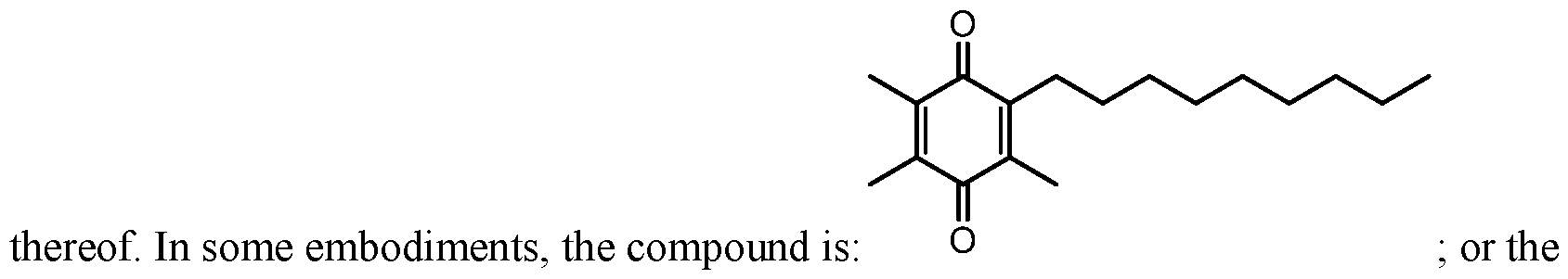WO2021077034A1 - 2,3,5-trimethyl-6-nonylcyclohexa-2,5-diene-1,4-dione for suppressing and treating hemoglobinopathy, thalassemia, sickle cell disease and other disorders - Google Patents
2,3,5-trimethyl-6-nonylcyclohexa-2,5-diene-1,4-dione for suppressing and treating hemoglobinopathy, thalassemia, sickle cell disease and other disorders Download PDFInfo
- Publication number
- WO2021077034A1 WO2021077034A1 PCT/US2020/056179 US2020056179W WO2021077034A1 WO 2021077034 A1 WO2021077034 A1 WO 2021077034A1 US 2020056179 W US2020056179 W US 2020056179W WO 2021077034 A1 WO2021077034 A1 WO 2021077034A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- thalassemia
- suppressing
- treating
- compound
- administered
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/045—Hydroxy compounds, e.g. alcohols; Salts thereof, e.g. alcoholates
- A61K31/05—Phenols
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/12—Ketones
- A61K31/122—Ketones having the oxygen directly attached to a ring, e.g. quinones, vitamin K1, anthralin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/04—Antihaemorrhagics; Procoagulants; Haemostatic agents; Antifibrinolytic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/127—Synthetic bilayered vehicles, e.g. liposomes or liposomes with cholesterol as the only non-phosphatidyl surfactant
Definitions
- Hemoglobinopathy refers to a group of blood disorders and diseases that affect red blood cells. The condition is associated with a genetic defect that results in an abnormal structure of one of the globin chains of the hemoglobin molecule. Thalassemia is also a type of inherited blood disorder, and characterized by a decrease in hemoglobin production.
- hemoglobinopathy is a structural abnormality in the globin protein itself
- thalassemia is a result of underproduction of normal globin proteins, typically through mutations in regulatory genes.
- the two conditions can overlap as some conditions that cause abnormalities in globin proteins (hemoglobinopathy) also affect their production (thalassemia).
- hemoglobinopathy also affect their production (thalassemia).
- thalassemia not all hemoglobinopathies are also thalassemias.
- Thalassemias can be divided into alpha-thalassemia (a-thalassemia) and beta- thalassemia (b-thalassemia). Hemoglobin consists of two alpha chains and two beta chains. In alpha-thalassemia, there is a decrease in the amount of alpha chains, resulting in fewer normal hemoglobin molecules. Analogously, beta-thalassemia refers to the condition wherein the amount of beta chains decrease. Both conditions can lead to microcytic anemia, resulting in insufficient red blood cell functioning, even red blood cell destruction. Patients may require multiple blood transfusions for treatment, which can result in severe health risks including iron overload.
- One well-known hemoglobinopathy is sickle-cell disease, the most common type being sickle-cell anemia.
- Sickle-cell disease is associated with rigid, sickle-like shaped hemoglobin.
- Adverse health issues associated with sickle cell disease include severe pain, anemia, swelling of the hands and feet, bacterial infections, and stroke. Further, people suffering with sickle-cell disease have a shortened life expectancy.
- Conventional treatments include antibiotics to prevent infections, high fluid intake, folic acid supplements, and pain medication. Patients also typically need to undergo multiple blood transfusions, and possibly transplants of bone marrow cells.
- U.S. Publication No. 2007/0072943 describes certain quinone compounds, and methods of treating certain mitochondrial disorders.
- U.S. Publication No. 2010/0063161 describes the compound 2,3,5-trimethyl-6-nonylcyclohexa-2,5-diene-l,4-dione, and methods for treating pervasive developmental disorders and ADHD.
- the compound is in the quinone form. In some embodiments, including any of the foregoing embodiments, the compound is in the hydroquinone form. In some embodiments, the method is for suppressing or treating hemoglobinopathy. In some embodiments, the method is for suppressing or treating thalassemia. In some embodiments, the thalassemia is a-thalassemia. In some embodiments, the thalassemia is a-thalassemia minima. In some embodiments, the thalassemia is a-thalassemia minor. In some embodiments, the thalassemia is b-thalassemia.
- the thalassemia is b- thalassemia minor. In some embodiments, the thalassemia is d-thalassemia In some embodiments, the method is for suppressing or treating sickle-cell disease. In some embodiments, the method is for suppressing or treating any combination of the foregoing diseases or disorders. In some embodiments, including any of the foregoing embodiments, the compound is administered orally. In some embodiments, including any of the foregoing embodiments, the compound is administered by injection. In some embodiments, including any of the foregoing embodiments, the compound is administered intravenously. In some embodiments, including any of the foregoing embodiments, the method is a method of suppressing the disorder. In some embodiments, including any of the foregoing embodiments, the method is a method of treating the disorder.
- any one or more of the compounds described herein, including all of the foregoing compounds can be used in a composition comprising a pharmaceutically acceptable carrier, pharmaceutically acceptable excipient, or pharmaceutically acceptable vehicle.
- the composition is formulated for internal use.
- Any one or more of the compounds described herein, including all of the foregoing compounds, can be formulated into a unit dose formulation.
- any compound in the quinone form can also be used in its reduced form (hydroquinone) when desired. That is, the compounds recited herein as cyclohexadienedione compounds (oxidized quinone) form can also be used in their benzenediol (reduced hydroquinone) form as desired.
- the compounds or compositions can either comprise the listed components or steps, or can “consist essentially of’ the listed components or steps, or can “consist of’ the listed components or steps.
- transitional phrase “comprising” or “comprises” can be replaced by the transitional phrase “consisting essentially of’ or “consists essentially of.”
- the transitional phrase “comprising” or “comprises” can be replaced, in some or any embodiments, by the transitional phrase “consisting of’ or “consists of.”
- the method contains the steps listed, and may contain other steps that do not substantially affect the condition being treated, but the method does not contain any other steps which substantially affect the condition being treated other than those steps expressly listed.
- the composition when a composition is described as ‘consisting essentially of a component, the composition may additionally contain any amount of pharmaceutically acceptable carriers, vehicles, excipients, or diluents and other such components which do not substantially affect the condition being treated.
- Figures 1A and IB provide exposure of Compound 3 (2,3,5-trimethyl-6- nonylcyclohexa-2,5-diene-l,4-dione) in control and sickle cell disease (SCD) mice.
- SCD sickle cell disease
- Figure 2 provides reduction of 12-HETE in whole blood from sickle cell disease (SCD) and control (C57NL/6) mice at 2 hours. Veh indicates vehicle control.
- Figure 3 provides reduction of 13-HODE in whole blood from sickle cell disease (SCD) and control (C57NL/6) mice at 2 hours. Veh indicates vehicle control.
- the present invention provides compounds, compositions, and methods for treating or suppressing certain disorders.
- Reference to “about” a value or parameter herein includes (and describes) variations that are directed to that value or parameter per se. For example, description referring to “about X” includes description of “X”.
- subject By “subject,” “individual,” or “patient” is meant an individual organism, preferably a vertebrate, more preferably a mammal, most preferably a human.
- Treating” a disorder with the compounds and methods discussed herein is defined as administering one or more of the compounds discussed herein, with or without additional therapeutic agents, in order to reduce or eliminate either the disorder or one or more symptoms of the disorder, or to retard the progression of the disorder or of one or more symptoms of the disorder, or to reduce the severity of the disorder or of one or more symptoms of the disorder.
- “Suppression” of a disorder with the compounds and methods discussed herein is defined as administering one or more of the compounds discussed herein, with or without additional therapeutic agents, in order to suppress the clinical manifestation of the disorder, or to suppress the manifestation of adverse symptoms of the disorder.
- treatment occurs after adverse symptoms of the disorder are manifest in a subject, while suppression occurs before adverse symptoms of the disorder are manifest in a subject. Suppression may be partial, substantially total, or total.
- genetic screening can be used to identify patients at risk of the disorder. The compounds and methods disclosed herein can then be administered to asymptomatic patients at risk of developing the clinical symptoms of the disorder, in order to suppress the appearance of any adverse symptoms.
- “Therapeutic use” of the compounds discussed herein is defined as using one or more of the compounds discussed herein to treat or suppress a disorder, as defined herein.
- a “therapeutically effective amount” of a compound is an amount of the compound, which, when administered to a subject, is sufficient to reduce or eliminate either a disorder or one or more symptoms of a disorder, or to retard the progression of a disorder or of one or more symptoms of a disorder, or to reduce the severity of a disorder or of one or more symptoms of a disorder, or to suppress the clinical manifestation of a disorder, or to suppress the manifestation of adverse symptoms of a disorder.
- a therapeutically effective amount can be given in one or more administrations.
- “Hydroquinone form” indicates the form of the compound when a two electron reduction of the quinone ring is effected, providing a net conversion of the two oxo groups to two hydroxy groups.
- the hydroquinone form of the quinone compound :
- Hemoglobinopathy includes genetic defects, diseases, and disorders related to an abnormal structure of one or more globin chains of hemoglobin.
- Thalassemia includes genetic defects, diseases, and disorders related to underproduction of globin proteins. As is known to those of skill in the art, thalassemia and hemoglobinopathy may overlap. In some embodiments, the thalassemia is an a-thalassemia. In some embodiments, the thalassemia is a b-thalassemia. In some embodiments, the thalassemia is a d-thalassemia.
- Sickle cell disease includes genetic defects, diseases, and disorders related to agene defect in the production of the hemoglobin S (HbS) protein.
- Sickle cell disease includes the common term sickle cell anemia.
- the compounds described herein can be formulated as pharmaceutical compositions by formulation with additives such as pharmaceutically acceptable excipients, pharmaceutically acceptable carriers, and pharmaceutically acceptable vehicles.
- suitable pharmaceutically acceptable excipients, carriers and vehicles include processing agents and drug delivery modifiers and enhancers, such as, in some embodiments, calcium phosphate, magnesium stearate, talc, monosaccharides, disaccharides, starch, gelatin, cellulose, methyl cellulose, sodium carboxymethyl cellulose, dextrose, hydroxypropyl-P-cyclodextrin, polyvinylpyrrolidone, low melting waxes, ion exchange resins, and the like, as well as combinations of any two or more thereof.
- a pharmaceutical composition can comprise a unit dose formulation, where the unit dose is a dose sufficient to have a therapeutic effect.
- compositions containing the compounds of the invention may be in any form suitable for the intended method of administration, including, in some embodiments, a solution, a suspension, or an emulsion.
- Liquid carriers are typically used in preparing solutions, suspensions, and emulsions.
- Liquid carriers contemplated for use in the practice of the present invention include, in some embodiments, water, saline, pharmaceutically acceptable organic solvent(s), pharmaceutically acceptable oils or fats, and the like, as well as mixtures of two or more thereof.
- the liquid carrier may contain other suitable pharmaceutically acceptable additives such as solubilizers, emulsifiers, nutrients, buffers, preservatives, suspending agents, thickening agents, viscosity regulators, stabilizers, and the like.
- suitable organic solvents include, in some embodiments, monohydric alcohols, such as ethanol, and polyhydric alcohols, such as glycols.
- Suitable oils include, in some embodiments, sesame oil, soybean oil, coconut oil, olive oil, safflower oil, cottonseed oil, and the like.
- the carrier can also be an oily ester such as ethyl oleate, isopropyl myristate, and the like.
- Compositions of the present invention may also be in the form of microparticles, microcapsules, liposomal encapsulates, and the like, as well as combinations of any two or more thereof.
- Time-release or controlled release delivery systems may be used, such as a diffusion controlled matrix system or an erodible system, as described for example in: Lee, “Diffusion-Controlled Matrix Systems”, pp. 155-198 and Ron and Langer, “Erodible Systems”, pp. 199-224, in “Treatise on Controlled Drug Delivery”, A. Kydonieus Ed., Marcel Dekker, Inc., New York 1992.
- the matrix may be, in some embodiments, a biodegradable material that can degrade spontaneously in situ and in vivo, in some embodiments, by hydrolysis or enzymatic cleavage, e.g., by proteases.
- the delivery system may be, in some embodiments, a naturally occurring or synthetic polymer or copolymer, in some embodiments, in the form of a hydrogel.
- exemplary polymers with cleavable linkages include polyesters, polyorthoesters, polyanhydrides, polysaccharides, poly(phosphoesters), polyamides, polyurethanes, poly(imidocarbonates) and poly(phosphazenes).
- the compounds of the invention may be administered enterally, orally, parenterally, sublingually, by inhalation (e.g. as mists or sprays), rectally, or topically in dosage unit formulations containing conventional nontoxic pharmaceutically acceptable carriers, adjuvants, and vehicles as desired.
- suitable modes of administration include oral, subcutaneous, transdermal, transmucosal, iontophoretic, intravenous, intraarterial, intramuscular, intraperitoneal, intranasal (e.g. via nasal mucosa), subdural, rectal, gastrointestinal, and the like, and directly to a specific or affected organ or tissue.
- Formulations for topical administration may include lotions, tinctures, creams, emulsions, ointments, sprays, gels, and the like, and may further be formulated in other suitable formulations such as sunscreens, moisturizing lotions and creams, facial gels and creams, etc.
- the active product is mixed with one or more inert excipients including, for example, water, acetone, ethanol, ethylene glycol, propylene glycol, butane 1,3 diol, isopropyl myristate, isopropyl palmitate, mineral oil, and mixtures thereof.
- Topical administration may also involve the use of transdermal administration such as transdermal patches or iontophoresis devices.
- parenteral as used herein includes subcutaneous, intravenous, intramuscular, and intrastemal injection or infusion techniques.
- the compounds are mixed with pharmaceutically acceptable carriers, adjuvants, and vehicles appropriate for the desired route of administration.
- Oral administration is a preferred route of administration, and formulations suitable for oral administration are preferred formulations.
- Topical administration is another preferred route of administration, and formulations suitable for topical administration are preferred formulations.
- the compounds described for use herein can be administered in solid form, in liquid form, in aerosol form, or in the form of tablets, pills, powder mixtures, capsules, granules, injectables, creams, solutions, suppositories, enemas, colonic irrigations, emulsions, dispersions, food premixes, and in other suitable forms.
- the compounds can also be administered in liposome formulations. Additional methods of administration are known in the art.
- compositions for topical administration can be emulsions or sterile solutions. Use may be made, as solvent or vehicle, of propylene glycol, a polyethylene glycol, vegetable oils, in particular olive oil, or injectable organic esters, in certain embodiments, ethyl oleate. These compositions can also contain adjuvants, in particular wetting, isotonizing, emulsifying, dispersing and stabilizing agents. Sterilization can be carried out in several ways, in certain embodiments, using a bacteriological filter, by radiation or by heating. They can also be prepared in the form of sterile solid compositions which can be dissolved at the time of use in sterile water or any other injectable sterile medium.
- sterile injectable aqueous or oleaginous suspensions may be formulated according to the known art using suitable dispersing or wetting agents and suspending agents.
- the sterile injectable preparation may also be a sterile injectable solution or suspension in a nontoxic parenterally acceptable diluent or solvent, in some embodiments, as a solution in propylene glycol.
- acceptable vehicles and solvents that may be employed are water, Ringer's solution, and isotonic sodium chloride solution.
- sterile, fixed oils are conventionally employed as a solvent or suspending medium.
- any bland fixed oil may be employed including synthetic mono- or diglycerides.
- fatty acids such as oleic acid find use in the preparation of injectables.
- Solid dosage forms for oral administration may include capsules, tablets, pills, powders, and granules.
- the active compound may be admixed with at least one inert diluent such as sucrose, lactose, or starch.
- Such dosage forms may also comprise additional substances other than inert diluents, e.g., lubricating agents such as magnesium stearate.
- the dosage forms may also comprise buffering agents. Tablets and pills can additionally be prepared with enteric coatings.
- Liquid dosage forms for oral administration may include pharmaceutically acceptable emulsions, solutions, suspensions, syrups, and elixirs containing inert diluents commonly used in the art, such as water.
- Such compositions may also comprise adjuvants, such as wetting agents, emulsifying and suspending agents, cyclodextrins, and sweetening, flavoring, and perfuming agents.
- the compounds of the present invention can also be administered in the form of liposomes.
- liposomes are generally derived from phospholipids or other lipid substances. Liposomes are formed by mono- or multilamellar hydrated liquid crystals that are dispersed in an aqueous medium. Any non-toxic, physiologically acceptable and metabolizable lipid capable of forming liposomes can be used.
- the present compositions in liposome form can contain, in addition to a compound of the present invention, stabilizers, preservatives, excipients, and the like.
- the preferred lipids are the phospholipids and phosphatidyl cholines (lecithins), both natural and synthetic. Methods to form liposomes are known in the art. See, for example, Prescott, Ed Methods in Cell Biology, Volume XIV, Academic Press, New York, N.W., p. 33 et seq. (1976).
- formulations of the present invention may comprise two or more compounds or compositions as described herein.
- the invention also provides articles of manufacture and kits comprising any one or more of the compounds of the invention, for use in any of the methods described herein.
- the amount of active ingredient that may be combined with the carrier materials to produce a single dosage form will vary depending upon the host to which the active ingredient is administered and the particular mode of administration. It will be understood, however, that the specific dose level for any particular individual will depend upon a variety of factors including the activity of the specific compound employed, the age, body weight, body area, body mass index (BMI), general health, sex, and diet of the patient; time of administration, route of administration, rate of excretion, or drug combination; and the type, progression, and severity of the particular disease or condition.
- the pharmaceutical unit dosage chosen may be fabricated and administered to provide a defined final concentration of drug in the targeted region of the body. The therapeutically effective amount for a given situation can be readily determined by routine experimentation and is within the skill and judgment of the ordinary clinician.
- the single or multiple dosages which can be used include an amount independently selected from about 0.1 mg/kg to about 600 mg/kg body weight, or about 1.0 mg/kg to about 500 mg/kg body weight, or about 1.0 mg/kg to about 400 mg/kg body weight, or about 1.0 mg/kg to about 300 mg/kg body weight, or about 1.0 mg/kg to about 200 mg/kg body weight, or about 1.0 mg/kg to about 100 mg/kg body weight, or about 1.0 mg/kg to about 50 mg/kg body weight, or about 1.0 mg/kg to about 30 mg/kg body weight, or about 1.0 mg/kg to about 10 mg/kg body weight, or about 10 mg/kg to about 600 mg/kg body weight, or about 10 mg/kg to about 500 mg/kg body weight, or about 10 mg/kg to about 400 mg/kg body weight, or about 10 mg/kg to about 300 mg/kg body weight, or about 10 mg/kg to about 200 mg/kg body weight, or about 10 mg/kg to about 100 mg/kg body weight, or about
- the dose is administered once, twice, three times, four times, five times, or six times. In some embodiments, the dose is administered once per day, twice per day, three times per day, or four times per day. In some embodiments, the dose is administered every hour, every two hours, every three hours, every four hours, every 6 hours, every 12 hours, or every 24 hours.
- the compounds of the invention can be administered as the sole active pharmaceutical agent, they can also be used in combination with one or more other agents used in the treatment of or suppression of the disorders described here. In some embodiments, the compound(s) of the invention are administered as the sole active pharmaceutical agent that is present in a therapeutically effective amount.
- the additional active agents may generally be employed in therapeutic amounts as indicated in the Physicians’ Desk Reference (PDR) 53rd Edition (1999), or such therapeutically useful amounts as would be known to one of ordinary skill in the art.
- the compounds of the invention and the other therapeutically active agents or prophylactically effective agents can be administered at the recommended maximum clinical dosage or at lower doses. Dosage levels of the active compounds in the compositions of the invention may be varied so as to obtain a desired response depending on the route of administration, severity of the disorder and the response of the individual.
- the therapeutic agents or prophylactic agents can be formulated as separate compositions that are given at the same time or different times, or the therapeutic agents or prophylactic agents can be given as a single composition.
- the compounds of this invention can be prepared from readily available starting materials using general methods and procedures that will be apparent to one skilled in the art in view of the disclosure provided herein. It will be appreciated that where typical or preferred process conditions (i.e., reaction temperatures, times, mole ratios of reactants, solvents, pressures, etc.) are given, other process conditions can also be used unless otherwise stated. Optimum reaction conditions may vary with the particular reactants or solvent used, but such conditions can be determined by one skilled in the art by routine optimization procedures.
- the quinone form can also be used in its reduced (hydroquinone) form when desired.
- the hydroquinone form can also be used in its oxidized (quinone) form when desired.
- the reduced (hydroquinone) form may readily be converted to the oxidized (quinone) form using methods known in the art.
- Compound 3 exposure was 10 times its 15-LO IC50 in SCD and control mice. See FIGS. 1 A and IB.
- the area under the curve (AUC) and maximum concentration (Cmax) were reduced in SCD mice compared to control mice, while Tmax and t were comparable.
- the ratio of Compound 3 in blood:plasma was nearly 1:1 in SCD and control mice.
- the lipoxygenase pathway is an inflammatory target in hemoglobinopathies including sickle cell disease. See, e.g., Owusu-Ansah et al, 2016, Transl. Res. 167(1)281-297.
- SCD Sickle Cell Disease
- 15-lipoxygenase (15- LO) contributes to activation of the endothelium and recruitment of adhesion molecules like VCAM and p-selectin. Furthermore, endothelium activation drives sickle red blood cells to aggregate causing the occlusive crisis. Thus 15-LO inhibition could provide a therapeutic benefit in sickle cell disease.
- the products of 15-LO were measured in plasma and whole blood.
- Compound 3 is dosed orally once over a dose range (low, medium, high). Endpoints related to lipoxygenase inhibition and inflammation are measured in whole blood, kidney, spleen and heart. These include 12-HETE, 13-HODE, various interleukins, vascular cell adhesion molecules, intercellular adhesion molecules, and other inflammation biomarkers. Compound 3 is also dosed repeatedly, once daily, over a dose range (low, medium, high). Endpoints related to lipoxygenase inhibition and inflammation are measured in whole blood, kidney, spleen and heart. These include 12-HETE, 13-HODE, various interleukins, vascular cell adhesion molecules, intercellular adhesion molecules, and other inflammation biomarkers. These experiments will reveal the dose at which Compound 3 maximally inhibits 15-LO and decreases inflammation.
- Assays are carried out for the amelioration of hemoglobinopathy and thalassemia.
- Test samples and solvent controls are tested for their ability to rescue hemoglobinopathy and thalassemia fibroblasts stressed by addition of L-buthionine-(S,R)-sulfoximine (BSO), or BSO plus iron (e.g. iron citrate), in a similar manner as that described in Jauslin et al, Hum. Mol. Genet. 11(24):3055 (2002), Jauslin et al., FASEB J. 17:1972-4 (2003), and International Patent Application WO 2004/003565.
- BSO L-buthionine-(S,R)-sulfoximine
- iron e.g. iron citrate
- MEM a medium enriched in amino acids and vitamins, catalog no. 1-31F24-I
- M199 Medium 199 with Earle’s Balanced Salts, without phenol red
- Fetal Calf Serum is obtained from PAA Laboratories.
- Basic fibroblast growth factor and epidermal growth factor are purchased from PeproTech. Penicillin-streptomycin-glutamine mix, L-buthionine (S,R)-sulfoximine, and insulin from bovine pancreas are purchased from Sigma. Calcein AM is purchased from Anaspec. Cell culture medium is made by combining 125 ml M199 EBS, 50 ml Fetal Calf Serum, 100 U/ml penicillin, 100 microgram/ml streptomycin, 2 mM glutamine, 10 microgram/ml insulin, 10 ng/ml EGF, and 10 ng/ml bFGF; MEM EBS is added to make the volume up to 500 ml.
- a 10 mM BSO solution is prepared by dissolving 444 mg BSO in 200 ml of medium (Invitrogen, Carlsbad, Ca.) with subsequent filter-sterilization. During the course of the experiments, this solution is stored at +4°C. The cells are grown in 10 cm tissue culture plates. Every third day, they are split at a 1 :3 ratio.
- test samples are supplied in 1.5 ml glass vials.
- the compounds are diluted with DMSO, ethanol or PBS to result in a 5 mM stock solution. Once dissolved, they are stored at -20°C.
- Test samples are screened according to the following protocol:
- a culture with hemoglobinopathy or thalassemia fibroblasts is started from a 1 ml vial with approximately 500,000 cells stored in liquid nitrogen. Cells are propagated in 10 cm cell culture dishes by splitting every third day in a ratio of 1:3 until nine plates are available. Once confluent, fibroblasts are harvested. For 54 micro titer plates (96 well-MTP) a total of 14.3 million cells (passage eight) are re-suspended in 480 ml medium, corresponding to 100 microliters medium with 3,000 cells/well. The remaining cells are distributed in 10 cm cell culture plates (500,000 cells/plate) for propagation. The plates are incubated overnight at 37°C in an atmosphere with 95% humidity and 5% C02 to allow attachment of the cells to the culture plate.
- 10% DMSO 242.5 microliters
- the test compounds are unfrozen, and 7.5 microliters of a 5 mM stock solution is dissolved in the well containing 242.5 microliters of 10% DMSO, resulting in a 150 micromolar master solution.
- Serial dilutions from the master solution are made. The period between the single dilution steps is kept as short as possible (generally less than 30 seconds). At least 4 hours after attachement into MTP, cells are then treated with the various compound dilutions.
- Plates are kept overnight in the cell culture incubator. The next day, 10 microliters of a 10 mM BSO solution are added to the wells, resulting in a 1 mM final BSO concentration. Forty-eight hours later, three plates are examined under a phase-contrast microscope to verify that the cells in the negative control (wells El-Hl) are clearly dead. The medium from all plates is discarded, and the remaining liquid is removed by gently tapping the plate inversed onto a paper towel. The plates are washed twice with lOOuL of PBS containing Calcium and Magnesium. [0064] 100 microliters of PBS +Ca +Mg containing 1.2 microM Calcein AM are then added to each well.
- the plates are incubated for 30 minutes at 37C. After that time fluorescence (excitation/emission wavelengths of 485 nm and 525 nm, respectively) is read on a Gemini fluorescence reader. Data is imported into Microsoft Excel (EXCEL is a registered trademark of Microsoft Corporation for a spreadsheet program) and ExcelFit is used to calculate the EC50 concentration for each compound.
- EXCEL is a registered trademark of Microsoft Corporation for a spreadsheet program
- the compounds are tested three times, i.e., the experiment is performed three times, the passage number of the cells increasing by one with every repetition.
- the solvents do not have a detrimental effect on the viability of non-BSO treated cells nor do they have a beneficial influence on BSO-treated fibroblasts even at the highest concentration tested (1%). None of the compounds show auto fluorescence. The viability of non-BSO treated fibroblasts is set as 100%, and the viability of the BSO- and compound-treated cells is calculated as relative to this value.
- Example 5 Evaluating Compounds in Vascular Occlusion Mouse Model
- Compound 3 is dosed repeatedly, once daily, over a dose range (low, medium, high) in a sickle cell disease mouse model implanted with a viewing chamber for assessing vaso- occlusion.
- This model is described in Wittig C et al. Geraniol Suppresses Angiogenesis by Downregulating Vascular Endothelial Growth Factor (VEGF)/VEGFR-2 Signaling.
- VEGF Vascular Endothelial Growth Factor
- Efficacy is determined by quantifying the extent of vaso-occlusion with and without compound treatment.
- Additional efficacy endpoints include the measurement of 12-HETE, 13-HODE, various interleukins, vascular cell adhesion molecules, intercellular adhesion molecules, and other inflammation biomarkers. These experiments will reveal the dose at which Compound 3 maximally inhibits 15-LO and resolves or prevents vaso-occlusion.
Landscapes
- Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Diabetes (AREA)
- Hematology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
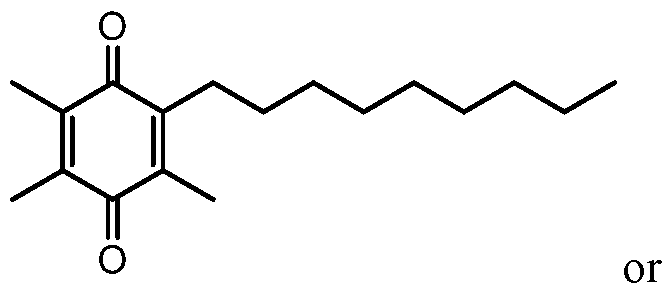
Disclosed herein are methods of treating or suppressing a disorder selected from the group consisting of hemoglobinopathy, thalassemia, α-thalassemia, α-thalassemia minima, α-thalassemia minor, β-thalassemia, β-thalassemia minor, β-thalassemia major, β-thalassemia intermedia, δ-thalassemia, sickle-cell disease, and combinations thereof, comprising administering to a subject in need thereof a therapeutically effective amount of a compound of the formula: (I) or the hydroquinone form thereof; or a solvate or hydrate thereof.
Description
2,3,5-TRIMETHYL-6-NONYLCYCLOHEXA-2,5-DIENE-l,4-DIONE FOR SUPPRESSING AND TREATING HEMOGUOBINOPATHY, THAUAS SEMIA, SICKLE CELL DISEASE AND OTHER DISORDERS
CROSS-REFERENCE TO RELATED APPLICATIONS [0001] This application claims priority to, and the benefit of, U.S. Provisional Patent Application No. 62/923,422, filed October 18, 2019, entitled 2,3,5-TRIMETHYL-6- NONYLCY CLOHEXA-2,5 -DIENE- 1 ,4-DIONE FOR SUPPRESSING AND TREATING HEMOGLOBINOPATHY, THALASSEMIA, SICKLE CELL DISEASE AND OTHER DISORDERS, the contents of which are herein incorporated by reference in their entirety for all purposes.
BACKGROUND
[0002] Hemoglobinopathy refers to a group of blood disorders and diseases that affect red blood cells. The condition is associated with a genetic defect that results in an abnormal structure of one of the globin chains of the hemoglobin molecule. Thalassemia is also a type of inherited blood disorder, and characterized by a decrease in hemoglobin production.
[0003] Whereas hemoglobinopathy is a structural abnormality in the globin protein itself, thalassemia is a result of underproduction of normal globin proteins, typically through mutations in regulatory genes. As such, the two conditions can overlap as some conditions that cause abnormalities in globin proteins (hemoglobinopathy) also affect their production (thalassemia). However, not all hemoglobinopathies are also thalassemias.
[0004] Thalassemias can be divided into alpha-thalassemia (a-thalassemia) and beta- thalassemia (b-thalassemia). Hemoglobin consists of two alpha chains and two beta chains. In alpha-thalassemia, there is a decrease in the amount of alpha chains, resulting in fewer normal hemoglobin molecules. Analogously, beta-thalassemia refers to the condition wherein the amount of beta chains decrease. Both conditions can lead to microcytic anemia, resulting in insufficient red blood cell functioning, even red blood cell destruction. Patients may require multiple blood transfusions for treatment, which can result in severe health risks including iron overload.
[0005] One well-known hemoglobinopathy is sickle-cell disease, the most common type being sickle-cell anemia. Sickle-cell disease is associated with rigid, sickle-like shaped hemoglobin. Adverse health issues associated with sickle cell disease include severe pain, anemia, swelling of the hands and feet, bacterial infections, and stroke. Further, people
suffering with sickle-cell disease have a shortened life expectancy. Conventional treatments include antibiotics to prevent infections, high fluid intake, folic acid supplements, and pain medication. Patients also typically need to undergo multiple blood transfusions, and possibly transplants of bone marrow cells.
[0006] U.S. Publication No. 2007/0072943 describes certain quinone compounds, and methods of treating certain mitochondrial disorders. U.S. Publication No. 2010/0063161 describes the compound 2,3,5-trimethyl-6-nonylcyclohexa-2,5-diene-l,4-dione, and methods for treating pervasive developmental disorders and ADHD.
[0007] Accordingly, there is a need for improved methods for treating hemoglobinopathy or thalassemia, including sickle cell disease.
BRIEF SUMMARY OF THE INVENTION [0008] In one aspect is a method of treating or suppressing a disorder selected from the group consisting of hemoglobinopathy, thalassemia, a-thalassemia, a-thalassemia minima, a-thalassemia minor, b-thalassemia, b-thalassemia minor, b-thalassemia major, b-thalassemia intermedia, d-thalassemia, sickle-cell disease, and combinations thereof, comprising administering to a subject in need thereof a therapeutically effective amount of a compound of the formula:
the hydroquinone form thereof; or a solvate or hydrate
hydroquinone form thereof. In some embodiments, including any of the foregoing embodiments, the compound is in the quinone form. In some embodiments, including any of the foregoing embodiments, the compound is in the hydroquinone form. In some embodiments, the method is for suppressing or treating hemoglobinopathy. In some embodiments, the method is for suppressing or treating thalassemia. In some embodiments, the thalassemia is a-thalassemia. In some embodiments, the thalassemia is a-thalassemia minima. In some embodiments, the thalassemia is a-thalassemia minor. In some embodiments, the thalassemia is b-thalassemia. In some embodiments, the thalassemia is b-
thalassemia minor. In some embodiments, the thalassemia is d-thalassemia In some embodiments, the method is for suppressing or treating sickle-cell disease. In some embodiments, the method is for suppressing or treating any combination of the foregoing diseases or disorders. In some embodiments, including any of the foregoing embodiments, the compound is administered orally. In some embodiments, including any of the foregoing embodiments, the compound is administered by injection. In some embodiments, including any of the foregoing embodiments, the compound is administered intravenously. In some embodiments, including any of the foregoing embodiments, the method is a method of suppressing the disorder. In some embodiments, including any of the foregoing embodiments, the method is a method of treating the disorder.
[0009] Any one or more of the compounds described herein, including all of the foregoing compounds, can be used in a composition comprising a pharmaceutically acceptable carrier, pharmaceutically acceptable excipient, or pharmaceutically acceptable vehicle. In some embodiments, the composition is formulated for internal use. Any one or more of the compounds described herein, including all of the foregoing compounds, can be formulated into a unit dose formulation.
[0010] For all the compounds, compositions, formulations and methods described herein, any compound in the quinone form can also be used in its reduced form (hydroquinone) when desired. That is, the compounds recited herein as cyclohexadienedione compounds (oxidized quinone) form can also be used in their benzenediol (reduced hydroquinone) form as desired. [0011] For all compounds, compositions, and formulations described herein, and all methods using a compound or composition or formulation described herein, the compounds or compositions can either comprise the listed components or steps, or can “consist essentially of’ the listed components or steps, or can “consist of’ the listed components or steps. That is, the transitional phrase “comprising” or “comprises” can be replaced by the transitional phrase “consisting essentially of’ or “consists essentially of.” Alternatively, the transitional phrase “comprising” or “comprises” can be replaced, in some or any embodiments, by the transitional phrase “consisting of’ or “consists of.” When a composition is described as “consisting essentially of’ the listed components, the composition contains the components listed, and may contain other components which do not substantially affect the condition being treated, but do not contain any other components which substantially affect the condition being treated other than those components expressly listed; or, if the composition does contain extra components other than those listed which substantially affect the condition being treated, the composition does not contain a sufficient concentration or
amount of the extra components to substantially affect the condition being treated. When a method is described as “consisting essentially of’ the listed steps, the method contains the steps listed, and may contain other steps that do not substantially affect the condition being treated, but the method does not contain any other steps which substantially affect the condition being treated other than those steps expressly listed. As a non-limiting specific example, when a composition is described as ‘consisting essentially of a component, the composition may additionally contain any amount of pharmaceutically acceptable carriers, vehicles, excipients, or diluents and other such components which do not substantially affect the condition being treated.
BRIEF DESCRIPTION OF THE FIGURES [0012] Figures 1A and IB provide exposure of Compound 3 (2,3,5-trimethyl-6- nonylcyclohexa-2,5-diene-l,4-dione) in control and sickle cell disease (SCD) mice.
[0013] Figure 2 provides reduction of 12-HETE in whole blood from sickle cell disease (SCD) and control (C57NL/6) mice at 2 hours. Veh indicates vehicle control.
[0014] Figure 3 provides reduction of 13-HODE in whole blood from sickle cell disease (SCD) and control (C57NL/6) mice at 2 hours. Veh indicates vehicle control.
DETAILED DESCRIPTION
[0015] The present invention provides compounds, compositions, and methods for treating or suppressing certain disorders.
[0016] The abbreviations used herein have their conventional meaning within the chemical and biological arts, unless otherwise specified.
[0017] Reference to “about” a value or parameter herein includes (and describes) variations that are directed to that value or parameter per se. For example, description referring to “about X” includes description of “X”.
[0018] As used herein, and unless otherwise specified, the terms “about” and “approximately,” when used in connection with various terms such as temperatures, doses, amounts, or weight percent of ingredients of a composition or a dosage form, mean e.g. a temperature, dose, amount, or weight percent that is recognized by those of ordinary skill in the art to provide an effect equivalent to that obtained from the specified temperature dose, amount, or weight percent. Specifically, the terms “about” and “approximately,” when used in this context, contemplate a temperature, dose, amount, or weight percent, etc. within 15%,
within 10%, within 5%, within 4%, within 3%, within 2%, within 1%, or within 0.5% of the specified temperature, dose, amount, or weight percent, etc.
[0019] The terms “a” or “an,” as used in herein means one or more, unless the context clearly dictates otherwise.
[0020] By “subject,” “individual,” or “patient” is meant an individual organism, preferably a vertebrate, more preferably a mammal, most preferably a human.
[0021] “Treating” a disorder with the compounds and methods discussed herein is defined as administering one or more of the compounds discussed herein, with or without additional therapeutic agents, in order to reduce or eliminate either the disorder or one or more symptoms of the disorder, or to retard the progression of the disorder or of one or more symptoms of the disorder, or to reduce the severity of the disorder or of one or more symptoms of the disorder. “Suppression” of a disorder with the compounds and methods discussed herein is defined as administering one or more of the compounds discussed herein, with or without additional therapeutic agents, in order to suppress the clinical manifestation of the disorder, or to suppress the manifestation of adverse symptoms of the disorder. The distinction between treatment and suppression is that treatment occurs after adverse symptoms of the disorder are manifest in a subject, while suppression occurs before adverse symptoms of the disorder are manifest in a subject. Suppression may be partial, substantially total, or total. In some embodiments, genetic screening can be used to identify patients at risk of the disorder. The compounds and methods disclosed herein can then be administered to asymptomatic patients at risk of developing the clinical symptoms of the disorder, in order to suppress the appearance of any adverse symptoms.
[0022] “Therapeutic use” of the compounds discussed herein is defined as using one or more of the compounds discussed herein to treat or suppress a disorder, as defined herein. A “therapeutically effective amount” of a compound is an amount of the compound, which, when administered to a subject, is sufficient to reduce or eliminate either a disorder or one or more symptoms of a disorder, or to retard the progression of a disorder or of one or more symptoms of a disorder, or to reduce the severity of a disorder or of one or more symptoms of a disorder, or to suppress the clinical manifestation of a disorder, or to suppress the manifestation of adverse symptoms of a disorder. A therapeutically effective amount can be given in one or more administrations.
[0023] “Hydroquinone form” indicates the form of the compound when a two electron reduction of the quinone ring is effected, providing a net conversion of the two oxo groups to
two hydroxy groups. For example, the hydroquinone form of the quinone compound:
[0024] The description of compounds herein also includes all isotopologues, in some embodiments, partially deuterated or perdeuterated analogs of all compounds herein.
[0025] “Hemoglobinopathy” includes genetic defects, diseases, and disorders related to an abnormal structure of one or more globin chains of hemoglobin.
[0026] “Thalassemia” includes genetic defects, diseases, and disorders related to underproduction of globin proteins. As is known to those of skill in the art, thalassemia and hemoglobinopathy may overlap. In some embodiments, the thalassemia is an a-thalassemia. In some embodiments, the thalassemia is a b-thalassemia. In some embodiments, the thalassemia is a d-thalassemia.
[0027] “Sickle cell disease” includes genetic defects, diseases, and disorders related to agene defect in the production of the hemoglobin S (HbS) protein. Sickle cell disease includes the common term sickle cell anemia.
Pharmaceutical formulations
[0028] The compounds described herein can be formulated as pharmaceutical compositions by formulation with additives such as pharmaceutically acceptable excipients, pharmaceutically acceptable carriers, and pharmaceutically acceptable vehicles. Suitable pharmaceutically acceptable excipients, carriers and vehicles include processing agents and drug delivery modifiers and enhancers, such as, in some embodiments, calcium phosphate, magnesium stearate, talc, monosaccharides, disaccharides, starch, gelatin, cellulose, methyl cellulose, sodium carboxymethyl cellulose, dextrose, hydroxypropyl-P-cyclodextrin, polyvinylpyrrolidone, low melting waxes, ion exchange resins, and the like, as well as combinations of any two or more thereof. Other suitable pharmaceutically acceptable excipients are described in “Remington’s Pharmaceutical Sciences,” Mack Pub. Co., New Jersey (1991), and “Remington: The Science and Practice of Pharmacy,” Lippincott Williams & Wilkins, Philadelphia, 20th edition (2003) and 21st edition (2005), incorporated herein by reference.
[0029] A pharmaceutical composition can comprise a unit dose formulation, where the unit dose is a dose sufficient to have a therapeutic effect.
[0030] Pharmaceutical compositions containing the compounds of the invention may be in any form suitable for the intended method of administration, including, in some embodiments, a solution, a suspension, or an emulsion. Liquid carriers are typically used in preparing solutions, suspensions, and emulsions. Liquid carriers contemplated for use in the practice of the present invention include, in some embodiments, water, saline, pharmaceutically acceptable organic solvent(s), pharmaceutically acceptable oils or fats, and the like, as well as mixtures of two or more thereof. The liquid carrier may contain other suitable pharmaceutically acceptable additives such as solubilizers, emulsifiers, nutrients, buffers, preservatives, suspending agents, thickening agents, viscosity regulators, stabilizers, and the like. Suitable organic solvents include, in some embodiments, monohydric alcohols, such as ethanol, and polyhydric alcohols, such as glycols. Suitable oils include, in some embodiments, sesame oil, soybean oil, coconut oil, olive oil, safflower oil, cottonseed oil, and the like. For parenteral administration, the carrier can also be an oily ester such as ethyl oleate, isopropyl myristate, and the like. Compositions of the present invention may also be in the form of microparticles, microcapsules, liposomal encapsulates, and the like, as well as combinations of any two or more thereof.
[0031] Time-release or controlled release delivery systems may be used, such as a diffusion controlled matrix system or an erodible system, as described for example in: Lee, “Diffusion-Controlled Matrix Systems”, pp. 155-198 and Ron and Langer, “Erodible Systems”, pp. 199-224, in “Treatise on Controlled Drug Delivery”, A. Kydonieus Ed., Marcel Dekker, Inc., New York 1992. The matrix may be, in some embodiments, a biodegradable material that can degrade spontaneously in situ and in vivo, in some embodiments, by hydrolysis or enzymatic cleavage, e.g., by proteases. The delivery system may be, in some embodiments, a naturally occurring or synthetic polymer or copolymer, in some embodiments, in the form of a hydrogel. Exemplary polymers with cleavable linkages include polyesters, polyorthoesters, polyanhydrides, polysaccharides, poly(phosphoesters), polyamides, polyurethanes, poly(imidocarbonates) and poly(phosphazenes).
[0032] The compounds of the invention may be administered enterally, orally, parenterally, sublingually, by inhalation (e.g. as mists or sprays), rectally, or topically in dosage unit formulations containing conventional nontoxic pharmaceutically acceptable carriers, adjuvants, and vehicles as desired. In some embodiments, suitable modes of administration include oral, subcutaneous, transdermal, transmucosal, iontophoretic,
intravenous, intraarterial, intramuscular, intraperitoneal, intranasal (e.g. via nasal mucosa), subdural, rectal, gastrointestinal, and the like, and directly to a specific or affected organ or tissue. Formulations for topical administration may include lotions, tinctures, creams, emulsions, ointments, sprays, gels, and the like, and may further be formulated in other suitable formulations such as sunscreens, moisturizing lotions and creams, facial gels and creams, etc. In these compositions, the active product is mixed with one or more inert excipients including, for example, water, acetone, ethanol, ethylene glycol, propylene glycol, butane 1,3 diol, isopropyl myristate, isopropyl palmitate, mineral oil, and mixtures thereof. Topical administration may also involve the use of transdermal administration such as transdermal patches or iontophoresis devices. The term parenteral as used herein includes subcutaneous, intravenous, intramuscular, and intrastemal injection or infusion techniques. The compounds are mixed with pharmaceutically acceptable carriers, adjuvants, and vehicles appropriate for the desired route of administration. Oral administration is a preferred route of administration, and formulations suitable for oral administration are preferred formulations. Topical administration is another preferred route of administration, and formulations suitable for topical administration are preferred formulations. The compounds described for use herein can be administered in solid form, in liquid form, in aerosol form, or in the form of tablets, pills, powder mixtures, capsules, granules, injectables, creams, solutions, suppositories, enemas, colonic irrigations, emulsions, dispersions, food premixes, and in other suitable forms. The compounds can also be administered in liposome formulations. Additional methods of administration are known in the art.
[0033] The compositions for topical administration can be emulsions or sterile solutions. Use may be made, as solvent or vehicle, of propylene glycol, a polyethylene glycol, vegetable oils, in particular olive oil, or injectable organic esters, in certain embodiments, ethyl oleate. These compositions can also contain adjuvants, in particular wetting, isotonizing, emulsifying, dispersing and stabilizing agents. Sterilization can be carried out in several ways, in certain embodiments, using a bacteriological filter, by radiation or by heating. They can also be prepared in the form of sterile solid compositions which can be dissolved at the time of use in sterile water or any other injectable sterile medium.
[0034] Injectable preparations, in some embodiments, sterile injectable aqueous or oleaginous suspensions, may be formulated according to the known art using suitable dispersing or wetting agents and suspending agents. The sterile injectable preparation may also be a sterile injectable solution or suspension in a nontoxic parenterally acceptable diluent or solvent, in some embodiments, as a solution in propylene glycol. Among the acceptable
vehicles and solvents that may be employed are water, Ringer's solution, and isotonic sodium chloride solution. In addition, sterile, fixed oils are conventionally employed as a solvent or suspending medium. For this purpose any bland fixed oil may be employed including synthetic mono- or diglycerides. In addition, fatty acids such as oleic acid find use in the preparation of injectables.
[0035] Solid dosage forms for oral administration may include capsules, tablets, pills, powders, and granules. In such solid dosage forms, the active compound may be admixed with at least one inert diluent such as sucrose, lactose, or starch. Such dosage forms may also comprise additional substances other than inert diluents, e.g., lubricating agents such as magnesium stearate. In the case of capsules, tablets, and pills, the dosage forms may also comprise buffering agents. Tablets and pills can additionally be prepared with enteric coatings.
[0036] Liquid dosage forms for oral administration may include pharmaceutically acceptable emulsions, solutions, suspensions, syrups, and elixirs containing inert diluents commonly used in the art, such as water. Such compositions may also comprise adjuvants, such as wetting agents, emulsifying and suspending agents, cyclodextrins, and sweetening, flavoring, and perfuming agents.
[0037] The compounds of the present invention can also be administered in the form of liposomes. As is known in the art, liposomes are generally derived from phospholipids or other lipid substances. Liposomes are formed by mono- or multilamellar hydrated liquid crystals that are dispersed in an aqueous medium. Any non-toxic, physiologically acceptable and metabolizable lipid capable of forming liposomes can be used. The present compositions in liposome form can contain, in addition to a compound of the present invention, stabilizers, preservatives, excipients, and the like. The preferred lipids are the phospholipids and phosphatidyl cholines (lecithins), both natural and synthetic. Methods to form liposomes are known in the art. See, for example, Prescott, Ed Methods in Cell Biology, Volume XIV, Academic Press, New York, N.W., p. 33 et seq. (1976).
[0038] The formulations of the present invention may comprise two or more compounds or compositions as described herein.
[0039] The invention also provides articles of manufacture and kits comprising any one or more of the compounds of the invention, for use in any of the methods described herein. [0040] The amount of active ingredient that may be combined with the carrier materials to produce a single dosage form will vary depending upon the host to which the active ingredient is administered and the particular mode of administration. It will be understood,
however, that the specific dose level for any particular individual will depend upon a variety of factors including the activity of the specific compound employed, the age, body weight, body area, body mass index (BMI), general health, sex, and diet of the patient; time of administration, route of administration, rate of excretion, or drug combination; and the type, progression, and severity of the particular disease or condition. The pharmaceutical unit dosage chosen may be fabricated and administered to provide a defined final concentration of drug in the targeted region of the body. The therapeutically effective amount for a given situation can be readily determined by routine experimentation and is within the skill and judgment of the ordinary clinician.
[0041] The single or multiple dosages which can be used include an amount independently selected from about 0.1 mg/kg to about 600 mg/kg body weight, or about 1.0 mg/kg to about 500 mg/kg body weight, or about 1.0 mg/kg to about 400 mg/kg body weight, or about 1.0 mg/kg to about 300 mg/kg body weight, or about 1.0 mg/kg to about 200 mg/kg body weight, or about 1.0 mg/kg to about 100 mg/kg body weight, or about 1.0 mg/kg to about 50 mg/kg body weight, or about 1.0 mg/kg to about 30 mg/kg body weight, or about 1.0 mg/kg to about 10 mg/kg body weight, or about 10 mg/kg to about 600 mg/kg body weight, or about 10 mg/kg to about 500 mg/kg body weight, or about 10 mg/kg to about 400 mg/kg body weight, or about 10 mg/kg to about 300 mg/kg body weight, or about 10 mg/kg to about 200 mg/kg body weight, or about 10 mg/kg to about 100 mg/kg body weight, or about 50 mg/kg to about 150 mg/kg body weight, or about 100 mg/kg to about 200 mg/kg body weight, or about 150 mg/kg to about 250 mg/kg body weight, or about 200 mg/kg to about 300 mg/kg body weight, or about 250 mg/kg to about 350 mg/kg body weight, or about 200 mg/kg to about 400 mg/kg body weight, or about 300 mg/kg to about 400 mg/kg body weight, or about 250 mg/kg to about 300 mg/kg body weight, or about 300 mg/kg body weight. Compounds of the present invention may be administered in a single daily dose, or the total daily dosage may be administered in divided dosage of two, three or four times daily.
[0042] Single or multiple doses can be administered. In some embodiments, the dose is administered once, twice, three times, four times, five times, or six times. In some embodiments, the dose is administered once per day, twice per day, three times per day, or four times per day. In some embodiments, the dose is administered every hour, every two hours, every three hours, every four hours, every 6 hours, every 12 hours, or every 24 hours. [0043] While the compounds of the invention can be administered as the sole active pharmaceutical agent, they can also be used in combination with one or more other agents
used in the treatment of or suppression of the disorders described here. In some embodiments, the compound(s) of the invention are administered as the sole active pharmaceutical agent that is present in a therapeutically effective amount.
[0044] When additional active agents are used in combination with the compounds of the present invention, the additional active agents may generally be employed in therapeutic amounts as indicated in the Physicians’ Desk Reference (PDR) 53rd Edition (1999), or such therapeutically useful amounts as would be known to one of ordinary skill in the art.
[0045] The compounds of the invention and the other therapeutically active agents or prophylactically effective agents can be administered at the recommended maximum clinical dosage or at lower doses. Dosage levels of the active compounds in the compositions of the invention may be varied so as to obtain a desired response depending on the route of administration, severity of the disorder and the response of the individual. When administered in combination with other therapeutic or prophylactic agents, the therapeutic agents or prophylactic agents can be formulated as separate compositions that are given at the same time or different times, or the therapeutic agents or prophylactic agents can be given as a single composition.
Preparation of Compounds of the Invention
[0046] The compounds of this invention can be prepared from readily available starting materials using general methods and procedures that will be apparent to one skilled in the art in view of the disclosure provided herein. It will be appreciated that where typical or preferred process conditions (i.e., reaction temperatures, times, mole ratios of reactants, solvents, pressures, etc.) are given, other process conditions can also be used unless otherwise stated. Optimum reaction conditions may vary with the particular reactants or solvent used, but such conditions can be determined by one skilled in the art by routine optimization procedures.
[0047] For all of the compounds and methods described herein, the quinone form can also be used in its reduced (hydroquinone) form when desired. Likewise, the hydroquinone form can also be used in its oxidized (quinone) form when desired. The reduced (hydroquinone) form may readily be converted to the oxidized (quinone) form using methods known in the art.
See, e.g., air, silica Miller et al PCT Inti Appl 2006130775 7 Dec 2006. The oxidized (quinone) form may readily be converted to the reduced hydroquinone form using methods known in the art. See, e.g., Zn, AcOH Fuchs et al EJOC 6 (2009) 833-40.
[0048] The invention is further described by the following non-limiting examples and embodiments.
[0049] Into a 50 L reactor with a thermometer and a stirrer was added 2, 3, 5-trimethyl- benzene- 1,4-diol (1) (1.39 kg, 9.1 mol) and ether (15 L) at 23 °C. It turned to a clear solution after stirring for 30 minutes. A solution of ferric chloride (5.6 kg, 34.5 mol) in water (20 L) was added dropwise over 3 h. The reaction mixture was stirred for another 2 hours at this temperature. The organic phase was separated. The drained aqueous layer was extracted with ether (3 x5 L). The combined organic phases were dried over sodium sulfate, and concentrated. The residue was diluted with dichloromethane (DCM) (1 L) and purified with silica gel chromatography (one column) to give the desired product 2 (1.27 kg, 95%). TLC (petroleum ether (PE)/ethyl acetate (EA) = 30/1). Rf (Compound 1) = 0.2. Rf (Product 2) = 0 6
[0050] Into a 50 L reactor with a thermometer and a stirrer was added 2,3,5-trimethyl- [l,4]benzoquinone (2) (780 g, 5.2 mol, 1.0 eq), decanoic acid (895 g, 5.2 mol, 1.0 eq), and acetonitrile (15 L). It turned to a clear solution after stirring at room temperature for 30 minutes. Silver nitrate (882 g, 5.2 mol, 1.0 eq) was added in one portion. The reaction mixture was heated up to 75 °C. A solution of potassium persulfate (1.54 kg, 5.7 mol, 1.1 eq) in water (30 L) was added dropwise over 2 hours. After the addition, the reaction was stirred for additional 3 hours at 75 °C. The solution was cooled to room temperature. The aqueous layer drained into 15 L of water, which was extracted with ethyl acetate (3 x 5 L). The combined organic phases were dried over sodium sulfate, filtered. The filtrate was concentrated to give a yellow residue, which was crystallized with hot methanol (800 mL). The solid was filtered and washed with small amount of methanol and ether, and dried in
vacuo to afford 2,3,5-trimethyl-6-nonylcyclohexa-2,5-diene-l,4-dione (3) (447 g) as a yellow crystal. The filtrate was concentrated and purified by flash column chromatography (petroleum ether : ethyl acetate, 100:1) to afford additional 155 g of 2,3,5-trimethyl-6- nonylcyclohexa-2,5-diene-l,4-dione (3) (total amount: 602 g, 42%). TLC (PE/EA = 30/1). Rf (Compound 2) = 0.5. Rr (Product 3) = 0.6. ¾ NMR (400 MHz, CDCb) d 2.46 (s, 2H), 2.02- 2.01(m, 9H), 1.35-1.26 (m, 22H), 0.88-0.86 (m, 5H).
Example 2. Pharmacokinetics in Sickle Cell Disease Mice
[0051] Compound 3 was evaluated in the Townes Sickle Cell Disease mouse model (Ryan et al, 1997, Science 278:873-6) and in control mice (C57BL/6). Compound 3 exposure was measured following a 300 mg/kg PO dose in SCD (n=32) and control (n=32) mice. Blood and plasma samples were obtained and frozen. Stable, isotopically labelled standards were added to samples after thawing. Liquid-liquid extraction was performed to extract and fractionate polar and non-polar metabolites. Extracts were analyzed using reverse-phase and HILIC liquid chromatography mass spectrometry.
[0052] Compound 3 exposure was 10 times its 15-LO IC50 in SCD and control mice. See FIGS. 1 A and IB. The area under the curve (AUC) and maximum concentration (Cmax) were reduced in SCD mice compared to control mice, while Tmax and t were comparable. The ratio of Compound 3 in blood:plasma was nearly 1:1 in SCD and control mice.
Example 3. Lipoxygenase Activity
[0053] The lipoxygenase pathway is an inflammatory target in hemoglobinopathies including sickle cell disease. See, e.g., Owusu-Ansah et al, 2016, Transl. Res. 167(1)281-297. One of the hallmarks of Sickle Cell Disease (SCD) is vaso-oclusive pain crises. 15-lipoxygenase (15- LO) contributes to activation of the endothelium and recruitment of adhesion molecules like VCAM and p-selectin. Furthermore, endothelium activation drives sickle red blood cells to aggregate causing the occlusive crisis. Thus 15-LO inhibition could provide a therapeutic benefit in sickle cell disease. To confirm lipoxygenase activity, the products of 15-LO were measured in plasma and whole blood.
[0054] 15-LO produces 12-hydroxy eicosatetraenoic acid (12-HETE) when arachidonic acid is its substrate. 12-HETE enhances red blood cell-endothelial cell adhesion and is significantly elevated in SCD animals. Compound 3 was evaluated in whole blood from SCD and control mice. Compound 3 significantly reduced 12-HETE in whole blood from both SCD and control mice. See FIG. 2.
[0055] 15-LO produces 13-hydroxy octadecadienoic acid (13-HODE) when linoleic acid is its substrate. 13-HODE damages cell membranes and is significantly elevated in SCD animals. Compound 3 was evaluated in whole blood from SCD and control mice. Compound 3 significantly reduced 13-HODE in whole blood from both SCD and control mice. See FIG. 3. [0056] Compound 3 is dosed orally once over a dose range (low, medium, high). Endpoints related to lipoxygenase inhibition and inflammation are measured in whole blood, kidney, spleen and heart. These include 12-HETE, 13-HODE, various interleukins, vascular cell adhesion molecules, intercellular adhesion molecules, and other inflammation biomarkers. Compound 3 is also dosed repeatedly, once daily, over a dose range (low, medium, high). Endpoints related to lipoxygenase inhibition and inflammation are measured in whole blood, kidney, spleen and heart. These include 12-HETE, 13-HODE, various interleukins, vascular cell adhesion molecules, intercellular adhesion molecules, and other inflammation biomarkers. These experiments will reveal the dose at which Compound 3 maximally inhibits 15-LO and decreases inflammation.
Example 4. Evaluating Compounds in Human Dermal Fibroblasts from Hemoglobinopathy and Thalassemia Patients
[0057] Assays are carried out for the amelioration of hemoglobinopathy and thalassemia.
Test samples and solvent controls are tested for their ability to rescue hemoglobinopathy and thalassemia fibroblasts stressed by addition of L-buthionine-(S,R)-sulfoximine (BSO), or BSO plus iron (e.g. iron citrate), in a similar manner as that described in Jauslin et al, Hum. Mol. Genet. 11(24):3055 (2002), Jauslin et al., FASEB J. 17:1972-4 (2003), and International Patent Application WO 2004/003565. Human dermal fibroblasts from hemoglobinopathy and thalassemia patients are more sensitive than fibroblast cells from a healthy patient to inhibition of the de novo synthesis of glutathione (GSH) with L-buthionine-(S,R)- sulfoximine (BSO), a specific inhibitor of GSH synthetase (Jauslin et al, Hum. Mol. Genet. 11(24):3055 (2002)). This specific BSO-mediated cell death is prevented by administration of compounds described herein.
[0058] MEM (a medium enriched in amino acids and vitamins, catalog no. 1-31F24-I) and Medium 199 (M199, catalog no. 1-21F22-I) with Earle’s Balanced Salts, without phenol red, are purchased from Bioconcept. Fetal Calf Serum is obtained from PAA Laboratories.
Basic fibroblast growth factor and epidermal growth factor are purchased from PeproTech. Penicillin-streptomycin-glutamine mix, L-buthionine (S,R)-sulfoximine, and insulin from bovine pancreas are purchased from Sigma. Calcein AM is purchased from Anaspec. Cell
culture medium is made by combining 125 ml M199 EBS, 50 ml Fetal Calf Serum, 100 U/ml penicillin, 100 microgram/ml streptomycin, 2 mM glutamine, 10 microgram/ml insulin, 10 ng/ml EGF, and 10 ng/ml bFGF; MEM EBS is added to make the volume up to 500 ml. A 10 mM BSO solution is prepared by dissolving 444 mg BSO in 200 ml of medium (Invitrogen, Carlsbad, Ca.) with subsequent filter-sterilization. During the course of the experiments, this solution is stored at +4°C. The cells are grown in 10 cm tissue culture plates. Every third day, they are split at a 1 :3 ratio.
[0059] The test samples are supplied in 1.5 ml glass vials. The compounds are diluted with DMSO, ethanol or PBS to result in a 5 mM stock solution. Once dissolved, they are stored at -20°C.
[0060] Test samples are screened according to the following protocol:
[0061] A culture with hemoglobinopathy or thalassemia fibroblasts is started from a 1 ml vial with approximately 500,000 cells stored in liquid nitrogen. Cells are propagated in 10 cm cell culture dishes by splitting every third day in a ratio of 1:3 until nine plates are available. Once confluent, fibroblasts are harvested. For 54 micro titer plates (96 well-MTP) a total of 14.3 million cells (passage eight) are re-suspended in 480 ml medium, corresponding to 100 microliters medium with 3,000 cells/well. The remaining cells are distributed in 10 cm cell culture plates (500,000 cells/plate) for propagation. The plates are incubated overnight at 37°C in an atmosphere with 95% humidity and 5% C02 to allow attachment of the cells to the culture plate.
[0062] 10% DMSO (242.5 microliters) is added to a well of the microtiter plate. The test compounds are unfrozen, and 7.5 microliters of a 5 mM stock solution is dissolved in the well containing 242.5 microliters of 10% DMSO, resulting in a 150 micromolar master solution. Serial dilutions from the master solution are made. The period between the single dilution steps is kept as short as possible (generally less than 30 seconds). At least 4 hours after attachement into MTP, cells are then treated with the various compound dilutions.
[0063] Plates are kept overnight in the cell culture incubator. The next day, 10 microliters of a 10 mM BSO solution are added to the wells, resulting in a 1 mM final BSO concentration. Forty-eight hours later, three plates are examined under a phase-contrast microscope to verify that the cells in the negative control (wells El-Hl) are clearly dead. The medium from all plates is discarded, and the remaining liquid is removed by gently tapping the plate inversed onto a paper towel. The plates are washed twice with lOOuL of PBS containing Calcium and Magnesium.
[0064] 100 microliters of PBS +Ca +Mg containing 1.2 microM Calcein AM are then added to each well. The plates are incubated for 30 minutes at 37C. After that time fluorescence (excitation/emission wavelengths of 485 nm and 525 nm, respectively) is read on a Gemini fluorescence reader. Data is imported into Microsoft Excel (EXCEL is a registered trademark of Microsoft Corporation for a spreadsheet program) and ExcelFit is used to calculate the EC50 concentration for each compound.
[0065] The compounds are tested three times, i.e., the experiment is performed three times, the passage number of the cells increasing by one with every repetition.
[0066] The solvents (DMSO, ethanol, PBS) do not have a detrimental effect on the viability of non-BSO treated cells nor do they have a beneficial influence on BSO-treated fibroblasts even at the highest concentration tested (1%). None of the compounds show auto fluorescence. The viability of non-BSO treated fibroblasts is set as 100%, and the viability of the BSO- and compound-treated cells is calculated as relative to this value.
[0067] Compounds described herein are found to rescue fibroblast cells from hemoglobinopathy and thalassemia patients from BSO-induced oxidative stress.
Example 5. Evaluating Compounds in Vascular Occlusion Mouse Model [0068] Compound 3 is dosed repeatedly, once daily, over a dose range (low, medium, high) in a sickle cell disease mouse model implanted with a viewing chamber for assessing vaso- occlusion. This model is described in Wittig C et al. Geraniol Suppresses Angiogenesis by Downregulating Vascular Endothelial Growth Factor (VEGF)/VEGFR-2 Signaling. PLoS ONE 10(7): e0131946. doi:10.1371/joumal.pone.0131946. Efficacy is determined by quantifying the extent of vaso-occlusion with and without compound treatment. Additional efficacy endpoints include the measurement of 12-HETE, 13-HODE, various interleukins, vascular cell adhesion molecules, intercellular adhesion molecules, and other inflammation biomarkers. These experiments will reveal the dose at which Compound 3 maximally inhibits 15-LO and resolves or prevents vaso-occlusion.
[0069] The disclosures of all publications, patents, patent applications and published patent applications referred to herein by an identifying citation are hereby incorporated herein by reference in their entirety.
[0070] Although the foregoing invention has been described in some detail by way of illustration and example for purposes of clarity of understanding, it is apparent to those
skilled in the art that certain minor changes and modifications will be practiced. Therefore, the description and examples should not be construed as limiting the scope of the invention.
Claims
1. A method of treating or suppressing a disorder selected from the group consisting of hemoglobinopathy, thalassemia, a-thalassemia, a-thalassemia minima, a-thalassemia minor, b-thalassemia, b-thalassemia minor, b-thalassemia major, b-thalassemia intermedia, d- thalassemia, and sickle-cell disease, comprising administering to a subject in need thereof a therapeutically effective amount of a compound of the formula:
or the hydroquinone form thereof; or a solvate or hydrate thereof.
2. The method of claim 1, wherein the compound is not a solvate or hydrate.
3. The method of claim 1 or 2, wherein the compound is in the quinone form.
4. The method of claim 1 or 2, wherein the compound is in the hydroquinone form.
5. The method of any one of claims 1-4, wherein the method is for treating or suppressing hemoglobinopathy.
6. The method of any one of claims 1-4, wherein the method is for treating or suppressing thalassemia.
7. The method of any one of claims 1-4, wherein the method is for treating or suppressing a-thalassemia.
8. The method of any one of claims 1-4, wherein the method is for treating or suppressing a-thalassemia minima.
9. The method of any one of claims 1-4, wherein the method is for treating or suppressing a-thalassemia minor.
10. The method of any one of claims 1-4, wherein the method is for treating or suppressing b-thalassemia.
11. The method of any one of claims 1 -4, wherein the method is for treating or suppressing b-thalassemia minor.
12. The method of any one of claims 1-4, wherein the method is for treating or suppressing b-thalassemia major.
13. The method of any one of claims 1-4, wherein the method is for treating or suppressing b-thalassemia intermedia.
14. The method of any one of claims 1-4, wherein the method is for treating or suppressing d-thalassemia.
15. The method of any one of claims 1-4, wherein the method is for treating or suppressing sickle-cell disease.
16. The method of any one of claims 1-15, wherein the method is for treating the disorder.
17. The method of any one of claims 1-15, wherein the method is for suppressing the disorder.
18. The method of any one of claims 1-17, wherein the compound is administered orally.
19. The method of any one of claims 1-17, wherein the compound is administered intravenously.
20. The method of any one of claims 1-19, wherein the compound is provided in a pharmaceutical composition.
21. The method of claim 20, wherein the pharmaceutical composition includes a pharmaceutically acceptable carrier.
22. The method of any one of claims 1-17, wherein the compound is administered in solid form, in liquid form, or in aerosol form.
23. The method of any one of claims 1-17, wherein the compound is administered in the form of a tablet, pill, powder mixture, capsule, granule, injectable, cream, solution, suppository, enema, colonic irrigation, emulsion, dispersion, food premix, or combinations thereof.
24. The method of any one of claims 1-17, wherein the compound is administered in a liposome formulation.
25. A pharmaceutical composition suitable for use in of treating or suppressing a disorder selected from the group consisting of hemoglobinopathy, thalassemia, a-thalassemia, a-thalassemia minima, a-thalassemia minor, b-thalassemia, b-thalassemia minor, b- thalassemia major, b-thalassemia intermedia, d-thalassemia, sickle-cell disease, wherein the composition comprises a therapeutically effective amount of a compound of the formula:
or the hydroquinone form thereof; or a solvate or hydrate thereof.
26. The pharmaceutical composition of claim 25 further comprising a pharmaceutical acceptable carrier.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201962923422P | 2019-10-18 | 2019-10-18 | |
| US62/923,422 | 2019-10-18 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2021077034A1 true WO2021077034A1 (en) | 2021-04-22 |
Family
ID=73139463
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2020/056179 Ceased WO2021077034A1 (en) | 2019-10-18 | 2020-10-16 | 2,3,5-trimethyl-6-nonylcyclohexa-2,5-diene-1,4-dione for suppressing and treating hemoglobinopathy, thalassemia, sickle cell disease and other disorders |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2021077034A1 (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11560364B2 (en) | 2015-12-16 | 2023-01-24 | Ptc Therapeutics, Inc. | Methods for enriching alpha-tocotrienol from mixed tocol compositions |
| US11667596B2 (en) | 2018-10-17 | 2023-06-06 | Ptc Therapeutics, Inc. | 2,3,5-trimethyl-6-nonylcyclohexa-2,5-diene-1,4-dione for suppressing and treating alpha-synucleinopathies, tauopathies, and other disorders |
| US11680034B2 (en) | 2015-12-17 | 2023-06-20 | Ptc Therapeutics, Inc. | Fluoroalkyl, fluoroalkoxy, phenoxy, heteroaryloxy, alkoxy, and amine 1,4-benzoquinone derivatives for treatment of oxidative stress disorders |
| US11786486B2 (en) | 2021-07-08 | 2023-10-17 | Ptc Therapeutics, Inc. | Pharmaceutical compositions comprising 2,3,5-trimethyl-6-nonylcyclohexa-2,5-diene-1,4-dione |
| US11840497B2 (en) | 2007-11-06 | 2023-12-12 | Ptc Therapeutics, Inc. | 4-(p-quinonyl)-2-hydroxybutanamide derivatives for treatment of mitochondrial diseases |
| WO2024050725A1 (en) | 2022-09-07 | 2024-03-14 | Ptc Therapeutics, Inc. | Methods of producing 2, 3, 5, 6-alkyl-1, 4-benzoquinones |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004003565A2 (en) | 2002-07-01 | 2004-01-08 | Santhera Pharmaceuticals (Schweiz) Gmbh | A screening method and compounds for treating friedreich ataxia |
| WO2006130775A2 (en) | 2005-06-01 | 2006-12-07 | Edison Pharmaceuticals, Inc. | Redox-active therapeutics for treatment of mitochondrial diseases and other conditions and modulation of energy biomakers |
| US20070072943A1 (en) | 2005-09-15 | 2007-03-29 | Miller Guy M | Tail variants of redox-active therapeutics for treatment of mitochondrial diseases and other conditions and modulation of energy biomarkers |
| US20100063161A1 (en) | 2008-09-10 | 2010-03-11 | Miller Guy M | Treatment of pervasive developmental disorders with redox-active therapeutics |
| WO2010045220A1 (en) * | 2008-10-14 | 2010-04-22 | Edison Pharmaceuticals, Inc. | Treatment of oxidative stress disorders including contrast nephropathy, radiation damage and disruptions in the function of red cells |
| WO2012170773A1 (en) * | 2011-06-08 | 2012-12-13 | Edison Pharmaceuticals, Inc. | Adjunctive therapy for the treatment of mitochondrial disorders with quinones and naphthoquinones |
| WO2018191732A1 (en) * | 2017-04-14 | 2018-10-18 | Bioelectron Technology Corporation | Methods and compositions for treatment of inflammation and oxidative stress |
| WO2020081879A2 (en) * | 2018-10-17 | 2020-04-23 | Ptc Therapeutics, Inc. | 2,3,5-TRIMETHYL-6-NONYLCYCLOHEXA-2,5-DIENE-1,4-DIONE FOR SUPPRESSING AND TREATING α-SYNUCLEINOPATHIES, TAUOPATHIES, AND OTHER DISORDERS |
-
2020
- 2020-10-16 WO PCT/US2020/056179 patent/WO2021077034A1/en not_active Ceased
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004003565A2 (en) | 2002-07-01 | 2004-01-08 | Santhera Pharmaceuticals (Schweiz) Gmbh | A screening method and compounds for treating friedreich ataxia |
| WO2006130775A2 (en) | 2005-06-01 | 2006-12-07 | Edison Pharmaceuticals, Inc. | Redox-active therapeutics for treatment of mitochondrial diseases and other conditions and modulation of energy biomakers |
| US20070072943A1 (en) | 2005-09-15 | 2007-03-29 | Miller Guy M | Tail variants of redox-active therapeutics for treatment of mitochondrial diseases and other conditions and modulation of energy biomarkers |
| US20100063161A1 (en) | 2008-09-10 | 2010-03-11 | Miller Guy M | Treatment of pervasive developmental disorders with redox-active therapeutics |
| WO2010045220A1 (en) * | 2008-10-14 | 2010-04-22 | Edison Pharmaceuticals, Inc. | Treatment of oxidative stress disorders including contrast nephropathy, radiation damage and disruptions in the function of red cells |
| WO2012170773A1 (en) * | 2011-06-08 | 2012-12-13 | Edison Pharmaceuticals, Inc. | Adjunctive therapy for the treatment of mitochondrial disorders with quinones and naphthoquinones |
| WO2018191732A1 (en) * | 2017-04-14 | 2018-10-18 | Bioelectron Technology Corporation | Methods and compositions for treatment of inflammation and oxidative stress |
| WO2020081879A2 (en) * | 2018-10-17 | 2020-04-23 | Ptc Therapeutics, Inc. | 2,3,5-TRIMETHYL-6-NONYLCYCLOHEXA-2,5-DIENE-1,4-DIONE FOR SUPPRESSING AND TREATING α-SYNUCLEINOPATHIES, TAUOPATHIES, AND OTHER DISORDERS |
Non-Patent Citations (14)
| Title |
|---|
| "Methods in Cell Biology", vol. XIV, 1976, ACADEMIC PRESS, pages: 33 |
| "Physicians' Desk Reference (PDR", 1999 |
| "Remington's Pharmaceutical Sciences", 1991, MACK PUB. CO. |
| "Treatise on Controlled Drug Delivery", 1992, MARCEL DEKKER, INC. |
| FUCHS ET AL., EJOC, vol. 6, 2009, pages 833 - 40 |
| JAUSLIN ET AL., FASEB J, vol. 17, 2003, pages 1972 - 4 |
| JAUSLIN ET AL., HUM. MOL. GENET., vol. 11, no. 24, 2002, pages 3055 |
| KALPRAVIDH RUCHANEEKORN W ET AL: "Effect of coenzyme Q(10) as an antioxidant in beta-thalassemia/Hb E patients", BIOFACTORS, OXFORD UNIVERSITY PRESS, OXFORD, GB, vol. 25, no. 1-4, 1 January 2005 (2005-01-01), pages 225 - 234, XP002674101, ISSN: 0951-6433, DOI: 10.1002/BIOF.5520250128 * |
| LEE, DIFFUSION-CONTROLLED MATRIX SYSTEMS, pages 155 - 198 |
| OWUSU-ANSAH AMMA ET AL: "Inflammatory targets of therapy in sickle cell disease", TRANSLATIONAL RESEARCH, vol. 167, no. 1, 1 January 2016 (2016-01-01), pages 281 - 297, XP029343534, ISSN: 1931-5244, DOI: 10.1016/J.TRSL.2015.07.001 * |
| OWUSU-ANSAH ET AL., TRANSL. RES., vol. 167, no. 1, 2016, pages 281 - 297 |
| RONLANGER, ERODIBLE SYSTEMS, pages 199 - 224 |
| RYAN ET AL., SCIENCE, vol. 278, 1997, pages 873 - 6 |
| WITTIG C ET AL.: "Geraniol Suppresses Angiogenesis by Downregulating Vascular Endothelial Growth Factor (VEGF)NEGFR-2 Signaling", PLOS ONE, vol. 10, no. 7, pages e0131946 |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11840497B2 (en) | 2007-11-06 | 2023-12-12 | Ptc Therapeutics, Inc. | 4-(p-quinonyl)-2-hydroxybutanamide derivatives for treatment of mitochondrial diseases |
| US11560364B2 (en) | 2015-12-16 | 2023-01-24 | Ptc Therapeutics, Inc. | Methods for enriching alpha-tocotrienol from mixed tocol compositions |
| US11680034B2 (en) | 2015-12-17 | 2023-06-20 | Ptc Therapeutics, Inc. | Fluoroalkyl, fluoroalkoxy, phenoxy, heteroaryloxy, alkoxy, and amine 1,4-benzoquinone derivatives for treatment of oxidative stress disorders |
| US11667596B2 (en) | 2018-10-17 | 2023-06-06 | Ptc Therapeutics, Inc. | 2,3,5-trimethyl-6-nonylcyclohexa-2,5-diene-1,4-dione for suppressing and treating alpha-synucleinopathies, tauopathies, and other disorders |
| US11746077B2 (en) | 2018-10-17 | 2023-09-05 | Ptc Therapeutics, Inc. | 2,3,5-trimethyl-6-nonylcyclohexa-2,5-diene-1,4-dione for suppressing and treating alpha-synucleinopathies, tauopathies, and other disorders |
| US11786486B2 (en) | 2021-07-08 | 2023-10-17 | Ptc Therapeutics, Inc. | Pharmaceutical compositions comprising 2,3,5-trimethyl-6-nonylcyclohexa-2,5-diene-1,4-dione |
| WO2024050725A1 (en) | 2022-09-07 | 2024-03-14 | Ptc Therapeutics, Inc. | Methods of producing 2, 3, 5, 6-alkyl-1, 4-benzoquinones |
| WO2024054871A1 (en) | 2022-09-07 | 2024-03-14 | Ptc Therapeutics, Inc. | Methods of producing 2,3,5,6-alkyl-1,4-benzoquinones |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2021077034A1 (en) | 2,3,5-trimethyl-6-nonylcyclohexa-2,5-diene-1,4-dione for suppressing and treating hemoglobinopathy, thalassemia, sickle cell disease and other disorders | |
| JP5881266B2 (en) | Methods and compositions for treating metabolic syndrome | |
| KR101929074B1 (en) | Anxiolytic effect of pterostilbene | |
| JP4236464B2 (en) | Combination of fenofibrate and coenzyme Q10 for the treatment of endothelial dysfunction | |
| KR102054038B1 (en) | Methods and Compositions For Treating Leukemia | |
| US10344001B2 (en) | Azolylacryloyl derivatives as therapeutic agents for sickle cell disease | |
| EP3568138B1 (en) | Compositions and methods for the treatment of myelin related and inflammation related diseases or disorders | |
| KR102106365B1 (en) | Geranyl geranyl acetone analogs and uses thereof | |
| US5710175A (en) | Growth stimulating factors | |
| KR20040049248A (en) | Anti-tumor pharmaceutical composition comprising n-vanillyl fatty acid amide | |
| AU710479B2 (en) | Antiglucocorticoid medicament | |
| EP0815858B1 (en) | Pharmaceutical composition for the treatment of leukemia containing 9-cis-retinoic acid-alpha-tocopherol ester | |
| US6806293B1 (en) | Use of pheromone compounds having MAP kinase modulating activity | |
| AU716817B2 (en) | (o-acyl-p-N-acylamino-phenyl)-O-phosphoethanolamines | |
| EP2557076A1 (en) | Anti-leishmania compound and anti-leishmania drug | |
| JP2004083465A (en) | Synostatin analogs as SMase inhibitors | |
| NZ515138A (en) | Vitamin D analogues and their pharmaceutical use | |
| CA3060365A1 (en) | Amide derivatives of polycaffeoylquinic acids, process for producing same and uses thereof | |
| CA2110336A1 (en) | Nonatetraenoic acid derivative | |
| US20260007640A1 (en) | Compositions for the treatment of inflammatory disorders | |
| CN101607954B (en) | Danshensu derivative, preparation method thereof, and application thereof in pharmacy | |
| JP2756941B2 (en) | Use of 2-chloro-3-arylamino-1,4-naphthoquinone derivatives as platelet aggregation inhibitors | |
| US20120108667A1 (en) | Hydroxy ceramides and analogs thereof and their use for preventing or treating cancer | |
| CA2880399C (en) | Use of bimatoprost free acid administered to the skin to reduce adipose tissue | |
| EP1779892A1 (en) | Antiprotozoal agent |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 20803396 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 20803396 Country of ref document: EP Kind code of ref document: A1 |