WO2021114691A1 - Nitrogen-containing ring-fused compound, preparation method therefor and use thereof - Google Patents
Nitrogen-containing ring-fused compound, preparation method therefor and use thereof Download PDFInfo
- Publication number
- WO2021114691A1 WO2021114691A1 PCT/CN2020/106220 CN2020106220W WO2021114691A1 WO 2021114691 A1 WO2021114691 A1 WO 2021114691A1 CN 2020106220 W CN2020106220 W CN 2020106220W WO 2021114691 A1 WO2021114691 A1 WO 2021114691A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- alkyl
- group
- membered
- aryl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 *c(nc1I*)nc(*2)c1S=C2I* Chemical compound *c(nc1I*)nc(*2)c1S=C2I* 0.000 description 4
- FNVOFDGAASRDQY-UHFFFAOYSA-N CC(C)(CN)CO Chemical compound CC(C)(CN)CO FNVOFDGAASRDQY-UHFFFAOYSA-N 0.000 description 2
- KWLOIDOKWUESNM-UHFFFAOYSA-N CC1(C)OB(c2ccn[nH]2)OC1(C)C Chemical compound CC1(C)OB(c2ccn[nH]2)OC1(C)C KWLOIDOKWUESNM-UHFFFAOYSA-N 0.000 description 2
- RWIVICVCHVMHMU-UHFFFAOYSA-N NCCN1CCOCC1 Chemical compound NCCN1CCOCC1 RWIVICVCHVMHMU-UHFFFAOYSA-N 0.000 description 2
- ZEZJZZPBSKSEOQ-UHFFFAOYSA-N Nc1nc(cc(cc2)Br)c2c(O)n1 Chemical compound Nc1nc(cc(cc2)Br)c2c(O)n1 ZEZJZZPBSKSEOQ-UHFFFAOYSA-N 0.000 description 2
- BSWQOODPVUTOIO-UHFFFAOYSA-N CC(C)(CNc1nc(N)nc2c1ccc(Br)c2)CO Chemical compound CC(C)(CNc1nc(N)nc2c1ccc(Br)c2)CO BSWQOODPVUTOIO-UHFFFAOYSA-N 0.000 description 1
- NTBIRUINHWWRGP-UHFFFAOYSA-N CCC(/N=C(/N)\NC)=C Chemical compound CCC(/N=C(/N)\NC)=C NTBIRUINHWWRGP-UHFFFAOYSA-N 0.000 description 1
- XHPJOYRUTMZHTA-SSDOTTSWSA-N CC[C@H](CO)Nc1c2[s]c(-c3ccn[nH]3)cc2nc(N)n1 Chemical compound CC[C@H](CO)Nc1c2[s]c(-c3ccn[nH]3)cc2nc(N)n1 XHPJOYRUTMZHTA-SSDOTTSWSA-N 0.000 description 1
- RUARECTWZFPDFK-UHFFFAOYSA-N CCc1cc2nc(N)nc(N)c2[s]1 Chemical compound CCc1cc2nc(N)nc(N)c2[s]1 RUARECTWZFPDFK-UHFFFAOYSA-N 0.000 description 1
- ZCOMWNGCZVAIHS-UHFFFAOYSA-N CCc1cc2nc(N)nc(O)c2[s]1 Chemical compound CCc1cc2nc(N)nc(O)c2[s]1 ZCOMWNGCZVAIHS-UHFFFAOYSA-N 0.000 description 1
- FFTDFJXFRVXAGD-UHFFFAOYSA-N Cc1ccc2OCCNc2n1 Chemical compound Cc1ccc2OCCNc2n1 FFTDFJXFRVXAGD-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/517—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with carbocyclic ring systems, e.g. quinazoline, perimidine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/70—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings condensed with carbocyclic rings or ring systems
- C07D239/72—Quinazolines; Hydrogenated quinazolines
- C07D239/95—Quinazolines; Hydrogenated quinazolines with hetero atoms directly attached in positions 2 and 4
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing three or more hetero rings
Definitions
- the present invention relates to a class of nitrogen-containing hexacyclic compounds, stereoisomers, tautomers or mixtures of said compounds, pharmaceutically acceptable salts, co-crystals, polymorphs or solvates of said compounds Or a stable isotope derivative, metabolite or prodrug of the compound.
- the compounds of the present invention are used as NLRP3 (NLR family pyrin domain containing 3) modulators (such as agonists or partial agonists) and can be used for the treatment of abnormal cell proliferation diseases (such as cancer).
- NLRP3 belongs to the family of NOD-like receptors. It is one of the most studied intracellular pattern recognition receptors in recent years. It is mainly expressed in macrophages and neutrophils. It participates in the body's innate immunity and resists pathogen infection and stress damage. NLRP3 inflammasome plays a very clear role in inflammatory and metabolic diseases, and its over-activation can lead to type 2 diabetes, immune diseases such as rheumatoid arthritis and atherosclerosis. However, recent studies have shown that NLRP3 has anti-tumor effects that inhibit tumor growth and metastasis.
- NLRP3 protein recognizes pathogen-related molecular patterns (PAMP) or endogenous damage-related molecular patterns (DAMP), its NOD domain oligomerizes and recruits proteins such as ASC and pro-caspase-1 to form functional NLRP3 Inflammatory bodies.
- pro-caspase-1 is cleaved and activated to caspase-1
- caspase-1 cleaves pro-IL-1 ⁇ and pro-IL-18 in a large amount to convert them into active forms of IL-1 ⁇ and IL-18 and release them to Extracellular, amplify the inflammatory response.
- NLRP3 inflammasome can significantly increase the levels of immune factors IL-1 ⁇ and IL-18 in the tumor microenvironment, initiate natural immune killing and subsequent adaptive immune responses to exert its anti-tumor effects.
- IL-1 ⁇ can induce CD8+ T cells to secrete interferon ⁇ (IFN- ⁇ ) and CD4+ cells to secrete IL-17, leading to effective anti-tumor immune effects; while IL-18 can promote the maturation of NK cells Activates the downstream signaling pathway of STAT1 in immune cells and enhances the killing function of immune cells.
- IFN- ⁇ interferon ⁇
- IL-18 can promote the maturation of NK cells Activates the downstream signaling pathway of STAT1 in immune cells and enhances the killing function of immune cells.
- Clinical studies have shown that the down-regulation of NLRP3 is significantly negatively correlated with the prognosis of liver cancer patients.
- NLRP3-deficient mice have a higher rate of colorectal tumor formation and worsen liver metastasis of colorectal cancer. It can be seen that NLRP3 plays an important role in the tumor microenvironment and can be used as a key target of tumor immunotherapy, as well as a tumor prognostic marker.
- NLRP3 modulators have the potential for tumor immunotherapy, and currently known compound BMS-986299 is in clinical phase I study. Therefore, it is necessary to develop new, high-efficiency and low-toxicity NLRP3 agonists to meet the needs of clinical treatment.
- NLRP3 modulators such as agonists
- directly binding or modifying NLRP3 at the protein level and by activating, stabilizing, and changing NLRP3 Distribution or other ways to enhance the function of NLRP3 inflammasome, thereby providing the following invention.
- the present invention provides compounds having the structure represented by formula X, stereoisomers, tautomers or mixtures thereof, pharmaceutically acceptable salts, co-crystals, polymorphs of the compounds Forms or solvates, or stable isotope derivatives, metabolites or prodrugs of said compounds:
- L is -(L 1 ) n -(L 2 ) p -(L 3 ) q -, wherein L 1 , L 2 and L 3 are the same or different and are each independently selected from C 1-8 alkylene, C 2 -8 alkenylene, C 2-8 alkynylene, C 1-8 alkyleneoxy, C 3-8 cycloalkylene, 4-10 membered heterocyclylene, C 6-12 arylene, 5 -10 membered heteroarylene, O, S, NR 33 , S(O), S(O) 2 , C(O) and C(R 36a R 36b ), wherein the C 1-8 alkylene group, C 2-8 alkenylene, C 2-8 alkynylene, C 1-8 alkyleneoxy, C 3-8 cycloalkylene, 4-10 membered heterocyclylene, C 6-12 arylene And 5-10 membered heteroarylene groups are each optionally substituted with one or more of the following substituents: halogen, OH
- n, p and q are each independently 0, 1 or 2;
- R 30 , R 37 , R 39 and R 40 are each independently selected from H, C 1-8 alkyl (e.g. C 1-6 alkyl or C 1-4 alkyl), C 1-8 alkoxy (e.g. C 1- 6 alkoxy or C 1-4 alkoxy), C 3-8 cycloalkyl, 4-10 membered heterocyclyl, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 alkyl-(5-10 membered heteroaryl), wherein the C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 Alkyl-(5-10 membered heteroaryl) are each optionally substituted with one or more of the following substituents: OH, CN, NO 2
- R 31 , R 32 , R 33 and R 34 are each independently selected from H, C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl and 5-10 membered heteroaryl, or R 31 and R 32 together with the N atom to which they are attached form a 4-8 membered heterocyclic group, or R 33 and R 34 and the C to which they are each attached Together with the N atom to form a 4-8 membered heterocyclic group, wherein the C 1-8 alkyl group, C 1-8 alkoxy group, C 3-8 cycloalkyl group, 4-8 membered heterocyclic group, 4-10
- Each of the membered heterocyclic group, C 6-12 aryl group and 5-10 membered heteroaryl group is optionally substituted by one or more of the following substituents: OH, CN, halogen, NO 2 , C 1-4 alkyl , C 1-4 alkoxy, C
- R 35 is selected from C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 alkyl-(5-10 membered heteroaryl), wherein the C 1-8 alkyl, C 1-8 alkoxy , C 3-8 cycloalkyl, 4-10 membered heterocyclyl, C 6-12 aryl and 5-10 membered heteroaryl are each optionally substituted by one or more of the following substituents: OH, CN , NO 2 , C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, halogen, C 1-4 haloalkoxy, CO 2 (C 1-6 alkyl), CONR 31 R 32 , NR 31 R 32 , NR 33 C(O)R 34 , S(O)Me, S(O
- R 36a and R 36b are the same or different and are each independently selected from H, C 1-8 alkyl and C 1-8 alkoxy, wherein the C 1-8 alkyl and C 1-8 alkoxy are each independently Optionally substituted by one or more of the following groups: OH, CN, halogen, NH 2 , NHCH 3 and N(CH 3 ) 2 , or R 36a and R 36b together with the C atom to which they are attached form a 3- 7-membered cycloalkyl or heterocyclic group;
- R 38 is selected from H, OH, CN, NO 2 , S(O)R 35 and S(O) 2 R 35 ;
- each R 30 may be the same or different;
- each R 31 may be the same or different;
- each R 32 may be the same or different;
- each R 33 may be the same or different;
- each R 34 may be the same or different;
- each R 35 may be the same or different;
- each R 37 may be the same or different;
- each R 38 may be the same or different;
- each R 39 may be the same or different;
- each R 40 may be the same or different;
- R is selected from among them:
- R 1 is selected from C 1-8 alkyl, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl and 9-12 membered aryl and hetero Cyclic group, wherein the C 1-8 alkyl group, C 3-8 cycloalkyl group, 4-10 membered heterocyclic group, C 6-12 aryl group, 5-10 membered heteroaryl group and 9-12 membered aryl group
- the heterocyclic groups are each optionally substituted by one or more of the following substituents: halogen, CN, NO 2 , C 1-4 alkyl, C 3-8 cycloalkyl, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-10 membered heterocyclyl, C 6-12 aryl, 5-10 membered heteroaryl, 9-12 membered heterocyclyl, and aryl group, CO 2 R 30, C ( O) R 30 , C(O)NR 31 R
- R 2 is selected from H, NR 41a R 41b , C 1-8 alkyl, C 2-8 alkenyl, C 2-8 alkynyl, C 3-8 cycloalkyl and 4-10 membered heterocyclic group, wherein said C 1- 8 alkyl, C 2-8 alkenyl, C 2-8 alkynyl, C 3-8 cycloalkyl and 4-10 membered heterocyclyl group each optionally substituted with a group of the following or One substitution: halogen, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-7 membered heterocyclic group, CN, NO 2 , OR 37 , SR 37 , C(O)R 30 , C(O) NR 31 R 32 , NR 33 C(O)R 34 , C(O)OR 30 , OC(O)R 30 , OC(O)NR 31 R 32 , NR 33 C(O)NR 31 R 32 and NR 31 R 32 ;
- R 5 is absent or selected from halogen, C 1-6 alkyl, C 1-4 alkoxy, C 3-8 cycloalkyl and 4-10 membered heterocyclic group, wherein the C 1-6 alkyl, C 1-4 alkoxy, C 3-8 cycloalkyl, and 4-10 membered heterocyclic group are each optionally substituted with one or more of the following groups: halogen, OH, CN, C 1-4 alkane Oxy, C 1-4 hydroxyalkyl and NR 31 R 32 ;
- n 0, 1 or 2, preferably 0 or 1;
- X 1 is selected from CR 6 and N;
- R 4 is selected from H, NR 41a R 41b , C 1-15 alkyl, C 1-8 alkoxy, C 2-8 alkenyl, C 2-8 alkynyl, C 3-8 cycloalkyl and 4- 10-membered heterocyclic group, wherein the C 1-15 alkyl group, C 1-8 alkoxy group, C 2-8 alkenyl group, C 2-8 alkynyl group, C 3-8 cycloalkyl group and 4-10 membered
- the heterocyclic groups are each optionally substituted by one or more of the following substituents: halogen, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-7 membered heterocyclyl, CN, NO 2 , OR 37 , SR 37 , C(O)R 30 , C(O)NR 31 R 32 , NR 33 C(O)R 34 , C(O)OR 30 , OC(O)R 30 , OC(O)NR 31 R 32 , NR 33
- R 6 is selected from H, halogen, C 1-6 alkyl, C 1-4 alkoxy, C 3-8 cycloalkyl and 4-10 membered heterocyclic group, wherein the C 1-6 alkyl, C 1-4 alkoxy, C 3-8 cycloalkyl, and 4-10 membered heterocyclic group are each optionally substituted with one or more of the following groups: halogen, OH, CN, C 1-4 alkoxy Group, C 1-4 hydroxyalkyl group and NR 31 R 32 ;
- R 41a and R 41b are each independently selected from H, C 1-6 alkyl, C 1-6 alkoxy and C 3-8 cycloalkyl, or R 41a and R 41b form together with the N atom to which they are attached 4-7 membered heterocyclic group, wherein the C 1-6 alkyl group, C 1-6 alkoxy group, C 3-8 cycloalkyl group and 4-7 membered heterocyclic group are each optionally selected from the following groups One or more of the substitutions: OH, CN and NR 31 R 32 .
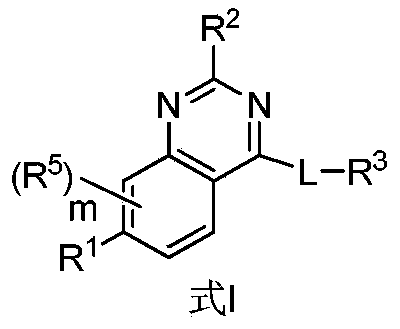
- the present invention provides a compound having a structure represented by formula I, a stereoisomer, a tautomer or a mixture of the compound, a pharmaceutically acceptable salt, co-crystal, Polymorphs or solvates, or stable isotope derivatives, metabolites or prodrugs of said compounds:
- R 1 is selected from C 1-8 alkyl, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl and 9-12 membered aryl and hetero Cyclic group, wherein the C 1-8 alkyl group, C 3-8 cycloalkyl group, 4-10 membered heterocyclic group, C 6-12 aryl group, 5-10 membered heteroaryl group and 9-12 membered aryl group
- the heterocyclic groups are each optionally substituted by one or more of the following substituents: halogen, CN, NO 2 , C 1-4 alkyl, C 3-8 cycloalkyl, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-10 membered heterocyclyl, C 6-12 aryl, 5-10 membered heteroaryl, 9-12 membered heterocyclyl, and aryl group, CO 2 R 30, C ( O) R 30 , C(O)NR 31 R
- R 2 is selected from H, NR 41a R 41b , C 1-8 alkyl, C 2-8 alkenyl, C 2-8 alkynyl, C 3-8 cycloalkyl and 4-10 membered heterocyclic group, wherein said C 1- 8 alkyl, C 2-8 alkenyl, C 2-8 alkynyl, C 3-8 cycloalkyl and 4-10 membered heterocyclyl group each optionally substituted with a group of the following or One substitution: halogen, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-7 membered heterocyclic group, CN, NO 2 , OR 37 , SR 37 , C(O)R 30 , C(O) NR 31 R 32 , NR 33 C(O)R 34 , C(O)OR 30 , OC(O)R 30 , OC(O)NR 31 R 32 , NR 33 C(O)NR 31 R 32 and NR 31 R 32 ;
- R 5 is absent or selected from halogen, C 1-6 alkyl, C 1-4 alkoxy, C 3-8 cycloalkyl and 4-10 membered heterocyclic group, wherein the C 1-6 alkyl, C 1-4 alkoxy, C 3-8 cycloalkyl, and 4-10 membered heterocyclic group are each optionally substituted with one or more of the following groups: halogen, OH, CN, C 1-4 alkane Oxy, C 1-4 hydroxyalkyl and NR 31 R 32 ;
- n 0, 1 or 2, preferably 0 or 1;
- L is -(L 1 ) n -(L 2 ) p -(L 3 ) q -, wherein L 1 , L 2 and L 3 are the same or different and each is independently selected from C 1-8 sub Alkyl, C 2-8 alkenylene, C 2-8 alkynylene, C 1-8 alkyleneoxy, C 3-8 cycloalkylene, 4-10 membered heterocyclylene, C 6-12 Arylene, 5-10 membered heteroarylene, O, S, NR 33 , S(O), S(O) 2 , C(O) and C(R 36a R 36b ), wherein the C 1- 8 alkylene, C 2-8 alkenylene, C 2-8 alkynylene, C 1-8 alkyleneoxy, C 3-8 cycloalkylene, 4-10 membered heterocyclylene, C 6
- the -12 arylene group and the 5-10 membered heteroarylene group are each optionally substituted with one or more of the following substituents: hal
- n, p, and q are each independently 0, 1 or 2 each time they appear;
- R 30 , R 37 , R 39 and R 40 are each independently selected from H, C 1-8 alkyl (e.g. C 1-6 alkyl or C 1-4 alkyl), C 1-8 alkoxy (e.g. C 1- 6 alkoxy or C 1-4 alkoxy), C 3-8 cycloalkyl, 4-10 membered heterocyclyl, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 alkyl-(5-10 membered heteroaryl), wherein the C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 Alkyl-(5-10 membered heteroaryl) are each optionally substituted with one or more of the following substituents: OH, CN, NO 2
- R 31 , R 32 , R 33 and R 34 are each independently selected from H, C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl and 5-10 membered heteroaryl, or R 31 and R 32 together with the N atom to which they are attached form a 4-8 membered heterocyclic group, or R 33 and R 34 and the C to which they are each attached Together with the N atom to form a 4-8 membered heterocyclic group, wherein the C 1-8 alkyl group, C 1-8 alkoxy group, C 3-8 cycloalkyl group, 4-8 membered heterocyclic group, 4-10
- Each of the membered heterocyclic group, C 6-12 aryl group and 5-10 membered heteroaryl group is optionally substituted by one or more of the following substituents: OH, CN, halogen, NO 2 , C 1-4 alkyl , C 1-4 alkoxy, C
- R 35 is selected from C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 alkyl-(5-10 membered heteroaryl), wherein the C 1-8 alkyl, C 1-8 alkoxy , C 3-8 cycloalkyl, 4-10 membered heterocyclyl, C 6-12 aryl and 5-10 membered heteroaryl are each optionally substituted by one or more of the following substituents: OH, CN , NO 2 , C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, halogen, C 1-4 haloalkoxy, CO 2 (C 1-6 alkyl), CONR 31 R 32 , NR 31 R 32 , NR 33 C(O)R 34 , S(O)Me, S(O
- R 36a and R 36b are the same or different and are each independently selected from H, C 1-8 alkyl and C 1-8 alkoxy, wherein the C 1-8 alkyl and C 1-8 alkoxy are each independently Optionally substituted by one or more of the following groups: OH, CN, halogen, NH 2 , NHCH 3 and N(CH 3 ) 2 , or R 36a and R 36b together with the C atom to which they are attached form a 3- 7-membered cycloalkyl or heterocyclic group;
- R 38 is selected from H, OH, CN, NO 2 , S(O)R 35 and S(O) 2 R 35 ;
- R 41a and R 41b are each independently selected from H, C 1-6 alkyl, C 1-6 alkoxy and C 3-8 cycloalkyl, or R 41a and R 41b form together with the N atom to which they are attached 4-7 membered heterocyclic group, wherein the C 1-6 alkyl group, C 1-6 alkoxy group, C 3-8 cycloalkyl group and 4-7 membered heterocyclic group are each optionally selected from the following groups One or more of the substitutions: OH, CN and NR 31 R 32 ; and
- each R 30 may be the same or different;
- each R 31 may be the same or different;
- each R 32 may be the same or different;
- each R 33 may be the same or different;
- each R 34 may be the same or different;
- each R 35 may be the same or different;
- each R 37 may be the same or different;
- each R 38 may be the same or different;
- each R 39 may be the same or different;
- each R 40 may be the same or different.
- the present invention provides a compound having a structure represented by Formula II, a stereoisomer, a tautomer or a mixture of the compound, a pharmaceutically acceptable salt, co-crystal, Polymorphs or solvates, or stable isotope derivatives, metabolites or prodrugs of said compounds:
- R 1 , L 2 and R 3 are as defined in formula I.
- the present invention provides a compound having a structure represented by Formula II-A, a stereoisomer, a tautomer or a mixture of the compound, a pharmaceutically acceptable salt, or a mixture of the compound Crystals, polymorphs or solvates, or stable isotope derivatives, metabolites or prodrugs of said compounds:
- the present invention provides a compound having a structure represented by formula III, a stereoisomer, a tautomer or a mixture of the compound, a pharmaceutically acceptable salt, co-crystal, Polymorphs or solvates, or stable isotope derivatives, metabolites or prodrugs of said compounds:
- X 1 is selected from CR 6 and N,
- R 1 is selected from C 1-8 alkyl, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl and 9-12 membered aryl and hetero Cyclic group, wherein the C 1-8 alkyl group, C 3-8 cycloalkyl group, 4-10 membered heterocyclic group, C 6-12 aryl group, 5-10 membered heteroaryl group and 9-12 membered aryl group
- the heterocyclic groups are each optionally substituted by one or more of the following substituents: halogen, CN, NO 2 , C 1-4 alkyl, C 3-8 cycloalkyl, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-10 membered heterocyclyl, C 6-12 aryl, 5-10 membered heteroaryl, 9-12 membered heterocyclyl, and aryl group, CO 2 R 30, C ( O) R 30 , C(O)NR 31 R
- R 4 is selected from H, NR 41a R 41b , C 1-15 alkyl, C 1-8 alkoxy, C 2-8 alkenyl, C 2-8 alkynyl, C 3-8 cycloalkyl and 4- 10-membered heterocyclic group, wherein the C 1-15 alkyl group, C 1-8 alkoxy group, C 2-8 alkenyl group, C 2-8 alkynyl group, C 3-8 cycloalkyl group and 4-10 membered
- the heterocyclic groups are each optionally substituted by one or more of the following substituents: halogen, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-7 membered heterocyclyl, CN, NO 2 , OR 37 , SR 37 , C(O)R 30 , C(O)NR 31 R 32 , NR 33 C(O)R 34 , C(O)OR 30 , OC(O)R 30 , OC(O)NR 31 R 32 , NR 33
- R 6 is selected from H, halogen, C 1-6 alkyl, C 1-4 alkoxy, C 3-8 cycloalkyl and 4-10 membered heterocyclic group, wherein the C 1-6 alkyl, C 1-4 alkoxy, C 3-8 cycloalkyl, and 4-10 membered heterocyclic group are each optionally substituted with one or more of the following groups: halogen, OH, CN, C 1-4 alkoxy Group, C 1-4 hydroxyalkyl group and NR 31 R 32 ;
- L is -(L 1 ) n -(L 2 ) p -(L 3 ) q -, wherein L 1 , L 2 and L 3 are the same or different and each is independently selected from C 1-8 sub Alkyl, C 2-8 alkenylene, C 2-8 alkynylene, C 1-8 alkyleneoxy, C 3-8 cycloalkylene, 4-10 membered heterocyclylene, C 6-12 Arylene, 5-10 membered heteroarylene, O, S, NR 33 , S(O), S(O) 2 , C(O) and C(R 36a R 36b ), wherein the C 1- 8 alkylene, C 2-8 alkenylene, C 2-8 alkynylene, C 1-8 alkyleneoxy, C 3-8 cycloalkylene, 4-10 membered heterocyclylene, C 6
- the -12 arylene group and the 5-10 membered heteroarylene group are each optionally substituted with one or more of the following substituents: hal
- n, p, and q are each independently 0, 1 or 2 each time they appear;
- R 30 , R 37 , R 39 and R 40 are each independently selected from H, C 1-8 alkyl (e.g. C 1-6 alkyl or C 1-4 alkyl), C 1-8 alkoxy (e.g. C 1- 6 alkoxy or C 1-4 alkoxy), C 3-8 cycloalkyl, 4-10 membered heterocyclyl, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 alkyl-(5-10 membered heteroaryl), wherein the C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 Alkyl-(5-10 membered heteroaryl) are each optionally substituted with one or more of the following substituents: OH, CN, NO 2
- R 31 , R 32 , R 33 and R 34 are each independently selected from H, C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl and 5-10 membered heteroaryl, or R 31 and R 32 together with the N atom to which they are attached form a 4-8 membered heterocyclic group, or R 33 and R 34 and the C to which they are each attached Together with the N atom to form a 4-8 membered heterocyclic group, wherein the C 1-8 alkyl group, C 1-8 alkoxy group, C 3-8 cycloalkyl group, 4-8 membered heterocyclic group, 4-10
- Each of the membered heterocyclic group, C 6-12 aryl group and 5-10 membered heteroaryl group is optionally substituted by one or more of the following substituents: OH, CN, halogen, NO 2 , C 1-4 alkyl , C 1-4 alkoxy, C
- R 35 is selected from C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 alkyl-(5-10 membered heteroaryl), wherein the C 1-8 alkyl, C 1-8 alkoxy , C 3-8 cycloalkyl, 4-10 membered heterocyclyl, C 6-12 aryl and 5-10 membered heteroaryl are each optionally substituted by one or more of the following substituents: OH, CN , NO 2 , C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, halogen, C 1-4 haloalkoxy, CO 2 (C 1-6 alkyl), CONR 31 R 32 , NR 31 R 32 , NR 33 C(O)R 34 , S(O)Me, S(O
- R 36a and R 36b are the same or different and are each independently selected from H, C 1-8 alkyl and C 1-8 alkoxy, wherein the C 1-8 alkyl and C 1-8 alkoxy are each independently Optionally substituted by one or more of the following groups: OH, CN, halogen, NH 2 , NHCH 3 and N(CH 3 ) 2 , or R 36a and R 36b together with the C atom to which they are attached form a 3- 7-membered cycloalkyl or heterocyclic group;
- R 38 is selected from H, OH, CN, NO 2 , S(O)R 35 and S(O) 2 R 35 ;
- R 41a and R 41b are each independently selected from H, C 1-6 alkyl, C 1-6 alkoxy and C 3-8 cycloalkyl, or R 41a and R 41b form together with the N atom to which they are attached 4-7 membered heterocyclic group, wherein the C 1-6 alkyl group, C 1-6 alkoxy group, C 3-8 cycloalkyl group and 4-7 membered heterocyclic group may optionally be selected from the following groups One or more of the substitutions: OH, CN and NR 31 R 32 ; and
- each R 30 may be the same or different;
- each R 31 may be the same or different;
- each R 32 may be the same or different;
- each R 33 may be the same or different;
- each R 34 may be the same or different;
- each R 35 may be the same or different;
- each R 37 may be the same or different;
- each R 38 may be the same or different;
- each R 39 may be the same or different;
- each R 40 may be the same or different.
- the present invention provides a compound having a structure represented by formula IV, stereoisomers, tautomers or mixtures thereof, pharmaceutically acceptable salts, co-crystals, Polymorphs or solvates, or stable isotope derivatives, metabolites or prodrugs of said compounds:
- R 1 , L 2 and R 3 are as defined in formula III.
- the present invention provides a compound having a structure represented by formula IV-A, a stereoisomer, a tautomer of the compound, or a mixture thereof, a pharmaceutically acceptable salt, or a mixture of the compound Crystals, polymorphs or solvates, or stable isotope derivatives, metabolites or prodrugs of said compounds:
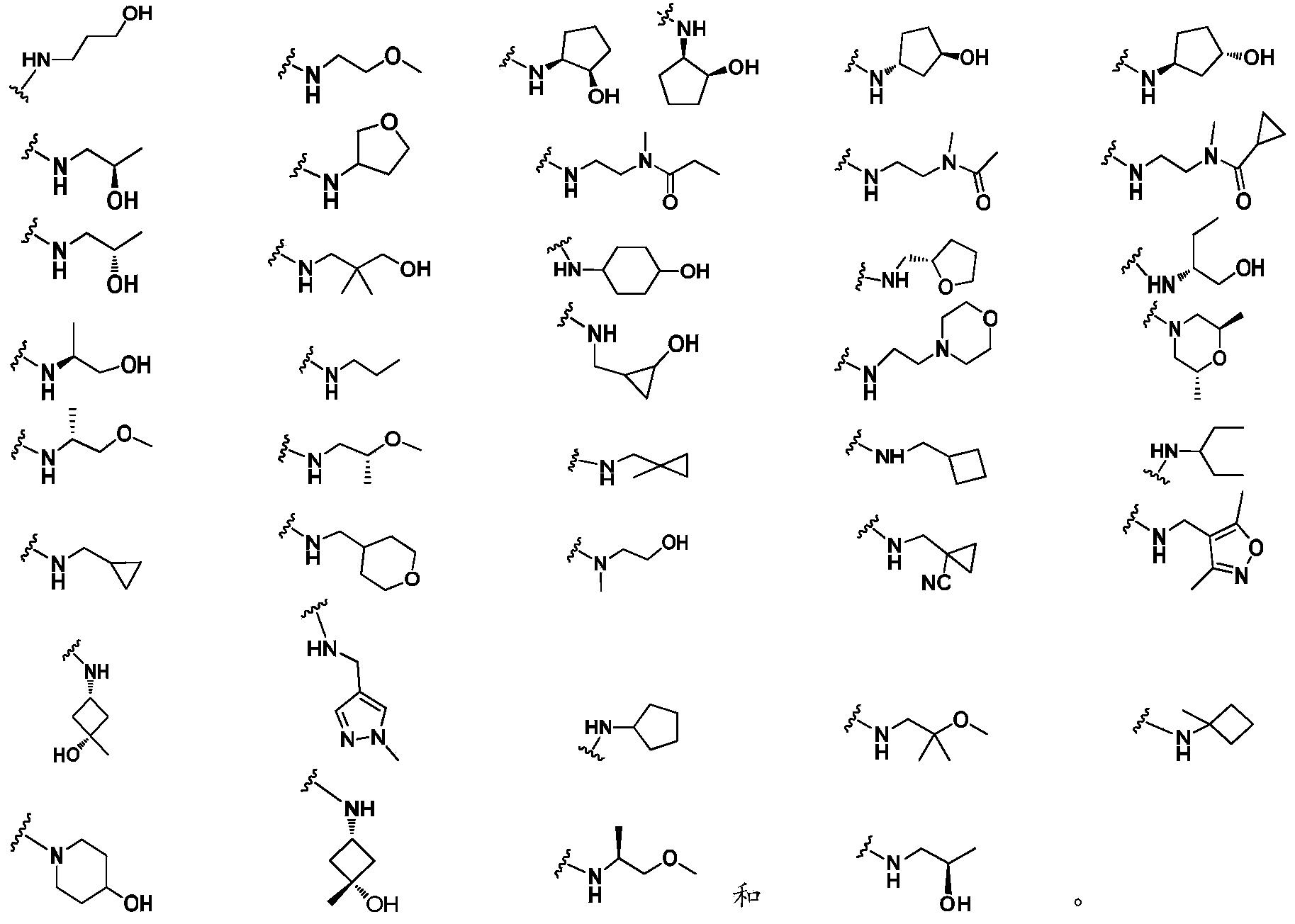
- R 3 is preferably independently selected from H, halogen, CN, C 1-6 alkyl, C 3-6 cycloalkyl, 4-7 membered heterocyclic group, C 6 each occurrence -10 aryl, 5-6 membered heteroaryl, CO 2 R 30 , C(O)NR 31 R 32 , NR 33 C(O)R 34 , NR 31 R 32 , S(O) 2 R 35 , OR 37 , SR 37 , C(O)R 30 , OC(O)R 30 , NR 33 C(O)NR 31 R 32 and S(O) 2 NR 31 R 32 , wherein the C 1-6 alkyl group, C 3-6 cycloalkyl, 4-7 membered heterocyclyl, C 6-10 aryl and 5-6 membered heteroaryl are each optionally substituted with one or more of the following substituents: halogen, CN, C 1-4 alkyl, C 3-6 cycloalkyl, C 1-4
- each occurrence of R 3 is independently selected from H, CN, C 1-6 alkyl, C 3-6 cycloalkyl, 4-7 membered heterocyclyl, C 6-10 aryl, 5- 6-membered heteroaryl, NR 33 C(O)R 34 , OR 37 , SR 37 , C(O)R 30 and OC(O)R 30 , wherein the C 1-6 alkyl group, C 3-6 ring
- the alkyl group, 4-7 membered heterocyclic group, C 6-10 aryl group and 5-6 membered heteroaryl group are each optionally substituted with one or more of the following substituents: CN, C 1-4 alkyl and CO 2 R 30 , R 30 and R 37 are each independently selected from H, C 1-6 alkyl, C 1-6 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6 -12 aryl group and 5-10 membered heteroaryl group, wherein the C 1-6 alkyl group
- R 3 is independently selected from H, OH, CN, N(CH 3 )C(O)CH 3 , N(CH 3 )C(O)CH 2 CH 3 , N(CH 3 ) C(O)-cyclopropyl and the following groups each optionally substituted with one or more of hydroxy, methyl and CN: methyl, ethyl, propyl, cyclopropyl, cyclobutyl , Cyclopentyl, tetrahydrofuranyl, morpholino, tetrahydropyranyl, isoxazolyl, pyrazolyl and piperidinyl.
- L does not exist or is -L 1 -L 2 -, wherein L 1 is NH or N(CH 3 ), and L 2 does not exist or is selected from C 1-4 alkylene , C 1-4 alkyleneoxy and C 3-6 cycloalkylene.
- L does not exist or is -L 1 -L 2 -, where L 1 is NH or N(CH 3 ), and L 2 does not exist or is selected from methylene, ethylene , Propylene, butylene, methyleneoxy, ethyleneoxy, n-propoxy, isopropoxy, n-butoxy, isobutoxy, tert-butoxy, ring Propyl, cyclobutylene, cyclopentylene and cyclohexylene.
- -LR 3 or -NH-L 2 -R 3 is preferably selected from:
- the compounds of the present invention and pharmaceutically acceptable salts thereof include, but are not limited to:
- composition preparation and use
- the present invention provides a pharmaceutical composition
- a pharmaceutical composition comprising a compound as described above, a stereoisomer, a tautomer of the compound, or a mixture thereof, a pharmaceutically acceptable salt, or a mixture of the compound Crystals, polymorphs or solvates, or stable isotope derivatives, metabolites or prodrugs of said compounds.
- the pharmaceutical composition further comprises one or more pharmaceutically acceptable carriers.
- the pharmaceutical composition is used to prevent and/or treat diseases related to NLRP3 inflammasome activity (e.g., tumor diseases).
- diseases related to NLRP3 inflammasome activity e.g., tumor diseases.
- the present invention provides a pharmaceutical preparation, which comprises the compound as described above, a stereoisomer, a tautomer of the compound, or a mixture thereof, a pharmaceutically acceptable salt, a co-crystal of the compound , Polymorphs or solvates, or stable isotope derivatives, metabolites or prodrugs of the compound, or comprise the above-mentioned pharmaceutical composition.
- the present invention provides a compound as described above, a stereoisomer, a tautomer or a mixture of the compound, a pharmaceutically acceptable salt, co-crystal, and polymorph of the compound Or solvates, stable isotope derivatives, metabolites or prodrugs of the compounds, or the use of the above-mentioned pharmaceutical composition for the preparation of medicines for the prevention and/or treatment of NLRP3 inflammasomes Activity-related diseases (e.g. tumor diseases).
- NLRP3 inflammasomes Activity-related diseases (e.g. tumor diseases).
- the present invention provides a compound as described above, a stereoisomer, a tautomer or a mixture of the compound, a pharmaceutically acceptable salt, co-crystal, and polymorph of the compound Or solvates, stable isotope derivatives, metabolites or prodrugs of the compounds, or pharmaceutical compositions as described above, which are used to prevent and/or treat diseases related to NLRP3 inflammasome activity (such as tumor diseases) ).
- the present invention provides a compound as described above, a stereoisomer, a tautomer or a mixture of the compound, a pharmaceutically acceptable salt, co-crystal, and polymorph of the compound Or solvates, stable isotope derivatives, metabolites or prodrugs of the compounds, or the use of the pharmaceutical composition as described above for the preparation of preparations for regulating (for example, increasing) NLRP3 inflammasome active.
- the formulation is administered to a subject (e.g., a mammal, including, for example, bovine, equine, ovine, swine, canine, feline, rodent, Primates (such as humans) in vivo to increase the NLRP3 inflammasome activity in the cells of the subject.
- a subject e.g., a mammal, including, for example, bovine, equine, ovine, swine, canine, feline, rodent, Primates (such as humans) in vivo to increase the NLRP3 inflammasome activity in the cells of the subject.
- cells in vitro e.g., cell lines or cells from a subject
- the present invention provides a method for modulating (e.g. increasing) the activity of NLRP3 inflammasome in a cell, which comprises administering to the cell an effective amount of a compound as described above, stereoisomers, tautomers of the compound Isomers or mixtures thereof, pharmaceutically acceptable salts, co-crystals, polymorphs or solvates of said compounds, stable isotope derivatives, metabolites or prodrugs of said compounds, drugs as described above Composition, or pharmaceutical formulation as described above.
- the present invention provides a kit for modulating (for example, increasing) the activity of NLRP3 inflammasomes, the kit comprising a compound as described above, stereoisomers and tautomers of the compound Or a mixture thereof, a pharmaceutically acceptable salt, co-crystal, polymorph or solvate of the compound, a stable isotope derivative, metabolite or prodrug of the compound, the pharmaceutical composition as described above, Or a pharmaceutical formulation as described above.
- the present invention provides a method for treating and/or preventing diseases (such as tumor diseases) associated with NLRP3 inflammasome activity, which comprises administering to a subject in need thereof a therapeutically and/or preventively effective amount of the above
- diseases such as tumor diseases
- the tumor diseases include, but are not limited to: brain tumor, lung cancer, squamous cell carcinoma, bladder cancer, gastric cancer, ovarian cancer, peritoneal cancer, pancreatic cancer, breast cancer, head and neck cancer, cervical cancer, intrauterine cancer Membranous cancer, rectal cancer, liver cancer, kidney cancer, esophageal adenocarcinoma, esophageal squamous cell carcinoma, prostate cancer, female reproductive tract cancer, carcinoma in situ, lymphoma, neurofibroma, thyroid cancer, bone cancer, skin cancer, brain Cancer, colon cancer, testicular cancer, gastrointestinal stromal tumor, prostate tumor, mast cell tumor, multiple myeloma, melanoma, glioma or sarcoma.
- the compounds of the invention are NLRP3 full agonists. In some embodiments, the compounds of the invention are NLRP3 partial agonists.
- agonist refers to a compound that binds to a receptor and activates it to trigger a downstream biological effect or response, including full agonist and partial agonist.
- Full agonists can activate the receptor and produce the greatest effect (maximal effect or E max ).
- a partial agonist can bind to the receptor and activate it, but compared to a full agonist, it only produces a partial effect.
- the partial agonist can sometimes become a partial antagonist by competing with the full agonist for binding sites on the receptor or by other mechanisms.
- Efficacy potential, when by EC 50 (producing 50% of the concentration of compound E max) measure) a possible partial agonist potency higher or lower than a full agonist.
- the NLRP3 agonists of the present invention include NLRP3 full agonists and NLRP3 partial agonists.
- NLRP3 NLR family pyrin domain containing 3, which is an inflammasome.
- the meaning includes NLRP3 nucleic acid, polynucleotide, oligonucleotide, sense and antisense polynucleotide chain, complementary sequence, short peptide, polypeptide , Proteins, homologous or heterologous molecules, subtypes, precursors, mutants, variants, derivatives, various splicing bodies, alleles, different species and activation fragments, etc.
- halo refers to substitution by a halogen atom, and the “halogen” includes F, Cl, Br, or I.
- alkyl refers to a linear or branched saturated aliphatic hydrocarbon group.
- C 1-15 alkyl C 1-8 alkyl
- C 1-6 alkyl C 1-6 alkyl
- C 1-4 alkyl respectively refer to having 1 to 15 carbon atoms, 1 to 8 Carbon atoms, linear or branched alkyl groups of 1 to 6 carbon atoms and 1-4 carbon atoms, such as methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl Group, tert-butyl, n-pentyl or n-hexyl.
- the alkyl group may be optionally substituted with one or more (such as 1 to 3) substituents which are the same or different.
- alkylene refers to a saturated divalent hydrocarbon group obtained by removing two hydrogen atoms from a linear or branched saturated hydrocarbon group, which contains the specified number of carbon atoms.
- C 1-8 alkylene refers to an alkylene group having 1 to 8 carbon atoms, such as methylene (-CH 2 -), ethylene (-CH 2 CH 2 -), isopropylidene Group (-CH(CH 3 )CH 2 -) and so on.
- the alkylene group may be optionally substituted with one or more (such as 1 to 3) substituents which are the same or different.
- haloalkyl refers to an alkyl group substituted with one or more (such as 1 to 3) identical or different halogen atoms.
- C 1-8 haloalkyl refers to having 1 to 8 carbon atoms, 1 to 6 carbon atoms and 1-4 carbon atoms, respectively For example, -CF 3 , -C 2 F 5 , -CHF 2 , -CH 2 F, -CH 2 CF 3 , -CH 2 Cl, -CH 2 CH 2 CF 3 and the like.
- hydroxyalkyl refers to a group formed by replacing the hydrogen atom of an alkyl group with one or more hydroxy groups, such as a C 1-4 hydroxyalkyl group or a C 1-3 hydroxyalkyl group, examples of which include but are not limited to hydroxy Methyl, hydroxyethyl, hydroxypropyl, hydroxybutyl, -CH(OH)CH 3 and so on.
- the alkenyl group may be optionally substituted with one or more (such as 1 to 3) substituents which are the same or different.
- the alkenylene group may be optionally substituted with one or more (such as 1 to 3) substituents which are the same or different.
- alkynyl refers to a monovalent linear or branched hydrocarbon group with one or more carbon-carbon triple bonds, including but not limited to ethynyl, 2-propynyl, 2-butynyl and 1,3-butynyl Dialkynyl and so on.
- the alkynyl group may be optionally substituted with one or more (such as 1 to 3) substituents which are the same or different.
- alkynylene refers to a divalent straight or branched chain hydrocarbon group with one or more carbon-carbon triple bonds, which contains the specified number of carbon atoms, such as 2 to 8 carbon atoms, including but not limited to Wait.
- the alkynylene group may be optionally substituted with one or more (such as 1 to 3) substituents which are the same or different.
- alkoxy means a group in which an oxygen atom is inserted at any reasonable position of an alkyl group (as defined above), for example, C 1-8 alkoxy, C 1-6 alkoxy, C 1-4 Alkoxy or C 1-3 alkoxy.
- Representative examples of C 1-6 alkoxy include, but are not limited to, methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, isobutoxy, tert-butoxy, pentoxy Group, hexyloxy group, -CH 2 -OCH 3 and so on.
- the alkoxy group may be optionally substituted with one or more (such as 1 to 3) substituents which are the same or different.
- alkyleneoxy refers to a divalent alkoxy group, such as -OCH 2 -, -OCH(CH 3 )CH 2 -, -OCH 2 CH 2 O-, -CH 2 CH 2 O-, etc.
- the alkyleneoxy group may be optionally substituted with one or more (such as 1 to 3) substituents which are the same or different.
- condensed ring or “fused ring” refers to a ring system formed by two or more ring structures sharing two adjacent atoms with each other.
- spirocyclic ring refers to a ring system formed by two or more cyclic structures sharing one ring atom with each other.
- bridged ring refers to a ring system formed by two or more ring structures sharing two atoms that are not directly connected to each other.
- cycloalkyl refers to saturated or unsaturated non-aromatic monocyclic or polycyclic (such as bicyclic) hydrocarbon ring groups, including but not limited to monocyclic alkyl groups (such as cyclopropyl, cyclobutyl, cyclopentyl, Cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl) and bicyclic alkyl groups, including spirocyclic, fused ring (fused ring) or bridged ring systems (ie, spirocyclic alkyl, fused ring (fused ring) alkyl And bridged cycloalkyl groups, such as bicyclo[1.1.1]pentyl, bicyclo[2.2.1]heptyl, etc.).
- monocyclic alkyl groups such as cyclopropyl, cyclobutyl, cyclopentyl, Cyclohexyl, cycloheptyl,
- the cycloalkyl group may be optionally substituted by one or more (such as 1 to 3) substituents which are the same or different.
- 3-7 membered cycloalkyl refers to a cycloalkyl group having 3 to 7 ring-forming carbon atoms, which may be a monocyclic alkyl group, such as cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl or cyclo
- the heptyl group may also be a bicyclic alkyl group, such as a C 5-7 spirocycloalkyl group, a C 5-7 bridged cycloalkyl group or a C 4-7 condensed cycloalkyl group.
- C 3-8 cycloalkyl refers to a cycloalkyl group having 3 to 8 ring-forming carbon atoms, such as a C 3-6 cycloalkyl group, which may be a monocyclic alkyl group, such as cyclopropyl, cyclobutyl , Cyclopentyl, cyclohexyl, cycloheptyl or cyclooctyl, it can also be a bicyclic alkyl group, such as C 3-8 spirocycloalkyl, C 3-8 bridged cycloalkyl, C 3-8 condensed cycloalkyl , C 3-6 spirocycloalkyl, C 3-6 bridged cycloalkyl or C 3-6 condensed cycloalkyl.
- cycloalkylene refers to a cycloalkyl group as defined herein, which has two monovalent group centers obtained by removing two hydrogen atoms from the same carbon atom or two different carbon atoms of the parent cycloalkyl group.
- Typical cycloalkylene groups include, but are not limited to, cyclopropylene, cyclobutylene, cyclopentylene, cyclohexylene, cycloheptylene, cyclooctylene, cyclononylidene, cyclohexenylene, etc. .
- aryl refers to an all-carbon monocyclic or fused polycyclic aromatic group having a conjugated ⁇ -electron system.
- C 6-12 aryl group means an aryl group containing 6 to 12 carbon atoms, such as a C 6-10 aryl group, and specific examples are phenyl or naphthyl.
- the aryl group is optionally substituted with one or more (such as 1 to 3) identical or different substituents (for example, halogen, OH, CN, NO 2 , C 1 -C 6 alkyl, etc.).
- arylene refers to an aryl group as defined herein, which has two monovalent group centers obtained by removing two hydrogen atoms from the same carbon atom or two different carbon atoms of the parent aryl group.
- Typical arylene groups include, but are not limited to, phenylene and naphthylene.
- aryl and cycloalkyl refers to a fused ring group formed by aryl and cycloalkyl (such as monocyclic alkyl) sharing two adjacent atoms with each other, wherein the point of attachment to other groups can be at the aromatic On the base or on the cycloalkyl group.
- 9-12 membered aryl and cycloalkyl refers to an aryl and cycloalkyl group containing 9-12 ring atoms in total, such as phenyl and cyclopentyl, phenyl and cyclohexyl, such as
- heterocyclylene refers to a heterocyclic group as defined herein, which has the same carbon atom or two different carbon atoms, one carbon atom and one heteroatom, or two heteroatoms removed from the parent heterocyclic group. Two monovalent group centers derived from a hydrogen atom.
- 3-14 membered heterocyclic group means a heterocyclic group containing 3-14 ring atoms, including but not limited to 4-10 membered heterocyclic group, 4-7 membered heterocyclic group, 5-6 membered heterocyclic group Group, 4-7 membered nitrogen-containing heterocyclic group, 4-7 membered oxygen-containing heterocyclic group, 4-7 membered sulfur-containing heterocyclic group, 5-6 membered nitrogen-containing heterocyclic group, 5-6 membered oxygen-containing heterocyclic group Group, 5-6 membered sulfur-containing heterocyclic group, etc., the "nitrogen-containing heterocyclic group", "oxygen-containing heterocyclic group” and "sulfur-containing heterocyclic group” each optionally further contain one or more Other heteroatoms of oxygen, nitrogen and sulfur.
- 3-14 membered heterocyclic groups include, but are not limited to, oxiranyl, aziridinyl, azetidinyl, oxetanyl, tetrahydrofuryl, pyrrolidinyl, pyrrolidonyl, imidazolidinyl, Pyrazolyl, tetrahydropyranyl, piperidinyl, morpholinyl, dithianyl, thiomorpholinyl, piperazinyl, trithianyl, Wait.
- the heterocyclic group also includes a fused ring structure, and the point of connection between the fused ring structure and other groups can be on any ring in the fused ring structure. Therefore, the heterocyclic group of the present invention also includes (but is not limited to) heterocyclic group and heterocyclic group, heterocyclic group and cycloalkyl group, single heterocyclic group and single heterocyclic group, single heterocyclic group and monocyclic alkyl group.
- heterocyclic groups also include bridged heterocyclic groups and spiro heterocyclic groups.
- bridged heterocyclyl refers to two saturated rings that share two ring atoms that are not directly connected and contain one or more (e.g., 1, 2, 3, or 4) heteroatoms (e.g., oxygen atoms, Nitrogen atom or sulfur atom), including but not limited to 7-10 membered heterocyclic group, 8-10 membered heterocyclic group, 7-10 membered nitrogen-containing bridged heterocyclic group, 7-10 membered oxygen-containing Bridged heterocyclic group, 7-10 membered sulfur-containing bridged heterocyclic group, etc., for example Wait.
- the "nitrogen-containing bridged heterocyclic group", "oxygen-containing bridged heterocyclic group” and “sulfur-containing bridged heterocyclic group” each optionally further contain one or more other heteroatoms selected from oxygen, nitrogen and sulfur.
- spiroheterocyclic group refers to two or more saturated rings that share one ring atom and contain one or more (e.g., 1, 2, 3, or 4) heteroatoms (e.g., oxygen atoms, Nitrogen atom, sulfur atom) ring structure, including but not limited to 5-10 membered spiro heterocyclic group, 6-10 membered spiro heterocyclic group, 6-10 membered nitrogen-containing spiro heterocyclic group, 6-10 membered oxygen-containing Spiro heterocyclic group, 6-10 membered sulfur-containing spiro heterocyclic group, etc., for example Wait.
- heteroatoms e.g., oxygen atoms, Nitrogen atom, sulfur atom
- nitrogen-containing spiro heterocyclic group each optionally further contain one or more other heteroatoms selected from oxygen, nitrogen and sulfur.
- oxygen-containing spiro heterocyclic group refers to a spiroheterocyclic group containing 6-10 ring atoms in total and at least one of which is a nitrogen atom.
- aryl and heterocyclic group refers to a cyclic group formed by an aryl group and a heterocyclic group sharing two adjacent carbon atoms with each other (wherein the aryl group and the heterocyclic group are as defined above), which is combined with other groups.
- the point of attachment of the group may be on an aryl group or a heterocyclic group.
- 9-12 membered aryl and heterocyclyl means an aryl and heterocyclyl group containing a total of 9-12 ring atoms, including but not limited to 9-10 membered Benzoheterocyclic group, such as benzo 5-8 membered heterocyclic group, such as benzo 5-6 membered heterocyclic group, such as benzo 5-6 membered monoheterocyclic group, benzo 5-6 membered nitrogen-containing monohetero Cyclic group, benzo 5-6 membered oxygen-containing monoheterocyclic group, benzo 5-6 membered sulfur-containing heterocyclic group, etc.
- Benzoheterocyclic group such as benzo 5-8 membered heterocyclic group
- benzo 5-6 membered heterocyclic group such as benzo 5-6 membered monoheterocyclic group, benzo 5-6 membered nitrogen-containing monohetero Cyclic group, benzo 5-6 membered oxygen-containing monoheterocyclic group, benzo 5-6 membere
- Each of the "nitrogen-containing heterocyclic group", “oxygen-containing heterocyclic group” and “sulfur-containing heterocyclic group” optionally further contains one or more other heteroatoms selected from oxygen, nitrogen and sulfur.
- aryl and heterocyclic groups include, but are not limited to: indazolyl,
- heteroaryl refers to a monocyclic or polycyclic aromatic group containing one or more identical or different heteroatoms, including monocyclic heteroaryl groups and aromatic groups containing at least one heteroaromatic ring (an aromatic group containing at least one heteroatom Ring system), which may have 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14 ring atoms, such as 5, 6, 7, 8, 9 or 10 Ring atom.
- the heteroatom may be oxygen, nitrogen or sulfur.
- heteroarylene refers to a heteroaryl group as described above, which has two hydrogen atoms removed from the same carbon atom or two different carbon atoms of the parent heteroaryl group or one hydrogen atom removed from the carbon atom And two monovalent group centers are obtained by removing one hydrogen atom from the nitrogen atom.
- 5-10 membered heteroaryl means a heteroaryl group containing 5 to 10 ring atoms, including 5-6 membered heteroaryl, 5-6 membered monoheteroaryl, 5-10 membered nitrogen-containing heteroaryl Group, 5-10 member oxygen-containing heteroaryl group, 5-10 member sulfur-containing heteroaryl group, 5-6 member nitrogen-containing heteroaryl group, 5-6 member oxygen-containing heteroaryl group, 5-6 member sulfur-containing heteroaryl group Group, 5-6 membered nitrogen-containing monoheteroaryl group, 5-6 membered oxygen-containing monoheteroaryl group, 5-6 membered sulfur-containing monoheteroaryl group.
- the “nitrogen-containing heteroaryl group”, “oxygen-containing heteroaryl group”, “sulfur-containing heteroaryl group”, “nitrogen-containing monoheteroaryl group”, “oxygen-containing monoheteroaryl group” and “sulfur-containing monoheteroaryl group” optionally further contains one or more other heteroatoms selected from oxygen, nitrogen, and sulfur.
- Examples of 5-10 membered heteroaryl groups include, but are not limited to, thienyl, furyl, pyrrolyl, oxazolyl, thiazolyl, imidazolyl, pyrazolyl, isoxazolyl, isothiazolyl, triazolyl, tetrazolyl, Azolyl, oxadiazolyl, thiadiazolyl, pyridyl, pyridazinyl, pyrimidinyl, pyrazinyl, triazinyl, etc., and 5-10 membered cyclic groups containing these groups.
- heteroaryl encompasses a bicyclic structure, that is, heteroaryl (e.g., monoheteroaryl) can be combined with aryl (e.g., monocyclic aryl, e.g., phenyl), heterocyclic group (e.g., monoheterocyclic group). Group), cycloalkyl (e.g. monocycloalkyl) or another heteroaryl group (e.g.
- Another monoheteroaryl sharing two adjacent atoms with each other to form a combined ring structure, which is the point of attachment to other groups
- It can be on any heteroaromatic ring or other ring, including but not limited to (mono)heteroaryl and (mono)heteroaryl, (mono)heteroaryl and (monocyclic)aryl, (mono)heteroaryl And (mono)heterocyclyl or (mono)heteroaryl and (mono)cycloalkyl, for example 5-6 membered (mono)heteroaryl and 5-6 membered (mono)heteroaryl, 5-6 membered ( Mono) heteroaryl phenyl, 5-6 membered (mono) heteroaryl and 5-6 membered (mono) heterocyclic group or 5-6 membered (mono) heteroaryl and C 4-6 (mono) ring Alkyl groups (e.g.
- heteroaryl encompasses aryl-containing fused ring structures, which are also referred to as "aryl and heteroaryl", which refers to aryl (e.g., monocyclic aryl, e.g., phenyl) and heteroaryl (e.g., monocyclic aryl) Heteroaryl groups, such as 5-6 membered mono-heteroaryl groups, can be connected to other groups on the aromatic ring or on the heteroaromatic ring.
- aryl and heteroaryl includes, but is not limited to, monocyclic aryl and monoheteroaryl.
- 9-12 membered aryl and heteroaryl group refers to an aryl and heteroaryl group containing a total of 9 to 12 ring atoms, such as a benzo 5-6 membered nitrogen-containing monoheteroaryl group.
- heteroaryl includes a cycloalkyl-containing bicyclic structure, which is also referred to as “heteroaryl and cycloalkyl", which refers to a heteroaryl group (such as a monoheteroaryl group, such as a 5-6 membered monoheteroaryl group). Group) and a cycloalkyl group (for example, a C 4-6 cycloalkyl group).
- a heteroaryl group such as a monoheteroaryl group, such as a 5-6 membered monoheteroaryl group).
- a cycloalkyl group for example, a C 4-6 cycloalkyl group.
- the point of connection with other groups can be on the heteroaromatic ring or on the cycloalkyl group.
- the "heteroaryl and cycloalkyl” includes, but is not limited to, monoheteroaryl and monocycloalkyl.
- 9-10 membered heteroaryl and cycloalkyl refers to a heteroaryl and cycloalkyl group containing a total of 9-10 ring atoms, such as 4-6 membered nitrogen-containing monoheteroaryl and C 4-6 mono Cycloalkyl.
- substitution means that one or more (for example, 1, 2, 3, or 4) hydrogens on the designated atom are replaced by a selection from the indicated group, provided that no more than the designated atom is The normal valence in the current situation and the substitution forms a stable compound. Combinations of substituents and/or variables are only permissible when such combinations form stable compounds.
- substituent can be (1) unsubstituted or (2) substituted. If the carbon of a substituent is described as being optionally substituted by one or more of the list of substituents, then one or more hydrogens on the carbon (to the extent of any hydrogens present) may each be independently selected optionally Substituent replacement. If the nitrogen of a substituent is described as being optionally substituted by one or more of the list of substituents, then one or more hydrogens on the nitrogen (to the extent of any hydrogens present) may each be independently selected optionally Substituent replacement.
- each substituent is selected independently of the other. Therefore, each substituent may be the same or different from another (other) substituent.
- one or more means one or more than one under reasonable conditions, such as two, three, four, five, six, seven, eight, nine Or 10.
- the point of attachment of a substituent can be from any suitable position of the substituent.
- the present invention also includes all pharmaceutically acceptable isotopic compounds of the compounds of the present invention, which are the same as the compounds of the present invention, except that one or more atoms have the same atomic number but the atomic mass or mass number is different from the one that is predominant in nature. Atomic substitution of atomic mass or mass number.
- isotopes suitable for inclusion in the compounds of the present invention include (but are not limited to) isotopes of hydrogen (e.g. 2 H, 3 H); isotopes of carbon (e.g. 11 C, 13 C, and 14 C); isotopes of chlorine (e.g.
- stable isotope derivative means that one or more atoms in the compound of the present invention are replaced by atoms having the same atomic number but whose atomic mass or mass number is different from the predominant atomic mass or mass number in nature. The stable compound.
- stereoisomer refers to an isomer formed by a compound containing at least one asymmetric center. In compounds with one or more (for example, 1, 2, 3, or 4) asymmetric centers, it can produce racemic mixtures, single enantiomers, diastereomeric mixtures, and Individual diastereomers. Certain individual molecules can also exist as geometric isomers (cis/trans).
- the compounds of the present invention may exist in a mixture of two or more different structural forms (commonly referred to as tautomers) in rapid equilibrium.
- Representative examples of tautomers include keto-enol tautomers, phenol-ketone tautomers, nitroso-oxime tautomers, imine-enamine tautomers Wait.
- nitroso-oximes can exist in equilibrium in the following tautomeric forms in solution:
- the pyrazole ring can exist in equilibrium in the following tautomeric forms:
- the compounds of the present invention are intended to be stereoisomers (including cis and trans isomers), optical isomers (such as R and S enantiomers), and diastereomers. , Geometric isomers, rotamers, conformational isomers, atropisomers or mixtures thereof.
- the compounds of the present invention can exhibit more than one type of isomerism, and are composed of mixtures thereof (e.g., racemic mixtures and diastereomeric pairs).
- the present invention covers all possible crystalline forms or polymorphs of the compounds of the present invention, which can be a single polymorph or a mixture of more than one polymorph in any ratio. It should also be understood that certain compounds of the present invention may exist in free form for treatment, or, when appropriate, in the form of their pharmaceutically acceptable derivatives.
- pharmaceutically acceptable derivatives include, but are not limited to: pharmaceutically acceptable salts, solvates, metabolites or prodrugs, which can be directly or indirectly administered to patients in need thereof.
- the compound of the present invention or its metabolite or residue is provided. Therefore, when the "compound of the present invention" is referred to herein, it is also intended to encompass the above-mentioned various derivative forms of the compound.
- the pharmaceutically acceptable salts of the compounds of the present invention include acid addition salts and base addition salts thereof.
- acid addition salts for example, hexafluorophosphate, meglumine salt and the like.
- suitable salts see “Handbook of Pharmaceutical Salts: Properties, Selection, and Use” by Stahl and Wermuth (Wiley-VCH, 2002).
- pharmaceutically acceptable carrier refers to a diluent, adjuvant, excipient or vehicle administered with the therapeutic agent, and it is suitable for contact with humans and/or within the scope of reasonable medical judgment Tissues of other animals without excessive toxicity, irritation, allergic reactions, or other problems or complications corresponding to a reasonable benefit/risk ratio.
- the pharmaceutically acceptable carriers that can be used in the pharmaceutical composition of the present invention include, but are not limited to, sterile liquids, such as water and oils, including those of petroleum, animal, vegetable, or synthetic origin, such as peanut oil, soybean oil, and mineral oil. Oil, sesame oil, etc.
- sterile liquids such as water and oils
- oils including those of petroleum, animal, vegetable, or synthetic origin, such as peanut oil, soybean oil, and mineral oil. Oil, sesame oil, etc.
- water is an exemplary carrier. It is also possible to use physiological saline and aqueous glucose and glycerol solutions as liquid carriers, especially for injections.
- Suitable pharmaceutical excipients include starch, glucose, lactose, sucrose, gelatin, maltose, chalk, silica gel, sodium stearate, glyceryl monostearate, talc, sodium chloride, skimmed milk powder, glycerin, propylene glycol, water, Ethanol, etc.
- the composition may also contain small amounts of wetting agents, emulsifiers or pH buffering agents as needed.
- Oral preparations may contain standard carriers such as pharmaceutical grades of mannitol, lactose, starch, magnesium stearate, sodium saccharin, cellulose, magnesium carbonate and the like. Examples of suitable pharmaceutically acceptable carriers are described in Remington's Pharmaceutical Sciences (1990).
- the pharmaceutical composition of the present invention can act systemically and/or locally.
- they can be administered by suitable routes, such as by injection, intravenous, intraarterial, subcutaneous, intraperitoneal, intramuscular or transdermal administration; or by oral, buccal, transnasal, transmucosal, topical, It is administered in the form of ophthalmic preparations or by inhalation.
- the pharmaceutical composition of the present invention can be administered in a suitable dosage form.
- the dosage form includes but not limited to tablet, capsule, lozenge, hard candy, powder, spray, cream, ointment, suppository, gel, paste, lotion, ointment, aqueous suspension , Injectable solutions, elixirs, syrups.
- an effective dose refers to the amount of a compound that will relieve one or more symptoms of the condition being treated to a certain extent after being administered.
- the dosage regimen can be adjusted to provide the best desired response. For example, a single bolus can be administered, several divided doses can be administered over time, or the dose can be proportionally reduced or increased as indicated by the urgent need of the treatment situation. It should be noted that the dose value may vary with the type and severity of the condition to be alleviated, and may include single or multiple doses. It should be further understood that for any particular individual, the specific dosing regimen should be adjusted over time according to the individual's needs and the professional judgment of the person administering the composition or supervising the administration of the composition.
- the amount of the compound of the present invention administered will depend on the individual being treated, the severity of the disorder or condition, the rate of administration, the treatment of the compound, and the judgment of the prescribing physician.
- the effective dose is about 0.0001 to about 50 mg per kg body weight per day, for example, about 0.01 to about 10 mg/kg/day (single or divided administration). For a 70 kg person, this would add up to about 0.007 mg/day to about 3500 mg/day, for example, about 0.7 mg/day to about 700 mg/day.
- a dose level not higher than the lower limit of the aforementioned range may be sufficient, while in other cases, a larger dose can still be used without causing any harmful side effects, provided that the larger dose is first
- the dose is divided into several smaller doses to be administered throughout the day.
- the content or amount of the compound of the present invention in the pharmaceutical composition may be about 0.01 mg to about 1000 mg.
- treating means reversing, alleviating, or inhibiting the progress of one or more symptoms of the disorder or condition to which such term is applied.
- prevention means being able to avoid the occurrence of the disease or condition to which such a term is applied.
- “Individual” as used herein includes human or non-human animals.
- Exemplary human individuals include human individuals (referred to as patients) or normal individuals suffering from diseases such as those described herein.
- non-human animals include all vertebrates, such as non-mammals (such as birds, amphibians, reptiles) and mammals, such as non-human primates, livestock and/or domesticated animals (such as sheep, dogs). , Cats, cows, pigs, etc.).
- the compound of the present invention may exist in the form of a solvate (preferably a hydrate), wherein the compound of the present invention contains a polar solvent as a structural element of the compound crystal lattice, in particular, for example, water, methanol or ethanol.
- a polar solvent as a structural element of the compound crystal lattice, in particular, for example, water, methanol or ethanol.
- the amount of polar solvent, especially water, can be present in a stoichiometric or non-stoichiometric ratio.
- metabolites of the compounds of the present invention that is, substances formed in the body when the compounds of the present invention are administered.
- Such products can be produced by, for example, oxidation, reduction, hydrolysis, amidation, deamidation, esterification, delipidation, enzymatic hydrolysis, etc. of the administered compound. Therefore, the present invention includes metabolites of the compounds of the present invention, including compounds obtained by contacting the compound of the present invention with a mammal for a time sufficient to produce its metabolites.
- the present invention further includes within its scope the prodrugs of the compounds of the present invention, which are certain derivatives of the compounds of the present invention that may themselves have little or no pharmacological activity, when administered to the body or The above can be converted into the compound of the present invention having the desired activity by, for example, hydrolytic cleavage.
- prodrugs will be functional group derivatives of the compound, which are easily converted into the desired therapeutically active compound in vivo.
- prodrugs please refer to "Pro-drugs as Novel Delivery Systems", Volume 14, ACS Symposium Series (T. Higuchi and V. Stella) and "Bioreversible Carriers in Drug Design," Pergamon Press, 1987 ( Edited by EBRoche, American Pharmaceutical Association).
- prodrugs of the present invention can be used, for example, by using certain parts known to those skilled in the art as “pro-moiety (for example, “Design of Prodrugs", described in H. Bundgaard (Elsevier, 1985))" It can be prepared by substituting appropriate functional groups present in the compounds of the present invention.
- the present invention also encompasses compounds of the present invention containing protecting groups.
- protection of sensitive groups or reactive groups on any relevant molecule may be necessary and/or desirable, thereby forming a chemically protected form of the compounds of the present invention.
- This can be achieved through conventional protective groups, such as those described in Protective Groups in Organic Chemistry, ed. JFW McOmie, Plenum Press, 1973; and TW Greene & P. GMWuts, Protective Groups in Organic Synthesis, John Wiley & Sons, 1991 Protective base. These references are incorporated herein by reference.
- the protecting group can be removed at an appropriate subsequent stage.
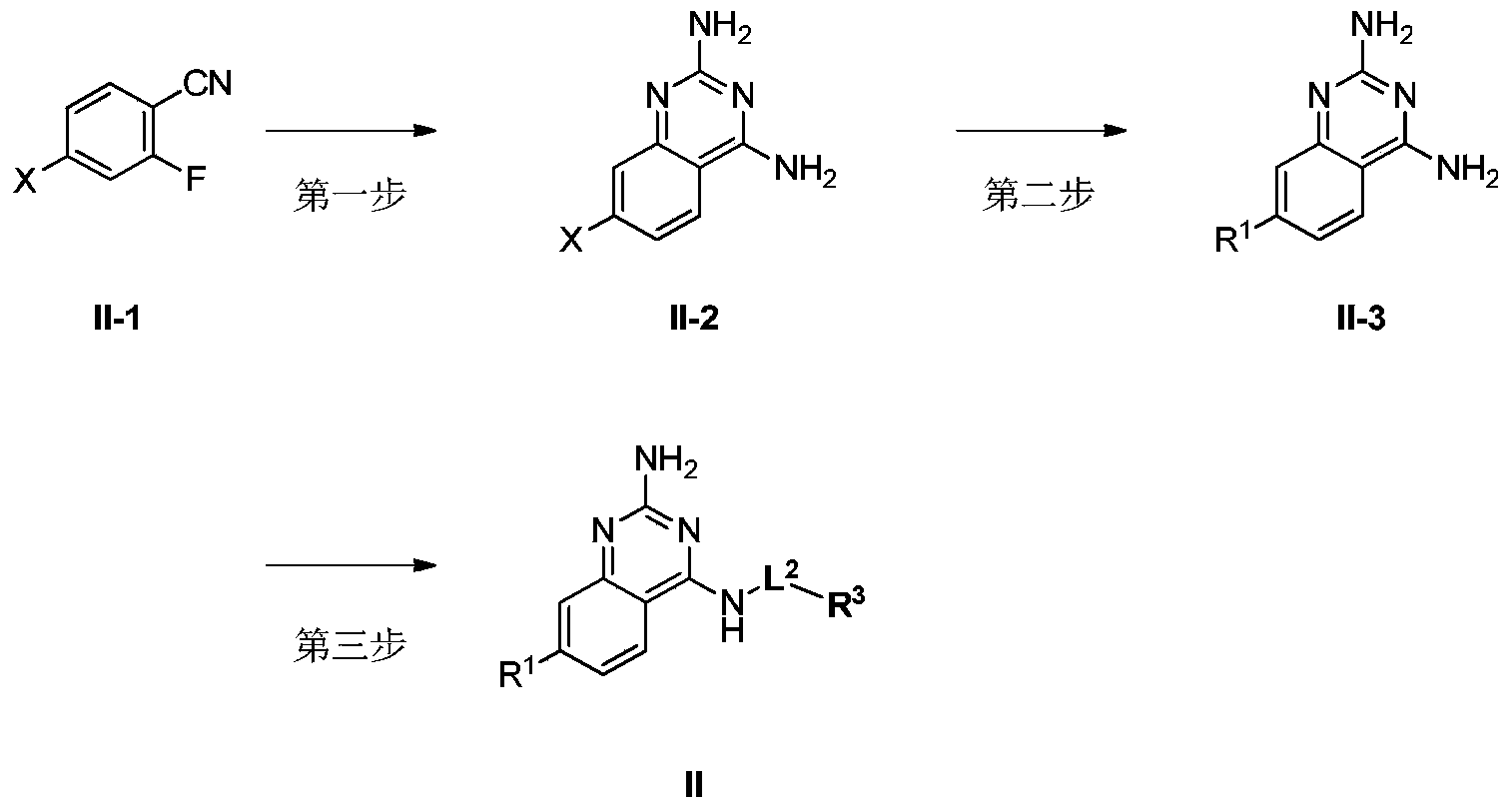
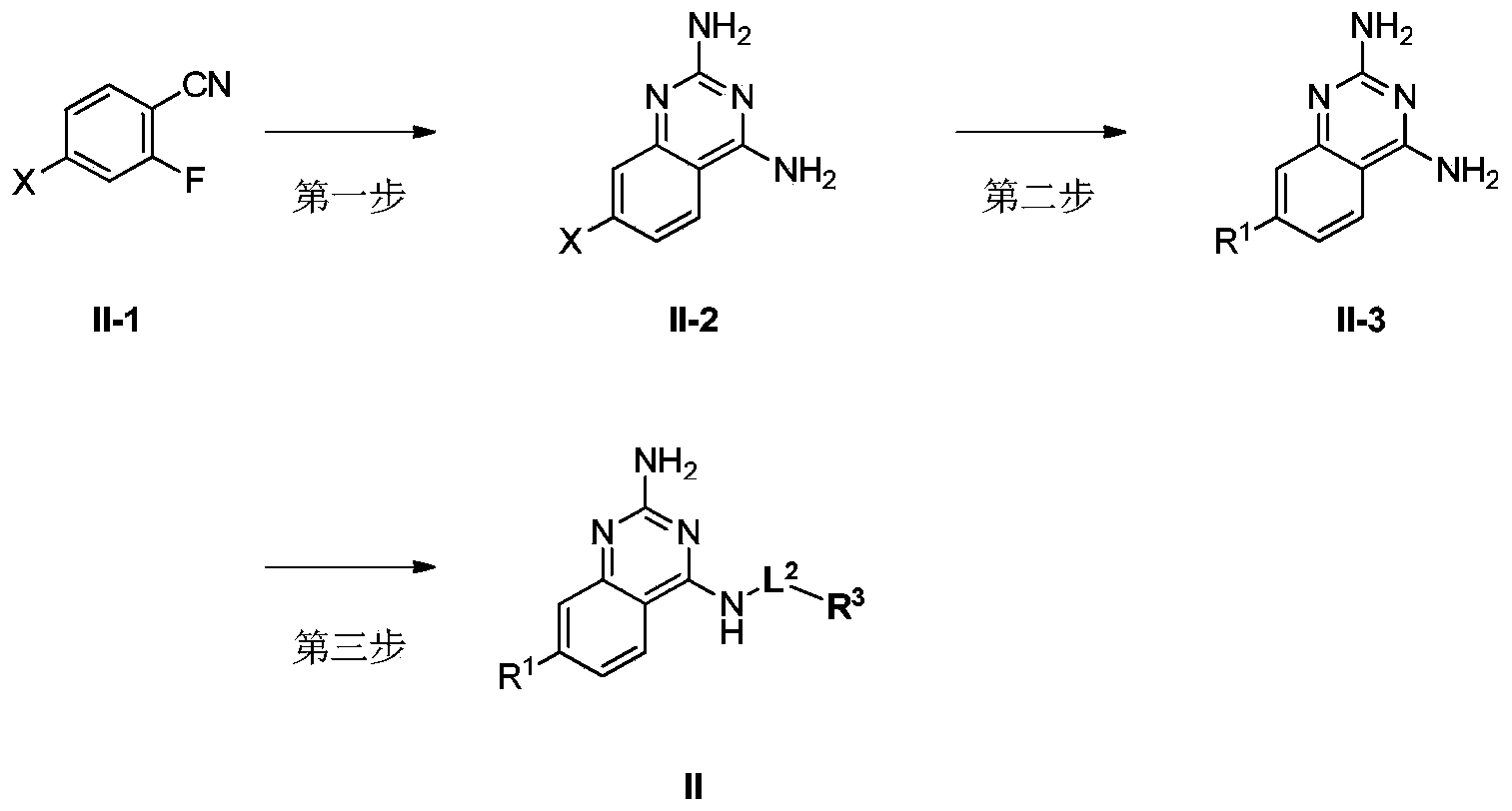
- R 1 , R 3 and L 2 are as defined in the above formula I, and X is selected from chlorine, bromine and iodine.
- the first step compound II-1 and guanidine carbonate undergo ring closure reaction to produce compound II-2.
- the solvent that can be used is 1,4-dioxane, dimethyl sulfoxide, DMF, N,N-dimethylacetamide, and the like.
- the reaction temperature is 0°C to 200°C.
- the second step Compound II-2 and R 1 -boronic acid or R 1 -boronic acid ester or R 1 -organotin compound (such as R 1 Sn(n-Bu) 3 ) undergo a coupling reaction (such as Suzuki reaction or Stille reaction) ) To produce compound II-3.
- a coupling reaction such as Suzuki reaction or Stille reaction
- the catalysts that can be used are Pd(PPh 3 ) 4 , Pd(dppf)Cl 2 ⁇ CH 2 Cl 2 and the like.
- the bases that can be used are Cs 2 CO 3 , K 3 PO 4 , Na 2 CO 3 , AcOK, NaHCO 3 , K 2 CO 3 and the like.
- the solvents that can be used are 1,4-dioxane/H 2 O, DMF/H 2 O, DMSO/H 2 O, CH 3 CN/H 2 O, toluene/H 2 O Wait.
- the reaction temperature is between 60°C and 180°C.
- the third step Compound II-3 and R 3 -L 2 -PG undergo substitution reaction to produce compound II, where PG is a leaving group, which can be halogen (for example, F, Cl, Br or I) or sulfonate.
- PG is a leaving group, which can be halogen (for example, F, Cl, Br or I) or sulfonate.
- the solvent that can be used is DMF, DMSO, THF, CH 3 CN, DCM, and the like.
- the base that can be used is sodium hydride, triethylamine, N,N-diisopropylethylamine, potassium tert-butoxide, sodium hydroxide, and the like.
- the reaction temperature is -20°C to 180°C.
- R 1 , R 3 and L 2 are as defined in the above formula I, and X is selected from chlorine, bromine and iodine.
- the first step compound II-4 and chloroformamidine hydrochloride undergo ring closure reaction to produce compound II-5.
- the solvent that can be used is 1,4-dioxane, dimethyl sulfone, and the like.
- the reaction temperature is 0°C to 200°C.
- the second step Compound II-5 and R 3 -L 2 -NH 2 undergo condensation reaction to produce compound II-6.
- condensing agents that can be used are BOP, pyBOP, HOBT, HATU, HBTU, TBTU, and the like.
- the bases that can be used are DBU, DIPEA, K 3 PO 4 , Na 2 CO 3 , AcOK, NaHCO 3 and the like.
- the reaction temperature is 0°C to 180°C.
- the third step Compound II-6 and R 1 -boronic acid or R 1 -boronic acid ester or R 1 -organotin compound (such as R 1 Sn(n-Bu) 3 ) undergo a coupling reaction (such as Suzuki reaction or Stille reaction) ), to produce compound II.
- a coupling reaction such as Suzuki reaction or Stille reaction
- the catalysts that can be used are Pd(PPh 3 ) 4 , Pd(dppf)Cl 2 ⁇ CH 2 Cl 2 and the like.
- the bases that can be used are Cs 2 CO 3 , K 3 PO 4 , Na 2 CO 3 , AcOK, NaHCO 3 , K 2 CO 3 and the like.
- the solvents that can be used are 1,4-dioxane/H 2 O, DMF/H 2 O, DMSO/H 2 O, CH 3 CN/H 2 O, toluene/H 2 O Wait.
- the reaction temperature is 60°C to 180°C.
- R 1 , R 3 and L 2 are as defined in the above formula III, and X is selected from chlorine, bromine and iodine.
- the first step compound IV-1 and chloroformamidine hydrochloride undergo ring closure reaction to produce compound IV-2.
- the solvent that can be used is 1,4-dioxane, dimethyl sulfone, and the like.
- the reaction temperature is 0°C to 200°C.
- the second step Compound IV-2 and R 3 -L 2 -NH 2 undergo condensation reaction to produce compound IV-3.
- condensing agents that can be used are BOP, pyBOP, HOBT, HATU, HBTU, TBTU, and the like.
- the bases that can be used are DBU, DIPEA, K 3 PO 4 , Na 2 CO 3 , AcOK, NaHCO 3 and the like.
- the reaction temperature is 0°C to 180°C.
- the third step Compound IV-3 and R 1 -boronic acid or R 1 -boronic acid ester or R 1 -organotin compound (such as R 1 Sn(n-Bu) 3 ) undergo coupling reaction (such as Suzuki reaction or Stille reaction ) To generate compound IV.
- R 1 -boronic acid or R 1 -boronic acid ester or R 1 -organotin compound such as R 1 Sn(n-Bu) 3
- coupling reaction such as Suzuki reaction or Stille reaction
- the catalysts that can be used are Pd(PPh 3 ) 4 , Pd(dppf)Cl 2 ⁇ CH 2 Cl 2 and the like.
- the bases that can be used are Cs 2 CO 3 , K 3 PO 4 , Na 2 CO 3 , AcOK, NaHCO 3 , K 2 CO 3 and the like.
- the solvents that can be used are 1,4-dioxane/H 2 O, DMF/H 2 O, DMSO/H 2 O, CH 3 CN/H 2 O, toluene/H 2 O Wait.
- the reaction temperature is between 60°C and 180°C.
- the compound of the present invention has obvious agonistic activity on NLRP3 and its signal pathway, without obvious toxic side effects, and can be used for the treatment of diseases related to NLRP3 inflammatory body activity, such as abnormal cell proliferation diseases (e.g. cancer).
- diseases related to NLRP3 inflammatory body activity such as abnormal cell proliferation diseases (e.g. cancer).
- the structure of the compound was identified by nuclear magnetic resonance spectroscopy ( 1 H NMR) and/or mass spectrometry (MS).
- the monitoring of the reaction is carried out by TLC or LC-MS.
- TLC uses silica gel GF 254 as the stationary phase.
- LC-MS uses Aglient 1260 Infinity/Aglient 6120 Quadrupole mass spectrometer to record.
- the compound can be separated and purified by preparative TLC, silica gel column chromatography, Prep-HPLC and/or flash column chromatography (Flash column chromatography).
- Prep-HPLC uses Agilent 1260 preparative liquid chromatograph, the detection wavelength is 214nm or 254nm; the chromatographic column is Waters SunFire Prep C18 OBD (19mm ⁇ 150mm ⁇ 5.0 ⁇ m); the column temperature is 25°C, and the elution conditions are as follows:
- Condition 1 10%-90% acetonitrile and 90%-10% ammonium formate aqueous solution (0.05%), 0-16min; flow rate: 24mL/min;
- Condition 3 10%-90% acetonitrile and 90%-10% formic acid aqueous solution (0.05%), 0-16min; flow rate: 28mL/min;
- Condition 4 10%-90% acetonitrile and 90%-10% ammonium bicarbonate aqueous solution (0.05%), 0-16min; flow rate: 28mL/min;
- Condition 7 0%-50% acetonitrile and 100%-50% ammonium bicarbonate aqueous solution (0.05%), 0-20min; flow rate: 28mL/min.
- Silica gel column chromatography generally uses 200-300 mesh silica gel (Qingdao Ocean) as the stationary phase.
- Eluent system A dichloromethane and methanol
- eluent system B petroleum ether and ethyl acetate; the volume ratio of the solvent is adjusted according to the polarity of the compound.
- the flash column chromatography uses the Biotage flash column chromatograph.
- the microwave reaction was carried out using BiotageInitiator+microwave reactor.
- reaction temperature is room temperature (15°C to 30°C).
- the reagents used in this application are purchased from companies such as Acros Organics, Aldrich Chemical Company, or Terb Chemical.
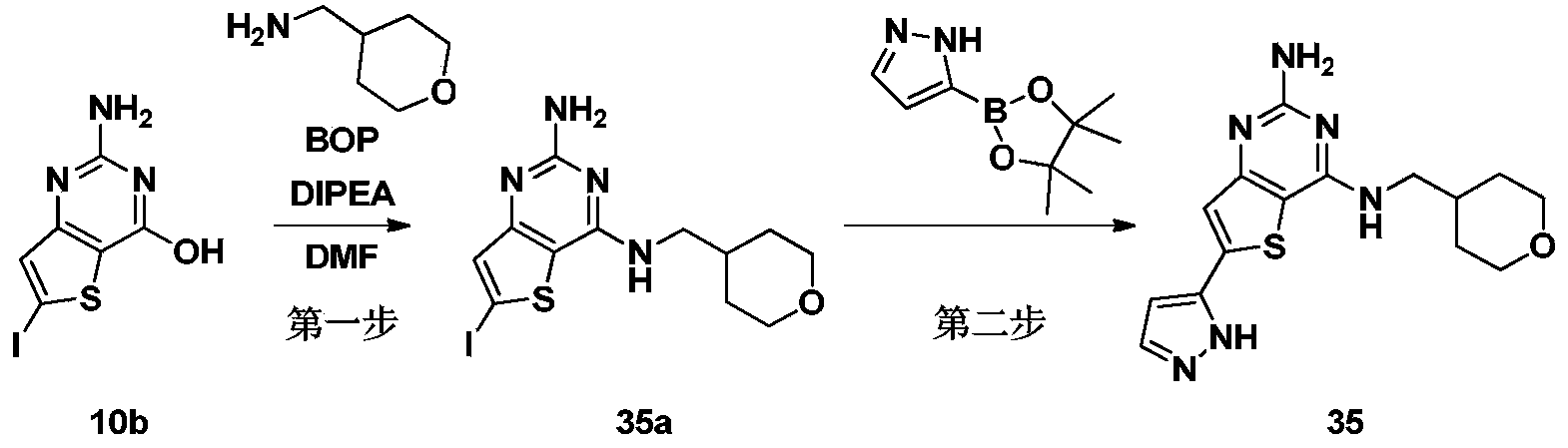
- the first step 7-bromoquinazoline-2,4-diamine (compound 1b)
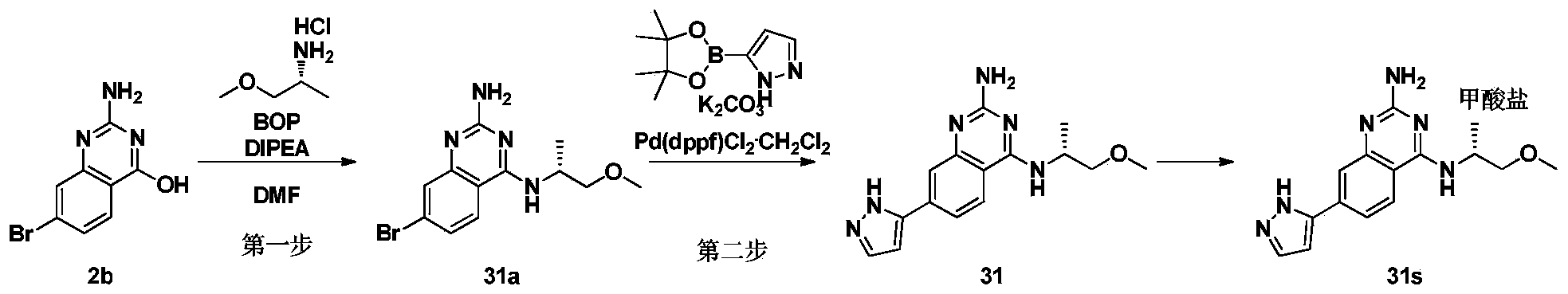
- the fourth step 3-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)-1-propanol formate (compound 1s)
- compound 2b 55 mg was added to DMF (3 mL), DBU (104.64 mg) and BOP (121.60 mg) were added successively, and the mixture was stirred for 5 min. Add 2-methoxyethylamine (127mg) and react for 16h. The reaction solution was suction filtered, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 2c (8 mg).
- the first step 3-((2-amino-7-bromoquinazolin-4-yl)amino)-2,2-dimethyl-1-propanol (compound 13a)
- the first step 4-((2-amino-7-bromoquinazolin-4-yl)amino)cyclohexanol (compound 14a)
- the first step 7-bromo-4-((2R,6R)-2,6-dimethylmorpholino)quinazolin-2-amine (compound 29a)
- the first step 7-bromo-N 4 -(cyclobutylmethyl)quinazoline-2,4-diamine (compound 30a)
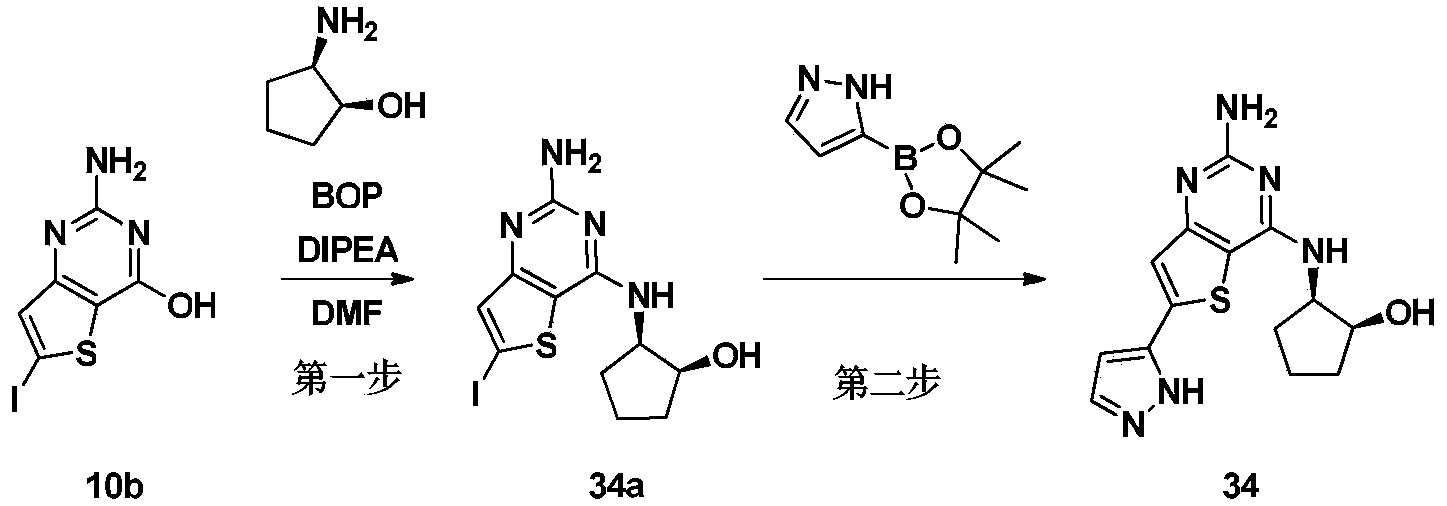
- Step 2 4-((2-Amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)cyclohexanol (Compound 38)
- Step 2 cis-3-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)-1-methyl ring Butanol (Compound 41)
- HTRF time-resolved fluorescence
- RPMI 1640 Hyclone
- FBS fetal bovine serum
- PMA Biyuntian
- Kit IL-1 ⁇ assay kit (CISBIO)
- THP-1 cells in the logarithmic growth phase were seeded in a T75 culture flask at a density of 5 ⁇ 10 5 cells/well, and then cultured in a 37°C, 5% CO 2 cell incubator for 24 hours.
- 1 ⁇ M PMA induces THP-1 suspension cells to become adherent macrophages.
- the medium was RPMI 1640 containing 10% heat-inactivated FBS and 0.05 mM ⁇ -mercaptoethanol.
- the HTRF detection method was used to test the effect of the compound of the present invention on the level of IL-1 ⁇ in THP1-def NLRP3 cells to evaluate the specificity of the compound on hNLRP3 inflammasome or hNLRP3 inflammasome pathway agonism.
- Kit IL-1 ⁇ assay kit (CISBIO)
- THP1-def NLRP3 cells in the logarithmic growth phase were seeded in a T75 culture flask at a density of 5 ⁇ 10 5 cells/well, and then cultured in a cell incubator at 37°C and 5% CO 2 for 24 hours.
- 1 ⁇ M PMA was used to induce THP1- def NLRP3 suspension cells to become adherent macrophages.
- the medium was RPMI 1640 containing 10% heat-inactivated FBS and 0.05 mM ⁇ -mercaptoethanol.
- the luciferase in HEK-hTLR7-NF- ⁇ B-luciferase reporter gene cells was tested to test the activating effect of the compound of the present invention on the TLR7 signaling pathway, so as to evaluate the specificity of the compound's agonistic effect on the NLRP3 pathway.
- DMEM High glucose
- FBS Gabco
- HEK-hTLR7-NF- ⁇ B-Luciferase reporter cells human TLR7NF- ⁇ B-luciferase reporter cells (Nanjing Kebai)
- DMEM High glucose
- FBS Gabco
- QUANTI-Blue InvivoGen
- HEK-Blue TM hTLR8 cells human TLR 8 cells (InvivoGen)
- the compounds of the present invention represented by the compounds listed in Table 1 have a significant agonistic effect on the expression of IL-1 ⁇ in THP-1 cells after PMA-induced differentiation, but on IL-1 ⁇ in THP-1 def NLRP3 cells.
- the expression has no agonistic effect even at the highest compound tested concentration (30 ⁇ M). All the compounds tested had no activating effect on hTLR7 and hTLR8 at 100 ⁇ M.
- the compounds of the present invention represented by the compounds listed in Table 1 have obvious specific agonistic activity on hNLRP3 and/or its signal pathway.
- HEK-293 human embryonic kidney cells
- Negative control extracellular fluid containing 0.1% DMSO (Sigma)
- CYP450 is the most important enzyme system in drug metabolism. Enzymes involved in metabolism interact with drugs, and the most important ones are CYP1A2, CYP2D6 and CYP3A4.
- This experiment uses P450-Glo TM CYP1A2 Screening System, CYP2D6Cyan Screening Kit and CYP3A4 Red Screening Kit, according to the kit instructions to determine the compound's inhibitory activity on CYP1A2, CYP2D6 and CYP3A4. The results are shown in Table 4.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Epidemiology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
相关申请的交叉引用Cross-references to related applications
本申请要求2019年12月13日提交的、名称为“含氮并环化合物、其制备方法及用途”的第201911279540.7号中国专利申请的优先权,将该专利申请的全部内容援引加入本文。This application claims the priority of the Chinese patent application No. 201911279540.7 filed on December 13, 2019 and titled "Nitrogen-containing hexacyclic compound, its preparation method and use", and the entire content of the patent application is incorporated herein by reference.
本发明涉及一类含氮并环化合物,所述化合物的立体异构体、互变异构体或其混合物,所述化合物的药学上可接受的盐、共晶、多晶型物或溶剂合物,或者所述化合物的稳定同位素衍生物、代谢物或前药。本发明的化合物作为NLRP3(NLR family pyrin domain containing 3)调节剂(例如激动剂或部分激动剂),可用于细胞增殖异常性疾病(例如癌症)的治疗。The present invention relates to a class of nitrogen-containing hexacyclic compounds, stereoisomers, tautomers or mixtures of said compounds, pharmaceutically acceptable salts, co-crystals, polymorphs or solvates of said compounds Or a stable isotope derivative, metabolite or prodrug of the compound. The compounds of the present invention are used as NLRP3 (NLR family pyrin domain containing 3) modulators (such as agonists or partial agonists) and can be used for the treatment of abnormal cell proliferation diseases (such as cancer).
发明背景Background of the invention
NLRP3属于NOD样受体家族,是近几年来研究最多的一种细胞内模式识别受体,主要表达于巨噬细胞和嗜中性粒细胞,参与机体固有免疫,抵抗病原体感染和应激损伤。NLRP3炎症小体在炎性和代谢类疾病中作用十分明确,其过度活化会导致2型糖尿病、诸如类风湿性关节炎的免疫性疾病和动脉粥样硬化等。然而,近年来的研究表明NLRP3有抑制肿瘤生长和转移的抗肿瘤作用。NLRP3 belongs to the family of NOD-like receptors. It is one of the most studied intracellular pattern recognition receptors in recent years. It is mainly expressed in macrophages and neutrophils. It participates in the body's innate immunity and resists pathogen infection and stress damage. NLRP3 inflammasome plays a very clear role in inflammatory and metabolic diseases, and its over-activation can lead to type 2 diabetes, immune diseases such as rheumatoid arthritis and atherosclerosis. However, recent studies have shown that NLRP3 has anti-tumor effects that inhibit tumor growth and metastasis.
NLRP3蛋白在识别病原相关分子模式(PAMP)或内源性的损伤相关分子模式(DAMP)后,其NOD结构域发生寡聚化并招募ASC和pro-caspase-1等蛋白,形成具有功能的NLRP3炎症小体。在pro-caspase-1被剪切活化成caspase-1之后,caspase-1大量剪切pro-IL-1β和pro-IL-18,使之转化成活性形式IL-1β和IL-18并释放到胞外,放大炎性反应。激动的NLRP3炎症小体可以显著提高肿瘤微环境中免疫因子IL-1β和IL-18的水平,启动天然免疫杀伤以及后续的获得性免疫应答以发挥其抗肿瘤作用。具体地,IL-1β可诱导CD8+T细胞分泌干扰素γ(IFN-γ),也可诱导CD4+细胞分泌IL-17,导致有效的抗肿瘤免疫效应;而IL-18则能够促进NK细胞成熟,激活免疫细胞内STAT1下游信号通路,增强免疫细胞的杀伤功能。临床研究显示,NLRP3的下调与肝癌病人的预后呈显著负相关。临床前研究也显示,NLRP3缺陷小鼠结直肠肿瘤的形成率更高且结直肠癌肝转移更加恶化。由此可见,NLRP3在肿瘤微环境中起着重要作用,可以作为肿瘤免疫治疗的一个关键靶点,也可作为肿瘤预后标志物。After NLRP3 protein recognizes pathogen-related molecular patterns (PAMP) or endogenous damage-related molecular patterns (DAMP), its NOD domain oligomerizes and recruits proteins such as ASC and pro-caspase-1 to form functional NLRP3 Inflammatory bodies. After pro-caspase-1 is cleaved and activated to caspase-1, caspase-1 cleaves pro-IL-1β and pro-IL-18 in a large amount to convert them into active forms of IL-1β and IL-18 and release them to Extracellular, amplify the inflammatory response. Agitated NLRP3 inflammasome can significantly increase the levels of immune factors IL-1β and IL-18 in the tumor microenvironment, initiate natural immune killing and subsequent adaptive immune responses to exert its anti-tumor effects. Specifically, IL-1β can induce CD8+ T cells to secrete interferon γ (IFN-γ) and CD4+ cells to secrete IL-17, leading to effective anti-tumor immune effects; while IL-18 can promote the maturation of NK cells Activates the downstream signaling pathway of STAT1 in immune cells and enhances the killing function of immune cells. Clinical studies have shown that the down-regulation of NLRP3 is significantly negatively correlated with the prognosis of liver cancer patients. Preclinical studies have also shown that NLRP3-deficient mice have a higher rate of colorectal tumor formation and worsen liver metastasis of colorectal cancer. It can be seen that NLRP3 plays an important role in the tumor microenvironment and can be used as a key target of tumor immunotherapy, as well as a tumor prognostic marker.
WO2017184746、WO2017184735、WO2018152396和WO2019014402公开了NLRP3调节剂。NLRP3激动剂有肿瘤免疫治疗的潜能,并且目前已知化合物BMS-986299处于临床I期研究。因此,需要开发新的、高效低毒的NLRP3激动剂来满足临床治疗需求。WO2017184746, WO2017184735, WO2018152396 and WO2019014402 disclose NLRP3 modulators. NLRP3 agonists have the potential for tumor immunotherapy, and currently known compound BMS-986299 is in clinical phase I study. Therefore, it is necessary to develop new, high-efficiency and low-toxicity NLRP3 agonists to meet the needs of clinical treatment.
发明概述Summary of the invention
本发明的发明人通过创造性的劳动,得到了一类新型含氮并环化合物,其可作为NLRP3调节剂(例如激动剂),在蛋白水平上直接结合或修饰NLRP3,通过活化、稳定、改变NLRP3分布或其它方式来增强NLRP3炎症小体的功能,从而提供了下述发明。Through creative work, the inventors of the present invention have obtained a new type of nitrogen-containing cyclic compounds, which can be used as NLRP3 modulators (such as agonists), directly binding or modifying NLRP3 at the protein level, and by activating, stabilizing, and changing NLRP3 Distribution or other ways to enhance the function of NLRP3 inflammasome, thereby providing the following invention.
化合物Compound
在一个方面,本发明提供具有式X所示结构的化合物,所述化合物的立体异构体、互变异构体或其混合物,所述化合物的药学上可接受的盐、共晶、多晶型物或溶剂合物,或者所述化合物的稳定同位素衍生物、代谢物或前药:In one aspect, the present invention provides compounds having the structure represented by formula X, stereoisomers, tautomers or mixtures thereof, pharmaceutically acceptable salts, co-crystals, polymorphs of the compounds Forms or solvates, or stable isotope derivatives, metabolites or prodrugs of said compounds:
R-L-R 3 RLR 3
式XFormula X
其中:among them:
R 3选自H、卤素、CN、NO 2、C 1-8烷基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、9-12元芳基并杂环基、9-12元芳基并杂芳基、9-12元芳基并环烷基、CO 2R 30、C(O)NR 31R 32、NR 33C(O)R 34、NR 31R 32、S(O) 2R 35、OR 37、SR 37、C(O)R 30、OC(O)R 30、OC(O)NR 31R 32、NR 33C(O)NR 31R 32、NR 33C(O)OR 30、C(=NR 38)NR 31R 32、NR 33C(=NR 38)NR 31R 32、P(R 39) 2、P(OR 39) 2、P(O)R 39R 40、P(O)OR 39OR 30、S(O)R 35、S(O)NR 31R 32和S(O) 2NR 31R 32,其中所述C 1-8烷基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、9-12元芳基并杂环基、9-12元芳基并杂芳基和9-12元芳基并环烷基各自任选地被下列取代基中的 一个或多个取代:卤素、CN、NO 2、C 1-4烷基、C 3-8环烷基、C 1-4卤代烷基、C 1-4羟烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、9-12元芳基并杂环基、CO 2R 30、C(O)R 30、C(O)NR 31R 32、NR 33C(O)R 34、NR 31R 32、S(O)R 35、S(O) 2R 35、S(O)NR 31R 32、S(O) 2NR 31R 32、OR 37、SR 37、OC(O)R 30、OC(O)NR 31R 32、NR 33C(O)NR 31R 32、NR 33C(O)OR 30、C(=NR 38)NR 31R 32、NR 33C(=NR 38)NR 31R 32、=NNR 31R 32、P(R 39) 2、P(OR 39) 2、P(O)R 39R 40和P(O)OR 39OR 30; R 3 is selected from H, halogen, CN, NO 2 , C 1-8 alkyl, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl , 9-12 membered aryl and heterocyclyl, 9-12 membered aryl and heteroaryl, 9-12 membered aryl and cycloalkyl, CO 2 R 30 , C(O)NR 31 R 32 , NR 33 C(O)R 34 , NR 31 R 32 , S(O) 2 R 35 , OR 37 , SR 37 , C(O)R 30 , OC(O)R 30 , OC(O)NR 31 R 32 , NR 33 C(O)NR 31 R 32 , NR 33 C(O)OR 30 , C(=NR 38 )NR 31 R 32 , NR 33 C(=NR 38 )NR 31 R 32 , P(R 39 ) 2 , P(OR 39 ) 2 , P(O)R 39 R 40 , P(O)OR 39 OR 30 , S(O)R 35 , S(O)NR 31 R 32 and S(O) 2 NR 31 R 32 , Wherein the C 1-8 alkyl group, C 3-8 cycloalkyl group, 4-10 membered heterocyclic group, C 6-12 aryl group, 5-10 membered heteroaryl group, 9-12 membered aryl group and hetero Cyclic, 9-12 membered aryl and heteroaryl and 9-12 membered aryl and cycloalkyl are each optionally substituted with one or more of the following substituents: halogen, CN, NO 2 , C 1- 4- alkyl, C 3-8 cycloalkyl, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl, 9-12 membered aryl and heterocyclic group, CO 2 R 30 , C(O)R 30 , C(O)NR 31 R 32 , NR 33 C(O)R 34 , NR 31 R 32 , S(O) R 35 , S(O) 2 R 35 , S(O)NR 31 R 32 , S(O) 2 NR 31 R 32 , OR 37 , SR 37 , OC(O)R 30 , OC(O)NR 31 R 32 , NR 33 C(O)NR 31 R 32 , NR 33 C(O)OR 30 , C(=NR 38 )NR 31 R 32 , NR 33 C(=NR 38 )NR 31 R 32 , =NNR 31 R 32 , P(R 39 ) 2 , P(OR 39 ) 2 , P(O)R 39 R 40 and P(O)OR 39 OR 30 ;
L为-(L 1) n-(L 2) p-(L 3) q-,其中L 1、L 2和L 3相同或不同且各自独立地选自C 1-8亚烷基、C 2-8亚烯基、C 2-8亚炔基、C 1-8亚烷氧基、C 3-8亚环烷基、4-10元亚杂环基、C 6-12亚芳基、5-10元亚杂芳基、O、S、NR 33、S(O)、S(O) 2、C(O)和C(R 36aR 36b),其中所述C 1-8亚烷基、C 2-8亚烯基、C 2-8亚炔基、C 1-8亚烷氧基、C 3-8亚环烷基、4-10元亚杂环基、C 6-12亚芳基和5-10元亚杂芳基各自任选地被下列取代基中的一个或多个取代:卤素、OH、CN、NO 2、C 1-6烷基、C 1-4卤代烷基、C 1-4羟烷基、C 1-4烷氧基和NR 31R 32; L is -(L 1 ) n -(L 2 ) p -(L 3 ) q -, wherein L 1 , L 2 and L 3 are the same or different and are each independently selected from C 1-8 alkylene, C 2 -8 alkenylene, C 2-8 alkynylene, C 1-8 alkyleneoxy, C 3-8 cycloalkylene, 4-10 membered heterocyclylene, C 6-12 arylene, 5 -10 membered heteroarylene, O, S, NR 33 , S(O), S(O) 2 , C(O) and C(R 36a R 36b ), wherein the C 1-8 alkylene group, C 2-8 alkenylene, C 2-8 alkynylene, C 1-8 alkyleneoxy, C 3-8 cycloalkylene, 4-10 membered heterocyclylene, C 6-12 arylene And 5-10 membered heteroarylene groups are each optionally substituted with one or more of the following substituents: halogen, OH, CN, NO 2 , C 1-6 alkyl, C 1-4 haloalkyl, C 1 -4 hydroxyalkyl, C 1-4 alkoxy and NR 31 R 32 ;
n、p和q各自独立地为0、1或2;n, p and q are each independently 0, 1 or 2;
R 30、R 37、R 39和R 40各自独立地选自H、C 1-8烷基(例如C 1-6烷基或C 1-4烷基)、C 1-8烷氧基(例如C 1- 6烷氧基或C 1-4烷氧基)、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、-C 1-8烷基-C 6-12芳基和-C 1-8烷基-(5-10元杂芳基),其中所述C 1-8烷基、C 1-8烷氧基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、-C 1-8烷基-C 6-12芳基和-C 1-8烷基-(5-10元杂芳基)各自任选地被下列取代基中的一个或多个取代:OH、CN、NO 2、C 1-4烷基、C 1-4烷氧基、C 1-4卤代烷基、卤素、C 1-4卤代烷氧基、CO 2(C 1-6烷基)、CONR 31R 32、NR 31R 32、NR 33C(O)R 34、S(O)R 35、S(O) 2R 35、S(O)NR 31R 32和S(O) 2NR 31R 32; R 30 , R 37 , R 39 and R 40 are each independently selected from H, C 1-8 alkyl (e.g. C 1-6 alkyl or C 1-4 alkyl), C 1-8 alkoxy (e.g. C 1- 6 alkoxy or C 1-4 alkoxy), C 3-8 cycloalkyl, 4-10 membered heterocyclyl, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 alkyl-(5-10 membered heteroaryl), wherein the C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 Alkyl-(5-10 membered heteroaryl) are each optionally substituted with one or more of the following substituents: OH, CN, NO 2 , C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, halogen, C 1-4 haloalkoxy, CO 2 (C 1-6 alkyl), CONR 31 R 32 , NR 31 R 32 , NR 33 C(O)R 34 , S(O ) R 35 , S(O) 2 R 35 , S(O)NR 31 R 32 and S(O) 2 NR 31 R 32 ;
R 31、R 32、R 33和R 34各自独立地选自H、C 1-8烷基、C 1-8烷氧基、C 3-8环烷基、4-10元杂环基、C 6-12芳基和5-10元杂芳基,或者R 31和R 32与它们所连接的N原子一起形成4-8元杂环基,或者R 33和R 34与它们各自所连接的C和N原子一起形成4-8元杂环基,其中所述C 1-8烷基、C 1-8烷氧基、C 3-8环烷基、4-8元杂环基、4-10元杂环基、C 6-12芳基和5-10元杂芳基各自任选地被下列取代基中的一个或多个取代:OH、CN、卤素、NO 2、C 1-4烷基、C 1-4烷氧基、C 1-4羟烷基、C 1-4卤代烷基、C 1-4卤代烷氧基、4-10元杂环基、C 6-12芳基和5-10元杂芳基; R 31 , R 32 , R 33 and R 34 are each independently selected from H, C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl and 5-10 membered heteroaryl, or R 31 and R 32 together with the N atom to which they are attached form a 4-8 membered heterocyclic group, or R 33 and R 34 and the C to which they are each attached Together with the N atom to form a 4-8 membered heterocyclic group, wherein the C 1-8 alkyl group, C 1-8 alkoxy group, C 3-8 cycloalkyl group, 4-8 membered heterocyclic group, 4-10 Each of the membered heterocyclic group, C 6-12 aryl group and 5-10 membered heteroaryl group is optionally substituted by one or more of the following substituents: OH, CN, halogen, NO 2 , C 1-4 alkyl , C 1-4 alkoxy, C 1-4 hydroxyalkyl, C 1-4 haloalkyl, C 1-4 haloalkoxy, 4-10 membered heterocyclic group, C 6-12 aryl and 5-10 Yuan heteroaryl
R 35选自C 1-8烷基、C 1-8烷氧基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、-C 1-8烷基-C 6-12芳基和-C 1-8烷基-(5-10元杂芳基),其中所述C 1-8烷基、C 1-8烷氧基、C 3-8环烷基、4-10元杂环基、C 6-12芳基和5-10元杂芳基各自任选地被下列取代基中的一个或多个取代:OH、CN、NO 2、C 1-4烷基、C 1-4烷氧基、C 1-4卤代烷基、卤素、C 1-4卤代烷氧基、CO 2(C 1-6烷基)、CONR 31R 32、NR 31R 32、NR 33C(O)R 34、S(O)Me、S(O) 2Me、S(O)NR 31R 32和S(O) 2NR 31R 32,其中R 31、R 32、R 33和R 34如上所定义; R 35 is selected from C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 alkyl-(5-10 membered heteroaryl), wherein the C 1-8 alkyl, C 1-8 alkoxy , C 3-8 cycloalkyl, 4-10 membered heterocyclyl, C 6-12 aryl and 5-10 membered heteroaryl are each optionally substituted by one or more of the following substituents: OH, CN , NO 2 , C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, halogen, C 1-4 haloalkoxy, CO 2 (C 1-6 alkyl), CONR 31 R 32 , NR 31 R 32 , NR 33 C(O)R 34 , S(O)Me, S(O) 2 Me, S(O)NR 31 R 32 and S(O) 2 NR 31 R 32 , where R 31 , R 32 , R 33 and R 34 are as defined above;
R 36a和R 36b相同或不同且各自独立地选自H、C 1-8烷基和C 1-8烷氧基,其中所述C 1-8烷基和C 1-8烷氧基各自任选地被下列基团中的一个或多个取代:OH、CN、卤素、NH 2、NHCH 3和N(CH 3) 2,或者R 36a和R 36b与它们所连接的C原子一起形成3-7元环烷基或杂环基; R 36a and R 36b are the same or different and are each independently selected from H, C 1-8 alkyl and C 1-8 alkoxy, wherein the C 1-8 alkyl and C 1-8 alkoxy are each independently Optionally substituted by one or more of the following groups: OH, CN, halogen, NH 2 , NHCH 3 and N(CH 3 ) 2 , or R 36a and R 36b together with the C atom to which they are attached form a 3- 7-membered cycloalkyl or heterocyclic group;
R 38选自H、OH、CN、NO 2、S(O)R 35和S(O) 2R 35; R 38 is selected from H, OH, CN, NO 2 , S(O)R 35 and S(O) 2 R 35 ;
当多个R 30同时存在时,各R 30可以相同或不同; When multiple R 30 exist at the same time, each R 30 may be the same or different;
当多个R 31同时存在时,各R 31可以相同或不同; When multiple R 31 exist at the same time, each R 31 may be the same or different;
当多个R 32同时存在时,各R 32可以相同或不同; When multiple R 32 exist at the same time, each R 32 may be the same or different;
当多个R 33同时存在时,各R 33可以相同或不同; When multiple R 33 exist at the same time, each R 33 may be the same or different;
当多个R 34同时存在时,各R 34可以相同或不同; When multiple R 34 exist at the same time, each R 34 may be the same or different;
当多个R 35同时存在时,各R 35可以相同或不同; When multiple R 35 exist at the same time, each R 35 may be the same or different;
当多个R 37同时存在时,各R 37可以相同或不同; When multiple R 37 exist at the same time, each R 37 may be the same or different;
当多个R 38同时存在时,各R 38可以相同或不同; When multiple R 38 exist at the same time, each R 38 may be the same or different;
当多个R 39同时存在时,各R 39可以相同或不同; When multiple R 39 exist at the same time, each R 39 may be the same or different;
当多个R 40同时存在时,各R 40可以相同或不同; When multiple R 40 exist at the same time, each R 40 may be the same or different;
R选自 其中: R is selected from among them:
R 1选自C 1-8烷基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基和9-12元芳基并杂环基,其中所述C 1-8烷基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基和9-12元芳基并杂环基各自任选地被下列取代基中的一个或多个取代:卤素、CN、NO 2、C 1-4烷基、C 3-8环烷基、C 1-4卤代烷基、C 1- 4羟烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、9-12元芳基并杂环基、CO 2R 30、C(O)R 30、C(O)NR 31R 32、NR 33C(O)R 34、NR 31R 32、OC(O)R 30、OC(O)NR 31R 32、NR 33C(O)NR 31R 32、NR 33C(O)OR 30、S(O)NR 31R 32、S(O) 2NR 31R 32、S(O)R 35、S(O) 2R 35、OR 37和SR 37; R 1 is selected from C 1-8 alkyl, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl and 9-12 membered aryl and hetero Cyclic group, wherein the C 1-8 alkyl group, C 3-8 cycloalkyl group, 4-10 membered heterocyclic group, C 6-12 aryl group, 5-10 membered heteroaryl group and 9-12 membered aryl group The heterocyclic groups are each optionally substituted by one or more of the following substituents: halogen, CN, NO 2 , C 1-4 alkyl, C 3-8 cycloalkyl, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-10 membered heterocyclyl, C 6-12 aryl, 5-10 membered heteroaryl, 9-12 membered heterocyclyl, and aryl group, CO 2 R 30, C ( O) R 30 , C(O)NR 31 R 32 , NR 33 C(O)R 34 , NR 31 R 32 , OC(O)R 30 , OC(O)NR 31 R 32 , NR 33 C(O)NR 31 R 32 , NR 33 C(O)OR 30 , S(O)NR 31 R 32 , S(O) 2 NR 31 R 32 , S(O)R 35 , S(O) 2 R 35 , OR 37 and SR 37 ;
R 2选自H、NR 41aR 41b、C 1-8烷基、C 2-8烯基、C 2-8炔基、C 3-8环烷基和4-10元杂环基,其中所述C 1- 8烷基、C 2-8烯基、C 2-8炔基、C 3-8环烷基和4-10元杂环基各自任选地被下列取代基中的一个或多个取代:卤素、C 1-4卤代烷基、C 1-4羟烷基、4-7元杂环基、CN、NO 2、OR 37、SR 37、C(O)R 30、C(O)NR 31R 32、NR 33C(O)R 34、C(O)OR 30、OC(O)R 30、OC(O)NR 31R 32、NR 33C(O)NR 31R 32和NR 31R 32; R 2 is selected from H, NR 41a R 41b , C 1-8 alkyl, C 2-8 alkenyl, C 2-8 alkynyl, C 3-8 cycloalkyl and 4-10 membered heterocyclic group, wherein said C 1- 8 alkyl, C 2-8 alkenyl, C 2-8 alkynyl, C 3-8 cycloalkyl and 4-10 membered heterocyclyl group each optionally substituted with a group of the following or One substitution: halogen, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-7 membered heterocyclic group, CN, NO 2 , OR 37 , SR 37 , C(O)R 30 , C(O) NR 31 R 32 , NR 33 C(O)R 34 , C(O)OR 30 , OC(O)R 30 , OC(O)NR 31 R 32 , NR 33 C(O)NR 31 R 32 and NR 31 R 32 ;
R 5不存在或者选自卤素、C 1-6烷基、C 1-4烷氧基、C 3-8环烷基和4-10元杂环基,其中所述C 1-6烷基、C 1-4烷氧基、C 3-8环烷基和4-10元杂环基各自任选地被下列基团中的一个或多个取代:卤素、OH、CN、C 1-4烷氧基、C 1-4羟烷基和NR 31R 32; R 5 is absent or selected from halogen, C 1-6 alkyl, C 1-4 alkoxy, C 3-8 cycloalkyl and 4-10 membered heterocyclic group, wherein the C 1-6 alkyl, C 1-4 alkoxy, C 3-8 cycloalkyl, and 4-10 membered heterocyclic group are each optionally substituted with one or more of the following groups: halogen, OH, CN, C 1-4 alkane Oxy, C 1-4 hydroxyalkyl and NR 31 R 32 ;
m为0、1或2,优选为0或1;m is 0, 1 or 2, preferably 0 or 1;
X 1选自CR 6和N; X 1 is selected from CR 6 and N;
R 4选自H、NR 41aR 41b、C 1-15烷基、C 1-8烷氧基、C 2-8烯基、C 2-8炔基、C 3-8环烷基和4-10元杂环基,其中所述C 1-15烷基、C 1-8烷氧基、C 2-8烯基、C 2-8炔基、C 3-8环烷基和4-10元杂环基各自任选地被下列取代基中的一个或多个取代:卤素、C 1-4卤代烷基、C 1-4羟烷基、4-7元杂环基、CN、NO 2、OR 37、SR 37、C(O)R 30、C(O)NR 31R 32、NR 33C(O)R 34、C(O)OR 30、OC(O)R 30、OC(O)NR 31R 32、NR 33C(O)NR 31R 32和NR 31R 32; R 4 is selected from H, NR 41a R 41b , C 1-15 alkyl, C 1-8 alkoxy, C 2-8 alkenyl, C 2-8 alkynyl, C 3-8 cycloalkyl and 4- 10-membered heterocyclic group, wherein the C 1-15 alkyl group, C 1-8 alkoxy group, C 2-8 alkenyl group, C 2-8 alkynyl group, C 3-8 cycloalkyl group and 4-10 membered The heterocyclic groups are each optionally substituted by one or more of the following substituents: halogen, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-7 membered heterocyclyl, CN, NO 2 , OR 37 , SR 37 , C(O)R 30 , C(O)NR 31 R 32 , NR 33 C(O)R 34 , C(O)OR 30 , OC(O)R 30 , OC(O)NR 31 R 32 , NR 33 C(O)NR 31 R 32 and NR 31 R 32 ;
R 6选自H、卤素、C 1-6烷基、C 1-4烷氧基、C 3-8环烷基和4-10元杂环基,其中所述C 1-6烷基、C 1-4烷氧基、C 3-8环烷基和4-10元杂环基各自任选地被下列基团中的一个或多个取代:卤素、OH、CN、C 1-4烷氧基、C 1-4羟烷基和NR 31R 32; R 6 is selected from H, halogen, C 1-6 alkyl, C 1-4 alkoxy, C 3-8 cycloalkyl and 4-10 membered heterocyclic group, wherein the C 1-6 alkyl, C 1-4 alkoxy, C 3-8 cycloalkyl, and 4-10 membered heterocyclic group are each optionally substituted with one or more of the following groups: halogen, OH, CN, C 1-4 alkoxy Group, C 1-4 hydroxyalkyl group and NR 31 R 32 ;
R 41a和R 41b各自独立地选自H、C 1-6烷基、C 1-6烷氧基和C 3-8环烷基,或者R 41a和R 41b与它们所连接的N原子一起形成4-7元杂环基,其中所述C 1-6烷基、C 1-6烷氧基、C 3-8环烷基和4-7元杂环基各自任选地被下列基团中的一个或多个取代:OH、CN和NR 31R 32。 R 41a and R 41b are each independently selected from H, C 1-6 alkyl, C 1-6 alkoxy and C 3-8 cycloalkyl, or R 41a and R 41b form together with the N atom to which they are attached 4-7 membered heterocyclic group, wherein the C 1-6 alkyl group, C 1-6 alkoxy group, C 3-8 cycloalkyl group and 4-7 membered heterocyclic group are each optionally selected from the following groups One or more of the substitutions: OH, CN and NR 31 R 32 .
在一些实施方案中,本发明提供具有式I所示结构的化合物,所述化合物的立体异构体、互变异构体或其混合物,所述化合物的药学上可接受的盐、共晶、多晶型物或溶剂合物,或者所述化合物的稳定同位素衍生物、代谢物或前药:In some embodiments, the present invention provides a compound having a structure represented by formula I, a stereoisomer, a tautomer or a mixture of the compound, a pharmaceutically acceptable salt, co-crystal, Polymorphs or solvates, or stable isotope derivatives, metabolites or prodrugs of said compounds:
其中:among them:
R 1选自C 1-8烷基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基和9-12元芳基并杂环基,其中所述C 1-8烷基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基和9-12元芳基并杂环基各自任选地被下列取代基中的一个或多个取代:卤素、CN、NO 2、C 1-4烷基、C 3-8环烷基、C 1-4卤代烷基、C 1- 4羟烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、9-12元芳基并杂环基、CO 2R 30、C(O)R 30、C(O)NR 31R 32、NR 33C(O)R 34、NR 31R 32、OC(O)R 30、OC(O)NR 31R 32、NR 33C(O)NR 31R 32、NR 33C(O)OR 30、S(O)NR 31R 32、S(O) 2NR 31R 32、S(O)R 35、S(O) 2R 35、OR 37和SR 37; R 1 is selected from C 1-8 alkyl, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl and 9-12 membered aryl and hetero Cyclic group, wherein the C 1-8 alkyl group, C 3-8 cycloalkyl group, 4-10 membered heterocyclic group, C 6-12 aryl group, 5-10 membered heteroaryl group and 9-12 membered aryl group The heterocyclic groups are each optionally substituted by one or more of the following substituents: halogen, CN, NO 2 , C 1-4 alkyl, C 3-8 cycloalkyl, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-10 membered heterocyclyl, C 6-12 aryl, 5-10 membered heteroaryl, 9-12 membered heterocyclyl, and aryl group, CO 2 R 30, C ( O) R 30 , C(O)NR 31 R 32 , NR 33 C(O)R 34 , NR 31 R 32 , OC(O)R 30 , OC(O)NR 31 R 32 , NR 33 C(O)NR 31 R 32 , NR 33 C(O)OR 30 , S(O)NR 31 R 32 , S(O) 2 NR 31 R 32 , S(O)R 35 , S(O) 2 R 35 , OR 37 and SR 37 ;
R 2选自H、NR 41aR 41b、C 1-8烷基、C 2-8烯基、C 2-8炔基、C 3-8环烷基和4-10元杂环基,其中所述C 1- 8烷基、C 2-8烯基、C 2-8炔基、C 3-8环烷基和4-10元杂环基各自任选地被下列取代基中的一个或多个取代:卤素、C 1-4卤代烷基、C 1-4羟烷基、4-7元杂环基、CN、NO 2、OR 37、SR 37、C(O)R 30、C(O)NR 31R 32、NR 33C(O)R 34、C(O)OR 30、OC(O)R 30、OC(O)NR 31R 32、NR 33C(O)NR 31R 32和NR 31R 32; R 2 is selected from H, NR 41a R 41b , C 1-8 alkyl, C 2-8 alkenyl, C 2-8 alkynyl, C 3-8 cycloalkyl and 4-10 membered heterocyclic group, wherein said C 1- 8 alkyl, C 2-8 alkenyl, C 2-8 alkynyl, C 3-8 cycloalkyl and 4-10 membered heterocyclyl group each optionally substituted with a group of the following or One substitution: halogen, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-7 membered heterocyclic group, CN, NO 2 , OR 37 , SR 37 , C(O)R 30 , C(O) NR 31 R 32 , NR 33 C(O)R 34 , C(O)OR 30 , OC(O)R 30 , OC(O)NR 31 R 32 , NR 33 C(O)NR 31 R 32 and NR 31 R 32 ;
R 3在每次出现时独立地选自H、卤素、CN、NO 2、C 1-8烷基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、9-12元芳基并杂环基、9-12元芳基并杂芳基、9-12元芳基并环烷基、CO 2R 30、C(O)NR 31R 32、NR 33C(O)R 34、NR 31R 32、S(O) 2R 35、OR 37、SR 37、C(O)R 30、OC(O)R 30、OC(O)NR 31R 32、NR 33C(O)NR 31R 32、NR 33C(O)OR 30、C(=NR 38)NR 31R 32、NR 33C(=NR 38)NR 31R 32、P(R 39) 2、P(OR 39) 2、P(O)R 39R 40、P(O)OR 39OR 30、S(O)R 35、S(O)NR 31R 32和S(O) 2NR 31R 32,其中所述C 1-8烷基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、9-12元芳基并杂环基、9-12元芳基并杂芳基和9-12元芳基并环烷基各自任选地被下列取代基中的一个或多个取代:卤素、CN、NO 2、C 1-4烷基、C 3-8环烷基、C 1-4卤代烷基、C 1-4羟烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、9-12元芳基并杂环基、CO 2R 30、C(O)R 30、C(O)NR 31R 32、NR 33C(O)R 34、NR 31R 32、S(O)R 35、S(O) 2R 35、S(O)NR 31R 32、S(O) 2NR 31R 32、OR 37、SR 37、OC(O)R 30、OC(O)NR 31R 32、NR 33C(O)NR 31R 32、NR 33C(O)OR 30、C(=NR 38)NR 31R 32、NR 33C(=NR 38)NR 31R 32、 =NNR 31R 32、P(R 39) 2、P(OR 39) 2、P(O)R 39R 40和P(O)OR 39OR 30; R 3 at each occurrence is independently selected from H, halogen, CN, NO 2 , C 1-8 alkyl, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl, 9-12 membered aryl and heterocyclic group, 9-12 membered aryl and heteroaryl, 9-12 membered aryl and cycloalkyl, CO 2 R 30 , C(O) NR 31 R 32 , NR 33 C(O)R 34 , NR 31 R 32 , S(O) 2 R 35 , OR 37 , SR 37 , C(O)R 30 , OC(O)R 30 , OC(O ) NR 31 R 32 , NR 33 C(O)NR 31 R 32 , NR 33 C(O)OR 30 , C(=NR 38 )NR 31 R 32 , NR 33 C(=NR 38 )NR 31 R 32 , P(R 39 ) 2 , P(OR 39 ) 2 , P(O)R 39 R 40 , P(O)OR 39 OR 30 , S(O)R 35 , S(O)NR 31 R 32 and S( O) 2 NR 31 R 32 , wherein the C 1-8 alkyl group, C 3-8 cycloalkyl group, 4-10 membered heterocyclic group, C 6-12 aryl group, 5-10 membered heteroaryl group, 9 The -12-membered aryl and heterocyclyl, 9-12-membered aryl and heteroaryl, and 9-12-membered aryl and cycloalkyl are each optionally substituted with one or more of the following substituents: halogen, CN , NO 2 , C 1-4 alkyl, C 3-8 cycloalkyl, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5 -10 membered heteroaryl, 9-12 membered aryl and heterocyclic group, CO 2 R 30 , C(O)R 30 , C(O)NR 31 R 32 , NR 33 C(O)R 34 , NR 31 R 32 , S(O)R 35 , S(O) 2 R 35 , S(O)NR 31 R 32 , S(O) 2 NR 31 R 32 , OR 37 , SR 37 , OC(O)R 30 , OC(O)NR 31 R 32 , NR 33 C(O)NR 31 R 32 , NR 33 C(O)OR 30 , C(=NR 38 )NR 31 R 32 , NR 33 C(=NR 38 )NR 31 R 32 , = NNR 31 R 32 , P(R 39 ) 2 , P(OR 39 ) 2 , P(O)R 39 R 40 and P(O)OR 39 OR 30 ;
R 5不存在或者选自卤素、C 1-6烷基、C 1-4烷氧基、C 3-8环烷基和4-10元杂环基,其中所述C 1-6烷基、C 1-4烷氧基、C 3-8环烷基和4-10元杂环基各自任选地被下列基团中的一个或多个取代:卤素、OH、CN、C 1-4烷氧基、C 1-4羟烷基和NR 31R 32; R 5 is absent or selected from halogen, C 1-6 alkyl, C 1-4 alkoxy, C 3-8 cycloalkyl and 4-10 membered heterocyclic group, wherein the C 1-6 alkyl, C 1-4 alkoxy, C 3-8 cycloalkyl, and 4-10 membered heterocyclic group are each optionally substituted with one or more of the following groups: halogen, OH, CN, C 1-4 alkane Oxy, C 1-4 hydroxyalkyl and NR 31 R 32 ;
m为0、1或2,优选为0或1;m is 0, 1 or 2, preferably 0 or 1;
L为-(L 1) n-(L 2) p-(L 3) q-,其中L 1、L 2和L 3相同或不同且各自在每次出现时独立地选自C 1-8亚烷基、C 2-8亚烯基、C 2-8亚炔基、C 1-8亚烷氧基、C 3-8亚环烷基、4-10元亚杂环基、C 6-12亚芳基、5-10元亚杂芳基、O、S、NR 33、S(O)、S(O) 2、C(O)和C(R 36aR 36b),其中所述C 1-8亚烷基、C 2-8亚烯基、C 2-8亚炔基、C 1-8亚烷氧基、C 3-8亚环烷基、4-10元亚杂环基、C 6-12亚芳基和5-10元亚杂芳基各自任选地被下列取代基中的一个或多个取代:卤素、OH、CN、NO 2、C 1-6烷基、C 1-4卤代烷基、C 1-4羟烷基、C 1-4烷氧基和NR 31R 32; L is -(L 1 ) n -(L 2 ) p -(L 3 ) q -, wherein L 1 , L 2 and L 3 are the same or different and each is independently selected from C 1-8 sub Alkyl, C 2-8 alkenylene, C 2-8 alkynylene, C 1-8 alkyleneoxy, C 3-8 cycloalkylene, 4-10 membered heterocyclylene, C 6-12 Arylene, 5-10 membered heteroarylene, O, S, NR 33 , S(O), S(O) 2 , C(O) and C(R 36a R 36b ), wherein the C 1- 8 alkylene, C 2-8 alkenylene, C 2-8 alkynylene, C 1-8 alkyleneoxy, C 3-8 cycloalkylene, 4-10 membered heterocyclylene, C 6 The -12 arylene group and the 5-10 membered heteroarylene group are each optionally substituted with one or more of the following substituents: halogen, OH, CN, NO 2 , C 1-6 alkyl, C 1-4 Haloalkyl, C 1-4 hydroxyalkyl, C 1-4 alkoxy and NR 31 R 32 ;
n、p和q各自在每次出现时独立地为0、1或2;n, p, and q are each independently 0, 1 or 2 each time they appear;
R 30、R 37、R 39和R 40各自独立地选自H、C 1-8烷基(例如C 1-6烷基或C 1-4烷基)、C 1-8烷氧基(例如C 1- 6烷氧基或C 1-4烷氧基)、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、-C 1-8烷基-C 6-12芳基和-C 1-8烷基-(5-10元杂芳基),其中所述C 1-8烷基、C 1-8烷氧基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、-C 1-8烷基-C 6-12芳基和-C 1-8烷基-(5-10元杂芳基)各自任选地被下列取代基中的一个或多个取代:OH、CN、NO 2、C 1-4烷基、C 1-4烷氧基、C 1-4卤代烷基、卤素、C 1-4卤代烷氧基、CO 2(C 1-6烷基)、CONR 31R 32、NR 31R 32、NR 33C(O)R 34、S(O)R 35、S(O) 2R 35、S(O)NR 31R 32和S(O) 2NR 31R 32; R 30 , R 37 , R 39 and R 40 are each independently selected from H, C 1-8 alkyl (e.g. C 1-6 alkyl or C 1-4 alkyl), C 1-8 alkoxy (e.g. C 1- 6 alkoxy or C 1-4 alkoxy), C 3-8 cycloalkyl, 4-10 membered heterocyclyl, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 alkyl-(5-10 membered heteroaryl), wherein the C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 Alkyl-(5-10 membered heteroaryl) are each optionally substituted with one or more of the following substituents: OH, CN, NO 2 , C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, halogen, C 1-4 haloalkoxy, CO 2 (C 1-6 alkyl), CONR 31 R 32 , NR 31 R 32 , NR 33 C(O)R 34 , S(O ) R 35 , S(O) 2 R 35 , S(O)NR 31 R 32 and S(O) 2 NR 31 R 32 ;
R 31、R 32、R 33和R 34各自独立地选自H、C 1-8烷基、C 1-8烷氧基、C 3-8环烷基、4-10元杂环基、C 6-12芳基和5-10元杂芳基,或者R 31和R 32与它们所连接的N原子一起形成4-8元杂环基,或者R 33和R 34与它们各自所连接的C和N原子一起形成4-8元杂环基,其中所述C 1-8烷基、C 1-8烷氧基、C 3-8环烷基、4-8元杂环基、4-10元杂环基、C 6-12芳基和5-10元杂芳基各自任选地被下列取代基中的一个或多个取代:OH、CN、卤素、NO 2、C 1-4烷基、C 1-4烷氧基、C 1-4羟烷基、C 1-4卤代烷基、C 1-4卤代烷氧基、4-10元杂环基、C 6-12芳基和5-10元杂芳基; R 31 , R 32 , R 33 and R 34 are each independently selected from H, C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl and 5-10 membered heteroaryl, or R 31 and R 32 together with the N atom to which they are attached form a 4-8 membered heterocyclic group, or R 33 and R 34 and the C to which they are each attached Together with the N atom to form a 4-8 membered heterocyclic group, wherein the C 1-8 alkyl group, C 1-8 alkoxy group, C 3-8 cycloalkyl group, 4-8 membered heterocyclic group, 4-10 Each of the membered heterocyclic group, C 6-12 aryl group and 5-10 membered heteroaryl group is optionally substituted by one or more of the following substituents: OH, CN, halogen, NO 2 , C 1-4 alkyl , C 1-4 alkoxy, C 1-4 hydroxyalkyl, C 1-4 haloalkyl, C 1-4 haloalkoxy, 4-10 membered heterocyclic group, C 6-12 aryl and 5-10 Yuan heteroaryl
R 35选自C 1-8烷基、C 1-8烷氧基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、-C 1-8烷基-C 6-12芳基和-C 1-8烷基-(5-10元杂芳基),其中所述C 1-8烷基、C 1-8烷氧基、C 3-8环烷基、4-10元杂环基、C 6-12芳基和5-10元杂芳基各自任选地被下列取代基中的一个或多个取代:OH、CN、NO 2、C 1-4烷基、C 1-4烷氧基、C 1-4卤代烷基、卤素、C 1-4卤代烷氧基、CO 2(C 1-6烷基)、CONR 31R 32、NR 31R 32、NR 33C(O)R 34、S(O)Me、S(O) 2Me、S(O)NR 31R 32和S(O) 2NR 31R 32,其中R 31、R 32、R 33和R 34如上所定义; R 35 is selected from C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 alkyl-(5-10 membered heteroaryl), wherein the C 1-8 alkyl, C 1-8 alkoxy , C 3-8 cycloalkyl, 4-10 membered heterocyclyl, C 6-12 aryl and 5-10 membered heteroaryl are each optionally substituted by one or more of the following substituents: OH, CN , NO 2 , C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, halogen, C 1-4 haloalkoxy, CO 2 (C 1-6 alkyl), CONR 31 R 32 , NR 31 R 32 , NR 33 C(O)R 34 , S(O)Me, S(O) 2 Me, S(O)NR 31 R 32 and S(O) 2 NR 31 R 32 , where R 31 , R 32 , R 33 and R 34 are as defined above;
R 36a和R 36b相同或不同且各自独立地选自H、C 1-8烷基和C 1-8烷氧基,其中所述C 1-8烷基和C 1-8烷氧基各自任选地被下列基团中的一个或多个取代:OH、CN、卤素、NH 2、NHCH 3和N(CH 3) 2,或者R 36a和R 36b与它们所连接的C原子一起形成3-7元环烷基或杂环基; R 36a and R 36b are the same or different and are each independently selected from H, C 1-8 alkyl and C 1-8 alkoxy, wherein the C 1-8 alkyl and C 1-8 alkoxy are each independently Optionally substituted by one or more of the following groups: OH, CN, halogen, NH 2 , NHCH 3 and N(CH 3 ) 2 , or R 36a and R 36b together with the C atom to which they are attached form a 3- 7-membered cycloalkyl or heterocyclic group;
R 38选自H、OH、CN、NO 2、S(O)R 35和S(O) 2R 35; R 38 is selected from H, OH, CN, NO 2 , S(O)R 35 and S(O) 2 R 35 ;
R 41a和R 41b各自独立地选自H、C 1-6烷基、C 1-6烷氧基和C 3-8环烷基,或者R 41a和R 41b与它们所连接的N原子一起形成4-7元杂环基,其中所述C 1-6烷基、C 1-6烷氧基、C 3-8环烷基和4-7元杂环基各自任选地被下列基团中的一个或多个取代:OH、CN和NR 31R 32;并且 R 41a and R 41b are each independently selected from H, C 1-6 alkyl, C 1-6 alkoxy and C 3-8 cycloalkyl, or R 41a and R 41b form together with the N atom to which they are attached 4-7 membered heterocyclic group, wherein the C 1-6 alkyl group, C 1-6 alkoxy group, C 3-8 cycloalkyl group and 4-7 membered heterocyclic group are each optionally selected from the following groups One or more of the substitutions: OH, CN and NR 31 R 32 ; and
当多个R 30同时存在时,各R 30可以相同或不同; When multiple R 30 exist at the same time, each R 30 may be the same or different;
当多个R 31同时存在时,各R 31可以相同或不同; When multiple R 31 exist at the same time, each R 31 may be the same or different;
当多个R 32同时存在时,各R 32可以相同或不同; When multiple R 32 exist at the same time, each R 32 may be the same or different;
当多个R 33同时存在时,各R 33可以相同或不同; When multiple R 33 exist at the same time, each R 33 may be the same or different;
当多个R 34同时存在时,各R 34可以相同或不同; When multiple R 34 exist at the same time, each R 34 may be the same or different;
当多个R 35同时存在时,各R 35可以相同或不同; When multiple R 35 exist at the same time, each R 35 may be the same or different;
当多个R 37同时存在时,各R 37可以相同或不同; When multiple R 37 exist at the same time, each R 37 may be the same or different;
当多个R 38同时存在时,各R 38可以相同或不同; When multiple R 38 exist at the same time, each R 38 may be the same or different;
当多个R 39同时存在时,各R 39可以相同或不同; When multiple R 39 exist at the same time, each R 39 may be the same or different;
当多个R 40同时存在时,各R 40可以相同或不同。 When multiple R 40 exist at the same time, each R 40 may be the same or different.
在一些实施方案中,本发明提供具有式II所示结构的化合物,所述化合物的立体异构体、互变异构体或其混合物,所述化合物的药学上可接受的盐、共晶、多晶型物或溶剂合物,或者所述化合物的稳定同位素衍生物、代谢物或前药:In some embodiments, the present invention provides a compound having a structure represented by Formula II, a stereoisomer, a tautomer or a mixture of the compound, a pharmaceutically acceptable salt, co-crystal, Polymorphs or solvates, or stable isotope derivatives, metabolites or prodrugs of said compounds:
其中R 1、L 2和R 3如式I中所定义。 Wherein R 1 , L 2 and R 3 are as defined in formula I.
在一些实施方案中,本发明提供具有式II-A所示结构的化合物,所述化合物的立体异构体、互变异构体或其混合物,所述化合物的药学上可接受的盐、共晶、多晶型物或溶剂合物,或者所述化合物的稳定同位素衍生物、代谢物或前药:In some embodiments, the present invention provides a compound having a structure represented by Formula II-A, a stereoisomer, a tautomer or a mixture of the compound, a pharmaceutically acceptable salt, or a mixture of the compound Crystals, polymorphs or solvates, or stable isotope derivatives, metabolites or prodrugs of said compounds:
其中L 2和R 3如式I中所定义。 Where L 2 and R 3 are as defined in formula I.
在一些实施方案中,本发明提供具有式III所示结构的化合物,所述化合物的立体异构体、互变异构体或其混合物,所述化合物的药学上可接受的盐、共晶、多晶型物或溶剂合物,或者所述化合物的稳定同位素衍生物、代谢物或前药:In some embodiments, the present invention provides a compound having a structure represented by formula III, a stereoisomer, a tautomer or a mixture of the compound, a pharmaceutically acceptable salt, co-crystal, Polymorphs or solvates, or stable isotope derivatives, metabolites or prodrugs of said compounds:
其中:among them:
X 1选自CR 6和N, X 1 is selected from CR 6 and N,
R 1选自C 1-8烷基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基和9-12元芳基并杂环基,其中所述C 1-8烷基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基和9-12元芳基并杂环基各自任选地被下列取代基中的一个或多个取代:卤素、CN、NO 2、C 1-4烷基、C 3-8环烷基、C 1-4卤代烷基、C 1- 4羟烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、9-12元芳基并杂环基、CO 2R 30、C(O)R 30、C(O)NR 31R 32、NR 33C(O)R 34、NR 31R 32、OC(O)R 30、OC(O)NR 31R 32、NR 33C(O)NR 31R 32、NR 33C(O)OR 30、S(O)NR 31R 32、S(O) 2NR 31R 32、S(O)R 35、S(O) 2R 35、OR 37和SR 37; R 1 is selected from C 1-8 alkyl, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl and 9-12 membered aryl and hetero Cyclic group, wherein the C 1-8 alkyl group, C 3-8 cycloalkyl group, 4-10 membered heterocyclic group, C 6-12 aryl group, 5-10 membered heteroaryl group and 9-12 membered aryl group The heterocyclic groups are each optionally substituted by one or more of the following substituents: halogen, CN, NO 2 , C 1-4 alkyl, C 3-8 cycloalkyl, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-10 membered heterocyclyl, C 6-12 aryl, 5-10 membered heteroaryl, 9-12 membered heterocyclyl, and aryl group, CO 2 R 30, C ( O) R 30 , C(O)NR 31 R 32 , NR 33 C(O)R 34 , NR 31 R 32 , OC(O)R 30 , OC(O)NR 31 R 32 , NR 33 C(O)NR 31 R 32 , NR 33 C(O)OR 30 , S(O)NR 31 R 32 , S(O) 2 NR 31 R 32 , S(O)R 35 , S(O) 2 R 35 , OR 37 and SR 37 ;
R 3在每次出现时独立地选自H、卤素、CN、NO 2、C 1-8烷基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、9-12元芳基并杂环基、9-12元芳基并杂芳基、9-12元芳基并环烷基、CO 2R 30、C(O)NR 31R 32、NR 33C(O)R 34、NR 31R 32、S(O) 2R 35、OR 37、SR 37、C(O)R 30、OC(O)R 30、OC(O)NR 31R 32、NR 33C(O)NR 31R 32、NR 33C(O)OR 30、C(=NR 38)NR 31R 32、NR 33C(=NR 38)NR 31R 32、P(R 39) 2、P(OR 39) 2、P(O)R 39R 40、P(O)OR 39OR 30、S(O)R 35、S(O)NR 31R 32和S(O) 2NR 31R 32,其中所述C 1-8烷基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、9-12元芳基并杂环基、9-12元芳基并杂芳基和9-12元芳基并环烷基各自任选地被下列取代基中的一个或多个取代:卤素、CN、NO 2、C 1-4烷基、C 3-8环烷基、C 1-4卤代烷基、C 1-4羟烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、9-12元芳基并杂环基、CO 2R 30、C(O)R 30、C(O)NR 31R 32、NR 33C(O)R 34、NR 31R 32、S(O)R 35、S(O) 2R 35、S(O)NR 31R 32、S(O) 2NR 31R 32、OR 37、SR 37、OC(O)R 30、OC(O)NR 31R 32、NR 33C(O)NR 31R 32、NR 33C(O)OR 30、C(=NR 38)NR 31R 32、NR 33C(=NR 38)NR 31R 32、=NNR 31R 32、P(R 39) 2、P(OR 39) 2、P(O)R 39R 40和P(O)OR 39OR 30; R 3 at each occurrence is independently selected from H, halogen, CN, NO 2 , C 1-8 alkyl, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl, 9-12 membered aryl and heterocyclic group, 9-12 membered aryl and heteroaryl, 9-12 membered aryl and cycloalkyl, CO 2 R 30 , C(O) NR 31 R 32 , NR 33 C(O)R 34 , NR 31 R 32 , S(O) 2 R 35 , OR 37 , SR 37 , C(O)R 30 , OC(O)R 30 , OC(O ) NR 31 R 32 , NR 33 C(O)NR 31 R 32 , NR 33 C(O)OR 30 , C(=NR 38 )NR 31 R 32 , NR 33 C(=NR 38 )NR 31 R 32 , P(R 39 ) 2 , P(OR 39 ) 2 , P(O)R 39 R 40 , P(O)OR 39 OR 30 , S(O)R 35 , S(O)NR 31 R 32 and S( O) 2 NR 31 R 32 , wherein the C 1-8 alkyl group, C 3-8 cycloalkyl group, 4-10 membered heterocyclic group, C 6-12 aryl group, 5-10 membered heteroaryl group, 9 The -12-membered aryl and heterocyclyl, 9-12-membered aryl and heteroaryl, and 9-12-membered aryl and cycloalkyl are each optionally substituted with one or more of the following substituents: halogen, CN , NO 2 , C 1-4 alkyl, C 3-8 cycloalkyl, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5 -10 membered heteroaryl, 9-12 membered aryl and heterocyclic group, CO 2 R 30 , C(O)R 30 , C(O)NR 31 R 32 , NR 33 C(O)R 34 , NR 31 R 32 , S(O)R 35 , S(O) 2 R 35 , S(O)NR 31 R 32 , S(O) 2 NR 31 R 32 , OR 37 , SR 37 , OC(O)R 30 , OC(O)NR 31 R 32 , NR 33 C(O)NR 31 R 32 , NR 33 C(O)OR 30 , C(=NR 38 )NR 31 R 32 , NR 33 C(=NR 38 )NR 31 R 32 , =NNR 31 R 32 , P(R 39 ) 2 , P(OR 39 ) 2 , P(O)R 39 R 40 and P(O)OR 39 OR 30 ;
R 4选自H、NR 41aR 41b、C 1-15烷基、C 1-8烷氧基、C 2-8烯基、C 2-8炔基、C 3-8环烷基和4-10元杂环基,其中所述C 1-15烷基、C 1-8烷氧基、C 2-8烯基、C 2-8炔基、C 3-8环烷基和4-10元杂环基各自任选地被下列取代基中的一个或多个取代:卤素、C 1-4卤代烷基、C 1-4羟烷基、4-7元杂环基、CN、NO 2、OR 37、SR 37、C(O)R 30、C(O)NR 31R 32、NR 33C(O)R 34、C(O)OR 30、OC(O)R 30、OC(O)NR 31R 32、NR 33C(O)NR 31R 32和NR 31R 32; R 4 is selected from H, NR 41a R 41b , C 1-15 alkyl, C 1-8 alkoxy, C 2-8 alkenyl, C 2-8 alkynyl, C 3-8 cycloalkyl and 4- 10-membered heterocyclic group, wherein the C 1-15 alkyl group, C 1-8 alkoxy group, C 2-8 alkenyl group, C 2-8 alkynyl group, C 3-8 cycloalkyl group and 4-10 membered The heterocyclic groups are each optionally substituted by one or more of the following substituents: halogen, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-7 membered heterocyclyl, CN, NO 2 , OR 37 , SR 37 , C(O)R 30 , C(O)NR 31 R 32 , NR 33 C(O)R 34 , C(O)OR 30 , OC(O)R 30 , OC(O)NR 31 R 32 , NR 33 C(O)NR 31 R 32 and NR 31 R 32 ;
R 6选自H、卤素、C 1-6烷基、C 1-4烷氧基、C 3-8环烷基和4-10元杂环基,其中所述C 1-6烷基、C 1-4烷 氧基、C 3-8环烷基和4-10元杂环基各自任选地被下列基团中的一个或多个取代:卤素、OH、CN、C 1-4烷氧基、C 1-4羟烷基和NR 31R 32; R 6 is selected from H, halogen, C 1-6 alkyl, C 1-4 alkoxy, C 3-8 cycloalkyl and 4-10 membered heterocyclic group, wherein the C 1-6 alkyl, C 1-4 alkoxy, C 3-8 cycloalkyl, and 4-10 membered heterocyclic group are each optionally substituted with one or more of the following groups: halogen, OH, CN, C 1-4 alkoxy Group, C 1-4 hydroxyalkyl group and NR 31 R 32 ;
L为-(L 1) n-(L 2) p-(L 3) q-,其中L 1、L 2和L 3相同或不同且各自在每次出现时独立地选自C 1-8亚烷基、C 2-8亚烯基、C 2-8亚炔基、C 1-8亚烷氧基、C 3-8亚环烷基、4-10元亚杂环基、C 6-12亚芳基、5-10元亚杂芳基、O、S、NR 33、S(O)、S(O) 2、C(O)和C(R 36aR 36b),其中所述C 1-8亚烷基、C 2-8亚烯基、C 2-8亚炔基、C 1-8亚烷氧基、C 3-8亚环烷基、4-10元亚杂环基、C 6-12亚芳基和5-10元亚杂芳基各自任选地被下列取代基中的一个或多个取代:卤素、OH、CN、NO 2、C 1-6烷基、C 1-4卤代烷基、C 1-4羟烷基、C 1-4烷氧基和NR 31R 32; L is -(L 1 ) n -(L 2 ) p -(L 3 ) q -, wherein L 1 , L 2 and L 3 are the same or different and each is independently selected from C 1-8 sub Alkyl, C 2-8 alkenylene, C 2-8 alkynylene, C 1-8 alkyleneoxy, C 3-8 cycloalkylene, 4-10 membered heterocyclylene, C 6-12 Arylene, 5-10 membered heteroarylene, O, S, NR 33 , S(O), S(O) 2 , C(O) and C(R 36a R 36b ), wherein the C 1- 8 alkylene, C 2-8 alkenylene, C 2-8 alkynylene, C 1-8 alkyleneoxy, C 3-8 cycloalkylene, 4-10 membered heterocyclylene, C 6 The -12 arylene group and the 5-10 membered heteroarylene group are each optionally substituted with one or more of the following substituents: halogen, OH, CN, NO 2 , C 1-6 alkyl, C 1-4 Haloalkyl, C 1-4 hydroxyalkyl, C 1-4 alkoxy and NR 31 R 32 ;
n、p和q各自在每次出现时独立地为0、1或2;n, p, and q are each independently 0, 1 or 2 each time they appear;
R 30、R 37、R 39和R 40各自独立地选自H、C 1-8烷基(例如C 1-6烷基或C 1-4烷基)、C 1-8烷氧基(例如C 1- 6烷氧基或C 1-4烷氧基)、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、-C 1-8烷基-C 6-12芳基和-C 1-8烷基-(5-10元杂芳基),其中所述C 1-8烷基、C 1-8烷氧基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、-C 1-8烷基-C 6-12芳基和-C 1-8烷基-(5-10元杂芳基)各自任选地被下列取代基中的一个或多个取代:OH、CN、NO 2、C 1-4烷基、C 1-4烷氧基、C 1-4卤代烷基、卤素、C 1-4卤代烷氧基、CO 2(C 1-6烷基)、CONR 31R 32、NR 31R 32、NR 33C(O)R 34、S(O)R 35、S(O) 2R 35、S(O)NR 31R 32和S(O) 2NR 31R 32; R 30 , R 37 , R 39 and R 40 are each independently selected from H, C 1-8 alkyl (e.g. C 1-6 alkyl or C 1-4 alkyl), C 1-8 alkoxy (e.g. C 1- 6 alkoxy or C 1-4 alkoxy), C 3-8 cycloalkyl, 4-10 membered heterocyclyl, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 alkyl-(5-10 membered heteroaryl), wherein the C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 Alkyl-(5-10 membered heteroaryl) are each optionally substituted with one or more of the following substituents: OH, CN, NO 2 , C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, halogen, C 1-4 haloalkoxy, CO 2 (C 1-6 alkyl), CONR 31 R 32 , NR 31 R 32 , NR 33 C(O)R 34 , S(O ) R 35 , S(O) 2 R 35 , S(O)NR 31 R 32 and S(O) 2 NR 31 R 32 ;
R 31、R 32、R 33和R 34各自独立地选自H、C 1-8烷基、C 1-8烷氧基、C 3-8环烷基、4-10元杂环基、C 6-12芳基和5-10元杂芳基,或者R 31和R 32与它们所连接的N原子一起形成4-8元杂环基,或者R 33和R 34与它们各自所连接的C和N原子一起形成4-8元杂环基,其中所述C 1-8烷基、C 1-8烷氧基、C 3-8环烷基、4-8元杂环基、4-10元杂环基、C 6-12芳基和5-10元杂芳基各自任选地被下列取代基中的一个或多个取代:OH、CN、卤素、NO 2、C 1-4烷基、C 1-4烷氧基、C 1-4羟烷基、C 1-4卤代烷基、C 1-4卤代烷氧基、4-10元杂环基、C 6-12芳基和5-10元杂芳基; R 31 , R 32 , R 33 and R 34 are each independently selected from H, C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl and 5-10 membered heteroaryl, or R 31 and R 32 together with the N atom to which they are attached form a 4-8 membered heterocyclic group, or R 33 and R 34 and the C to which they are each attached Together with the N atom to form a 4-8 membered heterocyclic group, wherein the C 1-8 alkyl group, C 1-8 alkoxy group, C 3-8 cycloalkyl group, 4-8 membered heterocyclic group, 4-10 Each of the membered heterocyclic group, C 6-12 aryl group and 5-10 membered heteroaryl group is optionally substituted by one or more of the following substituents: OH, CN, halogen, NO 2 , C 1-4 alkyl , C 1-4 alkoxy, C 1-4 hydroxyalkyl, C 1-4 haloalkyl, C 1-4 haloalkoxy, 4-10 membered heterocyclic group, C 6-12 aryl and 5-10 Yuan heteroaryl
R 35选自C 1-8烷基、C 1-8烷氧基、C 3-8环烷基、4-10元杂环基、C 6-12芳基、5-10元杂芳基、-C 1-8烷基-C 6-12芳基和-C 1-8烷基-(5-10元杂芳基),其中所述C 1-8烷基、C 1-8烷氧基、C 3-8环烷基、4-10元杂环基、C 6-12芳基和5-10元杂芳基各自任选地被下列取代基中的一个或多个取代:OH、CN、NO 2、C 1-4烷基、C 1-4烷氧基、C 1-4卤代烷基、卤素、C 1-4卤代烷氧基、CO 2(C 1-6烷基)、CONR 31R 32、NR 31R 32、NR 33C(O)R 34、S(O)Me、S(O) 2Me、S(O)NR 31R 32和S(O) 2NR 31R 32,其中R 31、R 32、R 33和R 34如上所定义; R 35 is selected from C 1-8 alkyl, C 1-8 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6-12 aryl, 5-10 membered heteroaryl, -C 1-8 alkyl-C 6-12 aryl and -C 1-8 alkyl-(5-10 membered heteroaryl), wherein the C 1-8 alkyl, C 1-8 alkoxy , C 3-8 cycloalkyl, 4-10 membered heterocyclyl, C 6-12 aryl and 5-10 membered heteroaryl are each optionally substituted by one or more of the following substituents: OH, CN , NO 2 , C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, halogen, C 1-4 haloalkoxy, CO 2 (C 1-6 alkyl), CONR 31 R 32 , NR 31 R 32 , NR 33 C(O)R 34 , S(O)Me, S(O) 2 Me, S(O)NR 31 R 32 and S(O) 2 NR 31 R 32 , where R 31 , R 32 , R 33 and R 34 are as defined above;
R 36a和R 36b相同或不同且各自独立地选自H、C 1-8烷基和C 1-8烷氧基,其中所述C 1-8烷基和C 1-8烷氧基各自任选地被下列基团中的一个或多个取代:OH、CN、卤素、NH 2、NHCH 3和N(CH 3) 2,或者R 36a和R 36b与它们所连接的C原子一起形成3-7元环烷基或杂环基; R 36a and R 36b are the same or different and are each independently selected from H, C 1-8 alkyl and C 1-8 alkoxy, wherein the C 1-8 alkyl and C 1-8 alkoxy are each independently Optionally substituted by one or more of the following groups: OH, CN, halogen, NH 2 , NHCH 3 and N(CH 3 ) 2 , or R 36a and R 36b together with the C atom to which they are attached form a 3- 7-membered cycloalkyl or heterocyclic group;
R 38选自H、OH、CN、NO 2、S(O)R 35和S(O) 2R 35; R 38 is selected from H, OH, CN, NO 2 , S(O)R 35 and S(O) 2 R 35 ;
R 41a和R 41b各自独立地选自H、C 1-6烷基、C 1-6烷氧基和C 3-8环烷基,或者R 41a和R 41b与它们所连接的N原子一起形成4-7元杂环基,其中所述C 1-6烷基、C 1-6烷氧基、C 3-8环烷基和4-7元杂环基可任选地被下列基团中的一个或多个取代:OH、CN和NR 31R 32;并且 R 41a and R 41b are each independently selected from H, C 1-6 alkyl, C 1-6 alkoxy and C 3-8 cycloalkyl, or R 41a and R 41b form together with the N atom to which they are attached 4-7 membered heterocyclic group, wherein the C 1-6 alkyl group, C 1-6 alkoxy group, C 3-8 cycloalkyl group and 4-7 membered heterocyclic group may optionally be selected from the following groups One or more of the substitutions: OH, CN and NR 31 R 32 ; and
当多个R 30同时存在时,各R 30可以相同或不同; When multiple R 30 exist at the same time, each R 30 may be the same or different;
当多个R 31同时存在时,各R 31可以相同或不同; When multiple R 31 exist at the same time, each R 31 may be the same or different;
当多个R 32同时存在时,各R 32可以相同或不同; When multiple R 32 exist at the same time, each R 32 may be the same or different;
当多个R 33同时存在时,各R 33可以相同或不同; When multiple R 33 exist at the same time, each R 33 may be the same or different;
当多个R 34同时存在时,各R 34可以相同或不同; When multiple R 34 exist at the same time, each R 34 may be the same or different;
当多个R 35同时存在时,各R 35可以相同或不同; When multiple R 35 exist at the same time, each R 35 may be the same or different;
当多个R 37同时存在时,各R 37可以相同或不同; When multiple R 37 exist at the same time, each R 37 may be the same or different;
当多个R 38同时存在时,各R 38可以相同或不同; When multiple R 38 exist at the same time, each R 38 may be the same or different;
当多个R 39同时存在时,各R 39可以相同或不同; When multiple R 39 exist at the same time, each R 39 may be the same or different;
当多个R 40同时存在时,各R 40可以相同或不同。 When multiple R 40 exist at the same time, each R 40 may be the same or different.
在一些实施方案中,本发明提供具有式IV所示结构的化合物,所述化合物的立体异构体、互变异构体或其混合物,所述化合物的药学上可接受的盐、共晶、多晶型物或溶剂合物,或者所述化合物的稳定同位素衍生物、代谢物或前药:In some embodiments, the present invention provides a compound having a structure represented by formula IV, stereoisomers, tautomers or mixtures thereof, pharmaceutically acceptable salts, co-crystals, Polymorphs or solvates, or stable isotope derivatives, metabolites or prodrugs of said compounds:
其中R 1、L 2和R 3如式III中所定义。 Wherein R 1 , L 2 and R 3 are as defined in formula III.
在一些实施方案中,本发明提供具有式IV-A所示结构的化合物,所述化合物的立体异构体、互变异构体或其混合物,所述化合物的药学上可接受的盐、共晶、多晶型物或溶剂合物,或者所述化合物的稳定同位素衍生物、代谢物或前药:In some embodiments, the present invention provides a compound having a structure represented by formula IV-A, a stereoisomer, a tautomer of the compound, or a mixture thereof, a pharmaceutically acceptable salt, or a mixture of the compound Crystals, polymorphs or solvates, or stable isotope derivatives, metabolites or prodrugs of said compounds:
其中L 2和R 3如式III中所定义。 Where L 2 and R 3 are as defined in formula III.
在以上各通式中,R 3在每次出现时优选独立地选自H、卤素、CN、C 1-6烷基、C 3-6环烷基、4-7元杂环基、C 6-10芳基、5-6元杂芳基、CO 2R 30、C(O)NR 31R 32、NR 33C(O)R 34、NR 31R 32、S(O) 2R 35、OR 37、SR 37、C(O)R 30、OC(O)R 30、NR 33C(O)NR 31R 32和S(O) 2NR 31R 32,其中所述C 1-6烷基、C 3-6环烷基、4-7元杂环基、C 6-10芳基和5-6元杂芳基各自任选地被下列取代基中的一个或多个取代:卤素、CN、C 1-4烷基、C 3-6环烷基、C 1-4卤代烷基、C 1-4羟烷基、4-7元杂环基、C 6-10芳基、5-6元杂芳基、CO 2R 30、C(O)R 30、C(O)NR 31R 32、NR 33C(O)R 34、NR 31R 32、S(O) 2R 35、S(O) 2NR 31R 32、OR 37、SR 37和NR 33C(O)NR 31R 32; In the above general formulas, R 3 is preferably independently selected from H, halogen, CN, C 1-6 alkyl, C 3-6 cycloalkyl, 4-7 membered heterocyclic group, C 6 each occurrence -10 aryl, 5-6 membered heteroaryl, CO 2 R 30 , C(O)NR 31 R 32 , NR 33 C(O)R 34 , NR 31 R 32 , S(O) 2 R 35 , OR 37 , SR 37 , C(O)R 30 , OC(O)R 30 , NR 33 C(O)NR 31 R 32 and S(O) 2 NR 31 R 32 , wherein the C 1-6 alkyl group, C 3-6 cycloalkyl, 4-7 membered heterocyclyl, C 6-10 aryl and 5-6 membered heteroaryl are each optionally substituted with one or more of the following substituents: halogen, CN, C 1-4 alkyl, C 3-6 cycloalkyl, C 1-4 haloalkyl, C 1-4 hydroxyalkyl, 4-7 membered heterocyclic group, C 6-10 aryl, 5-6 membered hetero Aryl, CO 2 R 30 , C(O)R 30 , C(O)NR 31 R 32 , NR 33 C(O)R 34 , NR 31 R 32 , S(O) 2 R 35 , S(O) 2 NR 31 R 32 , OR 37 , SR 37 and NR 33 C(O)NR 31 R 32 ;
优选地,R 3在每次出现时独立地选自H、CN、C 1-6烷基、C 3-6环烷基、4-7元杂环基、C 6-10芳基、5-6元杂芳基、NR 33C(O)R 34、OR 37、SR 37、C(O)R 30和OC(O)R 30,其中所述C 1-6烷基、C 3-6环烷基、4-7元杂环基、C 6-10芳基和5-6元杂芳基各自任选地被下列取代基中的一个或多个取代:CN、C 1-4烷基和CO 2R 30,R 30和R 37各自独立地选自H、C 1-6烷基、C 1-6烷氧基、C 3-8环烷基、4-10元杂环基、C 6-12芳基和5-10元杂芳基,其中所述C 1-6烷基、C 1-6烷氧基、C 3-8环烷基、4-10元杂环基、C 6-12芳基和5-10元杂芳基各自任选地被下列取代基中的一个或多个取代:OH、CN、NO 2、C 1-4烷基、C 1-4烷氧基、C 1-4卤代烷基、卤素和C 1-4卤代烷氧基,并且R 33和R 34各自独立地选自H、C 1-6烷基、C 1-6烷氧基和C 3-6环烷基,其中所述C 1-6烷基、C 1-6烷氧基和C 3-6环烷基各自任选地被下列取代基中的一个或多个取代:OH、CN、卤素、NO 2、C 1-4烷基、C 1-4烷氧基、C 1-4羟烷基、C 1-4卤代烷基、C 1-4卤代烷氧基、4-10元杂环基、C 6- 12芳基和5-10元杂芳基; Preferably, each occurrence of R 3 is independently selected from H, CN, C 1-6 alkyl, C 3-6 cycloalkyl, 4-7 membered heterocyclyl, C 6-10 aryl, 5- 6-membered heteroaryl, NR 33 C(O)R 34 , OR 37 , SR 37 , C(O)R 30 and OC(O)R 30 , wherein the C 1-6 alkyl group, C 3-6 ring The alkyl group, 4-7 membered heterocyclic group, C 6-10 aryl group and 5-6 membered heteroaryl group are each optionally substituted with one or more of the following substituents: CN, C 1-4 alkyl and CO 2 R 30 , R 30 and R 37 are each independently selected from H, C 1-6 alkyl, C 1-6 alkoxy, C 3-8 cycloalkyl, 4-10 membered heterocyclic group, C 6 -12 aryl group and 5-10 membered heteroaryl group, wherein the C 1-6 alkyl group, C 1-6 alkoxy group, C 3-8 cycloalkyl group, 4-10 membered heterocyclic group, C 6- 12 aryl and 5-10 membered heteroaryl are each optionally substituted by one or more of the following substituents: OH, CN, NO 2 , C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, halogen and C 1-4 haloalkoxy, and R 33 and R 34 are each independently selected from H, C 1-6 alkyl, C 1-6 alkoxy and C 3-6 cycloalkane Group, wherein the C 1-6 alkyl group, C 1-6 alkoxy group and C 3-6 cycloalkyl group are each optionally substituted with one or more of the following substituents: OH, CN, halogen, NO 2. C 1-4 alkyl, C 1-4 alkoxy, C 1-4 hydroxyalkyl, C 1-4 haloalkyl, C 1-4 haloalkoxy, 4-10 membered heterocyclic group, C 6 --12 aryl and 5-10 membered heteroaryl;
更优选地,R 3在每次出现时独立地选自H、OH、CN、N(CH 3)C(O)CH 3、N(CH 3)C(O)CH 2CH 3、N(CH 3)C(O)-环丙基和各自任选地被羟基、甲基和CN中的一个或多个取代的下列基团:甲基、乙基、丙基、环丙基、环丁基、环戊基、四氢呋喃基、吗啉代、四氢吡喃基、异噁唑基、吡唑基和哌啶基。 More preferably, R 3 is independently selected from H, OH, CN, N(CH 3 )C(O)CH 3 , N(CH 3 )C(O)CH 2 CH 3 , N(CH 3 ) C(O)-cyclopropyl and the following groups each optionally substituted with one or more of hydroxy, methyl and CN: methyl, ethyl, propyl, cyclopropyl, cyclobutyl , Cyclopentyl, tetrahydrofuranyl, morpholino, tetrahydropyranyl, isoxazolyl, pyrazolyl and piperidinyl.
在以上各通式中,优选地,L不存在或者为-L 1-L 2-,其中L 1为NH或N(CH 3),且L 2不存在或者选自C 1-4亚烷基、C 1-4亚烷氧基和C 3-6亚环烷基。 In the above general formulas, preferably, L does not exist or is -L 1 -L 2 -, wherein L 1 is NH or N(CH 3 ), and L 2 does not exist or is selected from C 1-4 alkylene , C 1-4 alkyleneoxy and C 3-6 cycloalkylene.
在以上各通式中,优选地,L不存在或者为-L 1-L 2-,其中L 1为NH或N(CH 3),且L 2不存在或者选自亚甲基、亚乙基、亚丙基、亚丁基、亚甲氧基、亚乙氧基、亚正丙氧基、亚异丙氧基、亚正丁氧基、亚异丁氧基、亚叔丁氧基、亚环丙基、亚环丁基、亚环戊基和亚环己基。 In the above general formulas, preferably, L does not exist or is -L 1 -L 2 -, where L 1 is NH or N(CH 3 ), and L 2 does not exist or is selected from methylene, ethylene , Propylene, butylene, methyleneoxy, ethyleneoxy, n-propoxy, isopropoxy, n-butoxy, isobutoxy, tert-butoxy, ring Propyl, cyclobutylene, cyclopentylene and cyclohexylene.
在以上各通式中,-L-R 3或-NH-L 2-R 3优选选自: In the above general formulas, -LR 3 or -NH-L 2 -R 3 is preferably selected from:
在本发明的某些实施方案中,本发明的化合物及其药学上可接受的盐包括,但不限于:In certain embodiments of the present invention, the compounds of the present invention and pharmaceutically acceptable salts thereof include, but are not limited to:
组合物、制剂和用途Composition, preparation and use
在另一方面,本发明提供药物组合物,其包含如上所述的化合物,所述化合物的立体异构体、互变异构体或其混合物,所述化合物的药学上可接受的盐、共晶、多晶型物或溶剂合物,或者所述化合物的稳定同位素衍生物、代谢物或前药。任选地,所述药物组合物还包含一种或多种药学上可接受的载体。In another aspect, the present invention provides a pharmaceutical composition comprising a compound as described above, a stereoisomer, a tautomer of the compound, or a mixture thereof, a pharmaceutically acceptable salt, or a mixture of the compound Crystals, polymorphs or solvates, or stable isotope derivatives, metabolites or prodrugs of said compounds. Optionally, the pharmaceutical composition further comprises one or more pharmaceutically acceptable carriers.
在一些实施方案中,所述药物组合物用于预防和/或治疗与NLRP3炎症小体活性相关的疾病(例如肿瘤疾病)。In some embodiments, the pharmaceutical composition is used to prevent and/or treat diseases related to NLRP3 inflammasome activity (e.g., tumor diseases).
在另一方面,本发明提供药物制剂,其包含如上所述的化合物,所述化合物的立体异构体、互变异构体或其混合物,所述化合物的药学上可接受的盐、共晶、多晶型物或溶剂合物,或者所述化合物的稳定同位素衍生物、代谢物或前药,或者包含如上所述的药物组合物。In another aspect, the present invention provides a pharmaceutical preparation, which comprises the compound as described above, a stereoisomer, a tautomer of the compound, or a mixture thereof, a pharmaceutically acceptable salt, a co-crystal of the compound , Polymorphs or solvates, or stable isotope derivatives, metabolites or prodrugs of the compound, or comprise the above-mentioned pharmaceutical composition.
在另一方面,本发明提供如上所述的化合物,所述化合物的立体异构体、互变异构体或其混合物,所述化合物的药学上可接受的盐、共晶、多晶型物或溶剂合物,所述化合物的稳定同位素衍生物、代谢物或前药,或者如上所述的药物组合物用于制备药物的用途,所述药物用于预防和/或治疗与NLRP3炎症小体活性相关的疾病(例如肿瘤疾病)。In another aspect, the present invention provides a compound as described above, a stereoisomer, a tautomer or a mixture of the compound, a pharmaceutically acceptable salt, co-crystal, and polymorph of the compound Or solvates, stable isotope derivatives, metabolites or prodrugs of the compounds, or the use of the above-mentioned pharmaceutical composition for the preparation of medicines for the prevention and/or treatment of NLRP3 inflammasomes Activity-related diseases (e.g. tumor diseases).
在另一方面,本发明提供如上所述的化合物,所述化合物的立体异构体、互变异构体或其混合物,所述化合物的药学上可接受的盐、共晶、多晶型物或溶剂合物,所述化合物的稳定同位素衍生物、代谢物或前药,或者如上所述的药物组合物,其用于预防和/或治疗与NLRP3炎症小体活性相关的疾病(例如肿瘤疾病)。In another aspect, the present invention provides a compound as described above, a stereoisomer, a tautomer or a mixture of the compound, a pharmaceutically acceptable salt, co-crystal, and polymorph of the compound Or solvates, stable isotope derivatives, metabolites or prodrugs of the compounds, or pharmaceutical compositions as described above, which are used to prevent and/or treat diseases related to NLRP3 inflammasome activity (such as tumor diseases) ).
在另一方面,本发明提供如上所述的化合物,所述化合物的立体异构体、互变异构体或其混合物,所述化合物的药学上可接受的盐、共晶、多晶型物或溶剂合物,所述化合物的稳定同位素衍生物、代谢物或前药,或者如上所述的药物组合物用于制备制剂的用途,所述制剂用于调节(例如增加)NLRP3炎症小体的活性。In another aspect, the present invention provides a compound as described above, a stereoisomer, a tautomer or a mixture of the compound, a pharmaceutically acceptable salt, co-crystal, and polymorph of the compound Or solvates, stable isotope derivatives, metabolites or prodrugs of the compounds, or the use of the pharmaceutical composition as described above for the preparation of preparations for regulating (for example, increasing) NLRP3 inflammasome active.
在一些实施方案中,所述制剂被施用至受试者(例如哺乳动物,包括例如牛科动物、马科动物、羊科动物、猪科动物、犬科动物、猫科动物、啮齿类动物、灵长类动物如人)体内,以增加受试者体内细胞中的NLRP3炎症小体活性。或者,所述制剂被施用至体外细胞(例如细胞系或者来自受试者的细胞),以增加细胞中NLRP3炎症小体的活性。In some embodiments, the formulation is administered to a subject (e.g., a mammal, including, for example, bovine, equine, ovine, swine, canine, feline, rodent, Primates (such as humans) in vivo to increase the NLRP3 inflammasome activity in the cells of the subject. Alternatively, the formulation is administered to cells in vitro (e.g., cell lines or cells from a subject) to increase the activity of NLRP3 inflammasomes in the cells.
在另一方面,本发明提供调节(例如增加)细胞中NLRP3炎症小体活性的方法,其包括给所述细胞施用有效量的如上所述的化合物,所述化合物的立体异构体、互变异构体或其混合物,所述化合物的药学上可接受的盐、共晶、多晶型物或溶剂合物,所述化合物的稳定同位素衍生物、代谢物或前药,如上所述的药物组合物,或者如上所述的药物制剂。In another aspect, the present invention provides a method for modulating (e.g. increasing) the activity of NLRP3 inflammasome in a cell, which comprises administering to the cell an effective amount of a compound as described above, stereoisomers, tautomers of the compound Isomers or mixtures thereof, pharmaceutically acceptable salts, co-crystals, polymorphs or solvates of said compounds, stable isotope derivatives, metabolites or prodrugs of said compounds, drugs as described above Composition, or pharmaceutical formulation as described above.
在另一方面,本发明提供用于调节(例如增加)NLRP3炎症小体的活性的药盒,所述药盒包含如上所述的化合物,所述化合物的立体异构体、互变异构体或其混合物,所述化合物的药学上可接受的盐、共晶、多晶型物或溶剂合物,所述化合物的稳定同位素衍生物、代谢物或前药,如上所述的药物组合物,或者如上所述的药物制剂。In another aspect, the present invention provides a kit for modulating (for example, increasing) the activity of NLRP3 inflammasomes, the kit comprising a compound as described above, stereoisomers and tautomers of the compound Or a mixture thereof, a pharmaceutically acceptable salt, co-crystal, polymorph or solvate of the compound, a stable isotope derivative, metabolite or prodrug of the compound, the pharmaceutical composition as described above, Or a pharmaceutical formulation as described above.
在另一方面,本发明提供治疗和/或预防与NLRP3炎症小体活性相关的疾病(例如肿瘤疾病)的方法,其包括给有此需要的受试者施用治疗和/或预防有效量的如上所述的化合物,所述化合物的立体异构体、互变异构体或其混合物,所述化合物的药学上可接受的盐、共晶、多晶型物或溶剂合物,所述化合物的稳定同位素衍生物、代谢物或前药,如上所述的药物组合物,或者如上所述的药物制剂。In another aspect, the present invention provides a method for treating and/or preventing diseases (such as tumor diseases) associated with NLRP3 inflammasome activity, which comprises administering to a subject in need thereof a therapeutically and/or preventively effective amount of the above The compound, the stereoisomer, tautomer or mixture of the compound, the pharmaceutically acceptable salt, co-crystal, polymorph or solvate of the compound, Stable isotope derivatives, metabolites or prodrugs, pharmaceutical compositions as described above, or pharmaceutical formulations as described above.
本发明中,所述肿瘤疾病包括但不限于:脑瘤、肺癌、鳞状上皮细胞癌、膀胱癌、胃癌、卵巢癌、腹膜癌、胰腺癌、乳腺癌、头颈癌、子宫颈癌、子宫内膜癌、直肠癌、肝癌、肾癌、食管腺癌、食管鳞状细胞癌、前列腺癌、雌性生殖道癌、原位癌、淋巴瘤、神经纤维瘤、甲状腺癌、骨癌、皮肤癌、脑癌、结肠癌、睾丸癌、胃肠道间质瘤、前列腺肿瘤、肥大细胞肿瘤、多发性骨髓瘤、黑色素瘤、胶质瘤或肉瘤。In the present invention, the tumor diseases include, but are not limited to: brain tumor, lung cancer, squamous cell carcinoma, bladder cancer, gastric cancer, ovarian cancer, peritoneal cancer, pancreatic cancer, breast cancer, head and neck cancer, cervical cancer, intrauterine cancer Membranous cancer, rectal cancer, liver cancer, kidney cancer, esophageal adenocarcinoma, esophageal squamous cell carcinoma, prostate cancer, female reproductive tract cancer, carcinoma in situ, lymphoma, neurofibroma, thyroid cancer, bone cancer, skin cancer, brain Cancer, colon cancer, testicular cancer, gastrointestinal stromal tumor, prostate tumor, mast cell tumor, multiple myeloma, melanoma, glioma or sarcoma.
在一些实施方案中,本发明的化合物为NLRP3全激动剂(full agonists)。在一些实施方案中,本发明的化合物为NLRP3部分激动剂(partial agonists)。In some embodiments, the compounds of the invention are NLRP3 full agonists. In some embodiments, the compounds of the invention are NLRP3 partial agonists.
发明详述Detailed description of the invention
术语定义Definition of Terms
除非在下文中另有定义,本文中所用的所有技术术语和科学术语的含义意图与本领域技术人员通常所理解的相同。提及本文中使用的技术意图指在本领域中通常所理解的技术,包括那些对本领域技术人员显而易见的技术的变化或等效技术的替换。虽然相信以下术语对于本领域技术人员很好理解,但仍然阐述以下定义以更好地解释本发明。Unless otherwise defined below, the meanings of all technical and scientific terms used herein are intended to be the same as those commonly understood by those skilled in the art. The reference to the technology used herein is intended to refer to the technology generally understood in the art, including those technical changes or equivalent technology substitutions that are obvious to those skilled in the art. Although it is believed that the following terms are well understood by those skilled in the art, the following definitions are still set forth to better explain the present invention.
术语“激动剂(agonist)”指的是一种与受体结合并将其激活而引发下游的生物学效应或应答的化合物,包括全激动剂(full agonist)和部分激动剂(partial agonist)。全激动剂能激活受体并产生最大的效应(maximal effect或E max)。部分激动剂可以和受体结合并将其激活,但相对于完全激动剂而言,只产生部分的效应(partial effect)。当全激动剂和部分激动剂共存时,部分激动剂有时可以通过与全激动剂竞争受体上的结合位点或其他机制而成为部分拮抗剂。一个部分激动剂的效力(potency,可由EC 50(产生50%的E max时的化合物浓度)衡量)有可能高于或低于一个全激动剂的效力。本发明的NLRP3激动剂包括NLRP3全激动剂和NLRP3部分激动剂。 The term "agonist" refers to a compound that binds to a receptor and activates it to trigger a downstream biological effect or response, including full agonist and partial agonist. Full agonists can activate the receptor and produce the greatest effect (maximal effect or E max ). A partial agonist can bind to the receptor and activate it, but compared to a full agonist, it only produces a partial effect. When a full agonist and a partial agonist coexist, the partial agonist can sometimes become a partial antagonist by competing with the full agonist for binding sites on the receptor or by other mechanisms. Efficacy (potency, when by EC 50 (producing 50% of the concentration of compound E max) measure) a possible partial agonist potency higher or lower than a full agonist. The NLRP3 agonists of the present invention include NLRP3 full agonists and NLRP3 partial agonists.
术语“NLRP3”的全称是NLR family pyrin domain containing 3,是一种炎症小体。本发明中,当提及“NLRP3”时,所指的含义包括NLRP3的核酸、多聚核苷酸、寡核苷酸、正义和反义多聚核苷酸链、互补序列、短肽、多肽、蛋白、同源或异源分子、亚型、前体、突变体、变体、衍生物、各种剪接体、等位基因、不同种属以及活化片段等。The full name of the term "NLRP3" is NLR family pyrin domain containing 3, which is an inflammasome. In the present invention, when referring to "NLRP3", the meaning includes NLRP3 nucleic acid, polynucleotide, oligonucleotide, sense and antisense polynucleotide chain, complementary sequence, short peptide, polypeptide , Proteins, homologous or heterologous molecules, subtypes, precursors, mutants, variants, derivatives, various splicing bodies, alleles, different species and activation fragments, etc.
术语“包括”、“包含”、“具有”、“含有”或“涉及”及其在本文中的其它变体形式为包含性的(inclusive)或开放式的,且不排除其它未列举的元素或方法步骤。The terms "including", "including", "having", "containing" or "involving" and other variants herein are inclusive or open-ended, and do not exclude other unlisted elements Or method steps.
术语“卤代”是指被卤素原子所取代,所述“卤素”包括F、Cl、Br或I。The term "halo" refers to substitution by a halogen atom, and the "halogen" includes F, Cl, Br, or I.
术语“烷基”为直链或支链的饱和脂肪烃基。术语“C 1-15烷基”、“C 1-8烷基”、“C 1-6烷基”和“C 1-4烷基”分别指具有1至15个碳原子、1至8个碳原子、1至6个碳原子和1-4个碳原子的直链或支链烷基,例如甲基、乙基、正丙基、异丙基、正丁基、异丁基、仲丁基、叔丁基、正戊基或正己基。所述烷基可以任选地被一个或多个(诸如1至3个)相同或不同的取代基取代。 The term "alkyl" refers to a linear or branched saturated aliphatic hydrocarbon group. The terms "C 1-15 alkyl", "C 1-8 alkyl", "C 1-6 alkyl" and "C 1-4 alkyl" respectively refer to having 1 to 15 carbon atoms, 1 to 8 Carbon atoms, linear or branched alkyl groups of 1 to 6 carbon atoms and 1-4 carbon atoms, such as methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl Group, tert-butyl, n-pentyl or n-hexyl. The alkyl group may be optionally substituted with one or more (such as 1 to 3) substituents which are the same or different.
术语“亚烷基”指从直链或支链的饱和烃基中去掉两个氢原子所得到的饱和的二价烃基,其含有指定的碳原子数目。例如术语“C 1-8亚烷基”指具有1至8个碳原子的亚烷基,例如亚甲基(-CH 2-)、亚乙基(-CH 2CH 2-)、亚异丙基(-CH(CH 3)CH 2-)等。所述亚烷基可以任选地被一个或多个(诸如1至3个)相同或不同的取代基取代。 The term "alkylene" refers to a saturated divalent hydrocarbon group obtained by removing two hydrogen atoms from a linear or branched saturated hydrocarbon group, which contains the specified number of carbon atoms. For example, the term "C 1-8 alkylene" refers to an alkylene group having 1 to 8 carbon atoms, such as methylene (-CH 2 -), ethylene (-CH 2 CH 2 -), isopropylidene Group (-CH(CH 3 )CH 2 -) and so on. The alkylene group may be optionally substituted with one or more (such as 1 to 3) substituents which are the same or different.
术语“卤代烷基”是指被一个或多个(诸如1至3个)相同或不同的卤素原子取代的烷基。术语“C 1-8卤代烷基”、“C 1-6卤代烷基”和“C 1-4卤代烷基”分别指具有1至8个碳原子、1至6个碳原子和1-4个碳原子的卤代烷基,例如-CF 3、-C 2F 5、-CHF 2、-CH 2F、-CH 2CF 3、-CH 2Cl、-CH 2CH 2CF 3等。 The term "haloalkyl" refers to an alkyl group substituted with one or more (such as 1 to 3) identical or different halogen atoms. The terms "C 1-8 haloalkyl", "C 1-6 haloalkyl" and "C 1-4 haloalkyl" refer to having 1 to 8 carbon atoms, 1 to 6 carbon atoms and 1-4 carbon atoms, respectively For example, -CF 3 , -C 2 F 5 , -CHF 2 , -CH 2 F, -CH 2 CF 3 , -CH 2 Cl, -CH 2 CH 2 CF 3 and the like.
术语“羟烷基”是指烷基的氢原子被一个或多个羟基取代所形成的基团,例如C 1-4羟烷基或C 1-3羟烷基,其实例包括但不限于羟甲基、羟乙基、羟丙基、羟丁基、-CH(OH)CH 3等。 The term "hydroxyalkyl" refers to a group formed by replacing the hydrogen atom of an alkyl group with one or more hydroxy groups, such as a C 1-4 hydroxyalkyl group or a C 1-3 hydroxyalkyl group, examples of which include but are not limited to hydroxy Methyl, hydroxyethyl, hydroxypropyl, hydroxybutyl, -CH(OH)CH 3 and so on.
术语“烯基”是指含有一个或多个碳-碳双键的单价直链或支链烃基,例如-CH=CH 2、-CH 2CH=CH 2、-C(CH 3)=CH 2、-CH 2-CH=CH-CH 3等。所述烯基可以任选地被一个或多个(诸如1至3个)相同或不同的取代基取代。 The term "alkenyl" refers to a monovalent linear or branched hydrocarbon group containing one or more carbon-carbon double bonds, such as -CH=CH 2 , -CH 2 CH=CH 2 , -C(CH 3 )=CH 2 , -CH 2 -CH=CH-CH 3 and so on. The alkenyl group may be optionally substituted with one or more (such as 1 to 3) substituents which are the same or different.
术语“亚烯基”指含有一个或多个碳-碳双键的二价直链或支链脂肪烃基团,其含有指定的碳原子数目,例如2至8个碳原子,例如-CH=CH-、-CH 2CH=CH-、-C(CH 3)=CH-等。所述亚烯基可以任选地被一个或多个(诸如1至3个)相同或不同的取代基取代。 The term "alkenylene" refers to a divalent linear or branched aliphatic hydrocarbon group containing one or more carbon-carbon double bonds, which contains the specified number of carbon atoms, for example, 2 to 8 carbon atoms, such as -CH=CH -, -CH 2 CH=CH-, -C(CH 3 )=CH-, etc. The alkenylene group may be optionally substituted with one or more (such as 1 to 3) substituents which are the same or different.
术语“炔基”是指具有一个或多个碳-碳三键的单价直链或支链烃基,包括但不限于乙炔基、2-丙炔基、2-丁炔基和1,3-丁二炔基等。所述炔基可以任选地被一个或多个(诸如1至3个)相同或不同的取代基取代。The term "alkynyl" refers to a monovalent linear or branched hydrocarbon group with one or more carbon-carbon triple bonds, including but not limited to ethynyl, 2-propynyl, 2-butynyl and 1,3-butynyl Dialkynyl and so on. The alkynyl group may be optionally substituted with one or more (such as 1 to 3) substituents which are the same or different.
术语“亚炔基”是指具有一个或多个碳-碳三键的二价直链或支链烃基,其含有指定的碳原子数目,例如2至8个碳原子,包括但不限于 等。所述亚炔基可以任选地被一个或多个(诸如1至3个)相同或不同的取代基取代。 The term "alkynylene" refers to a divalent straight or branched chain hydrocarbon group with one or more carbon-carbon triple bonds, which contains the specified number of carbon atoms, such as 2 to 8 carbon atoms, including but not limited to Wait. The alkynylene group may be optionally substituted with one or more (such as 1 to 3) substituents which are the same or different.
术语“烷氧基”意指在烷基(如上文所定义)任意合理的位置插入氧原子的基团,例如为C 1-8烷氧基、C 1-6烷氧基、C 1-4烷氧基或C 1-3烷氧基。C 1-6烷氧基的代表性实例包括但不限于甲氧基、乙氧基、正丙氧基、异丙氧基、正丁氧基、异丁氧基、叔丁氧基、戊氧基、己氧基、-CH 2-OCH 3等。所述烷氧基可以任选地被一个或多个(诸如1至3个)相同或不同的取代基取代。 The term "alkoxy" means a group in which an oxygen atom is inserted at any reasonable position of an alkyl group (as defined above), for example, C 1-8 alkoxy, C 1-6 alkoxy, C 1-4 Alkoxy or C 1-3 alkoxy. Representative examples of C 1-6 alkoxy include, but are not limited to, methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, isobutoxy, tert-butoxy, pentoxy Group, hexyloxy group, -CH 2 -OCH 3 and so on. The alkoxy group may be optionally substituted with one or more (such as 1 to 3) substituents which are the same or different.
术语“亚烷氧基”指二价的烷氧基,例如-OCH 2-、-OCH(CH 3)CH 2-、-OCH 2CH 2O-、-CH 2CH 2O-等,所 述亚烷氧基可以任选地被一个或多个(诸如1至3个)相同或不同的取代基取代。 The term "alkyleneoxy" refers to a divalent alkoxy group, such as -OCH 2 -, -OCH(CH 3 )CH 2 -, -OCH 2 CH 2 O-, -CH 2 CH 2 O-, etc., The alkyleneoxy group may be optionally substituted with one or more (such as 1 to 3) substituents which are the same or different.
术语“并环”或“稠环”指由两个或两个以上环状结构彼此共用两个相邻的原子所形成的环系。The term "condensed ring" or "fused ring" refers to a ring system formed by two or more ring structures sharing two adjacent atoms with each other.
术语“螺环”指由两个或两个以上环状结构彼此共用一个环原子所形成的环系。The term "spirocyclic ring" refers to a ring system formed by two or more cyclic structures sharing one ring atom with each other.
术语“桥环”指由两个或两个以上环状结构彼此共用两个不直接相连的原子所形成的环系。The term "bridged ring" refers to a ring system formed by two or more ring structures sharing two atoms that are not directly connected to each other.
术语“环烷基”指饱和或不饱和的非芳族单环或多环(诸如双环)烃环基,包括但不限于单环烷基(诸如环丙基、环丁基、环戊基、环己基、环庚基、环辛基、环壬基)和双环烷基,包括螺环、并环(稠环)或桥环系统(即,螺环烷基、并环(稠环)烷基和桥环烷基,诸如双环[1.1.1]戊基、双环[2.2.1]庚基等)。本发明中,环烷基可以任选地被一个或多个(诸如1至3个)相同或不同的取代基取代。环烷基上的碳原子任选地被氧代(oxo)基团取代(即形成C=O)。The term "cycloalkyl" refers to saturated or unsaturated non-aromatic monocyclic or polycyclic (such as bicyclic) hydrocarbon ring groups, including but not limited to monocyclic alkyl groups (such as cyclopropyl, cyclobutyl, cyclopentyl, Cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl) and bicyclic alkyl groups, including spirocyclic, fused ring (fused ring) or bridged ring systems (ie, spirocyclic alkyl, fused ring (fused ring) alkyl And bridged cycloalkyl groups, such as bicyclo[1.1.1]pentyl, bicyclo[2.2.1]heptyl, etc.). In the present invention, the cycloalkyl group may be optionally substituted by one or more (such as 1 to 3) substituents which are the same or different. The carbon atom on the cycloalkyl group is optionally substituted with an oxo group (ie forming C=O).
术语“3-7元环烷基”指具有3至7个成环碳原子的环烷基,其可以是单环烷基,例如环丙基、环丁基、环戊基、环己基或环庚基,也可以是双环烷基,例如C 5-7螺环烷基、C 5-7桥环烷基或C 4-7稠环烷基。 The term "3-7 membered cycloalkyl" refers to a cycloalkyl group having 3 to 7 ring-forming carbon atoms, which may be a monocyclic alkyl group, such as cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl or cyclo The heptyl group may also be a bicyclic alkyl group, such as a C 5-7 spirocycloalkyl group, a C 5-7 bridged cycloalkyl group or a C 4-7 condensed cycloalkyl group.
术语“C 3-8环烷基”指具有3至8个成环碳原子的环烷基,例如C 3-6环烷基,其可以是单环烷基,例如环丙基、环丁基、环戊基、环己基、环庚基或环辛基,也可以是双环烷基,例如C 3-8螺环烷基、C 3-8桥环烷基、C 3-8稠环烷基、C 3-6螺环烷基、C 3-6桥环烷基或C 3-6稠环烷基。 The term "C 3-8 cycloalkyl" refers to a cycloalkyl group having 3 to 8 ring-forming carbon atoms, such as a C 3-6 cycloalkyl group, which may be a monocyclic alkyl group, such as cyclopropyl, cyclobutyl , Cyclopentyl, cyclohexyl, cycloheptyl or cyclooctyl, it can also be a bicyclic alkyl group, such as C 3-8 spirocycloalkyl, C 3-8 bridged cycloalkyl, C 3-8 condensed cycloalkyl , C 3-6 spirocycloalkyl, C 3-6 bridged cycloalkyl or C 3-6 condensed cycloalkyl.
术语“亚环烷基”指如本文定义的环烷基,其具有自母体环烷基的相同碳原子或两个不同碳原子上去除两个氢原子而得到的两个单价基团中心。典型的亚环烷基包括但不限于亚环丙基、亚环丁基、亚环戊基、亚环己基、亚环庚基、亚环辛基、亚环壬基、亚环己烯基等。The term "cycloalkylene" refers to a cycloalkyl group as defined herein, which has two monovalent group centers obtained by removing two hydrogen atoms from the same carbon atom or two different carbon atoms of the parent cycloalkyl group. Typical cycloalkylene groups include, but are not limited to, cyclopropylene, cyclobutylene, cyclopentylene, cyclohexylene, cycloheptylene, cyclooctylene, cyclononylidene, cyclohexenylene, etc. .
术语“芳基”指具有共轭π电子系统的全碳单环或稠合多环芳族基团。如本文中所使用,术语“C 6-12芳基”意指含有6至12个碳原子的芳基,例如C 6-10芳基,并且具体实例为苯基或萘基。芳基任选地被一个或多个(诸如1至3个)相同或不同的取代基(例如卤素、OH、CN、NO 2、C 1-C 6烷基等)取代。 The term "aryl" refers to an all-carbon monocyclic or fused polycyclic aromatic group having a conjugated π-electron system. As used herein, the term "C 6-12 aryl group" means an aryl group containing 6 to 12 carbon atoms, such as a C 6-10 aryl group, and specific examples are phenyl or naphthyl. The aryl group is optionally substituted with one or more (such as 1 to 3) identical or different substituents (for example, halogen, OH, CN, NO 2 , C 1 -C 6 alkyl, etc.).
术语“亚芳基”指如本文定义的芳基,其具有自母体芳基的相同碳原子或两个不同碳原子上去除两个氢原子而得到的两个单价基团中心。典型的亚芳基包括但不限于亚苯基和亚萘基。The term "arylene" refers to an aryl group as defined herein, which has two monovalent group centers obtained by removing two hydrogen atoms from the same carbon atom or two different carbon atoms of the parent aryl group. Typical arylene groups include, but are not limited to, phenylene and naphthylene.
术语“芳基并环烷基”是指芳基与环烷基(例如单环烷基)彼此共用两个相邻的原子形成的并环基团,其中与其他基团的连接点可以在芳基上或在环烷基上。术语“9-12元芳基并环烷基”是指共计包含9-12个环原子的芳基并环烷基,例如苯基并环戊基、苯基并环己基,例如 The term "aryl and cycloalkyl" refers to a fused ring group formed by aryl and cycloalkyl (such as monocyclic alkyl) sharing two adjacent atoms with each other, wherein the point of attachment to other groups can be at the aromatic On the base or on the cycloalkyl group. The term "9-12 membered aryl and cycloalkyl" refers to an aryl and cycloalkyl group containing 9-12 ring atoms in total, such as phenyl and cyclopentyl, phenyl and cyclohexyl, such as
术语“杂环基”指具有2个或2个以上(例如3、4、5、6、7、8、9、10、11、12、13或14个)碳原子,以及一个或多个(例如1个、2个、3个或4个)杂原子的单环或多环(例如并环、螺环或桥环)基团,所述杂原子包括但不限于氧原子、氮原子或硫原子,所述杂环基上的碳原子和杂原子任选地被氧代基团取代(例如形成C=O、S(=O)或S(=O) 2)。 The term "heterocyclyl" refers to having 2 or more (for example, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, or 14) carbon atoms, and one or more ( For example, 1, 2, 3, or 4) heteroatom monocyclic or polycyclic (for example, fused ring, spiro ring or bridged ring) group, the heteroatom includes but not limited to oxygen atom, nitrogen atom or sulfur Atoms, carbon atoms and heteroatoms on the heterocyclic group are optionally substituted with oxo groups (for example, forming C=O, S(=O) or S(=O) 2 ).
术语“亚杂环基”指如本文定义的杂环基,其具有自母体杂环基的相同碳原子或两个不同碳原子、一个碳原子和一个杂原子、或者两个杂原子上去除两个氢原子而得到的两个单价基团中心。The term "heterocyclylene" refers to a heterocyclic group as defined herein, which has the same carbon atom or two different carbon atoms, one carbon atom and one heteroatom, or two heteroatoms removed from the parent heterocyclic group. Two monovalent group centers derived from a hydrogen atom.
术语“3-14元杂环基”意指含有3-14个环原子的杂环基,包括但不限于4-10元杂环基、4-7元杂环基、5-6元杂环基、4-7元含氮杂环基、4-7元含氧杂环基、4-7元含硫杂环基、5-6元含氮杂环基、5-6元含氧杂环基、5-6元含硫杂环基等,所述“含氮杂环基”、“含氧杂环基”和“含硫杂环基”各自任选地还含有一个或多个选自氧、氮和硫的其他杂原子。3-14元杂环基的实例包括但不限于环氧乙烷基、氮丙啶基、氮杂环丁基、氧杂环丁基、四氢呋喃基、吡咯烷基、吡咯烷酮基、咪唑烷基、吡唑烷基、四氢吡喃基、哌啶基、吗啉基、二噻烷基(dithianyl)、硫吗啉基、哌嗪基、三噻烷基(trithianyl)、 等。 The term "3-14 membered heterocyclic group" means a heterocyclic group containing 3-14 ring atoms, including but not limited to 4-10 membered heterocyclic group, 4-7 membered heterocyclic group, 5-6 membered heterocyclic group Group, 4-7 membered nitrogen-containing heterocyclic group, 4-7 membered oxygen-containing heterocyclic group, 4-7 membered sulfur-containing heterocyclic group, 5-6 membered nitrogen-containing heterocyclic group, 5-6 membered oxygen-containing heterocyclic group Group, 5-6 membered sulfur-containing heterocyclic group, etc., the "nitrogen-containing heterocyclic group", "oxygen-containing heterocyclic group" and "sulfur-containing heterocyclic group" each optionally further contain one or more Other heteroatoms of oxygen, nitrogen and sulfur. Examples of 3-14 membered heterocyclic groups include, but are not limited to, oxiranyl, aziridinyl, azetidinyl, oxetanyl, tetrahydrofuryl, pyrrolidinyl, pyrrolidonyl, imidazolidinyl, Pyrazolyl, tetrahydropyranyl, piperidinyl, morpholinyl, dithianyl, thiomorpholinyl, piperazinyl, trithianyl, Wait.
本发明中,杂环基还包括并环结构,所述并环结构与其他基团的连接点可以在并环结构中的任一环上。因此,本发明的杂环基还包括(但不限于)杂环基并杂环基、杂环基并环烷基、单杂环基并单杂环基、单杂环基并单环烷基,例如3-7元(单)杂环基并3-7元(单)杂环基、3-7元(单)杂环基并(单)环烷基、3-7元(单)杂环基并C 4-6(单)环烷基等,其实例包括但不限于吡咯烷基并环丙基、环戊基并氮杂环丙基、吡咯烷基并环丁基、吡咯烷基并吡咯烷基、吡咯烷基并哌啶基、吡咯烷基并哌嗪基、哌啶基并吗啉基、 等。 In the present invention, the heterocyclic group also includes a fused ring structure, and the point of connection between the fused ring structure and other groups can be on any ring in the fused ring structure. Therefore, the heterocyclic group of the present invention also includes (but is not limited to) heterocyclic group and heterocyclic group, heterocyclic group and cycloalkyl group, single heterocyclic group and single heterocyclic group, single heterocyclic group and monocyclic alkyl group. , Such as 3-7 membered (mono) heterocyclyl and 3-7 membered (mono) heterocyclyl, 3-7 membered (mono) heterocyclyl and (mono) cycloalkyl, 3-7 membered (mono) hetero Cyclic and C 4-6 (mono)cycloalkyl, etc., examples of which include, but are not limited to, pyrrolidinocyclopropyl, cyclopentylazacyclopropyl, pyrrolidinocyclobutyl, pyrrolidinyl Pyrrolidinyl, pyrrolidinopiperidinyl, pyrrolidinopiperazinyl, piperidinomorpholinyl, Wait.
本发明中,杂环基还包括桥杂环基和螺杂环基。In the present invention, heterocyclic groups also include bridged heterocyclic groups and spiro heterocyclic groups.
术语“桥杂环基”是指两个饱和环共用两个不直接相连的环原子形成的含有一个或多个(例如1个、2 个、3个或4个)杂原子(例如氧原子、氮原子或硫原子)的环状结构,包括但不限于7-10元桥杂环基、8-10元桥杂环基、7-10元含氮桥杂环基、7-10元含氧桥杂环基、7-10元含硫桥杂环基等,例如 等。所述“含氮桥杂环基”、“含氧桥杂环基”和“含硫桥杂环基”各自任选地还含有一个或多个选自氧、氮和硫的其他杂原子。 The term "bridged heterocyclyl" refers to two saturated rings that share two ring atoms that are not directly connected and contain one or more (e.g., 1, 2, 3, or 4) heteroatoms (e.g., oxygen atoms, Nitrogen atom or sulfur atom), including but not limited to 7-10 membered heterocyclic group, 8-10 membered heterocyclic group, 7-10 membered nitrogen-containing bridged heterocyclic group, 7-10 membered oxygen-containing Bridged heterocyclic group, 7-10 membered sulfur-containing bridged heterocyclic group, etc., for example Wait. The "nitrogen-containing bridged heterocyclic group", "oxygen-containing bridged heterocyclic group" and "sulfur-containing bridged heterocyclic group" each optionally further contain one or more other heteroatoms selected from oxygen, nitrogen and sulfur.
术语“螺杂环基”是指由两个或两个以上饱和环共用一个环原子形成的含有一个或多个(例如1个、2个、3个或4个)杂原子(例如氧原子、氮原子、硫原子)的环状结构,包括但不限于5-10元螺杂环基、6-10元螺杂环基、6-10元含氮螺杂环基、6-10元含氧螺杂环基、6-10元含硫螺杂环基等,例如 等。所述“含氮螺杂环基”、“含氧螺杂环基”和“含硫螺杂环基”各自任选地还含有一个或多个选自氧、氮和硫的其他杂原子。术语“6-10元含氮螺杂环基”是指含有共计6-10个环原子并且其中至少一个环原子为氮原子的螺杂环基。 The term "spiroheterocyclic group" refers to two or more saturated rings that share one ring atom and contain one or more (e.g., 1, 2, 3, or 4) heteroatoms (e.g., oxygen atoms, Nitrogen atom, sulfur atom) ring structure, including but not limited to 5-10 membered spiro heterocyclic group, 6-10 membered spiro heterocyclic group, 6-10 membered nitrogen-containing spiro heterocyclic group, 6-10 membered oxygen-containing Spiro heterocyclic group, 6-10 membered sulfur-containing spiro heterocyclic group, etc., for example Wait. The "nitrogen-containing spiro heterocyclic group", "oxygen-containing spiro heterocyclic group" and "sulfur-containing spiro heterocyclic group" each optionally further contain one or more other heteroatoms selected from oxygen, nitrogen and sulfur. The term "6-10 membered nitrogen-containing spiroheterocyclic group" refers to a spiroheterocyclic group containing 6-10 ring atoms in total and at least one of which is a nitrogen atom.
术语“芳基并杂环基”指芳基与杂环基彼此共用两个相邻的碳原子所形成的环状基团(其中芳基和杂环基如上文所定义),其与其他基团的连接点在可以在芳基或杂环基上。例如,如本文中所使用,术语“9-12元芳基并杂环基”意指含有共计9-12个环原子的芳基并杂环基的基团,包括但不限于9-10元苯并杂环基,例如苯并5-8元杂环基,例如苯并5-6元杂环基,例如苯并5-6元单杂环基、苯并5-6元含氮单杂环基、苯并5-6元含氧单杂环基、苯并5-6元含硫杂环基等。所述“含氮杂环基”、“含氧杂环基”和“含硫杂环基”各自任选地还含有一个或多个选自氧、氮和硫的其他杂原子。所述杂环基上的碳原子和杂原子任选地被氧代基团取代(例如形成C=O、S(=O)或S(=O) 2)。芳基并杂环基的实例包括但不限于:吲唑基、 The term "aryl and heterocyclic group" refers to a cyclic group formed by an aryl group and a heterocyclic group sharing two adjacent carbon atoms with each other (wherein the aryl group and the heterocyclic group are as defined above), which is combined with other groups. The point of attachment of the group may be on an aryl group or a heterocyclic group. For example, as used herein, the term "9-12 membered aryl and heterocyclyl" means an aryl and heterocyclyl group containing a total of 9-12 ring atoms, including but not limited to 9-10 membered Benzoheterocyclic group, such as benzo 5-8 membered heterocyclic group, such as benzo 5-6 membered heterocyclic group, such as benzo 5-6 membered monoheterocyclic group, benzo 5-6 membered nitrogen-containing monohetero Cyclic group, benzo 5-6 membered oxygen-containing monoheterocyclic group, benzo 5-6 membered sulfur-containing heterocyclic group, etc. Each of the "nitrogen-containing heterocyclic group", "oxygen-containing heterocyclic group" and "sulfur-containing heterocyclic group" optionally further contains one or more other heteroatoms selected from oxygen, nitrogen and sulfur. The carbon atoms and heteroatoms on the heterocyclic group are optionally substituted with oxo groups (for example, forming C=O, S(=O) or S(=O) 2 ). Examples of aryl and heterocyclic groups include, but are not limited to: indazolyl,
术语“杂芳基”指含有一个或多个相同或不同杂原子的单环或多环芳族基团,包括单环杂芳基和含有至少一个杂芳环(至少含有一个杂原子的芳族环系)的双环或多环环系,其可以具有5、6、7、8、9、10、11、12、13或14个环原子,例如5、6、7、8、9或10个环原子。所述杂原子可以是氧、氮或硫。所述杂芳基上的碳原子和杂原子任选地被氧代基团取代(例如形成C=O、S(=O)或S(=O) 2)。 The term "heteroaryl" refers to a monocyclic or polycyclic aromatic group containing one or more identical or different heteroatoms, including monocyclic heteroaryl groups and aromatic groups containing at least one heteroaromatic ring (an aromatic group containing at least one heteroatom Ring system), which may have 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14 ring atoms, such as 5, 6, 7, 8, 9 or 10 Ring atom. The heteroatom may be oxygen, nitrogen or sulfur. The carbon atoms and heteroatoms on the heteroaryl group are optionally substituted with oxo groups (for example, forming C=O, S(=O) or S(=O) 2 ).
术语“亚杂芳基”是指如上所述的杂芳基,其具有自母体杂芳基的相同碳原子或两个不同碳原子上去除两个氢原子或是自碳原子上去除一个氢原子且自氮原子上去除一个氢原子而得到的两个单价基团中心。The term "heteroarylene" refers to a heteroaryl group as described above, which has two hydrogen atoms removed from the same carbon atom or two different carbon atoms of the parent heteroaryl group or one hydrogen atom removed from the carbon atom And two monovalent group centers are obtained by removing one hydrogen atom from the nitrogen atom.
术语“5-10元杂芳基”意指含有5至10个环原子的杂芳基,包括5-6元杂芳基、5-6元单杂芳基、5-10元含氮杂芳基、5-10元含氧杂芳基、5-10元含硫杂芳基、5-6元含氮杂芳基、5-6元含氧杂芳基、5-6元含硫杂芳基、5-6元含氮单杂芳基、5-6元含氧单杂芳基、5-6元含硫单杂芳基。所述“含氮杂芳基”、“含氧杂芳基”、“含硫杂芳基”、“含氮单杂芳基”、“含氧单杂芳基”和“含硫单杂芳基”各自任选地还含有一个或多个选自氧、氮、硫的其他杂原子。5-10元杂芳基的实例包括但不限于噻吩基、呋喃基、吡咯基、噁唑基、噻唑基、咪唑基、吡唑基、异噁唑基、异噻唑基、三唑基、四唑基、噁二唑基、噻二唑基、吡啶基、哒嗪基、嘧啶基、吡嗪基、三嗪基等,以及包含这些基团的5-10元并环基团。The term "5-10 membered heteroaryl" means a heteroaryl group containing 5 to 10 ring atoms, including 5-6 membered heteroaryl, 5-6 membered monoheteroaryl, 5-10 membered nitrogen-containing heteroaryl Group, 5-10 member oxygen-containing heteroaryl group, 5-10 member sulfur-containing heteroaryl group, 5-6 member nitrogen-containing heteroaryl group, 5-6 member oxygen-containing heteroaryl group, 5-6 member sulfur-containing heteroaryl group Group, 5-6 membered nitrogen-containing monoheteroaryl group, 5-6 membered oxygen-containing monoheteroaryl group, 5-6 membered sulfur-containing monoheteroaryl group. The "nitrogen-containing heteroaryl group", "oxygen-containing heteroaryl group", "sulfur-containing heteroaryl group", "nitrogen-containing monoheteroaryl group", "oxygen-containing monoheteroaryl group" and "sulfur-containing monoheteroaryl group" Each of the "radicals" optionally further contains one or more other heteroatoms selected from oxygen, nitrogen, and sulfur. Examples of 5-10 membered heteroaryl groups include, but are not limited to, thienyl, furyl, pyrrolyl, oxazolyl, thiazolyl, imidazolyl, pyrazolyl, isoxazolyl, isothiazolyl, triazolyl, tetrazolyl, Azolyl, oxadiazolyl, thiadiazolyl, pyridyl, pyridazinyl, pyrimidinyl, pyrazinyl, triazinyl, etc., and 5-10 membered cyclic groups containing these groups.
本发明中,术语“杂芳基”涵盖并环结构,即杂芳基(例如单杂芳基)可以与芳基(例如单环芳基,例如苯基)、杂环基(例如单杂环基)、环烷基(例如单环烷基)或另一杂芳基(例如另一单杂芳基)彼此共用两个相邻的原子形成的并环结构,其与其他基团的连接点可以在任一杂芳环上或其它环上,包括但不限于(单)杂芳基并(单)杂芳基、(单)杂芳基并(单环)芳基、(单)杂芳基并(单)杂环基或(单)杂芳基并(单)环烷基,例如5-6元(单)杂芳基并5-6元(单)杂芳基、5-6元(单)杂芳基并苯基、5-6元(单)杂芳基并5-6元(单)杂环基或5-6元(单)杂芳基并C 4-6(单)环烷基(例如5-6元杂芳基并环丁基、5-6元杂芳基并环戊基或5-6元杂芳基并环己基),其实例包括但不限于吲哚基、异吲哚基、吲唑基、苯并咪唑、喹啉基、异喹啉基、 等。 In the present invention, the term "heteroaryl" encompasses a bicyclic structure, that is, heteroaryl (e.g., monoheteroaryl) can be combined with aryl (e.g., monocyclic aryl, e.g., phenyl), heterocyclic group (e.g., monoheterocyclic group). Group), cycloalkyl (e.g. monocycloalkyl) or another heteroaryl group (e.g. another monoheteroaryl) sharing two adjacent atoms with each other to form a combined ring structure, which is the point of attachment to other groups It can be on any heteroaromatic ring or other ring, including but not limited to (mono)heteroaryl and (mono)heteroaryl, (mono)heteroaryl and (monocyclic)aryl, (mono)heteroaryl And (mono)heterocyclyl or (mono)heteroaryl and (mono)cycloalkyl, for example 5-6 membered (mono)heteroaryl and 5-6 membered (mono)heteroaryl, 5-6 membered ( Mono) heteroaryl phenyl, 5-6 membered (mono) heteroaryl and 5-6 membered (mono) heterocyclic group or 5-6 membered (mono) heteroaryl and C 4-6 (mono) ring Alkyl groups (e.g. 5-6 membered heteroaryl and cyclobutyl, 5-6 membered heteroaryl and cyclopentyl or 5-6 membered heteroaryl and cyclohexyl), examples of which include but are not limited to indolyl, Isoindolyl, indazolyl, benzimidazole, quinolinyl, isoquinolinyl, Wait.
术语“杂芳基”所涵盖的含芳基的并环结构也称为“芳基并杂芳基”,是指芳基(例如单环芳基,例如苯基)与杂芳基(例如单杂芳基,例如5-6元单杂芳基)形成的并环基团,其与其他基团的连接点可以在芳环上,也可以在杂芳环上。所述“芳基并杂芳基”包括但不限于单环芳基并单杂芳基。术语“9-12元芳基并杂芳基”是指含有共计9-12个环原子的芳基并杂芳基,例如苯并5-6元含氮单杂芳基。The term "heteroaryl" encompasses aryl-containing fused ring structures, which are also referred to as "aryl and heteroaryl", which refers to aryl (e.g., monocyclic aryl, e.g., phenyl) and heteroaryl (e.g., monocyclic aryl) Heteroaryl groups, such as 5-6 membered mono-heteroaryl groups, can be connected to other groups on the aromatic ring or on the heteroaromatic ring. The "aryl and heteroaryl" includes, but is not limited to, monocyclic aryl and monoheteroaryl. The term "9-12 membered aryl and heteroaryl group" refers to an aryl and heteroaryl group containing a total of 9 to 12 ring atoms, such as a benzo 5-6 membered nitrogen-containing monoheteroaryl group.
术语“杂芳基”所涵盖的含环烷基的并环结构也称为“杂芳基并环烷基”,是指杂芳基(例如单杂芳基,例如5-6元单杂芳基)与环烷基(例如C 4-6环烷基)形成的并环基团,其与其他基团的连接点可以在杂芳环上,也可以在环烷基上。所述“杂芳基并环烷基”包括但不限于单杂芳基并单环烷基。术语“9-10元杂芳基并环烷基”是指含有共计9-10个环原子的杂芳基并环烷基,例如4-6元含氮单杂芳基并C 4-6单环烷基。 The term "heteroaryl" includes a cycloalkyl-containing bicyclic structure, which is also referred to as "heteroaryl and cycloalkyl", which refers to a heteroaryl group (such as a monoheteroaryl group, such as a 5-6 membered monoheteroaryl group). Group) and a cycloalkyl group (for example, a C 4-6 cycloalkyl group). The point of connection with other groups can be on the heteroaromatic ring or on the cycloalkyl group. The "heteroaryl and cycloalkyl" includes, but is not limited to, monoheteroaryl and monocycloalkyl. The term "9-10 membered heteroaryl and cycloalkyl" refers to a heteroaryl and cycloalkyl group containing a total of 9-10 ring atoms, such as 4-6 membered nitrogen-containing monoheteroaryl and C 4-6 mono Cycloalkyl.
术语“取代”指所指定的原子上的一个或多个(例如1个、2个、3个或4个)氢被从所指出的基团的选择代替,条件是未超过所指定的原子在当前情况下的正常原子价并且所述取代形成稳定的化合物。取代基和/或变量的组合仅仅当这种组合形成稳定的化合物时才是允许的。The term "substitution" means that one or more (for example, 1, 2, 3, or 4) hydrogens on the designated atom are replaced by a selection from the indicated group, provided that no more than the designated atom is The normal valence in the current situation and the substitution forms a stable compound. Combinations of substituents and/or variables are only permissible when such combinations form stable compounds.
如果取代基被描述为“任选地被……取代”,则取代基可(1)未被取代或(2)被取代。如果取代基的碳被描述为任选地被取代基列表中的一个或多个取代,则碳上的一个或多个氢(至存在的任何氢的程度)可各自被独立地选择的任选的取代基替代。如果取代基的氮被描述为任选地被取代基列表中的一个或多个取代,则氮上的一个或多个氢(至存在的任何氢的程度)可各自被独立地选择的任选的取代基替代。If a substituent is described as "optionally substituted by", the substituent can be (1) unsubstituted or (2) substituted. If the carbon of a substituent is described as being optionally substituted by one or more of the list of substituents, then one or more hydrogens on the carbon (to the extent of any hydrogens present) may each be independently selected optionally Substituent replacement. If the nitrogen of a substituent is described as being optionally substituted by one or more of the list of substituents, then one or more hydrogens on the nitrogen (to the extent of any hydrogens present) may each be independently selected optionally Substituent replacement.
如果取代基被描述为“独立地选自”一组,则各取代基独立于另一者被选择。因此,各取代基可与另一(其他)取代基相同或不同。If substituents are described as being "independently selected from" a group, then each substituent is selected independently of the other. Therefore, each substituent may be the same or different from another (other) substituent.
如本文中所使用,术语“一个或多个”意指在合理条件下的1个或超过1个,例如2个、3个、4个、5个、6个、7个、8个、9个或10个。As used herein, the term "one or more" means one or more than one under reasonable conditions, such as two, three, four, five, six, seven, eight, nine Or 10.
除非指明,否则如本文中所使用,取代基的连接点可来自取代基的任意适宜位置。Unless otherwise specified, as used herein, the point of attachment of a substituent can be from any suitable position of the substituent.
本发明还包括本发明化合物的所有药学上可接受的同位素化合物,其与本发明的化合物相同,除了一个或多个原子被具有相同原子序数但原子质量或质量数不同于在自然界中占优势的原子质量或质量数的原子替代。适合包含入本发明的化合物中的同位素的实例包括(但不限于)氢的同位素(例如 2H、 3H);碳的同位素(例如 11C、 13C及 14C);氯的同位素(例如 36Cl);氟的同位素(例如 18F);碘的同位素(例如 123I及 125I);氮的同位素(例如 13N及 15N);氧的同位素(例如 15O、 17O及 18O);磷的同位素(例如 32P);及硫的同位素(例如 35S)。术语“稳定的同位素衍生物”是指本发明的化合物中的一个或多个原子被具有相同原子序数但原子质量或质量数不同于在自然界中占优势的原子质量或质量数的原子替代所形成的稳定的化合物。 The present invention also includes all pharmaceutically acceptable isotopic compounds of the compounds of the present invention, which are the same as the compounds of the present invention, except that one or more atoms have the same atomic number but the atomic mass or mass number is different from the one that is predominant in nature. Atomic substitution of atomic mass or mass number. Examples of isotopes suitable for inclusion in the compounds of the present invention include (but are not limited to) isotopes of hydrogen (e.g. 2 H, 3 H); isotopes of carbon (e.g. 11 C, 13 C, and 14 C); isotopes of chlorine (e.g. 36 Cl); fluorine isotopes (such as 18 F); iodine isotopes (such as 123 I and 125 I); nitrogen isotopes (such as 13 N and 15 N); oxygen isotopes (such as 15 O, 17 O and 18 O ); Phosphorus isotopes (for example 32 P); and Sulfur isotopes (for example 35 S). The term "stable isotope derivative" means that one or more atoms in the compound of the present invention are replaced by atoms having the same atomic number but whose atomic mass or mass number is different from the predominant atomic mass or mass number in nature. The stable compound.
术语“立体异构体”表示化合物由于含有至少一个不对称中心而形成的异构体。在具有一个或多个(例如1个、2个、3个或4个)不对称中心的化合物中,其可产生外消旋混合物、单一对映异构体、非对映异构体混合物和单独的非对映异构体。特定个别分子也可以几何异构体(顺式/反式)存在。本发明的化合物可以两种或更多种处于快速平衡的不同结构形式的混合物(通常称作互变异构体)存在。互变异构体的代表性实例包括酮-烯醇互变异构体、苯酚-酮互变异构体、亚硝基-肟互变异构体、亚胺-烯胺互变异构体等。例如,亚硝基-肟在溶液中可以下列互变异构形式平衡存在: 而吡唑环可以下 列互变异构形式平衡存在: The term "stereoisomer" refers to an isomer formed by a compound containing at least one asymmetric center. In compounds with one or more (for example, 1, 2, 3, or 4) asymmetric centers, it can produce racemic mixtures, single enantiomers, diastereomeric mixtures, and Individual diastereomers. Certain individual molecules can also exist as geometric isomers (cis/trans). The compounds of the present invention may exist in a mixture of two or more different structural forms (commonly referred to as tautomers) in rapid equilibrium. Representative examples of tautomers include keto-enol tautomers, phenol-ketone tautomers, nitroso-oxime tautomers, imine-enamine tautomers Wait. For example, nitroso-oximes can exist in equilibrium in the following tautomeric forms in solution: The pyrazole ring can exist in equilibrium in the following tautomeric forms:
要理解,本申请的范围涵盖所有这样的以任意比例(例如60%、65%、70%、75%、80%、85%、90%、95%、96%、97%、98%或99%)的异构体或其混合物。It is to be understood that the scope of the present application covers all such proportions (for example, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98% or 99%). %) isomers or mixtures thereof.
除非另外指明,否则本发明的化合物意欲可以立体异构体(其包括顺式及反式异构体)、光学异构体(例如R及S对映异构体)、非对映异构体、几何异构体、旋转异构体、构象异构体、阻转异构体或其混合物的形式存在。本发明的化合物可表现一种以上类型的异构现象,且由其混合物(例如外消旋混合物及非对映异构体对)组成。Unless otherwise specified, the compounds of the present invention are intended to be stereoisomers (including cis and trans isomers), optical isomers (such as R and S enantiomers), and diastereomers. , Geometric isomers, rotamers, conformational isomers, atropisomers or mixtures thereof. The compounds of the present invention can exhibit more than one type of isomerism, and are composed of mixtures thereof (e.g., racemic mixtures and diastereomeric pairs).
本发明涵盖本发明的化合物的所有可能的结晶形式或多晶型物,其可为单一多晶型物或多于一种多晶型物的任意比例的混合物。还应当理解,本发明的某些化合物可以游离形式存在用于治疗,或适当时,以其药学上可接受的衍生物形式存在。在本发明中,药学上可接受的衍生物包括但不限于:药学上可接受的盐、溶剂合物、代谢物或前药,在将它们向需要其的患者给药后,能够直接或间接提供本发明的化合物或其代谢物或残余物。因此,当在本文中提及“本发明的化合物”时,也意在涵盖化合物的上述各种衍生物形式。The present invention covers all possible crystalline forms or polymorphs of the compounds of the present invention, which can be a single polymorph or a mixture of more than one polymorph in any ratio. It should also be understood that certain compounds of the present invention may exist in free form for treatment, or, when appropriate, in the form of their pharmaceutically acceptable derivatives. In the present invention, pharmaceutically acceptable derivatives include, but are not limited to: pharmaceutically acceptable salts, solvates, metabolites or prodrugs, which can be directly or indirectly administered to patients in need thereof. The compound of the present invention or its metabolite or residue is provided. Therefore, when the "compound of the present invention" is referred to herein, it is also intended to encompass the above-mentioned various derivative forms of the compound.
本发明的化合物的药学上可接受的盐包括其酸加成盐及碱加成盐。例如六氟磷酸盐、葡甲胺盐等。适合的盐的综述参见Stahl及Wermuth的“Handbook of Pharmaceutical Salts:Properties,Selection,and Use”(Wiley-VCH,2002)。The pharmaceutically acceptable salts of the compounds of the present invention include acid addition salts and base addition salts thereof. For example, hexafluorophosphate, meglumine salt and the like. For a review of suitable salts, see "Handbook of Pharmaceutical Salts: Properties, Selection, and Use" by Stahl and Wermuth (Wiley-VCH, 2002).
本发明中“药学上可接受的载体”是指与治疗剂一同给药的稀释剂、辅剂、赋形剂或媒介物,并且其在合理的医学判断的范围内适于接触人类和/或其它动物的组织而没有过度的毒性、刺激、过敏反应或与合理的益处/风险比相应的其它问题或并发症。In the present invention, "pharmaceutically acceptable carrier" refers to a diluent, adjuvant, excipient or vehicle administered with the therapeutic agent, and it is suitable for contact with humans and/or within the scope of reasonable medical judgment Tissues of other animals without excessive toxicity, irritation, allergic reactions, or other problems or complications corresponding to a reasonable benefit/risk ratio.
在本发明的药物组合物中可使用的药学上可接受的载体包括但不限于无菌液体,例如水和油,包括那些石油、动物、植物或合成来源的油,例如花生油、大豆油、矿物油、芝麻油等。当所述药物组合物通过静脉内给药时,水是示例性载体。还可以使用生理盐水和葡萄糖及甘油水溶液作为液体载体,特别是用于注射液。适合的药物赋形剂包括淀粉、葡萄糖、乳糖、蔗糖、明胶、麦芽糖、白垩、硅胶、硬脂酸钠、单硬脂酸甘油酯、滑石、氯化钠、脱脂奶粉、甘油、丙二醇、水、乙醇等。所述组合物还可以视需要包含少量的湿润剂、乳化剂或pH缓冲剂。口服制剂可以包含标准载体,如药物级的甘露醇、乳糖、淀粉、硬脂酸镁、糖精钠、纤维素、碳酸镁等。适合的药学上可接受的载体的实例如在Remington’s Pharmaceutical Sciences(1990)中所述。The pharmaceutically acceptable carriers that can be used in the pharmaceutical composition of the present invention include, but are not limited to, sterile liquids, such as water and oils, including those of petroleum, animal, vegetable, or synthetic origin, such as peanut oil, soybean oil, and mineral oil. Oil, sesame oil, etc. When the pharmaceutical composition is administered intravenously, water is an exemplary carrier. It is also possible to use physiological saline and aqueous glucose and glycerol solutions as liquid carriers, especially for injections. Suitable pharmaceutical excipients include starch, glucose, lactose, sucrose, gelatin, maltose, chalk, silica gel, sodium stearate, glyceryl monostearate, talc, sodium chloride, skimmed milk powder, glycerin, propylene glycol, water, Ethanol, etc. The composition may also contain small amounts of wetting agents, emulsifiers or pH buffering agents as needed. Oral preparations may contain standard carriers such as pharmaceutical grades of mannitol, lactose, starch, magnesium stearate, sodium saccharin, cellulose, magnesium carbonate and the like. Examples of suitable pharmaceutically acceptable carriers are described in Remington's Pharmaceutical Sciences (1990).
本发明的药物组合物可以系统地作用和/或局部地作用。为此目的,它们可以适合的途径给药,例如通过注射、静脉内、动脉内、皮下、腹膜内、肌内或经皮给药;或通过口服、含服、经鼻、透粘膜、局部、以眼用制剂的形式或通过吸入给药。The pharmaceutical composition of the present invention can act systemically and/or locally. For this purpose, they can be administered by suitable routes, such as by injection, intravenous, intraarterial, subcutaneous, intraperitoneal, intramuscular or transdermal administration; or by oral, buccal, transnasal, transmucosal, topical, It is administered in the form of ophthalmic preparations or by inhalation.
对于这些给药途径,可以适合的剂型给药本发明的药物组合物。For these administration routes, the pharmaceutical composition of the present invention can be administered in a suitable dosage form.
所述剂型包括但不限于片剂、胶囊剂、锭剂、硬糖剂、散剂、喷雾剂、乳膏剂、软膏剂、栓剂、凝胶剂、糊剂、洗剂、软膏剂、水性混悬剂、可注射溶液剂、酏剂、糖浆剂。The dosage form includes but not limited to tablet, capsule, lozenge, hard candy, powder, spray, cream, ointment, suppository, gel, paste, lotion, ointment, aqueous suspension , Injectable solutions, elixirs, syrups.
如本文中所使用的术语“有效剂量”指被给药后会在一定程度上缓解所治疗病症的一或多种症状的化合物的量。The term "effective dose" as used herein refers to the amount of a compound that will relieve one or more symptoms of the condition being treated to a certain extent after being administered.
可调整给药方案以提供最佳所需响应。例如,可给药单次推注,可随时间给药数个分剂量,或可如治疗情况的急需所表明而按比例减少或增加剂量。要注意,剂量值可随要减轻的病况的类型及严重性而变化,且可包括单次或多次剂量。要进一步理解,对于任何特定个体,具体的给药方案应根据个体需要及给药组合物或监督组合物的给药的人员的专业判断来随时间调整。The dosage regimen can be adjusted to provide the best desired response. For example, a single bolus can be administered, several divided doses can be administered over time, or the dose can be proportionally reduced or increased as indicated by the urgent need of the treatment situation. It should be noted that the dose value may vary with the type and severity of the condition to be alleviated, and may include single or multiple doses. It should be further understood that for any particular individual, the specific dosing regimen should be adjusted over time according to the individual's needs and the professional judgment of the person administering the composition or supervising the administration of the composition.
所给药的本发明的化合物的量会取决于所治疗的个体、病症或病况的严重性、给药的速率、化合物的处置及处方医师的判断。一般而言,有效剂量在每日每kg体重约0.0001至约50mg,例如约0.01至约10mg/kg/日(单次或分次给药)。对70kg的人而言,这会合计为约0.007mg/日至约3500mg/日,例如约0.7mg/日至约700mg/日。在一些情况下,不高于前述范围的下限的剂量水平可以是足够的,而在其它情况下,仍可在不引起任何有害副作用的情况下采用较大剂量,条件是首先将所述较大剂量分成数个较小剂量以在一整天中给药。The amount of the compound of the present invention administered will depend on the individual being treated, the severity of the disorder or condition, the rate of administration, the treatment of the compound, and the judgment of the prescribing physician. In general, the effective dose is about 0.0001 to about 50 mg per kg body weight per day, for example, about 0.01 to about 10 mg/kg/day (single or divided administration). For a 70 kg person, this would add up to about 0.007 mg/day to about 3500 mg/day, for example, about 0.7 mg/day to about 700 mg/day. In some cases, a dose level not higher than the lower limit of the aforementioned range may be sufficient, while in other cases, a larger dose can still be used without causing any harmful side effects, provided that the larger dose is first The dose is divided into several smaller doses to be administered throughout the day.
本发明的化合物在药物组合物中的含量或用量可以是约0.01mg至约1000mg。The content or amount of the compound of the present invention in the pharmaceutical composition may be about 0.01 mg to about 1000 mg.
除非另外说明,否则如本文中所使用,术语“治疗(treating)”意指逆转、减轻、抑制这样的术语所应用的病症或病况、或者这样的病症或病况的一或多种症状的进展。Unless otherwise specified, as used herein, the term "treating" means reversing, alleviating, or inhibiting the progress of one or more symptoms of the disorder or condition to which such term is applied.
术语“预防”意指能够避免这样的术语所应用的病症或病况的产生。The term "prevention" means being able to avoid the occurrence of the disease or condition to which such a term is applied.
如本文所使用的“个体”包括人或非人动物。示例性人个体包括患有疾病(例如本文所述的疾病)的人个体(称为患者)或正常个体。本发明中“非人动物”包括所有脊椎动物,例如非哺乳动物(例如鸟类、两栖动物、爬行动物)和哺乳动物,例如非人灵长类、家畜和/或驯化动物(例如绵羊、犬、猫、奶牛、猪等)。"Individual" as used herein includes human or non-human animals. Exemplary human individuals include human individuals (referred to as patients) or normal individuals suffering from diseases such as those described herein. In the present invention, "non-human animals" include all vertebrates, such as non-mammals (such as birds, amphibians, reptiles) and mammals, such as non-human primates, livestock and/or domesticated animals (such as sheep, dogs). , Cats, cows, pigs, etc.).
本发明的化合物可以溶剂合物(优选水合物)的形式存在,其中本发明的化合物包含作为所述化合物晶格的结构要素的极性溶剂,特别是例如水、甲醇或乙醇。极性溶剂特别是水的量可以化学计量比或非化学计量比存在。The compound of the present invention may exist in the form of a solvate (preferably a hydrate), wherein the compound of the present invention contains a polar solvent as a structural element of the compound crystal lattice, in particular, for example, water, methanol or ethanol. The amount of polar solvent, especially water, can be present in a stoichiometric or non-stoichiometric ratio.
在本发明的范围内还包括本发明的化合物的代谢物,即在给药本发明的化合物时体内形成的物质。这样的产物可由例如被给药的化合物的氧化、还原、水解、酰胺化、脱酰胺化、酯化、脱脂化、酶解等产生。因此,本发明包括本发明的化合物的代谢物,包括通过使本发明的化合物与哺乳动物接触足以产生其代谢产物的时间的方法得到的化合物。Also included within the scope of the present invention are metabolites of the compounds of the present invention, that is, substances formed in the body when the compounds of the present invention are administered. Such products can be produced by, for example, oxidation, reduction, hydrolysis, amidation, deamidation, esterification, delipidation, enzymatic hydrolysis, etc. of the administered compound. Therefore, the present invention includes metabolites of the compounds of the present invention, including compounds obtained by contacting the compound of the present invention with a mammal for a time sufficient to produce its metabolites.
本发明在其范围内进一步包括本发明的化合物的前药,其为自身可具有较小药理学活性或无药理学活性的本发明的化合物的某些衍生物,当被给药至身体中或其上时可通过例如水解裂解转化成具有期望活性的本发明的化合物。通常这样的前药会是所述化合物的官能团衍生物,其易于在体内转化成期望的治疗活性化合物。关于前药的使用的其他信息可参见“Pro-drugs as Novel Delivery Systems”,第14卷,ACS Symposium Series(T.Higuchi及V.Stella)及“Bioreversible Carriers in Drug Design,”Pergamon Press,1987(E.B.Roche编辑,American Pharmaceutical Association)。本发明的前药可例如通过用本领域技术人员已知作为“前-部分(pro-moiety)(例如“Design of Prodrugs”,H.Bundgaard(Elsevier,1985)中所述)”的某些部分替代本发明的化合物中存在的适当官能团来制备。The present invention further includes within its scope the prodrugs of the compounds of the present invention, which are certain derivatives of the compounds of the present invention that may themselves have little or no pharmacological activity, when administered to the body or The above can be converted into the compound of the present invention having the desired activity by, for example, hydrolytic cleavage. Usually such prodrugs will be functional group derivatives of the compound, which are easily converted into the desired therapeutically active compound in vivo. For other information about the use of prodrugs, please refer to "Pro-drugs as Novel Delivery Systems", Volume 14, ACS Symposium Series (T. Higuchi and V. Stella) and "Bioreversible Carriers in Drug Design," Pergamon Press, 1987 ( Edited by EBRoche, American Pharmaceutical Association). The prodrugs of the present invention can be used, for example, by using certain parts known to those skilled in the art as "pro-moiety (for example, "Design of Prodrugs", described in H. Bundgaard (Elsevier, 1985))" It can be prepared by substituting appropriate functional groups present in the compounds of the present invention.
本发明还涵盖含有保护基的本发明的化合物。在制备本发明的化合物的任何过程中,保护在任何有关分子上的敏感基团或反应基团可能是必需的和/或期望的,由此形成本发明的化合物的化学保护的形式。这可以通过常规的保护基实现,例如,在Protective Groups in Organic Chemistry,ed.J.F.W.McOmie,Plenum Press,1973;和T.W.Greene&P.G.M.Wuts,Protective Groups in Organic Synthesis,John Wiley&Sons,1991中所述的那些保护基。这些参考文献通过援引加入本文。使用本领域已知的方法,在适当的后续阶段可以移除保护基。The present invention also encompasses compounds of the present invention containing protecting groups. In any process of preparing the compounds of the present invention, protection of sensitive groups or reactive groups on any relevant molecule may be necessary and/or desirable, thereby forming a chemically protected form of the compounds of the present invention. This can be achieved through conventional protective groups, such as those described in Protective Groups in Organic Chemistry, ed. JFW McOmie, Plenum Press, 1973; and TW Greene & P. GMWuts, Protective Groups in Organic Synthesis, John Wiley & Sons, 1991 Protective base. These references are incorporated herein by reference. Using methods known in the art, the protecting group can be removed at an appropriate subsequent stage.
制备方法Preparation
化合物II的合成方法Synthesis method of compound II
路线1Route 1
其中R 1、R 3和L 2如上述式I中所定义,并且X选自氯、溴和碘。 Wherein R 1 , R 3 and L 2 are as defined in the above formula I, and X is selected from chlorine, bromine and iodine.
第一步:化合物II-1与碳酸胍经关环反应生成化合物II-2。The first step: compound II-1 and guanidine carbonate undergo ring closure reaction to produce compound II-2.
在某些实施方案中,可使用的溶剂为1,4-二氧六环、二甲基亚砜、DMF、N,N-二甲基乙酰胺等。在某些实施方案中,反应温度为0℃至200℃。In certain embodiments, the solvent that can be used is 1,4-dioxane, dimethyl sulfoxide, DMF, N,N-dimethylacetamide, and the like. In certain embodiments, the reaction temperature is 0°C to 200°C.
第二步:化合物II-2与R 1-硼酸或R 1-硼酸酯或R 1-有机锡化合物(例如R 1Sn(n-Bu) 3)经偶联反应(例如Suzuki反应或Stille反应),生成化合物II-3。 The second step: Compound II-2 and R 1 -boronic acid or R 1 -boronic acid ester or R 1 -organotin compound (such as R 1 Sn(n-Bu) 3 ) undergo a coupling reaction (such as Suzuki reaction or Stille reaction) ) To produce compound II-3.
在某些实施方案中,可使用的催化剂为Pd(PPh 3) 4、Pd(dppf)Cl 2·CH 2Cl 2等。在某些实施方案中,可使用的碱为Cs 2CO 3、K 3PO 4、Na 2CO 3、AcOK、NaHCO 3、K 2CO 3等。在某些实施方案中,可使用的溶剂为1,4-二氧六环/H 2O、DMF/H 2O、DMSO/H 2O、CH 3CN/H 2O、甲苯/H 2O等。在某些实施方案中,反应温度为60℃至180℃。 In certain embodiments, the catalysts that can be used are Pd(PPh 3 ) 4 , Pd(dppf)Cl 2 ·CH 2 Cl 2 and the like. In certain embodiments, the bases that can be used are Cs 2 CO 3 , K 3 PO 4 , Na 2 CO 3 , AcOK, NaHCO 3 , K 2 CO 3 and the like. In certain embodiments, the solvents that can be used are 1,4-dioxane/H 2 O, DMF/H 2 O, DMSO/H 2 O, CH 3 CN/H 2 O, toluene/H 2 O Wait. In certain embodiments, the reaction temperature is between 60°C and 180°C.
第三步:化合物II-3与R 3-L 2-PG经取代反应生成化合物II,其中PG为离去基团,其可为卤素(例如F、Cl、Br或I)或磺酸酯。 The third step: Compound II-3 and R 3 -L 2 -PG undergo substitution reaction to produce compound II, where PG is a leaving group, which can be halogen (for example, F, Cl, Br or I) or sulfonate.
在某些实施方案中,可使用的溶剂为DMF、DMSO、THF、CH 3CN、DCM等。在某些实施方案中,可使用的碱为氢化钠、三乙胺、N,N-二异丙基乙胺、叔丁醇钾、氢氧化钠等。在某些实施方案中,反应温度为-20℃至180℃。 In some embodiments, the solvent that can be used is DMF, DMSO, THF, CH 3 CN, DCM, and the like. In certain embodiments, the base that can be used is sodium hydride, triethylamine, N,N-diisopropylethylamine, potassium tert-butoxide, sodium hydroxide, and the like. In certain embodiments, the reaction temperature is -20°C to 180°C.
路线2Route 2
其中R 1、R 3和L 2如上述式I中所定义,并且X选自氯、溴和碘。 Wherein R 1 , R 3 and L 2 are as defined in the above formula I, and X is selected from chlorine, bromine and iodine.
第一步:化合物II-4与氯甲脒盐酸盐经关环反应生成化合物II-5。The first step: compound II-4 and chloroformamidine hydrochloride undergo ring closure reaction to produce compound II-5.
在某些实施方案中,可使用的溶剂为1,4-二氧六环、二甲基砜等。在某些实施方案中,反应温度为0℃至200℃。In certain embodiments, the solvent that can be used is 1,4-dioxane, dimethyl sulfone, and the like. In certain embodiments, the reaction temperature is 0°C to 200°C.
第二步:化合物II-5与R 3-L 2-NH 2经缩合反应生成化合物II-6。 The second step: Compound II-5 and R 3 -L 2 -NH 2 undergo condensation reaction to produce compound II-6.
在某些实施方案中,可使用的缩合剂为BOP、pyBOP、HOBT、HATU、HBTU、TBTU等。在某些实施方案中,可使用的碱为DBU、DIPEA、K 3PO 4、Na 2CO 3、AcOK、NaHCO 3等。在某些实施方案中,反应温度为0℃至180℃。 In certain embodiments, condensing agents that can be used are BOP, pyBOP, HOBT, HATU, HBTU, TBTU, and the like. In certain embodiments, the bases that can be used are DBU, DIPEA, K 3 PO 4 , Na 2 CO 3 , AcOK, NaHCO 3 and the like. In certain embodiments, the reaction temperature is 0°C to 180°C.
第三步:化合物II-6与R 1-硼酸或R 1-硼酸酯或R 1-有机锡化合物(例如R 1Sn(n-Bu) 3)经偶联反应(例如Suzuki反应或Stille反应),生成化合物II。 The third step: Compound II-6 and R 1 -boronic acid or R 1 -boronic acid ester or R 1 -organotin compound (such as R 1 Sn(n-Bu) 3 ) undergo a coupling reaction (such as Suzuki reaction or Stille reaction) ), to produce compound II.
在某些实施方案中,可使用的催化剂为Pd(PPh 3) 4、Pd(dppf)Cl 2·CH 2Cl 2等。在某些实施方案中,可使用的碱为Cs 2CO 3、K 3PO 4、Na 2CO 3、AcOK、NaHCO 3、K 2CO 3等。在某些实施方案中,可使用的溶剂为1,4-二氧六环/H 2O、DMF/H 2O、DMSO/H 2O、CH 3CN/H 2O、甲苯/H 2O等。在某些实施方案中,反应温度为60℃至180℃。 In certain embodiments, the catalysts that can be used are Pd(PPh 3 ) 4 , Pd(dppf)Cl 2 ·CH 2 Cl 2 and the like. In certain embodiments, the bases that can be used are Cs 2 CO 3 , K 3 PO 4 , Na 2 CO 3 , AcOK, NaHCO 3 , K 2 CO 3 and the like. In some embodiments, the solvents that can be used are 1,4-dioxane/H 2 O, DMF/H 2 O, DMSO/H 2 O, CH 3 CN/H 2 O, toluene/H 2 O Wait. In certain embodiments, the reaction temperature is 60°C to 180°C.
化合物IV的合成方法Synthesis method of compound IV
路线3Route 3
其中R 1、R 3和L 2如上述式III中所定义,并且X选自氯、溴和碘。 Wherein R 1 , R 3 and L 2 are as defined in the above formula III, and X is selected from chlorine, bromine and iodine.
第一步:化合物IV-1与氯甲脒盐酸盐经关环反应生成化合物IV-2。The first step: compound IV-1 and chloroformamidine hydrochloride undergo ring closure reaction to produce compound IV-2.
在某些实施方案中,可使用的溶剂为1,4-二氧六环、二甲基砜等。在某些实施方案中,反应温度为0℃至200℃。In certain embodiments, the solvent that can be used is 1,4-dioxane, dimethyl sulfone, and the like. In certain embodiments, the reaction temperature is 0°C to 200°C.
第二步:化合物IV-2与R 3-L 2-NH 2经缩合反应生成化合物IV-3。 The second step: Compound IV-2 and R 3 -L 2 -NH 2 undergo condensation reaction to produce compound IV-3.
在某些实施方案中,可使用的缩合剂为BOP、pyBOP、HOBT、HATU、HBTU、TBTU等。在某些实施方案中,可使用的碱为DBU、DIPEA、K 3PO 4、Na 2CO 3、AcOK、NaHCO 3等。在某些实施方案中,反应温度为0℃至180℃。 In certain embodiments, condensing agents that can be used are BOP, pyBOP, HOBT, HATU, HBTU, TBTU, and the like. In certain embodiments, the bases that can be used are DBU, DIPEA, K 3 PO 4 , Na 2 CO 3 , AcOK, NaHCO 3 and the like. In certain embodiments, the reaction temperature is 0°C to 180°C.
第三步:化合物IV-3与R 1-硼酸或R 1-硼酸酯或R 1-有机锡化合物(例如R 1Sn(n-Bu) 3)经偶联反应(例如Suzuki反应或Stille反应),生成化合物IV。 The third step: Compound IV-3 and R 1 -boronic acid or R 1 -boronic acid ester or R 1 -organotin compound (such as R 1 Sn(n-Bu) 3 ) undergo coupling reaction (such as Suzuki reaction or Stille reaction ) To generate compound IV.
在某些实施方案中,可使用的催化剂为Pd(PPh 3) 4、Pd(dppf)Cl 2·CH 2Cl 2等。在某些实施方案中,可使用的碱为Cs 2CO 3、K 3PO 4、Na 2CO 3、AcOK、NaHCO 3、K 2CO 3等。在某些实施方案中,可使用的溶剂为1,4-二氧六环/H 2O、DMF/H 2O、DMSO/H 2O、CH 3CN/H 2O、甲苯/H 2O等。在某些实施方案中,反应温度为60℃至180℃。 In certain embodiments, the catalysts that can be used are Pd(PPh 3 ) 4 , Pd(dppf)Cl 2 ·CH 2 Cl 2 and the like. In certain embodiments, the bases that can be used are Cs 2 CO 3 , K 3 PO 4 , Na 2 CO 3 , AcOK, NaHCO 3 , K 2 CO 3 and the like. In certain embodiments, the solvents that can be used are 1,4-dioxane/H 2 O, DMF/H 2 O, DMSO/H 2 O, CH 3 CN/H 2 O, toluene/H 2 O Wait. In certain embodiments, the reaction temperature is between 60°C and 180°C.
本发明的化合物对NLRP3及其信号通路有明显的激动活性,无明显毒副作用,可用于与NLRP3炎 症小体活性相关的疾病如细胞增殖异常性疾病(例如癌症)的治疗。The compound of the present invention has obvious agonistic activity on NLRP3 and its signal pathway, without obvious toxic side effects, and can be used for the treatment of diseases related to NLRP3 inflammatory body activity, such as abnormal cell proliferation diseases (e.g. cancer).
以下结合实施例进一步描述本发明,但提供这些实施例并非意图限制本发明的范围。The present invention is further described below in conjunction with examples, but the provision of these examples is not intended to limit the scope of the present invention.
本发明中的缩写具有以下含义:The abbreviations in the present invention have the following meanings:
化合物的结构通过核磁共振波谱( 1H NMR)和/或质谱(MS)鉴定。 The structure of the compound was identified by nuclear magnetic resonance spectroscopy ( 1 H NMR) and/or mass spectrometry (MS).
1H NMR化学位移(δ)以百万分之一(ppm)记录。 1H NMR通过Bruker超导核磁共振波谱仪(型号AVACE III HD 400MHz)测定,溶剂为CD 3OD、CDCl 3或DMSO-d 6,内标为TMS。s:单峰、d:二重峰、t:三重峰、q:四重峰、dd:双二重峰、qd:四二重峰、m:多重峰、br:宽峰(broad)、J:偶合常数、Hz:赫兹。 1 H NMR chemical shifts (δ) are reported in parts per million (ppm). 1 H NMR was measured by Bruker superconducting nuclear magnetic resonance spectrometer (model AVACE III HD 400MHz), the solvent was CD 3 OD, CDCl 3 or DMSO-d 6 , and the internal standard was TMS. s: singlet, d: doublet, t: triplet, q: quartet, dd: doublet, qd: quartet, m: multiplet, br: broad, J : Coupling constant, Hz: Hertz.
反应的监测采用TLC或LC-MS进行。The monitoring of the reaction is carried out by TLC or LC-MS.
TLC采用硅胶GF 254作为固定相。TLC uses silica gel GF 254 as the stationary phase.
LC-MS使用Aglient 1260 Infinity/Aglient 6120 Quadrupole质谱仪记录。LC-MS uses Aglient 1260 Infinity/Aglient 6120 Quadrupole mass spectrometer to record.
化合物可通过制备TLC、硅胶柱层析、Prep-HPLC和/或快速柱层析(Flash柱层析)分离纯化。The compound can be separated and purified by preparative TLC, silica gel column chromatography, Prep-HPLC and/or flash column chromatography (Flash column chromatography).
Prep-HPLC使用Agilent 1260制备液相色谱仪,检测波长为214nm或254nm;色谱柱为Waters SunFire Prep C18 OBD(19mm×150mm×5.0μm);柱温为25℃,并且洗脱条件如下:Prep-HPLC uses Agilent 1260 preparative liquid chromatograph, the detection wavelength is 214nm or 254nm; the chromatographic column is Waters SunFire Prep C18 OBD (19mm×150mm×5.0μm); the column temperature is 25℃, and the elution conditions are as follows:
条件1:10%-90%乙腈和90%-10%甲酸铵水溶液(0.05%),0-16min;流速:24mL/min;Condition 1: 10%-90% acetonitrile and 90%-10% ammonium formate aqueous solution (0.05%), 0-16min; flow rate: 24mL/min;
条件2:10%-46%乙腈和90%-54%碳酸氢铵水溶液(0.05%),0-7.2min;流速:24mL/min;Condition 2: 10%-46% acetonitrile and 90%-54% ammonium bicarbonate aqueous solution (0.05%), 0-7.2min; flow rate: 24mL/min;
条件3:10%-90%乙腈和90%-10%甲酸水溶液(0.05%),0-16min;流速:28mL/min;Condition 3: 10%-90% acetonitrile and 90%-10% formic acid aqueous solution (0.05%), 0-16min; flow rate: 28mL/min;
条件4:10%-90%乙腈和90%-10%碳酸氢铵水溶液(0.05%),0-16min;流速:28mL/min;Condition 4: 10%-90% acetonitrile and 90%-10% ammonium bicarbonate aqueous solution (0.05%), 0-16min; flow rate: 28mL/min;
条件5:10%-60%乙腈和90%-40%甲酸水溶液(0.05%),0-16min;流速:28mL/min;Condition 5: 10%-60% acetonitrile and 90%-40% formic acid aqueous solution (0.05%), 0-16min; flow rate: 28mL/min;
条件6:5%-95%乙腈和95%-5%甲酸水溶液(0.05%),0-20min;流速:28mL/min。Condition 6: 5%-95% acetonitrile and 95%-5% formic acid aqueous solution (0.05%), 0-20min; flow rate: 28mL/min.
条件7:0%-50%乙腈和100%-50%碳酸氢铵水溶液(0.05%),0-20min;流速:28mL/min。Condition 7: 0%-50% acetonitrile and 100%-50% ammonium bicarbonate aqueous solution (0.05%), 0-20min; flow rate: 28mL/min.
硅胶柱层析一般使用200~300目硅胶(青岛海洋)为固定相。洗脱剂体系A:二氯甲烷和甲醇;洗脱剂体系B:石油醚和乙酸乙酯;溶剂的体积比根据化合物的极性不同而进行调节。Silica gel column chromatography generally uses 200-300 mesh silica gel (Qingdao Ocean) as the stationary phase. Eluent system A: dichloromethane and methanol; eluent system B: petroleum ether and ethyl acetate; the volume ratio of the solvent is adjusted according to the polarity of the compound.
快速柱层析使用Biotage快速柱色谱仪。The flash column chromatography uses the Biotage flash column chromatograph.
微波反应使用BiotageInitiator+微波反应器进行。The microwave reaction was carried out using BiotageInitiator+microwave reactor.
在以下实施例中,如无特殊说明,反应的温度为室温(15℃~30℃)。In the following examples, unless otherwise specified, the reaction temperature is room temperature (15°C to 30°C).
本申请中所使用的试剂购自Acros Organics、Aldrich Chemical Company或特伯化学等公司。The reagents used in this application are purchased from companies such as Acros Organics, Aldrich Chemical Company, or Terb Chemical.
实施例1:3-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)-1-丙醇甲酸盐(化合物1s)Example 1: 3-((2-Amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)-1-propanol formate (Compound 1s)
第一步:7-溴喹唑啉-2,4-二胺(化合物1b)The first step: 7-bromoquinazoline-2,4-diamine (compound 1b)
将化合物1a(2g)、碳酸胍(4.68g)加入DMA(20mL)中,N 2保护下加热至150℃反应8h。加入水和乙酸乙酯萃取,有机相浓缩。加入适量的二氯甲烷搅拌数分钟,过滤。滤饼干燥,得到化合物1b(1.5g)。 Compound 1a (2g), guanidine carbonate (4.68 g) was added DMA (20mL), the under N 2 the reaction was heated to 150 deg.] C 8h. Water and ethyl acetate were added for extraction, and the organic phase was concentrated. Add appropriate amount of dichloromethane, stir for several minutes, and filter. The filter cake was dried to obtain compound 1b (1.5 g).
MS(ESI,m/z):241.2[M+H] +. MS(ESI,m/z):241.2[M+H] + .
第二步:7-(1-(四氢-2H-吡喃-2-基)-1H-吡唑-5-基)喹唑啉-2,4-二胺(化合物1c)Step 2: 7-(1-(Tetrahydro-2H-pyran-2-yl)-1H-pyrazol-5-yl)quinazoline-2,4-diamine (Compound 1c)
将化合物1b(500mg)、磷酸钾(1.11g)、Pd(dppf)Cl 2·CH 2Cl 2(0.19g)、1-(2-四氢吡喃基)-1H-吡唑-5-硼酸频哪酯(0.87g)加入DMF和水的混合溶剂(5mL)中,加热至100℃搅拌反应3h。加入水和乙酸乙酯萃取,有机相浓缩至干,再经快速柱层析分离纯化(洗脱剂体系A),得到化合物1c(300mg)。 Compound 1b (500mg), potassium phosphate (1.11g), Pd(dppf)Cl 2 ·CH 2 Cl 2 (0.19g), 1-(2-tetrahydropyranyl)-1H-pyrazole-5-boronic acid Pinacolate (0.87g) was added to the mixed solvent of DMF and water (5mL), heated to 100°C and stirred for 3h. Water and ethyl acetate were added for extraction, the organic phase was concentrated to dryness, and then separated and purified by flash column chromatography (eluent system A) to obtain compound 1c (300 mg).
MS(ESI,m/z):311.2[M+H] +. MS(ESI,m/z):311.2[M+H] + .
第三步:3-((2-氨基-7-(1-(四氢-2H-吡喃-2-基)-1H-吡唑-5-基)喹唑啉-4-基)氨基)-1-丙醇(化合物1d)The third step: 3-((2-amino-7-(1-(tetrahydro-2H-pyran-2-yl)-1H-pyrazol-5-yl)quinazolin-4-yl)amino) -1-propanol (Compound 1d)
将化合物1c(100mg)加入DMSO(2mL)中,降温至0℃。缓慢加入NaH(6.17mg),再加入3-溴-1-丙醇(88.96mg),并于0℃下反应16h。加水淬灭反应,反应液通过Prep-HPLC分离纯化(洗脱条件3),冻干得到化合物1d(30mg)。Compound 1c (100 mg) was added to DMSO (2 mL), and the temperature was lowered to 0°C. Slowly add NaH (6.17mg), then add 3-bromo-1-propanol (88.96mg), and react at 0°C for 16h. The reaction was quenched by adding water, and the reaction solution was separated and purified by Prep-HPLC (elution condition 3), and lyophilized to obtain compound 1d (30 mg).
MS(ESI,m/z):369.1[M+H] +. MS(ESI,m/z): 369.1[M+H] + .
第四步:3-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)-1-丙醇甲酸盐(化合物1s)The fourth step: 3-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)-1-propanol formate (compound 1s)
将化合物1d(30mg)加入TFA(2mL)中,室温搅拌反应2h。减压浓缩,得到粗品化合物1,再通过Prep-HPLC分离纯化(洗脱条件3),冻干得到化合物1s(20mg)。Compound 1d (30mg) was added to TFA (2mL), and the reaction was stirred at room temperature for 2h. Concentrated under reduced pressure to obtain crude compound 1, which was separated and purified by Prep-HPLC (elution condition 3), and lyophilized to obtain compound 1s (20 mg).
MS(ESI,m/z):285.1[M+H] +. MS(ESI,m/z):285.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.21(s,1H),9.02(s,1H),8.22(d,J=8.4Hz,1H),7.85-7.76(m,5H),6.86(d,1H),4.59(s,1H),3.64-3.59(m,2H),3.53-3.50(m,2H),1.85-1.79(m,2H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.21 (s, 1H), 9.02 (s, 1H), 8.22 (d, J = 8.4 Hz, 1H), 7.85-7.76 (m, 5H), 6.86 ( d, 1H), 4.59 (s, 1H), 3.64-3.59 (m, 2H), 3.53-3.50 (m, 2H), 1.85-1.79 (m, 2H).
实施例2:N 4-(2-甲氧基乙基)-7-(1H-吡唑-5-基)喹唑啉-2,4-二胺(化合物2) Example 2: N 4 -(2-methoxyethyl)-7-(1H-pyrazol-5-yl)quinazoline-2,4-diamine (Compound 2)
第一步:2-氨基-7-溴喹唑啉-4-醇(化合物2b)The first step: 2-amino-7-bromoquinazolin-4-ol (compound 2b)
向单口瓶中依次加入化合物2a(5.00g)、氯甲脒盐酸盐(3.90g)、二甲基砜(12.75g),加热至155℃反应2h。将反应液抽滤,滤饼移至水(0.5L)中搅碎并于室温下打浆0.5h。再次抽滤,滤饼用水洗涤,烘干后得到化合物2b(4.70g)。Compound 2a (5.00 g), chloroformamidine hydrochloride (3.90 g), and dimethyl sulfone (12.75 g) were sequentially added to a single-neck flask, and heated to 155° C. for reaction for 2 hours. The reaction solution was suction filtered, the filter cake was transferred to water (0.5L) and smashed and beaten at room temperature for 0.5h. Suction filtration was performed again, the filter cake was washed with water, and dried to obtain compound 2b (4.70 g).
MS(ESI,m/z):240.0[M+H] +. MS(ESI,m/z): 240.0[M+H] + .
第二步:7-溴-N 4-(2-甲氧基乙基)喹唑啉-2,4-二胺(化合物2c) The second step: 7-bromo-N 4 -(2-methoxyethyl)quinazoline-2,4-diamine (compound 2c)
室温下,将化合物2b(55mg)加入DMF(3mL)中,继续依次加入DBU(104.64mg)和BOP(121.60mg),并搅拌5min。加入2-甲氧基乙胺(127mg),反应16h。将反应液抽滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物2c(8mg)。At room temperature, compound 2b (55 mg) was added to DMF (3 mL), DBU (104.64 mg) and BOP (121.60 mg) were added successively, and the mixture was stirred for 5 min. Add 2-methoxyethylamine (127mg) and react for 16h. The reaction solution was suction filtered, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 2c (8 mg).
MS(ESI,m/z):296.9[M+H] +. MS(ESI,m/z): 296.9[M+H] + .
第三步:N 4-(2-甲氧基乙基)-7-(1H-吡唑-5-基)喹唑啉-2,4-二胺(化合物2) The third step: N 4 -(2-methoxyethyl)-7-(1H-pyrazol-5-yl)quinazoline-2,4-diamine (compound 2)
将化合物2c(8mg)、1H-吡唑-5-硼酸频哪酯(10.45mg)、Pd(dppf)Cl 2·CH 2Cl 2(2.20mg)和碳酸钾(11.16mg)加入DMF(3mL)和水(0.5mL)中,N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物2(4mg)。 Compound 2c (8mg), 1H-pyrazole-5-boronic acid pinacolate (10.45mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (2.20mg) and potassium carbonate (11.16mg) were added to DMF (3mL) In water (0.5mL), heated to 100°C under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 2 (4 mg).
MS(ESI,m/z):285.1[M+H] +. MS(ESI,m/z):285.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ12.96(s,1H),8.00(d,J=8.5Hz,1H),7.89(s,1H),7.79(s,1H),7.60(s,1H),7.49(s,1H),6.80(d,J=2.1Hz,1H),6.10(s,2H),3.69–3.60(m,2H),3.59–3.52(m,2H),3.29(s,3H). 1 H NMR (DMSO-d 6 , 400MHz) δ 12.96 (s, 1H), 8.00 (d, J = 8.5 Hz, 1H), 7.89 (s, 1H), 7.79 (s, 1H), 7.60 (s, 1H),7.49(s,1H),6.80(d,J=2.1Hz,1H),6.10(s,2H),3.69–3.60(m,2H),3.59–3.52(m,2H), 3.29(s ,3H).
实施例3:(1S,2R)-2-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)环戊醇(化合物3)Example 3: (1S,2R)-2-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)cyclopentanol (compound 3)
第一步:(1S,2R)-2-((2-氨基-7-溴喹唑啉-4-基)氨基)环戊醇(化合物3a)The first step: (1S,2R)-2-((2-amino-7-bromoquinazolin-4-yl)amino)cyclopentanol (compound 3a)
室温下,将化合物2b(50mg)加入DMF(3mL)中,继续依次加入DIPEA(134.60mg)和BOP(110.54mg),并搅拌5min。加入(1S,2R)-2-氨基环戊醇盐酸盐(86mg),反应16h,得到粗品化合物3a,将其直接用于下一步反应。At room temperature, compound 2b (50 mg) was added to DMF (3 mL), DIPEA (134.60 mg) and BOP (110.54 mg) were added successively, and stirred for 5 min. (1S, 2R)-2-aminocyclopentanol hydrochloride (86 mg) was added and reacted for 16 h to obtain crude compound 3a, which was directly used in the next reaction.
MS(ESI,m/z):323.0[M+H] +. MS(ESI,m/z):323.0[M+H] + .
第二步:(1S,2R)-2-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)环戊醇(化合物3)The second step: (1S,2R)-2-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)cyclopentanol (compound 3)
向第一步中得到的化合物3a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(80.82mg)、Pd(dppf)Cl 2·CH 2Cl 2(17mg)、K 2CO 3(86.35mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物3(15mg)。 To the reaction solution of compound 3a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (80.82mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (17mg), K 2 CO were sequentially added 3 (86.35mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 3 (15 mg).
MS(ESI,m/z):311.0[M+H] +. MS(ESI,m/z):311.0[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ12.97(s,1H),8.14–8.00(m,1H),7.81(s,1H),7.61(s,1H),7.58–7.50(m,1H),7.21(s,1H),6.80(s,1H),6.05(s,2H),4.77(d,J=3.6Hz,1H),4.38–4.28(m,1H),4.22–4.15(m,1H),2.00–1.87(m,1H),1.84–1.78(m,2H),1.70–1.60(m,1H),1.58–1.44(m,1H),1.27–1.12(m,1H). 1 H NMR(DMSO-d 6 ,400MHz)δ12.97(s,1H), 8.14-8.00(m,1H), 7.81(s,1H), 7.61(s,1H), 7.58-7.50(m,1H) ), 7.21 (s, 1H), 6.80 (s, 1H), 6.05 (s, 2H), 4.77 (d, J = 3.6 Hz, 1H), 4.38-4.28 (m, 1H), 4.22-4.15 (m, 1H), 2.00-1.87(m, 1H), 1.84--1.78(m, 2H), 1.70-1.60(m, 1H), 1.58-1.44(m, 1H), 1.27-1.12(m, 1H).
实施例4:(1R,3R)-3-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)环戊醇甲酸盐(化合物4s)Example 4: (1R,3R)-3-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)cyclopentanol formate (compound 4s)
第一步:(1R,3R)-3-((2-氨基-7-溴喹唑啉-4-基)氨基)环戊醇(化合物4a)The first step: (1R,3R)-3-((2-amino-7-bromoquinazolin-4-yl)amino)cyclopentanol (compound 4a)
室温下,将化合物2b(50mg)加入DMF(3mL)中,继续依次加入DIPEA(134.60mg)和BOP(110.54mg),搅拌5min。加入(1R,3R)-3-氨基环戊醇盐酸盐(86mg),反应16h,得到粗品化合物4a,直接用于下一步反应。At room temperature, compound 2b (50 mg) was added to DMF (3 mL), DIPEA (134.60 mg) and BOP (110.54 mg) were added successively, and the mixture was stirred for 5 min. (1R, 3R)-3-aminocyclopentanol hydrochloride (86 mg) was added and reacted for 16 h to obtain crude compound 4a, which was directly used in the next reaction.
MS(ESI,m/z):323.0[M+H] +. MS(ESI,m/z):323.0[M+H] + .
第二步:(1R,3R)-3-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)环戊醇甲酸盐(化合物4s)The second step: (1R,3R)-3-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)cyclopentanol formate (compound 4s)
向第一步中得到的化合物4a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(80.82mg)、Pd(dppf)Cl 2·CH 2Cl 2(17mg)、K 2CO 3(86.35mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,含化合物4的滤液通过Prep-HPLC分离纯化(洗脱条件3),冻干得到化合物4s(16mg)。 To the reaction solution of compound 4a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (80.82mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (17mg), K 2 CO were sequentially added 3 (86.35mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate containing compound 4 was separated and purified by Prep-HPLC (elution condition 3), and lyophilized to obtain compound 4s (16 mg).
MS(ESI,m/z):311.0[M+H] +. MS(ESI,m/z):311.0[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.12(s,1H),8.30(s,1H),8.15(d,J=8.5Hz,1H),8.07(s,1H),7.79(s,1H),7.68(s,1H),7.60(d,J=8.3Hz,1H),7.10(s,2H),6.82(d,J=1.9Hz,1H),4.90–4.77(m,1H),4.32–4.25(m,1H),2.24–2.10(m,1H),2.05–1.88(m,2H),1.88–1.74(m,1H),1.63–1.43(m,2H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.12 (s, 1H), 8.30 (s, 1H), 8.15 (d, J = 8.5 Hz, 1H), 8.07 (s, 1H), 7.79 (s, 1H), 7.68 (s, 1H), 7.60 (d, J = 8.3 Hz, 1H), 7.10 (s, 2H), 6.82 (d, J = 1.9 Hz, 1H), 4.90-4.77 (m, 1H), 4.32–4.25(m,1H), 2.24–2.10(m,1H), 2.05–1.88(m,2H), 1.88–1.74(m,1H), 1.63–1.43(m,2H).
实施例5:(1S,3S)-3-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)环戊醇甲酸盐(化合物5s)Example 5: (1S,3S)-3-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)cyclopentanol formate (compound 5s)
第一步:(1S,3S)-3-((2-氨基-7-溴喹唑啉-4-基)氨基)环戊醇(化合物5a)The first step: (1S,3S)-3-((2-amino-7-bromoquinazolin-4-yl)amino)cyclopentanol (compound 5a)
室温下,将化合物2b(50mg)加入DMF(3mL)中,继续依次加入DIPEA(134.60mg)和BOP(110.54mg),搅拌5min。加入(1S,3S)-3-氨基环戊醇盐酸盐(86mg),反应16h,得到粗品化合物5a,直接用于下一步反应。At room temperature, compound 2b (50 mg) was added to DMF (3 mL), DIPEA (134.60 mg) and BOP (110.54 mg) were added successively, and the mixture was stirred for 5 min. (1S,3S)-3-aminocyclopentanol hydrochloride (86 mg) was added and reacted for 16 h to obtain crude compound 5a, which was directly used in the next reaction.
MS(ESI,m/z):323.0[M+H] +. MS(ESI,m/z):323.0[M+H] + .
第二步:(1S,3S)-3-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)环戊醇甲酸盐(化合物5s)Step 2: (1S,3S)-3-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)cyclopentanol formate (compound 5s)
向第一步中得到的化合物5a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(80.82mg)、Pd(dppf)Cl 2·CH 2Cl 2(17mg)、K 2CO 3(86.35mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,含化合物5的滤液通过Prep-HPLC分离纯化(洗脱条件3),冻干得到化合物5s(2mg)。 To the reaction solution of compound 5a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (80.82mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (17mg), K 2 CO were sequentially added 3 (86.35mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate containing compound 5 was separated and purified by Prep-HPLC (elution condition 3), and lyophilized to obtain compound 5s (2 mg).
MS(ESI,m/z):311.0[M+H] +. MS(ESI,m/z):311.0[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.12(s,1H),8.27(s,1H),8.14(d,J=8.5Hz,1H),8.00(s,1H),7.78(s,1H),7.67(s,1H),7.58(d,J=7.8Hz,1H),7.25–6.70(m,3H),4.90–4.74(m,1H),4.34–4.23(m,1H),2.24–2.11(m,1H),2.04–1.88(m,2H),1.86–1.74(m,1H),1.62–1.43(m,2H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.12 (s, 1H), 8.27 (s, 1H), 8.14 (d, J = 8.5 Hz, 1H), 8.00 (s, 1H), 7.78 (s, 1H), 7.67(s,1H), 7.58(d,J=7.8Hz,1H), 7.25–6.70(m,3H), 4.90–4.74(m,1H), 4.34–4.23(m,1H), 2.24 --2.11(m,1H), 2.04–1.88(m,2H), 1.86–1.74(m,1H), 1.62–1.43(m,2H).
实施例6:(R)-1-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)-2-丙醇甲酸盐(化合物6s)Example 6: (R)-1-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)-2-propanol formate (compound 6s)
第一步:(R)-1-((2-氨基-7-溴喹唑啉-4-基)氨基)-2-丙醇(化合物6a)The first step: (R)-1-((2-amino-7-bromoquinazolin-4-yl)amino)-2-propanol (Compound 6a)
室温下,将化合物2b(200mg)、(R)-(-)-1-氨基-2-丙醇(187.73mg)、BOP(442.18mg)和DIPEA(538.37mg)加入DMF(5mL)中,反应16h,得到粗品化合物6a,直接用于下一步反应。At room temperature, compound 2b (200mg), (R)-(-)-1-amino-2-propanol (187.73mg), BOP (442.18mg) and DIPEA (538.37mg) were added to DMF (5mL) to react At 16h, the crude compound 6a was obtained, which was directly used in the next reaction.
MS(ESI,m/z):298.1[M+H] +. MS(ESI,m/z): 298.1[M+H] + .
第二步:(R)-1-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)-2-丙醇甲酸盐(化合物6s)The second step: (R)-1-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)-2-propanol formate (compound 6s)
向第一步中得到的化合物6a的反应液中,依次加入碳酸钾(111.62mg)、Pd(dppf)Cl 2·CH 2Cl 2(21.99mg)和1H-吡唑-5-硼酸频哪酯(104.48mg),N 2保护下加热至100℃反应3h。将反应液硅藻土过滤,含化合物6的滤液通过Prep-HPLC分离纯化(洗脱条件6),冻干得到化合物6s(20mg)。 To the reaction solution of compound 6a obtained in the first step, potassium carbonate (111.62 mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (21.99 mg) and 1H-pyrazole-5-boronic acid pinacolate were sequentially added (104.48mg), heated to 100°C under N 2 protection, and reacted for 3h. The reaction solution was filtered through Celite, and the filtrate containing compound 6 was separated and purified by Prep-HPLC (elution condition 6), and lyophilized to obtain compound 6s (20 mg).
MS(ESI,m/z):285.1[M+H] +. MS(ESI,m/z):285.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ12.93(s,1H),8.46(s,1H),8.35(s,1H),8.16(d,J=8.5Hz,1H),7.80(s,1H),7.72(s,1H),7.65(d,J=8.3Hz,1H),7.51–7.08(m,2H),6.84(d,J=1.7Hz,1H),3.98(dq,J=12.4,6.1Hz,1H),3.51(dt,J=10.1,5.1Hz,1H),3.45–3.36(m,1H),1.12(d,J=6.1Hz,3H). 1 H NMR (DMSO-d 6 , 400MHz) δ 12.93 (s, 1H), 8.46 (s, 1H), 8.35 (s, 1H), 8.16 (d, J = 8.5 Hz, 1H), 7.80 (s, 1H), 7.72 (s, 1H), 7.65 (d, J = 8.3 Hz, 1H), 7.51-7.08 (m, 2H), 6.84 (d, J = 1.7 Hz, 1H), 3.98 (dq, J = 12.4 ,6.1Hz,1H),3.51(dt,J=10.1,5.1Hz,1H),3.45-3.36(m,1H),1.12(d,J=6.1Hz,3H).
实施例7:7-(1H-吡唑-5-基)-N 4-(四氢呋喃-3-基)喹唑啉-2,4-二胺甲酸盐(化合物7s) Example 7: 7-(1H-pyrazol-5-yl)-N 4 -(tetrahydrofuran-3-yl)quinazoline-2,4-diamine formate (Compound 7s)
第一步:7-溴-N 4-(四氢呋喃-3-基)喹唑啉-2,4-二胺(化合物7a) The first step: 7-bromo-N 4 -(tetrahydrofuran-3-yl)quinazoline-2,4-diamine (compound 7a)
室温下,将化合物2b(200mg)、3-氨基四氢呋喃(217.75mg)、BOP(442.18mg)和DIPEA(538.37mg)加入DMF(5mL)中,反应16h,得到粗品化合物7a,直接用于下一步反应。At room temperature, compound 2b (200mg), 3-aminotetrahydrofuran (217.75mg), BOP (442.18mg) and DIPEA (538.37mg) were added to DMF (5mL) and reacted for 16h to obtain crude compound 7a, which was used directly in the next step reaction.
MS(ESI,m/z):310.1[M+H] +. MS(ESI,m/z): 310.1[M+H] + .
第二步:7-(1H-吡唑-5-基)-N 4-(四氢呋喃-3-基)喹唑啉-2,4-二胺甲酸盐(7s) The second step: 7-(1H-pyrazol-5-yl)-N 4 -(tetrahydrofuran-3-yl)quinazoline-2,4-diamine formate (7s)
向第一步中得到的化合物7a的反应液中,依次加入碳酸钾(107.29mg)、Pd(dppf)Cl 2·CH 2Cl 2(21.13mg)和1H-吡唑-5-硼酸频哪酯(100.42mg),N 2保护下加热至100℃反应3h。将反应液硅藻土过滤,含化合物7的滤液通过Prep-HPLC分离纯化(洗脱条件6),冻干得到化合物7s(35mg)。 To the reaction solution of compound 7a obtained in the first step, potassium carbonate (107.29mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (21.13mg) and 1H-pyrazole-5-boronic acid pinacolate were sequentially added (100.42mg), heated to 100°C under the protection of N 2 and reacted for 3h. The reaction solution was filtered through Celite, and the filtrate containing compound 7 was separated and purified by Prep-HPLC (elution condition 6), and lyophilized to obtain compound 7s (35 mg).
MS(ESI,m/z):297.1[M+H] +. MS(ESI,m/z): 297.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.08(s,1H),8.33(s,2H),8.24(d,J=8.5Hz,1H),7.80(s,1H),7.72(s,1H),7.64(d,J=8.3Hz,1H),7.42–7.12(m,2H),6.83(t,J=4.1Hz,1H),4.74(td,J=11.6,5.8Hz,1H),4.02–3.95(m,1H),3.95–3.89(m,1H),3.76(dd,J=14.4,7.9Hz,1H),3.68(dd,J=9.0,4.5Hz,1H),2.26(td,J=14.3,7.7Hz,1H),2.07(td,J=12.5,6.5Hz,1H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.08 (s, 1H), 8.33 (s, 2H), 8.24 (d, J = 8.5 Hz, 1H), 7.80 (s, 1H), 7.72 (s, 1H), 7.64 (d, J = 8.3 Hz, 1H), 7.42-7.12 (m, 2H), 6.83 (t, J = 4.1 Hz, 1H), 4.74 (td, J = 11.6, 5.8 Hz, 1H), 4.02–3.95(m,1H), 3.95–3.89(m,1H), 3.76(dd,J=14.4,7.9Hz,1H), 3.68(dd,J=9.0,4.5Hz,1H), 2.26(td, J = 14.3, 7.7 Hz, 1H), 2.07 (td, J = 12.5, 6.5 Hz, 1H).
实施例8:N-(2-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)乙基)-N-甲基丙酰胺(化合物8)Example 8: N-(2-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)ethyl)-N-methylpropionamide (Compound 8 )
第一步:叔丁基(2-((2-氨基-7-溴喹唑啉-4-基)氨基)乙基)(甲基)氨基甲酸酯(化合物8a)The first step: tert-butyl (2-((2-amino-7-bromoquinazolin-4-yl)amino)ethyl)(methyl)carbamate (compound 8a)
室温下,将化合物2b(0.50g)加入DMF(20mL)中,继续依次加入DIPEA(1.88g)和BOP(1.11g),搅拌5min。继续加入N-Boc-N-甲基乙二胺(1.09g),反应16h,得到粗品化合物8a,直接用于下一步反应。At room temperature, compound 2b (0.50 g) was added to DMF (20 mL), DIPEA (1.88 g) and BOP (1.11 g) were added successively, and the mixture was stirred for 5 min. Continue to add N-Boc-N-methylethylenediamine (1.09g) and react for 16h to obtain the crude compound 8a, which is directly used in the next reaction.
MS(ESI,m/z):396.1[M+H] +. MS(ESI,m/z): 396.1[M+H] + .
第二步:叔丁基(2-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)乙基)(甲基)氨基甲酸酯(化合物8b)The second step: tert-butyl (2-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)ethyl)(methyl)carbamate( Compound 8b)
向第一步中得到的化合物8a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(807.20mg)、Pd(dppf)Cl 2·CH 2Cl 2(170mg)、K 2CO 3(862.41mg)和水(3.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件7),浓缩后得到化合物8b(525mg)。 To the reaction solution of compound 8a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (807.20mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (170mg), K 2 CO were added sequentially. 3 (862.41mg) and water (3.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, the filtrate was separated and purified by Prep-HPLC (elution condition 7), and compound 8b (525 mg) was obtained after concentration.
MS(ESI,m/z):384.2[M+H] +. MS(ESI,m/z): 384.2[M+H] + .
第三步:N 4-(2-(甲基氨基)乙基)-7-(1H-吡唑-5-基)喹唑啉-2,4-二胺(化合物8c) The third step: N 4 -(2-(methylamino)ethyl)-7-(1H-pyrazol-5-yl)quinazoline-2,4-diamine (Compound 8c)
室温下,将化合物8b(525mg)加入DCM(6mL)中,继续加入TFA(2mL),反应1h,浓缩后得到粗品化合物8c(560mg)。At room temperature, compound 8b (525 mg) was added to DCM (6 mL), and TFA (2 mL) was continuously added, and reacted for 1 h. After concentration, the crude compound 8c (560 mg) was obtained.
MS(ESI,m/z):284.2[M+H] +. MS(ESI,m/z):284.2[M+H] + .
第四步:N-(2-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)乙基)-N-甲基丙酰胺(化合物8)The fourth step: N-(2-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)ethyl)-N-methylpropionamide (Compound 8 )
将化合物8c(100mg)、丙酸(28.76mg)、HBTU(134.40mg)和DIPEA(228.07mg)加入DMF(2mL)中,反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物8(3mg)。Compound 8c (100 mg), propionic acid (28.76 mg), HBTU (134.40 mg) and DIPEA (228.07 mg) were added to DMF (2 mL) and reacted for 4 hours. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 8 (3 mg).
MS(ESI,m/z):340.1[M+H] +. MS(ESI,m/z): 340.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.02(s,1H),8.06-8.04(m,1H),7.93-7.91(m,1H),7.75(s,1H),7.62(s,1H),7.52-7.50(m,1H),6.81-6.80(m,1H),6.30-6.20(m,2H),3.64-3.45(m,4H),3.01-2.88(m,3H),2.31-2.25(m,4H),0.97-0.87(m,3H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.02 (s, 1H), 8.06-8.04 (m, 1H), 7.93-7.91 (m, 1H), 7.75 (s, 1H), 7.62 (s, 1H) ),7.52-7.50(m,1H),6.81-6.80(m,1H),6.30-6.20(m,2H),3.64-3.45(m,4H),3.01-2.88(m,3H),2.31-2.25 (m,4H),0.97-0.87(m,3H).
实施例9:N-(2-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)乙基)-N-甲基乙酰胺(化合物9)Example 9: N-(2-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)ethyl)-N-methylacetamide (Compound 9 )
将化合物8c(100mg)、乙酸(23.31mg)、HBTU(134.40mg)和DIPEA(228.07mg)加入DMF(2mL)中,反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物9(3mg)。Compound 8c (100 mg), acetic acid (23.31 mg), HBTU (134.40 mg) and DIPEA (228.07 mg) were added to DMF (2 mL) and reacted for 4 hours. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 9 (3 mg).
MS(ESI,m/z):326.1[M+H] +. MS(ESI,m/z): 326.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.02(s,1H),8.04-7.91(m,2H),7.75(s,1H),7.62(s,1H),7.52-7.50(m,1H),6.81-6.80(m,1H),6.20-6.16(m,2H),3.66-3.45(m,4H),3.02-2.87(m,3H),1.97-1.95(m,3H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.02 (s, 1H), 8.04-7.91 (m, 2H), 7.75 (s, 1H), 7.62 (s, 1H), 7.52-7.50 (m, 1H) ), 6.81-6.80 (m, 1H), 6.20-6.16 (m, 2H), 3.66-3.45 (m, 4H), 3.02-2.87 (m, 3H), 1.97-1.95 (m, 3H).
实施例10:3-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)-1-丙醇(化合物10)Example 10: 3-((2-Amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)-1-propanol (Compound 10)
第一步:2-氨基-6-碘噻吩并[3,2-d]嘧啶-4-醇(化合物10b)The first step: 2-amino-6-iodothieno[3,2-d]pyrimidin-4-ol (compound 10b)
将化合物10a(1.00g)、氯甲脒盐酸盐(415.96mg)、二甲基砜(997.54mg)加至反应瓶内,加热至150℃反应4h。加水(6mL),加入氨水调节pH=9,继续搅拌0.5h。打浆过滤,滤饼干燥后得到化合物10b(0.98g)。Compound 10a (1.00 g), chloroformamidine hydrochloride (415.96 mg), and dimethyl sulfone (997.54 mg) were added to the reaction flask and heated to 150° C. for reaction for 4 hours. Add water (6 mL), add ammonia water to adjust pH=9, and continue stirring for 0.5 h. After beating and filtering, the filter cake was dried to obtain compound 10b (0.98 g).
MS(ESI,m/z):294[M+H] +. MS(ESI,m/z):294[M+H] + .
第二步:3-((2-氨基-6-碘噻吩并[3,2-d]嘧啶-4-基)氨基)-1-丙醇(化合物10c)The second step: 3-((2-amino-6-iodothieno[3,2-d]pyrimidin-4-yl)amino)-1-propanol (compound 10c)
将化合物10b(80.00mg)、3-氨基-1-丙醇(61.51mg)、PyBOP(184.66mg)、DIPEA(176.39mg)和DMF(5.0mL)加至反应瓶内,室温下搅拌16h。乙酸乙酯萃取3次,干燥浓缩,通过Prep-HPLC分离纯化(洗脱条件1),冻干得到化合物10c(25.0mg)。Compound 10b (80.00 mg), 3-amino-1-propanol (61.51 mg), PyBOP (184.66 mg), DIPEA (176.39 mg) and DMF (5.0 mL) were added to the reaction flask and stirred at room temperature for 16 h. It was extracted three times with ethyl acetate, dried and concentrated, separated and purified by Prep-HPLC (elution condition 1), and lyophilized to obtain compound 10c (25.0 mg).
MS(ESI,m/z):351.1[M+H] +. MS(ESI,m/z):351.1[M+H] + .
第三步:3-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)-1-丙醇(化合物10)The third step: 3-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)-1-propanol (Compound 10)
将化合物10c(20.00mg)、1H-吡唑-5-硼酸频哪酯(14.41mg)、Pd(dppf)Cl 2·CH 2Cl 2(9.33mg)、K 3PO 4(24.25mg)、水(1.0mL)和1,4-二氧六环(2.5mL)加至反应瓶内,N 2置换10min,加热至100℃反应5h。乙酸乙酯萃取,干燥浓缩,通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物10(14.0mg)。 Compound 10c (20.00mg), 1H-pyrazole-5-boronic acid pinacle ester (14.41mg), Pd(dppf)Cl 2 .CH 2 Cl 2 (9.33mg), K 3 PO 4 (24.25mg), water (1.0 mL) and 1,4-dioxane (2.5 mL) were added to the reaction flask, replaced with N 2 for 10 minutes, and heated to 100°C for 5 hours. It was extracted with ethyl acetate, dried and concentrated, separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 10 (14.0 mg).
MS(ESI,m/z):291[M+H] +. MS(ESI,m/z):291[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.06(s,1H),7.84(s,1H),7.24(s,1H),7.18(s,1H),6.78(s,1H),5.85(s,2H),4.51(s,1H),3.54–3.42(m,4H),1.74(p,J=6.6Hz,2H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.06 (s, 1H), 7.84 (s, 1H), 7.24 (s, 1H), 7.18 (s, 1H), 6.78 (s, 1H), 5.85 ( s,2H),4.51(s,1H),3.54-3.42(m,4H),1.74(p,J=6.6Hz,2H).
实施例11:N-(2-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)乙基)-N-甲基环丙酰胺(化合物11)Example 11: N-(2-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)ethyl)-N-methylcyclopropanamide (compound 11)
室温下,将化合物8c(53mg)、环丙甲酸(12.63mg)和HBTU(60.70mg)加入DMF(2mL)中,继续加入DIPEA(86.19mg),反应2h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物11(10mg)。At room temperature, compound 8c (53 mg), cyclopropanoic acid (12.63 mg) and HBTU (60.70 mg) were added to DMF (2 mL), DIPEA (86.19 mg) was added continuously, and the reaction was continued for 2 h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 11 (10 mg).
MS(ESI,m/z):351.9[M+H] +. MS(ESI,m/z):351.9[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.7–12.7(m,1H),8.45–7.97(m,1H),7.96–7.74(m,2H),7.61(s,1H),7.52(s,1H),6.80(s,1H),6.23–5.90(m,2H),3.79–3.64(m,2H),3.62–3.24(m,2H),3.22–2.86(m,3H),1.96–1.83(m,1H),0.74–0.47(m,4H). 1 H NMR(DMSO-d 6 ,400MHz)δ13.7--12.7(m,1H), 8.45-7.97(m,1H), 7.96-7.74(m,2H), 7.61(s,1H), 7.52(s ,1H),6.80(s,1H),6.23--5.90(m,2H),3.79-3.64(m,2H),3.62--3.24(m,2H),3.22-2.86(m,3H),1.96-1.83 (m,1H),0.74-0.47(m,4H).
实施例12:(2S)-1-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)-2-丙醇(化合物12)Example 12: (2S)-1-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)-2-propanol (Compound 12)
第一步:(2S)-1-[(2-氨基-6-碘噻吩并[3,2-d]嘧啶-4-基)氨基]-2-丙醇(化合物12a)The first step: (2S)-1-[(2-amino-6-iodothieno[3,2-d]pyrimidin-4-yl)amino]-2-propanol (Compound 12a)
将化合物10b(80.00mg)、(S)-1-氨基-2-丙醇(61.51mg)、PyBOP(184.66mg)、DIPEA(176.39mg)和DMF(5.0mL)加至反应瓶内,室温下搅拌16h。乙酸乙酯萃取3次,干燥浓缩,通过Prep-HPLC分离纯化(洗脱条件1),冻干得到化合物12a(25.0mg)。Compound 10b (80.00mg), (S)-1-amino-2-propanol (61.51mg), PyBOP (184.66mg), DIPEA (176.39mg) and DMF (5.0mL) were added to the reaction flask at room temperature Stir for 16h. It was extracted three times with ethyl acetate, dried and concentrated, separated and purified by Prep-HPLC (elution condition 1), and lyophilized to obtain compound 12a (25.0 mg).
MS(ESI,m/z):351.1[M+H] +. MS(ESI,m/z):351.1[M+H] + .
第二步:(2S)-1-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)-2-丙醇(化合物12)The second step: (2S)-1-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)-2-propanol (Compound 12)
将化合物12a(20.00mg)、1H-吡唑-5-硼酸频哪酯(14.41mg)、Pd(dppf)Cl 2·CH 2Cl 2(9.33mg)、K 3PO 4(24.25mg)、水(1.0mL)和1,4-二氧六环(2.5mL)加至反应瓶内,N 2置换10min,加热至100℃反应5h。乙酸乙酯萃取,干燥浓缩,通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物12(14.0mg)。 Compound 12a (20.00mg), 1H-pyrazole-5-boronic acid pinacle ester (14.41mg), Pd(dppf)Cl 2 .CH 2 Cl 2 (9.33mg), K 3 PO 4 (24.25mg), water (1.0 mL) and 1,4-dioxane (2.5 mL) were added to the reaction flask, replaced with N 2 for 10 minutes, and heated to 100°C for 5 hours. It was extracted with ethyl acetate, dried and concentrated, separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 12 (14.0 mg).
MS(ESI,m/z):291[M+H] +. MS(ESI,m/z):291[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.12(s,1H),8.14(s,1H),7.87(s,1H),7.61(s,1H),7.29(s,1H),6.82(s,1H),6.31(s,2H),3.89(q,J=6.2Hz,1H),3.94–3.42(m,2H),1.09(d,J=6.2Hz,3H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.12 (s, 1H), 8.14 (s, 1H), 7.87 (s, 1H), 7.61 (s, 1H), 7.29 (s, 1H), 6.82 ( s, 1H), 6.31 (s, 2H), 3.89 (q, J = 6.2 Hz, 1H), 3.94-3.42 (m, 2H), 1.09 (d, J = 6.2 Hz, 3H).
实施例13:3-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)-2,2-二甲基-1-丙醇(化合物13)Example 13: 3-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)-2,2-dimethyl-1-propanol (compound 13 )
第一步:3-((2-氨基-7-溴喹唑啉-4-基)氨基)-2,2-二甲基-1-丙醇(化合物13a)The first step: 3-((2-amino-7-bromoquinazolin-4-yl)amino)-2,2-dimethyl-1-propanol (compound 13a)
将化合物2b(100mg)、3-氨基-2,2-二甲基-1-丙醇(128.92mg)、BOP(276.18mg)和DIPEA(269.19mg)加入DMF(2mL)中,反应16h,得到粗品化合物13a,直接用于下一步反应。Compound 2b (100mg), 3-amino-2,2-dimethyl-1-propanol (128.92mg), BOP (276.18mg) and DIPEA (269.19mg) were added to DMF (2mL) and reacted for 16h to obtain The crude compound 13a was directly used in the next reaction.
MS(ESI,m/z):327.1[M+H] +. MS(ESI,m/z): 327.1[M+H] + .
第二步:3-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)-2,2-二甲基-1-丙醇(化合物13)The second step: 3-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)-2,2-dimethyl-1-propanol (compound 13 )
向第一步中得到的化合物13a的反应液中,依次加入碳酸钾(126.52mg)、Pd(dppf)Cl 2·CH 2Cl 2(49.87mg)、1H-吡唑-5-硼酸频哪酯(118.60mg)和水(1mL),N 2保护下加热至100℃反应3h。通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物13(5mg)。 To the reaction solution of compound 13a obtained in the first step, potassium carbonate (126.52mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (49.87mg), 1H-pyrazole-5-boronic acid pinacolate were sequentially added (118.60mg) and water (1mL), heated to 100°C under the protection of N 2 to react for 3h. It was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 13 (5 mg).
MS(ESI,m/z):313.1[M+H] +. MS(ESI,m/z): 313.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.01(s,1H),8.03(d,J=8.5Hz,1H),7.81-7.78(m,2H),7.61(d,J=1.7Hz,1H),7.52(s,1H),6.82(d,J=2.2Hz,1H),6.13(s,2H),5.01-4.98(m,1H),3.11(d,J=6.2Hz,2H),2.51-2.50(m,2H),0.90(s,6H). 1 H NMR(DMSO-d 6 ,400MHz)δ13.01(s,1H), 8.03(d,J=8.5Hz,1H),7.81-7.78(m,2H), 7.61(d,J=1.7Hz, 1H), 7.52 (s, 1H), 6.82 (d, J = 2.2 Hz, 1H), 6.13 (s, 2H), 5.01-4.98 (m, 1H), 3.11 (d, J = 6.2 Hz, 2H), 2.51-2.50 (m, 2H), 0.90 (s, 6H).
实施例14:4-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)环己醇(化合物14)Example 14: 4-((2-Amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)cyclohexanol (Compound 14)
第一步:4-((2-氨基-7-溴喹唑啉-4-基)氨基)环己醇(化合物14a)The first step: 4-((2-amino-7-bromoquinazolin-4-yl)amino)cyclohexanol (compound 14a)
将化合物2b(100mg)、4-氨基环己醇(143.93mg)、BOP(276.18mg)和DIPEA(269.19mg)加入DMF(2mL)中,反应16h,得到粗品化合物14a,直接用于下一步反应。Compound 2b (100mg), 4-aminocyclohexanol (143.93mg), BOP (276.18mg) and DIPEA (269.19mg) were added to DMF (2mL) and reacted for 16h to obtain crude compound 14a, which was directly used in the next reaction .
MS(ESI,m/z):338.8[M+H] +. MS(ESI,m/z): 338.8[M+H] + .
第二步:4-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)环己醇(化合物14)The second step: 4-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)cyclohexanol (compound 14)
向第一步中得到的化合物14a的反应液中,依次加入碳酸钾(122.04mg)、Pd(dppf)Cl 2·CH 2Cl 2(48.11mg)、1H-吡唑-5-硼酸频哪酯(114.40mg)和水(1mL),N 2保护下加热至100℃反应3h。通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物14(5mg)。 To the reaction solution of compound 14a obtained in the first step, potassium carbonate (122.04mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (48.11mg), 1H-pyrazole-5-boronic acid pinacolate were sequentially added (114.40mg) and water (1mL), heated to 100°C under the protection of N 2 to react for 3h. It was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 14 (5 mg).
MS(ESI,m/z):325.1[M+H] +. MS(ESI,m/z): 325.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.02(s,1H),8.04(d,J=8.5Hz,1H),7.75(s,1H),7.59(d,J=1.7Hz,1H),7.47(d,J=8.5Hz,1H),7.40(d,J=8.0Hz,1H),6.80(d,J=2.2Hz,1H),6.05(s,2H),4.62(d,J=4.5Hz,1H),4.14-4.08(m,1H),3.47-3.37(m,1H),1.93-1.88(m,4H),1.49-1.42(m,2H),1.32-1.22(m,2H). 1 H NMR(DMSO-d 6 ,400MHz)δ13.02(s,1H), 8.04(d,J=8.5Hz,1H), 7.75(s,1H), 7.59(d,J=1.7Hz,1H) ,7.47(d,J=8.5Hz,1H),7.40(d,J=8.0Hz,1H), 6.80(d,J=2.2Hz,1H), 6.05(s,2H), 4.62(d,J= 4.5Hz,1H),4.14-4.08(m,1H),3.47-3.37(m,1H),1.93-1.88(m,4H),1.49-1.42(m,2H),1.32-1.22(m,2H) .
实施例15:(S)-7-(1H-吡唑-5-基)-N 4-((四氢呋喃-2-基)甲基)喹唑啉-2,4-二胺甲酸盐(化合物15s) Example 15: (S)-7-(1H-pyrazol-5-yl)-N 4 -((tetrahydrofuran-2-yl)methyl)quinazoline-2,4-diamine formate (compound 15s)
第一步:(S)-7-溴-N 4-((四氢呋喃-2-基)甲基)喹唑啉-2,4-二胺(化合物15a) The first step: (S)-7-bromo-N 4 -((tetrahydrofuran-2-yl)methyl)quinazolin-2,4-diamine (compound 15a)
室温下,将化合物2b(50mg)加入DMF(2mL)中,继续依次加入DIPEA(134.60mg)和BOP(110.54mg),搅拌5min。加入(S)-(+)-四氢糠胺(63.20mg),反应16h,得到粗品化合物15a,直接用于下一步反应。At room temperature, compound 2b (50 mg) was added to DMF (2 mL), DIPEA (134.60 mg) and BOP (110.54 mg) were added successively, and the mixture was stirred for 5 min. (S)-(+)-tetrahydrofurfurylamine (63.20mg) was added and reacted for 16h to obtain the crude compound 15a, which was directly used in the next reaction.
MS(ESI,m/z):323.0[M+H] +. MS(ESI,m/z):323.0[M+H] + .
第二步:(S)-7-(1H-吡唑-5-基)-N 4-((四氢呋喃-2-基)甲基)喹唑啉-2,4-二胺甲酸盐(化合物15s) The second step: (S)-7-(1H-pyrazol-5-yl)-N 4 -((tetrahydrofuran-2-yl)methyl)quinazoline-2,4-diamine formate (compound 15s)
向第一步中得到的化合物15a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(80.82mg)、Pd(dppf)Cl 2·CH 2Cl 2(17mg)、K 2CO 3(86.35mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,含化合物15的滤液通过Prep-HPLC分离纯化(洗脱条件3),冻干得到化合物15s(30mg)。 To the reaction solution of compound 15a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (80.82mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (17mg), K 2 CO were added sequentially. 3 (86.35mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate containing compound 15 was separated and purified by Prep-HPLC (elution condition 3), and lyophilized to obtain compound 15s (30 mg).
MS(ESI,m/z):311.0[M+H] +. MS(ESI,m/z):311.0[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.04(s,1H),8.39–8.16(m,2H),8.09(d,J=8.6Hz,1H),7.85–7.72(m,1H),7.66(s,1H),7.57(d,J=7.6Hz,1H),7.05–6.45(m,3H),4.20–4.08(m,1H),3.85–3.78(m,1H),3.69–3.62(m,1H),3.59–3.50(m,2H),2.00–1.77(m,3H),1.68–1.57(m,1H). 1 H NMR(DMSO-d 6 ,400MHz)δ13.04(s,1H), 8.39–8.16(m,2H), 8.09(d,J=8.6Hz,1H), 7.85–7.72(m,1H), 7.66(s,1H),7.57(d,J=7.6Hz,1H), 7.05–6.45(m,3H), 4.20–4.08(m,1H), 3.85–3.78(m,1H), 3.69–3.62( m, 1H), 3.59--3.50 (m, 2H), 2.00--1.77 (m, 3H), 1.68--1.57 (m, 1H).
实施例16:(R)-1-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)-2-丙醇(化合物16)Example 16: (R)-1-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)-2-propanol (Compound 16)
第一步:(R)-1-[(2-氨基-6-碘噻吩并[3,2-d]嘧啶-4-基)氨基]-2-丙醇(化合物16a)The first step: (R)-1-[(2-amino-6-iodothieno[3,2-d]pyrimidin-4-yl)amino]-2-propanol (Compound 16a)
将化合物10b(80mg)、(R)-(-)-1-氨基-2-丙醇(61.51mg)、PyBOP(184.66mg、DIPEA(176.39mg)和DMF(5.0mL)加至反应瓶内,于室温下反应16h。乙酸乙酯萃取3次,干燥浓缩,通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物16a(21.0mg)。Compound 10b (80mg), (R)-(-)-1-amino-2-propanol (61.51mg), PyBOP (184.66mg, DIPEA (176.39mg) and DMF (5.0mL) were added to the reaction flask, The reaction was carried out at room temperature for 16 hours. It was extracted with ethyl acetate for 3 times, dried and concentrated, separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 16a (21.0 mg).
MS(ESI,m/z):351[M+H] +. MS(ESI,m/z):351[M+H] + .
第二步:(R)-1-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)-2-丙醇(化合物16)The second step: (R)-1-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)-2-propanol (Compound 16)
将化合物16a(29mg)、1H-吡唑-5-硼酸频哪酯(20.89mg)、Pd(dppf)Cl 2·CH 2Cl 2(13.53mg)、K 3PO 4(35.16mg)、水(1.0mL)和1,4-二氧六环(2.0mL)加至反应瓶内,N 2置换10min,加热至100℃反应5h。乙酸乙酯萃取,干燥浓缩,通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物16(21.0mg)。 Compound 16a (29mg), 1H-pyrazole-5-boronic acid pinacolate (20.89mg), Pd(dppf)Cl 2 .CH 2 Cl 2 (13.53mg), K 3 PO 4 (35.16mg), water ( 1.0 mL) and 1,4-dioxane (2.0 mL) were added to the reaction flask, replaced with N 2 for 10 minutes, and heated to 100°C for 5 hours. It was extracted with ethyl acetate, dried and concentrated, separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 16 (21.0 mg).
MS(ESI,m/z):291[M+H] +. MS(ESI,m/z):291[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.07(s,1H),7.84(s,1H),7.25(s,1H),7.14(s,1H),6.78(s,1H),5.86(s,2H),4.82(s,1H),4.05–3.77(m,1H),3.41-3.35(m,J=5.3Hz,2H),1.09(d,J=6.2Hz,3H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.07 (s, 1H), 7.84 (s, 1H), 7.25 (s, 1H), 7.14 (s, 1H), 6.78 (s, 1H), 5.86 ( s, 2H), 4.82 (s, 1H), 4.05-3.77 (m, 1H), 3.41-3.35 (m, J = 5.3 Hz, 2H), 1.09 (d, J = 6.2 Hz, 3H).
实施例17:(R)-2-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)-1-丁醇(化合物17)Example 17: (R)-2-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)-1-butanol (Compound 17)
第一步:(R)-2-[(2-氨基-6-碘噻吩并[3,2-d]嘧啶-4-基)氨基]-1-丁醇(化合物17a)The first step: (R)-2-[(2-amino-6-iodothieno[3,2-d]pyrimidin-4-yl)amino]-1-butanol (Compound 17a)
将化合物10b(80mg)、(R)-2-氨基-1-丁醇(72.99mg)、PyBOP(184.66mg)、DIPEA(176.39mg)和DMF(3.0mL)加至反应瓶内,于室温下反应16h。乙酸乙酯萃取3次,干燥浓缩,通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物17a(29.0mg)。Compound 10b (80mg), (R)-2-amino-1-butanol (72.99mg), PyBOP (184.66mg), DIPEA (176.39mg) and DMF (3.0mL) were added to the reaction flask and left at room temperature Reaction for 16h. It was extracted three times with ethyl acetate, dried and concentrated, separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 17a (29.0 mg).
MS(ESI,m/z):365[M+H] +. MS(ESI,m/z):365[M+H] + .
第二步:(R)-2-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)-1-丁醇(化合物17)The second step: (R)-2-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)-1-butanol (Compound 17)
将化合物17a(29mg)、1H-吡唑-5-硼酸频哪酯(20.09mg)、Pd(dppf)Cl 2·CH 2Cl 2(13.01mg)、K 3PO 4(33.80mg)、水(0.5mL)和1,4-二氧六环(2.0mL)加至反应瓶内,N 2置换10min,加热至100℃反应5h。乙酸乙酯萃取,干燥浓缩,通过Prep-HPLC分离纯化(洗脱条件2),冻干得到化合物17(8.0mg)。 Compound 17a (29mg), 1H-pyrazole-5-boronic acid pinacolate (20.09mg), Pd(dppf)Cl 2 .CH 2 Cl 2 (13.01mg), K 3 PO 4 (33.80 mg), water ( 0.5 mL) and 1,4-dioxane (2.0 mL) were added to the reaction flask, replaced with N 2 for 10 minutes, and heated to 100°C for 5 hours. It was extracted with ethyl acetate, dried and concentrated, separated and purified by Prep-HPLC (elution condition 2), and lyophilized to obtain compound 17 (8.0 mg).
MS(ESI,m/z):305[M+H] +. MS(ESI,m/z):305[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.09(s,1H),7.86(s,1H),7.26(s,1H),6.82(d,J=17.3Hz,2H),5.83(s,2H),4.70(s,1H),4.19(s,1H),3.50(d,2H),1.75(d,J=21.2Hz,1H),1.52(d,J=7.0Hz,1H),0.90(t,J=6.5Hz,3H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.09 (s, 1H), 7.86 (s, 1H), 7.26 (s, 1H), 6.82 (d, J = 17.3 Hz, 2H), 5.83 (s, 2H), 4.70 (s, 1H), 4.19 (s, 1H), 3.50 (d, 2H), 1.75 (d, J = 21.2 Hz, 1H), 1.52 (d, J = 7.0 Hz, 1H), 0.90 ( t,J=6.5Hz,3H).
实施例18:(S)-2-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)-1-丙醇(化合物18)Example 18: (S)-2-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)-1-propanol (Compound 18)
第一步:(S)-2-[(2-氨基-6-碘噻吩并[3,2-d]嘧啶-4-基)氨基]-1-丙醇(化合物18a)The first step: (S)-2-[(2-amino-6-iodothieno[3,2-d]pyrimidin-4-yl)amino]-1-propanol (Compound 18a)
将化合物10b(80mg)、L-氨基丙醇(61.51mg)、PyBOP(184.66mg)、DIPEA(176.39mg)和DMF(3.0mL)加至反应瓶内,于室温下反应16h。乙酸乙酯萃取3次,干燥浓缩,通过Prep-HPLC分离纯化(洗脱条件2),冻干得到化合物18a(28.0mg)。Compound 10b (80 mg), L-aminopropanol (61.51 mg), PyBOP (184.66 mg), DIPEA (176.39 mg) and DMF (3.0 mL) were added to the reaction flask and reacted at room temperature for 16 hours. It was extracted three times with ethyl acetate, dried and concentrated, separated and purified by Prep-HPLC (elution condition 2), and lyophilized to obtain compound 18a (28.0 mg).
MS(ESI,m/z):351[M+H] +. MS(ESI,m/z):351[M+H] + .
第二步:(S)-2-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)-1-丙醇(化合物18)The second step: (S)-2-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)-1-propanol (Compound 18)
将化合物18a(28.00mg)、1H-吡唑-5-硼酸频哪酯(20.17mg)、Pd(dppf)Cl 2·CH 2Cl 2(13.06mg)、K 3PO 4(33.94mg)、水(0.5mL)和1,4-二氧六环(2.0mL)加至反应瓶内,N 2置换10min,加热至100℃反应5h。乙酸乙酯萃取,干燥浓缩,通过Prep-HPLC分离纯化(洗脱条件2),冻干得到化合物18(17.0mg)。 Compound 18a (28.00mg), 1H-pyrazole-5-boronic acid pinacolate (20.17mg), Pd(dppf)Cl 2 .CH 2 Cl 2 (13.06mg), K 3 PO 4 (33.94 mg), water (0.5mL) and 1,4-dioxane (2.0mL) were added to the reaction flask, replaced with N 2 for 10 minutes, and heated to 100°C for 5 hours. It was extracted with ethyl acetate, dried and concentrated, separated and purified by Prep-HPLC (elution condition 2), and lyophilized to obtain compound 18 (17.0 mg).
MS(ESI,m/z):291[M+H] +. MS(ESI,m/z):291[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.08(s,1H),7.85(s,1H),7.25(s,1H),7.01(s,1H),6.79(s,1H),5.95(s,2H),4.74(s,1H),4.35-4.29(m,1H),3.51–3.48(m,1H),3.39(d,1H),1.16(d,J=6.6Hz,3H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.08 (s, 1H), 7.85 (s, 1H), 7.25 (s, 1H), 7.01 (s, 1H), 6.79 (s, 1H), 5.95 ( s, 2H), 4.74 (s, 1H), 4.35-4.29 (m, 1H), 3.51-3.48 (m, 1H), 3.39 (d, 1H), 1.16 (d, J = 6.6 Hz, 3H).
实施例19:N 4-丙基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物19) Example 19: N 4 -Propyl-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4-diamine (Compound 19)
第一步:6-碘-N 4-丙基噻吩并[3,2-d]嘧啶-2,4-二胺(化合物19a) The first step: 6-iodo-N 4 -propylthieno[3,2-d]pyrimidine-2,4-diamine (compound 19a)
将化合物10b(80mg)、1-丙胺(48.40mg)、PyBOP(184.66mg)、DIPEA(176.39mg)和DMF(3.0mL)加至反应瓶内,于室温下反应16h。乙酸乙酯萃取3次,干燥浓缩,通过Prep-HPLC分离纯化(洗脱条件2),冻干得到化合物19a(45.0mg)。Compound 10b (80mg), 1-propylamine (48.40mg), PyBOP (184.66mg), DIPEA (176.39mg) and DMF (3.0mL) were added to the reaction flask and reacted at room temperature for 16h. It was extracted three times with ethyl acetate, dried and concentrated, separated and purified by Prep-HPLC (elution condition 2), and lyophilized to obtain compound 19a (45.0 mg).
MS(ESI,m/z):335[M+H] +. MS(ESI,m/z):335[M+H] + .
第二步:N 4-丙基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物19) The second step: N 4 -propyl-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4-diamine (Compound 19)
将化合物19a(45.00mg)、1H-吡唑-5-硼酸频哪酯(33.97mg)、Pd(dppf)Cl 2·CH 2Cl 2(21.99mg)、K 3PO 4(57.17mg)、水(0.5mL)和1,4-二氧六环(2.0mL)加至反应瓶内,N 2置换10min,加热至100℃反应5h。乙酸乙酯萃取,干燥浓缩,通过Prep-HPLC分离纯化(洗脱条件2),冻干得到化合物19(35.0mg)。 Compound 19a (45.00mg), 1H-pyrazole-5-boronic acid pinacolate (33.97mg), Pd(dppf)Cl 2 .CH 2 Cl 2 (21.99 mg), K 3 PO 4 (57.17 mg), water (0.5mL) and 1,4-dioxane (2.0mL) were added to the reaction flask, replaced with N 2 for 10 minutes, and heated to 100°C for 5 hours. It was extracted with ethyl acetate, dried and concentrated, separated and purified by Prep-HPLC (elution condition 2), and lyophilized to obtain compound 19 (35.0 mg).
MS(ESI,m/z):276[M+H] +. MS(ESI,m/z):276[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.06(s,1H),7.84(s,1H),7.24(s,2H),6.78(s,1H),5.85(s,2H),3.38(d,J=6.2Hz,2H),1.74–1.46(m,2H),0.91(t,J=7.4Hz,3H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.06 (s, 1H), 7.84 (s, 1H), 7.24 (s, 2H), 6.78 (s, 1H), 5.85 (s, 2H), 3.38 ( d,J=6.2Hz,2H),1.74-1.46(m,2H),0.91(t,J=7.4Hz,3H).
实施例20:2-(((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)甲基)环丙醇(化合物20)Example 20: 2-(((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)methyl)cyclopropanol (compound 20)
第一步:2-(((2-氨基-6-碘噻吩并[3,2-d]嘧啶-4-基)氨基)甲基)环丙醇(化合物20a)The first step: 2-(((2-amino-6-iodothieno[3,2-d]pyrimidin-4-yl)amino)methyl)cyclopropanol (Compound 20a)
将化合物10b(80mg)、2-氨甲基环丙醇(71.34mg)、PyBOP(184.66mg)、DIPEA(176.39mg)和DMF(3.0mL)加至反应瓶内,于室温下反应16h。乙酸乙酯萃取3次,干燥浓缩,再经快速柱层析分离纯化(洗脱剂体系A),得到化合物20a(3.1mg)。Compound 10b (80mg), 2-aminomethylcyclopropanol (71.34mg), PyBOP (184.66mg), DIPEA (176.39mg) and DMF (3.0mL) were added to the reaction flask and reacted at room temperature for 16h. It was extracted three times with ethyl acetate, dried and concentrated, and then separated and purified by flash column chromatography (eluent system A) to obtain compound 20a (3.1 mg).
MS(ESI,m/z):362[M+H] +. MS(ESI,m/z):362[M+H] + .
第二步:2-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)甲基)环丙醇(化合物20)The second step: 2-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)methyl)cyclopropanol (Compound 20 )
将化合物20a(50.00mg)、1H-吡唑-5-硼酸频哪酯(41.71mg)、Pd(dppf)Cl 2·CH 2Cl 2(21.01mg)、K 3PO 4(70.20mg)、水(0.8mL)和1,4-二氧六环(2.3mL)加至反应瓶内,N 2置换10min,加热至100℃反应5h。乙酸乙酯萃取,干燥浓缩,通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物20(13.0mg)。 Compound 20a (50.00mg), 1H-pyrazole-5-boronic acid pinacolate (41.71mg), Pd(dppf)Cl 2 .CH 2 Cl 2 (21.01mg), K 3 PO 4 (70.20mg), water (0.8mL) and 1,4-dioxane (2.3mL) were added to the reaction flask, replaced with N 2 for 10 minutes, and heated to 100°C for 5 hours. It was extracted with ethyl acetate, dried and concentrated, separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 20 (13.0 mg).
MS(ESI,m/z):303[M+H] +. MS(ESI,m/z):303[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.10(s,1H),7.84(s,1H),7.23(d,J=23.0Hz,2H),6.78(s,1H),5.55(s,1H),3.57(d,J=5.1Hz,2H),0.59(d,J=7.4Hz,4H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.10 (s, 1H), 7.84 (s, 1H), 7.23 (d, J = 23.0 Hz, 2H), 6.78 (s, 1H), 5.55 (s, 1H), 3.57 (d, J = 5.1 Hz, 2H), 0.59 (d, J = 7.4 Hz, 4H).
实施例21:N 4-(2-吗啉代乙基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物21) Example 21: N 4 -(2-morpholinoethyl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4-diamine (Compound 21)
第一步:6-碘-N 4-(2-吗啉代乙基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物21a) The first step: 6-iodo-N 4 -(2-morpholinoethyl)thieno[3,2-d]pyrimidine-2,4-diamine (compound 21a)
将化合物10b(50mg)、2-吗啉代乙胺(66.63mg)、PyBOP(115.41mg)、DIPEA(22.05mg)和DMF(3.0mL)加至反应瓶内,于室温下反应16h。乙酸乙酯萃取3次,干燥浓缩,通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物21a(50.0mg)。Compound 10b (50mg), 2-morpholinoethylamine (66.63mg), PyBOP (115.41mg), DIPEA (22.05mg) and DMF (3.0mL) were added to the reaction flask and reacted at room temperature for 16h. It was extracted 3 times with ethyl acetate, dried and concentrated, separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 21a (50.0 mg).
MS(ESI,m/z):406[M+H] +. MS(ESI,m/z):406[M+H] + .
第二步:N 4-(2-吗啉代乙基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物21) The second step: N 4 -(2-morpholinoethyl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4-diamine (Compound 21)
将化合物21a(80.33mg)、1H-吡唑-5-硼酸频哪酯(50.00mg)、Pd(dppf)Cl 2·CH 2Cl 2(32.37mg)、K 3PO 4(84.15mg)、水(0.8mL)和1,4-二氧六环(2.0mL)加至反应瓶内,室温下搅拌16h。N 2置换10min,加热至100℃反应5h。乙酸乙酯萃取,干燥浓缩,通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物21(17.0mg)。 Compound 21a (80.33mg), 1H-pyrazole-5-boronic acid pinaline (50.00mg), Pd(dppf)Cl 2 .CH 2 Cl 2 (32.37 mg), K 3 PO 4 (84.15 mg), water (0.8mL) and 1,4-dioxane (2.0mL) were added to the reaction flask and stirred at room temperature for 16h. Replace with N 2 for 10 min, and heat to 100° C. to react for 5 h. It was extracted with ethyl acetate, dried and concentrated, separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 21 (17.0 mg).
MS(ESI,m/z):346[M+H] +. MS(ESI,m/z):346[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.10(s,1H),7.86(s,1H),7.27(s,1H),7.18(s,1H),6.80(s,1H),5.90(s,2H),3.69–3.49(m,8H),2.45(s,2H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.10 (s, 1H), 7.86 (s, 1H), 7.27 (s, 1H), 7.18 (s, 1H), 6.80 (s, 1H), 5.90 ( s, 2H), 3.69--3.49 (m, 8H), 2.45 (s, 2H).
实施例22:4-((2R,6R)-2,6-二甲基吗啉代)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2-胺(化合物22)Example 22: 4-((2R,6R)-2,6-dimethylmorpholino)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2- Amine (Compound 22)
第一步:4-((2R,6R)-2,6-二甲基吗啉代)-6-碘噻吩并[3,2-d]嘧啶-2-胺(化合物22a)The first step: 4-((2R,6R)-2,6-dimethylmorpholino)-6-iodothieno[3,2-d]pyrimidin-2-amine (Compound 22a)
室温下,将化合物10b(100mg)加入DMF(4mL)中,继续依次加入DIPEA(220.48mg)和BOP(181.09mg),搅拌5min。继续加入2R,6R-二甲基吗啉(117.89mg),反应16h,得到粗品化合物22a,直接用于下一步反应。At room temperature, compound 10b (100 mg) was added to DMF (4 mL), DIPEA (220.48 mg) and BOP (181.09 mg) were added successively, and stirred for 5 min. Continue to add 2R,6R-dimethylmorpholine (117.89mg) and react for 16h to obtain the crude compound 22a, which was directly used in the next reaction.
MS(ESI,m/z):391.0[M+H] +. MS(ESI,m/z): 391.0[M+H] + .
第二步:4-((2R,6R)-2,6-二甲基吗啉代)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2-胺(化合物22)The second step: 4-((2R,6R)-2,6-dimethylmorpholino)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2- Amine (Compound 22)
向第一步中得到的化合物22a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(132.41mg)、Pd(dppf)Cl 2·CH 2Cl 2(28mg)、K 2CO 3(141.47mg)和水(1mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件2),冻干得到化合物22(11mg)。 To the reaction solution of compound 22a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (132.41mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (28mg), K 2 CO were sequentially added 3 (141.47mg) and water (1mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 2), and lyophilized to obtain compound 22 (11 mg).
MS(ESI,m/z):331.1[M+H] +. MS(ESI,m/z):331.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.12(s,1H),7.86(s,1H),7.32(s,1H),6.83(s,1H),6.02(s,2H),4.12–4.02(m,2H),3.98–3.89(m,2H),3.52–3.44(m,2H),1.18(d,J=6.4Hz,6H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.12 (s, 1H), 7.86 (s, 1H), 7.32 (s, 1H), 6.83 (s, 1H), 6.02 (s, 2H), 4.12-- 4.02(m,2H), 3.98–3.89(m,2H), 3.52–3.44(m,2H), 1.18(d,J=6.4Hz,6H).
实施例23:(1R,3R)-3-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)环戊醇(化合物23)Example 23: (1R,3R)-3-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)cyclopentanol (Compound 23)
第一步:(1R,3R)-3-((2-氨基-6-碘噻吩并[3,2-d]嘧啶-4-基)氨基)环戊醇(化合物23a)The first step: (1R,3R)-3-((2-amino-6-iodothieno[3,2-d]pyrimidin-4-yl)amino)cyclopentanol (compound 23a)
室温下,将化合物10b(50mg)加入DMF(2mL)中,继续依次加入DIPEA(110.24mg)和BOP(90.69mg),搅拌5min。继续加入(1R,3R)-3-氨基环戊醇盐酸盐(117.89mg),反应16h,得到粗品化合物23a,直接用于下一步反应。At room temperature, compound 10b (50 mg) was added to DMF (2 mL), DIPEA (110.24 mg) and BOP (90.69 mg) were added successively, and the mixture was stirred for 5 min. (1R,3R)-3-aminocyclopentanol hydrochloride (117.89mg) was added continuously, and reacted for 16h to obtain crude compound 23a, which was directly used in the next reaction.
MS(ESI,m/z):377.0[M+H] +. MS(ESI,m/z): 377.0[M+H] + .
第二步:(1R,3R)-3-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)环戊醇(化合物23)Step 2: (1R,3R)-3-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)cyclopentanol (Compound 23)
向第一步中得到的化合物23a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(66.20mg)、Pd(dppf)Cl 2·CH 2Cl 2(14mg)、K 2CO 3(70.73mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物23(12mg)。 To the reaction solution of compound 23a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (66.20mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (14mg), K 2 CO were sequentially added 3 (70.73mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 23 (12 mg).
MS(ESI,m/z):317.2[M+H] +. MS(ESI,m/z): 317.2[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.07(s,1H),7.85(s,1H),7.34–7.11(m,2H),6.78(s,1H),5.91(s,2H),4.80–4.65(m,1H),4.52(d,J=3.4Hz,1H),4.28–4.17(m,1H),2.18–2.05(m,1H),2.00–1.84(m,2H),1.78–1.66(m,1H),1.54–1.41(m,2H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.07 (s, 1H), 7.85 (s, 1H), 7.34-7.11 (m, 2H), 6.78 (s, 1H), 5.91 (s, 2H), 4.80–4.65(m,1H), 4.52(d,J=3.4Hz,1H), 4.28–4.17(m,1H), 2.18–2.05(m,1H), 2.00–1.84(m,2H), 1.78– 1.66 (m, 1H), 1.54-1.41 (m, 2H).
实施例24:(1S,3S)-3-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)环戊醇(化合物24)Example 24: (1S,3S)-3-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)cyclopentanol (Compound 24)
第一步:(1S,3S)-3-((2-氨基-6-碘噻吩并[3,2-d]嘧啶-4-基)氨基)环戊醇(化合物24a)The first step: (1S,3S)-3-((2-amino-6-iodothieno[3,2-d]pyrimidin-4-yl)amino)cyclopentanol (Compound 24a)
室温下,将化合物10b(50mg)加入DMF(2mL)中,继续依次加入DIPEA(110.24mg)和BOP(90.69mg),搅拌5min。继续加入(1S,3S)-3-氨基环戊醇盐酸盐(117.89mg),反应16h,得到粗品化合物24a,直接用于下一步反应。At room temperature, compound 10b (50 mg) was added to DMF (2 mL), DIPEA (110.24 mg) and BOP (90.69 mg) were added successively, and the mixture was stirred for 5 min. (1S,3S)-3-aminocyclopentanol hydrochloride (117.89mg) was continuously added and reacted for 16h to obtain crude compound 24a, which was directly used in the next reaction.
MS(ESI,m/z):377.0[M+H] +. MS(ESI,m/z): 377.0[M+H] + .
第二步:(1S,3S)-3-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)环戊醇(化合物24)The second step: (1S,3S)-3-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)cyclopentanol (Compound 24)
向第一步中得到的化合物24a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(66.20mg)、Pd(dppf)Cl 2·CH 2Cl 2(14mg)、K 2CO 3(70.73mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物24(12mg)。 To the reaction solution of compound 24a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (66.20mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (14mg), K 2 CO were sequentially added 3 (70.73mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 24 (12 mg).
MS(ESI,m/z):317.2[M+H] +. MS(ESI,m/z): 317.2[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.09(s,1H),7.85(s,1H),7.41–7.21(m,2H),6.79(s,1H),6.16–5.97(m,2H),4.80–4.68(m,1H),4.53(d,J=3.4Hz,1H),4.28–4.18(m,1H),2.19–2.05(m,1H),2.01–1.82(m,2H),1.81–1.66(m,1H),1.58–1.39(m,2H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.09 (s, 1H), 7.85 (s, 1H), 7.41-7.21 (m, 2H), 6.79 (s, 1H), 6.16-5.97 (m, 2H) ), 4.80–4.68(m,1H), 4.53(d,J=3.4Hz,1H), 4.28–4.18(m,1H), 2.19–2.05(m,1H), 2.01–1.82(m,2H), 1.81-1.66(m,1H), 1.58-1.39(m,2H).
实施例25:(R)-N 4-(1-甲氧基丙-2-基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺甲酸盐(化合物25s) Example 25: (R)-N 4 -(1-methoxyprop-2-yl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4 -Diamine formate (Compound 25s)
第一步:(R)-6-碘-N 4-(1-甲氧基丙-2-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物25a) The first step: (R)-6-iodo-N 4 -(1-methoxyprop-2-yl)thieno[3,2-d]pyrimidine-2,4-diamine (Compound 25a)
室温下,将化合物10b(50mg)加入DMF(2mL)中,继续依次加入DIPEA(110.24mg)和BOP(90.69mg),搅拌5min。继续加入(R)-1-甲氧基丙-2-胺(50.94mg),反应16h,得到粗品化合物25a,直接用于下一步反应。At room temperature, compound 10b (50 mg) was added to DMF (2 mL), DIPEA (110.24 mg) and BOP (90.69 mg) were added successively, and the mixture was stirred for 5 min. (R)-1-methoxypropan-2-amine (50.94mg) was continuously added and reacted for 16h to obtain the crude compound 25a, which was directly used in the next reaction.
MS(ESI,m/z):365.0[M+H] +. MS(ESI,m/z): 365.0[M+H] + .
第二步:(R)-N 4-(1-甲氧基丙-2-基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺甲酸盐(化合物25s) The second step: (R)-N 4 -(1-methoxyprop-2-yl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4 -Diamine formate (Compound 25s)
向第一步中得到的化合物25a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(66.20mg)、Pd(dppf)Cl 2·CH 2Cl 2(14mg)、K 2CO 3(70.73mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,含化合物25的滤液通过Prep-HPLC分离纯化(洗脱条件3),冻干得到化合物25s(14mg)。 To the reaction solution of compound 25a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (66.20mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (14mg), K 2 CO were sequentially added 3 (70.73mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate containing compound 25 was separated and purified by Prep-HPLC (elution condition 3), and lyophilized to obtain compound 25s (14 mg).
MS(ESI,m/z):305.1[M+H] +. MS(ESI,m/z): 305.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.08(s,1H),8.21(s,1H),7.83(s,1H),7.24(s,1H),7.06(d,J=8.3Hz,1H),6.77(s,1H),5.91(s,2H),4.62–4.42(m,1H),3.48–3.41(m,2H),3.27(s,3H),1.16(d,J=6.7Hz,3H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.08 (s, 1H), 8.21 (s, 1H), 7.83 (s, 1H), 7.24 (s, 1H), 7.06 (d, J = 8.3 Hz, 1H), 6.77(s, 1H), 5.91(s, 2H), 4.62–4.42(m, 1H), 3.48–3.41(m, 2H), 3.27(s, 3H), 1.16(d,J=6.7Hz ,3H).
实施例26:(R)-N 4-(2-甲氧基丙基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物26) Example 26: (R)-N 4 -(2-Methoxypropyl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4-diamine (Compound 26)
第一步:(R)-6-碘-N 4-(2-甲氧基丙基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物26a) The first step: (R)-6-iodo-N 4 -(2-methoxypropyl)thieno[3,2-d]pyrimidine-2,4-diamine (compound 26a)
室温下,将化合物10b(50mg)加入DMF(2mL)中,继续依次加入DIPEA(110.24mg)和BOP(90.69mg),搅拌5min。继续加入(R)-2-甲氧基丙-1-胺盐酸盐(64.28mg),反应16h,得到粗品化合物26a,直接用于下一步反应。At room temperature, compound 10b (50 mg) was added to DMF (2 mL), DIPEA (110.24 mg) and BOP (90.69 mg) were added successively, and the mixture was stirred for 5 min. (R)-2-Methoxyprop-1-amine hydrochloride (64.28mg) was continuously added and reacted for 16h to obtain crude compound 26a, which was directly used in the next reaction.
MS(ESI,m/z):365.0[M+H] +. MS(ESI,m/z): 365.0[M+H] + .
第二步:(R)-N 4-(2-甲氧基丙基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物26) The second step: (R)-N 4 -(2-methoxypropyl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4-diamine (Compound 26)
向第一步中得到的化合物26a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(66.20mg)、Pd(dppf)Cl 2·CH 2Cl 2(14mg)、K 2CO 3(70.73mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物26(15mg)。 To the reaction solution of compound 26a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (66.20mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (14mg), K 2 CO were sequentially added 3 (70.73mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 26 (15 mg).
MS(ESI,m/z):305.1[M+H] +. MS(ESI,m/z): 305.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.07(s,1H),7.85(s,1H),7.32–7.21(m,2H),6.78(s,1H),5.86(s,2H),3.62–3.54(m,1H),3.53–3.45(m,1H),3.40–3.36(m,1H),3.29(s,3H),1.10(d,J=6.1Hz,3H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.07 (s, 1H), 7.85 (s, 1H), 7.32-7.21 (m, 2H), 6.78 (s, 1H), 5.86 (s, 2H), 3.62–3.54(m,1H), 3.53–3.45(m,1H), 3.40–3.36(m,1H), 3.29(s,3H), 1.10(d,J=6.1Hz,3H).
实施例27:N 4-((1-甲基环丙基)甲基)-7-(1H-吡唑-5-基)喹唑啉-2,4-二胺(化合物27) Example 27: N 4 -((1-methylcyclopropyl)methyl)-7-(1H-pyrazol-5-yl)quinazoline-2,4-diamine (Compound 27)
第一步:7-溴-N 4-((1-甲基环丙基)甲基)喹唑啉-2,4-二胺(化合物27a) The first step: 7-bromo-N 4 -((1-methylcyclopropyl)methyl)quinazoline-2,4-diamine (compound 27a)
室温下,将化合物2b(70mg)、1-甲基环丙基乙胺(70.92mg)、BOP(89.28mg)和DIPEA(188.43mg)加入DMF(2mL)中,反应16h,得到粗品化合物27a,直接用于下一步反应。At room temperature, compound 2b (70mg), 1-methylcyclopropylethylamine (70.92mg), BOP (89.28mg) and DIPEA (188.43mg) were added to DMF (2mL) and reacted for 16h to obtain crude compound 27a. Used directly in the next reaction.
MS(ESI,m/z):309.0[M+H] +. MS(ESI,m/z): 309.0[M+H] + .
第二步:N 4-((1-甲基环丙基)甲基)-7-(1H-吡唑-5-基)喹唑啉-2,4-二胺(化合物27) The second step: N 4 -((1-methylcyclopropyl)methyl)-7-(1H-pyrazol-5-yl)quinazoline-2,4-diamine (Compound 27)
向第一步中得到的化合物27a的反应液中,依次加入碳酸钾(93.72mg)、Pd(dppf)Cl 2·CH 2Cl 2(27.71mg)、1H-吡唑-5-硼酸频哪酯(87.86mg)和水(1mL),N 2保护下加热至100℃反应3h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物27(7mg)。 To the reaction solution of compound 27a obtained in the first step, potassium carbonate (93.72 mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (27.71 mg), 1H-pyrazole-5-boronic acid pinacolate were added sequentially (87.86mg) and water (1mL), heated to 100°C under the protection of N 2 to react for 3h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 27 (7 mg).
MS(ESI,m/z):295.1[M+H] +. MS(ESI,m/z): 295.1[M+H] + .
1H NMR(DMSO-d 6,400MHz))δ13.10(s,1H),8.37(s,1H),8.20(d,J=8.5Hz,1H),7.83(s,1H),7.69-7.64(m,2H),6.83-6.78(m,3H),3.48(d,J=5.6Hz,2H),1.11(s,3H),0.59-0.58(m,2H),0.30-0.27(m,2H). 1 H NMR (DMSO-d 6 , 400MHz)) δ 13.10 (s, 1H), 8.37 (s, 1H), 8.20 (d, J = 8.5 Hz, 1H), 7.83 (s, 1H), 7.69-7.64 (m,2H),6.83-6.78(m,3H),3.48(d,J=5.6Hz,2H),1.11(s,3H),0.59-0.58(m,2H),0.30-0.27(m,2H ).
实施例28:N 4-(2-吗啉代乙基)-7-(1H-吡唑-5-基)喹唑啉-2,4-二胺(化合物28) Example 28: N 4 -(2-morpholinoethyl)-7-(1H-pyrazol-5-yl)quinazoline-2,4-diamine (Compound 28)
第一步:7-溴-N 4-(2-吗啉代乙基)喹唑啉-2,4-二胺(化合物28a) The first step: 7-bromo-N 4 -(2-morpholinoethyl)quinazoline-2,4-diamine (compound 28a)
室温下,将化合物2b(70mg)、2-吗啉代乙胺(75.92mg)、BOP(89.28mg)和DIPEA(188.43mg)加入DMF(2mL)中,反应16h,得到粗品化合物28a,直接用于下一步反应。At room temperature, compound 2b (70mg), 2-morpholinoethylamine (75.92mg), BOP (89.28mg) and DIPEA (188.43mg) were added to DMF (2mL) and reacted for 16h to obtain crude compound 28a, which was used directly In the next step.
MS(ESI,m/z):352.1[M+H] +. MS(ESI,m/z):352.1[M+H] + .
第二步:N 4-(2-吗啉代乙基)-7-(1H-吡唑-5-基)喹唑啉-2,4-二胺(化合物28) The second step: N 4 -(2-morpholinoethyl)-7-(1H-pyrazol-5-yl)quinazoline-2,4-diamine (Compound 28)
向第一步中得到的化合物28a的反应液中,依次加入碳酸钾(93.72mg)、Pd(dppf)Cl 2·CH 2Cl 2(27.71mg)、1H-吡唑-5-硼酸频哪酯(87.86mg)和水(1mL),N 2保护下加热至100℃反应3h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物28(12mg)。 To the reaction solution of compound 28a obtained in the first step, potassium carbonate (93.72mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (27.71mg), 1H-pyrazole-5-boronic acid pinacolate were added sequentially (87.86mg) and water (1mL), heated to 100°C under the protection of N 2 to react for 3h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 28 (12 mg).
MS(ESI,m/z):340.1[M+H] +. MS(ESI,m/z): 340.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.00(s,1H),7.97(d,J=8.5Hz,1H),7.78-7.76(m,2H),7.61(d,J=1.6Hz,1H),7.57–7.45(m,1H),6.81(d,J=2.1Hz,1H),6.11(s,2H),3.63-3.57(m,6H),2.58–2.55(m,2H),2.47–2.44(m,4H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.00 (s, 1H), 7.97 (d, J = 8.5 Hz, 1H), 7.78-7.76 (m, 2H), 7.61 (d, J = 1.6 Hz, 1H), 7.57-7.45 (m, 1H), 6.81 (d, J = 2.1Hz, 1H), 6.11 (s, 2H), 3.63-3.57 (m, 6H), 2.58-2.55 (m, 2H), 2.47 –2.44(m,4H).
实施例29:4-((2R,6R)-2,6-二甲基吗啉代)-7-(1H-吡唑-5-基)喹唑啉-2-胺(化合物29)Example 29: 4-((2R,6R)-2,6-Dimethylmorpholino)-7-(1H-pyrazol-5-yl)quinazolin-2-amine (Compound 29)
第一步:7-溴-4-((2R,6R)-2,6-二甲基吗啉代)喹唑啉-2-胺(化合物29a)The first step: 7-bromo-4-((2R,6R)-2,6-dimethylmorpholino)quinazolin-2-amine (compound 29a)
室温下,将化合物2b(70mg)、(2R,6R)-2,6-二甲基吗啉(67.17mg)、BOP(89.28mg)和DIPEA(188.43mg)加入DMF(2mL)中,反应16h,得到粗品化合物29a,直接用于下一步反应。At room temperature, compound 2b (70mg), (2R,6R)-2,6-dimethylmorpholine (67.17mg), BOP (89.28mg) and DIPEA (188.43mg) were added to DMF (2mL) and reacted for 16h , The crude compound 29a was obtained, which was directly used in the next reaction.
MS(ESI,m/z):338.1[M+H] +. MS(ESI,m/z): 338.1[M+H] + .
第二步:4-((2R,6R)-2,6-二甲基吗啉代)-7-(1H-吡唑-5-基)喹唑啉-2-胺(化合物29)The second step: 4-((2R,6R)-2,6-dimethylmorpholino)-7-(1H-pyrazol-5-yl)quinazolin-2-amine (Compound 29)
向第一步中得到的化合物29a的反应液中,依次加入碳酸钾(93.72mg)、Pd(dppf)Cl 2·CH 2Cl 2(27.71mg)、1H-吡唑-5-硼酸频哪酯(87.86mg)和水(1mL),N 2保护下加热至100℃反应3h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物29(5mg)。 To the reaction solution of compound 29a obtained in the first step, potassium carbonate (93.72 mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (27.71 mg), 1H-pyrazole-5-boronic acid pinacolate were sequentially added (87.86mg) and water (1mL), heated to 100°C under the protection of N 2 to react for 3h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 29 (5 mg).
MS(ESI,m/z):325.1[M+H] +. MS(ESI,m/z): 325.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.03(s,1H),7.77(d,J=8.7Hz,2H),7.71(d,J=1.7Hz,1H),7.56(s,1H),6.82(d,J=2.3Hz,1H),6.34(s,2H),4.17-4.13(m,2H),3.63-3.59(m,2H),3.35-3.30(m,2H),1.24(d,J=6.4Hz,6H). 1 H NMR(DMSO-d 6 ,400MHz)δ13.03(s,1H),7.77(d,J=8.7Hz,2H),7.71(d,J=1.7Hz,1H),7.56(s,1H) ,6.82(d,J=2.3Hz,1H),6.34(s,2H),4.17-4.13(m,2H),3.63-3.59(m,2H),3.35-3.30(m,2H),1.24(d ,J=6.4Hz,6H).
实施例30:N 4-(环丁基甲基)-7-(1H-吡唑-5-基)喹唑啉-2,4-二胺(化合物30) Example 30: N 4 -(Cyclobutylmethyl)-7-(1H-pyrazol-5-yl)quinazoline-2,4-diamine (Compound 30)
第一步:7-溴-N 4-(环丁基甲基)喹唑啉-2,4-二胺(化合物30a) The first step: 7-bromo-N 4 -(cyclobutylmethyl)quinazoline-2,4-diamine (compound 30a)
室温下,将化合物2b(70mg)、环丁基甲胺(49.66mg)、BOP(89.28mg)和DIPEA(188.43mg)加入DMF(2mL)中,反应16h,得到粗品化合物30a,直接用于下一步反应。At room temperature, compound 2b (70mg), cyclobutylmethylamine (49.66mg), BOP (89.28mg) and DIPEA (188.43mg) were added to DMF (2mL) and reacted for 16h to obtain crude compound 30a, which was directly used in the next reaction .
MS(ESI,m/z):309.0[M+H] +. MS(ESI,m/z): 309.0[M+H] + .
第二步:N 4-(环丁基甲基)-7-(1H-吡唑-5-基)喹唑啉-2,4-二胺(化合物30) The second step: N 4 -(cyclobutylmethyl)-7-(1H-pyrazol-5-yl)quinazoline-2,4-diamine (compound 30)
向第一步中得到的化合物30a的反应液中,依次加入碳酸钾(93.72mg)、Pd(dppf)Cl 2·CH 2Cl 2(27.71mg)、1H-吡唑-5-硼酸频哪酯(87.86mg)和水(1mL),N 2保护下加热至100℃反应3h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件2),冻干得到化合物30(7mg)。 To the reaction solution of compound 30a obtained in the first step, potassium carbonate (93.72mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (27.71mg), 1H-pyrazole-5-boronic acid pinacolate were sequentially added (87.86mg) and water (1mL), heated to 100°C under the protection of N 2 to react for 3h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 2), and lyophilized to obtain compound 30 (7 mg).
MS(ESI,m/z):295.1[M+H] +. MS(ESI,m/z): 295.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ12.98(s,1H),8.02(d,J=8.5Hz,1H),7.86-7.83(m,1H),7.60(d,J=1.7Hz,1H),7.50(s,1H),6.80(d,J=2.2Hz,1H),6.13(s,2H),3.54-3.50(m,2H),2.72-2.65(m,1H),2.08-1.99(m,2H),1.91-1.83(m,2H),1.79-1.73(m 2H). 1 H NMR(DMSO-d 6 ,400MHz)δ12.98(s,1H), 8.02(d,J=8.5Hz,1H),7.86-7.83(m,1H), 7.60(d,J=1.7Hz, 1H), 7.50 (s, 1H), 6.80 (d, J = 2.2 Hz, 1H), 6.13 (s, 2H), 3.54-3.50 (m, 2H), 2.72-2.65 (m, 1H), 2.08-1.99 (m, 2H), 1.91-1.83 (m, 2H), 1.79-1.73 (m 2H).
实施例31:(R)-N 4-(1-甲氧基丙-2-基)-7-(1H-吡唑-5-基)喹唑啉-2,4-二胺甲酸盐(化合物31s) Example 31: (R)-N 4 -(1-methoxyprop-2-yl)-7-(1H-pyrazol-5-yl)quinazoline-2,4-diamine formate ( Compound 31s)
第一步:(R)-7-溴-N 4-(1-甲氧基丙-2-基)喹唑啉-2,4-二胺(化合物31a) The first step: (R)-7-bromo-N 4 -(1-methoxyprop-2-yl)quinazoline-2,4-diamine (compound 31a)
室温下,将化合物2b(70mg)、(R)-1-甲氧基丙-2-胺盐酸盐(73.25mg)、BOP(89.28mg)和DIPEA(188.43mg)加入DMF(2mL)中,反应16h,得到粗品化合物31a,直接用于下一步反应。At room temperature, compound 2b (70 mg), (R)-1-methoxypropan-2-amine hydrochloride (73.25 mg), BOP (89.28 mg) and DIPEA (188.43 mg) were added to DMF (2 mL), After 16 hours of reaction, the crude compound 31a was obtained, which was directly used in the next reaction.
MS(ESI,m/z):313.1[M+H] +. MS(ESI,m/z): 313.1[M+H] + .
第二步:(R)-N 4-(1-甲氧基丙-2-基)-7-(1H-吡唑-5-基)喹唑啉-2,4-二胺甲酸盐(化合物31s) The second step: (R)-N 4 -(1-methoxyprop-2-yl)-7-(1H-pyrazol-5-yl)quinazoline-2,4-diamine formate ( Compound 31s)
向第一步中得到的化合物31a的反应液中,依次加入碳酸钾(93.72mg)、Pd(dppf)Cl 2·CH 2Cl 2(27.71mg)、1H-吡唑-5-硼酸频哪酯(87.86mg)和水(1mL),N 2保护下加热至100℃反应3h。将反应液硅藻土过滤,含化合物31的滤液通过Prep-HPLC分离纯化(洗脱条件3),冻干得到化合物31s(2mg)。 To the reaction solution of compound 31a obtained in the first step, potassium carbonate (93.72mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (27.71mg), 1H-pyrazole-5-boronic acid pinacolate were sequentially added (87.86mg) and water (1mL), heated to 100°C under the protection of N 2 to react for 3h. The reaction solution was filtered through Celite, and the filtrate containing compound 31 was separated and purified by Prep-HPLC (elution condition 3), and lyophilized to obtain compound 31s (2 mg).
MS(ESI,m/z):299.1[M+H] +. MS(ESI,m/z): 299.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ8.32(s,1H),8.11(d,J=8.6Hz,1H),7.78(d,J=6.7Hz,2H),7.64(d,J=1.7Hz,1H),7.55(d,J=8.4Hz,1H),6.82(d,J=2.2Hz,1H),6.70(s,2H),4.66-4.59(m,1H),3.52-3.46(m,1H),3.38-3.34(m,1H),3.29(s,3H),1.22(d,J=6.7Hz,3H). 1 H NMR(DMSO-d 6 ,400MHz)δ8.32(s,1H), 8.11(d,J=8.6Hz,1H), 7.78(d,J=6.7Hz,2H), 7.64(d,J= 1.7Hz, 1H), 7.55 (d, J = 8.4 Hz, 1H), 6.82 (d, J = 2.2 Hz, 1H), 6.70 (s, 2H), 4.66-4.59 (m, 1H), 3.52-3.46 ( m, 1H), 3.38-3.34 (m, 1H), 3.29 (s, 3H), 1.22 (d, J = 6.7 Hz, 3H).
实施例32:N 4-(戊-3-基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物32) Example 32: N 4 -(pent-3-yl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4-diamine (Compound 32)
第一步:6-碘-N 4-(戊-3-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物32a) The first step: 6-iodo-N 4 -(pent-3-yl)thieno[3,2-d]pyrimidine-2,4-diamine (compound 32a)
室温下,将化合物10b(50mg)加入DMF(2mL)中,继续依次加入DIPEA(110.24mg)和BOP(90.69mg),搅拌5min。继续加入3-戊胺(44.61mg),反应16h,得到粗品化合物32a,直接用于下一步反应。At room temperature, compound 10b (50 mg) was added to DMF (2 mL), DIPEA (110.24 mg) and BOP (90.69 mg) were added successively, and the mixture was stirred for 5 min. Continue to add 3-pentylamine (44.61mg) and react for 16h to obtain the crude compound 32a, which was directly used in the next reaction.
MS(ESI,m/z):363.0[M+H] +. MS(ESI,m/z): 363.0[M+H] + .
第二步:N 4-(戊-3-基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物32) The second step: N 4 -(pent-3-yl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4-diamine (Compound 32)
向第一步中得到的化合物32a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(66.20mg)、Pd(dppf)Cl 2·CH 2Cl 2(14mg)、K 2CO 3(70.73mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件2),冻干得到化合物32(10mg)。 To the reaction solution of compound 32a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (66.20mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (14mg), K 2 CO were sequentially added 3 (70.73mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 2), and lyophilized to obtain compound 32 (10 mg).
MS(ESI,m/z):303.2[M+H] +. MS(ESI,m/z): 303.2[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.06(s,1H),7.84(s,1H),7.23(s,1H),6.88(d,J=8.6Hz,1H),6.77(s,1H),5.77(s,2H),4.19–4.04(m,1H),1.63–1.45(m,4H),0.87(t,J=7.4Hz,6H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.06 (s, 1H), 7.84 (s, 1H), 7.23 (s, 1H), 6.88 (d, J = 8.6 Hz, 1H), 6.77 (s, 1H), 5.77(s, 2H), 4.19–4.04(m,1H), 1.63–1.45(m,4H), 0.87(t,J=7.4Hz,6H).
实施例33:N 4-(环丙基甲基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物33) Example 33: N 4 -(Cyclopropylmethyl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4-diamine (Compound 33)
第一步:N 4-(环丙基甲基)-6-碘噻吩并[3,2-d]嘧啶-2,4-二胺(化合物33a) The first step: N 4 -(cyclopropylmethyl)-6-iodothieno[3,2-d]pyrimidine-2,4-diamine (compound 33a)
室温下,将化合物10b(50mg)加入DMF(2mL)中,继续依次加入DIPEA(110.24mg)和BOP(90.69mg),搅拌5min。继续加入环丙基甲胺(36.40mg),反应16h,得到粗品化合物33a,直接用于下一步反应。At room temperature, compound 10b (50 mg) was added to DMF (2 mL), DIPEA (110.24 mg) and BOP (90.69 mg) were added successively, and the mixture was stirred for 5 min. Cyclopropylmethylamine (36.40mg) was continuously added and reacted for 16h to obtain crude compound 33a, which was directly used in the next reaction.
MS(ESI,m/z):347.0[M+H] +. MS(ESI,m/z): 347.0[M+H] + .
第二步:N 4-(环丙基甲基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物33) Step 2: N 4 -(Cyclopropylmethyl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4-diamine (Compound 33)
向第一步中得到的化合物33a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(66.20mg)、Pd(dppf)Cl 2·CH 2Cl 2(14mg)、K 2CO 3(70.73mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件2),冻干得到化合物33(8mg)。 To the reaction solution of compound 33a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (66.20mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (14mg), K 2 CO were sequentially added 3 (70.73mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 2), and lyophilized to obtain compound 33 (8 mg).
MS(ESI,m/z):287.1[M+H] +. MS(ESI,m/z):287.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.08(s,1H),7.85(s,1H),7.49(s,1H),7.25(s,1H),6.79(s,1H),5.97(s,2H),3.31–3.27(m,2H),1.20–1.05(m,1H),0.48–0.41(m,2H),0.28–0.23(m,2H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.08 (s, 1H), 7.85 (s, 1H), 7.49 (s, 1H), 7.25 (s, 1H), 6.79 (s, 1H), 5.97 ( s, 2H), 3.31--3.27 (m, 2H), 1.20-1.05 (m, 1H), 0.48-0.41 (m, 2H), 0.28-0.23 (m, 2H).
实施例34:(1S,2R)-2-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)环戊醇(化合物34)Example 34: (1S,2R)-2-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)cyclopentanol (Compound 34)
第一步:(1S,2R)-2-((2-氨基-6-碘噻吩并[3,2-d]嘧啶-4-基)氨基)环戊醇(化合物34a)The first step: (1S,2R)-2-((2-amino-6-iodothieno[3,2-d]pyrimidin-4-yl)amino)cyclopentanol (Compound 34a)
室温下,将化合物10b(50mg)加入DMF(2mL)中,继续依次加入DIPEA(110.24mg)和BOP(90.69mg),搅拌5min。继续加入(1S,2R)-2-氨基环戊醇(51.77mg),反应16h,得到粗品化合物34a,直接用于下一步反应。At room temperature, compound 10b (50 mg) was added to DMF (2 mL), DIPEA (110.24 mg) and BOP (90.69 mg) were added successively, and the mixture was stirred for 5 min. (1S,2R)-2-aminocyclopentanol (51.77mg) was continuously added and reacted for 16h to obtain crude compound 34a, which was directly used in the next reaction.
MS(ESI,m/z):377.0[M+H] +. MS(ESI,m/z): 377.0[M+H] + .
第二步:(1S,2R)-2-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)环戊醇(化合物34)The second step: (1S,2R)-2-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)cyclopentanol (Compound 34)
向第一步中得到的化合物34a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(66.20mg)、Pd(dppf)Cl 2·CH 2Cl 2(14mg)、K 2CO 3(70.73mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物34(8mg)。 To the reaction solution of compound 34a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (66.20mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (14mg), K 2 CO were sequentially added 3 (70.73mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 34 (8 mg).
MS(ESI,m/z):317.1[M+H] +. MS(ESI,m/z): 317.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.07(s,1H),7.85(s,1H),7.26(s,1H),6.79(s,1H),6.60(s,1H),5.92(s,2H),4.77(d,J=3.9Hz,1H),4.32–4.20(m,1H),4.18–4.08(m,1H),1.95–1.86(m,1H),1.83–1.69(m,3H),1.66–1.57(m,1H),1.55–1.45(m,1H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.07 (s, 1H), 7.85 (s, 1H), 7.26 (s, 1H), 6.79 (s, 1H), 6.60 (s, 1H), 5.92 ( s,2H),4.77(d,J=3.9Hz,1H), 4.32–4.20(m,1H), 4.18–4.08(m,1H),1.95–1.86(m,1H),1.83–1.69(m, 3H), 1.66-1.57 (m, 1H), 1.55-1.45 (m, 1H).
实施例35:6-(1H-吡唑-5-基)-N 4-((四氢-2H-吡喃-4-基)甲基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物35) Example 35: 6-(1H-pyrazol-5-yl)-N 4 -((tetrahydro-2H-pyran-4-yl)methyl)thieno[3,2-d]pyrimidine-2, 4-Diamine (Compound 35)
第一步:6-碘-N 4-((四氢-2H-吡喃-4-基)甲基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物35a) The first step: 6-iodo-N 4 -((tetrahydro-2H-pyran-4-yl)methyl)thieno[3,2-d]pyrimidine-2,4-diamine (compound 35a)
室温下,将化合物10b(50mg)加入DMF(2mL)中,继续依次加入DIPEA(110.24mg)和BOP(90.69mg),搅拌5min。继续加入4-氨甲基四氢吡喃(58.95mg),反应16h,得到粗品化合物35a,直接用于下一步反应。At room temperature, compound 10b (50 mg) was added to DMF (2 mL), DIPEA (110.24 mg) and BOP (90.69 mg) were added successively, and the mixture was stirred for 5 min. Continue to add 4-aminomethyltetrahydropyran (58.95mg) and react for 16h to obtain the crude compound 35a, which was directly used in the next reaction.
MS(ESI,m/z):391.0[M+H] +. MS(ESI,m/z): 391.0[M+H] + .
第二步:6-(1H-吡唑-5-基)-N 4-((四氢-2H-吡喃-4-基)甲基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物35) The second step: 6-(1H-pyrazol-5-yl)-N 4 -((tetrahydro-2H-pyran-4-yl)methyl)thieno[3,2-d]pyrimidine-2, 4-Diamine (Compound 35)
向第一步中得到的化合物35a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(66.20mg)、Pd(dppf)Cl 2·CH 2Cl 2(14mg)、K 2CO 3(70.73mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物35(13mg)。 To the reaction solution of compound 35a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (66.20mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (14mg), K 2 CO were sequentially added 3 (70.73mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 35 (13 mg).
MS(ESI,m/z):331.1[M+H] +. MS(ESI,m/z):331.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.06(s,1H),7.84(s,1H),7.35–7.18(m,2H),6.78(s,1H),5.85(s,2H),3.85(dd,J=11.3,2.5Hz,2H),3.32–3.24(m,4H),1.97–1.82(m,1H),1.62(d,J=11.3Hz,2H),1.27– 1.14(m,2H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.06 (s, 1H), 7.84 (s, 1H), 7.35-7.18 (m, 2H), 6.78 (s, 1H), 5.85 (s, 2H), 3.85(dd,J=11.3,2.5Hz,2H),3.32–3.24(m,4H),1.97–1.82(m,1H),1.62(d,J=11.3Hz,2H),1.27– 1.14(m, 2H).
实施例36:N 4-(2-甲氧基乙基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺甲酸盐(化合物36s) Example 36: N 4 -(2-Methoxyethyl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4-diamine formate ( Compound 36s)
第一步:6-碘-N 4-(2-甲氧基乙基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物36a) The first step: 6-iodo-N 4 -(2-methoxyethyl)thieno[3,2-d]pyrimidine-2,4-diamine (compound 36a)
将化合物10b(40mg)、2-甲氧基乙胺(30.75mg)、BOP(27.86mg)、DIPEA(17.64mg)和DMF(2.0mL)加至反应瓶内,于室温下反应16h,得到粗品化合物36a,直接用于下一步反应。Compound 10b (40mg), 2-methoxyethylamine (30.75mg), BOP (27.86mg), DIPEA (17.64mg) and DMF (2.0mL) were added to the reaction flask and reacted at room temperature for 16h to obtain the crude product Compound 36a was directly used in the next reaction.
MS(ESI,m/z):350[M+H] +. MS(ESI,m/z): 350[M+H] + .
第二步:N 4-(2-甲氧基乙基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺甲酸盐(化合物36s) The second step: N 4 -(2-methoxyethyl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4-diamine formate ( Compound 36s)
向第一步中得到的化合物36a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(67.71mg)、Pd(dppf)Cl 2·CH 2Cl 2(43.84mg)、K 3PO 4(113.96mg)和水(0.5mL),N 2保护下于100℃下搅拌5h。将反应液硅藻土过滤,含化合物36的滤液通过Prep-HPLC分离纯化(洗脱条件6),冻干得到化合物36s(17.0mg)。 To the reaction solution of compound 36a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (67.71 mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (43.84 mg), K 3 were sequentially added PO 4 (113.96 mg) and water (0.5 mL) were stirred at 100°C for 5 h under the protection of N 2. The reaction solution was filtered through Celite, and the filtrate containing compound 36 was separated and purified by Prep-HPLC (elution condition 6), and lyophilized to obtain compound 36s (17.0 mg).
MS(ESI,m/z):337[M+H] +. MS(ESI,m/z):337[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.10(s,1H),8.16(s,1H),7.84(s,1H),7.47(t,J=5.3Hz,1H),6.79(d,J=1.9Hz,1H),6.10(s,2H),3.59(q,J=5.6Hz,2H),3.51(dd,J=6.2,4.8Hz,2H),3.28(s,3H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.10 (s, 1H), 8.16 (s, 1H), 7.84 (s, 1H), 7.47 (t, J = 5.3 Hz, 1H), 6.79 (d, J = 1.9Hz, 1H), 6.10 (s, 2H), 3.59 (q, J = 5.6 Hz, 2H), 3.51 (dd, J = 6.2, 4.8 Hz, 2H), 3.28 (s, 3H).
实施例37:2-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)(甲基)氨基)乙醇(化合物37)Example 37: 2-((2-Amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)(methyl)amino)ethanol (Compound 37)
第一步:2-((2-氨基-6-碘噻吩并[3,2-d]嘧啶-4-基)(甲基)氨基)乙醇(化合物37a)The first step: 2-((2-amino-6-iodothieno[3,2-d]pyrimidin-4-yl)(methyl)amino)ethanol (compound 37a)
室温下,将化合物10b(50mg)加入DMF(2mL)中,继续依次加入DIPEA(110.24mg)和BOP(90.69mg),搅拌5min。继续加入N-甲基-2-羟基乙胺(38.44mg),反应16h,得到粗品化合物37a,直接用于下一步反应。At room temperature, compound 10b (50 mg) was added to DMF (2 mL), DIPEA (110.24 mg) and BOP (90.69 mg) were added successively, and the mixture was stirred for 5 min. Continue to add N-methyl-2-hydroxyethylamine (38.44mg) and react for 16h to obtain the crude compound 37a, which was directly used in the next reaction.
MS(ESI,m/z):351.0[M+H] +. MS(ESI,m/z):351.0[M+H] + .
第二步:2-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)(甲基)氨基)乙醇(化合物37)Step 2: 2-((2-Amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)(methyl)amino)ethanol (Compound 37)
向第一步中得到的化合物37a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(66.20mg)、Pd(dppf)Cl 2·CH 2Cl 2(14mg)、K 2CO 3(70.73mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物37(22mg)。 To the reaction solution of compound 37a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (66.20mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (14mg), K 2 CO were sequentially added 3 (70.73mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 37 (22 mg).
MS(ESI,m/z):291.1[M+H] +. MS(ESI,m/z): 291.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.10(s,1H),7.83(s,1H),7.27(s,1H),6.79(d,J=1.9Hz,1H),5.84(s,2H),4.82(t,J=5.2Hz,1H),3.80–3.72(m,2H),3.70–3.62(m,2H),3.34(s,3H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.10 (s, 1H), 7.83 (s, 1H), 7.27 (s, 1H), 6.79 (d, J = 1.9 Hz, 1H), 5.84 (s, 2H), 4.82(t,J=5.2Hz,1H), 3.80–3.72(m,2H), 3.70–3.62(m,2H), 3.34(s,3H).
实施例38:4-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)环己醇(化合物38)Example 38: 4-((2-Amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)cyclohexanol (Compound 38)
第一步:4-((2-氨基-6-碘噻吩并[3,2-d]嘧啶-4-基)氨基)环己醇(化合物38a)The first step: 4-((2-amino-6-iodothieno[3,2-d]pyrimidin-4-yl)amino)cyclohexanol (compound 38a)
室温下,将化合物10b(50mg)加入DMF(2mL)中,继续依次加入DIPEA(110.24mg)和BOP(90.69mg),搅拌5min。继续加入4-氨基环己醇(58.95mg),反应16h,得到粗品化合物38a,直接用于下一步反应。At room temperature, compound 10b (50 mg) was added to DMF (2 mL), DIPEA (110.24 mg) and BOP (90.69 mg) were added successively, and the mixture was stirred for 5 min. Continue to add 4-aminocyclohexanol (58.95mg) and react for 16h to obtain crude compound 38a, which was directly used in the next reaction.
MS(ESI,m/z):391.0[M+H] +. MS(ESI,m/z): 391.0[M+H] + .
第二步:4-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)环己醇(化合物38)Step 2: 4-((2-Amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)cyclohexanol (Compound 38)
向第一步中得到的化合物38a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(66.20mg)、Pd(dppf)Cl 2·CH 2Cl 2(14mg)、K 2CO 3(70.73mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物38(16mg)。 To the reaction solution of compound 38a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (66.20mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (14mg), K 2 CO were sequentially added 3 (70.73mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 38 (16 mg).
MS(ESI,m/z):331.1[M+H] +. MS(ESI,m/z):331.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.07(s,1H),7.82(s,1H),7.22(s,1H),6.95(d,J=8.0Hz,1H),6.75(s,1H),5.81(s,2H),4.56(d,J=4.5Hz,1H),4.09–3.94(m,1H),3.45–3.36(m,1H),1.93–1.80(m,4H),1.46–1.32(m,2H),1.30–1.16(m,2H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.07 (s, 1H), 7.82 (s, 1H), 7.22 (s, 1H), 6.95 (d, J = 8.0 Hz, 1H), 6.75 (s, 1H),5.81(s,2H),4.56(d,J=4.5Hz,1H),4.09–3.94(m,1H),3.45–3.36(m,1H),1.93-1.80(m,4H),1.46 –1.32(m,2H),1.30–1.16(m,2H).
实施例39:1-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)甲基)环丙烷甲腈(化合物39)Example 39: 1-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)methyl)cyclopropanecarbonitrile (compound 39)
第一步:1-((2-氨基-6-碘噻吩并[3,2-d]嘧啶-4-基)氨基)甲基)环丙烷甲腈(化合物39a)The first step: 1-((2-amino-6-iodothieno[3,2-d]pyrimidin-4-yl)amino)methyl)cyclopropanecarbonitrile (compound 39a)
室温下,将化合物10b(50mg)加入DMF(2mL)中,继续依次加入DIPEA(110.24mg)和BOP(90.69mg),搅拌5min。继续加入1-(氨甲基)环丙烷甲腈盐酸盐(67.86mg),反应16h,得到粗品化合物39a,直接用于下一步反应。At room temperature, compound 10b (50 mg) was added to DMF (2 mL), DIPEA (110.24 mg) and BOP (90.69 mg) were added successively, and the mixture was stirred for 5 min. Continue to add 1-(aminomethyl)cyclopropanecarbonitrile hydrochloride (67.86mg) and react for 16h to obtain crude compound 39a, which was directly used in the next reaction.
MS(ESI,m/z):372.0[M+H] +. MS(ESI,m/z): 372.0[M+H] + .
第二步:1-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)甲基)环丙烷甲腈(化合物39)The second step: 1-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)methyl)cyclopropanecarbonitrile (compound 39)
向第一步中得到的化合物39a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(66.20mg)、Pd(dppf)Cl 2·CH 2Cl 2(14mg)、K 2CO 3(70.73mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物39(9mg)。 To the reaction solution of compound 39a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (66.20mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (14mg), K 2 CO were sequentially added 3 (70.73mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 39 (9 mg).
MS(ESI,m/z):312.1[M+H] +. MS(ESI,m/z):312.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.08(s,1H),7.85(s,1H),7.73(s,1H),7.28(s,1H),6.81(s,1H),5.99(s,2H),3.60(d,J=5.9Hz,2H),1.21(s,4H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.08 (s, 1H), 7.85 (s, 1H), 7.73 (s, 1H), 7.28 (s, 1H), 6.81 (s, 1H), 5.99 ( s, 2H), 3.60 (d, J = 5.9 Hz, 2H), 1.21 (s, 4H).
实施例40:N 4-((3,5-二甲基异噁唑-4-基)甲基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物40) Example 40: N 4 -((3,5-dimethylisoxazol-4-yl)methyl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine -2,4-Diamine (Compound 40)
第一步:N 4-((3,5-二甲基异噁唑-4-基)甲基)-6-碘噻吩并[3,2-d]嘧啶-2,4-二胺(化合物40a) The first step: N 4 -((3,5-dimethylisoxazol-4-yl)methyl)-6-iodothieno[3,2-d]pyrimidine-2,4-diamine (compound 40a)
室温下,将化合物10b(50mg)加入DMF(2mL)中,继续依次加入DIPEA(110.24mg)和BOP(90.69mg),搅拌5min。继续加入3,5-二甲基-4-氨甲基异噁唑盐酸盐(83.23mg),反应16h,得到粗品化合物40a,直接用于下一步反应。At room temperature, compound 10b (50 mg) was added to DMF (2 mL), DIPEA (110.24 mg) and BOP (90.69 mg) were added successively, and the mixture was stirred for 5 min. Continue to add 3,5-dimethyl-4-aminomethylisoxazole hydrochloride (83.23mg) and react for 16h to obtain the crude compound 40a, which was directly used in the next reaction.
MS(ESI,m/z):402.0[M+H] +. MS(ESI,m/z):402.0[M+H] + .
第二步:N 4-((3,5-二甲基异噁唑-4-基)甲基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物40) The second step: N 4 -((3,5-dimethylisoxazol-4-yl)methyl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine -2,4-Diamine (Compound 40)
向第一步中得到的化合物40a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(66.20mg)、Pd(dppf)Cl 2·CH 2Cl 2(14mg)、K 2CO 3(70.73mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物40(11mg)。 To the reaction solution of compound 40a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (66.20mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (14mg), K 2 CO were sequentially added 3 (70.73mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 40 (11 mg).
MS(ESI,m/z):342.1[M+H] +. MS(ESI,m/z):342.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.06(s,1H),7.84(s,1H),7.60(t,J=5.3Hz,1H),7.25(s,1H),6.78(s,1H),5.87(s,2H),4.36(d,J=5.4Hz,2H),2.43(s,3H),2.23(s,3H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.06 (s, 1H), 7.84 (s, 1H), 7.60 (t, J = 5.3 Hz, 1H), 7.25 (s, 1H), 6.78 (s, 1H), 5.87 (s, 2H), 4.36 (d, J = 5.4 Hz, 2H), 2.43 (s, 3H), 2.23 (s, 3H).
实施例41:顺式-3-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)-1-甲基环丁醇(化合物41)Example 41: cis-3-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)-1-methyl ring Butanol (Compound 41)
第一步:顺式-3-((2-氨基-6-碘噻吩并[3,2-d]嘧啶-4-基)氨基)-1-甲基环丁醇(化合物41a)The first step: cis-3-((2-amino-6-iodothieno[3,2-d]pyrimidin-4-yl)amino)-1-methylcyclobutanol (Compound 41a)
室温下,将化合物10b(50mg)加入DMF(2mL)中,继续依次加入DIPEA(110.24mg)和BOP(90.69mg),搅拌5min。继续加入顺式-3-氨基-1-甲基环丁醇盐酸盐(51.77mg),反应16h,得到粗品化合物41a,直接用于下一步反应。At room temperature, compound 10b (50 mg) was added to DMF (2 mL), DIPEA (110.24 mg) and BOP (90.69 mg) were added successively, and the mixture was stirred for 5 min. Continue to add cis-3-amino-1-methylcyclobutanol hydrochloride (51.77 mg) and react for 16 h to obtain the crude compound 41a, which was directly used in the next reaction.
MS(ESI,m/z):377.0[M+H] +. MS(ESI,m/z): 377.0[M+H] + .
第二步:顺式-3-((2-氨基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-4-基)氨基)-1-甲基环丁醇(化合物41)Step 2: cis-3-((2-amino-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidin-4-yl)amino)-1-methyl ring Butanol (Compound 41)
向第一步中得到的化合物41a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(66.20mg)、Pd(dppf)Cl 2·CH 2Cl 2(14mg)、K 2CO 3(70.73mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物494(13mg)。 To the reaction solution of compound 41a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (66.20mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (14mg), K 2 CO were sequentially added 3 (70.73mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 494 (13 mg).
MS(ESI,m/z):317.1[M+H] +. MS(ESI,m/z): 317.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.07(s,1H),7.85(s,1H),7.53(s,1H),7.24(s,1H),6.78(s,1H),5.95(s,2H),4.96(s,1H),4.20–4.04(m,1H),2.39–2.31(m,2H),2.16–2.05(m,2H),1.27(s,3H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.07 (s, 1H), 7.85 (s, 1H), 7.53 (s, 1H), 7.24 (s, 1H), 6.78 (s, 1H), 5.95 ( s, 2H), 4.96 (s, 1H), 4.20-4.04 (m, 1H), 2.39-2.31 (m, 2H), 2.16-2.05 (m, 2H), 1.27 (s, 3H).
实施例42:N 4-((1-甲基-1H-吡唑-4-基)甲基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物42) Example 42: N 4 -((1-methyl-1H-pyrazol-4-yl)methyl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine- 2,4-Diamine (Compound 42)
第一步:6-碘-N 4-((1-甲基-1H-吡唑-4-基)甲基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物42a) The first step: 6-iodo-N 4 -((1-methyl-1H-pyrazol-4-yl)methyl)thieno[3,2-d]pyrimidine-2,4-diamine (Compound 42a )
室温下,将化合物10b(50mg)加入DMF(2mL)中,继续依次加入DIPEA(110.24mg)和BOP(90.69mg),搅拌5min。继续加入4-(氨甲基)-1-甲基吡唑(56.88mg),反应16h,得到粗品化合物42a,直接用于下一步反应。At room temperature, compound 10b (50 mg) was added to DMF (2 mL), DIPEA (110.24 mg) and BOP (90.69 mg) were added successively, and the mixture was stirred for 5 min. Continue to add 4-(aminomethyl)-1-methylpyrazole (56.88mg) and react for 16h to obtain the crude compound 42a, which was directly used in the next reaction.
MS(ESI,m/z):387.0[M+H] +. MS(ESI,m/z): 387.0[M+H] + .
第二步:N 4-((1-甲基-1H-吡唑-4-基)甲基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物42) The second step: N 4 -((1-methyl-1H-pyrazol-4-yl)methyl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine- 2,4-Diamine (Compound 42)
向第一步中得到的化合物42a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(66.20mg)、Pd(dppf)Cl 2·CH 2Cl 2(14mg)、K 2CO 3(70.73mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物42(13mg)。 To the reaction solution of compound 42a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (66.20mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (14mg), K 2 CO were sequentially added 3 (70.73mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 42 (13 mg).
MS(ESI,m/z):327.1[M+H] +. MS(ESI,m/z): 327.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.06(s,1H),7.84(s,1H),7.71–7.62(m,2H),7.41(s,1H),7.25(s,1H),6.78(s,1H),6.01(s,2H),4.42(d,J=5.6Hz,2H),3.77(s,3H). 1 H NMR(DMSO-d 6 ,400MHz)δ13.06(s,1H),7.84(s,1H),7.71-7.62(m,2H),7.41(s,1H),7.25(s,1H), 6.78 (s, 1H), 6.01 (s, 2H), 4.42 (d, J = 5.6 Hz, 2H), 3.77 (s, 3H).
实施例43:N 4-环戊基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物43) Example 43: N 4 -Cyclopentyl-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4-diamine (Compound 43)
第一步:N 4-环戊基-6-碘噻吩并[3,2-d]嘧啶-2,4-二胺(化合物43a) The first step: N 4 -cyclopentyl-6-iodothieno[3,2-d]pyrimidine-2,4-diamine (compound 43a)
室温下,将化合物10b(50mg)加入DMF(2mL)中,继续依次加入DIPEA(110.24mg)和BOP(90.69mg),搅拌5min。继续加入环戊胺(43.58mg),反应16h,得到粗品化合物43a,直接用于下一步反应。At room temperature, compound 10b (50 mg) was added to DMF (2 mL), DIPEA (110.24 mg) and BOP (90.69 mg) were added successively, and the mixture was stirred for 5 min. Cyclopentylamine (43.58mg) was continuously added and reacted for 16h to obtain crude compound 43a, which was directly used in the next reaction.
MS(ESI,m/z):361.0[M+H] +. MS(ESI,m/z): 361.0[M+H] + .
第二步:N 4-环戊基-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物43) The second step: N 4 -cyclopentyl-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4-diamine (Compound 43)
向第一步中得到的化合物43a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(66.20mg)、Pd(dppf)Cl 2·CH 2Cl 2(14mg)、K 2CO 3(70.73mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物43(7mg)。 To the reaction solution of compound 43a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (66.20mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (14mg), K 2 CO were sequentially added 3 (70.73mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 43 (7 mg).
MS(ESI,m/z):301.1[M+H] +. MS(ESI,m/z): 301.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.06(s,1H),7.84(s,1H),7.23(s,1H),7.15(d,J=6.7Hz,1H),6.77(s,1H),5.84(s,2H),4.53–4.40(m,1H),2.01–1.88(m,2H),1.78–1.66(m,2H),1.60–1.47(m,4H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.06 (s, 1H), 7.84 (s, 1H), 7.23 (s, 1H), 7.15 (d, J = 6.7 Hz, 1H), 6.77 (s, 1H), 5.84 (s, 2H), 4.53--4.40 (m, 1H), 2.01--1.88 (m, 2H), 1.78--1.66 (m, 2H), 1.60--1.47 (m, 4H).
实施例44:N 4-(2-甲氧基-2-甲基丙基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物44) Example 44: N 4 -(2-methoxy-2-methylpropyl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4-di Amine (Compound 44)
第一步:6-碘-N 4-(2-甲氧基-2-甲基丙基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物44a) The first step: 6-iodo-N 4 -(2-methoxy-2-methylpropyl)thieno[3,2-d]pyrimidine-2,4-diamine (compound 44a)
室温下,将化合物10b(50mg)加入DMF(2mL)中,继续依次加入DIPEA(110.24mg)和BOP(90.69mg),搅拌5min。继续加入2-甲氧基-2-甲基丙胺盐酸盐(71.46mg),反应16h,得到粗品化合物44a,直接用于下一步反应。At room temperature, compound 10b (50 mg) was added to DMF (2 mL), DIPEA (110.24 mg) and BOP (90.69 mg) were added successively, and the mixture was stirred for 5 min. 2-Methoxy-2-methylpropylamine hydrochloride (71.46 mg) was continuously added and reacted for 16 h to obtain crude compound 44a, which was directly used in the next reaction.
MS(ESI,m/z):379.0[M+H] +. MS(ESI,m/z): 379.0[M+H] + .
第二步:N 4-(2-甲氧基-2-甲基丙基)-6-(1H-吡唑-5-基)噻吩并[3,2-d]嘧啶-2,4-二胺(化合物44) The second step: N 4 -(2-methoxy-2-methylpropyl)-6-(1H-pyrazol-5-yl)thieno[3,2-d]pyrimidine-2,4-di Amine (Compound 44)
向第一步中得到的化合物44a的反应液中,依次加入1H-吡唑-5-硼酸频哪酯(66.20mg)、Pd(dppf)Cl 2·CH 2Cl 2(14mg)、K 2CO 3(70.73mg)和水(0.5mL),N 2保护下加热至100℃反应4h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物44(8mg)。 To the reaction solution of compound 44a obtained in the first step, 1H-pyrazole-5-boronic acid pinacolate (66.20mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (14mg), K 2 CO were sequentially added 3 (70.73mg) and water (0.5mL), heated to 100℃ under the protection of N 2 to react for 4h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 44 (8 mg).
MS(ESI,m/z):319.1[M+H] +. MS(ESI,m/z):319.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.06(s,1H),7.84(s,1H),7.24(s,1H),6.92(t,J=5.6Hz,1H),6.78(s,1H),5.84(s,2H),3.53(d,J=6.0Hz,2H),3.17(s,3H),1.13(s,6H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.06 (s, 1H), 7.84 (s, 1H), 7.24 (s, 1H), 6.92 (t, J = 5.6 Hz, 1H), 6.78 (s, 1H), 5.84 (s, 2H), 3.53 (d, J = 6.0 Hz, 2H), 3.17 (s, 3H), 1.13 (s, 6H).
实施例45:N 4-(1-甲基环丁基)-7-(1H-吡唑-5-基)喹唑啉-2,4-二胺甲酸盐(化合物45s) Example 45: N 4 -(1-methylcyclobutyl)-7-(1H-pyrazol-5-yl)quinazoline-2,4-diamine formate (Compound 45s)
第一步:7-溴-N 4-(1-甲基环丁基)喹唑啉-2,4-二胺(化合物45a) The first step: 7-bromo-N 4 -(1-methylcyclobutyl)quinazoline-2,4-diamine (Compound 45a)
室温下,将化合物2b(70mg)、1-甲基环丁胺盐酸盐(70.92mg)、BOP(89.28mg)和DIPEA(188.43mg)加入DMF(2mL)中,反应16h,得到粗品化合物45a,直接用于下一步反应。At room temperature, compound 2b (70mg), 1-methylcyclobutylamine hydrochloride (70.92mg), BOP (89.28mg) and DIPEA (188.43mg) were added to DMF (2mL) and reacted for 16h to obtain crude compound 45a , Directly used in the next reaction.
MS(ESI,m/z):309.1[M+H] +. MS(ESI,m/z): 309.1[M+H] + .
第二步:N 4-(1-甲基环丁基)-7-(1H-吡唑-5-基)喹唑啉-2,4-二胺甲酸盐(化合物45s) The second step: N 4 -(1-methylcyclobutyl)-7-(1H-pyrazol-5-yl)quinazoline-2,4-diamine formate (Compound 45s)
向第一步中得到的化合物45a的反应液中,依次加入碳酸钾(94.34mgl)、Pd(dppf)Cl 2·CH 2Cl 2(27.89mg)、1H-吡唑-5-硼酸频哪酯(87.86mg)和水(1mL),N 2保护下加热至100℃反应3h。将反应液硅藻土过滤,含化合物45的滤液通过Prep-HPLC分离纯化(洗脱条件3),冻干得到化合物45s(2mg)。 To the reaction solution of compound 45a obtained in the first step, potassium carbonate (94.34mgl), Pd(dppf)Cl 2 ·CH 2 Cl 2 (27.89mg), 1H-pyrazole-5-boronic acid pinacol ester were sequentially added (87.86mg) and water (1mL), heated to 100°C under the protection of N 2 to react for 3h. The reaction solution was filtered through Celite, and the filtrate containing compound 45 was separated and purified by Prep-HPLC (elution condition 3), and lyophilized to obtain compound 45s (2 mg).
MS(ESI,m/z):295.1[M+H] +. MS(ESI,m/z): 295.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ8.32(s,1H),8.11(d,J=8.5Hz,1H),7.99(s,1H),7.76(s,1H),7.63(d,J=1.7Hz,1H),7.53(d,J=8.4Hz,1H),6.81(d,J=2.2Hz,1H),6.63(s,2H),2.42-2.34(m,2H),2.20-2.11(m,2H),1.91-1.80(m,2H),1.60(s,3H). 1 H NMR(DMSO-d 6 ,400MHz)δ8.32(s,1H),8.11(d,J=8.5Hz,1H),7.99(s,1H),7.76(s,1H),7.63(d, J = 1.7Hz, 1H), 7.53 (d, J = 8.4Hz, 1H), 6.81 (d, J = 2.2Hz, 1H), 6.63 (s, 2H), 2.42-2.34 (m, 2H), 2.20- 2.11 (m, 2H), 1.91-1.80 (m, 2H), 1.60 (s, 3H).
实施例46:1-(2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)哌啶-4-醇(化合物46)Example 46: 1-(2-Amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)piperidin-4-ol (Compound 46)
第一步:1-(2-氨基-7-溴喹唑啉-4-基)哌啶-4-醇(化合物46a)The first step: 1-(2-amino-7-bromoquinazolin-4-yl)piperidin-4-ol (compound 46a)
室温下,将化合物2b(70mg)、哌啶-4-醇(58.99mg)、BOP(89.28mg)和DIPEA(188.43mg)加入DMF(2mL)中,反应16h,得到粗品化合物46a,直接用于下一步反应。At room temperature, compound 2b (70mg), piperidin-4-ol (58.99mg), BOP (89.28mg) and DIPEA (188.43mg) were added to DMF (2mL) and reacted for 16h to obtain crude compound 46a, which was used directly Next reaction.
MS(ESI,m/z):325.1[M+H] +. MS(ESI,m/z): 325.1[M+H] + .
第二步:1-(2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)哌啶-4-醇(化合物46)Step 2: 1-(2-Amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)piperidin-4-ol (Compound 46)
向第一步中得到的化合物46a的反应液中,依次加入碳酸钾(94.34mg)、Pd(dppf)Cl 2·CH 2Cl 2(27.89mg)、1H-吡唑-5-硼酸频哪酯(87.86mg)和水(1mL),N 2保护下加热至100℃反应3h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物46(7mg)。 To the reaction solution of compound 46a obtained in the first step, potassium carbonate (94.34mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (27.89mg), 1H-pyrazole-5-boronic acid pinacolate were sequentially added (87.86mg) and water (1mL), heated to 100°C under the protection of N 2 to react for 3h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 46 (7 mg).
MS(ESI,m/z):311.1[M+H] +. MS(ESI,m/z):311.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ13.01(s,1H),7.82(s,1H),7.73-7.68(m,2H),7.56(s,1H),6.82(d,J= 2.2Hz,1H),6.29(s,2H),4.80(d,J=4.2Hz,1H),3.92-3.87(m,2H),3.79-3.74(m,1H),3.26-3.19(m,2H),1.93-1.88m,2H),1.62-1.53(m,2H). 1 H NMR (DMSO-d 6 , 400MHz) δ 13.01 (s, 1H), 7.82 (s, 1H), 7.73-7.68 (m, 2H), 7.56 (s, 1H), 6.82 (d, J = 2.2 Hz, 1H), 6.29 (s, 2H), 4.80 (d, J = 4.2 Hz, 1H), 3.92-3.87 (m, 2H), 3.79-3.74 (m, 1H), 3.26-3.19 (m, 2H) ,1.93-1.88m,2H),1.62-1.53(m,2H).
实施例47:顺式-3-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)-1-甲基环丁醇(化合物47)Example 47: cis-3-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)-1-methylcyclobutanol (Compound 47)
第一步:顺式-3-((2-氨基-7-溴喹唑啉-4-基)氨基)-1-甲基环丁醇(化合物47a)The first step: cis-3-((2-amino-7-bromoquinazolin-4-yl)amino)-1-methylcyclobutanol (Compound 47a)
室温下,将化合物2b(70mg)、顺式-3-氨基-1-甲基环丁醇盐酸盐(58.99mg)、BOP(89.28mg)和DIPEA(188.43mg)加入DMF(2mL)中,反应16h,得到粗品化合物47a,直接用于下一步反应。At room temperature, compound 2b (70mg), cis-3-amino-1-methylcyclobutanol hydrochloride (58.99mg), BOP (89.28mg) and DIPEA (188.43mg) were added to DMF (2mL), After 16 hours of reaction, the crude compound 47a was obtained, which was directly used in the next reaction.
MS(ESI,m/z):323.1[M+H] +. MS(ESI,m/z): 323.1[M+H] + .
第二步:顺式-3-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)-1-甲基环丁醇(化合物47)The second step: cis-3-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)-1-methylcyclobutanol (Compound 47)
向第一步中得到的化合物47a的反应液中,依次加入碳酸钾(94.34mg)、Pd(dppf)Cl 2·CH 2Cl 2(27.89mg)、1H-吡唑-5-硼酸频哪酯(87.86mg)和水(1mL),N 2保护下加热至100℃反应3h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物47(4mg)。 To the reaction solution of compound 47a obtained in the first step, potassium carbonate (94.34mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (27.89mg), 1H-pyrazole-5-boronic acid pinacolate were sequentially added (87.86mg) and water (1mL), heated to 100°C under the protection of N 2 to react for 3h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 47 (4 mg).
MS(ESI,m/z):311.1[M+H] +. MS(ESI,m/z):311.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ8.09(d,J=8.5Hz,1H),7.84(d,J=6.4Hz,1H),7.74(d,J=2.2Hz,1H),7.58(d,J=1.7Hz,1H),7.48-7.46(m,1H),6.80(d,J=2.2Hz,1H),6.04(s,2H),5.02(s,1H),4.21-4.15(m,1H),2.43-2.37(m,2H),2.20-2.14(m,2H),1.30(s,3H). 1 H NMR (DMSO-d 6 , 400MHz) δ 8.09 (d, J = 8.5 Hz, 1H), 7.84 (d, J = 6.4 Hz, 1H), 7.74 (d, J = 2.2 Hz, 1H), 7.58 (d,J=1.7Hz,1H),7.48-7.46(m,1H),6.80(d,J=2.2Hz,1H),6.04(s,2H),5.02(s,1H),4.21-4.15( m, 1H), 2.43-2.37 (m, 2H), 2.20-2.14 (m, 2H), 1.30 (s, 3H).
实施例48:(S)-2-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)-1-丙醇(化合物48)Example 48: (S)-2-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)-1-propanol (Compound 48)
第一步:(S)-2-((2-氨基-7-溴喹唑啉-4-基)氨基)-1-丙醇(化合物48a)The first step: (S)-2-((2-amino-7-bromoquinazolin-4-yl)amino)-1-propanol (Compound 48a)
室温下,将化合物2b(70mg)、(S)-2-氨基-1-丙醇(43.80mg)、BOP(89.28mg)和DIPEA(188.43mg)加入DMF(2mL)中,反应16h,得到粗品化合物48a,直接用于下一步反应。At room temperature, compound 2b (70mg), (S)-2-amino-1-propanol (43.80mg), BOP (89.28mg) and DIPEA (188.43mg) were added to DMF (2mL) and reacted for 16h to obtain crude product Compound 48a was directly used in the next reaction.
MS(ESI,m/z):299.1[M+H] +. MS(ESI,m/z): 299.1[M+H] + .
第二步:(S)-2-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)-1-丙醇(化合物48)The second step: (S)-2-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)-1-propanol (Compound 48)
向第一步中得到的化合物48a的反应液中,依次加入碳酸钾(94.34mg)、Pd(dppf)Cl 2·CH 2Cl 2(27.89mg)、1H-吡唑-5-硼酸频哪酯(87.86mg)和水(1mL),N 2保护下加热至100℃反应3h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物48(10mg) To the reaction solution of compound 48a obtained in the first step, potassium carbonate (94.34mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (27.89mg), 1H-pyrazole-5-boronic acid pinacolate were sequentially added (87.86mg) and water (1mL), heated to 100°C under the protection of N 2 to react for 3h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 48 (10 mg)
MS(ESI,m/z):285.1[M+H] +. MS(ESI,m/z):285.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ12.99(s,1H),8.06(d,J=8.5Hz,1H),7.80(s,1H),7.60(d,J=1.7Hz,1H),7.57-7.44(m,1H),7.38(d,J=8.0Hz,1H),6.81(d,J=2.2Hz,1H),6.06(s,2H),4.79-4.77(m,1H),4.44-4.37(m,1H),3.56-3.51(m,1H),3.50–3.43(m,1H),1.20(d,J=6.7Hz,3H). 1 H NMR(DMSO-d 6 ,400MHz)δ12.99(s,1H), 8.06(d,J=8.5Hz,1H), 7.80(s,1H), 7.60(d,J=1.7Hz,1H) ,7.57-7.44(m,1H),7.38(d,J=8.0Hz,1H), 6.81(d,J=2.2Hz,1H), 6.06(s,2H), 4.79-4.77(m,1H), 4.44-4.37(m,1H), 3.56-3.51(m,1H), 3.50-3.43(m,1H), 1.20(d,J=6.7Hz,3H).
实施例49:(R)-2-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)-1-丁醇(化合物49)Example 49: (R)-2-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)-1-butanol (Compound 49)
第一步:(R)-2-((2-氨基-7-溴喹唑啉-4-基)氨基)-1-丁醇(化合物49a)The first step: (R)-2-((2-amino-7-bromoquinazolin-4-yl)amino)-1-butanol (Compound 49a)
室温下,将化合物2b(70mg)、(R)-2-氨基-1-丁醇(51.98mg)、BOP(89.28mg)和DIPEA(188.43mg)加入DMF(2mL)中,反应16h,得到粗品化合物49a,直接用于下一步反应。At room temperature, compound 2b (70mg), (R)-2-amino-1-butanol (51.98mg), BOP (89.28mg) and DIPEA (188.43mg) were added to DMF (2mL) and reacted for 16h to obtain crude product Compound 49a was directly used in the next reaction.
MS(ESI,m/z):313.1[M+H] +. MS(ESI,m/z): 313.1[M+H] + .
第二步:(R)-2-((2-氨基-7-(1H-吡唑-5-基)喹唑啉-4-基)氨基)-1-丁醇(化合物49)The second step: (R)-2-((2-amino-7-(1H-pyrazol-5-yl)quinazolin-4-yl)amino)-1-butanol (Compound 49)
向第一步中得到的化合物49a的反应液中,依次加入碳酸钾(93.72mg)、Pd(dppf)Cl 2·CH 2Cl 2(27.71mg)、1H-吡唑-5-硼酸频哪酯(87.86mg)和水(1mL),N 2保护下加热至100℃反应3h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物49(10mg)。 To the reaction solution of compound 49a obtained in the first step, potassium carbonate (93.72mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (27.71mg), 1H-pyrazole-5-boronic acid pinacolate were sequentially added (87.86mg) and water (1mL), heated to 100°C under the protection of N 2 to react for 3h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 49 (10 mg).
MS(ESI,m/z):299.1[M+H] +. MS(ESI,m/z): 299.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ12.98(s,1H),8.10(d,J=8.4Hz,1H),7.81(s,1H),7.59(d,J=1.7Hz,1H),7.51(s,1H),7.29(d,J=8.2Hz,1H),6.81(d,J=2.2Hz,1H),6.00(s,2H),4.74-4.71(m,1H),4.32-4.23(m,1H),3.57-3.43(m,2H),1.82-1.51(m,2H),0.92-0.89(m,3H). 1 H NMR(DMSO-d 6 ,400MHz)δ12.98(s,1H), 8.10(d,J=8.4Hz,1H), 7.81(s,1H), 7.59(d,J=1.7Hz,1H) ,7.51(s,1H),7.29(d,J=8.2Hz,1H),6.81(d,J=2.2Hz,1H),6.00(s,2H),4.74-4.71(m,1H),4.32- 4.23 (m, 1H), 3.57-3.43 (m, 2H), 1.82-1.51 (m, 2H), 0.92-0.89 (m, 3H).
实施例50:(S)-N 4-(1-甲氧基丙-2-基)-7-(1H-吡唑-5-基)喹唑啉-2,4-二胺(化合物50) Example 50: (S)-N 4 -(1-methoxyprop-2-yl)-7-(1H-pyrazol-5-yl)quinazoline-2,4-diamine (Compound 50)
第一步:(S)-7-溴-N 4-(1-甲氧基丙-2-基)喹唑啉-2,4-二胺(化合物50a) The first step: (S)-7-bromo-N 4 -(1-methoxyprop-2-yl)quinazoline-2,4-diamine (compound 50a)
室温下,将化合物2b(70mg)、(S)-1-甲氧基丙-2-胺(51.98mg)、BOP(89.28mg)和DIPEA(188.43mg)加入DMF(2mL)中,反应16h,得到粗品化合物50a,直接用于下一步反应。At room temperature, compound 2b (70mg), (S)-1-methoxypropan-2-amine (51.98mg), BOP (89.28mg) and DIPEA (188.43mg) were added to DMF (2mL) and reacted for 16h. The crude compound 50a was obtained, which was directly used in the next reaction.
MS(ESI,m/z):313.1[M+H]+.MS(ESI, m/z): 313.1[M+H]+.
第二步:(S)-N 4-(1-甲氧基丙-2-基)-7-(1H-吡唑-5-基)喹唑啉-2,4-二胺(化合物50) The second step: (S)-N 4 -(1-methoxyprop-2-yl)-7-(1H-pyrazol-5-yl)quinazoline-2,4-diamine (Compound 50)
向第一步中得到的化合物50a的反应液中,依次加入碳酸钾(93.72mg)、Pd(dppf)Cl 2·CH 2Cl 2(27.71mg)、1H-吡唑-5-硼酸频哪酯(87.86mg)和水(1mL),N 2保护下加热至100℃反应3h。将反应液硅藻土过滤,滤液通过Prep-HPLC分离纯化(洗脱条件4),冻干得到化合物50。 To the reaction solution of compound 50a obtained in the first step, potassium carbonate (93.72 mg), Pd(dppf)Cl 2 ·CH 2 Cl 2 (27.71 mg), 1H-pyrazole-5-boronic acid pinacolate were sequentially added (87.86mg) and water (1mL), heated to 100°C under the protection of N 2 to react for 3h. The reaction solution was filtered through Celite, and the filtrate was separated and purified by Prep-HPLC (elution condition 4), and lyophilized to obtain compound 50.
MS(ESI,m/z):299.1[M+H] +. MS(ESI,m/z): 299.1[M+H] + .
1H NMR(DMSO-d 6,400MHz)δ12.98(s,1H),8.05(d,J=8.6Hz,1H),7.81(s,1H),7.59(d,J=1.7Hz,1H),7.46(d,J=9.3Hz,2H),6.81(d,J=2.2Hz,1H),6.04(s,2H),4.64-4.57(m,1H),3.51-3.47(m,1H),3.35-3.32(m,1H),3.29(s,3H),1.21(d,J=6.7Hz,3H). 1 H NMR(DMSO-d 6 ,400MHz)δ12.98(s,1H), 8.05(d,J=8.6Hz,1H), 7.81(s,1H), 7.59(d,J=1.7Hz,1H) ,7.46(d,J=9.3Hz,2H),6.81(d,J=2.2Hz,1H),6.04(s,2H),4.64-4.57(m,1H),3.51-3.47(m,1H), 3.35-3.32(m,1H), 3.29(s,3H), 1.21(d,J=6.7Hz,3H).
活性测试Activity test
实验例1:本发明化合物对PMA诱导分化后的THP-1细胞中的IL-1β表达的激动作用Experimental Example 1: The agonistic effect of the compound of the present invention on IL-1β expression in THP-1 cells after PMA-induced differentiation
本实验采用均相时间分辨荧光(HTRF)检测方法,测试本发明化合物对NLRP3下游细胞因子IL-1β水平的影响,以评估化合物在细胞水平上对hNLRP3炎症小体或hNLRP3炎症小体通路的激动作用。In this experiment, a homogeneous time-resolved fluorescence (HTRF) detection method was used to test the effect of the compound of the present invention on the level of the downstream cytokine IL-1β of NLRP3 to evaluate the compound's activation of hNLRP3 inflammasome or hNLRP3 inflammasome pathway at the cellular level effect.
试剂:RPMI 1640(Hyclone);热灭活的FBS(胎牛血清)(Gibco);PMA(碧云天)Reagents: RPMI 1640 (Hyclone); heat-inactivated FBS (fetal bovine serum) (Gibco); PMA (Biyuntian)
细胞:THP-1(南京科佰)Cell: THP-1 (Nanjing Kebai)
试剂盒:IL-1βassay kit(CISBIO)Kit: IL-1βassay kit (CISBIO)
实验步骤:Experimental steps:
1)将处于对数生长期的THP-1细胞以5×10 5个/孔的密度接种于T75培养瓶中,置于37℃、5%CO 2的细胞培养箱中培养24小时后,用1μM PMA诱导THP-1悬浮细胞成为贴壁状态的巨噬细胞。培养基为包含10%热灭活FBS和0.05mMβ-巯基乙醇的RPMI 1640。 1) The THP-1 cells in the logarithmic growth phase were seeded in a T75 culture flask at a density of 5×10 5 cells/well, and then cultured in a 37°C, 5% CO 2 cell incubator for 24 hours. 1μM PMA induces THP-1 suspension cells to become adherent macrophages. The medium was RPMI 1640 containing 10% heat-inactivated FBS and 0.05 mM β-mercaptoethanol.
2)细胞诱导培养24小时后,胰酶消化贴壁细胞。1000rpm离心5min,去上清液,使用包含2%热灭活FBS的RPMI 1640培养基重悬细胞,至密度为2×10 6个/mL。取50μL/孔的细胞重悬液铺至96孔板,每孔细胞数目为1×10 5个。 2) After the cells are induced and cultured for 24 hours, the adherent cells are trypsinized. Centrifuge at 1000 rpm for 5 min, remove the supernatant, and resuspend the cells in RPMI 1640 medium containing 2% heat-inactivated FBS to a density of 2×10 6 cells/mL. Take 50μL/well of cell resuspension and spread it on a 96-well plate. The number of cells per well is 1×10 5 cells.
3)取适量的10mM待测化合物的DMSO溶液,用含2%热灭活FBS的RPMI 1640培养基配制成2×测试浓度。取50μL/孔稀释液加至96孔板的细胞中,充分混匀后,将板放置于37℃、5%CO 2的细胞培养箱中培养6小时。收集上清液,按照IL-1β检测试剂盒说明书测量IL-1β的水平。 3) Take an appropriate amount of 10 mM DMSO solution of the compound to be tested, and use RPMI 1640 medium containing 2% heat-inactivated FBS to prepare a 2× test concentration. Take 50 μL/well of the dilution and add it to the cells in the 96-well plate, mix well, and place the plate in a 37°C, 5% CO 2 cell incubator for 6 hours. Collect the supernatant, and measure the level of IL-1β in accordance with the IL-1β detection kit instructions.
4)EC 50通过GraphPad软件log(agonist)vs.response--Variable slope四参数法拟合,结果见表1。 4) The EC 50 was fitted by the GraphPad software log(agonist) vs.response-Variable slope four-parameter method. The results are shown in Table 1.
实验例2:本发明化合物对PMA诱导分化后NLRP3缺失的THP1细胞(THP1- defNLRP3细胞)中的IL-1β表达的激动作用 Experimental Example 2: The agonistic effect of the compound of the present invention on the expression of IL-1β in THP1 cells (THP1-def NLRP3 cells) lacking NLRP3 after PMA-induced differentiation
本实验采用HTRF检测方法测试本发明化合物对THP1- defNLRP3细胞中IL-1β水平的影响,以评估化合物对hNLRP3炎症小体或hNLRP3炎症小体通路激动作用的特异性。 In this experiment, the HTRF detection method was used to test the effect of the compound of the present invention on the level of IL-1β in THP1-def NLRP3 cells to evaluate the specificity of the compound on hNLRP3 inflammasome or hNLRP3 inflammasome pathway agonism.
试剂:如实验例1所述Reagents: as described in Experimental Example 1
细胞:THP1- defNLRP3(InvivoGen) Cell: THP1- def NLRP3 (InvivoGen)
试剂盒:IL-1βassay kit(CISBIO)Kit: IL-1βassay kit (CISBIO)
实验步骤:Experimental steps:
1)将处于对数生长期的THP1- defNLRP3细胞以5×10 5个/孔的密度接种于T75培养瓶中,置于37℃、5%CO 2的细胞培养箱中培养24小时后,用1μM PMA诱导THP1- defNLRP3悬浮细胞成为贴壁状态的巨噬细胞。培养基为包含10%热灭活FBS和0.05mMβ-巯基乙醇的RPMI 1640。 1) THP1-def NLRP3 cells in the logarithmic growth phase were seeded in a T75 culture flask at a density of 5×10 5 cells/well, and then cultured in a cell incubator at 37°C and 5% CO 2 for 24 hours. 1μM PMA was used to induce THP1- def NLRP3 suspension cells to become adherent macrophages. The medium was RPMI 1640 containing 10% heat-inactivated FBS and 0.05 mM β-mercaptoethanol.
2)细胞诱导培养24小时后,胰酶消化贴壁细胞。1000rpm离心5min,去上清液,使用包含2%热灭活FBS的RPMI 1640培养基重悬细胞,至密度为2×10 6个/mL。取50μL/孔的细胞重悬液铺至96孔板,每孔细胞数目为1×10 5个。 2) After the cells are induced and cultured for 24 hours, the adherent cells are trypsinized. Centrifuge at 1000 rpm for 5 min, remove the supernatant, and resuspend the cells in RPMI 1640 medium containing 2% heat-inactivated FBS to a density of 2×10 6 cells/mL. Take 50μL/well of cell resuspension and spread it on a 96-well plate. The number of cells per well is 1×10 5 cells.
3)取适量的10mM待测化合物的DMSO溶液,用含2%热灭活FBS的RPMI 1640培养基配制成2×测试浓度。取50μL/孔稀释液加至96孔板的细胞中,充分混匀后,将板放置于37℃、5%CO 2的细胞培养箱中培养6小时。收集上清液,按照IL-1β检测试剂盒说明书测量IL-1β的水平。 3) Take an appropriate amount of 10 mM DMSO solution of the compound to be tested, and use RPMI 1640 medium containing 2% heat-inactivated FBS to prepare a 2× test concentration. Take 50 μL/well of the dilution and add it to the cells in the 96-well plate, mix well, and place the plate in a 37°C, 5% CO 2 cell incubator for 6 hours. Collect the supernatant, and measure the level of IL-1β in accordance with the IL-1β detection kit instructions.
4)EC 50通过GraphPad软件log(agonist)vs.response--Variable slope四参数法拟合,结果见表1。 4) The EC 50 was fitted by the GraphPad software log(agonist) vs.response-Variable slope four-parameter method. The results are shown in Table 1.
实验例3:本发明化合物对hTLR7的激动作用Experimental Example 3: The agonistic effect of the compound of the present invention on hTLR7
本实验通过检测HEK-hTLR7-NF-κB-荧光素酶报告基因细胞中的荧光素酶来测试本发明化合物对TLR7信号通路的激活作用,以评估化合物对NLRP3通路激动作用的特异性。In this experiment, the luciferase in HEK-hTLR7-NF-κB-luciferase reporter gene cells was tested to test the activating effect of the compound of the present invention on the TLR7 signaling pathway, so as to evaluate the specificity of the compound's agonistic effect on the NLRP3 pathway.
试剂:DMEM(High glucose),FBS(Gibco)Reagents: DMEM (High glucose), FBS (Gibco)
细胞:HEK-hTLR7-NF-κB-Luciferase报告基因细胞(人源TLR7NF-κB-荧光素酶报告基因细胞)(南京科佰)Cells: HEK-hTLR7-NF-κB-Luciferase reporter cells (human TLR7NF-κB-luciferase reporter cells) (Nanjing Kebai)
试剂盒:Bright-Glo TM Luciferase检测试剂盒(Promega) Kit: Bright-Glo TM Luciferase Detection Kit (Promega)
实验步骤:Experimental steps:
1)将处于对数生长期的HEK-hTLR7-NF-κB-Luciferase报告基因细胞胰酶消化,用培养基重悬至2×10 6个/mL的浓度。加入50μL/孔细胞重悬液于96孔板中,每孔细胞数为1×10 6个。取适量的10mM待测化合物的DMSO溶液,用培养基配制成2×测试浓度。加50μL/孔至96孔板的细胞中,将96孔板置于37℃、5%CO 2的细胞培养箱中培养16小时。培养基为包含10%FBS的DMEM(High glucose)。 1) Trypsinize HEK-hTLR7-NF-κB-Luciferase reporter gene cells in the logarithmic growth phase, and resuspend them to a concentration of 2×10 6 cells/mL in culture medium. Add 50μL/well of cell resuspension to a 96-well plate, the number of cells per well is 1×10 6 cells. Take an appropriate amount of 10 mM DMSO solution of the test compound, and use the culture medium to prepare a 2× test concentration. Add 50 μL/well to the cells in the 96-well plate, and place the 96-well plate in a 37°C, 5% CO 2 cell incubator for 16 hours. The medium is DMEM (High glucose) containing 10% FBS.
2)细胞孵育结束后,加入100μL/孔Bright-Glo TM Luciferase检测试剂,室温孵育5min。酶标仪读取相对荧光酶活性单位(Relative Luciferase Unit,RLU)。 2) After the cell incubation is over, add 100 μL/well of Bright-Glo TM Luciferase detection reagent and incubate at room temperature for 5 minutes. The microplate reader reads the relative luciferase activity unit (Relative Luciferase Unit, RLU).
3)所测试化合物对hTLR7的刺激作用EC 50通过GraphPad软件log(agonist)vs.response--Variable slope四参数法拟合,结果见表1。 3) The EC 50 of the stimulating effect of the tested compound on hTLR7 was fitted by the GraphPad software log(agonist) vs.response--Variable slope four-parameter method. The results are shown in Table 1.
实验例4:本发明化合物对hTLR8的激动作用Experimental Example 4: The agonistic effect of the compound of the present invention on hTLR8
本实验通过检测HEK-Blue细胞株中碱性磷酸酶的分泌量来测试本发明化合物对TLR8信号通路的激活作用,以评估化合物对NLRP3通路激动作用的特异性。In this experiment, the secretion of alkaline phosphatase in the HEK-Blue cell line was tested to test the activating effect of the compound of the present invention on the TLR8 signaling pathway, so as to evaluate the specificity of the compound's agonistic effect on the NLRP3 pathway.
试剂:DMEM(High glucose),FBS(Gibco),QUANTI-Blue(InvivoGen)Reagents: DMEM (High glucose), FBS (Gibco), QUANTI-Blue (InvivoGen)
细胞:HEK-Blue TM hTLR8细胞(人源TLR 8细胞)(InvivoGen) Cells: HEK-Blue TM hTLR8 cells (human TLR 8 cells) (InvivoGen)
实验步骤:Experimental steps:
1)将处于对数生长期的HEK-Blue TM hTLR8细胞胰酶消化,用培养基重悬至2×10 6个/mL的浓度。加入50μL/孔细胞悬液于96孔板中。取适量的10mM待测化合物的DMSO溶液,用培养基配制成2×测试浓度,加50μL/孔至96孔板的细胞中。将板置于37℃、5%CO 2的细胞培养箱中培养16小时。培养基为包含10%FBS的DMEM(High glucose)。 1) Trypsinize HEK-Blue™ hTLR8 cells in the logarithmic growth phase, and resuspend them to a concentration of 2×10 6 cells/mL in culture medium. Add 50μL/well of cell suspension to 96-well plate. Take an appropriate amount of 10 mM DMSO solution of the compound to be tested, prepare a 2× test concentration with culture medium, and add 50 μL/well to the cells in a 96-well plate. The plate was placed in a 37°C, 5% CO 2 cell incubator for 16 hours. The medium is DMEM (High glucose) containing 10% FBS.
2)细胞孵育结束后,取10μL细胞培养上清液转移至96孔板中。加入90μL/孔的QUANTI-Blue检测溶液,37℃孵育3小时。酶标仪OD 620读数。 2) After the cell incubation is over, take 10 μL of cell culture supernatant and transfer it to a 96-well plate. Add 90μL/well of QUANTI-Blue detection solution and incubate at 37°C for 3 hours. Microplate reader OD 620 reading.
3)所测试化合物对hTLR8的刺激作用EC 50通过GraphPad软件log(agonist)vs.response--Variable slope四参数法拟合,结果见表1。 3) The compound of hTLR8 EC stimulation test by GraphPad Software 50 log (agonist) vs.response - Variable slope fitting four parameters, the results are shown in Table 1.
表1Table 1
结果表明,以表1所列化合物为代表的本发明化合物对PMA诱导分化后的THP-1细胞中的IL-1β表达有明显的激动作用,而对THP-1 defNLRP3细胞中的IL-1β表达即使在最高化合物测试浓度(30μM)下也无激动作用。所测试的所有化合物在100μM下对hTLR7和hTLR8均无激活作用。综上,以表1所列化合物为代表的本发明化合物对hNLRP3和/或其信号通路有明显的特异性激动活性。 The results show that the compounds of the present invention represented by the compounds listed in Table 1 have a significant agonistic effect on the expression of IL-1β in THP-1 cells after PMA-induced differentiation, but on IL-1β in THP-1 def NLRP3 cells. The expression has no agonistic effect even at the highest compound tested concentration (30 μM). All the compounds tested had no activating effect on hTLR7 and hTLR8 at 100 μM. In summary, the compounds of the present invention represented by the compounds listed in Table 1 have obvious specific agonistic activity on hNLRP3 and/or its signal pathway.
实验例5:hERG实验Experimental example 5: hERG experiment
1、采用Predictor TM hERG Fluorescence Polarization Assay Kit(ThermoFisher),按照试剂盒说明测试化合物对hERG钾离子通道的抑制作用。化合物的测试浓度为1μM和10μM,结果见表2。 1. Using Predictor TM hERG Fluorescence Polarization Assay Kit (ThermoFisher), test the inhibitory effect of the compound on the hERG potassium ion channel according to the kit instructions. The test concentrations of the compounds were 1 μM and 10 μM, and the results are shown in Table 2.
表2Table 2
2、采用手动膜片钳方法测试了化合物对hERG的抑制作用。2. The manual patch clamp method was used to test the inhibitory effect of the compounds on hERG.
细胞株:过表达hERG钾离子通道HEK-293(人胚肾细胞)(科瑞斯生物)Cell line: over-expressing hERG potassium channel HEK-293 (human embryonic kidney cells) (Creece Bio)
阴性对照:含0.1%DMSO(Sigma)的细胞外液Negative control: extracellular fluid containing 0.1% DMSO (Sigma)
阳性对照:奎尼丁(aladdin)Positive control: Quinidine (aladdin)
测试化合物浓度:10μMTest compound concentration: 10μM
反应温度:22~24℃Reaction temperature: 22~24℃
数据采集:PATCHMASTER V2X60(HEKA Instruments Inc.)。Data collection: PATCHMASTER V2X60 (HEKA Instruments Inc.).
实验步骤:Experimental steps:
1)取化合物溶解于DMSO中,配制成10mM溶液。取30μL该溶液加入30mL细胞外液中,用于电生理检测。1) Dissolve the compound in DMSO and prepare a 10 mM solution. Take 30 μL of this solution and add it to 30 mL of extracellular fluid for electrophysiological testing.
2)将实验用细胞转移到一个嵌于倒置显微镜平台的细胞浴槽中,灌流细胞外液。稳定5分钟后,将膜电压钳制在-80mV。给予细胞持续2s,+20mV电压刺激,激活hERG钾通道,再复极化至-50mV,持续5s,产生外向尾电流。刺激频率为15秒/次。2) Transfer the experimental cells to a cell bath embedded in the platform of the inverted microscope, and perfuse the extracellular fluid. After stabilizing for 5 minutes, the membrane voltage was clamped to -80mV. The cells were stimulated with +20mV voltage for 2s to activate the hERG potassium channel, and then repolarized to -50mV for 5s to generate outward tail current. The stimulation frequency is 15 seconds/time.
3)灌流细胞外液(2毫升/分钟)并持续记录。电流稳定后灌流含待测化合物的细胞外液并持续记录直到化合物对hERG电流的抑制作用到达稳定状态。3) Perfuse extracellular fluid (2 ml/min) and record continuously. After the current is stable, the extracellular fluid containing the test compound is perfused and the recording is continued until the inhibitory effect of the compound on the hERG current reaches a steady state.
本实验中阳性对照奎尼丁在30μM时对hERG钾离子通道的抑制率为90.7%,说明试验系统正常。结果见表3。In this experiment, the positive control quinidine inhibited the hERG potassium channel by 90.7% at 30μM, indicating that the test system was normal. The results are shown in Table 3.
表3table 3
结果显示,表3所列化合物的IC 50>10μM,表明以表3所列化合物为代表的本发明化合物对hERG无 明显的抑制作用,导致心脏QT间期延长的可能性小。 The results show that the IC 50 of the compounds listed in Table 3> 10 μM, indicating that the compounds of the present invention represented by the compounds listed in Table 3 have no obvious inhibitory effect on hERG, and the possibility of prolonging the cardiac QT interval is small.
实验例6:CYP酶抑制试验Experimental example 6: CYP enzyme inhibition test
CYP450是药物代谢中最重要的酶系统。参与代谢的酶与药物相互作用,其中最主要的酶为CYP1A2、CYP2D6和CYP3A4。本实验使用P450-Glo TM CYP1A2 Screening System、 CYP2D6Cyan Screening Kit和 CYP3A4 Red Screening Kit,按照试剂盒说明分别测定化合物对CYP1A2、CYP2D6和CYP3A4的抑制活性。结果见表4。 CYP450 is the most important enzyme system in drug metabolism. Enzymes involved in metabolism interact with drugs, and the most important ones are CYP1A2, CYP2D6 and CYP3A4. This experiment uses P450-Glo TM CYP1A2 Screening System, CYP2D6Cyan Screening Kit and CYP3A4 Red Screening Kit, according to the kit instructions to determine the compound's inhibitory activity on CYP1A2, CYP2D6 and CYP3A4. The results are shown in Table 4.
表4Table 4
结果表明,以表4所列化合物为代表的本发明化合物对CYP1A2、CYP2D6和CYP3A4酶无明显抑制作用。The results show that the compounds of the present invention represented by the compounds listed in Table 4 have no obvious inhibitory effect on CYP1A2, CYP2D6 and CYP3A4 enzymes.
除本文中描述的那些外,根据前述描述,本发明的多种修改对本领域技术人员而言会是显而易见的。这样的修改也意图落入所附权利要求书的范围内。本申请中所引用的各参考文献(包括所有专利、专利申请、期刊文章、书籍及任何其它公开)均以其整体援引加入本文。In addition to those described herein, various modifications of the present invention will be apparent to those skilled in the art based on the foregoing description. Such modifications are also intended to fall within the scope of the appended claims. Each reference cited in this application (including all patents, patent applications, journal articles, books, and any other publications) is incorporated herein by reference in its entirety.
Claims (13)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202080071984.6A CN114555565B (en) | 2019-12-13 | 2020-07-31 | Nitrogen-containing cyclic compounds, their preparation methods and uses |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201911279540.7 | 2019-12-13 | ||
| CN201911279540 | 2019-12-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2021114691A1 true WO2021114691A1 (en) | 2021-06-17 |
Family
ID=76329456
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2020/106220 Ceased WO2021114691A1 (en) | 2019-12-13 | 2020-07-31 | Nitrogen-containing ring-fused compound, preparation method therefor and use thereof |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN114555565B (en) |
| WO (1) | WO2021114691A1 (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114269753B (en) * | 2019-09-29 | 2024-03-05 | 四川科伦博泰生物医药股份有限公司 | Nitrogen-containing bicyclic compound, pharmaceutical composition containing same, preparation method and application thereof |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101056865A (en) * | 2004-11-09 | 2007-10-17 | 霍夫曼-拉罗奇有限公司 | Aminoquinazolines compounds |
| WO2010146173A1 (en) * | 2009-06-18 | 2010-12-23 | Vereniging Voor Christelijk Hoger Onderwijs, Wetenschappelijk Onderzoek En Patientenzorg | Quinazoline derivatives as histamine h4-recept0r inhibitors for use in the treatment of inflammatory disorders |
| CN104582703A (en) * | 2012-06-08 | 2015-04-29 | 森索睿翁公司 | H4 receptor inhibitors for treating tinnitus |
| WO2018021977A1 (en) * | 2016-07-29 | 2018-02-01 | Agency For Science, Technology And Research | Glycine metabolism modulators and uses thereof |
| WO2019014402A1 (en) * | 2017-07-14 | 2019-01-17 | Innate Tumor Immunity, Inc. | Nlrp3 modulators |
| WO2019166532A1 (en) * | 2018-03-01 | 2019-09-06 | Janssen Sciences Ireland Unlimited Company | 2,4-diaminoquinazoline derivatives and medical uses thereof |
| WO2019214546A1 (en) * | 2018-05-11 | 2019-11-14 | 四川科伦博泰生物医药股份有限公司 | Fused ring compound, preparation method therefor and use thereof |
| WO2020150113A1 (en) * | 2019-01-14 | 2020-07-23 | Innate Tumor Immunity, Inc. | Substituted quinazolines as nlrp3 modulators, for use in the treatment of cancer |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1724268A4 (en) * | 2004-02-20 | 2010-04-21 | Kirin Pharma Kk | COMPOUND HAVING TGF-BETA INHIBITING ACTIVITY AND PHARMACEUTICAL COMPOSITION CONTAINING THE SAME |
-
2020
- 2020-07-31 WO PCT/CN2020/106220 patent/WO2021114691A1/en not_active Ceased
- 2020-07-31 CN CN202080071984.6A patent/CN114555565B/en active Active
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101056865A (en) * | 2004-11-09 | 2007-10-17 | 霍夫曼-拉罗奇有限公司 | Aminoquinazolines compounds |
| WO2010146173A1 (en) * | 2009-06-18 | 2010-12-23 | Vereniging Voor Christelijk Hoger Onderwijs, Wetenschappelijk Onderzoek En Patientenzorg | Quinazoline derivatives as histamine h4-recept0r inhibitors for use in the treatment of inflammatory disorders |
| CN104582703A (en) * | 2012-06-08 | 2015-04-29 | 森索睿翁公司 | H4 receptor inhibitors for treating tinnitus |
| WO2018021977A1 (en) * | 2016-07-29 | 2018-02-01 | Agency For Science, Technology And Research | Glycine metabolism modulators and uses thereof |
| WO2019014402A1 (en) * | 2017-07-14 | 2019-01-17 | Innate Tumor Immunity, Inc. | Nlrp3 modulators |
| WO2019166532A1 (en) * | 2018-03-01 | 2019-09-06 | Janssen Sciences Ireland Unlimited Company | 2,4-diaminoquinazoline derivatives and medical uses thereof |
| WO2019214546A1 (en) * | 2018-05-11 | 2019-11-14 | 四川科伦博泰生物医药股份有限公司 | Fused ring compound, preparation method therefor and use thereof |
| WO2020150113A1 (en) * | 2019-01-14 | 2020-07-23 | Innate Tumor Immunity, Inc. | Substituted quinazolines as nlrp3 modulators, for use in the treatment of cancer |
Non-Patent Citations (2)
| Title |
|---|
| DATABASE REGISTRY 8 June 2006 (2006-06-08), ANONYMOUS: "N - 2,4-Quinazolinediamine, N4,N4-dimethyl-7-(2-thienyl)- (CA INDEX NAME)", XP055820271, retrieved from STN Database accession no. 887232-89-5 * |
| SMITS ROGIER A.; LIM HERMAN D.; VAN DER MEER TIFFANY; KUHNE SEBASTIAAN; BESSEMBINDER KARIN; ZUIDERVELD OBBE P.; WIJTMANS MAIKEL; D: "Ligand based design of novel histamine H4 receptor antagonists; fragment optimization and analysis of binding kinetics", BIOORGANIC & MEDICINAL CHEMISTRY LETTERS, vol. 22, no. 1, 11 November 2011 (2011-11-11), pages 461 - 467, XP029121530, ISSN: 0960-894X, DOI: 10.1016/j.bmcl.2011.10.104 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN114555565B (en) | 2024-03-05 |
| CN114555565A (en) | 2022-05-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN115448910B (en) | A pyridazine derivative inhibitor, preparation method and application thereof | |
| CA2997051C (en) | Novel pyrazolo[3,4-d]pyrimidine compound or salt thereof | |
| CN111018861B (en) | Pyrrolopyrimidine compounds | |
| WO2020168927A1 (en) | Nitrogen-containing fused cyclic compound, preparation method therefor and use thereof | |
| JP6035423B2 (en) | Novel condensed pyrimidine compound or salt thereof | |
| JP2021512059A (en) | Degradation and use of BTK by conjugation of Bruton's tyrosine kinase (BTK) inhibitor with E3 ligase ligand | |
| CN115626919A (en) | Pyridazinyl thiazole carboxamides | |
| MX2012009541A (en) | Cyclobutane and methylcyclobutane derivatives as janus kinase inhibitors. | |
| WO2024046409A1 (en) | Heterocyclic compound, preparation method therefor, and pharmaceutical use thereof | |
| WO2016169421A1 (en) | Imidazo isoindole derivative, preparation method therefor and medical use thereof | |
| WO2020093905A1 (en) | Ring-fused compound, preparation method therefor and application thereof | |
| WO2019134707A1 (en) | Immunomodulator | |
| JP2022523477A (en) | A pharmaceutical composition for preventing or treating a pyrrolopyrimidine derivative and a protein kinase-related disease containing the derivative as an active ingredient. | |
| WO2019154294A1 (en) | Pyrazolo[1,5-a][1,3,5]triazine-2-amine derivative, preparation method therefor and medical use thereof | |
| US11267815B2 (en) | Class of amino-substituted nitrogen-containing fused ring compounds, preparation method therefor, and use thereof | |
| WO2019057112A1 (en) | 2-substituted pyrazole amino-4-substituted amino-5-pyrimidine formamide compound, composition, and application thereof | |
| TW202515544A (en) | Pharmaceutical compounds and compositions as c-kit kinase inhibitors | |
| CN109535132B (en) | 2-substituted pyrazole amino-4-substituted amino-5-pyrimidine carboxamide compounds, compositions and applications thereof | |
| CN117164583A (en) | Compound for targeting IRAK4 protein degradation and application thereof | |
| CN114555565B (en) | Nitrogen-containing cyclic compounds, their preparation methods and uses | |
| CN102421779B (en) | Dihydropteridinone derivatives, preparation method and pharmaceutical use thereof | |
| EP4653439A1 (en) | Azacycloalkane compound and pharmaceutical composition, and use thereof | |
| CN108299436B (en) | Xanthine compound and pharmaceutical composition and application thereof | |
| WO2016180334A1 (en) | Dual-site irreversible bruton's tyrosine kinase inhibitor, composition and application therefof | |
| EP4219453A1 (en) | Pyrazole compound and preparation method therefor and use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 20899042 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 20899042 Country of ref document: EP Kind code of ref document: A1 |