WO2022011531A1 - Anti-cld18a2 single-domain antibody - Google Patents
Anti-cld18a2 single-domain antibody Download PDFInfo
- Publication number
- WO2022011531A1 WO2022011531A1 PCT/CN2020/101789 CN2020101789W WO2022011531A1 WO 2022011531 A1 WO2022011531 A1 WO 2022011531A1 CN 2020101789 W CN2020101789 W CN 2020101789W WO 2022011531 A1 WO2022011531 A1 WO 2022011531A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- seq
- amino acid
- acid sequence
- cld18a2
- domain
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K40/00—Cellular immunotherapy
- A61K40/10—Cellular immunotherapy characterised by the cell type used
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K40/00—Cellular immunotherapy
- A61K40/10—Cellular immunotherapy characterised by the cell type used
- A61K40/11—T-cells, e.g. tumour infiltrating lymphocytes [TIL] or regulatory T [Treg] cells; Lymphokine-activated killer [LAK] cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K40/00—Cellular immunotherapy
- A61K40/30—Cellular immunotherapy characterised by the recombinant expression of specific molecules in the cells of the immune system
- A61K40/31—Chimeric antigen receptors [CAR]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K40/00—Cellular immunotherapy
- A61K40/40—Cellular immunotherapy characterised by antigens that are targeted or presented by cells of the immune system
- A61K40/41—Vertebrate antigens
- A61K40/42—Cancer antigens
- A61K40/4202—Receptors, cell surface antigens or cell surface determinants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/62—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being a protein, peptide or polyamino acid
- A61K47/64—Drug-peptide, drug-protein or drug-polyamino acid conjugates, i.e. the modifying agent being a peptide, protein or polyamino acid which is covalently bonded or complexed to a therapeutically active agent
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/30—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants from tumour cells
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/46—Hybrid immunoglobulins
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K19/00—Hybrid peptides, i.e. peptides covalently bound to nucleic acids, or non-covalently bound protein-protein complexes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/62—DNA sequences coding for fusion proteins
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/10—Cells modified by introduction of foreign genetic material
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2239/00—Indexing codes associated with cellular immunotherapy of group A61K40/00
- A61K2239/31—Indexing codes associated with cellular immunotherapy of group A61K40/00 characterized by the route of administration
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2239/00—Indexing codes associated with cellular immunotherapy of group A61K40/00
- A61K2239/38—Indexing codes associated with cellular immunotherapy of group A61K40/00 characterised by the dose, timing or administration schedule
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2239/00—Indexing codes associated with cellular immunotherapy of group A61K40/00
- A61K2239/46—Indexing codes associated with cellular immunotherapy of group A61K40/00 characterised by the cancer treated
- A61K2239/51—Stomach
Definitions
- the present invention relates to the field of biotechnology, in particular to an anti-CLD18A2 single-domain antibody.
- Claudin 18 (Claudin 18, CLD18) is a transmembrane protein with a molecular weight of about 28 kD, located in the tight junctions of the epithelium and the endothelium, which are tightly junctions between adjacent cells.
- the claudin on the cell surface is difficult to be contacted due to the tight intercellular space, while the intercellular space of tumor cells is relatively loose. Therefore, claudin on tumor cells becomes a potential target for extracellular antibodies and immunotherapy.
- CLD18 has four hydrophobic regions that act as transmembrane domains to form two ectodomains, wherein hydrophobic region 1 and hydrophobic region 2 surround to form ectodomain 1, and hydrophobic region 3 and hydrophobic region 4 surround to form ectodomain 2. Due to different splicing of genes, CLD18 forms two spliceosomes: CLD18A1 (Claudin18.1) and CLD18A2 (Claudin18.2).
- CLD18A1 is selectively expressed in the epithelium of normal lung and stomach, whereas CLD18A2 is expressed only in gastric cells; more importantly, CLD18A2 is localized in differentiated gastric epithelium short-lived cells but absent in gastric stem cells (Niimi T. , et al. Biol. 2001;21(21):7380–7390.). These properties suggest that CLD18A2 is a clinically valuable therapeutic target for the treatment of gastric cancer and other CLD18A2-positive tumors.
- the purpose of the present invention is to provide an anti-CLD18A2 single-domain antibody for solving the problems in the prior art.
- one aspect of the present invention provides an anti-CLD18A2 single-domain antibody
- the complementarity determining region of the anti-CLD18A2 single-domain antibody comprises an amino acid sequence as shown in one of SEQ ID No. 4-11.
- bispecific antibody comprises the above-mentioned anti-CLD18A2 single domain antibody and an anti-CD3 domain.
- Another aspect of the present invention provides an isolated polynucleotide encoding the above-mentioned anti-CLD18A2 single-domain antibody, or the above-mentioned bispecific antibody.
- Another aspect of the present invention provides a construct comprising the isolated polynucleotide described above.
- Another aspect of the present invention provides an antibody expression system, the expression system comprising the above-mentioned construct or the above-mentioned exogenous polynucleotide integrated into the genome.
- Another aspect of the present invention provides a method for preparing the above-mentioned anti-CLD18A2 single-domain antibody or the above-mentioned bispecific antibody, comprising the steps of: culturing the above-mentioned antibody expression system under conditions suitable for expressing the antibody, thereby expressing The antibody is obtained, and the antibody is purified and isolated.
- Another aspect of the present invention provides the use of the above-mentioned anti-CLD18A2 single-domain antibody or the above-mentioned bispecific antibody in the preparation of a medicament for treating a tumor.
- Another aspect of the present invention provides an antibody-drug complex comprising the above-mentioned anti-CLD18A2 single domain antibody and a cytotoxic drug.
- Another aspect of the present invention provides a cell comprising a membrane-bound chimeric antigen receptor, the chimeric antigen receptor comprising a transmembrane domain, an intracellular domain and an extracellular domain, the extracellular domain comprising the above-mentioned anti- Single domain antibody to CLD18A2.
- FIG. 1 is a schematic diagram showing the results of the activation test of the T cell fluorescent reporter system in Example 3 of the present invention.
- FIG. 2 is a schematic diagram showing the killing effect of anti-CLD18A2/anti-CD3 bispecific antibody on NUGC-4-Claudin18.2 in vitro in Example 3 of the present invention.
- Figure 3 shows a schematic diagram of the in vivo tumor inhibition test of the anti-CLD18A2/anti-CD3 bispecific antibody in Example 3 of the present invention.
- FIG. 4 is a schematic diagram showing the results of endocytosis mediated by anti-CLDN18A2 single-domain antibody fusion protein in Example 5 of the present invention.
- FIG. 5 is a schematic diagram showing the results of the in vitro cytotoxicity test in Example 8 of the present invention.
- FIG. 6 is a schematic diagram showing the results of the in vivo toxicity test in Example 9 of the present invention.
- Fig. 7 is a schematic diagram showing the results of in vitro cytokine release content detection after aC18.2-CAR-T cells act on target cells in Example 11 of the present invention.
- Figure 8 is a schematic diagram showing the inhibitory effect of aC18.2-CAR-T cells on tumor growth in mice in Example 11 of the present invention.
- the anti-CLD18A2 single-domain antibody has good affinity for Claudin18.2, and can be used to further construct bispecific antibodies, Antibody-drug complexes, T cell chimeric antigen receptors, etc., can be used to prepare drugs with good targeting and therapeutic effects.
- the first aspect of the present invention provides an anti-CLD18A2 single-domain antibody.
- the single-domain antibody generally refers to a type of antibody molecule that lacks the light chain of the antibody and only has the variable region of the heavy chain. Because of its small molecular weight, it is also commonly referred to as for nanobodies.
- the complementarity determining region (CDR, complementarity determining region) of the single-domain antibody of described anti-CLD18A2 includes amino acid sequence such as CDR-H1 shown in one of SEQ ID No.4 ⁇ 11, amino acid sequence such as SEQ ID No.19 ⁇ 26 CDR-H2 shown in one of them, and CDR-H3 whose amino acid sequence is shown in one of SEQ ID Nos. 33 to 39.
- the complementarity determining region of the anti-CLD18A2 single domain antibody comprises CDR-H1 whose amino acid sequence is shown in SEQ ID No. 4, and CDR-H1 whose amino acid sequence is shown in SEQ ID No. 19 H2, and CDR-H3 whose amino acid sequence is shown in SEQ ID No.33.
- the complementarity determining region of the anti-CLD18A2 single domain antibody includes CDR-H1 whose amino acid sequence is shown in SEQ ID No.5, and CDR-H1 whose amino acid sequence is shown in SEQ ID No.20 -H2, and CDR-H3 whose amino acid sequence is shown in SEQ ID No.34.
- the complementarity determining region of the anti-CLD18A2 single-domain antibody includes CDR-H1 whose amino acid sequence is shown in SEQ ID No. 6, and CDR-H1 whose amino acid sequence is shown in SEQ ID No. 21 -H2, and CDR-H3 whose amino acid sequence is shown in SEQ ID No.35.
- the complementarity determining region of the anti-CLD18A2 single-domain antibody comprises CDR-H1 whose amino acid sequence is shown in SEQ ID No.7, and CDR-H1 whose amino acid sequence is shown in SEQ ID No.22 -H2, and CDR-H3 whose amino acid sequence is shown in SEQ ID No.36.
- the complementarity determining region of the anti-CLD18A2 single-domain antibody includes CDR-H1 whose amino acid sequence is shown in SEQ ID No. 8, and CDR-H1 whose amino acid sequence is shown in SEQ ID No. 23 -H2, and CDR-H3 whose amino acid sequence is shown in SEQ ID No.37.
- the complementarity determining region of the anti-CLD18A2 single-domain antibody includes CDR-H1 whose amino acid sequence is shown in SEQ ID No. 9, and CDR-H1 whose amino acid sequence is shown in SEQ ID No. 24 -H2, and CDR-H3 whose amino acid sequence is shown in SEQ ID No.38.
- the complementarity determining region of the anti-CLD18A2 single domain antibody comprises CDR-H1 whose amino acid sequence is shown in SEQ ID No.10, and CDR-H1 whose amino acid sequence is shown in SEQ ID No.25 -H2, and CDR-H3 whose amino acid sequence is shown in SEQ ID No.36.
- the complementarity determining region of the anti-CLD18A2 single-domain antibody comprises CDR-H1 whose amino acid sequence is shown in SEQ ID No.11, and CDR-H1 whose amino acid sequence is shown in SEQ ID No.26 -H2, and CDR-H3 whose amino acid sequence is shown in SEQ ID No.39.
- the anti-CLD18A2 single-domain antibody further includes a framework region (FR, framework region), and the framework region FR includes an amino acid sequence such as one of SEQ ID No. 1-3 FR1 shown in one, FR2 whose amino acid sequence is shown in one of SEQ ID No. 12 to 18, FR3 whose amino acid sequence is shown in one of SEQ ID No. 27 to 32, and FR3 whose amino acid sequence is shown in SEQ ID No. 27 to 32.
- the framework region FR includes FR1 whose amino acid sequence is shown in SEQ ID No.1, FR2 whose amino acid sequence is shown in SEQ ID No.12, and whose amino acid sequence is shown in SEQ ID No.27 FR3 shown, and FR4 whose amino acid sequence is shown in SEQ ID No. 40.
- the framework region FR includes FR1 whose amino acid sequence is shown in SEQ ID No. 2, FR2 whose amino acid sequence is shown in SEQ ID No. 13, and whose amino acid sequence is shown in SEQ ID No. 28 The FR3 shown, and the amino acid sequence of FR4 shown in SEQ ID No. 41.
- the framework region FR includes FR1 whose amino acid sequence is shown in SEQ ID No.3, FR2 whose amino acid sequence is shown in SEQ ID No.14, and FR2 whose amino acid sequence is shown in SEQ ID No.29 The FR3 shown, and the amino acid sequence of FR4 shown in SEQ ID No. 41.
- the framework region FR includes FR1 whose amino acid sequence is shown in SEQ ID No. 1, FR2 whose amino acid sequence is shown in SEQ ID No. 15, and FR2 whose amino acid sequence is shown in SEQ ID No. 30 The FR3 shown, and the amino acid sequence of FR4 shown in SEQ ID No. 41.
- the framework region FR includes FR1 whose amino acid sequence is shown in SEQ ID No.2, FR2 whose amino acid sequence is shown in SEQ ID No.16, and whose amino acid sequence is shown in SEQ ID No.31 The FR3 shown, and the amino acid sequence of FR4 shown in SEQ ID No. 41.
- the FR in the framework region includes FR1 whose amino acid sequence is shown in SEQ ID No.2, FR2 whose amino acid sequence is shown in SEQ ID No.13, and whose amino acid sequence is shown in SEQ ID No.31 The FR3 shown, and the amino acid sequence of FR4 shown in SEQ ID No. 41.
- the framework region FR includes FR1 whose amino acid sequence is shown in SEQ ID No.1, FR2 whose amino acid sequence is shown in SEQ ID No.17, and FR2 whose amino acid sequence is shown in SEQ ID No.30 The FR3 shown, and the amino acid sequence of FR4 shown in SEQ ID No. 41.
- the framework region FR includes FR1 whose amino acid sequence is shown in SEQ ID No. 2, FR2 whose amino acid sequence is shown in SEQ ID No. 18, and FR2 whose amino acid sequence is shown in SEQ ID No. 32
- the shown FR3, and amino acid sequences are FR4 shown in SEQ ID No. 41.
- the amino acid sequence of the anti-CLD18A2 single-domain antibody may include: a) the amino acid sequence shown in one of SEQ ID Nos. 42 to 49; or, b) An amino acid sequence that has more than 80% sequence identity with the amino acid sequence shown in one of SEQ ID Nos. 42 to 49, and has the function of the amino acid sequence defined in a).
- the amino acid sequence in b) specifically refers to: the amino acid sequence shown in one of SEQ ID No.
- 42 to 49 is substituted, deleted or added one or more (specifically, 1-50, 1- 30, 1-20, 1-10, 1-5, or 1-3) amino acids, or by adding one or more (specifically, 1) to the N-terminal and/or C-terminal -50, 1-30, 1-20, 1-10, 1-5, or 1-3) amino acids, and have amino acids as one of SEQ ID No. 42-49
- Polypeptide fragments with the functions of the indicated polypeptide fragments, for example, may have specific binding ability to CLD18A2, so that they can specifically bind to cells expressing CLD18A2, but not to cells expressing only CLD18A1, that is, only recognize CLD18A2 and not Identify CLD18A1.
- the amino acid sequence in b) can be more than 80%, 85%, 90%, 93%, 95%, 97%, or 99% identical to one of SEQ ID Nos. 42-49.
- sequence identity refers to the percentage of identical residues in the sequences participating in the alignment. Sequence identity of two or more entry sequences can be calculated using computational software well known in the art, such software available from NCBI, for example.
- the anti-CLD18A2 single-domain antibody provided by the present invention can be derived from alpaca (Vicugna pacos), and its overall molecular weight can be about half that of ScFv single-chain antibody, so it can effectively reduce the molecular weight of the overall structure, thereby enhancing its organization. Penetration, reaching target tissues and organs more effectively, improving the therapeutic effect, and this structure is more convenient to prepare than the structure with two ScFv in series.
- the anti-CLD18A2 single domain antibody provided by the present invention can usually be a humanized antibody.
- the amino acid sequence of the anti-CLD18A2 single-domain antibody can include: c) the amino acid sequence shown in one of SEQ ID No. 67-90; or, d) and the amino acid sequence shown in one of SEQ ID No. 67-90
- the amino acid sequence has more than 80% sequence identity and has the amino acid sequence function defined by c).
- the amino acid sequence in d) specifically refers to: the amino acid sequence shown in one of SEQ ID No.
- 67-90 is substituted, deleted or added with one or more (specifically, 1-50, 1- 30, 1-20, 1-10, 1-5, or 1-3) amino acids, or by adding one or more (specifically, 1) to the N-terminal and/or C-terminal -50, 1-30, 1-20, 1-10, 1-5, or 1-3) amino acids, and have amino acids as one of SEQ ID No. 67-90
- Polypeptide fragments that exhibit the function of the polypeptide fragments shown, for example, may be of a relatively higher degree of humanization.
- the humanization of the anti-CLD18.2 single-domain antibody of the present invention is mainly aimed at transforming the framework region, for example, by adopting the method of CDR transplantation, the FR region of a suitable human-derived antibody is replaced with the original sequence, while the CDR region of the original antibody is retained.
- the sequences SEQ ID No. 75-90 are obtained by humanizing FR1, FR2, FR3, and FR4 based on the original sequences SEQ ID No. 42-49; the sequences SEQ ID No. 67-74 are obtained from the original sequences Based on SEQ ID Nos. 42 to 49, the corresponding FR1, FR3, and FR4 are obtained by humanization transformation, while FR2 retains the corresponding original sequence.
- the amino acid sequence in d) can be more than 80%, 85%, 90%, 93%, 95%, 97%, or 99% identical to one of SEQ ID Nos. 67-90.
- the second aspect of the present invention provides a bispecific antibody, and the bispecific antibody includes the anti-CLD18A2 single domain antibody and the anti-CD3 domain provided in the first aspect of the present invention.
- the bispecific antibody can simultaneously target the cell surface antigen CLD18A2 and the T cell surface triggering molecule CD3, thereby activating the T cells, resulting in the effect of killing the target cells.
- an anti-CD3 domain may be included.
- the structural form of the anti-CD3 domain can include, but is not limited to, single-chain antibody (scFv), antibody Fab fragments, and the like.
- the anti-CD3 domain can specifically bind to the triggering molecule CD3 on the surface of T cells, so that T cells can be activated to enhance the effect of killing target cells.
- the amino acid sequence of the heavy chain variable region of the anti-CD3 domain may include: e) as SEQ ID NO.91, SEQ ID NO.93, SEQ ID NO.95, SEQ ID NO.97, SEQ ID NO.99 , or the amino acid sequence shown in one of SEQ ID NO.101; or, f) with SEQ ID NO.91, SEQ ID NO.93, SEQ ID NO.95, SEQ ID NO.97, SEQ ID NO.99 , or the amino acid sequence shown in one of SEQ ID NO.101 has more than 80% sequence identity, and has the amino acid sequence function of the amino acid sequence defined in e).
- amino acid sequence in the f) specifically refers to: such as SEQ ID NO.91, SEQ ID NO.93, SEQ ID NO.95, SEQ ID NO.97, SEQ ID NO.99, or SEQ ID NO.99.
- the amino acid sequence shown in one of 101 has undergone substitution, deletion or addition of one or more (specifically, 1-50, 1-30, 1-20, 1-10, 1-5, or 1- 3) amino acids, or adding one or more (specifically 1-50, 1-30, 1-20, 1-10, 1- 5, or 1-3) amino acids, and have amino acids such as SEQ ID NO.91, SEQ ID NO.93, SEQ ID NO.95, SEQ ID NO.97, SEQ ID NO.99, or SEQ ID NO.99 A functional polypeptide fragment of one of the polypeptide fragments shown in ID NO.
- the amino acid sequence in said f) can have one of SEQ ID NO.91, SEQ ID NO.93, SEQ ID NO.95, SEQ ID NO.97, SEQ ID NO.99, or SEQ ID NO.101 Concordance above 80%, 85%, 90%, 93%, 95%, 97%, or 99%.
- the amino acid sequence of the light chain variable region of the anti-CD3 domain may include: g) as SEQ ID NO.92, SEQ ID NO.94, SEQ ID NO.96, SEQ ID NO.98, SEQ ID NO.100 and, or the amino acid sequence shown in one of SEQ ID NO.102; or, h) with SEQ ID NO.92, SEQ ID NO.94, SEQ ID NO.96, SEQ ID NO.98, SEQ ID NO. 100, or the amino acid sequence of one of the amino acid sequences shown in SEQ ID NO. 102 with more than 80% sequence identity and having the amino acid sequence function defined in g).
- amino acid sequence in the h specifically refers to: such as SEQ ID NO.92, SEQ ID NO.94, SEQ ID NO.96, SEQ ID NO.98, SEQ ID NO.100, or SEQ ID NO.
- the amino acid sequence shown in one of 102 has been substituted, deleted or added one or more (specifically can be 1-50, 1-30, 1-20, 1-10, 1-5, or 1- 3) amino acids, or adding one or more (specifically 1-50, 1-30, 1-20, 1-10, 1- 5, or 1-3) amino acids, and have amino acids such as SEQ ID NO.92, SEQ ID NO.94, SEQ ID NO.96, SEQ ID NO.98, SEQ ID NO.100, or SEQ ID NO.
- the amino acid sequence in h) can have one of SEQ ID NO.92, SEQ ID NO.94, SEQ ID NO.96, SEQ ID NO.98, SEQ ID NO.100, or SEQ ID NO.102 Concordance above 80%, 85%, 90%, 93%, 95%, 97%, or 99%.
- the heavy chain variable region sequence of the anti-CD3 structural domain can be SEQ ID NO.95
- the light chain variable region sequence of the anti-CD3 structural domain can be SEQ ID NO.96.
- the sequence of the ScFv form is shown in SEQ ID NO.103, or SEQ ID NO.104.
- sequence of the heavy chain variable region of the anti-CD3 domain can be SEQ ID NO.93
- sequence of the light chain variable region of the anti-CD3 domain can be SEQ ID NO.94.
- sequence of the ScFv form is shown in SEQ ID NO.105, or SEQ ID NO.106.
- the CD3 may be of human origin, non-human primate (eg, monkey) origin, and/or murine origin.
- the bispecific antibodies may also include a domain for prolonging serum half-life.
- the domain for prolonging serum half-life can be selected from the Fc domain of mammalian IgG, albumin, albumin binding domain (ABD) or polyethylene glycol (PEG) and the like.
- the domain for prolonging serum half-life can specifically include: i) a combination of amino acid sequences shown in SEQ ID No. 107, SEQ ID No. 108, or SEQ ID No. 156; or, j) and SEQ ID No. 107.
- the amino acid sequence shown in one of SEQ ID No. 108, or SEQ ID No. 156 has more than 80% sequence identity and has the amino acid sequence function as defined in i).
- the amino acid sequence in j) specifically refers to: the amino acid sequence shown in one of SEQ ID No. 107, SEQ ID No. 108, or SEQ ID No. 156 is substituted, deleted, or added with one or more (specifically can be 1-50, 1-30, 1-20, 1-10, 1-5, or 1-3) amino acids, or at the N-terminal and/or C- It is obtained by adding one or more (specifically, 1-50, 1-30, 1-20, 1-10, 1-5, or 1-3) amino acids to the end, and has amino acids such as The functional polypeptide fragment of one of SEQ ID No. 107, SEQ ID No. 108, or SEQ ID No.
- the amino acid sequence in said j) can have 80%, 85%, 90%, 93%, 95%, 97%, 80%, 85%, 90%, 93%, 95%, 97%, SEQ ID No. or more than 99% consistency.
- the anti-CLD18A2 single-domain antibody, the anti-CD3 domain and the domain for prolonging serum half-life further include a linker peptide.
- a plurality of linker peptide fragments can be included, for example, an anti-CLD18A2 single-domain antibody, a linker peptide can be included between the anti-CD3 domains, for example, an anti-CD3 domain, a domain for prolonging serum half-life Linking peptides may be included in between.
- the connecting peptide fragment can usually be a flexible polypeptide of suitable length composed of glycine (G) and/or serine (S) and/or alanine (A) and/or threonine (T), which can maintain the bispecific antibody
- the amino acid sequence of the linker peptide fragment may include, for example, (GS)n, (GGS)n, (GGSG)n, (GGGS)nA, (GGGGS) Sequences of nA, (GGGGA)nA, (GGGGG)nA, etc., wherein n is selected from an integer between 1-10.
- the amino acid sequence of the connecting peptide may include: GGGGSGGGS (SEQ ID NO. 157), GGGGGGSGGSGGSGGSGG (SEQ ID NO. 158), GGGGSGGGGSGGGGS (SEQ ID NO. 159) and the like.
- the bispecific antibody provided by the present invention can be one of the following:
- the bispecific antibody can include, from N-terminal to C-terminal, an anti-CLD18A2 single domain antibody, a linking peptide, an anti-CD3 single-chain antibody (ie , the structure of VHH/CLD18A2 -linking peptide-scFv CD3 ), and, for example, anti-CD3
- the single-chain antibody may sequentially include its heavy chain variable region fragment, connecting peptide, light chain variable region fragment (ie , the structure of VH -connecting peptide- VL ), or sequentially include its light chain.
- variable region fragment, connecting peptide, a heavy chain variable region fragment i.e., V L - linker peptide structure -V H).
- a bispecific antibody can include, from N-terminal to C-terminal, anti-CLD18A2 single domain antibody, linker peptide, ABD, linker peptide, anti-CD3 single chain antibody (ie VHH/CLD18A2 -linker peptide-ABD-linker peptide-scFv
- the structure of CD3 for another example, the anti-CD3 single-chain antibody can sequentially include its heavy chain variable region fragment, connecting peptide, light chain variable region fragment (ie, VH -connecting peptide- VL ) from the N-terminus to the C-terminus. structure), or sequentially including its light chain variable region fragment, connecting peptide, and heavy chain variable region fragment (ie, the structure of VL -connecting peptide- VH).
- a bispecific antibody may include, in order from N-terminus to C-terminus, an anti-CLD18A2 single domain antibody, a linker peptide, an anti-CD3 single chain antibody, a linker peptide, an ABD (ie, VHH/CLD18A2 -linker peptide-scFv CD3 -linker peptide- The structure of ABD), for another example, the anti-CD3 single-chain antibody can sequentially include its heavy chain variable region fragment, connecting peptide, and light chain variable region fragment from the N-terminus to the C-terminus (ie, the VH -connecting peptide- VL) . structure), or sequentially including its light chain variable region fragment, connecting peptide, and heavy chain variable region fragment (ie, the structure of VL -connecting peptide- VH).
- S4 structure a heterodimer structure composed of anti-CLD18A2 single-domain antibody (V HH/CLD18A2 ), anti-CD3 domain and IgG Fc fragment.
- the amino acid sequence of one of the IgG1 Fc variants in the S4 structure of the bispecific antibody may include SEQ ID NO. 107, and the amino acid sequence of the other may include SEQ ID NO. 108.
- the CH3 domains of the two variants are modified to form asymmetrical pestle-hole structure and form a stable heterodimeric structure.
- Fc has been mutated, such as L234A and L235A mutations, the purpose is to reduce a biological effect of Fc, that is, to reduce the affinity of Fc with Fc ⁇ RIII on NK cells, so as to avoid possible NK cells. kill.
- the amino acid sequence of the bispecific antibody may include the sequence shown in one of SEQ ID NO. 109-118, wherein SEQ ID NO. Single domain antibody ABD (SEQ ID NO.156) structure to prolong drug half-life, while SEQ ID NO.115 and SEQ ID NO.116 form a heterodimer to prolong half-life through Fc, SEQ ID NO.117 and SEQ ID NO. 118 forms heterodimers, extending half-life by Fc.
- SEQ ID NO. 109-118 Single domain antibody ABD (SEQ ID NO.156) structure to prolong drug half-life
- SEQ ID NO.115 and SEQ ID NO.116 form a heterodimer to prolong half-life through Fc
- SEQ ID NO.117 and SEQ ID NO. 118 forms heterodimers, extending half-life by Fc.
- the third aspect of the present invention provides an isolated polynucleotide encoding the anti-CLD18A2 single domain antibody provided by the first aspect of the present invention, or the bispecific antibody provided by the second aspect of the present invention.
- the fourth aspect of the present invention provides a construct comprising the isolated polynucleotide provided by the third aspect of the present invention.
- the construct contains the isolated polynucleotide provided by the second aspect of the present invention.
- the construct can generally be constructed by inserting the isolated polynucleotide into a suitable vector, and those skilled in the art can select a suitable expression vector.
- the type of vector may include, but is not limited to, plasmids, phagemids, phage derivatives, animal viruses, cosmids, and the like.
- the vector may be an expression vector or a cloning vector.
- the fifth aspect of the present invention provides an antibody expression system
- the expression system contains the construct provided by the fourth aspect of the present invention or the exogenous polynucleotide provided by the third aspect of the present invention is integrated into the genome, so that it can be Express the single-domain antibody or bispecific antibody of the anti-CLD18A2.
- the expression system can be a host cell, which can be a prokaryotic cell, such as a bacterial cell; or a lower eukaryotic cell, such as a yeast cell; a filamentous fungal cell, or a higher eukaryotic cell, such as a mammalian cell .
- Escherichia coli, Streptomyces bacterial cells of Salmonella typhimurium
- fungal cells such as yeast, filamentous fungi, plant cells
- insect cells of Drosophila S2 or Sf9 CHO, COS, 293 cells, or Bowes black melanoma cells of animal cells, etc.
- Methods for introducing constructs into host cells should be known to those skilled in the art, for example, microinjection, biolistic, electroporation, virus-mediated transformation, electron bombardment, calcium phosphate precipitation can be used method, etc.
- the plasmid may be pPIC9K.
- the expression system uses Pichia pastoris cells as a host.
- Yeast has unique advantages in expressing exogenous proteins.
- yeast has a complete eukaryotic expression system, which can realize the correct folding and post-translational modification of antibody structure; yeast can also realize high-density fermentation to realize exogenous protein. high expression and obtain high unit yield.
- the plasmid contains a DHFR (dihydrofolate reductase) gene or a GS (glutamine synthase) gene, which is both a selection marker and a gene co-amplification.
- DHFR dihydrofolate reductase
- GS glucose synthase
- the selected mammalian host cells can be NSO, HEK293, PERC6 or CHO, and CHO cells are preferably used as the expression host of the bispecific antibody.
- CHO cells have a complete set of cellular machinery for synthesizing, assembling and secreting proteins. Therefore, the produced antibody molecules can maintain the correct protein conformation and post-translational glycosylation processing, become functional antibody molecules, and be secreted outside the cell, which is convenient for Isolation and Purification.
- CHO cells also have the characteristics of secreting and producing less self-proteins and proteases.
- CHO cells can be grown in suspension after serum-free acclimation, which can achieve high-density culture. The target protein yield can reach a high yield of 2-10g/L. Industrial application, and therefore, most of the antibody drugs on the market in the past 30 years have used the CHO cell expression system.
- the sixth aspect of the present invention provides a method for preparing the anti-CLD18A2 single domain antibody provided in the first aspect of the present invention, or the bispecific antibody provided in the second aspect of the present invention, comprising the following steps: under conditions suitable for expressing the antibody Next, the antibody expression system provided by the fifth aspect of the present invention is cultured to express the anti-CLD18A2 single domain antibody or bispecific antibody, and the antibody is purified and isolated.
- the seventh aspect of the present invention provides an antibody-drug complex, the drug complex comprising the anti-CLD18A2 single domain antibody and the cytotoxic drug provided in the first aspect of the present invention.
- the structure of the antibody-drug complex provided by the present invention can be shown in formula 1:
- VHH/CLD18A2 is a single domain antibody against CLD18A2;
- Z is an accessory functional region, and Z is selected from a domain and/or a drug-conjugating domain for prolonging serum half-life, or is absent;
- D is a cytotoxic drug molecule
- n represents the average number of D coupled, and 0 ⁇ n ⁇ 10, preferably 2 ⁇ n ⁇ 7; more preferably 3 ⁇ n ⁇ 6; most preferably 4.
- the VHH/CLD18A2-connecting peptide-Z part can be a fusion protein, and the C-terminus of the anti-CLD18A2 single-domain antibody (VHH/CLD18A2 ) is connected with the connecting peptide through the connecting peptide.
- Accessory functional domains are linked, or linked to linker chains, and cytotoxic drug molecules.
- Z when Z is not present, it can include anti-CLD18A2 single domain antibody and linker peptide in order from N-terminal to C-terminal, and for example, when Z is present, it can include anti-CLD18A2 single-domain antibody from N-terminal to C-terminal in order , linker peptides, domains for extending serum half-life and/or drug-conjugation domains.
- VHH/CLD18A2 is a single-domain antibody against CLD18A2, and the VHH/CLD18A2 can be monovalent, that is, it includes an antigen-binding site, and the VHH/CLD18A2 can also be To be multivalent, that is, it may include two or more antigen-binding sites of the same or different sequences, and these antigen-binding sites may be in a tandem structure.
- an accessory functional region may be included.
- the accessory functional region may include, but is not limited to, an immunoglobulin Fc region, a serum albumin fragment, a polyethylene glycol fragment (PEG), a serum albumin binding domain (ABD), a polypeptide chain, an antibody or a gelatin-like unit A combination of one or more of the above, as well as derivatives of the above structures, including mutants and fusion proteins, and the like.
- Z is ABD-(GGC) n , wherein n is an integer greater than or equal to 1, preferably n is 4, 5, 6, 7, 8, 9, or 10.
- ABD is a single-domain antibody that binds human serum albumin, and (GGC) n provides a drug-conjugated cysteine group.
- Z is (PAEC)n, wherein, n is an integer greater than or equal to 1, preferably n is 4, 5, 6, 7, 8, 9, 10, PAEC is composed of proline (P) , alanine (A) and glutamic acid (E) and cysteine (C). More specifically, the amino acid sequence of Z can include the sequence shown in SEQ ID NO.124. This sequence can significantly prolong the half-life in vivo and increase the conjugation site of the drug.
- Z is an immunoglobulin Fc region.
- the human immunoglobulin Fc region includes a mutation for altering Fc-mediated effector function, the effector function including one of CDC activity, ADCC activity, ADCP activity or various combinations.
- the immunoglobulin is selected from a combination of one or more of IgG, IgA1, IgA2, IgD, IgE, and IgM, and the IgG is selected from IgG1, IgG2, IgG3 or IgG4 A combination of one or more of the subtypes.
- the amino acid sequence of the immunoglobulin Fc region may include the sequence shown in one of SEQ ID NO. 119-123.
- the accessory functional region is selected from the Fc portion of the human immunoglobulin IgG1 constant region, and the sequence comprises the hinge region and CH2 and CH3, such that the antibody-drug complex forms the dimerization of formula 1 body structure, and maintain the CDC and ADCC roles of IgG1 constant region Fc.
- the accessory functional region is selected from a single domain antibody that specifically binds to human serum albumin.
- the connecting peptide fragment can usually be a segment of suitable length composed of glycine (G) and/or serine (S) and/or alanine (A) and/or threonine ( T) is a flexible polypeptide that can maintain the correct folding of each domain of the bispecific antibody molecule and the flexibility of each other.
- the amino acid sequence of the connecting peptide fragment can include such as (GS)n, (GGS)n, ( GGSG)n, (GGGS)nA, (GGGGS)nA, (GGGGA)nA, (GGGGG)nA and other sequences, wherein n is selected from an integer between 1-10.
- the amino acid sequence of the linking peptide may include: GGGGSGGGS (SEQ ID NO. 160).
- a linker chain may be included.
- a linker can usually be used to connect the cytotoxic small molecule drug and the anti-CLD18A2 single domain antibody fusion protein, and a linker that can bind the reactive functional group of the drug molecule and the amino acid side chain can usually be used.
- Linking chains can generally be divided into two categories: degradable linking chains and non-degradable linking chains.
- the degradable linking chain may include: hydrazones, cis-aconiyl, hydrazide bonds, disulfide bonds, peptide bonds, ⁇ -glucuronic acid-based bonds, etc.;
- Linking chains of the non-degradable type may include succinimide-thioether linkages and the like.
- SPP 2-pyridyldithio)valerate
- SIAB N-succinimidyl(4-iodoacetyl)aminobenzoate
- SMCC 6-maleimidohexanoyl
- MC 6-maleimidohexanoyl
- B is a covalent binding unit covalently bound to the amino acid on the anti-CLD18A2 single-domain antibody fusion protein, and b is 0 or 1;
- S and S ⁇ are interval units, and s and s ⁇ are each independently an integer from 0 to 3;
- A is an amino acid unit; a is an integer of 0-12.
- a covalent binding unit may be included.
- a group with electrophilic properties on the linking chain can react with nucleophilic cysteine or selenocysteine on the anti-CLD18A2 single domain antibody fusion protein to form a co- price key.
- Groups of electrophilic nature include, but are not limited to, maleimide and haloacetamide groups.
- the group with electrophilic properties on the intermediate of the drug-linking chain can also react with the nucleophilic cysteine or selenocysteine on the anti-CLD18A2 single domain antibody fusion protein to form a covalent bond.
- the linker chain can be a branched structure for covalently binding more than one drug moiety to the hydrophilic polypeptide bound to the targeting ligand. Branched linker chains can increase the molar ratio of drug to antibody, ie, loading.
- the linking chain may have a reactive functional group that has the ability to react with an electrophilic group present on the anti-CLD18A2 single-domain antibody fusion protein Nucleophilic group.
- Useful electrophilic groups on anti-CLD18A2 single domain antibody fusion proteins include, but are not limited to, aldehyde and ketone carbonyl groups.
- the heteroatoms of the nucleophilic group linking the chain can react with the electrophilic group on the anti-CLD18A2 single domain antibody fusion protein and form a covalent bond.
- nucleophilic groups on the linking chain include, but are not limited to, hydrazides, oximes, amino groups, hydrazines, thiosemicarbazones, hydrazine carboxylates, and aryl hydrazides.
- the electrophilic group on the anti-CLD18A2 single domain antibody fusion protein provides a convenient location for binding (attaching) the linker chain.
- the bis-maleimide reagent enables the sulfhydryl group on the Nanobody derivative to bind to the sulfhydryl group-containing drug moiety or linker intermediate in a sequential or simultaneous manner.
- amino acid units may be included, and the amino acid units usually include amino acid residues.
- A may be a dipeptide, tripeptide, tetrapeptide, pentapeptide, hexapeptide, heptapeptide, octapeptide, nonapeptide, decapeptide, undecapeptide or dodecapeptide unit.
- Amino acid residues comprising amino acid units include those naturally occurring as well as minimal amino acids and non-naturally occurring amino acid analogs, such as citrulline.
- the amino acid unit can be enzymatically cleaved with one or more enzymes, including tumor-associated proteases, to release the drug moiety (-D).
- a spacer unit (S or S') may be included.
- the spacer unit may typically link the amino acid unit (-A-) to the drug moiety (D) in the presence of the spacer unit; alternatively, the spacer unit may link the Bb unit of formula II to the drug moiety in the absence of the amino acid unit.
- the spacer unit also links the drug moiety to the targeting ligand derivative unit when neither the amino acid unit nor the B b unit is present.
- Spacer units are generally of two types: self-immolative and non-self-eliminating.
- a non-self-eliminating spacer unit is one in which some or all of the spacer unit remains bound to the drug moiety after cleavage from the ligand derivative-drug complex, especially after enzymatic cleavage of the amino acid unit.
- the antibody-drug complexes provided by the present invention may include cytotoxic drug molecules.
- the cytotoxic molecule is generally any compound or group that has a cytotoxic or cytostatic effect. These cytotoxic molecules can include: (i) chemotherapeutic agents that can act as tubulin inhibitors, mitotic inhibitors, topoisomerase inhibitors, or DNA intercalators; (ii) protein toxins that can act enzymatically ; and (iii) radioisotopes.
- Typical cytotoxic molecules may include, but are not limited to, maytansinoids, auristatin, dolastatin, trichothecene, CC1065, calicheamicin ( Calicheamicin and other enediyne antibiotics, taxanes, anthracyclines and their stereoisomers, isosteres, analogs or derivatives or combinations thereof.
- the monomethyl auristatin can be monomethyl auristatin E (MMAE) or monomethyl auristatin F (MMAF)
- the maytansinoids can be N2'-deacetyl -N2'-(3-mercapto-1-oxopropyl)-maytansine (DM1), N2'-deacetyl-N2'-(4-mercapto-1-oxopentyl)-maytansine (DM3) and N2'-deacetyl-N2'-(4-mercapto-4-methyl-1-oxopentyl)-maytansine (DM4).
- MMAE monomethyl auristatin E
- MMAF monomethyl auristatin F
- the drug moiety (D) can also include a camptothecin derivative 7-ethyl-10-hydroxycamptothecin (SN38), which is based on topoisomerase as an action target Anticancer drugs that inhibit DNA synthesis.
- SN38 camptothecin derivative 7-ethyl-10-hydroxycamptothecin
- protein toxins include but are not limited to: diphtheria toxin A chain, non-binding active fragment of diphtheria toxin, exotoxin A chain, ricin A chain, acacia soybean protein A chain ( abrin A chain), modeccin A chain, alpha-sarcin, Aleurites fordii protein, dianthin proteins, curcin , crotonin (crotin), gelonin (gelonin), mitogellin (mitogellin), restricted aspergillin (restrictocin), phenomycin (phenomycin), enomycin (enomycin) and so on.
- therapeutic radionuclides include, but are not limited to: radioisotopes of 32P, 33P, 90Y, 125I, 131I, 131In, 153Sm, 186Re, 188Re, 211At, 212Bi, 212Pb and Lu.
- the eighth aspect of the present invention provides the preparation method of the antibody-drug complex provided by the seventh aspect of the present invention, including: on the basis of the anti-CLD18A2 single-domain antibody, it can be formed by increasing the tandem connection of the single-domain antibody and the setting of the accessory functional region.
- a fusion protein of an anti-CLD18A2 single domain antibody the fusion protein is cross-linked with a cytotoxic molecule to provide the antibody-drug complex.
- the multivalent tandem of anti-CLD18A2 single-domain antibody can increase the binding ability to human CLD18A2, and the accessory functional region can be designed according to different purposes, which can prolong the half-life in vivo, or improve the hydrophilicity, or increase the coupling of toxic chemical groups site.
- two anti-CLD18A2 single-domain antibodies of the same sequence are connected in series, and a six-repeat structure of three amino acids of human serum albumin binding sequence (ABD) and GGC (SEQ ID NO. 125), after the formed single-domain antibody fusion protein is cross-linked with linker-MMAE through sulfhydryl groups, a single-domain antibody-drug complex with a DAR of 4.42 is obtained.
- ABS human serum albumin binding sequence
- GGC SEQ ID NO. 125
- an anti-CLD18A2 single-domain antibody of the same sequence is connected in series at the C-terminus of the anti-CLD18A2 single-domain antibody, and a highly hydrophilic polypeptide is fused at the C-terminus, and 6 Cys amino acids are introduced to form
- the single-domain antibody derivative (SEQ ID NO. 126) was cross-linked with linker-MMAE through sulfhydryl groups to obtain an antibody-drug complex with a DAR of 4.10.
- a single-domain antibody derivative (SEQ ID NO. 147) formed by fusing human IgG1FC to the C-terminus of anti-CLD18A2 single-domain antibody, after cross-linking with linker-MMAE through interchain sulfhydryl groups , an antibody-drug complex with a DAR of 4.21 was obtained.
- the cross-linking amino acid is cysteine
- it can include: (1) The cysteine group on the single-domain antibody derivative reacts with the linking chain reagent, thereby forming a ligand derivative-linking through a covalent bond chain intermediate, which is subsequently reacted with the activated drug moiety D; or, (2) the nucleophilic group of the drug moiety is reacted with the linking chain reagent, thereby forming a drug-linking chain intermediate by covalent bonding, which is subsequently reacted with the single domain antibody
- the cysteine group on the derivative reacts.
- Coupling methods (1) and (2) can be used for a variety of targeting ligands, drug moieties and linkers to prepare ligand derivative-drug complexes of formula I.
- Cysteine sulfhydryl groups are nucleophilic and can react with electrophilic groups on linker reagents and drug-linker intermediates to form covalent bonds, said drug-linker intermediates including: (i) Active esters such as NHS esters, HOBt esters, haloformates and acid halides; (ii) alkyl and benzyl halides such as haloacetamides; (iii) aldehydes, ketones, Carboxyl and maleimide groups; and (iv) disulfides exchanged by sulfide, including pyridyl disulfides.
- Nucleophilic groups on the drug moiety can include, but are not limited to, amine, sulfhydryl, hydroxyl, hydrazide, oxime, hydrazine, thiosemicarbazide, hydrazine carboxylate, and arylhydrazide groups, which can interact with the linker moiety and The electrophilic groups on the linker reagent react to form a covalent bond.
- TCEP tris (2-carboxyethyl) phosphine hydrochloride treatment to reduce the interchain disulfide bond of the antibody, and then react with the linking chain reagent.
- cross-linking amino acid is selenocysteine
- cross-linking amino acid is an unnatural amino acid
- Han The protocol of Xiao et al. Angew Chem Int Ed Engl. 2013 Dec 23;52(52):14080-3
- the ninth aspect of the present invention provides a pharmaceutical composition, comprising the anti-CLD18A2 single domain antibody provided by the first aspect of the present invention, the bispecific antibody provided by the second aspect of the present invention, and the antibody provided by the fifth aspect of the present invention
- the culture of the expression system, or the antibody-drug complex provided by the seventh aspect of the present invention is usually a therapeutically effective amount.
- the pharmaceutical composition may also include a pharmaceutically acceptable carrier.
- the carrier may include various excipients and diluents which are not themselves necessary for the active ingredient and which are not unduly toxic after administration.
- the pharmaceutical composition can be administered by injection route, especially intravitreal injection, so the pharmaceutical composition is preferably a powder for injection (such as freeze-dried powder for injection) and a liquid preparation .
- a tenth aspect of the present invention provides a cell comprising a membrane-bound chimeric antigen receptor, wherein the chimeric antigen receptor includes a transmembrane domain, an intracellular domain and an extracellular domain, and the extracellular domain includes the first
- the anti-CLD18A2 single domain antibodies are provided.
- the surface of the cells provided by the present invention can express the anti-CLD18A2 single-domain antibody, and the cells can usually bind to their corresponding antigens through the extracellular domain, and more specifically can bind to the anti-CLD18A2 single-domain antibody through the extracellular domain.
- the cells when the polypeptide binds to its corresponding antigen, the cells can be activated and/or stimulated to proliferate and kill the corresponding target cells.
- the cells can be T lymphocytes, NK cells, macrophages and the like.
- the chimeric antigen receptor may include an extracellular domain, and the extracellular domain may generally include an antibody targeting the target antigen, that is, the above-mentioned anti-CLD18A2 single domain antibody.
- the chimeric antigen receptor can also include a transmembrane domain, and the transmembrane domain can mainly fix the chimeric antigen receptor on the cell membrane of T cells.
- the transmembrane domain may include sequences of CD4, CD8, CD8b, CD28 transmembrane domains, subunits of T cell receptors such as alpha, beta, gamma or delta, subunits of IL-2 receptors (alpha chain), low Affinity Nerve growth factor receptor (LNGFR or p75) subunit (beta chain or gamma chain), or the transmembrane domain of the subunit chain of Fc receptors.
- the transmembrane domain may comprise the amino acid sequence shown in SEQ ID NO:128. In another specific embodiment of the present invention, the transmembrane domain may comprise the amino acid sequence shown in SEQ ID NO:129.
- the chimeric antigen receptor may also include an intracellular domain.
- the intracellular domain may be the cytoplasmic sequences of native T cell receptors and coreceptors that cooperate to initiate signal transduction upon antigen binding, as well as any derivatives or variants of these sequences, and any synthetic functional equivalents sequence.
- the intracellular domains can generally be divided into two broad categories, and can include, for example, costimulatory domains and/or signaling domains.
- the co-stimulatory domain can typically provide a secondary or co-stimulatory signal for full cell activation in an antigen-independent manner, which can bind to a cognate co-stimulatory ligand on an antigen-presenting cell to enhance T cell responses, for example by increasing Proliferation activation, differentiation, etc.
- the costimulatory domain may include CD28, CD27, 4-1BB (CD137), OX40 (CD134), ICOS (CD278), CD30, CD40, PD-1 (CD279), CD2, CD7, NKG2C (CD94), B7 - Intracellular domain of H3 (CD276).
- the costimulatory domain may include CD28 and/or CD137, and the amino acid sequence thereof may include the amino acid sequences shown in SEQ ID NO: 130 and SEQ ID NO: 131.
- the signaling domain generally refers to a region capable of transducing a signal into the cell when the above-mentioned antibody recognizes an antigen on the surface of the target cell.
- the signaling domain may be an immunoreceptor tyrosine-based activation motif (ITAM), which is a well-defined signaling motif typically present in the intracytoplasmic tail of various receptors and used as syk/ Binding site for zap70-like tyrosine kinases.
- ITAM immunoreceptor tyrosine-based activation motif
- the signaling domains may include the signaling domains of CD3 ⁇ , FcR ⁇ , FcR ⁇ , FcR ⁇ , CD3 ⁇ , CD3 ⁇ , CD5, CD22, CD79a, CD79b, and CD66d.
- the signaling domain includes the signaling domain of CD3 ⁇ , and its amino acid sequence may include the amino acid sequence shown in SEQ ID NO: 132.
- Natural TCRs contain the CD3 ⁇ signaling molecule, so the use of this effector domain is closest to the TCR constructs that occur in nature.
- the chimeric antigen receptor may also include a hinge region, and the hinge region includes the CD8 dumpling region or the CH2 and/or CH3 domains of IgG1 or IgG4, preferably the dumpling chain of CD8b Area.
- the hinge region may comprise the amino acid sequence shown in SEQ ID NO:127.
- the chimeric antigen receptor generally includes an anti-CLD18A2 single domain antibody, a transmembrane domain (TM) and an intracellular domain in sequence from the N-terminal to the C-terminal, and the intracellular domain starts from the N-terminal To the C-terminus, it includes a costimulatory domain (ITAM) and a signaling domain ( ⁇ ).
- TM transmembrane domain
- ITAM costimulatory domain
- ⁇ signaling domain
- a hinge region may also be included, and the hinge region may typically be located between the anti-CLD18A2 single domain antibody, transmembrane domains.
- the chimeric antigen receptor may include anti-CLD18A2 single domain antibody, CD8bhinge, CD8bTM, CD28ITAM, and CD3 ⁇ in sequence from the N-terminus to the C-terminus.
- the chimeric antigen receptor may include anti-CLD18A2 single domain antibody, CD8bhinge, CD8bTM, CD137ITAM, CD3 ⁇ in sequence from the N-terminus to the C-terminus.
- the chimeric antigen receptor may include anti-CLD18A2 single domain antibody, CD8bhinge, CD28TM, CD28ITAM, CD137ITAM, and CD3 ⁇ in sequence from the N-terminus to the C-terminus.
- the chimeric antigen receptor may further comprise a signal peptide, and the signal peptide is mainly used to express the chimeric antigen receptor on the cell membrane.
- the amino acid sequence shown in SEQ ID NO: 136 is shown in SEQ ID NO: 136.
- the amino acid sequence of the chimeric antigen receptor includes the sequence shown in one of SEQ ID NO. 137-140.
- the eleventh aspect of the present invention provides a culture of the anti-CLD18A2 single domain antibody provided by the first aspect of the present invention, the bispecific antibody provided by the second aspect of the present invention, and the expression system of the antibody provided by the fifth aspect of the present invention , or the use of the antibody-drug complex provided by the seventh aspect of the present invention, the pharmaceutical composition provided by the ninth aspect of the present invention, or the cell provided by the tenth aspect of the present invention in preparing a medicine.
- the drug may be a drug for treating tumors.
- the tumor may be a solid tumor or a hematological tumor, more specifically, colon cancer, lung cancer, liver cancer, breast cancer, esophageal cancer, head and neck cancer, skin cancer, kidney cancer, leukemia, coad (colon cancer), lihc (hepatocellular carcinoma) ), ov (ovarian serous cystadenocarcinoma), ucec (endometrial cancer), thca (thyroid cancer), skcm (skin melanoma), luad (lung adenocarcinoma), hnsc (head and neck squamous cell carcinoma), gbm (glioma multiforme), prad (prostate cancer), thym (thymic carcinoma), lgg (brain low-grade glioma), read (rectal adenocarcinoma), pcpg (pheochromocytoma and paraganglioma) ), esca (esophageal cancer), kirc
- the above-mentioned substances can be a single medicinal component, or can be combined with other active components to jointly treat tumors.
- the twelfth aspect of the present invention provides a treatment method, comprising administering to an individual a therapeutically effective amount of the anti-CLD18A2 single domain antibody provided in the first aspect, the bispecific antibody provided in the second aspect of the present invention, and the fifth aspect of the present invention.
- the treatment methods provided by the present invention can be used to treat indications including but not limited to tumors.
- the "individual” generally includes mammals, which may be rodents, artiodactyls, odd ungulates, lagomorphs, non-human primates, primates, etc., which may be be monkeys, apes or humans, such as mammals, dogs, cats, horses, sheep, pigs, cattle, etc., which may benefit from treatment with the T lymphocytes, or compositions.
- the "therapeutically effective amount” generally refers to an amount which, after an appropriate period of administration, is effective in treating the diseases listed above.
- Selection of a preferred therapeutically effective amount can be determined by one of ordinary skill in the art based on a variety of factors (eg, through clinical trials), such as tumor growth, proliferation, recurrence and/or metastasis when the above-mentioned substances are used in the individual to which they are administered Can be inhibited, more specifically, at least 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95% of tumor growth, proliferation, recurrence and/or metastasis or 99% partially suppressed.
- factors eg, through clinical trials
- the anti-CLD18A2 single domain antibody provided by the present invention has good affinity for Claudin18.2, and can be used to further construct bispecific antibodies, antibody-drug complexes, chimeric antigen receptors, etc.
- Antibodies, antibody-drug complexes, CAR-T cells, etc. have good targeting and killing effects on target cells, and have good industrialization prospects.
- the experimental methods, detection methods and preparation methods disclosed in the present invention all adopt the conventional molecular biology, biochemistry, chromatin structure and analysis, analytical chemistry, cell culture, recombinant DNA technology and related fields in the technical field. conventional technology. These techniques have been well described in the existing literature. For details, please refer to Sambrook et al.
- MOLECULAR CLONING A LABORATORY MANUAL, Second edition, Cold Spring Harbor Laboratory Press, 1989 and Third edition, 2001; Ausubel et al., CURRENT PROTOCOLS IN MOLECULAR BIOLOGY, John Wiley & Sons, New York, 1987 and periodic updates; the series METHODS IN ENZYMOLOGY, Academic Press, San Diego; Wolfe, CHROMATIN STRUCTURE AND FUNCTION, Third edition, Academic Press, San Diego, 1998; METHODS IN ENZYMOLOGY, Vol.304, Chromatin (PMWassarman and APWolffe, eds.), Academic Press, San Diego, 1999; and METHODS IN MOLECULAR BIOLOGY, Vol. 119, Chromatin Protocols (PBBecker, ed.) Humana Press, Totowa, 1999, et al.
- the human Claudin18.2 full-length gene expression vector pCDNA3.1/CLDN18A2 was constructed and electrotransfected into CHO-S cells, and then positive clones were screened by G418 to obtain the CHO-S cell line CHO-S-Claudin18 expressing Claudin18.2.
- CHO-S-Claudin18.2 cells were subcutaneously immunized with alpaca at multiple points and supplemented with adjuvant. After 3 to 4 times of immunization, blood was drawn to check that the antibody titer met the requirements, and then blood was drawn to extract PBMC, and total RNA was extracted and reverse transcribed.
- the packaged phage was subjected to subtractive hybridization between CHO-S and CHO-S expressing Claudin18.2, as well as multiple rounds of panning, and finally multiple high-affinity single-domain antibodies that specifically bind to Claudin18.2 were obtained. Finally, a group of humanized high-affinity single-domain antibodies that specifically bind to Claudin18.2 are obtained, as shown in the sequences of SEQ ID NOs: 67-90.
- the high-fidelity enzyme GVP8 (General Biosystems (Anhui) Co., Ltd.) was used for PCR amplification with the synthetic humanized sequence as a template, a signal peptide sequence was introduced at the 5' end of the sequence, and a 6xHis histidine tag code was introduced at the 3' end. Sequence, PCR product electrophoresis and gel cutting to recover a band of about 500 bp, and the recovered PCR product was recombined with the pET32a+ vector (Novagen) digested with endonuclease NdeI and EcoRI with a recombination kit (Nearshore Protein Technology Co., Ltd.) Connected, constructed E. coli expression plasmid, transformed into E.
- GVP8 General Biosystems (Anhui) Co., Ltd.
- the E. coli expression plasmid determined by sequencing was transformed into the E. coli expression host Rosetta (DE3) to construct an E. coli expression strain. Recombinant clones were picked on ampicillin-resistant plates, cultured, and induced to express overnight with 1 mM IPTG at 30°C. The bacterial liquid induced to express overnight was sonicated, centrifuged at 12,000g at 4°C for 10 minutes, and the supernatant was taken and purified with a Ni column (Borgron Biotechnology Co., Ltd.), and the final protein purity reached more than 90%.
- CHO-S-CLD18A2 was plated in a 96-well plate at 5 ⁇ 10 5 /ml cells per well, and blocked with 3% BSA for 1 hour at room temperature.
- the purified histidine tag-fused CLD18A2 nanobody was used 1% BSA gradient dilution was added to the blocked cells, and incubated at room temperature for 1 hour. After washing, 100 ⁇ l/well of mouse anti his tag antibody (R&D Systems, Inc) diluted at 1:5000 was added and incubated for 1 hour at room temperature.
- the S4-14 bispecific antibody converted the amino acid sequence into each base sequence according to the codon preference of CHO cells, and obtained the full-length DNA by gene synthesis (Nanjing GenScript Biotechnology Co., Ltd.). Take each DNA as a template, carry out PCR amplification with high-fidelity enzyme GVP8 (Anhui General Biotechnology Co., Ltd.), insert pCDNA3.1 vector after HindIII/EcoRI digestion, and recombine and connect to construct two expression plasmids (expressing SEQ ID NO.
- the two plasmids were extracted with the endotoxin-removing plasmid large extraction kit (Biomiga) and mixed 1:1, and the mixed plasmid was mixed with the transfection reagent PEI (Polysciences, Inc.) 1: 3 Mix well and let it stand for 30 min, then add it to HEK293F cells for co-transfection, culture in a 37°C, 5% CO 2 shaker incubator for 7 days, and then centrifuge to get the supernatant.
- PEI Polysciences, Inc.
- Affinity chromatography 50ml Protein A chromatography column (Borgron, AA0273) was treated with 0.1M NaOH at a flow rate of 10ml/min for 30min to remove residual protein on the column. It was then equilibrated with 150 mM NaCl 20 mM PB (pH 6.5) for at least 3 column volumes until the pH reached 6.5 and the conductance was around 15 mS/cm. The culture filtrate was passed through a Protein A chromatography column at a flow rate of 10 ml/min with a residence time of about 5 min on the column, and then equilibrated with 150 mM NaCl 20 mM PB (pH 6.5) for at least 3 column volumes.
- Anion exchange chromatography 50ml Q FF chromatography column (Borgron, AI0024) was treated with 0.5M NaOH at a flow rate of 10ml/min for 30min to remove residual protein on the column. It was then equilibrated with 20 mM PB (pH 6.5) for at least 5 column volumes until the pH reached 6.5. The sample obtained by affinity chromatography was passed through the Q FF chromatography column at a flow rate of 10ml/min, the residence time on the column was about 5min, and the flow-through sample was collected and stored. It was then washed with 500 mM NaCl 20 mM PB (pH 6.5) to remove impurities bound to the column and identified.
- Cation exchange chromatography 50ml SP mustang chromatography column (Borgron, AI0192) was treated with 0.5M NaOH at a flow rate of 10ml/min for 30min to remove residual protein on the column. It was then equilibrated with 20 mM PB (pH 6.5) for at least 5 column volumes until the pH reached 6.5. The flow-through sample obtained by anion exchange chromatography was passed through the SP mustang chromatography column at a flow rate of 10ml/min, and the residence time on the column was about 5min. It was then eluted with 200 mM NaCl 20 mM PB (pH 6.5) and eluted samples were collected.
- the S2-12 bispecific antibody, and the negative controls Anti-C18.2-hu19V3 and Anti-CD3 were prepared according to the vector construction, expression and purification methods provided in Example 1.
- Detector Agilent 1100LC; Detection wavelength: 214nm; Mobile phase: 150mM pH7.0PB+5% isopropanol; Chromatographic column: Superdex 200Increase 5/150GL; Running time: 15 minutes; Column temperature 25°C. Purity is greater than 95%.
- luciferase detection cells Construction of luciferase detection cells: Jurkat cells were transformed into NFAT-responsive luciferase system, and the detection cell line Jurkat-PB-NFAT-luc2p was obtained.
- Target cell plating Take an appropriate amount of CHO-S-Claudin18.2 cells cultured in suspension, centrifuge at 800 rpm for 5 min, discard the supernatant, add RPMI1640 medium (containing FBS) to resuspend, count, take the required cells and dilute to the specified concentration, Add to 96-well cell culture plates.
- Protein dilution S2-12 and S4-14 bispecific antibodies prepared by gradient dilution in RPMI1640 medium (containing FBS), as well as negative controls Anti-C18.2-hu19V3 and Anti-CD3 to the specified concentration, and then add an appropriate volume In a 96-well plate, target cells were incubated with antibody protein for 30 min at 37°C, and Jurkat-PB-NFAT-luc2p cells were added.
- Jurkat-PB-NFAT-luc2p plating Dilute an appropriate amount of Jurkat-PB-NFAT-luc2p cells to the specified concentration and add them to a 96-well cell culture plate. Incubate for about 20h for detection.
- Photometric value detection add 10 ⁇ l/well of the detection solution provided by the luciferase detection kit (Promega, E2620), shake for 2 min, transfer 60 ⁇ l of the solution to a 96-well white plate, and detect in a microplate reader.
- the software GraphPad Prism v5.0 was used for data processing and graph analysis, and the activation curves and EC50 values of various anti-CLD18A2/anti-CD3 bispecific antibodies for the detection system were obtained, as shown in Figure 1, S2-12, S4- 14
- Two bispecific antibodies with different structures produced similar activation ability of the in vitro T cell system, while the negative controls Anti-C18.2-hu19V3 and Anti-CD3 did not have the ability to activate the detection system. It shows that anti-CLD18A2 antibody and anti-CD3 antibody can activate the Claudin18.2 antigen target cells and T cell effector system only when bispecific antibodies are formed, but neither antibody alone has this function.
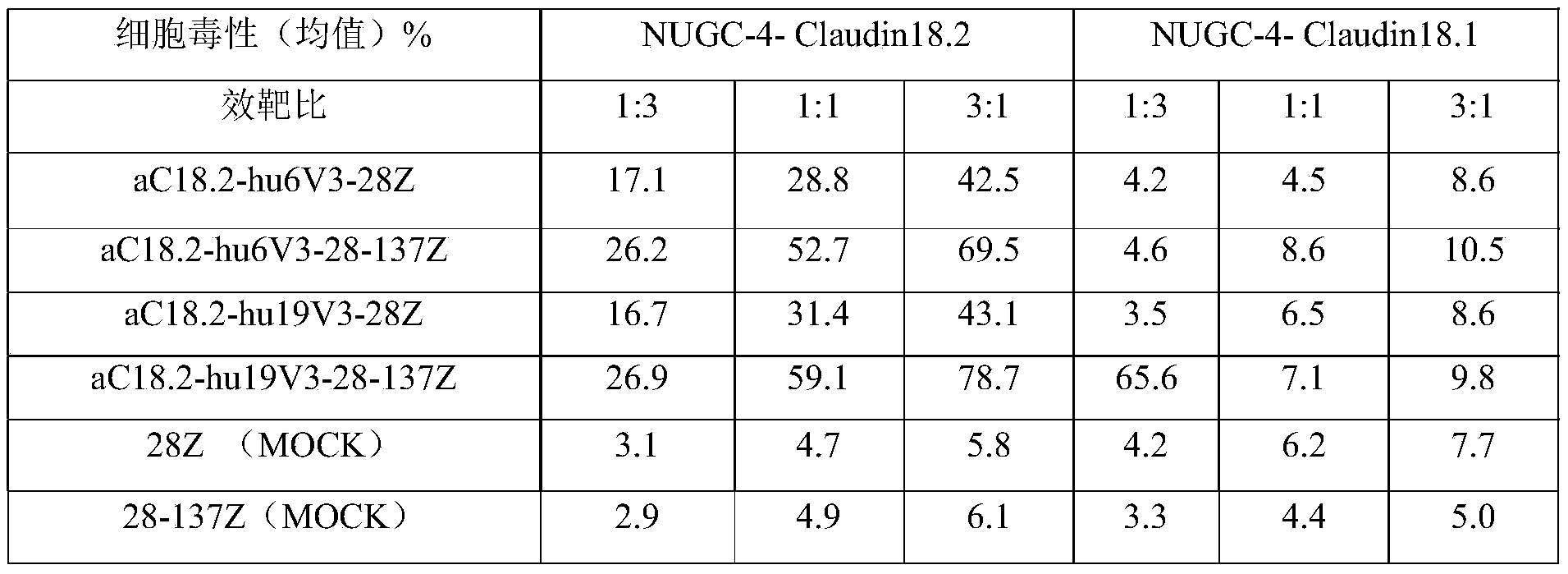
- the present invention uses T cells (Miaotong Biotechnology) as effector cells to conduct a cytotoxicity test.
- Anti-S2-12, S4-14 bispecific antibodies, as well as negative controls Anti-C18.2-hu19V3 and Anti-CD3 were serially diluted, and 50 ⁇ l was added to each well.
- Claudin18.2 and Claudin18.1 stably transfected cells NUGC-4-Claudin18.2 and NUGC-4-Claudin18.1 were washed and resuspended in 5% FBS 1640 medium (Gibco), respectively, to a concentration of about 2 ⁇ 10 5 /ml For cell density, add 50 ⁇ l per well to the corresponding 96-well plate.
- Human T lymphocytes from healthy donors were resuspended in 5% FBS 1640 medium, and 1 ⁇ 10 5 cells were added to each well to make the effect-target ratio 10:1. After incubating at 37°C for 4 hours, press The LDH detection kit (Dongren Chemical Technology (Shanghai) Co., Ltd.) detected the release of LDH and evaluated the in vitro cell killing effect of anti-Claudin18.2/anti-CD3 bispecific antibodies.
- the LDH detection kit Dongren Chemical Technology (Shanghai) Co., Ltd.
- the anti-CLD18A2/anti-CD3 bispecific antibodies S2-12 and S4-14 had a very significant killing effect on NUGC-4-Claudin18.2 with high Claudin18.2 expression, with EC50 values of 24.65pM and 21.15pM, respectively. ;
- anti-CLD18A2/anti-CD3 bispecific antibodies had no obvious killing effect (as shown in Figure 2).
- tumor-bearing NSG mice of a xenograft tumor model (patientl derived xenograft, PDX) established by gastric cancer tissue derived from patients are used to analyze the tumor suppressive effect of anti-Claudin18.2/anti-CD3 bispecific antibody.
- PDX patientl derived xenograft
- the tumor-bearing mice were randomly assigned to 5 mice in each group, and 2 ⁇ 10 7 healthy human PBMC cells were injected intraperitoneally.
- tumor-bearing mice were intraperitoneally injected with 5 ⁇ g (25 ⁇ g/ml, 200 ⁇ l PBS), bispecific antibody S2-12, once every two days for 4 weeks, and the tumor volume was recorded twice a week.
- S4-14 10 ⁇ g (25 ⁇ g/ml, 200 ⁇ l PBS) once a week for 4 weeks, tumor volume was recorded twice a week.
- Example 4 Preparation of anti-CLD18A2 single-domain antibody fusion protein and control antibody
- the specific positive sequences and humanized sequences obtained by screening were used as templates, the upstream primer 5'-gtgctgctgctgtgggtgccaggatccaccgggcaggtgcagctcgtggagtc-3' (SEQ ID NO.141) and the downstream primer 5'-gcaggacttgggctcagaagacacggtgaccagggtccctggcc-3' (SEQ ID NO.142) , use high-fidelity enzyme GVP8 (Anhui General Biotechnology Co., Ltd.) to carry out PCR amplification, the PCR product is electrophoresed and cut into the gel to recover a band of about 400bp, and the recovered PCR product is mixed with a signal peptide and human IgG1Fc sequence (amino acid sequence SEQ NO.119) pCDNA3.1 vector was recombined to construct a cell expression plasmid fused with Anti-CLD18A2 single domain antibody and human Ig
- the fused cells express the plasmid, mix the plasmid with the transfection reagent PEI (Polysciences, Inc.) 1:3 and let stand for 30 min, then add it to HEK293F cells, and culture at 37°C, 5% CO 2 in a shaking incubator for 7 Days later, the supernatant was collected by centrifugation. The supernatant was adjusted to pH 7.0 and loaded onto a ProteinA affinity chromatography column (Borgron Biotechnology Co., Ltd.), eluted with 100% 0.1M Gly-HCl (pH 3.0); the eluent was pre-added with 10% 1M Tris-HCl (pH 8.5).
- PEI Polysciences, Inc.
- the 100% eluate was diluted to a conductivity of 4ms/cm, adjusted to pH 5.5, centrifuged (8000rpm, 4°C, 10min), and the supernatant was adjusted to pH 5.0 and loaded onto a DSP column (Borgron Biotechnology Co., Ltd. Company), 0-60% eluent (20 mM NaAc, 0.5 M NaCl, pH 5.0) linear elution, flow rate 2 ml/min, 15 min.
- the chimeric antibody (named ch-175D10 in the US9751934B2 patent) composed of the heavy chain of SEQ ID NO: 118 and the light chain of SEQ ID NO: 125 in US9751934B2 was used as a control antibody, and the corresponding polynucleotide sequence of its amino acid sequence was compared with that of pCDNA3. 1.
- the vector was recombined and ligated, and the transient transfection, expression and purification of HEK293F cells were carried out by the same method as in Example 2.
- Example 5 Detection of endocytic activity of anti-CLD18A2 single-domain antibody fusion protein
- the full-length gene of CLD18A2 (SEQ ID NO. 143) and the gfp gene were linked with an internal ribosome entry site (IRES) sequence and constructed in pCDNA3.1 vector (Life Technologies) to achieve co-expression of CLD18A2 and gfp.
- the expression plasmid was extracted, and Lipofectamine 3000 (Invitrogen, L3000001) transfection reagent was used to transfect CHO-K1 cells according to the instructions. The next day, the transfected CHO-K1 cells were trypsinized and then seeded in a 96-well plate at a final concentration of 2 ⁇ 10 6 /ml cells.
- Conjugate pH-sensitive fluorescent dyes ( ⁇ pH7 can be excited to fluoresce) with antibodies.
- the labeled antibody When the labeled antibody is endocytosed through receptors, etc., it can be excited to generate fluorescence in an acidic environment, and the endocytosis can be judged by the intensity of fluorescence generated. efficient.
- the humanized antibody selected in the present invention is an endocytic antibody, which can specifically bind to CLD18A2 and mediate endocytosis, while the isotype control antibody has no obvious endocytosis.
- the design was based on the humanized sequence Anti-C18.2-hu19V3 of the specific positive sequence Anti-C18.2-19 obtained by screening, and the Anti-C18.2-hu19V3 was concatenated twice, and the C-terminal A six-repeat structure of three amino acids of human serum albumin binding sequence (ABD) and GGC is introduced, and the complete sequence is shown in SEQ ID NO.125.
- the complete base sequence encoding the amino acid sequence shown in SEQ ID NO.125 was synthesized in Universal Biosystems (Anhui) Co., Ltd., and constructed in the pET32a vector, and the expression plasmid was transformed into E. coli expression host Rosetta (DE3) to construct E. coli expression strains.
- Recombinant clones were picked on ampicillin-resistant plates, cultured, and induced to express overnight with 1 mM IPTG at 30°C.
- the bacterial liquid induced to express overnight was sonicated, centrifuged at 12,000g at 4°C for 10 minutes, and the supernatant was taken and purified with a Ni column (Borgron Biotechnology Co., Ltd.), and the final protein purity reached more than 90%.
- the design was based on the humanized sequence Anti-C18.2-hu19V3 of the specific positive sequence Anti-C18.2-19 obtained by screening, and the Anti-C18.2-hu19V3 was concatenated twice. (Anhui) Co., Ltd.
- Recombinant clones were picked on ampicillin-resistant plates, cultured, and induced to express overnight with 1 mM IPTG at 30°C.
- the bacterial liquid induced to express overnight was sonicated, centrifuged at 12,000g at 4°C for 10 minutes, and the supernatant was taken and purified with a Ni column (Borgron Biotechnology Co., Ltd.), and the final protein purity reached more than 90%.
- SEQ ID NO. 147 The sequence of SEQ ID NO. 147 was prepared as shown in Example 4.
- the purified anti-CLDN18A2 single-domain antibody fusion protein and ch-175D10 were dissolved in PBS solution, and 10-fold excess TCEP was added to reduce the interchain disulfide bonds at 25°C, and the reducing agent was removed by dialysis. Re-formation of interchain disulfide bonds can be obtained by adding 2 times the molar concentration of TCEP to CuSO4 at 25 °C. Then, MC-VC-PAB-MMAE with a molar concentration of 10 times the antibody was added and reacted at 25°C for 1 hour to form the antibody derivative Ab-MC-VC-PAB-MMAE complex. The complex was dialyzed or ultrafiltered to remove unbound MC-VC-PAB-MMAE. The DAR of the final product obtained was determined by LC/MS.
- the final concentration of the obtained Ab1-MC-VC-PAB-MMAE was 1.2 mg/ml.
- the LC-MS method was used to detect and analyze, and it was proved that there were no free toxin small molecules in the obtained sample.
- the final concentration of the obtained Ab2-MC-VC-PAB-MMAE was 2.5 mg/ml.
- the LC-MS method was used to detect and analyze, and it was proved that there were no free toxin small molecules in the obtained sample.
- the final concentration of the obtained Ab3-MC-VC-PAB-MMAE was 5.4 mg/ml.
- the LC-MS method was used for detection and analysis, which proved that there were no free toxin small molecules in the obtained sample.
- the final concentration of the obtained ch175D10-MC-VC-PAB-MMAE was 4.4 mg/ml.
- the LC-MS method was used to detect and analyze, and it was proved that there were no free toxin small molecules in the obtained sample.
- CLD18A2 protein (Kactus Biosystems) was coated on a 96-well microtiter plate, 100 ng per well, overnight at 4°C, 10 mM Na 2 CO 3 -NaHCO 3 ( After washing at pH 10.0), 5% nonfat milk powder was coated for 1 hour, and after washing again, the single-domain antibody-drug complex was added; after incubation at 37°C for 2 hours, biotin-labeled anti-single-domain antibody rabbit polyclonal antibody (self-made) was added. ), and then added Strep-HRP to incubate at 37°C for 1 h. After washing, TMB developed color. The results are shown in Table 4. The binding activity of Ab3 before and after drug conjugation was not affected, and the remaining two structures, Ab1 and Ab2, still had good binding activity after drug conjugation.
- NUGC-4-CLD18A2 human CLD18A2
- original NUGC-4 cells grown to 90% confluence were cultured, washed with PBS, and trypsinized , after termination of digestion, cells were collected and seeded in 96-well plates, 2x10 4 /well, DMEM medium with 10% fetal bovine serum, 2 mM glutamate, and cultured overnight in a 5% CO 2 incubator.
- the above results show that the antibody-drug complex described in the present invention can specifically target human CLD18A2, and the positive cytotoxicity is strong. cell killing.
- the unconjugated single domain antibody fusion protein Ab3 had no obvious cytotoxic effect.
- mice inoculated with the antibody-drug complex were well controlled relative to the PBS control group, compared with the positive control group ch175D10-MC-VC.
- -PAB-MMAE also has better tumor suppressive effect.
- Ab1-MC-VC-PAB-MMAE and Ab2-MC-VC-PAB-MMAE rebounded to a certain extent in the later stage, they eventually maintained a lower tumor volume without further significant increase, indicating that there was a significant increase in tumor volume. tumor suppressor effect.
- the anti-CLD18A2 single-domain antibody of the present invention is used for the construction of chimeric antigen receptors.
- Table 6 lists the constructed chimeric antigen receptors comprising signal peptides and their structures (signal peptide-antigen recognition domain-hinge region-trans membrane domain - intracellular signaling domain).
- the present invention uses the third-generation self-inactivating lentiviral vector system, which has four plasmids, namely, the enveloped plasmid pLP/VSVG (purchased from Addgene) encoding the VSV-G protein; Packaging plasmid pLP1; packaging plasmid pLP2 (purchased from Addgene) encoding Rev protein and recombinant expression vector encoding the target gene CAR constructed based on the empty vector pLVX-IRES-ZsGreen1 (purchased from Addgene).
- the enveloped plasmid pLP/VSVG purchased from Addgene
- Packaging plasmid pLP1 packaging plasmid pLP2
- Rev protein Rev protein
- recombinant expression vector encoding the target gene CAR constructed based on the empty vector pLVX-IRES-ZsGreen1 (purchased from Addgene).
- Synthetic structural sequences (SEQ ID NO: 137, SEQ ID NO: 138, SEQ ID NO: 139, SEQ ID NO: 140), the synthesized sequences pass through the EcoRI and BamHI restriction sites at both ends, and the same restriction vector pLVX -IRES-ZsGreen1 (Addgene) was ligated by T4 ligase (Takara).
- the ligation product was transformed into Top10F', coated with ampicillin-resistant plate, cloned, cultured and sequenced to confirm, and the expression vectors expressing the above CAR were obtained: pLVX-aC18.2-hu6V3-28Z, pLVX-aC18.2-hu6V3-28-137Z , pLVX-aC18.2-hu19V3-28Z and pLVX-aC18.2-hu19V3-28-137Z,
- Lentiviral packaging follows the conventional methods, as follows: 5 ⁇ 10 6 cell density cultivation of HEK-293T cells (ATCC) cells in 10cm dishes, 37 °C, 5% CO 2 incubator overnight, the medium containing 10% fetal bovine Serum (Gibco) in DMEM (Gibco). Change the culture medium to serum-free DMEM about 2 hours before transfection.
- plasmids providing viral membrane proteins and structural proteins
- the lentiviral plasmid expressing the target sequence CAR or empty vector used 5 ⁇ g
- pLP1 used 2.5 ⁇ g
- pLP2 used 2.5 ⁇ g
- pLP/VSVG used 1.25 ⁇ g.
- the mixture of the above four plasmids was added to 500 ⁇ l MEM medium, and 25 ⁇ L Lipofectamine 2000 transfection reagent (thermo fisher) was added to 500 ⁇ L MEM medium in another microcentrifuge tube.
- HEK-293T cells were seeded in a 96-well culture plate in DMEM with 10% fetal bovine serum at a cell density of 1 ⁇ 10 5 /mL at a cell density of 100 ⁇ L/well, and cultured overnight at 37°C, 5% CO 2 . The next day, discard 50 ⁇ L/well of the culture supernatant, and add 50 ⁇ L/well of fresh culture medium containing polybrene with a final concentration of 6 ⁇ g/mL, incubate at 37°C, 5% CO 2 for 30 min. Add 10 ⁇ L/well of virus stock solution, incubate at 37°C, 5% CO 2 . 48h after infection, GFP was detected by flow cytometer, and the number of cells with a positive rate of 5-20% was appropriate, and the calculated titer was about 2 ⁇ 10 6 U/mL.
- Example 11 CAR-T cells specifically targeting Claudin18.2
- Human peripheral blood mononuclear cells (Shanghai Miaotong) were obtained from healthy human peripheral blood by density gradient centrifugation, and sorted by CD3 MicroBeads (Miltenyi Biotec GmbH) according to the instructions. Quantum007 lymphocyte culture medium (purchased from PAA Laboratories GmbH) was added at a density of about 1 ⁇ 10 6 /mL, and DynabeadsTM Human T-Activator CD3/CD28 (thermofisher) was added at a cell:magnetic bead ratio of 1:1 and the final cells were added. Recombinant human IL-2 (Shanghai nearshore) at a concentration of 100 U/mL was stimulated and cultured for 24 h.
- Quantum007 lymphocyte culture medium purchased from PAA Laboratories GmbH
- DynabeadsTM Human T-Activator CD3/CD28 thermofisher
- T cells were then infected with the recombinant lentivirus described above (Example 10.3) at MOI ⁇ 5.
- the infected cells were passaged at a density of 5 ⁇ 10 5 /mL every other day, and the lymphocyte culture medium was supplemented with recombinant human IL-2 at a final concentration of 100 U/mL.
- the positive cells detected by GFP were regarded as positive cells expressing the chimeric antigen receptor.
- Uninfected T cells were used as negative control, and the positive rate of virus-infected T cells expressing different chimeric antigen receptors was about 64.2%.
- Results Table 7 shows that when the effector-target ratio is 3:1, specific aC18.2-CAR-T cells can effectively kill Claudin18.2-positive cells, but almost no Claudin18.2-negative cells.
- the above results show that aC18.2-CAR-T can specifically kill Claudin18.2 positive cells, and the killing effect is positively correlated with the effector-target ratio.
- NUGC-4-Claudin18.2 and aC18.2-CAR-T cells were co-cultured at a ratio of 1:1, and the culture supernatant was collected after 24 hours of incubation, and IL-2 (R&D Systems, Inc.), TNF- ⁇ (R&D Systems, Inc.) and IFN- ⁇ (R&D Systems, Inc.) were detected according to the kit instructions.
- Figure 7 shows that in NUGC-4-Claudin18.2, aC18.2-CAR-T cells aC18.2-hu19V3-28-137Z co-incubated the secretion of cytokines such as IL-2, TNF- ⁇ and IFN- ⁇ significantly higher than negative cells NUGC-4-Claudin18.1.
- NUGC-4-Claudin18.2 a subcutaneous xenograft model was established.
- 3 ⁇ 10 6 th NUGC-4-Claudin18.2 inoculated subcutaneously NOD / SCID mice.
- 100 mg/kg of cyclophosphamide was injected intraperitoneally to clear the immune cells of NOD/SCID mice, so that the adoptively transferred transgenic T lymphocytes could play a better anti-tumor function.
- the present invention effectively overcomes various shortcomings in the prior art and has high industrial utilization value.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Genetics & Genomics (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Engineering & Computer Science (AREA)
- Medicinal Chemistry (AREA)
- Molecular Biology (AREA)
- Immunology (AREA)
- Biochemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Biophysics (AREA)
- Biomedical Technology (AREA)
- Biotechnology (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Microbiology (AREA)
- General Engineering & Computer Science (AREA)
- Pharmacology & Pharmacy (AREA)
- Cell Biology (AREA)
- Physics & Mathematics (AREA)
- Plant Pathology (AREA)
- Mycology (AREA)
- Peptides Or Proteins (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
Description
本发明涉及生物技术领域,特别是涉及一种抗CLD18A2的单域抗体。The present invention relates to the field of biotechnology, in particular to an anti-CLD18A2 single-domain antibody.
密蛋白18(Claudin 18,CLD18)是分子量约为28kD,位于上皮和内皮的紧密连接中的跨膜蛋白,紧密连接在相邻细胞之间。正常上皮组织中,由于细胞间隙紧密导致细胞表面的密蛋白难以被接触,而肿瘤细胞的间隙却较为疏松,因此肿瘤细胞上的密蛋白成为胞外抗体及免疫疗法的潜在靶点。CLD18具有四个疏水区,其作为跨膜域形成两个胞外域,其中疏水区1和疏水区2环绕形成胞外域1,疏水区3和疏水区4环绕形成胞外域2。由于基因不同剪切,CLD18形成两种剪切体:CLD18A1(Claudin18.1)和CLD18A2(Claudin18.2)。CLD18A1在正常肺和胃的上皮中选择性表达,而CLD18A2仅在胃细胞中表达;更重要的是,CLD18A2局限在已分化的胃上皮短寿细胞中,但在胃干细胞中不存在(Niimi T,et al.Biol.2001;21(21):7380–7390.)。这些特性显示CLD18A2是一个用于治疗胃癌和其他CLD18A2阳性肿瘤的有临床价值的治疗靶点。Claudin 18 (Claudin 18, CLD18) is a transmembrane protein with a molecular weight of about 28 kD, located in the tight junctions of the epithelium and the endothelium, which are tightly junctions between adjacent cells. In normal epithelial tissues, the claudin on the cell surface is difficult to be contacted due to the tight intercellular space, while the intercellular space of tumor cells is relatively loose. Therefore, claudin on tumor cells becomes a potential target for extracellular antibodies and immunotherapy. CLD18 has four hydrophobic regions that act as transmembrane domains to form two ectodomains, wherein
由于单一靶点的临床效果常常有限,目前更倾向于采用双靶点的形式,比如基于CD3杀伤的双特异性抗体(T-cell-engaging bispecific antibody,TCB),或者ADC等。但是目前仍未出现安全有效的基于CLD18A2的双特异性抗体或ADC等衍生物。Since the clinical effect of a single target is often limited, it is more inclined to adopt the form of dual targets, such as CD3-based bispecific antibody (T-cell-engaging bispecific antibody, TCB), or ADC, etc. However, there is still no safe and effective CLD18A2-based bispecific antibody or derivatives such as ADC.
发明内容SUMMARY OF THE INVENTION
鉴于以上所述现有技术的缺点,本发明的目的在于提供一种抗CLD18A2的单域抗体,用于解决现有技术中的问题。In view of the above-mentioned shortcomings of the prior art, the purpose of the present invention is to provide an anti-CLD18A2 single-domain antibody for solving the problems in the prior art.
为实现上述目的及其他相关目的,本发明一方面提供一种抗CLD18A2的单域抗体,所述抗CLD18A2的单域抗体的互补决定区包括氨基酸序列如SEQ ID No.4~11其中之一所示的CDR-H1、氨基酸序列如SEQ ID No.19~26其中之一所示的CDR-H2、和氨基酸序列如SEQ ID No.33~39其中之一所示的CDR-H3。In order to achieve the above purpose and other related purposes, one aspect of the present invention provides an anti-CLD18A2 single-domain antibody, the complementarity determining region of the anti-CLD18A2 single-domain antibody comprises an amino acid sequence as shown in one of SEQ ID No. 4-11. CDR-H1 shown in one of the amino acid sequence, CDR-H2 shown in one of SEQ ID No. 19-26, and CDR-H3 whose amino acid sequence is shown in one of SEQ ID No. 33-39.
本发明另一方面提供一种双特异性抗体,所述双特异性抗体包括上述的抗CLD18A2的单域抗体和抗CD3结构域。Another aspect of the present invention provides a bispecific antibody, the bispecific antibody comprises the above-mentioned anti-CLD18A2 single domain antibody and an anti-CD3 domain.
本发明另一方面提供一种分离的多核苷酸,编码上述的抗CLD18A2的单域抗体、或上述的双特异性抗体。Another aspect of the present invention provides an isolated polynucleotide encoding the above-mentioned anti-CLD18A2 single-domain antibody, or the above-mentioned bispecific antibody.
本发明另一方面提供一种构建体,含有上述的分离的多核苷酸。Another aspect of the present invention provides a construct comprising the isolated polynucleotide described above.
本发明另一方面提供一种抗体的表达系统,所述表达系统含有上述的构建体或基因组中整合有外源的上述的多核苷酸。Another aspect of the present invention provides an antibody expression system, the expression system comprising the above-mentioned construct or the above-mentioned exogenous polynucleotide integrated into the genome.
本发明另一方面提供上述的抗CLD18A2的单域抗体、或上述的双特异性抗体的制备方法,包括如下步骤:在适合表达所述抗体的条件下,培养上述的抗体的表达系统,从而表达出所述的抗体,纯化分离出所述的抗体。Another aspect of the present invention provides a method for preparing the above-mentioned anti-CLD18A2 single-domain antibody or the above-mentioned bispecific antibody, comprising the steps of: culturing the above-mentioned antibody expression system under conditions suitable for expressing the antibody, thereby expressing The antibody is obtained, and the antibody is purified and isolated.
本发明另一方面提供上述的抗CLD18A2的单域抗体、或上述的双特异性抗体在制备药物中的用途,所述药物用于治疗肿瘤。Another aspect of the present invention provides the use of the above-mentioned anti-CLD18A2 single-domain antibody or the above-mentioned bispecific antibody in the preparation of a medicament for treating a tumor.
本发明另一方面提供一种抗体-药物复合物,所述药物复合物包括上述的抗CLD18A2的单域抗体和细胞毒性药物。Another aspect of the present invention provides an antibody-drug complex comprising the above-mentioned anti-CLD18A2 single domain antibody and a cytotoxic drug.
本发明另一方面提供一种细胞,所述细胞含膜结合的嵌合抗原受体,所述嵌合抗原受体包括跨膜域、胞内域和胞外域,所述胞外域包括上述的抗CLD18A2的单域抗体。Another aspect of the present invention provides a cell comprising a membrane-bound chimeric antigen receptor, the chimeric antigen receptor comprising a transmembrane domain, an intracellular domain and an extracellular domain, the extracellular domain comprising the above-mentioned anti- Single domain antibody to CLD18A2.
图1显示为本发明实施例3中T细胞荧光报告体系激活试验结果示意图。FIG. 1 is a schematic diagram showing the results of the activation test of the T cell fluorescent reporter system in Example 3 of the present invention.
图2显示为本发明实施例3中抗CLD18A2/抗CD3双特异抗体体外对NUGC-4-Claudin18.2的杀伤效果示意图。FIG. 2 is a schematic diagram showing the killing effect of anti-CLD18A2/anti-CD3 bispecific antibody on NUGC-4-Claudin18.2 in vitro in Example 3 of the present invention.
图3显示为本发明实施例3中抗CLD18A2/抗CD3双特异抗体的体内肿瘤抑制试验示意图。Figure 3 shows a schematic diagram of the in vivo tumor inhibition test of the anti-CLD18A2/anti-CD3 bispecific antibody in Example 3 of the present invention.
图4显示为本发明实施例5中抗CLDN18A2单域抗体融合蛋白介导的内吞结果示意图。FIG. 4 is a schematic diagram showing the results of endocytosis mediated by anti-CLDN18A2 single-domain antibody fusion protein in Example 5 of the present invention.
图5显示为本发明实施例8中体外细胞毒性测试结果示意图。FIG. 5 is a schematic diagram showing the results of the in vitro cytotoxicity test in Example 8 of the present invention.
图6显示为本发明实施例9中体内毒性测试结果示意图。FIG. 6 is a schematic diagram showing the results of the in vivo toxicity test in Example 9 of the present invention.
图7显示为本发明实施例11中aC18.2-CAR-T细胞作用于靶细胞后的体外细胞因子释放含量检测结果示意图。Fig. 7 is a schematic diagram showing the results of in vitro cytokine release content detection after aC18.2-CAR-T cells act on target cells in Example 11 of the present invention.
图8显示为本发明实施例11中aC18.2-CAR-T细胞在小鼠体内对肿瘤生长的抑制作用示意图。Figure 8 is a schematic diagram showing the inhibitory effect of aC18.2-CAR-T cells on tumor growth in mice in Example 11 of the present invention.
为了使本发明的发明目的、技术方案和有益技术效果更加清晰,以下结合实施例对本发明进行进一步详细说明,熟悉此技术的人士可由本说明书所揭露的内容容易地了解本申请发明的其他优点及功效。In order to make the invention purpose, technical solution and beneficial technical effect of the present invention clearer, the present invention will be described in further detail below in conjunction with the embodiments. Those who are familiar with this technology can easily understand other advantages and other advantages of the present invention from the content disclosed in this specification. effect.
本发明发明人经过大量探索性研究,意外发现了一种抗CLD18A2的单域抗体,所述抗CLD18A2的单域抗体对于Claudin18.2具有良好的亲和力,且可以用于进一步构建双特异性抗体、抗体-药物复合物、T细胞嵌合抗原受体等,从而可以被用于制备具有良好靶向性和治疗效果的药物。After a lot of exploratory research, the inventors of the present invention unexpectedly discovered an anti-CLD18A2 single-domain antibody. The anti-CLD18A2 single-domain antibody has good affinity for Claudin18.2, and can be used to further construct bispecific antibodies, Antibody-drug complexes, T cell chimeric antigen receptors, etc., can be used to prepare drugs with good targeting and therapeutic effects.
本发明第一方面提供一种抗CLD18A2的单域抗体,所述单域抗体通常是指缺失抗体轻链,而只有重链可变区的一类抗体分子,因其分子量小,通常也被称为纳米抗体。所述抗CLD18A2的单域抗体的互补决定区(CDR,complementarity determining region)包括氨基酸序列如SEQ ID No.4~11其中之一所示的CDR-H1、氨基酸序列如SEQ ID No.19~26其中之一所示的CDR-H2、和氨基酸序列如SEQ ID No.33~39其中之一所示的CDR-H3。The first aspect of the present invention provides an anti-CLD18A2 single-domain antibody. The single-domain antibody generally refers to a type of antibody molecule that lacks the light chain of the antibody and only has the variable region of the heavy chain. Because of its small molecular weight, it is also commonly referred to as for nanobodies. The complementarity determining region (CDR, complementarity determining region) of the single-domain antibody of described anti-CLD18A2 includes amino acid sequence such as CDR-H1 shown in one of SEQ ID No.4~11, amino acid sequence such as SEQ ID No.19~26 CDR-H2 shown in one of them, and CDR-H3 whose amino acid sequence is shown in one of SEQ ID Nos. 33 to 39.
在本发明一具体实施例中,所述抗CLD18A2的单域抗体的互补决定区包括氨基酸序列如SEQ ID No.4所示的CDR-H1、氨基酸序列如SEQ ID No.19所示的CDR-H2、和氨基酸序列如SEQ ID No.33所示的CDR-H3。In a specific embodiment of the present invention, the complementarity determining region of the anti-CLD18A2 single domain antibody comprises CDR-H1 whose amino acid sequence is shown in SEQ ID No. 4, and CDR-H1 whose amino acid sequence is shown in SEQ ID No. 19 H2, and CDR-H3 whose amino acid sequence is shown in SEQ ID No.33.
在本发明另一具体实施例中,所述抗CLD18A2的单域抗体的互补决定区包括氨基酸序列如SEQ ID No.5所示的CDR-H1、氨基酸序列如SEQ ID No.20所示的CDR-H2、和氨基酸序列如SEQ ID No.34所示的CDR-H3。In another specific embodiment of the present invention, the complementarity determining region of the anti-CLD18A2 single domain antibody includes CDR-H1 whose amino acid sequence is shown in SEQ ID No.5, and CDR-H1 whose amino acid sequence is shown in SEQ ID No.20 -H2, and CDR-H3 whose amino acid sequence is shown in SEQ ID No.34.
在本发明另一具体实施例中,所述抗CLD18A2的单域抗体的互补决定区包括氨基酸序列如SEQ ID No.6所示的CDR-H1、氨基酸序列如SEQ ID No.21所示的CDR-H2、和氨基酸序列如SEQ ID No.35所示的CDR-H3。In another specific embodiment of the present invention, the complementarity determining region of the anti-CLD18A2 single-domain antibody includes CDR-H1 whose amino acid sequence is shown in SEQ ID No. 6, and CDR-H1 whose amino acid sequence is shown in SEQ ID No. 21 -H2, and CDR-H3 whose amino acid sequence is shown in SEQ ID No.35.
在本发明另一具体实施例中,所述抗CLD18A2的单域抗体的互补决定区包括氨基酸序列如SEQ ID No.7所示的CDR-H1、氨基酸序列如SEQ ID No.22所示的CDR-H2、和氨基酸序列如SEQ ID No.36所示的CDR-H3。In another specific embodiment of the present invention, the complementarity determining region of the anti-CLD18A2 single-domain antibody comprises CDR-H1 whose amino acid sequence is shown in SEQ ID No.7, and CDR-H1 whose amino acid sequence is shown in SEQ ID No.22 -H2, and CDR-H3 whose amino acid sequence is shown in SEQ ID No.36.
在本发明另一具体实施例中,所述抗CLD18A2的单域抗体的互补决定区包括氨基酸序列如SEQ ID No.8所示的CDR-H1、氨基酸序列如SEQ ID No.23所示的CDR-H2、和氨基酸序列如SEQ ID No.37所示的CDR-H3。In another specific embodiment of the present invention, the complementarity determining region of the anti-CLD18A2 single-domain antibody includes CDR-H1 whose amino acid sequence is shown in SEQ ID No. 8, and CDR-H1 whose amino acid sequence is shown in SEQ ID No. 23 -H2, and CDR-H3 whose amino acid sequence is shown in SEQ ID No.37.
在本发明另一具体实施例中,所述抗CLD18A2的单域抗体的互补决定区包括氨基酸序列如SEQ ID No.9所示的CDR-H1、氨基酸序列如SEQ ID No.24所示的CDR-H2、和氨基酸序列如SEQ ID No.38所示的CDR-H3。In another specific embodiment of the present invention, the complementarity determining region of the anti-CLD18A2 single-domain antibody includes CDR-H1 whose amino acid sequence is shown in SEQ ID No. 9, and CDR-H1 whose amino acid sequence is shown in SEQ ID No. 24 -H2, and CDR-H3 whose amino acid sequence is shown in SEQ ID No.38.
在本发明另一具体实施例中,所述抗CLD18A2的单域抗体的互补决定区包括氨基酸序列如SEQ ID No.10所示的CDR-H1、氨基酸序列如SEQ ID No.25所示的CDR-H2、和氨基酸序列如SEQ ID No.36所示的CDR-H3。In another specific embodiment of the present invention, the complementarity determining region of the anti-CLD18A2 single domain antibody comprises CDR-H1 whose amino acid sequence is shown in SEQ ID No.10, and CDR-H1 whose amino acid sequence is shown in SEQ ID No.25 -H2, and CDR-H3 whose amino acid sequence is shown in SEQ ID No.36.
在本发明另一具体实施例中,所述抗CLD18A2的单域抗体的互补决定区包括氨基酸序列如SEQ ID No.11所示的CDR-H1、氨基酸序列如SEQ ID No.26所示的CDR-H2、和氨基酸序列如SEQ ID No.39所示的CDR-H3。In another specific embodiment of the present invention, the complementarity determining region of the anti-CLD18A2 single-domain antibody comprises CDR-H1 whose amino acid sequence is shown in SEQ ID No.11, and CDR-H1 whose amino acid sequence is shown in SEQ ID No.26 -H2, and CDR-H3 whose amino acid sequence is shown in SEQ ID No.39.
本发明所提供的抗CLD18A2的单域抗体中,所述抗CLD18A2的单域抗体还包括框架区(FR,framework region),所述框架区FR包括氨基酸序列如SEQ ID No.1~3其中之一所示的FR1、氨基酸序列如SEQ ID No.12~18其中之一所示的FR2、氨基酸序列如SEQ ID No.27~32其中之一所示的FR3、和氨基酸序列如SEQ ID No.40~41其中之一所示的FR4。In the anti-CLD18A2 single-domain antibody provided by the present invention, the anti-CLD18A2 single-domain antibody further includes a framework region (FR, framework region), and the framework region FR includes an amino acid sequence such as one of SEQ ID No. 1-3 FR1 shown in one, FR2 whose amino acid sequence is shown in one of SEQ ID No. 12 to 18, FR3 whose amino acid sequence is shown in one of SEQ ID No. 27 to 32, and FR3 whose amino acid sequence is shown in SEQ ID No. 27 to 32. FR4 shown in one of 40 to 41.
在本发明一具体实施例中,所述框架区FR包括氨基酸序列如SEQ ID No.1所示的FR1、氨基酸序列如SEQ ID No.12所示的FR2、氨基酸序列如SEQ ID No.27所示的FR3、和氨基酸序列如SEQ ID No.40所示的FR4。In a specific embodiment of the present invention, the framework region FR includes FR1 whose amino acid sequence is shown in SEQ ID No.1, FR2 whose amino acid sequence is shown in SEQ ID No.12, and whose amino acid sequence is shown in SEQ ID No.27 FR3 shown, and FR4 whose amino acid sequence is shown in SEQ ID No. 40.
在本发明另一具体实施例中,所述框架区FR包括氨基酸序列如SEQ ID No.2所示的FR1、氨基酸序列如SEQ ID No.13所示的FR2、氨基酸序列如SEQ ID No.28所示的FR3、和氨基酸序列如SEQ ID No.41所示的FR4。In another specific embodiment of the present invention, the framework region FR includes FR1 whose amino acid sequence is shown in SEQ ID No. 2, FR2 whose amino acid sequence is shown in SEQ ID No. 13, and whose amino acid sequence is shown in SEQ ID No. 28 The FR3 shown, and the amino acid sequence of FR4 shown in SEQ ID No. 41.
在本发明另一具体实施例中,所述框架区FR包括氨基酸序列如SEQ ID No.3所示的FR1、氨基酸序列如SEQ ID No.14所示的FR2、氨基酸序列如SEQ ID No.29所示的FR3、和氨基酸序列如SEQ ID No.41所示的FR4。In another specific embodiment of the present invention, the framework region FR includes FR1 whose amino acid sequence is shown in SEQ ID No.3, FR2 whose amino acid sequence is shown in SEQ ID No.14, and FR2 whose amino acid sequence is shown in SEQ ID No.29 The FR3 shown, and the amino acid sequence of FR4 shown in SEQ ID No. 41.
在本发明另一具体实施例中,所述框架区FR包括氨基酸序列如SEQ ID No.1所示的FR1、氨基酸序列如SEQ ID No.15所示的FR2、氨基酸序列如SEQ ID No.30所示的FR3、和氨基酸序列如SEQ ID No.41所示的FR4。In another specific embodiment of the present invention, the framework region FR includes FR1 whose amino acid sequence is shown in SEQ ID No. 1, FR2 whose amino acid sequence is shown in SEQ ID No. 15, and FR2 whose amino acid sequence is shown in SEQ ID No. 30 The FR3 shown, and the amino acid sequence of FR4 shown in SEQ ID No. 41.
在本发明另一具体实施例中,所述框架区FR包括氨基酸序列如SEQ ID No.2所示的FR1、氨基酸序列如SEQ ID No.16所示的FR2、氨基酸序列如SEQ ID No.31所示的FR3、和氨基酸序列如SEQ ID No.41所示的FR4。In another specific embodiment of the present invention, the framework region FR includes FR1 whose amino acid sequence is shown in SEQ ID No.2, FR2 whose amino acid sequence is shown in SEQ ID No.16, and whose amino acid sequence is shown in SEQ ID No.31 The FR3 shown, and the amino acid sequence of FR4 shown in SEQ ID No. 41.
在本发明另一具体实施例中,所述框架区FR包括氨基酸序列如SEQ ID No.2所示的FR1、氨基酸序列如SEQ ID No.13所示的FR2、氨基酸序列如SEQ ID No.31所示的FR3、和氨基酸序列如SEQ ID No.41所示的FR4。In another specific embodiment of the present invention, the FR in the framework region includes FR1 whose amino acid sequence is shown in SEQ ID No.2, FR2 whose amino acid sequence is shown in SEQ ID No.13, and whose amino acid sequence is shown in SEQ ID No.31 The FR3 shown, and the amino acid sequence of FR4 shown in SEQ ID No. 41.
在本发明另一具体实施例中,所述框架区FR包括氨基酸序列如SEQ ID No.1所示的FR1、氨基酸序列如SEQ ID No.17所示的FR2、氨基酸序列如SEQ ID No.30所示的FR3、和氨基酸序列如SEQ ID No.41所示的FR4。In another specific embodiment of the present invention, the framework region FR includes FR1 whose amino acid sequence is shown in SEQ ID No.1, FR2 whose amino acid sequence is shown in SEQ ID No.17, and FR2 whose amino acid sequence is shown in SEQ ID No.30 The FR3 shown, and the amino acid sequence of FR4 shown in SEQ ID No. 41.
在本发明另一具体实施例中,所述框架区FR包括氨基酸序列如SEQ ID No.2所示的FR1、氨基酸序列如SEQ ID No.18所示的FR2、氨基酸序列如SEQ ID No.32所示的FR3、 和氨基酸序列如SEQ ID No.41所示的FR4。In another specific embodiment of the present invention, the framework region FR includes FR1 whose amino acid sequence is shown in SEQ ID No. 2, FR2 whose amino acid sequence is shown in SEQ ID No. 18, and FR2 whose amino acid sequence is shown in SEQ ID No. 32 The shown FR3, and amino acid sequences are FR4 shown in SEQ ID No. 41.
本发明所提供的抗CLD18A2的单域抗体中,所述抗CLD18A2的单域抗体的氨基酸序列可以包括:a)如SEQ ID No.42~49其中之一所示的氨基酸序列;或,b)与SEQ ID No.42~49其中之一所示的氨基酸序列具有80%以上序列一致性、且具有a)所限定的氨基酸序列功能的氨基酸序列。具体的,所述b)中的氨基酸序列具体指:如SEQ ID No.42~49其中之一所示的氨基酸序列经过取代、缺失或者添加一个或多个(具体可以是1-50、1-30个、1-20个、1-10个、1-5个、或1-3个)氨基酸而得到的,或者在N-末端和/或C-末端添加一个或多个(具体可以是1-50个、1-30个、1-20个、1-10个、1-5个、或1-3个)氨基酸而得到的,且具有氨基酸如SEQ ID No.42~49其中之一所示的多肽片段的功能的多肽片段,例如,可以是与CLD18A2的特异性结合能力,从而可以与表达CLD18A2的细胞特异性结合,而不与只表达CLD18A1的细胞结合,亦即只识别CLD18A2而不识别CLD18A1。所述b)中的氨基酸序列可以与SEQ ID No.42~49其中之一具有80%、85%、90%、93%、95%、97%、或99%以上的一致性。In the anti-CLD18A2 single-domain antibody provided by the present invention, the amino acid sequence of the anti-CLD18A2 single-domain antibody may include: a) the amino acid sequence shown in one of SEQ ID Nos. 42 to 49; or, b) An amino acid sequence that has more than 80% sequence identity with the amino acid sequence shown in one of SEQ ID Nos. 42 to 49, and has the function of the amino acid sequence defined in a). Specifically, the amino acid sequence in b) specifically refers to: the amino acid sequence shown in one of SEQ ID No. 42 to 49 is substituted, deleted or added one or more (specifically, 1-50, 1- 30, 1-20, 1-10, 1-5, or 1-3) amino acids, or by adding one or more (specifically, 1) to the N-terminal and/or C-terminal -50, 1-30, 1-20, 1-10, 1-5, or 1-3) amino acids, and have amino acids as one of SEQ ID No. 42-49 Polypeptide fragments with the functions of the indicated polypeptide fragments, for example, may have specific binding ability to CLD18A2, so that they can specifically bind to cells expressing CLD18A2, but not to cells expressing only CLD18A1, that is, only recognize CLD18A2 and not Identify CLD18A1. The amino acid sequence in b) can be more than 80%, 85%, 90%, 93%, 95%, 97%, or 99% identical to one of SEQ ID Nos. 42-49.
本文中,序列一致性(sequence identity)指参与对比的序列中相同残基的百分比。可采用本领域周知的计算软件计算两条或多条目的序列的序列一致性,这些软件可获自如NCBI。As used herein, sequence identity refers to the percentage of identical residues in the sequences participating in the alignment. Sequence identity of two or more entry sequences can be calculated using computational software well known in the art, such software available from NCBI, for example.
本发明所提供的抗CLD18A2的单域抗体可以来源于羊驼(Vicugna pacos),其整体分子量可以是ScFv单链抗体的二分之一左右,因此可以有效降低整体结构的分子量,从而增强其组织穿透性,更有效的到达靶组织和器官,提高治疗效果,且这种结构比起具有两个ScFv串联的结构更便于制备。The anti-CLD18A2 single-domain antibody provided by the present invention can be derived from alpaca (Vicugna pacos), and its overall molecular weight can be about half that of ScFv single-chain antibody, so it can effectively reduce the molecular weight of the overall structure, thereby enhancing its organization. Penetration, reaching target tissues and organs more effectively, improving the therapeutic effect, and this structure is more convenient to prepare than the structure with two ScFv in series.
本发明所提供的抗CLD18A2的单域抗体,通常可以是人源化抗体。所述抗CLD18A2的单域抗体的氨基酸序列可以包括:c)如SEQ ID No.67~90其中之一所示的氨基酸序列;或,d)与SEQ ID No.67~90其中之一所示的氨基酸序列具有80%以上序列一致性、且具有c)所限定的氨基酸序列功能的氨基酸序列。具体的,所述d)中的氨基酸序列具体指:如SEQ ID No.67~90其中之一所示的氨基酸序列经过取代、缺失或者添加一个或多个(具体可以是1-50、1-30个、1-20个、1-10个、1-5个、或1-3个)氨基酸而得到的,或者在N-末端和/或C-末端添加一个或多个(具体可以是1-50个、1-30个、1-20个、1-10个、1-5个、或1-3个)氨基酸而得到的,且具有氨基酸如SEQ ID No.67~90其中之一所示的多肽片段的功能的多肽片段,例如,可以是具有相对更高的人源化程度。本发明对抗CLD18.2单域抗体人源化主要针对框架区进行改造,例如采用CDR移植的方法,将合适的来源于人的抗体的FR区替换原来的序列,同时保留原始抗体的CDR区。其中序列SEQ ID No.75~90是在原始序列SEQ ID No.42~49基础上对各自FR1、FR2、FR3、FR4人源化改造得到;其中序列SEQ ID No.67~74是在原始序列SEQ ID No.42~49基础上对各自相应的FR1、FR3、FR4人源化改造得到,FR2则保留各自相应的原始序列。所述d)中的氨基酸序列可以与SEQ ID No.67~90其中之一具有80%、85%、90%、93%、95%、97%、或99%以上的一致性。The anti-CLD18A2 single domain antibody provided by the present invention can usually be a humanized antibody. The amino acid sequence of the anti-CLD18A2 single-domain antibody can include: c) the amino acid sequence shown in one of SEQ ID No. 67-90; or, d) and the amino acid sequence shown in one of SEQ ID No. 67-90 The amino acid sequence has more than 80% sequence identity and has the amino acid sequence function defined by c). Specifically, the amino acid sequence in d) specifically refers to: the amino acid sequence shown in one of SEQ ID No. 67-90 is substituted, deleted or added with one or more (specifically, 1-50, 1- 30, 1-20, 1-10, 1-5, or 1-3) amino acids, or by adding one or more (specifically, 1) to the N-terminal and/or C-terminal -50, 1-30, 1-20, 1-10, 1-5, or 1-3) amino acids, and have amino acids as one of SEQ ID No. 67-90 Polypeptide fragments that exhibit the function of the polypeptide fragments shown, for example, may be of a relatively higher degree of humanization. The humanization of the anti-CLD18.2 single-domain antibody of the present invention is mainly aimed at transforming the framework region, for example, by adopting the method of CDR transplantation, the FR region of a suitable human-derived antibody is replaced with the original sequence, while the CDR region of the original antibody is retained. The sequences SEQ ID No. 75-90 are obtained by humanizing FR1, FR2, FR3, and FR4 based on the original sequences SEQ ID No. 42-49; the sequences SEQ ID No. 67-74 are obtained from the original sequences Based on SEQ ID Nos. 42 to 49, the corresponding FR1, FR3, and FR4 are obtained by humanization transformation, while FR2 retains the corresponding original sequence. The amino acid sequence in d) can be more than 80%, 85%, 90%, 93%, 95%, 97%, or 99% identical to one of SEQ ID Nos. 67-90.
本发明第二方面提供一种双特异性抗体,双特异性抗体包括本发明第一方面所提供的抗CLD18A2的单域抗体和抗CD3结构域。所述双特异抗体可以同时靶向细胞表面抗原CLD18A2以及T细胞表面触发分子CD3,从而可以激活T细胞,导致靶细胞被杀伤的效果。The second aspect of the present invention provides a bispecific antibody, and the bispecific antibody includes the anti-CLD18A2 single domain antibody and the anti-CD3 domain provided in the first aspect of the present invention. The bispecific antibody can simultaneously target the cell surface antigen CLD18A2 and the T cell surface triggering molecule CD3, thereby activating the T cells, resulting in the effect of killing the target cells.
本发明所提供的双特异性抗体中,可以包括抗CD3结构域。抗CD3结构域其结构形式可以是包括但不限于单链抗体(scFv)和抗体Fab片段等。所述抗CD3结构域可以与T细胞表面触发分子CD3的特异性结合,从而可以激活T细胞,以增强靶细胞被杀伤的效果。所述抗CD3结构域的重链可变区的氨基酸序列可以包括:e)如SEQ ID NO.91、SEQ ID NO.93、SEQ ID NO.95、SEQ ID NO.97、SEQ ID NO.99、或SEQ ID NO.101其中之一所示的氨基酸序列;或,f)与SEQ ID NO.91、SEQ ID NO.93、SEQ ID NO.95、SEQ ID NO.97、SEQ ID NO.99、或SEQ ID NO.101其中之一所示的氨基酸序列具有80%以上序列一致性、且具有e)所限定的氨基酸序列功能的氨基酸序列。具体的,所述f)中的氨基酸序列具体指:如SEQ ID NO.91、SEQ ID NO.93、SEQ ID NO.95、SEQ ID NO.97、SEQ ID NO.99、或SEQ ID NO.101其中之一所示的氨基酸序列经过取代、缺失或者添加一个或多个(具体可以是1-50、1-30个、1-20个、1-10个、1-5个、或1-3个)氨基酸而得到的,或者在N-末端和/或C-末端添加一个或多个(具体可以是1-50个、1-30个、1-20个、1-10个、1-5个、或1-3个)氨基酸而得到的,且具有氨基酸如SEQ ID NO.91、SEQ ID NO.93、SEQ ID NO.95、SEQ ID NO.97、SEQ ID NO.99、或SEQ ID NO.101其中之一所示的多肽片段的功能的多肽片段。所述f)中的氨基酸序列可以与SEQ ID NO.91、SEQ ID NO.93、SEQ ID NO.95、SEQ ID NO.97、SEQ ID NO.99、或SEQ ID NO.101其中之一具有80%、85%、90%、93%、95%、97%、或99%以上的一致性。所述抗CD3结构域的轻链可变区的氨基酸序列可以包括:g)如SEQ ID NO.92、SEQ ID NO.94、SEQ ID NO.96、SEQ ID NO.98、SEQ ID NO.100和、或SEQ ID NO.102其中之一所示的氨基酸序列;或,h)与SEQ ID NO.92、SEQ ID NO.94、SEQ ID NO.96、SEQ ID NO.98、SEQ ID NO.100、或SEQ ID NO.102其中之一所示的氨基酸序列具有80%以上序列一致性、且具有g)所限定的氨基酸序列功能的氨基酸序列。具体的,所述h)中的氨基酸序列具体指:如SEQ ID NO.92、SEQ ID NO.94、SEQ ID NO.96、SEQ ID NO.98、SEQ ID NO.100、或SEQ ID NO.102其中之一所示的氨基酸序列 经过取代、缺失或者添加一个或多个(具体可以是1-50、1-30个、1-20个、1-10个、1-5个、或1-3个)氨基酸而得到的,或者在N-末端和/或C-末端添加一个或多个(具体可以是1-50个、1-30个、1-20个、1-10个、1-5个、或1-3个)氨基酸而得到的,且具有氨基酸如SEQ ID NO.92、SEQ ID NO.94、SEQ ID NO.96、SEQ ID NO.98、SEQ ID NO.100、或SEQ ID NO.102其中之一所示的多肽片段的功能的多肽片段。所述h)中的氨基酸序列可以与SEQ ID NO.92、SEQ ID NO.94、SEQ ID NO.96、SEQ ID NO.98、SEQ ID NO.100、或SEQ ID NO.102其中之一具有80%、85%、90%、93%、95%、97%、或99%以上的一致性。在本发明一具体实施例中,抗CD3结构域的重链可变区序列可以为SEQ ID NO.95,抗CD3结构域的轻链可变区序列可以为SEQ ID NO.96,最终产生的ScFv形式的序列如SEQ ID NO.103、或SEQ ID NO.104所示。在本发明另一具体实施例中,抗CD3结构域的重链可变区序列可以为SEQ ID NO.93,抗CD3结构域的轻链可变区序列可以为SEQ ID NO.94,最终产生的ScFv形式的序列如SEQ ID NO.105、或SEQ ID NO.106所示。所述CD3可以是人源、非人灵长类(如猴)来源和/或鼠源。In the bispecific antibody provided by the present invention, an anti-CD3 domain may be included. The structural form of the anti-CD3 domain can include, but is not limited to, single-chain antibody (scFv), antibody Fab fragments, and the like. The anti-CD3 domain can specifically bind to the triggering molecule CD3 on the surface of T cells, so that T cells can be activated to enhance the effect of killing target cells. The amino acid sequence of the heavy chain variable region of the anti-CD3 domain may include: e) as SEQ ID NO.91, SEQ ID NO.93, SEQ ID NO.95, SEQ ID NO.97, SEQ ID NO.99 , or the amino acid sequence shown in one of SEQ ID NO.101; or, f) with SEQ ID NO.91, SEQ ID NO.93, SEQ ID NO.95, SEQ ID NO.97, SEQ ID NO.99 , or the amino acid sequence shown in one of SEQ ID NO.101 has more than 80% sequence identity, and has the amino acid sequence function of the amino acid sequence defined in e). Specifically, the amino acid sequence in the f) specifically refers to: such as SEQ ID NO.91, SEQ ID NO.93, SEQ ID NO.95, SEQ ID NO.97, SEQ ID NO.99, or SEQ ID NO.99. The amino acid sequence shown in one of 101 has undergone substitution, deletion or addition of one or more (specifically, 1-50, 1-30, 1-20, 1-10, 1-5, or 1- 3) amino acids, or adding one or more (specifically 1-50, 1-30, 1-20, 1-10, 1- 5, or 1-3) amino acids, and have amino acids such as SEQ ID NO.91, SEQ ID NO.93, SEQ ID NO.95, SEQ ID NO.97, SEQ ID NO.99, or SEQ ID NO.99 A functional polypeptide fragment of one of the polypeptide fragments shown in ID NO. 101. The amino acid sequence in said f) can have one of SEQ ID NO.91, SEQ ID NO.93, SEQ ID NO.95, SEQ ID NO.97, SEQ ID NO.99, or SEQ ID NO.101 Concordance above 80%, 85%, 90%, 93%, 95%, 97%, or 99%. The amino acid sequence of the light chain variable region of the anti-CD3 domain may include: g) as SEQ ID NO.92, SEQ ID NO.94, SEQ ID NO.96, SEQ ID NO.98, SEQ ID NO.100 and, or the amino acid sequence shown in one of SEQ ID NO.102; or, h) with SEQ ID NO.92, SEQ ID NO.94, SEQ ID NO.96, SEQ ID NO.98, SEQ ID NO. 100, or the amino acid sequence of one of the amino acid sequences shown in SEQ ID NO. 102 with more than 80% sequence identity and having the amino acid sequence function defined in g). Specifically, the amino acid sequence in the h) specifically refers to: such as SEQ ID NO.92, SEQ ID NO.94, SEQ ID NO.96, SEQ ID NO.98, SEQ ID NO.100, or SEQ ID NO. The amino acid sequence shown in one of 102 has been substituted, deleted or added one or more (specifically can be 1-50, 1-30, 1-20, 1-10, 1-5, or 1- 3) amino acids, or adding one or more (specifically 1-50, 1-30, 1-20, 1-10, 1- 5, or 1-3) amino acids, and have amino acids such as SEQ ID NO.92, SEQ ID NO.94, SEQ ID NO.96, SEQ ID NO.98, SEQ ID NO.100, or SEQ ID NO. A functional polypeptide fragment of one of the polypeptide fragments shown in ID NO. 102. The amino acid sequence in h) can have one of SEQ ID NO.92, SEQ ID NO.94, SEQ ID NO.96, SEQ ID NO.98, SEQ ID NO.100, or SEQ ID NO.102 Concordance above 80%, 85%, 90%, 93%, 95%, 97%, or 99%. In a specific embodiment of the present invention, the heavy chain variable region sequence of the anti-CD3 structural domain can be SEQ ID NO.95, and the light chain variable region sequence of the anti-CD3 structural domain can be SEQ ID NO.96. The sequence of the ScFv form is shown in SEQ ID NO.103, or SEQ ID NO.104. In another specific embodiment of the present invention, the sequence of the heavy chain variable region of the anti-CD3 domain can be SEQ ID NO.93, and the sequence of the light chain variable region of the anti-CD3 domain can be SEQ ID NO.94. The sequence of the ScFv form is shown in SEQ ID NO.105, or SEQ ID NO.106. The CD3 may be of human origin, non-human primate (eg, monkey) origin, and/or murine origin.
本发明所提供的双特异性抗体中,双特异性抗体还可以包括用于延长血清半衰期的结构域。用于延长血清半衰期的结构域可以选自哺乳动物IgG的Fc结构域、白蛋白、结合白蛋白的结构域(ABD)或者聚乙二醇(PEG)等。用于延长血清半衰期的结构域具体可以包括:i)如SEQ ID No.107、SEQ ID No.108所示的氨基酸序列的组合,或SEQ ID No.156;或,j)与SEQ ID No.107、SEQ ID No.108,或SEQ ID No.156其中之一所示的氨基酸序列具有80%以上序列一致性、且具有i)所限定的氨基酸序列功能的氨基酸序列。具体的,所述j)中的氨基酸序列具体指:如SEQ ID No.107、SEQ ID No.108,或SEQ ID No.156其中之一所示的氨基酸序列经过取代、缺失或者添加一个或多个(具体可以是1-50、1-30个、1-20个、1-10个、1-5个、或1-3个)氨基酸而得到的,或者在N-末端和/或C-末端添加一个或多个(具体可以是1-50个、1-30个、1-20个、1-10个、1-5个、或1-3个)氨基酸而得到的,且具有氨基酸如SEQ ID No.107、SEQ ID No.108,或SEQ ID No.156其中之一所示的多肽片段的功能的多肽片段,例如,可以是能够延长双特异性抗体的血清半衰期。所述j)中的氨基酸序列可以与SEQ ID No.107、SEQ ID No.108,或SEQ ID No.156其中之一具有80%、85%、90%、93%、95%、97%、或99%以上的一致性。Among the bispecific antibodies provided by the present invention, the bispecific antibodies may also include a domain for prolonging serum half-life. The domain for prolonging serum half-life can be selected from the Fc domain of mammalian IgG, albumin, albumin binding domain (ABD) or polyethylene glycol (PEG) and the like. The domain for prolonging serum half-life can specifically include: i) a combination of amino acid sequences shown in SEQ ID No. 107, SEQ ID No. 108, or SEQ ID No. 156; or, j) and SEQ ID No. 107. The amino acid sequence shown in one of SEQ ID No. 108, or SEQ ID No. 156 has more than 80% sequence identity and has the amino acid sequence function as defined in i). Specifically, the amino acid sequence in j) specifically refers to: the amino acid sequence shown in one of SEQ ID No. 107, SEQ ID No. 108, or SEQ ID No. 156 is substituted, deleted, or added with one or more (specifically can be 1-50, 1-30, 1-20, 1-10, 1-5, or 1-3) amino acids, or at the N-terminal and/or C- It is obtained by adding one or more (specifically, 1-50, 1-30, 1-20, 1-10, 1-5, or 1-3) amino acids to the end, and has amino acids such as The functional polypeptide fragment of one of SEQ ID No. 107, SEQ ID No. 108, or SEQ ID No. 156 may, for example, be capable of extending the serum half-life of a bispecific antibody. The amino acid sequence in said j) can have 80%, 85%, 90%, 93%, 95%, 97%, 80%, 85%, 90%, 93%, 95%, 97%, SEQ ID No. or more than 99% consistency.
本发明所提供的双特异性抗体中,所述抗CLD18A2的单域抗体、抗CD3结构域和用于延长血清半衰期的结构域之间还包括连接肽。双特异性抗体中,可以包括多个连接肽片段,例如,抗CLD18A2的单域抗体、抗CD3结构域之间可以包括连接肽,再例如,抗CD3结构 域、用于延长血清半衰期的结构域之间可以包括连接肽。连接肽片段通常可以为一段长度合适的由甘氨酸(G)和/或丝氨酸(S)和/或丙氨酸(A)和/或苏氨酸(T)构成的柔性多肽,能够保持双特异抗体分子各结构域的正确折叠,以及相互的柔韧性,例如,所述连接肽片段的氨基酸序列可以包括如(GS)n、(GGS)n、(GGSG)n、(GGGS)nA、(GGGGS)nA、(GGGGA)nA、(GGGGG)nA等序列,其中,n选自1-10之间的整数。在本发明一具体实施例中,连接肽的氨基酸序列可以包括:GGGGSGGGS(SEQ ID NO.157)、GGGGGGSGGSGGSGGSGG(SEQ ID NO.158)、GGGGSGGGGSGGGGS(SEQ ID NO.159)等。In the bispecific antibody provided by the present invention, the anti-CLD18A2 single-domain antibody, the anti-CD3 domain and the domain for prolonging serum half-life further include a linker peptide. In the bispecific antibody, a plurality of linker peptide fragments can be included, for example, an anti-CLD18A2 single-domain antibody, a linker peptide can be included between the anti-CD3 domains, for example, an anti-CD3 domain, a domain for prolonging serum half-life Linking peptides may be included in between. The connecting peptide fragment can usually be a flexible polypeptide of suitable length composed of glycine (G) and/or serine (S) and/or alanine (A) and/or threonine (T), which can maintain the bispecific antibody Correct folding of each domain of the molecule, and mutual flexibility, for example, the amino acid sequence of the linker peptide fragment may include, for example, (GS)n, (GGS)n, (GGSG)n, (GGGS)nA, (GGGGS) Sequences of nA, (GGGGA)nA, (GGGGG)nA, etc., wherein n is selected from an integer between 1-10. In a specific embodiment of the present invention, the amino acid sequence of the connecting peptide may include: GGGGSGGGS (SEQ ID NO. 157), GGGGGGSGGSGGSGGSGG (SEQ ID NO. 158), GGGGSGGGGSGGGGS (SEQ ID NO. 159) and the like.
本发明所提供的双特异性抗体,其结构可以是如下之一:The bispecific antibody provided by the present invention, its structure can be one of the following:
(a)S1结构:抗CLD18A2单域抗体(V HH/CLD18A2)的C端通过连接肽与抗CD3单链抗体(scFv CD3)连接。例如,双特异性抗体自N端至C端可以依次包括抗CLD18A2单域抗体、连接肽、抗CD3单链抗体(即V HH/CLD18A2-连接肽-scFv CD3的结构),再例如,抗CD3单链抗体自N端至C端可以依次包括其重链可变区片段、连接肽、轻链可变区片段(即V H-连接肽-V L的结构),或依次包括其轻链可变区片段、连接肽、重链可变区片段(即V L-连接肽-V H的结构)。 (a) S1 structure: the C-terminus of anti-CLD18A2 single-domain antibody (V HH/CLD18A2 ) was linked to anti-CD3 single-chain antibody (scFv CD3 ) through a linking peptide. For example, the bispecific antibody can include, from N-terminal to C-terminal, an anti-CLD18A2 single domain antibody, a linking peptide, an anti-CD3 single-chain antibody (ie , the structure of VHH/CLD18A2 -linking peptide-scFv CD3 ), and, for example, anti-CD3 From the N-terminal to the C-terminal, the single-chain antibody may sequentially include its heavy chain variable region fragment, connecting peptide, light chain variable region fragment (ie , the structure of VH -connecting peptide- VL ), or sequentially include its light chain. variable region fragment, connecting peptide, a heavy chain variable region fragment (i.e., V L - linker peptide structure -V H).
(b)S2结构:抗CLD18A2单域抗体(V HH/CLD18A2)的C端通过连接肽与白蛋白结合结构域(ABD)连接,然后通过连接肽与抗CD3单链抗体(scFv CD3)连接。例如,双特异性抗体自N端至C端可以依次包括抗CLD18A2单域抗体、连接肽、ABD、连接肽、抗CD3单链抗体(即V HH/CLD18A2-连接肽-ABD-连接肽-scFv CD3的结构),再例如,抗CD3单链抗体自N端至C端可以依次包括其重链可变区片段、连接肽、轻链可变区片段(即V H-连接肽-V L的结构),或依次包括其轻链可变区片段、连接肽、重链可变区片段(即V L-连接肽-V H的结构)。 (b) S2 structure: the C-terminus of the anti-CLD18A2 single-domain antibody ( VHH/CLD18A2 ) was linked to the albumin-binding domain (ABD) through a linker peptide, and then to the anti-CD3 single-chain antibody (scFv CD3 ) through the linker peptide. For example, a bispecific antibody can include, from N-terminal to C-terminal, anti-CLD18A2 single domain antibody, linker peptide, ABD, linker peptide, anti-CD3 single chain antibody (ie VHH/CLD18A2 -linker peptide-ABD-linker peptide-scFv The structure of CD3 ), for another example, the anti-CD3 single-chain antibody can sequentially include its heavy chain variable region fragment, connecting peptide, light chain variable region fragment (ie, VH -connecting peptide- VL ) from the N-terminus to the C-terminus. structure), or sequentially including its light chain variable region fragment, connecting peptide, and heavy chain variable region fragment (ie, the structure of VL -connecting peptide- VH).
(c)S3结构:抗CLD18A2单域抗体(V HH/CLD18A2)的C端通过连接肽与抗CD3单链抗体(scFv CD3)连接,然后通过连接肽与白蛋白结合结构域(ABD)连接。例如,双特异性抗体自N端至C端可以依次包括抗CLD18A2单域抗体、连接肽、抗CD3单链抗体、连接肽、ABD(即V HH/CLD18A2-连接肽-scFv CD3-连接肽-ABD的结构),再例如,抗CD3单链抗体自N端至C端可以依次包括其重链可变区片段、连接肽、轻链可变区片段(即V H-连接肽-V L的结构),或依次包括其轻链可变区片段、连接肽、重链可变区片段(即V L-连接肽-V H的结构)。 (c) S3 structure: anti CLD18A2 single domain antibodies (V HH / CLD18A2) connected via the C-terminus of the peptide to anti-CD3 single-chain antibody (the scFv CD3) is connected, and then connected binding domain (ABD) to albumin via a linker peptide. For example, a bispecific antibody may include, in order from N-terminus to C-terminus, an anti-CLD18A2 single domain antibody, a linker peptide, an anti-CD3 single chain antibody, a linker peptide, an ABD (ie, VHH/CLD18A2 -linker peptide-scFv CD3 -linker peptide- The structure of ABD), for another example, the anti-CD3 single-chain antibody can sequentially include its heavy chain variable region fragment, connecting peptide, and light chain variable region fragment from the N-terminus to the C-terminus (ie, the VH -connecting peptide- VL) . structure), or sequentially including its light chain variable region fragment, connecting peptide, and heavy chain variable region fragment (ie, the structure of VL -connecting peptide- VH).
(d)S4结构:抗CLD18A2单域抗体(V HH/CLD18A2)、抗CD3结构域与IgG Fc片段组成的异二聚体结构。双特异抗体的S4结构中IgG1Fc变体的其中一个的氨基酸序列可以包括SEQ ID NO.107,另一个的氨基酸序列可以包括SEQ ID NO.108,两种变体CH3结构域经过修饰后形成非对称杵臼结构,并形成稳固异二聚体结构。同时,对Fc进行了突变,比如L234A和L235A突变,其目的在于降低Fc的一种生物学效应,即降低Fc与NK细胞上FcγRIII的亲和力,从而可以避免NK细胞引起的可能的对T细胞的杀伤。 (d) S4 structure: a heterodimer structure composed of anti-CLD18A2 single-domain antibody (V HH/CLD18A2 ), anti-CD3 domain and IgG Fc fragment. The amino acid sequence of one of the IgG1 Fc variants in the S4 structure of the bispecific antibody may include SEQ ID NO. 107, and the amino acid sequence of the other may include SEQ ID NO. 108. The CH3 domains of the two variants are modified to form asymmetrical pestle-hole structure and form a stable heterodimeric structure. At the same time, Fc has been mutated, such as L234A and L235A mutations, the purpose is to reduce a biological effect of Fc, that is, to reduce the affinity of Fc with FcγRIII on NK cells, so as to avoid possible NK cells. kill.
在本发明一具体实施例中,双特异性抗体的氨基酸序列可以包括SEQ ID NO.109-118其中之一所示的序列,其中SEQ ID NO.109-114是通过能结合人血白蛋白的单域抗体ABD(SEQ ID NO.156)结构来延长药物半衰期,而SEQ ID NO.115和SEQ ID NO.116形成异二聚体,通过Fc延长半衰期,SEQ ID NO.117和SEQ ID NO.118形成异二聚体,通过Fc延长半衰期。In a specific embodiment of the present invention, the amino acid sequence of the bispecific antibody may include the sequence shown in one of SEQ ID NO. 109-118, wherein SEQ ID NO. Single domain antibody ABD (SEQ ID NO.156) structure to prolong drug half-life, while SEQ ID NO.115 and SEQ ID NO.116 form a heterodimer to prolong half-life through Fc, SEQ ID NO.117 and SEQ ID NO. 118 forms heterodimers, extending half-life by Fc.
本发明第三方面提供一种分离的多核苷酸,编码本发明第一方面所提供的抗CLD18A2的单域抗体、或本发明第二方面所提供的双特异性抗体。The third aspect of the present invention provides an isolated polynucleotide encoding the anti-CLD18A2 single domain antibody provided by the first aspect of the present invention, or the bispecific antibody provided by the second aspect of the present invention.
本发明第四方面提供一种构建体,含有本发明第三方面所提供的分离的多核苷酸。所述构建体含有本发明第二方面所提供的分离的多核苷酸。所述构建体通常可以通过将所述分离的多核苷酸插入合适的载体中构建获得,本领域技术人员可选择合适的表达载体。例如,所述载体的类型可以是包括但不限于质粒、噬菌粒、噬菌体衍生物、动物病毒和粘粒等。再例如,所述载体可以是表达载体,也可以是克隆载体。The fourth aspect of the present invention provides a construct comprising the isolated polynucleotide provided by the third aspect of the present invention. The construct contains the isolated polynucleotide provided by the second aspect of the present invention. The construct can generally be constructed by inserting the isolated polynucleotide into a suitable vector, and those skilled in the art can select a suitable expression vector. For example, the type of vector may include, but is not limited to, plasmids, phagemids, phage derivatives, animal viruses, cosmids, and the like. For another example, the vector may be an expression vector or a cloning vector.
本发明第五方面提供一种抗体的表达系统,所述表达系统含有本发明第四方面所提供的构建体或基因组中整合有外源的本发明第三方面所提供的多核苷酸,从而可表达所述的抗CLD18A2的单域抗体、或双特异性抗体。所述表达系统可以是宿主细胞,所述宿主细胞可以是原核细胞,如细菌细胞;或是低等真核细胞,如酵母细胞;丝状真菌细胞、或是高等真核细胞,如哺乳动物细胞。代表性例子有:大肠杆菌,链霉菌属;鼠伤寒沙门氏菌的细菌细胞;真菌细胞如酵母、丝状真菌、植物细胞;果蝇S2或Sf9的昆虫细胞;CHO、COS、293细胞、或Bowes黑素瘤细胞的动物细胞等。将构建体导入宿主细胞的方法对于本领域技术人员来说应该是已知的,例如,可以显微注射法、基因枪法、电穿孔法、病毒介导的转化法、电子轰击法、磷酸钙沉淀法等方法。The fifth aspect of the present invention provides an antibody expression system, the expression system contains the construct provided by the fourth aspect of the present invention or the exogenous polynucleotide provided by the third aspect of the present invention is integrated into the genome, so that it can be Express the single-domain antibody or bispecific antibody of the anti-CLD18A2. The expression system can be a host cell, which can be a prokaryotic cell, such as a bacterial cell; or a lower eukaryotic cell, such as a yeast cell; a filamentous fungal cell, or a higher eukaryotic cell, such as a mammalian cell . Representative examples are: Escherichia coli, Streptomyces; bacterial cells of Salmonella typhimurium; fungal cells such as yeast, filamentous fungi, plant cells; insect cells of Drosophila S2 or Sf9; CHO, COS, 293 cells, or Bowes black melanoma cells of animal cells, etc. Methods for introducing constructs into host cells should be known to those skilled in the art, for example, microinjection, biolistic, electroporation, virus-mediated transformation, electron bombardment, calcium phosphate precipitation can be used method, etc.
在本发明的一具体实施例中,所述质粒可以是pPIC9K。In a specific embodiment of the present invention, the plasmid may be pPIC9K.
在本发明的另一具体实施例中,所述表达系统是以毕赤酵母细胞为宿主。酵母在用于表达外源性蛋白时具有独特优势,比如酵母具有一套完整的真核表达系统,可以实现抗体结构的正确折叠和翻译后修饰;酵母还可以实现高密度发酵,实现外源蛋白的高表达和获得高的单位产量。In another specific embodiment of the present invention, the expression system uses Pichia pastoris cells as a host. Yeast has unique advantages in expressing exogenous proteins. For example, yeast has a complete eukaryotic expression system, which can realize the correct folding and post-translational modification of antibody structure; yeast can also realize high-density fermentation to realize exogenous protein. high expression and obtain high unit yield.
在本发明的另一具体实施例中,所述质粒含有DHFR(二氢叶酸还原酶)基因或GS(谷氨酰胺合成酶)基因,该类基因既是一种选择标记,也是通过基因共扩增使得引入的目的基因在宿主中的基因拷贝数得到提高,产物表达量也会因此提高。In another specific embodiment of the present invention, the plasmid contains a DHFR (dihydrofolate reductase) gene or a GS (glutamine synthase) gene, which is both a selection marker and a gene co-amplification. The gene copy number of the introduced target gene in the host is increased, and the product expression level is also increased accordingly.
在本发明的另一种实施方式中,所选用的哺乳动物宿主细胞可以为NS0、HEK293、PERC6或CHO,优选CHO细胞作为双特异抗体的表达宿主。CHO细胞具有一套完整的合成、组装和分泌蛋白质的细胞装置,因此,产生的抗体分子能够维持正确的蛋白构象以及翻译后糖基化加工,成为功能性抗体分子,并分泌到细胞外,便于分离纯化。CHO细胞还具有分泌产生较少自身蛋白以及蛋白酶的特点,同时CHO细胞可以经过无血清驯化成为悬浮状态生长,可以实现高密度的培养,目的蛋白产量可以达到2-10g/L的高产,特别适合工业化应用,也因此,过去30多年绝大多数上市的抗体药物都是采用CHO细胞表达体系。In another embodiment of the present invention, the selected mammalian host cells can be NSO, HEK293, PERC6 or CHO, and CHO cells are preferably used as the expression host of the bispecific antibody. CHO cells have a complete set of cellular machinery for synthesizing, assembling and secreting proteins. Therefore, the produced antibody molecules can maintain the correct protein conformation and post-translational glycosylation processing, become functional antibody molecules, and be secreted outside the cell, which is convenient for Isolation and Purification. CHO cells also have the characteristics of secreting and producing less self-proteins and proteases. At the same time, CHO cells can be grown in suspension after serum-free acclimation, which can achieve high-density culture. The target protein yield can reach a high yield of 2-10g/L. Industrial application, and therefore, most of the antibody drugs on the market in the past 30 years have used the CHO cell expression system.
本发明第六方面提供本发明第一方面所提供的抗CLD18A2的单域抗体、或本发明第二方面所提供的双特异性抗体的制备方法,包括如下步骤:在适合表达所述抗体的条件下,培养本发明第五方面所提供的抗体的表达系统,从而表达出所述的抗CLD18A2的单域抗体、或双特异性抗体,纯化分离出所述的抗体。The sixth aspect of the present invention provides a method for preparing the anti-CLD18A2 single domain antibody provided in the first aspect of the present invention, or the bispecific antibody provided in the second aspect of the present invention, comprising the following steps: under conditions suitable for expressing the antibody Next, the antibody expression system provided by the fifth aspect of the present invention is cultured to express the anti-CLD18A2 single domain antibody or bispecific antibody, and the antibody is purified and isolated.
本发明第七方面提供一种抗体-药物复合物,所述药物复合物包括本发明第一方面所提供的抗CLD18A2的单域抗体和细胞毒性药物。The seventh aspect of the present invention provides an antibody-drug complex, the drug complex comprising the anti-CLD18A2 single domain antibody and the cytotoxic drug provided in the first aspect of the present invention.
本发明所提供的抗体-药物复合物的结构可以如式1所示:The structure of the antibody-drug complex provided by the present invention can be shown in formula 1:
V HH/CLD18A2-连接肽-Z-[L-D]n I V HH/CLD18A2 -Linking Peptide-Z-[LD]n I
其中,V HH/CLD18A2为抗CLD18A2的单域抗体; Wherein, VHH/CLD18A2 is a single domain antibody against CLD18A2;
Z为附属功能区域,Z选自用于延长血清半衰期的结构域和/或药物偶联结构域、或不存在;Z is an accessory functional region, and Z is selected from a domain and/or a drug-conjugating domain for prolonging serum half-life, or is absent;
D为细胞毒性药物分子;D is a cytotoxic drug molecule;
L为连接链;L is the connection chain;
n表示偶联的D的平均数,且0<n≤10,优选2≤n≤7;更优选3≤n≤6;最优选4。n represents the average number of D coupled, and 0<n≤10, preferably 2≤n≤7; more preferably 3≤n≤6; most preferably 4.
本发明所提供的抗体-药物复合物中,式I中,VHH/CLD18A2-连接肽-Z部分为可以是一个融合蛋白,抗CLD18A2单域抗体(V HH/CLD18A2)的C端通过连接肽与附属功能区域相连、或者与连接链和细胞毒性药物分子相连。例如,当Z不存在时,其从N端至C端可以依次包括抗CLD18A2单域抗体、连接肽,再例如,当Z存在时,其从N端至C端可以依次包括抗CLD18A2单域抗体、连接肽、用于延长血清半衰期的结构域和/或药物偶联结构域。 In the antibody-drug complex provided by the present invention, in formula I, the VHH/CLD18A2-connecting peptide-Z part can be a fusion protein, and the C-terminus of the anti-CLD18A2 single-domain antibody (VHH/CLD18A2 ) is connected with the connecting peptide through the connecting peptide. Accessory functional domains are linked, or linked to linker chains, and cytotoxic drug molecules. For example, when Z is not present, it can include anti-CLD18A2 single domain antibody and linker peptide in order from N-terminal to C-terminal, and for example, when Z is present, it can include anti-CLD18A2 single-domain antibody from N-terminal to C-terminal in order , linker peptides, domains for extending serum half-life and/or drug-conjugation domains.
本发明所提供的抗体-药物复合物中,V HH/CLD18A2为抗CLD18A2的单域抗体,所述 V HH/CLD18A2可以为单价的,即包括一个抗原结合部位,所述V HH/CLD18A2也可以为多价的,即可以包括2个以上相同或不同序列的抗原结合部位,这些抗原结合部位可以是串联结构。 In the antibody-drug complex provided by the present invention, VHH/CLD18A2 is a single-domain antibody against CLD18A2, and the VHH/CLD18A2 can be monovalent, that is, it includes an antigen-binding site, and the VHH/CLD18A2 can also be To be multivalent, that is, it may include two or more antigen-binding sites of the same or different sequences, and these antigen-binding sites may be in a tandem structure.
本发明所提供的抗体-药物复合物中,可以包括附属功能区域(Z)。所述的附属功能区可以包括但不限于免疫球蛋白Fc区、血清白蛋白片段、聚乙二醇片段(PEG)、结合血清白蛋白的结构域(ABD)、多肽链、抗体或明胶样单元等中的一种或多种的组合,以及上述结构的衍生物,包括突变体和融合蛋白等。In the antibody-drug complex provided by the present invention, an accessory functional region (Z) may be included. The accessory functional region may include, but is not limited to, an immunoglobulin Fc region, a serum albumin fragment, a polyethylene glycol fragment (PEG), a serum albumin binding domain (ABD), a polypeptide chain, an antibody or a gelatin-like unit A combination of one or more of the above, as well as derivatives of the above structures, including mutants and fusion proteins, and the like.
在本发明一具体实施例中,Z为ABD-(GGC) n,其中,n为≥1的整数,优选n为4、5、6、7、8、9、10。ABD为结合人血清白蛋白的单域抗体,(GGC) n为提供药物偶联半胱氨酸基团的。 In a specific embodiment of the present invention, Z is ABD-(GGC) n , wherein n is an integer greater than or equal to 1, preferably n is 4, 5, 6, 7, 8, 9, or 10. ABD is a single-domain antibody that binds human serum albumin, and (GGC) n provides a drug-conjugated cysteine group.
在本发明一具体实施例中,Z为(PAEC)n,其中,n为≥1的整数,优选n为4、5、6、7、8、9、10,PAEC由脯氨酸(P)、丙氨酸(A)和谷氨酸(E)和半胱氨酸(C)组成。更具体的,Z的氨基酸序列可以包括如SEQ ID NO.124所示的序列。该序列能明显地延长在体内的半衰期,以及增加药物的偶联位点。In a specific embodiment of the present invention, Z is (PAEC)n, wherein, n is an integer greater than or equal to 1, preferably n is 4, 5, 6, 7, 8, 9, 10, PAEC is composed of proline (P) , alanine (A) and glutamic acid (E) and cysteine (C). More specifically, the amino acid sequence of Z can include the sequence shown in SEQ ID NO.124. This sequence can significantly prolong the half-life in vivo and increase the conjugation site of the drug.
在本发明一具体实施例中,Z为免疫球蛋白Fc区。In a specific embodiment of the present invention, Z is an immunoglobulin Fc region.
在本发明另一具体实施例中,所述人免疫球蛋白Fc区中包括用于改变Fc介导的效应功能的突变,所述效应功能包括CDC活性、ADCC活性、ADCP活性中的一种或多种的组合。In another specific embodiment of the present invention, the human immunoglobulin Fc region includes a mutation for altering Fc-mediated effector function, the effector function including one of CDC activity, ADCC activity, ADCP activity or various combinations.
在本发明另一具体实施例中,所述免疫球蛋白选自IgG、IgA1、IgA2、IgD、IgE、IgM中的一种或多种的组合,所述IgG选自IgG1、IgG2、IgG3或IgG4亚型中的一种或多种的组合。In another specific embodiment of the present invention, the immunoglobulin is selected from a combination of one or more of IgG, IgA1, IgA2, IgD, IgE, and IgM, and the IgG is selected from IgG1, IgG2, IgG3 or IgG4 A combination of one or more of the subtypes.
在本发明另一具体实施例中,免疫球蛋白Fc区的氨基酸序列可以包括SEQ ID NO.119~123其中之一所示的序列。In another specific embodiment of the present invention, the amino acid sequence of the immunoglobulin Fc region may include the sequence shown in one of SEQ ID NO. 119-123.
在本发明另一具体实施例中,所述附属功能区选自人免疫球蛋白IgG1恒定区Fc部分,所述序列包含铰链区及CH2和CH3,使得抗体-药物复合物形成式1的二聚体结构,并保持IgG1恒定区Fc所具有的CDC和ADCC作用。在本发明另一具体实施例中,所述附属功能区选自特异性结合人血清白蛋白的单域抗体。In another specific embodiment of the present invention, the accessory functional region is selected from the Fc portion of the human immunoglobulin IgG1 constant region, and the sequence comprises the hinge region and CH2 and CH3, such that the antibody-drug complex forms the dimerization of
本发明所提供的抗体-药物复合物中,连接肽片段通常可以为一段长度合适的由甘氨酸(G)和/或丝氨酸(S)和/或丙氨酸(A)和/或苏氨酸(T)构成的柔性多肽,能够保持双特异抗体分子各结构域的正确折叠,以及相互的柔韧性,例如,所述连接肽片段的氨基酸序列可以包括如(GS)n、(GGS)n、(GGSG)n、(GGGS)nA、(GGGGS)nA、(GGGGA)nA、(GGGGG)nA等序列,其中,n选自1-10之间的整数。在本发明一具体实施例中,连接肽的 氨基酸序列可以包括:GGGGSGGGS(SEQ ID NO.160)。In the antibody-drug complex provided by the present invention, the connecting peptide fragment can usually be a segment of suitable length composed of glycine (G) and/or serine (S) and/or alanine (A) and/or threonine ( T) is a flexible polypeptide that can maintain the correct folding of each domain of the bispecific antibody molecule and the flexibility of each other. For example, the amino acid sequence of the connecting peptide fragment can include such as (GS)n, (GGS)n, ( GGSG)n, (GGGS)nA, (GGGGS)nA, (GGGGA)nA, (GGGGG)nA and other sequences, wherein n is selected from an integer between 1-10. In a specific embodiment of the present invention, the amino acid sequence of the linking peptide may include: GGGGSGGGS (SEQ ID NO. 160).
本发明所提供的抗体-药物复合物中,可以包括连接链。连接链(linker)通常可以用于连接所述细胞毒性小分子药物和抗CLD18A2单域抗体融合蛋白,通常可以使用能够结合药物分子和氨基酸侧链的反应性官能基的连接链。连接链一般可分为可降解连接链和不可降解连接链两大类。可降解连接链可以包括:腙键(hydrazones)、顺式乌头酰基(cis-aconityl)、酰肼键、二硫键、肽键、β-葡萄糖醛酸(glucuronic acid-based)类键等;不可降解类的连接链可以包括琥珀酰亚胺-硫醚键等。具体可以是例如,N-琥珀酰亚胺基4-(2-吡啶基二硫代)戊酸酯(SPP)、N-琥珀酰亚胺基(4-碘乙酰基)氨基苯甲酸酯(SIAB)、N-琥珀酰亚胺基4-(马来酰亚胺甲基)环己烷羧酸酯(SMCC)、6-马来酰亚氨基己酰基(MC)、马来酰亚氨基丙酰基(MP)、缬氨酸-瓜氨酸(VC)、丙氨酸-苯丙氨酸(ala-phe)、对氨基苄氧羰基(PAB)等以及上述接头的任意组合。这些连接链可以单独使用,也可以互相组合使用,例如,MC-VC-PAB等。在本发明一具体实施中,L的连接链的化学结构式可以如下所示:In the antibody-drug complex provided by the present invention, a linker chain may be included. A linker can usually be used to connect the cytotoxic small molecule drug and the anti-CLD18A2 single domain antibody fusion protein, and a linker that can bind the reactive functional group of the drug molecule and the amino acid side chain can usually be used. Linking chains can generally be divided into two categories: degradable linking chains and non-degradable linking chains. The degradable linking chain may include: hydrazones, cis-aconiyl, hydrazide bonds, disulfide bonds, peptide bonds, β-glucuronic acid-based bonds, etc.; Linking chains of the non-degradable type may include succinimide-thioether linkages and the like. Specifically, for example, N-succinimidyl 4-(2-pyridyldithio)valerate (SPP), N-succinimidyl(4-iodoacetyl)aminobenzoate ( SIAB), N-succinimidyl 4-(maleimidomethyl)cyclohexanecarboxylate (SMCC), 6-maleimidohexanoyl (MC), maleimidopropyl Acyl (MP), valine-citrulline (VC), alanine-phenylalanine (ala-phe), p-aminobenzyloxycarbonyl (PAB), etc. and any combination of the above linkers. These linking chains can be used alone or in combination with each other, for example, MC-VC-PAB, etc. In a specific implementation of the present invention, the chemical structural formula of the linking chain of L can be as follows:
-B b-S s-A a-S` s`- II -B b -S s -A a -S` s` - II
其中,B为与抗CLD18A2单域抗体融合蛋白上氨基酸共价结合的共价结合单元,b为0或1;Wherein, B is a covalent binding unit covalently bound to the amino acid on the anti-CLD18A2 single-domain antibody fusion protein, and b is 0 or 1;
S、S`为间隔单元,且s、s`各自独立地为0-3的整数;S and S` are interval units, and s and s` are each independently an integer from 0 to 3;
A为氨基酸单元;a为0-12的整数。A is an amino acid unit; a is an integer of 0-12.
所述连接链中,可以包括共价结合单元。例如,当b为1时,连接链上可以带有亲电性质的基团,可与抗CLD18A2单域抗体融合蛋白上的亲核半胱氨酸或硒代半胱氨酸反应,从而形成共价键。亲电子性质的基团包括但不限于马来酰亚胺和卤代乙酰胺基团。药物-连接链中间体上带有亲电性质的基团,也可与抗CLD18A2单域抗体融合蛋白上的亲核半胱氨酸或硒代半胱氨酸反应,从而形成共价键。连接链可以为分支状结构,用于使一种以上药物部分共价结合到与靶向配体结合的亲水性多肽上。分支状连接链可以增加药物与抗体的摩尔比,即负荷。In the linking chain, a covalent binding unit may be included. For example, when b is 1, a group with electrophilic properties on the linking chain can react with nucleophilic cysteine or selenocysteine on the anti-CLD18A2 single domain antibody fusion protein to form a co- price key. Groups of electrophilic nature include, but are not limited to, maleimide and haloacetamide groups. The group with electrophilic properties on the intermediate of the drug-linking chain can also react with the nucleophilic cysteine or selenocysteine on the anti-CLD18A2 single domain antibody fusion protein to form a covalent bond. The linker chain can be a branched structure for covalently binding more than one drug moiety to the hydrophilic polypeptide bound to the targeting ligand. Branched linker chains can increase the molar ratio of drug to antibody, ie, loading.
当抗CLD18A2单域抗体融合蛋白序列中含有非天然氨基酸时,连接链可以带有反应性官能基,该反应性官能基具有与存在于抗CLD18A2单域抗体融合蛋白上的亲电子基团反应的亲核基团。抗CLD18A2单域抗体融合蛋白上有用的亲电子基团包括,但不限于醛和酮羰基。连接链的亲核基团的杂原子可以与抗CLD18A2单域抗体融合蛋白上的亲电子基团反应并且形成共价键。连接链上有用的亲核基团包括,但不限于酰肼、肟、氨基、肼、缩氨基硫脲(thiosemicarbazone)、肼羧酸酯和芳基酰肼。抗CLD18A2单域抗体融合蛋白上的亲电子基 团提供了用于结合(附着)连接链的便利位置。When the anti-CLD18A2 single-domain antibody fusion protein sequence contains an unnatural amino acid, the linking chain may have a reactive functional group that has the ability to react with an electrophilic group present on the anti-CLD18A2 single-domain antibody fusion protein Nucleophilic group. Useful electrophilic groups on anti-CLD18A2 single domain antibody fusion proteins include, but are not limited to, aldehyde and ketone carbonyl groups. The heteroatoms of the nucleophilic group linking the chain can react with the electrophilic group on the anti-CLD18A2 single domain antibody fusion protein and form a covalent bond. Useful nucleophilic groups on the linking chain include, but are not limited to, hydrazides, oximes, amino groups, hydrazines, thiosemicarbazones, hydrazine carboxylates, and aryl hydrazides. The electrophilic group on the anti-CLD18A2 single domain antibody fusion protein provides a convenient location for binding (attaching) the linker chain.
双-马来酰亚胺试剂能够使纳米抗体衍生物上的巯基与含巯基的药物部分或连接基中间体按照依次或同时的方式结合。The bis-maleimide reagent enables the sulfhydryl group on the Nanobody derivative to bind to the sulfhydryl group-containing drug moiety or linker intermediate in a sequential or simultaneous manner.
所述连接链中,可以包括氨基酸单元,所述氨基酸单元通常包括氨基酸残基。式II中,A可以为二肽、三肽、四肽、五肽、六肽、七肽、八肽、九肽、十肽、十一肽或十二肽单元。包含氨基酸单元的氨基酸残基包括那些天然存在的以及最小的氨基酸和非-天然存在的氨基酸类似物,诸如瓜氨酸。可以用一种或多种酶,包括肿瘤相关蛋白酶以酶促方式裂解氨基酸单元,以便释放药物部分(-D)。In the linking chain, amino acid units may be included, and the amino acid units usually include amino acid residues. In formula II, A may be a dipeptide, tripeptide, tetrapeptide, pentapeptide, hexapeptide, heptapeptide, octapeptide, nonapeptide, decapeptide, undecapeptide or dodecapeptide unit. Amino acid residues comprising amino acid units include those naturally occurring as well as minimal amino acids and non-naturally occurring amino acid analogs, such as citrulline. The amino acid unit can be enzymatically cleaved with one or more enzymes, including tumor-associated proteases, to release the drug moiety (-D).
所述连接链中,可以包括间隔单元(S或S`)。所述间隔单元存在时通常可以使氨基酸单元(-A-)与药物部分(D)连接;或者,在氨基酸单元不存在时,间隔单元可以使式II中的Bb单元与药物部分连接。在氨基酸单元和B b单元都不存在时,间隔单元还使药物部分与靶向配体衍生物单元连接。间隔单元一般具有两种类型:自我消除(self-immolative)和非自我消除的。非自我消除的间隔单元为部分或所有间隔单元在从配体衍生物-药物复合物裂解后,特别是酶促裂解氨基酸单元后保持与药物部分结合的间隔单元。 In the connecting chain, a spacer unit (S or S') may be included. The spacer unit may typically link the amino acid unit (-A-) to the drug moiety (D) in the presence of the spacer unit; alternatively, the spacer unit may link the Bb unit of formula II to the drug moiety in the absence of the amino acid unit. The spacer unit also links the drug moiety to the targeting ligand derivative unit when neither the amino acid unit nor the B b unit is present. Spacer units are generally of two types: self-immolative and non-self-eliminating. A non-self-eliminating spacer unit is one in which some or all of the spacer unit remains bound to the drug moiety after cleavage from the ligand derivative-drug complex, especially after enzymatic cleavage of the amino acid unit.
本发明所提供的抗体-药物复合物中,可以包括细胞毒性药物分子。所述细胞毒性分子通常是具有细胞毒性或抑制细胞效应的任意化合物或基团。这些细胞毒性分子可以包括:(i)可以起微管蛋白抑制剂、有丝分裂抑制剂、拓扑异构酶抑制剂或DNA嵌入剂作用的化疗剂;(ii)可以通过酶促方式起作用的蛋白毒素;和(iii)放射性同位素。The antibody-drug complexes provided by the present invention may include cytotoxic drug molecules. The cytotoxic molecule is generally any compound or group that has a cytotoxic or cytostatic effect. These cytotoxic molecules can include: (i) chemotherapeutic agents that can act as tubulin inhibitors, mitotic inhibitors, topoisomerase inhibitors, or DNA intercalators; (ii) protein toxins that can act enzymatically ; and (iii) radioisotopes.
典型的细胞毒性分子可以是包括但不限于美登木素生物碱、澳瑞他汀(auristatin)、多拉司他汀(dolastatin)、单端孢霉烯(trichothecene)、CC1065、加利车霉素(Calicheamicin)和其它烯二炔类(enediyne)抗生素、紫杉醇(taxane)、蒽环类抗生素(anthracycline)及其立体异构体、同电子排列体(isostere)、类似物或衍生物或其组合。更具体的,所述单甲基澳瑞他汀可以为单甲基澳瑞他汀E(MMAE)或单甲基澳瑞他汀F(MMAF),所述美登素类可以为N2'-脱乙酰基-N2'-(3-巯基-1-氧代丙基)-美登素(DM1),N2'-脱乙酰基-N2'-(4-巯基-1-氧代戊基)-美登素(DM3)和N2'-脱乙酰基-N2'-(4-巯基-4-甲基-1-氧代戊基)-美登素(DM4)。Typical cytotoxic molecules may include, but are not limited to, maytansinoids, auristatin, dolastatin, trichothecene, CC1065, calicheamicin ( Calicheamicin and other enediyne antibiotics, taxanes, anthracyclines and their stereoisomers, isosteres, analogs or derivatives or combinations thereof. More specifically, the monomethyl auristatin can be monomethyl auristatin E (MMAE) or monomethyl auristatin F (MMAF), and the maytansinoids can be N2'-deacetyl -N2'-(3-mercapto-1-oxopropyl)-maytansine (DM1), N2'-deacetyl-N2'-(4-mercapto-1-oxopentyl)-maytansine (DM3) and N2'-deacetyl-N2'-(4-mercapto-4-methyl-1-oxopentyl)-maytansine (DM4).
式I所示的抗体-药物复合物中,药物部分(D)还可以包括喜树碱衍生物7-乙基-10-羟基喜树碱(SN38),其是以拓扑异构酶为作用靶抑制DNA的合成而发挥作用的抗癌药物。In the antibody-drug complex shown in formula I, the drug moiety (D) can also include a camptothecin derivative 7-ethyl-10-hydroxycamptothecin (SN38), which is based on topoisomerase as an action target Anticancer drugs that inhibit DNA synthesis.
式I所示的抗体-药物复合物中,蛋白毒素包括但不限于:白喉毒素A链、白喉毒素的非结合活性片段、外毒素A链、蓖麻毒素A链、相思豆毒蛋白A链(abrin A chain)、蒴莲根毒素A链(modeccin A chain)、α-八叠球菌(alpha-sarcin)、油桐(Aleurites fordii)蛋白、石竹素蛋白 (dianthin proteins)、泻果素(curcin)、巴豆毒蛋白(crotin)、白树毒素(gelonin)、米托洁林(mitogellin)、局限曲菌素(restrictocin)、酚霉素(phenomycin)、依诺霉素(enomycin)等。In the antibody-drug complex shown in formula I, protein toxins include but are not limited to: diphtheria toxin A chain, non-binding active fragment of diphtheria toxin, exotoxin A chain, ricin A chain, acacia soybean protein A chain ( abrin A chain), modeccin A chain, alpha-sarcin, Aleurites fordii protein, dianthin proteins, curcin , crotonin (crotin), gelonin (gelonin), mitogellin (mitogellin), restricted aspergillin (restrictocin), phenomycin (phenomycin), enomycin (enomycin) and so on.
式I所示的抗体-药物复合物中,治疗放射性核素包括但不限于:32P、33P、90Y、125I、131I、131In、153Sm、186Re、188Re、211At、212Bi、212Pb和Lu的放射性同位素。In the antibody-drug complex of formula I, therapeutic radionuclides include, but are not limited to: radioisotopes of 32P, 33P, 90Y, 125I, 131I, 131In, 153Sm, 186Re, 188Re, 211At, 212Bi, 212Pb and Lu.
本发明第八方面提供本发明第七方面所提供的抗体-药物复合物的制备方法,包括:在抗CLD18A2单域抗体的基础上,可以通过增加单域抗体的串联和附属功能区的设置形成抗CLD18A2单域抗体的融合蛋白,将融合蛋白与细胞毒性分子交联以提供所述抗体-药物复合物。抗CLD18A2单域抗体的多价串联可以增加对人CLD18A2的结合能力,附属功能区根据不同的目的设计,可以延长在体内的半衰期,或者提高亲水性,或者增加毒性化药基团的偶联位点。The eighth aspect of the present invention provides the preparation method of the antibody-drug complex provided by the seventh aspect of the present invention, including: on the basis of the anti-CLD18A2 single-domain antibody, it can be formed by increasing the tandem connection of the single-domain antibody and the setting of the accessory functional region. A fusion protein of an anti-CLD18A2 single domain antibody, the fusion protein is cross-linked with a cytotoxic molecule to provide the antibody-drug complex. The multivalent tandem of anti-CLD18A2 single-domain antibody can increase the binding ability to human CLD18A2, and the accessory functional region can be designed according to different purposes, which can prolong the half-life in vivo, or improve the hydrophilicity, or increase the coupling of toxic chemical groups site.
在本发明一具体实施例中,两个相同序列的抗CLD18A2单域抗体串联,并在C末端引入人血清白蛋白结合序列(ABD)和GGC三个氨基酸的6个重复结构(SEQ ID NO.125),形成的单域抗体融合蛋白在与linker-MMAE通过巯基交联后,得到了DAR为4.42的单域抗体-药物复合物。In a specific embodiment of the present invention, two anti-CLD18A2 single-domain antibodies of the same sequence are connected in series, and a six-repeat structure of three amino acids of human serum albumin binding sequence (ABD) and GGC (SEQ ID NO. 125), after the formed single-domain antibody fusion protein is cross-linked with linker-MMAE through sulfhydryl groups, a single-domain antibody-drug complex with a DAR of 4.42 is obtained.
在本发明另一具体实施例中,在抗CLD18A2单域抗体的C端再串联同样序列的抗CLD18A2单域抗体,并在C端融合了高亲水性多肽,引入了6个Cys氨基酸,形成的单域抗体衍生物(SEQ ID NO.126)在与linker-MMAE通过巯基交联后,得到了DAR为4.10的抗体-药物复合物。In another specific embodiment of the present invention, an anti-CLD18A2 single-domain antibody of the same sequence is connected in series at the C-terminus of the anti-CLD18A2 single-domain antibody, and a highly hydrophilic polypeptide is fused at the C-terminus, and 6 Cys amino acids are introduced to form The single-domain antibody derivative (SEQ ID NO. 126) was cross-linked with linker-MMAE through sulfhydryl groups to obtain an antibody-drug complex with a DAR of 4.10.
在本发明另一具体实施例中,在抗CLD18A2单域抗体的C端融合了人IgG1FC形成的单域抗体衍生物(SEQ ID NO.147),在与linker-MMAE通过链间巯基交联后,得到了DAR为4.21的抗体-药物复合物。In another specific embodiment of the present invention, a single-domain antibody derivative (SEQ ID NO. 147) formed by fusing human IgG1FC to the C-terminus of anti-CLD18A2 single-domain antibody, after cross-linking with linker-MMAE through interchain sulfhydryl groups , an antibody-drug complex with a DAR of 4.21 was obtained.
本发明所提供的抗体-药物复合物的制备方法中,本领域技术人员可选用各种公知的有机化学反应、条件和试剂以提供本发明第七方面所提供的抗体-药物复合物。例如,当交联氨基酸为半胱氨酸时,可以包括:(1)单域抗体衍生物上的半胱氨酸基团与连接链试剂反应,从而通过共价键形成配体衍生物-连接链中间体,随后与活化的药物部分D反应;或,(2)使药物部分的亲核基团与连接链试剂反应,从而通过共价键形成药物-连接链中间体,随后与单域抗体衍生物上的半胱氨酸基团反应。偶联方法(1)和(2)可以用于各种靶向配体、药物部分和连接链以便制备式I的配体衍生物-药物复合物。半胱氨酸巯基为亲核性的并且能够与连接链试剂和药物-连接链中间体上的亲电子基团反应而形成共价键,所述的药物-连接链中间体包括:(i)活性酯类,诸如NHS酯类、HOBt酯类、卤代甲酸酯类和酰基卤类; (ii)烷基和苄基卤化物,诸如卤代乙酰胺类;(iii)醛类、酮类、羧基和马来酰亚胺基;和(iv)通过硫化物交换的二硫化物,包括吡啶基二硫化物。药物部分上的亲核基团可以包括但不限于胺、巯基、羟基、酰肼、肟、肼、缩氨基硫脲、肼羧酸酯和芳基酰肼基团,它们能够与连接链部分和连接链试剂上的亲电子基团反应而形成共价键。In the preparation method of the antibody-drug complex provided by the present invention, those skilled in the art can select various well-known organic chemical reactions, conditions and reagents to provide the antibody-drug complex provided by the seventh aspect of the present invention. For example, when the cross-linking amino acid is cysteine, it can include: (1) The cysteine group on the single-domain antibody derivative reacts with the linking chain reagent, thereby forming a ligand derivative-linking through a covalent bond chain intermediate, which is subsequently reacted with the activated drug moiety D; or, (2) the nucleophilic group of the drug moiety is reacted with the linking chain reagent, thereby forming a drug-linking chain intermediate by covalent bonding, which is subsequently reacted with the single domain antibody The cysteine group on the derivative reacts. Coupling methods (1) and (2) can be used for a variety of targeting ligands, drug moieties and linkers to prepare ligand derivative-drug complexes of formula I. Cysteine sulfhydryl groups are nucleophilic and can react with electrophilic groups on linker reagents and drug-linker intermediates to form covalent bonds, said drug-linker intermediates including: (i) Active esters such as NHS esters, HOBt esters, haloformates and acid halides; (ii) alkyl and benzyl halides such as haloacetamides; (iii) aldehydes, ketones, Carboxyl and maleimide groups; and (iv) disulfides exchanged by sulfide, including pyridyl disulfides. Nucleophilic groups on the drug moiety can include, but are not limited to, amine, sulfhydryl, hydroxyl, hydrazide, oxime, hydrazine, thiosemicarbazide, hydrazine carboxylate, and arylhydrazide groups, which can interact with the linker moiety and The electrophilic groups on the linker reagent react to form a covalent bond.
在某些条件下,例如细胞表达的全长抗体,分子内存在着链内与链间二硫键的情况下,可以通过用低浓度的还原剂,诸如DTT(Cleland试剂,二硫苏糖醇)或TCEP(三(2-羧乙基)膦盐酸盐处理使抗体的链间二硫键还原,再与连接链试剂反应。Under certain conditions, such as cell-expressed full-length antibodies, intra- and inter-chain disulfide bonds exist within the molecule. ) or TCEP (tris (2-carboxyethyl) phosphine hydrochloride treatment to reduce the interchain disulfide bond of the antibody, and then react with the linking chain reagent.
当交联氨基酸为硒代半胱氨酸时,可参考Xiuling Li等的方案(Bioconjug Chem.201518;26(11):2243-8.),当交联氨基酸为非天然氨基酸时,可参考Han Xiao等的方案(Angew Chem Int Ed Engl.2013 Dec 23;52(52):14080-3)。When the cross-linking amino acid is selenocysteine, refer to the scheme of Xiuling Li et al. (Bioconjug Chem. 201518; 26(11): 2243-8.), when the cross-linking amino acid is an unnatural amino acid, refer to Han The protocol of Xiao et al. (Angew Chem Int Ed Engl. 2013 Dec 23;52(52):14080-3).
本发明第九方面提供一种药物组合物,包括本发明第一方面所提供的抗CLD18A2的单域抗体、本发明第二方面所提供的双特异性抗体、本发明第五方面所提供的抗体的表达系统的培养物、或本发明第七方面所提供的抗体-药物复合物。所述药物组合物中,所述抗CLD18A2的单域抗体、双特异性抗体、培养物、或抗体-药物复合物的含量通常为治疗有效量的。所述药物组合物还可以包括药学上可接受的载体。所述载体可以包括各种赋形剂和稀释剂,这些载体本身并不是必要的活性成分,且施用后没有过分的毒性。合适的载体对于本领域技术人员来说应该是熟知的,例如,在Remington's Pharmaceutical Sciences(Mack Pub.Co.,N.J.,1991)中可找到关于药学上可接受的载体的充分讨论。在本发明一优选具体实施例中,所述药物组合物可以通过注射途径来给药,尤其是玻璃体腔内注射,因此所述药物组合物优选是粉针剂(如冻干粉针剂)和液体制剂。The ninth aspect of the present invention provides a pharmaceutical composition, comprising the anti-CLD18A2 single domain antibody provided by the first aspect of the present invention, the bispecific antibody provided by the second aspect of the present invention, and the antibody provided by the fifth aspect of the present invention The culture of the expression system, or the antibody-drug complex provided by the seventh aspect of the present invention. In the pharmaceutical composition, the content of the anti-CLD18A2 single domain antibody, bispecific antibody, culture, or antibody-drug complex is usually a therapeutically effective amount. The pharmaceutical composition may also include a pharmaceutically acceptable carrier. The carrier may include various excipients and diluents which are not themselves necessary for the active ingredient and which are not unduly toxic after administration. Suitable carriers will be well known to those skilled in the art, for example, a thorough discussion of pharmaceutically acceptable carriers can be found in Remington's Pharmaceutical Sciences (Mack Pub. Co., N.J., 1991). In a preferred embodiment of the present invention, the pharmaceutical composition can be administered by injection route, especially intravitreal injection, so the pharmaceutical composition is preferably a powder for injection (such as freeze-dried powder for injection) and a liquid preparation .
本发明第十方面提供一种细胞,所述细胞含膜结合的嵌合抗原受体,所述嵌合抗原受体包括跨膜域、胞内域和胞外域,所述胞外域包括本发明第一方面所提供的抗CLD18A2的单域抗体。本发明所提供的细胞表面可以表达所述抗CLD18A2的单域抗体,所述细胞通常可以通过胞外域结合于它们对应的抗原,更具体可以是通过包含抗CLD18A2的单域抗体的胞外域结合于Claudin18.2抗原,当所述多肽结合于其对应的抗原时,所述细胞可以活化和/或刺激从而得以增殖,并杀伤相应靶细胞。所述细胞具体可以为T淋巴细胞、NK细胞、巨噬细胞等。A tenth aspect of the present invention provides a cell comprising a membrane-bound chimeric antigen receptor, wherein the chimeric antigen receptor includes a transmembrane domain, an intracellular domain and an extracellular domain, and the extracellular domain includes the first In one aspect the anti-CLD18A2 single domain antibodies are provided. The surface of the cells provided by the present invention can express the anti-CLD18A2 single-domain antibody, and the cells can usually bind to their corresponding antigens through the extracellular domain, and more specifically can bind to the anti-CLD18A2 single-domain antibody through the extracellular domain. Claudin18.2 antigen, when the polypeptide binds to its corresponding antigen, the cells can be activated and/or stimulated to proliferate and kill the corresponding target cells. Specifically, the cells can be T lymphocytes, NK cells, macrophages and the like.
本发明所提供的细胞中,嵌合抗原受体可以包括胞外域,胞外域通常可以包括靶向目标抗原的抗体,即上述抗CLD18A2的单域抗体。In the cells provided by the present invention, the chimeric antigen receptor may include an extracellular domain, and the extracellular domain may generally include an antibody targeting the target antigen, that is, the above-mentioned anti-CLD18A2 single domain antibody.
本发明所提供的细胞中,所述嵌合抗原受体还可以包括跨膜域,所述跨膜域主要可以将 嵌合抗原受体固定于T细胞的细胞膜。所述跨膜域可以包括CD4、CD8、CD8b、CD28跨膜域的序列,T细胞受体的亚基诸如α、β、γ或δ,IL-2受体的亚基(α链),低亲和力神经生长因子受体(LNGFR或p75)的亚基(β链或γ链),或Fc受体的亚基链的跨膜域。在本发明一具体实施例中,所述跨膜域可以包括如SEQ ID NO:128所示的氨基酸序列。在本发明另一具体实施例中,所述跨膜域可以包括如SEQ ID NO:129所示的氨基酸序列。In the cells provided by the present invention, the chimeric antigen receptor can also include a transmembrane domain, and the transmembrane domain can mainly fix the chimeric antigen receptor on the cell membrane of T cells. The transmembrane domain may include sequences of CD4, CD8, CD8b, CD28 transmembrane domains, subunits of T cell receptors such as alpha, beta, gamma or delta, subunits of IL-2 receptors (alpha chain), low Affinity Nerve growth factor receptor (LNGFR or p75) subunit (beta chain or gamma chain), or the transmembrane domain of the subunit chain of Fc receptors. In a specific embodiment of the present invention, the transmembrane domain may comprise the amino acid sequence shown in SEQ ID NO:128. In another specific embodiment of the present invention, the transmembrane domain may comprise the amino acid sequence shown in SEQ ID NO:129.
本发明所提供的细胞中,所述嵌合抗原受体还可以包括胞内域。所述胞内域可以是天然T细胞受体和协同作用以在抗原结合后启动信号转导的辅助受体的细胞质序列,以及这些序列的任何衍生物或变体,以及任何具有相同功能的合成序列。所述胞内域通常可以分为两大类,例如,可以包括共刺激结构域和/或信号结构域。所述共刺激结构域通常可以抗原非依赖性地为细胞完全活化提供二级或共刺激信号,其可以与抗原呈递细胞上的同源共刺激配体结合,以增强T细胞应答,例如通过增加增殖活化、分化等。所述共刺激结构域可以包括CD28、CD27、4-1BB(CD137)、OX40(CD134)、ICOS(CD278)、CD30、CD40、PD-1(CD279)、CD2、CD7、NKG2C(CD94)、B7-H3(CD276)的细胞内结构域。在本发明一具体实施例中,所述共刺激结构域可以包括CD28和/或CD137,其氨基酸序列可以包括SEQ ID NO:130和SEQ ID NO:131所示的氨基酸序列。所述信号结构域通常指上述抗体识别目标细胞表面的抗原时能够向细胞内转导信号的区域。所述信号结构域可以是基于免疫受体酪氨酸的活化基序(ITAM),ITAM是明确定义的信号传导基序,通常存在于多种受体的胞质内尾部,并且用作syk/zap70类酪氨酸激酶的结合位点。具体来说,所述信号结构域可以包括CD3ζ、FcRγ、FcRβ、FcRε、CD3γ、CD3δ、CD3ε、CD5、CD22、CD79a、CD79b和CD66d的信号传导结构域。在本发明一具体实施例中,所述信号结构域包括CD3ζ的信号传导结构域,其氨基酸序列可以包括如SEQ ID NO:132所示的氨基酸序列。天然TCR含有CD3ζ信号传导分子,因此该效应结构域的使用最接近自然界中发生的TCR构建体。In the cells provided by the present invention, the chimeric antigen receptor may also include an intracellular domain. The intracellular domain may be the cytoplasmic sequences of native T cell receptors and coreceptors that cooperate to initiate signal transduction upon antigen binding, as well as any derivatives or variants of these sequences, and any synthetic functional equivalents sequence. The intracellular domains can generally be divided into two broad categories, and can include, for example, costimulatory domains and/or signaling domains. The co-stimulatory domain can typically provide a secondary or co-stimulatory signal for full cell activation in an antigen-independent manner, which can bind to a cognate co-stimulatory ligand on an antigen-presenting cell to enhance T cell responses, for example by increasing Proliferation activation, differentiation, etc. The costimulatory domain may include CD28, CD27, 4-1BB (CD137), OX40 (CD134), ICOS (CD278), CD30, CD40, PD-1 (CD279), CD2, CD7, NKG2C (CD94), B7 - Intracellular domain of H3 (CD276). In a specific embodiment of the present invention, the costimulatory domain may include CD28 and/or CD137, and the amino acid sequence thereof may include the amino acid sequences shown in SEQ ID NO: 130 and SEQ ID NO: 131. The signaling domain generally refers to a region capable of transducing a signal into the cell when the above-mentioned antibody recognizes an antigen on the surface of the target cell. The signaling domain may be an immunoreceptor tyrosine-based activation motif (ITAM), which is a well-defined signaling motif typically present in the intracytoplasmic tail of various receptors and used as syk/ Binding site for zap70-like tyrosine kinases. Specifically, the signaling domains may include the signaling domains of CD3ζ, FcRγ, FcRβ, FcRε, CD3γ, CD3δ, CD3ε, CD5, CD22, CD79a, CD79b, and CD66d. In a specific embodiment of the present invention, the signaling domain includes the signaling domain of CD3ζ, and its amino acid sequence may include the amino acid sequence shown in SEQ ID NO: 132. Natural TCRs contain the CD3ζ signaling molecule, so the use of this effector domain is closest to the TCR constructs that occur in nature.
本发明所提供的细胞中,所述嵌合抗原受体还可以包括铰链区,所述铰链区包括CD8饺链区或IgG1或IgG4的CH2和/或CH3结构域,优选可以是CD8b的饺链区。在本发明一具体实施例中,铰链区可以包括如SEQ ID NO:127所示的氨基酸序列。In the cells provided by the present invention, the chimeric antigen receptor may also include a hinge region, and the hinge region includes the CD8 dumpling region or the CH2 and/or CH3 domains of IgG1 or IgG4, preferably the dumpling chain of CD8b Area. In a specific embodiment of the present invention, the hinge region may comprise the amino acid sequence shown in SEQ ID NO:127.
本发明所提供的细胞中,所述嵌合抗原受体自N端至C端通常依次包括抗CLD18A2的单域抗体、跨膜域(TM)和胞内域,所述胞内域自N端至C端依次包括共刺激结构域(ITAM)和信号结构域(ζ)。还可以包括铰链区(hinge),铰链区通常可以位于抗CLD18A2的单域抗体、跨膜域之间。In the cells provided by the present invention, the chimeric antigen receptor generally includes an anti-CLD18A2 single domain antibody, a transmembrane domain (TM) and an intracellular domain in sequence from the N-terminal to the C-terminal, and the intracellular domain starts from the N-terminal To the C-terminus, it includes a costimulatory domain (ITAM) and a signaling domain (ζ). A hinge region may also be included, and the hinge region may typically be located between the anti-CLD18A2 single domain antibody, transmembrane domains.
在本发明一具体实施例中,所述嵌合抗原受体自N端至C端可以依次包括抗CLD18A2 单域抗体、CD8b hinge、CD8bTM、CD28ITAM、CD3ζ。In a specific embodiment of the present invention, the chimeric antigen receptor may include anti-CLD18A2 single domain antibody, CD8bhinge, CD8bTM, CD28ITAM, and CD3ζ in sequence from the N-terminus to the C-terminus.
在本发明另一具体实施例中,所述嵌合抗原受体自N端至C端可以依次包括抗CLD18A2单域抗体、CD8b hinge、CD8bTM、CD137ITAM、CD3ζ。In another specific embodiment of the present invention, the chimeric antigen receptor may include anti-CLD18A2 single domain antibody, CD8bhinge, CD8bTM, CD137ITAM, CD3ζ in sequence from the N-terminus to the C-terminus.
在本发明另一具体实施例中,所述嵌合抗原受体自N端至C端可以依次包括抗CLD18A2单域抗体、CD8b hinge、CD28TM、CD28ITAM、CD137ITAM、CD3ζ。In another specific embodiment of the present invention, the chimeric antigen receptor may include anti-CLD18A2 single domain antibody, CD8bhinge, CD28TM, CD28ITAM, CD137ITAM, and CD3ζ in sequence from the N-terminus to the C-terminus.
本发明所提供的细胞中,所述的嵌合抗原受体还可以包含信号肽,所述信号肽主要用于将嵌合抗原受体表达于细胞膜上。在本发明一具体实施例中,SEQ ID NO:136所示的氨基酸序列。In the cells provided by the present invention, the chimeric antigen receptor may further comprise a signal peptide, and the signal peptide is mainly used to express the chimeric antigen receptor on the cell membrane. In a specific embodiment of the present invention, the amino acid sequence shown in SEQ ID NO: 136.
在本发明另一具体实施例中,所述嵌合抗原受体的氨基酸序列包括SEQ ID NO.137-140其中之一所示序列。In another specific embodiment of the present invention, the amino acid sequence of the chimeric antigen receptor includes the sequence shown in one of SEQ ID NO. 137-140.
本发明第十一方面提供本发明第一方面所提供的抗CLD18A2的单域抗体、本发明第二方面所提供的双特异性抗体、本发明第五方面所提供的抗体的表达系统的培养物、或本发明第七方面所提供的抗体-药物复合物、本发明第九方面所提供的药物组合物、或本发明第十方面所提供的细胞在制备药物中的用途。所述药物具体可以是用于治疗肿瘤的药物。所述肿瘤可以是实体瘤或血液肿瘤,更具体可以是肠癌、肺癌、肝癌、乳腺癌、食管癌、头颈癌、皮肤癌、肾癌、白血病、coad(结肠癌),lihc(肝细胞肝癌),ov(卵巢浆液性囊腺癌),ucec(子宫内膜癌),thca(甲状腺癌),skcm(皮肤黑色素瘤),luad(肺腺癌),hnsc(头颈鳞状细胞癌),gbm(多形成性胶质细胞瘤),prad(前列腺癌),thym(胸腺癌),lgg(脑低级别胶质瘤),read(直肠腺癌),pcpg(嗜铬细胞瘤和副神经节瘤),esca(食管癌),kirc(肾透明细胞癌),cesc(宫颈鳞癌和腺癌),blca(膀胱尿路上皮癌),kirp(肾乳头状细胞癌),paad(胰腺癌),stad(胃癌),kich(肾嫌色细胞癌),brca(乳腺浸润癌),lusc(肺鳞癌),sarc(肉瘤),LAML(急性髓细胞样白血病)等。The eleventh aspect of the present invention provides a culture of the anti-CLD18A2 single domain antibody provided by the first aspect of the present invention, the bispecific antibody provided by the second aspect of the present invention, and the expression system of the antibody provided by the fifth aspect of the present invention , or the use of the antibody-drug complex provided by the seventh aspect of the present invention, the pharmaceutical composition provided by the ninth aspect of the present invention, or the cell provided by the tenth aspect of the present invention in preparing a medicine. Specifically, the drug may be a drug for treating tumors. The tumor may be a solid tumor or a hematological tumor, more specifically, colon cancer, lung cancer, liver cancer, breast cancer, esophageal cancer, head and neck cancer, skin cancer, kidney cancer, leukemia, coad (colon cancer), lihc (hepatocellular carcinoma) ), ov (ovarian serous cystadenocarcinoma), ucec (endometrial cancer), thca (thyroid cancer), skcm (skin melanoma), luad (lung adenocarcinoma), hnsc (head and neck squamous cell carcinoma), gbm (glioma multiforme), prad (prostate cancer), thym (thymic carcinoma), lgg (brain low-grade glioma), read (rectal adenocarcinoma), pcpg (pheochromocytoma and paraganglioma) ), esca (esophageal cancer), kirc (renal clear cell carcinoma), csc (cervical squamous and adenocarcinoma), blca (bladder urothelial carcinoma), kirp (renal papillary cell carcinoma), paad (pancreatic carcinoma), stad (gastric cancer), kich (renal chromophobe carcinoma), brca (breast invasive carcinoma), lusc (lung squamous cell carcinoma), sarc (sarcoma), LAML (acute myeloid leukemia), etc.
本发明所提供的用途中,上述物质可以是单一药效成分,也可以与其他活性组分进行组合,共同地用于治疗肿瘤。In the use provided by the present invention, the above-mentioned substances can be a single medicinal component, or can be combined with other active components to jointly treat tumors.
本发明第十二方面提供一种治疗方法,包括向个体施用治疗有效量的第一方面所提供的抗CLD18A2的单域抗体、本发明第二方面所提供的双特异性抗体、本发明第五方面所提供的抗体的表达系统的培养物、或本发明第七方面所提供的抗体-药物复合物、本发明第九方面所提供的药物组合物、或本发明第十方面所提供的细胞。本发明所提供的治疗方法可以用于治疗包括但不限于肿瘤等的适应症。所述“个体”通常包括哺乳动物,所述哺乳动物可以为啮齿目动物、偶蹄目动物、奇蹄目动物、兔形目动物、非人灵长类动物、灵长目动物等, 所述灵长目动物可以为猴、猿或人,如哺乳动物、狗、猫、马、羊、猪、牛等,其可因利用所述T淋巴细胞、或组合物进行治疗而获益。所述“治疗有效量”通常指一用量在经过适当的给药期间后,能够达到治疗如上所列出的疾病的效果。优选的治疗有效量的选择可以由本领域普通技术人员根据各种因素来确定(例如通过临床试验),例如,当上述物质用于被施用的个体时,肿瘤的生长、增殖、复发和/或转移可以被抑制,更具体的,肿瘤的生长、增殖、复发和/或转移的至少10%、20%、30%、40%、50%、60%、70%、80%、90%、95%或99%的部分被抑制。The twelfth aspect of the present invention provides a treatment method, comprising administering to an individual a therapeutically effective amount of the anti-CLD18A2 single domain antibody provided in the first aspect, the bispecific antibody provided in the second aspect of the present invention, and the fifth aspect of the present invention. The culture of the antibody expression system provided by the aspect, or the antibody-drug complex provided by the seventh aspect of the present invention, the pharmaceutical composition provided by the ninth aspect of the present invention, or the cell provided by the tenth aspect of the present invention. The treatment methods provided by the present invention can be used to treat indications including but not limited to tumors. The "individual" generally includes mammals, which may be rodents, artiodactyls, odd ungulates, lagomorphs, non-human primates, primates, etc., which may be be monkeys, apes or humans, such as mammals, dogs, cats, horses, sheep, pigs, cattle, etc., which may benefit from treatment with the T lymphocytes, or compositions. The "therapeutically effective amount" generally refers to an amount which, after an appropriate period of administration, is effective in treating the diseases listed above. Selection of a preferred therapeutically effective amount can be determined by one of ordinary skill in the art based on a variety of factors (eg, through clinical trials), such as tumor growth, proliferation, recurrence and/or metastasis when the above-mentioned substances are used in the individual to which they are administered Can be inhibited, more specifically, at least 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95% of tumor growth, proliferation, recurrence and/or metastasis or 99% partially suppressed.
本发明所提供的抗CLD18A2的单域抗体对于Claudin18.2具有良好的亲和力,且可以用于进一步构建双特异性抗体、抗体-药物复合物、嵌合抗原受体等,且构建获得的双特异性抗体、抗体-药物复合物、CAR-T细胞等对于靶标细胞具有良好的靶向性和杀伤效果,具有良好的产业化前景。The anti-CLD18A2 single domain antibody provided by the present invention has good affinity for Claudin18.2, and can be used to further construct bispecific antibodies, antibody-drug complexes, chimeric antigen receptors, etc. Antibodies, antibody-drug complexes, CAR-T cells, etc. have good targeting and killing effects on target cells, and have good industrialization prospects.
下面通过实施例对本申请的发明予以进一步说明,但并不因此而限制本申请的范围。The invention of the present application is further illustrated by the following examples, but the scope of the present application is not limited thereby.
除非另外说明,本发明中所公开的实验方法、检测方法、制备方法均采用本技术领域常规的分子生物学、生物化学、染色质结构和分析、分析化学、细胞培养、重组DNA技术及相关领域的常规技术。这些技术在现有文献中已有完善说明,具体可参见Sambrook等MOLECULAR CLONING:A LABORATORY MANUAL,Second edition,Cold Spring Harbor Laboratory Press,1989 and Third edition,2001;Ausubel等,CURRENT PROTOCOLS IN MOLECULAR BIOLOGY,John Wiley & Sons,New York,1987 and periodic updates;the series METHODS IN ENZYMOLOGY,Academic Press,San Diego;Wolffe,CHROMATIN STRUCTURE AND FUNCTION,Third edition,Academic Press,San Diego,1998;METHODS IN ENZYMOLOGY,Vol.304,Chromatin(P.M.Wassarman and A.P.Wolffe,eds.),Academic Press,San Diego,1999;和METHODS IN MOLECULAR BIOLOGY,Vol.119,Chromatin Protocols(P.B.Becker,ed.)Humana Press,Totowa,1999等。Unless otherwise specified, the experimental methods, detection methods and preparation methods disclosed in the present invention all adopt the conventional molecular biology, biochemistry, chromatin structure and analysis, analytical chemistry, cell culture, recombinant DNA technology and related fields in the technical field. conventional technology. These techniques have been well described in the existing literature. For details, please refer to Sambrook et al. MOLECULAR CLONING: A LABORATORY MANUAL, Second edition, Cold Spring Harbor Laboratory Press, 1989 and Third edition, 2001; Ausubel et al., CURRENT PROTOCOLS IN MOLECULAR BIOLOGY, John Wiley & Sons, New York, 1987 and periodic updates; the series METHODS IN ENZYMOLOGY, Academic Press, San Diego; Wolfe, CHROMATIN STRUCTURE AND FUNCTION, Third edition, Academic Press, San Diego, 1998; METHODS IN ENZYMOLOGY, Vol.304, Chromatin (PMWassarman and APWolffe, eds.), Academic Press, San Diego, 1999; and METHODS IN MOLECULAR BIOLOGY, Vol. 119, Chromatin Protocols (PBBecker, ed.) Humana Press, Totowa, 1999, et al.
实施例1:抗CLD18A2的单域抗体的制备Example 1: Preparation of anti-CLD18A2 single domain antibody
构建人源Claudin18.2全长基因表达载体pCDNA3.1/CLDN18A2并用电转染CHO-S细胞,再通过G418筛选阳性克隆,获得表达Claudin18.2的CHO-S细胞株CHO-S-Claudin18.2。将CHO-S-Claudin18.2细胞皮下多点免疫羊驼并辅以佐剂,经过3到4次免疫,抽血检验抗体滴度满足要求后即采血提取PBMC,提取总RNA,反转录获取 cDNA,再用两轮PCR获得针对单域抗体VHH的基因序列,随后与噬菌体展示载体连接,转化展示载体pcomb3xss(Addgene plasmid#63890;RRID:Addgene_63890)中,通过T4连接酶(Takara)连接。将连接产物转化至电转感受态细胞ER2738中,构建抗Claudin18.2单域抗体文库。将构建的Claudin18.2单域抗体文库采用辅助噬菌体M13KO7(NEB)对其进行包装。将包装的噬菌体经过CHO-S和表达Claudin18.2的CHO-S进行差减杂交,以及多轮次的淘选,最终获得多个特异结合Claudin18.2的高亲和力单域抗体,在经过人源化改造,最后得到一组人源化的特异结合Claudin18.2的高亲和力单域抗体,如序列SEQ ID NO:67~90所示。The human Claudin18.2 full-length gene expression vector pCDNA3.1/CLDN18A2 was constructed and electrotransfected into CHO-S cells, and then positive clones were screened by G418 to obtain the CHO-S cell line CHO-S-Claudin18 expressing Claudin18.2. 2. CHO-S-Claudin18.2 cells were subcutaneously immunized with alpaca at multiple points and supplemented with adjuvant. After 3 to 4 times of immunization, blood was drawn to check that the antibody titer met the requirements, and then blood was drawn to extract PBMC, and total RNA was extracted and reverse transcribed. cDNA, and then two rounds of PCR were used to obtain the gene sequence against the single-domain antibody VHH, which was then ligated with a phage display vector, transformed into the display vector pcomb3xss (Addgene plasmad#63890; RRID:Addgene_63890), and ligated by T4 ligase (Takara). The ligation product was transformed into electrotransformation competent cells ER2738 to construct an anti-Claudin18.2 single domain antibody library. The constructed Claudin18.2 single domain antibody library was packaged with helper phage M13KO7 (NEB). The packaged phage was subjected to subtractive hybridization between CHO-S and CHO-S expressing Claudin18.2, as well as multiple rounds of panning, and finally multiple high-affinity single-domain antibodies that specifically bind to Claudin18.2 were obtained. Finally, a group of humanized high-affinity single-domain antibodies that specifically bind to Claudin18.2 are obtained, as shown in the sequences of SEQ ID NOs: 67-90.
用高保真酶GVP8(通用生物系统(安徽)有限公司)分别以合成的人源化序列为模板进行PCR扩增,在序列5’端引入信号肽序列,3’端引入6xHis组氨酸标签编码序列,PCR产物电泳并切胶回收约500bp左右的条带,将回收的PCR产物与内切酶NdeI和EcoRI酶切的pET32a+载体(Novagen)用重组试剂盒(近岸蛋白质科技有限公司)进行重组连接,构建大肠杆菌表达质粒,转化大肠杆菌感受态Top10F’,涂布氨苄抗性平板,培养箱37℃培养过夜。分别挑取氨苄抗性平板上的克隆,提质粒测序,确定序列在pET32a+载体上的正确插入。The high-fidelity enzyme GVP8 (General Biosystems (Anhui) Co., Ltd.) was used for PCR amplification with the synthetic humanized sequence as a template, a signal peptide sequence was introduced at the 5' end of the sequence, and a 6xHis histidine tag code was introduced at the 3' end. Sequence, PCR product electrophoresis and gel cutting to recover a band of about 500 bp, and the recovered PCR product was recombined with the pET32a+ vector (Novagen) digested with endonuclease NdeI and EcoRI with a recombination kit (Nearshore Protein Technology Co., Ltd.) Connected, constructed E. coli expression plasmid, transformed into E. coli competent Top10F', coated with ampicillin resistance plate, and cultivated overnight in an incubator at 37°C. The clones on the ampicillin-resistant plate were respectively picked, and the plasmids were extracted and sequenced to confirm the correct insertion of the sequence on the pET32a+ vector.
将测序确定的大肠杆菌表达质粒,转化大肠杆菌表达宿主Rosetta(DE3),构建大肠杆菌表达菌株。在氨苄抗性平板上挑取重组克隆、培养,并用1mM的IPTG 30℃诱导表达过夜。将诱导表达过夜的菌液进行超声破碎,12000g 4℃离心10分钟后,取上清,用Ni柱(博格隆生物技术有限公司)进行纯化,最终蛋白纯度达到90%以上。The E. coli expression plasmid determined by sequencing was transformed into the E. coli expression host Rosetta (DE3) to construct an E. coli expression strain. Recombinant clones were picked on ampicillin-resistant plates, cultured, and induced to express overnight with 1 mM IPTG at 30°C. The bacterial liquid induced to express overnight was sonicated, centrifuged at 12,000g at 4°C for 10 minutes, and the supernatant was taken and purified with a Ni column (Borgron Biotechnology Co., Ltd.), and the final protein purity reached more than 90%.
通过细胞ELISA测定的亲和力见表1。具体的,将CHO-S-CLD18A2以5×10 5/ml个细胞每孔在96孔板进行铺板,并用3%BSA室温封闭1小时,将纯化的组氨酸标签融合的CLD18A2纳米抗体经用1%BSA梯度稀释后分别加入封闭后的细胞,室温孵育1小时。洗涤之后加入100μl/孔1:5000稀释的mouse anti his tag抗体(R&D Systems,Inc),室温孵育1小时。洗涤后加入1:10000稀释的HRP-Goat anti mouse IgG抗体(Thermo Scientific),每孔100μl加入,室温孵育1小时。洗涤之后加TMB显色,450nm下检测OD值。应用软件GraphPad Prism v5.0进行数据处理和作图分析,得到Anti-CLD18A2的人源化纳米抗体对细胞中的CLD18A2的EC50值以反映抗体对CLD18A2的亲和能力。 Affinities determined by cellular ELISA are shown in Table 1. Specifically, CHO-S-CLD18A2 was plated in a 96-well plate at 5×10 5 /ml cells per well, and blocked with 3% BSA for 1 hour at room temperature. The purified histidine tag-fused CLD18A2 nanobody was used 1% BSA gradient dilution was added to the blocked cells, and incubated at room temperature for 1 hour. After washing, 100 μl/well of mouse anti his tag antibody (R&D Systems, Inc) diluted at 1:5000 was added and incubated for 1 hour at room temperature. After washing, 1:10000 diluted HRP-Goat anti mouse IgG antibody (Thermo Scientific) was added, 100 μl per well, and incubated at room temperature for 1 hour. After washing, TMB was added for color development, and the OD value was detected at 450 nm. The software GraphPad Prism v5.0 was used for data processing and graph analysis, and the EC50 value of Anti-CLD18A2 humanized nanobody to CLD18A2 in cells was obtained to reflect the affinity of the antibody to CLD18A2.
表1Table 1
实施例2:抗CLD18A2/抗CD3双特异性抗体及对照品的制备Example 2: Preparation of anti-CLD18A2/anti-CD3 bispecific antibody and reference substance
表2Table 2
按照表2所示相关序列,S4-14双特异抗体根据CHO细胞密码子偏好性分别将氨基酸序列转换成各碱基序列,通过基因合成(南京金斯瑞生物科技有限公司)获得全长DNA。以各个DNA为模板,用高保真酶GVP8(安徽通用生物技术有限公司)进行PCR扩增,经HindIII/EcoRI酶切后插入pCDNA3.1载体进行重组连接构建两个表达质粒(分别表达SEQ ID NO:115和SEQ ID NO:116序列),用去内毒素质粒大抽试剂盒(Biomiga)提取两个质粒并按1:1混合,将混合质粒与转染试剂PEI(Polysciences,Inc.) 1:3混合均匀后静置30min,然后加入到HEK293F细胞中共转染,37℃,5%CO 2摇床培养箱中培养7天后,离心取上清。 According to the relevant sequences shown in Table 2, the S4-14 bispecific antibody converted the amino acid sequence into each base sequence according to the codon preference of CHO cells, and obtained the full-length DNA by gene synthesis (Nanjing GenScript Biotechnology Co., Ltd.). Take each DNA as a template, carry out PCR amplification with high-fidelity enzyme GVP8 (Anhui General Biotechnology Co., Ltd.), insert pCDNA3.1 vector after HindIII/EcoRI digestion, and recombine and connect to construct two expression plasmids (expressing SEQ ID NO. : 115 and SEQ ID NO: 116 sequence), the two plasmids were extracted with the endotoxin-removing plasmid large extraction kit (Biomiga) and mixed 1:1, and the mixed plasmid was mixed with the transfection reagent PEI (Polysciences, Inc.) 1: 3 Mix well and let it stand for 30 min, then add it to HEK293F cells for co-transfection, culture in a 37°C, 5% CO 2 shaker incubator for 7 days, and then centrifuge to get the supernatant.
准备样品:取培养液1L,5000rcf离心20分钟,分离沉淀,取上清。然后,用0.22μm微孔滤膜过滤除去细胞碎片,检测样品浊度在20NTU以下,留样1ml;Prepare samples: take 1L of culture medium, centrifuge at 5000rcf for 20 minutes, separate the precipitate, and take the supernatant. Then, filter the cell debris with a 0.22 μm microporous membrane, and check that the turbidity of the sample is below 20 NTU, and retain 1 ml of the sample;
亲和层析:用0.1M NaOH以10ml/min流速处理50ml Protein A层析柱(博格隆,AA0273),30min,以除去柱上残留的蛋白。然后用150mM NaCl 20mM PB(pH6.5)平衡至少3个柱体积,直至pH达到6.5,电导在15mS/cm左右。将培养液滤液以10ml/min的流速经过Protein A层析柱,柱上停留时间约5min,然后用150mM NaCl 20mM PB(pH6.5)平衡至少3个柱体积。用500mM NaCl 20mM PB(pH6.5)平衡3个柱体积,然后用100mM Arg-HAc(pH5.0)平衡3个柱体积。用100mM Gly-HCl(pH3.2)洗脱,根据280nm吸收值收集洗脱峰,得到的样品,按10%的体积加入1.0M NaCit-HCit(pH6.5),轻轻混匀。Affinity chromatography: 50ml Protein A chromatography column (Borgron, AA0273) was treated with 0.1M NaOH at a flow rate of 10ml/min for 30min to remove residual protein on the column. It was then equilibrated with 150
阴离子交换层析:用0.5M NaOH以10ml/min流速处理50ml Q FF层析柱(博格隆,AI0024),30min,以除去柱上残留的蛋白。然后用20mM PB(pH6.5)平衡至少5个柱体积,直至pH达到6.5。将亲和层析得到的样品,以10ml/min的流速经过Q FF层析柱,柱上停留时间约5min,收集并保存流穿样品。然后用500mM NaCl 20mM PB(pH6.5)清洗,除去结合在柱上的杂质,并鉴定。Anion exchange chromatography: 50ml Q FF chromatography column (Borgron, AI0024) was treated with 0.5M NaOH at a flow rate of 10ml/min for 30min to remove residual protein on the column. It was then equilibrated with 20 mM PB (pH 6.5) for at least 5 column volumes until the pH reached 6.5. The sample obtained by affinity chromatography was passed through the Q FF chromatography column at a flow rate of 10ml/min, the residence time on the column was about 5min, and the flow-through sample was collected and stored. It was then washed with 500
阳离子交换层析:用0.5M NaOH以10ml/min流速处理50ml SP mustang层析柱(博格隆,AI0192),30min,以除去柱上残留的蛋白。然后用20mM PB(pH6.5)平衡至少5个柱体积,直至pH达到6.5。将阴离子交换层析得到的流穿样品,以10ml/min的流速经过SP mustang层析柱,柱上停留时间约5min。然后用200mM NaCl 20mM PB(pH6.5)洗脱,收集洗脱样品。Cation exchange chromatography: 50ml SP mustang chromatography column (Borgron, AI0192) was treated with 0.5M NaOH at a flow rate of 10ml/min for 30min to remove residual protein on the column. It was then equilibrated with 20 mM PB (pH 6.5) for at least 5 column volumes until the pH reached 6.5. The flow-through sample obtained by anion exchange chromatography was passed through the SP mustang chromatography column at a flow rate of 10ml/min, and the residence time on the column was about 5min. It was then eluted with 200
保存样品:将阳离子交换层析的洗脱样品,0.22μm无菌过滤,4℃保存。Preservation of samples: The eluted samples of cation exchange chromatography were sterile filtered at 0.22 μm and stored at 4°C.
按照表2所示相关序列,S2-12双特异抗体,以及阴性对照品Anti-C18.2-hu19V3和Anti-CD3根据实施例1中提供的载体构建、表达和纯化方法进行样品制备。According to the related sequences shown in Table 2, the S2-12 bispecific antibody, and the negative controls Anti-C18.2-hu19V3 and Anti-CD3 were prepared according to the vector construction, expression and purification methods provided in Example 1.
上述所有制备的4个样品,其纯度检测使用SEC-HPLC-UV分析。检测器:Agilent 1100LC;检测波长:214nm;流动相:150mM pH7.0PB+5%异丙醇;色谱柱:Superdex 200Increase 5/150GL;运行时间:15分钟;柱温25℃。纯度都大于95%。All 4 samples prepared above were tested for purity using SEC-HPLC-UV analysis. Detector: Agilent 1100LC; Detection wavelength: 214nm; Mobile phase: 150mM pH7.0PB+5% isopropanol; Chromatographic column: Superdex 200Increase 5/150GL; Running time: 15 minutes;
实施例3:鉴定抗CLD18A2/抗CD3双特异性抗体的功能Example 3: Identification of the function of anti-CLD18A2/anti-CD3 bispecific antibodies
3.1抗CLD18A2/抗CD3双特异性抗体的细胞活性3.1 Cellular activity of anti-CLD18A2/anti-CD3 bispecific antibodies
荧光素酶检测细胞的构建:Jurkat细胞中转入NFAT响应的荧光素酶系统,获得检测细胞株Jurkat-PB-NFAT-luc2p。Construction of luciferase detection cells: Jurkat cells were transformed into NFAT-responsive luciferase system, and the detection cell line Jurkat-PB-NFAT-luc2p was obtained.
靶细胞铺板:取适量悬浮培养的的细胞CHO-S-Claudin18.2,800rpm离心5min,弃去上清,加入RPMI1640培养基(含FBS)重悬,计数,取所需细胞稀释至指定浓度,加入到96孔细胞培养板中。Target cell plating: Take an appropriate amount of CHO-S-Claudin18.2 cells cultured in suspension, centrifuge at 800 rpm for 5 min, discard the supernatant, add RPMI1640 medium (containing FBS) to resuspend, count, take the required cells and dilute to the specified concentration, Add to 96-well cell culture plates.
蛋白稀释:用RPMI1640培养基(含FBS)梯度稀释制备的S2-12、S4-14双特异抗体,以及阴性对照品Anti-C18.2-hu19V3和Anti-CD3至指定浓度,然后取适量体积加入到96孔板内,靶细胞与抗体蛋白37℃孵育30min后加入Jurkat-PB-NFAT-luc2p细胞。Protein dilution: S2-12 and S4-14 bispecific antibodies prepared by gradient dilution in RPMI1640 medium (containing FBS), as well as negative controls Anti-C18.2-hu19V3 and Anti-CD3 to the specified concentration, and then add an appropriate volume In a 96-well plate, target cells were incubated with antibody protein for 30 min at 37°C, and Jurkat-PB-NFAT-luc2p cells were added.
Jurkat-PB-NFAT-luc2p铺板:取适量Jurkat-PB-NFAT-luc2p细胞稀释至指定浓度,加入到96孔细胞培养板中。孵育20h左右进行检测。Jurkat-PB-NFAT-luc2p plating: Dilute an appropriate amount of Jurkat-PB-NFAT-luc2p cells to the specified concentration and add them to a 96-well cell culture plate. Incubate for about 20h for detection.
光度值检测:加入萤荧光素酶检测试剂盒(Promega,E2620)提供的检测溶液10μl/孔,震荡2min,转移溶液60μl至96孔白板中,于酶标仪中检测。应用软件GraphPad Prism v5.0进行数据处理和作图分析,得到各种抗CLD18A2/抗CD3双特异性抗体对该检测体系的激活曲线和EC50值,如图1所示,S2-12、S4-14两种不同结构的双特异抗体产生而相似的体外T细胞系统的激活能力,而阴性对照Anti-C18.2-hu19V3和Anti-CD3不具有激活该检测系统的能力。说明抗CLD18A2抗体和抗CD3抗体只有在形成双特异抗体时才具有对Claudin18.2抗原靶细胞和T细胞效应系统产生激活作用,而单独的抗体都不具备该功能。Photometric value detection: add 10 μl/well of the detection solution provided by the luciferase detection kit (Promega, E2620), shake for 2 min,
3.2抗CLD18A2/抗CD3双特异抗体的体外细胞杀伤检测3.2 In vitro cell killing assay of anti-CLD18A2/anti-CD3 bispecific antibodies
为了评估抗CLD18A2/抗CD3双特异抗体的细胞杀伤效果,本发明采用T细胞(妙通生物)作为效应细胞进行细胞毒性试验。In order to evaluate the cell killing effect of the anti-CLD18A2/anti-CD3 bispecific antibody, the present invention uses T cells (Miaotong Biotechnology) as effector cells to conduct a cytotoxicity test.
抗S2-12、S4-14双特异抗体,以及阴性对照品Anti-C18.2-hu19V3和Anti-CD3分别梯度稀释,每孔加入50μl。Claudin18.2及Claudin18.1稳转细胞NUGC-4-Claudin18.2和NUGC-4-Claudin18.1分别用5%FBS 1640培养基(Gibco)清洗重悬,配置成约2×10 5/ml的细胞密度,每孔加入50μl至对应的96孔板中。将来自健康供体的人T淋巴细胞重悬在5%FBS 1640培养基中,每孔加入1×10 5个每孔,使效靶比为10:1,37℃培养箱孵育4小时后按LDH检测试剂盒(东仁化学科技(上海)有限公司)检测LDH释放量,评估抗Claudin18.2/抗CD3双特异抗体的体外细胞杀伤效果。 Anti-S2-12, S4-14 bispecific antibodies, as well as negative controls Anti-C18.2-hu19V3 and Anti-CD3 were serially diluted, and 50 μl was added to each well. Claudin18.2 and Claudin18.1 stably transfected cells NUGC-4-Claudin18.2 and NUGC-4-Claudin18.1 were washed and resuspended in 5% FBS 1640 medium (Gibco), respectively, to a concentration of about 2×10 5 /ml For cell density, add 50 μl per well to the corresponding 96-well plate. Human T lymphocytes from healthy donors were resuspended in 5% FBS 1640 medium, and 1 × 10 5 cells were added to each well to make the effect-target ratio 10:1. After incubating at 37°C for 4 hours, press The LDH detection kit (Dongren Chemical Technology (Shanghai) Co., Ltd.) detected the release of LDH and evaluated the in vitro cell killing effect of anti-Claudin18.2/anti-CD3 bispecific antibodies.
在体外细胞毒性实验中,抗CLD18A2/抗CD3双特异抗体S2-12和S4-14对Claudin18.2 高表达的NUGC-4-Claudin18.2杀伤效果非常明显,EC50值分别为24.65pM、21.15pM;对于NUGC-4-Claudin18.1细胞,抗CLD18A2/抗CD3双特异抗体均没有明显的杀伤效果(如图2所示)。说明抗S2-12和S4-14双特异抗体在体外实验中,在T淋巴细胞参与下,对NUGC-4-Claudin18.2细胞具有特异性杀伤效果,对不表达Claudin18.2的细胞不产生毒性,体现了高度杀伤特异性。In the in vitro cytotoxicity experiments, the anti-CLD18A2/anti-CD3 bispecific antibodies S2-12 and S4-14 had a very significant killing effect on NUGC-4-Claudin18.2 with high Claudin18.2 expression, with EC50 values of 24.65pM and 21.15pM, respectively. ; For NUGC-4-Claudin18.1 cells, anti-CLD18A2/anti-CD3 bispecific antibodies had no obvious killing effect (as shown in Figure 2). It shows that the anti-S2-12 and S4-14 bispecific antibodies have specific killing effect on NUGC-4-Claudin18.2 cells in the in vitro experiments with the participation of T lymphocytes, and have no toxicity to cells that do not express Claudin18.2 , reflecting a high degree of killing specificity.
3.3抗CLD18A2/抗CD3双特异抗体的肿瘤抑制活性3.3 Tumor inhibitory activity of anti-CLD18A2/anti-CD3 bispecific antibodies
本发明采用病人来源的胃癌组织建立的异种移植瘤模型(patientl derived xenograft,PDX)的荷瘤NSG小鼠,分析抗Claudin18.2/抗CD3双特异抗体的肿瘤抑制作用。肿瘤长至100mm 3左右时,将荷瘤小鼠随机分配,每组5只,腹腔注射给予2x10 7健康人PBMC细胞。一天后,荷瘤小鼠腹腔注射5μg(25μg/ml,200μl PBS),双特异抗体S2-12,每两日一次,持续4周,每周记录两次肿瘤体积。以及注射S4-14,10μg(25μg/ml,200μl PBS)每周一次,持续4周,每周记录两次肿瘤体积。 In the present invention, tumor-bearing NSG mice of a xenograft tumor model (patientl derived xenograft, PDX) established by gastric cancer tissue derived from patients are used to analyze the tumor suppressive effect of anti-Claudin18.2/anti-CD3 bispecific antibody. When the tumor grew to about 100mm 3 , the tumor-bearing mice were randomly assigned to 5 mice in each group, and 2×10 7 healthy human PBMC cells were injected intraperitoneally. One day later, tumor-bearing mice were intraperitoneally injected with 5 μg (25 μg/ml, 200 μl PBS), bispecific antibody S2-12, once every two days for 4 weeks, and the tumor volume was recorded twice a week. As well as injection of S4-14, 10 μg (25 μg/ml, 200 μl PBS) once a week for 4 weeks, tumor volume was recorded twice a week.
由实验结果图3可以看出随着时间的推移,实验组肿瘤体积逐渐减小,双特异抗体S2-12和S4-14对移植瘤都具有明显的生长抑制作用。It can be seen from the experimental results in Figure 3 that the tumor volume of the experimental group gradually decreased over time, and both bispecific antibodies S2-12 and S4-14 had obvious growth inhibitory effects on the transplanted tumors.
实施例4:抗CLD18A2单域抗体融合蛋白与对照抗体的制备Example 4: Preparation of anti-CLD18A2 single-domain antibody fusion protein and control antibody
4.1抗CLD18A2单域抗体融合蛋白的制备4.1 Preparation of anti-CLD18A2 single-domain antibody fusion protein
分别以筛选获得的特异性阳性序列和人源化序列为模板,上游引物5’-gtgctgctgctgtgggtgccaggatccaccgggcaggtgcagctcgtggagtc-3’(SEQ ID NO.141)和下游引物5’-gcaggacttgggctcagaagacacggtgaccagggtcccctggcc-3’(SEQ ID NO.142),用高保真酶GVP8(安徽通用生物技术有限公司)进行PCR扩增,PCR产物电泳并切胶回收约400bp左右的条带,将回收的PCR产物与含有信号肽和人IgG1Fc序列(氨基酸序列SEQ NO.119)的pCDNA3.1载体进行重组连接,构建Anti-CLD18A2的单域抗体与人IgG1Fc融合的细胞表达质粒,用去内毒素质粒大抽试剂盒(Biomiga)提取抗CLD18A2的纳米抗体人IgG1Fc融合的细胞表达质粒,将质粒与转染试剂PEI(Polysciences,Inc.)1:3混合均匀后静置30min,然后加入到HEK293F细胞中,37℃,5%CO
2摇床培养箱中培养7天后,离心取上清。将上清调至pH7.0后上样ProteinA亲和层析柱(博格隆生物技术有限公司),100%0.1M Gly-HCl(pH3.0)洗脱;洗脱液预先加入10%1M Tris-HCl(pH8.5)。100%洗脱液稀释至电导4ms/cm,调pH5.5后,离心(8000rpm,4℃,10min),上清液调pH至5.0后上样至DSP层析柱(博格隆生物技术有限公司),0-60%洗脱液(20mM NaAc,0.5M NaCl,pH5.0)线 性洗脱,流速2ml/min,15min。
The specific positive sequences and humanized sequences obtained by screening were used as templates, the upstream primer 5'-gtgctgctgctgtgggtgccaggatccaccgggcaggtgcagctcgtggagtc-3' (SEQ ID NO.141) and the downstream primer 5'-gcaggacttgggctcagaagacacggtgaccagggtcccctggcc-3' (SEQ ID NO.142) , use high-fidelity enzyme GVP8 (Anhui General Biotechnology Co., Ltd.) to carry out PCR amplification, the PCR product is electrophoresed and cut into the gel to recover a band of about 400bp, and the recovered PCR product is mixed with a signal peptide and human IgG1Fc sequence (amino acid sequence SEQ NO.119) pCDNA3.1 vector was recombined to construct a cell expression plasmid fused with Anti-CLD18A2 single domain antibody and human IgG1Fc, and the anti-CLD18A2 nanobody human IgG1Fc was extracted with endotoxin removal plasmid kit (Biomiga). The fused cells express the plasmid, mix the plasmid with the transfection reagent PEI (Polysciences, Inc.) 1:3 and let stand for 30 min, then add it to HEK293F cells, and culture at 37°C, 5% CO 2 in a shaking incubator for 7 Days later, the supernatant was collected by centrifugation. The supernatant was adjusted to pH 7.0 and loaded onto a ProteinA affinity chromatography column (Borgron Biotechnology Co., Ltd.), eluted with 100% 0.1M Gly-HCl (pH 3.0); the eluent was pre-added with 10% 1M Tris-HCl (pH 8.5). The 100% eluate was diluted to a conductivity of 4ms/cm, adjusted to pH 5.5, centrifuged (8000rpm, 4°C, 10min), and the supernatant was adjusted to pH 5.0 and loaded onto a DSP column (Borgron Biotechnology Co., Ltd. Company), 0-60% eluent (20 mM NaAc, 0.5 M NaCl, pH 5.0) linear elution, flow
4.2阳性对照抗体ch-175D10的表达和纯化4.2 Expression and purification of positive control antibody ch-175D10
以US9751934B2中的序列号118的重链和序列号125的轻链构成的嵌合抗体(US9751934B2专利中的名称为ch-175D10)作为对照抗体,将其氨基酸序列的对应多核苷酸序列与pCDNA3.1载体进行重组连接,通过实施例2相同的方法进行HEK293F细胞的瞬时转染表达和纯化。The chimeric antibody (named ch-175D10 in the US9751934B2 patent) composed of the heavy chain of SEQ ID NO: 118 and the light chain of SEQ ID NO: 125 in US9751934B2 was used as a control antibody, and the corresponding polynucleotide sequence of its amino acid sequence was compared with that of pCDNA3. 1. The vector was recombined and ligated, and the transient transfection, expression and purification of HEK293F cells were carried out by the same method as in Example 2.
实施例5:抗CLD18A2单域抗体融合蛋白内吞活性检测Example 5: Detection of endocytic activity of anti-CLD18A2 single-domain antibody fusion protein
将CLD18A2(SEQ ID NO.143)全长基因和gfp基因用内部核糖体进入位点(IRES)序列连接,并构建在pCDNA3.1载体(Life Technologies),以实现CLD18A2和gfp的共表达。提取表达质粒,使用Lipofectamine 3000(Invitrogen,L3000001)转染试剂,根据说明书的操作转染CHO-K1细胞。第二天,将转染的CHO-K1细胞用胰酶消化后,以终浓度为2×10 6/ml细胞接种在96孔板培养。将pH敏感的荧光染料(<pH7可被激发出荧光)与抗体偶联,当标记抗体通过受体等介导内吞后在酸性环境下可以被激发产生荧光,通过产生荧光强弱判断内吞效率。第三天,去除96孔板培养上清,并加入经pH敏感荧光素标记的Anti-C18.2-Fc融合蛋白(所有8个纳米抗体的人源化抗体V3的FC融合蛋白)和对照抗体(步骤均按照pHAb Amine Reactive Dye试剂盒中的说明书进行)的DMEM稀释液,荧光标记抗体的终浓度为10μg/ml,冰上孵育1小时,用预冷的DMEM洗涤三次。其中一块板上的孔在冰上保持,做为内吞0小时样品,余下的在37℃培养箱孵育,分别在3小时取出,冰上预冷,终止内吞。将所有样品进行荧光检测。结果见图4和表3: The full-length gene of CLD18A2 (SEQ ID NO. 143) and the gfp gene were linked with an internal ribosome entry site (IRES) sequence and constructed in pCDNA3.1 vector (Life Technologies) to achieve co-expression of CLD18A2 and gfp. The expression plasmid was extracted, and Lipofectamine 3000 (Invitrogen, L3000001) transfection reagent was used to transfect CHO-K1 cells according to the instructions. The next day, the transfected CHO-K1 cells were trypsinized and then seeded in a 96-well plate at a final concentration of 2×10 6 /ml cells. Conjugate pH-sensitive fluorescent dyes (<pH7 can be excited to fluoresce) with antibodies. When the labeled antibody is endocytosed through receptors, etc., it can be excited to generate fluorescence in an acidic environment, and the endocytosis can be judged by the intensity of fluorescence generated. efficient. On the third day, the culture supernatant of the 96-well plate was removed, and pH-sensitive fluorescein-labeled Anti-C18.2-Fc fusion protein (FC fusion protein of humanized antibody V3 of all 8 Nanobodies) and control antibody were added (The steps were all performed according to the instructions in the pHAb Amine Reactive Dye kit) DMEM dilution, the final concentration of fluorescently labeled antibody was 10 μg/ml, incubated on ice for 1 hour, and washed three times with pre-cooled DMEM. The wells on one of the plates were kept on ice as samples for endocytosis for 0 hours, and the rest were incubated in a 37°C incubator for 3 hours, respectively, and pre-cooled on ice to stop endocytosis. All samples were subjected to fluorescence detection. The results are shown in Figure 4 and Table 3:
表3table 3
上述这些结果表明,本发明中选取的人源化抗体是内吞抗体,能特异性结合CLD18A2并介导内吞,而同型对照抗体无明显内吞。The above results show that the humanized antibody selected in the present invention is an endocytic antibody, which can specifically bind to CLD18A2 and mediate endocytosis, while the isotype control antibody has no obvious endocytosis.
实施例6抗CLDN18A2单域抗体-药物复合物的制备Example 6 Preparation of anti-CLDN18A2 single domain antibody-drug complex
6.1抗CLDN18A2单域抗体融合蛋白的制备6.1 Preparation of anti-CLDN18A2 single-domain antibody fusion protein
hu19V3-hu19V3-ABD-(GGC) 6(Ab1) hu19V3-hu19V3-ABD-(GGC) 6 (Ab1)
分别以筛选获得的特异性阳性序列Anti-C18.2-19的人源化序列Anti-C18.2-hu19V3为基础进行设计,将Anti-C18.2-hu19V3进行两次串联,并在C末端引入人血清白蛋白结合序列(ABD)和GGC三个氨基酸的6个重复结构,且完整序列如SEQ ID NO.125所示。在通用生物系统(安徽)有限公司合成编码SEQ ID NO.125所示氨基酸序列的完整碱基序列,并构建在pET32a载体,将表达质粒,转化大肠杆菌表达宿主Rosetta(DE3),构建大肠杆菌表达菌株。在氨苄抗性平板上挑取重组克隆、培养,并用1mM的IPTG 30℃诱导表达过夜。将诱导表达过夜的菌液进行超声破碎,12000g 4℃离心10分钟后,取上清,用Ni柱(博格隆生物技术有限公司)进行纯化,最终蛋白纯度达到90%以上。The design was based on the humanized sequence Anti-C18.2-hu19V3 of the specific positive sequence Anti-C18.2-19 obtained by screening, and the Anti-C18.2-hu19V3 was concatenated twice, and the C-terminal A six-repeat structure of three amino acids of human serum albumin binding sequence (ABD) and GGC is introduced, and the complete sequence is shown in SEQ ID NO.125. The complete base sequence encoding the amino acid sequence shown in SEQ ID NO.125 was synthesized in Universal Biosystems (Anhui) Co., Ltd., and constructed in the pET32a vector, and the expression plasmid was transformed into E. coli expression host Rosetta (DE3) to construct E. coli expression strains. Recombinant clones were picked on ampicillin-resistant plates, cultured, and induced to express overnight with 1 mM IPTG at 30°C. The bacterial liquid induced to express overnight was sonicated, centrifuged at 12,000g at 4°C for 10 minutes, and the supernatant was taken and purified with a Ni column (Borgron Biotechnology Co., Ltd.), and the final protein purity reached more than 90%.
hu19V3-hu19V3-PAEC6(Ab2)hu19V3-hu19V3-PAEC6(Ab2)
分别以筛选获得的特异性阳性序列Anti-C18.2-19的人源化序列Anti-C18.2-hu19V3为基础进行设计,将Anti-C18.2-hu19V3进行两次串联,在通用生物系统(安徽)有限公司合成编码如SEQ ID NO.155所示氨基酸序列的碱基序列,同时合成编码如SEQ ID NO.124所示的PAEC序列,将PAEC序列进行6个重复串联的构建(PAEC6),将PAEC6连接在Anti-C18.2-hu19V3序列的C端,构建成如SEQ ID NO.126所示完整氨基酸的碱基序列,并将完整序列构建在pET32a载体,将表达质粒,转化大肠杆菌表达宿主Rosetta(DE3),构建大肠杆菌表达菌株。在氨苄抗性平板上挑取重组克隆、培养,并用1mM的IPTG 30℃诱导表达过夜。将诱导表达过夜的菌液进行超声破碎,12000g 4℃离心10分钟后,取上清,用Ni柱(博格隆生物技术有限公司)进行纯化,最终蛋白纯度达到90%以上。The design was based on the humanized sequence Anti-C18.2-hu19V3 of the specific positive sequence Anti-C18.2-19 obtained by screening, and the Anti-C18.2-hu19V3 was concatenated twice. (Anhui) Co., Ltd. synthesized the base sequence encoding the amino acid sequence shown in SEQ ID NO.155, and at the same time synthesized the PAEC sequence encoding the amino acid sequence shown in SEQ ID NO.124, and constructed the PAEC sequence with 6 repeats in series (PAEC6) , connect PAEC6 to the C-terminus of Anti-C18.2-hu19V3 sequence, construct a complete amino acid base sequence as shown in SEQ ID NO.126, and construct the complete sequence in pET32a vector, and transform the expression plasmid into Escherichia coli The expression host Rosetta (DE3) was used to construct an E. coli expression strain. Recombinant clones were picked on ampicillin-resistant plates, cultured, and induced to express overnight with 1 mM IPTG at 30°C. The bacterial liquid induced to express overnight was sonicated, centrifuged at 12,000g at 4°C for 10 minutes, and the supernatant was taken and purified with a Ni column (Borgron Biotechnology Co., Ltd.), and the final protein purity reached more than 90%.
Anti-C18.2-hu19V3-Fc(Ab3)Anti-C18.2-hu19V3-Fc(Ab3)
制备如实施例4所示的SEQ ID NO.147的序列。The sequence of SEQ ID NO. 147 was prepared as shown in Example 4.
6.2抗体-药物复合物的制备6.2 Preparation of antibody-drug complexes
纯化的抗CLDN18A2单域抗体融合蛋白以及ch-175D10溶于PBS溶液,在25℃下,加入10倍过量的TCEP还原链间二硫键,透析除去还原剂。链间二硫键重新形成可通过在25℃下加入2倍TCEP摩尔浓度的CuSO4获得。然后加入10倍抗体摩尔浓度的MC-VC-PAB-MMAE,25℃下反应1小时,形成抗体衍生物Ab-MC-VC-PAB-MMAE复合物。复合物经透析或超滤去除未结合的MC-VC-PAB-MMAE。最终获得的产物用LC/MS测定DAR。The purified anti-CLDN18A2 single-domain antibody fusion protein and ch-175D10 were dissolved in PBS solution, and 10-fold excess TCEP was added to reduce the interchain disulfide bonds at 25°C, and the reducing agent was removed by dialysis. Re-formation of interchain disulfide bonds can be obtained by adding 2 times the molar concentration of TCEP to CuSO4 at 25 °C. Then, MC-VC-PAB-MMAE with a molar concentration of 10 times the antibody was added and reacted at 25°C for 1 hour to form the antibody derivative Ab-MC-VC-PAB-MMAE complex. The complex was dialyzed or ultrafiltered to remove unbound MC-VC-PAB-MMAE. The DAR of the final product obtained was determined by LC/MS.
所得Ab1-MC-VC-PAB-MMAE的终浓度为1.2mg/ml,用LC-MS方法检测分析,证明所得样品中没有游离的毒素小分子。用分光光度计(UV方法)检测A252,A280吸收峰,测定所得毒素和抗体的比例DAR即y=4.42。分装于4℃储存备用。The final concentration of the obtained Ab1-MC-VC-PAB-MMAE was 1.2 mg/ml. The LC-MS method was used to detect and analyze, and it was proved that there were no free toxin small molecules in the obtained sample. The absorption peaks of A252 and A280 were detected by a spectrophotometer (UV method), and the ratio DAR of the obtained toxin and antibody was determined, that is, y=4.42. Store in aliquots at 4°C.
所得Ab2-MC-VC-PAB-MMAE的终浓度为2.5mg/ml,用LC-MS方法检测分析,证明所得样品中没有游离的毒素小分子。用分光光度计(UV方法)检测A252,A280吸收峰,测定所得毒素和抗体的比例DAR即y=4.10。分装于4℃储存备用。The final concentration of the obtained Ab2-MC-VC-PAB-MMAE was 2.5 mg/ml. The LC-MS method was used to detect and analyze, and it was proved that there were no free toxin small molecules in the obtained sample. The absorption peaks of A252 and A280 were detected by spectrophotometer (UV method), and the ratio DAR of the obtained toxin and antibody was determined, that is, y=4.10. Store in aliquots at 4°C.
所得Ab3-MC-VC-PAB-MMAE的终浓度为5.4mg/ml,用LC-MS方法检测分析,证明所得样品中没有游离的毒素小分子。用分光光度计(UV方法)检测A252,A280吸收峰,测定所得毒素和抗体的比例DAR即y=4.21。分装于4℃储存备用。The final concentration of the obtained Ab3-MC-VC-PAB-MMAE was 5.4 mg/ml. The LC-MS method was used for detection and analysis, which proved that there were no free toxin small molecules in the obtained sample. The absorption peaks of A252 and A280 were detected by spectrophotometer (UV method), and the ratio DAR of the obtained toxin and antibody was determined, that is, y=4.21. Store in aliquots at 4°C.
所得ch175D10-MC-VC-PAB-MMAE的终浓度为4.4mg/ml,用LC-MS方法检测分析,证明所得样品中没有游离的毒素小分子。用分光光度计(UV方法)检测A252,A280吸收峰,测定所得毒素和抗体的比例DAR即y=4.31。分装于4℃储存备用。The final concentration of the obtained ch175D10-MC-VC-PAB-MMAE was 4.4 mg/ml. The LC-MS method was used to detect and analyze, and it was proved that there were no free toxin small molecules in the obtained sample. The absorption peaks of A252 and A280 were detected by a spectrophotometer (UV method), and the ratio DAR of the obtained toxin and antibody was determined, that is, y=4.31. Store in aliquots at 4°C.
实施例7抗CLDN18A2单域抗体-药物复合物的结合活性检测Example 7 Detection of binding activity of anti-CLDN18A2 single-domain antibody-drug complexes
对制备的单域抗体-药物复合物进行ELISA结合活性检测:CLD18A2蛋白(Kactus Biosystems)包被于96孔酶标板,每孔100ng,4℃包被过夜,10mM Na 2CO 3-NaHCO 3(pH10.0)洗涤后,5%脱脂奶粉包被1小时,再次洗涤后,加入单域抗体-药物复合物;37℃孵育2小时后,加入生物素标记的抗单域抗体兔多抗(自制),再加入Strep-HRP孵育37℃,1h,洗涤后TMB显色。结果如表4所示,Ab3在药物偶联前后的结合活性未受影响,其余两个结构Ab1和Ab2在药物偶联后,依然具有较好的结合活性。 ELISA binding activity detection of the prepared single-domain antibody-drug complexes: CLD18A2 protein (Kactus Biosystems) was coated on a 96-well microtiter plate, 100 ng per well, overnight at 4°C, 10 mM Na 2 CO 3 -NaHCO 3 ( After washing at pH 10.0), 5% nonfat milk powder was coated for 1 hour, and after washing again, the single-domain antibody-drug complex was added; after incubation at 37°C for 2 hours, biotin-labeled anti-single-domain antibody rabbit polyclonal antibody (self-made) was added. ), and then added Strep-HRP to incubate at 37°C for 1 h. After washing, TMB developed color. The results are shown in Table 4. The binding activity of Ab3 before and after drug conjugation was not affected, and the remaining two structures, Ab1 and Ab2, still had good binding activity after drug conjugation.
表4Table 4
实施例8体外细胞毒性测试Example 8 In vitro cytotoxicity test
体外细胞毒性实验的检测采用过表达人CLD18A2的NUGC-4细胞(NUGC-4-CLD18A2)和原始NUGC-4进行:将生长至90%汇合度的细胞培养基,用PBS清洗,并用胰酶消化,终止消化后,收集细胞,并接种于96孔板,2x10 4/well,10%胎牛血清、2mM谷氨酸盐的DMEM培养基,于5%CO 2培养箱中培养过夜。第二天,去除培养基,并用含有不同浓度的抗CLD18A2单域抗体-药物复合物及其相应的单域抗体融合蛋白的新鲜培养基代替,每孔50μl,每个浓度三个复孔;37℃,5%CO 2孵育72小时,取出上清用LDH试剂盒(购自东仁化学科技(上海)有限公司,货号CK12)检测LDH的释放。检测方法按照说明书进行。细胞杀伤百分比(%)=100*(OD490待测样品-OD490对照孔)/(OD490细胞全部裂解-OD490对照孔)。实验结果如图5和表5所示。 In vitro cytotoxicity assays were performed using NUGC-4 cells overexpressing human CLD18A2 (NUGC-4-CLD18A2) and original NUGC-4: cells grown to 90% confluence were cultured, washed with PBS, and trypsinized , after termination of digestion, cells were collected and seeded in 96-well plates, 2x10 4 /well, DMEM medium with 10% fetal bovine serum, 2 mM glutamate, and cultured overnight in a 5% CO 2 incubator. The next day, the medium was removed and replaced with fresh medium containing different concentrations of anti-CLD18A2 single-domain antibody-drug complexes and their corresponding single-domain antibody fusion proteins, 50 μl per well, three replicate wells per concentration; 37 Incubate for 72 hours at ℃, 5% CO 2 , remove the supernatant and use LDH kit (purchased from Dongren Chemical Technology (Shanghai) Co., Ltd., product number CK12) to detect the release of LDH. The detection method is carried out according to the instruction manual. Cell killing percentage (%)=100*(OD490 test sample-OD490 control well)/(OD490 cells were completely lysed-OD490 control well). The experimental results are shown in Figure 5 and Table 5.
表5table 5
上述结果表明,本发明中所述的抗体-药物复合物能特异靶向人CLD18A2,阳性细胞毒性强,相比阳性对照抗体药物-复合物ch175D10-MC-VC-PAB-MMAE更能显示对肿瘤细胞的杀伤作用。未偶联药物的单域抗体融合蛋白Ab3的并无明显细胞毒性作用。The above results show that the antibody-drug complex described in the present invention can specifically target human CLD18A2, and the positive cytotoxicity is strong. cell killing. The unconjugated single domain antibody fusion protein Ab3 had no obvious cytotoxic effect.
实施例9体内毒性测试Example 9 In vivo toxicity test
本实验采用病人来源的胃癌组织建立的异种移植瘤模型(patientl derived xenograft,PDX)的荷瘤小鼠,测定抗体-药物化合物的抗肿瘤作用。如实施例7.4所描述的方法类似,将100mm 3左右大小的荷瘤小鼠随机分组,每组实验组4-6只小鼠。肿瘤移植15d后,给予6mg/kg剂量不同抗体-药物复合物的静脉注射治疗,监测给药期间各组小鼠瘤体积,给药频率为单次给药,监测频率均为3天1次,连续监测6周。肿瘤体积测定:采用游标卡尺测定肿瘤的最大长轴(L)和最大宽轴(W),肿瘤体积按如下公式计算:V=L×W 2/2。 In this experiment, the tumor-bearing mice of the xenograft model (patientl derived xenograft, PDX) established by patient-derived gastric cancer tissue were used to determine the anti-tumor effects of antibody-drug compounds. Similar to the method described in Example 7.4, tumor-bearing mice with a size of about 100 mm 3 were randomly divided into groups, with 4-6 mice in each experimental group. 15 days after tumor transplantation, intravenous injection of different antibody-drug complexes at a dose of 6 mg/kg was given. During the administration period, the tumor volume of mice in each group was monitored. The administration frequency was a single administration, and the monitoring frequency was once every 3 days. Continuous monitoring for 6 weeks. Determination of tumor volume: The maximum long axis (L) and the maximum width axis (W) of the tumor were measured with vernier calipers, and the tumor volume was calculated according to the following formula: V=L×W 2 /2.
实验结果如图6所示,随着时间的推移,接种了抗体-药物复合物的小鼠,其肿瘤体积相对于PBS对照组得到了很好的控制,相比阳性对照组ch175D10-MC-VC-PAB-MMAE也具有更好的肿瘤抑制作用。Ab1-MC-VC-PAB-MMAE和Ab2-MC-VC-PAB-MMAE尽管在后期有一定的反弹,但最终也维持在较低的肿瘤体积,并没有出现进一步显著增加的情况,说明有明显的肿瘤抑制作用。The experimental results are shown in Figure 6. Over time, the tumor volume of mice inoculated with the antibody-drug complex was well controlled relative to the PBS control group, compared with the positive control group ch175D10-MC-VC. -PAB-MMAE also has better tumor suppressive effect. Although Ab1-MC-VC-PAB-MMAE and Ab2-MC-VC-PAB-MMAE rebounded to a certain extent in the later stage, they eventually maintained a lower tumor volume without further significant increase, indicating that there was a significant increase in tumor volume. tumor suppressor effect.
实施例10抗CLD18A2单域抗体用于嵌合抗原受体Example 10 Anti-CLD18A2 Single Domain Antibody for Chimeric Antigen Receptor
将本发明中的抗CLD18A2单域抗体用于嵌合抗原受体的构建,表6列举了构建的包含信号肽的嵌合抗原受体及其结构(信号肽-抗原识别域-铰链区-跨膜域-胞内信号域)。The anti-CLD18A2 single-domain antibody of the present invention is used for the construction of chimeric antigen receptors. Table 6 lists the constructed chimeric antigen receptors comprising signal peptides and their structures (signal peptide-antigen recognition domain-hinge region-trans membrane domain - intracellular signaling domain).
表6Table 6
1.用于表达特异性抗CLD18A2单域抗体慢病毒质粒载体的构建1. Construction of lentiviral plasmid vector for expressing specific anti-CLD18A2 single-domain antibody
作为示例的构建,本发明使用第三代自灭活慢病毒载体系统,该系统共有四个质粒即编码VSV-G蛋白的包膜质粒pLP/VSVG(购自Addgene);编码蛋白Gag/Pol的包装质粒pLP1;编码Rev蛋白的包装质粒pLP2(购自Addgene)及基于空载体pLVX-IRES-ZsGreen1(购自Addgene)构建的编码目的基因CAR的重组表达载体。As an example construction, the present invention uses the third-generation self-inactivating lentiviral vector system, which has four plasmids, namely, the enveloped plasmid pLP/VSVG (purchased from Addgene) encoding the VSV-G protein; Packaging plasmid pLP1; packaging plasmid pLP2 (purchased from Addgene) encoding Rev protein and recombinant expression vector encoding the target gene CAR constructed based on the empty vector pLVX-IRES-ZsGreen1 (purchased from Addgene).
合成结构序列(SEQ ID NO:137、SEQ ID NO:138、SEQ ID NO:139、SEQ ID NO:140),合成的序列通过两端的EcoRI和BamHI酶切位点,与同样酶切的载体pLVX-IRES-ZsGreen1(Addgene)通过T4连接酶(Takara)连接。将连接产物转化Top10F’,涂布氨苄青霉素抗性平板,挑克隆培养测序确认,获得表达上述CAR的表达载体:pLVX-aC18.2-hu6V3-28Z、pLVX-aC18.2-hu6V3-28-137Z、pLVX-aC18.2-hu19V3-28Z和pLVX-aC18.2-hu19V3-28-137Z,Synthetic structural sequences (SEQ ID NO: 137, SEQ ID NO: 138, SEQ ID NO: 139, SEQ ID NO: 140), the synthesized sequences pass through the EcoRI and BamHI restriction sites at both ends, and the same restriction vector pLVX -IRES-ZsGreen1 (Addgene) was ligated by T4 ligase (Takara). The ligation product was transformed into Top10F', coated with ampicillin-resistant plate, cloned, cultured and sequenced to confirm, and the expression vectors expressing the above CAR were obtained: pLVX-aC18.2-hu6V3-28Z, pLVX-aC18.2-hu6V3-28-137Z , pLVX-aC18.2-hu19V3-28Z and pLVX-aC18.2-hu19V3-28-137Z,
2.质粒转染293T包装慢病毒2. Plasmid transfection of 293T packaging lentivirus
慢病毒包装遵循常规方法,大致如下:5×10
6细胞密度种植HEK-293T细胞(ATCC)细胞于10cm培养皿,37℃,5%CO
2培养箱培养过夜,培养基为含10%胎牛血清(Gibco)的DMEM(Gibco)。转染前约2小时更换培养液为无血清DMEM,细胞转染时,除了使用表达CAR的慢病毒质粒外,还需要质粒(提供病毒膜蛋白和结构蛋白)pLP/VSVG、pLP1、pLP2。其中表达目的序列CAR或空载体的慢病毒质粒使用5μg,pLP1使用2.5μg,pLP2使用2.5μg,pLP/VSVG使用1.25μg。转染时,将以上四个种质粒的混合物加入500μl MEM培养基内,在另一个微型离心管内将25μL Lipofectamine 2000转染试剂(thermo fisher)加入500μL MEM培养基内,然后,将稀释后的转染试剂加入稀释后的质粒上方,混匀,室温静置20分钟后,将质粒和转染试剂的混合物加入10cm培养皿内,摇晃、混匀,放入37℃培养箱,6小时后,更换成10%胎牛血清的DMEM培养基。细胞转染3天后,可以收获病毒,将含病毒的培养上清转入离心管内,4℃,1500rpm,离心5分钟,去除细胞,然后,将含病毒培养基过滤分装,-80℃冻存。在10%胎牛血清的DMEM以1×10
5/mL细胞密度100μL/孔接种HEK-293T细胞于96孔培养板,37℃,5%CO
2培养过夜。第二天,弃50μL/孔培养上清,补加50μL/孔新鲜上述培养液,并含终浓度为6μg/mL的polybrene,37℃,5%CO
2孵育30min。加10μL/孔的病毒原液,37℃,5%CO
2培养。感染48h后,流式细胞仪检测GFP,以阳性率为5~20%的细胞数为宜,计算滴度约为2×10
6U/mL。
Lentiviral packaging follows the conventional methods, as follows: 5 × 10 6 cell density cultivation of HEK-293T cells (ATCC) cells in 10cm dishes, 37 ℃, 5% CO 2 incubator overnight, the medium containing 10% fetal bovine Serum (Gibco) in DMEM (Gibco). Change the culture medium to serum-free DMEM about 2 hours before transfection. When transfecting cells, in addition to using CAR-expressing lentiviral plasmids, plasmids (providing viral membrane proteins and structural proteins) pLP/VSVG, pLP1, and pLP2 are also required. The lentiviral plasmid expressing the target sequence CAR or empty vector used 5 μg, pLP1 used 2.5 μg, pLP2 used 2.5 μg, and pLP/VSVG used 1.25 μg. During transfection, the mixture of the above four plasmids was added to 500 μl MEM medium, and 25
实施例11:特异性靶向Claudin18.2的CAR-T细胞Example 11: CAR-T cells specifically targeting Claudin18.2
1.aC18.2-CAR-T制备1. Preparation of aC18.2-CAR-T
由健康人外周血通过密度梯度离心法获得人外周血单个核细胞(上海妙通),并通过CD3MicroBeads(美天旎,Miltenyi Biotec GmbH)根据说明书进行分选。以约1×10 6/mL密度加入Quantum007淋巴细胞培养基液(购自PAA Laboratories GmbH公司)培养并以细胞:磁珠比例为1:1加入DynabeadsTM Human T-Activator CD3/CD28(thermofisher)和终浓度100U/mL的重组人IL-2(上海近岸)刺激培养24h。然后以MOI≈5用上述重组慢病毒(实施例10.3)感染T细胞。感染后的细胞每隔一天采用5×10 5/mL的密度进行传代,同时在淋巴细胞培养液中补加终浓度100U/mL的重组人IL-2。在培养第8天时通过流式细胞检测,由于GFP与CAR共表达,检测GFP的阳性细胞认为表达嵌合抗原受体的阳性细胞。未感染的T细胞作为阴性对照,表达不同嵌合抗原受体的病毒感染T细胞其阳性率约为64.2%。 Human peripheral blood mononuclear cells (Shanghai Miaotong) were obtained from healthy human peripheral blood by density gradient centrifugation, and sorted by CD3 MicroBeads (Miltenyi Biotec GmbH) according to the instructions. Quantum007 lymphocyte culture medium (purchased from PAA Laboratories GmbH) was added at a density of about 1 × 10 6 /mL, and DynabeadsTM Human T-Activator CD3/CD28 (thermofisher) was added at a cell:magnetic bead ratio of 1:1 and the final cells were added. Recombinant human IL-2 (Shanghai nearshore) at a concentration of 100 U/mL was stimulated and cultured for 24 h. T cells were then infected with the recombinant lentivirus described above (Example 10.3) at MOI ≈ 5. The infected cells were passaged at a density of 5×10 5 /mL every other day, and the lymphocyte culture medium was supplemented with recombinant human IL-2 at a final concentration of 100 U/mL. On the 8th day of culture, by flow cytometry, due to the co-expression of GFP and CAR, the positive cells detected by GFP were regarded as positive cells expressing the chimeric antigen receptor. Uninfected T cells were used as negative control, and the positive rate of virus-infected T cells expressing different chimeric antigen receptors was about 64.2%.
2.aC18.2-CAR-T的杀伤实验2. Killing experiments of aC18.2-CAR-T
我们观察了不同aC18.2-CAR-T细胞在体外对NUGC-4-Claudin18.2细胞和CLD18A2阴性细胞系NUGC-4-Claudin18.1的杀伤作用。效靶比分别设置为3:1、1:1和1:3,靶细胞数量为10000/孔。各组均设5个复孔,取5个复孔的平均值,共培养16h后,用LDH检测试剂盒(上海东仁)检测上清的LDH含量评价杀伤。结果表7显示在效靶比为3:1时,特异性aC18.2-CAR-T细胞能够有效杀伤Claudin18.2表达阳性的细胞,而对Claudin18.2阴性的细胞则几乎没有杀伤。以上结果显示,aC18.2-CAR-T能够特异性的杀伤Claudin18.2阳性的细胞,且杀伤作用与效靶比呈正相关。We observed the killing effect of different aC18.2-CAR-T cells on NUGC-4-Claudin18.2 cells and CLD18A2 negative cell line NUGC-4-Claudin18.1 in vitro. The effector-target ratios were set as 3:1, 1:1 and 1:3, respectively, and the number of target cells was 10,000/well. Five duplicate wells were set in each group, and the average value of the five duplicate wells was taken. After co-cultivation for 16 hours, the LDH content of the supernatant was detected by LDH detection kit (Shanghai Dongren) to evaluate killing. Results Table 7 shows that when the effector-target ratio is 3:1, specific aC18.2-CAR-T cells can effectively kill Claudin18.2-positive cells, but almost no Claudin18.2-negative cells. The above results show that aC18.2-CAR-T can specifically kill Claudin18.2 positive cells, and the killing effect is positively correlated with the effector-target ratio.
表7Table 7
3.体外细胞因子释放3. In vitro cytokine release
将Claudin18.2表达阳性的细胞NUGC-4-Claudin18.2与aC18.2-CAR-T细胞以1:1的比例共同培养,孵育24h后收集培养上清,分别用IL-2(R&D Systems,Inc.)、TNF-α(R&D Systems,Inc.)和IFN-γ(R&D Systems,Inc.)按试剂盒说明书检测细胞因子。图7结果显示 NUGC-4-Claudin18.2中,aC18.2-CAR-T细胞aC18.2-hu19V3-28-137Z共孵育时IL-2、TNF-α和IFN-γ等细胞因子的分泌显著高于阴性细胞NUGC-4-Claudin18.1。Claudin18.2-positive cells NUGC-4-Claudin18.2 and aC18.2-CAR-T cells were co-cultured at a ratio of 1:1, and the culture supernatant was collected after 24 hours of incubation, and IL-2 (R&D Systems, Inc.), TNF-α (R&D Systems, Inc.) and IFN-γ (R&D Systems, Inc.) were detected according to the kit instructions. Figure 7 shows that in NUGC-4-Claudin18.2, aC18.2-CAR-T cells aC18.2-hu19V3-28-137Z co-incubated the secretion of cytokines such as IL-2, TNF-α and IFN-γ significantly higher than negative cells NUGC-4-Claudin18.1.
4.aC18.2-CAR-T的体内药效学研究4. In vivo pharmacodynamic study of aC18.2-CAR-T
以NUGC-4-Claudin18.2,建立了皮下移植瘤模型。将3×10 6个NUGC-4-Claudin18.2皮下接种NOD/SCID小鼠。至小鼠肿瘤平均体积达100-150mm 3时,腹腔注射100mg/kg的环磷酰胺以清除NOD/SCID小鼠的免疫细胞,使得过继转移的转基因的T淋巴细胞更好的发挥抗肿瘤功能。第二日,通过尾静脉输注1.0×10 7个aC18.2-CAR-T细胞aC18.2-hu19V3-28-137Z,同时以表达28-137Z的Mock组作为对照,观察测量皮下移植瘤的生长。结果图8显示aC18.2-CAR-T细胞能够显著抑制NUGC-4-Claudin18.2移植瘤的生长。 With NUGC-4-Claudin18.2, a subcutaneous xenograft model was established. To 3 × 10 6 th NUGC-4-Claudin18.2 inoculated subcutaneously NOD / SCID mice. When the average tumor volume in mice reached 100-150 mm 3 , 100 mg/kg of cyclophosphamide was injected intraperitoneally to clear the immune cells of NOD/SCID mice, so that the adoptively transferred transgenic T lymphocytes could play a better anti-tumor function. On the second day, 1.0×10 7 aC18.2-CAR-T cells aC18.2-hu19V3-28-137Z were infused through the tail vein, and the Mock group expressing 28-137Z was used as a control to observe and measure the subcutaneous transplanted tumor. grow. Results Figure 8 shows that aC18.2-CAR-T cells can significantly inhibit the growth of NUGC-4-Claudin18.2 xenografts.
综上所述,本发明有效克服了现有技术中的种种缺点而具高度产业利用价值。To sum up, the present invention effectively overcomes various shortcomings in the prior art and has high industrial utilization value.
上述实施例仅例示性说明本发明的原理及其功效,而非用于限制本发明。任何熟悉此技术的人士皆可在不违背本发明的精神及范畴下,对上述实施例进行修饰或改变。因此,举凡所属技术领域中具有通常知识者在未脱离本发明所揭示的精神与技术思想下所完成的一切等效修饰或改变,仍应由本发明的权利要求所涵盖。The above-mentioned embodiments merely illustrate the principles and effects of the present invention, but are not intended to limit the present invention. Anyone skilled in the art can modify or change the above embodiments without departing from the spirit and scope of the present invention. Therefore, all equivalent modifications or changes made by those with ordinary knowledge in the technical field without departing from the spirit and technical idea disclosed in the present invention should still be covered by the claims of the present invention.
Claims (20)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202080041701.3A CN114222761B (en) | 2020-07-14 | 2020-07-14 | A single domain antibody against CLD18A2 |
| PCT/CN2020/101789 WO2022011531A1 (en) | 2020-07-14 | 2020-07-14 | Anti-cld18a2 single-domain antibody |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/CN2020/101789 WO2022011531A1 (en) | 2020-07-14 | 2020-07-14 | Anti-cld18a2 single-domain antibody |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022011531A1 true WO2022011531A1 (en) | 2022-01-20 |
Family
ID=79554401
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2020/101789 Ceased WO2022011531A1 (en) | 2020-07-14 | 2020-07-14 | Anti-cld18a2 single-domain antibody |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN114222761B (en) |
| WO (1) | WO2022011531A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023134742A1 (en) * | 2022-01-14 | 2023-07-20 | 浙江道尔生物科技有限公司 | Three-target anti-tumor drug, and preparation method therefor and use thereof |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN119591708A (en) * | 2023-09-08 | 2025-03-11 | 成都临界点生物科技有限公司 | Humanized single domain antibodies against Claudin18.2 |
| CN117551199B (en) * | 2023-11-16 | 2024-04-19 | 杭州荣谷生物科技有限公司 | Preparation method and application of Claudin18.2 nano antibody |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107667118A (en) * | 2015-04-15 | 2018-02-06 | 加尼梅德药物有限公司 | Include the drug conjugate of the antibody for sealing protein 18 .2 |
| CN107960056A (en) * | 2015-05-11 | 2018-04-24 | 拜恩科技细胞&基因治疗有限公司 | Ocludin-18.2 specific immune receptor and T cell epitope |
| CN108047331A (en) * | 2012-05-09 | 2018-05-18 | 加尼梅德药物公司 | For the antibody for claudin 18.2 of cancer diagnosis |
| CN110862454A (en) * | 2018-08-27 | 2020-03-06 | 南京圣和药业股份有限公司 | anti-Claudin 18_2 antibody and application thereof |
| EP3656793A1 (en) * | 2005-11-24 | 2020-05-27 | Astellas Pharma Inc. | Monoclonal antibodies against claudin-18 for treatment of cancer |
| CN111434692A (en) * | 2019-01-15 | 2020-07-21 | 浙江道尔生物科技有限公司 | Anti-CLD18A2 Nanobody and Its Application |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1997832A1 (en) * | 2007-05-29 | 2008-12-03 | Ganymed Pharmaceuticals AG | Monoclonal antibodies against Claudin-18 for treatment of cancer |
| EA035033B1 (en) * | 2010-11-22 | 2020-04-20 | Иннейт Фарма Са | Method for treatment of hematological pre-malignancy or hematological malignancy |
| WO2015113576A1 (en) * | 2014-01-29 | 2015-08-06 | Biontech Ag | Peptide mimotopes of claudin 18.2 and uses thereof |
| CN120329449A (en) * | 2014-07-17 | 2025-07-18 | 恺兴生命科技(上海)有限公司 | T lymphocytes targeting CLD18A2 and preparation method and application thereof |
| CN105384825B (en) * | 2015-08-11 | 2018-06-01 | 南京传奇生物科技有限公司 | A kind of bispecific chimeric antigen receptor and its application based on single domain antibody |
| JP7587921B2 (en) * | 2016-07-08 | 2024-11-21 | クレージュ メディカル カンパニー,リミテッド | Anti-claudin 18A2 antibody and its applications |
| US11912763B2 (en) * | 2018-06-17 | 2024-02-27 | L & L Biopharma Co., Ltd. | Antibody targeting CLDN18.2, bispecific antibody, ADC, and CAR, and applications thereof |
| CN110606891B (en) * | 2018-06-17 | 2022-12-06 | 上海健信生物医药科技有限公司 | Antibody molecule aiming at human CLDN18.2, antigen binding fragment and medical application thereof |
| CN118955712A (en) * | 2018-07-25 | 2024-11-15 | 阿克罗斯生物科学公司 | CLDN 18.2-specific monoclonal antibodies and methods of use thereof |
| CN110857322A (en) * | 2018-08-22 | 2020-03-03 | 瑞阳(苏州)生物科技有限公司 | Anti-human claudin18.2 monoclonal antibody and application thereof |
| CN109762067B (en) * | 2019-01-17 | 2020-02-28 | 北京天广实生物技术股份有限公司 | Antibodies that bind human Claudin18.2 and uses thereof |
| CN111235113A (en) * | 2020-01-21 | 2020-06-05 | 南京北恒生物科技有限公司 | Immune cells comprising chimeric antigen receptors and uses thereof |
-
2020
- 2020-07-14 WO PCT/CN2020/101789 patent/WO2022011531A1/en not_active Ceased
- 2020-07-14 CN CN202080041701.3A patent/CN114222761B/en active Active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3656793A1 (en) * | 2005-11-24 | 2020-05-27 | Astellas Pharma Inc. | Monoclonal antibodies against claudin-18 for treatment of cancer |
| CN108047331A (en) * | 2012-05-09 | 2018-05-18 | 加尼梅德药物公司 | For the antibody for claudin 18.2 of cancer diagnosis |
| CN107667118A (en) * | 2015-04-15 | 2018-02-06 | 加尼梅德药物有限公司 | Include the drug conjugate of the antibody for sealing protein 18 .2 |
| CN107960056A (en) * | 2015-05-11 | 2018-04-24 | 拜恩科技细胞&基因治疗有限公司 | Ocludin-18.2 specific immune receptor and T cell epitope |
| CN110862454A (en) * | 2018-08-27 | 2020-03-06 | 南京圣和药业股份有限公司 | anti-Claudin 18_2 antibody and application thereof |
| CN111434692A (en) * | 2019-01-15 | 2020-07-21 | 浙江道尔生物科技有限公司 | Anti-CLD18A2 Nanobody and Its Application |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023134742A1 (en) * | 2022-01-14 | 2023-07-20 | 浙江道尔生物科技有限公司 | Three-target anti-tumor drug, and preparation method therefor and use thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| CN114222761B (en) | 2024-02-20 |
| CN114222761A (en) | 2022-03-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20240100180A1 (en) | Tumor-specific claudin 18.2 antibody-drug conjugates | |
| US11845793B2 (en) | Anti-ROR1 antibodies | |
| KR102629905B1 (en) | Anti-PD-L1/anti-PD-1 natural antibody structure-like heterodimeric bispecific antibody and preparation thereof | |
| WO2020147451A1 (en) | Anti-cld18a2 nanobody and application thereof | |
| US20230348627A1 (en) | Anti-4-1bb-anti-pd-l1 bispecific antibody, and pharmaceutical composition and use thereof | |
| TWI812645B (en) | Novel Anti-CD19 Antibody | |
| CN113527487A (en) | Monoclonal antibody of anti-human B7-H3 and application thereof | |
| KR20180129684A (en) | Anti-Human Interleukin-2 Antibodies and Uses thereof | |
| AU2022232674B2 (en) | Anti-her2 antibody-immune agonist conjugate and applications thereof | |
| CN113943370A (en) | Heterodimeric bispecific antibody with anti-PD-1/anti-HER 2 natural antibody structure and preparation method thereof | |
| US20220177573A1 (en) | Two chimeric antigen receptors specifically binding cd19 and igkappa | |
| CN114222761B (en) | A single domain antibody against CLD18A2 | |
| CN113754766A (en) | anti-B7-H3 antibody and preparation and application thereof | |
| CN113166251A (en) | Anti-human TIM-3 monoclonal antibody and application thereof | |
| CN112996816B (en) | Anti-glycemic-MUC1 antibodies and their uses | |
| CN110799211B (en) | Drug delivery of anti-RON monoclonal antibodies that specifically recognize the plexin-semaphorin-integrin domain and its application in tumor therapy | |
| KR20230171465A (en) | Anti-CLDN4-anti-CD137 bispecific antibody | |
| WO2022122709A1 (en) | Antibody-drug conjugates based on humanized cldn18.2 antibodies | |
| TW202428603A (en) | Novel fusion protein specific for cd137 and cd228 | |
| EP4613774A1 (en) | Single domain antibody targeting human ror1 | |
| WO2024199294A1 (en) | Antibody or antigen-binding fragment thereof targeting cd3 and use thereof | |
| HK40057929A (en) | Anti-pd-1/anti-her2 natural antibody structural heterodimeric bispecific antibody and method of preparing same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 20944965 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 20944965 Country of ref document: EP Kind code of ref document: A1 |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 20944965 Country of ref document: EP Kind code of ref document: A1 |