WO2021136462A1 - Furan derivatives and application thereof in medicine - Google Patents
Furan derivatives and application thereof in medicine Download PDFInfo
- Publication number
- WO2021136462A1 WO2021136462A1 PCT/CN2020/141860 CN2020141860W WO2021136462A1 WO 2021136462 A1 WO2021136462 A1 WO 2021136462A1 CN 2020141860 W CN2020141860 W CN 2020141860W WO 2021136462 A1 WO2021136462 A1 WO 2021136462A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- methyl
- alkyl
- chloro
- amino
- dihydro
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 *C1C(CCCC2)C2CCC1 Chemical compound *C1C(CCCC2)C2CCC1 0.000 description 2
- RMWQCJMQJIRCRO-UHFFFAOYSA-N CN(c1cnc(Nc(cc(CCO2)c2c2)c2Cl)nc1N1C2=CC(O3)=CCC3=C2)C1=O Chemical compound CN(c1cnc(Nc(cc(CCO2)c2c2)c2Cl)nc1N1C2=CC(O3)=CCC3=C2)C1=O RMWQCJMQJIRCRO-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D473/00—Heterocyclic compounds containing purine ring systems
- C07D473/26—Heterocyclic compounds containing purine ring systems with an oxygen, sulphur, or nitrogen atom directly attached in position 2 or 6, but not in both
- C07D473/32—Nitrogen atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/519—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/519—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with heterocyclic rings
- A61K31/52—Purines, e.g. adenine
- A61K31/522—Purines, e.g. adenine having oxo groups directly attached to the heterocyclic ring, e.g. hypoxanthine, guanine, acyclovir
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/28—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to ring carbon atoms
- C07D239/30—Halogen atoms or nitro radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/28—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to ring carbon atoms
- C07D239/32—One oxygen, sulfur or nitrogen atom
- C07D239/42—One nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/12—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D473/00—Heterocyclic compounds containing purine ring systems
- C07D473/40—Heterocyclic compounds containing purine ring systems with halogen atoms or perhalogeno-alkyl radicals directly attached in position 2 or 6
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D519/00—Heterocyclic compounds containing more than one system of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring system not provided for in groups C07D453/00 or C07D455/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/18—Compounds having one or more C—Si linkages as well as one or more C—O—Si linkages
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/18—Compounds having one or more C—Si linkages as well as one or more C—O—Si linkages
- C07F7/1804—Compounds having Si-O-C linkages
Definitions
- the present invention relates to furan derivatives, or their stereoisomers, solvates, prodrugs, metabolites, deuterated substances, pharmaceutically acceptable salts or co-crystals, their pharmaceutical compositions and their use in the preparation of DNA-PK inhibitors .
- DNA-dependent protein kinase is a DNA-PK enzyme complex composed of Ku70/Ku80 heterodimer and DNA-dependent protein kinase catalytic subunit (DNA-PKcs). The enzyme complex needs to be activated with the participation of DNA to perform its corresponding functions (George et al., 2019). As a serine/threonine protein kinase, DNA-PK belongs to the PIKK (phosphatidylinositol 3-kinase-related kinase) family.
- PIKK phosphatidylinositol 3-kinase-related kinase
- DSBs In the normal physiological process, a variety of factors may lead to the occurrence of DSBs in DNA: For example, DSBs often appear as intermediate products in the process of somatic DNA recombination. This physiological process is very important for the formation of the functional immune system of all vertebrates; DNA replication is in progress. When the replication fork encounters damaged bases, it may also cause single-strand or double-strand breaks; DNA may also generate DSBs due to the attack of reactive oxygen species (ROS) during normal metabolism (Cannan & Pederson, 2016).
- ROS reactive oxygen species
- DSBs ionizing radiation (IR) and chemotherapeutic agents (such as topoisomerase II inhibitors)
- IR ionizing radiation
- chemotherapeutic agents such as topoisomerase II inhibitors
- NHEJ non-homologous end-joining
- NHEJ is a dynamic process mediated by DNA-PK that requires the participation of multiple proteins and signaling pathways.
- the basic process is as follows: (1) Ku70/Ku80 heterodimer recognizes and binds to the ends of double-stranded DNA breaks; (2) Recruitment DNA-PKcs, XRCC4-DNA ligase IV complex and other proteins to both sides of the DNA break double-strand; (3) DNA-PKcs autophosphorylate and activate its own kinase activity; (4) DNA-PKcs as an adhesive to connect Break both ends of the DNA to prevent exonuclease from degrading DN A; (5) Process the DNA to remove the unlinkable ends or other forms of damage at the break; (6) XRCC4-DNA ligase IV complex Repair DNA ends (in some cases, DNA polymerase may be required to synthesize new ends before ligation).
- DNA-PKcs When DNA-PKcs is phosphorylated, it can induce protein conformation to change and regulate the activity of various proteins in the NHEJ process (such as Artemis, Ku70, Ku80, DNA ligase), which is essential for the repair process of DN A. Therefore, phosphorylated DNA-PKcs (pDNA-PKcs) is often used as a marker of cellular DSBs.
- DNA-PK activity is related to the occurrence and development of a variety of tumors: for example, DNA-PKcs in melanoma can promote angiogenesis and tumor metastasis; DNA-PKcs expression in multiple myeloma is significantly up-regulated; radiotherapy The content of Ku protein in tolerant thyroid tumors is significantly increased (Ihara, Ashizawa, Shichijo, & Kudo, 2019). Therefore, it can be considered to combine DNA-PK inhibitors with anti-tumor therapies that cause DNA damage (such as IR, chemotherapeutic agents, etc.) to improve the effect.
- the use of DNA-PK inhibitors can interfere with the DNA repair function of normal cells to a certain extent. However, there are many DNA repair pathways in normal cells as a supplement, and tumor cells face strong DNA replication pressure and lack effective DNA repair methods. . By inhibiting the activity of tumor cell DNA-PK, the killing effect of other anti-tumor drugs on tumor cells can be improved.
- DNA-PK inhibitors After years of research, several DNA-PK inhibitors have been discovered.
- the first compound found to have DNA-PK kinase inhibitory activity is a fungal metabolite-Wortmannin, with an IC50 (DNA-PK) of about 15nM.
- DNA-PK IC50
- This compound also plays an important role in the acetylation and phosphorylation of p53 protein ( Sarkaria et al., 1998);
- the quercetin derivative LY294002 reported later also has DNA-PK inhibitory activity (Maira, Stauffer, Schnell, & Garcia-Echeverria, 2009); later based on the structure of LY294002, NU7026 and NU7441 were developed A generation of DNA-PK inhibitors.
- DNA-PK inhibitors have also been reported, such as OK1035, SU11752, PP121, KU-0060648 and other small molecule compounds, but these compounds also have defects such as low specificity for DNA-PK (George et al., 2019). Therefore, there is still a need to develop DNA-PK inhibitors with high activity, high specificity, and low toxicity to better meet clinical needs.
- One or more embodiments of the present application provide furan derivatives, or their stereoisomers, solvates, prodrugs, metabolites, deuterated products, pharmaceutically acceptable salts or co-crystals, and their pharmaceutical compositions and Preparation of DNA-PK inhibitors; these compounds have high inhibitory activity and high selectivity to DNA-PK, and can be used as chemotherapy and radiotherapy sensitizers to effectively treat cancer, improve the curative effect of the prior art, and reduce toxic and side effects.
- One or more embodiments of the present application provide a compound of general formula (I'), or its stereoisomers, solvates, prodrugs, metabolites, deuterated products, pharmaceutically acceptable salts or co-crystals:
- R 0 and R 1 are each independently selected from H, -OH, cyano, halogen, -NH 2 , C 1-6 alkyl or C 1-6 alkoxy, the C 1-6 alkyl optionally Further substituted by 1-3 substituents selected from D or halogen;
- R 0 and the atoms to which they are connected form a 3- to 8-membered ring, and the 3- to 8-membered ring optionally contains 1 to 3 heteroatoms selected from N, O or S, and the 3- to 8-membered ring
- R 2 is selected from H or C 1-6 alkyl
- R 3 is selected from C 1-6 alkyl, C 3-12 carbocyclic group, C 3 heterocyclic group, C 4-12 heterocyclic group, -C 1-6 alkylene-C 3-12 carbocyclic group, -C 1-6 alkylene-C 3 heterocyclic group, -C 1-6 alkylene-C 4-12 heterocyclic group, C 6-12 spiro compound or C 6-12 heterospiro compound, so
- the C 3 heterocyclic group and C 4-12 heterocyclic group contain 1 to 3 heteroatoms selected from N, O or S
- R 4 is selected from H, C 1-6 alkyl, C 3-12 cycloalkyl, said C 1-6 alkyl and C 3-8 cycloalkyl are optionally further selected from 1-3 from D or Substituted by halogen substituents;
- R a3 is selected from C 1-6 alkyl, C 1-6 alkoxy or C 6-12 aryl;
- n 0, 1, 2 or 3;
- p 0, 1, 2, or 3.
- One or more embodiments of the application provide a compound, or its stereoisomer, solvate, prodrug, metabolite, deuterated product, pharmaceutically acceptable salt or co-crystal, wherein the compound is selected from the general formula ( I) The compound shown:
- R 0 and R 1 are selected from H, -OH, cyano, halogen, -NH 2 , C 1-6 alkyl or C 1-6 alkoxy, and the alkyl group is optionally further selected from 1-3 Substituted by D or halogen substituent;
- R 2 is selected from H or C 1-6 alkyl
- R a3 is selected from C 1-6 alkyl, C 1-6 alkoxy or C 6-12 aryl;
- R a4 and R a5 are selected from H or C 1-6 alkyl
- n and p are each independently selected from 0, 1, 2 or 3.
- One or more embodiments of the application provide a compound, or its stereoisomer, solvate, prodrug, metabolite, deuterated product, pharmaceutically acceptable salt or co-crystal, wherein the compound is selected from the general formula ( IA) The compound shown:
- R 0 and R 1 are each independently selected from halogen or C 1-6 alkyl, and said C 1-6 alkyl is optionally further substituted with 1-3 substituents selected from D or halogen;
- R 0 and the atoms to which they are connected form a 3- to 8-membered ring, and the 3- to 8-membered ring optionally contains 1 to 3 heteroatoms selected from N, O or S, and the 3- to 8-membered ring
- R 3 is selected from C 1-6 alkyl, C 3-12 carbocyclic group, C 3 heterocyclic group, C 4-12 heterocyclic group, -C 1-6 alkylene-C 3-12 carbocyclic group, -C 1-6 alkylene-C 3 heterocyclic group, -C 1-6 alkylene-C 4-12 heterocyclic group, C 6-12 spiro compound or C 6-12 heterospiro compound, so
- the C 3 heterocyclic group and C 4-12 heterocyclic group contain 1 to 3 heteroatoms selected from N, O or S
- R 4 is selected from H, C1-6 alkyl, C3-12 cycloalkyl, said C 1-6 alkyl and C 3-8 cycloalkyl are optionally further selected from 1-3 selected from D or halogen Substituent substituted;
- R a3 is selected from C 1-6 alkyl, C 1-6 alkoxy or C 6-12 aryl;
- R a4 and R a5 are each independently selected from H or C 1-6 alkyl
- n 0, 1, 2 or 3;
- p 0, 1, 2, or 3.
- One or more embodiments of the application provide a compound, or its stereoisomer, solvate, prodrug, metabolite, deuterated product, pharmaceutically acceptable salt or co-crystal, wherein the compound is selected from the general formula ( II) The compound shown:
- R 0 and R 1 are selected from halogen or C 1-6 alkyl, and the alkyl is optionally further substituted with 1-3 substituents selected from D or halogen;
- R a3 is selected from C 1-6 alkyl, C 1-6 alkoxy or C 6-12 aryl;
- R a4 and R a5 are selected from H or C 1-6 alkyl
- n and p are each independently selected from 0, 1, 2 or 3.
- One or more embodiments of the application provide a compound, or its stereoisomer, solvate, prodrug, metabolite, deuterated product, pharmaceutically acceptable salt or co-crystal, wherein the compound is selected from the general formula ( I'), (IA), (I) or (II), wherein:
- R 0 is selected from methyl
- R 1 is selected from halogen or C 1-4 alkyl, and the alkyl is optionally further substituted with 1-3 substituents selected from D or halogen;
- n is selected from 0, 1 or 2;
- p is selected from 1.
- One or more embodiments of the present application provide a compound, or its stereoisomer, solvate, metabolite, prodrug, deuterium, pharmaceutically acceptable salt or co-crystal, wherein the compound is selected from but not limited to :
- One or more embodiments of the present application provide an intermediate for preparing the compound of the present invention, and the intermediate is selected from but not limited to:
- composition comprising:
- compositions of the present application or the compound or its stereoisomers, solvates, prodrugs, metabolites, deuterated substances, pharmaceutically acceptable salts or co-crystals are prepared in Use in medicines for the treatment of cancer.
- compositions of the present application or the compound or its stereoisomers, solvates, prodrugs, metabolites, deuterated substances, pharmaceutically acceptable salts or co-crystals are prepared in Use in DNA-PK inhibitors.
- One or more embodiments of the present application provide the compound of the present application for use as a medicine.
- One or more embodiments of the present application provide the compound of the present application for use as a DNA-PK inhibitor.
- One or more embodiments of the present application provide a compound of the present application for use in a method of treating, preventing, or inhibiting cancer.
- One or more embodiments of the present application provide a compound of the present application for use in a method of inhibiting DNA-PK.
- One or more embodiments of the present application provide a method of treating, preventing or inhibiting cancer, which comprises administering the compound of the present application to a subject in need.
- One or more embodiments of the present application provide a method for inhibiting DNA-PK, which includes administering the compound of the present application to a subject in need.
- the carbon, hydrogen, oxygen, sulfur, nitrogen or F, Cl, Br, and I involved in the groups and compounds of the present invention include their isotopes, and the carbon involved in the groups and compounds of the present invention , Hydrogen, oxygen, sulfur or nitrogen are optionally further replaced by one or more of their corresponding isotopes, wherein carbon isotopes include 12 C, 13 C and 14 C, and hydrogen isotopes include protium (H), deuterium (D, Also called heavy hydrogen), tritium (T, also called super heavy hydrogen), oxygen isotopes include 16 O, 17 O and 18 O, sulfur isotopes include 32 S, 33 S, 34 S and 36 S, and nitrogen isotopes include 14 N and 15 N, fluorine isotopes include 17 F and 19 F, chlorine isotopes include 35 Cl and 37 Cl, and bromine isotopes include 79 Br and 81 Br.
- carbon isotopes include 12 C, 13 C and 14 C
- hydrogen isotopes include
- Alkyl refers to a linear or branched saturated aliphatic hydrocarbon group of 1 to 20 carbon atoms, preferably 1 to 8 (for example, 1, 2, 3, 4, 5, 6, 7, 8) carbon atoms
- the alkyl group of is more preferably an alkyl group of 1 to 6 carbon atoms, and still more preferably an alkyl group of 1 to 4 carbon atoms.
- Non-limiting examples include methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, neobutyl, tert-butyl, n-pentyl, isopentyl, neopentyl, n-hexyl And its various branched isomers; when the alkyl group is substituted, it may be optionally further substituted with one or more substituents.
- Alkoxy refers to a group formed by replacing at least one carbon atom in an alkyl group with an oxygen atom.
- Non-limiting examples include methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, sec-butoxy, tert-butoxy, n-pentoxy, n-hexoxy, cyclopropyl Oxy and cyclobutoxy.
- the definition of the alkyl group is the same as the definition of "alkyl" mentioned above.
- Alkenyl refers to a straight line consisting of 1 to 10 (for example, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10) carbon-carbon double bonds consisting of 2 to 20 carbon atoms. Chain or branched unsaturated aliphatic hydrocarbon group, preferably 2 to 12 (for example, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12) carbon atoms alkenyl group, more preferably 2 to The alkenyl group of 8 carbon atoms is more preferably the alkenyl group of 2 to 6 carbon atoms.
- Non-limiting examples include vinyl, propen-2-yl, buten-2-yl, buten-2-yl, penten-2-yl, penten-4-yl, hexen-2-yl, Hexen-3-yl, hepten-2-yl, hepten-3-yl, hepten-4-yl, octen-3-yl, nonen-3-yl, decen-4-yl and undecenyl En-3-yl.
- the alkenyl group may be further substituted with one or more substituents.
- Alkynyl refers to those containing 1 to 10 (for example, 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10) carbon-carbon triple bonds, consisting of 2 to 20 carbon atoms Straight or branched chain unsaturated aliphatic hydrocarbon group, preferably 2 to 12 (for example, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 or 12) carbon atom alkynyl group, more preferably 2 An alkynyl group having to 8 carbon atoms, and an alkynyl group having 2 to 6 carbon atoms is more preferable.
- Non-limiting examples include ethynyl, propyn-1-yl, propyn-2-yl, butyn-1-yl, butyn-2-yl, butyn-3-yl, 3,3-dimethyl Butyn-2-yl, pentyn-1-yl, pentyn-2-yl, hexyn-1-yl, 1-heptyn-1-yl, heptyn-3-yl, heptyn-4- Base, octyn-3-yl, nonyn-3-yl, decyn-4-yl, undecyn-3-yl, dodecyn-4-yl.
- the alkynyl group may be optionally further substituted with one to more substituents.
- Aryl refers to a substituted or unsubstituted aromatic ring, which can be a 5- to 8-membered (e.g., 5, 6, 7, 8-membered) monocyclic ring, 5 to 12-membered (e.g., 5, 6, 7 , 8, 9, 10, 11, 12 membered) bicyclic or 10 to 15 membered (for example, 10, 11, 12, 13, 14, 15 membered) tricyclic ring system, which can be bridged or spiro ring, non-limiting implementation Examples include phenyl and naphthyl. The aryl group may be further substituted with one or more substituents.
- Heteroaryl refers to a substituted or unsubstituted aromatic ring, which can be 3 to 8 membered (e.g. 3, 4, 5, 6, 7, 8 membered) monocyclic, 5 to 12 membered (e.g. 5, 6, 7, 8, 9, 10, 11, 12 membered) bicyclic or 10 to 15 membered (e.g. 10, 11, 12, 13, 14, 15 membered) tricyclic ring system, and contains 1 to 6 (e.g. 1, 2, 3, 4, 5, 6) heteroatoms selected from N, O or S, preferably 5 to 8 membered heteroaryl groups, and 1 to 4 (e.g. 1, 2 , 3, 4) N and S can be oxidized into various oxidation states.
- 3 to 8 membered e.g. 3, 4, 5, 6, 7, 8 membered
- monocyclic e.g. 5, 6, 7, 8, 9, 10, 11, 12 membered
- 10 to 15 membered e.g. 10, 11, 12, 13, 14, 15 membered
- tricyclic ring system contains 1 to
- the heterocyclic group can be attached to a hetero atom or a carbon atom, and the heteroaryl group can be a bridged ring or a spiro ring.
- Non-limiting examples include cyclopyridyl, furyl, thienyl, pyranyl, pyrrolyl, pyrimidinyl, Pyrazinyl, pyridazinyl, imidazolyl, piperidinyl benzimidazolyl, benzopyridyl, pyrrolopyridyl.
- the heteroaryl group is optionally further substituted with one or more substituents.
- Carbocyclic group or “carbocyclic ring” refers to a saturated or unsaturated aromatic ring or a non-aromatic ring.
- aromatic ring When it is an aromatic ring, its definition is the same as the definition of "aryl”above; when it is a non-aromatic ring, it can be 3 to 10 members (for example, 3, 4, 5, 6, 7, 8, 9, 10 Yuan), 4 to 12 yuan (e.g. 4, 5, 6, 7, 8, 9, 10, 11, 12 yuan) bicyclic ring or 10 to 15 yuan (e.g.
- tricyclic ring system which can be bridged or spiro ring
- non-limiting examples include cyclopropyl, cyclobutyl, cyclopentyl, 1-cyclopentyl-1-enyl, 1-cyclopentyl-2 -Alkenyl, 1-cyclopentyl-3-alkenyl, cyclohexyl, 1-cyclohexyl-2-alkenyl, 1-cyclohexyl-3-alkenyl, cyclohexenyl, cyclohexadienyl, cyclo Heptyl, cyclooctyl, cyclononyl, cyclodecyl, cycloundecyl, cyclododecyl,
- the "carbocyclic group” or "carbocyclic ring” is optionally further substituted with one or more substituents.
- Heterocyclic group or “heterocyclic ring” refers to a saturated or unsaturated aromatic heterocyclic ring or non-aromatic heterocyclic ring. When it is an aromatic heterocyclic ring, its definition is the same as the definition of "heteroaryl” above; when When it is a non-aromatic heterocyclic ring, it can be a 3- to 10-membered (e.g. 3, 4, 5, 6, 7, 8, 9, 10-membered) monocyclic ring, 4 to 12-membered (e.g. 4, 5, 6, 7, 8, 9, 10, 11, 12 membered) bicyclic or 10 to 15 membered (e.g. 10, 11, 12, 13, 14, 15 membered) tricyclic ring system, and contains 1 to 4 (e.g.
- heteroatoms selected from N, O or S preferably 3 to 8 membered heterocyclic groups.
- One to four (for example, 1, 2, 3, 4) N and S optionally substituted in the "heterocyclic group” or “heterocyclic ring” can be oxidized to various oxidation states;
- heterocyclic group” or “Heterocycle” can be attached to a heteroatom or carbon atom;
- heterocyclic group” or “heterocycle” can be a bridged ring or a spiro ring.
- heterocyclic group or “heterocyclic ring” include oxirane, glycidyl, aziridinyl, oxetanyl, azetidinyl, thietanyl , 1,3-dioxolane, 1,4-dioxolane, 1,3-dioxanyl, azepanyl, oxepanyl, thiepanyl, oxygen Azepine, diazepine, thiazepine, pyridinyl, piperidinyl, homopiperidinyl, furyl, thienyl, pyranyl, N-alkylpyrrolyl, pyrimidinyl, pyridine Azinyl, pyridazinyl, piperazinyl, homopiperazinyl, imidazolyl, piperidinyl, morpholinyl, thiomorpholinyl, thiazinyl, 1,3-
- Cycloalkyl refers to a saturated cyclic hydrocarbon group whose ring can be 3 to 10 membered (e.g. 3, 4, 5, 6, 7, 8, 9, 10 membered) monocyclic, 4 to 12 membered (e.g. 4 , 5, 6, 7, 8, 9, 10, 11, 12 yuan) bicyclic or 10 to 20 yuan (e.g. 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20 yuan) more
- the ring carbon atoms preferably have 3 to 10 carbon atoms, more preferably 3 to 8 carbon atoms.
- Non-limiting examples of "cycloalkyl” include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclopropenyl, cyclobutenyl, cyclopentenyl, cyclohexyl Alkenyl, cycloheptenyl, 1,5-cyclooctadienyl, 1,4-cyclohexadienyl and cycloheptatrienyl, etc. When the cycloalkyl group is substituted, it may be further substituted with one or more substituents.
- Heterocycloalkyl refers to a substituted or unsubstituted saturated non-aromatic ring group, which can be 3 to 8 membered (for example, 3, 4, 5, 6, 7, 8 membered) monocyclic, 4 to 12 membered (E.g. 4, 5, 6, 7, 8, 9, 10, 11, 12-membered) bicyclic or 10 to 15-membered (e.g. 10, 11, 12, 13, 14, 15-membered) tricyclic ring system, including 1, 2 or 3 heteroatoms selected from N, O or S, preferably 3 to 8 membered heterocyclic group.
- the selectively substituted N and S in the "heterocycloalkyl" ring can be oxidized to various oxidation states; the "heterocycloalkyl” can be connected to a heteroatom or a carbon atom; the “heterocycloalkyl” can be a bridge Ring or spiro ring.
- heterocycloalkyl include oxirane ethyl, aziridinyl, oxetanyl, azetidinyl, 1,3-dioxolane, 1,4-dioxolane, Oxolane, 1,3-dioxanyl, azepanyl, piperidinyl, piperidinyl, morpholinyl, thiomorpholinyl, 1,3-dithianyl, tetrahydrofuranyl , Tetrahydropyrrolyl, tetrahydroimidazolyl, tetrahydrothiazolyl, tetrahydropyranyl, azabicyclo[3.2.1]octyl, azabicyclo[5.2.0]nonyl, oxa Tricyclic[5.3.1.1]dodecyl, azaadamantyl and oxaspiro[3.3]heptyl.
- “Pharmaceutically acceptable salt” or “pharmaceutically acceptable salt thereof” means that the compound of the present invention maintains the biological effectiveness and characteristics of the free acid or free base, and the free acid is combined with a non-toxic inorganic base or An organic base is a salt obtained by reacting the free base with a non-toxic inorganic acid or organic acid.
- “Pharmaceutical composition” refers to a mixture of one or more compounds of the present invention, their pharmaceutically acceptable salts or prodrugs, and other chemical components, where "other chemical components” refer to pharmaceutically acceptable compounds. Accepted carriers, excipients and/or one or more other therapeutic agents.
- Carrier refers to a material that does not cause significant irritation to the organism and does not eliminate the biological activity and characteristics of the administered compound.
- Excipient refers to an inert substance added to a pharmaceutical composition to facilitate the administration of a compound.
- Non-limiting examples include calcium carbonate, calcium phosphate, sugar, starch, cellulose derivatives (including microcrystalline cellulose), gelatin, vegetable oils, polyethylene glycols, diluents, granulating agents, lubricants, adhesives Agent and disintegrant.
- a “prodrug” refers to a compound of the present invention that can be converted into a biologically active compound by metabolism in the body.
- the prodrug of the present invention is prepared by modifying the amino or carboxyl group in the compound of the present invention, and this modification can be removed by conventional operations or in vivo to obtain the parent compound.
- the prodrug of the present invention is administered to a mammalian individual, the prodrug is split to form free amino or carboxyl groups.
- Co-crystal refers to the crystal formed by the combination of active pharmaceutical ingredient (API) and co-crystal former (CCF) under the action of hydrogen bonds or other non-covalent bonds.
- API active pharmaceutical ingredient
- CCF co-crystal former
- the pure state of API and CCF are both at room temperature. Solid, and there is a fixed stoichiometric ratio between the components.
- a eutectic is a multi-component crystal, which includes both a binary eutectic formed between two neutral solids and a multi-element eutectic formed between a neutral solid and a salt or solvate.
- Stepoisomers refer to isomers produced by the different arrangements of atoms in a molecule in space, including cis-trans isomers, enantiomers and conformational isomers.
- Optional or “optionally” or “selective” or “selectively” means that the event or condition described later can but does not necessarily occur, and the description includes the situation in which the event or condition occurs and its failures. What happened.
- heterocyclic group optionally substituted by an alkyl group means that the alkyl group may but does not necessarily exist, and the description includes the case where the heterocyclic group is substituted by an alkyl group and the case where the heterocyclic group is not substituted by an alkyl group.
- the structure of the compound is determined by nuclear magnetic resonance (NMR) or (and) mass spectrometry (MS).
- NMR shift ( ⁇ ) is given in units of 10 -6 (ppm).
- NMR is measured with (Bruker Avance III 400 and Bruker Avance 300) nuclear magnetic instrument, the solvent is deuterated dimethyl sulfoxide (DMSO-d 6 ), deuterated chloroform (CDCl 3 ), deuterated methanol (CD 3 OD) ), the internal standard is tetramethylsilane (TMS);
- HPLC determination uses Agilent 1260DAD high pressure liquid chromatograph (Zorbax SB-C18 100 ⁇ 4.6mm, 3.5 ⁇ M);
- the thin layer chromatography silica gel plate uses Yantai Huanghai HSGF254 or Qingdao GF254 silica gel plate.
- the size of the silica gel plate used for thin layer chromatography (TLC) is 0.15mm-0.20mm, and the size used for thin layer chromatography separation and purification products is 0.4mm. -0.5mm;
- the known starting materials of the present invention can be synthesized by or according to methods known in the art, or can be purchased from Titan Technology, Anaiji Chemical, Shanghai Demo, Chengdu Kelon Chemical, Shaoyuan Chemical Technology, Bailingwei Technology, etc. the company;
- Nitrogen atmosphere means that the reaction flask is connected to a nitrogen balloon with a volume of about 1L;
- the hydrogen atmosphere refers to the reaction flask connected to a hydrogen balloon with a volume of about 1L;
- the hydrogenation reaction is usually evacuated and filled with hydrogen, and the operation is repeated 3 times;
- the reaction temperature is room temperature, and the most suitable reaction temperature for room temperature is 20°C-30°C;
- THF Tetrahydrofuran
- PE petroleum ether
- NCS N-chlorosuccinimide
- Pd(dppf)Cl 2 [1,1'-bis(diphenylphosphine)ferrocene]palladium dichloride;
- DMSO dimethyl sulfoxide
- DNA Deoxyribonucleotide
- IC50 refers to the concentration of the compound when the activity of DNA-PK kinase is inhibited by 50%;
- X-Phos 2-Dicyclohexylphosphorus-2,4,6-triisopropylbiphenyl.
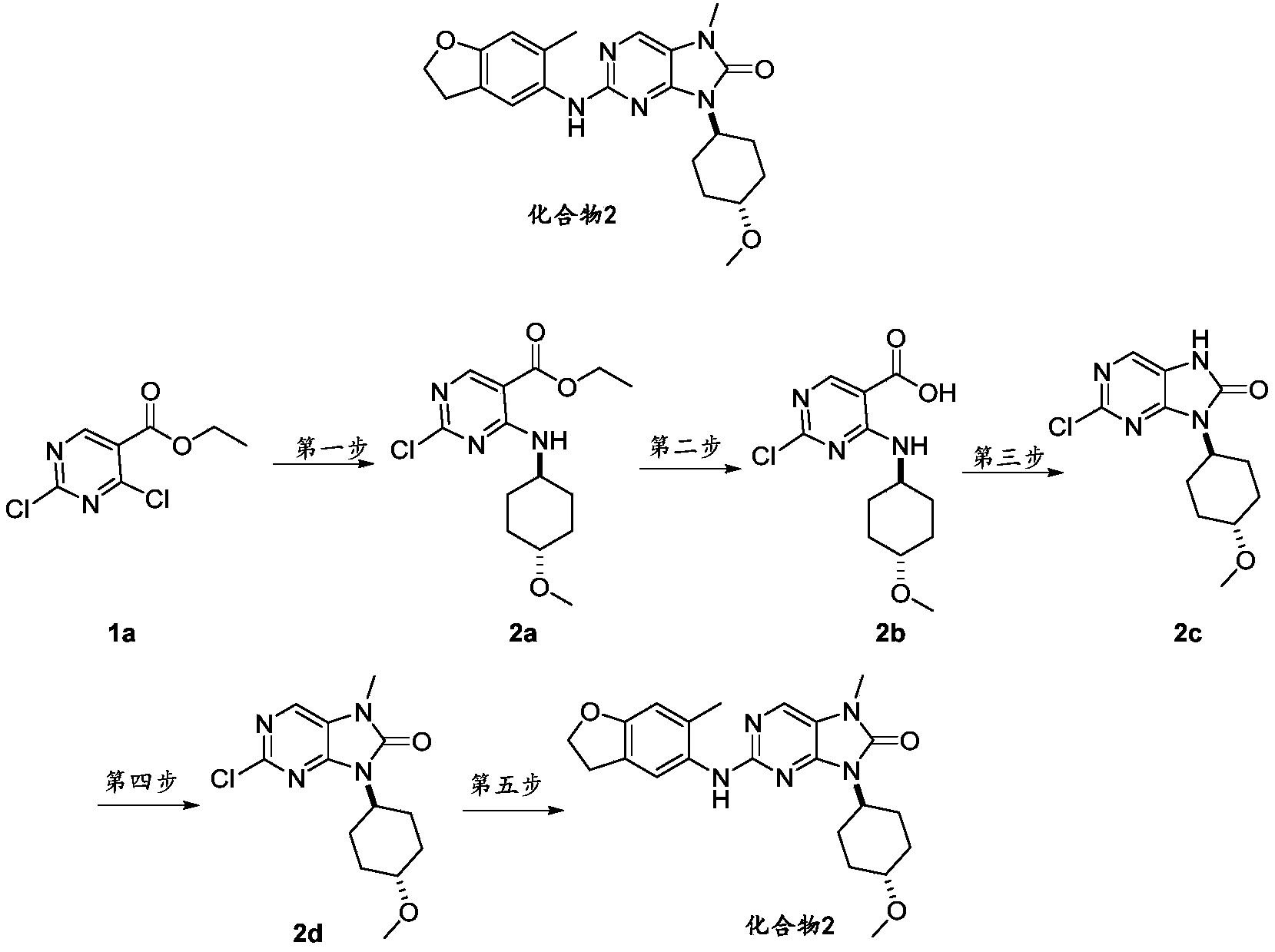
- reaction solution was quenched by adding water (20 mL), removing the organic solvent under reduced pressure, extracting twice with ethyl acetate, combining the organic phases, drying and concentrating, and using silica gel column chromatography to separate and purify (pure petroleum ether) to obtain The title compound 2C (colorless liquid, 15g, yield 98.42%)
- 2,4-Dichloropyrimidine-5-carboxylic acid ethyl ester 1a (30.00g, 136.4mmol), tetrahydro-2H-pyran-4-amine hydrochloride (18.66g, 136.4mmol) were dissolved in acetonitrile (600mL ), adding potassium carbonate (46.92g, 340.9mmol) several times with stirring, and stirring at room temperature for 4h. After the reaction was monitored by TLC, it was filtered. The residue was washed with ethyl acetate (300 mL). The filtrate was concentrated to obtain a crude product.
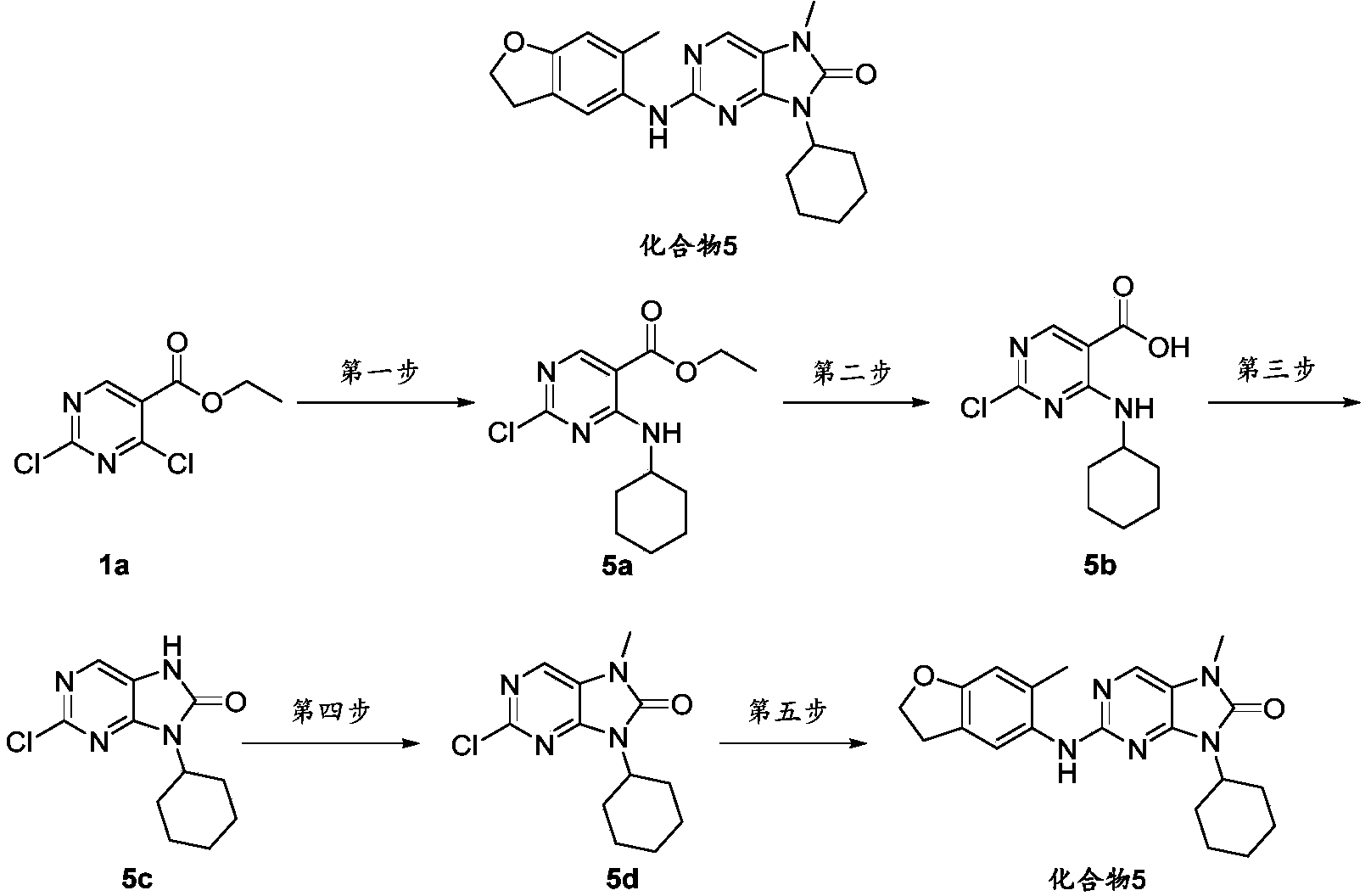
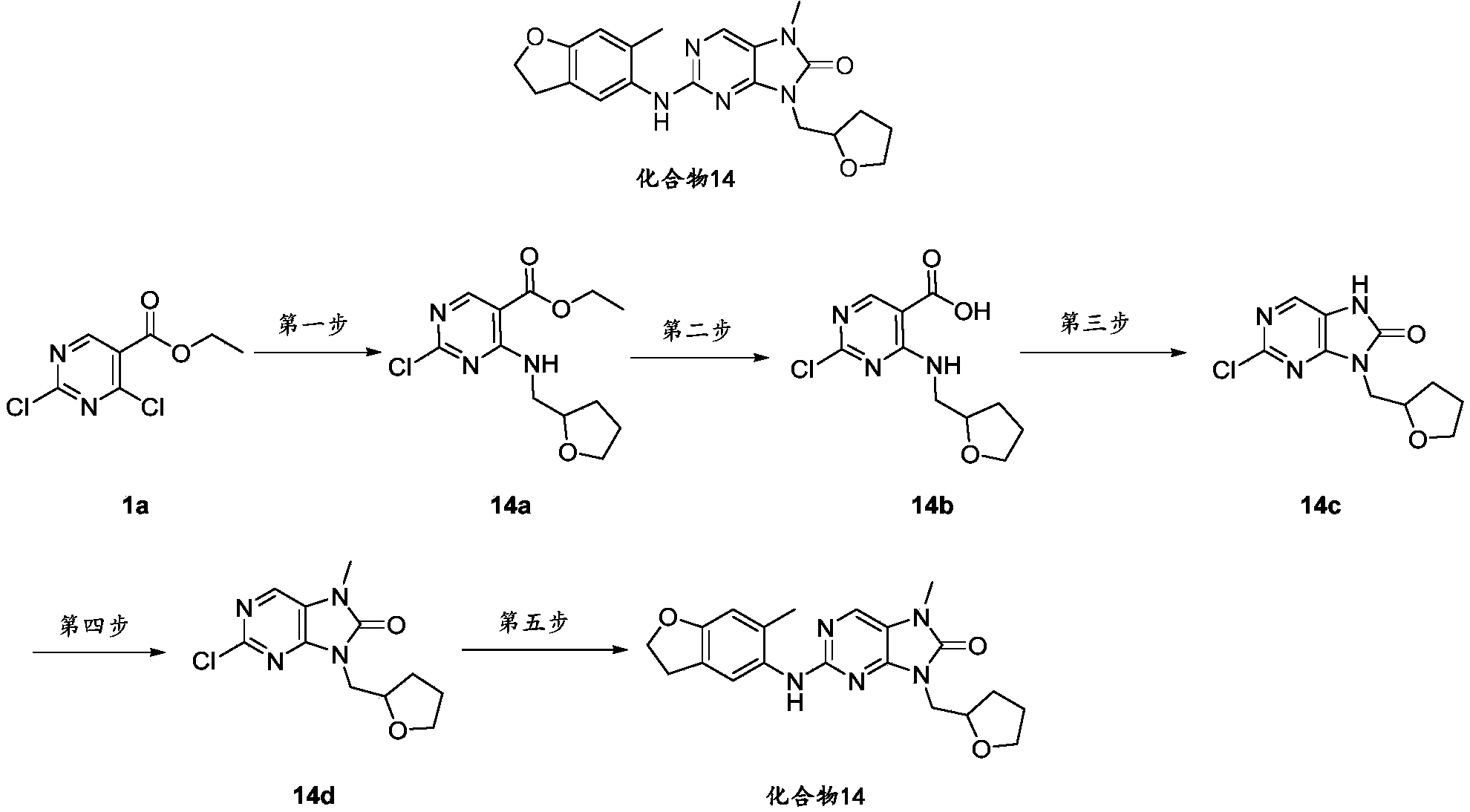
- the 2-chloro-7-methyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one 1e (126mg, 0.47mmol), 6- Methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (70mg, 0.47mmol), cesium carbonate (305mg, 0.94mmol), tris(dibenzylideneacetone) two palladium (42mg, 0.047mmol) ) 2,2'-bis(diphenylphosphino)-1,1'-binaphthyl (58mg, 0.094mmol) was dissolved in dioxane (10mL), protected with nitrogen and ventilated, and stirred at 100°C for 4h.
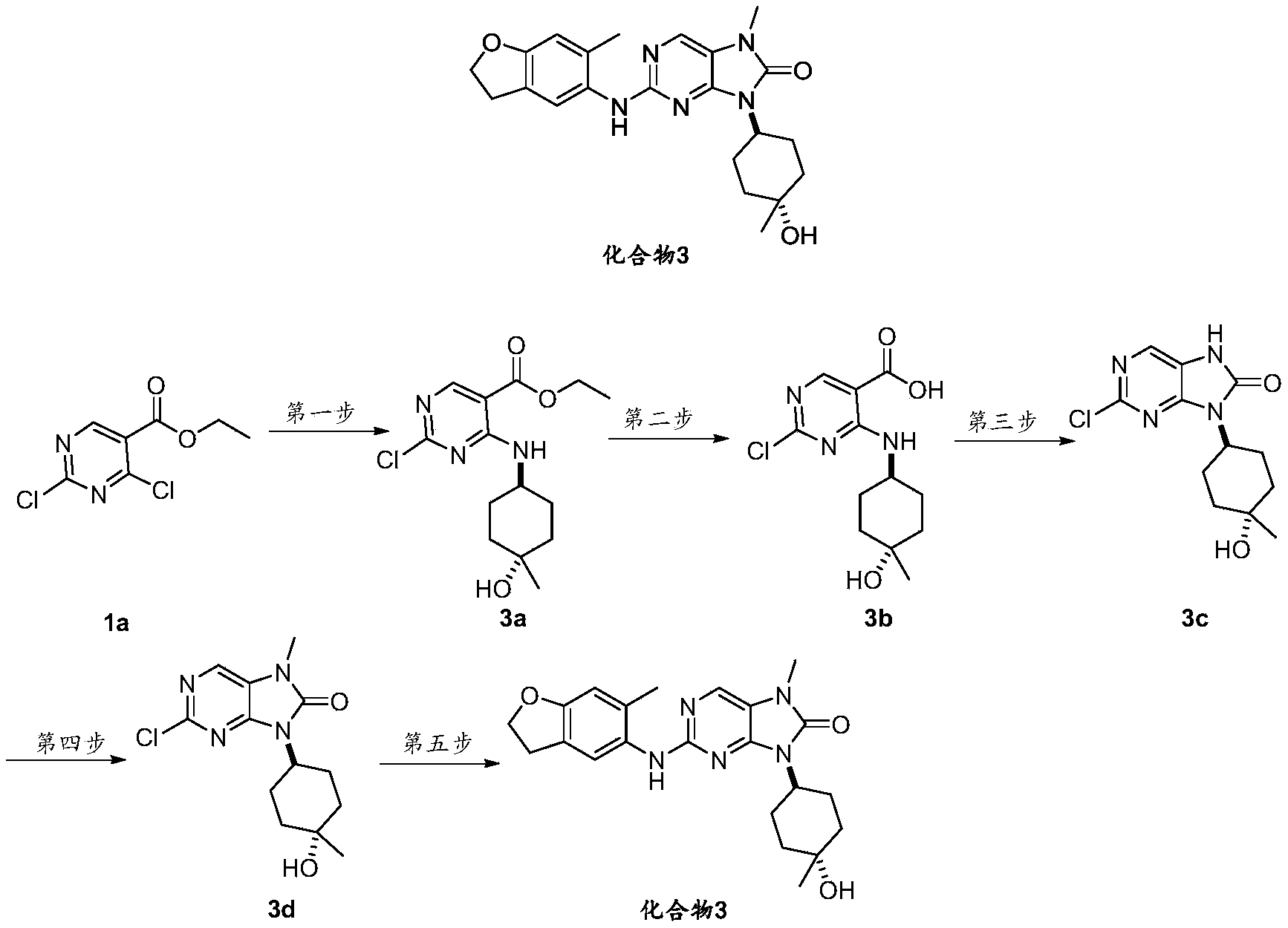
- Ethyl 2,4-dichloro-5-pyrimidinecarboxylate 1a (5.00g, 30.18mmol) was dissolved in acetonitrile (30mL), potassium carbonate (12.51g, 90.55mmol) was added with stirring at 0°C, and the reaction was slowly added dropwise.
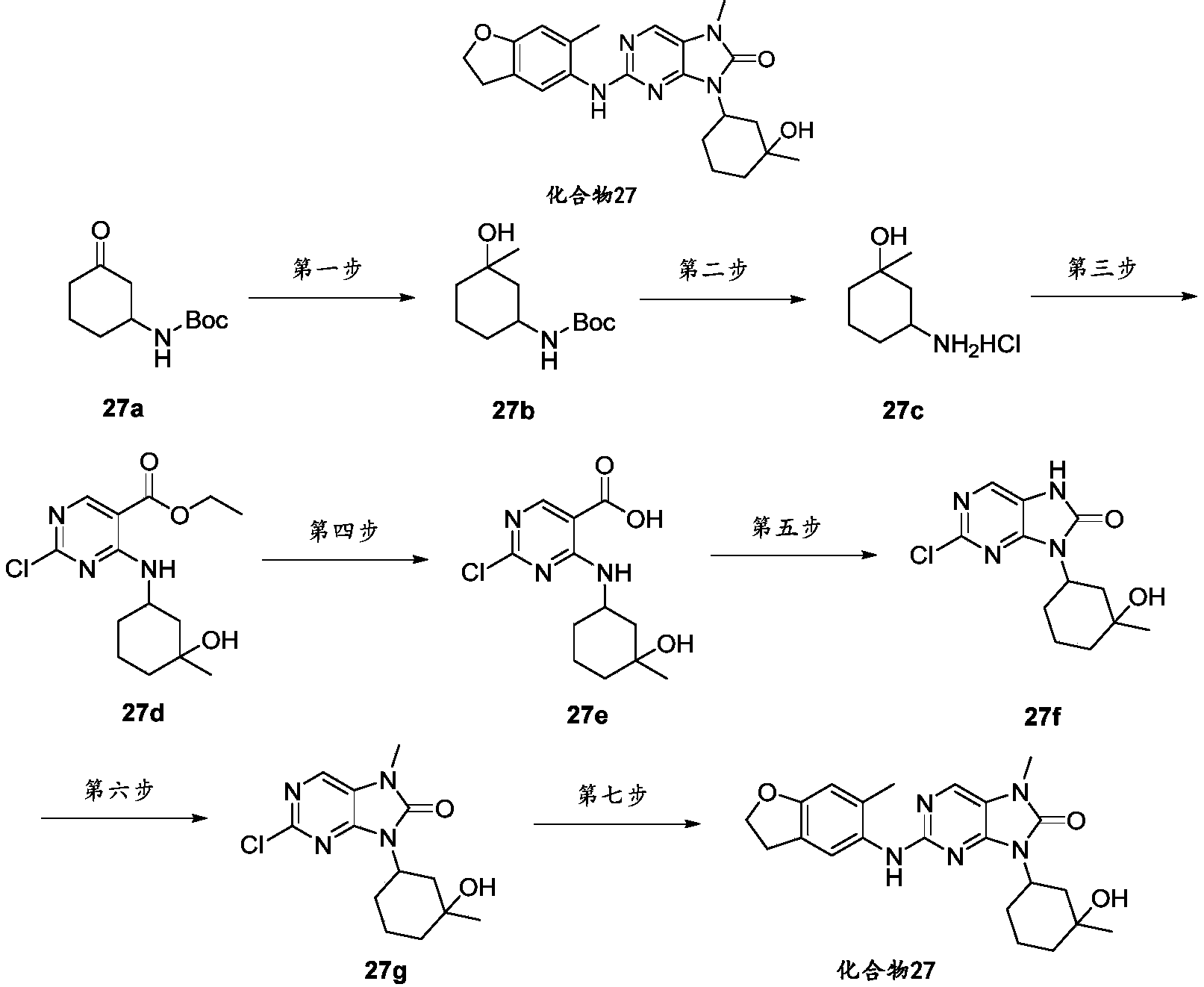
- the acetonitrile solution (10 mL) of -4-amino-1-methylcyclohexanol hydrochloride (10.01 g, 45.27 mmol) was heated to room temperature and the reaction was stirred for 1 h, monitored by TLC until the reaction was over, filtered through diatomaceous earth, and chromatographed on a silica gel column.
- the seventh step is a first step.
- the 2-chloro-9-(cis-4-methoxycyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one 12d (100mg, 0.34mmol), 6-methyl -2,3-Dihydrobenzofuran-5-amine intermediate 1 (100mg, 0.68mmol), cesium carbonate (220mg, 0.68mmol) and Brettphos G3 Pd (30mg, 0.034mmol) were added to the dry reaction flask and replaced with nitrogen After three times, dry 1,4-dioxane (1 mL) was added and reacted at 110° C. for 5 h. The reaction was monitored by TLC until the end of the reaction.
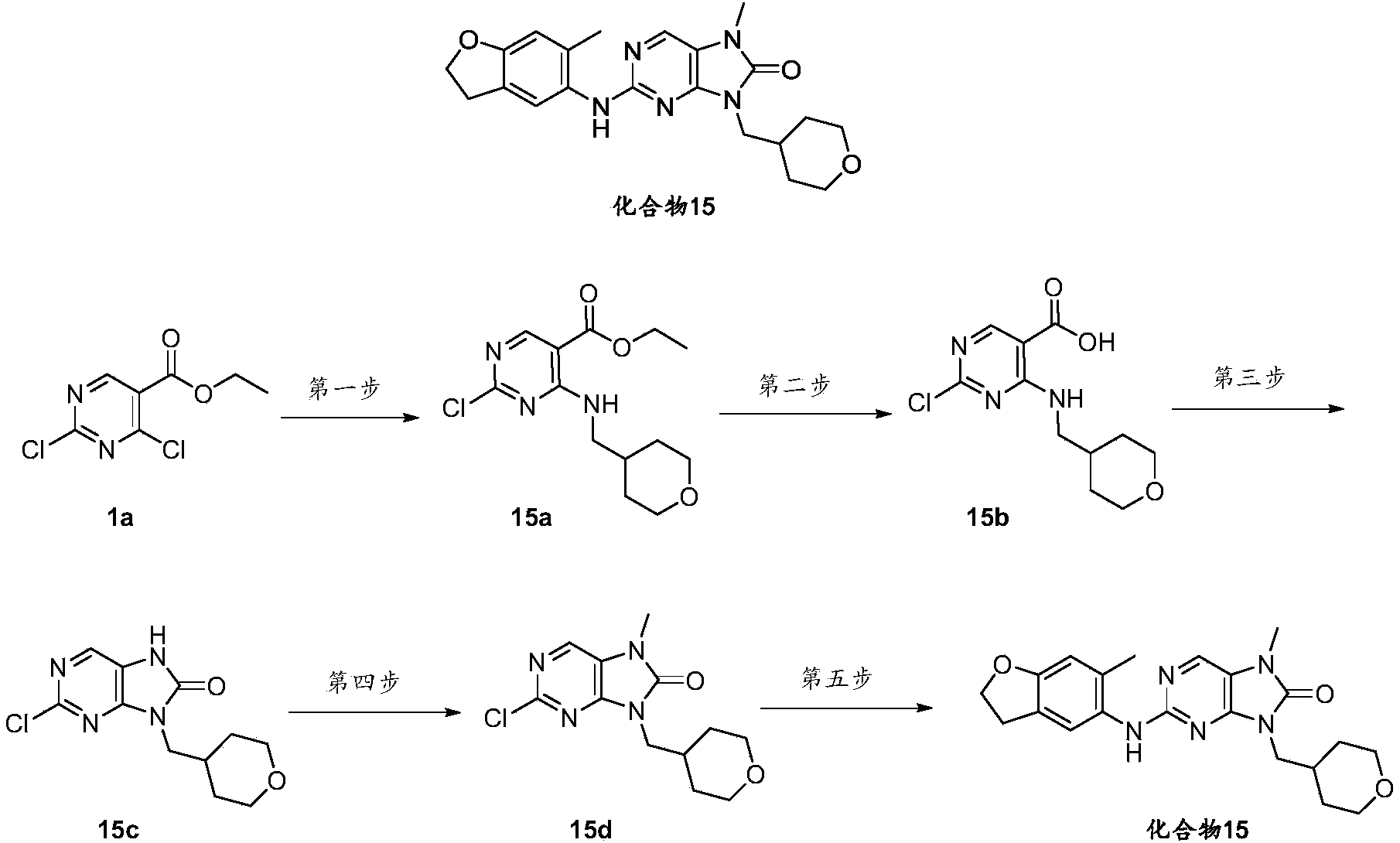
- the 2-chloro-7-methyl-9-((tetrahydro-2H-pyran-4-yl)methyl)-7,9-dihydro-8H-purin-8-one 15d (100mg, 0.35mmol ), 6-methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (106mg, 0.71mmol), cesium carbonate (230mg, 0.71mmol) and Brettphos G3 Pd (32mg, 0.035mmol) were added to the dry After replacing the reaction flask with nitrogen for three times, add dry 1,4-dioxane (1 mL), and react at 110°C for 5 hours. The reaction was monitored by TLC until the end of the reaction.
- the seventh step is a first step.
- the seventh step is a first step.
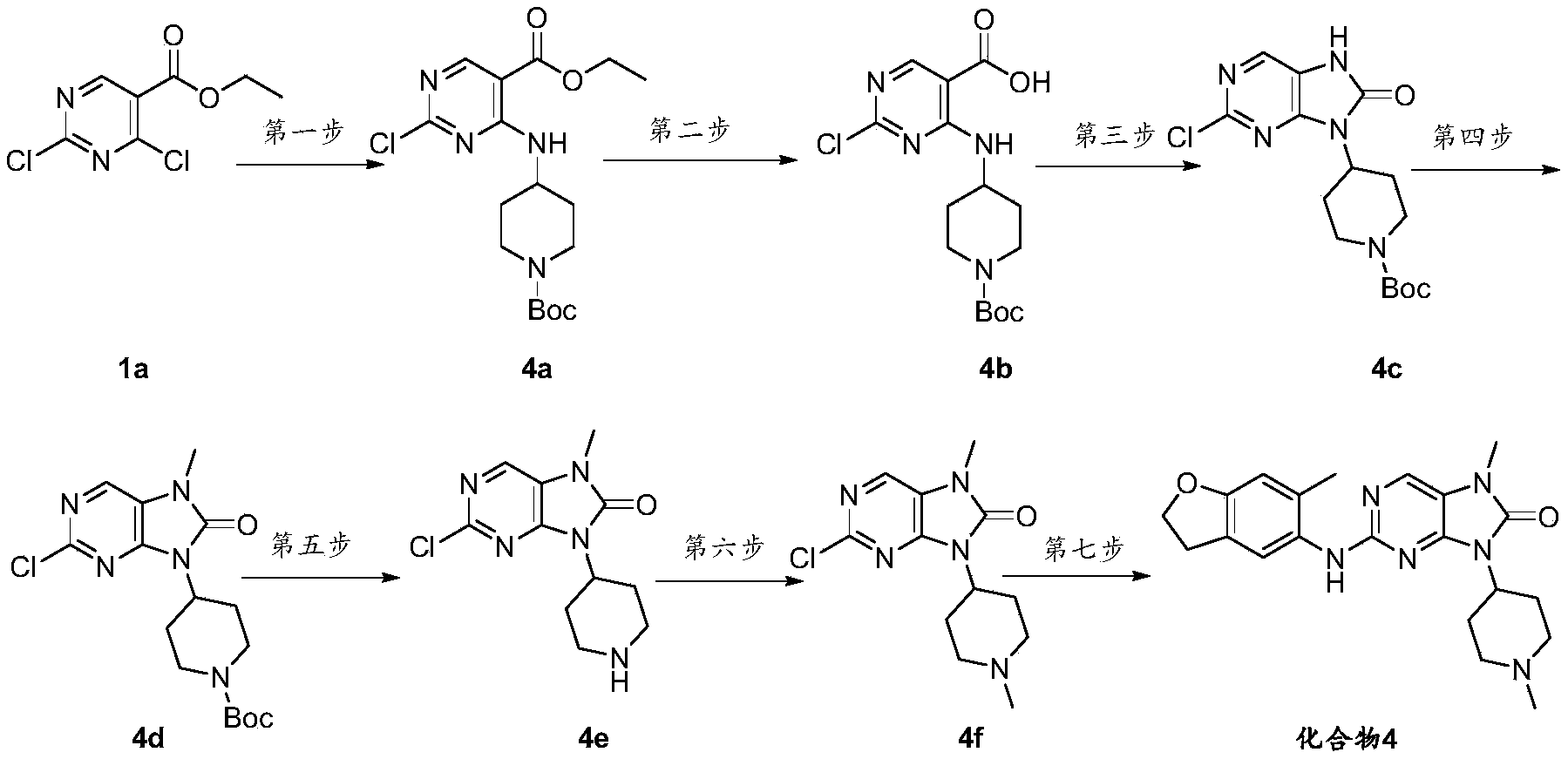
- tert-butyl (3-oxocyclohexyl) carbamate 18a (8.20g, 38.45mmol) in tetrahydrofuran (40mL), add sodium borohydride (4.36g, 115.35mmol) under stirring at 0°C and react for 3h, TLC monitors to the end of the reaction, slowly add saturated sodium carbonate solution to the reaction solution, stir at room temperature for 3 hours, add ethyl acetate for extraction, concentrate the organic layer to obtain the title compound tert-butyl (3-hydroxycyclohexyl) carbamate (18b) (Yellow liquid, 8.00 g, yield 96.95%).
- the seventh step is a first step.
- TLC monitors to the end of the reaction add 10 mL of water to the reaction solution, separate the organic phase, wash with saturated brine once, dry with anhydrous sodium sulfate and mix the sample with silica gel, pass the product with a normal phase column passer, and concentrate to obtain the title compound tert-butyl Group (trans-4-carbamoylcyclohexyl) carbamate (19b) (white solid, 4.4 g, yield 83%).
- the seventh step is a first step.
- TLC monitors to the end of the reaction spin-dry the tetrahydrofuran, adjust the pH to 1-2 with 2N hydrochloric acid, precipitate a solid, filter, and wash the filter cake with water to dry to obtain the title compound 2-chloro-4-((2-isopropyl-4- Methylpyridin-3-yl)amino)pyrimidine-5-carboxylic acid (20b) (brown solid, 29.29 g, yield 72.66%).
- the 2-chloro-9-(2-isopropyl-4-methylpyridin-3-yl)-7-methyl-7,9-dihydro-8H-purin-8-one 20d (80mg, 0.25mmol ), 6-methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (75mg, 0.50mmol), cesium carbonate (164mg, 0.50mmol) and Brettphos G3 Pd (23mg, 0.025mmol) were added to the dry The reaction tube was replaced with N 2 three times, then dry 1,4-dioxane (1 mL) was added, and the reaction was carried out at 110°C for 5 hours.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Epidemiology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
本发明涉及呋喃衍生物,或者其立体异构体、溶剂化物、前药、代谢产物、氘代物、药学上可接受的盐或共晶,其药物组合物以及在制备DNA-PK抑制剂的用途。The present invention relates to furan derivatives, or their stereoisomers, solvates, prodrugs, metabolites, deuterated substances, pharmaceutically acceptable salts or co-crystals, their pharmaceutical compositions and their use in the preparation of DNA-PK inhibitors .
DNA依赖的蛋白激酶(DNA-dependent protein kinase,DNA-PK)是由Ku70/Ku80异二聚体和DNA依赖的蛋白激酶催化亚基(DNA-PKcs)构成的DNA-PK酶复合物。该酶复合物需要在DNA参与下才能被激活发挥出相应的功能(George et al.,2019)。作为一种丝氨酸/苏氨酸蛋白激酶,DNA-PK属于PIKK(phosphatidylinositol 3-kinase-related kinase)家族成员,它不仅在修复细胞内DNA双链断裂(double-strand breaks;DSBs)和细胞DNA重组或抗体DNA重排(V(D)J重组)过程中具有重要作用,还参与染色体修饰、转录调节、端粒维持等生理过程。DNA-dependent protein kinase (DNA-PK) is a DNA-PK enzyme complex composed of Ku70/Ku80 heterodimer and DNA-dependent protein kinase catalytic subunit (DNA-PKcs). The enzyme complex needs to be activated with the participation of DNA to perform its corresponding functions (George et al., 2019). As a serine/threonine protein kinase, DNA-PK belongs to the PIKK (phosphatidylinositol 3-kinase-related kinase) family. It not only repairs intracellular DNA double-strand breaks (DSBs) and cell DNA recombination Or it plays an important role in the process of antibody DNA rearrangement (V(D)J recombination), and also participates in physiological processes such as chromosome modification, transcription regulation, and telomere maintenance.
在正常生理过程中,多种因素可能导致DNA发生DSBs:如体细胞DNA重组过程中DSBs常常作为中间产物出现,这一生理过程对所有脊椎动物的功能性免疫系统的形成十分重要;DNA复制中复制叉遇到受损的碱基,也可能造成单链或双链断裂;DNA也可能因为正常代谢过程中活性氧(reactive oxygen species;ROS)的攻击而产生DSBs(Cannan&Pederson,2016)。此外,还有多种外源性因素也可能导致DSBs,如电离辐射(Ionizing radiation,IR)和化疗试剂(如拓扑异构酶II抑制剂)等(George et al.,2019)。如果DSBs未被修复或者错误地修复,将会产生突变和/或染色体畸变,最终导致细胞死亡。为了应对DSBs带来的危害,真核细胞已进化出多种机制来修复受损的DNA以维持细胞的活力和基因组的稳定性。在真核细胞中,最主要的DNA修复方式是非同源末端连接(non-homologous end-joining,NHEJ)。这种直接将断裂DNA连接起来的方式并不需要有同源DNA片段参与,可以发生在细胞周期的任何阶段。NHEJ是由DNA-PK介导的需要多种蛋白与信号通路共同参与的动态过程,基本过程如下:(1)Ku70/Ku80异二聚体识别并结合至双链DNA断裂末端;(2)募集DNA-PKcs、XRCC4-DNA连接酶IV复合体等蛋白至DNA断裂双链的两侧;(3)DNA-PKcs自身磷酸化,激活自身的激酶活性;(4)DNA-PKcs作为粘合剂连接断裂DNA的两端,防止核酸外切酶对DN A的降解作用;(5)对DNA进行加工以移除断裂处的不可连接末端或其他损伤形式;(6)XRCC4-DNA连接酶IV复合体修复DNA末端(某些情况下,在连接之前可能还需要D NA聚合酶来合成新的末端)。当DNA-PKcs发生磷酸化后,可诱导蛋白构象发生改变,调节NHEJ过程中多种蛋白的活性(如Artemis、Ku70、Ku80、DNA ligase),这对DN A修复过程至关重要。因此,磷酸化的DNA-PKcs(pDNA-PKcs)常常作为细胞DSBs的标志物。In the normal physiological process, a variety of factors may lead to the occurrence of DSBs in DNA: For example, DSBs often appear as intermediate products in the process of somatic DNA recombination. This physiological process is very important for the formation of the functional immune system of all vertebrates; DNA replication is in progress. When the replication fork encounters damaged bases, it may also cause single-strand or double-strand breaks; DNA may also generate DSBs due to the attack of reactive oxygen species (ROS) during normal metabolism (Cannan & Pederson, 2016). In addition, there are a variety of exogenous factors that may also cause DSBs, such as ionizing radiation (IR) and chemotherapeutic agents (such as topoisomerase II inhibitors) (George et al., 2019). If DSBs are not repaired or repaired incorrectly, mutations and/or chromosomal aberrations will occur, eventually leading to cell death. In order to cope with the harm caused by DSBs, eukaryotic cells have evolved a variety of mechanisms to repair damaged DNA to maintain cell viability and genome stability. In eukaryotic cells, the most important way of DNA repair is non-homologous end-joining (NHEJ). This method of directly connecting broken DNA does not require the participation of homologous DNA fragments, and can occur at any stage of the cell cycle. NHEJ is a dynamic process mediated by DNA-PK that requires the participation of multiple proteins and signaling pathways. The basic process is as follows: (1) Ku70/Ku80 heterodimer recognizes and binds to the ends of double-stranded DNA breaks; (2) Recruitment DNA-PKcs, XRCC4-DNA ligase IV complex and other proteins to both sides of the DNA break double-strand; (3) DNA-PKcs autophosphorylate and activate its own kinase activity; (4) DNA-PKcs as an adhesive to connect Break both ends of the DNA to prevent exonuclease from degrading DN A; (5) Process the DNA to remove the unlinkable ends or other forms of damage at the break; (6) XRCC4-DNA ligase IV complex Repair DNA ends (in some cases, DNA polymerase may be required to synthesize new ends before ligation). When DNA-PKcs is phosphorylated, it can induce protein conformation to change and regulate the activity of various proteins in the NHEJ process (such as Artemis, Ku70, Ku80, DNA ligase), which is essential for the repair process of DN A. Therefore, phosphorylated DNA-PKcs (pDNA-PKcs) is often used as a marker of cellular DSBs.
已有研究表明,DNA-PK活性与多种肿瘤的发生发展有关:如黑色素瘤中的DNA-PKcs可以促进血管再生和肿瘤的转移;多发性骨髓瘤中的DNA-PKcs表达量显著上调;放疗耐受的甲状腺肿瘤中的Ku蛋白的含量明显增加(Ihara,Ashizawa,Shichijo,&Kudo,2019)。因此,可以考虑将DNA-PK抑制剂与引起DNA损伤的抗肿瘤疗法(如IR、化疗试剂等)联用来提高效果。DNA-PK抑制剂的使用在一定程度上会干扰正常细胞的DNA修复功能,然而正常细胞体内还存在多种DNA修复途径作为补充,而肿瘤细胞面临强大的DNA复制压力且缺乏有效的DNA修复方式。通过抑制肿瘤细胞DNA-PK的活性能够提高其他抗肿瘤药物对肿瘤细胞的杀伤效果。Studies have shown that DNA-PK activity is related to the occurrence and development of a variety of tumors: for example, DNA-PKcs in melanoma can promote angiogenesis and tumor metastasis; DNA-PKcs expression in multiple myeloma is significantly up-regulated; radiotherapy The content of Ku protein in tolerant thyroid tumors is significantly increased (Ihara, Ashizawa, Shichijo, & Kudo, 2019). Therefore, it can be considered to combine DNA-PK inhibitors with anti-tumor therapies that cause DNA damage (such as IR, chemotherapeutic agents, etc.) to improve the effect. The use of DNA-PK inhibitors can interfere with the DNA repair function of normal cells to a certain extent. However, there are many DNA repair pathways in normal cells as a supplement, and tumor cells face strong DNA replication pressure and lack effective DNA repair methods. . By inhibiting the activity of tumor cell DNA-PK, the killing effect of other anti-tumor drugs on tumor cells can be improved.
经多年研究,目前已经发现了多个DNA-PK抑制剂。最早发现具有DNA-PK激酶抑制活性的化合物是一种真菌代谢产物——Wortmannin,IC50(DNA-PK)约15nM,该化合物同时在p53蛋白的乙酰化和磷酸化过程中也发挥着重要作用(Sarkaria et al.,1998); 之后报道的槲皮素衍生物LY294002也具有DNA-PK抑制活性(Maira,Stauffer,Schnell,&Garcia-Echeverria,2009);后来基于LY294002结构又研发了NU7026、NU7441等新一代DNA-PK抑制剂。虽然已经证实了这些化合物对肿瘤细胞有着良好的杀伤效果,但它们存在高毒性、选择性差等问题而无法进入临床开发(Maira et al.,2009)。还曾报道过其他DNA-PK抑制剂,如OK1035、SU11752、PP121、KU-0060648等小分子化合物,但这些化合物同样存在对DNA-PK特异性较低等缺陷(George et al.,2019)。所以,目前仍然需要开发高活性、高特异性、低毒性的DNA-PK抑制剂,以更好满足临床需求。After years of research, several DNA-PK inhibitors have been discovered. The first compound found to have DNA-PK kinase inhibitory activity is a fungal metabolite-Wortmannin, with an IC50 (DNA-PK) of about 15nM. This compound also plays an important role in the acetylation and phosphorylation of p53 protein ( Sarkaria et al., 1998); The quercetin derivative LY294002 reported later also has DNA-PK inhibitory activity (Maira, Stauffer, Schnell, & Garcia-Echeverria, 2009); later based on the structure of LY294002, NU7026 and NU7441 were developed A generation of DNA-PK inhibitors. Although it has been confirmed that these compounds have a good killing effect on tumor cells, they have problems such as high toxicity and poor selectivity and cannot enter clinical development (Maira et al., 2009). Other DNA-PK inhibitors have also been reported, such as OK1035, SU11752, PP121, KU-0060648 and other small molecule compounds, but these compounds also have defects such as low specificity for DNA-PK (George et al., 2019). Therefore, there is still a need to develop DNA-PK inhibitors with high activity, high specificity, and low toxicity to better meet clinical needs.
发明内容Summary of the invention
本申请的一个或多个实施方式提供呋喃衍生物,或者其立体异构体、溶剂化物、前药、代谢产物、氘代物、药学上可接受的盐或共晶,其药物组合物以及其在制备DNA-PK抑制剂;这些化合物对DNA-PK具有高抑制活性且高选择性,且能够作为化疗和放疗增敏剂有效治疗癌症,改善现有技术的疗效,同时降低毒副作用。One or more embodiments of the present application provide furan derivatives, or their stereoisomers, solvates, prodrugs, metabolites, deuterated products, pharmaceutically acceptable salts or co-crystals, and their pharmaceutical compositions and Preparation of DNA-PK inhibitors; these compounds have high inhibitory activity and high selectivity to DNA-PK, and can be used as chemotherapy and radiotherapy sensitizers to effectively treat cancer, improve the curative effect of the prior art, and reduce toxic and side effects.
本申请的一个或多个实施方式提供通式(I’)的化合物,或者其立体异构体、溶剂化物、前药、代谢产物、氘代物、药学上可接受的盐或共晶:One or more embodiments of the present application provide a compound of general formula (I'), or its stereoisomers, solvates, prodrugs, metabolites, deuterated products, pharmaceutically acceptable salts or co-crystals:
其中:among them:
R 0、R 1各自独立地选自H、-OH、氰基、卤素、-NH 2、C 1-6烷基或者C 1-6烷氧基,所述的C 1-6烷基任选进一步被1-3个选自D或者卤素的取代基所取代; R 0 and R 1 are each independently selected from H, -OH, cyano, halogen, -NH 2 , C 1-6 alkyl or C 1-6 alkoxy, the C 1-6 alkyl optionally Further substituted by 1-3 substituents selected from D or halogen;
或者两个R 0与其相连的原子形成3至8元环,所述的3至8元环任选地包含1至3个选自N、O或者S的杂原子,所述的3至8元环任选进一步被1个或者多个选自-OH、羧基、卤素、氰基、=O、C 1-6烷基或者氨基的取代基所取代; Or two R 0 and the atoms to which they are connected form a 3- to 8-membered ring, and the 3- to 8-membered ring optionally contains 1 to 3 heteroatoms selected from N, O or S, and the 3- to 8-membered ring The ring is optionally further substituted with one or more substituents selected from -OH, carboxy, halogen, cyano, =0, C 1-6 alkyl or amino;
R 2选自H或者C 1-6烷基; R 2 is selected from H or C 1-6 alkyl;
R 3选自C 1-6烷基、C 3-12碳环基、C 3杂环基、C 4-12杂环基、-C 1-6亚烷基-C 3-12碳环基、-C 1-6亚烷基-C 3杂环基、-C 1-6亚烷基-C 4-12杂环基、C 6-12螺环化合物或者C 6-12杂螺环化合物,所述的C 3杂环基和C 4-12杂环基包含1至3个选自N、O或者S的杂原子,所述的C 1-6烷基、C 3-12碳环基、C 3杂环基、C 4-12杂环基、C 1-6亚烷基任选进一步被1个或者多个选自-OH、-OR a1、羧基、卤素、氰基、=O、C 1-6烷基、C 1-6杂烷基、C 2-6烯基、C 2-6炔基、-NR a1R a2、-C(=O)OC 1-6烷基、-C(=O)NR a1R a2、C 3-12环烷基、C 3杂环烷基、C 4-12杂环烷基、C 6-12芳基或者C 5-12杂芳基的取代基所取代;且所述取代基中的所述的C 1-6烷基、C 1-6杂烷基、C 2-6烯基或者C 2-6炔基任选进一步被1个或者多个选自-OH、羧基、氰基、卤素、-O-R a1、-NR a1R a2、C 3-12环烷基、C 3杂环烷基、C 4-12杂环烷基、C 6-12芳基或者C 5-12杂芳基的取代基所取代; R 3 is selected from C 1-6 alkyl, C 3-12 carbocyclic group, C 3 heterocyclic group, C 4-12 heterocyclic group, -C 1-6 alkylene-C 3-12 carbocyclic group, -C 1-6 alkylene-C 3 heterocyclic group, -C 1-6 alkylene-C 4-12 heterocyclic group, C 6-12 spiro compound or C 6-12 heterospiro compound, so The C 3 heterocyclic group and C 4-12 heterocyclic group contain 1 to 3 heteroatoms selected from N, O or S, the C 1-6 alkyl group, C 3-12 carbocyclic group, C 3 Heterocyclic group, C 4-12 heterocyclic group, C 1-6 alkylene group may be further selected by one or more selected from -OH, -OR a1 , carboxy, halogen, cyano, =O, C 1 -6 alkyl, C 1-6 heteroalkyl, C 2-6 alkenyl, C 2-6 alkynyl, -NR a1 R a2 , -C(=O)OC 1-6 alkyl, -C(= O) NR a1 R a2 , C 3-12 cycloalkyl, C 3 heterocycloalkyl, C 4-12 heterocycloalkyl, C 6-12 aryl or C 5-12 heteroaryl substituent substituted And the C 1-6 alkyl group, C 1-6 heteroalkyl group, C 2-6 alkenyl group or C 2-6 alkynyl group in the substituent is optionally further selected from one or more -OH, carboxyl, cyano, halogen, -OR a1 , -NR a1 R a2 , C 3-12 cycloalkyl, C 3 heterocycloalkyl, C 4-12 heterocycloalkyl, C 6-12 aryl Or substituted by a substituent of a C 5-12 heteroaryl group;
R 4选自H、C 1-6烷基、C 3-12环烷基,所述的C 1-6烷基和C 3-8环烷基任选进一步被1-3个选自D或者卤素的取代基所取代; R 4 is selected from H, C 1-6 alkyl, C 3-12 cycloalkyl, said C 1-6 alkyl and C 3-8 cycloalkyl are optionally further selected from 1-3 from D or Substituted by halogen substituents;
R a1、R a2各自独立地选自H、C 1-6烷基、-C(=O)R a3或者-C(=O)NR a4R a5,其中所述的C 1-6烷基任选进一步被1个或者多个选自OH、卤素、C 1-6烷基、C 1-6烷氧基、C 6-12芳基、C 5-12杂芳基、C 3-12环烷基、C 3杂环烷基、或者C 4-12杂环烷基的取代基所取代;或者R a4与R a5及N原子形成3至8元杂环,所述的3至8元杂环包含1个至3个选自N、O或者S的杂原子; R a1 and R a2 are each independently selected from H, C 1-6 alkyl, -C(=O)R a3 or -C(=O)NR a4 R a5 , wherein the C 1-6 alkyl is any Optionally, one or more selected from OH, halogen, C 1-6 alkyl, C 1-6 alkoxy, C 6-12 aryl, C 5-12 heteroaryl, C 3-12 cycloalkane Group, C 3 heterocycloalkyl, or C 4-12 heterocycloalkyl substituent; or Ra4, Ra5 and N atom form a 3- to 8-membered heterocyclic ring, said 3- to 8-membered heterocyclic ring Contain 1 to 3 heteroatoms selected from N, O or S;
R a3选自C 1-6烷基、C 1-6烷氧基或者C 6-12芳基; R a3 is selected from C 1-6 alkyl, C 1-6 alkoxy or C 6-12 aryl;
n为0、1、2或者3;n is 0, 1, 2 or 3;
p为0、1、2或者3。p is 0, 1, 2, or 3.
本申请的一个或多个实施方式提供了化合物,或者其立体异构体、溶剂化物、前药、代谢产物、氘代物、药学上可接受的盐或共晶,其中该化合物选自通式(I)所示的化合物:One or more embodiments of the application provide a compound, or its stereoisomer, solvate, prodrug, metabolite, deuterated product, pharmaceutically acceptable salt or co-crystal, wherein the compound is selected from the general formula ( I) The compound shown:
其中:among them:
R 0、R 1选自H、-OH、氰基、卤素、-NH 2、C 1-6烷基或者C 1-6烷氧基,所述的烷基任选进一步被1-3个选自D或者卤素的取代基所取代; R 0 and R 1 are selected from H, -OH, cyano, halogen, -NH 2 , C 1-6 alkyl or C 1-6 alkoxy, and the alkyl group is optionally further selected from 1-3 Substituted by D or halogen substituent;
或者两个R 0与其相连的原子可以形成3至8元环,所述的环可含有1至3个选自N、O或者S的杂原子,所述的环任选进一步被1个或者多个选自-OH、羧基、卤素、氰基、=O、C 1-6烷基或者氨基的取代基所取代; Or two R 0 and the atoms to which they are connected can form a 3- to 8-membered ring, the ring may contain 1 to 3 heteroatoms selected from N, O or S, and the ring is optionally further divided by 1 or more Substituted by a substituent selected from -OH, carboxyl, halogen, cyano, =O, C 1-6 alkyl or amino;
R 2选自H或者C 1-6烷基; R 2 is selected from H or C 1-6 alkyl;
R 3选自C 1-6烷基、C 3-12碳环基、C 3杂环基、C 4-12杂环基、-C 1-6亚烷基-C 3-12碳环基、-C 1-6亚烷基-C 3杂环基或者-C 1-6亚烷基-C 4-12杂环基,所述的杂环基可以含有1至3个选自N、O或者S的杂原子,所述的烷基、碳环基、杂环基、亚烷基任选进一步被1个或者多个选自-OH、羧基、卤素、氰基、=O、C 1-6烷基、C 1-6杂烷基、C 2-6烯基、C 2-6炔基、-NR a1R a2、-C(=O)OC 1-6烷基、-C(=O)NR a1R a2、C 3-12环烷基、C 3杂环烷基、C 4-12杂环烷基、C 6-12芳基或者C 5-12杂芳基的取代基所取代;且所述的烷基、杂烷基、烯基或者炔基任选进一步被1个或者多个选自-OH、羧基、氰基、卤素、-O-R a1、-NR a1R a2、C 3-12环烷基、C 3杂环烷基、C 4-12杂环烷基、C 6-12芳基或者C 5-12杂芳基的取代基所取代; R 3 is selected from C 1-6 alkyl, C 3-12 carbocyclic group, C 3 heterocyclic group, C 4-12 heterocyclic group, -C 1-6 alkylene-C 3-12 carbocyclic group, -C 1-6 alkylene-C 3 heterocyclic group or -C 1-6 alkylene-C 4-12 heterocyclic group, said heterocyclic group may contain 1 to 3 selected from N, O or S heteroatom, the alkyl group, carbocyclic group, heterocyclic group, alkylene group may be further selected by one or more selected from -OH, carboxyl, halogen, cyano, =0, C 1-6 Alkyl, C 1-6 heteroalkyl, C 2-6 alkenyl, C 2-6 alkynyl, -NR a1 R a2 , -C(=O)OC 1-6 alkyl, -C(=O) NR a1 R a2 , C 3-12 cycloalkyl, C 3 heterocycloalkyl, C 4-12 heterocycloalkyl, C 6-12 aryl or C 5-12 heteroaryl substituents are substituted; and The alkyl, heteroalkyl, alkenyl or alkynyl group is optionally further selected by one or more selected from -OH, carboxyl, cyano, halogen, -OR a1 , -NR a1 R a2 , C 3-12 Cycloalkyl, C 3 heterocycloalkyl, C 4-12 heterocycloalkyl, C 6-12 aryl or C 5-12 heteroaryl substituents;
R a1、R a2选自H、C 1-6烷基、-C(=O)R a3或者-C(=O)NR a4R a5,其中所述的C 1-6烷基任选进一步被1个或者多个选自OH、卤素、C 1-6烷基、C 1-6烷氧基、C 6-12芳基、C 5-12杂芳基、C 3-12环烷基、C 3杂环烷基、或者C 4-12杂环烷基的取代基所取代;或者R a4与R a5及N原子形成一个3至8元杂环,所述的环可以含有1个或者多个选自N、O或者S的杂原子; R a1 and R a2 are selected from H, C 1-6 alkyl, -C(=O)R a3 or -C(=O)NR a4 R a5 , wherein the C 1-6 alkyl is optionally further One or more selected from OH, halogen, C 1-6 alkyl, C 1-6 alkoxy, C 6-12 aryl, C 5-12 heteroaryl, C 3-12 cycloalkyl, C 3 heterocycloalkyl, or substituted by a substituent of C 4-12 heterocycloalkyl; or R a4 and R a5 and N atoms form a 3- to 8-membered heterocyclic ring, and the ring may contain one or more Heteroatom selected from N, O or S;
R a3选自C 1-6烷基、C 1-6烷氧基或者C 6-12芳基; R a3 is selected from C 1-6 alkyl, C 1-6 alkoxy or C 6-12 aryl;
R a4、R a5选自H或者C 1-6烷基; R a4 and R a5 are selected from H or C 1-6 alkyl;
n、p各自独立地选自0、1、2或者3。n and p are each independently selected from 0, 1, 2 or 3.
本申请的一个或多个实施方式提供了化合物,或者其立体异构体、溶剂化物、前药、代谢产物、氘代物、药学上可接受的盐或共晶,其中该化合物选自通式(I-A)所示的化合物:One or more embodiments of the application provide a compound, or its stereoisomer, solvate, prodrug, metabolite, deuterated product, pharmaceutically acceptable salt or co-crystal, wherein the compound is selected from the general formula ( IA) The compound shown:
其中:among them:
R 0、R 1各自独立地选自卤素或者C 1-6烷基,所述的C 1-6烷基任选进一步被1-3个选自D或者卤素的取代基所取代; R 0 and R 1 are each independently selected from halogen or C 1-6 alkyl, and said C 1-6 alkyl is optionally further substituted with 1-3 substituents selected from D or halogen;
或者两个R 0与其相连的原子形成3至8元环,所述的3至8元环任选地包含1至3个选自N、O或者S的杂原子,所述的3至8元环任选进一步被1个或者多个选自-OH、羧基、卤素、氰基、=O、C 1-6烷基或者氨基的取代基所取代; Or two R 0 and the atoms to which they are connected form a 3- to 8-membered ring, and the 3- to 8-membered ring optionally contains 1 to 3 heteroatoms selected from N, O or S, and the 3- to 8-membered ring The ring is optionally further substituted with one or more substituents selected from -OH, carboxy, halogen, cyano, =0, C 1-6 alkyl or amino;
R 3选自C 1-6烷基、C 3-12碳环基、C 3杂环基、C 4-12杂环基、-C 1-6亚烷基-C 3-12碳环基、-C 1-6亚烷基-C 3杂环基、-C 1-6亚烷基-C 4-12杂环基、C 6-12螺环化合物或者C 6-12杂螺环化合物,所述的C 3杂环基和C 4-12杂环基包含1至3个选自N、O或者S的杂原子,所述的C 1-6烷基、C 3-12碳环基、C 3杂环基、C 4-12杂环基、C 1-6亚烷基任选进一步被1个或者多个选自-OH、-OR a1、羧基、卤素、氰基、=O、C 1-6烷基、C 1-6杂烷基、C 2-6烯基、C 2-6炔基、-NR a1R a2、-C(=O)OC 1-6烷基、-C(=O)NR a1R a2、C 3-12环烷基、C 3杂环烷基、C 4-12杂环烷基、C 6-12芳基或者C 5-12杂芳基的取代基所取代;且所述取代基中的所述的C 1-6烷基、C 1-6杂烷基、C 2-6烯基或者C 2-6炔基任选进一步被1个或者多个选自-OH、羧基、氰基、卤素、-O-R a1、-NR a1R a2、C 3-12环烷基、C 3杂环烷基、C 4-12杂环烷基、C 6-12芳基或者C 5-12杂芳基的取代基所取代; R 3 is selected from C 1-6 alkyl, C 3-12 carbocyclic group, C 3 heterocyclic group, C 4-12 heterocyclic group, -C 1-6 alkylene-C 3-12 carbocyclic group, -C 1-6 alkylene-C 3 heterocyclic group, -C 1-6 alkylene-C 4-12 heterocyclic group, C 6-12 spiro compound or C 6-12 heterospiro compound, so The C 3 heterocyclic group and C 4-12 heterocyclic group contain 1 to 3 heteroatoms selected from N, O or S, the C 1-6 alkyl group, C 3-12 carbocyclic group, C 3 heterocyclic group, C 4-12 heterocyclic group, C 1-6 alkylene group may be further selected by one or more selected from -OH, -OR a1 , carboxy, halogen, cyano, =O, C 1 -6 alkyl, C 1-6 heteroalkyl, C 2-6 alkenyl, C 2-6 alkynyl, -NR a1 R a2 , -C(=O)OC 1-6 alkyl, -C(= O) NR a1 R a2 , C 3-12 cycloalkyl, C 3 heterocycloalkyl, C 4-12 heterocycloalkyl, C 6-12 aryl or C 5-12 heteroaryl substituent substituted And the C 1-6 alkyl group, C 1-6 heteroalkyl group, C 2-6 alkenyl group or C 2-6 alkynyl group in the substituent is optionally further selected from one or more -OH, carboxyl, cyano, halogen, -OR a1 , -NR a1 R a2 , C 3-12 cycloalkyl, C 3 heterocycloalkyl, C 4-12 heterocycloalkyl, C 6-12 aryl Or substituted by a substituent of a C 5-12 heteroaryl group;
R 4选自H、C1-6烷基、C3-12环烷基,所述的C 1-6烷基和C 3-8环烷基任选进一步被1-3个选自D或者卤素的取代基所取代; R 4 is selected from H, C1-6 alkyl, C3-12 cycloalkyl, said C 1-6 alkyl and C 3-8 cycloalkyl are optionally further selected from 1-3 selected from D or halogen Substituent substituted;
R a1、R a2各自独立地选自H、C 1-6烷基、-C(=O)R a3或者-C(=O)NR a4R a5,其中所述的C 1-6烷基任选进一步被1个或者多个选自OH、卤素、C 1-6烷基、C 1-6烷氧基、C 6-12芳基、C 5-12杂芳基、C 3-12环烷基、C 3杂环烷基或者C 4-12杂环烷基的取代基所取代;或者R a4与R a5及N原子形成3至8元杂环,所述的3至8元杂环包含1个至3个选自N、O或者S的杂原子; R a1 and R a2 are each independently selected from H, C 1-6 alkyl, -C(=O)R a3 or -C(=O)NR a4 R a5 , wherein the C 1-6 alkyl is any Optionally, one or more selected from OH, halogen, C 1-6 alkyl, C 1-6 alkoxy, C 6-12 aryl, C 5-12 heteroaryl, C 3-12 cycloalkane Group, C 3 heterocycloalkyl or C 4-12 heterocycloalkyl substituent; or Ra4, Ra5 and N atom form a 3- to 8-membered heterocyclic ring, and the 3- to 8-membered heterocyclic ring includes 1 to 3 heteroatoms selected from N, O or S;
R a3选自C 1-6烷基、C 1-6烷氧基或者C 6-12芳基; R a3 is selected from C 1-6 alkyl, C 1-6 alkoxy or C 6-12 aryl;
R a4、R a5各自独立地选自H或者C 1-6烷基; R a4 and R a5 are each independently selected from H or C 1-6 alkyl;
n为0、1、2或者3;n is 0, 1, 2 or 3;
p为0、1、2或者3。p is 0, 1, 2, or 3.
本申请的一个或多个实施方式提供了化合物,或者其立体异构体、溶剂化物、前药、代谢产物、氘代物、药学上可接受的盐或共晶,其中该化合物选自通式(II)所示的化合物:One or more embodiments of the application provide a compound, or its stereoisomer, solvate, prodrug, metabolite, deuterated product, pharmaceutically acceptable salt or co-crystal, wherein the compound is selected from the general formula ( II) The compound shown:
其中:among them:
R 0、R 1选自卤素或者C 1-6烷基,所述的烷基任选进一步被1-3个选自D或者卤素的取代基所取代; R 0 and R 1 are selected from halogen or C 1-6 alkyl, and the alkyl is optionally further substituted with 1-3 substituents selected from D or halogen;
或者两个R 0与其相连的原子可以形成3至8元环,所述的环可含有1至3个选自N、O或者S的杂原子,所述的环任选进一步被1个或者多个选自-OH、羧基、卤素、氰基、=O、C 1-6烷基或者氨基的取代基所取代; Or two R 0 and the atoms to which they are connected can form a 3- to 8-membered ring, and the ring may contain 1 to 3 heteroatoms selected from N, O or S, and the ring is optionally further divided by 1 or more. Substituted by a substituent selected from -OH, carboxyl, halogen, cyano, =O, C 1-6 alkyl or amino;
R 3选自C 1-6烷基、C 3-12碳环基、C 3杂环基、C 4-12杂环基、-C 1-6亚烷基-C 3-12碳环基、-C 1-6亚烷基-C 3杂环基或者-C 1-6亚烷基-C 4-12杂环基,所述的杂环基可以含有1至3个选自N、O或者S的杂原子,所述的烷基、碳环基、杂环基、亚烷基任选进一步被1个或者多个选自-OH、羧基、卤素、氰基、=O、C 1-6烷基、C 1-6杂烷基、C 2-6烯基、C 2-6炔基、-NR a1R a2、-C(=O)OC 1-6烷基、-C(=O)NR a1R a2、C 3-12环烷基、C 3杂环烷基、C 4-12杂环烷基、C 6-12芳基或者C 5-12杂芳基的取代基所取代;且所述的烷基、杂烷基、烯基或者炔基任选进一步被1个或者多个选自-OH、羧基、氰基、卤素、-O-R a1、-NR a1R a2、C 3-12环烷基、C 3杂环烷基、C 4-12杂环烷基、C 6-12芳基或者C 5-12杂芳基的取代基所取代; R 3 is selected from C 1-6 alkyl, C 3-12 carbocyclic group, C 3 heterocyclic group, C 4-12 heterocyclic group, -C 1-6 alkylene-C 3-12 carbocyclic group, -C 1-6 alkylene-C 3 heterocyclic group or -C 1-6 alkylene-C 4-12 heterocyclic group, said heterocyclic group may contain 1 to 3 selected from N, O or S heteroatom, the alkyl group, carbocyclic group, heterocyclic group, alkylene group may be further selected by one or more selected from -OH, carboxyl, halogen, cyano, =0, C 1-6 Alkyl, C 1-6 heteroalkyl, C 2-6 alkenyl, C 2-6 alkynyl, -NR a1 R a2 , -C(=O)OC 1-6 alkyl, -C(=O) NR a1 R a2 , C 3-12 cycloalkyl, C 3 heterocycloalkyl, C 4-12 heterocycloalkyl, C 6-12 aryl or C 5-12 heteroaryl substituents are substituted; and The alkyl, heteroalkyl, alkenyl or alkynyl group is optionally further selected by one or more selected from -OH, carboxyl, cyano, halogen, -OR a1 , -NR a1 R a2 , C 3-12 Cycloalkyl, C 3 heterocycloalkyl, C 4-12 heterocycloalkyl, C 6-12 aryl or C 5-12 heteroaryl substituents;
R a1、R a2选自H、C 1-6烷基、-C(=O)R a3或者-C(=O)NR a4R a5,其中所述的C 1-6烷基任选进一步被1个或者多个选自OH、卤素、C 1-6烷基、C 1-6烷氧基、C 6-12芳基、C 5-12杂芳基、C 3-12环烷基、C 3杂环烷基或者C 4-12杂环烷基的取代基所取代;或者R a4与R a5 及N原子形成一个3至8元杂环,所述的环可以含有1个或者多个选自N、O或者S的杂原子; R a1 and R a2 are selected from H, C 1-6 alkyl, -C(=O)R a3 or -C(=O)NR a4 R a5 , wherein the C 1-6 alkyl is optionally further One or more selected from OH, halogen, C 1-6 alkyl, C 1-6 alkoxy, C 6-12 aryl, C 5-12 heteroaryl, C 3-12 cycloalkyl, C 3 Heterocycloalkyl or C 4-12 heterocycloalkyl substituents; or R a4 and R a5 and N atoms form a 3- to 8-membered heterocyclic ring, the ring may contain one or more options Heteroatoms from N, O or S;
R a3选自C 1-6烷基、C 1-6烷氧基或者C 6-12芳基; R a3 is selected from C 1-6 alkyl, C 1-6 alkoxy or C 6-12 aryl;
R a4、R a5选自H或者C 1-6烷基; R a4 and R a5 are selected from H or C 1-6 alkyl;
n、p各自独立地选自0、1、2或者3。n and p are each independently selected from 0, 1, 2 or 3.
本申请的一个或多个实施方式提供了化合物,或者其立体异构体、溶剂化物、前药、代谢产物、氘代物、药学上可接受的盐或共晶,其中该化合物选自通式(I’)、(I-A)、(I)或者(II)所示的化合物,其中:One or more embodiments of the application provide a compound, or its stereoisomer, solvate, prodrug, metabolite, deuterated product, pharmaceutically acceptable salt or co-crystal, wherein the compound is selected from the general formula ( I'), (IA), (I) or (II), wherein:
R 0选自甲基; R 0 is selected from methyl;
R 1选自卤素或者C 1-4烷基,所述的烷基任选进一步被1-3个选自D或者卤素的取代基所取代; R 1 is selected from halogen or C 1-4 alkyl, and the alkyl is optionally further substituted with 1-3 substituents selected from D or halogen;
R 3选自C 1-6烷基、C 3-6碳环基、C 4-8杂环基、-C 1-2亚烷基-C 3-8碳环基或者-C 1-2亚烷基-C 4-8杂环基,所述的杂环基可以含有1至3个选自N或者O的杂原子,所述的烷基、碳环基、杂环基、亚烷基任选进一步被1个或者多个选自-OH、卤素、氰基、=OC 1-4烷氧基或者C 1-4烷基的取代基所取代;且所述的取代基中的烷基任选进一步被1个或者多个选自-OH、羧基、氰基或者卤素的取代基所取代; R 3 is selected from C 1-6 alkyl, C 3-6 carbocyclic group, C 4-8 heterocyclic group, -C 1-2 alkylene-C 3-8 carbocyclic group or -C 1-2 alkylene Alkyl-C 4-8 heterocyclic group, the heterocyclic group may contain 1 to 3 heteroatoms selected from N or O, and the alkyl group, carbocyclic group, heterocyclic group, and alkylene group may be any Optionally further substituted by one or more substituents selected from -OH, halogen, cyano, =OC 1-4 alkoxy or C 1-4 alkyl; and the alkyl in the substituents is any Optionally further substituted by one or more substituents selected from -OH, carboxyl, cyano or halogen;
n选自0、1或者2;n is selected from 0, 1 or 2;
p选自1。p is selected from 1.
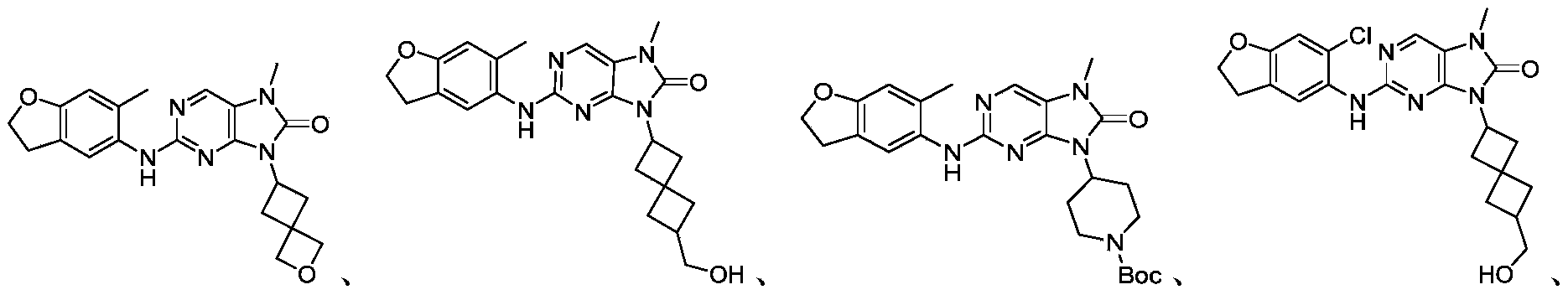
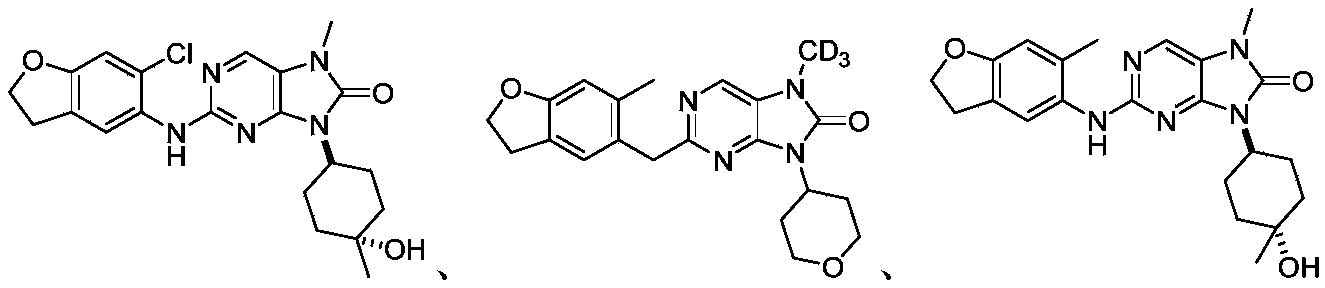
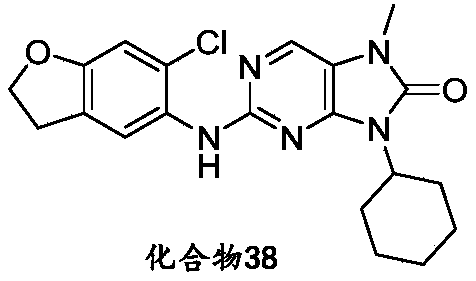
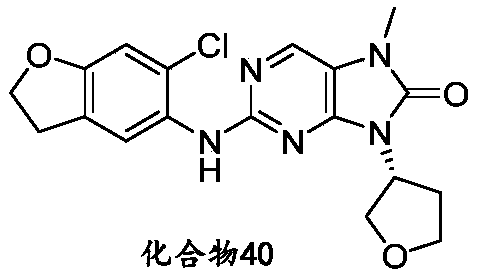
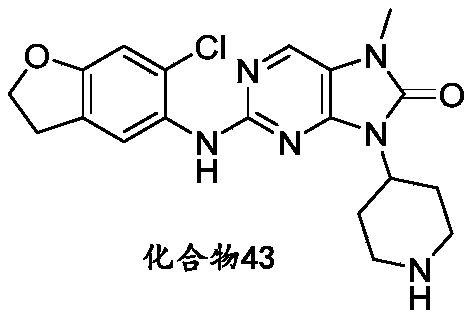
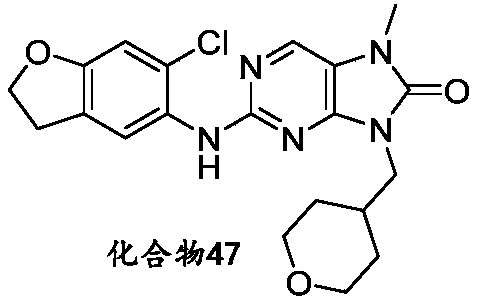
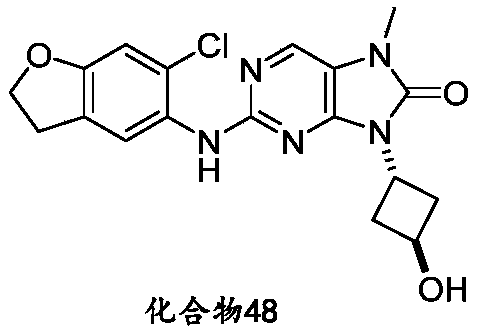
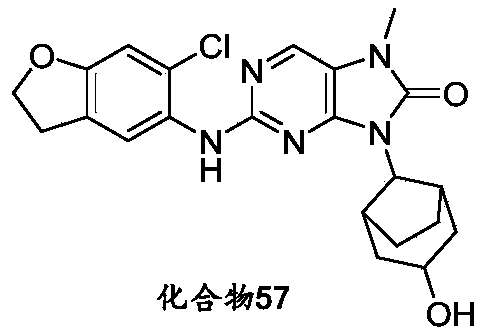
本申请的一个或多个实施方式提供了化合物,或者其立体异构体、溶剂化物、代谢产物、前药、氘代物、药学上可接受的盐或共晶,其中该化合物选自但不限于:One or more embodiments of the present application provide a compound, or its stereoisomer, solvate, metabolite, prodrug, deuterium, pharmaceutically acceptable salt or co-crystal, wherein the compound is selected from but not limited to :
或者 or
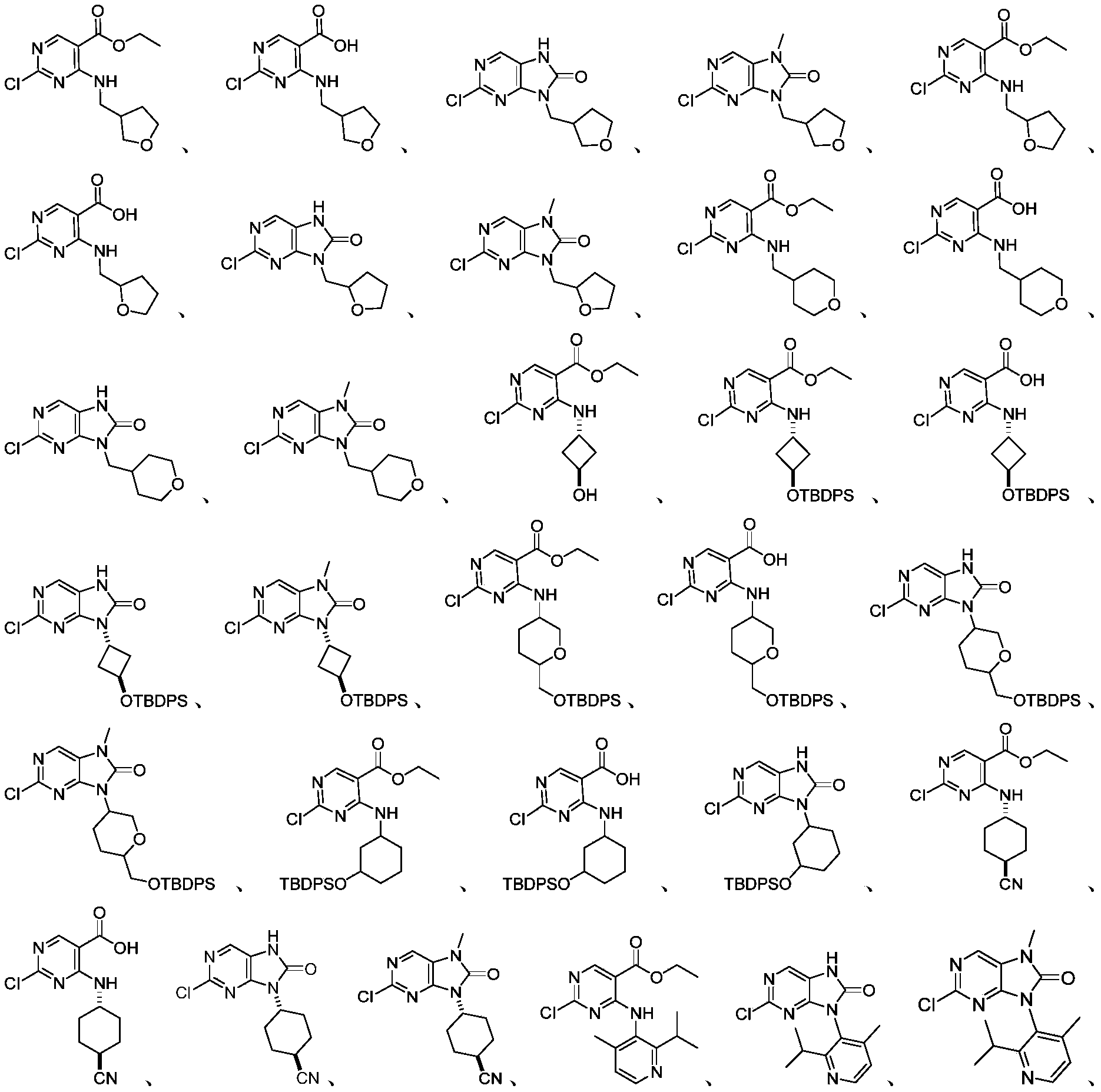
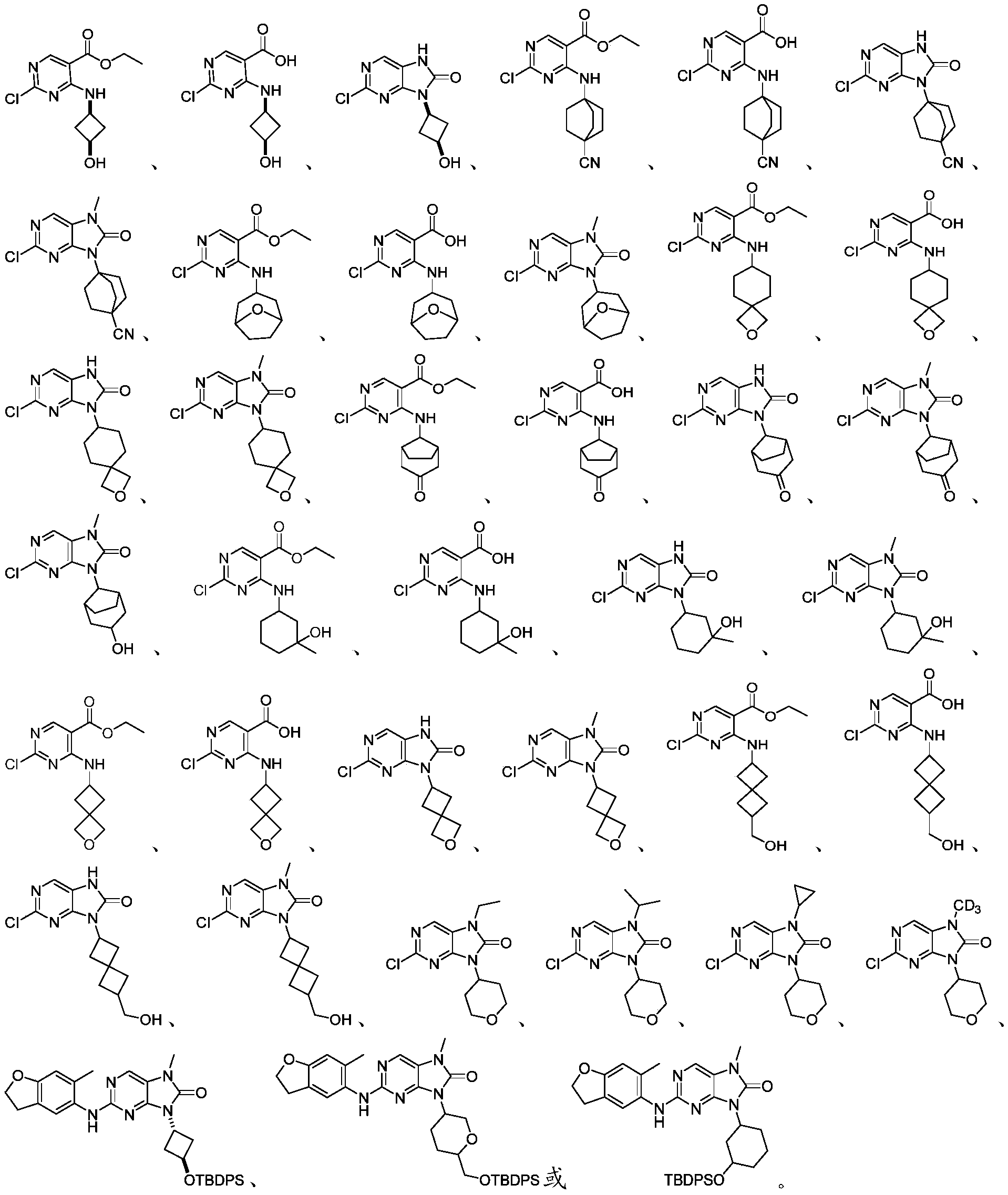
本申请的一个或多个实施方式提供了制备本发明所述化合物的中间体,所述的中间体选自但不限于:One or more embodiments of the present application provide an intermediate for preparing the compound of the present invention, and the intermediate is selected from but not limited to:
本申请的一个或多个实施方式提供了药物组合物,所述药物组合物包括:One or more embodiments of the present application provide a pharmaceutical composition, the pharmaceutical composition comprising:
(1)所述的本发明化合物或者其立体异构体、溶剂化物、前药、代谢产物、氘代物、药学上可接受的盐或共晶;(1) The compound of the present invention or its stereoisomers, solvates, prodrugs, metabolites, deuterated products, pharmaceutically acceptable salts or co-crystals;
(2)任选的一种或者多种其他活性成分;以及(2) Optional one or more other active ingredients; and
(3)药学上可接受的载体和/或赋形剂。(3) A pharmaceutically acceptable carrier and/or excipient.
本申请的一个或多个实施方式提供了本申请的药物组合物、或者化合物或者其立体异构体、溶剂化物、前药、代谢产物、氘代物、药学上可接受的盐或共晶在制备用于治疗癌症的药物中的用途。One or more embodiments of the present application provide that the pharmaceutical composition of the present application, or the compound or its stereoisomers, solvates, prodrugs, metabolites, deuterated substances, pharmaceutically acceptable salts or co-crystals are prepared in Use in medicines for the treatment of cancer.
本申请的一个或多个实施方式提供了本申请的药物组合物、或者化合物或者其立体异构体、溶剂化物、前药、代谢产物、氘代物、药学上可接受的盐或共晶在制备 DNA-PK抑制剂中的用途。One or more embodiments of the present application provide that the pharmaceutical composition of the present application, or the compound or its stereoisomers, solvates, prodrugs, metabolites, deuterated substances, pharmaceutically acceptable salts or co-crystals are prepared in Use in DNA-PK inhibitors.
本申请一个或多个实施方式提供了作为药物使用的本申请的化合物。One or more embodiments of the present application provide the compound of the present application for use as a medicine.
本申请一个或多个实施方式提供了作为DNA-PK抑制剂使用的本申请的化合物。One or more embodiments of the present application provide the compound of the present application for use as a DNA-PK inhibitor.
本申请一个或多个实施方式提供了在治疗、预防或抑制癌症的方法中使用的本申请的化合物。One or more embodiments of the present application provide a compound of the present application for use in a method of treating, preventing, or inhibiting cancer.
本申请一个或多个实施方式提供了在抑制DNA-PK的方法中使用的本申请的化合物。One or more embodiments of the present application provide a compound of the present application for use in a method of inhibiting DNA-PK.
本申请一个或多个实施方式提供了治疗、预防或抑制癌症的方法,其包括向有需要的对象施用本申请的化合物。One or more embodiments of the present application provide a method of treating, preventing or inhibiting cancer, which comprises administering the compound of the present application to a subject in need.
本申请一个或多个实施方式提供了抑制DNA-PK的方法,其包括向有需要的对象施用本申请的化合物。One or more embodiments of the present application provide a method for inhibiting DNA-PK, which includes administering the compound of the present application to a subject in need.
除非有相反的陈述,在说明书和权利要求书中使用的术语具有下述含义。Unless stated to the contrary, the terms used in the specification and claims have the following meanings.
本发明所述基团和化合物中所涉及的碳、氢、氧、硫、氮或F、Cl、Br、I均包括它们的同位素情况,及本发明所述基团和化合物中所涉及的碳、氢、氧、硫或氮任选进一步被一个或多个它们对应的同位素所替代,其中碳的同位素包括 12C、 13C和 14C,氢的同位素包括氕(H)、氘(D,又叫重氢)、氚(T,又叫超重氢),氧的同位素包括 16O、 17O和 18O,硫的同位素包括 32S、 33S、 34S和 36S,氮的同位素包括 14N和 15N,氟的同位素包括 17F和 19F,氯的同位素包括 35Cl和 37Cl,溴的同位素包括 79Br和 81Br。 The carbon, hydrogen, oxygen, sulfur, nitrogen or F, Cl, Br, and I involved in the groups and compounds of the present invention include their isotopes, and the carbon involved in the groups and compounds of the present invention , Hydrogen, oxygen, sulfur or nitrogen are optionally further replaced by one or more of their corresponding isotopes, wherein carbon isotopes include 12 C, 13 C and 14 C, and hydrogen isotopes include protium (H), deuterium (D, Also called heavy hydrogen), tritium (T, also called super heavy hydrogen), oxygen isotopes include 16 O, 17 O and 18 O, sulfur isotopes include 32 S, 33 S, 34 S and 36 S, and nitrogen isotopes include 14 N and 15 N, fluorine isotopes include 17 F and 19 F, chlorine isotopes include 35 Cl and 37 Cl, and bromine isotopes include 79 Br and 81 Br.
“烷基”是指1至20个碳原子的直链或支链饱和脂肪族烃基,优选为1至8个(例如1、2、3、4、5、6、7、8个)碳原子的烷基,更优选为1至6个碳原子的烷基,进一步优选为1至4个碳原子的烷基。非限制性实施例包括甲基、乙基、正丙基、异丙基、正丁基、仲丁基、新丁基、叔丁基、正戊基、异戊基、新戊基、正己基及其各种支链异构体;当烷基被取代基时,可以任选进一步被1个或者多个取代基所取代。"Alkyl" refers to a linear or branched saturated aliphatic hydrocarbon group of 1 to 20 carbon atoms, preferably 1 to 8 (for example, 1, 2, 3, 4, 5, 6, 7, 8) carbon atoms The alkyl group of is more preferably an alkyl group of 1 to 6 carbon atoms, and still more preferably an alkyl group of 1 to 4 carbon atoms. Non-limiting examples include methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, neobutyl, tert-butyl, n-pentyl, isopentyl, neopentyl, n-hexyl And its various branched isomers; when the alkyl group is substituted, it may be optionally further substituted with one or more substituents.
“烷氧基”是指烷基中至少1个碳原子被氧原子取代所形成的基团。非限制性实施例包括甲氧基、乙氧基、正丙氧基、异丙氧基、正丁氧基、仲丁氧基、叔丁氧基、正戊氧基、正己氧基、环丙氧基和环丁氧基。所述的烷基定义与上文所述的“烷基”定义相同。"Alkoxy" refers to a group formed by replacing at least one carbon atom in an alkyl group with an oxygen atom. Non-limiting examples include methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, sec-butoxy, tert-butoxy, n-pentoxy, n-hexoxy, cyclopropyl Oxy and cyclobutoxy. The definition of the alkyl group is the same as the definition of "alkyl" mentioned above.
“烯基”是指包含1至10个(例如1、2、3、4、5、6、7、8、9、10个)碳-碳双键,由2至20个碳原子组成的直链或者支链不饱和脂肪族烃基,优选2至12个(例如2、3、4、5、6、7、8、9、10、11、12个)碳原子的烯基,更优选2至8个碳原子的烯基,进一步优选2至6个碳原子的烯基。非限制性实施例包括乙烯基、丙烯-2-基、丁烯-2-基、丁烯-2-基、戊烯-2-基、戊烯-4-基、己烯-2-基、己烯-3基、庚烯-2-基、庚烯-3-基、庚烯-4-基、辛烯-3-基、壬烯-3-基、癸烯-4-基和十一烯-3-基。所述的烯基可以任选进一步被1个或者多个取代基所取代。"Alkenyl" refers to a straight line consisting of 1 to 10 (for example, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10) carbon-carbon double bonds consisting of 2 to 20 carbon atoms. Chain or branched unsaturated aliphatic hydrocarbon group, preferably 2 to 12 (for example, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12) carbon atoms alkenyl group, more preferably 2 to The alkenyl group of 8 carbon atoms is more preferably the alkenyl group of 2 to 6 carbon atoms. Non-limiting examples include vinyl, propen-2-yl, buten-2-yl, buten-2-yl, penten-2-yl, penten-4-yl, hexen-2-yl, Hexen-3-yl, hepten-2-yl, hepten-3-yl, hepten-4-yl, octen-3-yl, nonen-3-yl, decen-4-yl and undecenyl En-3-yl. The alkenyl group may be further substituted with one or more substituents.
“炔基”是指包含1至10个(例如1、2、3、4、5、6、7、8、9、或10个)碳-碳叁键,由2至20个碳原子组成的直链或者支链不饱和脂肪族烃基,优选2至12个(例如2、3、4、5、6、7、8、9、10、11或12个)碳原子的炔基,更优选2至8个碳原子的炔基,进一步优选2至6个碳原子的炔基。非限制性实施例包括乙炔基、丙炔-1-基、丙炔-2-基、丁炔-1-基、丁炔-2-基、丁炔-3-基、3,3-二甲基丁炔-2-基、戊炔-1-基、戊炔-2-基、己炔-1-基、1-庚炔-1-基、庚炔-3-基、庚炔-4-基、辛炔-3-基、壬炔-3-基、癸炔-4-基、十一炔-3-基、十二炔-4-基。所述的炔基可以任选进一步被一个至多个取代基所取代。"Alkynyl" refers to those containing 1 to 10 (for example, 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10) carbon-carbon triple bonds, consisting of 2 to 20 carbon atoms Straight or branched chain unsaturated aliphatic hydrocarbon group, preferably 2 to 12 (for example, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 or 12) carbon atom alkynyl group, more preferably 2 An alkynyl group having to 8 carbon atoms, and an alkynyl group having 2 to 6 carbon atoms is more preferable. Non-limiting examples include ethynyl, propyn-1-yl, propyn-2-yl, butyn-1-yl, butyn-2-yl, butyn-3-yl, 3,3-dimethyl Butyn-2-yl, pentyn-1-yl, pentyn-2-yl, hexyn-1-yl, 1-heptyn-1-yl, heptyn-3-yl, heptyn-4- Base, octyn-3-yl, nonyn-3-yl, decyn-4-yl, undecyn-3-yl, dodecyn-4-yl. The alkynyl group may be optionally further substituted with one to more substituents.
“芳基”是指是指取代的或未取代的芳香环,其可以是5至8元(例如5、6、7、8元)的单环、5至12元(例如5、6、7、8、9、10、11、12元)双环或者10至15元(例如10、11、12、13、14、15元)三环体系,其可以是桥环或者螺环,非限制性实施例包括苯基、萘基。所述的芳基可以任选进一步被1个或者多个取代基所取代。"Aryl" refers to a substituted or unsubstituted aromatic ring, which can be a 5- to 8-membered (e.g., 5, 6, 7, 8-membered) monocyclic ring, 5 to 12-membered (e.g., 5, 6, 7 , 8, 9, 10, 11, 12 membered) bicyclic or 10 to 15 membered (for example, 10, 11, 12, 13, 14, 15 membered) tricyclic ring system, which can be bridged or spiro ring, non-limiting implementation Examples include phenyl and naphthyl. The aryl group may be further substituted with one or more substituents.
“杂芳基”是指取代的或未取代的芳香环,其可以是3至8元(例如3、4、5、6、7、8元)的单环、5至12元(例如5、6、7、8、9、10、11、12元)双环或者10至15元(例如10、11、12、13、14、15元)三环体系,且包含1至6个(例如1、2、3、4、5、6个)选自N、O或S的杂原子,优选5至8元杂芳基,杂芳基的环中选择性取代的1至4个(例如1、2、3、4个)N、S可被氧化成各种氧化态。杂环基可以连接在杂原子或者碳原子上,杂芳基可以是桥环或者螺环,非限制性实施例包括环吡啶基、呋喃基、噻吩基、吡喃基、吡咯基、嘧啶基、吡嗪基、哒嗪基、咪唑基、哌啶基苯并咪唑基、苯并吡啶基、吡咯并吡啶基。杂芳基任选进一步被1个或多个取代基所取代。"Heteroaryl" refers to a substituted or unsubstituted aromatic ring, which can be 3 to 8 membered (e.g. 3, 4, 5, 6, 7, 8 membered) monocyclic, 5 to 12 membered (e.g. 5, 6, 7, 8, 9, 10, 11, 12 membered) bicyclic or 10 to 15 membered (e.g. 10, 11, 12, 13, 14, 15 membered) tricyclic ring system, and contains 1 to 6 (e.g. 1, 2, 3, 4, 5, 6) heteroatoms selected from N, O or S, preferably 5 to 8 membered heteroaryl groups, and 1 to 4 (e.g. 1, 2 , 3, 4) N and S can be oxidized into various oxidation states. The heterocyclic group can be attached to a hetero atom or a carbon atom, and the heteroaryl group can be a bridged ring or a spiro ring. Non-limiting examples include cyclopyridyl, furyl, thienyl, pyranyl, pyrrolyl, pyrimidinyl, Pyrazinyl, pyridazinyl, imidazolyl, piperidinyl benzimidazolyl, benzopyridyl, pyrrolopyridyl. The heteroaryl group is optionally further substituted with one or more substituents.
“碳环基”或“碳环”是指饱和或者不饱和的芳香环或者非芳香环。当为芳香环时,其定义与上文“芳基”的定义相同;当为非芳香环时,其可以是3至10元(例如3、4、5、6、7、8、9、10元)的单环、4至12元(例如4、5、6、7、8、9、10、11、12元)双环或者10至15元(例如10、11、12、13、14、15元)三环体系,可以是桥环或者螺环,非限制性实施例包括环丙基、环丁基、环戊基、1-环戊基-1-烯基、1-环戊基-2-烯基、1-环戊基-3-烯基、环己基、1-环己基-2-烯基、1-环己基-3-烯基、环己烯基、环己二烯基、环庚基、环辛基、环壬基、环癸基、环十一烷基、环十二烷基、 所述的“碳环基”或“碳环”任选进一步被1个或者多个取代基所取代。 "Carbocyclic group" or "carbocyclic ring" refers to a saturated or unsaturated aromatic ring or a non-aromatic ring. When it is an aromatic ring, its definition is the same as the definition of "aryl"above; when it is a non-aromatic ring, it can be 3 to 10 members (for example, 3, 4, 5, 6, 7, 8, 9, 10 Yuan), 4 to 12 yuan (e.g. 4, 5, 6, 7, 8, 9, 10, 11, 12 yuan) bicyclic ring or 10 to 15 yuan (e.g. 10, 11, 12, 13, 14, 15 Member) tricyclic ring system, which can be bridged or spiro ring, non-limiting examples include cyclopropyl, cyclobutyl, cyclopentyl, 1-cyclopentyl-1-enyl, 1-cyclopentyl-2 -Alkenyl, 1-cyclopentyl-3-alkenyl, cyclohexyl, 1-cyclohexyl-2-alkenyl, 1-cyclohexyl-3-alkenyl, cyclohexenyl, cyclohexadienyl, cyclo Heptyl, cyclooctyl, cyclononyl, cyclodecyl, cycloundecyl, cyclododecyl, The "carbocyclic group" or "carbocyclic ring" is optionally further substituted with one or more substituents.
“杂环基”或“杂环”是指饱和或不饱和的芳香性杂环或者非芳香性杂环,当为芳香性杂环时,其定义与上文“杂芳基”定义相同;当为非芳香性杂环时,其可以是3至10元(例如3、4、5、6、7、8、9、10元)的单环、4至12元(例如4、5、6、7、8、9、10、11、12元)双环或者10至15元(例如10、11、12、13、14、15元)三环体系,且包含1至4个(例如1、2、3、4个)选自N、O或S的杂原子,优选3至8元杂环基。“杂环基”或“杂环”的环中选择性取代的1至4个(例如1、2、3、4个)N、S可被氧化成各种氧化态;“杂环基”或“杂环”可以连接在杂原子或者碳原子上;“杂环基”或“杂环”可以为桥环或者螺环。“杂环基”或“杂环”的非限制性实施例包括环氧乙基、环氧丙基、氮杂环丙基、氧杂环丁基、氮杂环丁基、硫杂环丁基、1,3-二氧戊环基、1,4-二氧戊环基、1,3-二氧六环基、氮杂环庚基、氧杂环庚基、硫杂环庚基、氧氮杂卓基、二氮杂卓基、硫氮杂卓基、吡啶基、哌啶基、高哌啶基、呋喃基、噻吩基、吡喃基、N-烷基吡咯基、嘧啶基、吡嗪基、哒嗪基、哌嗪基、高哌嗪基、咪唑基、哌啶基、吗啉基、硫代吗啉基、噻噁烷基、1,3-二噻烷基、二氢呋喃基、二噻戊环基、四氢呋喃基、四氢噻吩基、四氢吡喃基、四氢噻喃基、四氢吡咯基、四氢咪唑基、四氢噻唑基、四氢吡喃基、苯并咪唑基、苯并吡啶基、吡咯并吡啶基、苯并二氢呋喃基、2-吡咯啉基、3-吡咯啉基、二氢吲哚基、2H-吡喃基、4H-吡喃基、二氧杂环己基、1,3-二氧戊基、吡唑啉基、二噻烷基、二噻茂烷基、二氢噻吩基、吡唑烷基、咪唑啉基、咪唑烷基、1,2,3,4-四氢异喹啉基、3-氮杂双环[3.1.0]己基、3-氮杂双环[4.1.0]庚基、氮杂双环[2.2.2]己基、3H-吲哚基喹嗪基、N-吡啶基尿素、1,1-二氧硫代吗啉基、氮杂二环[3.2.1]辛烷基、氮杂二环[5.2.0]壬烷基、氧杂三环[5.3.1.1]十二烷基、氮杂金刚烷基和氧杂螺[3.3]庚烷基。所述的“杂环基”或“杂环”可以任选进一步被1个或者多个取代基所取代。"Heterocyclic group" or "heterocyclic ring" refers to a saturated or unsaturated aromatic heterocyclic ring or non-aromatic heterocyclic ring. When it is an aromatic heterocyclic ring, its definition is the same as the definition of "heteroaryl" above; when When it is a non-aromatic heterocyclic ring, it can be a 3- to 10-membered (e.g. 3, 4, 5, 6, 7, 8, 9, 10-membered) monocyclic ring, 4 to 12-membered (e.g. 4, 5, 6, 7, 8, 9, 10, 11, 12 membered) bicyclic or 10 to 15 membered (e.g. 10, 11, 12, 13, 14, 15 membered) tricyclic ring system, and contains 1 to 4 (e.g. 1, 2, 3, 4) heteroatoms selected from N, O or S, preferably 3 to 8 membered heterocyclic groups. One to four (for example, 1, 2, 3, 4) N and S optionally substituted in the "heterocyclic group" or "heterocyclic ring" can be oxidized to various oxidation states; "heterocyclic group" or "Heterocycle" can be attached to a heteroatom or carbon atom; "heterocyclic group" or "heterocycle" can be a bridged ring or a spiro ring. Non-limiting examples of "heterocyclic group" or "heterocyclic ring" include oxirane, glycidyl, aziridinyl, oxetanyl, azetidinyl, thietanyl , 1,3-dioxolane, 1,4-dioxolane, 1,3-dioxanyl, azepanyl, oxepanyl, thiepanyl, oxygen Azepine, diazepine, thiazepine, pyridinyl, piperidinyl, homopiperidinyl, furyl, thienyl, pyranyl, N-alkylpyrrolyl, pyrimidinyl, pyridine Azinyl, pyridazinyl, piperazinyl, homopiperazinyl, imidazolyl, piperidinyl, morpholinyl, thiomorpholinyl, thiazinyl, 1,3-dithiazinyl, dihydrofuran Group, dithiopentyl, tetrahydrofuranyl, tetrahydrothienyl, tetrahydropyranyl, tetrahydrothiopyranyl, tetrahydropyrrolyl, tetrahydroimidazolyl, tetrahydrothiazolyl, tetrahydropyranyl, benzene Bisimidazolyl, benzopyridyl, pyrrolopyridyl, chromanyl, 2-pyrrolinyl, 3-pyrrolinyl, indolinyl, 2H-pyranyl, 4H-pyranyl , Dioxanyl, 1,3-dioxolyl, pyrazolinyl, dithianyl, dithiazolinyl, dihydrothienyl, pyrazolidinyl, imidazolinyl, imidazolinyl, 1,2,3,4-Tetrahydroisoquinolinyl, 3-azabicyclo[3.1.0]hexyl, 3-azabicyclo[4.1.0]heptyl, azabicyclo[2.2.2]hexyl, 3H-indolylquinazinyl, N-pyridylurea, 1,1-dioxothiomorpholinyl, azabicyclo[3.2.1]octyl, azabicyclo[5.2.0]non Alkyl, oxatricyclo[5.3.1.1]dodecyl, azaadamantyl and oxaspiro[3.3]heptyl. The "heterocyclic group" or "heterocyclic ring" may be optionally further substituted with one or more substituents.
“环烷基”是指饱和的环烃基,其环可以为3至10元(例如3、4、5、6、7、8、9、10元)的单环、4至12元(例如4、5、6、7、8、9、10、11、12元)双环或者10至20元(例如10、11、12、13、14、15、16、17、18、19、20元)多环体系,环碳原子优选3至10个碳原子,进一步优选3至8个碳原子。“环烷基”非限制性实施例包括环丙基、环丁基、环戊基、环己基、环庚基、环辛基、环丙烯基、环丁烯基、环戊烯基、环己 烯基、环庚烯基、1,5-环辛二烯基、1,4-环己二烯基和环庚三烯基等。当环烷基被取代时,可以任选进一步被1个或者多个取代基所取代。"Cycloalkyl" refers to a saturated cyclic hydrocarbon group whose ring can be 3 to 10 membered (e.g. 3, 4, 5, 6, 7, 8, 9, 10 membered) monocyclic, 4 to 12 membered (e.g. 4 , 5, 6, 7, 8, 9, 10, 11, 12 yuan) bicyclic or 10 to 20 yuan (e.g. 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20 yuan) more In the ring system, the ring carbon atoms preferably have 3 to 10 carbon atoms, more preferably 3 to 8 carbon atoms. Non-limiting examples of "cycloalkyl" include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclopropenyl, cyclobutenyl, cyclopentenyl, cyclohexyl Alkenyl, cycloheptenyl, 1,5-cyclooctadienyl, 1,4-cyclohexadienyl and cycloheptatrienyl, etc. When the cycloalkyl group is substituted, it may be further substituted with one or more substituents.
“杂环烷基”是指取代的或未取代的饱和非芳香环基,其可以是3至8元(例如3、4、5、6、7、8元)的单环、4至12元(例如4、5、6、7、8、9、10、11、12元)双环或者10至15元(例如10、11、12、13、14、15元)三环体系,且包含1、2或3个选自N、O或S的杂原子,优选3至8元杂环基。“杂环烷基”的环中选择性取代的N、S可被氧化成各种氧化态;“杂环烷基”可以连接在杂原子或者碳原子上;“杂环烷基”可以为桥环或者螺环。“杂环烷基”非限制性实施例包括环氧乙基、氮杂环丙基、氧杂环丁基、氮杂环丁基、1,3-二氧戊环基、1,4-二氧戊环基、1,3-二氧六环基、氮杂环庚基、哌啶基、哌叮基、吗啉基、硫代吗啉基、1,3-二噻烷基、四氢呋喃基、四氢吡咯基、四氢咪唑基、四氢噻唑基、四氢吡喃基、氮杂二环[3.2.1]辛烷基、氮杂二环[5.2.0]壬烷基、氧杂三环[5.3.1.1]十二烷基、氮杂金刚烷基和氧杂螺[3.3]庚烷基。"Heterocycloalkyl" refers to a substituted or unsubstituted saturated non-aromatic ring group, which can be 3 to 8 membered (for example, 3, 4, 5, 6, 7, 8 membered) monocyclic, 4 to 12 membered (E.g. 4, 5, 6, 7, 8, 9, 10, 11, 12-membered) bicyclic or 10 to 15-membered (e.g. 10, 11, 12, 13, 14, 15-membered) tricyclic ring system, including 1, 2 or 3 heteroatoms selected from N, O or S, preferably 3 to 8 membered heterocyclic group. The selectively substituted N and S in the "heterocycloalkyl" ring can be oxidized to various oxidation states; the "heterocycloalkyl" can be connected to a heteroatom or a carbon atom; the "heterocycloalkyl" can be a bridge Ring or spiro ring. Non-limiting examples of "heterocycloalkyl" include oxirane ethyl, aziridinyl, oxetanyl, azetidinyl, 1,3-dioxolane, 1,4-dioxolane, Oxolane, 1,3-dioxanyl, azepanyl, piperidinyl, piperidinyl, morpholinyl, thiomorpholinyl, 1,3-dithianyl, tetrahydrofuranyl , Tetrahydropyrrolyl, tetrahydroimidazolyl, tetrahydrothiazolyl, tetrahydropyranyl, azabicyclo[3.2.1]octyl, azabicyclo[5.2.0]nonyl, oxa Tricyclic[5.3.1.1]dodecyl, azaadamantyl and oxaspiro[3.3]heptyl.
当上文所述的“烷基”、“烷氧基”、“烯基”、“炔基”、“芳基”、“杂芳基”、“碳环基”、“碳环”、“杂环基”、“杂环”、“环烷基”、“杂环烷基”或者“杂环基”被取代时,可以任选进一步被0、1、2、3、4、5、6、7、8、9或者10个选自F、Cl、Br、I、羟基、巯基、硝基、氰基、氨基、C 1-6烷基氨基、=O、C 1-6烷基、C 1-6烷氧基、C 2-6烯基、C 2-6炔基、-NR q4R q5、=NR q6、-C(=O)OC 1-6烷基、-OC(=O)C 1-6烷基、-C(=O)NR q4R q5、C 3-12环烷基、C 3杂环烷基、C 4-12杂环烷基、C 6-12芳基、C 5-12杂芳基、-C(=O)OC 6-12芳基、-OC(=O)C 6-12芳基、-OC(=O)C 5-12杂芳基、-C(=O)OC 5-12杂芳基、-OC(=O)C 3杂环烷基、-OC(=O)C 4-12杂环烷基、-C(=O)OC 3杂环烷基、-C(=O)OC 4-12杂环烷基、-OC(=O)C 3-12环烷基、-C(=O)OC 3-12环烷基、-NHC(=O)C 3杂环烷基、-NHC(=O)C 4-12杂环烷基、-NHC(=O)C 6-12芳基、-NHC(=O)C 5-12杂芳基、-NHC(=O)C 3-12环烷基、-NHC(=O)C 2-6烯基或者-NHC(=O)C 2-6炔基的取代基所取代,且其中所述的取代基C 1-6烷基、C 1-6烷氧基、C 2-6烯基、C 2-6炔基、C 3-12环烷基、C 3杂环烷基、C 4-12杂环烷基、C 6-12芳基、C 5-12杂芳基、-NHC(=O)C 6-12芳基、-NHC(=O)C 5-12杂芳基、-NHC(=O)C 3杂环烷基、-NHC(=O)C 4-12杂环烷基或者-NHC(=O)C 3-12环烷基任选进一步被1至3个选自OH、F、Cl、Br、I、C 1-6烷基、C 1-6烷氧基、-NR q4R q5或者=O的取代基所取代;R q1选自C 1-6烷基、C 1-6烷氧基或者C 6-12芳基;R q2、R q3选自H或者C 1-6烷基;其中,R q4、R q5选自H、C 1-6烷基、-NH(C=NR q1)NR q2R q3、-S(=O) 2NR q2R q3、-C(=O)R q1或者-C(=O)NR q2R q3,其中所述的C 1-6烷基任选进一步被1个或者多个选自OH、F、Cl、Br、I、C 1-6烷基、C 1-6烷氧基、C 6-12芳基、C 5-12杂芳基、C 3-12环烷基、C 3杂环烷基或者C 4-12杂环烷基的取代基所取代;或者R q4与R q5及N原子形成一个3至8元杂环,所述的杂环可以包含1个或者多个选自N、O或者S的杂原子。 When the above-mentioned "alkyl", "alkoxy", "alkenyl", "alkynyl", "aryl", "heteroaryl", "carbocyclyl", "carbocyclic", " When "heterocyclyl", "heterocycle", "cycloalkyl", "heterocycloalkyl" or "heterocyclyl" are substituted, they may optionally be further substituted with 0, 1, 2, 3, 4, 5, 6 , 7, 8, 9 or 10 selected from F, Cl, Br, I, hydroxyl, mercapto, nitro, cyano, amino, C 1-6 alkylamino, =0, C 1-6 alkyl, C 1-6 alkoxy, C 2-6 alkenyl, C 2-6 alkynyl, -NR q4 R q5 , =NR q6 , -C(=O)OC 1-6 alkyl, -OC(=O) C 1-6 alkyl, -C(=O)NR q4 R q5 , C 3-12 cycloalkyl, C 3 heterocycloalkyl, C 4-12 heterocycloalkyl, C 6-12 aryl, C 5-12 heteroaryl, -C(=O)OC 6-12 aryl, -OC(=O)C 6-12 aryl, -OC(=O)C 5-12 heteroaryl, -C( =O)OC 5-12 heteroaryl, -OC(=O)C 3 heterocycloalkyl, -OC(=O)C 4-12 heterocycloalkyl, -C(=O)OC 3 heterocycloalkane Group, -C(=O)OC 4-12 heterocycloalkyl, -OC(=O)C 3-12 cycloalkyl, -C(=O)OC 3-12 cycloalkyl, -NHC(=O )C 3 heterocycloalkyl, -NHC(=O)C 4-12 heterocycloalkyl, -NHC(=O)C 6-12 aryl, -NHC(=O)C 5-12 heteroaryl, -NHC(=O)C 3-12 cycloalkyl, -NHC(=O)C 2-6 alkenyl or -NHC(=O)C 2-6 alkynyl substituents, and the said Substituents C 1-6 alkyl, C 1-6 alkoxy, C 2-6 alkenyl, C 2-6 alkynyl, C 3-12 cycloalkyl, C 3 heterocycloalkyl, C 4-12 Heterocycloalkyl, C 6-12 aryl, C 5-12 heteroaryl, -NHC(=O)C 6-12 aryl, -NHC(=O)C 5-12 heteroaryl, -NHC( =0)C 3 heterocycloalkyl, -NHC(=O)C 4-12 heterocycloalkyl or -NHC(=O)C 3-12 cycloalkyl is optionally further selected from 1 to 3 OH, F, Cl, Br, I, C 1-6 alkyl, C 1-6 alkoxy, -NR q4 R q5 or =0 substituents; R q1 is selected from C 1-6 alkyl, C 1 -6 alkoxy or C 6-12 aryl; R q2 and R q3 are selected from H or C 1-6 alkyl; wherein R q4 and R q5 are selected from H, C 1-6 alkyl, -NH( C=NR q1 )NR q2 R q3 , -S(=O) 2 NR q2 R q3 , -C(=O)R q1 or -C(=O)NR q2 R q3 , Wherein, the C 1-6 alkyl group is optionally further substituted by one or more selected from OH, F, Cl, Br, I, C 1-6 alkyl, C 1-6 alkoxy, C 6-12 Substituted by aryl, C 5-12 heteroaryl, C 3-12 cycloalkyl, C 3 heterocycloalkyl or C 4-12 heterocycloalkyl substituent; or R q4 is formed with R q5 and N atom A 3- to 8-membered heterocyclic ring, said heterocyclic ring may contain one or more heteroatoms selected from N, O or S.
“药学上可接受的盐”或者“其药学上可接受的盐”是指本发明化合物保持游离酸或者游离碱的生物有效性和特性,且所述的游离酸通过与无毒的无机碱或者有机碱,所述的游离碱通过与无毒的无机酸或者有机酸反应获得的盐。"Pharmaceutically acceptable salt" or "pharmaceutically acceptable salt thereof" means that the compound of the present invention maintains the biological effectiveness and characteristics of the free acid or free base, and the free acid is combined with a non-toxic inorganic base or An organic base is a salt obtained by reacting the free base with a non-toxic inorganic acid or organic acid.
“药物组合物”是指一种或多种本发明所述化合物、其药学上可接受的盐或前药和其它化学组分形成的混合物,其中,“其它化学组分”是指药学上可接受的载体、赋形剂和/或一种或多种其它治疗剂。"Pharmaceutical composition" refers to a mixture of one or more compounds of the present invention, their pharmaceutically acceptable salts or prodrugs, and other chemical components, where "other chemical components" refer to pharmaceutically acceptable compounds. Accepted carriers, excipients and/or one or more other therapeutic agents.
“载体”是指不会对生物体产生明显刺激且不会消除所给予化合物的生物活性和特性的材料。"Carrier" refers to a material that does not cause significant irritation to the organism and does not eliminate the biological activity and characteristics of the administered compound.
“赋形剂”是指加入到药物组合物中以促进化合物给药的惰性物质。非限制性实施例包括碳酸钙、磷酸钙、糖、淀粉、纤维素衍生物(包括微晶纤维素)、明胶、植物油、聚乙二醇类、稀释剂、成粒剂、润滑剂、粘合剂和崩解剂。"Excipient" refers to an inert substance added to a pharmaceutical composition to facilitate the administration of a compound. Non-limiting examples include calcium carbonate, calcium phosphate, sugar, starch, cellulose derivatives (including microcrystalline cellulose), gelatin, vegetable oils, polyethylene glycols, diluents, granulating agents, lubricants, adhesives Agent and disintegrant.
“前药”是指可经体内代谢转化为具有生物活性的本发明化合物。本发明的前药通 过修饰本发明化合物中的氨基或者羧基来制备,该修饰可以通过常规的操作或者在体内被除去,而得到母体化合物。当本发明的前药被施予哺乳动物个体时,前药被割裂形成游离的氨基或者羧基。A "prodrug" refers to a compound of the present invention that can be converted into a biologically active compound by metabolism in the body. The prodrug of the present invention is prepared by modifying the amino or carboxyl group in the compound of the present invention, and this modification can be removed by conventional operations or in vivo to obtain the parent compound. When the prodrug of the present invention is administered to a mammalian individual, the prodrug is split to form free amino or carboxyl groups.
“共晶”是指活性药物成分(API)和共晶形成物(CCF)在氢键或其他非共价键的作用下结合而成的晶体,其中API和CCF的纯态在室温下均为固体,并且各组分间存在固定的化学计量比。共晶是一种多组分晶体,既包含两种中性固体之间形成的二元共晶,也包含中性固体与盐或溶剂化物形成的多元共晶。"Co-crystal" refers to the crystal formed by the combination of active pharmaceutical ingredient (API) and co-crystal former (CCF) under the action of hydrogen bonds or other non-covalent bonds. The pure state of API and CCF are both at room temperature. Solid, and there is a fixed stoichiometric ratio between the components. A eutectic is a multi-component crystal, which includes both a binary eutectic formed between two neutral solids and a multi-element eutectic formed between a neutral solid and a salt or solvate.
“立体异构体”是指由分子中原子在空间上排列方式不同所产生的异构体,包括顺反异构体、对映异构体和构象异构体。"Stereoisomers" refer to isomers produced by the different arrangements of atoms in a molecule in space, including cis-trans isomers, enantiomers and conformational isomers.
“任选”或“任选地”或“选择性的”或“选择性地”是指随后所述的事件或状况可以但未必发生,该描述包括其中发生该事件或状况的情况及其中未发生的情况。例如,“任选地被烷基取代的杂环基”是指该烷基可以但未必存在,该描述包括其中杂环基被烷基取代的情况,及其中杂环基未被烷基取代的情况。"Optional" or "optionally" or "selective" or "selectively" means that the event or condition described later can but does not necessarily occur, and the description includes the situation in which the event or condition occurs and its failures. What happened. For example, "heterocyclic group optionally substituted by an alkyl group" means that the alkyl group may but does not necessarily exist, and the description includes the case where the heterocyclic group is substituted by an alkyl group and the case where the heterocyclic group is not substituted by an alkyl group. Happening.
以下实施例详细说明本发明的技术方案,但本发明的保护范围包括但是不限于此。The following embodiments illustrate the technical solutions of the present invention in detail, but the protection scope of the present invention includes but is not limited thereto.
化合物的结构是通过核磁共振(NMR)或(和)质谱(MS)来确定的。NMR位移(δ)以10 -6(ppm)的单位给出。NMR的测定是用(Bruker Avance III 400和Bruker Avance 300)核磁仪,测定溶剂为氘代二甲基亚砜(DMSO-d 6),氘代氯仿(CDCl 3),氘代甲醇(CD 3OD),内标为四甲基硅烷(TMS); The structure of the compound is determined by nuclear magnetic resonance (NMR) or (and) mass spectrometry (MS). The NMR shift (δ) is given in units of 10 -6 (ppm). NMR is measured with (Bruker Avance III 400 and Bruker Avance 300) nuclear magnetic instrument, the solvent is deuterated dimethyl sulfoxide (DMSO-d 6 ), deuterated chloroform (CDCl 3 ), deuterated methanol (CD 3 OD) ), the internal standard is tetramethylsilane (TMS);
MS的测定用(Agilent 6120B(ESI)和Agilent 6120B(APCI));For MS measurement (Agilent 6120B (ESI) and Agilent 6120B (APCI));
HPLC的测定使用Agilent 1260DAD高压液相色谱仪(Zorbax SB-C18 100×4.6mm,3.5μM);HPLC determination uses Agilent 1260DAD high pressure liquid chromatograph (Zorbax SB-C18 100×4.6mm, 3.5μM);
薄层层析硅胶板使用烟台黄海HSGF254或青岛GF254硅胶板,薄层色谱法(TLC)使用的硅胶板采用的规格是0.15mm-0.20mm,薄层层析分离纯化产品采用的规格是0.4mm-0.5mm;The thin layer chromatography silica gel plate uses Yantai Huanghai HSGF254 or Qingdao GF254 silica gel plate. The size of the silica gel plate used for thin layer chromatography (TLC) is 0.15mm-0.20mm, and the size used for thin layer chromatography separation and purification products is 0.4mm. -0.5mm;
柱层析一般使用烟台黄海硅胶200-300目硅胶为载体;Column chromatography generally uses Yantai Huanghai silica gel 200-300 mesh silica gel as the carrier;
本发明的己知起始原料可以采用或按照本领域已知的方法来合成,或可购买于泰坦科技、安耐吉化学、上海德默、成都科龙化工、韶远化学科技、百灵威科技等公司;The known starting materials of the present invention can be synthesized by or according to methods known in the art, or can be purchased from Titan Technology, Anaiji Chemical, Shanghai Demo, Chengdu Kelon Chemical, Shaoyuan Chemical Technology, Bailingwei Technology, etc. the company;
氮气氛是指反应瓶连接约1L容积的氮气气球;Nitrogen atmosphere means that the reaction flask is connected to a nitrogen balloon with a volume of about 1L;
氢气氛是指反应瓶连接约1L容积的氢气气球;The hydrogen atmosphere refers to the reaction flask connected to a hydrogen balloon with a volume of about 1L;
氢化反应通常抽真空,充入氢气,反复操作3次;The hydrogenation reaction is usually evacuated and filled with hydrogen, and the operation is repeated 3 times;
实施例中无特殊说明,反应在氮气氛下进行;There are no special instructions in the examples, and the reaction is carried out under a nitrogen atmosphere;
实施例中无特殊说明,溶液是指水溶液;There is no special description in the examples, and the solution refers to an aqueous solution;
实施例中无特殊说明,反应的温度为室温,室温最适宜的反应温度,为20℃-30℃;No special instructions in the examples, the reaction temperature is room temperature, and the most suitable reaction temperature for room temperature is 20°C-30°C;
DCM:二氯甲烷;DCM: dichloromethane;
EA:乙酸乙酯;EA: ethyl acetate;
HCl:盐酸;HCl: hydrochloric acid;
THF:四氢呋喃;THF: Tetrahydrofuran;
DMF:N,N-二甲基甲酰胺;DMF: N,N-dimethylformamide;
PE:石油醚;PE: petroleum ether;
TLC:薄层色谱;TLC: thin layer chromatography;
SFC:超临界流体色谱法;SFC: Supercritical fluid chromatography;
NCS:N-氯代丁二酰亚胺;NCS: N-chlorosuccinimide;
Pd(dppf)Cl 2:[1,1'-双(二苯基膦)二茂铁]二氯化钯; Pd(dppf)Cl 2 : [1,1'-bis(diphenylphosphine)ferrocene]palladium dichloride;
DMSO:二甲基亚砜;DMSO: dimethyl sulfoxide;
DTT:二硫苏糖醇;DTT: Dithiothreitol;
ATP:三磷酸腺苷;ATP: Adenosine triphosphate;
DNA:脱氧核糖核苷酸;DNA: Deoxyribonucleotide;
IC50:是指DNA-PK激酶的活性受到50%抑制时化合物的浓度;IC50: refers to the concentration of the compound when the activity of DNA-PK kinase is inhibited by 50%;
X-Phos:2-二环己基磷-2,4,6-三异丙基联苯。X-Phos: 2-Dicyclohexylphosphorus-2,4,6-triisopropylbiphenyl.
实施例Example
中间体1Intermediate 1
6-甲基-2,3-二氢苯并呋喃-5-胺(中间体1)6-Methyl-2,3-dihydrobenzofuran-5-amine (Intermediate 1)
6-methyl-2,3-dihydrobenzofuran-5-amine6-methyl-2,3-dihydrobenzofuran-5-amine
第一步:first step:
1-溴-2-(2-溴乙氧基)-4-甲基苯(1B)1-Bromo-2-(2-bromoethoxy)-4-methylbenzene (1B)
1-bromo-2-(2-bromoethoxy)-4-methylbenzene1-bromo-2-(2-bromoethoxy)-4-methylbenzene
将1,2-二溴乙烷(100.4g,534.67mmol)用乙腈(100mL)混匀,然后加入2-溴-5-甲基苯酚1A(25g,133.67mmol),最后加入碳酸钾(55.42g,401.01mmol),在80℃下反应5h。反应完成后过滤,浓缩滤液,硅胶柱色谱分离提纯(纯石油醚),得到标题化合物1B(无色液体,34g,产率86.51%)。Mix 1,2-dibromoethane (100.4g, 534.67mmol) with acetonitrile (100mL), then add 2-bromo-5-methylphenol 1A (25g, 133.67mmol), and finally add potassium carbonate (55.42g) , 401.01mmol), reacted at 80°C for 5h. After the reaction was completed, it was filtered, the filtrate was concentrated, and the silica gel column chromatography (pure petroleum ether) was separated and purified to obtain the title compound 1B (colorless liquid, 34 g, yield 86.51%).
第二步:The second step:
6-甲基-2,3-二氢苯并呋喃(1C)6-Methyl-2,3-dihydrobenzofuran (1C)
6-methyl-2,3-dihydrobenzofuran6-methyl-2,3-dihydrobenzofuran
将1-溴-2-(2-溴乙氧基)-4-甲基苯1B(34g,115.65mmol)加入干燥的反应瓶,用干燥的四氢呋喃(160mL)溶解,然后在-78℃下滴加正丁基锂(55mL,138.78mmol),滴加完成后继续反应1.5h。反应完成后,向反应液中加入水(20ml)淬灭,减压除去有机溶剂,用乙酸乙酯萃取两次,合并有机相,干燥浓缩,使用硅胶柱色谱分离提纯(纯石油醚),得到标题化合物1C(无色液体,10g,产率64.43%)。Add 1-bromo-2-(2-bromoethoxy)-4-methylbenzene 1B (34g, 115.65mmol) into a dry reaction flask, dissolve it with dry tetrahydrofuran (160mL), and then drop at -78℃ Add n-butyllithium (55mL, 138.78mmol), and continue the reaction for 1.5h after the addition is complete. After the reaction is complete, add water (20ml) to the reaction solution for quenching, remove the organic solvent under reduced pressure, extract twice with ethyl acetate, combine the organic phases, dry and concentrate, and use silica gel column chromatography to separate and purify (pure petroleum ether) to obtain The title compound 1C (colorless liquid, 10 g, yield 64.43%).
第三步:third step:
6-甲基-5-硝基-2,3-二氢苯并呋喃(1D)6-Methyl-5-nitro-2,3-dihydrobenzofuran (1D)
6-methyl-5-nitro-2,3-dihydrobenzofuran6-methyl-5-nitro-2,3-dihydrobenzofuran
将6-甲基-2,3-二氢苯并呋喃1C(10g,74.53mmol)溶于醋酸(50mL)中,室温下滴入硝酸(11.8mL,178.87mmol,68%纯度),滴加完成后继续反应10min。TLC监测反应完全,将反应液倒入冰水中,用乙酸乙酯萃取三次,干燥浓缩有机相,使用硅胶柱色谱分离提纯(石油醚/乙酸乙酯=15/1),得到标题化合物1D(黄色固体,7.0g,产率52.43%)。Dissolve 6-methyl-2,3-dihydrobenzofuran 1C (10g, 74.53mmol) in acetic acid (50mL), add nitric acid (11.8mL, 178.87mmol, 68% purity) dropwise at room temperature, and the addition is complete Then continue to react for 10 minutes. TLC monitored the completion of the reaction. The reaction solution was poured into ice water, extracted three times with ethyl acetate, dried and concentrated the organic phase, separated and purified by silica gel column chromatography (petroleum ether/ethyl acetate = 15/1) to obtain the title compound 1D (yellow) Solid, 7.0 g, yield 52.43%).
第四步:the fourth step:
6-甲基-2,3-二氢苯并呋喃-5-胺(中间体1)6-Methyl-2,3-dihydrobenzofuran-5-amine (Intermediate 1)
6-methyl-2,3-dihydrobenzofuran-5-amine6-methyl-2,3-dihydrobenzofuran-5-amine
将6-甲基-5-硝基-2,3-二氢苯并呋喃1D(7.0g,39.07mmol)用110mL(乙醇/水=10/1) 混匀,加入铁粉(10.9g,195.33mmol),最后加入稀盐酸(9.8mL,2mol/L),在85℃反应2h。将反应液过滤除去铁粉,浓缩滤液,然后用饱和碳酸氢钠溶液调节pH至弱碱性,然后用乙酸乙酯萃取三次,合并有机相,干燥浓缩,使用硅胶柱色谱分离纯化(石油醚/乙酸乙酯=5/1),得标题化合物中间体1(褐色固体,4.5g,产率77.05%)。Mix 6-methyl-5-nitro-2,3-dihydrobenzofuran 1D (7.0g, 39.07mmol) with 110mL (ethanol/water=10/1) and add iron powder (10.9g, 195.33). mmol), and finally add dilute hydrochloric acid (9.8mL, 2mol/L), and react at 85°C for 2h. The reaction solution was filtered to remove iron powder, the filtrate was concentrated, and the pH was adjusted to weakly alkaline with saturated sodium bicarbonate solution, and then extracted three times with ethyl acetate. The organic phases were combined, dried and concentrated, and separated and purified by silica gel column chromatography (petroleum ether/ Ethyl acetate = 5/1) to obtain the title compound Intermediate 1 (brown solid, 4.5 g, yield 77.05%).
1H NMR(400MHz DMSO)δ6.52(s,1H),6.38(s,1H),4.36-4.31(t,2H),4.25(s,2H),3.02-2.98(t,2H),1.98(s,3H)。1H NMR(400MHz DMSO)δ6.52(s,1H),6.38(s,1H),4.36-4.31(t,2H),4.25(s,2H),3.02-2.98(t,2H),1.98(s ,3H).
LC-MS m/z(ESI)=150.10[M+1]。LC-MS m/z(ESI)=150.10[M+1].
中间体2Intermediate 2
6-氯-2,3-二氢苯并呋喃-5-胺(中间体2)6-Chloro-2,3-dihydrobenzofuran-5-amine (Intermediate 2)
6-chloro-2,3-dihydrobenzofuran-5-amine6-chloro-2,3-dihydrobenzofuran-5-amine
第一步:first step:
1-溴-2-(2-溴乙氧基)-4-氯苯(2B)1-Bromo-2-(2-bromoethoxy)-4-chlorobenzene (2B)
1-bromo-2-(2-bromoethoxy)-4-chlorobenzene1-bromo-2-(2-bromoethoxy)-4-chlorobenzene
将1,2-二溴乙烷(109.2g,581.28mmol)用乙腈(120mL)混匀,然后加入2-溴-5-氯苯酚2A(30g,144.61mmol),最后加入碳酸钾(60g,434.12mmol),在80℃下反应5h。反应完成后过滤,浓缩滤液,硅胶柱色谱分离提纯(石油醚/乙酸乙酯=200/1),得到标题化合物2B(白色固体,31g,产率68.19%)。Mix 1,2-dibromoethane (109.2g, 581.28mmol) with acetonitrile (120mL), then add 2-bromo-5-chlorophenol 2A (30g, 144.61mmol), and finally add potassium carbonate (60g, 434.12 mmol), react at 80°C for 5h. After the reaction was completed, it was filtered, the filtrate was concentrated, and the silica gel column chromatography was separated and purified (petroleum ether/ethyl acetate=200/1) to obtain the title compound 2B (white solid, 31 g, yield 68.19%).
第二步:The second step:
6-氯-2,3-二氢苯并呋喃(2C)6-Chloro-2,3-dihydrobenzofuran (2C)
6-chloro-2,3-dihydrobenzofuran6-chloro-2,3-dihydrobenzofuran
将1-溴-2-(2-溴乙氧基)-4-氯苯2B(31g,98.60mmol)加入干燥的反应瓶,用干燥的四氢呋喃(160mL)溶解,然后在-78℃下滴加正丁基锂(45.5mL,118.32mmol),滴加完成后继续反应1.5h。反应完成后,向反应液中加入水(20mL)淬灭,减压除去有机溶剂,用乙酸乙酯萃取两次,合并有机相,干燥浓缩,使用硅胶柱色谱分离提纯(纯石油醚),得到标题化合物2C(无色液体,15g,产率98.42%)Add 1-bromo-2-(2-bromoethoxy)-4-chlorobenzene 2B (31g, 98.60mmol) into a dry reaction flask, dissolve with dry tetrahydrofuran (160mL), and then add dropwise at -78℃ N-Butyllithium (45.5mL, 118.32mmol), the reaction was continued for 1.5h after the addition was completed. After the reaction was completed, the reaction solution was quenched by adding water (20 mL), removing the organic solvent under reduced pressure, extracting twice with ethyl acetate, combining the organic phases, drying and concentrating, and using silica gel column chromatography to separate and purify (pure petroleum ether) to obtain The title compound 2C (colorless liquid, 15g, yield 98.42%)
1H NMR(400MHz CDCl3)δ6.98-6.96(dt,1H),6.72-6.69(dd,1H),6.67(d,1H),4.50-4.46(t,2H),3.08-3.03(t,2H)。1H NMR (400MHz CDCl3) δ 6.98-6.96 (dt, 1H), 6.72-6.69 (dd, 1H), 6.67 (d, 1H), 4.50-4.46 (t, 2H), 3.08-3.03 (t, 2H) .
第三步:third step:
6-氯-5-硝基-2,3-二氢苯并呋喃(2D)6-chloro-5-nitro-2,3-dihydrobenzofuran (2D)
6-chloro-5-nitro-2,3-dihydrobenzofuran6-chloro-5-nitro-2,3-dihydrobenzofuran
将6-氯-2,3-二氢苯并呋喃2C(15g,97.03mmol)溶于醋酸(110mL)中,在70℃下滴入硝酸(15.5mL,232.87mmol,68%纯度),滴加完成后继续反应30min。TLC监测反应完全,将反应液倒入冰水中,用乙酸乙酯萃取三次,干燥浓缩有机相,使用硅胶柱色谱分离提纯(石油醚/乙酸乙酯=20/1),得到标题化合物2D(黄色固体,12.5g,产率64.55%)。6-Chloro-2,3-dihydrobenzofuran 2C (15g, 97.03mmol) was dissolved in acetic acid (110mL), and nitric acid (15.5mL, 232.87mmol, 68% purity) was added dropwise at 70°C. After completion, the reaction was continued for 30 minutes. TLC monitored the completion of the reaction, the reaction solution was poured into ice water, extracted with ethyl acetate three times, dried and concentrated the organic phase, separated and purified by silica gel column chromatography (petroleum ether/ethyl acetate = 20/1) to obtain the title compound 2D (yellow) Solid, 12.5 g, yield 64.55%).
LC-MS m/z(ESI)=200.00[M+1]。LC-MS m/z(ESI)=200.00[M+1].
第四步:the fourth step:
6-氯-2,3-二氢苯并呋喃-5-胺(中间体2)6-Chloro-2,3-dihydrobenzofuran-5-amine (Intermediate 2)
6-chloro-2,3-dihydrobenzofuran-5-amine6-chloro-2,3-dihydrobenzofuran-5-amine
将6-氯-5-硝基-2,3-二氢苯并呋喃2D(12.5g,62.63mmol)用110mL(乙醇/水=10/1)混匀,加入铁粉(17.8g,318.77mmol),最后加入稀盐酸(16.5ml,2mol/L),在85℃反应2h。将反应液过滤除去铁粉,浓缩滤液,然后用饱和碳酸氢钠溶液调节pH至弱碱性,然后用乙酸乙酯萃取三次,合并有机相,干燥浓缩,使用硅胶柱色谱分离纯化(石油醚/乙酸乙酯=15/1),得标题化合物中间体2(黄色固体,7.0g,产率65.91%)。Mix 6-chloro-5-nitro-2,3-dihydrobenzofuran 2D (12.5g, 62.63mmol) with 110mL (ethanol/water=10/1), add iron powder (17.8g, 318.77mmol) ), finally add dilute hydrochloric acid (16.5ml, 2mol/L), and react at 85°C for 2h. The reaction solution was filtered to remove iron powder, the filtrate was concentrated, and the pH was adjusted to weakly alkaline with saturated sodium bicarbonate solution, and then extracted three times with ethyl acetate. The organic phases were combined, dried and concentrated, and separated and purified by silica gel column chromatography (petroleum ether/ Ethyl acetate = 15/1) to obtain the title compound Intermediate 2 (yellow solid, 7.0 g, yield 65.91%).
1H NMR(400MHz DMSO)δ6.72(s,1H),6.64(s,1H),4.73(s,2H),4.43-4.39(t,2H),3.07-3.02(t,2H)。1H NMR (400MHz DMSO) δ 6.72 (s, 1H), 6.64 (s, 1H), 4.73 (s, 2H), 4.43-4.39 (t, 2H), 3.07-3.02 (t, 2H).
LC-MS m/z(ESI)=170.00[M+1]。LC-MS m/z(ESI)=170.00[M+1].
实施例1Example 1
7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物1)7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9 -Dihydro-8H-purin-8-one (Compound 1)
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8 -one
第一步:first step:
2-氯-4-((四氢-2H-吡喃-4-基)氨基)嘧啶-5-羧酸乙酯(1b)Ethyl 2-chloro-4-((tetrahydro-2H-pyran-4-yl)amino)pyrimidine-5-carboxylate (1b)
ethyl 2-chloro-4-((tetrahydro-2H-pyran-4-yl)amino)pyrimidine-5-carboxylateethyl 2-chloro-4-((tetrahydro-2H-pyran-4-yl)amino)pyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(30.00g,136.4mmol)、四氢-2H-吡喃-4-胺盐酸盐(18.66g,136.4mmol)溶解于乙腈(600mL),搅拌多次加入碳酸钾(46.92g,340.9mmol),在室温搅拌4h。TLC监测反应完毕后过滤,滤渣用乙酸乙酯(300mL)清洗,将滤液浓缩得粗品,粗品通过柱分离提纯(正己烷:乙酸乙酯(v/v)=1:1)得标题化合物2-氯-4-((四氢-2H-吡喃-4-基)氨基)嘧啶-5-羧酸乙酯(1b)(白色固体,30.0g,产率77.4%)。2,4-Dichloropyrimidine-5-carboxylic acid ethyl ester 1a (30.00g, 136.4mmol), tetrahydro-2H-pyran-4-amine hydrochloride (18.66g, 136.4mmol) were dissolved in acetonitrile (600mL ), adding potassium carbonate (46.92g, 340.9mmol) several times with stirring, and stirring at room temperature for 4h. After the reaction was monitored by TLC, it was filtered. The residue was washed with ethyl acetate (300 mL). The filtrate was concentrated to obtain a crude product. The crude product was separated and purified by column (n-hexane: ethyl acetate (v/v) = 1:1) to obtain the title compound 2- Ethyl chloro-4-((tetrahydro-2H-pyran-4-yl)amino)pyrimidine-5-carboxylate (1b) (white solid, 30.0 g, yield 77.4%).
1H NMR(400MHz DMSO)δ8.62(s,1H),8.32(d,1H),4.30(q,2H),4.21-4.16(m,1H),3.86-3.83(m,2H),3.48-3.42(m,2H),1.88-1.85(m,2H),1.62-1.53(m,2H),1.31(t,3H)。1H NMR(400MHz DMSO)δ8.62(s,1H),8.32(d,1H),4.30(q,2H),4.21-4.16(m,1H),3.86-3.83(m,2H),3.48-3.42 (m, 2H), 1.88-1.85 (m, 2H), 1.62-1.53 (m, 2H), 1.31 (t, 3H).
LC-MS m/z(ESI)=286.10[M+1]。LC-MS m/z(ESI)=286.10[M+1].
第二步:The second step:
2-氯-4-((四氢-2H-吡喃-4-基)氨基)嘧啶-5-羧酸(1c)2-chloro-4-((tetrahydro-2H-pyran-4-yl)amino)pyrimidine-5-carboxylic acid (1c)
2-chloro-4-((tetrahydro-2H-pyran-4-yl)amino)pyrimidine-5-carboxylic acid2-chloro-4-((tetrahydro-2H-pyran-4-yl)amino)pyrimidine-5-carboxylic acid
将2-氯-4-((四氢-2H-吡喃-4-基)氨基)嘧啶-5-羧酸乙酯1b(30g,104.99mmol)溶解于四氢呋喃/水(200mL/200mL)中,加入氢氧化锂(5.03g,209.99mmol),室温搅拌1h。TLC监测反应完全,浓缩除去四氢呋喃,用6N盐酸调pH为5,有白色固体析出,过滤,滤饼用石油醚洗两次,搜集固体得到标题化合物2-氯-4-((四氢-2H-吡喃-4-基)氨基)嘧啶-5-羧酸(1c)(白色固体,15.0g,产率55.44%)。Dissolve 2-chloro-4-((tetrahydro-2H-pyran-4-yl)amino)pyrimidine-5-carboxylic acid ethyl ester 1b (30g, 104.99mmol) in tetrahydrofuran/water (200mL/200mL), Lithium hydroxide (5.03 g, 209.99 mmol) was added, and the mixture was stirred at room temperature for 1 h. TLC monitored the reaction to complete, concentrated to remove tetrahydrofuran, adjusted the pH to 5 with 6N hydrochloric acid, a white solid precipitated out, filtered, the filter cake was washed twice with petroleum ether, and the solid was collected to obtain the title compound 2-chloro-4-((tetrahydro-2H -Pyran-4-yl)amino)pyrimidine-5-carboxylic acid (1c) (white solid, 15.0 g, yield 55.44%).
1H NMR(400MHz DMSO)δ8.60(s,1H),8.54(d,1H),4.20-4.15(m,1H),3.86-3.83(m,2H),3.48-3.42(m,2H),1.89-1.86(m,2H),1.60-1.50(m,2H)。1H NMR(400MHz DMSO) δ8.60(s,1H), 8.54(d,1H), 4.20-4.15(m,1H), 3.86-3.83(m,2H), 3.48-3.42(m,2H), 1.89 -1.86 (m, 2H), 1.60-1.50 (m, 2H).
LC-MS m/z(ESI)=258.10[M+1]。LC-MS m/z(ESI)=258.10[M+1].
第三步:third step:
2-氯-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(1d)2-chloro-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one (1d)
2-chloro-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one2-chloro-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one
将2-氯-4-((四氢-2H-吡喃-4-基)氨基)嘧啶-5-羧酸1c(15g,58.21mmol)溶解于二甲基乙酰胺(150mL),加入三乙胺(7.38mL,58.21mmol)、叠氮磷酸二苯酯(12.06mL,58.21mmol),随后逐步升温至120℃搅拌反应1.5h。TLC监测反应完毕,将反应液倒入冰水中,过滤搜集固体,水洗3次,真空浓缩干燥得到标题化合物2-氯-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(1d)(白色固体,13.0g,产率87.69%)。Dissolve 2-chloro-4-((tetrahydro-2H-pyran-4-yl)amino)pyrimidine-5-carboxylic acid 1c (15g, 58.21mmol) in dimethylacetamide (150mL) and add triethyl Amine (7.38mL, 58.21mmol), diphenyl azide phosphate (12.06mL, 58.21mmol), and then gradually heated to 120°C and stirred for 1.5h. TLC monitors the completion of the reaction, the reaction solution is poured into ice water, the solid is collected by filtration, washed with water 3 times, and concentrated and dried in vacuo to obtain the title compound 2-chloro-9-(tetrahydro-2H-pyran-4-yl)-7,9 -Dihydro-8H-purin-8-one (1d) (white solid, 13.0 g, yield 87.69%).
1H NMR(400MHz DMSO)δ11.63(s,1H),8.11(s,1H),4.43-4.37(m,1H),3.98-3.94(m,2H),2.59-2.38(m,2H),1.73-1.65(m,2H)。1H NMR (400MHz DMSO) δ 11.63 (s, 1H), 8.11 (s, 1H), 4.43-4.37 (m, 1H), 3.98-3.94 (m, 2H), 2.59-2.38 (m, 2H), 1.73 -1.65 (m, 2H).
LC-MS m/z(ESI)=255.10[M+1]。LC-MS m/z(ESI)=255.10[M+1].
第四步:the fourth step:
2-氯-7-甲基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(1e)2-chloro-7-methyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one (1e)
2-chloro-7-methyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one2-chloro-7-methyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one
将2-氯-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮1d(5g,19.63mmol)溶解于二甲基甲酰胺(50mL),在0℃下加入硫酸二甲酯(2.48g,19.63mmol)和碳酸铯(9.5g,29.445mmol),0℃搅拌1h。TLC监测反应结束,将反应液倒入冰水中,有固体析出,过滤、搜集固体得到标题化合物2-氯-7-甲基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(1e)(白色固体,3.0g,产率56.87%)。Dissolve 2-chloro-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one 1d (5g, 19.63mmol) in dimethylformamide ( 50mL), add dimethyl sulfate (2.48g, 19.63mmol) and cesium carbonate (9.5g, 29.445mmol) at 0°C, and stir at 0°C for 1h. TLC monitors the end of the reaction. Pour the reaction solution into ice water. A solid precipitates out. Filter and collect the solid to obtain the title compound 2-chloro-7-methyl-9-(tetrahydro-2H-pyran-4-yl)-7 , 9-dihydro-8H-purin-8-one (1e) (white solid, 3.0 g, yield 56.87%).
1HNMR(400MHz DMSO)δ8.36(s,1H),4.50-4.41(m,1H),3.99-3.95(m,2H),3.48-3.42(m,2H),3.34(s,3H),2.47-2.38(m,2H),1.70-1.66(m,2H)。1HNMR(400MHz DMSO)δ8.36(s,1H), 4.50-4.41(m,1H), 3.99-3.95(m,2H), 3.48-3.42(m,2H), 3.34(s,3H), 2.47- 2.38 (m, 2H), 1.70 to 1.66 (m, 2H).
LC-MS m/z(ESI)=268.10[M+1]。LC-MS m/z(ESI)=268.10[M+1].
第五步:the fifth step:
7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物1)7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9 -Dihydro-8H-purin-8-one (Compound 1)
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8 -one
将2-氯-7-甲基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮1e(126mg,0.47mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(70mg,0.47mmol)、碳酸铯(305mg,0.94mmol)、三(二亚苄基丙酮)二钯(42mg,0.047mmol)2,2'-双(二苯基膦基)-1,1'-联萘(58mg,0.094mmol)溶解于二氧六环(10mL),氮气保护并换气,在100℃搅拌4h。TLC监测反应结束,将反应液倒入冰水中,收集固体,将固体用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v)=30:1)纯化得标题化合物7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物1)(白色固体,40mg,产率22.35%)。The 2-chloro-7-methyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one 1e (126mg, 0.47mmol), 6- Methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (70mg, 0.47mmol), cesium carbonate (305mg, 0.94mmol), tris(dibenzylideneacetone) two palladium (42mg, 0.047mmol) ) 2,2'-bis(diphenylphosphino)-1,1'-binaphthyl (58mg, 0.094mmol) was dissolved in dioxane (10mL), protected with nitrogen and ventilated, and stirred at 100°C for 4h. TLC monitors the end of the reaction, the reaction solution is poured into ice water, the solid is collected, and the solid is separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v)=30:1) to obtain the title compound 7-methyl-2 -((6-Methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H- Purine-8-one (Compound 1) (white solid, 40 mg, yield 22.35%).
1H NMR(400MHz DMSO)δ8.25(s,1H),7.95(s,1H),7.23(s,1H),6.6(s,1H),4.48(t,2H),4.32-4.50(m,1H),3.92-3.97(dd,2H),3.40(t,2H),3.27(s,3H),3.11(t,2H),2.45- 2.54(m,2H),2.11(s,3H),1.61-1.64(m,2H)。1H NMR (400MHz DMSO) δ 8.25 (s, 1H), 7.95 (s, 1H), 7.23 (s, 1H), 6.6 (s, 1H), 4.48 (t, 2H), 4.32-4.50 (m, 1H) ),3.92-3.97(dd,2H),3.40(t,2H),3.27(s,3H),3.11(t,2H),2.45-2.54(m,2H),2.11(s,3H),1.61- 1.64 (m, 2H).
LC-MS m/z(ESI)=382.30[M+1]。LC-MS m/z(ESI)=382.30[M+1].
实施例2Example 2
9-(((1r,4r)-4-甲氧基环己基)-7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物2)9-(((1r,4r)-4-methoxycyclohexyl)-7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino) -7,9-dihydro-8H-purin-8-one (compound 2)
9-((1r,4r)-4-methoxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-((1r,4r)-4-methoxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8 -one
第一步:first step:
2-氯-4-(反-4-甲氧基环己基)氨基)嘧啶-5-羧酸乙酯(2a)2-Chloro-4-(trans-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylic acid ethyl ester (2a)
ethyl 2-chloro-4-((trans-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylateethyl 2-chloro-4-((trans-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylate
将2,4-二氯-5-嘧啶甲酸乙酯1a(5.13g,23.22mmol)溶于乙腈(20mL)中,0℃搅拌下加入碳酸钾(6.42g,46.44mmol),再缓慢滴加(1r,4r)-4-甲氧基环己烷-1-胺(2.00g,15.48mmol)的乙腈溶液(10mL)升温至常温搅拌反应1h,TLC监测至反应结束,硅藻土过滤后用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=2:1)纯化得标题化合物2-氯-4-((反-4-甲氧基环己基)氨基)嘧啶-5-羧酸乙酯(2a)(白色固体,2.00g,产率41.26%)。Ethyl 2,4-dichloro-5-pyrimidinecarboxylate 1a (5.13g, 23.22mmol) was dissolved in acetonitrile (20mL), potassium carbonate (6.42g, 46.44mmol) was added with stirring at 0°C, and then slowly added dropwise ( 1r,4r)-4-methoxycyclohexane-1-amine (2.00g, 15.48mmol) in acetonitrile (10mL) was heated to room temperature and stirred for 1h, monitored by TLC until the reaction was over, filtered through diatomaceous earth and filtered with silica gel Column chromatography separation and purification (petroleum ether/ethyl acetate (v/v)=2:1) to obtain the title compound 2-chloro-4-((trans-4-methoxycyclohexyl)amino)pyrimidine-5-carboxy Ethyl acid (2a) (white solid, 2.00 g, yield 41.26%).
LC-MS m/z(ESI)=314.10[M+1]。LC-MS m/z(ESI)=314.10[M+1].
第二步:The second step:
2-氯-4-((反-4-甲氧基环己基)氨基)嘧啶-5-羧酸(2b)2-chloro-4-((trans-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylic acid (2b)
2-chloro-4-((trans-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylate2-chloro-4-((trans-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylate
将2-氯-4-((反-4-甲氧基环己基)氨基)嘧啶-5-羧酸乙酯2a(2.00g,6.37mmol)溶解于四氢呋喃/水(15mL/15mL)中,加入氢氧化锂一水合物(0.80g,19.12mmol),常温搅拌反应2h,TLC监测至反应结束,浓缩蒸发掉四氢呋喃后加入2N HCl调节pH至3-4左右,有白色固体析出,过滤,滤饼用水以及石油醚/乙酸乙酯(v/v=10/1)洗两次得标题化合物2-氯-4-((反-4-甲氧基环己基)氨基)嘧啶-5-羧酸(2b)(白色固体,1.67g,产率91.70%)。Dissolve 2-chloro-4-((trans-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylic acid ethyl ester 2a (2.00g, 6.37mmol) in tetrahydrofuran/water (15mL/15mL) and add Lithium hydroxide monohydrate (0.80g, 19.12mmol), stirring at room temperature for 2h, TLC monitoring until the end of the reaction, concentration and evaporation of tetrahydrofuran, adding 2N HCl to adjust the pH to about 3-4, a white solid precipitated, filter, filter cake Wash twice with water and petroleum ether/ethyl acetate (v/v=10/1) to obtain the title compound 2-chloro-4-((trans-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylic acid ( 2b) (white solid, 1.67 g, yield 91.70%).
LC-MS m/z(ESI)=286.10[M+1]。LC-MS m/z(ESI)=286.10[M+1].
第三步:third step:
2-氯-9-(反-4-甲氧基环己基)-7,9-二氢-8H-嘌呤-8-酮(2c)2-chloro-9-(trans-4-methoxycyclohexyl)-7,9-dihydro-8H-purin-8-one (2c)
2-chloro-9-(trans-4-methoxycyclohexyl)-7,9-dihydro-8H-purin-8-one2-chloro-9-(trans-4-methoxycyclohexyl)-7,9-dihydro-8H-purin-8-one
将2-氯-4-(反-4-甲氧基环己基)氨基)嘧啶-5-羧酸2b(1.57g,5.49mmol)用N,N-二甲基乙酰胺(16mL)溶解,常温搅拌下加入三乙胺(0.76mL,5.49mmol)和叠氮磷酸二苯酯(1.18mL,5.49mmol)反应2h后,升温至110℃回流反应2.5h。TLC监测至反应结束,反应液中加水二氯甲烷进行萃取,浓缩有机层得标题化合物2-氯-9-(反-4-甲氧基环己基)-7,9-二氢-8H-嘌呤-8-酮(2c)(白色固体,1.70g,粗品,产率109.47%)。Dissolve 2-chloro-4-(trans-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylic acid 2b (1.57g, 5.49mmol) with N,N-dimethylacetamide (16mL) at room temperature Under stirring, triethylamine (0.76 mL, 5.49 mmol) and diphenyl azide phosphate (1.18 mL, 5.49 mmol) were added and reacted for 2 h, and then the temperature was raised to 110° C. and refluxed for 2.5 h. TLC monitors to the end of the reaction, the reaction solution is extracted with water and dichloromethane, and the organic layer is concentrated to obtain the title compound 2-chloro-9-(trans-4-methoxycyclohexyl)-7,9-dihydro-8H-purine -8-one (2c) (white solid, 1.70 g, crude product, yield 109.47%).
1H NMR(400MHz,DMSO-d6)δ11.61(s,1H),8.11(s,1H),4.20–4.10(m,1H),3.25(s,3H),3.22–3.14(m,1H),2.30–2.18(m,2H),2.15–2.06(m,2H),1.81–1.73(d,2H),1.31–1.18(m,2H)。1H NMR(400MHz,DMSO-d6)δ11.61(s,1H),8.11(s,1H),4.20-4.10(m,1H),3.25(s,3H),3.22-3.14(m,1H), 2.30–2.18(m,2H), 2.15–2.06(m,2H), 1.81–1.73(d,2H), 1.31–1.18(m,2H).
LC-MS m/z(ESI)=283.00[M+1]。LC-MS m/z(ESI)=283.00[M+1].
第四步:the fourth step:
2-氯-9-(反-4-甲氧基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(2d)2-Chloro-9-(trans-4-methoxycyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one (2d)
2-chloro-9-(trans-4-methoxycyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one2-chloro-9-(trans-4-methoxycyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one
将2-氯-9-(反-4-甲氧基环己基)-7,9-二氢-8H-嘌呤-8-酮2c(1.70g,6.01mmol)用N,N-二甲基甲酰胺(20mL)溶解,0℃搅拌下加入硫酸二甲酯(0.57mL,6.01mmol)和碳酸铯(3.92g,12.03mmol)反应2h。TLC监测至反应结束,把反应液缓慢滴加到冰水中搅拌,析出白色固体,用水和石油醚洗3次过滤得到标题化合物2-氯-9-(反-4-甲氧基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(2d)(白色固体,1.2g,产率67.25%)。Use 2-chloro-9-(trans-4-methoxycyclohexyl)-7,9-dihydro-8H-purin-8-one 2c (1.70g, 6.01mmol) with N,N-dimethylformaldehyde The amide (20 mL) was dissolved, and dimethyl sulfate (0.57 mL, 6.01 mmol) and cesium carbonate (3.92 g, 12.03 mmol) were added under stirring at 0°C to react for 2 h. TLC monitored until the reaction was over, the reaction solution was slowly added dropwise to ice water and stirred, a white solid precipitated out, washed with water and petroleum ether three times and filtered to obtain the title compound 2-chloro-9-(trans-4-methoxycyclohexyl)- 7-Methyl-7,9-dihydro-8H-purin-8-one (2d) (white solid, 1.2 g, yield 67.25%).
1H NMR(400MHz,DMSO-d6)δ8.34(s,1H),4.26–4.15(m,1H),3.35(s,3H),3.27(s,3H),3.25–3.16(m,1H),2.31–2.19(m,2H),2.15–2.08(m,2H),1.83–1.75(m,2H),1.34–1.21(m,2H)。1H NMR (400MHz, DMSO-d6) δ 8.34 (s, 1H), 4.26-4.15 (m, 1H), 3.35 (s, 3H), 3.27 (s, 3H), 3.25-3.16 (m, 1H), 2.31–2.19(m,2H), 2.15–2.08(m,2H), 1.83–1.75(m,2H), 1.34–1.21(m,2H).
LC-MS m/z(ESI)=297.10[M+1]。LC-MS m/z(ESI)=297.10[M+1].
第五步:the fifth step:
9-(反-4-甲氧基环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物2)9-(trans-4-methoxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-di Hydrogen-8H-purin-8-one (Compound 2)
9-(trans-4-methoxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(trans-4-methoxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one
将2-氯-9-(反-4-甲氧基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮2d(100mg,0.34mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(101mg,0.68mmol)、碳酸铯(240mg,0.68mmol)和Brettphos G3 Pd(31mg,0.034mmol)加入干燥的反应瓶,氮气置换三次后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化得到标题化合物9-(反-4-甲氧基环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物2)(浅粉色固体,65mg,产率47.11%)。The 2-chloro-9-(trans-4-methoxycyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one 2d (100mg, 0.34mmol), 6-methyl -2,3-Dihydrobenzofuran-5-amine intermediate 1 (101mg, 0.68mmol), cesium carbonate (240mg, 0.68mmol) and Brettphos G3 Pd (31mg, 0.034mmol) were added to the dry reaction flask and replaced with nitrogen After three times, dry 1,4-dioxane (1 mL) was added and reacted at 110° C. for 5 h. The reaction was monitored by TLC until the end of the reaction. After cooling to room temperature, it was separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 9-(trans-4-methoxycyclohexyl)- 7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one (compound 2) ( Light pink solid, 65 mg, yield 47.11%).
1H NMR(400MHz,DMSO-d6)δ8.20(s,1H),7.95(s,1H),7.23(s,1H),6.62(s,1H),4.49(t,2H),4.16–4.07(m,1H),3.25(s,6H),3.12(t,2H),3.09–3.02(m,1H),2.34–2.22(m,2H),2.14–2.04(m,5H),1.71(d,2H),1.27–1.15(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.20(s,1H),7.95(s,1H),7.23(s,1H),6.62(s,1H),4.49(t,2H),4.16-4.07( m, 1H), 3.25 (s, 6H), 3.12 (t, 2H), 3.09-3.02 (m, 1H), 2.34-2.22 (m, 2H), 2.14-2.04 (m, 5H), 1.71 (d, 2H), 1.27–1.15 (m, 2H).
LC-MS m/z(ESI)=410.20[M+1]。LC-MS m/z(ESI)=410.20[M+1].
实施例3Example 3
9-(反-4-羟基-4-甲基环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物3)9-(trans-4-hydroxy-4-methylcyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7, 9-dihydro-8H-purin-8-one (compound 3)
9-(trans-4-hydroxy-4-methylcyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydro-benzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(trans-4-hydroxy-4-methylcyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydro-benzofuran-5-yl)amino)-7,9-dihydro-8H-purin -8-one
第一步:first step:
2-氯-4-(反-4-羟基-4-甲基环己基)氨基)嘧啶-5-羧酸乙酯(3a)Ethyl 2-chloro-4-(trans-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylate (3a)
ethyl 2-chloro-4-((trans-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylateethyl 2-chloro-4-((trans-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylate
将2,4-二氯-5-嘧啶甲酸乙酯1a(5.00g,30.18mmol)溶于乙腈(30mL)中,0℃搅拌下加入碳酸钾(12.51g,90.55mmol),再缓慢滴加反-4-氨基-1-甲基环己醇盐酸盐(10.01g,45.27mmol)的乙腈溶液(10mL)升温至常温搅拌反应1h,TLC监测至反应结束,硅藻土过滤后用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=2:1)纯化得标题化合物2-氯-4-((反-4-羟基-4-甲基环己基)氨基)嘧啶-5-羧酸乙酯(3a)(白色固体,4.70g,产率49.63%)。Ethyl 2,4-dichloro-5-pyrimidinecarboxylate 1a (5.00g, 30.18mmol) was dissolved in acetonitrile (30mL), potassium carbonate (12.51g, 90.55mmol) was added with stirring at 0°C, and the reaction was slowly added dropwise. The acetonitrile solution (10 mL) of -4-amino-1-methylcyclohexanol hydrochloride (10.01 g, 45.27 mmol) was heated to room temperature and the reaction was stirred for 1 h, monitored by TLC until the reaction was over, filtered through diatomaceous earth, and chromatographed on a silica gel column. Separation and purification (petroleum ether/ethyl acetate (v/v)=2:1) to obtain the title compound 2-chloro-4-((trans-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5- Ethyl carboxylate (3a) (white solid, 4.70 g, yield 49.63%).
1H NMR(400MHz,Chloroform-d)δ8.66(s,1H),4.36(q,2H),4.30–4.21(m,1H),2.10–2.00(m,2H),1.74–1.63(m,5H),1.62–1.50(m,3H),1.40(t,3H),1.31(s,3H)。1H NMR(400MHz,Chloroform-d)δ8.66(s,1H), 4.36(q,2H), 4.30-4.21(m,1H), 2.10-2.00(m,2H), 1.74-1.63(m,5H) ), 1.62–1.50 (m, 3H), 1.40 (t, 3H), 1.31 (s, 3H).
LC-MS m/z(ESI)=314.10[M+1]。LC-MS m/z(ESI)=314.10[M+1].
第二步:The second step:
2-氯-4-((反-4-羟基-4-甲基环己基)氨基)嘧啶-5-羧酸(3b)2-chloro-4-((trans-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylic acid (3b)
2-chloro-4-((trans-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylic acid2-chloro-4-((trans-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylic acid
将2-氯-4-((反-4-羟基-4-甲基环己基)氨基)嘧啶-5-羧酸乙酯3a(4.70g,14.98mmol)溶解于四氢呋喃/水(40mL/20mL)中,加入氢氧化锂一水合物(1.89g,44.94mmol),常温搅拌反应2h,TLC监测至反应结束,浓缩蒸发掉四氢呋喃后加入2N HCl调节pH至3-4左右,有白色固体析出,过滤,滤饼用水以及石油醚/乙酸乙酯(v/v=10/1)洗两次得标题化合物2-氯-4-((反-4-羟基-4-甲基环己基)氨基)嘧啶-5-羧酸(3b)(白色固体,4.20g,产率98.15%)。Dissolve 2-chloro-4-((trans-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylic acid ethyl ester 3a (4.70g, 14.98mmol) in tetrahydrofuran/water (40mL/20mL) Add lithium hydroxide monohydrate (1.89g, 44.94mmol), stir the reaction at room temperature for 2h, monitor by TLC until the reaction is over, concentrate and evaporate the tetrahydrofuran, add 2N HCl to adjust the pH to about 3-4, a white solid precipitates out, filter , The filter cake was washed twice with water and petroleum ether/ethyl acetate (v/v=10/1) to obtain the title compound 2-chloro-4-((trans-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine -5-carboxylic acid (3b) (white solid, 4.20 g, yield 98.15%).
1H NMR(400MHz,DMSO-d6)δ8.68(d,1H),8.56(s,1H),4.02(s,1H),3.16(s,1H),1.91–1.83(m,2H),1.51–1.45(m,6H),1.15(s,3H)。1H NMR(400MHz,DMSO-d6)δ8.68(d,1H),8.56(s,1H),4.02(s,1H),3.16(s,1H),1.91-1.83(m,2H),1.51-- 1.45 (m, 6H), 1.15 (s, 3H).
LC-MS m/z(ESI)=314.10[M+1]。LC-MS m/z(ESI)=314.10[M+1].
第三步:third step:
2-氯-9-(反-4-羟基-4-甲基环己基)-7,9-二氢-8H-嘌呤-8-酮(3c)2-chloro-9-(trans-4-hydroxy-4-methylcyclohexyl)-7,9-dihydro-8H-purin-8-one (3c)
2-chloro-9-(trans-4-hydroxy-4-methylcyclohexyl)-7,9-dihydro-8H-purin-8-one2-chloro-9-(trans-4-hydroxy-4-methylcyclohexyl)-7,9-dihydro-8H-purin-8-one
将2-氯-4-((反-4-羟基-4-甲基环己基)氨基)嘧啶-5-羧酸3b(4.34g,15.19mmol)用N,N-二甲基乙酰胺(30mL)溶解,常温搅拌下加入三乙胺(2.11mL,15.19mmol)和叠氮磷酸二苯酯(3.27mL,15.19mmol)反应2h后,升温至110℃回流反应2.5h。TLC监测至反应结束,反应液中加水有白色固体析出,过滤,滤饼用水以及石油醚/乙酸乙酯(v/v=10/1)洗两次得标题化合物2-氯-9-(反-4-羟基-4-甲基环己基)-7,9-二氢-8H-嘌呤-8-酮(3c)(白色固体,1.82g,产率42.36%)。Use 2-chloro-4-((trans-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylic acid 3b (4.34g, 15.19mmol) with N,N-dimethylacetamide (30mL ) Was dissolved, and triethylamine (2.11 mL, 15.19 mmol) and diphenyl azide phosphate (3.27 mL, 15.19 mmol) were added under stirring at room temperature and reacted for 2 hours, and then heated to 110° C. and refluxed for 2.5 hours. TLC monitoring to the end of the reaction, the reaction solution was added with water and a white solid precipitated, filtered, the filter cake was washed twice with water and petroleum ether/ethyl acetate (v/v=10/1) to obtain the title compound 2-chloro-9-(anti- -4-hydroxy-4-methylcyclohexyl)-7,9-dihydro-8H-purin-8-one (3c) (white solid, 1.82 g, yield 42.36%).
LC-MS m/z(ESI)=283.00[M+1]。LC-MS m/z(ESI)=283.00[M+1].
第四步:the fourth step:
2-氯-9-(反-4-羟基-4-甲基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(3d)2-chloro-9-(trans-4-hydroxy-4-methylcyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one (3d)
2-chloro-9-(trans-4-hydroxy-4-methylcyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one2-chloro-9-(trans-4-hydroxy-4-methylcyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one
将2-氯-9-(反-4-羟基-4-甲基环己基)-7,9-二氢-8H-嘌呤-8-酮3c(1.80g,6.37mmol)用N,N-二甲基甲酰胺(20mL)溶解,0℃搅拌下加入硫酸二甲酯(0.60mL,6.37mmol)和碳酸铯(3.11g,9.55mmol)反应2h。TLC监测至反应结束,把反应液缓慢滴加到冰水中搅拌,析出白色固体,用水和石油醚洗3次过滤得到标题化合物2-氯-9-(反-4-羟基-4-甲基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(3d)(白色固体,0.80g,产率42.34%)。Use 2-chloro-9-(trans-4-hydroxy-4-methylcyclohexyl)-7,9-dihydro-8H-purin-8-one 3c (1.80g, 6.37mmol) with N,N-two Methylformamide (20 mL) was dissolved, and dimethyl sulfate (0.60 mL, 6.37 mmol) and cesium carbonate (3.11 g, 9.55 mmol) were added under stirring at 0°C to react for 2 h. The reaction was monitored by TLC until the reaction was over. The reaction solution was slowly added dropwise to ice water and stirred, a white solid precipitated out, washed with water and petroleum ether three times, and filtered to obtain the title compound 2-chloro-9-(trans-4-hydroxy-4-methyl ring Hexyl)-7-methyl-7,9-dihydro-8H-purin-8-one (3d) (white solid, 0.80 g, yield 42.34%).
1H NMR(400MHz,DMSO-d6)δ8.35(s,1H),4.48(s,1H),4.23–4.12(m,1H),3.35(s,3H),2.42–2.25(m,2H),1.65(d,4H),1.58–1.47(m,2H),1.26(s,3H)。1H NMR(400MHz,DMSO-d6)δ8.35(s,1H),4.48(s,1H),4.23-4.12(m,1H),3.35(s,3H),2.42-2.25(m,2H), 1.65 (d, 4H), 1.58-1.47 (m, 2H), 1.26 (s, 3H).
LC-MS m/z(ESI)=297.10[M+1]。LC-MS m/z(ESI)=297.10[M+1].
第五步:the fifth step:
9-(反-4-羟基-4-甲基环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物3)9-(trans-4-hydroxy-4-methylcyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7, 9-dihydro-8H-purin-8-one (compound 3)
9-(trans-4-hydroxy-4-methylcyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydro-benzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(trans-4-hydroxy-4-methylcyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydro-benzofuran-5-yl)amino)-7,9-dihydro-8H-purin -8-one
将2-氯-9-((1r,4r)-4-羟基-4-甲基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮3d(100mg,0.34mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(101mg,0.68mmol)、碳酸铯(240mg,0.68mmol)和Brettphos G3 Pd(31mg,0.034mmol)加入干燥的反应瓶,氮气置换三次后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化得到标题化合物9-(反-4-羟基-4-甲基环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物3)(浅粉色固体,42mg,产率30.44%)。The 2-chloro-9-((1r,4r)-4-hydroxy-4-methylcyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one 3d (100mg, 0.34 mmol), 6-methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (101mg, 0.68mmol), cesium carbonate (240mg, 0.68mmol) and Brettphos G3 Pd (31mg, 0.034mmol) were added Dry the reaction flask, replace it with nitrogen three times, add dry 1,4-dioxane (1 mL), and react at 110°C for 5 hours. The reaction was monitored by TLC until the end of the reaction. After cooling to room temperature, it was separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 9-(trans-4-hydroxy-4-methyl ring Hexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one (compound 3) (Light pink solid, 42 mg, yield 30.44%).
1H NMR(400MHz,DMSO-d6)δ8.22(s,1H),7.97(s,1H),7.10(s,1H),6.60(s,1H),4.48(t,2H),4.35(s,1H),4.10–3.99(m,1H),3.26(s,3H),3.10(t,2H),2.30–2.19(m,2H),2.09(s,3H),1.58–1.39(m,6H),0.98(s,3H)。1H NMR (400MHz, DMSO-d6) δ 8.22 (s, 1H), 7.97 (s, 1H), 7.10 (s, 1H), 6.60 (s, 1H), 4.48 (t, 2H), 4.35 (s, 1H), 4.10–3.99(m,1H), 3.26(s,3H), 3.10(t,2H), 2.30–2.19(m,2H), 2.09(s,3H), 1.58–1.39(m,6H) ,0.98(s,3H).
LC-MS m/z(ESI)=410.20[M+1]。LC-MS m/z(ESI)=410.20[M+1].
实施例4Example 4
7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(1-甲基哌啶-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物4)7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(1-methylpiperidin-4-yl)-7,9 -Dihydro-8H-purin-8-one (Compound 4)
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(1-methylpiperidin-4-yl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(1-methylpiperidin-4-yl)-7,9-dihydro-8H-purin-8-one
第一步:first step:
4-((1-(叔丁氧基羰基)哌啶-4-基)氨基)-2-氯嘧啶-5-羧酸乙酯(4a)4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester (4a)
ethyl 4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxylateethyl 4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(5g,22.6mmol)、碳酸钾(6.2g,45.2mmol)溶解于乙腈(20mL),在0℃下加入4-氨基哌啶-1-羧酸叔丁酯(4.5g,22.6mmol),在室温搅拌20h,TLC监测至反应完全,加水和乙酸乙酯萃取三次,无水硫酸钠干燥后用硅胶柱色谱分离提纯(正己烷/乙酸乙酯(v/v)=10:)),浓缩得到标题化合物4-((1-(叔丁氧基羰基)哌啶-4-基)氨基)-2-氯嘧啶-5-羧酸乙酯(4a)(白色固体,8.2g,产率95%)。Dissolve 2,4-dichloropyrimidine-5-carboxylic acid ethyl ester 1a (5g, 22.6mmol), potassium carbonate (6.2g, 45.2mmol) in acetonitrile (20mL), add 4-aminopiperidine- Tert-butyl 1-carboxylate (4.5g, 22.6mmol), stirred at room temperature for 20h, monitored by TLC until the reaction was complete, added water and ethyl acetate for extraction three times, dried over anhydrous sodium sulfate and purified by silica gel column chromatography (n-hexane/ Ethyl acetate (v/v)=10:)), and concentrated to obtain the title compound 4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxylic acid Ethyl ester (4a) (white solid, 8.2 g, yield 95%).
1H NMR(400MHz DMSO)δ8.63(s,1H),8.32(d,1H),4.33-4.28(m,2H),4.17-4.14(m,1H),3.85(d,2H),2.94(s,2H),1.88-1.80(m,2H),1.51-1.44(m,2H),1.41(s,9H),1.32-1.29(m,3H)。1H NMR(400MHz DMSO) δ8.63(s,1H), 8.32(d,1H), 4.33-4.28(m,2H), 4.17-4.14(m,1H), 3.85(d,2H), 2.94(s , 2H), 1.88-1.80 (m, 2H), 1.51-1.44 (m, 2H), 1.41 (s, 9H), 1.32-1.29 (m, 3H).
LC-MS m/z(ESI)=385.10[M+1]。LC-MS m/z(ESI)=385.10[M+1].
第二步:The second step:
4-((1-(叔丁氧基羰基)哌啶-4-基)氨基)-2-氯嘧啶-5-羧酸(4b)4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxylic acid (4b)
4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxy-lic acid4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxy-lic acid
将4-((1-(叔丁氧基羰基)哌啶-4-基)氨基)-2-氯嘧啶-5-羧酸乙酯4a(8.2g,21.3mmol)溶解于四氢呋喃/水(10mL/5mL)中,加入氢氧化锂(1.8g,42.7mmol),室温搅拌1h。TLC监测至反应完全,浓缩蒸发掉四氢呋喃,调pH至4-5,有白色固体析出,过滤,滤饼用石油醚/乙酸乙酯(v/v=10/1)洗两次,浓缩得到标题化合物4-((1-(叔丁氧基羰基)哌啶-4-基)氨基)-2-氯嘧啶-5-羧酸(4b)(白色固体,7g,产率86%)。Dissolve 4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester 4a (8.2g, 21.3mmol) in tetrahydrofuran/water (10mL /5mL), lithium hydroxide (1.8g, 42.7mmol) was added, and the mixture was stirred at room temperature for 1h. TLC monitors the reaction until the reaction is complete, concentrates and evaporates the tetrahydrofuran, adjusts the pH to 4-5, and a white solid precipitates out. It is filtered. The filter cake is washed twice with petroleum ether/ethyl acetate (v/v=10/1) and concentrated to obtain the title Compound 4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxylic acid (4b) (white solid, 7g, yield 86%).
1H NMR(400MHz DMSO)δ8.59(s,1H),8.54(d,1H),4.20-4.10(m,1H),3.87-3.84(m,2H),2.96(s,3H),1.91-1.73(m,3H),1.41(s,9H)。1H NMR(400MHz DMSO)δ8.59(s,1H), 8.54(d,1H), 4.20-4.10(m,1H), 3.87-3.84(m,2H), 2.96(s,3H), 1.91-1.73 (m, 3H), 1.41 (s, 9H).
LC-MS m/z(ESI)=357.10[M+1]。LC-MS m/z(ESI)=357.10[M+1].
第三步third step
4-(2-氯-8-氧代-7,8-二氢-9H-嘌呤-9-基)哌啶-1-甲酸叔丁酯(4c)Tert-Butyl 4-(2-chloro-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine-1-carboxylate (4c)
tert-butyl 4-(2-chloro-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine-1-carboxylatetert-butyl 4-(2-chloro-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine-1-carboxylate
将4-((1-(叔丁氧基羰基)哌啶-4-基)氨基)-2-氯嘧啶-5-羧酸4b(7g,19.6mmol)溶解于二甲基乙酰胺(10mL),加入三乙胺(1.96g,19.6mmol)、叠氮磷酸二苯酯(5.4g,19.6mmol),随后逐步升温至110℃搅拌1.5h,TLC监测至反应完全,浓缩反应液,残留物用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=1:10),得到标题化合物4-(2-氯-8-氧代-7,8-二氢-9H-嘌呤-9-基)哌啶-1-甲酸叔丁酯(4c)(白色固体,6.4g,产率87%)。Dissolve 4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxylic acid 4b (7g, 19.6mmol) in dimethylacetamide (10mL) , Add triethylamine (1.96g, 19.6mmol) and diphenyl azide phosphate (5.4g, 19.6mmol), then gradually increase the temperature to 110°C and stir for 1.5h. TLC monitors until the reaction is complete. The reaction solution is concentrated and the residue is used Separation and purification by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)=1:10) gave the title compound 4-(2-chloro-8-oxo-7,8-dihydro-9H-purine-9) -Yl)piperidine-1-carboxylic acid tert-butyl ester (4c) (white solid, 6.4 g, yield 87%).
1H NMR(400MHz DMSO)δ8.14(s,1H),4.41-4.33(m,1H),4.06(d,2H),2.88(s,2H), 2.30-2.20(m,2H),1.84-1.71(m,2H),1.43(s,9H)。1H NMR(400MHz DMSO)δ8.14(s,1H),4.41-4.33(m,1H),4.06(d,2H),2.88(s,2H), 2.30-2.20(m,2H),1.84-1.71 (m, 2H), 1.43 (s, 9H).
LC-MS m/z(ESI)=354.10[M+1]。LC-MS m/z(ESI)=354.10[M+1].
第四步the fourth step
4-(2-氯-7-甲基-8-氧代-7,8-二氢-9H-嘌呤-9-基)哌啶-1-甲酸叔丁酯(4d)Tert-Butyl 4-(2-chloro-7-methyl-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine-1-carboxylate (4d)
tert-butyl4-(2-chloro-7-methyl-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine-1-carboxylatetert-butyl4-(2-chloro-7-methyl-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine-1-carboxylate
将4-(2-氯-8-氧代-7,8-二氢-9H-嘌呤-9-基)哌啶-1-甲酸叔丁酯6c(6.4g,18.1mmol)溶解于二甲基甲酰胺(10mL),在0℃下加入硫酸二甲酯(2.28g,18.1mmol)和碳酸铯(8.5g,27.1mmol),0℃搅拌0.5h。TLC监测至反应完全,向反应液中加入水,有固体析出,过滤得到标题化合物4-(2-氯-7-甲基-8-氧代-7,8-二氢-9H-嘌呤-9-基)哌啶-1-甲酸叔丁酯(4d)(白色固体,5.4g,产率79%)。Dissolve tert-butyl 4-(2-chloro-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine-1-carboxylate 6c (6.4g, 18.1mmol) in dimethyl Formamide (10 mL), dimethyl sulfate (2.28 g, 18.1 mmol) and cesium carbonate (8.5 g, 27.1 mmol) were added at 0°C, and stirred at 0°C for 0.5 h. TLC monitors until the reaction is complete, water is added to the reaction solution, a solid precipitates out, filtered to obtain the title compound 4-(2-chloro-7-methyl-8-oxo-7,8-dihydro-9H-purine-9 -Yl)piperidine-1-carboxylic acid tert-butyl ester (4d) (white solid, 5.4 g, yield 79%).
1H NMR(400MHz DMSO)δ8.36(s,1H),4.46-4.37(m,1H),4.05(d,2H),3.35(s,3H),2.87(s,2H),2.33-2.19(m,2H),1.74-1.72(m,2H),1.43(s,9H)。1H NMR (400MHz DMSO) δ 8.36 (s, 1H), 4.46-4.37 (m, 1H), 4.05 (d, 2H), 3.35 (s, 3H), 2.87 (s, 2H), 2.33-2.19 (m , 2H), 1.74-1.72 (m, 2H), 1.43 (s, 9H).
LC-MS m/z(ESI)=368.10[M+1]。LC-MS m/z(ESI)=368.10[M+1].
第五步:the fifth step:
2-氯-7-甲基-9-(哌啶-4-基)-7,9-二氢-8H-嘌呤-8-酮(4e)2-chloro-7-methyl-9-(piperidin-4-yl)-7,9-dihydro-8H-purin-8-one (4e)
2-chloro-7-methyl-9-(piperidin-4-yl)-7,9-dihydro-8H-purin-8-one2-chloro-7-methyl-9-(piperidin-4-yl)-7,9-dihydro-8H-purin-8-one
将4-(2-氯-8-氧代-7,8-二氢-9H-嘌呤-9-基)哌啶-1-甲酸叔丁酯4d(2g,5.4mmol)加入到反应瓶中,常温搅拌下加入2M盐酸-乙酸乙酯溶液(10mL),常温搅拌4h。TLC监测至反应完全,浓缩反应液,得到标题化合物2-氯-7-甲基-9-(哌啶-4-基)-7,9-二氢-8H-嘌呤-8-酮的盐酸盐(4e)(白色固体,1.4g,产率91%)。Add tert-butyl 4-(2-chloro-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine-1-carboxylate 4d (2g, 5.4mmol) into the reaction flask, 2M hydrochloric acid-ethyl acetate solution (10 mL) was added with stirring at room temperature, and stirred at room temperature for 4 hours. TLC monitoring until the reaction is complete, the reaction solution is concentrated to obtain the title compound 2-chloro-7-methyl-9-(piperidin-4-yl)-7,9-dihydro-8H-purin-8-one hydrochloric acid Salt (4e) (white solid, 1.4 g, yield 91%).
1H NMR(400MHz DMSO)δ8.39(s,1H),6.42(s,1H),4.59-4.53(m,1H),3.39(s,2H),3.36(s,3H),3.16-3.04(m,2H),2.62-2.50(m,2H),2.07–1.93(m,2H)。1H NMR (400MHz DMSO) δ 8.39 (s, 1H), 6.42 (s, 1H), 4.59-4.53 (m, 1H), 3.39 (s, 2H), 3.36 (s, 3H), 3.16-3.04 (m , 2H), 2.62-2.50 (m, 2H), 2.07-1.93 (m, 2H).
LC-MS m/z(ESI)=268.10[M+1]。LC-MS m/z(ESI)=268.10[M+1].
第六步:The sixth step:
2-氯-7-甲基-9-(1-甲基哌啶-4-基)-7,9-二氢-8H-嘌呤-8-酮(4f)2-chloro-7-methyl-9-(1-methylpiperidin-4-yl)-7,9-dihydro-8H-purin-8-one (4f)
2-chloro-7-methyl-9-(1-methylpiperidin-4-yl)-7,9-dihydro-8H-purin-8-one2-chloro-7-methyl-9-(1-methylpiperidin-4-yl)-7,9-dihydro-8H-purin-8-one
将2-氯-7-甲基-9-(哌啶-4-基)-7,9-二氢-8H-嘌呤-8-酮4e(1.4g,5.4mmol)溶解于甲醇(20mL),加入4A分子筛(100mg),然后加入多聚甲醛(783mg,27mmol),在室温搅拌反应6h,再加入氰基硼氢化钠(1g,16.2mmol)。TLC监测至反应完全,过滤并浓缩得到标题化合物2-氯-7-甲基-9-(1-甲基哌啶-4-基)-7,9-二氢-8H-嘌呤-8-酮(4f)(白色固体,600mg,产率62%)。Dissolve 2-chloro-7-methyl-9-(piperidin-4-yl)-7,9-dihydro-8H-purin-8-one 4e (1.4g, 5.4mmol) in methanol (20mL), 4A molecular sieve (100mg) was added, then paraformaldehyde (783mg, 27mmol) was added, the reaction was stirred at room temperature for 6h, and then sodium cyanoborohydride (1g, 16.2mmol) was added. TLC monitored until the reaction was complete, filtered and concentrated to obtain the title compound 2-chloro-7-methyl-9-(1-methylpiperidin-4-yl)-7,9-dihydro-8H-purin-8-one (4f) (white solid, 600 mg, yield 62%).
1H NMR(400MHz DMSO)δ8.35(s,1H),4.21-4.13(m,1H),3.35(s,3H),2.92(d,2H),2.45-2.41(m,2H),2.23(s,3H),2.09-2.03(m,2H),1.70-1.67(m,2H)。1H NMR(400MHz DMSO)δ8.35(s,1H), 4.21-4.13(m,1H), 3.35(s,3H), 2.92(d,2H), 2.45-2.41(m,2H), 2.23(s , 3H), 2.09-2.03 (m, 2H), 1.70-1.67 (m, 2H).
LC-MS m/z(ESI)=282.10[M+1]。LC-MS m/z(ESI)=282.10[M+1].
第七步:The seventh step:
7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(1-甲基哌啶-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物4)7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(1-methylpiperidin-4-yl)-7,9 -Dihydro-8H-purin-8-one (Compound 4)
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(1-methylpiperidin-4-yl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(1-methylpiperidin-4-yl)-7,9-dihydro-8H-purin-8-one
将2-氯-7-甲基-9-(1-甲基哌啶-4-基)-7,9-二氢-8H-嘌呤-8-酮4f(100mg,0.35mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(106mg,0.71mmol)、碳酸铯(231mg,0.71mmol)和Brettphos G3 Pd(32mg,0.035mmol)加入干燥的反应瓶,氮气置换三次后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化得到标题化合物7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(1-甲基哌啶-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物 4)(白色固体,35mg,产率25.00%)。The 2-chloro-7-methyl-9-(1-methylpiperidin-4-yl)-7,9-dihydro-8H-purin-8-one 4f (100mg, 0.35mmol), 6-methyl 2-,3-dihydrobenzofuran-5-amine intermediate 1 (106mg, 0.71mmol), cesium carbonate (231mg, 0.71mmol) and Brettphos G3 Pd (32mg, 0.035mmol) into the dry reaction flask, nitrogen After three replacements, dry 1,4-dioxane (1 mL) was added and reacted at 110° C. for 5 h. The reaction was monitored by TLC until the end of the reaction. After cooling to room temperature, it was separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 7-methyl-2-(((6-methyl -2,3-Dihydrobenzofuran-5-yl)amino)-9-(1-methylpiperidin-4-yl)-7,9-dihydro-8H-purin-8-one (compound 4 ) (White solid, 35 mg, yield 25.00%).
1H NMR(400MHz,DMSO-d6)δ8.20(s,1H),7.94(s,1H),7.25(s,1H),6.60(s,1H),4.49(t,2H),4.13–4.03(m,1H),3.26(s,3H),3.12(t,2H),2.86(d,2H),2.59–2.47(m,2H),2.19(s,3H),2.12(s,3H),1.94(t,2H),1.61(d,2H)。1H NMR(400MHz,DMSO-d6)δ8.20(s,1H),7.94(s,1H),7.25(s,1H),6.60(s,1H),4.49(t,2H),4.13-4.03( m, 1H), 3.26 (s, 3H), 3.12 (t, 2H), 2.86 (d, 2H), 2.59-2.47 (m, 2H), 2.19 (s, 3H), 2.12 (s, 3H), 1.94 (t, 2H), 1.61 (d, 2H).
LC-MS m/z(ESI)=395.20[M+1]。LC-MS m/z(ESI)=395.20[M+1].
实施例5Example 5
9-环己基-7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物5)9-Cyclohexyl-7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purine-8- Ketone (Compound 5)
9-cyclohexyl-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-cyclohexyl-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one
第一步:first step:
2-氯-4-(环己基氨基)嘧啶-5-羧酸乙酯(5a)Ethyl 2-chloro-4-(cyclohexylamino)pyrimidine-5-carboxylate (5a)
ethyl 2-chloro-4-(cyclohexylamino)pyrimidine-5-carboxylateethyl 2-chloro-4-(cyclohexylamino)pyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(5.0g,22.6mmol)、碳酸钾(6.2g,45.2mmol)溶解于乙腈(50mL),在0℃下加入环己氨(2.2g,22.6mmol),在室温搅拌20h。TLC监测反应结束,加水和乙酸乙酯萃取三次,无水硫酸钠干燥并用硅胶柱色谱分离提纯(正己烷/乙酸乙酯(v/v)=10:1),浓缩得到标题化合物2-氯-4-(环己基氨基)嘧啶-5-羧酸乙酯(5a)(白色固体,4.1g,产率64%)。Dissolve 2,4-dichloropyrimidine-5-carboxylic acid ethyl ester 1a (5.0g, 22.6mmol), potassium carbonate (6.2g, 45.2mmol) in acetonitrile (50mL), add cyclohexylamine (2.2 g, 22.6mmol), stirred at room temperature for 20h. TLC monitors the completion of the reaction, adds water and ethyl acetate to extract three times, dry with anhydrous sodium sulfate and separate and purify by silica gel column chromatography (n-hexane/ethyl acetate (v/v)=10:1), and concentrate to obtain the title compound 2-chloro- Ethyl 4-(cyclohexylamino)pyrimidine-5-carboxylate (5a) (white solid, 4.1 g, yield 64%).
1H NMR(400MHz,Chloroform-d)δ8.64(s,1H),8.53–8.23(m,1H),4.35(q,2H),4.25–4.05(m,1H),2.01-1.95(m,2H),1.77-1.72(m,2H),1.64-1.61(m,1H),1.50-1.41(m,3H),1.39(t,3H),1.35–1.18(m,2H)。1H NMR(400MHz,Chloroform-d)δ8.64(s,1H), 8.53-8.23(m,1H), 4.35(q,2H), 4.25-4.05(m,1H), 2.01-1.95(m,2H) ),1.77-1.72(m,2H),1.64-1.61(m,1H),1.50-1.41(m,3H),1.39(t,3H),1.35-1.18(m,2H).
LC-MS m/z(ESI)=284.10[M+1]。LC-MS m/z(ESI)=284.10[M+1].
第二步:The second step:
2-氯-4-(环己基氨基)嘧啶-5-羧酸(5b)2-Chloro-4-(cyclohexylamino)pyrimidine-5-carboxylic acid (5b)
2-chloro-4-(cyclohexylamino)pyrimidine-5-carboxylic acid2-chloro-4-(cyclohexylamino)pyrimidine-5-carboxylic acid
将2-氯-4-(环己基氨基)嘧啶-5-羧酸乙酯5a(4.1g,14.4mmol)溶解于四氢呋喃/水(20mL/20mL)中,加入氢氧化锂(691mg,28.8mmol),室温搅拌1h。TLC监测反应结束,旋干四氢呋喃,调pH至4-5,有白色固体析出,过滤,滤饼用石油醚/乙酸乙酯(v/v=10/1)洗两次,浓缩得到标题化合物2-氯-4-(环己基氨基)嘧啶-5-羧酸(5b)(白色固体,3.0g, 产率81%)。Dissolve 2-chloro-4-(cyclohexylamino)pyrimidine-5-carboxylic acid ethyl ester 5a (4.1g, 14.4mmol) in tetrahydrofuran/water (20mL/20mL), add lithium hydroxide (691mg, 28.8mmol) , Stir at room temperature for 1h. The reaction was monitored by TLC, the tetrahydrofuran was spin-dried, and the pH was adjusted to 4-5. A white solid precipitated out. The filter cake was washed twice with petroleum ether/ethyl acetate (v/v=10/1) and concentrated to obtain the title compound 2. -Chloro-4-(cyclohexylamino)pyrimidine-5-carboxylic acid (5b) (white solid, 3.0 g, yield 81%).
1H NMR(400MHz,DMSO-d6)δ13.77(s,1H),8.76–8.45(m,2H),4.01-3.92(m,1H),2.05–1.79(m,2H),1.79–1.65(m,2H),1.60-1.54(m,1H),1.42–1.19(m,5H)。1H NMR(400MHz,DMSO-d6)δ13.77(s,1H),8.76-8.45(m,2H),4.01-3.92(m,1H),2.05-1.79(m,2H),1.79-1.65(m ,2H),1.60-1.54(m,1H),1.42-1.19(m,5H).
LC-MS m/z(ESI)=256.10[M+1]。LC-MS m/z(ESI)=256.10[M+1].
第三步:third step:
2-氯-9-环己基-7,9-二氢-8H-嘌呤-8-酮(5c)2-chloro-9-cyclohexyl-7,9-dihydro-8H-purin-8-one (5c)
2-chloro-9-cyclohexyl-7,9-dihydro-8H-purin-8-one2-chloro-9-cyclohexyl-7,9-dihydro-8H-purin-8-one
将2-氯-4-(环己基氨基)嘧啶-5-羧酸5b(3.0g,11.7mmol)溶解于二甲基乙酰胺(30mL),加入三乙胺(1.18g,11.7mmol)、叠氮磷酸二苯酯(3.22g,11.7mmol),随后逐步升温至110℃搅拌1.5h。TLC监测反应结束,浓缩反应液,残留物用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=1:10)得到标题化合物2-氯-9-环己基-7,9-二氢-8H-嘌呤-8-酮(5c)(白色固体,2.2g,产率74%)。Dissolve 2-chloro-4-(cyclohexylamino)pyrimidine-5-carboxylic acid 5b (3.0g, 11.7mmol) in dimethylacetamide (30mL), add triethylamine (1.18g, 11.7mmol), stack Diphenyl nitrogen phosphate (3.22g, 11.7mmol), then gradually heated to 110°C and stirred for 1.5h. The end of the reaction was monitored by TLC, the reaction solution was concentrated, and the residue was separated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)=1:10) to obtain the title compound 2-chloro-9-cyclohexyl-7,9- Dihydro-8H-purin-8-one (5c) (white solid, 2.2 g, yield 74%).
1H NMR(400MHz,DMSO-d6)δ11.60(s,1H),8.12(s,1H),4.19-4.11(m,1H),1.92–1.57(m,6H),1.52–0.93(m,4H)。1H NMR(400MHz,DMSO-d6)δ11.60(s,1H),8.12(s,1H),4.19-4.11(m,1H),1.92-1.57(m,6H),1.52-0.93(m,4H) ).
LC-MS m/z(ESI)=253.10[M+1]。LC-MS m/z(ESI)=253.10[M+1].
第四步:the fourth step:
2-氯-9-环己基-7-甲基-7,9-二氢-8H-嘌呤-8-酮(5d)2-chloro-9-cyclohexyl-7-methyl-7,9-dihydro-8H-purin-8-one (5d)
2-chloro-9-cyclohexyl-7-methyl-7,9-dihydro-8H-purin-8-one2-chloro-9-cyclohexyl-7-methyl-7,9-dihydro-8H-purin-8-one
将2-氯-9-环己基-7,9-二氢-8H-嘌呤-8-酮5c(2.2g,8.7mmol)溶解于二甲基甲酰胺(15mL),在0℃下加入硫酸二甲酯(1.1g,8.7mmol)和碳酸铯(4.25g,13mmol)搅拌反应0.5h。TLC监测反应结束,向反应液中加入水,有固体析出,过滤得到标题化合物2-氯-9-环己基-7-甲基-7,9-二氢-8H-嘌呤-8-酮(5d)(黄色固体,1.1g,产率48%)。Dissolve 2-chloro-9-cyclohexyl-7,9-dihydro-8H-purin-8-one 5c (2.2g, 8.7mmol) in dimethylformamide (15mL), add two sulfuric acid at 0℃ Methyl ester (1.1g, 8.7mmol) and cesium carbonate (4.25g, 13mmol) were stirred and reacted for 0.5h. The end of the reaction was monitored by TLC, water was added to the reaction solution, a solid precipitated out, and the title compound 2-chloro-9-cyclohexyl-7-methyl-7,9-dihydro-8H-purin-8-one (5d ) (Yellow solid, 1.1 g, yield 48%).
1H NMR(400MHz,DMSO-d6)δ8.34(s,1H),4.22-4.41(m,1H),3.35(s,3H),2.22-2.12(m,2H),1.91–1.60(m,5H),1.43-1.33(m,2H),1.27-1.14(m,1H)。1H NMR(400MHz,DMSO-d6)δ8.34(s,1H),4.22-4.41(m,1H),3.35(s,3H),2.22-2.12(m,2H),1.91-160(m,5H) ), 1.43-1.33 (m, 2H), 1.27-1.14 (m, 1H).
LC-MS m/z(ESI)=267.10[M+1]。LC-MS m/z(ESI)=267.10[M+1].
第五步:the fifth step:
9-环己基-7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物5)9-Cyclohexyl-7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purine-8- Ketone (Compound 5)
9-cyclohexyl-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-cyclohexyl-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one
将2-氯-9-环己基-7-甲基-7,9-二氢-8H-嘌呤-8-酮5d(100mg,0.37mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(112mg,0.75mmol)、碳酸铯(244mg,0.75mmol)和Brettphos G3 Pd(34mg,0.037mmol)加入干燥的反应瓶,氮气置换三次后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化得到标题化合物9-环己基-7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物5)(浅黄色固体,34mg,产率23.90%)。The 2-chloro-9-cyclohexyl-7-methyl-7,9-dihydro-8H-purin-8-one 5d (100mg, 0.37mmol), 6-methyl-2,3-dihydrobenzo Furan-5-amine Intermediate 1 (112mg, 0.75mmol), Cesium Carbonate (244mg, 0.75mmol) and Brettphos G3 Pd (34mg, 0.037mmol) were added to the dry reaction flask, replaced with nitrogen three times and then added to the dry 1,4- Dioxane (1 mL) was reacted at 110°C for 5 hours. The reaction was monitored by TLC until the end of the reaction. After cooling to room temperature, it was separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 9-cyclohexyl-7-methyl-2-(( (6-Methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one (compound 5) (pale yellow solid, 34mg, yield 23.90%).
1H NMR(400MHz,DMSO-d6)δ8.18(s,1H),7.95(s,1H),7.26(s,1H),6.61(s,1H),4.49(t,2H),4.16–4.05(m,1H),3.26(s,3H),3.11(t,2H),2.27–2.15(m,2H),2.12(s,3H),1.83–1.76(m,2H),1.71–1.65(m,2H),1.38–1.22(m,3H),1.17–1.04(m,1H)。1H NMR(400MHz,DMSO-d6)δ8.18(s,1H),7.95(s,1H),7.26(s,1H),6.61(s,1H),4.49(t,2H),4.16-4.05( m, 1H), 3.26 (s, 3H), 3.11 (t, 2H), 2.27-2.15 (m, 2H), 2.12 (s, 3H), 1.83-1.76 (m, 2H), 1.71-1.65 (m, 2H), 1.38–1.22 (m, 3H), 1.17–1.04 (m, 1H).
LC-MS m/z(ESI)=380.20[M+1]。LC-MS m/z(ESI)=380.20[M+1].
实施例6Example 6
7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(化合物6)7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-3-yl)-7, 9-dihydro-8H-purin-8-one (compound 6)
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-3-yl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-3-yl)-7,9-dihydro-8H-purin-8 -one
第一步:first step:
2-氯-4-((四氢-2H-吡喃-3-基)氨基)嘧啶-5-羧酸乙酯(6a)Ethyl 2-chloro-4-((tetrahydro-2H-pyran-3-yl)amino)pyrimidine-5-carboxylate (6a)
ethyl 2-chloro-4-((tetrahydro-2H-pyran-3-yl)amino)pyrimidine-5-carboxylateethyl 2-chloro-4-((tetrahydro-2H-pyran-3-yl)amino)pyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(5.0g,22.6mmol)、碳酸钾(6.2g,45.2mmol)溶解于乙腈(50mL),在0℃下加入四氢-2H-吡喃-3-胺(2.3g,22.6mmol),在室温搅拌20h。加水和乙酸乙酯萃取三次,无水硫酸钠干燥并用硅胶柱色谱分离提纯(正己烷/乙酸乙酯(v/v)=10:1),浓缩得到标题化合物2-氯-4-((四氢-2H-吡喃-3-基)氨基)嘧啶-5-羧酸乙酯(6a)(白色固体,4.1g,产率64%)。Dissolve 2,4-dichloropyrimidine-5-carboxylic acid ethyl ester 1a (5.0g, 22.6mmol), potassium carbonate (6.2g, 45.2mmol) in acetonitrile (50mL), add tetrahydro-2H- at 0℃ Pyran-3-amine (2.3g, 22.6mmol), stirred at room temperature for 20h. Add water and ethyl acetate to extract three times, dry with anhydrous sodium sulfate and separate and purify by silica gel column chromatography (n-hexane/ethyl acetate (v/v)=10:1), and concentrate to obtain the title compound 2-chloro-4-((四Hydrogen-2H-pyran-3-yl)amino)pyrimidine-5-carboxylic acid ethyl ester (6a) (white solid, 4.1 g, yield 64%).
1H NMR(400MHz,Chloroform-d)δ8.69(s,1H),8.68(s,1H),4.37(q,2H),4.34–4.24(m,1H),3.89-3.84(m,1H),3.77-3.66(m,2H),3.58-3.54(m,1H),2.01-1.95(m,1H),1.86-1.65(m,2H),1.69-1.63(m,1H),1.40(t,3H)。1H NMR(400MHz,Chloroform-d)δ8.69(s,1H),8.68(s,1H),4.37(q,2H),4.34-4.24(m,1H),3.89-3.84(m,1H), 3.77-3.66(m,2H),3.58-3.54(m,1H),2.01-1.95(m,1H),1.86-1.65(m,2H),1.69-1.63(m,1H),1.40(t,3H) ).
LC-MS m/z(ESI)=286.10[M+1]。LC-MS m/z(ESI)=286.10[M+1].
第二步:The second step:
2-氯-4-((四氢-2H-吡喃-3-基)氨基)嘧啶-5-羧酸(6b)2-chloro-4-((tetrahydro-2H-pyran-3-yl)amino)pyrimidine-5-carboxylic acid (6b)
2-chloro-4-((tetrahydro-2H-pyran-3-yl)amino)pyrimidine-5-carboxylic acid2-chloro-4-((tetrahydro-2H-pyran-3-yl)amino)pyrimidine-5-carboxylic acid
将2-氯-4-((四氢-2H-吡喃-3-基)氨基)嘧啶-5-羧酸乙酯6a(4.1g,14.3mmol)溶解于四氢呋喃/水(20mL/20mL)中,加入氢氧化锂(686mg,28.6mmol),室温搅拌反应1h。TLC监测至反应结束,旋干四氢呋喃,调pH至4-5,有白色固体析出,过滤,滤饼用石油醚/乙酸乙酯(v/v=10/1)洗两次,浓缩得到标题化合物2-氯-4-((四氢-2H-吡喃-3-基)氨基)嘧啶-5-羧酸(6b)(白色固体,3.5g,产率95%)。Dissolve 2-chloro-4-((tetrahydro-2H-pyran-3-yl)amino)pyrimidine-5-carboxylic acid ethyl ester 6a (4.1g, 14.3mmol) in tetrahydrofuran/water (20mL/20mL) , Lithium hydroxide (686 mg, 28.6 mmol) was added, and the reaction was stirred at room temperature for 1 h. TLC monitors to the end of the reaction, spin-dry the tetrahydrofuran, adjust the pH to 4-5, a white solid is precipitated, filtered, the filter cake is washed twice with petroleum ether/ethyl acetate (v/v=10/1), and concentrated to obtain the title compound 2-Chloro-4-((tetrahydro-2H-pyran-3-yl)amino)pyrimidine-5-carboxylic acid (6b) (white solid, 3.5 g, yield 95%).
1H NMR(400MHz,DMSO-d6)δ13.83(s,1H),8.75(d,1H),8.59(s,1H),4.14-4.07(m,1H),3.76-3.72(m,1H),3.59(t,1H),3.47-3.42(m,2H),1.93-1.86(m,1H),1.79–1.62(m,2H),1.58-1.49(m,1H)。1H NMR(400MHz,DMSO-d6)δ13.83(s,1H),8.75(d,1H),8.59(s,1H),4.14-4.07(m,1H),3.76-3.72(m,1H), 3.59 (t, 1H), 3.47-3.42 (m, 2H), 1.93-1.86 (m, 1H), 1.79-1.62 (m, 2H), 1.58-1.49 (m, 1H).
LC-MS m/z(ESI)=258.10[M+1]。LC-MS m/z(ESI)=258.10[M+1].
第三步:third step:
2-氯-9-(四氢-2H-吡喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(6c)2-chloro-9-(tetrahydro-2H-pyran-3-yl)-7,9-dihydro-8H-purin-8-one (6c)
2-chloro-9-(tetrahydro-2H-pyran-3-yl)-7,9-dihydro-8H-purin-8-one2-chloro-9-(tetrahydro-2H-pyran-3-yl)-7,9-dihydro-8H-purin-8-one
将2-氯-4-((四氢-2H-吡喃-3-基)氨基)嘧啶-5-羧酸6b(3.5g,13.6mmol)溶解于二甲基乙酰胺(30mL)中,再加入三乙胺(1.37g,13.6mmol)和叠氮磷酸二苯酯(3.74g,13.6mmol)随后逐步升温至110℃搅拌1.5h。TLC监测至反应结束,浓缩反应液,残留物用 硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=1:10)得到标题化合物2-氯-9-(四氢-2H-吡喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(6c)(白色固体,2.2g,产率63%)。Dissolve 2-chloro-4-((tetrahydro-2H-pyran-3-yl)amino)pyrimidine-5-carboxylic acid 6b (3.5g, 13.6mmol) in dimethylacetamide (30mL), and then Triethylamine (1.37g, 13.6mmol) and diphenyl azide phosphate (3.74g, 13.6mmol) were added and then gradually heated to 110°C and stirred for 1.5h. The reaction was monitored by TLC until the end of the reaction. The reaction solution was concentrated. The residue was separated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)=1:10) to obtain the title compound 2-chloro-9-(tetrahydro-2H- Pyran-3-yl)-7,9-dihydro-8H-purin-8-one (6c) (white solid, 2.2 g, yield 63%).
1H NMR(400MHz,DMSO-d6)δ11.67(s,1H),8.13(s,1H),4.29-4.21(m,1H),3.86–3.77(m,2H),3.37-3.29(m,2H),2.04–1.39(m,4H)。1H NMR(400MHz,DMSO-d6)δ11.67(s,1H),8.13(s,1H),4.29-4.21(m,1H),3.86-3.77(m,2H),3.37-3.29(m,2H) ), 2.04–1.39 (m, 4H).
LC-MS m/z(ESI)=255.10[M+1]。LC-MS m/z(ESI)=255.10[M+1].
第四步:the fourth step:
2-氯-7-甲基-9-(四氢-2H-吡喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(6d)2-chloro-7-methyl-9-(tetrahydro-2H-pyran-3-yl)-7,9-dihydro-8H-purin-8-one (6d)
2-chloro-7-methyl-9-(tetrahydro-2H-pyran-3-yl)-7,9-dihydro-8H-purin-8-one2-chloro-7-methyl-9-(tetrahydro-2H-pyran-3-yl)-7,9-dihydro-8H-purin-8-one
将2-氯-9-(四氢-2H-吡喃-3-基)-7,9-二氢-8H-嘌呤-8-酮6c(2.2g,8.7mmol)溶解于二甲基甲酰胺(15mL),在0℃下加入硫酸二甲酯(1.1g,8.7mmol)和碳酸铯(4.25g,13mmol)搅拌反应0.5h。TLC监测至反应结束,向反应液中加入水,有固体析出,过滤得到标题化合物2-氯-9-环己基-7-甲基-7,9-二氢-8H-嘌呤-8-酮(6d)(黄色固体,1.7g,产率73%)。Dissolve 2-chloro-9-(tetrahydro-2H-pyran-3-yl)-7,9-dihydro-8H-purin-8-one 6c (2.2g, 8.7mmol) in dimethylformamide (15mL), add dimethyl sulfate (1.1g, 8.7mmol) and cesium carbonate (4.25g, 13mmol) at 0°C and stir for 0.5h. TLC monitored to the end of the reaction, water was added to the reaction solution, a solid precipitated out, filtered to obtain the title compound 2-chloro-9-cyclohexyl-7-methyl-7,9-dihydro-8H-purin-8-one ( 6d) (yellow solid, 1.7 g, yield 73%).
1H NMR(400MHz,DMSO-d6)δ8.36(s,1H),4.35–4.23(m,1H),3.91–3.77(m,2H),3.35(s,3H),3.34-3.33(m,2H),2.50-2.44(m,1H),1.98–1.89(m,1H),1.83–1.62(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.36(s,1H), 4.35-4.23(m,1H), 3.91-3.77(m,2H), 3.35(s,3H),3.34-3.33(m,2H) ), 2.50-2.44 (m, 1H), 1.98-1.89 (m, 1H), 1.83-1.62 (m, 2H).
LC-MS m/z(ESI)=269.10[M+1]。LC-MS m/z(ESI)=269.10[M+1].
第五步:the fifth step:
7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(化合物6)7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-3-yl)-7, 9-dihydro-8H-purin-8-one (compound 6)
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-3-yl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-3-yl)-7,9-dihydro-8H-purin-8 -one
将2-氯-9-环己基-7-甲基-7,9-二氢-8H-嘌呤-8-酮6d(100mg,0.37mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(112mg,0.75mmol)、碳酸铯(242mg,0.75mmol)和Brettphos G3 Pd(34mg,0.037mmol)加入干燥的反应瓶,氮气置换三次后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化得到标题化合物7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(化合物6)(浅粉色固体,42mg,产率29.59%)。The 2-chloro-9-cyclohexyl-7-methyl-7,9-dihydro-8H-purin-8-one 6d (100mg, 0.37mmol), 6-methyl-2,3-dihydrobenzo Furan-5-amine intermediate 1 (112mg, 0.75mmol), cesium carbonate (242mg, 0.75mmol) and Brettphos G3 Pd (34mg, 0.037mmol) were added to the dry reaction flask, replaced with nitrogen three times and then added to the dry 1,4- Dioxane (1 mL) was reacted at 110°C for 5 hours. The reaction was monitored by TLC until the end of the reaction. After cooling to room temperature, it was separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 7-methyl-2-(((6-methyl -2,3-Dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-3-yl)-7,9-dihydro-8H-purin-8-one (compound 6) (Light pink solid, 42 mg, yield 29.59%).
1H NMR(400MHz,DMSO-d6)δ8.25(s,1H),7.97(s,1H),7.22(s,1H),6.62(s,1H),4.49(t,2H),4.27–4.16(m,1H),3.94–3.82(m,2H),3.75(dd,1H),3.26(s,3H),3.23–3.16(m,1H),3.11(t,2H),2.49–2.40(m,1H),2.12(s,3H),1.85(d,1H),1.75–1.61(m,2H)。1H NMR (400MHz, DMSO-d6) δ 8.25 (s, 1H), 7.97 (s, 1H), 7.22 (s, 1H), 6.62 (s, 1H), 4.49 (t, 2H), 4.27-4.16 ( m,1H),3.94-3.82(m,2H), 3.75(dd,1H), 3.26(s,3H), 3.23-3.16(m,1H), 3.11(t,2H), 2.49-2.40(m, 1H), 2.12 (s, 3H), 1.85 (d, 1H), 1.75-1.61 (m, 2H).
LC-MS m/z(ESI)=382.20[M+1]。LC-MS m/z(ESI)=382.20[M+1].
实施例7Example 7
(S)-7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(化合物7)(S)-7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydrofuran-3-yl)-7,9- Dihydro-8H-purin-8-one (Compound 7)
(S)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-furan-3-yl)-7,9-dihydro-8H-purin-8-one(S)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-furan-3-yl)-7,9-dihydro-8H-purin -8-one
第一步:first step:
(S)-2-氯-4-((四氢呋喃-3-基)氨基)嘧啶-5-羧酸乙酯(7a)(S)-2-Chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylic acid ethyl ester (7a)
ethyl(S)-2-chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylateethyl(S)-2-chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(5g,5.35mmol)、碳酸钾(1.4g,22.6mmol)溶解于乙腈(20mL),在0℃下加入(S)-四氢呋喃-3-胺(660mg,5.35mmol),在室温搅拌20h。加水和乙酸乙酯萃取三次,无水硫酸钠干燥并用硅胶柱色谱分离提纯(正己烷/乙酸乙酯(v/v)=10:1),浓缩得到标题化合物(S)-2-氯-4-((四氢呋喃-3-基)氨基)嘧啶-5-羧酸乙酯(7a)(白色固体,2.2g,产率36%)。Dissolve 2,4-dichloropyrimidine-5-carboxylic acid ethyl ester 1a (5g, 5.35mmol) and potassium carbonate (1.4g, 22.6mmol) in acetonitrile (20mL), add (S)-tetrahydrofuran- at 0°C 3-amine (660mg, 5.35mmol), stirred at room temperature for 20h. Add water and ethyl acetate to extract three times, dry with anhydrous sodium sulfate and separate and purify by silica gel column chromatography (n-hexane/ethyl acetate (v/v)=10:1), and concentrate to obtain the title compound (S)-2-chloro-4 -((Tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylic acid ethyl ester (7a) (white solid, 2.2 g, yield 36%).
1H NMR(400MHz DMSO)δ8.68(s,1H),8.58(d,1H),4.79-4.77(m,1H),4.39-4.33(m,2H),4.04–3.98(m,2H),3.90-3.87(m,1H),3.76-3.73(m,1H),2.43-2.33(m,1H),1.95-1.88(m,1H),1.39(t,3H)。1H NMR(400MHz DMSO)δ8.68(s,1H), 8.58(d,1H), 4.79-4.77(m,1H), 4.39-4.33(m,2H), 4.04-3.98(m,2H), 3.90 -3.87 (m, 1H), 3.76-3.73 (m, 1H), 2.43-2.33 (m, 1H), 1.95-1.88 (m, 1H), 1.39 (t, 3H).
LC-MS m/z(ESI)=272.00[M+1]。LC-MS m/z(ESI)=272.00[M+1].
第二步:The second step:
(S)-2-氯-4-((四氢呋喃-3-基)氨基)嘧啶-5-羧酸(7b)(S)-2-Chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylic acid (7b)
(S)-2-chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylic acid(S)-2-chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylic acid
将(S)-2-氯-4-((四氢呋喃-3-基)氨基)嘧啶-5-羧酸乙酯7a(2.2g,8.1mmol)溶解于四氢呋喃/水(5mL/5mL)中,加入氢氧化锂(681mg,16.2mmol),室温搅拌反应1h。TLC监测至反应结束,旋干四氢呋喃,调pH至4-5,有白色固体析出,过滤,滤饼用石油醚/乙酸乙酯(v/v=10/1)洗两次,浓缩得到标题化合物(S)-2-氯-4-((四氢呋喃-3-基)氨基)嘧啶-5-羧酸(7b)(白色固体,1.7g,产率89%)。Dissolve (S)-2-chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylic acid ethyl ester 7a (2.2g, 8.1mmol) in tetrahydrofuran/water (5mL/5mL) and add Lithium hydroxide (681 mg, 16.2 mmol) was stirred at room temperature for 1 h. TLC monitors to the end of the reaction, spin-dry the tetrahydrofuran, adjust the pH to 4-5, a white solid is precipitated, filtered, the filter cake is washed twice with petroleum ether/ethyl acetate (v/v=10/1), and concentrated to obtain the title compound (S)-2-Chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylic acid (7b) (white solid, 1.7 g, yield 89%).
1H NMR(400MHz DMSO)δ13.83(s,1H),8.65(s,1H),8.63(d,1H),4.61(s,1H),3.89-3.80(m,2H),3.76-3.70(m,1H),3.65-3.63(m,1H),2.50-2.25(m,1H),1.87-1.86(m,1H)。1H NMR (400MHz DMSO) δ 13.83 (s, 1H), 8.65 (s, 1H), 8.63 (d, 1H), 4.61 (s, 1H), 3.89-3.80 (m, 2H), 3.76-3.70 (m , 1H), 3.65-3.63 (m, 1H), 2.50-2.25 (m, 1H), 1.87-1.86 (m, 1H).
LC-MS m/z(ESI)=244.20[M+1]。LC-MS m/z(ESI)=244.20[M+1].
第三步:third step:
(S)-2-氯-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(7c)(S)-2-chloro-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one (7c)
(S)-2-chloro-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one(S)-2-chloro-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one
将(S)-2-氯-4-((四氢呋喃-3-基)氨基)嘧啶-5-羧酸7b(1.7g,7mmol)溶解于二甲基乙酰胺(10mL),加入三乙胺(707mg,7mmol)、叠氮磷酸二苯酯(1.9g,7mmol),随后逐步升温至110℃搅拌1.5h。TLC监测至反应结束,浓缩反应液,残留物用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=1:6)得到标题化合物(S)-2-氯-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(7c)(白色固体,1.4g,产率87%)。(S)-2-chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylic acid 7b (1.7g, 7mmol) was dissolved in dimethylacetamide (10mL), and triethylamine ( 707 mg, 7 mmol), diphenyl azide phosphate (1.9 g, 7 mmol), and then gradually heated to 110° C. and stirred for 1.5 h. The reaction was monitored by TLC until the end of the reaction. The reaction solution was concentrated. The residue was separated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)=1:6) to obtain the title compound (S)-2-chloro-9-(tetrahydrofuran) -3-yl)-7,9-dihydro-8H-purin-8-one (7c) (white solid, 1.4g, yield 87%).
1H NMR(400MHz DMSO)δ8.13(s,1H),5.00–4.93(m,1H),4.12-4.06(dd,1H),3.97(t,1H),3.88-3.87(m,2H),3.33(s,3H),2.43-2.37(m,1H),2.25-2.20(m,1H)。1H NMR(400MHz DMSO)δ8.13(s,1H), 5.00-4.93(m,1H), 4.12-4.06(dd,1H), 3.97(t,1H), 3.88-3.87(m,2H), 3.33 (s, 3H), 2.43-2.37 (m, 1H), 2.25-2.20 (m, 1H).
LC-MS m/z(ESI)=241.20[M+1]。LC-MS m/z(ESI)=241.20[M+1].
第四步:the fourth step:
(S)-2-氯-7-甲基-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(7d)(S)-2-Chloro-7-methyl-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one (7d)
(S)-2-chloro-7-methyl-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one(S)-2-chloro-7-methyl-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one
将(S)-2-氯-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮7c(1.4g,5.8mmol)溶解于二甲基甲酰胺(10mL)中,在0℃下加入硫酸二甲酯(735mg,5.8mmol)和碳酸铯(2.85g,8.7mmol)搅拌反应0.5h。TLC监测至反应结束,向反应液中加入水,有固体析出,过滤得到标题化合物(S)-2-氯-7-甲基-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(7d)(白色固体,1.2g,产率85%)。Dissolve (S)-2-chloro-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one 7c (1.4g, 5.8mmol) in dimethylformamide (10mL ), add dimethyl sulfate (735 mg, 5.8 mmol) and cesium carbonate (2.85 g, 8.7 mmol) at 0° C. and stir and react for 0.5 h. TLC monitors to the end of the reaction, water is added to the reaction solution, a solid is precipitated, and the title compound (S)-2-chloro-7-methyl-9-(tetrahydrofuran-3-yl)-7,9-dihydro is obtained by filtration. -8H-purin-8-one (7d) (white solid, 1.2g, yield 85%).
1H NMR(400MHz DMSO)δ8.35(s,1H),5.04–4.97(m,1H),4.13-4.07(dd,1H),3.98(t,1H),3.90-3.84(m,2H),3.35(s,3H),2.44-2.37(m,1H),2.28-2.27(m,1H)。1H NMR(400MHz DMSO)δ8.35(s,1H),5.04-4.97(m,1H),4.13-4.07(dd,1H),3.98(t,1H),3.90-3.84(m,2H), 3.35 (s,3H),2.44-2.37(m,1H),2.28-2.27(m,1H).
LC-MS m/z(ESI)=255.20[M+1]。LC-MS m/z(ESI)=255.20[M+1].
第五步:the fifth step:
(S)-7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(化合物7)(S)-7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydrofuran-3-yl)-7,9- Dihydro-8H-purin-8-one (Compound 7)
(S)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-furan-3-yl)-7,9-dihydro-8H-purin-8-one(S)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-furan-3-yl)-7,9-dihydro-8H-purin -8-one
将(S)-2-氯-7-甲基-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮7d(100mg,0.39mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(117mg,0.79mmol)、碳酸铯(256mg,0.75mmol)和Brettphos G3 Pd(36mg,0.039mmol)加入干燥的反应瓶,氮气置换三次后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化得到标题化合物(S)-7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(化合物7)(浅黄色固体,43mg,产率20.79%)。Add (S)-2-chloro-7-methyl-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one 7d (100mg, 0.39mmol), 6-methyl -2,3-Dihydrobenzofuran-5-amine intermediate 1 (117mg, 0.79mmol), cesium carbonate (256mg, 0.75mmol) and Brettphos G3 Pd (36mg, 0.039mmol) were added to the dry reaction flask and replaced with nitrogen After three times, dry 1,4-dioxane (1 mL) was added and reacted at 110° C. for 5 h. The reaction was monitored by TLC until the end of the reaction. After cooling to room temperature, it was separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound (S)-7-methyl-2-((( 6-Methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one (compound 7) (Light yellow solid, 43 mg, yield 20.79%).
1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),7.98(s,1H),7.19(s,1H),6.61(s,1H),4.93-4.83(m,1H),4.49(t,2H),3.96–3.87(m,2H),3.82–3.75(m,2H),3.27(s,3H),3.11(t,2H),2.41–2.32(m,1H),2.21-2.12(m,1H),2.11(s,3H)。1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),7.98(s,1H),7.19(s,1H),6.61(s,1H),4.93-4.83(m,1H),4.49( t, 2H), 3.96--3.87(m, 2H), 3.82--3.75(m, 2H), 3.27(s, 3H), 3.11(t, 2H), 2.41--2.32(m, 1H), 2.21-2.12( m, 1H), 2.11 (s, 3H).
LC-MS m/z(ESI)=368.20[M+1]。LC-MS m/z(ESI)=368.20[M+1].
实施例8Example 8
(R)-7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(化合物8)(R)-7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydrofuran-3-yl)-7,9- Dihydro-8H-purin-8-one (Compound 8)
(R)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-furan-3-yl)-7,9-dihydro-8H-purin-8-one(R)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-furan-3-yl)-7,9-dihydro-8H-purin -8-one
第一步:first step:
(R)-2-氯-4-((四氢呋喃-3-基)氨基)嘧啶-5-羧酸乙酯(8a)(R)-2-Chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylic acid ethyl ester (8a)
ethyl(R)-2-chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylateethyl(R)-2-chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(5g,5.35mmol)、碳酸钾(1.4g,22.6mmol)溶解于乙腈(20mL),在0℃下加入(R)-四氢呋喃-3-胺(660mg,5.35mmol),在室温搅拌20h。加水和乙酸乙酯萃取三次,无水硫酸钠干燥并用硅胶柱色谱分离提纯(正己烷/乙酸乙酯(v/v)=10:1),浓缩得到标题化合物(R)-2-氯-4-((四氢呋喃-3-基)氨基)嘧啶-5-羧酸乙酯(8a)(白色固体,4.3g,产率65%)。Dissolve 2,4-dichloropyrimidine-5-carboxylic acid ethyl ester 1a (5g, 5.35mmol) and potassium carbonate (1.4g, 22.6mmol) in acetonitrile (20mL), add (R)-tetrahydrofuran- at 0°C 3-amine (660mg, 5.35mmol), stirred at room temperature for 20h. Add water and ethyl acetate to extract three times, dry with anhydrous sodium sulfate and separate and purify by silica gel column chromatography (n-hexane/ethyl acetate (v/v)=10:1), and concentrate to obtain the title compound (R)-2-chloro-4 -((Tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylic acid ethyl ester (8a) (white solid, 4.3 g, yield 65%).
1H NMR(400MHz DMSO)δ8.68(s,1H),8.58(d,1H),4.83-4.77(m,1H),4.39-4.33(m,2H),4.02–3.98(m,2H),3.90-3.86(m,1H),3.76-3.73(m,1H),2.43-2.34(m,1H),1.98-1.88(m,1H),1.39(t,3H)。1H NMR(400MHz DMSO)δ8.68(s,1H),8.58(d,1H),4.83-4.77(m,1H),4.39-4.33(m,2H),4.02-3.98(m,2H),3.90 -3.86 (m, 1H), 3.76-3.73 (m, 1H), 2.43-2.34 (m, 1H), 1.98-1.88 (m, 1H), 1.39 (t, 3H).
LC-MS m/z(ESI)=272.00[M+1]。LC-MS m/z(ESI)=272.00[M+1].
第二步:The second step:
(R)-2-氯-4-((四氢呋喃-3-基)氨基)嘧啶-5-羧酸(8b)(R)-2-chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylic acid (8b)
(R)-2-chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylic acid(R)-2-chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylic acid
将(R)-2-氯-4-((四氢呋喃-3-基)氨基)嘧啶-5-羧酸乙酯8a(4.3g,15.8mmol)溶解于四氢呋喃/水(5mL/5mL)中,加入氢氧化锂(1.3g,31.7mmol),室温搅拌反应1h。TLC监测至反应结束,旋干四氢呋喃,调pH至4-5,有白色固体析出,过滤,滤饼用石油醚/乙酸乙酯(v/v=10/1)洗两次,浓缩得到标题化合物(R)-2-氯-4-((四氢呋喃-3-基)氨基)嘧啶-5-羧酸(8b)(白色固体,2.9g,产率87%)。(R)-2-chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylic acid ethyl ester 8a (4.3g, 15.8mmol) was dissolved in tetrahydrofuran/water (5mL/5mL) and added Lithium hydroxide (1.3g, 31.7mmol) was stirred at room temperature for 1h. TLC monitors to the end of the reaction, spin-dry the tetrahydrofuran, adjust the pH to 4-5, a white solid is precipitated, filtered, the filter cake is washed twice with petroleum ether/ethyl acetate (v/v=10/1), and concentrated to obtain the title compound (R)-2-chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylic acid (8b) (white solid, 2.9 g, yield 87%).
1H NMR(400MHz DMSO)δ13.83(s,1H),8.65(s,1H),8.59(d,1H),4.62-4.60(m,1H),3.89-3.80(m,2H),3.76-3.70(m,1H),3.64-3.56(m,1H),2.33-2.23(m,1H),1.88-1.83(m,1H)。1H NMR(400MHz DMSO)δ13.83(s,1H),8.65(s,1H),8.59(d,1H),4.62-4.60(m,1H),3.89-3.80(m,2H),3.76-3.70 (m, 1H), 3.64-3.56 (m, 1H), 2.33-2.23 (m, 1H), 1.88-1.83 (m, 1H).
LC-MS m/z(ESI)=244.20[M+1]。LC-MS m/z(ESI)=244.20[M+1].
第三步:third step:
(R)-2-氯-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(8c)(R)-2-chloro-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one (8c)
(R)-2-chloro-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one(R)-2-chloro-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one
将(R)-2-氯-4-((四氢呋喃-3-基)氨基)嘧啶-5-羧酸8b(2.9g,11.9mmol)溶解于二甲基乙酰胺(20mL),加入三乙胺(1.2g,11.9mmol)、叠氮磷酸二苯酯(3.3g,11.9mmol),随后逐步升温至110℃搅拌1.5h。TLC监测至反应结束,浓缩反应液,残留物用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=1:6)得到标题化合物(R)-2-氯-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(8c)(白色固体,2.2g,产率68%)。Dissolve (R)-2-chloro-4-((tetrahydrofuran-3-yl)amino)pyrimidine-5-carboxylic acid 8b (2.9g, 11.9mmol) in dimethylacetamide (20mL) and add triethylamine (1.2g, 11.9mmol), diphenyl azide phosphate (3.3g, 11.9mmol), and then gradually heated to 110°C and stirred for 1.5h. The reaction was monitored by TLC and the reaction solution was concentrated. The residue was separated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)=1:6) to obtain the title compound (R)-2-chloro-9-(tetrahydrofuran) -3-yl)-7,9-dihydro-8H-purin-8-one (8c) (white solid, 2.2 g, yield 68%).
1H NMR(400MHz DMSO)δ8.36(s,1H),5.04–4.96(m,1H),4.13-4.07(dd,1H),3.97(t,1H),3.87-3.84(m,2H),3.33(s,3H),2.40-2.36(m,1H),2.29-2.27(m,1H)。1H NMR(400MHz DMSO)δ8.36(s,1H),5.04-4.96(m,1H),4.13-4.07(dd,1H),3.97(t,1H),3.87-3.84(m,2H),3.33 (s, 3H), 2.40-2.36 (m, 1H), 2.29-2.27 (m, 1H).
LC-MS m/z(ESI)=241.20[M+1]。LC-MS m/z(ESI)=241.20[M+1].
第四步:the fourth step:
(R)-2-氯-7-甲基-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(8d)(R)-2-chloro-7-methyl-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one (8d)
(R)-2-chloro-7-methyl-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one(R)-2-chloro-7-methyl-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one
将(R)-2-氯-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮8c(2.2g,9.1mmol)溶解于二甲基甲酰胺(10mL)中,在0℃下加入硫酸二甲酯(1.1g,9.1mmol)和碳酸铯(4.5g,13.7mmol)搅拌反应0.5h。TLC监测至反应结束,向反应液中加入水,有固体析出,过滤得到标题化合物(R)-2-氯-7-甲基-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(8d)(白色固体,1.7g,产率82%)。(R)-2-chloro-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one 8c (2.2g, 9.1mmol) was dissolved in dimethylformamide (10mL ), add dimethyl sulfate (1.1 g, 9.1 mmol) and cesium carbonate (4.5 g, 13.7 mmol) at 0° C. and stir and react for 0.5 h. TLC monitors to the end of the reaction, water is added to the reaction solution, a solid is precipitated, and the title compound (R)-2-chloro-7-methyl-9-(tetrahydrofuran-3-yl)-7,9-dihydro is obtained by filtration. -8H-purin-8-one (8d) (white solid, 1.7 g, yield 82%).
1H NMR(400MHz DMSO)δ8.41(s,1H),5.05–4.97(m,1H),4.13-4.08(dd,1H),3.98(t,1H),3.91-3.84(m,2H),3.33(s,3H),2.45-2.27(m,1H),2.26-2.20(m,1H)。1H NMR (400MHz DMSO) δ8.41 (s, 1H), 5.05-4.97 (m, 1H), 4.13-4.08 (dd, 1H), 3.98 (t, 1H), 3.91-3.84 (m, 2H), 3.33 (s, 3H), 2.45-2.27 (m, 1H), 2.26-2.20 (m, 1H).
LC-MS m/z(ESI)=255.20[M+1]。LC-MS m/z(ESI)=255.20[M+1].
第五步:the fifth step:
(R)-7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(化合物8)(R)-7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydrofuran-3-yl)-7,9- Dihydro-8H-purin-8-one (Compound 8)
(R)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-furan-3-yl)-7,9-dihydro-8H-purin-8-one(R)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-furan-3-yl)-7,9-dihydro-8H-purin -8-one
将(R)-2-氯-7-甲基-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮8d(100mg,0.39mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(117mg,0.79mmol)、碳酸铯(256mg,0.75mmol)和Brettphos G3 Pd(36mg,0.039mmol)加入干燥的反应瓶,氮气置换三次后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化得到标题化合物(R)-7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(化合物8)(浅黄色固体,60mg,产率41.59%)。Add (R)-2-chloro-7-methyl-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one 8d (100mg, 0.39mmol), 6-methyl -2,3-Dihydrobenzofuran-5-amine intermediate 1 (117mg, 0.79mmol), cesium carbonate (256mg, 0.75mmol) and Brettphos G3 Pd (36mg, 0.039mmol) were added to the dry reaction flask and replaced with nitrogen After three times, dry 1,4-dioxane (1 mL) was added and reacted at 110° C. for 5 h. The reaction was monitored by TLC until the end of the reaction. After cooling to room temperature, it was separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound (R)-7-methyl-2-(( 6-Methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one (compound 8) (Light yellow solid, 60 mg, yield 41.59%).
1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),7.98(s,1H),7.19(s,1H),6.61(s,1H),4.93-4.82(m,1H),4.49(t,2H),3.95-3.88(m,2H),3.81–3.75(m,2H),3.27(s,3H),3.11(t,2H),2.40-2.32(m,1H),2.18–2.12(m,1H),2.11(s,3H)。1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),7.98(s,1H),7.19(s,1H),6.61(s,1H),4.93-4.82(m,1H),4.49( t, 2H), 3.95-3.88(m, 2H), 3.81-3.75(m, 2H), 3.27(s, 3H), 3.11(t, 2H), 2.40-2.32(m, 1H), 2.18-2.12( m, 1H), 2.11 (s, 3H).
LC-MS m/z(ESI)=368.20[M+1]。LC-MS m/z(ESI)=368.20[M+1].
实施例9Example 9
7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(((四氢呋喃-3-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(化合物9)7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(((tetrahydrofuran-3-yl)methyl)-7,9 -Dihydro-8H-purin-8-one (compound 9)
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-((tetrahydrofuran-3-yl)methyl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-((tetrahydrofuran-3-yl)methyl)-7,9-dihydro-8H-purin-8- one
第一步:first step:
2-氯-4-((四氢呋喃-3-基)甲基)氨基)嘧啶-5-羧酸乙酯(9a)Ethyl 2-chloro-4-((tetrahydrofuran-3-yl)methyl)amino)pyrimidine-5-carboxylate (9a)
ethyl 2-chloro-4-(((tetrahydrofuran-3-yl)methyl)amino)pyrimidine-5-carboxylateethyl 2-chloro-4-(((tetrahydrofuran-3-yl)methyl)amino)pyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(6.2g,22.6mmol)、碳酸钾(6.2g,44.8mmol)溶解于乙腈(20mL),在0℃下加入(四氢呋喃-3-基)甲酰胺(2.3g,22.6mmol),在室温搅拌20h。加水和乙酸乙酯萃取三次,无水硫酸钠干燥并用硅胶柱色谱分离提纯(正己烷/乙酸乙酯(v/v)=10:1),浓缩得到标题化合物2-氯-4-((四氢呋喃-3-基)甲基)氨基)嘧啶-5-羧酸乙酯(9a)(白色固体,4.4g,产率85%)。Dissolve 2,4-dichloropyrimidine-5-carboxylic acid ethyl ester 1a (6.2g, 22.6mmol), potassium carbonate (6.2g, 44.8mmol) in acetonitrile (20mL), add (tetrahydrofuran-3- Methyl) formamide (2.3g, 22.6mmol), stirred at room temperature for 20h. Add water and ethyl acetate to extract three times, dry with anhydrous sodium sulfate and separate and purify by silica gel column chromatography (n-hexane/ethyl acetate (v/v)=10:1), and concentrate to obtain the title compound 2-chloro-4-((tetrahydrofuran) -3-yl)methyl)amino)pyrimidine-5-carboxylic acid ethyl ester (9a) (white solid, 4.4 g, yield 85%).
1H NMR(400MHz DMSO)δ8.58-8.61(m,2H),4.31(q,2H),3.78-3.73(m,1H),3.69-3.58(m,2H),3.48-3.44(m,3H),2.57-2.53(m,1H),1.98-1.91(m,1H),1.62-1.58(m,1H),1.31(t,3H)。1H NMR (400MHz DMSO) δ 8.58-8.61 (m, 2H), 4.31 (q, 2H), 3.78-3.73 (m, 1H), 3.69-3.58 (m, 2H), 3.48-3.44 (m, 3H) , 2.57-2.53 (m, 1H), 1.98-1.91 (m, 1H), 1.62-1.58 (m, 1H), 1.31 (t, 3H).
LC-MS m/z(ESI)=286.20[M+1]。LC-MS m/z(ESI)=286.20[M+1].
第二步:The second step:
2-氯-4-((四氢呋喃-3-基)甲基)氨基)嘧啶-5-羧酸(9b)2-Chloro-4-((tetrahydrofuran-3-yl)methyl)amino)pyrimidine-5-carboxylic acid (9b)
2-chloro-4-(((tetrahydrofuran-3-yl)methyl)amino)pyrimidine-5-carboxylate2-chloro-4-(((tetrahydrofuran-3-yl)methyl)amino)pyrimidine-5-carboxylate
将2-氯-4-((四氢呋喃-3-基)甲基)氨基)嘧啶-5-羧酸乙酯9a(4.4g,15.3mmol)溶解于四氢呋喃/水(10mL/5mL)中,加入氢氧化锂(736mg,30.6mmol),室温搅拌反应1h。TLC监测至反应结束,旋干四氢呋喃,调pH至4-5,有白色固体析出,过滤,滤饼用石油醚/乙酸乙酯(v/v=10/1)洗两次,浓缩得到标题化合物2-氯-4-((四氢呋喃-3-基)甲基)氨基)嘧啶-5-羧酸(9b)(白色固体,3.4g,产率80%)。Dissolve 2-chloro-4-((tetrahydrofuran-3-yl)methyl)amino)pyrimidine-5-carboxylic acid ethyl ester 9a (4.4g, 15.3mmol) in tetrahydrofuran/water (10mL/5mL), add hydrogen Lithium oxide (736 mg, 30.6 mmol) was stirred at room temperature for 1 h. TLC monitors to the end of the reaction, spin-dry the tetrahydrofuran, adjust the pH to 4-5, a white solid is precipitated, filtered, the filter cake is washed twice with petroleum ether/ethyl acetate (v/v=10/1), and concentrated to obtain the title compound 2-Chloro-4-((tetrahydrofuran-3-yl)methyl)amino)pyrimidine-5-carboxylic acid (9b) (white solid, 3.4 g, yield 80%).
1H NMR(400MHz DMSO)δ13.75(s,1H),8.75(s,1H),8.57(s,1H),3.78-3.73(m,3H),3.47-3.43(m,3H),2.58-2.52(m,1H),1.98-1.89(m,3H),1.63-1.55(m,1H)。1H NMR (400MHz DMSO) δ 13.75 (s, 1H), 8.75 (s, 1H), 8.57 (s, 1H), 3.78-3.73 (m, 3H), 3.47-3.43 (m, 3H), 2.58-2.52 (m, 1H), 1.98-1.89 (m, 3H), 1.63-1.55 (m, 1H).
LC-MS m/z(ESI)=259.20[M+1]。LC-MS m/z(ESI)=259.20[M+1].
第三步:third step:
2-氯-9-((四氢呋喃-3-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(9c)2-Chloro-9-((tetrahydrofuran-3-yl)methyl)-7,9-dihydro-8H-purin-8-one (9c)
2-chloro-9-((tetrahydrofuran-3-yl)methyl)-7,9-dihydro-8H-purin-8-one2-chloro-9-((tetrahydrofuran-3-yl)methyl)-7,9-dihydro-8H-purin-8-one
将2-氯-4-((四氢呋喃-3-基)甲基)氨基)嘧啶-5-羧酸9b(3.4g,13.0mmol)溶解于二甲基乙酰胺(20mL),加入三乙胺(1.58g,13.0mmol)、叠氮磷酸二苯酯(3.6g,13.0mmol),随后逐步升温至110℃搅拌1.5h。TLC监测至反应结束,浓缩反应液,残留物用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=1:10)得到标题化合物2-氯-9-((四氢呋喃-3-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(9c)(白色固体,1.46g,产率50%)。Dissolve 2-chloro-4-((tetrahydrofuran-3-yl)methyl)amino)pyrimidine-5-carboxylic acid 9b (3.4g, 13.0mmol) in dimethylacetamide (20mL), add triethylamine ( 1.58g, 13.0mmol), diphenyl azide phosphate (3.6g, 13.0mmol), and then gradually heated to 110°C and stirred for 1.5h. The reaction was monitored by TLC until the end of the reaction. The reaction solution was concentrated. The residue was separated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)=1:10) to obtain the title compound 2-chloro-9-((tetrahydrofuran-3- (Yl)methyl)-7,9-dihydro-8H-purin-8-one (9c) (white solid, 1.46 g, yield 50%).
1H NMR(400MHz DMSO)δ11.65(s,1H),8.14(s,1H),3.80-3.74(m,3H),3.66-3.57(m,2H),3.52-3.49(m,1H),2.73-2.69(m,1H),1.98-1.89(m,1H),1.67-1.60(m,1H)。1H NMR (400MHz DMSO) δ 11.65 (s, 1H), 8.14 (s, 1H), 3.80-3.74 (m, 3H), 3.66-3.57 (m, 2H), 3.52-3.49 (m, 1H), 2.73 -2.69 (m, 1H), 1.98-1.89 (m, 1H), 1.67-1.60 (m, 1H).
LC-MS m/z(ESI)=255.20[M+1]。LC-MS m/z(ESI)=255.20[M+1].
第四步:the fourth step:
2-氯-7-甲基-9-((四氢呋喃-3-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(9d)2-Chloro-7-methyl-9-((tetrahydrofuran-3-yl)methyl)-7,9-dihydro-8H-purin-8-one (9d)
2-chloro-7-methyl-9-((tetrahydrofuran-3-yl)methyl)-7,9-dihydro-8H-purin-8-one2-chloro-7-methyl-9-((tetrahydrofuran-3-yl)methyl)-7,9-dihydro-8H-purin-8-one
将2-氯-9-((四氢呋喃-3-基)甲基)-7,9-二氢-8H-嘌呤-8-酮9c(1.6g,5.2mmol)溶解于二甲基甲酰胺(10mL)中,在0℃下加入硫酸二甲酯(663mg,5.2mmol)和碳酸铯(2.4g,7.7mmol)搅拌反应1h。TLC监测至反应结束,向反应液中加入水,有固体析出,过滤得到标题化合物2-氯-7-甲基-9-((四氢呋喃-3-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(9d)(白色固体,1.2g,产率80%)。Dissolve 2-chloro-9-((tetrahydrofuran-3-yl)methyl)-7,9-dihydro-8H-purin-8-one 9c (1.6g, 5.2mmol) in dimethylformamide (10mL ), dimethyl sulfate (663 mg, 5.2 mmol) and cesium carbonate (2.4 g, 7.7 mmol) were added at 0° C. and the reaction was stirred for 1 h. TLC monitors to the end of the reaction, water is added to the reaction solution, a solid is precipitated, and the title compound 2-chloro-7-methyl-9-((tetrahydrofuran-3-yl)methyl)-7,9-dihydro is obtained by filtration. -8H-purin-8-one (9d) (white solid, 1.2 g, yield 80%).
1H NMR(400MHz DMSO)δ8.35(s,1H),3.81-3.75(m,3H),3.65-3.57(m,2H),3.52-3.49(m,1H),3.37(s,3H),2.73-2.70(m,1H),1.96-1.91(m,1H),1.67-1.61(m,1H)。1H NMR(400MHz DMSO)δ8.35(s,1H),3.81-3.75(m,3H),3.65-3.57(m,2H),3.52-3.49(m,1H),3.37(s,3H),2.73 -2.70 (m, 1H), 1.96-1.91 (m, 1H), 1.67-1.61 (m, 1H).
LC-MS m/z(ESI)=268.20[M+1]。LC-MS m/z(ESI)=268.20[M+1].
第五步:the fifth step:
7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(((四氢呋喃-3-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(化合物9)7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(((tetrahydrofuran-3-yl)methyl)-7,9 -Dihydro-8H-purin-8-one (compound 9)
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-((tetrahydrofuran-3-yl)methyl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-((tetrahydrofuran-3-yl)methyl)-7,9-dihydro-8H-purin-8- one
将2-氯-7-甲基-9-((四氢呋喃-3-基)甲基)-7,9-二氢-8H-嘌呤-8-酮9d(100mg,0.37mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(112mg,0.74mmol)、碳酸铯(242mg,0.74mmol)和Brettphos G3 Pd(34mg,0.037mmol)加入干燥的反应瓶,氮气置换三次后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化得到标题化合物7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(((四氢呋喃-3-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(化合物9)(灰白色固体,22mg,产率15.50%)。The 2-chloro-7-methyl-9-((tetrahydrofuran-3-yl)methyl)-7,9-dihydro-8H-purin-8-one 9d (100mg, 0.37mmol), 6-methyl -2,3-Dihydrobenzofuran-5-amine intermediate 1 (112mg, 0.74mmol), cesium carbonate (242mg, 0.74mmol) and Brettphos G3 Pd (34mg, 0.037mmol) were added to the dry reaction flask and replaced with nitrogen After three times, dry 1,4-dioxane (1 mL) was added and reacted at 110° C. for 5 h. The reaction was monitored by TLC until the end of the reaction. After cooling to room temperature, it was separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 7-methyl-2-(((6-methyl -2,3-Dihydrobenzofuran-5-yl)amino)-9-(((tetrahydrofuran-3-yl)methyl)-7,9-dihydro-8H-purin-8-one (compound 9 ) (Off-white solid, 22 mg, yield 15.50%).
1H NMR(400MHz,DMSO-d6)δ8.25(s,1H),7.95(s,1H),7.18(s,1H),6.60(s,1H),4.49(t,2H),4.24(p,1H),3.81-3.72(m,2H),3.66-3.57(m,2H),3.28(s,3H),3.11(t,2H),2.10(s,3H),1.93-1.77(m,3H),1.70-1.60(m,1H)。1H NMR (400MHz, DMSO-d6) δ 8.25 (s, 1H), 7.95 (s, 1H), 7.18 (s, 1H), 6.60 (s, 1H), 4.49 (t, 2H), 4.24 (p, 1H), 3.81-3.72 (m, 2H), 3.66-3.57 (m, 2H), 3.28 (s, 3H), 3.11 (t, 2H), 2.10 (s, 3H), 1.93-1.77 (m, 3H) ,1.70-1.60(m,1H).
LC-MS m/z(ESI)=382.20[M+1]。LC-MS m/z(ESI)=382.20[M+1].
实施例10Example 10
7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(哌啶-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物10)7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(piperidin-4-yl)-7,9-dihydro-8H -Purin-8-one (Compound 10)
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(piperidin-4-yl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(piperidin-4-yl)-7,9-dihydro-8H-purin-8-one
第一步:first step:
4-((1-(叔丁氧基羰基)哌啶-4-基)氨基)-2-氯嘧啶-5-羧酸乙酯(10a)4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester (10a)
ethyl 4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxylateethyl 4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(5g,22.6mmol)、碳酸钾(6.2g,45.2mmol)溶解于乙腈(20mL),在0℃下加入4-氨基哌啶-1-羧酸叔丁酯(4.5g,22.6mmol),在室温搅拌20h,TLC监测至反应完全,加水和乙酸乙酯萃取三次,无水硫酸钠干燥后用硅胶柱色谱分离提纯(正己烷/乙酸乙酯(v/v)=10:)),浓缩得到标题化合物4-((1-(叔丁氧基羰基)哌啶-4-基)氨基)-2-氯嘧啶-5-羧酸乙酯(10a)(白色固体,8.2g,产率95%)。Dissolve 2,4-dichloropyrimidine-5-carboxylic acid ethyl ester 1a (5g, 22.6mmol), potassium carbonate (6.2g, 45.2mmol) in acetonitrile (20mL), add 4-aminopiperidine- Tert-Butyl 1-carboxylate (4.5g, 22.6mmol), stirred at room temperature for 20h, TLC monitored until the reaction was complete, added water and ethyl acetate for three times, dried over anhydrous sodium sulfate and purified by silica gel column chromatography (n-hexane/ Ethyl acetate (v/v)=10:)), and concentrated to obtain the title compound 4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxylic acid Ethyl ester (10a) (white solid, 8.2 g, yield 95%).
1H NMR(400MHz DMSO)δ8.63(s,1H),8.32(d,1H),4.33-4.28(m,2H),4.17-4.14(m,1H),3.85(d,2H),2.94(s,2H),1.88-1.80(m,2H),1.51-1.44(m,2H),1.41(s,9H),1.32-1.29(m,3H)。1H NMR(400MHz DMSO) δ8.63(s,1H), 8.32(d,1H), 4.33-4.28(m,2H), 4.17-4.14(m,1H), 3.85(d,2H), 2.94(s , 2H), 1.88-1.80 (m, 2H), 1.51-1.44 (m, 2H), 1.41 (s, 9H), 1.32-1.29 (m, 3H).
LC-MS m/z(ESI)=385.10[M+1]。LC-MS m/z(ESI)=385.10[M+1].
第二步:The second step:
4-((1-(叔丁氧基羰基)哌啶-4-基)氨基)-2-氯嘧啶-5-羧酸(10b)4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxylic acid (10b)
4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxy-lic acid4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxy-lic acid
将4-((1-(叔丁氧基羰基)哌啶-4-基)氨基)-2-氯嘧啶-5-羧酸乙酯10a(8.2g,21.3mmol)溶解于四氢呋喃/水(10mL/5mL)中,加入氢氧化锂(1.8g,42.7mmol),室温搅拌1h。TLC监测至反应完全,浓缩蒸发掉四氢呋喃,调pH至4-5,有白色固体析出,过滤,滤饼用石油醚/乙酸乙酯(v/v=10/1)洗两次,浓缩得到标题化合物4-((1-(叔丁氧基羰基)哌啶-4-基)氨基)-2-氯嘧啶-5-羧酸(10b)(白色固体,7g,产率86%)。Dissolve 4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester 10a (8.2g, 21.3mmol) in tetrahydrofuran/water (10mL /5mL), lithium hydroxide (1.8g, 42.7mmol) was added, and the mixture was stirred at room temperature for 1h. TLC monitors the reaction until the reaction is complete, concentrates and evaporates the tetrahydrofuran, adjusts the pH to 4-5, and a white solid precipitates out, filtered, the filter cake was washed twice with petroleum ether/ethyl acetate (v/v=10/1), and concentrated to obtain the title Compound 4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxylic acid (10b) (white solid, 7g, yield 86%).
1H NMR(400MHz DMSO)δ8.59(s,1H),8.54(d,1H),4.20-4.10(m,1H),3.87-3.84(m,2H),2.96(s,3H),1.91-1.73(m,3H),1.41(s,9H)。1H NMR(400MHz DMSO)δ8.59(s,1H), 8.54(d,1H), 4.20-4.10(m,1H), 3.87-3.84(m,2H), 2.96(s,3H), 1.91-1.73 (m, 3H), 1.41 (s, 9H).
LC-MS m/z(ESI)=357.10[M+1]。LC-MS m/z(ESI)=357.10[M+1].
第三步third step
4-(2-氯-8-氧代-7,8-二氢-9H-嘌呤-9-基)哌啶-1-甲酸叔丁酯(10c)Tert-Butyl 4-(2-chloro-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine-1-carboxylate (10c)
tert-butyl 4-(2-chloro-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine-1-carboxylatetert-butyl 4-(2-chloro-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine-1-carboxylate
将4-((1-(叔丁氧基羰基)哌啶-4-基)氨基)-2-氯嘧啶-5-羧酸10b(7g,19.6mmol)溶解于二甲基乙酰胺(10mL),加入三乙胺(1.96g,19.6mmol)、叠氮磷酸二苯酯(5.4g,19.6mmol),随后逐步升温至110℃搅拌1.5h,TLC监测至反应完全,浓缩反应液,残留物用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=1:10),得到标题化合物4-(2-氯-8-氧代-7,8-二氢-9H-嘌呤-9-基)哌啶-1-甲酸叔丁酯(10c)(白色固体,6.4g,产率87%)。Dissolve 4-((1-(tert-butoxycarbonyl)piperidin-4-yl)amino)-2-chloropyrimidine-5-carboxylic acid 10b (7g, 19.6mmol) in dimethylacetamide (10mL) , Add triethylamine (1.96g, 19.6mmol) and diphenyl azide phosphate (5.4g, 19.6mmol), then gradually increase the temperature to 110°C and stir for 1.5h. TLC monitors until the reaction is complete. The reaction solution is concentrated and the residue is used Separation and purification by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)=1:10) gave the title compound 4-(2-chloro-8-oxo-7,8-dihydro-9H-purine-9) -Yl)piperidine-1-carboxylic acid tert-butyl ester (10c) (white solid, 6.4 g, yield 87%).
1H NMR(400MHz DMSO)δ8.14(s,1H),4.41-4.33(m,1H),4.06(d,2H),2.88(s,2H), 2.30-2.20(m,2H),1.84-1.71(m,2H),1.43(s,9H)。1H NMR(400MHz DMSO)δ8.14(s,1H),4.41-4.33(m,1H),4.06(d,2H),2.88(s,2H), 2.30-2.20(m,2H),1.84-1.71 (m, 2H), 1.43 (s, 9H).
LC-MS m/z(ESI)=354.10[M+1]。LC-MS m/z(ESI)=354.10[M+1].
第四步:the fourth step:
4-(2-氯-7-甲基-8-氧代-7,8-二氢-9H-嘌呤-9-基)哌啶-1-甲酸叔丁酯(10d)Tert-Butyl 4-(2-chloro-7-methyl-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine-1-carboxylate (10d)
tert-butyl4-(2-chloro-7-methyl-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine-1-carboxylatetert-butyl4-(2-chloro-7-methyl-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine-1-carboxylate
将4-(2-氯-8-氧代-7,8-二氢-9H-嘌呤-9-基)哌啶-1-甲酸叔丁酯10c(6.4g,18.1mmol)溶解于二甲基甲酰胺(10mL),在0℃下加入硫酸二甲酯(2.28g,18.1mmol)和碳酸铯(8.5g,27.1mmol),0℃搅拌0.5h。TLC监测至反应完全,向反应液中加入水,有固体析出,过滤得到标题化合物4-(2-氯-7-甲基-8-氧代-7,8-二氢-9H-嘌呤-9-基)哌啶-1-甲酸叔丁酯(10d)(白色固体,5.4g,产率79%)。Dissolve tert-butyl 4-(2-chloro-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine-1-carboxylate 10c (6.4g, 18.1mmol) in dimethyl Formamide (10 mL), dimethyl sulfate (2.28 g, 18.1 mmol) and cesium carbonate (8.5 g, 27.1 mmol) were added at 0°C, and stirred at 0°C for 0.5 h. TLC monitors until the reaction is complete, water is added to the reaction solution, a solid is precipitated, and the title compound 4-(2-chloro-7-methyl-8-oxo-7,8-dihydro-9H-purine-9 is obtained by filtration. -Yl)piperidine-1-carboxylic acid tert-butyl ester (10d) (white solid, 5.4 g, yield 79%).
1H NMR(400MHz DMSO)δ8.36(s,1H),4.46-4.37(m,1H),4.05(d,2H),3.35(s,3H),2.87(s,2H),2.33-2.19(m,2H),1.74-1.72(m,2H),1.43(s,9H)。1H NMR (400MHz DMSO) δ 8.36 (s, 1H), 4.46-4.37 (m, 1H), 4.05 (d, 2H), 3.35 (s, 3H), 2.87 (s, 2H), 2.33-2.19 (m , 2H), 1.74-1.72 (m, 2H), 1.43 (s, 9H).
LC-MS m/z(ESI)=368.10[M+1]。LC-MS m/z(ESI)=368.10[M+1].
第五步:the fifth step:
4-(7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-8-氧代-7,8-二氢-9H-嘌呤-9-基)哌啶-1-羧酸叔丁酯(10e)4-(7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-8-oxo-7,8-dihydro-9H-purine- 9-yl) piperidine-1-carboxylic acid tert-butyl ester (10e)
tert-butyl 4-(7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine-1-carboxylatetert-butyl 4-(7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine -1-carboxylate
将4-(2-氯-7-甲基-8-氧代-7,8-二氢-9H-嘌呤-9-基)哌啶-1-甲酸叔丁酯10d(200mg,0.54mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(161mg,1.08mmol)、碳酸铯(381mg,1.08mmol)和Brettphos G3 Pd(49mg,0.054mmol)加入干燥的反应瓶,氮气置换三次后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后使用硅藻土过滤,滤液浓缩得到粗品4-(7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-8-氧代-7,8-二氢-9H-嘌呤-9-基)哌啶-1-羧酸叔丁酯(10e),未经纯化直接投下一步。The 4-(2-chloro-7-methyl-8-oxo-7,8-dihydro-9H-purin-9-yl) piperidine-1-carboxylic acid tert-butyl ester 10d (200mg, 0.54mmol), 6-Methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (161mg, 1.08mmol), cesium carbonate (381mg, 1.08mmol) and Brettphos G3 Pd (49mg, 0.054mmol) were added to the dry reaction After replacing the flask with nitrogen three times, add dry 1,4-dioxane (1 mL), and react at 110°C for 5 hours. The reaction was monitored by TLC until the end of the reaction. After cooling to room temperature, it was filtered with celite. The filtrate was concentrated to obtain crude 4-(7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl) )Amino)-8-oxo-7,8-dihydro-9H-purin-9-yl)piperidine-1-carboxylic acid tert-butyl ester (10e), directly cast to the next step without purification.
第六步:The sixth step:
7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(哌啶-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物10)7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(piperidin-4-yl)-7,9-dihydro-8H -Purin-8-one (Compound 10)
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(piperidin-4-yl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(piperidin-4-yl)-7,9-dihydro-8H-purin-8-one
将粗品4-(7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-8-氧代-7,8-二氢-9H-嘌呤-9-基)哌啶-1-羧酸叔丁酯10e用二氯甲烷(2mL)溶解,再加入三氟乙酸(0.19mL,2.5mmol),室温反应1h。TLC监测至反应结束,用饱和碳酸氢钠溶液调节pH至8-9,加入二氯甲烷萃取三次,浓缩有机相,使用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v)=40:1))纯化得到标题化合物7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(哌啶-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物10)(白色固体,40mg,产率19.38%)。The crude 4-(7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-8-oxo-7,8-dihydro-9H- Purin-9-yl)piperidine-1-carboxylic acid tert-butyl ester 10e was dissolved in dichloromethane (2 mL), and then trifluoroacetic acid (0.19 mL, 2.5 mmol) was added, and the reaction was carried out at room temperature for 1 h. TLC monitors to the end of the reaction, adjust the pH to 8-9 with saturated sodium bicarbonate solution, add dichloromethane for extraction three times, concentrate the organic phase, and use silica gel column chromatography to separate and purify (dichloromethane/methanol (v/v)=40: 1)) Purified to obtain the title compound 7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(piperidin-4-yl)-7 , 9-dihydro-8H-purin-8-one (compound 10) (white solid, 40 mg, yield 19.38%).
1H NMR(400MHz,DMSO-d6)δ8.18(s,1H),7.93(s,1H),7.24(s,1H),6.60(s,1H),4.48(t,2H),4.22–4.12(m,1H),3.26(s,3H),3.12(t,2H),3.03(d,2H),2.55-2.48(m,2H),2.38–2.27(m,2H),2.12(s,3H),1.60(d,2H)。1H NMR(400MHz,DMSO-d6)δ8.18(s,1H),7.93(s,1H),7.24(s,1H),6.60(s,1H),4.48(t,2H),4.22-4.12( m, 1H), 3.26 (s, 3H), 3.12 (t, 2H), 3.03 (d, 2H), 2.55-2.48 (m, 2H), 2.38-2.27 (m, 2H), 2.12 (s, 3H) ,1.60(d,2H).
LC-MS m/z(ESI)=381.20[M+1]。LC-MS m/z(ESI)=381.20[M+1].
实施例11Example 11
9-(顺-4-羟基-4-甲基环己基)-7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物11)9-(cis-4-hydroxy-4-methylcyclohexyl)-7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7 ,9-Dihydro-8H-purin-8-one (Compound 11)
9-(cis-4-hydroxy-4-methylcyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(cis-4-hydroxy-4-methylcyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8 -one
第一步:first step:
2-氯-4-((顺-4-羟基-4-甲基环己基)氨基)嘧啶-5-羧酸乙酯(11a)Ethyl 2-chloro-4-((cis-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylate (11a)
ethyl 2-chloro-4-((cis-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylateethyl 2-chloro-4-((cis-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(5.0g,22.6mmol)、碳酸钾(6.2g,45.2mmol)溶解于乙腈(50mL),在0℃下加入(1s,4s)-4-氨基-1-甲基环己-1-醇(2.9g,22.6mmol),在室温搅拌20h。加水和乙酸乙酯萃取三次,无水硫酸钠干燥并用硅胶柱色谱分离提纯(正己烷/乙酸乙酯(v/v)=10:1),浓缩得到标题化合物2-氯-4-((顺-4-羟基-4-甲基环己基)氨基)嘧啶-5-羧酸乙酯(11a)(白色固体,4.1g,产率58%)。Dissolve 2,4-dichloropyrimidine-5-carboxylic acid ethyl ester 1a (5.0g, 22.6mmol) and potassium carbonate (6.2g, 45.2mmol) in acetonitrile (50mL), add (1s, 4s) at 0℃ -4-amino-1-methylcyclohexan-1-ol (2.9g, 22.6mmol), stirred at room temperature for 20h. Add water and ethyl acetate to extract three times, dry with anhydrous sodium sulfate and separate and purify by silica gel column chromatography (n-hexane/ethyl acetate (v/v) = 10:1), and concentrate to obtain the title compound 2-chloro-4-((cis Ethyl 4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylate (11a) (white solid, 4.1 g, yield 58%).
1H NMR(400MHz,Chloroform-d)δ8.65(s,1H),8.37(d,1H),4.35(q,2H),4.09–4.04(m,1H),1.94-1.82(m,2H),1.82-1.45(m,6H),1.38(t,3H),1.28(s,3H)。1H NMR(400MHz,Chloroform-d)δ8.65(s,1H),8.37(d,1H),4.35(q,2H),4.09-4.04(m,1H),1.94-1.82(m,2H), 1.82-1.45 (m, 6H), 1.38 (t, 3H), 1.28 (s, 3H).
LC-MS m/z(ESI)=314.10[M+1]。LC-MS m/z(ESI)=314.10[M+1].
第二步:The second step:
2-氯-4-((顺-4-羟基-4-甲基环己基)氨基)嘧啶-5-羧酸(13b)2-chloro-4-((cis-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylic acid (13b)
2-chloro-4-((cis-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylate2-chloro-4-((cis-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylate
将2-氯-4-((顺-4-羟基-4-甲基环己基)氨基)嘧啶-5-羧酸乙酯11a(4.1g,13.1mmol)溶解于四氢呋喃/水(20mL/20mL)中,加入氢氧化锂(629mg,26.2mmol),室温搅拌反应1h。TLC监测至反应结束,旋干四氢呋喃,调pH至4-5,有白色固体析出,过滤,滤饼用石油醚/乙酸乙酯(v/v=10/1)洗两次,浓缩得到标题化合物2-氯-4-((顺-4-羟基-4-甲基环己基)氨基)嘧啶-5-羧酸(11b)(白色固体,3.5g,产率93%)。Dissolve 2-chloro-4-((cis-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylic acid ethyl ester 11a (4.1g, 13.1mmol) in tetrahydrofuran/water (20mL/20mL) Lithium hydroxide (629 mg, 26.2 mmol) was added to the mixture, and the reaction was stirred at room temperature for 1 h. TLC monitors to the end of the reaction, spin-dry the tetrahydrofuran, adjust the pH to 4-5, a white solid is precipitated, filtered, the filter cake is washed twice with petroleum ether/ethyl acetate (v/v=10/1), and concentrated to obtain the title compound 2-Chloro-4-((cis-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylic acid (11b) (white solid, 3.5 g, yield 93%).
1H NMR(400MHz,DMSO-d6)δ13.77(s,1H),8.56(s,1H),8.49(d,1H),3.93–3.83(m,2H),1.78-1.50(m,6H),1.41(td,2H),1.11(s,3H)。1H NMR(400MHz,DMSO-d6)δ13.77(s,1H),8.56(s,1H),8.49(d,1H),3.93-3.83(m,2H),1.78-1.50(m,6H), 1.41 (td, 2H), 1.11 (s, 3H).
LC-MS m/z(ESI)=286.10[M+1]。LC-MS m/z(ESI)=286.10[M+1].
第三步:third step:
2-氯-9-顺-4-羟基-4-甲基环己基)-7,9-二氢-8H-嘌呤-8-酮(11c)2-chloro-9-cis-4-hydroxy-4-methylcyclohexyl)-7,9-dihydro-8H-purin-8-one (11c)
2-chloro-9-(cis-4-hydroxy-4-methylcyclohexyl)-7,9-dihydro-8H-purin-8-one2-chloro-9-(cis-4-hydroxy-4-methylcyclohexyl)-7,9-dihydro-8H-purin-8-one
将2-氯-4-((顺-4-羟基-4-甲基环己基)氨基)嘧啶-5-羧酸11b(3.5g,12.2mmol)溶解于二甲基乙酰胺(30mL),加入三乙胺(1.24g,12.2mmol)、叠氮磷酸二苯酯(3.36g,12.2mmol),随后逐步升温至110℃搅拌1.5h。TLC监测至反应结束,浓缩反应液,残留物 用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=1:10)得到标题化合物2-氯-9-顺-4-羟基-4-甲基环己基)-7,9-二氢-8H-嘌呤-8-酮(11c)(白色固体,1.4g,产率41%)。Dissolve 2-chloro-4-((cis-4-hydroxy-4-methylcyclohexyl)amino)pyrimidine-5-carboxylic acid 11b (3.5g, 12.2mmol) in dimethylacetamide (30mL) and add Triethylamine (1.24g, 12.2mmol), diphenyl azide phosphate (3.36g, 12.2mmol), then gradually heated to 110°C and stirred for 1.5h. The reaction was monitored by TLC until the end of the reaction. The reaction solution was concentrated. The residue was separated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)=1:10) to obtain the title compound 2-chloro-9-cis-4-hydroxy- 4-methylcyclohexyl)-7,9-dihydro-8H-purin-8-one (11c) (white solid, 1.4g, yield 41%).
1H NMR(400MHz,DMSO-d6)δ11.58(s,1H),8.11(s,1H),4.22(s,1H),4.22–4.02(m,1H),2.65-2.56(m,2H),1.83-1.60(m,2H),1.47–1.43(m,4H),1.18(s,3H)。1H NMR(400MHz,DMSO-d6)δ11.58(s,1H),8.11(s,1H),4.22(s,1H),4.22-4.02(m,1H),2.65-2.56(m,2H), 1.83-1.60 (m, 2H), 1.47-1.43 (m, 4H), 1.18 (s, 3H).
LC-MS m/z(ESI)=283.10[M+1]。LC-MS m/z(ESI)=283.10[M+1].
第四步:the fourth step:
2-氯-9-(顺-4-羟基-4-甲基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(11d)2-Chloro-9-(cis-4-hydroxy-4-methylcyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one (11d)
2-chloro-9-(cis-4-hydroxy-4-methylcyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one2-chloro-9-(cis-4-hydroxy-4-methylcyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one
将2-氯-9-顺-4-羟基-4-甲基环己基)-7,9-二氢-8H-嘌呤-8-酮11c(1.4g,4.9mmol)溶解于二甲基甲酰胺(15mL),在0℃下加入硫酸二甲酯(618mg,4.9mmol)和碳酸铯(3.2g,9.8mmol)搅拌反应0.5h。TLC监测至反应结束,向反应液中加入水,有固体析出,过滤得到标题化合物2-氯-9-顺-4-羟基-4-甲基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(11d)(黄色固体,502mg,产率34%)。Dissolve 2-chloro-9-cis-4-hydroxy-4-methylcyclohexyl)-7,9-dihydro-8H-purin-8-one 11c (1.4g, 4.9mmol) in dimethylformamide (15mL), add dimethyl sulfate (618mg, 4.9mmol) and cesium carbonate (3.2g, 9.8mmol) at 0°C and stir and react for 0.5h. TLC monitored to the end of the reaction, water was added to the reaction solution, a solid precipitated out, filtered to obtain the title compound 2-chloro-9-cis-4-hydroxy-4-methylcyclohexyl)-7-methyl-7,9- Dihydro-8H-purin-8-one (11d) (yellow solid, 502 mg, yield 34%).
LC-MS m/z(ESI)=297.10[M+1]。LC-MS m/z(ESI)=297.10[M+1].
第五步:the fifth step:
9-(顺-4-羟基-4-甲基环己基)-7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物11)9-(cis-4-hydroxy-4-methylcyclohexyl)-7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7 ,9-Dihydro-8H-purin-8-one (Compound 11)
9-(cis-4-hydroxy-4-methylcyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(cis-4-hydroxy-4-methylcyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8 -one
将2-氯-9-顺-4-羟基-4-甲基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮11d(80mg,0.27mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(80mg,0.54mmol)、碳酸铯(176mg,0.54mmol)和Brettphos G3 Pd(24mg,0.027mmol)加入干燥的反应瓶,氮气置换三次后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化得到标题化合物9-(顺-4-羟基-4-甲基环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物11)(灰白色固体,46mg,产率41.67%)。The 2-chloro-9-cis-4-hydroxy-4-methylcyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one 11d (80mg, 0.27mmol), 6- Methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (80mg, 0.54mmol), cesium carbonate (176mg, 0.54mmol) and Brettphos G3 Pd (24mg, 0.027mmol) were added to the dry reaction flask, After nitrogen replacement three times, dry 1,4-dioxane (1 mL) was added, and the reaction was carried out at 110° C. for 5 h. The reaction was monitored by TLC until the reaction was over. After cooling to room temperature, it was separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 9-(cis-4-hydroxy-4-methyl ring Hexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one (compound 11) (Off-white solid, 46 mg, yield 41.67%).
1H NMR(400MHz,DMSO-d6)δ8.09(s,1H),7.92(s,1H),7.27(s,1H),6.59(s,1H),4.48(t,2H),4.14–4.04(m,1H),4.02(s,1H),3.25(s,3H),3.13(t,2H),2.63(q,2H),2.12(s,3H),1.66(d,2H),1.46–1.34(m,4H),1.14(s,3H)。1H NMR(400MHz,DMSO-d6)δ8.09(s,1H),7.92(s,1H),7.27(s,1H),6.59(s,1H),4.48(t,2H),4.14-4.04( m, 1H), 4.02 (s, 1H), 3.25 (s, 3H), 3.13 (t, 2H), 2.63 (q, 2H), 2.12 (s, 3H), 1.66 (d, 2H), 1.46-1.34 (m, 4H), 1.14 (s, 3H).
LC-MS m/z(ESI)=410.20[M+1]。LC-MS m/z(ESI)=410.20[M+1].
实施例12Example 12
9-(顺-4-甲氧基环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物12)9-(cis-4-methoxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-di Hydrogen-8H-purin-8-one (Compound 12)
9-(cis-4-methoxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(cis-4-methoxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one
第一步:first step:
2-氯-4-(顺-4-甲氧基环己基)氨基)嘧啶-5-羧酸乙酯(12a)Ethyl 2-chloro-4-(cis-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylate (12a)
ethyl 2-chloro-4-((cis-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylateethyl 2-chloro-4-((cis-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylate
将2,4-二氯-5-嘧啶甲酸乙酯1a(5.13g,23.22mmol)溶于乙腈(20mL)中,0℃搅拌下加入碳酸钾(6.42g,46.44mmol),再缓慢滴加(1s,4s)-4-甲氧基环己烷-1-胺(2.00g,15.48mmol)的乙腈溶液(10mL)升温至常温搅拌反应1h,TLC监测至反应结束,硅藻土过滤后用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=2:1)纯化得标题化合物2-氯-4-((1s,4s)-4-甲氧基环己基)氨基)嘧啶-5-羧酸乙酯(12a)(白色固体,2.00g,产率41.26%)。Ethyl 2,4-dichloro-5-pyrimidinecarboxylate 1a (5.13g, 23.22mmol) was dissolved in acetonitrile (20mL), potassium carbonate (6.42g, 46.44mmol) was added with stirring at 0°C, and then slowly added dropwise ( 1s,4s)-4-methoxycyclohexane-1-amine (2.00g, 15.48mmol) in acetonitrile (10mL) was heated to room temperature and stirred for 1h, monitored by TLC until the reaction was over, filtered through diatomaceous earth and filtered with silica gel Column chromatography separation and purification (petroleum ether/ethyl acetate (v/v)=2:1) was purified to obtain the title compound 2-chloro-4-((1s,4s)-4-methoxycyclohexyl)amino)pyrimidine- Ethyl 5-carboxylate (12a) (white solid, 2.00 g, yield 41.26%).
LC-MS m/z(ESI)=314.10[M+1]。LC-MS m/z(ESI)=314.10[M+1].
第二步:The second step:
2-氯-4-(顺-4-甲氧基环己基)氨基)嘧啶-5-羧酸(12b)2-Chloro-4-(cis-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylic acid (12b)
2-chloro-4-((cis-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylate2-chloro-4-((cis-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylate
将2-氯-4-((顺-4-甲氧基环己基)氨基)嘧啶-5-羧酸乙酯12a(2.00g,6.37mmol)溶解于四氢呋喃/水(15mL/15mL)中,加入氢氧化锂一水合物(0.80g,19.12mmol),常温搅拌反应2h,TLC监测至反应结束,浓缩蒸发掉四氢呋喃后加入2N HCl调节pH至3-4左右,有白色固体析出,过滤,滤饼用水以及石油醚/乙酸乙酯(v/v=10/1)洗两次得标题化合物2-氯-4-((顺-4-甲氧基环己基)氨基)嘧啶-5-羧酸(12b)(白色固体,1.67g,产率91.70%)。Dissolve 2-chloro-4-((cis-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylic acid ethyl ester 12a (2.00g, 6.37mmol) in tetrahydrofuran/water (15mL/15mL) and add Lithium hydroxide monohydrate (0.80g, 19.12mmol), stirred at room temperature for 2h, monitored by TLC until the end of the reaction, concentrated and evaporated off the tetrahydrofuran, then added 2N HCl to adjust the pH to about 3-4, a white solid precipitated, filtered, filter cake Wash twice with water and petroleum ether/ethyl acetate (v/v=10/1) to obtain the title compound 2-chloro-4-((cis-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylic acid ( 12b) (white solid, 1.67 g, yield 91.70%).
LC-MS m/z(ESI)=286.10[M+1]。LC-MS m/z(ESI)=286.10[M+1].
第三步:third step:
2-氯-9-(顺-4-甲氧基环己基)-7,9-二氢-8H-嘌呤-8-酮(12c)2-Chloro-9-(cis-4-methoxycyclohexyl)-7,9-dihydro-8H-purin-8-one (12c)
2-chloro-9-(cis-4-methoxycyclohexyl)-7,9-dihydro-8H-purin-8-one2-chloro-9-(cis-4-methoxycyclohexyl)-7,9-dihydro-8H-purin-8-one
将2-氯-4-((顺-4-甲氧基环己基)氨基)嘧啶-5-羧酸12b(1.57g,5.49mmol)用N,N-二甲基乙酰胺(16mL)溶解,常温搅拌下加入三乙胺(0.76mL,5.49mmol)和叠氮磷酸二苯酯(1.18mL,5.49mmol)反应2h后,升温至110℃回流反应2.5h。TLC监测至反应结束,反应液中加水二氯甲烷进行萃取,浓缩有机层得标题化合物2-氯-9-(顺-4-甲氧基环己基)-7,9-二氢-8H-嘌呤-8-酮(12c)(白色固体,1.70g,粗品,产率109.47%)。Dissolve 2-chloro-4-((cis-4-methoxycyclohexyl)amino)pyrimidine-5-carboxylic acid 12b (1.57g, 5.49mmol) with N,N-dimethylacetamide (16mL), Triethylamine (0.76 mL, 5.49 mmol) and diphenyl azide phosphate (1.18 mL, 5.49 mmol) were added under stirring at room temperature and reacted for 2 hours, then the temperature was raised to 110° C. and the reaction was refluxed for 2.5 hours. TLC monitors to the end of the reaction, the reaction solution is extracted with water and dichloromethane, and the organic layer is concentrated to obtain the title compound 2-chloro-9-(cis-4-methoxycyclohexyl)-7,9-dihydro-8H-purine -8-one (12c) (white solid, 1.70 g, crude product, yield 109.47%).
LC-MS m/z(ESI)=283.00[M+1]。LC-MS m/z(ESI)=283.00[M+1].
第四步:the fourth step:
2-氯-9-(顺-4-甲氧基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(12d)2-Chloro-9-(cis-4-methoxycyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one (12d)
2-chloro-9-(cis-4-methoxycyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one2-chloro-9-(cis-4-methoxycyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one
将2-氯-9-(顺-4-甲氧基环己基)-7,9-二氢-8H-嘌呤-8-酮12c(1.70g,6.01mmol)用N,N- 二甲基甲酰胺(20mL)溶解,0℃搅拌下加入硫酸二甲酯(0.57mL,6.01mmol)和碳酸铯(3.92g,12.03mmol)反应2h。TLC监测至反应结束,把反应液缓慢滴加到冰水中搅拌,析出白色固体,用水和石油醚洗3次过滤得到标题化合物2-氯-9-(顺-4-甲氧基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(12d)(白色固体,1.2g,产率67.25%)。Use 2-chloro-9-(cis-4-methoxycyclohexyl)-7,9-dihydro-8H-purin-8-one 12c (1.70g, 6.01mmol) with N,N-dimethylformaldehyde The amide (20 mL) was dissolved, and dimethyl sulfate (0.57 mL, 6.01 mmol) and cesium carbonate (3.92 g, 12.03 mmol) were added under stirring at 0°C to react for 2 h. TLC monitored until the reaction was over, the reaction solution was slowly added dropwise to ice water and stirred, a white solid precipitated out, washed with water and petroleum ether three times and filtered to obtain the title compound 2-chloro-9-(cis-4-methoxycyclohexyl)- 7-Methyl-7,9-dihydro-8H-purin-8-one (12d) (white solid, 1.2 g, yield 67.25%).
LC-MS m/z(ESI)=297.10[M+1]。LC-MS m/z(ESI)=297.10[M+1].
第五步:the fifth step:
9-(顺-4-甲氧基环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物12)9-(cis-4-methoxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-di Hydrogen-8H-purin-8-one (Compound 12)
9-(cis-4-methoxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(cis-4-methoxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one
将2-氯-9-(顺-4-甲氧基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮12d(100mg,0.34mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(100mg,0.68mmol)、碳酸铯(220mg,0.68mmol)和Brettphos G3 Pd(30mg,0.034mmol)加入干燥的反应瓶,氮气置换三次后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化得到标题化合物9-(顺-4-甲氧基环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物12)(浅黄色固体,47mg,产率34.06%)。The 2-chloro-9-(cis-4-methoxycyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one 12d (100mg, 0.34mmol), 6-methyl -2,3-Dihydrobenzofuran-5-amine intermediate 1 (100mg, 0.68mmol), cesium carbonate (220mg, 0.68mmol) and Brettphos G3 Pd (30mg, 0.034mmol) were added to the dry reaction flask and replaced with nitrogen After three times, dry 1,4-dioxane (1 mL) was added and reacted at 110° C. for 5 h. The reaction was monitored by TLC until the end of the reaction. After cooling to room temperature, it was separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 9-(cis-4-methoxycyclohexyl)- 7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one (compound 12) ( Light yellow solid, 47 mg, yield 34.06%).
1H NMR(400MHz,DMSO-d6)δ8.16(s,1H),7.93(s,1H),7.18(s,1H),6.60(s,1H),4.48(t,2H),4.18–4.09(m,1H),3.39(s,1H),3.25(s,3H),3.19(s,3H),3.12(t,2H),2.48-2.39(m,2H),2.11(s,3H),2.01–1.95(m,2H),1.50-1.39(m,4H)。1H NMR(400MHz,DMSO-d6)δ8.16(s,1H),7.93(s,1H), 7.18(s,1H), 6.60(s,1H), 4.48(t,2H), 4.18-4.09( m, 1H), 3.39(s, 1H), 3.25(s, 3H), 3.19(s, 3H), 3.12(t, 2H), 2.48-2.39(m, 2H), 2.11(s, 3H), 2.01 –1.95(m,2H),1.50-1.39(m,4H).
LC-MS m/z(ESI)=410.20[M+1]。LC-MS m/z(ESI)=410.20[M+1].
实施例13Example 13
9-(1,1-二氧四氢-2H-噻喃-4-基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物13)9-(1,1-Dioxytetrahydro-2H-thiopyran-4-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl) Amino)-7,9-dihydro-8H-purin-8-one (compound 13)
9-(1,1-dioxidotetrahydro-2H-thiopyran-4-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(1,1-dioxidotetrahydro-2H-thiopyran-4-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H -purin-8-one
第一步:first step:
2-氯-4-((1,1-二氧基四氢-2H-硫代吡喃-4-基)氨基)嘧啶-5-羧酸乙酯(13a)2-chloro-4-((1,1-dioxytetrahydro-2H-thiopyran-4-yl)amino)pyrimidine-5-carboxylic acid ethyl ester (13a)
ethyl 2-chloro-4-((1,1-dioxidotetrahydro-2H-thiopyran-4-yl)amino)pyrimidine-5-carboxylateethyl 2-chloro-4-((1,1-dioxidotetrahydro-2H-thiopyran-4-yl)amino)pyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(8g,36mmol)、碳酸钾(9.9g,72mmol)溶解于乙腈(30mL),0℃搅拌下缓慢滴加4-氨基四氢-2H-硫代吡喃1,1-二氧化物(6.6g,36mmol),升温至常温搅拌反应20h。TLC监测至反应结束,向反应液中加入水,析出固体,过滤得到标题化合物2-氯-4-((1,1-二氧基四氢-2H-硫代吡喃-4-基)氨基)嘧啶-5-羧酸乙酯(13a)(褐色固体,6.3g,产率76%)。Dissolve 2,4-dichloropyrimidine-5-carboxylic acid ethyl ester 1a (8g, 36mmol) and potassium carbonate (9.9g, 72mmol) in acetonitrile (30mL), and slowly add 4-aminotetrahydro- 2H-thiopyran 1,1-dioxide (6.6g, 36mmol), heated to room temperature and stirred for 20h. TLC monitors to the end of the reaction, water is added to the reaction solution, a solid is precipitated, and the title compound 2-chloro-4-((1,1-dioxytetrahydro-2H-thiopyran-4-yl)amino is obtained by filtration. ) Ethyl pyrimidine-5-carboxylate (13a) (brown solid, 6.3 g, yield 76%).
1H NMR(400MHz DMSO)δ8.64(s,1H),8.33(d,1H),4.34-4.30(m,3H),3.42-3.35(m,1H),3.08(d,1H),2.24-2.06(m,4H),1.31(t,3H)。1H NMR(400MHz DMSO)δ8.64(s,1H),8.33(d,1H),4.34-4.30(m,3H),3.42-3.35(m,1H),3.08(d,1H),2.24-2.06 (m, 4H), 1.31 (t, 3H).
LC-MS m/z(ESI)=334.20[M+1]。LC-MS m/z(ESI)=334.20[M+1].
第二步:The second step:
2-氯-4-((1,1-二氧基四氢-2H-硫代吡喃-4-基)氨基)嘧啶-5-羧酸(13b)2-chloro-4-((1,1-dioxytetrahydro-2H-thiopyran-4-yl)amino)pyrimidine-5-carboxylic acid (13b)
2-chloro-4-((1,1-dioxidotetrahydro-2H-thiopyran-4-yl)amino)pyrimidine-5-carboxylic acid2-chloro-4-((1,1-dioxidotetrahydro-2H-thiopyran-4-yl)amino)pyrimidine-5-carboxylic acid
将2-氯-4-((1,1-二氧基四氢-2H-硫代吡喃-4-基)氨基)嘧啶-5-羧酸乙酯13a(3g,9mmol)溶解于四氢呋喃/水(15mL/15mL)中,加入氢氧化锂一水合物(756mg,18mmol),常温搅拌反应2h,TLC监测至反应结束,浓缩蒸发掉四氢呋喃后加入2N HCl调节pH至3-4左右,有白色固体析出,过滤,滤饼用水以及石油醚/乙酸乙酯(v/v=10/1)洗两次得标题化合物2-氯-4-((1,1-二氧基四氢-2H-硫代吡喃-4-基)氨基)嘧啶-5-羧酸(13b)(褐色固体,2.3g,产率84%)。Dissolve 2-chloro-4-((1,1-dioxytetrahydro-2H-thiopyran-4-yl)amino)pyrimidine-5-carboxylic acid ethyl ester 13a (3g, 9mmol) in tetrahydrofuran/ Lithium hydroxide monohydrate (756mg, 18mmol) was added to water (15mL/15mL), and the reaction was stirred at room temperature for 2h. TLC monitored until the reaction was over, concentrated and evaporated off the tetrahydrofuran, then added 2N HCl to adjust the pH to about 3-4, white color The solid precipitated out, filtered, and the filter cake was washed twice with water and petroleum ether/ethyl acetate (v/v=10/1) to obtain the title compound 2-chloro-4-((1,1-dioxytetrahydro-2H- Thiopyran-4-yl)amino)pyrimidine-5-carboxylic acid (13b) (brown solid, 2.3 g, yield 84%).
1H NMR(400MHz DMSO)δ13.80(s,1H),8.60(s,1H),8.57(d,1H),4.37-4.29(m,1H),3.50-3.27(m,2H),3.07(d,2H),2.25-2.21(m,2H),2.08-1.98(m,2H)。1H NMR (400MHz DMSO) δ 13.80 (s, 1H), 8.60 (s, 1H), 8.57 (d, 1H), 4.37-4.29 (m, 1H), 3.50-3.27 (m, 2H), 3.07 (d ,2H),2.25-2.21(m,2H),2.08-1.98(m,2H).
LC-MS m/z(ESI)=306.20[M+1]。LC-MS m/z(ESI)=306.20[M+1].
第三步:third step:
2-氯-9-(1,1-二氧基四氢-2H-硫代吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(13c)2-chloro-9-(1,1-dioxytetrahydro-2H-thiopyran-4-yl)-7,9-dihydro-8H-purin-8-one (13c)
2-chloro-9-(1,1-dioxidotetrahydro-2H-thiopyran-4-yl)-7,9-dihydro-8H-purin-8-one2-chloro-9-(1,1-dioxidotetrahydro-2H-thiopyran-4-yl)-7,9-dihydro-8H-purin-8-one
将2-氯-4-((1,1-二氧基四氢-2H-硫代吡喃-4-基)氨基)嘧啶-5-羧酸13b(2.3g,7.5mmol)溶解于二甲基乙酰胺(10mL),加入三乙胺(757mg,7.5mmol)、叠氮磷酸二苯酯(2g,7.5mmol),随后逐步升温至120℃搅拌1.5h。TLC监测至反应结束,将反应液倒入水中有固体析出,过滤即得到标题化合物2-氯-9-(1,1-二氧基四氢-2H-硫代吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(13c)(白色固体,1.7g,产率58%)。Dissolve 2-chloro-4-((1,1-dioxytetrahydro-2H-thiopyran-4-yl)amino)pyrimidine-5-carboxylic acid 13b (2.3g, 7.5mmol) in dimethyl Acetamide (10 mL), triethylamine (757 mg, 7.5 mmol) and diphenyl azide phosphate (2 g, 7.5 mmol) were added, and then gradually heated to 120° C. and stirred for 1.5 h. TLC monitors to the end of the reaction, the reaction solution is poured into water and a solid precipitates out, and it is filtered to obtain the title compound 2-chloro-9-(1,1-dioxytetrahydro-2H-thiopyran-4-yl)- 7,9-dihydro-8H-purin-8-one (13c) (white solid, 1.7 g, yield 58%).
1H NMR(400MHz DMSO)δ11.67(s,1H),8.14(s,1H),4.69-4.61(m,1H),3.55-3.45(m,2H),3.13(d,2H),2.94-2.77(m,2H),2.14-2.02(m,2H)。1H NMR (400MHz DMSO) δ 11.67 (s, 1H), 8.14 (s, 1H), 4.69-4.61 (m, 1H), 3.55-3.45 (m, 2H), 3.13 (d, 2H), 2.94-2.77 (m,2H),2.14-2.02(m,2H).
LC-MS m/z(ESI)=303.20[M+1]。LC-MS m/z(ESI)=303.20[M+1].
第四步:the fourth step:
2-氯-9-(1,1-二氧基四氢-2H-硫代吡喃-4-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(13d)2-chloro-9-(1,1-dioxytetrahydro-2H-thiopyran-4-yl)-7-methyl-7,9-dihydro-8H-purin-8-one (13d )
2-chloro-9-(1,1-dioxidotetrahydro-2H-thiopyran-4-yl)-7-methyl-7,9-dihydro-8H-purin-8-one2-chloro-9-(1,1-dioxidotetrahydro-2H-thiopyran-4-yl)-7-methyl-7,9-dihydro-8H-purin-8-one
将2-氯-9-(1,1-二氧基四氢-2H-硫代吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮13c(1.7g,5.6mmol)溶解于二甲基甲酰胺(10mL),在0℃下加入硫酸二甲酯(709mg,4.9mmol)和碳酸铯(2.7g,9.8mmol)搅拌反应1h。TLC监测至反应结束,向反应液中加入水,有固体析出,过滤得到标题化合物2-氯-9-(1,1-二氧基四氢-2H-硫代吡喃-4-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(13d)(白色固体,1.3g,产率82%)。The 2-chloro-9-(1,1-dioxytetrahydro-2H-thiopyran-4-yl)-7,9-dihydro-8H-purin-8-one 13c (1.7g, 5.6 mmol) was dissolved in dimethylformamide (10 mL), dimethyl sulfate (709 mg, 4.9 mmol) and cesium carbonate (2.7 g, 9.8 mmol) were added at 0° C. and the reaction was stirred for 1 h. TLC monitored to the end of the reaction, water was added to the reaction solution, a solid precipitated out, filtered to obtain the title compound 2-chloro-9-(1,1-dioxytetrahydro-2H-thiopyran-4-yl)- 7-Methyl-7,9-dihydro-8H-purin-8-one (13d) (white solid, 1.3 g, yield 82%).
1H NMR(400MHz DMSO)δ8.33(s,1H),4.26(s,1H),3.33(s,3H),2.99(s,2H),2.11-2.04(m,2H),1.80-1.61(m,2H),1.44(d,2H)。1H NMR (400MHz DMSO) δ 8.33 (s, 1H), 4.26 (s, 1H), 3.33 (s, 3H), 2.99 (s, 2H), 2.11-2.04 (m, 2H), 1.80-1.61 (m ,2H),1.44(d,2H).
LC-MS m/z(ESI)=317.20[M+1]。LC-MS m/z(ESI)=317.20[M+1].
第五步:the fifth step:
9-(1,1-二氧四氢-2H-噻喃-4-基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物13)9-(1,1-Dioxytetrahydro-2H-thiopyran-4-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl) Amino)-7,9-dihydro-8H-purin-8-one (compound 13)
9-(1,1-dioxidotetrahydro-2H-thiopyran-4-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(1,1-dioxidotetrahydro-2H-thiopyran-4-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H -purin-8-one
将2-氯-9-(1,1-二氧基四氢-2H-硫代吡喃-4-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮13d(100mg,0.32mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(94mg,0.63mmol)、碳酸铯(206mg,0.63mmol)和Brettphos G3 Pd(29mg,0.032mmol)加入干燥的反应瓶,氮气置换三次后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化得到标题化合物9-(((1s,4s)-4-甲氧基环己基)-7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物13)(灰白色固体,28mg,产率20.65%)。Combine 2-chloro-9-(1,1-dioxytetrahydro-2H-thiopyran-4-yl)-7-methyl-7,9-dihydro-8H-purin-8-one 13d (100mg, 0.32mmol), 6-methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (94mg, 0.63mmol), cesium carbonate (206mg, 0.63mmol) and Brettphos G3 Pd (29mg, 0.032mmol) was added to a dry reaction flask, replaced with nitrogen three times and then dry 1,4-dioxane (1mL) was added, and reacted at 110°C for 5h. TLC monitors until the reaction is complete. After cooling to room temperature, it is separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 9-(((1s,4s)-4-methoxy Cyclohexyl)-7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purine-8- Ketone (Compound 13) (off-white solid, 28 mg, yield 20.65%).
1H NMR(400MHz,DMSO-d6)δ8.14(s,1H),7.98(s,1H),7.25(s,1H),6.59(s,1H),4.61–4.52(m,1H),4.48(t,2H),3.47-3.39(m,2H),3.27(s,3H),3.20–3.08(m,4H),2.98–2.86(m,2H),2.12(s,3H),2.04(d,2H)。1H NMR(400MHz,DMSO-d6)δ8.14(s,1H),7.98(s,1H),7.25(s,1H),6.59(s,1H),4.61-4.52(m,1H), 4.48( t, 2H), 3.47-3.39 (m, 2H), 3.27 (s, 3H), 3.20-3.08 (m, 4H), 2.98-2.86 (m, 2H), 2.12 (s, 3H), 2.04 (d, 2H).
LC-MS m/z(ESI)=430.10[M+1]。LC-MS m/z(ESI)=430.10[M+1].
实施例14Example 14
7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-((四氢呋喃-2-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(化合物14)7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-((tetrahydrofuran-2-yl)methyl)-7,9-bis Hydrogen-8H-purin-8-one (Compound 14)
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-((tetrahydrofuran-2-yl)methyl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-((tetrahydrofuran-2-yl)methyl)-7,9-dihydro-8H-purin-8- one
第一步:first step:
2-氯-4-((四氢呋喃-2-基)甲基)氨基)嘧啶-5-羧酸乙酯(14a)Ethyl 2-chloro-4-((tetrahydrofuran-2-yl)methyl)amino)pyrimidine-5-carboxylate (14a)
ethyl 2-chloro-4-(((tetrahydrofuran-2-yl)methyl)amino)pyrimidine-5-carboxylateethyl 2-chloro-4-(((tetrahydrofuran-2-yl)methyl)amino)pyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(5g,22.6mmol)、碳酸钾(6.2g,44.8mmol)溶解于乙腈(20mL),0℃搅拌下缓慢滴加4-氨基四氢-2H-硫代吡喃1,1-二氧化物(2.3g,22.6mmol),升温至常温搅拌反应20h。TLC监测至反应结束,向反应液中加入水和乙酸乙酯进行萃取,浓缩有机层经硅胶柱色谱分离提纯(正己烷/乙酸乙酯(v/v)=10:1)纯化得到 标题化合物2-氯-4-((四氢呋喃-2-基)甲基)氨基)嘧啶-5-羧酸乙酯(14a)(白色固体,4.7g,产率87%)。Dissolve 2,4-dichloropyrimidine-5-carboxylic acid ethyl ester 1a (5g, 22.6mmol) and potassium carbonate (6.2g, 44.8mmol) in acetonitrile (20mL), and slowly add 4-aminotetramethylene dropwise with stirring at 0°C. Hydrogen-2H-thiopyran 1,1-dioxide (2.3g, 22.6mmol), heated to room temperature and stirred for 20h. TLC monitoring until the end of the reaction, water and ethyl acetate were added to the reaction solution for extraction, and the organic layer was concentrated and purified by silica gel column chromatography (n-hexane/ethyl acetate (v/v) = 10:1) to obtain the title compound 2. -Chloro-4-((tetrahydrofuran-2-yl)methyl)amino)pyrimidine-5-carboxylic acid ethyl ester (14a) (white solid, 4.7 g, yield 87%).
1H NMR(400MHz DMSO)δ8.62(s,1H),8.57(s,1H),4.33-4.28(m,2H),4.04-4.01(m,1H),3.82-3.77(m,1H),3.68-3.58(m,1H),3.47-3.42(m,1H),1.96-1.81(m,2H),1.59-1.54(m,1H),1.32-1.29(m,3H)。1H NMR(400MHz DMSO)δ8.62(s,1H),8.57(s,1H),4.33-4.28(m,2H),4.04-4.01(m,1H),3.82-3.77(m,1H), 3.68 -3.58 (m, 1H), 3.47-3.42 (m, 1H), 1.96-1.81 (m, 2H), 1.59-1.54 (m, 1H), 1.32-1.29 (m, 3H).
LC-MS m/z(ESI)=286.20[M+1]。LC-MS m/z(ESI)=286.20[M+1].
第二步:The second step:
2-氯-4-((四氢呋喃-2-基)甲基)氨基)嘧啶-5-羧酸(14b)2-Chloro-4-((tetrahydrofuran-2-yl)methyl)amino)pyrimidine-5-carboxylic acid (14b)
2-chloro-4-(((tetrahydrofuran-2-yl)methyl)amino)pyrimidine-5-carboxylic acid2-chloro-4-(((tetrahydrofuran-2-yl)methyl)amino)pyrimidine-5-carboxylic acid
将2-氯-4-((四氢呋喃-2-基)甲基)氨基)嘧啶-5-羧酸乙酯14a(4.7g,16.4mmol)溶解于四氢呋喃/水(15mL/15mL)中,加入氢氧化锂一水合物(788mg,32.8mmol),常温搅拌反应1h。TLC监测至反应结束,浓缩蒸发掉四氢呋喃后加入2N HCl调节pH至3-4左右,有白色固体析出,过滤,滤饼用水以及石油醚/乙酸乙酯(v/v=10/1)洗两次得标题化合物2-氯-4-((四氢呋喃-2-基)甲基)氨基)嘧啶-5-羧酸(14b)(白色固体,3.8g,产率83%)。Dissolve 2-chloro-4-((tetrahydrofuran-2-yl)methyl)amino)pyrimidine-5-carboxylic acid ethyl ester 14a (4.7g, 16.4mmol) in tetrahydrofuran/water (15mL/15mL), add hydrogen Lithium oxide monohydrate (788mg, 32.8mmol), stirred at room temperature for 1h. TLC monitors to the end of the reaction, concentrates and evaporates the tetrahydrofuran, adds 2N HCl to adjust the pH to about 3-4, a white solid is precipitated, filtered, and the filter cake is washed with water and petroleum ether/ethyl acetate (v/v=10/1) The title compound 2-chloro-4-((tetrahydrofuran-2-yl)methyl)amino)pyrimidine-5-carboxylic acid (14b) (white solid, 3.8 g, yield 83%) was obtained in the second time.
1H NMR(400MHz DMSO)δ13.75(s,1H),8.74(t,1H),8.58(s,1H),4.40-4.01(m,1H),3.82-3.76(m,1H),3.69-3.58(m,2H),3.46-3.40(m,1H),1.95-1.90(m,1H),1.86-1.81(m,1H),1.58-1.53(m,1H)。1H NMR (400MHz DMSO) δ 13.75 (s, 1H), 8.74 (t, 1H), 8.58 (s, 1H), 4.40-4.01 (m, 1H), 3.82-3.76 (m, 1H), 3.69-3.58 (m, 2H), 3.46-3.40 (m, 1H), 1.95-1.90 (m, 1H), 1.86-1.81 (m, 1H), 1.58-1.53 (m, 1H).
LC-MS m/z(ESI)=259.2[M+1]。LC-MS m/z(ESI)=259.2[M+1].
第三步:third step:
2-氯-9-((四氢呋喃-2-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(14c)2-Chloro-9-((tetrahydrofuran-2-yl)methyl)-7,9-dihydro-8H-purin-8-one (14c)
2-chloro-9-((tetrahydrofuran-2-yl)methyl)-7,9-dihydro-8H-purin-8-one2-chloro-9-((tetrahydrofuran-2-yl)methyl)-7,9-dihydro-8H-purin-8-one
将2-氯-4-((四氢呋喃-2-基)甲基)氨基)嘧啶-5-羧酸14b(3.8g,14.7mmol)溶解于二甲基乙酰胺(20mL),加入三乙胺(1.8g,17.6mmol)、叠氮磷酸二苯酯(4.4g,16.2mmol),随后逐步升温至120℃搅拌1.5h。TLC监测至反应结束,浓缩反应液,残留物用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=1:10)得到标题化合物2-氯-9-((四氢呋喃-2-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(14c)(白色固体,1.3g,产率46%)。Dissolve 2-chloro-4-((tetrahydrofuran-2-yl)methyl)amino)pyrimidine-5-carboxylic acid 14b (3.8g, 14.7mmol) in dimethylacetamide (20mL), add triethylamine ( 1.8g, 17.6mmol), diphenyl azide phosphate (4.4g, 16.2mmol), and then gradually heated to 120°C and stirred for 1.5h. The reaction was monitored by TLC until the end of the reaction. The reaction solution was concentrated. The residue was separated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)=1:10) to obtain the title compound 2-chloro-9-((tetrahydrofuran-2- (Yl)methyl)-7,9-dihydro-8H-purin-8-one (14c) (white solid, 1.3 g, yield 46%).
LC-MS m/z(ESI)=255.20[M+1]。LC-MS m/z(ESI)=255.20[M+1].
第四步:the fourth step:
2-氯-7-甲基-9-((四氢呋喃-2-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(14d)2-Chloro-7-methyl-9-((tetrahydrofuran-2-yl)methyl)-7,9-dihydro-8H-purin-8-one (14d)
2-chloro-7-methyl-9-((tetrahydrofuran-2-yl)methyl)-7,9-dihydro-8H-purin-8-one2-chloro-7-methyl-9-((tetrahydrofuran-2-yl)methyl)-7,9-dihydro-8H-purin-8-one
将2-氯-9-((四氢呋喃-2-基)甲基)-7,9-二氢-8H-嘌呤-8-酮14c(1.3g,5.1mmol)溶解于二甲基甲酰胺(10mL),在0℃下加入硫酸二甲酯(709mg,4.9mmol)和碳酸铯(2.7g,9.8mmol)搅拌反应1h。TLC监测至反应结束,向反应液中加入水,有固体析出,过滤得到标题化合物2-氯-7-甲基-9-((四氢呋喃-2-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(14d)(白色固体,600mg,产率45%)。Dissolve 2-chloro-9-((tetrahydrofuran-2-yl)methyl)-7,9-dihydro-8H-purin-8-one 14c (1.3g, 5.1mmol) in dimethylformamide (10mL ), dimethyl sulfate (709 mg, 4.9 mmol) and cesium carbonate (2.7 g, 9.8 mmol) were added at 0° C. and the reaction was stirred for 1 h. TLC monitors to the end of the reaction, water is added to the reaction solution, a solid is precipitated, and the title compound 2-chloro-7-methyl-9-((tetrahydrofuran-2-yl)methyl)-7,9-dihydro is obtained by filtration. -8H-purin-8-one (14d) (white solid, 600 mg, yield 45%).
1H NMR(400MHz DMSO)δ8.35(s,1H),4.24-4.20(m,1H),3.90-3.85(m,1H),3.79-3.73(m,2H),3.60(q,1H),3.37(s,3H),1.98-1.77(m,3H),1.70-1.63(m,1H)。1H NMR (400MHz DMSO) δ 8.35 (s, 1H), 4.24-4.20 (m, 1H), 3.90-3.85 (m, 1H), 3.79-3.73 (m, 2H), 3.60 (q, 1H), 3.37 (s, 3H), 1.98-1.77 (m, 3H), 1.70-1.63 (m, 1H).
LC-MS m/z(ESI)=268.20[M+1]。LC-MS m/z(ESI)=268.20[M+1].
第五步:the fifth step:
7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(((四氢呋喃-2-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(化合物14)7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(((tetrahydrofuran-2-yl)methyl)-7,9 -Dihydro-8H-purin-8-one (Compound 14)
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-((tetrahydrofuran-2-yl)methyl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-((tetrahydrofuran-2-yl)methyl)-7,9-dihydro-8H-purin-8- one
将2-氯-7-甲基-9-((四氢呋喃-2-基)甲基)-7,9-二氢-8H-嘌呤-8-酮14d(50mg,0.19mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(56mg,0.37mmol)、碳酸铯(121mg,0.37mmol)和Brettphos G3 Pd(17mg,0.019mmol)加入干燥的反应瓶,氮气置换三次后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室 温后用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化得到标题化合物7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-((四氢呋喃-2-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(化合物14)(淡紫色固体,64mg,产率90.14%)。The 2-chloro-7-methyl-9-((tetrahydrofuran-2-yl)methyl)-7,9-dihydro-8H-purin-8-one 14d (50mg, 0.19mmol), 6-methyl -2,3-Dihydrobenzofuran-5-amine intermediate 1 (56mg, 0.37mmol), cesium carbonate (121mg, 0.37mmol) and Brettphos G3 Pd (17mg, 0.019mmol) were added to the dry reaction flask and replaced with nitrogen After three times, dry 1,4-dioxane (1 mL) was added and reacted at 110° C. for 5 h. The reaction was monitored by TLC until the end of the reaction. After cooling to room temperature, it was separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 7-methyl-2-((6-methyl- 2,3-Dihydrobenzofuran-5-yl)amino)-9-((tetrahydrofuran-2-yl)methyl)-7,9-dihydro-8H-purin-8-one (compound 14) ( Lilac solid, 64 mg, yield 90.14%).
1H NMR(400MHz,DMSO-d6)δ8.26(s,1H),7.95(s,1H),7.19(s,1H),6.60(s,1H),4.49(t,2H),3.76-3.68(m,3H),3.66-3.59(m,2H),3.55–3.50(m,1H),3.28(s,3H),3.11(t,2H),2.78-2.67(m,1H),2.11(s,3H),1.96-1.85(m,1H),1.68–1.58(m,1H)。1H NMR(400MHz,DMSO-d6)δ8.26(s,1H),7.95(s,1H),7.19(s,1H),6.60(s,1H),4.49(t,2H),3.76-3.68( m,3H),3.66-3.59(m,2H),3.55-3.50(m,1H), 3.28(s,3H), 3.11(t,2H), 2.78-2.67(m,1H), 2.11(s, 3H), 1.96-1.85 (m, 1H), 1.68-1.58 (m, 1H).
LC-MS m/z(ESI)=382.20[M+1]。LC-MS m/z(ESI)=382.20[M+1].
实施例15Example 15
7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-((四氢-2H-吡喃-4-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(化合物15)7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-((tetrahydro-2H-pyran-4-yl)methyl) -7,9-dihydro-8H-purin-8-one (Compound 15)
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-((tetrahydro-2H-pyran-4-yl)methyl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-((tetrahydro-2H-pyran-4-yl)methyl)-7,9-dihydro-8H- purin-8-one
第一步:first step:
2-氯-4-(((四氢-2H-吡喃-4-基)甲基)氨基)嘧啶-5-羧酸乙酯(15a)2-chloro-4-(((tetrahydro-2H-pyran-4-yl)methyl)amino)pyrimidine-5-carboxylic acid ethyl ester (15a)
ethyl 2-chloro-4-(((tetrahydro-2H-pyran-4-yl)methyl)amino)pyrimidine-5-carbo-xylateethyl 2-chloro-4-(((tetrahydro-2H-pyran-4-yl)methyl)amino)pyrimidine-5-carbo-xylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(5g,22.6mmol)、碳酸钾(6.2g,44.8mmol)溶解于乙腈(20mL),0℃搅拌下缓慢滴加(四氢-2H-吡喃-4-基)甲酰胺(2.6g,2.6mmol),升温至常温搅拌反应20h。TLC监测至反应结束,向反应液中加入水和乙酸乙酯进行萃取,浓缩有机层经硅胶柱色谱分离提纯(正己烷/乙酸乙酯(v/v)=10:1)纯化得到标题化合物2-氯-4-((四氢-2H-吡喃-4-基)甲基)氨基)嘧啶-5-羧酸乙酯(15a)(白色固体,3.6g,产率70%)。Dissolve 2,4-dichloropyrimidine-5-carboxylic acid ethyl ester 1a (5g, 22.6mmol), potassium carbonate (6.2g, 44.8mmol) in acetonitrile (20mL), slowly add dropwise (tetrahydro- 2H-pyran-4-yl)formamide (2.6g, 2.6mmol) was heated to room temperature and stirred for 20h. TLC monitoring until the end of the reaction, water and ethyl acetate were added to the reaction solution for extraction, and the organic layer was concentrated and purified by silica gel column chromatography (n-hexane/ethyl acetate (v/v) = 10:1) to obtain the title compound 2. -Chloro-4-((tetrahydro-2H-pyran-4-yl)methyl)amino)pyrimidine-5-carboxylic acid ethyl ester (15a) (white solid, 3.6 g, yield 70%).
1H NMR(400MHz DMSO)δ8.60(s,1H),8.54(t,1H),4.31(q,2H),3.84(dd,2H),3.38(t,2H),3.29-3.23(m,2H),1.87-1.82(m,1H),1.55(dd,2H),1.30(t,3H),1.25-1.17(m,2H)。1H NMR(400MHz DMSO)δ8.60(s,1H),8.54(t,1H),4.31(q,2H), 3.84(dd,2H), 3.38(t,2H), 3.29-3.23(m,2H) ), 1.87-1.82 (m, 1H), 1.55 (dd, 2H), 1.30 (t, 3H), 1.25-1.17 (m, 2H).
LC-MS m/z(ESI)=301.20[M+1]。LC-MS m/z(ESI)=301.20[M+1].
第二步:The second step:
2-氯-4-(((四氢-2H-吡喃-4-基)甲基)氨基)嘧啶-5-羧酸(15b)2-chloro-4-(((tetrahydro-2H-pyran-4-yl)methyl)amino)pyrimidine-5-carboxylic acid (15b)
2-chloro-4-(((tetrahydro-2H-pyran-4-yl)methyl)amino)pyrimidine-5-carboxylic acid2-chloro-4-(((tetrahydro-2H-pyran-4-yl)methyl)amino)pyrimidine-5-carboxylic acid
将2-氯-4-((四氢-2H-吡喃-4-基)甲基)氨基)嘧啶-5-羧酸乙酯15a(4.6g,15.3mmol)溶解于四氢呋喃/水(15mL/15mL)中,加入氢氧化锂一水合物(788mg,32.8mmol),常温搅拌反应1h。TLC监测至反应结束,浓缩蒸发掉四氢呋喃后加入2N HCl调节pH至3-4左右,有白色固体析出,过滤,滤饼用水以及石油醚/乙酸乙酯(v/v=10/1)洗两次得标题化合物2-氯-4-((四氢-2H-吡喃-4-基)甲基)氨基)嘧啶-5-羧酸(15b)(白色固体,4.0g,产率83%)。2-Chloro-4-((tetrahydro-2H-pyran-4-yl)methyl)amino)pyrimidine-5-carboxylic acid ethyl ester 15a (4.6g, 15.3mmol) was dissolved in tetrahydrofuran/water (15mL/ 15mL), lithium hydroxide monohydrate (788mg, 32.8mmol) was added, and the reaction was stirred at room temperature for 1h. TLC monitors to the end of the reaction, concentrates and evaporates the tetrahydrofuran, adds 2N HCl to adjust the pH to about 3-4, a white solid is precipitated, filtered, and the filter cake is washed with water and petroleum ether/ethyl acetate (v/v=10/1) The title compound 2-chloro-4-((tetrahydro-2H-pyran-4-yl)methyl)amino)pyrimidine-5-carboxylic acid (15b) (white solid, 4.0g, yield 83%) .
1H NMR(400MHz DMSO)δ13.72(s,1H),8.71(s,1H),8.56(s,1H),3.84(dd,2H),3.37(t,2H),3.29-3.23(m,2H),1.87-1.80(m,1H),1.54(dd,2H),1.26-1.17(m,2H)。1H NMR (400MHz DMSO) δ 13.72 (s, 1H), 8.71 (s, 1H), 8.56 (s, 1H), 3.84 (dd, 2H), 3.37 (t, 2H), 3.29-3.23 (m, 2H) ), 1.87-1.80 (m, 1H), 1.54 (dd, 2H), 1.26-1.17 (m, 2H).
LC-MS m/z(ESI)=273.20[M+1]。LC-MS m/z(ESI)=273.20[M+1].
第三步:third step:
2-氯-9-((四氢-2H-吡喃-4-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(15c)2-chloro-9-((tetrahydro-2H-pyran-4-yl)methyl)-7,9-dihydro-8H-purin-8-one (15c)
2-chloro-9-((tetrahydro-2H-pyran-4-yl)methyl)-7,9-dihydro-8H-purin-8-one2-chloro-9-((tetrahydro-2H-pyran-4-yl)methyl)-7,9-dihydro-8H-purin-8-one
将2-氯-4-((四氢-2H-吡喃-4-基)甲基)氨基)嘧啶-5-羧酸15b(4g,14.6mmol)溶解于二甲基乙酰胺(20mL),加入三乙胺(2.9g,29mmol)、叠氮磷酸二苯酯(5.2g,19mmol),随后逐步升温至120℃搅拌1.5h。TLC监测至反应结束,浓缩反应液,残留物用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=1:10)得到标题化合物2-氯-9-((四氢-2H-吡喃-4-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(15c)(白色固体,1.46g,产率50%)。Dissolve 2-chloro-4-((tetrahydro-2H-pyran-4-yl)methyl)amino)pyrimidine-5-carboxylic acid 15b (4g, 14.6mmol) in dimethylacetamide (20mL), Triethylamine (2.9 g, 29 mmol) and diphenyl azide phosphate (5.2 g, 19 mmol) were added, and then gradually heated to 120° C. and stirred for 1.5 h. The reaction was monitored by TLC until the end of the reaction. The reaction solution was concentrated. The residue was separated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)=1:10) to obtain the title compound 2-chloro-9-((tetrahydro-2H -Pyran-4-yl)methyl)-7,9-dihydro-8H-purin-8-one (15c) (white solid, 1.46 g, yield 50%).
1H NMR(400MHz DMSO)δ11.63(s,1H),8.13(s,1H),3.81(dd,2H),3.66(d,2H),3.25-3.15(m,2H),2.07-2.00(m,1H),1.50(dd,2H),1.29-1.21(m,2H)。1H NMR(400MHz DMSO)δ11.63(s,1H),8.13(s,1H),3.81(dd,2H),3.66(d,2H),3.25-3.15(m,2H),2.07-2.00(m ,1H), 1.50 (dd, 2H), 1.29-1.21 (m, 2H).
LC-MS m/z(ESI)=269.20[M+1]。LC-MS m/z(ESI)=269.20[M+1].
第四步:the fourth step:
2-氯-7-甲基-9-((四氢-2H-吡喃-4-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(15d)2-chloro-7-methyl-9-((tetrahydro-2H-pyran-4-yl)methyl)-7,9-dihydro-8H-purin-8-one (15d)
2-chloro-7-methyl-9-((tetrahydro-2H-pyran-4-yl)methyl)-7,9-dihydro-8H-purin-8-one2-chloro-7-methyl-9-((tetrahydro-2H-pyran-4-yl)methyl)-7,9-dihydro-8H-purin-8-one
将2-氯-9-((四氢-2H-吡喃-4-基)甲基)-7,9-二氢-8H-嘌呤-8-酮15c(1.4g,5.4mmol)溶解于二甲基甲酰胺(10mL),在0℃下加入硫酸二甲酯(688mg,5.4mmol)和碳酸铯(2.6g,8.0mmol)搅拌反应1h。TLC监测至反应结束,向反应液中加入水,有固体析出,过滤得到标题化合物2-氯-7-甲基-9-((四氢-2H-吡喃-4-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(15d)(白色固体,1.2g,产率80%)。The 2-chloro-9-((tetrahydro-2H-pyran-4-yl)methyl)-7,9-dihydro-8H-purin-8-one 15c (1.4g, 5.4mmol) was dissolved in two Methylformamide (10 mL), dimethyl sulfate (688 mg, 5.4 mmol) and cesium carbonate (2.6 g, 8.0 mmol) were added at 0° C. and the reaction was stirred for 1 h. TLC monitored to the end of the reaction, water was added to the reaction solution, a solid precipitated out, filtered to obtain the title compound 2-chloro-7-methyl-9-((tetrahydro-2H-pyran-4-yl)methyl)- 7,9-dihydro-8H-purin-8-one (15d) (white solid, 1.2g, yield 80%).
1H NMR(400MHz DMSO)δ8.34(s,1H),3.81(dd,2H),3.70(d,2H),3.36(s,3H),3.25-3.19(m,2H),2.07-2.01(m,1H),1.51(dd,2H),1.29-1.19(m,2H)。1H NMR (400MHz DMSO) δ 8.34 (s, 1H), 3.81 (dd, 2H), 3.70 (d, 2H), 3.36 (s, 3H), 3.25-3.19 (m, 2H), 2.07-2.01 (m ,1H),1.51(dd,2H),1.29-1.19(m,2H).
LC-MS m/z(ESI)=283.20[M+1]。LC-MS m/z(ESI)=283.20[M+1].
第五步:the fifth step:
7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-((四氢-2H-吡喃-4-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(化合物15)7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-((tetrahydro-2H-pyran-4-yl)methyl) -7,9-dihydro-8H-purin-8-one (Compound 15)
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-((tetrahydro-2H-pyran-4-yl)methyl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-((tetrahydro-2H-pyran-4-yl)methyl)-7,9-dihydro-8H- purin-8-one
将2-氯-7-甲基-9-((四氢-2H-吡喃-4-基)甲基)-7,9-二氢-8H-嘌呤-8-酮15d(100mg,0.35mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(106mg,0.71mmol)、碳酸铯(230mg,0.71mmol)和Brettphos G3 Pd(32mg,0.035mmol)加入干燥的反应瓶,氮气置换三次后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化得到标题化合物7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-((四氢-2H-吡喃-4-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(化合物15)(黄色固体,22mg,产率90.14%)。The 2-chloro-7-methyl-9-((tetrahydro-2H-pyran-4-yl)methyl)-7,9-dihydro-8H-purin-8-one 15d (100mg, 0.35mmol ), 6-methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (106mg, 0.71mmol), cesium carbonate (230mg, 0.71mmol) and Brettphos G3 Pd (32mg, 0.035mmol) were added to the dry After replacing the reaction flask with nitrogen for three times, add dry 1,4-dioxane (1 mL), and react at 110°C for 5 hours. The reaction was monitored by TLC until the end of the reaction. After cooling to room temperature, it was separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 7-methyl-2-(((6-methyl -2,3-Dihydrobenzofuran-5-yl)amino)-9-((tetrahydro-2H-pyran-4-yl)methyl)-7,9-dihydro-8H-purine-8 -Ketone (Compound 15) (yellow solid, 22 mg, yield 90.14%).
1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),7.95(s,1H),7.20(s,1H),6.61(s,1H),4.49(t,2H),3.85-3.80(m,2H),3.60(d,2H),3.28(s,3H),3.26-3.20(m,2H),3.11(t,2H),2.11(s,3H),2.09–2.02(m,1H),1.50–1.45(m,2H),1.25–1.22(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),7.95(s,1H),7.20(s,1H),6.61(s,1H),4.49(t,2H),3.85-3.80( m, 2H), 3.60 (d, 2H), 3.28 (s, 3H), 3.26-3.20 (m, 2H), 3.11 (t, 2H), 2.11 (s, 3H), 2.09-2.02 (m, 1H) ,1.50–1.45(m,2H),1.25–1.22(m,2H).
LC-MS m/z(ESI)=396.20[M+1]。LC-MS m/z(ESI)=396.20[M+1].
实施例16Example 16
9-((反-3-羟基环丁基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物16)9-((trans-3-hydroxycyclobutyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-di Hydrogen-8H-purin-8-one (Compound 16)
9-(trans-3-hydroxycyclobutyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(trans-3-hydroxycyclobutyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one
第一步:first step:
2-氯-4-((反-3-羟基环丁基)氨基)嘧啶-5-羧酸乙酯(16a)Ethyl 2-chloro-4-((trans-3-hydroxycyclobutyl)amino)pyrimidine-5-carboxylate (16a)
ethyl 2-chloro-4-((trans-3-hydroxycyclobutyl)amino)pyrimidine-5-carboxylateethyl 2-chloro-4-((trans-3-hydroxycyclobutyl)amino)pyrimidine-5-carboxylate
将(反-3-氨基环丁烷-1-醇盐酸盐(3.00g,34.48mmol)溶于乙腈(25mL)中,0℃搅拌下加入碳酸钾(14.30g,103.45mmol)、2,4-二氯-5-嘧啶甲酸乙酯1a(11.43g,51.72mmol),升温至常温搅拌反应1h,TLC监测至反应结束,硅藻土过滤后用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=2:1)纯化得标题化合物2-氯-4-((反-3-羟基环丁基)氨基)嘧啶-5-羧酸乙酯(16a)(白色固体,2.9g,产率30.95%)。Dissolve (trans-3-aminocyclobutane-1-ol hydrochloride (3.00g, 34.48mmol) in acetonitrile (25mL), add potassium carbonate (14.30g, 103.45mmol), 2,4 -Dichloro-5-pyrimidinecarboxylic acid ethyl ester 1a (11.43g, 51.72mmol), heated to room temperature and stirred for 1h, monitored by TLC until the reaction is over, filtered through diatomaceous earth and purified by silica gel column chromatography (petroleum ether/ethyl acetate) (v/v)=2:1) Purified to obtain the title compound 2-chloro-4-((trans-3-hydroxycyclobutyl)amino)pyrimidine-5-carboxylic acid ethyl ester (16a) (white solid, 2.9g , The yield is 30.95%).
1H NMR(400MHz,DMSO-d6)δ8.61(s,1H),8.48(d,1H),5.17-5.11(m,1H),4.61–4.51(m,1H),4.36–4.27(m,3H),2.32-2.20(m,4H),1.32(t,3H)。1H NMR(400MHz,DMSO-d6)δ8.61(s,1H), 8.48(d,1H), 5.17-5.11(m,1H), 4.61-4.51(m,1H), 4.36-4.27(m,3H) ), 2.32-2.20 (m, 4H), 1.32 (t, 3H).
LC-MS m/z(ESI)=272.00[M+1]。LC-MS m/z(ESI)=272.00[M+1].
第二步:The second step:
4-((反-3-((叔丁基二苯基硅烷基)氧基)环丁基)氨基)-2-氯嘧啶-5-羧酸乙酯(16b)4-((trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester (16b)
ethyl 4-((trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)amino)-2-chloropyrimidine-5-carboxylateethyl 4-((trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)amino)-2-chloropyrimidine-5-carboxylate
将2-氯-4-((反-3-羟基环丁基)氨基)嘧啶-5-羧酸乙酯18a(3.10g,11.41mmol)溶解于 二氯甲烷(20mL)中,加入叔丁基二苯基氯硅烷(4.45mL,17.11mmol)、咪唑(1.94g,28.52mmol),氮气保护并换气,常温搅拌反应2h,TLC监测至反应结束,硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=10:1)纯化得标题化合物4-(反-3-((叔丁基二苯基硅烷基)氧基)环丁基)氨基)-2-氯嘧啶-5-羧酸乙酯(16b)(无色液体,4.18g,产率64.26%)。Dissolve 2-chloro-4-((trans-3-hydroxycyclobutyl)amino)pyrimidine-5-carboxylic acid ethyl ester 18a (3.10g, 11.41mmol) in dichloromethane (20mL), add tert-butyl Diphenylchlorosilane (4.45mL, 17.11mmol), imidazole (1.94g, 28.52mmol), protected by nitrogen and ventilated, stirred at room temperature for 2h, monitored by TLC until the end of the reaction, and purified by silica gel column chromatography (petroleum ether/ethyl acetate) Ester (v/v)=10:1) was purified to obtain the title compound 4-(trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)amino)-2-chloropyrimidine-5- Ethyl carboxylate (16b) (colorless liquid, 4.18 g, yield 64.26%).
LC-MS m/z(ESI)=510.20[M+1]。LC-MS m/z(ESI)=510.20[M+1].
第三步:third step:
4-((反-3-((叔丁基二苯基硅烷基)氧基)环丁基)氨基)-2-氯嘧啶-5-羧酸(16c)4-((trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)amino)-2-chloropyrimidine-5-carboxylic acid (16c)
4-((trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)amino)-2-chloropyrimidine-5-carboxylic acid4-((trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)amino)-2-chloropyrimidine-5-carboxylic acid
将4-((反-3-((叔丁基二苯基硅烷基)氧基)环丁基)氨基)-2-氯嘧啶-5-羧酸乙酯16b(4.18g,8.19mmol)溶解于四氢呋喃/水(30mL/15mL)中,加入氢氧化锂一水合物(1.03g,24.58mmol),常温搅拌反应2h,TLC监测至反应结束,浓缩蒸发掉四氢呋喃后加入2N HCl调节pH至3-4,加水和乙酸乙酯萃取两次,浓缩有机层用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v)=20:1)纯化得标题化合物4-((反-3-((叔丁基二苯基硅烷基)氧基)环丁基)氨基)-2-氯嘧啶-5-羧酸(16c)(白色固体,3.87g,产率97.97%)。Dissolve 4-((trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester 16b (4.18g, 8.19mmol) In tetrahydrofuran/water (30mL/15mL), lithium hydroxide monohydrate (1.03g, 24.58mmol) was added, the reaction was stirred at room temperature for 2h, TLC monitored until the reaction was over, concentrated and evaporated off the tetrahydrofuran, and then 2N HCl was added to adjust the pH to 3 4. Add water and ethyl acetate to extract twice, concentrate the organic layer and use silica gel column chromatography to separate and purify (dichloromethane/methanol (v/v)=20:1) to obtain the title compound 4-((反-3-(( Tert-butyldiphenylsilyl)oxy)cyclobutyl)amino)-2-chloropyrimidine-5-carboxylic acid (16c) (white solid, 3.87 g, yield 97.97%).
LC-MS m/z(ESI)=482.20[M+1]。LC-MS m/z(ESI)=482.20[M+1].
第四步:the fourth step:
9-(反-3-((叔丁基二苯基硅烷基)氧基)环丁基)-2-氯-7,9-二氢-8H-嘌呤-8-酮(18d)9-(trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)-2-chloro-7,9-dihydro-8H-purin-8-one (18d)
9-(trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)-2-chloro-7,9-dihydro-8H-purin-8-one9-(trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)-2-chloro-7,9-dihydro-8H-purin-8-one
将4-(反-3-((叔丁基二苯基硅烷基)氧基)环丁基)氨基)-2-氯嘧啶-5-羧酸16c(3.87g,8.03mmol)用N,N-二甲基乙酰胺(38mL)溶解,常温搅拌下加入三乙胺(1.11mL,8.03mmol)和叠氮磷酸二苯酯(1.73mL,8.03mmol)反应2h后,升温至110℃回流反应2.5h。TLC监测至反应结束,反应液中加水、乙酸乙酯进行萃取,浓缩有机层用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=1:1)纯化得标题化合物9-(反-3-((叔丁基二苯基硅烷基)氧基)环丁基)-2-氯-7,9-二氢-8H-嘌呤-8-酮(16d)(白色固体,1.81g,产率47.06%)。Use 4-(trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)amino)-2-chloropyrimidine-5-carboxylic acid 16c (3.87g, 8.03mmol) with N, N -Dimethylacetamide (38mL) was dissolved, triethylamine (1.11mL, 8.03mmol) and diphenyl azide phosphate (1.73mL, 8.03mmol) were added under stirring at room temperature, and then reacted for 2h, then heated to 110℃ and refluxed for 2.5 h. TLC monitoring until the end of the reaction, the reaction solution was added with water and ethyl acetate for extraction, and the organic layer was concentrated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v) = 1:1) to obtain the title compound 9-( Trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)-2-chloro-7,9-dihydro-8H-purin-8-one (16d) (white solid, 1.81g , The yield is 47.06%).
1H NMR(400MHz,DMSO-d6)δ11.59(s,1H),8.09(s,1H),7.65–7.62(m,2H),7.62–7.60(m,2H),7.47-7.39(m,6H),5.05–4.97(m,1H),4.96–4.89(m,1H),2.98-2.84(m,2H),2.54-2.46(m,2H),1.02(s,9H)。1H NMR (400MHz, DMSO-d6) δ 11.59 (s, 1H), 8.09 (s, 1H), 7.65-7.62 (m, 2H), 7.62-7.60 (m, 2H), 7.47-7.39 (m, 6H) ), 5.05–4.97(m,1H), 4.96–4.89(m,1H), 2.98-2.84(m,2H), 2.54-2.46(m,2H), 1.02(s,9H).
LC-MS m/z(ESI)=479.20[M+1]。LC-MS m/z(ESI)=479.20[M+1].
第五步:the fifth step:
9-(反-3-((叔丁基二苯基硅烷基)氧基)环丁基)-2-氯-7-甲基-7,9-二氢-8H-嘌呤-8-酮(18e)9-(trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)-2-chloro-7-methyl-7,9-dihydro-8H-purin-8-one( 18e)
9-(trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)-2-chloro-7-methyl-7,9-dihydro-8H-purin-8-one9-(trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)-2-chloro-7-methyl-7,9-dihydro-8H-purin-8-one
将9-(反-3-((叔丁基二苯基硅烷基)氧基)环丁基)-2-氯-7,9-二氢-8H-嘌呤-8-酮16d(1.81g,3.78mmol)用N,N-二甲基甲酰胺(20mL)溶解,0℃搅拌下加入硫酸二甲酯(0.36mL,3.78mmol)和碳酸铯(2.47g,7.57mmol)反应2h。TLC监测至反应结束,把反应液缓慢滴加到冰水中搅拌,加入乙酸乙酯进行萃取,浓缩有机层经硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=3:1)纯化得标题化合物9-(反-3-((叔丁基二苯基硅烷基)氧基)环丁基)-2-氯-7-甲基-7,9-二氢-8H-嘌呤-8-酮(16e)(白色固体,1.80g,产率96.62%)。9-(trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)-2-chloro-7,9-dihydro-8H-purine-8-one 16d (1.81g, 3.78mmol) was dissolved in N,N-dimethylformamide (20mL), dimethyl sulfate (0.36mL, 3.78mmol) and cesium carbonate (2.47g, 7.57mmol) were added and reacted for 2h under stirring at 0°C. TLC monitors until the reaction is over, the reaction solution is slowly added dropwise to ice water and stirred, ethyl acetate is added for extraction, the concentrated organic layer is separated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v) = 3:1) Purified to obtain the title compound 9-(trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)-2-chloro-7-methyl-7,9-dihydro-8H-purine- 8-ketone (16e) (white solid, 1.80 g, yield 96.62%).
LC-MS m/z(ESI)=493.20[M+1]。LC-MS m/z(ESI)=493.20[M+1].
第六步:The sixth step:
9-(反-3-((叔丁基二苯基甲硅烷基)氧基)环丁基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(16f)9-(trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran -5-yl)amino)-7,9-dihydro-8H-purin-8-one (16f)
9-(trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H -purin-8-one
将9-(反-3-((叔丁基二苯基硅烷基)氧基)环丁基)-2-氯-7-甲基-7,9-二氢-8H-嘌呤-8-酮16e(200mg,0.41mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1、(121mg,0.82mmol)、碳酸铯(264mg,0.82mmol)和Brettphos G3 Pd(37mg,0.041mmol)加入干燥的反应瓶,N 2置换三次,然加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。冷却至室温,过滤,浓缩滤液,得到粗品(16f),未经纯化直接投下一步。 9-(trans-3-((tert-butyldiphenylsilyl)oxy)cyclobutyl)-2-chloro-7-methyl-7,9-dihydro-8H-purin-8-one 16e (200mg, 0.41mmol), 6-methyl-2,3-dihydrobenzofuran-5-amine intermediate 1, (121mg, 0.82mmol), cesium carbonate (264mg, 0.82mmol) and Brettphos G3 Pd ( 37 mg, 0.041 mmol) was added to a dry reaction flask, and N 2 was replaced three times, then dry 1,4-dioxane (1 mL) was added, and the reaction was carried out at 110° C. for 5 hours. Cool to room temperature, filter, and concentrate the filtrate to obtain crude product (16f), which is directly used for the next step without purification.
第七步:The seventh step:
9-((反-3-羟基环丁基)-7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物16)9-((trans-3-hydroxycyclobutyl)-7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9- Dihydro-8H-purin-8-one (Compound 16)
9-(trans-3-hydroxycyclobutyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(trans-3-hydroxycyclobutyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one
将第六步所得粗品16f用1,4-二氧六环(5mL)溶解,然后滴加浓盐酸(1mL),室温反应2小时。TLC监测至反应结束,用饱和碳酸氢钠溶液调节pH至7左右,加入二氯甲烷萃取三次,合并有机相,浓缩,使用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化,得到标题化合物9-((反-3-羟基环丁基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物16)(白色固体,54mg,产率36.23%)。The crude product 16f obtained in the sixth step was dissolved in 1,4-dioxane (5 mL), then concentrated hydrochloric acid (1 mL) was added dropwise, and the reaction was carried out at room temperature for 2 hours. TLC monitors to the end of the reaction, adjusts the pH to about 7 with saturated sodium bicarbonate solution, adds dichloromethane for extraction three times, combines the organic phases, concentrates, and uses silica gel column chromatography to separate and purify (dichloromethane/methanol (v/v=40: 1)) Purification to obtain the title compound 9-((trans-3-hydroxycyclobutyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl) Amino)-7,9-dihydro-8H-purin-8-one (compound 16) (white solid, 54 mg, yield 36.23%).
1H NMR(400MHz,DMSO-d6)δ8.24(s,1H),7.97(s,1H),7.22(s,1H),6.61(s,1H),5.07(d,1H),4.99(p,1H),4.49(t,2H),4.40–4.33(m,1H),3.25(s,3H),3.11(t,2H),3.02-2.92(m,2H),2.19-2.13(m,2H),2.12(s,3H)。1H NMR(400MHz,DMSO-d6)δ8.24(s,1H),7.97(s,1H),7.22(s,1H),6.61(s,1H),5.07(d,1H),4.99(p, 1H), 4.49 (t, 2H), 4.40--4.33 (m, 1H), 3.25 (s, 3H), 3.11 (t, 2H), 3.02-2.92 (m, 2H), 2.19-2.13 (m, 2H) , 2.12 (s, 3H).
LC-MS m/z(ESI)=368.10[M+1]。LC-MS m/z(ESI)=368.10[M+1].
实施例17Example 17
9-(6-(羟甲基)四氢-2H-吡喃-3-基)-7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物17)9-(6-(Hydroxymethyl)tetrahydro-2H-pyran-3-yl)-7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5- (Yl)amino)-7,9-dihydro-8H-purin-8-one (compound 17)
9-(6-(hydroxymethyl)tetrahydro-2H-pyran-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(6-(hydroxymethyl)tetrahydro-2H-pyran-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro- 8H-purin-8-one
第一步:first step:
叔丁基((3,4-二氢-2H-吡喃-2-基)甲氧基)二苯基硅烷(17b)Tert-butyl((3,4-dihydro-2H-pyran-2-yl)methoxy)diphenylsilane (17b)
tert-butyl((3,4-dihydro-2H-pyran-2-yl)methoxy)diphenylsilanetert-butyl((3,4-dihydro-2H-pyran-2-yl)methoxy)diphenylsilane
将(3,4-二氢-2H-吡喃-2-基)甲醇17a(20.0g,175.22mmol)溶解到二氯甲烷(400mL)中,加入叔丁基二苯基氯硅烷(68.35mL,262.84mmol)、咪唑(29.82g,438.06mmol),氮气保护并换气,常温搅拌反应过夜,TLC监测至反应结束,用水洗涤,有机相用无水硫酸钠干燥,过滤,滤液减压浓缩经硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=100:1)纯化得标题化合物叔丁基((3,4-二氢-2H-吡喃-2-基)甲氧基)二苯基硅烷(17b)(无色液体,42.78g,产率75.00%)。Dissolve (3,4-dihydro-2H-pyran-2-yl)methanol 17a (20.0g, 175.22mmol) in dichloromethane (400mL), add tert-butyldiphenylchlorosilane (68.35mL, 262.84mmol), imidazole (29.82g, 438.06mmol), protected by nitrogen and ventilated, stirred overnight at room temperature, monitored by TLC until the end of the reaction, washed with water, dried over anhydrous sodium sulfate, filtered, and the filtrate was concentrated under reduced pressure on silica gel Column chromatography separation and purification (petroleum ether/ethyl acetate (v/v) = 100:1) to obtain the title compound tert-butyl ((3,4-dihydro-2H-pyran-2-yl)methoxy) Diphenylsilane (17b) (colorless liquid, 42.78 g, yield 75.00%).
1H NMR(400MHz,CDCl 3):δ7.71-7.68(m,4H),7.45-7.37(m,6H),6.37-6.35(d,1H),4.68-4.65(m,1H),3.96-3.93(m,1H),3.83-3.77(m,1H),3.71-3.67(m,1H),2.08-2.00(m,1H),1.99-1.92(m,2H),1.74-1.71(m,1H),1.02(s,9H)。 1 H NMR (400MHz, CDCl 3 ): δ7.71-7.68 (m, 4H), 7.45-7.37 (m, 6H), 6.37-6.35 (d, 1H), 4.68-4.65 (m, 1H), 3.96 3.93(m,1H),3.83-3.77(m,1H),3.71-3.67(m,1H),2.08-2.00(m,1H),1.99-1.92(m,2H),1.74-1.71(m,1H) ), 1.02(s, 9H).
LC-MS m/z(ESI)=353.20[M+1]。LC-MS m/z(ESI)=353.20[M+1].
第二步:The second step:
6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-醇(17c)6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-ol(17c)
6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-ol6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-ol
将叔丁基((3,4-二氢-2H-吡喃-2-基)甲氧基)二苯基硅烷17b(23.5g,66.7mmol)加入到四氢呋喃(200mL)中,氮气保护下,降温至-78℃后,将硼烷二甲硫醚络合物(100mL,2M,200.00mmol)滴加到反应液中,滴加完毕后,自然升至室温搅拌过夜。向反应体系中缓慢滴加1N的氢氧化钠水溶液至无硼烷气体放出后,加入30%双氧水(90mL)至反应液中,反应液在45℃搅拌2h。向反应体系中加入水和乙酸乙酯萃取,有机相用无水硫酸钠干燥,过滤,减压浓缩经反相柱层析色谱分离提纯(乙腈/水(v/v)=5:95)纯化得标题化合物6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-醇(17c)(无色液体,18.03g,产率73.00%)。Add tert-butyl((3,4-dihydro-2H-pyran-2-yl)methoxy)diphenylsilane 17b (23.5g, 66.7mmol) to tetrahydrofuran (200mL), under nitrogen protection, After cooling to -78°C, borane dimethyl sulfide complex (100 mL, 2M, 200.00 mmol) was added dropwise to the reaction solution, and after the addition was completed, it was naturally warmed to room temperature and stirred overnight. After slowly adding 1N sodium hydroxide aqueous solution to the reaction system until no borane gas was released, 30% hydrogen peroxide (90 mL) was added to the reaction solution, and the reaction solution was stirred at 45° C. for 2 h. Water and ethyl acetate were added to the reaction system for extraction, the organic phase was dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure, and purified by reverse phase column chromatography (acetonitrile/water (v/v) = 5:95) The title compound 6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-ol (17c) (colorless liquid, 18.03g, yield 73.00%) .
LC-MS m/z(ESI)=371.30[M+1]。LC-MS m/z(ESI)=371.30[M+1].
第三步:third step:
2-(6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-基)异吲哚啉-1,3-二酮(17d)2-(6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)isoindoline-1,3-dione (17d)
2-(6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)isoindoline-1,3-dione2-(6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)isoindoline-1,3-dione
将6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-醇17c(13.0g,35.1mmol)溶于四氢呋喃中(150mL),常温搅拌下加入邻苯二甲酰亚胺(5.16g,35.1mmol)和三苯基膦(13.8g,52.7mmol),再缓慢滴加偶氮二甲酸二异丙酯(10.7g,52.7mmol),升温至70℃回流反应1h,TLC监测至反应结束,反应液浓缩经硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=100:1)纯化得标题2-(6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-基)异吲哚啉-1,3-二酮(17d)(白色固体,9.0g,产率51.34%)。Dissolve 6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-ol 17c (13.0g, 35.1mmol) in tetrahydrofuran (150mL), stir at room temperature Add phthalimide (5.16g, 35.1mmol) and triphenylphosphine (13.8g, 52.7mmol), then slowly add diisopropyl azodicarboxylate (10.7g, 52.7mmol) dropwise and increase the temperature The reaction was refluxed at 70°C for 1 hour, monitored by TLC until the reaction was over, the reaction solution was concentrated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v) = 100:1) to obtain the title 2-(6-((( Tert-Butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)isoindoline-1,3-dione (17d) (white solid, 9.0g, yield 51.34%).
LC-MS m/z(ESI)=500.20[M+1]。LC-MS m/z(ESI)=500.20[M+1].
第四步:the fourth step:
6-(((叔丁基二苯基硅烷基)氧基)甲基四氢-2H-吡喃-3-胺(17e)6-(((tert-butyldiphenylsilyl)oxy)methyltetrahydro-2H-pyran-3-amine(17e)
6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-amine6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-amine
将2-(6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-基)异吲哚啉-1,3-二酮19d(9.0g,18.0mmol)溶于甲醇(50mL)中,再加入水合联氨(1.8g,36.0mmol),反应液回流反应2h,反应完成后,加入二氯甲烷(50mL)大量固体析出,过滤后旋干,得到的粘稠液固体再用二氯甲烷洗涤两次后过滤,合并有机相,减压浓缩得标题化合物6-((叔丁基二苯基硅烷基)氧基)甲基四氢-2H-吡喃-3-胺(17e)(无色液体,6.3g,产率94.10%)。The 2-(6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)isoindoline-1,3-dione 19d(9.0 g, 18.0mmol) was dissolved in methanol (50mL), and then hydrazine hydrate (1.8g, 36.0mmol) was added. The reaction solution was refluxed for 2h. After the reaction was completed, dichloromethane (50mL) was added to precipitate a large amount of solids, filtered and rotated. After drying, the viscous liquid solid obtained was washed twice with dichloromethane and filtered. The organic phases were combined and concentrated under reduced pressure to obtain the title compound 6-((tert-butyldiphenylsilyl)oxy)methyltetrahydro- 2H-pyran-3-amine (17e) (colorless liquid, 6.3 g, yield 94.10%).
LC-MS m/z(ESI)=370.20[M+1]。LC-MS m/z(ESI)=370.20[M+1].
第五步:the fifth step:
4-((6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-基)氨基)-2-氯嘧啶-5-羧酸乙酯(19f)4-((6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester (19f)
ethyl 4-((6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)amino)-2-chloropyrimidine-5-carboxylateethyl 4-((6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)amino)-2-chloropyrimidine-5-carboxylate
将2,4-二氯-5-嘧啶甲酸乙酯1a(6.94g,31.40mmol)溶于乙腈(25mL)中,常温搅拌下加入碳酸钾(8.68g,62.80mmol),再缓慢滴加6-((叔丁基二苯基硅烷基)氧基)甲基四氢-2H-吡喃-3-胺17e(7.40g,20.93mmol)的乙腈溶液(15mL),搅拌反应2h,TLC监测至反应结束,硅藻土过滤后经硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=20:1)纯化得标题化合物4-((6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-基)氨基)-2-氯嘧啶-5-羧酸乙酯(17f)(淡黄色固体,5.20g,产率46.86%)。Dissolve 2,4-dichloro-5-pyrimidine ethyl carboxylate 1a (6.94g, 31.40mmol) in acetonitrile (25mL), add potassium carbonate (8.68g, 62.80mmol) while stirring at room temperature, and slowly add 6- ((Tert-butyldiphenylsilyl)oxy)methyltetrahydro-2H-pyran-3-amine 17e (7.40g, 20.93mmol) in acetonitrile (15mL), the reaction was stirred for 2h, TLC monitored until the reaction At the end, after filtration through diatomaceous earth, it was separated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)=20:1) to obtain the title compound 4-((6-(((tert-butyldiphenylsilane) (Yl)oxy)methyl)tetrahydro-2H-pyran-3-yl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester (17f) (light yellow solid, 5.20g, yield 46.86%) .
LC-MS m/z(ESI)=554.30[M+1]。LC-MS m/z(ESI)=554.30[M+1].
第六步:The sixth step:
4-((6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-基)氨基)-2-氯嘧啶-5-羧酸(17g)4-((6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)amino)-2-chloropyrimidine-5-carboxylic acid (17g )
4-((6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)amino)-2-chloropyrimidine-5-carboxylate4-((6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)amino)-2-chloropyrimidine-5-carboxylate
将4-((6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-基)氨基)-2-氯嘧啶-5-羧酸乙酯17f(5.20g,9.383mmol)溶解于四氢呋喃/水(30mL/15mL)中,加入氢氧化锂一水合物(1.18g,28.15mmol),常温搅拌反应3h后升温至50℃反应2h,TLC监测至反应结束,浓缩蒸发掉四氢呋喃后加入2N HCl调节pH至3-4,加水和乙酸乙酯萃取两次,浓缩有机层经硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=4:1)纯化得标题化合物4-((6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-基)氨基)-2-氯嘧啶-5-羧酸(17g)(白色固体,2.57g,产率52.06%)。4-((6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl Ester 17f (5.20g, 9.383mmol) was dissolved in tetrahydrofuran/water (30mL/15mL), lithium hydroxide monohydrate (1.18g, 28.15mmol) was added, the reaction was stirred at room temperature for 3 hours, and then heated to 50°C for 2 hours, monitored by TLC To the end of the reaction, concentrate and evaporate the tetrahydrofuran, add 2N HCl to adjust the pH to 3-4, add water and ethyl acetate for extraction twice, concentrate the organic layer and separate and purify by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)= 4:1) Purified to obtain the title compound 4-((6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)amino)-2-chloro Pyrimidine-5-carboxylic acid (17 g) (white solid, 2.57 g, yield 52.06%).
LC-MS m/z(ESI)=526.20[M+1]。LC-MS m/z(ESI)=526.20[M+1].
第七步:The seventh step:
9-(6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-基)-2-氯-7,9-二氢-8H-嘌呤-8-酮(17h)9-(6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)-2-chloro-7,9-dihydro-8H-purine -8-ketone (17h)
9-(6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)-2-chloro-7,9-dihydro-8H-purin-8-one9-(6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)-2-chloro-7,9-dihydro-8H-purin-8-one
将4-((6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-基)氨基)-2-氯嘧啶-5-羧酸17g(2.57g,4.88mmol)用N,N-二甲基乙酰胺(24mL)溶解,常温搅拌下加入三乙胺(0.68mL,4.88mmol)和叠氮磷酸二苯酯(1.05mL,4.88mmol)反应2h后,升温至110℃回流反应2.5h。TLC监测至反应结束,反应液中加水、乙酸乙酯进行萃取,浓缩有机层用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=1:1)纯化得标题化合物9-(6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-基)-2-氯-7,9-二氢-8H-嘌呤-8-酮(17h)(淡粉色固体,1.60g,产率62.66%)。The 4-((6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)amino)-2-chloropyrimidine-5-carboxylic acid 17g (2.57g, 4.88mmol) dissolved in N,N-dimethylacetamide (24mL), add triethylamine (0.68mL, 4.88mmol) and diphenyl azide phosphate (1.05mL, 4.88mmol) under stirring at room temperature After reacting for 2h, the temperature was raised to 110°C and refluxed for 2.5h. TLC monitoring until the end of the reaction, the reaction solution was added with water and ethyl acetate for extraction, and the organic layer was concentrated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v) = 1:1) to obtain the title compound 9-( 6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)-2-chloro-7,9-dihydro-8H-purine-8- Ketone (17h) (light pink solid, 1.60 g, yield 62.66%).
1H NMR(400MHz,DMSO-d6)δ11.65(s,1H),8.12(s,1H),7.67-7.62(m,4H),7.46-7.39(m,6H),4.32–4.24(m,1H),4.13–4.06(m,1H),4.00-3.93(m,1H),3.91-3.81(m,2H),3.56(dd,1H),2.53-2.47(m,1H),2.07-1.98(m,1H),1.85-1.72(m,2H),1.01(s,9H)。1H NMR (400MHz, DMSO-d6) δ 11.65 (s, 1H), 8.12 (s, 1H), 7.67-7.62 (m, 4H), 7.46-7.39 (m, 6H), 4.32-4.24 (m, 1H) ), 4.13-4.06 (m, 1H), 4.00-3.93 (m, 1H), 3.91-3.81 (m, 2H), 3.56 (dd, 1H), 2.53-2.47 (m, 1H), 2.07-1.98 (m ,1H),1.85-1.72(m,2H),1.01(s,9H).
LC-MS m/z(ESI)=523.20[M+1]。LC-MS m/z(ESI)=523.20[M+1].
第八步:The eighth step:
9-(6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-基)-2-氯-7-甲基-7,9-二氢-8H-嘌呤-8-酮(17i)9-(6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)-2-chloro-7-methyl-7,9-bis Hydrogen-8H-purin-8-one (17i)
9-(6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)-2-chloro-7-methyl-7,9-dihydro-8H-purin-8-one9-(6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)-2-chloro-7-methyl-7,9-dihydro-8H-purin-8-one
将9-(6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-基)-2-氯-7,9-二氢-8H-嘌呤-8-酮17h(1.60g,3.06mmol)用N,N-二甲基甲酰胺(15mL)溶解,常温搅拌下加入硫酸二甲酯(0.29mL,3.06mmol)和碳酸铯(2.00g,6.13mmol)反应1h,TLC监测至反应结束,加入水和乙酸乙酯进行萃取,浓缩有机层用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=3:2)纯化得标题化合物9-(6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-基)-2-氯-7-甲基-7,9-二氢-8H-嘌呤-8-酮(17i)(淡粉色液体,1.6g,产率97.21%)。Add 9-(6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)-2-chloro-7,9-dihydro-8H- Purine-8-one 17h (1.60g, 3.06mmol) was dissolved with N,N-dimethylformamide (15mL), and dimethyl sulfate (0.29mL, 3.06mmol) and cesium carbonate (2.00g, 6.13mmol) reacted for 1h, monitored by TLC to the end of the reaction, added water and ethyl acetate for extraction, concentrated the organic layer and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)=3:2) to obtain the title Compound 9-(6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)-2-chloro-7-methyl-7,9- Dihydro-8H-purin-8-one (17i) (light pink liquid, 1.6 g, yield 97.21%).
1H NMR(400MHz,DMSO-d6)δ8.35(s,1H),7.67-7.62(m,4H),7.49-7.39(m,6H),4.36–4.28(m,1H),4.14–4.07(m,1H),4.00-3.93(m,1H),3.90-3.81(m,2H),3.57(dd,1H),3.35(s,3H),2.55-2.44(m,1H),2.09-1.98(m,1H),1.87-1.73(m,2H),1.01(s,9H)。1H NMR(400MHz,DMSO-d6)δ8.35(s,1H),7.67-7.62(m,4H),7.49-7.39(m,6H),4.36-4.28(m,1H),4.14-4.07(m ,1H),4.00-3.93(m,1H),3.90-3.81(m,2H),3.57(dd,1H),3.35(s,3H),2.55-2.44(m,1H),2.09-1.98(m ,1H),1.87-1.73(m,2H),1.01(s,9H).
LC-MS m/z(ESI)=537.30[M+1]。LC-MS m/z(ESI)=537.30[M+1].
第九步:Step 9:
9-(6-(((叔丁基二苯基甲硅烷基)氧基)甲基)四氢-2H-吡喃-3-基)-7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(17j)9-(6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)-7-methyl-2-(((6-methyl (2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one (17j)
9-(6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino )-7,9-dihydro-8H-purin-8-one
将9-(6-(((叔丁基二苯基硅烷基)氧基)甲基)四氢-2H-吡喃-3-基)-2-氯-7-甲基-7,9-二氢-8H-嘌呤-8-酮17i(150mg,0.28mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(83mg,0.56mmol)、碳酸铯(182mg,0.56mmol)和Brettphos G3 Pd(25mg,0.028mmol)加入干燥的反应瓶,N2置换三次,然后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后过滤,浓缩滤液得到粗品17j,未经纯化直接投下一步。Add 9-(6-(((tert-butyldiphenylsilyl)oxy)methyl)tetrahydro-2H-pyran-3-yl)-2-chloro-7-methyl-7,9- Dihydro-8H-purin-8-one 17i (150mg, 0.28mmol), 6-methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (83mg, 0.56mmol), cesium carbonate (182mg , 0.56mmol) and Brettphos G3 Pd (25mg, 0.028mmol) were added to a dry reaction flask, replaced with N2 three times, and then added dry 1,4-dioxane (1mL), and reacted at 110°C for 5h. TLC monitored to the end of the reaction, cooled to room temperature and filtered, and the filtrate was concentrated to obtain the crude product 17j, which was directly used for the next step without purification.
第十步:The tenth step:
9-(6-(羟甲基)四氢-2H-吡喃-3-基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物17)9-(6-(Hydroxymethyl)tetrahydro-2H-pyran-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl )Amino)-7,9-dihydro-8H-purin-8-one (compound 17)
9-(6-(hydroxymethyl)tetrahydro-2H-pyran-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(6-(hydroxymethyl)tetrahydro-2H-pyran-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro- 8H-purin-8-one
将第九步所得粗品17j用1,4-二氧六环(5mL)溶解,然后滴加浓盐酸(1mL),室温 反应2h。TLC监测至反应结束,用饱和碳酸氢钠溶液调节pH至7左右,加入DCM萃取三次,合并有机相,浓缩后使用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化,得到标题化合物9-(6-(羟甲基)四氢-2H-吡喃-3-基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物17)(白色固体,33mg,产率28.72%)。The crude product 17j obtained in the ninth step was dissolved with 1,4-dioxane (5 mL), then concentrated hydrochloric acid (1 mL) was added dropwise, and the reaction was carried out at room temperature for 2 h. TLC monitors until the reaction is over, adjust the pH to about 7 with saturated sodium bicarbonate solution, add DCM for extraction three times, combine the organic phases, concentrate and use silica gel column chromatography for separation and purification (dichloromethane/methanol (v/v=40:1) ) To obtain the title compound 9-(6-(hydroxymethyl)tetrahydro-2H-pyran-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzene P-furan-5-yl)amino)-7,9-dihydro-8H-purin-8-one (compound 17) (white solid, 33 mg, yield 28.72%).
1H NMR(400MHz,DMSO-d6)δ8.25(s,1H),7.97(s,1H),7.13(s,1H),6.62(s,1H),4.55-4.46(m,3H),4.24–4.15(m,1H),4.07(t,1H),3.68-3.60(m,1H),3.59–3.52(m,1H),3.44(dd,1H),3.38-3.31(m,2H),3.25(s,3H),3.13(t,2H),2.09(s,3H),1.85-1.76(m,1H),1.71-1.58(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.25(s,1H),7.97(s,1H),7.13(s,1H),6.62(s,1H),4.55-4.46(m,3H), 4.24-- 4.15 (m, 1H), 4.07 (t, 1H), 3.68-3.60 (m, 1H), 3.59-3.52 (m, 1H), 3.44 (dd, 1H), 3.38-3.31 (m, 2H), 3.25 ( s, 3H), 3.13 (t, 2H), 2.09 (s, 3H), 1.85-1.76 (m, 1H), 1.71-1.58 (m, 2H).
LC-MS m/z(ESI)=412.20[M+1]。LC-MS m/z(ESI)=412.20[M+1].
实施例18Example 18
9-(3-羟基环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物18)9-(3-Hydroxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H- Purine-8-one (Compound 18)
9-(3-hydroxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(3-hydroxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one
第一步:first step:
叔丁基(3-羟基环己基)氨基甲酸酯(18b)Tert-Butyl (3-hydroxycyclohexyl) carbamate (18b)
tert-butyl(3-hydroxycyclohexyl)carbamatetert-butyl(3-hydroxycyclohexyl)carbamate
将叔丁基(3-氧代环己基)氨基甲酸酯18a(8.20g,38.45mmol)溶于四氢呋喃(40mL)中,0℃搅拌下加入硼氢化钠(4.36g,115.35mmol)反应3h,TLC监测至反应结束,向反应液中缓慢加入饱和碳酸钠溶液,常温搅拌3h,加入乙酸乙酯萃取,浓缩有机层得 标题化合物叔丁基(3-羟基环己基)氨基甲酸酯(18b)(黄色液体,8.00g,产率96.95%)。Dissolve tert-butyl (3-oxocyclohexyl) carbamate 18a (8.20g, 38.45mmol) in tetrahydrofuran (40mL), add sodium borohydride (4.36g, 115.35mmol) under stirring at 0°C and react for 3h, TLC monitors to the end of the reaction, slowly add saturated sodium carbonate solution to the reaction solution, stir at room temperature for 3 hours, add ethyl acetate for extraction, concentrate the organic layer to obtain the title compound tert-butyl (3-hydroxycyclohexyl) carbamate (18b) (Yellow liquid, 8.00 g, yield 96.95%).
LC-MS m/z(ESI)=216.20[M+1]。LC-MS m/z(ESI)=216.20[M+1].
第二步:The second step:
3-氨基环己烷-1-醇(20c)3-Aminocyclohexane-1-ol (20c)
3-aminocyclohexan-1-ol hydrochloride3-aminocyclohexan-1-ol hydrochloride
将叔丁基(3-羟基环己基)氨基甲酸酯18b(8.00g,37.16mmol)溶于2M氯化氢-乙酸乙酯(35mL)中,常温搅拌反应4h,TLC监测至反应结束,浓缩蒸发掉溶剂得标题化合物3-氨基环己烷-1-醇盐酸盐(18c)(黄色液体,粗品,4.00g,产率71.05%)。Dissolve tert-butyl (3-hydroxycyclohexyl) carbamate 18b (8.00g, 37.16mmol) in 2M hydrogen chloride-ethyl acetate (35mL), stir the reaction at room temperature for 4h, monitor by TLC until the reaction is complete, concentrate and evaporate The solvent was used to obtain the title compound 3-aminocyclohexane-1-ol hydrochloride (18c) (yellow liquid, crude product, 4.00 g, yield 71.05%).
LC-MS m/z(ESI)=116.10[M+1]。LC-MS m/z(ESI)=116.10[M+1].
第三步:third step:
2-氯-4-((3-羟基环己基)氨基)嘧啶-5-羧酸乙酯(18d)Ethyl 2-chloro-4-((3-hydroxycyclohexyl)amino)pyrimidine-5-carboxylate (18d)
ethyl 2-chloro-4-((3-hydroxycyclohexyl)amino)pyrimidine-5-carboxylateethyl 2-chloro-4-((3-hydroxycyclohexyl)amino)pyrimidine-5-carboxylate
将3-氨基环己烷-1-醇盐酸盐18c(4.00g,26.38mmol)溶于乙腈(30mL)中,0℃搅拌下加入2,4-二氯-5-嘧啶甲酸乙酯1a(8.75g,39.57mmol)、碳酸钾(10.94g,79.14mmol),升温至常温搅拌反应2h,TLC监测至反应结束,硅藻土过滤后用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=2:1)纯化得标题化合物2-氯-4-((3-羟基环己基)氨基)嘧啶-5-羧酸乙酯(18d)(淡黄色固体,1.40g,产率17.71%)。Dissolve 3-aminocyclohexane-1-ol hydrochloride 18c (4.00g, 26.38mmol) in acetonitrile (30mL), add 2,4-dichloro-5-pyrimidinecarboxylic acid ethyl ester 1a( 8.75g, 39.57mmol), potassium carbonate (10.94g, 79.14mmol), heated to room temperature and stirred for 2h, monitored by TLC until the reaction was over, filtered through diatomaceous earth and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v /v)=2:1) Purified to obtain the title compound 2-chloro-4-((3-hydroxycyclohexyl)amino)pyrimidine-5-carboxylic acid ethyl ester (18d) (light yellow solid, 1.40g, yield 17.71 %).
LC-MS m/z(ESI)=300.10[M+1]。LC-MS m/z(ESI)=300.10[M+1].
第四步:the fourth step:
4-((3-((叔丁基二苯基硅烷基)氧基)环己基)氨基)-2-氯嘧啶-5-羧酸乙酯(18e)4-((3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester (18e)
ethyl 4-((3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)amino)-2-chloropyrimidine-5-carboxylateethyl 4-((3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)amino)-2-chloropyrimidine-5-carboxylate
将2-氯-4-((3-羟基环己基)氨基)嘧啶-5-羧酸乙酯18d(1.10g,3.67mmol)溶解于二氯甲烷(20mL)中,加入叔丁基二苯基氯硅烷(1.43mL,5.50mmol)、咪唑(0.62g,9.17mmol),氮气保护并换气,常温搅拌反应2h,TLC监测至反应结束,硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=8:1)纯化得标题化合物4-((3-((叔丁基二苯基硅烷基)氧基)环己基)氨基)-2-氯嘧啶-5-羧酸乙酯(18e)(无色液体,1.90g,产率96.21%)。Dissolve 2-chloro-4-((3-hydroxycyclohexyl)amino)pyrimidine-5-carboxylic acid ethyl ester 18d (1.10g, 3.67mmol) in dichloromethane (20mL), add tert-butyldiphenyl Chlorosilane (1.43mL, 5.50mmol), imidazole (0.62g, 9.17mmol), protected by nitrogen and ventilated, stirred at room temperature for 2h, monitored by TLC until the end of the reaction, and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v /v)=8:1) was purified to obtain the title compound 4-((3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester ( 18e) (colorless liquid, 1.90 g, yield 96.21%).
LC-MS m/z(ESI)=538.20[M+1]。LC-MS m/z(ESI)=538.20[M+1].
第五步:the fifth step:
4-((3-((叔丁基二苯基硅烷基)氧基)环己基)氨基)-2-氯嘧啶-5-羧酸(18f)4-((3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)amino)-2-chloropyrimidine-5-carboxylic acid (18f)
4-((3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)amino)-2-chloropyrimidine-5-carboxylate4-((3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)amino)-2-chloropyrimidine-5-carboxylate
将4-((3-((叔丁基二苯基硅烷基)氧基)环己基)氨基)-2-氯嘧啶-5-羧酸乙酯18e(1.90g,3.53mmol)溶解于四氢呋喃/水(20mL/20mL)中,加入氢氧化锂一水合物(0.44g,10.59mmol),常温搅拌反应2h,TLC监测至反应结束,浓缩蒸发掉四氢呋喃后加入2N HCl调节pH至3-4,加水和乙酸乙酯萃取两次,浓缩有机层得标题化合物4-((3-((叔丁基二苯基硅烷基)氧基)环己基)氨基)-2-氯嘧啶-5-羧酸18f(白色固体,1.10g,产率61.08%)。4-((3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester 18e (1.90g, 3.53mmol) was dissolved in tetrahydrofuran/ In water (20mL/20mL), add lithium hydroxide monohydrate (0.44g, 10.59mmol), stir the reaction at room temperature for 2h, monitor by TLC until the reaction is over, concentrate and evaporate the tetrahydrofuran, add 2N HCl to adjust the pH to 3-4, add water It was extracted twice with ethyl acetate, and the organic layer was concentrated to obtain the title compound 4-((3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)amino)-2-chloropyrimidine-5-carboxylic acid 18f (White solid, 1.10g, yield 61.08%).
LC-MS m/z(ESI)=510.20[M+1]。LC-MS m/z(ESI)=510.20[M+1].
第六步:The sixth step:
9-(3-((叔丁基二苯基硅基)氧基)环己基)-2-氯-7,9-二氢-8H-嘌呤-8-酮(18g)9-(3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)-2-chloro-7,9-dihydro-8H-purin-8-one (18g)
9-(3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)-2-chloro-7,9-dihydro-8H-purin-8-one9-(3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)-2-chloro-7,9-dihydro-8H-purin-8-one
将4-((3-((叔丁基二苯基硅烷基)氧基)环己基)氨基)-2-氯嘧啶-5-羧酸18f(1.10g,2.16mmol)用N,N-二甲基乙酰胺(23mL)溶解,常温搅拌下加入三乙胺(0.30mL,2.16mmol)和叠氮磷酸二苯酯(0.46mL,2.16mmol)反应2h后,升温至110℃回流反应2.5h。TLC监测至反应结束,反应液中加水、乙酸乙酯进行萃取,浓缩有机层用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=1:1)纯化得标题化合物9-(3-((叔丁基二苯基硅基)氧基)环己基)-2-氯-7,9-二氢-8H-嘌呤-8-酮18g(白色固体,0.31g,产率28.35%)。Use 4-((3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)amino)-2-chloropyrimidine-5-carboxylic acid 18f (1.10g, 2.16mmol) with N,N-di Methyl acetamide (23 mL) was dissolved, triethylamine (0.30 mL, 2.16 mmol) and diphenyl azide phosphate (0.46 mL, 2.16 mmol) were added under stirring at room temperature and reacted for 2 h, then the temperature was raised to 110° C. and refluxed for 2.5 h. TLC monitoring until the end of the reaction, the reaction solution was added with water and ethyl acetate for extraction, and the organic layer was concentrated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v) = 1:1) to obtain the title compound 9-( 3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)-2-chloro-7,9-dihydro-8H-purin-8-one 18g (white solid, 0.31g, yield 28.35% ).
LC-MS m/z(ESI)=507.20[M+1]。LC-MS m/z(ESI)=507.20[M+1].
第七步:The seventh step:
9-(3-((叔丁基二苯基硅烷基)氧基)环己基)-2-氯-7-甲基-7,9-二氢-8H-嘌呤-8-酮(18h)9-(3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)-2-chloro-7-methyl-7,9-dihydro-8H-purin-8-one (18h)
9-(3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)-2-chloro-7-methyl-7,9-dihydro-8H-purin-8-one9-(3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)-2-chloro-7-methyl-7,9-dihydro-8H-purin-8-one
将9-(3-((叔丁基二苯基硅基)氧基)环己基)-2-氯-7,9-二氢-8H-嘌呤-8-酮18g(0.30g,0.59mmol)用N,N-二甲基甲酰胺(10mL)溶解,0℃搅拌下加入硫酸二甲酯(0.06mL,0.59mmol)和碳酸铯(0.39g,1.18mmol)反应1h。TLC监测至反应结束,把反应液缓慢滴加到冰水中搅拌,加入乙酸乙酯进行萃取,浓缩有机层得标题化合物9-(3-((叔丁基二苯基硅烷基)氧基)环己基)-2-氯-7-甲基-7,9-二氢-8H-嘌呤-8-酮(18h)(无色液体,0.30g,产率97.31%)。9-(3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)-2-chloro-7,9-dihydro-8H-purin-8-one 18g (0.30g, 0.59mmol) Dissolve with N,N-dimethylformamide (10mL), add dimethyl sulfate (0.06mL, 0.59mmol) and cesium carbonate (0.39g, 1.18mmol) under stirring at 0°C and react for 1h. TLC monitors until the reaction is over, the reaction solution is slowly added dropwise to ice water and stirred, ethyl acetate is added for extraction, the organic layer is concentrated to obtain the title compound 9-(3-((tert-butyldiphenylsilyl)oxy) Hexyl)-2-chloro-7-methyl-7,9-dihydro-8H-purin-8-one (18h) (colorless liquid, 0.30 g, yield 97.31%).
LC-MS m/z(ESI)=521.20[M+1]。LC-MS m/z(ESI)=521.20[M+1].
第八步:Step 8:
9-(3-((叔丁基二苯基甲硅烷基)氧基)环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(20i)9-(3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5- (Yl)amino)-7,9-dihydro-8H-purin-8-one (20i)
9-(3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin -8-one
将9-(3-((叔丁基二苯基硅烷基)氧基)环己基)-2-氯-7-甲基-7,9-二氢-8H-嘌呤-8-酮18h(200mg,0.38mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(115mg,0.76mmol)、碳酸铯(251mg,0.76mmol)和Brettphos G3 Pd(35mg,0.038mmol)加入干燥的反应瓶,N 2置换三次,然后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后过滤,浓缩滤液得到粗品18i,未经纯化直接投下一步。 Add 9-(3-((tert-butyldiphenylsilyl)oxy)cyclohexyl)-2-chloro-7-methyl-7,9-dihydro-8H-purin-8-one 18h (200mg , 0.38mmol), 6-methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (115mg, 0.76mmol), cesium carbonate (251mg, 0.76mmol) and Brettphos G3 Pd (35mg, 0.038mmol) ) Add a dry reaction flask, replace with N 2 three times, then add dry 1,4-dioxane (1 mL), and react at 110° C. for 5 hours. TLC monitored to the end of the reaction, cooled to room temperature and filtered. The filtrate was concentrated to obtain the crude product 18i, which was directly used in the next step without purification.
第九步:Step 9:
9-(3-羟基环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物18)9-(3-Hydroxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H- Purine-8-one (Compound 18)
9-(3-hydroxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(3-hydroxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one
将第八步所得粗品18i用1,4-二氧六环(5mL)溶解,然后滴加浓盐酸(1mL),室温反应2h。TLC监测至反应结束,用饱和碳酸氢钠溶液调节pH至7左右,加入二氯甲烷萃取三次,合并有机相,浓缩后使用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化,得到标题化合物9-(3-羟基环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物18)(白色固体,22mg,产率14.50%)。The crude product 18i obtained in the eighth step was dissolved with 1,4-dioxane (5 mL), then concentrated hydrochloric acid (1 mL) was added dropwise, and the reaction was carried out at room temperature for 2 h. TLC monitors to the end of the reaction, adjust the pH to about 7 with saturated sodium bicarbonate solution, add dichloromethane for extraction three times, combine the organic phases, concentrate and use silica gel column chromatography to separate and purify (dichloromethane/methanol (v/v=40: 1)) Purification to obtain the title compound 9-(3-hydroxycyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7 , 9-dihydro-8H-purin-8-one (Compound 18) (white solid, 22 mg, yield 14.50%).
1H NMR(400MHz,DMSO-d6)δ8.20(s,1H),7.96(s,1H),7.26(s,1H),6.60(s,1H),4.78(d,1H),4.48(t,2H),4.17–4.08(m,1H),3.52–3.42(m,1H),3.26(s,3H),3.21–3.05(m,2H),2.20-2.03(m,5H),1.90–1.79(m,2H),1.78-1.70(m,1H),1.59(d,1H),1.34-1.21(m,1H),1.11–0.99(m,1H)。1H NMR (400MHz, DMSO-d6) δ 8.20 (s, 1H), 7.96 (s, 1H), 7.26 (s, 1H), 6.60 (s, 1H), 4.78 (d, 1H), 4.48 (t, 2H), 4.17-4.08(m, 1H), 3.52-3.42(m, 1H), 3.26(s, 3H), 3.21-3.05(m, 2H), 2.20-2.03(m, 5H), 1.90-1.79( m, 2H), 1.78-1.70 (m, 1H), 1.59 (d, 1H), 1.34-1.21 (m, 1H), 1.11-0.99 (m, 1H).
LC-MS m/z(ESI)=396.20[M+1]。LC-MS m/z(ESI)=396.20[M+1].
实施例19Example 19
反-4-(7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-8-氧代-7,8-二氢-9H-嘌呤-9-基)环己烷-1-腈(化合物19)Trans-4-(7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-8-oxo-7,8-dihydro-9H- Purin-9-yl)cyclohexane-1-carbonitrile (Compound 19)
trans-4-(7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-8-oxo-7,8-dihydro-9H-purin-9-yl)cyclohexane-1-carbonitriletrans-4-(7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-8-oxo-7,8-dihydro-9H-purin-9-yl)cyclohexane- 1-carbonitrile
第一步:first step:
叔丁基(反-4-氨甲酰环己基)氨基甲酸酯(19b)Tert-Butyl (trans-4-carbamoylcyclohexyl) carbamate (19b)
tert-butyl(trans-4-carbamoylcyclohexyl)carbamatetert-butyl(trans-4-carbamoylcyclohexyl)carbamate
反-4-((叔丁氧羰基)氨基)环己烷-1-羧酸19a(5.0g,20.5mmol)、O-(7-氮杂苯并三氮唑基)-N,N,N',N'-四甲基脲鎓六氟磷酸盐(9.4g,24.7mmol)溶解于二氯甲烷(15mL),在0℃下搅拌20min,加入N,N二异丙基乙胺(10.5g,82mmol)及氯化铵(3.3g,61.5mmol),在室温搅拌4h。TLC监测至反应结束,向反应液中加入水10mL,分离有机相,饱和盐水洗一次,用无水硫酸钠干燥并用硅胶拌样,用正相过柱仪过出产物,浓缩得到标题化合物叔丁基(反-4-氨甲酰环己基)氨基甲酸酯(19b)(白色固体,4.4g,产率83%)。Trans-4-((tert-butoxycarbonyl)amino)cyclohexane-1-carboxylic acid 19a (5.0g, 20.5mmol), O-(7-azabenzotriazole)-N,N,N ',N'-Tetramethyluronium hexafluorophosphate (9.4g, 24.7mmol) was dissolved in dichloromethane (15mL), stirred at 0℃ for 20min, and N,N diisopropylethylamine (10.5g , 82mmol) and ammonium chloride (3.3g, 61.5mmol), stirred at room temperature for 4h. TLC monitors to the end of the reaction, add 10 mL of water to the reaction solution, separate the organic phase, wash with saturated brine once, dry with anhydrous sodium sulfate and mix the sample with silica gel, pass the product with a normal phase column passer, and concentrate to obtain the title compound tert-butyl Group (trans-4-carbamoylcyclohexyl) carbamate (19b) (white solid, 4.4 g, yield 83%).
LC-MS m/z(ESI)=243.30[M+1]。LC-MS m/z(ESI)=243.30[M+1].
第二步:The second step:
叔丁基(反-4-氰基环己基)氨基甲酸酯(19c)Tert-butyl (trans-4-cyanocyclohexyl) carbamate (19c)
tert-butyl(trans-4-cyanocyclohexyl)carbamatetert-butyl(trans-4-cyanocyclohexyl)carbamate
叔丁基(反-4-氨甲酰环己基)氨基甲酸酯19b(4.4g,18.0mmol)溶解于70mL吡啶中,冰浴降温,然后将三氯氧磷(7.7mL)滴加进反应液中,冰浴搅拌1h。TLC监测反应完全,在冰浴下加入水20mL,用乙酸乙酯萃取4次,然后用酸水洗有机相7次,最后用饱和盐水洗两次,无水硫酸钠干燥,过滤并浓缩至干得到化合物叔丁基(反-4-氰基环己基)氨基甲酸酯(19c)(黄色固体,1.2g,产率30%)。Tert-butyl (trans-4-carbamoylcyclohexyl) carbamate 19b (4.4g, 18.0mmol) was dissolved in 70mL of pyridine, cooled in an ice bath, and then phosphorus oxychloride (7.7mL) was added dropwise to the reaction In the solution, stir in an ice bath for 1 hour. TLC monitors the completion of the reaction, adds 20 mL of water in an ice bath, extracts 4 times with ethyl acetate, then washes the organic phase 7 times with acid water, and finally washes twice with saturated brine, dried with anhydrous sodium sulfate, filtered and concentrated to dryness. The compound tert-butyl (trans-4-cyanocyclohexyl) carbamate (19c) (yellow solid, 1.2 g, yield 30%).
1H NMR(400MHz DMSO)δ6.80(m,1H),3.24-3.22(m,1H),2.63-2.55(m,1H),1.99-1.95(m,2H),1.77-1.73(m,2H),1.56-1.46(m,2H),1.36(s,9H),1.21-1.11(m,2H)。1H NMR(400MHz DMSO)δ6.80(m,1H),3.24-3.22(m,1H),2.63-2.55(m,1H),1.99-1.95(m,2H),1.77-1.73(m,2H) , 1.56-1.46 (m, 2H), 1.36 (s, 9H), 1.21-1.11 (m, 2H).
LC-MS m/z(ESI)=225.30[M+1]。LC-MS m/z(ESI)=225.30[M+1].
第三步:third step:
反-4-氨基环己烷-1-碳腈(19d)Trans-4-aminocyclohexane-1-carbonitrile (19d)
trans-4-aminocyclohexane-1-carbonitriletrans-4-aminocyclohexane-1-carbonitrile
将叔丁基(反-4-氰基环己基)氨基甲酸酯19c(1.2g,9.6mmol)溶解于盐酸乙酸乙酯溶液中(20mL),加热至45℃,搅拌2h。TLC监测至反应结束,有白色固体析出,浓缩干燥得到标题化合物反-4-氨基环己烷-1-碳腈(19d)(白色固体,660mg,产率60%)。Dissolve tert-butyl (trans-4-cyanocyclohexyl) carbamate 19c (1.2 g, 9.6 mmol) in hydrochloric acid ethyl acetate solution (20 mL), heat to 45° C., and stir for 2 h. TLC monitored to the end of the reaction, a white solid precipitated, concentrated and dried to obtain the title compound trans-4-aminocyclohexane-1-carbonitrile (19d) (white solid, 660 mg, yield 60%).
LC-MS m/z(ESI)=125.30[M+1]。LC-MS m/z(ESI)=125.30[M+1].
第四步:the fourth step:
2-氯-4-(反-4-氰基环己基)氨基)嘧啶-5-羧酸乙酯(19e)2-Chloro-4-(trans-4-cyanocyclohexyl)amino)pyrimidine-5-carboxylic acid ethyl ester (19e)
ethyl 2-chloro-4-((trans-4-cyanocyclohexyl)amino)pyrimidine-5-carboxylateethyl 2-chloro-4-((trans-4-cyanocyclohexyl)amino)pyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(1.4g,5.35mmol)、碳酸钾(1.4g,10.7mmol)溶解于乙腈(10mL),在0℃下加入(1r,4r)-4-氨基环己烷-1-碳腈19d(660mg,5.35mmol),在室温搅拌20h。TLC监测至反应结束,向反应液中加入水和乙酸乙酯萃取三次,饱和盐水洗一次,用无水硫酸钠干燥并用硅胶柱色谱分离(正己烷/乙酸乙酯(v/v)=10:1)纯化,得到标题化合物2-氯-4-((1r,4r)-4-氰基环己基)氨基)嘧啶-5-羧酸乙酯(19e)(白色固体,810g,产率62%)。Dissolve 2,4-dichloropyrimidine-5-carboxylic acid ethyl ester 1a (1.4g, 5.35mmol), potassium carbonate (1.4g, 10.7mmol) in acetonitrile (10mL), add (1r, 4r) at 0℃ -4-Aminocyclohexane-1-carbonitrile 19d (660 mg, 5.35 mmol) was stirred at room temperature for 20 h. TLC monitors until the reaction is complete, the reaction solution is added with water and ethyl acetate for extraction three times, washed with saturated brine once, dried with anhydrous sodium sulfate and separated by silica gel column chromatography (n-hexane/ethyl acetate (v/v)=10: 1) Purification to obtain the title compound 2-chloro-4-((1r,4r)-4-cyanocyclohexyl)amino)pyrimidine-5-carboxylic acid ethyl ester (19e) (white solid, 810g, yield 62% ).
1H NMR(400MHz DMSO)δ8.62(s,1H),8.27(d,1H),4.30(q,2H),4.04-3.96(m,1H),4.04-3.67(m,1H),2.76-2.70(m,1H),2.04-2.00(m,2H),1.96-1.92(m,2H),1.72-1.62(m,2H),1.47-1.37(m,2H),1.30(t,3H)。1H NMR (400MHz DMSO) δ8.62 (s, 1H), 8.27 (d, 1H), 4.30 (q, 2H), 4.04-3.96 (m, 1H), 4.04-3.67 (m, 1H), 2.76-2.70 (m, 1H), 2.04-2.00 (m, 2H), 1.96-1.92 (m, 2H), 1.72-1.62 (m, 2H), 1.47-1.37 (m, 2H), 1.30 (t, 3H).
LC-MS m/z(ESI)=310.20[M+1]。LC-MS m/z(ESI)=310.20[M+1].
第五步:the fifth step:
2-氯-4-((反-4-氰基环己基)氨基)嘧啶-5-羧酸(19f)2-chloro-4-((trans-4-cyanocyclohexyl)amino)pyrimidine-5-carboxylic acid (19f)
2-chloro-4-((trans-4-cyanocyclohexyl)amino)pyrimidine-5-carboxylic acide2-chloro-4-((trans-4-cyanocyclohexyl)amino)pyrimidine-5-carboxylic acid
将2-氯-4-((反-4-氰基环己基)氨基)嘧啶-5-羧酸乙酯19e(810mg,2.6mmol)溶解于四氢呋喃/水(4mL/4mL)中,加入氢氧化锂(247mg,5.2mmol),室温搅拌1h。TLC监测至反应结束,旋干四氢呋喃,调pH至4-5,有白色固体析出,过滤,滤饼用石油醚/乙酸乙酯(v/v=10/1)洗两次,浓缩得到标题化合物2-氯-4-((反-4-氰基环己基)氨基)嘧啶-5-羧酸(19f)(白色固体,790mg,产率86%)Dissolve 2-chloro-4-((trans-4-cyanocyclohexyl)amino)pyrimidine-5-carboxylic acid ethyl ester 19e (810mg, 2.6mmol) in tetrahydrofuran/water (4mL/4mL) and add hydroxide Lithium (247mg, 5.2mmol), stirred at room temperature for 1h. TLC monitors to the end of the reaction, spin-dry the tetrahydrofuran, adjust the pH to 4-5, a white solid is precipitated, filtered, the filter cake is washed twice with petroleum ether/ethyl acetate (v/v=10/1), and concentrated to obtain the title compound 2-Chloro-4-((trans-4-cyanocyclohexyl)amino)pyrimidine-5-carboxylic acid (19f) (white solid, 790mg, yield 86%)
1H NMR(400MHz DMSO)δ8.58(s,1H),8.49(d,1H),4.01-3.87(m,1H),2.76-2.71(m,1H),2.03-1.93(m,4H),1.72-1.63(m,2H),1.44-1.35(m,2H)。1H NMR(400MHz DMSO)δ8.58(s,1H),8.49(d,1H),4.01-3.87(m,1H),2.76-2.71(m,1H),2.03-1.93(m,4H),1.72 -1.63 (m, 2H), 1.44-1.35 (m, 2H).
LC-MS m/z(ESI)=282.30[M+1]。LC-MS m/z(ESI)=282.30[M+1].
第六步:The sixth step:
反-4-(2-氯-8-氧代-7,8-二氢-9H-嘌呤-9-基)环己烷-1-碳腈(19g)Trans-4-(2-chloro-8-oxo-7,8-dihydro-9H-purin-9-yl)cyclohexane-1-carbonitrile (19g)
trans-4-(2-chloro-8-oxo-7,8-dihydro-9H-purin-9-yl)cyclohexane-1-carbonitriletrans-4-(2-chloro-8-oxo-7,8-dihydro-9H-purin-9-yl)cyclohexane-1-carbonitrile
将2-氯-4-(反-4-氰基环己基)氨基)嘧啶-5-羧酸19f(730mg,2.6mmol)溶解于二甲基乙酰胺(10mL),加入三乙胺(263mg,2.6mmol)、叠氮磷酸二苯酯(715mg,2.6mmol),随后逐步升温至110℃,搅拌1.5h。TLC监测至反应结束,浓缩反应液,残留物用硅胶柱色谱分离(石油醚/乙酸乙酯(v/v)=1:10),得到标题化合物反-4-(2-氯-8-氧代-7,8-二氢-9H-嘌呤-9-基)环己烷-1-碳腈(19g)(白色固体,553mg,产率68%)。2-Chloro-4-(trans-4-cyanocyclohexyl)amino)pyrimidine-5-carboxylic acid 19f (730mg, 2.6mmol) was dissolved in dimethylacetamide (10mL), and triethylamine (263mg, 2.6mmol), diphenyl azide phosphate (715mg, 2.6mmol), then gradually heated to 110°C and stirred for 1.5h. The reaction was monitored by TLC until the end of the reaction. The reaction solution was concentrated. The residue was separated by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)=1:10) to obtain the title compound trans-4-(2-chloro-8-oxygen). Di-7,8-dihydro-9H-purin-9-yl)cyclohexane-1-carbonitrile (19 g) (white solid, 553 mg, yield 68%).
1H NMR(400MHz DMSO)δ11.63(s,1H),8.12(s,1H),4.23-4.16(m,1H),2.78-2.72(m,1H),2.25-2.12(m,4H),1.82-1.68(m,4H)。1H NMR (400MHz DMSO) δ 11.63 (s, 1H), 8.12 (s, 1H), 4.23-4.16 (m, 1H), 2.78-2.72 (m, 1H), 2.25-2.12 (m, 4H), 1.82 -1.68(m,4H).
LC-MS m/z(ESI)=278.30[M+1]。LC-MS m/z(ESI)=278.30[M+1].
第七步:The seventh step:
反-4-(2-氯-7-甲基-8-氧代-7,8-二氢-9H-嘌呤-9-基)环己烷-1-碳腈(19h)Trans-4-(2-chloro-7-methyl-8-oxo-7,8-dihydro-9H-purin-9-yl)cyclohexane-1-carbonitrile (19h)
trans-4-(2-chloro-7-methyl-8-oxo-7,8-dihydro-9H-purin-9-yl)cyclohexane-1-carbonitriletrans-4-(2-chloro-7-methyl-8-oxo-7,8-dihydro-9H-purin-9-yl)cyclohexane-1-carbonitrile
将反-4-(2-氯-8-氧代-7,8-二氢-9H-嘌呤-9-基)环己烷-1-碳腈19g(553mg,2mmol)溶解于二甲基甲酰胺(5mL),在0℃下加入硫酸二甲酯(252mg,2mmol)和碳酸铯(977mg,3mmol),0℃搅拌30min。TLC监测至反应结束,向反应液中加入水10mL,有固体析出,过滤得到标题化合物反-4-(2-氯-7-甲基-8-氧代-7,8-二氢-9H-嘌呤-9-基)环己烷-1-碳腈(19h)(黄色固体,420mg,产率72%)。Dissolve 19 g (553 mg, 2 mmol) of trans-4-(2-chloro-8-oxo-7,8-dihydro-9H-purin-9-yl)cyclohexane-1-carbonitrile in dimethylform Amide (5 mL), dimethyl sulfate (252 mg, 2 mmol) and cesium carbonate (977 mg, 3 mmol) were added at 0°C, and stirred at 0°C for 30 min. TLC monitored to the end of the reaction, 10mL of water was added to the reaction solution, a solid precipitated out, filtered to obtain the title compound trans-4-(2-chloro-7-methyl-8-oxo-7,8-dihydro-9H- Purin-9-yl)cyclohexane-1-carbonitrile (19h) (yellow solid, 420 mg, yield 72%).
1H NMR(400MHz DMSO)δ8.34(s,1H),4.28-4.21(m,1H),2.79-2.72(m,1H),2.22-2.13(m,4H),1.82-1.70(m,4H)。1H NMR (400MHz DMSO) δ8.34 (s, 1H), 4.28-4.21 (m, 1H), 2.79-2.72 (m, 1H), 2.22-2.13 (m, 4H), 1.82-1.70 (m, 4H) .
LC-MS m/z(ESI)=292.30[M+1]。LC-MS m/z(ESI)=292.30[M+1].
第八步:The eighth step:
反-4-(7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-8-氧代-7,8-二氢-9H-嘌呤-9-基)环己烷-1-腈(化合物19)Trans-4-(7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-8-oxo-7,8-dihydro-9H- Purin-9-yl)cyclohexane-1-carbonitrile (Compound 19)
trans-4-(7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-8-oxo-7,8-dihydro-9H-purin-9-yl)cyclohexane-1-carbonitriletrans-4-(7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-8-oxo-7,8-dihydro-9H-purin-9-yl)cyclohexane- 1-carbonitrile
将反-4-(2-氯-7-甲基-8-氧代-7,8-二氢-9H-嘌呤-9-基)环己烷-1-碳腈19h(100mg,0.34mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(102mg,0.68mmol)、碳酸铯(223mg,0.68mmol)和Brettphos G3 Pd(31mg,0.034mmol)加入干燥的反应管,N 2置换三次,然后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温,使用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化,得到标题化合物反-4-(7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-8-氧代-7,8-二氢-9H-嘌呤-9-基)环己烷-1-腈(化合物19)(浅黄色固体,34mg,产率24.52%)。 Trans-4-(2-chloro-7-methyl-8-oxo-7,8-dihydro-9H-purin-9-yl)cyclohexane-1-carbonitrile 19h (100mg, 0.34mmol) , 6-Methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (102mg, 0.68mmol), cesium carbonate (223mg, 0.68mmol) and Brettphos G3 Pd (31mg, 0.034mmol) were added to the dry In the reaction tube, replace with N 2 three times, then add dry 1,4-dioxane (1 mL), and react at 110° C. for 5 h. TLC monitors until the reaction is complete, cools to room temperature, and uses silica gel column chromatography to separate and purify (dichloromethane/methanol (v/v=40:1)) to obtain the title compound trans-4-(7-methyl-2-( (6-Methyl-2,3-dihydrobenzofuran-5-yl)amino)-8-oxo-7,8-dihydro-9H-purin-9-yl)cyclohexane-1-carbonitrile (Compound 19) (pale yellow solid, 34 mg, yield 24.52%).
1H NMR(400MHz,DMSO-d6)δ8.18(s,1H),7.94(s,1H),7.21(s,1H),6.62(s,1H),4.50(t,2H),4.21–4.12(m,1H),3.25(s,3H),3.12(t,2H),2.59–2.53(m,1H),2.31–2.19(m,2H),2.18-2.09(m,5H),1.80–1.73(m,2H),1.71–1.61(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.18(s,1H),7.94(s,1H),7.21(s,1H),6.62(s,1H),4.50(t,2H),4.21-4.12( m, 1H), 3.25 (s, 3H), 3.12 (t, 2H), 2.59-2.53 (m, 1H), 2.31-2.19 (m, 2H), 2.18-2.09 (m, 5H), 1.80-1.73 ( m,2H),1.71-1.61(m,2H).
LC-MS m/z(ESI)=405.20[M+1]。LC-MS m/z(ESI)=405.20[M+1].
实施例20Example 20
9-(2-异丙基-4-甲基吡啶-3-基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8酮(化合物20)9-(2-isopropyl-4-methylpyridin-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino) -7,9-dihydro-8H-purin-8one (Compound 20)
9-(2-isopropyl-4-methylpyridin-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(2-isopropyl-4-methylpyridin-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin -8-one
第一步:first step:
2-氯-4-((2-异丙基-4-甲基吡啶-3-基)氨基)嘧啶-5-羧酸乙酯(20a)Ethyl 2-chloro-4-((2-isopropyl-4-methylpyridin-3-yl)amino)pyrimidine-5-carboxylate (20a)
2-chloro-4-((2-isopropyl-4-methylpyridin-3-yl)amino)pyrimidine-5-carboxylate2-chloro-4-((2-isopropyl-4-methylpyridin-3-yl)amino)pyrimidine-5-carboxylate
将2-异丙基-4-甲基吡啶-3-胺(21.61g,143.92mmol)、2,4-二氯嘧啶-5-羧酸乙酯1a(26.51g,119.93mmol),溶于乙腈(400mL)中,0℃搅拌下加入碳酸钾(33.15g,239.86mmol),随后逐步升温至90℃搅拌72h。TLC监测至反应结束,向反应液中加入水和乙酸乙酯萃取,合并有机相,用无水硫酸钠干燥,过滤浓缩得到标题化合物2-氯-4-((2-异丙基-4-甲基吡啶-3-基)氨基)嘧啶-5-羧酸乙酯(20a)(棕色固体,44.23g,产率91.95%)。Dissolve 2-isopropyl-4-methylpyridine-3-amine (21.61g, 143.92mmol), 2,4-dichloropyrimidine-5-carboxylic acid ethyl ester 1a (26.51g, 119.93mmol) in acetonitrile (400mL), add potassium carbonate (33.15g, 239.86mmol) under stirring at 0°C, and then gradually increase the temperature to 90°C and stir for 72h. The reaction was monitored by TLC until the end of the reaction. Water and ethyl acetate were added to the reaction solution for extraction. The organic phases were combined, dried over anhydrous sodium sulfate, filtered and concentrated to obtain the title compound 2-chloro-4-((2-isopropyl-4- Methylpyridin-3-yl)amino)pyrimidine-5-carboxylic acid ethyl ester (20a) (brown solid, 44.23 g, yield 91.95%).
1H NMR(400MHz,DMSO-d6)δ9.83(s,1H),8.77(s,1H),8.39(d,1H),7.20(d,1H),4.39(q,2H),3.10-3.03(m,1H),2.13(s,3H),1.36(t,3H),1.12(d,6H)。1H NMR(400MHz,DMSO-d6)δ9.83(s,1H), 8.77(s,1H), 8.39(d,1H), 7.20(d,1H), 4.39(q,2H), 3.10-3.03( m, 1H), 2.13 (s, 3H), 1.36 (t, 3H), 1.12 (d, 6H).
LC-MS m/z(ESI)=335.10[M+1]。LC-MS m/z(ESI)=335.10[M+1].
第二步:The second step:
2-氯-4-((2-异丙基-4-甲基吡啶-3-基)氨基)嘧啶-5-羧酸(20b)2-chloro-4-((2-isopropyl-4-methylpyridin-3-yl)amino)pyrimidine-5-carboxylic acid (20b)
2-chloro-4-((2-isopropyl-4-methylpyridin-3-yl)amino)pyrimidine-5-carboxylic acid2-chloro-4-((2-isopropyl-4-methylpyridin-3-yl)amino)pyrimidine-5-carboxylic acid
将2-氯-4-((2-异丙基-4-甲基吡啶-3-基)氨基)嘧啶-5-羧酸乙酯20a(44.23g,131.42mmol),用四氢呋喃/水(200mL/200mL)溶解,再加入氢氧化锂(11.03g,262.84mmol)常温搅拌反应3h。TLC监测至反应结束,旋干四氢呋喃,用2N盐酸调节pH至1-2,析出固体,过滤,并用水洗涤滤饼干燥得到标题化合物2-氯-4-((2-异丙基-4-甲基吡啶-3-基)氨基)嘧啶-5-羧酸(20b)(棕色固体,29.29g,产率72.66%)。The 2-chloro-4-((2-isopropyl-4-methylpyridin-3-yl)amino)pyrimidine-5-carboxylic acid ethyl ester 20a (44.23g, 131.42mmol) was mixed with tetrahydrofuran/water (200mL /200mL) was dissolved, and then lithium hydroxide (11.03g, 262.84mmol) was added and stirred at room temperature for 3h. TLC monitors to the end of the reaction, spin-dry the tetrahydrofuran, adjust the pH to 1-2 with 2N hydrochloric acid, precipitate a solid, filter, and wash the filter cake with water to dry to obtain the title compound 2-chloro-4-((2-isopropyl-4- Methylpyridin-3-yl)amino)pyrimidine-5-carboxylic acid (20b) (brown solid, 29.29 g, yield 72.66%).
1H NMR(400MHz,DMSO-d6)δ10.23(s,1H),8.72(s,1H),8.38(d,1H),7.20(d,1H),3.11-3.04(m,1H),2.13(s,3H),1.26(d,1H),1.12(d,6H)。1H NMR(400MHz,DMSO-d6)δ10.23(s,1H),8.72(s,1H),8.38(d,1H),7.20(d,1H),3.11-3.04(m,1H),2.13( s, 3H), 1.26 (d, 1H), 1.12 (d, 6H).
LC-MS m/z(ESI)=307.09[M+1]。LC-MS m/z(ESI)=307.09[M+1].
第三步:third step:
2-氯-9-(2-异丙基-4-甲基吡啶-3-基)-7,9-二氢-8H-嘌呤-8-酮(20c)2-Chloro-9-(2-isopropyl-4-methylpyridin-3-yl)-7,9-dihydro-8H-purin-8-one (20c)
2-chloro-9-(2-isopropyl-4-methylpyridin-3-yl)-7,9-dihydro-8H-purin-8-one2-chloro-9-(2-isopropyl-4-methylpyridin-3-yl)-7,9-dihydro-8H-purin-8-one
将2-氯-4-((2-异丙基-4-甲基吡啶-3-基)氨基)嘧啶-5-羧酸20b(25.00g,81.5mmol)溶解于四氢呋喃中,再将温度降至0℃,加入叠氮磷酸二苯酯(24.6g,89.5mmol),三乙胺(9.8g,97.75mmol)。室温搅拌1h后逐步升温至120℃反应2h。TLC监测至反应结束,在反应液加入水和乙酸乙酯萃取,合并有机相,用无水硫酸钠干燥,过滤浓缩经硅胶柱色谱分离(石油醚/乙酸乙酯(v/v)=1/1)纯化得到标题化合物2-氯-9-(2-异丙基-4-甲基吡啶-3-基)-7,9-二氢-8H-嘌呤-8-酮(20c)(白色固体,18g,产率75%)。Dissolve 2-chloro-4-((2-isopropyl-4-methylpyridin-3-yl)amino)pyrimidine-5-carboxylic acid 20b (25.00g, 81.5mmol) in tetrahydrofuran, and then lower the temperature To 0°C, add diphenyl azide phosphate (24.6 g, 89.5 mmol) and triethylamine (9.8 g, 97.75 mmol). After stirring at room temperature for 1 hour, the temperature was gradually increased to 120°C for 2 hours. The reaction was monitored by TLC until the end of the reaction. Water and ethyl acetate were added to the reaction solution for extraction, the organic phases were combined, dried over anhydrous sodium sulfate, filtered and concentrated and separated by silica gel column chromatography (petroleum ether/ethyl acetate (v/v) = 1/ 1) Purified to obtain the title compound 2-chloro-9-(2-isopropyl-4-methylpyridin-3-yl)-7,9-dihydro-8H-purin-8-one (20c) (white solid , 18g, yield 75%).
1H NMR(400MHz,DMSO-d6)δ12.06(s,1H),8.59(d,1H),8.33(s,1H),7.35(dd,1H),2.80-2.73(m,1H),2.05(s,3H),1.09(dd,6H)。1H NMR(400MHz,DMSO-d6)δ12.06(s,1H),8.59(d,1H),8.33(s,1H),7.35(dd,1H),2.80-2.73(m,1H),2.05( s, 3H), 1.09 (dd, 6H).
LC-MS m/z(ESI)=304.10[M+1]。LC-MS m/z(ESI)=304.10[M+1].
第四步:the fourth step:
2-氯-9-(2-异丙基-4-甲基吡啶-3-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(20d)2-Chloro-9-(2-isopropyl-4-methylpyridin-3-yl)-7-methyl-7,9-dihydro-8H-purin-8-one (20d)
2-chloro-9-(2-isopropyl-4-methylpyridin-3-yl)-7-methyl-7,9-dihydro-8H-purin-8-one2-chloro-9-(2-isopropyl-4-methylpyridin-3-yl)-7-methyl-7,9-dihydro-8H-purin-8-one
将2-氯-9-(2-异丙基-4-甲基吡啶-3-基)-7,9-二氢-8H-嘌呤-8-酮20c(500mg,1.65mmol),溶解于N,N-二甲基甲酰胺(10mL),在0℃下加入碳酸铯(536mg,1.65mmol)和硫酸二甲酯(269mg,2.14mmol),0℃搅拌1.5h。TLC监测至反应结束,向反应液中加入水和乙酸乙酯萃取3次,有机相用无水硫酸钠干燥,浓缩后用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=1/1)得到标题化合物2-氯-9-(2-异丙基-4-甲基吡啶-3-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(20d)(白色固体,258mg,产率49.32%)。Dissolve 2-chloro-9-(2-isopropyl-4-methylpyridin-3-yl)-7,9-dihydro-8H-purin-8-one 20c (500mg, 1.65mmol) in N , N-Dimethylformamide (10mL), add cesium carbonate (536mg, 1.65mmol) and dimethyl sulfate (269mg, 2.14mmol) at 0°C, and stir for 1.5h at 0°C. TLC monitors to the end of the reaction, adds water and ethyl acetate to the reaction solution to extract 3 times, the organic phase is dried over anhydrous sodium sulfate, concentrated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v) = 1/1) Obtain the title compound 2-chloro-9-(2-isopropyl-4-methylpyridin-3-yl)-7-methyl-7,9-dihydro-8H-purin-8-one (20d) (white solid, 258 mg, yield 49.32%).
1H NMR(400MHz,DMSO-d6)δ8.60(d,1H),8.56(s,1H),7.36(d,1H),3.49(s,3H),2.71-2.78(m,1H),2.04(s,3H),1.10(d,3H),1.06(d,3H)。1H NMR(400MHz,DMSO-d6)δ8.60(d,1H),8.56(s,1H),7.36(d,1H),3.49(s,3H),2.71-2.78(m,1H),2.04( s, 3H), 1.10 (d, 3H), 1.06 (d, 3H).
LC-MS m/z(ESI)=318.10[M+1]。LC-MS m/z(ESI)=318.10[M+1].
第五步:the fifth step:
9-(2-异丙基-4-甲基吡啶-3-基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8酮(化合物20)9-(2-isopropyl-4-methylpyridin-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino) -7,9-dihydro-8H-purin-8one (Compound 20)
9-(2-isopropyl-4-methylpyridin-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(2-isopropyl-4-methylpyridin-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin -8-one
将2-氯-9-(2-异丙基-4-甲基吡啶-3-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮20d(80mg,0.25mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(75mg,0.50mmol)、碳酸铯(164mg,0.50mmol)和Brettphos G3 Pd(23mg,0.025mmol)加入干燥的反应管,N 2置换三次,然后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温,使用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化,得到标题化合物9-(2-异丙基-4-甲基吡啶-3-基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8酮(化合物20)(浅粉色固体,32mg,产率29.52%)。 The 2-chloro-9-(2-isopropyl-4-methylpyridin-3-yl)-7-methyl-7,9-dihydro-8H-purin-8-one 20d (80mg, 0.25mmol ), 6-methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (75mg, 0.50mmol), cesium carbonate (164mg, 0.50mmol) and Brettphos G3 Pd (23mg, 0.025mmol) were added to the dry The reaction tube was replaced with N 2 three times, then dry 1,4-dioxane (1 mL) was added, and the reaction was carried out at 110°C for 5 hours. TLC monitors until the reaction is complete, cools to room temperature, uses silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to separate and purify to obtain the title compound 9-(2-isopropyl-4-methyl) Pyridin-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purine-8 Ketone (Compound 20) (light pink solid, 32 mg, yield 29.52%).
1H NMR(600MHz,DMSO-d6)δ8.53(d,1H),8.37(s,1H),8.08(s,1H),7.31(d,1H),7.07(s,1H),6.56(s,1H),4.46(t,2H),3.39(s,3H),3.06(t,2H),2.83-2.75(m,1H),2.06(s,3H),2.03(s,3H),1.14–1.08(m,6H)。1H NMR(600MHz,DMSO-d6)δ8.53(d,1H), 8.37(s,1H), 8.08(s,1H), 7.31(d,1H), 7.07(s,1H), 6.56(s, 1H), 4.46(t, 2H), 3.39(s, 3H), 3.06(t, 2H), 2.83-2.75(m, 1H), 2.06(s, 3H), 2.03(s, 3H), 1.14-1.08 (m,6H).
LC-MS m/z(ESI)=431.20[M+1]。LC-MS m/z(ESI)=431.20[M+1].
实施例21Example 21
9-(顺-3-羟基环丁基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物21)9-(cis-3-hydroxycyclobutyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro -8H-purin-8-one (Compound 21)
9-(cis-3-hydroxycyclobutyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(cis-3-hydroxycyclobutyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one
第一步:first step:
2-氯-4-((顺-3-羟基环丁基)氨基)嘧啶-5-羧酸乙酯(21a)Ethyl 2-chloro-4-((cis-3-hydroxycyclobutyl)amino)pyrimidine-5-carboxylate (21a)
ethyl 2-chloro-4-((cis-3-hydroxycyclobutyl)amino)pyrimidine-5-carboxylateethyl 2-chloro-4-((cis-3-hydroxycyclobutyl)amino)pyrimidine-5-carboxylate
将(1s,3s)-3-氨基环丁烷-1-醇盐酸盐(3.00g,34.48mmol)溶于乙腈(25mL)中,0℃搅拌下加入碳酸钾(14.30g,103.45mmol)、2,4-二氯-5-嘧啶甲酸乙酯1a(11.43g,51.72mmol),升温至常温搅拌反应1h,TLC监测至反应结束,硅藻土过滤后用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=2:1)纯化得标题化合物2-氯-4-((顺-3-羟基环丁基)氨 基)嘧啶-5-羧酸乙酯(21a)(白色固体,3.3g,产率32.91%)。(1s, 3s)-3-aminocyclobutane-1-ol hydrochloride (3.00g, 34.48mmol) was dissolved in acetonitrile (25mL), and potassium carbonate (14.30g, 103.45mmol) was added while stirring at 0°C, Ethyl 2,4-dichloro-5-pyrimidinecarboxylate 1a (11.43g, 51.72mmol), heated to room temperature and stirred for 1h, monitored by TLC until the end of the reaction, filtered through diatomaceous earth and purified by silica gel column chromatography (petroleum ether/ Ethyl acetate (v/v)=2:1) was purified to obtain the title compound 2-chloro-4-((cis-3-hydroxycyclobutyl)amino)pyrimidine-5-carboxylic acid ethyl ester (21a) (white solid) , 3.3g, yield 32.91%).
1H NMR(400MHz,DMSO-d6)δ8.60(s,1H),8.41(d,1H),4.96(s,1H),4.31(q,2H),4.08–3.97(m,1H),3.94–3.84(m,1H),2.71-2.63(m,2H),1.88–1.80(m,2H),1.33–1.29(m,3H)。1H NMR(400MHz,DMSO-d6)δ8.60(s,1H),8.41(d,1H),4.96(s,1H),4.31(q,2H),4.08--3.97(m,1H),3.94-- 3.84 (m, 1H), 2.71-2.63 (m, 2H), 1.88-1.80 (m, 2H), 1.33-1.29 (m, 3H).
LC-MS m/z(ESI)=272.00[M+1]。LC-MS m/z(ESI)=272.00[M+1].
第二步:The second step:
2-氯-4-((顺-3-羟基环丁基)氨基)嘧啶-5-羧酸(21b)2-chloro-4-((cis-3-hydroxycyclobutyl)amino)pyrimidine-5-carboxylic acid (21b)
2-chloro-4-((cis-3-hydroxycyclobutyl)amino)pyrimidine-5-carboxylate2-chloro-4-((cis-3-hydroxycyclobutyl)amino)pyrimidine-5-carboxylate
将2-氯-4-(((1s,3s)-3-羟基环丁基)氨基)嘧啶-5-羧酸乙酯21a(3.30g,12.14mmol)溶解于四氢呋喃/水(30mL/15mL)中,加入氢氧化锂一水合物(1.53g,36.44mmol),常温搅拌反应2h,TLC监测至反应结束,浓缩蒸发掉四氢呋喃后加入2N HCl调节pH至3-4左右,有白色固体析出,过滤,滤饼用水以及石油醚/乙酸乙酯(v/v=10/1)洗两次得标题化合物2-氯-4-((顺-3-羟基环丁基)氨基)嘧啶-5-羧酸(21b)(白色固体,2.44g,产率82.45%)。Dissolve 2-chloro-4-(((1s,3s)-3-hydroxycyclobutyl)amino)pyrimidine-5-carboxylic acid ethyl ester 21a (3.30g, 12.14mmol) in tetrahydrofuran/water (30mL/15mL) Add lithium hydroxide monohydrate (1.53g, 36.44mmol), stir the reaction at room temperature for 2h, monitor by TLC until the reaction is over, concentrate and evaporate the tetrahydrofuran, add 2N HCl to adjust the pH to about 3-4, white solid precipitates out, filter , The filter cake was washed twice with water and petroleum ether/ethyl acetate (v/v=10/1) to obtain the title compound 2-chloro-4-((cis-3-hydroxycyclobutyl)amino)pyrimidine-5-carboxy Acid (21b) (white solid, 2.44 g, yield 82.45%).
1H NMR(400MHz,DMSO-d6)δ8.61(d,1H),8.57(s,1H),4.43-4.20(m,1H),4.07–3.96(m,1H),3.93–3.83(m,1H),2.72-2.63(m,2H),1.87–1.77(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.61(d,1H),8.57(s,1H),4.43-4.20(m,1H),4.07-3.96(m,1H),3.93-3.83(m,1H) ), 2.72-2.63 (m, 2H), 1.87-1.77 (m, 2H).
LC-MS m/z(ESI)=244.00[M+1]。LC-MS m/z(ESI)=244.00[M+1].
第三步:third step:
2-氯-9-(顺-3-羟基环丁基)-7,9-二氢-8H-嘌呤-8-酮(21c)2-Chloro-9-(cis-3-hydroxycyclobutyl)-7,9-dihydro-8H-purin-8-one (21c)
2-chloro-9-(cis-3-hydroxycyclobutyl)-7,9-dihydro-8H-purin-8-one2-chloro-9-(cis-3-hydroxycyclobutyl)-7,9-dihydro-8H-purin-8-one
将2-氯-4-(((1s,3s)-3-羟基环丁基)氨基)嘧啶-5-羧酸21b(2.44g,10.01mmol)用N,N-二甲基乙酰胺(20mL)溶解,常温搅拌下加入三乙胺(1.39mL,10.01mmol)和叠氮磷酸二苯酯(2.16mL,10.01mmol)反应2h,升温至110℃回流反应2.5h。TLC监测至反应结束,反应液中加水、二氯甲烷进行萃取,浓缩有机层得标题化合物2-氯-9-(顺-3-羟基环丁基)-7,9-二氢-8H-嘌呤-8-酮(21c)(白色固体,0.90g,产率37.35%)。Use 2-chloro-4-(((1s,3s)-3-hydroxycyclobutyl)amino)pyrimidine-5-carboxylic acid 21b (2.44g, 10.01mmol) with N,N-dimethylacetamide (20mL ) Was dissolved, triethylamine (1.39mL, 10.01mmol) and diphenyl azide phosphate (2.16mL, 10.01mmol) were added under stirring at room temperature and reacted for 2h, and the temperature was raised to 110°C and refluxed for 2.5h. TLC monitors to the end of the reaction, the reaction solution is extracted with water and dichloromethane, and the organic layer is concentrated to obtain the title compound 2-chloro-9-(cis-3-hydroxycyclobutyl)-7,9-dihydro-8H-purine -8-one (21c) (white solid, 0.90 g, yield 37.35%).
1H NMR(400MHz,DMSO-d6)δ11.59(s,1H),8.12(s,1H),4.31–4.20(m,1H),4.00-3.92(m,1H),3.80-3.54(m,1H),2.83–2.73(m,2H),2.58–2.51(m,2H)。1H NMR(400MHz,DMSO-d6)δ11.59(s,1H), 8.12(s,1H), 4.31-4.20(m,1H), 4.00-3.92(m,1H), 3.80-3.54(m,1H) ), 2.83–2.73(m,2H), 2.58–2.51(m,2H).
LC-MS m/z(ESI)=241.00[M+1]。LC-MS m/z(ESI)=241.00[M+1].
第四步:the fourth step:
2-氯-9-(顺-3-羟基环丁基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(21d)2-Chloro-9-(cis-3-hydroxycyclobutyl)-7-methyl-7,9-dihydro-8H-purin-8-one (21d)
2-chloro-9-(cis-3-hydroxycyclobutyl)-7-methyl-7,9-dihydro-8H-purin-8-one2-chloro-9-(cis-3-hydroxycyclobutyl)-7-methyl-7,9-dihydro-8H-purin-8-one
将2-氯-9-((1s,3s)-3-羟基环丁基)-7,9-二氢-8H-嘌呤-8-酮21c(0.70g,1.25mmol)用N,N-二甲基甲酰胺(10mL)溶解,0℃搅拌下加入硫酸二甲酯(0.28mL,1.25mmol)和碳酸铯(1.42g,4.36mmol)反应2h。TLC监测至反应结束,把反应液缓慢滴加到冰水中搅拌,加入乙酸乙酯进行萃取,浓缩有机层经硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=1:1)纯化得标题化合物2-氯-9-(顺-3-羟基环丁基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(21d)(白色固体,0.32g,产率41.20%)。Use 2-chloro-9-((1s,3s)-3-hydroxycyclobutyl)-7,9-dihydro-8H-purin-8-one 21c (0.70g, 1.25mmol) with N,N-two Methylformamide (10 mL) was dissolved, and dimethyl sulfate (0.28 mL, 1.25 mmol) and cesium carbonate (1.42 g, 4.36 mmol) were added under stirring at 0°C to react for 2 h. TLC monitors until the reaction is over, the reaction solution is slowly added dropwise to ice water and stirred, ethyl acetate is added for extraction, the concentrated organic layer is separated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)=1:1) Purified to obtain the title compound 2-chloro-9-(cis-3-hydroxycyclobutyl)-7-methyl-7,9-dihydro-8H-purin-8-one (21d) (white solid, 0.32g, Yield 41.20%).
1H NMR(400MHz,DMSO-d6)δ8.33(s,1H),5.31(d,1H),4.34–4.23(m,1H),4.02-3.91(m,1H),3.34(s,3H),2.81-2.72(m,2H),2.58–2.51(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.33(s,1H),5.31(d,1H),4.34-4.23(m,1H),4.02-3.91(m,1H),3.34(s,3H), 2.81-2.72 (m, 2H), 2.58-2.51 (m, 2H).
LC-MS m/z(ESI)=255.00[M+1]。LC-MS m/z(ESI)=255.00[M+1].
第五步:the fifth step:
9-(顺-3-羟基环丁基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物21)9-(cis-3-hydroxycyclobutyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro -8H-purin-8-one (Compound 21)
9-(cis-3-hydroxycyclobutyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(cis-3-hydroxycyclobutyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one
将2-氯-9-((1s,3s)-3-羟基环丁基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮21d(50mg,0.20mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(59mg,0.40mmol)、碳酸铯(128mg, 0.4mmol)和Brettphos G3 Pd(19mg,0.020mmol)加入干燥的反应管,N 2置换三次,然后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温,使用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化,得到标题化合物9-((1s,3s)-3-羟基环丁基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物21)(浅黄色固体,22mg,产率30.50%)。 The 2-chloro-9-((1s,3s)-3-hydroxycyclobutyl)-7-methyl-7,9-dihydro-8H-purin-8-one 21d (50mg, 0.20mmol), 6 -Methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (59mg, 0.40mmol), cesium carbonate (128mg, 0.4mmol) and Brettphos G3 Pd (19mg, 0.020mmol) were added to the dry reaction tube , N 2 was replaced three times, and then dry 1,4-dioxane (1 mL) was added, and the reaction was carried out at 110° C. for 5 h. TLC monitors until the reaction is complete, cool to room temperature, use silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to separate and purify to obtain the title compound 9-((1s, 3s)-3-hydroxy Butyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one( Compound 21) (light yellow solid, 22 mg, yield 30.50%).
1H NMR(400MHz,DMSO-d6)δ8.14(s,1H),7.95(s,1H),7.26(s,1H),6.60(s,1H),5.11(d,1H),4.48(t,2H),4.23–4.14(m,1H),3.94-3.88(m,1H),3.25(s,3H),3.13(t,2H),2.82–2.73(m,2H),2.54-2.44(m,2H),2.12(s,3H)。1H NMR (400MHz, DMSO-d6) δ 8.14 (s, 1H), 7.95 (s, 1H), 7.26 (s, 1H), 6.60 (s, 1H), 5.11 (d, 1H), 4.48 (t, 2H), 4.23 - 4.14 (m, 1H), 3.94-3.88 (m, 1H), 3.25 (s, 3H), 3.13 (t, 2H), 2.82 - 2.73 (m, 2H), 2.54-2.44 (m, 2H), 2.12(s, 3H).
LC-MS m/z(ESI)=368.10[M+1]。LC-MS m/z(ESI)=368.10[M+1].
实施例22Example 22
9-(4,4-二氟环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物22)9-(4,4-Difluorocyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro -8H-purin-8-one (Compound 22)
9-(4,4-difluorocyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(4,4-difluorocyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one
第一步:first step:
2-氯-4-((4,4-二氟环己基)氨基)嘧啶-5-羧酸乙酯(22a)Ethyl 2-chloro-4-((4,4-difluorocyclohexyl)amino)pyrimidine-5-carboxylate (22a)
ethyl-2-chloro-4-((4,4-difluorocyclohexyl)amino)pyrimidine-5-carboxylateethyl-2-chloro-4-((4,4-difluorocyclohexyl)amino)pyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(6.5g,29.41mmol)、4,4-二氟环己-1-胺盐酸盐(5.0g,29.41mmol)溶解于乙腈(100mL),常温搅拌下加入碳酸钾(10.16g,73.52mmol)反应4h。TLC监测至反应完毕后过滤,滤渣用乙酸乙酯清洗,将滤液浓缩通过硅胶柱色谱分离提纯(正己烷/乙酸乙酯(v/v)=1:1)得标题化合物2-氯-4-((四氢-2H-吡喃-4-基)氨基)嘧啶-5-羧酸乙酯(22a)(白色固体,8.0g,产率85.10%)。2,4-Dichloropyrimidine-5-carboxylic acid ethyl ester 1a (6.5g, 29.41mmol), 4,4-difluorocyclohexyl-1-amine hydrochloride (5.0g, 29.41mmol) were dissolved in acetonitrile ( 100mL), potassium carbonate (10.16g, 73.52mmol) was added under stirring at room temperature to react for 4h. After the reaction was monitored by TLC, it was filtered and the residue was washed with ethyl acetate. The filtrate was concentrated and separated and purified by silica gel column chromatography (n-hexane/ethyl acetate (v/v)=1:1) to obtain the title compound 2-chloro-4- ((Tetrahydro-2H-pyran-4-yl)amino)pyrimidine-5-carboxylic acid ethyl ester (22a) (white solid, 8.0 g, yield 85.10%).
1H NMR(400MHz,DMSO-d6)δ8.85(s,1H),8.11(d,1H),4.34-4.25(m,2H),4.21-4.10(m,1H),2.11-1.91(m,6H),1.71-1.57(m,2H),1.23(t,3H)。1H NMR(400MHz,DMSO-d6)δ8.85(s,1H),8.11(d,1H),4.34-4.25(m,2H),4.21-4.10(m,1H),2.11-1.91(m,6H) ), 1.71-1.57 (m, 2H), 1.23 (t, 3H).
LCMS m/z(ESI)=320.10[M+l]。LCMS m/z(ESI)=320.10[M+1].
第二步:The second step:
2-氯-4-((4,4-二氟环己基)氨基)嘧啶-5-羧酸(22b)2-chloro-4-((4,4-difluorocyclohexyl)amino)pyrimidine-5-carboxylic acid (22b)
2-chloro-4-((4,4-difluorocyclohexyl)amino)pyrimidine-5-carboxylic acid2-chloro-4-((4,4-difluorocyclohexyl)amino)pyrimidine-5-carboxylic acid
将2-氯-4-((4,4-二氟环己基)氨基)嘧啶-5-羧酸乙酯22a(4g,12.47mmol)溶解于四氢呋喃/水(50mL/50mL)中,加入氢氧化锂(597.22mg,24.94mmol),室温搅拌1h。TLC监测至反应完全,旋干四氢呋喃,用2N盐酸调pH为5,有白色固体析出,过滤,滤饼用石油醚洗两次,搜集固体得到标题化合物2-氯-4-((4,4-二氟环己基)氨基)嘧啶-5-羧酸(22b)(白色固体,3.4g,产率91.53%)。Dissolve 2-chloro-4-((4,4-difluorocyclohexyl)amino)pyrimidine-5-carboxylic acid ethyl ester 22a (4g, 12.47mmol) in tetrahydrofuran/water (50mL/50mL) and add hydroxide Lithium (597.22mg, 24.94mmol), stirred at room temperature for 1h. TLC monitors until the reaction is complete, spin dry the tetrahydrofuran, adjust the pH to 5 with 2N hydrochloric acid, a white solid precipitates out, filter, wash the filter cake twice with petroleum ether, collect the solid to obtain the title compound 2-chloro-4-((4,4 -Difluorocyclohexyl)amino)pyrimidine-5-carboxylic acid (22b) (white solid, 3.4g, yield 91.53%).
1H NMR(400MHz,DMSO-d6)δ13.79(s,1H),8.60(s,1H),8.55(d,1H),4.22-4.06(m,1H),2.12-1.89(m,6H),1.72-1.56(m,2H)。1H NMR(400MHz,DMSO-d6)δ13.79(s,1H),8.60(s,1H),8.55(d,1H),4.22-4.06(m,1H),2.12-1.89(m,6H), 1.72-1.56 (m, 2H).
LCMS m/z(ESI)=292.00[M+l]。LCMS m/z(ESI)=292.00[M+1].
第三步:third step:
2-氯-9-(4,4-二氟环己基)-7,9-二氢-8H-嘌呤-8-酮(22c)2-chloro-9-(4,4-difluorocyclohexyl)-7,9-dihydro-8H-purin-8-one (22c)
2-chloro-9-(4,4-difluorocyclohexyl)-7,9-dihydro-8H-purin-8-one2-chloro-9-(4,4-difluorocyclohexyl)-7,9-dihydro-8H-purin-8-one
将2-氯-4-((四氢-2H-吡喃-4-基)氨基)嘧啶-5-羧酸22b(3.34g,11.45mmol)溶解于二甲基乙酰胺(50mL),加入三乙胺(1.6mL,11.45mmol)、叠氮磷酸二苯酯(1.22mL,11.45mmol),随后逐步升温至120℃,搅拌1.5h。TLC监测反应完毕,将反应液倒入冰水中,过滤搜集固体,水洗3次,真空浓缩干燥得标题化合物2-氯-9-(4,4-二氟环己基)-7,9-二氢-8H-嘌呤-8-酮(22c)(白色固体,2.2g,产率78.5%)。Dissolve 2-chloro-4-((tetrahydro-2H-pyran-4-yl)amino)pyrimidine-5-carboxylic acid 22b (3.34g, 11.45mmol) in dimethylacetamide (50mL), add three Ethylamine (1.6mL, 11.45mmol), diphenyl azide phosphate (1.22mL, 11.45mmol), then gradually heated to 120°C and stirred for 1.5h. TLC monitors the completion of the reaction, the reaction solution is poured into ice water, the solid is collected by filtration, washed with water 3 times, concentrated and dried in vacuo to obtain the title compound 2-chloro-9-(4,4-difluorocyclohexyl)-7,9-dihydro -8H-purin-8-one (22c) (white solid, 2.2 g, yield 78.5%).
1H NMR(400MHz,DMSO-d6)δ11.66(s,1H),8.13(s,1H),4.46-4.36(m,1H),2.17-1.94(m,6H),1.87-1.80(m,2H)。1H NMR(400MHz,DMSO-d6)δ11.66(s,1H),8.13(s,1H),4.46-4.36(m,1H),2.17-1.94(m,6H),1.87-1.80(m,2H) ).
LCMS m/z(ESI)=289.10[M+l]。LCMS m/z(ESI)=289.10[M+1].
第四步:the fourth step:
2-氯-7-甲基-9-(4,4-二氟环己基)-7,9-二氢-8H-嘌呤-8-酮(22d)2-chloro-7-methyl-9-(4,4-difluorocyclohexyl)-7,9-dihydro-8H-purin-8-one (22d)
2-chloro-9-(4,4-difluorocyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one2-chloro-9-(4,4-difluorocyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one
将2-氯-9-(4,4-二氟环己基)-7,9-二氢-8H-嘌呤-8-酮22c(2.62g,9.08mmol)溶解于二甲基甲酰胺(50mL),在0℃下加入硫酸二甲酯(1.14g,9.08mmol)和碳酸铯(4.44g,13.61mmol),0℃搅拌1h。TLC监测反应结束,将反应液倒入冰水中,有固体析出,过滤、搜集固体得到标题化合物2-氯-7-甲基-9-(4,4-二氟环己基)-7,9-二氢-8H-嘌呤-8-酮(22d)(白色固体,2.2g,产率80%)。Dissolve 2-chloro-9-(4,4-difluorocyclohexyl)-7,9-dihydro-8H-purin-8-one 22c (2.62g, 9.08mmol) in dimethylformamide (50mL) Add dimethyl sulfate (1.14g, 9.08mmol) and cesium carbonate (4.44g, 13.61mmol) at 0°C, and stir at 0°C for 1h. TLC monitors the end of the reaction. Pour the reaction solution into ice water. A solid precipitates out. The solid is filtered and collected to obtain the title compound 2-chloro-7-methyl-9-(4,4-difluorocyclohexyl)-7,9- Dihydro-8H-purin-8-one (22d) (white solid, 2.2 g, yield 80%).
1H NMR(400MHz,DMSO-d6)δ8.37(s,1H),4.55–4.39(m,1H),3.33(s,3H),2.21–2.01(m,6H),1.90–1.79(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.37(s,1H),4.55-4.39(m,1H),3.33(s,3H),2.21-2.01(m,6H),1.90-1.79(m,2H) ).
LCMS m/z(ESI)=303.10[M+l]。LCMS m/z(ESI)=303.10[M+1].
第五步:the fifth step:
9-(4,4-二氟环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物22)9-(4,4-Difluorocyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro -8H-purin-8-one (Compound 22)
9-(4,4-difluorocyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(4,4-difluorocyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one
将2-氯-7-甲基-9-(4,4-二氟环己基)-7,9-二氢-8H-嘌呤-8-酮22d(100mg,0.33mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(99mg,0.66mmol)、碳酸铯(215mg,0.66mmol)和Brettphos G3 Pd(30mg,0.033mmol)加入干燥的反应管,N 2置换三次,然后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。冷却至室温,使用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化,得到标题化合物9-(4,4-二氟环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物22)(灰白色固体,62mg,产率45.18%)。 The 2-chloro-7-methyl-9-(4,4-difluorocyclohexyl)-7,9-dihydro-8H-purin-8-one 22d (100mg, 0.33mmol), 6-methyl- 2,3-Dihydrobenzofuran-5-amine intermediate 1 (99mg, 0.66mmol), cesium carbonate (215mg, 0.66mmol) and Brettphos G3 Pd (30mg, 0.033mmol) were added to the dry reaction tube and replaced with N 2 Three times, then dry 1,4-dioxane (1 mL) was added and reacted at 110° C. for 5 h. Cooled to room temperature, and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 9-(4,4-difluorocyclohexyl)-7-methyl-2 -((6-Methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one (Compound 22) (off-white solid, 62mg, product Rate 45.18%).
1H NMR(400MHz,DMSO-d6)δ8.18(s,1H),7.98(s,1H),7.24(s,1H),6.60(s,1H),4.48(t,2H),4.38–4.28(m,1H),3.27(s,3H),3.11(t,2H),2.56–2.43(m,2H),2.12(s,3H),2.11–1.92(m,4H),1.77(d,2H)。1H NMR(400MHz,DMSO-d6)δ8.18(s,1H),7.98(s,1H), 7.24(s,1H), 6.60(s,1H), 4.48(t,2H), 4.38-4.28( m, 1H), 3.27 (s, 3H), 3.11 (t, 2H), 2.56-2.43 (m, 2H), 2.12 (s, 3H), 2.11-1.92 (m, 4H), 1.77 (d, 2H) .
LC-MS m/z(ESI)=416.20[M+1]。LC-MS m/z(ESI)=416.20[M+1].
实施例23Example 23
4-(7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-8-氧代7,8-二氢-9H-嘌呤-9-基)双环[2.2.2]辛烷-1-腈(化合物23)4-(7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-8-oxo 7,8-dihydro-9H-purine-9 -Yl)bicyclo[2.2.2]octane-1-carbonitrile (compound 23)
4-(7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-8-oxo-7,8-dihydro-9H-purin-9-yl)bicyclo[2.2.2]octane-1-carbonitrile4-(7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-8-oxo-7,8-dihydro-9H-purin-9-yl)bicyclo[2.2. 2]octane-1-carbonitrile
第一步:first step:
4-氨基甲酰基双环[2.2.2]辛烷-1-甲酸甲酯(23b)Methyl 4-carbamoylbicyclo[2.2.2]octane-1-carboxylate (23b)
methyl 4-carbamoylbicyclo[2.2.2]octane-1-carboxylatemethyl 4-carbamoylbicyclo[2.2.2]octane-1-carboxylate
将4-(甲氧羰基)双环[2.2.2]辛烷-1-羧酸23a(10.0g,47.12mmol)溶解于无水二氯甲烷(200mL),冰浴下加入2-(7-氮杂苯并三氮唑)-N,N,N',N'-四甲基脲六氟磷酸酯(18.81g,49.47mmol),保持温度反应30min,加入N,N-二异丙基乙胺(24.62mL,141.35mmol),随后缓慢多次加入氯化铵固体(3.78g,70.67mmol)。反应逐步恢复至室温并过夜反应。TLC监测反应结束,反应液直接用0.5N盐酸溶液100mL,分液后有机相依次用水和饱和食盐水各100mL,干燥,浓缩后得目标化合物4-氨基甲酰基双环[2.2.2]辛烷-1-甲酸甲酯(23b)(白色固体,21g,粗品)。Dissolve 4-(methoxycarbonyl)bicyclo[2.2.2]octane-1-carboxylic acid 23a (10.0g, 47.12mmol) in anhydrous dichloromethane (200mL), add 2-(7-nitrogen under ice bath) Heterobenzotriazole)-N,N,N',N'-tetramethylurea hexafluorophosphate (18.81g, 49.47mmol), keep the temperature for 30min, add N,N-diisopropylethylamine (24.62mL, 141.35mmol), then slowly added ammonium chloride solid (3.78g, 70.67mmol) several times. The reaction gradually returned to room temperature and reacted overnight. TLC monitors the end of the reaction, the reaction solution is directly used with 0.5N hydrochloric acid solution 100mL, after separation, the organic phase is successively 100mL with water and saturated brine, dried, and concentrated to obtain the target compound 4-carbamoylbicyclo[2.2.2]octane- Methyl 1-formate (23b) (white solid, 21 g, crude product).
1H NMR(400MHz,DMSO-d6)δ6.94(s,1H),6.73(s,1H),3.57(s,3H),1.72-1.63(m,12H)。1H NMR (400MHz, DMSO-d6) δ 6.94 (s, 1H), 6.73 (s, 1H), 3.57 (s, 3H), 1.72-1.63 (m, 12H).
LC-MS m/z(ESI)=212.10[M+1]。LC-MS m/z(ESI)=212.10[M+1].
第二步:The second step:
4-氰基双环[2.2.2]辛烷-1-羧酸甲酯(23c)4-cyanobicyclo[2.2.2]octane-1-carboxylic acid methyl ester (23c)
methyl 4-cyanobicyclo[2.2.2]octane-1-carboxylatemethyl 4-cyanobicyclo[2.2.2]octane-1-carboxylate
将4-氨基甲酰基双环[2.2.2]辛烷-1-甲酸甲酯23b(21g,99.40mmol)溶于二氯甲烷(200mL),冰浴加入吡啶(16.02mL,198.81mmol),三氟乙酸酐(21.00ml,149.10mmol), 保持温度继续反应1h。TLC监测反应结束,直接将反应液过滤,滤饼用100ml二氯甲烷清洗,合并有机相,随后用1N盐酸溶液,水,饱和食盐水各100mL,干燥,浓缩后经柱层析分离后得目标产物4-氰基双环[2.2.2]辛烷-1-羧酸甲酯(23c)(白色固体,5.3g,60mg,两步产率58.21%)。Dissolve 4-carbamoyl bicyclo[2.2.2]octane-1-carboxylic acid methyl ester 23b (21g, 99.40mmol) in dichloromethane (200mL), add pyridine (16.02mL, 198.81mmol) in an ice bath, trifluoro Acetic anhydride (21.00ml, 149.10mmol), keep the temperature and continue the reaction for 1h. TLC monitors the end of the reaction, directly filter the reaction solution, wash the filter cake with 100ml of dichloromethane, combine the organic phases, then use 1N hydrochloric acid solution, water, and saturated brine each with 100ml, dry, concentrate and separate by column chromatography to obtain the target The product 4-cyanobicyclo[2.2.2]octane-1-carboxylic acid methyl ester (23c) (white solid, 5.3g, 60mg, two-step yield 58.21%).
1H NMR(400MHz,CDCl3-d6)δ3.66(s,3H),1.98-1.94(m,6H),1.86-1.82(m,6H)。1H NMR (400MHz, CDCl3-d6) δ 3.66 (s, 3H), 1.98-1.94 (m, 6H), 1.86-1.82 (m, 6H).
LC-MS m/z(ESI)=194.10[M+1]。LC-MS m/z(ESI)=194.10[M+1].
第三步:third step:
4-氰基双环[2.2.2]辛烷-1-羧酸(23d)4-cyanobicyclo[2.2.2]octane-1-carboxylic acid (23d)
4-cyanobicyclo[2.2.2]octane-1-carboxylic acid4-cyanobicyclo[2.2.2]octane-1-carboxylic acid
将4-氰基双环[2.2.2]辛烷-1-羧酸甲酯23c(5.3g,27.43mmol)溶解于四氢呋喃50mL,水50mL中,加入氢氧化锂(1.73g,41.14mmol),室温搅拌1h。TLC监测反应完全,浓缩除去四氢呋喃,用2N盐酸调pH为5,有白色固体析出,过滤,滤饼用石油醚洗两次,搜集固体得到标题化合物4-氰基双环[2.2.2]辛烷-1-羧酸(23d)(白色固体,5.0g,粗品)。Dissolve 4-cyanobicyclo[2.2.2]octane-1-carboxylic acid methyl ester 23c (5.3g, 27.43mmol) in tetrahydrofuran 50mL, water 50mL, add lithium hydroxide (1.73g, 41.14mmol), room temperature Stir for 1h. TLC monitors that the reaction is complete, concentrates to remove tetrahydrofuran, adjusts the pH to 5 with 2N hydrochloric acid, a white solid is precipitated, filtered, the filter cake is washed twice with petroleum ether, and the solid is collected to obtain the title compound 4-cyanobicyclo[2.2.2]octane -1-carboxylic acid (23d) (white solid, 5.0 g, crude product).
1H NMR(400MHz,DMSO-d6)δ12.23(s,1H),1.89-1.85(m,6H),1.72-1.68(m,6H)。1H NMR (400MHz, DMSO-d6) δ 12.23 (s, 1H), 1.89-1.85 (m, 6H), 1.72-1.68 (m, 6H).
LC-MS m/z(ESI)=180.10[M+1]。LC-MS m/z(ESI)=180.10[M+1].
第四步:the fourth step:
4-氨基双环[2.2.2]辛烷-1-腈盐酸盐(23e)4-aminobicyclo[2.2.2]octane-1-carbonitrile hydrochloride (23e)
4-aminobicyclo[2.2.2]octane-1-carbonitrile hydrochloride4-aminobicyclo[2.2.2]octane-1-carbonitrile hydrochloride
将4-氰基双环[2.2.2]辛烷-1-羧酸23d(5.0g,27.90mmol)溶于甲苯(60mL)中,冰浴加入叠氮磷酸二苯酯(6.01mL,27.90mmol)和三乙胺(3.88mL,27.90mmol),将反应液室温搅拌1h后升温至90度继续反应3h。TLC监测至反应结束,冷却至室温,缓慢倒入100mL 1N盐酸溶液,出现大量固体,过滤,收集固体,且用乙酸乙酯(150mL)打浆,真空干燥得标题化合物4-氨基双环[2.2.2]辛烷-1-腈盐酸盐(23e)(白色固体,7.3g,粗品,收率80%)。Dissolve 4-cyanobicyclo[2.2.2]octane-1-carboxylic acid 23d (5.0g, 27.90mmol) in toluene (60mL), add diphenyl azide phosphate (6.01mL, 27.90mmol) in an ice bath And triethylamine (3.88mL, 27.90mmol), the reaction solution was stirred at room temperature for 1h, then the temperature was raised to 90°C and the reaction was continued for 3h. TLC monitors until the reaction is complete, cool to room temperature, slowly pour 100mL 1N hydrochloric acid solution, a large amount of solids appear, filter, collect the solids, beat with ethyl acetate (150mL), and dry in vacuo to obtain the title compound 4-aminobicyclo[2.2.2 ] Octane-1-nitrile hydrochloride (23e) (white solid, 7.3 g, crude product, yield 80%).
1H NMR(401MHz,DMSO-d6)δ8.45(s,2H),2.01–1.97(m,6H),1.81-1.76(m,6H)。1H NMR(401MHz,DMSO-d6)δ8.45(s,2H),2.01–1.97(m,6H),1.81-1.76(m,6H).
LC-MS m/z(ESI)=151.20[M+1]。LC-MS m/z(ESI)=151.20[M+1].
第五步:the fifth step:
2-氯-4-((4-氰基双环[2.2.2]辛烷-1-基)氨基)嘧啶-5-羧酸乙酯(23f)2-chloro-4-((4-cyanobicyclo[2.2.2]octane-1-yl)amino)pyrimidine-5-carboxylic acid ethyl ester (23f)
ethyl 2-chloro-4-((4-cyanobicyclo[2.2.2]octan-1-yl)amino)pyrimidine-5-carboxylateethyl 2-chloro-4-((4-cyanobicyclo[2.2.2]octan-1-yl)amino)pyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(8.74g,39.53mmol),4-氨基双环[2.2.2]辛烷-1-腈盐酸盐23e(7.38g,39.53mmol,60%纯度),碳酸钾(21.85g,158.13mmol)溶于乙腈(200mL),反应液在室温反应16h。TLC监测反应结束,过滤,并用少量乙腈清洗固体,将滤液合并后浓缩,粗品经硅胶柱色谱分离(石油醚/乙酸乙酯(v/v)=1:1)后得标题化合物2-氯-4-((4-氰基双环[2.2.2]辛烷-1-基)氨基)嘧啶-5-羧酸乙酯(23f)(白色固体,4.0g,产率30.23%)。The 2,4-dichloropyrimidine-5-carboxylic acid ethyl ester 1a (8.74g, 39.53mmol), 4-aminobicyclo[2.2.2]octane-1-carbonitrile hydrochloride 23e (7.38g, 39.53mmol, 60% purity), potassium carbonate (21.85g, 158.13mmol) was dissolved in acetonitrile (200mL), and the reaction solution was reacted at room temperature for 16h. The end of the reaction was monitored by TLC, filtered, and the solid was washed with a small amount of acetonitrile. The filtrate was combined and concentrated. The crude product was separated by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)=1:1) to obtain the title compound 2-chloro- 4-((4-Cyanobicyclo[2.2.2]octan-1-yl)amino)pyrimidine-5-carboxylic acid ethyl ester (23f) (white solid, 4.0 g, yield 30.23%).
1H NMR(400MHz,DMSO-d6)δ8.63(s,1H),8.29(s,1H),4.30(q,2H),2.10–2.00(m,12H),1.30(t,3H)。1H NMR(400MHz,DMSO-d6)δ8.63(s,1H), 8.29(s,1H), 4.30(q,2H), 2.10–2.00(m,12H), 1.30(t,3H).
LC-MS m/z(ESI)=335.10[M+1]。LC-MS m/z(ESI)=335.10[M+1].
第六步:The sixth step:
2-氯-4-((4-氰基双环[2.2.2]辛烷-1-基)氨基)嘧啶-5-羧酸(23g)2-chloro-4-((4-cyanobicyclo[2.2.2]octane-1-yl)amino)pyrimidine-5-carboxylic acid (23g)
2-chloro-4-((4-cyanobicyclo[2.2.2]octan-1-yl)amino)pyrimidine-5-carboxylic acid2-chloro-4-((4-cyanobicyclo[2.2.2]octan-1-yl)amino)pyrimidine-5-carboxylic acid
将2-氯-4-((4-氰基双环[2.2.2]辛烷-1-基)氨基)嘧啶-5-羧酸乙酯23f(4g,11.95mmol)溶解于四氢呋喃50mL,水50mL中,加入氢氧化锂(1.01g,23.90mmol),室温搅拌1h 。TLC监测反应完全,浓缩除去四氢呋喃,用2N盐酸调pH为5,有白色固体析出,过滤,滤饼用石油醚洗两次,搜集固体得到标题化合物2-氯-4-((4-氰基双环[2.2.2]辛烷-1-基)氨基)嘧啶-5-羧酸(23g)(3.6g,白色固体,产率98.23%),直接进行下一步实验。Dissolve 2-chloro-4-((4-cyanobicyclo[2.2.2]octan-1-yl)amino)pyrimidine-5-carboxylic acid ethyl ester 23f (4g, 11.95mmol) in tetrahydrofuran 50mL, water 50mL Lithium hydroxide (1.01 g, 23.90 mmol) was added to the mixture, and stirred at room temperature for 1 h. TLC monitored the reaction to complete, concentrated to remove tetrahydrofuran, adjusted the pH to 5 with 2N hydrochloric acid, a white solid precipitated out, filtered, the filter cake was washed twice with petroleum ether, the solid was collected to obtain the title compound 2-chloro-4-((4-cyano Bicyclo[2.2.2]octane-1-yl)amino)pyrimidine-5-carboxylic acid (23g) (3.6g, white solid, yield 98.23%), proceed directly to the next experiment.
1H NMR(600MHz,DMSO-d6)δ13.85(s,1H),8.59(s,1H),8.56(s,1H),2.07–1.98(m,12H)。1H NMR (600MHz, DMSO-d6) δ 13.85 (s, 1H), 8.59 (s, 1H), 8.56 (s, 1H), 2.07-1.98 (m, 12H).
LCMS m/z(ESI)=307.10[M+l]。LCMS m/z(ESI)=307.10[M+1].
第七步:The seventh step:
4-(2-氯-8-氧代-7,8-二氢-9H-嘌呤-9-基)双环[2.2.2]辛烷-1-腈(23h)4-(2-Chloro-8-oxo-7,8-dihydro-9H-purin-9-yl)bicyclo[2.2.2]octane-1-carbonitrile (23h)
4-(2-chloro-8-oxo-7,8-dihydro-9H-purin-9-yl)bicyclo[2.2.2]octane-1-carbonitrile4-(2-chloro-8-oxo-7,8-dihydro-9H-purin-9-yl)bicyclo[2.2.2]octane-1-carbonitrile
将2-氯-4-((4-氰基双环[2.2.2]辛烷-1-基)氨基)嘧啶-5-羧酸23g(3.8g,12.39mmol)溶解于二甲基乙酰胺(50mL),加入三乙胺(1.72mL,12.39mmol)、叠氮磷酸二苯酯(2.67mL,12.39mmol),随后逐步升温至120℃,搅拌1.5h。TLC监测反应完毕,将反应液倒入冰水中,过滤搜集固体,水洗3次,真空浓缩干燥得4-(2-氯-8-氧代-7,8-二氢-9H-嘌呤-9-基)双环[2.2.2]辛烷-1-腈(23h)(3.3g,白色固体,产率87.7%)。Dissolve 23g (3.8g, 12.39mmol) of 2-chloro-4-((4-cyanobicyclo[2.2.2]octan-1-yl)amino)pyrimidine-5-carboxylic acid in dimethylacetamide ( 50mL), triethylamine (1.72mL, 12.39mmol) and diphenyl azide phosphate (2.67mL, 12.39mmol) were added, and then gradually heated to 120°C and stirred for 1.5h. TLC monitors the completion of the reaction, pour the reaction solution into ice water, filter to collect the solid, wash 3 times with water, concentrate and dry in vacuo to obtain 4-(2-chloro-8-oxo-7,8-dihydro-9H-purine-9- Yl)bicyclo[2.2.2]octane-1-carbonitrile (23h) (3.3g, white solid, yield 87.7%).
1H NMR(400MHz,DMSO-d6)δ11.61(s,1H),8.09(s,1H),2.48–2.40(m,6H),2.09–2.03(m,6H)。1H NMR(400MHz,DMSO-d6)δ11.61(s,1H), 8.09(s,1H), 2.48–2.40(m,6H), 2.09–2.03(m,6H).
LC-MS m/z(ESI)=304.20[M+l]。LC-MS m/z(ESI)=304.20[M+1].
第八步:The eighth step:
4-(2-氯-7-甲基-8-氧代7,8-二氢-9H-嘌呤-9-基)双环[2.2.2]辛烷-1-腈(23i)4-(2-Chloro-7-methyl-8-oxo 7,8-dihydro-9H-purin-9-yl)bicyclo[2.2.2]octane-1-carbonitrile (23i)
4-(2-chloro-7-methyl-8-oxo-7,8-dihydro-9H-purin-9-yl)bicyclo[2.2.2]octane-1-carbonitrile4-(2-chloro-7-methyl-8-oxo-7,8-dihydro-9H-purin-9-yl)bicyclo[2.2.2]octane-1-carbonitrile
将4-(2-氯-8-氧代-7,8-二氢-9H-嘌呤-9-基)双环[2.2.2]辛烷-1-腈23h(3.3g,11.43mmol)溶解于二甲基甲酰胺(50mL),在0℃下加入硫酸二甲酯(1.44g,11.43mmol)和碳酸铯(5.59g,17.15mmol),0度搅拌1h。TLC监测反应结束,将反应液倒入冰水中,有固体析出,过滤、搜集固体得到标题化合物4-(2-氯-7-甲基-8-氧代7,8-二氢-9H-嘌呤-9-基)双环[2.2.2]辛烷-1-腈(23i)(3.1g,白色固体,产率89.59%)。Dissolve 4-(2-chloro-8-oxo-7,8-dihydro-9H-purin-9-yl)bicyclo[2.2.2]octane-1-nitrile 23h (3.3g, 11.43mmol) in Dimethylformamide (50mL), dimethyl sulfate (1.44g, 11.43mmol) and cesium carbonate (5.59g, 17.15mmol) were added at 0°C, and stirred at 0°C for 1h. TLC monitors the end of the reaction. Pour the reaction solution into ice water. A solid precipitates out. The solid is filtered and collected to obtain the title compound 4-(2-chloro-7-methyl-8-oxo7,8-dihydro-9H-purine -9-yl)bicyclo[2.2.2]octane-1-carbonitrile (23i) (3.1 g, white solid, yield 89.59%).
1H NMR(401MHz,DMSO-d6)δ8.33(s,1H),3.29(s,3H),2.48–2.41(m,6H),2.10–2.04(m,6H)。1H NMR(401MHz,DMSO-d6)δ8.33(s,1H), 3.29(s,3H), 2.48–2.41(m,6H), 2.10–2.04(m,6H).
LC-MS m/z(ESI)=318.20[M+l]。LC-MS m/z(ESI)=318.20[M+1].
第九步:Step 9:
4-(7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-8-氧代7,8-二氢-9H-嘌呤-9-基)双环[2.2.2]辛烷-1-腈(化合物23)4-(7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-8-oxo 7,8-dihydro-9H-purine-9 -Yl)bicyclo[2.2.2]octane-1-carbonitrile (compound 23)
4-(7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-8-oxo-7,8-dihydro-9H-purin-9-yl)bicyclo[2.2.2]octane-1-carbonitrile4-(7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-8-oxo-7,8-dihydro-9H-purin-9-yl)bicyclo[2.2. 2]octane-1-carbonitrile
将4-(2-氯-7-甲基-8-氧代7,8-二氢-9H-嘌呤-9-基)双环[2.2.2]辛烷-1-腈23i(100mg,0.31mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(94mg,0.62mmol)、碳酸铯(205mg,0.62mmol)和Brettphos G3 Pd(28mg,0.031mmol)加入干燥的反应管,N 2置换三次,然后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。冷却至室温,使用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化,得到标题化合物4-(7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-8-氧代7,8-二氢-9H-嘌呤-9-基)双环[2.2.2]辛烷-1-酮(化合物23)(灰白色固体,53mg,产率39.12%)。 The 4-(2-chloro-7-methyl-8-oxo 7,8-dihydro-9H-purin-9-yl)bicyclo[2.2.2]octane-1-carbonitrile 23i (100mg, 0.31mmol ), 6-methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (94mg, 0.62mmol), cesium carbonate (205mg, 0.62mmol) and Brettphos G3 Pd (28mg, 0.031mmol) were added to the dry The reaction tube was replaced with N 2 three times, then dry 1,4-dioxane (1 mL) was added, and the reaction was carried out at 110°C for 5 hours. After cooling to room temperature, silica gel column chromatography was used for separation and purification (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 4-(7-methyl-2-((6-methyl-2, 3-Dihydrobenzofuran-5-yl)amino)-8-oxo7,8-dihydro-9H-purin-9-yl)bicyclo[2.2.2]octan-1-one (compound 23) (Off-white solid, 53 mg, yield 39.12%).
1H NMR(400MHz,DMSO-d6)δ8.16(s,1H),7.95(s,1H),7.23(s,1H),6.61(s,1H),4.50(t,2H),3.20(s,3H),3.12(t,2H),2.45–2.37(M,6H),2.12(s,3H),2.04–1.96(m,6H)。1H NMR (400MHz, DMSO-d6) δ 8.16 (s, 1H), 7.95 (s, 1H), 7.23 (s, 1H), 6.61 (s, 1H), 4.50 (t, 2H), 3.20 (s, 3H), 3.12(t,2H), 2.45–2.37(M,6H), 2.12(s,3H), 2.04–1.96(m,6H).
LC-MS m/z(ESI)=431.20[M+1]。LC-MS m/z(ESI)=431.20[M+1].
实施例24Example 24
9-(8-氧杂双环[3.2.1]辛烷-3-基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮9-(8-oxabicyclo[3.2.1]octan-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino )-7,9-dihydro-8H-purin-8-one
9-(8-oxabicyclo[3.2.1]octan-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(8-oxabicyclo[3.2.1]octan-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H -purin-8-one
第一步:first step:
8-氧杂双环[3.2.1]辛-3-酮肟(24b)8-oxabicyclo[3.2.1]octan-3-one oxime (24b)
8-oxabicyclo[3.2.1]octan-3-one oxime8-oxabicyclo[3.2.1]octan-3-one oxime
将8-氧杂双环[3.2.1]辛烷-3-酮24a(1.4g,11.1mmol)、盐酸羟胺(925mg,13.3mmol)和碳酸钾(3.1g,22.2mmol)溶解于乙醇/水(10mL/5mL)混合溶剂中,在80℃下反应2h。反应结束后,直接将反应液浓缩,加水淬灭,乙酸乙酯萃取,无水硫酸钠干燥、过滤,减压移除有机溶剂得到标题化合物8-氧杂双环[3.2.1]辛-3-酮肟(24b)(淡黄色固体,1.56g,产率100%)。Dissolve 8-oxabicyclo[3.2.1]octan-3-one 24a (1.4g, 11.1mmol), hydroxylamine hydrochloride (925mg, 13.3mmol) and potassium carbonate (3.1g, 22.2mmol) in ethanol/water ( 10mL/5mL) in a mixed solvent, react at 80°C for 2h. After the reaction, the reaction solution was directly concentrated, quenched with water, extracted with ethyl acetate, dried with anhydrous sodium sulfate, filtered, and the organic solvent was removed under reduced pressure to obtain the title compound 8-oxabicyclo[3.2.1]oct-3- Ketone oxime (24b) (light yellow solid, 1.56 g, yield 100%).
LC-MS m/z(ESI)=142.10[M+1]。LC-MS m/z(ESI)=142.10[M+1].
第二步:The second step:
8-氧杂双环[3.2.1]辛-3-胺(24c)8-oxabicyclo[3.2.1]oct-3-amine(24c)
8-oxabicyclo[3.2.1]octan-3-amine8-oxabicyclo[3.2.1]octan-3-amine
将8-氧杂双环[3.2.1]辛-3-酮肟24b(1.5g,10.64mmol)溶解于甲醇(30mL)中,室温下加入六水氯化镍(2.53g,10.64mmol),在室温下反应0.5h。将反应液冷却至-30℃,缓慢添加硼氢化钠(6.0g,159.6mmol),添加完毕缓慢升至室温反应过夜;反应结束,加水淬灭,乙酸乙酯萃取,无水硫酸钠干燥、过滤,减压移除有机溶剂得到标题化合物8-氧杂双环[3.2.1]辛-3-胺(24c)(淡黄色油状物,639mg,产率45%)。Dissolve 8-oxabicyclo[3.2.1]octan-3-one oxime 24b (1.5g, 10.64mmol) in methanol (30mL), add nickel chloride hexahydrate (2.53g, 10.64mmol) at room temperature, React at room temperature for 0.5h. The reaction solution was cooled to -30°C, and sodium borohydride (6.0g, 159.6mmol) was slowly added. After the addition, it was slowly raised to room temperature and reacted overnight; the reaction was completed, quenched with water, extracted with ethyl acetate, dried over anhydrous sodium sulfate, filtered The organic solvent was removed under reduced pressure to obtain the title compound 8-oxabicyclo[3.2.1]oct-3-amine (24c) (light yellow oil, 639 mg, yield 45%).
LC-MS m/z(ESI)=128.10[M+1]。LC-MS m/z(ESI)=128.10[M+1].
第三步:third step:
4-((8-氧杂双环[3.2.1]辛烷-3-基)氨基)-2-氯嘧啶-5-羧酸乙酯(24d)4-((8-oxabicyclo[3.2.1]octan-3-yl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester (24d)
ethyl 4-((8-oxabicyclo[3.2.1]octan-3-yl)amino)-2-chloropyrimidine-5-carboxylateethyl 4-((8-oxabicyclo[3.2.1]octan-3-yl)amino)-2-chloropyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(1.1g,5.03mmol)、碳酸钾(1.74g,12.58mmol)溶解于乙腈(20mL),在0℃下加入8-氧杂双环[3.2.1]辛-3-胺24c(639mg,5.03mmol),在室温下搅拌20h。加水30mL,有固体析出,过滤并用水洗3次,浓缩得到标题化合物4-((8-氧杂双环[3.2.1]辛烷-3-基)氨基)-2-氯嘧啶-5-羧酸乙酯(24d)(白色固体,1.01g,产率64.3%)。Dissolve 2,4-dichloropyrimidine-5-carboxylic acid ethyl ester 1a (1.1g, 5.03mmol), potassium carbonate (1.74g, 12.58mmol) in acetonitrile (20mL), add 8-oxabicyclo at 0℃ [3.2.1] Octan-3-amine 24c (639 mg, 5.03 mmol), stirred at room temperature for 20 h. Add 30 mL of water, a solid precipitated out, filtered and washed with water 3 times, and concentrated to obtain the title compound 4-((8-oxabicyclo[3.2.1]octane-3-yl)amino)-2-chloropyrimidine-5-carboxylic acid Ethyl ester (24d) (white solid, 1.01 g, yield 64.3%).
1H NMR(400MHz,DMSO)δ8.91(d,1H),8.64(s,1H),4.38–4.31(m,4H),4.30–4.24(m,1H),2.20–2.08(m,2H),2.02–1.90(m,4H),1.66(d,2H),1.32(t,3H)。1H NMR(400MHz,DMSO)δ8.91(d,1H),8.64(s,1H), 4.38-4.31(m,4H), 4.30-4.24(m,1H), 2.20-2.08(m,2H), 2.02–1.90 (m, 4H), 1.66 (d, 2H), 1.32 (t, 3H).
LC-MS m/z(ESI)=312.10[M+1]。LC-MS m/z(ESI)=312.10[M+1].
第四步:the fourth step:
4-((8-氧杂双环[3.2.1]辛-3-基)氨基)-2-氯嘧啶-5-羧酸(24e)4-((8-oxabicyclo[3.2.1]oct-3-yl)amino)-2-chloropyrimidine-5-carboxylic acid (24e)
4-((8-oxabicyclo[3.2.1]octan-3-yl)amino)-2-chloropyrimidine-5-carboxylic acid4-((8-oxabicyclo[3.2.1]octan-3-yl)amino)-2-chloropyrimidine-5-carboxylic acid
将4-((8-氧杂双环[3.2.1]辛烷-3-基)氨基)-2-氯嘧啶-5-羧酸乙酯24d(1.0g,3.21mmol)溶解于四氢呋喃10mL,水5mL中,加入氢氧化锂(308mg,12.83mmol),室温下搅拌1h。将四氢呋喃旋干,调PH为4-5,有白色固体析出,过滤,滤饼用石油醚/乙酸乙酯(v/v=10/1)洗两次,浓缩得到标题化合物4-((8-氧杂双环[3.2.1]辛-3-基)氨基)-2-氯嘧啶-5-羧酸(24e)(白色固体,808mg,产率87.9%)。Dissolve 4-((8-oxabicyclo[3.2.1]octane-3-yl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester 24d (1.0g, 3.21mmol) in 10mL of tetrahydrofuran, water In 5 mL, lithium hydroxide (308 mg, 12.83 mmol) was added, and the mixture was stirred at room temperature for 1 h. The tetrahydrofuran was spin-dried and the pH was adjusted to 4-5. A white solid precipitated out and filtered. The filter cake was washed twice with petroleum ether/ethyl acetate (v/v=10/1) and concentrated to obtain the title compound 4-((8 -Oxabicyclo[3.2.1]oct-3-yl)amino)-2-chloropyrimidine-5-carboxylic acid (24e) (white solid, 808 mg, yield 87.9%).
1H NMR(400MHz,DMSO)δ13.83(s,1H),9.17(d,1H),8.59(s,1H),4.32(s,2H),4.29–4.22(m,1H),2.17–2.07(m,2H),2.00–1.89(m,4H),1.65(d,2H)。1H NMR (400MHz, DMSO) δ 13.83 (s, 1H), 9.17 (d, 1H), 8.59 (s, 1H), 4.32 (s, 2H), 4.29-4.22 (m, 1H), 2.17-2.07 ( m, 2H), 2.00–1.89 (m, 4H), 1.65 (d, 2H).
LC-MS m/z(ESI)=284.20[M+1]。LC-MS m/z(ESI)=284.20[M+1].
第五步:the fifth step:
9-(8-氧杂双环[3.2.1]辛-3-基)-2-氯-7,9-二氢-8H-嘌呤-8-酮(24f)9-(8-oxabicyclo[3.2.1]oct-3-yl)-2-chloro-7,9-dihydro-8H-purin-8-one (24f)
9-(8-oxabicyclo[3.2.1]octan-3-yl)-2-chloro-7,9-dihydro-8H-purin-8-one9-(8-oxabicyclo[3.2.1]octan-3-yl)-2-chloro-7,9-dihydro-8H-purin-8-one
将4-((8-氧杂双环[3.2.1]辛-3-基)氨基)-2-氯嘧啶-5-羧酸24e(808mg,2.85mmol)溶解于二甲基乙酰胺(20mL)中,加入三乙胺(288mg,2.85mmol)、叠氮磷酸二苯酯(784mg,2.85mmol),随后逐步升温至120℃,搅拌1.5h。浓缩反应液,残留物用硅胶柱色谱分离提纯,(石油醚/乙酸乙酯(v/v)=5:1~1:10),得到标题化合物9-(8-氧杂双环[3.2.1]辛-3-基)-2-氯-7,9-二氢-8H-嘌呤-8-酮(24f)(白色固体,653mg,产率71%)。Dissolve 4-((8-oxabicyclo[3.2.1]oct-3-yl)amino)-2-chloropyrimidine-5-carboxylic acid 24e (808mg, 2.85mmol) in dimethylacetamide (20mL) Add triethylamine (288mg, 2.85mmol) and diphenyl azide phosphate (784mg, 2.85mmol), then gradually increase the temperature to 120°C and stir for 1.5h. The reaction solution was concentrated, and the residue was separated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)=5:1~1:10) to obtain the title compound 9-(8-oxabicyclo[3.2.1 ]Oct-3-yl)-2-chloro-7,9-dihydro-8H-purin-8-one (24f) (white solid, 653 mg, yield 71%).
1H NMR(600MHz,DMSO)δ11.64(s,1H),8.12(s,1H),4.47–4.44(m,2H),4.42–4.40(m,1H),2.28–2.20(m,2H),2.07–2.01(m,2H),1.96–1.90(m,2H),1.81–1.75(m,2H)。1H NMR (600MHz, DMSO) δ 11.64 (s, 1H), 8.12 (s, 1H), 4.47-4.44 (m, 2H), 4.42-4.40 (m, 1H), 2.28-2.20 (m, 2H), 2.07–2.01(m,2H), 1.96–1.90(m,2H), 1.81–1.75(m,2H).
LC-MS m/z(ESI)=281.10[M+1]。LC-MS m/z(ESI)=281.10[M+1].
第六步:The sixth step:
9-(8-氧杂双环[3.2.1]辛-3-基)-2-氯-7-甲基-7,9-二氢-8H-嘌呤-8-酮(24g)9-(8-oxabicyclo[3.2.1]oct-3-yl)-2-chloro-7-methyl-7,9-dihydro-8H-purin-8-one (24g)
9-(8-oxabicyclo[3.2.1]octan-3-yl)-2-chloro-7-methyl-7,9-dihydro-8H-purin-8-one9-(8-oxabicyclo[3.2.1]octan-3-yl)-2-chloro-7-methyl-7,9-dihydro-8H-purin-8-one
将9-(8-氧杂双环[3.2.1]辛-3-基)-2-氯-7,9-二氢-8H-嘌呤-8-酮24f(653mg,2.33mmol)溶解于二甲基甲酰胺(10mL)中,在0℃下加入硫酸二甲酯(293mg,2.33mmol)和碳酸铯(1.52g,4.65mmol),在0℃下搅拌1h。随后加入10mL水,用乙酸乙酯萃取3次,有机相用无水硫酸钠干燥,浓缩,有固体析出,过滤得到标题化合物9-(8-氧杂双环[3.2.1]辛-3-基)-2-氯-7-甲基-7,9-二氢-8H-嘌呤-8-酮(24g)(白色固体,533mg,产率81.6%)。Dissolve 9-(8-oxabicyclo[3.2.1]oct-3-yl)-2-chloro-7,9-dihydro-8H-purin-8-one 24f (653mg, 2.33mmol) in dimethyl Add dimethyl sulfate (293 mg, 2.33 mmol) and cesium carbonate (1.52 g, 4.65 mmol) to methyl formamide (10 mL) at 0°C, and stir at 0°C for 1 h. Then 10mL of water was added, extracted with ethyl acetate 3 times, the organic phase was dried over anhydrous sodium sulfate, concentrated, a solid precipitated out, filtered to obtain the title compound 9-(8-oxabicyclo[3.2.1]oct-3-yl )-2-chloro-7-methyl-7,9-dihydro-8H-purin-8-one (24g) (white solid, 533mg, yield 81.6%).
1H NMR(400MHz,DMSO)δ8.36–8.34(m,1H),4.51-4.47(m,2H),4.45–4.42(m,1H),3.34(s,3H),2.31–2.22(m,2H),2.07–1.98(m,2H),1.98–1.90(m,2H),1.79(t,2H)。1H NMR (400MHz, DMSO) δ 8.36 - 8.34 (m, 1H), 4.51-4.47 (m, 2H), 4.45 - 4.42 (m, 1H), 3.34 (s, 3H), 2.31 - 2.22 (m, 2H) ), 2.07–1.98(m,2H), 1.98–1.90(m,2H), 1.79(t,2H).
LC-MS m/z(ESI)=295.20[M+1]。LC-MS m/z(ESI)=295.20[M+1].
第七步:The seventh step:
9-(8-氧杂双环[3.2.1]辛烷-3-基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物24)9-(8-oxabicyclo[3.2.1]octane-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino )-7,9-dihydro-8H-purin-8-one (Compound 24)
9-(8-oxabicyclo[3.2.1]octan-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(8-oxabicyclo[3.2.1]octan-3-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H -purin-8-one
将9-(8-氧杂双环[3.2.1]辛-3-基)-2-氯-7-甲基-7,9-二氢-8H-嘌呤-8-酮24g(100mg,0.34mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(101mg,0.68mmol)、碳酸铯(221mg,0.68mmol)和Brettphos G3 Pd(31mg,0.034mmol)加入干燥的反应管,N 2置换三次,然后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。冷却至室温,使用硅胶柱色 谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化,得到标题化合物9-(8-氧杂双环[3.2.1]辛烷-3-基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物24)(灰白色固体,60mg,产率43.40%)。 The 9-(8-oxabicyclo[3.2.1]oct-3-yl)-2-chloro-7-methyl-7,9-dihydro-8H-purin-8-one 24g (100mg, 0.34mmol ), 6-methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (101mg, 0.68mmol), cesium carbonate (221mg, 0.68mmol) and Brettphos G3 Pd (31mg, 0.034mmol) were added to the dry The reaction tube was replaced with N 2 three times, then dry 1,4-dioxane (1 mL) was added, and the reaction was carried out at 110°C for 5 hours. Cooled to room temperature, and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 9-(8-oxabicyclo[3.2.1]octan-3-yl )-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one (compound 24 ) (Off-white solid, 60 mg, yield 43.40%).
1H NMR(400MHz,DMSO-d6)δ8.27(s,1H),7.98(s,1H),7.09(s,1H),6.64(s,1H),4.49(t,2H),4.38–4.29(m,3H),3.25(s,3H),3.11(t,2H),2.15–2.05(m,5H),1.95(t,2H),1.81–1.74(m,2H),1.50–1.40(d,2H)。1H NMR(400MHz,DMSO-d6)δ8.27(s,1H),7.98(s,1H), 7.09(s,1H), 6.64(s,1H), 4.49(t,2H), 4.38-4.29( m, 3H), 3.25 (s, 3H), 3.11 (t, 2H), 2.15-2.05 (m, 5H), 1.95 (t, 2H), 1.81-1.74 (m, 2H), 1.50-1.40 (d, 2H).
LC-MS m/z(ESI)=408.20[M+1]。LC-MS m/z(ESI)=408.20[M+1].
实施例25Example 25
7-甲基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(2-氧杂螺[3.5]壬基-7-基)-7,9-二氢-8H-嘌呤-8-酮7-methyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(2-oxaspiro[3.5]nonyl-7-yl) -7,9-dihydro-8H-purin-8-one
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(2-oxaspiro[3.5]nonan-7-yl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(2-oxaspiro[3.5]nonan-7-yl)-7,9-dihydro-8H-purin -8-one
第一步:first step:
2-氧杂螺[3.5]壬-7-酮肟(25b)2-oxaspiro[3.5]non-7-one oxime (25b)
2-oxaspiro[3.5]nonan-7-one oxime2-oxaspiro[3.5]nonan-7-one oxime
将2-氧杂螺[3.5]壬基-7-酮25a(1.5g,10.7mmol)、盐酸羟胺(744mg,10.7mmol)和碳酸钾(2.95g,21.4mmol)溶解于乙醇/水(10mL/5mL)混合溶剂中,在80℃下反应2h。反应结束后,直接将反应液浓缩,加水淬灭,乙酸乙酯萃取,无水硫酸钠干燥、过滤,减压移除有机溶剂得到标题化合物2-氧杂螺[3.5]壬-7-酮肟(25b)(淡黄色固体,1.45g,产率89%)。Dissolve 2-oxaspiro[3.5]nonyl-7-one 25a (1.5g, 10.7mmol), hydroxylamine hydrochloride (744mg, 10.7mmol) and potassium carbonate (2.95g, 21.4mmol) in ethanol/water (10mL/ 5mL) in a mixed solvent, react at 80°C for 2h. After the reaction, the reaction solution was directly concentrated, quenched with water, extracted with ethyl acetate, dried over anhydrous sodium sulfate, filtered, and the organic solvent was removed under reduced pressure to obtain the title compound 2-oxaspiro[3.5]non-7-one oxime (25b) (Light yellow solid, 1.45 g, yield 89%).
LC-MS m/z(ESI)=156.10[M+1]。LC-MS m/z(ESI)=156.10[M+1].
第二步:The second step:
2-氧杂螺[3.5]壬-7-胺(25c)2-oxaspiro[3.5]non-7-amine (25c)
2-oxaspiro[3.5]nonan-7-amine2-oxaspiro[3.5]nonan-7-amine
将2-氧杂螺[3.5]壬-7-酮肟25b(1.45g,9.34mmol)溶解于甲醇(30mL)中,室温下加入六水氯化镍(2.22g,9.34mmol),在室温下反应0.5h。将反应液冷却至-30℃,缓慢 添加硼氢化钠(5.3g,140.1mmol),添加完毕缓慢升至室温反应过夜;反应结束,加水淬灭,乙酸乙酯萃取,无水硫酸钠干燥、过滤,减压移除有机溶剂得到标题化合物2-氧杂螺[3.5]壬-7-胺(25c)(淡黄色油状物,481mg,产率37.6%)。Dissolve 2-oxaspiro[3.5]non-7-one oxime 25b (1.45g, 9.34mmol) in methanol (30mL) and add nickel chloride hexahydrate (2.22g, 9.34mmol) at room temperature. Reaction for 0.5h. The reaction solution was cooled to -30°C, and sodium borohydride (5.3g, 140.1mmol) was slowly added. After the addition, the temperature was slowly raised to room temperature and reacted overnight; the reaction was completed, quenched with water, extracted with ethyl acetate, dried over anhydrous sodium sulfate, filtered The organic solvent was removed under reduced pressure to obtain the title compound 2-oxaspiro[3.5]non-7-amine (25c) (light yellow oil, 481 mg, yield 37.6%).
LC-MS m/z(ESI)=142.20[M+1]。LC-MS m/z(ESI)=142.20[M+1].
第三步:third step:
乙基4-((2-氧杂螺[3.5]壬-7-基)氨基)-2-氯嘧啶-5-甲酸乙酯(25d)Ethyl 4-((2-oxaspiro[3.5]non-7-yl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester (25d)
ethyl 4-((2-oxaspiro[3.5]nonan-7-yl)amino)-2-chloropyrimidine-5-carboxylateethyl 4-((2-oxaspiro[3.5]nonan-7-yl)amino)-2-chloropyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(753mg,3.41mmol)、碳酸钾(940mg,6.81mmol)溶解于乙腈(20mL),在0℃下加入8-氧杂双环[3.2.1]辛-3-胺25c(481mg,3.41mmol),在室温下搅拌20h。加水30mL,有固体析出,过滤并用水洗3次,浓缩得到标题化合物乙基4-((2-氧杂螺[3.5]壬-7-基)氨基)-2-氯嘧啶-5-甲酸乙酯(25d)(白色固体,640mg,产率57.6%)。Dissolve 2,4-dichloropyrimidine-5-carboxylic acid ethyl ester 1a (753mg, 3.41mmol), potassium carbonate (940mg, 6.81mmol) in acetonitrile (20mL), add 8-oxabicyclo[3.2 .1] Octan-3-amine 25c (481mg, 3.41mmol), stirred at room temperature for 20h. Add 30 mL of water, a solid precipitated out, filtered and washed with water 3 times, and concentrated to obtain the title compound ethyl 4-((2-oxaspiro[3.5]non-7-yl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester (25d) (white solid, 640 mg, yield 57.6%).
1H NMR(400MHz,DMSO)δ8.61(s,1H),8.29(d,1H),4.34–4.29(m,4H),4.24(s,2H),3.98–3.89(m,1H),2.05–1.92(m,2H),1.86–1.75(m,2H),1.65–1.56(m,2H),1.45–1.35(m,2H),1.34–1.27(m,3H)。1H NMR (400MHz, DMSO) δ8.61 (s, 1H), 8.29 (d, 1H), 4.34 - 4.29 (m, 4H), 4.24 (s, 2H), 3.98 - 3.89 (m, 1H), 2.05 - 1.92(m,2H), 1.86–1.75(m,2H), 1.65–1.56(m,2H), 1.45–1.35(m,2H), 1.34–1.27(m,3H).
LC-MS m/z(ESI)=326.20[M+1]。LC-MS m/z(ESI)=326.20[M+1].
第四步:the fourth step:
4-((2-氧杂螺[3.5]壬基-7-基)氨基)-2-氯嘧啶-5-羧酸(25e)4-((2-oxaspiro[3.5]nonyl-7-yl)amino)-2-chloropyrimidine-5-carboxylic acid (25e)
4-((2-oxaspiro[3.5]nonan-7-yl)amino)-2-chloropyrimidine-5-carboxylic acid4-((2-oxaspiro[3.5]nonan-7-yl)amino)-2-chloropyrimidine-5-carboxylic acid
将乙基4-((2-氧杂螺[3.5]壬-7-基)氨基)-2-氯嘧啶-5-甲酸乙酯25d(640mg,1.96mmol)溶解于四氢呋喃10mL,水5mL中,加入氢氧化锂(189mg,7.86mmol),室温下搅拌1小时。将四氢呋喃旋干,调PH为4-5,有白色固体析出,过滤,滤饼用石油醚/乙酸乙酯(v/v=10/1)洗两次,浓缩得到标题化合物4-((2-氧杂螺[3.5]壬基-7-基)氨基)-2-氯嘧啶-5-羧酸(25e)(白色固体,518mg,产率88.5%)。Ethyl 4-((2-oxaspiro[3.5]non-7-yl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester 25d (640mg, 1.96mmol) was dissolved in tetrahydrofuran 10mL, water 5mL, Lithium hydroxide (189 mg, 7.86 mmol) was added, and the mixture was stirred at room temperature for 1 hour. The tetrahydrofuran was spin-dried and adjusted to pH 4-5. A white solid precipitated out. Filtered. The filter cake was washed twice with petroleum ether/ethyl acetate (v/v=10/1) and concentrated to obtain the title compound 4-((2 -Oxaspiro[3.5]nonyl-7-yl)amino)-2-chloropyrimidine-5-carboxylic acid (25e) (white solid, 518 mg, yield 88.5%).
1H NMR(400MHz,DMSO)δ13.74(s,1H),8.57(s,1H),8.48(d,1H),4.32(s,2H),4.24(s,2H),3.97–3.84(m,1H),1.99(d,2H),1.86–1.76(m,2H),1.66–1.54(m,2H),1.42–1.26(m,2H)。1H NMR (400MHz, DMSO) δ 13.74 (s, 1H), 8.57 (s, 1H), 8.48 (d, 1H), 4.32 (s, 2H), 4.24 (s, 2H), 3.97-3.84 (m, 1H), 1.99(d,2H), 1.86–1.76(m,2H), 1.66–1.54(m,2H), 1.42–1.26(m,2H).
LC-MS m/z(ESI)=298.10[M+1]。LC-MS m/z(ESI)=298.10[M+1].
第五步:the fifth step:
2-氯-9-(2-氧杂螺[3.5]壬-7-基)-7,9-二氢-8H-嘌呤-8-酮(25f)2-Chloro-9-(2-oxaspiro[3.5]non-7-yl)-7,9-dihydro-8H-purin-8-one (25f)
2-chloro-9-(2-oxaspiro[3.5]nonan-7-yl)-7,9-dihydro-8H-purin-8-one2-chloro-9-(2-oxaspiro[3.5]nonan-7-yl)-7,9-dihydro-8H-purin-8-one
将4-((2-氧杂螺[3.5]壬基-7-基)氨基)-2-氯嘧啶-5-羧酸25e(518mg,1.74mmol)溶解于二甲基乙酰胺(20mL)中,加入三乙胺(175mg,1.74mmol)、叠氮磷酸二苯酯(479mg,1.74mmol),随后逐步升温至120℃,搅拌1.5h。浓缩反应液,残留物用硅胶柱色谱分离提纯,(石油醚/乙酸乙酯(v/v)=5:1~1:10),得到标题化合物2-氯-9-(2-氧杂螺[3.5]壬-7-基)-7,9-二氢-8H-嘌呤-8-酮(25f)(白色固体,353mg,产率72.8%)。Dissolve 4-((2-oxaspiro[3.5]nonyl-7-yl)amino)-2-chloropyrimidine-5-carboxylic acid 25e (518mg, 1.74mmol) in dimethylacetamide (20mL) Add triethylamine (175 mg, 1.74 mmol) and diphenyl azide phosphate (479 mg, 1.74 mmol), then gradually increase the temperature to 120° C. and stir for 1.5 h. The reaction solution was concentrated, and the residue was separated and purified by silica gel column chromatography (petroleum ether/ethyl acetate (v/v)=5:1~1:10) to obtain the title compound 2-chloro-9-(2-oxaspiro [3.5] Non-7-yl)-7,9-dihydro-8H-purin-8-one (25f) (white solid, 353 mg, yield 72.8%).
1H NMR(600MHz,DMSO)δ11.61(s,1H),8.11(s,1H),4.41(s,2H),4.24(s,2H),4.15–4.08(m,1H),2.24–2.08(m,4H),1.69(d,2H),1.64–1.51(m,2H)。1H NMR (600MHz, DMSO) δ 11.61 (s, 1H), 8.11 (s, 1H), 4.41 (s, 2H), 4.24 (s, 2H), 4.15-4.08 (m, 1H), 2.24-2.08 ( m, 4H), 1.69 (d, 2H), 1.64-1.51 (m, 2H).
LC-MS m/z(ESI)=295.10[M+1]。LC-MS m/z(ESI)=295.10[M+1].
第六步:The sixth step:
2-氯-7-甲基-9-(2-氧杂螺[3.5]壬-7-基)-7,9-二氢-8H-嘌呤-8-酮(25g)2-Chloro-7-methyl-9-(2-oxaspiro[3.5]non-7-yl)-7,9-dihydro-8H-purin-8-one (25g)
2-chloro-7-methyl-9-(2-oxaspiro[3.5]nonan-7-yl)-7,9-dihydro-8H-purin-8-one2-chloro-7-methyl-9-(2-oxaspiro[3.5]nonan-7-yl)-7,9-dihydro-8H-purin-8-one
将2-氯-9-(2-氧杂螺[3.5]壬-7-基)-7,9-二氢-8H-嘌呤-8-酮25f(353mg,1.18mmol)溶解于二甲基甲酰胺(10mL)中,在0℃下加入硫酸二甲酯(150mg,1.18mmol)和碳酸铯(464mg,3.36mmol),在0℃下搅拌1h。随后加入10mL水,用乙酸乙酯萃取3次,有机相用无水硫酸钠干燥,浓缩,有固体析出,过滤得到标题化合物2-氯-7-甲基-9-(2-氧 杂螺[3.5]壬-7-基)-7,9-二氢-8H-嘌呤-8-酮(25g)(白色固体,313mg,产率85.5%)。Dissolve 2-chloro-9-(2-oxaspiro[3.5]non-7-yl)-7,9-dihydro-8H-purin-8-one 25f (353mg, 1.18mmol) in dimethylform To the amide (10 mL), dimethyl sulfate (150 mg, 1.18 mmol) and cesium carbonate (464 mg, 3.36 mmol) were added at 0°C, and the mixture was stirred at 0°C for 1 h. Then 10 mL of water was added, extracted with ethyl acetate 3 times, the organic phase was dried over anhydrous sodium sulfate, concentrated, a solid precipitated out, filtered to obtain the title compound 2-chloro-7-methyl-9-(2-oxaspiro[ 3.5] Non-7-yl)-7,9-dihydro-8H-purin-8-one (25 g) (white solid, 313 mg, yield 85.5%).
1H NMR(400MHz,DMSO)δ8.34(s,1H),4.41(s,2H),4.25(s,2H),4.22–4.08(m,1H),3.34(s,3H),2.22–2.05(m,4H),1.69(d,2H),1.64–1.50(m,2H)。1H NMR (400MHz, DMSO) δ 8.34 (s, 1H), 4.41 (s, 2H), 4.25 (s, 2H), 4.22-4.08 (m, 1H), 3.34 (s, 3H), 2.22-2.05 ( m, 4H), 1.69 (d, 2H), 1.64-1.50 (m, 2H).
LC-MS m/z(ESI)=309.20[M+1]。LC-MS m/z(ESI)=309.20[M+1].
第七步:The seventh step:
7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(2-氧杂螺[3.5]壬基-7-基)-7,9-二氢-8H-嘌呤-8酮(化合物25)7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(2-oxaspiro[3.5]nonyl-7-yl)- 7,9-dihydro-8H-purin-8one (Compound 25)
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(2-oxaspiro[3.5]nonan-7-yl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(2-oxaspiro[3.5]nonan-7-yl)-7,9-dihydro-8H-purin -8-one
将2-氯-7-甲基-9-(2-氧杂螺[3.5]壬-7-基)-7,9-二氢-8H-嘌呤-8-酮25g(100mg,0.32mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(97mg,0.64mmol)、碳酸铯(211mg,0.64mmol)和Brettphos G3 Pd(29mg,0.032mmol)加入干燥的反应管,N2置换三次,然后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。冷却至室温,使用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化,得到标题化合物7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(2-氧杂螺[3.5]壬基-7-基)-7,9-二氢-8H-嘌呤-8酮(化合物25)(灰白色固体,50mg,产率36.63%)。25g (100mg, 0.32mmol) of 2-chloro-7-methyl-9-(2-oxaspiro[3.5]non-7-yl)-7,9-dihydro-8H-purin-8-one, 6-Methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (97mg, 0.64mmol), cesium carbonate (211mg, 0.64mmol) and Brettphos G3 Pd (29mg, 0.032mmol) were added to the dry reaction Replace the tube with N2 three times, then add dry 1,4-dioxane (1 mL), and react at 110°C for 5 hours. Cool to room temperature, use silica gel column chromatography to separate and purify (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 7-methyl-2-((6-methyl-2,3-di Hydrobenzofuran-5-yl)amino)-9-(2-oxaspiro[3.5]nonyl-7-yl)-7,9-dihydro-8H-purin-8one (compound 25) (off-white Solid, 50 mg, yield 36.63%).
1H NMR(400MHz,DMSO-d6)δ8.24(s,1H),7.96(s,1H),7.07(s,1H),6.63(s,1H),4.47(t,2H),4.19(s,2H),4.10(s,2H),4.06–3.96(m,1H),3.25(s,3H),3.12(t,2H),2.13–1.99(m,7H),1.55(d,2H),1.52–1.41(m,2H)。1H NMR (400MHz, DMSO-d6) δ 8.24 (s, 1H), 7.96 (s, 1H), 7.07 (s, 1H), 6.63 (s, 1H), 4.47 (t, 2H), 4.19 (s, 2H), 4.10 (s, 2H), 4.06-3.96 (m, 1H), 3.25 (s, 3H), 3.12 (t, 2H), 2.13-1.99 (m, 7H), 1.55 (d, 2H), 1.52 –1.41(m,2H).
LC-MS m/z(ESI)=422.20[M+1]。LC-MS m/z(ESI)=422.20[M+1].
实施例26Example 26
9-(3-羟基双环[3.2.1]辛基-8-基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮9-(3-Hydroxybicyclo[3.2.1]octyl-8-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino) -7,9-dihydro-8H-purin-8-one
9-(3-hydroxybicyclo[3.2.1]octan-8-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(3-hydroxybicyclo[3.2.1]octan-8-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H -purin-8-one
第一步:first step:
8-氨基双环[3.2.1]辛基-3-酮盐酸盐(26b)8-aminobicyclo[3.2.1]octyl-3-one hydrochloride (26b)
8-aminobicyclo[3.2.1]octan-3-one hydrochloride8-aminobicyclo[3.2.1]octan-3-one hydrochloride
将3-氧代双环[3.2.1]辛-8-基)氨基甲酸叔丁酯26a(1.0g,4.18mmol)溶于4N盐酸二氧六环(40mL),室温反应1h。TLC监测反应结束,直接将反应液浓缩至干,得目标化合物8-氨基双环[3.2.1]octan-3-one盐酸盐(26b)(白色固体,30mg,产率99.42%)。Dissolve tert-butyl 3-oxobicyclo[3.2.1]oct-8-yl)carbamate 26a (1.0 g, 4.18 mmol) in 4N dioxane hydrochloride (40 mL), and react at room temperature for 1 h. TLC monitored the completion of the reaction, and the reaction solution was directly concentrated to dryness to obtain the target compound 8-aminobicyclo[3.2.1]octan-3-one hydrochloride (26b) (white solid, 30mg, yield 99.42%).
1H NMR(401MHz,DMSO-d6)δ8.77(s,2H),3.36(m,2H),2.80-2.77(m,2H),2.16-2.13(m,2H),1.87-1.85(m,2H),1.43-1.42(m,2H)。1H NMR (401MHz, DMSO-d6) δ8.77 (s, 2H), 3.36 (m, 2H), 2.80-2.77 (m, 2H), 2.16-2.13 (m, 2H), 1.87-1.85 (m, 2H) ), 1.43-1.42 (m, 2H).
LC-MS m/z(ESI)=176.20[M+1]。LC-MS m/z(ESI)=176.20[M+1].
第二步:The second step:
2-氯-4-(((3-氧双环[3.2.1]辛-8-基)氨基)嘧啶-5-羧酸乙酯(26c)Ethyl 2-chloro-4-(((3-oxobicyclo[3.2.1]oct-8-yl)amino)pyrimidine-5-carboxylate (26c)
ethyl 2-chloro-4-((3-oxobicyclo[3.2.1]octan-8-yl)amino)pyrimidine-5-carboxylateethyl 2-chloro-4-((3-oxobicyclo[3.2.1]octan-8-yl)amino)pyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(1.38g,6.26mmol),(1R,5S,8s)-8-氨基双环[3.2.1]octan-3-one盐酸盐26b(730mg,4.17mmol),碳酸钾(1.73g,12.52mmol)溶于乙腈(20mL),反应液在室温反应16h。TLC监测反应结束,过滤,并用少量乙腈清洗固体,将滤液合并后浓缩,粗品经柱层析分离(石油醚:乙酸乙酯=1:1)后得目标化合物2-氯-4-(((3-氧双环[3.2.1]octan-8-yl)氨基)嘧啶-5-羧酸乙酯(26c)(白色固体,1.0g,产率74.01%)。Ethyl 2,4-dichloropyrimidine-5-carboxylate 1a (1.38g, 6.26mmol), (1R, 5S, 8s)-8-aminobicyclo[3.2.1]octan-3-one hydrochloride 26b (730mg, 4.17mmol), potassium carbonate (1.73g, 12.52mmol) was dissolved in acetonitrile (20mL), and the reaction solution was reacted at room temperature for 16h. The end of the reaction was monitored by TLC, filtered, and the solid was washed with a small amount of acetonitrile. The filtrate was combined and concentrated. The crude product was separated by column chromatography (petroleum ether: ethyl acetate = 1:1) to obtain the target compound 2-chloro-4-((( 3-Oxybicyclo[3.2.1]octan-8-yl)amino)pyrimidine-5-carboxylic acid ethyl ester (26c) (white solid, 1.0 g, yield 74.01%).
1H NMR(400MHz,DMSO-d6)δ8.86(d,2H),8.68(s,1H),4.36-4.30(q,2H),4.20-4.16(m,1H),2.66-2.65(m,2H),2.56-2.55(m,2H),2.21-2.17(m,2H),1.97-1.93(m,2H),1.53-1.33(m,2H),1.31(t,3H)。1H NMR(400MHz,DMSO-d6)δ8.86(d,2H),8.68(s,1H),4.36-4.30(q,2H),4.20-4.16(m,1H),2.66-2.65(m,2H) ), 2.56-2.55 (m, 2H), 2.21-2.17 (m, 2H), 1.97-1.93 (m, 2H), 1.53-1.33 (m, 2H), 1.31 (t, 3H).
LC-MS m/z(ESI)=324.10[M+1]。LC-MS m/z(ESI)=324.10[M+1].
第三步:third step:
2-氯-4-((3--3-氧双环[3.2.1]辛-8-基)氨基]嘧啶-5-羧酸(26d)2-chloro-4-((3--3-oxobicyclo[3.2.1]oct-8-yl)amino]pyrimidine-5-carboxylic acid (26d)
2-chloro-4-((3-oxobicyclo[3.2.1]octan-8-yl)amino)pyrimidine-5-carboxylic acid2-chloro-4-((3-oxobicyclo[3.2.1]octan-8-yl)amino)pyrimidine-5-carboxylic acid
将2-氯-4-((3-氧双环[3.2.1]辛-8-基)氨基)嘧啶-5-羧酸乙酯26c(1.0g,3.09mmol)溶解于四氢呋喃20mL,水20mlL中,加入氢氧化锂(259mg,6.18mmol),室温搅拌1h。TLC监测反应完全,浓缩除去四氢呋喃,用2N盐酸调pH为5,有白色固体析出,过滤,滤饼用石油醚洗两次,搜集固体得到标题化合物2-氯-4-((3-3-氧双环[3.2.1]octan-8-yl)氨 基]嘧啶-5-羧酸(26d)(白色固体,800mg,产率87.59%)。Dissolve 2-chloro-4-((3-oxobicyclo[3.2.1]oct-8-yl)amino)pyrimidine-5-carboxylic acid ethyl ester 26c (1.0g, 3.09mmol) in 20mL of tetrahydrofuran and 20mL of water , Lithium hydroxide (259mg, 6.18mmol) was added and stirred at room temperature for 1h. TLC monitored the reaction to complete, concentrated to remove tetrahydrofuran, adjusted the pH to 5 with 2N hydrochloric acid, a white solid precipitated out, filtered, the filter cake was washed twice with petroleum ether, the solid was collected to obtain the title compound 2-chloro-4-((3-3- Oxybicyclo[3.2.1]octan-8-yl)amino]pyrimidine-5-carboxylic acid (26d) (white solid, 800mg, yield 87.59%).
1H NMR(400MHz,DMSO-d6)δ13.93(s,1H),9.20(d,2H),8.64(s,1H),4.20-4.16(m,1H),2.66-2.64(m,2H),2.54-2.53(m,2H),2.21-2.16(m,2H),1.96-1.93(m,2H),1.53-1.47(m,2H)。1H NMR(400MHz,DMSO-d6)δ13.93(s,1H),9.20(d,2H),8.64(s,1H),4.20-4.16(m,1H),2.66-2.64(m,2H), 2.54-2.53 (m, 2H), 2.21-2.16 (m, 2H), 1.96-1.93 (m, 2H), 1.53-1.47 (m, 2H).
LC-MS m/z(ESI)=296.10[M+1]。LC-MS m/z(ESI)=296.10[M+1].
第四步:the fourth step:
2-氯-9-(3-氧代双环[3.2.1]辛-8-基)-7,9-二氢-8H-嘌呤-8-酮(26e)2-chloro-9-(3-oxobicyclo[3.2.1]oct-8-yl)-7,9-dihydro-8H-purin-8-one (26e)
2-chloro-9-(3-oxobicyclo[3.2.1]octan-8-yl)-7,9-dihydro-8H-purin-8-one2-chloro-9-(3-oxobicyclo[3.2.1]octan-8-yl)-7,9-dihydro-8H-purin-8-one
将2-氯-4-((3-3-氧双环[3.2.1]辛-8-基)氨基)嘧啶-5-羧酸26d(800mg,2.71mmol)溶解于二甲基乙酰胺(20mL),加入三乙胺(0.37mL,2.7mmol)、叠氮磷酸二苯酯(0.58mL,2.7mmol),随后逐步升温至90℃,搅拌2h。TLC监测反应完毕,将反应液倒入冰水中,过滤搜集固体,水洗3次,真空浓缩干燥得标题化合物2-氯-9-(3-氧代双环[3.2.1]辛-8-基)-7,9-二氢-8H-嘌呤-8-酮(26e)(白色固体,630mg,产率94.71%)。Dissolve 2-chloro-4-((3-3-oxobicyclo[3.2.1]oct-8-yl)amino)pyrimidine-5-carboxylic acid 26d (800mg, 2.71mmol) in dimethylacetamide (20mL ), add triethylamine (0.37mL, 2.7mmol) and diphenyl azide phosphate (0.58mL, 2.7mmol), then gradually increase the temperature to 90°C and stir for 2h. TLC monitors the completion of the reaction, the reaction solution is poured into ice water, the solid is collected by filtration, washed with water 3 times, and concentrated and dried in vacuo to obtain the title compound 2-chloro-9-(3-oxobicyclo[3.2.1]oct-8-yl) -7,9-dihydro-8H-purin-8-one (26e) (white solid, 630 mg, yield 94.71%).
1H NMR(400MHz,DMSO-d6)δ11.66(s,1H),8.13(s,1H),4.01-3.99(m,1H),3.65-3.64(m,2H),2.66-2.61(m,2H),2.12-2.08(m,2H),1.89-1.86(m,2H),1.54-1.49(m,2H)。1H NMR(400MHz,DMSO-d6)δ11.66(s,1H),8.13(s,1H),4.01-3.99(m,1H),3.65-3.64(m,2H),2.66-2.61(m,2H) ), 2.12-2.08 (m, 2H), 1.89-1.86 (m, 2H), 1.54-1.49 (m, 2H).
LC-MS m/z(ESI)=293.00[M+l]。LC-MS m/z(ESI)=293.00[M+1].
第五步:the fifth step:
2-氯-7-甲基-9-(3-氧代双环[3.2.1]辛-8-基)-7,9-二氢-8H-嘌呤-8-酮(26f)2-chloro-7-methyl-9-(3-oxobicyclo[3.2.1]oct-8-yl)-7,9-dihydro-8H-purin-8-one(26f)
2-chloro-7-methyl-9-(3-oxobicyclo[3.2.1]octan-8-yl)-7,9-dihydro-8H-purin-8-one2-chloro-7-methyl-9-(3-oxobicyclo[3.2.1]octan-8-yl)-7,9-dihydro-8H-purin-8-one
将2-氯-9-(3-氧代双环[3.2.1]辛-8-基)-7,9-二氢-8H-嘌呤-8-酮26e(630mg,2.15mmol)溶解于二甲基甲酰胺(10mL),在0℃下加入硫酸二甲酯(0.2mL,2.15mmol)和碳酸铯(1.4g,4.3mmol),0℃搅拌1h。TLC监测反应结束,将反应液倒入冰水中,有固体析出,过滤、搜集固体得到标题化合物2-氯-7-甲基-9-(3-氧代双环[3.2.1]辛-8-基)-7,9-二氢-8H-嘌呤-8-酮(26f)(白色固体,490mg,产率74.22%)。Dissolve 2-chloro-9-(3-oxobicyclo[3.2.1]oct-8-yl)-7,9-dihydro-8H-purin-8-one 26e (630mg, 2.15mmol) in dimethyl Methyl formamide (10 mL), dimethyl sulfate (0.2 mL, 2.15 mmol) and cesium carbonate (1.4 g, 4.3 mmol) were added at 0°C, and the mixture was stirred at 0°C for 1 h. TLC monitors the end of the reaction. Pour the reaction liquid into ice water. A solid precipitates out. The solid is filtered and collected to obtain the title compound 2-chloro-7-methyl-9-(3-oxobicyclo[3.2.1]oct-8- Yl)-7,9-dihydro-8H-purin-8-one (26f) (white solid, 490 mg, yield 74.22%).
1H NMR(400MHz,DMSO-d6)δ8.37(s,1H),4.03-4.01(m,1H),3.66-3.65(m,2H),3.36(s,3H),2.65-2.59(m,2H),2.13-2.08(m,2H),1.89-1.87(m,2H),1.55-1.50(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.37(s,1H),4.03-4.01(m,1H),3.66-3.65(m,2H),3.36(s,3H),2.65-2.59(m,2H) ), 2.13-2.08 (m, 2H), 1.89-1.87 (m, 2H), 1.55-1.50 (m, 2H).
LC-MS m/z(ESI)=307.10[M+l]。LC-MS m/z(ESI)=307.10[M+1].
第六步:The sixth step:
2-氯-9-(3-羟基双环[3.2.1]辛-8-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(26g)2-chloro-9-(3-hydroxybicyclo[3.2.1]oct-8-yl)-7-methyl-7,9-dihydro-8H-purin-8-one (26g)
2-chloro-9-(3-hydroxybicyclo[3.2.1]octan-8-yl)-7-methyl-7,9-dihydro-8H-purin-8-one2-chloro-9-(3-hydroxybicyclo[3.2.1]octan-8-yl)-7-methyl-7,9-dihydro-8H-purin-8-one
将2-氯-7-甲基-9-(3-氧代双环[3.2.1]辛-8-基)-7,9-二氢-8H-嘌呤-8-酮26f(490mg,1.6mmol)溶解于四氢呋喃(30mL)和甲醇(30mL),在0℃下加入硼氢化钠(181.30mg,4.79mmol)。TLC监测反应结束,将反应液倒入冰水中,有固体析出,过滤、搜集固体得到标题化合物2-氯-9-(3-羟基双环[3.2.1]辛-8-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(26g)(白色固体,380mg,产率77.04%)。The 2-chloro-7-methyl-9-(3-oxobicyclo[3.2.1]oct-8-yl)-7,9-dihydro-8H-purin-8-one 26f (490mg, 1.6mmol ) Was dissolved in tetrahydrofuran (30 mL) and methanol (30 mL), and sodium borohydride (181.30 mg, 4.79 mmol) was added at 0°C. TLC monitors the end of the reaction. Pour the reaction solution into ice water. A solid precipitates out. The solid is filtered and collected to obtain the title compound 2-chloro-9-(3-hydroxybicyclo[3.2.1]oct-8-yl)-7-methyl Glyc-7,9-dihydro-8H-purin-8-one (26 g) (white solid, 380 mg, yield 77.04%).
1H NMR(400MHz,DMSO-d6)δ8.37(s,1H),4.28-4.20(m,1H),3.74-3.66(m,2H),3.45-3.43(m,2H),3.37(s,3H),2.14-2.07(m,1H),1.74-1.50(m,5H),1.28-1.22(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.37(s,1H),4.28-4.20(m,1H),3.74-3.66(m,2H),3.45-3.43(m,2H),3.37(s,3H) ), 2.14-2.07 (m, 1H), 1.74-1.50 (m, 5H), 1.28-1.22 (m, 2H).
LC-MS m/z(ESI)=309.10[M+l]。LC-MS m/z(ESI)=309.10[M+1].
第七步:The seventh step:
9-(3-羟基双环[3.2.1]辛基-8-基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物26)9-(3-Hydroxybicyclo[3.2.1]octyl-8-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino) -7,9-dihydro-8H-purin-8-one (compound 26)
9-(3-hydroxybicyclo[3.2.1]octan-8-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(3-hydroxybicyclo[3.2.1]octan-8-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H -purin-8-one
将2-氯-9-(3-羟基双环[3.2.1]辛-8-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮26g(50mg,0.16mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(49mg,0.32mmol)、碳酸铯(106mg,0.32mmol)和Brettphos G3 Pd(15mg,0.016mmol)加入干燥的反应管,N 2置换三次,然后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。冷却至室温,使用硅胶柱色谱 分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化,得到标题化合物9-(3-羟基双环[3.2.1]辛基-8-基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物26)(灰白色固体,25mg,产率36.63%)。 The 2-chloro-9-(3-hydroxybicyclo[3.2.1]oct-8-yl)-7-methyl-7,9-dihydro-8H-purin-8-one 26g (50mg, 0.16mmol) , 6-Methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (49mg, 0.32mmol), cesium carbonate (106mg, 0.32mmol) and Brettphos G3 Pd (15mg, 0.016mmol) were added to the dry In the reaction tube, replace with N 2 three times, then add dry 1,4-dioxane (1 mL), and react at 110° C. for 5 h. Cooled to room temperature, and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 9-(3-hydroxybicyclo[3.2.1]octyl-8-yl) -7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one (compound 26) (Off-white solid, 25mg, yield 36.63%).
1H NMR(400MHz,DMSO-d6)δ8.18(s,1H),8.00(s,1H),7.20(s,1H),6.60(s,1H),4.49(t,2H),4.24(d,1H),3.74–3.63(m,1H),3.53–3.49(m,1H),3.45–3.39(m,2H),3.28(s,3H),3.09(t,2H),2.12(s,3H),1.66-1.53(m,4H),1.52–1.46(m,2H),1.33–1.25(m,2H)。1H NMR (400MHz, DMSO-d6) δ 8.18 (s, 1H), 8.00 (s, 1H), 7.20 (s, 1H), 6.60 (s, 1H), 4.49 (t, 2H), 4.24 (d, 1H), 3.74--3.63(m,1H), 3.53--3.49(m,1H), 3.45--3.39(m,2H), 3.28(s,3H), 3.09(t,2H), 2.12(s,3H) ,1.66-1.53(m,4H), 1.52-1.46(m,2H),1.33-1.25(m,2H).
LC-MS m/z(ESI)=422.20[M+1]。LC-MS m/z(ESI)=422.20[M+1].
实施例27Example 27
9-(3-羟基-3-甲基环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物27)9-(3-hydroxy-3-methylcyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9- Dihydro-8H-purin-8-one (Compound 27)
9-(3-hydroxy-3-methylcyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(3-hydroxy-3-methylcyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one
第一步:first step:
叔丁基(3-羟基-3-甲基环己基)氨基甲酸酯(27b)Tert-Butyl (3-hydroxy-3-methylcyclohexyl) carbamate (27b)
tert-butyl(3-hydroxy-3-methylcyclohexyl)carbamatetert-butyl(3-hydroxy-3-methylcyclohexyl)carbamate
将叔丁基(3-氧代环己基)氨基甲酸酯27a(5.00g,23.4mmol)溶解于四氢呋喃(80mL)在-78℃下预冷,然后经45min缓慢滴加1.6M甲基锂-乙醚溶液(61.5mL,98.28mmol),反应液在-78℃下搅拌反应1h后加入剩余1.6M甲基锂-乙醚溶液(61.5mL,98.28mmol),再在-78℃搅拌反应1h。TLC监测至反应结束,加入饱和氯化铵溶液淬灭反应,再加入水和乙酸乙酯萃取,浓缩有机层用硅胶柱色谱分离提纯(石油醚/乙酸乙酯(v/v)=8:1)纯化得标题化合物叔丁基(3-羟基-3-甲基环己基)氨基甲酸酯(27b)(白色固体,2.07g,产率38.53%)。Dissolve tert-butyl (3-oxocyclohexyl) carbamate 27a (5.00g, 23.4mmol) in tetrahydrofuran (80mL) and pre-cool at -78℃, then slowly add 1.6M methyl lithium dropwise over 45min. Ether solution (61.5mL, 98.28mmol), the reaction solution was stirred at -78°C for 1h, then the remaining 1.6M methyl lithium-ether solution (61.5mL, 98.28mmol) was added, and the reaction was stirred at -78°C for 1h. TLC monitors to the end of the reaction, adds saturated ammonium chloride solution to quench the reaction, adds water and ethyl acetate for extraction, concentrates the organic layer and uses silica gel column chromatography to separate and purify (petroleum ether/ethyl acetate (v/v)=8:1 ) Purified to obtain the title compound tert-butyl (3-hydroxy-3-methylcyclohexyl) carbamate (27b) (white solid, 2.07 g, yield 38.53%).
1H NMR(400MHz,Chloroform-d)δ4.30(s,1H),3.68(s,1H),1.90(d,3H),1.67(q,1H),1.59–1.48(m,2H),1.37(s,9H),1.19–1.12(m,4H),1.06(t,1H),0.96–0.84(m,1H) 。1H NMR (400MHz, Chloroform-d) δ 4.30 (s, 1H), 3.68 (s, 1H), 1.90 (d, 3H), 1.67 (q, 1H), 1.59-1.48 (m, 2H), 1.37 ( s,9H),1.19–1.12(m,4H),1.06(t,1H),0.96–0.84(m,1H).
LC-MS m/z(ESI)=230.20[M+1]。LC-MS m/z(ESI)=230.20[M+1].
第二步:The second step:
3-氨基-1-甲基环己烷-1-醇盐酸盐(27c)3-amino-1-methylcyclohexane-1-ol hydrochloride (27c)
3-amino-1-methylcyclohexan-1-ol hydrochloride3-amino-1-methylcyclohexan-1-ol hydrochloride
将叔丁基(3-羟基-3-甲基环己基)氨基甲酸酯27b(2.07g,9.03mmol)用4M氯化氢-1,4-二氧六环(10mL)溶解,常温搅拌反应。TLC监测至反应结束,浓缩蒸发掉1,4-二氧六环溶液得标题化合物3-氨基-1-甲基环己烷-1-醇盐酸盐(27c)(淡黄色固体,粗品,1.49g,产率99.60%)。Tert-butyl (3-hydroxy-3-methylcyclohexyl) carbamate 27b (2.07 g, 9.03 mmol) was dissolved in 4M hydrogen chloride-1,4-dioxane (10 mL), and the reaction was stirred at room temperature. TLC monitors to the end of the reaction, concentrates and evaporates the 1,4-dioxane solution to obtain the title compound 3-amino-1-methylcyclohexane-1-ol hydrochloride (27c) (light yellow solid, crude product, 1.49) g, yield 99.60%).
LC-MS m/z(ESI)=130.20[M+1]。LC-MS m/z(ESI)=130.20[M+1].
第三步:third step:
2-氯-4-((3-羟基-3-甲基环己基)氨基)嘧啶-5-羧酸乙酯(27d)Ethyl 2-chloro-4-((3-hydroxy-3-methylcyclohexyl)amino)pyrimidine-5-carboxylate (27d)
ethyl 2-chloro-4-((3-hydroxy-3-methylcyclohexyl)amino)pyrimidine-5-carboxyl-ateethyl 2-chloro-4-((3-hydroxy-3-methylcyclohexyl)amino)pyrimidine-5-carboxyl-ate
将2,4-二氯嘧啶-5-羧酸乙酯1a(2.99g,13.54mmol)、3-氨基-1-甲基环己烷-1-醇盐酸盐27c(1.49g,9.03mmol)溶解于乙腈(20mL),搅拌加入碳酸钾(3.74g,27.08mmol),在室温搅拌4h。TLC监测至反应结束,浓缩通过柱分离提纯(石油醚:乙酸乙酯(v/v)=4:1)得标题化合物2-氯-4-((3-羟基-3-甲基环己基)氨基)嘧啶-5-羧酸乙酯(27d)(白色固体,2.23g,产率78.74%)。2,4-Dichloropyrimidine-5-carboxylic acid ethyl ester 1a (2.99g, 13.54mmol), 3-amino-1-methylcyclohexane-1-ol hydrochloride 27c (1.49g, 9.03mmol) Dissolved in acetonitrile (20 mL), stirred and added potassium carbonate (3.74 g, 27.08 mmol), and stirred at room temperature for 4 h. TLC monitors to the end of the reaction, concentrates and purifies by column separation (petroleum ether: ethyl acetate (v/v)=4:1) to obtain the title compound 2-chloro-4-((3-hydroxy-3-methylcyclohexyl) Amino)pyrimidine-5-carboxylic acid ethyl ester (27d) (white solid, 2.23 g, yield 78.74%).
1H NMR(400MHz,Chloroform-d)δ8.57(s,1H),8.19(d,1H),4.43–4.32(m,1H),4.27(q,2H),2.07–1.95(m,2H),1.83–1.69(m,2H),1.64–1.56(m,2H),1.31(t,4H),1.26(d,1H),1.21(s,3H),1.10–1.05(m,1H)。1H NMR(400MHz,Chloroform-d)δ8.57(s,1H), 8.19(d,1H), 4.43-4.32(m,1H), 4.27(q,2H), 2.07-1.95(m,2H), 1.83–1.69(m,2H), 1.64–1.56(m,2H), 1.31(t,4H), 1.26(d,1H), 1.21(s,3H), 1.10–1.05(m,1H).
LC-MS m/z(ESI)=314.10[M+1]。LC-MS m/z(ESI)=314.10[M+1].
第四步:the fourth step:
2-氯-4-((3-羟基-3-甲基环己基)氨基)嘧啶-5-羧酸(27e)2-chloro-4-((3-hydroxy-3-methylcyclohexyl)amino)pyrimidine-5-carboxylic acid (27e)
2-chloro-4-((3-hydroxy-3-methylcyclohexyl)amino)pyrimidine-5-carboxylate2-chloro-4-((3-hydroxy-3-methylcyclohexyl)amino)pyrimidine-5-carboxylate
将2-氯-4-((3-羟基-3-甲基环己基)氨基)嘧啶-5-羧酸乙酯27d(2.23g,7.11mmol)溶解于四氢呋喃/水(15mL/15mL)中,加入氢氧化锂(895mg,21.32mmol),室温搅拌1h。TLC监测至反应结束,浓缩除去四氢呋喃,用2N盐酸调pH至3-4,有白色固体析出,过滤,滤饼用石油醚洗两次,搜集固体得到标题化合物2-氯-4-((3-羟基-3-甲基环己基)氨基)嘧啶-5-羧酸(27e)(白色固体,1.6g,产率78.79%)。Dissolve 2-chloro-4-((3-hydroxy-3-methylcyclohexyl)amino)pyrimidine-5-carboxylic acid ethyl ester 27d (2.23g, 7.11mmol) in tetrahydrofuran/water (15mL/15mL), Lithium hydroxide (895 mg, 21.32 mmol) was added, and the mixture was stirred at room temperature for 1 h. TLC monitors until the reaction is complete, concentrates to remove tetrahydrofuran, adjusts the pH to 3-4 with 2N hydrochloric acid, a white solid is precipitated, filtered, the filter cake is washed twice with petroleum ether, and the solid is collected to obtain the title compound 2-chloro-4-((3 -Hydroxy-3-methylcyclohexyl)amino)pyrimidine-5-carboxylic acid (27e) (white solid, 1.6g, yield 78.79%).
LC-MS m/z(ESI)=286.10[M+1]。LC-MS m/z(ESI)=286.10[M+1].
第五步:the fifth step:
2-氯-9-(3-羟基-3-甲基环己基)-7,9-二氢-8H-嘌呤-8-酮(27e)2-chloro-9-(3-hydroxy-3-methylcyclohexyl)-7,9-dihydro-8H-purin-8-one (27e)
2-chloro-9-(3-hydroxy-3-methylcyclohexyl)-7,9-dihydro-8H-purin-8-one2-chloro-9-(3-hydroxy-3-methylcyclohexyl)-7,9-dihydro-8H-purin-8-one
将2-氯-4-((3-羟基-3-甲基环己基)氨基)嘧啶-5-羧酸27d(1.38g,4.83mmol)溶解于二甲基乙酰胺(20mL),加入三乙胺(0.67mL,4.83mmol)、叠氮磷酸二苯酯(1.04mL,4.83mmol),随后逐步升温至90℃搅拌2h。TLC监测至反应结束,将反应液倒入冰水中,过滤搜集固体,水洗3次,真空浓缩干燥得到目标化合物2-氯-9-(3-羟基-3-甲基环己基)-7,9-二氢-8H-嘌呤-8-酮(27e)(白色固体,1.35g,产率98.86%)。Dissolve 2-chloro-4-((3-hydroxy-3-methylcyclohexyl)amino)pyrimidine-5-carboxylic acid 27d (1.38g, 4.83mmol) in dimethylacetamide (20mL) and add triethyl Amine (0.67mL, 4.83mmol), diphenyl azide phosphate (1.04mL, 4.83mmol), then gradually heated to 90°C and stirred for 2h. TLC monitors to the end of the reaction, the reaction solution is poured into ice water, the solid is collected by filtration, washed with water 3 times, and concentrated and dried in vacuo to obtain the target compound 2-chloro-9-(3-hydroxy-3-methylcyclohexyl)-7,9 -Dihydro-8H-purin-8-one (27e) (white solid, 1.35 g, yield 98.86%).
1H NMR(400MHz,DMSO-d6)δ11.60(s,1H),8.10(s,1H),4.62-4.54(m,1H),4.35(s,1H),2.19(t,1H),2.10-1.99(m,1H),1.70-1.53(m,5H),1.30-1.22(m,1H),1.16(s,3H)。1H NMR (400MHz, DMSO-d6) δ 11.60 (s, 1H), 8.10 (s, 1H), 4.62-4.54 (m, 1H), 4.35 (s, 1H), 2.19 (t, 1H), 2.10- 1.99 (m, 1H), 1.70 to 1.53 (m, 5H), 1.30 to 1.22 (m, 1H), 1.16 (s, 3H).
LC-MS m/z(ESI)=283.10[M+1]。LC-MS m/z(ESI)=283.10[M+1].
第六步:The sixth step:
2-氯-9-(3-羟基-3-甲基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(27f)2-chloro-9-(3-hydroxy-3-methylcyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one (27f)
2-chloro-9-(3-hydroxy-3-methylcyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one2-chloro-9-(3-hydroxy-3-methylcyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one
将2-氯-9-(3-羟基-3-甲基环己基)-7,9-二氢-8H-嘌呤-8-酮27e(1.35g,4.77mmol)溶 解于二甲基甲酰胺(10mL),在0℃下加入硫酸二甲酯(0.45mL,4.77mmol)和碳酸铯(1.56g,4.77mmol),搅拌反应1h。TLC监测至反应结束,将反应液倒入冰水中,有固体析出,过滤、搜集固体得到标题化合物2-氯-9-(3-羟基-3-甲基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(27f)(白色固体,873mg,产率61.61%)。Dissolve 2-chloro-9-(3-hydroxy-3-methylcyclohexyl)-7,9-dihydro-8H-purin-8-one 27e (1.35g, 4.77mmol) in dimethylformamide ( 10 mL), dimethyl sulfate (0.45 mL, 4.77 mmol) and cesium carbonate (1.56 g, 4.77 mmol) were added at 0°C, and the reaction was stirred for 1 h. TLC monitors to the end of the reaction, the reaction solution is poured into ice water, a solid precipitates out, filtered and collected the solid to obtain the title compound 2-chloro-9-(3-hydroxy-3-methylcyclohexyl)-7-methyl-7 , 9-dihydro-8H-purin-8-one (27f) (white solid, 873 mg, yield 61.61%).
1H NMR(400MHz,DMSO-d6)δ8.34(s,1H),4.66-4.58(m,1H),4.37(s,1H),3.35(s,3H),2.20(t,1H),2.11-2.00(m,1H),1.72-1.54(m,5H),1.32-1.24(m,1H),1.17(s,3H)。1H NMR (400MHz, DMSO-d6) δ 8.34 (s, 1H), 4.66-4.58 (m, 1H), 4.37 (s, 1H), 3.35 (s, 3H), 2.20 (t, 1H), 2.11 2.00 (m, 1H), 1.72-1.54 (m, 5H), 1.32-1.24 (m, 1H), 1.17 (s, 3H).
LC-MS m/z(ESI)=297.10[M+1]。LC-MS m/z(ESI)=297.10[M+1].
第七步:The seventh step:
9-(3-羟基-3-甲基环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物27)9-(3-hydroxy-3-methylcyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9- Dihydro-8H-purin-8-one (Compound 27)
9-(3-hydroxy-3-methylcyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(3-hydroxy-3-methylcyclohexyl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one
将2-氯-9-(3-羟基-3-甲基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮27f(100mg,0.34mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(100mg,0.67mmol)、碳酸铯(220mg,0.67mmol)、Brettphos G3 Pd(30mg,0.034mmol)加入干燥的反应瓶,氮气置换三次后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化得到标题化合物9-(3-羟基-3-甲基环己基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物27)(浅粉色固体,80mg,产率58.01%)。The 2-chloro-9-(3-hydroxy-3-methylcyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one 27f (100mg, 0.34mmol), 6-methyl 2-,3-dihydrobenzofuran-5-amine intermediate 1 (100mg, 0.67mmol), cesium carbonate (220mg, 0.67mmol), Brettphos G3 Pd (30mg, 0.034mmol) into the dry reaction flask, nitrogen After three replacements, dry 1,4-dioxane (1 mL) was added and reacted at 110° C. for 5 h. TLC monitors until the reaction is complete. After cooling to room temperature, it is separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 9-(3-hydroxy-3-methylcyclohexyl) -7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one (compound 27) (Light pink solid, 80 mg, yield 58.01%).
1H NMR(400MHz,DMSO-d6)δ8.21(s,1H),7.94(s,1H),7.21(s,1H),6.60(s,1H),4.58–4.50(m,1H),4.48(t,2H),4.27(s,1H),3.25(s,3H),3.10(t,2H),2.26(t,1H),2.11(s,3H),2.04–1.96(m,1H),1.74–1.60(m,2H),1.58–1.49(m,3H),1.19–1.13(m,1H),1.12(s,3H)。1H NMR(400MHz,DMSO-d6)δ8.21(s,1H),7.94(s,1H),7.21(s,1H),6.60(s,1H),4.58-4.50(m,1H), 4.48( t, 2H), 4.27(s, 1H), 3.25(s, 3H), 3.10(t, 2H), 2.26(t, 1H), 2.11(s, 3H), 2.04-1.96(m, 1H), 1.74 –1.60(m,2H), 1.58–1.49(m,3H), 1.19–1.13(m,1H), 1.12(s,3H).
LC-MS m/z(ESI)=410.20[M+1]。LC-MS m/z(ESI)=410.20[M+1].
实施例28Example 28
7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(2-氧杂菲罗[3.3]庚烷-6-基)-7,9-二氢-8H-嘌呤-8-酮(化合物28)7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(2-oxapheno[3.3]heptane-6-yl) -7,9-dihydro-8H-purin-8-one (compound 28)
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(2-oxaspiro[3.3]heptan-6-yl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(2-oxaspiro[3.3]heptan-6-yl)-7,9-dihydro-8H-purin -8-one
第一步:first step:
4-((2-氧杂螺[3.3]庚-6-6基)氨基)-2-氯嘧啶-5-羧酸乙酯(28a)4-((2-oxaspiro[3.3]hept-6-6 yl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester (28a)
ethyl 4-((2-oxaspiro[3.3]heptan-6-yl)amino)-2-chloropyrimidine-5-carboxylateethyl 4-((2-oxaspiro[3.3]heptan-6-yl)amino)-2-chloropyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(4.43g,20.05mmol),2-氧杂螺[3.3]庚-6-胺盐酸盐(2.0g,13.37mmol),碳酸钾(5.54g,40.10mmol)溶于乙腈(60mL),反应液在室温反应16h。TLC监测反应结束,过滤,并用少量乙腈清洗固体,将滤液合并后浓缩,粗品经柱层析分离(石油醚:乙酸乙酯=1:1)后得目标化合4-((2-氧杂螺[3.3]庚-6-6基)氨基)-2-氯嘧啶-5-羧酸乙酯(28a)(白色固体,3.0g,产率75.38%)。2,4-Dichloropyrimidine-5-carboxylic acid ethyl ester 1a (4.43g, 20.05mmol), 2-oxaspiro[3.3]heptan-6-amine hydrochloride (2.0g, 13.37mmol), potassium carbonate (5.54g, 40.10mmol) was dissolved in acetonitrile (60mL), and the reaction solution was reacted at room temperature for 16h. The end of the reaction was monitored by TLC, filtered, and the solid was washed with a small amount of acetonitrile. The filtrate was combined and concentrated. The crude product was separated by column chromatography (petroleum ether: ethyl acetate = 1:1) to obtain the target compound 4-((2-oxaspiro) [3.3]Heptan-6-6yl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester (28a) (white solid, 3.0 g, yield 75.38%).
1H NMR(400MHz,DMSO-d6)δ8.60(s,1H),8.47(d,1H),4.63(s,2H),4.49(s,2H),4.39-4.33(m,1H),4.30-4.27(q,2H),2.66-2.61(m,2H),2.31-2.26(m,2H),1.30(t,3H)。1H NMR(400MHz,DMSO-d6)δ8.60(s,1H), 8.47(d,1H), 4.63(s,2H), 4.49(s,2H), 4.39-4.33(m,1H), 4.30- 4.27 (q, 2H), 2.66-2.61 (m, 2H), 2.31-2.26 (m, 2H), 1.30 (t, 3H).
LC-MS m/z(ESI)=298.10[M+1]。LC-MS m/z(ESI)=298.10[M+1].
第二步:The second step:
4-((2-氧杂螺[3.3]庚-6-6基)氨基)-2-氯嘧啶-5-羧酸(28b)4-((2-oxaspiro[3.3]hept-6-6 yl)amino)-2-chloropyrimidine-5-carboxylic acid (28b)
4-((2-oxaspiro[3.3]heptan-6-yl)amino)-2-chloropyrimidine-5-carboxylic acid4-((2-oxaspiro[3.3]heptan-6-yl)amino)-2-chloropyrimidine-5-carboxylic acid
将4-((2-氧杂螺[3.3]庚-6-6基)氨基)-2-氯嘧啶-5-羧酸乙酯280a(3.0g,10.08mmol)溶解于四氢呋喃/水(30mL/30mL)中,加入氢氧化锂(845mg,20.15mmol),室温搅拌1h。TLC监测至反应结束,浓缩除去四氢呋喃,用2N盐酸调pH为5,有白色固体析出,过滤,滤饼用石油醚洗两次,搜集固体得到标题化合物4-((2-氧杂螺[3.3]庚-6-6基)氨基)-2-氯嘧啶-5-羧酸(28b)(白色固体,1.8g,产率66.24%)。4-((2-oxaspiro[3.3]hept-6-6 yl)amino)-2-chloropyrimidine-5-carboxylic acid ethyl ester 280a (3.0g, 10.08mmol) was dissolved in tetrahydrofuran/water (30mL/ 30 mL), lithium hydroxide (845 mg, 20.15 mmol) was added, and the mixture was stirred at room temperature for 1 h. TLC monitors until the reaction is complete, concentrates to remove tetrahydrofuran, adjusts the pH to 5 with 2N hydrochloric acid, a white solid is precipitated, filtered, the filter cake is washed twice with petroleum ether, and the solid is collected to obtain the title compound 4-((2-oxaspiro[3.3 ]Hept-6-6yl)amino)-2-chloropyrimidine-5-carboxylic acid (28b) (white solid, 1.8g, yield 66.24%).
1H NMR(400MHz,DMSO-d6)δ13.76(s,1H),8.64(d,1H),8.57(s,1H),4.64(s,2H),4.49(s,2H),4.40-4.30(m,1H),2.67-2.61(m,2H),2.28-2.23(m,2H)。1H NMR (400MHz, DMSO-d6) δ 13.76 (s, 1H), 8.64 (d, 1H), 8.57 (s, 1H), 4.64 (s, 2H), 4.49 (s, 2H), 4.40-4.30 ( m, 1H), 2.67-2.61 (m, 2H), 2.28-2.23 (m, 2H).
LC-MS m/z(ESI)=270.20[M+1]。LC-MS m/z(ESI)=270.20[M+1].
第三步:third step:
2-氯-9-(2-氧杂螺[3.3]庚-6-基)-7,9-二氢-8H-嘌呤-8-酮(28c)2-Chloro-9-(2-oxaspiro[3.3]heptan-6-yl)-7,9-dihydro-8H-purin-8-one (28c)
2-chloro-9-(2-oxaspiro[3.3]heptan-6-yl)-7,9-dihydro-8H-purin-8-one2-chloro-9-(2-oxaspiro[3.3]heptan-6-yl)-7,9-dihydro-8H-purin-8-one
将4-((2-氧杂螺[3.3]庚-6-6基)氨基)-2-氯嘧啶-5-羧酸28b(1.8g,6.67mmol)溶解于二甲基乙酰胺(40mL),加入三乙胺(0.92mL,6.67mmol)、叠氮磷酸二苯酯(1.4mL,6.67mmol),随后逐步升温至90℃搅拌反应2h。TLC监测至反应结束,将反应液倒入冰水中,过滤搜集固体,水洗3次,真空浓缩干燥得2-氯-9-(2-氧杂螺[3.3]庚-6-基)-7,9-二氢-8H-嘌呤-8-酮(28c)(白色固体,1.3g,产率73.03%)。Dissolve 4-((2-oxaspiro[3.3]hept-6-6 yl)amino)-2-chloropyrimidine-5-carboxylic acid 28b (1.8g, 6.67mmol) in dimethylacetamide (40mL) , Triethylamine (0.92mL, 6.67mmol) and diphenyl azide phosphate (1.4mL, 6.67mmol) were added, and then gradually heated to 90°C and stirred for 2h. TLC monitors to the end of the reaction, the reaction solution is poured into ice water, the solid is collected by filtration, washed 3 times with water, concentrated and dried in vacuo to obtain 2-chloro-9-(2-oxaspiro[3.3]heptan-6-yl)-7, 9-dihydro-8H-purin-8-one (28c) (white solid, 1.3 g, yield 73.03%).
1H NMR(400MHz,DMSO-d6)δ11.61(s,1H),8.11(s,1H),4.71-4.63(m,5H),3.02-2.94(m,2H),2.69-2.66(m,2H)。1H NMR(400MHz,DMSO-d6)δ11.61(s,1H),8.11(s,1H),4.71-4.63(m,5H),3.02-2.94(m,2H),2.69-2.66(m,2H) ).
LC-MS m/z(ESI)=267.10[M+1]。LC-MS m/z(ESI)=267.10[M+1].
第四步:the fourth step:
2-氯-7-甲基-9-(2-氧杂螺[3.3]庚-6-基)-7,9-二氢-8H-嘌呤-8-酮(28d)2-Chloro-7-methyl-9-(2-oxaspiro[3.3]heptan-6-yl)-7,9-dihydro-8H-purin-8-one (28d)
2-chloro-7-methyl-9-(2-oxaspiro[3.3]heptan-6-yl)-7,9-dihydro-8H-purin-8-one2-chloro-7-methyl-9-(2-oxaspiro[3.3]heptan-6-yl)-7,9-dihydro-8H-purin-8-one
将2-氯-9-(2-氧杂螺[3.3]庚-6-基)-7,9-二氢-8H-嘌呤-8-酮28c(1.3g,4.87mmol)溶解于二甲基甲酰胺(10mL),在0℃下加入硫酸二甲酯(0.46mL,4.87mmol)和碳酸铯(3.18g,9.75mmol),搅拌反应1h。TLC监测至反应结束,将反应液倒入冰水中,有固体析出,过滤、搜集固体得到标题化合物2-氯-7-甲基-9-(2-氧杂螺[3.3]庚-6-基)-7,9-二氢-8H-嘌呤-8-酮(28d)(白色固体,875mg,产率63.94%)。Dissolve 2-chloro-9-(2-oxaspiro[3.3]hept-6-yl)-7,9-dihydro-8H-purin-8-one 28c (1.3g, 4.87mmol) in dimethyl Formamide (10 mL), dimethyl sulfate (0.46 mL, 4.87 mmol) and cesium carbonate (3.18 g, 9.75 mmol) were added at 0°C, and the reaction was stirred for 1 h. TLC monitors to the end of the reaction, the reaction solution is poured into ice water, a solid precipitates out, filtered and collected the solid to obtain the title compound 2-chloro-7-methyl-9-(2-oxaspiro[3.3]hept-6-yl )-7,9-dihydro-8H-purin-8-one (28d) (white solid, 875mg, yield 63.94%).
1H NMR(400MHz,DMSO-d6)δ8.35(s,1H),4.76-4.69(m,1H),4.66-4.63(m,4H),3.33(s,3H),3.01-2.95(m,2H),2.71-2.66(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.35(s,1H),4.76-4.69(m,1H),4.66-4.63(m,4H),3.33(s,3H),3.01-2.95(m,2H) ), 2.71-2.66 (m, 2H).
LC-MS m/z(ESI)=281.10[M+1]。LC-MS m/z(ESI)=281.10[M+1].
第五步:the fifth step:
7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(2-氧杂菲罗[3.3]庚烷-6-基)-7,9-二氢-8H-嘌呤-8-酮(化合物28)7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(2-oxapheno[3.3]heptane-6-yl) -7,9-dihydro-8H-purin-8-one (compound 28)
7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(2-oxaspiro[3.3]heptan-6-yl)-7,9-dihydro-8H-purin-8-one7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(2-oxaspiro[3.3]heptan-6-yl)-7,9-dihydro-8H-purin -8-one
将2-氯-7-甲基-9-(2-氧杂螺[3.3]庚-6-基)-7,9-二氢-8H-嘌呤-8-酮28d(50mg,0.18mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(53mg,0.36mmol)、碳酸铯(116mg,0.36mmol)、Brettphos G3 Pd(16mg,0.018mmol)加入干燥的反应瓶,氮气置换三次后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化得到标题化合物7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(2-氧杂菲罗[3.3]庚烷-6-基)-7,9-二氢-8H-嘌呤-8-酮(化合物28)(浅粉色固体,37mg,产率52.80%)。The 2-chloro-7-methyl-9-(2-oxaspiro[3.3]heptan-6-yl)-7,9-dihydro-8H-purin-8-one 28d (50mg, 0.18mmol), 6-Methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (53mg, 0.36mmol), cesium carbonate (116mg, 0.36mmol), Brettphos G3 Pd (16mg, 0.018mmol) were added to the dry reaction After replacing the flask with nitrogen three times, add dry 1,4-dioxane (1 mL), and react at 110°C for 5 hours. The reaction was monitored by TLC until the end of the reaction. After cooling to room temperature, it was separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 7-methyl-2-((6-methyl- 2,3-Dihydrobenzofuran-5-yl)amino)-9-(2-oxafero[3.3]heptane-6-yl)-7,9-dihydro-8H-purine-8- Ketone (Compound 28) (light pink solid, 37 mg, yield 52.80%).
1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),7.97(s,1H),7.09(s,1H),6.63(s,1H),4.63–4.55(m,3H),4.48(t,2H),4.31(s,2H),3.23(s,3H),3.12(t,2H),2.99–2.90(m,2H),2.55–2.48(m,2H),2.08(s,3H)。1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),7.97(s,1H),7.09(s,1H),6.63(s,1H),4.63-4.55(m,3H),4.48( t, 2H), 4.31 (s, 2H), 3.23 (s, 3H), 3.12 (t, 2H), 2.99-2.90 (m, 2H), 2.55-2.48 (m, 2H), 2.08 (s, 3H) .
LC-MS m/z(ESI)=394.20[M+1]。LC-MS m/z(ESI)=394.20[M+1].
实施例29Example 29
9-(6-(羟甲基)螺[3.3]庚烷-2-基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮9-(6-(Hydroxymethyl)spiro[3.3]heptan-2-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl) Amino)-7,9-dihydro-8H-purin-8-one
9-(6-(hydroxymethyl)spiro[3.3]heptan-2-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(6-(hydroxymethyl)spiro[3.3]heptan-2-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro- 8H-purin-8-one
第一步:first step:
(6-氨基螺[3.3]庚烷-2-基)甲醇(29b)(6-Aminospiro[3.3]heptan-2-yl)methanol (29b)
(6-aminospiro[3.3]heptan-2-yl)methanol(6-aminospiro[3.3]heptan-2-yl)methanol
将6-氨基螺[3.3]庚烷-2-羧酸甲酯29a(2.0g,11.8mmol)溶于四氢呋喃(15mL)中,混合液降温至0℃,缓慢滴加1N的四氢铝锂溶液(23.6mL,1mol/L的四氢呋喃溶液),滴加完成后,缓慢升温至室温搅拌2h。TLC监测至反应结束,加入甲醇淬灭反应,减压浓缩蒸发掉溶剂,加入乙腈(50mL)打浆过滤,滤液旋干得到标题化合物(6-氨基螺[3.3]庚烷-2-基)甲醇(29b)(白色固体,1.3g,产率77.8%)。Methyl 6-aminospiro[3.3]heptane-2-carboxylate 29a (2.0g, 11.8mmol) was dissolved in tetrahydrofuran (15mL), the mixture was cooled to 0℃, and 1N lithium aluminum tetrahydrogen solution was slowly added dropwise (23.6mL, 1mol/L tetrahydrofuran solution), after the dripping is completed, slowly warm up to room temperature and stir for 2h. The reaction was monitored by TLC until the reaction was over, methanol was added to quench the reaction, concentrated under reduced pressure to evaporate the solvent, acetonitrile (50 mL) was added to make a slurry filter, and the filtrate was spin-dried to obtain the title compound (6-aminospiro[3.3]heptan-2-yl)methanol ( 29b) (white solid, 1.3 g, yield 77.8%).
LC-MS m/z(ESI)=142.10[M+1]。LC-MS m/z(ESI)=142.10[M+1].
第二步:The second step:
2-氯-4-((6-(羟甲基)螺[3.3]庚烷-2-基)氨基)嘧啶-5-羧酸乙酯(29c)Ethyl 2-chloro-4-((6-(hydroxymethyl)spiro[3.3]heptan-2-yl)amino)pyrimidine-5-carboxylate (29c)
ethyl 2-chloro-4-((6-(hydroxymethyl)spiro[3.3]heptan-2-yl)amino)pyrimidine-5-carboxylateethyl 2-chloro-4-((6-(hydroxymethyl)spiro[3.3]heptan-2-yl)amino)pyrimidine-5-carboxylate
将2,4-二氯嘧啶-5-羧酸乙酯1a(2.03g,9.19mmol),(6-氨基螺[3.3]庚烷-2-基)甲醇29b(1.3g,9.19mmol),碳酸钾(1.27g,9.19mmol)溶于乙腈(25mL)中,室温搅拌反应16h。TLC监测至反应结束,滤液浓缩经硅胶柱色谱分离提纯(石油醚:乙酸乙酯(v/v)=1:1)纯化得到标题化合物2-氯-4-((6-(羟甲基)螺[3.3]庚烷-2-基)氨基)嘧啶-5-羧酸乙酯(29c)(黄色固体,1.56g,产率52.2%)。Ethyl 2,4-dichloropyrimidine-5-carboxylate 1a (2.03g, 9.19mmol), (6-aminospiro[3.3]heptan-2-yl)methanol 29b (1.3g, 9.19mmol), carbonic acid Potassium (1.27g, 9.19mmol) was dissolved in acetonitrile (25mL), and the reaction was stirred at room temperature for 16h. TLC monitoring until the end of the reaction, the filtrate was concentrated and purified by silica gel column chromatography (petroleum ether: ethyl acetate (v/v) = 1: 1) to obtain the title compound 2-chloro-4-((6-(hydroxymethyl) Spiro[3.3]heptan-2-yl)amino)pyrimidine-5-carboxylic acid ethyl ester (29c) (yellow solid, 1.56 g, yield 52.2%).
1H NMR(400MHz,Chloroform-d)δ8.65(s,1H),8.54(d,1H),4.59–4.48(m,1H),4.35(q,2H),3.59(dd,2H),2.66–2.58(m,1H),2.51–2.38(m,2H),2.27–2.19(m,1H),2.11–2.04(m,2H),2.03–1.91(m,2H),1.91–1.85(m,1H),1.84–1.76(m,1H),1.39(t,3H)。1H NMR(400MHz,Chloroform-d)δ8.65(s,1H),8.54(d,1H),4.59--4.48(m,1H),4.35(q,2H),3.59(dd,2H),2.66-- 2.58 (m, 1H), 2.51-2.38 (m, 2H), 2.27-2.19 (m, 1H), 2.11-2.04 (m, 2H), 2.03-1.91 (m, 2H), 1.91-1.85 (m, 1H) ), 1.84-1.76 (m, 1H), 1.39 (t, 3H).
LCMS m/z(ESI)=326.10[M+1]。LCMS m/z(ESI)=326.10[M+1].
第三步:third step:
2-氯-4-((6-(羟甲基)螺[3.3]庚烷-2-基)氨基)嘧啶-5-羧酸(29d)2-chloro-4-((6-(hydroxymethyl)spiro[3.3]heptan-2-yl)amino)pyrimidine-5-carboxylic acid (29d)
2-chloro-4-((6-(hydroxymethyl)spiro[3.3]heptan-2-yl)amino)pyrimidine-5-carboxylic acid2-chloro-4-((6-(hydroxymethyl)spiro[3.3]heptan-2-yl)amino)pyrimidine-5-carboxylic acid
将2-氯-4-((6-(羟甲基)螺[3.3]庚烷-2-基)氨基)嘧啶-5-羧酸乙酯29c(1.07g,3.28 mmol)溶解于四氢呋喃/水(10mL/10mL)中,加入氢氧化锂一水合物(0.41g,9.85mmol),常温搅拌反应1h。TLC监测至反应结束,浓缩蒸发掉四氢呋喃后加入2N HCl调节pH至3-4左右,有白色固体析出,过滤,滤饼用水以及石油醚/乙酸乙酯(v/v=10/1)洗两次得标题化合物2-氯-4-((6-(羟甲基)螺[3.3]庚烷-2-基)氨基)嘧啶-5-羧酸(29d)(白色固体,918mg,产率93.88%)。Dissolve 2-chloro-4-((6-(hydroxymethyl)spiro[3.3]heptan-2-yl)amino)pyrimidine-5-carboxylic acid ethyl ester 29c (1.07g, 3.28 mmol) in tetrahydrofuran/water (10mL/10mL), lithium hydroxide monohydrate (0.41g, 9.85mmol) was added, and the reaction was stirred at room temperature for 1h. TLC monitors to the end of the reaction, concentrates and evaporates the tetrahydrofuran, adds 2N HCl to adjust the pH to about 3-4, a white solid is precipitated, filtered, and the filter cake is washed twice with water and petroleum ether/ethyl acetate (v/v=10/1) The title compound 2-chloro-4-((6-(hydroxymethyl)spiro[3.3]heptan-2-yl)amino)pyrimidine-5-carboxylic acid (29d) (white solid, 918mg, yield 93.88) %).
1H NMR(400MHz,DMSO-d6)δ12.69(s,1H),9.67(d,1H),8.35(s,1H),4.42–4.31(m,1H),3.30(s,1H),2.55–2.46(m,3H),2.40–2.31(m,1H),2.28–2.19(m,1H),2.10–2.00(m,2H),2.00–1.91(m,2H),1.83–1.69(m,2H)。1H NMR(400MHz,DMSO-d6)δ12.69(s,1H),9.67(d,1H),8.35(s,1H),4.42-4.31(m,1H),3.30(s,1H),2.55-- 2.46 (m, 3H), 2.40-2.31 (m, 1H), 2.28-2.19 (m, 1H), 2.10-2.00 (m, 2H), 2.00-1.91 (m, 2H), 1.83-1.69 (m, 2H) ).
LC-MS m/z(ESI)=298.10[M+1]。LC-MS m/z(ESI)=298.10[M+1].
第四步:the fourth step:
2-氯-9-(6-(羟甲基)螺[3.3]庚烷-2-基)-7,9-二氢-8H-嘌呤-8-酮(29e)2-chloro-9-(6-(hydroxymethyl)spiro[3.3]heptan-2-yl)-7,9-dihydro-8H-purin-8-one (29e)
2-chloro-9-(6-(hydroxymethyl)spiro[3.3]heptan-2-yl)-7,9-dihydro-8H-purin-8-one2-chloro-9-(6-(hydroxymethyl)spiro[3.3]heptan-2-yl)-7,9-dihydro-8H-purin-8-one
将2-氯-4-((6-(羟甲基)螺[3.3]庚烷-2-基)氨基)嘧啶-5-羧酸29d(0.91g,3.06mmol)用N,N-二甲基乙酰胺(12mL)溶解,常温搅拌下加入三乙胺(0.42mL,3.06mmol)和叠氮磷酸二苯酯(0.66mL,10.01mmol)反应2h,升温至110℃回流反应2.5h。TLC监测至反应结束,反应液中加水和乙酸乙酯进行萃取,浓缩有机层经硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v)=20:1)纯化得标题化合物2-氯-9-(6-(羟甲基)螺[3.3]庚烷-2-基)-7,9-二氢-8H-嘌呤-8-酮(29e)(白色固体,516mg,产率57.28%)。Use 2-chloro-4-((6-(hydroxymethyl)spiro[3.3]heptan-2-yl)amino)pyrimidine-5-carboxylic acid 29d (0.91g, 3.06mmol) with N,N-dimethyl Acetamide (12 mL) was dissolved, triethylamine (0.42 mL, 3.06 mmol) and diphenyl azide phosphate (0.66 mL, 10.01 mmol) were added under stirring at room temperature for 2 hours, and the temperature was raised to 110° C. and refluxed for 2.5 hours. TLC monitors to the end of the reaction, the reaction solution is added with water and ethyl acetate for extraction, the organic layer is concentrated and purified by silica gel column chromatography (dichloromethane/methanol (v/v)=20:1) to obtain the title compound 2-chloro- 9-(6-(hydroxymethyl)spiro[3.3]heptan-2-yl)-7,9-dihydro-8H-purin-8-one (29e) (white solid, 516mg, yield 57.28%) .
1H NMR(400MHz,DMSO-d6)δ11.60(s,1H),8.11(s,1H),4.68–4.57(m,1H),4.46(t,1H),3.37–3.32(m,2H),2.95–2.81(m,2H),2.42–2.35(m,1H),2.32–2.19(m,2H),2.19–2.12(m,1H),2.06–1.98(m,1H),1.89–1.77(m,2H)。1H NMR(400MHz,DMSO-d6)δ11.60(s,1H),8.11(s,1H), 4.68-4.57(m,1H), 4.46(t,1H), 3.37-3.32(m,2H), 2.95–2.81(m,2H), 2.42–2.35(m,1H), 2.32–2.19(m,2H), 2.19–2.12(m,1H), 2.06–1.98(m,1H), 1.89–1.77(m ,2H).
LC-MS m/z(ESI)=295.10[M+1]。LC-MS m/z(ESI)=295.10[M+1].
第五步:the fifth step:
2-氯-9-(6-(羟甲基)螺[3.3]庚烷-2-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(29f)2-chloro-9-(6-(hydroxymethyl)spiro[3.3]heptan-2-yl)-7-methyl-7,9-dihydro-8H-purin-8-one (29f)
2-chloro-9-(6-(hydroxymethyl)spiro[3.3]heptan-2-yl)-7-methyl-7,9-dihydro-8H-purin-8-one2-chloro-9-(6-(hydroxymethyl)spiro[3.3]heptan-2-yl)-7-methyl-7,9-dihydro-8H-purin-8-one
将2-氯-9-(6-(羟甲基)螺[3.3]庚烷-2-基)-7,9-二氢-8H-嘌呤-8-酮29e(516mg,1.75mmol)用N,N-二甲基甲酰胺(6mL)溶解,0℃搅拌下加入硫酸二甲酯(0.17mL,1.75mmol)和碳酸铯(1.14g,3.50mmol)反应1h。TLC监测至反应结束,把反应液缓慢滴加到冰水中搅拌,加入乙酸乙酯进行萃取,浓缩有机层得标题化合物2-氯-9-(6-(羟甲基)螺[3.3]庚烷-2-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(29f)(白色固体,470mg,产率86.95%)。Use 2-chloro-9-(6-(hydroxymethyl)spiro[3.3]heptan-2-yl)-7,9-dihydro-8H-purin-8-one 29e (516mg, 1.75mmol) with N , N-dimethylformamide (6mL) was dissolved, and dimethyl sulfate (0.17mL, 1.75mmol) and cesium carbonate (1.14g, 3.50mmol) were added under stirring at 0°C and reacted for 1h. TLC monitors until the reaction is over, the reaction solution is slowly added dropwise to ice water and stirred, ethyl acetate is added for extraction, and the organic layer is concentrated to obtain the title compound 2-chloro-9-(6-(hydroxymethyl)spiro[3.3]heptane -2-yl)-7-methyl-7,9-dihydro-8H-purin-8-one (29f) (white solid, 470 mg, yield 86.95%).
1H NMR(400MHz,DMSO-d6)δ8.33(s,1H),4.71–4.60(m,1H),4.10–3.96(m,2H),3.35(s,1H),3.33(s,3H),2.93–2.81(m,2H),2.44–2.36(m,1H),2.32–2.21(m,2H),2.20–2.13(m,1H),2.07–1.99(m,1H),1.89–1.78(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.33(s,1H), 4.71-4.60(m,1H), 4.10-3.96(m,2H), 3.35(s,1H), 3.33(s,3H), 2.93–2.81(m, 2H), 2.44–2.36(m, 1H), 2.32–2.21(m, 2H), 2.20–2.13(m, 1H), 2.07–1.99(m, 1H), 1.89–1.78(m ,2H).
LC-MS m/z(ESI)=309.10[M+1]。LC-MS m/z(ESI)=309.10[M+1].
第六步:The sixth step:
9-(6-(羟甲基)螺[3.3]庚烷-2-基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物29)9-(6-(Hydroxymethyl)spiro[3.3]heptan-2-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl) Amino)-7,9-dihydro-8H-purin-8-one (Compound 29)
9-(6-(hydroxymethyl)spiro[3.3]heptan-2-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purin-8-one9-(6-(hydroxymethyl)spiro[3.3]heptan-2-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro- 8H-purin-8-one
将2-氯-9-(6-(羟甲基)螺[3.3]庚烷-2-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮29f(100mg,0.32mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(97mg,0.65mmol)、碳酸铯(211mg,0.65mmol)、Brettphos G3 Pd(29mg,0.032mmol)加入干燥的反应瓶,氮气置换三次后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。TLC监测至反应结束,冷却至室温后用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化得到标题化合物9-(6-(羟甲基)螺[3.3]庚烷-2-基)-7-甲基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-7,9-二氢-8H-嘌呤-8-酮(化合物29)(浅粉色固体,70mg,产率51.28%)。The 2-chloro-9-(6-(hydroxymethyl)spiro[3.3]heptan-2-yl)-7-methyl-7,9-dihydro-8H-purin-8-one 29f (100mg, 0.32mmol), 6-methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (97mg, 0.65mmol), cesium carbonate (211mg, 0.65mmol), Brettphos G3 Pd (29mg, 0.032mmol) Add a dry reaction flask, replace with nitrogen three times, add dry 1,4-dioxane (1 mL), and react at 110° C. for 5 hours. The reaction was monitored by TLC until the end of the reaction. After cooling to room temperature, it was separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 9-(6-(hydroxymethyl)spiro[3.3] Heptan-2-yl)-7-methyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-7,9-dihydro-8H-purine- 8-ketone (compound 29) (light pink solid, 70 mg, yield 51.28%).
1H NMR(400MHz,DMSO-d6)δ8.22(s,1H),7.96(s,1H),7.14(s,1H),6.61(s,1H),4.61–4.51(m,1H),4.48(t,2H),4.43(t,1H),3.33–3.29(m,2H),3.23(s,3H),3.11(t,2H),2.87(t,1H),2.80(t,1H),2.29–2.17(m,2H),2.14–2.06(m,5H),1.85–1.76(m,2H),1.67–1.59(m,1H)。1H NMR(400MHz,DMSO-d6)δ8.22(s,1H),7.96(s,1H),7.14(s,1H),6.61(s,1H),4.61-4.51(m,1H), 4.48( t, 2H), 4.43 (t, 1H), 3.33--3.29 (m, 2H), 3.23 (s, 3H), 3.11 (t, 2H), 2.87 (t, 1H), 2.80 (t, 1H), 2.29 –2.17(m,2H), 2.14–2.06(m,5H), 1.85–1.76(m,2H), 1.67–1.59(m,1H).
LC-MS m/z(ESI)=422.20[M+1]。LC-MS m/z(ESI)=422.20[M+1].
实施例30Example 30
7-乙基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物30)7-Ethyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7, 9-dihydro-8H-purin-8-one (Compound 30)
7-ethyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one7-ethyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8 -one
第一步:first step:
2-氯-7-乙基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(30a)2-chloro-7-ethyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one (30a)
2-chloro-7-ethyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one2-chloro-7-ethyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one
将2-氯-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮1d(400mg,1.57mmol),溶解于N,N-二甲基甲酰胺(8mL),在0℃下加入碳酸铯(511mg,1.57mmol)和碘乙烷(293mg,1.88mmol),0℃搅拌1h后,随后加入10mL水,用乙酸乙酯萃取3次,有机相用无水硫酸钠干燥,浓缩,旋蒸除去有机溶剂,得到目标产物2-氯-7-乙基-9-(四氢-2H-吡喃-4-基(-7,9-二氢-8H-嘌呤-8-酮(30a)(白色固体,290mg,65.32%)。Dissolve 2-chloro-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one 1d (400mg, 1.57mmol) in N,N-di Methylformamide (8mL), add cesium carbonate (511mg, 1.57mmol) and iodoethane (293mg, 1.88mmol) at 0℃, stir for 1h at 0℃, then add 10mL of water and extract 3 times with ethyl acetate , The organic phase was dried with anhydrous sodium sulfate, concentrated, and the organic solvent was removed by rotary evaporation to obtain the target product 2-chloro-7-ethyl-9-(tetrahydro-2H-pyran-4-yl(-7,9- Dihydro-8H-purin-8-one (30a) (white solid, 290 mg, 65.32%).
1H NMR(400MHz,DMSO-d6)δ8.44(s,1H),4.50–4.41(m,1H),3.99–3.95(m,2H),3.89(q,2H),3.45(t,2H),2.46–2.41(m,2H),1.67–1.71(m,2H),1.25(t,3H)。1H NMR(400MHz,DMSO-d6)δ8.44(s,1H), 4.50-4.41(m,1H), 3.99-3.95(m,2H), 3.89(q,2H), 3.45(t,2H), 2.46–2.41(m,2H), 1.67–1.71(m,2H), 1.25(t,3H).
LC-MS m/z(ESI)=283.00[M+1]。LC-MS m/z(ESI)=283.00[M+1].
第二步:The second step:
7-乙基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物30)7-Ethyl-2-(((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7, 9-dihydro-8H-purin-8-one (Compound 30)
7-ethyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one7-ethyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8 -one
将2-氯-7-乙基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮30a(100mg,0.35mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(106mg,0.70mmol)、碳酸铯(230mg,0.70mmol)和Brettphos G3 Pd(32mg,0.035mmol)加入干燥的反应管,N 2置换三次,然后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。冷却至室温,使用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化,得到标题化合物7-乙基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物30)(浅粉色固体,44mg,产率31.46%)。 The 2-chloro-7-ethyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one 30a (100mg, 0.35mmol), 6- Methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (106mg, 0.70mmol), cesium carbonate (230mg, 0.70mmol) and Brettphos G3 Pd (32mg, 0.035mmol) were added to the dry reaction tube, Replace with N 2 three times, then add dry 1,4-dioxane (1 mL), and react at 110° C. for 5 h. Cooled to room temperature, and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 7-ethyl-2-(((6-methyl-2,3- Dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one (compound 30) (light pink Solid, 44 mg, yield 31.46%).
1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),8.02(s,1H),7.24(s,1H),6.60(s,1H),4.49(t,2H),4.41–4.31(m,1H),3.95(dd,2H),3.79(q,2H),3.40(t,2H),3.11(t,2H),2.58– 2.45(m,2H),2.12(s,3H),1.68-1.60(m,2H),1.21(t,3H)。1H NMR(400MHz,DMSO-d6)δ8.23(s,1H), 8.02(s,1H), 7.24(s,1H), 6.60(s,1H), 4.49(t,2H), 4.41--4.31( m, 1H), 3.95 (dd, 2H), 3.79 (q, 2H), 3.40 (t, 2H), 3.11 (t, 2H), 2.58-2.45 (m, 2H), 2.12 (s, 3H), 1.68 -1.60(m,2H),1.21(t,3H).
LC-MS m/z(ESI)=396.20[M+1]。LC-MS m/z(ESI)=396.20[M+1].
实施例31Example 31
7-异丙基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8–酮7-isopropyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7, 9-dihydro-8H-purin-8--one
7-isopropyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one7-isopropyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8 -one
第一步:first step:
2-氯-7-异丙基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(31a)2-Chloro-7-isopropyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one (31a)
2-chloro-7-isopropyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one2-chloro-7-isopropyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one
将2-氯-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮1d(520mg,2.04mmol),溶解于N,N-二甲基甲酰胺(10mL),在0℃下加入碳酸铯(665mg,2.04mmol)和2-碘丙烷(416mg,2.45mmol),0℃搅拌2h后,随后加入20mL水,用乙酸乙酯萃取3次,有机相用无水硫酸钠干燥,浓缩,旋蒸除去有机溶剂,得到目标产物2-氯-7-异丙基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(31a)(白色固体,465mg,76.74%)。Dissolve 2-chloro-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one 1d (520mg, 2.04mmol) in N,N-di Methylformamide (10mL), add cesium carbonate (665mg, 2.04mmol) and 2-iodopropane (416mg, 2.45mmol) at 0℃, stir for 2h at 0℃, then add 20mL of water, and extract 3 with ethyl acetate. Next, the organic phase was dried with anhydrous sodium sulfate, concentrated, and the organic solvent was removed by rotary evaporation to obtain the target product 2-chloro-7-isopropyl-9-(tetrahydro-2H-pyran-4-yl)-7, 9-Dihydro-8H-purin-8-one (31a) (white solid, 465 mg, 76.74%).
1H NMR(400MHz,DMSO-d6)δ8.52(s,1H),4.65–4.56(m,1H),4.49–4.40(m,1H),3.99–3.95(m,2H),3.44(t,2H),2.46–2.42(m,2H),1.70–1.67(m,2H),1.43(d,6H)。1H NMR(400MHz,DMSO-d6)δ8.52(s,1H), 4.65-4.56(m,1H), 4.49-4.40(m,1H), 3.99-3.95(m,2H), 3.44(t,2H) ), 2.46–2.42(m,2H), 1.70–1.67(m,2H), 1.43(d,6H).
LC-MS m/z(ESI)=297.10[M+1]。LC-MS m/z(ESI)=297.10[M+1].
第二步:The second step:
7-异丙基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8–酮(化合物31)7-isopropyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7, 9-dihydro-8H-purin-8-one (compound 31)
7-isopropyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one7-isopropyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8 -one
将2-氯-7-异丙基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮31a(100mg,0.37mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(101mg,0.74mmol)、碳酸铯(220mg,0.74mmol)和Brettphos G3 Pd(30mg,0.037mmol)加入干燥的反应管,N2置换三次,然后加入干燥的1,4-二氧六环(1ml),在110℃下反应5h。冷却至室温,使用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化,得到标题化合物7-异丙基-2-(((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8–酮(化合物31)(浅黄色固体,41mg,产率29.71%)。The 2-chloro-7-isopropyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one 31a (100mg, 0.37mmol), 6 -Methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (101mg, 0.74mmol), cesium carbonate (220mg, 0.74mmol) and Brettphos G3 Pd (30mg, 0.037mmol) were added to the dry reaction tube , N2 was replaced three times, and then dry 1,4-dioxane (1ml) was added and reacted at 110°C for 5h. Cool to room temperature, use silica gel column chromatography to separate and purify (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 7-isopropyl-2-(((6-methyl-2,3 -Dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one (compound 31) (shallow Yellow solid, 41 mg, yield 29.71%).
1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),8.13(s,1H),7.24(s,1H),6.60(s,1H),4.57–4.51(m,1H),4.49(t,2H),4.41–4.32(m,1H),3.95(dd,2H),3.40(t,2H),3.11(t,2H),2.58–2.45(m,2H),2.13(s,3H),1.66-1.60(m,2H),1.38(d,6H)。1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),8.13(s,1H),7.24(s,1H),6.60(s,1H),4.57-4.51(m,1H), 4.49( t, 2H), 4.41--4.32 (m, 1H), 3.95 (dd, 2H), 3.40 (t, 2H), 3.11 (t, 2H), 2.58 - 2.45 (m, 2H), 2.13 (s, 3H) ,1.66-1.60(m,2H),1.38(d,6H).
LC-MS m/z(ESI)=410.20[M+1]。LC-MS m/z(ESI)=410.20[M+1].
实施例32Example 32
7-环丙基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物32)7-Cyclopropyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7, 9-dihydro-8H-purin-8-one (Compound 32)
7-cyclopropyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one7-cyclopropyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8 -one
第一步:first step:
2-氯-7-环丙基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(32a)2-Chloro-7-cyclopropyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one (32a)
2-chloro-7-cyclopropyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one2-chloro-7-cyclopropyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one
将2-氯-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮1d(1g,3.9mmol)、环丙基硼酸(676mg,7.8mmol)、无水醋酸铜(715mg,3.9mmol)、碳酸钾(1.058g,7.8mmol)、1,10-菲啰啉)710mg,3.9mmol)溶解于1,2-二氯乙烷12mL中,敞口反应,加热至70℃搅拌6h。TLC监测至反应完全,浓缩反应液,残留物用硅胶柱色谱分离提纯)石油醚/乙酸乙酯(v/v)=3/1),得到标题化合物2-氯-7-环丙基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(34a)(白色固体,690mg,产率56%)。Combine 2-chloro-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one 1d (1g, 3.9mmol), cyclopropylboronic acid (676mg, 7.8mmol), anhydrous copper acetate (715mg, 3.9mmol), potassium carbonate (1.058g, 7.8mmol), 1,10-phenanthroline) 710mg, 3.9mmol) dissolved in 1,2-dichloroethane 12mL , Open reaction, heat to 70 ℃ and stir for 6h. TLC monitors until the reaction is complete, the reaction solution is concentrated, and the residue is separated and purified by silica gel column chromatography) petroleum ether/ethyl acetate (v/v) = 3/1) to obtain the title compound 2-chloro-7-cyclopropyl-9 -(Tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one (34a) (white solid, 690 mg, yield 56%).
1H NMR(400MHz CDCI3)δ8.15(s,1H),4.51-4.59(m,1H),4.12(dd,2H),3.49-3.55(m,2H),2.91-2.96(m,1H),2.67-2.77(m,2H),1.68-1.77(m,2H),1.13-1.26(m,2H),1.00-1.04(m,2H)。1H NMR (400MHz CDCI3) δ8.15 (s, 1H), 4.51-4.59 (m, 1H), 4.12 (dd, 2H), 3.49-3.55 (m, 2H), 2.91-2.96 (m, 1H), 2.67 -2.77 (m, 2H), 1.68-1.77 (m, 2H), 1.13-1.26 (m, 2H), 1.00-1.04 (m, 2H).
LC-MS m/z(ESI)=295.2[M+1]。LC-MS m/z(ESI)=295.2[M+1].
第二步:The second step:
7-环丙基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物32)7-Cyclopropyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7, 9-dihydro-8H-purin-8-one (Compound 32)
7-cyclopropyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one7-cyclopropyl-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8 -one
将2-氯-7-环丙基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮32a(100mg,0.34mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺(102mg,0.68mmol)、碳酸铯(222mg,0.68mmol)和Brettphos G3 Pd(32mg,0.034mmol)加入干燥的反应管,N 2置换三次,然后加入干燥的1,4-二氧六环(1mL),在110℃下反应5h。冷却至室温,使用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化,得到标题化合物7-环丙基-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物32)(浅黄色固体,28mg,产率20.25%)。 The 2-chloro-7-cyclopropyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one 32a (100mg, 0.34mmol), 6 -Methyl-2,3-dihydrobenzofuran-5-amine (102mg, 0.68mmol), cesium carbonate (222mg, 0.68mmol) and Brettphos G3 Pd (32mg, 0.034mmol) were added to the dry reaction tube, N 2 Replace three times, then add dry 1,4-dioxane (1 mL), and react at 110° C. for 5 h. Cooled to room temperature, and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 7-cyclopropyl-2-((6-methyl-2,3- Dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one (compound 32) (light yellow Solid, 28 mg, yield 20.25%).
1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),7.94(s,1H),7.23(s,1H),6.60(s,1H),4.48(t,2H),4.38–4.28(m,1H),3.94(dd,2H),3.39(t,2H),3.11(t,2H),2.90–2.84(m,1H),2.56–2.43(m,2H),2.12(s,3H),1.65-1.57(m,2H),0.99–0.92(m,2H),0.90–0.84(m,2H) 。1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),7.94(s,1H),7.23(s,1H),6.60(s,1H), 4.48(t,2H), 4.38-4.28( m, 1H), 3.94 (dd, 2H), 3.39 (t, 2H), 3.11 (t, 2H), 2.90-2.84 (m, 1H), 2.56-2.43 (m, 2H), 2.12 (s, 3H) ,1.65-1.57(m,2H),0.99–0.92(m,2H),0.90–0.84(m,2H).
LC-MS m/z(ESI)=408.20[M+1]。LC-MS m/z(ESI)=408.20[M+1].
实施例33Example 33
7-(甲基-d3)-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物33)7-(methyl-d3)-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl) -7,9-dihydro-8H-purin-8-one (Compound 33)
7-(methyl-d3)-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one7-(methyl-d3)-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H -purin-8-one
第一步:first step:
2-氯-7-(甲基-d3)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(33a)2-Chloro-7-(methyl-d3)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one (33a)
2-chloro-7-(methyl-d3)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one2-chloro-7-(methyl-d3)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one
将2-氯-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮1d(1.0g,3.93mmol)溶解于二甲基亚砜(20mL)中,室温下加入碳酸铯(2.03g,7.86mmol),然后在0℃加入氘代碘甲烷(0.45g,3.93mmol),室温反应2h。反应结束后加入5mL水,用乙酸乙酯萃取3次,有机相用无水硫酸钠干燥,浓缩,有固体析出,使用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=20:1))纯化,得到化合物2-氯-7-(甲基-d3)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(33a)(白色固体,0.36g,产率34.66%)。Dissolve 2-chloro-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one 1d (1.0g, 3.93mmol) in dimethyl sulfoxide (20mL), add cesium carbonate (2.03g, 7.86mmol) at room temperature, then add deuterated methyl iodide (0.45g, 3.93mmol) at 0°C, and react at room temperature for 2h. After the reaction, 5 mL of water was added and extracted with ethyl acetate for 3 times. The organic phase was dried with anhydrous sodium sulfate and concentrated. A solid precipitated out. It was separated and purified by silica gel column chromatography (dichloromethane/methanol (v/v=20:1) )) Purification to obtain the compound 2-chloro-7-(methyl-d3)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one ( 33a) (white solid, 0.36 g, yield 34.66%).
1HNMR(400MHz DMSO)δ8.36(s,1H),4.50-4.41(m,1H),3.99-3.95(m,2H),3.48-3.42(m,2H),2.47-2.38(m,2H),1.70-1.66(m,2H)。1HNMR(400MHz DMSO)δ8.36(s,1H), 4.50-4.41(m,1H), 3.99-3.95(m,2H), 3.48-3.42(m,2H), 2.47-2.38(m,2H), 1.70-1.66 (m, 2H).
LC-MS m/z(ESI)=272.10[M+1]。LC-MS m/z(ESI)=272.10[M+1].
第二步:The second step:
7-(甲基-d3)-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物33)7-(methyl-d3)-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl) -7,9-dihydro-8H-purin-8-one (Compound 33)
7-(methyl-d3)-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one7-(methyl-d3)-2-((6-methyl-2,3-dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H -purin-8-one
将2-氯-7-(甲基-d3)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮33a(100mg,0.37mmol)、6-甲基-2,3-二氢苯并呋喃-5-胺中间体1(110mg,0.74mmol)、碳酸铯(240mg,0.74mmol)和Brettphos G3 Pd(34mg,0.037mmol)加入干燥的反应管,N2置换三次,然后加入干燥的1,4-二氧六环(1ml),在110℃下反应5h。冷却至室温,使用硅胶柱色谱分离提纯(二氯甲烷/甲醇(v/v=40:1))纯化,得到标题化合物7-(甲基-d3)-2-((6-甲基-2,3-二氢苯并呋喃-5-基)氨基)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物33)(灰白色固体,50mg,产率35.34%)。The 2-chloro-7-(methyl-d3)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one 33a (100mg, 0.37mmol ), 6-methyl-2,3-dihydrobenzofuran-5-amine intermediate 1 (110mg, 0.74mmol), cesium carbonate (240mg, 0.74mmol) and Brettphos G3 Pd (34mg, 0.037mmol) were added to the dry Replace the reaction tube with N2 three times, then add dry 1,4-dioxane (1ml), and react at 110°C for 5h. Cooled to room temperature, and purified by silica gel column chromatography (dichloromethane/methanol (v/v=40:1)) to obtain the title compound 7-(methyl-d3)-2-((6-methyl-2 ,3-Dihydrobenzofuran-5-yl)amino)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one (Compound 33) (Off-white solid, 50 mg, yield 35.34%).
1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),7.95(s,1H),7.24(s,1H),6.60(s,1H),4.49(t,2H),4.41–4.32(m,1H),3.95(dd,2H),3.40(t,2H),3.11(t,2H),2.57–2.44(m,2H),2.12(s,3H),1.68–1.59(m,2H)。1H NMR (400MHz, DMSO-d6) δ 8.23 (s, 1H), 7.95 (s, 1H), 7.24 (s, 1H), 6.60 (s, 1H), 4.49 (t, 2H), 4.41-4.32 ( m, 1H), 3.95 (dd, 2H), 3.40 (t, 2H), 3.11 (t, 2H), 2.57-2.44 (m, 2H), 2.12 (s, 3H), 1.68-1.59 (m, 2H) .
LC-MS m/z(ESI)=385.20[M+1]。LC-MS m/z(ESI)=385.20[M+1].
实施例34Example 34
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物34)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydro-2H-pyran-4-yl)-7,9- Dihydro-8H-purin-8-one (Compound 34)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8 -one
化合物34合成方法与化合物1相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物34)(白色固体,15mg,产率10.55%)。The synthesis method of compound 34 is the same as that of compound 1, and the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydro-2H- Pyran-4-yl)-7,9-dihydro-8H-purin-8-one (Compound 34) (white solid, 15 mg, yield 10.55%).
1H NMR(400MHz MeOD)δ7.86(s,1H),7.41(s,1H),6.99(s,1H),4.67(t,2H),4.51-4.59(m,1H),4.03-4.07(m,2H),3.52(m,2H),3.38(s,3H),3.25-3.30(m,2H),2.55-2.66(m,2H),1.75-1.78(m,2H)1H NMR (400MHz MeOD) δ 7.86 (s, 1H), 7.41 (s, 1H), 6.99 (s, 1H), 4.67 (t, 2H), 4.51-4.59 (m, 1H), 4.03-4.07 (m ,2H),3.52(m,2H),3.38(s,3H),3.25-3.30(m,2H),2.55-2.66(m,2H),1.75-1.78(m,2H)
LC-MS m/z(ESI)=402.20[M+1]。LC-MS m/z(ESI)=402.20[M+1].
实施例35Example 35
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-((反-4-甲氧基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物35)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-((trans-4-methoxycyclohexyl)-7-methyl-7,9-di Hydrogen-8H-purin-8-one (Compound 35)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-((trans-4-methoxy cyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-((trans-4-methoxycyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one
参考化合物2的制备方法,制备得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-((反-4-甲氧基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8–酮(化合物35)(灰白色固体,65mg,产率44.88%)。Refer to the preparation method of compound 2, and prepare the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-((trans-4-methoxycyclohexyl) -7-Methyl-7,9-dihydro-8H-purin-8-one (Compound 35) (off-white solid, 65 mg, yield 44.88%).
1H NMR(400MHz,DMSO-d6)δ8.28(s,1H),8.00(s,1H),7.53(s,1H),6.91(s,1H),4.57(t,2H),4.16-4.08(m,1H),3.27(s,3H),3.25(s,3H),3.18(t,2H),3.09-3.02(m,1H),2.34-2.22(m,2H),2.08(d,2H),1.71(d,2H),1.26-1.20(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.28(s,1H),8.00(s,1H),7.53(s,1H),6.91(s,1H),4.57(t,2H),4.16-4.08( m, 1H), 3.27 (s, 3H), 3.25 (s, 3H), 3.18 (t, 2H), 3.09-3.02 (m, 1H), 2.34-2.22 (m, 2H), 2.08 (d, 2H) , 1.71 (d, 2H), 1.26-1.20 (m, 2H).
LC-MS m/z(ESI)=430.10[M+1]。LC-MS m/z(ESI)=430.10[M+1].
实施例36Example 36
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(反-4-羟基-4-甲基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物36)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(trans-4-hydroxy-4-methylcyclohexyl)-7-methyl-7,9 -Dihydro-8H-purin-8-one (Compound 36)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(trans-4-hydroxy-4-methylcyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(trans-4-hydroxy-4-methylcyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8 -one
采用化合物3的制备方法,制备得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-((反-4-羟基-4-甲基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物36)(浅黄色固体,58mg,产率40.04%)。Using the preparation method of compound 3, the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-((trans-4-hydroxy-4-methyl) was prepared. Cyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one (Compound 36) (light yellow solid, 58 mg, yield 40.04%).
1H NMR(400MHz,DMSO-d6)δ8.38(s,1H),8.01(s,1H),7.34(s,1H),6.88(s,1H),4.56(t,2H),4.38(s,1H),4.08-4.01(m,1H),3.27(s,3H),3.15(t,2H),2.32-2.19(m,2H),1.59-1.39(m,6H),0.98(s,3H)。1H NMR (400MHz, DMSO-d6) δ 8.38 (s, 1H), 8.01 (s, 1H), 7.34 (s, 1H), 6.88 (s, 1H), 4.56 (t, 2H), 4.38 (s, 1H), 4.08-4.01 (m, 1H), 3.27 (s, 3H), 3.15 (t, 2H), 2.32-2.19 (m, 2H), 1.59-1.39 (m, 6H), 0.98 (s, 3H) .
LC-MS m/z(ESI)=430.10[M+1]。LC-MS m/z(ESI)=430.10[M+1].
实施例37Example 37
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(1-甲基哌啶-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物37)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(1-methylpiperidin-4-yl)-7,9-bis Hydrogen-8H-purin-8-one (Compound 37)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(1-methylpiperidin-4-yl)-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(1-methylpiperidin-4-yl)-7,9-dihydro-8H-purin-8-one
化合物37合成方法与化合物4相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(1-甲基哌啶-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物37)(浅黄色固体,36mg,产率44.45%)。The synthesis method of compound 37 is the same as that of compound 4 to obtain the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(1-methylpiper (Pyridin-4-yl)-7,9-dihydro-8H-purin-8-one (Compound 37) (pale yellow solid, 36 mg, yield 44.45%).
1H NMR(400MHz,DMSO-d6)δ8.25(s,1H),7.99(s,1H),7.59(s,1H),6.89(s,1H),4.57(t,2H),4.13-4.05(m,1H),3.27(s,3H),3.18(t,2H),2.86(d,2H),2.58-2.47(m,2H),2.19(s,3H),1.95(t,2H),1.62(d,2H)。1H NMR(400MHz,DMSO-d6)δ8.25(s,1H),7.99(s,1H),7.59(s,1H),6.89(s,1H),4.57(t,2H),4.13-4.05( m, 1H), 3.27(s, 3H), 3.18(t, 2H), 2.86(d, 2H), 2.58-2.47(m, 2H), 2.19(s, 3H), 1.95(t, 2H), 1.62 (d, 2H).
LC-MS m/z(ESI)=415.10[M+1]。LC-MS m/z(ESI)=415.10[M+1].
实施例38Example 38
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-环己基-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物38)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-cyclohexyl-7-methyl-7,9-dihydro-8H-purin-8-one( Compound 38)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-cyclohexyl-7-methyl-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-cyclohexyl-7-methyl-7,9-dihydro-8H-purin-8-one
化合物38合成方法与化合物5相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-环己基-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物38)(浅黄色固体,40mg,产率26.68%)。The synthesis method of compound 38 is the same as that of compound 5 to obtain the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-cyclohexyl-7-methyl-7,9 -Dihydro-8H-purin-8-one (Compound 38) (light yellow solid, 40 mg, yield 26.68%).
1H NMR(400MHz,DMSO-d6)δ8.24(s,1H),8.00(s,1H),7.57(s,1H),6.90(s,1H),4.57(t,J=8.6Hz,2H),4.15-4.07(m,1H),3.27(s,3H),3.17(t,2H),2.26-2.16(m,2H),1.80(d,2H),1.68(d,3H),1.36-1.27(m,2H),1.15-1.05(m,1H)。1H NMR(400MHz,DMSO-d6)δ8.24(s,1H),8.00(s,1H),7.57(s,1H),6.90(s,1H),4.57(t,J=8.6Hz,2H) ,4.15-4.07(m,1H),3.27(s,3H),3.17(t,2H),2.26-2.16(m,2H),1.80(d,2H),1.68(d,3H),1.36-1.27 (m, 2H), 1.15-1.05 (m, 1H).
LC-MS m/z(ESI)=400.10[M+1]。LC-MS m/z(ESI)=400.10[M+1].
实施例39Example 39
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(四氢-2H-吡喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(化合物39)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydro-2H-pyran-3-yl)-7,9- Dihydro-8H-purin-8-one (Compound 39)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydro-2H-pyran-3- yl)-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydro-2H-pyran-3- yl)-7,9-dihydro-8H-purin-8 -one
化合物39合成方法与化合物6相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(四氢-2H-吡喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(化合物39)(浅黄色固体,49mg,产率32.76%)。The synthesis method of compound 39 is the same as that of compound 6, and the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydro-2H- Pyran-3-yl)-7,9-dihydro-8H-purin-8-one (Compound 39) (pale yellow solid, 49 mg, yield 32.76%).
1H NMR(400MHz,DMSO-d6)δ8.35(s,1H),8.01(s,1H),7.51(s,1H),6.91(s,1H),4.57(t,2H),4.26-4.18(m,1H),3.95-3.82(m,2H),3.77-3.73(m,1H),3.27(s,3H),3.17(t,3H),2.48-2.40(m,1H),1.85(d,1H),1.77-1.61(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.35(s,1H),8.01(s,1H),7.51(s,1H),6.91(s,1H),4.57(t,2H),4.26-4.18( m, 1H), 3.95-3.82 (m, 2H), 3.77-3.73 (m, 1H), 3.27 (s, 3H), 3.17 (t, 3H), 2.48-2.40 (m, 1H), 1.85 (d, 1H), 1.77-1.61 (m, 2H).
LC-MS m/z(ESI)=402.10[M+1]。LC-MS m/z(ESI)=402.10[M+1].
实施例40Example 40
(R)-2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(化合物40)(R)-2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydrofuran-3-yl)-7,9-dihydro -8H-purin-8-one (Compound 40)
(R)-2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydro furan-3-yl)-7,9-dihydro-8H-purin-8-one(R)-2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin- 8-one
化合物40合成方法与化合物7相同,得到标题化合物(R)-2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(化合物40)(浅黄色固体,65mg,产率42.68%)。The synthesis method of compound 40 is the same as that of compound 7, to obtain the title compound (R)-2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydrofuran) -3-yl)-7,9-dihydro-8H-purin-8-one (Compound 40) (pale yellow solid, 65 mg, yield 42.68%).
1H NMR(400MHz,DMSO-d6)δ8.32(s,1H),8.03(s,1H),7.51(s,1H),6.90(s,1H),4.94-4.85(m,1H),4.57(t,2H),3.95-3.90(m,2H),3.83-3.76(m,2H),3.28(s,3H),3.20-3.14(m,2H),2.40-2.33(m,1H),2.22-2.12(m,1H)。1H NMR(400MHz,DMSO-d6)δ8.32(s,1H),8.03(s,1H),7.51(s,1H),6.90(s,1H),4.94-4.85(m,1H),4.57( t,2H),3.95-3.90(m,2H),3.83-3.76(m,2H), 3.28(s,3H), 3.20-3.14(m,2H), 2.40-2.33(m,1H),2.22- 2.12 (m, 1H).
LC-MS m/z(ESI)=388.10[M+1]。LC-MS m/z(ESI)=388.10[M+1].
实施例41Example 41
(S)-2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(化合物41)(S)-2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydrofuran-3-yl)-7,9-dihydro -8H-purin-8-one (Compound 41)
(S)-2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8-one(S)-2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydrofuran-3-yl)-7,9-dihydro-8H-purin-8 -one
化合物41合成方法与化合物8相同,得到标题化合物(S)-2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(四氢呋喃-3-基)-7,9-二氢-8H-嘌呤-8-酮(化合物41)(浅黄色固体,67mg,产率44.00%)。The synthesis method of compound 41 is the same as that of compound 8, to obtain the title compound (S)-2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydrofuran) -3-yl)-7,9-dihydro-8H-purin-8-one (Compound 41) (light yellow solid, 67 mg, yield 44.00%).
1H NMR(400MHz,DMSO-d6)δ8.32(s,1H),8.03(s,1H),7.52(s,1H),6.90(s,1H),4.93-4.86(m,1H),4.57(t,2H),3.95-3.88(m,2H),3.83-3.76(m,2H),3.28(s,3H),3.17(t,2H),2.40-2.33(m,1H),2.22-2.12(m,1H)。1H NMR(400MHz,DMSO-d6)δ8.32(s,1H),8.03(s,1H),7.52(s,1H),6.90(s,1H),4.93-4.86(m,1H),4.57( t,2H),3.95-3.88(m,2H),3.83-3.76(m,2H), 3.28(s,3H), 3.17(t,2H), 2.40-2.33(m,1H),2.22-2.12( m,1H).
LC-MS m/z(ESI)=388.10[M+1]。LC-MS m/z(ESI)=388.10[M+1].
实施例42Example 42
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(((四氢呋喃-3-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(化合物42)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(((tetrahydrofuran-3-yl)methyl)-7,9-bis Hydrogen-8H-purin-8-one (Compound 42)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-((tetrahydrofuran-3-yl)methyl)-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-((tetrahydrofuran-3-yl)methyl)-7,9-dihydro-8H-purin-8- one
化合物42合成方法与化合物9相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(((四氢呋喃-3-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(化合物42)(灰白色固体,48mg,产率32.10%)。The synthesis method of compound 42 is the same as that of compound 9, and the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(((tetrahydrofuran-3 -Yl)methyl)-7,9-dihydro-8H-purin-8-one (Compound 42) (off-white solid, 48 mg, yield 32.10%).
1H NMR(400MHz,DMSO-d6)δ8.34(s,1H),8.01(s,1H),7.50(s,1H),6.90(s,1H),4.57(t,2H),3.78-3.73(m,1H),3.71(d,2H),3.65-3.60(m,2H),3.52(dd,1H),3.29(s,3H),3.17(t,2H),2.78-2.68(m,1H),1.94-1.86(m,1H),1.68-1.60(m,1H)。1H NMR(400MHz,DMSO-d6)δ8.34(s,1H),8.01(s,1H),7.50(s,1H),6.90(s,1H),4.57(t,2H),3.78-3.73( m, 1H), 3.71 (d, 2H), 3.65-3.60 (m, 2H), 3.52 (dd, 1H), 3.29 (s, 3H), 3.17 (t, 2H), 2.78-2.68 (m, 1H) ,1.94-1.86(m,1H),1.68-1.60(m,1H).
LC-MS m/z(ESI)=402.10[M+1]。LC-MS m/z(ESI)=402.10[M+1].
实施例43Example 43
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(哌啶-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物43)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(piperidin-4-yl)-7,9-dihydro-8H- Purine-8-one (Compound 43)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(piperidin-4-yl)-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(piperidin-4-yl)-7,9-dihydro-8H-purin-8-one
化合物43合成方法与化合物10相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(哌啶-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物43)(白色固体,30mg,产率13.76%)。The synthesis method of compound 43 is the same as that of compound 10 to obtain the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(piperidine-4- Yl)-7,9-dihydro-8H-purin-8-one (Compound 43) (white solid, 30 mg, yield 13.76%).
1H NMR(400MHz,DMSO-d6)δ8.24(s,1H),7.99(s,1H),7.59(s,1H),6.90(s,1H),4.57(t,2H),4.18(tt,1H),3.27(s,3H),3.19(t,2H),3.04(d,2H),2.55-2.48(m,2H),2.38-2.28(m,2H),1.61(d,2H)。1H NMR(400MHz,DMSO-d6)δ8.24(s,1H),7.99(s,1H),7.59(s,1H),6.90(s,1H),4.57(t,2H),4.18(tt, 1H), 3.27 (s, 3H), 3.19 (t, 2H), 3.04 (d, 2H), 2.55-2.48 (m, 2H), 2.38-2.28 (m, 2H), 1.61 (d, 2H).
LC-MS m/z(ESI)=401.10[M+1]。LC-MS m/z(ESI)=401.10[M+1].
实施例44Example 44
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-((顺-4-羟基-4-甲基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物44)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-((cis-4-hydroxy-4-methylcyclohexyl)-7-methyl-7, 9-dihydro-8H-purin-8-one (Compound 44)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(cis-4-hydroxy-4-methylcyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(cis-4-hydroxy-4-methylcyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8 -one
化合物44合成方法与化合物11相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-((顺-4-羟基-4-甲基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物44)(灰白色固体,23mg,产率19.84%)。The synthetic method of compound 44 is the same as that of compound 11 to obtain the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-((cis-4-hydroxy-4-methyl) Cyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one (Compound 44) (off-white solid, 23 mg, yield 19.84%).
1H NMR(400MHz,DMSO-d6)δ8.09(s,1H),8.00(s,1H),7.68(s,1H),6.89(s,1H),4.56(t,2H),4.14-4.07(m,1H),4.05(s,1H),3.28(s,3H),3.20(t,2H),2.70-2.59(m,2H),1.66(d,2H),1.46-1.37(m,4H),1.15(s,3H)。1H NMR(400MHz,DMSO-d6)δ8.09(s,1H),8.00(s,1H),7.68(s,1H),6.89(s,1H),4.56(t,2H),4.14-4.07( m, 1H), 4.05 (s, 1H), 3.28 (s, 3H), 3.20 (t, 2H), 2.70-2.59 (m, 2H), 1.66 (d, 2H), 1.46-1.37 (m, 4H) , 1.15 (s, 3H).
LC-MS m/z(ESI)=430.10[M+1]。LC-MS m/z(ESI)=430.10[M+1].
实施例45Example 45
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(顺-4-甲氧基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物45)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(cis-4-methoxycyclohexyl)-7-methyl-7,9-dihydro -8H-purin-8-one (Compound 45)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(cis-4-methoxy cyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(cis-4-methoxycyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one
化合物45合成方法与化合物12相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(顺-4-甲氧基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物45)(浅黄色固体,33mg,产率22.78%)。The synthesis method of compound 45 is the same as that of compound 12 to obtain the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(cis-4-methoxycyclohexyl) -7-Methyl-7,9-dihydro-8H-purin-8-one (Compound 45) (pale yellow solid, 33 mg, yield 22.78%).
1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),7.99(s,1H),7.51(s,1H),6.89(s,1H),4.57(t,2H),4.19-4.10(m,1H),3.40(s,1H),3.27(s,3H),3.25-3.15(m,5H),2.49-2.40(m,2H),1.98(d,2H),1.50-1.40(m,4H)。1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),7.99(s,1H),7.51(s,1H),6.89(s,1H),4.57(t,2H),4.19-4.10( m,1H), 3.40 (s, 1H), 3.27 (s, 3H), 3.25-3.15 (m, 5H), 2.49-2.40 (m, 2H), 1.98 (d, 2H), 1.50-1.40 (m, 4H).
LC-MS m/z(ESI)=430.20[M+1]。LC-MS m/z(ESI)=430.20[M+1].
实施例46Example 46
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(((四氢呋喃-2-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(化合物46)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(((tetrahydrofuran-2-yl)methyl)-7,9-bis Hydrogen-8H-purin-8-one (Compound 46)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-((tetrahydrofuran-2-yl)methyl)-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-((tetrahydrofuran-2-yl)methyl)-7,9-dihydro-8H-purin-8- one
化合物46合成方法与化合物14相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(((四氢呋喃-2-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(化合物46)(棕色固体,29mg,产率38.78%)。The synthesis method of compound 46 is the same as that of compound 14, and the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(((tetrahydrofuran-2 -Yl)methyl)-7,9-dihydro-8H-purin-8-one (Compound 46) (brown solid, 29 mg, yield 38.78%).
1H NMR(400MHz,DMSO-d6)δ8.32(s,1H),8.00(s,1H),7.51(s,1H),6.90(s,1H),4.57(t,2H),4.28-4.22(m,1H),3.81-3.73(m,2H),3.68-3.58(m,2H),3.29(s,3H),3.17(t,2H),1.95-1.75(m,3H),1.71-1.61(m,1H)。1H NMR(400MHz,DMSO-d6)δ8.32(s,1H),8.00(s,1H),7.51(s,1H),6.90(s,1H),4.57(t,2H),4.28-4.22( m,1H),3.81-3.73(m,2H),3.68-3.58(m,2H), 3.29(s,3H), 3.17(t,2H),1.95-1.75(m,3H),1.71-1.61( m,1H).
LC-MS m/z(ESI)=402.10[M+1]。LC-MS m/z(ESI)=402.10[M+1].
实施例47Example 47
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-((四氢-2H-吡喃-4-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(化合物47)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-((tetrahydro-2H-pyran-4-yl)methyl)- 7,9-dihydro-8H-purin-8-one (Compound 47)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-((tetrahydro-2H-pyran-4-yl)methyl)-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-((tetrahydro-2H-pyran-4-yl)methyl)-7,9-dihydro-8H- purin-8-one
化合物47合成方法与化合物15相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-((四氢-2H-吡喃-4-基)甲基)-7,9-二氢-8H-嘌呤-8-酮(化合物47)(白色固体,48mg,产率32.63%)。The synthesis method of compound 47 is the same as that of compound 15, and the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-((tetrahydro-2H -Pyran-4-yl)methyl)-7,9-dihydro-8H-purin-8-one (compound 47) (white solid, 48 mg, yield 32.63%).
1H NMR(400MHz,DMSO-d6)δ8.31(s,1H),8.00(s,1H),7.52(s,1H),6.90(s,1H),4.57(t,2H),3.85-3.80(m,2H),3.61(d,2H),3.29(s,3H),3.27-3.14(m,4H),2.12-2.03(m,1H),1.48(d,2H),1.28-1.18(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.31(s,1H),8.00(s,1H),7.52(s,1H),6.90(s,1H),4.57(t,2H),3.85-3.80( m, 2H), 3.61 (d, 2H), 3.29 (s, 3H), 3.27-3.14 (m, 4H), 2.12-2.03 (m, 1H), 1.48 (d, 2H), 1.28-1.18 (m, 2H).
LC-MS m/z(ESI)=416.10[M+1]。LC-MS m/z(ESI)=416.10[M+1].
实施例48Example 48
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(反-3-羟基环丁基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物48)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(trans-3-hydroxycyclobutyl)-7-methyl-7,9-dihydro- 8H-purin-8-one (Compound 48)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(trans-3-hydroxycyclobutyl)-7-methyl-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(trans-3-hydroxycyclobutyl)-7-methyl-7,9-dihydro-8H-purin-8-one
化合物48合成方法与化合物16相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(反-3-羟基环丁基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物48)(白色固体,40mg,产率25.43%)。The synthesis method of compound 48 is the same as that of compound 16, to obtain the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(trans-3-hydroxycyclobutyl)- 7-Methyl-7,9-dihydro-8H-purin-8-one (Compound 48) (white solid, 40 mg, yield 25.43%).
1H NMR(400MHz,DMSO-d6)δ8.34(s,1H),8.02(s,1H),7.50(s,1H),6.91(s,1H),5.09(d,1H),5.03-4.95(m,1H),4.57(t,2H),4.39-4.33(m,1H),3.26(s,3H),3.17(t,2H),3.03-2.94(m,2H),2.20-2.14(m,2H)。1H NMR (400MHz, DMSO-d6) δ 8.34 (s, 1H), 8.02 (s, 1H), 7.50 (s, 1H), 6.91 (s, 1H), 5.09 (d, 1H), 5.03-4.95 ( m, 1H), 4.57 (t, 2H), 4.39-4.33 (m, 1H), 3.26 (s, 3H), 3.17 (t, 2H), 3.03-2.94 (m, 2H), 2.20-2.14 (m, 2H).
LC-MS m/z(ESI)=388.10[M+1]。LC-MS m/z(ESI)=388.10[M+1].
实施例49Example 49
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(6-(羟甲基)四氢-2H-吡喃-3-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物49)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(6-(hydroxymethyl)tetrahydro-2H-pyran-3-yl)-7- Methyl-7,9-dihydro-8H-purin-8-one (Compound 49)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(6-(hydroxymethyl)tetrahydro-2H-pyran-3-yl)-7-methyl-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(6-(hydroxymethyl)tetrahydro-2H-pyran-3-yl)-7-methyl-7,9-dihydro- 8H-purin-8-one
化合物49合成方法与化合物17相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(6-(羟甲基)四氢-2H-吡喃-3-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物49)(白色固体,25mg,产率20.73%)。The synthesis method of compound 49 is the same as that of compound 17, and the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(6-(hydroxymethyl)tetrahydro- 2H-pyran-3-yl)-7-methyl-7,9-dihydro-8H-purin-8-one (compound 49) (white solid, 25 mg, yield 20.73%).
1H NMR(400MHz,DMSO-d6)δ8.38(s,1H),8.02(s,1H),7.39(s,1H),6.91(s,1H),4.58(t,2H),4.53(t,1H),4.24-4.16(m,1H),4.08(t,1H),3.69-3.61(m,1H),3.58-3.52(m,1H),3.44(dd,1H),3.38-3.31(m,1H),3.27(s,3H),3.19(t,2H),2.56-2.44(m,1H),1.84-1.78(m,1H),1.69-1.60(m,2H)。1H NMR (400MHz, DMSO-d6) δ 8.38 (s, 1H), 8.02 (s, 1H), 7.39 (s, 1H), 6.91 (s, 1H), 4.58 (t, 2H), 4.53 (t, 1H), 4.24-4.16 (m, 1H), 4.08 (t, 1H), 3.69-3.61 (m, 1H), 3.58-3.52 (m, 1H), 3.44 (dd, 1H), 3.38-3.31 (m, 1H), 3.27 (s, 3H), 3.19 (t, 2H), 2.56-2.44 (m, 1H), 1.84-1.78 (m, 1H), 1.69-1.60 (m, 2H).
LC-MS m/z(ESI)=432.20[M+1]。LC-MS m/z(ESI)=432.20[M+1].
实施例50Example 50
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(3-羟基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物50)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(3-hydroxycyclohexyl)-7-methyl-7,9-dihydro-8H-purine -8-one (Compound 50)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(3-hydroxycyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(3-hydroxycyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one
化合物50合成方法与化合物18相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(3-羟基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物50)(白色固体,30mg,产率18.80%)。The synthesis method of compound 50 is the same as that of compound 18 to obtain the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(3-hydroxycyclohexyl)-7-methyl Cyclo-7,9-dihydro-8H-purin-8-one (Compound 50) (white solid, 30 mg, yield 18.80%).
1H NMR(400MHz,DMSO-d6)δ8.24(s,1H),8.01(s,1H),7.58(s,1H),6.90(s,1H),4.79(d,1H),4.57(t,2H),4.18-4.10(m,1H),3.55-3.43(m,1H),3.27(s,3H),3.25-3.14(m,2H),2.21-2.02(m,2H),1.93-1.79(m,2H),1.79-1.71(m,1H),1.69-1.57(m,1H),1.35-1.26(m,1H),1.12-1.00(m,1H)。1H NMR (400MHz, DMSO-d6) δ 8.24 (s, 1H), 8.01 (s, 1H), 7.58 (s, 1H), 6.90 (s, 1H), 4.79 (d, 1H), 4.57 (t, 2H), 4.18-4.10 (m, 1H), 3.55-3.43 (m, 1H), 3.27 (s, 3H), 3.25-3.14 (m, 2H), 2.21-2.02 (m, 2H), 1.93-1.79 ( m, 2H), 1.79-1.71 (m, 1H), 1.69-1.57 (m, 1H), 1.35-1.26 (m, 1H), 1.12-1.00 (m, 1H).
LC-MS m/z(ESI)=416.10[M+1]。LC-MS m/z(ESI)=416.10[M+1].
实施例51Example 51
反-4-(2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-8-氧代-7,8-二氢-9H-嘌呤-9-yl)环己烷-1-腈(化合物51)Trans-4-(2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-8-oxo-7,8-dihydro-9H-purine -9-yl)cyclohexane-1-nitrile (Compound 51)
trans-4-(2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-8-oxo-7,8-dihydro-9H-purin-9-yl)cyclohexane-1-carbonitriletrans-4-(2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-8-oxo-7,8-dihydro-9H-purin-9-yl)cyclohexane- 1-carbonitrile
化合物51合成方法与化合物19相同,得到标题化合物反-4-(2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-8-氧代-7,8-二氢-9H-嘌呤-9-yl)环己烷-1-腈化合物51(白色固体,60mg,产率41.20%)。The synthesis method of compound 51 is the same as that of compound 19 to obtain the title compound trans-4-(2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-8-oxy (Dihydro-7,8-dihydro-9H-purine-9-yl)cyclohexane-1-carbonitrile compound 51 (white solid, 60 mg, yield 41.20%).
1H NMR(400MHz,DMSO-d6)δ8.25(s,1H),8.00(s,1H),7.54(s,1H),6.92(s,1H),4.58(t,2H),4.22-4.14(m,1H),3.27(s,3H),3.18(t,2H),2.60-2.52(m,1H),2.31-2.20(m,2H),2.15(d,2H),1.77(d,2H),1.73-1.61(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.25(s,1H),8.00(s,1H),7.54(s,1H),6.92(s,1H),4.58(t,2H),4.22-4.14( m, 1H), 3.27 (s, 3H), 3.18 (t, 2H), 2.60-2.52 (m, 1H), 2.31-2.20 (m, 2H), 2.15 (d, 2H), 1.77 (d, 2H) ,1.73-1.61(m,2H).
LC-MS m/z(ESI)=425.20[M+1]。LC-MS m/z(ESI)=425.20[M+1].
实施例52Example 52
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(2-异丙基-4-甲基吡啶-3-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物52)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(2-isopropyl-4-methylpyridin-3-yl)-7-methyl- 7,9-dihydro-8H-purin-8-one (Compound 52)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(2-isopropyl-4-methyl pyridin-3-yl)- 7-methyl-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(2-isopropyl-4-methylpyridin-3-yl)- 7-methyl-7,9-dihydro-8H- purin-8-one
化合物52合成方法与化合物20相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(2-异丙基-4-甲基吡啶-3-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物52)(白色固体,20mg,产率17.62%)。The synthesis method of compound 52 is the same as that of compound 20 to obtain the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(2-isopropyl-4-methyl Pyridin-3-yl)-7-methyl-7,9-dihydro-8H-purin-8-one (Compound 52) (white solid, 20 mg, yield 17.62%).
1H NMR(400MHz,DMSO-d6)δ8.54(d,1H),8.50(s,1H),8.13(s,1H),7.32(d,2H),6.85(s,1H),4.55(t,2H),3.40(s,3H),3.11(t,2H),2.83-2.75(m,1H),2.05(s,3H),1.15-1.08(m,6H)。1H NMR(400MHz,DMSO-d6)δ8.54(d,1H),8.50(s,1H),8.13(s,1H),7.32(d,2H),6.85(s,1H),4.55(t, 2H), 3.40 (s, 3H), 3.11 (t, 2H), 2.83-2.75 (m, 1H), 2.05 (s, 3H), 1.15-1.08 (m, 6H).
LC-MS m/z(ESI)=451.10[M+1]。LC-MS m/z(ESI)=451.10[M+1].
实施例53Example 53
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(4,4-二氟环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物53)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(4,4-difluorocyclohexyl)-7-methyl-7,9-dihydro- 8H-purine-8-one (Compound 53)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(4,4-difluorocyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(4,4-difluorocyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one
化合物53合成方法与化合物22相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(4,4-二氟环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物53)(浅黄色固体,25mg,产率17.36%)。The synthesis method of compound 53 is the same as that of compound 22 to obtain the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(4,4-difluorocyclohexyl)- 7-Methyl-7,9-dihydro-8H-purin-8-one (Compound 53) (light yellow solid, 25 mg, yield 17.36%).
1H NMR(400MHz,DMSO-d6)δ8.22(s,1H),8.04(s,1H),7.56(s,1H),6.89(s,1H),4.56(t,2H),4.37-4.29(m,1H),3.29(s,3H),3.17(t,2H),2.56-2.43(m,2H),2.10-1.95(m,4H),1.77(d,2H)。1H NMR(400MHz,DMSO-d6)δ8.22(s,1H), 8.04(s,1H), 7.56(s,1H), 6.89(s,1H), 4.56(t,2H), 4.37-4.29( m, 1H), 3.29 (s, 3H), 3.17 (t, 2H), 2.56-2.43 (m, 2H), 2.10-1.95 (m, 4H), 1.77 (d, 2H).
LC-MS m/z(ESI)=436.10[M+1]。LC-MS m/z(ESI)=436.10[M+1].
实施例54Example 54
4-(2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-8-氧代-7,8-二氢-9H-嘌呤-9-基)双环[2.2.2]辛烷-1-腈(化合物54)4-(2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-8-oxo-7,8-dihydro-9H-purine-9 -Yl)bicyclo[2.2.2]octane-1-carbonitrile (compound 54)
4-(2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-8-oxo-7,8-dihydro-9H-purin-9-yl)bicyclo[2.2.2]octane-1-carbonitrile4-(2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-8-oxo-7,8-dihydro-9H-purin-9-yl)bicyclo[2.2. 2]octane-1-carbonitrile
化合物54合成方法与化合物23相同,得到标题化合物4-(2-(((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-8-氧代-7,8-二氢-9H-嘌呤-9-基)双环[2.2.2]辛烷-1-腈(化合物54)(白色固体,60mg,产率42.28%)。The synthetic method of compound 54 is the same as that of compound 23 to obtain the title compound 4-(2-(((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-8-oxo -7,8-Dihydro-9H-purin-9-yl)bicyclo[2.2.2]octane-1-carbonitrile (Compound 54) (white solid, 60 mg, yield 42.28%).
1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),8.00(s,1H),7.51(s,1H),6.91(s,1H), 4.58(t,2H),3.22(s,3H),3.18(t,2H),2.44-2.39(m,6H),2.013-1.98(m,6H)。1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),8.00(s,1H),7.51(s,1H),6.91(s,1H), 4.58(t,2H),3.22(s, 3H), 3.18 (t, 2H), 2.44-2.39 (m, 6H), 2.013-1.98 (m, 6H).
LC-MS m/z(ESI)=451.10[M+1]。LC-MS m/z(ESI)=451.10[M+1].
实施例55Example 55
9-(8-氧杂双环[3.2.1]辛基-3-基)-2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物55)9-(8-oxabicyclo[3.2.1]octyl-3-yl)-2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl -7,9-dihydro-8H-purin-8-one (Compound 55)
9-(8-oxabicyclo[3.2.1]octan-3-yl)-2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-7,9-dihydro-8H-purin-8-one9-(8-oxabicyclo[3.2.1]octan-3-yl)-2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-7,9-dihydro-8H -purin-8-one
化合物55合成方法与化合物24相同,得到标题化合物9-(8-氧杂双环[3.2.1]辛基-3-基)-2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物55)(浅黄色固体,28mg,产率19.29%)。The synthesis method of compound 55 is the same as that of compound 24 to obtain the title compound 9-(8-oxabicyclo[3.2.1]octyl-3-yl)-2-((6-chloro-2,3-dihydrobenzofuran) -5-yl)amino)-7-methyl-7,9-dihydro-8H-purin-8-one (compound 55) (pale yellow solid, 28 mg, yield 19.29%).
1H NMR(400MHz,DMSO-d6)δ8.43(s,1H),8.02(s,1H),7.34(s,1H),6.93(s,1H),4.57(t,2H),4.39-4.27(m,3H),3.26(s,3H),3.17(t,2H),2.15-2.05(m,2H),1.97(t,2H),1.82-1.76(m,2H),1.48-1.43(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.43(s,1H), 8.02(s,1H), 7.34(s,1H), 6.93(s,1H), 4.57(t,2H), 4.39-4.27( m, 3H), 3.26 (s, 3H), 3.17 (t, 2H), 2.15-2.05 (m, 2H), 1.97 (t, 2H), 1.82-1.76 (m, 2H), 1.48-1.43 (m, 2H).
LC-MS m/z(ESI)=428.10[M+1]。LC-MS m/z(ESI)=428.10[M+1].
实施例56Example 56
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(2-氧杂螺[3.5]壬基-7-基)-7,9-二氢-8H-嘌呤-8-酮(化合物56)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(2-oxaspiro[3.5]nonyl-7-yl)-7 ,9-Dihydro-8H-purin-8-one (Compound 56)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(2-oxaspiro[3.5]。nonan-7-yl)-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(2-oxaspiro[3.5].nonan-7-yl)-7,9-dihydro-8H- purin-8-one
化合物56合成方法与化合物25相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(2-氧杂螺[3.5]壬基-7-基)-7,9-二氢-8H-嘌呤-8-酮(化合物56)(白色固体,38mg,产率26.55%)。The synthesis method of compound 56 is the same as that of compound 25 to obtain the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(2-oxaspiro [3.5] Nonyl-7-yl)-7,9-dihydro-8H-purin-8-one (Compound 56) (white solid, 38 mg, yield 26.55%).
1H NMR(400MHz,DMSO-d6)δ8.38(s,1H),8.00(s,1H),7.33(s,1H),6.92(s,1H),4.56(t,2H),4.20(s,2H),4.12(s,2H),4.08-3.98(m,1H),3.26(s,3H),3.18(t,2H),2.13-2.04(m,4H),1.60-1.43(m,4H)。1H NMR(400MHz,DMSO-d6)δ8.38(s,1H),8.00(s,1H),7.33(s,1H),6.92(s,1H),4.56(t,2H),4.20(s, 2H), 4.12 (s, 2H), 4.08-3.98 (m, 1H), 3.26 (s, 3H), 3.18 (t, 2H), 2.13-2.04 (m, 4H), 1.60-1.43 (m, 4H) .
LC-MS m/z(ESI)=442.20[M+1]。LC-MS m/z(ESI)=442.20[M+1].
实施例57Example 57
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(3-羟基双环[3.2.1]辛基-8-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物57)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(3-hydroxybicyclo[3.2.1]octyl-8-yl)-7-methyl- 7,9-dihydro-8H-purin-8-one (Compound 57)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(3-hydroxybicyclo[3.2.1]octan-8-yl)-7-methyl-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(3-hydroxybicyclo[3.2.1]octan-8-yl)-7-methyl-7,9-dihydro-8H -purin-8-one
化合物57合成方法与化合物26相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(3-羟基双环[3.2.1]辛基-8-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物57)(白色固体,22mg,产率30.74%)。The synthesis method of compound 57 is the same as that of compound 26 to obtain the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(3-hydroxybicyclo[3.2.1]octane (Yl-8-yl)-7-methyl-7,9-dihydro-8H-purin-8-one (Compound 57) (white solid, 22 mg, yield 30.74%).
1H NMR(400MHz,DMSO-d6)δ8.23(s,1H),8.04(s,1H),7.46(s,1H),6.89(s,1H),4.57(t,2H),4.24(d,1H),3.73-3.63(m,1H),3.54-3.52(m,1H),3.42(s,2H),3.29(s,3H),3.15(t,2H),1.65-1.54(m,4H),1.52-1.48(m,2H),1.32-1.26(m,2H)。1H NMR (400MHz, DMSO-d6) δ 8.23 (s, 1H), 8.04 (s, 1H), 7.46 (s, 1H), 6.89 (s, 1H), 4.57 (t, 2H), 4.24 (d, 1H), 3.73-3.63(m, 1H), 3.54-3.52(m, 1H), 3.42(s, 2H), 3.29(s, 3H), 3.15(t, 2H), 1.65-1.54(m, 4H) , 1.52-1.48 (m, 2H), 1.32-1.26 (m, 2H).
LC-MS m/z(ESI)=442.10[M+1]。LC-MS m/z(ESI)=442.10[M+1].
实施例58Example 58
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(3-羟基-3-甲基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物58)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(3-hydroxy-3-methylcyclohexyl)-7-methyl-7,9-di Hydrogen-8H-purin-8-one (Compound 58)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(3-hydroxy-3-methyl cyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(3-hydroxy-3-methylcyclohexyl)-7-methyl-7,9-dihydro-8H-purin-8- one
化合物58合成方法与化合物27相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(3-羟基-3-甲基环己基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物58)(灰色固体,20mg,产率13.81%)。The synthesis method of compound 58 is the same as that of compound 27 to obtain the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(3-hydroxy-3-methylcyclohexyl) )-7-methyl-7,9-dihydro-8H-purin-8-one (Compound 58) (gray solid, 20 mg, yield 13.81%).
1H NMR(400MHz,DMSO-d6)δ8.28(s,1H),7.99(s,1H),7.51(s,1H),6.90(s,1H),4.60-4.49(m,3H),4.28(s,1H),3.26(s,3H),3.16(t,2H),2.26(t,1H),2.09–1.96(m,1H),1.72-1.59(m,2H),1.54(d,3H),1.19-1.13(m,1H),1.12(s,3H)。1H NMR (400MHz, DMSO-d6) δ 8.28 (s, 1H), 7.99 (s, 1H), 7.51 (s, 1H), 6.90 (s, 1H), 4.60-4.49 (m, 3H), 4.28 ( s, 1H), 3.26 (s, 3H), 3.16 (t, 2H), 2.26 (t, 1H), 2.09-1.96 (m, 1H), 1.72-1.59 (m, 2H), 1.54 (d, 3H) , 1.19-1.13 (m, 1H), 1.12 (s, 3H).
LC-MS m/z(ESI)=430.20[M+1]。LC-MS m/z(ESI)=430.20[M+1].
实施例59Example 59
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(6-(羟甲基)螺[3.3]庚烷-2-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物59)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(6-(hydroxymethyl)spiro[3.3]heptan-2-yl)-7-methyl Gyl-7,9-dihydro-8H-purin-8-one (Compound 59)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(6-(hydroxymethyl)spiro[3.3]heptan-2-yl)-7-methyl-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(6-(hydroxymethyl)spiro[3.3]heptan-2-yl)-7-methyl-7,9-dihydro- 8H-purin-8-one
化合物59合成方法与化合物29相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-9-(6-(羟甲基)螺[3.3]庚烷-2-基)-7-甲基-7,9-二氢-8H-嘌呤-8-酮(化合物59)(灰色固体,40mg,产率27.93%)。The synthesis method of compound 59 is the same as that of compound 29 to obtain the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-9-(6-(hydroxymethyl)spiro[3.3 ]Heptan-2-yl)-7-methyl-7,9-dihydro-8H-purin-8-one (Compound 59) (gray solid, 40 mg, yield 27.93%).
1H NMR(400MHz,DMSO-d 6)δ8.34(s,1H),8.00(s,1H),7.42(s,1H),6.89(s,1H),4.56(t,3H),4.45(t,1H),3.33–3.29(m,2H),3.24(s,3H),3.17(t,2H),2.89-2.79(m,2H),2.29–2.17(m,2H),2.13-2.07(m,2H),1.82-1.77(m,2H),1.65-1.59(m,1H)。 1 H NMR (400MHz, DMSO-d 6 ) δ 8.34 (s, 1H), 8.00 (s, 1H), 7.42 (s, 1H), 6.89 (s, 1H), 4.56 (t, 3H), 4.45 ( t,1H),3.33–3.29(m,2H), 3.24(s,3H), 3.17(t,2H), 2.89-2.79(m,2H), 2.29–2.17(m,2H),2.13-2.07( m, 2H), 1.82-1.77 (m, 2H), 1.65-1.59 (m, 1H).
LC-MS m/z(ESI)=416.10[M+1]。LC-MS m/z(ESI)=416.10[M+1].
实施例60Example 60
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-乙基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物60)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-ethyl-9-(tetrahydro-2H-pyran-4-yl)-7,9- Dihydro-8H-purin-8-one (Compound 60)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-ethyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-ethyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8 -one
化合物60合成方法与化合物30相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-乙基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物60)(浅黄色固体,31mg,产率21.08%)。The synthesis method of compound 60 is the same as that of compound 30, and the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-ethyl-9-(tetrahydro-2H- Pyran-4-yl)-7,9-dihydro-8H-purin-8-one (Compound 60) (pale yellow solid, 31 mg, yield 21.08%).
1H NMR(400MHz,DMSO-d6)δ8.30(s,1H),8.07(s,1H),7.55(s,1H),6.89(s,1H),4.57(t,2H),4.38(tt,1H),3.95(dd,2H),3.80(q,2H),3.41(t,2H),3.17(t,2H),2.57-2.44(m,2H),1.69-1.60(m,2H),1.21(t,3H)。1H NMR (400MHz, DMSO-d6) δ 8.30 (s, 1H), 8.07 (s, 1H), 7.55 (s, 1H), 6.89 (s, 1H), 4.57 (t, 2H), 4.38 (tt, 1H), 3.95 (dd, 2H), 3.80 (q, 2H), 3.41 (t, 2H), 3.17 (t, 2H), 2.57-2.44 (m, 2H), 1.69-1.60 (m, 2H), 1.21 (t, 3H).
LC-MS m/z(ESI)=416.10[M+1]。LC-MS m/z(ESI)=416.10[M+1].
实施例61Example 61
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-异丙基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物61)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-isopropyl-9-(tetrahydro-2H-pyran-4-yl)-7,9 -Dihydro-8H-purin-8-one (Compound 61)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-isopropyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-isopropyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8 -one
化合物61合成方法与化合物31相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-异丙基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物61)(白色固体,15mg,产率10.35%)。The synthesis method of compound 61 is the same as that of compound 31 to obtain the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-isopropyl-9-(tetrahydro-2H -Pyran-4-yl)-7,9-dihydro-8H-purin-8-one (compound 61) (white solid, 15 mg, yield 10.35%).
1H NMR(400MHz,DMSO-d6)δ8.31(s,1H),8.19(s,1H),7.55(s,1H),6.90(s,1H),4.61-4.50(m,3H),4.38(tt,1H),3.96(dd,2H),3.41(t,2H),3.17(t,2H),2.51(d,6H),1.68-1.60(m,2H),1.45-1.36(m,6H)。1H NMR(400MHz,DMSO-d6)δ8.31(s,1H), 8.19(s,1H), 7.55(s,1H), 6.90(s,1H), 4.61-4.50(m,3H), 4.38( tt,1H),3.96(dd,2H),3.41(t,2H),3.17(t,2H),2.51(d,6H),1.68-1.60(m,2H),1.45-1.36(m,6H) .
LC-MS m/z(ESI)=430.20[M+1]。LC-MS m/z(ESI)=430.20[M+1].
实施例62Example 62
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-环丙基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物62)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-cyclopropyl-9-(tetrahydro-2H-pyran-4-yl)-7,9 -Dihydro-8H-purin-8-one (Compound 62)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-cyclopropyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-cyclopropyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8 -one
化合物62合成方法与化合物32相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-环丙基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物62)(灰白色固体,14mg,产率9.64%)。The synthesis method of compound 62 is the same as that of compound 32 to obtain the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-cyclopropyl-9-(tetrahydro-2H -Pyran-4-yl)-7,9-dihydro-8H-purin-8-one (Compound 62) (off-white solid, 14 mg, yield 9.64%).
1H NMR(400MHz,DMSO-d6)δ8.31(s,1H),7.99(s,1H),7.54(s,1H),6.90(s,1H),4.57(t,2H),4.34(tt,1H),3.95(dd,2H),3.40(t,2H),3.17(t,2H),2.89(tt,1H),2.55-2.46(s,2H),1.66-1.58(m,2H),1.00-0.93(m,2H),0.91-0.85(m,2H)。1H NMR(400MHz,DMSO-d6)δ8.31(s,1H),7.99(s,1H),7.54(s,1H),6.90(s,1H),4.57(t,2H), 4.34(tt, 1H), 3.95(dd, 2H), 3.40(t, 2H), 3.17(t, 2H), 2.89(tt, 1H), 2.55-2.46(s, 2H), 1.66-1.58(m, 2H), 1.00 -0.93 (m, 2H), 0.91-0.85 (m, 2H).
LC-MS m/z(ESI)=428.20[M+1]。LC-MS m/z(ESI)=428.20[M+1].
实施例63Example 63
2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-(甲基-d3)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物63)2-((6-Chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-(methyl-d3)-9-(tetrahydro-2H-pyran-4-yl)- 7,9-dihydro-8H-purin-8-one (Compound 63)
2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-(methyl-d3)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-(methyl-d3)-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H -purin-8-one
化合物63合成方法与化合物33相同,得到标题化合物2-((6-氯-2,3-二氢苯并呋喃-5-基)氨基)-7-(甲基-d3)-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物63)(白色固体,45mg,产率30.20%)。The synthesis method of compound 63 is the same as that of compound 33 to obtain the title compound 2-((6-chloro-2,3-dihydrobenzofuran-5-yl)amino)-7-(methyl-d3)-9-(tetra Hydrogen-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one (Compound 63) (white solid, 45 mg, yield 30.20%).
1H NMR(400MHz,DMSO-d6)δ8.29(s,1H),8.01(s,1H),7.56(s,1H),6.89(s,1H),4.57(t,2H),4.38(tt,1H),3.96(dd,2H),3.41(t,2H),3.20-3.16(m,2H),2.56-2.44(m,2H),1.67-1.61(m,2H)。1H NMR (400MHz, DMSO-d6) δ 8.29 (s, 1H), 8.01 (s, 1H), 7.56 (s, 1H), 6.89 (s, 1H), 4.57 (t, 2H), 4.38 (tt, 1H), 3.96 (dd, 2H), 3.41 (t, 2H), 3.20-3.16 (m, 2H), 2.56-2.44 (m, 2H), 1.67-1.61 (m, 2H).
LC-MS m/z(ESI)=405.10[M+1]。LC-MS m/z(ESI)=405.10[M+1].
实施例64Example 64
2-((6-氟-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物64)2-((6-Fluoro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydro-2H-pyran-4-yl)-7,9- Dihydro-8H-purin-8-one (Compound 64)
2-((6-fluoro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one2-((6-fluoro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8 -one
第一步:first step:
6-氟-5-硝基-2,3-二氢苯并呋喃(64b)6-Fluoro-5-nitro-2,3-dihydrobenzofuran (64b)
6-fluoro-5-nitro-2,3-dihydrobenzofuran6-fluoro-5-nitro-2,3-dihydrobenzofuran
将6-氟-2,3-二氢苯并呋喃64a(0.8g,5.79mmol)溶于30mL醋酸中,冰浴下滴入硝酸(660.6mg,69%纯度),反应在70℃搅拌1h。TLC监测反应完全,将反应液倒入冰水中,萃取,浓缩有机相得黄色油状粗品,粗品经过硅胶柱层析分离(石油醚:乙酸乙酯(v/v)=10/1)得到无色透明固体状的标题化合物,6-氟-5-硝基-2,3-二氢苯并呋喃(64b)(白色固体,0.5g,产率47.14%)。6-Fluoro-2,3-dihydrobenzofuran 64a (0.8g, 5.79mmol) was dissolved in 30mL of acetic acid, nitric acid (660.6mg, 69% purity) was added dropwise under ice bath, and the reaction was stirred at 70°C for 1h. TLC monitors that the reaction is complete, the reaction solution is poured into ice water, extracted, and the organic phase is concentrated to obtain a yellow oily crude product. The crude product is separated by silica gel column chromatography (petroleum ether: ethyl acetate (v/v) = 10/1) to obtain colorless The title compound as a clear solid, 6-fluoro-5-nitro-2,3-dihydrobenzofuran (64b) (white solid, 0.5 g, yield 47.14%).
1H NMR(400MHz CDCl 3)δ7.97(d,1H),6.62(d,1H),4.77(t,2H),3.27(t,2H)。 1 H NMR (400MHz CDCl 3 ) δ 7.97 (d, 1H), 6.62 (d, 1H), 4.77 (t, 2H), 3.27 (t, 2H).
第二步:The second step:
6-氟-2,3-二氢苯并呋喃-5-胺(64c)6-Fluoro-2,3-dihydrobenzofuran-5-amine (64c)
6-fluoro-2,3-dihydrobenzofuran-5-amine6-fluoro-2,3-dihydrobenzofuran-5-amine
将Pd/C(10%,湿载体)(50mg)添加到6-氟-5-硝基-2,3-二氢苯并呋喃64b(0.5g,2.73mmol)的甲醇(100mL)溶液中,在氢气氛围下室温反应过夜。过滤,浓缩得标题化合物6-氟-2,3-二氢苯并呋喃-5-胺(64c)(无色固体,0.4g,产率95.66%)。Pd/C (10%, wet carrier) (50mg) was added to a solution of 6-fluoro-5-nitro-2,3-dihydrobenzofuran 64b (0.5g, 2.73mmol) in methanol (100mL), React overnight at room temperature under hydrogen atmosphere. It was filtered and concentrated to obtain the title compound 6-fluoro-2,3-dihydrobenzofuran-5-amine (64c) (colorless solid, 0.4g, yield 95.66%).
1H NMR(400MHz CDCl 3)δ6.66(d,1H),6.50(d,1H),4.52(t,2H),3.1(t,2H)。 1 H NMR (400MHz CDCl 3 ) δ 6.66 (d, 1H), 6.50 (d, 1H), 4.52 (t, 2H), 3.1 (t, 2H).
第三步:third step:
2-((6-氟-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物64)2-((6-Fluoro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydro-2H-pyran-4-yl)-7,9- Dihydro-8H-purin-8-one (Compound 64)
2-((6-fluoro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one2-((6-fluoro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8 -one
将2-氯-7-甲基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮3e(315mg,1.18mmol,参考WO2018114999中间体20方法制备)、6-氟-2,3-二氢苯并呋喃-5-胺36c(150mg,0.979mmol)、碳酸铯(638.21g,1.96mmol)、三(二亚苄基丙酮)二钯(89.57mg,0.09mmol)和2,2'-双(二苯基膦基)-1,1'-联萘(121.84mg,0.18mmol)溶解于二氧六环,氮气保护并换气,在100℃搅拌4h。TLC监测反应结束,将反应液倒入冰水中,收集固体,将固体用硅胶柱色谱分离提纯(二氯甲烷:甲醇(v/v)=30/1),得到标题化合物,2-((6-氟-2,3-二氢苯并呋喃-5-基)氨基)-7-甲基-9-(四氢-2H-吡喃-4-基)-7,9-二氢-8H-嘌呤-8-酮(化合物64)(白色固体,140mg,产率39.74%)。The 2-chloro-7-methyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H-purin-8-one 3e (315mg, 1.18mmol, refer to the middle of WO2018114999) Body 20 method preparation), 6-fluoro-2,3-dihydrobenzofuran-5-amine 36c (150mg, 0.979mmol), cesium carbonate (638.21g, 1.96mmol), three (dibenzylidene acetone) two Palladium (89.57mg, 0.09mmol) and 2,2'-bis(diphenylphosphino)-1,1'-binaphthalene (121.84mg, 0.18mmol) were dissolved in dioxane, protected by nitrogen and ventilated, Stir at 100°C for 4h. TLC monitors the end of the reaction, the reaction solution is poured into ice water, the solid is collected, and the solid is separated and purified by silica gel column chromatography (dichloromethane: methanol (v/v) = 30/1) to obtain the title compound, 2-((6 -Fluoro-2,3-dihydrobenzofuran-5-yl)amino)-7-methyl-9-(tetrahydro-2H-pyran-4-yl)-7,9-dihydro-8H- Purin-8-one (Compound 64) (white solid, 140 mg, yield 39.74%).
1H NMR(400MHz DMSO)δ8.50(s,1H),8.0(s,1H),7.43(d,1H),6.80(d,2H),4.56(t,2H),4.33-4.39(m,1H),3.93-3.97(m,2H),3.37-3.45(m,2H),3.27(s,3H),3.15(t,2H),1.64(t,2H)。 1 H NMR(400MHz DMSO)δ8.50(s,1H),8.0(s,1H),7.43(d,1H),6.80(d,2H),4.56(t,2H),4.33-4.39(m, 1H), 3.93-3.97 (m, 2H), 3.37-3.45 (m, 2H), 3.27 (s, 3H), 3.15 (t, 2H), 1.64 (t, 2H).
LC-MS m/z(ESI)=386.10[M+1]。LC-MS m/z(ESI)=386.10[M+1].
生物学试验Biological test
1、DNA-PK激酶抑制试验1. DNA-PK kinase inhibition test
通过DNA-PK激酶检测试剂盒(DNA-PK kinase assay kit)(购买自Promega公司,货号:V4107,批号:0000366495)检测化合物对DNA-PK激酶的抑制活性。利用化学发光对结果进行定量,具体实验方案如下:The DNA-PK kinase assay kit (purchased from Promega, product number: V4107, batch number: 0000366495) was used to detect the inhibitory activity of the compound against DNA-PK kinase. Using chemiluminescence to quantify the results, the specific experimental plan is as follows:
i.按照试剂盒说明书构建不同浓度ADP-荧光标准曲线;i. Construct different concentrations of ADP-fluorescence standard curve according to the kit instructions;
ii.于384孔白色板中制备5μL反应体系,每孔中分别加入1μL化合物(分别设定浓度梯度1μM、200nM、40nM、8nM、1.6nM、0.32nM、0.064nM、0.013nM)、20units DNA-PK激酶、0.2μg/μL底物、10μg/μL DNA、50μM ATP、1%DMSO;ii. Prepare a 5μL reaction system in a 384-well white plate, and add 1μL of compound to each well (set concentration gradients of 1μM, 200nM, 40nM, 8nM, 1.6nM, 0.32nM, 0.064nM, 0.013nM), 20units DNA- PK kinase, 0.2μg/μL substrate, 10μg/μL DNA, 50μM ATP, 1% DMSO;
iii.混匀,离心(1000rpm,30s),37℃孵育60min;iii. Mix well, centrifuge (1000rpm, 30s), and incubate at 37°C for 60min;
iv.加入5μL ADP‐Glo TM Reagent终止反应,混匀,离心(1000rpm,30s),室温孵育40min; iv. Add 5μL ADP-Glo TM Reagent to terminate the reaction, mix well, centrifuge (1000rpm, 30s), and incubate at room temperature for 40min;
v.加入10μL Kinase Detection Reagent,震荡混匀,离心(1000rpm,30s),室温孵育30min;v. Add 10μL Kinase Detection Reagent, shake and mix, centrifuge (1000rpm, 30s), and incubate at room temperature for 30min;
vi.利用酶标仪(Thermo fisher,Varioskan LUX)测定荧光值。利用GraphPad Pris m 8进行IC 50的计算,结果见表1。 vi. Measure the fluorescence value using a microplate reader (Thermo fisher, Varioskan LUX). Using GraphPad Pris m 8 to calculate IC 50 , the results are shown in Table 1.
表1本发明化合物DNA-PK激酶抑制活性Table 1 DNA-PK kinase inhibitory activity of the compounds of the present invention
注:对照例为参考J.Med.Chem(2020),63(7),3461-3471的化合物3,对照例按照其制备方法制备得到。Note: The comparative example refers to compound 3 of J. Med. Chem (2020), 63(7), 3461-3471, and the comparative example is prepared according to its preparation method.
结论:结果表明,与对照例相比,本发明化合物对DNA-PK激酶具有更显著的抑制效果。Conclusion: The results show that, compared with the control example, the compound of the present invention has a more significant inhibitory effect on DNA-PK kinase.
2、增殖抑制实验2. Proliferation inhibition experiment
2.1、实验材料2.1. Experimental materials
A549细胞(购自ATCC)、Doxorubicin hydrochloride(多柔比星盐酸盐)(购自上海陶素生化科技有限公司,货号:T1020)、DMEM(10%FBS)培养基、96孔白板、细胞活力检测试剂盒(购自Promega,货号:G9241)。A549 cells (purchased from ATCC), Doxorubicin hydrochloride (purchased from Shanghai Taosu Biochemical Technology Co., Ltd., catalog number: T1020), DMEM (10% FBS) medium, 96-well white board, cell viability Detection kit (purchased from Promega, article number: G9241).
2.2、实验步骤2.2. Experimental steps
A549细胞重悬后进行计数,铺96孔板,500个/孔(80μL);过夜培养。The A549 cells were resuspended and counted, and then spread in a 96-well plate, 500 cells/well (80 μL); cultured overnight.
配置10×药物浓度梯度(1:5):初始浓度为100μM,用培养基1:5倍比稀释为20μM、4μM、800nM、160nM、32nM、6.4nM、1.3nM(终浓度10μM、2μM、0.4μM、80nM、16nM、3.2nM、0.64nM、0.13nM)。Configure 10× drug concentration gradient (1:5): the initial concentration is 100μM, diluted with medium 1:5 to 20μM, 4μM, 800nM, 160nM, 32nM, 6.4nM, 1.3nM (final concentration 10μM, 2μM, 0.4 μM, 80nM, 16nM, 3.2nM, 0.64nM, 0.13nM).
3.每孔中加入10μL待测化合物,每个浓度梯度做2个重复,设置单药组(待测化合物或者Doxorubicin)、联用组、培养基对照组:3. Add 10 μL of the test compound to each well, do 2 replicates for each concentration gradient, set up the single-drug group (test compound or Doxorubicin), combination group, and medium control group:
单药组:加入待测化合物(终浓度10μM、2μM、0.4μM、80nM、16nM、3.2nM、0.64nM、0.13nM),或者只加入doxorubicin(终浓度10μM、2μM、0.4μM、80nM、16nM、3.2nM、0.64nM、0.13nM),37℃温箱继续培养120h;Single-drug group: add the test compound (final concentration 10μM, 2μM, 0.4μM, 80nM, 16nM, 3.2nM, 0.64nM, 0.13nM), or only add doxorubicin (final concentration 10μM, 2μM, 0.4μM, 80nM, 16nM, 3.2nM, 0.64nM, 0.13nM), continue to incubate for 120h in a 37°C incubator;
联用组:加入待测化合物(终浓度10μM、2μM、0.4μM、80nM、16nM、3.2nM、0.64nM、0.13nM),37℃温箱孵育1h后,每孔再加入10nM Doxorubicin(10μL/孔),37℃温箱继续培养120h;Combination group: add test compound (final concentration 10μM, 2μM, 0.4μM, 80nM, 16nM, 3.2nM, 0.64nM, 0.13nM), incubate at 37℃ for 1h, add 10nM Doxorubicin (10μL/well) ), continue to incubate for 120h in 37℃ incubator;
对照组:加入20μL培养基,37℃温箱继续培养120h。Control group: add 20μL of culture medium and continue to incubate for 120h in 37℃ incubator.
4.取出细胞培养板,加入细胞培养液等体积的检测液(100μL培养基,加入100μL检测液)。4. Take out the cell culture plate and add an equal volume of cell culture medium to the test solution (100 μL medium, add 100 μL test solution).
5.震板机上震动2min,裂解细胞。5. Vibrate on the shaker for 2 minutes to lyse the cells.
6.室温放置10min,使信号稳定。6. Leave at room temperature for 10 minutes to stabilize the signal.
7.酶标仪(Thermo fisher;Varioskan LUX)测定化学发光值。7. Microplate reader (Thermo fisher; Varioskan LUX) measures the chemiluminescence value.
结论:本发明化合物与Doxorubicin联用对A549细胞具有更显著增殖抑制作用,且细胞毒性降低。Conclusion: The combination of the compound of the present invention and Doxorubicin has a more significant inhibitory effect on the proliferation of A549 cells and reduces cytotoxicity.
3、移植瘤抑制实验3. The transplantation tumor inhibition experiment
3.1实验材料:A549细胞(购自ATCC);Doxorubicin脂质体(Dox)(Lipo Doxorubicin,商品名“里葆多”,购自上海复旦张江生物医药股份有限公司);6周龄雌性裸鼠(体重18-20g)(北京维通利华实验动物技术有限公司),每组10只鼠。3.1 Experimental materials: A549 cells (purchased from ATCC); Doxorubicin liposomes (Dox) (Lipo Doxorubicin, trade name "Libaoduo", purchased from Shanghai Fudan Zhangjiang Biomedical Co., Ltd.); 6-week-old female nude mice ( Body weight 18-20g) (Beijing Weitong Lihua Experimental Animal Technology Co., Ltd.), 10 rats in each group.
3.2 Doxorubicin与待筛选化合物联用对A549移植瘤的抑制效果测定:3.2 Determination of the inhibitory effect of Doxorubicin in combination with the compound to be screened on A549 xenograft tumors:
3.2.1收集处于生长对数期的A549细胞,预冷PBS洗2次后备用;3.2.1 Collect the A549 cells in the logarithmic growth phase, wash them twice with pre-cooled PBS and use them for later use;
3.2.2 Balb/c裸小鼠实验室环境适应3天,于右肋部皮下接种A549细胞,接种细胞量为5×10 6/只,待肿瘤生长至200mm 3左右时进行药效实验; 3.2.2 Balb/c nude mice were adapted to the laboratory environment for 3 days, and A549 cells were subcutaneously inoculated on the right ribs with a cell amount of 5×10 6 per mouse. When the tumor grows to about 200 mm 3 , the efficacy experiment will be carried out;
3.2.3将成功长瘤的小鼠进行随机分组,设置单独Doxorubicin(Dox)组、待测化合物和Dox联用组、对照组(Vehicle),给药21天;小鼠灌胃给药(i.g.),每日2次(BID,给药体积5mL/kg;溶剂为5%DMSO+30%2-羟丙基-β-环糊精)。早上灌胃给药1h后,由尾静脉注射Lipo Doxorubicin(2.5mg/kg);每周1次(QW,给药体积5mL/kg;);3.2.3 Randomly group the mice that have successfully grown tumors, and set up a single Doxorubicin (Dox) group, a test compound and Dox combined group, and a control group (Vehicle) for 21 days; mice are given intragastric administration (ig ), twice a day (BID, administration volume 5mL/kg; solvent: 5% DMSO+30% 2-hydroxypropyl-β-cyclodextrin). After intragastric administration in the morning for 1 hour, Lipo Doxorubicin (2.5mg/kg) was injected from the tail vein; once a week (QW, administration volume 5mL/kg;);
3.2.4每周称量2次小鼠体重,并同时测定肿瘤体积:肿瘤体积(V)计算公式为:V=1/2×L 长×L 短 2,并计算抑瘤率,抑瘤率(%)=(D21肿瘤体积(Vehicle)-D21肿瘤体积(给药组))/D21肿瘤体积(Vehicle)×100; 3.2.4 Weigh the weight of the mice twice a week, and measure the tumor volume at the same time: the tumor volume (V) is calculated as: V = 1/2 × L long × L short 2 , and calculate the tumor inhibition rate and tumor inhibition rate (%)=(D21 tumor volume (Vehicle)-D21 tumor volume (administration group))/D21 tumor volume (Vehicle)×100;
3.2.5给药21天后,分离肿瘤并称重,并计算体重变化率,体重变化率(%)=(D21体重-D0体重)/D0体重×100。3.2.5 After 21 days of administration, the tumor was isolated and weighed, and the weight change rate was calculated. The weight change rate (%)=(D21 weight-D0 weight)/D0 weight×100.
结论:实验结果表明,本发明化合物与Doxorubicin联用均显著提高Doxorubicin的肿瘤抑制效果,且不会引起明显的体重减轻。Conclusion: The experimental results show that the combination of the compound of the present invention and Doxorubicin can significantly improve the tumor suppressive effect of Doxorubicin, and will not cause significant weight loss.
本发明说明书对具体实施方案进行了详细描述,本领域技术人员应认识到,上述实施方案是示例性的,不能理解为对本发明的限制,对于本领域技术人员来说,在不脱离本发明原理的前提下,通过对本发明进行若干改进和修饰,这些改进和修饰获得技术方案也落在本发明的权利要求书的保护范围内。The specification of the present invention describes the specific embodiments in detail. Those skilled in the art should realize that the above-mentioned embodiments are exemplary and should not be construed as limiting the present invention. For those skilled in the art, the principle of the present invention is not deviated from Under the premise of making several improvements and modifications to the present invention, the technical solutions obtained by these improvements and modifications also fall within the protection scope of the claims of the present invention.
Claims (10)
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201911410949.8 | 2019-12-31 | ||
| CN201911410949 | 2019-12-31 | ||
| CN202010154743 | 2020-03-11 | ||
| CN202010154743.X | 2020-03-11 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2021136462A1 true WO2021136462A1 (en) | 2021-07-08 |
Family
ID=76686500
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2020/141860 Ceased WO2021136462A1 (en) | 2019-12-31 | 2020-12-30 | Furan derivatives and application thereof in medicine |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN113121538B (en) |
| WO (1) | WO2021136462A1 (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022135360A1 (en) * | 2020-12-21 | 2022-06-30 | 江苏恒瑞医药股份有限公司 | Purinone derivative, preparation method therefor, and application thereof in medicine |
| WO2022135555A1 (en) * | 2020-12-24 | 2022-06-30 | 江苏恒瑞医药股份有限公司 | Purinone compound, preparation method therefor, and pharmaceutical application thereof |
| WO2022199547A1 (en) * | 2021-03-22 | 2022-09-29 | 成都赜灵生物医药科技有限公司 | 7,9-dihydropurine derivative and pharmaceutical purpose thereof |
| WO2025049250A1 (en) * | 2023-08-25 | 2025-03-06 | Juno Therapeutics, Inc. | 6,6-core silacycle based inhibitors of dna-dependent protein kinase and compositions and application in gene editing |
| WO2025049247A1 (en) * | 2023-08-25 | 2025-03-06 | Juno Therapeutics, Inc. | 5,6-core silacycle based inhibitors of dna-dependent protein kinase and compositions and application in gene editing |
| WO2025049253A1 (en) * | 2023-08-25 | 2025-03-06 | Juno Therapeutics, Inc. | Bridged cycle‑based inhibitors of dna‑dependent protein kinase and compositions and application in gene editing |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN116406272A (en) * | 2020-07-20 | 2023-07-07 | 首药控股(北京)股份有限公司 | DNA-PK selective inhibitor and its preparation method and use |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103649085A (en) * | 2011-05-25 | 2014-03-19 | 阿尔米雷尔有限公司 | Pyridin-2(1h)-one derivatives useful as medicaments for the treatment of myeloproliferative disorders, transplant rejection, immune-mediated and inflammatory diseases |
| CN105246883A (en) * | 2013-03-12 | 2016-01-13 | 沃泰克斯药物股份有限公司 | DNA-PK inhibitors |
| CN110177791A (en) * | 2016-12-20 | 2019-08-27 | 阿斯利康(瑞典)有限公司 | Amino-triazolopyridine compound and its purposes in treating cancer |
| CN110386932A (en) * | 2018-04-20 | 2019-10-29 | 艾科思莱德制药公司 | Dual ATM and DNA-PK inhibitors for use in antitumor therapy |
| WO2019238929A1 (en) * | 2018-06-15 | 2019-12-19 | Astrazeneca Ab | Purinone compounds and their use in treating cancer |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105384695B (en) * | 2014-08-22 | 2019-12-20 | 四川海思科制药有限公司 | Pyrimidine derivative, preparation method and medical application thereof |
| WO2019201283A1 (en) * | 2018-04-20 | 2019-10-24 | Xrad Therapeutics, Inc. | Dual atm and dna-pk inhibitors for use in anti-tumor therapy |
-
2020
- 2020-12-30 WO PCT/CN2020/141860 patent/WO2021136462A1/en not_active Ceased
- 2020-12-30 CN CN202011615932.9A patent/CN113121538B/en active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103649085A (en) * | 2011-05-25 | 2014-03-19 | 阿尔米雷尔有限公司 | Pyridin-2(1h)-one derivatives useful as medicaments for the treatment of myeloproliferative disorders, transplant rejection, immune-mediated and inflammatory diseases |
| CN105246883A (en) * | 2013-03-12 | 2016-01-13 | 沃泰克斯药物股份有限公司 | DNA-PK inhibitors |
| CN110177791A (en) * | 2016-12-20 | 2019-08-27 | 阿斯利康(瑞典)有限公司 | Amino-triazolopyridine compound and its purposes in treating cancer |
| CN110386932A (en) * | 2018-04-20 | 2019-10-29 | 艾科思莱德制药公司 | Dual ATM and DNA-PK inhibitors for use in antitumor therapy |
| WO2019238929A1 (en) * | 2018-06-15 | 2019-12-19 | Astrazeneca Ab | Purinone compounds and their use in treating cancer |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022135360A1 (en) * | 2020-12-21 | 2022-06-30 | 江苏恒瑞医药股份有限公司 | Purinone derivative, preparation method therefor, and application thereof in medicine |
| WO2022135555A1 (en) * | 2020-12-24 | 2022-06-30 | 江苏恒瑞医药股份有限公司 | Purinone compound, preparation method therefor, and pharmaceutical application thereof |
| WO2022199547A1 (en) * | 2021-03-22 | 2022-09-29 | 成都赜灵生物医药科技有限公司 | 7,9-dihydropurine derivative and pharmaceutical purpose thereof |
| WO2025049250A1 (en) * | 2023-08-25 | 2025-03-06 | Juno Therapeutics, Inc. | 6,6-core silacycle based inhibitors of dna-dependent protein kinase and compositions and application in gene editing |
| WO2025049247A1 (en) * | 2023-08-25 | 2025-03-06 | Juno Therapeutics, Inc. | 5,6-core silacycle based inhibitors of dna-dependent protein kinase and compositions and application in gene editing |
| WO2025049253A1 (en) * | 2023-08-25 | 2025-03-06 | Juno Therapeutics, Inc. | Bridged cycle‑based inhibitors of dna‑dependent protein kinase and compositions and application in gene editing |
Also Published As
| Publication number | Publication date |
|---|---|
| CN113121538B (en) | 2023-04-21 |
| CN113121538A (en) | 2021-07-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN113121538B (en) | Furan derivative and application thereof in medicine | |
| WO2021209055A1 (en) | Imidazolinone derivative and use thereof in medicine | |
| WO2021136461A1 (en) | Purine derivative and medical use thereof | |
| WO2019158019A1 (en) | Pyrimidine-fused cyclic compound, preparation method therefor and application thereof | |
| CN113121574B (en) | Purine derivatives and their use in medicine | |
| CN115073474A (en) | Pyrrolo [2,3-b ] pyrazines as HPK1 inhibitors and uses thereof | |
| CN117157297A (en) | PRMT5 inhibitors | |
| CN112566916A (en) | Substituted thienopyrroles as PAD4 inhibitors | |
| TWI850321B (en) | Polycyclic derivative modulator, preparation method and application thereof | |
| WO2025040170A1 (en) | Novel fused heterocyclic compound serving as cdk inhibitor and use thereof | |
| TWI866007B (en) | p38 MAPK/MK2 PATHWAY REGULATOR, AND COMPOSITION, PREPARATION METHOD, AND USE THEREOF | |
| CN112778183A (en) | Nitrogen-containing ring derivative regulator, preparation method and application thereof | |
| WO2021051899A1 (en) | Cyano-substituted cyclic hydrazine derivative and application thereof | |
| WO2022134641A1 (en) | Aromatic heterocyclic compound, pharmaceutical composition and use thereof | |
| CN113874354A (en) | Pyridone derivatives, their preparation method and their application in medicine | |
| CN114075200A (en) | JAK inhibitor compounds for the treatment of severe pneumonia | |
| WO2024125532A1 (en) | Novel heterocyclic compound acting as cdks inhibitor, and use thereof | |
| WO2017156177A1 (en) | 3-phosphoglycerate dehydrogenase inhibitors and uses thereof | |
| WO2022156779A1 (en) | Substituted pyrazolo[1,5-a]pyrimidine-7-amine derivative, and compositions and medical use thereof | |
| WO2024175024A1 (en) | New fused heterocyclic compound as cdks inhibitor and use thereof | |
| WO2024188143A1 (en) | Nk-3 receptor antagonist compound, pharmaceutical composition, preparation method therefore and use thereof | |
| CN111867584A (en) | Ethylenediamine-heterocyclic derivatives as inhibitors of protein arginine methyltransferases | |
| WO2019085996A1 (en) | Pyridopyrimidine compounds acting as mtorc 1/2 double-kinase inhibitors | |
| KR20250004015A (en) | Substituted 7-(pyrimidin-4-yl)quinolin-4(1H)-one compounds as cyclin-dependent kinase inhibitors | |
| HK40046823A (en) | Furan derivatives and application thereof in medicine |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 20909815 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 20909815 Country of ref document: EP Kind code of ref document: A1 |