a target antigen that acts as a cell surface marker on target cells associated with a particular disease state.
- examples of cell surface markers that may act as target antigens for the antigen binding domain in a TFP of the present disclosure include those associated with viral, bacterial and parasitic infections; autoimmune diseases; and cancerous diseases (e.g., malignant diseases).
- the TFP-mediated T cell response can be directed to an antigen of interest by way of engineering an antigen-binding domain into the TFP that specifically binds a desired antigen.
- the antigen binding domain can be any domain that binds to the antigen including but not limited to a monoclonal antibody, a polyclonal antibody, a recombinant antibody, a human antibody, a humanized antibody, and a functional fragment thereof, including but not limited to a single-domain antibody such as a heavy chain variable domain (V H ), a light chain variable domain (VL) and a variable domain (VHH) of a camelid derived nanobody, and to an alternative scaffold known in the art to function as antigen binding domain, such as a recombinant fibronectin domain, anticalin, DARPIN and the like.
- V H heavy chain variable domain
- VL light chain variable domain
- VHH variable domain of a camelid derived nanobody
- the antigen binding domain for the TFP.
- the antigen binding domain of the TFP for use in humans, it may be beneficial for the antigen binding domain of the TFP to comprise human or humanized residues for the antigen binding domain of an antibody or antibody fragment.
- the antigen-binding domain comprises a humanized or human antibody or an antibody fragment, or a murine antibody or antibody fragment.
- the murine, humanized or human anti-TAA binding domain comprises one or more (e.g., all three) light chain complementary determining region 1 (LC CDR1), light chain complementary determining region 2 (LC CDR2), and light chain complementary determining region 3 (LC CDR3) of a murine, humanized or human anti-TAA binding domain described herein, and/or one or more (e.g., all three) heavy chain complementary determining region 1 (HC CDR1), heavy chain complementary determining region 2 (HC CDR2), and heavy chain complementary determining region 3 (HC CDR3) of a murine, humanized or human anti-CD19 binding domain described herein, e.g., a murine, humanized or human anti-TAA binding domain comprising one or more, e.g., all three, LC CDRs and one or more, e.g., all three, HC CDRs.
- LC CDR1 light chain complementary determining region 1
- HC CDR2 light chain complementary determining region 2
- the murine, humanized or human anti-CD19 binding domain comprises one or more (e.g., all three) heavy chain complementary determining region 1 (HC CDR1), heavy chain complementary determining region 2 (HC CDR2), and heavy chain complementary determining region 3 (HC CDR3) of a murine, humanized or human anti-TAA binding domain described herein, e.g., the murine, humanized or human anti-TAA binding domain has two variable heavy chain regions, each comprising a HC CDR1, a HC CDR2 and a HC CDR3 described herein.
- HC CDR1 heavy chain complementary determining region 1
- HC CDR2 heavy chain complementary determining region 2
- HC CDR3 heavy chain complementary determining region 3
- the murine, humanized or human anti-TAA binding domain comprises a humanized or human light chain variable region described herein and/or a murine, humanized or human heavy chain variable region described herein.
- the murine, humanized or human anti-TAA binding domain comprises a murine, humanized or human heavy chain variable region described herein, e.g., at least two murine, humanized or human heavy chain variable regions described herein.
- the anti-TAA binding domain is a scFv comprising a light chain and a heavy chain of an amino acid sequence provided herein.
- the anti-TAA binding domain (e.g., a scFv) comprises: a light chain variable region comprising an amino acid sequence having at least one, two or three modifications (e.g., substitutions) but not more than 30, 20 or 10 modifications (e.g., substitutions) of an amino acid sequence of a light chain variable region provided herein, or a sequence with 95-99% identity with an amino acid sequence provided herein; and/or a heavy chain variable region comprising an amino acid sequence having at least one, two or three modifications (e.g., substitutions) but not more than 30, 20 or 10 modifications (e.g., substitutions) of an amino acid sequence of a heavy chain variable region provided herein, or a sequence with 95-99% identity to an amino acid sequence provided herein.
- a light chain variable region comprising an amino acid sequence having at least one, two or three modifications (e.g., substitutions) but not more than 30, 20 or 10 modifications (e.g., substitutions) of an amino acid sequence of a heavy chain variable region provided here
- the murine, humanized or human anti-TAA binding domain is a scFv, and a light chain variable region comprising an amino acid sequence described herein, is attached to a heavy chain variable region comprising an amino acid sequence described herein, via a linker, e.g., a linker described herein.
- the murine, humanized, or human anti-TAA binding domain includes a (Gly 4 -Ser) n linker, wherein n is 1, 2, 3, 4, 5, or 6, preferably 3 or 4 (SEQ ID NO: 400).
- the light chain variable region and heavy chain variable region of a scFv can be, e.g., in any of the following orientations: light chain variable region- linker-heavy chain variable region or heavy chain variable region-linker-light chain variable region.
- the linker sequence comprises a long linker (LL) sequence.
- the linker sequence comprises a short linker (SL) sequence.
- the antigen binding domain is an antibody or a fragment thereof.
- the antigen binding domain is a camelid antibody or a binding fragment thereof. In some embodiments, the antigen binding domain is a murine antibody or a binding fragment thereof. In some embodiments, the antigen binding domain is a human or humanized antibody or a binding fragment thereof. In some embodiments, the antigen binding domain is a single-chain variable fragment (scFv) or a single domain antibody (sdAb) domain. In some embodiments, the sdAb is a V HH .
- the antigen binding domain is selected from the group consisting of an anti-CD19 binding domain, an anti-B-cell maturation antigen (BCMA) binding domain, an anti-mesothelin (MSLN) binding domain, an anti-CD20 binding domain, an anti-CD70 binding domain, anti-MUC16 binding domain, an anti-Nectin-4 binding domain, an anti-GPC3 binding domain, and an anti-TROP-2 binding domain.
- BCMA anti-B-cell maturation antigen
- MSLN anti-mesothelin
- the antigen-binding domain comprises an anti-CD19 humanized or human antibody or an antibody fragment, or a murine antibody or antibody fragment having a light chain CDR1 of SEQ ID NO:73, a CDR2 of SEQ ID NO:75, and a CDR3 of SEQ ID NO:77 and a heavy chain CDR1 of SEQ ID NO:79, a CDR2 of SEQ ID NO:81, and a CDR3 of SEQ ID NO:83.
- the anti-CD19 antibody is a murine scFv.
- the anti-CD-19 antibody comprises a VL of SEQ ID NO:85 and a VH of SEQ ID NO:87.
- the antigen-binding domain comprises an anti-mesothelin humanized or human single domain antibody or an antibody fragment having a CDR1 of SEQ ID NO:60, a CDR2 of SEQ ID NO:61, and a CDR3 of SEQ ID NO:62 or a CDR1 of SEQ ID NO:63, a CDR2 of SEQ ID NO:64, and a CDR3 of SEQ ID NO:65 or a CDR1 of SEQ ID NO:66, a CDR2 of SEQ ID NO:67, and a CDR3 of SEQ ID NO:68.
- the anti-mesothelin antibody has a variable domain of SEQ ID NO:69, SEQ ID NO:70, or SEQ ID NO:71.
- the antigen-binding domain comprises an anti-CD70 humanized or human single domain antibody or an antibody fragment having a CDR1 of SEQ ID NO:88, a CDR2 of SEQ ID NO:89, and a CDR3 of SEQ ID NO:90, or a CDR1 of SEQ ID NO:92, a CDR2 of SEQ ID NO:93, and a CDR3 of SEQ ID NO:94, or a CDR1 of SEQ ID NO:96, a CDR2 of SEQ ID NO:97, and a CDR3 of SEQ ID NO:98, or a CDR1 of SEQ ID NO:100, a CDR2 of SEQ ID NO:101, and a CDR3 of SEQ ID NO:102, or a CDR1 of SEQ ID NO:104, a
- the antigen-binding domain comprises an anti-CD70 single chain Fv (scFv) or an antibody fragment thereof.
- the anti-CD70 scFv or antibody fragment thereof can comprise a heavy chain complementary determining region 1 (CDRH1) having a sequence of SEQ ID NO: 361, a CDRH2 having a sequence of SEQ ID NO: 362, and a CDRH3 having a sequence of SEQ ID NOs: 363.
- the anti-CD70 scFv or antibody fragment thereof can comprise a light chain complementary determining region 1 (CDRL1) having a sequence of SEQ ID NO: 365 , a CDRL2 having a sequence of SEQ ID NO: 366, and a CDRL3 having a sequence of SEQ ID NO: 367.
- the anti-CD70 scFv or antibody fragment thereof can comprise a heavy chain variable (VH) domain having at least 70%, 75%, 80%, 85%, 90%, 95%, or 100% sequence identity to SEQ ID NO: 364.
- the anti-CD70 scFv or antibody fragment thereof can comprise a light chain variable (VL) domain having at least 70%, 75%, 80%, 85%, 90%, 95%, or 100% sequence identity to SEQ ID NO: 368.
- VL light chain variable
- a non-human antibody is humanized, where specific sequences or regions of the antibody are modified to increase similarity to an antibody naturally produced in a human or fragment thereof.
- the antigen binding domain is humanized.
- a humanized antibody can be produced using a variety of techniques known in the art, including but not limited to, CDR-grafting (see, e.g., European Patent No. EP 239,400; International Publication No. WO 91/09967; and U.S. Pat.
- a humanized antibody or antibody fragment has one or more amino acid residues remaining in it from a source which is nonhuman. These nonhuman amino acid residues are often referred to as “import” residues, which are typically taken from an “import” variable domain.
- humanized antibodies or antibody fragments comprise one or more CDRs from nonhuman immunoglobulin molecules and framework regions wherein the amino acid residues comprising the framework are derived completely or mostly from human germline.
- Multiple techniques for humanization of antibodies or antibody fragments are well- known in the art and can essentially be performed following the method of Winter and co- workers (Jones et al., Nature, 321:522-525 (1986); Riechmann et al., Nature, 332:323-327 (1988); Verhoeyen et al., Science, 239:1534-1536 (1988)), by substituting rodent CDRs or CDR sequences for the corresponding sequences of a human antibody, i.e., CDR-grafting (EP 239,400; PCT Publication No.
- WO 91/09967 and U.S. Pat. Nos.4,816,567; 6,331,415; 5,225,539; 5,530,101; 5,585,089; 6,548,640, the contents of which are incorporated herein by reference in their entirety).
- Humanized antibodies and antibody fragments substantially less than an intact human variable domain has been substituted by the corresponding sequence from a nonhuman species.
- Humanized antibodies are often human antibodies in which some CDR residues and possibly some framework (FR) residues are substituted by residues from analogous sites in rodent antibodies.
- the sequence of the variable domain of a rodent antibody is screened against the entire library of known human variable-domain sequences.
- the human sequence which is closest to that of the rodent is then accepted as the human framework (FR) for the humanized antibody (Sims et al., J. Immunol., 151:2296 (1993); Chothia et al., J. Mol. Biol., 196:901 (1987), the contents of which are incorporated herein by reference herein in their entirety).
- Another method uses a particular framework derived from the consensus sequence of all human antibodies of a particular subgroup of light or heavy chains.
- the same framework may be used for several different humanized antibodies (see, e.g., Nicholson et al., Mol. Immun.34 (16-17): 1157-1165 (1997); Carter et al., Proc. Natl. Acad. Sci. USA, 89:4285 (1992); Presta et al., J. Immunol., 151:2623 (1993), the contents of which are incorporated herein by reference herein in their entirety).
- the framework region e.g., all four framework regions, of the heavy chain variable region are derived from a VH4-4-59 germline sequence.
- the framework region can comprise, one, two, three, four or five modifications, e.g., substitutions, e.g., from the amino acid at the corresponding murine sequence.
- the framework region e.g., all four framework regions of the light chain variable region are derived from a VK3-1.25 germline sequence.
- the framework region can comprise, one, two, three, four or five modifications, e.g., substitutions, e.g., from the amino acid at the corresponding murine sequence.
- the portion of a TFP composition of the present disclosure that comprises an antibody fragment is humanized with retention of high affinity for the target antigen and other favorable biological properties.
- humanized antibodies and antibody fragments are prepared by a process of analysis of the parental sequences and various conceptual humanized products using three- dimensional models of the parental and humanized sequences.
- Three-dimensional immunoglobulin models are commonly available and are familiar to those skilled in the art.
- Computer programs are available which illustrate and display probable three-dimensional conformational structures of selected candidate immunoglobulin sequences. Inspection of these displays permits analysis of the likely role of the residues in the functioning of the candidate immunoglobulin sequence, e.g., the analysis of residues that influence the ability of the candidate immunoglobulin to bind the target antigen.
- a humanized antibody or antibody fragment may retain a similar antigenic specificity as the original antibody, e.g., in the present disclosure, the ability to bind human a tumor associated antigen (TAA).
- TAA tumor associated antigen
- a humanized antibody or antibody fragment may have improved affinity and/or specificity of binding to, e.g., human CD19, human BCMA, or another tumor associated antigen.
- the binding domain is characterized by particular functional features or properties of an antibody or antibody fragment.
- the portion of a TFP composition of the present disclosure that comprises an antigen binding domain specifically binds human CD19.
- the antigen binding domain has the same or a similar binding specificity to human CD19 as the FMC63 scFv described in Nicholson et al., Mol. Immun.34 (16-17): 1157-1165 (1997).
- the present disclosure relates to an antigen binding domain comprising an antibody or antibody fragment, wherein the antibody binding domain specifically binds to a CD19 or BCMA protein or fragment thereof, wherein the antibody or antibody fragment comprises a variable light chain and/or a variable heavy chain that includes an amino acid sequence provided herein.
- the scFv is contiguous with and in the same reading frame as a leader sequence.
- the anti-tumor-associated antigen binding domain is a fragment, e.g., a single chain variable fragment (scFv).
- the anti-TAA binding domain is a Fv, a Fab, a (Fab’)2, or a bi-functional (e.g., bi-specific) hybrid antibody (e.g., Lanzavecchia et al., Eur. J. Immunol.17, 105 (1987)).
- the antibodies and fragments thereof of the present disclosure binds a CD19 protein with wild-type or enhanced affinity.
- the anti-TAA binding domain comprises a single domain antibody (sdAb or VHH).
- a target antigen e.g., CD19, BCMA, MSLN, CD70, or any target antigen described elsewhere herein for targets of fusion moiety binding domains
- the method comprising providing by way of addition, deletion, substitution or insertion of one or more amino acids in the amino acid sequence of a V H domain set out herein a V H domain which is an amino acid sequence variant of the VH domain, optionally combining the VH domain thus provided with one or more VL domains, and testing the VH domain or VH/VL combination or combinations to identify a specific binding member or an antibody antigen binding domain specific for a target antigen of interest (e.g., MSLN, CD79B, etc.) and optionally with one or more desired properties.
- a target antigen of interest e.g., MSLN, CD79B, etc.
- V H domains and scFvs can be prepared according to method known in the art (see, for example, Bird et al., (1988) Science 242:423-426 and Huston et al., (1988) Proc. Natl. Acad. Sci. USA 85:5879-5883).
- scFv molecules can be produced by linking VH and VL regions together using flexible polypeptide linkers.
- the scFv molecules comprise a linker (e.g., a Ser-Gly linker) with an optimized length and/or amino acid composition. The linker length can greatly affect how the variable regions of a scFv fold and interact.
- the linker sequence comprises a linker sequence.
- linker orientation and size see, e.g., Hollinger et al., 1993 Proc Natl Acad. Sci.
- An scFv can comprise a linker of about 10, 11, 12, 13, 14, 15 or greater than 15 residues between its VL and VH regions.
- the linker sequence may comprise any naturally occurring amino acid.
- the linker sequence comprises amino acids glycine and serine.
- the linker sequence comprises sets of glycine and serine repeats such as (Gly4Ser)n, where n is a positive integer equal to or greater than 1 (SEQ ID NO: 402).
- the linker can be (Gly4Ser)4 (SEQ ID NO: 394) or (Gly 4 Ser) 3 (SEQ ID NO: 395). Variation in the linker length may retain or enhance activity, giving rise to superior efficacy in activity studies.
- the linker sequence comprises GGSGGSGGSGGS (SEQ ID NO: 369).
- Stability and Mutations [0397] The stability of a tumor associated antigen binding domain, e.g., scFv molecules (e.g., soluble scFv) can be evaluated in reference to the biophysical properties (e.g., thermal stability) of a conventional control scFv molecule or a full-length antibody.
- the humanized or human scFv has a thermal stability that is greater than about 0.1, about 0.25, about 0.5, about 0.75, about 1, about 1.25, about 1.5, about 1.75, about 2, about 2.5, about 3, about 3.5, about 4, about 4.5, about 5, about 5.5, about 6, about 6.5, about 7, about 7.5, about 8, about 8.5, about 9, about 9.5, about 10 degrees, about 11 degrees, about 12 degrees, about 13 degrees, about 14 degrees, or about 15 degrees Celsius than a parent scFv in the described assays.
- the improved thermal stability of the anti-TAA binding domain e.g., scFv is subsequently conferred to the entire TAA-TFP construct, leading to improved therapeutic properties of the anti-TAA TFP construct.
- the thermal stability of the binding domain e.g., scFv or sdAb, can be improved by at least about 2 °C or 3 °C as compared to a conventional antibody.
- the binding domain has a 1 °C improved thermal stability as compared to a conventional antibody.
- the binding domain has a 2 °C improved thermal stability as compared to a conventional antibody.
- the scFv has a 4 °C, 5 °C, 6 °C, 7 °C, 8 °C, 9 °C, 10 °C, 11 °C, 12 °C, 13 °C, 14 °C, or 15 °C improved thermal stability as compared to a conventional antibody. Comparisons can be made, for example, between the scFv molecules as described herein and scFv molecules or Fab fragments of an antibody from which the scFv VH and VL were derived. Thermal stability can be measured using methods known in the art. For example, in one embodiment, TM can be measured. Methods for measuring T M and other methods of determining protein stability are described in more detail below.
- the binding domain e.g., a scFv or sdAb, comprises at least one mutation arising from the humanization process such that the mutated scFv confers improved stability to the anti-TAA TFP construct.
- the anti-TAA binding domain e.g., scFv or sdAb
- the antigen binding domain of the TFP comprises an amino acid sequence that is homologous to an antigen binding domain amino acid sequence described herein, and the antigen binding domain retains the desired functional properties of the anti- tumor-associated antigen antibody fragments described herein.
- the TFP composition of the present disclosure comprises an antibody fragment. In a further aspect, that antibody fragment comprises a scFv.
- the antigen binding domain of the TFP is engineered by modifying one or more amino acids within one or both variable regions (e.g., VH and/or VL), for example within one or more CDR regions and/or within one or more framework regions.
- the TFP composition of the present disclosure comprises an antibody fragment.
- that antibody fragment comprises a scFv.
- the antibody or antibody fragment of the present disclosure may further be modified such that they vary in amino acid sequence (e.g., from wild-type), but not in desired activity.
- nucleotide substitutions leading to amino acid substitutions at “non-essential” amino acid residues may be made to the protein.
- a nonessential amino acid residue in a molecule may be replaced with another amino acid residue from the same side chain family.
- a string of amino acids can be replaced with a structurally similar string that differs in order and/or composition of side chain family members, e.g., a conservative substitution, in which an amino acid residue is replaced with an amino acid residue having a similar side chain, may be made.
- Families of amino acid residues having similar side chains have been defined in the art, including basic side chains (e.g., lysine, arginine, histidine), acidic side chains (e.g., aspartic acid, glutamic acid), uncharged polar side chains (e.g., glycine, asparagine, glutamine, serine, threonine, tyrosine, cysteine), nonpolar side chains (e.g., alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine, tryptophan), beta-branched side chains (e.g., threonine, valine, isoleucine) and aromatic side chains (e.g., tyrosine, phenylalanine, tryptophan, histidine).
- basic side chains e.g., lysine, arginine, histidine
- acidic side chains e.g., aspartic acid
- Percent identity in the context of two or more nucleic acids or polypeptide sequences refers to two or more sequences that are the same. Two sequences are “substantially identical” if two sequences have a specified percentage of amino acid residues or nucleotides that are the same (e.g., 60% identity, optionally 70%, 71% , 72%, 73%, 74%, 75%, 76%, 77%, 78%, 79%, 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity over a specified region, or, when not specified, over the entire sequence), when compared and aligned for maximum correspondence over a comparison window, or designated region as measured using one of the following sequence comparison algorithms or by manual alignment and visual inspection.
- the identity exists over a region that is at least about 50 nucleotides (or 10 amino acids) in length, or more preferably over a region that is 100 to 500 or 1000 or more nucleotides (or 20, 50, 200 or more amino acids) in length.
- sequence comparison algorithm typically one sequence acts as a reference sequence, to which test sequences are compared.
- test and reference sequences are entered into a computer, subsequence coordinates are designated, if necessary, and sequence algorithm program parameters are designated. Default program parameters can be used, or alternative parameters can be designated.
- the sequence comparison algorithm then calculates the percent sequence identities for the test sequences relative to the reference sequence, based on the program parameters.
- Optimal alignment of sequences for comparison can be conducted, e.g., by the local homology algorithm of Smith and Waterman, (1970) Adv. Appl. Math.2:482c, by the homology alignment algorithm of Needleman and Wunsch, (1970) J. Mol. Biol.48:443, by the search for similarity method of Pearson and Lipman, (1988) Proc. Natl. Acad. Sci.
- the present disclosure contemplates modifications of the starting antibody or fragment (e.g., scFv) amino acid sequence that generate functionally equivalent molecules.
- the VH or VL of a binding domain, e.g., scFv, comprised in the TFP can be modified to retain at least about 70%, 71%.72%.73%, 74%, 75%, 76%, 77%, 78%, 79%, 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% identity of the starting VH or VL framework region of the anti- CD19 binding domain, e.g., scFv.
- the present disclosure contemplates modifications of the entire TFP construct, e.g., modifications in one or more amino acid sequences of the various domains of the TFP construct in order to generate functionally equivalent molecules.
- the TFP construct can be modified to retain at least about 70%, 71%.72%.73%, 74%, 75%, 76%, 77%, 78%, 79%, 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% identity of the starting TFP construct.
- Extracellular Domain [0407] The extracellular domain may be derived either from a natural or from a recombinant source.
- the domain may be derived from any protein, but in particular a membrane-bound or transmembrane protein.
- the extracellular domain is capable of associating with the transmembrane domain.
- An extracellular domain of particular use in this present disclosure may include at least the extracellular region(s) of e.g., the alpha, beta or zeta chain of the T cell receptor, or CD3 epsilon, CD3 gamma, or CD3 delta, or in alternative embodiments, CD28, CD45, CD4, CD5, CD8, CD9, CD16, CD22, CD33, CD37, CD64, CD80, CD86, CD134, CD137, or CD154.
- the extracellular domain is a TCR extracellular domain.
- the TCR extracellular domain comprises an extracellular domain or portion thereof of a protein selected from the group consisting of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, a CD3 delta TCR subunit, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the TCR extracellular domain comprises an extracellular domain or portion thereof of a TCR alpha chain, a TCR beta chain, a TCR delta chain, or a TCR gamma chain.
- the TCR extracellular domain comprises the extracellular portion of a constant (an IgC) domain of a TCR alpha chain, a TCR beta chain, a TCR delta chain, or a TCR gamma chain.
- the extracellular domain comprises, or comprises at least 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92
- the extracellular domain comprises a sequence having at least about 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99% or more sequence identity to a sequence encoding the extracellular domain of a TCR alpha chain, a TCR beta chain, a TCR delta chain, or a TCR gamma chain.
- the extracellular domain comprises a sequence encoding the extracellular domain of a TCR alpha chain, a TCR beta chain, a TCR delta chain, or a TCR gamma chain having a truncation of at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25 or more amino acids at the N- or C-terminus or at both the N- and C-terminus.
- the extracellular domain comprises, or comprises at least 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100 or more consecutive amino acid residues of the extracellular portion of a constant (an IgC) domain of TCR alpha, a TCR beta, a TCR delta
- the extracellular domain comprises a sequence having at least about 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99% or more sequence identity to a sequence encoding the extracellular portion of a constant (an IgC) domain of TCR alpha, a TCR beta, a TCR delta, or a TCR gamma.
- a constant (an IgC) domain of TCR alpha, a TCR beta, a TCR delta, or a TCR gamma a sequence having at least about 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99% or more sequence identity to a sequence encoding the extracellular portion of a constant (an IgC) domain of TCR alpha, a TCR beta, a TCR delta, or a TCR gamma.
- the extracellular domain comprises a sequence encoding the extracellular portion of a constant (an IgC) domain of TCR alpha, TCR beta, TCR delta, or TCR gamma having a truncation of at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25 or more amino acids at the N- or C-terminus or at both the N- and C-terminus.
- a constant (an IgC) domain of TCR alpha, TCR beta, TCR delta, or TCR gamma having a truncation of at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25 or more amino acids at the N- or C-terminus or at both the N- and C-terminus.
- the extracellular domain comprises, or comprises at least 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100 or more consecutive amino acid residues of the extracellular domain of a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit
- the extracellular domain comprises a sequence having at least about 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99% or more sequence identity to a sequence encoding the extracellular domain of a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit.
- the extracellular domain comprises a sequence encoding the extracellular domain of a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit having a truncation of at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25 or more amino acids at the N- or C- terminus or at both the N- and C-terminus.
- the extracellular domain can be a TCR extracellular domain.
- the TCR extracellular domain can be derived from a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit or a CD3 delta TCR subunit.
- the extracellular domain can be a full-length TCR extracellular domain or fragment (e.g., functional fragment) thereof.
- the extracellular domain can comprise a variable domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain.
- the extracellular domain can comprise a variable domain and a constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain. In some cases, the extracellular domain may not comprise a variable domain. [0413]
- the extracellular domain can comprise a constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain.
- the extracellular domain can comprise a full-length constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain.
- the extracellular domain can comprise a fragment (e.g., functional fragment) of the full-length constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain.
- the extracellular domain can comprise at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of the constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain.
- the TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain described herein can be derived from various species.
- the TCR chain can be a murine or human TCR chain.
- the extracellular domain can comprise a constant domain of a murine TCR alpha chain, a murine TCR beta chain, a human TCR gamma chain or a human TCR delta chain.
- Transmembrane Domain [0415]
- a TFP sequence contains an extracellular domain and a transmembrane domain encoded by a single genomic sequence.
- a TFP can be designed to comprise a transmembrane domain that is heterologous to the extracellular domain of the TFP.
- a transmembrane domain can include one or more additional amino acids adjacent to the transmembrane region, e.g., one or more amino acid associated with the extracellular region of the protein from which the transmembrane was derived (e.g., at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, or more amino acids of the extracellular region) and/or one or more additional amino acids associated with the intracellular region of the protein from which the transmembrane protein is derived (e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, or more amino acids of the intracellular region).
- the transmembrane domain can include at least 30, 35, 40, 45, 50, 55, 60 or more amino acids of the extracellular region. In some cases, the transmembrane domain can include at least 30, 35, 40, 45, 50, 55, 60 or more amino acids of the intracellular region. In one aspect, the transmembrane domain is one that is associated with one of the other domains of the TFP is used. In some instances, the transmembrane domain can be selected or modified by amino acid substitution to avoid binding of such domains to the transmembrane domains of the same or different surface membrane proteins, e.g., to minimize interactions with other members of the receptor complex.
- the transmembrane domain is capable of homodimerization with another TFP on the TFP-T cell surface.
- the amino acid sequence of the transmembrane domain may be modified or substituted so as to minimize interactions with the binding domains of the native binding partner present in the same TFP.
- the transmembrane domain may be derived either from a natural or from a recombinant source. Where the source is natural, the domain may be derived from any membrane-bound or transmembrane protein.
- the transmembrane domain is capable of signaling to the intracellular domain(s) whenever the TFP has bound to a target.
- the TCR-integrating subunit comprises a transmembrane domain comprising a transmembrane domain of a protein selected from the group consisting of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, a TCR zeta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, a CD3 delta TCR subunit, CD45, CD4, CD5, CD8, CD9, CD16, CD22, CD33, CD28, CD37, CD64, CD80, CD86, CD134, CD137, CD154, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the transmembrane domain comprises, or comprises at least 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, or 30 or more consecutive amino acid residues of the transmembrane domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit.
- the transmembrane domain comprises a sequence having at least about 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99% or more sequence identity to a sequence encoding the transmembrane domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit.
- the transmembrane domain comprises a sequence encoding the transmembrane domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit having a truncation of at least 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 or more amino acids at the N- or C-terminus or at both the N- and C-terminus.
- the transmembrane domain can be attached to the extracellular region of the TFP, e.g., the antigen binding domain of the TFP, via a hinge, e.g., a hinge from a human protein.
- the hinge can be a human immunoglobulin (Ig) hinge, e.g., an IgG4 hinge, or a CD8a hinge.
- Ig immunoglobulin
- Linkers [0419]
- a short oligo- or polypeptide linker between 2 and 10 amino acids in length may form the linkage between the binding element and the TCR extracellular domain of the TFP.
- a glycine-serine doublet provides a particularly suitable linker.
- the linker may be at least about 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, or more in length.
- the linker comprises the amino acid sequence of GGGGSGGGGS (SEQ ID NO: 403) or a sequence (GGGGS)x (SEQ ID NO: 390) or (G4S)n (SEQ ID NO: 390), wherein X or n is 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 or more.
- X or n is an integer from 1 to 10.
- X or n is an integer from 1 to 4.
- X or n is 2. In some embodiments, X or n is 4.
- the linker is encoded by a nucleotide sequence of GGTGGCGGAGGTTCTGGAGGTGGAGGTTCC (SEQ ID NO: 404).
- Cytoplasmic Domain [0420]
- the cytoplasmic domain of the TFP can include an intracellular domain.
- the intracellular domain is from CD3 gamma, CD3 delta, CD3 epsilon, TCR alpha, TCR beta, TCR gamma, or TCR delta.
- the intracellular domain comprises a signaling domain, if the TFP contains CD3 gamma, delta or epsilon polypeptides; TCR alpha, TCR beta, TCR gamma, and TCR delta subunits generally have short (e.g., 1-19 amino acids in length) intracellular domains and are generally lacking in a signaling domain.
- An intracellular signaling domain is generally responsible for activation of at least one of the normal effector functions of the immune cell in which the TFP has been introduced.
- intracellular domains of TCR alpha, TCR beta, TCR gamma, and TCR delta do not have signaling domains, they are able to recruit proteins having a primary intracellular signaling domain described herein, e.g., CD3 zeta, which functions as an intracellular signaling domain.
- effector function refers to a specialized function of a cell. Effector function of a T cell, for example, may be cytolytic activity or helper activity including the secretion of cytokines.
- intracellular signaling domain refers to the portion of a protein which transduces the effector function signal and directs the cell to perform a specialized function.
- intracellular signaling domain While usually the entire intracellular signaling domain can be employed, in many cases it is not necessary to use the entire chain. To the extent that a truncated portion of the intracellular signaling domain is used, such truncated portion may be used in place of the intact chain as long as it transduces the effector function signal.
- the term intracellular signaling domain is thus meant to include any truncated portion of the intracellular signaling domain sufficient to transduce the effector function signal.
- intracellular domains for use in the TFP of the present disclosure include the cytoplasmic sequences of the T cell receptor (TCR) and co-receptors that are able to act in concert to initiate signal transduction following antigen receptor engagement, as well as any derivative or variant of these sequences and any recombinant sequence that has the same functional capability.
- the intracellular domain comprises the intracellular domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit.
- the intracellular domain comprises, or comprises at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 or more consecutive amino acid residues of the intracellular domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, or a TCR delta chain.
- the intracellular domain comprises a sequence having at least about 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99% or more sequence identity to a sequence encoding the intracellular domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, or a TCR delta chain.
- the transmembrane domain comprises a sequence encoding the intracellular domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, or a TCR delta chain having a truncation of at least 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 or more amino acids at the N- or C-terminus or at both the N- and C-terminus.
- the intracellular domain comprises, or comprises at least 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, or 62 or more consecutive amino acid residues of the intracellular domain of a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit.
- the intracellular domain comprises a sequence having at least about 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99% or more sequence identity to a sequence encoding the intracellular domain of a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit.
- the intracellular domain comprises a sequence encoding the intracellular domain of a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit having a truncation of at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25 or more amino acids at the N- or C-terminus or at both the N- and C-terminus.
- na ⁇ ve T cell activation can be said to be mediated by two distinct classes of cytoplasmic signaling sequences: those that initiate antigen-dependent primary activation through the TCR (primary intracellular signaling domains) and those that act in an antigen-independent manner to provide a secondary or costimulatory signal (secondary cytoplasmic domain, e.g., a costimulatory domain).
- primary intracellular signaling domains that act in a stimulatory manner may contain signaling motifs which are known as immunoreceptor tyrosine-based activation motifs (ITAMs).
- ITAMs immunoreceptor tyrosine-based activation motifs
- ITAMs containing primary intracellular signaling domains include those of CD3 zeta, FcR gamma, FcR beta, CD3 gamma, CD3 delta, CD3 epsilon, CD5, CD22, CD79a, CD79b, and CD66d.
- a TFP of the present disclosure comprises an intracellular signaling domain, e.g., a primary signaling domain of CD3-epsilon.
- a primary signaling domain comprises a modified ITAM domain, e.g., a mutated ITAM domain which has altered (e.g., increased or decreased) activity as compared to the native ITAM domain.
- a primary signaling domain comprises a modified ITAM-containing primary intracellular signaling domain, e.g., an optimized and/or truncated ITAM-containing primary intracellular signaling domain.
- a primary signaling domain comprises one, two, three, four or more ITAM motifs.
- the intracellular signaling domain of the TFP can comprise a CD3 signaling domain, e.g., CD3 epsilon, CD3 delta, CD3 gamma, or CD3 zeta, by itself or it can be combined with any other desired intracellular signaling domain(s) useful in the context of a TFP of the present disclosure.
- the intracellular signaling domain of the TFP can comprise a CD3 epsilon chain portion and a costimulatory signaling domain.
- the costimulatory signaling domain refers to a portion of the TFP comprising the intracellular domain of a costimulatory molecule.
- a costimulatory molecule is a cell surface molecule other than an antigen receptor or its ligands that is required for an efficient response of lymphocytes to an antigen.
- CD27 costimulation has been demonstrated to enhance expansion, effector function, and survival of human TFP-T cells in vitro and augments human T cell persistence and antitumor activity in vivo (Song et al., Blood.2012; 119(3):696-706).
- the intracellular signaling sequences within the cytoplasmic portion of the TFP of the present disclosure may be linked to each other in a random or specified order.
- a short oligo- or polypeptide linker for example, between 2 and 10 amino acids (e.g., 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids) in length may form the linkage between intracellular signaling sequences.
- a glycine-serine doublet can be used as a suitable linker.
- a single amino acid e.g., an alanine, a glycine, can be used as a suitable linker.
- the TFPs described herein may comprise a TCR extracellular domain, a TCR transmembrane domain, and a TCR intracellular domain, wherein at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from the same TCR subunit.
- at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from TCR alpha.
- at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from TCR beta.
- At least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from TCR gamma. In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from TCR delta. In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from CD3 epsilon. In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from CD3 delta.
- the TFPs described herein may comprise a TCR extracellular domain, a TCR transmembrane domain, and a TCR intracellular domain, wherein all three of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from the same TCR subunit.
- the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from CD3 epsilon.
- the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from CD3 delta. In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from CD3 gamma. In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain may comprise the constant domain of TCR alpha. In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain may comprise the constant domain of TCR beta.
- the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain may comprise the constant domain of TCR gamma. In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain may comprise the constant domain of TCR delta. In some embodiments, the constant domain of TCR alpha or the constant domain of TCR beta may be murine.
- the TFP-expressing cell co-expressing a switch polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof; and/or the TFP-expressing cell co-expressing a DN TGFBr2 as described herein can further comprise a second TFP, e.g., a second TFP that includes a different antigen binding domain, e.g., to the same target or a different target.
- the antigen binding domains of the different TFPs can be such that the antigen binding domains do not interact with one another.
- a cell expressing a first and second TFP can have an antigen binding domain of the first TFP, e.g., as a fragment, e.g., a scFv, that does not form an association with the antigen binding domain of the second TFP, e.g., the antigen binding domain of the second TFP is a V HH .
- TFP constructs can be generated as previously described.
- An anti-MSLN or CD19 binder can be linked to a CD3 or TCR DNA fragment by either a DNA sequence encoding a short linker (SL): AAAGGGGSGGGGSGGGGSLE (SEQ ID NO: 387) or a long linker (LL): AAAIEVMYPPPYLGGGGSGGGGSGGGGSLE (SEQ ID NO: 388) into pLRPO or p510 vector.
- the TFP used is TC-210 (e.g., an anti-MSLN MH1e VHH antibody linked to CD3 epsilon) having the sequence of SEQ ID NO: 195.
- the TC-210 sequence can comprise a CSF2RA signal peptide, an anti-MSLN sdAb, A3(G4S)3LE linker (SEQ ID NO: 387) and/or a CD3 epsilon.
- TC-210 comprises SEQ ID NO: 201, SEQ ID NO: 69, SEQ ID NO: 387, and SEQ ID NO: 258.
- the TFP used is TC-110 (e.g., an anti-CD19 FMC63 scFv antibody linked to CD3 epsilon) having the sequence of SEQ ID NO: 196.
- Anti-MSLN-CD3 epsilon (SEQ ID NO: 195) MLLLVTSLLLCELPHPAFLLIPEVQLVESGGGLVQPGGSLRLSCAASGGDWSANFMY WYRQAPGKQRELVARISGRGVVDYVESVKGRFTISRDNSKNTLYLQMNSLRAEDTA VYYCAVASYWGQGTLVTVSSAAAGGGGSGGGGSGGGGSLEDGNEEMGGITQTPYK VSISGTTVILTCPQYPGSEILWQHNDKNIGGDEDDKNIGSDEDHLSLKEFSELEQSGYY VCYPRGSKPEDANFYLYLRARVCENCMEMDVMSVATIVIVDICITGGLLLLVYYWS KNRKAKAKPVTRGAGAGGRQRGQNKERPPPVPNPDYEPIRKGQRDLYSGLNQRRI [0434] Anti-CD19-CD3 epsilon (SEQ ID NO: 196) MLLLVTSLLLCELPHPAFL
- recombinant nucleic acid molecules comprising a first nucleic acid sequence encoding a T cell receptor (TCR) fusion protein (TFP) as described herein and a second nucleic acid sequence encoding a switch polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof as described herein.
- TCR T cell receptor
- TGFBr2 transforming growth factor beta receptor II
- the TFP can comprise a TCR subunit comprising at least a portion of a TCR extracellular domain.
- the TCR subunit can further comprise a transmembrane domain.
- the TCR subunit can further comprise an intracellular domain of TCR gamma, TCR delta, TCR alpha or TCR beta or an intracellular domain comprising a stimulatory domain from an intracellular signaling domain of CD3 epsilon, CD3 gamma, CD3 delta.
- the TFP can further comprise an antibody (e.g., a human, humanized, or murine antibody) comprising an antigen binding domain.
- the recombinant nucleic acid molecule can further comprise a sequence encoding a TCR constant domain, wherein the TCR constant domain is a TCR alpha constant domain, a TCR beta constant domain, a TCR alpha constant domain and a TCR beta constant domain, a TCR gamma constant domain, a TCR delta constant domain, or a TCR gamma constant domain and a TCR delta constant domain.
- the TCR subunit and the antibody can be operatively linked.
- the TFP can functionally incorporate into a TCR complex (e.g., an endogenous TCR complex) when expressed in a T cell.
- the constant domain can comprise a constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain.
- the constant domain can comprise a full-length constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain.
- the constant domain can comprise a fragment (e.g., functional fragment) of the full-length constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain.
- the constant domain can comprise at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of the constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain.
- the sequence encoding the TCR constant domain can further encode the transmembrane domain and/or intracellular region of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain.
- the sequence encoding the TCR constant domain can encode a full-length constant region of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain.
- the constant region of a TCR chain can comprise a constant domain, a transmembrane domain, and an intracellular region.
- the constant region of a TCR chain can also exclude the transmembrane domain and the intracellular region of the TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain.
- the TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain described herein can be derived from various species.
- the TCR chain can be a murine or human TCR chain.
- the constant domain can comprise a constant domain of a murine or human TCR alpha chain, TCR beta chain, TCR gamma chain or TCR delta chain.
- the constant domain can comprise truncations, additions, or substitutions of a sequence of a constant domain described herein.
- the constant domain can comprise a truncated version of a constant domain described herein having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 19, SEQ ID NO: 20, SEQ ID NO: 21, SEQ ID NO: 22, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, SEQ ID NO:155, SEQ ID NO:207, SEQ ID NO:209, SEQ ID NO:243 or SEQ ID NO:265.
- the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more additional amino acid residues of SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 19, SEQ ID NO: 20, SEQ ID NO: 21, SEQ ID NO: 22, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, SEQ ID NO:155, SEQ ID NO:207, SEQ ID NO:209, SEQ ID NO:243 or SEQ ID NO:265.
- the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid substitutions of SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 19, SEQ ID NO: 20, SEQ ID NO: 21, SEQ ID NO: 22, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, SEQ ID NO:155, SEQ ID NO:207, SEQ ID NO:209, SEQ ID NO:243 or SEQ ID NO:265.
- the constant domain can comprise a sequence or fragment thereof of SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 19, SEQ ID NO: 20, SEQ ID NO: 21, SEQ ID NO: 22, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, SEQ ID NO:155, SEQ ID NO:207, SEQ ID NO:209, SEQ ID NO:243 or SEQ ID NO:265.
- the constant domain can comprise at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more modifications, mutations or deletions of the sequence of SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 19, SEQ ID NO: 20, SEQ ID NO: 21, SEQ ID NO: 22, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, SEQ ID NO:155, SEQ ID NO:207, SEQ ID NO:209, SEQ ID NO:243 or SEQ ID NO:265.
- the constant domain can comprise at most 20, 19, 18, 17, 16, 15, 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 modification, mutations or deletions of the sequence of SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 19, SEQ ID NO: 20, SEQ ID NO: 21, SEQ ID NO: 22, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, SEQ ID NO:155, SEQ ID NO:207, SEQ ID NO:209, SEQ ID NO:243 or SEQ ID NO:265.
- the constant domain can comprise a sequence having a sequence identity of at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 99% or 100% to the sequence of SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 19, SEQ ID NO: 20, SEQ ID NO: 21, or SEQ ID NO: 22, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, SEQ ID NO:155, SEQ ID NO:207, SEQ ID NO:209, SEQ ID NO:243 or SEQ ID NO:265.
- the murine TCR alpha constant domain can comprise positions 2-137 of SEQ ID NO:146.
- the murine TCR alpha constant domain can comprise truncations, additions, or substitutions of a sequence of a constant domain described herein.
- the constant domain can comprise a truncated version of a constant domain described herein having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of positions 2-137 of SEQ ID NO:146.
- the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more additional amino acid residues of positions 2-137 of SEQ ID NO:146.
- the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid substitutions of positions 2-137 of SEQ ID NO:146.
- the constant domain can comprise a sequence or fragment thereof of positions 2-137 of SEQ ID NO:146.
- the constant domain can comprise at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more modifications, mutations or deletions of the sequence of positions 2-137 of SEQ ID NO:146.
- the constant domain can comprise at most 20, 19, 18, 17, 16, 15, 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 modification, mutations or deletions of the sequence of positions 2-137 of SEQ ID NO:146.
- the constant domain can comprise a sequence having a sequence identity of at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 99% or 100% to the sequence of positions 2-137 of SEQ ID NO:146.
- the murine TCR beta constant domain can comprise positions 2-173 of SEQ ID NO:152.
- the murine TCR beta constant domain can comprise truncations, additions, or substitutions of a sequence of a constant domain described herein.
- the constant domain can comprise a truncated version of a constant domain described herein having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of positions 2-173 of SEQ ID NO:152.
- the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more additional amino acid residues of positions 2-173 of SEQ ID NO:152.
- the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid substitutions of positions 2-173 of SEQ ID NO:152.
- the constant domain can comprise a sequence or fragment thereof of positions 22-173 of SEQ ID NO:152.
- the constant domain can comprise at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more modifications, mutations or deletions of the sequence of positions 2-173 of SEQ ID NO:152.
- the constant domain can comprise at most 20, 19, 18, 17, 16, 15, 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 modification, mutations or deletions of the sequence of positions 2-173 of SEQ ID NO:152.
- the constant domain can comprise a sequence having a sequence identity of at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 99% or 100% to the sequence of positions 2-173 of SEQ ID NO:152.
- the TCR constant domain is a TCR delta constant domain.
- the TCR delta constant domain can comprise SEQ ID NO:20, SEQ ID NO:22, SEQ ID NO:243 or SEQ ID NO:265, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modification.
- the TCR delta constant domain can comprise SEQ ID NO:243.
- the TCR delta constant domain can comprise truncations, additions, or substitutions of a sequence of a constant domain described herein.
- the constant domain can comprise a truncated version of a constant domain described herein having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of SEQ ID NO:243.
- the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more additional amino acid residues of SEQ ID NO:243.
- the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid substitutions of SEQ ID NO:243.
- the constant domain can comprise a sequence or fragment thereof of SEQ ID NO:243.
- the constant domain can comprise at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more modifications, mutations or deletions of the sequence of SEQ ID NO:243.
- the constant domain can comprise at most 20, 19, 18, 17, 16, 15, 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 modification, mutations or deletions of the sequence of SEQ ID NO:243.
- the constant domain can comprise a sequence having a sequence identity of at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 99% or 100% to the sequence of SEQ ID NO:243.
- the TCR delta constant domain can comprise SEQ ID NO:20, SEQ ID NO:22, SEQ ID NO:243 or SEQ ID NO:265, functional fragments thereof, or amino acid sequences thereof having at least one but not more than 20 modifications.
- the sequence encoding a TCR delta constant domain further encodes a TCR delta variable domain, thereby encoding a full TCR delta domain.
- the full TCR delta domain can be delta 2 or delta 1.
- the full TCR delta constant domain can comprise SEQ ID NO:256, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the full TCR delta domain can comprise truncations, additions, or substitutions of a sequence of a constant domain described herein.
- the delta domain can comprise a truncated version of a delta domain described herein having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of SEQ ID NO:256.
- the delta domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more additional amino acid residues of SEQ ID NO:256.
- the delta domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid substitutions of SEQ ID NO:256.
- the delta domain can comprise a sequence or fragment thereof of SEQ ID NO:256.
- the delta domain can comprise at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more modifications, mutations or deletions of the sequence of SEQ ID NO:256.
- the delta domain can comprise at most 20, 19, 18, 17, 16, 15, 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 modification, mutations or deletions of the sequence of SEQ ID NO:256.
- the delta domain can comprise a sequence having a sequence identity of at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 99% or 100% to the sequence of SEQ ID NO:256.
- the TCR gamma constant domain can comprise SEQ ID NO:21.
- the TCR gamma constant domain can comprise truncations, additions, or substitutions of a sequence of a constant domain described herein.
- the constant domain can comprise a truncated version of a constant domain described herein having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of SEQ ID NO:21.
- the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more additional amino acid residues of SEQ ID NO:21.
- the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid substitutions of SEQ ID NO:21.
- the constant domain can comprise a sequence or fragment thereof of SEQ ID NO:21.
- the constant domain can comprise at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more modifications, mutations or deletions of the sequence of SEQ ID NO:21.
- the constant domain can comprise at most 20, 19, 18, 17, 16, 15, 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 modification, mutations or deletions of the sequence of SEQ ID NO:21.
- the constant domain can comprise a sequence having a sequence identity of at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 99% or 100% to the sequence of SEQ ID NO:243.
- the TCR gamma constant domain can comprise SEQ ID NO:21 or SEQ ID NO:155, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the sequence encoding the TCR gamma constant domain further encodes a TCR gamma variable domain, thereby encoding a full TCR gamma domain.
- the full TCR gamma domain can be gamma 9 or gamma 4.
- the full TCR gamma domain can comprise SEQ ID NO:255, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the full TCR gamma domain can comprise truncations, additions, or substitutions of a sequence of a constant domain described herein.
- the gamma domain can comprise a truncated version of a gamma domain described herein having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of SEQ ID NO:255.
- the gamma domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more additional amino acid residues of SEQ ID NO:255.
- the gamma domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid substitutions of SEQ ID NO:255.
- the gamma domain can comprise a sequence or fragment thereof of SEQ ID NO:255.
- the gamma domain can comprise at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more modifications, mutations or gamma of the sequence of SEQ ID NO:255.
- the gamma domain can comprise at most 20, 19, 18, 17, 16, 15, 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 modification, mutations or deletions of the sequence of SEQ ID NO:255.
- the gamma domain can comprise a sequence having a sequence identity of at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 99% or 100% to the sequence of SEQ ID NO:255.
- TCR beta chain (Homo sapiens): VEDLNKVFPPEVAVFEPSEAEISHTQKATLVCLATGFFPDHVELSWWVNGKEVHSGV STDPQPLKEQPALNDSRYCLSSRLRVSATFWQNPRNHFRCQVQFYGLSENDEWTQD RAKPVTQIVSAEAWGRADCGFTSVSYQQGVLSATILYEILLGKATLYAVLVSALVLM AMVKRKDF (SEQ ID NO: 16).
- the murine TCR beta chain constant region canonical sequence is: EDLRNVTPPKVSLFEPSKAEIANKQKATLVCLARGFFPDHVELSWWVNGKEVHSGV STDPQAYKESNYSYCLSSRLRVSATFWHNPRNHFRCQVQFHGLSEEDKWPEGSPKP VTQNISAEAWGRADCGITSASYQQGVLSATILYEILLGKATLYAVLVSTLVVMAMV KRKNS (SEQ ID NO:152).
- TCR alpha constant region (Mus musculus) (or [mm]TRAC(82-137)): ATYPSSDVPCDATLTEKSFETDMNLNFQNLSVMGLRILLLKVAGFNLLMTLRLWSS (SEQ ID NO: 17).
- the murine TCR alpha chain constant (mTRAC) region canonical sequence is: XIQNPEPAVYQLKDPRSQDSTLCLFTDFDSQINVPKTMESGTFITDKTVLDMKAMDS KSNGAIAWSNQTSFTCQDIFKETNATYPSSDVPCDATLTEKSFETDMNLNFQNLSVM GLRILLLKVAGFNLLMTLRLWSS (SEQ ID NO:146).
- TCR beta constant region (Mus musculus) (or [mm]TRBC1(123-173)): GRADCGITSASYQQGVLSATILYEILLGKATLYAVLVSTLVVMAMVKRKNS (SEQ ID NO: 18).
- the murine TCR beta chain constant region canonical sequence is: EDLRNVTPPKVSLFEPSKAEIANKQKATLVCLARGFFPDHVELSWWVNGKEVHSGV STDPQAYKESNYSYCLSSRLRVSATFWHNPRNHFRCQVQFHGLSEEDKWPEGSPKP VTQNISAEAWGRADCGITSASYQQGVLSATILYEILLGKATLYAVLVSTLVVMAMV KRKNS (SEQ ID NO:152).
- TCR beta chain (Homo sapiens): PVDSGVTQTPKHLITATGQRVTLRCSPRSGDLSVSWYQQSLDQGLQFLIQYYNGEER AKGNILERFSAQQFPDLHSELNLSSLELGDSALYFCASSPRTGLNTEAFFGQGTRLTV VEDLNKVFPPEVAVFEPSEAEISHTQKATLVCLATGFFPDHVELSWWVNGKEVHSGV STDPQPLKEQPALNDSRYCLSSRLRVSATFWQNPRNHFRCQVQFYGLSENDEWTQD RAKPVTQIVSAEAWGRADCGFTSVSYQQGVLSATILYEILLGKATLYAVLVSALVLM AMVKRKDF (SEQ ID NO: 19).
- TCR delta constant region version 1 (Homo sapiens): SQPHTKPSVFVMKNGTNVACLVKEFYPKDIRINLVSSKKITEFDPAIVISPSGKYNAV KLGKYEDSNSVTCSVQHDNKTVHSTDFEVKTDSTDHVKPKETENTKQPSKSCHKPK AIVHTEKVNMMSLTVLGLRMLFAKTVAVNFLLTAKLFF (SEQ ID NO: 20).
- TCR gamma constant region (Homo sapiens) (or [hs]TRGC(1-173)): DKQLDADVSPKPTIFLPSIAETKLQKAGTYLCLLEKFFPDVIKIHWQEKKSNTILGSQE GNTMKTNDTYMKFSWLTVPEKSLDKEHRCIVRHENNKNGVDQEIIFPPIKTDVITMD PKDNCSKDANDTLLLQLTNTSAYYMYLLLLLKSVVYFAIITCCLLRRTAFCCNGEKS (SEQ ID NO: 21).
- TCR delta constant region version 2 (Homo sapiens): SQPHTKPSVFVMKNGTNVACLVKEFYPKDIRINLVSSKKITEFDPAIVISPSGKYNAV KLGKYEDSNSVTCSVQHDNKTVHSTDFEVKTDSTDHVKPKETENTKQPSKSCHKPK AIVHTEKVNMMSLTVLGLRMLFAKTVAVNFLLTAK (SEQ ID NO: 22).
- the TCR constant domain is a TCR delta constant domain.
- the sequence encoding the TCR delta constant domain can further encode a second antigen binding domain or ligand binding domain that is operatively linked to the sequence encoding the TCR delta constant domain.
- the second antigen binding domain or ligand binding domain can be the same or different as the antigen binding domain or ligand binding domain of the TFP.
- the TCR constant domain is a TCR gamma constant domain.
- the sequence encoding the TCR gamma constant domain can further encode a second antigen binding domain or ligand binding domain that is operatively linked to the sequence encoding the TCR gamma constant domain.
- the second antigen binding domain or ligand binding domain can be the same or different as the antigen binding domain or ligand binding domain of the TFP.
- the recombinant nucleic acid comprises a sequence encoding a TCR gamma constant domain and a TCR delta constant domain.
- the TCR gamma constant domain can comprise SEQ ID NO:21 or SEQ ID NO:155, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the sequence encoding the TCR gamma constant domain can further encode a TCR gamma variable domain, thereby encoding a full TCR gamma domain.
- the TCR gamma domain can be gamma 9 or gamma 4.
- the full TCR gamma domain comprises SEQ ID NO:255, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the TCR delta constant domain can comprise SEQ ID NO:20, SEQ ID NO:22, SEQ ID NO:243 or SEQ ID NO:265, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the sequence encoding the TCR delta constant domain can further encode a TCR delta variable domain, thereby encoding a full TCR delta domain.
- the TCR delta domain can be delta 2 or delta 1.
- the full TCR delta domain can comprise SEQ ID NO:256, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. [0461]
- the TCR constant domain incorporates into a functional TCR complex when expressed in a T cell.
- the TCR constant domain incorporates into a same functional TCR complex as the functional TCR complex that incorporates the TFP when expressed in a T cell.
- the sequence encoding the TFP and the sequence encoding the TCR constant domain are contained within a same nucleic acid molecule.
- the sequence encoding the TFP and the sequence encoding the TCR constant domain are contained within different nucleic acid molecules.
- the sequence can further encode a cleavage site (e.g., a protease cleavage site) between the encoded TFP and the TCR constant domain.
- the cleavage site can be a protease cleavage site.
- the cleavage site can be a self-cleaving peptide such as a T2A, P2A, E2A or F2A cleavage site.
- the cleavage site can comprise a sequence of SEQ ID NO: 23.
- T2A cleavage site EGRGSLLTCGDVEENPGP (SEQ ID NO: 23).
- the TCR subunit of the TFP and the constant domain can comprise a sequence derived from a same TCR chain or a different TCR chain. In some cases, the TCR subunit of the TFP and the constant domain are derived from different TCR chains.
- the TCR subunit can comprise (1) at least a portion of a TCR extracellular domain, (2) a transmembrane domain, and (3) an intracellular domain, where the TCR extracellular domain, the transmembrane domain and the intracellular domain are derived from a TCR alpha chain, and the constant domain can comprise a constant domain of a TCR beta chain.
- the TCR subunit can comprise (1) at least a portion of a TCR extracellular domain, (2) a transmembrane domain, and (3) an intracellular domain, where the TCR extracellular domain, the transmembrane domain and the intracellular domain are derived from a TCR beta chain, and the constant domain can comprise a constant domain of a TCR alpha chain.
- the TCR subunit can comprise (1) at least a portion of a TCR extracellular domain, (2) a transmembrane domain, and (3) an intracellular domain, where the TCR extracellular domain, the transmembrane domain and the intracellular domain are derived from a TCR gamma chain, and the constant domain can comprise a constant domain of a TCR delta chain.
- the TCR subunit can comprise (1) at least a portion of a TCR extracellular domain, (2) a transmembrane domain, and (3) an intracellular domain, where the TCR extracellular domain, the transmembrane domain and the intracellular domain are derived from a TCR delta chain, and the constant domain can comprise a constant domain of a TCR gamma chain.
- the TCR subunit and the antibody domain, the antigen domain or the binding ligand or fragment thereof are operatively linked by a linker sequence.
- the transmembrane domain is a TCR transmembrane domain from CD3 epsilon, CD3 gamma, CD3 delta, TCR gamma, TCR delta, TCR alpha or TCR beta.
- the intracellular domain is derived from only CD3 epsilon, only CD3 gamma, only CD3 delta, only TCR gamma, only TCR delta, only TCR alpha or only TCR beta.
- the TCR subunit comprises (i) at least a portion of a TCR extracellular domain, (ii) a TCR transmembrane domain, and (iii) a TCR intracellular domain, wherein at least two or all of (i), (ii), and (iii) are from the same TCR subunit.
- the TCR extracellular domain comprises an extracellular domain or portion thereof of a protein selected from the group consisting of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, a CD3 delta TCR subunit, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the TCR subunit comprises a transmembrane domain comprising a transmembrane domain of a protein selected from the group consisting of a TCR alpha chain, a TCR beta chain, a TCR zeta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, a CD3 delta TCR subunit, CD45, CD4, CD5, CD8, CD9, CD16, CD22, CD33, CD28, CD37, CD64, CD80, CD86, CD134, CD137, CD154, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the TCR subunit comprises a TCR intracellular domain of TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, or a fragment thereof.
- the TCR subunit comprises an intracellular domain comprising a stimulatory domain of a protein selected from an intracellular signaling domain of CD3 epsilon, CD3 gamma or CD3 delta, or an amino acid sequence having at least one modification thereto.
- the TCR subunit can comprise (i) at least a portion of a TCR extracellular domain, (ii) a TCR transmembrane domain, and (iii) a TCR intracellular domain of a TCR gamma chain or a TCR delta chain.
- the TCR extracellular domain can comprise the extracellular portion of a constant domain of a TCR gamma chain or a TCR delta chain, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the TCR subunit comprising (i) at least a portion of a TCR extracellular domain, (ii) a TCR transmembrane domain, and (iii) a TCR intracellular domain is or comprises a delta constant domain, or a fragment thereof, e.g., a delta constant domain described herein.
- the delta constant domain can have the sequence of SEQ ID NO:20, SEQ ID NO:22, SEQ ID NO:243 or SEQ ID NO:265, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the TCR subunit comprising (i) at least a portion of a TCR extracellular domain, (ii) a TCR transmembrane domain, and (iii) a TCR intracellular domain is or comprises a gamma constant domain, e.g., a gamma constant domain described herein.
- the gamma constant domain can have the sequence of SEQ ID NO:21 or SEQ ID NO:155, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the extracellular domain of the TFP may not comprise the variable domain of a gamma chain or a delta chain.
- the TCR subunit of the TFP can comprise the extracellular, transmembrane and intracellular domain of CD3 epsilon, CD3 gamma, or CD3 delta.
- recombinant nucleic acid comprises a TFP comprising the extracellular, transmembrane and intracellular domain of CD3 epsilon, CD3 gamma, or CD3 delta and the constant domains of TCR beta and TCR alpha.
- recombinant nucleic acid comprises a TFP comprising the extracellular, transmembrane and intracellular domain of CD3 epsilon and the constant domains of TCR gamma and TCR delta.
- recombinant nucleic acid comprises a TFP comprising the extracellular, transmembrane and intracellular domain of CD3 epsilon and full length TCF gamma and full length TCR delta.
- the TCR subunit of the TFP comprises CD3 epsilon.
- the TCR subunit of CD3 epsilon can comprise the sequence of SEQ ID NO:258 functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the TCR subunit comprising at least a portion of a murine TCR alpha or murine TCR beta extracellular domain and a murine TCR alpha or murine TCR beta transmembrane domain is or comprises a TCR alpha constant domain or a TCR beta constant domain.
- the TCR subunit can comprise an intracellular domain of murine TCR alpha or murine TCR beta.
- the TCR constant domain can be a TCR alpha constant domain, e.g., a TCR alpha constant domain described herein.
- the TCR alpha constant domain can comprise SEQ ID NO:17, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, or SEQ ID NO:207, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the sequence encoding the TCR alpha constant domain can further encode a second antigen binding domain or ligand binding domain that is operatively linked to the sequence encoding the TCR alpha constant domain.
- the second antigen binding domain or ligand binding domain can be the same or different as the antigen binding domain or ligand binding domain of the TFP.
- the TCR alpha constant domain can comprise a murine TCR alpha constant domain.
- the murine TCR alpha constant domain can comprise amino acids 2-137 of the murine TCR alpha constant domain.
- the murine TCR alpha constant domain can comprise amino acids 2-137 of SEQ ID NO:146.
- the murine TCR alpha constant domain can comprise a sequence of SEQ ID NO:207.
- the murine TCR alpha constant domain can comprise amino acids 82-137 of SEQ ID NO:146.
- the murine TCR alpha constant domain comprises a sequence of SEQ ID NO:17.
- the TCR constant domain can be a TCR beta constant domain, e.g., a TCR beta constant domain described herein.
- the TCR beta constant domain can comprise SEQ ID NO:18, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, or SEQ ID NO:209, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the sequence encoding the TCR beta constant domain can further encode a second antigen binding domain or ligand binding domain that is operatively linked to the sequence encoding the TCR beta constant domain.
- the second antigen binding domain or ligand binding domain can be the same or different as the antigen binding domain or ligand binding domain of the TFP.
- TCR beta constant domain can comprise a murine TCR beta constant domain.
- the murine TCR beta constant domain can comprise amino acids 2-173 of the murine TCR beta constant domain.
- the murine TCR beta constant domain can comprise amino acids 2-173 of SEQ ID NO:152.
- the murine TCR beta constant domain can comprise SEQ ID NO:209.
- the TCR beta constant domain can comprise amino acids 123-173 of SEQ ID NO:152.
- the TCR beta constant domain can comprise SEQ ID NO:18 [0473]
- the recombinant nucleic acid can comprise sequence encoding a TCR alpha constant domain and a TCR beta constant domain.
- the TCR alpha constant domain can comprise SEQ ID NO:17, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, or SEQ ID NO:207, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the TCR beta constant domain can comprise SEQ ID NO:18, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, or SEQ ID NO:209, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the intracellular signaling domain can be CD3 epsilon, CD3 gamma, or CD3 delta.
- the intracellular signaling domain can be CD3 epsilon.
- the sequence encoding the TCR constant domain can comprise from 5’ to 3’, a first leader sequence, an antigen binding domain sequence, a linker, a TRAC gene sequence, a cleavable linker sequence, a second leader sequence, and a TRBC gene sequence.
- the sequence encoding the TCR constant domain can comprise, from 5’ to 3’, a first leader sequence, an antigen binding domain sequence, a linker, a TRAC gene sequence, a cleavable linker sequence, a second leader sequence, and a TRBC gene sequence.
- the sequence encoding the TCR constant domain can comprise, from 5’ to 3’, a first leader sequence, a TRAC gene sequence, a cleavable linker sequence, a second leader sequence, an antigen binding domain sequence, a linker, and a TRBC gene sequence.
- the sequence encoding the TCR constant domain can comprise, from 5’ to 3’, a first leader sequence, an antigen binding domain sequence, a linker, a TRAC gene sequence, a cleavable linker sequence, a second leader sequence, an antigen binding domain sequence, a linker, and a TRBC gene sequence.
- the sequence encoding the TCR constant domain can comprise, from 5’-3’, a first leader sequence, a TRAC gene sequence, a first cleavable linker sequence, a second leader sequence, a TRBC gene sequence, a second cleavable linker sequence, a third leader sequence, an antigen binding domain sequence, a linker sequence, and a CD3 epsilon gene sequence.
- the at least one but not more than 20 modifications thereto of a sequence described herein can comprise a modification of an amino acid that mediates cell signaling or a modification of an amino acid that is phosphorylated in response to a ligand binding to the TFP.
- the TCR subunit comprises an intracellular domain comprising a stimulatory domain of a protein selected from a functional signaling domain of 4-1BB and/or a functional signaling domain of CD3 zeta, or an amino acid sequence having at least one modification thereto.
- the recombinant nucleic acid further comprises a sequence encoding a costimulatory domain.
- the costimulatory domain comprises a functional signaling domain of a protein selected from the group consisting of OX40, CD2, CD27, CD28, CD5, ICAM-1, LFA-1 (CD11a/CD18), ICOS (CD278), 4-1BB (CD137), IL- 15Ra, IL12R, IL18R, IL21R, GITR, CD30, CD40, BAFFR, HVEM, CD7, LIGHT, NKG2C, SLAMF7, NKp80, CD160, CD226, Fc ⁇ RI, Fc ⁇ RII, and Fc ⁇ RIII, and amino acid sequences thereof having at least one but not more than 20 modifications thereto.
- a protein selected from the group consisting of OX40, CD2, CD27, CD28, CD5, ICAM-1, LFA-1 (CD11a/CD18), ICOS (CD278), 4-1BB (CD137), IL- 15Ra, IL12R, IL18R, IL21R, GITR,
- the TCR subunit comprises an immunoreceptor tyrosine-based activation motif (ITAM) of a TCR subunit that comprises an ITAM or portion thereof of a protein selected from the group consisting of CD3 zeta TCR subunit, CD3 epsilon TCR subunit, CD3 gamma TCR subunit, CD3 delta TCR subunit, TCR zeta chain, Fc epsilon receptor 1 chain, Fc epsilon receptor 2 chain, Fc gamma receptor 1 chain, Fc gamma receptor 2a chain, Fc gamma receptor 2b1 chain, Fc gamma receptor 2b2 chain, Fc gamma receptor 3a chain, Fc gamma receptor 3b chain, Fc beta receptor 1 chain, TYROBP (DAP12), CD5, CD16a, CD16b, CD22, CD23, CD32, CD64, CD79a, CD79b, CD89, CD278, CD66
- ITAM immunorecept
- the ITAM replaces an ITAM of CD3 gamma, CD3 delta, or CD3 epsilon.
- the ITAM is selected from the group consisting of CD3 zeta TCR subunit, CD3 epsilon TCR subunit, CD3 gamma TCR subunit, and CD3 delta TCR subunit and replaces a different ITAM selected from the group consisting of CD3 zeta TCR subunit, CD3 epsilon TCR subunit, CD3 gamma TCR subunit, and CD3 delta TCR subunit.
- the TFP, the TCR gamma constant domain, the TCR delta constant domain, and any combination thereof is capable of functionally interacting with an endogenous TCR complex and/or at least one endogenous TCR polypeptide.
- the TCR constant domain is a TCR gamma constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of TCR delta, CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof
- the TCR constant domain is a TCR delta constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of TCR gamma, CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof
- the TCR constant domain is a TCR gamma constant domain and a TCR delta constant domain and the TFP functionally integrates into a TCR complex comprising
- the at least one but not more than 20 modifications thereto comprise a modification of an amino acid that mediates cell signaling or a modification of an amino acid that is phosphorylated in response to a ligand binding to the TFP.
- the antibody or antigen binding domain can be an antibody fragment.
- the antibody or antigen binding domain can be murine, human or humanized. In some instances, the murine, human or humanized antibody is an antibody fragment. In some instances, the antibody fragment is a scFv, a single domain antibody domain, a VH domain or a VL domain.
- An antigen binding domain described herein can be selected from a group consisting of an anti-CD19 binding domain, an anti-B-cell maturation antigen (BCMA) binding domain, an anti-mesothelin (MSLN) binding domain, an anti-CD20 binding domain, an anti-CD70 binding domain, an anti-CD79b binding domain, an anti-PMSA binding domain, an anti- MUC16 binding domain, an anti-CD22 binding domain, an anti-PD-L1 binding domain, an anti BAFF receptor binding domain, an anti-Nectin-4 binding domain, an anti-TROP-2 binding domain, an anti-GPC3 binding domain, and anti-ROR-1 binding domain.
- BCMA anti-B-cell maturation antigen
- MSLN anti-mesothelin
- the nucleic acid is selected from the group consisting of a DNA and an RNA. In some instances, the nucleic acid is an mRNA. In some instances, the recombinant nucleic acid comprises a nucleic acid analog, wherein the nucleic acid analog is not in an encoding sequence of the recombinant nucleic acid.
- the nucleic analog is selected from the group consisting of 2’-O-methyl, 2’-O-methoxyethyl (2’-O- MOE), 2’-O-aminopropyl, 2’-deoxy, T-deoxy-2’-fluoro, 2’-O-aminopropyl (2’-O-AP), 2'-O- dimethylaminoethyl (2’-O-DMAOE), 2’-O-dimethylaminopropyl (2’-O-DMAP), T-O- dimethylaminoethyloxyethyl (2’-O-DMAEOE), 2’-O-N-methylacetamido (2’-O-NMA) modified, a locked nucleic acid (LNA), an ethylene nucleic acid (ENA), a peptide nucleic acid (PNA), a 1’,5’- anhydrohexitol nucleic acid (HNA), a morpholino, a morpholino
- the recombinant nucleic acid further comprises a leader sequence. In some instances, the recombinant nucleic acid further comprises a promoter sequence. In some instances, the recombinant nucleic acid further comprises a sequence encoding a poly(A) tail. In some instances, the recombinant nucleic acid further comprises a 3’UTR sequence. In some instances, the nucleic acid is an isolated nucleic acid or a non-naturally occurring nucleic acid. In some instances, the nucleic acid is an in vitro transcribed nucleic acid. [0485] In some instances, the recombinant nucleic acid further comprises a sequence encoding a TCR alpha transmembrane domain.
- the recombinant nucleic acid further comprises a sequence encoding a TCR beta transmembrane domain. In some instances, the recombinant nucleic acid further comprises a sequence encoding a TCR alpha transmembrane domain and a sequence encoding a TCR beta transmembrane domain.
- the TCR subunit comprises an immunoreceptor tyrosine-based activation motif (ITAM) of a TCR subunit that comprises an ITAM or portion thereof of a protein selected from the group consisting of CD3 zeta TCR subunit, CD3 epsilon TCR subunit, CD3 gamma TCR subunit, CD3 delta TCR subunit, TCR zeta chain, Fc epsilon receptor 1 chain, Fc epsilon receptor 2 chain, Fc gamma receptor 1 chain, Fc gamma receptor 2a chain, Fc gamma receptor 2b1 chain, Fc gamma receptor 2b2 chain, Fc gamma receptor 3a chain, Fc gamma receptor 3b chain, Fc beta receptor 1 chain, TYROBP (DAP12), CD5, CD16a, CD16b, CD22, CD23, CD32, CD64, CD79a, CD79b, CD89, CD278, CD66
- ITAM immunorecept
- the ITAM replaces an ITAM of CD3 gamma, CD3 delta, or CD3 epsilon.
- the ITAM is selected from the group consisting of CD3 zeta TCR subunit, CD3 epsilon TCR subunit, CD3 gamma TCR subunit, and CD3 delta TCR subunit and replaces a different ITAM selected from the group consisting of CD3 zeta TCR subunit, CD3 epsilon TCR subunit, CD3 gamma TCR subunit, and CD3 delta TCR subunit.
- the TFP, the TCR gamma constant domain, the TCR delta constant domain, the TCR alpha constant domain, the TCR beta constant domain, and any combination thereof is capable of functionally interacting with an endogenous TCR complex and/or at least one endogenous TCR polypeptide.
- the TCR constant domain is a TCR gamma constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of TCR beta, CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof;
- the TCR constant domain is a TCR delta constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of TCR alpha, CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof;
- the TCR constant domain is a TCR gamma constant domain and a TCR delta constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof;
- the TCR constant domain is a TCR alpha constant domain and the TFP functionally integrates into a TCR complex comprising
- the at least one but not more than 20 modifications thereto comprise a modification of an amino acid that mediates cell signaling or a modification of an amino acid that is phosphorylated in response to a ligand binding to the TFP.
- murine, human or humanized antibody comprising an antigen binding domain.
- the antigen binding domain described herein can be selected from a group consisting of an anti-CD19 binding domain, an anti-B-cell maturation antigen (BCMA) binding domain, an anti-mesothelin (MSLN) binding domain, an anti-CD20 binding domain, an anti-CD70 binding domain, an anti-CD79b binding domain, , an anti-PMSA binding domain, an anti-MUC16 binding domain, an anti-CD22 binding domain, an anti-PD-1 binding domain, an anti-PD-L1 binding domain, an anti IL13R ⁇ 2 binding domain, anti BAFF receptor binding domain, an anti-Nectin-4 binding domain, an anti-TROP-2 binding domain, an anti-GPC3 binding domain, and anti-ROR-1 binding domain.
- BCMA anti-B-cell maturation antigen
- MSLN anti-mesothelin
- the antigen binding domain comprises a ligand.
- the ligand binds to the receptor of a cell.
- the ligand binds to the polypeptide expressed on a surface of a cell.
- the receptor or polypeptide expressed on a surface of a cell comprises a stress response receptor or polypeptide.
- the receptor or polypeptide expressed on a surface of a cell is an MHC class I- related glycoprotein.
- the MHC class I-related glycoprotein is selected from the group consisting of MICA, MICB, RAET1E, RAET1G, ULBP1, ULBP2, ULBP3, ULBP4 and combinations thereof.
- the antigen domain comprises a monomer, a dimer, a trimer, a tetramer, a pentamer, a hexamer, a heptamer, an octomer, a nonamer, or a decamer.
- the antigen domain comprises a monomer or a dimer of the ligand or fragment thereof.
- the ligand or fragment thereof is a monomer, a dimer, a trimer, a tetramer, a pentamer, a hexamer, a heptamer, an octomer, a nonamer, or a decamer.
- the ligand or fragment thereof is a monomer or a dimer.
- the antigen domain does not comprise an antibody or fragment thereof.
- the antigen domain does not comprise a variable region.
- the antigen domain does not comprise a CDR.
- the ligand or fragment thereof is a Natural Killer Group 2D (NKG2D) ligand or a fragment thereof.
- the TCR subunit and the antibody domain, the antigen domain or the binding ligand or fragment thereof are operatively linked by a linker sequence.
- the transmembrane domain is a TCR transmembrane domain from CD3 epsilon, CD3 gamma, CD3 delta, TCR alpha, TCR beta, TCR delta, or TCR gamma.
- the intracellular domain is derived from only CD3 epsilon, only CD3 gamma, only CD3 delta, only TCR alpha, only TCR beta, only TCR delta, or only TCR gamma.
- the TCR subunit comprises (i) at least a portion of a TCR extracellular domain, (ii) a TCR transmembrane domain, and (iii) a TCR intracellular domain, wherein at least two of (i), (ii), and (iii) are from the same TCR subunit.
- the TCR extracellular domain comprises an extracellular domain or portion thereof of a protein selected from the group consisting of a TCR alpha chain, a TCR beta chain, a TCR delta chain, a TCR gamma chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, a CD3 delta TCR subunit, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
- the TFP, the TCR gamma constant domain, the TCR delta constant domain, the TCR alpha constant domain, the TCR beta constant domain, and any combination thereof is capable of functionally interacting with an endogenous TCR complex and/or at least one endogenous TCR polypeptide.
- the TCR constant domain is a TCR gamma constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of TCR beta, CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof;
- the TCR constant domain is a TCR delta constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of TCR gamma, CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof;
- the TCR constant domain is a TCR gamma constant domain and a TCR delta constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof;
- the TCR constant domain is a TCR alpha constant domain and the TFP functionally integrates into a TCR complex
- the at least one but not more than 20 modifications thereto comprise a modification of an amino acid that mediates cell signaling or a modification of an amino acid that is phosphorylated in response to a ligand binding to the TFP.
- the murine, human or humanized antibody is an antibody fragment.
- the antibody fragment is a scFv, a single domain antibody domain, a VH domain or a VL domain.
- An antigen binding domain described herein can be selected from a group consisting of an anti-CD19 binding domain, an anti-B-cell maturation antigen (BCMA) binding domain, an anti-mesothelin (MSLN) binding domain, an anti-CD20 binding domain, an anti-CD70 binding domain, an anti-CD79b binding domain, , an anti-PMSA binding domain, an anti-MUC16 binding domain, an anti-CD22 binding domain, an anti-PD-L1 binding domain, an anti-BAFF receptor binding domain, an anti-Nectin-4 binding domain, an anti-TROP-2 binding domain, an anti-GPC3 binding domain, and anti-ROR-1 binding domain.
- BCMA anti-B-cell maturation antigen
- MSLN anti-mesothelin
- a sequence encoding the antigen binding domain or ligand binding domain is operatively linked to a sequence encoding a delta constant domain.
- the intracellular domain is an intracellular domain of TCR gamma.
- a sequence encoding the antigen binding domain or ligand binding domain is operatively linked to a sequence encoding a gamma constant domain.
- the intracellular domain is an intracellular domain of TCR delta.
- a sequence encoding the antigen binding domain or ligand binding domain is operatively linked to both a sequence encoding a TCR delta constant domain or fragment thereof and a TCR gamma constant domain or fragment thereof.
- the intracellular signaling domain is CD3 epsilon, CD3 gamma, or CD3 delta. In some embodiments, the intracellular signaling domain is CD3 epsilon. In some embodiments, the recombinant nucleic acid further comprises at least one leader sequence and at least one linker. In some embodiments, the recombinant nucleic acid further comprises a portion of a TCR alpha constant domain, a portion of a TCR beta domain, or both.
- the sequence comprises, from 5’ to 3’, a first leader sequence, an antigen binding domain sequence, a linker, a TRDC gene sequence, a cleavable linker sequence, a second leader sequence, and a TRGC gene sequence. In some embodiments, the sequence comprises, from 5’-3’, a first leader sequence, a TRDC gene sequence, a cleavable linker sequence, a second leader sequence, an antigen binding domain sequence, a linker sequence, and a TRGC gene sequence.
- the sequence comprises, from 5’-3’, a first leader sequence, an antigen binding domain sequence, a first linker sequence, a TRDC gene sequence, a cleavable linker, a second leader sequence, a second antigen binding domain sequence, a second linker sequence, and a TRGC gene sequence.
- the sequence comprises, from 5’-3’, a first leader sequence, a TRDC gene sequence, a first cleavable linker sequence, a second leader sequence, a TRGC gene sequence, a second cleavable linker sequence, a third leader sequence, an antigen binding domain sequence, a linker sequence, and a CD3 epsilon gene sequence.
- the sequence comprises, from 5’- 3’, a first leader sequence, a first antigen binding domain sequence, a first linker sequence, a TRDC gene sequence or fragment thereof, a TRAC gene sequence or fragment thereof, a cleavable linker sequence, a second leader sequence, a second antigen binding domain sequence, a second linker sequence, a TRGC gene sequence or fragment thereof, and a TRBC gene sequence or fragment thereof.
- the binding ligand is capable of binding an Fc domain of the antibody.
- the binding ligand is capable of selectively binding an IgG1 antibody.
- the binding ligand is capable of specifically binding an IgG4 antibody.
- the antibody or fragment thereof binds to a cell surface antigen. In some embodiments, the antibody or fragment thereof is murine, human or humanized. In some embodiments, the antibody or fragment thereof binds to a cell surface antigen on the surface of a tumor cell. In some embodiments, the binding ligand comprises a monomer, a dimer, a trimer, a tetramer, a pentamer, a hexamer, a heptamer, an octomer, a nonamer, or a decamer. In some embodiments, the binding ligand does not comprise an antibody or fragment thereof. In some embodiments, the binding ligand comprises a CD16 polypeptide or fragment thereof.
- the binding ligand comprises a CD16-binding polypeptide. In some embodiments, the binding ligand is human or humanized. In some embodiments, the recombinant nucleic acid further comprises a nucleic acid sequence encoding an antibody or fragment thereof capable of being bound by the binding ligand. In some embodiments, the antibody or fragment thereof is capable of being secreted from a cell.
- the present disclosure in some cases, provides a recombinant nucleic acid comprising: a first nucleic acid sequence encoding a T cell receptor (TCR) fusion protein (TFP) wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain, wherein the TCR subunit and the antigen binding domain are operatively linked; and a second nucleic acid sequence encoding a polypeptide comprising a dominant negative transforming growth factor beta receptor II (TGFBr2) as described herein.
- TCR T cell receptor
- TFP T cell receptor fusion protein
- TGFBr2 dominant negative transforming growth factor beta receptor II
- the recombinant nucleic acid further comprises a sequence encoding a TCR constant domain, wherein the TCR constant domain is a TCR gamma constant domain, a TCR delta constant domain, or a TCR gamma constant domain and a TCR delta constant domain.
- a modified T cell further comprises a functional disruption of an endogenous TCR.
- the present disclosure provides a recombinant nucleic acid comprising: a first nucleic acid sequence encoding a T cell receptor (TCR) fusion protein (TFP) wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain, wherein the TCR subunit and the antigen binding domain are operatively linked; and a second nucleic acid sequence encoding a switch polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof as described herein.
- TCR T cell receptor
- TFP TFP fusion protein
- TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain, wherein the
- the recombinant nucleic acid further comprises a sequence encoding a TCR constant domain, wherein the TCR constant domain is a TCR gamma constant domain, a TCR delta constant domain, or a TCR gamma constant domain and a TCR delta constant domain.
- a modified T cell further comprises a functional disruption of an endogenous TCR.
- the present disclosure in some cases, provides a recombinant nucleic acid comprising (a) a sequence encoding a T cell receptor (TCR) fusion protein (TFP) comprising (i) a TCR subunit comprising (1) at least a portion of a TCR extracellular domain, (2) a transmembrane domain, and (3) an intracellular domain of TCR alpha, TCR beta, TCR gamma, or TCR delta or an intracellular domain comprising a stimulatory domain from an intracellular signaling domain of CD3 epsilon, CD3 gamma, or CD3 delta, and (ii) a binding ligand or a fragment thereof that is capable of binding to an antibody or fragment thereof; and (b) an antigen binding domain, wherein the TCR subunit and the antigen binding domain are operatively linked; and a second nucleic acid sequence encoding (i) a switch polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or
- the recombinant nucleic acid further comprises a sequence encoding a TCR constant domain, wherein the TCR constant domain is a TCR gamma constant domain, a TCR delta constant domain, or a TCR gamma constant domain and a TCR delta constant domain.
- a modified T cell further comprises a functional disruption of an endogenous TCR.
- recombinant nucleic acid molecules described herein further comprise a leader sequence.
- the recombinant nucleic acid molecule is selected from the group consisting of a DNA and an RNA.
- the recombinant nucleic acid molecule is an mRNA.
- the recombinant nucleic acid molecule is a circRNA. In some embodiments, the recombinant nucleic acid molecule comprises a nucleic acid analog. In some embodiments, the nucleic acid analog is not in an encoding sequence of the recombinant nucleic acid.
- the nucleic analog is selected from the group consisting of 2’-O-methyl, 2’-O-methoxyethyl (2’- O-MOE), 2’-O-aminopropyl, 2’-deoxy, T-deoxy-2’-fluoro, 2’-O-aminopropyl (2’-O-AP), 2'- O-dimethylaminoethyl (2’-O-DMAOE), 2’-O-dimethylaminopropyl (2’-O-DMAP), T-O- dimethylaminoethyloxyethyl (2’-O-DMAEOE), 2’-O-N-methylacetamido (2’-O-NMA) modified, a locked nucleic acid (LNA), an ethylene nucleic acid (ENA), a peptide nucleic acid (PNA), a 1’,5’- anhydrohexitol nucleic acid (HNA), a morpholino,
- LNA locked
- the recombinant nucleic acid molecule further comprises a leader sequence. In some embodiments, the recombinant nucleic acid molecule further comprises a promoter sequence. In some embodiments, the recombinant nucleic acid molecule further comprises a sequence encoding a poly(A) tail. In some embodiments, the recombinant nucleic acid molecule further comprises a 3’UTR sequence. In some embodiments, the recombinant nucleic acid molecule is an isolated nucleic acid or a non-naturally occurring nucleic acid. In some embodiments, the nucleic acid is an in vitro transcribed nucleic acid.
- Vectors comprising the recombinant nucleic acid molecules as described herein.
- the vector can be directly transduced into a cell, e.g., a T cell.
- the vector is a cloning or expression vector, e.g., a vector including, but not limited to, one or more plasmids (e.g., expression plasmids, cloning vectors, minicircles, minivectors, double minute chromosomes), retroviral and lentiviral vector constructs.
- the vector is capable of expressing the TFP construct as described herein and a switch polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, or a dominant negative TGFBr2 comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, in mammalian T cells.
- the mammalian T cell is a human T cell.
- the vector is selected from the group consisting of a DNA, a RNA, a plasmid, a lentivirus vector, adenoviral vector, an adeno-associated viral vector (AAV), a Rous sarcoma viral (RSV) vector, or a retrovirus vector.
- the vector is an AAV6 vector.
- the vector further comprises a promoter.
- the vector is an in vitro transcribed vector.
- the nucleic acid sequences coding for the desired molecules can be obtained using recombinant methods known in the art, such as, for example by screening libraries from cells expressing the gene, by deriving the gene from a vector known to include the same, or by isolating directly from cells and tissues containing the same, using standard techniques. Alternatively, the gene of interest can be produced synthetically, rather than cloned.
- the present disclosure also provides vectors in which a DNA of the present disclosure is inserted. Vectors derived from retroviruses such as the lentivirus are suitable tools to achieve long-term gene transfer since they allow long-term, stable integration of a transgene and its propagation in daughter cells.
- Lentiviral vectors have the added advantage over vectors derived from onco-retroviruses such as murine leukemia viruses in that they can transduce non-proliferating cells, such as hepatocytes. They also have the added advantage of low immunogenicity.
- the vector comprising the nucleic acid encoding the desired TFP and (i) a switch polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof or (ii) a dominant negative TGFBr2 of the present disclosure is an adenoviral vector (A5/35).
- nucleic acids encoding TFPs as described herein and (i) a switch polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein or (ii) a dominant negative TGFBr2 comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein can be accomplished using of transposons such as sleeping beauty, crisper, CAS9, and zinc finger nucleases. See below June et al.2009 Nature Reviews Immunology 9.10: 704-716, is incorporated herein by reference. [0508]
- the expression constructs of the present disclosure may also be used for nucleic acid immunization and gene therapy, using standard gene delivery protocols.
- the present disclosure provides a gene therapy vector.
- the nucleic acid can be cloned into a number of types of vectors.
- the nucleic acid can be cloned into a vector including, but not limited to a plasmid, a phagemid, a phage derivative, an animal virus, and a cosmid.
- Vectors of particular interest include expression vectors, replication vectors, probe generation vectors, and sequencing vectors.
- the expression vector may be provided to a cell in the form of a viral vector.
- Viral vector technology is well known in the art and is described, for example, in Sambrook et al., 2012, Molecular Cloning: A Laboratory Manual, volumes 1-4, Cold Spring Harbor Press, NY), and in other virology and molecular biology manuals.
- Viruses, which are useful as vectors include, but are not limited to, retroviruses, adenoviruses, adeno-associated viruses, herpes viruses, and lentiviruses.
- a suitable vector contains an origin of replication functional in at least one organism, a promoter sequence, convenient restriction endonuclease sites, and one or more selectable markers, (e.g., WO 01/96584; WO 01/29058; and U.S. Pat. No.6,326,193).
- selectable markers e.g., WO 01/96584; WO 01/29058; and U.S. Pat. No.6,326,193
- retroviruses provide a convenient platform for gene delivery systems.
- a selected gene can be inserted into a vector and packaged in retroviral particles using techniques known in the art.
- the recombinant virus can then be isolated and delivered to cells of the subject either in vivo or ex vivo.
- retroviral systems are known in the art.
- adenovirus vectors are used.
- a number of adenovirus vectors are known in the art.
- lentivirus vectors are used.
- Additional promoter elements e.g., enhancers, regulate the frequency of transcriptional initiation. Typically, these are located in the region 30-110 bp upstream of the start site, although a number of promoters have been shown to contain functional elements downstream of the start site as well.
- the spacing between promoter elements frequently is flexible, so that promoter function is preserved when elements are inverted or moved relative to one another. In the thymidine kinase (tk) promoter, the spacing between promoter elements can be increased to 50 bp apart before activity begins to decline.
- tk thymidine kinase
- a promoter that is capable of expressing a TFP transgene as described herein and a (i) switch polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein or (ii) a dominant negative TGFBr2 comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein in a mammalian T cell is the EF1a promoter.
- the native EF1a promoter drives expression of the alpha subunit of the elongation factor-1 complex, which is responsible for the enzymatic delivery of aminoacyl tRNAs to the ribosome.
- the EF1a promoter has been extensively used in mammalian expression plasmids and has been shown to be effective in driving TFP and expression from transgenes cloned into a lentiviral vector (see, e.g., Milone et al., Mol. Ther. 17(8): 1453-1464 (2009)).
- Another example of a promoter is the immediate early cytomegalovirus (CMV) promoter sequence. This promoter sequence is a strong constitutive promoter sequence capable of driving high levels of expression of any polynucleotide sequence operatively linked thereto.
- CMV immediate early cytomegalovirus
- constitutive promoter sequences may also be used, including, but not limited to the simian virus 40 (SV40) early promoter, mouse mammary tumor virus (MMTV), human immunodeficiency virus (HIV) long terminal repeat (LTR) promoter, MoMuLV promoter, an avian leukemia virus promoter, an Epstein-Barr virus immediate early promoter, a Rous sarcoma virus promoter, as well as human gene promoters such as, but not limited to, the actin promoter, the myosin promoter, the elongation factor-1a promoter, the hemoglobin promoter, and the creatine kinase promoter. Further, the present disclosure should not be limited to the use of constitutive promoters.
- inducible promoters are also contemplated as part of the present disclosure.
- the use of an inducible promoter provides a molecular switch capable of turning on expression of the polynucleotide sequence which it is operatively linked when such expression is desired or turning off the expression when expression is not desired.
- inducible promoters include, but are not limited to a metallothionine promoter, a glucocorticoid promoter, a progesterone promoter, and a tetracycline-regulated promoter.
- the expression vector to be introduced into a cell can also contain either a selectable marker gene or a reporter gene or both to facilitate identification and selection of expressing cells from the population of cells sought to be transfected or infected through viral vectors.
- the selectable marker may be carried on a separate piece of DNA and used in a co-transfection procedure. Both selectable markers and reporter genes may be flanked with appropriate regulatory sequences to enable expression in the host cells.
- Reporter genes are used for identifying potentially transfected cells and for evaluating the functionality of regulatory sequences.
- a reporter gene is a gene that is not present in or expressed by the recipient organism or tissue and that encodes a polypeptide whose expression is manifested by some easily detectable property, e.g., enzymatic activity. Expression of the reporter gene is assayed at a suitable time after the DNA has been introduced into the recipient cells.
- Suitable reporter genes may include genes encoding luciferase, beta-galactosidase, chloramphenicol acetyl transferase, secreted alkaline phosphatase, or the green fluorescent protein gene (e.g., Ui-Tei et al., 2000 FEBS Letters 479: 79-82). Suitable expression systems are well known and may be prepared using known techniques or obtained commercially. In general, the construct with the minimal 5’ flanking region showing the highest level of expression of reporter gene is identified as the promoter. Such promoter regions may be linked to a reporter gene and used to evaluate agents for the ability to modulate promoter-driven transcription. [0516] Methods of introducing and expressing genes into a cell are known in the art.
- the vector can be readily introduced into a host cell, e.g., mammalian, bacterial, yeast, or insect cell by any method in the art.
- the expression vector can be transferred into a host cell by physical, chemical, or biological means.
- Physical methods for introducing a polynucleotide into a host cell include calcium phosphate precipitation, lipofection, particle bombardment, microinjection, electroporation, and the like. Methods for producing cells comprising vectors and/or exogenous nucleic acids are well-known in the art. See, for example, Sambrook et al., 2012, Molecular Cloning: A Laboratory Manual, volumes 1-4, Cold Spring Harbor Press, NY).
- a preferred method for the introduction of a polynucleotide into a host cell is calcium phosphate transfection
- Biological methods for introducing a polynucleotide of interest into a host cell include the use of DNA and RNA vectors.
- Viral vectors, and especially retroviral vectors have become the most widely used method for inserting genes into mammalian, e.g., human cells.
- Other viral vectors can be derived from lentivirus, poxviruses, herpes simplex virus I, adenoviruses and adeno-associated viruses, and the like (see, e.g., U.S. Pat. Nos.5,350,674 and 5,585,362.
- Chemical means for introducing a polynucleotide into a host cell include colloidal dispersion systems, such as macromolecule complexes, nanocapsules, microspheres, beads, and lipid-based systems including oil-in-water emulsions, micelles, mixed micelles, and liposomes.
- An exemplary colloidal system for use as a delivery vehicle in vitro and in vivo is a liposome (e.g., an artificial membrane vesicle).
- Other methods of state-of-the-art targeted delivery of nucleic acids are available, such as delivery of polynucleotides with targeted nanoparticles or other suitable sub-micron sized delivery system.
- an exemplary delivery vehicle is a liposome.
- lipid formulations is contemplated for the introduction of the nucleic acids into a host cell (in vitro, ex vivo or in vivo).
- the nucleic acid may be associated with a lipid.
- the nucleic acid associated with a lipid may be encapsulated in the aqueous interior of a liposome, interspersed within the lipid bilayer of a liposome, attached to a liposome via a linking molecule that is associated with both the liposome and the oligonucleotide, entrapped in a liposome, complexed with a liposome, dispersed in a solution containing a lipid, mixed with a lipid, combined with a lipid, contained as a suspension in a lipid, contained or complexed with a micelle, or otherwise associated with a lipid.
- Lipid, lipid/DNA or lipid/expression vector associated compositions are not limited to any particular structure in solution.
- Lipids are fatty substances which may be naturally occurring or synthetic lipids.
- lipids include the fatty droplets that naturally occur in the cytoplasm as well as the class of compounds which contain long-chain aliphatic hydrocarbons and their derivatives, such as fatty acids, alcohols, amines, amino alcohols, and aldehydes.
- DMPC dimyristyl phosphatidylcholine
- DCP dicetyl phosphate
- Choi cholesterol
- DMPG dimyristyl phosphatidylglycerol
- Stock solutions of lipids in chloroform or chloroform/methanol can be stored at about -20 °C. Chloroform is used as the only solvent since it is more readily evaporated than methanol.
- Liposome is a generic term encompassing a variety of single and multilamellar lipid vehicles formed by the generation of enclosed lipid bilayers or aggregates. Liposomes can be characterized as having vesicular structures with a phospholipid bilayer membrane and an inner aqueous medium. Multilamellar liposomes have multiple lipid layers separated by aqueous medium. They form spontaneously when phospholipids are suspended in an excess of aqueous solution. The lipid components undergo self-rearrangement before the formation of closed structures and entrap water and dissolved solutes between the lipid bilayers (Ghosh et al., 1991 Glycobiology 5: 505-10).
- compositions that have different structures in solution than the normal vesicular structure are also encompassed.
- the lipids may assume a micellar structure or merely exist as nonuniform aggregates of lipid molecules.
- lipofectamine-nucleic acid complexes are also contemplated.
- Such assays include, for example, “molecular biological” assays well known to those of skill in the art, such as Southern and Northern blotting, RT-PCR and PCR; “biochemical” assays, such as detecting the presence or absence of a particular peptide, e.g., by immunological means (ELISAs and western blots) or by assays described herein to identify agents falling within the scope of the present disclosure.
- the present disclosure further provides a vector comprising a nucleic acid molecule encoding a TFP as described herein and a switch polypeptide or a dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- a vector encoding a TFP as described herein and a switch or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein can be directly transduced into a cell, e.g., a T cell.
- the vector is a cloning or expression vector, e.g., a vector including, but not limited to, one or more plasmids (e.g., expression plasmids, cloning vectors, minicircles, minivectors, double minute chromosomes), retroviral and lentiviral vector constructs.
- the vector is capable of expressing the TFP construct as described herein and a switch polypeptide or a dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein in mammalian T cells.
- the mammalian T cell is a human T cell.
- Expression vectors include: a promoter (e.g., an EF1a promoter), a signal sequence to enable secretion, a polyadenylation signal and transcription terminator (Bovine Growth Hormone (BGH) gene), an element allowing episomal replication and replication in prokaryotes (e.g., SV40 origin and ColE1 or others known in the art) and elements to allow selection (ampicillin resistance gene and zeocin marker).
- a promoter e.g., an EF1a promoter
- a signal sequence to enable secretion e.g., a polyadenylation signal and transcription terminator (Bovine Growth Hormone (BGH) gene
- BGH Bovine Growth Hormone
- the TFP-encoding nucleic acid construct as described herein with or without a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein can be cloned into a lentiviral expression vector and expression validated based on the quantity and quality of the effector T cell response of transduced T cells in response to MSLN+ target cells.
- Effector T cell responses include, but are not limited to, cellular expansion, proliferation, doubling, cytokine production and target cell lysis or cytolytic activity (i.e., degranulation).
- RNAs [0526] Disclosed herein are methods for producing in vitro transcribed RNA encoding TFPs as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the present disclosure also includes a TFP as described herein encoding RNA construct and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein that can be directly transfected into a cell.
- a method for generating mRNA for use in transfection can involve in vitro transcription (IVT) of a template with specially designed primers, followed by polyA addition, to produce a construct containing 3’ and 5’ untranslated sequence (“UTR”), a 5’ cap and/or Internal Ribosome Entry Site (IRES), the nucleic acid to be expressed, and a polyA tail, typically 50-2000 bases in length (SEQ ID NO: 407).
- RNA so produced can efficiently transfect different kinds of cells.
- the template includes sequences for the TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein as described herein.
- the anti-TAA TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein is encoded by a messenger RNA (mRNA).
- mRNA messenger RNA
- the mRNA encoding the anti-TAA TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein is introduced into a T cell for production of a T cell expressing the TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the in vitro transcribed RNA encoding a TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein can be introduced to a cell as a form of transient transfection.
- the RNA is produced by in vitro transcription using a polymerase chain reaction (PCR)-generated template.
- DNA of interest from any source can be directly converted by PCR into a template for in vitro mRNA synthesis using appropriate primers and RNA polymerase.
- the source of the DNA can be, for example, genomic DNA, plasmid DNA, phage DNA, cDNA, synthetic DNA sequence or any other appropriate source of DNA.
- the desired template for in vitro transcription is a TFP and/or a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof of the present disclosure.
- the DNA to be used for PCR contains an open reading frame.
- the DNA can be from a naturally occurring DNA sequence from the genome of an organism.
- the nucleic acid can include some or all of the 5’ and/or 3’ untranslated regions (UTRs).
- the nucleic acid can include exons and introns.
- the DNA to be used for PCR is a human nucleic acid sequence.
- the DNA to be used for PCR is a human nucleic acid sequence including the 5’ and 3’ UTRs.
- the DNA can alternatively be an artificial DNA sequence that is not normally expressed in a naturally occurring organism.
- An exemplary artificial DNA sequence is one that contains portions of genes that are ligated together to form an open reading frame that encodes a fusion protein.
- the portions of DNA that are ligated together can be from a single organism or from more than one organism.
- PCR can be used to generate a template for in vitro transcription of mRNA which is used for transfection. Methods for performing PCR are well known in the art. Primers for use in PCR are designed to have regions that are substantially complementary to regions of the DNA to be used as a template for the PCR.
- “Substantially complementary,” as used herein, refers to sequences of nucleotides where a majority or all of the bases in the primer sequence are complementary, or one or more bases are non-complementary, or mismatched. Substantially complementary sequences are able to anneal or hybridize with the intended DNA target under annealing conditions used for PCR.
- the primers can be designed to be substantially complementary to any portion of the DNA template. For example, the primers can be designed to amplify the portion of a nucleic acid that is normally transcribed in cells (the open reading frame), including 5’ and 3’ UTRs. The primers can also be designed to amplify a portion of a nucleic acid that encodes a particular domain of interest.
- the primers are designed to amplify the coding region of a human cDNA, including all or portions of the 5’ and 3’ UTRs.
- Primers useful for PCR can be generated by synthetic methods that are well known in the art.
- “Forward primers” are primers that contain a region of nucleotides that are substantially complementary to nucleotides on the DNA template that are upstream of the DNA sequence that is to be amplified.
- Upstream is used herein to refer to a location 5, to the DNA sequence to be amplified relative to the coding strand.
- “Reverse primers” are primers that contain a region of nucleotides that are substantially complementary to a double-stranded DNA template that are downstream of the DNA sequence that is to be amplified.
- Downstream is used herein to refer to a location 3’ to the DNA sequence to be amplified relative to the coding strand.
- Any DNA polymerase useful for PCR can be used in the methods as described herein. The reagents and polymerase are commercially available from a number of sources.
- Chemical structures with the ability to promote stability and/or translation efficiency may also be used.
- the RNA preferably has 5’ and 3’ UTRs. In one embodiment, the 5’ UTR is between one and 3000 nucleotides in length. The length of 5’ and 3’ UTR sequences to be added to the coding region can be altered by different methods, including, but not limited to, designing primers for PCR that anneal to different regions of the UTRs.
- the 5’ and 3’ UTRs can be the naturally occurring, endogenous 5’ and 3’ UTRs for the nucleic acid of interest.
- UTR sequences that are not endogenous to the nucleic acid of interest can be added by incorporating the UTR sequences into the forward and reverse primers or by any other modifications of the template.
- the use of UTR sequences that are not endogenous to the nucleic acid of interest can be useful for modifying the stability and/or translation efficiency of the RNA.
- 3’ UTRs can be selected or designed to increase the stability of the transcribed RNA based on properties of UTRs that are well known in the art.
- the 5’ UTR can contain the Kozak sequence of the endogenous nucleic acid.
- a consensus Kozak sequence can be redesigned by adding the 5’ UTR sequence.
- Kozak sequences can increase the efficiency of translation of some RNA transcripts but do not appear to be required for all RNAs to enable efficient translation.
- the 5’ UTR can be 5’UTR of an RNA virus whose RNA genome is stable in cells.
- various nucleotide analogues can be used in the 3’ or 5’ UTR to impede exonuclease degradation of the mRNA.
- the RNA polymerase promoter When a sequence that functions as a promoter for an RNA polymerase is added to the 5’ end of the forward primer, the RNA polymerase promoter becomes incorporated into the PCR product upstream of the open reading frame that is to be transcribed.
- the promoter is a T7 polymerase promoter, as described elsewhere herein.
- Other useful promoters include, but are not limited to, T3 and SP6 RNA polymerase promoters. Consensus nucleotide sequences for T7, T3 and SP6 promoters are known in the art.
- the mRNA has both a cap on the 5’ end and a 3’ poly(A) tail which determine ribosome binding, initiation of translation and stability mRNA in the cell.
- a circular DNA template for instance, plasmid DNA
- RNA polymerase produces a long concatemeric product which is not suitable for expression in eukaryotic cells.
- the transcription of plasmid DNA linearized at the end of the 3’ UTR results in normal sized mRNA which is not effective in eukaryotic transfection even if it is polyadenylated after transcription.
- phage T7 RNA polymerase can extend the 3’ end of the transcript beyond the last base of the template (Schenborn and Mierendorf, Nuc Acids Res., 13:6223-36 (1985); Nacheva and Berzal-Herranz, Eur. J. Biochem., 270:1485-65 (2003).
- the conventional method of integration of polyA/T stretches into a DNA template is molecular cloning.
- polyA/T sequence integrated into plasmid DNA can cause plasmid instability, which is why plasmid DNA templates obtained from bacterial cells are often highly contaminated with deletions and other aberrations. This makes cloning procedures not only laborious and time consuming but often not reliable.
- the polyA/T segment of the transcriptional DNA template can be produced during PCR by using a reverse primer containing a poly-T tail, such as 100 T tail (SEQ ID NO: 408) (size can be 50-5000 T (SEQ ID NO: 409)), or after PCR by any other method, including, but not limited to, DNA ligation or in vitro recombination.
- Poly(A) tails also provide stability to RNAs and reduce their degradation. Generally, the length of a poly(A) tail positively correlates with the stability of the transcribed RNA.
- the poly(A) tail is between 100 and 5000 adenosines (SEQ ID NO: 410).
- Poly(A) tails of RNAs can be further extended following in vitro transcription with the use of a poly(A) polymerase, such as E. coli polyA polymerase (E-PAP).
- E-PAP E. coli polyA polymerase
- increasing the length of a poly(A) tail from 100 nucleotides to between 300 and 400 nucleotides (SEQ ID NO: 411) results in about a two-fold increase in the translation efficiency of the RNA.
- the attachment of different chemical groups to the 3’ end can increase mRNA stability. Such attachment can contain modified/artificial nucleotides, aptamers and other compounds.
- RNAs produced by the methods as described herein include a 5’ cap.
- the 5’ cap is provided using techniques known in the art and described herein (Cougot, et al., Trends in Biochem. Sci., 29:436-444 (2001); Stepinski, et al., RNA, 7:1468-95 (2001); Elango, et al., Biochim. Biophys. Res. Commun., 330:958-966 (2005)).
- RNAs produced by the methods as described herein can also contain an internal ribosome entry site (IRES) sequence.
- IRES sequence may be any viral, chromosomal or artificially designed sequence which initiates cap-independent ribosome binding to mRNA and facilitates the initiation of translation. Any solutes suitable for cell electroporation, which can contain factors facilitating cellular permeability and viability such as sugars, peptides, lipids, proteins, antioxidants, and surfactants can be included.
- RNA can be introduced into target cells using any of a number of different methods, for instance, commercially available methods which include, but are not limited to, electroporation (Amaxa Nucleofector®-II (Amaxa Biosystems, Cologne, Germany)), ECM 830 (BTX) (Harvard Instruments, Boston, Mass.) or the Gene Pulser® II (BioRad, Denver, Colo.), Multiporator® (Eppendorf, Hamburg Germany), cationic liposome mediated transfection using lipofection, polymer encapsulation, peptide mediated transfection, or biolistic particle delivery systems such as “gene guns” (see, for example, Nishikawa, et al.
- cells comprising the recombinant nucleic acid as described herein, the polypeptide as described herein, or the vectors as described herein.
- cells comprising the recombinant nucleic acid as described herein, the polypeptide as described herein, or the vectors as described herein, wherein cells comprise the sequence encoding a TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the cell is a T cell.
- the T cell is a human T cell. In some embodiments, the T cell is a CD8+ or CD4+ T cell. In some embodiments, the T cell is a human ⁇ T cell. In some embodiments, the T cell is a human ⁇ T cell. In some embodiments, the cell is a human NKT cell. In some embodiments, the cell is an allogeneic cell or an autologous cell. In some embodiments, the T cell is modified to comprise a functional disruption of the TCR. In some embodiments, the modified T cells are ⁇ T cells and do not comprise a functional disruption of an endogenous TCR. In some embodiments, the ⁇ T cells are V 1+ V 2- T cells.
- the ⁇ T cells are V 1- V 2+ T cells. In some embodiments, the ⁇ T cells are V 1- V 2- T cells.
- the present disclosure provides genetically-modified immune cells and populations thereof and methods for producing the same.
- the genetically-modified immune cells of the presently disclosed compositions and methods are human immune cells.
- the immune cells are T cells, or cells derived therefrom.
- the immune cells are natural killer (NK) cells, or cells derived therefrom.
- the immune cells are B cells, or cells derived therefrom.
- the immune cells are monocyte or macrophage cells or cells derived therefrom.
- cells comprising the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, wherein the cells may have enhanced survival rate, enhanced effector function, and/or enhanced cytotoxicity compared to cells that do not comprise the sequence encoding TFP as described herein, and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the cell has enhanced survival rate compared to a cell that does not have a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In some embodiments, the cell has enhanced effector function compared to a cell that does not have a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In some embodiments, the cell has enhanced cytotoxicity compared to a cell that does not have a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- cells comprising the sequence encoding TFP as described herein, and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof, wherein the cells as described herein may have increased longevity compared to cells that do not comprise the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the longevity of the cell is increased compared to a cell that does not comprise a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- cells comprising the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, wherein the cells may have increased persistence compared to cells that do not comprise the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the persistence of the cell is increased compared to a cell that does not comprise a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof.
- cells comprising the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, wherein the cells may have increased cytotoxicity compared to cells that do not comprise the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the cytotoxicity of the cell is increased compared to a cell that does not comprise a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- cells comprising the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, wherein the cells may have increased cytokine production compared to cells that do not comprise the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the cytokine production of the cell is increased compared to a cell that does not comprise a nucleic acid sequence encoding herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- a cell that does not comprise a nucleic acid sequence encoding herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- cells comprising the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, wherein the cells have increased ability to resist, blunt, and/or overcome TGF ⁇ - mediated suppression of T cell function.
- the cells have increased ability to resist, blunt, and/or overcome TGF ⁇ -mediated suppression of cytokine production (e.g., IFN ⁇ , IL-2, GM-CSF, and/or TNF ⁇ ) and/or TGF ⁇ -mediated suppression of cytotoxicity and/or TGF ⁇ -mediated exhaustion.
- cytokine production e.g., IFN ⁇ , IL-2, GM-CSF, and/or TNF ⁇
- TGF ⁇ -mediated suppression of cytotoxicity and/or TGF ⁇ -mediated exhaustion e.g., IFN ⁇ , IL-2, GM-CSF, and/or TNF ⁇
- the ability to resist, blunt, and/or overcome TGF ⁇ -mediated effects is as compared to a cell that does not comprise a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, for example, is as compared to a cell that comprises the same TFP as the cell comprising the TGFBr2 switch polypeptide or TGFBr2 dominant negative polypeptide but that lacks a TGFBr2 switch polypeptide and TGFBr2 dominant negative polypeptide.
- cells as described herein retain na ⁇ ve and/or central memory phenotypes.
- cells as described herein have not differentiated into terminal effector cells. In some embodiments, the cells as described herein expand in the presence of TGF ⁇ .
- a population of cells comprising any of the cell described herein.
- a population of cells comprising any of the cell described herein, wherein the population of cells has an increased proportion of cells having a central memory phenotype relative to a population of cells that do not comprise the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the population of cells has an increased proportion of cells having a central memory phenotype relative to a population of cells that do not comprise a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- population of cells comprising any of the cell described herein, wherein the population of cells has an increased proportion of cells having a na ⁇ ve phenotype relative to a population of cells that do not comprise the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the population of cells has an increased proportion of cells having a na ⁇ ve phenotype relative to a population of cells that do not comprise a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- population of cells comprising any of the cell described herein, wherein the population of cells has a reduced proportion of cells having a terminal effector phenotype relative to a population of cells that do not comprise the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the population of cells has a reduced proportion of cells having a terminal effector phenotype relative to a population of cells that do not comprise a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the population of cells comprising any of the cells described herein has a similar phenotype (e.g., CD4:CD8 ratio, proportion of cells having a central memory phenotype, proportion of cells having a na ⁇ ve phenotype, and/or proportion of cells having a terminal effector phenotype) as a population of cells that do not comprise a nucleic acid encoding a TGFBr2 switch polypeptide or TGFBR2 dominant negative polypeptide, but exhibits increased survival rate, effector function, cytotoxicity, longevity, persistence, and/or cytokine production, compared to a population of cells that do not comprise a nucleic acid encoding a TGFBr2 switch polypeptide or TGFBr2 dominant negative polypeptide.
- a similar phenotype e.g., CD4:CD8 ratio, proportion of cells having a central memory phenotype, proportion of cells having a na ⁇ ve phenotype, and/or proportion of cells having a terminal
- modified immune cells e.g., T cells comprising the recombinant nucleic acid as described herein, or the vectors as described herein.
- the modified T cell further comprises a functional disruption of an endogenous TCR.
- modified T cells comprising the sequence encoding the TFP of the nucleic acid as described herein or a TFP encoded by the sequence of the nucleic acid as described herein and a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the modified T cell further comprises a functional disruption of an endogenous TCR.
- modified allogenic T cells comprising the sequence encoding the TFP as described herein or a TFP encoded by the sequence of the nucleic acid as described herein and a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- modified T cell comprising the recombinant nucleic acid disclosed above, or the vector disclosed above; wherein the modified T cell further comprises a functional disruption of an endogenous TCR.
- modified T cells comprising the sequence encoding the TFP of the nucleic acid disclosed above or a TFP encoded by the sequence of the nucleic acid disclosed above encoding the TFP as described herein and a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, wherein the modified T cell further comprises a functional disruption of an endogenous TCR.
- modified allogenic T cell comprising the sequence encoding the TFP disclosed above or a TFP encoded by the sequence of the nucleic acid disclosed above encoding the TFP as described herein and a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the T cell further comprises a heterologous sequence encoding a TCR constant domain, wherein the TCR constant domain is a TCR alpha constant domain, a TCR beta constant domain, a TCR alpha constant domain and a TCR beta constant domain, a TCR gamma constant domain, a TCR delta constant domain or a TCR gamma constant domain and a TCR delta constant domain.
- the TCR constant domain is a TCR alpha constant domain, a TCR beta constant domain, a TCR alpha constant domain and a TCR beta constant domain, a TCR gamma constant domain, a TCR delta constant domain or a TCR gamma constant domain and a TCR delta constant domain.
- the endogenous TCR that is functionally disrupted is an endogenous TCR alpha chain, an endogenous TCR beta constant domain, an endogenous TCR alpha constant domain and an endogenous TCR beta constant domain, an endogenous TCR gamma chain, an endogenous TCR delta chain, or an endogenous TCR gamma chain and an endogenous TCR delta chain.
- the endogenous TCR that is functionally disrupted has reduced binding to MHC-peptide complex compared to that of an unmodified control T cell.
- the functional disruption is a disruption of a gene encoding the endogenous TCR.
- the disruption of a gene encoding the endogenous TCR is a removal of a sequence of the gene encoding the endogenous TCR from the genome of a T cell.
- the T cell is a CD8+ T cell, a CD4+ T cell, a na ⁇ ve T cell, a memory stem T cell, a central memory T cell, a double negative T cell, an effector memory T cell, an effector T cell, a ThO cell, a TcO cell, a Th1 cell, a Tc1 cell, a Th2 cell, a Tc2 cell, a Th17 cell, a Th22 cell, a gamma delta T cell, a natural killer (NK) cell, a natural killer T (NKT) cell, a hematopoietic stem cell, or a pluripotent stem cell.
- NK natural killer
- NKT natural killer T
- the T cell is a human T cell. In some instances, the T cell is a CD8+ or CD4+ T cell. In some embodiments, the T cell is a CD4+CD8+ T cell. In some embodiments, the T cell is a human ⁇ T cell. In some embodiments, the T cell is a human ⁇ T cell. In some embodiments, the cell is a human NKT cell. In some embodiments, the cell is an allogeneic cell or an autologous cell. In some embodiments, the T cell is modified to comprise a functional disruption of the TCR. In some embodiments, the modified T cells are ⁇ T cells and do not comprise a functional disruption of an endogenous TCR.
- the ⁇ T cells are V 1+ V 2- T cells. In some embodiments, the ⁇ T cells are V 1- V 2+ T cells. In some embodiments, the ⁇ T cells are V 1- V 2- T cells. In some instances, the T cell is an allogenic T cell.
- the modified T cells further comprise a nucleic acid encoding an inhibitory molecule that comprises a first polypeptide comprising at least a portion of an inhibitory molecule, associated with a second polypeptide comprising a positive signal from an intracellular signaling domain. In some instances, the inhibitory molecule comprises the first polypeptide comprising at least a portion of PD-1 and the second polypeptide comprising a costimulatory domain and primary signaling domain.
- a T cell expressing the TFP as descried herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as descried herein can inhibit tumor growth when expressed in a T cell.
- genetically-modified immune cells of the invention comprise an inactivated TCR alpha gene and/or an inactivated TCR beta gene. Inactivation of the TCR alpha gene and/or TCR beta gene to generate the genetically-modified cells of the present disclosure occurs in at least one or both alleles where the TCR alpha gene and/or TCR beta gene is being expressed.
- inactivation of one or both genes prevents expression of the endogenous TCR alpha chain or the endogenous TCR beta chain protein. Expression of these proteins is required for assembly of the endogenous alpha/beta TCR on the cell surface.
- inactivation of the TCR alpha gene and/or the TCR beta gene results in genetically-modified immune that have no detectable cell surface expression of the endogenous alpha/beta TCR.
- the inactivated gene is a TCR alpha constant region (TRAC) gene.
- genetically-modified immune cells of the invention comprise an inactivated B2M gene.
- the TCR alpha gene, the TRAC gene, or the TCR beta gene is inactivated by insertion of a template nucleic acid into a cleavage site in the gene. Insertion of the template nucleic acid disrupts expression of the endogenous TCR alpha chain or TCR beta chain and, therefore, prevents assembly of an endogenous alpha/beta TCR on the T cell surface.
- the template nucleic acid is inserted into the TRAC gene.
- the genetically-modified immune cell expresses a TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, such cells have no detectable cell-surface expression of an endogenous T cell receptor (e.g., an alpha/beta T cell receptor).
- an endogenous T cell receptor e.g., an alpha/beta T cell receptor
- the disclosure further provides a population of genetically-modified immune cells that express a TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein and have no detectable cell- surface expression of an endogenous T cell receptor (e.g., an alpha/beta T cell receptor), and in some embodiments also express a TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- an endogenous T cell receptor e.g., an alpha/beta T cell receptor
- the population can include a plurality of genetically-modified immune cells of the disclosure which express a TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, and have no cell- surface expression of an endogenous T cell receptor (i.e., are TCR-).
- “detectable cell-surface expression of an endogenous TCR” refers to the ability to detect one or more components of the TCR complex (e.g., an alpha/beta TCR complex) on the cell surface of an immune cell using standard experimental methods.
- Such methods can include, for example, immunostaining and/or flow cytometry specific for components of the TCR itself, such as a TCR alpha or TCR beta chain, or for components of the assembled cell-surface TCR complex, such as CD3.
- Methods for detecting cell-surface expression of an endogenous TCR (e.g., an alpha/beta TCR) on an immune cell include those described in the examples herein, and, for example, those described in MacLeod et al. (2017) Molecular Therapy 25(4): 949-961.
- the present disclosure provides a modified allogenic immune cell, e.g., T cell comprising the sequence encoding the TFP described herein or a TFP encoded by the sequence of the nucleic acid as described herein encoding the TFP and the sequence encoding the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- Sources of T cells e.g., T cell comprising the sequence encoding the TFP described herein or a TFP encoded by the sequence of the nucleic acid as described herein encoding the TFP and the sequence encoding the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- Sources of T cells Prior to expansion and genetic modification, a source of T cells is obtained from a subject. Examples of subjects include humans, dogs, cats, mice, rats, and transgenic species thereof.
- T cells can be obtained from a number of sources, including peripheral blood mononuclear cells, bone marrow, lymph node tissue, cord blood, thymus tissue, tissue from a site of infection, ascites, pleural effusion, spleen tissue, and tumors. In certain aspects of the present disclosure, any number of T cell lines available in the art, may be used. In certain aspects of the present disclosure, T cells can be obtained from a unit of blood collected from a subject using any number of techniques known to the skilled artisan, such as Ficoll TM separation. In one preferred aspect, cells from the circulating blood of an individual are obtained by apheresis.
- the apheresis product typically contains lymphocytes, including T cells, monocytes, granulocytes, B cells, other nucleated white blood cells, red blood cells, and platelets.
- the cells collected by apheresis may be washed to remove the plasma fraction and to place the cells in an appropriate buffer or media for subsequent processing steps.
- the cells are washed with phosphate buffered saline (PBS).
- PBS phosphate buffered saline
- the wash solution lacks calcium and may lack magnesium or may lack many if not all divalent cations. Initial activation steps in the absence of calcium can lead to magnified activation.
- a washing step may be accomplished by methods known to those in the art, such as by using a semi-automated “flow-through” centrifuge (for example, the Cobe® 2991 cell processor, the Baxter Oncology CytoMateTM, or the Haemonetics® Cell Saver® 5) according to the manufacturer’s instructions.
- a semi-automated “flow-through” centrifuge for example, the Cobe® 2991 cell processor, the Baxter Oncology CytoMateTM, or the Haemonetics® Cell Saver® 5
- the cells may be resuspended in a variety of biocompatible buffers, such as, for example, Ca-free, Mg-free PBS, PlasmaLyte A, or other saline solution with or without buffer.
- the undesirable components of the apheresis sample may be removed, and the cells directly resuspended in culture media.
- T cells are isolated from peripheral blood lymphocytes by lysing the red blood cells and depleting the monocytes, for example, by centrifugation through a PERCOLL ® gradient or by counterflow centrifugal elutriation.
- a specific subpopulation of T cells such as CD3+, CD28+, CD4+, CD8+, CD45RA+, and CD45RO+ T cells, can be further isolated by positive or negative selection techniques.
- T cells are isolated by incubation with anti-CD3/anti-CD28 (e.g., 3x28)-conjugated beads, such as DYNABEADS ® M-450 CD3/CD28 T, for a time period sufficient for positive selection of the desired T cells.
- the time period is about 30 minutes.
- the time period ranges from 30 minutes to 36 hours or longer and all integer values there between.
- the time period is at least 1, 2, 3, 4, 5, or 6 hours.
- the time period is 10 to 24 hours.
- the incubation time period is 24 hours.
- TIL tumor infiltrating lymphocytes
- subpopulations of T cells can be preferentially selected for or against at culture initiation or at other desired time points.
- multiple rounds of selection can also be used in the context of this present disclosure.
- Unselected” cells can also be subjected to further rounds of selection.
- Enrichment of a T cell population by negative selection can be accomplished with a combination of antibodies directed to surface markers unique to the negatively selected cells.
- One method is cell sorting and/or selection via negative magnetic immunoadherence or flow cytometry that uses a cocktail of monoclonal antibodies directed to cell surface markers present on the cells negatively selected.
- a monoclonal antibody cocktail typically includes antibodies to CD14, CD20, CD11b, CD16, HLA-DR, and CD8.
- T regulatory cells are depleted by anti- C25 conjugated beads or other similar method of selection.
- a T cell population can be selected that expresses one or more of IFN- ⁇ TNF-alpha, IL-17A, IL-2, IL-3, IL-4, GM-CSF, IL-10, IL-13, granzyme B, and perforin, or other appropriate molecules, e.g., other cytokines.
- Methods for screening for cell expression can be determined, e.g., by the methods described in PCT Publication No.: WO 2013/126712.
- the concentration of cells and surface e.g., particles such as beads
- the concentration of cells and surface can be varied.
- a concentration of 2 billion cells/mL is used.
- a concentration of 1 billion cells/mL is used.
- greater than 100 million cells/mL is used.
- a concentration of cells of 10, 15, 20, 25, 30, 35, 40, 45, or 50 million cells/mL is used.
- a concentration of cells from 75, 80, 85, 90, 95, or 100 million cells/mL is used.
- concentrations of 125 or 150 million cells/mL can be used.
- Using high concentrations can result in increased cell yield, cell activation, and cell expansion. Further, use of high cell concentrations allows more efficient capture of cells that may weakly express target antigens of interest, such as CD28-negative T cells, or from samples where there are many tumor cells present (e.g., leukemic blood, tumor tissue, etc.). Such populations of cells may have therapeutic value and would be desirable to obtain. For example, using high concentration of cells allows more efficient selection of CD8+ T cells that normally have weaker CD28 expression. [0569] In a related aspect, it may be desirable to use lower concentrations of cells. By significantly diluting the mixture of T cells and surface (e.g., particles such as beads), interactions between the particles and cells are minimized.
- target antigens of interest such as CD28-negative T cells
- CD4+ T cells express higher levels of CD28 and are more efficiently captured than CD8+ T cells in dilute concentrations.
- the concentration of cells used is 5x10 6 /mL. In other aspects, the concentration used can be from about 1x10 5 /mL to 1x10 6 /mL, and any integer value in between.
- the cells may be incubated on a rotator for varying lengths of time at varying speeds at either 2-10 °C or at room temperature. [0570] T cells for stimulation can also be frozen after a washing step.
- the freeze and subsequent thaw step provides a more uniform product by removing granulocytes and to some extent monocytes in the cell population.
- the cells may be suspended in a freezing solution.
- one method involves using PBS containing 20% DMSO and 8% human serum albumin, or culture media containing 10% Dextran 40 and 5% Dextrose, 20% Human Serum Albumin and 7.5% DMSO, or 31.25% Plasmalyte-A, 31.25% Dextrose 5%, 0.45% NaCl, 10% Dextran 40 and 5% Dextrose, 20% Human Serum Albumin, and 7.5% DMSO or other suitable cell freezing media containing for example, Hespan and PlasmaLyte A, the cells then are frozen to -80 °C at a rate of 1 per minute and stored in the vapor phase of a liquid nitrogen storage tank.
- cryopreserved cells are thawed and washed as described herein and allowed to rest for one hour at room temperature prior to activation using the methods of the present disclosure.
- the collection of blood samples or apheresis product from a subject at a time period prior to when the expanded cells as described herein might be needed.
- the source of the cells to be expanded can be collected at any time point necessary, and desired cells, such as T cells, isolated and frozen for later use in T cell therapy for any number of diseases or conditions that would benefit from T cell therapy, such as those described herein.
- a blood sample or an apheresis is taken from a generally healthy subject.
- a blood sample or an apheresis is taken from a generally healthy subject who is at risk of developing a disease, but who has not yet developed a disease, and the cells of interest are isolated and frozen for later use.
- the T cells may be expanded, frozen, and used at a later time.
- samples are collected from a patient shortly after diagnosis of a particular disease as described herein but prior to any treatments.
- the cells are isolated from a blood sample or an apheresis from a subject prior to any number of relevant treatment modalities, including but not limited to treatment with agents such as natalizumab, efalizumab, antiviral agents, chemotherapy, radiation, immunosuppressive agents such as cyclosporin, azathioprine, methotrexate, and mycophenolate, antibodies, or other immunoablative agents such as alemtuzumab, anti-CD3 antibodies, cytoxan, fludarabine, cyclosporin, tacrolimus, rapamycin, mycophenolic acid, steroids, romidepsin, and irradiation.
- agents such as natalizumab, efalizumab, antiviral agents, chemotherapy, radiation, immunosuppressive agents such as cyclosporin, azathioprine, methotrexate, and mycophenolate, antibodies, or other immunoablative agents such as alemtuzumab, anti-CD3
- T cells are obtained from a patient directly following treatment that leaves the subject with functional T cells.
- the quality of T cells obtained may be optimal or improved for their ability to expand ex vivo.
- these cells may be in a preferred state for enhanced engraftment and in vivo expansion.
- mobilization for example, mobilization with GM-CSF
- conditioning regimens can be used to create a condition in a subject wherein repopulation, recirculation, regeneration, and/or expansion of particular cell types is favored, especially during a defined window of time following therapy.
- Illustrative cell types include T cells, B cells, dendritic cells, and other cells of the immune system.
- Activation and Expansion of T Cells [0573] T cells may be activated and expanded generally using methods as described, for example, in U.S. Pat.
- the T cells of the present disclosure may be expanded by contact with a surface having attached thereto an agent that stimulates a CD3/TCR complex associated signal and a ligand that stimulates a costimulatory molecule on the surface of the T cells.
- T cell populations may be stimulated as described herein, such as by contact with an anti-CD3 antibody, or antigen-binding fragment thereof, or an anti-CD2 antibody immobilized on a surface, or by contact with a protein kinase C activator (e.g., bryostatin) in conjunction with a calcium ionophore.
- a protein kinase C activator e.g., bryostatin
- a ligand that binds the accessory molecule is used for co-stimulation of an accessory molecule on the surface of the T cells.
- a population of T cells can be contacted with an anti-CD3 antibody and an anti-CD28 antibody, under conditions appropriate for stimulating proliferation of the T cells.
- an anti-CD3 antibody and an anti-CD28 antibody To stimulate proliferation of either CD4+ T cells, CD8+ T cells, or CD4+CD8+ T cells, an anti-CD3 antibody and an anti-CD28 antibody.
- an anti-CD28 antibody include 9.3, B-T3, XR-CD28 (Diaclone, Besancon, France) can be used as can other methods commonly known in the art (Berg et al., Transplant Proc.30(8):3975-3977, 1998; Haanen et al., J. Exp. Med. 190(9):13191328, 1999; Garland et al., J. Immunol. Meth.227(1-2):53-63, 1999).
- T cells may additionally be activated and expanded in the presence of a cytokine with or without an anti-CD3 and/or CD28 antibody.
- cytokines include IL-2, IL-7, IL-15, and IL-21.
- T cells are activated by incubation with anti-CD3/anti-CD28- conjugated beads, such as DYNABEADS® or Trans-Act® beads, for a time period sufficient for activation of the T cells.
- the time period is at least 1, 2, 3, 4, 5, or 6 hours.
- the time period is 10 to 24 hours, e.g., 24 hours.
- T cells are activated by stimulation with an anti-CD3 antibody and an anti- CD28 antibody in combination with cytokines that bind the common gamma-chain (e.g., IL- 2, IL-7, IL-12, IL-15, IL-21, and others).
- T cells are activated by stimulation with an anti-CD3 antibody and an anti-CD28 antibody in combination with 10, 20, 30, 40, 50, 60, 70, 80, 90, 100, 200, 300, 400, 500, 600, 700, 800, 900, or 100 U/mL of IL-2, IL-7, and/or IL-15.
- the cells are activated for 24 hours.
- the cells after transduction, are expanded in the presence of anti-CD3 antibody, anti-CD28 antibody in combination with the same cytokines.
- cells activated in the presence of an anti-CD3 antibody and an anti-CD28 antibody in combination with cytokines that bind the common gamma-chain are expanded in the presence of the same cytokines in the absence of the anti-CD3 antibody and anti-CD28 antibody after transduction.
- the cells after transduction, the cells are expanded in the presence of anti-CD3 antibody, anti-CD28 antibody in combination with the same cytokines up to a first washing step, when the cells are sub-cultured in media that includes the cytokines but does not include the anti-CD3 antibody and anti-CD28 antibody.
- the cells are subcultured every 1, 2, 3, 4, 5, or 6 days. In some embodiments, cells are expanded for 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, or 30 days.
- the expansion of T cells may be stimulated with zoledronic acid (Zometa), alendronic acid (Fosamax) or other related bisphosphonate drugs at concentrations of 0.1, 0.25, 0.5, 1.0, 2.0, 3.0, 4.0, 5.0, 7.5, 10, or 100 ⁇ M in the presence of feeder cells (irradiated cancer cells, PBMCs, artificial antigen presenting cells).
- T cells may be stimulated with isopentyl pyrophosphate (IPP), (E)-4-Hydroxy-3-methyl-but-2-enyl pyrophosphate (HMBPP or HMB-PP) or other structurally related compounds at concentrations of 0.1, 0.25, 0.5, 1.0, 2.0, 3.0, 4.0, 5.0, 7.5, 10, or 100 ⁇ M in the presence of feeder cells (irradiated cancer cells, PBMCs, artificial antigen presenting cells).
- IPP isopentyl pyrophosphate
- HMBPP or HMB-PP HMB-4-Hydroxy-3-methyl-but-2-enyl pyrophosphate
- feeder cells irradiated cancer cells, PBMCs, artificial antigen presenting cells.
- the expansion of T cells may be stimulated with synthetic phosphoantigens (e.g., bromohydrin pyrophosphate; BrHPP), 2M3B1 PP, or 2-methyl-3-butenyl-1 -pyrophosphate in the presence of IL-2 for one- to-two weeks.
- the expansion of T cells may be stimulated with immobilized anti-TCRyd (e.g., pan TCRY6) in the presence of IL-2, e.g., for approximately 14 days.
- the expansion of T cells may be stimulated with culture of immobilized anti-CD3 antibodies (e.g., OKT3) in the presence of IL-2.
- the aforementioned culture is maintained for about seven days prior to subculture in soluble anti-CD3, and IL-2.
- T cells that have been exposed to varied stimulation times may exhibit different characteristics.
- typical blood or apheresed peripheral blood mononuclear cell products have a helper T cell population (TH, CD4+) that is greater than the cytotoxic or suppressor T cell population (TC, CD8+).
- TH, CD4+ helper T cell population
- TC cytotoxic or suppressor T cell population
- Ex vivo expansion of T cells by stimulating CD3 and CD28 receptors produces a population of T cells that prior to about days 8-9 consists predominately of TH cells, while after about days 8-9, the population of T cells comprises an increasingly greater population of TC cells.
- infusing a subject with a T cell population comprising predominately of TH cells may be advantageous.
- an antigen-specific subset of TC cells has been isolated it may be beneficial to expand this subset to a greater degree.
- other phenotypic markers vary significantly, but in large part, reproducibly during the course of the cell expansion process. Thus, such reproducibility enables the ability to tailor an activated T cell product for specific purposes.
- anti-CD19, anti-BCMA, anti-CD22, anti-ROR1, anti-PD-1, or anti-BAFFR, anti-MUC16, anti-mesothelin, anti-HER2, anti-PMSA, anti-CD20, anti-CD70, anti-GPC3, anti-Nectin-4, anti-Trop2, or antiCD79b TFP is constructed, various assays can be used to evaluate the activity of the molecule, such as but not limited to, the ability to expand T cells following antigen stimulation, sustain T cell expansion in the absence of re-stimulation, and anti-cancer activities in appropriate in vitro and animal models.
- T cells (1:1 mixture of CD4 + and CD8 + T cells) expressing the TFPs are expanded in vitro for more than 10 days followed by lysis and SDS-PAGE under reducing conditions. TFPs are detected by western blotting using an antibody to a TCR chain. The same T cell subsets are used for SDS-PAGE analysis under non-reducing conditions to permit evaluation of covalent dimer formation.
- TFT as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein following antigen stimulation can be measured by flow cytometry.
- a mixture of CD4 + and CD8 + T cells are stimulated with alphaCD3/alphaCD28 and APCs followed by transduction with lentiviral vectors expressing GFP under the control of the promoters to be analyzed.
- promoters include the CMV IE gene, EF-1alpha, ubiquitin C, or phosphoglycerokinase (PGK) promoters.
- GFP fluorescence is evaluated on day 6 of culture in the CD4+ and/or CD8+ T cell subsets by flow cytometry (see, e.g., Milone et al., Molecular Therapy 17(8): 1453-1464 (2009)).
- a mixture of CD4+ and CD8+ T cells are stimulated with alphaCD3/alphaCD28 coated magnetic beads on day 0 and transduced with TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein on day 1 using, e.g., a bicistronic lentiviral vector expressing TFP as described herein along with eGFP using a 2A ribosomal skipping sequence and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- TAA+ K562 cells K562-TAA
- wild-type K562 cells K562 wild type
- K562 cells expressing hCD32 and 4-1BBL in the presence of anti-CD3 and anti-CD28 antibody (K562-BBL-3/28) following washing.
- Exogenous IL-2 is added to the cultures every other day at 100 IU/mL.
- GFP+ T cells are enumerated by flow cytometry using bead-based counting (see, e.g., Milone et al., Molecular Therapy 17(8): 1453-1464 (2009)).
- T cells expressing TFT as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein in the absence of re-stimulation can also be measured (see, e.g., Milone et al., Molecular Therapy 17(8): 1453-1464 (2009)).
- mean T cell volume (fl) is measured on day 8 of culture using a Coulter Multisizer III particle counter following stimulation with alphaCD3/alphaCD28 coated magnetic beads on day 0, and transduction with the indicated TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein on day 1.
- Animal models can also be used to measure an activity of T cells expressing TFT as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- xenograft model using, e.g., human CD19-specific TFP+ T cells co-expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein to treat a primary human pre-B ALL in immunodeficient mice can be used (see, e.g., Milone et al., Molecular Therapy 17(8): 1453-1464 (2009)).
- mice are randomized as to treatment groups. Different numbers of engineered T cells are coinjected at a 1:1 ratio into NOD/SCID/ ⁇ -/- mice bearing B-ALL. The number of copies of each vector in spleen DNA from mice is evaluated at various times following T cell injection.
- Peripheral blood CD19+ B-ALL blast cell counts are measured in mice that are injected with alphaCD19-zeta TFP+ T cells co-expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein or mock-transduced T cells. Survival curves for the groups are compared using the log-rank test. In addition, absolute peripheral blood CD4+ and CD8+ T cell counts 4 weeks following T cell injection in NOD/SCID/ ⁇ -/- mice can also be analyzed.
- mice are injected with leukemic cells and 3 weeks later are injected with T cells engineered to express TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein by, e.g., a bicistronic lentiviral vector that encodes the TFP as described herein linked to eGFP and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein by, e.g., a bicistronic lentiviral vector that encodes the TFP as described herein linked to eGFP and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- T cells are normalized to 45-50% input GFP+ T cells co-expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein by mixing with mock-transduced cells prior to injection and confirmed by flow cytometry. Animals are assessed for leukemia at 1-week intervals. Survival curves for the groups of TFP+ T cell co-expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein are compared using the log-rank test.
- Dose dependent TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein treatment response can be evaluated (see, e.g., Milone et al., Molecular Therapy 17(8): 1453-1464 (2009)).
- peripheral blood is obtained 35- 70 days after establishing leukemia in mice injected on day 21 with TFP T cells co- expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, an equivalent number of mock-transduced T cells, or no T cells.
- mice from each group are randomly bled for determination of peripheral blood CD19+ ALL blast counts and then killed on days 35 and 49. The remaining animals are evaluated on days 57 and 70. [0584] Assessment of cell proliferation and cytokine production has been previously described, e.g., at Milone et al., Molecular Therapy 17(8): 1453-1464 (2009).
- TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein-mediated proliferation is performed in microtiter plates by mixing washed T cells with K562 cells expressing the tumor associated antigen (TAA, e.g., CD19) CD19 (K19) or CD32 and CD137 (KT32-BBL) for a final T cell:K562 ratio of 2:1. K562 cells are irradiated with gamma-radiation prior to use.
- TAA tumor associated antigen
- K19 CD19
- KT32-BBL CD137
- Anti-CD3 (clone OKT3) and anti-CD28 (clone 9.3) monoclonal antibodies are added to cultures with KT32-BBL cells to serve as a positive control for stimulating T cell proliferation since these signals support long-term CD8+ T cell expansion ex vivo.
- T cells are enumerated in cultures using CountBright TM fluorescent beads (Invitrogen) and flow cytometry as described by the manufacturer.
- TFP+ T cells co- expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein are identified by GFP expression using T cells that are engineered with eGFP-2A linked TFP-expressing lentiviral vectors.
- TFP+ T cells co-expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein but not expressing GFP
- the TFP+ T cells co-expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein are detected with biotinylated recombinant CD19 protein and a secondary avidin-PE conjugate.
- CD4+ and CD8+ expression on T cells are also simultaneously detected with specific monoclonal antibodies (BD Biosciences).
- Cytokine measurements are performed on supernatants collected 24 hours following re- stimulation using the human TH1/TH2 cytokine cytometric bead array kit (BD Biosciences) according the manufacturer’s instructions. Fluorescence is assessed using a FACScaliburTM flow cytometer (BD Biosciences), and data are analyzed according to the manufacturer’s instructions. [0585] Cytotoxicity can be assessed by a standard 51 Cr-release assay (see, e.g., Milone et al., Molecular Therapy 17(8): 1453-1464 (2009)).
- Target cells K562 lines and primary pro-B- ALL cells are loaded with 51 Cr (as NaCrO4, New England Nuclear) at 37 °C for 2 hours with frequent agitation, washed twice in complete RPMI and plated into microtiter plates. Effector T cells are mixed with target cells in the wells in complete RPMI at varying ratios of effector cell:target cell (E:T). Additional wells containing media only (spontaneous release, SR) or a 1% solution of Triton-X 100 detergent (total release, TR) are also prepared. After 4 hours of incubation at 37 °C, supernatant from each well is harvested.
- 51 Cr as NaCrO4, New England Nuclear
- % Lysis (ER-SR)/(TR-SR), where ER represents the average 51 Cr released for each experimental condition.
- Imaging technologies can be used to evaluate specific trafficking and proliferation of TFPs in tumor-bearing animal models. Such assays have been described, e.g., in Barrett et al., Human Gene Therapy 22:1575-1586 (2011).
- NOD/SCID/ ⁇ c-/- (NSG) mice are injected IV with Nalm-6 cells (ATCC® CRL-3273TM) followed 7 days later with T cells 4 hour after electroporation with the TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein constructs.
- the T cells are stably transfected with a lentiviral construct to express firefly luciferase, and mice are imaged for bioluminescence.
- therapeutic efficacy and specificity of a single injection of TFP+ T cells co- expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein in Nalm-6 xenograft model can be measured as the following: NSG mice are injected with Nalm-6 transduced to stably express firefly luciferase, followed by a single tail-vein injection of T cells electroporated with a TAA-TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein 7 days later. Animals are imaged at various time points post injection.
- photon-density heat maps of firefly luciferase positive leukemia in representative mice at day 5 (2 days before treatment) and day 8 (24 hours post TFP+ PBLs) can be generated.
- Other assays, including those described herein as well as those that are known in the art can also be used to evaluate the anti-CD19, anti-BCMA, anti-CD22, anti-MSLN, anti- CD79B, anti-GPC3, anti-Nectin-4, anti-Trop2, anti-IL13Ra2, anti-PD-1, anti-ROR1, anti-PD- L1, or anti-BAFFR TFP constructs as described herein.
- nucleic acid molecules comprising a first sequence encoding a TFP as described herein and a second sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, and a third nucleic acid sequence encoding an agent that can enhance the activity of a modified T cell expressing the TFP as described herein and the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the third nucleic acid sequence is included in a separate nucleic acid sequence.
- the third nucleic acid sequence is included in the same nucleic acid molecule as the recombinant nucleic acid molecules.
- the agent that can enhance the activity of a modified T cell can be a PD-1 polypeptide.
- the PD-1 polypeptide may be operably linked to the N- terminus of an intracellular domain of a costimulatory polypeptide via the C-terminus of the PD-1 polypeptide.
- the agent that can enhance the activity of a modified T cell can be an anti-PD-1 antibody, or antigen binding fragment thereof.
- the anti-PD-1 antibody or antigen binding fragment thereof may be operably linked to the N-terminus of an intracellular domain of a costimulatory polypeptide via the C-terminus of the anti-PD-1 antibody, or antigen binding fragment thereof.
- the PD-1 polypeptide or anti-PD-1 antibody is linked to the intracellular domain of the costimulatory polypeptide via the transmembrane domain of PD- 1.
- the costimulatory polypeptide is selected from the group consisting of OX40, CD2, CD27, CD5, ICAM-1, ICOS (CD278), 4-1BB (CD137), GITR, CD28, CD30, CD40, IL-15Ra, IL12R, IL18R, IL21R, BAFFR, HVEM, CD7, LIGHT, NKG2C, SLAMF7, NKp80, CD160, CD226, Fc ⁇ RI, Fc ⁇ RII, and Fc ⁇ RIII.
- the costimulatory peptide is CD28.
- recombinant nucleic acid molecules comprising a first sequence encoding a TFP as described herein and a second sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, wherein the recombinant nucleic acid molecules further comprising an agent that can enhance the activity of a modified T cell expressing the TFP as described herein and the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the cells expressing TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein can further express another agent, e.g., an agent which enhances the activity of a modified T cell.
- the agent can be an agent which inhibits an inhibitory molecule.
- Inhibitory molecules, e.g., PD-1 can, in some embodiments, decrease the ability of a modified T cell to mount an immune effector response. Examples of inhibitory molecules include PD-1, PD-L1, CTLA4, TIM3, LAG3, VISTA, BTLA, TIGIT, LAIR1, CD160, and 2B4.
- the agent which inhibits an inhibitory molecule comprises a first polypeptide, e.g., an inhibitory molecule, associated with a second polypeptide that provides a positive signal to the cell, e.g., an intracellular signaling domain as described herein.
- the agent comprises a first polypeptide, e.g., of an inhibitory molecule such as PD-1, LAG3, CTLA4, CD160, BTLA, LAIR1, TIM3, 2B4, and TIGIT, or a fragment of any of these (e.g., at least a portion of an extracellular domain of any of these), and a second polypeptide which is an intracellular signaling domain described herein (e.g., comprising a costimulatory domain (e.g., 4-1BB, CD27 or CD28, e.g., as described herein) and/or a primary signaling domain (e.g., a CD3 zeta signaling domain described herein).
- an inhibitory molecule such as PD-1, LAG3, CTLA4, CD160, BTLA, LAIR1, TIM3, 2B4, and TIGIT
- a fragment of any of these e.g., at least a portion of an extracellular domain of any of these
- a second polypeptide which is an intra
- the agent comprises a first polypeptide of PD-1 or a fragment thereof (e.g., at least a portion of an extracellular domain of PD-1), and a second polypeptide of an intracellular signaling domain described herein (e.g., a CD28 signaling domain described herein and/or a CD3 zeta signaling domain described herein).
- the recombinant nucleic acid molecules as described herein further comprises a sequence encoding PD-1 or a fragment thereof.
- the recombinant nucleic acid molecules as described herein further comprises a sequence encoding the extracellular domain of PD-1.
- the recombinant nucleic acid molecules as described herein comprises a sequence encoding the extracellular domain and transmembrane domain of PD-1. In some embodiments, the recombinant nucleic acid molecules as described herein may further comprise a sequence encoding CD28 or a fragment thereof. In some embodiments, the recombinant nucleic acid molecules as described herein comprises a sequence encoding the intracellular domain of CD28. In some embodiments, the recombinant nucleic acid molecules as described herein comprises a sequence encoding a fusion protein comprising the PD-1 extracellular domain and transmembrane domain linked to the CD28 intracellular domain linked to intracellular domain.
- the agent comprises the extracellular and transmembrane domain of PD-1 fused to the intracellular signaling domain of CD28.
- the agent comprises SEQ ID NO: 370.
- PD1 is an inhibitory member of the CD28 family of receptors that also includes CD28, CTLA-4, ICOS, and BTLA.
- PD-1 is expressed on activated B cells, T cells and myeloid cells (Agata et al., 1996, Int. Immunol 8:765-75). Two ligands for PD1, PD-L1 and PD-L2, have been shown to downregulate T cell activation upon binding to PD1 (Freeman et al., 2000 J. Exp.
- PD-L1 is abundant in human cancers (Dong et al., 2003 J. Mol. Med.81:281-7; Blank et al., 2005 Cancer Immunol. Immunother.54:307-314; Konishi et al., 2004 Clin. Cancer Res.10:5094). Immune suppression can be reversed by inhibiting the local interaction of PD1 with PD-L1.
- the agent comprises the extracellular domain (ECD) of an inhibitory molecule, e.g., Programmed Death 1 (PD-1) can be fused to a transmembrane domain and optionally an intracellular signaling domain such as 41BB and CD3 zeta (also referred to herein as a PD
- the PD-1 TFP when used in combinations with an anti-TAA TFP described herein, improves the persistence of the T cell.
- the TFP is a PD-1 TFP comprising the extracellular domain of PD-1.
- TFPs containing an antibody or antibody fragment such as a scFv that specifically binds to the Programmed Death-Ligand 1 (PD-L1) or Programmed Death- Ligand 2 (PD-L2).
- the present disclosure provides a population of TFP-expressing T cells, e.g., TFP-T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the population of TFP-expressing T cells co- expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein comprises a mixture of cells expressing different TFPs.
- the population of TFP-T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein can include a first cell expressing a TFP having a binding domain described herein, and a second cell expressing a TFP having a different anti-TAA binding domain, e.g., a binding domain described herein that differs from the binding domain in the TFP expressed by the first cell.
- the population of TFP-expressing cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein can include a first cell expressing a TFP that includes a first binding domain binding domain, e.g., as described herein, and a second cell expressing a TFP that includes an antigen binding domain to a target other than the binding domain of the first cell (e.g., another tumor-associated antigen).
- the present disclosure provides a population of cells wherein at least one cell in the population expresses a TFP having a domain described herein, and a second cell expressing another agent, e.g., an agent which enhances the activity of a modified T cell.
- the agent can be an agent which inhibits an inhibitory molecule.
- Inhibitory molecules e.g., can, in some embodiments, decrease the ability of a modified T cell to mount an immune effector response. Examples of inhibitory molecules include PD-1, PD-L1, PD-L2, CTLA4, TIM3, LAG3, VISTA, BTLA, TIGIT, LAIR1, CD160, and 2B4.
- the agent that inhibits an inhibitory molecule comprises a first polypeptide, e.g., an inhibitory molecule, associated with a second polypeptide that provides a positive signal to the cell, e.g., an intracellular signaling domain described herein.
- Pharmaceutical Compositions [0594] Disclosed herein, in some embodiments, are pharmaceutical compositions comprising: (a) the cells of the disclosure; and (b) a pharmaceutically acceptable carrier.
- pharmaceutical compositions comprising: (a) the modified T cells of the disclosure; and (b) a pharmaceutically acceptable carrier.
- compositions comprising: (a) the nucleic acid molecules of the disclosure; and (b) a pharmaceutically acceptable carrier.
- Such compositions may comprise buffers such as neutral buffered saline, phosphate buffered saline and the like; carbohydrates such as glucose, mannose, sucrose or dextrans, mannitol; proteins; polypeptides or amino acids such as glycine; antioxidants; chelating agents such as EDTA or glutathione; adjuvants (e.g., aluminum hydroxide); and preservatives.
- buffers such as neutral buffered saline, phosphate buffered saline and the like
- carbohydrates such as glucose, mannose, sucrose or dextrans, mannitol
- proteins such as glucose, mannose, sucrose or dextrans, mannitol
- proteins such as glucose, mannose, sucrose or dextrans, mannitol
- proteins such as glucose, mannose, sucrose or dextrans, mannito
- compositions of the present disclosure may be administered in a manner appropriate to the disease to be treated (or prevented).
- the quantity and frequency of administration will be determined by such factors as the condition of the patient, and the type and severity of the patient’s disease, although appropriate dosages may be determined by clinical trials.
- the pharmaceutical composition is substantially free of, e.g., there are no detectable levels of a contaminant, e.g., selected from the group consisting of endotoxin, mycoplasma, replication competent lentivirus (RCL), p24, VSV-G nucleic acid, HIV gag, residual anti-CD3/anti-CD28 coated beads, mouse antibodies, pooled human serum, bovine serum albumin, bovine serum, culture media components, vector packaging cell or plasmid components, a bacterium and a fungus.
- a contaminant e.g., selected from the group consisting of endotoxin, mycoplasma, replication competent lentivirus (RCL), p24, VSV-G nucleic acid, HIV gag, residual anti-CD3/anti-CD28 coated beads, mouse antibodies, pooled human serum, bovine serum albumin, bovine serum, culture media components, vector packaging cell or plasmid components, a bacterium and a fungus.
- the bacterium is at least one selected from the group consisting of Alcaligenes faecalis, Candida albicans, Escherichia coli, Haemophilus influenza, Neisseria meningitides, Pseudomonas aeruginosa, Staphylococcus aureus, Streptococcus pneumonia, and Streptococcus pyogenes group A.
- an immunologically effective amount When “an immunologically effective amount,” “an anti-tumor effective amount,” “a tumor-inhibiting effective amount,” or “therapeutic amount” is indicated, the precise amount of the compositions of the present disclosure to be administered can be determined by a physician with consideration of individual differences in age, weight, tumor size, extent of infection or metastasis, and condition of the patient (subject). It can generally be stated that a pharmaceutical composition comprising the T cells as described herein may be administered at a dosage of 10 4 to 10 9 cells/kg body weight, in some instances 10 5 to 10 6 cells/kg body weight, including all integer values within those ranges. T cell compositions may also be administered multiple times at these dosages.
- the cells can be administered by using infusion techniques that are commonly known in immunotherapy (see, e.g., Rosenberg et al., New Eng. J. Med.319:1676, 1988).
- T cells can be activated from blood draws of from 10 cc to 400 cc.
- T cells are activated from blood draws of 20 cc, 30 cc, 40 cc, 50 cc, 60 cc, 70 cc, 80 cc, 90 cc, or 100 cc.
- the administration of the subject compositions may be carried out in any convenient manner, including by aerosol inhalation, injection, ingestion, transfusion, implantation or transplantation.
- the compositions described herein may be administered to a patient trans arterially, subcutaneously, intradermally, intratumorally, intranodally, intramedullary, intramuscularly, by intravenous (i.v.) injection, or intraperitoneally.
- the T cell compositions of the present disclosure are administered to a patient by intradermal or subcutaneous injection. In one aspect, the T cell compositions of the present disclosure are administered by i.v. injection.
- the compositions of T cells may be injected directly into a tumor, lymph node, or site of infection.
- subjects may undergo leukapheresis, wherein leukocytes are collected, enriched, or depleted ex vivo to select and/or isolate the cells of interest, e.g., T cells.
- T cell isolates may be expanded by methods known in the art and treated such that one or more TFP and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof constructs of the present disclosure may be introduced, thereby creating a modified T-T cell of the present disclosure.
- Subjects in need thereof may subsequently undergo standard treatment with high dose chemotherapy followed by peripheral blood stem cell transplantation.
- subjects receive an infusion of the expanded modified T cells of the present disclosure.
- expanded cells are administered before or following surgery.
- the dosage of the above treatments to be administered to a patient will vary with the precise nature of the condition being treated and the recipient of the treatment.
- the scaling of dosages for human administration can be performed according to art-accepted practices.
- the dose for alemtuzumab for example, will generally be in the range 1 to about 100 mg for an adult patient, usually administered daily for a period between 1 and 30 days.
- the preferred daily dose is 1 to 10 mg per day although in some instances larger doses of up to 40 mg per day may be used (described in U.S. Pat. No.6,120,766).
- the TFP as described herein and the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein is introduced into T cells, e.g., using in vitro transcription, and the subject (e.g., human) receives an initial administration of TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof of the present disclosure, and one or more subsequent administrations of the TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof of the present disclosure, wherein the one or more subsequent administrations are administered less than 15 days, e.g., 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, or 2 days after the previous administration.
- more than one administration of the TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof of the present disclosure are administered to the subject (e.g., human) per week, e.g., 2, 3, or 4 administrations of the TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof of the present disclosure are administered per week.
- the subject receives more than one administration of the TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein per week (e.g., 2, 3 or 4 administrations per week) (also referred to herein as a cycle), followed by a week of no TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein administrations, and then one or more additional administration of the TFP T cells co- expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein (e.g., more than one administration of the TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein per week) is administered to the subject.
- the subject receives more than one cycle of TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, and the time between each cycle is less than 10, 9, 8, 7, 6, 5, 4, or 3 days.
- the TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein are administered every other day for 3 administrations per week.
- the TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof of the present disclosure are administered for at least two, three, four, five, six, seven, eight or more weeks.
- CD19 TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein are generated using lentiviral viral vectors, such as lentivirus.
- TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein transiently express TFP as described herein and/or switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein vectors for 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15 days after transduction.
- Transient expression of TFPs as described herein and/or the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein can be affected by RNA TFP as described herein and/or switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein vector delivery.
- the TFP as described herein and/or the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein RNA is transduced into the T cell by electroporation.
- a potential issue that can arise in patients being treated using T cells transiently expressing TFP as described herein and the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein (particularly with T cells expressing murine scFv bearing TFP) is anaphylaxis after multiple treatments.
- an anaphylactic response might be caused by a patient developing humoral anti-TFP response, i.e., anti-TFP antibodies having an anti-IgE isotype.
- TFP T cell infusion breaks should not last more than ten to fourteen days.
- the recombinant nucleic acid can comprise a sequence encoding a TFP as described herein and/or a sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the method can further comprise (a) disrupting an endogenous TCR gene encoding a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, or any combination thereof; thereby producing a T cell containing a functional disruption of an endogenous TCR gene; and (b) transducing the T cell containing a functional disruption of an endogenous TCR gene with the recombinant nucleic acid of the disclosure, or the vectors as described herein.
- disrupting comprises transducing the T cell with a nuclease protein or a nucleic acid sequence encoding a nuclease protein that targets the endogenous gene encoding a TCR alpha chain, a TCR beta chain, or a TCR alpha chain and a TCR beta chain.
- methods of producing the modified T cell of the disclosure comprising transducing a T cell containing a functional disruption of an endogenous TCR gene with the recombinant nucleic acid as described herein, or the vectors as described herein.
- the T cell containing a functional disruption of an endogenous TCR gene is a T cell containing a functional disruption of an endogenous TCR gene encoding a TCR alpha chain, a TCR beta chain, or a TCR alpha chain and a TCR beta chain.
- the T cell is a human T cell.
- the T cell containing a functional disruption of an endogenous TCR gene has reduced binding to MHC- peptide complex compared to that of an unmodified control T cell.
- the nuclease is a meganuclease, a zinc-finger nuclease (ZFN), a transcription activator-like effector nuclease (TALEN), a CRISPR/Cas nuclease, CRISPR/Cas nickase, or a megaTAL nuclease.
- the sequence comprised by the recombinant nucleic acid or the vector is inserted into the endogenous TCR subunit gene at the cleavage site, and wherein the insertion of the sequence into the endogenous TCR subunit gene functionally disrupts the endogenous TCR subunit.
- the nuclease is a meganuclease.
- the meganuclease comprises a first subunit and a second subunit, wherein the first subunit binds to a first recognition half-site of the recognition sequence, and wherein the second subunit binds to a second recognition half-site of the recognition sequence.
- the meganuclease is a single-chain meganuclease comprising a linker, wherein the linker covalently joins the first subunit and the second subunit.
- the modified immune cells e.g., T cells, as described herein are further engineered using a gene editing technique such as clustered regularly interspaced short palindromic repeats (CRISPR®, see, e.g., US Patent No.8,697,359), transcription activator-like effector (TALE) nucleases (TALENs, see, e.g., U.S. Patent No.9,393,257), meganucleases (endodeoxyribonucleases having large recognition sites comprising double- stranded DNA sequences of 12 to 40 base pairs), zinc finger nuclease (ZFN, see, e.g., Urnov et al., Nat.
- CRISPR® clustered regularly interspaced short palindromic repeats
- TALE transcription activator-like effector
- TALENs transcription activator-like effector
- meganucleases endodeoxyribonucleases having large recognition sites comprising double- stranded DNA sequence
- a chimeric construct may be engineered to combine desirable characteristics of each subunit, such as conformation or signaling capabilities. See also Sander & Joung, Nat. Biotech. (2014) v32, 347-55; and June et al., 2009 Nature Reviews Immunol.9.10: 704-716, each incorporated herein by reference.
- one or more of the extracellular domain, the transmembrane domain, or the cytoplasmic domain of a TFP subunit are engineered to have aspects of more than one natural TCR subunit domain (i.e., are chimeric).
- TCR subunit domain i.e., are chimeric.
- the inactivation can include disruption of genomic gene locus, gene silencing, inhibition or reduction of transcription, or inhibition or reduction of translation.
- the endogenous TCR gene can be silenced, for example, by inhibitory nucleic acids such as siRNA and shRNA.
- the translation of the endogenous TCR gene can be inhibited by inhibitory nucleic acids such as microRNA.
- gene editing techniques are employed to disrupt an endogenous TCR gene.
- mentioned endogenous TCR gene encodes a TCR alpha chain, a TCR beta chain, or a TCR alpha chain and a TCR beta chain.
- gene editing techniques pave the way for multiplex genomic editing, which allows simultaneous disruption of multiple genomic loci in endogenous TCR gene.
- multiplex genomic editing techniques are applied to generate gene-disrupted T cells that are deficient in the expression of endogenous TCR, and/or B2M, and/or human leukocyte antigens (HLAs), and/or programmed cell death protein 1 (PD-1), and/or other genes.
- Current gene editing technologies comprise meganucleases, zinc-finger nucleases (ZFN), TAL effector nucleases (TALEN), and clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated (Cas) system.
- DSB double-stranded DNA break
- NHEJ non-homologous end joining
- HR homologous recombination
- DSBs may be repaired by single strand DNA incorporation (ssDI) or single strand template repair (ssTR), an event that introduces the homologous sequence from a donor DNA.
- Geno modification of genomic DNA can be performed using site-specific, rare- cutting endonucleases that are engineered to recognize DNA sequences in the locus of interest.
- Methods for producing engineered, site-specific endonucleases are known in the art.
- ZFNs zinc-finger nucleases
- ZFNs are chimeric proteins comprising a zinc finger DNA- binding domain fused to the nuclease domain of the Fokl restriction enzyme.
- the zinc finger domain can be redesigned through rational or experimental means to produce a protein that binds to a pre-determined DNA sequence -18 base pairs in length.
- TAL-effector nucleases can be generated to cleave specific sites in genomic DNA.
- a TALEN comprises an engineered, site- specific DNA-binding domain fused to the Fokl nuclease domain (reviewed in Mak et al. (2013), Curr Opin Struct Biol.23:93-9).
- the DNA binding domain comprises a tandem array of TAL-effector domains, each of which specifically recognizes a single DNA base pair.
- Compact TALENs have an alternative endonuclease architecture that avoids the need for dimerization (Beurdeley et al. (2013), Nat Commun.4: 1762).
- a Compact TALEN comprises an engineered, site-specific TAL-effector DNA-binding domain fused to the nuclease domain from the I-TevI homing endonuclease. Unlike Fokl, I-TevI does not need to dimerize to produce a double-strand DNA break so a Compact TALEN is functional as a monomer.
- Engineered endonucleases based on the CRISPR/Cas9 system are also known in the art (Ran et al. (2013), Nat Protoc.8:2281-2308; Mali et al. (2013), Nat Methods 10:957-63).
- the CRISPR gene-editing technology is composed of an endonuclease protein whose DNA- targeting specificity and cutting activity can be programmed by a short guide RNA or a duplex crRNA/TracrRNA.
- a CRISPR endonuclease comprises two components: (1) a caspase effector nuclease, typically microbial Cas9; and (2) a short “guide RNA” or an RNA duplex comprising an 18 to 20 nucleotide targeting sequence that directs the nuclease to a location of interest in the genome.
- a caspase effector nuclease typically microbial Cas9
- a short “guide RNA” or an RNA duplex comprising an 18 to 20 nucleotide targeting sequence that directs the nuclease to a location of interest in the genome.
- Class II contains type II, IV, V, and VI CRISPR systems.
- CRISPR/Cas system is the type II CRISPR-Cas9 system
- CRISPR/Cas systems have been repurposed by researchers for genome editing. More than 10 different CRISPR/Cas proteins have been remodeled within last few years (Adli (2016) Nat. Commun.9:1911).
- Cas12a (Cpf1) proteins from Acid- aminococcus sp (AsCpf1) and Lachnospiraceae bacterium (LbCpf1) are particularly interesting.
- Homing endonucleases are a group of naturally-occurring nucleases that recognize 15-40 base-pair cleavage sites commonly found in the genomes of plants and fungi. They are frequently associated with parasitic DNA elements, such as group 1 self-splicing introns and inteins. They naturally promote homologous recombination or gene insertion at specific locations in the host genome by producing a double -stranded break in the chromosome, which recruits the cellular DNA-repair machinery (Stoddard (2006), Q. Rev. Biophys.38: 49- 95).
- meganucleases are monomeric proteins with innate nuclease activity that are derived from bacterial homing endonucleases and engineered for a unique target site (Gersbach (2016), Molecular Therapy. 24: 430–446).
- meganuclease is engineered I-CreI homing endonuclease. In other embodiments, meganuclease is engineered I-SceI homing endonuclease.
- chimeric proteins comprising fusions of meganucleases, ZFNs, and TALENs have been engineered to generate novel monomeric enzymes that take advantage of the binding affinity of ZFNs and TALENs and the cleavage specificity of meganucleases (Gersbach (2016), Molecular Therapy 24: 430–446).
- a megaTAL is a single chimeric protein, which is the combination of the easy-to-tailor DNA binding domains from TALENs with the high cleavage efficiency of meganucleases.
- the nucleases and in the case of the CRISPR/ Cas9 system, a gRNA, may need to be efficiently delivered to the cells of interest. Delivery methods such as physical, chemical, and viral methods are also know in the art (Mali (2013). Indian J. Hum. Genet.19: 3-8.). In some instances, physical delivery methods can be selected from the methods but not limited to electroporation, microinjection, or use of ballistic particles. On the other hand, chemical delivery methods require use of complex molecules such calcium phosphate, lipid, or protein. In some embodiments, viral delivery methods are applied for gene editing techniques using viruses such as but not limited to adenovirus, lentivirus, and retrovirus.
- viruses such as but not limited to adenovirus, lentivirus, and retrovirus.
- the endogenous TCR gene (e.g., a TRAC locus or a TRBC locus) encoding a TCR alpha chain, a TCR beta chain, or a TCR alpha chain and a TCR beta chain can be inactivated by CRISPR/Cas9 system.
- the gRNA used to inactivate (e.g., disrupt) the TRAC locus can comprise a sequence of SEQ ID: 159.
- the gRNA used to disrupt the TRBC locus can comprise a sequence of SEQ ID: 197.
- CTCGACCAGCTTGACATCAC (SEQ ID NO: 159).
- ACACTGGTGTGCCTGGCCAC (SEQ ID NO: 197).
- Methods of Treatment are a method of treating a disease or a condition in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of the pharmaceutical compositions as described herein. Further disclosed herein, in some embodiments, are methods of treating a disease or a condition in a subject in need thereof, the method comprising administering to the subject a therapeutically effective amount of a pharmaceutical composition comprising (a) a cell produced according to the methods as described herein; and (b) a pharmaceutically acceptable carrier.
- the disease or the condition is a cancer or a disease or a condition associated with expression of CD19, B-cell maturation antigen (BCMA), mesothelin (MSLN), CD20, CD70, MUC16, Trop-2, Nectin-4, or GPC3.
- the cancer is a hematologic cancer.
- B-cell acute lymphoid leukemia B-ALL
- T-ALL T cell acute lymphoid leukemia
- ALL acute lymphoblastic leukemia
- CML chronic myelogenous leukemia
- CLL chronic lymphocytic leukemia
- B-ALL B-cell acute lymphoid leukemia
- T-ALL T cell acute lymphoid leukemia
- ALL acute lymphoblastic leukemia
- CML chronic myelogenous leukemia
- CLL chronic lymphocytic leukemia
- B cell prolymphocytic leukemia blastic plasmacytoid dendritic cell neoplasm
- Burkitt s lymphoma
- diffuse large B cell lymphoma follicular lymphoma
- hairy cell leukemia small cell-follicular lymphoma
- large cell-follicular lymphoma malignant lymphoproliferative conditions
- MALT lymphoma mantle cell lymphoma
- the subject is a mammal. In some embodiments, the subject is a human. [0627] Disclosed herein, in some embodiments, are methods of increasing the activity or persistence of a cell expressing a recombinant nucleic acid molecule comprising a sequence encoding the TFP as described herein by expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein in the cell. In some embodiments, the cell is any one of cells described herein.
- a pharmaceutical composition comprising (a) a modified T cell produced according to the methods as described herein; and (b) a pharmaceutically acceptable carrier.
- the modified T cell is an autologous T cell.
- the T cell is an allogeneic T cell. In some instances, less cytokines are released in the subject compared a subject administered an effective amount of an unmodified control T cell.
- the method comprises administering the pharmaceutical composition as described herein in combination with an agent that increases the efficacy of the pharmaceutical composition. In some instances, the method comprises administering the pharmaceutical composition in combination with an agent that ameliorates one or more side effects associated with the pharmaceutical composition.
- the cancer is a solid cancer, a lymphoma or a leukemia.
- the cancer is selected from the group consisting of renal cell carcinoma, breast cancer, lung cancer, ovarian cancer, prostate cancer, colon cancer, cervical cancer, brain cancer, liver cancer, pancreatic cancer, kidney and stomach cancer.
- the present disclosure includes a type of cellular therapy where T cells are genetically modified to express a TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein and the modified T cell is infused to a recipient in need thereof.
- the infused cell is able to kill tumor cells in the recipient.
- modified T cells are able to replicate in vivo resulting in long-term persistence that can lead to sustained tumor control.
- the T cells administered to the patient, or their progeny persist in the patient for at least four months, five months, six months, seven months, eight months, nine months, ten months, eleven months, twelve months, thirteen months, fourteen month, fifteen months, sixteen months, seventeen months, eighteen months, nineteen months, twenty months, twenty-one months, twenty-two months, twenty-three months, two years, three years, four years, or five years after administration of the T cell to the patient.
- the present disclosure also includes a type of cellular therapy where T cells are modified, e.g., by in vitro transcribed RNA, to transiently express a TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein and the modified T cell is infused to a recipient in need thereof.
- the infused cell is able to kill tumor cells in the recipient.
- the T cells administered to the patient is present for less than one month, e.g., three weeks, two weeks, or one week, after administration of the T cell to the patient.
- the anti-tumor immunity response elicited by the modified T cells may be an active or a passive immune response, or alternatively may be due to a direct vs indirect immune response.
- the human modified T cells of the disclosure may be a type of vaccine for ex vivo immunization and/or in vivo therapy in a mammal.
- the mammal is a human.
- a nucleic acid encoding a TCR gamma and/or delta constant domain is further introduced to the cells.
- cells are isolated from a mammal (e.g., a human) and genetically modified (i.e., transduced or transfected in vitro) with a vector as described herein.
- the modified T cell can be administered to a mammalian recipient to provide a therapeutic benefit.
- the mammalian recipient may be a human and the modified cell can be autologous with respect to the recipient.
- the cells can be allogeneic, syngeneic or xenogeneic with respect to the recipient.
- the procedure for ex vivo expansion of hematopoietic stem and progenitor cells is described in U.S. Pat. No.5,199,942, incorporated herein by reference, can be applied to the cells of the present disclosure.
- ex vivo culture and expansion of immune cells comprises: (1) collecting CD34+ hematopoietic stem and progenitor cells from a mammal from peripheral blood harvest or bone marrow explants; and (2) expanding such cells ex vivo.
- T cells e.g., TGF-derived progenitor cells
- other factors such as flt3-L, IL- 1, IL-3 and c-kit ligand, can be used for culturing and expansion of the cells.
- the present disclosure also provides compositions and methods for in vivo immunization to elicit an immune response directed against an antigen in a patient.
- the cells activated and expanded as described herein may be utilized in the treatment and prevention of diseases that arise in individuals who are immunocompromised.
- the modified T cells of the present disclosure may be administered either alone, or as a pharmaceutical composition in combination with diluents and/or with other components such as IL-2 or other cytokines or cell populations.
- Combination Therapies [0642] A modified immune cells, e.g., T cell described herein may be used in combination with other known agents and therapies.
- Administered “in combination”, as used herein, means that two (or more) different treatments are delivered to the subject during the course of the subject’s affliction with the disorder, e.g., the two or more treatments are delivered after the subject has been diagnosed with the disorder and before the disorder has been cured or eliminated or treatment has ceased for other reasons.
- the delivery of one treatment is still occurring when the delivery of the second begins, so that there is overlap in terms of administration. This is sometimes referred to herein as “simultaneous” or “concurrent delivery”.
- the delivery of one treatment ends before the delivery of the other treatment begins. In some embodiments of either case, the treatment is more effective because of combined administration.
- the second treatment is more effective, e.g., an equivalent effect is seen with less of the second treatment, or the second treatment reduces symptoms to a greater extent, than would be seen if the second treatment were administered in the absence of the first treatment or the analogous situation is seen with the first treatment.
- delivery is such that the reduction in a symptom, or other parameter related to the disorder is greater than what would be observed with one treatment delivered in the absence of the other.
- the effect of the two treatments can be partially additive, wholly additive, or greater than additive.
- the delivery can be such that an effect of the first treatment delivered is still detectable when the second is delivered.
- the “at least one additional therapeutic agent” includes a modified immune cell, e.g., T cell.
- T cells that express multiple TFPs as described herein, which bind to the same or different target antigens, or same or different epitopes on the same target antigen, and multiple switch polypeptides comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein; T cells that express multiple TFPs as described herein, which bind to the same or different target antigens, or same or different epitopes on the same target antigen, and multiple dominant negative polypeptides comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein; and T cells that express multiple TFPs as described herein, which bind to the same or different target antigens, or same or different epitopes on the same target antigen, and multiple switch polypeptides comprising a TGFBr2 extracellular domain or a functional fragment thereof and multiple dominant negative polypeptides comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- a modified immune cell e.g., T cell, described herein and the at least one additional therapeutic agent can be administered simultaneously, in the same or in separate compositions, or sequentially.
- a modified immune cell e.g., T cell described herein can be administered first, and the additional agent can be administered second, or the order of administration can be reversed.
- a modified immune cell, e.g., T cell, as described herein may be used in a treatment regimen in combination with surgery, chemotherapy, radiation, immunosuppressive agents, such as cyclosporin, azathioprine, methotrexate, mycophenolate, and tacrolimus, antibodies, or other immunoablative agents such as alemtuzumab, anti-CD3 antibodies or other antibody therapies, cytoxin, fludarabine, cyclosporin, tacrolimus, rapamycin, mycophenolic acid, steroids, romidepsin, cytokines, and irradiation.
- immunosuppressive agents such as cyclosporin, azathioprine, methotrexate, mycophenolate, and tacrolimus
- immunoablative agents such as alemtuzumab, anti-CD
- the subject can be administered an agent which reduces or ameliorates a side effect associated with the administration of a modified immune cell, e.g., T cell.
- a modified immune cell e.g., T cell.
- Side effects associated with the administration of a modified T cell include but are not limited to cytokine release syndrome (CRS), and hemophagocytic lymphohistiocytosis (HLH), also termed Macrophage Activation Syndrome (MAS).
- CRS cytokine release syndrome
- HHLH hemophagocytic lymphohistiocytosis
- MAS Macrophage Activation Syndrome
- Symptoms of CRS include high fevers, nausea, transient hypotension, hypoxia, and the like.
- the methods as described herein can comprise administering a modified T cell described herein to a subject and further administering an agent to manage elevated levels of a soluble factor resulting from treatment with a modified T cell.
- the soluble factor elevated in the subject is one or more of IFN- ⁇ , TNF ⁇ , IL-2 and IL-6. Therefore, an agent administered to treat this side effect can be an agent that neutralizes one or more of these soluble factors.
- agents include, but are not limited to a steroid, an inhibitor of TNF ⁇ , and an inhibitor of IL-6.
- An example of a TNF ⁇ inhibitor is entanercept.
- An example of an IL-6 inhibitor is tocilizumab (toc).
- the subject can be administered an agent which enhances the activity of a modified immune cell, e.g., T cell.
- the agent can be an agent which inhibits an inhibitory molecule.
- Inhibitory molecules e.g., Programmed Death 1 (PD-1)
- PD-1 Programmed Death 1
- Examples of inhibitory molecules include PD-1, PD-L1, CTLA4, TIM3, LAG3, VISTA, BTLA, TIGIT, LAIR1, CD160, and 2B4.
- Inhibition of an inhibitory molecule e.g., by inhibition at the DNA, RNA or protein level, can optimize a modified T cell performance.
- an inhibitory nucleic acid e.g., an inhibitory nucleic acid, e.g., a dsRNA, e.g., an siRNA or shRNA
- an inhibitory nucleic acid e.g., a dsRNA, e.g., an siRNA or shRNA
- the inhibitor is a shRNA.
- the inhibitory molecule is inhibited within a modified T cell.
- a dsRNA molecule that inhibits expression of the inhibitory molecule is linked to the nucleic acid that encodes a component, e.g., all of the components, of the TFP as described herein and/or the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the inhibitor of an inhibitory signal can be, e.g., an antibody or antibody fragment that binds to an inhibitory molecule.
- the agent can be an antibody or antibody fragment that binds to PD-1, PD-L1, PD-L2 or CTLA4 (e.g., ipilimumab (also referred to as MDX-010 and MDX-101 and marketed as Yervoy ® ; Bristol-Myers Squibb; tremelimumab (IgG2 monoclonal antibody available from Pfizer, formerly known as ticilimumab, CP-675,206)).
- the agent is an antibody or antibody fragment that binds to TIM3.
- the agent is an antibody or antibody fragment that binds to LAG3.
- the agent which enhances the activity of a modified T cell can be, e.g., a fusion protein comprising a first domain and a second domain, wherein the first domain is an inhibitory molecule, or fragment thereof, and the second domain is a polypeptide that is associated with a positive signal, e.g., a polypeptide comprising an intracellular signaling domain as described herein.
- the polypeptide that is associated with a positive signal can include a costimulatory domain of CD28, CD27,
- the fusion protein is expressed by the same cell that expresses the TFP as described herein and the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- the fusion protein is expressed by a cell, e.g. , a T cell that does not express an anti-TAA TFP as described herein and the switch polypeptide or dominant negative polypeptide comprising a
- TGFBr2 extracellular domain or a functional fragment thereof as described herein.
- T-Cell Receptor is formed by a complex of dimers TCR ⁇ / ⁇ , CD3 ⁇ / ⁇ , CD3 ⁇ / ⁇ and the homodimer CD3 ⁇ / ⁇ .
- TCR ⁇ / ⁇ are expressed instead of TCR ⁇ / ⁇ to form a functional TCR.
- TCR ⁇ / ⁇ / ⁇ / ⁇ have a constant domain common to all T- cells and a variable domain specific to an antigen.
- TRAC, TRBC, TRGC and TRDC genes encode for the constant C-terminal region of TCR ⁇ , TCR ⁇ , TCR ⁇ and TCR ⁇ respectively.
- TCRa only pairs with TCRb and TCR ⁇ only pairs with TCR ⁇ .
- a TCR complex is formed with TCR ⁇ / ⁇ in ⁇ / ⁇ T cells or with TCR ⁇ / ⁇ in ⁇ / ⁇ T cells.
- TFP constructs are generated as previously described.
- An anti-MSLN binder is linked to a CD3 or TCR DNA fragment by either a DNA sequence encoding a short linker (SL): AAAGGGGSGGGGSGGGGSLE (SEQ ID NO:387) or a long linker (LL): AAAIEVMYPPPYLGGGGSGGGGSGGGGSLE (SEQ ID NO:388) into a lentiviral vector (pLRPO, pLRPC, pLCUS, or pLKaUS).
- SL short linker
- LL long linker
- pLRPO lentiviral vector
- pLRPC pLRPC
- pLCUS pLCUS
- pLKaUS lentiviral vector
- the TFP used is TC-210 (an anti-MSLN MH1e VHH antibody linked to CD3 epsilon) having the sequence of SEQ ID NO: 195.
- a TCR complex contains the CD3-epsilon polypeptide, the CD3-gamma poly peptide, the CD3-delta polypeptide, and the TCR alpha chain polypeptide and the TCR beta chain polypeptide or the TCR delta chain polypeptide and the TCR gamma chain polypeptide.
- TCR alpha, TCR beta, TCR gamma, and TCR delta recruit the CD3 zeta polypeptide.
- the human CD3-epsilon polypeptide canonical sequence is Uniprot Accession No. P07766.
- the human CD3-gamma polypeptide canonical sequence is Uniprot Accession No. P09693.
- the human CD3-delta polypeptide canonical sequence is Uniprot Accession No. P043234.
- the human CD3-zeta polypeptide canonical sequence is Uniprot Accession No. P20963.
- the human TCR alpha chain canonical sequence is Uniprot Accession No. Q6ISU1.
- the murine TCR alpha chain canonical sequence is Uniprot Accession No. A0A075B662.
- the human TCR beta chain constant region canonical sequence is Uniprot Accession No. P01850.
- the murine TCR beta chain constant region canonical sequence is Uniprot Accession No. P01852.
- the human CD3-epsilon polypeptide canonical sequence is: MQSGTHWRVLGLCLLSVGVWGQDGNEEMGGITQTPYKVSISGTTVILTCPQYPGSEI LWQHNDKNIGGDEDDKNIGSDEDHLSLKEFSELEQSGYYVCYPRGSKPEDANFYLY LRARVCENCMEMDVMSVATIVIVDICITGGLLLLVYYWSKNRKAKAKPVTRGAGAG GRQRGQNKERPPPVPNPDYEPIRKGQRDLYSGLNQRRI (SEQ ID NO:124).
- the mature human CD3-epsilon polypeptide sequence is: DGNEEMGGITQTPYKVSISGTTVILTCPQYPGSEILWQHNDKNIGGDEDDKNIGSDED HLSLKEFSELEQSGYYVCYPRGSKPEDANFYLYLRARVCENCMEMDVMSVATIVIV DICITGGLLLLVYYWSKNRKAKAKPVTRGAGAGGRQRGQNKERPPPVPNPDYEPIR KGQRDLYSGLNQRRI (SEQ ID NO:258).
- the signal peptide of human CD3 ⁇ is: MQSGTHWRVLGLCLLSVGVWGQ (SEQ ID NO:125).
- the extracellular domain of human CD3 ⁇ is: DGNEEMGGITQTPYKVSISGTTVILTCPQYPGSEILWQHNDKNIGGDEDDKNIGSDED HLSLKEFSELEQSGYYVCYPRGSKPEDANFYLYLRARVCENCMEMD (SEQ ID NO:126).
- the transmembrane domain of human CD3 ⁇ is: VMSVATIVIVDICITGGLLLLVYYWS (SEQ ID NO:127).
- the intracellular domain of human CD3 ⁇ is: KNRKAKAKPVTRGAGAGGRQRGQNKERPPPVPNPDYEPIRKGQRDLYSGLNQRRI (SEQ ID NO:128).
- the human CD3-gamma polypeptide canonical sequence is: MEQGKGLAVLILAIILLQGTLAQSIKGNHLVKVYDYQEDGSVLLTCDAEAKNITWFK DGKMIGFLTEDKKKWNLGSNAKDPRGMYQCKGSQNKSKPLQVYYRMCQNCIELNA ATISGFLFAEIVSIFVLAVGVYFIAGQDGVRQSRASDKQTLLPNDQLYQPLKDREDDQ YSHLQGNQLRRN (SEQ ID NO:129).
- the mature human CD3-gamma polypeptide sequence is: QSIKGNHLVKVYDYQEDGSVLLTCDAEAKNITWFKDGKMIGFLTEDKKKWNLGSN AKDPRGMYQCKGSQNKSKPLQVYYRMCQNCIELNAATISGFLFAEIVSIFVLAVGVY FIAGQDGVRQSRASDKQTLLPNDQLYQPLKDREDDQYSHLQGNQLRRN (SEQ ID NO:130).
- the signal peptide of human CD3 ⁇ is: MEQGKGLAVLILAIILLQGTLA (SEQ ID NO:131).
- the extracellular domain of human CD3 ⁇ is: QSIKGNHLVKVYDYQEDGSVLLTCDAEAKNITWFKDGKMIGFLTEDKKKWNLGSN AKDPRGMYQCKGSQNKSKPLQVYYRMCQNCIELNAATIS (SEQ ID NO:132).
- the transmembrane domain of human CD3 ⁇ is: GFLFAEIVSIFVLAVGVYFIA (SEQ ID NO:133).
- the intracellular domain of human CD3 ⁇ is: GQDGVRQSRASDKQTLLPNDQLYQPLKDREDDQYSHLQGNQLRRN (SEQ ID NO:134).
- the human CD3-delta polypeptide canonical sequence is: MEHSTFLSGLVLATLLSQVSPFKIPIEELEDRVFVNCNTSITWVEGTVGTLLSDITRLD LGKRILDPRGIYRCNGTDIYKDKESTVQVHYRMCQSCVELDPATVAGIIVTDVIATLL LALGVFCFAGHETGRLSGAADTQALLRNDQVYQPLRDRDDAQYSHLGGNWARNKS (SEQ ID NO:135).
- the mature human CD3-delta polypeptide sequence is: FKIPIEELEDRVFVNCNTSITWVEGTVGTLLSDITRLDLGKRILDPRGIYRCNGTDIYK DKESTVQVHYRMCQSCVELDPATVAGIIVTDVIATLLLALGVFCFAGHETGRLSGAA DTQALLRNDQVYQPLRDRDDAQYSHLGGNWARNKS (SEQ ID NO:136).
- the signal peptide of human CD3 ⁇ is: MEHSTFLSGLVLATLLSQVSP (SEQ ID NO:137).
- the extracellular domain of human CD3 ⁇ is: FKIPIEELEDRVFVNCNTSITWVEGTVGTLLSDITRLDLGKRILDPRGIYRCNGTDIYK DKESTVQVHYRMCQSCVELDPATVA (SEQ ID NO:138).
- the transmembrane domain of human CD3 ⁇ is: GIIVTDVIATLLLALGVFCFA (SEQ ID NO:139).
- the intracellular domain of human CD3 ⁇ is: GHETGRLSGAADTQALLRNDQVYQPLRDRDDAQYSHLGGNWARNK (SEQ ID NO:140).
- the human CD3-zeta polypeptide canonical sequence is: MKWKALFTAAILQAQLPITEAQSFGLLDPKLCYLLDGILFIYGVILTALFLRVKFSRSA DAPAYQQGQNQLYNELNLGRREEYDVLDKRRGRDPEMGGKPQRRKNPQEGLYNEL QKDKMAEAYSEIGMKGERRRGKGHDGLYQGLSTATKDTYDALHMQALPPR (SEQ ID NO:141).
- the human TCR alpha chain constant region canonical sequence is: IQNPDPAVYQLRDSKSSDKSVCLFTDFDSQTNVSQSKDSDVYITDKTVLDMRSMDFK SNSAVAWSNKSDFACANAFNNSIIPEDTFFPSPESSCDVKLVEKSFETDTNLNFQNLS VIGFRILLLKVAGFNLLMTLRLWSS (SEQ ID NO:142).
- the human TCR alpha chain human IgC sequence is: IQNPDPAVYQLRDSKSSDKSVCLFTDFDSQTNVSQSKDSDVYITDKTVLDMRSMDFK SNSAVAWSNKSDFACANAFNNSIIPEDTFFPSPESSCDVKLVEKSFETDTNLNFQNLS (SEQ ID NO:143).
- the transmembrane domain of the human TCR alpha chain is: VIGFRILLLKVAGFNLLMTLRLW (SEQ ID NO:144).
- the intracellular domain of the human TCR alpha chain is: SS (SEQ ID NO:145).
- the murine TCR alpha chain constant (mTRAC) region canonical sequence is: XIQNPEPAVYQLKDPRSQDSTLCLFTDFDSQINVPKTMESGTFITDKTVLDMKAMDS KSNGAIAWSNQTSFTCQDIFKETNATYPSSDVPCDATLTEKSFETDMNLNFQNLSVM GLRILLLKVAGFNLLMTLRLWSS (SEQ ID NO:146).
- the transmembrane domain of the murine TCR alpha chain is: MGLRILLLKVAGFNLLMTLRLW (SEQ ID NO:147).
- the intracellular domain of the murine TCR alpha chain is: SS (SEQ ID NO: 405).
- the human TCR beta chain constant region 1 (hTRBC1) canonical sequence is: EDLNKVFPPEVAVFEPSEAEISHTQKATLVCLATGFFPDHVELSWWVNGKEVHSGVS TDPQPLKEQPALNDSRYCLSSRLRVSATFWQNPRNHFRCQVQFYGLSENDEWTQDR AKPVTQIVSAEAWGRADCGFTSVSYQQGVLSATILYEILLGKATLYAVLVSALVLMA MVKRKDF (SEQ ID NO:148).
- the human TCR beta chain 1 human IgC sequence is: EDLNKVFPPEVAVFEPSEAEISHTQKATLVCLATGFFPDHVELSWWVNGKEVHSGVS TDPQPLKEQPALNDSRYCLSSRLRVSATFWQNPRNHFRCQVQFYGLSENDEWTQDR AKPVTQIVSAEAWGRADCGFTSVSYQQGVLSATILYE (SEQ ID NO: 149).
- the transmembrane domain of the human TCR beta chain 1 is: ILLGKATLYAVLVSALVLMAM (SEQ ID NO:150).
- the intracellular domain of the human TCR beta chain 1 is: VKRKDF (SEQ ID NO: 151).
- the human TCR beta chain 2 constant region (hTRBC2) canonical sequence is: DLKNVFPPKVAVFEPSEAEISHTQKATLVCLATGFYPDHVELSWWVNGKEVHSGVS TDPQPLKEQPALNDSRYCLSSRLRVSATFWQNPRNHFRCQVQFYGLSENDEWTQDR AKPVTQIVSAEAWGRADCGFTSESYQQGVLSATILYEILLGKATLYAVLVSALVLMA MVKRKDSRG (SEQ ID NO:371).
- the murine TCR beta chain constant region 1 canonical sequence is: EDLRNVTPPKVSLFEPSKAEIANKQKATLVCLARGFFPDHVELSWWVNGKEVHSGV STDPQAYKESNYSYCLSSRLRVSATFWHNPRNHFRCQVQFHGLSEEDKWPEGSPKP VTQNISAEAWGRADCGITSASYQQGVLSATILYEILLGKATLYAVLVSTLVVMAMV KRKNS (SEQ ID NO:152).
- the transmembrane domain of the murine TCR beta chain 1 is: ILYEILLGKATLYAVLVS TLVVMAMVK (SEQ ID NO:153).
- the intracellular domain of the murine TCR beta chain 1 is: KRKNS (SEQ ID NO:154).
- the murine TCR beta chain constant 2 region canonical sequence is: XDLRNVTPPKVSLFEPSKAEIANKQKATLVCLARGFFPDHVELSWWVNGKEVHSGV STDPQAYKESNYSYCLSSRLRVSATFWHNPRNHFRCQVQFHGLSEEDKWPEGSPKP VTQNISAEAWGRADCGITSASYHQGVLSATILYEILLGKATLYAVLVSGLVLMAMV KKKNS (SEQ ID NO:282).
- the human TCR gamma chain constant region canonical sequence is: DKQLDADVSPKPTIFLPSIAETKLQKAGTYLCLLEKFFPDVIKIHWQEKKSNTILGSQE GNTMKTNDTYMKFSWLTVPEKSLDKEHRCIVRHENNKNGVDQEIIFPPIKTDVITMD PKDNCSKDANDTLLLQLTNTSAYYMYLLLLLKSVVYFAIITCCLLRRTAFCCNGEKS (SEQ ID NO:21).
- the human TCR gamma human IgC sequence is: DKQLDADVSPKPTIFLPSIAETKLQKAGTYLCLLEKFFPDVIKIHWQEKKSNTILGSQE GNTMKTNDTYMKFSWLTVPEKSLDKEHRCIVRHENNKNGVDQEIIFPPIKTDVITMD PKDNCSKDANDTLLLQLTNTSA (SEQ ID NO: 155).
- the transmembrane domain of the human TCR gamma chain is: YYMYLLLLLKSVVYFAIITCCLL (SEQ ID NO:156).
- the intracellular domain of the human TCR gamma chain is: RRTAFCCNGEKS (SEQ ID NO: 157).
- the human TCR delta chain C region canonical sequence is: SQPHTKPSVFVMKNGTNVACLVKEFYPKDIRINLVSSKKITEFDPAIVISPSGKYNAV KLGKYEDSNSVTCSVQHDNKTVHSTDFEVKTDSTDHVKPKETENTKQPSKSCHKPK AIVHTEKVNMMSLTVLGLRMLFAKTVAVNFLLTAKLFFL (SEQ ID NO:243).
- the human TCR delta human IgC sequence is: SQPHTKPSVFVMKNGTNVACLVKEFYPKDIRINLVSSKKITEFDPAIVISPSGKYNAV KLGKYEDSNSVTCSVQHDNKTVHSTDFEVKTDSTDHVKPKETENTKQPSKSCHKPK AIVHTEKVNMMSLTV (SEQ ID NO: 265).
- the transmembrane domain of the human TCR delta chain is: LGLRMLFAKTVAVNFLLTAKLFF (SEQ ID NO:158).
- the intracellular domain of the human TCR delta chain is: L.
- TFP constructs are in a vector that further contains a sequence encoding a dominant negative TGF ⁇ RII.
- the dominant negative TGF ⁇ RII may be encoded in the same open reading frame and separated by a self-cleaving peptide (e.g., a P2A or a T2A self-cleaving peptide).
- the dominant negative TGF ⁇ RII comprises the extracellular domain of TGF ⁇ RII and lacks a functional kinase domain.
- the dominant negative TGF ⁇ RII comprises a TGF ⁇ RII extracellular domain comprising SEQ ID NO: 271 and a TGF ⁇ RII transmembrane domain comprising SEQ ID NO: 272. In some embodiments, the dominant negative TGF ⁇ RII comprises a TGF ⁇ RII extracellular domain, a TGF ⁇ RII transmembrane domain, and a portion of a TGF ⁇ RII intracellular domain, wherein the portion of the intracellular domain is not capable of kinase activity. In some embodiments, the dominant negative TGF ⁇ RII comprises SEQ ID NO: 68.
- TFP constructs are in a vector that further contains a sequence encoding a TGF ⁇ RII switch.
- the TGF ⁇ RII switch may be encoded in the same open reading frame and separated by a self-cleaving peptide (e.g., a P2AW or a T2A self- cleaving peptide).
- the TGF ⁇ RII comprises the extracellular domain of TGF ⁇ RII fused to the intracellular domain of CD28.
- the transmembrane domain is from TGF ⁇ RII and the switch comprises SEQ ID NO:273.
- the transmembrane domain is from CD28 and the switch comprises SEQ ID NO:273.
- the TGF ⁇ RII comprises the extracellular domain of TGF ⁇ RII fused to the intracellular domain of 4-1BB.
- the transmembrane domain is from TGF ⁇ RII and the switch comprises SEQ ID NO:277. In some embodiments, the transmembrane domain is from 4-1BB and the switch comprises SEQ ID NO:277.
- TFP Expression vectors include: a promoter (e.g., an EF1a promoter), a signal sequence to enable secretion, a polyadenylation signal and transcription terminator (Bovine Growth Hormone (BGH) gene), an element allowing episomal replication and replication in prokaryotes (e.g., SV40 origin and ColE1 or others known in the art) and elements to allow selection (ampicillin resistance gene and zeocin marker).
- a promoter e.g., an EF1a promoter
- BGH Bovine Growth Hormone
- T cells were purified from healthy donor leukopak or PBMCs via positive selection of CD4+ and CD8+ T cells with CD4 and CD8 microbeads from Miltenyi Biotech.
- T cells freshly isolated or thawed from previously prepared frozen vials, were activated by MACS GMP T cell TransAct (Miltenyi Biotech), in the presence of human IL-7 and IL-15 (both from Miltenyi Biotech, premium grade).
- activated T cells were transduced with lentivirus encoding the TFP.
- T cells were transduced with vectors expressing anti- MSLN TFP (referred to as MH1e, MH1e TFP, or TC-210), or the anti-MSLN TFP expressing the TGF ⁇ RII switch (or PD-1 switch) described above.
- MH1e anti- MSLN TFP
- MH1e TFP anti- MSLN TFP
- TC-210 vectors expressing anti- MSLN TFP
- TGF ⁇ RII switch or PD-1 switch
- FIG. 3 shows that although the ratio differed between donors, there was no significant difference in CD4:CD8 ratio between the different TFP constructs (with or without a TGF ⁇ RII or PD-1 switch).
- the memory status of the T cells was determined by flow cytometry to detect cell surface levels of CD45RA and CCR7 as is shown in Figure 4. Again, while the memory status differed between donors, there was no significant difference in memory status between the different TFP constructs (with or without a TGF ⁇ RII or PD-1 switch).
- Figure 5 shows detection of TGF ⁇ RII on the surface of TFP expressing cells.
- TGF ⁇ RII switch having the TGF ⁇ RII extracellular and transmembrane domains with the CD28 intracellular domain is expressed at the highest level while the TGF ⁇ RII switch having the TGF ⁇ RII extracellular and transmembrane domains with the 4-1BB transmembrane domain is expressed at low levels.
- Example 3 Plate bound stimulation assay [0703] TFP-expressing T cells expressing the constructs shown in Table 1 were expanded to day 10, as described above.2 x 10 5 TFP transduced T cells or untransduced control T cells in 200uL of R10 media were then added to one of four plates for 24 hours.
- the plates contained: (i) PBS control; (ii) Dynabeads at a 1:1 ratio with T cells (Positive Stimulation); (iii) 1ug/mL plate bound MSLN; or (iv) 1ug/mL plate bound MSLN with plate bound 5ug/mL PDL1-Fc.0, 1, 5, 25, or 200 ng/mL of TGF ⁇ was added to wells.
- FIG. 6 shows cytokine production in cells contacted with plate bound mesothelin in the presence or absence of 25 ng/mL TGF ⁇ .
- TGF ⁇ cytokines
- FIG. 7 shows IL-2 and IFN- ⁇ production in TFP-expressing cells or non- transduced cells contacted with plate bound mesothelin in the presence of TGF ⁇ across a range of TGF ⁇ .
- the data show that even 1 ng/mL of TGF ⁇ is sufficient to suppress cytokine production in TC-210 expressing cells and MH1e PD-1 switch expressing cells, and that increasing levels of TGF ⁇ do not further suppress cytokine production.
- increasing levels of TGF ⁇ increase the level of cytokines produced in cells expressing TGF ⁇ RII switches.
- FIG. 8 shows cytokine production in TFP expressing cells contacted with plate bound mesothelin and PD-L1 in the presence or absence of 25 ng/mL TGF ⁇ .
- TGF ⁇ cytokine production in TFP expressing cells contacted with plate bound mesothelin and PD-L1 in the presence or absence of 25 ng/mL TGF ⁇ .
- cytokines IFN- ⁇ , IL-2, TNF ⁇ , and GM-CSF
- TGF ⁇ inhibited cytokine production in PD-1 switch expressing TFP T cells.
- the expression of TGF ⁇ RII switches was not able to overcome PD-L1 mediated repression of cytokine expression.
- Example 4 Co-culture assay [0707] TFP-expressing T cells expanded to day 10, as described above, were contacted with MLSN-expressing MSTO-mlsn target cells, MSLN and TGF ⁇ -expressing OVCAR3 target cells, or C30 control cells, which do not express mesothelin, at a 1:1 ratio in 200uL TexMACs media in a 96 well flat bottom plate with or without added TGF ⁇ .24 hours after initiation of co-culture 100uL of supernatant was collect and frozen for cytokine analysis and 100uL of new media was added.72 hours after initiation of co-culture 100uL of supernatant was collect and frozen for cytokine analysis.
- Target-specific cytokine production including IFN- ⁇ , IL-2, TNF ⁇ , and GM-CSF by TFP T cells was measured from supernatants using the U-PLEX® Biomarker Group I (hu) Assays (Meso Scale Diagnostics®, LLC, catalog number: K15067L-4).
- U-PLEX® Biomarker Group I (hu) Assays Meso Scale Diagnostics®, LLC, catalog number: K15067L-4.
- Figure 9 shows IL-2 production for Donors R020 in Figures 10 and 11.
- Figure 10 shows IL-2 production in cells contacted with MSTO-msln, OVCAR3, or C30 cells in the presence or absence of 10 ng/mL TGF ⁇ .
- TGF ⁇ inhibited cytokine production in cells expressing TC-210 or MH1e TFPs with the PD-1 switch.
- Expression of TGF ⁇ RII switches rescued cytokine production in MH1e expressing cells when contacted with TGF ⁇ , with the TGF ⁇ RII switch having the TGF ⁇ RII transmembrane domain and the CD28 intracellular domain having the greatest effect.
- OVCAR3 target cells cytokine production is low in cells expressing TC-210 or MH1e TFPs with the PD-1 switch, likely due to the production of TGF ⁇ of the OVCAR3 cells.
- TGF ⁇ RII switches increased cytokine production in MH1e expressing cells, with the TGF ⁇ RII switch having the TGF ⁇ RII transmembrane domain and the CD28 intracellular domain having the greatest effect.
- cytokine production by cells expressing TGF ⁇ RII switches further increased. Very low cytokine levels were observed in all TFP-expressing cells contacted with C30 control target cells.
- FIG 11 shows IFN- ⁇ , IL-2, TNF ⁇ , and GM-CSF production in TFP-expressing cells or non-transduced cells contacted with contacted with MSTO-msln or OVCAR3 cells and TGF ⁇ across a range of TGF ⁇ concentrations (0.1, 1, 10, or 25 ng/mL).
- TGF ⁇ concentrations 0.1, 1, 10, or 25 ng/mL.
- the data show that even 0.1 ng/mL of TGF ⁇ is sufficient to suppress cytokine production in TC-210 expressing cells and MH1e PD-1 switch expressing cells, and that increasing levels of TGF ⁇ do not further suppress cytokine production.
- increasing levels of TGF ⁇ increase the level of cytokines produced in cells expressing TGF ⁇ RII switches, having the greatest impact on IL-2 production.
- TFP-expressing T cells from Donor 22 expressing the constructs shown in Table 1 were expanded to day 10, as described above. Cells were suspended at concentration of 9 x 10 6 cell/mL in R10 media with 100U/mL IL-2. After 24 hours (on day 2), cells were washed 4x with 15mL of Serum Free Media and resuspended at a concentration of 1 x 10 5 cells in 100uL of serum-free media. On day 3, 200ng/mL, 10ng/mL, or 0.1ng/mL of TGF ⁇ was added.
- TGF ⁇ RII was detected on the surface of cells transduced with the vector encoding MH1e TFP and each of the TGF ⁇ RII switches.
- the levels of pSMAD2/3, as determined by flow cytometry after fixation, are shown in Figure 14. In the absence of TGF ⁇ , all TFP+ cells (VHH+) had similar levels of pSMAD2/3. At increasing levels of TGF ⁇ , the levels of pSMAD2/3 were shown to increase in TC-210 cells and in cells containing MH1e with the PD-1 switch. Expression of the TGF ⁇ RII switches suppressed this increase in pSMAD2/3, with the switch having the TGF ⁇ RII extracellular and transmembrane domains and the CD28 intracellular domain being particularly effective.
- pSMAD2/3 also increased in non-transduced cells and in TFP- cells from the cell populations transduced with TC-210 or MH1e with the PD-1 switch at increasing TGF ⁇ concentrations.
- TFP- cells from the cell populations transduced with the TGF ⁇ RII switches also showed a suppression of this increase in pSMAD2/3, indicating that the TGF ⁇ RII switches on TFP+ cells may act as a dominant negative receptor, also affecting pSMAD2/3 levels in TFP- cells.
- Figure 15 shows pSMAD2/3 levels in non-transduced CD4+ and CD8+ T cells, indicating that there is no significant difference in pSMAD2/3 signaling between CD4 and CD8+ T cells Example 6.
- T cells Phenotype of cells expressing TGF ⁇ RII Switch or DN TGF ⁇ RII TFP
- T cells were purified from healthy donors, activated, and transduced with lentivirus encoding a TFP as described above.
- T cells were transduced with vectors encoding an anti-MSLN TFP (TC-210); anti-MSLN TFP with PD-CD28 Switch (TC-510); anti-MSLN TFP and a TGF ⁇ RII Switch (41BB(IC) or CD28(IC)); anti-MSLN TFP and a full length TGF ⁇ RII (FL); or anti-MSLN TFP and a dominant negative TGF ⁇ RII lacking the functional kinase domain (DN).
- TC-210 anti-MSLN TFP
- TC-510 anti-MSLN TFP with PD-CD28 Switch
- TGF ⁇ RII Switch 41BB(IC) or CD28(IC)
- FL full length TGF ⁇ RII
- FL functional kina
- Non-transduced T cells were also included as a control (NT). Exemplary constructs used in the study are shown below in Table 2. Table 2: Constructs [0716] Expression of TFPs by transduced T cells was confirmed by flow cytometry, using an anti-VHH antibody. As shown in FIG.16A, binding of the anti-VHH antibody was detected in each of the TFP-transduced T cell groups, but not in non-transduced control T cells. The T cells transduced with a TGF ⁇ RII Switch, DN, or FL each exhibited similar VHH expression compared to TC-210 and TC-510 (FIG.16A).
- TGF ⁇ RII Switch In the cells transduced with the TGF ⁇ RII Switch, DN, and FL, VHH+ cells expressed high levels of TGF ⁇ RII (FIG.16B). The highest levels of surface TGF ⁇ RII were detected on the cells transduced with the DN construct; little TGF ⁇ RII was detected on the surface of TC-210 or TC-510 transduced cells.
- the CD4 and CD8 memory phenotype was determined by detecting surface levels of CD45RA and CCR7 by flow cytometry. CD4 and CD8 T cell memory phenotypes were similar among the groups, as shown in FIG.17 and FIG 18, respectively. The CD4:CD8 ratio of transduced cells was also determined by flow cytometry.
- Example 7 Functional assessment of cells transduced with TFP-TGF ⁇ RII Switch or TFP-DN TGF ⁇ RII [0718] In this study, T cell cytotoxicity against tumor cell lines, cytokine production, capacity for TGF ⁇ signaling, and ability to overcome TGF ⁇ -mediated suppression were assessed in the T cells transduced with vectors encoding the constructs shown above in Table 2.
- transduced cells were incubated with a tumor cell line at effector (transduced T cells) to target (tumor cell line) ratios (E:T) of 9:1, 3:1, or 1:1. After 24 hours, a cytotoxicity assay was carried out to determine % tumor lysis, and supernatants were collected to measure cytokine production.
- Target tumor cell lines included MSTO-MSLN (high MSLN expression), Suit-2 (low MSLN expression), and control C30 target cells (no MSLN expression). Prior to incubation with the tumor cell lines, transduced cells were normalized to 25-40% transduction.
- TGF ⁇ RII Switch and TGF ⁇ RII DN transduced cells exhibited higher IFN ⁇ , IL-2, GM-CSF, and TNF ⁇ production compared to TC-210 transduced cells after incubation with MSTO-MSLN or Suit- 2 cell lines, as shown in FIGs.21A and 21B.
- pSMAD2/3 levels were determined to assess levels of TGF ⁇ signaling in transduced T cells.
- the T cells were plated at 2 x 10 6 cells/mL in R10 in the presence of 100U/mL IL-2, washed three times, resuspended in media without serum, and plated with 100 ng/mL, 10 ng/mL, 1 ng/mL, 0.1 ng/mL, or 0.01 ng/mL of TGF ⁇ for 30 minutes. Cells were then fixed and stained for expression of CD4, CD8, VHH, and pSMAD2/3, and analyzed by flow cytometry.
- FIG.22 Representative histograms are provided in FIG.22 and show reduced pSMAD2/3 staining in VHH+ cells upon addition of TGF ⁇ in the TGF ⁇ RII Switch and TGF ⁇ RII DN transduced cells, compared to TC-210 and TC-510 transduced cells.
- pSMAD2/3 MFI in VHH+ and VHH- cells at increasing concentrations of TGF ⁇ are provided in FIG.23.
- TGF ⁇ RII Switch 41BB(IC) and CD28(IC)
- TGF ⁇ RII DN expressing cells exhibited significantly reduced TGF ⁇ signaling compared to TC-210, TC- 510, and FL cells.
- TGF ⁇ RII Switch and TGF ⁇ RII DN constructs acted as potent dominant negative receptors for TGF ⁇ signaling (FIG.23).
- TGF ⁇ RII Switch and TGF ⁇ RII DN constructs acted as potent dominant negative receptors for TGF ⁇ signaling (FIG.23).
- 1 x 10 5 cells were normalized to 40% VHH+ and co-cultured with 1 x 10 5 tumor cells in a 200 ⁇ L volume in R10 for 24 hours, with or without exogenous TGF ⁇ .
- TGF ⁇ RII Switch and TGF ⁇ RII DN constructs maintained cytokine production upon addition of exogenous TGF ⁇ (FIGs.24A and 24B) or when cultured with Suit2, a TGF ⁇ secreting cell line (FIG.25).
- TGF ⁇ RII Switch and TGF ⁇ RII DN constructs also maintained cytokine production upon addition of increasing levels of exogenous TGF ⁇ in the Suit2 culture.
- 2 x 10 5 T cells were normalized to 25% VHH+ and placed in a 96 well plate coated with 1ng/mL of recombinant mesothelin in R10, with or without exogenous TGF ⁇ .
- TGF ⁇ RII Switch and TGF ⁇ RII DN constructs blunted the TGF ⁇ -mediated suppression of MSLN cytokine production, in cells from two different donors.
- higher levels of exogenous TGF ⁇ increased cytokine production in both TGF ⁇ RII Switch and TGF ⁇ RII DN cells, compared to TC-210, TC-510, and TGF ⁇ RII FL cells (FIGs.28A and 28B).
- TGF ⁇ RII Switch and TGF ⁇ RII DN cells increased in cell number upon repeated stimulation with the TGF ⁇ -secreting cell line Suit2 (FIG.29). In contrast, TC-210 and TC-510 cells did not grow in the presence of Suit2 cells (FIG.29). In addition, both TGF ⁇ RII Switch and TGF ⁇ RII DN cells expressed cytokines upon co-culture with Suit2 cells (FIG.30).
- TGF ⁇ RII Switch and TGF ⁇ RII DN cells expressed high amounts of IFN ⁇ and GM-CSF on days 8, 12, and 16, in contrast to TC-210, TC-510, and TGF ⁇ RII FL cells, which expressed little to no cytokine at those time points (FIG.30).
- T cells expressing the TGF ⁇ RII Switch or TGF ⁇ RII DN constructs express extracellular TGF ⁇ RII, exhibit high cytotoxicity and cytokine production in response to tumor cells, have dominant negative TGF ⁇ RII signaling activity, produce higher levels of cytokines function in the presence of TGF ⁇ , and expand upon repeated stimulation with a TGF ⁇ secreting cell line.
- TGF ⁇ RII Switch and TGF ⁇ RII DN efficacy An in vivo study is conducted to assess efficacy of TGF ⁇ RII Switch and TGF ⁇ RII DN cells in a tumor cell model. NSG mice are implanted at Day -10 with 1 x 10 6 Suit2-luc cells. When tumor size reaches 80-150mm 3 , transduced T cells are injected. Exemplary cells tested in the study include TC-210, TGF ⁇ RII DN, and non-transduced (NT) control cells. Tumors are measured every 3-4 days to determine anti-tumor efficacy. Persistence of the cells is tracked by weekly bleeds, and blood, tumor, liver, and spleen are collected at the end of the study (e.g., Day 12) to assess tumor infiltration. Table 3. Antigen binding domain sequences.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical & Material Sciences (AREA)
- Epidemiology (AREA)
- Immunology (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Toxicology (AREA)
- Gastroenterology & Hepatology (AREA)
- Cell Biology (AREA)
- Biophysics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Molecular Biology (AREA)
- Biochemistry (AREA)
- Genetics & Genomics (AREA)
- Zoology (AREA)
- Engineering & Computer Science (AREA)
- Hematology (AREA)
- Oncology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Peptides Or Proteins (AREA)
Abstract
Provided herein are recombinant nucleic acids comprising a sequence encoding T cell receptor (TCR) fusion proteins (TFPs) and a dominant negative and/or switch polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof, cells comprising the recombinant nucleic acids, methods of using the recombinant nucleic acids and/or the cells, including the methods of treating diseases or conditions, including cancer.
Description
COMPOSITIONS AND METHODS FOR TCR REPROGRAMMING USING FUSION PROTEINS AND TGFβR SWITCH CROSS REFERENCE TO RELATED APPLICATIONS [0001] This application claims the benefit of priority to U.S. Provisional Application Nos. 63/180,292, filed on April 27, 2021, and 63/318,053, filed on March 9, 2022, each of which is hereby incorporated by reference in its entirety. SEQUENCE LISTING [0001.1] The instant application contains a Sequence Listing which has been submitted electronically in ASCII format and is hereby incorporated by reference in its entirety. Said ASCII copy, created on April 25, 2022, is named 48538752601_SL.txt and is 278,366 bytes in size. BACKGROUND OF THE INVENTION [0002] Most patients with hematological malignancies or with late-stage solid tumors are incurable with standard therapy. In addition, traditional treatment options often have serious side effects. Numerous attempts have been made to engage a patient’s immune system for rejecting cancerous cells, an approach collectively referred to as cancer immunotherapy. However, several obstacles make it rather difficult to achieve clinical effectiveness. Although hundreds of so-called tumor antigens have been identified, these are often derived from self and thus can direct the cancer immunotherapy against healthy tissue, or are poorly immunogenic. Furthermore, cancer cells use multiple mechanisms to render themselves invisible or hostile to the initiation and propagation of an immune attack by cancer immunotherapies. [0003] Recent developments using chimeric antigen receptor (CAR) modified autologous T cell therapy, which relies on redirecting genetically engineered T cells to a suitable cell- surface molecule on cancer cells, show promising results in harnessing the power of the immune system to treat B cell malignancies (see, e.g., Sadelain et al., Cancer Discovery 3:388-398 (2013)). The clinical results with CD19-specific CAR T cells (called CTL019) have shown complete remissions in patients suffering from chronic lymphocytic leukemia (CLL) as well as in childhood acute lymphoblastic leukemia (ALL) (see, e.g., Kalos et al., Sci Transl Med 3:95ra73 (2011), Porter et al., NEJM 365:725-733 (2011), Grupp et al., NEJM 368:1509-1518 (2013)). An alternative approach is the use of T cell receptor (TCR) alpha and beta chains selected for a tumor-associated peptide antigen for genetically engineering autologous T cells. These TCR chains will form complete TCR complexes and
provide the T cells with a TCR for a second defined specificity. Encouraging results were obtained with engineered autologous T cells expressing NY-ESO-1-specific TCR alpha and beta chains in patients with synovial carcinoma. [0004] Besides the ability for genetically modified T cells expressing a CAR or a second TCR to recognize and destroy respective target cells in vitro/ex vivo, successful patient therapy with engineered T cells may require the T cells to be capable of strong activation, expansion, persistence over time, effective tumor targeting, and, in case of relapsing disease, enabling a ‘memory’ response. SUMMARY OF THE INVENTION [0005] There is a dire need to improve genetically engineered T cells to more broadly act against various human malignancies and to enhance longevity of genetically engineered T cells to generate durable responses in cancer patients. [0006] Provided herein are recombinant nucleic acids comprising: a first nucleic acid sequence encoding a T cell receptor (TCR) fusion protein (TFP), wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain; and wherein the TCR subunit and the antigen binding domain are operatively linked, and a second nucleic acid sequence encoding a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof. In some embodiments, the second nucleic acid encodes a dominant negative TGFBr2 comprising a TGFBr2 extracellular domain or a functional fragment thereof. In some embodiments, the second nucleic acid encodes a switch polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof. [0007] In some embodiments, the TCR subunit further comprises a TCR intracellular domain. [0008] In some embodiments, the first nucleic acid sequence and the second nucleic acid sequence are operatively linked by a linker. [0009] In some embodiments, the linker comprises a protease cleavage site. [0010] In some embodiments, the protease cleavage site is a 2A cleavage site. [0011] In some embodiments, the 2A cleavage site is a T2A cleavage site or a P2A cleavage site. [0012] In some embodiments, the first nucleic acid sequence and the second nucleic acid sequence are present on different nucleic acid molecules. [0013] In some embodiments, the TGFBr2 extracellular domain comprises a sequence with at least 80% sequence identity to SEQ ID NO:271. In some embodiments, the TGFBr2 extracellular domain comprises a sequence according to SEQ ID NO: 271.
[0014] In some embodiments, the dominant negative TGFBr2 comprises a TGFBr2 extracellular domain and a TGFBr2 transmembrane domain and lacks a functional kinase domain. In some embodiments, the dominant negative TGFBr2 comprises a truncated TGFBr2, for example, a TGFBr2 with a truncated intracellular domain. In some embodiments, the dominant negative TGFBr2 comprises a TGFBr2 extracellular domain, a TGFBr2 transmembrane domain, and a truncated portion of a TGFBr2 intracellular domain. In some embodiments, the dominant negative TGFBr2 comprises a sequence with at least 80% sequence identity to SEQ ID NO: 68. In some embodiments, the dominant negative TGFBr2 comprises a sequence according to SEQ ID NO: 68. [0015] In some embodiments, the switch polypeptide further comprises a switch intracellular domain. [0016] In some embodiments, the TGFBr2 extracellular domain is operably linked to the switch intracellular domain. [0017] In some embodiments, the switch intracellular domain comprises an intracellular domain of a costimulatory polypeptide. [0018] In some embodiments, the costimulatory polypeptide is selected from the group consisting of CD28, 4-1BB, IL-15Ra, IL12R, IL18R, IL21R, OX40, CD2, CD27, CD5, ICAM-1, ICOS (CD278), GITR, CD30, CD40, BAFFR, HVEM, CD7, LIGHT, NKG2C, SLAMF7, NKp80, CD160, CD226, FcγRI, FcγRII, and FcγRIII. [0019] In some embodiments, the costimulatory polypeptide is CD28. [0020] In some embodiments, the costimulatory polypeptide is 4-1BB. [0021] In some embodiments, the costimulatory polypeptide is IL-15Ra. [0022] In some embodiments, the switch intracellular domain comprises a sequence with at least 80% sequence identity to SEQ ID NO:273 or SEQ ID NO:277. [0023] In some embodiments, the switch polypeptide further comprises a switch transmembrane domain. [0024] In some embodiments, the TGFBr2 extracellular domain is operably linked to the switch intracellular domain via the switch transmembrane domain. [0025] In some embodiments, the switch transmembrane domain is derived from a TGFBr2 transmembrane domain. [0026] In some embodiments, the switch transmembrane domain comprises a sequence with at least 80% sequence identity to SEQ ID NO:272. [0027] In some embodiments, the switch transmembrane domain is derived from a transmembrane domain of the costimulatory polypeptide.
[0028] In some embodiments, the switch transmembrane domain is derived from a transmembrane domain of CD28. [0029] In some embodiments, the switch transmembrane domain is derived from a transmembrane domain of 4-1BB. [0030] In some embodiments, the switch transmembrane domain is derived from a transmembrane domain of IL-15Ra. [0031] In some embodiments, the switch transmembrane domain comprises a sequence with at least 80% sequence identity to SEQ ID NO:275 or SEQ ID NO:279. [0032] In some embodiments, the switch polypeptide further comprises an additional intracellular domain. [0033] In some embodiments, the additional intracellular domain is operably linked to C- terminus of the switch intracellular domain. [0034] In some embodiments, the additional intracellular domain is derived from an intracellular domain of IL-15Ra. [0035] In some embodiments, the additional intracellular domain comprises a sequence with at least 80% sequence identity to SEQ ID NO:372 or SEQ ID NO:383. [0036] In some embodiments, the switch polypeptide comprises a transmembrane domain derived from a TGFBr2 transmembrane domain and an intracellular signaling domain of 4- 1BB. [0037] In some embodiments, the switch polypeptide comprises a transmembrane domain derived from a 4-1BB transmembrane domain and an intracellular signaling domain of 4- 1BB. [0038] In some embodiments, the switch polypeptide comprises a transmembrane domain derived from a TGFBr2 transmembrane domain and an intracellular signaling domain of CD28. [0039] In some embodiments, the switch polypeptide comprises a transmembrane domain derived from a CD28 transmembrane domain and an intracellular signaling domain of CD28. [0040] In some embodiments, the switch polypeptide comprises a sequence with at least 80% sequence identity to a sequence selected from SEQ ID NOs: 283, 284, 285, and 286. [0041] In some embodiments, the TFP functionally interacts with an endogenous TCR complex when expressed in a T cell. [0042] In some embodiments, the TCR intracellular domain comprises a stimulatory domain from an intracellular signaling domain of CD3 gamma, CD3 delta, or CD3 epsilon. [0043] In some embodiments, the TCR intracellular domain comprises an intracellular domain from TCR alpha, TCR beta, TCR gamma, or TCR delta.
[0044] In some embodiments, the antigen binding domain is connected to the TCR extracellular domain by a linker sequence. [0045] In some embodiments, the linker sequence is 120 amino acids in length or less. [0046] In some embodiments, the linker sequence comprises (G4S)n, wherein G is glycine, S is serine, and n is an integer from 1 to 10 (SEQ ID NO: 390). [0047] In some embodiments, n is an integer from 1 to 4. [0048] In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from the same TCR subunit. [0049] In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from TCR alpha. [0050] In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from TCR beta. [0051] In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from TCR gamma. [0052] In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from TCR delta. [0053] In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 epsilon. [0054] In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 delta. [0055] In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 gamma. [0056] In some embodiments, all three of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from the same TCR subunit. [0057] In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 epsilon. [0058] In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 delta. [0059] In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 gamma. [0060] In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain comprise the constant domain of TCR alpha. [0061] In some embodiments, the constant domain of TCR alpha is murine. [0062] In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain comprise the constant domain of TCR beta.
[0063] In some embodiments, the constant domain of TCR beta is murine. [0064] In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain comprise the constant domain of TCR gamma. [0065] In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain comprise the constant domain of TCR delta. [0066] In some embodiments, the antigen binding domain is a camelid antibody or binding fragment thereof. [0067] In some embodiments, the antigen binding domain is a murine antibody or binding fragment thereof. [0068] In some embodiments, the antigen binding domain is a human or humanized antibody or binding fragment thereof. [0069] In some embodiments, the antigen binding domain is a single-chain variable fragment (scFv) or a single domain antibody (sdAb) domain. [0070] In some embodiments, the antigen binding domain is a single domain antibody (sdAb). [0071] In some embodiments, the sdAb is a VH or VHH. [0072] In some embodiments, the antigen binding domain is selected from the group consisting of an anti-CD19 binding domain, an anti-B-cell maturation antigen (BCMA) binding domain, and an anti-mesothelin (MSLN) binding domain, an anti-CD20 binding domain, an anti-CD70 binding domain, anti-MUC16 binding domain, an anti-Nectin-4 binding domain, an anti-GPC3 binding domain, and an anti-TROP-2 binding domain. [0073] In some embodiments, a T cell expressing the TFP inhibits tumor growth when expressed in a T cell. [0074] In some embodiments, the recombinant nucleic acid as provided herein further comprises a leader sequence. In some embodiments, the leader sequence comprises SEQ ID NO: 270. [0075] In some embodiments, the recombinant nucleic acid is selected from the group consisting of a DNA and an RNA. [0076] In some embodiments, the recombinant nucleic acid is an mRNA. [0077] In some embodiments, the recombinant nucleic acid is a circRNA. [0078] In some embodiments, the recombinant nucleic acid comprises a nucleotide analog. [0079] In some embodiments, the nucleotide analog is selected from the group consisting of 2’-O-methyl, 2’-O-methoxyethyl (2’-O-MOE), 2’-O-aminopropyl, 2’-deoxy, T-deoxy-2’- fluoro, 2’-O-aminopropyl (2’-O-AP), 2'-O-dimethylaminoethyl (2’-O-DMAOE), 2’-O- dimethylaminopropyl (2’-O-DMAP), T-O-dimethylaminoethyloxyethyl (2’-O-DMAEOE),
2’-O-N-methylacetamido (2’-O-NMA) modified, a locked nucleic acid (LNA), an ethylene nucleic acid (ENA), a peptide nucleic acid (PNA), a 1’,5’- anhydrohexitol nucleic acid (HNA), a morpholino, a methylphosphonate nucleotide, a thiolphosphonate nucleotide, and a 2’-fluoro N3-P5’-phosphoramidite. [0080] In some embodiments, the recombinant nucleic acid as provided herein further comprises a promoter. [0081] In some embodiments, the recombinant nucleic acid is an in vitro transcribed nucleic acid. [0082] In some embodiments, the recombinant nucleic acid further comprises a sequence encoding a poly(A) tail. [0083] In some embodiments, the recombinant nucleic acid further comprises a 3’UTR sequence. [0084] In an aspect, the present disclosure provides a polypeptide encoded by the recombinant nucleic acid as provided herein. [0085] In an aspect, the present disclosure provides a vector comprising a recombinant nucleic acid as provided herein. [0086] In some embodiments, the vector is a lentiviral vector. [0087] In an aspect, the present disclosure provides a cell comprising the recombinant nucleic acid as provided herein, the polypeptide as provided herein, or the vector as provided herein. [0088] In some embodiments, the cell is a T cell. [0089] In some embodiments, the T cell is a human T cell. [0090] In some embodiments, the T cell is a CD8+ or CD4+ T cell. [0091] In some embodiments, the T cell is a human αβ T cell. [0092] In some embodiments, the T cell is a human γδ T cell. [0093] In some embodiments, the cell is a human NKT cell. [0094] In some embodiments, the cell is an allogeneic cell or an autologous cell.In some embodiments, the present disclosure provides a modified T cell or a population of modified T cells comprising a dominant negative TGFBr2 receptor and a T cell receptor (TCR) fusion protein (TFP), wherein the TFP comprises (a) a TCR subunit comprising (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain; and (b) an antigen binding domain, wherein the TCR subunit and the antigen binding domain are operatively linked. In some embodiments, the modified T cell or population of modified T cells comprises a dominant negative TGFBr2 comprising a sequence with at least 80% sequence identity to SEQ ID NO: 68. In some embodiments, the modified T cell or population of
modified T cells comprises a dominant negative TGFBr2 comprising a sequence according to SEQ ID NO: 68. [0095] In some embodiments, the present disclosure provides a modified T cell or a population of modified T cells comprising a TGFBr2 switch receptor and a T cell receptor (TCR) fusion protein (TFP), wherein the TFP comprises (a) a TCR subunit comprising (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain; and (b) an antigen binding domain, wherein the TCR subunit and the antigen binding domain are operatively linked. In some embodiments, the modified T cell or population of modified T cells comprises a switch receptor comprising an amino acid sequence with at least 80% sequence identity to any one of SEQ ID NOs: 283, 284, 285, or 286. In some embodiments, the modified T cell or population of modified T cells comprises a switch receptor comprising an amino acid sequence selected from the group consisting of SEQ ID Nos: 283, 284, 285, and 286. [0096] In some embodiments, anti-tumor efficacy of the cell is increased compared to a cell that does not comprise the second nucleic acid sequence encoding a polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a portion thereof. [0097] In some embodiments, expression of an exhaustion marker of the cell is decreased compared to a cell that comprises the first nucleic acid sequence and does not comprise the second nucleic acid sequence. [0098] In some embodiments, cytotoxicity of the cell is increased compared to a cell that comprises the first nucleic acid sequence and does not comprise the second nucleic acid sequence . [0099] In some embodiments, the cytokine production of the cell is increased compared to a cell that comprises the first nucleic acid sequence and does not comprise the second nucleic acid sequence. [0100] In some embodiments, the cytokine is selected from the group consisting of GM-CSF, IFNgamma, IL-2, and TNFalpha. [0101] In some embodiments, pSMAD2/3 signaling is decreased compared to a cell that comprises the first nucleic acid sequence and does not comprise the second nucleic acid sequence. [0102] In some embodiments, the cell comprises a population of cells. [0103] In an aspect, the present disclosure provides a pharmaceutical composition comprising the cell as provided herein and a pharmaceutically acceptable carrier.
[0104] In an aspect, the present disclosure provides a method of increasing an activity or cytotoxicity of a cell expressing a recombinant nucleic acid comprising a sequence encoding a T cell receptor (TCR) fusion protein (TFP), the method comprising expressing a dominant negative TGFBr2 in the cell, wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain; and wherein the TCR subunit and the antigen binding domain are operatively linked. [0105] In an aspect, the present disclosure provides a method of increasing an activity or cytotoxicity of a cell expressing a recombinant nucleic acid comprising a sequence encoding a T cell receptor (TCR) fusion protein (TFP), the method comprising expressing a switch polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof in the cell: wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain; and wherein the TCR subunit and the antigen binding domain are operatively linked. [0106] In some embodiments, the cell is the cell as provided herein. [0107] In an aspect, the present disclosure provides a method of treating a disease or a condition in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of the pharmaceutical composition as provided herein. [0108] In some embodiments, the disease or the condition is a cancer or a disease or a condition associated with expression of CD19, B-cell maturation antigen (BCMA), mesothelin (MSLN), CD20, CD70, MUC16, Trop-2, Nectin-4, or GPC3. [0109] In some embodiments, the cancer is a hematologic cancer selected from the group consisting of B-cell acute lymphoid leukemia (B-ALL), T cell acute lymphoid leukemia (T- ALL), acute lymphoblastic leukemia (ALL), chronic myelogenous leukemia (CML), chronic lymphocytic leukemia (CLL), B cell prolymphocytic leukemia, blastic plasmacytoid dendritic cell neoplasm, Burkitt’s lymphoma, diffuse large B cell lymphoma, follicular lymphoma, hairy cell leukemia, small cell-follicular lymphoma, large cell-follicular lymphoma, malignant lymphoproliferative conditions, MALT lymphoma, mantle cell lymphoma, Marginal zone lymphoma, multiple myeloma, myelodysplasia, myelodysplastic syndrome, non-Hodgkin’s lymphoma, plasmablastic lymphoma, plasmacytoid dendritic cell neoplasm, Waldenstrom macroglobulinemia, and preleukemia. [0110] In some embodiments, the cancer is mesothelioma, renal cell carcinoma, stomach cancer, breast cancer, lung cancer, ovarian cancer, prostate cancer, colon cancer, cervical cancer, brain cancer, liver cancer, pancreatic cancer, thyroid cancer, bladder cancer, ureter
cancer, kidney cancer, endometrial cancer, esophageal cancer, gastric cancer, thymic carcinoma or cholangiocarcinoma. [0111] In some embodiments, the subject is a human. INCORPORATION BY REFERENCE [0112] All publications, patents, and patent applications mentioned in this specification are herein incorporated by reference in their entireties and to the same extent as if each individual publication, patent, or patent application was specifically and individually indicated to be incorporated by reference. BRIEF DESCRIPTION OF THE DRAWINGS [0113] FIG.1 is a series of graphs showing expansion of T cells expressing the TFP constructs shown over 10 days. [0114] FIG.2 is a series of flow plots showing VHH (anti-MSLN TFP) expression (transduction efficiency) in T cells expressing the TFP constructs shown in a representative donor. Also shown are graph with average % VHH and VHH MFI in T cells expressing the TFP constructs shown averaged across three donors. [0115] FIG.3 is a series of graphs showing the CD4:CD8 T cell ratio in T cells expressing the TFP constructs shown in three donors. [0116] FIG 4 is a series of flow plots showing TGFβRII and VHH expression in T cells expressing the TFP constructs shown in a representative donor. Also shown is a graph with average TGFβRII MFI in T cells expressing the TFP constructs shown averaged across three donors. [0117] FIG.5 is a series of graphs showing the memory phenotype of T cells expressing the TFP constructs shown in three donors. [0118] FIG.6 is a series of graphs showing cytokine expression (IFNγ, GM-CSF, IL-2, and TNF-α) by T cells expressing the TFP constructs shown when contacted with plate bound mesothelin in the presence or absence of 25 ng/ml TGFβ, as described in Example 3. [0119] FIG.7 is a series of graphs showing cytokine expression (IL-2 and IFNγ) by T cells expressing the TFP constructs shown when contacted with plate bound mesothelin in the presence of TGFβ at the concentrations shown, as described in Example 3. [0120] FIG.8 is a series of graphs showing cytokine expression (IFNγ, GM-CSF, IL-2, and TNF-α) by T cells expressing the TFP constructs shown when contacted with plate bound
mesothelin and PD-L1 in the presence and absence of 25 ng/ml TGFβ, as described in Example 3. [0121] FIG.9 is a schematic illustration of the protocol described in Example 4. [0122] FIG.10 is a series of graphs showing IL-2 expression by T cells expressing the TFP constructs shown when contacted with MSTO-MSLN, OVCAR3, or C30 target cells, in the presence or absence of 10 ng/ml TGFβ, as described in Example 4. [0123] FIG.11 is a series of graphs showing cytokine expression (IFNγ, GM-CSF, IL-2, and TNF-α) by T cells expressing the TFP constructs shown when contacted with MSTO-MSLN in the presence of TGFβ at the concentrations shown, as described in Example 4. [0124] FIG.12 is a schematic illustration of the protocol described in Example 5. [0125] FIG.13 is a series of flow plots showing expression of TGFβRII, PD-1, and VHH expression in T cells expressing the TFP constructs shown at 10 days of expansion. [0126] FIG.14 is a series of graphs showing pSMAD2/3 staining in CD4+ and CD8+ T cells incubated with 0, 0.1, 10, or 200 ng/ml TGFβ, as described in Example 5. [0127] FIGs.15A and 15B are a series of graphs showing the level of pSMAD2/3 staining in T cells expressing the TFP constructs shown (VHH+) or untransduced cells from the same population (VHH-) incubated with 0, 0.1, 10, or 200 ng/ml TGFβ, as described in Example 5. [0128] FIGs.16A and 16B show VHH expression and TGFβRII expression in T cells expressing the TFP constructs. FIG.16A provides a series of flow plots showing CD3 and VHH expression in cells expressing the TFP constructs from a representative donor and a bar graph with the %VHH+ cells, averaged across six donors. FIG.16B provides a series of flow plots showing TGFβRII and VHH expression in cells from the representative donor and a bar graph showing the TGFβRII MFI in VHH+ cells, averaged across six donors. [0129] FIG.17 shows the memory phenotype of CD4+ T cells expressing the TFP constructs. The flow plots provide the CD45RA and CCR7 staining of VHH+ CD4+ cells from a representative donor. Also shown is a bar graph providing the frequencies of naïve, TCM, TEM, and TEMRA cells in the CD4+ T cell compartment of cells expressing the TFP constructs, from six donors. [0130] FIG.18 shows the memory phenotype of CD8+ T cells expressing the TFP constructs. The flow plots provide the CD45RA and CCR7 staining of VHH+ CD8+ cells from a representative donor. Also shown is a bar graph providing the frequencies of naïve, TCM, TEM, and TEMRA cells in the CD8+ T cell compartment of cells expressing the TFP constructs, from six donors. [0131] FIG.19 shows the CD4:CD8 T cell ratio in T cells expressing the TFP constructs. The flow plots provide the results from a representative donor. Also shown is a bar graph
providing the average ratio of CD4:CD8 T cells in T cells expressing the TFP constructs, from six donors. [0132] FIG.20 is a series of bar graphs showing cytotoxicity of T cells expressing the TFP constructs against tumor cell lines C30 (ovarian cancer; no expression of MSLN), MSTO- MSLN (mesothelioma cancer; high expression of MSLN), and Suit-2 (pancreatic cancer, low expression of MSLN), at the indicated effector to target ratios (E:T). [0133] FIG.21A and FIG.21B are each a series of bar graphs showing cytokine production by T cells expressing the TFP constructs upon incubation with MSTO-MSLN (top row) or Suit-2 (bottom row) cell lines at decreasing E:T. Production of IFNγ (left panels) and IL-2 (right panels) is shown in FIG.21A; and production of GM-CSF (left panels) and TNF-α (right panels) is shown in FIG.21B. [0134] FIG.22 is a representative set of histograms showing pSMAD2/3 staining in T cells expressing the indicated TFP constructs after incubation with TGFβ, as described in Example 6. In each histogram from a TFP group, the Δ indicates the shift in MFI of VHH+ cells upon addition of cytokine. [0135] FIG.23 shows the pSMAD2/3 staining in T cells expressing the indicated TFP constructs after incubation with 0.01, 0.1, 1, 10, or 100 ng/mL TGFβ. [0136] FIG.24A and FIG.24B are graphs showing cytokine expression by T cells expressing the TFP constructs shown, when contacted with MSTO-MSLN in the absence (0 ng/mL) or presence (1 ng/mL) of TGFβ. FIG.24A shows the expression of IFNγ (left panels), and IL-2 (right panels); FIG.24B shows the expression of GM-CSF (left panels) and TNF-α (right panels). [0137] FIG.25 shows the cytokine expression (IFNγ, GM-CSF, IL-2, and TNF-α) by T cells expressing the indicated TFP construct, when cultured with Suit-2, a TGFβ secreting cell line. [0138] FIG.26 is a series of graphs showing cytokine expression (IFNγ, GM-CSF, IL-2, and TNF-α) by T cells expressing the indicated TFP construct, when cultured with Suit-2 cells and in the presence of increasing concentrations of TGFβ. [0139] FIG.27A and FIG.27B are each a series of graphs showing the fold change in cytokine expression (GM-CSF, IFNγ, IL-2, and TNF-α) by T cells expressing the TFP constructs shown, when cultured with plate bound MSLN upon addition of 10 ng/mL TGFβ. The data in FIG.27A and FIG 27B are from two different donors and show the fold change in cytokine levels comparing the 0 ng/mL to 10 ng/mL TGFβ culture conditions. [0140] FIG.28A and FIG.28B are each a series of graphs showing cytokine expression (IFNγ, GM-CSF, IL-2, and TNF-α) by T cells expressing the TFP constructs shown, when
cultured with plate bound MSLN in the presence the indicated increasing concentrations of TGFβ. The data in FIG.28A and FIG 28B are from two different donors. [0141] FIG.29 shows the fold change in cell number (left panel) and VHH+ cell count (right panel) over time of T cells expressing the TFP constructs shown, upon repeated stimulation with Suit-2 cells. [0142] FIG.30 shows cytokine expression (IFNγ, GM-CSF, IL-2, and TNF-α) over time by T cells expressing the TFP constructs shown, upon repeated stimulation with Suit-2 cells. DETAILED DESCRIPTION OF THE INVENTION [0143] The present disclosure provides a recombinant nucleic acid comprising a first nucleic acid sequence encoding a T cell receptor (TCR) fusion protein (TFP). The TFP can comprise (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain. The TCR subunit and the antigen binding domain can be operatively linked. The recombinant nucleic acid can comprise a second nucleic acid sequence encoding (i) a dominant negative form of transforming growth factor beta receptor II (TGFBr2) comprising a TGFBr2 extracellular domain or a functional fragment thereof, or (ii) a switch polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof. The first nucleic acid sequence and the second nucleic acid sequence can be present on the same or different nucleic acid molecules. The present disclosure further provides a vector comprising the recombinant nucleic acid, a cell comprising the recombinant nucleic acid or the vector described herein, or a pharmaceutical composition comprising the cell (e.g., modified cell). [0144] The present disclosure also provides a method of increasing an activity or cytotoxicity of a cell expressing a recombinant nucleic acid comprising a sequence encoding a T cell receptor (TCR) fusion protein (TFP). The method can comprise expressing a dominant negative and/or switch polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof in the cell. The present disclosure also provides a method of treating a disease such as cancer using the cell described herein. In an aspect, the compositions and cells provided herein provide improved efficacy against tumors relative to modified T cells that lack a TGFBr2 switch or TGFBr2 dominant negative polypeptide or compositions thereof. Without wishing to be bound by theory, the cells provided herein are capable of resisting and/or overcoming TGFβ-mediated suppression and thus provide an effective therapy against solid tumors, given that TGFβ is commonly found in the tumor microenvironment (TME) and is a potent suppressor of effector T cell
function (Costanza et. al. IJC 145(6):1570-1584; 2019) (Flavell, et al. Nat Rev Immunol 10(8); 2010) (Gunderson et al, Nat Comm 11(1):1749; 2020). Definitions [0145] Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which the invention pertains. [0146] The term “a” and “an” refers to one or to more than one (i.e., to at least one) of the grammatical object of the article. By way of example, “an element” means one element or more than one element. [0147] As used herein, “about” can mean plus or minus less than 1 or 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 25, 30, or greater than 30 percent, depending upon the situation and known or knowable by one skilled in the art. [0148] As used herein the specification, “subject” or “subjects” or “individuals” may include, but are not limited to, mammals such as humans or non-human mammals, e.g., domesticated, agricultural or wild, animals, as well as birds, and aquatic animals. “Patients” are subjects suffering from or at risk of developing a disease, disorder or condition or otherwise in need of the compositions and methods provided herein. The term “subject” is intended to include living organisms in which an immune response can be elicited (e.g., mammals, human). [0149] As used herein, “treating” or “treatment” refers to any indicia of success in the treatment or amelioration of the disease or condition. Treating can include, for example, reducing, delaying or alleviating the severity of one or more symptoms of the disease or condition, or it can include reducing the frequency with which symptoms of a disease, defect, disorder, or adverse condition, and the like, are experienced by a patient. As used herein, “treat or prevent” is sometimes used herein to refer to a method that results in some level of treatment or amelioration of the disease or condition, and contemplates a range of results directed to that end, including but not restricted to prevention of the condition entirely. [0150] As used herein, “preventing” refers to the prevention of the disease or condition, e.g., tumor formation, in the patient. For example, if an individual at risk of developing a tumor or other form of cancer is treated with the methods of the present disclosure and does not later develop the tumor or other form of cancer, then the disease has been prevented, at least over a period of time, in that individual. [0151] As used herein, a “therapeutically effective amount” is the amount of a composition or an active component thereof sufficient to provide a beneficial effect or to otherwise reduce a detrimental non-beneficial event to the individual to whom the composition is
administered. By “therapeutically effective dose” herein is meant a dose that produces one or more desired or desirable (e.g., beneficial) effects for which it is administered, such administration occurring one or more times over a given period of time. The exact dose will depend on the purpose of the treatment, and will be ascertainable by one skilled in the art using known techniques (see, e.g. Lieberman, Pharmaceutical Dosage Forms (vols.1-3, 1992); Lloyd, The Art, Science and Technology of Pharmaceutical Compounding (1999); and Pickar, Dosage Calculations (1999)). [0152] As used herein, the terms “dominant negative” or “dominant negative receptor” refer to a gene product, protein, or polypeptide that is designed to reduce the effect of a negative signal. For example, a dominant negative polypeptide of the present disclosure reduces or eliminates the effect of a negative signal on a modified immune cell. In some embodiments, a dominant negative receptor of the present disclosure may bind a signaling molecule (e.g. TGFβ) by way of an extracellular domain that associates with the molecule, and reduce or eliminate the effect of the molecule by way of a truncated, mutated, or otherwise disrupted signaling or kinase domain. In some embodiments of the present disclosure, a modified immune cell comprising a dominant negative receptor may bind to a signaling molecule (e.g., TGFβ), and reduce the effect the molecule would have on the cell in the absence of the dominant negative receptor. [0153] As used herein, the term “fusion protein” relates to a protein which is made of polypeptide parts from different sources. Accordingly, in some embodiments, it may be also understood as a chimeric protein. In the context of the TGFBr2 fusion proteins as described herein, the term “fusion protein” is used interchangeably with the term “switch polypeptide” or “switch-receptor” or “TGFBr2 switch” and the like. Usually, fusion proteins are proteins created through the joining of two or more genes (or, for example, cDNAs) that originally coded for separate proteins. Translation of this fusion gene (or, for example, fusion cDNA) results in a single polypeptide, for example, with functional properties derived from each of the original proteins. In some embodiments, recombinant fusion proteins are created artificially by recombinant DNA technology for use in biological research or therapeutics. Further details to the exemplary production of the fusion protein of the present disclosure are described herein. [0154] In some embodiments, the fusion protein further comprises an epitope tag. An epitope tag as described herein can be a peptide epitope tag or a protein epitope tag. Examples of a peptide epitope tag includes, but are not limited to, 6X His (also known as His-tag or hexahistidine tag (SEQ ID NO: 391)), FLAG (e.g., 3X FLAG), HA, Myc, and V5. Examples of a protein epitope tag include, but are not limited to, green fluorescent protein (GFP),
glutathione-S-transferase (GST), β-galactosidase (β-GAL), Luciferase, Maltose Binding Protein (MBP), Red Fluorescence Protein (RFP), and Vesicular Stomatitis Virus Glycoprotein (VSV-G). In some embodiments, the fusion protein further comprises a FLAG tag. In some embodiments, the fusion protein further comprises a 3X FLAG tag. [0155] The term “TGFBr2 switch,” “TGFBr2 switch polypeptide,” “TGFBr2 fusion protein,” or “TGFBr2 switch receptor,” as used herein, refers to the TGFBr2 fusion proteins as described herein that receive an inhibitory signal by binding to, e.g., TGF-beta, and transform (e.g., “switch”) the signal via the co-stimulatory domain of the fusion protein into an activating signal. In some aspects, the TGFBr2 switch polypeptides provided herein lack a functional TGFBr2 kinase domain, and may function as a dominant negative receptor or exhibit functional characteristics of a dominant negative receptor. [0156] The term “TGFBr2” or “transforming growth factor beta receptor II,” also known as transforming growth factor, beta receptor II, TGF beta receptor 2, TGFBR2, TGFβRII, TGFBRII, AAT3, FAA3, LDS1B, LDS2, LDS2B, MFS2, RIIC, TAAD2, TGFR-2, TGFbeta-RII, transforming growth factor beta receptor 2, TBR-ii, TBRII, refers to a protein that is a member of the serine/threonine protein kinase family and the TGFB receptor subfamily. TGFBr2, when used in the context of the functional or wild-type protein, refers to a transmembrane protein that has a protein kinase domain, forms a heterodimeric complex with another receptor protein (e.g., TGFBr1), and binds TGF-beta. TGFBr2, as used herein, includes any of the recombinant or naturally-occurring forms of TGFBr2 or variants or homologs thereof that have or maintain TGFBr2 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring TGFBr2. In some embodiments, TGFBr2 is substantially identical to the protein identified by the UniProt reference number P37173 or a variant or homolog having substantial identity thereto. [0157] The term “TGFBr2 dominant negative,” “dominant negative TGFBr2,” “DN TGFBr2,” and the like, as used herein, refer to a TGFBr2 protein that lacks a functional kinase domain. In some embodiments, the dominant negative TGFBr2 comprises a TGFBr2 extracellular domain that is capable of binding to or capturing TGFβ; and lacks a kinase domain. For example, in some embodiments, the dominant negative TGFBr2 comprises a truncated or mutated intracellular domain that is not capable of phosphorylation of the GS domain of TGFBr1. Thus, in some embodiments, the dominant negative TGFBr2 is capable
of binding to TGFβ and complexing with TGFBr1, but is incapable of phosphorylating the GS domain of TGFBr1 such that the effect of TGFβ on a cell expressing the dominant negative TGFBr2 is reduced or eliminated. Exemplary dominant negative forms of TGFBr2 are provided herein. [0158] The term “GMCSFRa,” also known as CSF2RA, CD116, Cluster of Differentiation 116, CDw116, CSF2R, CSF2RAX, CSF2RAY, CSF2RX, CSF2RY, GM-CSF-R-alpha, GMCSFR, GMR, SMDP4, colony stimulating factor 2 receptor alpha subunit, alphaGMR, colony stimulating factor 2 receptor subunit alpha, GMR-alpha, GMCSFR-alpha, granulocyte-macrophage colony-stimulating factor receptor, as used herein, refers to a receptor for granulocyte-macrophage colony-stimulating factor, which stimulates the production of white blood cells. In some embodiments, GM-CSF and its receptor play a role in earlier stages of development. In some embodiments, GMCSFRa is associated with Surfactant metabolism dysfunction type 4. GMCSFRa, as used herein, includes any of the recombinant or naturally-occurring forms of GMCSFRa or variants or homologs thereof that have or maintain GMCSFRa activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring GMCSFRa. In some embodiments, GMCSFRa is substantially identical to the protein identified by the UniProt reference number P15509 or a variant or homolog having substantial identity thereto. [0159] The term “CD28,” also known as Cluster of Differentiation 28, CD28, Tp44, and CD28 molecule, as used herein, refers to a protein expressed on T cells that provides co- stimulatory signals required for T cell activation and survival. In some embodiments, CD28 is the receptor for CD80 (B7.1) and CD86 (B7.2) proteins. “CD28,” as used herein, includes any of the recombinant or naturally-occurring forms of CD28 or variants or homologs thereof that have or maintain CD28 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD28. In some embodiments, CD28 is substantially identical to the protein identified by the UniProt reference number P10747 or a variant or homolog having substantial identity thereto.
[0160] The term “2A” “2A self-cleaving peptide,” or “2A peptide,” as used herein, refers to a class of peptides, which can induce ribosomal skipping during translation of a protein in a cell. These peptides share a core sequence motif of DxExNPGP (SEQ ID NO: 392), and are found in a wide range of viral families. Exemplary members of 2A include, but are not limited to, P2A, E2A, F2A, and T2A. “T2A” refers to the 2A derived from thosea asigna virus, and the sequence is EGRGSLLTCGDVEENPGP (SEQ ID NO: 23). “P2A” (or “P2AW”) refers to the 2A derived from porcine teschovirus-12A, and the sequence is ATNFSLLKQAGDVEENPGP (SEQ ID NO: 269). “E2A” refers to the 2A derived from quine rhinitis A virus, and the sequence is QCTNYALLKLAGDVESNPGP (SEQ ID NO: 280). F2A is derived from foot-and-mouth disease virus 18, and the sequence is VKQTLNFDLLKLAGDVESNPGP (SEQ ID NO: 281). In some embodiments, adding the l linker “GSG” (Gly-Ser-Gly) on the N-terminal of a 2A peptide helps with efficiency. [0161] As used herein, a “T cell receptor (TCR) fusion protein” or “TFP,” as used herein, includes a recombinant polypeptide derived from the various polypeptides comprising the TCR that is generally capable of i) binding to a surface antigen on target cells and ii) interacting with other polypeptide components of the intact TCR complex, typically when co-located in or on the surface of a T cell. [0162] The term “stimulation,” as used herein, refers to a primary response induced by binding of a stimulatory domain or stimulatory molecule (e.g., a TCR/CD3 complex) with its cognate ligand thereby mediating a signal transduction event, such as, but not limited to, signal transduction via the TCR/CD3 complex. Stimulation can mediate altered expression of certain molecules, and/or reorganization of cytoskeletal structures, and the like. [0163] The term “stimulatory molecule” or “stimulatory domain,” as used herein, refers to a molecule or portion thereof expressed by a T cell that provides the primary cytoplasmic signaling sequence(s) that regulate primary activation of the TCR complex in a stimulatory way for at least some aspect of the T cell signaling pathway. In some embodiments, the primary signal is initiated by, for instance, binding of a TCR/CD3 complex with an MHC molecule loaded with peptide, and which leads to mediation of a T cell response, including, but not limited to, proliferation, activation, differentiation, and the like. A primary cytoplasmic signaling sequence (also referred to as a “primary signaling domain”) that acts in a stimulatory manner may contain a signaling motif which is known as immunoreceptor tyrosine-based activation motif or “ITAM”. Examples of an ITAM containing primary cytoplasmic signaling sequence that is of particular use in the invention includes, but is not limited to, those derived from TCR zeta, FcR gamma, FcR beta, CD3 gamma, CD3 delta, CD3 epsilon, CD5, CD22, CD79a, CD79b, CD278 (also known as “ICOS”) and CD66d.
[0164] The term “antigen presenting cell” or “APC,” as used herein, refers to an immune system cell such as an accessory cell (e.g., a B-cell, a dendritic cell, and the like) that displays a foreign antigen complexed with major histocompatibility complexes (MHC’s) on its surface. T cells may recognize these complexes using their T cell receptors (TCRs). APCs process antigens and present them to T cells. [0165] “Major histocompatibility complex (MHC) molecules,” as used herein, are typically bound by TCRs as part of peptide:MHC complex. The MHC molecule may be an MHC class I or II molecule. The complex may be on the surface of an antigen presenting cell, such as a dendritic cell or a B cell, or any other cell, including cancer cells, or it may be immobilized by, for example, coating on to a bead or plate. [0166] The “human leukocyte antigen system (HLA),” as used herein, refers to the gene complex which encodes major histocompatibility complex (MHC) in humans and includes HLA class I antigens (A, B & C) and HLA class II antigens (DP, DQ, & DR). HLA alleles A, B and C present peptides derived mainly from intracellular proteins, e.g., proteins expressed within the cell. [0167] During T cell development in vivo, T cells undergo a positive selection step to ensure recognition of self MHCs followed by a negative step to remove T cells that bind too strongly to MHC which present self-antigens. As a consequence, certain T cells and the TCRs they express will only recognize peptides presented by certain types of MHC molecules - i.e., those encoded by particular HLA alleles. This is known as HLA restriction. [0168] In some embodiments, one HLA allele of interest is HLA-A*0201, which is expressed in the vast majority (>50%) of the Caucasian population. Accordingly, TCRs which bind WT1 peptides presented by MHC encoded by HLA-A*0201 (i.e. are HLA- A*0201 restricted) are advantageous since an immunotherapy making use of such TCRs will be suitable for treating a large proportion of the Caucasian population. [0169] In some embodiments, other HLA- A alleles of interest are HLA-A*0101, HLA- A*2402, and HLA-A*0301. [0170] In some embodiments, widely expressed HLA-B alleles of interest are HLA-B*3501, HLA-B*0702 and HLA-B*3502. [0171] The term “intracellular signaling domain,” as used herein, refers to an intracellular portion of a molecule. The intracellular signaling domain generates a signal that promotes an immune effector function of the TFP containing cell, e.g., a modified T-T cell. Examples of immune effector function, e.g., in a modified T-T cell, include, but are not limited to, cytolytic activity and T helper cell activity, including the secretion of cytokines. In some embodiments, the intracellular signaling domain comprises a primary intracellular signaling
domain. Exemplary primary intracellular signaling domains include, but are not limited to, those derived from the molecules responsible for primary stimulation, or antigen dependent simulation. In some embodiments, the intracellular signaling domain comprises a costimulatory intracellular domain. Exemplary costimulatory intracellular signaling domains include, but are not limited to, those derived from molecules responsible for costimulatory signals, or antigen independent stimulation. [0172] In some embodiments, a primary intracellular signaling domain comprises an ITAM (“immunoreceptor tyrosine-based activation motif”). Examples of ITAM containing primary cytoplasmic signaling sequences include, but are not limited to, those derived from CD3 zeta, FcR gamma, FcR beta, CD3 gamma, CD3 delta, CD3 epsilon, CD5, CD22, CD79a, CD79b, and CD66d DAP10 and DAP12. [0173] The term “costimulatory molecule,” as used herein, refers to the cognate binding partner on a T cell that specifically binds with a costimulatory ligand, thereby mediating a costimulatory response by the T cell, such as, but not limited to, proliferation. In some embodiments, costimulatory molecules are cell surface molecules other than antigen receptors or their ligands that are required for an efficient immune response. Exemplary costimulatory molecules include, but are not limited to, an MHC class 1 molecule, BTLA and a Toll ligand receptor, as well as OX40, CD2, CD27, CD28, CD5, ICAM-1, LFA-1 (CD11a/CD18), 4-1BB (CD137), IL-15Ra, IL12R, IL18R, IL21R, ICOS (CD278), GITR, CD30, CD40, BAFFR, HVEM, CD7, LIGHT, NKG2C, SLAMF7, NKp80, CD160, CD226, FcγRI, FcγRII, and FcγRIII. In some embodiments, a costimulatory intracellular signaling domain is the intracellular portion of a costimulatory molecule. In some embodiments, a costimulatory molecule is represented in the following protein families: TNF receptor proteins, Immunoglobulin-like proteins, cytokine receptors, integrins, signaling lymphocytic activation molecules (SLAM proteins), and activating NK cell receptors. Examples of such molecules include CD27, CD28, 4-1BB (CD137), OX40, GITR, CD30, CD40, ICOS, BAFFR, HVEM, lymphocyte function-associated antigen-1 (LFA-1), CD2, CD7, LIGHT, NKG2C, SLAMF7, NKp80, CD160, B7-H3, and a ligand that specifically binds with CD83, IL-15Ra, IL12R, IL18R, IL21R, CD27, CD5, ICAM-1, CD7, CD226, FcγRI, FcγRII, FcγRIII, and the like. In some embodiments, the intracellular signaling domain comprises the entire intracellular portion or the entire native intracellular signaling domain of the molecule from which it is derived, or a functional fragment thereof. [0174] The term “4-1BB,” as used herein, refers to a member of the TNFR superfamily with an amino acid sequence provided as GenBank Acc. No. AAA62478.2, or the equivalent residues from a non-human species, e.g., mouse, rodent, monkey, ape and the like; and a “4-
1BB costimulatory domain,” as used herein, refers to amino acid residues 214-255 of GenBank Acc. No. AAA62478.2, or the equivalent residues from a non-human species, e.g., mouse, rodent, monkey, ape and the like. “4-1BB,” also known as TNFRSF9, 4-1BB, CD137, Cluster of Differentiation 137, CDw137, ILA, tumor necrosis factor receptor superfamily member 9, and TNF receptor superfamily member 9, as used herein, includes any of the recombinant or naturally-occurring forms of 4-1BB or variants or homologs thereof that have or maintain 4-1BB activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring 4-1BB. In some embodiments, 4-1BB is substantially identical to the protein identified by the UniProt reference number Q07011 or a variant or homolog having substantial identity thereto. [0175] The term “antibody,” as used herein, refers to a protein, or polypeptide sequences derived from an immunoglobulin molecule, which specifically binds to an antigen. Antibodies can be intact immunoglobulins of polyclonal or monoclonal origin, or fragments thereof and can be derived from natural or from recombinant sources. [0176] The terms “antibody fragment,” as used herein, refers to at least one portion of an antibody, or recombinant variants thereof, that contains the antigen binding domain, i.e., an antigenic determining variable region of an intact antibody, that is sufficient to confer recognition and specific binding of the antibody fragment to a target, such as an antigen and its defined epitope. Examples of antibody fragments include, but are not limited to, Fab, Fab’, F(ab’)2, and Fv fragments, single-chain (sc)Fv (“scFv”) antibody fragments, linear antibodies, single domain antibodies such as sdAb (either VL or VH), camelid VHH domains, and multi-specific antibodies formed from antibody fragments. [0177] The term “scFv,” as used herein, refers to a fusion protein comprising at least one antibody fragment comprising a variable region of a light chain and at least one antibody fragment comprising a variable region of a heavy chain, wherein the light and heavy chain variable regions are contiguously linked via a short flexible polypeptide linker, and capable of being expressed as a single polypeptide chain, and wherein the scFv retains the specificity of the intact antibody from which it is derived. [0178] The term “Heavy chain variable region” or “VH” with regard to an antibody, as used herein, refers to the fragment of the heavy chain that contains three CDRs interposed between flanking stretches known as framework regions. These framework regions are
generally more highly conserved than the CDRs and form a scaffold to support the CDRs. A camelid “VHH” domain, as used herein, refers to a heavy chain comprising a single variable antibody domain. [0179] Unless specified, as used herein a scFv may have the VL and VH variable regions in either order, e.g., with respect to the N-terminal and C-terminal ends of the polypeptide. In some embodiments, the scFv may comprise VL-linker-VH or may comprise VH-linker-VL. [0180] In some embodiments, the portion of the TFP composition of the disclosure comprising an antibody or antibody fragment thereof may exist in a variety of forms where the antigen binding domain is expressed as part of a contiguous polypeptide chain including, for example, a single domain antibody fragment (sdAb), or a single chain antibody (scFv) derived from a murine, humanized or human antibody (Harlow et al., 1999, In: Using Antibodies: A Laboratory Manual, Cold Spring Harbor Laboratory Press, N.Y.; Harlow et al., 1989, In: Antibodies: A Laboratory Manual, Cold Spring Harbor, N.Y.; Houston et al., 1988, Proc. Natl. Acad. Sci. USA 85:5879-5883; Bird et al., 1988, Science 242:423-426). In one aspect, the antigen binding domain of a TFP composition of the disclosure comprises an antibody fragment. In a further aspect, the TFP comprises an antibody fragment that comprises a scFv or a sdAb. [0181] The term “recombinant antibody,” as used herein, refers to an antibody that is generated using recombinant DNA technology, such as, for example, an antibody expressed by a bacteriophage or yeast expression system. The term should also be construed to mean an antibody which has been generated by the synthesis of a DNA molecule encoding the antibody and which DNA molecule expresses an antibody protein, or an amino acid sequence specifying the antibody, wherein the DNA or amino acid sequence has been obtained using recombinant DNA or amino acid sequence technology which is available and well known in the art. [0182] The term “antigen” or “Ag,” as used herein, refers to a molecule that is capable of being bound specifically by an antibody, or otherwise provokes an immune response. In some embodiments, this immune response may involve either antibody production, or the activation of specific immunologically-competent cells, or both. [0183] The skilled artisan will understand that any macromolecule, including virtually all proteins or peptides, can serve as an antigen. Furthermore, antigens can be derived from recombinant or genomic DNA. A skilled artisan will understand that any DNA, which comprises a nucleotide sequences or a partial nucleotide sequence encoding a protein that elicits an immune response therefore encodes an “antigen” as that term is used herein. Furthermore, one skilled in the art will understand that an antigen need not be encoded solely
by a full length nucleotide sequence of a gene. It is readily apparent that the present disclosure includes, but is not limited to, the use of partial nucleotide sequences of more than one gene and that these nucleotide sequences are arranged in various combinations to encode polypeptides that elicit the desired immune response. Moreover, a skilled artisan will understand that an antigen need not be encoded by a “gene” at all. It is readily apparent that an antigen can be generated synthesized or can be derived from a biological sample, or might be macromolecule besides a polypeptide. Such a biological sample can include, but is not limited to a tissue sample, a tumor sample, a cell or a fluid with other biological components. [0184] The term “CD3” or “Cluster of Differentiation 3,” as used herein, refers to a protein complex and T cell co-receptor that is involved in activating both the cytotoxic T cell and T helper cells. In some embodiments, it is composed of four distinct chains. For example, in some embodiments, the complex contains a CD3γ chain, a CD3δ chain, and two CD3ε chains in mammals. [0185] “CD3ε,” “CD3ε chain,” or “T-cell surface glycoprotein CD3 epsilon chain,” as used herein, includes any of the recombinant or naturally-occurring forms of CD3ε or variants or homologs thereof that have or maintain CD3ε activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD3ε. In some embodiments, CD3ε is substantially identical to the protein identified by the UniProt reference number P07766 or a variant or homolog having substantial identity thereto. [0186] “CD3δ,” “CD3δ chain,” or “T-cell surface glycoprotein CD3 delta chain,” as used herein, includes any of the recombinant or naturally-occurring forms of CD3δ or variants or homologs thereof that have or maintain CD3δ activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD3δ. In some embodiments, CD3δ is substantially identical to the protein identified by the UniProt reference number P04234 or a variant or homolog having substantial identity thereto. [0187] “CD3γ,” “CD3γ chain,” or “T-cell surface glycoprotein CD3 gamma chain,” as used herein, includes any of the recombinant or naturally-occurring forms of CD3γ or variants or
homologs thereof that have or maintain CD3γ activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD3γ. In some embodiments, CD3γ is substantially identical to the protein identified by the UniProt reference number P09693 or a variant or homolog having substantial identity thereto. [0188] As used herein, the term “CD19,” also known as B-lymphocyte antigen CD19, B4, CVID3, and CD19 molecule, refers to the Cluster of Differentiation 19 protein, which is an antigenic determinant detectable on B cell leukemia precursor cells, other malignant B cells and most cells of the normal B cell lineage. CD19, as used herein, includes any of the recombinant or naturally-occurring forms of CD19 or variants or homologs thereof that have or maintain CD19 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD19. In some embodiments, CD19 is substantially identical to the protein identified by the UniProt reference number P15391 or a variant or homolog having substantial identity thereto. [0189] As used herein, the term “BCMA” refers to the B-cell maturation antigen, also known as tumor necrosis factor receptor superfamily member 17 (TNFRSF17), Cluster of Differentiation 269 protein (CD269), BCM, TNFRSF13A, tumor necrosis factor receptor superfamily member 17, and TNF receptor superfamily member 17, which is a protein that in humans is encoded by the TNFRSF17 gene. TNFRSF17 is a cell surface receptor of the TNF receptor superfamily which recognizes B-cell activating factor (BAFF) (see, e.g., Laabi et al., EMBO 11 (11): 3897–904 (1992). This receptor is expressed in mature B lymphocytes, and may be important for B-cell development and autoimmune response. BCMA, as used herein, includes any of the recombinant or naturally-occurring forms of BCMA or variants or homologs thereof that have or maintain BCMA activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared
to a naturally occurring BCMA. In some embodiments, BCMA is substantially identical to the protein identified by the UniProt reference number Q02223 or a variant or homolog having substantial identity thereto. [0190] As used herein, the term “CD16,” also known as FcγRIII, refers to a cluster of differentiation molecule found on the surface of natural killer cells, neutrophil polymorphonuclear leukocytes, monocytes, and macrophages. CD16 has been identified as Fc receptors FcγRIIIa (CD16a) and FcγRIIIb (CD16b), which participate in signal transduction. In some embodiments, CD16 is a molecule of the immunoglobulin superfamily (IgSF) involved in antibody-dependent cellular cytotoxicity (ADCC). CD16, as used herein, includes any of the recombinant or naturally-occurring forms of CD16 or variants or homologs thereof that have or maintain CD16 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD16. In some embodiments, CD16 is substantially identical to the protein identified by the UniProt reference number P08637 (CD16a) or a variant or homolog having substantial identity thereto or the protein identified by the UniProt reference number O7501 (CD16b) or a variant or homolog having substantial identity thereto. [0191] As used herein, the term “NKG2D,” also known as KLRK1, CD314, D12S2489E, KLR, NKG2-D, NKG2D, natural killer group 2D, killer cell lectin-like receptor K1, and killer cell lectin like receptor K1, refers to a transmembrane protein belonging to the CD94/NKG2 family of C-type lectin-like receptors. In some embodiments, in humans, NKG2D is expressed by NK cells, γδ T cells and CD8+ αβ T cells. In some embodiments, NKG2D recognizes induced-self proteins from MIC and RAET1/ULBP families which appear on the surface of stressed, malignant transformed, and infected cells. NKG2D, as used herein, includes any of the recombinant or naturally-occurring forms of NKG2D or variants or homologs thereof that have or maintain NKG2D activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring NKG2D. In some embodiments, NKG2D is substantially identical to the protein identified by the UniProt reference number P26718 or a variant or homolog having substantial identity thereto.
[0192] The term “mesothelin (MSLN),” also known as MPF and SMRP, refers to a tumor differentiation antigen that is normally present on the mesothelial cells lining the pleura, peritoneum and pericardium. In some embodiments, mesothelin is over-expressed in several human tumors, including mesothelioma and ovarian and pancreatic adenocarcinoma. MSLN, as used herein, includes any of the recombinant or naturally-occurring forms of MSLN or variants or homologs thereof that have or maintain MSLN activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring MSLN. In some embodiments, MSLN is substantially identical to the protein identified by the UniProt reference number Q13421 or a variant or homolog having substantial identity thereto. [0193] The term “tyrosine-protein kinase transmembrane receptor ROR1”, also known as ROR1, neurotrophic tyrosine kinase, receptor-related 1 (NTRKR1), dJ537F10.1, receptor tyrosine kinase-like orphan receptor 1, and receptor tyrosine kinase like orphan receptor 1, refers to a member of the receptor tyrosine kinase-like orphan receptor (ROR) family. It plays a role in metastasis of cancer. ROR1, as used herein, includes any of the recombinant or naturally-occurring forms of ROR1 or variants or homologs thereof that have or maintain ROR1 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring ROR1. In some embodiments, ROR1 is substantially identical to the protein identified by the UniProt reference number Q01973 or a variant or homolog having substantial identity thereto. [0194] The term “MUC16”, also known as mucin 16, cell-surface associated, ovarian cancer- related tumor marker CA125, CA-125 (cancer antigen 125, carcinoma antigen 125, or carbohydrate antigen 125), mucin 16, and CA125, refers to a membrane-tethered mucin that contains an extracellular domain at its amino terminus, a large tandem repeat domain, and a transmembrane domain with a short cytoplasmic domain. In some embodiments, products of this gene have been used as a marker for different cancers, with higher expression levels associated with poorer outcomes. MUC16, as used herein, includes any of the recombinant or naturally-occurring forms of MUC16 or variants or homologs thereof that have or maintain MUC16 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99%
or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring MUC16. In some embodiments, MUC16 is substantially identical to the protein identified by the UniProt reference number Q8WXI7 or a variant or homolog having substantial identity thereto. [0195] The term “CD22,” also known as cluster of differentiation-22, sialic acid binding Ig- like lectin 2, SIGLEC-2, SIGLEC2, CD22 molecule, T cell surface antigen leu-14, and B cell receptor CD22, refers to a protein that mediates B cell/B cell interactions, and is thought to be involved in, e.g., the localization of B cells in lymphoid tissues. In some embodiments, CD22 is associated with diseases including, but not limited to, refractory hematologic cancer and hairy cell leukemia. An exemplary fully human anti-CD22 monoclonal antibody (“M971”) suitable for use with the methods as described herein is described, e.g., in Xiao et al., MAbs.2009 May-Jun; 1(3): 297–303. CD22, as used herein, includes any of the recombinant or naturally-occurring forms of CD22 or variants or homologs thereof that have or maintain CD22 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD22. In some embodiments, CD22 is substantially identical to the protein identified by the UniProt reference number P20273 or a variant or homolog having substantial identity thereto. [0196] “Programmed cell death protein 1,” also known as PD-1, CD279 (cluster of differentiation 279), PDCD1, PD1, SLEB2, hPD-1, hSLE1, and Programmed cell death 1, refers to a protein on the surface of cells that has a role in regulating the immune system's response to the cells of the human body by down-regulating the immune system and promoting self-tolerance by suppressing T cell inflammatory activity. This prevents autoimmune diseases, but it can also prevent the immune system from killing cancer cells. PD-1 is an immune checkpoint and guards against autoimmunity, e.g., through two mechanisms. First, it promotes apoptosis (programmed cell death) of antigen-specific T-cells in lymph nodes. Second, it reduces apoptosis in regulatory T cells (anti-inflammatory, suppressive T cells). PD-1 is a cell surface receptor that belongs to the immunoglobulin superfamily and is expressed on T cells and pro-B cells. PD-1 binds two ligands, PD-L1 and
PD-L2. PD-1, as used herein, includes any of the recombinant or naturally-occurring forms of PD-1 or variants or homologs thereof that have or maintain PD-1 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring PD-1. In some embodiments, PD-1 is substantially identical to the protein identified by the UniProt reference number Q15116 or a variant or homolog having substantial identity thereto. [0197] “Programmed death-ligand 1 (PD-L1),” also known as cluster of differentiation 274, CD274, B7 homolog 1, B7-H, B7-H1, B7H1, PDCD1L1, PDCD1LG1, PDL1, hPD-L1, and CD274 molecule, refers to a 40kDa type 1 transmembrane protein. In some embodiments, PD-L1 may play a major role in suppressing the adaptive arm of immune system during particular events such as, e.g., pregnancy, tissue allografts, autoimmune disease and other disease states such as, e.g., hepatitis. Normally the adaptive immune system reacts to antigens that are associated with immune system activation by exogenous or endogenous danger signals. In turn, clonal expansion of antigen-specific CD8+ T cells and/or CD4+ helper cells is propagated. The binding of PD-L1 to the inhibitory checkpoint molecule PD-1 transmits an inhibitory signal based on interaction with phosphatases (SHP-1 or SHP-2) via Immunoreceptor Tyrosine-Based Switch Motif (ITSM) motif. This reduces the proliferation of antigen-specific T-cells in lymph nodes, while simultaneously reducing apoptosis in regulatory T cells (anti-inflammatory, suppressive T cells) - further mediated by a lower regulation of the gene Bcl-2. PD-L1, as used herein, includes any of the recombinant or naturally-occurring forms of PD-L1 or variants or homologs thereof that have or maintain PD-L1 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring PD-L1. In some embodiments, PD-L1 is substantially identical to the protein identified by the UniProt reference number Q9NZQ7 or a variant or homolog having substantial identity thereto. [0198] The “CD79α (Cluster of Differentiation 79α)” and “CD79β (Cluster of Differentiation 79β)” genes encode proteins that make up the B lymphocyte antigen receptor, a multimeric complex that includes the antigen-specific component, surface immunoglobulin (Ig). Surface Ig non-covalently associates with two other proteins, Ig-alpha and Ig-beta (encoded by
CD79α and its paralog CD79β, respectively) which are necessary for expression and function of the B-cell antigen receptor. Functional disruption of this complex can lead to, e.g., human B-cell chronic lymphocytic leukemias. CD79α protein, as used herein, includes any of the recombinant or naturally-occurring forms of CD79α protein or variants or homologs thereof that have or maintain CD79α protein activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD79α protein. In some embodiments, CD79α protein is substantially identical to the protein identified by the UniProt reference number P11912 or a variant or homolog having substantial identity thereto. CD79β protein, as used herein, includes any of the recombinant or naturally-occurring forms of CD79β protein or variants or homologs thereof that have or maintain CD79β protein activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD79β protein. In some embodiments, CD79β protein is substantially identical to the protein identified by the UniProt reference number P40259 or a variant or homolog having substantial identity thereto. [0199] “B cell activating factor” or “BAFF,” also known as tumor necrosis factor ligand superfamily member 13B, TNFSF13B, BLYS, CD257, DTL, TALL-1, TALL1, THANK, TNFSF20, ZTNF4, TNLG7A, tumor necrosis factor superfamily member 13b, and TNF superfamily member 13b, refers to a cytokine that belongs to the tumor necrosis factor (TNF) ligand family. This cytokine is a ligand for receptors TNFRSF13B/TACI, TNFRSF17/BCMA, and TNFRSF13C/BAFF-R. This cytokine is expressed in B cell lineage cells, and acts as a potent B cell activator. In some embodiments, BAFF plays an important role in the proliferation and differentiation of B cells. BAFF, as used herein, includes any of the recombinant or naturally-occurring forms of BAFF or variants or homologs thereof that have or maintain BAFF activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally
occurring BAFF. In some embodiments, BAFF is substantially identical to the protein identified by the UniProt reference number Q9Y275 or a variant or homolog having substantial identity thereto. [0200] “CD70,” also known as CD27LG and TNFSF7, as referred herein, refers to a cytokine that is the ligand for CD27. The CD70-CD27 pathway plays an important role in the generation and maintenance of T cell immunity, in particular, during antiviral responses. Upon CD27 binding, CD70 induces the proliferation of co-stimulated T-cells and enhances the generation of cytolytic T-cells. CD70, as referred herein, includes any of the recombinant or naturally-occurring forms of CD70 or variants or homologs thereof that have or maintain CD70 activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring CD70. In some embodiments, CD70 is substantially identical to the protein identified by the UniProt reference number P32970 or a variant or homolog having substantial identity thereto. [0201] The term “interleukin 15 receptor” or “IL-15R” refers to a type I cytokine receptor that IL-15 binds to and signals through. In some embodiments, IL-15R is composed of three subunits: IL-15 receptor alpha chain (“IL-15Rα” or CD215), IL-2 receptor beta chain (“IL- 2Rβ” or CD122) and IL-2 receptor gamma/the common gamma chain (“IL-2Rγ/γc” or CD132). For example, in some embodiments, human IL-15Rα precursor protein has a 30 amino acid signal peptide, a 175 amino acid extracellular domain, a 23 amino acid single membrane-spanning transmembrane stretch, and a 39 amino acid cytoplasmic (or intracellular) domain and contains N- and O-linked glycosylation sites. In some embodiments, IL-15Rα contains a Sushi domain (amino acid 31-95), which is essential for IL-15 binding. In some embodiments, IL-15Rα exists as a soluble form (sIL-15Rα). In some embodiments, sIL-15Rα is constitutively generated from the transmembrane receptor through a defined proteolytic cleavage, and this process can be enhanced by certain chemical agents, such as PMA. In some embodiments, the human sIL-15Rα, about 42 kDa in size, may prolong the half-life of IL-15 or potentiate IL-15 signaling through IL-15 binding and IL- 2Rβ/γc heterodimer. Although IL-15R shares subunits with IL-2R that contain the cytoplasmic motifs required for signal transduction, in some embodiments, IL-15 signaling has separate biological effects in vivo apart from many biological activities overlapping with IL-2 signaling due to IL-15Rα subunit that is unique to IL-15R, availability and concentration of IL-15, the kinetics and affinity of IL-15-IL-15Rα binding. In some
embodiments, IL-15 binds to IL-15Rα specifically with high affinity, which then associates with a complex composed of IL-2Rβ and IL-2Rγ/γc subunits, expressed on the same cell (“cis-presentation”) or on a different cell (“trans-presentation”). In some embodiments, the interaction between IL-15 and IL-15Rα is independent of the complex composed of IL-2Rβ and IL-2Rγ/γc subunits. In some embodiments, IL-15 binding to the IL-2Rβ/γc heterodimeric receptor induces JAK1 activation that phosphorylates STAT3 via the beta chain, and JAK3 activation that phosphorylates STAT5 via the gamma chain. In some embodiments, the IL- 15/IL-15R interaction modulates T-cell development and homeostasis in memory CD8+ T- cell. In some embodiments, the IL-15/IL-15R interaction also modulates NK cell development, maintenance, expansion and activities. [0202] “IL-15Rα,” also known as CD215, IL-15 receptor subunit alpha, IL-15R-alpha, IL- 15RA, and Interleukin-15 receptor subunit alpha, as used herein, includes any of the recombinant or naturally-occurring forms of IL-15Rα or variants or homologs thereof that have or maintain IL-15Rα activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring IL-15Rα. In some embodiments, IL-15Rα is substantially identical to the protein identified by the UniProt reference number Q13261 or a variant or homolog having substantial identity thereto. [0203] “IL-2Rβ,” also known as CD122, IL-2 receptor subunit beta, IL-2R subunit beta, IL- 2RB, P70-75, IMD63, and Interleukin-2 receptor subunit beta, as used herein, includes any of the recombinant or naturally-occurring forms of IL-2Rβ or variants or homologs thereof that have or maintain IL-2Rβ activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring IL-2Rβ. In some embodiments, IL-2Rβ is substantially identical to the protein identified by the UniProt reference number P14784 or a variant or homolog having substantial identity thereto. [0204] “IL-2 receptor gamma/the common gamma chain,” also known as IL-2Rγ/γc, IL2RG, CIDX, IL-2RG, IMD4, P64, SCIDX, SCIDX1, interleukin 2 receptor subunit gamma, or CD132, as used herein, includes any of the recombinant or naturally-occurring forms of IL-
2Rγ/γc or variants or homologs thereof that have or maintain IL-2Rγ/γc activity (e.g., at least 40% 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or 100% activity). In some aspects, the variants or homologs have at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% or 100% amino acid sequence identity across the whole sequence or a portion of the sequence (e.g., a 50, 100, 150 or 200 continuous amino acid portion) compared to a naturally occurring IL-2Rγ/γc. In some embodiments, IL- 2Rγ/γc is substantially identical to the protein identified by the UniProt reference number P31785 or a variant or homolog having substantial identity thereto. [0205] In some embodiments, IL-15Rα cytoplasmic (or intracellular) domain comprises amino acids 229-267 of IL-15Rα protein. In some embodiments, IL-15Rα cytoplasmic (or intracellular) domain comprises a sequence of SEQ ID NO: 372. In some embodiments, IL- 15Rα Sushi domain comprises amino acids 31-95 of IL-15Rα protein. In some embodiments, IL-15Rα Sushi domain comprises a sequence of SEQ ID NO: 382. In some embodiments, IL- 15Rα comprises the transmembrane domain and the cytoplasmic (intracellular) domain of IL- 15Rα protein. In some embodiments, IL-15Rα comprises amino acids 96-267 of IL-15Rα protein. In some embodiments, IL-15Rα comprises a sequence of SEQ ID NO: 383. In some embodiments, sIL-15Rα comprises amino acids 21-205 of IL-15Rα protein. In some embodiments, sIL-15Rα comprises a sequence of SEQ ID NO: 379. [0206] SEQ ID NO: 372 (IL-15Rα intracellular domain) KSRQTPPLASVEMEAMEALPVTWGTSSRDEDLENCSHHL [0207] SEQ ID NO: 379 (Soluble IL-15Rα (sIL-15Rα)) ITCPPPMSVEHADIWVKSYSLYSRERYICNSGFKRKAGTSSLTECVLNKATNVAHWT TPSLKCIRDPALVHQRPAPPSTVTTAGVTPQPESLSPSGKEPAASSPSSNNTAATTAAI VPGSQLMPSKSPSTGTTEISSHESSHGTPSQTTAKNWELTASASHQPPGVYPQGHSDT T [0208] SEQ ID NO: 382 (IL-15 Sushi domain) ITCPPPMSVEHADIWVKSYSLYSRERYICNSGFKRKAGTSSLTECVLNKATNVAHWT TPSLKCIR [0209] SEQ ID NO: 383 (IL-15Rα region downstream of Sushi domain) DPALVHQRPAPPSTVTTAGVTPQPESLSPSGKEPAASSPSSNNTAATTAAIVPGSQLM PSKSPSTGTTEISSHESSHGTPSQTTAKNWELTASASHQPPGVYPQGHSDTTVAISTST VLLCGLSAVSLLACYLKSRQTPPLASVEMEAMEALPVTWGTSSRDEDLENCSHHL [0210] SEQ ID NO: 386 (IL-15Rα full-length protein sequence) MAPRRARGCRTLGLPALLLLLLLRPPATRGITCPPPMSVEHADIWVKSYSLYSRERYI CNSGFKRKAGTSSLTECVLNKATNVAHWTTPSLKCIRDPALVHQRPAPPSTVTTAGV
TPQPESLSPSGKEPAASSPSSNNTAATTAAIVPGSQLMPSKSPSTGTTEISSHESSHGTP SQTTAKNWELTASASHQPPGVYPQGHSDTTVAISTSTVLLCGLSAVSLLACYLKSRQ TPPLASVEMEAMEALPVTWGTSSRDEDLENCSHHL [0211] SEQ ID NO: 300 (IL-15Rα transmembrane domain) VAISTSTVLLCGLSAVSLLACYL [0212] As used herein, the terms “cleave” or “cleavage” refer to the hydrolysis of phosphodiester bonds within the backbone of a recognition sequence within a target sequence that results in a double-stranded break within the target sequence, referred to herein as a “cleavage site”. [0213] The term “transfer vector,” as used herein, refers to a composition of matter which comprises an isolated nucleic acid and which can be used to deliver the isolated nucleic acid to the interior of a cell. Numerous vectors are known in the art including, but not limited to, linear polynucleotides, polynucleotides associated with ionic or amphiphilic compounds, plasmids, and viruses. Thus, the term “transfer vector” includes an autonomously replicating plasmid or a virus. The term should also be construed to further include non-plasmid and non-viral compounds which facilitate transfer of nucleic acid into cells, such as, for example, a polylysine compound, liposome, and the like. Examples of viral transfer vectors include, but are not limited to, adenoviral vectors, adeno-associated virus vectors, retroviral vectors, lentiviral vectors, and the like. [0214] The term “expression vector,” as used herein, refers to a vector comprising a recombinant polynucleotide comprising expression control sequences operatively linked to a nucleotide sequence to be expressed. An expression vector comprises sufficient cis-acting elements for expression; other elements for expression can be supplied by the host cell or in an in vitro expression system. Expression vectors include all those known in the art, including cosmids, plasmids (e.g., naked or contained in liposomes) and viruses (e.g., lentiviruses, retroviruses, adenoviruses, and adeno-associated viruses) that incorporate the recombinant polynucleotide. [0215] The term “lentivirus,” as used herein, refers to a genus of the Retroviridae family. Lentiviruses are unique among the retroviruses in being able to infect non-dividing cells; they can deliver a significant amount of genetic information into the DNA of the host cell, so they are one of the most efficient methods of a gene delivery vector. HIV, SIV, and FIV are all examples of lentiviruses. [0216] The term “lentiviral vector,” as used herein, refers to a vector derived from at least a portion of a lentivirus genome, including especially a self-inactivating lentiviral vector as
provided in Milone et al., Mol. Ther.17(8): 1453-1464 (2009). Other examples of lentivirus vectors that may be used in the clinic, include but are not limited to, e.g., the LENTIVECTORTM gene delivery technology from Oxford BioMedica, the LENTIMAXTM vector system from Lentigen, and the like. Nonclinical types of lentiviral vectors are also available and would be known to one skilled in the art. [0217] The term “homologous” or “identity,” as used herein, refers to the subunit sequence identity between two polymeric molecules, e.g., between two nucleic acid molecules, such as, two DNA molecules or two RNA molecules, or between two polypeptide molecules. When a subunit position in both of the two molecules is occupied by the same monomeric subunit; e.g., if a position in each of two DNA molecules is occupied by adenine, then they are homologous or identical at that position. The homology between two sequences is a direct function of the number of matching or homologous positions; e.g., if half (e.g., five positions in a polymer ten subunits in length) of the positions in two sequences are homologous, the two sequences are 50% homologous; if 90% of the positions (e.g., 9 of 10), are matched or homologous, the two sequences are 90% homologous. [0218] “Humanized” forms of non-human (e.g., murine) antibodies, as described herein, are chimeric immunoglobulins, immunoglobulin chains or fragments thereof (such as Fv, Fab, Fab’, F(ab’)2 or other antigen-binding subsequences of antibodies) which contain minimal sequence derived from non-human immunoglobulin. For the most part, humanized antibodies and antibody fragments thereof are human immunoglobulins (recipient antibody or antibody fragment) in which residues from a complementary-determining region (CDR) of the recipient are replaced by residues from a CDR of a non-human species (donor antibody) such as mouse, rat or rabbit having the desired specificity, affinity, and capacity. In some instances, Fv framework region (FR) residues of the human immunoglobulin are replaced by corresponding non-human residues. Furthermore, a humanized antibody/antibody fragment can comprise residues which are found neither in the recipient antibody nor in the imported CDR or framework sequences. These modifications can further refine and optimize antibody or antibody fragment performance. In general, the humanized antibody or antibody fragment thereof will comprise substantially all of at least one, and typically two, variable domains, in which all or substantially all of the CDR regions correspond to those of a non-human immunoglobulin and all or a significant portion of the FR regions are those of a human immunoglobulin sequence. The humanized antibody or antibody fragment can also comprise at least a portion of an immunoglobulin constant region (Fc), typically that of a human immunoglobulin. For further details, see Jones et al., Nature, 321: 522-525, 1986; Reichmann et al., Nature, 332: 323-329, 1988; Presta, Curr. Op. Struct. Biol., 2: 593-596, 1992.
[0219] “Human” or “fully human,” as used herein, refers to an immunoglobulin, such as an antibody or antibody fragment, where the whole molecule is of human origin or consists of an amino acid sequence identical to a human form of the antibody or immunoglobulin. [0220] The term “isolated,” as used herein, means altered or removed from the natural state. For example, a nucleic acid or a peptide naturally present in a living animal is not “isolated,” but the same nucleic acid or peptide partially or completely separated from the coexisting materials of its natural state is “isolated.” An isolated nucleic acid or protein can exist in substantially purified form, or can exist in a non-native environment such as, for example, a host cell. [0221] In the context of the present disclosure, the following abbreviations for the commonly occurring nucleic acid bases are used. “A” refers to adenosine, “C” refers to cytosine, “G” refers to guanosine, “T” refers to thymidine, and “U” refers to uridine. [0222] The term “conservative sequence modifications,” as used herein, refers to amino acid modifications that do not significantly affect or alter the binding characteristics of the antibody or antibody fragment containing the amino acid sequence. Such conservative modifications include amino acid substitutions, additions and deletions. Modifications can be introduced into an antibody or antibody fragment of the present disclosure by standard techniques known in the art, such as site-directed mutagenesis and PCR-mediated mutagenesis. Conservative amino acid substitutions are ones in which the amino acid residue is replaced with an amino acid residue having a similar side chain. Families of amino acid residues having similar side chains have been defined in the art. These families include amino acids with basic side chains (e.g., lysine, arginine, histidine), acidic side chains (e.g., aspartic acid, glutamic acid), uncharged polar side chains (e.g., glycine, asparagine, glutamine, serine, threonine, tyrosine, cysteine, tryptophan), nonpolar side chains (e.g., alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine), beta-branched side chains (e.g., threonine, valine, isoleucine) and aromatic side chains (e.g., tyrosine, phenylalanine, tryptophan, histidine). Thus, one or more amino acid residues within a TFP of the present disclosure can be replaced with other amino acid residues from the same side chain family and the altered TFP can be tested using the functional assays described herein. [0223] The term “operably linked” or “transcriptional control,” as used herein, refers to functional linkage between a regulatory sequence and a heterologous nucleic acid sequence resulting in expression of the latter. For example, a first nucleic acid sequence is operably linked with a second nucleic acid sequence when the first nucleic acid sequence is placed in a functional relationship with the second nucleic acid sequence. For instance, a promoter is operably linked to a coding sequence if the promoter affects the transcription or expression of
the coding sequence. Operably linked DNA sequences can be contiguous with each other and, e.g., where necessary to join two protein coding regions, are in the same reading frame. [0224] The terms “nucleotide,” “nucleic acid” and “polynucleotide,” as used herein, are used interchangeably, and refer to deoxyribonucleic acids (DNA) or ribonucleic acids (RNA) and polymers thereof in either single- or double-stranded form. Unless specifically limited, the term encompasses nucleic acids containing known analogues of natural nucleotides that have similar binding properties as the reference nucleic acid and are metabolized in a manner similar to naturally occurring nucleotides. Unless otherwise indicated, a particular nucleic acid sequence also implicitly encompasses conservatively modified variants thereof (e.g., degenerate codon substitutions), alleles, orthologs, SNPs, and complementary sequences as well as the sequence explicitly indicated. Specifically, degenerate codon substitutions may be achieved by generating sequences in which the third position of one or more selected (or all) codons is substituted with mixed-base and/or deoxyinosine residues (Batzer et al., Nucleic Acid Res.19:5081 (1991); Ohtsuka et al., J. Biol. Chem.260:2605-2608 (1985); and Rossolini et al., Mol. Cell. Probes 8:91-98 (1994)). [0225] The terms “peptide,” “polypeptide,” and “protein,” as used herein, are used interchangeably, and refer to a compound comprised of amino acid residues covalently linked by peptide bonds. A protein or peptide must contain at least two amino acids, and no limitation is placed on the maximum number of amino acids that can comprise a protein’s or peptide’s sequence. Polypeptides include any peptide or protein comprising two or more amino acids joined to each other by peptide bonds. As used herein, the term refers to both short chains, which also commonly are referred to in the art as peptides, oligopeptides and oligomers, for example, and to longer chains, which generally are referred to in the art as proteins, of which there are many types. “Polypeptides” include, for example, biologically active fragments, substantially homologous polypeptides, oligopeptides, homodimers, heterodimers, variants of polypeptides, modified polypeptides, derivatives, analogs, fusion proteins, among others. A polypeptide includes a natural peptide, a recombinant peptide, or a combination thereof. [0226] The term “promoter,” as used herein, refers to a DNA sequence recognized by the transcription machinery of the cell, or introduced synthetic machinery, required to initiate the specific transcription of a polynucleotide sequence. [0227] The term “promoter/regulatory sequence,” as used herein, refers to a nucleic acid sequence which is required for expression of a gene product operably linked to the promoter/regulatory sequence. In some instances, this sequence may be the core promoter sequence and in other instances, this sequence may also include an enhancer sequence and
other regulatory elements which are required for expression of the gene product. The promoter/regulatory sequence may, for example, be one which expresses the gene product in a tissue specific manner. [0228] The term “constitutive” promoter, as used herein, refers to a nucleotide sequence which, when operably linked with a polynucleotide which encodes or specifies a gene product, causes the gene product to be produced in a cell under most or all physiological conditions of the cell. [0229] The term “inducible” promoter, as used herein, refers to a nucleotide sequence which, when operably linked with a polynucleotide which encodes or specifies a gene product, causes the gene product to be produced in a cell substantially only when an inducer which corresponds to the promoter is present in the cell. [0230] The term “tissue-specific” promoter, as used herein, refers to a nucleotide sequence which, when operably linked with a polynucleotide encodes or specified by a gene, causes the gene product to be produced in a cell substantially only if the cell is a cell of the tissue type corresponding to the promoter. [0231] The terms “linker” and “flexible polypeptide linker” as used in the context of a scFv refers to a peptide linker that consists of amino acids such as glycine and/or serine residues used alone or in combination, to link variable heavy and variable light chain regions together. In one embodiment, the flexible polypeptide linker is a Gly/Ser linker and comprises the amino acid sequence (Gly-Gly-Gly-Ser)n, where n is a positive integer equal to or greater than 1 (SEQ ID NO: 393). For example, n=1, n=2, n=3, n=4, n=5, n=6, n=7, n=8, n=9 and n=10. In one embodiment, the flexible polypeptide linkers include, but are not limited to, (Gly4Ser)4 (SEQ ID NO: 394) or (Gly4Ser)3 (SEQ ID NO: 395). In another embodiment, the linkers include multiple repeats of (Gly2Ser), (GlySer) or (Gly3Ser) (SEQ ID NO: 396). Also included within the scope of the present disclosure are linkers described in WO2012/138475 (incorporated herein by reference). In some instances, the linker sequence comprises (G4S)n, wherein n=2 to 5 (SEQ ID NO: 397). In some instances, the linker sequence comprises (G4S)n, wherein n=1 to 3 (SEQ ID NO: 398). [0232] As used herein, a 5’ cap (also termed an RNA cap, an RNA 7-methylguanosine cap or an RNA m7G cap) is a modified guanine nucleotide that has been added to the “front” or 5’ end of a eukaryotic messenger RNA shortly after the start of transcription. The 5’ cap consists of a terminal group which is linked to the first transcribed nucleotide. Its presence is critical for recognition by the ribosome and protection from RNases. Cap addition is coupled to transcription, and occurs co-transcriptionally, such that each influences the other. Shortly after the start of transcription, the 5’ end of the mRNA being synthesized is bound by a cap-
synthesizing complex associated with RNA polymerase. This enzymatic complex catalyzes the chemical reactions that are required for mRNA capping. Synthesis proceeds as a multi- step biochemical reaction. The capping moiety can be modified to modulate functionality of mRNA such as its stability or efficiency of translation. [0233] As used herein, “in vitro transcribed RNA” refers to RNA, preferably mRNA, which has been synthesized in vitro. Generally, the in vitro transcribed RNA is generated from an in vitro transcription vector. The in vitro transcription vector comprises a template that is used to generate the in vitro transcribed RNA. [0234] As used herein, a “poly(A)” refers to a series of adenosines attached by polyadenylation to the mRNA. In the preferred embodiment of a construct for transient expression, the polyA is between 50 and 5000, preferably greater than 64, more preferably greater than 100, most preferably greater than 300 or 400 (SEQ ID NO: 399). Poly(A) sequences can be modified chemically or enzymatically to modulate mRNA functionality such as localization, stability or efficiency of translation. [0235] As used herein, “polyadenylation” refers to the covalent linkage of a polyadenylyl moiety, or its modified variant, to a messenger RNA molecule. In eukaryotic organisms, most messenger RNA (mRNA) molecules are polyadenylated at the 3’ end. The 3’ poly(A) tail is a long sequence of adenine nucleotides (often several hundred) added to the pre-mRNA through the action of an enzyme, polyadenylate polymerase. In higher eukaryotes, the poly(A) tail is added onto transcripts that contain a specific sequence, the polyadenylation signal. The poly(A) tail and the protein bound to it aid in protecting mRNA from degradation by exonucleases. Polyadenylation is also important for transcription termination, export of the mRNA from the nucleus, and translation. Polyadenylation occurs in the nucleus immediately after transcription of DNA into RNA, but additionally can also occur later in the cytoplasm. After transcription has been terminated, the mRNA chain is cleaved through the action of an endonuclease complex associated with RNA polymerase. The cleavage site is usually characterized by the presence of the base sequence AAUAAA near the cleavage site. After the mRNA has been cleaved, adenosine residues are added to the free 3’ end at the cleavage site. [0236] As used herein, “transient” refers to expression of a non-integrated transgene for a period of hours, days or weeks, wherein the period of time of expression is less than the period of time for expression of the gene if integrated into the genome or contained within a stable plasmid replicon in the host cell. [0237] The term “signal transduction pathway,” as used herein, refers to the biochemical relationship between a variety of signal transduction molecules that play a role in the
transmission of a signal from one portion of a cell to another portion of a cell. The phrase “cell surface receptor” includes molecules and complexes of molecules capable of receiving a signal and transmitting signal across the membrane of a cell. [0238] The term, a “substantially purified” cell, as used herein, refers to a cell that is essentially free of other cell types. A substantially purified cell also refers to a cell which has been separated from other cell types with which it is normally associated in its naturally occurring state. In some instances, a population of substantially purified cells refers to a homogenous population of cells. In other instances, this term refers simply to cell that have been separated from the cells with which they are naturally associated in their natural state. In some aspects, the cells are cultured in vitro. In other aspects, the cells are not cultured in vitro. [0239] The term “nucleic acid” or “polynucleotide” refers to deoxyribonucleic acids (DNA) or ribonucleic acids (RNA) and polymers thereof in either single- or double-stranded form. Unless specifically limited, the term encompasses nucleic acids containing known analogues of natural nucleotides that have similar binding properties as the reference nucleic acid and are metabolized in a manner similar to naturally occurring nucleotides. Unless otherwise indicated, a particular nucleic acid sequence also implicitly encompasses conservatively modified variants thereof (e.g., degenerate codon substitutions), alleles, orthologs, SNPs, and complementary sequences as well as the sequence explicitly indicated. Specifically, degenerate codon substitutions may be achieved by generating sequences in which the third position of one or more selected (or all) codons is substituted with mixed-base and/or deoxyinosine residues (Batzer et al., Nucleic Acid Res.19:5081 (1991); Ohtsuka et al., J. Biol. Chem.260:2605-2608 (1985); and Rossolini et al., Mol. Cell. Probes 8:91-98 (1994)). [0240] The term “transfected” or “transformed” or “transduced,” as used herein, refers to a process by which exogenous nucleic acid is transferred or introduced into the host cell. A “transfected” or “transformed” or “transduced” cell is one which has been transfected, transformed or transduced with exogenous nucleic acid. The cell includes the primary subject cell and its progeny. [0241] The term “specifically binds,” as used herein, refers to an antibody, an antibody fragment or a specific ligand, which recognizes and binds a cognate binding partner (e.g., CD19) present in a sample, but which does not necessarily and substantially recognize or bind other molecules in the sample. [0242] In the context of the present disclosure, “tumor antigen” or “hyperproliferative disorder antigen” or “antigen associated with a hyperproliferative disorder” refers to antigens that are common to specific hyperproliferative disorders. In certain aspects, the
hyperproliferative disorder antigens of the present disclosure are derived from, cancers including but not limited to primary or metastatic melanoma, thymoma, lymphoma, sarcoma, lung cancer, liver cancer, NHL, leukemias, uterine cancer, cervical cancer, bladder cancer, kidney cancer and adenocarcinomas such as breast cancer, prostate cancer, ovarian cancer, pancreatic cancer, and the like. [0243] The term “anti-tumor effect,” as used herein, refers to a biological effect which can be manifested by various means, including but not limited to, e.g., a decrease in tumor volume, a decrease in the number of tumor cells, a decrease in the number of metastases, an increase in life expectancy, decrease in tumor cell proliferation, decrease in tumor cell survival, or amelioration of various physiological symptoms associated with the cancerous condition. In some embodiments, an “anti-tumor effect” can also be manifested by the ability of the peptides, polynucleotides, cells and antibodies of the present disclosure in prevention of the occurrence of tumor in the first place. [0244] The term “autologous,” as used herein, refers to any material derived from the same individual to whom it is later to be re-introduced into the individual. [0245] The term “allogeneic” or, alternatively, “allogenic,” as used herein, refers to any material derived from a different animal of the same species or different patient as the individual to whom the material is introduced. Two or more individuals are said to be allogeneic to one another when the genes at one or more loci are not identical. In some aspects, allogeneic material from individuals of the same species may be sufficiently unlike genetically to interact antigenically. [0246] The term “xenogeneic,” as used herein, refers to a graft derived from an animal of a different species. [0247] The term “cancer,” as used herein, refers to a disease characterized by the rapid and uncontrolled growth of aberrant cells. Cancer cells can spread locally or through the bloodstream and lymphatic system to other parts of the body. Examples of various cancers are described herein and include but are not limited to, breast cancer, prostate cancer, ovarian cancer, cervical cancer, skin cancer, pancreatic cancer, colorectal cancer, renal cancer, liver cancer, brain cancer, lymphoma, leukemia, lung cancer and the like. [0248] The term “encoding,” as used herein, refers to the inherent property of specific sequences of nucleotides in a polynucleotide, such as a gene, a cDNA, or an mRNA, to serve as templates for synthesis of other polymers and macromolecules in biological processes having either a defined sequence of nucleotides (e.g., rRNA, tRNA and mRNA) or a defined sequence of amino acids and the biological properties resulting therefrom. Thus, a gene, cDNA, or RNA, encodes a protein if transcription and translation of mRNA corresponding to
that gene produces the protein in a cell or other biological system. Both the coding strand, the nucleotide sequence of which is identical to the mRNA sequence and is usually provided in sequence listings, and the non-coding strand, used as the template for transcription of a gene or cDNA, can be referred to as encoding the protein or other product of that gene or cDNA. [0249] Unless otherwise specified, a “nucleotide sequence encoding an amino acid sequence” includes all nucleotide sequences that are degenerate versions of each other and that encode the same amino acid sequence. The phrase nucleotide sequence that encodes a protein or an RNA may also include introns to the extent that the nucleotide sequence encoding the protein may in some versions contain one or more introns. [0250] The terms “effective amount” and “therapeutically effective amount,” as used herein, are used interchangeably herein, and refer to an amount of a compound, formulation, material, or composition, as described herein effective to achieve a particular biological or therapeutic result. [0251] The term “endogenous,” as used herein, refers to any material from or produced inside an organism, cell, tissue or system. [0252] The term “exogenous,” as used herein, refers to any material introduced from or produced outside an organism, cell, tissue or system. [0253] The term “expression,” as used herein, refers to the transcription and/or translation of a particular nucleotide sequence driven by a promoter. [0254] The term “parenteral” administration of an immunogenic composition, as used herein, includes, e.g., subcutaneous (s.c.), intravenous (i.v.), intramuscular (i.m.), or intrasternal injection, intratumoral, or infusion techniques. [0255] The term “therapeutic” as used herein, means a treatment. A therapeutic effect is obtained by reduction, suppression, remission, or eradication of a disease state. [0256] The term “prophylaxis” as used herein, means the prevention of or protective treatment for a disease or disease state. [0257] The term “functional disruption,” as used herein, refers to a physical or biochemical change to a specific (e.g., target) nucleic acid (e.g., gene, RNA transcript, of protein encoded thereby) that prevents its normal expression and/or behavior in the cell. In one embodiment, a functional disruption refers to a modification of the gene via a gene editing method. In one embodiment, a functional disruption prevents expression of a target gene (e.g., an endogenous gene). [0258] As used herein, the term “meganuclease” refers to an endonuclease that binds double- stranded DNA at a recognition sequence that is greater than 12 base pairs. In some embodiments, the recognition sequence for a meganuclease of the present disclosure is 22
base pairs. In some embodiments, a meganuclease may be an endonuclease that is derived from I-Crel and may refer to an engineered variant of I-Crel that has been modified relative to natural I-Crel with respect to, for example, DNA-binding specificity, DNA cleavage activity, DNA-binding affinity, or dimerization properties. Methods for producing such modified variants of I-Crel are known in the art (e.g., WO 2007/047859). In some embodiments, a meganuclease binds to double-stranded DNA as a heterodimer or as a “single-chain meganuclease” in which a pair of DNA-binding domains are joined into a single polypeptide using a peptide linker. The term “homing endonuclease” is synonymous with the term “meganuclease.” In some embodiments, meganucleases are substantially non-toxic when expressed in cells, particularly in human T cells, such that cells may be transfected and maintained at 37°C without observing deleterious effects on cell viability or significant reductions in meganuclease cleavage activity when measured using the methods described herein. [0259] As used herein, the term “single-chain meganuclease” refers to a polypeptide comprising a pair of nuclease subunits joined by a linker. A single-chain meganuclease has the organization: N-terminal subunit - Linker - C-terminal subunit. In some embodiments, the two meganuclease subunits may generally be non-identical in amino acid sequence and may recognize non-identical DNA sequences. Thus, in some embodiments, single-chain meganucleases typically cleave pseudo-palindromic or non-palindromic recognition sequences. In some embodiments, a single-chain meganuclease may be referred to as a “single-chain heterodimer” or “single-chain heterodimeric meganuclease” although it is not, in fact, dimeric. For clarity, unless otherwise specified, the term “meganuclease” can refer to a dimeric or single-chain meganuclease. [0260] As used herein, the term “TALEN” refers to an endonuclease comprising a DNA- binding domain comprising 16-22 TAL domain repeats fused to any portion of the Fokl nuclease domain. [0261] As used herein, the term “Compact TALEN” refers to an endonuclease comprising a DNA-binding domain with 16-22 TAL domain repeats fused in any orientation to any catalytically active portion of nuclease domain of the I-Tevl homing endonuclease. [0262] As used herein, the term “CRISPR” refers to a caspase-based endonuclease comprising a caspase, such as Cas9, and a guide RNA that directs DNA cleavage of the caspase by hybridizing to a recognition site in the genomic DNA. [0263] As used herein, the term “megaTAL” refers to a single-chain nuclease comprising a transcription activator-like effector (TALE) DNA binding domain with an engineered, sequence-specific homing endonuclease.
[0264] As is used herein, the terms “T cell receptor” and “T cell receptor complex” are used interchangeably to refer to a molecule found on the surface of T cells that is, in general, responsible for recognizing antigens. The TCR comprises a heterodimer consisting of a TCR alpha and TCR beta chain in 95% of T cells, whereas 5% of T cells have TCRs consisting of TCR gamma and TCR delta chains. The TCR further comprises one or more of CD3ε, CD3γ, and CD3δ. In some embodiments, the TCR comprises CD3ε. In some embodiments, the TCR comprises CD3γ. In some embodiments, the TCR comprises CD3δ. In some embodiments, the TCR comprises CD3ζ. Engagement of the TCR with antigen, e.g., with antigen and MHC, results in activation of its T cells through a series of biochemical events mediated by associated enzymes, co-receptors, and specialized accessory molecules. In some embodiments, the constant domain of human TCR alpha has a sequence of SEQ ID NO: 142. In some embodiments, the constant domain of human TCR alpha has an IgC domain having a sequence of SEQ ID NO: 143, a transmembrane domain having a sequence of SEQ ID NO: 144, and an intracellular domain having a sequence of SS (SEQ ID NO: 145). In some embodiments, the constant domain of murine TCR alpha has a sequence of SEQ ID NO: 147. In some embodiments, the constant domain of murine TCR alpha has a transmembrane domain having a sequence of SEQ ID NO: 144, and an intracellular domain having a sequence of SS (SEQ ID NO: 145). In some embodiments, the constant domain of human TCR beta has a sequence of SEQ ID NO: 148. In some embodiments, the constant domain of human TCR beta has an IgC domain having a sequence of SEQ ID NO: 149, a transmembrane domain having a sequence of SEQ ID NO: 150, and an intracellular domain having a sequence of SEQ ID NO: 151. In some embodiments, the constant domain of murine TCR beta has a sequence of SEQ ID NO: 152. In some embodiments, the constant domain of murine TCR beta has a transmembrane domain having a sequence of SEQ ID NO: 152, and an intracellular domain having a sequence of SEQ ID NO: 153. In some embodiments, the constant domain of human TCR delta has a sequence of SEQ ID NO: 243. In some embodiments, the constant domain of human TCR delta has an IgC domain having a sequence of SEQ ID NO: 265, a transmembrane domain having a sequence of SEQ ID NO: 158, and an intracellular domain having a sequence of L. In some embodiments, the constant domain of human TCR gamma has a sequence of SEQ ID NO: 21. In some embodiments, the constant domain of human TCR gamma has an IgC domain having a sequence of SEQ ID NO: 155, a transmembrane domain having a sequence of SEQ ID NO: 156, and an intracellular domain having a sequence of SEQ ID NO: 157. [0265] In some embodiments, human CD3 epsilon has a sequence of SEQ ID NO: 258. In some embodiments, human CD3 epsilon has an extracellular domain having a sequence of
SEQ ID NO: 126, a transmembrane domain having a sequence of SEQ ID NO: 127, and an intracellular domain, e.g., an intracellular signaling domain, having a sequence of SEQ ID NO: 128. In some embodiments, human CD3 delta has a sequence of SEQ ID NO: 136. In some embodiments, human CD3 delta has an extracellular domain having a sequence of SEQ ID NO: 138, a transmembrane domain having a sequence of SEQ ID NO: 139, and an intracellular domain, e.g., an intracellular signaling domain, having a sequence of SEQ ID NO: 140. In some embodiments, human CD3 gamma has a sequence of SEQ ID NO: 130. In some embodiments, human CD3 gamma has an extracellular domain having a sequence of SEQ ID NO: 132, a transmembrane domain having a sequence of SEQ ID NO: 133, and an intracellular domain, e.g., an intracellular signaling domain, having a sequence of SEQ ID NO: 134. [0266] Ranges: throughout this disclosure, various aspects of the present disclosure can be presented in a range format. It should be understood that the description in range format is merely for convenience and brevity and should not be construed as an inflexible limitation on the scope of the present disclosure. Accordingly, the description of a range should be considered to have specifically disclosed all the possible subranges as well as individual numerical values within that range. For example, description of a range such as from 1 to 6 should be considered to have specifically disclosed subranges such as from 1 to 3, from 1 to 4, from 1 to 5, from 2 to 4, from 2 to 6, from 3 to 6 etc., as well as individual numbers within that range, for example, 1, 2, 2.7, 3, 4, 5, 5.3, and 6. As another example, a range such as 95- 99% identity, includes something with 95%, 96%, 97%, 98% or 99% identity, and includes subranges such as 96-99%, 96-98%, 96-97%, 97-99%, 97-98% and 98-99% identity. This applies regardless of the breadth of the range. [0267] Provided herein are compositions of matter and methods of use for the treatment of a disease such as cancer, using recombinant nucleic acids comprising: a first nucleic acid sequence encoding a T cell receptor (TCR) fusion protein (TFP), wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain; and wherein the TCR subunit and the antigen binding domain are operatively linked, and a second nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof. As used herein, a “T cell receptor (TCR) fusion protein” or “TFP” includes a recombinant polypeptide derived from the various polypeptides comprising the TCR that is generally capable of i) binding to a surface antigen on target cells and ii) interacting with other polypeptide components of the intact TCR complex, typically when co-located in or on
the surface of a T cell. As provided herein, TFPs provide substantial benefits as compared to Chimeric Antigen Receptors. The term “Chimeric Antigen Receptor” or alternatively a “CAR” refers to a recombinant polypeptide comprising an extracellular antigen binding domain in the form of, e.g., a single domain antibody or scFv, a transmembrane domain, and cytoplasmic signaling domains (also referred to herein as “intracellular signaling domains”) comprising a functional signaling domain derived from a stimulatory molecule as defined below. Generally, the central intracellular signaling domain of a CAR is derived from the CD3 zeta chain that is normally found associated with the TCR complex. The CD3 zeta signaling domain can be fused with one or more functional signaling domains derived from at least one co-stimulatory molecule such as 4-1BB (i.e., CD137), CD27 and/or CD28. Dominant Negative TGFBR2 polypeptides [0268] Provided herein are recombinant nucleic acids comprising: a first nucleic acid sequence encoding a T cell receptor (TCR) fusion protein (TFP) wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain, wherein the TCR subunit and the antigen binding domain are operatively linked; and a second nucleic acid sequence encoding a dominant negative form of transforming growth factor beta receptor II (TGFBr2). In some embodiments, the first nucleic acid sequence and the second nucleic acid sequence are operatively linked by a linker. In some embodiments, the linker comprises a protease cleavage site. In some embodiments, the protease cleavage site is a 2A cleavage site. In some embodiments, the cleavage site can be a self-cleaving peptide such as a T2A, P2A, E2A or F2A cleavage site. In some embodiments, the 2A cleavage site is a T2A cleavage site or a P2A cleavage site. [0269] In some embodiments, the dominant negative TGFBr2 comprises a TGFBr2 extracellular domain and does not comprise a functional kinase domain. In some embodiments, the dominant negative TGFBr2 comprises a truncated TGFBr2 intracellular domain. In some embodiments, the dominant negative TGFBr2 comprises or consists of a TGFBr2 extracellular domain and a TGFBR2 transmembrane domain. In some embodiments, the dominant negative TGFBr2 comprises a truncated or non-functional kinase domain. In some embodiments, the dominant negative TGFBr2 comprises or consists of about 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 50, 75, 100, 150, or 200 amino acids of a TGFBr2 intracellular domain. For example, in some embodiments, the dominant negative TGFBr2 comprises no more than about 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28,
39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids of SEQ ID NO: 294. In some embodiments, the intracellular domain portion of the dominant negative TGFBr2 consists of SEQ ID NO: 389. [0270] In some embodiments, the dominant negative TGFBr2 comprises a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to SEQ ID NO: 68. In some embodiments, the dominant negative TGFBr2 comprises the sequence of SEQ ID NO: 68. In some embodiments, the sequence of the dominant negative TGFBr2 extracellular domain is the sequence of SEQ ID NO:68. [0271] In some embodiments, the dominant negative TGFBr2 polypeptide comprises an addition of amino acid residue(s) to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of a sequence of a dominant negative TGFBr2 polypeptide as described herein. For example, in some embodiments, the dominant negative TGFBr2 polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C- terminal ends of a sequence of a dominant negative TGFBr2 polypeptide as described herein. In some embodiments, the dominant negative TGFBr2 polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N- terminal end, C-terminal end, or both N-terminal and C-terminal ends of a sequence of a dominant negative TGFBr2 polypeptide as described herein. In some embodiments, the dominant negative TGFBr2 polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C- terminal end, or both N-terminal and C-terminal ends, of SEQ ID NO: 68. In some embodiments, the dominant negative TGFBr2 polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55,
60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N- terminal end, C-terminal end, or both N-terminal and C-terminal ends of SEQ ID NO: 68. [0272] In some embodiments, the dominant negative TGFBr2 polypeptide comprises amino acid residue deletions from the N-terminal end, C-terminal end, or both N-terminal and C- terminal ends of a sequence of a dominant negative TGFBr2 polypeptide as described herein. For example, in some embodiments, the dominant negative TGFBr2 polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids deleted from the N-terminal or C-terminal end of a sequence of a dominant negative TGFBr2 polypeptide as described herein. In some embodiments, the dominant negative TGFBr2 polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids deleted from the N-terminal or C-terminal end of a sequence of a dominant negative TGFBr2 polypeptide as described herein. In some embodiments, the dominant negative TGFBr2 polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids deleted from the N-terminal or C-terminal end of SEQ ID NO: 68. In some embodiments, the dominant negative TGFBr2 polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids deleted from the N-terminal or C-terminal end of SEQ ID NO: 68. In some embodiments, the dominant negative TGFBr2 polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently deleted from both N-terminal and C-terminal ends of a sequence
of a dominant negative TGFBr2 polypeptide as described herein. In some embodiments, the dominant negative TGFBr2 polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently deleted from both N-terminal and C- terminal ends of a sequence of a dominant negative TGFBr2 polypeptide as described herein. In some embodiments, the dominant negative TGFBr2 polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently deleted from both N-terminal and C-terminal ends of SEQ ID NO: 68. In some embodiments, the dominant negative TGFBr2 polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently deleted from both N-terminal and C- terminal ends of SEQ ID NO: 68. [0273] In some embodiments, the dominant negative TGFBr2 polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently mutated as compared to SEQ ID NO: 68. In some embodiments, the dominant negative TGFBr2 polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids mutated as compared to SEQ ID NO: 68. Switch Polypeptides [0274] Provided herein are recombinant nucleic acids comprising: a first nucleic acid sequence encoding a T cell receptor (TCR) fusion protein (TFP) wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain; and wherein the TCR
subunit and the antigen binding domain are operatively linked, and a second nucleic acid sequence encoding a switch polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof. In some embodiments, the first nucleic acid sequence and the second nucleic acid sequence are operatively linked by a linker. In some embodiments, the linker comprises a protease cleavage site. In some embodiments, the protease cleavage site is a 2A cleavage site. In some embodiments, the cleavage site can be a self-cleaving peptide such as a T2A, P2A, E2A or F2A cleavage site. In some embodiments, the 2A cleavage site is a T2A cleavage site or a P2A cleavage site. [0275] In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286. In some embodiments, the switch polypeptide comprises any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286. In some embodiments, the sequence of the switch polypeptide is a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, or 99.9% sequence identity to any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286. In some embodiments, the sequence of the switch polypeptide is any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286. [0276] In some embodiments, the switch polypeptide comprises an addition of amino acid residue(s) to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of a sequence of a switch polypeptide as described herein. For example, in some embodiments, the switch polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of a sequence of a switch polypeptide as described herein. In some embodiments, the switch polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of a sequence of a switch polypeptide as described herein. In some embodiments, the switch polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19,
20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286. In some embodiments, the switch polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286. [0277] In some embodiments, the switch polypeptide comprises amino acid residue deletions from the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of a sequence of a switch polypeptide as described herein. For example, in some embodiments, the switch polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids deleted from the N-terminal or C-terminal end of a sequence of a switch polypeptide as described herein. In some embodiments, the switch polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids deleted from the N-terminal or C-terminal end of a sequence of a switch polypeptide as described herein. In some embodiments, the switch polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids deleted from the N-terminal or C-terminal end of any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286. In some embodiments, the switch polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75,
80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids deleted from the N-terminal or C-terminal end of any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286. In some embodiments, the switch polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently deleted from both N-terminal and C-terminal ends of a sequence of a switch polypeptide as described herein. In some embodiments, the switch polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently deleted from both N-terminal and C-terminal ends of a sequence of a switch polypeptide as described herein. In some embodiments, the switch polypeptide comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently deleted from both N-terminal and C-terminal ends of any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286. In some embodiments, the switch polypeptide comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, or 200 amino acids independently deleted from both N-terminal and C-terminal ends of any one sequence selected from SEQ ID NOs: 283, 284, 285, and 286. TGFBr2 extracellular domain [0278] In some embodiments, the TGFBr2 extracellular domain comprises a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to SEQ ID NO:271. In some embodiments, the TGFBr2 extracellular domain comprises the sequence of SEQ ID NO:271. In some embodiments, the sequence of the TGFBr2 extracellular domain is a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, or 99.9% sequence identity to SEQ ID NO:271.
In some embodiments, the sequence of the TGFBr2 extracellular domain is the sequence of SEQ ID NO:271. [0279] In some embodiments, the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having an addition of amino acid residue(s) to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of the TGFBr2 extracellular domain as described herein. For example, in some embodiments, the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of the TGFBr2 extracellular domain as described herein. In some embodiments, the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C- terminal ends of the sequence of the TGFBr2 extracellular domain as described herein. In some embodiments, the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:271. In some embodiments, the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:271.
[0280] In some embodiments, the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having a deletion of amino acid residue(s) from the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of the TGFBr2 extracellular domain as described herein. For example, in some embodiments, the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, or 130 amino acids deleted from the N-terminal or C-terminal end of the sequence of the TGFBr2 extracellular domain as described herein. In some embodiments, the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, or 130 amino acids deleted from the N-terminal or C-terminal end of the sequence of the TGFBr2 extracellular domain as described herein. In some embodiments, the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, or 130 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:271. In some embodiments, the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, or 130 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:271. In some embodiments, in some embodiments, the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, or 130 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of the TGFBr2 extracellular domain as described herein. In some embodiments, the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22,
23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, or 130 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of the TGFBr2 extracellular domain as described herein. In some embodiments, the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, or 130 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:271. In some embodiments, the switch polypeptide and/or dominant negative polypeptide comprises an extracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, or 130 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:271. [0281] TGFBr2 extracellular domain (TGFBR2 ecto): TIPPHVQKSVNNDMIVTDNNGAVKFPQLCKFCDVRFSTCDNQKSCMSNCSITSICEKP QEVCVAVWRKNDENITLETVCHDPKLPYHDFILEDAASPKCIMKEKKKPGETFFMCS CSSDECNDNIIFSEEYNTSNPDLLLVIFQ (SEQ ID NO:271) Switch Intracellular Domain [0282] In some embodiments, the switch polypeptide further comprises a switch intracellular domain. In some embodiments, the TGFBr2 extracellular domain is operably linked to the switch intracellular domain. In some embodiments, the switch intracellular domain comprises an intracellular domain of a costimulatory polypeptide. In some embodiments, the costimulatory polypeptide is selected from the group consisting of CD28, 4-1BB(CD137), IL-15Ra, IL12R, IL18R, IL21R, OX40, CD2, CD27, CD5, ICAM-1, ICOS (CD278), GITR, CD30, CD40, BAFFR, HVEM, CD7, LIGHT, NKG2C, SLAMF7, NKp80, CD160, CD226, FcγRI, FcγRII, and FcγRIII. In some embodiments, the costimulatory polypeptide is CD28. In some embodiments, the costimulatory polypeptide is 4-1BB. In some embodiments, the costimulatory polypeptide is IL-15Ra. [0283] In some embodiments, the switch intracellular domain comprises a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to or SEQ ID NO:273 or SEQ ID NO:277. In some embodiments, the switch intracellular domain comprises the sequence of SEQ ID NO:273 or SEQ ID NO:277. In some embodiments, the sequence of the switch intracellular domain is a sequence
with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:273 or SEQ ID NO:277. In some embodiments, the sequence of the switch intracellular domain is the sequence of SEQ ID NO:273 or SEQ ID NO:277. [0284] In some embodiments, the switch polypeptide comprises an intracellular domain sequence having an addition of amino acid residue(s) to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the intracellular domain sequence as described herein. For example, in some embodiments, the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the intracellular domain sequence as described herein. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the intracellular domain sequence as described herein. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:273 or SEQ ID NO:277. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:273 or SEQ ID NO:277.
[0285] In some embodiments, the switch polypeptide comprises an intracellular domain sequence having a deletion of amino acid residue(s) from the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the intracellular domain sequence as described herein. For example, in some embodiments, the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids deleted from the N-terminal or C-terminal end of the intracellular domain sequence as described herein. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids deleted from the N-terminal or C-terminal end of the intracellular domain sequence as described herein. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids deleted from the N-terminal or C- terminal end of the sequence of SEQ ID NO:273 or SEQ ID NO:277. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:273 or SEQ ID NO:277. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids independently deleted from both N-terminal and C-terminal ends of the intracellular domain sequence as described herein. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids independently deleted from both N-terminal and C-terminal ends of the intracellular domain sequence as described herein. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:273 or SEQ ID NO:277. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:273 or SEQ ID NO:277.
[0286] In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a 4-IBB intracellular domain or a fragment thereof, a MyD88 intracellular domain or a fragment thereof, an ICOS intracellular domain or a fragment thereof, a CTLA4 intracellular domain or a fragment thereof, a CD28 intracellular domain or a fragment thereof, a CD200R intracellular domain or a fragment thereof, a BTLA intracellular domain or a fragment thereof, a TIM-3 intracellular domain or a fragment thereof, a TIGIT intracellular domain or a fragment thereof, a 4-1BB intracellular domain or a fragment thereof, an IL-10RA intracellular domain or a fragment thereof, an IL-7RA intracellular domain or a fragment thereof, a Fas intracellular domain or a fragment thereof, a TRAIL-R2 intracellular domain or a fragment thereof, a PD-1 intracellular domain or a fragment thereof, an IL-4RA intracellular domain or a fragment thereof, an IL12R intracellular domain or a fragment thereof, an IL18R intracellular domain or a fragment thereof, an IL21R intracellular domain or a fragment thereof, or a combination thereof. [0287] In some embodiments, an IL12R intracellular domain or a fragment thereof is an IL- 12 receptor subunit beta-1 intracellular domain or a fragment thereof. In some embodiments, an IL12R intracellular domain or a fragment thereof is an IL-12 receptor subunit beta-2 intracellular domain or a fragment thereof. In some embodiments, an IL18R intracellular domain or a fragment thereof is an interleukin-18 receptor 1 intracellular domain or a fragment thereof. [0288] In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human 4-IBB intracellular domain or a fragment thereof, a human MyD88 intracellular domain or a fragment thereof, a human ICOS intracellular domain or a fragment thereof, a human CTLA4 intracellular domain or a fragment thereof, a human CD28 intracellular domain or a fragment thereof, a human CD200R intracellular domain or a fragment thereof, a human BTLA intracellular domain or a fragment thereof, a human TIM-3 intracellular domain or a fragment thereof, a human TIGIT intracellular domain or a fragment thereof, a human 4-1BB intracellular domain or a fragment thereof, a human IL-10RA intracellular domain or a fragment thereof, a human IL-7RA intracellular domain or a fragment thereof, a Fas intracellular domain or a fragment thereof, a TRAIL-R2 intracellular domain or a fragment thereof, a human PD-1 intracellular domain or a fragment thereof, a human IL-4RA intracellular domain or a fragment thereof, a human IL12R intracellular domain or a fragment thereof, a human IL18R intracellular domain or a fragment thereof, a human IL21R intracellular domain or a fragment thereof, or a combination thereof.
[0289] In some embodiments, a human IL12R intracellular domain or a fragment thereof is a human IL-12 receptor subunit beta-1 intracellular domain or a fragment thereof. In some embodiments, a human IL12R intracellular domain or a fragment thereof is a human IL-12 receptor subunit beta-2 intracellular domain or a fragment thereof. In some embodiments, a human IL18R intracellular domain or a fragment thereof is a human interleukin-18 receptor 1 intracellular domain or a fragment thereof. [0290] In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a CD28 intracellular domain or a fragment thereof and a CTLA4 intracellular domain or a fragment thereof. In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human CD28 intracellular domain or a fragment thereof and a human CTLA4 intracellular domain or a fragment thereof. [0291] In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a Myd88 intracellular domain or a fragment thereof and a TGFβR2 intracellular domain or a fragment thereof. In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human Myd88 intracellular domain or a fragment thereof and a human TGFβR2 intracellular domain or a fragment thereof. [0292] In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a CD28 intracellular domain or a fragment thereof and a Fas intracellular domain or a fragment thereof. In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human CD28 intracellular domain or a fragment thereof and a human Fas intracellular domain or a fragment thereof. [0293] In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a 4-1BB intracellular domain or a fragment thereof and a Fas intracellular domain or a fragment thereof. In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human 4-1BB intracellular domain or a fragment thereof and a human Fas intracellular domain or a fragment thereof. [0294] In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a MyD88 intracellular domain or a fragment thereof and a Fas intracellular domain or a fragment thereof. In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human MyD88
intracellular domain or a fragment thereof and a human Fas intracellular domain or a fragment thereof. [0295] In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to an ICOS intracellular domain or a fragment thereof and a Fas intracellular domain or a fragment thereof. In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human ICOS intracellular domain or a fragment thereof and a human Fas intracellular domain or a fragment thereof. [0296] In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a CD28 intracellular domain or a fragment thereof and a TRAIL-R2 intracellular domain or a fragment thereof. In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human CD28 intracellular domain or a fragment thereof and a human TRAIL-R2 intracellular domain or a fragment thereof. [0297] In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a MyD88 intracellular domain or a fragment thereof and a PD-1 intracellular domain or a fragment thereof. In some embodiments, the switch polypeptide comprises a TGFBr2 extracellular domain or portion thereof linked to a human MyD88 intracellular domain or a fragment thereof and a human PD-1 intracellular domain or a fragment thereof. [0298] In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to any one selected from SEQ ID NOs:287, 277, 288, 289, 273, 290, 291, 292, 293, 294, 295, 296, 297, 298, 299, 313, 314, 315, 316, and a combination thereof. In some embodiments, the switch polypeptide comprises any one sequence selected from the group consisting of SEQ ID NOs:287, 277, 288, 289, 273, 290, 291, 292, 293, 294, 295, 296, 297, 298, 299, 313, 314, 315, 316, and a combination thereof. In some embodiments, the sequence of the switch intracellular domain is a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to any one selected from SEQ ID NOs:287, 277, 288, 289, 273, 290, 291, 292, 293, 294, 295, 296, 297, 298, 299, 313, 314, 315, 316, and a combination thereof. In some embodiments, the sequence of the switch intracellular domain is any one sequence selected from the group consisting of SEQ ID NOs:287, 277, 288, 289, 273, 290, 291, 292, 293, 294, 295, 296, 297, 298, 299, 313, 314, 315, 316, and a combination thereof.
[0299] PD-1 Intracellular domain CSRAARGTIGARRTGQPLKEDPSAVPVFSVDYGELDFQWREKTPEPPVPCVPEQTEY ATIVFPSGMGTSSPARRGSADGPRSAQPLRPEDGHCSWPL (SEQ ID NO:287) [0300] 4-1BB Intracellular domain KRGRKKLLYIFKQPFMRPVQTTQEEDGCSCRFPEEEEGGCEL (SEQ ID NO:277) [0301] ICOS Intracellular domain CWLTKKKYSSSVHDPNGEYMFMRAVNTAKKSRLTDVTL (SEQ ID NO:288) [0302] CTLA4 Intracellular domain AVSLSKMLKKRSPLTTGVYVKMPPTEPECEKQFQPYFIPIN (SEQ ID NO:289) [0303] CD28 Intracellular domain RSKRSRLLHSDYMNMTPRRPGPTRKHYQPYAPPRDFAAYRS (SEQ ID NO:273) [0304] CD200R Intracellular domain KVNGCRKYKLNKTESTPVVEEDEMQPYASYTEKNNPLYDTTNKVKASEALQSEVDT DLHTL (SEQ ID NO:290) [0305] BTLA Intracellular domain RRHQGKQNELSDTAGREINLVDAHLKSEQTEASTRQNSQVLLSETGIYDNDPDLCFR MQEGSEVYSNPCLEENKPGIVYASLNHSVIGPNSRLARNVKEAPTEYASICVRS (SEQ ID NO:291) [0306] TIM-3 Intracellular domain FKWYSHSKEKIQNLSLISLANLPPSGLANAVAEGIRSEENIYTIEENVYEVEEPNEYYC YVSSRQQPSQPLGCRFAMP (SEQ ID NO:292) [0307] TIGIT Intracellular domain LTRKKKALRIHSVEGDLRRKSAGQEEWSPSAPSPPGSCVQAEAAPAGLCGEQRGEDC AELHDYFNVLSYRSLGNCSFFTETG (SEQ ID NO:293) [0308] TGFβR2 Intracellular domain CYRVNRQQKLSSTWETGKTRKLMEFSEHCAIILEDDRSDISSTCANNINHNTELLPIEL DTLVGKGRFAEVYKAKLKQNTSEQFETVAVKIFPYEEYASWKTEKDIFSDINLKHENI LQFLTAEERKTELGKQYWLITAFHAKGNLQEYLTRHVISWEDLRKLGSSLARGIAHL HSDHTPCGRPKMPIVHRDLKSSNILVKNDLTCCLCDFGLSLRLDPTLSVDDLANSGQ VGTARYMAPEVLESRMNLENVESFKQTDVYSMALVLWEMTSRCNAVGEVKDYEPP FGSKVREHPCVESMKDNVLRDRGRPEIPSFWLNHQGIQMVCETLTECWDHDPEARL TAQCVAERFSELEHLDRLSGRSCSEEKIPEDGSLNTTK (SEQ ID NO:294) [0309] IL-10RA Intracellular domain QLYVRRRKKLPSVLLFKKPSPFIFISQRPSPETQDTIHPLDEEAFLKVSPELKNLDLHGS TDSGFGSTKPSLQTEEPQFLLPDPHPQADRTLGNREPPVLGDSCSSGSSNSTDSGICLQ
EPSLSPSTGPTWEQQVGSNSRGQDDSGIDLVQNSEGRAGDTQGGSALGHHSPPEPEV PGEEDPAAVAFQGYLRQTRCAEEKATKTGCLEEESPLTDGLGPKFGRCLVDEAGLHP PALAKGYLKQDPLEMTLASSGAPTGQWNQPTEEWSLLALSSCSDLGISDWSFAHDL APLGCVAAPGGLLGSFNSDLVTLPLISSLQSSE (SEQ ID NO:295) [0310] IL-4RA Intracellular domain KIKKEWWDQIPNPARSRLVAIIIQDAQGSQWEKRSRGQEPAKCPHWKNCLTKLLPCF LEHNMKRDEDPHKAAKEMPFQGSGKSAWCPVEISKTVLWPESISVVRCVELFEAPV ECEEEEEVEEEKGSFCASPESSRDDFQEGREGIVARLTESLFLDLLGEENGGFCQQDM GESCLLPPSGSTSAHMPWDEFPSAGPKEAPPWGKEQPLHLEPSPPASPTQSPDNLTCT ETPLVIAGNPAYRSFSNSLSQSPCPRELGPDPLLARHLEEVEPEMPCVPQLSEPTTVPQ PEPETWEQILRRNVLQHGAAAAPVSAPTSGYQEFVHAVEQGGTQASAVVGLGPPGE AGYKAFSSLLASSAVSPEKCGFGASSGEEGYKPFQDLIPGCPGDPAPVPVPLFTFGLD REPPRSPQSSHLPSSSPEHLGLEPGEKVEDMPKPPLPQEQATDPLVDSLGSGIVYSALT CHLCGHLKQCHGQEDGGQTPVMASPCCGCCCGDRSSPPTTPLRAPDPSPGGVPLEAS LCPASLAPSGISEKSKSSSSFHPAPGNAQSSSQTPKIVNFVSVGPTYMRVS (SEQ ID NO:296) [0311] IL-7RA Intracellular domain KKRIKPIVWPSLPDHKKTLEHLCKKPRKNLNVSFNPESFLDCQIHRVDDIQARDEVEG FLQDTFPQQLEESEKQRLGGDVQSPNCPSEDVVITPESFGRDSSLTCLAGNVSACDAP ILSSSRSLDCRESGKNGPHVYQDLLLSLGTTNSTLPPPFSLQSGILTLNPVAQGQPILTS LGSNQEEAYVTMSSFYQNQ (SEQ ID NO:297) [0312] Fas Intracellular domain KRKEVQKTCRKHRKENQGSHESPTLNPETVAINLSDVDLSKYITTIAGVMTLSQVKG FVRKNGVNEAKIDEIKNDNVQDTAEQKVQLLRNWHQLHGKKEAYDTLIKDLKKAN LCTLAEKIQTIILKDITSDSENSNFRNEIQSLV (SEQ ID NO:298) [0313] TRAILR2 Intracellular domain CKSLLWKKVLPYLKGICSGGGGDPERVDRSSQRPGAEDNVLNEIVSILQPTQVPEQE MEVQEPAEPTGVNMLSPGESEHLLEPAEAERSQRRRLLVPANEGDPTETLRQCFDDF ADLVPFDSWEPLMRKLGLMDNEIKVAKAEAAGHRDTLYTMLIKWVNKTGRDASVH TLLDALETLGERLAKQKIEDHLLSSGKFMYLEGNADSAMS (SEQ ID NO:299) [0314] IL12R (IL-12 receptor) subunit beta-1 intracellular domain NRAARHLCPPLPTPCASSAIEFPGGKETWQWINPVDFQEEASLQEALVVEMSWDKGE RTEPLEKTELPEGAPELALDTELSLEDGDRCKAKM (SEQ ID NO:313) [0315] IL12R (IL-12 receptor) subunit beta-2 intracellular domain HYFQQKVFVLLAALRPQWCSREIPDPANSTCAKKYPIAEEKTQLPLDRLLIDWPTPED
PEPLVISEVLHQVTPVFRHPPCSNWPQREKGIQGHQASEKDMMHSASSPPPPRALQA ESRQLVDLYKVLESRGSDPKPENPACPWTVLPAGDLPTHDGYLPSNIDDLPSHEAPL ADSLEELEPQHISLSVFPSSSLHPLTFSCGDKLTLDQLKMRCDSLML (SEQ ID NO:314) [0316] IL18R1 (Interleukin-18 receptor 1) intracellular domain YRVDLVLFYRHLTRRDETLTDGKTYDAFVSYLKECRPENGEEHTFAVEILPRVLEKH FGYKLCIFERDVVPGGAVVDEIHSLIEKSRRLIIVLSKSYMSNEVRYELESGLHEALVE RKIKIILIEFTPVTDFTFLPQSLKLLKSHRVLKWKADKSLSYNSRFWKNLLYLMPAKT VKPGRDEPEVLPVLSES (SEQ ID NO:315) [0317] IL21R intracellular domain SLKTHPLWRLWKKIWAVPSPERFFMPLYKGCSGDFKKWVGAPFTGSSLELGPWSPE VPSTLEVYSCHPPRSPAKRLQLTELQEPAELVESDGVPKPSFWPTAQNSGGSAYSEER DRPYGLVSIDTVTVLDAEGPCTWPCSCEDDGYPALDLDAGLEPSPGLEDPLLDAGTT VLSCGCVSAGSPGLGGPLGSLLDRLKPPLADGEDWAGGLPWGGRSPGGVSESEAGS PLAGLDMDTFDSGFVGSDCSSPVECDFTSPGDEGPPRSYLRQWVVIPPPLSSPGPQAS (SEQ ID NO:316) [0318] In some embodiments, the costimulatory polypeptide is selected from the group consisting of OX40, CD2, CD27, CD5, IL12R, IL18R, IL21R, ICAM-1, ICOS (CD278), 4- 1BB (CD137), GITR, CD28, CD30, CD40, BAFFR, HVEM, CD7, LIGHT, NKG2C, SLAMF7, NKp80, CD160, CD226, FcγRI, FcγRII, FcγRIII, CD3 zeta, CD28, CD27, ICOS, DAP10, DAP12, LFA-1 (CD11a/CD18) an MHC class 1 molecule, BTLA and a Toll ligand receptor, lymphocyte function-associated antigen-1 (LFA-1, also known as CD11a/CD18), CD276 (B7-H3), IL-15Ra, and a ligand that specifically binds with CD83. [0319] In some embodiments, the costimulatory polypeptide is IL-15Ra. In some embodiments, the sequence of the switch intracellular domain is from the intracellular domain of IL-15Ra. For example, in some embodiments, the sequence of the switch intracellular domain comprises a sequence having at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to SEQ ID NO:372. For another example, in some embodiments, the sequence of the switch intracellular domain comprises a sequence or portion thereof of SEQ ID NO:372. In some embodiments, the sequence of the switch intracellular domain comprises a sequence having at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to SEQ ID NO:383. In some embodiments, the sequence of the switch intracellular domain comprises a sequence or portion thereof of SEQ ID NO:383. [0320] In some embodiments, the switch polypeptide comprises an intracellular domain sequence having an addition of amino acid residue(s) to the N-terminal end, C-terminal end,
or both N-terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein. For example, in some embodiments, the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C- terminal ends of the sequence of an IL-15Ra intracellular domain as described herein. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N- terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:372 or SEQ ID NO:383. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N- terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:372 or SEQ ID NO:383. [0321] In some embodiments, the switch polypeptide comprises an intracellular domain sequence having a deletion of amino acid residue(s) from the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein. For example, in some embodiments, the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115,
120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids deleted from the N-terminal or C-terminal end of the sequence of an IL-15Ra intracellular domain as described herein. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids deleted from the N-terminal or C-terminal end of the sequence of an IL-15Ra intracellular domain as described herein. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of an IL- 15Ra intracellular domain as described herein. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:372. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:372. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:372. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:372. In some embodiments, the switch polypeptide comprises an intracellular domain
sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:383. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids deleted from the N-terminal or C- terminal end of the sequence of SEQ ID NO:383. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:383. In some embodiments, the switch polypeptide comprises an intracellular domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids independently deleted from both N-terminal and C- terminal ends of the sequence of SEQ ID NO:383. Transmembrane Domain [0322] In some embodiments, the switch polypeptide and/or dominant negative polypeptide further comprises a transmembrane domain. In some embodiments, the TGFBr2 extracellular domain of the switch polypeptide is operably linked to the switch intracellular domain via a switch transmembrane domain. [0323] In some embodiments, the transmembrane domain is derived from a TGFBr2 transmembrane domain. In some embodiments, the transmembrane domain comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:272. In some embodiments, the transmembrane domain comprises the sequence of SEQ ID NO:272. In some embodiments, the sequence of the transmembrane domain is a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:272. In some embodiments, the sequence of the transmembrane domain is the sequence of SEQ ID NO:272.
[0324] In some embodiments, the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having an addition of amino acid residue(s) to the N- terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of a TGFBr2 transmembrane domain as described herein. For example, in some embodiments, the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of a TGFBr2 transmembrane domain as described herein. In some embodiments, the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of a TGFBr2 transmembrane domain as described herein. In some embodiments, the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:272. In some embodiments, the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C- terminal ends of the sequence of SEQ ID NO:272. [0325] In some embodiments, the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having a deletion of amino acid residue(s) from the N- terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of a TGFBr2 transmembrane domain as described herein. For example, in some embodiments, the
switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, or 18 amino acids deleted from the N-terminal or C-terminal end of the sequence of a TGFBr2 transmembrane domain as described herein. In some embodiments, the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, or 18 amino acids deleted from the N-terminal or C-terminal end of the sequence of a TGFBr2 transmembrane domain as described herein. In some embodiments, the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, or 18 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:272. In some embodiments, the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, or 18 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:272. In some embodiments, the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, or 18 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of a TGFBr2 transmembrane domain as described herein. In some embodiments, the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, or 18 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of a TGFBr2 transmembrane domain as described herein. In some embodiments, the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, or 18 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:272. In some embodiments, the switch and/or dominant negative polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, or 18 amino acids independently deleted from both N- terminal and C-terminal ends of the sequence of SEQ ID NO:272. [0326] In some embodiments, the switch transmembrane domain is derived from a transmembrane domain of the costimulatory polypeptide. In some embodiments, the switch transmembrane domain is derived from a transmembrane domain of CD28. In some embodiments, the switch transmembrane domain is derived from a transmembrane domain of 4-1BB. In some embodiments, the switch transmembrane domain comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence to SEQ ID NO:275 or SEQ ID NO:279. In some embodiments,
the switch transmembrane domain comprises the sequence of SEQ ID NO:275 or SEQ ID NO:279. In some embodiments, the sequence of the switch transmembrane domain is a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence to SEQ ID NO:275 or SEQ ID NO:279. In some embodiments, the sequence of the switch transmembrane domain is the sequence of SEQ ID NO:275 or SEQ ID NO:279. [0327] In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having an addition of amino acid residue(s) to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of a CD28 transmembrane domain or a 4-1BB transmembrane domain as described herein. For example, in some embodiments, the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of a CD28 transmembrane domain or a 4-1BB transmembrane domain as described herein. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of a CD28 transmembrane domain or a 4-1BB transmembrane domain as described herein. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:275 or SEQ ID NO:279. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to
the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:275 or SEQ ID NO:279. [0328] In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having a deletion of amino acid residue(s) from the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of a CD28 transmembrane domain or a 4-1BB transmembrane domain as described herein. For example, in some embodiments, the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 amino acids deleted from the N-terminal or C-terminal end of the sequence of a CD28 transmembrane domain or a 4-1BB transmembrane domain as described herein. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 amino acids deleted from the N-terminal or C-terminal end of the sequence of a CD28 transmembrane domain or a 4- 1BB transmembrane domain as described herein. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:275 or SEQ ID NO:279. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:275 or SEQ ID NO:279. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 amino acids independently deleted from both N- terminal and C-terminal ends of the sequence of a CD28 transmembrane domain or a 4-1BB transmembrane domain as described herein. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of a CD28 transmembrane domain or a 4- 1BB transmembrane domain as described herein. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, or 24 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:275 or SEQ ID NO:279. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15,
16, 17, 18, 19, 20, 21, 22, 23, or 24 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:275 or SEQ ID NO:279. [0329] In some embodiments, the switch transmembrane domain is derived from a transmembrane domain of IL-15Ra. In some embodiments, the switch transmembrane domain comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence to SEQ ID NO:300. In some embodiments, the switch transmembrane domain comprises the sequence of SEQ ID NO:300. In some embodiments, the sequence of the switch transmembrane domain is a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence to SEQ ID NO:300. In some embodiments, the sequence of the switch transmembrane domain is the sequence of SEQ ID NO:300. [0330] In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having an addition of amino acid residue(s) to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra transmembrane domain as described herein. For example, in some embodiments, the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra transmembrane domain as described herein. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra transmembrane domain as described herein. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C- terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:300. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence
having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N- terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:300. [0331] In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having a deletion of amino acid residue(s) from the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra transmembrane domain as described herein. For example, in some embodiments, the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 amino acids deleted from the N-terminal or C-terminal end of the sequence of an IL-15Ra transmembrane domain as described herein. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 amino acids deleted from the N-terminal or C-terminal end of the sequence of an IL-15Ra transmembrane domain as described herein. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:300. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:300. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of an IL-15Ra transmembrane domain as described herein. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 amino acids independently deleted from both N- terminal and C-terminal ends of the sequence of an IL-15Ra transmembrane domain as described herein. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:300. In some embodiments, the switch polypeptide comprises a transmembrane domain sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15,
16, 17, 18, or 19 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:300. [0332] In some embodiments, the switch transmembrane domain is derived from any one selected from the group consisting of an ICOS transmembrane domain or a fragment thereof, a PD-1 transmembrane domain or a fragment thereof, a CTLA4 transmembrane domain or a fragment thereof, a CD200R transmembrane domain or a fragment thereof, a BTLA transmembrane domain or a fragment thereof, a TIM-3 transmembrane domain or a fragment thereof, a TIGIT transmembrane domain or a fragment thereof, a CD28 transmembrane domain or a fragment thereof, a TGFβR2 transmembrane domain or a fragment thereof, a 4- IBB transmembrane domain or a fragment thereof, an IL-10RA transmembrane domain or a fragment thereof, an IL-7RA transmembrane domain or a fragment thereof, an IL-4RA transmembrane domain or a fragment thereof, a Fas transmembrane domain or a fragment thereof, a MyD88 transmembrane domain or a fragment thereof, an TRAIL-R2 transmembrane domain or a fragment thereof, an IL12R transmembrane domain or a fragment thereof, an IL18R transmembrane domain or a fragment thereof, an IL21R transmembrane domain or a fragment thereof, and a combination thereof. [0333] In some embodiments, an IL12R transmembrane domain or a fragment thereof is an IL-12 receptor subunit beta-1 transmembrane domain or a fragment thereof. In some embodiments, an IL12R transmembrane domain or a fragment thereof is an IL-12 receptor subunit beta-2 transmembrane domain or a fragment thereof. In some embodiments, an IL18R transmembrane domain or a fragment thereof is an interleukin-18 receptor 1 transmembrane domain or a fragment thereof [0334] In some embodiments, the switch transmembrane domain is derived from any one selected from the group consisting of a human ICOS transmembrane domain or a fragment thereof, a human PD-1 transmembrane domain or a fragment thereof, a human CTLA4 transmembrane domain or a fragment thereof, a human CD200R transmembrane domain or a fragment thereof, a human BTLA transmembrane domain or a fragment thereof, a human TIM-3 transmembrane domain or a fragment thereof, a human TIGIT transmembrane domain or a fragment thereof, a human CD28 transmembrane domain or a fragment thereof, a human TGFβR2 transmembrane domain or a fragment thereof, a human 4-IBB transmembrane domain or a fragment thereof, a human IL-10RA transmembrane domain or a fragment thereof, a human IL-7RA transmembrane domain or a fragment thereof, a human IL-4RA transmembrane domain or a fragment thereof, a human Fas transmembrane domain or a fragment thereof, a human MyD88 transmembrane domain or a fragment thereof, a human TRAIL-R2 transmembrane domain or a fragment thereof, a human IL12R transmembrane
domain or a fragment thereof, a human IL18R transmembrane domain or a fragment thereof, a human IL21R transmembrane domain or a fragment thereof, and a combination thereof. [0335] In some embodiments, a human IL12R transmembrane domain or a fragment thereof is a human IL-12 receptor subunit beta-1 transmembrane domain or a fragment thereof. In some embodiments, a human IL12R transmembrane domain or a fragment thereof is a human IL-12 receptor subunit beta-2 transmembrane domain or a fragment thereof. In some embodiments, a human IL18R transmembrane domain or a fragment thereof is a human interleukin-18 receptor 1 trans-membrane domain or a fragment thereof. [0336] In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to any one selected from SEQ ID NOs:301, 279, 302, 303, 275, 304, 305, 306, 307, 272, 308, 309, 310, 311, 312, 317, 318, 319, 320, and a combination thereof. In some embodiments, the switch polypeptide comprises any one sequence selected from the group consisting of SEQ ID NOs:301, 279, 302, 303, 275, 304, 305, 306, 307, 272, 308, 309, 310, 311, 312, 317, 318, 319, 320, and a combination thereof. In some embodiments, the sequence of the switch intracellular domain is a sequence with at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 95%, 97%, 99.0%, 99.5%, 99.8, or 99.9% sequence identity to any one selected from SEQ ID NOs:301, 279, 302, 303, 275, 304, 305, 306, 307, 272, 308, 309, 310, 311, 312, 317, 318, 319, 320, and a combination thereof. In some embodiments, the sequence of the switch intracellular domain is any one sequence selected from the group consisting of SEQ ID NOs:301, 279, 302, 303, 275, 304, 305, 306, 307, 272, 308, 309, 310, 311, 312, 317, 318, 319, 320, and a combination thereof. [0337] PD-1 Transmembrane domain VGVVGGLLGSLVLLVWVLAVI (SEQ ID NO:301) [0338] 4-1BB Transmembrane domain IISFFLALTSTALLFLLFFLTLRFSVV (SEQ ID NO:279) [0339] ICOS Transmembrane FWLPIGCAAFVVVCILGCILI (SEQ ID NO:302) [0340] CTLA4 Transmembrane domain FLLWILAAVSSGLFFYSFLLT (SEQ ID NO:303) [0341] CD28 Transmembrane domain FWVLVVVGGVLACYSLLVTVAFIIFWV (SEQ ID NO:275) [0342] CD200R Transmembrane domain LTGNKSLYIELLPVPGAKKSA (SEQ ID NO:304)
[0343] BTLA Transmembrane domain LLPLGGLPLLITTCFCLFCCL (SEQ ID NO:305) [0344] TIM-3 Transmembrane domain IYIGAGICAGLALALIFGALI (SEQ ID NO:306) [0345] TIGIT Transmembrane domain LLGAMAATLVVICTAVIVVVA (SEQ ID NO:307) [0346] TGFβR2 Transmembrane domain VTGISLLPPLGVAISVIIIFY (SEQ ID NO:272) [0347] IL-10RA Transmembrane domain VIIFFAFVLLLSGALAYCLAL (SEQ ID NO:308) [0348] IL-4RA Transmembrane domain LLLGVSVSCIVILAVCLLCYVSIT (SEQ ID NO:309) [0349] IL-7RA Transmembrane domain PILLTISILSFFSVALLVILACVLW (SEQ ID NO:310) [0350] Fas Transmembrane domain LGWLCLLLLPIPLIVWV (SEQ ID NO:311) [0351] TRAILR2 Transmembrane domain LSGIIIGVTVAAVVLIVAVFV (SEQ ID NO:312) [0352] IL12R subunit beta-1 transmembrane domain WLIFFASLGSFLSILLVGVLGYLGL (SEQ ID NO:317) [0353] IL12R subunit beta-2 transmembrane domain WMAFVAPSICIAIIMVGIFST (SEQ ID NO:318) [0354] IL18R1 (Interleukin-18 receptor 1) transmembrane domain GMIIAVLILVAVVCLVTVCVI (SEQ ID NO:319) [0355] IL21R transmembrane domain GWNPHLLLLLLLVIVFIPAFW (SEQ ID NO:320) Additional Intracellular Domain [0356] In some embodiments, the switch polypeptide further comprises an additional intracellular domain. In some embodiments, the additional intracellular domain is operably linked to the C-terminus of the switch intracellular domain. In some embodiments, the additional intracellular domain is operably linked to the N-terminus of the switch intracellular domain. In some embodiments, the additional intracellular domain is operably linked to the C-terminus or the N-terminus of the switch intracellular domain. In some embodiments, the additional intracellular domain is operably linked to the C-terminus or the N-terminus of the switch intracellular domain via a linker. In some embodiments, the switch polypeptide may
comprise two or more additional intracellular domains. In some embodiments, the intracellular domains are operably linked to the C-terminus and the N-terminus of the switch intracellular domain, independently. In some embodiments, the additional intracellular domains are operably linked to the C-terminus, to the N-terminus, or independently to the C- terminus and the N-terminus of the switch intracellular domain via a linker. [0357] In some embodiments, the switch polypeptide further comprises one or more additional intracellular domains. In some embodiments, one or more additional intracellular domains are the same intracellular domain. In some embodiments, one or more additional intracellular domains are different intracellular domains. [0358] In some embodiments, the additional intracellular domain is derived from an intracellular domain of IL-15Ra. In some embodiments, the additional intracellular domain comprises an intracellular domain of IL-15Ra or a fragment thereof. [0359] In some embodiments, IL-15Ra or a fragment thereof comprises 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100, 101, 102, 103, 104, 105, 106, 107, 108, 109, 110, 111, 112, 113, 114, 115, 116, 117, 118, 119, 120, 121, 122, 123, 124, 125, 126, 127, 128, 129, 130, 131, 132, 133, 134, 135, 136, 137, 138, 139, 140, 141, 142, 143, 144, 145, 146, 147, 148, 149, 150, 151, 152, 153, 154, 155, 156, 157, 158, 159, 160, 161, 162, 163, 164, 165, 166, 167, 168, 169, 170, 171, 172, 173, 174, 175, 176, 177, 178, 179, 180, 181, 182, 183, 184, 185, 186, 187, 188, 189, 190, 191, 192, 193, 194, 195, 196, 197, 198, 199, 200, 201, 202, 203, 204, 205, 206, 207, 208, 209, 210, 211, 212, 213, 214, 215, 216, 217, 218, 219, 220, 221, 222, 223, 224, 225, 226, 227, 228, 229, 230, 231, 232, 233, 234, 235, 236, 237, 238, 239, 240, 241, 242, 243, 244, 245, 246, 247, 248, 249, 250, or more consecutive amino acid residues of IL-15Ra. In some embodiments, IL-15Ra or a fragment thereof comprises a sequence having at least about 50%, 55%, 60%, 6%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99% or more sequence identity to a sequence encoding IL-15Ra. In some embodiments, IL-15Ra or a fragment thereof comprises a sequence encoding IL-15Rα having a truncation of at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100, or more amino acids at the N- or C-terminus or at both the N- and C- terminus.
[0360] In some embodiments, the additional intracellular domain comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:372 or SEQ ID NO:383. In some embodiments, the additional intracellular domain comprises the sequence of SEQ ID NO:372 or SEQ ID NO:383. In some embodiments, the sequence of the additional intracellular domain is a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:372 or SEQ ID NO:383. In some embodiments, the sequence of the additional intracellular domain is the sequence of SEQ ID NO:372 or SEQ ID NO:383. [0361] In some embodiments, the additional intracellular domain comprises a sequence having an addition of amino acid residue(s) to the N-terminal end, C-terminal end, or both N- terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein. For example, in some embodiments, the additional intracellular domain comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein. In some embodiments, the additional intracellular domain comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein. In some embodiments, the additional intracellular domain comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of SEQ ID NO:372 or SEQ ID NO:383. In some embodiments, the additional intracellular domain comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115,
120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200 or more amino acid residues added to the N-terminal end, C-terminal end, or both N-terminal and C- terminal ends of the sequence of SEQ ID NO:372 or SEQ ID NO:383. [0362] In some embodiments, the additional intracellular domain comprises a sequence having a deletion of amino acid residue(s) from the N-terminal end, C-terminal end, or both N-terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein. For example, in some embodiments, the additional intracellular domain comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids deleted from the N-terminal or C- terminal end of the sequence of an IL-15Ra intracellular domain as described herein. In some embodiments, the additional intracellular domain comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids deleted from the N-terminal or C-terminal end of the sequence of an IL-15Ra intracellular domain as described herein. In some embodiments, the additional intracellular domain comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of an IL-15Ra intracellular domain as described herein. In some embodiments, the additional intracellular domain comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids independently deleted from both N-terminal and C- terminal ends of the sequence of an IL-15Ra intracellular domain as described herein. In some embodiments, the additional intracellular domain comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:372. In some embodiments, the additional intracellular domain comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids deleted
from the N-terminal or C-terminal end of the sequence of SEQ ID NO:372. In some embodiments, the additional intracellular domain comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids independently deleted from both N-terminal and C- terminal ends of the sequence of SEQ ID NO:372. In some embodiments, the additional intracellular domain comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:372. In some embodiments, the additional intracellular domain comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:383. In some embodiments, the additional intracellular domain comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids deleted from the N-terminal or C-terminal end of the sequence of SEQ ID NO:383. In some embodiments, the additional intracellular domain comprises a sequence having at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:383. In some embodiments, the additional intracellular domain comprises a sequence having about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 28, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 55, 60 , 65, 70, 75, 80, 85, 90, 95, 100, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, or 165 amino acids independently deleted from both N-terminal and C-terminal ends of the sequence of SEQ ID NO:383. Examples of Switch Polypeptides [0363] In some embodiments, the switch polypeptide comprises a transmembrane domain derived from a TGFBr2 transmembrane domain and an intracellular signaling domain of 4- 1BB.
[0364] In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:272 and a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:277. In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:272 operatively linked a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:277. In some embodiments, the switch polypeptide comprises the sequence of SEQ ID NO:272 and the sequence of SEQ ID NO:277. In some embodiments, the switch polypeptide comprises the sequence of SEQ ID NO:272 operatively linked to the sequence of SEQ ID NO:277. [0365] In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:285. In some embodiments, the switch polypeptide comprises the sequence of SEQ ID NO:285. In some embodiments, the sequence of the switch polypeptide is a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:285. In some embodiments, the sequence of the switch polypeptide is the sequence of SEQ ID NO:285. [0366] In some embodiments, the switch polypeptide comprises a transmembrane domain derived from a 4-1BB transmembrane domain and an intracellular signaling domain of 4- 1BB. [0367] In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:279 and a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:277. In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:279 operatively linked a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:277. In some embodiments, the switch polypeptide comprises the sequence of SEQ ID NO:279 and the sequence of SEQ ID NO:277. In some embodiments, the switch polypeptide comprises the sequence of SEQ ID NO:279 operatively linked to the sequence of SEQ ID NO:277.
[0368] In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:286. In some embodiments, the switch polypeptide comprises the sequence of SEQ ID NO:286. In some embodiments, the sequence of the switch polypeptide is a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:286. In some embodiments, the sequence of the switch polypeptide is the sequence of SEQ ID NO:286. [0369] In some embodiments, the switch polypeptide comprises a transmembrane domain derived from a TGFBr2 transmembrane domain and an intracellular signaling domain of CD28. [0370] In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:272 and a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:273. In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:272 operatively linked a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:273. In some embodiments, the switch polypeptide comprises the sequence of SEQ ID NO:272 and the sequence of SEQ ID NO:273. In some embodiments, the switch polypeptide comprises the sequence of SEQ ID NO:272 operatively linked to the sequence of SEQ ID NO:273. [0371] In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:283. In some embodiments, the switch polypeptide comprises the sequence of SEQ ID NO:283. In some embodiments, the sequence of the switch polypeptide is a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:283. In some embodiments, the sequence of the switch polypeptide is the sequence of SEQ ID NO:283. [0372] In some embodiments, the switch polypeptide comprises a transmembrane domain derived from a CD28 transmembrane domain and an intracellular signaling domain of CD28. [0373] In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or
99.9% sequence identity to SEQ ID NO:275 and a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:273. In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:275 operatively linked a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:273. In some embodiments, the switch polypeptide comprises the sequence of SEQ ID NO:275 and the sequence of SEQ ID NO:273. In some embodiments, the switch polypeptide comprises the sequence of SEQ ID NO:275 operatively linked to the sequence of SEQ ID NO:273. [0374] In some embodiments, the switch polypeptide comprises a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:284. In some embodiments, the switch polypeptide comprises the sequence of SEQ ID NO:284. In some embodiments, the sequence of the switch polypeptide is a sequence with at least 50%, 55%, 60%, 65%, 90%, 75%, 80%, 85%, 90%, 95%, 97%, 99.0%, 99.5%, 99.7%, or 99.9% sequence identity to SEQ ID NO:284. In some embodiments, the sequence of the switch polypeptide is the sequence of SEQ ID NO:284. T-cell Receptor (TCR) Fusion Proteins (TFPs) [0375] The present disclosure encompasses recombinant nucleic acid constructs encoding TFPs, wherein the TFP comprises a binding domain, e.g., an antibody or antibody fragment, a ligand, or a ligand binding protein, wherein the sequence of the binding domain is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof. The antibody or antibody fragment can comprise an antigen binding domain selected from a group consisting of an anti-CD19 binding domain, an anti-B-cell maturation antigen (BCMA) binding domain, an anti-mesothelin (MSLN) binding domain, an anti-CD20 binding domain, an anti-CD70 binding domain, an anti-CD79b binding domain, , an anti- PMSA binding domain, an anti-MUC16 binding domain, an anti-CD22 binding domain, an anti-PD-L1 binding domain, an anti BAFF receptor binding domain, an anti-Nectin-4 binding domain, an anti-TROP-2 binding domain, an anti-GPC3 binding domain, and anti-ROR-1 binding domain. The present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to a tumor associated antigen (a TAA) wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion
thereof. The present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to CD19, e.g., human CD19, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof. The present disclosure encompasses recombinant nucleic acid, e.g., DNA, constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to mesothelin, e.g., human mesothelin, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof. The present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to MUC16, e.g., human MUC16, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof. The present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to CD20, e.g., human CD20, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof. The present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to CD70, e.g., human CD70, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof. The present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to CD79B, e.g., human CD79B, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof. The present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to HER2, e.g., human HER2, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof. The present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to PSMA, e.g., human PSMA, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof. The present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to BCMA, e.g., human BCMA, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a
nucleic acid sequence encoding a TCR subunit or portion thereof. The present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to ROR1, e.g., human ROR1, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof. The present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to CD22, e.g., human CD22, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof. The present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to GPC3, e.g., human GPC3, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof. The present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to Nectin-4, e.g., human Nectin-4, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof. The present disclosure encompasses recombinant DNA constructs encoding TFPs, wherein the TFP comprises an antibody fragment that binds specifically to Trop-2, e.g., human Trop-2, wherein the sequence of the antibody fragment is contiguous with and in the same reading frame as a nucleic acid sequence encoding a TCR subunit or portion thereof. The TFPs provided herein are able to associate with one or more endogenous (or alternatively, one or more exogenous, or a combination of endogenous and exogenous) TCR subunits in order to form a functional TCR complex. [0376] In one aspect, the TFP of the present disclosure comprises a target-specific binding element otherwise referred to as an antigen binding domain. The choice of moiety depends upon the type and number of target antigen that define the surface of a target cell. For example, the antigen binding domain may be chosen to recognize a target antigen that acts as a cell surface marker on target cells associated with a particular disease state. Thus, examples of cell surface markers that may act as target antigens for the antigen binding domain in a TFP of the present disclosure include those associated with viral, bacterial and parasitic infections; autoimmune diseases; and cancerous diseases (e.g., malignant diseases). [0377] In one aspect, the TFP-mediated T cell response can be directed to an antigen of interest by way of engineering an antigen-binding domain into the TFP that specifically binds a desired antigen.
[0378] The antigen binding domain can be any domain that binds to the antigen including but not limited to a monoclonal antibody, a polyclonal antibody, a recombinant antibody, a human antibody, a humanized antibody, and a functional fragment thereof, including but not limited to a single-domain antibody such as a heavy chain variable domain (VH), a light chain variable domain (VL) and a variable domain (VHH) of a camelid derived nanobody, and to an alternative scaffold known in the art to function as antigen binding domain, such as a recombinant fibronectin domain, anticalin, DARPIN and the like. Likewise, a natural or synthetic ligand specifically recognizing and binding the target antigen can be used as antigen binding domain for the TFP. In some instances, it is beneficial for the antigen binding domain to be derived from the same species in which the TFP will ultimately be used in. For example, for use in humans, it may be beneficial for the antigen binding domain of the TFP to comprise human or humanized residues for the antigen binding domain of an antibody or antibody fragment. [0379] Thus, in one aspect, the antigen-binding domain comprises a humanized or human antibody or an antibody fragment, or a murine antibody or antibody fragment. In one embodiment, the murine, humanized or human anti-TAA binding domain comprises one or more (e.g., all three) light chain complementary determining region 1 (LC CDR1), light chain complementary determining region 2 (LC CDR2), and light chain complementary determining region 3 (LC CDR3) of a murine, humanized or human anti-TAA binding domain described herein, and/or one or more (e.g., all three) heavy chain complementary determining region 1 (HC CDR1), heavy chain complementary determining region 2 (HC CDR2), and heavy chain complementary determining region 3 (HC CDR3) of a murine, humanized or human anti-CD19 binding domain described herein, e.g., a murine, humanized or human anti-TAA binding domain comprising one or more, e.g., all three, LC CDRs and one or more, e.g., all three, HC CDRs. In one embodiment, the murine, humanized or human anti-CD19 binding domain comprises one or more (e.g., all three) heavy chain complementary determining region 1 (HC CDR1), heavy chain complementary determining region 2 (HC CDR2), and heavy chain complementary determining region 3 (HC CDR3) of a murine, humanized or human anti-TAA binding domain described herein, e.g., the murine, humanized or human anti-TAA binding domain has two variable heavy chain regions, each comprising a HC CDR1, a HC CDR2 and a HC CDR3 described herein. In one embodiment, the murine, humanized or human anti-TAA binding domain comprises a humanized or human light chain variable region described herein and/or a murine, humanized or human heavy chain variable region described herein. In one embodiment, the murine, humanized or human anti-TAA binding domain comprises a murine, humanized or human heavy chain
variable region described herein, e.g., at least two murine, humanized or human heavy chain variable regions described herein. In one embodiment, the anti-TAA binding domain is a scFv comprising a light chain and a heavy chain of an amino acid sequence provided herein. In an embodiment, the anti-TAA binding domain (e.g., a scFv) comprises: a light chain variable region comprising an amino acid sequence having at least one, two or three modifications (e.g., substitutions) but not more than 30, 20 or 10 modifications (e.g., substitutions) of an amino acid sequence of a light chain variable region provided herein, or a sequence with 95-99% identity with an amino acid sequence provided herein; and/or a heavy chain variable region comprising an amino acid sequence having at least one, two or three modifications (e.g., substitutions) but not more than 30, 20 or 10 modifications (e.g., substitutions) of an amino acid sequence of a heavy chain variable region provided herein, or a sequence with 95-99% identity to an amino acid sequence provided herein. In one embodiment, the murine, humanized or human anti-TAA binding domain is a scFv, and a light chain variable region comprising an amino acid sequence described herein, is attached to a heavy chain variable region comprising an amino acid sequence described herein, via a linker, e.g., a linker described herein. In one embodiment, the murine, humanized, or human anti-TAA binding domain includes a (Gly4-Ser)n linker, wherein n is 1, 2, 3, 4, 5, or 6, preferably 3 or 4 (SEQ ID NO: 400). The light chain variable region and heavy chain variable region of a scFv can be, e.g., in any of the following orientations: light chain variable region- linker-heavy chain variable region or heavy chain variable region-linker-light chain variable region. In some instances, the linker sequence comprises a long linker (LL) sequence. In some instances, the long linker sequence comprises (G4S)n, wherein n=2 to 4 (SEQ ID NO: 401). In some instances, the linker sequence comprises a short linker (SL) sequence. In some instances, the short linker sequence comprises (G4S)n, wherein n=1 to 3 (SEQ ID NO: 398). [0380] In some embodiments, the antigen binding domain is an antibody or a fragment thereof. In some embodiments, the antigen binding domain is a camelid antibody or a binding fragment thereof. In some embodiments, the antigen binding domain is a murine antibody or a binding fragment thereof. In some embodiments, the antigen binding domain is a human or humanized antibody or a binding fragment thereof. In some embodiments, the antigen binding domain is a single-chain variable fragment (scFv) or a single domain antibody (sdAb) domain. In some embodiments, the sdAb is a VHH. [0381] In some embodiments, the antigen binding domain is selected from the group consisting of an anti-CD19 binding domain, an anti-B-cell maturation antigen (BCMA) binding domain, an anti-mesothelin (MSLN) binding domain, an anti-CD20 binding domain,
an anti-CD70 binding domain, anti-MUC16 binding domain, an anti-Nectin-4 binding domain, an anti-GPC3 binding domain, and an anti-TROP-2 binding domain. [0382] In some embodiments, the antigen-binding domain comprises an anti-CD19 humanized or human antibody or an antibody fragment, or a murine antibody or antibody fragment having a light chain CDR1 of SEQ ID NO:73, a CDR2 of SEQ ID NO:75, and a CDR3 of SEQ ID NO:77 and a heavy chain CDR1 of SEQ ID NO:79, a CDR2 of SEQ ID NO:81, and a CDR3 of SEQ ID NO:83. In some embodiments, the anti-CD19 antibody is a murine scFv. In some embodiments, the anti-CD-19 antibody comprises a VL of SEQ ID NO:85 and a VH of SEQ ID NO:87. [0383] In some embodiments, the antigen-binding domain comprises an anti-mesothelin humanized or human single domain antibody or an antibody fragment having a CDR1 of SEQ ID NO:60, a CDR2 of SEQ ID NO:61, and a CDR3 of SEQ ID NO:62 or a CDR1 of SEQ ID NO:63, a CDR2 of SEQ ID NO:64, and a CDR3 of SEQ ID NO:65 or a CDR1 of SEQ ID NO:66, a CDR2 of SEQ ID NO:67, and a CDR3 of SEQ ID NO:68. In some embodiments, the anti-mesothelin antibody has a variable domain of SEQ ID NO:69, SEQ ID NO:70, or SEQ ID NO:71. [0384] In some embodiments, the antigen-binding domain comprises an anti-CD70 humanized or human single domain antibody or an antibody fragment having a CDR1 of SEQ ID NO:88, a CDR2 of SEQ ID NO:89, and a CDR3 of SEQ ID NO:90, or a CDR1 of SEQ ID NO:92, a CDR2 of SEQ ID NO:93, and a CDR3 of SEQ ID NO:94, or a CDR1 of SEQ ID NO:96, a CDR2 of SEQ ID NO:97, and a CDR3 of SEQ ID NO:98, or a CDR1 of SEQ ID NO:100, a CDR2 of SEQ ID NO:101, and a CDR3 of SEQ ID NO:102, or a CDR1 of SEQ ID NO:104, a CDR2 of SEQ ID NO:105, and a CDR3 of SEQ ID NO:106, or a CDR1 of SEQ ID NO:108, a CDR2 of SEQ ID NO:109, and a CDR3 of SEQ ID NO:110, or a CDR1 of SEQ ID NO:112, a CDR2 of SEQ ID NO:113, and a CDR3 of SEQ ID NO:114, or a CDR1 of SEQ ID NO: 116, a CDR2 of SEQ ID NO:117, and a CDR3 of SEQ ID NO:118, or a CDR1 of SEQ ID NO:120, a CDR2 of SEQ ID NO:121, and a CDR3 of SEQ ID NO:122. [0385] In some embodiments, the antigen-binding domain comprises an anti-CD70 single chain Fv (scFv) or an antibody fragment thereof. The anti-CD70 scFv or antibody fragment thereof can comprise a heavy chain complementary determining region 1 (CDRH1) having a sequence of SEQ ID NO: 361, a CDRH2 having a sequence of SEQ ID NO: 362, and a CDRH3 having a sequence of SEQ ID NOs: 363. The anti-CD70 scFv or antibody fragment thereof can comprise a light chain complementary determining region 1 (CDRL1) having a sequence of SEQ ID NO: 365 , a CDRL2 having a sequence of SEQ ID NO: 366, and a
CDRL3 having a sequence of SEQ ID NO: 367. The anti-CD70 scFv or antibody fragment thereof can comprise a heavy chain variable (VH) domain having at least 70%, 75%, 80%, 85%, 90%, 95%, or 100% sequence identity to SEQ ID NO: 364. The anti-CD70 scFv or antibody fragment thereof can comprise a light chain variable (VL) domain having at least 70%, 75%, 80%, 85%, 90%, 95%, or 100% sequence identity to SEQ ID NO: 368. [0386] In some aspects, a non-human antibody is humanized, where specific sequences or regions of the antibody are modified to increase similarity to an antibody naturally produced in a human or fragment thereof. In one aspect, the antigen binding domain is humanized. [0387] A humanized antibody can be produced using a variety of techniques known in the art, including but not limited to, CDR-grafting (see, e.g., European Patent No. EP 239,400; International Publication No. WO 91/09967; and U.S. Pat. Nos.5,225,539, 5,530,101, and 5,585,089, each of which is incorporated herein in its entirety by reference), veneering or resurfacing (see, e.g., European Patent Nos. EP 592,106 and EP 519,596; Padlan, 1991, Molecular Immunology, 28(4/5):489-498; Studnicka et al., 1994, Protein Engineering, 7(6):805-814; and Roguska et al., 1994, PNAS, 91:969-973, each of which is incorporated herein by its entirety by reference), chain shuffling (see, e.g., U.S. Pat. No.5,565,332, which is incorporated herein in its entirety by reference), and techniques disclosed in, e.g., U.S. Patent Application Publication No. US2005/0042664, U.S. Patent Application Publication No. US2005/0048617, U.S. Pat. No.6,407,213, U.S. Pat. No.5,766,886, International Publication No. WO 9317105, Tan et al., J. Immunol., 169:1119-25 (2002), Caldas et al., Protein Eng., 13(5):353-60 (2000), Morea et al., Methods, 20(3):267-79 (2000), Baca et al., J. Biol. Chem., 272(16):10678-84 (1997), Roguska et al., Protein Eng., 9(10):895-904 (1996), Couto et al., Cancer Res., 55 (23 Supp):5973s-5977s (1995), Couto et al., Cancer Res., 55(8):1717-22 (1995), Sandhu J S, Gene, 150(2):409-10 (1994), and Pedersen et al., J. Mol. Biol., 235(3):959-73 (1994), each of which is incorporated herein in its entirety by reference. Often, framework residues in the framework regions will be substituted with the corresponding residue from the CDR donor antibody to alter, for example improve, antigen binding. These framework substitutions are identified by methods well-known in the art, e.g., by modeling of the interactions of the CDR and framework residues to identify framework residues important for antigen binding and sequence comparison to identify unusual framework residues at particular positions (see, e.g., Queen et al., U.S. Pat. No.5,585,089; and Riechmann et al., 1988, Nature, 332:323, which are incorporated herein by reference in their entireties.) [0388] A humanized antibody or antibody fragment has one or more amino acid residues remaining in it from a source which is nonhuman. These nonhuman amino acid residues are
often referred to as “import” residues, which are typically taken from an “import” variable domain. As provided herein, humanized antibodies or antibody fragments comprise one or more CDRs from nonhuman immunoglobulin molecules and framework regions wherein the amino acid residues comprising the framework are derived completely or mostly from human germline. Multiple techniques for humanization of antibodies or antibody fragments are well- known in the art and can essentially be performed following the method of Winter and co- workers (Jones et al., Nature, 321:522-525 (1986); Riechmann et al., Nature, 332:323-327 (1988); Verhoeyen et al., Science, 239:1534-1536 (1988)), by substituting rodent CDRs or CDR sequences for the corresponding sequences of a human antibody, i.e., CDR-grafting (EP 239,400; PCT Publication No. WO 91/09967; and U.S. Pat. Nos.4,816,567; 6,331,415; 5,225,539; 5,530,101; 5,585,089; 6,548,640, the contents of which are incorporated herein by reference in their entirety). In such humanized antibodies and antibody fragments, substantially less than an intact human variable domain has been substituted by the corresponding sequence from a nonhuman species. Humanized antibodies are often human antibodies in which some CDR residues and possibly some framework (FR) residues are substituted by residues from analogous sites in rodent antibodies. Humanization of antibodies and antibody fragments can also be achieved by veneering or resurfacing (EP 592,106; EP 519,596; Padlan, 1991, Molecular Immunology, 28(4/5):489-498; Studnicka et al., Protein Engineering, 7(6):805-814 (1994); and Roguska et al., Proc. Natl. Acad. Sci. USA, 91:969- 973 (1994)) or chain shuffling (U.S. Pat. No.5,565,332), the contents of which are incorporated herein by reference in their entirety. [0389] The choice of human variable domains, both light and heavy, to be used in making the humanized antibodies is to reduce antigenicity. According to the so-called “best-fit” method, the sequence of the variable domain of a rodent antibody is screened against the entire library of known human variable-domain sequences. The human sequence which is closest to that of the rodent is then accepted as the human framework (FR) for the humanized antibody (Sims et al., J. Immunol., 151:2296 (1993); Chothia et al., J. Mol. Biol., 196:901 (1987), the contents of which are incorporated herein by reference herein in their entirety). Another method uses a particular framework derived from the consensus sequence of all human antibodies of a particular subgroup of light or heavy chains. The same framework may be used for several different humanized antibodies (see, e.g., Nicholson et al., Mol. Immun.34 (16-17): 1157-1165 (1997); Carter et al., Proc. Natl. Acad. Sci. USA, 89:4285 (1992); Presta et al., J. Immunol., 151:2623 (1993), the contents of which are incorporated herein by reference herein in their entirety). In some embodiments, the framework region, e.g., all four framework regions, of the heavy chain variable region are derived from a VH4-4-59 germline
sequence. In one embodiment, the framework region can comprise, one, two, three, four or five modifications, e.g., substitutions, e.g., from the amino acid at the corresponding murine sequence. In one embodiment, the framework region, e.g., all four framework regions of the light chain variable region are derived from a VK3-1.25 germline sequence. In one embodiment, the framework region can comprise, one, two, three, four or five modifications, e.g., substitutions, e.g., from the amino acid at the corresponding murine sequence. [0390] In some aspects, the portion of a TFP composition of the present disclosure that comprises an antibody fragment is humanized with retention of high affinity for the target antigen and other favorable biological properties. According to one aspect of the present disclosure, humanized antibodies and antibody fragments are prepared by a process of analysis of the parental sequences and various conceptual humanized products using three- dimensional models of the parental and humanized sequences. Three-dimensional immunoglobulin models are commonly available and are familiar to those skilled in the art. Computer programs are available which illustrate and display probable three-dimensional conformational structures of selected candidate immunoglobulin sequences. Inspection of these displays permits analysis of the likely role of the residues in the functioning of the candidate immunoglobulin sequence, e.g., the analysis of residues that influence the ability of the candidate immunoglobulin to bind the target antigen. In this way, FR residues can be selected and combined from the recipient and import sequences so that the desired antibody or antibody fragment characteristic, such as increased affinity for the target antigen, is achieved. In general, the CDR residues are directly and most substantially involved in influencing antigen binding. [0391] A humanized antibody or antibody fragment may retain a similar antigenic specificity as the original antibody, e.g., in the present disclosure, the ability to bind human a tumor associated antigen (TAA). In some embodiments, a humanized antibody or antibody fragment may have improved affinity and/or specificity of binding to, e.g., human CD19, human BCMA, or another tumor associated antigen. [0392] In one aspect, the binding domain is characterized by particular functional features or properties of an antibody or antibody fragment. For example, in one aspect, the portion of a TFP composition of the present disclosure that comprises an antigen binding domain specifically binds human CD19. In one aspect, the antigen binding domain has the same or a similar binding specificity to human CD19 as the FMC63 scFv described in Nicholson et al., Mol. Immun.34 (16-17): 1157-1165 (1997). In one aspect, the present disclosure relates to an antigen binding domain comprising an antibody or antibody fragment, wherein the antibody binding domain specifically binds to a CD19 or BCMA protein or fragment thereof, wherein
the antibody or antibody fragment comprises a variable light chain and/or a variable heavy chain that includes an amino acid sequence provided herein. In certain aspects, the scFv is contiguous with and in the same reading frame as a leader sequence. [0393] In one aspect, the anti-tumor-associated antigen binding domain is a fragment, e.g., a single chain variable fragment (scFv). In one aspect, the anti-TAA binding domain is a Fv, a Fab, a (Fab’)2, or a bi-functional (e.g., bi-specific) hybrid antibody (e.g., Lanzavecchia et al., Eur. J. Immunol.17, 105 (1987)). In one aspect, the antibodies and fragments thereof of the present disclosure binds a CD19 protein with wild-type or enhanced affinity. In another aspect, the anti-TAA binding domain comprises a single domain antibody (sdAb or VHH). [0394] Also provided herein are methods for obtaining an antibody antigen binding domain specific for a target antigen (e.g., CD19, BCMA, MSLN, CD70, or any target antigen described elsewhere herein for targets of fusion moiety binding domains), the method comprising providing by way of addition, deletion, substitution or insertion of one or more amino acids in the amino acid sequence of a VH domain set out herein a VH domain which is an amino acid sequence variant of the VH domain, optionally combining the VH domain thus provided with one or more VL domains, and testing the VH domain or VH/VL combination or combinations to identify a specific binding member or an antibody antigen binding domain specific for a target antigen of interest (e.g., MSLN, CD79B, etc.) and optionally with one or more desired properties. [0395] In some instances, VH domains and scFvs can be prepared according to method known in the art (see, for example, Bird et al., (1988) Science 242:423-426 and Huston et al., (1988) Proc. Natl. Acad. Sci. USA 85:5879-5883). scFv molecules can be produced by linking VH and VL regions together using flexible polypeptide linkers. The scFv molecules comprise a linker (e.g., a Ser-Gly linker) with an optimized length and/or amino acid composition. The linker length can greatly affect how the variable regions of a scFv fold and interact. In fact, if a short polypeptide linker is employed (e.g., between 5-10 amino acids) intra-chain folding is prevented. Inter-chain folding is also required to bring the two variable regions together to form a functional epitope binding site. In some instances, the linker sequence comprises a linker sequence. In some instances, the long linker sequence comprises (G4S)n, wherein n=2 to 4 (SEQ ID NO: 401). In some instances, the linker sequence comprises (G4S)n, wherein n=1 to 3 (SEQ ID NO: 398). For examples of linker orientation and size see, e.g., Hollinger et al., 1993 Proc Natl Acad. Sci. U.S.A.90:6444-6448, U.S. Patent Application Publication Nos.2005/0100543, 2005/0175606, 2007/0014794, and PCT publication Nos. WO2006/020258 and WO2007/024715, each of which is incorporated herein by reference.
[0396] An scFv can comprise a linker of about 10, 11, 12, 13, 14, 15 or greater than 15 residues between its VL and VH regions. The linker sequence may comprise any naturally occurring amino acid. In some embodiments, the linker sequence comprises amino acids glycine and serine. In another embodiment, the linker sequence comprises sets of glycine and serine repeats such as (Gly4Ser)n, where n is a positive integer equal to or greater than 1 (SEQ ID NO: 402). In one embodiment, the linker can be (Gly4Ser)4 (SEQ ID NO: 394) or (Gly4Ser)3 (SEQ ID NO: 395). Variation in the linker length may retain or enhance activity, giving rise to superior efficacy in activity studies. In some instances, the linker sequence comprises (G4S)n, wherein n=2 to 4 (SEQ ID NO: 401). In some instances, the linker sequence comprises (G4S)n, wherein n=1 to 3 (SEQ ID NO: 398). In some embodiments, the linker sequence comprises GGSGGSGGSGGS (SEQ ID NO: 369). Stability and Mutations [0397] The stability of a tumor associated antigen binding domain, e.g., scFv molecules (e.g., soluble scFv) can be evaluated in reference to the biophysical properties (e.g., thermal stability) of a conventional control scFv molecule or a full-length antibody. In one embodiment, the humanized or human scFv has a thermal stability that is greater than about 0.1, about 0.25, about 0.5, about 0.75, about 1, about 1.25, about 1.5, about 1.75, about 2, about 2.5, about 3, about 3.5, about 4, about 4.5, about 5, about 5.5, about 6, about 6.5, about 7, about 7.5, about 8, about 8.5, about 9, about 9.5, about 10 degrees, about 11 degrees, about 12 degrees, about 13 degrees, about 14 degrees, or about 15 degrees Celsius than a parent scFv in the described assays. [0398] The improved thermal stability of the anti-TAA binding domain, e.g., scFv is subsequently conferred to the entire TAA-TFP construct, leading to improved therapeutic properties of the anti-TAA TFP construct. The thermal stability of the binding domain, e.g., scFv or sdAb, can be improved by at least about 2 °C or 3 °C as compared to a conventional antibody. In one embodiment, the binding domain, has a 1 °C improved thermal stability as compared to a conventional antibody. In another embodiment, the binding domain, has a 2 °C improved thermal stability as compared to a conventional antibody. In another embodiment, the scFv has a 4 °C, 5 °C, 6 °C, 7 °C, 8 °C, 9 °C, 10 °C, 11 °C, 12 °C, 13 °C, 14 °C, or 15 °C improved thermal stability as compared to a conventional antibody. Comparisons can be made, for example, between the scFv molecules as described herein and scFv molecules or Fab fragments of an antibody from which the scFv VH and VL were derived. Thermal stability can be measured using methods known in the art. For example, in one embodiment, TM can be measured. Methods for measuring TM and other methods of determining protein stability are described in more detail below.
[0399] Mutations in antibody sequences (arising through humanization or direct mutagenesis of the soluble scFv) alter the stability of the antibody or fragment thereof and improve the overall stability of the antibody and the TFP construct. Stability of the humanized antibody or fragment thereof is compared against the murine antibody or fragment thereof using measurements such as TM, temperature denaturation and temperature aggregation. In one embodiment, the binding domain, e.g., a scFv or sdAb, comprises at least one mutation arising from the humanization process such that the mutated scFv confers improved stability to the anti-TAA TFP construct. In another embodiment, the anti-TAA binding domain, e.g., scFv or sdAb, comprises at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 mutations arising from the humanization process such that the mutated scFv or sdAb confers improved stability to the TAA-TFP construct. [0400] In one aspect, the antigen binding domain of the TFP comprises an amino acid sequence that is homologous to an antigen binding domain amino acid sequence described herein, and the antigen binding domain retains the desired functional properties of the anti- tumor-associated antigen antibody fragments described herein. In one specific aspect, the TFP composition of the present disclosure comprises an antibody fragment. In a further aspect, that antibody fragment comprises a scFv. [0401] In various aspects, the antigen binding domain of the TFP is engineered by modifying one or more amino acids within one or both variable regions (e.g., VH and/or VL), for example within one or more CDR regions and/or within one or more framework regions. In one specific aspect, the TFP composition of the present disclosure comprises an antibody fragment. In a further aspect, that antibody fragment comprises a scFv. [0402] It will be understood by one of ordinary skill in the art that the antibody or antibody fragment of the present disclosure may further be modified such that they vary in amino acid sequence (e.g., from wild-type), but not in desired activity. For example, additional nucleotide substitutions leading to amino acid substitutions at “non-essential” amino acid residues may be made to the protein. For example, a nonessential amino acid residue in a molecule may be replaced with another amino acid residue from the same side chain family. In another embodiment, a string of amino acids can be replaced with a structurally similar string that differs in order and/or composition of side chain family members, e.g., a conservative substitution, in which an amino acid residue is replaced with an amino acid residue having a similar side chain, may be made. [0403] Families of amino acid residues having similar side chains have been defined in the art, including basic side chains (e.g., lysine, arginine, histidine), acidic side chains (e.g., aspartic acid, glutamic acid), uncharged polar side chains (e.g., glycine, asparagine,
glutamine, serine, threonine, tyrosine, cysteine), nonpolar side chains (e.g., alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine, tryptophan), beta-branched side chains (e.g., threonine, valine, isoleucine) and aromatic side chains (e.g., tyrosine, phenylalanine, tryptophan, histidine). [0404] Percent identity in the context of two or more nucleic acids or polypeptide sequences refers to two or more sequences that are the same. Two sequences are “substantially identical” if two sequences have a specified percentage of amino acid residues or nucleotides that are the same (e.g., 60% identity, optionally 70%, 71% , 72%, 73%, 74%, 75%, 76%, 77%, 78%, 79%, 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity over a specified region, or, when not specified, over the entire sequence), when compared and aligned for maximum correspondence over a comparison window, or designated region as measured using one of the following sequence comparison algorithms or by manual alignment and visual inspection. Optionally, the identity exists over a region that is at least about 50 nucleotides (or 10 amino acids) in length, or more preferably over a region that is 100 to 500 or 1000 or more nucleotides (or 20, 50, 200 or more amino acids) in length. [0405] For sequence comparison, typically one sequence acts as a reference sequence, to which test sequences are compared. When using a sequence comparison algorithm, test and reference sequences are entered into a computer, subsequence coordinates are designated, if necessary, and sequence algorithm program parameters are designated. Default program parameters can be used, or alternative parameters can be designated. The sequence comparison algorithm then calculates the percent sequence identities for the test sequences relative to the reference sequence, based on the program parameters. Methods of alignment of sequences for comparison are well known in the art. Optimal alignment of sequences for comparison can be conducted, e.g., by the local homology algorithm of Smith and Waterman, (1970) Adv. Appl. Math.2:482c, by the homology alignment algorithm of Needleman and Wunsch, (1970) J. Mol. Biol.48:443, by the search for similarity method of Pearson and Lipman, (1988) Proc. Natl. Acad. Sci. USA 85:2444, by computerized implementations of these algorithms (GAP, BESTFIT, FASTA, and TFASTA in the Wisconsin Genetics Software Package, Genetics Computer Group, 575 Science Dr., Madison, Wis.), or by manual alignment and visual inspection (see, e.g., Brent et al., (2003) Current Protocols in Molecular Biology). Two examples of algorithms that are suitable for determining percent sequence identity and sequence similarity are the BLAST and BLAST 2.0 algorithms, which are described in Altschul et al., (1977) Nuc. Acids Res.25:3389-3402; and Altschul et al.,
(1990) J. Mol. Biol.215:403-410, respectively. Software for performing BLAST analyses is publicly available through the National Center for Biotechnology Information. [0406] In one aspect, the present disclosure contemplates modifications of the starting antibody or fragment (e.g., scFv) amino acid sequence that generate functionally equivalent molecules. For example, the VH or VL of a binding domain, e.g., scFv, comprised in the TFP can be modified to retain at least about 70%, 71%.72%.73%, 74%, 75%, 76%, 77%, 78%, 79%, 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% identity of the starting VH or VL framework region of the anti- CD19 binding domain, e.g., scFv. The present disclosure contemplates modifications of the entire TFP construct, e.g., modifications in one or more amino acid sequences of the various domains of the TFP construct in order to generate functionally equivalent molecules. The TFP construct can be modified to retain at least about 70%, 71%.72%.73%, 74%, 75%, 76%, 77%, 78%, 79%, 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% identity of the starting TFP construct. Extracellular Domain [0407] The extracellular domain may be derived either from a natural or from a recombinant source. Where the source is natural, the domain may be derived from any protein, but in particular a membrane-bound or transmembrane protein. In one aspect the extracellular domain is capable of associating with the transmembrane domain. An extracellular domain of particular use in this present disclosure may include at least the extracellular region(s) of e.g., the alpha, beta or zeta chain of the T cell receptor, or CD3 epsilon, CD3 gamma, or CD3 delta, or in alternative embodiments, CD28, CD45, CD4, CD5, CD8, CD9, CD16, CD22, CD33, CD37, CD64, CD80, CD86, CD134, CD137, or CD154. In some embodiments, the extracellular domain is a TCR extracellular domain. In some instances, the TCR extracellular domain comprises an extracellular domain or portion thereof of a protein selected from the group consisting of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, a CD3 delta TCR subunit, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. [0408] In some embodiments, the TCR extracellular domain comprises an extracellular domain or portion thereof of a TCR alpha chain, a TCR beta chain, a TCR delta chain, or a TCR gamma chain. In some embodiments, the TCR extracellular domain comprises the extracellular portion of a constant (an IgC) domain of a TCR alpha chain, a TCR beta chain, a TCR delta chain, or a TCR gamma chain.
[0409] In some embodiments, the extracellular domain comprises, or comprises at least 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100 or more consecutive amino acid residues of the extracellular domain of a TCR alpha chain, a TCR beta chain, a TCR delta chain, or a TCR gamma chain. In some embodiments, the extracellular domain comprises a sequence having at least about 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99% or more sequence identity to a sequence encoding the extracellular domain of a TCR alpha chain, a TCR beta chain, a TCR delta chain, or a TCR gamma chain. In some embodiments, the extracellular domain comprises a sequence encoding the extracellular domain of a TCR alpha chain, a TCR beta chain, a TCR delta chain, or a TCR gamma chain having a truncation of at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25 or more amino acids at the N- or C-terminus or at both the N- and C-terminus. [0410] In some embodiments, the extracellular domain comprises, or comprises at least 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100 or more consecutive amino acid residues of the extracellular portion of a constant (an IgC) domain of TCR alpha, a TCR beta, a TCR delta, or a TCR gamma. In some embodiments, the extracellular domain comprises a sequence having at least about 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99% or more sequence identity to a sequence encoding the extracellular portion of a constant (an IgC) domain of TCR alpha, a TCR beta, a TCR delta, or a TCR gamma. In some embodiments, the extracellular domain comprises a sequence encoding the extracellular portion of a constant (an IgC) domain of TCR alpha, TCR beta, TCR delta, or TCR gamma having a truncation of at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25 or more amino acids at the N- or C-terminus or at both the N- and C-terminus. [0411] In some embodiments, the extracellular domain comprises, or comprises at least 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100 or more consecutive
amino acid residues of the extracellular domain of a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit. In some embodiments, the extracellular domain comprises a sequence having at least about 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99% or more sequence identity to a sequence encoding the extracellular domain of a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit. In some embodiments, the extracellular domain comprises a sequence encoding the extracellular domain of a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit having a truncation of at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25 or more amino acids at the N- or C- terminus or at both the N- and C-terminus. [0412] The extracellular domain can be a TCR extracellular domain. The TCR extracellular domain can be derived from a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit or a CD3 delta TCR subunit. The extracellular domain can be a full-length TCR extracellular domain or fragment (e.g., functional fragment) thereof. The extracellular domain can comprise a variable domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain. The extracellular domain can comprise a variable domain and a constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain. In some cases, the extracellular domain may not comprise a variable domain. [0413] The extracellular domain can comprise a constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain. The extracellular domain can comprise a full-length constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain. The extracellular domain can comprise a fragment (e.g., functional fragment) of the full-length constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain. For example, the extracellular domain can comprise at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of the constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain. [0414] The TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain described herein can be derived from various species. The TCR chain can be a murine or human TCR chain. For example, the extracellular domain can comprise a constant domain of a murine TCR alpha chain, a murine TCR beta chain, a human TCR gamma chain or a human TCR delta chain. Transmembrane Domain
[0415] In general, a TFP sequence contains an extracellular domain and a transmembrane domain encoded by a single genomic sequence. In alternative embodiments, a TFP can be designed to comprise a transmembrane domain that is heterologous to the extracellular domain of the TFP. A transmembrane domain can include one or more additional amino acids adjacent to the transmembrane region, e.g., one or more amino acid associated with the extracellular region of the protein from which the transmembrane was derived (e.g., at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, or more amino acids of the extracellular region) and/or one or more additional amino acids associated with the intracellular region of the protein from which the transmembrane protein is derived (e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, or more amino acids of the intracellular region). In some cases, the transmembrane domain can include at least 30, 35, 40, 45, 50, 55, 60 or more amino acids of the extracellular region. In some cases, the transmembrane domain can include at least 30, 35, 40, 45, 50, 55, 60 or more amino acids of the intracellular region. In one aspect, the transmembrane domain is one that is associated with one of the other domains of the TFP is used. In some instances, the transmembrane domain can be selected or modified by amino acid substitution to avoid binding of such domains to the transmembrane domains of the same or different surface membrane proteins, e.g., to minimize interactions with other members of the receptor complex. In one aspect, the transmembrane domain is capable of homodimerization with another TFP on the TFP-T cell surface. In a different aspect the amino acid sequence of the transmembrane domain may be modified or substituted so as to minimize interactions with the binding domains of the native binding partner present in the same TFP. [0416] The transmembrane domain may be derived either from a natural or from a recombinant source. Where the source is natural, the domain may be derived from any membrane-bound or transmembrane protein. In one aspect the transmembrane domain is capable of signaling to the intracellular domain(s) whenever the TFP has bound to a target. In some instances, the TCR-integrating subunit comprises a transmembrane domain comprising a transmembrane domain of a protein selected from the group consisting of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, a TCR zeta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, a CD3 delta TCR subunit, CD45, CD4, CD5, CD8, CD9, CD16, CD22, CD33, CD28, CD37, CD64, CD80, CD86, CD134, CD137, CD154, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications.
[0417] In some embodiments, the transmembrane domain comprises, or comprises at least 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, or 30 or more consecutive amino acid residues of the transmembrane domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit. In some embodiments, the transmembrane domain comprises a sequence having at least about 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99% or more sequence identity to a sequence encoding the transmembrane domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit. In some embodiments, the transmembrane domain comprises a sequence encoding the transmembrane domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit having a truncation of at least 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 or more amino acids at the N- or C-terminus or at both the N- and C-terminus. [0418] In some instances, the transmembrane domain can be attached to the extracellular region of the TFP, e.g., the antigen binding domain of the TFP, via a hinge, e.g., a hinge from a human protein. For example, in one embodiment, the hinge can be a human immunoglobulin (Ig) hinge, e.g., an IgG4 hinge, or a CD8a hinge. Linkers [0419] Optionally, a short oligo- or polypeptide linker, between 2 and 10 amino acids in length may form the linkage between the binding element and the TCR extracellular domain of the TFP. A glycine-serine doublet provides a particularly suitable linker. In some cases, the linker may be at least about 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, or more in length. For example, in one aspect, the linker comprises the amino acid sequence of GGGGSGGGGS (SEQ ID NO: 403) or a sequence (GGGGS)x (SEQ ID NO: 390) or (G4S)n (SEQ ID NO: 390), wherein X or n is 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 or more. In some embodiments, X or n is an integer from 1 to 10. In some embodiments, X or n is an integer from 1 to 4. In some embodiments, X or n is 2. In some embodiments, X or n is 4. In some embodiments, the linker is encoded by a nucleotide sequence of GGTGGCGGAGGTTCTGGAGGTGGAGGTTCC (SEQ ID NO: 404). Cytoplasmic Domain [0420] The cytoplasmic domain of the TFP can include an intracellular domain. In some embodiments, the intracellular domain is from CD3 gamma, CD3 delta, CD3 epsilon, TCR alpha, TCR beta, TCR gamma, or TCR delta. In some embodiments, the intracellular domain comprises a signaling domain, if the TFP contains CD3 gamma, delta or epsilon
polypeptides; TCR alpha, TCR beta, TCR gamma, and TCR delta subunits generally have short (e.g., 1-19 amino acids in length) intracellular domains and are generally lacking in a signaling domain. An intracellular signaling domain is generally responsible for activation of at least one of the normal effector functions of the immune cell in which the TFP has been introduced. While the intracellular domains of TCR alpha, TCR beta, TCR gamma, and TCR delta do not have signaling domains, they are able to recruit proteins having a primary intracellular signaling domain described herein, e.g., CD3 zeta, which functions as an intracellular signaling domain. The term “effector function” refers to a specialized function of a cell. Effector function of a T cell, for example, may be cytolytic activity or helper activity including the secretion of cytokines. Thus, the term “intracellular signaling domain,” as used herein, refers to the portion of a protein which transduces the effector function signal and directs the cell to perform a specialized function. While usually the entire intracellular signaling domain can be employed, in many cases it is not necessary to use the entire chain. To the extent that a truncated portion of the intracellular signaling domain is used, such truncated portion may be used in place of the intact chain as long as it transduces the effector function signal. The term intracellular signaling domain is thus meant to include any truncated portion of the intracellular signaling domain sufficient to transduce the effector function signal. [0421] Examples of intracellular domains for use in the TFP of the present disclosure include the cytoplasmic sequences of the T cell receptor (TCR) and co-receptors that are able to act in concert to initiate signal transduction following antigen receptor engagement, as well as any derivative or variant of these sequences and any recombinant sequence that has the same functional capability. In some embodiments, the intracellular domain comprises the intracellular domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit. In some embodiments, the intracellular domain comprises, or comprises at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 or more consecutive amino acid residues of the intracellular domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, or a TCR delta chain. In some embodiments, the intracellular domain comprises a sequence having at least about 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99% or more sequence identity to a sequence encoding the intracellular domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, or a TCR delta chain. In some embodiments, the transmembrane domain comprises a sequence encoding the intracellular domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, or a TCR delta chain
having a truncation of at least 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 or more amino acids at the N- or C-terminus or at both the N- and C-terminus. [0422] In some embodiments, the intracellular domain comprises, or comprises at least 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, or 62 or more consecutive amino acid residues of the intracellular domain of a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit. In some embodiments, the intracellular domain comprises a sequence having at least about 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99% or more sequence identity to a sequence encoding the intracellular domain of a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit. In some embodiments, the intracellular domain comprises a sequence encoding the intracellular domain of a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, or a CD3 delta TCR subunit having a truncation of at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25 or more amino acids at the N- or C-terminus or at both the N- and C-terminus. [0423] It is known that signals generated through the TCR alone are insufficient for full activation of naive T cells and that a secondary and/or costimulatory signal is required. Thus, naïve T cell activation can be said to be mediated by two distinct classes of cytoplasmic signaling sequences: those that initiate antigen-dependent primary activation through the TCR (primary intracellular signaling domains) and those that act in an antigen-independent manner to provide a secondary or costimulatory signal (secondary cytoplasmic domain, e.g., a costimulatory domain). [0424] A primary signaling domain regulates primary activation of the TCR complex either in a stimulatory way, or in an inhibitory way. Primary intracellular signaling domains that act in a stimulatory manner may contain signaling motifs which are known as immunoreceptor tyrosine-based activation motifs (ITAMs). [0425] Examples of ITAMs containing primary intracellular signaling domains that are of particular use in the present disclosure include those of CD3 zeta, FcR gamma, FcR beta, CD3 gamma, CD3 delta, CD3 epsilon, CD5, CD22, CD79a, CD79b, and CD66d. In one embodiment, a TFP of the present disclosure comprises an intracellular signaling domain, e.g., a primary signaling domain of CD3-epsilon. In one embodiment, a primary signaling domain comprises a modified ITAM domain, e.g., a mutated ITAM domain which has altered (e.g., increased or decreased) activity as compared to the native ITAM domain. In one embodiment, a primary signaling domain comprises a modified ITAM-containing primary intracellular signaling domain, e.g., an optimized and/or truncated ITAM-containing primary
intracellular signaling domain. In an embodiment, a primary signaling domain comprises one, two, three, four or more ITAM motifs. [0426] The intracellular signaling domain of the TFP can comprise a CD3 signaling domain, e.g., CD3 epsilon, CD3 delta, CD3 gamma, or CD3 zeta, by itself or it can be combined with any other desired intracellular signaling domain(s) useful in the context of a TFP of the present disclosure. For example, the intracellular signaling domain of the TFP can comprise a CD3 epsilon chain portion and a costimulatory signaling domain. The costimulatory signaling domain refers to a portion of the TFP comprising the intracellular domain of a costimulatory molecule. A costimulatory molecule is a cell surface molecule other than an antigen receptor or its ligands that is required for an efficient response of lymphocytes to an antigen. Examples of such molecules include CD27, CD28, 4-1BB (CD137), OX40, CD30, CD40, PD-1, ICOS, lymphocyte function-associated antigen-1 (LFA-1), CD2, CD7, LIGHT, NKG2C, B7-H3, and a ligand that specifically binds with CD83, and the like. For example, CD27 costimulation has been demonstrated to enhance expansion, effector function, and survival of human TFP-T cells in vitro and augments human T cell persistence and antitumor activity in vivo (Song et al., Blood.2012; 119(3):696-706). [0427] The intracellular signaling sequences within the cytoplasmic portion of the TFP of the present disclosure may be linked to each other in a random or specified order. Optionally, a short oligo- or polypeptide linker, for example, between 2 and 10 amino acids (e.g., 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids) in length may form the linkage between intracellular signaling sequences. [0428] In one embodiment, a glycine-serine doublet can be used as a suitable linker. In one embodiment, a single amino acid, e.g., an alanine, a glycine, can be used as a suitable linker. [0429] In one aspect, the TFPs described herein may comprise a TCR extracellular domain, a TCR transmembrane domain, and a TCR intracellular domain, wherein at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from the same TCR subunit. In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from TCR alpha. In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from TCR beta. In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from TCR gamma. In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from TCR delta. In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can
be from CD3 epsilon. In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from CD3 delta. In some embodiments, at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from CD3 gamma. [0430] In one aspect, the TFPs described herein may comprise a TCR extracellular domain, a TCR transmembrane domain, and a TCR intracellular domain, wherein all three of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from the same TCR subunit. In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from CD3 epsilon. In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from CD3 delta. In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain can be from CD3 gamma. In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain may comprise the constant domain of TCR alpha. In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain may comprise the constant domain of TCR beta. In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain may comprise the constant domain of TCR gamma. In some embodiments, the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain may comprise the constant domain of TCR delta. In some embodiments, the constant domain of TCR alpha or the constant domain of TCR beta may be murine. [0431] In one aspect, the TFP-expressing cell co-expressing a switch polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof; and/or the TFP-expressing cell co-expressing a DN TGFBr2 as described herein can further comprise a second TFP, e.g., a second TFP that includes a different antigen binding domain, e.g., to the same target or a different target. In one embodiment, when the TFP-expressing cell comprises two or more different TFPs, the antigen binding domains of the different TFPs can be such that the antigen binding domains do not interact with one another. For example, a cell expressing a first and second TFP can have an antigen binding domain of the first TFP, e.g., as a fragment, e.g., a scFv, that does not form an association with the antigen binding domain of the second TFP, e.g., the antigen binding domain of the second TFP is a VHH. [0432] TFP constructs can be generated as previously described. An anti-MSLN or CD19 binder can be linked to a CD3 or TCR DNA fragment by either a DNA sequence encoding a short linker (SL): AAAGGGGSGGGGSGGGGSLE (SEQ ID NO: 387) or a long linker (LL): AAAIEVMYPPPYLGGGGSGGGGSGGGGSLE (SEQ ID NO: 388) into pLRPO or p510
vector. In some embodiments, the TFP used is TC-210 (e.g., an anti-MSLN MH1e VHH antibody linked to CD3 epsilon) having the sequence of SEQ ID NO: 195. The TC-210 sequence can comprise a CSF2RA signal peptide, an anti-MSLN sdAb, A3(G4S)3LE linker (SEQ ID NO: 387) and/or a CD3 epsilon. In some embodiments, TC-210 comprises SEQ ID NO: 201, SEQ ID NO: 69, SEQ ID NO: 387, and SEQ ID NO: 258. In some embodiments, the TFP used is TC-110 (e.g., an anti-CD19 FMC63 scFv antibody linked to CD3 epsilon) having the sequence of SEQ ID NO: 196. [0433] Anti-MSLN-CD3 epsilon (SEQ ID NO: 195) MLLLVTSLLLCELPHPAFLLIPEVQLVESGGGLVQPGGSLRLSCAASGGDWSANFMY WYRQAPGKQRELVARISGRGVVDYVESVKGRFTISRDNSKNTLYLQMNSLRAEDTA VYYCAVASYWGQGTLVTVSSAAAGGGGSGGGGSGGGGSLEDGNEEMGGITQTPYK VSISGTTVILTCPQYPGSEILWQHNDKNIGGDEDDKNIGSDEDHLSLKEFSELEQSGYY VCYPRGSKPEDANFYLYLRARVCENCMEMDVMSVATIVIVDICITGGLLLLVYYWS KNRKAKAKPVTRGAGAGGRQRGQNKERPPPVPNPDYEPIRKGQRDLYSGLNQRRI [0434] Anti-CD19-CD3 epsilon (SEQ ID NO: 196) MLLLVTSLLLCELPHPAFLLIPDIQMTQTTSSLSASLGDRVTISCRASQDISKYLNWYQ QKPDGTVKLLIYHTSRLHSGVPSRFSGSGSGTDYSLTISNLEQEDIATYFCQQGNTLPY TFGGGTKLEITGGGGSGGGGSGGGGSEVKLQESGPGLVAPSQSLSVTCTVSGVSLPD YGVSWIRQPPRKGLEWLGVIWGSETTYYNSALKSRLTIIKDNSKSQVFLKMNSLQTD DTAIYYCAKHYYYGGSYAMDYWGQGTSVTVSSAAAGGGGSGGGGSGGGGSLEDG NEEMGGITQTPYKVSISGTTVILTCPQYPGSEILWQHNDKNIGGDEDDKNIGSDEDHL SLKEFSELEQSGYYVCYPRGSKPEDANFYLYLRARVCENCMEMDVMSVATIVIVDIC ITGGLLLLVYYWSKNRKAKAKPVTRGAGAGGRQRGQNKERPPPVPNPDYEPIRKGQ RDLYSGLNQRRI Recombinant Nucleic Acid Molecules [0435] Disclosed herein are recombinant nucleic acid molecules comprising a first nucleic acid sequence encoding a T cell receptor (TCR) fusion protein (TFP) as described herein and a second nucleic acid sequence encoding a dominant negative transforming growth factor beta receptor II (TGFBr2) as described herein. Disclosed herein are recombinant nucleic acid molecules comprising a first nucleic acid sequence encoding a T cell receptor (TCR) fusion protein (TFP) as described herein and a second nucleic acid sequence encoding a switch polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof as described herein. Recombinant Nucleic Acid Encoding a TFP and a TCR Constant Domain
[0436] Disclosed herein, in some embodiments, are recombinant nucleic acid molecules comprising a sequence encoding a T cell receptor (TCR) fusion protein (TFP). The TFP can comprise a TCR subunit comprising at least a portion of a TCR extracellular domain. The TCR subunit can further comprise a transmembrane domain. The TCR subunit can further comprise an intracellular domain of TCR gamma, TCR delta, TCR alpha or TCR beta or an intracellular domain comprising a stimulatory domain from an intracellular signaling domain of CD3 epsilon, CD3 gamma, CD3 delta. The TFP can further comprise an antibody (e.g., a human, humanized, or murine antibody) comprising an antigen binding domain. The recombinant nucleic acid molecule can further comprise a sequence encoding a TCR constant domain, wherein the TCR constant domain is a TCR alpha constant domain, a TCR beta constant domain, a TCR alpha constant domain and a TCR beta constant domain, a TCR gamma constant domain, a TCR delta constant domain, or a TCR gamma constant domain and a TCR delta constant domain. The TCR subunit and the antibody can be operatively linked. The TFP can functionally incorporate into a TCR complex (e.g., an endogenous TCR complex) when expressed in a T cell. [0437] The constant domain can comprise a constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain. The constant domain can comprise a full-length constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain. The constant domain can comprise a fragment (e.g., functional fragment) of the full-length constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain. For example, the constant domain can comprise at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of the constant domain of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain. The sequence encoding the TCR constant domain can further encode the transmembrane domain and/or intracellular region of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain. The sequence encoding the TCR constant domain can encode a full-length constant region of a TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain. The constant region of a TCR chain can comprise a constant domain, a transmembrane domain, and an intracellular region. The constant region of a TCR chain can also exclude the transmembrane domain and the intracellular region of the TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain. [0438] The TCR alpha chain, a TCR beta chain, a TCR gamma chain or a TCR delta chain described herein can be derived from various species. The TCR chain can be a murine or
human TCR chain. For example, the constant domain can comprise a constant domain of a murine or human TCR alpha chain, TCR beta chain, TCR gamma chain or TCR delta chain. [0439] The constant domain can comprise truncations, additions, or substitutions of a sequence of a constant domain described herein. For example, the constant domain can comprise a truncated version of a constant domain described herein having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 19, SEQ ID NO: 20, SEQ ID NO: 21, SEQ ID NO: 22, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, SEQ ID NO:155, SEQ ID NO:207, SEQ ID NO:209, SEQ ID NO:243 or SEQ ID NO:265. For example, the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more additional amino acid residues of SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 19, SEQ ID NO: 20, SEQ ID NO: 21, SEQ ID NO: 22, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, SEQ ID NO:155, SEQ ID NO:207, SEQ ID NO:209, SEQ ID NO:243 or SEQ ID NO:265. For example, the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid substitutions of SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 19, SEQ ID NO: 20, SEQ ID NO: 21, SEQ ID NO: 22, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, SEQ ID NO:155, SEQ ID NO:207, SEQ ID NO:209, SEQ ID NO:243 or SEQ ID NO:265. The constant domain can comprise a sequence or fragment thereof of SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 19, SEQ ID NO: 20, SEQ ID NO: 21, SEQ ID NO: 22, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, SEQ ID NO:155, SEQ ID NO:207, SEQ ID NO:209, SEQ ID NO:243 or SEQ ID NO:265. The constant domain can comprise at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more modifications, mutations or deletions of the sequence of SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 19, SEQ ID NO: 20, SEQ ID NO: 21, SEQ ID NO: 22, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, SEQ ID NO:155, SEQ ID NO:207, SEQ ID NO:209, SEQ ID NO:243 or SEQ ID NO:265. The constant domain can comprise at most 20, 19, 18, 17, 16, 15, 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 modification, mutations or deletions of the sequence of SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 19, SEQ ID NO: 20, SEQ ID NO: 21, SEQ ID NO: 22, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, SEQ ID NO:155, SEQ ID NO:207, SEQ ID
NO:209, SEQ ID NO:243 or SEQ ID NO:265. The constant domain can comprise a sequence having a sequence identity of at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 99% or 100% to the sequence of SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 19, SEQ ID NO: 20, SEQ ID NO: 21, or SEQ ID NO: 22, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, SEQ ID NO:155, SEQ ID NO:207, SEQ ID NO:209, SEQ ID NO:243 or SEQ ID NO:265. [0440] The murine TCR alpha constant domain can comprise positions 2-137 of SEQ ID NO:146. The murine TCR alpha constant domain can comprise truncations, additions, or substitutions of a sequence of a constant domain described herein. For example, the constant domain can comprise a truncated version of a constant domain described herein having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of positions 2-137 of SEQ ID NO:146. For example, the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more additional amino acid residues of positions 2-137 of SEQ ID NO:146. For example, the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid substitutions of positions 2-137 of SEQ ID NO:146. The constant domain can comprise a sequence or fragment thereof of positions 2-137 of SEQ ID NO:146. The constant domain can comprise at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more modifications, mutations or deletions of the sequence of positions 2-137 of SEQ ID NO:146. The constant domain can comprise at most 20, 19, 18, 17, 16, 15, 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 modification, mutations or deletions of the sequence of positions 2-137 of SEQ ID NO:146. The constant domain can comprise a sequence having a sequence identity of at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 99% or 100% to the sequence of positions 2-137 of SEQ ID NO:146. [0441] The murine TCR beta constant domain can comprise positions 2-173 of SEQ ID NO:152. The murine TCR beta constant domain can comprise truncations, additions, or substitutions of a sequence of a constant domain described herein. For example, the constant domain can comprise a truncated version of a constant domain described herein having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of positions 2-173 of SEQ ID NO:152. For example, the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more additional amino acid residues of positions 2-173 of SEQ ID NO:152. For example, the constant domain can comprise a
sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid substitutions of positions 2-173 of SEQ ID NO:152. The constant domain can comprise a sequence or fragment thereof of positions 22-173 of SEQ ID NO:152. The constant domain can comprise at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more modifications, mutations or deletions of the sequence of positions 2-173 of SEQ ID NO:152. The constant domain can comprise at most 20, 19, 18, 17, 16, 15, 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 modification, mutations or deletions of the sequence of positions 2-173 of SEQ ID NO:152. The constant domain can comprise a sequence having a sequence identity of at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 99% or 100% to the sequence of positions 2-173 of SEQ ID NO:152. [0442] In some instances, the TCR constant domain is a TCR delta constant domain. The TCR delta constant domain can comprise SEQ ID NO:20, SEQ ID NO:22, SEQ ID NO:243 or SEQ ID NO:265, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modification. In some embodiments, the TCR delta constant domain can comprise SEQ ID NO:243. The TCR delta constant domain can comprise truncations, additions, or substitutions of a sequence of a constant domain described herein. For example, the constant domain can comprise a truncated version of a constant domain described herein having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of SEQ ID NO:243. For example, the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more additional amino acid residues of SEQ ID NO:243. For example, the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid substitutions of SEQ ID NO:243. The constant domain can comprise a sequence or fragment thereof of SEQ ID NO:243. The constant domain can comprise at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more modifications, mutations or deletions of the sequence of SEQ ID NO:243. The constant domain can comprise at most 20, 19, 18, 17, 16, 15, 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 modification, mutations or deletions of the sequence of SEQ ID NO:243. The constant domain can comprise a sequence having a sequence identity of at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 99% or 100% to the sequence of SEQ ID NO:243. [0443] The TCR delta constant domain can comprise SEQ ID NO:20, SEQ ID NO:22, SEQ ID NO:243 or SEQ ID NO:265, functional fragments thereof, or amino acid sequences thereof having at least one but not more than 20 modifications. In some cases, the sequence encoding a TCR delta constant domain further encodes a TCR delta variable domain, thereby
encoding a full TCR delta domain. The full TCR delta domain can be delta 2 or delta 1. The full TCR delta constant domain can comprise SEQ ID NO:256, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. [0444] The full TCR delta domain can comprise truncations, additions, or substitutions of a sequence of a constant domain described herein. For example, the delta domain can comprise a truncated version of a delta domain described herein having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of SEQ ID NO:256. For example, the delta domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more additional amino acid residues of SEQ ID NO:256. For example, the delta domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid substitutions of SEQ ID NO:256. The delta domain can comprise a sequence or fragment thereof of SEQ ID NO:256. The delta domain can comprise at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more modifications, mutations or deletions of the sequence of SEQ ID NO:256. The delta domain can comprise at most 20, 19, 18, 17, 16, 15, 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 modification, mutations or deletions of the sequence of SEQ ID NO:256. The delta domain can comprise a sequence having a sequence identity of at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 99% or 100% to the sequence of SEQ ID NO:256. [0445] The TCR gamma constant domain can comprise SEQ ID NO:21. The TCR gamma constant domain can comprise truncations, additions, or substitutions of a sequence of a constant domain described herein. For example, the constant domain can comprise a truncated version of a constant domain described herein having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of SEQ ID NO:21. For example, the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more additional amino acid residues of SEQ ID NO:21. For example, the constant domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid substitutions of SEQ ID NO:21. The constant domain can comprise a sequence or fragment thereof of SEQ ID NO:21. The constant domain can comprise at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more modifications, mutations or deletions of the sequence of SEQ ID NO:21. The constant domain can comprise at most 20, 19, 18, 17, 16, 15, 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 modification, mutations or deletions of the sequence of SEQ ID NO:21. The constant domain can comprise a sequence having a
sequence identity of at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 99% or 100% to the sequence of SEQ ID NO:243. [0446] The TCR gamma constant domain can comprise SEQ ID NO:21 or SEQ ID NO:155, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. In some cases, the sequence encoding the TCR gamma constant domain further encodes a TCR gamma variable domain, thereby encoding a full TCR gamma domain. The full TCR gamma domain can be gamma 9 or gamma 4. The full TCR gamma domain can comprise SEQ ID NO:255, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. [0447] The full TCR gamma domain can comprise truncations, additions, or substitutions of a sequence of a constant domain described herein. For example, the gamma domain can comprise a truncated version of a gamma domain described herein having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid residues of SEQ ID NO:255. For example, the gamma domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more additional amino acid residues of SEQ ID NO:255. For example, the gamma domain can comprise a sequence having at least about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 150 or more amino acid substitutions of SEQ ID NO:255. The gamma domain can comprise a sequence or fragment thereof of SEQ ID NO:255. The gamma domain can comprise at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more modifications, mutations or gamma of the sequence of SEQ ID NO:255. The gamma domain can comprise at most 20, 19, 18, 17, 16, 15, 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, 2 or 1 modification, mutations or deletions of the sequence of SEQ ID NO:255. The gamma domain can comprise a sequence having a sequence identity of at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 99% or 100% to the sequence of SEQ ID NO:255. [0448] TCR beta chain (Homo sapiens): VEDLNKVFPPEVAVFEPSEAEISHTQKATLVCLATGFFPDHVELSWWVNGKEVHSGV STDPQPLKEQPALNDSRYCLSSRLRVSATFWQNPRNHFRCQVQFYGLSENDEWTQD RAKPVTQIVSAEAWGRADCGFTSVSYQQGVLSATILYEILLGKATLYAVLVSALVLM AMVKRKDF (SEQ ID NO: 16). [0449] The murine TCR beta chain constant region canonical sequence is: EDLRNVTPPKVSLFEPSKAEIANKQKATLVCLARGFFPDHVELSWWVNGKEVHSGV STDPQAYKESNYSYCLSSRLRVSATFWHNPRNHFRCQVQFHGLSEEDKWPEGSPKP
VTQNISAEAWGRADCGITSASYQQGVLSATILYEILLGKATLYAVLVSTLVVMAMV KRKNS (SEQ ID NO:152). [0450] TCR alpha constant region (Mus musculus) (or [mm]TRAC(82-137)): ATYPSSDVPCDATLTEKSFETDMNLNFQNLSVMGLRILLLKVAGFNLLMTLRLWSS (SEQ ID NO: 17). [0451] The murine TCR alpha chain constant (mTRAC) region canonical sequence is: XIQNPEPAVYQLKDPRSQDSTLCLFTDFDSQINVPKTMESGTFITDKTVLDMKAMDS KSNGAIAWSNQTSFTCQDIFKETNATYPSSDVPCDATLTEKSFETDMNLNFQNLSVM GLRILLLKVAGFNLLMTLRLWSS (SEQ ID NO:146). [0452] TCR beta constant region (Mus musculus) (or [mm]TRBC1(123-173)): GRADCGITSASYQQGVLSATILYEILLGKATLYAVLVSTLVVMAMVKRKNS (SEQ ID NO: 18). [0453] The murine TCR beta chain constant region canonical sequence is: EDLRNVTPPKVSLFEPSKAEIANKQKATLVCLARGFFPDHVELSWWVNGKEVHSGV STDPQAYKESNYSYCLSSRLRVSATFWHNPRNHFRCQVQFHGLSEEDKWPEGSPKP VTQNISAEAWGRADCGITSASYQQGVLSATILYEILLGKATLYAVLVSTLVVMAMV KRKNS (SEQ ID NO:152). [0454] TCR beta chain (Homo sapiens): PVDSGVTQTPKHLITATGQRVTLRCSPRSGDLSVSWYQQSLDQGLQFLIQYYNGEER AKGNILERFSAQQFPDLHSELNLSSLELGDSALYFCASSPRTGLNTEAFFGQGTRLTV VEDLNKVFPPEVAVFEPSEAEISHTQKATLVCLATGFFPDHVELSWWVNGKEVHSGV STDPQPLKEQPALNDSRYCLSSRLRVSATFWQNPRNHFRCQVQFYGLSENDEWTQD RAKPVTQIVSAEAWGRADCGFTSVSYQQGVLSATILYEILLGKATLYAVLVSALVLM AMVKRKDF (SEQ ID NO: 19). [0455] TCR delta constant region version 1 (Homo sapiens): SQPHTKPSVFVMKNGTNVACLVKEFYPKDIRINLVSSKKITEFDPAIVISPSGKYNAV KLGKYEDSNSVTCSVQHDNKTVHSTDFEVKTDSTDHVKPKETENTKQPSKSCHKPK AIVHTEKVNMMSLTVLGLRMLFAKTVAVNFLLTAKLFF (SEQ ID NO: 20). [0456] TCR gamma constant region (Homo sapiens) (or [hs]TRGC(1-173)): DKQLDADVSPKPTIFLPSIAETKLQKAGTYLCLLEKFFPDVIKIHWQEKKSNTILGSQE GNTMKTNDTYMKFSWLTVPEKSLDKEHRCIVRHENNKNGVDQEIIFPPIKTDVITMD PKDNCSKDANDTLLLQLTNTSAYYMYLLLLLKSVVYFAIITCCLLRRTAFCCNGEKS (SEQ ID NO: 21). [0457] TCR delta constant region version 2 (Homo sapiens): SQPHTKPSVFVMKNGTNVACLVKEFYPKDIRINLVSSKKITEFDPAIVISPSGKYNAV
KLGKYEDSNSVTCSVQHDNKTVHSTDFEVKTDSTDHVKPKETENTKQPSKSCHKPK AIVHTEKVNMMSLTVLGLRMLFAKTVAVNFLLTAK (SEQ ID NO: 22). [0458] In some instances, the TCR constant domain is a TCR delta constant domain. The sequence encoding the TCR delta constant domain can further encode a second antigen binding domain or ligand binding domain that is operatively linked to the sequence encoding the TCR delta constant domain. The second antigen binding domain or ligand binding domain can be the same or different as the antigen binding domain or ligand binding domain of the TFP. [0459] In some instances, the TCR constant domain is a TCR gamma constant domain. The sequence encoding the TCR gamma constant domain can further encode a second antigen binding domain or ligand binding domain that is operatively linked to the sequence encoding the TCR gamma constant domain. The second antigen binding domain or ligand binding domain can be the same or different as the antigen binding domain or ligand binding domain of the TFP. [0460] In some instances, the recombinant nucleic acid comprises a sequence encoding a TCR gamma constant domain and a TCR delta constant domain. The TCR gamma constant domain can comprise SEQ ID NO:21 or SEQ ID NO:155, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. The sequence encoding the TCR gamma constant domain can further encode a TCR gamma variable domain, thereby encoding a full TCR gamma domain. The TCR gamma domain can be gamma 9 or gamma 4. The full TCR gamma domain comprises SEQ ID NO:255, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. The TCR delta constant domain can comprise SEQ ID NO:20, SEQ ID NO:22, SEQ ID NO:243 or SEQ ID NO:265, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. The sequence encoding the TCR delta constant domain can further encode a TCR delta variable domain, thereby encoding a full TCR delta domain. The TCR delta domain can be delta 2 or delta 1. The full TCR delta domain can comprise SEQ ID NO:256, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. [0461] In some instances, the TCR constant domain incorporates into a functional TCR complex when expressed in a T cell. In some instances, the TCR constant domain incorporates into a same functional TCR complex as the functional TCR complex that incorporates the TFP when expressed in a T cell. In some instances, the sequence encoding the TFP and the sequence encoding the TCR constant domain are contained within a same
nucleic acid molecule. In some instances, the sequence encoding the TFP and the sequence encoding the TCR constant domain are contained within different nucleic acid molecules. The sequence can further encode a cleavage site (e.g., a protease cleavage site) between the encoded TFP and the TCR constant domain. The cleavage site can be a protease cleavage site. The cleavage site can be a self-cleaving peptide such as a T2A, P2A, E2A or F2A cleavage site. The cleavage site can comprise a sequence of SEQ ID NO: 23. [0462] T2A cleavage site: EGRGSLLTCGDVEENPGP (SEQ ID NO: 23). [0463] The TCR subunit of the TFP and the constant domain can comprise a sequence derived from a same TCR chain or a different TCR chain. In some cases, the TCR subunit of the TFP and the constant domain are derived from different TCR chains. For example, the TCR subunit can comprise (1) at least a portion of a TCR extracellular domain, (2) a transmembrane domain, and (3) an intracellular domain, where the TCR extracellular domain, the transmembrane domain and the intracellular domain are derived from a TCR alpha chain, and the constant domain can comprise a constant domain of a TCR beta chain. For another example, the TCR subunit can comprise (1) at least a portion of a TCR extracellular domain, (2) a transmembrane domain, and (3) an intracellular domain, where the TCR extracellular domain, the transmembrane domain and the intracellular domain are derived from a TCR beta chain, and the constant domain can comprise a constant domain of a TCR alpha chain. For another example, the TCR subunit can comprise (1) at least a portion of a TCR extracellular domain, (2) a transmembrane domain, and (3) an intracellular domain, where the TCR extracellular domain, the transmembrane domain and the intracellular domain are derived from a TCR gamma chain, and the constant domain can comprise a constant domain of a TCR delta chain. For yet another example, the TCR subunit can comprise (1) at least a portion of a TCR extracellular domain, (2) a transmembrane domain, and (3) an intracellular domain, where the TCR extracellular domain, the transmembrane domain and the intracellular domain are derived from a TCR delta chain, and the constant domain can comprise a constant domain of a TCR gamma chain. [0464] In some instances, the TCR subunit and the antibody domain, the antigen domain or the binding ligand or fragment thereof are operatively linked by a linker sequence. In some instances, the linker sequence comprises (G4S)n, wherein n=1 to 4 (SEQ ID NO: 406). [0465] In some instances, the transmembrane domain is a TCR transmembrane domain from CD3 epsilon, CD3 gamma, CD3 delta, TCR gamma, TCR delta, TCR alpha or TCR beta. In some instances, the intracellular domain is derived from only CD3 epsilon, only CD3 gamma, only CD3 delta, only TCR gamma, only TCR delta, only TCR alpha or only TCR beta.
[0466] In some instances, the TCR subunit comprises (i) at least a portion of a TCR extracellular domain, (ii) a TCR transmembrane domain, and (iii) a TCR intracellular domain, wherein at least two or all of (i), (ii), and (iii) are from the same TCR subunit. [0467] In some instances, the TCR extracellular domain comprises an extracellular domain or portion thereof of a protein selected from the group consisting of a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, a CD3 delta TCR subunit, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. [0468] In some instances, the TCR subunit comprises a transmembrane domain comprising a transmembrane domain of a protein selected from the group consisting of a TCR alpha chain, a TCR beta chain, a TCR zeta chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, a CD3 delta TCR subunit, CD45, CD4, CD5, CD8, CD9, CD16, CD22, CD33, CD28, CD37, CD64, CD80, CD86, CD134, CD137, CD154, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. [0469] In some instances, the TCR subunit comprises a TCR intracellular domain of TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, or a fragment thereof. In some instances, the TCR subunit comprises an intracellular domain comprising a stimulatory domain of a protein selected from an intracellular signaling domain of CD3 epsilon, CD3 gamma or CD3 delta, or an amino acid sequence having at least one modification thereto. [0470] In some instances, the TCR subunit can comprise (i) at least a portion of a TCR extracellular domain, (ii) a TCR transmembrane domain, and (iii) a TCR intracellular domain of a TCR gamma chain or a TCR delta chain. The TCR extracellular domain can comprise the extracellular portion of a constant domain of a TCR gamma chain or a TCR delta chain, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. In some embodiments, the TCR subunit comprising (i) at least a portion of a TCR extracellular domain, (ii) a TCR transmembrane domain, and (iii) a TCR intracellular domain is or comprises a delta constant domain, or a fragment thereof, e.g., a delta constant domain described herein. The delta constant domain can have the sequence of SEQ ID NO:20, SEQ ID NO:22, SEQ ID NO:243 or SEQ ID NO:265, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. In some embodiments, the TCR subunit comprising (i) at least a portion of a TCR extracellular domain, (ii) a TCR transmembrane domain, and (iii) a TCR intracellular domain is or comprises a gamma constant domain, e.g., a gamma constant domain described herein. The gamma constant domain can have the sequence of SEQ ID NO:21 or SEQ ID
NO:155, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. The extracellular domain of the TFP may not comprise the variable domain of a gamma chain or a delta chain. [0471] The TCR subunit of the TFP can comprise the extracellular, transmembrane and intracellular domain of CD3 epsilon, CD3 gamma, or CD3 delta. In some embodiments, recombinant nucleic acid comprises a TFP comprising the extracellular, transmembrane and intracellular domain of CD3 epsilon, CD3 gamma, or CD3 delta and the constant domains of TCR beta and TCR alpha. In some embodiments, recombinant nucleic acid comprises a TFP comprising the extracellular, transmembrane and intracellular domain of CD3 epsilon and the constant domains of TCR gamma and TCR delta. In some embodiments, recombinant nucleic acid comprises a TFP comprising the extracellular, transmembrane and intracellular domain of CD3 epsilon and full length TCF gamma and full length TCR delta. In some embodiments, the TCR subunit of the TFP comprises CD3 epsilon. The TCR subunit of CD3 epsilon can comprise the sequence of SEQ ID NO:258 functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. [0472] In some instances, the TCR subunit comprising at least a portion of a murine TCR alpha or murine TCR beta extracellular domain and a murine TCR alpha or murine TCR beta transmembrane domain is or comprises a TCR alpha constant domain or a TCR beta constant domain. The TCR subunit can comprise an intracellular domain of murine TCR alpha or murine TCR beta. The TCR constant domain can be a TCR alpha constant domain, e.g., a TCR alpha constant domain described herein. The TCR alpha constant domain can comprise SEQ ID NO:17, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, or SEQ ID NO:207, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. The sequence encoding the TCR alpha constant domain can further encode a second antigen binding domain or ligand binding domain that is operatively linked to the sequence encoding the TCR alpha constant domain. The second antigen binding domain or ligand binding domain can be the same or different as the antigen binding domain or ligand binding domain of the TFP. The TCR alpha constant domain can comprise a murine TCR alpha constant domain. The murine TCR alpha constant domain can comprise amino acids 2-137 of the murine TCR alpha constant domain. The murine TCR alpha constant domain can comprise amino acids 2-137 of SEQ ID NO:146. The murine TCR alpha constant domain can comprise a sequence of SEQ ID NO:207. The murine TCR alpha constant domain can comprise amino acids 82-137 of SEQ ID NO:146. The murine TCR alpha constant domain comprises a sequence of SEQ ID NO:17. The TCR constant domain can be a TCR beta constant domain, e.g., a TCR beta constant domain described herein. The TCR beta
constant domain can comprise SEQ ID NO:18, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, or SEQ ID NO:209, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. The sequence encoding the TCR beta constant domain can further encode a second antigen binding domain or ligand binding domain that is operatively linked to the sequence encoding the TCR beta constant domain. The second antigen binding domain or ligand binding domain can be the same or different as the antigen binding domain or ligand binding domain of the TFP. TCR beta constant domain can comprise a murine TCR beta constant domain. The murine TCR beta constant domain can comprise amino acids 2-173 of the murine TCR beta constant domain. The murine TCR beta constant domain can comprise amino acids 2-173 of SEQ ID NO:152. The murine TCR beta constant domain can comprise SEQ ID NO:209. The TCR beta constant domain can comprise amino acids 123-173 of SEQ ID NO:152. The TCR beta constant domain can comprise SEQ ID NO:18 [0473] The recombinant nucleic acid can comprise sequence encoding a TCR alpha constant domain and a TCR beta constant domain. The TCR alpha constant domain can comprise SEQ ID NO:17, SEQ ID NO:142, SEQ ID NO:143, SEQ ID NO:146, or SEQ ID NO:207, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. The TCR beta constant domain can comprise SEQ ID NO:18, SEQ ID NO:148, SEQ ID NO:149, SEQ ID NO:152, or SEQ ID NO:209, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. The intracellular signaling domain can be CD3 epsilon, CD3 gamma, or CD3 delta. The intracellular signaling domain can be CD3 epsilon. [0474] The sequence encoding the TCR constant domain can comprise from 5’ to 3’, a first leader sequence, an antigen binding domain sequence, a linker, a TRAC gene sequence, a cleavable linker sequence, a second leader sequence, and a TRBC gene sequence. The sequence encoding the TCR constant domain can comprise, from 5’ to 3’, a first leader sequence, an antigen binding domain sequence, a linker, a TRAC gene sequence, a cleavable linker sequence, a second leader sequence, and a TRBC gene sequence. The sequence encoding the TCR constant domain can comprise, from 5’ to 3’, a first leader sequence, a TRAC gene sequence, a cleavable linker sequence, a second leader sequence, an antigen binding domain sequence, a linker, and a TRBC gene sequence. The sequence encoding the TCR constant domain can comprise, from 5’ to 3’, a first leader sequence, an antigen binding domain sequence, a linker, a TRAC gene sequence, a cleavable linker sequence, a second leader sequence, an antigen binding domain sequence, a linker, and a TRBC gene sequence. The sequence encoding the TCR constant domain can comprise, from 5’-3’, a first leader
sequence, a TRAC gene sequence, a first cleavable linker sequence, a second leader sequence, a TRBC gene sequence, a second cleavable linker sequence, a third leader sequence, an antigen binding domain sequence, a linker sequence, and a CD3 epsilon gene sequence. [0475] As described herein, the at least one but not more than 20 modifications thereto of a sequence described herein can comprise a modification of an amino acid that mediates cell signaling or a modification of an amino acid that is phosphorylated in response to a ligand binding to the TFP. [0476] In some instances, the TCR subunit comprises an intracellular domain comprising a stimulatory domain of a protein selected from a functional signaling domain of 4-1BB and/or a functional signaling domain of CD3 zeta, or an amino acid sequence having at least one modification thereto. [0477] In some instances, the recombinant nucleic acid further comprises a sequence encoding a costimulatory domain. In some instances, the costimulatory domain comprises a functional signaling domain of a protein selected from the group consisting of OX40, CD2, CD27, CD28, CD5, ICAM-1, LFA-1 (CD11a/CD18), ICOS (CD278), 4-1BB (CD137), IL- 15Ra, IL12R, IL18R, IL21R, GITR, CD30, CD40, BAFFR, HVEM, CD7, LIGHT, NKG2C, SLAMF7, NKp80, CD160, CD226, FcγRI, FcγRII, and FcγRIII, and amino acid sequences thereof having at least one but not more than 20 modifications thereto. [0478] In some instances, the TCR subunit comprises an immunoreceptor tyrosine-based activation motif (ITAM) of a TCR subunit that comprises an ITAM or portion thereof of a protein selected from the group consisting of CD3 zeta TCR subunit, CD3 epsilon TCR subunit, CD3 gamma TCR subunit, CD3 delta TCR subunit, TCR zeta chain, Fc epsilon receptor 1 chain, Fc epsilon receptor 2 chain, Fc gamma receptor 1 chain, Fc gamma receptor 2a chain, Fc gamma receptor 2b1 chain, Fc gamma receptor 2b2 chain, Fc gamma receptor 3a chain, Fc gamma receptor 3b chain, Fc beta receptor 1 chain, TYROBP (DAP12), CD5, CD16a, CD16b, CD22, CD23, CD32, CD64, CD79a, CD79b, CD89, CD278, CD66d, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications thereto. In some instances, the ITAM replaces an ITAM of CD3 gamma, CD3 delta, or CD3 epsilon. In some instances, the ITAM is selected from the group consisting of CD3 zeta TCR subunit, CD3 epsilon TCR subunit, CD3 gamma TCR subunit, and CD3 delta TCR subunit and replaces a different ITAM selected from the group consisting of CD3 zeta TCR subunit, CD3 epsilon TCR subunit, CD3 gamma TCR subunit, and CD3 delta TCR subunit.
[0479] In some instances, the TFP, the TCR gamma constant domain, the TCR delta constant domain, and any combination thereof is capable of functionally interacting with an endogenous TCR complex and/or at least one endogenous TCR polypeptide. In some instances, (a) the TCR constant domain is a TCR gamma constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of TCR delta, CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof; (b) the TCR constant domain is a TCR delta constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of TCR gamma, CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof; or (c) the TCR constant domain is a TCR gamma constant domain and a TCR delta constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof. [0480] In some instances, the at least one but not more than 20 modifications thereto comprise a modification of an amino acid that mediates cell signaling or a modification of an amino acid that is phosphorylated in response to a ligand binding to the TFP. [0481] The antibody or antigen binding domain can be an antibody fragment. The antibody or antigen binding domain can be murine, human or humanized. In some instances, the murine, human or humanized antibody is an antibody fragment. In some instances, the antibody fragment is a scFv, a single domain antibody domain, a VH domain or a VL domain. [0482] An antigen binding domain described herein can be selected from a group consisting of an anti-CD19 binding domain, an anti-B-cell maturation antigen (BCMA) binding domain, an anti-mesothelin (MSLN) binding domain, an anti-CD20 binding domain, an anti-CD70 binding domain, an anti-CD79b binding domain, an anti-PMSA binding domain, an anti- MUC16 binding domain, an anti-CD22 binding domain, an anti-PD-L1 binding domain, an anti BAFF receptor binding domain, an anti-Nectin-4 binding domain, an anti-TROP-2 binding domain, an anti-GPC3 binding domain, and anti-ROR-1 binding domain. [0483] In some instances, the nucleic acid is selected from the group consisting of a DNA and an RNA. In some instances, the nucleic acid is an mRNA. In some instances, the recombinant nucleic acid comprises a nucleic acid analog, wherein the nucleic acid analog is not in an encoding sequence of the recombinant nucleic acid. In some instances, the nucleic analog is selected from the group consisting of 2’-O-methyl, 2’-O-methoxyethyl (2’-O- MOE), 2’-O-aminopropyl, 2’-deoxy, T-deoxy-2’-fluoro, 2’-O-aminopropyl (2’-O-AP), 2'-O- dimethylaminoethyl (2’-O-DMAOE), 2’-O-dimethylaminopropyl (2’-O-DMAP), T-O- dimethylaminoethyloxyethyl (2’-O-DMAEOE), 2’-O-N-methylacetamido (2’-O-NMA)
modified, a locked nucleic acid (LNA), an ethylene nucleic acid (ENA), a peptide nucleic acid (PNA), a 1’,5’- anhydrohexitol nucleic acid (HNA), a morpholino, a methylphosphonate nucleotide, a thiolphosphonate nucleotide, and a 2’-fluoro N3-P5’-phosphoramidite. [0484] In some instances, the recombinant nucleic acid further comprises a leader sequence. In some instances, the recombinant nucleic acid further comprises a promoter sequence. In some instances, the recombinant nucleic acid further comprises a sequence encoding a poly(A) tail. In some instances, the recombinant nucleic acid further comprises a 3’UTR sequence. In some instances, the nucleic acid is an isolated nucleic acid or a non-naturally occurring nucleic acid. In some instances, the nucleic acid is an in vitro transcribed nucleic acid. [0485] In some instances, the recombinant nucleic acid further comprises a sequence encoding a TCR alpha transmembrane domain. In some instances, the recombinant nucleic acid further comprises a sequence encoding a TCR beta transmembrane domain. In some instances, the recombinant nucleic acid further comprises a sequence encoding a TCR alpha transmembrane domain and a sequence encoding a TCR beta transmembrane domain. [0486] In some instances, the TCR subunit comprises an immunoreceptor tyrosine-based activation motif (ITAM) of a TCR subunit that comprises an ITAM or portion thereof of a protein selected from the group consisting of CD3 zeta TCR subunit, CD3 epsilon TCR subunit, CD3 gamma TCR subunit, CD3 delta TCR subunit, TCR zeta chain, Fc epsilon receptor 1 chain, Fc epsilon receptor 2 chain, Fc gamma receptor 1 chain, Fc gamma receptor 2a chain, Fc gamma receptor 2b1 chain, Fc gamma receptor 2b2 chain, Fc gamma receptor 3a chain, Fc gamma receptor 3b chain, Fc beta receptor 1 chain, TYROBP (DAP12), CD5, CD16a, CD16b, CD22, CD23, CD32, CD64, CD79a, CD79b, CD89, CD278, CD66d, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications thereto. In some instances, the ITAM replaces an ITAM of CD3 gamma, CD3 delta, or CD3 epsilon. In some instances, the ITAM is selected from the group consisting of CD3 zeta TCR subunit, CD3 epsilon TCR subunit, CD3 gamma TCR subunit, and CD3 delta TCR subunit and replaces a different ITAM selected from the group consisting of CD3 zeta TCR subunit, CD3 epsilon TCR subunit, CD3 gamma TCR subunit, and CD3 delta TCR subunit. [0487] In some instances, the TFP, the TCR gamma constant domain, the TCR delta constant domain, the TCR alpha constant domain, the TCR beta constant domain, and any combination thereof is capable of functionally interacting with an endogenous TCR complex and/or at least one endogenous TCR polypeptide. In some instances, (a) the TCR constant domain is a TCR gamma constant domain and the TFP functionally integrates into a TCR
complex comprising an endogenous subunit of TCR beta, CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof; (b) the TCR constant domain is a TCR delta constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of TCR alpha, CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof; (c) the TCR constant domain is a TCR gamma constant domain and a TCR delta constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof; (d) the TCR constant domain is a TCR alpha constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of TCR beta, CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof; or (e) the TCR constant domain is a TCR beta constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of TCR alpha, CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof. [0488] In some instances, the at least one but not more than 20 modifications thereto comprise a modification of an amino acid that mediates cell signaling or a modification of an amino acid that is phosphorylated in response to a ligand binding to the TFP. [0489] In some instances, murine, human or humanized antibody comprising an antigen binding domain. The antigen binding domain described herein can be selected from a group consisting of an anti-CD19 binding domain, an anti-B-cell maturation antigen (BCMA) binding domain, an anti-mesothelin (MSLN) binding domain, an anti-CD20 binding domain, an anti-CD70 binding domain, an anti-CD79b binding domain, , an anti-PMSA binding domain, an anti-MUC16 binding domain, an anti-CD22 binding domain, an anti-PD-1 binding domain, an anti-PD-L1 binding domain, an anti IL13Rα2 binding domain, anti BAFF receptor binding domain, an anti-Nectin-4 binding domain, an anti-TROP-2 binding domain, an anti-GPC3 binding domain, and anti-ROR-1 binding domain. [0490] In some instances, the antigen binding domain comprises a ligand. In some instances, the ligand binds to the receptor of a cell. In some instances, the ligand binds to the polypeptide expressed on a surface of a cell. In some instances, the receptor or polypeptide expressed on a surface of a cell comprises a stress response receptor or polypeptide. In some instances, the receptor or polypeptide expressed on a surface of a cell is an MHC class I- related glycoprotein. In some instances, the MHC class I-related glycoprotein is selected from the group consisting of MICA, MICB, RAET1E, RAET1G, ULBP1, ULBP2, ULBP3, ULBP4 and combinations thereof. In some instances, the antigen domain comprises a monomer, a dimer, a trimer, a tetramer, a pentamer, a hexamer, a heptamer, an octomer, a nonamer, or a decamer. In some instances, the antigen domain comprises a monomer or a dimer of the ligand or fragment thereof. In some instances, the ligand or fragment thereof is a
monomer, a dimer, a trimer, a tetramer, a pentamer, a hexamer, a heptamer, an octomer, a nonamer, or a decamer. In some instances, the ligand or fragment thereof is a monomer or a dimer. In some instances, the antigen domain does not comprise an antibody or fragment thereof. In some instances, the antigen domain does not comprise a variable region. In some instances, the antigen domain does not comprise a CDR. In some instances, the ligand or fragment thereof is a Natural Killer Group 2D (NKG2D) ligand or a fragment thereof. [0491] In some instances, the TCR subunit and the antibody domain, the antigen domain or the binding ligand or fragment thereof are operatively linked by a linker sequence. In some instances, the linker sequence comprises (G4S)n, wherein n=1 to 4 (SEQ ID NO: 406). [0492] In some instances, the transmembrane domain is a TCR transmembrane domain from CD3 epsilon, CD3 gamma, CD3 delta, TCR alpha, TCR beta, TCR delta, or TCR gamma. In some instances, the intracellular domain is derived from only CD3 epsilon, only CD3 gamma, only CD3 delta, only TCR alpha, only TCR beta, only TCR delta, or only TCR gamma. [0493] In some instances, the TCR subunit comprises (i) at least a portion of a TCR extracellular domain, (ii) a TCR transmembrane domain, and (iii) a TCR intracellular domain, wherein at least two of (i), (ii), and (iii) are from the same TCR subunit. [0494] In some instances, the TCR extracellular domain comprises an extracellular domain or portion thereof of a protein selected from the group consisting of a TCR alpha chain, a TCR beta chain, a TCR delta chain, a TCR gamma chain, a CD3 epsilon TCR subunit, a CD3 gamma TCR subunit, a CD3 delta TCR subunit, functional fragments thereof, and amino acid sequences thereof having at least one but not more than 20 modifications. [0495] In some instances, the TFP, the TCR gamma constant domain, the TCR delta constant domain, the TCR alpha constant domain, the TCR beta constant domain, and any combination thereof is capable of functionally interacting with an endogenous TCR complex and/or at least one endogenous TCR polypeptide. In some instances, (a) the TCR constant domain is a TCR gamma constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of TCR beta, CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof; (b) the TCR constant domain is a TCR delta constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of TCR gamma, CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof; (c) the TCR constant domain is a TCR gamma constant domain and a TCR delta constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof; (d) the TCR constant domain is a TCR alpha constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of TCR beta, CD3 epsilon, CD3 gamma, CD3 delta, or a
combination thereof; or (e) the TCR constant domain is a TCR beta constant domain and the TFP functionally integrates into a TCR complex comprising an endogenous subunit of TCR alpha, CD3 epsilon, CD3 gamma, CD3 delta, or a combination thereof. [0496] In some instances, the at least one but not more than 20 modifications thereto comprise a modification of an amino acid that mediates cell signaling or a modification of an amino acid that is phosphorylated in response to a ligand binding to the TFP. [0497] In some instances, the murine, human or humanized antibody is an antibody fragment. In some instances, the antibody fragment is a scFv, a single domain antibody domain, a VH domain or a VL domain. An antigen binding domain described herein can be selected from a group consisting of an anti-CD19 binding domain, an anti-B-cell maturation antigen (BCMA) binding domain, an anti-mesothelin (MSLN) binding domain, an anti-CD20 binding domain, an anti-CD70 binding domain, an anti-CD79b binding domain, , an anti-PMSA binding domain, an anti-MUC16 binding domain, an anti-CD22 binding domain, an anti-PD-L1 binding domain, an anti-BAFF receptor binding domain, an anti-Nectin-4 binding domain, an anti-TROP-2 binding domain, an anti-GPC3 binding domain, and anti-ROR-1 binding domain. [0498] In some embodiments, a sequence encoding the antigen binding domain or ligand binding domain is operatively linked to a sequence encoding a delta constant domain. In some embodiments, the intracellular domain is an intracellular domain of TCR gamma. In some embodiments, a sequence encoding the antigen binding domain or ligand binding domain is operatively linked to a sequence encoding a gamma constant domain. In some embodiments, the intracellular domain is an intracellular domain of TCR delta. In some embodiments, a sequence encoding the antigen binding domain or ligand binding domain is operatively linked to both a sequence encoding a TCR delta constant domain or fragment thereof and a TCR gamma constant domain or fragment thereof. In some embodiments, the intracellular signaling domain is CD3 epsilon, CD3 gamma, or CD3 delta. In some embodiments, the intracellular signaling domain is CD3 epsilon. In some embodiments, the recombinant nucleic acid further comprises at least one leader sequence and at least one linker. In some embodiments, the recombinant nucleic acid further comprises a portion of a TCR alpha constant domain, a portion of a TCR beta domain, or both. In some embodiments, the sequence comprises, from 5’ to 3’, a first leader sequence, an antigen binding domain sequence, a linker, a TRDC gene sequence, a cleavable linker sequence, a second leader sequence, and a TRGC gene sequence. In some embodiments, the sequence comprises, from 5’-3’, a first leader sequence, a TRDC gene sequence, a cleavable linker sequence, a second leader sequence, an antigen binding domain sequence, a linker sequence, and a TRGC gene
sequence. In some embodiments, the sequence comprises, from 5’-3’, a first leader sequence, an antigen binding domain sequence, a first linker sequence, a TRDC gene sequence, a cleavable linker, a second leader sequence, a second antigen binding domain sequence, a second linker sequence, and a TRGC gene sequence. In some embodiments, the sequence comprises, from 5’-3’, a first leader sequence, a TRDC gene sequence, a first cleavable linker sequence, a second leader sequence, a TRGC gene sequence, a second cleavable linker sequence, a third leader sequence, an antigen binding domain sequence, a linker sequence, and a CD3 epsilon gene sequence. In some embodiments, the sequence comprises, from 5’- 3’, a first leader sequence, a first antigen binding domain sequence, a first linker sequence, a TRDC gene sequence or fragment thereof, a TRAC gene sequence or fragment thereof, a cleavable linker sequence, a second leader sequence, a second antigen binding domain sequence, a second linker sequence, a TRGC gene sequence or fragment thereof, and a TRBC gene sequence or fragment thereof. In some embodiments, the binding ligand is capable of binding an Fc domain of the antibody. In some embodiments, the binding ligand is capable of selectively binding an IgG1 antibody. In some embodiments, the binding ligand is capable of specifically binding an IgG4 antibody. In some embodiments, the antibody or fragment thereof binds to a cell surface antigen. In some embodiments, the antibody or fragment thereof is murine, human or humanized. In some embodiments, the antibody or fragment thereof binds to a cell surface antigen on the surface of a tumor cell. In some embodiments, the binding ligand comprises a monomer, a dimer, a trimer, a tetramer, a pentamer, a hexamer, a heptamer, an octomer, a nonamer, or a decamer. In some embodiments, the binding ligand does not comprise an antibody or fragment thereof. In some embodiments, the binding ligand comprises a CD16 polypeptide or fragment thereof. In some embodiments, the binding ligand comprises a CD16-binding polypeptide. In some embodiments, the binding ligand is human or humanized. In some embodiments, the recombinant nucleic acid further comprises a nucleic acid sequence encoding an antibody or fragment thereof capable of being bound by the binding ligand. In some embodiments, the antibody or fragment thereof is capable of being secreted from a cell. [0499] The present disclosure, in some cases, provides a recombinant nucleic acid comprising: a first nucleic acid sequence encoding a T cell receptor (TCR) fusion protein (TFP) wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain, wherein the TCR subunit and the antigen binding domain are operatively linked; and a second nucleic acid sequence encoding a polypeptide comprising a dominant negative transforming growth factor beta receptor II (TGFBr2) as described herein. In some
embodiments, the recombinant nucleic acid further comprises a sequence encoding a TCR constant domain, wherein the TCR constant domain is a TCR gamma constant domain, a TCR delta constant domain, or a TCR gamma constant domain and a TCR delta constant domain. In some embodiment, a modified T cell further comprises a functional disruption of an endogenous TCR. [0500] The present disclosure, in some cases, provides a recombinant nucleic acid comprising: a first nucleic acid sequence encoding a T cell receptor (TCR) fusion protein (TFP) wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain, wherein the TCR subunit and the antigen binding domain are operatively linked; and a second nucleic acid sequence encoding a switch polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof as described herein. In some embodiments, the recombinant nucleic acid further comprises a sequence encoding a TCR constant domain, wherein the TCR constant domain is a TCR gamma constant domain, a TCR delta constant domain, or a TCR gamma constant domain and a TCR delta constant domain. In some embodiment, a modified T cell further comprises a functional disruption of an endogenous TCR. [0501] The present disclosure, in some cases, provides a recombinant nucleic acid comprising (a) a sequence encoding a T cell receptor (TCR) fusion protein (TFP) comprising (i) a TCR subunit comprising (1) at least a portion of a TCR extracellular domain, (2) a transmembrane domain, and (3) an intracellular domain of TCR alpha, TCR beta, TCR gamma, or TCR delta or an intracellular domain comprising a stimulatory domain from an intracellular signaling domain of CD3 epsilon, CD3 gamma, or CD3 delta, and (ii) a binding ligand or a fragment thereof that is capable of binding to an antibody or fragment thereof; and (b) an antigen binding domain, wherein the TCR subunit and the antigen binding domain are operatively linked; and a second nucleic acid sequence encoding (i) a switch polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof as described herein; or (ii) a dominant negative TGFBr2 as described herein. In some embodiments, the recombinant nucleic acid further comprises a sequence encoding a TCR constant domain, wherein the TCR constant domain is a TCR gamma constant domain, a TCR delta constant domain, or a TCR gamma constant domain and a TCR delta constant domain. In some embodiment, a modified T cell further comprises a functional disruption of an endogenous TCR. [0502] In some embodiments, recombinant nucleic acid molecules described herein further comprise a leader sequence. In some embodiments, the recombinant nucleic acid molecule is
selected from the group consisting of a DNA and an RNA. In some embodiments, the recombinant nucleic acid molecule is an mRNA. In some embodiments, the recombinant nucleic acid molecule is a circRNA. In some embodiments, the recombinant nucleic acid molecule comprises a nucleic acid analog. In some embodiments, the nucleic acid analog is not in an encoding sequence of the recombinant nucleic acid. In some embodiments, the nucleic analog is selected from the group consisting of 2’-O-methyl, 2’-O-methoxyethyl (2’- O-MOE), 2’-O-aminopropyl, 2’-deoxy, T-deoxy-2’-fluoro, 2’-O-aminopropyl (2’-O-AP), 2'- O-dimethylaminoethyl (2’-O-DMAOE), 2’-O-dimethylaminopropyl (2’-O-DMAP), T-O- dimethylaminoethyloxyethyl (2’-O-DMAEOE), 2’-O-N-methylacetamido (2’-O-NMA) modified, a locked nucleic acid (LNA), an ethylene nucleic acid (ENA), a peptide nucleic acid (PNA), a 1’,5’- anhydrohexitol nucleic acid (HNA), a morpholino, a methylphosphonate nucleotide, a thiolphosphonate nucleotide, and a 2’-fluoro N3-P5’-phosphoramidite. In some embodiments, the recombinant nucleic acid molecule further comprises a leader sequence. In some embodiments, the recombinant nucleic acid molecule further comprises a promoter sequence. In some embodiments, the recombinant nucleic acid molecule further comprises a sequence encoding a poly(A) tail. In some embodiments, the recombinant nucleic acid molecule further comprises a 3’UTR sequence. In some embodiments, the recombinant nucleic acid molecule is an isolated nucleic acid or a non-naturally occurring nucleic acid. In some embodiments, the nucleic acid is an in vitro transcribed nucleic acid. Vectors [0503] Further disclosed herein, in some embodiments, are vectors comprising the recombinant nucleic acid molecules as described herein. In one aspect, the vector can be directly transduced into a cell, e.g., a T cell. In one aspect, the vector is a cloning or expression vector, e.g., a vector including, but not limited to, one or more plasmids (e.g., expression plasmids, cloning vectors, minicircles, minivectors, double minute chromosomes), retroviral and lentiviral vector constructs. In one aspect, the vector is capable of expressing the TFP construct as described herein and a switch polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, or a dominant negative TGFBr2 comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, in mammalian T cells. In one aspect, the mammalian T cell is a human T cell. [0504] In some instances, the vector is selected from the group consisting of a DNA, a RNA, a plasmid, a lentivirus vector, adenoviral vector, an adeno-associated viral vector (AAV), a Rous sarcoma viral (RSV) vector, or a retrovirus vector. In some instances, the vector is an
AAV6 vector. In some instances, the vector further comprises a promoter. In some instances, the vector is an in vitro transcribed vector. [0505] The nucleic acid sequences coding for the desired molecules can be obtained using recombinant methods known in the art, such as, for example by screening libraries from cells expressing the gene, by deriving the gene from a vector known to include the same, or by isolating directly from cells and tissues containing the same, using standard techniques. Alternatively, the gene of interest can be produced synthetically, rather than cloned. [0506] The present disclosure also provides vectors in which a DNA of the present disclosure is inserted. Vectors derived from retroviruses such as the lentivirus are suitable tools to achieve long-term gene transfer since they allow long-term, stable integration of a transgene and its propagation in daughter cells. Lentiviral vectors have the added advantage over vectors derived from onco-retroviruses such as murine leukemia viruses in that they can transduce non-proliferating cells, such as hepatocytes. They also have the added advantage of low immunogenicity. [0507] In another embodiment, the vector comprising the nucleic acid encoding the desired TFP and (i) a switch polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof or (ii) a dominant negative TGFBr2 of the present disclosure is an adenoviral vector (A5/35). In another embodiment, the expression of nucleic acids encoding TFPs as described herein and (i) a switch polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein or (ii) a dominant negative TGFBr2 comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein can be accomplished using of transposons such as sleeping beauty, crisper, CAS9, and zinc finger nucleases. See below June et al.2009 Nature Reviews Immunology 9.10: 704-716, is incorporated herein by reference. [0508] The expression constructs of the present disclosure may also be used for nucleic acid immunization and gene therapy, using standard gene delivery protocols. Methods for gene delivery are known in the art (see, e.g., U.S. Pat. Nos.5,399,346, 5,580,859, 5,589,466, incorporated by reference herein in their entireties). In another embodiment, the present disclosure provides a gene therapy vector. [0509] The nucleic acid can be cloned into a number of types of vectors. For example, the nucleic acid can be cloned into a vector including, but not limited to a plasmid, a phagemid, a phage derivative, an animal virus, and a cosmid. Vectors of particular interest include expression vectors, replication vectors, probe generation vectors, and sequencing vectors. [0510] Further, the expression vector may be provided to a cell in the form of a viral vector. Viral vector technology is well known in the art and is described, for example, in Sambrook
et al., 2012, Molecular Cloning: A Laboratory Manual, volumes 1-4, Cold Spring Harbor Press, NY), and in other virology and molecular biology manuals. Viruses, which are useful as vectors include, but are not limited to, retroviruses, adenoviruses, adeno-associated viruses, herpes viruses, and lentiviruses. In general, a suitable vector contains an origin of replication functional in at least one organism, a promoter sequence, convenient restriction endonuclease sites, and one or more selectable markers, (e.g., WO 01/96584; WO 01/29058; and U.S. Pat. No.6,326,193). [0511] A number of virally based systems have been developed for gene transfer into mammalian cells. For example, retroviruses provide a convenient platform for gene delivery systems. A selected gene can be inserted into a vector and packaged in retroviral particles using techniques known in the art. The recombinant virus can then be isolated and delivered to cells of the subject either in vivo or ex vivo. A number of retroviral systems are known in the art. In some embodiments, adenovirus vectors are used. A number of adenovirus vectors are known in the art. In one embodiment, lentivirus vectors are used. [0512] Additional promoter elements, e.g., enhancers, regulate the frequency of transcriptional initiation. Typically, these are located in the region 30-110 bp upstream of the start site, although a number of promoters have been shown to contain functional elements downstream of the start site as well. The spacing between promoter elements frequently is flexible, so that promoter function is preserved when elements are inverted or moved relative to one another. In the thymidine kinase (tk) promoter, the spacing between promoter elements can be increased to 50 bp apart before activity begins to decline. Depending on the promoter, it appears that individual elements can function either cooperatively or independently to activate transcription. [0513] An example of a promoter that is capable of expressing a TFP transgene as described herein and a (i) switch polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein or (ii) a dominant negative TGFBr2 comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein in a mammalian T cell is the EF1a promoter. The native EF1a promoter drives expression of the alpha subunit of the elongation factor-1 complex, which is responsible for the enzymatic delivery of aminoacyl tRNAs to the ribosome. The EF1a promoter has been extensively used in mammalian expression plasmids and has been shown to be effective in driving TFP and expression from transgenes cloned into a lentiviral vector (see, e.g., Milone et al., Mol. Ther. 17(8): 1453-1464 (2009)). Another example of a promoter is the immediate early cytomegalovirus (CMV) promoter sequence. This promoter sequence is a strong constitutive promoter sequence capable of driving high levels of expression of any polynucleotide
sequence operatively linked thereto. However, other constitutive promoter sequences may also be used, including, but not limited to the simian virus 40 (SV40) early promoter, mouse mammary tumor virus (MMTV), human immunodeficiency virus (HIV) long terminal repeat (LTR) promoter, MoMuLV promoter, an avian leukemia virus promoter, an Epstein-Barr virus immediate early promoter, a Rous sarcoma virus promoter, as well as human gene promoters such as, but not limited to, the actin promoter, the myosin promoter, the elongation factor-1a promoter, the hemoglobin promoter, and the creatine kinase promoter. Further, the present disclosure should not be limited to the use of constitutive promoters. Inducible promoters are also contemplated as part of the present disclosure. The use of an inducible promoter provides a molecular switch capable of turning on expression of the polynucleotide sequence which it is operatively linked when such expression is desired or turning off the expression when expression is not desired. Examples of inducible promoters include, but are not limited to a metallothionine promoter, a glucocorticoid promoter, a progesterone promoter, and a tetracycline-regulated promoter. [0514] In order to assess the expression of a TFP polypeptide as described herein and a switch polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, or a dominant negative TGFBr2 comprising a TGFBr2 extracellular domain or a functional fragment thereof, the expression vector to be introduced into a cell can also contain either a selectable marker gene or a reporter gene or both to facilitate identification and selection of expressing cells from the population of cells sought to be transfected or infected through viral vectors. In other aspects, the selectable marker may be carried on a separate piece of DNA and used in a co-transfection procedure. Both selectable markers and reporter genes may be flanked with appropriate regulatory sequences to enable expression in the host cells. Useful selectable markers include, for example, antibiotic-resistance genes, such as neo and the like. [0515] Reporter genes are used for identifying potentially transfected cells and for evaluating the functionality of regulatory sequences. In general, a reporter gene is a gene that is not present in or expressed by the recipient organism or tissue and that encodes a polypeptide whose expression is manifested by some easily detectable property, e.g., enzymatic activity. Expression of the reporter gene is assayed at a suitable time after the DNA has been introduced into the recipient cells. Suitable reporter genes may include genes encoding luciferase, beta-galactosidase, chloramphenicol acetyl transferase, secreted alkaline phosphatase, or the green fluorescent protein gene (e.g., Ui-Tei et al., 2000 FEBS Letters 479: 79-82). Suitable expression systems are well known and may be prepared using known techniques or obtained commercially. In general, the construct with the minimal 5’ flanking
region showing the highest level of expression of reporter gene is identified as the promoter. Such promoter regions may be linked to a reporter gene and used to evaluate agents for the ability to modulate promoter-driven transcription. [0516] Methods of introducing and expressing genes into a cell are known in the art. In the context of an expression vector, the vector can be readily introduced into a host cell, e.g., mammalian, bacterial, yeast, or insect cell by any method in the art. For example, the expression vector can be transferred into a host cell by physical, chemical, or biological means. [0517] Physical methods for introducing a polynucleotide into a host cell include calcium phosphate precipitation, lipofection, particle bombardment, microinjection, electroporation, and the like. Methods for producing cells comprising vectors and/or exogenous nucleic acids are well-known in the art. See, for example, Sambrook et al., 2012, Molecular Cloning: A Laboratory Manual, volumes 1-4, Cold Spring Harbor Press, NY). A preferred method for the introduction of a polynucleotide into a host cell is calcium phosphate transfection [0518] Biological methods for introducing a polynucleotide of interest into a host cell include the use of DNA and RNA vectors. Viral vectors, and especially retroviral vectors, have become the most widely used method for inserting genes into mammalian, e.g., human cells. Other viral vectors can be derived from lentivirus, poxviruses, herpes simplex virus I, adenoviruses and adeno-associated viruses, and the like (see, e.g., U.S. Pat. Nos.5,350,674 and 5,585,362. [0519] Chemical means for introducing a polynucleotide into a host cell include colloidal dispersion systems, such as macromolecule complexes, nanocapsules, microspheres, beads, and lipid-based systems including oil-in-water emulsions, micelles, mixed micelles, and liposomes. An exemplary colloidal system for use as a delivery vehicle in vitro and in vivo is a liposome (e.g., an artificial membrane vesicle). Other methods of state-of-the-art targeted delivery of nucleic acids are available, such as delivery of polynucleotides with targeted nanoparticles or other suitable sub-micron sized delivery system. [0520] In the case where a non-viral delivery system is utilized, an exemplary delivery vehicle is a liposome. The use of lipid formulations is contemplated for the introduction of the nucleic acids into a host cell (in vitro, ex vivo or in vivo). In another aspect, the nucleic acid may be associated with a lipid. The nucleic acid associated with a lipid may be encapsulated in the aqueous interior of a liposome, interspersed within the lipid bilayer of a liposome, attached to a liposome via a linking molecule that is associated with both the liposome and the oligonucleotide, entrapped in a liposome, complexed with a liposome, dispersed in a solution containing a lipid, mixed with a lipid, combined with a lipid,
contained as a suspension in a lipid, contained or complexed with a micelle, or otherwise associated with a lipid. Lipid, lipid/DNA or lipid/expression vector associated compositions are not limited to any particular structure in solution. For example, they may be present in a bilayer structure, as micelles, or with a “collapsed” structure. They may also simply be interspersed in a solution, possibly forming aggregates that are not uniform in size or shape. Lipids are fatty substances which may be naturally occurring or synthetic lipids. For example, lipids include the fatty droplets that naturally occur in the cytoplasm as well as the class of compounds which contain long-chain aliphatic hydrocarbons and their derivatives, such as fatty acids, alcohols, amines, amino alcohols, and aldehydes. [0521] Lipids suitable for use can be obtained from commercial sources. For example, dimyristyl phosphatidylcholine (“DMPC”) can be obtained from Sigma, St. Louis, Mo.; dicetyl phosphate (“DCP”) can be obtained from K & K Laboratories (Plainview, N.Y.); cholesterol (“Choi”) can be obtained from Calbiochem-Behring; dimyristyl phosphatidylglycerol (“DMPG”) and other lipids may be obtained from Avanti Polar Lipids, Inc. (Birmingham, Ala.). Stock solutions of lipids in chloroform or chloroform/methanol can be stored at about -20 °C. Chloroform is used as the only solvent since it is more readily evaporated than methanol. “Liposome” is a generic term encompassing a variety of single and multilamellar lipid vehicles formed by the generation of enclosed lipid bilayers or aggregates. Liposomes can be characterized as having vesicular structures with a phospholipid bilayer membrane and an inner aqueous medium. Multilamellar liposomes have multiple lipid layers separated by aqueous medium. They form spontaneously when phospholipids are suspended in an excess of aqueous solution. The lipid components undergo self-rearrangement before the formation of closed structures and entrap water and dissolved solutes between the lipid bilayers (Ghosh et al., 1991 Glycobiology 5: 505-10). However, compositions that have different structures in solution than the normal vesicular structure are also encompassed. For example, the lipids may assume a micellar structure or merely exist as nonuniform aggregates of lipid molecules. Also contemplated are lipofectamine-nucleic acid complexes. [0522] Regardless of the method used to introduce exogenous nucleic acids into a host cell or otherwise expose a cell to the inhibitor of the present disclosure, in order to confirm the presence of the recombinant DNA sequence in the host cell, a variety of assays may be performed. Such assays include, for example, “molecular biological” assays well known to those of skill in the art, such as Southern and Northern blotting, RT-PCR and PCR; “biochemical” assays, such as detecting the presence or absence of a particular peptide, e.g.,
by immunological means (ELISAs and western blots) or by assays described herein to identify agents falling within the scope of the present disclosure. [0523] The present disclosure further provides a vector comprising a nucleic acid molecule encoding a TFP as described herein and a switch polypeptide or a dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In one aspect, a vector encoding a TFP as described herein and a switch or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein can be directly transduced into a cell, e.g., a T cell. In one aspect, the vector is a cloning or expression vector, e.g., a vector including, but not limited to, one or more plasmids (e.g., expression plasmids, cloning vectors, minicircles, minivectors, double minute chromosomes), retroviral and lentiviral vector constructs. In one aspect, the vector is capable of expressing the TFP construct as described herein and a switch polypeptide or a dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein in mammalian T cells. In one aspect, the mammalian T cell is a human T cell. [0524] Expression vectors are provided that include: a promoter (e.g., an EF1a promoter), a signal sequence to enable secretion, a polyadenylation signal and transcription terminator (Bovine Growth Hormone (BGH) gene), an element allowing episomal replication and replication in prokaryotes (e.g., SV40 origin and ColE1 or others known in the art) and elements to allow selection (ampicillin resistance gene and zeocin marker). [0525] The TFP-encoding nucleic acid construct as described herein with or without a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein can be cloned into a lentiviral expression vector and expression validated based on the quantity and quality of the effector T cell response of transduced T cells in response to MSLN+ target cells. Effector T cell responses include, but are not limited to, cellular expansion, proliferation, doubling, cytokine production and target cell lysis or cytolytic activity (i.e., degranulation). Recombinant RNAs [0526] Disclosed herein are methods for producing in vitro transcribed RNA encoding TFPs as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. The present disclosure also includes a TFP as described herein encoding RNA construct and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein that can be directly transfected into a cell. A method for generating mRNA for use in transfection can involve in vitro
transcription (IVT) of a template with specially designed primers, followed by polyA addition, to produce a construct containing 3’ and 5’ untranslated sequence (“UTR”), a 5’ cap and/or Internal Ribosome Entry Site (IRES), the nucleic acid to be expressed, and a polyA tail, typically 50-2000 bases in length (SEQ ID NO: 407). RNA so produced can efficiently transfect different kinds of cells. In one aspect, the template includes sequences for the TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein as described herein. [0527] In one aspect, the anti-TAA TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein is encoded by a messenger RNA (mRNA). In one aspect, the mRNA encoding the anti-TAA TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein is introduced into a T cell for production of a T cell expressing the TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In one embodiment, the in vitro transcribed RNA encoding a TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein can be introduced to a cell as a form of transient transfection. The RNA is produced by in vitro transcription using a polymerase chain reaction (PCR)-generated template. DNA of interest from any source can be directly converted by PCR into a template for in vitro mRNA synthesis using appropriate primers and RNA polymerase. The source of the DNA can be, for example, genomic DNA, plasmid DNA, phage DNA, cDNA, synthetic DNA sequence or any other appropriate source of DNA. The desired template for in vitro transcription is a TFP and/or a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof of the present disclosure. In one embodiment, the DNA to be used for PCR contains an open reading frame. The DNA can be from a naturally occurring DNA sequence from the genome of an organism. In one embodiment, the nucleic acid can include some or all of the 5’ and/or 3’ untranslated regions (UTRs). The nucleic acid can include exons and introns. In one embodiment, the DNA to be used for PCR is a human nucleic acid sequence. In another embodiment, the DNA to be used for PCR is a human nucleic acid sequence including the 5’ and 3’ UTRs. The DNA can alternatively be an artificial DNA sequence that is not normally expressed in a naturally occurring organism. An exemplary artificial DNA sequence is one that contains portions of
genes that are ligated together to form an open reading frame that encodes a fusion protein. The portions of DNA that are ligated together can be from a single organism or from more than one organism. [0528] PCR can be used to generate a template for in vitro transcription of mRNA which is used for transfection. Methods for performing PCR are well known in the art. Primers for use in PCR are designed to have regions that are substantially complementary to regions of the DNA to be used as a template for the PCR. “Substantially complementary,” as used herein, refers to sequences of nucleotides where a majority or all of the bases in the primer sequence are complementary, or one or more bases are non-complementary, or mismatched. Substantially complementary sequences are able to anneal or hybridize with the intended DNA target under annealing conditions used for PCR. The primers can be designed to be substantially complementary to any portion of the DNA template. For example, the primers can be designed to amplify the portion of a nucleic acid that is normally transcribed in cells (the open reading frame), including 5’ and 3’ UTRs. The primers can also be designed to amplify a portion of a nucleic acid that encodes a particular domain of interest. In one embodiment, the primers are designed to amplify the coding region of a human cDNA, including all or portions of the 5’ and 3’ UTRs. Primers useful for PCR can be generated by synthetic methods that are well known in the art. “Forward primers” are primers that contain a region of nucleotides that are substantially complementary to nucleotides on the DNA template that are upstream of the DNA sequence that is to be amplified. “Upstream” is used herein to refer to a location 5, to the DNA sequence to be amplified relative to the coding strand. “Reverse primers” are primers that contain a region of nucleotides that are substantially complementary to a double-stranded DNA template that are downstream of the DNA sequence that is to be amplified. “Downstream” is used herein to refer to a location 3’ to the DNA sequence to be amplified relative to the coding strand. [0529] Any DNA polymerase useful for PCR can be used in the methods as described herein. The reagents and polymerase are commercially available from a number of sources. [0530] Chemical structures with the ability to promote stability and/or translation efficiency may also be used. The RNA preferably has 5’ and 3’ UTRs. In one embodiment, the 5’ UTR is between one and 3000 nucleotides in length. The length of 5’ and 3’ UTR sequences to be added to the coding region can be altered by different methods, including, but not limited to, designing primers for PCR that anneal to different regions of the UTRs. Using this approach, one of ordinary skill in the art can modify the 5’ and 3’ UTR lengths required to achieve optimal translation efficiency following transfection of the transcribed RNA.
[0531] The 5’ and 3’ UTRs can be the naturally occurring, endogenous 5’ and 3’ UTRs for the nucleic acid of interest. Alternatively, UTR sequences that are not endogenous to the nucleic acid of interest can be added by incorporating the UTR sequences into the forward and reverse primers or by any other modifications of the template. The use of UTR sequences that are not endogenous to the nucleic acid of interest can be useful for modifying the stability and/or translation efficiency of the RNA. For example, it is known that AU-rich elements in 3’UTR sequences can decrease the stability of mRNA. Therefore, 3’ UTRs can be selected or designed to increase the stability of the transcribed RNA based on properties of UTRs that are well known in the art. [0532] In one embodiment, the 5’ UTR can contain the Kozak sequence of the endogenous nucleic acid. Alternatively, when a 5’ UTR that is not endogenous to the nucleic acid of interest is being added by PCR as described above, a consensus Kozak sequence can be redesigned by adding the 5’ UTR sequence. Kozak sequences can increase the efficiency of translation of some RNA transcripts but do not appear to be required for all RNAs to enable efficient translation. The requirement for Kozak sequences for many mRNAs is known in the art. In other embodiments the 5’ UTR can be 5’UTR of an RNA virus whose RNA genome is stable in cells. In other embodiments various nucleotide analogues can be used in the 3’ or 5’ UTR to impede exonuclease degradation of the mRNA. [0533] To enable synthesis of RNA from a DNA template without the need for gene cloning, a promoter of transcription should be attached to the DNA template upstream of the sequence to be transcribed. When a sequence that functions as a promoter for an RNA polymerase is added to the 5’ end of the forward primer, the RNA polymerase promoter becomes incorporated into the PCR product upstream of the open reading frame that is to be transcribed. In one preferred embodiment, the promoter is a T7 polymerase promoter, as described elsewhere herein. Other useful promoters include, but are not limited to, T3 and SP6 RNA polymerase promoters. Consensus nucleotide sequences for T7, T3 and SP6 promoters are known in the art. [0534] In some embodiments, the mRNA has both a cap on the 5’ end and a 3’ poly(A) tail which determine ribosome binding, initiation of translation and stability mRNA in the cell. On a circular DNA template, for instance, plasmid DNA, RNA polymerase produces a long concatemeric product which is not suitable for expression in eukaryotic cells. The transcription of plasmid DNA linearized at the end of the 3’ UTR results in normal sized mRNA which is not effective in eukaryotic transfection even if it is polyadenylated after transcription.
[0535] On a linear DNA template, phage T7 RNA polymerase can extend the 3’ end of the transcript beyond the last base of the template (Schenborn and Mierendorf, Nuc Acids Res., 13:6223-36 (1985); Nacheva and Berzal-Herranz, Eur. J. Biochem., 270:1485-65 (2003). [0536] The conventional method of integration of polyA/T stretches into a DNA template is molecular cloning. However, polyA/T sequence integrated into plasmid DNA can cause plasmid instability, which is why plasmid DNA templates obtained from bacterial cells are often highly contaminated with deletions and other aberrations. This makes cloning procedures not only laborious and time consuming but often not reliable. That is why a method which allows construction of DNA templates with polyA/T 3’ stretch without cloning highly desirable. [0537] The polyA/T segment of the transcriptional DNA template can be produced during PCR by using a reverse primer containing a poly-T tail, such as 100 T tail (SEQ ID NO: 408) (size can be 50-5000 T (SEQ ID NO: 409)), or after PCR by any other method, including, but not limited to, DNA ligation or in vitro recombination. Poly(A) tails also provide stability to RNAs and reduce their degradation. Generally, the length of a poly(A) tail positively correlates with the stability of the transcribed RNA. In one embodiment, the poly(A) tail is between 100 and 5000 adenosines (SEQ ID NO: 410). [0538] Poly(A) tails of RNAs can be further extended following in vitro transcription with the use of a poly(A) polymerase, such as E. coli polyA polymerase (E-PAP). In one embodiment, increasing the length of a poly(A) tail from 100 nucleotides to between 300 and 400 nucleotides (SEQ ID NO: 411) results in about a two-fold increase in the translation efficiency of the RNA. Additionally, the attachment of different chemical groups to the 3’ end can increase mRNA stability. Such attachment can contain modified/artificial nucleotides, aptamers and other compounds. For example, ATP analogs can be incorporated into the poly(A) tail using poly(A) polymerase. ATP analogs can further increase the stability of the RNA. [0539] 5’ caps can also provide stability to RNA molecules. In some embodiments, RNAs produced by the methods as described herein include a 5’ cap. The 5’ cap is provided using techniques known in the art and described herein (Cougot, et al., Trends in Biochem. Sci., 29:436-444 (2001); Stepinski, et al., RNA, 7:1468-95 (2001); Elango, et al., Biochim. Biophys. Res. Commun., 330:958-966 (2005)). [0540] The RNAs produced by the methods as described herein can also contain an internal ribosome entry site (IRES) sequence. The IRES sequence may be any viral, chromosomal or artificially designed sequence which initiates cap-independent ribosome binding to mRNA and facilitates the initiation of translation. Any solutes suitable for cell electroporation, which
can contain factors facilitating cellular permeability and viability such as sugars, peptides, lipids, proteins, antioxidants, and surfactants can be included. [0541] RNA can be introduced into target cells using any of a number of different methods, for instance, commercially available methods which include, but are not limited to, electroporation (Amaxa Nucleofector®-II (Amaxa Biosystems, Cologne, Germany)), ECM 830 (BTX) (Harvard Instruments, Boston, Mass.) or the Gene Pulser® II (BioRad, Denver, Colo.), Multiporator® (Eppendorf, Hamburg Germany), cationic liposome mediated transfection using lipofection, polymer encapsulation, peptide mediated transfection, or biolistic particle delivery systems such as “gene guns” (see, for example, Nishikawa, et al. Hum Gene Ther., 12(8):861-70 (2001). Modified Cells [0542] Disclosed herein, in some embodiments, are cells comprising the recombinant nucleic acid as described herein, the polypeptide as described herein, or the vectors as described herein. Disclosed herein, in some embodiments, are cells comprising the recombinant nucleic acid as described herein, the polypeptide as described herein, or the vectors as described herein, wherein cells comprise the sequence encoding a TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In some embodiments, the cell is a T cell. In some embodiments, the T cell is a human T cell. In some embodiments, the T cell is a CD8+ or CD4+ T cell. In some embodiments, the T cell is a human αβ T cell. In some embodiments, the T cell is a human γδ T cell. In some embodiments, the cell is a human NKT cell. In some embodiments, the cell is an allogeneic cell or an autologous cell. In some embodiments, the T cell is modified to comprise a functional disruption of the TCR. In some embodiments, the modified T cells are γδ T cells and do not comprise a functional disruption of an endogenous TCR. In some embodiments, the γδ T cells are V 1+ V 2- T cells. In some embodiments, the γδ T cells are V 1- V 2+ T cells. In some embodiments, the γδ T cells are V 1- V 2- T cells. [0543] The present disclosure provides genetically-modified immune cells and populations thereof and methods for producing the same. In some embodiments, the genetically-modified immune cells of the presently disclosed compositions and methods are human immune cells. In some embodiments, the immune cells are T cells, or cells derived therefrom. In other embodiments, the immune cells are natural killer (NK) cells, or cells derived therefrom. In still other embodiments, the immune cells are B cells, or cells derived therefrom. In yet other embodiments, the immune cells are monocyte or macrophage cells or cells derived therefrom.
[0544] Disclosed herein, in some embodiments, are cells comprising the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, wherein the cells may have enhanced survival rate, enhanced effector function, and/or enhanced cytotoxicity compared to cells that do not comprise the sequence encoding TFP as described herein, and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In some embodiments, the cell has enhanced survival rate compared to a cell that does not have a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In some embodiments, the cell has enhanced effector function compared to a cell that does not have a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In some embodiments, the cell has enhanced cytotoxicity compared to a cell that does not have a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. [0545] Disclosed herein, in some embodiments, are cells comprising the sequence encoding TFP as described herein, and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof, wherein the cells as described herein may have increased longevity compared to cells that do not comprise the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In some embodiments, the longevity of the cell is increased compared to a cell that does not comprise a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. [0546] Disclosed herein, in some embodiments, are cells comprising the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, wherein the cells may have increased persistence compared to cells that do not comprise the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In some embodiments, the persistence of the cell is increased compared to a cell that does not comprise a nucleic acid sequence encoding a
switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof. [0547] Disclosed herein, in some embodiments, are cells comprising the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, wherein the cells may have increased cytotoxicity compared to cells that do not comprise the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In some embodiments, the cytotoxicity of the cell is increased compared to a cell that does not comprise a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. [0548] Disclosed herein, in some embodiments, are cells comprising the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, wherein the cells may have increased cytokine production compared to cells that do not comprise the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In some embodiments, the cytokine production of the cell is increased compared to a cell that does not comprise a nucleic acid sequence encoding herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. [0549] Disclosed herein, in some embodiments, are cells comprising the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, wherein the cells have increased ability to resist, blunt, and/or overcome TGFβ- mediated suppression of T cell function. For example, in some embodiments, the cells have increased ability to resist, blunt, and/or overcome TGFβ-mediated suppression of cytokine production (e.g., IFNγ, IL-2, GM-CSF, and/or TNFα) and/or TGFβ-mediated suppression of cytotoxicity and/or TGFβ-mediated exhaustion. In some embodiments, the ability to resist, blunt, and/or overcome TGFβ-mediated effects is as compared to a cell that does not comprise a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, for example, is as compared to a cell that comprises the same TFP as the
cell comprising the TGFBr2 switch polypeptide or TGFBr2 dominant negative polypeptide but that lacks a TGFBr2 switch polypeptide and TGFBr2 dominant negative polypeptide. [0550] In some embodiments, cells as described herein retain naïve and/or central memory phenotypes. In some embodiments, cells as described herein have not differentiated into terminal effector cells. In some embodiments, the cells as described herein expand in the presence of TGFβ. [0551] Disclosed herein, in some embodiments, is a population of cells comprising any of the cell described herein. Disclosed herein, in some embodiments, is a population of cells comprising any of the cell described herein, wherein the population of cells has an increased proportion of cells having a central memory phenotype relative to a population of cells that do not comprise the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In some embodiments, the population of cells has an increased proportion of cells having a central memory phenotype relative to a population of cells that do not comprise a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. [0552] Disclosed herein, in some embodiments, is population of cells comprising any of the cell described herein, wherein the population of cells has an increased proportion of cells having a naïve phenotype relative to a population of cells that do not comprise the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In some embodiments, the population of cells has an increased proportion of cells having a naïve phenotype relative to a population of cells that do not comprise a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. [0553] Disclosed herein, in some embodiments, is population of cells comprising any of the cell described herein, wherein the population of cells has a reduced proportion of cells having a terminal effector phenotype relative to a population of cells that do not comprise the sequence encoding TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In some embodiments, the population of cells has a reduced proportion of cells having a terminal effector phenotype relative to a population of cells that do not comprise a nucleic acid sequence encoding a switch polypeptide or dominant negative
polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. [0554] In some embodiments, the population of cells comprising any of the cells described herein has a similar phenotype (e.g., CD4:CD8 ratio, proportion of cells having a central memory phenotype, proportion of cells having a naïve phenotype, and/or proportion of cells having a terminal effector phenotype) as a population of cells that do not comprise a nucleic acid encoding a TGFBr2 switch polypeptide or TGFBR2 dominant negative polypeptide, but exhibits increased survival rate, effector function, cytotoxicity, longevity, persistence, and/or cytokine production, compared to a population of cells that do not comprise a nucleic acid encoding a TGFBr2 switch polypeptide or TGFBr2 dominant negative polypeptide. [0555] Disclosed herein, in some embodiments, are modified immune cells, e.g., T cells comprising the recombinant nucleic acid as described herein, or the vectors as described herein. In some embodiments, the modified T cell further comprises a functional disruption of an endogenous TCR. Also disclosed herein, in some embodiments, are modified T cells comprising the sequence encoding the TFP of the nucleic acid as described herein or a TFP encoded by the sequence of the nucleic acid as described herein and a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In some embodiments, the modified T cell further comprises a functional disruption of an endogenous TCR. Further disclosed herein, in some embodiments, are modified allogenic T cells comprising the sequence encoding the TFP as described herein or a TFP encoded by the sequence of the nucleic acid as described herein and a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. [0556] Disclosed herein, in some embodiments, are modified T cell comprising the recombinant nucleic acid disclosed above, or the vector disclosed above; wherein the modified T cell further comprises a functional disruption of an endogenous TCR. Further disclosed herein, in some embodiments, are modified T cells comprising the sequence encoding the TFP of the nucleic acid disclosed above or a TFP encoded by the sequence of the nucleic acid disclosed above encoding the TFP as described herein and a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, wherein the modified T cell further comprises a functional disruption of an endogenous TCR. Also disclosed herein, are modified allogenic T cell comprising the sequence encoding the TFP disclosed above or a TFP encoded by the sequence of the nucleic acid disclosed above
encoding the TFP as described herein and a nucleic acid sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. [0557] In some instances, the T cell further comprises a heterologous sequence encoding a TCR constant domain, wherein the TCR constant domain is a TCR alpha constant domain, a TCR beta constant domain, a TCR alpha constant domain and a TCR beta constant domain, a TCR gamma constant domain, a TCR delta constant domain or a TCR gamma constant domain and a TCR delta constant domain. In some instances, the endogenous TCR that is functionally disrupted is an endogenous TCR alpha chain, an endogenous TCR beta constant domain, an endogenous TCR alpha constant domain and an endogenous TCR beta constant domain, an endogenous TCR gamma chain, an endogenous TCR delta chain, or an endogenous TCR gamma chain and an endogenous TCR delta chain. In some instances, the endogenous TCR that is functionally disrupted has reduced binding to MHC-peptide complex compared to that of an unmodified control T cell. In some instances, the functional disruption is a disruption of a gene encoding the endogenous TCR. In some instances, the disruption of a gene encoding the endogenous TCR is a removal of a sequence of the gene encoding the endogenous TCR from the genome of a T cell. In some instances, the T cell is a CD8+ T cell, a CD4+ T cell, a naïve T cell, a memory stem T cell, a central memory T cell, a double negative T cell, an effector memory T cell, an effector T cell, a ThO cell, a TcO cell, a Th1 cell, a Tc1 cell, a Th2 cell, a Tc2 cell, a Th17 cell, a Th22 cell, a gamma delta T cell, a natural killer (NK) cell, a natural killer T (NKT) cell, a hematopoietic stem cell, or a pluripotent stem cell. In some instances, the T cell is a human T cell. In some instances, the T cell is a CD8+ or CD4+ T cell. In some embodiments, the T cell is a CD4+CD8+ T cell. In some embodiments, the T cell is a human αβ T cell. In some embodiments, the T cell is a human γδ T cell. In some embodiments, the cell is a human NKT cell. In some embodiments, the cell is an allogeneic cell or an autologous cell. In some embodiments, the T cell is modified to comprise a functional disruption of the TCR. In some embodiments, the modified T cells are γδ T cells and do not comprise a functional disruption of an endogenous TCR. In some embodiments, the γδ T cells are V 1+ V 2- T cells. In some embodiments, the γδ T cells are V 1- V 2+ T cells. In some embodiments, the γδ T cells are V 1- V 2- T cells. In some instances, the T cell is an allogenic T cell. [0558] In some instances, the modified T cells further comprise a nucleic acid encoding an inhibitory molecule that comprises a first polypeptide comprising at least a portion of an inhibitory molecule, associated with a second polypeptide comprising a positive signal from an intracellular signaling domain. In some instances, the inhibitory molecule comprises the
first polypeptide comprising at least a portion of PD-1 and the second polypeptide comprising a costimulatory domain and primary signaling domain. In some embodiments, a T cell expressing the TFP as descried herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as descried herein can inhibit tumor growth when expressed in a T cell. [0559] In some embodiments, genetically-modified immune cells of the invention comprise an inactivated TCR alpha gene and/or an inactivated TCR beta gene. Inactivation of the TCR alpha gene and/or TCR beta gene to generate the genetically-modified cells of the present disclosure occurs in at least one or both alleles where the TCR alpha gene and/or TCR beta gene is being expressed. Accordingly, inactivation of one or both genes prevents expression of the endogenous TCR alpha chain or the endogenous TCR beta chain protein. Expression of these proteins is required for assembly of the endogenous alpha/beta TCR on the cell surface. Thus, inactivation of the TCR alpha gene and/or the TCR beta gene results in genetically-modified immune that have no detectable cell surface expression of the endogenous alpha/beta TCR. In particular embodiments, the inactivated gene is a TCR alpha constant region (TRAC) gene. In some embodiments, genetically-modified immune cells of the invention comprise an inactivated B2M gene. [0560] In some examples, the TCR alpha gene, the TRAC gene, or the TCR beta gene is inactivated by insertion of a template nucleic acid into a cleavage site in the gene. Insertion of the template nucleic acid disrupts expression of the endogenous TCR alpha chain or TCR beta chain and, therefore, prevents assembly of an endogenous alpha/beta TCR on the T cell surface. In some examples, the template nucleic acid is inserted into the TRAC gene. [0561] In some of those embodiments wherein the genetically-modified immune cell expresses a TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, such cells have no detectable cell-surface expression of an endogenous T cell receptor (e.g., an alpha/beta T cell receptor). Thus, the disclosure further provides a population of genetically-modified immune cells that express a TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein and have no detectable cell- surface expression of an endogenous T cell receptor (e.g., an alpha/beta T cell receptor), and in some embodiments also express a TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. For example, the population can include a plurality of genetically-modified immune cells of the disclosure which express a TFP as described herein
and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, and have no cell- surface expression of an endogenous T cell receptor (i.e., are TCR-). [0562] As used herein, “detectable cell-surface expression of an endogenous TCR” refers to the ability to detect one or more components of the TCR complex (e.g., an alpha/beta TCR complex) on the cell surface of an immune cell using standard experimental methods. Such methods can include, for example, immunostaining and/or flow cytometry specific for components of the TCR itself, such as a TCR alpha or TCR beta chain, or for components of the assembled cell-surface TCR complex, such as CD3. Methods for detecting cell-surface expression of an endogenous TCR (e.g., an alpha/beta TCR) on an immune cell include those described in the examples herein, and, for example, those described in MacLeod et al. (2017) Molecular Therapy 25(4): 949-961. [0563] In some cases, the present disclosure provides a modified allogenic immune cell, e.g., T cell comprising the sequence encoding the TFP described herein or a TFP encoded by the sequence of the nucleic acid as described herein encoding the TFP and the sequence encoding the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. Sources of T cells [0564] Prior to expansion and genetic modification, a source of T cells is obtained from a subject. Examples of subjects include humans, dogs, cats, mice, rats, and transgenic species thereof. T cells can be obtained from a number of sources, including peripheral blood mononuclear cells, bone marrow, lymph node tissue, cord blood, thymus tissue, tissue from a site of infection, ascites, pleural effusion, spleen tissue, and tumors. In certain aspects of the present disclosure, any number of T cell lines available in the art, may be used. In certain aspects of the present disclosure, T cells can be obtained from a unit of blood collected from a subject using any number of techniques known to the skilled artisan, such as FicollTM separation. In one preferred aspect, cells from the circulating blood of an individual are obtained by apheresis. The apheresis product typically contains lymphocytes, including T cells, monocytes, granulocytes, B cells, other nucleated white blood cells, red blood cells, and platelets. In one aspect, the cells collected by apheresis may be washed to remove the plasma fraction and to place the cells in an appropriate buffer or media for subsequent processing steps. In one aspect of the present disclosure, the cells are washed with phosphate buffered saline (PBS). In an alternative aspect, the wash solution lacks calcium and may lack magnesium or may lack many if not all divalent cations. Initial activation steps in the absence of calcium can lead to magnified activation. As those of ordinary skill in the art would readily
appreciate a washing step may be accomplished by methods known to those in the art, such as by using a semi-automated “flow-through” centrifuge (for example, the Cobe® 2991 cell processor, the Baxter Oncology CytoMate™, or the Haemonetics® Cell Saver® 5) according to the manufacturer’s instructions. After washing, the cells may be resuspended in a variety of biocompatible buffers, such as, for example, Ca-free, Mg-free PBS, PlasmaLyte A, or other saline solution with or without buffer. Alternatively, the undesirable components of the apheresis sample may be removed, and the cells directly resuspended in culture media. [0565] In one aspect, T cells are isolated from peripheral blood lymphocytes by lysing the red blood cells and depleting the monocytes, for example, by centrifugation through a PERCOLL® gradient or by counterflow centrifugal elutriation. A specific subpopulation of T cells, such as CD3+, CD28+, CD4+, CD8+, CD45RA+, and CD45RO+ T cells, can be further isolated by positive or negative selection techniques. For example, in one aspect, T cells are isolated by incubation with anti-CD3/anti-CD28 (e.g., 3x28)-conjugated beads, such as DYNABEADS® M-450 CD3/CD28 T, for a time period sufficient for positive selection of the desired T cells. In one aspect, the time period is about 30 minutes. In a further aspect, the time period ranges from 30 minutes to 36 hours or longer and all integer values there between. In a further aspect, the time period is at least 1, 2, 3, 4, 5, or 6 hours. In yet another preferred aspect, the time period is 10 to 24 hours. In one aspect, the incubation time period is 24 hours. Longer incubation times may be used to isolate T cells in any situation where there are few T cells as compared to other cell types, such in isolating tumor infiltrating lymphocytes (TIL) from tumor tissue or from immunocompromised individuals. Further, use of longer incubation times can increase the efficiency of capture of CD8+ T cells. Thus, by simply shortening or lengthening the time T cells are allowed to bind to the CD3/CD28 beads and/or by increasing or decreasing the ratio of beads to T cells (as described further herein), subpopulations of T cells can be preferentially selected for or against at culture initiation or at other time points during the process. Additionally, by increasing or decreasing the ratio of anti-CD3 and/or anti-CD28 antibodies on the beads or other surface, subpopulations of T cells can be preferentially selected for or against at culture initiation or at other desired time points. The skilled artisan would recognize that multiple rounds of selection can also be used in the context of this present disclosure. In certain aspects, it may be desirable to perform the selection procedure and use the “unselected” cells in the activation and expansion process. “Unselected” cells can also be subjected to further rounds of selection. [0566] Enrichment of a T cell population by negative selection can be accomplished with a combination of antibodies directed to surface markers unique to the negatively selected cells. One method is cell sorting and/or selection via negative magnetic immunoadherence or flow
cytometry that uses a cocktail of monoclonal antibodies directed to cell surface markers present on the cells negatively selected. For example, to enrich for CD4+ cells by negative selection, a monoclonal antibody cocktail typically includes antibodies to CD14, CD20, CD11b, CD16, HLA-DR, and CD8. In certain aspects, it may be desirable to enrich for or positively select for regulatory T cells which typically express CD4+, CD25+, CD62Lhi, GITR+, and FoxP3+. Alternatively, in certain aspects, T regulatory cells are depleted by anti- C25 conjugated beads or other similar method of selection. [0567] In one embodiment, a T cell population can be selected that expresses one or more of IFN-γ TNF-alpha, IL-17A, IL-2, IL-3, IL-4, GM-CSF, IL-10, IL-13, granzyme B, and perforin, or other appropriate molecules, e.g., other cytokines. Methods for screening for cell expression can be determined, e.g., by the methods described in PCT Publication No.: WO 2013/126712. [0568] For isolation of a desired population of cells by positive or negative selection, the concentration of cells and surface (e.g., particles such as beads) can be varied. In certain aspects, it may be desirable to significantly decrease the volume in which beads and cells are mixed together (e.g., increase the concentration of cells), to ensure maximum contact of cells and beads. For example, in one aspect, a concentration of 2 billion cells/mL is used. In one aspect, a concentration of 1 billion cells/mL is used. In a further aspect, greater than 100 million cells/mL is used. In a further aspect, a concentration of cells of 10, 15, 20, 25, 30, 35, 40, 45, or 50 million cells/mL is used. In yet one aspect, a concentration of cells from 75, 80, 85, 90, 95, or 100 million cells/mL is used. In further aspects, concentrations of 125 or 150 million cells/mL can be used. Using high concentrations can result in increased cell yield, cell activation, and cell expansion. Further, use of high cell concentrations allows more efficient capture of cells that may weakly express target antigens of interest, such as CD28-negative T cells, or from samples where there are many tumor cells present (e.g., leukemic blood, tumor tissue, etc.). Such populations of cells may have therapeutic value and would be desirable to obtain. For example, using high concentration of cells allows more efficient selection of CD8+ T cells that normally have weaker CD28 expression. [0569] In a related aspect, it may be desirable to use lower concentrations of cells. By significantly diluting the mixture of T cells and surface (e.g., particles such as beads), interactions between the particles and cells are minimized. This selects for cells that express high amounts of desired antigens to be bound to the particles. For example, CD4+ T cells express higher levels of CD28 and are more efficiently captured than CD8+ T cells in dilute concentrations. In one aspect, the concentration of cells used is 5x106/mL. In other aspects, the concentration used can be from about 1x105/mL to 1x106/mL, and any integer value in
between. In other aspects, the cells may be incubated on a rotator for varying lengths of time at varying speeds at either 2-10 °C or at room temperature. [0570] T cells for stimulation can also be frozen after a washing step. Wishing not to be bound by theory, the freeze and subsequent thaw step provides a more uniform product by removing granulocytes and to some extent monocytes in the cell population. After the washing step that removes plasma and platelets, the cells may be suspended in a freezing solution. While many freezing solutions and parameters are known in the art and will be useful in this context, one method involves using PBS containing 20% DMSO and 8% human serum albumin, or culture media containing 10% Dextran 40 and 5% Dextrose, 20% Human Serum Albumin and 7.5% DMSO, or 31.25% Plasmalyte-A, 31.25% Dextrose 5%, 0.45% NaCl, 10% Dextran 40 and 5% Dextrose, 20% Human Serum Albumin, and 7.5% DMSO or other suitable cell freezing media containing for example, Hespan and PlasmaLyte A, the cells then are frozen to -80 °C at a rate of 1 per minute and stored in the vapor phase of a liquid nitrogen storage tank. Other methods of controlled freezing may be used as well as uncontrolled freezing immediately at -20 °C or in liquid nitrogen. In certain aspects, cryopreserved cells are thawed and washed as described herein and allowed to rest for one hour at room temperature prior to activation using the methods of the present disclosure. [0571] Also contemplated in the context of the present disclosure is the collection of blood samples or apheresis product from a subject at a time period prior to when the expanded cells as described herein might be needed. As such, the source of the cells to be expanded can be collected at any time point necessary, and desired cells, such as T cells, isolated and frozen for later use in T cell therapy for any number of diseases or conditions that would benefit from T cell therapy, such as those described herein. In one aspect a blood sample or an apheresis is taken from a generally healthy subject. In certain aspects, a blood sample or an apheresis is taken from a generally healthy subject who is at risk of developing a disease, but who has not yet developed a disease, and the cells of interest are isolated and frozen for later use. In certain aspects, the T cells may be expanded, frozen, and used at a later time. In certain aspects, samples are collected from a patient shortly after diagnosis of a particular disease as described herein but prior to any treatments. In a further aspect, the cells are isolated from a blood sample or an apheresis from a subject prior to any number of relevant treatment modalities, including but not limited to treatment with agents such as natalizumab, efalizumab, antiviral agents, chemotherapy, radiation, immunosuppressive agents such as cyclosporin, azathioprine, methotrexate, and mycophenolate, antibodies, or other immunoablative agents such as alemtuzumab, anti-CD3 antibodies, cytoxan, fludarabine, cyclosporin, tacrolimus, rapamycin, mycophenolic acid, steroids, romidepsin, and irradiation.
[0572] In a further aspect of the present disclosure, T cells are obtained from a patient directly following treatment that leaves the subject with functional T cells. In this regard, it has been observed that following certain cancer treatments, in particular treatments with drugs that damage the immune system, shortly after treatment during the period when patients would normally be recovering from the treatment, the quality of T cells obtained may be optimal or improved for their ability to expand ex vivo. Likewise, following ex vivo manipulation using the methods described herein, these cells may be in a preferred state for enhanced engraftment and in vivo expansion. Thus, it is contemplated within the context of the present disclosure to collect blood cells, including T cells, dendritic cells, or other cells of the hematopoietic lineage, during this recovery phase. Further, in certain aspects, mobilization (for example, mobilization with GM-CSF) and conditioning regimens can be used to create a condition in a subject wherein repopulation, recirculation, regeneration, and/or expansion of particular cell types is favored, especially during a defined window of time following therapy. Illustrative cell types include T cells, B cells, dendritic cells, and other cells of the immune system. Activation and Expansion of T Cells [0573] T cells may be activated and expanded generally using methods as described, for example, in U.S. Pat. Nos.6,352,694; 6,534,055; 6,905,680; 6,692,964; 5,858,358; 6,887,466; 6,905,681; 7,144,575; 7,067,318; 7,172,869; 7,232,566; 7,175,843; 5,883,223; 6,905,874; 6,797,514; 6,867,041; and 7,572,631. [0574] Generally, the T cells of the present disclosure may be expanded by contact with a surface having attached thereto an agent that stimulates a CD3/TCR complex associated signal and a ligand that stimulates a costimulatory molecule on the surface of the T cells. In particular, T cell populations may be stimulated as described herein, such as by contact with an anti-CD3 antibody, or antigen-binding fragment thereof, or an anti-CD2 antibody immobilized on a surface, or by contact with a protein kinase C activator (e.g., bryostatin) in conjunction with a calcium ionophore. For co-stimulation of an accessory molecule on the surface of the T cells, a ligand that binds the accessory molecule is used. For example, a population of T cells can be contacted with an anti-CD3 antibody and an anti-CD28 antibody, under conditions appropriate for stimulating proliferation of the T cells. To stimulate proliferation of either CD4+ T cells, CD8+ T cells, or CD4+CD8+ T cells, an anti-CD3 antibody and an anti-CD28 antibody. Examples of an anti-CD28 antibody include 9.3, B-T3, XR-CD28 (Diaclone, Besancon, France) can be used as can other methods commonly known in the art (Berg et al., Transplant Proc.30(8):3975-3977, 1998; Haanen et al., J. Exp. Med.
190(9):13191328, 1999; Garland et al., J. Immunol. Meth.227(1-2):53-63, 1999). T cells may additionally be activated and expanded in the presence of a cytokine with or without an anti-CD3 and/or CD28 antibody. Exemplary cytokines include IL-2, IL-7, IL-15, and IL-21. In some embodiments, T cells are activated by incubation with anti-CD3/anti-CD28- conjugated beads, such as DYNABEADS® or Trans-Act® beads, for a time period sufficient for activation of the T cells. In one aspect, the time period is at least 1, 2, 3, 4, 5, or 6 hours. In yet another preferred aspect, the time period is 10 to 24 hours, e.g., 24 hours. In some embodiments, T cells are activated by stimulation with an anti-CD3 antibody and an anti- CD28 antibody in combination with cytokines that bind the common gamma-chain (e.g., IL- 2, IL-7, IL-12, IL-15, IL-21, and others). In some embodiments, T cells are activated by stimulation with an anti-CD3 antibody and an anti-CD28 antibody in combination with 10, 20, 30, 40, 50, 60, 70, 80, 90, 100, 200, 300, 400, 500, 600, 700, 800, 900, or 100 U/mL of IL-2, IL-7, and/or IL-15. In some embodiments, the cells are activated for 24 hours. In some embodiments, after transduction, the cells are expanded in the presence of anti-CD3 antibody, anti-CD28 antibody in combination with the same cytokines. In some embodiments, cells activated in the presence of an anti-CD3 antibody and an anti-CD28 antibody in combination with cytokines that bind the common gamma-chain are expanded in the presence of the same cytokines in the absence of the anti-CD3 antibody and anti-CD28 antibody after transduction. In some embodiments, after transduction, the cells are expanded in the presence of anti-CD3 antibody, anti-CD28 antibody in combination with the same cytokines up to a first washing step, when the cells are sub-cultured in media that includes the cytokines but does not include the anti-CD3 antibody and anti-CD28 antibody. In some embodiments, the cells are subcultured every 1, 2, 3, 4, 5, or 6 days. In some embodiments, cells are expanded for 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, or 30 days. [0575] The expansion of T cells may be stimulated with zoledronic acid (Zometa), alendronic acid (Fosamax) or other related bisphosphonate drugs at concentrations of 0.1, 0.25, 0.5, 1.0, 2.0, 3.0, 4.0, 5.0, 7.5, 10, or 100 µM in the presence of feeder cells (irradiated cancer cells, PBMCs, artificial antigen presenting cells). The expansion of T cells may be stimulated with isopentyl pyrophosphate (IPP), (E)-4-Hydroxy-3-methyl-but-2-enyl pyrophosphate (HMBPP or HMB-PP) or other structurally related compounds at concentrations of 0.1, 0.25, 0.5, 1.0, 2.0, 3.0, 4.0, 5.0, 7.5, 10, or 100 µM in the presence of feeder cells (irradiated cancer cells, PBMCs, artificial antigen presenting cells). In some embodiments, the expansion of T cells may be stimulated with synthetic phosphoantigens (e.g., bromohydrin pyrophosphate; BrHPP), 2M3B1 PP, or 2-methyl-3-butenyl-1 -pyrophosphate in the presence of IL-2 for one-
to-two weeks. In some embodiments, the expansion of T cells may be stimulated with immobilized anti-TCRyd (e.g., pan TCRY6) in the presence of IL-2, e.g., for approximately 14 days. In some embodiments, the expansion of T cells may be stimulated with culture of immobilized anti-CD3 antibodies (e.g., OKT3) in the presence of IL-2. In some embodiments, the aforementioned culture is maintained for about seven days prior to subculture in soluble anti-CD3, and IL-2. [0576] T cells that have been exposed to varied stimulation times may exhibit different characteristics. For example, typical blood or apheresed peripheral blood mononuclear cell products have a helper T cell population (TH, CD4+) that is greater than the cytotoxic or suppressor T cell population (TC, CD8+). Ex vivo expansion of T cells by stimulating CD3 and CD28 receptors produces a population of T cells that prior to about days 8-9 consists predominately of TH cells, while after about days 8-9, the population of T cells comprises an increasingly greater population of TC cells. Accordingly, depending on the purpose of treatment, infusing a subject with a T cell population comprising predominately of TH cells may be advantageous. Similarly, if an antigen-specific subset of TC cells has been isolated it may be beneficial to expand this subset to a greater degree. [0577] Further, in addition to CD4 and CD8 markers, other phenotypic markers vary significantly, but in large part, reproducibly during the course of the cell expansion process. Thus, such reproducibility enables the ability to tailor an activated T cell product for specific purposes. [0578] Once an anti-CD19, anti-BCMA, anti-CD22, anti-ROR1, anti-PD-1, or anti-BAFFR, anti-MUC16, anti-mesothelin, anti-HER2, anti-PMSA, anti-CD20, anti-CD70, anti-GPC3, anti-Nectin-4, anti-Trop2, or antiCD79b TFP is constructed, various assays can be used to evaluate the activity of the molecule, such as but not limited to, the ability to expand T cells following antigen stimulation, sustain T cell expansion in the absence of re-stimulation, and anti-cancer activities in appropriate in vitro and animal models. Assays to evaluate the effects of an anti-CD19, anti-BCMA, anti-GPC3, anti-Nectin-4, anti-Trop2, anti-CD22, anti-MSLN, anti-CD79B, anti-ROR1, anti-PD-1, anti-IL13Ra2, anti-PD-L1, anti-CD20, anti-CD70, or anti-BAFFR TFP are described in further detail below. [0579] Western blot analysis of TFP expression in primary T cells can be used to detect the presence of monomers and dimers (see, e.g., Milone et al., Molecular Therapy 17(8): 1453- 1464 (2009)). Very briefly, T cells (1:1 mixture of CD4+ and CD8+ T cells) expressing the TFPs are expanded in vitro for more than 10 days followed by lysis and SDS-PAGE under reducing conditions. TFPs are detected by western blotting using an antibody to a TCR chain.
The same T cell subsets are used for SDS-PAGE analysis under non-reducing conditions to permit evaluation of covalent dimer formation. [0580] In vitro expansion of T cells expressing TFT as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein following antigen stimulation can be measured by flow cytometry. For example, a mixture of CD4+ and CD8+ T cells are stimulated with alphaCD3/alphaCD28 and APCs followed by transduction with lentiviral vectors expressing GFP under the control of the promoters to be analyzed. Exemplary promoters include the CMV IE gene, EF-1alpha, ubiquitin C, or phosphoglycerokinase (PGK) promoters. GFP fluorescence is evaluated on day 6 of culture in the CD4+ and/or CD8+ T cell subsets by flow cytometry (see, e.g., Milone et al., Molecular Therapy 17(8): 1453-1464 (2009)). Alternatively, a mixture of CD4+ and CD8+ T cells are stimulated with alphaCD3/alphaCD28 coated magnetic beads on day 0 and transduced with TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein on day 1 using, e.g., a bicistronic lentiviral vector expressing TFP as described herein along with eGFP using a 2A ribosomal skipping sequence and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. Cultures are re-stimulated with either TAA+ K562 cells (K562-TAA), wild-type K562 cells (K562 wild type) or K562 cells expressing hCD32 and 4-1BBL in the presence of anti-CD3 and anti-CD28 antibody (K562-BBL-3/28) following washing. Exogenous IL-2 is added to the cultures every other day at 100 IU/mL. GFP+ T cells are enumerated by flow cytometry using bead-based counting (see, e.g., Milone et al., Molecular Therapy 17(8): 1453-1464 (2009)). [0581] Sustained expansion of T cells expressing TFT as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein in the absence of re-stimulation can also be measured (see, e.g., Milone et al., Molecular Therapy 17(8): 1453-1464 (2009)). Briefly, mean T cell volume (fl) is measured on day 8 of culture using a Coulter Multisizer III particle counter following stimulation with alphaCD3/alphaCD28 coated magnetic beads on day 0, and transduction with the indicated TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein on day 1. [0582] Animal models can also be used to measure an activity of T cells expressing TFT as described herein and a switch polypeptide or dominant negative polypeptide comprising a
TGFBr2 extracellular domain or a functional fragment thereof as described herein. For example, xenograft model using, e.g., human CD19-specific TFP+ T cells co-expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein to treat a primary human pre-B ALL in immunodeficient mice can be used (see, e.g., Milone et al., Molecular Therapy 17(8): 1453-1464 (2009)). After establishment of ALL, mice are randomized as to treatment groups. Different numbers of engineered T cells are coinjected at a 1:1 ratio into NOD/SCID/γ-/- mice bearing B-ALL. The number of copies of each vector in spleen DNA from mice is evaluated at various times following T cell injection. Animals are assessed for leukemia at weekly intervals. Peripheral blood CD19+ B-ALL blast cell counts are measured in mice that are injected with alphaCD19-zeta TFP+ T cells co-expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein or mock-transduced T cells. Survival curves for the groups are compared using the log-rank test. In addition, absolute peripheral blood CD4+ and CD8+ T cell counts 4 weeks following T cell injection in NOD/SCID/γ-/- mice can also be analyzed. Mice are injected with leukemic cells and 3 weeks later are injected with T cells engineered to express TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein by, e.g., a bicistronic lentiviral vector that encodes the TFP as described herein linked to eGFP and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. T cells are normalized to 45-50% input GFP+ T cells co-expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein by mixing with mock-transduced cells prior to injection and confirmed by flow cytometry. Animals are assessed for leukemia at 1-week intervals. Survival curves for the groups of TFP+ T cell co-expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein are compared using the log-rank test. [0583] Dose dependent TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein treatment response can be evaluated (see, e.g., Milone et al., Molecular Therapy 17(8): 1453-1464 (2009)). For example, peripheral blood is obtained 35- 70 days after establishing leukemia in mice injected on day 21 with TFP T cells co- expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, an equivalent
number of mock-transduced T cells, or no T cells. Mice from each group are randomly bled for determination of peripheral blood CD19+ ALL blast counts and then killed on days 35 and 49. The remaining animals are evaluated on days 57 and 70. [0584] Assessment of cell proliferation and cytokine production has been previously described, e.g., at Milone et al., Molecular Therapy 17(8): 1453-1464 (2009). Briefly, assessment of TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein-mediated proliferation is performed in microtiter plates by mixing washed T cells with K562 cells expressing the tumor associated antigen (TAA, e.g., CD19) CD19 (K19) or CD32 and CD137 (KT32-BBL) for a final T cell:K562 ratio of 2:1. K562 cells are irradiated with gamma-radiation prior to use. Anti-CD3 (clone OKT3) and anti-CD28 (clone 9.3) monoclonal antibodies are added to cultures with KT32-BBL cells to serve as a positive control for stimulating T cell proliferation since these signals support long-term CD8+ T cell expansion ex vivo. T cells are enumerated in cultures using CountBrightTM fluorescent beads (Invitrogen) and flow cytometry as described by the manufacturer. TFP+ T cells co- expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein are identified by GFP expression using T cells that are engineered with eGFP-2A linked TFP-expressing lentiviral vectors. For TFP+ T cells co-expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, but not expressing GFP, the TFP+ T cells co-expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein are detected with biotinylated recombinant CD19 protein and a secondary avidin-PE conjugate. CD4+ and CD8+ expression on T cells are also simultaneously detected with specific monoclonal antibodies (BD Biosciences). Cytokine measurements are performed on supernatants collected 24 hours following re- stimulation using the human TH1/TH2 cytokine cytometric bead array kit (BD Biosciences) according the manufacturer’s instructions. Fluorescence is assessed using a FACScalibur™ flow cytometer (BD Biosciences), and data are analyzed according to the manufacturer’s instructions. [0585] Cytotoxicity can be assessed by a standard 51Cr-release assay (see, e.g., Milone et al., Molecular Therapy 17(8): 1453-1464 (2009)). Target cells (K562 lines and primary pro-B- ALL cells) are loaded with 51Cr (as NaCrO4, New England Nuclear) at 37 °C for 2 hours with frequent agitation, washed twice in complete RPMI and plated into microtiter plates. Effector T cells are mixed with target cells in the wells in complete RPMI at varying ratios of effector
cell:target cell (E:T). Additional wells containing media only (spontaneous release, SR) or a 1% solution of Triton-X 100 detergent (total release, TR) are also prepared. After 4 hours of incubation at 37 °C, supernatant from each well is harvested. Released 51Cr is then measured using a gamma particle counter (Packard Instrument Co., Waltham, Mass.). Each condition is performed in at least triplicate, and the percentage of lysis is calculated using the formula: % Lysis=(ER-SR)/(TR-SR), where ER represents the average 51Cr released for each experimental condition. [0586] Imaging technologies can be used to evaluate specific trafficking and proliferation of TFPs in tumor-bearing animal models. Such assays have been described, e.g., in Barrett et al., Human Gene Therapy 22:1575-1586 (2011). NOD/SCID/γc-/- (NSG) mice are injected IV with Nalm-6 cells (ATCC® CRL-3273™) followed 7 days later with T cells 4 hour after electroporation with the TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein constructs. The T cells are stably transfected with a lentiviral construct to express firefly luciferase, and mice are imaged for bioluminescence. Alternatively, therapeutic efficacy and specificity of a single injection of TFP+ T cells co- expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein in Nalm-6 xenograft model can be measured as the following: NSG mice are injected with Nalm-6 transduced to stably express firefly luciferase, followed by a single tail-vein injection of T cells electroporated with a TAA-TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein 7 days later. Animals are imaged at various time points post injection. For example, photon-density heat maps of firefly luciferase positive leukemia in representative mice at day 5 (2 days before treatment) and day 8 (24 hours post TFP+ PBLs) can be generated. [0587] Other assays, including those described herein as well as those that are known in the art can also be used to evaluate the anti-CD19, anti-BCMA, anti-CD22, anti-MSLN, anti- CD79B, anti-GPC3, anti-Nectin-4, anti-Trop2, anti-IL13Ra2, anti-PD-1, anti-ROR1, anti-PD- L1, or anti-BAFFR TFP constructs as described herein. Other Agents [0588] Disclosed herein, in some embodiments, are recombinant nucleic acid molecules comprising a first sequence encoding a TFP as described herein and a second sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2
extracellular domain or a functional fragment thereof as described herein, and a third nucleic acid sequence encoding an agent that can enhance the activity of a modified T cell expressing the TFP as described herein and the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In some embodiments, the third nucleic acid sequence is included in a separate nucleic acid sequence. In some embodiments, the third nucleic acid sequence is included in the same nucleic acid molecule as the recombinant nucleic acid molecules. For example, in one embodiment, the agent that can enhance the activity of a modified T cell can be a PD-1 polypeptide. In these embodiments, the PD-1 polypeptide may be operably linked to the N- terminus of an intracellular domain of a costimulatory polypeptide via the C-terminus of the PD-1 polypeptide. For example, in another embodiment, the agent that can enhance the activity of a modified T cell can be an anti-PD-1 antibody, or antigen binding fragment thereof. In this embodiment, the anti-PD-1 antibody or antigen binding fragment thereof may be operably linked to the N-terminus of an intracellular domain of a costimulatory polypeptide via the C-terminus of the anti-PD-1 antibody, or antigen binding fragment thereof. In some embodiments, the PD-1 polypeptide or anti-PD-1 antibody is linked to the intracellular domain of the costimulatory polypeptide via the transmembrane domain of PD- 1. In some embodiments, the costimulatory polypeptide is selected from the group consisting of OX40, CD2, CD27, CD5, ICAM-1, ICOS (CD278), 4-1BB (CD137), GITR, CD28, CD30, CD40, IL-15Ra, IL12R, IL18R, IL21R, BAFFR, HVEM, CD7, LIGHT, NKG2C, SLAMF7, NKp80, CD160, CD226, FcγRI, FcγRII, and FcγRIII. In some embodiments, the costimulatory peptide is CD28. [0589] Disclosed herein, in some embodiments, are recombinant nucleic acid molecules comprising a first sequence encoding a TFP as described herein and a second sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, wherein the recombinant nucleic acid molecules further comprising an agent that can enhance the activity of a modified T cell expressing the TFP as described herein and the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In another aspect, the cells expressing TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein can further express another agent, e.g., an agent which enhances the activity of a modified T cell. For example, in one embodiment, the agent can be an agent which inhibits an inhibitory molecule. Inhibitory molecules, e.g., PD-1, can, in some embodiments, decrease the ability of a modified T cell to
mount an immune effector response. Examples of inhibitory molecules include PD-1, PD-L1, CTLA4, TIM3, LAG3, VISTA, BTLA, TIGIT, LAIR1, CD160, and 2B4. In one embodiment, the agent which inhibits an inhibitory molecule comprises a first polypeptide, e.g., an inhibitory molecule, associated with a second polypeptide that provides a positive signal to the cell, e.g., an intracellular signaling domain as described herein. In one embodiment, the agent comprises a first polypeptide, e.g., of an inhibitory molecule such as PD-1, LAG3, CTLA4, CD160, BTLA, LAIR1, TIM3, 2B4, and TIGIT, or a fragment of any of these (e.g., at least a portion of an extracellular domain of any of these), and a second polypeptide which is an intracellular signaling domain described herein (e.g., comprising a costimulatory domain (e.g., 4-1BB, CD27 or CD28, e.g., as described herein) and/or a primary signaling domain (e.g., a CD3 zeta signaling domain described herein). In one embodiment, the agent comprises a first polypeptide of PD-1 or a fragment thereof (e.g., at least a portion of an extracellular domain of PD-1), and a second polypeptide of an intracellular signaling domain described herein (e.g., a CD28 signaling domain described herein and/or a CD3 zeta signaling domain described herein). In some embodiments, the recombinant nucleic acid molecules as described herein further comprises a sequence encoding PD-1 or a fragment thereof. In some embodiments, the recombinant nucleic acid molecules as described herein further comprises a sequence encoding the extracellular domain of PD-1. In some embodiments, the recombinant nucleic acid molecules as described herein comprises a sequence encoding the extracellular domain and transmembrane domain of PD-1. In some embodiments, the recombinant nucleic acid molecules as described herein may further comprise a sequence encoding CD28 or a fragment thereof. In some embodiments, the recombinant nucleic acid molecules as described herein comprises a sequence encoding the intracellular domain of CD28. In some embodiments, the recombinant nucleic acid molecules as described herein comprises a sequence encoding a fusion protein comprising the PD-1 extracellular domain and transmembrane domain linked to the CD28 intracellular domain linked to intracellular domain. In some embodiments, the agent comprises the extracellular and transmembrane domain of PD-1 fused to the intracellular signaling domain of CD28. In some embodiments, the agent comprises SEQ ID NO: 370. PD1 is an inhibitory member of the CD28 family of receptors that also includes CD28, CTLA-4, ICOS, and BTLA. PD-1 is expressed on activated B cells, T cells and myeloid cells (Agata et al., 1996, Int. Immunol 8:765-75). Two ligands for PD1, PD-L1 and PD-L2, have been shown to downregulate T cell activation upon binding to PD1 (Freeman et al., 2000 J. Exp. Med.192:1027-34; Latchman et al., 2001 Nat. Immunol.2:261-8; Carter et al., 2002 Eur. J. Immunol.32:634-43). PD-L1 is abundant in human cancers (Dong et al., 2003 J. Mol.
Med.81:281-7; Blank et al., 2005 Cancer Immunol. Immunother.54:307-314; Konishi et al., 2004 Clin. Cancer Res.10:5094). Immune suppression can be reversed by inhibiting the local interaction of PD1 with PD-L1. [0590] PD1CD28 fusion protein/switch-receptor, amino acid sequence (SEQ ID NO: 370) MQIPQAPWPVVWAVLQLGWRPGWFLDSPDRPWNPPTFSPALLVVTEGDNATFTCSF SNTSESFVLNWYRMSPSNQTDKLAAFPEDRSQPGQDCRFRVTQLPNGRDFHMSVVR ARRNDSGTYLCGAISLAPKAQIKESLRAELRVTERRAEVPTAHPSPSPRPAGQFQTLV VGVVGGLLGSLVLLVWVLAVIRSKRSRLLHSDYMNMTPRRPGPTRKHYQPYAPPRD FAAYRS [0591] In one embodiment, the agent comprises the extracellular domain (ECD) of an inhibitory molecule, e.g., Programmed Death 1 (PD-1) can be fused to a transmembrane domain and optionally an intracellular signaling domain such as 41BB and CD3 zeta (also referred to herein as a PD-1 TFP). In one embodiment, the PD-1 TFP, when used in combinations with an anti-TAA TFP described herein, improves the persistence of the T cell. In one embodiment, the TFP is a PD-1 TFP comprising the extracellular domain of PD-1. Alternatively, provided are TFPs containing an antibody or antibody fragment such as a scFv that specifically binds to the Programmed Death-Ligand 1 (PD-L1) or Programmed Death- Ligand 2 (PD-L2). [0592] In another aspect, the present disclosure provides a population of TFP-expressing T cells, e.g., TFP-T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In some embodiments, the population of TFP-expressing T cells co- expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein comprises a mixture of cells expressing different TFPs. For example, in one embodiment, the population of TFP-T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein can include a first cell expressing a TFP having a binding domain described herein, and a second cell expressing a TFP having a different anti-TAA binding domain, e.g., a binding domain described herein that differs from the binding domain in the TFP expressed by the first cell. As another example, the population of TFP-expressing cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein can include a first cell expressing a TFP that includes a first binding domain binding domain, e.g., as described herein, and a second
cell expressing a TFP that includes an antigen binding domain to a target other than the binding domain of the first cell (e.g., another tumor-associated antigen). [0593] In another aspect, the present disclosure provides a population of cells wherein at least one cell in the population expresses a TFP having a domain described herein, and a second cell expressing another agent, e.g., an agent which enhances the activity of a modified T cell. For example, in one embodiment, the agent can be an agent which inhibits an inhibitory molecule. Inhibitory molecules, e.g., can, in some embodiments, decrease the ability of a modified T cell to mount an immune effector response. Examples of inhibitory molecules include PD-1, PD-L1, PD-L2, CTLA4, TIM3, LAG3, VISTA, BTLA, TIGIT, LAIR1, CD160, and 2B4. In one embodiment, the agent that inhibits an inhibitory molecule comprises a first polypeptide, e.g., an inhibitory molecule, associated with a second polypeptide that provides a positive signal to the cell, e.g., an intracellular signaling domain described herein. Pharmaceutical Compositions [0594] Disclosed herein, in some embodiments, are pharmaceutical compositions comprising: (a) the cells of the disclosure; and (b) a pharmaceutically acceptable carrier. Disclosed herein, in some embodiments, are pharmaceutical compositions comprising: (a) the modified T cells of the disclosure; and (b) a pharmaceutically acceptable carrier. Disclosed herein, in some embodiments, are pharmaceutical compositions comprising: (a) the nucleic acid molecules of the disclosure; and (b) a pharmaceutically acceptable carrier. Such compositions may comprise buffers such as neutral buffered saline, phosphate buffered saline and the like; carbohydrates such as glucose, mannose, sucrose or dextrans, mannitol; proteins; polypeptides or amino acids such as glycine; antioxidants; chelating agents such as EDTA or glutathione; adjuvants (e.g., aluminum hydroxide); and preservatives. Compositions of the present disclosure are in one aspect formulated for intravenous administration. [0595] Pharmaceutical compositions of the present disclosure may be administered in a manner appropriate to the disease to be treated (or prevented). The quantity and frequency of administration will be determined by such factors as the condition of the patient, and the type and severity of the patient’s disease, although appropriate dosages may be determined by clinical trials. [0596] In one embodiment, the pharmaceutical composition is substantially free of, e.g., there are no detectable levels of a contaminant, e.g., selected from the group consisting of endotoxin, mycoplasma, replication competent lentivirus (RCL), p24, VSV-G nucleic acid,
HIV gag, residual anti-CD3/anti-CD28 coated beads, mouse antibodies, pooled human serum, bovine serum albumin, bovine serum, culture media components, vector packaging cell or plasmid components, a bacterium and a fungus. In one embodiment, the bacterium is at least one selected from the group consisting of Alcaligenes faecalis, Candida albicans, Escherichia coli, Haemophilus influenza, Neisseria meningitides, Pseudomonas aeruginosa, Staphylococcus aureus, Streptococcus pneumonia, and Streptococcus pyogenes group A. [0597] When “an immunologically effective amount,” “an anti-tumor effective amount,” “a tumor-inhibiting effective amount,” or “therapeutic amount” is indicated, the precise amount of the compositions of the present disclosure to be administered can be determined by a physician with consideration of individual differences in age, weight, tumor size, extent of infection or metastasis, and condition of the patient (subject). It can generally be stated that a pharmaceutical composition comprising the T cells as described herein may be administered at a dosage of 104 to 109 cells/kg body weight, in some instances 105 to 106 cells/kg body weight, including all integer values within those ranges. T cell compositions may also be administered multiple times at these dosages. The cells can be administered by using infusion techniques that are commonly known in immunotherapy (see, e.g., Rosenberg et al., New Eng. J. Med.319:1676, 1988). [0598] In certain aspects, it may be desired to administer activated T cells to a subject and then subsequently redraw blood (or have an apheresis performed), activate T cells therefrom according to the present disclosure, and reinfuse the patient with these activated and expanded T cells. This process can be carried out multiple times every few weeks. In certain aspects, T cells can be activated from blood draws of from 10 cc to 400 cc. In certain aspects, T cells are activated from blood draws of 20 cc, 30 cc, 40 cc, 50 cc, 60 cc, 70 cc, 80 cc, 90 cc, or 100 cc. [0599] The administration of the subject compositions may be carried out in any convenient manner, including by aerosol inhalation, injection, ingestion, transfusion, implantation or transplantation. The compositions described herein may be administered to a patient trans arterially, subcutaneously, intradermally, intratumorally, intranodally, intramedullary, intramuscularly, by intravenous (i.v.) injection, or intraperitoneally. In one aspect, the T cell compositions of the present disclosure are administered to a patient by intradermal or subcutaneous injection. In one aspect, the T cell compositions of the present disclosure are administered by i.v. injection. The compositions of T cells may be injected directly into a tumor, lymph node, or site of infection. [0600] In a particular exemplary aspect, subjects may undergo leukapheresis, wherein leukocytes are collected, enriched, or depleted ex vivo to select and/or isolate the cells of
interest, e.g., T cells. These T cell isolates may be expanded by methods known in the art and treated such that one or more TFP and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof constructs of the present disclosure may be introduced, thereby creating a modified T-T cell of the present disclosure. Subjects in need thereof may subsequently undergo standard treatment with high dose chemotherapy followed by peripheral blood stem cell transplantation. In certain aspects, following or concurrent with the transplant, subjects receive an infusion of the expanded modified T cells of the present disclosure. In an additional aspect, expanded cells are administered before or following surgery. [0601] The dosage of the above treatments to be administered to a patient will vary with the precise nature of the condition being treated and the recipient of the treatment. The scaling of dosages for human administration can be performed according to art-accepted practices. The dose for alemtuzumab, for example, will generally be in the range 1 to about 100 mg for an adult patient, usually administered daily for a period between 1 and 30 days. The preferred daily dose is 1 to 10 mg per day although in some instances larger doses of up to 40 mg per day may be used (described in U.S. Pat. No.6,120,766). [0602] In one embodiment, the TFP as described herein and the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein is introduced into T cells, e.g., using in vitro transcription, and the subject (e.g., human) receives an initial administration of TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof of the present disclosure, and one or more subsequent administrations of the TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof of the present disclosure, wherein the one or more subsequent administrations are administered less than 15 days, e.g., 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3, or 2 days after the previous administration. In one embodiment, more than one administration of the TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof of the present disclosure are administered to the subject (e.g., human) per week, e.g., 2, 3, or 4 administrations of the TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof of the present disclosure are administered per week. In one embodiment, the subject (e.g., human subject) receives more than one administration of the TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular
domain or a functional fragment thereof as described herein per week (e.g., 2, 3 or 4 administrations per week) (also referred to herein as a cycle), followed by a week of no TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein administrations, and then one or more additional administration of the TFP T cells co- expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein (e.g., more than one administration of the TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein per week) is administered to the subject. In another embodiment, the subject (e.g., human subject) receives more than one cycle of TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, and the time between each cycle is less than 10, 9, 8, 7, 6, 5, 4, or 3 days. In one embodiment, the TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein are administered every other day for 3 administrations per week. In one embodiment, the TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof of the present disclosure are administered for at least two, three, four, five, six, seven, eight or more weeks. [0603] In one aspect, CD19 TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein are generated using lentiviral viral vectors, such as lentivirus. TFP-T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein generated that way will have stable TFP as described herein and the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein expression. [0604] In one aspect, TFP T cells co-expressing the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein transiently express TFP as described herein and/or switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein vectors for 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15 days after transduction. Transient expression of TFPs as described herein and/or the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or
a functional fragment thereof as described herein can be affected by RNA TFP as described herein and/or switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein vector delivery. In one aspect, the TFP as described herein and/or the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein RNA is transduced into the T cell by electroporation. [0605] A potential issue that can arise in patients being treated using T cells transiently expressing TFP as described herein and the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein (particularly with T cells expressing murine scFv bearing TFP) is anaphylaxis after multiple treatments. [0606] Without being bound by this theory, it is believed that such an anaphylactic response might be caused by a patient developing humoral anti-TFP response, i.e., anti-TFP antibodies having an anti-IgE isotype. It is thought that a patient’s antibody producing cells undergo a class switch from IgG isotype (that does not cause anaphylaxis) to IgE isotype when there is a ten to fourteen day break in exposure to antigen. [0607] If a patient is at high risk of generating an anti-TFP antibody response during the course of transient TFP therapy (such as those generated by RNA transductions), TFP T cell infusion breaks should not last more than ten to fourteen days. Methods of Producing Modified T cells [0608] Disclosed herein, in some embodiments, are methods of producing the modified T cells of the disclosure, the method comprising introducing the recombinant nucleic acid as described herein or the vectors as described herein into the cells. The recombinant nucleic acid can comprise a sequence encoding a TFP as described herein and/or a sequence encoding a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In some cases, the method can further comprise (a) disrupting an endogenous TCR gene encoding a TCR alpha chain, a TCR beta chain, a TCR gamma chain, a TCR delta chain, or any combination thereof; thereby producing a T cell containing a functional disruption of an endogenous TCR gene; and (b) transducing the T cell containing a functional disruption of an endogenous TCR gene with the recombinant nucleic acid of the disclosure, or the vectors as described herein. In some instances, disrupting comprises transducing the T cell with a nuclease protein or a nucleic acid sequence encoding a nuclease protein that targets the endogenous gene encoding a TCR alpha chain, a TCR beta chain, or a TCR alpha chain and a TCR beta chain.
[0609] Further disclosed herein, in some embodiments, are methods of producing the modified T cell of the disclosure, the method comprising transducing a T cell containing a functional disruption of an endogenous TCR gene with the recombinant nucleic acid as described herein, or the vectors as described herein. In some instances, the T cell containing a functional disruption of an endogenous TCR gene is a T cell containing a functional disruption of an endogenous TCR gene encoding a TCR alpha chain, a TCR beta chain, or a TCR alpha chain and a TCR beta chain. [0610] In some instances, the T cell is a human T cell. In some instances, the T cell containing a functional disruption of an endogenous TCR gene has reduced binding to MHC- peptide complex compared to that of an unmodified control T cell. [0611] In some instances, the nuclease is a meganuclease, a zinc-finger nuclease (ZFN), a transcription activator-like effector nuclease (TALEN), a CRISPR/Cas nuclease, CRISPR/Cas nickase, or a megaTAL nuclease. In some instances, the sequence comprised by the recombinant nucleic acid or the vector is inserted into the endogenous TCR subunit gene at the cleavage site, and wherein the insertion of the sequence into the endogenous TCR subunit gene functionally disrupts the endogenous TCR subunit. In some instances, the nuclease is a meganuclease. In some instances, the meganuclease comprises a first subunit and a second subunit, wherein the first subunit binds to a first recognition half-site of the recognition sequence, and wherein the second subunit binds to a second recognition half-site of the recognition sequence. In some instances, the meganuclease is a single-chain meganuclease comprising a linker, wherein the linker covalently joins the first subunit and the second subunit. Gene Editing Technologies [0612] In some embodiments, the modified immune cells, e.g., T cells, as described herein are further engineered using a gene editing technique such as clustered regularly interspaced short palindromic repeats (CRISPR®, see, e.g., US Patent No.8,697,359), transcription activator-like effector (TALE) nucleases (TALENs, see, e.g., U.S. Patent No.9,393,257), meganucleases (endodeoxyribonucleases having large recognition sites comprising double- stranded DNA sequences of 12 to 40 base pairs), zinc finger nuclease (ZFN, see, e.g., Urnov et al., Nat. Rev. Genetics (2010) v11, 636-646), or megaTAL nucleases (a fusion protein of a meganuclease to TAL repeats) methods. In this way, a chimeric construct may be engineered to combine desirable characteristics of each subunit, such as conformation or signaling capabilities. See also Sander & Joung, Nat. Biotech. (2014) v32, 347-55; and June et al., 2009 Nature Reviews Immunol.9.10: 704-716, each incorporated herein by reference. In some embodiments, one or more of the extracellular domain, the transmembrane domain, or the
cytoplasmic domain of a TFP subunit are engineered to have aspects of more than one natural TCR subunit domain (i.e., are chimeric). [0613] Recent developments of technologies to permanently alter the human genome and to introduce site-specific genome modifications in disease relevant genes lay the foundation for therapeutic applications. These technologies are now commonly known as “genome editing. [0614] The endogenous TCR gene encoding a TCR alpha chain, a TCR beta chain, or a TCR alpha chain and a TCR beta chain can be inactivated in the modified cell (e.g., modified T cell) described herein. The inactivation can include disruption of genomic gene locus, gene silencing, inhibition or reduction of transcription, or inhibition or reduction of translation. The endogenous TCR gene can be silenced, for example, by inhibitory nucleic acids such as siRNA and shRNA. The translation of the endogenous TCR gene can be inhibited by inhibitory nucleic acids such as microRNA. In some embodiments, gene editing techniques are employed to disrupt an endogenous TCR gene. In some embodiments, mentioned endogenous TCR gene encodes a TCR alpha chain, a TCR beta chain, or a TCR alpha chain and a TCR beta chain. In some embodiments, gene editing techniques pave the way for multiplex genomic editing, which allows simultaneous disruption of multiple genomic loci in endogenous TCR gene. In some embodiments, multiplex genomic editing techniques are applied to generate gene-disrupted T cells that are deficient in the expression of endogenous TCR, and/or B2M, and/or human leukocyte antigens (HLAs), and/or programmed cell death protein 1 (PD-1), and/or other genes. [0615] Current gene editing technologies comprise meganucleases, zinc-finger nucleases (ZFN), TAL effector nucleases (TALEN), and clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated (Cas) system. These four major classes of gene-editing techniques share a common mode of action in binding a user-defined sequence of DNA and mediating a double-stranded DNA break (DSB). DSB may then be repaired by either non-homologous end joining (NHEJ) or –when donor DNA is present- homologous recombination (HR), an event that introduces the homologous sequence from a donor DNA fragment. Additionally, nickase nucleases generate single-stranded DNA breaks (SSB). DSBs may be repaired by single strand DNA incorporation (ssDI) or single strand template repair (ssTR), an event that introduces the homologous sequence from a donor DNA. [0616] Genetic modification of genomic DNA can be performed using site-specific, rare- cutting endonucleases that are engineered to recognize DNA sequences in the locus of interest. Methods for producing engineered, site-specific endonucleases are known in the art. For example, zinc-finger nucleases (ZFNs) can be engineered to recognize and cut predetermined sites in a genome. ZFNs are chimeric proteins comprising a zinc finger DNA-
binding domain fused to the nuclease domain of the Fokl restriction enzyme. The zinc finger domain can be redesigned through rational or experimental means to produce a protein that binds to a pre-determined DNA sequence -18 base pairs in length. By fusing this engineered protein domain to the Fokl nuclease, it is possible to target DNA breaks with genome-level specificity. ZFNs have been used extensively to target gene addition, removal, and substitution in a wide range of eukaryotic organisms (reviewed in Durai et al. (2005), Nucleic Acids Res 33, 5978). Likewise, TAL-effector nucleases (TALENs) can be generated to cleave specific sites in genomic DNA. Like a ZFN, a TALEN comprises an engineered, site- specific DNA-binding domain fused to the Fokl nuclease domain (reviewed in Mak et al. (2013), Curr Opin Struct Biol.23:93-9). In this case, however, the DNA binding domain comprises a tandem array of TAL-effector domains, each of which specifically recognizes a single DNA base pair. Compact TALENs have an alternative endonuclease architecture that avoids the need for dimerization (Beurdeley et al. (2013), Nat Commun.4: 1762). A Compact TALEN comprises an engineered, site-specific TAL-effector DNA-binding domain fused to the nuclease domain from the I-TevI homing endonuclease. Unlike Fokl, I-TevI does not need to dimerize to produce a double-strand DNA break so a Compact TALEN is functional as a monomer. [0617] Engineered endonucleases based on the CRISPR/Cas9 system are also known in the art (Ran et al. (2013), Nat Protoc.8:2281-2308; Mali et al. (2013), Nat Methods 10:957-63). The CRISPR gene-editing technology is composed of an endonuclease protein whose DNA- targeting specificity and cutting activity can be programmed by a short guide RNA or a duplex crRNA/TracrRNA. A CRISPR endonuclease comprises two components: (1) a caspase effector nuclease, typically microbial Cas9; and (2) a short “guide RNA” or an RNA duplex comprising an 18 to 20 nucleotide targeting sequence that directs the nuclease to a location of interest in the genome. By expressing multiple guide RNAs in the same cell, each having a different targeting sequence, it is possible to target DNA breaks simultaneously to multiple sites in the genome (multiplex genomic editing). [0618] There are two classes of CRISPR systems known in the art (Adli (2018) Nat. Commun.9:1911), each containing multiple CRISPR types. Class 1 contains type I and type III CRISPR systems that are commonly found in Archaea. And, Class II contains type II, IV, V, and VI CRISPR systems. Although the most widely used CRISPR/Cas system is the type II CRISPR-Cas9 system, CRISPR/Cas systems have been repurposed by researchers for genome editing. More than 10 different CRISPR/Cas proteins have been remodeled within last few years (Adli (2018) Nat. Commun.9:1911). Among these, such as Cas12a (Cpf1)
proteins from Acid- aminococcus sp (AsCpf1) and Lachnospiraceae bacterium (LbCpf1), are particularly interesting. [0619] Homing endonucleases are a group of naturally-occurring nucleases that recognize 15-40 base-pair cleavage sites commonly found in the genomes of plants and fungi. They are frequently associated with parasitic DNA elements, such as group 1 self-splicing introns and inteins. They naturally promote homologous recombination or gene insertion at specific locations in the host genome by producing a double -stranded break in the chromosome, which recruits the cellular DNA-repair machinery (Stoddard (2006), Q. Rev. Biophys.38: 49- 95). Specific amino acid substations could reprogram DNA cleavage specificity of homing nucleases (Niyonzima (2017), Protein Eng Des Sel.30(7): 503–522). Meganucleases (MN) are monomeric proteins with innate nuclease activity that are derived from bacterial homing endonucleases and engineered for a unique target site (Gersbach (2016), Molecular Therapy. 24: 430–446). In some embodiments, meganuclease is engineered I-CreI homing endonuclease. In other embodiments, meganuclease is engineered I-SceI homing endonuclease. [0620] In addition to mentioned four major gene editing technologies, chimeric proteins comprising fusions of meganucleases, ZFNs, and TALENs have been engineered to generate novel monomeric enzymes that take advantage of the binding affinity of ZFNs and TALENs and the cleavage specificity of meganucleases (Gersbach (2016), Molecular Therapy 24: 430–446). For example, A megaTAL is a single chimeric protein, which is the combination of the easy-to-tailor DNA binding domains from TALENs with the high cleavage efficiency of meganucleases. [0621] In order to perform the gene editing technique, the nucleases, and in the case of the CRISPR/ Cas9 system, a gRNA, may need to be efficiently delivered to the cells of interest. Delivery methods such as physical, chemical, and viral methods are also know in the art (Mali (2013). Indian J. Hum. Genet.19: 3-8.). In some instances, physical delivery methods can be selected from the methods but not limited to electroporation, microinjection, or use of ballistic particles. On the other hand, chemical delivery methods require use of complex molecules such calcium phosphate, lipid, or protein. In some embodiments, viral delivery methods are applied for gene editing techniques using viruses such as but not limited to adenovirus, lentivirus, and retrovirus. [0622] As an example, the endogenous TCR gene (e.g., a TRAC locus or a TRBC locus) encoding a TCR alpha chain, a TCR beta chain, or a TCR alpha chain and a TCR beta chain can be inactivated by CRISPR/Cas9 system. The gRNA used to inactivate (e.g., disrupt) the
TRAC locus can comprise a sequence of SEQ ID: 159. The gRNA used to disrupt the TRBC locus can comprise a sequence of SEQ ID: 197. [0623] CTCGACCAGCTTGACATCAC (SEQ ID NO: 159). [0624] ACACTGGTGTGCCTGGCCAC (SEQ ID NO: 197). Methods of Treatment [0625] Disclosed herein, in some embodiments, is a method of treating a disease or a condition in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of the pharmaceutical compositions as described herein. Further disclosed herein, in some embodiments, are methods of treating a disease or a condition in a subject in need thereof, the method comprising administering to the subject a therapeutically effective amount of a pharmaceutical composition comprising (a) a cell produced according to the methods as described herein; and (b) a pharmaceutically acceptable carrier. [0626] In some embodiments, the disease or the condition is a cancer or a disease or a condition associated with expression of CD19, B-cell maturation antigen (BCMA), mesothelin (MSLN), CD20, CD70, MUC16, Trop-2, Nectin-4, or GPC3. In some embodiments, the cancer is a hematologic cancer. Examples of a hematologic cancer include, but are not limited to, B-cell acute lymphoid leukemia (B-ALL), T cell acute lymphoid leukemia (T-ALL), acute lymphoblastic leukemia (ALL), chronic myelogenous leukemia (CML), chronic lymphocytic leukemia (CLL), B cell prolymphocytic leukemia, blastic plasmacytoid dendritic cell neoplasm, Burkitt’s lymphoma, diffuse large B cell lymphoma, follicular lymphoma, hairy cell leukemia, small cell-follicular lymphoma, large cell-follicular lymphoma, malignant lymphoproliferative conditions, MALT lymphoma, mantle cell lymphoma, Marginal zone lymphoma, multiple myeloma, myelodysplasia, myelodysplastic syndrome, non-Hodgkin’s lymphoma, plasmablastic lymphoma, plasmacytoid dendritic cell neoplasm, Waldenstrom macroglobulinemia, and preleukemia. In some embodiments, the subject is a mammal. In some embodiments, the subject is a human. [0627] Disclosed herein, in some embodiments, are methods of increasing the activity or persistence of a cell expressing a recombinant nucleic acid molecule comprising a sequence encoding the TFP as described herein by expressing a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein in the cell. In some embodiments, the cell is any one of cells described herein.
[0628] Disclosed herein, in some embodiments, are methods of treating cancer in a subject in need thereof, the method comprising administering to the subject a therapeutically effective amount of the pharmaceutical compositions as described herein. Further disclosed herein, in some embodiments, are methods of treating cancer in a subject in need thereof, the method comprising administering to the subject a pharmaceutical composition comprising (a) a modified T cell produced according to the methods as described herein; and (b) a pharmaceutically acceptable carrier. [0629] In some instances, the modified T cell is an autologous T cell. In some embodiments, the T cell is an allogeneic T cell. In some instances, less cytokines are released in the subject compared a subject administered an effective amount of an unmodified control T cell. In some instances, less cytokines are released in the subject compared a subject administered an effective amount of a modified T cell comprising the recombinant nucleic acid as described herein, or the vector as described herein. [0630] In some instances, the method comprises administering the pharmaceutical composition as described herein in combination with an agent that increases the efficacy of the pharmaceutical composition. In some instances, the method comprises administering the pharmaceutical composition in combination with an agent that ameliorates one or more side effects associated with the pharmaceutical composition. [0631] In some instances, the cancer is a solid cancer, a lymphoma or a leukemia. In some instances, the cancer is selected from the group consisting of renal cell carcinoma, breast cancer, lung cancer, ovarian cancer, prostate cancer, colon cancer, cervical cancer, brain cancer, liver cancer, pancreatic cancer, kidney and stomach cancer. [0632] The present disclosure includes a type of cellular therapy where T cells are genetically modified to express a TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein and the modified T cell is infused to a recipient in need thereof. The infused cell is able to kill tumor cells in the recipient. Unlike antibody therapies, modified T cells are able to replicate in vivo resulting in long-term persistence that can lead to sustained tumor control. In various aspects, the T cells administered to the patient, or their progeny, persist in the patient for at least four months, five months, six months, seven months, eight months, nine months, ten months, eleven months, twelve months, thirteen months, fourteen month, fifteen months, sixteen months, seventeen months, eighteen months, nineteen months, twenty months, twenty-one months, twenty-two months, twenty-three months, two years, three years, four years, or five years after administration of the T cell to the patient.
[0633] The present disclosure also includes a type of cellular therapy where T cells are modified, e.g., by in vitro transcribed RNA, to transiently express a TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein and the modified T cell is infused to a recipient in need thereof. The infused cell is able to kill tumor cells in the recipient. Thus, in various aspects, the T cells administered to the patient, is present for less than one month, e.g., three weeks, two weeks, or one week, after administration of the T cell to the patient. [0634] Without wishing to be bound by any particular theory, the anti-tumor immunity response elicited by the modified T cells may be an active or a passive immune response, or alternatively may be due to a direct vs indirect immune response. [0635] In one aspect, the human modified T cells of the disclosure may be a type of vaccine for ex vivo immunization and/or in vivo therapy in a mammal. In one aspect, the mammal is a human. [0636] With respect to ex vivo immunization, at least one of the following occurs in vitro prior to administering the cell into a mammal: i) expansion of the cells, ii) introducing a nucleic acid encoding a TFP as described herein and a switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein to the cells or iii) cryopreservation of the cells. In some embodiments, a nucleic acid encoding a TCR gamma and/or delta constant domain is further introduced to the cells. [0637] Ex vivo procedures are well known in the art and are discussed more fully below. Briefly, cells are isolated from a mammal (e.g., a human) and genetically modified (i.e., transduced or transfected in vitro) with a vector as described herein. The modified T cell can be administered to a mammalian recipient to provide a therapeutic benefit. The mammalian recipient may be a human and the modified cell can be autologous with respect to the recipient. Alternatively, the cells can be allogeneic, syngeneic or xenogeneic with respect to the recipient. [0638] The procedure for ex vivo expansion of hematopoietic stem and progenitor cells is described in U.S. Pat. No.5,199,942, incorporated herein by reference, can be applied to the cells of the present disclosure. Other suitable methods are known in the art; therefore, the present disclosure is not limited to any particular method of ex vivo expansion of the cells. Briefly, ex vivo culture and expansion of immune cells, e.g., T cells, comprises: (1) collecting CD34+ hematopoietic stem and progenitor cells from a mammal from peripheral blood harvest or bone marrow explants; and (2) expanding such cells ex vivo. In addition to the
cellular growth factors described in U.S. Pat. No.5,199,942, other factors such as flt3-L, IL- 1, IL-3 and c-kit ligand, can be used for culturing and expansion of the cells. [0639] In addition to using a cell-based vaccine in terms of ex vivo immunization, the present disclosure also provides compositions and methods for in vivo immunization to elicit an immune response directed against an antigen in a patient. [0640] Generally, the cells activated and expanded as described herein may be utilized in the treatment and prevention of diseases that arise in individuals who are immunocompromised. [0641] The modified T cells of the present disclosure may be administered either alone, or as a pharmaceutical composition in combination with diluents and/or with other components such as IL-2 or other cytokines or cell populations. Combination Therapies [0642] A modified immune cells, e.g., T cell described herein may be used in combination with other known agents and therapies. Administered “in combination”, as used herein, means that two (or more) different treatments are delivered to the subject during the course of the subject’s affliction with the disorder, e.g., the two or more treatments are delivered after the subject has been diagnosed with the disorder and before the disorder has been cured or eliminated or treatment has ceased for other reasons. In some embodiments, the delivery of one treatment is still occurring when the delivery of the second begins, so that there is overlap in terms of administration. This is sometimes referred to herein as “simultaneous” or “concurrent delivery”. In other embodiments, the delivery of one treatment ends before the delivery of the other treatment begins. In some embodiments of either case, the treatment is more effective because of combined administration. For example, the second treatment is more effective, e.g., an equivalent effect is seen with less of the second treatment, or the second treatment reduces symptoms to a greater extent, than would be seen if the second treatment were administered in the absence of the first treatment or the analogous situation is seen with the first treatment. In some embodiments, delivery is such that the reduction in a symptom, or other parameter related to the disorder is greater than what would be observed with one treatment delivered in the absence of the other. The effect of the two treatments can be partially additive, wholly additive, or greater than additive. The delivery can be such that an effect of the first treatment delivered is still detectable when the second is delivered. [0643] In some embodiments, the “at least one additional therapeutic agent” includes a modified immune cell, e.g., T cell. Also provided are T cells that express multiple TFPs as described herein, which bind to the same or different target antigens, or same or different epitopes on the same target antigen, and multiple switch polypeptides comprising a TGFBr2
extracellular domain or a functional fragment thereof as described herein; T cells that express multiple TFPs as described herein, which bind to the same or different target antigens, or same or different epitopes on the same target antigen, and multiple dominant negative polypeptides comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein; and T cells that express multiple TFPs as described herein, which bind to the same or different target antigens, or same or different epitopes on the same target antigen, and multiple switch polypeptides comprising a TGFBr2 extracellular domain or a functional fragment thereof and multiple dominant negative polypeptides comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. Also provided are populations of T cells in which a first subset of T cells express a first TFP and a first switch polypeptide and/or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein, and a second subset of T cells express a second TFP and a second switch polypeptide and/or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. [0644] A modified immune cell, e.g., T cell, described herein and the at least one additional therapeutic agent can be administered simultaneously, in the same or in separate compositions, or sequentially. For sequential administration, the modified immune cell, e.g., T cell described herein can be administered first, and the additional agent can be administered second, or the order of administration can be reversed. [0645] In further aspects, a modified immune cell, e.g., T cell, as described herein may be used in a treatment regimen in combination with surgery, chemotherapy, radiation, immunosuppressive agents, such as cyclosporin, azathioprine, methotrexate, mycophenolate, and tacrolimus, antibodies, or other immunoablative agents such as alemtuzumab, anti-CD3 antibodies or other antibody therapies, cytoxin, fludarabine, cyclosporin, tacrolimus, rapamycin, mycophenolic acid, steroids, romidepsin, cytokines, and irradiation. peptide vaccine, such as that described in Izumoto et al., 2008 J. Neurosurg.108:963-971. [0646] In one embodiment, the subject can be administered an agent which reduces or ameliorates a side effect associated with the administration of a modified immune cell, e.g., T cell. Side effects associated with the administration of a modified T cell include but are not limited to cytokine release syndrome (CRS), and hemophagocytic lymphohistiocytosis (HLH), also termed Macrophage Activation Syndrome (MAS). Symptoms of CRS include high fevers, nausea, transient hypotension, hypoxia, and the like. Accordingly, the methods as described herein can comprise administering a modified T cell described herein to a subject and further administering an agent to manage elevated levels of a soluble factor resulting
from treatment with a modified T cell. In one embodiment, the soluble factor elevated in the subject is one or more of IFN-γ, TNFα, IL-2 and IL-6. Therefore, an agent administered to treat this side effect can be an agent that neutralizes one or more of these soluble factors. Such agents include, but are not limited to a steroid, an inhibitor of TNFα, and an inhibitor of IL-6. An example of a TNFα inhibitor is entanercept. An example of an IL-6 inhibitor is tocilizumab (toc). [0647] In one embodiment, the subject can be administered an agent which enhances the activity of a modified immune cell, e.g., T cell. For example, in one embodiment, the agent can be an agent which inhibits an inhibitory molecule. Inhibitory molecules, e.g., Programmed Death 1 (PD-1), can, in some embodiments, decrease the ability of a modified T cell to mount an immune effector response. Examples of inhibitory molecules include PD-1, PD-L1, CTLA4, TIM3, LAG3, VISTA, BTLA, TIGIT, LAIR1, CD160, and 2B4. Inhibition of an inhibitory molecule, e.g., by inhibition at the DNA, RNA or protein level, can optimize a modified T cell performance. In embodiments, an inhibitory nucleic acid, e.g., an inhibitory nucleic acid, e.g., a dsRNA, e.g., an siRNA or shRNA, can be used to inhibit expression of an inhibitory molecule in the TFP as described herein and the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein-expressing cell. In an embodiment the inhibitor is a shRNA. In an embodiment, the inhibitory molecule is inhibited within a modified T cell. In these embodiments, a dsRNA molecule that inhibits expression of the inhibitory molecule is linked to the nucleic acid that encodes a component, e.g., all of the components, of the TFP as described herein and/or the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In one embodiment, the inhibitor of an inhibitory signal can be, e.g., an antibody or antibody fragment that binds to an inhibitory molecule. For example, the agent can be an antibody or antibody fragment that binds to PD-1, PD-L1, PD-L2 or CTLA4 (e.g., ipilimumab (also referred to as MDX-010 and MDX-101 and marketed as Yervoy®; Bristol-Myers Squibb; tremelimumab (IgG2 monoclonal antibody available from Pfizer, formerly known as ticilimumab, CP-675,206)). In an embodiment, the agent is an antibody or antibody fragment that binds to TIM3. In an embodiment, the agent is an antibody or antibody fragment that binds to LAG3. [0648] In some embodiments, the agent which enhances the activity of a modified T cell can be, e.g., a fusion protein comprising a first domain and a second domain, wherein the first domain is an inhibitory molecule, or fragment thereof, and the second domain is a polypeptide that is associated with a positive signal, e.g., a polypeptide comprising an
intracellular signaling domain as described herein. In some embodiments, the polypeptide that is associated with a positive signal can include a costimulatory domain of CD28, CD27,
ICOS, e.g ., an intracellular signaling domain of CD28, CD27 and/or ICOS, and/or a primary signaling domain, e.g. , of CD3 zeta, e.g. , described herein. In one embodiment, the fusion protein is expressed by the same cell that expresses the TFP as described herein and the switch polypeptide or dominant negative polypeptide comprising a TGFBr2 extracellular domain or a functional fragment thereof as described herein. In another embodiment, the fusion protein is expressed by a cell, e.g. , a T cell that does not express an anti-TAA TFP as described herein and the switch polypeptide or dominant negative polypeptide comprising a
TGFBr2 extracellular domain or a functional fragment thereof as described herein.
EXAMPLES
[0649] The invention is further described in detail by reference to the following experimental examples. These examples are provided for purposes of illustration only, and are not intended to be limiting unless otherwise specified. Thus, the invention should in no way be construed as being limited to the following examples, but rather, should be construed to encompass any and all variations which become evident as a result of the teaching provided herein. Without further description, it is believed that one of ordinary skill in the art can, using the preceding description and the following illustrative examples, make and utilize the compounds of the present invention and practice the claimed methods. The following working examples specifically point out various aspects of the present invention, and are not to be construed as limiting in any way the remainder of the disclosure.
Background for Examples
[0650] T-Cell Receptor (TCR) is formed by a complex of dimers TCRα/β, CD3γ/ε, CD3δ/ε and the homodimer CD3ζ/ζ. In some particular T cells, TCRγ/δ are expressed instead of TCRα/β to form a functional TCR. TCRα/β/γ/δ have a constant domain common to all T- cells and a variable domain specific to an antigen. TRAC, TRBC, TRGC and TRDC genes encode for the constant C-terminal region of TCRα, TCRβ, TCRγ and TCRδ respectively. Despite high structural homology between those molecules, TCRa only pairs with TCRb and TCRγ only pairs with TCRδ. Hence, a TCR complex is formed with TCRα/β in α/β T cells or with TCRγ/δ in γ/δ T cells.
Example 1: TFP Constructs
[0651] TFP constructs are generated as previously described. An anti-MSLN binder is linked to a CD3 or TCR DNA fragment by either a DNA sequence encoding a short linker (SL): AAAGGGGSGGGGSGGGGSLE (SEQ ID NO:387) or a long linker (LL): AAAIEVMYPPPYLGGGGSGGGGSGGGGSLE (SEQ ID NO:388) into a lentiviral vector (pLRPO, pLRPC, pLCUS, or pLKaUS). In some embodiments, the TFP used is TC-210 (an anti-MSLN MH1e VHH antibody linked to CD3 epsilon) having the sequence of SEQ ID NO: 195. Source of TCR Subunits [0652] A TCR complex contains the CD3-epsilon polypeptide, the CD3-gamma poly peptide, the CD3-delta polypeptide, and the TCR alpha chain polypeptide and the TCR beta chain polypeptide or the TCR delta chain polypeptide and the TCR gamma chain polypeptide. TCR alpha, TCR beta, TCR gamma, and TCR delta recruit the CD3 zeta polypeptide. The human CD3-epsilon polypeptide canonical sequence is Uniprot Accession No. P07766. The human CD3-gamma polypeptide canonical sequence is Uniprot Accession No. P09693. The human CD3-delta polypeptide canonical sequence is Uniprot Accession No. P043234. The human CD3-zeta polypeptide canonical sequence is Uniprot Accession No. P20963. The human TCR alpha chain canonical sequence is Uniprot Accession No. Q6ISU1. The murine TCR alpha chain canonical sequence is Uniprot Accession No. A0A075B662. The human TCR beta chain constant region canonical sequence is Uniprot Accession No. P01850. The murine TCR beta chain constant region canonical sequence is Uniprot Accession No. P01852. [0653] The human CD3-epsilon polypeptide canonical sequence is: MQSGTHWRVLGLCLLSVGVWGQDGNEEMGGITQTPYKVSISGTTVILTCPQYPGSEI LWQHNDKNIGGDEDDKNIGSDEDHLSLKEFSELEQSGYYVCYPRGSKPEDANFYLY LRARVCENCMEMDVMSVATIVIVDICITGGLLLLVYYWSKNRKAKAKPVTRGAGAG GRQRGQNKERPPPVPNPDYEPIRKGQRDLYSGLNQRRI (SEQ ID NO:124). [0654] The mature human CD3-epsilon polypeptide sequence is: DGNEEMGGITQTPYKVSISGTTVILTCPQYPGSEILWQHNDKNIGGDEDDKNIGSDED HLSLKEFSELEQSGYYVCYPRGSKPEDANFYLYLRARVCENCMEMDVMSVATIVIV DICITGGLLLLVYYWSKNRKAKAKPVTRGAGAGGRQRGQNKERPPPVPNPDYEPIR KGQRDLYSGLNQRRI (SEQ ID NO:258). [0655] The signal peptide of human CD3ε is: MQSGTHWRVLGLCLLSVGVWGQ (SEQ ID NO:125).
[0656] The extracellular domain of human CD3ε is: DGNEEMGGITQTPYKVSISGTTVILTCPQYPGSEILWQHNDKNIGGDEDDKNIGSDED HLSLKEFSELEQSGYYVCYPRGSKPEDANFYLYLRARVCENCMEMD (SEQ ID NO:126). [0657] The transmembrane domain of human CD3ε is: VMSVATIVIVDICITGGLLLLVYYWS (SEQ ID NO:127). [0658] The intracellular domain of human CD3ε is: KNRKAKAKPVTRGAGAGGRQRGQNKERPPPVPNPDYEPIRKGQRDLYSGLNQRRI (SEQ ID NO:128). [0659] The human CD3-gamma polypeptide canonical sequence is: MEQGKGLAVLILAIILLQGTLAQSIKGNHLVKVYDYQEDGSVLLTCDAEAKNITWFK DGKMIGFLTEDKKKWNLGSNAKDPRGMYQCKGSQNKSKPLQVYYRMCQNCIELNA ATISGFLFAEIVSIFVLAVGVYFIAGQDGVRQSRASDKQTLLPNDQLYQPLKDREDDQ YSHLQGNQLRRN (SEQ ID NO:129). [0660] The mature human CD3-gamma polypeptide sequence is: QSIKGNHLVKVYDYQEDGSVLLTCDAEAKNITWFKDGKMIGFLTEDKKKWNLGSN AKDPRGMYQCKGSQNKSKPLQVYYRMCQNCIELNAATISGFLFAEIVSIFVLAVGVY FIAGQDGVRQSRASDKQTLLPNDQLYQPLKDREDDQYSHLQGNQLRRN (SEQ ID NO:130). [0661] The signal peptide of human CD3γ is: MEQGKGLAVLILAIILLQGTLA (SEQ ID NO:131). [0662] The extracellular domain of human CD3γ is: QSIKGNHLVKVYDYQEDGSVLLTCDAEAKNITWFKDGKMIGFLTEDKKKWNLGSN AKDPRGMYQCKGSQNKSKPLQVYYRMCQNCIELNAATIS (SEQ ID NO:132). [0663] The transmembrane domain of human CD3 γ is: GFLFAEIVSIFVLAVGVYFIA (SEQ ID NO:133). [0664] The intracellular domain of human CD3γ is: GQDGVRQSRASDKQTLLPNDQLYQPLKDREDDQYSHLQGNQLRRN (SEQ ID NO:134). [0665] The human CD3-delta polypeptide canonical sequence is: MEHSTFLSGLVLATLLSQVSPFKIPIEELEDRVFVNCNTSITWVEGTVGTLLSDITRLD LGKRILDPRGIYRCNGTDIYKDKESTVQVHYRMCQSCVELDPATVAGIIVTDVIATLL LALGVFCFAGHETGRLSGAADTQALLRNDQVYQPLRDRDDAQYSHLGGNWARNKS (SEQ ID NO:135).
[0666] The mature human CD3-delta polypeptide sequence is: FKIPIEELEDRVFVNCNTSITWVEGTVGTLLSDITRLDLGKRILDPRGIYRCNGTDIYK DKESTVQVHYRMCQSCVELDPATVAGIIVTDVIATLLLALGVFCFAGHETGRLSGAA DTQALLRNDQVYQPLRDRDDAQYSHLGGNWARNKS (SEQ ID NO:136). [0667] The signal peptide of human CD3δ is: MEHSTFLSGLVLATLLSQVSP (SEQ ID NO:137). [0668] The extracellular domain of human CD3δ is: FKIPIEELEDRVFVNCNTSITWVEGTVGTLLSDITRLDLGKRILDPRGIYRCNGTDIYK DKESTVQVHYRMCQSCVELDPATVA (SEQ ID NO:138). [0669] The transmembrane domain of human CD3δ is: GIIVTDVIATLLLALGVFCFA (SEQ ID NO:139). [0670] The intracellular domain of human CD3δ is: GHETGRLSGAADTQALLRNDQVYQPLRDRDDAQYSHLGGNWARNK (SEQ ID NO:140). [0671] The human CD3-zeta polypeptide canonical sequence is: MKWKALFTAAILQAQLPITEAQSFGLLDPKLCYLLDGILFIYGVILTALFLRVKFSRSA DAPAYQQGQNQLYNELNLGRREEYDVLDKRRGRDPEMGGKPQRRKNPQEGLYNEL QKDKMAEAYSEIGMKGERRRGKGHDGLYQGLSTATKDTYDALHMQALPPR (SEQ ID NO:141). [0672] The human TCR alpha chain constant region canonical sequence is: IQNPDPAVYQLRDSKSSDKSVCLFTDFDSQTNVSQSKDSDVYITDKTVLDMRSMDFK SNSAVAWSNKSDFACANAFNNSIIPEDTFFPSPESSCDVKLVEKSFETDTNLNFQNLS VIGFRILLLKVAGFNLLMTLRLWSS (SEQ ID NO:142). [0673] The human TCR alpha chain human IgC sequence is: IQNPDPAVYQLRDSKSSDKSVCLFTDFDSQTNVSQSKDSDVYITDKTVLDMRSMDFK SNSAVAWSNKSDFACANAFNNSIIPEDTFFPSPESSCDVKLVEKSFETDTNLNFQNLS (SEQ ID NO:143). [0674] The transmembrane domain of the human TCR alpha chain is: VIGFRILLLKVAGFNLLMTLRLW (SEQ ID NO:144). [0675] The intracellular domain of the human TCR alpha chain is: SS (SEQ ID NO:145). [0676] The murine TCR alpha chain constant (mTRAC) region canonical sequence is: XIQNPEPAVYQLKDPRSQDSTLCLFTDFDSQINVPKTMESGTFITDKTVLDMKAMDS KSNGAIAWSNQTSFTCQDIFKETNATYPSSDVPCDATLTEKSFETDMNLNFQNLSVM GLRILLLKVAGFNLLMTLRLWSS (SEQ ID NO:146).
[0677] The transmembrane domain of the murine TCR alpha chain is: MGLRILLLKVAGFNLLMTLRLW (SEQ ID NO:147). [0678] The intracellular domain of the murine TCR alpha chain is: SS (SEQ ID NO: 405). [0679] The human TCR beta chain constant region 1 (hTRBC1) canonical sequence is: EDLNKVFPPEVAVFEPSEAEISHTQKATLVCLATGFFPDHVELSWWVNGKEVHSGVS TDPQPLKEQPALNDSRYCLSSRLRVSATFWQNPRNHFRCQVQFYGLSENDEWTQDR AKPVTQIVSAEAWGRADCGFTSVSYQQGVLSATILYEILLGKATLYAVLVSALVLMA MVKRKDF (SEQ ID NO:148). [0680] The human TCR beta chain 1 human IgC sequence is: EDLNKVFPPEVAVFEPSEAEISHTQKATLVCLATGFFPDHVELSWWVNGKEVHSGVS TDPQPLKEQPALNDSRYCLSSRLRVSATFWQNPRNHFRCQVQFYGLSENDEWTQDR AKPVTQIVSAEAWGRADCGFTSVSYQQGVLSATILYE (SEQ ID NO: 149). [0681] The transmembrane domain of the human TCR beta chain 1 is: ILLGKATLYAVLVSALVLMAM (SEQ ID NO:150). [0682] The intracellular domain of the human TCR beta chain 1 is: VKRKDF (SEQ ID NO: 151). [0683] The human TCR beta chain 2 constant region (hTRBC2) canonical sequence is: DLKNVFPPKVAVFEPSEAEISHTQKATLVCLATGFYPDHVELSWWVNGKEVHSGVS TDPQPLKEQPALNDSRYCLSSRLRVSATFWQNPRNHFRCQVQFYGLSENDEWTQDR AKPVTQIVSAEAWGRADCGFTSESYQQGVLSATILYEILLGKATLYAVLVSALVLMA MVKRKDSRG (SEQ ID NO:371). [0684] The murine TCR beta chain constant region 1 canonical sequence is: EDLRNVTPPKVSLFEPSKAEIANKQKATLVCLARGFFPDHVELSWWVNGKEVHSGV STDPQAYKESNYSYCLSSRLRVSATFWHNPRNHFRCQVQFHGLSEEDKWPEGSPKP VTQNISAEAWGRADCGITSASYQQGVLSATILYEILLGKATLYAVLVSTLVVMAMV KRKNS (SEQ ID NO:152). [0685] The transmembrane domain of the murine TCR beta chain 1 is: ILYEILLGKATLYAVLVS TLVVMAMVK (SEQ ID NO:153). [0686] The intracellular domain of the murine TCR beta chain 1 is: KRKNS (SEQ ID NO:154). [0687] The murine TCR beta chain constant 2 region canonical sequence is: XDLRNVTPPKVSLFEPSKAEIANKQKATLVCLARGFFPDHVELSWWVNGKEVHSGV STDPQAYKESNYSYCLSSRLRVSATFWHNPRNHFRCQVQFHGLSEEDKWPEGSPKP VTQNISAEAWGRADCGITSASYHQGVLSATILYEILLGKATLYAVLVSGLVLMAMV KKKNS (SEQ ID NO:282).
[0688] The human TCR gamma chain constant region canonical sequence is: DKQLDADVSPKPTIFLPSIAETKLQKAGTYLCLLEKFFPDVIKIHWQEKKSNTILGSQE GNTMKTNDTYMKFSWLTVPEKSLDKEHRCIVRHENNKNGVDQEIIFPPIKTDVITMD PKDNCSKDANDTLLLQLTNTSAYYMYLLLLLKSVVYFAIITCCLLRRTAFCCNGEKS (SEQ ID NO:21). [0689] The human TCR gamma human IgC sequence is: DKQLDADVSPKPTIFLPSIAETKLQKAGTYLCLLEKFFPDVIKIHWQEKKSNTILGSQE GNTMKTNDTYMKFSWLTVPEKSLDKEHRCIVRHENNKNGVDQEIIFPPIKTDVITMD PKDNCSKDANDTLLLQLTNTSA (SEQ ID NO: 155). [0690] The transmembrane domain of the human TCR gamma chain is: YYMYLLLLLKSVVYFAIITCCLL (SEQ ID NO:156). [0691] The intracellular domain of the human TCR gamma chain is: RRTAFCCNGEKS (SEQ ID NO: 157). [0692] The human TCR delta chain C region canonical sequence is: SQPHTKPSVFVMKNGTNVACLVKEFYPKDIRINLVSSKKITEFDPAIVISPSGKYNAV KLGKYEDSNSVTCSVQHDNKTVHSTDFEVKTDSTDHVKPKETENTKQPSKSCHKPK AIVHTEKVNMMSLTVLGLRMLFAKTVAVNFLLTAKLFFL (SEQ ID NO:243). [0693] The human TCR delta human IgC sequence is: SQPHTKPSVFVMKNGTNVACLVKEFYPKDIRINLVSSKKITEFDPAIVISPSGKYNAV KLGKYEDSNSVTCSVQHDNKTVHSTDFEVKTDSTDHVKPKETENTKQPSKSCHKPK AIVHTEKVNMMSLTV (SEQ ID NO: 265). [0694] The transmembrane domain of the human TCR delta chain is: LGLRMLFAKTVAVNFLLTAKLFF (SEQ ID NO:158). [0695] The intracellular domain of the human TCR delta chain is: L. Dominant Negative TGFβRII [0696] In some embodiments, TFP constructs are in a vector that further contains a sequence encoding a dominant negative TGFβRII. The dominant negative TGFβRII may be encoded in the same open reading frame and separated by a self-cleaving peptide (e.g., a P2A or a T2A self-cleaving peptide). In some embodiments, the dominant negative TGFβRII comprises the extracellular domain of TGFβRII and lacks a functional kinase domain. In some embodiments, the dominant negative TGFβRII comprises a TGFβRII extracellular domain comprising SEQ ID NO: 271 and a TGFβRII transmembrane domain comprising SEQ ID NO: 272. In some embodiments, the dominant negative TGFβRII comprises a TGFβRII extracellular domain, a TGFβRII transmembrane domain, and a portion of a
TGFβRII intracellular domain, wherein the portion of the intracellular domain is not capable of kinase activity. In some embodiments, the dominant negative TGFβRII comprises SEQ ID NO: 68. TGFβRII switches [0697] In some embodiments, TFP constructs are in a vector that further contains a sequence encoding a TGFβRII switch. The TGFβRII switch may be encoded in the same open reading frame and separated by a self-cleaving peptide (e.g., a P2AW or a T2A self- cleaving peptide). In some embodiments, the TGFβRII comprises the extracellular domain of TGFβRII fused to the intracellular domain of CD28. In some embodiments, the transmembrane domain is from TGFβRII and the switch comprises SEQ ID NO:273. In some embodiments, the transmembrane domain is from CD28 and the switch comprises SEQ ID NO:273. In some embodiments, the TGFβRII comprises the extracellular domain of TGFβRII fused to the intracellular domain of 4-1BB. In some embodiments, the transmembrane domain is from TGFβRII and the switch comprises SEQ ID NO:277. In some embodiments, the transmembrane domain is from 4-1BB and the switch comprises SEQ ID NO:277. [0698] Exemplary constructs used in this study are shown in Table 1 below: Table 1: Constructs
TFP Expression Vectors [0699] Expression vectors are provided that include: a promoter (e.g., an EF1a promoter), a signal sequence to enable secretion, a polyadenylation signal and transcription terminator
(Bovine Growth Hormone (BGH) gene), an element allowing episomal replication and replication in prokaryotes (e.g., SV40 origin and ColE1 or others known in the art) and elements to allow selection (ampicillin resistance gene and zeocin marker). [0700] The TFP-encoding nucleic acid constructs shown above were cloned into a lentiviral expression vector. Example 2: Generation of T cell receptor fusion protein T Cells T-cell activation, transduction, and expansion [0701] T cells were purified from healthy donor leukopak or PBMCs via positive selection of CD4+ and CD8+ T cells with CD4 and CD8 microbeads from Miltenyi Biotech. On day 0, T cells, freshly isolated or thawed from previously prepared frozen vials, were activated by MACS GMP T cell TransAct (Miltenyi Biotech), in the presence of human IL-7 and IL-15 (both from Miltenyi Biotech, premium grade). On day 1, activated T cells were transduced with lentivirus encoding the TFP. T cells were transduced with vectors expressing anti- MSLN TFP (referred to as MH1e, MH1e TFP, or TC-210), or the anti-MSLN TFP expressing the TGFβRII switch (or PD-1 switch) described above. On day 4, the cells were washed, subcultured in fresh medium with cytokines and then expanded up to day 10 by supplementing fresh medium every 2 days. At each day of subculture, fresh medium with cytokines were added to maintain the cell suspension at 0.5x106 cells/mL. The expansion of T cells, and non-transduced controls, is shown in Figure 1 for three donors (Donors R020, R021, and R022). No dramatic difference in expansion was observed between donors or between transduced and non-transduced T cells. Verification of TFP expression and phenotyping of TFP-expressing cells by cell staining [0702] Following lentiviral transduction, expression of TFPs by transduced T cells was confirmed by flow cytometry, using an anti-VHH antibody, on day 10 of cell expansion. As is shown in Figure 2, binding of the anti-VHH antibody was detected TFP-transduced T cells, but not in non-transduced control T cells from the same donors. All TFP transduced cells showed at least 40% transduction efficiency. The CD4:CD8 ratio of transduced cells was also determined by flow cytometry. As is shown in Figure 3, although the ratio differed between donors, there was no significant difference in CD4:CD8 ratio between the different TFP constructs (with or without a TGFβRII or PD-1 switch). The memory status of the T cells was determined by flow cytometry to detect cell surface levels of CD45RA and CCR7 as is shown in Figure 4. Again, while the memory status differed between donors, there was no significant difference in memory status between the different TFP constructs (with or without
a TGFβRII or PD-1 switch). Figure 5 shows detection of TGFβRII on the surface of TFP expressing cells. Little TGFβRII is detected on the surface of TC-210 or MH1e + PD-1 switch transduced cells, while TGFβRII is detected on the surface of cells transduced with constructs containing TGFβRII switches. The TGFβRII switch having the TGFβRII extracellular and transmembrane domains with the CD28 intracellular domain is expressed at the highest level while the TGFβRII switch having the TGFβRII extracellular and transmembrane domains with the 4-1BB transmembrane domain is expressed at low levels. Example 3: Plate bound stimulation assay [0703] TFP-expressing T cells expressing the constructs shown in Table 1 were expanded to day 10, as described above.2 x 105 TFP transduced T cells or untransduced control T cells in 200uL of R10 media were then added to one of four plates for 24 hours. The plates contained: (i) PBS control; (ii) Dynabeads at a 1:1 ratio with T cells (Positive Stimulation); (iii) 1ug/mL plate bound MSLN; or (iv) 1ug/mL plate bound MSLN with plate bound 5ug/mL PDL1-Fc.0, 1, 5, 25, or 200 ng/mL of TGFβ was added to wells. Cytokine production was then measured after 24 hours [0704] Representative data for Donor R022 is shown in Figures 6-8. Figure 6 shows cytokine production in cells contacted with plate bound mesothelin in the presence or absence of 25 ng/mL TGFβ. For all cytokines (IFN-γ, IL-2, TNFα, and GM-CSF), addition of TGFβ inhibited cytokine production in cells expressing TC-210 or MH1e TFPs with the PD-1 switch. Expression of TGFβRII switches rescued cytokine production in MH1e expressing cells, with the TGFβRII switch having the TGFβRII transmembrane domain and the CD28 intracellular domain having the greatest effect. [0705] Figure 7 shows IL-2 and IFN-γ production in TFP-expressing cells or non- transduced cells contacted with plate bound mesothelin in the presence of TGFβ across a range of TGFβ. The data show that even 1 ng/mL of TGFβ is sufficient to suppress cytokine production in TC-210 expressing cells and MH1e PD-1 switch expressing cells, and that increasing levels of TGFβ do not further suppress cytokine production. However, increasing levels of TGFβ increase the level of cytokines produced in cells expressing TGFβRII switches. [0706] Figure 8 shows cytokine production in TFP expressing cells contacted with plate bound mesothelin and PD-L1 in the presence or absence of 25 ng/mL TGFβ. For all cytokines (IFN-γ, IL-2, TNFα, and GM-CSF), the addition of PD-L1 reduced cytokine levels, except in cells expressing MH1e and the PD-1 switch, regardless of the presence or absence of TGFβ. TGFβ inhibited cytokine production in PD-1 switch expressing TFP T cells. The
expression of TGFβRII switches was not able to overcome PD-L1 mediated repression of cytokine expression. Example 4: Co-culture assay [0707] TFP-expressing T cells expanded to day 10, as described above, were contacted with MLSN-expressing MSTO-mlsn target cells, MSLN and TGFβ-expressing OVCAR3 target cells, or C30 control cells, which do not express mesothelin, at a 1:1 ratio in 200uL TexMACs media in a 96 well flat bottom plate with or without added TGFβ.24 hours after initiation of co-culture 100uL of supernatant was collect and frozen for cytokine analysis and 100uL of new media was added.72 hours after initiation of co-culture 100uL of supernatant was collect and frozen for cytokine analysis. [0708] Target-specific cytokine production including IFN-γ, IL-2, TNFα, and GM-CSF by TFP T cells was measured from supernatants using the U-PLEX® Biomarker Group I (hu) Assays (Meso Scale Diagnostics®, LLC, catalog number: K15067L-4). A schematic of the experimental setup is shown in Figure 9. [0709] IL-2 production for Donors R020 is shown in Figures 10 and 11. [0710] Figure 10 shows IL-2 production in cells contacted with MSTO-msln, OVCAR3, or C30 cells in the presence or absence of 10 ng/mL TGFβ. With msto-MSLN target cells, the addition of TGFβ inhibited cytokine production in cells expressing TC-210 or MH1e TFPs with the PD-1 switch. Expression of TGFβRII switches rescued cytokine production in MH1e expressing cells when contacted with TGFβ, with the TGFβRII switch having the TGFβRII transmembrane domain and the CD28 intracellular domain having the greatest effect. With OVCAR3 target cells, cytokine production is low in cells expressing TC-210 or MH1e TFPs with the PD-1 switch, likely due to the production of TGFβ of the OVCAR3 cells. Expression of TGFβRII switches increased cytokine production in MH1e expressing cells, with the TGFβRII switch having the TGFβRII transmembrane domain and the CD28 intracellular domain having the greatest effect. When exogenous TGFβ was added, cytokine production by cells expressing TGFβRII switches further increased. Very low cytokine levels were observed in all TFP-expressing cells contacted with C30 control target cells. [0711] Figure 11 shows IFN-γ, IL-2, TNFα, and GM-CSF production in TFP-expressing cells or non-transduced cells contacted with contacted with MSTO-msln or OVCAR3 cells and TGFβ across a range of TGFβ concentrations (0.1, 1, 10, or 25 ng/mL). The data show that even 0.1 ng/mL of TGFβ is sufficient to suppress cytokine production in TC-210 expressing cells and MH1e PD-1 switch expressing cells, and that increasing levels of TGFβ do not further suppress cytokine production. However, increasing levels of TGFβ increase the
level of cytokines produced in cells expressing TGFβRII switches, having the greatest impact on IL-2 production. Example 5: pSMAD2/3 staining of TGFβRII switch expressing TFP T cells [0712] TFP-expressing T cells from Donor 22 expressing the constructs shown in Table 1 were expanded to day 10, as described above. Cells were suspended at concentration of 9 x 106 cell/mL in R10 media with 100U/mL IL-2. After 24 hours (on day 2), cells were washed 4x with 15mL of Serum Free Media and resuspended at a concentration of 1 x 105 cells in 100uL of serum-free media. On day 3, 200ng/mL, 10ng/mL, or 0.1ng/mL of TGFβ was added. Cells were then fixed with BD 1x Fixation Buffer and resuspended in BD Buffer III. Cells were then stained for CD4, CD8, pSMAD2/3, and VHH (MH1e expression). A schematic illustrating the experimental protocol is shown in Figure 12. [0713] The phenotype of the day 10 T cells used (prior to resuspension in R10 media with IL-2) was determined as described in Example 2 and results are shown in Figure 13. Transduction efficiency (VHH levels), PD-1, and TGFβRII were measured. High transduction efficiency was observed with all TFP constructs. PD-1 was detected on the surface of cells transduced with the vector encoding MH1e TFP and the PD-1 switch. TGFβRII was detected on the surface of cells transduced with the vector encoding MH1e TFP and each of the TGFβRII switches. [0714] The levels of pSMAD2/3, as determined by flow cytometry after fixation, are shown in Figure 14. In the absence of TGFβ, all TFP+ cells (VHH+) had similar levels of pSMAD2/3. At increasing levels of TGFβ, the levels of pSMAD2/3 were shown to increase in TC-210 cells and in cells containing MH1e with the PD-1 switch. Expression of the TGFβRII switches suppressed this increase in pSMAD2/3, with the switch having the TGFβRII extracellular and transmembrane domains and the CD28 intracellular domain being particularly effective. pSMAD2/3 also increased in non-transduced cells and in TFP- cells from the cell populations transduced with TC-210 or MH1e with the PD-1 switch at increasing TGFβ concentrations. TFP- cells from the cell populations transduced with the TGFβRII switches also showed a suppression of this increase in pSMAD2/3, indicating that the TGFβRII switches on TFP+ cells may act as a dominant negative receptor, also affecting pSMAD2/3 levels in TFP- cells. Figure 15 shows pSMAD2/3 levels in non-transduced CD4+ and CD8+ T cells, indicating that there is no significant difference in pSMAD2/3 signaling between CD4 and CD8+ T cells Example 6. Phenotype of cells expressing TGFβRII Switch or DN TGFβRII TFP
[0715] T cells were purified from healthy donors, activated, and transduced with lentivirus encoding a TFP as described above. In this study, T cells were transduced with vectors encoding an anti-MSLN TFP (TC-210); anti-MSLN TFP with PD-CD28 Switch (TC-510); anti-MSLN TFP and a TGFβRII Switch (41BB(IC) or CD28(IC)); anti-MSLN TFP and a full length TGFβRII (FL); or anti-MSLN TFP and a dominant negative TGFβRII lacking the functional kinase domain (DN). Non-transduced T cells were also included as a control (NT). Exemplary constructs used in the study are shown below in Table 2. Table 2: Constructs
[0716] Expression of TFPs by transduced T cells was confirmed by flow cytometry, using an anti-VHH antibody. As shown in FIG.16A, binding of the anti-VHH antibody was detected in each of the TFP-transduced T cell groups, but not in non-transduced control T cells. The T cells transduced with a TGFβRII Switch, DN, or FL each exhibited similar VHH expression compared to TC-210 and TC-510 (FIG.16A). In the cells transduced with the TGFβRII Switch, DN, and FL, VHH+ cells expressed high levels of TGFβRII (FIG.16B). The highest levels of surface TGFβRII were detected on the cells transduced with the DN construct; little TGFβRII was detected on the surface of TC-210 or TC-510 transduced cells. [0717] The CD4 and CD8 memory phenotype was determined by detecting surface levels of CD45RA and CCR7 by flow cytometry. CD4 and CD8 T cell memory phenotypes were similar among the groups, as shown in FIG.17 and FIG 18, respectively. The CD4:CD8 ratio of transduced cells was also determined by flow cytometry. As shown in FIG.19, there
were no significant differences in CD4:CD8 ratio among the T cells transduced with vectors encoding the different constructs. Example 7. Functional assessment of cells transduced with TFP-TGFβRII Switch or TFP-DN TGFβRII [0718] In this study, T cell cytotoxicity against tumor cell lines, cytokine production, capacity for TGFβ signaling, and ability to overcome TGFβ-mediated suppression were assessed in the T cells transduced with vectors encoding the constructs shown above in Table 2. [0719] To compare cytotoxicity of the TGFβRII Switch and TGFβRII DN cells to TC- 210 and TC-510, transduced cells were incubated with a tumor cell line at effector (transduced T cells) to target (tumor cell line) ratios (E:T) of 9:1, 3:1, or 1:1. After 24 hours, a cytotoxicity assay was carried out to determine % tumor lysis, and supernatants were collected to measure cytokine production. Target tumor cell lines included MSTO-MSLN (high MSLN expression), Suit-2 (low MSLN expression), and control C30 target cells (no MSLN expression). Prior to incubation with the tumor cell lines, transduced cells were normalized to 25-40% transduction. [0720] As shown in FIG.20, none of the transduced cells exhibited cytotoxicity against control C30 cells. In MSTO-MSLN and Suit-2 cells, the TGFβRII Switch, TGFβRII DN, and TGFβRII FL transduced cells were similarly cytotoxic when compared to TC-210 and TC- 510, as measured by % tumor lysis in vitro after the 24 hour incubation (FIG.20). TGFβRII Switch and TGFβRII DN transduced cells exhibited higher IFNγ, IL-2, GM-CSF, and TNFα production compared to TC-210 transduced cells after incubation with MSTO-MSLN or Suit- 2 cell lines, as shown in FIGs.21A and 21B. [0721] pSMAD2/3 levels were determined to assess levels of TGFβ signaling in transduced T cells. The T cells were plated at 2 x 106 cells/mL in R10 in the presence of 100U/mL IL-2, washed three times, resuspended in media without serum, and plated with 100 ng/mL, 10 ng/mL, 1 ng/mL, 0.1 ng/mL, or 0.01 ng/mL of TGFβ for 30 minutes. Cells were then fixed and stained for expression of CD4, CD8, VHH, and pSMAD2/3, and analyzed by flow cytometry. Representative histograms are provided in FIG.22 and show reduced pSMAD2/3 staining in VHH+ cells upon addition of TGFβ in the TGFβRII Switch and TGFβRII DN transduced cells, compared to TC-210 and TC-510 transduced cells. pSMAD2/3 MFI in VHH+ and VHH- cells at increasing concentrations of TGFβ are provided in FIG.23. TGFβRII Switch (41BB(IC) and CD28(IC)) and TGFβRII DN expressing cells exhibited significantly reduced TGFβ signaling compared to TC-210, TC-
510, and FL cells. Particularly at high TGFβ concentrations, the TGFβRII Switch and TGFβRII DN constructs acted as potent dominant negative receptors for TGFβ signaling (FIG.23). [0722] To assess the ability of the TGFβRII Switch and TGFβRII DN cells to overcome TGFβ-induced suppression of T cell function, 1 x 105 cells were normalized to 40% VHH+ and co-cultured with 1 x 105 tumor cells in a 200µL volume in R10 for 24 hours, with or without exogenous TGFβ. The cells transduced with TGFβRII Switch and TGFβRII DN constructs maintained cytokine production upon addition of exogenous TGFβ (FIGs.24A and 24B) or when cultured with Suit2, a TGFβ secreting cell line (FIG.25). As shown in FIG.26, TGFβRII Switch and TGFβRII DN constructs also maintained cytokine production upon addition of increasing levels of exogenous TGFβ in the Suit2 culture. [0723] In another study, 2 x 105 T cells were normalized to 25% VHH+ and placed in a 96 well plate coated with 1ng/mL of recombinant mesothelin in R10, with or without exogenous TGFβ. Twenty-four hours after initiation of culture, 100µL of supernatant was collected for analysis of cytokine production. As shown in FIGs.27A and 27B, TGFβRII Switch and TGFβRII DN constructs blunted the TGFβ-mediated suppression of MSLN cytokine production, in cells from two different donors. Moreover, higher levels of exogenous TGFβ increased cytokine production in both TGFβRII Switch and TGFβRII DN cells, compared to TC-210, TC-510, and TGFβRII FL cells (FIGs.28A and 28B). [0724] The effects of repeated stimulation of TGFβRII Switch and TGFβRII DN with the TGFβ secreting cell line Suit2 were analyzed.2.7 x 104 cells were normalized to 27% VHH+ and co-cultured with 2.7 x 104 Suit2 tumor cells in a 200µL volume in R10. Four days after initiation of culture, cells and 100µL of supernatant were collected from a first group of plates, for flow cytometry and cytokine analyses. In remaining plates, 100µL of media was removed and 100uL of new tumor cells were added. The process was repeated on days 8, 12, and 16. [0725] Both TGFβRII Switch and TGFβRII DN cells increased in cell number upon repeated stimulation with the TGFβ-secreting cell line Suit2 (FIG.29). In contrast, TC-210 and TC-510 cells did not grow in the presence of Suit2 cells (FIG.29). In addition, both TGFβRII Switch and TGFβRII DN cells expressed cytokines upon co-culture with Suit2 cells (FIG.30). In particular, TGFβRII Switch and TGFβRII DN cells expressed high amounts of IFNγ and GM-CSF on days 8, 12, and 16, in contrast to TC-210, TC-510, and TGFβRII FL cells, which expressed little to no cytokine at those time points (FIG.30). [0726] Together, the studies showed that T cells expressing the TGFβRII Switch or TGFβRII DN constructs express extracellular TGFβRII, exhibit high cytotoxicity and
cytokine production in response to tumor cells, have dominant negative TGFβRII signaling activity, produce higher levels of cytokines function in the presence of TGFβ, and expand upon repeated stimulation with a TGFβ secreting cell line. The TGFβRII DN constructs in particular resulted in high expression of extracellular TGFBRII. Example 8. In vivo study of TGFβRII Switch and TGFβRII DN efficacy [0727] An in vivo study is conducted to assess efficacy of TGFβRII Switch and TGFβRII DN cells in a tumor cell model. NSG mice are implanted at Day -10 with 1 x 106 Suit2-luc cells. When tumor size reaches 80-150mm3, transduced T cells are injected. Exemplary cells tested in the study include TC-210, TGFβRII DN, and non-transduced (NT) control cells. Tumors are measured every 3-4 days to determine anti-tumor efficacy. Persistence of the cells is tracked by weekly bleeds, and blood, tumor, liver, and spleen are collected at the end of the study (e.g., Day 12) to assess tumor infiltration. Table 3. Antigen binding domain sequences.
OTHER EMBODIMENTS
[0728] The disclosure set forth above may encompass multiple distinct inventions with independent utility. Although each of these inventions has been disclosed in its preferred form(s), the specific embodiments thereof as disclosed and illustrated herein are not to be considered in a limiting sense, because numerous variations are possible. The subject matter of the inventions includes all novel and nonobvious combinations and subcombinations of the various elements, features, functions, and/or properties as described herein. The following claims particularly point out certain combinations and subcombinations regarded as novel and nonobvious. Inventions embodied in other combinations and subcombinations of features, functions, elements, and/or properties may be claimed in this application, in applications claiming priority from this application, or in related applications. Such claims, whether directed to a different invention or to the same invention, and whether broader, narrower, equal, or different in scope in comparison to the original claims, also are regarded as included within the subject matter of the inventions of the present disclosure.
Claims
CLAIMS WHAT IS CLAIMED IS: 1. A recombinant nucleic acid comprising: a first nucleic acid sequence encoding a T cell receptor (TCR) fusion protein (TFP) wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain; and wherein the TCR subunit and the antigen binding domain are operatively linked, and a second nucleic acid sequence encoding a polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof.
2. The recombinant nucleic acid of claim 1, wherein the TCR subunit further comprises a TCR intracellular domain.
3. The recombinant nucleic acid of claim 1 or 2, wherein the first nucleic acid sequence and the second nucleic acid sequence are operatively linked by a linker.
4. The recombinant nucleic acid of claim 3, wherein the linker comprises a protease cleavage site.
5. The recombinant nucleic acid of claim 4, wherein the protease cleavage site is a 2A cleavage site.
6. The recombinant nucleic acid of claim 5, wherein the 2A cleavage site is a T2A cleavage site or a P2A cleavage site.
7. The recombinant nucleic acid of any one of claims 1-6, wherein the first nucleic acid sequence and the second nucleic acid sequence are present on different nucleic acid molecules.
8. The recombinant nucleic acid of any one of claims 1-7, wherein the TGFBr2 extracellular domain comprises a sequence with at least 80% sequence identity to SEQ ID NO:271.
9. The recombinant nucleic acid of any one of claims 1-8, wherein the TGFBr2 extracellular domain comprises a sequence according to SEQ ID NO: 271.
10. The recombinant nucleic acid of any one of claims 1-9, wherein polypeptide comprising a TGFBr2 extracellular domain is a dominant negative TGFBr2.
11. The recombinant nucleic acid of claim 10, wherein the dominant negative TGFBr2 comprises a sequence with at least 80% identity to SEQ ID NO: 271 and a sequence with at least 80% sequence identity to SEQ ID NO: 272.
12. The recombinant nucleic acid of claim 10 or 11, wherein the dominant negative TGFBr2 comprises a sequence according to SEQ ID NO: 271 and a sequence according to SEQ ID NO: 272.
13. The recombinant nucleic acid of any one of claims 10-12, wherein the dominant negative TGFBr2 comprises a truncated portion of a TGFBr2 intracellular domain.
14. The recombinant nucleic acid of any one of claims 10-13, wherein the dominant negative TGFBr2 comprises a sequence with at least 80% sequence identity to SEQ ID NO: 68.
15. The recombinant nucleic acid of any one of claims 10-14, wherein the dominant negative TGFBr2 comprises a sequence according to SEQ ID NO: 68.
16. The recombinant nucleic acid of any one of claims 1-9, wherein the polypeptide comprising a TGFBr2 extracellular domain or fragment thereof is a switch polypeptide.
17. The recombinant nucleic acid of claim 16, wherein the switch polypeptide further comprises a switch intracellular domain.
18. The recombinant nucleic acid of claim 17, wherein the TGFBr2 extracellular domain is operably linked to the switch intracellular domain.
19. The recombinant nucleic acid of claim 17 or 18, wherein the switch intracellular domain comprises an intracellular domain of a costimulatory polypeptide.
20. The recombinant nucleic acid of claim 19, wherein the costimulatory polypeptide is selected from the group consisting of CD28, 4-1BB, IL-15Ra, IL12R, IL18R, IL21R, OX40, CD2, CD27, CD5, ICAM-1, ICOS (CD278), GITR, CD30, CD40, BAFFR, HVEM, CD7, LIGHT, NKG2C, SLAMF7, NKp80, CD160, CD226, FcγRI, FcγRII, and FcγRIII.
21. The recombinant nucleic acid of claim 20, wherein the costimulatory polypeptide is CD28.
22. The recombinant nucleic acid of claim 20, wherein the costimulatory polypeptide is 4- 1BB.
23. The recombinant nucleic acid of claim 20, wherein the costimulatory polypeptide is IL-15Ra.
24. The recombinant nucleic acid of claim 20, wherein the switch intracellular domain comprises a sequence with at least 80% sequence identity to SEQ ID NO:273 or SEQ ID NO:277.
25. The recombinant nucleic acid of any one of claims 16-24, wherein the switch polypeptide further comprises a switch transmembrane domain.
26. The recombinant nucleic acid of claim 25, wherein the TGFBr2 extracellular domain is operably linked to the switch intracellular domain via the switch transmembrane domain.
27. The recombinant nucleic acid of claim 25 or 26, wherein the switch transmembrane domain is derived from a TGFBr2 transmembrane domain.
28. The recombinant nucleic acid of claim 26, wherein the switch transmembrane domain comprises a sequence with at least 80% sequence identity to SEQ ID NO:272.
29. The recombinant nucleic acid of claim 25 or 26, wherein the switch transmembrane domain is derived from a transmembrane domain of the costimulatory polypeptide.
30. The recombinant nucleic acid of claim 29, wherein the switch transmembrane domain is derived from a transmembrane domain of CD28.
31. The recombinant nucleic acid of claim 29, wherein the switch transmembrane domain is derived from a transmembrane domain of 4-1BB.
32. The recombinant nucleic acid of claim 29, wherein the switch transmembrane domain is derived from a transmembrane domain of IL-15Ra.
33. The recombinant nucleic acid of claim 29, wherein the switch transmembrane domain comprises a sequence with at least 80% sequence identity to SEQ ID NO:275 or SEQ ID NO:279.
34. The recombinant nucleic acid of any one of claims 16-33, wherein the switch polypeptide further comprises an additional intracellular domain.
35. The recombinant nucleic acid of claim 34, wherein the additional intracellular domain is operably linked to C-terminus of the switch intracellular domain.
36. The recombinant nucleic acid of claim 34 or 35, wherein the additional intracellular domain is derived from an intracellular domain of IL-15Ra.
37. The recombinant nucleic acid of any one of claims 34-36, wherein the additional intracellular domain comprises a sequence with at least 80% sequence identity to SEQ ID NO:372 or SEQ ID NO:383.
38. The recombinant nucleic acid of any one of claims 16-37, wherein the switch polypeptide comprises a transmembrane domain derived from a TGFBr2 transmembrane domain and an intracellular signaling domain of 4-1BB.
39. The recombinant nucleic acid of any one of claims 16-37, wherein the switch polypeptide comprises a transmembrane domain derived from a 4-1BB transmembrane domain and an intracellular signaling domain of 4-1BB.
40. The recombinant nucleic acid of any one of claims 16-37, wherein the switch polypeptide comprises a transmembrane domain derived from a TGFBr2 transmembrane domain and an intracellular signaling domain of CD28.
41. The recombinant nucleic acid of any one of claims 16-37, wherein the switch polypeptide comprises a transmembrane domain derived from a CD28 transmembrane domain and an intracellular signaling domain of CD28.
42. The recombinant nucleic acid of any one of claims 16-37, wherein the switch polypeptide comprises a sequence with at least 80% sequence identity to any one selected from SEQ ID NOs: 283, 284, 285, and 286.
43. The recombinant nucleic acid of any one of claims 1-42, wherein the TFP functionally interacts with an endogenous TCR complex when expressed in a T cell.
44. The recombinant nucleic acid of any one of claims 2-43, wherein the TCR intracellular domain comprises a stimulatory domain from an intracellular signaling domain of CD3 gamma, CD3 delta, or CD3 epsilon.
45. The recombinant nucleic acid of any one of claims 2-44, wherein the TCR intracellular domain comprises an intracellular domain from TCR alpha, TCR beta, TCR gamma, or TCR delta.
46. The recombinant nucleic acid of any one of claims 1-45, wherein the antigen binding domain is connected to the TCR extracellular domain by a linker sequence.
47. The recombinant nucleic acid of claim 46, wherein the linker sequence is 120 amino acids in length or less.
48. The recombinant nucleic acid of claim 46 or 47, wherein the linker sequence comprises (G4S)n, wherein G is glycine, S is serine, and n is an integer from 1 to 10 (SEQ ID NO: 390).
49. The recombinant nucleic acid of claim 48, wherein n is an integer from 1 to 4.
50. The recombinant nucleic acid of any one of claims 2-49, wherein at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from the same TCR subunit.
51. The recombinant nucleic acid of claim 50, wherein at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from TCR alpha.
52. The recombinant nucleic acid of claim 50, wherein at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from TCR beta.
53. The recombinant nucleic acid of claim 50, wherein at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from TCR gamma.
54. The recombinant nucleic acid of claim 50, wherein at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from TCR delta.
55. The recombinant nucleic acid of claim 50, wherein at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 epsilon.
56. The recombinant nucleic acid of claim 50, wherein at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 delta.
57. The recombinant nucleic acid of claim 50, wherein at least two of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 gamma.
58. The recombinant nucleic acid of any one of claims 50-57, wherein all three of the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from the same TCR subunit.
59. The recombinant nucleic acid of claim 58, wherein the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 epsilon.
60. The recombinant nucleic acid of claim 58, wherein the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 delta.
61. The recombinant nucleic acid of claim 58, wherein the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain are from CD3 gamma.
62. The recombinant nucleic acid of claim 58, wherein the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain comprise the constant domain of TCR alpha.
63. The recombinant nucleic acid of claim 62, wherein the constant domain of TCR alpha is murine.
64. The recombinant nucleic acid of claim 58, wherein the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain comprise the constant domain of TCR beta.
65. The recombinant nucleic acid of claim 64, wherein the constant domain of TCR beta is murine.
66. The recombinant nucleic acid of claim 58, wherein the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain comprise the constant domain of TCR gamma.
67. The recombinant nucleic acid of claim 58, wherein the TCR extracellular domain, the TCR transmembrane domain, and the TCR intracellular domain comprise the constant domain of TCR delta.
68. The recombinant nucleic acid of any one of claims 1-67, wherein the antigen binding domain is a camelid antibody or binding fragment thereof.
69. The recombinant nucleic acid of any one of claims 1-67, wherein the antigen binding domain is a murine antibody or binding fragment thereof.
70. The recombinant nucleic acid of any one of claims 1-67, wherein the antigen binding domain is a human or humanized antibody or binding fragment thereof.
71. The recombinant nucleic acid of any one of claims 1-70, wherein the antigen binding domain is a single-chain variable fragment (scFv) or a single domain antibody (sdAb) domain.
72. The recombinant nucleic acid of claim 71, wherein the antigen binding domain is a single domain antibody (sdAb).
73. The recombinant nucleic acid of claim 72, wherein the sdAb is a VH or VHH.
74. The recombinant nucleic acid of any one of claims 1-73, wherein the antigen binding domain is selected from the group consisting of an anti-CD19 binding domain, an anti-B-cell maturation antigen (BCMA) binding domain, and an anti-mesothelin (MSLN) binding domain, an anti-CD20 binding domain, an anti-CD70 binding domain, anti-MUC16 binding domain, an anti-Nectin-4 binding domain, an anti- GPC3 binding domain, and an anti-TROP-2 binding domain.
75. The recombinant nucleic acid of any one of claims 1-74, wherein a T cell expressing the TFP inhibits tumor growth when expressed in a T cell.
76. The recombinant nucleic acid of any one of claims 1-75, further comprising a leader sequence.
77. The recombinant nucleic acid of any one of claims 1-76, wherein the recombinant nucleic acid is selected from the group consisting of a DNA and an RNA.
78. The recombinant nucleic acid of claim 77, wherein the recombinant nucleic acid is an mRNA.
79. The recombinant nucleic acid of claim 77, wherein the recombinant nucleic acid is a circRNA.
80. The recombinant nucleic acid of any one of claims 1-79, wherein the recombinant nucleic acid comprises a nucleotide analog.
81. The recombinant nucleic acid of claim 80, wherein the nucleotide analog is selected from the group consisting of 2’-O-methyl, 2’-O-methoxyethyl (2’-O-MOE), 2’-O- aminopropyl, 2’-deoxy, T-deoxy-2’-fluoro, 2’-O-aminopropyl (2’-O-AP), 2'-O- dimethylaminoethyl (2’-O-DMAOE), 2’-O-dimethylaminopropyl (2’-O-DMAP), T- O-dimethylaminoethyloxyethyl (2’-O-DMAEOE), 2’-O-N-methylacetamido (2’-O- NMA) modified, a locked nucleic acid (LNA), an ethylene nucleic acid (ENA), a peptide nucleic acid (PNA), a 1’,5’- anhydrohexitol nucleic acid (HNA), a morpholino, a methylphosphonate nucleotide, a thiolphosphonate nucleotide, and a 2’-fluoro N3-P5’-phosphoramidite.
82. The recombinant nucleic acid of any one of claims 1-81, further comprising a promoter.
83. The recombinant nucleic acid of any one of claims 1-82, wherein the recombinant nucleic acid is an in vitro transcribed nucleic acid.
84. The recombinant nucleic acid of any one of claims 1-83, wherein the recombinant nucleic acid further comprises a sequence encoding a poly(A) tail.
85. The recombinant nucleic acid of any one of claims 1-84, wherein the recombinant nucleic acid further comprises a 3’UTR sequence.
86. A polypeptide encoded by the recombinant nucleic acid of any one of claims 1-85.
87. A vector comprising a recombinant nucleic acid of any one of claims 1-85.
88. The vector of claim 87, wherein the vector is a lentiviral vector.
89. A cell comprising the recombinant nucleic acid of any one of claims 1-85, the polypeptide of claim 86, or the vector of claim 87 or 88.
90. The cell of claim 89, wherein the cell is a T cell.
91. The cell of claim 90, wherein the T cell is a human T cell.
92. The cell of claim 90 or 91, wherein the T cell is a CD8+ or CD4+ T cell.
93. The cell of claim 90, wherein the T cell is a human αβ T cell.
94. The cell of claim 90, wherein the T cell is a human γδ T cell.
95. The cell of claim 89, wherein the cell is a human NKT cell.
96. The cell of any one of claims 89-95, wherein the cell is an allogeneic cell or an autologous cell.
97. The cell of any one of claims 89-96, wherein anti-tumor efficacy of the cell is increased compared to a cell that does not comprise the second nucleic acid sequence encoding a polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a portion thereof.
98. The cell of any one of claims 89-97, wherein expression of an exhaustion marker of the cell is decreased compared to a cell that comprises the first nucleic acid sequence and does not comprise the second nucleic acid sequence.
99. The cell of any one of claims 89-98, wherein cytotoxicity of the cell is increased compared to a cell that comprises the first nucleic acid sequence and does not comprise the second nucleic acid sequence .
100. The cell of any one of claims 89-99, wherein the cytokine production of the cell is increased compared to a cell that comprises the first nucleic acid sequence and does not comprise the second nucleic acid sequence.
101. The cell of claim 100, wherein the cytokine is selected from the group consisting of GM-CSF, IFNgamma, IL-2, and TNFalpha.
102. The cell of any one of claims 89-101, wherein pSMAD2/3 signaling is decreased compared to a cell that comprises the first nucleic acid sequence and does not comprise the second nucleic acid sequence.
103. The cell of any one of claims 89-102, wherein the cell comprises a population of cells.
104. A pharmaceutical composition comprising the cell of any one of claims 89-103 and a pharmaceutically acceptable carrier.
105. A modified T cell comprising a dominant negative receptor comprising the amino acid sequence of SEQ ID NO: 68, and a T cell receptor (TCR) fusion protein (TFP), wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain; wherein the TCR subunit and the antigen binding domain are operatively linked.
106. A modified T cell comprising a switch receptor comprising an amino acid sequence selected from the group consisting of SEQ ID NOs: 283, 284, 285, and 286; and a T cell receptor (TCR) fusion protein (TFP), wherein the TFP comprises: (a) a TCR subunit comprising:
(i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain; wherein the TCR subunit and the antigen binding domain are operatively linked.
107. A method of increasing an activity or cytotoxicity of a cell expressing a recombinant nucleic acid comprising a sequence encoding a T cell receptor (TCR) fusion protein (TFP), the method comprising expressing a polypeptide comprising a transforming growth factor beta receptor II (TGFBr2) extracellular domain or a functional fragment thereof in the cell: wherein the TFP comprises: (a) a TCR subunit comprising: (i) at least a portion of a TCR extracellular domain, and (ii) a TCR transmembrane domain, and (b) an antigen binding domain; and wherein the TCR subunit and the antigen binding domain are operatively linked.
108. The method of claim 107, wherein the polypeptide comprising a TGFBr2 extracellular domain or functional fragment thereof is a dominant negative TGFBr2 or a TGFBR2 switch.
109. The method of claim 108, wherein the cell is the cell of any one of claims 89-103.
110. A method of treating a disease or a condition in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of the pharmaceutical composition of claim 104.
111. The method of claim 110, wherein the disease or the condition is a cancer or a disease or a condition associated with expression of CD19, B-cell maturation antigen (BCMA), mesothelin (MSLN), CD20, CD70, MUC16, Trop-2, Nectin-4, or GPC3.
112. The method of claim 110 or 111, wherein the cancer is a hematologic cancer selected from the group consisting of B-cell acute lymphoid leukemia (B-ALL), T cell acute lymphoid leukemia (T-ALL), acute lymphoblastic leukemia (ALL), chronic myelogenous leukemia (CML), chronic lymphocytic leukemia (CLL), B cell prolymphocytic leukemia, blastic plasmacytoid dendritic cell neoplasm, Burkitt’s lymphoma, diffuse large B cell lymphoma, follicular lymphoma, hairy cell leukemia, small cell-follicular lymphoma, large cell-follicular lymphoma, malignant lymphoproliferative conditions, MALT lymphoma, mantle cell lymphoma, Marginal zone lymphoma, multiple myeloma, myelodysplasia, myelodysplastic syndrome, non- Hodgkin’s lymphoma, plasmablastic lymphoma, plasmacytoid dendritic cell
neoplasm, Waldenstrom macroglobulinemia, and preleukemia.
113. The method of claim 110 or 111, wherein the cancer is mesothelioma, renal cell carcinoma, stomach cancer, breast cancer, lung cancer, ovarian cancer, prostate cancer, colon cancer, cervical cancer, brain cancer, liver cancer, pancreatic cancer, thyroid cancer, bladder cancer, ureter cancer, kidney cancer, endometrial cancer, esophageal cancer, gastric cancer, thymic carcinoma or cholangiocarcinoma.
114. The method of any one of claims 110-113, wherein the subject is a human.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202163180292P | 2021-04-27 | 2021-04-27 | |
| US63/180,292 | 2021-04-27 | ||
| US202263318053P | 2022-03-09 | 2022-03-09 | |
| US63/318,053 | 2022-03-09 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022232277A1 true WO2022232277A1 (en) | 2022-11-03 |
Family
ID=83847459
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2022/026542 Ceased WO2022232277A1 (en) | 2021-04-27 | 2022-04-27 | COMPOSITIONS AND METHODS FOR TCR REPROGRAMMING USING FUSION PROTEINS AND TGFβR SWITCH |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2022232277A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP4146233A4 (en) * | 2020-05-05 | 2024-05-22 | TCR2 Therapeutics Inc. | COMPOSITIONS AND METHODS FOR TCR REPROGRAMMING USING CD70-SPECIFIC FUSION PROTEINS |
| WO2024103107A1 (en) * | 2022-11-14 | 2024-05-23 | Peter Maccallum Cancer Institute | Fusion proteins and uses thereof |
| EP4509522A1 (en) * | 2023-08-16 | 2025-02-19 | Max-Delbrück-Centrum für Molekulare Medizin in der Helmholtz-Gemeinschaft | A tgfbeta switch receptor, a nucleic acid encoding it, cells and pharmaceutical compositions comprising the same |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2020043152A1 (en) * | 2018-08-29 | 2020-03-05 | Nanjing Legend Biotech Co., Ltd. | Anti-mesothelin chimeric antigen receptor (car) constructs and uses thereof |
| WO2020223535A1 (en) * | 2019-05-01 | 2020-11-05 | Juno Therapeutics, Inc. | Cells expressing a recombinant receptor from a modified tgfbr2 locus, related polynucleotides and methods |
-
2022
- 2022-04-27 WO PCT/US2022/026542 patent/WO2022232277A1/en not_active Ceased
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2020043152A1 (en) * | 2018-08-29 | 2020-03-05 | Nanjing Legend Biotech Co., Ltd. | Anti-mesothelin chimeric antigen receptor (car) constructs and uses thereof |
| WO2020223535A1 (en) * | 2019-05-01 | 2020-11-05 | Juno Therapeutics, Inc. | Cells expressing a recombinant receptor from a modified tgfbr2 locus, related polynucleotides and methods |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP4146233A4 (en) * | 2020-05-05 | 2024-05-22 | TCR2 Therapeutics Inc. | COMPOSITIONS AND METHODS FOR TCR REPROGRAMMING USING CD70-SPECIFIC FUSION PROTEINS |
| WO2024103107A1 (en) * | 2022-11-14 | 2024-05-23 | Peter Maccallum Cancer Institute | Fusion proteins and uses thereof |
| EP4509522A1 (en) * | 2023-08-16 | 2025-02-19 | Max-Delbrück-Centrum für Molekulare Medizin in der Helmholtz-Gemeinschaft | A tgfbeta switch receptor, a nucleic acid encoding it, cells and pharmaceutical compositions comprising the same |
| WO2025036957A1 (en) * | 2023-08-16 | 2025-02-20 | Max-Delbrück-Centrum Für Molekulare Medizin In Der Helmholtz-Gemeinschaft | A tgfbeta switch receptor, a nucleic acid encoding it, cells and a pharmaceutical composition comprising the same |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7262535B2 (en) | Compositions and methods for TCR reprogramming using fusion proteins | |
| US11242376B2 (en) | Compositions and methods for TCR reprogramming using fusion proteins | |
| US20210079057A1 (en) | Compositions and methods for tcr reprogramming using fusion proteins | |
| US20210361704A1 (en) | Compositions and methods for tcr reprogramming using fusion proteins | |
| JP2022188163A (en) | Engineered t cells for the treatment of cancer | |
| US20210315933A1 (en) | Compositions and methods for tcr reprogramming using target specific fusion proteins | |
| WO2022006451A2 (en) | Compositions and methods for tcr reprogramming using fusion proteins and pd-1 antibodies | |
| WO2021035170A1 (en) | Compositions and methods for tcr reprogramming using fusion proteins | |
| WO2021133959A2 (en) | Compositions and methods for gamma delta tcr reprogramming using fusion proteins | |
| CA3154287A1 (en) | Compositions and methods for tcr reprogramming using fusion proteins | |
| WO2022232277A1 (en) | COMPOSITIONS AND METHODS FOR TCR REPROGRAMMING USING FUSION PROTEINS AND TGFβR SWITCH | |
| WO2022056321A1 (en) | Compositions and methods for tcr reprogramming using gpc3 specific fusion proteins | |
| US20240117002A1 (en) | Compositions and methods for tcr reprogramming using fusion proteins | |
| WO2022056304A1 (en) | Compositions and methods for tcr reprogramming using nectin-4 specific fusion proteins | |
| WO2022192286A1 (en) | Compositions and methods for tcr reprogramming using fusion proteins and rna interference | |
| CN117222738A (en) | Compositions and methods for TCR reprogramming using fusion proteins | |
| WO2021155034A1 (en) | Compositions and methods for tcr reprogramming using muc16 specific fusion proteins | |
| WO2023034220A2 (en) | Compositions and methods for tcr reprogramming using fusion proteins and cxcr6 | |
| WO2023091420A2 (en) | Compositions and methods for t cell engineering | |
| WO2023133424A2 (en) | Compositions and methods for tcr reprogramming using fusion proteins and anti-pd-1 fusion peptides | |
| WO2023172967A2 (en) | Compositions and methods for tcr reprogramming using gpc3 specific fusion proteins | |
| WO2023086379A2 (en) | Compositions and methods for tcr reprogramming using fusion proteins |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22796644 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 22796644 Country of ref document: EP Kind code of ref document: A1 |