WO2022040469A1 - Spiro compounds as kras inhibitors - Google Patents
Spiro compounds as kras inhibitors Download PDFInfo
- Publication number
- WO2022040469A1 WO2022040469A1 PCT/US2021/046773 US2021046773W WO2022040469A1 WO 2022040469 A1 WO2022040469 A1 WO 2022040469A1 US 2021046773 W US2021046773 W US 2021046773W WO 2022040469 A1 WO2022040469 A1 WO 2022040469A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- substituted
- group
- ring
- groups
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 CC(CCCC=C[*@](C)*CCCCC*C1)C1N Chemical compound CC(CCCC=C[*@](C)*CCCCC*C1)C1N 0.000 description 32
- RLWGSYMGUGNMLE-UHFFFAOYSA-N CC(C)(NC)N1CCNCC1 Chemical compound CC(C)(NC)N1CCNCC1 RLWGSYMGUGNMLE-UHFFFAOYSA-N 0.000 description 1
- BIVMIOYYNOGXDY-SECBINFHSA-N CC(C)(NC)N1C[C@@H](CC#N)NCC1 Chemical compound CC(C)(NC)N1C[C@@H](CC#N)NCC1 BIVMIOYYNOGXDY-SECBINFHSA-N 0.000 description 1
- YCODLESHHLJCFX-JTQLQIEISA-N CC(C)OCC[C@H]1N(C)CCC1 Chemical compound CC(C)OCC[C@H]1N(C)CCC1 YCODLESHHLJCFX-JTQLQIEISA-N 0.000 description 1
- XSAUQIUBMRFHTH-UHFFFAOYSA-N CC(C)OCc1c[n](C[IH]OCCCN2CCOCC2)cn1 Chemical compound CC(C)OCc1c[n](C[IH]OCCCN2CCOCC2)cn1 XSAUQIUBMRFHTH-UHFFFAOYSA-N 0.000 description 1
- QCPGHYUINVUVOR-IHXHZNKUSA-N CC(C[F]C1CN(C)C(COS)C1)OC[C@H](C1)N(C)CC1F Chemical compound CC(C[F]C1CN(C)C(COS)C1)OC[C@H](C1)N(C)CC1F QCPGHYUINVUVOR-IHXHZNKUSA-N 0.000 description 1
- NNBQJEGXDNOJOB-UHFFFAOYSA-N CC(Cc(c(C1)n2)c(N(CC3)CC(CC#N)N3C(C=C)=O)nc2OCC(C2)N(C)CC2F)C1(Cc1c2c(C)c[s]1)NC2=O Chemical compound CC(Cc(c(C1)n2)c(N(CC3)CC(CC#N)N3C(C=C)=O)nc2OCC(C2)N(C)CC2F)C1(Cc1c2c(C)c[s]1)NC2=O NNBQJEGXDNOJOB-UHFFFAOYSA-N 0.000 description 1
- RWMILEFOJIOVFF-UHFFFAOYSA-N CC(Cc(c(C1)n2)c(N(CC3)CC(CC#N)N3C(C=C)=O)nc2OCC(C2)N(C)CC2F)C1(Cc1c2ccc(C)c1)NC2=O Chemical compound CC(Cc(c(C1)n2)c(N(CC3)CC(CC#N)N3C(C=C)=O)nc2OCC(C2)N(C)CC2F)C1(Cc1c2ccc(C)c1)NC2=O RWMILEFOJIOVFF-UHFFFAOYSA-N 0.000 description 1
- IMOSVODAZXDWPN-UHFFFAOYSA-N CC(NC)N(CC1C2)CC2N1N(C)C Chemical compound CC(NC)N(CC1C2)CC2N1N(C)C IMOSVODAZXDWPN-UHFFFAOYSA-N 0.000 description 1
- DZZVHQHELMADSH-SSDOTTSWSA-N CCN1C[C@@H](C)NCC1 Chemical compound CCN1C[C@@H](C)NCC1 DZZVHQHELMADSH-SSDOTTSWSA-N 0.000 description 1
- JNPARXPEIYOFGQ-JTQLQIEISA-N CC[C@@H](CN(CC1)C(C)(C)C)N1N(C)N Chemical compound CC[C@@H](CN(CC1)C(C)(C)C)N1N(C)N JNPARXPEIYOFGQ-JTQLQIEISA-N 0.000 description 1
- ZBVKAQBAYPSQLX-RQJHMYQMSA-N CN(C1)[C@H](COC)C[C@H]1F Chemical compound CN(C1)[C@H](COC)C[C@H]1F ZBVKAQBAYPSQLX-RQJHMYQMSA-N 0.000 description 1
- DBCXCUYGMLZUNT-YFKPBYRVSA-N CN([C@@H](COC)CO1)C1=O Chemical compound CN([C@@H](COC)CO1)C1=O DBCXCUYGMLZUNT-YFKPBYRVSA-N 0.000 description 1
- DQWZAGFXJVIOMU-UHFFFAOYSA-N CN1C(COC)CCC1 Chemical compound CN1C(COC)CCC1 DQWZAGFXJVIOMU-UHFFFAOYSA-N 0.000 description 1
- XPEMFBZYHOMBNM-UHFFFAOYSA-N CN1C(COS)CCC1 Chemical compound CN1C(COS)CCC1 XPEMFBZYHOMBNM-UHFFFAOYSA-N 0.000 description 1
- DQWZAGFXJVIOMU-ZETCQYMHSA-N CN1[C@H](COC)CCC1 Chemical compound CN1[C@H](COC)CCC1 DQWZAGFXJVIOMU-ZETCQYMHSA-N 0.000 description 1
- LEMKXWWLBRHGAZ-UHFFFAOYSA-N COC(C(CCC1(C2)Cc3ccccc3CC1)C2=O)=O Chemical compound COC(C(CCC1(C2)Cc3ccccc3CC1)C2=O)=O LEMKXWWLBRHGAZ-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing three or more hetero rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/10—Spiro-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D491/00—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00
- C07D491/02—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00 in which the condensed system contains two hetero rings
- C07D491/10—Spiro-condensed systems
- C07D491/107—Spiro-condensed systems with only one oxygen atom as ring hetero atom in the oxygen-containing ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D495/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms
- C07D495/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms in which the condensed system contains two hetero rings
- C07D495/10—Spiro-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D495/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms
- C07D495/12—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms in which the condensed system contains three hetero rings
- C07D495/20—Spiro-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D498/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D498/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms in which the condensed system contains two hetero rings
- C07D498/10—Spiro-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D498/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D498/12—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms in which the condensed system contains three hetero rings
- C07D498/20—Spiro-condensed systems
Definitions
- the mitogen-activated protein kinase (MAPK) signaling pathway controls many essential cellular functions, such as cytoskeleton organization, aging and programmed cell death, calcium signaling, trafficking of vesicles, cellular proliferation, and cell division.
- MAPK mitogen-activated protein kinase
- FIG. 1 The mitogen-activated protein kinase (MAPK) signaling pathway controls many essential cellular functions, such as cytoskeleton organization, aging and programmed cell death, calcium signaling, trafficking of vesicles, cellular proliferation, and cell division.
- MAPK pathway plays a role in certain cancers.
- cell proliferation has been found to be a critical component of the MAPK pathway, because when perturbed it leads to many different types of solid tumors as well as blood cancers.
- Available FDA-approved drugs that function in the MAPK pathway generally target BRAF and MEK, and are estimated to exceed $10 billion per year with the right combination of drugs.
- these drugs that target the BRAF and MEK proteins have either
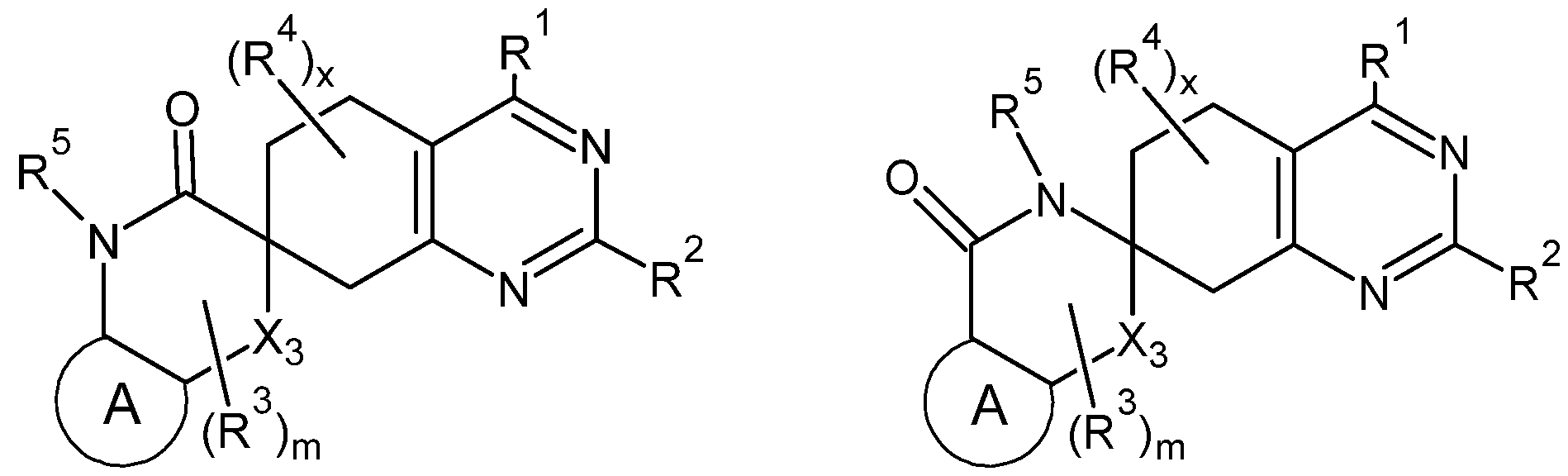
- a compound capable of inhibiting a RAS protein form comprises a compound represented by Formula 1.
- Two geminal R 3 s or two geminal R 4 s bonded to a common carbon atom may optionally combine to form an additional spirocycle from the common carbon atom.
- Each of X1-X3 is independently a bond, a moiety containing a ring C atom, or a moiety containing a ring heteroatom.
- the ring A is a cyclic, heterocyclic, aryl or heteroaryl ring.
- each of x and m is an integer from 1 to 6.
- R 1 may be represented by one of Formulae 4A and 4B:
- X 4 may be selected from substituted or unsubstituted, saturated or partially saturated monocyclic, bridged or spirocyclic rings having 2 nitrogen ring atoms.
- R 14 may be represented by one of Formulae 5A through 5E:
- the compound of Formula 1 may be represented by one of Formulae 2A to 2B:
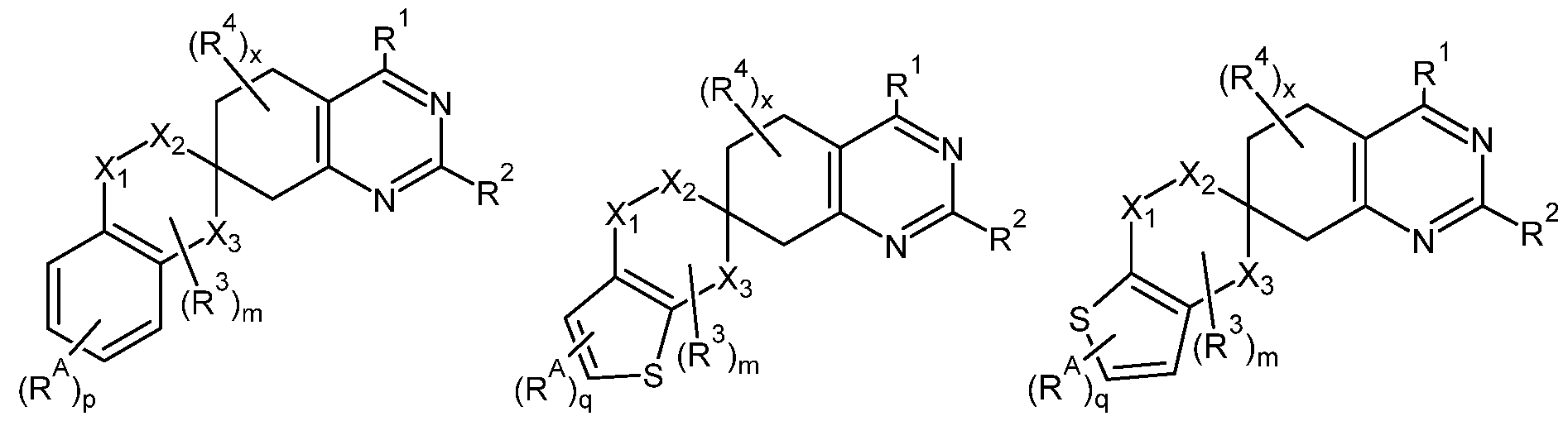
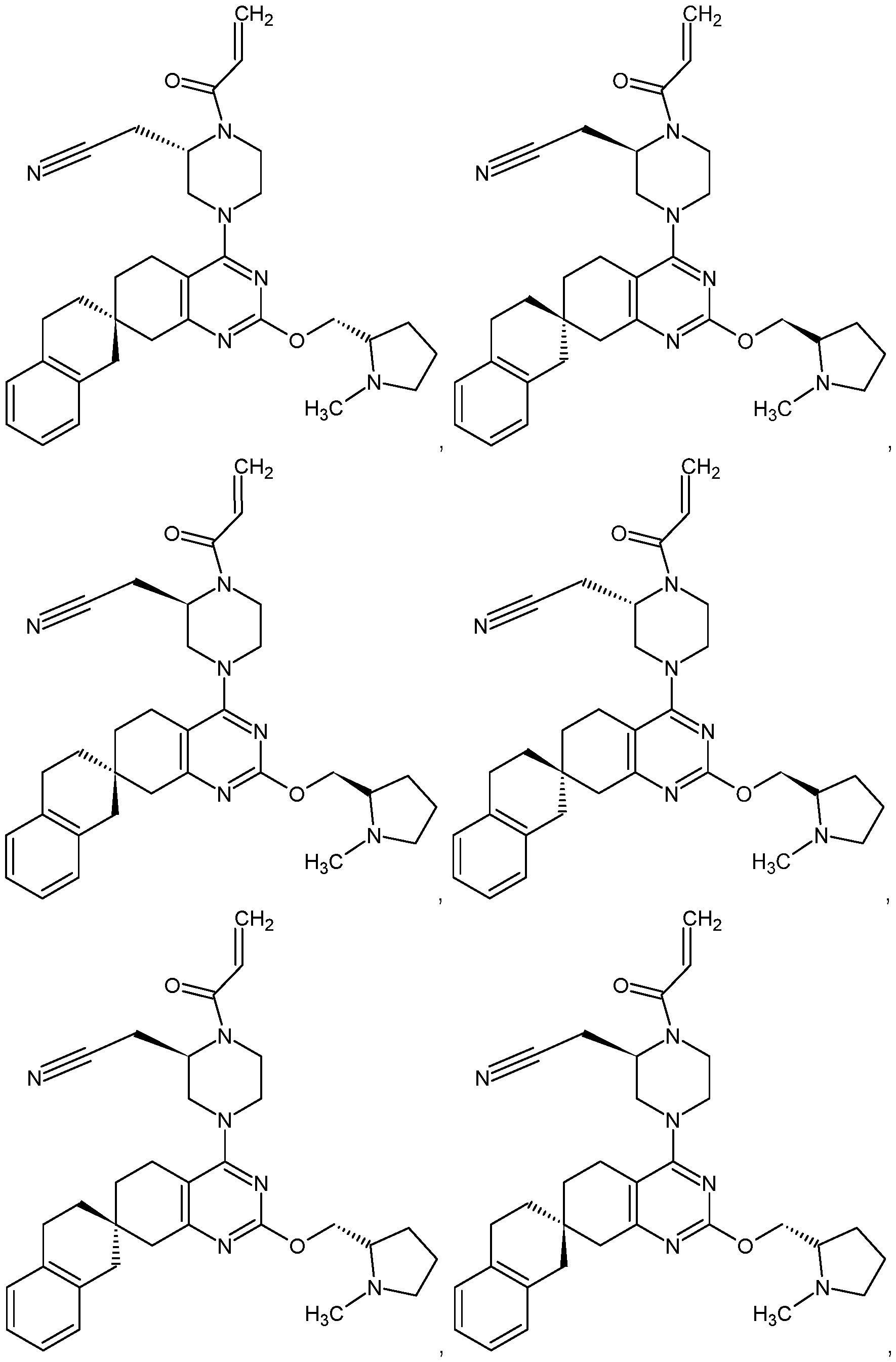
- the ring A may be a substituted or unsubstituted aryl ring or a substituted or unsubstituted heteroaryl ring. And in some embodiments, the ring A has 5 or 6 ring atoms. In some embodiments, the ring A may be selected from benzene, thiophene, furan, pyran, thiopyran, pyrrole, imidazole, pyrazole, triazole, pyridine, pyrimidine, pyridazine, pyrazine, oxazole, thiazole and cyclopentadiene. [0012] In some embodiments, the compound of Formula 1 may be selected from one of the following compounds:
- a pharmaceutical composition comprises at least one compound represented by Formula 1 or a pharmaceutically acceptable salt or derivative thereof.
- the pharmaceutical composition may further comprise a pharmaceutically acceptable carrier, excipient, additive, adjuvant or diluent.
- a method of inhibiting a RAS protein form comprises contacting a cell in which inhibition is desired with at least one compound represented by Formula 1 or a pharmaceutically acceptable salt or derivative thereof.
- a method of inhibiting a RAS protein form comprises administering to a patient or subject a pharmaceutical composition comprising at least one compound represented by Formula 1 or a pharmaceutically acceptable salt or derivative thereof.
- a method of treating a disease or condition comprises administering to a patient or subject having the disease or condition at least one compound represented by Formula 1 or a pharmaceutically acceptable salt or derivative thereof.
- the disease or condition may be a cancer.
- a method of treating a disease or condition comprises administering to a patient or subject having the disease or condition a pharmaceutical composition comprising at least one compound represented by Formula 1 or a pharmaceutically acceptable salt or derivative thereof.
- the disease or condition may be a cancer.
- FIG. 1 is a schematic depicting the mitogen-activated protein kinase (MAPK) signaling pathway, reproduced from Nature Reviews, as indicated in the drawing.
- MAPK mitogen-activated protein kinase
- RAS is the last key protein involved in the signaling cascade in the MAPK pathway.
- a drug against RAS would create a new effective first-line treatment for certain cancers, e.g., lung, colon, and pancreatic cancer, and would provide new hope for patients with BRAF and MEK resistant tumors.
- cancers e.g., lung, colon, and pancreatic cancer
- Currently, 30% of all cancers display mutations in the MAPK pathway, including mutations in KRAS.
- drugs have been developed for other proteins (such as BRAF and MEK, as discussed above) in this pathway, there currently is no drug on the market to treat KRAS, which is the key driver of this pathway.
- R 3 and R 4 are not particularly limited, and may be any suitable hydrocarbon substituent known in this field.
- alkyl refers to straight and branched chain aliphatic groups.
- the length of the alkyl group is not particularly limited, but in some embodiments, the alkyl group may have from 1 to 12 carbon atoms.
- the alkyl group may be a lower alkyl group, i.e. , a straight or branched chain aliphatic group having from 1 to 8 carbon atoms, for example, 1 to 6 carbon atoms, or 1 to 3 carbon atoms.
- alkyl group When the alkyl group is substituted, it may be substituted with one or more substituents selected from substituted or unsubstituted hydrocarbon substituents, hydrogen atoms, F atoms, hydroxyl groups, cyano groups, amino groups, or oxo groups.
- substituents selected from substituted or unsubstituted hydrocarbon substituents, hydrogen atoms, F atoms, hydroxyl groups, cyano groups, amino groups, or oxo groups.
- suitable alkyl groups include methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, te/Y-butyl, pentyl, and hexyl, which may be unsubstituted or substituted as discussed above.
- alkyl groups include methyl (Me, -CH 3 ), ethyl (Et, -CH 2 CH 3 ), 1 -propyl (n-Pr, n-propyl, -CH 2 CH 2 CH 3 ), 2-propyl (i- Pr, i-propyl, -CH(CH 3 )2), i-butyl (n-Bu, n-butyl, -CH 2 CH 2 CH 2 CH 3 ), 2-methyl-1 -propyl (i-Bu, i-butyl, -CH 2 CH(CH 3 )2), 2-butyl (s-Bu, s-butyl, -CH(CH 3 )CH 2 CH 3 ), 2-methyl-2- propyl (t-Bu, t-butyl, -C(CH 3 )3), 1 -pentyl (n-pentyl, -CH 2 CH 2 CH 2 CH 3 ), 2-pentyl (n-CH 2 CH 2 CH 2 CH 3
- haloalkyl refers to an alkyl chain in which one or more hydrogen atoms have been replaced by a F atom.
- suitable haloalkyl groups include trifluoromethyl, difluoromethyl, and fluoromethyl.
- haloalkyloxy refers to a haloalkyl group bonded to an oxygen atom.
- the oxygen atom is bonded to the relevant position of Formula 1 .
- the haloalkyloxy group has the formula *-O-haloalkyl, where * represents the binding site to Formula 1 .
- alkoxy refers to an alkyl group bonded to an oxygen atom.
- the oxygen atom is bonded to the relevant position of Formula 1 .
- the alkoxy group has the formula *-O- alkyl, where * represents the binding site to Formula 1 .
- hydroxy a Iky I refers to an alkyl group bonded to a hydroxy group (-OH).
- a carbon atom of the alkyl group is bonded to the relevant position of Formula 1 .
- the hydroxyalkyl group has the formula *- alkyl-OH, where * represents the binding site to Formula 1 .
- dihydroxyalkyl refers to an alkyl group in which two carbon atoms of the alkyl chain are each substituted with a hydroxyl group.
- the alkylaminoalkyl group a carbon atom of one of the alkyl groups is bonded to the relevant position of Formula 1 .
- the alkylaminoalkyl group has the formula *-alkyl-NH-alkyl, where * represents the binding site to Formula 1.
- dialkylaminoalkyl refers to an alkyl group that has been substituted with a tertiary amino group.
- a carbon atom of one of the three alkyl groups (which may be the same or different from each other) is bonded to the relevant position of Formula 1 .
- the dialkylaminoalkyl group has the formula *-alkyl-N(R a iki)(Raik2), where * represents the binding site to Formula 1 , and Raiki and Raik2 represent alkyl groups which may be the same or different from each other.
- cycloalkyl refers to saturated and partially unsaturated cyclic hydrocarbon groups.
- the partially unsaturated cyclic groups are also referred to herein as “cycloalkenyls” or “cycloalkenyl groups” and are encompassed both in this definition of cycloalkyl as well as the definition herein of cycloalkenyl.
- the cycloalkyl group may have from 3 to 12 carbons, for example 3 to 8 carbons, or 3 to 6 carbons.
- cycloalkyl group When the cycloalkyl group is substituted, it may be substituted with one or more substituents selected from substituted or unsubstituted hydrocarbon substituents, hydrogen atoms, F atoms, hydroxyl groups, cyano groups, amino groups, or oxo groups.
- substituents selected from substituted or unsubstituted hydrocarbon substituents, hydrogen atoms, F atoms, hydroxyl groups, cyano groups, amino groups, or oxo groups.
- suitable cycloalkyl groups include cyclopropyl, cyclobutyl, cyclopentyl, cyclopentenyl, cyclohexyl, cyclohexenyl, cycloheptyl, and cyclooctyl.
- cycloalkyl also encompasses bridged cyclic groups, such as, for example, bicyclo[1 .1 ,1]pentanyl.
- monocyclic cycloalkyls include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl, cyclodecyl, cycloundecyl and cyclododecyl.
- bicyclic cycloalkyls include [4,4], [4,5], [5,5], [5,6] or [6,6] ring systems.
- heteroalkyl refers to an alkyl group, as defined herein, in which one or more carbon atoms in the chain are replaced by a heteroatom.
- the heteroatom is not particularly limited, and may be any atom other than carbon or nitrogen. In some embodiments, for example, the heteroatom may be selected from O, S, and N.
- the “aryl” group is an aromatic moiety having one or more aromatic rings, for example, from 1 to 3 aromatic rings. Each of the rings of the aryl group may optionally be substituted, as noted generally above. In some embodiments, the aryl group may have from 6 to 14 ring carbon atoms, for example, from 6 to 10 ring carbon atoms.
- aralkyl and arylalkyl refer to an aryl group covalently linked to an alkyl group. Either the alkyl group or the aryl group, or both, may be substituted as noted generally above. In contrast to an aryl group substituted with an alkyl group (in which the aryl group is bonded to the relevant position of Formula 1 ), in the aralkyl group, the alkyl component is bonded to the relevant position of Formula 1 .
- the aralkyl group has the formula *-alkyl-aryl, where * represents the binding site to Formula 1 .
- halo-arylalkyl or “halo-aralkyl” refers to an aryl alkyl group (or aralkyl group), as defined herein, substituted with at least one halogen atom. Either the alkyl component or the aryl component may be substituted with the halogen. However, when the alkyl component is substituted, the halogen atom is F. When the aryl component is substituted with the halogen, the halogen atom is not particularly limited, and may be any of F, Cl, I or Br. The halo-arylalkyl (or halo-aralkyl) group may be further substituted with any of the other substituents described herein.
- cycloalkenyl refers to a cyclic structure having at least one ring that is non-aromatic, and that has at least one carbon-carbon double bond.
- the ring structure of the cycloalkenyl group is not particularly limited, and may have any number of rings in any configuration.
- the cycloalkenyl group may include from 1 to 3 rings, which may be arranged relative to each other in any way.
- the rings of the group may be either bonded to each other or fused to each other, and in some embodiments, some rings of the group may be bonded while other rings may be fused.
- alkynyl refers to a linear or branched hydrocarbon group with at least one carbon-carbon triple bond.
- the size of the alkynyl group is not particularly limited. However, in some embodiments, the alkynyl group may have 2 to 18 carbon atoms, for example 2 to 13 carbon atoms, 2 to 12 carbon atoms, 2 to 10 carbon atoms, 2 to 6 carbon atoms, or 2 to 3 carbon atoms.
- the ring structure of the heterocyclic group is not particularly limited, and may have any number of rings in any configuration.
- the heterocyclic group may include from 1 to 3 rings, which may be arranged relative to each other in any way.
- the rings of the heterocyclic group may be either bonded to each other or fused to each other, and in some embodiments, some rings of the heterocycle may be bonded while other rings may be fused.
- the heterocyclic group may be spirocyclic or may have a bridged ring structure.
- the size of the heterocyclic group is also not particularly limited. However, in some embodiments, the heterocyclic group may have from 3 to 12 ring atoms, for example from 4 to 8 ring atoms.
- heterocyclic groups include epoxy, azetidinyl, aziridinyl, tetrahydrofuranyl, tetrahydropyranyl, pyrrolidinyl, pyrrolidinonyl, piperidinyl, piperazinyl, imidazolidinyl, thiazolidinyl, dithianyl, trithianyl, dioxolanyl, oxazolidinyl, oxazolidinonyl, decahydroquinolinyl, piperidonyl, 4-piperidinonyl, quinuclidinyl, thiomorpholinyl, thiomorpholinyl[1 , 1 ]dioxide, morpholinyl, azepanyl, oxazepanyl, azabicyclohexanes, azabicycloheptanes, azabicyclooctanes, azabicyclononanes
- heterocycloalkyl refers to a heterocyclic group, as defined herein, linked to an alkyl group. Either the alkyl group or the heterocyclic group, or both, may be substituted as noted generally above. In contrast to a heterocyclic group substituted with an alkyl group (in which the heterocyclic group is bonded to the relevant position of Formula 1), in the heterocycloalkyl group, the alkyl component is bonded to the relevant position of Formula 1 .
- the heterocycloalkyl group has the formula *-alkyl-heterocycle, where * represents the binding site to Formula 1 .
- heteroaryl refers to an aromatic moiety having one or more aromatic rings, in which at least one atom of at least one ring is a heteroatom. In some embodiments, for example, more than one atom of at least one ring, or more than one atom of more than one ring, may be a heteroatom.
- the heteroatom is not particularly limited, and may be any atom other than carbon or nitrogen. In some embodiments, for example, the heteroatom may be selected from O, S, and N. Any of the ring atoms (including the heteroatoms) may also be substituted, as generally noted above. When substituted, the heteroaryl group may be substituted with any halogen atom (e.g., F, I, Cl or Br) or any of the other substituents described herein.
- any halogen atom e.g., F, I, Cl or Br
- the size and configuration of the heteroaryl group is also not particularly limited, and the heteroaryl group may have any number of rings in any configuration.
- the heteroaryl group may include from 1 to 3 rings, which may be arranged relative to each other in any way.
- the rings of the heteroaryl group may be either bonded to each other or fused to each other, and in some embodiments, some rings of the group may be bonded while other rings may be fused.
- the aryl group may have from 5 to 14 ring atoms, for example 5, 6, 9, or 10 ring atoms, and/or 6, 10, or 14 electrons shared in a cyclic array.
- the heteroaryl group may include thienyl, furyl, imidazolyl, pyrazolyl, thiazolyl, isothiazolyl, oxazolyl, isoxazolyl, triazolyl, thiadiazolyl, oxadiazolyl, tetrazolyl, thiatriazolyl, oxatriazolyl, pyridyl, pyrimidyl, pyrazinyl, pyridazinyl, triazinyl, tetrazinyl, tetrazolo[1 ,5-b]pyridazinyl, imidazol[1 ,2-a]pyrimidinyl and purinyl, as well as benzo-fused derivatives, such as, for example benzoxazolyl, benzofuryl, benzothiazolyl, benzothiadiazolyl, benzotriazolyl, benzoimidazolyl,
- halo-heteroaryl refers to a heteroaryl group substituted with at least one halogen atom.
- the halogen atom substituent on the “halo-heteroaryl group” is not particularly limited, and in some embodiments, may be any of F, Cl, I or Br.
- the halo-heteroaryl group may be further substituted with any of the other substituents described herein.
- heteroaryl alkyl refers to a heteroaryl group, as defined herein, linked to an alkyl group. Either the alkyl group or the heteroaryl group, or both, may be substituted as noted generally above. In contrast to a heteroaryl group substituted with an alkyl group (in which the heteroaryl group is bonded to the relevant position of Formula 1 ), in the heteroarylalkyl group, the alkyl component is bonded to the relevant position of Formula 1 .
- the heteroarylalkyl group has the formula *-alkyl-heteroaryl, where * represents the binding site to Formula 1 .
- halo-heteroaryl alkyl refers to a heteroaryl alkyl group, as defined herein, substituted with at least one halogen atom. Either the alkyl component or the heteroaryl component may be substituted with the halogen. However, when the alkyl component is substituted, the halogen atom is F. When the heteroaryl component is substituted with the halogen, the halogen atom is not particularly limited, and may be any of F, Cl, I or Br. The halo-heteroaryl alkyl group may be further substituted with any of the other substituents described herein.
- each of X1-X3 is independently a bond, a moiety containing a ring C atom, or a moiety containing a ring heteroatom.
- the heteroatom is not particularly limited, and may be any suitable non-carbon, non-hydrogen atom.
- each of X1-X3 is independently a moiety containing a ring C atom or a ring heteroatom selected from N, O and S. Additionally, as shown in Formula 1 , the ring to which X1-X3 belong is non-aromatic.
- each of X1-X3 may be bonded within the ring either to each other (e.g., X1-X2) or to an adjacent ring carbon atom by a single or double bond, the ring containing X1-X3 remains non-aromatic.
- one or more of X1-X3 may independently be a bond such that the ring containing X1-X3 may be a 3 to 6 membered non-aromatic ring. In some embodiments, for example, when both Xi and X2 are a bond, they combine to form one bond to the ring A.
- the ring containing X1-X3 when X3 is also a bond, the ring containing X1-X3 is a 3-membered ring; and when X3 is not a bond (e.g., is a ring C atom or ring heteroatom), the ring containing X1-X3 is a 4-membered ring.
- any one or more of X1-X3 may be a bond (yielding a 3 to 5 membered ring), or none of X1-X3 may be a bond (yielding a 6 membered ring).
- X1-X3 moieties containing the ring C atoms or ring heteroatoms are not particularly limited, and may be any suitable such moiety.
- X3 may be an oxygen atom (-O-) or a divalent methyl (e.g., -CH 2 -, -CHF-, -CF 2 -, -CHRme-, -C(Rmei)(Rme2)-, or -CFRme- and in some embodiments -CH 2 -, -CHF- or -CF 2 -).
- a divalent methyl e.g., -CH 2 -, -CHF-, -CF 2 -, -CHRme-, -C(Rmei)(Rme2)-, or -CFRme- and in some embodiments -CH 2 -, -CHF- or -CF 2 -).
- each of R 1 -R 4 , A, x and m are as defined herein with respect to Formula 1 , and R 5 is equivalent to Ram described above with respect to the amino moiety.
- R 5 (or Ram) may be hydrogen or a lower alkyl group (unsubstituted or substituted with, e.g., one or more halogen atoms).
- suitable rings for the ring A include benzene, thiophene, furan, pyran, thiopyran, pyrrole, imidazole, pyrazole, triazole, pyridine, pyrimidine, pyridazine, pyrazine, oxazole, thiazole and cyclopentadiene.
- Formulae 3A through 3K depict certain example configurations of the fusion of the ring A to the spirocyclic core of Formula 1 , it is understood that the ring A may be fused to the spirocyclic core in any suitable configuration. Those of ordinary skill in the art would readily recognize the available fusion configurations, all of which are encompassed by this disclosure.
- some example alternative configurations include: 1 ) Formula 3D (furan) in which the O of the furan ring points downward rather than upward (depicted below as Formula 3L); 2) Formula 3E (pyran) in which the O atom is any other available ring position) (depicted below as Formulae 3M(i) through 3M(iii); 3) Formula 3F (thiopyran) in which the S is in any other available ring position) (depicted below as Formulae 3N(i) through 3N(iii)); 4) Formula 3G (imidazole) in which the N-R A1 moiety points upward rather than downward (depicted below as Formula 30); 5) Formula 3H (pyridine) in which the N atom is in any other available ring position (depicted below as Formulae 3P(i) through 3P(iii)); 6) Formula 3I (oxazole) in which the 0 atom points upward rather than downward (depicted below as Formula 3Q);
- each of R 1 -R 4 , x and m are as defined herein with respect to Formula 1 .
- R A , R A1 and R A2 may each independently be any halogen (e.g., F, I, Cl or Br) or any of the substituents described above in connection with R 3 -R 4 of Formula 1 .
- R A , R A1 and R A2 may be hydrogen, a halogen, or a lower alkyl group (unsubstituted or substituted with, e.g., one or more F atoms).
- p is an integer from 1 to 4 and q is an integer from 1 to 2.
- X4 may be selected from the same substituents described above with respect to R3 and R4 of Formula 1 . In some embodiments, however, X4 may be selected from 4 to 12 membered heterocyclic rings (or groups), for example, 4 to 7 membered heterocyclic rings (or groups).
- the heterocyclic group may be unsubstituted, or may optionally be substituted with one or more substituents, such as those described above in connection with R 3 and R 4 .
- each substituent may be independently selected from alkyl groups, cyano groups, cyanoalkyl groups, and hydroxyalkyl groups.
- two geminal substituents of the heterocyclic group may optionally combine together to form a 4 to 12 membered (or, in some embodiments, a 4 to 7 membered) spiroheterocyclic group.
- X4 may be a 4 to 7 membered heterocylcoamino group, which may be substituted or unsubstituted.
- X4 may be , wherein * represents binding sites to
- ⁇ represents binding sites to Formula 1 and R 14 of Formula 4A or 4B, and either of the in each of the above structures may be bonded to either Formula 1 or R 14 of Formula 4A or 4B. In some embodiments, however, the lower represents the binding site to Formula 1 , and the upper represents the binding site to R 14 .
- X4 may be selected from the following structures:
- R 14 in Formula 4A and 4B is also not particularly limited, and may be any of the substituents described above in connection with R 3 and R 4 of Formula 1 .
- R 14 may be represented by one of Formulae 5A through 5E.
- R 15 and R 16 may be H or F
- R 17 may be hydrogen, a halogen, a substituted or unsubstituted lower alkyl group (e.g., -CH 3 , -CH 2 CH 3 , -CF 3 , -CH 2 F, -CHF 2 , -CH 2 CF 3 , -CH 2 CHF 2 , -CH 2 CH 2 F, -CHFCF 3 , -CF 2 CF 3 , -CHFCHF 2 , -CF 2 CHF 2 , -CHFCH 2 F, or -CF 2 CH 2 F), a substituted or unsubstituted alkylamino group (e.g., in which the alkyl component is a lower alkyl), and a substituted or unsubstituted alkylaminoalkyl group (e.g., in which the alkyl component is a lower alkyl), and a substituted or unsubstituted alkylaminoal
- the * represents the binding site to X4 of Formula 4A or 4B.
- R 15 through R 17 and R 19 are as described above in connection with Formulae 5A through 5E and Formulae 6A through 6D, and the * represents the binding site to Formula 1 .
- R 11A , R 11B and R 11 C are equivalent to R 11 described above in connection with Formulae 6A through 6D.
- R 17 may be selected from substituted or unsubstituted lower alkyl groups, and substituted or unsubstituted haloalkyl groups (e.g., in which the alkyl component is a lower alkyl).
- R 18 may be hydrogen, or a halogen atom.
- suitable R 1 groups include the [0084] In some embodiments, for example, R 1 may be
- X 5 may be -O- or -NR X5 -
- R 20 may be a substituted or unsubstituted heterocyclic group, or a substituted or unsubstituted heterocycolalkyl group.
- the compound represented by Formula 1 may be a compound represented by one of Formula 11A, 11 B and 11 C.
- R 2 groups can be found in the discussion of -Y-R 2 in U.S. Patent Publication No. 2019/0270743 to Mirati Therapeutics, Inc. and Array BioPharma Inc., titled “KRAS G12C Inhibitors,” filed on May 14, 2019 and published on September 5, 2019, the entire content of which is incorporated herein by reference, and in the discussion of Z 2 and its optional substituents in WO 2020/035031 to Genentech, Inc. and F. Hoffman-La Roche AG, titled “Fused Ring Compounds,” published on February 20, 2020 and having an international filing date of August 15, 2019, the entire content of which is also incorporated herein by reference.
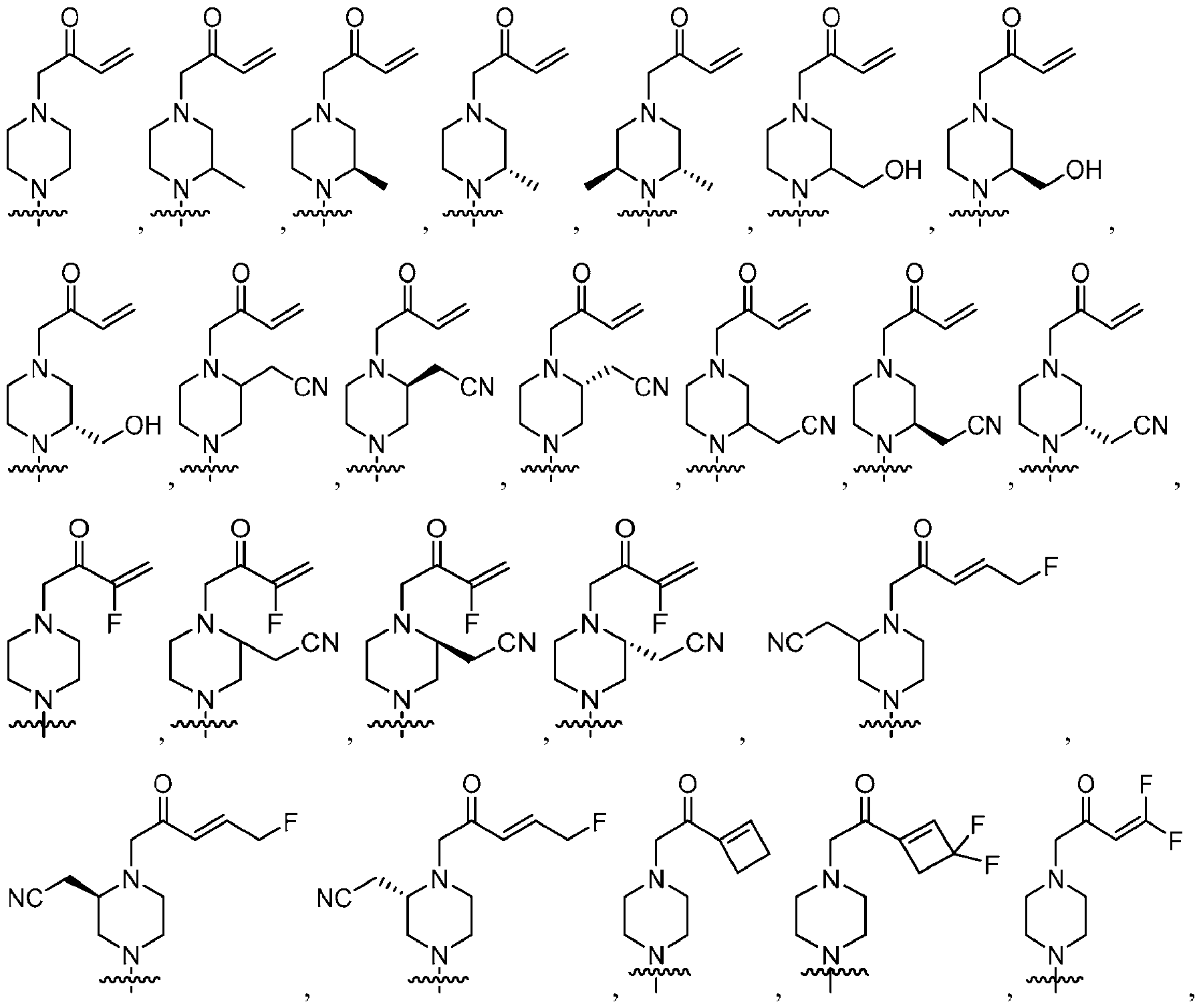
- suitable compounds satisfying Formula 1 include the following compounds:
- the compound represented by Formula 1 may be selected from the following compounds:
- a pharmaceutical composition may include one or more of the compounds described herein (or pharmaceutically acceptable salts or derivatives thereof) in a therapeutically effective amount.
- the pharmaceutical composition may include one or more compound represented by Formula 1 (or pharmaceutically acceptable salt thereof), as well as one or more pharmaceutically acceptable carriers, excipients, adjuvants and/or diluents. Acceptable carriers, excipients and diluents are well known in the art and can be selected with regard to the intended route of administration and standard practice.
- Some non-limiting examples include binders, lubricants, suspending agents, coating agents, solubilizing agents, preserving agents, wetting agents, emulsifiers, surfactants, sweeteners, colorants, flavoring agents, odorants, buffers, antioxidants, stabilizing agents and/or salts.
- Compounds of the present disclosure may be formulated by any method well known in the art and may be prepared for administration by any route, including, without limitation, parenteral, oral, sublingual, transdermal, topical, intranasal, intratracheal, or intrarectal.
- compounds of the invention are administered intravenously in a hospital setting.
- administration may be by the oral route.
- the compounds described herein may also be administered as pharmaceutically acceptable quaternary salts known to those skilled in the art, e.g., quaternary ammonium salts represented by -N(R)2- , wherein R is hydrogen, alkyl, or benzyl, and Z is a counterion (non-limiting examples of which include chloride, bromide, iodide, O-alkyl, toluenesulfonate, methylsulfonate, sulfonate, phosphate, or carboxylate (non-limiting examples of which include benzoate, succinate, acetate, glycolate, maleate, malate, citrate, tartrate, ascorbate, benzoate, cinnamoate, mandeloate, benzyloate, and diphenylacetate).
- R is hydrogen, alkyl, or benzyl
- Z is a counterion
- R is hydrogen, alkyl, or benzyl
- Z is a
- a method of inhibiting RAS protein forms includes contacting a cell in which inhibition is desired with an effective amount of one or more compounds represented by Formula 1 (or one or more pharmaceutically acceptable salts or derivatives thereof).
- the method of inhibiting RAS protein forms may include administering a pharmaceutical composition as described herein to a patient or subject in need of RAS protein form inhibition. Contacting the cell with the compound(s), salt(s) or derivative(s) thereof, or the pharmaceutical composition may occur in vivo or in vitro. The contacting (or administration) may be accomplished in a single dose or over multiple doses.
- the method of treating a condition or disease may further comprise first determining if the patient or subject to be treated has a KRAS mutation. If the patient or subject to be treated is determined to have such a mutation, then the method includes administering to the patient or subject the therapeutically effective amount of one or more compound(s) described herein, a pharmaceutically acceptable salt or derivative thereof, or a pharmaceutical composition described herein.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Compounds according to Formula 1 are capable of inhibiting a RAS protein form.
Description
SPIRO COMPOUNDS AS KRAS INHIBITORS
BACKGROUND
[0001] The mitogen-activated protein kinase (MAPK) signaling pathway (shown generally in FIG. 1 ) controls many essential cellular functions, such as cytoskeleton organization, aging and programmed cell death, calcium signaling, trafficking of vesicles, cellular proliferation, and cell division. Recent research suggests that this MAPK pathway plays a role in certain cancers. For example, cell proliferation has been found to be a critical component of the MAPK pathway, because when perturbed it leads to many different types of solid tumors as well as blood cancers. Available FDA-approved drugs that function in the MAPK pathway generally target BRAF and MEK, and are estimated to exceed $10 billion per year with the right combination of drugs. However, these drugs that target the BRAF and MEK proteins have either failed as single agents in certain cancers, or patients have developed resistance to their therapeutic effects within several months and the caner returns. Accordingly, there is a desire to develop new drugs against other targets in the MAPK pathway.
SUMMARY
[0002] According to embodiments of the present disclosure, a compound capable of inhibiting a RAS protein form comprises a compound represented by Formula 1. Formula 1
[0003] In Formula 1 , each of R1-R2 is independently selected from halogens, substituted or unsubstituted hydrocarbon substituents, hydrogen, deuterium, hydroxyl groups, cyano groups, amino groups, and oxo groups (=0). Each of R3-R4 is independently selected from substituted or unsubstituted hydrocarbon substituents, hydrogen, deuterium, F, hydroxyl groups, cyano groups, amino groups, or oxo groups (=0). Two geminal R3s or two geminal R4s bonded to a common carbon atom may optionally combine to form an additional spirocycle from the
common carbon atom. Each of X1-X3 is independently a bond, a moiety containing a ring C atom, or a moiety containing a ring heteroatom. The ring A is a cyclic, heterocyclic, aryl or heteroaryl ring. And each of x and m is an integer from 1 to 6.
[0004] In some embodiments, R1 may be represented by one of Formulae 4A and 4B:
[0005] In Formulae 4A and 4B, * represents a binding site to Formula 1. In Formula 4A, X4 may be selected from substituted or unsubstituted hydrocarbon substituents, hydrogen, deuterium, halogens, hydroxyl groups, cyano groups, amino groups, or oxo groups (=0). In Formula 4B, X4 may be selected from substituted or unsubstituted, saturated or partially saturated monocyclic, bridged or spirocyclic rings having 2 nitrogen ring atoms. And in Formula 4A and 4B, R14 may be selected from substituted or unsubstituted hydrocarbon substituents, hydrogen, deuterium, halogens, hydroxyl groups, cyano groups, amino groups, or oxo groups (=0).
[0006] In some embodiments, in Formulae 4A and 4B, R14 may be represented by one of Formulae 5A through 5E:
independently selected from substituted or unsubstituted hydrocarbon substituents, hydrogen, deuterium, halogens, hydroxyl groups, cyano groups, amino groups, or oxo groups (=0).
[0008] In some embodiments, in Formula 1 , each of X1-X3 may independently be a moiety containing a ring C atom or a ring heteroatom selected from N, 0 and S. [0009] According to some embodiments, the compound of Formula 1 may be represented by one of Formulae 1A to 1 H:
[0010] In some embodiments, the compound of Formula 1 may be represented by one of Formulae 2A to 2B:
In Formulae 2A and 2B, R5 is independently selected from substituted or unsubstituted hydrocarbon substituents, hydrogen, deuterium, F, hydroxyl groups, cyano groups, amino groups, or oxo groups (=0).
[0011] According to some embodiments, the ring A may be a substituted or unsubstituted aryl ring or a substituted or unsubstituted heteroaryl ring. And in some embodiments, the ring A has 5 or 6 ring atoms. In some embodiments, the ring A may be selected from benzene, thiophene, furan, pyran, thiopyran, pyrrole, imidazole, pyrazole, triazole, pyridine, pyrimidine, pyridazine, pyrazine, oxazole, thiazole and cyclopentadiene.
[0012] In some embodiments, the compound of Formula 1 may be selected from one of the following compounds:
[0013] In some embodiments, for example, the compound of Formula 1 may be represented by one of the following compounds:
[0014] According to some embodiments, a pharmaceutical composition comprises at least one compound represented by Formula 1 or a pharmaceutically acceptable salt or derivative thereof. In some embodiments, the pharmaceutical composition may further comprise a pharmaceutically acceptable carrier, excipient, additive, adjuvant or diluent.
[0015] In some embodiments, a method of inhibiting a RAS protein form comprises contacting a cell in which inhibition is desired with at least one compound represented by Formula 1 or a pharmaceutically acceptable salt or derivative thereof. [0016] According to some embodiments, a method of inhibiting a RAS protein form comprises administering to a patient or subject a pharmaceutical composition comprising at least one compound represented by Formula 1 or a pharmaceutically acceptable salt or derivative thereof.
[0017] In some embodiments, a method of treating a disease or condition comprises administering to a patient or subject having the disease or condition at
least one compound represented by Formula 1 or a pharmaceutically acceptable salt or derivative thereof. In some embodiments, the disease or condition may be a cancer.
[0018] According to some embodiments, a method of treating a disease or condition comprises administering to a patient or subject having the disease or condition a pharmaceutical composition comprising at least one compound represented by Formula 1 or a pharmaceutically acceptable salt or derivative thereof. In some embodiments, the disease or condition may be a cancer.
BRIEF DESCRIPTION OF THE DRAWINGS
[0019] The following description of embodiments of the present invention will be better understood when considered in conjunction with the following drawing, in which:
[0020] FIG. 1 is a schematic depicting the mitogen-activated protein kinase (MAPK) signaling pathway, reproduced from Nature Reviews, as indicated in the drawing.
DETAILED DESCRIPTION
[0021] RAS is the last key protein involved in the signaling cascade in the MAPK pathway. As such, a drug against RAS would create a new effective first-line treatment for certain cancers, e.g., lung, colon, and pancreatic cancer, and would provide new hope for patients with BRAF and MEK resistant tumors. Currently, 30% of all cancers display mutations in the MAPK pathway, including mutations in KRAS. Although drugs have been developed for other proteins (such as BRAF and MEK, as discussed above) in this pathway, there currently is no drug on the market to treat KRAS, which is the key driver of this pathway.
[0022] According to some embodiments, compounds capable of inhibiting RAS protein forms include compounds represented by Formula 1 , and pharmaceutically acceptable salts thereof.
In Formula 1 , R3-R4 may each independently be a substituted or unsubstituted hydrocarbon substituent, hydrogen, deuterium, F, hydroxyl group, cyano group, amino group, or oxo group (=0). In some embodiments, for example R3-R4 may each independently be hydrogen, F, or a substituted or unsubstituted alkyl group. For example, in some embodiments, each of R3-R4 may each independently be hydrogen, F, or a substituted or unsubstituted lower alkyl (i.e., a substituted or unsubstituted alkyl group having from 1 to 8 carbon atoms). In some embodiments, for example, each of R3-R4 may each independently be hydrogen, F, or a F- substituted lower alkyl group. For example, in some embodiments, each of R3-R4 may each independently be hydrogen, F, methyl, ethyl, isopropyl, F-substituted methyl (e.g., -CH2F, -CHF2, or CF3), or F-substituted ethyl (e.g., -CH2CH2F, - CH2CHF2, -CH2CF3, -CHFCH3, -CF2CH3, -CHFCH2F, -CHFCHF2, -CHFCF3, - CF2CH2F, -CF2CHF2, -CF2CF3).
[0023] Each of x and m is an integer from 1 to 6, and when either x or m (or both) are greater than 1 , each of the plurality of R3s or R4s may be the same or different from each other. Additionally, as shown in Formula 1 , any one or more of the R3s may be attached to any of the ring atoms, including any ring carbon atoms or ring heteroatoms (i.e., when one or more of Xi to X3 is a heteroatom). Also, in some embodiments, two geminal R3s or two geminal R4s (i.e., two R3s or two R4s bonded to a common carbon atom in the spiro ring of Formula 1 ) may combine to form an additional spirocycle from the common carbon atom, as shown generally in the example Formulae 1 A through 1 H below. This additional ring formed from the two geminal R groups (and forming the additional spirocycle) is not particularly limited, and may include any suitable number of carbon atoms. In some embodiments, however, the additional ring (including the common carbon atom) may have from 3 to 10 carbon atoms, for example from 3 to 6 carbon atoms. Also, the additional ring may include ring heteroatoms, as generally described herein, and may be unsubstituted or substituted with any of the substituents described above in
connection with R3 and R4. In some embodiments, for example, the additional ring may be substituted with one or more F atoms, or substituted or unsubstituted alkyl groups (e.g., unsubstituted or substituted lower alkyl groups). Indeed, while the example Formulae 1 A through 1 H below depict examples of 3-6 membered rings with no heteroatoms and no substitutions, it is understood that the present disclosure is not limited thereto, as described above.
[0024] As used herein, the term “
is used in its art-recognized sense to refer to substituents or moieties having any combination or arrangement of carbon atoms, hydrogen atoms, and heteroatoms. Indeed, the hydrocarbon substituent(s) of R3 and R4 are not particularly limited, and may be any suitable hydrocarbon substituent known in this field. In some embodiments, for example, the hydrocarbon substituent may include a substituted or unsubstituted alkyl group, substituted or unsubstituted cycloalkyl group, substituted or unsubstituted alkoxy group, substituted or unsubstituted alkyl carbonyl (or
alkanoyl) group (-C(=O)-alkyl), substituted or unsubstituted alkyl carbonyl amino (or alkanoyl amino) group (-NH-C(=O)-alkyl) substituted or unsubstituted acyl group, substituted or unsubstituted carbamoyl group (-O(C=O)NH2), substituted or unsubstituted carbamoylalkyl (-alkyl-O(C=O)NH2), substituted or unsubstituted carboxyalkyl (-C(=O)-alkyl), substituted or unsubstituted cyanoalkyl (-alkyl — C=N), substituted or unsubstituted alkylsulfanyl (-S(=O)-alkyl), substituted or unsubstituted alkylsulfonyl (-S(=O)2-alkyl), substituted or unsubstituted alkylsulfonylamino (-NH- S(=O)2-alkyl), substituted or unsubstituted alkylthio (-S-alkyl), substituted or unsubstituted alkoxy carbonyl group (-C(=O)-O-alkyl), substituted or unsubstituted haloalkyl group, substituted or unsubstituted haloalkyloxy group, substituted or unsubstituted heteroalkyl, substituted or unsubstituted hydroxyalkyl, substituted or unsubstituted dihydroxyalkyl, substituted or unsubstituted alkylamino group, substituted or unsubstituted alkylaminoalkyl group, substituted or unsubstituted dialkylaminoalkyl group, substituted or unsubstituted alkenyl group, substituted or unsubstituted cycloalkenyl, substituted or unsubstituted alkynyl group, substituted or unsubstituted heterocyclic group, a substituted or unsubstituted heterocycloalkyl group, a substituted or unsubstituted heterocycloamino group, substituted or unsubstituted heterocylcoalkyl group, substituted or unsubstituted aryl group, substituted or unsubstituted halo-aryl group, substituted or unsubstituted aralkyl (or arylalkyl) group, substituted or unsubstituted halo-aralkyl (or arylalkyl) group, substituted or unsubstituted heteroaryl group, substituted or unsubstituted halo- heteroaryl group, substituted or unsubstituted heteroaryl alkyl group, or substituted or unsubstituted halo-heteroaryl alkyl group.
[0025] As used herein, the term “alkyl” refers to straight and branched chain aliphatic groups. The length of the alkyl group is not particularly limited, but in some embodiments, the alkyl group may have from 1 to 12 carbon atoms. In some embodiments, the alkyl group may be a lower alkyl group, i.e. , a straight or branched chain aliphatic group having from 1 to 8 carbon atoms, for example, 1 to 6 carbon atoms, or 1 to 3 carbon atoms. When the alkyl group is substituted, it may be substituted with one or more substituents selected from substituted or unsubstituted hydrocarbon substituents, hydrogen atoms, F atoms, hydroxyl groups, cyano groups, amino groups, or oxo groups. Some nonlimiting examples of suitable alkyl groups include methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, te/Y-butyl, pentyl, and hexyl, which may be unsubstituted or substituted as discussed above. Some additional specific, and non-limiting, examples of suitable alkyl groups include methyl (Me, -CH3), ethyl (Et, -CH2CH3), 1 -propyl (n-Pr, n-propyl, -CH2CH2CH3), 2-propyl (i- Pr, i-propyl, -CH(CH3)2), i-butyl (n-Bu, n-butyl, -CH2CH2CH2CH3), 2-methyl-1 -propyl (i-Bu, i-butyl, -CH2CH(CH3)2), 2-butyl (s-Bu, s-butyl, -CH(CH3)CH2CH3), 2-methyl-2-
propyl (t-Bu, t-butyl, -C(CH3)3), 1 -pentyl (n-pentyl, -CH2CH2CH2CH2CH3), 2-pentyl (- CH(CH3)CH2CH2CH3), 3-pentyl (-CH(CH2CH3)2), 2-methyl-2-butyl (C(CH3)2CH2CH3), 3-methyl-2-butyl (-CH(CH3)CH(CH3)2), 3-methyl-1 -butyl (CH2CH2CH(CH3)2), 2- methyl-1 -butyl (-CH2CH(CH3)CH2CH3), 1 -hexyl (CH2CH2CH2CH2CH2CH3), 2-hexyl (- CH(CH3)CH2CH2CH2CH3), 3-hexyl (CH(CH2CH3)(CH2CH2CH3)), 2-methyl-2-pentyl (- C(CH3)2CH2CH2CH3), 3-methyl-2-pentyl (CH(CH3)CH(CH3)CH2CH3), 4-methyl-2- pentyl (-CH(CH3)CH2CH(CH3)2), 3-methyl-3-pentyl(-C(CH3)(CH2CH3)2), 2-methyl-3- pentyl (-CH(CH2CH3)CH(CH3)2), 2,3-dimethyl-2-butyl (C(CH3)2CH(CH3)2), 3,3- dimethyl-2-butyl (-CH(CH3)C(CH3)3, 1 -heptyl and 1 -octyl.
[0026] Also, as used herein, the term “haloalkyl” refers to an alkyl chain in which one or more hydrogen atoms have been replaced by a F atom. Some nonlimiting examples of suitable haloalkyl groups include trifluoromethyl, difluoromethyl, and fluoromethyl.
[0027] The term “haloalkyloxy” refers to a haloalkyl group bonded to an oxygen atom. In the haloalkyloxy group, the oxygen atom is bonded to the relevant position of Formula 1 . By way of illustration, for example, the haloalkyloxy group has the formula *-O-haloalkyl, where * represents the binding site to Formula 1 .
[0028] Similarly, the term “alkoxy” refers to an alkyl group bonded to an oxygen atom. In the alkoxy group, the oxygen atom is bonded to the relevant position of Formula 1 . By way of illustration, for example, the alkoxy group has the formula *-O- alkyl, where * represents the binding site to Formula 1 .
[0029] The term “hydroxy a Iky I,” however, refers to an alkyl group bonded to a hydroxy group (-OH). In the hydroxyalkyl group, a carbon atom of the alkyl group is bonded to the relevant position of Formula 1 . By way of illustration (and contrast with the alkoxy and haloalkyloxy groups), the hydroxyalkyl group has the formula *- alkyl-OH, where * represents the binding site to Formula 1 . Similarly, the term “dihydroxyalkyl” refers to an alkyl group in which two carbon atoms of the alkyl chain are each substituted with a hydroxyl group.
[0030] The term “alkylamino” refers to a secondary amino group in which the N atom of the amino group is bonded to Formula 1 , one hydrogen atom, and one alkyl group. By way of illustration, the alkylamino group has the formula *-NH-alkyl, where * represents the binding site to Formula 1 .
[0031] The term “dialkylamino,” however, refers to a tertiary amino group in which the N atom of the amino group is bonded to Formula 1 and two alkyl groups (which may be the same or different from each other). By way of illustration, the dialkylamino group has the formula *-N(Raiki)(Raik2), where * represents the binding site to Formula 1 , and Raiki and Raik2 represent alkyl groups which may be the same or different from each other.
[0032] The term “alkylaminoalkyl” refers to an alkyl group that has been substituted with a secondary amino group. In the alkylaminoalkyl group, a carbon atom of one of the alkyl groups is bonded to the relevant position of Formula 1 . By way of illustration, the alkylaminoalkyl group has the formula *-alkyl-NH-alkyl, where * represents the binding site to Formula 1.
[0033] The term “dialkylaminoalkyl” refers to an alkyl group that has been substituted with a tertiary amino group. In the dialkylaminoalkyl group, a carbon atom of one of the three alkyl groups (which may be the same or different from each other) is bonded to the relevant position of Formula 1 . By way of illustration, the dialkylaminoalkyl group has the formula *-alkyl-N(Raiki)(Raik2), where * represents the binding site to Formula 1 , and Raiki and Raik2 represent alkyl groups which may be the same or different from each other.
[0034] The term “cycloalkyl” refers to saturated and partially unsaturated cyclic hydrocarbon groups. The partially unsaturated cyclic groups are also referred to herein as “cycloalkenyls” or “cycloalkenyl groups” and are encompassed both in this definition of cycloalkyl as well as the definition herein of cycloalkenyl. The cycloalkyl group may have from 3 to 12 carbons, for example 3 to 8 carbons, or 3 to 6 carbons. When the cycloalkyl group is substituted, it may be substituted with one or more substituents selected from substituted or unsubstituted hydrocarbon substituents, hydrogen atoms, F atoms, hydroxyl groups, cyano groups, amino groups, or oxo groups. Some nonlimiting examples of suitable cycloalkyl groups include cyclopropyl, cyclobutyl, cyclopentyl, cyclopentenyl, cyclohexyl, cyclohexenyl, cycloheptyl, and cyclooctyl. The term “cycloalkyl” also encompasses bridged cyclic groups, such as, for example, bicyclo[1 .1 ,1]pentanyl. Additional non-limiting examples of monocyclic cycloalkyls include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl, cyclodecyl, cycloundecyl and cyclododecyl. Additional non-limiting examples of bicyclic cycloalkyls include [4,4], [4,5], [5,5], [5,6] or [6,6] ring systems. Additional non-limiting examples of suitable bridged bicyclic cycloalkyls include bicyclo[2.2.1]heptane, bicyclo[2.2.2]octane and bicyclo[3.2.2]nonane. And additional non-limiting examples of suitable spirocycloalkyls include spiro[2.2]pentane, spiro[2.3]hexane, spiro[2.4]heptane, spiro[2.5]octane and spiro[4.5]decane.
[0035] The term “heteroalkyl” refers to an alkyl group, as defined herein, in which one or more carbon atoms in the chain are replaced by a heteroatom. The heteroatom is not particularly limited, and may be any atom other than carbon or nitrogen. In some embodiments, for example, the heteroatom may be selected from O, S, and N.
[0036] As used herein, the “aryl” group is an aromatic moiety having one or more aromatic rings, for example, from 1 to 3 aromatic rings. Each of the rings of the aryl group may optionally be substituted, as noted generally above. In some embodiments, the aryl group may have from 6 to 14 ring carbon atoms, for example, from 6 to 10 ring carbon atoms. Some nonlimiting examples of suitable aryl groups include phenyl, naphthyl, anthracenyl, fluorenyl, dihydrobenzofuranyl, biphenyl, phenanthrenyl, naphthacenyl, 1 ,2,3,4-tetrahydronaphthalenyl, IH-indenyl, 2,3- dihydro-IH-indenyl, and the like.
[0037] The term “halo-aryl” refers to an aryl group substituted with at least one halogen atom. The halogen atom substituent on the “halo-aryl group” is not particularly limited, and in some embodiments, may be any of F, Cl, I or Br. The halo-aryl group may be further substituted with any of the other substituents described herein.
[0038] The terms “aralkyl” and “arylalkyl” refer to an aryl group covalently linked to an alkyl group. Either the alkyl group or the aryl group, or both, may be substituted as noted generally above. In contrast to an aryl group substituted with an alkyl group (in which the aryl group is bonded to the relevant position of Formula 1 ), in the aralkyl group, the alkyl component is bonded to the relevant position of Formula 1 . By way of illustration, the aralkyl group has the formula *-alkyl-aryl, where * represents the binding site to Formula 1 . In some embodiments, the alkyl component of the aralkyl group may include from 1 to 6 carbon atoms, and the aryl component may include from 6 to 12 (e.g., from 6 to 10) carbon atoms. Some nonlimiting examples of suitable aralkyl groups include benzyl, phenethyl, and naphthylmethyl, and a nonlimiting example of a suitable substituted aralkyl includes an aralkyl group in which the alkyl component is substituted with hydroxy or hydroxyalkyl.
[0039] The term “halo-arylalkyl” or “halo-aralkyl” refers to an aryl alkyl group (or aralkyl group), as defined herein, substituted with at least one halogen atom. Either the alkyl component or the aryl component may be substituted with the halogen. However, when the alkyl component is substituted, the halogen atom is F. When the aryl component is substituted with the halogen, the halogen atom is not particularly limited, and may be any of F, Cl, I or Br. The halo-arylalkyl (or halo-aralkyl) group may be further substituted with any of the other substituents described herein.
[0040] The term "alkenyl" refers to linear or branched hydrocarbon groups with at least one carbon-carbon double bond, and includes groups having “c/s” and “trans" orientations, or alternatively, “E” and “Z” orientations. The size of the alkenyl group is not particularly limited, but in some embodiments, the alkenyl group may have 2 to 18 carbon atoms, for example, 2 to 12 carbon atoms, 2 to 10 carbon atoms, 2 to 8
carbon atoms, 2 to 6 carbon atoms, or 2 to 3 carbon atoms. Some non-limiting examples of suitable alkenyl groups include ethenyl or vinyl (-CH=CH2), prop-1 -enyl (-CH=CHCH3), prop-2 -enyl (-CH2CH=CH2), 2-methylprop-1-enyl, but-1-enyl, but-2- enyl, but-3-enyl, buta-1 ,3-dienyl, 2-methylbuta-1 ,3-dieneyl, hex-1 -enyl, hex-2-enyl, hex-3-enyl, hex-4-enyl, and hexa-1 ,3-dienyl.
[0041] The term “cycloalkenyl” refers to a cyclic structure having at least one ring that is non-aromatic, and that has at least one carbon-carbon double bond. The ring structure of the cycloalkenyl group is not particularly limited, and may have any number of rings in any configuration. In some embodiments, for example, the cycloalkenyl group may include from 1 to 3 rings, which may be arranged relative to each other in any way. For example, in some embodiments, the rings of the group may be either bonded to each other or fused to each other, and in some embodiments, some rings of the group may be bonded while other rings may be fused. Additionally, in some embodiments, the cycloalkenyl group may be spirocyclic or may have a bridged ring structure. Some non-limiting examples of suitable monocyclic cycloalkenyl groups include l-cyclopent-1-enyl, l-cyclopent-2- enyl, l-cyclopent-3-enyl, l-cyclohex-1-enyl, l-cyclohex-2-enyl, l-cyclohex-3-enyl, and cyclohexadienyl. Some non-limiting examples of suitable bicyclic cycloalkenyls include [4,4], [4,5], [5,5], [5,6] or [6,6] ring systems. Some non-limiting examples of suitable bridged bi-cyclic cycloalkenyl groups include bicyclo[2.2.1]heptene, bicyclo[2.2.2]octene and bicyclo[3.2.2]nonene. Some non-limiting examples of suitable spiro cycloalkenyl groups include, spiro[2.2]pentene, spiro[2.3]hexene, spiro[2.4]heptene, spiro[2.5]octene and spiro[4.5]decene.
[0042] The term "alkynyl" refers to a linear or branched hydrocarbon group with at least one carbon-carbon triple bond. The size of the alkynyl group is not particularly limited. However, in some embodiments, the alkynyl group may have 2 to 18 carbon atoms, for example 2 to 13 carbon atoms, 2 to 12 carbon atoms, 2 to 10 carbon atoms, 2 to 6 carbon atoms, or 2 to 3 carbon atoms. Some non-limiting examples of suitable alkynyl groups include ethynyl (-C=CH), prop-1 -ynyl (-C=CCH3), prop-2 -ynyl (propargyl, -CH2C=CH), but-1-ynyl, but-2-ynyl, and but-3-ynyl.
[0043] The term "acyl" refers to a carbonyl containing substituent represented by *-C(=O)-Racyi in which Racyi is a hydrocarbon substituent (e.g., hydrogen, alkyl, cycloalkyl, aryl or heterocyclic group), and * represents the binding site to the relevant position of Formula 1 . Some non-limiting examples of suitable acyl groups include alkanoyl (e.g., acetyl), aroyl (e.g., benzoyl), and heteroaroyl (e.g., pyridinoyl). [0044] As used herein, a “heterocyclic” group is a cyclic structure having one or more rings in which at least one ring atom of at least one of the rings is a heteroatom. In some embodiments, for example, more than one atom of at least one
ring, or more than one atom of more than one ring, may be a heteroatom. The heteroatom is not particularly limited, and may be any atom other than carbon or nitrogen. In some embodiments, for example, the heteroatom may be selected from O, S, and N. Any of the ring atoms (including the heteroatoms) may also be substituted, as generally noted above.
[0045] The ring structure of the heterocyclic group is not particularly limited, and may have any number of rings in any configuration. In some embodiments, for example, the heterocyclic group may include from 1 to 3 rings, which may be arranged relative to each other in any way. For example, in some embodiments, the rings of the heterocyclic group may be either bonded to each other or fused to each other, and in some embodiments, some rings of the heterocycle may be bonded while other rings may be fused. Additionally, in some embodiments, the heterocyclic group may be spirocyclic or may have a bridged ring structure.
[0046] The size of the heterocyclic group is also not particularly limited. However, in some embodiments, the heterocyclic group may have from 3 to 12 ring atoms, for example from 4 to 8 ring atoms.
[0047] Some nonlimiting examples of suitable heterocyclic groups include epoxy, azetidinyl, aziridinyl, tetrahydrofuranyl, tetrahydropyranyl, pyrrolidinyl, pyrrolidinonyl, piperidinyl, piperazinyl, imidazolidinyl, thiazolidinyl, dithianyl, trithianyl, dioxolanyl, oxazolidinyl, oxazolidinonyl, decahydroquinolinyl, piperidonyl, 4-piperidinonyl, quinuclidinyl, thiomorpholinyl, thiomorpholinyl[1 , 1 ]dioxide, morpholinyl, azepanyl, oxazepanyl, azabicyclohexanes, azabicycloheptanes, azabicyclooctanes, azabicyclononanes (e.g., octahydroindolizinyl), azaspiroheptanes, dihydro- 1 H,3H,5H-oxazolo[3,4-c]oxazolyl, tetrahydro-1'H,3'H-spiro[cyclopropane-1 ,2'- pyrrolizine), hexahydro-1 H-pyrrolo[2,1-c][1 ,4]oxazine, octahydroindolizinyl, oxaazaspirononanes, diazaspirononanes, and oxaazabiocycloheptanes. In some embodiments, for example, the heterocyclic group may include oxiranyl, aziridinyl, thiiranyl, azetidinyl, oxetanyl, thietanyl, 1 ,2-dithietanyl, 1 ,3-dithietanyl, pyrrolidinyl, dihydro- IH-pyrrolyl, dihydrofuranyl, tetrahydrofuranyl, dihydrothienyl, tetrahydrothienyl, imidazolidinyl, piperidinyl, piperazinyl, isoquinolinyl, tetrahydroisoquinolinyl, morpholinyl, thiomorpholinyl, 1 ,1-dioxothiomorpholinyl, dihydropyranyl, tetrahydropyranyl, hexahydrothiopyranyl, hexahydropyrimidinyl, oxazinanyl, thiazinanyl, thioxanyl, homopiperazinyl, homopiperidinyl, azepanyl, oxepanyl, thiepanyl, oxazepinyl, oxazepanyl, diazepanyl, 1 ,4-diazepanyl, diazepinyl, thiazepinyl, thiazepanyl, tetrahydrothiopyranyl, oxazolidinyl, thiazolidinyl, isothiazolidinyl, 1 ,1-dioxoisothiazolidinonyl, 1 ,1 -dioxoisothiazolyl, oxazolidinonyl, imidazolidinonyl, 4,5,6,7-tetrahydro[2H]indazolyl, tetrahydrobenzoimidazolyl, 4, 5,6,7- tetrahydrobenzo[d]imidazolyl, thiazinyl, oxazinyl, thiadiazinyl, oxadiazinyl, dithiazinyl,
dioxazinyl, oxathiazinyl, thiatriazinyl, oxatriazinyl, dithiadiazinyl, imidazolinyl, dihydropyrimidyl, tetrahydropyrimidyl, 1-pyrrolinyl, 2-pyrrolinyl, 3-pyrrolinyl, indolinyl, thiapyranyl, 2H-pyranyl, 4H-pyranyl, dioxanyl, 1 ,3-dioxolanyl, pyrazolinyl, pyrazolidinyl, dithianyl, dithiolanyl, pyrimidinonyl, pyrimidindionyl, pyrimidin-2,4- dionyl, piperazinonyl, piperazindionyl, pyrazolidinylimidazolinyl, 3- azabicyclo[3.1 .0]hexanyl, 3,6-diazabicyclo[3.1.1]heptanyl, 6- azabicyclo[3.1 .1 ]heptanyl, 3-azabicyclo[3.1.1 ]heptanyl, 3-azabicyclo[4.1 ,0]heptanyl, azabicyclo[2.2.2]hexanyl, 2-azabicyclo[3.2.1]octanyl, 8-azabicyclo[3.2.1]octanyl, 2- azabicyclo[2.2.2]octanyl, 8-azabicyclo[2.2.2]octanyl, 7-oxabicyclo[2.2.1 ]heptane, azaspiro[3.5]nonanyl, azaspiro[2.5]octanyl, azaspiro[4.5]decanyl, 1- azaspiro[4.5]decan-2-onyl, azaspiro[5.5]undecanyl, tetrahydroindolyl, octahydroindolyl, tetrahydroisoindolyl, tetrahydroindazolyl, 1 ,1- dioxohexahydrothiopyranyl.
[0048] The term “heterocycloalkyl” refers to a heterocyclic group, as defined herein, linked to an alkyl group. Either the alkyl group or the heterocyclic group, or both, may be substituted as noted generally above. In contrast to a heterocyclic group substituted with an alkyl group (in which the heterocyclic group is bonded to the relevant position of Formula 1), in the heterocycloalkyl group, the alkyl component is bonded to the relevant position of Formula 1 . By way of illustration, the heterocycloalkyl group has the formula *-alkyl-heterocycle, where * represents the binding site to Formula 1 .
[0049] The term “heterocycloamino” refers to a heterocyclic group, as defined herein, linked to an amino group. Either the amino group or the heterocyclic group, or both, may be substituted as noted generally above. In contrast to a heterocyclic group substituted with an amino group (in which the heterocyclic group is bonded to the relevant position of Formula 1), in the heterocycloamino group, the amino component is bonded to the relevant position of Formula 1 . By way of illustration, the heterocycloamino group has the formula *-NH-heterocycle, where * represents the binding site to Formula 1 .
[0050] As used herein, the term “heteroaryl” refers to an aromatic moiety having one or more aromatic rings, in which at least one atom of at least one ring is a heteroatom. In some embodiments, for example, more than one atom of at least one ring, or more than one atom of more than one ring, may be a heteroatom. The heteroatom is not particularly limited, and may be any atom other than carbon or nitrogen. In some embodiments, for example, the heteroatom may be selected from O, S, and N. Any of the ring atoms (including the heteroatoms) may also be substituted, as generally noted above. When substituted, the heteroaryl group may
be substituted with any halogen atom (e.g., F, I, Cl or Br) or any of the other substituents described herein.
[0051] The size and configuration of the heteroaryl group is also not particularly limited, and the heteroaryl group may have any number of rings in any configuration. In some embodiments, for example, the heteroaryl group may include from 1 to 3 rings, which may be arranged relative to each other in any way. For example, in some embodiments, the rings of the heteroaryl group may be either bonded to each other or fused to each other, and in some embodiments, some rings of the group may be bonded while other rings may be fused. In some embodiments, for example, the aryl group may have from 5 to 14 ring atoms, for example 5, 6, 9, or 10 ring atoms, and/or 6, 10, or 14 electrons shared in a cyclic array.
[0052] Some nonlimiting examples of suitable heteroaryl groups include acridinyl, azocinyl, benzimidazolyl, benzofuranyl, benzothiofuranyl, benzothiophenyl, benzoxazolyl, benzthiazolyl, benztriazolyl, benztetrazolyl, benzisoxazolyl, benzisothiazolyl, benzimidazolinyl, carbazolyl, 4aH-carbazolyl, carbolinyl, chromanyl, chromenyl, cinnolinyl, 6,7-dihydro-5H-pyrrolo[1 ,2-a]imidazole, furanyl, furazanyl, imidazolinyl, imidazolyl, 1 H-indazolyl, indolenyl, indolinyl, indolizinyl, indolyl, 3H- indolyl, isobenzofuranyl, isochromanyl, isoindazolyl, isoindolinyl, isoindolyl, isoquinolinyl, isothiazolyl, isoxazolyl, methylenedioxyphenyl, naphthyridinyl, octahydroisoquinolinyl, oxadiazolyl, 1 ,2,3-oxadiazolyl, 1 ,2,4-oxadiazolyl, 1 ,2,5- oxadiazolyl, 1 ,3,4-oxadiazolyl, oxazolidinyl, oxazolyl, oxazolidinyl, pyrimidinyl, phenanthridinyl, phenanthrolinyl, phenazinyl, phenothiazinyl, phenoxathiinyl, phenoxazinyl, phthalazinyl, piperonyl, pteridinyl, purinyl, pyranyl, pyrazinyl, pyrazolidinyl, pyrazolinyl, pyrazolyl, pyridazinyl, pyridooxazole, pyridoimidazole, pyridothiazole, pyridinyl, pyridyl, pyrimidinyl, pyrrolinyl, 2H-pyrrolyl, pyrrolyl, quinazolinyl, quinolinyl, 4H-quinolizinyl, quinoxalinyl, quinuclidinyl, tetrahydroisoquinolinyl, tetrahydroquinolinyl, tetrazolyl, 6H-1 ,2,5-thiadiazinyl, 1 ,2,3- thiadiazolyl, 1 ,2,4-thiadiazolyl, 1 ,2,5-thiadiazolyl, 1 ,3,4-thiadiazolyl, thianthrenyl, thiazolyl, thienyl, thienothiazolyl, thienooxazolyl, thienoimidazolyl, thiophenyl, triazinyl, 1 ,2,3-triazolyl, 1 ,2,4-triazolyl, 1 ,2,5-triazolyl, 1 ,3,4-triazolyl, and xanthenyl. For example, in some embodiments, the heteroaryl group may include thienyl, furyl, imidazolyl, pyrazolyl, thiazolyl, isothiazolyl, oxazolyl, isoxazolyl, triazolyl, thiadiazolyl, oxadiazolyl, tetrazolyl, thiatriazolyl, oxatriazolyl, pyridyl, pyrimidyl, pyrazinyl, pyridazinyl, triazinyl, tetrazinyl, tetrazolo[1 ,5-b]pyridazinyl, imidazol[1 ,2-a]pyrimidinyl and purinyl, as well as benzo-fused derivatives, such as, for example benzoxazolyl, benzofuryl, benzothiazolyl, benzothiadiazolyl, benzotriazolyl, benzoimidazolyl, indazolyl or indolyl.
[0053] The term “halo-heteroaryl” refers to a heteroaryl group substituted with at least one halogen atom. The halogen atom substituent on the “halo-heteroaryl group” is not particularly limited, and in some embodiments, may be any of F, Cl, I or Br. The halo-heteroaryl group may be further substituted with any of the other substituents described herein.
[0054] The term “heteroaryl alkyl” refers to a heteroaryl group, as defined herein, linked to an alkyl group. Either the alkyl group or the heteroaryl group, or both, may be substituted as noted generally above. In contrast to a heteroaryl group substituted with an alkyl group (in which the heteroaryl group is bonded to the relevant position of Formula 1 ), in the heteroarylalkyl group, the alkyl component is bonded to the relevant position of Formula 1 . By way of illustration, the heteroarylalkyl group has the formula *-alkyl-heteroaryl, where * represents the binding site to Formula 1 .
[0055] The term “halo-heteroaryl alkyl” refers to a heteroaryl alkyl group, as defined herein, substituted with at least one halogen atom. Either the alkyl component or the heteroaryl component may be substituted with the halogen. However, when the alkyl component is substituted, the halogen atom is F. When the heteroaryl component is substituted with the halogen, the halogen atom is not particularly limited, and may be any of F, Cl, I or Br. The halo-heteroaryl alkyl group may be further substituted with any of the other substituents described herein.
[0056] Returning to Formula 1 , each of X1-X3 is independently a bond, a moiety containing a ring C atom, or a moiety containing a ring heteroatom. As noted generally above, the heteroatom is not particularly limited, and may be any suitable non-carbon, non-hydrogen atom. For example, in some embodiments, each of X1-X3 is independently a moiety containing a ring C atom or a ring heteroatom selected from N, O and S. Additionally, as shown in Formula 1 , the ring to which X1-X3 belong is non-aromatic. Accordingly, in some embodiments, while each of X1-X3 may be bonded within the ring either to each other (e.g., X1-X2) or to an adjacent ring carbon atom by a single or double bond, the ring containing X1-X3 remains non-aromatic. [0057] Additionally, as noted above, in some embodiments, one or more of X1-X3 may independently be a bond such that the ring containing X1-X3 may be a 3 to 6 membered non-aromatic ring. In some embodiments, for example, when both Xi and X2 are a bond, they combine to form one bond to the ring A. In such embodiments, when X3 is also a bond, the ring containing X1-X3 is a 3-membered ring; and when X3 is not a bond (e.g., is a ring C atom or ring heteroatom), the ring containing X1-X3 is a 4-membered ring. However, as noted above, any one or more of X1-X3 may be a bond (yielding a 3 to 5 membered ring), or none of X1-X3 may be a bond (yielding a 6 membered ring).
[0058] The X1-X3 moieties containing the ring C atoms or ring heteroatoms are not particularly limited, and may be any suitable such moiety. For example, in some embodiments, X1-X3 may each independently be carbonyl (-C(=O)-), an oxygen atom (-O-), amino (-NH-, or -N(Ram)-, where Ram is a substituent as defined above with respect to R3 and R4 in Formula 1 , and in some embodiments, is a lower alkyl group), or a divalent methyl which may be substituted or unsubstituted (e.g., -CH2-, - CHF-, -CF2-, -CHRme-, -C(Rmei )(Rme2)-, or -CFRme-, where Rme, Rmei, and Rme2 are each independently a substituent as defined above with respect to Ram of the amino, and R3 and R4 of Formula 1 , and in some embodiments are each independently a lower alkyl group). In the divalent methyl having two R groups (i.e. , Rmei, and Rme2), the two R groups may be the same or different from each other. And in some embodiments, two geminal R groups (e.g., Rmei , and Rme2) may combine to form an additional substituted or unsubstituted spirocycle (which can include one or more heteroatoms) from the common carbon atom, as described above in connection with R3 and R4, as well as Formulae 1 A through 1 H. However, in some embodiments, Xi through X3 may be selected such that no more than one of Xi through X3 is a ring oxygen atom. In some embodiments, Xi and X2 may be double-bonded to each other within the ring, and may each therefore be -CH- or -CR (resulting in a structure within the ring represented by -CH=CH-).
[0059] Any number of different configurations are possible in selecting the moieties for X1-X3, and the selection of these moieties is not particularly limited. In some embodiments, however, one of X1-X3 may be selected from a carbonyl (-C(= O)-), an oxygen atom (-O-), or an amino (-NH-, or -N(Ram)-), while the remaining two of X1-X3 are divalent methyl (i.e., -CH2-, -CHF-, -CF2-, -CHRme- -C(Rmei)(Rme2)-, or -CFRme-, and in some embodiments -CH2-). In other embodiments, Xi and X2 may be double-bonded to each other (resulting in a structure within the ring of -CH=CH-, or -C(Rmei)= C(Rme2)-, wherein each of Rmei and Rme2 are independently H, a halogen (e.g., F) or an R group as defined above), while X3 is an oxygen atom or a divalent methyl (e.g., -CH2-, -CHF-, -CF2-, -CHRme-, -C(Rmei)(Rme2)-, or -CFRme-, and in some embodiments -CH2-). In still other embodiments, each of X1-X3 may independently be a divalent methyl (e.g., -CH2-, -CHF-, -CF2-, -CHRme-, -C(Rmei)(Rme2)-, or -CFRme-, and in some embodiments -CH2-). In embodiments in which two or more of X1-X3 may be a divalent methyl, the plurality of divalent methyls may be the same or different from each other.
[0060] In some embodiments, X1-X3 may be selected to provide a lactam ring structure. Specifically, as shown in the below Formulae 2A and 2B, one of Xi and X2 in these embodiments is an amino (-NH-, or -N(Ram)-), and the other of Xi and X2 is a carbonyl (-C(=O)-). Also, in these embodiments, X3 may be an oxygen atom (-O-)
or a divalent methyl (e.g., -CH2-, -CHF-, -CF2-, -CHRme-, -C(Rmei)(Rme2)-, or -CFRme- and in some embodiments -CH2-, -CHF- or -CF2-).
[0061] In Formula 2A and 2B, each of R1-R4, A, x and m are as defined herein with respect to Formula 1 , and R5 is equivalent to Ram described above with respect to the amino moiety. In some embodiments, however, R5 (or Ram) may be hydrogen or a lower alkyl group (unsubstituted or substituted with, e.g., one or more halogen atoms). Additionally, X3 in Formulae 2A and 2B are also as defined herein with respect to Formula 1 , however in Formula 2B, X3 is not a heteroatom (i.e., X3 in Formula 2B is either a bond or a ring C atom (which may be substituted or unsubstituted as discussed herein).
[0062] Returning again to Formula 1 , the compound has a spirocyclic core structure having a pyrimidine ring fused to the spirocyclic core on one side, and the ring A fused to another side of the spirocyclic core. Within this structure, the ring A is not particularly limited, and may be any cyclic (e.g., cycloalkyl or cycloalkenyl as defined herein), heterocyclic, aryl or heteroaryl ring. For example, in some embodiments, the ring A is an aryl or heteroaryl ring. The size of the ring A is also not particularly limited. However, in some embodiments, the ring A may be a 5- or 6- membered ring. And in some embodiments, the ring A may be a 5- or 6-membered substituted or unsubstituted aryl or heteroaryl ring. Some nonlimiting examples of suitable rings for the ring A include substituted and unsubstituted benzene rings, substituted and unsubstituted thiophene rings, substituted and unsubstituted furan rings, substituted and unsubstituted azole rings (e.g., including mono-, di-, tri- and tetra-azoles), substituted and unsubstituted oxazole rings (e.g., including mono-, di-, tri- and tetra-azoles), substituted and unsubstituted thiazoles (e.g., including mono-, di-, tri- and tetra-azoles), substituted and unsubstituted azine rings, substituted and unsubstituted pyran rings, substituted and unsubstituted thiopyran rings, and substituted and unsubstituted cyclopentadiene rings. Some nonlimiting examples of suitable rings for the ring A include benzene, thiophene, furan, pyran, thiopyran, pyrrole, imidazole, pyrazole, triazole, pyridine, pyrimidine, pyridazine, pyrazine,
oxazole, thiazole and cyclopentadiene. Formulae 3A through 3K below show the general structure of the compounds when the ring A is benzene (Formula 3A), thiophene (Formula 3B and 3C), furan (an example configuration in Formula 3D), pyran (an example configuration in Formula 3E), thiopyran (an example configuration in Formula 3F), imidazole (an example configuration in Formula 3G), pyridine (an example configuration in Formula 3H), oxazole (an example configuration in Formula 3I), thiazole (an example configuration in Formula 3J), and cyclopentadiene (an example configuration in Formula 3K).
[0063] While Formulae 3A through 3K depict certain example configurations of the fusion of the ring A to the spirocyclic core of Formula 1 , it is understood that the ring A may be fused to the spirocyclic core in any suitable configuration. Those of ordinary skill in the art would readily recognize the available fusion configurations, all of which are encompassed by this disclosure. By way of example only, and without limitation, some example alternative configurations include: 1 ) Formula 3D (furan) in which the O of the furan ring points downward rather than upward (depicted below as Formula 3L); 2) Formula 3E (pyran) in which the O atom is any other available ring position) (depicted below as Formulae 3M(i) through 3M(iii); 3) Formula 3F (thiopyran) in which the S is in any other available ring position) (depicted below as
Formulae 3N(i) through 3N(iii)); 4) Formula 3G (imidazole) in which the N-RA1 moiety points upward rather than downward (depicted below as Formula 30); 5) Formula 3H (pyridine) in which the N atom is in any other available ring position (depicted below as Formulae 3P(i) through 3P(iii)); 6) Formula 3I (oxazole) in which the 0 atom points upward rather than downward (depicted below as Formula 3Q); 7) Formula 3J (thiazole) in which the S atom points upward rather than downward (depicted below as Formula 3R); 8) Formula 3K (cyclopentadiene) in which the CH2 moiety points downward rather than upward (depicted below as Formula 3S).
[0064] In Formulae 3A through 3K and 3L through 3S, each of R1-R4, x and m are as defined herein with respect to Formula 1 . RA, RA1 and RA2 may each independently be any halogen (e.g., F, I, Cl or Br) or any of the substituents described above in connection with R3-R4 of Formula 1 . For example, in some embodiments, RA, RA1 and RA2 may be hydrogen, a halogen, or a lower alkyl group (unsubstituted or substituted with, e.g., one or more F atoms). Additionally, p is an integer from 1 to 4 and q is an integer from 1 to 2. When either p or q are greater than 1 , each of the plurality of RAs may be the same or different from each other. Additionally, as shown in Formulae 3A through 3K and 3L through 3S, any one or more of the RAs, RA1s and RA2s may be attached to any of the ring atoms, including any ring carbon atoms or ring heteroatoms (i.e. , when the ring A is heterocyclic or heteroaryl).
[0065] As generally noted above, the ring A may be substituted with one or more substituents RA, RA1 and RA2. The number and type of substituents RA, RA1 and RA2 is not particularly limited, and may be any suitable number and type. In some embodiments, for example, the ring A may be substituted with one or more halogen atoms (e.g., F, Cl, I or Br) or lower alkyl groups (which may themselves be unsubstituted or substituted with, e.g., one or more F atoms).
[0066] Turning back to Formula 1 again, the pyrimidine ring fused to the spirocyclic core may also be substituted with substituents R1 and R2. R1 and R2 are not particularly limited, and may be any halogen (e.g., F, I, Cl or Br) or any of the substituted or unsubstituted substituents described above with respect to R3 and R4. [0067] In some embodiments, however, R1 may be represented by one of Formulae 4A and 4B.
[0068] In Formula 4A, * represents the binding site to Formula 1 , and X4 may be selected from the same substituents described above with respect to R3 and R4 of Formula 1 . In some embodiments, however, X4 may be selected from 4 to 12 membered heterocyclic rings (or groups), for example, 4 to 7 membered heterocyclic rings (or groups). The heterocyclic group may be unsubstituted, or may optionally be substituted with one or more substituents, such as those described above in connection with R3 and R4. For example, in some embodiments, each substituent may be independently selected from alkyl groups, cyano groups, cyanoalkyl groups, and hydroxyalkyl groups. Additionally, two geminal substituents of the heterocyclic group may optionally combine together to form a 4 to 12 membered (or, in some embodiments, a 4 to 7 membered) spiroheterocyclic group.
[0069] In some embodiments, for example, X4 may be a 4 to 7 membered heterocylcoamino group, which may be substituted or unsubstituted. For example, in some embodiments, X4 may be , wherein * represents binding sites to
Formula 1 and to R14 in Formula 4A. Also, the ring in the
moiety may be unsubstituted or substituted with the substituents described above in connection with
f Formula 1 . For example, in some embodiments, the ring in the
moiety may be substituted with one or more of -CH3, -CH2CH3, -CH2CN, -CH2OH, -CN, -CF3, -CH2F, -CHF2, -CH2CF3, -CH2CHF2, -CH2CH2F, -CHFCF3, -CF2CF3, -CHFCHF2, -CF2CHF2, -CHFCH2F, or -CF2CH2F.
[0070] In some embodiments, as shown generally in Formula 4B, X4 may be a 4 to 12 membered saturated or partially saturated monocyclic, bridged or spirocyclic ring having 2 nitrogen ring atoms, which ring may optionally be substituted with one or more substituents (such as those described above in connection with R3 and R4 of Formula 1 ). In some embodiments, for example, X4 may be selected from the following structures:
In each of the above structures, the < represents binding sites to Formula 1 and R14 of Formula 4A or 4B, and either of the in each of the above structures may be bonded to either Formula 1 or R14 of Formula 4A or 4B. In some embodiments, however, the lower represents the binding site to Formula 1 , and the upper represents the binding site to R14.
[0071] For example, in some embodiments, X4 may be selected from the following structures:
In each of the above structures, the represents binding sites to Formula 1 and R14 of Formula 4A or 4B. And again, in each of the above structures, the represents binding sites to Formula 1 and R14 of Formula 4A or 4B, and either of the in each of the above structures may be bonded to either Formula 1 or R14 of Formula 4A or 4B. In some embodiments, however, the lower represents the binding site to Formula 1 , and the upper represents the binding site to R14.
[0072] In some embodiments, for example, X4 may be a substituted or unsubstituted piperazine or homopiperazine ring. When substituted, the piperazine or homopiperazine ring may be substituted with any of the substituents discussed above in connection with R3 and R4 of Formula 1 . In some embodiments, however, the piperazine or homopiperazine ring may be substituted with one or more of -CH3, -CH2CH3, -CH2CN, -CH2OH, -CN, -CF3, -CH2F, -CHF2, -CH2CF3, -CH2CHF2, -CH2CH2F, -CHFCF3, -CF2CF3, -CHFCHF2, -CF2CHF2, -CHFCH2F, or -CF2CH2F. [0073] R14 in Formula 4A and 4B is also not particularly limited, and may be any of the substituents described above in connection with R3 and R4 of Formula 1 . In some embodiments, for example, R14 may be represented by one of Formulae 5A through 5E.
Formula 5A Formula 5B
In Formulae 5A through 5E, n is 0, 1 or 2, and in some embodiments, n is 0. R15 through R19 are not particularly limited, and may be independently selected from the substituents described above in connection with R3 and R4 of Formula 1 . In some embodiments, for example, R15 through R19 may each independently be hydrogen, a halogen, a substituted or unsubstituted lower alkyl group (e.g., -CH3, -CH2CH3, -CF3, -CH2F, -CHF2, -CH2CF3, -CH2CHF2, -CH2CH2F, -CHFCF3, -CF2CF3, -CHFCHF2, -CF2CHF2, -CHFCH2F, or -CF2CH2F), a substituted or unsubstituted alkylamino group (e.g., in which the alkyl component is a lower alkyl), a substituted or unsubstituted alkylaminoalkyl group (e.g., in which the alkyl component is a lower alkyl), a substituted or unsubstituted cyano group, or a substituted or unsubstituted cyanoalkyl group. In some embodiments, the substituted lower alkyl, substituted alkylamino, and substituted alkylaminoalkyl group may be substituted with one or more halogen atoms (e.g., one or more F atoms).
[0074] For example, in some embodiments, in Formulae 5B and 5C, R15 and R16 may be H or F, and R17 may be hydrogen, a halogen, a substituted or unsubstituted lower alkyl group (e.g., -CH3, -CH2CH3, -CF3, -CH2F, -CHF2, -CH2CF3, -CH2CHF2,
-CH2CH2F, -CHFCF3, -CF2CF3, -CHFCHF2, -CF2CHF2, -CHFCH2F, or -CF2CH2F), a substituted or unsubstituted alkylamino group (e.g., in which the alkyl component is a lower alkyl), and a substituted or unsubstituted alkylaminoalkyl group (e.g., in which the alkyl component is a lower alkyl).
In the above structures, the represents the binding site to X4 of Formula 4A or 4B. And in the above structures, R16 through R18 may be independently selected from the substituents described above in connection with R3 and R4 of Formula 1 . For example, in some embodiments, R16 may be selected from substituted and unsubstituted acyl groups (e.g., in which the alkyl component is a lower alkyl), substituted or unsubstituted lower alkyl groups, substituted or unsubstituted alkylsulfonyl groups (e.g., in which the alkyl component is a lower alkyl), and substituted or unsubstituted cycloalkyl (e.g., having from 3 to 6 carbon atoms). Also, in some embodiments, R17 may be selected from substituted or unsubstituted lower alkyl groups, and substituted or unsubstituted haloalkyl groups (e.g., in which the alkyl component is a lower alkyl). And in some embodiments, R18 may be hydrogen, or a halogen atom.
[0077] In the above structures, the * represents the binding site to X4 of Formula 4A or 4B.
And in some embodiments, for example, R may be represented by |n the above structures, the represents the binding site to X4 of Formula 4A or 4B. [0079] In some embodiments, R1 (including both X4 and R14) may be represented by one of Formulae 6A-6D.
In Formula 6A through 6D, z is 0, 1 , 2, 3 or 4, and in some embodiments, z is 1 , 2 or 3, for example, 1 or 2. R15 through R17 and R19 are as described above in connection with Formula 5A through 5E. And in Formulae 6A through 6D, the * represents the binding site to Formula 1 .
[0080] R11 is not particularly limited, and may be selected from the substituents described above in connection with R3 and R4 of Formula 1 . In some embodiments, for example, R11 may be hydrogen, a halogen, a substituted or unsubstituted cyano, a substituted or unsubstituted alkyl cyano, a substituted or unsubstituted lower alkyl group (e.g., -CH3, -CH2CH3, -CF3, -CH2F, -CHF2, -CH2CF3, -CH2CHF2, -CH2CH2F, -CHFCF3, -CF2CF3, -CHFCHF2, -CF2CHF2, -CHFCH2F, or -CF2CH2F), a substituted or unsubstituted alkynyl group (e.g., an alkynyl group having from 2 to 4, for example 2 to 3 carbon atoms), a substituted or unsubstituted alkylamino group (e.g., in which the alkyl component is a lower alkyl), and a substituted or unsubstituted alkylaminoalkyl group (e.g., in which the alkyl component is a lower alkyl). In some
embodiments, the substituted lower alkyl, substituted alkylamino, and substituted alkylaminoalkyl group may be substituted with one or more halogen atoms (e.g., one or more F atoms). In some embodiments, for example, z is 1 , 2, 3 or 4, and at least one of the R11s is a substituted or unsubstituted cyano group, or a substituted or unsubstituted alkylcyano group.
In Formula 7A through 7D, R15 through R17 and R19 are as described above in connection with Formulae 5A through 5E and Formulae 6A through 6D, and the * represents the binding site to Formula 1 . Also, R11A, R11B and R11 C are equivalent to R11 described above in connection with Formulae 6A through 6D. However, in some embodiments, R11A and R11C are independently selected from hydrogen, a halogen atom (e.g., F), a substituted or unsubstituted alkyl group (e.g., a lower alkyl group that is optionally substituted with one or more halogen atoms), a substituted or unsubstituted alkynyl group (e.g., substituted with one or more halogen atoms, e.g., F), a substituted or unsubstituted cyano group, or a substituted or unsubstituted alkyl
cyano group (e.g., in which the alkyl component is a lower alkyl that is optionally substituted with one or more halogen atoms, e.g., F). Additionally, in some embodiments, R11B is selected from hydrogen, a halogen atom (e.g., F), and a substituted or unsubstituted alkyl group (e.g., a lower alkyl group which may optionally be substituted with one or more halogen atoms, e.g., F). And in some embodiments, at least one of R11A and R11 C includes a substituted or unsubstituted cyano group, or a substituted or unsubstituted alkyl cyano group.
In the above structures, the represents the binding site to Formula 1 , and R16 through R18 may be independently selected from the substituents described above in connection with R3 and R4 of Formula 1 . For example, in some embodiments, R16 may be selected from substituted and unsubstituted acyl groups (e.g., in which the alkyl component is a lower alkyl), substituted or unsubstituted lower alkyl groups, substituted or unsubstituted alkylsulfonyl groups (e.g., in which the alkyl component is a lower alkyl), and substituted or unsubstituted cycloalkyl (e.g., having from 3 to 6 carbon atoms). Also, in some embodiments, R17 may be selected from substituted or unsubstituted lower alkyl groups, and substituted or unsubstituted haloalkyl groups (e.g., in which the alkyl component is a lower alkyl). And in some embodiments, R18 may be hydrogen, or a halogen atom.
[0083] Some additional non-limiting examples of suitable R1 groups include the
[0084] In some embodiments, for example, R1 may be
[0085] Additional non-limiting examples and disclosure of suitable R1 groups can be found in the discussion of -X-R1 in U.S. Patent Publication No. 2019/0270743 to Mirati Therapeutics, Inc. and Array BioPharma Inc., titled “KRAS G12C Inhibitors,” filed on May 14, 2019 and published on September 5, 2019, the entire content of which is incorporated herein by reference, and in the discussion of -X-(CH2)n-R2 and -X-(CH2)n-C(=O)-C(R13)=C(R14)(R15) in WO 2020/035031 to Genentech, Inc. and F. Hoffman-La Roche AG, titled “Fused Ring Compounds,” published on February 20, 2020 and having an international filing date of August 15, 2019, the entire content of which is also incorporated herein by reference.
[0086] Returning back to Formula 1 , R2 on the pyrimidine ring fused to the spirocyclic core is not particularly limited, and may be any of the substituted or unsubstituted substituents described above with respect to R3 and R4. In some embodiments, however, R2 may be represented by Formula 8.
In Formula 8, * denotes the binding site to Formula 1 . Xs is a bond, -O-, -S- or -NRX5-. RX5 is not particularly limited, and may be any substituent described above in connection with R3 and R4 of Formula 1 . However, in some embodiments, RX5 may be hydrogen, or a substituted or unsubstituted alkyl group (e.g., a lower alkyl group which may optionally be substituted with one or more halogen atoms). [0087] R20 in Formula 8 is also not particularly limited, and may be any substituent described above in connection with R3 and R4 of Formula 1. However, in some embodiments, R20 may be hydrogen, a substituted or unsubstituted alkyl group, substituted or unsubstituted haloalkyl group, substituted or unsubstituted hydroxyalkyl group, substituted or unsubstituted dihydroxyalkyl group, substituted or
unsubstituted alkylaminoalkyl group, substituted or unsubstituted dialkylam inylalkyl group, -Zi-N(RX5R20A)
substituted or unsubstituted heterocyclic group, substituted or unsubstituted cycloalkyl group, substituted or unsubstituted heterocycloalkyl substituted or unsubstituted, substituted or unsubstituted aryl group, substituted or unsubstituted heteroaryl group, or substituted or unsubstituted heteroarylalkyl group. In the -Zi-N(RX5R20A) (RX5R20B) moiety, Zi may be a substituted or unsubstituted alkyl group (e.g., a lower alkyl group optionally substituted with one or more halogen atoms). Each of RX5R20A and RX5R20B may be hydrogen, a substituted or unsubstituted acyl group, substituted or unsubstituted alkyl group (e.g., a lower alkyl group optionally substituted with, e.g., one or more halogen atoms), substituted or unsubstituted haloalkyl group, substituted or unsubstituted heteroalkyl group, or substituted or unsubstituted hydroxyalkyl group. In some embodiments, RX5R20A may be hydrogen or a substituted or unsubstituted lower alkyl group, and RX5R20B may be hydrogen, a substituted or unsubstituted acyl group, substituted or unsubstituted alkyl group (e.g., a lower alkyl group optionally substituted with, e.g., one or more halogen atoms), substituted or unsubstituted haloalkyl group, substituted or unsubstituted heteroalkyl group, or substituted or unsubstituted hydroxyalkyl group.
[0088] In some embodiments, for example, X5 may be -O- or -NRX5-, and R20 may be a substituted or unsubstituted heterocyclic group, or a substituted or unsubstituted heterocycolalkyl group. Some non-limiting examples of suitable heterocyclic or heterocycloalkyl groups for R20 include azetidinyl, alkyl-substituted azetidinyl (e.g., lower alkyl azetidinyl, e.g., methylazetidinyl), halo-substituted azetidinyl (e.g., difluoroazetidinyl), tetrahydropyran, pyrrolidinyl, alkyl substituted pyrrolidinyl (e.g., lower alkyl pyrrolidinyl, e.g., methylpyrrolidinyl, dimethyl pyrrolidinyl, and isopropylpyrrolidinyl), cycloalkylalkylpyrrolidinyl, hydroxypyrrolindinyl, halo substituted pyrrolidinyl (e.g., fluoropyrrolidinyl and difluoropyrrolidinyl), halo- substituted N-methylpyrrolidinyl (e.g., N-methylfluoropyrrolidinyl and N- methyldifluoropyrrolidinyl), methoxyethylpyrrolidinyl, alkoxy-substituted N- methylpyrrolidinyl (e.g., (N-methyl) methoxypyrrolidinyl), piperazinyl, dimethylaminylpyrrolidinyl, morpholinyl, methylmorpholinyl, 1 ,4-oxazepanyl, piperidinyl, alkyl-substituted piperidinyl (e.g., lower alkyl substituted piperidinyl, e.g., methylpiperidinyl), acylpiperidinyl, cyanopiperidinyl, cycloalkylpiperidinyl, halopiperidinyl (e.g., fluoropiperidinyl), dihalopiperidinyl (e.g., difluoropiperidinyl), alkoxypiperidinyl, pyrrolidonyl, piperidonyl, thiomorpholinyl-1 ,1 -dioxide, 3- azabicyclo[3.1 .0]hexanyl, oxa-5-azabicyclo[2.2.1]heptan-5-yl, and azabicyclo[2.2.1]heptan-2-yl. For example, in some embodiments, the heterocyclic
group or heterocycloalkyl group of R20 may be N-methylpyrrolidinyl or 3,3-difluoro-1 - methylpyrrolidinyl.
[0089] In some embodiments, for example, X5 may be -O-, and R20 may be a substituted or unsubstituted heterocycloalkyl group. And in some embodiments, the heterocycloalkyl group has one or more ring heteroatoms selected from N and O. Some non-limiting examples of suitable R20 groups include substituted or unsubstituted groups such as azetidinyl, methylazetidinyl, difluoroazetidinyl, tetrahydropyran, pyrrolidinyl, methylpyrrolidinyl, diemethylpyrrolidinyl, isopropylpyrrolidinyl, cycloalkylalkylpyrrolidinyl, hydroxypyrrolindinyl, fluoropyrrolidinyl, difluoropyrrolidinyl, (N-methyl)fluoropyrrolidinyl, (N- methyl)difluoropyrrolidinyl, methoxyethylpyrrolidinyl, alkoxy-substituted N- methylpyrrolidinyl (e.g., (N-methyl)methoxypyrrolidinyl), piperazinyl, dimethylaminylpyrrolidinyl, morpholinyl, methylmorpholinyl, 1 ,4-oxazepanyl, piperidinyl, methylpiperidinyl, acylpiperidinyl, cyanopiperidinyl, cycloalkylpiperidinyl, halopiperidinyl, dihalopiperidinyl, fluoropiperidinyl, difluoropiperidinyl, alkoxypiperidinyl, pyrrolidonyl, piperidinonyl, thiomorpholinyl-1 ,1 -dioxide, 3- azabicyclo[3.1 .0]hexanyl, oxa-5-azabicyclo[2.2.1 ]heptan-5-yl, or azabicyclo[2.2.1 ]heptan-2-yl. In some embodiments, for example, R20 may be selected from substituted or unsubstituted hexahydro-1 H-pyrrol iziny I and substituted or unsubstituted octahydroindolizinyl.
In Formula 9, * represents the binding site to Formula 1 , RX5R20A and RX5R20B are as described above in connection with Formula 8, and RR2 is equivalent to R3 and R4 in Formula 1 , and may be any of the moieties described above in connection with R3 and R4 in Formula 1. However, in some embodiments, RR2 may be hydrogen, a halogen, a substituted or unsubstituted alkyl group (e.g., a lower alkyl optionally substituted with, e.g., one or more halogen atoms), or a substituted or unsubstituted hydroxyalkyl group (e.g., in which the alkyl component is a lower alkyl optionally substituted with, e.g., one or more halogen atoms).
[0091] Some nonlimiting examples of suitable structures for R2 of Formula 1 include the following:
[0092] In the above structures, the ? represents the binding site to Formula 1. [0093] Some additional non-limiting examples of suitable structures for R2 of Formula 1 include the following:
[0095] In some embodiments, for example, R2 may
[0096] According to some embodiments, the compound represented by Formula
1 may be a compound represented by one of Formula 10A, 10B and 10C.
In Formulae 10A, 10B and 10C, R3, R4, R5, R11, x, m, z and ring A are as described above in connection with Formulae 1 through 7D.
[0097] According to some embodiments, the compound represented by Formula 1 may be a compound represented by one of Formula 11A, 11 B and 11 C.
In Formulae
are as described above with respect to Formulae 1 through 9. However, in some embodiments, R4 may be selected from hydrogen, methyl, ethyl and isopropyl. Also, in some embodiments, R5 may be selected from hydrogen and lower alkyl groups, e.g., methyl, ethyl, and isopropyl. And in some embodiments, R11A may be selected from hydrogen, -CH2-C=N and -CH2-C=CH. Additionally, in some embodiments, R11 B may be selected from hydrogen and methyl. In some embodiments, R11 C may be selected from hydrogen, methyl and -CH2-C=N. According to some embodiments, R16 may be selected from hydrogen, -CH2F, -CHF2, -CF3, methyl, ethyl and -CH2-NH-(lower alkyl) (e.g., -CH2-NH-methyl, -CH2-NH-ethyl, -CH2-NH- isopropyl). And in some embodiments, R17 and RR2 may be selected from hydrogen and a halogen (e.g., F).
[0098] Additional non-limiting examples and disclosure of suitable R2 groups can be found in the discussion of -Y-R2 in U.S. Patent Publication No. 2019/0270743 to Mirati Therapeutics, Inc. and Array BioPharma Inc., titled “KRAS G12C Inhibitors,” filed on May 14, 2019 and published on September 5, 2019, the entire content of which is incorporated herein by reference, and in the discussion of Z2 and its optional substituents in WO 2020/035031 to Genentech, Inc. and F. Hoffman-La Roche AG, titled “Fused Ring Compounds,” published on February 20, 2020 and having an international filing date of August 15, 2019, the entire content of which is also incorporated herein by reference.
[0099] Some non-limiting examples of suitable compounds satisfying Formula 1 include the following compounds:
In the above structures (and indeed, in any of the structures and Formulae in this disclosure), if no stereochemistry is shown, it is understood that the compound (or moiety) may have any suitable stereochemistry without limitation.
[00100] In some embodiments, for example, the compound represented by Formula 1 may be selected from the following compounds:
25
30
35
[00101] According to some embodiments, a pharmaceutical composition may include one or more of the compounds described herein (or pharmaceutically acceptable salts or derivatives thereof) in a therapeutically effective amount. In some embodiments, the pharmaceutical composition may include one or more compound represented by Formula 1 (or pharmaceutically acceptable salt thereof), as well as one or more pharmaceutically acceptable carriers, excipients, adjuvants and/or diluents. Acceptable carriers, excipients and diluents are well known in the art and can be selected with regard to the intended route of administration and standard practice. Some non-limiting examples include binders, lubricants, suspending agents, coating agents, solubilizing agents, preserving agents, wetting agents, emulsifiers, surfactants, sweeteners, colorants, flavoring agents, odorants, buffers, antioxidants, stabilizing agents and/or salts.
[00102] Compounds of the present disclosure may be formulated by any method well known in the art and may be prepared for administration by any route, including, without limitation, parenteral, oral, sublingual, transdermal, topical, intranasal,
intratracheal, or intrarectal. In some embodiments, compounds of the invention are administered intravenously in a hospital setting. In some embodiment, administration may be by the oral route.
[00103] The characteristics of the carrier will depend on the route of administration. As used herein, the term “pharmaceutically acceptable” means a non-toxic material that is compatible with a biological system such as a cell, cell culture, tissue, or organism, and that does not interfere with the effectiveness of the biological activity of the active ingredient(s). Thus, compositions according to the present disclosure may contain, in addition to the inhibitor, diluents, fillers, salts, buffers, stabilizers, solubilizers, and other materials well known in the art. The preparation of pharmaceutically acceptable formulations is described in, e.g., Remington's Pharmaceutical Sciences, Mack Publishing Company, Easton, Pa., 18th edition, 1990.
[00104] As used herein, the term “pharmaceutically acceptable salt” refers to salts that retain the desired biological activity of the compounds and exhibit minimal or no undesired toxicological effects. Non-limiting examples of suitable such pharmaceutically acceptable salts include acid addition salts formed with inorganic acids (for example, hydrochloric acid, hydrobromic acid, sulfuric acid, phosphoric acid, nitric acid, and the like), and salts formed with organic acids (such as, e.g., acetic acid, oxalic acid, tartaric acid, succinic acid, malic acid, ascorbic acid, benzoic acid, tannic acid, pamoic acid, alginic acid, polyglutamic acid, naphthalenesulfonic acid, naphthalene disulfonic acid, and polygalacturonic acid).
[00105] The compounds described herein may also be administered as pharmaceutically acceptable quaternary salts known to those skilled in the art, e.g., quaternary ammonium salts represented by -N(R)2- , wherein R is hydrogen, alkyl, or benzyl, and Z is a counterion (non-limiting examples of which include chloride, bromide, iodide, O-alkyl, toluenesulfonate, methylsulfonate, sulfonate, phosphate, or carboxylate (non-limiting examples of which include benzoate, succinate, acetate, glycolate, maleate, malate, citrate, tartrate, ascorbate, benzoate, cinnamoate, mandeloate, benzyloate, and diphenylacetate).
[00106] According to some embodiments, the compound(s) described herein (e.g., the one or more compound represented by Formula 1) may be present in the pharmaceutical compositions together with the pharmaceutically acceptable carrier or diluent in any suitable amount. For example, the compound(s) may be present in an amount sufficient to deliver a therapeutically effective amount of the compound(s) to a patient or subject without causing (or minimizing the risk of) serious toxic effects in the patient or subject. In some embodiments, for example, the active compound (i.e., the compound(s) of Formula 1 ) may be administered by any route in a dose of
about 0.01 to 300 mg/kg per day, for example about 0.1 to 100 mg/kg per day, or about 0.5 to about 25 mg/kg per day. In some embodiments, the active compound may be administered via a topical route in a dose of about 0.01-3% wt/wt in a suitable carrier. As would be understood by those of ordinary skill in the art, the effective dosage range of pharmaceutically acceptable derivatives or salts can be calculated based on the weight of the parent compound to be delivered. And if the derivative or salt exhibits activity in itself, the effective dosage can be estimated as above using the weight of the derivative, or by any other means known to those of ordinary skill in the art.
[00107] According to some embodiments of the present disclosure, a method of inhibiting RAS protein forms includes contacting a cell in which inhibition is desired with an effective amount of one or more compounds represented by Formula 1 (or one or more pharmaceutically acceptable salts or derivatives thereof). For example, in some embodiments, the method of inhibiting RAS protein forms may include administering a pharmaceutical composition as described herein to a patient or subject in need of RAS protein form inhibition. Contacting the cell with the compound(s), salt(s) or derivative(s) thereof, or the pharmaceutical composition may occur in vivo or in vitro. The contacting (or administration) may be accomplished in a single dose or over multiple doses.
[00108] According to some embodiments, a method of treating a condition or disease includes administering to a patient or subject having the disease or condition a therapeutically effective amount of one or more compounds represented by Formula 1 (or one or more pharmaceutically acceptable salts or derivatives thereof). For example, in some embodiments, the method may include administering a pharmaceutical composition as described herein to a patient or subject having the disease or condition. The disease or condition treated using these methods is not particularly limited, and may be any disease or condition. In some embodiments, however, the disease or condition treated is one that is caused by a mutation in KRAS, including various solid and blood cancers. Some non-limiting examples of cancers to be treated using methods according to embodiments of the present disclosure include lung, pancreatic, colon, thyroid and melanoma cancers, as well as the cancers listed in: 1 ) WO 2020/035031 to Genentech, Inc. and F. Hoffman-La Roche AG, titled “Fused Ring Compounds,” published on February 20, 2020 and having an international filing date of August 15, 2019; and 2) U.S. Patent Publication No. 2019/0270743 to Mirati Therapeutics, Inc. and Array BioPharma Inc., titled “KRAS G12C Inhibitors,” filed on May 14, 2019 and published on September 5, 2019, the entire contents of both of which are incorporated herein by reference.
[00109] In some embodiments, the method of treating a condition or disease may further comprise first determining if the patient or subject to be treated has a KRAS mutation. If the patient or subject to be treated is determined to have such a mutation, then the method includes administering to the patient or subject the therapeutically effective amount of one or more compound(s) described herein, a pharmaceutically acceptable salt or derivative thereof, or a pharmaceutical composition described herein.
[00110] As would be understood by those of ordinary skill in the art, the concentration, treatment protocol, and administration route will vary depending on the particular patient and type of cancer to be treated. Additionally, the compounds described herein, pharmaceutically acceptable salts or derivatives thereof, and pharmaceutical compositions described herein may be co-administered with other therapies (e.g., anti-neoplastic compounds, such as chemotherapy agents), or administered in combination with other treatments (such as radiation or surgical intervention). When used in combination with surgical intervention, for example, the compounds and pharmaceutical compositions described herein may be used either as an adjuvant prior to surgery or may be used post-operatively.
Examples
[00111] The following examples and comparative examples are provided for illustrative purposes only, and do not limit the scope or content of this disclosure.
[00113] NaH (8.21 g, 205 mmol, 60% purity, 1.50 eq) was added to a solution of Compound 1 (20.0 g, 136 mmol, 18.2 mL, 1 eq) in THF (120 mL) at 0 °C under N2. And then dimethyl carbonate (14.8 g, 164 mmol, 13.8 mL, 1.20 eq) was added to the mixture. The mixture was stirred at 60 °C for 12 hrs. TLC (Petroleum ether/Ethyl acetate = 5/1 , Rf = 0.76) indicated the reactant was consumed completely and two new spots formed. The reaction was clean according to TLC. The mixture was added to NH4CI (200 mL) and extracted with ethyl acetate (200 mL), and the organic phase was concentrated under reduced pressure to give the residue. The residue was purified by silica gel chromatography (column height: 250 mm, diameter: 100 mm, 100-200 mesh silica gel, Petroleum ether/Ethyl acetate = 20/1 , 10/1 ) to afford compound 17 (25.0 g, 122 mmol, 89.4% yield) as a light yellow oil. Reaction 2
17 18
[00114] K2CO3 (25.4 g, 183 mmol, 1 .50 eq) was added to a solution of compound 17 (25.0 g, 122 mmol, 1 eq) in DMSO (150 mL) at 20 °C under N2. Then 5- bromopent-1 -ene (21 .9 g, 146 mmol, 1 .20 eq) was added to the mixture. The mixture was stirred at 20 °C for 12 hrs. TLC indicated the reactant was consumed completely and two new spots formed. The reaction was clean according to TLC. The mixture was quenched with NH4CI (50 mL) and extracted with ethyl acetate (100 mL), the organic phase was concentrated under reduced pressure to give the residue. The residue was purified by silica gel chromatography (column height: 250 mm, diameter: 100 mm, 100-200 mesh silica gel, Petroleum ether/Ethyl acetate = 20/1 , 3/1 ) to afford compound 18 (26.0 g, 95.5 mmol, 77.9% yield) as a light yellow oil.
18 14
[00115] NaOH (7.64 g, 190 mmol, 2.00 eq) was added to a solution of Compound 18 (26.0 g, 95.5 mmol, 1 .00 eq) in EtOH (26.0 mL) and H2O (180 mL) with stirring. The mixture was stirred at 100 °C for 1 hr. TLC indicated the reactant was consumed completely and three new spots formed. The reaction was clean according to TLC. The mixture was concentrated under reduced pressure to give the residue, and the residue was extracted with ethyl acetate (150 ml). The residue was purified by silica gel chromatography (column height: 250 mm, diameter: 100 mm, 100-200 mesh silica gel, Petroleum ether/Ethyl acetate = 20/1 , 5/1 ) to afford compound 14 (19.5 g, 90.9 mmol, 95.3% yield) as a light yellow oil.
[00116] NaH (5.32 g, 133 mmol, 60% purity, 1.50 eq) was added to a solution of Compound 14 (19.0 g, 88.6 mmol, 1.00 eq) in THF (114 mL) at 0 °C under N2. Then 3-bromoprop-1-ene (12.8 g, 106 mmol, 1.20 eq) was added to the mixture and stirred at 20 °C for 2 hrs. The mixture was stirred at 70 °C for 12 hrs. TLC indicated the reactant was consumed completely and two new spots formed. The reaction was clean according to TLC. The reaction was quenched with saturated NH4CI (150 mL) and extracted with ethyl acetate (150 mL), and washed with saturated brines (100 mL*2). The organic phase was concentrated under reduced pressure to give the residue. The residue was purified by silica gel chromatography (column height: 250 mm, diameter: 100 mm, 100-200 mesh silica gel, Petroleum ether/Ethyl acetate = 1/0, 20/1 ) to afford the compound. Compound 15 (20.1 g, 79.0 mmol, 89.1% yield) was obtained as a light yellow oil. Reaction 5
[00117] To a solution of compound 15 (19.0 g, 74.7 mmol, 1 .00 eq) in acetone (76 mL) and H2O (38 mL) was added NalO4 (95.8 g, 448 mmol, 24.8 mL, 6.00 eq) at 20 °C under N2. KMnO4 (11.8 g, 74.7 mmol, 1 .00 eq) was added to the mixture at 20 °C. The mixture was stirred at 20 °C for 2 hrs. LCMS (ET35223-22-P1 A) showed
reactant was consumed completely and the reaction was worked well. The mixture was filtered to give the filtrate and concentrated under reduced pressure to give the residue. The pH of the residue was adjusted to 9 by saturated Na2COs, and extracted with ethyl acetate (100 mL). And the pH of the aqueous phase was adjusted to 4 by 3N HCI. The aqueous phase was extracted with ethyl acetate (100 ml), and concentrated under reduced pressure give the residue. Compound 16 (19.0 g, crude) was obtained as a light yellow oil. Reaction 6
[00118] To a solution of compound 16 (19.0 g, 65.4 mmol, 1 .00 eq) in DMF (114 mL) was added K2CO3 (27.1 g, 196 mmol, 3.00 eq) at 20 °C under N2. Mel (20.4 g,
143 mmol, 8.96 mL, 2.20 eq) was added to the mixture and the mixture was stirred at 20 °C for 12 hrs. LCMS (ET35223-23-P1A) showed the reactant was consumed completely and the reaction was worked well. TLC indicated the reactant was consumed completely and three new spots formed. The reaction was clean according to TLC. The mixture was concentrated under reduced pressure to give the residue. The residue was purified by silica gel chromatography (column height:
250 mm, diameter: 100 mm, 100-200 mesh silica gel, Petroleum ether/Ethyl acetate=1/0, 5/1 ) to afford compound 3 (10.3 g, 32.3 mmol, 49.4% yield) as a light yellow oil.
[00119] To a solution of compound 3 (10.0 g, 31.4 mmol, 1.00 eq) was added EtsSiH (36.5 g, 314 mmol, 50.1 mL, 10.0 eq) and TFA (35.8 g, 314 mmol, 23.2 mL, 10.0 eq). The mixture was stirred at 50 °C for 12 hrs. TLC indicated the reactant
was consumed completely and three new spots formed. The reaction was clean according to TLC. LCMS (ET35223-25-P1A) showed the reaction was worked well. The mixture was quenched with saturated Na2COs (50.0 mL) and extracted with ethyl acetate (50.0 mL*2). The organic phase was concentrated under reduced pressure to give the residue. The residue was purified by silica gel chromatography
(column height: 250 mm, diameter: 100 mm, 100-200 mesh silica gel, Petroleum ether/Ethyl acetate = 10/1 , 3/1 ) to afford compound 4 (4.00 g, 13.1 mmol, 41.8% yield) as a yellow oil.
[00120] To a solution of compound 4 (4.00 g, 13.1 mmol, 1 .00 eq) in THF (24.0 mL) was added t-BuOK (2.21 g, 19.7 mmol, 1 .50 eq) at 0 °C. The mixture was stirred at 20 °C for 12 hrs. TLC indicated the reactant was consumed completely and one new spot formed. The reaction was clean according to TLC. The mixture was quenched by NH4CI (30.0 mL) and extracted with ethyl acetate (50.0 mL), and concentrated under reduced pressure to give the residue. Compound 5 (3.60 g, crude) was obtained as a light yellow oil and checked by HNMR (ET35223-27-P1A).
[00121] To a solution of compound 5 (3.20 g, 11.7 mmol, 1 .00 eq), urea (16.0 g, 266 mmol, 14.2 mL, 22.6 eq) was added with stirring. The mixture was stirred at 190°C for 1 hr. LCMS (ET35223-30-P1A) showed the reactant was consumed completely and the reaction was worked well. The mixture was cooled to 20 °C, and then H2O (20.0 mL) was added to the mixture. Then the mixture was filtered to give
the filter cake. Compound 6 (2.20 g, crude) was obtained as a white solid and checked by HNMR (ET35223-30-P1A).
[00122] Compound 6 (2.20 g, 7.79 mmol, 1 .00 eq) was dissolved in POCI3 (36.3 g, 236 mmol, 22.0 mL, 30.3 eq) and the mixture was stirred at 120 °C for 1 hr. TLC and LCMS (ET35223-31-P1A) showed the reaction was worked well. The mixture was concentrated under reduced pressure to remove POCI3, then the mixture was quenched with H2O (20.0 ml) and extracted with ethyl acetate (30.0 ml). The residue was purified by silica gel chromatography (column height: 250 mm, diameter: 100 mm, 100-200 mesh silica gel, Petroleum ether/Ethyl acetate = 20/1 , 5/1 ) to afford compound 7 (1 .20 g, 3.76 mmol, 48.2% yield) as a light yellow oil.
[00123] To a solution of compound 7 (1.00 g, 3.13 mmol, 1.00 eq) in DMF (2.00 mL) was added compound 8 (705 mg, 3.13 mmol, 1 .00 eq), and then DIEA (809 mg, 6.27 mmol, 1 .09 mL, 2.00 eq) was added. The mixture was stirred at 70 °C for 12 hrs. TLC indicated the reactant was consumed completely and two new spots formed. The reaction was clean according to TLC. The mixture was washed with H2O (20.0 ml) and the organic phase was concentrated under reduced pressure to give the residue. The residue was purified by silica gel chromatography (column
height: 250 mm, diameter: 100 mm, 100-200 mesh silica gel, Petroleum ether/Ethyl acetate = 20/1 , 3/1 ) to afford compound 9 (1.10 g, 2.17 mmol, 69.1 % yield) as a white solid. The product was checked by HNMR (ET35223-37-P1 A).
[00124] Compound 9 (0.30 g, 590 umol, 1.00 eq) was dissolved in toluene (10.0 mL), and then compound 10 (136 mg, 1.18 mmol, 2.00 eq) and Pd2(dba)3 (27.0 mg, 29.5 umol, 0.05 eq), t-BuONa (170 mg, 1.77 mmol, 2.00 eq), and BINAP (73.5 mg, 118.1 umol, 0.20 eq) were added to the solution. The mixture was stirred at 110 °C for 0.5 hr. TLC indicated the reactant was consumed completely and three new spots formed. The reaction was clean according to TLC. The mixture was washed with H2O (20.0 mL) and extracted with Ethyl acetate (20.0 mL), and then the organic phase was concentrated under reduced pressure to give the residue. The residue was purified by silica gel chromatography (column height: 250 mm, diameter: 100 mm, 100-200 mesh silica gel, Petroleum ether/Ethyl acetate = 10/1 , 0/1 ) to afford compound 11 (0.25 g, 426 umol, 72.1 % yield) as a white solid.
[00125] To a solution of compound 11 (0.20 g, 340 umol, 1 .00 eq) in MeOH (5.00 mL) was added HCI/MeOH (4 M, 5.00 mL, 58.6 eq) at 20 °C. The mixture was stirred at 20 °C for 1 hr. LCMS (ET35223-41-P1A) showed the reactant was
consumed completely and the reaction worked well. The mixture was concentrated under reduced pressure to give the residue. Compound 12 (0.20 g, crude, HCI) was obtained as a white solid.
[00126] To a solution of compound 12 (0.20 g, 382 umol, 1 .00 eq, HCI) in DCM (2.00 mL) was added Et3N (116 mg, 1.15 mmol, 159 uL, 3.00 eq) at 0 °C under N2. Prop-2-enoyl chloride (41.5 mg, 458 umol, 37.4 uL, 1.20 eq) was added to the mixture at 0 °C and the mixture was stirred at 20 °C for 6 hrs. LCMS (ET35223-42- P1A) and HPLC (ET35223-42-P1 B) showed the reactant was consumed completely and the reaction worked well. The mixture was washed with H2O (10.0 ml), and separated to give the organic phase. The residue was purified by Pre-HPLC (column: Phenomenex Gemini-NX 80*40mm*3um; mobile phase: [water (10m M NH4HCO3)-ACN]; B%: 25%-55%, 8 min). Compound 13 (0.07 g, 129 umol, 33.8% yield) was obtained as a white solid.
[00127] The product was purified by SFC (column: DAICEL CHIRALCEL OD (250mm*30mm, 10 um)) mobile phase: [0.1 %NH3H2O IPA]; B%: 50%-50%, 15 min). [00128] A first diastereomer of purified Compound 13 (0.011 g, 20.3 umol, 22.0% yield) was obtained as a white solid and checked by LCMS (ET35223-43-P1 H) and HNMR (SIT-002_P1_NMR), SFC (SIT-002_P1_SFC). LCMS: ET35223-43-P1 H; Product Rt = 1.870min, m/z = 541.2 (M+H)+
[00129] 1H NMR (400MHz, CDCI3-d6) ET35223-43-P1 D: 5 = 7.18 - 6.98 (m, 4H), 6.56 (br d, J = 10.5 Hz, 1 H), 6.43 - 6.32 (m, 1 H), 5.81 (br d, J = 10.3 Hz, 1 H), 4.34 (dd, J = 5.0, 10.5 Hz, 1 H), 4.20 - 3.80 (m, 4H), 3.35 - 3.21 (m, 1 H), 3.15 - 2.56 (m, 14H), 2.35 - 2.21 (m, 1 H), 2.09 - 1 .57 (m, 12H)
[00130] A second diastereomer of Compound 13 (0.012 g, 22.1 umol, 24.0% yield) was obtained as a white solid and checked by LCMS (ET35223-43-P2E) and HNMR (SIT-002_P2_NMR), SFC (SIT-002_P2_SFC). LCMS: ET35223-43-P2E; Product Rt = 1 .941 min, m/z = 541 .2 (M+H)+
[00131] 1 H NMR (400MHz, CDCI3-d6) ET35223-43-P2B
[00132] δ = 7.14 - 7.03 (m, 4H), 6.58 (br s, 1H), 6.44 - 6.33 (m, 1H), 5.81 (br d, J = 10.1 Hz, 1H), 4.42 (brs, 1H), 4.20-4.11 (m, 1H), 4.08-3.91 (m, 2H), 3.30 (br dd, J = 3.7, 14.0 Hz, 1H), 3.17 (brs, 1H), 2.92-2.82 (m, 3H), 2.79-2.48 (m, 10H), 2.35 (brs, 1H), 2.14-1.58 (m, 13H)
[00133] Table 1 below shows the IC50 values or inhibition behavior achieved by the racemate and two stereoisomers of Example 1 under the conditions indicated in the table.
* In each of the stereoisomers shown in Table 1 , the absolute configuration of the asterisked spiro-carbon atom is arbitrarily assigned for illustrative purposes.
[00134] As can be seen in Table 1 , the Example 1 compound (and stereoisomers) achieved 50% inhibition after incubation at 21 °C for 15 minutes at concentrations under 0.3 pM, and achieved 50% inhibition after incubation at 4°C for 18 hours at
concentrations of 0.06 pM or below. In contrast, as shown in Table 2 on page 473 of WO 2020/035031 to Genentech, Inc. and F. Hoffman-La Roche AG, titled “Fused Ring Compounds,” published on February 20, 2020 and having an international filing date of August 15, 2019, the entire content of which is also incorporated herein by reference, the following Comparative Compound 1 achieved 50% inhibition (albeit at different incubation conditions which are described on page 471 ) at a significantly higher concentration of 5.4 pM.
As can be generally seen by this comparison, the compounds of embodiments of the present disclosure achieve significantly greater KRAS inhibition at much lower concentrations than, e.g., Comparative Compound 1.
[00135] While certain exemplary embodiments of the present disclosure have been illustrated and described, those of ordinary skill in the art will recognize that various changes and modifications can be made to the described embodiments without departing from the spirit and scope of the present invention, and equivalents thereof, as defined in the claims that follow this description. For example, although certain components may have been described in the singular, i.e. , "a" compound, "an" excipient, and the like, one or more of these components in any combination can be used according to the present disclosure.
[00136] Also, although certain embodiments have been described as "comprising" or "including" the specified components, embodiments "consisting essentially of" or "consisting of' the listed components are also within the scope of this disclosure. For example, while embodiments of the present invention are described as comprising a compound represented by Formula 1 or a pharmaceutical composition comprising a compound represented by Formula 1 and an excipient, adjuvant or carrier, embodiments consisting essentially of or consisting of these components are also
within the scope of this disclosure. Accordingly, a pharmaceutical composition may consist essentially of a compound represented by Formula 1 and one or more excipients, adjuvants or carriers. In this context, "consisting essentially of" means that any additional components will not materially affect the chemical, physical, therapeutic or pharmaceutical properties of the pharmaceutical composition.
[00137] As used herein, unless otherwise expressly specified, all numbers such as those expressing values, ranges, amounts or percentages may be read as if prefaced by the word "about," even if the term does not expressly appear. Further, the word "about" is used as a term of approximation, and not as a term of degree, and reflects the penumbra of variation associated with measurement, significant figures, and interchangeability, all as understood by a person having ordinary skill in the art to which this disclosure pertains. Any numerical range recited herein is intended to include all sub-ranges subsumed therein. Plural encompasses singular and vice versa. For example, while the present disclosure may describe "a" compound or "an" excipient, a mixture of such materials can be used. When ranges are given, any endpoints of those ranges and/or numbers within those ranges can be combined within the scope of the present disclosure. The terms "including" and like terms mean "including but not limited to," unless specified to the contrary.
[00138] Notwithstanding that the numerical ranges and parameters set forth herein may be approximations, numerical values set forth in the Examples are reported as precisely as is practical. Any numerical value, however, inherently contains certain errors necessarily resulting from the standard variation found in their respective testing measurements. The word "comprising" and variations thereof as used in this description and in the claims do not limit the disclosure to exclude any variants or additions.
Claims
[00139] WHAT IS CLAIMED IS:
1 . A compound capable of inhibiting a RAS protein form, the compound being represented by Formula 1 :
Formula 1
wherein, in Formula 1 : each of R1-R2 is independently selected from the group consisting of halogens, substituted or unsubstituted hydrocarbon substituents, hydrogen, deuterium, hydroxyl groups, cyano groups, amino groups, and oxo groups (=0); each of R3-R4 is independently selected from the group consisting of substituted or unsubstituted hydrocarbon substituents, hydrogen, deuterium, F, hydroxyl groups, cyano groups, amino groups, or oxo groups (=0), wherein two geminal R3s or two geminal R4s bonded to a common carbon atom may optionally combine to form an additional spirocycle from the common carbon atom; each of X1-X3 is independently a bond, a moiety containing a ring C atom, or a moiety containing a ring heteroatom the ring A is a cyclic, heterocyclic, aryl or heteroaryl ring; and each of x and m is an integer from 1 to 6.
2. The compound according to claim 1 , wherein R1 is represented by one of Formulae 4A and 4B:
Formula 4A Formula 4B
wherein: in Formulae 4A and 4B, * represents a binding site to Formula 1 ; in Formula 4A, X4 is selected from the group consisting of substituted or unsubstituted hydrocarbon substituents, hydrogen, deuterium, halogens, hydroxyl groups, cyano groups, amino groups, or oxo groups (=0); in Formula 4B, X4 is selected from the group consisting of substituted or unsubstitutd, saturated or partially saturated monocyclic, bridged or spirocyclic rings having 2 nitrogen ring atoms; and in Formula 4A and 4B, R14 is selected from the group consisting of substituted or unsubstituted hydrocarbon substituents, hydrogen, deuterium, halogens, hydroxyl groups, cyano groups, amino groups, or oxo groups (=0).
3. The compound according to claim 2, wherein R14 is represented by one of Formulae 5A through 5E:
R15 through R19 are each independently selected from the group consisting of substituted or unsubstituted hydrocarbon substituents, hydrogen, deuterium, halogens, hydroxyl groups, cyano groups, amino groups, or oxo groups (=0).
4. The compound according to claim 1 , wherein each of X1-X3 is independently a moiety containing a ring C atom or a ring heteroatom selected from N, O and S.
5. The compound according to claim 1 , wherein the compound of Formula
1 is represented by one of Formulae 1 A to 1 H:
6. The compound according to claim 1 , wherein the compound of Formula
1 is represented by one of Formulae 2A to 2B:
Formula 2A Formula 2B
wherein R5 is independently selected from the group consisting of substituted or unsubstituted hydrocarbon substituents, hydrogen, deuterium, F, hydroxyl groups, cyano groups, amino groups, or oxo groups (=0).
7. The compound according to claim 1 , wherein the ring A is a substituted or unsubstituted aryl ring or a substituted or unsubstituted heteroaryl ring.
-110-
8. The compound according to claim 1 , wherein the ring A has 5 or 6 ring atoms.
9. The compound according to claim 1 , wherein the ring A is selected from the group consisting of benzene, thiophene, furan, pyran, thiopyran, pyrrole, imidazole, pyrazole, triazole, pyridine, pyrimidine, pyridazine, pyrazine, oxazole, thiazole and cyclopentadiene.
10. The compound according to claim 1 , wherein the compound of Formula
-119-
-122-
-123-
-125-
-126-
-127-
-128-
-129-
12. A pharmaceutical composition, comprising at least one compound according to claim 1 or a pharmaceutically acceptable salt or derivative thereof.
13. The pharmaceutical composition according to claim 12, further comprising a pharmaceutically acceptable carrier, excipient, additive, adjuvant or diluent.
14. A method of inhibiting a RAS protein form, the method comprising contacting a cell in which inhibition is desired with at least one compound according to claim 1 or a pharmaceutically acceptable salt or derivative thereof.
-130-
15. A method of inhibiting a RAS protein form, the method comprising administering to a patient or subject the pharmaceutical composition according to claim 12.
16. A method of treating a disease or condition, the method comprising administering to a patient or subject having the disease or condition at least one compound according to claim 1 or a pharmaceutically acceptable salt or derivative thereof.
17. The method of claim 16, wherein the disease or condition is a cancer.
18. A method of treating a disease or condition, the method comprising administering to a patient or subject having the disease or condition the pharmaceutical composition according to claim 11 .
19. The method according to claim 17, wherein the disease or condition is a cancer.
-131-
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202063067812P | 2020-08-19 | 2020-08-19 | |
| US63/067,812 | 2020-08-19 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022040469A1 true WO2022040469A1 (en) | 2022-02-24 |
Family
ID=77774984
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2021/046773 Ceased WO2022040469A1 (en) | 2020-08-19 | 2021-08-19 | Spiro compounds as kras inhibitors |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2022040469A1 (en) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022235870A1 (en) | 2021-05-05 | 2022-11-10 | Revolution Medicines, Inc. | Ras inhibitors for the treatment of cancer |
| WO2022235864A1 (en) | 2021-05-05 | 2022-11-10 | Revolution Medicines, Inc. | Ras inhibitors |
| US11845761B2 (en) | 2020-12-18 | 2023-12-19 | Erasca, Inc. | Tricyclic pyridones and pyrimidones |
| US11912723B2 (en) | 2022-02-09 | 2024-02-27 | Quanta Therapeutics, Inc. | KRAS modulators and uses thereof |
| US12145947B2 (en) | 2022-05-25 | 2024-11-19 | Quanta Therapeutics, Inc. | Pyrimidine based modulators and uses thereof |
| US12162893B2 (en) | 2020-09-23 | 2024-12-10 | Erasca, Inc. | Tricyclic pyridones and pyrimidones |
| US12421254B2 (en) | 2023-03-15 | 2025-09-23 | Quanta Therapeutics, Inc. | KRAS modulators and uses thereof |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20190270743A1 (en) | 2017-11-15 | 2019-09-05 | Mirati Therapeutics, Inc. | Kras g12c inhibitors |
| WO2020035031A1 (en) | 2018-08-16 | 2020-02-20 | Genentech, Inc. | Fused ring compounds |
| WO2020236940A1 (en) * | 2019-05-20 | 2020-11-26 | California Institute Of Technology | Kras g12c inhibitors and uses thereof |
| WO2021093758A1 (en) * | 2019-11-15 | 2021-05-20 | 四川海思科制药有限公司 | Pyrimido derivative and application thereof in medicine |
| WO2021139748A1 (en) * | 2020-01-08 | 2021-07-15 | Ascentage Pharma (Suzhou) Co., Ltd. | Spirocyclic tetrahydroquinazolines |
-
2021
- 2021-08-19 WO PCT/US2021/046773 patent/WO2022040469A1/en not_active Ceased
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20190270743A1 (en) | 2017-11-15 | 2019-09-05 | Mirati Therapeutics, Inc. | Kras g12c inhibitors |
| WO2020035031A1 (en) | 2018-08-16 | 2020-02-20 | Genentech, Inc. | Fused ring compounds |
| WO2020236940A1 (en) * | 2019-05-20 | 2020-11-26 | California Institute Of Technology | Kras g12c inhibitors and uses thereof |
| WO2021093758A1 (en) * | 2019-11-15 | 2021-05-20 | 四川海思科制药有限公司 | Pyrimido derivative and application thereof in medicine |
| WO2021139748A1 (en) * | 2020-01-08 | 2021-07-15 | Ascentage Pharma (Suzhou) Co., Ltd. | Spirocyclic tetrahydroquinazolines |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12162893B2 (en) | 2020-09-23 | 2024-12-10 | Erasca, Inc. | Tricyclic pyridones and pyrimidones |
| US11845761B2 (en) | 2020-12-18 | 2023-12-19 | Erasca, Inc. | Tricyclic pyridones and pyrimidones |
| WO2022235870A1 (en) | 2021-05-05 | 2022-11-10 | Revolution Medicines, Inc. | Ras inhibitors for the treatment of cancer |
| WO2022235864A1 (en) | 2021-05-05 | 2022-11-10 | Revolution Medicines, Inc. | Ras inhibitors |
| US11912723B2 (en) | 2022-02-09 | 2024-02-27 | Quanta Therapeutics, Inc. | KRAS modulators and uses thereof |
| US12145947B2 (en) | 2022-05-25 | 2024-11-19 | Quanta Therapeutics, Inc. | Pyrimidine based modulators and uses thereof |
| US12421254B2 (en) | 2023-03-15 | 2025-09-23 | Quanta Therapeutics, Inc. | KRAS modulators and uses thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2022040469A1 (en) | Spiro compounds as kras inhibitors | |
| AU2019262927B2 (en) | Pyridazinones as PARP7 inhibitors | |
| RU2691629C1 (en) | Imidazopyrimidine derivatives as modulators of tnf activity | |
| ES2743208T3 (en) | Tetrahydroimidazopyridine derivatives as modulators of TNF activity | |
| US6809199B2 (en) | (Halo-benzo carbonyl)heterocyclo fused phenyl p38 kinase inhibiting agents | |
| CN103987700B (en) | 4-quinazoline amine derivant and application thereof | |
| RU2684637C1 (en) | Tetrahydrobenzimidazole derivatives as tnf activity modulators | |
| WO2020181283A1 (en) | Carboxamide-pyrimidine derivatives as shp2 antagonists | |
| EP3483158B1 (en) | Heterocyclic compound serving as fgfr4 inhibitor | |
| WO2021083135A1 (en) | Bcl-2 INHIBITORS | |
| WO2000044743A1 (en) | Amide derivatives and drug compositions | |
| AU2011288876A2 (en) | Phthalazinone ketone derivative, preparation method thereof, and pharmaceutical use thereof | |
| AU2009331179A1 (en) | Novel bicyclic heterocyclic compound | |
| WO2024193439A1 (en) | Shp2 phosphatase allosteric inhibitors | |
| EP3478675A1 (en) | Heteroaromatic derivatives as nik inhibitors | |
| JP6483700B2 (en) | Imidazothiazole Derivatives as Modulators of TNF Activity | |
| EP1515965A1 (en) | Phenylaminopyrimidines and their use as rho-kinase inhibitors | |
| CN116635377A (en) | Ketokinase inhibitors and uses thereof | |
| WO2010026113A1 (en) | Acyl- and sufonyl tetrahydronaphthyridines and aza derivatives thereof as histamine h3 receptor antagonists | |
| CA3090876C (en) | Dioxinoquinoline compounds, preparation method and uses thereof | |
| EP2989104B1 (en) | Pyrazino[1,2-a]indole compounds, their preparation and use in medicaments | |
| CN117736205A (en) | Fused ring compounds as MC4R antagonists, pharmaceutical compositions and their use in medicine | |
| KR102882224B1 (en) | ROCK kinase inhibitors | |
| WO2011078226A1 (en) | Tricyclic compound | |
| US5081128A (en) | 2,3-dihydro-1h-isoindole derivatives and their application in therapy |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 21770349 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 21770349 Country of ref document: EP Kind code of ref document: A1 |