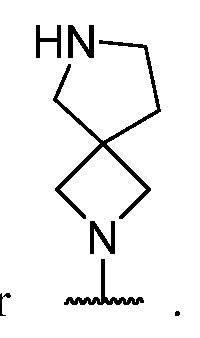WO2022217042A1 - Naphthyl-substituted quinoline-4(1h)-ones and related compounds and their use in treating medical conditions - Google Patents
Naphthyl-substituted quinoline-4(1h)-ones and related compounds and their use in treating medical conditions Download PDFInfo
- Publication number
- WO2022217042A1 WO2022217042A1 PCT/US2022/024006 US2022024006W WO2022217042A1 WO 2022217042 A1 WO2022217042 A1 WO 2022217042A1 US 2022024006 W US2022024006 W US 2022024006W WO 2022217042 A1 WO2022217042 A1 WO 2022217042A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- certain embodiments
- nitrogen
- substituted
- occurrences
- heterocyclyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/08—Bridged systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D519/00—Heterocyclic compounds containing more than one system of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring system not provided for in groups C07D453/00 or C07D455/00
Definitions
- the invention provides naphthyl-substituted quinolin-4(1H)-oncs and related compounds, pharmaceutical compositions, their use for inhibiting K-Ras G12D activity, and their use in the treatment of medical disorders, such as cancer.
- K-Ras Viral Oncogene Homolog
- K-Ras G12D and G12C activating mutations of K-Ras
- K-Ras G12D inhibitor compounds for treating cancer has been described in, for example, international patent application WO 2021/041671, the contents of which are herein incorporated by reference in its entirety.
- the invention provides naphthyl-substituted quinolin-4( 1H)-oncs and related compounds, pharmaceutical compositions, their use for inhibiting K-Ras G12D activity, and their use in the treatment of medical disorders, such as cancer.
- one aspect of the invention provides a collection of naphthyl-substituted 3,4-dihydroisoquinolin- 1 (2H)-oncs and related compounds, such as a compound represented by Formula I: or a pharmaceutically acceptable salt thereof, where the variables are as defined in the detailed description. Further description of additional collections of naphthyl-substituted quinolin-4( 1H)- ones and related compounds are described in the detailed description.
- the compounds may be part of a pharmaceutical composition comprising a pharmaceutically acceptable carrier.
- Another aspect of the invention provides a collection of naphthyl-substituted quinolin- 4(1 H)-oncs and related compounds, such as a compound represented by Formula II: or a pharmaceutically acceptable salt thereof, where the variables are as defined in the detailed description. Further description of additional collections of naphthyl-substituted quinolin-4( 1H)- ones and related compounds are described in the detailed description.
- the compounds may be part of a pharmaceutical composition comprising a pharmaceutically acceptable carrier.
- Another aspect of the invention provides a collection of naphthyl-substituted AH- pyrazino[l,2-a]pyrazin-4-ones and related compounds, such as a compound represented by Formula III: or a pharmaceutically acceptable salt thereof, where the variables are as defined in the detailed description. Further description of additional collections of naphthyl-substituted AH- pyrazino[l,2-a]pyrazin-4-ones and related compounds are described in the detailed description.
- the compounds may be part of a pharmaceutical composition comprising a pharmaceutically acceptable carrier.
- Another aspect of the invention provides a method of treating a disorder mediated by K- Ras G12D in a subject.
- the method comprises administering a therapeutically effective amount of a compound described herein, such as a compound of Formula I, II, or III, to a subject in need thereof to treat the disorder, as further described in the detailed description.
- Another aspect of the invention provides a method of inhibiting K-Ras G12D activity.
- the method comprises contacting K-Ras G12D with an effective amount of a compound described herein, such as a compound of Formula I, II, or III, to inhibit K-Ras G12D activity, as further described in the detailed description.
- the invention provides naphthyl-substituted quinolin-4( 1H)-oncs and related compounds, pharmaceutical compositions, their use for inhibiting K-Ras G12D activity, and their use in the treatment of medical disorders, such as cancer.
- the practice of the present invention employs, unless otherwise indicated, conventional techniques of organic chemistry, pharmacology, molecular biology (including recombinant techniques), cell biology, biochemistry, and immunology. Such techniques are explained in the literature, such as in “Comprehensive Organic Synthesis” (B.M. Trost & I. Fleming, eds., 1991-1992); “Handbook of experimental immunology” (D.M. Weir & C.C.
- aliphatic or “aliphatic group”, as used herein, means a straight-chain (i.e., unbranched) or branched, substituted or unsubstituted hydrocarbon chain that is completely saturated or that contains one or more units of unsaturation, or a monocyclic hydrocarbon or bicyclic hydrocarbon that is completely saturated or that contains one or more units of unsaturation, but which is not aromatic (also referred to herein as “cycloaliphatic”), that has a single point of attachment to the rest of the molecule.
- aliphatic groups contain 1-6 aliphatic carbon atoms. In some embodiments, aliphatic groups contain 1-5 aliphatic carbon atoms.
- aliphatic groups contain 1-4 aliphatic carbon atoms. In still other embodiments, aliphatic groups contain 1-3 aliphatic carbon atoms, and in yet other embodiments, aliphatic groups contain 1-2 aliphatic carbon atoms.
- “cycloaliphatic” refers to a monocyclic C3-C6 hydrocarbon that is completely saturated or that contains one or more units of unsaturation, but which is not aromatic, that has a single point of attachment to the rest of the molecule.
- Suitable aliphatic groups include, but are not limited to, linear or branched, substituted or unsubstituted alkyl, alkenyl, alkynyl groups and hybrids thereof such as (cycloalkyl)alkyl, (cycloalkenyl)alkyl or (cycloalkyl)alkenyl.
- bicyclic ring or “bicyclic ring system” refers to any bicyclic ring system, i.e. carbocyclic or heterocyclic, saturated or having one or more units of unsaturation, having one or more atoms in common between the two rings of the ring system.
- the term includes any permissible ring fusion, such as ortho -fused or spirocyclic.
- heterocyclic is a subset of “bicyclic” that requires that one or more heteroatoms are present in one or both rings of the bicycle.
- Such heteroatoms may be present at ring junctions and are optionally substituted, and may be selected from nitrogen (including N- oxides), oxygen, sulfur (including oxidized forms such as sulfones and sulfonates), phosphorus (including oxidized forms such as phosphates), boron, etc.
- a bicyclic group has 7-12 ring members and 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur.
- bridged bicyclic refers to any bicyclic ring system, i.e. carbocyclic or heterocyclic, saturated or partially unsaturated, having at least one bridge.
- a “bridge” is an unbranched chain of atoms or an atom or a valence bond connecting two bridgeheads, where a “bridgehead” is any skeletal atom of the ring system which is bonded to three or more skeletal atoms (excluding hydrogen).
- a bridged bicyclic group has 7-12 ring members and 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur.
- Such bridged bicyclic groups are well known in the art and include those groups set forth below where each group is attached to the rest of the molecule at any substitutable carbon or nitrogen atom.
- a bridged bicyclic group is optionally substituted with one or more substituents as set forth for aliphatic groups. Additionally or alternatively, any substitutable nitrogen of a bridged bicyclic group is optionally substituted.
- Exemplary bicyclic rings include:
- Exemplary bridged bicyclics include:
- lower alkyl refers to a C 1-4 straight or branched alkyl group.
- exemplary lower alkyl groups are methyl, ethyl, propyl, isopropyl, butyl, isobutyl, and tert-butyl.
- lower haloalkyl refers to a CIM straight or branched alkyl group that is substituted with one or more halogen atoms.
- heteroatom means one or more of oxygen, sulfur, nitrogen, phosphorus, or silicon (including, any oxidized form of nitrogen, sulfur, phosphorus, or silicon; the quatemized form of any basic nitrogen or; a substitutable nitrogen of a heterocyclic ring, for example N (as in 3 , 4 - d i h y d ro - 2 H- p y rrp l y l ) , NH (as in pyrrolidinyl) or NR + (as in N-substituted pyrrolidinyl)).
- unsaturated as used herein, means that a moiety has one or more units of unsaturation.
- bivalent C1-8 saturated or unsaturated, straight or branched, hydrocarbon chain
- bivalent alkylene, alkenylene, and alkynylene chains that are straight or branched as defined herein.
- alkylene refers to a bivalent alkyl group.
- An “alkylene chain” is a polymethylene group, i.e., -(CH2) n- , wherein n is a positive integer, preferably from 1 to 6, from 1 to 4, from 1 to 3, from 1 to 2, or from 2 to 3.
- a substituted alkylene chain is a polymethylene group in which one or more methylene hydrogen atoms are replaced with a substituent. Suitable substituents include those described below for a substituted aliphatic group.
- -(Co alkylene)- refers to a bond. Accordingly, the term “-(Co-3 alkylene)-” encompasses a bond (i.e., Co) and a -(C1-3 alkylene)- group.
- alkenylene refers to a bivalent alkenyl group.
- a substituted alkenylene chain is a polymethylene group containing at least one double bond in which one or more hydrogen atoms are replaced with a substituent. Suitable substituents include those described below for a substituted aliphatic group.
- halogen means F, Cl, Br, or I.
- aryl used alone or as part of a larger moiety as in “aralkyl,” “aralkoxy,” or “aryloxyalkyl,” refers to monocyclic or bicyclic ring systems having a total of five to fourteen ring members, wherein at least one ring in the system is aromatic and wherein each ring in the system contains 3 to 7 ring members.
- aryl may be used interchangeably with the term “aryl ring.”
- aryl refers to an aromatic ring system which includes, but not limited to, phenyl, biphenyl, naphthyl, anthracyl and the like, which may bear one or more substituents.
- aryl is a group in which an aromatic ring is fused to one or more non-aromatic rings, such as indanyl, phthalimidyl, naphthimidyl, phenanthridinyl, or tetrahydronaphthyl, and the like.
- phenylene refers to a multivalent phenyl group having the appropriate number of open valences to account for groups attached to it. For example, “phenylene” is a bivalent phenyl group when it has two groups attached trivalent phenyl group when it has three groups attached to it (e.g., ).
- arylene refers to a bivalent aryl group.
- heteroaryl and “heteroar-,” used alone or as part of a larger moiety, e.g., “heteroaralkyl,” or “heteroaralkoxy,” refer to groups having 5 to 10 ring atoms, preferably 5, 6, or 9 ring atoms; having 6, 10, or 14 p electrons shared in a cyclic array; and having, in addition to carbon atoms, from one to five heteroatoms.
- heteroatom refers to nitrogen, oxygen, or sulfur, and includes any oxidized form of nitrogen or sulfur, and any quaternized form of a basic nitrogen.
- Heteroaryl groups include, without limitation, thienyl, furanyl, pyrrolyl, imidazolyl, pyrazolyl, triazolyl, tetrazolyl, oxazolyl, isoxazolyl, oxadiazolyl, thiazolyl, isothiazolyl, thiadiazolyl, pyridyl, pyridazinyl, pyrimidinyl, pyrazinyl, indolizinyl, purinyl, naphthyridinyl, and pteridinyl.
- heteroaryl and “heteroar-”, as used herein, also include groups in which a hetero aromatic ring is fused to one or more aryl, cycloaliphatic, or heterocyclyl rings, where unless otherwise specified, the radical or point of attachment is on the heteroaromatic ring or on one of the rings to which the hetero aromatic ring is fused.
- Nonlimiting examples include indolyl, isoindolyl, benzothienyl, benzofuranyl, dibenzofuranyl, indazolyl, benzimidazolyl, benzthiazolyl, quinolyl, isoquinolyl, cinnolinyl, phthalazinyl, quinazolinyl, quinoxalinyl, 4/7-quinolizinyl, carbazolyl, acridinyl, phenazinyl, phenothiazinyl, phenoxazinyl, tetrahydroquinolinyl, and tetrahydroisoquinolinyl.
- a heteroaryl group may be mono- or bicyclic.
- heteroaryl may be used interchangeably with the terms “heteroaryl ring,” “heteroaryl group,” or “heteroaromatic,” any of which terms include rings that are optionally substituted.
- heteroarylkyl refers to an alkyl group substituted by a heteroaryl, wherein the alkyl and heteroaryl portions independently are optionally substituted.
- heteroarylene refers to a multivalent heteroaryl group having the appropriate number of open valences to account for groups attached to it.
- heteroarylene is a bivalent heteroaryl group when it has two groups attached to it; “heteroarylene” is a trivalent heteroaryl group when it has three groups attached to it.
- pyridinylene refers to a multivalent pyridine radical having the appropriate number of open valences to account for groups attached to it.
- pyridinylene is a bivalent pyridine radical when it has two
- heterocycle As used herein, the terms “heterocycle,” “heterocyclyl,” “heterocyclic radical,” and “heterocyclic ring” are used interchangeably and refer to a stable 5- to 7-membered monocyclic or 7-10-membered bicyclic heterocyclic moiety that is either saturated or partially unsaturated, and having, in addition to carbon atoms, one or more, preferably one to four, heteroatoms, as defined above.
- nitrogen includes a substituted nitrogen.
- the nitrogen in a saturated or partially unsaturated ring having 0-3 heteroatoms selected from oxygen, sulfur or nitrogen, the nitrogen may be N (as in 3,4- di h y dro-2H-py rro l y l ) , NH (as in pyrrolidinyl), or + NR (as in A-substitutcd pyrrolidinyl).
- a heterocyclic ring can be attached to its pendant group at any heteroatom or carbon atom that results in a stable structure and any of the ring atoms can be optionally substituted.
- saturated or partially unsaturated heterocyclic radicals include, without limitation, tetrahydrofuranyl, tetrahydrothiophenyl pyrrolidinyl, piperidinyl, pyrrolinyl, tetrahydroquinolinyl, tetrahydroisoquinolinyl, decahydroquinolinyl, oxazolidinyl, piperazinyl, dioxanyl, dioxolanyl, diazepinyl, oxazepinyl, thiazepinyl, morpholinyl, 2-oxa-6- azaspiro[3.3]heptane, and quinuclidinyl.
- heterocycle used interchangeably herein, and also include groups in which a heterocyclyl ring is fused to one or more aryl, heteroaryl, or cycloaliphatic rings, such as indolinyl, 3H-indolyl, chromanyl, phenanthridinyl, or tetrahydroquinolinyl.
- a heterocyclyl group may be mono- or bicyclic.
- heterocyclylalkyl refers to an alkyl group substituted by a heterocyclyl, wherein the alkyl and heterocyclyl portions independently are optionally substituted.
- oxo-heterocyclyl refers to a heterocyclyl substituted by an oxo group.
- heterocyclylene refers to a multivalent heterocyclyl group having the appropriate number of open valences to account for groups attached to it. For example, “heterocyclylene” is a bivalent heterocyclyl group when it has two groups attached to it; “heterocyclylene” is a trivalent heterocyclyl group when it has three groups attached to it.
- partially unsaturated refers to a ring moiety that includes at least one double or triple bond.
- partially unsaturated is intended to encompass rings having multiple sites of unsaturation, but is not intended to include aryl or heteroaryl moieties, as herein defined.
- compounds of the invention may contain “optionally substituted” moieties.
- substituted whether preceded by the term “optionally” or not, means that one or more hydrogens of the designated moiety are replaced with a suitable substituent.
- an “optionally substituted” group may have a suitable substituent at each substitutable position of the group, and when more than one position in any given structure may be substituted with more than one substituent selected from a specified group, the substituent may be either the same or different at every position.
- Combinations of substituents envisioned by this invention are preferably those that result in the formation of stable or chemically feasible compounds.
- stable refers to compounds that are not substantially altered when subjected to conditions to allow for their production, detection, and, in certain embodiments, their recovery, purification, and use for one or more of the purposes disclosed herein.
- S(O) 2 N C(NR° 2 ) 2 ; -(CH 2 ) 0-4 S(O)R°; -N(R°)S(O) 2 NRo 2 ; -N(R°)S(O) 2 R°; -N(OR°)R°; - C(NH)NR° 2 ; -P(0) 2 R°; -P(0)R° 2 ; -0P(0)R° 2 ; -0P(0)(0R°) 2 ; SiR° 3 ; -(C1-4 straight or branched alkylene)0-N(R°) 2 ; or — (C i 4 straight or branched alkylene)C(0)0-N(R°) 2 .
- R * is C1-6 aliphatic
- R * is optionally substituted with halogen, - R*, -(haloR*), -OH, -OR*, -0(haloR*), -CN, -C(0)OH, -C(0)OR*, -NH 2 , -NHR*, -NR* 2 , or -N0 2
- each R* is independently selected from Ci 4 aliphatic, -CH 2 Ph, -0(CH 2 )o iPh, or a 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur, and wherein each R* is unsubstituted or where preceded by halo is substituted only with one or more halogens.
- An optional substituent on a substitutable nitrogen is independently -R ⁇ , -NR ⁇ 2 , - C(0)R ⁇ , -C(0)0R ⁇ , -C(0)C(0)R ⁇ , -C(0)CH 2 C(0)R ⁇ , -S(0) 2 R ⁇ , -S(0) 2 NR ⁇ 2 , -C(S)NR ⁇ 2 , - C(NH)NR ⁇ 2 , or -N(R ⁇ )S(0) 2 R ⁇ ; wherein each R ⁇ is independently hydrogen, C1-6 aliphatic, unsubstituted -OPh, or an unsubstituted 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur, or, two independent occurrences of R ⁇ , taken together with their intervening atom(s) form an unsubstituted 3-12-membered saturated, partially unsaturated,
- the term "pharmaceutically acceptable salt” refers to those salts which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of humans and lower animals without undue toxicity, irritation, allergic response and the like, and are commensurate with a reasonable benefit/risk ratio.
- Pharmaceutically acceptable salts are well known in the art. For example, S. M. Berge et al., describe pharmaceutically acceptable salts in detail in J. Pharmaceutical Sciences, 1977, 66, 1-19, incorporated herein by reference.
- Pharmaceutically acceptable salts of the compounds of this invention include those derived from suitable inorganic and organic acids and bases.
- Examples of pharmaceutically acceptable, nontoxic acid addition salts are salts of an amino group formed with inorganic acids such as hydrochloric acid, hydrobromic acid, phosphoric acid, sulfuric acid and perchloric acid or with organic acids such as acetic acid, oxalic acid, maleic acid, tartaric acid, citric acid, succinic acid or malonic acid or by using other methods used in the art such as ion exchange.
- inorganic acids such as hydrochloric acid, hydrobromic acid, phosphoric acid, sulfuric acid and perchloric acid
- organic acids such as acetic acid, oxalic acid, maleic acid, tartaric acid, citric acid, succinic acid or malonic acid or by using other methods used in the art such as ion exchange.
- salts include adipate, alginate, ascorbate, aspartate, benzenesulfonate, benzoate, bisulfate, borate, butyrate, camphorate, camphorsulfonate, citrate, cyclopentanepropionate, digluconate, dodecylsulfate, ethanesulfonate, formate, fumarate, glucoheptonate, glycerophosphate, gluconate, hemisulfate, heptanoate, hexanoate, hydroiodide, 2-hydroxy-ethanesulfonate, lactobionate, lactate, laurate, lauryl sulfate, malate, maleate, malonate, methanesulfonate, 2-naphthalenesulfonate, nicotinate, nitrate, oleate, oxalate, palmitate, pamoate, pectinate,
- Salts derived from appropriate bases include alkali metal, alkaline earth metal, ammonium and N + (Ci 4alkyl)4 salts.
- Representative alkali or alkaline earth metal salts include sodium, lithium, potassium, calcium, magnesium, and the like.
- Further pharmaceutically acceptable salts include, when appropriate, nontoxic ammonium, quaternary ammonium, and amine cations formed using counterions such as halide, hydroxide, carboxylate, sulfate, phosphate, nitrate, loweralkyl sulfonate and aryl sulfonate.
- structures depicted herein are also meant to include all isomeric (e.g ., enantiomeric, diastereomeric, and geometric (or conformational)) forms of the structure; for example, the R and S configurations for each asymmetric center, Z and E double bond isomers, and Z and E conformational isomers. Therefore, single stereochemical isomers as well as enantiomeric, diastereomeric, and geometric (or conformational) mixtures of the present compounds are within the scope of the invention. Unless otherwise stated, all tautomeric forms of the compounds of the invention are within the scope of the invention.
- structures depicted herein are also meant to include compounds that differ only in the presence of one or more isotopically enriched atoms.
- compounds having the present structures including the replacement of hydrogen by deuterium or tritium, or the replacement of a carbon by a 13 C- or 14 C-enriched carbon are within the scope of this invention.
- Such compounds are useful, for example, as analytical tools, as probes in biological assays, or as therapeutic agents in accordance with the present invention.
- Diastereomeric mixtures can be separated into their individual diastereomers on the basis of their physical chemical differences by methods known to those skilled in the art, such as, for example, by chromatography and/or fractional crystallization.
- Enantiomers can be separated by converting the enantiomeric mixture into a diastereomeric mixture by reaction with an appropriate optically active compound (e.g ., chiral auxiliary such as a chiral alcohol or Mosher’s acid chloride), separating the diastereomers and converting (e.g., hydrolyzing) the individual diastereomers to the corresponding pure enantiomers.
- an appropriate optically active compound e.g ., chiral auxiliary such as a chiral alcohol or Mosher’s acid chloride
- a particular enantiomer of a compound of the present invention may be prepared by asymmetric synthesis.
- diastereomeric salts are formed with an appropriate optically- active acid or base, followed by resolution of the diastereomers thus formed by fractional crystallization or chromatographic means known in the art, and subsequent recovery of the pure enantiomers.
- Individual stereoisomers of the compounds of the invention may, for example, be substantially free of other isomers, or may be admixed, for example, as racemates or with all other, or other selected, stereoisomers.
- Chiral center(s) in a compound of the present invention can have the S or R configuration as defined by the IUPAC 1974 Recommendations.
- a compound described herein may exist as an atropisomer (e.g., substituted biaryls)
- all forms of such atropisomer are considered part of this invention.
- Chemical names, common names, and chemical structures may be used interchangeably to describe the same structure. If a chemical compound is referred to using both a chemical structure and a chemical name, and an ambiguity exists between the structure and the name, the structure predominates. It should also be noted that any carbon as well as heteroatom with unsatisfied valences in the text, schemes, examples and tables herein is assumed to have the sufficient number of hydrogen atom(s) to satisfy the valences.
- alkyl refers to a saturated straight or branched hydrocarbon, such as a straight or branched group of 1-12, 1-10, or 1-6 carbon atoms, referred to herein as C1-C12 alkyl, C1-C10 alkyl, and C1-C6 alkyl, respectively.
- Exemplary alkyl groups include, but are not limited to, methyl, ethyl, propyl, isopropyl, 2-methyl- 1 -propyl, 2-methyl-2-propyl, 2-methyl- 1 -butyl, 3- methyl-1 -butyl, 2-methyl-3-butyl, 2,2-dimethyl- 1 -propyl, 2-methyl- 1 -pentyl, 3 -methyl- 1 -pentyl, 4-methyl- 1 -pentyl, 2-methyl-2-pentyl, 3-methyl-2-pentyl, 4-methyl-2-pentyl, 2,2-dimethyl- 1- butyl, 3, 3 -dimethyl- 1 -butyl, 2-ethyl- 1 -butyl, butyl, isobutyl, t-butyl, pentyl, isopentyl, neopentyl, hexyl, heptyl, octyl, etc.
- cycloalkyl refers to a monovalent saturated cyclic, bicyclic, or bridged cyclic (e.g ., adamantyl) hydrocarbon group of 3-12, 3-8, 4-8, or 4-6 carbons, referred to herein, e.g., as “C3-C6 cycloalkyl,” derived from a cycloalkane.
- exemplary cycloalkyl groups include cyclohexyl, cyclopentyl, cyclobutyl, and cyclopropyl.
- cycloalkylene refers to a bivalent cycloalkyl group.
- haloalkyl refers to an alkyl group that is substituted with at least one halogen.
- exemplary haloalkyl groups include -CH2F, -CHF2, -CF3, -CH2CF3, -CF2CF3, and the like.
- haloalkylene refers to a bivalent haloalkyl group.
- hydroxy alkyl refers to an alkyl group that is substituted with at least one hydroxyl.
- exemplary hydroxyalkyl groups include -CH2CH2OH, -C(H)(OH)CH3, -CH 2 C(H)(OH)CH 2 CH 2 OH, and the like.
- alkenyl and alkynyl are art-recognized and refer to unsaturated aliphatic groups analogous in length and possible substitution to the alkyls described above, but that contain at least one double or triple bond respectively.
- Carbocyclylene refers to a multivalent carbocyclyl group having the appropriate number of open valences to account for groups attached to it.
- “carbocyclylene” is a bivalent carbocyclyl group when it has two groups attached to it; “carbocyclylene” is a trivalent carbocyclyl group when it has three groups attached to it.
- alkoxyl or “alkoxy” are art-recognized and refer to an alkyl group, as defined above, having an oxygen radical attached thereto.
- Representative alkoxyl groups include methoxy, ethoxy, propyloxy, ieri-butoxy and the like.
- haloalkoxyl refers to an alkoxyl group that is substituted with at least one halogen.
- Exemplary haloalkoxyl groups include -OCH2F, -OCHF2, -OCF3, -OCH2CF3, -OCF2CF3, and the like.
- hydroxyalkoxyl refers to an alkoxyl group that is substituted with at least one hydroxyl.
- hydroxyalkoxyl groups include -OCH2CH2OH, -0CH 2 C(H)(0H)CH 2 CH 2 0H, and the like.
- alkoxylene refers to a bivalent alkoxyl group.
- a cyclopentane susbsituted with an oxo group is cyclopentanone.
- the chemical structure encompasses and .
- a chemical structure containing a polycyclic fused ring when depicted with one or more substituent(s) having a bond that crosses multiple rings, the one or more substituent(s) may be independently attached to any of the rings crossed by the bond.
- the chemical structure encompasses, for example,
- One or more compounds of the invention may exist in unsolvated as well as solvated forms with pharmaceutically acceptable solvents such as water, ethanol, and the like, and it is intended that the invention embrace both solvated and unsolvated forms.
- “Solvate” means a physical association of a compound of this invention with one or more solvent molecules. This physical association involves varying degrees of ionic and covalent bonding, including hydrogen bonding. In certain instances the solvate will be capable of isolation, for example when one or more solvent molecules are incorporated in the crystal lattice of the crystalline solid. “Solvate” encompasses both solution-phase and isolatable solvates. Non-limiting examples of suitable solvates include ethanolates, methanolates, and the like. “Hydrate” is a solvate wherein the solvent molecule is H2O.
- the terms “subject” and “patient” are used interchangeably and refer to organisms to be treated by the methods of the present invention.
- Such organisms preferably include, but are not limited to, mammals (e.g ., murines, simians, equines, bovines, porcines, canines, felines, and the like), and, most preferably, includes humans.
- an inhibitor has an IC50 and/or binding constant of less than about 100 mM, less than about 50 mM, less than about 1 mM, less than about 500 nM, less than about 100 nM, less than about 10 nM, or less than about 1 nM.
- K-Ras refers to a mammalian K-Ras protein.
- the assignment of amino acid codon and residue positions for human K-Ras is based on the amino acid sequence identified by UniProtKB/Swiss-Prot P01116.
- K-Ras G12D refers to a mutant form of a mammalian K-Ras protein that contains an amino acid substitution of an aspartic acid, instead of a glycine, at amino acid position 12.
- the assignment of amino acid codon and residue positions for human K-Ras is based on the amino acid sequence identified by UniProtKB/Swiss-Prot P01116:
- V ariantp Gly 12 Asp .
- K-Ras inhibitor or “K-Ras antagonist” are defined as a compound that binds to and/or inhibits K-Ras with measurable affinity. In some embodiments, inhibition in the presence of the inhibitor is observed in a dose-dependent manner.
- the measured signal (e.g., signaling activity or biological activity) is at least about 5%, at least about 10%, at least about 15%, at least about 20%, at least about 25%, at least about 30%, at least about 35%, at least about 40%, at least about 45%, at least about 50%, at least about 55%, at least about 60%, at least about 65%, at least about 70%, at least about 75%, at least about 80%, at least about 85%, at least about 90%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, at least about 99%, or at least about 100% lower than the signal measured with a negative control under comparable conditions.
- inhibitor or “K-Ras G12D inhibitor” or “K-Ras G12D antagonist” are defined as a compound that binds to and/or inhibits K-Ras G12D with measurable affinity. In some embodiments, inhibition in the presence of the inhibitor is observed in a dose-dependent manner.
- the measured signal (e.g ., signaling activity or biological activity) is at least about 5%, at least about 10%, at least about 15%, at least about 20%, at least about 25%, at least about 30%, at least about 35%, at least about 40%, at least about 45%, at least about 50%, at least about 55%, at least about 60%, at least about 65%, at least about 70%, at least about 75%, at least about 80%, at least about 85%, at least about 90%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, at least about 99%, or at least about 100% lower than the signal measured with a negative control under comparable conditions.
- measurable affinity and “measurably inhibit,” as used herein, means a measurable change or inhibition in K-Ras activity, such as K-Ras G12D activity, between a sample comprising a compound of the present invention, or composition thereof, and K-Ras, such as K-Ras G12D, and an equivalent sample comprising K-Ras, such as K-Ras G12D, in the absence of said compound, or composition thereof.
- an effective amount refers to the amount of a compound sufficient to effect beneficial or desired results (e.g., a therapeutic, ameliorative, inhibitory, or preventative result).
- An effective amount can be administered in one or more administrations, applications, or dosages and is not intended to be limited to a particular formulation or administration route.
- treating includes any effect, e.g., lessening, reducing, modulating, ameliorating or eliminating, that results in the improvement of the condition, disease, disorder, and the like, or ameliorating a symptom thereof.
- treatment can be administered after one or more symptoms have developed.
- treatment can be administered in the absence of symptoms.
- treatment can be administered to a susceptible individual prior to the onset of symptoms (e.g., in light of a history of symptoms and/or in light of genetic or other susceptibility factors). Treatment can also be continued after symptoms have resolved, for example, to prevent or delay their recurrence.
- K-Ras-associated disease or disorder means any disease or other deleterious condition in which K-Ras, or a variant or mutant thereof, is known to play a role.
- a non-limiting example of a K-Ras-associated disease or disorder is a K-Ras-associated cancer.
- K-Ras G12D disorder mediated by K-Ras G12D
- disease mediated by K-Ras G12D or “disease mediated by K-Ras G12D” or "K-Ras G12D-associated disease or disorder,” as used herein, refer to diseases or disorders associated with, or mediated by, or having a K-Ras G12D mutation.
- a non-limiting example of a K-Ras G12D-associated disease or disorder is a K-Ras G12D-associated cancer.
- composition refers to the combination of an active agent with a carrier, inert or active, making the composition especially suitable for diagnostic or therapeutic use in vivo or ex vivo.
- the term “pharmaceutically acceptable carrier” refers to any of the standard pharmaceutical carriers, such as a phosphate buffered saline solution, water, emulsions ( e.g ., such as an oil/water or water/oil emulsions), and various types of wetting agents.
- the compositions also can include stabilizers and preservatives.
- stabilizers and adjuvants see e.g., Martin, Remington’s Pharmaceutical Sciences, 15th Ed., Mack Publ. Co., Easton, PA [1975].
- salts of the compounds of the present invention are contemplated as being pharmaceutically acceptable.
- salts of acids and bases that are non- pharmaceutically acceptable may also find use, for example, in the preparation or purification of a pharmaceutically acceptable compound.
- a compound of the invention contains both a basic moiety (such as, but not limited to, a pyridine or imidazole) and an acidic moiety (such as, but not limited to, a carboxylic acid) zwitterions (“inner salts”) may be formed.
- acidic and basic salts used within the scope of the invention are pharmaceutically acceptable (i.e., non-toxic, physiologically acceptable) salts.
- Such salts of the compounds of the invention may be formed, for example, by reacting a compound of the invention with an amount of acid or base, such as an equivalent amount, in a medium such as one in which the salt precipitates or in an aqueous medium followed by lyophilization.
- compositions are described as having, including, or comprising specific components, or where processes and methods are described as having, including, or comprising specific steps, it is contemplated that, additionally, there are compositions of the present invention that consist essentially of, or consist of, the recited components, and that there are processes and methods according to the present invention that consist essentially of, or consist of, the recited processing steps.
- compositions specifying a percentage are by weight unless otherwise specified.
- the invention provides naphthyl-substituted quinolin-4( 1H)-oncs and related compounds.
- the compounds may be used in the pharmaceutical compositions and therapeutic methods described herein. Exemplary compounds are described in the following sections, along with exemplary procedures for making the compounds.
- One aspect of the invention provides a compound represented by Formula I: or a pharmaceutically acceptable salt thereof; wherein:
- R 1 is naphthyl; an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with m occurrences of R 5 ;
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci- 6 aliphatic; each of which is substituted with n occurrences of R 6 ;
- R 3 is hydrogen; or one occurrence of R 3 is hydrogen, and the other occurrence of R 3 is taken together with R 4 to form a double bond between the atoms to which R 3 and R 4 are attached; or both occurrences of R 3 are taken together to form an oxo substituent;
- R 4 is taken together with one occurrence of R 3 to form a double bond between the atoms to which R 3 and R 4 are attached;
- R 5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, Ci- 6 alkyl, hydroxyl, Ci- 6 alkoxyl, halo, or cyano;
- R 6 and R 7 each represent independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 haloalkoxyl, Ci- 6 alkyl, Ci- 6 haloalkyl, Ci- 6 hydroxyalkyl, or -N(R 9 )2;
- R 8 and R 9 each represent independently for each occurrence hydrogen, C M alkyl, or C3-5 cycloalkyl; or two occurrences of R 9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom;
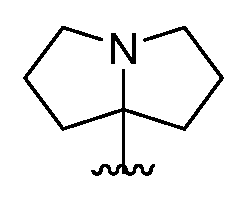
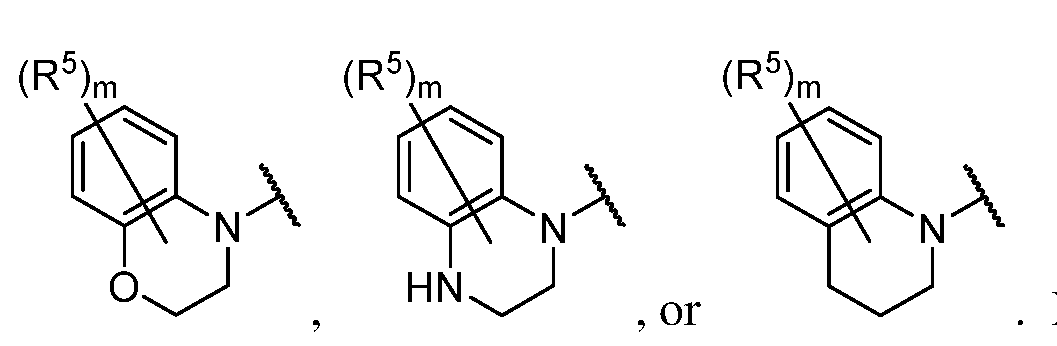
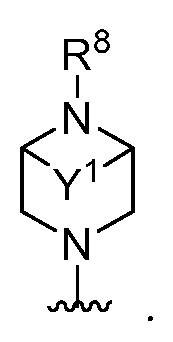
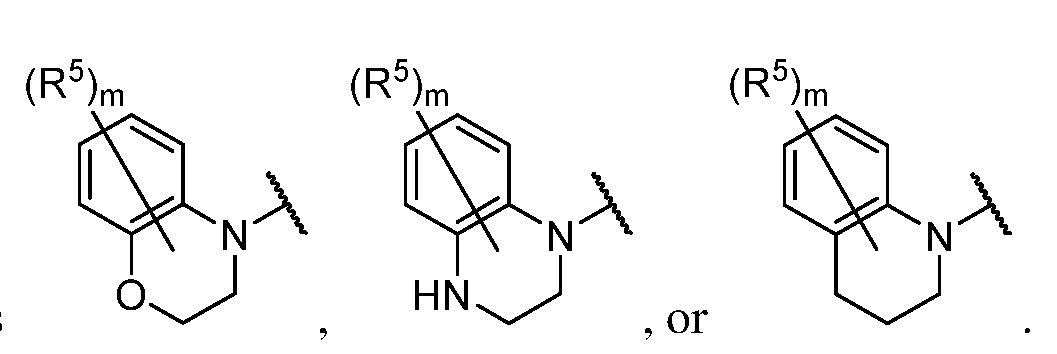
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences
- X 1 is C(H) 2 , C(R 4 )(H), or N(R 4 );
- X 2 is N or C(CN);
- X 3 is N or C(H);
- X 4 is C(O) or C(H) 2 ;
- L is a covalent bond or a C 1-4 bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R 9 )-, or -C(O)-;
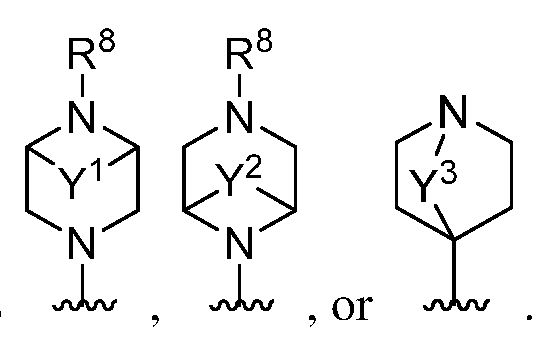
- Y 1 , Y 2 , and Y 3 represent independently a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -; and m, n, and p are each independently 0, 1, 2, or 3.
- the definition of a variable is a single chemical group selected from those chemical groups set forth above
- the definition of a variable is a collection of two or more of the chemical groups selected from those set forth above
- the compound is defined by a combination of variables in which the variables are defined by (i) or (ii).
- the compound is a compound of Formula I.
- R 1 is naphthyl; an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with m occurrences of R 5 .
- R 1 is naphthyl, an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur, or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R 5 .
- R 1 is phenyl or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R 5 .
- R 1 is naphthyl or phenyl; each of which is substituted with m occurrences of R 5 .
- R 1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R 5 .
- R 1 is naphthyl or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R 5 .
- R 1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur, or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R 5 .
- R 1 is naphthyl substituted with m occurrences of R 5 . In certain embodiments, R 1 is 1-naphthyl substituted with m occurrences of R 5 . In certain embodiments,
- R 1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with m occurrences of R 5 .
- R 1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with m occurrences of R 5 .
- R 1 is In certain embodiments, R 1 is
- R 1 is an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R 5 .
- R 1 is an 8-10 membered bicyclic heteroaryl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R 5 .
- R 1 is quinolinyl, isoquinolinyl, or indazolyl; each of which is substituted with m occurrences of R 5 .
- R 1 is quinolinyl substituted with m occurrences of R 5 .
- R 1 is quinoline-4-yl substituted with m occurrences of R 5 .
- R 1 is isoquinolinyl substituted with m occurrences of R 5 .
- R 1 is isoquinolin-l-yl substituted with m occurrences of R 5 .
- R 1 is indazolyl substituted with m occurrences of R 5 .
- R 1 is phenyl substituted with m occurrences of R 5 . In certain embodiments, R 1 is In certain embodiments, R 1 is [0089] In certain embodiments, R 1 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R 5 . In certain embodiments, R 1 is a 6-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with m occurrences of R 5 . In certain embodiments, R 1 is a 5-membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R 5 .
- R 1 is selected from the groups depicted in the compounds in Table 1, below. In certain embodiments, R 1 is selected from the groups depicted in the compounds in Table 1 or 1-A, below.
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci- 6 aliphatic; each of which is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R 6 .
- R 2 is phenyl or Ci- 6 aliphatic; each of which is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci- 6 aliphatic; each of which is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or Ci- 6 aliphatic; each of which is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 nitrogen atom; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated bicyclic heterocyclyl having 1 nitrogen atom; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated bicyclic heterocyclyl having 1 nitrogen atom; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur. In certain embodiments, R 2 is an 8-10 membered saturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur. In certain embodiments, R 2 is an 8-10 membered partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur.
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen. In certain embodiments, R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 nitrogen atom. In certain embodiments, R 2 is an 8-10 membered saturated bicyclic heterocyclyl having 1 nitrogen atom. In certain embodiments, R 2 is
- R 2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 4-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 6-membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is azetidinyl, pyrrolidinyl, piperidinyl, or morpholinyl; each of which is substituted with n occurrences of R 6 .
- R 2 is azetidinyl substituted with n occurrences of R 6 .
- R 2 is pyrrolidinyl substituted with n occurrences of R 6 .
- R 2 is pyrrolidin-2-yl substituted with n occurrences of R 6 .
- R 2 is pyrrolidin-2-yl substituted with a single occurrence of R 6 at the 1-position.
- R 2 is piperidinyl substituted with n occurrences of R 6 .
- R 2 is morpholinyl substituted with n occurrences of R 6 .
- R 2 is azetidinyl, pyrrolidinyl, piperidinyl, or morpholinyl. In certain embodiments, R 2 is azetidinyl. In certain embodiments, R 2 is pyrrolidinyl. In certain embodiments, R 2 is pyrrolidin-2-yl. In certain embodiments, R 2 is l-methyl-pyrrolidin-2-yl. In certain embodiments, R 2 is piperidinyl. In certain embodiments, R 2 is morpholinyl.
- R 2 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with n occurrences of R 6 .
- R 2 is phenyl substituted with n occurrences of R 6 . In certain embodiments, R 2 is phenyl.
- R 2 is Ci- 6 aliphatic substituted with n occurrences of R 6 . In certain embodiments, R 2 is C3-6 cycloaliphatic substituted with n occurrences of R 6 . In certain embodiments, R 2 is a Ci- 6 aliphatic chain substituted with n occurrences of R 6 . In certain embodiments, R 2 is C3-6 cycloalkyl substituted with n occurrences of R 6 . In certain embodiments, R 2 is Ci- 6 alkyl substituted with n occurrences of R 6 .
- R 2 is Ci- 6 aliphatic. In certain embodiments, R 2 is C3-6 cycloaliphatic. In certain embodiments, R 2 is a Ci- 6 aliphatic chain. In certain embodiments, R 2 is C3-6 cycloalkyl. In certain embodiments, R 2 is Ci- 6 alkyl.
- R 2 is selected from the groups depicted in the compounds in Table 1, below. In certain embodiments, R 2 is selected from the groups depicted in the compounds in Table 1 or 1-A, below.
- R 3 is hydrogen; or one occurrence of R 3 is hydrogen, and the other occurrence of R 3 is taken together with R 4 to form a double bond between the atoms to which R 3 and R 4 are attached; or both occurrences of R 3 are taken together to form an oxo substituent.
- R 3 is hydrogen.
- one occurrence of R 3 is hydrogen, and the other occurrence of R 3 is taken together with R 4 to form a double bond between the atoms to which R 3 and R 4 are attached.
- both occurrences of R 3 are taken together to form an oxo substituent.
- R 3 is selected from the groups depicted in the compounds in Table 1, below.
- R 4 is taken together with one occurrence of R 3 to form a double bond between the atoms to which R 3 and R 4 are attached.
- R 4 is selected from the groups depicted in the compounds in Table 1, below.
- R 5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, Ci- 6 alkyl, hydroxyl, Ci- 6 alkoxyl, halo, or cyano.
- R 5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, Ci- 6 alkyl, or hydroxyl. In certain embodiments, R 5 represents independently for each occurrence ethynyl, ethenyl, C1-3 alkyl, or hydroxyl. In certain embodiments, R 5 represents independently for each occurrence C2-6 alkynyl or hydroxyl. In certain embodiments, R 5 represents independently for each occurrence ethynyl or hydroxyl. In certain embodiments, one occurrence of R 5 is ethynyl. In certain embodiments, one occurrence of R 5 is hydroxyl.
- R 5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, or Ci- 6 alkyl. In certain embodiments, R 5 represents independently for each occurrence ethynyl, ethenyl, or C1-3 alkyl. In certain embodiments, R 5 represents independently for each occurrence hydroxyl or Ci- 6 alkoxyl. In certain embodiments, R 5 represents independently for each occurrence halo or cyano.
- R 5 represents independently for each occurrence C2-6 alkynyl. In certain embodiments, R 5 is ethynyl. In certain embodiments, R 5 represents independently for each occurrence C2-6 alkenyl. In certain embodiments, R 5 is ethenyl. In certain embodiments, R 5 represents independently for each occurrence Ci- 6 alkyl. In certain embodiments, R 5 represents independently for each occurrence C1-3 alkyl. In certain embodiments, R 5 is methyl. In certain embodiments, R 5 is hydroxyl. In certain embodiments, R 5 represents independently for each occurrence Ci- 6 alkoxyl. In certain embodiments, R 5 represents independently for each occurrence halo.
- R 5 represents independently for each occurrence fluoro or chloro. In certain embodiments, R 5 is fluoro. In certain embodiments, R 5 is chloro. In certain embodiments, R 5 is cyano. In certain embodiments, R 5 is selected from the groups depicted in the compounds in Table 1, below. In certain embodiments, R 5 is selected from the groups depicted in the compounds in Table 1 or 1-A, below.
- R 6 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 haloalkoxyl, Ci- 6 alkyl, Ci- 6 haloalkyl, Ci- 6 hydroxyalkyl, or -N(R 9 )2.
- R 6 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 alkyl, or Ci- 6 haloalkyl. In certain embodiments, R 6 represents independently for each occurrence halo, Ci- 6 alkyl, or Ci- 6 haloalkyl. In certain embodiments, R 6 represents independently for each occurrence halo or Ci- 6 alkyl. In certain embodiments, R 6 represents independently for each occurrence halo, hydroxyl, or Ci- 6 alkyl.
- R 6 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 haloalkoxyl, or -N(R 9 )2. In certain embodiments, R 6 represents independently for each occurrence halo, hydroxyl, or -N(R 9 )2. In certain embodiments, R 6 represents independently for each occurrence halo, Ci- 6 alkoxyl, or Ci- 6 haloalkoxyl. In certain embodiments, R 6 represents independently for each occurrence halo, Ci- 6 alkyl, Ci- 6 haloalkyl, or Ci- 6 hydroxyalkyl.
- R 6 represents independently for each occurrence Ci- 6 alkyl, Ci- 6 haloalkyl, or Ci- 6 hydroxyalkyl. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 alkyl or Ci- 6 haloalkyl.
- R 6 represents independently for each occurrence halo. In certain embodiments, R 6 represents independently for each occurrence fluoro or chloro. In certain embodiments, R 6 is fluoro. In certain embodiments, R 6 is chloro. In certain embodiments, R 6 is hydroxyl. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 alkoxyl. In certain embodiments, R 6 is methoxy. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 haloalkoxyl. In certain embodiments, R 6 is trifluoromethoxy. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 alkyl. In certain embodiments, R 6 represents independently for each occurrence C1-3 alkyl.
- R 6 is methyl. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 haloalkyl. In certain embodiments, R 6 is trifluoromethyl. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 hydroxyalkyl. In certain embodiments, R 6 represents independently for each occurrence -N(R 9 )2. In certain embodiments, R 6 is -NH2. In certain embodiments, R 6 is selected from the groups depicted in the compounds in Table 1, below. In certain embodiments, R 6 is selected from the groups depicted in the compounds in Table 1 or 1-A, below.
- R 7 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 haloalkoxyl, Ci- 6 alkyl, Ci- 6 haloalkyl, Ci- 6 hydroxyalkyl, or -N(R 9 )2.
- R 7 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 alkyl, or Ci- 6 haloalkyl. In certain embodiments, R 7 represents independently for each occurrence halo, Ci- 6 alkyl, or Ci- 6 haloalkyl. In certain embodiments, R 7 represents independently for each occurrence halo or Ci- 6 alkyl. In certain embodiments, R 7 represents independently for each occurrence halo, hydroxyl, or Ci- 6 alkyl.
- R 7 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 haloalkoxyl, or -N(R 9 )2. In certain embodiments, R 7 represents independently for each occurrence halo, hydroxyl, or -N(R 9 )2. In certain embodiments, R 7 represents independently for each occurrence halo, Ci- 6 alkoxyl, or Ci- 6 haloalkoxyl. In certain embodiments, R 7 represents independently for each occurrence halo, Ci- 6 alkyl, Ci- 6 haloalkyl, or Ci- 6 hydroxyalkyl.
- R 7 represents independently for each occurrence Ci- 6 alkyl, Ci- 6 haloalkyl, or Ci- 6 hydroxyalkyl. In certain embodiments, R 7 represents independently for each occurrence Ci- 6 alkyl or Ci- 6 haloalkyl.
- R 7 represents independently for each occurrence halo. In certain embodiments, R 7 represents independently for each occurrence fluoro or chloro. In certain embodiments, R 7 is fluoro. In certain embodiments, R 7 is chloro. In certain embodiments, R 7 is hydroxyl. In certain embodiments, R 7 represents independently for each occurrence Ci- 6 alkoxyl. In certain embodiments, R 7 is methoxy. In certain embodiments, R 7 represents independently for each occurrence Ci- 6 haloalkoxyl. In certain embodiments, R 7 is trifluoromethoxy. In certain embodiments, R 7 represents independently for each occurrence C1-6 alkyl. In certain embodiments, R 7 represents independently for each occurrence C1-3 alkyl.
- R 7 is methyl. In certain embodiments, R 7 represents independently for each occurrence Ci- 6 haloalkyl. In certain embodiments, R 7 is trifluoromethyl. In certain embodiments, R 7 represents independently for each occurrence Ci- 6 hydroxyalkyl. In certain embodiments, R 7 represents independently for each occurrence -N(R 9 )2. In certain embodiments, R 7 is -NH2. In certain embodiments, R 7 is selected from the groups depicted in the compounds in Table 1, below.
- R 8 is hydrogen, C1-4 alkyl, or C3-5 cycloalkyl. In certain embodiments, R 8 is hydrogen or C 1-4 alkyl. In certain embodiments, R 8 is hydrogen or methyl.
- R 8 is CIM alkyl or C3-5 cycloalkyl.
- R 8 is hydrogen. In certain embodiments, R 8 is CIM alkyl. In certain embodiments, R 8 is methyl. In certain embodiments, R 8 is C3-5 cycloalkyl. In certain embodiments, R 8 is cyclopropyl. In certain embodiments, R 8 is selected from the groups depicted in the compounds in Table 1, below.
- R 9 represents independently for each occurrence hydrogen, Ci-4 alkyl, or C3-5 cycloalkyl; or two occurrences of R 9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom.
- R 9 represents independently for each occurrence hydrogen, C1-4 alkyl, or C3-5 cycloalkyl. In certain embodiments, R 9 represents independently for each occurrence hydrogen or C1-4 alkyl. In certain embodiments, R 9 represents independently for each occurrence hydrogen or methyl. In certain embodiments, R 9 represents independently for each occurrence C1-4 alkyl or C3-5 cycloalkyl.
- two occurrences of R 9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom.
- R 9 is hydrogen. In certain embodiments, R 9 represents independently for each occurrence C M alkyl. In certain embodiments, R 9 is methyl. In certain embodiments, R 9 represents independently for each occurrence C3-5 cycloalkyl. In certain embodiments, R 9 is cyclopropyl. In certain embodiments, R 9 is selected from the groups depicted in the compounds in Table 1, below.
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R 7 ; or A 1 is , [0130] In certain embodiments, A 1 is In certain embodiments, A 1 is In certain embodiments, A 1 is In certain embodiments, A 1 is In certain embodiments, A 1 is
- a 1 is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5- 6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R 7 .
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R 7 ; or A 1 is
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R 7 .
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R 7 ; or A 1 is or
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R 7 .
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R 7 .
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5- 6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur.
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur.
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is 1,4-diazepanyl, piperidinyl, piperazinyl, pyrrolidinyl, or azetidinyl; each of which is substituted with p occurrences of R 7 .
- a 1 is 1,4-diazepanyl substituted with p occurrences of R 7 .
- a 1 is piperidinyl substituted with p occurrences of R 7 .
- a 1 is piperazinyl substituted with p occurrences of R 7 .
- a 1 is pyrrolidinyl substituted with p occurrences of R 7 .
- a 1 is azetidinyl substituted with p occurrences of R 7 .
- a 1 is 1,4-diazepanyl, piperidinyl, piperazinyl, pyrrolidinyl, or azetidinyl. In certain embodiments, A 1 is 1,4-diazepanyl. In certain embodiments, A 1 is piperidinyl. In certain embodiments, A 1 is piperazinyl. In certain embodiments, A 1 is pyrrolidinyl. In certain embodiments, A 1 is azetidinyl.
- a 1 is 1,4-diazepan-l-yl, piperidin-4-yl, piperazin-l-yl, pyrrolidin-3-yl, or azetidin-3-yl; each of which is substituted with p occurrences of R 7 .
- a 1 is 1,4-diazepan-l-yl substituted with p occurrences of R 7 .
- a 1 is piperidin-4-yl substituted with p occurrences of R 7 .
- a 1 is piperazin-l-yl substituted with p occurrences of R 7 .
- a 1 is pyrrolidin-3-yl substituted with p occurrences of R 7 .
- a 1 is azetidin-3-yl substituted with p occurrences of R 7 .
- a 1 is 1,4-diazepan-l-yl, piperidin-4-yl, piperazin-l-yl, pyrrolidin-3-yl, or azetidin-3-yl. In certain embodiments, A 1 is 1,4-diazepan-l-yl. In certain embodiments, A 1 is piperidin-4-yl. In certain embodiments, A 1 is piperazin-l-yl. In certain embodiments, A 1 is pyrrolidin-3-yl. In certain embodiments, A 1 is azetidin-3-yl.
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 7-10 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A 1 is a 7-10 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 2 nitrogen atoms. In certain embodiments, A 1 is or In certain embodiments, A 1 is In certain embodiments, A 1 i is
- a 1 is a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 7-10 membered saturated ori/zo-fused heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 6-11 membered saturated ori/zo-fused heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is a 7-10 membered saturated ori zo-fused heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with p occurrences of R 7 .
- a 1 is a 6-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with p occurrences of R 7 .
- a 1 is pyridinyl or pyrimidinyl; each of which is substituted with p occurrences of R 7 .
- a 1 is pyridinyl substituted with p occurrences of R 7 .
- a 1 is pyridin-4-yl substituted with p occurrences of R 7 .
- a 1 is pyrimidinyl substituted with p occurrences of R 7 .
- a 1 is a 5-membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with p occurrences of R 7 .
- a 1 is a 5-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with p occurrences of R 7 .
- a 1 is imidazolyl or pyrazolyl; each of which is substituted with p occurrences of R 7 .
- a 1 is imidazolyl substituted with p occurrences of R 7 .
- a 1 is pyrazolyl substituted with p occurrences of R 7 .
- a 1 is selected from the groups depicted in the compounds in Table 1, below.
- X 1 is C(H)2, C(R 4 )(H), or N(R 4 ). In certain embodiments,
- X 1 is C(H) 2 or C(R 4 )(H). In certain embodiments, X 1 is C(R 4 )(H) or N(R 4 ). In certain embodiments, X 1 is C(H)2. In certain embodiments, X 1 is C(R 4 )(H). In certain embodiments, X 1 is N(R 4 ). In certain embodiments, X 1 is selected from the groups depicted in the compounds in Table 1, below. [0156] As defined generally above, X 2 is N or C(CN). In certain embodiments, X 2 is N. In certain embodiments, X 2 is C(CN). In certain embodiments, X 2 is selected from the groups depicted in the compounds in Table 1, below.
- X 3 is N or C(H). In certain embodiments, X 3 is N. In certain embodiments, X 3 is C(H). In certain embodiments, X 3 is selected from the groups depicted in the compounds in Table 1, below.
- X 4 is C(O) or C(H)2. In certain embodiments, X 4 is C(O).
- X 4 is C(H)2. In certain embodiments, X 4 is selected from the groups depicted in the compounds in Table 1, below.
- L is a covalent bond or a Ci-4 bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R 9 )-, or -C(O)-. In certain embodiments, L is a covalent bond.
- L is a Ci-4 bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R 9 )-, or -C(O)-.
- L is a Ci-4 bivalent saturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R 9 )-, or -C(O)-.
- L is a C1-4 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R 9 )-, or - C(O)-. In certain embodiments, L is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -N(R 9 )-, or -C(O)-.
- L is a C 1-4 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -N(R 9 )-, or -C(O)-.
- L is a C 2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -N(R 9 )-, or -C(O)-.
- L is a Ci- 4 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -O- or -N(R 9 )-.
- L is a C 2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -O- or -N(R 9 )-.
- L is -0-CH 2 -, -0-(CH 2 ) 2 -, -N(R 9 )-CH 2 -, or -N(R 9 )-(CH 2 ) 2 -, wherein the terminal carbon atom of L is attached to R 2 .
- L is -0-CH 2 - or -0-(CH 2 ) 2 -, wherein the terminal carbon atom of L is attached to R 2 .
- L is -N(R 9 )-CH 2 - or -N(R 9 )-(CH 2 ) 2 -, wherein the terminal carbon atom of L is attached to R 2 .
- L is -0-CH 2 -, wherein the carbon atom of L is attached to R 2 .
- L is -0-(CH 2 ) 2 -, wherein the terminal carbon atom of L is attached to R 2 .
- L is -N(R 9 )-CH 2 -, wherein the carbon atom of L is attached to R 2 .
- L is -N(R 9 )-(CH 2 ) 2 -, wherein the terminal carbon atom of L is attached to R 2 .
- L is selected from the groups depicted in the compounds in Table 1, below.
- Y 1 is a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 1 is a C 2 -3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -. In certain embodiments, Y 1 is a C 2 -3 bivalent unsaturated, straight hydrocarbon chain.
- Y 1 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -. In certain embodiments, Y 1 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 1 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 1 is a C1-3 bivalent saturated, straight hydrocarbon chain.
- Y 1 is a C 2 -3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 1 is a C 2 -3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 1 is a C 2 -3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 1 is a C 2-3 bivalent saturated, straight hydrocarbon chain.
- Y 1 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) 3 -, -CH 2 -0- CH 2 -, or -CH 2 -CF 2 -CH 2 -.
- Y 1 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, - (CH 2 ) 3 -, or -CH 2 -CF 2 -CH 2 -.
- Y 1 is -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) 3 -, or -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 1 is -(CH 2 ) 2 - or -(CH 2 ) 3 -. In certain embodiments, Y 1 is -CH 2 -C(H)(OH)- or -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 1 is -(CH 2 )-, -(CH 2 ) 2 -, or - (CH 2 ) 3 -.
- Y 1 is -(CH 2 ) 2 - or -CH 2 -C(H)(OH)-. In certain embodiments, Y 1 is -(CH 2 ) 3 -, -CH 2 -0-CH 2 -, or -CH 2 -CF 2 -CH 2 -.
- Y 1 is -(CH 2 )-. In certain embodiments, Y 1 is -(CH 2 ) 2 -. In certain embodiments, Y 1 is -CH 2 -C(H)(OH)-. In certain embodiments, Y 1 is -(CH 2 ) 3 -. In certain embodiments, Y 1 is -CH 2 -0-CH 2 -. In certain embodiments, Y 1 is -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 1 is selected from the groups depicted in the compounds in Table 1, below.
- Y 2 is a C 1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 2 is a C 2-3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 2 is a C 2-3 bivalent unsaturated, straight hydrocarbon chain.
- Y 2 is a C 1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 2 is a C 1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 2 is a C 1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 2 is a C 1-3 bivalent saturated, straight hydrocarbon chain.
- Y 2 is a C 2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -. In certain embodiments, Y 2 is a C 2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 2 is a C 2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 2 is a C 2-3 bivalent saturated, straight hydrocarbon chain.
- Y 2 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) 3 -, -CH 2 -0- CH 2 -, or -CH 2 -CF 2 -CH 2 -.
- Y 2 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, - (CH 2 ) 3 -, or -CH 2 -CF 2 -CH 2 -.
- Y 2 is -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) 3 -, or -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 2 is -(CH 2 ) 2 - or -(CH 2 ) 3 -. In certain embodiments, Y 2 is -CH 2 -C(H)(OH)- or -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 2 is -(CH 2 )-, -(CH 2 ) 2 -, or - (CH 2 ) 3 -.
- Y 2 is -(CH 2 ) 2 - or -CH 2 -C(H)(OH)-. In certain embodiments, Y 2 is -(CH 2 ) 3 -, -CH 2 -0-CH 2 -, or -CH 2 -CF 2 -CH 2 -.
- Y 2 is -(CH 2 )-. In certain embodiments, Y 2 is -(CH 2 ) 2 -. In certain embodiments, Y 2 is -CH 2 -C(H)(OH)-. In certain embodiments, Y 2 is -(CH 2 ) 3 -. In certain embodiments, Y 2 is -CH 2 -0-CH 2 -. In certain embodiments, Y 2 is -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 2 is selected from the groups depicted in the compounds in Table 1, below.
- Y 3 is a C 1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 3 is a C 2-3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 3 is a C 2-3 bivalent unsaturated, straight hydrocarbon chain.
- Y 3 is a C 1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -. In certain embodiments, Y 3 is a C 1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 3 is a C 1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 3 is a C 1-3 bivalent saturated, straight hydrocarbon chain. [0179] In certain embodiments, Y 3 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 3 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R 7 )-, or -C(R 7 ) 2 -. In certain embodiments, Y 3 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 3 is a C2-3 bivalent saturated, straight hydrocarbon chain.
- Y 3 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) 3 -, -CH2-O- CH2-, or -CH2-CF2-CH2-.
- Y 3 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, - (CH 2 ) 3 -, or -CH2-CF2-CH2-.
- Y 3 is -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) 3 -, or -CH2-CF2-CH2-. In certain embodiments, Y 3 is -(CH2)2- or -(CH2)3-. In certain embodiments, Y 3 is -CH 2 -C(H)(OH)- or -CH2-CF2-CH2-. In certain embodiments, Y 3 is -(CH 2 )-, -(CH 2 ) 2 -, or - (CH2)3-. In certain embodiments, Y 3 is -(CH2)2- or -CH2-C(H)(OH)-. In certain embodiments, Y 3 is -(CH 2 ) 3 -, -CH2-O-CH2-, or -CH2-CF2-CH2-.
- Y 3 is -(CH2)-. In certain embodiments, Y 3 is -(CH2)2-. In certain embodiments, Y 3 is -CH2-C(H)(OH)-. In certain embodiments, Y 3 is -(CH2)3-. In certain embodiments, Y 3 is -CH2-O-CH2-. In certain embodiments, Y 3 is -CH2-CF2-CH2-. In certain embodiments, Y 3 is selected from the groups depicted in the compounds in Table 1, below.
- m is 0, 1, 2, or 3. In certain embodiments, m is 0. In certain embodiments, m is 1. In certain embodiments, m is 2. In certain embodiments, m is 3. In certain embodiments, m is 0 or 1. In certain embodiments, m is 1 or 2. In certain embodiments, m is 2 or 3. In certain embodiments, m is 0, 1, or 2. In certain embodiments, m is 1, 2, or 3. In certain embodiments, m is selected from the values represented in the compounds in Table 1, below.
- n is 0, 1, 2, or 3. In certain embodiments, n is 0. In certain embodiments, n is 1. In certain embodiments, n is 2. In certain embodiments, n is 3. In certain embodiments, n is 0 or 1. In certain embodiments, n is 1 or 2. In certain embodiments, n is 2 or 3. In certain embodiments, n is 0, 1, or 2. In certain embodiments, n is 1, 2, or 3. In certain embodiments, n is selected from the values represented in the compounds in Table 1, below. [0184] As defined generally above, p is 0, 1, 2, or 3. In certain embodiments, p is 0. In certain embodiments, p is 1. In certain embodiments, p is 2. In certain embodiments, p is 3.
- p is 0 or 1. In certain embodiments, p is 1 or 2. In certain embodiments, p is 2 or 3. In certain embodiments, p is 0, 1, or 2. In certain embodiments, p is 1, 2, or 3. In certain embodiments, p is selected from the values represented in the compounds in Table 1, below.
- Another aspect of the invention provides a compound represented by Formula II: or a pharmaceutically acceptable salt thereof; wherein:
- R 1 is naphthyl; an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with m occurrences of R 5 ;
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci- 6 aliphatic; each of which is substituted with n occurrences of R 6 ;
- R 3 is C M alkyl
- R 4 is hydrogen, halo, or C1-4 alkyl
- R 5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, Ci- 6 alkyl, hydroxyl, Ci- 6 alkoxyl, halo, or cyano;
- R 6 and R 7 each represent independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 haloalkoxyl, Ci- 6 alkyl, Ci- 6 haloalkyl, Ci- 6 hydroxyalkyl, or -N(R 9 )2;
- R 8 and R 9 each represent independently for each occurrence hydrogen, C 1-4 alkyl, or C3-5 cycloalkyl; or two occurrences of R 9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom;
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p
- X 1 is C(H) or N
- X 2 is N, C(CN), or C(R 4 );
- L is a covalent bond or a C1-4 bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R 9 )-, or -C(O)-;
- Y 1 , Y 2 , and Y 3 represent independently a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R 7 )-, or -C(R 7 )2-; and m, n, and p are each independently 0, 1, 2, or 3.
- the definitions of variables in Formula II above encompass multiple chemical groups.
- the definition of a variable is a single chemical group selected from those chemical groups set forth above
- the definition of a variable is a collection of two or more of the chemical groups selected from those set forth above
- the compound is defined by a combination of variables in which the variables are defined by (i) or (ii).
- the compound is a compound of Formula II.
- R 1 is naphthyl; an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with m occurrences of R 5 .
- R 1 is naphthyl, an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur, or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R 5 .
- R 1 is phenyl or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R 5 .
- R 1 is naphthyl or phenyl; each of which is substituted with m occurrences of R 5 .
- R 1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R 5 .
- R 1 is naphthyl or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R 5 .
- R 1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur, or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R 5 .
- R 1 is naphthyl substituted with m occurrences of R 5 . In certain embodiments, R 1 is 1-naphthyl substituted with m occurrences of R 5 . In certain embodiments,
- R 1 is In certain embodiments, R 1 is
- R 1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with m occurrences of R 5 .
- R 1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with m occurrences of R 5 .
- R 1 is certain embodiments, R 1 is In certain embodiments, R 1 is In certain embodiments, R 1 is In certain embodiments, R 1 is
- R 1 is an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R 5 .
- R 1 is an 8-10 membered bicyclic heteroaryl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R 5 .
- R 1 is quinolinyl, isoquinolinyl, or indazolyl; each of which is substituted with m occurrences of R 5 .
- R 1 is quinolinyl substituted with m occurrences of R 5 .
- R 1 is quinoline-4-yl substituted with m occurrences of R 5 .
- R 1 is isoquinolinyl substituted with m occurrences of R 5 .
- R 1 is isoquinolin-l-yl substituted with m occurrences of R 5 .
- R 1 is indazolyl substituted with m occurrences of R 5 .
- R 1 is phenyl substituted with m occurrences of R 5 . In certain embodiments, R 1 is In certain embodiments, R 1 is
- R 1 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R 5 .
- R 1 is a 6-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with m occurrences of R 5 .
- R 1 is a 5-membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R 5 .
- R 1 is selected from the groups depicted in the compounds in Table 2, below.
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci- 6 aliphatic; each of which is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R 6 .
- R 2 is phenyl or Ci- 6 aliphatic; each of which is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci- 6 aliphatic; each of which is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or Ci- 6 aliphatic; each of which is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 nitrogen atom; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated bicyclic heterocyclyl having 1 nitrogen atom; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated bicyclic heterocyclyl having 1 nitrogen atom; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur. In certain embodiments, R 2 is an 8-10 membered saturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur. In certain embodiments, R 2 is an 8-10 membered partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur.
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen. In certain embodiments, R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 nitrogen atom. In certain embodiments, R 2 is an 8-10 membered saturated bicyclic heterocyclyl having 1 nitrogen atom. In certain embodiments, R 2 is [0209] In certain embodiments, R 2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 4-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 6-membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is azetidinyl, pyrrolidinyl, piperidinyl, or morpholinyl; each of which is substituted with n occurrences of R 6 .
- R 2 is azetidinyl substituted with n occurrences of R 6 .
- R 2 is pyrrolidinyl substituted with n occurrences of R 6 .
- R 2 is pyrrolidin-2-yl substituted with n occurrences of R 6 .
- R 2 is pyrrolidin-2-yl substituted with a single occurrence of R 6 at the 1-position.
- R 2 is piperidinyl substituted with n occurrences of R 6 .
- R 2 is morpholinyl substituted with n occurrences of R 6 .
- R 2 is azetidinyl, pyrrolidinyl, piperidinyl, or morpholinyl. In certain embodiments, R 2 is azetidinyl. In certain embodiments, R 2 is pyrrolidinyl. In certain embodiments, R 2 is pyrrolidin-2-yl. In certain embodiments, R 2 is l-methyl-pyrrolidin-2-yl. In certain embodiments, R 2 is piperidinyl. In certain embodiments, R 2 is morpholinyl.
- R 2 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with n occurrences of R 6 .
- R 2 is phenyl substituted with n occurrences of R 6 . In certain embodiments, R 2 is phenyl.
- R 2 is Ci- 6 aliphatic substituted with n occurrences of R 6 . In certain embodiments, R 2 is C3-6 cycloaliphatic substituted with n occurrences of R 6 . In certain embodiments, R 2 is a Ci- 6 aliphatic chain substituted with n occurrences of R 6 . In certain embodiments, R 2 is C3-6 cycloalkyl substituted with n occurrences of R 6 . In certain embodiments, R 2 is Ci- 6 alkyl substituted with n occurrences of R 6 .
- R 2 is Ci- 6 aliphatic. In certain embodiments, R 2 is C3-6 cycloaliphatic. In certain embodiments, R 2 is a Ci- 6 aliphatic chain. In certain embodiments, R 2 is C3-6 cycloalkyl. In certain embodiments, R 2 is Ci- 6 alkyl.
- R 2 is selected from the groups depicted in the compounds in Table 2, below.
- R 3 is Ci-4 alkyl. In certain embodiments, R 3 is methyl. In certain embodiments, R 3 is selected from the groups depicted in the compounds in Table 2, below.
- R 4 is hydrogen, halo, or Ci-4 alkyl. In certain embodiments, R 4 is hydrogen. In certain embodiments, R 4 is halo. In certain embodiments, R 4 is fluoro or chloro. In certain embodiments, R 4 is fluoro. In certain embodiments, R 4 is chloro. In certain embodiments, R 4 is C M alkyl. In certain embodiments, R 4 is methyl. In certain embodiments,
- R 4 is selected from the groups depicted in the compounds in Table 2, below.
- R 5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, Ci- 6 alkyl, hydroxyl, Ci- 6 alkoxyl, halo, or cyano.
- R 5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, Ci- 6 alkyl, or hydroxyl. In certain embodiments, R 5 represents independently for each occurrence ethynyl, ethenyl, C1-3 alkyl, or hydroxyl. In certain embodiments, R 5 represents independently for each occurrence C2-6 alkynyl or hydroxyl. In certain embodiments, R 5 represents independently for each occurrence ethynyl or hydroxyl. In certain embodiments, one occurrence of R 5 is ethynyl. In certain embodiments, one occurrence of R 5 is hydroxyl.
- R 5 represents independently for each occurrence C 2-6 alkynyl, C 2-6 alkenyl, or Ci- 6 alkyl. In certain embodiments, R 5 represents independently for each occurrence ethynyl, ethenyl, or C 1-3 alkyl. In certain embodiments, R 5 represents independently for each occurrence hydroxyl or Ci- 6 alkoxyl. In certain embodiments, R 5 represents independently for each occurrence halo or cyano.
- R 5 represents independently for each occurrence C 2-6 alkynyl. In certain embodiments, R 5 is ethynyl. In certain embodiments, R 5 represents independently for each occurrence C 2-6 alkenyl. In certain embodiments, R 5 is ethenyl. In certain embodiments, R 5 represents independently for each occurrence Ci- 6 alkyl. In certain embodiments, R 5 represents independently for each occurrence C 1-3 alkyl. In certain embodiments, R 5 is methyl. In certain embodiments, R 5 is hydroxyl. In certain embodiments, R 5 represents independently for each occurrence Ci- 6 alkoxyl. In certain embodiments, R 5 represents independently for each occurrence halo.
- R 5 represents independently for each occurrence fluoro or chloro. In certain embodiments, R 5 is fluoro. In certain embodiments, R 5 is chloro. In certain embodiments, R 5 is cyano. In certain embodiments, R 5 is selected from the groups depicted in the compounds in Table 2, below.
- R 6 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 haloalkoxyl, Ci- 6 alkyl, Ci- 6 haloalkyl, Ci- 6 hydroxyalkyl, or -N(R 9 )2.
- R 6 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 alkyl, or Ci- 6 haloalkyl. In certain embodiments, R 6 represents independently for each occurrence halo, Ci- 6 alkyl, or Ci- 6 haloalkyl. In certain embodiments, R 6 represents independently for each occurrence halo or Ci- 6 alkyl. In certain embodiments, R 6 represents independently for each occurrence halo, hydroxyl, or Ci- 6 alkyl.
- R 6 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 haloalkoxyl, or -N(R 9 )2. In certain embodiments, R 6 represents independently for each occurrence halo, hydroxyl, or -N(R 9 )2. In certain embodiments, R 6 represents independently for each occurrence halo, Ci- 6 alkoxyl, or Ci- 6 haloalkoxyl. In certain embodiments, R 6 represents independently for each occurrence halo, Ci- 6 alkyl, Ci- 6 haloalkyl, or Ci- 6 hydroxyalkyl.
- R 6 represents independently for each occurrence Ci- 6 alkyl, Ci- 6 haloalkyl, or Ci- 6 hydroxyalkyl. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 alkyl or Ci- 6 haloalkyl.
- R 6 represents independently for each occurrence halo. In certain embodiments, R 6 represents independently for each occurrence fluoro or chloro. In certain embodiments, R 6 is fluoro. In certain embodiments, R 6 is chloro. In certain embodiments, R 6 is hydroxyl. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 alkoxyl. In certain embodiments, R 6 is methoxy. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 haloalkoxyl. In certain embodiments, R 6 is trifluoromethoxy. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 alkyl. In certain embodiments, R 6 represents independently for each occurrence C1-3 alkyl.
- R 6 is methyl. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 haloalkyl. In certain embodiments, R 6 is trifluoromethyl. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 hydroxyalkyl. In certain embodiments, R 6 represents independently for each occurrence -N(R 9 )2. In certain embodiments, R 6 is -NH2. In certain embodiments, R 6 is selected from the groups depicted in the compounds in Table 2, below.
- R 7 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 haloalkoxyl, Ci- 6 alkyl, Ci- 6 haloalkyl, Ci- 6 hydroxyalkyl, or -N(R 9 )2.
- R 7 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 alkyl, or Ci- 6 haloalkyl. In certain embodiments, R 7 represents independently for each occurrence halo, Ci- 6 alkyl, or Ci- 6 haloalkyl. In certain embodiments, R 7 represents independently for each occurrence halo or Ci- 6 alkyl. In certain embodiments, R 7 represents independently for each occurrence halo, hydroxyl, or Ci- 6 alkyl.
- R 7 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 haloalkoxyl, or -N(R 9 )2. In certain embodiments, R 7 represents independently for each occurrence halo, hydroxyl, or -N(R 9 )2. In certain embodiments, R 7 represents independently for each occurrence halo, Ci- 6 alkoxyl, or Ci- 6 haloalkoxyl. In certain embodiments, R 7 represents independently for each occurrence halo, Ci- 6 alkyl, Ci- 6 haloalkyl, or Ci- 6 hydroxyalkyl.
- R 7 represents independently for each occurrence Ci- 6 alkyl, Ci- 6 haloalkyl, or Ci- 6 hydroxyalkyl. In certain embodiments, R 7 represents independently for each occurrence Ci- 6 alkyl or Ci- 6 haloalkyl.
- R 7 represents independently for each occurrence halo. In certain embodiments, R 7 represents independently for each occurrence fluoro or chloro. In certain embodiments, R 7 is fluoro. In certain embodiments, R 7 is chloro. In certain embodiments, R 7 is hydroxyl. In certain embodiments, R 7 represents independently for each occurrence Ci- 6 alkoxyl. In certain embodiments, R 7 is methoxy. In certain embodiments, R 7 represents independently for each occurrence Ci- 6 haloalkoxyl. In certain embodiments, R 7 is trifluoromethoxy. In certain embodiments, R 7 represents independently for each occurrence Ci- 6 alkyl. In certain embodiments, R 7 represents independently for each occurrence C1-3 alkyl.
- R 7 is methyl. In certain embodiments, R 7 represents independently for each occurrence Ci- 6 haloalkyl. In certain embodiments, R 7 is trifluoromethyl. In certain embodiments, R 7 represents independently for each occurrence Ci- 6 hydroxyalkyl. In certain embodiments, R 7 represents independently for each occurrence -N(R 9 )2. In certain embodiments, R 7 is -NH2. In certain embodiments, R 7 is selected from the groups depicted in the compounds in Table 2, below.
- R 8 is hydrogen, C1-4 alkyl, or C3-5 cycloalkyl. In certain embodiments, R 8 is hydrogen or C 1-4 alkyl. In certain embodiments, R 8 is hydrogen or methyl.
- R 8 is C IM alkyl or C3-5 cycloalkyl.
- R 8 is hydrogen. In certain embodiments, R 8 is C IM alkyl. In certain embodiments, R 8 is methyl. In certain embodiments, R 8 is C3-5 cycloalkyl. In certain embodiments, R 8 is cyclopropyl. In certain embodiments, R 8 is selected from the groups depicted in the compounds in Table 2, below.
- R 9 represents independently for each occurrence hydrogen, Ci-4 alkyl, or C3-5 cycloalkyl; or two occurrences of R 9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom.
- R 9 represents independently for each occurrence hydrogen, C1-4 alkyl, or C3-5 cycloalkyl. In certain embodiments, R 9 represents independently for each occurrence hydrogen or C1-4 alkyl. In certain embodiments, R 9 represents independently for each occurrence hydrogen or methyl. In certain embodiments, R 9 represents independently for each occurrence C1-4 alkyl or C3-5 cycloalkyl.
- two occurrences of R 9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom.
- R 9 is hydrogen. In certain embodiments, R 9 represents independently for each occurrence C1-4 alkyl. In certain embodiments, R 9 is methyl. In certain embodiments, R 9 represents independently for each occurrence C3-5 cycloalkyl. In certain embodiments, R 9 is cyclopropyl. In certain embodiments, R 9 is selected from the groups depicted in the compounds in Table 2, below.
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R 7 ; or A 1 is , ,
- a 1 is In certain embodiments, A 1 is i n certain embodiments, A 1 is In certain embodiments, A 1 is
- a 1 is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5- 6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R 7 .
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R 7 ; or A 1 is [0243] In certain embodiments, A 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R 7 .
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R 7 ; or A 1 is or
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R 7 .
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R 7 .
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5- 6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur.
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur.
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is 1,4-diazepanyl, piperidinyl, piperazinyl, pyrrolidinyl, or azetidinyl; each of which is substituted with p occurrences of R 7 .
- a 1 is 1,4-diazepanyl substituted with p occurrences of R 7 .
- a 1 is piperidinyl substituted with p occurrences of R 7 .
- a 1 is piperazinyl substituted with p occurrences of R 7 .
- a 1 is pyrrolidinyl substituted with p occurrences of R 7 .
- a 1 is azetidinyl substituted with p occurrences of R 7 .
- a 1 is 1,4-diazepanyl, piperidinyl, piperazinyl, pyrrolidinyl, or azetidinyl. In certain embodiments, A 1 is 1,4-diazepanyl. In certain embodiments, A 1 is piperidinyl. In certain embodiments, A 1 is piperazinyl. In certain embodiments, A 1 is pyrrolidinyl. In certain embodiments, A 1 is azetidinyl.
- a 1 is 1,4-diazepan-l-yl, piperidin-4-yl, piperazin-l-yl, pyrrolidin-3-yl, or azetidin-3-yl; each of which is substituted with p occurrences of R 7 .
- a 1 is 1,4-diazepan-l-yl substituted with p occurrences of R 7 .
- a 1 is piperidin-4-yl substituted with p occurrences of R 7 .
- a 1 is piperazin-l-yl substituted with p occurrences of R 7 .
- a 1 is pyrrolidin-3-yl substituted with p occurrences of R 7 .
- a 1 is azetidin-3-yl substituted with p occurrences of R 7 .
- a 1 is 1,4-diazepan-l-yl, piperidin-4-yl, piperazin-l-yl, pyrrolidin-3-yl, or azetidin-3-yl. In certain embodiments, A 1 is 1,4-diazepan-l-yl. In certain embodiments, A 1 is piperidin-4-yl. In certain embodiments, A 1 is piperazin-l-yl. In certain embodiments, A 1 is pyrrolidin-3-yl. In certain embodiments, A 1 is azetidin-3-yl.
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 7-10 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A 1 is a 7-10 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 2 nitrogen atoms. In certain embodiments, A 1 is or In certain embodiments, A 1 is In certain embodiments, A 1 is
- a 1 is a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 7-10 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is a 7-10 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with p occurrences of R 7 .
- a 1 is a 6-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with p occurrences of R 7 .
- a 1 is pyridinyl or pyrimidinyl; each of which is substituted with p occurrences of R 7 .
- a 1 is pyridinyl substituted with p occurrences of R 7 .
- a 1 is pyridin-4-yl substituted with p occurrences of R 7 .
- a 1 is pyrimidinyl substituted with p occurrences of R 7 .
- a 1 is a 5-membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with p occurrences of R 7 .
- a 1 is a 5-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with p occurrences of R 7 .
- a 1 is imidazolyl or pyrazolyl; each of which is substituted with p occurrences of R 7 .
- a 1 is imidazolyl substituted with p occurrences of R 7 .
- a 1 is pyrazolyl substituted with p occurrences of R 7 .
- a 1 is selected from the groups depicted in the compounds in Table 2, below.
- X 1 is C(H) or N. In certain embodiments, X 1 is C(H). In certain embodiments, X 1 is N. In certain embodiments, X 1 is selected from the groups depicted in the compounds in Table 2, below.
- X 2 is N, C(CN), or C(R 4 ). In certain embodiments, X 2 is N. In certain embodiments, X 2 is C(CN) or C(R 4 ). In certain embodiments, X 2 is C(CN) or C(H).
- X 2 is C(CN). In certain embodiments, X 2 is C(R 4 ). In certain embodiments, X 2 is C(H). In certain embodiments, X 2 is selected from the groups depicted in the compounds in Table 2, below.
- L is a covalent bond or a Ci-4 bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R 9 )-, or -C(O)-. In certain embodiments, L is a covalent bond.
- L is a Ci-4 bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R 9 )-, or -C(O)-.
- L is a Ci-4 bivalent saturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R 9 )-, or -C(O)-.
- L is a CM bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R 9 )-, or - C(O)-.
- L is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -N(R 9 )-, or -C(O)-.
- L is a C1-4 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -N(R 9 )-, or -C(O)-.
- L is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -N(R 9 )-, or -C(O)-.
- L is a Ci- 4 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -O- or -N(R 9 )-.
- L is a C 2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -O- or -N(R 9 )-.
- L is -O-CH2-, -0-(CH 2 ) 2 -, -N(R 9 )-CH 2 -, or -N(R 9 )-(CH 2 ) 2 -, wherein the terminal carbon atom of L is attached to R 2 .
- L is -O-CH2- or -0-(CH 2 ) 2 -, wherein the terminal carbon atom of L is attached to R 2 .
- L is -N(R 9 )-CH2- or -N(R 9 )-(CH2)2-, wherein the terminal carbon atom of L is attached to R 2 .
- L is -O-CH2-, wherein the carbon atom of L is attached to R 2 .
- L is -0-(CH 2 ) 2 -, wherein the terminal carbon atom of L is attached to R 2 .
- L is -N(R 9 )-CH2-, wherein the carbon atom of L is attached to R 2 .
- L is -N(R 9 )-(CH2)2-, wherein the terminal carbon atom of L is attached to R 2 .
- L is selected from the groups depicted in the compounds in Table 2, below.
- Y 1 is a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -O-, -C(H)(R 7 )-, or -C(R 7 )2-.
- Y 1 is a C 2-3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -O-, -C(H)(R 7 )-, or -C(R 7 ) 2 -. In certain embodiments, Y 1 is a C 2-3 bivalent unsaturated, straight hydrocarbon chain.
- Y 1 is a C 1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - O-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 1 is a C 1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -O-, - C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 1 is a C 1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 1 is a C 1-3 bivalent saturated, straight hydrocarbon chain. [0275] In certain embodiments, Y 1 is a C 2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 1 is a C 2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R 7 )-, or -C(R 7 ) 2 -. In certain embodiments, Y 1 is a C 2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 1 is a C 2-3 bivalent saturated, straight hydrocarbon chain.
- Y 1 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) 3 -, -CH 2 -O- CH 2 -, or -CH 2 -CF 2 -CH 2 -.
- Y 1 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, - (CH 2 ) 3 -, or -CH2-CF2-CH2-.
- Y 1 is -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) 3 -, or -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 1 is -(CH2) 2 - or -(CH2) 3 -. In certain embodiments, Y 1 is -CH 2 -C(H)(OH)- or -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 1 is -(CH 2 )-, -(CH 2 ) 2 -, or - (CH2) 3 -.
- Y 1 is -(CH 2 ) 2 - or -CH 2 -C(H)(OH)-. In certain embodiments, Y 1 is -(CH 2 ) 3 -, -CH2-O-CH2-, or -CH2-CF2-CH2-.
- Y 1 is -(CH2)-. In certain embodiments, Y 1 is -(CH2) 2 -. In certain embodiments, Y 1 is -CH 2 -C(H)(OH)-. In certain embodiments, Y 1 is -(CH2) 3 -. In certain embodiments, Y 1 is -CH 2 -O-CH 2 -. In certain embodiments, Y 1 is -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 1 is selected from the groups depicted in the compounds in Table 2, below.
- Y 2 is a C 1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 2 is a C 2-3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 2 is a C 2-3 bivalent unsaturated, straight hydrocarbon chain.
- Y 2 is a C 1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 2 is a C 1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 2 is a C 1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 2 is a C1-3 bivalent saturated, straight hydrocarbon chain.
- Y 2 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 2 is a C 2 -3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 2 is a C 2 -3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 2 is a C 2 -3 bivalent saturated, straight hydrocarbon chain.
- Y 2 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) -, -CH 2 -0- CH 2 -, or -CH 2 -CF 2 -CH 2 -.
- Y 2 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, - (CH 2 ) -, or -CH 2 -CF 2 -CH 2 -.
- Y 2 is -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) -, or -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 2 is -(CH 2 ) 2 - or -(CH 2 )3-. In certain embodiments, Y 2 is -CH 2 -C(H)(OH)- or -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 2 is -(CH 2 )-, -(CH 2 ) 2 -, or - (CH 2 ) -.
- Y 2 is -(CH 2 ) 2 - or -CH 2 -C(H)(OH)-. In certain embodiments, Y 2 is -(CH 2 ) -, -CH 2 -0-CH 2 -, or -CH 2 -CF 2 -CH 2 -.
- Y 2 is -(CH 2 )-. In certain embodiments, Y 2 is -(CH 2 ) 2 -. In certain embodiments, Y 2 is -CH 2 -C(H)(OH)-. In certain embodiments, Y 2 is -(CH 2 )3-. In certain embodiments, Y 2 is -CH 2 -0-CH 2 -. In certain embodiments, Y 2 is -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 2 is selected from the groups depicted in the compounds in Table 2, below.
- Y 3 is a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 3 is a C 2 -3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 3 is a C 2 -3 bivalent unsaturated, straight hydrocarbon chain.
- Y 3 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -. In certain embodiments, Y 3 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 3 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 3 is a C1-3 bivalent saturated, straight hydrocarbon chain.
- Y 3 is a C 2 -3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -. In certain embodiments, Y 3 is a C 2 -3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 3 is a C 2 -3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 3 is a C 2 -3 bivalent saturated, straight hydrocarbon chain.
- Y 3 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) 3 -, -CH 2 -0- CH 2 -, or -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 3 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, - (CH 2 )3-, or -CH 2 -CF 2 -CH 2 -.
- Y 3 is -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) 3 -, or -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 3 is -(CH 2 ) 2 - or -(CH 2 )3-. In certain embodiments, Y 3 is -CH 2 -C(H)(OH)- or -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 3 is -(CH 2 )-, -(CH 2 ) 2 -, or - (CH 2 ) 3 -.
- Y 3 is -(CH 2 ) 2 - or -CH 2 -C(H)(OH)-. In certain embodiments, Y 3 is -(CH 2 ) 3 -, -CH 2 -0-CH 2 -, or -CH 2 -CF 2 -CH 2 -.
- Y 3 is -(CH 2 )-. In certain embodiments, Y 3 is -(CH 2 ) 2 -. In certain embodiments, Y 3 is -CH 2 -C(H)(OH)-. In certain embodiments, Y 3 is -(CH 2 )3-. In certain embodiments, Y 3 is -CH 2 -0-CH 2 -. In certain embodiments, Y 3 is -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 3 is selected from the groups depicted in the compounds in Table 2, below.
- m is 0, 1, 2, or 3. In certain embodiments, m is 0. In certain embodiments, m is 1. In certain embodiments, m is 2. In certain embodiments, m is 3. In certain embodiments, m is 0 or 1. In certain embodiments, m is 1 or 2. In certain embodiments, m is 2 or 3. In certain embodiments, m is 0, 1, or 2. In certain embodiments, m is 1, 2, or 3. In certain embodiments, m is selected from the values represented in the compounds in Table 2, below.
- n is 0, 1, 2, or 3. In certain embodiments, n is 0. In certain embodiments, n is 1. In certain embodiments, n is 2. In certain embodiments, n is 3. In certain embodiments, n is 0 or 1. In certain embodiments, n is 1 or 2. In certain embodiments, n is 2 or 3. In certain embodiments, n is 0, 1, or 2. In certain embodiments, n is 1, 2, or 3. In certain embodiments, n is selected from the values represented in the compounds in Table 2, below.
- p is 0, 1, 2, or 3. In certain embodiments, p is 0. In certain embodiments, p is 1. In certain embodiments, p is 2. In certain embodiments, p is 3. In certain embodiments, p is 0 or 1. In certain embodiments, p is 1 or 2. In certain embodiments, p is 2 or 3. In certain embodiments, p is 0, 1, or 2. In certain embodiments, p is 1, 2, or 3. In certain embodiments, p is selected from the values represented in the compounds in Table 2, below.
- Another aspect of the invention provides a compound represented by Formula III: or a pharmaceutically acceptable salt thereof; wherein:
- R 1 is naphthyl; an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with m occurrences of R 5 ;
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci- 6 aliphatic; each of which is substituted with n occurrences of R 6 ;
- R 4 is hydrogen, halo, or C1-4 alkyl
- R 5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, Ci- 6 alkyl, hydroxyl, Ci- 6 alkoxyl, halo, or cyano;
- R 6 and R 7 each represent independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 haloalkoxyl, Ci- 6 alkyl, Ci- 6 haloalkyl, Ci- 6 hydroxyalkyl, or -N(R 9 )2;
- R 8 and R 9 each represent independently for each occurrence hydrogen, C1-4 alkyl, or C3-5 cycloalkyl; or two occurrences of R 9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom;
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p
- X 2 is N, C(CN), or C(R 4 );
- L is a covalent bond or a CM bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R 9 )-, or -C(O)-;
- Y 1 , Y 2 , and Y 3 represent independently a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R 7 )-, or -C(R 7 )2-; and m, n, and p are each independently 0, 1, 2, or 3.
- the definitions of variables in Formula III above encompass multiple chemical groups.
- the definition of a variable is a single chemical group selected from those chemical groups set forth above
- the definition of a variable is a collection of two or more of the chemical groups selected from those set forth above
- the compound is defined by a combination of variables in which the variables are defined by (i) or (ii).
- the compound is a compound of Formula III.
- R 1 is naphthyl; an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with m occurrences of R 5 .
- R 1 is naphthyl, an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur, or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R 5 .
- R 1 is phenyl or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R 5 .
- R 1 is naphthyl or phenyl; each of which is substituted with m occurrences of R 5 .
- R 1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R 5 .
- R 1 is naphthyl or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R 5 .
- R 1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur, or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R 5 .
- R 1 is naphthyl substituted with m occurrences of R 5 . In certain embodiments, R 1 is 1-naphthyl substituted with m occurrences of R 5 . In certain embodiments,
- R 1 is In certain embodiments, R 1 is
- R 1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with m occurrences of R 5 .
- R 1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with m occurrences of R 5 .
- R 1 is certain embodiments, R 1 is In certain embodiments, R 1 is In certain embodiments, R 1 is In certain embodiments, R 1 is
- R 1 is an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R 5 .
- R 1 is an 8-10 membered bicyclic heteroaryl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R 5 .
- R 1 is quinolinyl, isoquinolinyl, or indazolyl; each of which is substituted with m occurrences of R 5 .
- R 1 is quinolinyl substituted with m occurrences of R 5 .
- R 1 is quinoline-4-yl substituted with m occurrences of R 5 .
- R 1 is isoquinolinyl substituted with m occurrences of R 5 .
- R 1 is isoquinolin-l-yl substituted with m occurrences of R 5 .
- R 1 is indazolyl substituted with m occurrences of R 5 .
- R 1 is phenyl substituted with m occurrences of R 5 . In certain embodiments, R 1 is In certain embodiments, R 1 is
- R 1 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R 5 .
- R 1 is a 6-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with m occurrences of R 5 .
- R 1 is a 5-membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R 5 .
- R 1 is selected from the groups depicted in the compounds in Table 3, below.
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci- 6 aliphatic; each of which is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R 6 .
- R 2 is phenyl or Ci- 6 aliphatic; each of which is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci- 6 aliphatic; each of which is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or Ci- 6 aliphatic; each of which is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 nitrogen atom; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated bicyclic heterocyclyl having 1 nitrogen atom; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated bicyclic heterocyclyl having 1 nitrogen atom; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is substituted with n occurrences of R 6 .
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur. In certain embodiments, R 2 is an 8-10 membered saturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur. In certain embodiments, R 2 is an 8-10 membered partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur.
- R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen. In certain embodiments, R 2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 nitrogen atom. In certain embodiments, R 2 is an 8-10 membered saturated bicyclic heterocyclyl having 1 nitrogen atom. In certain embodiments, R 2 is [0317] In certain embodiments, R 2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 4-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 6-membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R 6 .
- R 2 is azetidinyl, pyrrolidinyl, piperidinyl, or morpholinyl; each of which is substituted with n occurrences of R 6 .
- R 2 is azetidinyl substituted with n occurrences of R 6 .
- R 2 is pyrrolidinyl substituted with n occurrences of R 6 .
- R 2 is pyrrolidin-2-yl substituted with n occurrences of R 6 .
- R 2 is pyrrolidin-2-yl substituted with a single occurrence of R 6 at the 1-position.
- R 2 is piperidinyl substituted with n occurrences of R 6 .
- R 2 is morpholinyl substituted with n occurrences of R 6 .
- R 2 is azetidinyl, pyrrolidinyl, piperidinyl, or morpholinyl. In certain embodiments, R 2 is azetidinyl. In certain embodiments, R 2 is pyrrolidinyl. In certain embodiments, R 2 is pyrrolidin-2-yl. In certain embodiments, R 2 is l-methyl-pyrrolidin-2-yl. In certain embodiments, R 2 is piperidinyl. In certain embodiments, R 2 is morpholinyl.
- R 2 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with n occurrences of R 6 .
- R 2 is phenyl substituted with n occurrences of R 6 . In certain embodiments, R 2 is phenyl.
- R 2 is Ci- 6 aliphatic substituted with n occurrences of R 6 . In certain embodiments, R 2 is C3-6 cycloaliphatic substituted with n occurrences of R 6 . In certain embodiments, R 2 is a Ci- 6 aliphatic chain substituted with n occurrences of R 6 . In certain embodiments, R 2 is C3-6 cycloalkyl substituted with n occurrences of R 6 . In certain embodiments, R 2 is Ci- 6 alkyl substituted with n occurrences of R 6 .
- R 2 is Ci- 6 aliphatic. In certain embodiments, R 2 is C3-6 cycloaliphatic. In certain embodiments, R 2 is a Ci- 6 aliphatic chain. In certain embodiments, R 2 is C3-6 cycloalkyl. In certain embodiments, R 2 is Ci- 6 alkyl.
- R 2 is selected from the groups depicted in the compounds in Table 3, below.
- R 4 is hydrogen, halo, or Ci- 4 alkyl. In certain embodiments, R 4 is hydrogen. In certain embodiments, R 4 is halo. In certain embodiments, R 4 is fluoro or chloro. In certain embodiments, R 4 is fluoro. In certain embodiments, R 4 is chloro. In certain embodiments, R 4 is C 1-4 alkyl. In certain embodiments, R 4 is methyl. In certain embodiments,
- R 4 is selected from the groups depicted in the compounds in Table 3, below.
- R 5 represents independently for each occurrence C 2-6 alkynyl, C 2-6 alkenyl, C1-6 alkyl, hydroxyl, C1-6 alkoxyl, halo, or cyano.
- R 5 represents independently for each occurrence C 2-6 alkynyl, C 2-6 alkenyl, C1-6 alkyl, or hydroxyl. In certain embodiments, R 5 represents independently for each occurrence ethynyl, ethenyl, C 1-3 alkyl, or hydroxyl. In certain embodiments, R 5 represents independently for each occurrence C 2-6 alkynyl or hydroxyl. In certain embodiments, R 5 represents independently for each occurrence ethynyl or hydroxyl. In certain embodiments, one occurrence of R 5 is ethynyl. In certain embodiments, one occurrence of R 5 is hydroxyl.
- R 5 represents independently for each occurrence C 2-6 alkynyl, C 2-6 alkenyl, or C1-6 alkyl. In certain embodiments, R 5 represents independently for each occurrence ethynyl, ethenyl, or C 1-3 alkyl. In certain embodiments, R 5 represents independently for each occurrence hydroxyl or C1-6 alkoxyl. In certain embodiments, R 5 represents independently for each occurrence halo or cyano.
- R 5 represents independently for each occurrence C 2-6 alkynyl. In certain embodiments, R 5 is ethynyl. In certain embodiments, R 5 represents independently for each occurrence C 2-6 alkenyl. In certain embodiments, R 5 is ethenyl. In certain embodiments, R 5 represents independently for each occurrence Ci-6 alkyl. In certain embodiments, R 5 represents independently for each occurrence C 1-3 alkyl. In certain embodiments, R 5 is methyl. In certain embodiments, R 5 is hydroxyl. In certain embodiments, R 5 represents independently for each occurrence C1-6 alkoxyl. In certain embodiments, R 5 represents independently for each occurrence halo.
- R 5 represents independently for each occurrence fluoro or chloro. In certain embodiments, R 5 is fluoro. In certain embodiments, R 5 is chloro. In certain embodiments, R 5 is cyano. In certain embodiments, R 5 is selected from the groups depicted in the compounds in Table 3, below. [0332] As defined generally above, R 6 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 haloalkoxyl, Ci- 6 alkyl, Ci- 6 haloalkyl, Ci- 6 hydroxyalkyl, or -N(R 9 )2.
- R 6 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 alkyl, or Ci- 6 haloalkyl. In certain embodiments, R 6 represents independently for each occurrence halo, Ci- 6 alkyl, or Ci- 6 haloalkyl. In certain embodiments, R 6 represents independently for each occurrence halo or Ci- 6 alkyl. In certain embodiments, R 6 represents independently for each occurrence halo, hydroxyl, or Ci- 6 alkyl.
- R 6 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 haloalkoxyl, or -N(R 9 )2. In certain embodiments, R 6 represents independently for each occurrence halo, hydroxyl, or -N(R 9 )2. In certain embodiments, R 6 represents independently for each occurrence halo, Ci- 6 alkoxyl, or Ci- 6 haloalkoxyl. In certain embodiments, R 6 represents independently for each occurrence halo, Ci- 6 alkyl, Ci- 6 haloalkyl, or Ci- 6 hydroxyalkyl.
- R 6 represents independently for each occurrence Ci- 6 alkyl, Ci- 6 haloalkyl, or Ci- 6 hydroxyalkyl. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 alkyl or Ci- 6 haloalkyl.
- R 6 represents independently for each occurrence halo. In certain embodiments, R 6 represents independently for each occurrence fluoro or chloro. In certain embodiments, R 6 is fluoro. In certain embodiments, R 6 is chloro. In certain embodiments, R 6 is hydroxyl. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 alkoxyl. In certain embodiments, R 6 is methoxy. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 haloalkoxyl. In certain embodiments, R 6 is trifluoromethoxy. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 alkyl. In certain embodiments, R 6 represents independently for each occurrence C1-3 alkyl.
- R 6 is methyl. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 haloalkyl. In certain embodiments, R 6 is trifluoromethyl. In certain embodiments, R 6 represents independently for each occurrence Ci- 6 hydroxyalkyl. In certain embodiments, R 6 represents independently for each occurrence -N(R 9 )2. In certain embodiments, R 6 is -NH2. In certain embodiments, R 6 is selected from the groups depicted in the compounds in Table 3, below.
- R 7 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 haloalkoxyl, Ci- 6 alkyl, Ci- 6 haloalkyl, Ci- 6 hydroxyalkyl, or -N(R 9 )2.
- R 7 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 alkyl, or Ci- 6 haloalkyl. In certain embodiments, R 7 represents independently for each occurrence halo, Ci- 6 alkyl, or Ci- 6 haloalkyl. In certain embodiments, R 7 represents independently for each occurrence halo or Ci- 6 alkyl. In certain embodiments, R 7 represents independently for each occurrence halo, hydroxyl, or Ci- 6 alkyl.
- R 7 represents independently for each occurrence halo, hydroxyl, Ci- 6 alkoxyl, Ci- 6 haloalkoxyl, or -N(R 9 )2. In certain embodiments, R 7 represents independently for each occurrence halo, hydroxyl, or -N(R 9 )2. In certain embodiments, R 7 represents independently for each occurrence halo, Ci- 6 alkoxyl, or Ci- 6 haloalkoxyl. In certain embodiments, R 7 represents independently for each occurrence halo, Ci- 6 alkyl, Ci- 6 haloalkyl, or Ci- 6 hydroxyalkyl.
- R 7 represents independently for each occurrence Ci- 6 alkyl, Ci- 6 haloalkyl, or Ci- 6 hydroxyalkyl. In certain embodiments, R 7 represents independently for each occurrence Ci- 6 alkyl or Ci- 6 haloalkyl.
- R 7 represents independently for each occurrence halo. In certain embodiments, R 7 represents independently for each occurrence fluoro or chloro. In certain embodiments, R 7 is fluoro. In certain embodiments, R 7 is chloro. In certain embodiments, R 7 is hydroxyl. In certain embodiments, R 7 represents independently for each occurrence Ci- 6 alkoxyl. In certain embodiments, R 7 is methoxy. In certain embodiments, R 7 represents independently for each occurrence Ci- 6 haloalkoxyl. In certain embodiments, R 7 is trifluoromethoxy. In certain embodiments, R 7 represents independently for each occurrence Ci- 6 alkyl. In certain embodiments, R 7 represents independently for each occurrence C1-3 alkyl.
- R 7 is methyl. In certain embodiments, R 7 represents independently for each occurrence Ci- 6 haloalkyl. In certain embodiments, R 7 is trifluoromethyl. In certain embodiments, R 7 represents independently for each occurrence Ci- 6 hydroxyalkyl. In certain embodiments, R 7 represents independently for each occurrence -N(R 9 )2. In certain embodiments, R 7 is -NH2. In certain embodiments, R 7 is selected from the groups depicted in the compounds in Table 3, below. [0340] As defined generally above, R 8 is hydrogen, Ci alkyl, or C3-5 cycloalkyl. In certain embodiments, R 8 is hydrogen or C M alkyl. In certain embodiments, R 8 is hydrogen or methyl.
- R 8 is C IM alkyl or C3-5 cycloalkyl.
- R 8 is hydrogen. In certain embodiments, R 8 is C IM alkyl. In certain embodiments, R 8 is methyl. In certain embodiments, R 8 is C3-5 cycloalkyl. In certain embodiments, R 8 is cyclopropyl. In certain embodiments, R 8 is selected from the groups depicted in the compounds in Table 3, below.
- R 9 represents independently for each occurrence hydrogen, Ci-4 alkyl, or C3-5 cycloalkyl; or two occurrences of R 9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom.
- R 9 represents independently for each occurrence hydrogen, C1-4 alkyl, or C3-5 cycloalkyl. In certain embodiments, R 9 represents independently for each occurrence hydrogen or C1-4 alkyl. In certain embodiments, R 9 represents independently for each occurrence hydrogen or methyl. In certain embodiments, R 9 represents independently for each occurrence C1-4 alkyl or C3-5 cycloalkyl.
- two occurrences of R 9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom.
- R 9 is hydrogen. In certain embodiments, R 9 represents independently for each occurrence C1-4 alkyl. In certain embodiments, R 9 is methyl. In certain embodiments, R 9 represents independently for each occurrence C3-5 cycloalkyl. In certain embodiments, R 9 is cyclopropyl. In certain embodiments, R 9 is selected from the groups depicted in the compounds in Table 3, below.
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R 7 ; or A 1 is
- a 1 is In certain embodiments, A 1 is In certain embodiments, A 1 is In certain embodiments, A 1 is in certain embodiments, A 1 is
- a 1 is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5- 6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R 7 .
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R 7 ; or A 1 is [0350] In certain embodiments, A 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R 7 .
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R 7 ; or A 1 is or
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R 7 .
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R 7 .
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5- 6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur.
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur.
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is 1,4-diazepanyl, piperidinyl, piperazinyl, pyrrolidinyl, or azetidinyl; each of which is substituted with p occurrences of R 7 .
- a 1 is 1,4-diazepanyl substituted with p occurrences of R 7 .
- a 1 is piperidinyl substituted with p occurrences of R 7 .
- a 1 is piperazinyl substituted with p occurrences of R 7 .
- a 1 is pyrrolidinyl substituted with p occurrences of R 7 .
- a 1 is azetidinyl substituted with p occurrences of R 7 .
- a 1 is 1,4-diazepanyl, piperidinyl, piperazinyl, pyrrolidinyl, or azetidinyl. In certain embodiments, A 1 is 1,4-diazepanyl. In certain embodiments, A 1 is piperidinyl. In certain embodiments, A 1 is piperazinyl. In certain embodiments, A 1 is pyrrolidinyl. In certain embodiments, A 1 is azetidinyl.
- a 1 is 1,4-diazepan-l-yl, piperidin-4-yl, piperazin-l-yl, pyrrolidin-3-yl, or azetidin-3-yl; each of which is substituted with p occurrences of R 7 .
- a 1 is 1,4-diazepan-l-yl substituted with p occurrences of R 7 .
- a 1 is piperidin-4-yl substituted with p occurrences of R 7 .
- a 1 is piperazin-l-yl substituted with p occurrences of R 7 .
- a 1 is pyrrolidin-3-yl substituted with p occurrences of R 7 .
- a 1 is azetidin-3-yl substituted with p occurrences of R 7 .
- a 1 is 1,4-diazepan-l-yl, piperidin-4-yl, piperazin-l-yl, pyrrolidin-3-yl, or azetidin-3-yl. In certain embodiments, A 1 is 1,4-diazepan-l-yl. In certain embodiments, A 1 is piperidin-4-yl. In certain embodiments, A 1 is piperazin-l-yl. In certain embodiments, A 1 is pyrrolidin-3-yl. In certain embodiments, A 1 is azetidin-3-yl.
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 7-10 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A 1 is a 7-10 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A 1 is a 6-11 membered saturated spirocyclic heterocyclyl having 2 nitrogen atoms. In certain embodiments, A 1 is or In certain embodiments, A 1 is In certain embodiments, A 1 i s
- a 1 is a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 7-10 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R 7 .
- a 1 is a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is a 7-10 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
- a 1 is a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with p occurrences of R 7 .
- a 1 is a 6-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with p occurrences of R 7 .
- a 1 is pyridinyl or pyrimidinyl; each of which is substituted with p occurrences of R 7 .
- a 1 is pyridinyl substituted with p occurrences of R 7 .
- a 1 is pyridin-4-yl substituted with p occurrences of R 7 .
- a 1 is pyrimidinyl substituted with p occurrences of R 7 .
- a 1 is a 5-membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with p occurrences of R 7 .
- a 1 is a 5-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with p occurrences of R 7 .
- a 1 is imidazolyl or pyrazolyl; each of which is substituted with p occurrences of R 7 .
- a 1 is imidazolyl substituted with p occurrences of R 7 .
- a 1 is pyrazolyl substituted with p occurrences of R 7 .
- a 1 is selected from the groups depicted in the compounds in Table 3, below.
- X 2 is N, C(CN), or C(R 4 ). In certain embodiments, X 2 is N. In certain embodiments, X 2 is C(CN) or C(R 4 ). In certain embodiments, X 2 is C(CN) or C(H).
- X 2 is C(CN). In certain embodiments, X 2 is C(R 4 ). In certain embodiments, X 2 is C(H). In certain embodiments, X 2 is selected from the groups depicted in the compounds in Table 3, below.
- L is a covalent bond or a Ci-4 bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R 9 )-, or -C(O)-. In certain embodiments, L is a covalent bond.
- L is a Ci-4 bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R 9 )-, or -C(O)-.
- L is a Ci-4 bivalent saturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R 9 )-, or -C(O)-.
- L is a C 1-4 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R 9 )-, or - C(O)-. In certain embodiments, L is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -N(R 9 )-, or -C(O)-.
- L is a C1-4 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -N(R 9 )-, or -C(O)-.
- L is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -N(R 9 )-, or -C(O)-.
- L is a Ci-4 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -O- or -N(R 9 )-.
- L is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -O- or -N(R 9 )-.
- L is -0-CH 2 -, -0-(CH 2 ) 2 -, -N(R 9 )-CH 2 -, or -N(R 9 )-(CH 2 ) 2 -, wherein the terminal carbon atom of L is attached to R 2 .
- L is -0-CH 2 - or -0-(CH 2 ) 2 -, wherein the terminal carbon atom of L is attached to R 2 .
- L is -N(R 9 )-CH 2 - or -N(R 9 )-(CH 2 ) 2 -, wherein the terminal carbon atom of L is attached to R 2 .
- L is -0-CH 2 -, wherein the carbon atom of L is attached to R 2 .
- L is -0-(CH 2 ) 2 -, wherein the terminal carbon atom of L is attached to R 2 .
- L is -N(R 9 )-CH 2 -, wherein the carbon atom of L is attached to R 2 .
- L is -N(R 9 )-(CH 2 ) 2 -, wherein the terminal carbon atom of L is attached to R 2 .
- L is selected from the groups depicted in the compounds in Table 3, below.
- Y 1 is a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 1 is a C 2 -3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -. In certain embodiments, Y 1 is a C 2 -3 bivalent unsaturated, straight hydrocarbon chain.
- Y 1 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -. In certain embodiments, Y 1 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 1 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 1 is a C1-3 bivalent saturated, straight hydrocarbon chain.
- Y 1 is a C 2 -3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 1 is a C 2 -3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 1 is a C 2 -3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 1 is a C2-3 bivalent saturated, straight hydrocarbon chain.
- Y 1 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) 3 -, -CH 2 -0- CH 2 -, or -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 1 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, - (CH 2 )3-, or -CH 2 -CF 2 -CH 2 -.
- Y 1 is -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) 3 -, or -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 1 is -(CH 2 ) 2 - or -(CH 2 )3-. In certain embodiments, Y 1 is -CH 2 -C(H)(OH)- or -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 1 is -(CH 2 )-, -(CH 2 ) 2 -, or - (CH 2 ) 3 -.
- Y 1 is -(CH 2 ) 2 - or -CH 2 -C(H)(OH)-. In certain embodiments, Y 1 is -(CH 2 ) 3 -, -CH 2 -0-CH 2 -, or -CH 2 -CF 2 -CH 2 -.
- Y 1 is -(CH 2 )-. In certain embodiments, Y 1 is -(CH 2 ) 2 -. In certain embodiments, Y 1 is -CH 2 -C(H)(OH)-. In certain embodiments, Y 1 is -(CH 2 )3-. In certain embodiments, Y 1 is -CH 2 -0-CH 2 -. In certain embodiments, Y 1 is -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 1 is selected from the groups depicted in the compounds in Table 3, below.
- Y 2 is a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 2 is a C 2 -3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 2 is a C 2 -3 bivalent unsaturated, straight hydrocarbon chain.
- Y 2 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -. In certain embodiments, Y 2 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 2 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 2 is a C1-3 bivalent saturated, straight hydrocarbon chain.
- Y 2 is a C 2 -3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 2 is a C 2 -3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 2 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 2 is a C 2 -3 bivalent saturated, straight hydrocarbon chain.
- Y 2 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) 3 -, -CH 2 -0- CH 2 -, or -CH 2 -CF 2 -CH 2 -.
- Y 2 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, - (CH 2 ) 3 -, or -CH 2 -CF 2 -CH 2 -.
- Y 2 is -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) 3 -, or -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 2 is -(CH 2 ) 2 - or -(CH 2 )3-. In certain embodiments, Y 2 is -CH 2 -C(H)(OH)- or -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 2 is -(CH 2 )-, -(CH 2 ) 2 -, or - (CH 2 ) 3 -.
- Y 2 is -(CH 2 ) 2 - or -CH 2 -C(H)(OH)-. In certain embodiments, Y 2 is -(CH 2 ) 3 -, -CH 2 -0-CH 2 -, or -CH 2 -CF 2 -CH 2 -.
- Y 2 is -(CH 2 )-. In certain embodiments, Y 2 is -(CH 2 ) 2 -. In certain embodiments, Y 2 is -CH 2 -C(H)(OH)-. In certain embodiments, Y 2 is -(CH 2 )3-. In certain embodiments, Y 2 is -CH 2 -0-CH 2 -. In certain embodiments, Y 2 is -CH 2 -CF 2 -CH 2 -. In certain embodiments, Y 2 is selected from the groups depicted in the compounds in Table 3, below.
- Y 3 is a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 3 is a C 2 -3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 3 is a C 2 -3 bivalent unsaturated, straight hydrocarbon chain.
- Y 3 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -. In certain embodiments, Y 3 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 3 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 3 is a C1-3 bivalent saturated, straight hydrocarbon chain. [0393] In certain embodiments, Y 3 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R 7 )-, or -C(R 7 ) 2 -.
- Y 3 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R 7 )-, or -C(R 7 ) 2 -. In certain embodiments, Y 3 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R 7 )-, or - C(R 7 ) 2 -. In certain embodiments, Y 3 is a C2-3 bivalent saturated, straight hydrocarbon chain.
- Y 3 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) 3 -, -CH2-O- CH2-, or -CH2-CF2-CH2-.
- Y 3 is -(CH 2 )-, -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, - (CH 2 ) 3 -, or -CH2-CF2-CH2-.
- Y 3 is -(CH 2 ) 2 -, -CH 2 -C(H)(OH)-, -(CH 2 ) 3 -, or -CH2-CF2-CH2-. In certain embodiments, Y 3 is -(CH2)2- or -(CH2)3-. In certain embodiments, Y 3 is -CH 2 -C(H)(OH)- or -CH2-CF2-CH2-. In certain embodiments, Y 3 is -(CH 2 )-, -(CH 2 ) 2 -, or - (CH2)3-. In certain embodiments, Y 3 is -(CH2)2- or -CH2-C(H)(OH)-. In certain embodiments, Y 3 is -(CH 2 ) 3 -, -CH2-O-CH2-, or -CH2-CF2-CH2-.
- Y 3 is -(CH2)-. In certain embodiments, Y 3 is -(CH2)2-. In certain embodiments, Y 3 is -CH2-C(H)(OH)-. In certain embodiments, Y 3 is -(CH2)3-. In certain embodiments, Y 3 is -CH2-O-CH2-. In certain embodiments, Y 3 is -CH2-CF2-CH2-. In certain embodiments, Y 3 is selected from the groups depicted in the compounds in Table 3, below.
- m is 0, 1, 2, or 3. In certain embodiments, m is 0. In certain embodiments, m is 1. In certain embodiments, m is 2. In certain embodiments, m is 3. In certain embodiments, m is 0 or 1. In certain embodiments, m is 1 or 2. In certain embodiments, m is 2 or 3. In certain embodiments, m is 0, 1, or 2. In certain embodiments, m is 1, 2, or 3. In certain embodiments, m is selected from the values represented in the compounds in Table 3, below.
- n is 0, 1, 2, or 3. In certain embodiments, n is 0. In certain embodiments, n is 1. In certain embodiments, n is 2. In certain embodiments, n is 3. In certain embodiments, n is 0 or 1. In certain embodiments, n is 1 or 2. In certain embodiments, n is 2 or 3. In certain embodiments, n is 0, 1, or 2. In certain embodiments, n is 1, 2, or 3. In certain embodiments, n is selected from the values represented in the compounds in Table 3, below. [0398] As defined generally above, p is 0, 1, 2, or 3. In certain embodiments, p is 0. In certain embodiments, p is 1. In certain embodiments, p is 2. In certain embodiments, p is 3.
- p is 0 or 1. In certain embodiments, p is 1 or 2. In certain embodiments, p is 2 or 3. In certain embodiments, p is 0, 1, or 2. In certain embodiments, p is 1, 2, or 3. In certain embodiments, p is selected from the values represented in the compounds in Table 3, below.
- the compound is a compound in Table 1, 1-A, 2, or 3 below, or a pharmaceutically acceptable salt thereof. In certain embodiments, the compound is a compound in Table 1, 1-A, 2, or 3 below. In certain embodiments, the compound is a compound in Table 1, 2, or 3 below, or a pharmaceutically acceptable salt thereof. In certain embodiments, the compound is a compound in Table 1, 2, or 3 below. In certain embodiments, the compound is a compound in Table 1 or 2 below, or a pharmaceutically acceptable salt thereof. In certain embodiments, the compound is a compound in Table 1 or 2 below. In certain embodiments, the compound is a compound in Table 1 or 1-A below, or a pharmaceutically acceptable salt thereof. In certain embodiments, the compound is a compound in Table 1 or 1-A below.
- the compound is a compound in Table 1 below, or a pharmaceutically acceptable salt thereof. In certain embodiments, the compound is a compound in Table 1 below. In certain embodiments, the compound is a compound in Table 1-A below, or a pharmaceutically acceptable salt thereof. In certain embodiments, the compound is a compound in Table 1-A below. In certain embodiments, the compound is a compound in Table 2 below, or a pharmaceutically acceptable salt thereof. In certain embodiments, the compound is a compound in Table 2 below. In certain embodiments, the compound is a compound in Table 3 below, or a pharmaceutically acceptable salt thereof. In certain embodiments, the compound is a compound in Table 3 below. TABLE 1.
- Schemes 4 and 5 depict methods that may be used for preparing specific compounds described herein, based on methods described in Schemes 1-3 above, and in the Examples below.
- Scheme 4 depicts methods that may be used to prepare compounds 1-5 and 1-8.
- compound 1-26 may be prepared according to the methods depicted for compound 1-8, but using tert - butyl azetidin-3-ylcarbamate instead of tert -butyl 3,8-diazabicyclo[3.2.1]octane-8- carboxylate in the first step.
- compound 1-23 may be prepared according to the methods depicted for compound 1-5, but using 4-bromo-2-(methoxymethoxy)quinoline instead of the substituted naphthyl triflate in the Pd coupling step.
- Scheme 5 depicts methods that may be used to prepare compounds II-3 and II-4.
- compounds II-9 and II- 10 may be prepared according to the methods depicted for compounds II-3 and II-4, respectively, but using tert-butyl 1,4-diazepane-l-carboxylate or tert- butyl azetidin-3-ylcarbamate instead of tert-butyl 3,8-diazabicyclo[3.2.1]octane-8-carboxylate in the first step.
- SCHEME 5 tert-butyl 1,4-diazepane-l-carboxylate or tert- butyl azetidin-3-ylcarbamate instead of tert-butyl 3,8-diazabicyclo[3.2.1]octane-8-carboxylate in the first step.
- one aspect of the invention provides a method of treating a disorder mediated by K- Ras in a subject.
- the method comprises administering a therapeutically effective amount of a compound described herein, such as a compound of Formula I, II, or III, to a subject in need thereof to treat the disorder.
- Another aspect of the invention provides a method of treating a disorder mediated by K-Ras G12D in a subject.
- the method comprises administering a therapeutically effective amount of a compound described herein, such as a compound of Formula I, II, or III, to a subject in need thereof to treat the disorder.
- a compound described herein such as a compound of Formula I, II, or III
- the compound is a compound of Formula I, II, or III defined by one of the embodiments described above.
- K-Ras Kirsten Rat Sarcoma 2 Viral Oncogene Homolog
- GDP-bound inactive
- GTP-bound active
- cellular proliferation e.g ., see Alamgeer et al., (2013) Current Opin Pharmcol.13:394-401).
- K-Ras The role of activated K-Ras in malignancy was observed over thirty years ago (e.g., see Santos et al., (1984) Science 223:661-664). Aberrant expression of K-Ras has been reported to account for up to 20% of all cancers, and oncogenic K-Ras mutations that stabilize GTP binding and lead to constitutive activation of K-Ras and downstream signaling have been reported in 25 - 30% of lung adenocarcinomas (e.g., see Samatar and Poulikakos (2014) Nat Rev Drug Disc 13(12): 928- 942 doi: 10.1038/nrd428).
- K-Ras G12D mutation has been reported to be present in 25.0% of all pancreatic ductal adenocarcinoma patients, 13.3% of all colorectal carcinoma patients, 10.1% of all rectal carcinoma patients, 4.1% of all non- small cell lung carcinoma patients, and 1.7% of all small cell lung carcinoma patients (e.g., see The AACR Project GENIE Consortium, (2017) Cancer Discovery;7(8):818-831. Dataset Version 4).
- the provided compounds are inhibitors of K-Ras and are therefore useful for treating one or more disorders associated with activity of K-Ras.
- the present invention provides a method for treating a K-Ras -mediated disorder comprising the step of administering to a patient in need thereof a therapeutically effective compound of the present invention, or pharmaceutically acceptable composition thereof.
- K-Ras-mediated disorders, diseases, and/or conditions means any disease or other deleterious condition in which K-Ras, or a variant or mutant thereof, is known to play a role.
- another aspect or embodiment of the present invention relates to treating or lessening the severity of one or more diseases in which K-Ras is known to play a role.
- provided herein are methods of treating, reducing the severity of, delaying the onset of, or inhibiting the progress of a disease or disorder, or one or more symptoms thereof, of a disease or disorder characterized by or associated with increased K- Ras expression and/or increased K-Ras activity, comprising the step of administering to a patient in need thereof a therapeutically effective amount of a compound of the present invention, or pharmaceutically acceptable composition thereof.
- provided herein are methods of treating, reducing the severity of, delaying the onset of, or inhibiting the progress of a disease or disorder, or one or more symptoms thereof of a disease or disorder in which inhibition or antagonizing of K-Ras activity is beneficial, comprising the step of administering to a patient in need thereof a therapeutically effective amount of a compound of the present invention, or pharmaceutically acceptable composition thereof.
- the present invention provides a method for treating one or more disorders, diseases, and/or conditions wherein the disorder, disease, or condition includes, but is not limited to, a cellular proliferative disorder, comprising administering to a patient in need thereof, a K-Ras inhibitor compound as described herein, or a pharmaceutical salt or composition thereof.
- the cellular proliferative disorder is cancer.
- the cancer is characterized by increased K-Ras expression and/or increased K-Ras activity, i.e., “increased activated K-Ras.”
- the K-Ras is a mutant K-Ras. In some embodiments, the K- Ras is an activated mutant K-Ras. In some embodiments, the K-Ras is a G12 mutant K-Ras. In some embodiments, the K-Ras is K-Ras G12D, K-Ras G12V, or K-Ras G12C. In some embodiments, the K-Ras is K-Ras G12D. In some embodiments, the K-Ras is K-Ras G12V. In some embodiments, the K-Ras is K-Ras G12C.
- the provided compounds are inhibitors of K-Ras G12D and are therefore useful for treating one or more disorders associated with activity of K-Ras G12D.
- the present invention provides a method for treating a K-Ras G12D-mediated disorder comprising the step of administering to a patient in need thereof a therapeutically effective compound of the present invention, or pharmaceutically acceptable composition thereof.
- K-Ras G12D-mediated disorders, diseases, and/or conditions means any disease or other deleterious condition in which K-Ras G12D is known to play a role.
- another aspect or embodiment of the present invention relates to treating or lessening the severity of one or more diseases in which K-Ras G12D is known to play a role.
- kits for treating, reducing the severity of, delaying the onset of, or inhibiting the progress of a disease or disorder, or one or more symptoms thereof, of a disease or disorder characterized by or associated with increased K- Ras G12D expression and/or increased K-Ras G12D activity comprising the step of administering to a patient in need thereof a therapeutically effective amount of a compound of the present invention, or pharmaceutically acceptable composition thereof.
- provided herein are methods of treating, reducing the severity of, delaying the onset of, or inhibiting the progress of a disease or disorder, or one or more symptoms thereof of a disease or disorder in which inhibition or antagonizing of K-Ras G12D activity is beneficial, comprising the step of administering to a patient in need thereof a therapeutically effective amount of a compound of the present invention, or pharmaceutically acceptable composition thereof.
- the present invention provides a method for treating one or more disorders, diseases, and/or conditions wherein the disorder, disease, or condition includes, but is not limited to, a cellular proliferative disorder, comprising administering to a patient in need thereof, an K-Ras G12D inhibitor compound as described herein, or a pharmaceutical salt or composition thereof.
- the cellular proliferative disorder is cancer.
- the cancer is characterized by increased K-Ras G12D expression and/or increased K-Ras G12D activity, i.e., “increased activated K-Ras G12D.”
- an increase can be by at least about 10%, about 15%, about 20%, about 25%, about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 96%, about 97%, about 98%, about 99%, about 100%, about 2-fold, about 3- fold, about 4-fold, about 5-fold, about 6-fold, about 7-fold, about 8-fold, about 9-fold, about 10- fold, about 20-fold, about 25-fold, about 50-fold, about 100-fold, or higher, relative to a control or baseline amount of a function, or activity, or concentration.
- the terms "increased expression” and/or “increased activity" of a substance, such as K-Ras G12D, in a sample or cancer or patient refers to an increase in the amount of the substance, such as K-Ras G12D, of about 5%, about 10%, about 15%, about 20%, about 25%, about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 96%, about 97%, about 98%, about 99%, about 100%, about 2-fold, about 3-fold, about 4-fold, about 5-fold, about 6-fold, about 7-fold, about 8-fold, about 9-fold, about 10-fold, about 20-fold, about 25-fold, about 50-fold, about 100-fold, or higher, relative to the amount of the substance, such as K-Ras G12D, in a control sample or control samples, such as an individual or
- a subject can also be determined to have an "increased expression” or “increased activity” of a substance, such as K-Ras G12D, if the expression and/or activity of the substance, such as K-Ras G12D, is increased by one standard deviation, two standard deviations, three standard deviations, four standard deviations, five standard deviations, or more, relative to the mean (average) or median amount of the substance, such as K-Ras G12D, in a control group of samples or a baseline group of samples or a retrospective analysis of patient samples.
- control or baseline expression levels can be previously determined, or measured prior to the measurement in the sample or cancer or subject, or can be obtained from a database of such control samples.
- a proliferative disease refers to a disease that occurs due to abnormal growth or extension by the multiplication of cells (Walker, Cambridge Dictionary of Biology, Cambridge University Press: Cambridge, UK, 1990).
- a proliferative disease can be associated with: 1) the pathological proliferation of normally quiescent cells; 2) the pathological migration of cells from their normal location ( e.g ., metastasis of neoplastic cells); 3) the pathological expression of proteolytic enzymes, such as the matrix metalloproteinases (e.g., collagenases, gelatinases, and elastases); or 4) the pathological angiogenesis, as in proliferative retinopathy and tumor metastasis.
- proteolytic enzymes such as the matrix metalloproteinases (e.g., collagenases, gelatinases, and elastases)
- the pathological angiogenesis as in proliferative retinopathy and tumor metastasis.
- Exemplary proliferative diseases include cancers (i.e., "malignant neoplasms"), benign neoplasms, angiogenesis, inflammatory diseases, and autoimmune diseases.
- the disorder is cancer.
- the cancer or proliferative disorder or tumor to be treated using the compounds and methods and uses described herein include, but are not limited to, a hematological cancer, a lymphoma, a myeloma, a leukemia, a neurological cancer, skin cancer, breast cancer, a prostate cancer, a colorectal cancer, lung cancer, head and neck cancer, a gastrointestinal cancer, a liver cancer, a pancreatic cancer, a genitourinary cancer, a bone cancer, renal cancer, and a vascular cancer.
- a cancer is treated by inhibiting or reducing or decreasing or arresting further growth or spread of the cancer or tumor.
- a cancer is treated by inhibiting or reducing the size (e.g., volume or mass) of the cancer or tumor by at least 5%, at least 10%, at least 25%, at least 50%, at least 75%, at least 90% or at least 99% relative to the size of the cancer or tumor prior to treatment.
- a cancer is treated by reducing the quantity of the cancers or tumors in the patient by at least 5%, at least 10%, at least 25%, at least 50%, at least 75%, at least 90% or at least 99% relative to the quantity of the cancers or tumors prior to treatment.
- the cancer is selected from is non-small cell lung cancer (NSCLC), small cell lung cancer, colorectal cancer, rectal cancer, and pancreatic cancer. In some embodiments, the cancer is selected from non-small cell lung cancer (NSCLC), pancreatic cancer, and colorectal cancer. In some embodiments, the cancer is selected from non-small cell lung cancer (NSCLC) and pancreatic cancer.
- the cancer is a solid tumor. In certain embodiments, the cancer is a melanoma, carcinoma, or blastoma. In certain embodiments, the cancer is a melanoma. In certain embodiments, the cancer is a carcinoma. In certain embodiments, the cancer is an adenocarcinoma. In certain embodiments, the cancer is a blastoma.
- the cancer is lung cancer, pancreatic cancer, colorectal cancer, breast cancer, cervical cancer, prostate cancer, gastric cancer, skin cancer, liver cancer, bile duct cancer, or nervous system cancer.
- the cancer is lung cancer.
- the cancer is pancreatic cancer.
- the cancer is colorectal cancer.
- the cancer is breast cancer.
- the cancer is cervical cancer.
- the cancer is prostate cancer.
- the cancer is gastric cancer.
- the cancer is skin cancer.
- the cancer is liver cancer.
- the cancer is bile duct cancer.
- the cancer is nervous system cancer.
- the cancer is breast adenocarcinoma, lung adenocarcinoma, pancreatic adenocarcinoma, cervical adenocarcinoma, colorectal adenocarcinoma, prostate adenocarcinoma, gastric adenocarcinoma, melanoma, lung squamous cell carcinoma, hepatocellular carcinoma, cholangiocarcinoma, glioblastoma, or neuroblastoma.
- the cancer is breast adenocarcinoma.
- the cancer is lung adenocarcinoma.
- the cancer is pancreatic adenocarcinoma.
- the cancer is cervical adenocarcinoma.
- the cancer is prostate adenocarcinoma.
- the cancer is gastric adenocarcinoma.
- the cancer is melanoma.
- the cancer is lung squamous cell carcinoma, hepatocellular carcinoma, or cholangiocarcinoma. In certain embodiments, the cancer is lung squamous cell carcinoma. In certain embodiments, the cancer is hepatocellular carcinoma. In certain embodiments, the cancer is cholangiocarcinoma.
- the cancer is glioblastoma or neuroblastoma. In certain embodiments, the cancer is glioblastoma. In certain embodiments, the cancer is neuroblastoma. [0429] In certain embodiments, the cancer is lung cancer, pancreatic cancer, or colorectal cancer. In certain embodiments, the cancer is non-small cell lung cancer, pancreatic cancer, or colorectal cancer. In certain embodiments, the cancer is lung cancer. In certain embodiments, the cancer is non-small cell lung cancer.
- the cancer has elevated K-Ras activity. In certain embodiments, the cancer overexpresses K-Ras. In some embodiments, a subject having a disorder associated with or mediated by K-Ras has been identified or diagnosed as having a cancer having a K-Ras mutation (e.g., as determined using a regulatory agency-approved, e.g., FDA-approved, assay or kit). In some embodiments, the subject has a tumor that is positive for a K-Ras mutation (e.g., as determined using a regulatory agency-approved assay or kit).
- the subject can be a subject who has one or more tumor that is positive for a K-Ras mutation (e.g., identified as positive using a regulatory agency-approved, e.g., FDA-approved, assay or kit).
- the subject can be a subject whose tumor(s) have a K-Ras mutation (e.g., where the tumor is identified as such using a regulatory agency-approved, e.g., FDA-approved, kit or assay).
- the subject is suspected of having a K-Ras gene-associated cancer.
- the subject has a clinical record indicating that the subject has a tumor that has a K-Ras mutation (and, for example, the clinical record indicates that the subject should be treated with any of the compounds or compositions provided herein).
- an assay is used to determine whether the patient has a K-Ras mutation using a sample (e.g., a biological sample or a biopsy sample (e.g., a paraffin-embedded biopsy sample) from a patient (e.g., a patient suspected of having a K-Ras-associated cancer, a patient having one or more symptoms of a K- Ras-associated cancer, and/or a patient that has an increased risk of developing a K-Ras- associated cancer).
- a sample e.g., a biological sample or a biopsy sample (e.g., a paraffin-embedded biopsy sample) from a patient (e.g., a patient suspected of having a K-Ras-associated cancer, a patient having one or more symptoms of a K- Ras-associated cancer, and/or a patient that has an increased risk of developing a K-Ras- associated cancer).
- Non-limiting examples of such assays that can be used to determine whether a patient or subject has a K-Ras mutation include next generation sequencing (NGS), immunohistochemistry, fluorescence microscopy, break apart FISH analysis, Southern blotting, Western blotting, FACS analysis, Northern blotting, and PCR-based amplification (e.g., RT-PCR and quantitative real-time RT-PCR).
- NGS next generation sequencing
- the assays are typically performed, e.g., with at least one labelled nucleic acid probe or at least one labelled antibody or antigen-binding fragment thereof.
- regulatory agency is a country’s agency for the approval of the medical use of pharmaceutical agents with the country.
- a non limiting example of a regulatory agency is the U.S. Food and Drug Administration (FDA).
- the K-Ras mutation is a G12 mutation. In some embodiments, the K-Ras mutation is G12D, G12V, or G12C. In some embodiments, the K-Ras mutation is G12D. In some embodiments, the K-Ras mutation is G12V. In some embodiments, the K-Ras mutation is G12C.
- the cancer has elevated K-Ras G12D activity. In certain embodiments, the cancer overexpresses K-Ras G12D.
- a subject having a disorder associated with or mediated by K-Ras G12D has been identified or diagnosed as having a cancer having a K-Ras G12D mutation (e.g ., as determined using a regulatory agency- approved, e.g., FDA-approved, assay or kit).
- the subject has a tumor that is positive for a K-Ras G12D mutation (e.g., as determined using a regulatory agency-approved assay or kit).
- the subject can be a subject who has one or more tumor that is positive for a K-Ras G12D mutation (e.g., identified as positive using a regulatory agency-approved, e.g., FDA- approved, assay or kit).
- the subject can be a subject whose tumor(s) have a K-Ras G12D mutation (e.g., where the tumor is identified as such using a regulatory agency-approved, e.g., FDA-approved, kit or assay).
- the subject is suspected of having a K-Ras G12D gene-associated cancer.
- the subject has a clinical record indicating that the subject has a tumor that has a K-Ras G12D mutation (and, for example, the clinical record indicates that the subject should be treated with any of the compounds or compositions provided herein).
- an assay is used to determine whether the patient has a K-Ras G12D mutation using a sample (e.g., a biological sample or a biopsy sample (e.g., a paraffin-embedded biopsy sample) from a patient (e.g., a patient suspected of having a K-Ras G12D-associated cancer, a patient having one or more symptoms of a K-Ras G12D-associated cancer, and/or a patient that has an increased risk of developing a K-Ras G12D-associated cancer).
- a sample e.g., a biological sample or a biopsy sample (e.g., a paraffin-embedded biopsy sample) from a patient (e.g., a patient suspected of having a K-Ras G12D-associated cancer, a patient having one or more symptoms of a K-Ras G12D-associated cancer, and/or a patient that has an increased risk of developing a K-Ras G12D-
- Non-limiting examples of such assays that can be used to determine whether a patient or subject has a K-Ras G12D mutation include next generation sequencing (NGS), immunohistochemistry, fluorescence microscopy, break apart FISH analysis, Southern blotting, Western blotting, FACS analysis, Northern blotting, and PCR- based amplification (e.g ., RT-PCR and quantitative real-time RT-PCR).
- NGS next generation sequencing
- the assays are typically performed, e.g., with at least one labelled nucleic acid probe or at least one labelled antibody or antigen-binding fragment thereof.
- regulatory agency is a country’s agency for the approval of the medical use of pharmaceutical agents with the country.
- a non-limiting example of a regulatory agency is the U.S. Food and Drug Administration (FDA).
- the cancer is a leukemia (e.g., acute leukemia, acute lymphocytic leukemia, acute myelocytic leukemia, acute myeloblastic leukemia, acute promyelocytic leukemia, acute myelomonocytic leukemia, acute monocytic leukemia, acute erythroleukemia, chronic leukemia, chronic myelocytic leukemia, chronic lymphocytic leukemia), polycythemia vera, lymphoma (e.g., Hodgkin’s disease or non-Hodgkin’s disease), Waldenstrom's macroglobulinemia, multiple myeloma, heavy chain disease, or a solid tumor such as a sarcoma or carcinoma (e.g., fibrosarcoma, myxosarcoma, liposarcoma, chondrosarcoma, osteogenic sarcoma, chordoma, angiosarcoma
- a leukemia
- the cancer is glioma, astrocytoma, glioblastoma multiforme (GBM, also known as glioblastoma), medulloblastoma, craniopharyngioma, ependymoma, pinealoma, hemangioblastoma, acoustic neuroma, oligodendroglioma, schwannoma, neurofibrosarcoma, meningioma, melanoma, neuroblastoma, or retinoblastoma.
- the cancer is acoustic neuroma, astrocytoma ( e.g .
- Grade I Pilocytic Astrocytoma, Grade II - Low-grade Astrocytoma, Grade III - Anaplastic Astrocytoma, or Grade IV - Glioblastoma (GBM)), chordoma, CNS lymphoma, craniopharyngioma, brain stem glioma, ependymoma, mixed glioma, optic nerve glioma, subependymoma, medulloblastoma, meningioma, metastatic brain tumor, oligodendroglioma, pituitary tumors, primitive neuroectodermal (PNET) tumor, or schwannoma.
- GBM Glioblastoma
- the cancer is a type found more commonly in children than adults, such as brain stem glioma, craniopharyngioma, ependymoma, juvenile pilocytic astrocytoma (JPA), medulloblastoma, optic nerve glioma, pineal tumor, primitive neuroectodermal tumors (PNET), or rhabdoid tumor.
- the cancer is mesothelioma, hepatobilliary (hepatic and billiary duct), bone cancer, pancreatic cancer, skin cancer, cancer of the head or neck, cutaneous or intraocular melanoma, ovarian cancer, colon cancer, rectal cancer, cancer of the anal region, stomach cancer, gastrointestinal (gastric, colorectal, and duodenal), uterine cancer, carcinoma of the fallopian tubes, carcinoma of the endometrium, carcinoma of the cervix, carcinoma of the vagina, carcinoma of the vulva, Hodgkin’s Disease, cancer of the esophagus, cancer of the small intestine, cancer of the endocrine system, cancer of the thyroid gland, cancer of the parathyroid gland, cancer of the adrenal gland, sarcoma of soft tissue, cancer of the urethra, cancer of the penis, prostate cancer, testicular cancer, chronic or acute leukemia, chronic myeloid leukemia, lymphoc
- the cancer is hepatocellular carcinoma, ovarian cancer, ovarian epithelial cancer, fallopian tube cancer, papillary serous cystadenocarcinoma, uterine papillary serous carcinoma (UPSC), prostate cancer, testicular cancer, gallbladder cancer, hepatocholangiocarcinoma, soft tissue and bone synovial sarcoma, rhabdomyosarcoma, osteosarcoma, chondrosarcoma, Ewing sarcoma, anaplastic thyroid cancer, adrenocortical adenoma, pancreatic cancer, pancreatic ductal carcinoma, pancreatic adenocarcinoma, gastrointestinal/stomach (GIST) cancer, lymphoma, squamous cell carcinoma of the head and neck (SCCHN), salivary gland cancer, glioma, or brain cancer, neurofibromatosis- 1 associated malignant peripheral nerve sheath tumors (MPNST),
- MPNST neurofibromat
- the cancer is hepatocellular carcinoma (HCC), hepatoblastoma, colon cancer, rectal cancer, ovarian cancer, ovarian epithelial cancer, fallopian tube cancer, papillary serous cystadenocarcinoma, uterine papillary serous carcinoma (UPSC), hepatocholangiocarcinoma, soft tissue and bone synovial sarcoma, rhabdomyosarcoma, osteosarcoma, anaplastic thyroid cancer, adrenocortical adenoma, pancreatic cancer, pancreatic ductal carcinoma, pancreatic adenocarcinoma, glioma, neurofibromatosis- 1 associated malignant peripheral nerve sheath tumors (MPNST), Waldenstrom’s macroglobulinemia, or medulloblastoma.
- HCC hepatocellular carcinoma
- hepatoblastoma colon cancer
- rectal cancer ovarian cancer
- ovarian epithelial cancer
- the cancer is selected from renal cell carcinoma, or kidney cancer; hepatocellular carcinoma (HCC) or hepatoblastoma, or liver cancer; melanoma; breast cancer; colorectal carcinoma, or colorectal cancer; colon cancer; rectal cancer; anal cancer; lung cancer, such as non-small cell lung cancer (NSCLC) or small cell lung cancer (SCLC); ovarian cancer, ovarian epithelial cancer, ovarian carcinoma, or fallopian tube cancer; papillary serous cystadenocarcinoma or uterine papillary serous carcinoma (UPSC); prostate cancer; testicular cancer; gallbladder cancer; hepatocholangiocarcinoma; soft tissue and bone synovial sarcoma; rhabdomyosarcoma; osteosarcoma; chondrosarcoma; Ewing sarcoma; anaplastic thyroid cancer; adrenocortical carcinoma; pancreatic cancer; pancreatic duct
- the cancer is renal cell carcinoma, hepatocellular carcinoma (HCC), hepatoblastoma, colorectal carcinoma, colorectal cancer, colon cancer, rectal cancer, anal cancer, ovarian cancer, ovarian epithelial cancer, ovarian carcinoma, fallopian tube cancer, papillary serous cystadenocarcinoma, uterine papillary serous carcinoma (UPSC), hepatocholangiocarcinoma, soft tissue and bone synovial sarcoma, rhabdomyosarcoma, osteosarcoma, chondrosarcoma, anaplastic thyroid cancer, adrenocortical carcinoma, pancreatic cancer, pancreatic ductal carcinoma, pancreatic adenocarcinoma, glioma, brain cancer, neurofibromatosis- 1 associated malignant peripheral nerve sheath tumors (MPNST), Waldenstrom’s macroglobulinemia, or medulloblastoma.
- HCC hepatocellular
- the cancer is hepatocellular carcinoma (HCC), hepatoblastoma, colon cancer, rectal cancer, ovarian cancer, ovarian epithelial cancer, ovarian carcinoma, fallopian tube cancer, papillary serous cystadenocarcinoma, uterine papillary serous carcinoma (UPSC), hepatocholangiocarcinoma, soft tissue and bone synovial sarcoma, rhabdomyosarcoma, osteosarcoma, anaplastic thyroid cancer, adrenocortical carcinoma, pancreatic cancer, pancreatic ductal carcinoma, pancreatic adenocarcinoma, glioma, neurofibromatosis- 1 associated malignant peripheral nerve sheath tumors (MPNST), Waldenstrom’s macroglobulinemia, or medulloblastoma.
- HCC hepatocellular carcinoma
- hepatoblastoma colon cancer
- rectal cancer ovarian cancer
- ovarian cancer ovarian
- the cancer is hepatocellular carcinoma (HCC). In some embodiments, the cancer is hepatoblastoma. In some embodiments, the cancer is colon cancer.
- the cancer is rectal cancer. In some embodiments, the cancer is ovarian cancer, or ovarian carcinoma. In some embodiments, the cancer is ovarian epithelial cancer. In some embodiments, the cancer is fallopian tube cancer. In some embodiments, the cancer is papillary serous cystadenocarcinoma. In some embodiments, the cancer is uterine papillary serous carcinoma (UPSC). In some embodiments, the cancer is hepatocholangiocarcinoma. In some embodiments, the cancer is soft tissue and bone synovial sarcoma. In some embodiments, the cancer is rhabdomyosarcoma. In some embodiments, the cancer is osteosarcoma.
- UPSC papillary serous carcinoma
- the cancer is anaplastic thyroid cancer. In some embodiments, the cancer is adrenocortical carcinoma. In some embodiments, the cancer is pancreatic cancer, or pancreatic ductal carcinoma. In some embodiments, the cancer is pancreatic adenocarcinoma. In some embodiments, the cancer is glioma. In some embodiments, the cancer is malignant peripheral nerve sheath tumors (MPNST). In some embodiments, the cancer is neurofibromatosis- 1 associated MPNST. In some embodiments, the cancer is Waldenstrom’s macroglobulinemia. In some embodiments, the cancer is medulloblastoma.
- MPNST peripheral nerve sheath tumors
- the cancer is neurofibromatosis- 1 associated MPNST.
- the cancer is Waldenstrom’s macroglobulinemia. In some embodiments, the cancer is medulloblastoma.
- Another aspect of the invention provides for the use of a compound described herein (such as a compound of Formula I, II, or III, or other compounds in Section I) for treating a medical disorder, such as a medical disorder described herein (for example, cancer).
- a compound described herein such as a compound of Formula I, II, or III, or other compounds in Section I
- a medical disorder such as a medical disorder described herein (for example, cancer).
- the compounds described herein can inhibit K-Ras activity.
- another aspect of the invention provides a method of inhibiting K-Ras activity. The method comprises contacting K-Ras with an effective amount of a naphthyl-substituted quinolin-4( 1H)- one or related compound described herein, such as a compound of Formula I, II, or III, or other compounds in Section I, to inhibit K-Ras activity.
- the compound is a compound of Formula I, II, or III defined by one of the embodiments described above.
- the K-Ras is a mutant K-Ras. In some embodiments, the K-Ras is an activated mutant K-Ras. In some embodiments, the K-Ras is a G 12 mutant K-Ras. In some embodiments, the K-Ras is K-Ras G12D, K-Ras G12V, or K-Ras G12C. In some embodiments, the K-Ras is K-Ras G12D. In some embodiments, the K-Ras is K-Ras G12V. In some embodiments, the K-Ras is K-Ras G12C.
- the compounds described herein can inhibit K-Ras G12D activity.
- another aspect of the invention provides a method of inhibiting K-Ras G12D activity.
- the method comprises contacting K-Ras G12D with an effective amount of a naphthyl- substituted quinolin-4( 1H)-onc or related compound described herein, such as a compound of Formula I, II, or III, or other compounds in Section I, to inhibit K-Ras G12D activity.
- the compound is a compound of Formula I, II, or III defined by one of the embodiments described above.
- Compounds may be tested for ability to bind to and/or inhibit K-Ras activity according to any of various assays known in the art, including, for example, surface plasmon resonance binding assays, TR-FRET displacement binding assays, TR-FRET assays that detect Ras-Raf interaction, and cell-based phosphorylation inhibition assays.
- Compounds may be tested for ability to bind to and/or inhibit K-Ras G12D activity according to any of various assays known in the art, including, for example, surface plasmon resonance binding assays, TR-FRET displacement binding assays, TR-FRET assays that detect Ras-Raf interaction, and cell-based phosphorylation inhibition assays.
- Naphthyl-substituted quinolin-4( l//)-onc or related compounds described herein e.g., a compound of Formula I, II, or III, or other compounds in Section I
- additional therapeutic agents to treat medical disorders, such as a cancer.
- the present invention provides a method of treating a disclosed disease or condition comprising administering to a patient in need thereof an effective amount of a compound disclosed herein or a pharmaceutically acceptable salt thereof and co-administering simultaneously or sequentially an effective amount of one or more additional therapeutic agents, such as those described herein.
- the method includes co-administering one additional therapeutic agent.
- the method includes co-administering two additional therapeutic agents.
- the combination of the disclosed compound and the additional therapeutic agent or agents acts synergistically.
- One or more other therapeutic agents may be administered separately from a compound or composition of the invention, as part of a multiple dosage regimen.
- one or more other therapeutic agents may be part of a single dosage form, mixed together with a compound of this invention in a single composition.
- one or more other therapeutic agent and a compound or composition of the invention may be administered simultaneously, sequentially or within a period of time from one another, for example within 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 18, 20, 21, 22, 23, or 24 hours from one another.
- one or more other therapeutic agent and a compound or composition of the invention are administerd as a multiple dosage regimen more than 24 hours aparts.
- the term “combination,” “combined,” and related terms refers to the simultaneous or sequential administration of therapeutic agents in accordance with this invention.
- a compound of the present invention can be administered with one or more other therapeutic agent(s) simultaneously or sequentially in separate unit dosage forms or together in a single unit dosage form.
- the present invention provides a single unit dosage form comprising a compound of the current invention, one or more other therapeutic agent(s), and a pharmaceutically acceptable carrier, adjuvant, or vehicle.
- compositions which comprise an additional therapeutic agent, such as a second anti-cancer agent, as described above varies depending upon the host treated and the particular mode of administration.
- a composition of the invention should be formulated so that a dosage of between 0.01 - 100 mg/kg body weight/day of a compound of the invention can be administered.
- compositions which comprise one or more other therapeutic agent(s) can act synergistically. Therefore, the amount of the one or more other therapeutic agent(s) in such compositions may be less than that required in a monotherapy utilizing only that therapeutic agent. In such compositions a dosage of between 0.01 - 1,000 g/kg body weight/day of the one or more other therapeutic agent(s) can be administered.
- the amount of one or more other therapeutic agent(s) present in the compositions of this invention is preferably no more than the amount that would normally be administered in a composition comprising that therapeutic agent as the only active agent.
- the amount of one or more other therapeutic agent(s) in the presently disclosed compositions ranges from about 50% to 100% of the amount normally present in a composition comprising that agent as the only therapeutically active agent.
- one or more other therapeutic agent(s) is administered at a dosage of about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, or about 95% of the amount normally administered for that agent.
- the phrase "normally administered” means the amount an FDA- approved therapeutic agent is approved for dosing per the FDA label insert.
- another aspect of the invention provides a method of treating cancer in a patient.
- the method comprises administering to a subject in need thereof (i) a therapeutically effective amount of a naphthyl-substituted quinolin-4( l /7)-onc or related compound described herein and (ii) a second anti-cancer agent, in order to treat the cancer.
- the second anti-cancer agent is an EGFR inhibitor, a FAK inhibitor, a RAF/ERK/MAPK inhibitor, a mTOR Inhibitor, a MEK Inhibitor, or a Phosphoinositide 3-Kinase Inhibitor.
- the second anti-cancer agent is an ALK Inhibitor, an ATR Inhibitor, an A2A Antagonist, a Base Excision Repair Inhibitor, a Bcr-Abl Tyrosine Kinase Inhibitor, a Bruton's Tyrosine Kinase Inhibitor, a CDC7 Inhibitor, a CHK1 Inhibitor, a Cyclin-Dependent Kinase Inhibitor, a DNA-PK Inhibitor, an Inhibitor of both DNA-PK and mTOR, a DNMT1 Inhibitor, a DNMT1 Inhibitor plus 2-chloro-deoxy adenosine, an HD AC Inhibitor, a Hedgehog Signaling Pathway Inhibitor, an IDO Inhibitor, a JAK Inhibitor, a mTOR Inhibitor, a MEK Inhibitor, a MELK Inhibitor, a
- the second anti-cancer agent is an ALK Inhibitor. In certain embodiments, the second anti-cancer agent is an ALK Inhibitor comprisng ceritinib or crizotinib. In certain embodiments, the second anti-cancer agent is an ATR Inhibitor. In certain embodiments, the second anti-cancer agent is an ATR Inhibitor comprising AZD6738 or VX- 970. In certain embodiments, the second anti-cancer agent is an A2A Antagonist. In certain embodiments, the second anti-cancer agent is a Base Excision Repair Inhibitor comprising methoxyamine.
- the second anti-cancer agent is a Base Excision Repair Inhibitor, such as methoxyamine.
- the second anti-cancer agent is a Bcr- Abl Tyrosine Kinase Inhibitor.
- the second anti-cancer agent is a Bcr- Abl Tyrosine Kinase Inhibitor comprising dasatinib or nilotinib.
- the second anti-cancer agent is a Bruton's Tyrosine Kinase Inhibitor.
- the second anti-cancer agent is a Bruton's Tyrosine Kinase Inhibitor comprising ibrutinib.
- the second anti-cancer agent is a CDC7 Inhibitor.
- the second anti-cancer agent is a CDC7 Inhibitor comprising RXDX-103 or AS- 141.
- the second anti-cancer agent is a CHK1 Inhibitor. In certain embodiments, the second anti-cancer agent is a CHK1 Inhibitor comprising MK-8776, ARRY- 575, or SAR-020106. In certain embodiments, the second anti-cancer agent is a Cyclin- Dependent Kinase Inhibitor. In certain embodiments, the second anti-cancer agent is a Cyclin- Dependent Kinase Inhibitor comprising palbociclib. In certain embodiments, the second anti cancer agent is a DNA-PK Inhibitor. In certain embodiments, the second anti-cancer agent is a DNA-PK Inhibitor comprising MSC2490484A. In certain embodiments, the second anti-cancer agent is Inhibitor of both DNA-PK and mTOR. In certain embodiments, the second anti-cancer agent comprises CC-115.
- the second anti-cancer agent is a DNMT1 Inhibitor.
- the second anti-cancer agent is a DNMT1 Inhibitor comprising decitabine, RX- 3117, guadecitabine, NUC-8000, or azacytidine.
- the second anti-cancer agent comprises a DNMT1 Inhibitor and 2-chloro-deoxyadenosine.
- the second anti-cancer agent comprises ASTX-727.
- the second anti-cancer agent is a HD AC Inhibitor.
- the second anti-cancer agent is a HDAC Inhibitor comprising OBP-801, CHR- 3996, etinostate, resminostate, pracinostat, CG-200745, panobinostat, romidepsin, mocetinostat, belinostat, AR-42, ricolinostat, KA-3000, or ACY-241.
- the second anti-cancer agent is a Hedgehog Signaling Pathway Inhibitor. In certain embodiments, the second anti-cancer agent is a Hedgehog Signaling Pathway Inhibitor comprising sonidegib or vismodegib. In certain embodiments, the second anti-cancer agent is an IDO Inhibitor. In certain embodiments, the second anti-cancer agent is an IDO Inhibitor comprising INCB024360. In certain embodiments, the second anti-cancer agent is a JAK Inhibitor. In certain embodiments, the second anti-cancer agent is a JAK Inhibitor comprising ruxolitinib or tofacitinib.
- the second anti-cancer agent is a mTOR Inhibitor. In certain embodiments, the second anti-cancer agent is a mTOR Inhibitor comprising everolimus or temsirolimus. In certain embodiments, the second anti-cancer agent is a MEK Inhibitor. In certain embodiments, the second anti-cancer agent is a MEK Inhibitor comprising cobimetinib or trametinib. In certain embodiments, the second anti-cancer agent is a MELK Inhibitor. In certain embodiments, the second anti-cancer agent is a MELK Inhibitor comprising ARN-7016, APTO-500, or OTS-167.
- the second anti cancer agent is a MTH1 Inhibitor. In certain embodiments, the second anti-cancer agent is a MTH1 Inhibitor comprising (S)-crizotinib, TH287, or TH588.
- the second anti-cancer agent is a PARP Inhibitor.
- the second anti-cancer agent is a PARP Inhibitor comprising MP-124, olaparib, BGB-290, talazoparib, veliparib, niraparib, E7449, rucaparb, or ABT-767.
- the second anti-cancer agent is a Phosphoinositide 3-Kinase Inhibitor.
- the second anti-cancer agent is a Phosphoinositide 3-Kinase Inhibitor comprising idelalisib.
- the second anti-cancer agent is an inhibitor of both PARP1 and DHODH (i.e., an agent that inhibits both poly ADP ribose polymerase 1 and dihydroorotate dehydrogenase).
- the second anti-cancer agent is a Proteasome Inhibitor. In certain embodiments, the second anti-cancer agent is a Proteasome Inhibitor comprising bortezomib or carfilzomib. In certain embodiments, the second anti-cancer agent is a Topoisomerase-II Inhibitor. In certain embodiments, the second anti-cancer agent is a Topoisomerase-II Inhibitor comprising vosaroxin.
- the second anti-cancer agent is a Tyrosine Kinase Inhibitor. In certain embodiments, the second anti-cancer agent is a Tyrosine Kinase Inhibitor comprising bosutinib, cabozantinib, imatinib or ponatinib. In certain embodiments, the second anti-cancer agent is a VEGFR Inhibitor. In certain embodiments, the second anti-cancer agent is a VEGFR Inhibitor comprising regorafenib. In certain embodiments, the second anti-cancer agent is a WEE1 Inhibitor. In certain embodiments, the second anti-cancer agent is a WEE1 Inhibitor comprising AZD1775.
- the second anti-cancer agent is a compound targeting, decreasing or inhibiting the activity of members of the protein kinase C (PKC) and Raf family of serine/threonine kinases, members of the MEK, SRC, JAK/pan-JAK, FAK, PDK1, PKB/Akt, Ras/MAPK, PI3K, SYK, TYK2, BTK and TEC family, and/or members of the cyclin-dependent kinase family (CDK) including staurosporine derivatives, such as midostaurin; examples of further compounds include UCN-01, safingol, BAY 43-9006, Bryostatin 1, Perifosine; llmofosine; RO 318220 and RO 320432; GO 6976; Isis 3521; LY333531/LY379196; isochinoline compounds; FTIs; PD184352 or QAN697 (a P13K inhibitor) or
- the second anti-cancer agent is an agonist of 0X40, CD137, CD40, GITR, CD27, HVEM, TNFRSF25, or ICOS.
- the second anti cancer agent is an agonist of 0X40, CD137, CD40, or GITR.
- the second anti-cancer agent is an agonist of CD27, HVEM, TNFRSF25, or ICOS.
- the second anti-cancer agent is a therapeutic antibody.
- the therapeutic antibody targets one of the following: CD20, CD30, CD33, CD52, EpCAM, CEA, gpA33, a mucin, TAG-72, CAIX, PSMA, a folate-binding protein, a ganglioside, Le, VEGF, VEGFR, VEGFR2, integrin aVp3, integrin a5b1, EGFR, ERBB2, ERBB3, MET, IGF1R, EPHA3, TRAILR1, TRAILR2, RANKL, FAP, tenascin, CD19, KIR, NKG2A, CD47, CEACAM1, c-MET, VISTA, CD73, CD38, BAFF, interleukin- 1 beta, B4GALNT1, interleukin- 6, and interleukin-6 receptor.
- the second anti-cancer agent is a therapeutic antibody selected from the group consisting of rituximab, ibritumomab tiuxetan, tositumomab, obinutuzumab, ofatumumab, brentuximab vedotin, gemtuzumab ozogamicin, alemtuzumab, IGN101, adecatumumab, labetuzumab, huA33, pemtumomab, oregovomab, minetumomab, cG250, J591, Movl8, farletuzumab, 3F8, chl4.18, KW-2871, hu3S193, lgN311, bevacizumab, IM-2C6, pazopanib, sorafenib, axitinib, CDP791, lenvatinib, ramuci
- the second anti-cancer agent is a cytokine.
- the cytokine is IL-12, IL-15, GM-CSF, or G-CSF.
- the second anti-cancer agent is sipuleucel-T, aldesleukin (a human recombinant interleukin-2 product having the chemical name des-alanyl-1, serine- 125 human interleukin-2), dabrafenib (a kinase inhibitor having the chemical name N- ⁇ 3-[5-(2- aminopyri midi n-4-yl)-2-ter t -butyl- 1 ,3-thiazol-4-yl] -2- fluorophenyl ⁇ -2,6- difluorobenzenesulfonamide), vemurafenib (a kinase inhibitor having the chemical name propane- 1- sulfonic acid ⁇ 3-[5-(4-ch)
- the second anti-cancer agent is a placental growth factor, an antibody-drug conjugate, an oncolytic vims, or an anti-cancer vaccine.
- the second anti-cancer agent is a placental growth factor.
- the second anti-cancer agent is a placental growth factor comprising ziv-aflibercept.
- the second anti-cancer agent is an antibody-drug conjugate.
- the second anti-cancer agent is an antibody-drug conjugate selected from the group consisting of brentoxumab vedotin and trastuzumab emtransine.
- the second anti-cancer agent is an oncolytic virus. In certain embodiments, the second anti-cancer agent is the oncolytic vims talimogene laherparepvec. In certain embodiments, the second anti-cancer agent is an anti-cancer vaccine. In certain embodiments, the second anti-cancer agent is an anti-cancer vaccine selected from the group consistint of a GM-CSF tumor vaccine, a STING/GM-CSF tumor vaccine, and NY-ESO-1. In certain embodiments, the second anti-cancer agent is a cytokine selected from IL-12, IL-15, GM- CSF, and G-CSF.
- the second anti-cancer agent is an immune checkpoint inhibitor (also referred to as immune checkpoint blockers).
- Immune checkpoint inhibitors are a class of therapeutic agents that have the effect of blocking immune checkpoints. See, for example, Pardoll in Nature Reviews Cancer (2012) vol. 12, pages 252-264.
- the immune checkpoint inhibitor is an agent that inhibits one or more of (i) cytotoxic T- lymphocyte- associated antigen 4 (CTLA4), (ii) programmed cell death protein 1 (PD1), (iii) PDL1, (iv) LAB3, (v) B7-H3, (vi) B7-H4, and (vii) TIM3.
- CTLA4 cytotoxic T- lymphocyte- associated antigen 4
- PD1 programmed cell death protein 1
- PDL1 programmed cell death protein 1
- PDL1 programmed cell death protein 1
- PDL1 programmed cell death protein 1
- PD1 programmed cell death protein 1
- PDL1 programmed cell death protein 1
- PD1 programmed cell death
- the immune checkpoint inhibitor is pembrolizumab.
- the second anti-cancer agent is a monoclonal antibody that targets a non-checkpoint target (e.g., herceptin).
- the second anti-cancer agent is a non-cytoxic agent (e.g., a tyrosine -kinase inhibitor).
- the second anti-cancer agent is selected from mitomycin, ribomustin, vincristine, tretinoin, etoposide, cladribine, gemcitabine, mitobronitol, methotrexate, doxorubicin, carboquone, pentostatin, nitracrine, zinostatin, cetrorelix, letrozole, raltitrexed, daunombicin, fadrozole, fotemustine, thymalfasin, sobuzoxane, nedaplatin, aminoglutethimide, amsacrine, proglumide, elliptinium acetate, ketanserin, doxifluridine, etretinate, isotretinoin, streptozocin, nimustine, vindesine, cytarabine, bicalutamide, vinorelbine, vesnarin
- the second anti-cancer agent is radiation therapy.
- the second anti-cancer agent is a MEK Inhibitor.
- the second anti-cancer agent is binimetinib, cobimetinib, refametinib, selumetinib, trametinib, or a pharmaceutically acceptable salt and/or solvate of any of the foregoing.
- the second anti-cancer agent is binimetinib, cobimetinib, selumetinib, trametinib, or a pharmaceutically acceptable salt and/or solvate of any of the foregoing.
- the second anti-cancer agent is trametinib or a pharmaceutically acceptable salt and/or solvate thereof. In certain embodiments, the second anti-cancer agent is trametinib or a pharmaceutically acceptable solvate thereof. In certain embodiments, the second anti-cancer agent is trametinib dimethylsulfoxide. In certain embodiments, the second anti cancer agent is trametinib.
- the second anti-cancer agent is binimetinib or a pharmaceutically acceptable salt and/or solvate thereof. In certain embodiments, the second anti-cancer agent is cobimetinib or a pharmaceutically acceptable salt and/or solvate thereof. In certain embodiments, the second anti-cancer agent is refametinib or a pharmaceutically acceptable salt and/or solvate thereof. In certain embodiments, the second anti-cancer agent is selumetinib or a pharmaceutically acceptable salt and/or solvate thereof.
- the second anti-cancer agent is binimetinib. In certain embodiments, the second anti-cancer agent is cobimetinib. In certain embodiments, the second anti-cancer agent is refametinib. In certain embodiments, the second anti-cancer agent is selumetinib.
- the second anti-cancer agent is a TEAD inhibitor.
- the TEAD inhibitor is selected from those described in WO 2020/243415, the contents of which are herein incorporated by reference in their entirety.
- the TEAD inhibitor is selected from those described in WO 2020/243423, the contents of which are herein incorporated by reference in their entirety.
- the TEAD inhibitor is selected from those described in US Patent No. 11,274,082, the contents of which are herein incorporated by reference in their entirety.
- the second anti-cancer agent is an ERK5 inhibitor.
- the ERK5 inhibitor is selected from those described in WO 2022/051567, the contents of which are herein incorporated by reference in their entirety.
- the ERK5 inhibitor is selected from those described in WO 2022/051565, the contents of which are herein incorporated by reference in their entirety.
- the ERK5 inhibitor is selected from those described in WO 2022/051569, the contents of which are herein incorporated by reference in their entirety.
- the ERK5 inhibitor is selected from those described in WO 2022/051568, the contents of which are herein incorporated by reference in their entirety.
- the method further comprises administering to the subject a third anti-cancer agent. In certain embodiments, the method further comprises administering to the subject a fourth anti-cancer agent. In certain embodiments, the method further comprises administering to the subject a fifth anti-cancer agent.
- the third anti-cancer agent is one of the second anti-cancer agents described above.
- the fourth anti-cancer agent is one of the second anti-cancer agents described above.
- the fifth anti-cancer agent is one of the second anti-cancer agents described above.
- the doses and dosage regimen of the active ingredients used in the combination therapy may be determined by an attending clinician.
- the naphthyl-substituted quinolin-4( 1H)-onc or related compound described herein e.g., a compound of Formula I, II, or III, or other compounds in Section I
- the additional therapeutic agent(s) e.g. the second, third, or fourth, or fifth anti-cancer agent, described above
- the naphthyl-substituted quinolin-4( 1 H)-onc or related compound described herein e.g., a compound of Formula I, II, or III, or other compounds in Section I
- the additional therapeutic agent(s) e.g. the second, third, or fourth, or fifth anti-cancer agent, described above
- the naphthyl-substituted quinolin-4( 1H)-onc or related compound described herein e.g., a compound of Formula I, II, or III, or other compounds in Section I
- the additional therapeutic agent(s) e.g. the second, third, or fourth, or fifth anti-cancer agent, described above
- the naphthyl-substituted quinolin-4( 1 H)-onc or related compound described herein e.g., a compound of Formula I, II, or III, or other compounds in Section I
- the additional therapeutic agent(s) e.g. the second, third, or fourth, or fifth anticancer agent, described above
- a synergistic combination may allow the use of lower dosages of one or more agents and/or less frequent administration of one or more agents of a combination therapy.
- a lower dosage or less frequent administration of one or more agents may lower toxicity of the therapy without reducing the efficacy of the therapy.
- Another aspect of this invention is a kit comprising a therapeutically effective amount of the naphthyl-substituted quinolin-4( 1 H)-onc or related compound described herein (e.g., a compound of Formula I, II, or III, or other compounds in Section I), a pharmaceutically acceptable carrier, vehicle or diluent, and optionally at least one additional therapeutic agent listed above.
- a pharmaceutically acceptable carrier e.g., a compound of Formula I, II, or III, or other compounds in Section I
- vehicle or diluent e.g., a compound of Formula I, II, or III, or other compounds in Section I
- the invention provides pharmaceutical compositions, which comprise a therapeutically-effective amount of one or more of the compounds described above, formulated together with one or more pharmaceutically acceptable carriers (additives) and/or diluents.
- the pharmaceutical compositions may be specially formulated for administration in solid or liquid form, including those adapted for the following: (1) oral administration, for example, drenches (aqueous or non-aqueous solutions or suspensions), tablets, e.g., those targeted for buccal, sublingual, and systemic absorption, boluses, powders, granules, pastes for application to the tongue; (2) parenteral administration, for example, by subcutaneous, intramuscular, intravenous or epidural injection as, for example, a sterile solution or suspension, or sustained-release formulation; (3) topical application, for example, as a cream, ointment, or a controlled-release patch or spray applied to the skin; (4) intravaginally or intrarectally, for example, as a pessary
- a therapeutically effective amount means that amount of a compound, material, or composition comprising a compound of the present invention which is effective for producing some desired therapeutic effect in at least a sub-population of cells in an animal at a reasonable benefit/risk ratio applicable to any medical treatment.
- a therapeutically effective amount is an amount sufficient for inhibition of K-Ras G12D.
- a therapeutically effective amount is an amount sufficient for treating a proliferative disease, such as cancer.
- phrases “pharmaceutically acceptable” is employed herein to refer to those compounds, materials, compositions, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio.
- Wetting agents, emulsifiers and lubricants, such as sodium lauryl sulfate and magnesium stearate, as well as coloring agents, release agents, coating agents, sweetening, flavoring and perfuming agents, preservatives and antioxidants can also be present in the compositions.
- antioxidants examples include: (1) water soluble antioxidants, such as ascorbic acid, cysteine hydrochloride, sodium bisulfate, sodium metabisulfite, sodium sulfite and the like; (2) oil-soluble antioxidants, such as ascorbyl palmitate, butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), lecithin, propyl gallate, alpha-tocopherol, and the like; and (3) metal chelating agents, such as citric acid, ethylenediamine tetraacetic acid (EDTA), sorbitol, tartaric acid, phosphoric acid, and the like.
- water soluble antioxidants such as ascorbic acid, cysteine hydrochloride, sodium bisulfate, sodium metabisulfite, sodium sulfite and the like
- oil-soluble antioxidants such as ascorbyl palmitate, butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), le
- Formulations of the present invention include those suitable for oral, nasal, topical (including buccal and sublingual), rectal, vaginal and/or parenteral administration.
- the formulations may conveniently be presented in unit dosage form and may be prepared by any methods well known in the art of pharmacy.
- the amount of active ingredient which can be combined with a carrier material to produce a single dosage form will vary depending upon the host being treated, the particular mode of administration.
- the amount of active ingredient which can be combined with a carrier material to produce a single dosage form will generally be that amount of the compound which produces a therapeutic effect. Generally, out of one hundred percent, this amount will range from about 0.1 percent to about ninety-nine percent of active ingredient, preferably from about 5 percent to about 70 percent, most preferably from about 10 percent to about 30 percent.
- a formulation of the present invention comprises an excipient selected from the group consisting of cyclodextrins, celluloses, liposomes, micelle forming agents, e.g., bile acids, and polymeric carriers, e.g., polyesters and poly anhydrides; and a compound of the present invention.
- an aforementioned formulation renders orally bioavailable a compound of the present invention.
- Methods of preparing these formulations or compositions include the step of bringing into association a compound of the present invention with the carrier and, optionally, one or more accessory ingredients.
- the formulations are prepared by uniformly and intimately bringing into association a compound of the present invention with liquid carriers, or finely divided solid carriers, or both, and then, if necessary, shaping the product.
- Formulations of the invention suitable for oral administration may be in the form of capsules, cachets, pills, tablets, lozenges (using a flavored basis, usually sucrose and acacia or tragacanth), powders, granules, or as a solution or a suspension in an aqueous or non-aqueous liquid, or as an oil-in-water or water-in-oil liquid emulsion, or as an elixir or syrup, or as pastilles (using an inert base, such as gelatin and glycerin, or sucrose and acacia) and/or as mouth washes and the like, each containing a predetermined amount of a compound of the present invention as an active ingredient.
- a compound of the present invention may also be administered as a bolus, electuary or paste.
- the active ingredient is mixed with one or more pharmaceutically-acceptable carriers, such as sodium citrate or dicalcium phosphate, and/or any of the following: (1) fillers or extenders, such as starches, lactose, sucrose, glucose, mannitol, and/or silicic acid; (2) binders, such as, for example, carboxymethylcellulose, alginates, gelatin, polyvinyl pyrrolidone, sucrose and/or acacia; (3) humectants, such as glycerol; (4) disintegrating agents, such as agar-agar, calcium carbonate, potato or tapioca starch, alginic acid, certain silicates, and sodium carbonate; (5) solution retarding agents, such as paraffin; (6) absorption accelerators, such as quaternary ammonium compounds and surfactants, such
- compositions may also comprise buffering agents.
- Solid compositions of a similar type may also be employed as fillers in soft and hard-shelled gelatin capsules using such excipients as lactose or milk sugars, as well as high molecular weight polyethylene glycols and the like.
- a tablet may be made by compression or molding, optionally with one or more accessory ingredients.
- Compressed tablets may be prepared using binder (for example, gelatin or hydroxypropylmethyl cellulose), lubricant, inert diluent, preservative, disintegrant (for example, sodium starch glycolate or cross-linked sodium carboxymethyl cellulose), surface-active or dispersing agent.
- Molded tablets may be made by molding in a suitable machine a mixture of the powdered compound moistened with an inert liquid diluent.
- the tablets, and other solid dosage forms of the pharmaceutical compositions of the present invention may optionally be scored or prepared with coatings and shells, such as enteric coatings and other coatings well known in the pharmaceutical-formulating art. They may also be formulated so as to provide slow or controlled release of the active ingredient therein using, for example, hydroxypropylmethyl cellulose in varying proportions to provide the desired release profile, other polymer matrices, liposomes and/or microspheres. They may be formulated for rapid release, e.g., freeze-dried.
- compositions may be sterilized by, for example, filtration through a bacteria-retaining filter, or by incorporating sterilizing agents in the form of sterile solid compositions which can be dissolved in sterile water, or some other sterile injectable medium immediately before use.
- These compositions may also optionally contain opacifying agents and may be of a composition that they release the active ingredient(s) only, or preferentially, in a certain portion of the gastrointestinal tract, optionally, in a delayed manner.
- embedding compositions which can be used include polymeric substances and waxes.
- the active ingredient can also be in micro-encapsulated form, if appropriate, with one or more of the above-described excipients.
- Liquid dosage forms for oral administration of the compounds of the invention include pharmaceutically acceptable emulsions, microemulsions, solutions, suspensions, syrups and elixirs.
- the liquid dosage forms may contain inert diluents commonly used in the art, such as, for example, water or other solvents, solubilizing agents and emulsifiers, such as ethyl alcohol, isopropyl alcohol, ethyl carbonate, ethyl acetate, benzyl alcohol, benzyl benzoate, propylene glycol, 1,3-butylene glycol, oils (in particular, cottonseed, groundnut, corn, germ, olive, castor and sesame oils), glycerol, tetrahydrofuryl alcohol, polyethylene glycols and fatty acid esters of sorbitan, and mixtures thereof.
- inert diluents commonly used in the art, such as, for example, water or other solvents, solubilizing agents and
- the oral compositions can also include adjuvants such as wetting agents, emulsifying and suspending agents, sweetening, flavoring, coloring, perfuming and preservative agents.
- adjuvants such as wetting agents, emulsifying and suspending agents, sweetening, flavoring, coloring, perfuming and preservative agents.
- Suspensions in addition to the active compounds, may contain suspending agents as, for example, ethoxylated isostearyl alcohols, polyoxyethylene sorbitol and sorbitan esters, microcrystalline cellulose, aluminum metahydroxide, bentonite, agar-agar and tragacanth, and mixtures thereof.
- suspending agents as, for example, ethoxylated isostearyl alcohols, polyoxyethylene sorbitol and sorbitan esters, microcrystalline cellulose, aluminum metahydroxide, bentonite, agar-agar and tragacanth, and mixtures thereof.
- Formulations of the pharmaceutical compositions of the invention for rectal or vaginal administration may be presented as a suppository, which may be prepared by mixing one or more compounds of the invention with one or more suitable nonirritating excipients or carriers comprising, for example, cocoa butter, polyethylene glycol, a suppository wax or a salicylate, and which is solid at room temperature, but liquid at body temperature and, therefore, will melt in the rectum or vaginal cavity and release the active compound.
- suitable nonirritating excipients or carriers comprising, for example, cocoa butter, polyethylene glycol, a suppository wax or a salicylate, and which is solid at room temperature, but liquid at body temperature and, therefore, will melt in the rectum or vaginal cavity and release the active compound.
- Formulations of the present invention which are suitable for vaginal administration also include pessaries, tampons, creams, gels, pastes, foams or spray formulations containing such carriers as are known in the art to be appropriate.
- Dosage forms for the topical or transdermal administration of a compound of this invention include powders, sprays, ointments, pastes, creams, lotions, gels, solutions, patches and inhalants.
- the active compound may be mixed under sterile conditions with a pharmaceutically-acceptable carrier, and with any preservatives, buffers, or propellants which may be required.
- the ointments, pastes, creams and gels may contain, in addition to an active compound of this invention, excipients, such as animal and vegetable fats, oils, waxes, paraffins, starch, tragacanth, cellulose derivatives, polyethylene glycols, silicones, bentonites, silicic acid, talc and zinc oxide, or mixtures thereof.
- excipients such as animal and vegetable fats, oils, waxes, paraffins, starch, tragacanth, cellulose derivatives, polyethylene glycols, silicones, bentonites, silicic acid, talc and zinc oxide, or mixtures thereof.
- Powders and sprays can contain, in addition to a compound of this invention, excipients such as lactose, talc, silicic acid, aluminum hydroxide, calcium silicates and polyamide powder, or mixtures of these substances.
- Sprays can additionally contain customary propellants, such as chlorofluorohydrocarbons and volatile unsubstituted hydrocarbons, such as butane and propane.
- Transdermal patches have the added advantage of providing controlled delivery of a compound of the present invention to the body.
- dosage forms can be made by dissolving or dispersing the compound in the proper medium.
- Absorption enhancers can also be used to increase the flux of the compound across the skin. The rate of such flux can be controlled by either providing a rate controlling membrane or dispersing the compound in a polymer matrix or gel.
- Ophthalmic formulations are also contemplated as being within the scope of this invention.
- compositions of this invention suitable for parenteral administration comprise one or more compounds of the invention in combination with one or more pharmaceutically-acceptable sterile isotonic aqueous or nonaqueous solutions, dispersions, suspensions or emulsions, or sterile powders which may be reconstituted into sterile injectable solutions or dispersions just prior to use, which may contain sugars, alcohols, antioxidants, buffers, bacteriostats, solutes which render the formulation isotonic with the blood of the intended recipient or suspending or thickening agents.
- aqueous and nonaqueous carriers examples include water, ethanol, polyols (such as glycerol, propylene glycol, polyethylene glycol, and the like), and suitable mixtures thereof, vegetable oils, such as olive oil, and injectable organic esters, such as ethyl oleate.
- polyols such as glycerol, propylene glycol, polyethylene glycol, and the like
- vegetable oils such as olive oil
- injectable organic esters such as ethyl oleate.
- Proper fluidity can be maintained, for example, by the use of coating materials, such as lecithin, by the maintenance of the required particle size in the case of dispersions, and by the use of surfactants.
- compositions may also contain adjuvants such as preservatives, wetting agents, emulsifying agents and dispersing agents. Prevention of the action of microorganisms upon the subject compounds may be ensured by the inclusion of various antibacterial and antifungal agents, for example, paraben, chlorobutanol, phenol sorbic acid, and the like. It may also be desirable to include isotonic agents, such as sugars, sodium chloride, and the like into the compositions. In addition, prolonged absorption of the injectable pharmaceutical form may be brought about by the inclusion of agents which delay absorption such as aluminum monostearate and gelatin.
- adjuvants such as preservatives, wetting agents, emulsifying agents and dispersing agents.
- Injectable depot forms are made by forming microencapsule matrices of the subject compounds in biodegradable polymers such as polylactide-polyglycolide. Depending on the ratio of drug to polymer, and the nature of the particular polymer employed, the rate of drug release can be controlled. Examples of other biodegradable polymers include poly(orthoesters) and poly(anhydrides). Depot injectable formulations are also prepared by entrapping the drug in liposomes or microemulsions which are compatible with body tissue.
- biodegradable polymers such as polylactide-polyglycolide.
- Depot injectable formulations are also prepared by entrapping the drug in liposomes or microemulsions which are compatible with body tissue.
- the compounds of the present invention are administered as pharmaceuticals, to humans and animals, they can be given per se or as a pharmaceutical composition containing, for example, 0.1 to 99% (more preferably, 10 to 30%) of active ingredient in combination with a pharmaceutically acceptable carrier.
- the preparations of the present invention may be given orally, parenterahy, topically, or rectahy. They are of course given in forms suitable for each administration route. For example, they are administered in tablets or capsule form, by injection, inhalation, eye lotion, ointment, suppository, etc. administration by injection, infusion or inhalation; topical by lotion or ointment; and rectal by suppositories. Oral administrations are preferred.
- parenteral administration and “administered parenterahy” as used herein means modes of administration other than enteral and topical administration, usually by injection, and includes, without limitation, intravenous, intramuscular, intraarterial, intrathecal, intracapsular, intraorbital, intracardiac, intradermal, intraperitoneal, transtracheal, subcutaneous, subcuticular, intraarticular, subcapsular, subarachnoid, intraspinal and intrastemal injection and infusion.
- systemic administration means the administration of a compound, drug or other material other than directly into the central nervous system, such that it enters the patient’s system and, thus, is subject to metabolism and other like processes, for example, subcutaneous administration.
- These compounds may be administered to humans and other animals for therapy by any suitable route of administration, including orally, nasally, as by, for example, a spray, rectally, intravaginally, parenterally, intracisternally and topically, as by powders, ointments or drops, including buccally and sublingually.
- the compounds of the present invention which may be used in a suitable hydrated form, and/or the pharmaceutical compositions of the present invention, are formulated into pharmaceutically-acceptable dosage forms by conventional methods known to those of skill in the art.
- the selected dosage level will depend upon a variety of factors including the activity of the particular compound of the present invention employed, or the ester, salt or amide thereof, the route of administration, the time of administration, the rate of excretion or metabolism of the particular compound being employed, the rate and extent of absorption, the duration of the treatment, other drugs, compounds and/or materials used in combination with the particular compound employed, the age, sex, weight, condition, general health and prior medical history of the patient being treated, and like factors well known in the medical arts.
- a physician or veterinarian having ordinary skill in the art can readily determine and prescribe the effective amount of the pharmaceutical composition required.
- the physician or veterinarian could start doses of the compounds of the invention employed in the pharmaceutical composition at levels lower than that required in order to achieve the desired therapeutic effect and gradually increase the dosage until the desired effect is achieved.
- a suitable daily dose of a compound of the invention will be that amount of the compound which is the lowest dose effective to produce a therapeutic effect. Such an effective dose will generally depend upon the factors described above.
- the compounds are administered at about 0.01 mg/kg to about 200 mg/kg, more preferably at about 0.1 mg/kg to about 100 mg/kg, even more preferably at about 0.5 mg/kg to about 50 mg/kg.
- the effective amount may be less than when the agent is used alone.
- the effective daily dose of the active compound may be administered as two, three, four, five, six or more sub-doses administered separately at appropriate intervals throughout the day, optionally, in unit dosage forms. Preferred dosing is one administration per day.
- the invention further provides a unit dosage form (such as a tablet or capsule) comprising a naphthyl-substituted quinolin-4( 1H)-onc or related compound described herein in a therapeutically effective amount for the treatment of a medical disorder described herein.
- a unit dosage form such as a tablet or capsule
- a naphthyl-substituted quinolin-4( 1H)-onc or related compound described herein in a therapeutically effective amount for the treatment of a medical disorder described herein.
- Step 1 Benzyl 4-(8-tert-butoxycarbonyl-3,8-diazabicyclo[3.2.1]octan-3-yl)-2-chloro-6,8- dihydro-5H-pyrido[3,4-d]pyrimidine-7-carboxylate
- Step 2 Benzyl 4-(8-tert-butoxycarbonyl-3,8-diazabicyclo[3.2.1]octan-3-yl)-2-[[(2R,8S)-2- fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]-6,8-dihydro-5H-pyrido[3,4- d]pyrimidine-7-carboxylate
- Step 3 tert- Butyl 3-[2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]- 5,6,7,8-tetrahydropyrido[3,4-d]pyrimidin-4-yl]-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
- Step 4 tert -Butyl 3-[2-[[(2/?,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]-7- [3-(methoxymethoxy)-8-methyl-l-naphthyl]-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-4-yl]- 3,8-diazabicyclo[3.2.1]octane-8-carboxylate
- Step 5 4-[4-(3,8-Diazabicyclo[3.2.1]octan-3-yl)-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-7-yl]-5-methyl- naphthalen-2-ol (1-31)
- the assay quantifies the inhibition of SOSl-mediated nucleotide exchange of K-Ras (G12D, G12V, G12C, or wild-type).
- K-Ras K-Ras
- Tb-streptavidin Tb-streptavidin
- the K-Ras complex was allowed to incubate with varied concentrations of test compounds before the addition of the mixture of SOS1 and fluorescently- labeled GTP (BODIPY-GTP) to initiate the reaction.
- BODIPY-GTP fluorescently- labeled GTP
- the TR-FRET signal was monitored after 30 min.
- the TR-FRET signal was generated by Tb and BODIPY-GTP, that displaced unlabeled GDP.
- Biotinylated K-Ras enzyme G12D, G12V, G12C, and wild-type
- SOS1 were purchased from GenScript.
- Other materials were: Tb-streptavidin (Cisbio-610SATLB), BODIPY FL GTP (Thermo-G12411), and white 384-well ProxiPlus Assay Plates (Perkin-Elmer- 6008289).
- the assay buffer was 20 mM HEPES pH 7.5, 50 mM NaCl, 10 mM MgCl2, 0.01% Tween-20, 1 mM DTT.
- Total assay volume was 15 ⁇ L, and final assay concentrations were biotinylated K-Ras enzyme (G12D, G12V, or wild-type; 1.0 nM), SOS1 (200 nM), Tb-streptavidin (1 nM), and BODIPY-GTP (100 nM).
- the assay was conducted at 23 °C. Controls included all the reagents, but replaced test compound solution with DMSO (0% inhibition control) or 100 mM GDP (100% inhibition control).
- Test compound was dissolved and diluted in DMSO (100 mM top concentration, 3-fold dilution, 11 doses), then transferred to the assay plate using Echo. The final DMSO concentration was 1.5%.
- the mixture of 1.5x K-Ras enzyme (G12D, G12V, G12C, or wild-type) and Streptavidin-Tb in assay buffer was prepared, and then pre-incubated at 23°C for 1 hour. The enzyme mixture was then dispensed into the assay plate (10 ⁇ L/weII). The plate was allowed to react with enzyme and compound for 30 minutes. A mixture of 3x SOS1 and BODIPY-GTP in assay buffer was added (5 ⁇ L/well).
- the plate was spun at 1000 rpm for 30 seconds and then sealed. After incubating for 30 min, the assay signal was detected with Envision plate reader. The TR-FRET ratios were used to calculate percent inhibition values for each concentration of compound, using the no inhibition and 100% inhibition controls. IC50 values were calculated by Xlfit from the percent inhibition values using the 4-parameter logistical equation. Part II - Results
- An exemplary compound was tested for its ability to inhibit phosphorylation of ERK downstream of K-Ras G12D using a homogeneous time-resolved fluorescence assay. Assay procedures and results are described below.
- the culture/assay medium was RPMI-1640 +10% FBS. Cells were seeded at 6,000 cells/well, 50 mE/well. Compound treatment time was 4 hours. Controls replaced test compound solution with 0.8% DMSO (max control) or 0.2 mM trametinib (min control).
- Procedure Cells were harvested from T150 growthflask using 0.05% trypsin/EDTA solution. Medium (10 mL) was added to stop trypsinizing. The cells were pipetted into a conical-bottomed, 50-mL centrifuge tube and centrifuged for 5 minutes at 1,000 rpm. The cell pellet was resuspended in medium, and cell count was determined (ViCell counter). Cell density was adjusted using fresh medium, then 384-well poly-D-lysine coated plates were seeded with 50 ⁇ L/well of the cell suspension. The cell plate was incubated overnight in a 37 °C, 5% CO2 incubator.
- Test compounds were dissolved and diluted in DMSO (20 mM top concentration, 3-fold dilution, 10 doses) using Tecan. Compounds were added to the cell plate. The final DMSO concentration was 0.8%. The cell plate was incubated for 4 hours in the incubator.
- Detection was conducted using a CisBio pERK kit. Kit-supplied lysis buffer was prepared and kept on ice. Concentrated (4x) lysis buffer was diluted with 3 volumes of deionized water, then lOOx of the blocking agent was added. Media was removed from the cell plate. Lysis buffer (35 mE/well) was added, and then the plate was placed on a plate agitator and shaken at 300 rpm, 4 °C for 40 minutes. Upon completion, the resulting lysate plate was centrifuged for 3 minutes at 1500 rpm.
- Assay plates were prepared containing the HTRF antibody buffer. For each assay plate, 50 ⁇ L of d2-conjugate antibody was mixed with 950 ⁇ L of detection buffer. For each assay plate, 50 ⁇ L of cryptate antibody was mixed with 950 ⁇ L of detection buffer. The two diluted antibodies were then mixed together, and 3.4 ⁇ L of the resulting antibody buffer was dispensed to wells of an empty 384-well white Proxi plate. The plate was sealed and centrifuged 30 minutes at 1,000 rpm.
- An exemplary compound was tested for its ability to inhibit the binding of GMPPNP- bound KRas (G12D or WT) to the Ras binding domain of RAF.
- KRas protein was biotinylated and RAF protein was FLAG-tagged. Compounds were incubated with KRas-GMPPNP for 30 minutes at room temperature, followed by addition of RAF protein, Tb-anti-FLAG and SA-XL665 HTRF reagents. Fluorescence detection was carried out after 1.5 hours of incubation. IC50 values were calculated using a four-parameter fit.
- GMPPNP-loaded KRAS-G12D Biotinylated GMPPNP-loaded KRAS-G12D, GMPPNP-loaded KRAS-WT and FLAG-tagged-Raf-RBD were purchased from GenScript. GMPPNP was obtained from Sigma. Perkin Elmer and Cisbio provided the Tb-anti-FLAG and streptavidin XL665 reagents and the white 384W ProxiPlus assay plates.
- the assay buffer was 20 mM HEPES, pH 7.5, 150 mM NaCl,
- HTRF ratios were used to calculate percent inhibition values for each concentration of compound, using the no inhibition and 100% inhibition controls.
- IC50 values were calculated by Xlfit from the percent inhibition values using the 4-parameter logistical equation.
- EXAMPLE 5 Assay for Inhibition of K-Ras G12D-mediated Phosphorylation of ERK
- Compounds may be tested for ability to inhibit phosphorylation of ERK downstream of K-Ras G12D according to the following experimental procedure.
- AGS cells (ATCC CRL-1739) expressing G12D are grown in DMEM medium supplemented with 10% fetal bovine serum, 10 mM HEPES, and Penicillin/Streptomycin. Cells are plated in tissue culture treated 96 well plates at a density of 40,000 cells/well and allowed to attach for 12-14 hours. Diluted test compound is then added in a final concentration of 0.5% DMSO. After 3 hours, the medium is removed, 150 pL of 4.0% formaldehyde is added and the plates incubated at room temperature for 20 minutes. Then, the plates are washed with PBS, and permeabilized with 150 pL of ice cold 100% methanol for 10 minutes. Non-specific antibody binding to the plates is blocked using 100 pL Licor blocking buffer (Li-Cor Biotechnology, Lincoln NE) for 1 hour at room temperature.
- Licor blocking buffer Li-Cor Biotechnology, Lincoln NE
- the amount of phospho-ERK is determined using an antibody specific for the phosphorylated form of ERK and compared to the amount of GAPDH. Primary antibodies used for the detection are added as follows: Phospho-ERK (Cell Signaling cs-9101) diluted 1:500 and GAPDH(Millipore MAB374) diluted 1:5000 in Licor block + 0.05%Tween 20. The plates are incubated for 2 hours at room temperature. Then, the plates are washed with PBS + 0.05%
- Secondary antibodies used to visualize primary antibodies are added as follows: Anti- rabbit-680 diluted 1 : 1000 and Anti-mouse-800 diluted 1 : 1000 both in Licor block +0.05% TweeN20, and are incubated for 1 hour at room temperature. Then, the plates are washed with PBS + 0.05% Tween 20. A 100 pL aliquot of PBS is added to each well and the plates are read on a Li-Cor Odyssey CLX plate reader.
- the phospho-ERK Thr202/Tyr204 signal is normalized to the GAPDH signal for each well and percent of DMSO control values are calculated. IC50 values are generated using a 4- parameter fit of the dose response curve
- EXAMPLE 6 Biological Assay for Inhibiting 3D Cancer Cell Cultures Using Combination Therapy
- Exemplary compounds having K-Ras G12D inhibitory activity may be tested for ability to inhibit 3D cancer cell cultures of pancreatic cancer and lung cancer. Assay procedures are described below.
- cells are seeded in 231 soft agar in 96-well, low-attachment tissue-culture plates. After 24 hours, cells are treated with a concentration range of one or more K-Ras G12D inhibitors. At the time of treatment, assay plates that did not receive treatment are collected and cell viability is measured using alamarBlue reagent to establish baseline cell viability. Treated plates are incubated for 7 days, and alamarBlue readout is then collected.
- EXAMPLE 7 Biological Assay for Inhibiting Mouse Xenograft Tumor Growth
- Exemplary compounds having K-Ras G12D inhibitory activity may be tested, alone or in combination with other agents, such as trametinib, for ability to inhibit tumor growth in patient- derived mouse xenograft models of pancreatic cancer and lung cancer. Assay procedures are described below.
- mice (6-8 weeks old) are inoculated subcutaneously in the right flank with a primary human tumor xenograft model tumor fragment (2-3 mm 3 in diameter) for tumor development.
- mice are inoculated with a human lung adenocarcinoma tumor model (MSCLC, ADC model LU6424) that harbors a BRAF mutation (LU6424);
- mice are inoculated with a human pancreatic tumor model (adenosquamous carcinoma model PA6258) with a K-Ras G12D mutation (PA6258).
- mean tumor volume reaches approximately 150-200 mm 3 , animals are randomly allocated to appropriate treatment groups.
- mice are treated with one of the following: (l)Vehicle control, (2) Exemplary K-Ras G12D inhibitor alone, (3) Second anti-cancer agent alone, or (4) Combination of K-Ras G12D inhibitor and second anti-cancer agent. Tumors are measured twice per week using calipers.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The invention provides naphthyl-substituted quinolin-4(1H)-ones and related compounds, pharmaceutical compositions, their use for inhibiting K-Ras G12D activity, and their use in the treatment of medical disorders, such as cancer.
Description
NAPHTHYL-SUBSTITUTED QUINOLINE-4(1H)-ONES AND RELATED COMPOUNDS AND THEIR USE IN TREATING MEDICAL CONDITIONS
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This application claims the benefit of and priority to United States Provisional Patent Application serial number 63/173,274, filed April 9, 2021, the contents of which are hereby incorporated by reference in their entirety.
FIELD OF THE INVENTION
[0002] The invention provides naphthyl-substituted quinolin-4(1H)-oncs and related compounds, pharmaceutical compositions, their use for inhibiting K-Ras G12D activity, and their use in the treatment of medical disorders, such as cancer.
BACKGROUND
[0003] Cancer continues to be a significant health problem despite the substantial research efforts and scientific advances reported in the literature for treating this disease. Solid tumors, including breast cancer, lung cancer, and pancreatic cancer remain prevalent among the world population. Current treatment options for these cancers are not effective for all patients and/or can have substantial adverse side effects. New therapies are needed to address this unmet need in cancer therapy.
[0004] Kirsten rat sarcoma 2 Viral Oncogene Homolog (K-Ras) is a GTPase that has been reported to impact cell survival, proliferation, migration, and differentiation. Evidence suggests that activating mutations of K-Ras (for example, K-Ras G12D and G12C) may be involved in the onset and progression of multiple cancers, and that inhibition of such mutant K-Ras proteins may provide a therapeutic benefit for cancer. Use of certain K-Ras G12D inhibitor compounds for treating cancer has been described in, for example, international patent application WO 2021/041671, the contents of which are herein incorporated by reference in its entirety.
[0005] New compounds having inhibitory activity toward K-Ras G12D are therefore needed as therapeutic agents for the treatment of medical disorders, such as cancer. The present invention addresses the foregoing needs and provides other related advantages.
SUMMARY
[0006] The invention provides naphthyl-substituted quinolin-4( 1H)-oncs and related compounds, pharmaceutical compositions, their use for inhibiting K-Ras G12D activity, and their use in the treatment of medical disorders, such as cancer. In particular, one aspect of the invention provides a collection of naphthyl-substituted 3,4-dihydroisoquinolin- 1 (2H)-oncs and related compounds, such as a compound represented by Formula I:
or a pharmaceutically acceptable salt thereof, where the variables are as defined in the detailed description. Further description of additional collections of naphthyl-substituted quinolin-4( 1H)- ones and related compounds are described in the detailed description. The compounds may be part of a pharmaceutical composition comprising a pharmaceutically acceptable carrier.
[0007] Another aspect of the invention provides a collection of naphthyl-substituted quinolin- 4(1 H)-oncs and related compounds, such as a compound represented by Formula II:
or a pharmaceutically acceptable salt thereof, where the variables are as defined in the detailed description. Further description of additional collections of naphthyl-substituted quinolin-4( 1H)- ones and related compounds are described in the detailed description. The compounds may be part of a pharmaceutical composition comprising a pharmaceutically acceptable carrier.
[0008] Another aspect of the invention provides a collection of naphthyl-substituted AH- pyrazino[l,2-a]pyrazin-4-ones and related compounds, such as a compound represented by Formula III:
or a pharmaceutically acceptable salt thereof, where the variables are as defined in the detailed description. Further description of additional collections of naphthyl-substituted AH- pyrazino[l,2-a]pyrazin-4-ones and related compounds are described in the detailed description. The compounds may be part of a pharmaceutical composition comprising a pharmaceutically acceptable carrier.
[0009] Another aspect of the invention provides a method of treating a disorder mediated by K- Ras G12D in a subject. The method comprises administering a therapeutically effective amount of a compound described herein, such as a compound of Formula I, II, or III, to a subject in need thereof to treat the disorder, as further described in the detailed description.
[0010] Another aspect of the invention provides a method of inhibiting K-Ras G12D activity. The method comprises contacting K-Ras G12D with an effective amount of a compound described herein, such as a compound of Formula I, II, or III, to inhibit K-Ras G12D activity, as further described in the detailed description.
DETAILED DESCRIPTION
[0011] The invention provides naphthyl-substituted quinolin-4( 1H)-oncs and related compounds, pharmaceutical compositions, their use for inhibiting K-Ras G12D activity, and their use in the treatment of medical disorders, such as cancer. The practice of the present invention employs, unless otherwise indicated, conventional techniques of organic chemistry, pharmacology, molecular biology (including recombinant techniques), cell biology, biochemistry, and immunology. Such techniques are explained in the literature, such as in “Comprehensive
Organic Synthesis” (B.M. Trost & I. Fleming, eds., 1991-1992); “Handbook of experimental immunology” (D.M. Weir & C.C. Blackwell, eds.); “Current protocols in molecular biology” (F.M. Ausubel el al., eds., 1987, and periodic updates); and “Current protocols in immunology” (J.E. Coligan et al., eds., 1991), each of which is herein incorporated by reference in its entirety.
[0012] Various aspects of the invention are set forth below in sections; however, aspects of the invention described in one particular section are not to be limited to any particular section. Further, when a variable is not accompanied by a definition, the previous definition of the variable controls.
Definitions
[0013] Compounds of the present invention include those described generally herein, and are further illustrated by the classes, subclasses, and species disclosed herein. As used herein, the following definitions shall apply unless otherwise indicated. These definitions apply regardless of whether a term is used by itself or in combination with other terms, unless otherwise indicated. Hence, the definition of “alkyl” applies to “alkyl” as well as the “alkyl” portions of “- O-alkyl” etc. For purposes of this invention, the chemical elements are identified in accordance with the Periodic Table of the Elements, CAS version, Handbook of Chemistry and Physics, 75th Ed. Additionally, general principles of organic chemistry are described in “Organic Chemistry”, Thomas Sorrell, University Science Books, Sausalito: 1999, and “March’s Advanced Organic Chemistry”, 5th Ed., Ed.: Smith, M.B. and March, J., John Wiley & Sons, New York: 2001, the entire contents of which are hereby incorporated by reference.
[0014] The term “aliphatic” or “aliphatic group”, as used herein, means a straight-chain (i.e., unbranched) or branched, substituted or unsubstituted hydrocarbon chain that is completely saturated or that contains one or more units of unsaturation, or a monocyclic hydrocarbon or bicyclic hydrocarbon that is completely saturated or that contains one or more units of unsaturation, but which is not aromatic (also referred to herein as “cycloaliphatic”), that has a single point of attachment to the rest of the molecule. Unless otherwise specified, aliphatic groups contain 1-6 aliphatic carbon atoms. In some embodiments, aliphatic groups contain 1-5 aliphatic carbon atoms. In other embodiments, aliphatic groups contain 1-4 aliphatic carbon atoms. In still other embodiments, aliphatic groups contain 1-3 aliphatic carbon atoms, and in yet other embodiments, aliphatic groups contain 1-2 aliphatic carbon atoms. In some
embodiments, “cycloaliphatic” refers to a monocyclic C3-C6 hydrocarbon that is completely saturated or that contains one or more units of unsaturation, but which is not aromatic, that has a single point of attachment to the rest of the molecule. Suitable aliphatic groups include, but are not limited to, linear or branched, substituted or unsubstituted alkyl, alkenyl, alkynyl groups and hybrids thereof such as (cycloalkyl)alkyl, (cycloalkenyl)alkyl or (cycloalkyl)alkenyl.
[0015] As used herein, the term “bicyclic ring” or “bicyclic ring system” refers to any bicyclic ring system, i.e. carbocyclic or heterocyclic, saturated or having one or more units of unsaturation, having one or more atoms in common between the two rings of the ring system. Thus, the term includes any permissible ring fusion, such as ortho -fused or spirocyclic. As used herein, the term “heterobicyclic” is a subset of “bicyclic” that requires that one or more heteroatoms are present in one or both rings of the bicycle. Such heteroatoms may be present at ring junctions and are optionally substituted, and may be selected from nitrogen (including N- oxides), oxygen, sulfur (including oxidized forms such as sulfones and sulfonates), phosphorus (including oxidized forms such as phosphates), boron, etc. In some embodiments, a bicyclic group has 7-12 ring members and 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur. As used herein, the term “bridged bicyclic” refers to any bicyclic ring system, i.e. carbocyclic or heterocyclic, saturated or partially unsaturated, having at least one bridge. As defined by IUPAC, a “bridge” is an unbranched chain of atoms or an atom or a valence bond connecting two bridgeheads, where a “bridgehead” is any skeletal atom of the ring system which is bonded to three or more skeletal atoms (excluding hydrogen). In some embodiments, a bridged bicyclic group has 7-12 ring members and 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur. Such bridged bicyclic groups are well known in the art and include those groups set forth below where each group is attached to the rest of the molecule at any substitutable carbon or nitrogen atom. Unless otherwise specified, a bridged bicyclic group is optionally substituted with one or more substituents as set forth for aliphatic groups. Additionally or alternatively, any substitutable nitrogen of a bridged bicyclic group is optionally substituted. Exemplary bicyclic rings include:
[0017] The term “lower alkyl” refers to a C 1-4 straight or branched alkyl group. Exemplary lower alkyl groups are methyl, ethyl, propyl, isopropyl, butyl, isobutyl, and tert-butyl.
[0018] The term “lower haloalkyl” refers to a CIM straight or branched alkyl group that is substituted with one or more halogen atoms.
[0019] The term “heteroatom” means one or more of oxygen, sulfur, nitrogen, phosphorus, or silicon (including, any oxidized form of nitrogen, sulfur, phosphorus, or silicon; the quatemized form of any basic nitrogen or; a substitutable nitrogen of a heterocyclic ring, for example N (as in 3 , 4 - d i h y d ro - 2 H- p y rrp l y l ) , NH (as in pyrrolidinyl) or NR+ (as in N-substituted pyrrolidinyl)).
[0020] The term “unsaturated,” as used herein, means that a moiety has one or more units of unsaturation.
[0021] As used herein, the term “bivalent C1-8 (or C1-6,) saturated or unsaturated, straight or branched, hydrocarbon chain”, refers to bivalent alkylene, alkenylene, and alkynylene chains that are straight or branched as defined herein.
[0022] The term “alkylene” refers to a bivalent alkyl group. An “alkylene chain” is a polymethylene group, i.e., -(CH2)n-, wherein n is a positive integer, preferably from 1 to 6, from 1 to 4, from 1 to 3, from 1 to 2, or from 2 to 3. A substituted alkylene chain is a polymethylene group in which one or more methylene hydrogen atoms are replaced with a substituent. Suitable substituents include those described below for a substituted aliphatic group.
[0023] The term “-(Co alkylene)-" refers to a bond. Accordingly, the term “-(Co-3 alkylene)-” encompasses a bond (i.e., Co) and a -(C1-3 alkylene)- group.
[0024] The term “alkenylene” refers to a bivalent alkenyl group. A substituted alkenylene chain is a polymethylene group containing at least one double bond in which one or more hydrogen atoms are replaced with a substituent. Suitable substituents include those described below for a substituted aliphatic group.
[0025] The term “halogen” means F, Cl, Br, or I.
[0026] The term “aryl” used alone or as part of a larger moiety as in “aralkyl,” “aralkoxy,” or “aryloxyalkyl,” refers to monocyclic or bicyclic ring systems having a total of five to fourteen ring members, wherein at least one ring in the system is aromatic and wherein each ring in the system contains 3 to 7 ring members. The term “aryl” may be used interchangeably with the term “aryl ring.” In certain embodiments of the present invention, “aryl” refers to an aromatic ring system which includes, but not limited to, phenyl, biphenyl, naphthyl, anthracyl and the like, which may bear one or more substituents. Also included within the scope of the term “aryl,” as it is used herein, is a group in which an aromatic ring is fused to one or more non-aromatic rings, such as indanyl, phthalimidyl, naphthimidyl, phenanthridinyl, or tetrahydronaphthyl, and the like. The term “phenylene” refers to a multivalent phenyl group having the appropriate number of open valences to account for groups attached to it. For example, “phenylene” is a bivalent phenyl group when it has two groups attached
trivalent phenyl group when it has three groups attached to it (e.g.,
). The term
“arylene” refers to a bivalent aryl group.
[0027] The terms “heteroaryl” and “heteroar-,” used alone or as part of a larger moiety, e.g., “heteroaralkyl,” or “heteroaralkoxy,” refer to groups having 5 to 10 ring atoms, preferably 5, 6, or 9 ring atoms; having 6, 10, or 14 p electrons shared in a cyclic array; and having, in addition to carbon atoms, from one to five heteroatoms. The term “heteroatom” refers to nitrogen, oxygen, or sulfur, and includes any oxidized form of nitrogen or sulfur, and any quaternized form of a basic nitrogen. Heteroaryl groups include, without limitation, thienyl, furanyl, pyrrolyl, imidazolyl, pyrazolyl, triazolyl, tetrazolyl, oxazolyl, isoxazolyl, oxadiazolyl, thiazolyl, isothiazolyl, thiadiazolyl, pyridyl, pyridazinyl, pyrimidinyl, pyrazinyl, indolizinyl, purinyl, naphthyridinyl, and pteridinyl. The terms “heteroaryl” and “heteroar-”, as used herein, also include groups in which a hetero aromatic ring is fused to one or more aryl, cycloaliphatic, or heterocyclyl rings, where unless otherwise specified, the radical or point of attachment is on the heteroaromatic ring or on one of the rings to which the hetero aromatic ring is fused. Nonlimiting examples include indolyl, isoindolyl, benzothienyl, benzofuranyl, dibenzofuranyl, indazolyl, benzimidazolyl, benzthiazolyl, quinolyl, isoquinolyl, cinnolinyl, phthalazinyl, quinazolinyl, quinoxalinyl, 4/7-quinolizinyl, carbazolyl, acridinyl, phenazinyl, phenothiazinyl, phenoxazinyl, tetrahydroquinolinyl, and tetrahydroisoquinolinyl. A heteroaryl group may be mono- or bicyclic. The term “heteroaryl” may be used interchangeably with the terms “heteroaryl ring,” “heteroaryl group,” or “heteroaromatic,” any of which terms include rings that are optionally substituted. The term “heteroaralkyl” refers to an alkyl group substituted by a heteroaryl, wherein the alkyl and heteroaryl portions independently are optionally substituted.
[0028] The term “heteroarylene” refers to a multivalent heteroaryl group having the appropriate number of open valences to account for groups attached to it. For example, “heteroarylene” is a bivalent heteroaryl group when it has two groups attached to it; “heteroarylene” is a trivalent heteroaryl group when it has three groups attached to it. The term “pyridinylene” refers to a multivalent pyridine radical having the appropriate number of open valences to account for groups attached to it. For example, “pyridinylene” is a bivalent pyridine radical when it has two
[0029] As used herein, the terms “heterocycle,” “heterocyclyl,” “heterocyclic radical,” and “heterocyclic ring” are used interchangeably and refer to a stable 5- to 7-membered monocyclic or 7-10-membered bicyclic heterocyclic moiety that is either saturated or partially unsaturated, and having, in addition to carbon atoms, one or more, preferably one to four, heteroatoms, as defined above. When used in reference to a ring atom of a heterocycle, the term "nitrogen" includes a substituted nitrogen. As an example, in a saturated or partially unsaturated ring having 0-3 heteroatoms selected from oxygen, sulfur or nitrogen, the nitrogen may be N (as in 3,4- di h y dro-2H-py rro l y l ) , NH (as in pyrrolidinyl), or +NR (as in A-substitutcd pyrrolidinyl).
[0030] A heterocyclic ring can be attached to its pendant group at any heteroatom or carbon atom that results in a stable structure and any of the ring atoms can be optionally substituted. Examples of such saturated or partially unsaturated heterocyclic radicals include, without limitation, tetrahydrofuranyl, tetrahydrothiophenyl pyrrolidinyl, piperidinyl, pyrrolinyl, tetrahydroquinolinyl, tetrahydroisoquinolinyl, decahydroquinolinyl, oxazolidinyl, piperazinyl, dioxanyl, dioxolanyl, diazepinyl, oxazepinyl, thiazepinyl, morpholinyl, 2-oxa-6- azaspiro[3.3]heptane, and quinuclidinyl. The terms “heterocycle,” “heterocyclyl,” “heterocyclyl ring,” “heterocyclic group,” “heterocyclic moiety,” and “heterocyclic radical,” are used interchangeably herein, and also include groups in which a heterocyclyl ring is fused to one or more aryl, heteroaryl, or cycloaliphatic rings, such as indolinyl, 3H-indolyl, chromanyl, phenanthridinyl, or tetrahydroquinolinyl. A heterocyclyl group may be mono- or bicyclic. The term “heterocyclylalkyl” refers to an alkyl group substituted by a heterocyclyl, wherein the alkyl and heterocyclyl portions independently are optionally substituted. The term “oxo-heterocyclyl” refers to a heterocyclyl substituted by an oxo group. The term “heterocyclylene” refers to a multivalent heterocyclyl group having the appropriate number of open valences to account for groups attached to it. For example, “heterocyclylene” is a bivalent heterocyclyl group when it has two groups attached to it; “heterocyclylene” is a trivalent heterocyclyl group when it has three groups attached to it.
[0031] As used herein, the term “partially unsaturated” refers to a ring moiety that includes at least one double or triple bond. The term “partially unsaturated” is intended to encompass rings having multiple sites of unsaturation, but is not intended to include aryl or heteroaryl moieties, as herein defined.
[0032] As described herein, compounds of the invention may contain “optionally substituted” moieties. In general, the term “substituted,” whether preceded by the term “optionally” or not, means that one or more hydrogens of the designated moiety are replaced with a suitable substituent. Unless otherwise indicated, an “optionally substituted” group may have a suitable substituent at each substitutable position of the group, and when more than one position in any given structure may be substituted with more than one substituent selected from a specified group, the substituent may be either the same or different at every position. Combinations of substituents envisioned by this invention are preferably those that result in the formation of stable or chemically feasible compounds. The term “stable,” as used herein, refers to compounds that are not substantially altered when subjected to conditions to allow for their production, detection, and, in certain embodiments, their recovery, purification, and use for one or more of the purposes disclosed herein.
[0033] Each optional substituent on a substitutable carbon is a monovalent substituent independently selected from halogen; -(CH2)0-4Rº; -(CH2)0-4OR°; -O(CH2)0-4R°, -O-(CH2)0- 4C(O)OR°; -(CH2)0-4CH(OR°)2; -(CH2)0-4SR°; - (CH2)0-4Ph, which may be substituted with R°; -(CH2)0-4O(CH2)o-iPh which may be substituted with R°; -CH=CHPh, which may be substituted with R°; -(CH2)0-4O(CH2)O-I -pyridyl which may be substituted with R°; -NCh; -CN; - N3; -(CH2)0-4N(R°)2; -(CH2)0-4N(R°X:(O)R°; -N(R°)C(S)R°; -(CH2)0-4N(R°)C(O)NR°2;
-N(R°)C(S)NR°2; -(CH2)0-4N(R°)C(O)OR°; -N(R°)N(R°)C(O)R°; -N(R°)N(R°)C(O)NRº2;
-N(R°)N(R°)C(O)OR°; -(CH2)0-4C(O)R°; -C(S)R°; -(CH2)0-4C(O)OR°; -(CH2)0-4C(O)SR°; -(CH2)0-4C(O)OSiR°3; -(CH2)0-4OC(O)R°; -OC(O) (CH2)0-4SR- SC(S)SR°; -(CH2)0-4SC(O)R°; -(CH2)0-4C(O)NR°2; -C(S)NR°2; -C(S)SR°; -SC(S)SR°, -(CH2)0-4OC(O)NR°2;
-C(O)N(OR°)R°; -C(O)C(O)R°; -C(O)CH2C(O)R°; -C(NOR°)R°; -(CH2)0-4SSR°; -(CH2)0- 4S(O)2R°; -(CH2)0-4S(O)2OR°; - -(CH2)0-4OS(O)2R°; -S(O)2NR°2; -S(O)(NR°)R°; -
S(O)2N=C(NR°2)2; -(CH2)0-4S(O)R°; -N(R°)S(O)2NRº2 ; -N(R°)S(O)2R°; -N(OR°)R°; -
C(NH)NR°2; -P(0)2R°; -P(0)R°2; -0P(0)R°2; -0P(0)(0R°)2; SiR°3; -(C1-4 straight or branched alkylene)0-N(R°)2; or — (C i 4 straight or branched alkylene)C(0)0-N(R°)2.
[0034] Each R° is independently hydrogen, Ci-6 aliphatic, -CH2Ph, -0(CH2)o-iPh, -CH2-(5-6 membered heteroaryl ring), or a 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur, or, notwithstanding the definition above, two independent occurrences of R°, taken together with their intervening atom(s), form a 3-12-membered saturated, partially unsaturated, or aryl mono- or bicyclic ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur, which may be substituted by a divalent substituent on a saturated carbon atom of R° selected from =0 and =S; or each R° is optionally substituted with a monovalent substituent independently selected from halogen, -(CH2)0-2R*, -(haloR*), -(CH2)0-2OH, -(CH2)0-2OR*, - (CH2)0-2CH(OR*)2; -0(haloR·), -CN, -N3, -(CH2)0-2C(0)R*, -(CH2)0 2C(0)OH, -(CH2)0 2C(0)OR·, -(CH2)0-2SR·, -(CH2)0-2SH, -(CH2)0-2NH2, -(CH2)O 2NHR* , -(CH2)O 2NR*2, -NO2, -SiR*3, -OSiR*3, -C(0)SR* — (C 1 4 straight or branched alkylene)C(0)OR*, or -SSR*.
[0035] Each R* is independently selected from C1-4 aliphatic, -CH2Ph, -0(CH2)o iPh, or a 5-6- membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur, and wherein each R* is unsubstituted or where preceded by halo is substituted only with one or more halogens; or wherein an optional substituent on a saturated carbon is a divalent substituent independently selected from =0, =S, =NNR* 2, =NNHC(0)R*, =NNHC(0)OR*, =NNHS(0)2R*, =NR*, =NOR *, -0(C(R* 2))2 3O-, or - S(C(R* 2))2-3S-, or a divalent substituent bound to vicinal substitutable carbons of an “optionally substituted” group is -0(CR* 2)2-3O-, wherein each independent occurrence of R* is selected from hydrogen, Ci-6 aliphatic or an unsubstituted 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur.
[0036] When R* is C1-6 aliphatic, R* is optionally substituted with halogen, - R*, -(haloR*), -OH, -OR*, -0(haloR*), -CN, -C(0)OH, -C(0)OR*, -NH2, -NHR*, -NR*2, or -N02, wherein each R* is independently selected from Ci 4 aliphatic, -CH2Ph, -0(CH2)o iPh, or a 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur, and wherein each R* is unsubstituted or where preceded by halo is substituted only with one or more halogens.
[0037] An optional substituent on a substitutable nitrogen is independently -R†, -NR† 2, - C(0)R†, -C(0)0R†, -C(0)C(0)R†, -C(0)CH2C(0)R†, -S(0)2R†, -S(0)2NR† 2, -C(S)NR† 2, - C(NH)NR† 2, or -N(R†)S(0)2R†; wherein each R† is independently hydrogen, C1-6 aliphatic, unsubstituted -OPh, or an unsubstituted 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur, or, two independent occurrences of R†, taken together with their intervening atom(s) form an unsubstituted 3-12-membered saturated, partially unsaturated, or aryl mono- or bicyclic ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur; wherein when R† is Ci 6 aliphatic, R† is optionally substituted with halogen, -R*, -(haloR*), -OH, -OR*, - 0(haloR*), -CN, -C(0)OH, -C(0)OR*, -NH2, -NHR*, -NR*2, or -N02, wherein each R* is independently selected from C1-4 aliphatic, -CH2Ph, -0(CH2)0-1Ph, or a 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur, and wherein each R* is unsubstituted or where preceded by halo is substituted only with one or more halogens.
[0038] As used herein, the term "pharmaceutically acceptable salt" refers to those salts which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of humans and lower animals without undue toxicity, irritation, allergic response and the like, and are commensurate with a reasonable benefit/risk ratio. Pharmaceutically acceptable salts are well known in the art. For example, S. M. Berge et al., describe pharmaceutically acceptable salts in detail in J. Pharmaceutical Sciences, 1977, 66, 1-19, incorporated herein by reference. Pharmaceutically acceptable salts of the compounds of this invention include those derived from suitable inorganic and organic acids and bases. Examples of pharmaceutically acceptable, nontoxic acid addition salts are salts of an amino group formed with inorganic acids such as hydrochloric acid, hydrobromic acid, phosphoric acid, sulfuric acid and perchloric acid or with organic acids such as acetic acid, oxalic acid, maleic acid, tartaric acid, citric acid, succinic acid or malonic acid or by using other methods used in the art such as ion exchange. Other pharmaceutically acceptable salts include adipate, alginate, ascorbate, aspartate, benzenesulfonate, benzoate, bisulfate, borate, butyrate, camphorate, camphorsulfonate, citrate, cyclopentanepropionate, digluconate, dodecylsulfate, ethanesulfonate, formate, fumarate, glucoheptonate, glycerophosphate, gluconate, hemisulfate, heptanoate, hexanoate, hydroiodide, 2-hydroxy-ethanesulfonate, lactobionate, lactate, laurate, lauryl sulfate, malate, maleate,
malonate, methanesulfonate, 2-naphthalenesulfonate, nicotinate, nitrate, oleate, oxalate, palmitate, pamoate, pectinate, persulfate, 3-phenylpropionate, phosphate, pivalate, propionate, stearate, succinate, sulfate, tartrate, thiocyanate, p-toluenesulfonate, undecanoate, valerate salts, and the like.
[0039] Further, acids which are generally considered suitable for the formation of pharmaceutically useful salts from basic pharmaceutical compounds are discussed, for example, by P. Stahl et al., Camille G. (eds.) Handbook of Pharmaceutical Salts. Properties, Selection and Use. (2002) Zurich: Wiley-VCH; S. Berge et al., Journal of Pharmaceutical Sciences (1977) 66(1) 1-19; P. Gould, International J. of Pharmaceutics (1986) 33 201-217; Anderson et al, The Practice of Medicinal Chemistry (1996), Academic Press, New York; and in The Orange Book (Food & Drug Administration, Washington, D.C. on their website). These disclosures are incorporated herein by reference.
[0040] Salts derived from appropriate bases include alkali metal, alkaline earth metal, ammonium and N+(Ci 4alkyl)4 salts. Representative alkali or alkaline earth metal salts include sodium, lithium, potassium, calcium, magnesium, and the like. Further pharmaceutically acceptable salts include, when appropriate, nontoxic ammonium, quaternary ammonium, and amine cations formed using counterions such as halide, hydroxide, carboxylate, sulfate, phosphate, nitrate, loweralkyl sulfonate and aryl sulfonate.
[0041] Unless otherwise stated, structures depicted herein are also meant to include all isomeric ( e.g ., enantiomeric, diastereomeric, and geometric (or conformational)) forms of the structure; for example, the R and S configurations for each asymmetric center, Z and E double bond isomers, and Z and E conformational isomers. Therefore, single stereochemical isomers as well as enantiomeric, diastereomeric, and geometric (or conformational) mixtures of the present compounds are within the scope of the invention. Unless otherwise stated, all tautomeric forms of the compounds of the invention are within the scope of the invention. Additionally, unless otherwise stated, structures depicted herein are also meant to include compounds that differ only in the presence of one or more isotopically enriched atoms. For example, compounds having the present structures including the replacement of hydrogen by deuterium or tritium, or the replacement of a carbon by a 13C- or 14C-enriched carbon are within the scope of this invention. Such compounds are useful, for example, as analytical tools, as probes in biological assays, or as therapeutic agents in accordance with the present invention.
[0042] Diastereomeric mixtures can be separated into their individual diastereomers on the basis of their physical chemical differences by methods known to those skilled in the art, such as, for example, by chromatography and/or fractional crystallization. Enantiomers can be separated by converting the enantiomeric mixture into a diastereomeric mixture by reaction with an appropriate optically active compound ( e.g ., chiral auxiliary such as a chiral alcohol or Mosher’s acid chloride), separating the diastereomers and converting (e.g., hydrolyzing) the individual diastereomers to the corresponding pure enantiomers. Alternatively, a particular enantiomer of a compound of the present invention may be prepared by asymmetric synthesis. Still further, where the molecule contains a basic functional group (such as amino) or an acidic functional group (such as carboxylic acid) diastereomeric salts are formed with an appropriate optically- active acid or base, followed by resolution of the diastereomers thus formed by fractional crystallization or chromatographic means known in the art, and subsequent recovery of the pure enantiomers.
[0043] Individual stereoisomers of the compounds of the invention may, for example, be substantially free of other isomers, or may be admixed, for example, as racemates or with all other, or other selected, stereoisomers. Chiral center(s) in a compound of the present invention can have the S or R configuration as defined by the IUPAC 1974 Recommendations. Further, to the extent a compound described herein may exist as an atropisomer (e.g., substituted biaryls), all forms of such atropisomer are considered part of this invention.
[0044] Chemical names, common names, and chemical structures may be used interchangeably to describe the same structure. If a chemical compound is referred to using both a chemical structure and a chemical name, and an ambiguity exists between the structure and the name, the structure predominates. It should also be noted that any carbon as well as heteroatom with unsatisfied valences in the text, schemes, examples and tables herein is assumed to have the sufficient number of hydrogen atom(s) to satisfy the valences.
[0045] The terms “a” and “an” as used herein mean “one or more” and include the plural unless the context is inappropriate.
[0046] The term “alkyl” refers to a saturated straight or branched hydrocarbon, such as a straight or branched group of 1-12, 1-10, or 1-6 carbon atoms, referred to herein as C1-C12 alkyl, C1-C10 alkyl, and C1-C6 alkyl, respectively. Exemplary alkyl groups include, but are not limited to,
methyl, ethyl, propyl, isopropyl, 2-methyl- 1 -propyl, 2-methyl-2-propyl, 2-methyl- 1 -butyl, 3- methyl-1 -butyl, 2-methyl-3-butyl, 2,2-dimethyl- 1 -propyl, 2-methyl- 1 -pentyl, 3 -methyl- 1 -pentyl, 4-methyl- 1 -pentyl, 2-methyl-2-pentyl, 3-methyl-2-pentyl, 4-methyl-2-pentyl, 2,2-dimethyl- 1- butyl, 3, 3 -dimethyl- 1 -butyl, 2-ethyl- 1 -butyl, butyl, isobutyl, t-butyl, pentyl, isopentyl, neopentyl, hexyl, heptyl, octyl, etc.
[0047] The term “cycloalkyl” refers to a monovalent saturated cyclic, bicyclic, or bridged cyclic ( e.g ., adamantyl) hydrocarbon group of 3-12, 3-8, 4-8, or 4-6 carbons, referred to herein, e.g., as “C3-C6 cycloalkyl,” derived from a cycloalkane. Exemplary cycloalkyl groups include cyclohexyl, cyclopentyl, cyclobutyl, and cyclopropyl. The term “cycloalkylene” refers to a bivalent cycloalkyl group.
[0048] The term “haloalkyl” refers to an alkyl group that is substituted with at least one halogen. Exemplary haloalkyl groups include -CH2F, -CHF2, -CF3, -CH2CF3, -CF2CF3, and the like. The term “haloalkylene” refers to a bivalent haloalkyl group.
[0049] The term “hydroxy alkyl” refers to an alkyl group that is substituted with at least one hydroxyl. Exemplary hydroxyalkyl groups include -CH2CH2OH, -C(H)(OH)CH3, -CH2C(H)(OH)CH2CH2OH, and the like.
[0050] The terms “alkenyl” and “alkynyl” are art-recognized and refer to unsaturated aliphatic groups analogous in length and possible substitution to the alkyls described above, but that contain at least one double or triple bond respectively.
[0051] The term “carbocyclylene” refers to a multivalent carbocyclyl group having the appropriate number of open valences to account for groups attached to it. For example, “carbocyclylene” is a bivalent carbocyclyl group when it has two groups attached to it; “carbocyclylene” is a trivalent carbocyclyl group when it has three groups attached to it.
[0052] The terms “alkoxyl” or “alkoxy” are art-recognized and refer to an alkyl group, as defined above, having an oxygen radical attached thereto. Representative alkoxyl groups include methoxy, ethoxy, propyloxy, ieri-butoxy and the like. The term “haloalkoxyl” refers to an alkoxyl group that is substituted with at least one halogen. Exemplary haloalkoxyl groups include -OCH2F, -OCHF2, -OCF3, -OCH2CF3, -OCF2CF3, and the like. The term “hydroxyalkoxyl” refers to an alkoxyl group that is substituted with at least one hydroxyl.
Exemplary hydroxyalkoxyl groups include -OCH2CH2OH, -0CH2C(H)(0H)CH2CH20H, and the like. The term “alkoxylene” refers to a bivalent alkoxyl group.
[0053] The term “oxo” is art-recognized and refers to a “=0” substituent. For example, a cyclopentane susbsituted with an oxo group is cyclopentanone.
[0054] The symbol “ ” indicates a point of attachment.
[0055] When a chemical structure containing a ring is depicted with a substituent having a bond that crosses a ring bond, the substituent may be attached at any available position on the ring.
For example, the chemical structure
encompasses and
. In the context of a polycyclic fused ring, when a chemical structure containing a polycyclic fused ring is depicted with one or more substituent(s) having a bond that crosses multiple rings, the one or more substituent(s) may be independently attached to any of the rings crossed by the bond. To illustrate, the chemical structure
encompasses, for example,
[0056] When any substituent or variable occurs more than one time in any constituent or the compound of the invention, its definition on each occurrence is independent of its definition at every other occurrence, unless otherwise indicated.
[0057] One or more compounds of the invention may exist in unsolvated as well as solvated forms with pharmaceutically acceptable solvents such as water, ethanol, and the like, and it is intended that the invention embrace both solvated and unsolvated forms. “Solvate” means a physical association of a compound of this invention with one or more solvent molecules. This physical association involves varying degrees of ionic and covalent bonding, including hydrogen bonding. In certain instances the solvate will be capable of isolation, for example when one or more solvent molecules are incorporated in the crystal lattice of the crystalline solid. “Solvate”
encompasses both solution-phase and isolatable solvates. Non-limiting examples of suitable solvates include ethanolates, methanolates, and the like. “Hydrate” is a solvate wherein the solvent molecule is H2O.
[0058] As used herein, the terms “subject” and “patient” are used interchangeably and refer to organisms to be treated by the methods of the present invention. Such organisms preferably include, but are not limited to, mammals ( e.g ., murines, simians, equines, bovines, porcines, canines, felines, and the like), and, most preferably, includes humans.
[0059] The term “IC50” is art-recognized and refers to the concentration of a compound that is required to achieve 50% inhibition of the target. The potency of an inhibitor is usually defined by its IC50 value. The lower the IC50 value the greater the potency of the antagonist and the lower the concentration that is required to inhibit the maximum biological response. In certain embodiments, an inhibitor has an IC50 and/or binding constant of less than about 100 mM, less than about 50 mM, less than about 1 mM, less than about 500 nM, less than about 100 nM, less than about 10 nM, or less than about 1 nM.
[0060] As used herein, “K-Ras” refers to a mammalian K-Ras protein. The assignment of amino acid codon and residue positions for human K-Ras is based on the amino acid sequence identified by UniProtKB/Swiss-Prot P01116.
[0061] As used herein, “K-Ras G12D” refers to a mutant form of a mammalian K-Ras protein that contains an amino acid substitution of an aspartic acid, instead of a glycine, at amino acid position 12. The assignment of amino acid codon and residue positions for human K-Ras is based on the amino acid sequence identified by UniProtKB/Swiss-Prot P01116:
V ariantp . Gly 12 Asp .
[0062] As used herein, the terms “K-Ras inhibitor” or “K-Ras antagonist” are defined as a compound that binds to and/or inhibits K-Ras with measurable affinity. In some embodiments, inhibition in the presence of the inhibitor is observed in a dose-dependent manner. In some embodiments, the measured signal (e.g., signaling activity or biological activity) is at least about 5%, at least about 10%, at least about 15%, at least about 20%, at least about 25%, at least about 30%, at least about 35%, at least about 40%, at least about 45%, at least about 50%, at least about 55%, at least about 60%, at least about 65%, at least about 70%, at least about 75%, at least about 80%, at least about 85%, at least about 90%, at least about 95%, at least about 96%,
at least about 97%, at least about 98%, at least about 99%, or at least about 100% lower than the signal measured with a negative control under comparable conditions.
[0063] As used herein, the terms “inhibitor” or “K-Ras G12D inhibitor” or “K-Ras G12D antagonist” are defined as a compound that binds to and/or inhibits K-Ras G12D with measurable affinity. In some embodiments, inhibition in the presence of the inhibitor is observed in a dose-dependent manner. In some embodiments, the measured signal ( e.g ., signaling activity or biological activity) is at least about 5%, at least about 10%, at least about 15%, at least about 20%, at least about 25%, at least about 30%, at least about 35%, at least about 40%, at least about 45%, at least about 50%, at least about 55%, at least about 60%, at least about 65%, at least about 70%, at least about 75%, at least about 80%, at least about 85%, at least about 90%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, at least about 99%, or at least about 100% lower than the signal measured with a negative control under comparable conditions.
[0064] The terms “measurable affinity” and “measurably inhibit,” as used herein, means a measurable change or inhibition in K-Ras activity, such as K-Ras G12D activity, between a sample comprising a compound of the present invention, or composition thereof, and K-Ras, such as K-Ras G12D, and an equivalent sample comprising K-Ras, such as K-Ras G12D, in the absence of said compound, or composition thereof.
[0065] As used herein, the term “effective amount” refers to the amount of a compound sufficient to effect beneficial or desired results (e.g., a therapeutic, ameliorative, inhibitory, or preventative result). An effective amount can be administered in one or more administrations, applications, or dosages and is not intended to be limited to a particular formulation or administration route.
[0066] As used herein, the term “treating” includes any effect, e.g., lessening, reducing, modulating, ameliorating or eliminating, that results in the improvement of the condition, disease, disorder, and the like, or ameliorating a symptom thereof. In some embodiments, treatment can be administered after one or more symptoms have developed. In other embodiments, treatment can be administered in the absence of symptoms. For example, treatment can be administered to a susceptible individual prior to the onset of symptoms (e.g., in light of a history of symptoms and/or in light of genetic or other susceptibility factors).
Treatment can also be continued after symptoms have resolved, for example, to prevent or delay their recurrence.
[0067] The phrases “disorder mediated by K-Ras” or “disease mediated by K-Ras” or "K-Ras- associated disease or disorder," as used herein, means any disease or other deleterious condition in which K-Ras, or a variant or mutant thereof, is known to play a role. A non-limiting example of a K-Ras-associated disease or disorder is a K-Ras-associated cancer.
[0068] The phrases “disorder mediated by K-Ras G12D” or “disease mediated by K-Ras G12D” or "K-Ras G12D-associated disease or disorder," as used herein, refer to diseases or disorders associated with, or mediated by, or having a K-Ras G12D mutation. A non-limiting example of a K-Ras G12D-associated disease or disorder is a K-Ras G12D-associated cancer.
[0069] As used herein, the term “pharmaceutical composition” refers to the combination of an active agent with a carrier, inert or active, making the composition especially suitable for diagnostic or therapeutic use in vivo or ex vivo.
[0070] As used herein, the term “pharmaceutically acceptable carrier” refers to any of the standard pharmaceutical carriers, such as a phosphate buffered saline solution, water, emulsions ( e.g ., such as an oil/water or water/oil emulsions), and various types of wetting agents. The compositions also can include stabilizers and preservatives. For examples of carriers, stabilizers and adjuvants, see e.g., Martin, Remington’s Pharmaceutical Sciences, 15th Ed., Mack Publ. Co., Easton, PA [1975].
[0071] For therapeutic use, salts of the compounds of the present invention are contemplated as being pharmaceutically acceptable. However, salts of acids and bases that are non- pharmaceutically acceptable may also find use, for example, in the preparation or purification of a pharmaceutically acceptable compound.
[0072] In addition, when a compound of the invention contains both a basic moiety (such as, but not limited to, a pyridine or imidazole) and an acidic moiety (such as, but not limited to, a carboxylic acid) zwitterions (“inner salts”) may be formed. Such acidic and basic salts used within the scope of the invention are pharmaceutically acceptable (i.e., non-toxic, physiologically acceptable) salts. Such salts of the compounds of the invention may be formed, for example, by reacting a compound of the invention with an amount of acid or base, such as an
equivalent amount, in a medium such as one in which the salt precipitates or in an aqueous medium followed by lyophilization.
[0073] Throughout the description, where compositions are described as having, including, or comprising specific components, or where processes and methods are described as having, including, or comprising specific steps, it is contemplated that, additionally, there are compositions of the present invention that consist essentially of, or consist of, the recited components, and that there are processes and methods according to the present invention that consist essentially of, or consist of, the recited processing steps.
[0074] As a general matter, compositions specifying a percentage are by weight unless otherwise specified.
I. Naphthyl-substituted Quinolin-4(1H)-ones and Related Compounds
[0075] The invention provides naphthyl-substituted quinolin-4( 1H)-oncs and related compounds. The compounds may be used in the pharmaceutical compositions and therapeutic methods described herein. Exemplary compounds are described in the following sections, along with exemplary procedures for making the compounds.
[0076] One aspect of the invention provides a compound represented by Formula I:
or a pharmaceutically acceptable salt thereof; wherein:
R1 is naphthyl; an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3
heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with m occurrences of R5;
R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci-6 aliphatic; each of which is substituted with n occurrences of R6;
R3 is hydrogen; or one occurrence of R3 is hydrogen, and the other occurrence of R3 is taken together with R4 to form a double bond between the atoms to which R3 and R4 are attached; or both occurrences of R3 are taken together to form an oxo substituent;
R4 is taken together with one occurrence of R3 to form a double bond between the atoms to which R3 and R4 are attached;
R5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, Ci-6 alkyl, hydroxyl, Ci-6 alkoxyl, halo, or cyano;
R6 and R7 each represent independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, Ci-6 alkyl, Ci-6 haloalkyl, Ci-6 hydroxyalkyl, or -N(R9)2;
R8 and R9 each represent independently for each occurrence hydrogen, CM alkyl, or C3-5 cycloalkyl; or two occurrences of R9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom;
A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p
occurrences
X1 is C(H)2, C(R4)(H), or N(R4);
X2 is N or C(CN);
X3 is N or C(H);
X4 is C(O) or C(H)2;
L is a covalent bond or a C1-4 bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or -C(O)-;
Y1, Y2, and Y3 represent independently a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R7)-, or -C(R7)2-; and m, n, and p are each independently 0, 1, 2, or 3.
[0077] The definitions of variables in Formula I above encompass multiple chemical groups.
The application contemplates embodiments where, for example, i) the definition of a variable is a single chemical group selected from those chemical groups set forth above, ii) the definition of a variable is a collection of two or more of the chemical groups selected from those set forth above, and iii) the compound is defined by a combination of variables in which the variables are defined by (i) or (ii).
[0078] In certain embodiments, the compound is a compound of Formula I.
[0079] As defined generally above, R1 is naphthyl; an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with m occurrences of R5.
[0080] In certain embodiments, R1 is naphthyl, an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur, or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R5. In certain embodiments, R1 is phenyl or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R5.
[0081] In certain embodiments, R1 is naphthyl or phenyl; each of which is substituted with m occurrences of R5. In certain embodiments, R1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R5.
[0082] In certain embodiments, R1 is naphthyl or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R5. In certain embodiments, R1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur, or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R5.
[0083] In certain embodiments, R1 is naphthyl substituted with m occurrences of R5. In certain embodiments, R1 is 1-naphthyl substituted with m occurrences of R5. In certain embodiments,
[0084] In certain embodiments, R1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with m occurrences of R5. In certain
embodiments, R1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with m occurrences of R5.
[0086] In certain embodiments, R1 is an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R5. In certain embodiments, R1 is an 8-10 membered bicyclic heteroaryl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R5.
[0087] In certain embodiments, R1 is quinolinyl, isoquinolinyl, or indazolyl; each of which is substituted with m occurrences of R5. In certain embodiments, R1 is quinolinyl substituted with m occurrences of R5. In certain embodiments, R1 is quinoline-4-yl substituted with m occurrences of R5. In certain embodiments, R1 is isoquinolinyl substituted with m occurrences of R5. In certain embodiments, R1 is isoquinolin-l-yl substituted with m occurrences of R5. In certain embodiments, R1 is indazolyl substituted with m occurrences of R5.
[0088] In certain embodiments, R1 is phenyl substituted with m occurrences of R5. In certain embodiments, R1 is
In certain embodiments, R1 is
[0089] In certain embodiments, R1 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R5. In certain embodiments, R1 is a 6-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with m occurrences of R5. In certain embodiments, R1 is a 5-membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R5.
[0090] In certain embodiments, R1 is selected from the groups depicted in the compounds in Table 1, below. In certain embodiments, R1 is selected from the groups depicted in the compounds in Table 1 or 1-A, below.
[0091] As defined generally above, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci-6 aliphatic; each of which is substituted with n occurrences of R6.
[0092] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R6. In certain embodiments, R2 is phenyl or Ci-6 aliphatic; each of which is substituted with n occurrences of R6.
[0093] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R6. In certain embodiments, R2 is a 3-7
membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R6.
[0094] In certain embodiments, R2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci-6 aliphatic; each of which is substituted with n occurrences of R6. In certain embodiments, R2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or Ci-6 aliphatic; each of which is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with n occurrences of R6.
[0095] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is an 8-10 membered saturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is an 8-10 membered partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6.
[0096] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 nitrogen atom; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is an 8-10 membered saturated bicyclic heterocyclyl having 1 nitrogen
atom; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments,
[0097] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur. In certain embodiments, R2 is an 8-10 membered saturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur. In certain embodiments, R2 is an 8-10 membered partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur.
[0098] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen. In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 nitrogen atom. In certain embodiments, R2 is an 8-10 membered saturated bicyclic heterocyclyl having 1 nitrogen atom. In certain embodiments, R2 is
[0099] In certain embodiments, R2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 4-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 6-membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6.
[0100] In certain embodiments, R2 is a 3-7 membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 3-7 membered partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6 membered partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6.
[0101] In certain embodiments, R2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R6.
[0102] In certain embodiments, R2 is azetidinyl, pyrrolidinyl, piperidinyl, or morpholinyl; each of which is substituted with n occurrences of R6. In certain embodiments, R2 is azetidinyl substituted with n occurrences of R6. In certain embodiments, R2 is pyrrolidinyl substituted with n occurrences of R6. In certain embodiments, R2 is pyrrolidin-2-yl substituted with n occurrences of R6. In certain embodiments, R2 is pyrrolidin-2-yl substituted with a single occurrence of R6 at the 1-position. In certain embodiments, R2 is piperidinyl substituted with n occurrences of R6. In certain embodiments, R2 is morpholinyl substituted with n occurrences of R6.
[0103] In certain embodiments, R2 is azetidinyl, pyrrolidinyl, piperidinyl, or morpholinyl. In certain embodiments, R2 is azetidinyl. In certain embodiments, R2 is pyrrolidinyl. In certain
embodiments, R2 is pyrrolidin-2-yl. In certain embodiments, R2 is l-methyl-pyrrolidin-2-yl. In certain embodiments, R2 is piperidinyl. In certain embodiments, R2 is morpholinyl.
[0104] In certain embodiments, R2 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with n occurrences of R6.
[0105] In certain embodiments, R2 is phenyl substituted with n occurrences of R6. In certain embodiments, R2 is phenyl.
[0106] In certain embodiments, R2 is Ci-6 aliphatic substituted with n occurrences of R6. In certain embodiments, R2 is C3-6 cycloaliphatic substituted with n occurrences of R6. In certain embodiments, R2 is a Ci-6 aliphatic chain substituted with n occurrences of R6. In certain embodiments, R2 is C3-6 cycloalkyl substituted with n occurrences of R6. In certain embodiments, R2 is Ci-6 alkyl substituted with n occurrences of R6.
[0107] In certain embodiments, R2 is Ci-6 aliphatic. In certain embodiments, R2 is C3-6 cycloaliphatic. In certain embodiments, R2 is a Ci-6 aliphatic chain. In certain embodiments, R2 is C3-6 cycloalkyl. In certain embodiments, R2 is Ci-6 alkyl.
[0108] In certain embodiments, R2 is selected from the groups depicted in the compounds in Table 1, below. In certain embodiments, R2 is selected from the groups depicted in the compounds in Table 1 or 1-A, below.
[0109] As defined generally above, R3 is hydrogen; or one occurrence of R3 is hydrogen, and the other occurrence of R3 is taken together with R4 to form a double bond between the atoms to which R3 and R4 are attached; or both occurrences of R3 are taken together to form an oxo substituent. In certain embodiments, R3 is hydrogen. In certain embodiments, one occurrence of R3 is hydrogen, and the other occurrence of R3 is taken together with R4 to form a double bond between the atoms to which R3 and R4 are attached. In certain embodiments, both occurrences of R3 are taken together to form an oxo substituent. In certain embodiments, R3 is selected from the groups depicted in the compounds in Table 1, below.
[0110] As defined generally above, R4 is taken together with one occurrence of R3 to form a double bond between the atoms to which R3 and R4 are attached. In certain embodiments, R4 is selected from the groups depicted in the compounds in Table 1, below.
[0111] As defined generally above, R5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, Ci-6 alkyl, hydroxyl, Ci-6 alkoxyl, halo, or cyano.
[0112] In certain embodiments, R5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, Ci-6 alkyl, or hydroxyl. In certain embodiments, R5 represents independently for each occurrence ethynyl, ethenyl, C1-3 alkyl, or hydroxyl. In certain embodiments, R5 represents independently for each occurrence C2-6 alkynyl or hydroxyl. In certain embodiments, R5 represents independently for each occurrence ethynyl or hydroxyl. In certain embodiments, one occurrence of R5 is ethynyl. In certain embodiments, one occurrence of R5 is hydroxyl.
[0113] In certain embodiments, R5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, or Ci-6 alkyl. In certain embodiments, R5 represents independently for each occurrence ethynyl, ethenyl, or C1-3 alkyl. In certain embodiments, R5 represents independently for each occurrence hydroxyl or Ci-6 alkoxyl. In certain embodiments, R5 represents independently for each occurrence halo or cyano.
[0114] In certain embodiments, R5 represents independently for each occurrence C2-6 alkynyl. In certain embodiments, R5 is ethynyl. In certain embodiments, R5 represents independently for each occurrence C2-6 alkenyl. In certain embodiments, R5 is ethenyl. In certain embodiments, R5 represents independently for each occurrence Ci-6 alkyl. In certain embodiments, R5 represents independently for each occurrence C1-3 alkyl. In certain embodiments, R5 is methyl. In certain embodiments, R5 is hydroxyl. In certain embodiments, R5 represents independently for each occurrence Ci-6 alkoxyl. In certain embodiments, R5 represents independently for each occurrence halo. In certain embodiments, R5 represents independently for each occurrence fluoro or chloro. In certain embodiments, R5 is fluoro. In certain embodiments, R5 is chloro. In certain embodiments, R5 is cyano. In certain embodiments, R5 is selected from the groups depicted in the compounds in Table 1, below. In certain embodiments, R5 is selected from the groups depicted in the compounds in Table 1 or 1-A, below.
[0115] As defined generally above, R6 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, Ci-6 alkyl, Ci-6 haloalkyl, Ci-6 hydroxyalkyl, or -N(R9)2.
[0116] In certain embodiments, R6 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 alkyl, or Ci-6 haloalkyl. In certain embodiments, R6 represents independently for each occurrence halo, Ci-6 alkyl, or Ci-6 haloalkyl. In certain embodiments, R6 represents
independently for each occurrence halo or Ci-6 alkyl. In certain embodiments, R6 represents independently for each occurrence halo, hydroxyl, or Ci-6 alkyl.
[0117] In certain embodiments, R6 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, or -N(R9)2. In certain embodiments, R6 represents independently for each occurrence halo, hydroxyl, or -N(R9)2. In certain embodiments, R6 represents independently for each occurrence halo, Ci-6 alkoxyl, or Ci-6 haloalkoxyl. In certain embodiments, R6 represents independently for each occurrence halo, Ci-6 alkyl, Ci-6 haloalkyl, or Ci-6 hydroxyalkyl. In certain embodiments, R6 represents independently for each occurrence Ci-6 alkyl, Ci-6 haloalkyl, or Ci-6 hydroxyalkyl. In certain embodiments, R6 represents independently for each occurrence Ci-6 alkyl or Ci-6 haloalkyl.
[0118] In certain embodiments, R6 represents independently for each occurrence halo. In certain embodiments, R6 represents independently for each occurrence fluoro or chloro. In certain embodiments, R6 is fluoro. In certain embodiments, R6 is chloro. In certain embodiments, R6 is hydroxyl. In certain embodiments, R6 represents independently for each occurrence Ci-6 alkoxyl. In certain embodiments, R6 is methoxy. In certain embodiments, R6 represents independently for each occurrence Ci-6 haloalkoxyl. In certain embodiments, R6 is trifluoromethoxy. In certain embodiments, R6 represents independently for each occurrence Ci-6 alkyl. In certain embodiments, R6 represents independently for each occurrence C1-3 alkyl. In certain embodiments, R6 is methyl. In certain embodiments, R6 represents independently for each occurrence Ci-6 haloalkyl. In certain embodiments, R6 is trifluoromethyl. In certain embodiments, R6 represents independently for each occurrence Ci-6 hydroxyalkyl. In certain embodiments, R6 represents independently for each occurrence -N(R9)2. In certain embodiments, R6 is -NH2. In certain embodiments, R6 is selected from the groups depicted in the compounds in Table 1, below. In certain embodiments, R6 is selected from the groups depicted in the compounds in Table 1 or 1-A, below.
[0119] As defined generally above, R7 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, Ci-6 alkyl, Ci-6 haloalkyl, Ci-6 hydroxyalkyl, or -N(R9)2.
[0120] In certain embodiments, R7 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 alkyl, or Ci-6 haloalkyl. In certain embodiments, R7 represents independently for each occurrence halo, Ci-6 alkyl, or Ci-6 haloalkyl. In certain embodiments, R7 represents
independently for each occurrence halo or Ci-6 alkyl. In certain embodiments, R7 represents independently for each occurrence halo, hydroxyl, or Ci-6 alkyl.
[0121] In certain embodiments, R7 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, or -N(R9)2. In certain embodiments, R7 represents independently for each occurrence halo, hydroxyl, or -N(R9)2. In certain embodiments, R7 represents independently for each occurrence halo, Ci-6 alkoxyl, or Ci-6 haloalkoxyl. In certain embodiments, R7 represents independently for each occurrence halo, Ci-6 alkyl, Ci-6 haloalkyl, or Ci-6 hydroxyalkyl. In certain embodiments, R7 represents independently for each occurrence Ci-6 alkyl, Ci-6 haloalkyl, or Ci-6 hydroxyalkyl. In certain embodiments, R7 represents independently for each occurrence Ci-6 alkyl or Ci-6 haloalkyl.
[0122] In certain embodiments, R7 represents independently for each occurrence halo. In certain embodiments, R7 represents independently for each occurrence fluoro or chloro. In certain embodiments, R7 is fluoro. In certain embodiments, R7 is chloro. In certain embodiments, R7 is hydroxyl. In certain embodiments, R7 represents independently for each occurrence Ci-6 alkoxyl. In certain embodiments, R7 is methoxy. In certain embodiments, R7 represents independently for each occurrence Ci-6 haloalkoxyl. In certain embodiments, R7 is trifluoromethoxy. In certain embodiments, R7 represents independently for each occurrence C1-6 alkyl. In certain embodiments, R7 represents independently for each occurrence C1-3 alkyl. In certain embodiments, R7 is methyl. In certain embodiments, R7 represents independently for each occurrence Ci-6 haloalkyl. In certain embodiments, R7 is trifluoromethyl. In certain embodiments, R7 represents independently for each occurrence Ci-6 hydroxyalkyl. In certain embodiments, R7 represents independently for each occurrence -N(R9)2. In certain embodiments, R7 is -NH2. In certain embodiments, R7 is selected from the groups depicted in the compounds in Table 1, below.
[0123] As defined generally above, R8 is hydrogen, C1-4 alkyl, or C3-5 cycloalkyl. In certain embodiments, R8 is hydrogen or C 1-4 alkyl. In certain embodiments, R8 is hydrogen or methyl.
In certain embodiments, R8 is CIM alkyl or C3-5 cycloalkyl.
[0124] In certain embodiments, R8 is hydrogen. In certain embodiments, R8 is CIM alkyl. In certain embodiments, R8 is methyl. In certain embodiments, R8 is C3-5 cycloalkyl. In certain
embodiments, R8 is cyclopropyl. In certain embodiments, R8 is selected from the groups depicted in the compounds in Table 1, below.
[0125] As defined generally above, R9 represents independently for each occurrence hydrogen, Ci-4 alkyl, or C3-5 cycloalkyl; or two occurrences of R9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom.
[0126] In certain embodiments, R9 represents independently for each occurrence hydrogen, C1-4 alkyl, or C3-5 cycloalkyl. In certain embodiments, R9 represents independently for each occurrence hydrogen or C1-4 alkyl. In certain embodiments, R9 represents independently for each occurrence hydrogen or methyl. In certain embodiments, R9 represents independently for each occurrence C1-4 alkyl or C3-5 cycloalkyl.
[0127] In certain embodiments, two occurrences of R9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom.
[0128] In certain embodiments, R9 is hydrogen. In certain embodiments, R9 represents independently for each occurrence CM alkyl. In certain embodiments, R9 is methyl. In certain embodiments, R9 represents independently for each occurrence C3-5 cycloalkyl. In certain embodiments, R9 is cyclopropyl. In certain embodiments, R9 is selected from the groups depicted in the compounds in Table 1, below.
[0129] As defined generally above, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R7; or A1 is
,
[0130] In certain embodiments, A1 is
In certain embodiments, A1 is
In certain embodiments, A1 is In certain embodiments, A1 is
[0131] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5- 6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R7.
[0132] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R7; or A1 is
[0133] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R7.
[0134] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2
nitrogen atoms; each of which is substituted with p occurrences of R7; or A1 is
or
[0135] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R7.
[0136] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R7.
[0137] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5- 6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur.
[0138] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or
[0139] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
[0140] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2
[0141] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
[0142] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur.
[0143] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7. In certain embodiments, A1 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7. In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A1 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms.
[0144] In certain embodiments, A1 is 1,4-diazepanyl, piperidinyl, piperazinyl, pyrrolidinyl, or azetidinyl; each of which is substituted with p occurrences of R7. In certain embodiments, A1 is 1,4-diazepanyl substituted with p occurrences of R7. In certain embodiments, A1 is piperidinyl substituted with p occurrences of R7. In certain embodiments, A1 is piperazinyl substituted with p occurrences of R7. In certain embodiments, A1 is pyrrolidinyl substituted with p occurrences of R7. In certain embodiments, A1 is azetidinyl substituted with p occurrences of R7.
[0145] In certain embodiments, A1 is 1,4-diazepanyl, piperidinyl, piperazinyl, pyrrolidinyl, or azetidinyl. In certain embodiments, A1 is 1,4-diazepanyl. In certain embodiments, A1 is piperidinyl. In certain embodiments, A1 is piperazinyl. In certain embodiments, A1 is pyrrolidinyl. In certain embodiments, A1 is azetidinyl.
[0146] In certain embodiments, A1 is 1,4-diazepan-l-yl, piperidin-4-yl, piperazin-l-yl, pyrrolidin-3-yl, or azetidin-3-yl; each of which is substituted with p occurrences of R7. In certain embodiments, A1 is 1,4-diazepan-l-yl substituted with p occurrences of R7. In certain embodiments, A1 is piperidin-4-yl substituted with p occurrences of R7. In certain embodiments, A1 is piperazin-l-yl substituted with p occurrences of R7. In certain embodiments, A1 is pyrrolidin-3-yl substituted with p occurrences of R7. In certain embodiments, A1 is azetidin-3-yl substituted with p occurrences of R7.
[0147] In certain embodiments, A1 is 1,4-diazepan-l-yl, piperidin-4-yl, piperazin-l-yl, pyrrolidin-3-yl, or azetidin-3-yl. In certain embodiments, A1 is 1,4-diazepan-l-yl. In certain embodiments, A1 is piperidin-4-yl. In certain embodiments, A1 is piperazin-l-yl. In certain embodiments, A1 is pyrrolidin-3-yl. In certain embodiments, A1 is azetidin-3-yl.
[0148] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7. In certain embodiments, A1 is a 7-10 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7. In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7.
[0149] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A1 is a 7-10 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 2 nitrogen atoms. In certain embodiments, A1 is
or
In certain embodiments, A1 is
In certain embodiments, A1 i is
[0150] In certain embodiments, A1 is a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7.
In certain embodiments, A1 is a 7-10 membered saturated ori/zo-fused heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7. In certain embodiments, A1 is a 6-11 membered saturated ori/zo-fused heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A1 is a 7-10 membered saturated ori zo-fused heterocyclyl having 1 or 2 nitrogen atoms.
[0151] In certain embodiments, A1 is a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with p occurrences of R7.
[0152] In certain embodiments, A1 is a 6-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with p occurrences of R7. In certain embodiments, A1 is pyridinyl or pyrimidinyl; each of which is substituted with p occurrences of R7. In certain embodiments, A1 is pyridinyl substituted with p occurrences of R7. In certain embodiments, A1 is pyridin-4-yl substituted with p occurrences of R7. In certain embodiments, A1 is pyrimidinyl substituted with p occurrences of R7.
[0153] In certain embodiments, A1 is a 5-membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with p occurrences of R7. In certain embodiments, A1 is a 5-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with p occurrences of R7. In certain embodiments, A1 is imidazolyl or pyrazolyl; each of which is substituted with p occurrences of R7. In certain embodiments, A1 is imidazolyl substituted with p occurrences of R7. In certain embodiments, A1 is pyrazolyl substituted with p occurrences of R7.
[0154] In certain embodiments, A1 is selected from the groups depicted in the compounds in Table 1, below.
[0155] As defined generally above, X1 is C(H)2, C(R4)(H), or N(R4). In certain embodiments,
X1 is C(H)2 or C(R4)(H). In certain embodiments, X1 is C(R4)(H) or N(R4). In certain embodiments, X1 is C(H)2. In certain embodiments, X1 is C(R4)(H). In certain embodiments, X1 is N(R4). In certain embodiments, X1 is selected from the groups depicted in the compounds in Table 1, below.
[0156] As defined generally above, X2 is N or C(CN). In certain embodiments, X2 is N. In certain embodiments, X2 is C(CN). In certain embodiments, X2 is selected from the groups depicted in the compounds in Table 1, below.
[0157] As defined generally above, X3 is N or C(H). In certain embodiments, X3 is N. In certain embodiments, X3 is C(H). In certain embodiments, X3 is selected from the groups depicted in the compounds in Table 1, below.
[0158] As defined generally above, X4 is C(O) or C(H)2. In certain embodiments, X4 is C(O).
In certain embodiments, X4 is C(H)2. In certain embodiments, X4 is selected from the groups depicted in the compounds in Table 1, below.
[0159] As defined generally above, L is a covalent bond or a Ci-4 bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or -C(O)-. In certain embodiments, L is a covalent bond.
[0160] In certain embodiments, L is a Ci-4 bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or -C(O)-. In certain embodiments, L is a Ci-4 bivalent saturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or -C(O)-. In certain embodiments, L is a C1-4 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or - C(O)-. In certain embodiments, L is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -N(R9)-, or -C(O)-.
[0161] In certain embodiments, L is a C1-4 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -N(R9)-, or -C(O)-. In certain embodiments, L is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -N(R9)-, or -C(O)-. In certain embodiments, L is a Ci-4 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -O- or -N(R9)-. In certain embodiments, L is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -O- or -N(R9)-.
[0162] In certain embodiments, L is -0-CH2-, -0-(CH2)2-, -N(R9)-CH2-, or -N(R9)-(CH2)2-, wherein the terminal carbon atom of L is attached to R2. In certain embodiments, L is -0-CH2- or -0-(CH2)2-, wherein the terminal carbon atom of L is attached to R2. In certain embodiments, L is -N(R9)-CH2- or -N(R9)-(CH2)2-, wherein the terminal carbon atom of L is attached to R2. In certain embodiments, L is -0-CH2-, wherein the carbon atom of L is attached to R2. In certain embodiments, L is -0-(CH2)2-, wherein the terminal carbon atom of L is attached to R2. In certain embodiments, L is -N(R9)-CH2-, wherein the carbon atom of L is attached to R2. In certain embodiments, L is -N(R9)-(CH2)2-, wherein the terminal carbon atom of L is attached to R2.
[0163] In certain embodiments, L is selected from the groups depicted in the compounds in Table 1, below.
[0164] As defined generally above, Y1 is a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R7)-, or -C(R7)2-.
[0165] In certain embodiments, Y1 is a C2-3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y1 is a C2-3 bivalent unsaturated, straight hydrocarbon chain.
[0166] In certain embodiments, Y1 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y1 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y1 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y1 is a C1-3 bivalent saturated, straight hydrocarbon chain.
[0167] In certain embodiments, Y1 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y1 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y1 is a C2-3 bivalent saturated, straight
hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y1 is a C2-3 bivalent saturated, straight hydrocarbon chain.
[0168] In certain embodiments, Y1 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, -(CH2)3-, -CH2-0- CH2-, or -CH2-CF2-CH2-. In certain embodiments, Y1 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, - (CH2)3-, or -CH2-CF2-CH2-. In certain embodiments, Y1 is -(CH2)2-, -CH2-C(H)(OH)-, -(CH2)3-, or -CH2-CF2-CH2-. In certain embodiments, Y1 is -(CH2)2- or -(CH2)3-. In certain embodiments, Y1 is -CH2-C(H)(OH)- or -CH2-CF2-CH2-. In certain embodiments, Y1 is -(CH2)-, -(CH2)2-, or - (CH2)3-. In certain embodiments, Y1 is -(CH2)2- or -CH2-C(H)(OH)-. In certain embodiments, Y1 is -(CH2)3-, -CH2-0-CH2-, or -CH2-CF2-CH2-.
[0169] In certain embodiments, Y1 is -(CH2)-. In certain embodiments, Y1 is -(CH2)2-. In certain embodiments, Y1 is -CH2-C(H)(OH)-. In certain embodiments, Y1 is -(CH2)3-. In certain embodiments, Y1 is -CH2-0-CH2-. In certain embodiments, Y1 is -CH2-CF2-CH2-. In certain embodiments, Y1 is selected from the groups depicted in the compounds in Table 1, below.
[0170] As defined generally above, Y2 is a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R7)-, or -C(R7)2-.
[0171] In certain embodiments, Y2 is a C2-3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -C(H)(R7)-, or -C(R7)2-.
In certain embodiments, Y2 is a C2-3 bivalent unsaturated, straight hydrocarbon chain.
[0172] In certain embodiments, Y2 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y2 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y2 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y2 is a C1-3 bivalent saturated, straight hydrocarbon chain.
[0173] In certain embodiments, Y2 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y2 is a C2-3 bivalent saturated, straight
hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y2 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y2 is a C2-3 bivalent saturated, straight hydrocarbon chain.
[0174] In certain embodiments, Y2 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, -(CH2)3-, -CH2-0- CH2-, or -CH2-CF2-CH2-. In certain embodiments, Y2 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, - (CH2)3-, or -CH2-CF2-CH2-. In certain embodiments, Y2 is -(CH2)2-, -CH2-C(H)(OH)-, -(CH2)3-, or -CH2-CF2-CH2-. In certain embodiments, Y2 is -(CH2)2- or -(CH2)3-. In certain embodiments, Y2 is -CH2-C(H)(OH)- or -CH2-CF2-CH2-. In certain embodiments, Y2 is -(CH2)-, -(CH2)2-, or - (CH2)3-. In certain embodiments, Y2 is -(CH2)2- or -CH2-C(H)(OH)-. In certain embodiments, Y2 is -(CH2)3-, -CH2-0-CH2-, or -CH2-CF2-CH2-.
[0175] In certain embodiments, Y2 is -(CH2)-. In certain embodiments, Y2 is -(CH2)2-. In certain embodiments, Y2 is -CH2-C(H)(OH)-. In certain embodiments, Y2 is -(CH2)3-. In certain embodiments, Y2 is -CH2-0-CH2-. In certain embodiments, Y2 is -CH2-CF2-CH2-. In certain embodiments, Y2 is selected from the groups depicted in the compounds in Table 1, below.
[0176] As defined generally above, Y3 is a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R7)-, or -C(R7)2-.
[0177] In certain embodiments, Y3 is a C2-3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -C(H)(R7)-, or -C(R7)2-.
In certain embodiments, Y3 is a C2-3 bivalent unsaturated, straight hydrocarbon chain.
[0178] In certain embodiments, Y3 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y3 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y3 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y3 is a C1-3 bivalent saturated, straight hydrocarbon chain.
[0179] In certain embodiments, Y3 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y3 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y3 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y3 is a C2-3 bivalent saturated, straight hydrocarbon chain.
[0180] In certain embodiments, Y3 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, -(CH2)3-, -CH2-O- CH2-, or -CH2-CF2-CH2-. In certain embodiments, Y3 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, - (CH2)3-, or -CH2-CF2-CH2-. In certain embodiments, Y3 is -(CH2)2-, -CH2-C(H)(OH)-, -(CH2)3-, or -CH2-CF2-CH2-. In certain embodiments, Y3 is -(CH2)2- or -(CH2)3-. In certain embodiments, Y3 is -CH2-C(H)(OH)- or -CH2-CF2-CH2-. In certain embodiments, Y3 is -(CH2)-, -(CH2)2-, or - (CH2)3-. In certain embodiments, Y3 is -(CH2)2- or -CH2-C(H)(OH)-. In certain embodiments, Y3 is -(CH2)3-, -CH2-O-CH2-, or -CH2-CF2-CH2-.
[0181] In certain embodiments, Y3 is -(CH2)-. In certain embodiments, Y3 is -(CH2)2-. In certain embodiments, Y3 is -CH2-C(H)(OH)-. In certain embodiments, Y3 is -(CH2)3-. In certain embodiments, Y3 is -CH2-O-CH2-. In certain embodiments, Y3 is -CH2-CF2-CH2-. In certain embodiments, Y3 is selected from the groups depicted in the compounds in Table 1, below.
[0182] As defined generally above, m is 0, 1, 2, or 3. In certain embodiments, m is 0. In certain embodiments, m is 1. In certain embodiments, m is 2. In certain embodiments, m is 3. In certain embodiments, m is 0 or 1. In certain embodiments, m is 1 or 2. In certain embodiments, m is 2 or 3. In certain embodiments, m is 0, 1, or 2. In certain embodiments, m is 1, 2, or 3. In certain embodiments, m is selected from the values represented in the compounds in Table 1, below.
[0183] As defined generally above, n is 0, 1, 2, or 3. In certain embodiments, n is 0. In certain embodiments, n is 1. In certain embodiments, n is 2. In certain embodiments, n is 3. In certain embodiments, n is 0 or 1. In certain embodiments, n is 1 or 2. In certain embodiments, n is 2 or 3. In certain embodiments, n is 0, 1, or 2. In certain embodiments, n is 1, 2, or 3. In certain embodiments, n is selected from the values represented in the compounds in Table 1, below.
[0184] As defined generally above, p is 0, 1, 2, or 3. In certain embodiments, p is 0. In certain embodiments, p is 1. In certain embodiments, p is 2. In certain embodiments, p is 3. In certain embodiments, p is 0 or 1. In certain embodiments, p is 1 or 2. In certain embodiments, p is 2 or 3. In certain embodiments, p is 0, 1, or 2. In certain embodiments, p is 1, 2, or 3. In certain embodiments, p is selected from the values represented in the compounds in Table 1, below.
[0185] The description above describes multiple embodiments relating to compounds of Formula I. The patent application specifically contemplates all combinations of the embodiments.
[0186] Another aspect of the invention provides a compound represented by Formula II:
or a pharmaceutically acceptable salt thereof; wherein:
R1 is naphthyl; an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with m occurrences of R5;
R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci-6 aliphatic; each of which is substituted with n occurrences of R6;
R3 is CM alkyl;
R4 is hydrogen, halo, or C1-4 alkyl;
R5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, Ci-6 alkyl, hydroxyl, Ci-6 alkoxyl, halo, or cyano;
R6 and R7 each represent independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, Ci-6 alkyl, Ci-6 haloalkyl, Ci-6 hydroxyalkyl, or -N(R9)2;
R8 and R9 each represent independently for each occurrence hydrogen, C 1-4 alkyl, or C3-5 cycloalkyl; or two occurrences of R9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom;
A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p
X1 is C(H) or N;
X2 is N, C(CN), or C(R4);
L is a covalent bond or a C1-4 bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or -C(O)-;
Y1, Y2, and Y3 represent independently a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R7)-, or -C(R7)2-; and m, n, and p are each independently 0, 1, 2, or 3.
[0187] The definitions of variables in Formula II above encompass multiple chemical groups.
The application contemplates embodiments where, for example, i) the definition of a variable is a single chemical group selected from those chemical groups set forth above, ii) the definition of a variable is a collection of two or more of the chemical groups selected from those set forth above, and iii) the compound is defined by a combination of variables in which the variables are defined by (i) or (ii).
[0188] In certain embodiments, the compound is a compound of Formula II.
[0189] As defined generally above, R1 is naphthyl; an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with m occurrences of R5.
[0190] In certain embodiments, R1 is naphthyl, an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur, or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R5. In certain embodiments, R1 is phenyl or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R5.
[0191] In certain embodiments, R1 is naphthyl or phenyl; each of which is substituted with m occurrences of R5. In certain embodiments, R1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R5.
[0192] In certain embodiments, R1 is naphthyl or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R5. In certain embodiments, R1 is an 8-10 membered saturated
or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur, or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R5.
[0193] In certain embodiments, R1 is naphthyl substituted with m occurrences of R5. In certain embodiments, R1 is 1-naphthyl substituted with m occurrences of R5. In certain embodiments,
[0194] In certain embodiments, R1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with m occurrences of R5. In certain embodiments, R1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with m occurrences of R5.
[0195] In certain embodiments, R1 is
certain embodiments, R1 is
In certain embodiments, R1 is
In certain embodiments, R1 is
[0196] In certain embodiments, R1 is an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is
substituted with m occurrences of R5. In certain embodiments, R1 is an 8-10 membered bicyclic heteroaryl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R5.
[0197] In certain embodiments, R1 is quinolinyl, isoquinolinyl, or indazolyl; each of which is substituted with m occurrences of R5. In certain embodiments, R1 is quinolinyl substituted with m occurrences of R5. In certain embodiments, R1 is quinoline-4-yl substituted with m occurrences of R5. In certain embodiments, R1 is isoquinolinyl substituted with m occurrences of R5. In certain embodiments, R1 is isoquinolin-l-yl substituted with m occurrences of R5. In certain embodiments, R1 is indazolyl substituted with m occurrences of R5.
[0198] In certain embodiments, R1 is phenyl substituted with m occurrences of R5. In certain embodiments, R1 is
In certain embodiments, R1 is
[0199] In certain embodiments, R1 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R5. In certain embodiments, R1 is a 6-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with m occurrences of R5. In certain embodiments, R1 is a 5-membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R5.
[0200] In certain embodiments, R1 is selected from the groups depicted in the compounds in Table 2, below.
[0201] As defined generally above, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci-6 aliphatic; each of which is substituted with n occurrences of R6.
[0202] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R6. In certain embodiments, R2 is phenyl or Ci-6 aliphatic; each of which is substituted with n occurrences of R6.
[0203] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R6. In certain embodiments, R2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R6.
[0204] In certain embodiments, R2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci-6 aliphatic; each of which is substituted with n occurrences of R6. In certain embodiments, R2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or Ci-6 aliphatic; each of which is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with n occurrences of R6.
[0205] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain
embodiments, R2 is an 8-10 membered saturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is an 8-10 membered partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6.
[0206] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 nitrogen atom; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is an 8-10 membered saturated bicyclic heterocyclyl having 1 nitrogen atom; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments,
[0207] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur. In certain embodiments, R2 is an 8-10 membered saturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur. In certain embodiments, R2 is an 8-10 membered partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur.
[0208] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen. In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 nitrogen atom. In certain embodiments, R2 is an 8-10 membered saturated bicyclic heterocyclyl having 1 nitrogen atom. In certain embodiments, R2 is
[0209] In certain embodiments, R2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 4-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 6-membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6.
[0210] In certain embodiments, R2 is a 3-7 membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 3-7 membered partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6 membered partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6.
[0211] In certain embodiments, R2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6
membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R6.
[0212] In certain embodiments, R2 is azetidinyl, pyrrolidinyl, piperidinyl, or morpholinyl; each of which is substituted with n occurrences of R6. In certain embodiments, R2 is azetidinyl substituted with n occurrences of R6. In certain embodiments, R2 is pyrrolidinyl substituted with n occurrences of R6. In certain embodiments, R2 is pyrrolidin-2-yl substituted with n occurrences of R6. In certain embodiments, R2 is pyrrolidin-2-yl substituted with a single occurrence of R6 at the 1-position. In certain embodiments, R2 is piperidinyl substituted with n occurrences of R6. In certain embodiments, R2 is morpholinyl substituted with n occurrences of R6.
[0213] In certain embodiments, R2 is azetidinyl, pyrrolidinyl, piperidinyl, or morpholinyl. In certain embodiments, R2 is azetidinyl. In certain embodiments, R2 is pyrrolidinyl. In certain embodiments, R2 is pyrrolidin-2-yl. In certain embodiments, R2 is l-methyl-pyrrolidin-2-yl. In certain embodiments, R2 is piperidinyl. In certain embodiments, R2 is morpholinyl.
[0214] In certain embodiments, R2 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with n occurrences of R6.
[0215] In certain embodiments, R2 is phenyl substituted with n occurrences of R6. In certain embodiments, R2 is phenyl.
[0216] In certain embodiments, R2 is Ci-6 aliphatic substituted with n occurrences of R6. In certain embodiments, R2 is C3-6 cycloaliphatic substituted with n occurrences of R6. In certain embodiments, R2 is a Ci-6 aliphatic chain substituted with n occurrences of R6. In certain embodiments, R2 is C3-6 cycloalkyl substituted with n occurrences of R6. In certain embodiments, R2 is Ci-6 alkyl substituted with n occurrences of R6.
[0217] In certain embodiments, R2 is Ci-6 aliphatic. In certain embodiments, R2 is C3-6 cycloaliphatic. In certain embodiments, R2 is a Ci-6 aliphatic chain. In certain embodiments, R2 is C3-6 cycloalkyl. In certain embodiments, R2 is Ci-6 alkyl.
[0218] In certain embodiments, R2 is selected from the groups depicted in the compounds in Table 2, below.
[0219] As defined generally above, R3 is Ci-4 alkyl. In certain embodiments, R3 is methyl. In certain embodiments, R3 is selected from the groups depicted in the compounds in Table 2, below.
[0220] As defined generally above, R4 is hydrogen, halo, or Ci-4 alkyl. In certain embodiments, R4 is hydrogen. In certain embodiments, R4 is halo. In certain embodiments, R4 is fluoro or chloro. In certain embodiments, R4 is fluoro. In certain embodiments, R4 is chloro. In certain embodiments, R4 is CM alkyl. In certain embodiments, R4 is methyl. In certain embodiments,
R4 is selected from the groups depicted in the compounds in Table 2, below.
[0221] As defined generally above, R5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, Ci-6 alkyl, hydroxyl, Ci-6 alkoxyl, halo, or cyano.
[0222] In certain embodiments, R5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, Ci-6 alkyl, or hydroxyl. In certain embodiments, R5 represents independently for each occurrence ethynyl, ethenyl, C1-3 alkyl, or hydroxyl. In certain embodiments, R5 represents independently for each occurrence C2-6 alkynyl or hydroxyl. In certain embodiments, R5 represents independently for each occurrence ethynyl or hydroxyl. In certain embodiments, one occurrence of R5 is ethynyl. In certain embodiments, one occurrence of R5 is hydroxyl.
[0223] In certain embodiments, R5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, or Ci-6 alkyl. In certain embodiments, R5 represents independently for each occurrence ethynyl, ethenyl, or C1-3 alkyl. In certain embodiments, R5 represents independently for each occurrence hydroxyl or Ci-6 alkoxyl. In certain embodiments, R5 represents independently for each occurrence halo or cyano.
[0224] In certain embodiments, R5 represents independently for each occurrence C2-6 alkynyl. In certain embodiments, R5 is ethynyl. In certain embodiments, R5 represents independently for each occurrence C2-6 alkenyl. In certain embodiments, R5 is ethenyl. In certain embodiments, R5 represents independently for each occurrence Ci-6 alkyl. In certain embodiments, R5 represents independently for each occurrence C1-3 alkyl. In certain embodiments, R5 is methyl. In certain embodiments, R5 is hydroxyl. In certain embodiments, R5 represents independently for each occurrence Ci-6 alkoxyl. In certain embodiments, R5 represents independently for each occurrence halo. In certain embodiments, R5 represents independently for each occurrence fluoro or chloro. In certain embodiments, R5 is fluoro. In certain embodiments, R5 is chloro. In
certain embodiments, R5 is cyano. In certain embodiments, R5 is selected from the groups depicted in the compounds in Table 2, below.
[0225] As defined generally above, R6 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, Ci-6 alkyl, Ci-6 haloalkyl, Ci-6 hydroxyalkyl, or -N(R9)2.
[0226] In certain embodiments, R6 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 alkyl, or Ci-6 haloalkyl. In certain embodiments, R6 represents independently for each occurrence halo, Ci-6 alkyl, or Ci-6 haloalkyl. In certain embodiments, R6 represents independently for each occurrence halo or Ci-6 alkyl. In certain embodiments, R6 represents independently for each occurrence halo, hydroxyl, or Ci-6 alkyl.
[0227] In certain embodiments, R6 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, or -N(R9)2. In certain embodiments, R6 represents independently for each occurrence halo, hydroxyl, or -N(R9)2. In certain embodiments, R6 represents independently for each occurrence halo, Ci-6 alkoxyl, or Ci-6 haloalkoxyl. In certain embodiments, R6 represents independently for each occurrence halo, Ci-6 alkyl, Ci-6 haloalkyl, or Ci-6 hydroxyalkyl. In certain embodiments, R6 represents independently for each occurrence Ci-6 alkyl, Ci-6 haloalkyl, or Ci-6 hydroxyalkyl. In certain embodiments, R6 represents independently for each occurrence Ci-6 alkyl or Ci-6 haloalkyl.
[0228] In certain embodiments, R6 represents independently for each occurrence halo. In certain embodiments, R6 represents independently for each occurrence fluoro or chloro. In certain embodiments, R6 is fluoro. In certain embodiments, R6 is chloro. In certain embodiments, R6 is hydroxyl. In certain embodiments, R6 represents independently for each occurrence Ci-6 alkoxyl. In certain embodiments, R6 is methoxy. In certain embodiments, R6 represents independently for each occurrence Ci-6 haloalkoxyl. In certain embodiments, R6 is trifluoromethoxy. In certain embodiments, R6 represents independently for each occurrence Ci-6 alkyl. In certain embodiments, R6 represents independently for each occurrence C1-3 alkyl. In certain embodiments, R6 is methyl. In certain embodiments, R6 represents independently for each occurrence Ci-6 haloalkyl. In certain embodiments, R6 is trifluoromethyl. In certain embodiments, R6 represents independently for each occurrence Ci-6 hydroxyalkyl. In certain embodiments, R6 represents independently for each occurrence -N(R9)2. In certain embodiments,
R6 is -NH2. In certain embodiments, R6 is selected from the groups depicted in the compounds in Table 2, below.
[0229] As defined generally above, R7 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, Ci-6 alkyl, Ci-6 haloalkyl, Ci-6 hydroxyalkyl, or -N(R9)2.
[0230] In certain embodiments, R7 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 alkyl, or Ci-6 haloalkyl. In certain embodiments, R7 represents independently for each occurrence halo, Ci-6 alkyl, or Ci-6 haloalkyl. In certain embodiments, R7 represents independently for each occurrence halo or Ci-6 alkyl. In certain embodiments, R7 represents independently for each occurrence halo, hydroxyl, or Ci-6 alkyl.
[0231] In certain embodiments, R7 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, or -N(R9)2. In certain embodiments, R7 represents independently for each occurrence halo, hydroxyl, or -N(R9)2. In certain embodiments, R7 represents independently for each occurrence halo, Ci-6 alkoxyl, or Ci-6 haloalkoxyl. In certain embodiments, R7 represents independently for each occurrence halo, Ci-6 alkyl, Ci-6 haloalkyl, or Ci-6 hydroxyalkyl. In certain embodiments, R7 represents independently for each occurrence Ci-6 alkyl, Ci-6 haloalkyl, or Ci-6 hydroxyalkyl. In certain embodiments, R7 represents independently for each occurrence Ci-6 alkyl or Ci-6 haloalkyl.
[0232] In certain embodiments, R7 represents independently for each occurrence halo. In certain embodiments, R7 represents independently for each occurrence fluoro or chloro. In certain embodiments, R7 is fluoro. In certain embodiments, R7 is chloro. In certain embodiments, R7 is hydroxyl. In certain embodiments, R7 represents independently for each occurrence Ci-6 alkoxyl. In certain embodiments, R7 is methoxy. In certain embodiments, R7 represents independently for each occurrence Ci-6 haloalkoxyl. In certain embodiments, R7 is trifluoromethoxy. In certain embodiments, R7 represents independently for each occurrence Ci-6 alkyl. In certain embodiments, R7 represents independently for each occurrence C1-3 alkyl. In certain embodiments, R7 is methyl. In certain embodiments, R7 represents independently for each occurrence Ci-6 haloalkyl. In certain embodiments, R7 is trifluoromethyl. In certain embodiments, R7 represents independently for each occurrence Ci-6 hydroxyalkyl. In certain embodiments, R7 represents independently for each occurrence -N(R9)2. In certain embodiments,
R7 is -NH2. In certain embodiments, R7 is selected from the groups depicted in the compounds in Table 2, below.
[0233] As defined generally above, R8 is hydrogen, C1-4 alkyl, or C3-5 cycloalkyl. In certain embodiments, R8 is hydrogen or C1-4 alkyl. In certain embodiments, R8 is hydrogen or methyl.
In certain embodiments, R8 is CIM alkyl or C3-5 cycloalkyl.
[0234] In certain embodiments, R8 is hydrogen. In certain embodiments, R8 is CIM alkyl. In certain embodiments, R8 is methyl. In certain embodiments, R8 is C3-5 cycloalkyl. In certain embodiments, R8 is cyclopropyl. In certain embodiments, R8 is selected from the groups depicted in the compounds in Table 2, below.
[0235] As defined generally above, R9 represents independently for each occurrence hydrogen, Ci-4 alkyl, or C3-5 cycloalkyl; or two occurrences of R9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom.
[0236] In certain embodiments, R9 represents independently for each occurrence hydrogen, C1-4 alkyl, or C3-5 cycloalkyl. In certain embodiments, R9 represents independently for each occurrence hydrogen or C1-4 alkyl. In certain embodiments, R9 represents independently for each occurrence hydrogen or methyl. In certain embodiments, R9 represents independently for each occurrence C1-4 alkyl or C3-5 cycloalkyl.
[0237] In certain embodiments, two occurrences of R9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom.
[0238] In certain embodiments, R9 is hydrogen. In certain embodiments, R9 represents independently for each occurrence C1-4 alkyl. In certain embodiments, R9 is methyl. In certain embodiments, R9 represents independently for each occurrence C3-5 cycloalkyl. In certain embodiments, R9 is cyclopropyl. In certain embodiments, R9 is selected from the groups depicted in the compounds in Table 2, below.
[0239] As defined generally above, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen
atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R7; or A1 is
, ,
[0240] In certain embodiments, A1 is
In certain embodiments, A1 is
in certain embodiments, A1 is
In certain embodiments, A1 is
[0241] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5- 6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R7.
[0242] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R7; or A1 is
[0243] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R7.
[0244] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R7; or A1 is
or
[0245] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R7.
[0246] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R7.
[0247] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5- 6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur.
[0248] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen
atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or
[0249] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
[0250] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2
[0251] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
[0252] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur.
[0253] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7. In certain embodiments, A1 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7. In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A1 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms.
[0254] In certain embodiments, A1 is 1,4-diazepanyl, piperidinyl, piperazinyl, pyrrolidinyl, or azetidinyl; each of which is substituted with p occurrences of R7. In certain embodiments, A1 is
1,4-diazepanyl substituted with p occurrences of R7. In certain embodiments, A1 is piperidinyl substituted with p occurrences of R7. In certain embodiments, A1 is piperazinyl substituted with p occurrences of R7. In certain embodiments, A1 is pyrrolidinyl substituted with p occurrences of R7. In certain embodiments, A1 is azetidinyl substituted with p occurrences of R7.
[0255] In certain embodiments, A1 is 1,4-diazepanyl, piperidinyl, piperazinyl, pyrrolidinyl, or azetidinyl. In certain embodiments, A1 is 1,4-diazepanyl. In certain embodiments, A1 is piperidinyl. In certain embodiments, A1 is piperazinyl. In certain embodiments, A1 is pyrrolidinyl. In certain embodiments, A1 is azetidinyl.
[0256] In certain embodiments, A1 is 1,4-diazepan-l-yl, piperidin-4-yl, piperazin-l-yl, pyrrolidin-3-yl, or azetidin-3-yl; each of which is substituted with p occurrences of R7. In certain embodiments, A1 is 1,4-diazepan-l-yl substituted with p occurrences of R7. In certain embodiments, A1 is piperidin-4-yl substituted with p occurrences of R7. In certain embodiments, A1 is piperazin-l-yl substituted with p occurrences of R7. In certain embodiments, A1 is pyrrolidin-3-yl substituted with p occurrences of R7. In certain embodiments, A1 is azetidin-3-yl substituted with p occurrences of R7.
[0257] In certain embodiments, A1 is 1,4-diazepan-l-yl, piperidin-4-yl, piperazin-l-yl, pyrrolidin-3-yl, or azetidin-3-yl. In certain embodiments, A1 is 1,4-diazepan-l-yl. In certain embodiments, A1 is piperidin-4-yl. In certain embodiments, A1 is piperazin-l-yl. In certain embodiments, A1 is pyrrolidin-3-yl. In certain embodiments, A1 is azetidin-3-yl.
[0258] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7. In certain embodiments, A1 is a 7-10 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7. In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7.
[0259] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A1 is a 7-10 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A1 is a 6-11 membered
saturated spirocyclic heterocyclyl having 2 nitrogen atoms. In certain embodiments, A1 is
or
In certain embodiments, A1 is
In certain embodiments, A1 is
[0260] In certain embodiments, A1 is a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7. In certain embodiments, A1 is a 7-10 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7. In certain embodiments, A1 is a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A1 is a 7-10 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
[0261] In certain embodiments, A1 is a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with p occurrences of R7.
[0262] In certain embodiments, A1 is a 6-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with p occurrences of R7. In certain embodiments, A1 is pyridinyl or pyrimidinyl; each of which is substituted with p occurrences of R7. In certain embodiments, A1 is pyridinyl substituted with p occurrences of R7. In certain embodiments, A1 is pyridin-4-yl substituted with p occurrences of R7. In certain embodiments, A1 is pyrimidinyl substituted with p occurrences of R7.
[0263] In certain embodiments, A1 is a 5-membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with p occurrences of R7. In certain embodiments, A1 is a 5-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with p occurrences of R7. In certain embodiments, A1 is imidazolyl or pyrazolyl; each of which is substituted with p occurrences of R7. In certain embodiments, A1 is
imidazolyl substituted with p occurrences of R7. In certain embodiments, A1 is pyrazolyl substituted with p occurrences of R7.
[0264] In certain embodiments, A1 is selected from the groups depicted in the compounds in Table 2, below.
[0265] As defined generally above, X1 is C(H) or N. In certain embodiments, X1 is C(H). In certain embodiments, X1 is N. In certain embodiments, X1 is selected from the groups depicted in the compounds in Table 2, below.
[0266] As defined generally above, X2 is N, C(CN), or C(R4). In certain embodiments, X2 is N. In certain embodiments, X2 is C(CN) or C(R4). In certain embodiments, X2 is C(CN) or C(H).
In certain embodiments, X2 is C(CN). In certain embodiments, X2 is C(R4). In certain embodiments, X2 is C(H). In certain embodiments, X2 is selected from the groups depicted in the compounds in Table 2, below.
[0267] As defined generally above, L is a covalent bond or a Ci-4 bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or -C(O)-. In certain embodiments, L is a covalent bond.
[0268] In certain embodiments, L is a Ci-4 bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or -C(O)-. In certain embodiments, L is a Ci-4 bivalent saturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or -C(O)-. In certain embodiments, L is a CM bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or - C(O)-. In certain embodiments, L is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -N(R9)-, or -C(O)-.
[0269] In certain embodiments, L is a C1-4 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -N(R9)-, or -C(O)-. In certain embodiments, L is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene
unit of the chain is optionally replaced by -0-, -N(R9)-, or -C(O)-. In certain embodiments, L is a Ci-4 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -O- or -N(R9)-. In certain embodiments, L is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -O- or -N(R9)-.
[0270] In certain embodiments, L is -O-CH2-, -0-(CH2)2-, -N(R9)-CH2-, or -N(R9)-(CH2)2-, wherein the terminal carbon atom of L is attached to R2. In certain embodiments, L is -O-CH2- or -0-(CH2)2-, wherein the terminal carbon atom of L is attached to R2. In certain embodiments, L is -N(R9)-CH2- or -N(R9)-(CH2)2-, wherein the terminal carbon atom of L is attached to R2. In certain embodiments, L is -O-CH2-, wherein the carbon atom of L is attached to R2. In certain embodiments, L is -0-(CH2)2-, wherein the terminal carbon atom of L is attached to R2. In certain embodiments, L is -N(R9)-CH2-, wherein the carbon atom of L is attached to R2. In certain embodiments, L is -N(R9)-(CH2)2-, wherein the terminal carbon atom of L is attached to R2.
[0271] In certain embodiments, L is selected from the groups depicted in the compounds in Table 2, below.
[0272] As defined generally above, Y1 is a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -O-, -C(H)(R7)-, or -C(R7)2-.
[0273] In certain embodiments, Y1 is a C2-3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -O-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y1 is a C2-3 bivalent unsaturated, straight hydrocarbon chain.
[0274] In certain embodiments, Y1 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - O-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y1 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -O-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y1 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y1 is a C1-3 bivalent saturated, straight hydrocarbon chain.
[0275] In certain embodiments, Y1 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y1 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y1 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y1 is a C2-3 bivalent saturated, straight hydrocarbon chain.
[0276] In certain embodiments, Y1 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, -(CH2)3-, -CH2-O- CH2-, or -CH2-CF2-CH2-. In certain embodiments, Y1 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, - (CH2)3-, or -CH2-CF2-CH2-. In certain embodiments, Y1 is -(CH2)2-, -CH2-C(H)(OH)-, -(CH2)3-, or -CH2-CF2-CH2-. In certain embodiments, Y1 is -(CH2)2- or -(CH2)3-. In certain embodiments, Y1 is -CH2-C(H)(OH)- or -CH2-CF2-CH2-. In certain embodiments, Y1 is -(CH2)-, -(CH2)2-, or - (CH2)3-. In certain embodiments, Y1 is -(CH2)2- or -CH2-C(H)(OH)-. In certain embodiments, Y1 is -(CH2)3-, -CH2-O-CH2-, or -CH2-CF2-CH2-.
[0277] In certain embodiments, Y1 is -(CH2)-. In certain embodiments, Y1 is -(CH2)2-. In certain embodiments, Y1 is -CH2-C(H)(OH)-. In certain embodiments, Y1 is -(CH2)3-. In certain embodiments, Y1 is -CH2-O-CH2-. In certain embodiments, Y1 is -CH2-CF2-CH2-. In certain embodiments, Y1 is selected from the groups depicted in the compounds in Table 2, below.
[0278] As defined generally above, Y2 is a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R7)-, or -C(R7)2-.
[0279] In certain embodiments, Y2 is a C2-3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -C(H)(R7)-, or -C(R7)2-.
In certain embodiments, Y2 is a C2-3 bivalent unsaturated, straight hydrocarbon chain.
[0280] In certain embodiments, Y2 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y2 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y2 is a C1-3 bivalent saturated, straight
hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y2 is a C1-3 bivalent saturated, straight hydrocarbon chain.
[0281] In certain embodiments, Y2 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y2 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y2 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y2 is a C2-3 bivalent saturated, straight hydrocarbon chain.
[0282] In certain embodiments, Y2 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, -(CH2) -, -CH2-0- CH2-, or -CH2-CF2-CH2-. In certain embodiments, Y2 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, - (CH2) -, or -CH2-CF2-CH2-. In certain embodiments, Y2 is -(CH2)2-, -CH2-C(H)(OH)-, -(CH2) -, or -CH2-CF2-CH2-. In certain embodiments, Y2 is -(CH2)2- or -(CH2)3-. In certain embodiments, Y2 is -CH2-C(H)(OH)- or -CH2-CF2-CH2-. In certain embodiments, Y2 is -(CH2)-, -(CH2)2-, or - (CH2) -. In certain embodiments, Y2 is -(CH2)2- or -CH2-C(H)(OH)-. In certain embodiments, Y2 is -(CH2) -, -CH2-0-CH2-, or -CH2-CF2-CH2-.
[0283] In certain embodiments, Y2 is -(CH2)-. In certain embodiments, Y2 is -(CH2)2-. In certain embodiments, Y2 is -CH2-C(H)(OH)-. In certain embodiments, Y2 is -(CH2)3-. In certain embodiments, Y2 is -CH2-0-CH2-. In certain embodiments, Y2 is -CH2-CF2-CH2-. In certain embodiments, Y2 is selected from the groups depicted in the compounds in Table 2, below.
[0284] As defined generally above, Y3 is a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R7)-, or -C(R7)2-.
[0285] In certain embodiments, Y3 is a C2-3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -C(H)(R7)-, or -C(R7)2-.
In certain embodiments, Y3 is a C2-3 bivalent unsaturated, straight hydrocarbon chain.
[0286] In certain embodiments, Y3 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y3 is a C1-3 bivalent saturated, straight
hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y3 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y3 is a C1-3 bivalent saturated, straight hydrocarbon chain.
[0287] In certain embodiments, Y3 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y3 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y3 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y3 is a C2-3 bivalent saturated, straight hydrocarbon chain.
[0288] In certain embodiments, Y3 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, -(CH2)3-, -CH2-0- CH2-, or -CH2-CF2-CH2-. In certain embodiments, Y3 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, - (CH2)3-, or -CH2-CF2-CH2-. In certain embodiments, Y3 is -(CH2)2-, -CH2-C(H)(OH)-, -(CH2)3-, or -CH2-CF2-CH2-. In certain embodiments, Y3 is -(CH2)2- or -(CH2)3-. In certain embodiments, Y3 is -CH2-C(H)(OH)- or -CH2-CF2-CH2-. In certain embodiments, Y3 is -(CH2)-, -(CH2)2-, or - (CH2)3-. In certain embodiments, Y3 is -(CH2)2- or -CH2-C(H)(OH)-. In certain embodiments, Y3 is -(CH2)3-, -CH2-0-CH2-, or -CH2-CF2-CH2-.
[0289] In certain embodiments, Y3 is -(CH2)-. In certain embodiments, Y3 is -(CH2)2-. In certain embodiments, Y3 is -CH2-C(H)(OH)-. In certain embodiments, Y3 is -(CH2)3-. In certain embodiments, Y3 is -CH2-0-CH2-. In certain embodiments, Y3 is -CH2-CF2-CH2-. In certain embodiments, Y3 is selected from the groups depicted in the compounds in Table 2, below.
[0290] As defined generally above, m is 0, 1, 2, or 3. In certain embodiments, m is 0. In certain embodiments, m is 1. In certain embodiments, m is 2. In certain embodiments, m is 3. In certain embodiments, m is 0 or 1. In certain embodiments, m is 1 or 2. In certain embodiments, m is 2 or 3. In certain embodiments, m is 0, 1, or 2. In certain embodiments, m is 1, 2, or 3. In certain embodiments, m is selected from the values represented in the compounds in Table 2, below.
[0291] As defined generally above, n is 0, 1, 2, or 3. In certain embodiments, n is 0. In certain embodiments, n is 1. In certain embodiments, n is 2. In certain embodiments, n is 3. In certain
embodiments, n is 0 or 1. In certain embodiments, n is 1 or 2. In certain embodiments, n is 2 or 3. In certain embodiments, n is 0, 1, or 2. In certain embodiments, n is 1, 2, or 3. In certain embodiments, n is selected from the values represented in the compounds in Table 2, below.
[0292] As defined generally above, p is 0, 1, 2, or 3. In certain embodiments, p is 0. In certain embodiments, p is 1. In certain embodiments, p is 2. In certain embodiments, p is 3. In certain embodiments, p is 0 or 1. In certain embodiments, p is 1 or 2. In certain embodiments, p is 2 or 3. In certain embodiments, p is 0, 1, or 2. In certain embodiments, p is 1, 2, or 3. In certain embodiments, p is selected from the values represented in the compounds in Table 2, below.
[0293] The description above describes multiple embodiments relating to compounds of Formula II. The patent application specifically contemplates all combinations of the embodiments.
[0294] Another aspect of the invention provides a compound represented by Formula III:
or a pharmaceutically acceptable salt thereof; wherein:
R1 is naphthyl; an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with m occurrences of R5;
R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic
heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci-6 aliphatic; each of which is substituted with n occurrences of R6;
R4 is hydrogen, halo, or C1-4 alkyl;
R5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, Ci-6 alkyl, hydroxyl, Ci-6 alkoxyl, halo, or cyano;
R6 and R7 each represent independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, Ci-6 alkyl, Ci-6 haloalkyl, Ci-6 hydroxyalkyl, or -N(R9)2;
R8 and R9 each represent independently for each occurrence hydrogen, C1-4 alkyl, or C3-5 cycloalkyl; or two occurrences of R9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom;
A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p
X2 is N, C(CN), or C(R4);
L is a covalent bond or a CM bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or -C(O)-;
Y1, Y2, and Y3 represent independently a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R7)-, or -C(R7)2-; and m, n, and p are each independently 0, 1, 2, or 3.
[0295] The definitions of variables in Formula III above encompass multiple chemical groups. The application contemplates embodiments where, for example, i) the definition of a variable is a single chemical group selected from those chemical groups set forth above, ii) the definition of a variable is a collection of two or more of the chemical groups selected from those set forth above, and iii) the compound is defined by a combination of variables in which the variables are defined by (i) or (ii).
[0296] In certain embodiments, the compound is a compound of Formula III.
[0297] As defined generally above, R1 is naphthyl; an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with m occurrences of R5.
[0298] In certain embodiments, R1 is naphthyl, an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur, or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R5. In certain embodiments, R1 is phenyl or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R5.
[0299] In certain embodiments, R1 is naphthyl or phenyl; each of which is substituted with m occurrences of R5. In certain embodiments, R1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R5.
[0300] In certain embodiments, R1 is naphthyl or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R5. In certain embodiments, R1 is an 8-10 membered saturated
or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur, or an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with m occurrences of R5.
[0301] In certain embodiments, R1 is naphthyl substituted with m occurrences of R5. In certain embodiments, R1 is 1-naphthyl substituted with m occurrences of R5. In certain embodiments,
[0302] In certain embodiments, R1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with m occurrences of R5. In certain embodiments, R1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with m occurrences of R5.
[0303] In certain embodiments, R1 is
certain embodiments, R1 is
In certain embodiments, R1 is
In certain embodiments, R1 is
[0304] In certain embodiments, R1 is an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is
substituted with m occurrences of R5. In certain embodiments, R1 is an 8-10 membered bicyclic heteroaryl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R5.
[0305] In certain embodiments, R1 is quinolinyl, isoquinolinyl, or indazolyl; each of which is substituted with m occurrences of R5. In certain embodiments, R1 is quinolinyl substituted with m occurrences of R5. In certain embodiments, R1 is quinoline-4-yl substituted with m occurrences of R5. In certain embodiments, R1 is isoquinolinyl substituted with m occurrences of R5. In certain embodiments, R1 is isoquinolin-l-yl substituted with m occurrences of R5. In certain embodiments, R1 is indazolyl substituted with m occurrences of R5.
[0306] In certain embodiments, R1 is phenyl substituted with m occurrences of R5. In certain embodiments, R1 is In certain embodiments, R1 is
[0307] In certain embodiments, R1 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R5. In certain embodiments, R1 is a 6-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with m occurrences of R5. In certain embodiments, R1 is a 5-membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R5.
[0308] In certain embodiments, R1 is selected from the groups depicted in the compounds in Table 3, below.
[0309] As defined generally above, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci-6 aliphatic; each of which is substituted with n occurrences of R6.
[0310] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R6. In certain embodiments, R2 is phenyl or Ci-6 aliphatic; each of which is substituted with n occurrences of R6.
[0311] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R6. In certain embodiments, R2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with n occurrences of R6.
[0312] In certain embodiments, R2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci-6 aliphatic; each of which is substituted with n occurrences of R6. In certain embodiments, R2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or Ci-6 aliphatic; each of which is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with n occurrences of R6.
[0313] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain
embodiments, R2 is an 8-10 membered saturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is an 8-10 membered partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6.
[0314] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 nitrogen atom; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is an 8-10 membered saturated bicyclic heterocyclyl having 1 nitrogen atom; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments,
[0315] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur. In certain embodiments, R2 is an 8-10 membered saturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur. In certain embodiments, R2 is an 8-10 membered partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur.
[0316] In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen. In certain embodiments, R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1 nitrogen atom. In certain embodiments, R2 is an 8-10 membered saturated bicyclic heterocyclyl having 1 nitrogen atom. In certain embodiments, R2 is
[0317] In certain embodiments, R2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 4-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 6-membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6.
[0318] In certain embodiments, R2 is a 3-7 membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 3-7 membered partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6 membered partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6.
[0319] In certain embodiments, R2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R6. In certain embodiments, R2 is a 5-6
membered saturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen and oxygen; wherein said heterocyclyl is substituted with n occurrences of R6.
[0320] In certain embodiments, R2 is azetidinyl, pyrrolidinyl, piperidinyl, or morpholinyl; each of which is substituted with n occurrences of R6. In certain embodiments, R2 is azetidinyl substituted with n occurrences of R6. In certain embodiments, R2 is pyrrolidinyl substituted with n occurrences of R6. In certain embodiments, R2 is pyrrolidin-2-yl substituted with n occurrences of R6. In certain embodiments, R2 is pyrrolidin-2-yl substituted with a single occurrence of R6 at the 1-position. In certain embodiments, R2 is piperidinyl substituted with n occurrences of R6. In certain embodiments, R2 is morpholinyl substituted with n occurrences of R6.
[0321] In certain embodiments, R2 is azetidinyl, pyrrolidinyl, piperidinyl, or morpholinyl. In certain embodiments, R2 is azetidinyl. In certain embodiments, R2 is pyrrolidinyl. In certain embodiments, R2 is pyrrolidin-2-yl. In certain embodiments, R2 is l-methyl-pyrrolidin-2-yl. In certain embodiments, R2 is piperidinyl. In certain embodiments, R2 is morpholinyl.
[0322] In certain embodiments, R2 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with n occurrences of R6.
[0323] In certain embodiments, R2 is phenyl substituted with n occurrences of R6. In certain embodiments, R2 is phenyl.
[0324] In certain embodiments, R2 is Ci-6 aliphatic substituted with n occurrences of R6. In certain embodiments, R2 is C3-6 cycloaliphatic substituted with n occurrences of R6. In certain embodiments, R2 is a Ci-6 aliphatic chain substituted with n occurrences of R6. In certain embodiments, R2 is C3-6 cycloalkyl substituted with n occurrences of R6. In certain embodiments, R2 is Ci-6 alkyl substituted with n occurrences of R6.
[0325] In certain embodiments, R2 is Ci-6 aliphatic. In certain embodiments, R2 is C3-6 cycloaliphatic. In certain embodiments, R2 is a Ci-6 aliphatic chain. In certain embodiments, R2 is C3-6 cycloalkyl. In certain embodiments, R2 is Ci-6 alkyl.
[0326] In certain embodiments, R2 is selected from the groups depicted in the compounds in Table 3, below.
[0327] As defined generally above, R4 is hydrogen, halo, or Ci-4 alkyl. In certain embodiments, R4 is hydrogen. In certain embodiments, R4 is halo. In certain embodiments, R4 is fluoro or chloro. In certain embodiments, R4 is fluoro. In certain embodiments, R4 is chloro. In certain embodiments, R4 is C1-4 alkyl. In certain embodiments, R4 is methyl. In certain embodiments,
R4 is selected from the groups depicted in the compounds in Table 3, below.
[0328] As defined generally above, R5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, C1-6 alkyl, hydroxyl, C1-6 alkoxyl, halo, or cyano.
[0329] In certain embodiments, R5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, C1-6 alkyl, or hydroxyl. In certain embodiments, R5 represents independently for each occurrence ethynyl, ethenyl, C1-3 alkyl, or hydroxyl. In certain embodiments, R5 represents independently for each occurrence C2-6 alkynyl or hydroxyl. In certain embodiments, R5 represents independently for each occurrence ethynyl or hydroxyl. In certain embodiments, one occurrence of R5 is ethynyl. In certain embodiments, one occurrence of R5 is hydroxyl.
[0330] In certain embodiments, R5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, or C1-6 alkyl. In certain embodiments, R5 represents independently for each occurrence ethynyl, ethenyl, or C1-3 alkyl. In certain embodiments, R5 represents independently for each occurrence hydroxyl or C1-6 alkoxyl. In certain embodiments, R5 represents independently for each occurrence halo or cyano.
[0331] In certain embodiments, R5 represents independently for each occurrence C2-6 alkynyl. In certain embodiments, R5 is ethynyl. In certain embodiments, R5 represents independently for each occurrence C2-6 alkenyl. In certain embodiments, R5 is ethenyl. In certain embodiments, R5 represents independently for each occurrence Ci-6 alkyl. In certain embodiments, R5 represents independently for each occurrence C1-3 alkyl. In certain embodiments, R5 is methyl. In certain embodiments, R5 is hydroxyl. In certain embodiments, R5 represents independently for each occurrence C1-6 alkoxyl. In certain embodiments, R5 represents independently for each occurrence halo. In certain embodiments, R5 represents independently for each occurrence fluoro or chloro. In certain embodiments, R5 is fluoro. In certain embodiments, R5 is chloro. In certain embodiments, R5 is cyano. In certain embodiments, R5 is selected from the groups depicted in the compounds in Table 3, below.
[0332] As defined generally above, R6 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, Ci-6 alkyl, Ci-6 haloalkyl, Ci-6 hydroxyalkyl, or -N(R9)2.
[0333] In certain embodiments, R6 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 alkyl, or Ci-6 haloalkyl. In certain embodiments, R6 represents independently for each occurrence halo, Ci-6 alkyl, or Ci-6 haloalkyl. In certain embodiments, R6 represents independently for each occurrence halo or Ci-6 alkyl. In certain embodiments, R6 represents independently for each occurrence halo, hydroxyl, or Ci-6 alkyl.
[0334] In certain embodiments, R6 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, or -N(R9)2. In certain embodiments, R6 represents independently for each occurrence halo, hydroxyl, or -N(R9)2. In certain embodiments, R6 represents independently for each occurrence halo, Ci-6 alkoxyl, or Ci-6 haloalkoxyl. In certain embodiments, R6 represents independently for each occurrence halo, Ci-6 alkyl, Ci-6 haloalkyl, or Ci-6 hydroxyalkyl. In certain embodiments, R6 represents independently for each occurrence Ci-6 alkyl, Ci-6 haloalkyl, or Ci-6 hydroxyalkyl. In certain embodiments, R6 represents independently for each occurrence Ci-6 alkyl or Ci-6 haloalkyl.
[0335] In certain embodiments, R6 represents independently for each occurrence halo. In certain embodiments, R6 represents independently for each occurrence fluoro or chloro. In certain embodiments, R6 is fluoro. In certain embodiments, R6 is chloro. In certain embodiments, R6 is hydroxyl. In certain embodiments, R6 represents independently for each occurrence Ci-6 alkoxyl. In certain embodiments, R6 is methoxy. In certain embodiments, R6 represents independently for each occurrence Ci-6 haloalkoxyl. In certain embodiments, R6 is trifluoromethoxy. In certain embodiments, R6 represents independently for each occurrence Ci-6 alkyl. In certain embodiments, R6 represents independently for each occurrence C1-3 alkyl. In certain embodiments, R6 is methyl. In certain embodiments, R6 represents independently for each occurrence Ci-6 haloalkyl. In certain embodiments, R6 is trifluoromethyl. In certain embodiments, R6 represents independently for each occurrence Ci-6 hydroxyalkyl. In certain embodiments, R6 represents independently for each occurrence -N(R9)2. In certain embodiments, R6 is -NH2. In certain embodiments, R6 is selected from the groups depicted in the compounds in Table 3, below.
[0336] As defined generally above, R7 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, Ci-6 alkyl, Ci-6 haloalkyl, Ci-6 hydroxyalkyl, or -N(R9)2.
[0337] In certain embodiments, R7 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 alkyl, or Ci-6 haloalkyl. In certain embodiments, R7 represents independently for each occurrence halo, Ci-6 alkyl, or Ci-6 haloalkyl. In certain embodiments, R7 represents independently for each occurrence halo or Ci-6 alkyl. In certain embodiments, R7 represents independently for each occurrence halo, hydroxyl, or Ci-6 alkyl.
[0338] In certain embodiments, R7 represents independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, or -N(R9)2. In certain embodiments, R7 represents independently for each occurrence halo, hydroxyl, or -N(R9)2. In certain embodiments, R7 represents independently for each occurrence halo, Ci-6 alkoxyl, or Ci-6 haloalkoxyl. In certain embodiments, R7 represents independently for each occurrence halo, Ci-6 alkyl, Ci-6 haloalkyl, or Ci-6 hydroxyalkyl. In certain embodiments, R7 represents independently for each occurrence Ci-6 alkyl, Ci-6 haloalkyl, or Ci-6 hydroxyalkyl. In certain embodiments, R7 represents independently for each occurrence Ci-6 alkyl or Ci-6 haloalkyl.
[0339] In certain embodiments, R7 represents independently for each occurrence halo. In certain embodiments, R7 represents independently for each occurrence fluoro or chloro. In certain embodiments, R7 is fluoro. In certain embodiments, R7 is chloro. In certain embodiments, R7 is hydroxyl. In certain embodiments, R7 represents independently for each occurrence Ci-6 alkoxyl. In certain embodiments, R7 is methoxy. In certain embodiments, R7 represents independently for each occurrence Ci-6 haloalkoxyl. In certain embodiments, R7 is trifluoromethoxy. In certain embodiments, R7 represents independently for each occurrence Ci-6 alkyl. In certain embodiments, R7 represents independently for each occurrence C1-3 alkyl. In certain embodiments, R7 is methyl. In certain embodiments, R7 represents independently for each occurrence Ci-6 haloalkyl. In certain embodiments, R7 is trifluoromethyl. In certain embodiments, R7 represents independently for each occurrence Ci-6 hydroxyalkyl. In certain embodiments, R7 represents independently for each occurrence -N(R9)2. In certain embodiments, R7 is -NH2. In certain embodiments, R7 is selected from the groups depicted in the compounds in Table 3, below.
[0340] As defined generally above, R8 is hydrogen, Ci alkyl, or C3-5 cycloalkyl. In certain embodiments, R8 is hydrogen or CM alkyl. In certain embodiments, R8 is hydrogen or methyl.
In certain embodiments, R8 is CIM alkyl or C3-5 cycloalkyl.
[0341] In certain embodiments, R8 is hydrogen. In certain embodiments, R8 is CIM alkyl. In certain embodiments, R8 is methyl. In certain embodiments, R8 is C3-5 cycloalkyl. In certain embodiments, R8 is cyclopropyl. In certain embodiments, R8 is selected from the groups depicted in the compounds in Table 3, below.
[0342] As defined generally above, R9 represents independently for each occurrence hydrogen, Ci-4 alkyl, or C3-5 cycloalkyl; or two occurrences of R9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom.
[0343] In certain embodiments, R9 represents independently for each occurrence hydrogen, C1-4 alkyl, or C3-5 cycloalkyl. In certain embodiments, R9 represents independently for each occurrence hydrogen or C1-4 alkyl. In certain embodiments, R9 represents independently for each occurrence hydrogen or methyl. In certain embodiments, R9 represents independently for each occurrence C1-4 alkyl or C3-5 cycloalkyl.
[0344] In certain embodiments, two occurrences of R9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom.
[0345] In certain embodiments, R9 is hydrogen. In certain embodiments, R9 represents independently for each occurrence C1-4 alkyl. In certain embodiments, R9 is methyl. In certain embodiments, R9 represents independently for each occurrence C3-5 cycloalkyl. In certain embodiments, R9 is cyclopropyl. In certain embodiments, R9 is selected from the groups depicted in the compounds in Table 3, below.
[0346] As defined generally above, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2
additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R7; or A1 is
[0347] In certain embodiments, A1 is
In certain embodiments, A1 is
In certain embodiments, A1 is
In certain embodiments, A1 is
[0348] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5- 6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R7.
[0349] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R7; or A1 is
[0350] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R7.
[0351] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R7; or A1 is
or
[0352] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; each of which is substituted with p occurrences of R7.
[0353] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p occurrences of R7.
[0354] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5- 6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur.
[0355] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen
atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or
[0356] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
[0357] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2
[0358] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
[0359] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur.
[0360] In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7. In certain embodiments, A1 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7. In certain embodiments, A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A1 is a 5-6 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms.
[0361] In certain embodiments, A1 is 1,4-diazepanyl, piperidinyl, piperazinyl, pyrrolidinyl, or azetidinyl; each of which is substituted with p occurrences of R7. In certain embodiments, A1 is
1,4-diazepanyl substituted with p occurrences of R7. In certain embodiments, A1 is piperidinyl substituted with p occurrences of R7. In certain embodiments, A1 is piperazinyl substituted with p occurrences of R7. In certain embodiments, A1 is pyrrolidinyl substituted with p occurrences of R7. In certain embodiments, A1 is azetidinyl substituted with p occurrences of R7.
[0362] In certain embodiments, A1 is 1,4-diazepanyl, piperidinyl, piperazinyl, pyrrolidinyl, or azetidinyl. In certain embodiments, A1 is 1,4-diazepanyl. In certain embodiments, A1 is piperidinyl. In certain embodiments, A1 is piperazinyl. In certain embodiments, A1 is pyrrolidinyl. In certain embodiments, A1 is azetidinyl.
[0363] In certain embodiments, A1 is 1,4-diazepan-l-yl, piperidin-4-yl, piperazin-l-yl, pyrrolidin-3-yl, or azetidin-3-yl; each of which is substituted with p occurrences of R7. In certain embodiments, A1 is 1,4-diazepan-l-yl substituted with p occurrences of R7. In certain embodiments, A1 is piperidin-4-yl substituted with p occurrences of R7. In certain embodiments, A1 is piperazin-l-yl substituted with p occurrences of R7. In certain embodiments, A1 is pyrrolidin-3-yl substituted with p occurrences of R7. In certain embodiments, A1 is azetidin-3-yl substituted with p occurrences of R7.
[0364] In certain embodiments, A1 is 1,4-diazepan-l-yl, piperidin-4-yl, piperazin-l-yl, pyrrolidin-3-yl, or azetidin-3-yl. In certain embodiments, A1 is 1,4-diazepan-l-yl. In certain embodiments, A1 is piperidin-4-yl. In certain embodiments, A1 is piperazin-l-yl. In certain embodiments, A1 is pyrrolidin-3-yl. In certain embodiments, A1 is azetidin-3-yl.
[0365] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7. In certain embodiments, A1 is a 7-10 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7. In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7.
[0366] In certain embodiments, A1 is a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A1 is a 7-10 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A1 is a 6-11 membered
saturated spirocyclic heterocyclyl having 2 nitrogen atoms. In certain embodiments, A1 is
or In certain embodiments, A1 is In certain embodiments, A1 i
s
[0367] In certain embodiments, A1 is a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7. In certain embodiments, A1 is a 7-10 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7. In certain embodiments, A1 is a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms. In certain embodiments, A1 is a 7-10 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms.
[0368] In certain embodiments, A1 is a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with p occurrences of R7.
[0369] In certain embodiments, A1 is a 6-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with p occurrences of R7. In certain embodiments, A1 is pyridinyl or pyrimidinyl; each of which is substituted with p occurrences of R7. In certain embodiments, A1 is pyridinyl substituted with p occurrences of R7. In certain embodiments, A1 is pyridin-4-yl substituted with p occurrences of R7. In certain embodiments, A1 is pyrimidinyl substituted with p occurrences of R7.
[0370] In certain embodiments, A1 is a 5-membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with p occurrences of R7. In certain embodiments, A1 is a 5-membered monocyclic heteroaryl having 1, 2, or 3 nitrogen atoms; wherein said heteroaryl is substituted with p occurrences of R7. In certain embodiments, A1 is imidazolyl or pyrazolyl; each of which is substituted with p occurrences of R7. In certain embodiments, A1 is
imidazolyl substituted with p occurrences of R7. In certain embodiments, A1 is pyrazolyl substituted with p occurrences of R7.
[0371] In certain embodiments, A1 is selected from the groups depicted in the compounds in Table 3, below.
[0372] As defined generally above, X2 is N, C(CN), or C(R4). In certain embodiments, X2 is N. In certain embodiments, X2 is C(CN) or C(R4). In certain embodiments, X2 is C(CN) or C(H).
In certain embodiments, X2 is C(CN). In certain embodiments, X2 is C(R4). In certain embodiments, X2 is C(H). In certain embodiments, X2 is selected from the groups depicted in the compounds in Table 3, below.
[0373] As defined generally above, L is a covalent bond or a Ci-4 bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or -C(O)-. In certain embodiments, L is a covalent bond.
[0374] In certain embodiments, L is a Ci-4 bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or -C(O)-. In certain embodiments, L is a Ci-4 bivalent saturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or -C(O)-. In certain embodiments, L is a C1-4 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or - C(O)-. In certain embodiments, L is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -N(R9)-, or -C(O)-.
[0375] In certain embodiments, L is a C1-4 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -N(R9)-, or -C(O)-. In certain embodiments, L is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -N(R9)-, or -C(O)-. In certain embodiments, L is a Ci-4 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -O- or -N(R9)-. In certain embodiments, L is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -O- or -N(R9)-.
[0376] In certain embodiments, L is -0-CH2-, -0-(CH2)2-, -N(R9)-CH2-, or -N(R9)-(CH2)2-, wherein the terminal carbon atom of L is attached to R2. In certain embodiments, L is -0-CH2- or -0-(CH2)2-, wherein the terminal carbon atom of L is attached to R2. In certain embodiments, L is -N(R9)-CH2- or -N(R9)-(CH2)2-, wherein the terminal carbon atom of L is attached to R2. In certain embodiments, L is -0-CH2-, wherein the carbon atom of L is attached to R2. In certain embodiments, L is -0-(CH2)2-, wherein the terminal carbon atom of L is attached to R2. In certain embodiments, L is -N(R9)-CH2-, wherein the carbon atom of L is attached to R2. In certain embodiments, L is -N(R9)-(CH2)2-, wherein the terminal carbon atom of L is attached to R2.
[0377] In certain embodiments, L is selected from the groups depicted in the compounds in Table 3, below.
[0378] As defined generally above, Y1 is a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R7)-, or -C(R7)2-.
[0379] In certain embodiments, Y1 is a C2-3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y1 is a C2-3 bivalent unsaturated, straight hydrocarbon chain.
[0380] In certain embodiments, Y1 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y1 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y1 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y1 is a C1-3 bivalent saturated, straight hydrocarbon chain.
[0381] In certain embodiments, Y1 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y1 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y1 is a C2-3 bivalent saturated, straight
hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y1 is a C2-3 bivalent saturated, straight hydrocarbon chain.
[0382] In certain embodiments, Y1 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, -(CH2)3-, -CH2-0- CH2-, or -CH2-CF2-CH2-. In certain embodiments, Y1 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, - (CH2)3-, or -CH2-CF2-CH2-. In certain embodiments, Y1 is -(CH2)2-, -CH2-C(H)(OH)-, -(CH2)3-, or -CH2-CF2-CH2-. In certain embodiments, Y1 is -(CH2)2- or -(CH2)3-. In certain embodiments, Y1 is -CH2-C(H)(OH)- or -CH2-CF2-CH2-. In certain embodiments, Y1 is -(CH2)-, -(CH2)2-, or - (CH2)3-. In certain embodiments, Y1 is -(CH2)2- or -CH2-C(H)(OH)-. In certain embodiments, Y1 is -(CH2)3-, -CH2-0-CH2-, or -CH2-CF2-CH2-.
[0383] In certain embodiments, Y1 is -(CH2)-. In certain embodiments, Y1 is -(CH2)2-. In certain embodiments, Y1 is -CH2-C(H)(OH)-. In certain embodiments, Y1 is -(CH2)3-. In certain embodiments, Y1 is -CH2-0-CH2-. In certain embodiments, Y1 is -CH2-CF2-CH2-. In certain embodiments, Y1 is selected from the groups depicted in the compounds in Table 3, below.
[0384] As defined generally above, Y2 is a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R7)-, or -C(R7)2-.
[0385] In certain embodiments, Y2 is a C2-3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -C(H)(R7)-, or -C(R7)2-.
In certain embodiments, Y2 is a C2-3 bivalent unsaturated, straight hydrocarbon chain.
[0386] In certain embodiments, Y2 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y2 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y2 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y2 is a C1-3 bivalent saturated, straight hydrocarbon chain.
[0387] In certain embodiments, Y2 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y2 is a C2-3 bivalent saturated, straight
hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y2 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y2 is a C2-3 bivalent saturated, straight hydrocarbon chain.
[0388] In certain embodiments, Y2 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, -(CH2)3-, -CH2-0- CH2-, or -CH2-CF2-CH2-. In certain embodiments, Y2 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, - (CH2)3-, or -CH2-CF2-CH2-. In certain embodiments, Y2 is -(CH2)2-, -CH2-C(H)(OH)-, -(CH2)3-, or -CH2-CF2-CH2-. In certain embodiments, Y2 is -(CH2)2- or -(CH2)3-. In certain embodiments, Y2 is -CH2-C(H)(OH)- or -CH2-CF2-CH2-. In certain embodiments, Y2 is -(CH2)-, -(CH2)2-, or - (CH2)3-. In certain embodiments, Y2 is -(CH2)2- or -CH2-C(H)(OH)-. In certain embodiments, Y2 is -(CH2)3-, -CH2-0-CH2-, or -CH2-CF2-CH2-.
[0389] In certain embodiments, Y2 is -(CH2)-. In certain embodiments, Y2 is -(CH2)2-. In certain embodiments, Y2 is -CH2-C(H)(OH)-. In certain embodiments, Y2 is -(CH2)3-. In certain embodiments, Y2 is -CH2-0-CH2-. In certain embodiments, Y2 is -CH2-CF2-CH2-. In certain embodiments, Y2 is selected from the groups depicted in the compounds in Table 3, below.
[0390] As defined generally above, Y3 is a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R7)-, or -C(R7)2-.
[0391] In certain embodiments, Y3 is a C2-3 bivalent unsaturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, -C(H)(R7)-, or -C(R7)2-.
In certain embodiments, Y3 is a C2-3 bivalent unsaturated, straight hydrocarbon chain.
[0392] In certain embodiments, Y3 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y3 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y3 is a C1-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y3 is a C1-3 bivalent saturated, straight hydrocarbon chain.
[0393] In certain embodiments, Y3 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by - 0-, -C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y3 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is optionally replaced by -0-, - C(H)(R7)-, or -C(R7)2-. In certain embodiments, Y3 is a C2-3 bivalent saturated, straight hydrocarbon chain wherein one methylene unit of the chain is replaced by -0-, -C(H)(R7)-, or - C(R7)2-. In certain embodiments, Y3 is a C2-3 bivalent saturated, straight hydrocarbon chain.
[0394] In certain embodiments, Y3 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, -(CH2)3-, -CH2-O- CH2-, or -CH2-CF2-CH2-. In certain embodiments, Y3 is -(CH2)-, -(CH2)2-, -CH2-C(H)(OH)-, - (CH2)3-, or -CH2-CF2-CH2-. In certain embodiments, Y3 is -(CH2)2-, -CH2-C(H)(OH)-, -(CH2)3-, or -CH2-CF2-CH2-. In certain embodiments, Y3 is -(CH2)2- or -(CH2)3-. In certain embodiments, Y3 is -CH2-C(H)(OH)- or -CH2-CF2-CH2-. In certain embodiments, Y3 is -(CH2)-, -(CH2)2-, or - (CH2)3-. In certain embodiments, Y3 is -(CH2)2- or -CH2-C(H)(OH)-. In certain embodiments, Y3 is -(CH2)3-, -CH2-O-CH2-, or -CH2-CF2-CH2-.
[0395] In certain embodiments, Y3 is -(CH2)-. In certain embodiments, Y3 is -(CH2)2-. In certain embodiments, Y3 is -CH2-C(H)(OH)-. In certain embodiments, Y3 is -(CH2)3-. In certain embodiments, Y3 is -CH2-O-CH2-. In certain embodiments, Y3 is -CH2-CF2-CH2-. In certain embodiments, Y3 is selected from the groups depicted in the compounds in Table 3, below.
[0396] As defined generally above, m is 0, 1, 2, or 3. In certain embodiments, m is 0. In certain embodiments, m is 1. In certain embodiments, m is 2. In certain embodiments, m is 3. In certain embodiments, m is 0 or 1. In certain embodiments, m is 1 or 2. In certain embodiments, m is 2 or 3. In certain embodiments, m is 0, 1, or 2. In certain embodiments, m is 1, 2, or 3. In certain embodiments, m is selected from the values represented in the compounds in Table 3, below.
[0397] As defined generally above, n is 0, 1, 2, or 3. In certain embodiments, n is 0. In certain embodiments, n is 1. In certain embodiments, n is 2. In certain embodiments, n is 3. In certain embodiments, n is 0 or 1. In certain embodiments, n is 1 or 2. In certain embodiments, n is 2 or 3. In certain embodiments, n is 0, 1, or 2. In certain embodiments, n is 1, 2, or 3. In certain embodiments, n is selected from the values represented in the compounds in Table 3, below.
[0398] As defined generally above, p is 0, 1, 2, or 3. In certain embodiments, p is 0. In certain embodiments, p is 1. In certain embodiments, p is 2. In certain embodiments, p is 3. In certain embodiments, p is 0 or 1. In certain embodiments, p is 1 or 2. In certain embodiments, p is 2 or 3. In certain embodiments, p is 0, 1, or 2. In certain embodiments, p is 1, 2, or 3. In certain embodiments, p is selected from the values represented in the compounds in Table 3, below.
[0399] The description above describes multiple embodiments relating to compounds of Formula III. The patent application specifically contemplates all combinations of the embodiments.
[0400] In certain embodiments, the compound is a compound in Table 1, 1-A, 2, or 3 below, or a pharmaceutically acceptable salt thereof. In certain embodiments, the compound is a compound in Table 1, 1-A, 2, or 3 below. In certain embodiments, the compound is a compound in Table 1, 2, or 3 below, or a pharmaceutically acceptable salt thereof. In certain embodiments, the compound is a compound in Table 1, 2, or 3 below. In certain embodiments, the compound is a compound in Table 1 or 2 below, or a pharmaceutically acceptable salt thereof. In certain embodiments, the compound is a compound in Table 1 or 2 below. In certain embodiments, the compound is a compound in Table 1 or 1-A below, or a pharmaceutically acceptable salt thereof. In certain embodiments, the compound is a compound in Table 1 or 1-A below.
[0401] In certain embodiments, the compound is a compound in Table 1 below, or a pharmaceutically acceptable salt thereof. In certain embodiments, the compound is a compound in Table 1 below. In certain embodiments, the compound is a compound in Table 1-A below, or a pharmaceutically acceptable salt thereof. In certain embodiments, the compound is a compound in Table 1-A below. In certain embodiments, the compound is a compound in Table 2 below, or a pharmaceutically acceptable salt thereof. In certain embodiments, the compound is a compound in Table 2 below. In certain embodiments, the compound is a compound in Table 3 below, or a pharmaceutically acceptable salt thereof. In certain embodiments, the compound is a compound in Table 3 below.
TABLE 1.
[0402] Methods for preparing compounds described herein are illustrated in the following synthetic Schemes and in the Examples, below. The Schemes are given for the purpose of illustrating the invention, and are not intended to limit the scope or spirit of the invention.
Starting materials shown in the Schemes can be obtained from commercial sources or can be prepared based on procedures described in the literature.
[0403] In the Schemes, it is understood by one skilled in the art of organic synthesis that the functionality present on various portions of the molecule should be compatible with the reagents and reactions proposed. Substituents not compatible with the reaction conditions will be apparent to one skilled in the art, and alternate methods are therefore indicated (for example, use of protecting groups or alternative reactions). Protecting group chemistry and strategy is well known in the art, for example, as described in detail in “Protecting Groups in Organic Synthesis”, T. W. Greene and P. G. M. Wuts, 3rd edition, John Wiley & Sons, 1999, the entire contents of which are hereby incorporated by reference.
[0404] The modular synthetic routes illustrated in Schemes 1, 2, and 3 can also be readily modified by one of skill in the art to provide additional naphthyl-substituted quinolin-4( 1 H)-oncs and related compounds by conducting functional group transformations on the intermediate and final compounds. Such functional group transformations are well known in the art, as described in, for example, “Comprehensive Organic Synthesis” (B.M. Trost & I. Fleming, eds., 1991- 1992).
[0405] Schemes 4 and 5 depict methods that may be used for preparing specific compounds described herein, based on methods described in Schemes 1-3 above, and in the Examples below.
For example, Scheme 4 depicts methods that may be used to prepare compounds 1-5 and 1-8. In addition, compound 1-26 may be prepared according to the methods depicted for compound 1-8, but using tert - butyl azetidin-3-ylcarbamate instead of tert -butyl 3,8-diazabicyclo[3.2.1]octane-8- carboxylate in the first step. Additionally, compound 1-23 may be prepared according to the methods depicted for compound 1-5, but using 4-bromo-2-(methoxymethoxy)quinoline instead of the substituted naphthyl triflate in the Pd coupling step.
[0406] Scheme 5 depicts methods that may be used to prepare compounds II-3 and II-4. In addition, compounds II-9 and II- 10 may be prepared according to the methods depicted for compounds II-3 and II-4, respectively, but using tert-butyl 1,4-diazepane-l-carboxylate or tert- butyl azetidin-3-ylcarbamate instead of tert-butyl 3,8-diazabicyclo[3.2.1]octane-8-carboxylate in the first step.
SCHEME 5.
II. Therapeutic Applications of Naphthyl-substituted Quinolin-4(1H )-ones and Related Compounds
[0407] It is contemplated that the naphthyl-substituted quinolin-4(1 H)-oncs and related compounds described herein, such as a compound of Formula I, II, or III, or other compounds in Section I, provide therapeutic benefits to subjects suffering from cancer and other disorders. Accordingly, one aspect of the invention provides a method of treating a disorder mediated by K- Ras in a subject. The method comprises administering a therapeutically effective amount of a compound described herein, such as a compound of Formula I, II, or III, to a subject in need thereof to treat the disorder. Another aspect of the invention provides a method of treating a disorder mediated by K-Ras G12D in a subject. The method comprises administering a therapeutically effective amount of a compound described herein, such as a compound of
Formula I, II, or III, to a subject in need thereof to treat the disorder. In certain embodiments, the compound is a compound of Formula I, II, or III defined by one of the embodiments described above.
[0408] Kirsten Rat Sarcoma 2 Viral Oncogene Homolog (“K-Ras”) is a small GTPase and a member of the Ras family of oncogenes. K-Ras has been reported to serve as a molecular switch cycling between inactive (GDP-bound) and active (GTP-bound) states to transduce upstream cellular signals received from multiple tyrosine kinases to downstream effectors to regulate a wide variety of processes, including cellular proliferation ( e.g ., see Alamgeer et al., (2013) Current Opin Pharmcol.13:394-401).
[0409] The role of activated K-Ras in malignancy was observed over thirty years ago (e.g., see Santos et al., (1984) Science 223:661-664). Aberrant expression of K-Ras has been reported to account for up to 20% of all cancers, and oncogenic K-Ras mutations that stabilize GTP binding and lead to constitutive activation of K-Ras and downstream signaling have been reported in 25 - 30% of lung adenocarcinomas (e.g., see Samatar and Poulikakos (2014) Nat Rev Drug Disc 13(12): 928- 942 doi: 10.1038/nrd428). Single nucleotide substitutions that result in missense mutations at codons 12 and 13 of the K-Ras primary amino acid sequence have been reported to comprise approximately 40% of these K-Ras driver mutations in lung adenocarcinoma. K-Ras G12D mutation has been reported to be present in 25.0% of all pancreatic ductal adenocarcinoma patients, 13.3% of all colorectal carcinoma patients, 10.1% of all rectal carcinoma patients, 4.1% of all non- small cell lung carcinoma patients, and 1.7% of all small cell lung carcinoma patients (e.g., see The AACR Project GENIE Consortium, (2017) Cancer Discovery;7(8):818-831. Dataset Version 4).
[0410] Despite efforts to develop inhibitors of K-Ras for treating cancer, through year 2020, no K-Ras inhibitor had demonstrated sufficient safety and/or efficacy to obtain regulatory approval from the U.S. FDA (e.g., see McCormick (2015) Clin Cancer Res.21 (8): 1797-1801).
Compounds that inhibit K-Ras activity are under investigation, including those that disrupt effectors such as guanine nucleotide exchange factors (e.g., see Sun et al., (2012) Angew Chem Int Ed Engl.51(25):6140-6143 doi: 10.1002/anie201201358) as well recent advances in the covalent targeting of an allosteric pocket of K-Ras G12C (e.g., see Ostrem et al., (2013) Nature 503:548-551 and Fell et al., (2018) ACS Med. Chem. Lett.9: 1230- 1234).
[0411] Accordingly, the provided compounds are inhibitors of K-Ras and are therefore useful for treating one or more disorders associated with activity of K-Ras. Thus, in certain aspects and embodiments, the present invention provides a method for treating a K-Ras -mediated disorder comprising the step of administering to a patient in need thereof a therapeutically effective compound of the present invention, or pharmaceutically acceptable composition thereof. As used herein, the term “K-Ras-mediated” disorders, diseases, and/or conditions means any disease or other deleterious condition in which K-Ras, or a variant or mutant thereof, is known to play a role. Accordingly, another aspect or embodiment of the present invention relates to treating or lessening the severity of one or more diseases in which K-Ras is known to play a role.
[0412] In some aspects and embodiments, provided herein are methods of treating, reducing the severity of, delaying the onset of, or inhibiting the progress of a disease or disorder, or one or more symptoms thereof, of a disease or disorder characterized by or associated with increased K- Ras expression and/or increased K-Ras activity, comprising the step of administering to a patient in need thereof a therapeutically effective amount of a compound of the present invention, or pharmaceutically acceptable composition thereof. In some aspects and embodiments, provided herein are methods of treating, reducing the severity of, delaying the onset of, or inhibiting the progress of a disease or disorder, or one or more symptoms thereof of a disease or disorder in which inhibition or antagonizing of K-Ras activity is beneficial, comprising the step of administering to a patient in need thereof a therapeutically effective amount of a compound of the present invention, or pharmaceutically acceptable composition thereof.
[0413] Accordingly, in some aspects and embodiments, the present invention provides a method for treating one or more disorders, diseases, and/or conditions wherein the disorder, disease, or condition includes, but is not limited to, a cellular proliferative disorder, comprising administering to a patient in need thereof, a K-Ras inhibitor compound as described herein, or a pharmaceutical salt or composition thereof. In some embodiments, the cellular proliferative disorder is cancer. In some embodiments, the cancer is characterized by increased K-Ras expression and/or increased K-Ras activity, i.e., “increased activated K-Ras.”
[0414] In some embodiments, the K-Ras is a mutant K-Ras. In some embodiments, the K- Ras is an activated mutant K-Ras. In some embodiments, the K-Ras is a G12 mutant K-Ras. In some embodiments, the K-Ras is K-Ras G12D, K-Ras G12V, or K-Ras G12C. In some
embodiments, the K-Ras is K-Ras G12D. In some embodiments, the K-Ras is K-Ras G12V. In some embodiments, the K-Ras is K-Ras G12C.
[0415] Accordingly, the provided compounds are inhibitors of K-Ras G12D and are therefore useful for treating one or more disorders associated with activity of K-Ras G12D. Thus, in certain aspects and embodiments, the present invention provides a method for treating a K-Ras G12D-mediated disorder comprising the step of administering to a patient in need thereof a therapeutically effective compound of the present invention, or pharmaceutically acceptable composition thereof. As used herein, the term “K-Ras G12D-mediated” disorders, diseases, and/or conditions means any disease or other deleterious condition in which K-Ras G12D is known to play a role. Accordingly, another aspect or embodiment of the present invention relates to treating or lessening the severity of one or more diseases in which K-Ras G12D is known to play a role.
[0416] In some aspects and embodiments, provided herein are methods of treating, reducing the severity of, delaying the onset of, or inhibiting the progress of a disease or disorder, or one or more symptoms thereof, of a disease or disorder characterized by or associated with increased K- Ras G12D expression and/or increased K-Ras G12D activity, comprising the step of administering to a patient in need thereof a therapeutically effective amount of a compound of the present invention, or pharmaceutically acceptable composition thereof. In some aspects and embodiments, provided herein are methods of treating, reducing the severity of, delaying the onset of, or inhibiting the progress of a disease or disorder, or one or more symptoms thereof of a disease or disorder in which inhibition or antagonizing of K-Ras G12D activity is beneficial, comprising the step of administering to a patient in need thereof a therapeutically effective amount of a compound of the present invention, or pharmaceutically acceptable composition thereof.
[0417] Accordingly, in some aspects and embodiments, the present invention provides a method for treating one or more disorders, diseases, and/or conditions wherein the disorder, disease, or condition includes, but is not limited to, a cellular proliferative disorder, comprising administering to a patient in need thereof, an K-Ras G12D inhibitor compound as described herein, or a pharmaceutical salt or composition thereof. In some embodiments, the cellular proliferative disorder is cancer. In some embodiments, the cancer is characterized by increased
K-Ras G12D expression and/or increased K-Ras G12D activity, i.e., “increased activated K-Ras G12D.”
[0418] As used herein, the terms "increased," "elevated," or "enhanced," are used interchangeably and encompass any measurable increase in a biological function and/or biological activity and/or a concentration. For example, an increase can be by at least about 10%, about 15%, about 20%, about 25%, about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 96%, about 97%, about 98%, about 99%, about 100%, about 2-fold, about 3- fold, about 4-fold, about 5-fold, about 6-fold, about 7-fold, about 8-fold, about 9-fold, about 10- fold, about 20-fold, about 25-fold, about 50-fold, about 100-fold, or higher, relative to a control or baseline amount of a function, or activity, or concentration.
[0419] As used herein, the terms "increased expression" and/or "increased activity" of a substance, such as K-Ras G12D, in a sample or cancer or patient, refers to an increase in the amount of the substance, such as K-Ras G12D, of about 5%, about 10%, about 15%, about 20%, about 25%, about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 96%, about 97%, about 98%, about 99%, about 100%, about 2-fold, about 3-fold, about 4-fold, about 5-fold, about 6-fold, about 7-fold, about 8-fold, about 9-fold, about 10-fold, about 20-fold, about 25-fold, about 50-fold, about 100-fold, or higher, relative to the amount of the substance, such as K-Ras G12D, in a control sample or control samples, such as an individual or group of individuals who are not suffering from the disease or disorder ( e.g ., cancer) or an internal control, as determined by techniques known in the art. A subject can also be determined to have an "increased expression" or "increased activity" of a substance, such as K-Ras G12D, if the expression and/or activity of the substance, such as K-Ras G12D, is increased by one standard deviation, two standard deviations, three standard deviations, four standard deviations, five standard deviations, or more, relative to the mean (average) or median amount of the substance, such as K-Ras G12D, in a control group of samples or a baseline group of samples or a retrospective analysis of patient samples. As practiced in the art, such control or baseline expression levels can be previously determined, or measured prior to the measurement in the sample or cancer or subject, or can be obtained from a database of such control samples.
[0420] As used herein, a "proliferative disease" refers to a disease that occurs due to abnormal growth or extension by the multiplication of cells (Walker, Cambridge Dictionary of Biology, Cambridge University Press: Cambridge, UK, 1990). A proliferative disease can be associated with: 1) the pathological proliferation of normally quiescent cells; 2) the pathological migration of cells from their normal location ( e.g ., metastasis of neoplastic cells); 3) the pathological expression of proteolytic enzymes, such as the matrix metalloproteinases (e.g., collagenases, gelatinases, and elastases); or 4) the pathological angiogenesis, as in proliferative retinopathy and tumor metastasis. Exemplary proliferative diseases include cancers (i.e., "malignant neoplasms"), benign neoplasms, angiogenesis, inflammatory diseases, and autoimmune diseases. Accordingly, in certain aspects and embodiments, the disorder is cancer. The cancer or proliferative disorder or tumor to be treated using the compounds and methods and uses described herein include, but are not limited to, a hematological cancer, a lymphoma, a myeloma, a leukemia, a neurological cancer, skin cancer, breast cancer, a prostate cancer, a colorectal cancer, lung cancer, head and neck cancer, a gastrointestinal cancer, a liver cancer, a pancreatic cancer, a genitourinary cancer, a bone cancer, renal cancer, and a vascular cancer.
[0421] In some embodiments of the methods and uses described herein, a cancer is treated by inhibiting or reducing or decreasing or arresting further growth or spread of the cancer or tumor. In some embodiments of the methods and uses described herein, a cancer is treated by inhibiting or reducing the size (e.g., volume or mass) of the cancer or tumor by at least 5%, at least 10%, at least 25%, at least 50%, at least 75%, at least 90% or at least 99% relative to the size of the cancer or tumor prior to treatment. In some embodiments of the methods and uses described herein, a cancer is treated by reducing the quantity of the cancers or tumors in the patient by at least 5%, at least 10%, at least 25%, at least 50%, at least 75%, at least 90% or at least 99% relative to the quantity of the cancers or tumors prior to treatment.
[0422] In some embodiments, the cancer is selected from is non-small cell lung cancer (NSCLC), small cell lung cancer, colorectal cancer, rectal cancer, and pancreatic cancer. In some embodiments, the cancer is selected from non-small cell lung cancer (NSCLC), pancreatic cancer, and colorectal cancer. In some embodiments, the cancer is selected from non-small cell lung cancer (NSCLC) and pancreatic cancer.
[0423] In certain embodiments, the cancer is a solid tumor. In certain embodiments, the cancer is a melanoma, carcinoma, or blastoma. In certain embodiments, the cancer is a melanoma. In certain embodiments, the cancer is a carcinoma. In certain embodiments, the cancer is an adenocarcinoma. In certain embodiments, the cancer is a blastoma.
[0424] In certain embodiments, the cancer is lung cancer, pancreatic cancer, colorectal cancer, breast cancer, cervical cancer, prostate cancer, gastric cancer, skin cancer, liver cancer, bile duct cancer, or nervous system cancer. In certain embodiments, the cancer is lung cancer. In certain embodiments, the cancer is pancreatic cancer. In certain embodiments, the cancer is colorectal cancer. In certain embodiments, the cancer is breast cancer. In certain embodiments, the cancer is cervical cancer. In certain embodiments, the cancer is prostate cancer. In certain embodiments, the cancer is gastric cancer. In certain embodiments, the cancer is skin cancer. In certain embodiments, the cancer is liver cancer. In certain embodiments, the cancer is bile duct cancer. In certain embodiments, the cancer is nervous system cancer.
[0425] In certain embodiments, the cancer is breast adenocarcinoma, lung adenocarcinoma, pancreatic adenocarcinoma, cervical adenocarcinoma, colorectal adenocarcinoma, prostate adenocarcinoma, gastric adenocarcinoma, melanoma, lung squamous cell carcinoma, hepatocellular carcinoma, cholangiocarcinoma, glioblastoma, or neuroblastoma. In certain embodiments, the cancer is breast adenocarcinoma. In certain embodiments, the cancer is lung adenocarcinoma. In certain embodiments, the cancer is pancreatic adenocarcinoma. In certain embodiments, the cancer is cervical adenocarcinoma. In certain embodiments, the cancer is prostate adenocarcinoma. In certain embodiments, the cancer is gastric adenocarcinoma.
[0426] In certain embodiments, the cancer is melanoma.
[0427] In certain embodiments, the cancer is lung squamous cell carcinoma, hepatocellular carcinoma, or cholangiocarcinoma. In certain embodiments, the cancer is lung squamous cell carcinoma. In certain embodiments, the cancer is hepatocellular carcinoma. In certain embodiments, the cancer is cholangiocarcinoma.
[0428] In certain embodiments, the cancer is glioblastoma or neuroblastoma. In certain embodiments, the cancer is glioblastoma. In certain embodiments, the cancer is neuroblastoma.
[0429] In certain embodiments, the cancer is lung cancer, pancreatic cancer, or colorectal cancer. In certain embodiments, the cancer is non-small cell lung cancer, pancreatic cancer, or colorectal cancer. In certain embodiments, the cancer is lung cancer. In certain embodiments, the cancer is non-small cell lung cancer.
[0430] In certain embodiments, the cancer has elevated K-Ras activity. In certain embodiments, the cancer overexpresses K-Ras. In some embodiments, a subject having a disorder associated with or mediated by K-Ras has been identified or diagnosed as having a cancer having a K-Ras mutation (e.g., as determined using a regulatory agency-approved, e.g., FDA-approved, assay or kit). In some embodiments, the subject has a tumor that is positive for a K-Ras mutation (e.g., as determined using a regulatory agency-approved assay or kit). The subject can be a subject who has one or more tumor that is positive for a K-Ras mutation (e.g., identified as positive using a regulatory agency-approved, e.g., FDA-approved, assay or kit). The subject can be a subject whose tumor(s) have a K-Ras mutation (e.g., where the tumor is identified as such using a regulatory agency-approved, e.g., FDA-approved, kit or assay). In some embodiments, the subject is suspected of having a K-Ras gene-associated cancer. In some embodiments, the subject has a clinical record indicating that the subject has a tumor that has a K-Ras mutation (and, for example, the clinical record indicates that the subject should be treated with any of the compounds or compositions provided herein).
[0431] In some embodiments of any of the methods or uses described herein, an assay is used to determine whether the patient has a K-Ras mutation using a sample (e.g., a biological sample or a biopsy sample (e.g., a paraffin-embedded biopsy sample) from a patient (e.g., a patient suspected of having a K-Ras-associated cancer, a patient having one or more symptoms of a K- Ras-associated cancer, and/or a patient that has an increased risk of developing a K-Ras- associated cancer). Non-limiting examples of such assays that can be used to determine whether a patient or subject has a K-Ras mutation include next generation sequencing (NGS), immunohistochemistry, fluorescence microscopy, break apart FISH analysis, Southern blotting, Western blotting, FACS analysis, Northern blotting, and PCR-based amplification (e.g., RT-PCR and quantitative real-time RT-PCR). As is well-known in the art, the assays are typically performed, e.g., with at least one labelled nucleic acid probe or at least one labelled antibody or antigen-binding fragment thereof. The term “regulatory agency” is a country’s agency for the
approval of the medical use of pharmaceutical agents with the country. For example, a non limiting example of a regulatory agency is the U.S. Food and Drug Administration (FDA).
[0432] In some embodiments, the K-Ras mutation is a G12 mutation. In some embodiments, the K-Ras mutation is G12D, G12V, or G12C. In some embodiments, the K-Ras mutation is G12D. In some embodiments, the K-Ras mutation is G12V. In some embodiments, the K-Ras mutation is G12C.
[0433] In certain embodiments, the cancer has elevated K-Ras G12D activity. In certain embodiments, the cancer overexpresses K-Ras G12D. In some embodiments, a subject having a disorder associated with or mediated by K-Ras G12D has been identified or diagnosed as having a cancer having a K-Ras G12D mutation ( e.g ., as determined using a regulatory agency- approved, e.g., FDA-approved, assay or kit). In some embodiments, the subject has a tumor that is positive for a K-Ras G12D mutation (e.g., as determined using a regulatory agency-approved assay or kit). The subject can be a subject who has one or more tumor that is positive for a K-Ras G12D mutation (e.g., identified as positive using a regulatory agency-approved, e.g., FDA- approved, assay or kit). The subject can be a subject whose tumor(s) have a K-Ras G12D mutation (e.g., where the tumor is identified as such using a regulatory agency-approved, e.g., FDA-approved, kit or assay). In some embodiments, the subject is suspected of having a K-Ras G12D gene-associated cancer. In some embodiments, the subject has a clinical record indicating that the subject has a tumor that has a K-Ras G12D mutation (and, for example, the clinical record indicates that the subject should be treated with any of the compounds or compositions provided herein).
[0434] In some embodiments of any of the methods or uses described herein, an assay is used to determine whether the patient has a K-Ras G12D mutation using a sample (e.g., a biological sample or a biopsy sample (e.g., a paraffin-embedded biopsy sample) from a patient (e.g., a patient suspected of having a K-Ras G12D-associated cancer, a patient having one or more symptoms of a K-Ras G12D-associated cancer, and/or a patient that has an increased risk of developing a K-Ras G12D-associated cancer). Non-limiting examples of such assays that can be used to determine whether a patient or subject has a K-Ras G12D mutation include next generation sequencing (NGS), immunohistochemistry, fluorescence microscopy, break apart FISH analysis, Southern blotting, Western blotting, FACS analysis, Northern blotting, and PCR-
based amplification ( e.g ., RT-PCR and quantitative real-time RT-PCR). As is well-known in the art, the assays are typically performed, e.g., with at least one labelled nucleic acid probe or at least one labelled antibody or antigen-binding fragment thereof. The term “regulatory agency” is a country’s agency for the approval of the medical use of pharmaceutical agents with the country. For example, a non-limiting example of a regulatory agency is the U.S. Food and Drug Administration (FDA).
[0435] In certain embodiments, the cancer is a leukemia (e.g., acute leukemia, acute lymphocytic leukemia, acute myelocytic leukemia, acute myeloblastic leukemia, acute promyelocytic leukemia, acute myelomonocytic leukemia, acute monocytic leukemia, acute erythroleukemia, chronic leukemia, chronic myelocytic leukemia, chronic lymphocytic leukemia), polycythemia vera, lymphoma (e.g., Hodgkin’s disease or non-Hodgkin’s disease), Waldenstrom's macroglobulinemia, multiple myeloma, heavy chain disease, or a solid tumor such as a sarcoma or carcinoma (e.g., fibrosarcoma, myxosarcoma, liposarcoma, chondrosarcoma, osteogenic sarcoma, chordoma, angiosarcoma, endotheliosarcoma, lymphangio sarcoma, lymphangioendotheliosarcoma, synovioma, mesothelioma, Ewing’s tumor, leiomyosarcoma, rhabdomyosarcoma, colon carcinoma, pancreatic cancer, breast cancer, ovarian cancer, prostate cancer, squamous cell carcinoma, basal cell carcinoma, adenocarcinoma, sweat gland carcinoma, sebaceous gland carcinoma, papillary carcinoma, papillary adenocarcinomas, cystadenocarcinoma, medullary carcinoma, bronchogenic carcinoma, renal cell carcinoma, hepatoma, bile duct carcinoma, choriocarcinoma, seminoma, embryonal carcinoma, Wilm's tumor, cervical cancer, uterine cancer, testicular cancer, lung carcinoma, small cell lung carcinoma, bladder carcinoma, epithelial carcinoma, glioma, astrocytoma, glioblastoma multiforme (GBM, also known as glioblastoma), medulloblastoma, craniopharyngioma, ependymoma, pinealoma, hemangioblastoma, acoustic neuroma, oligodendroglioma, schwannoma, neurofibrosarcoma, meningioma, melanoma, neuroblastoma, and retinoblastoma).
[0436] In some embodiments, the cancer is glioma, astrocytoma, glioblastoma multiforme (GBM, also known as glioblastoma), medulloblastoma, craniopharyngioma, ependymoma, pinealoma, hemangioblastoma, acoustic neuroma, oligodendroglioma, schwannoma, neurofibrosarcoma, meningioma, melanoma, neuroblastoma, or retinoblastoma.
[0437] In some embodiments, the cancer is acoustic neuroma, astrocytoma ( e.g . Grade I - Pilocytic Astrocytoma, Grade II - Low-grade Astrocytoma, Grade III - Anaplastic Astrocytoma, or Grade IV - Glioblastoma (GBM)), chordoma, CNS lymphoma, craniopharyngioma, brain stem glioma, ependymoma, mixed glioma, optic nerve glioma, subependymoma, medulloblastoma, meningioma, metastatic brain tumor, oligodendroglioma, pituitary tumors, primitive neuroectodermal (PNET) tumor, or schwannoma. In some embodiments, the cancer is a type found more commonly in children than adults, such as brain stem glioma, craniopharyngioma, ependymoma, juvenile pilocytic astrocytoma (JPA), medulloblastoma, optic nerve glioma, pineal tumor, primitive neuroectodermal tumors (PNET), or rhabdoid tumor.
[0438] In some embodiments, the cancer is mesothelioma, hepatobilliary (hepatic and billiary duct), bone cancer, pancreatic cancer, skin cancer, cancer of the head or neck, cutaneous or intraocular melanoma, ovarian cancer, colon cancer, rectal cancer, cancer of the anal region, stomach cancer, gastrointestinal (gastric, colorectal, and duodenal), uterine cancer, carcinoma of the fallopian tubes, carcinoma of the endometrium, carcinoma of the cervix, carcinoma of the vagina, carcinoma of the vulva, Hodgkin’s Disease, cancer of the esophagus, cancer of the small intestine, cancer of the endocrine system, cancer of the thyroid gland, cancer of the parathyroid gland, cancer of the adrenal gland, sarcoma of soft tissue, cancer of the urethra, cancer of the penis, prostate cancer, testicular cancer, chronic or acute leukemia, chronic myeloid leukemia, lymphocytic lymphomas, cancer of the bladder, cancer of the kidney or ureter, renal cell carcinoma, carcinoma of the renal pelvis, non-Hodgkins’ s lymphoma, spinal axis tumors, brain stem glioma, pituitary adenoma, adrenocortical cancer, gall bladder cancer, multiple myeloma, cholangiocarcinoma, fibrosarcoma, neuroblastoma, retinoblastoma, or a combination of one or more of the foregoing cancers.
[0439] In some embodiments, the cancer is hepatocellular carcinoma, ovarian cancer, ovarian epithelial cancer, fallopian tube cancer, papillary serous cystadenocarcinoma, uterine papillary serous carcinoma (UPSC), prostate cancer, testicular cancer, gallbladder cancer, hepatocholangiocarcinoma, soft tissue and bone synovial sarcoma, rhabdomyosarcoma, osteosarcoma, chondrosarcoma, Ewing sarcoma, anaplastic thyroid cancer, adrenocortical adenoma, pancreatic cancer, pancreatic ductal carcinoma, pancreatic adenocarcinoma, gastrointestinal/stomach (GIST) cancer, lymphoma, squamous cell carcinoma of the head and neck (SCCHN), salivary gland cancer, glioma, or brain cancer, neurofibromatosis- 1 associated
malignant peripheral nerve sheath tumors (MPNST), Waldenstrom’s macroglobulinemia, or medulloblastoma.
[0440] In some embodiments, the cancer is hepatocellular carcinoma (HCC), hepatoblastoma, colon cancer, rectal cancer, ovarian cancer, ovarian epithelial cancer, fallopian tube cancer, papillary serous cystadenocarcinoma, uterine papillary serous carcinoma (UPSC), hepatocholangiocarcinoma, soft tissue and bone synovial sarcoma, rhabdomyosarcoma, osteosarcoma, anaplastic thyroid cancer, adrenocortical adenoma, pancreatic cancer, pancreatic ductal carcinoma, pancreatic adenocarcinoma, glioma, neurofibromatosis- 1 associated malignant peripheral nerve sheath tumors (MPNST), Waldenstrom’s macroglobulinemia, or medulloblastoma.
[0441] In some embodiments, the cancer is selected from renal cell carcinoma, or kidney cancer; hepatocellular carcinoma (HCC) or hepatoblastoma, or liver cancer; melanoma; breast cancer; colorectal carcinoma, or colorectal cancer; colon cancer; rectal cancer; anal cancer; lung cancer, such as non-small cell lung cancer (NSCLC) or small cell lung cancer (SCLC); ovarian cancer, ovarian epithelial cancer, ovarian carcinoma, or fallopian tube cancer; papillary serous cystadenocarcinoma or uterine papillary serous carcinoma (UPSC); prostate cancer; testicular cancer; gallbladder cancer; hepatocholangiocarcinoma; soft tissue and bone synovial sarcoma; rhabdomyosarcoma; osteosarcoma; chondrosarcoma; Ewing sarcoma; anaplastic thyroid cancer; adrenocortical carcinoma; pancreatic cancer; pancreatic ductal carcinoma or pancreatic adenocarcinoma; gastrointestinal/stomach (GIST) cancer; lymphoma; squamous cell carcinoma of the head and neck (SCCHN); salivary gland cancer; glioma, or brain cancer; neurofibromatosis- 1 associated malignant peripheral nerve sheath tumors (MPNST); Waldenstrom’s macroglobulinemia; and medulloblastoma.
[0442] In some embodiments, the cancer is renal cell carcinoma, hepatocellular carcinoma (HCC), hepatoblastoma, colorectal carcinoma, colorectal cancer, colon cancer, rectal cancer, anal cancer, ovarian cancer, ovarian epithelial cancer, ovarian carcinoma, fallopian tube cancer, papillary serous cystadenocarcinoma, uterine papillary serous carcinoma (UPSC), hepatocholangiocarcinoma, soft tissue and bone synovial sarcoma, rhabdomyosarcoma, osteosarcoma, chondrosarcoma, anaplastic thyroid cancer, adrenocortical carcinoma, pancreatic cancer, pancreatic ductal carcinoma, pancreatic adenocarcinoma, glioma, brain cancer,
neurofibromatosis- 1 associated malignant peripheral nerve sheath tumors (MPNST), Waldenstrom’s macroglobulinemia, or medulloblastoma.
[0443] In some embodiments, the cancer is hepatocellular carcinoma (HCC), hepatoblastoma, colon cancer, rectal cancer, ovarian cancer, ovarian epithelial cancer, ovarian carcinoma, fallopian tube cancer, papillary serous cystadenocarcinoma, uterine papillary serous carcinoma (UPSC), hepatocholangiocarcinoma, soft tissue and bone synovial sarcoma, rhabdomyosarcoma, osteosarcoma, anaplastic thyroid cancer, adrenocortical carcinoma, pancreatic cancer, pancreatic ductal carcinoma, pancreatic adenocarcinoma, glioma, neurofibromatosis- 1 associated malignant peripheral nerve sheath tumors (MPNST), Waldenstrom’s macroglobulinemia, or medulloblastoma.
[0444] In some embodiments, the cancer is hepatocellular carcinoma (HCC). In some embodiments, the cancer is hepatoblastoma. In some embodiments, the cancer is colon cancer.
In some embodiments, the cancer is rectal cancer. In some embodiments, the cancer is ovarian cancer, or ovarian carcinoma. In some embodiments, the cancer is ovarian epithelial cancer. In some embodiments, the cancer is fallopian tube cancer. In some embodiments, the cancer is papillary serous cystadenocarcinoma. In some embodiments, the cancer is uterine papillary serous carcinoma (UPSC). In some embodiments, the cancer is hepatocholangiocarcinoma. In some embodiments, the cancer is soft tissue and bone synovial sarcoma. In some embodiments, the cancer is rhabdomyosarcoma. In some embodiments, the cancer is osteosarcoma. In some embodiments, the cancer is anaplastic thyroid cancer. In some embodiments, the cancer is adrenocortical carcinoma. In some embodiments, the cancer is pancreatic cancer, or pancreatic ductal carcinoma. In some embodiments, the cancer is pancreatic adenocarcinoma. In some embodiments, the cancer is glioma. In some embodiments, the cancer is malignant peripheral nerve sheath tumors (MPNST). In some embodiments, the cancer is neurofibromatosis- 1 associated MPNST. In some embodiments, the cancer is Waldenstrom’s macroglobulinemia. In some embodiments, the cancer is medulloblastoma.
[0445] In certain embodiments, the subject is a human. In certain embodiments, the subject is an adult human. In certain embodiments, the subject is a pediatric human. In certain embodiments, the subject is a companion animal. In certain embodiments, the subject is a canine, feline, or equine.
[0446] Another aspect of the invention provides for the use of a compound described herein (such as a compound of Formula I, II, or III, or other compounds in Section I) in the manufacture of a medicament. In certain embodiments, the medicament is for treating a disorder described herein, such as cancer.
[0447] Another aspect of the invention provides for the use of a compound described herein (such as a compound of Formula I, II, or III, or other compounds in Section I) for treating a medical disorder, such as a medical disorder described herein (for example, cancer).
[0448] Further, it is contemplated that the compounds described herein, such as a compound of Formula I, II, or III, or other compounds in Section I, can inhibit K-Ras activity. Accordingly, another aspect of the invention provides a method of inhibiting K-Ras activity. The method comprises contacting K-Ras with an effective amount of a naphthyl-substituted quinolin-4( 1H)- one or related compound described herein, such as a compound of Formula I, II, or III, or other compounds in Section I, to inhibit K-Ras activity. In certain embodiments, the compound is a compound of Formula I, II, or III defined by one of the embodiments described above.
[0449] In some embodiments, the K-Ras is a mutant K-Ras. In some embodiments, the K-Ras is an activated mutant K-Ras. In some embodiments, the K-Ras is a G 12 mutant K-Ras. In some embodiments, the K-Ras is K-Ras G12D, K-Ras G12V, or K-Ras G12C. In some embodiments, the K-Ras is K-Ras G12D. In some embodiments, the K-Ras is K-Ras G12V. In some embodiments, the K-Ras is K-Ras G12C.
[0450] Further, it is contemplated that the compounds described herein, such as a compound of Formula I, II, or III, or other compounds in Section I, can inhibit K-Ras G12D activity. Accordingly, another aspect of the invention provides a method of inhibiting K-Ras G12D activity. The method comprises contacting K-Ras G12D with an effective amount of a naphthyl- substituted quinolin-4( 1H)-onc or related compound described herein, such as a compound of Formula I, II, or III, or other compounds in Section I, to inhibit K-Ras G12D activity. In certain embodiments, the compound is a compound of Formula I, II, or III defined by one of the embodiments described above.
[0451] Compounds may be tested for ability to bind to and/or inhibit K-Ras activity according to any of various assays known in the art, including, for example, surface plasmon resonance binding assays, TR-FRET displacement binding assays, TR-FRET assays that detect Ras-Raf
interaction, and cell-based phosphorylation inhibition assays. Compounds may be tested for ability to bind to and/or inhibit K-Ras G12D activity according to any of various assays known in the art, including, for example, surface plasmon resonance binding assays, TR-FRET displacement binding assays, TR-FRET assays that detect Ras-Raf interaction, and cell-based phosphorylation inhibition assays. Such assays are described in, for example, WO 2021/041671 and Zhang, Z., el al. “GTP-State-Selective Cyclic Peptide Ligands of K-Ras(G12D) Block Its Interaction with Raf,” ACS Cent. Sci. (2020) Vol. 6, No. 10, pp. 1753-1761; the contents of each of which are herein incorporated by reference in their entirety.
III. Combination Therapy
[0452] Another aspect of the invention provides for combination therapy. Naphthyl-substituted quinolin-4( l//)-onc or related compounds described herein (e.g., a compound of Formula I, II, or III, or other compounds in Section I) or their pharmaceutically acceptable salts may be used in combination with additional therapeutic agents to treat medical disorders, such as a cancer.
[0453] In some embodiments, the present invention provides a method of treating a disclosed disease or condition comprising administering to a patient in need thereof an effective amount of a compound disclosed herein or a pharmaceutically acceptable salt thereof and co-administering simultaneously or sequentially an effective amount of one or more additional therapeutic agents, such as those described herein. In some embodiments, the method includes co-administering one additional therapeutic agent. In some embodiments, the method includes co-administering two additional therapeutic agents. In some embodiments, the combination of the disclosed compound and the additional therapeutic agent or agents acts synergistically.
[0454] One or more other therapeutic agents may be administered separately from a compound or composition of the invention, as part of a multiple dosage regimen. Alternatively, one or more other therapeutic agents may be part of a single dosage form, mixed together with a compound of this invention in a single composition. If administered as a multiple dosage regime, one or more other therapeutic agent and a compound or composition of the invention may be administered simultaneously, sequentially or within a period of time from one another, for example within 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 18, 20, 21, 22, 23, or 24 hours from one another. In some embodiments, one or more other therapeutic agent and a
compound or composition of the invention are administerd as a multiple dosage regimen more than 24 hours aparts.
[0455] As used herein, the term “combination,” “combined,” and related terms refers to the simultaneous or sequential administration of therapeutic agents in accordance with this invention. For example, a compound of the present invention can be administered with one or more other therapeutic agent(s) simultaneously or sequentially in separate unit dosage forms or together in a single unit dosage form. Accordingly, the present invention provides a single unit dosage form comprising a compound of the current invention, one or more other therapeutic agent(s), and a pharmaceutically acceptable carrier, adjuvant, or vehicle.
[0456] The amount of a compound of the invention and one or more other therapeutic agent(s) (in those compositions which comprise an additional therapeutic agent, such as a second anti-cancer agent, as described above) that can be combined with the carrier materials to produce a single dosage form varies depending upon the host treated and the particular mode of administration. Preferably, a composition of the invention should be formulated so that a dosage of between 0.01 - 100 mg/kg body weight/day of a compound of the invention can be administered.
[0457] In those compositions which comprise one or more other therapeutic agent(s), the one or more other therapeutic agent(s) and a compound of the invention can act synergistically. Therefore, the amount of the one or more other therapeutic agent(s) in such compositions may be less than that required in a monotherapy utilizing only that therapeutic agent. In such compositions a dosage of between 0.01 - 1,000 g/kg body weight/day of the one or more other therapeutic agent(s) can be administered.
[0458] The amount of one or more other therapeutic agent(s) present in the compositions of this invention is preferably no more than the amount that would normally be administered in a composition comprising that therapeutic agent as the only active agent. Preferably the amount of one or more other therapeutic agent(s) in the presently disclosed compositions ranges from about 50% to 100% of the amount normally present in a composition comprising that agent as the only therapeutically active agent. In some embodiments, one or more other therapeutic agent(s) is administered at a dosage of about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, or about 95% of the amount normally administered for
that agent. As used herein, the phrase "normally administered" means the amount an FDA- approved therapeutic agent is approved for dosing per the FDA label insert.
[0459] Accordingly, another aspect of the invention provides a method of treating cancer in a patient. The method comprises administering to a subject in need thereof (i) a therapeutically effective amount of a naphthyl-substituted quinolin-4( l /7)-onc or related compound described herein and (ii) a second anti-cancer agent, in order to treat the cancer.
[0460] In certain embodiments, the second anti-cancer agent is an EGFR inhibitor, a FAK inhibitor, a RAF/ERK/MAPK inhibitor, a mTOR Inhibitor, a MEK Inhibitor, or a Phosphoinositide 3-Kinase Inhibitor.
In certain embodiments, the second anti-cancer agent is an ALK Inhibitor, an ATR Inhibitor, an A2A Antagonist, a Base Excision Repair Inhibitor, a Bcr-Abl Tyrosine Kinase Inhibitor, a Bruton's Tyrosine Kinase Inhibitor, a CDC7 Inhibitor, a CHK1 Inhibitor, a Cyclin-Dependent Kinase Inhibitor, a DNA-PK Inhibitor, an Inhibitor of both DNA-PK and mTOR, a DNMT1 Inhibitor, a DNMT1 Inhibitor plus 2-chloro-deoxy adenosine, an HD AC Inhibitor, a Hedgehog Signaling Pathway Inhibitor, an IDO Inhibitor, a JAK Inhibitor, a mTOR Inhibitor, a MEK Inhibitor, a MELK Inhibitor, a MTH1 Inhibitor, a PARP Inhibitor, a Phosphoinositide 3-Kinase Inhibitor, an Inhibitor of both PARP1 and DHODH, a Proteasome Inhibitor, a Topoisomerase-II Inhibitor, a Tyrosine Kinase Inhibitor, a VEGFR Inhibitor, or a WEE1 Inhibitor.
[0461] In certain embodiments, the second anti-cancer agent is an ALK Inhibitor. In certain embodiments, the second anti-cancer agent is an ALK Inhibitor comprisng ceritinib or crizotinib. In certain embodiments, the second anti-cancer agent is an ATR Inhibitor. In certain embodiments, the second anti-cancer agent is an ATR Inhibitor comprising AZD6738 or VX- 970. In certain embodiments, the second anti-cancer agent is an A2A Antagonist. In certain embodiments, the second anti-cancer agent is a Base Excision Repair Inhibitor comprising methoxyamine. In certain embodiments, the second anti-cancer agent is a Base Excision Repair Inhibitor, such as methoxyamine. In certain embodiments, the second anti-cancer agent is a Bcr- Abl Tyrosine Kinase Inhibitor. In certain embodiments, the second anti-cancer agent is a Bcr- Abl Tyrosine Kinase Inhibitor comprising dasatinib or nilotinib. In certain embodiments, the second anti-cancer agent is a Bruton's Tyrosine Kinase Inhibitor. In certain embodiments, the second anti-cancer agent is a Bruton's Tyrosine Kinase Inhibitor comprising ibrutinib. In certain
embodiments, the second anti-cancer agent is a CDC7 Inhibitor. In certain embodiments, the second anti-cancer agent is a CDC7 Inhibitor comprising RXDX-103 or AS- 141.
[0462] In certain embodiments, the second anti-cancer agent is a CHK1 Inhibitor. In certain embodiments, the second anti-cancer agent is a CHK1 Inhibitor comprising MK-8776, ARRY- 575, or SAR-020106. In certain embodiments, the second anti-cancer agent is a Cyclin- Dependent Kinase Inhibitor. In certain embodiments, the second anti-cancer agent is a Cyclin- Dependent Kinase Inhibitor comprising palbociclib. In certain embodiments, the second anti cancer agent is a DNA-PK Inhibitor. In certain embodiments, the second anti-cancer agent is a DNA-PK Inhibitor comprising MSC2490484A. In certain embodiments, the second anti-cancer agent is Inhibitor of both DNA-PK and mTOR. In certain embodiments, the second anti-cancer agent comprises CC-115.
[0463] In certain embodiments, the second anti-cancer agent is a DNMT1 Inhibitor. In certain embodiments, the second anti-cancer agent is a DNMT1 Inhibitor comprising decitabine, RX- 3117, guadecitabine, NUC-8000, or azacytidine. In certain embodiments, the second anti-cancer agent comprises a DNMT1 Inhibitor and 2-chloro-deoxyadenosine. In certain embodiments, the second anti-cancer agent comprises ASTX-727.
[0464] In certain embodiments, the second anti-cancer agent is a HD AC Inhibitor. In certain embodiments, the second anti-cancer agent is a HDAC Inhibitor comprising OBP-801, CHR- 3996, etinostate, resminostate, pracinostat, CG-200745, panobinostat, romidepsin, mocetinostat, belinostat, AR-42, ricolinostat, KA-3000, or ACY-241.
[0465] In certain embodiments, the second anti-cancer agent is a Hedgehog Signaling Pathway Inhibitor. In certain embodiments, the second anti-cancer agent is a Hedgehog Signaling Pathway Inhibitor comprising sonidegib or vismodegib. In certain embodiments, the second anti-cancer agent is an IDO Inhibitor. In certain embodiments, the second anti-cancer agent is an IDO Inhibitor comprising INCB024360. In certain embodiments, the second anti-cancer agent is a JAK Inhibitor. In certain embodiments, the second anti-cancer agent is a JAK Inhibitor comprising ruxolitinib or tofacitinib. In certain embodiments, the second anti-cancer agent is a mTOR Inhibitor. In certain embodiments, the second anti-cancer agent is a mTOR Inhibitor comprising everolimus or temsirolimus. In certain embodiments, the second anti-cancer agent is a MEK Inhibitor. In certain embodiments, the second anti-cancer agent is a MEK Inhibitor
comprising cobimetinib or trametinib. In certain embodiments, the second anti-cancer agent is a MELK Inhibitor. In certain embodiments, the second anti-cancer agent is a MELK Inhibitor comprising ARN-7016, APTO-500, or OTS-167. In certain embodiments, the second anti cancer agent is a MTH1 Inhibitor. In certain embodiments, the second anti-cancer agent is a MTH1 Inhibitor comprising (S)-crizotinib, TH287, or TH588.
[0466] In certain embodiments, the second anti-cancer agent is a PARP Inhibitor. In certain embodiments, the second anti-cancer agent is a PARP Inhibitor comprising MP-124, olaparib, BGB-290, talazoparib, veliparib, niraparib, E7449, rucaparb, or ABT-767. In certain embodiments, the second anti-cancer agent is a Phosphoinositide 3-Kinase Inhibitor. In certain embodiments, the second anti-cancer agent is a Phosphoinositide 3-Kinase Inhibitor comprising idelalisib. In certain embodiments, the second anti-cancer agent is an inhibitor of both PARP1 and DHODH (i.e., an agent that inhibits both poly ADP ribose polymerase 1 and dihydroorotate dehydrogenase).
[0467] In certain embodiments, the second anti-cancer agent is a Proteasome Inhibitor. In certain embodiments, the second anti-cancer agent is a Proteasome Inhibitor comprising bortezomib or carfilzomib. In certain embodiments, the second anti-cancer agent is a Topoisomerase-II Inhibitor. In certain embodiments, the second anti-cancer agent is a Topoisomerase-II Inhibitor comprising vosaroxin.
[0468] In certain embodiments, the second anti-cancer agent is a Tyrosine Kinase Inhibitor. In certain embodiments, the second anti-cancer agent is a Tyrosine Kinase Inhibitor comprising bosutinib, cabozantinib, imatinib or ponatinib. In certain embodiments, the second anti-cancer agent is a VEGFR Inhibitor. In certain embodiments, the second anti-cancer agent is a VEGFR Inhibitor comprising regorafenib. In certain embodiments, the second anti-cancer agent is a WEE1 Inhibitor. In certain embodiments, the second anti-cancer agent is a WEE1 Inhibitor comprising AZD1775.
[0469] In some embodiments, the second anti-cancer agent is a compound targeting, decreasing or inhibiting the activity of members of the protein kinase C (PKC) and Raf family of serine/threonine kinases, members of the MEK, SRC, JAK/pan-JAK, FAK, PDK1, PKB/Akt, Ras/MAPK, PI3K, SYK, TYK2, BTK and TEC family, and/or members of the cyclin-dependent kinase family (CDK) including staurosporine derivatives, such as midostaurin; examples of
further compounds include UCN-01, safingol, BAY 43-9006, Bryostatin 1, Perifosine; llmofosine; RO 318220 and RO 320432; GO 6976; Isis 3521; LY333531/LY379196; isochinoline compounds; FTIs; PD184352 or QAN697 (a P13K inhibitor) or AT7519 (CDK inhibitor). In some embodiments, the second anti-cancer agent is a KRAS inhibitor, such as MRTX849 (Mirati Therapeutics) or sotorasib (Amgen).
[0470] In certain embodiments, the second anti-cancer agent is an agonist of 0X40, CD137, CD40, GITR, CD27, HVEM, TNFRSF25, or ICOS. In certain embodiments, the second anti cancer agent is an agonist of 0X40, CD137, CD40, or GITR. In certain embodiments, the second anti-cancer agent is an agonist of CD27, HVEM, TNFRSF25, or ICOS.
[0471] In certain embodiments, the second anti-cancer agent is a therapeutic antibody. In certain embodiments, the therapeutic antibody targets one of the following: CD20, CD30, CD33, CD52, EpCAM, CEA, gpA33, a mucin, TAG-72, CAIX, PSMA, a folate-binding protein, a ganglioside, Le, VEGF, VEGFR, VEGFR2, integrin aVp3, integrin a5b1, EGFR, ERBB2, ERBB3, MET, IGF1R, EPHA3, TRAILR1, TRAILR2, RANKL, FAP, tenascin, CD19, KIR, NKG2A, CD47, CEACAM1, c-MET, VISTA, CD73, CD38, BAFF, interleukin- 1 beta, B4GALNT1, interleukin- 6, and interleukin-6 receptor.
[0472] In certain embodiments, the second anti-cancer agent is a therapeutic antibody selected from the group consisting of rituximab, ibritumomab tiuxetan, tositumomab, obinutuzumab, ofatumumab, brentuximab vedotin, gemtuzumab ozogamicin, alemtuzumab, IGN101, adecatumumab, labetuzumab, huA33, pemtumomab, oregovomab, minetumomab, cG250, J591, Movl8, farletuzumab, 3F8, chl4.18, KW-2871, hu3S193, lgN311, bevacizumab, IM-2C6, pazopanib, sorafenib, axitinib, CDP791, lenvatinib, ramucirumab, etaracizumab, volociximab, cetuximab, panitumumab, nimotuzumab, 806, afatinib, erlotinib, gefitinib, osimertinib, vandetanib, trastuzumab, pertuzumab, MM-121, AMG 102, METMAB, SCH 900105,
AVE1642, IMC-A12, MK-0646, R1507, CP 751871, KB004, IIIA-4, mapatumumab, HGS- ETR2, CS-1008, denosumab, sibrotuzumab, F19, 81C6, MEDI551, lirilumab, MED 19447, daratumumab, belimumab, canakinumab, dinutuximab, siltuximab, and tocilizumab.
[0473] In certain embodiments, the second anti-cancer agent is a cytokine. In certain embodiments, the cytokine is IL-12, IL-15, GM-CSF, or G-CSF.
[0474] In certain embodiments, the second anti-cancer agent is sipuleucel-T, aldesleukin (a human recombinant interleukin-2 product having the chemical name des-alanyl-1, serine- 125 human interleukin-2), dabrafenib (a kinase inhibitor having the chemical name N-{3-[5-(2- aminopyri midi n-4-yl)-2-ter t -butyl- 1 ,3-thiazol-4-yl] -2- fluorophenyl } -2,6- difluorobenzenesulfonamide), vemurafenib (a kinase inhibitor having the chemical name propane- 1- sulfonic acid {3-[5-(4-chlorophcnyl)-1 H-pyrazolo[3,4-b]pyridine-3-carbonyl]-2,4- difluoro-phenyl}-amide), or 2-chloro-deoxyadenosine.
[0475] In certain embodiments, the second anti-cancer agent is a placental growth factor, an antibody-drug conjugate, an oncolytic vims, or an anti-cancer vaccine. In certain embodiments, the second anti-cancer agent is a placental growth factor. In certain embodiments, the second anti-cancer agent is a placental growth factor comprising ziv-aflibercept. In certain embodiments, the second anti-cancer agent is an antibody-drug conjugate. In certain embodiments, the second anti-cancer agent is an antibody-drug conjugate selected from the group consisting of brentoxumab vedotin and trastuzumab emtransine.
[0476] In certain embodiments, the second anti-cancer agent is an oncolytic virus. In certain embodiments, the second anti-cancer agent is the oncolytic vims talimogene laherparepvec. In certain embodiments, the second anti-cancer agent is an anti-cancer vaccine. In certain embodiments, the second anti-cancer agent is an anti-cancer vaccine selected from the group consistint of a GM-CSF tumor vaccine, a STING/GM-CSF tumor vaccine, and NY-ESO-1. In certain embodiments, the second anti-cancer agent is a cytokine selected from IL-12, IL-15, GM- CSF, and G-CSF.
[0477] In certain embodiments, the second anti-cancer agent is an immune checkpoint inhibitor (also referred to as immune checkpoint blockers). Immune checkpoint inhibitors are a class of therapeutic agents that have the effect of blocking immune checkpoints. See, for example, Pardoll in Nature Reviews Cancer (2012) vol. 12, pages 252-264. In certain embodiments, the immune checkpoint inhibitor is an agent that inhibits one or more of (i) cytotoxic T- lymphocyte- associated antigen 4 (CTLA4), (ii) programmed cell death protein 1 (PD1), (iii) PDL1, (iv) LAB3, (v) B7-H3, (vi) B7-H4, and (vii) TIM3. In certain embodiments, the immune checkpoint inhibitor is ipilumumab. In certain embodiments, the immune checkpoint inhibitor is pembrolizumab.
[0478] In certain embodiments, the second anti-cancer agent is a monoclonal antibody that targets a non-checkpoint target (e.g., herceptin). In certain embodiments, the second anti-cancer agent is a non-cytoxic agent (e.g., a tyrosine -kinase inhibitor).
[0479] In certain embodiments, the second anti-cancer agent is selected from mitomycin, ribomustin, vincristine, tretinoin, etoposide, cladribine, gemcitabine, mitobronitol, methotrexate, doxorubicin, carboquone, pentostatin, nitracrine, zinostatin, cetrorelix, letrozole, raltitrexed, daunombicin, fadrozole, fotemustine, thymalfasin, sobuzoxane, nedaplatin, aminoglutethimide, amsacrine, proglumide, elliptinium acetate, ketanserin, doxifluridine, etretinate, isotretinoin, streptozocin, nimustine, vindesine, cytarabine, bicalutamide, vinorelbine, vesnarinone, flutamide, drogenil, butocin, carmofur, razoxane, sizofilan, carboplatin, mitolactol, tegafur, ifosfamide, prednimustine, picibanil, levamisole, teniposide, improsulfan, enocitabine, lisuride, oxymetholone, tamoxifen, progesterone, mepitiostane, epitiostanol, formestane, colony stimulating factor- 1, colony stimulating factor-2, denileukin diftitox, interleukin-2, leutinizing hormone releasing factor, interferon-alpha, interferon-2 alpha, interferon-beta, interferon- gamma.
[0480] In certain embodiments, the second anti-cancer agent is radiation therapy.
[0481] In certain embodiments, the second anti-cancer agent is a MEK Inhibitor. In certain embodiments, the second anti-cancer agent is binimetinib, cobimetinib, refametinib, selumetinib, trametinib, or a pharmaceutically acceptable salt and/or solvate of any of the foregoing. In certain embodiments, the second anti-cancer agent is binimetinib, cobimetinib, selumetinib, trametinib, or a pharmaceutically acceptable salt and/or solvate of any of the foregoing.
[0482] In certain embodiments, the second anti-cancer agent is trametinib or a pharmaceutically acceptable salt and/or solvate thereof. In certain embodiments, the second anti-cancer agent is trametinib or a pharmaceutically acceptable solvate thereof. In certain embodiments, the second anti-cancer agent is trametinib dimethylsulfoxide. In certain embodiments, the second anti cancer agent is trametinib.
[0483] In certain embodiments, the second anti-cancer agent is binimetinib or a pharmaceutically acceptable salt and/or solvate thereof. In certain embodiments, the second anti-cancer agent is cobimetinib or a pharmaceutically acceptable salt and/or solvate thereof. In certain embodiments, the second anti-cancer agent is refametinib or a pharmaceutically acceptable salt and/or solvate
thereof. In certain embodiments, the second anti-cancer agent is selumetinib or a pharmaceutically acceptable salt and/or solvate thereof.
[0484] In certain embodiments, the second anti-cancer agent is binimetinib. In certain embodiments, the second anti-cancer agent is cobimetinib. In certain embodiments, the second anti-cancer agent is refametinib. In certain embodiments, the second anti-cancer agent is selumetinib.
[0485] In certain embodiments, the second anti-cancer agent is a TEAD inhibitor. In certain embodiments, the TEAD inhibitor is selected from those described in WO 2020/243415, the contents of which are herein incorporated by reference in their entirety. In certain embodiments, the TEAD inhibitor is selected from those described in WO 2020/243423, the contents of which are herein incorporated by reference in their entirety. In certain embodiments, the TEAD inhibitor is selected from those described in US Patent No. 11,274,082, the contents of which are herein incorporated by reference in their entirety.
[0486] In certain embodiments, the second anti-cancer agent is an ERK5 inhibitor. In certain embodiments, the ERK5 inhibitor is selected from those described in WO 2022/051567, the contents of which are herein incorporated by reference in their entirety. In certain embodiments, the ERK5 inhibitor is selected from those described in WO 2022/051565, the contents of which are herein incorporated by reference in their entirety. In certain embodiments, the ERK5 inhibitor is selected from those described in WO 2022/051569, the contents of which are herein incorporated by reference in their entirety. In certain embodiments, the ERK5 inhibitor is selected from those described in WO 2022/051568, the contents of which are herein incorporated by reference in their entirety.
Additional Combination Therapy Considerations
[0487] In certain embodiments, the method further comprises administering to the subject a third anti-cancer agent. In certain embodiments, the method further comprises administering to the subject a fourth anti-cancer agent. In certain embodiments, the method further comprises administering to the subject a fifth anti-cancer agent.
[0488] In certain embodiments, the third anti-cancer agent is one of the second anti-cancer agents described above. In certain embodiments, the fourth anti-cancer agent is one of the
second anti-cancer agents described above. In certain embodiments, the fifth anti-cancer agent is one of the second anti-cancer agents described above.
[0489] The doses and dosage regimen of the active ingredients used in the combination therapy may be determined by an attending clinician. In certain embodiments, the naphthyl-substituted quinolin-4( 1H)-onc or related compound described herein (e.g., a compound of Formula I, II, or III, or other compounds in Section I) and the additional therapeutic agent(s) (e.g. the second, third, or fourth, or fifth anti-cancer agent, described above) are administered in doses commonly employed when such agents are used as monotherapy for treating the disorder. In other embodiments, the naphthyl-substituted quinolin-4( 1 H)-onc or related compound described herein (e.g., a compound of Formula I, II, or III, or other compounds in Section I) and the additional therapeutic agent(s) (e.g. the second, third, or fourth, or fifth anti-cancer agent, described above) are administered in doses lower than the doses commonly employed when such agents are used as monotherapy for treating the disorder. In certain embodiments, the naphthyl-substituted quinolin-4( 1H)-onc or related compound described herein (e.g., a compound of Formula I, II, or III, or other compounds in Section I) and the additional therapeutic agent(s) (e.g. the second, third, or fourth, or fifth anti-cancer agent, described above) are present in the same composition, which is suitable for oral administration.
[0490] In certain embodiments, the naphthyl-substituted quinolin-4( 1 H)-onc or related compound described herein (e.g., a compound of Formula I, II, or III, or other compounds in Section I) and the additional therapeutic agent(s) (e.g. the second, third, or fourth, or fifth anticancer agent, described above) may act additively or synergistically. A synergistic combination may allow the use of lower dosages of one or more agents and/or less frequent administration of one or more agents of a combination therapy. A lower dosage or less frequent administration of one or more agents may lower toxicity of the therapy without reducing the efficacy of the therapy.
[0491] Another aspect of this invention is a kit comprising a therapeutically effective amount of the naphthyl-substituted quinolin-4( 1 H)-onc or related compound described herein (e.g., a compound of Formula I, II, or III, or other compounds in Section I), a pharmaceutically acceptable carrier, vehicle or diluent, and optionally at least one additional therapeutic agent listed above.
IV. Pharmaceutical Compositions and Dosing Considerations
[0492] As indicated above, the invention provides pharmaceutical compositions, which comprise a therapeutically-effective amount of one or more of the compounds described above, formulated together with one or more pharmaceutically acceptable carriers (additives) and/or diluents. The pharmaceutical compositions may be specially formulated for administration in solid or liquid form, including those adapted for the following: (1) oral administration, for example, drenches (aqueous or non-aqueous solutions or suspensions), tablets, e.g., those targeted for buccal, sublingual, and systemic absorption, boluses, powders, granules, pastes for application to the tongue; (2) parenteral administration, for example, by subcutaneous, intramuscular, intravenous or epidural injection as, for example, a sterile solution or suspension, or sustained-release formulation; (3) topical application, for example, as a cream, ointment, or a controlled-release patch or spray applied to the skin; (4) intravaginally or intrarectally, for example, as a pessary, cream or foam; (5) sublingually; (6) ocularly; (7) transdermally; or (8) nasally. In certain embodiments, the invention provides a pharmaceutical composition comprising a compound described herein (e.g., a compound of Formula I, II, or III) and a pharmaceutically acceptable carrier.
[0493] The phrase “therapeutically effective amount” as used herein means that amount of a compound, material, or composition comprising a compound of the present invention which is effective for producing some desired therapeutic effect in at least a sub-population of cells in an animal at a reasonable benefit/risk ratio applicable to any medical treatment. In certain embodiments, a therapeutically effective amount is an amount sufficient for inhibition of K-Ras G12D. In certain embodiments, a therapeutically effective amount is an amount sufficient for treating a proliferative disease, such as cancer.
[0494] The phrase “pharmaceutically acceptable” is employed herein to refer to those compounds, materials, compositions, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio.
[0495] Wetting agents, emulsifiers and lubricants, such as sodium lauryl sulfate and magnesium stearate, as well as coloring agents, release agents, coating agents, sweetening, flavoring and perfuming agents, preservatives and antioxidants can also be present in the compositions.
[0496] Examples of pharmaceutically-acceptable antioxidants include: (1) water soluble antioxidants, such as ascorbic acid, cysteine hydrochloride, sodium bisulfate, sodium metabisulfite, sodium sulfite and the like; (2) oil-soluble antioxidants, such as ascorbyl palmitate, butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), lecithin, propyl gallate, alpha-tocopherol, and the like; and (3) metal chelating agents, such as citric acid, ethylenediamine tetraacetic acid (EDTA), sorbitol, tartaric acid, phosphoric acid, and the like.
[0497] Formulations of the present invention include those suitable for oral, nasal, topical (including buccal and sublingual), rectal, vaginal and/or parenteral administration. The formulations may conveniently be presented in unit dosage form and may be prepared by any methods well known in the art of pharmacy. The amount of active ingredient which can be combined with a carrier material to produce a single dosage form will vary depending upon the host being treated, the particular mode of administration. The amount of active ingredient which can be combined with a carrier material to produce a single dosage form will generally be that amount of the compound which produces a therapeutic effect. Generally, out of one hundred percent, this amount will range from about 0.1 percent to about ninety-nine percent of active ingredient, preferably from about 5 percent to about 70 percent, most preferably from about 10 percent to about 30 percent.
[0498] In certain embodiments, a formulation of the present invention comprises an excipient selected from the group consisting of cyclodextrins, celluloses, liposomes, micelle forming agents, e.g., bile acids, and polymeric carriers, e.g., polyesters and poly anhydrides; and a compound of the present invention. In certain embodiments, an aforementioned formulation renders orally bioavailable a compound of the present invention.
[0499] Methods of preparing these formulations or compositions include the step of bringing into association a compound of the present invention with the carrier and, optionally, one or more accessory ingredients. In general, the formulations are prepared by uniformly and intimately bringing into association a compound of the present invention with liquid carriers, or finely divided solid carriers, or both, and then, if necessary, shaping the product.
[0500] Formulations of the invention suitable for oral administration may be in the form of capsules, cachets, pills, tablets, lozenges (using a flavored basis, usually sucrose and acacia or tragacanth), powders, granules, or as a solution or a suspension in an aqueous or non-aqueous liquid, or as an oil-in-water or water-in-oil liquid emulsion, or as an elixir or syrup, or as pastilles (using an inert base, such as gelatin and glycerin, or sucrose and acacia) and/or as mouth washes and the like, each containing a predetermined amount of a compound of the present invention as an active ingredient. A compound of the present invention may also be administered as a bolus, electuary or paste.
[0501] In solid dosage forms of the invention for oral administration (capsules, tablets, pills, dragees, powders, granules, trouches and the like), the active ingredient is mixed with one or more pharmaceutically-acceptable carriers, such as sodium citrate or dicalcium phosphate, and/or any of the following: (1) fillers or extenders, such as starches, lactose, sucrose, glucose, mannitol, and/or silicic acid; (2) binders, such as, for example, carboxymethylcellulose, alginates, gelatin, polyvinyl pyrrolidone, sucrose and/or acacia; (3) humectants, such as glycerol; (4) disintegrating agents, such as agar-agar, calcium carbonate, potato or tapioca starch, alginic acid, certain silicates, and sodium carbonate; (5) solution retarding agents, such as paraffin; (6) absorption accelerators, such as quaternary ammonium compounds and surfactants, such as poloxamer and sodium lauryl sulfate; (7) wetting agents, such as, for example, cetyl alcohol, glycerol monostearate, and non-ionic surfactants; (8) absorbents, such as kaolin and bentonite clay; (9) lubricants, such as talc, calcium stearate, magnesium stearate, solid polyethylene glycols, sodium lauryl sulfate, zinc stearate, sodium stearate, stearic acid, and mixtures thereof; (10) coloring agents; and (11) controlled release agents such as crospovidone or ethyl cellulose.
In the case of capsules, tablets and pills, the pharmaceutical compositions may also comprise buffering agents. Solid compositions of a similar type may also be employed as fillers in soft and hard-shelled gelatin capsules using such excipients as lactose or milk sugars, as well as high molecular weight polyethylene glycols and the like.
[0502] A tablet may be made by compression or molding, optionally with one or more accessory ingredients. Compressed tablets may be prepared using binder (for example, gelatin or hydroxypropylmethyl cellulose), lubricant, inert diluent, preservative, disintegrant (for example, sodium starch glycolate or cross-linked sodium carboxymethyl cellulose), surface-active or
dispersing agent. Molded tablets may be made by molding in a suitable machine a mixture of the powdered compound moistened with an inert liquid diluent.
[0503] The tablets, and other solid dosage forms of the pharmaceutical compositions of the present invention, such as dragees, capsules, pills and granules, may optionally be scored or prepared with coatings and shells, such as enteric coatings and other coatings well known in the pharmaceutical-formulating art. They may also be formulated so as to provide slow or controlled release of the active ingredient therein using, for example, hydroxypropylmethyl cellulose in varying proportions to provide the desired release profile, other polymer matrices, liposomes and/or microspheres. They may be formulated for rapid release, e.g., freeze-dried. They may be sterilized by, for example, filtration through a bacteria-retaining filter, or by incorporating sterilizing agents in the form of sterile solid compositions which can be dissolved in sterile water, or some other sterile injectable medium immediately before use. These compositions may also optionally contain opacifying agents and may be of a composition that they release the active ingredient(s) only, or preferentially, in a certain portion of the gastrointestinal tract, optionally, in a delayed manner. Examples of embedding compositions which can be used include polymeric substances and waxes. The active ingredient can also be in micro-encapsulated form, if appropriate, with one or more of the above-described excipients.
[0504] Liquid dosage forms for oral administration of the compounds of the invention include pharmaceutically acceptable emulsions, microemulsions, solutions, suspensions, syrups and elixirs. In addition to the active ingredient, the liquid dosage forms may contain inert diluents commonly used in the art, such as, for example, water or other solvents, solubilizing agents and emulsifiers, such as ethyl alcohol, isopropyl alcohol, ethyl carbonate, ethyl acetate, benzyl alcohol, benzyl benzoate, propylene glycol, 1,3-butylene glycol, oils (in particular, cottonseed, groundnut, corn, germ, olive, castor and sesame oils), glycerol, tetrahydrofuryl alcohol, polyethylene glycols and fatty acid esters of sorbitan, and mixtures thereof.
[0505] Besides inert diluents, the oral compositions can also include adjuvants such as wetting agents, emulsifying and suspending agents, sweetening, flavoring, coloring, perfuming and preservative agents.
[0506] Suspensions, in addition to the active compounds, may contain suspending agents as, for example, ethoxylated isostearyl alcohols, polyoxyethylene sorbitol and sorbitan esters,
microcrystalline cellulose, aluminum metahydroxide, bentonite, agar-agar and tragacanth, and mixtures thereof.
[0507] Formulations of the pharmaceutical compositions of the invention for rectal or vaginal administration may be presented as a suppository, which may be prepared by mixing one or more compounds of the invention with one or more suitable nonirritating excipients or carriers comprising, for example, cocoa butter, polyethylene glycol, a suppository wax or a salicylate, and which is solid at room temperature, but liquid at body temperature and, therefore, will melt in the rectum or vaginal cavity and release the active compound.
[0508] Formulations of the present invention which are suitable for vaginal administration also include pessaries, tampons, creams, gels, pastes, foams or spray formulations containing such carriers as are known in the art to be appropriate.
[0509] Dosage forms for the topical or transdermal administration of a compound of this invention include powders, sprays, ointments, pastes, creams, lotions, gels, solutions, patches and inhalants. The active compound may be mixed under sterile conditions with a pharmaceutically-acceptable carrier, and with any preservatives, buffers, or propellants which may be required.
[0510] The ointments, pastes, creams and gels may contain, in addition to an active compound of this invention, excipients, such as animal and vegetable fats, oils, waxes, paraffins, starch, tragacanth, cellulose derivatives, polyethylene glycols, silicones, bentonites, silicic acid, talc and zinc oxide, or mixtures thereof.
[0511] Powders and sprays can contain, in addition to a compound of this invention, excipients such as lactose, talc, silicic acid, aluminum hydroxide, calcium silicates and polyamide powder, or mixtures of these substances. Sprays can additionally contain customary propellants, such as chlorofluorohydrocarbons and volatile unsubstituted hydrocarbons, such as butane and propane.
[0512] Transdermal patches have the added advantage of providing controlled delivery of a compound of the present invention to the body. Such dosage forms can be made by dissolving or dispersing the compound in the proper medium. Absorption enhancers can also be used to increase the flux of the compound across the skin. The rate of such flux can be controlled by
either providing a rate controlling membrane or dispersing the compound in a polymer matrix or gel.
[0513] Ophthalmic formulations, eye ointments, powders, solutions and the like, are also contemplated as being within the scope of this invention.
[0514] Pharmaceutical compositions of this invention suitable for parenteral administration comprise one or more compounds of the invention in combination with one or more pharmaceutically-acceptable sterile isotonic aqueous or nonaqueous solutions, dispersions, suspensions or emulsions, or sterile powders which may be reconstituted into sterile injectable solutions or dispersions just prior to use, which may contain sugars, alcohols, antioxidants, buffers, bacteriostats, solutes which render the formulation isotonic with the blood of the intended recipient or suspending or thickening agents.
[0515] Examples of suitable aqueous and nonaqueous carriers which may be employed in the pharmaceutical compositions of the invention include water, ethanol, polyols (such as glycerol, propylene glycol, polyethylene glycol, and the like), and suitable mixtures thereof, vegetable oils, such as olive oil, and injectable organic esters, such as ethyl oleate. Proper fluidity can be maintained, for example, by the use of coating materials, such as lecithin, by the maintenance of the required particle size in the case of dispersions, and by the use of surfactants.
[0516] These compositions may also contain adjuvants such as preservatives, wetting agents, emulsifying agents and dispersing agents. Prevention of the action of microorganisms upon the subject compounds may be ensured by the inclusion of various antibacterial and antifungal agents, for example, paraben, chlorobutanol, phenol sorbic acid, and the like. It may also be desirable to include isotonic agents, such as sugars, sodium chloride, and the like into the compositions. In addition, prolonged absorption of the injectable pharmaceutical form may be brought about by the inclusion of agents which delay absorption such as aluminum monostearate and gelatin.
[0517] In some cases, in order to prolong the effect of a drug, it is desirable to slow the absorption of the drug from subcutaneous or intramuscular injection. This may be accomplished by the use of a liquid suspension of crystalline or amorphous material having poor water solubility. The rate of absorption of the drug then depends upon its rate of dissolution which, in turn, may depend upon crystal size and crystalline form. Alternatively, delayed absorption of a
parenterally-administered drug form is accomplished by dissolving or suspending the drug in an oil vehicle.
[0518] Injectable depot forms are made by forming microencapsule matrices of the subject compounds in biodegradable polymers such as polylactide-polyglycolide. Depending on the ratio of drug to polymer, and the nature of the particular polymer employed, the rate of drug release can be controlled. Examples of other biodegradable polymers include poly(orthoesters) and poly(anhydrides). Depot injectable formulations are also prepared by entrapping the drug in liposomes or microemulsions which are compatible with body tissue.
[0519] When the compounds of the present invention are administered as pharmaceuticals, to humans and animals, they can be given per se or as a pharmaceutical composition containing, for example, 0.1 to 99% (more preferably, 10 to 30%) of active ingredient in combination with a pharmaceutically acceptable carrier.
[0520] The preparations of the present invention may be given orally, parenterahy, topically, or rectahy. They are of course given in forms suitable for each administration route. For example, they are administered in tablets or capsule form, by injection, inhalation, eye lotion, ointment, suppository, etc. administration by injection, infusion or inhalation; topical by lotion or ointment; and rectal by suppositories. Oral administrations are preferred.
[0521] The phrases “parenteral administration” and “administered parenterahy” as used herein means modes of administration other than enteral and topical administration, usually by injection, and includes, without limitation, intravenous, intramuscular, intraarterial, intrathecal, intracapsular, intraorbital, intracardiac, intradermal, intraperitoneal, transtracheal, subcutaneous, subcuticular, intraarticular, subcapsular, subarachnoid, intraspinal and intrastemal injection and infusion.
[0522] The phrases “systemic administration,” “administered systemicahy,” “peripheral administration” and “administered peripherally” as used herein mean the administration of a compound, drug or other material other than directly into the central nervous system, such that it enters the patient’s system and, thus, is subject to metabolism and other like processes, for example, subcutaneous administration.
[0523] These compounds may be administered to humans and other animals for therapy by any suitable route of administration, including orally, nasally, as by, for example, a spray, rectally, intravaginally, parenterally, intracisternally and topically, as by powders, ointments or drops, including buccally and sublingually.
[0524] Regardless of the route of administration selected, the compounds of the present invention, which may be used in a suitable hydrated form, and/or the pharmaceutical compositions of the present invention, are formulated into pharmaceutically-acceptable dosage forms by conventional methods known to those of skill in the art.
[0525] Actual dosage levels of the active ingredients in the pharmaceutical compositions of this invention may be varied so as to obtain an amount of the active ingredient which is effective to achieve the desired therapeutic response for a particular patient, composition, and mode of administration, without being toxic to the patient.
[0526] The selected dosage level will depend upon a variety of factors including the activity of the particular compound of the present invention employed, or the ester, salt or amide thereof, the route of administration, the time of administration, the rate of excretion or metabolism of the particular compound being employed, the rate and extent of absorption, the duration of the treatment, other drugs, compounds and/or materials used in combination with the particular compound employed, the age, sex, weight, condition, general health and prior medical history of the patient being treated, and like factors well known in the medical arts.
[0527] A physician or veterinarian having ordinary skill in the art can readily determine and prescribe the effective amount of the pharmaceutical composition required. For example, the physician or veterinarian could start doses of the compounds of the invention employed in the pharmaceutical composition at levels lower than that required in order to achieve the desired therapeutic effect and gradually increase the dosage until the desired effect is achieved.
[0528] In general, a suitable daily dose of a compound of the invention will be that amount of the compound which is the lowest dose effective to produce a therapeutic effect. Such an effective dose will generally depend upon the factors described above. Preferably, the compounds are administered at about 0.01 mg/kg to about 200 mg/kg, more preferably at about 0.1 mg/kg to about 100 mg/kg, even more preferably at about 0.5 mg/kg to about 50 mg/kg.
When the compounds described herein are co-administered with another agent ( e.g ., as sensitizing agents), the effective amount may be less than when the agent is used alone.
[0529] If desired, the effective daily dose of the active compound may be administered as two, three, four, five, six or more sub-doses administered separately at appropriate intervals throughout the day, optionally, in unit dosage forms. Preferred dosing is one administration per day.
[0530] The invention further provides a unit dosage form (such as a tablet or capsule) comprising a naphthyl-substituted quinolin-4( 1H)-onc or related compound described herein in a therapeutically effective amount for the treatment of a medical disorder described herein.
EXAMPLES
[0531] The invention now being generally described, will be more readily understood by reference to the following examples, which are included merely for purposes of illustration of certain aspects and embodiments of the present invention, and are not intended to limit the invention. Starting materials described herein can be obtained from commercial sources or may be readily prepared from commercially available materials using transformations known to those of skill in the art.
EXAMPLE 1 - Synthesis of 4-[4-(3,8-Diazabicyclo[3.2.1]octan-3-yl)-2-[[(2R,8S)-2-fluoro- l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-7- yl]-5-methyl-naphthalen-2-ol (1-31)
Step 1: Benzyl 4-(8-tert-butoxycarbonyl-3,8-diazabicyclo[3.2.1]octan-3-yl)-2-chloro-6,8- dihydro-5H-pyrido[3,4-d]pyrimidine-7-carboxylate
[0532] To a solution of benzyl 2, 4-dich loro-6, 8-di hydro-5 H-pyrido[ 3, 4-dJpyri midinc-7- carboxylate (1 g, 2.96 mmol, 1 eq) and tert-butyl 3,8-diazabicyclo[3.2.1]octane-8-carboxylate (564.95 mg, 2.66 mmol, 0.9 eq) in THF (10 mL) was added TEA (897.63 mg, 8.87 mmol, 1.23 mL, 3 eq). The mixture was stirred at 25 °C for 12 h. The reaction mixture was diluted with H2O
(20 mL) and extracted with EtOAc (20 mL x 3). The combined organics were concentrated to yield the crude product which was purified by flash silica gel chromatography (from PE/EtOAc = 100/1 to 3/1, TLC: PE/EtOAc = 3/1, Rf = 0.42) to yeild benzyl 4-(8-tert-butoxycarbonyl-3,8- diazabicyclo[3.2.1]octan-3-yl)-2-chloro-6,8-dihydro-5H-pyrido[3,4-d]pyrimidine-7-carboxylate (1 g, 1.88 mmol, 63.6% yield, 96.6% purity) as a gray solid. 1H NMR (400 MHz, DMSO-d6) d ppm 7.41-7.32 (m, 5H), 5.13 (s, 2H), 4.44 (d, J = 19.6 Hz, 2H), 4.16 (s, 2H), 3.90 (d, J = 12.3 Hz, 2H), 3.53 (s, 2H), 3.11 (d, J= 12.4 Hz, 2H), 2.68 (s, 2H), 1.78 (d, J = 4.4 Hz, 2H), 1.69 (d, J = 8.0 Hz, 2H), 1.42 (s, 9H); ES-LCMS m/z 514.7, 516.7 [M+H]+.
Step 2: Benzyl 4-(8-tert-butoxycarbonyl-3,8-diazabicyclo[3.2.1]octan-3-yl)-2-[[(2R,8S)-2- fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]-6,8-dihydro-5H-pyrido[3,4- d]pyrimidine-7-carboxylate
[0533] To a solution of benzyl 4-(8-tert-butoxycarbonyl-3,8-diazabicyclo[3.2.1]octan-3-yl)-2- ch loro-6, 8-di hydro-5 H-pyrido[ 3, 4-dJpyri midi nc-7-carboxy late (500 mg, 939.66 μmol, 96.6%, 1 eq) and [(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methanol (373.99 mg, 2.35 mmol, 2.5 eq) in THF (10 mL) was added CS2CO3 (918.48 mg, 2.82 mmol, 3 eq) and 1,4- diazabicyclo[2.2.2]octane (10.54 mg, 93.97 pmol, 10.33 pL, 0.1 eq). The mixture was stirred at 100 °C for 12 h. The reaction mixture was diluted with H2O (30 mL) and extracted with EtOAc (30 mL x 3). The combined organics were concentrated to yield the crude product which was purified by preparative TLC (PE/EtOAc = 0/1, TLC: PE/EtOAc = 0/1, Rf = 0.08) to yield benzyl 4-(8-tert-butoxycarbonyl-3,8-diazabicyclo[3.2.1]octan-3-yl)-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]-6,8-dihydro-5H-pyrido[3,4-d]pyrimidine-7-carboxylate (330 mg, 502.71 pmol, 53.5% yield, 97.0% purity) as a yellow solid. 1H NMR (500 MHz, DMSO-d6 ) d ppm 7.40-7.31 (m, 5H), 5.32-5.18 (m, 1H), 5.12 (s, 2H), 4.40-4.33 (m, 2H), 4.15 (s, 2H), 3.93 (d, / = 10.2 Hz, 1H), 3.85-3.80 (m, 3H), 3.52 (s, 2H), 3.06-2.99 (m, 4H), 2.83-2.78 (m, 1H),
2.65-2.59 (m, 2H), 2.07-1.98 (m, 2H), 1.93 (s, 1H), 1.84-1.67 (m, 8H), 1.42 (s, 9H); ES-LCMS m/z 637.7 [M+H]+.
Step 3: tert- Butyl 3-[2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]- 5,6,7,8-tetrahydropyrido[3,4-d]pyrimidin-4-yl]-3,8-diazabicyclo[3.2.1]octane-8-carboxylate
[0534] To a solution of benzyl 4-(8-tert-butoxycarbonyl-3,8-diazabicyclo[3.2.1]octan-3-yl)-2- [[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]-6,8-dihydro-5H-pyrido[3,4- d]pyrimidine-7-carboxylate (330 mg, 502.71 μmol, 97.0%, 1 eq) in i-PrOH (10 mL) was added NH3Ή2O (943.81 mg, 7.54 mmol, 1.04 mL, 28.0%, 15 eq) and Pd/C (330 mg, 10.0%). The mixture was stirred at 25 °C for 1 h under ¾ (15 Psi). The reaction mixture was filtered and dried under reduced pressure to yield ter t -butyl 3-[2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]-5,6,7,8-tetrahydropyrido[3,4-d]pyrimidin-4-yl]-3,8- diazabicyclo[3.2.1]octane-8-carboxylate (260 mg, 491.42 pmol, 97.8% yield, 95.0% purity) as yellow oil which was used in the next step without further purification. 'H NMR (400 MHz, DMSO-d6) d ppm 4.17 (s, 2H), 3.92-3.89 (m, 1H), 3.82-3.74 (m, 4H), 3.65 (s,2H), 3.06-2.97 (m, 6H), 2.81-2.77 (m, 3H), 2.08-2.05 (m, 1H), 1.95-1.91 (m, 1H), 1.82-1.72 (m, 8H), 1.42 (s, 9H), 1.27-1.22 (m, 1H); ES-LCMS m/z 503.3 [M+H]+.
Step 4: tert -Butyl 3-[2-[[(2/?,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]-7- [3-(methoxymethoxy)-8-methyl-l-naphthyl]-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-4-yl]- 3,8-diazabicyclo[3.2.1]octane-8-carboxylate
[0535] A mixture of tert- butyl 3-[2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8- yl]methoxy]-5,6,7,8-tetrahydropyrido[3,4-d]pyrimidin-4-yl]-3,8-diazabicyclo[3.2.1]octane-8- carboxylate (260 mg, 491.42 μmol, 95.0%, 1 eq), l-bromo-3-(methoxymethoxy)-8-methyl- naphthalene (290.86 mg, 982.84 pmol, 95.0%, 2 eq), Pd2(dba)3 (67.50 mg, 73.71 pmol, 0.15 eq), RuPhos (45.86 mg, 98.28 pmol, 0.2 eq) and CS2CO3 (320.23 mg, 982.84 pmol, 2 eq) in toluene (8 mL) was degassed and purged with N2 for 3 times, and then the mixture was stirred at 110 °C for 12 h under N2 atmosphere. The reaction mixture was diluted with H2O (30 mL) and extracted with EtOAc (30 mL x 3). The combined organics were concentrated to yield the crude product which was purified by preparative TLC (PE/EtOAc = 0/1, TLC: PE/EtOAc = 0/1, Rf= 0.03) to yield ter t -butyl 3-[2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]-7-[3- (mcthoxymcthoxy)-8- methyl- 1 -naphthylJ-6,8-dihydiO-5H-pyrido[3,4-d]pyri midi n-4-yl] -3,8- diazabicyclo[3.2.1]octane-8-carboxylate (230 mg, 163.62 pmol, 33.3% yield, 50.0% purity) as a black brown solid.
NMR (400 MHz, CDCI3) S ppm 8.25 (d, J = 9.2 Hz, 1H), 7.70 (d, J = 8.2 Hz, 1H), 7.41 (d, J = 2.5 Hz, 1H), 7.19 (d, J = 2.5 Hz, 1H), 6.92 (s, 1H), 5.31 (s, 2H), 5.18 (s, 1H), 4.42-4.27 (m, 5H), 4.24-4.20 (m, 2H), 4.10-4.05 (m, 2H), 3.53 (d, J= 1.9 Hz, 4H), 3.22 (d, J= 5.4 Hz, 2H), 3.15 (s, 2H), 3.07 (d, J= 9.8 Hz, 2H), 2.98-2.94 (m, 2H), 2.86 (s, 2H), 2.67-2.57 (m, 4H), 2.26 (t, J= 7.3 Hz, 2H), 2.05 (s, 3H), 1.51 (s, 2H), 1.48 (s, 9H); ES-LCMS m/z 703.3 [M+H]+.
Step 5: 4-[4-(3,8-Diazabicyclo[3.2.1]octan-3-yl)-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-7-yl]-5-methyl- naphthalen-2-ol (1-31)
[0536] A mixture of tert- butyl 3-[2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8- yljmcthoxy] -7- [ 3 -(mcthoxymcthoxy)-8- methyl- 1 -naphthyl] -6,8-di hydro-5 H-pyrido[ 3, 4- d]pyrimidin-4-yl]-3,8-diazabicyclo[3.2.1]octane-8-carboxylate (100 mg, 71.14 μmol, 50.0%, 1 eq) in HC1 (1 M, 1.50 mL, 21.09 eq) and ACN (1 mL) was lyophilized to yield the residue which was purified by preparative HPLC (column: Boston Prime C18 150*30mm*5um; mobile phase: [water(FA)-ACN]; B%: 18%-38%, 14 min) to yield 4-[4-(3,8-diazabicyclo[3.2.1]octan-3-yl)-2- [[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]-6,8-dihydro-5H-pyrido[3,4- d]pyrimidin-7-yl]-5-methyl-naphthalen-2-ol (12.75 mg, 19.32 pmol, 27.2% yield, 98.6% purity, 2FA) as a yellow solid.
NMR (500 MHz, CD3OD) d ppm 8.29 (s, 1H), 8.20 (d, 7 = 9.0 Hz, 1H), 7.62 (d, J= 8.1 Hz, 1H), 7.35-7.31 (m, 1H), 7.29-7.26 (m, 1H), 7.12 (d, 7 = 2.3 Hz, 1H), 7.08 (dd, 7 = 2.4, 9.2 Hz, 1H), 5.58-5.46 (m, 1H), 4.43 (q, 7= 11.8 Hz, 2H), 4.18-4.11 (m, 4H), 4.08 (s, 2H), 3.96-3.72 (m, 4H), 3.54 (s, 2H), 3.41 (d, 7= 13.6 Hz, 2H), 3.38-3.32 (m, 1H), 2.83- 2.77 (m, 2H), 2.72 (d, 7 = 4.9 Hz, 2H), 2.65-2.46 (m, 2H), 2.35-2.24 (m, 3H), 2.13-2.07 (m, 5H); ES-LCMS m/z 559.3 [M+H]+.
EXAMPLE 2 — Biochemical Assay for Inhibiting Isolated K-Ras
[0537] An exemplary compound was tested for its ability to inhibit isolated K-Ras G12D, G12V, G12C, and wild-type using a homogeneous time-resolved fluorescence assay. Assay procedures and results are described below.
Part I - Procedures for Homogeneous Time-Resolved Fluorescence Assay
[0538] The assay quantifies the inhibition of SOSl-mediated nucleotide exchange of K-Ras (G12D, G12V, G12C, or wild-type). The K-Ras was incubated with Tb-streptavidin to pre equilibrate the K-Ras complex. The K-Ras complex was allowed to incubate with varied concentrations of test compounds before the addition of the mixture of SOS1 and fluorescently- labeled GTP (BODIPY-GTP) to initiate the reaction. Then, the TR-FRET signal was monitored after 30 min. The TR-FRET signal was generated by Tb and BODIPY-GTP, that displaced unlabeled GDP.
[0539] Materials: Biotinylated K-Ras enzyme (G12D, G12V, G12C, and wild-type), and SOS1 were purchased from GenScript. Other materials were: Tb-streptavidin (Cisbio-610SATLB), BODIPY FL GTP (Thermo-G12411), and white 384-well ProxiPlus Assay Plates (Perkin-Elmer- 6008289).
[0540] Final Assay Conditions: The assay buffer was 20 mM HEPES pH 7.5, 50 mM NaCl, 10 mM MgCl2, 0.01% Tween-20, 1 mM DTT. Total assay volume was 15 μL, and final assay concentrations were biotinylated K-Ras enzyme (G12D, G12V, or wild-type; 1.0 nM), SOS1 (200 nM), Tb-streptavidin (1 nM), and BODIPY-GTP (100 nM). The assay was conducted at 23 °C. Controls included all the reagents, but replaced test compound solution with DMSO (0% inhibition control) or 100 mM GDP (100% inhibition control).
[0541] Procedure: Test compound was dissolved and diluted in DMSO (100 mM top concentration, 3-fold dilution, 11 doses), then transferred to the assay plate using Echo. The final DMSO concentration was 1.5%. The mixture of 1.5x K-Ras enzyme (G12D, G12V, G12C, or wild-type) and Streptavidin-Tb in assay buffer was prepared, and then pre-incubated at 23°C for 1 hour. The enzyme mixture was then dispensed into the assay plate (10 μL/weII). The plate was allowed to react with enzyme and compound for 30 minutes. A mixture of 3x SOS1 and BODIPY-GTP in assay buffer was added (5 μL/well). The plate was spun at 1000 rpm for 30 seconds and then sealed. After incubating for 30 min, the assay signal was detected with Envision plate reader. The TR-FRET ratios were used to calculate percent inhibition values for each concentration of compound, using the no inhibition and 100% inhibition controls. IC50 values were calculated by Xlfit from the percent inhibition values using the 4-parameter logistical equation.
Part II - Results
[0542] Experimental results are provided in Table 4, below. The symbol “+++” indicates an IC50 less than or equal to 0.05 mM. The symbol “++” indicates an IC50 in the range of 0.5 mM to greater than 0.05 mM. The symbol “+” indicates an IC50 greater than 0.5 mM.
EXAMPLE 3 - Cellular Assay for Inhibiting K-Ras G12D-mediated Phosphorylation of ERK
[0543] An exemplary compound was tested for its ability to inhibit phosphorylation of ERK downstream of K-Ras G12D using a homogeneous time-resolved fluorescence assay. Assay procedures and results are described below.
Part I - Procedures for Homogeneous Time-Resolved Fluorescence Assay
[0544] Human AsPc-1 cells expressing K-Ras G12D are seeded in 384-well culture plates and grown overnight. Cells are pretreated with compound (10-pt titration) for 4 hrs. Then, cell lysates are prepared and assayed for levels of phosphorylated ERK1/2 using a homogeneous TR-FRET assay.
[0545] Materials: Human AsPc-1 cells were provided from WuXi. Other materials were: RPMI-1640 medium (Invitrogen-22400089), Fetal bovine serum (Excell-FCP500), Coming® BioCoat™ Microplate (Coming-356663), Advanced phospho-ERK HTRF Assay (CisBio- 64AERPEH), and assay plate (Perkin Elmer-6008289).
[0546] Assay Conditions: The culture/assay medium was RPMI-1640 +10% FBS. Cells were seeded at 6,000 cells/well, 50 mE/well. Compound treatment time was 4 hours. Controls replaced test compound solution with 0.8% DMSO (max control) or 0.2 mM trametinib (min control).
[0547] Procedure: Cells were harvested from T150 growthflask using 0.05% trypsin/EDTA solution. Medium (10 mL) was added to stop trypsinizing. The cells were pipetted into a
conical-bottomed, 50-mL centrifuge tube and centrifuged for 5 minutes at 1,000 rpm. The cell pellet was resuspended in medium, and cell count was determined (ViCell counter). Cell density was adjusted using fresh medium, then 384-well poly-D-lysine coated plates were seeded with 50 μL/well of the cell suspension. The cell plate was incubated overnight in a 37 °C, 5% CO2 incubator.
[0548] Test compounds were dissolved and diluted in DMSO (20 mM top concentration, 3-fold dilution, 10 doses) using Tecan. Compounds were added to the cell plate. The final DMSO concentration was 0.8%. The cell plate was incubated for 4 hours in the incubator.
[0549] Detection was conducted using a CisBio pERK kit. Kit-supplied lysis buffer was prepared and kept on ice. Concentrated (4x) lysis buffer was diluted with 3 volumes of deionized water, then lOOx of the blocking agent was added. Media was removed from the cell plate. Lysis buffer (35 mE/well) was added, and then the plate was placed on a plate agitator and shaken at 300 rpm, 4 °C for 40 minutes. Upon completion, the resulting lysate plate was centrifuged for 3 minutes at 1500 rpm.
[0550] Assay plates were prepared containing the HTRF antibody buffer. For each assay plate, 50 μL of d2-conjugate antibody was mixed with 950 μL of detection buffer. For each assay plate, 50 μL of cryptate antibody was mixed with 950 μL of detection buffer. The two diluted antibodies were then mixed together, and 3.4 μL of the resulting antibody buffer was dispensed to wells of an empty 384-well white Proxi plate. The plate was sealed and centrifuged 30 minutes at 1,000 rpm.
[0551] Lysate (13.6 μL ) from each well of the lysate plate was added using Bravo to each well of the antibody plate (prepared as described above). The plate was centrifuged 30 seconds at 1,000 rpm, and then incubated 2 hours at room temperature. Fluorescence was detected with Envision plate reader (665 nm/615 nm). The TR-FRET ratios were used to calculate percent inhibition values for each concentration of compound: % Inhibition = ((max - sample) / (max - min)) * 100. Curves were fit by XLFIT5 as % inhibition vs. log [compound concentration] using a 4-parameter fit.
Part II - Results
[0552] Experimental results are provided in Table 5, below. The symbol “***” indicates an IC50 less than or equal to 0.5 mM. The symbol
indicates an IC50 in the range of 10 mM to greater than 0.5 mM. The symbol
indicates an IC50 greater than 10 mM.
EXAMPLE 4 - Biological Assay for Inhibiting GMPPNP-bound KRas to RAF
[0553] An exemplary compound was tested for its ability to inhibit the binding of GMPPNP- bound KRas (G12D or WT) to the Ras binding domain of RAF.
Part I - Procedure
[0554] KRas protein was biotinylated and RAF protein was FLAG-tagged. Compounds were incubated with KRas-GMPPNP for 30 minutes at room temperature, followed by addition of RAF protein, Tb-anti-FLAG and SA-XL665 HTRF reagents. Fluorescence detection was carried out after 1.5 hours of incubation. IC50 values were calculated using a four-parameter fit.
[0555] Materials: Biotinylated GMPPNP-loaded KRAS-G12D, GMPPNP-loaded KRAS-WT and FLAG-tagged-Raf-RBD were purchased from GenScript. GMPPNP was obtained from Sigma. Perkin Elmer and Cisbio provided the Tb-anti-FLAG and streptavidin XL665 reagents and the white 384W ProxiPlus assay plates.
[0556] Final Assay Conditions: The assay buffer was 20 mM HEPES, pH 7.5, 150 mM NaCl,
1 mM MgCh, 0.01% BGG, 0.01% Tween-20, 1 mM DTT. Final assay concentrations were biotinylated KRAS G12D/WT (20 nM), FLAG-Raf-RBD (70 nM), Tb-anti-FLAG (1 nM) and SA-XL665 (20 nM). The assay was executed at 23°C in a total volume of 15 mE. Controls included all the reagents, but replaced test compound solution with DMSO (0% inhibition control) or removal of KRAS protein (100% inhibition control).
[0557] Procedure: Test compounds were dissolved and diluted in DMSO (100 mM top concentration, 3-fold dilution, 11 doses), then transferred to the assay plate using Echo. The final DMSO concentration was 1.5%.
[0558] Ten mE of a mixture of 1.5x GMPPNP-KRAS was added to the assay plate and the plate was spun at 1000 rpm for 30 sec and sealed. The plate was incubated for 30 minutes at 23°C.
Five mE of a mixture of 3x Raf-RBD containing Tb-anti-FLAG and SA-XF665 was added. The plate was spun at 1000 rpm for 30 sec and sealed. HTRF assay signal was measured after a 90 min incubation.
[0559] The HTRF ratios were used to calculate percent inhibition values for each concentration of compound, using the no inhibition and 100% inhibition controls. IC50 values were calculated by Xlfit from the percent inhibition values using the 4-parameter logistical equation.
Part II - Results
[0560] Experimental results are provided in Table 6, below. The symbol “***” indicates an IC50 less than or equal to 0.5 mM. The symbol
indicates an IC50 in the range of 10 mM to greater than 0.5 mM. The symbol
indicates an IC50 greater than 10 mM.
EXAMPLE 5 — Assay for Inhibition of K-Ras G12D-mediated Phosphorylation of ERK
[0561] Compounds may be tested for ability to inhibit phosphorylation of ERK downstream of K-Ras G12D according to the following experimental procedure.
[0562] AGS cells (ATCC CRL-1739) expressing G12D are grown in DMEM medium supplemented with 10% fetal bovine serum, 10 mM HEPES, and Penicillin/Streptomycin. Cells are plated in tissue culture treated 96 well plates at a density of 40,000 cells/well and allowed to attach for 12-14 hours. Diluted test compound is then added in a final concentration of 0.5% DMSO. After 3 hours, the medium is removed, 150 pL of 4.0% formaldehyde is added and the plates incubated at room temperature for 20 minutes. Then, the plates are washed with PBS, and
permeabilized with 150 pL of ice cold 100% methanol for 10 minutes. Non-specific antibody binding to the plates is blocked using 100 pL Licor blocking buffer (Li-Cor Biotechnology, Lincoln NE) for 1 hour at room temperature.
[0563] The amount of phospho-ERK is determined using an antibody specific for the phosphorylated form of ERK and compared to the amount of GAPDH. Primary antibodies used for the detection are added as follows: Phospho-ERK (Cell Signaling cs-9101) diluted 1:500 and GAPDH(Millipore MAB374) diluted 1:5000 in Licor block + 0.05%Tween 20. The plates are incubated for 2 hours at room temperature. Then, the plates are washed with PBS + 0.05%
Tween 20.
[0564] Secondary antibodies used to visualize primary antibodies are added as follows: Anti- rabbit-680 diluted 1 : 1000 and Anti-mouse-800 diluted 1 : 1000 both in Licor block +0.05% TweeN20, and are incubated for 1 hour at room temperature. Then, the plates are washed with PBS + 0.05% Tween 20. A 100 pL aliquot of PBS is added to each well and the plates are read on a Li-Cor Odyssey CLX plate reader.
[0565] The phospho-ERK Thr202/Tyr204 signal is normalized to the GAPDH signal for each well and percent of DMSO control values are calculated. IC50 values are generated using a 4- parameter fit of the dose response curve
EXAMPLE 6 — Biological Assay for Inhibiting 3D Cancer Cell Cultures Using Combination Therapy
[0566] Exemplary compounds having K-Ras G12D inhibitory activity may be tested for ability to inhibit 3D cancer cell cultures of pancreatic cancer and lung cancer. Assay procedures are described below.
Part I -Procedures for 3D Cancer Cell Culture Assay
[0567] Assays are conducted using K-Ras mutant (NCI- H2122 and A549) and K-Ras wild-type (NCI-H520) human lung cancer cell lines according to the procedures described in Haagensen,
E. L, et al. Eur. J. Cancer (2016) Vol. 56, pp. 69-76. Briefly, cells are seeded in 231 soft agar in 96-well, low-attachment tissue-culture plates. After 24 hours, cells are treated with a concentration range of one or more K-Ras G12D inhibitors. At the time of treatment, assay plates that did not receive treatment are collected and cell viability is measured using alamarBlue
reagent to establish baseline cell viability. Treated plates are incubated for 7 days, and alamarBlue readout is then collected.
[0568] Inhibition (%) is used as a measure of cell viability and is calculated using the equation I = 1 -T/U, where T is treated and U is untreated control. Single-agent response curve fitting, and the analysis of combination effects, are performed using CHALICE™ software from Horizon. The calculated synergy scores are based on the Loewe Additivity model and characterize the strength of synergistic interaction. A score greater than 1.5 represents synergistic activity between treatment agents.
EXAMPLE 7 — Biological Assay for Inhibiting Mouse Xenograft Tumor Growth
[0569] Exemplary compounds having K-Ras G12D inhibitory activity may be tested, alone or in combination with other agents, such as trametinib, for ability to inhibit tumor growth in patient- derived mouse xenograft models of pancreatic cancer and lung cancer. Assay procedures are described below.
Part I -Procedures for Patient-Derived Mouse Xenograft (PDX) Tumor Growth Assay
[0570] Balb/c mice (6-8 weeks old) are inoculated subcutaneously in the right flank with a primary human tumor xenograft model tumor fragment (2-3 mm3 in diameter) for tumor development. In one study, mice are inoculated with a human lung adenocarcinoma tumor model (MSCLC, ADC model LU6424) that harbors a BRAF mutation (LU6424); in a second study, mice are inoculated with a human pancreatic tumor model (adenosquamous carcinoma model PA6258) with a K-Ras G12D mutation (PA6258). When mean tumor volume reaches approximately 150-200 mm3, animals are randomly allocated to appropriate treatment groups. Mice are treated with one of the following: (l)Vehicle control, (2) Exemplary K-Ras G12D inhibitor alone, (3) Second anti-cancer agent alone, or (4) Combination of K-Ras G12D inhibitor and second anti-cancer agent. Tumors are measured twice per week using calipers.
INCORPORATION BY REFERENCE
[0571] The entire disclosure of each of the patent documents and scientific articles referred to herein is incorporated by reference for all purposes.
EQUIVALENTS
[0572] The invention may be embodied in other specific forms without departing from the spirit or essential characteristics thereof. The foregoing embodiments are therefore to be considered in all respects illustrative rather than limiting the invention described herein. Scope of the invention is thus indicated by the appended claims rather than by the foregoing description, and all changes that come within the meaning and range of equivalency of the claims are intended to be embraced therein.
Claims
R1 is naphthyl; an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with m occurrences of R5;
R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci-6 aliphatic; each of which is substituted with n occurrences of R6;
R3 is hydrogen; or one occurrence of R3 is hydrogen, and the other occurrence of R3 is taken together with R4 to form a double bond between the atoms to which R3 and R4 are attached; or both occurrences of R3 are taken together to form an oxo substituent;
R4 is taken together with one occurrence of R3 to form a double bond between the atoms to which R3 and R4 are attached;
R5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, Ci-6 alkyl, hydroxyl, Ci-6 alkoxyl, halo, or cyano;
R6 and R7 each represent independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, Ci-6 alkyl, Ci-6 haloalkyl, Ci-6 hydroxyalkyl, or -N(R9)2;
R8 and R9 each represent independently for each occurrence hydrogen, C1-4 alkyl, or C3-5 cycloalkyl; or two occurrences of R9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom;
A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p
X1 is C(H)2, C(R4)(H), or N(R4);
X2 is N or C(CN);
X3 is N or C(H);
X4 is C(O) or C(H)2;
L is a covalent bond or a C 1-4 bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or -C(O)-;
Y1, Y2, and Y3 represent independently a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R7)-, or -C(R7)2-; and m, n, and p are each independently 0, 1, 2, or 3.
2. The compound of claim 1, wherein the compound is a compound of Formula I.
3. The compound of claim 1 or 2, wherein X1 is C(H)2.
4. The compound of any one of claims 1-3, wherein X3 is N.
5. The compound of any one of claims 1-3, wherein X3 is C(H).
6. The compound of any one of claims 1-5, wherein X4 is C(O).
7. The compound of any one of claims 1-5, wherein X4 is C(H)2.
8. The compound of any one of claims 1-7, wherein R3 is hydrogen.
9. The compound of any one of claims 1-7, wherein both occurrences of R3 are taken together to form an oxo substituent.
R1 is naphthyl; an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with m occurrences of R5;
R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci-6 aliphatic; each of which is substituted with n occurrences of R6;
R3 is CM alkyl;
R4 is hydrogen, halo, or CM alkyl;
R5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, Ci-6 alkyl, hydroxyl, Ci-6 alkoxyl, halo, or cyano;
R6 and R7 each represent independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, Ci-6 alkyl, Ci-6 haloalkyl, Ci-6 hydroxyalkyl, or -N(R9)2;
R8 and R9 each represent independently for each occurrence hydrogen, CM alkyl, or C3-5 cycloalkyl; or two occurrences of R9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom;
A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p
X1 is C(H) or N;
X2 is N, C(CN), or C(R4);
L is a covalent bond or a CM bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or -C(O)-;
Y1, Y2, and Y3 represent independently a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R7)-, or -C(R7)2-; and m, n, and p are each independently 0, 1, 2, or 3.
11. The compound of claim 10, wherein the compound is a compound of Formula II.
12. The compound of claim 10 or 11, wherein X1 is C(H).
R1 is naphthyl; an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; or phenyl; each of which is substituted with m occurrences of R5;
R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; phenyl; or Ci-6 aliphatic; each of which is substituted with n occurrences of R6;
R4 is hydrogen, halo, or Ci-4 alkyl;
R5 represents independently for each occurrence C2-6 alkynyl, C2-6 alkenyl, Ci-6 alkyl, hydroxyl, Ci-6 alkoxyl, halo, or cyano;
R6 and R7 each represent independently for each occurrence halo, hydroxyl, Ci-6 alkoxyl, Ci-6 haloalkoxyl, Ci-6 alkyl, Ci-6 haloalkyl, Ci-6 hydroxyalkyl, or -N(R9)2;
R8 and R9 each represent independently for each occurrence hydrogen, C1-4 alkyl, or C3-5 cycloalkyl; or two occurrences of R9 attached to the same nitrogen atom are taken together with the nitrogen atom to which they are attached to form a 4-7 membered saturated ring having one nitrogen atom;
A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated spirocyclic heterocyclyl having 1 or 2 nitrogen atoms; a 6-11 membered saturated ortho- fused heterocyclyl having 1 or 2 nitrogen atoms; or a 5-6 membered monocyclic heteroaryl having one nitrogen atom and 0, 1, or 2 additional heteroatoms independently selected from nitrogen, oxygen, and sulfur; each of which is substituted with p
X2 is N, C(CN), or C(R4);
L is a covalent bond or a CM bivalent saturated or unsaturated, straight or branched hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -N(R9)-, or -C(O)-;
Y1, Y2, and Y3 represent independently a C1-3 bivalent saturated or unsaturated, straight hydrocarbon chain wherein one or two methylene units of the chain are optionally and independently replaced by -0-, -C(H)(R7)-, or -C(R7)2-; and m, n, and p are each independently 0, 1, 2, or 3.
14. The compound of claim 13, wherein the compound is a compound of Formula III.
15. The compound of any one of claims 10-14, wherein X2 is C(R4).
16. The compound of any one of claims 10-15, wherein R4 is hydrogen.
17. The compound of any one of claims 1-14, wherein X2 is N.
18. The compound of any one of claims 1-14, wherein X2 is C(CN).
19. The compound of any one of claims 1-18, wherein R1 is naphthyl substituted with m occurrences of R5.
20. The compound of any one of claims 1-18, wherein R1 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with m occurrences of R5.
21. The compound of any one of claims 1-18, wherein R1 is an 8-10 membered bicyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with m occurrences of R5.
22. The compound of any one of claims 1-21, wherein R2 is an 8-10 membered saturated or partially unsaturated bicyclic heterocyclyl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6.
24. The compound of any one of claims 1-21, wherein R2 is a 3-7 membered saturated or partially unsaturated monocyclic heterocyclyl having 1 or 2 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heterocyclyl is substituted with n occurrences of R6.
25. The compound of any one of claims 1-21, wherein R2 is a 5-6 membered monocyclic heteroaryl having 1, 2, or 3 heteroatoms independently selected from nitrogen, oxygen, and sulfur; wherein said heteroaryl is substituted with n occurrences of R6.
26. The compound of any one of claims 1-25, wherein L is -O-CH2-, -0-(CH2)2-, -N(R9)-CH2-, or -N(R9)-(CH2)2-, wherein the terminal carbon atom of L is attached to R2.
27. The compound of any one of claims 1-25, wherein L is -O-CH2-, wherein the carbon atom of L is attached to R2.
29. The compound of any one of claims 1-28, wherein Y1 is -(CH2)-, -(CH2)2-, -CH2- C(H)(OH)-, -(CH2)3-, -CH2-O-CH2-, or -CH2-CF2-CH2-.
31. The compound of any one of claims 1-27 or 30, wherein Y2 is -(CH2)-, -(CH2)2-, -CH2- C(H)(OH)-, -(CH2)3-, -CH2-O-CH2-, or -CH2-CF2-CH2-.
32. The compound of any one of claims 1-31, wherein R8 is hydrogen.
33. The compound of any one of claims 1-27, wherein A1 is a 4-7 membered saturated monocyclic heterocyclyl having 1 or 2 nitrogen atoms; wherein said heterocyclyl is substituted with p occurrences of R7.
34. A compound in Table 1, 1-A, 2, or 3 herein, or a pharmaceutically acceptable salt thereof.
35. A pharmaceutical composition comprising a compound of any one of claims 1-34 and a pharmaceutically acceptable carrier.
36. A method of treating a disorder mediated by K-Ras G12D, comprising administering a therapeutically effective amount of a compound of any one of claims 1-34 to a subject in need thereof to treat the disorder.
37. The method of claim 36, wherein the disorder is cancer.
38. The method of claim 37, wherein the cancer is lung cancer, pancreatic cancer, colorectal cancer, breast cancer, cervical cancer, prostate cancer, gastric cancer, skin cancer, liver cancer, bile duct cancer, or nervous system cancer.
39. The method of claim 37, wherein the cancer is non-small cell lung cancer (NSCLC) or pancreatic cancer.
40. The method of any one of claims 36-39, wherein the subject is a human.
41. A method of inhibiting K-Ras G12D activity, comprising contacting K-Ras G12D with an effective amount of a compound of any one of claims 1-34 to inhibit K-Ras G12D activity.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202163173274P | 2021-04-09 | 2021-04-09 | |
| US63/173,274 | 2021-04-09 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022217042A1 true WO2022217042A1 (en) | 2022-10-13 |
Family
ID=83545779
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2022/024006 Ceased WO2022217042A1 (en) | 2021-04-09 | 2022-04-08 | Naphthyl-substituted quinoline-4(1h)-ones and related compounds and their use in treating medical conditions |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2022217042A1 (en) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN116120315A (en) * | 2023-04-19 | 2023-05-16 | 山东绿叶制药有限公司 | KRAS G12C inhibitor and application thereof |
| WO2023198191A1 (en) * | 2022-04-15 | 2023-10-19 | 杭州多域生物技术有限公司 | Six- and six-membered compound, preparation method, pharmaceutical composition, and application |
| WO2024115890A1 (en) * | 2022-11-28 | 2024-06-06 | Redx Pharma Plc | Compounds |
| WO2024243025A1 (en) * | 2023-05-19 | 2024-11-28 | Ikena Oncology, Inc. | Naphthyl-substituted pyrido[4,3-d]pyrimidines and related compounds and their use in treating medical conditions |
| WO2025080592A1 (en) | 2023-10-09 | 2025-04-17 | Incyte Corporation | Combination comprising a kras g12d inhibitor and an egfr inhibitor for use in the treatment of cancer |
| WO2025080593A1 (en) | 2023-10-09 | 2025-04-17 | Incyte Corporation | Combination therapy using a kras g12d inhibitor and pd-1 inhibitor or pd-l1 inhibitor |
| US12448400B2 (en) | 2023-09-08 | 2025-10-21 | Gilead Sciences, Inc. | KRAS G12D modulating compounds |
Citations (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009047255A1 (en) * | 2007-10-09 | 2009-04-16 | Ucb Pharma, S.A. | Heterobicyclic compounds as histamine h4-receptor antagonists |
| WO2017201161A1 (en) * | 2016-05-18 | 2017-11-23 | Mirati Therapeutics, Inc. | Kras g12c inhibitors |
| US20190144444A1 (en) * | 2017-11-15 | 2019-05-16 | Mirati Therapeutics, Inc. | Kras g12c inhibitors |
| CN112142735A (en) * | 2020-04-09 | 2020-12-29 | 上海凌达生物医药有限公司 | Condensed cyanopyridine compound, preparation method and application |
| CN112430234A (en) * | 2019-08-26 | 2021-03-02 | 信达生物制药(苏州)有限公司 | Novel KRAS G12C protein inhibitor and preparation method and application thereof |
| WO2021041671A1 (en) * | 2019-08-29 | 2021-03-04 | Mirati Therapeutics, Inc. | Kras g12d inhibitors |
| US20210094919A1 (en) * | 2019-09-25 | 2021-04-01 | Jacobio Pharmaceuticals Co., Ltd. | Kras mutant protein inhibitors |
| CN112830928A (en) * | 2019-11-22 | 2021-05-25 | 四川海思科制药有限公司 | Pyrimido-cyclic derivative and application thereof in medicine |
| WO2021102288A1 (en) * | 2019-11-22 | 2021-05-27 | Senda Biosciences, Inc. | Pyridopyrimidinone derivatives as ahr antagonists |
| WO2021108683A1 (en) * | 2019-11-27 | 2021-06-03 | Revolution Medicines, Inc. | Covalent ras inhibitors and uses thereof |
| WO2021107160A1 (en) * | 2019-11-29 | 2021-06-03 | Taiho Pharmaceutical Co., Ltd. | A compound having inhibitory activity against kras g12d mutation |
| WO2021210970A1 (en) * | 2020-04-17 | 2021-10-21 | Dong-A St Co., Ltd. | Pyridopyrimidinone derivatives and their use as aryl hydrocarbon receptor modulators |
| CN113683616A (en) * | 2020-05-18 | 2021-11-23 | 广州百霆医药科技有限公司 | KRAS G12C mutein inhibitors |
| WO2021244603A1 (en) * | 2020-06-04 | 2021-12-09 | Shanghai Antengene Corporation Limited | Inhibitors of kras g12c protein and uses thereof |
| WO2022042630A1 (en) * | 2020-08-26 | 2022-03-03 | InventisBio Co., Ltd. | Heteroaryl compounds, preparation methods and uses thereof |
| WO2022066646A1 (en) * | 2020-09-22 | 2022-03-31 | Mirati Therapeutics, Inc. | Kras g12d inhibitors |
| WO2022068921A1 (en) * | 2020-09-30 | 2022-04-07 | 上海医药集团股份有限公司 | Quinazoline compound and application thereof |
-
2022
- 2022-04-08 WO PCT/US2022/024006 patent/WO2022217042A1/en not_active Ceased
Patent Citations (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009047255A1 (en) * | 2007-10-09 | 2009-04-16 | Ucb Pharma, S.A. | Heterobicyclic compounds as histamine h4-receptor antagonists |
| WO2017201161A1 (en) * | 2016-05-18 | 2017-11-23 | Mirati Therapeutics, Inc. | Kras g12c inhibitors |
| US20190144444A1 (en) * | 2017-11-15 | 2019-05-16 | Mirati Therapeutics, Inc. | Kras g12c inhibitors |
| CN112430234A (en) * | 2019-08-26 | 2021-03-02 | 信达生物制药(苏州)有限公司 | Novel KRAS G12C protein inhibitor and preparation method and application thereof |
| WO2021041671A1 (en) * | 2019-08-29 | 2021-03-04 | Mirati Therapeutics, Inc. | Kras g12d inhibitors |
| US20210094919A1 (en) * | 2019-09-25 | 2021-04-01 | Jacobio Pharmaceuticals Co., Ltd. | Kras mutant protein inhibitors |
| WO2021102288A1 (en) * | 2019-11-22 | 2021-05-27 | Senda Biosciences, Inc. | Pyridopyrimidinone derivatives as ahr antagonists |
| CN112830928A (en) * | 2019-11-22 | 2021-05-25 | 四川海思科制药有限公司 | Pyrimido-cyclic derivative and application thereof in medicine |
| WO2021108683A1 (en) * | 2019-11-27 | 2021-06-03 | Revolution Medicines, Inc. | Covalent ras inhibitors and uses thereof |
| WO2021107160A1 (en) * | 2019-11-29 | 2021-06-03 | Taiho Pharmaceutical Co., Ltd. | A compound having inhibitory activity against kras g12d mutation |
| CN112142735A (en) * | 2020-04-09 | 2020-12-29 | 上海凌达生物医药有限公司 | Condensed cyanopyridine compound, preparation method and application |
| WO2021210970A1 (en) * | 2020-04-17 | 2021-10-21 | Dong-A St Co., Ltd. | Pyridopyrimidinone derivatives and their use as aryl hydrocarbon receptor modulators |
| CN113683616A (en) * | 2020-05-18 | 2021-11-23 | 广州百霆医药科技有限公司 | KRAS G12C mutein inhibitors |
| WO2021244603A1 (en) * | 2020-06-04 | 2021-12-09 | Shanghai Antengene Corporation Limited | Inhibitors of kras g12c protein and uses thereof |
| WO2022042630A1 (en) * | 2020-08-26 | 2022-03-03 | InventisBio Co., Ltd. | Heteroaryl compounds, preparation methods and uses thereof |
| WO2022066646A1 (en) * | 2020-09-22 | 2022-03-31 | Mirati Therapeutics, Inc. | Kras g12d inhibitors |
| WO2022068921A1 (en) * | 2020-09-30 | 2022-04-07 | 上海医药集团股份有限公司 | Quinazoline compound and application thereof |
Non-Patent Citations (2)
| Title |
|---|
| FELL, J. B ET AL.: "Identification of the Clinical Development Candidate MRTX849, a Covalent KRASG12C Inhibitor for the Treatment of Cancer", JOURNAL OF MEDICINAL CHEMISTRY, vol. 63, 2020, pages 6679 - 6693, XP055788094, DOI: 10.1021/acs.jmedchem.9b02052 * |
| PAIN, C. ET AL.: "Synthesis of 5-substituted 2-(4- or 3-methoxyphenyl)-4(1H)-quinolones", TETRAHEDRON, vol. 59, 2003, pages 9627 - 9633, XP085016395, DOI: 10.1016/j.tet.2003.09.094 * |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023198191A1 (en) * | 2022-04-15 | 2023-10-19 | 杭州多域生物技术有限公司 | Six- and six-membered compound, preparation method, pharmaceutical composition, and application |
| WO2024115890A1 (en) * | 2022-11-28 | 2024-06-06 | Redx Pharma Plc | Compounds |
| CN116120315A (en) * | 2023-04-19 | 2023-05-16 | 山东绿叶制药有限公司 | KRAS G12C inhibitor and application thereof |
| CN116120315B (en) * | 2023-04-19 | 2023-06-09 | 山东绿叶制药有限公司 | KRAS G12C inhibitor and application thereof |
| WO2024243025A1 (en) * | 2023-05-19 | 2024-11-28 | Ikena Oncology, Inc. | Naphthyl-substituted pyrido[4,3-d]pyrimidines and related compounds and their use in treating medical conditions |
| US12448400B2 (en) | 2023-09-08 | 2025-10-21 | Gilead Sciences, Inc. | KRAS G12D modulating compounds |
| WO2025080592A1 (en) | 2023-10-09 | 2025-04-17 | Incyte Corporation | Combination comprising a kras g12d inhibitor and an egfr inhibitor for use in the treatment of cancer |
| WO2025080593A1 (en) | 2023-10-09 | 2025-04-17 | Incyte Corporation | Combination therapy using a kras g12d inhibitor and pd-1 inhibitor or pd-l1 inhibitor |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12403196B2 (en) | Ras inhibitors | |
| WO2022217042A1 (en) | Naphthyl-substituted quinoline-4(1h)-ones and related compounds and their use in treating medical conditions | |
| WO2024064335A1 (en) | Naphthyl-substituted pyranopyrimidinones and related compounds and their use in treating medical conditions | |
| AU2021344830A2 (en) | Use of SOS1 inhibitors to treat malignancies with SHP2 mutations | |
| CN114867735A (en) | RAS inhibitors | |
| CA3050188C (en) | Imidazopyrazine compounds, preparation methods and uses thereof | |
| CN115916194A (en) | Methods for delaying, preventing and treating acquired resistance to RAS inhibitors | |
| CN106536503A (en) | Tyrosine kinase inhibitor and uses thereof | |
| WO2022051569A1 (en) | Substituted 3-piperidinyl-pyrrolo[2,3-b]pyridines and related compounds and their use in treating medical conditions | |
| WO2023059609A1 (en) | Heterobifunctional compounds and their use in treating disease | |
| CN116600808A (en) | Tetrahydronaphthyridine derivative serving as KRAS mutant G12C inhibitor, and preparation method and application thereof | |
| WO2022051567A1 (en) | Substituted pyrido[2,3-b]pyrazinones and reuated compounds and their use in treating medicau conditions | |
| US20240238424A1 (en) | Heterobifunctional compounds and methods of treating disease | |
| TW202332429A (en) | Therapeutic compounds and methods of use | |
| ES3039638T3 (en) | Melanocortin 4 receptor antagonists and uses thereof | |
| WO2023059582A1 (en) | Heterobifunctional compounds and their use in treating disease | |
| WO2023059605A1 (en) | Heterobifunctional compounds and their use in treating disease | |
| CN115707705A (en) | Aromatic heterocyclic compound, preparation method and application thereof | |
| JP2023503322A (en) | Novel compounds for treating diseases associated with DUX4 expression | |
| JP7808616B2 (en) | Cyclic compounds and methods of using same | |
| US20250136611A1 (en) | Heterobifunctional compounds and methods of treating disease | |
| WO2025050016A1 (en) | Tricyclic quinolone bcl6 bifunctional degraders | |
| WO2025222158A1 (en) | Compounds and uses thereof | |
| WO2025147514A1 (en) | Piperidinyl-methyl-purineamine nsd2 inhibitor therapeutic methods and compositions for treating cancer | |
| WO2025160079A1 (en) | Indazolo-amino-pyrimidinone compounds |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22785517 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 22785517 Country of ref document: EP Kind code of ref document: A1 |