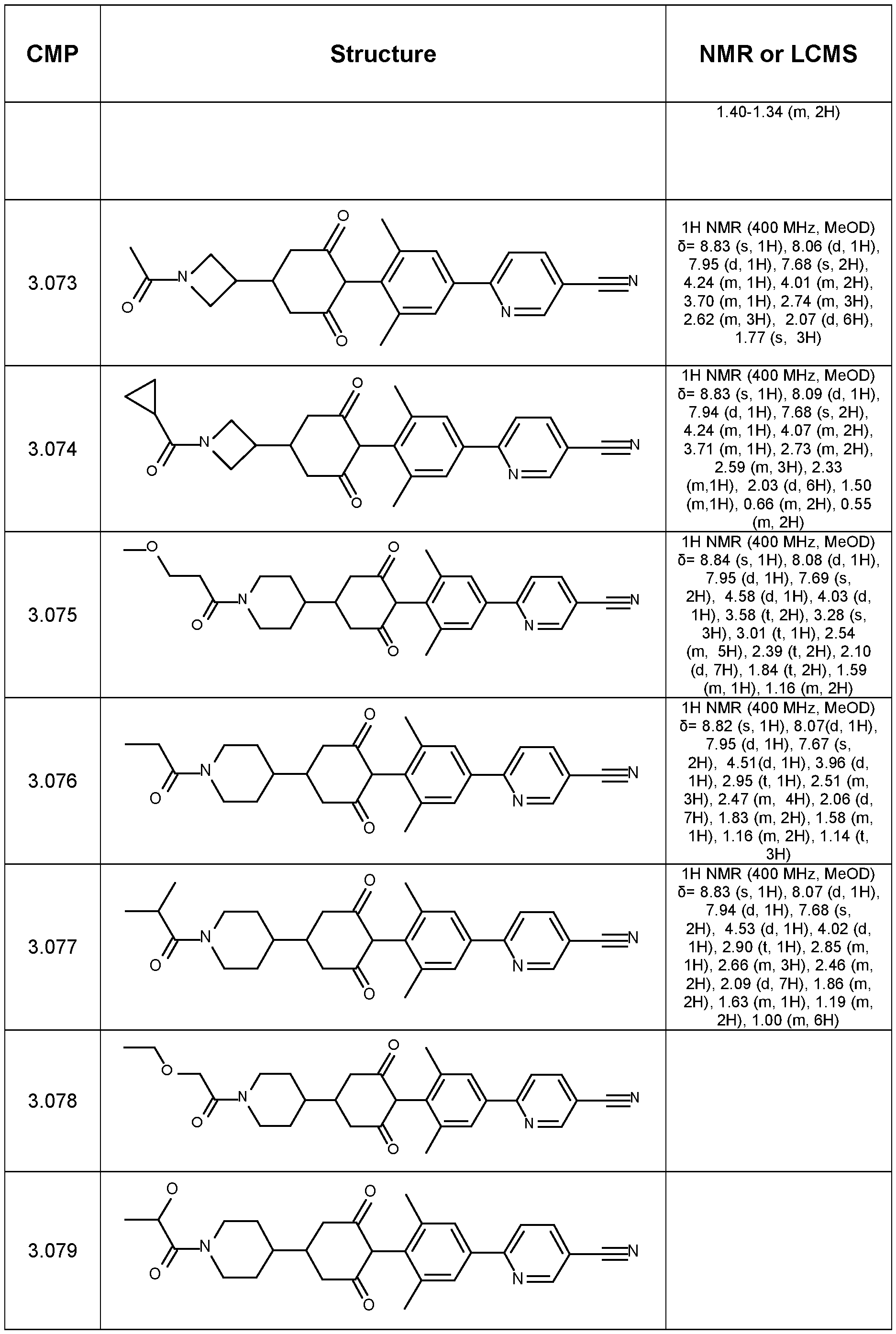
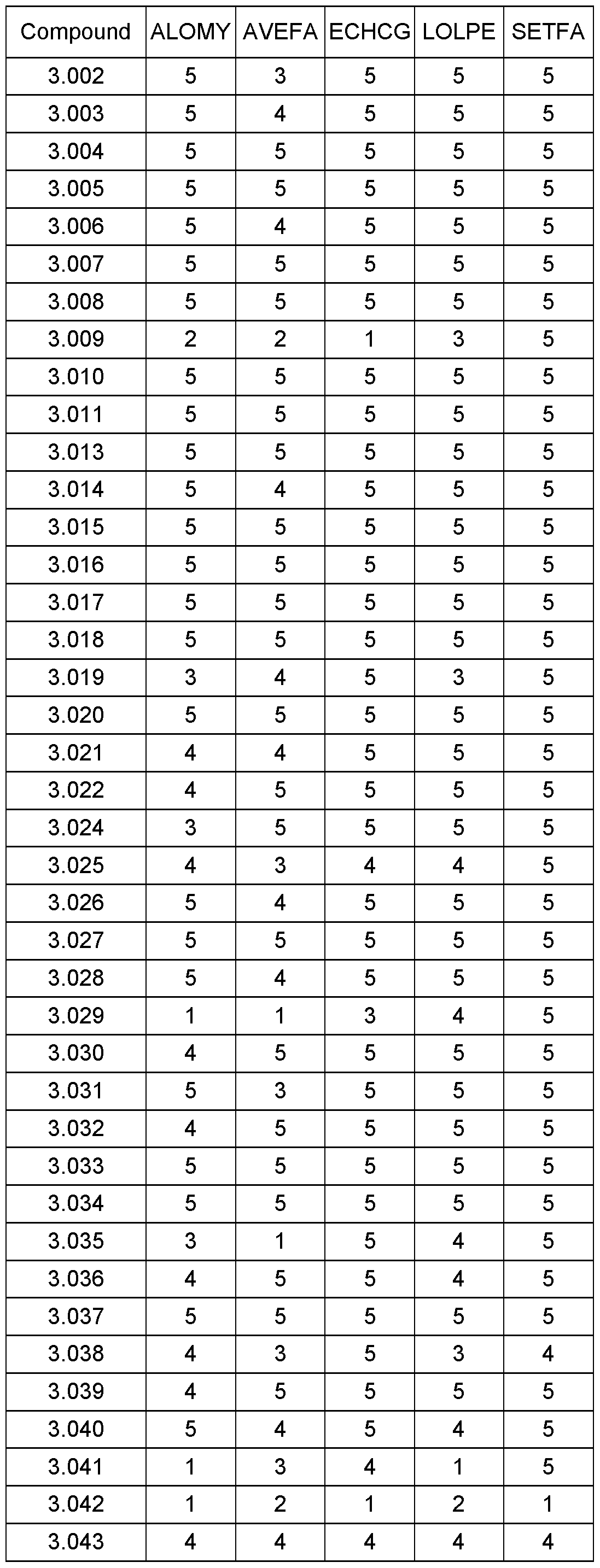
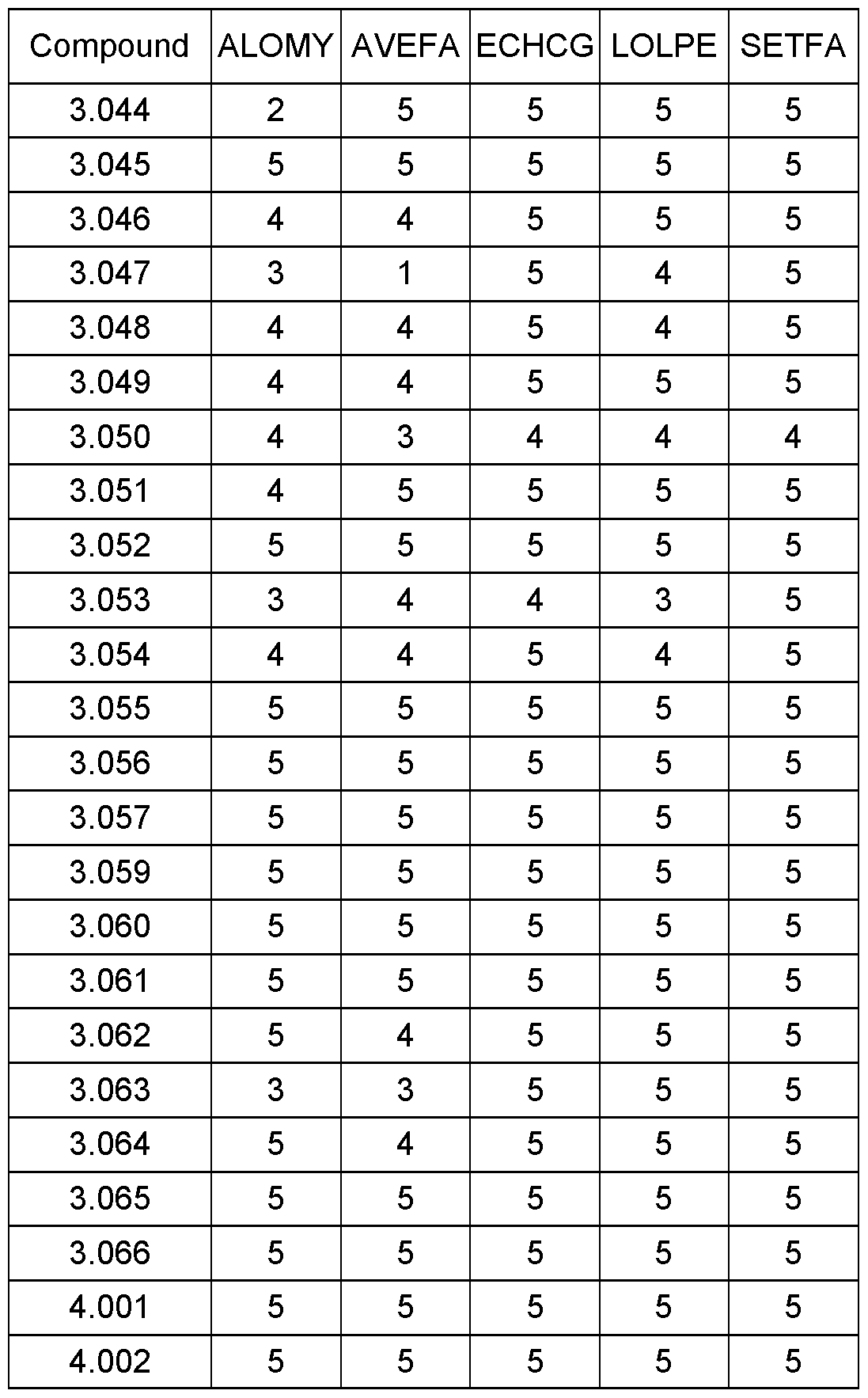
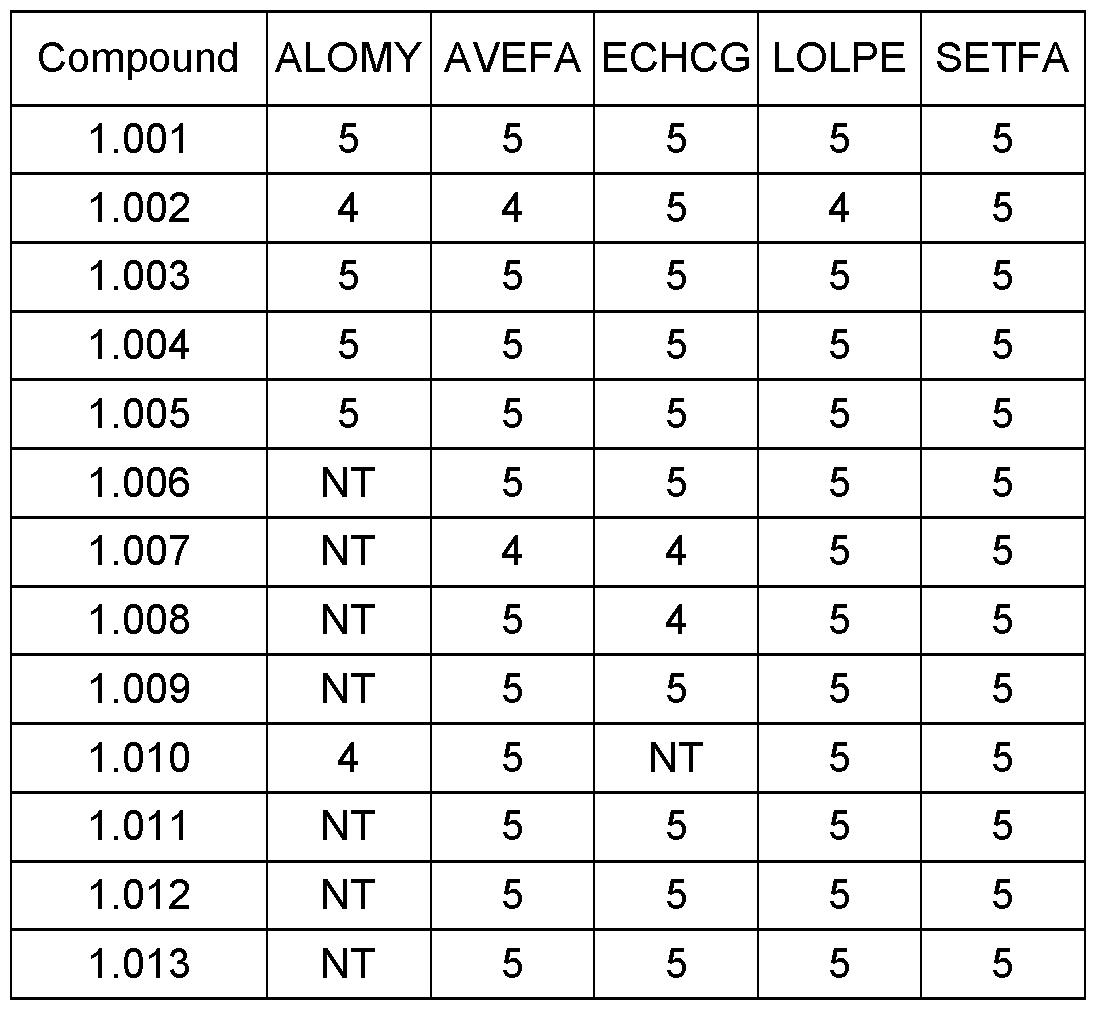
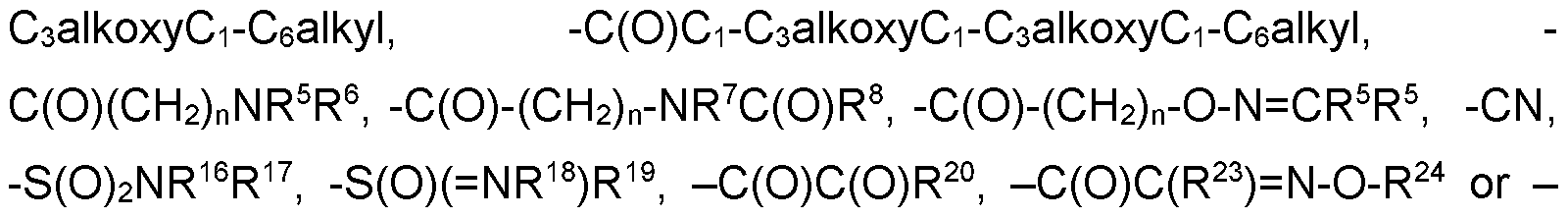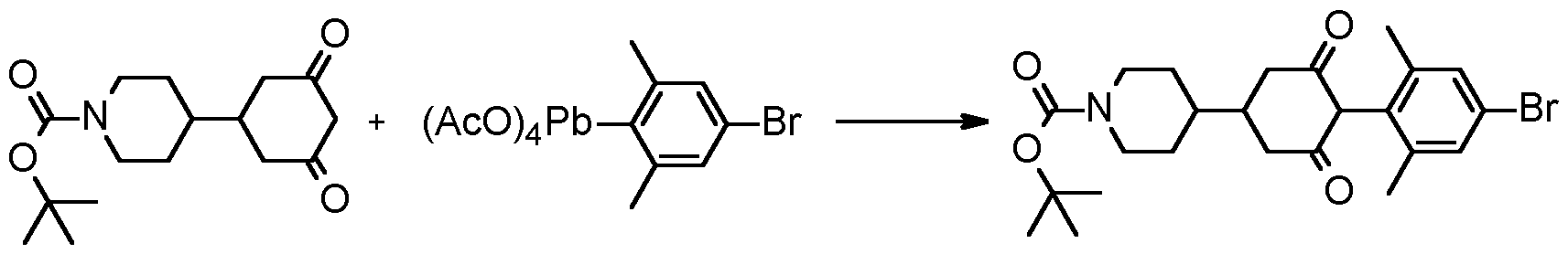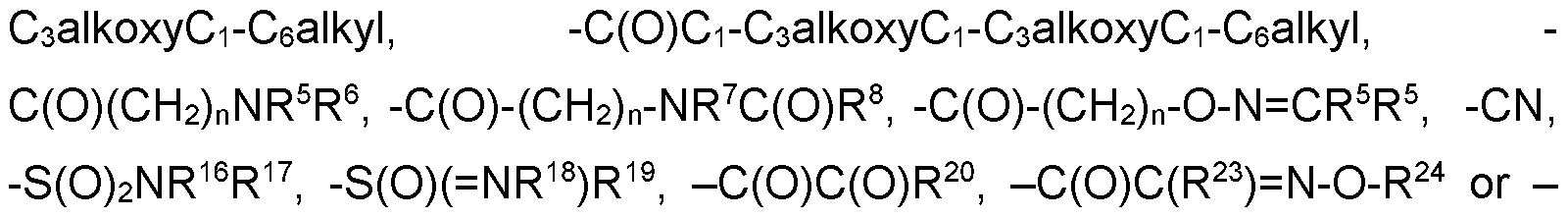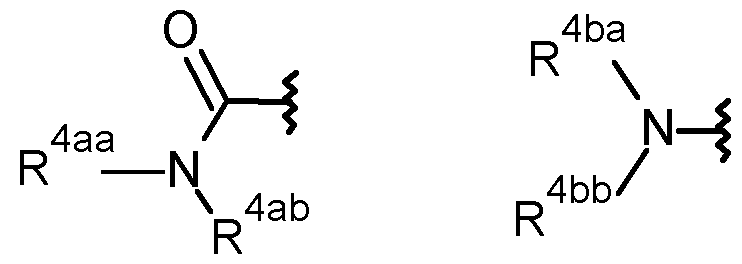WO2021018833A1 - Cyclohexanediones as herbicidal compounds - Google Patents
Cyclohexanediones as herbicidal compounds Download PDFInfo
- Publication number
- WO2021018833A1 WO2021018833A1 PCT/EP2020/071130 EP2020071130W WO2021018833A1 WO 2021018833 A1 WO2021018833 A1 WO 2021018833A1 EP 2020071130 W EP2020071130 W EP 2020071130W WO 2021018833 A1 WO2021018833 A1 WO 2021018833A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- alkyl
- hydrogen
- phenyl
- pyridyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 Cc1cc(*)cc(C)c1C(C(CC(*)(C1)N(C)C)=O)=C1ON Chemical compound Cc1cc(*)cc(C)c1C(C(CC(*)(C1)N(C)C)=O)=C1ON 0.000 description 2
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D211/00—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings
- C07D211/04—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D211/06—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members
- C07D211/08—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hydrocarbon or substituted hydrocarbon radicals directly attached to ring carbon atoms
- C07D211/18—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hydrocarbon or substituted hydrocarbon radicals directly attached to ring carbon atoms with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D211/30—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hydrocarbon or substituted hydrocarbon radicals directly attached to ring carbon atoms with substituted hydrocarbon radicals attached to ring carbon atoms with hydrocarbon radicals, substituted by doubly bound oxygen or sulfur atoms or by two oxygen or sulfur atoms singly bound to the same carbon atom
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N35/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having two bonds to hetero atoms with at the most one bond to halogen, e.g. aldehyde radical
- A01N35/06—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having two bonds to hetero atoms with at the most one bond to halogen, e.g. aldehyde radical containing keto or thioketo groups as part of a ring, e.g. cyclohexanone, quinone; Derivatives thereof, e.g. ketals
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/18—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing the group —CO—N<, e.g. carboxylic acid amides or imides; Thio analogues thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/40—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom six-membered rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/44—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom three- or four-membered rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/54—1,3-Diazines; Hydrogenated 1,3-diazines
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/56—1,2-Diazoles; Hydrogenated 1,2-diazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/02—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having no bond to a nitrogen atom
- A01N47/06—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having no bond to a nitrogen atom containing —O—CO—O— groups; Thio analogues thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/08—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having one or more single bonds to nitrogen atoms
- A01N47/28—Ureas or thioureas containing the groups >N—CO—N< or >N—CS—N<
- A01N47/38—Ureas or thioureas containing the groups >N—CO—N< or >N—CS—N< containing the group >N—CO—N< where at least one nitrogen atom is part of a heterocyclic ring; Thio analogues thereof
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C233/00—Carboxylic acid amides
- C07C233/01—Carboxylic acid amides having carbon atoms of carboxamide groups bound to hydrogen atoms or to acyclic carbon atoms
- C07C233/30—Carboxylic acid amides having carbon atoms of carboxamide groups bound to hydrogen atoms or to acyclic carbon atoms having the nitrogen atom of at least one of the carboxamide groups bound to a carbon atom of a hydrocarbon radical substituted by doubly-bound oxygen atoms
- C07C233/32—Carboxylic acid amides having carbon atoms of carboxamide groups bound to hydrogen atoms or to acyclic carbon atoms having the nitrogen atom of at least one of the carboxamide groups bound to a carbon atom of a hydrocarbon radical substituted by doubly-bound oxygen atoms with the substituted hydrocarbon radical bound to the nitrogen atom of the carboxamide group by a carbon atom of a ring other than a six-membered aromatic ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C235/00—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by oxygen atoms
- C07C235/70—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by oxygen atoms having carbon atoms of carboxamide groups and doubly-bound oxygen atoms bound to the same carbon skeleton
- C07C235/82—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by oxygen atoms having carbon atoms of carboxamide groups and doubly-bound oxygen atoms bound to the same carbon skeleton with the carbon atom of at least one of the carboxamide groups bound to a carbon atom of a ring other than a six-membered aromatic ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C235/00—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by oxygen atoms
- C07C235/88—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by oxygen atoms having the nitrogen atom of at least one of the carboxamide groups further acylated
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C259/00—Compounds containing carboxyl groups, an oxygen atom of a carboxyl group being replaced by a nitrogen atom, this nitrogen atom being further bound to an oxygen atom and not being part of nitro or nitroso groups
- C07C259/04—Compounds containing carboxyl groups, an oxygen atom of a carboxyl group being replaced by a nitrogen atom, this nitrogen atom being further bound to an oxygen atom and not being part of nitro or nitroso groups without replacement of the other oxygen atom of the carboxyl group, e.g. hydroxamic acids
- C07C259/08—Compounds containing carboxyl groups, an oxygen atom of a carboxyl group being replaced by a nitrogen atom, this nitrogen atom being further bound to an oxygen atom and not being part of nitro or nitroso groups without replacement of the other oxygen atom of the carboxyl group, e.g. hydroxamic acids having carbon atoms of hydroxamic groups bound to carbon atoms of rings other than six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D205/00—Heterocyclic compounds containing four-membered rings with one nitrogen atom as the only ring hetero atom
- C07D205/02—Heterocyclic compounds containing four-membered rings with one nitrogen atom as the only ring hetero atom not condensed with other rings
- C07D205/04—Heterocyclic compounds containing four-membered rings with one nitrogen atom as the only ring hetero atom not condensed with other rings having no double bonds between ring members or between ring members and non-ring members
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/24—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D213/36—Radicals substituted by singly-bound nitrogen atoms
- C07D213/40—Acylated substituent nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/61—Halogen atoms or nitro radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/72—Nitrogen atoms
- C07D213/75—Amino or imino radicals, acylated by carboxylic or carbonic acids, or by sulfur or nitrogen analogues thereof, e.g. carbamates
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/78—Carbon atoms having three bonds to hetero atoms, with at the most one bond to halogen, e.g. ester or nitrile radicals
- C07D213/81—Amides; Imides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/78—Carbon atoms having three bonds to hetero atoms, with at the most one bond to halogen, e.g. ester or nitrile radicals
- C07D213/84—Nitriles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/28—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to ring carbon atoms
- C07D239/30—Halogen atoms or nitro radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/28—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to ring carbon atoms
- C07D239/32—One oxygen, sulfur or nitrogen atom
- C07D239/42—One nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D261/00—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings
- C07D261/02—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings not condensed with other rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D263/00—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings
- C07D263/02—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings not condensed with other rings
- C07D263/04—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings not condensed with other rings having no double bonds between ring members or between ring members and non-ring members
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D295/00—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms
- C07D295/16—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms acylated on ring nitrogen atoms
- C07D295/18—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms acylated on ring nitrogen atoms by radicals derived from carboxylic acids, or sulfur or nitrogen analogues thereof
- C07D295/182—Radicals derived from carboxylic acids
- C07D295/185—Radicals derived from carboxylic acids from aliphatic carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/06—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/10—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/02—Systems containing only non-condensed rings with a three-membered ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/04—Systems containing only non-condensed rings with a four-membered ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/06—Systems containing only non-condensed rings with a five-membered ring
- C07C2601/08—Systems containing only non-condensed rings with a five-membered ring the ring being saturated
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/12—Systems containing only non-condensed rings with a six-membered ring
- C07C2601/14—The ring being saturated
Definitions
- the present invention relates to novel herbicidal cyclohexanedione compounds, to processes for their preparation, to herbicidal compositions which comprise the novel compounds, and to their use for controlling weeds.
- Herbicidal cyclic dione compounds substituted by a phenyl which has various substituents are disclosed in, for example, W02008/110308.
- the present invention relates to novel herbicidal cyclohexanedione derivatives with improved properties.
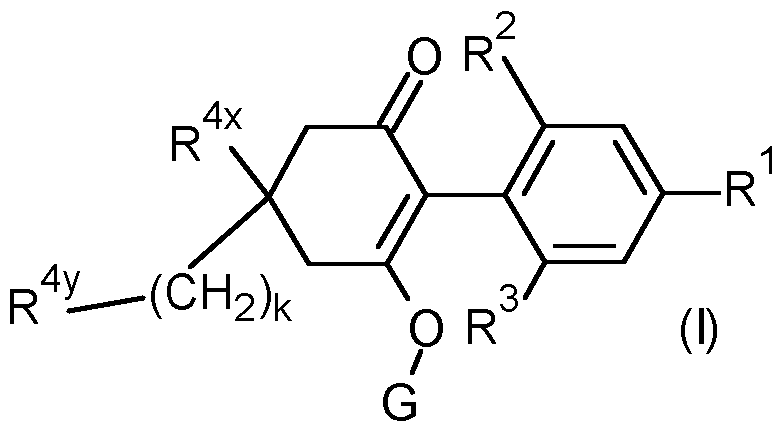
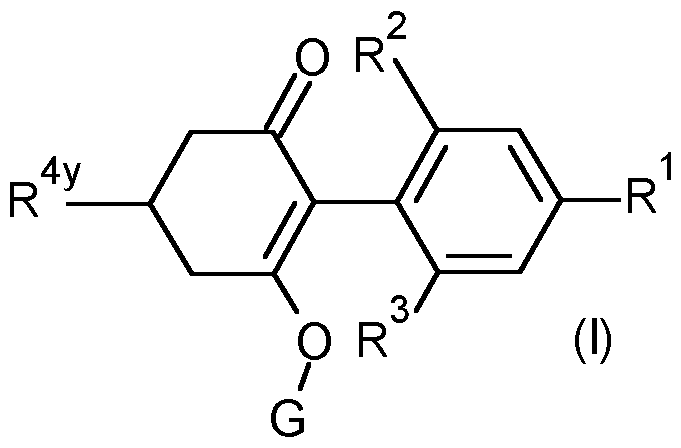
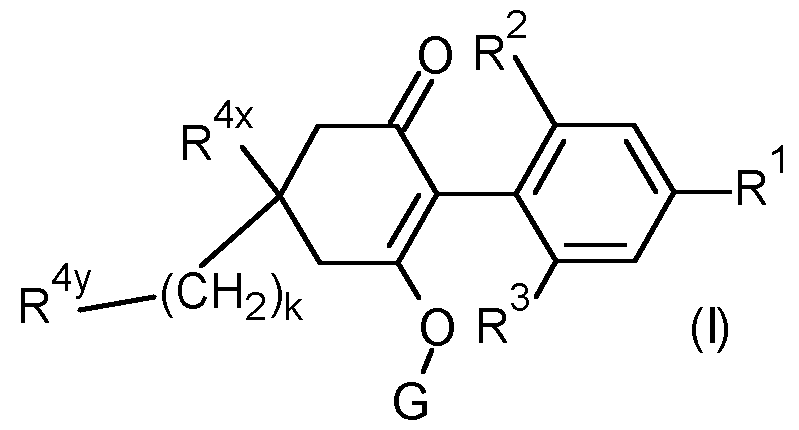
- R 1 is selected from methyl, ethynyl, 1-propynyl, phenyl and a 5 or 6 membered heteroaryl which comprises one or two nitrogen heteroatoms, said phenyl and heteroaryl optionally substituted by one or two R 15 substituents;
- R 2 is selected from the group consisting of methyl, ethyl, methoxy and chloro;
- R 3 is selected from the group consisting of methyl, ethyl, methoxy and chloro; k is O, 1 or 2;
- R 4x is selected from the group consisting of hydrogen, C1-C6 alkyl, Ci- Cehaloalkyl, Ci-CealkoxyCrCsalkyl- and Ci-CehaloalkoxyCrCsalkyl-;
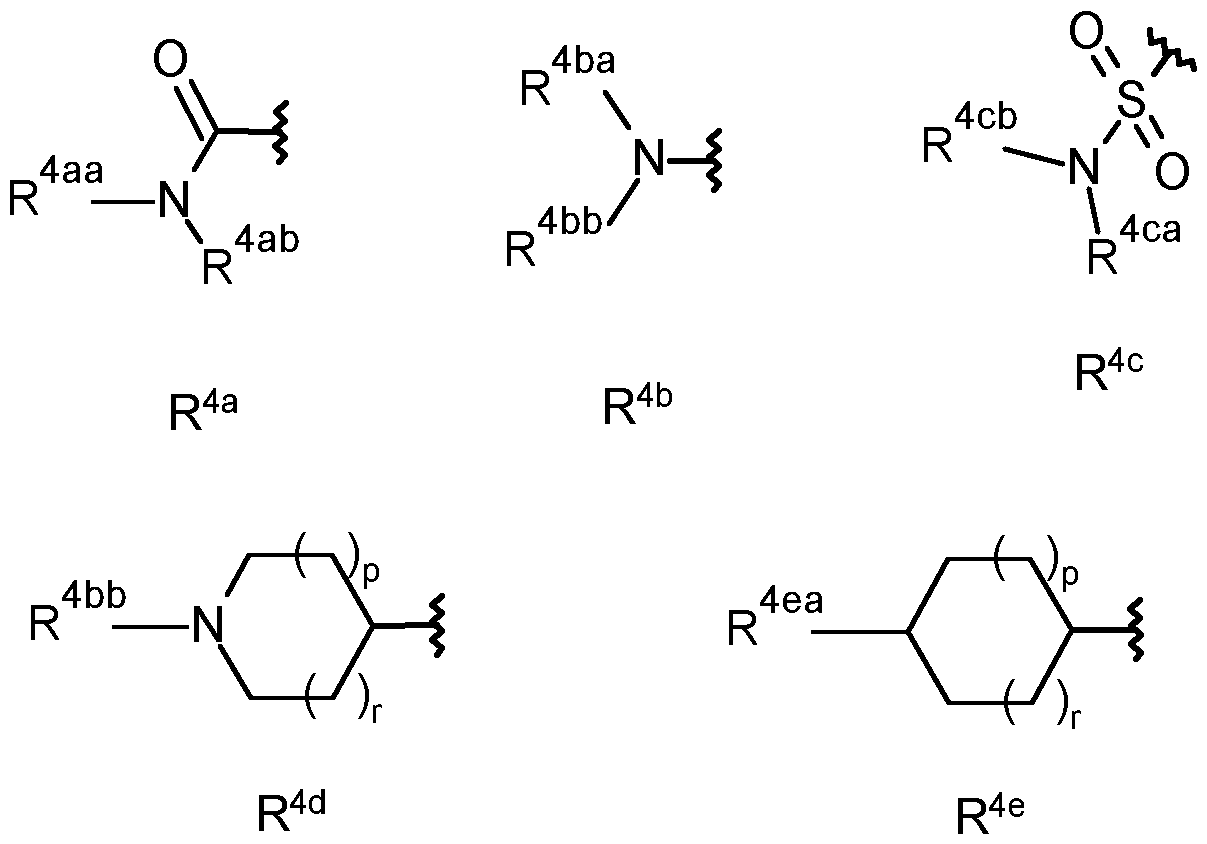
- R 4y is selected from the group consisting of R4 a , R4 b , R4 C , R 4d and R 4e ;
- R 4aa is selected from the group consisting of hydrogen, C1-C6 alkyl and Ci- Cealkoxy-;
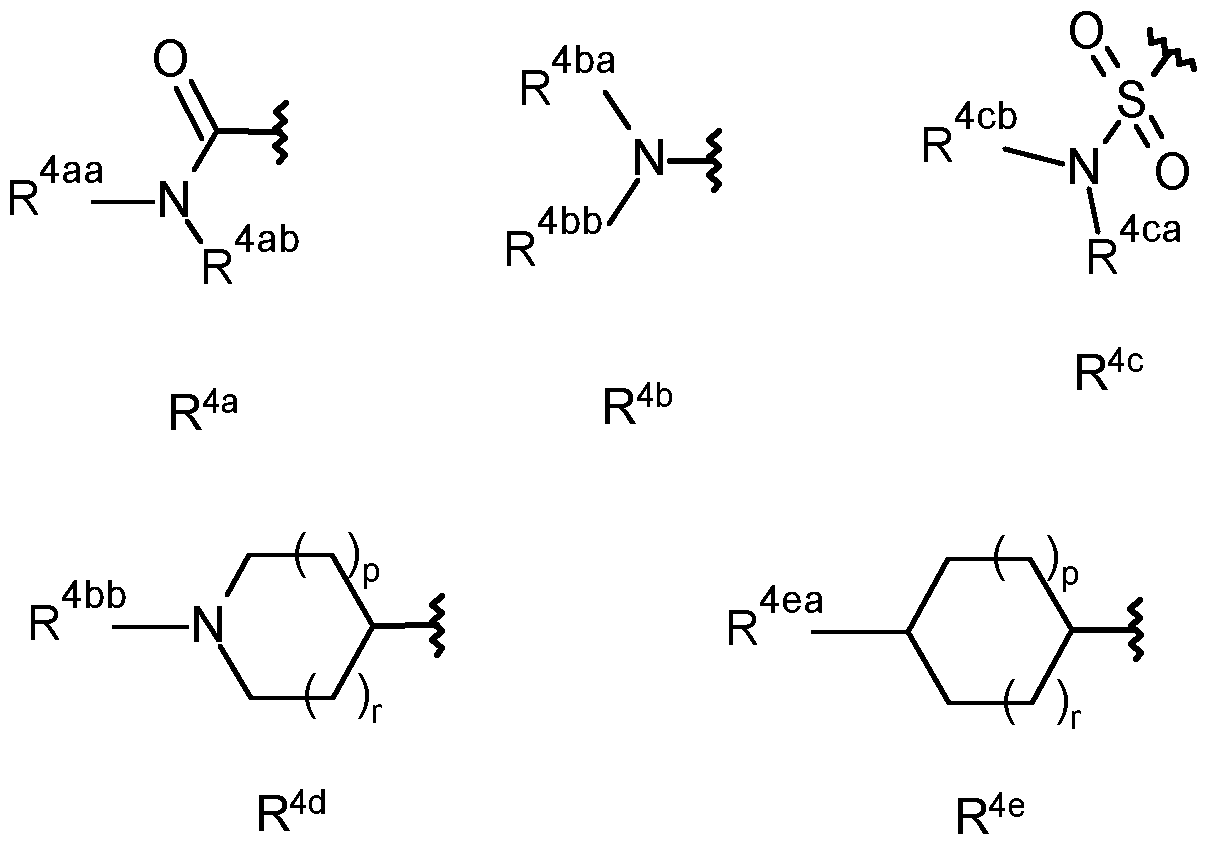
- R 4ab is selected from the group consisting of hydrogen, CrCealkyl, C2- Cealkenyl, C2-C6alkynyl, CrCehaloalkyl, hydroxy-, hydroxyCi-Cealkyl-, Ci- Cealkoxy, C3-C6cycloalkyl, -Ci-C4alkoxyCi-Cealkyl, -Ci-CsalkoxyCrCehaloalkyl, cyanoCrCealkyl-, C(0)R 27 , S(0) n R 27 , phenyl, -pyridyl and pyrimidinyl, wherein the phenyl, pyridyl or pyrimidinyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, C2-C3 alkenyl, C2-C3 al
- R 4aa and R 4ab together form— (CH 2 ) q -, -OCH2CH2CH2CH2-, -CH 2 OCH 2 C(CH 3 )2-, -CH2CH 2 X 1 CH 2 CH2- or -C(0)CH 2 X 1 CH 2 CH 2 - wherein X 1 is selected from the group consisting of O, S(0) n and N-R 28 ; and
- R 4ba is selected from the group consisting of hydrogen, C1-C4 alkyl and Ci- C4alkoxy-;
- R 4bb is selected from the group consisting of CrC4alkyl, Ci-C4alkoxy-, Ci- C4haloalkyl, -C(0)CrC 4 alkyl, -C(0)CrC 4 haloalkyl, hydroxyCi-C 6 alkyl-C(0)-, - S(0) n Ci-Cealkyl, -S(0) n Ci-C 6 haloalkyl, -S(0) n -(CH 2 ) n -C 3 -C 6 cycloalkyl, S(0) n C(R 11 )R 12 R 13 , -C(0)H, -C(0)-(CH 2 ) n -C 3 -C 6 cycloalkyl, -C(0)C(R 11 )R 12 R 13 , -C(0)C 2 -C 4 alkenyl, -C(O)(CR 9 R 10 )CN, -C(O)(CR 9 R 10 )(CR 9 R 10
- C(0)C(R 23 ) N-NR 25 R 26 , -(CH 2 ) n -phenyl, -C(0)-(CH 2 ) n -phenyl, -S(0) n -(CH 2 ) n - phenyl, -heterocyclyl, -C(0)-(CH 2 ) n -heterocyclyl, -C(0)(CH 2 ) n 0-(CH 2 ) n - heterocyclyl, -S(0) n -(CH 2 ) n -heterocyclyl, wherein each heterocyclyl is a 5- or 6- membered heterocyclyl which may be aromatic, saturated or partially saturated and can contain from 1 to 4 heteroatoms each independently selected from the group consisting of oxygen, nitrogen and sulphur, and wherein said heterocyclyl or phenyl groups are optionally substituted by one, two or three substituents independently selected from the group consisting of CrC3alkyl, CrC
- R 5 is independently selected from the group consisting of hydrogen and C1-C6 alkyl
- R 6 is selected from the group consisting of hydrogen, CrCealkyl, C 2 -C 6 alkenyl, C 2 -C 6 alkynyl, CrCehaloalkyl, hydroxyl-, CrCealkoxy, C 3 -C 6 cycloalkyl, , -Ci- C 4 alkoxyCi-C 6 alkyl, -Ci-CsalkoxyCrCehaloalkyl, -(CR 9 R 10 )Ci-C 6 haloalkyl, - (CR 9 R 10 )C(O)NR 5 R 5 , phenyl, -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C 1 -C 3 alkyl, C 1 -C 3 haloalkyl, C 1 -C 3 alkoxy, C 2 -C 3 alkenyl, C 2 -
- R 5 and R 6 together form -CH 2 CH 2 OCH 2 CH 2 -;
- R 7 is selected from the group consisting of hydrogen and C1-C6 alkyl
- R 8 is selected from the group consisting of hydrogen, C 1 -C 6 alkyl, C 1 -C 6 alkoxy, C 3 -C 6 cycloalkyl, phenyl, -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C 1 -C 3 alkyl, C 1 -C 3 haloalkyl, C 1 -C 3 alkoxy, C 2 -C 3 alkenyl, C 2 -C 3 alkynyl, halogen, cyano and nitro;
- R 9 is hydrogen or methyl
- R 1 0 is hydrogen or methyl
- R 9 and R 1 0 together form -CH 2 CH 2 -;
- R 1 1 is hydrogen or methyl
- R 1 2 is selected from the group consisting of hydrogen, C 1 -C 6 alkyl, hydroxyl and C 1 -C 6 alkoxy-;
- R 1 3 is selected from the group consisting of hydrogen, C 1 -C 6 alkyl, hydroxyl and C 1 -C 6 alkoxy; or
- R 1 2 and R 1 3 together form -CH 2 -X 2 -CH 2 - wherein X 2 is selected from the group consisting of O, S and N-R 14 ; and
- R 1 4 is selected from the group consisting of hydrogen, C 1 -C 3 alkyl and C 1 -C 3 alkoxy-;
- R 1 5 is independently selected from the group consisting of C 1 -C 4 alkyl, C 1 -C 4 haloalkyl, cyano and halogen;
- R 16 is hydrogen or CrCealkyl
- R 17 is selected from the group consisting of hydrogen, CrCealkyl, C 3 - Cecycloalkyl, CrCe alkoxy-CrCsalkyl-, -C(0)CrCealkyl, -C(0)OCrCealkyl and CH 2 CN; or
- R 1 6 and R 1 7 together form -CH2CH2OCH2CH2-, -CH 2 CH2S(0)2CH 2 CH2-; and R 18 is hydrogen or CrCealkyl;
- R 1 9 is selected from the group consisting of hydrogen, C 1 -C 6 alkyl, C 1 -C 6 alkoxy, C 3 -C 6 cycloalkyl, phenyl, -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C 1 -C 3 alkyl, C 1 -C 3 haloalkyl, C 1 -C 3 alkoxy, C 2 -C 3 alkenyl, C 2 -C 3 alkynyl, halogen, cyano and nitro;
- R 20 is selected from the group consisting of CrCealkyl, CrCehaloalkyl, Cr Cealkoxy-, CrCehaloalkoxy, -NR 21 R 22 , phenyl and -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C 1 -C 3 alkyl, C 1 -C 3 haloalkyl, C 1 -C 3 alkoxy, C 2 -C 3 alkenyl, C 2 -C 3 alkynyl, halogen, cyano and nitro;
- R 21 is selected from the group consisting of hydrogen, C 1 -C 6 alkyl, C 1 -C 6 alkoxy, CrCealkoxyCrCsalkyl-, C 3 -C 6 cycloalkyl, CrCehaloalkyl- and Cr Cehaloalkoxy-, -C(0)CrCealkyl, phenyl, -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C 1 -C 3 alkyl, C 1 -C 3 haloalkyl, C 1 -C 3 alkoxy, C 2 -C 3 alkenyl, C 2 -C 3 alkynyl, halogen, cyano and nitro;
- R 22 is hydrogen or CrCealkyl
- R 21 and R 22 together form -CH 2 CH 2 OCH 2 CH 2 -;
- R 23 is selected from the group consisting of hydrogen, CrCealkyl, Cr
- R 24 is selected from the group consisting of hydrogen, CrCealkyl, Cr
- R 25 is hydrogen or C 1 -C 6 alkyl
- R 26 is hydrogen or C1-C6 alkyl
- R 4ca is selected from hydrogen, CrCealkyl, C2-Cealkenyl, C2-C6alkynyl, Ci- Cehaloalkyl, hydroxyl-, CrCealkoxy, C3-Cecycloalkyl, -Ci-C4alkoxyCi-Cealkyl, - Ci-C3alkoxyCrC6haloalkyl, cyanoC1-C6alkyl-.
- R 4cb is selected from the group consisting of hydrogen, CrCe alkyl and Ci- Cealkoxy-; or
- R 4ca and R 4cb together form — (CH 2 ) q -, -CH2CH 2 X 1 CH 2 CH2- or - C(0)CH 2 X 1 CH 2 CH 2 - wherein X 1 is selected from the group consisting of O, S(0) n and N-R 28 ;
- R 27 is selected from the group consisting of hydrogen, CrCe alkyl, CrCe haloalkyl, C2-Cealkenyl, C2-Cealkynyl, CrCe alkoxy, C 3 -C 6 cycloalkyl, phenyl, -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of Ci- C 3 alkyl, C1-C 3 haloalkyl, C1-C 3 alkoxy, C2-C 3 alkenyl, C2-C 3 alkynyl, halogen, cyano and nitro;
- R 28 is selected from the group consisting of hydrogen, C1-C 3 alkyl, -C(0)Ci- Csalkyl and C1-C 3 alkoxy-;
- R 4ea is R4 ea1 or R4 ea2
- p4ea1 p4ea2 n is independently 0, 1 or 2; q is 3, 4 or 5; and
- G is selected from the group consisting of hydrogen, -(Chb R 3 , -C(0)-R a , - C(0)-(CR c R d ) n -0-R b , -C(0)-(CR c R d ) n -S-R b , -C(0)NR a R a , -S(0) 2 -R a and Cr Csalkoxy-Ci-Csalkyl-;
- R a is independently selected from the group consisting of hydrogen, Cr Csalkyl, CrCshaloalkyl, C2-Csalkenyl, C2-Csalkynyl, C3-C6cycloalkyl, heterocyclyl and phenyl wherein said heterocyclyl and phenyl groups are optionally substituted by one, two or three substituents independently selected from the group consisting of CrCsalkyl, CrCshaloalkyl, CrCsalkoxy, C2-Csalkenyl, C2-Csalkynyl, halogen, cyano and nitro;
- R b is selected from the group consisting of CrCsalkyl, CrCshaloalkyl, C 2 - Csalkenyl, C2-Csalkynyl, C3-C6 cycloalkyl, heterocyclyl and phenyl wherein said heterocyclyl and phenyl groups are optionally substituted by one, two or three substituents independently selected from the group consisting of Cr Csalkyl, CrCshaloalkyl, CrCsalkoxy, C2-C3alkenyl, C2-C3alkynyl, halogen, cyano and nitro;
- R c is hydrogen or C 1 -C 3 alkyl
- R d is hydrogen or C 1 -C 3 alkyl; or an agriculturally acceptable salt thereof.
- Alkyl groups include, for example, methyl (Me, CH 3 ), ethyl (Et, C2H5), n-propyl (n-Pr), isopropyl (/-Pr), n-butyl (n-Bu), isobutyl (/- Bu), sec-butyl (s-Bu) and tert- butyl (f-Bu).
- Alkenyl and alkynyl moieties can be in the form of straight or branched chains, and the alkenyl moieties, where appropriate, can be of either the (£)- or (Z)-configuration. Examples are vinyl, allyl and propargyl. Alkenyl and alkynyl moieties can contain one or more double and/or triple bonds in any combination.
- Halogen or halo encompasses fluorine, chlorine, bromine or iodine. The same correspondingly applies to halogen in the context of other definitions, such as haloalkyl.
- Haloalkyl groups are, for example, fluoromethyl, difluoromethyl, trifluoromethyl, chloromethyl, dichloromethyl, trichloromethyl, 2,2,2- trifluoroethyl, 2-fluoroethyl, 2-chloroethyl, pentafluoroethyl, 1 , 1 -difluoro-2,2,2- trichloroethyl, 2,2,3,3-tetrafluoroethyl and 2,2,2-trichloroethyl, heptafluoro-n-propyl and peril uoro-n-hexyl.
- Alkoxy groups are, for example, methoxy, ethoxy, propoxy, isopropoxy, n-butoxy, isobutoxy, sec-butoxy or tert-butoxy, preferably methoxy and ethoxy.
- Alkoxyalkyl groups (e.g Ci-Csalkoxy-CrCsalkyl-) includes, for example, methoxymethyl, methoxyethyl, ethoxymethyl, ethoxyethyl, n-propoxymethyl, n- propoxyethyl, isopropoxymethyl or isopropoxyethyl.
- Cycloalkyl groups include, for example cyclopropyl (c- propyl, c-Pr), cyclobutyl (c-butyl, c-Bu), cyclopentyl (c-pentyl) and cyclohexyl (c-hexyl) and may be substituted or unsubstituted as indicated.
- CrC 6 alkyl-S- (alkylthio) is, for example, methylthio, ethylthio, propylthio, isopropylthio, n-butylthio, isobutylthio, sec-butylthio or tert-butylthio, preferably methylthio or ethylthio.
- Ci-C 6 alkyl-S(0)- (alkylsulfinyl) is, for example, methylsulfinyl, ethylsulfinyl, propylsulfinyl, isopropylsulfinyl, n-butylsulfinyl, isobutylsulfinyl, sec-butylsulfinyl or tert-butylsulfinyl, preferably methylsulfinyl or ethylsulfinyl.
- alkyl-S(0) 2 - (alkylsulfonyl) is, for example, methylsulfonyl, ethylsulfonyl, propylsulfonyl, isopropylsulfonyl, n-butylsulfonyl, isobutylsulfonyl, sec-butylsulfonyl or tert-butylsulfonyl, preferably methylsulfonyl or ethylsulfonyl.
- Heterocyclyl is a 5- or 6- membered heterocyclyl which may be aromatic, saturated or partially saturated and can contain from 1 to 4 heteroatoms each independently selected from the group consisting of oxygen, nitrogen and sulphur.
- the invention also relates agriculturally acceptable salts of the compounds of Formula (I).
- Such salts include those which are able to form with amines, alkali metal and alkaline earth metal bases or quaternary ammonium bases.
- alkali metal and alkaline earth metal hydroxides as salt formers special mention should be made of the hydroxides of lithium, sodium, potassium, magnesium and calcium, but especially the hydroxides of sodium and potassium.
- the compounds of Formula (I) according to the invention also include hydrates which may be formed during the salt formation.
- amines suitable for ammonium salt formation include ammonia as well as primary, secondary and tertiary CrCisalkylamines, Ci- C4hydroxyalkylamines and C2-C4alkoxyalkylamines, for example methylamine, ethylamine, n-propylamine, isopropylamine, the four butylamine isomers, n- amylamine, isoamylamine, hexylamine, heptylamine, octylamine, nonylamine, decylamine, pentadecylamine, hexadecylamine, heptadecylamine, octadecylamine, methylethylamine, methylisopropylamine, methylhexylamine, methylnonylamine, methylpentadecylamine, methyloctadecylamine, ethylbutylamine, e
- R 1 is 1-propynyl. In another embodiment of the present invention R 1 is phenyl optionally substituted by one or two R 15 substituents, e.g selected from the group consisting of cyano, chloro and fluoro.
- R 1 is a 5 or 6 membered heteroaryl which comprises one or two nitrogen heteroatoms, said heteroaryl optionally substituted by one or two R 15 substituents, e.g selected from the group consisting of cyano, chloro and fluoro.
- said heteroaryl is selected from the group consisting of pyridyl, pyrimidinyl, and pyrazolyl.
- R 2 is methoxy, chloro or methyl. In a preferred embodiment of the present invention R 2 is methyl.
- R 3 is methyl or methoxy, preferably methyl.
- R 2 is methyl and R 3 is methyl, R 2 is methoxy and R 3 is methyl or R 2 is chloro and R 3 is methoxy. In a particularly preferred embodiment, R 2 is methyl and R 3 is methyl.
- k is 0 or 1. In one embodiment of the present invention k is 0. In another embodiment of the present invention k is 1.
- R 4x is hydrogen or C1-C6 alkyl (e.g methyl), most preferably hydrogen.
- R 4y is R 4a .
- R 4aa is preferably hydrogen or methyl and R 4ab is preferably selected from the group consisting of C Cealkyl (e.g methyl, f-butyl), pyridyl, -C(0)CH2X 1 CH2CH2- (wherein X 1 is O), -CH2CH 2 X 1 CH 2 CH2- (wherein X 1 is O, N-R 28 wherein R 28 is -C(0)CrC 3 alkyl (preferablky methyl), or S(0) n wherein n is 2), hydroxyCi-Cealkyl- (e.g HO-CH2CH2-), CrCealkoxy (e.g MeO), -Ci-C4alkoxyCrC6alkyl (e.g CH 3 OCH2CH2-) and cyanoCr Cealkyl- (e.g -C(CH 3 )(C 2 H 5 )(CN).
- C Cealkyl e.
- R 4y is R 4b .
- R 4ba is preferably hydrogen or methyl and R 4bb is selected from the group consisting of -C(0)Ci-C 4 alkyi (e.g -C(0)CH 3 , -C(O)f-butyl), -C(0)-(CH 2 ) n -C 3 -C 6 cycloalkyl (e.g - C(O)-cyclopropyl), -C(0)-heterocyclyl (e.g pyridyl), -C(0)-(CH 2 ) n -phenyl, C(0)(CH 2 ) n NR 5 R 6 , -C(0)Ci-C 3 alkoxyCi-C 6 haloalkyl (e.g CHF 2 -0-CH 2 -), -
- R 4y is R 4d .
- p and r are preferably both 0 or both 1.
- R4 bb is preferably selected from the group consisting of is selected from the group consisting of CrC4alkoxy- (e.g methoxy-), -C(0)CrC 4 alkyl (e.g -C(0)CH 3 , -C(O)f-butyl), -C(0)-(CH 2 ) n -C 3 -C 6 cycloalkyl (e.g -C(O)-cyclopropyl), -C(O)- heterocyclyl (e.g pyridyl), -C(0)-(CH 2 ) n -phenyl, -C(0)(CH 2 ) n NR 5 R 6 , -C(0)Ci- CsalkoxyCrCehaloalkyl (e.g CHF 2 -0-CH 2 -), -C(
- R 4y is R 4e .
- each aforementioned heterocyclyl is an aromatic heterocyclyl (i.e heteroaryl), more preferably selected from the group consisting of furanyl, pyrrolyl, thiophenyl, imidazolyl, pyrazolyl, oxazolyl, isoxazolyl, thiazolyl, pyranyl, pyridyl, pyrazinyl, pyrimidinyl, pyridazinyl, and triazolyl more preferably selected from the group consisting of pyridyl, pyridazinyl, pyrimidinyl and pyrazinyl each of which is optionally substituted as defined previously.
- aromatic heterocyclyl i.e heteroaryl
- each aforementioned heterocyclyl is a partially saturated heterocyclyl, more preferably selected from the group consisting of imidazolinyl, isoxazolinyl and thiazolinyl each of which is optionally substituted as defined previously.
- each aforementioned heterocyclyl is a saturated heterocyclyl more preferably selected from the group consisting of morpholinyl, tetrahydrofuryl and tetrahydropyranyl each of which is optionally substituted as defined previously.
- R 5 is hydrogen or methyl, preferably hydrogen.
- R 6 is C1-C6 alkyl (e.g methyl) or pyridyl.
- R 7 is hydrogen or methyl, preferably hydrogen.
- R 8 is C1-C6 alkyl (e.g f-butyl) or pyridyl.
- R 9 is hydrogen
- R 10 is hydrogen
- G is selected from the group consisting of hydrogen, CrCsalkyl (e.g methyl, ethyl, n-propyl, /-propyl, n-butyl, f-butyl, -C2-Csalkenyl (e.g vinyl), C2-Csalkynyl (e.g propargyl), -C(0)Ci-Csalkyl (more preferably -C(0)Ci-C 6 alkyl e.g -C(0)/-propyl and -C(O)f-butyl) and -C(0)-0-Ci- Csalkyl (more preferably -C(0)-0-Ci-C 6 alkyl e.g -C(O)-O-methyl).
- G is hydrogen.
- compounds of Formula (I) may exist in different isomeric forms.
- G is hydrogen, for example, compounds of Formula (I) may exist in different tautomeric forms.
- This invention covers all such isomers and tautomers and mixtures thereof in all proportions. Also, when substituents contain double bonds, cis- and trans- isomers can exist. These isomers, too, are within the scope of the claimed compounds of the Formula (I).
- Compounds of Formula (I) may contain asymmetric centres and may be present as a single enantiomer, pairs of enantiomers in any proportion or, where more than one asymmetric centre are present, contain diastereoisomers in all possible ratios. Typically one of the enantiomers has enhanced biological activity compared to the other possibilities.
- the compounds of Formula (I) according to the invention can be used as herbicides by themselves, but they are generally formulated into herbicidal compositions using formulation adjuvants, such as carriers, solvents and surface- active agents (SFAs).
- formulation adjuvants such as carriers, solvents and surface- active agents (SFAs).
- the present invention further provides a herbicidal composition comprising a herbicidal compound according to any one of the previous claims and an agriculturally acceptable formulation adjuvant.
- the composition can be in the form of concentrates which are diluted prior to use, although ready-to-use compositions can also be made. The final dilution is usually made with water, but can be made instead of, or in addition to, water, with, for example, liquid fertilisers, micronutrients, biological organisms, oil or solvents.
- the herbicidal compositions generally comprise from 0.1 to 99 % by weight, especially from 0.1 to 95 % by weight, compounds of Formula (I) and from 1 to 99.9 % by weight of a formulation adjuvant which preferably includes from 0 to 25 % by weight of a surface-active substance.
- compositions can be chosen from a number of formulation types, many of which are known from the Manual on Development and Use of FAO Specifications for Plant Protection Products, 5th Edition, 1999. These include dustable powders (DP), soluble powders (SP), water soluble granules (SG), water dispersible granules (WG), wettable powders (WP), granules (GR) (slow or fast release), soluble concentrates (SL), oil miscible liquids (OL), ultra low volume liquids (UL), emulsifiable concentrates (EC), dispersible concentrates (DC), emulsions (both oil in water (EW) and water in oil (EO)), micro-emulsions (ME), suspension concentrates (SC), aerosols, capsule suspensions (CS) and seed treatment formulations.
- the formulation type chosen in any instance will depend upon the particular purpose envisaged and the physical, chemical and biological properties of the compound of Formula (I).
- Dustable powders may be prepared by mixing a compound of Formula (I) with one or more solid diluents (for example natural clays, kaolin, pyrophyllite, bentonite, alumina, montmorillonite, kieselguhr, chalk, diatomaceous earths, calcium phosphates, calcium and magnesium carbonates, sulphur, lime, flours, talc and other organic and inorganic solid carriers) and mechanically grinding the mixture to a fine powder.
- solid diluents for example natural clays, kaolin, pyrophyllite, bentonite, alumina, montmorillonite, kieselguhr, chalk, diatomaceous earths, calcium phosphates, calcium and magnesium carbonates, sulphur, lime, flours, talc and other organic and inorganic solid carriers
- Soluble powders may be prepared by mixing a compound of Formula (I) with one or more water-soluble inorganic salts (such as sodium bicarbonate, sodium carbonate or magnesium sulphate) or one or more water-soluble organic solids (such as a polysaccharide) and, optionally, one or more wetting agents, one or more dispersing agents or a mixture of said agents to improve water dispersibility/solubility. The mixture is then ground to a fine powder. Similar compositions may also be granulated to form water soluble granules (SG).
- water-soluble inorganic salts such as sodium bicarbonate, sodium carbonate or magnesium sulphate
- water-soluble organic solids such as a polysaccharide
- WP Wettable powders
- WG Water dispersible granules
- Granules may be formed either by granulating a mixture of a compound of Formula (I) and one or more powdered solid diluents or carriers, or from pre formed blank granules by absorbing a compound of Formula (I) (or a solution thereof, in a suitable agent) in a porous granular material (such as pumice, attapulgite clays, fuller's earth, kieselguhr, diatomaceous earths or ground corn cobs) or by adsorbing a compound of Formula (I) (or a solution thereof, in a suitable agent) on to a hard core material (such as sands, silicates, mineral carbonates, sulphates or phosphates) and drying if necessary.
- a hard core material such as sands, silicates, mineral carbonates, sulphates or phosphates
- Agents which are commonly used to aid absorption or adsorption include solvents (such as aliphatic and aromatic petroleum solvents, alcohols, ethers, ketones and esters) and sticking agents (such as polyvinyl acetates, polyvinyl alcohols, dextrins, sugars and vegetable oils).
- solvents such as aliphatic and aromatic petroleum solvents, alcohols, ethers, ketones and esters
- sticking agents such as polyvinyl acetates, polyvinyl alcohols, dextrins, sugars and vegetable oils.
- One or more other additives may also be included in granules (for example an emulsifying agent, wetting agent or dispersing agent).
- DC Dispersible Concentrates
- a compound of Formula (I) may be prepared by dissolving a compound of Formula (I) in water or an organic solvent, such as a ketone, alcohol or glycol ether.
- organic solvent such as a ketone, alcohol or glycol ether.
- surface active agent for example to improve water dilution or prevent crystallisation in a spray tank.
- Emulsifiable concentrates or oil-in-water emulsions (EW) may be prepared by dissolving a compound of Formula (I) in an organic solvent (optionally containing one or more wetting agents, one or more emulsifying agents or a mixture of said agents).
- Suitable organic solvents for use in ECs include aromatic hydrocarbons (such as alkylbenzenes or alkylnaphthalenes, exemplified by SOLVESSO 100, SOLVESSO 150 and SOLVESSO 200; SOLVESSO is a Registered Trade Mark), ketones (such as cyclohexanone or methylcyclohexanone) and alcohols (such as benzyl alcohol, furfuryl alcohol or butanol), N-alkylpyrrolidones (such as N-methylpyrrolidone or N-octylpyrrolidone), dimethyl amides of fatty acids (such as Cs-Cio fatty acid dimethylamide) and chlorinated hydrocarbons.
- An EC product may spontaneously emulsify on addition to water, to produce an emulsion with sufficient stability to allow spray application through appropriate equipment.
- Preparation of an EW involves obtaining a compound of Formula (I) either as a liquid (if it is not a liquid at room temperature, it may be melted at a reasonable temperature, typically below 70°C) or in solution (by dissolving it in an appropriate solvent) and then emulsifying the resultant liquid or solution into water containing one or more SFAs, under high shear, to produce an emulsion.
- Suitable solvents for use in EWs include vegetable oils, chlorinated hydrocarbons (such as chlorobenzenes), aromatic solvents (such as alkylbenzenes or alkylnaphthalenes) and other appropriate organic solvents which have a low solubility in water.
- Microemulsions may be prepared by mixing water with a blend of one or more solvents with one or more SFAs, to produce spontaneously a thermodynamically stable isotropic liquid formulation.
- a compound of Formula (I) is present initially in either the water or the solvent/SFA blend.
- Suitable solvents for use in MEs include those hereinbefore described for use in in ECs or in EWs.
- An ME may be either an oil-in-water or a water-in-oil system (which system is present may be determined by conductivity measurements) and may be suitable for mixing water- soluble and oil-soluble pesticides in the same formulation.
- An ME is suitable for dilution into water, either remaining as a microemulsion or forming a conventional oil- in-water emulsion.
- SC Suspension concentrates
- SCs may comprise aqueous or non-aqueous suspensions of finely divided insoluble solid particles of a compound of Formula (I).
- SCs may be prepared by ball or bead milling the solid compound of Formula (I) in a suitable medium, optionally with one or more dispersing agents, to produce a fine particle suspension of the compound.
- One or more wetting agents may be included in the composition and a suspending agent may be included to reduce the rate at which the particles settle.
- a compound of Formula (I) may be dry milled and added to water, containing agents hereinbefore described, to produce the desired end product.
- Aerosol formulations comprise a compound of Formula (I) and a suitable propellant (for example n-butane).
- a compound of Formula (I) may also be dissolved or dispersed in a suitable medium (for example water or a water miscible liquid, such as n-propanol) to provide compositions for use in non-pressurised, hand-actuated spray pumps.
- Capsule suspensions (CS) may be prepared in a manner similar to the preparation of EW formulations but with an additional polymerisation stage such that an aqueous dispersion of oil droplets is obtained, in which each oil droplet is encapsulated by a polymeric shell and contains a compound of Formula (I) and, optionally, a carrier or diluent therefor.
- the polymeric shell may be produced by either an interfacial polycondensation reaction or by a coacervation procedure.
- the compositions may provide for controlled release of the compound of Formula (I) and they may be used for seed treatment.
- a compound of Formula (I) may also be formulated in a biodegradable polymeric matrix to provide a slow, controlled release of the compound.
- the composition may include one or more additives to improve the biological performance of the composition, for example by improving wetting, retention or distribution on surfaces; resistance to rain on treated surfaces; or uptake or mobility of a compound of Formula (I).
- additives include surface active agents (SFAs), spray additives based on oils, for example certain mineral oils or natural plant oils (such as soy bean and rape seed oil), and blends of these with other bio-enhancing adjuvants (ingredients which may aid or modify the action of a compound of Formula
- Wetting agents, dispersing agents and emulsifying agents may be SFAs of the cationic, anionic, amphoteric or non-ionic type.
- Suitable SFAs of the cationic type include quaternary ammonium compounds (for example cetyltrimethyl ammonium bromide), imidazolines and amine salts.
- Suitable anionic SFAs include alkali metals salts of fatty acids, salts of aliphatic monoesters of sulphuric acid (for example sodium lauryl sulphate), salts of sulphonated aromatic compounds (for example sodium dodecylbenzenesulphonate, calcium dodecylbenzenesulphonate, butylnaphthalene sulphonate and mixtures of sodium di-/sopropyl- and tri-/sopropyl-naphthalene sulphonates), ether sulphates, alcohol ether sulphates (for example sodium laureth-3-sulphate), ether carboxylates (for example sodium laureth-3-carboxylate), phosphate esters (products from the reaction between one or more fatty alcohols and phosphoric acid (predominately mono-esters) or phosphorus pentoxide (predominately di-esters), for example the reaction between lauryl alcohol and tetraphosphoric acid
- Suitable SFAs of the non-ionic type include condensation products of alkylene oxides, such as ethylene oxide, propylene oxide, butylene oxide or mixtures thereof, with fatty alcohols (such as oleyl alcohol or cetyl alcohol) or with alkylphenols (such as octylphenol, nonylphenol or octylcresol); partial esters derived from long chain fatty acids or hexitol anhydrides; condensation products of said partial esters with ethylene oxide; block polymers (comprising ethylene oxide and propylene oxide); alkanolamides; simple esters (for example fatty acid polyethylene glycol esters); amine oxides (for example lauryl dimethyl amine oxide); and lecithins.
- alkylene oxides such as ethylene oxide, propylene oxide, butylene oxide or mixtures thereof
- fatty alcohols such as oleyl alcohol or cetyl alcohol
- alkylphenols such as octylphenol, nonyl
- Suitable suspending agents include hydrophilic colloids (such as polysaccharides, polyvinylpyrrolidone or sodium carboxymethylcellulose) and swelling clays (such as bentonite or attapulgite).
- hydrophilic colloids such as polysaccharides, polyvinylpyrrolidone or sodium carboxymethylcellulose
- swelling clays such as bentonite or attapulgite
- the herbicidal compounds of the present invention can also be used in mixture with one or more additional herbicides and/or plant growth regulators.
- additional herbicides or plant growth regulators include acetochlor, acifluorfen (including acifluorfen-sodium), aclonifen, ametryn, amicarbazone, aminopyralid, aminotriazole, atrazine, beflubutamid-M, bensulfuron
- trifloxysulfuron including trifloxysulfuron-sodium
- trifludimoxazin
- the mixing partners of the compound of Formula (I) may also be in the form of esters or salts, as mentioned e.g. in The Pesticide Manual, Sixteenth Edition, British Crop Protection Council, 2012.
- the compound of Formula (I) can also be used in mixtures with other agrochemicals such as fungicides, nematicides or insecticides, examples of which are given in The Pesticide Manual.
- the mixing ratio of the compound of Formula (I) to the mixing partner is preferably from 1 : 100 to 1000:1.
- the mixtures can advantageously be used in the above-mentioned formulations (in which case "active ingredient” relates to the respective mixture of compound of Formula (I) with the mixing partner).
- active ingredient relates to the respective mixture of compound of Formula (I) with the mixing partner.
- the compounds or mixtures of the present invention can also be used in combination with one or more herbicide safeners.
- safeners examples include benoxacor, cloquintocet (including cloquintocet-mexyl), cyprosulfamide, dichlormid, fenchlorazole (including fenchlorazole-ethyl), fenclorim, fluxofenim, furilazole, isoxadifen (including isoxadifen-ethyl), mefenpyr (including mefenpyr-diethyl), metcamifen and oxabetrinil.
- mixtures of a compound of Formula (I) with cyprosulfamide, isoxadifen-ethyl, cloquintocet-mexyl and/or metcamifen are particularly preferred.
- the safeners of the compound of Formula (I) may also be in the form of esters or salts, as mentioned e.g. in The Pesticide Manual, 16 th Edition (BCPC), 2012.
- the reference to cloquintocet-mexyl also applies to a lithium, sodium, potassium, calcium, magnesium, aluminium, iron, ammonium, quaternary ammonium, sulfonium or phosphonium salt thereof as disclosed in WO 02/34048.
- the mixing ratio of compound of Formula (I) to safener is from 100:1 to 1 : 10, especially from 20: 1 to 1 : 1.
- mixtures can advantageously be used in the above-mentioned formulations (in which case“active ingredient” relates to the respective mixture of compound of Formula (I) with the safener).
- the present invention still further provides a method of controlling weeds at a locus comprising crop plants and weeds, wherein the method comprises application to the locus of a weed controlling amount of a composition according to the present invention.
- Controlling means killing, reducing or retarding growth or preventing or reducing germination.
- the plants to be controlled are unwanted plants (weeds).
- Locus means the area in which the plants are growing or will grow.
- the rates of application of compounds of Formula (I) may vary within wide limits and depend on the nature of the soil, the method of application (pre- or post emergence; seed dressing; application to the seed furrow; no tillage application etc.), the crop plant, the weed(s) to be controlled, the prevailing climatic conditions, and other factors governed by the method of application, the time of application and the target crop.
- the compounds of Formula (I) according to the invention are generally applied at a rate of from 10 to 2000 g/ha, especially from 50 to 1000 g/ha.
- the application is generally made by spraying the composition, typically by tractor mounted sprayer for large areas, but other methods such as dusting (for powders), drip or drench can also be used.
- Useful plants in which the composition according to the invention can be used include crops such as cereals, for example barley and wheat, cotton, oilseed rape, sunflower, maize, rice, soybeans, sugar beet, sugar cane and turf.
- Crop plants can also include trees, such as fruit trees, palm trees, coconut trees or other nuts. Also included are vines such as grapes, fruit bushes, fruit plants and vegetables.
- Crops are to be understood as also including those crops which have been rendered tolerant to herbicides or classes of herbicides (e.g. ALS-, GS-, EPSPS-, PPO-, ACCase- and HPPD-inhibitors) by conventional methods of breeding or by genetic engineering.
- herbicides or classes of herbicides e.g. ALS-, GS-, EPSPS-, PPO-, ACCase- and HPPD-inhibitors
- An example of a crop that has been rendered tolerant to imidazolinones, e.g. imazamox, by conventional methods of breeding is Clearfield® summer rape (canola).
- crops that have been rendered tolerant to herbicides by genetic engineering methods include e.g. glyphosate- and glufosinate- resistant maize varieties commercially available under the trade names RoundupReady® and LibertyLink®.
- Crops are also to be understood as being those which have been rendered resistant to harmful insects by genetic engineering methods, for example Bt maize (resistant to European corn borer), Bt cotton (resistant to cotton boll weevil) and also Bt potatoes (resistant to Colorado beetle).
- Bt maize are the Bt 176 maize hybrids of NK® (Syngenta Seeds).
- the Bt toxin is a protein that is formed naturally by Bacillus thuringiensis soil bacteria.
- Examples of toxins, or transgenic plants able to synthesise such toxins are described in EP-A-451 878, EP-A-374 753, WO 93/07278, WO 95/34656, WO 03/052073 and EP-A-427 529.
- transgenic plants comprising one or more genes that code for an insecticidal resistance and express one or more toxins are KnockOut® (maize), Yield Gard® (maize), NuCOTIN33B® (cotton), Bollgard® (cotton), NewLeaf® (potatoes), NatureGard® and Protexcta®.
- Plant crops or seed material thereof can be both resistant to herbicides and, at the same time, resistant to insect feeding (“stacked” transgenic events).
- seed can have the ability to express an insecticidal Cry3 protein while at the same time being tolerant to glyphosate.
- Crops are also to be understood to include those which are obtained by conventional methods of breeding or genetic engineering and contain so-called output traits (e.g. improved storage stability, higher nutritional value and improved flavour).
- output traits e.g. improved storage stability, higher nutritional value and improved flavour.
- Other useful plants include turf grass for example in golf-courses, lawns, parks and roadsides, or grown commercially for sod, and ornamental plants such as flowers or bushes.
- the compositions can be used to control unwanted plants (collectively, ‘weeds’).
- weeds to be controlled may be both monocotyledonous species, for example Agrostis, Alopecurus, Avena, Brachiaria, Bromus, Cenchrus, Cyperus, Digitaria, Echinochloa, Eleusine, Lolium, Monochoria, Rottboellia, Sagittaria, Scirpus, Setaria and Sorghum, and dicotyledonous species, for example Abutilon, Amaranthus, Ambrosia, Chenopodium, Chrysanthemum, Conyza, Galium, Ipomoea, Nasturtium, Sida, Sinapis, Solanum, Stellaria, Veronica, Viola and Xanthium.
- Agrostis Alopecurus
- Avena Brachiaria
- Bromus Cenchrus
- Cyperus Digitaria
- Echinochloa Eleusine
- Lolium Monochoria
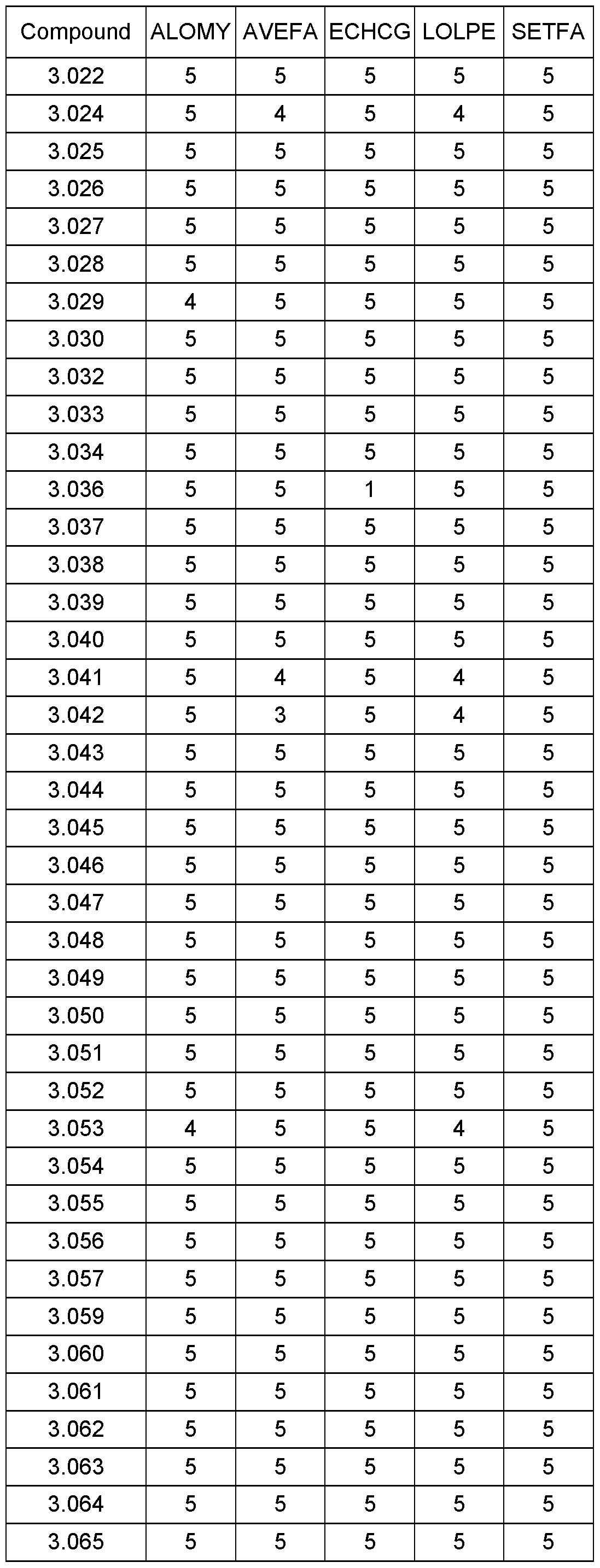
- the compounds of the present invention have been shown to exhibit particularly good activity against certain grass weed species, especially Lolium Perenne. Weeds can also include plants which may be considered crop plants but which are growing outside a crop area (‘escapes’), or which grow from seed left over from a previous planting of a different crop (‘volunteers’). Such volunteers or escapes may be tolerant to certain other herbicides.
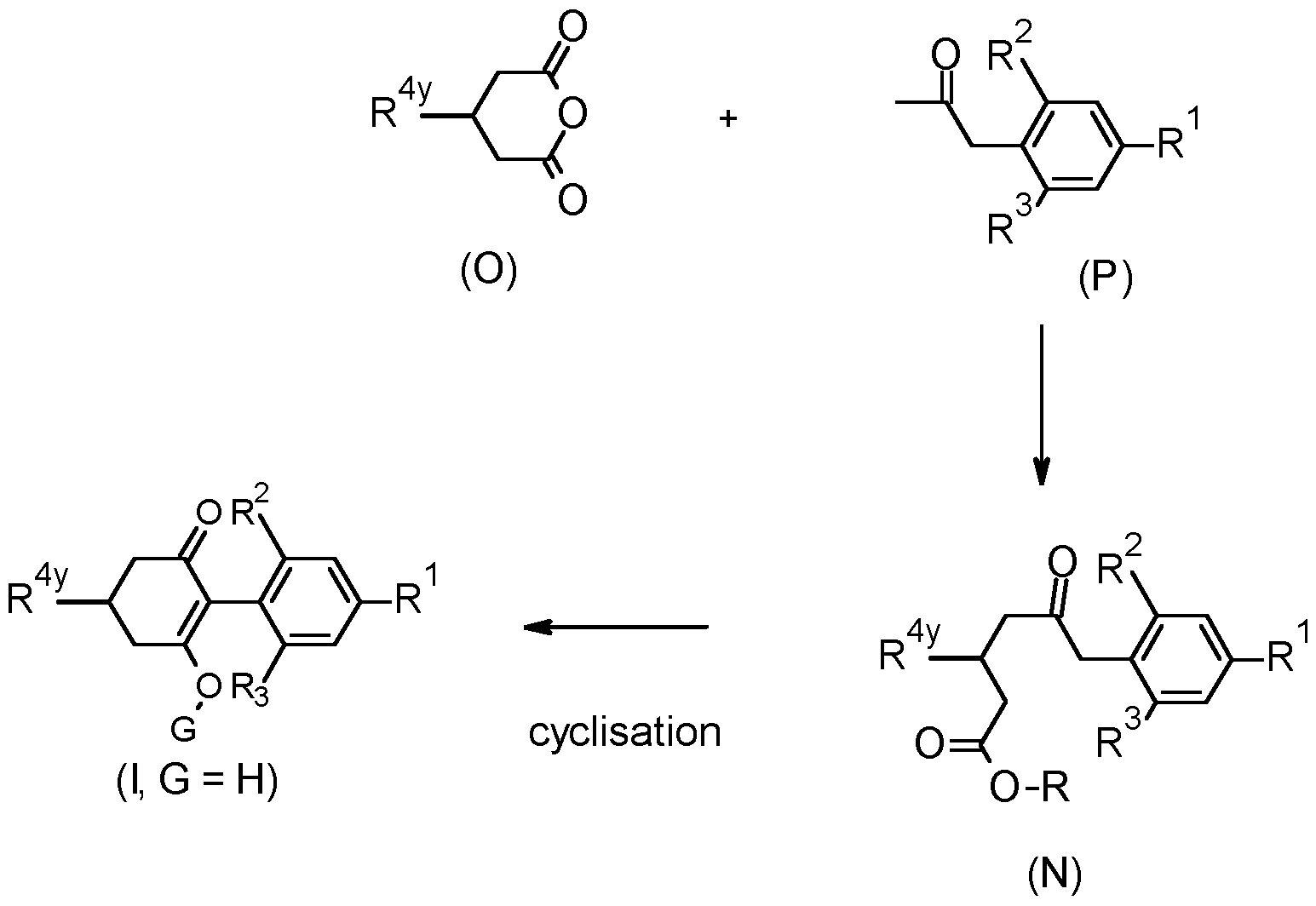
- the compounds of the present invention can be prepared according to the following schemes.
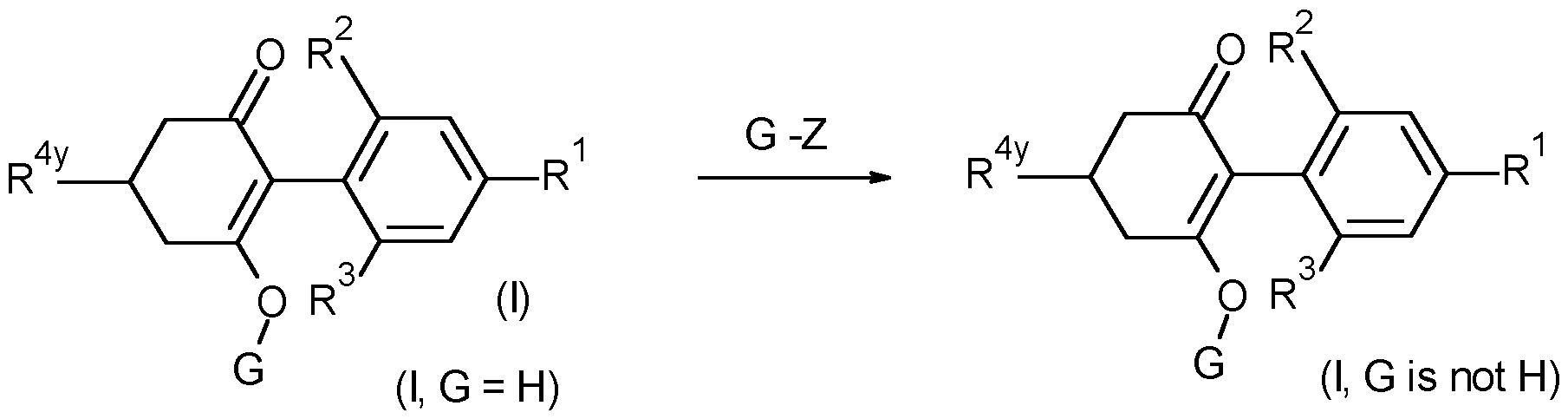
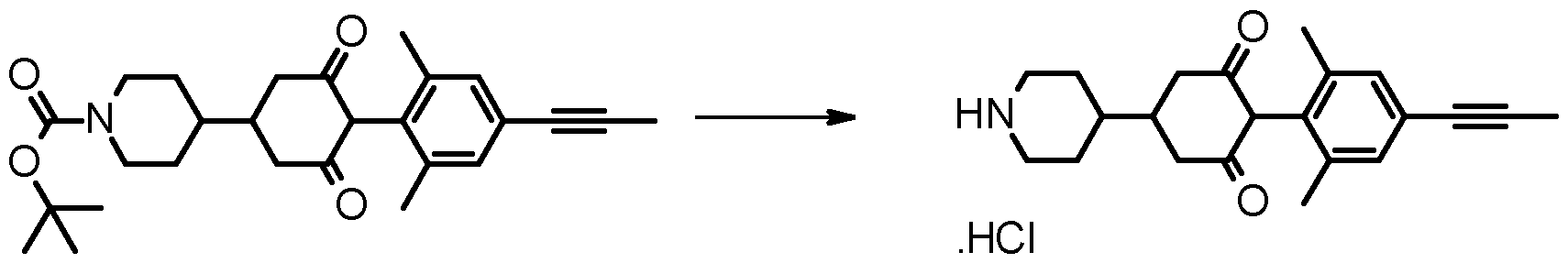
- Compounds of formula (I) wherein G is other than hydrogen may be prepared by treating a compound of formula (I) wherein G is hydrogen, with a reagent G-Z, wherein G-Z is an alkylating agent such as an alkyl halide, acylating agent such as an acid chloride or anhydride, sulfonylating agent such as a sulfonyl chloride, carbamylating agent such as a carbamoyl chloride, or carbonating agent such as a chloroformate, using known methods.
- G-Z is an alkylating agent such as an alkyl halide, acylating agent such as an acid chloride or anhydride, sulfonylating agent such as a sulfonyl chloride, carbamylating agent such as a carbamoyl chloride, or carbonating agent such as a chloroformate, using known methods.
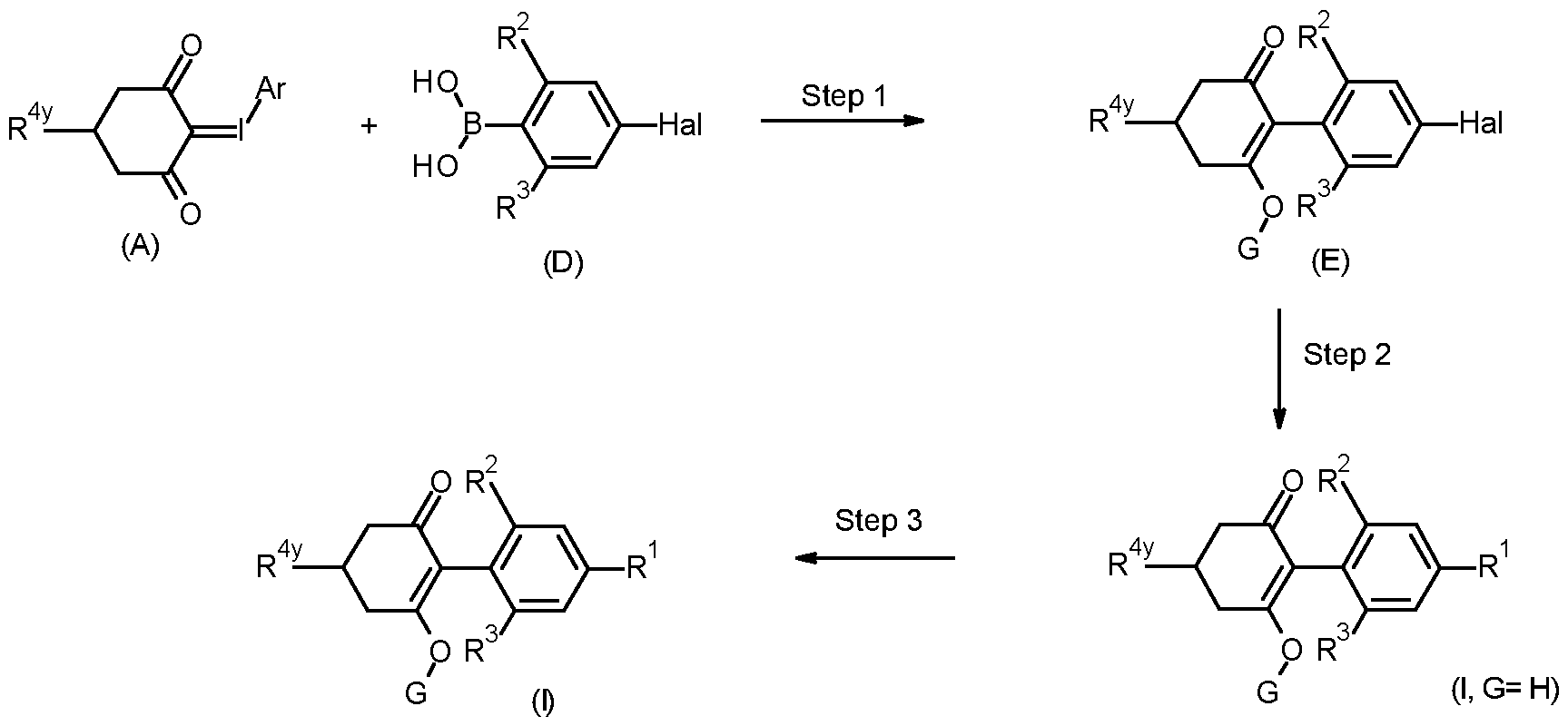
- Compounds of formula (I) may be prepared by reacting an iodonium ylide of formula (A), wherein Ar is an optionally substituted phenyl group, and an aryl boronic acid of formula (B), in the presence of a suitable palladium catalyst, a base and in a suitable solvent.
- Suitable palladium catalysts are generally palladium(ll) or palladium(O) complexes, for example palladium(ll) dihalides, palladium(ll) acetate, palladium(ll) sulfate, bis(triphenylphosphine)-palladium(ll) dichloride, bis(tricyclopentylphosphine)- palladium(ll) dichloride, bis(tricyclohexyl-phosphine)palladium(ll) dichloride, bis(dibenzylideneacetone)palladium(0) or tetrakis-(triphenylphosphine)palladium(0).
- palladium(ll) dihalides palladium(ll) acetate, palladium(ll) sulfate, bis(triphenylphosphine)-palladium(ll) dichloride, bis(tricyclopentylphosphine)- palladium(ll) dichloride, bis(tricyclo
- the palladium catalyst can also be prepared "in situ" from palladium(ll) or palladium(O) compounds by complexing with the desired ligands, by, for example, combining the palladium(ll) salt to be complexed, for example palladium(ll) dichloride (PdCh) or palladium(ll) acetate (Pd(OAc)2), together with the desired ligand, for example triphenylphosphine (PPhi3), tricyclopentylphosphine, tricyclohexylphosphine, 2-dicyclohexylphosphino-2’,6’-dimethoxybiphenyl or 2-dicyclohexylphosphino-2’,4’,6’- triisopropylbiphenyl and the selected solvent, with a compound of formula (N), the arylboronic acid of formula (O), and a base.
- PdCh palladium(ll) dichloride
- bidendate ligands for example 1 ,T-bis(diphenylphosphino)ferrocene or 1 ,2-bis(diphenylphosphino)ethane.
- the palladium(ll) complex or palladium(O) complex desired for the C-C coupling reaction is thus formed "in situ", and then initiates the C- C coupling reaction.
- the palladium catalysts are used in an amount of from 0.001 to 50 mol %, preferably in an amount of from 0.1 to 15 mol %, based on the compound of formula (N).
- the reaction may also be carried out in the presence of other additives, such as tetralkylammonium salts, for example, tetrabutylammonium bromide.
- tetralkylammonium salts for example, tetrabutylammonium bromide.
- the palladium catalyst is palladium acetate
- the base is lithium hydroxide
- the solvent is aqueous 1 ,2-dimethoxyethane.
- a compound of formula (A) may be prepared from a 1 ,3 dione compound of formula (C) by treatment with a hypervalent iodine reagent such as a (diacetoxy)iodobenzene or an iodosylbenzene and a base such as aqueous sodium carbonate, lithium hydroxide or sodium hydroxide in a solvent such as water or an aqueous alcohol such as aqueous ethanol using known procedures.
- a hypervalent iodine reagent such as a (diacetoxy)iodobenzene or an iodosylbenzene
- a base such as aqueous sodium carbonate, lithium hydroxide or sodium hydroxide in a solvent such as water or an aqueous alcohol such as aqueous ethanol using known procedures.
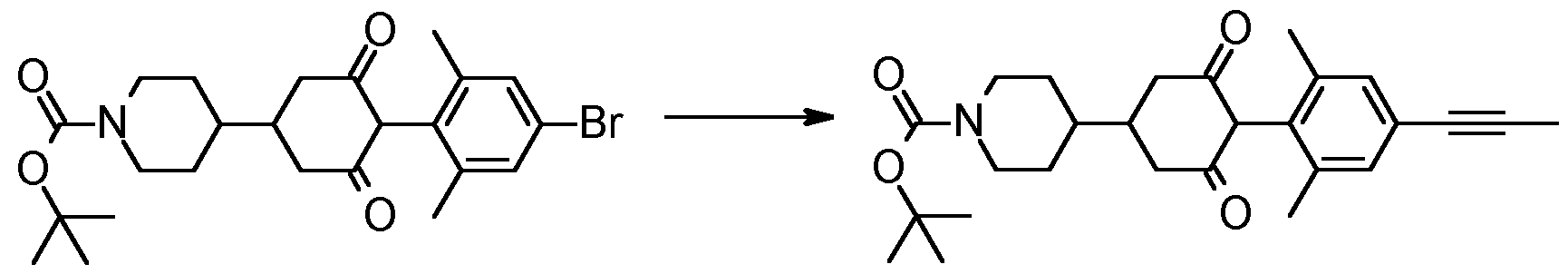
- the R 1 group may be added later in the synthetic sequence by decarboxylative propynylation (where R 1 is propyne, or alternative Suzuki/Stille or similar cross coupling where R 1 is phenyl or heteroaryl) such as in step 2 below.
- a compound of formula (B) or (D) may be prepared from an aryl halide of formula (F) or (H) by known methods.
- an aryl halide of formula (F) or (H) may be treated with an alkyl lithium or alkyl magnesium halide in a suitable solvent, preferably diethyl ether or tetrahydrofuran, at a temperature of between -80 °C and 30 °C, and the aryl magnesium or aryl lithium reagent obtained may then be reacted with a trialkyl borate (preferably tri methyl borate) to give an aryl dialkylboronate which may be hydrolysed to provide a boronic acid of formula (B) or (D) under acidic conditions.
- a trialkyl borate preferably tri methyl borate
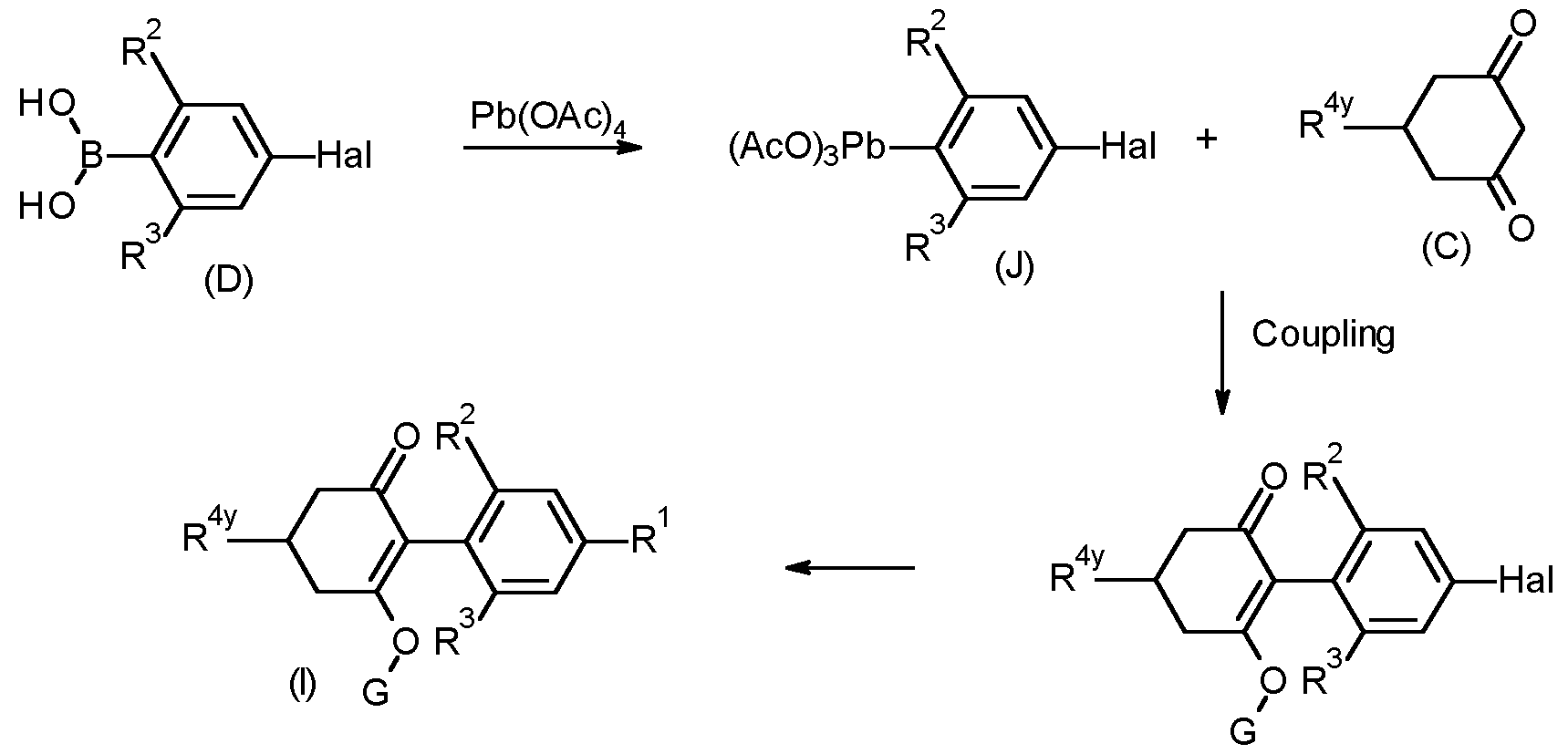
- Compounds of formula (I) can also be prepared via Pb coupling as shown in the scheme below by reacting a compound of formula (D), to form an organolead reagent of formula (J) and subsequent reaction with 1 ,3 dione (C) under conditions described, for example, by J. Pinhey, Pure and Appl. Chem., (1996), 68 (4), 819 and by M. Moloney et al., Tetrahedron Lett., (2002), 43, 3407.
- a suitable triarylbismuth compound under conditions described, for example, by A. Yu. Fedorov et al., Russ. Chem. Bull. Int. Ed., (2005), 54 (11), 2602, and by P. Koech and M. Kharide, J. Am. Chem. Soc., (2004), 126 (17), 5350 and references therein may be used as a related procedure.
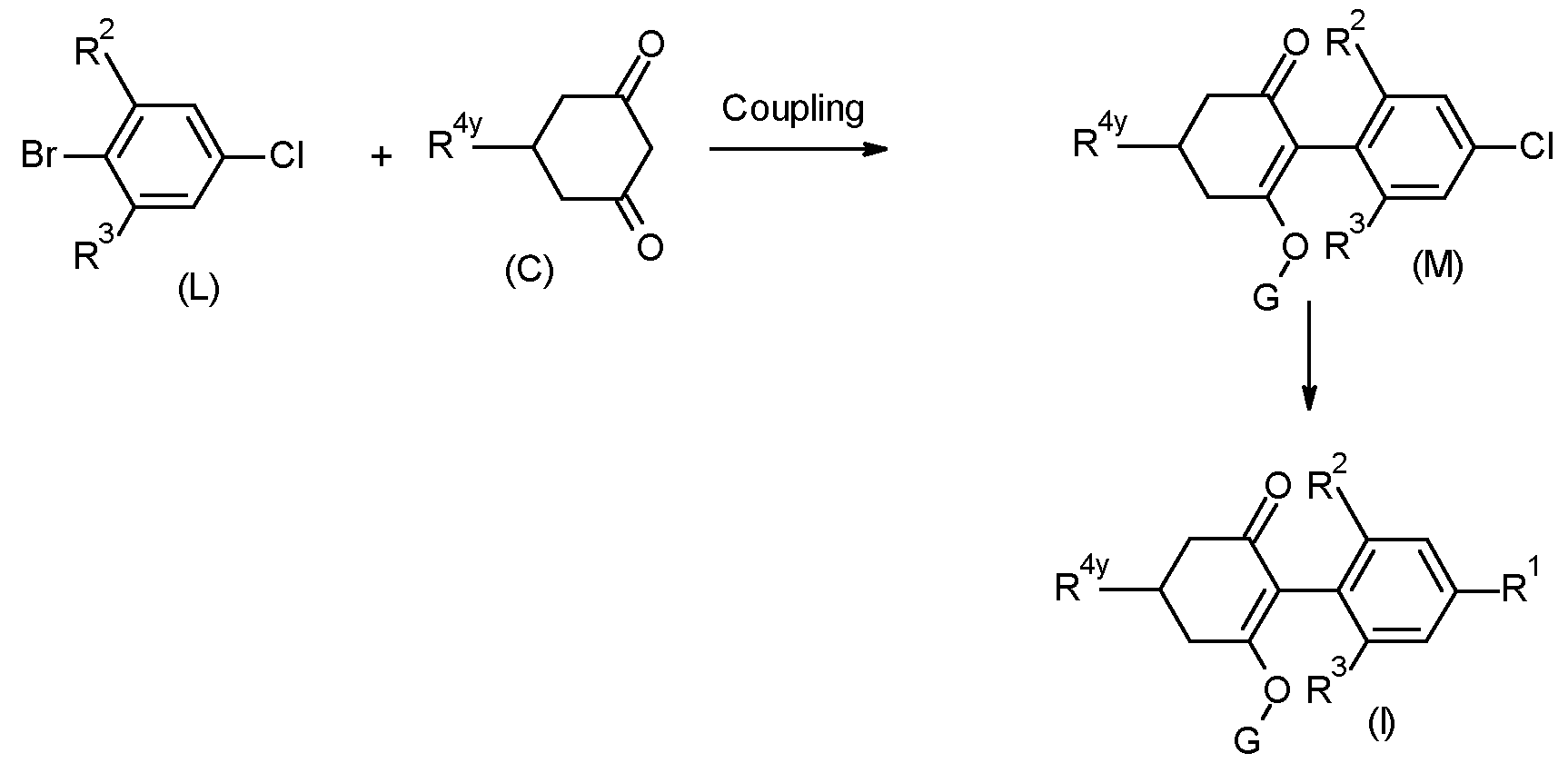
- the compounds of type (I) can also be prepared via palladium coupling as shown in the scheme below, where boronic acid of type (B) is coupled to the suitably protected halo-alkene of type (K) in a Suzuki type coupling.
- a suitable 1 ,3 dione may also be directly coupled to a Halo- compound (for example of formula (L)) with palladium catalysis.
- a Halo- compound for example of formula (L)
- Pladium catalysis Propynylation or arylation/Heteroarylation of intermediate (M) as described earlier gives compounds of type (I).
- the compounds of formula (N) have been particularly designed as intermediates in the synthesis of the compounds of the Formula (I).
- a compound of formula (N) wherein R is hydrogen may be cyclised under acidic conditions, preferably in the presence of a strong acid such as sulfuric acid, polyphosphoric acid or Eaton’s reagent, optionally in the presence of a suitable solvent such as acetic acid, toluene or dichloromethane.
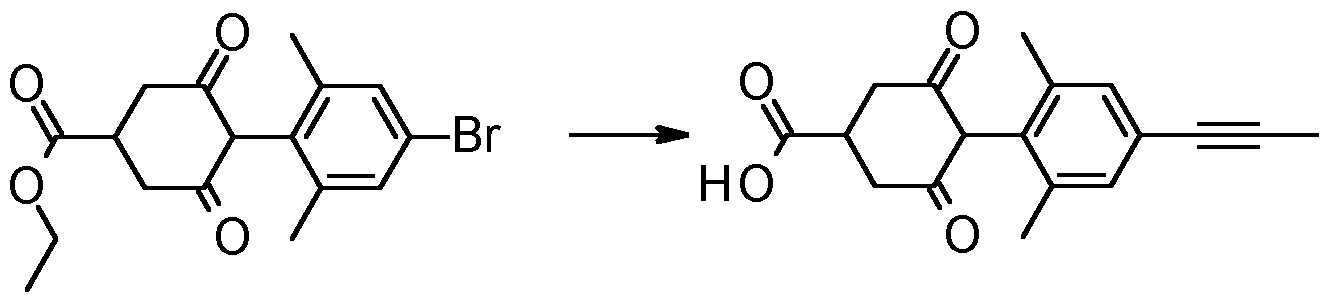
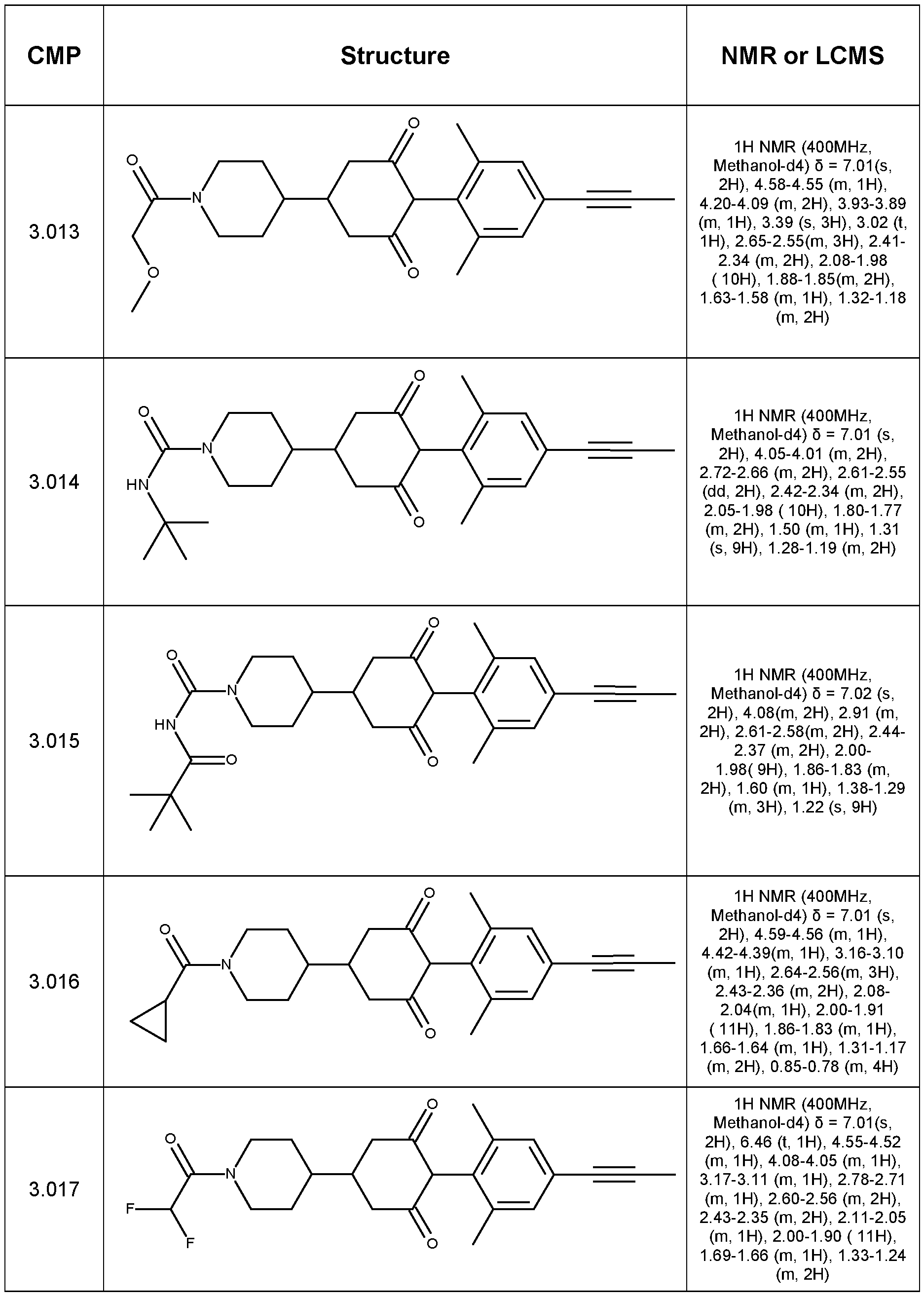
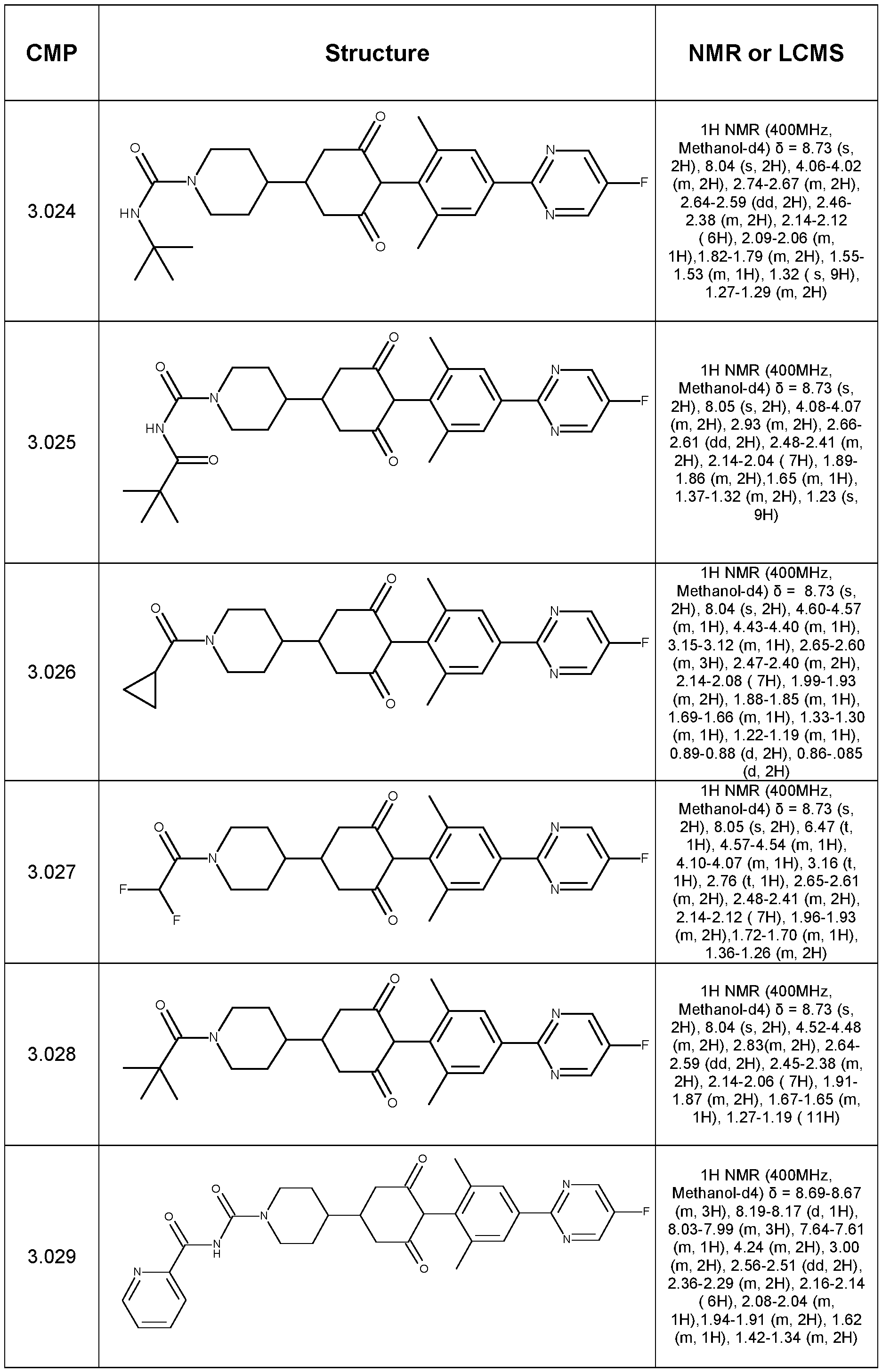
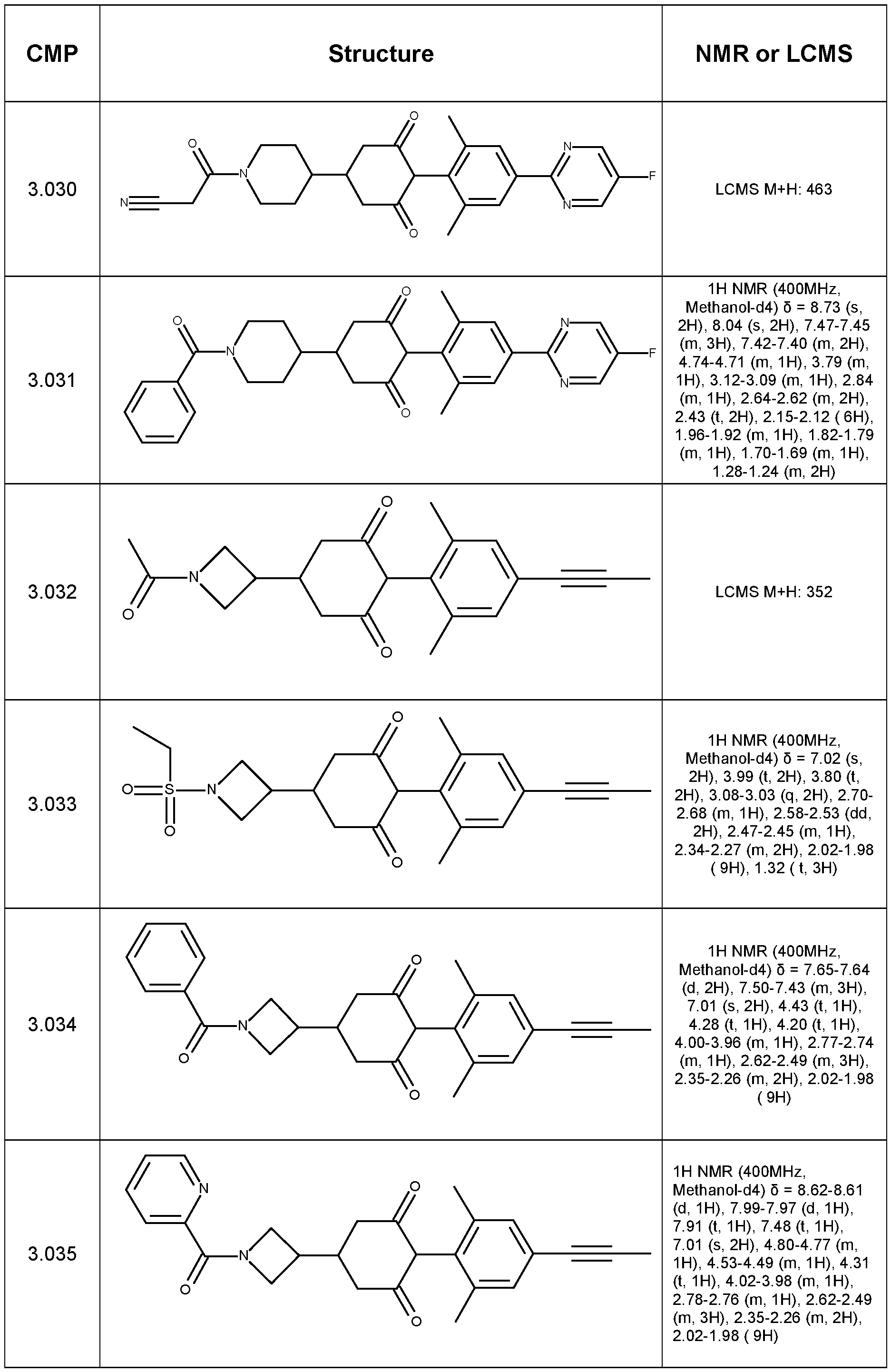
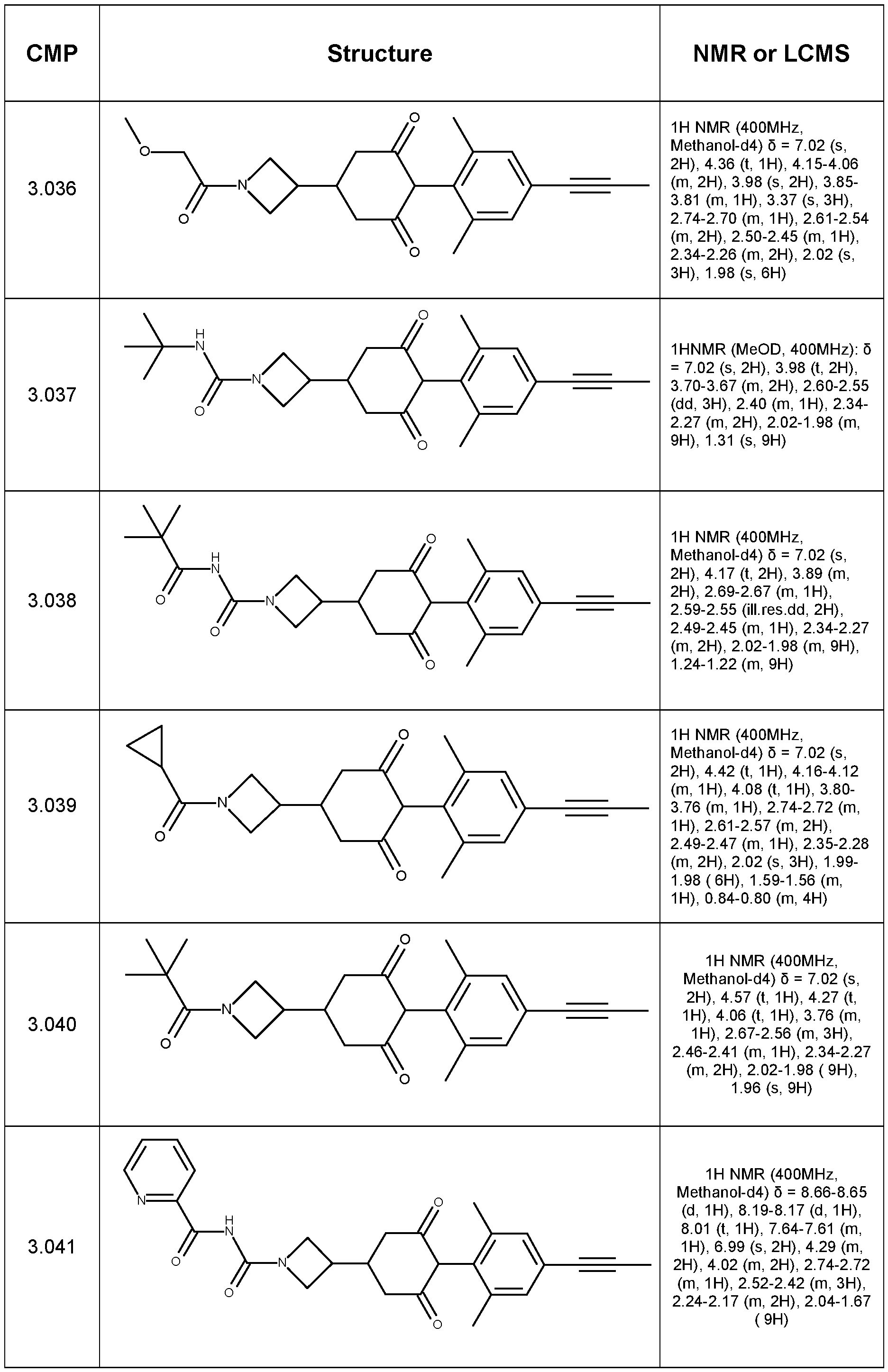
- Step 1 Synthesis of tert-butyl 9-(4-bromo-2,6-dimethyl-phenyl)-8,10-dioxo-3- azaspiro[5.5]undecane-3-carboxylate
- Step 2 Synthesis of ethyl 4-(2,6-dimethyl-4-prop-1-ynyl-phenyl)-3,5-dioxo- cyclohexanecarboxylate
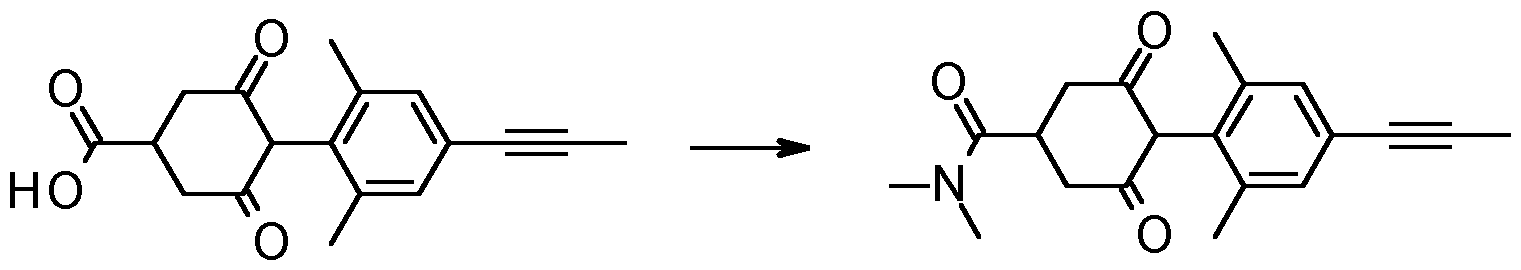
- Step 3 Synthesis of 4-(2,6-dimethyl-4-prop-1-ynyl-phenyl)-3,5-dioxo- cyclohexanecarboxylic acid
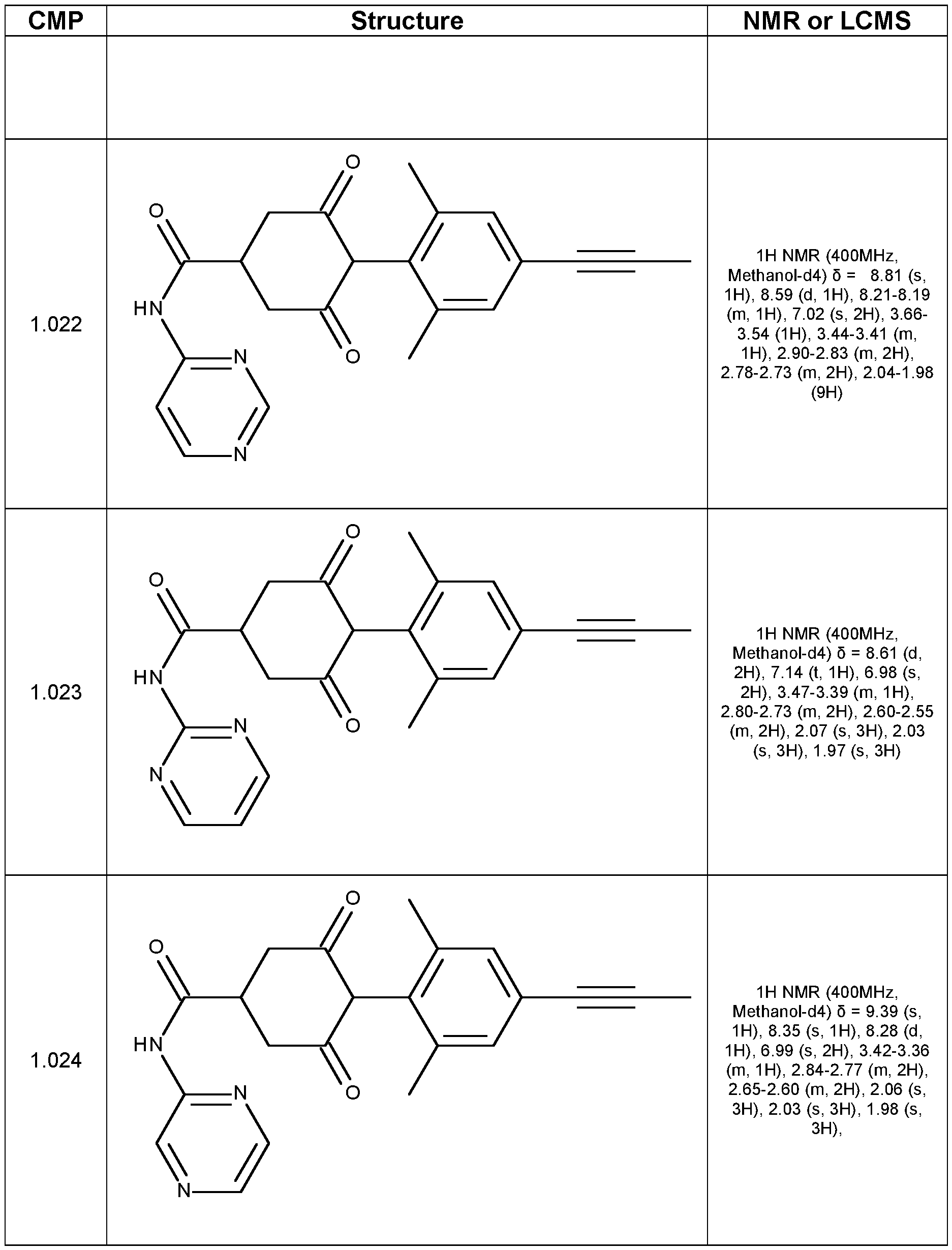
- Step 4 4-(2,6-dimethyl-4-prop-1-ynyl-phenyl)-N,N-dimethyl-3,5-dioxo- cyclohexanecarboxamide (Compound 1.001)
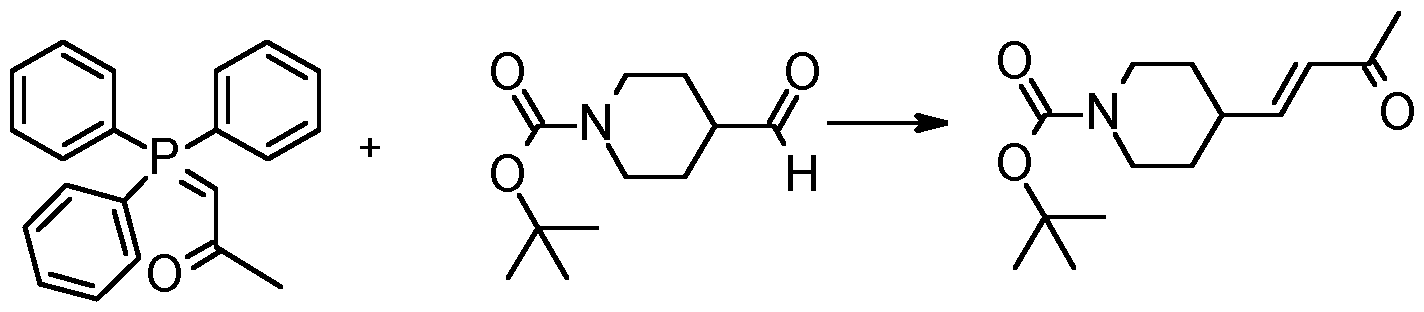
- Step 1 Te/f-butyl 4-[(E)-3-oxobut-1-enyl]piperidine-1-carboxylate
- Te/f-butyl 4-formylpiperidine-1-carboxylate (25.0 g, 117 mmol and 1- (triphenylphosphoranylidene)-2-propanone (37.3 g, 117 mmol) were dissolved in DCM (300ml_) and refluxed overnight. The reaction mixture was cooled and evaporated. The crude was purified by flash chromatography to give product as a yellow liquid (26.8 g).
- Step 2 Tert-butyl 4-(2-ethoxycarbonyl-3,5-dioxo-cyclohexyl)piperidine-1-carboxylate
- Te/f-butyl 4-(2-ethoxycarbonyl-3,5-dioxo-cyclohexyl)piperidine-1-carboxylate (36.0 g, 98.0 mmol) was taken in ethanol (88 ml_).
- Sodium hydroxide (12.0 M, 173 ml_, 2078 mmol) was added at RT and the reaction mixture was stirred over the weekend.
- the reaction mixture was concentrated and cooled in an ice bath then acidified with 10% citric acid solution.
- the mixture was saturated with solid NaCI then extracted with EtOAc (1 L x 3).
- the combined organics were washed with brine and dried over Na2SC>4.
- Step 4 Tert- butyl 4-[4-(4-bromo-2,6-dimethyl-phenyl)-3,5-dioxo- cyclohexyl]piperidine-1-carboxylate
- reaction mixture was acidified with 10% citric acid and the mixuture was filtered through a celite-bed.
- the celite-bed was washed with chloroform.
- the aqueous layer was separated and extracted with chloroform (x2).
- the combined organics were washed with brine, dried over Na2SC>4, filtered and evaporated to give a crude material , which was purified by flash chromatography to give product as a light brown foam.
- Step 5 Te/f-butyl 4-[4-(2,6-dimethyl-4-prop-1-ynyl-phenyl)-3,5-dioxo- cyclohexyl]piperidine-1-carboxylate
- a vacuum/argon back fill cycle was repeated twice, then anhydrous dimethyl sulfoxide (5.3 ml_) was added and again a vacuum/argon back fill cycle was repeated three times then 1 ,8-diazabicyclo[5.4.0]undec-7-ene (0.955 g, 6.27 mmol) was added to the reaction mixture. This was stirred at 120°C for 40h and then allowed stand at RT. The reaction mixture was diluted with water (10 ml) and extracted with ethyl acetate (20 ml x 1). The aqueous layer was then acidified with 10% citric acid to pH4, then saturated with NaCI and extracted with ethyl acetate (25 ml_ x 3).
- Step 6 2-(2,6-dimethyl-4-prop-1-ynyl-phenyl)-5-(4-piperidyl)cyclohexane-1 ,3-dione hydrochloride
- Step 7 [5-(1 -acetyl-4-piperidyl)-2-(2,6-dimethyl-4-prop-1 -ynyl-phenyl)-3-oxo- cyclohexen-1-yl] acetate
- Step 8 5-(1-acetyl-4-piperidyl)-2-(2,6-dimethyl-4-prop-1-ynyl-phenyl)cyclohexane- 1 ,3-dione (Compound 3.008)
- Seeds of a variety of test species are sown in standard soil in pots ( Lolium perenne (LOLPE), Setaria faberi (SETFA), Alopecurus myosuroides (ALOMY), Echinochloa crus-galli (ECHCG), Avena fatua (AVEFA)).
- LPE Lolium perenne
- SETFA Setaria faberi
- EHCG Echinochloa crus-galli
- AVEFA Avena fatua
- the plants After cultivation for one day (pre emergence) or after 8 days cultivation (post-emergence) under controlled conditions in a glasshouse (at 24/16°C, day/night; 14 hours light; 65 % humidity), the plants are sprayed with an aqueous spray solution derived from the formulation of the technical active ingredient in acetone / water (50:50) solution containing 0.5% Tween 20 (polyoxyethelyene sorbitan monolaurate, CAS RN 9005-64-5). Compounds are applied at 250 g/h. The test plants are then grown in a glasshouse under controlled conditions in a glasshouse (at 24/16°C, day/night; 14 hours light; 65 % humidity) and watered twice daily.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Environmental Sciences (AREA)
- Wood Science & Technology (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Zoology (AREA)
- Pest Control & Pesticides (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Plural Heterocyclic Compounds (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Pyridine Compounds (AREA)
Abstract
The present invention relates to compounds of Formula (I), wherein R1, R2, R3, R4x, R4y, k and G are as defined herein. The invention further relates to herbicidal compositions which comprise a compound of Formula (I), to their use for controlling weeds, in particular in crops of useful plants.
Description
CYCLOHEXANEDIONES AS HERBICIDAL COMPOUNDS
The present invention relates to novel herbicidal cyclohexanedione compounds, to processes for their preparation, to herbicidal compositions which comprise the novel compounds, and to their use for controlling weeds.
Herbicidal cyclic dione compounds substituted by a phenyl which has various substituents are disclosed in, for example, W02008/110308. The present invention relates to novel herbicidal cyclohexanedione derivatives with improved properties.
Thus, according to the present invention there is provided a compound of Formula (I)
R1 is selected from methyl, ethynyl, 1-propynyl, phenyl and a 5 or 6 membered heteroaryl which comprises one or two nitrogen heteroatoms, said phenyl and heteroaryl optionally substituted by one or two R15 substituents;
R2 is selected from the group consisting of methyl, ethyl, methoxy and chloro;
R3 is selected from the group consisting of methyl, ethyl, methoxy and chloro; k is O, 1 or 2;
R4x is selected from the group consisting of hydrogen, C1-C6 alkyl, Ci- Cehaloalkyl, Ci-CealkoxyCrCsalkyl- and Ci-CehaloalkoxyCrCsalkyl-;
R4y is selected from the group consisting of R4a, R4b, R4C, R4d and R4e;
R4aa is selected from the group consisting of hydrogen, C1-C6 alkyl and Ci- Cealkoxy-;
R4ab is selected from the group consisting of hydrogen, CrCealkyl, C2- Cealkenyl, C2-C6alkynyl, CrCehaloalkyl, hydroxy-, hydroxyCi-Cealkyl-, Ci- Cealkoxy, C3-C6cycloalkyl, -Ci-C4alkoxyCi-Cealkyl, -Ci-CsalkoxyCrCehaloalkyl, cyanoCrCealkyl-, C(0)R27, S(0)nR27, phenyl, -pyridyl and pyrimidinyl, wherein the phenyl, pyridyl or pyrimidinyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, C2-C3 alkenyl, C2-C3 alkynyl, halogen, cyano and nitro; or
R4aa and R4ab together form— (CH2)q-, -OCH2CH2CH2CH2-, -CH2OCH2C(CH3)2-, -CH2CH2X1CH2CH2- or -C(0)CH2X1CH2CH2- wherein X1 is selected from the group consisting of O, S(0)n and N-R28; and
R4ba is selected from the group consisting of hydrogen, C1-C4 alkyl and Ci- C4alkoxy-;
R4bb is selected from the group consisting of CrC4alkyl, Ci-C4alkoxy-, Ci- C4haloalkyl, -C(0)CrC4alkyl, -C(0)CrC4haloalkyl, hydroxyCi-C6alkyl-C(0)-, - S(0)nCi-Cealkyl, -S(0)nCi-C6haloalkyl, -S(0)n-(CH2)n-C3-C6cycloalkyl, S(0)nC(R11)R12R13, -C(0)H, -C(0)-(CH2)n-C3-C6cycloalkyl, -C(0)C(R11)R12R13, -C(0)C2-C4alkenyl, -C(O)(CR9R10)CN, -C(O)(CR9R10)(CR9R10)CN,
C(0)CH2C(0)-Ci-C6alkyl, -C(0)CH20C(0)-Ci-C6alkyl, -C(0)OCi-C6alkyl, -
C(0)OCi-C6haloalkyl, -C(0)(CH2)nS(0)nC1-C6alkyl -C(0)Ci-C3aikoxyCi- Cealkyl, -C(0)Ci-C3alkoxyC2-C6alkenyl, -C(0)Ci-C3alkoxyC2-C6alkynyl, - C(0)Ci-C3alkoxyCi-C6haloalkyl, -C(0)Ci-C3alkoxyC3-C6cycloalkyl, -C(0)OCi-
C(0)C(R23)=N-NR25R26, -(CH2)n-phenyl, -C(0)-(CH2)n-phenyl, -S(0)n-(CH2)n- phenyl, -heterocyclyl, -C(0)-(CH2)n-heterocyclyl, -C(0)(CH2)n0-(CH2)n- heterocyclyl, -S(0)n-(CH2)n-heterocyclyl, wherein each heterocyclyl is a 5- or 6- membered heterocyclyl which may be aromatic, saturated or partially saturated and can contain from 1 to 4 heteroatoms each independently selected from the group consisting of oxygen, nitrogen and sulphur, and wherein said heterocyclyl or phenyl groups are optionally substituted by one, two or three substituents independently selected from the group consisting of CrC3alkyl, CrCshaloalkyl, CrCsalkoxy, C2-C3alkenyl, C2-C3alkynyl, halogen, cyano and nitro; p is 0, 1 or 2; r is 0, 1 or 2; wherein p+r = 0, 1 or 2;
R5 is independently selected from the group consisting of hydrogen and C1-C6 alkyl;
R6 is selected from the group consisting of hydrogen, CrCealkyl, C2-C6alkenyl, C2-C6alkynyl, CrCehaloalkyl, hydroxyl-, CrCealkoxy, C3-C6 cycloalkyl, , -Ci- C4alkoxyCi-C6alkyl, -Ci-CsalkoxyCrCehaloalkyl, -(CR9R10)Ci-C6haloalkyl, - (CR9R10)C(O)NR5R5, phenyl, -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, C2-C3 alkenyl, C2-C3 alkynyl, halogen, cyano and nitro; or
R5 and R6 together form -CH2CH2OCH2CH2-; and
R7 is selected from the group consisting of hydrogen and C1-C6 alkyl;
R8 is selected from the group consisting of hydrogen, C1-C6 alkyl, C1-C6 alkoxy, C3-C6 cycloalkyl, phenyl, -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, C2-C3 alkenyl, C2-C3 alkynyl, halogen, cyano and nitro;
R9 is hydrogen or methyl;
R1 0 is hydrogen or methyl; or
R9 and R1 0 together form -CH2CH2-; and
R1 1 is hydrogen or methyl;
R1 2 is selected from the group consisting of hydrogen, C1-C6 alkyl, hydroxyl and C1-C6 alkoxy-;
R1 3 is selected from the group consisting of hydrogen, C1-C6 alkyl, hydroxyl and C1-C6 alkoxy; or
R1 2 and R1 3 together form -CH2-X2-CH2- wherein X2 is selected from the group consisting of O, S and N-R14; and
R1 4 is selected from the group consisting of hydrogen, C1-C3 alkyl and C1-C3 alkoxy-;
R1 5 is independently selected from the group consisting of C1-C4 alkyl, C1-C4 haloalkyl, cyano and halogen;
R16 is hydrogen or CrCealkyl; and
R17 is selected from the group consisting of hydrogen, CrCealkyl, C3- Cecycloalkyl, CrCe alkoxy-CrCsalkyl-, -C(0)CrCealkyl, -C(0)OCrCealkyl and CH2CN; or
R1 6 and R1 7 together form -CH2CH2OCH2CH2-, -CH2CH2S(0)2CH2CH2-; and
R18 is hydrogen or CrCealkyl;
R1 9 is selected from the group consisting of hydrogen, C1-C6 alkyl, C1-C6 alkoxy, C3-C6cycloalkyl, phenyl, -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, C2-C3 alkenyl, C2-C3 alkynyl, halogen, cyano and nitro;
R20 is selected from the group consisting of CrCealkyl, CrCehaloalkyl, Cr Cealkoxy-, CrCehaloalkoxy, -NR21R22, phenyl and -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, C2-C3 alkenyl, C2-C3 alkynyl, halogen, cyano and nitro;
R21 is selected from the group consisting of hydrogen, C1-C6 alkyl, C1-C6 alkoxy, CrCealkoxyCrCsalkyl-, C3-C6 cycloalkyl, CrCehaloalkyl- and Cr Cehaloalkoxy-, -C(0)CrCealkyl, phenyl, -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, C2-C3 alkenyl, C2-C3 alkynyl, halogen, cyano and nitro;
R22 is hydrogen or CrCealkyl; or
R21 and R22 together form -CH2CH2OCH2CH2-; and
R23 is selected from the group consisting of hydrogen, CrCealkyl, Cr
Cehaloalkyl, CrCealkoxy- and CrCehaloalkoxy-;
R24 is selected from the group consisting of hydrogen, CrCealkyl, Cr
C6alkoxyCrC3alkyl-, C3-Cecycloalkyl, -CH2CN, tetrahydropyranyl-, phenyl and -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of Cr Cealkyl, CrCehaloalkyl, CrCealkoxy, C2-C3alkenyl, C2-C3alkynyl, halogen, cyano and nitro;
R25 is hydrogen or C1-C6 alkyl;
R26 is hydrogen or C1-C6 alkyl; and
R4ca is selected from hydrogen, CrCealkyl, C2-Cealkenyl, C2-C6alkynyl, Ci- Cehaloalkyl, hydroxyl-, CrCealkoxy, C3-Cecycloalkyl, -Ci-C4alkoxyCi-Cealkyl, - Ci-C3alkoxyCrC6haloalkyl, cyanoC1-C6alkyl-. C(0)R27, S(0)nR27, phenyl, - pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of Ci- C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, C2-C3 alkenyl, C2-C3 alkynyl, halogen, cyano and nitro;
R4cb is selected from the group consisting of hydrogen, CrCe alkyl and Ci- Cealkoxy-; or
R4ca and R4cb together form — (CH2)q-, -CH2CH2X1CH2CH2- or - C(0)CH2X1CH2CH2- wherein X1 is selected from the group consisting of O, S(0)n and N-R28;
R27 is selected from the group consisting of hydrogen, CrCe alkyl, CrCe haloalkyl, C2-Cealkenyl, C2-Cealkynyl, CrCe alkoxy, C3-C6 cycloalkyl, phenyl, -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of Ci- C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, C2-C3 alkenyl, C2-C3 alkynyl, halogen, cyano and nitro;
R28 is selected from the group consisting of hydrogen, C1-C3 alkyl, -C(0)Ci- Csalkyl and C1-C3 alkoxy-;
R4ea is R4ea1 or R4ea2
G is selected from the group consisting of hydrogen, -(Chb R3, -C(0)-Ra, - C(0)-(CRcRd)n-0-Rb, -C(0)-(CRcRd)n-S-Rb, -C(0)NRaRa, -S(0)2-Ra and Cr Csalkoxy-Ci-Csalkyl-;
Ra is independently selected from the group consisting of hydrogen, Cr Csalkyl, CrCshaloalkyl, C2-Csalkenyl, C2-Csalkynyl, C3-C6cycloalkyl, heterocyclyl and phenyl wherein said heterocyclyl and phenyl groups are optionally substituted by one, two or three substituents independently selected from the group consisting of CrCsalkyl, CrCshaloalkyl, CrCsalkoxy, C2-Csalkenyl, C2-Csalkynyl, halogen, cyano and nitro;
Rb is selected from the group consisting of CrCsalkyl, CrCshaloalkyl, C2- Csalkenyl, C2-Csalkynyl, C3-C6 cycloalkyl, heterocyclyl and phenyl wherein said heterocyclyl and phenyl groups are optionally substituted by one, two or three substituents independently selected from the group consisting of Cr Csalkyl, CrCshaloalkyl, CrCsalkoxy, C2-C3alkenyl, C2-C3alkynyl, halogen, cyano and nitro;
Rc is hydrogen or C1-C3 alkyl; and
Rd is hydrogen or C1-C3 alkyl; or an agriculturally acceptable salt thereof.
Alkyl groups (e.g CrCsalkyl) include, for example, methyl (Me, CH3), ethyl (Et, C2H5), n-propyl (n-Pr), isopropyl (/-Pr), n-butyl (n-Bu), isobutyl (/- Bu), sec-butyl (s-Bu) and tert- butyl (f-Bu).
Alkenyl and alkynyl moieties can be in the form of straight or branched chains, and the alkenyl moieties, where appropriate, can be of either the (£)- or (Z)-configuration. Examples are vinyl, allyl and propargyl. Alkenyl and alkynyl moieties can contain one or more double and/or triple bonds in any combination.
Halogen (or halo) encompasses fluorine, chlorine, bromine or iodine. The same correspondingly applies to halogen in the context of other definitions, such as haloalkyl.
Haloalkyl groups (e.g CrCehaloalkyl) are, for example, fluoromethyl, difluoromethyl, trifluoromethyl, chloromethyl, dichloromethyl, trichloromethyl, 2,2,2- trifluoroethyl, 2-fluoroethyl, 2-chloroethyl, pentafluoroethyl, 1 , 1 -difluoro-2,2,2- trichloroethyl, 2,2,3,3-tetrafluoroethyl and 2,2,2-trichloroethyl, heptafluoro-n-propyl and peril uoro-n-hexyl.
Alkoxy groups (e.g Ci-C4alkoxy-) are, for example, methoxy, ethoxy, propoxy, isopropoxy, n-butoxy, isobutoxy, sec-butoxy or tert-butoxy, preferably methoxy and ethoxy.
Alkoxyalkyl groups (e.g Ci-Csalkoxy-CrCsalkyl-) includes, for example, methoxymethyl, methoxyethyl, ethoxymethyl, ethoxyethyl, n-propoxymethyl, n- propoxyethyl, isopropoxymethyl or isopropoxyethyl.
Cycloalkyl groups (e.g C3-C6cycloalkyl-) include, for example cyclopropyl (c- propyl, c-Pr), cyclobutyl (c-butyl, c-Bu), cyclopentyl (c-pentyl) and cyclohexyl (c-hexyl) and may be substituted or unsubstituted as indicated.
CrC6alkyl-S- (alkylthio) is, for example, methylthio, ethylthio, propylthio, isopropylthio, n-butylthio, isobutylthio, sec-butylthio or tert-butylthio, preferably methylthio or ethylthio.
Ci-C6alkyl-S(0)- (alkylsulfinyl) is, for example, methylsulfinyl, ethylsulfinyl, propylsulfinyl, isopropylsulfinyl, n-butylsulfinyl, isobutylsulfinyl, sec-butylsulfinyl or tert-butylsulfinyl, preferably methylsulfinyl or ethylsulfinyl.
CrC6alkyl-S(0)2- (alkylsulfonyl) is, for example, methylsulfonyl, ethylsulfonyl, propylsulfonyl, isopropylsulfonyl, n-butylsulfonyl, isobutylsulfonyl, sec-butylsulfonyl or tert-butylsulfonyl, preferably methylsulfonyl or ethylsulfonyl.
Heterocyclyl, unless stated otherwise, is a 5- or 6- membered heterocyclyl which may be aromatic, saturated or partially saturated and can contain from 1 to 4
heteroatoms each independently selected from the group consisting of oxygen, nitrogen and sulphur.
The invention also relates agriculturally acceptable salts of the compounds of Formula (I). Such salts include those which are able to form with amines, alkali metal and alkaline earth metal bases or quaternary ammonium bases. Among the alkali metal and alkaline earth metal hydroxides as salt formers, special mention should be made of the hydroxides of lithium, sodium, potassium, magnesium and calcium, but especially the hydroxides of sodium and potassium. The compounds of Formula (I) according to the invention also include hydrates which may be formed during the salt formation.
Examples of amines suitable for ammonium salt formation include ammonia as well as primary, secondary and tertiary CrCisalkylamines, Ci- C4hydroxyalkylamines and C2-C4alkoxyalkylamines, for example methylamine, ethylamine, n-propylamine, isopropylamine, the four butylamine isomers, n- amylamine, isoamylamine, hexylamine, heptylamine, octylamine, nonylamine, decylamine, pentadecylamine, hexadecylamine, heptadecylamine, octadecylamine, methylethylamine, methylisopropylamine, methylhexylamine, methylnonylamine, methylpentadecylamine, methyloctadecylamine, ethylbutylamine, ethylheptylamine, ethyloctylamine, hexylheptylamine, hexyloctylamine, dimethylamine, diethylamine, di- n-propylamine, diisopropylamine, di-n-butylamine, di-n-amylamine, diisoamylamine, dihexylamine, diheptylamine, dioctylamine, ethanolamine, n-propanolamine, isopropanolamine, A/./V-diethanolamine, /V-ethylpropanolamine, /V-butylethanolamine, allylamine, n-but-2-enylamine, n-pent-2-enylamine, 2,3-dimethylbut-2-enylamine, dibut-2-enylamine, n-hex-2-enylamine, propylenediamine, trimethylamine, triethylamine, tri-n-propylamine, triisopropylamine, tri-n-butylamine, triisobutylamine, tri-sec-butylamine, tri-n-amylamine, methoxyethylamine and ethoxyethylamine; heterocyclic amines, for example pyridine, quinoline, isoquinoline, morpholine, piperidine, pyrrolidine, indoline, quinuclidine and azepine; primary arylamines, for example anilines, methoxyanilines, ethoxyanilines, o-, m- and p-toluidines, phenylenediamines, benzidines, naphthylamines and o-, m- and p-chloroanilines; but especially triethylamine, isopropylamine and diisopropylamine.
In one embodiment of the present invention R1 is 1-propynyl.
In another embodiment of the present invention R1 is phenyl optionally substituted by one or two R15 substituents, e.g selected from the group consisting of cyano, chloro and fluoro.
In another embodiment of the present invention R1 is a 5 or 6 membered heteroaryl which comprises one or two nitrogen heteroatoms, said heteroaryl optionally substituted by one or two R15 substituents, e.g selected from the group consisting of cyano, chloro and fluoro. In a preferred embodiment, said heteroaryl is selected from the group consisting of pyridyl, pyrimidinyl, and pyrazolyl.
In one embodiment of the present invention R2 is methoxy, chloro or methyl. In a preferred embodiment of the present invention R2 is methyl.
In one embodiment of the present invention R3 is methyl or methoxy, preferably methyl.
In a preferred embodiment of the present invention R2 is methyl and R3 is methyl, R2 is methoxy and R3 is methyl or R2 is chloro and R3 is methoxy. In a particularly preferred embodiment, R2 is methyl and R3 is methyl.
In one embodiment of the present invention k is 0 or 1. In one embodiment of the present invention k is 0. In another embodiment of the present invention k is 1.
In one embodiment of the present invention, R4x is hydrogen or C1-C6 alkyl (e.g methyl), most preferably hydrogen.
In one embodiment of the present invention, R4y is R4a. In this embodiment R4aa is preferably hydrogen or methyl and R4ab is preferably selected from the group consisting of C Cealkyl (e.g methyl, f-butyl), pyridyl, -C(0)CH2X1CH2CH2- (wherein X1 is O), -CH2CH2X1CH2CH2- (wherein X1 is O, N-R28 wherein R28 is -C(0)CrC3alkyl (preferablky methyl), or S(0)n wherein n is 2), hydroxyCi-Cealkyl- (e.g HO-CH2CH2-), CrCealkoxy (e.g MeO), -Ci-C4alkoxyCrC6alkyl (e.g CH3OCH2CH2-) and cyanoCr Cealkyl- (e.g -C(CH3)(C2H5)(CN).
In another embodiment of the present invention R4y is R4b . In this embodiment R4ba is preferably hydrogen or methyl and R4bb is selected from the group consisting of -C(0)Ci-C4alkyi (e.g -C(0)CH3, -C(O)f-butyl), -C(0)-(CH2)n-C3-C6cycloalkyl (e.g -
C(O)-cyclopropyl), -C(0)-heterocyclyl (e.g pyridyl), -C(0)-(CH2)n-phenyl, C(0)(CH2)nNR5R6, -C(0)Ci-C3alkoxyCi-C6haloalkyl (e.g CHF2-0-CH2-), -
C(O)(CR9R10)CN (e.g -C(0)CH2CN), -C(O)(CR9R10)(CR9R10)CN (e.g -
C(0)CH2CH2CN), -C(0)Ci-C4haloalkyl (e.g -C(0)CHF2), hydroxyCrCealkyl-C O)- C Cealkyl (e.g CH30CH2C(0)-) and -C(O)-
In another embodiment of the present invention, R4y is R4d. In this embodiment, p and r are preferably both 0 or both 1. Furthermore, in this embodiment R4bb is preferably selected from the group consisting of is selected from the group consisting of CrC4alkoxy- (e.g methoxy-), -C(0)CrC4alkyl (e.g -C(0)CH3, -C(O)f-butyl), -C(0)-(CH2)n-C3-C6cycloalkyl (e.g -C(O)-cyclopropyl), -C(O)- heterocyclyl (e.g pyridyl), -C(0)-(CH2)n-phenyl, -C(0)(CH2)nNR5R6, -C(0)Ci- CsalkoxyCrCehaloalkyl (e.g CHF2-0-CH2-), -C(O)(CR9R10)CN (e.g -C(0)CH2CN), - C(O)(CR9R10)(CR9R10)CN (e.g -C(0)CH2CH2CN), -C^CrC^aloalkyl (e.g - C(0)CHF2), hydroxyCi-C6alkyi-C(0)- (e.g HOCH(CH3)C(0)-), -C(0)CH20C(0)-Cr Cealkyl (e.g CH30CH2C(0)-) and -C(0)-(CH2)n-NR7C(0)R8.
In another embodiment of the present invention, R4y is R4e.
In one embodiment, each aforementioned heterocyclyl is an aromatic heterocyclyl (i.e heteroaryl), more preferably selected from the group consisting of furanyl, pyrrolyl, thiophenyl, imidazolyl, pyrazolyl, oxazolyl, isoxazolyl, thiazolyl, pyranyl, pyridyl, pyrazinyl, pyrimidinyl, pyridazinyl, and triazolyl more preferably selected from the group consisting of pyridyl, pyridazinyl, pyrimidinyl and pyrazinyl each of which is optionally substituted as defined previously. In another embodiment, each aforementioned heterocyclyl is a partially saturated heterocyclyl, more preferably selected from the group consisting of imidazolinyl, isoxazolinyl and thiazolinyl each of which is optionally substituted as defined previously. In another embodiment, each aforementioned heterocyclyl is a saturated heterocyclyl more preferably selected from the group consisting of morpholinyl, tetrahydrofuryl and tetrahydropyranyl each of which is optionally substituted as defined previously.
In one embodiment of the present invention R5 is hydrogen or methyl, preferably hydrogen.
In one embodiment of the present invention R6 is C1-C6 alkyl (e.g methyl) or pyridyl.
In one embodiment of the present invention R7 is hydrogen or methyl, preferably hydrogen.
In one embodiment of the present invention R8 is C1-C6 alkyl (e.g f-butyl) or pyridyl.
In a preferred embodiment of the present invention R9 is hydrogen.
In a preferred embodiment of the present invention R10 is hydrogen.
In one embodiment of the present invention, G is selected from the group consisting of hydrogen, CrCsalkyl (e.g methyl, ethyl, n-propyl, /-propyl, n-butyl, f-butyl, -C2-Csalkenyl (e.g vinyl), C2-Csalkynyl (e.g propargyl), -C(0)Ci-Csalkyl (more preferably -C(0)Ci-C6alkyl e.g -C(0)/-propyl and -C(O)f-butyl) and -C(0)-0-Ci- Csalkyl (more preferably -C(0)-0-Ci-C6alkyl e.g -C(O)-O-methyl). In a preferred embodiment, G is hydrogen.
Depending on the nature of the substituents, compounds of Formula (I) may exist in different isomeric forms. When G is hydrogen, for example, compounds of Formula (I) may exist in different tautomeric forms.
This invention covers all such isomers and tautomers and mixtures thereof in all proportions. Also, when substituents contain double bonds, cis- and trans- isomers can exist. These isomers, too, are within the scope of the claimed compounds of the Formula (I). Compounds of Formula (I) may contain asymmetric centres and may be present as a single enantiomer, pairs of enantiomers in any proportion or, where more than one asymmetric centre are present, contain diastereoisomers in all possible ratios. Typically one of the enantiomers has enhanced biological activity compared to the other possibilities.
The compounds of Formula (I) according to the invention can be used as herbicides by themselves, but they are generally formulated into herbicidal compositions using formulation adjuvants, such as carriers, solvents and surface- active agents (SFAs). Thus, the present invention further provides a herbicidal
composition comprising a herbicidal compound according to any one of the previous claims and an agriculturally acceptable formulation adjuvant. The composition can be in the form of concentrates which are diluted prior to use, although ready-to-use compositions can also be made. The final dilution is usually made with water, but can be made instead of, or in addition to, water, with, for example, liquid fertilisers, micronutrients, biological organisms, oil or solvents.
The herbicidal compositions generally comprise from 0.1 to 99 % by weight, especially from 0.1 to 95 % by weight, compounds of Formula (I) and from 1 to 99.9 % by weight of a formulation adjuvant which preferably includes from 0 to 25 % by weight of a surface-active substance.
The compositions can be chosen from a number of formulation types, many of which are known from the Manual on Development and Use of FAO Specifications for Plant Protection Products, 5th Edition, 1999. These include dustable powders (DP), soluble powders (SP), water soluble granules (SG), water dispersible granules (WG), wettable powders (WP), granules (GR) (slow or fast release), soluble concentrates (SL), oil miscible liquids (OL), ultra low volume liquids (UL), emulsifiable concentrates (EC), dispersible concentrates (DC), emulsions (both oil in water (EW) and water in oil (EO)), micro-emulsions (ME), suspension concentrates (SC), aerosols, capsule suspensions (CS) and seed treatment formulations. The formulation type chosen in any instance will depend upon the particular purpose envisaged and the physical, chemical and biological properties of the compound of Formula (I).
Dustable powders (DP) may be prepared by mixing a compound of Formula (I) with one or more solid diluents (for example natural clays, kaolin, pyrophyllite, bentonite, alumina, montmorillonite, kieselguhr, chalk, diatomaceous earths, calcium phosphates, calcium and magnesium carbonates, sulphur, lime, flours, talc and other organic and inorganic solid carriers) and mechanically grinding the mixture to a fine powder.
Soluble powders (SP) may be prepared by mixing a compound of Formula (I) with one or more water-soluble inorganic salts (such as sodium bicarbonate, sodium carbonate or magnesium sulphate) or one or more water-soluble organic solids (such as a polysaccharide) and, optionally, one or more wetting agents, one or more dispersing agents or a mixture of said agents to improve water dispersibility/solubility.
The mixture is then ground to a fine powder. Similar compositions may also be granulated to form water soluble granules (SG).
Wettable powders (WP) may be prepared by mixing a compound of Formula (I) with one or more solid diluents or carriers, one or more wetting agents and, preferably, one or more dispersing agents and, optionally, one or more suspending agents to facilitate the dispersion in liquids. The mixture is then ground to a fine powder. Similar compositions may also be granulated to form water dispersible granules (WG).
Granules (GR) may be formed either by granulating a mixture of a compound of Formula (I) and one or more powdered solid diluents or carriers, or from pre formed blank granules by absorbing a compound of Formula (I) (or a solution thereof, in a suitable agent) in a porous granular material (such as pumice, attapulgite clays, fuller's earth, kieselguhr, diatomaceous earths or ground corn cobs) or by adsorbing a compound of Formula (I) (or a solution thereof, in a suitable agent) on to a hard core material (such as sands, silicates, mineral carbonates, sulphates or phosphates) and drying if necessary. Agents which are commonly used to aid absorption or adsorption include solvents (such as aliphatic and aromatic petroleum solvents, alcohols, ethers, ketones and esters) and sticking agents (such as polyvinyl acetates, polyvinyl alcohols, dextrins, sugars and vegetable oils). One or more other additives may also be included in granules (for example an emulsifying agent, wetting agent or dispersing agent).
Dispersible Concentrates (DC) may be prepared by dissolving a compound of Formula (I) in water or an organic solvent, such as a ketone, alcohol or glycol ether. These solutions may contain a surface active agent (for example to improve water dilution or prevent crystallisation in a spray tank).
Emulsifiable concentrates (EC) or oil-in-water emulsions (EW) may be prepared by dissolving a compound of Formula (I) in an organic solvent (optionally containing one or more wetting agents, one or more emulsifying agents or a mixture of said agents). Suitable organic solvents for use in ECs include aromatic hydrocarbons (such as alkylbenzenes or alkylnaphthalenes, exemplified by SOLVESSO 100, SOLVESSO 150 and SOLVESSO 200; SOLVESSO is a Registered Trade Mark), ketones (such as cyclohexanone or methylcyclohexanone) and alcohols (such as benzyl alcohol, furfuryl alcohol or butanol), N-alkylpyrrolidones (such as N-methylpyrrolidone or N-octylpyrrolidone), dimethyl amides of fatty acids (such as Cs-Cio fatty acid dimethylamide) and chlorinated hydrocarbons. An EC
product may spontaneously emulsify on addition to water, to produce an emulsion with sufficient stability to allow spray application through appropriate equipment.
Preparation of an EW involves obtaining a compound of Formula (I) either as a liquid (if it is not a liquid at room temperature, it may be melted at a reasonable temperature, typically below 70°C) or in solution (by dissolving it in an appropriate solvent) and then emulsifying the resultant liquid or solution into water containing one or more SFAs, under high shear, to produce an emulsion. Suitable solvents for use in EWs include vegetable oils, chlorinated hydrocarbons (such as chlorobenzenes), aromatic solvents (such as alkylbenzenes or alkylnaphthalenes) and other appropriate organic solvents which have a low solubility in water.
Microemulsions (ME) may be prepared by mixing water with a blend of one or more solvents with one or more SFAs, to produce spontaneously a thermodynamically stable isotropic liquid formulation. A compound of Formula (I) is present initially in either the water or the solvent/SFA blend. Suitable solvents for use in MEs include those hereinbefore described for use in in ECs or in EWs. An ME may be either an oil-in-water or a water-in-oil system (which system is present may be determined by conductivity measurements) and may be suitable for mixing water- soluble and oil-soluble pesticides in the same formulation. An ME is suitable for dilution into water, either remaining as a microemulsion or forming a conventional oil- in-water emulsion.
Suspension concentrates (SC) may comprise aqueous or non-aqueous suspensions of finely divided insoluble solid particles of a compound of Formula (I). SCs may be prepared by ball or bead milling the solid compound of Formula (I) in a suitable medium, optionally with one or more dispersing agents, to produce a fine particle suspension of the compound. One or more wetting agents may be included in the composition and a suspending agent may be included to reduce the rate at which the particles settle. Alternatively, a compound of Formula (I) may be dry milled and added to water, containing agents hereinbefore described, to produce the desired end product.
Aerosol formulations comprise a compound of Formula (I) and a suitable propellant (for example n-butane). A compound of Formula (I) may also be dissolved or dispersed in a suitable medium (for example water or a water miscible liquid, such as n-propanol) to provide compositions for use in non-pressurised, hand-actuated spray pumps.
Capsule suspensions (CS) may be prepared in a manner similar to the preparation of EW formulations but with an additional polymerisation stage such that an aqueous dispersion of oil droplets is obtained, in which each oil droplet is encapsulated by a polymeric shell and contains a compound of Formula (I) and, optionally, a carrier or diluent therefor. The polymeric shell may be produced by either an interfacial polycondensation reaction or by a coacervation procedure. The compositions may provide for controlled release of the compound of Formula (I) and they may be used for seed treatment. A compound of Formula (I) may also be formulated in a biodegradable polymeric matrix to provide a slow, controlled release of the compound.
The composition may include one or more additives to improve the biological performance of the composition, for example by improving wetting, retention or distribution on surfaces; resistance to rain on treated surfaces; or uptake or mobility of a compound of Formula (I). Such additives include surface active agents (SFAs), spray additives based on oils, for example certain mineral oils or natural plant oils (such as soy bean and rape seed oil), and blends of these with other bio-enhancing adjuvants (ingredients which may aid or modify the action of a compound of Formula
(I)·
Wetting agents, dispersing agents and emulsifying agents may be SFAs of the cationic, anionic, amphoteric or non-ionic type.
Suitable SFAs of the cationic type include quaternary ammonium compounds (for example cetyltrimethyl ammonium bromide), imidazolines and amine salts.
Suitable anionic SFAs include alkali metals salts of fatty acids, salts of aliphatic monoesters of sulphuric acid (for example sodium lauryl sulphate), salts of sulphonated aromatic compounds (for example sodium dodecylbenzenesulphonate, calcium dodecylbenzenesulphonate, butylnaphthalene sulphonate and mixtures of sodium di-/sopropyl- and tri-/sopropyl-naphthalene sulphonates), ether sulphates, alcohol ether sulphates (for example sodium laureth-3-sulphate), ether carboxylates (for example sodium laureth-3-carboxylate), phosphate esters (products from the reaction between one or more fatty alcohols and phosphoric acid (predominately mono-esters) or phosphorus pentoxide (predominately di-esters), for example the reaction between lauryl alcohol and tetraphosphoric acid; additionally these products may be ethoxylated), sulphosuccinamates, paraffin or olefine sulphonates, taurates and lignosulphonates.
Suitable SFAs of the amphoteric type include betaines, propionates and glycinates.
Suitable SFAs of the non-ionic type include condensation products of alkylene oxides, such as ethylene oxide, propylene oxide, butylene oxide or mixtures thereof, with fatty alcohols (such as oleyl alcohol or cetyl alcohol) or with alkylphenols (such as octylphenol, nonylphenol or octylcresol); partial esters derived from long chain fatty acids or hexitol anhydrides; condensation products of said partial esters with ethylene oxide; block polymers (comprising ethylene oxide and propylene oxide); alkanolamides; simple esters (for example fatty acid polyethylene glycol esters); amine oxides (for example lauryl dimethyl amine oxide); and lecithins.
Suitable suspending agents include hydrophilic colloids (such as polysaccharides, polyvinylpyrrolidone or sodium carboxymethylcellulose) and swelling clays (such as bentonite or attapulgite).
The herbicidal compounds of the present invention can also be used in mixture with one or more additional herbicides and/or plant growth regulators. Examples of such additional herbicides or plant growth regulators include acetochlor, acifluorfen (including acifluorfen-sodium), aclonifen, ametryn, amicarbazone, aminopyralid, aminotriazole, atrazine, beflubutamid-M, bensulfuron
(including bensulfuron-methyl), bentazone, bicyclopyrone, bilanafos, bispyribac- sodium, bixlozone, bromacil, bromoxynil, butachlor, butafenacil, carfentrazone (including carfentrazone-ethyl), cloransulam (including cloransulam- methyl), chlorimuron (including chlorimuron-ethyl), chlorotoluron, chlorsulfuron, cinmethylin, clacyfos, clethodim, clodinafop (including clodinafop- propargyl), clomazone, clopyralid, cyclopyranil, cyclopyrimorate, cyclosulfamuron, cyhalofop (including cyhalofop-butyl), 2,4-D (including the choline salt and 2- ethylhexyl ester thereof), 2,4-DB, desmedipham, dicamba (including the aluminium, aminopropyl, bis-aminopropylmethyl, choline, dichloroprop, diglycolamine, dimethylamine, dimethylammonium, potassium and sodium salts thereof) diclosulam, diflufenican, diflufenzopyr, dimethachlor, dimethenamid-P, diquat dibromide, diuron, epyrifenacil, ethalfluralin, ethofumesate, fenoxaprop (including fenoxaprop-P- ethyl), fenoxasulfone, fenquinotrione, fentrazamide, flazasulfuron, florasulam, florpyrauxifen (including florpyrauxifen-benzyl), fluazifop (including fluazifop-P- butyl), flucarbazone (including flucarbazone-sodium), flufenacet, flumetsulam, flumioxazin, fluometuron, flupyrsulfuron (including flupyrsulfuron-methyl-sodium), fluroxypyr (including fluroxypyr-meptyl), fomesafen, foramsulfuron, glufosinate (including the ammonium salt thereof), glyphosate (including the
diammonium, isopropylammonium and potassium salts thereof), halauxifen (including halauxifen-methyl), haloxyfop (including haloxyfop- methyl), hexazinone, hydantocidin, imazamox, imazapic, imazapyr, imazethapyr, indaziflam, iodosulfuron (including iodosulfuron-methyl- sodium), iofensulfuron (including iofensulfuron-sodium), ioxynil, isoproturon, isoxaflutole, lancotrione, MCPA, MCPB, mecoprop-P, mesosulfuron (including mesosulfuron-methyl), mesotrione, metamitron, metazachlor, methiozolin, metolachlor, metosulam, metribuzin, metsulfuron, napropamide, nicosulfuron, norflurazon, oxadiazon, oxasulfuron, oxyfluorfen, paraquat dichloride, pendimethalin, penoxsulam, phenmedipham, picloram, pinoxaden, pretilachlor, primisulfuron-methyl, prometryne, propanil, propaquizafop, propyrisulfuron, propyzamide, prosulfocarb, prosulfuron, pyraclonil, pyraflufen (including pyraflufen- ethyl), pyrasulfotole, pyridate, pyriftalid, pyrimisulfan, pyroxasulfone, pyroxsulam, quinclorac, quinmerac, quizalofop (including quizalofop-P-ethyl and quizalofop-P- tefuryl), rimsulfuron, saflufenacil, sethoxydim, simazine, S-metalochlor, sulfentrazone, sulfosulfuron, tebuthiuron, tefuryltrione, tembotrione, terbuthylazine, terbutryn, tetflupyrolimet, thiencarbazone, thifensulfuron, tiafenacil, tolpyralate, topramezone, tralkoxydim, triafamone, triallate, triasulfuron, tribenuron (including tribenuron-methyl), triclopyr,
trifloxysulfuron (including trifloxysulfuron-sodium), trifludimoxazin,
trifluralin, triflusulfuron, 3-(2-chloro-4-fluoro-5-(3-methyl-2,6-dioxo-4-trifluoromethyl- 3,6-dihydropyrimidin-1 (2H)-yl)phenyl)-5-methyl-4,5-dihydroisoxazole-5-carboxylic acid ethyl ester, 4-hydroxy-1-methoxy-5-methyl-3-[4-(trifluoromethyl)-2- pyridyl]imidazolidin-2-one, 4-hydroxy-1 ,5-dimethyl-3-[4-(trifluoromethyl)-2- pyridyl]imidazolidin-2-one, 5-ethoxy-4-hydroxy-1-methyl-3-[4-(trifluoromethyl)-2- pyridyl]imidazolidin-2-one, 4-hydroxy-1-methyl-3-[4-(trifluoromethyl)-2- pyridyl]imidazolidin-2-one, 4-hydroxy-1 ,5-dimethyl-3-[1-methyl-5-
(trifluoromethyl)pyrazol-3-yl]imidazolidin-2-one, (4R)1-(5-tert-butylisoxazol-3-yl)-4- ethoxy-5-hydroxy-3-methyl-imidazolidin-2-one, 3-[2-(3,4-dimethoxyphenyl)-6-methyl- 3-oxo-pyridazine-4-carbonyl]bicyclo[3.2.1]octane-2,4-dione, 2-[2-(3,4- dimethoxyphenyl)-6-methyl-3-oxo-pyridazine-4-carbonyl]-5-methyl-cyclohexane-1 ,3- dione, 2-[2-(3,4-dimethoxyphenyl)-6-methyl-3-oxo-pyridazine-4- carbonyl]cyclohexane-1 ,3-dione, 2-[2-(3,4-dimethoxyphenyl)-6-methyl-3-oxo- pyridazine-4-carbonyl]-5,5-dimethyl-cyclohexane-1 ,3-dione, 6-[2-(3,4- dimethoxyphenyl)-6-methyl-3-oxo-pyridazine-4-carbonyl]-2,2,4,4-tetramethyl- cyclohexane-1 ,3,5-trione, 2-[2-(3,4-dimethoxyphenyl)-6-methyl-3-oxo-pyridazine-4- carbonyl]-5-ethyl-cyclohexane-1 ,3-dione, 2-[2-(3,4-dimethoxyphenyl)-6-methyl-3-oxo-
pyridazine-4-carbonyl]-4,4,6,6-tetramethyl-cyclohexane-1 ,3-dione, 2-[6-cyclopropyl-2- (3,4-dimethoxyphenyl)-3-oxo-pyridazine-4-carbonyl]-5-methyl-cyclohexane-1 ,3-dione,
3-[6-cyclopropyl-2-(3,4-dimethoxyphenyl)-3-oxo-pyridazine-4- carbonyl]bicyclo[3.2.1]octane-2,4-dione, 2-[6-cyclopropyl-2-(3,4-dimethoxyphenyl)-3- oxo-pyridazine-4-carbonyl]-5,5-dimethyl-cyclohexane-1 ,3-dione, 6-[6-cyclopropyl-2- (3,4-dimethoxyphenyl)-3-oxo-pyridazine-4-carbonyl]-2,2,4,4-tetramethyl- cyclohexane-1 ,3,5-trione, 2-[6-cyclopropyl-2-(3,4-dimethoxyphenyl)-3-oxo-pyridazine-
4-carbonyl]cyclohexane-1 ,3-dione, 4-[2-(3,4-dimethoxyphenyl)-6-methyl-3-oxo- pyridazine-4-carbonyl]-2,2,6,6-tetramethyl-tetrahydropyran-3,5-dione, 4-[6- cyclopropyl-2-(3,4-dimethoxyphenyl)-3-oxo-pyridazine-4-carbonyl]-2, 2,6,6- tetramethyl-tetrahydropyran-3,5-dione, 4-amino-3-chloro-5-fluoro-6-(7-fluoro-1 H- indol-6-yl)pyridine-2-carboxylic acid (including agrochemically acceptable esters thereof, for example, methyl 4-amino-3-chloro-5-fluoro-6-(7-fluoro-1 H-indol-6- yl)pyridine-2-carboxylate, prop-2-ynyl 4-amino-3-chloro-5-fluoro-6-(7-fluoro-1 H-indol- 6-yl)pyridine-2-carboxylate and cyanomethyl 4-amino-3-chloro-5-fluoro-6-(7-fluoro- 1 H-indol-6-yl)pyridine-2-carboxylate), 3-ethylsulfanyl-N-(1 ,3,4-oxadiazol-2-yl)-5- (trifluoromethyl)-[1 ,2,4]triazolo[4,3-a]pyridine-8-carboxamide, 3-
(isopropylsulfanylmethyl)-N-(5-methyl-1 ,3,4-oxadiazol-2-yl)-5-(trifluoromethyl)- [1 ,2,4]triazolo[4,3-a]pyridine-8-carboxamide, 3-(isopropylsulfonylmethyl)-N-(5-methyl- 1 ,3,4-oxadiazol-2-yl)-5-(trifluoromethyl)-[1 ,2,4]triazolo[4,3-a]pyridine-8-carboxamide, 3-(ethylsulfonylmethyl)-N-(5-methyl-1 ,3,4-oxadiazol-2-yl)-5-(trifluoromethyl)-
[1 ,2,4]triazolo[4,3-a]pyridine-8-carboxamide, ethyl 2-[[3-[[3-chloro-5-fluoro-6-[3- methyl-2,6-dioxo-4-(trifluoromethyl)pyrimidin-1-yl]-2-pyridyl]oxy]acetate, 6-chloro-4- (2,7-dimethyl-1-naphthyl)-5-hydroxy-2-methyl-pyridazin-3-one, 1-[2-chloro-6-(5- chloropyrimidin-2-yl)oxy-phenyl]-4,4,4-trifluoro-butan-1-one and 5-[2-chloro-6-(5- chloropyrimidin-2-yl)oxy-phenyl]-3-(difluoromethyl)isoxazole.
The mixing partners of the compound of Formula (I) may also be in the form of esters or salts, as mentioned e.g. in The Pesticide Manual, Sixteenth Edition, British Crop Protection Council, 2012.
The compound of Formula (I) can also be used in mixtures with other agrochemicals such as fungicides, nematicides or insecticides, examples of which are given in The Pesticide Manual.
The mixing ratio of the compound of Formula (I) to the mixing partner is preferably from 1 : 100 to 1000:1.
The mixtures can advantageously be used in the above-mentioned formulations (in which case "active ingredient" relates to the respective mixture of compound of Formula (I) with the mixing partner).
The compounds or mixtures of the present invention can also be used in combination with one or more herbicide safeners. Examples of such safeners include benoxacor, cloquintocet (including cloquintocet-mexyl), cyprosulfamide, dichlormid, fenchlorazole (including fenchlorazole-ethyl), fenclorim, fluxofenim, furilazole, isoxadifen (including isoxadifen-ethyl), mefenpyr (including mefenpyr-diethyl), metcamifen and oxabetrinil.
Particularly preferred are mixtures of a compound of Formula (I) with cyprosulfamide, isoxadifen-ethyl, cloquintocet-mexyl and/or metcamifen.
The safeners of the compound of Formula (I) may also be in the form of esters or salts, as mentioned e.g. in The Pesticide Manual, 16th Edition (BCPC), 2012. The reference to cloquintocet-mexyl also applies to a lithium, sodium, potassium, calcium, magnesium, aluminium, iron, ammonium, quaternary ammonium, sulfonium or phosphonium salt thereof as disclosed in WO 02/34048.
Preferably the mixing ratio of compound of Formula (I) to safener is from 100:1 to 1 : 10, especially from 20: 1 to 1 : 1.
The mixtures can advantageously be used in the above-mentioned formulations (in which case“active ingredient” relates to the respective mixture of compound of Formula (I) with the safener).
The present invention still further provides a method of controlling weeds at a locus comprising crop plants and weeds, wherein the method comprises application to the locus of a weed controlling amount of a composition according to the present invention. ‘Controlling’ means killing, reducing or retarding growth or preventing or reducing germination. Generally the plants to be controlled are unwanted plants (weeds).‘Locus’ means the area in which the plants are growing or will grow.
The rates of application of compounds of Formula (I) may vary within wide limits and depend on the nature of the soil, the method of application (pre- or post emergence; seed dressing; application to the seed furrow; no tillage application etc.), the crop plant, the weed(s) to be controlled, the prevailing climatic conditions, and other factors governed by the method of application, the time of application and the target crop. The compounds of Formula (I) according to the invention are generally applied at a rate of from 10 to 2000 g/ha, especially from 50 to 1000 g/ha.
The application is generally made by spraying the composition, typically by tractor mounted sprayer for large areas, but other methods such as dusting (for powders), drip or drench can also be used.
Useful plants in which the composition according to the invention can be used include crops such as cereals, for example barley and wheat, cotton, oilseed rape, sunflower, maize, rice, soybeans, sugar beet, sugar cane and turf.
Crop plants can also include trees, such as fruit trees, palm trees, coconut trees or other nuts. Also included are vines such as grapes, fruit bushes, fruit plants and vegetables.
Crops are to be understood as also including those crops which have been rendered tolerant to herbicides or classes of herbicides (e.g. ALS-, GS-, EPSPS-, PPO-, ACCase- and HPPD-inhibitors) by conventional methods of breeding or by genetic engineering. An example of a crop that has been rendered tolerant to imidazolinones, e.g. imazamox, by conventional methods of breeding is Clearfield® summer rape (canola). Examples of crops that have been rendered tolerant to herbicides by genetic engineering methods include e.g. glyphosate- and glufosinate- resistant maize varieties commercially available under the trade names RoundupReady® and LibertyLink®.
Crops are also to be understood as being those which have been rendered resistant to harmful insects by genetic engineering methods, for example Bt maize (resistant to European corn borer), Bt cotton (resistant to cotton boll weevil) and also Bt potatoes (resistant to Colorado beetle). Examples of Bt maize are the Bt 176 maize hybrids of NK® (Syngenta Seeds). The Bt toxin is a protein that is formed naturally by Bacillus thuringiensis soil bacteria. Examples of toxins, or transgenic plants able to synthesise such toxins, are described in EP-A-451 878, EP-A-374 753, WO 93/07278, WO 95/34656, WO 03/052073 and EP-A-427 529. Examples of transgenic plants comprising one or more genes that code for an insecticidal resistance and express one or more toxins are KnockOut® (maize), Yield Gard® (maize), NuCOTIN33B® (cotton), Bollgard® (cotton), NewLeaf® (potatoes), NatureGard® and Protexcta®. Plant crops or seed material thereof can be both resistant to herbicides and, at the same time, resistant to insect feeding (“stacked” transgenic events). For example, seed can have the ability to express an insecticidal Cry3 protein while at the same time being tolerant to glyphosate.
Crops are also to be understood to include those which are obtained by conventional methods of breeding or genetic engineering and contain so-called output traits (e.g. improved storage stability, higher nutritional value and improved flavour).
Other useful plants include turf grass for example in golf-courses, lawns, parks and roadsides, or grown commercially for sod, and ornamental plants such as flowers or bushes.
The compositions can be used to control unwanted plants (collectively, ‘weeds’). The weeds to be controlled may be both monocotyledonous species, for example Agrostis, Alopecurus, Avena, Brachiaria, Bromus, Cenchrus, Cyperus, Digitaria, Echinochloa, Eleusine, Lolium, Monochoria, Rottboellia, Sagittaria, Scirpus, Setaria and Sorghum, and dicotyledonous species, for example Abutilon, Amaranthus, Ambrosia, Chenopodium, Chrysanthemum, Conyza, Galium, Ipomoea, Nasturtium, Sida, Sinapis, Solanum, Stellaria, Veronica, Viola and Xanthium. The compounds of the present invention have been shown to exhibit particularly good activity against certain grass weed species, especially Lolium Perenne. Weeds can also include plants which may be considered crop plants but which are growing outside a crop area (‘escapes’), or which grow from seed left over from a previous planting of a different crop (‘volunteers’). Such volunteers or escapes may be tolerant to certain other herbicides.
The compounds of the present invention can be prepared according to the following schemes.
Compounds of formula (I) wherein G is other than hydrogen may be prepared by treating a compound of formula (I) wherein G is hydrogen, with a reagent G-Z, wherein G-Z is an alkylating agent such as an alkyl halide, acylating agent such as an acid chloride or anhydride, sulfonylating agent such as a sulfonyl chloride, carbamylating agent such as a carbamoyl chloride, or carbonating agent such as a chloroformate, using known methods.
Scheme 1
Compounds of formula (I) may be prepared by reacting an iodonium ylide of formula (A), wherein Ar is an optionally substituted phenyl group, and an aryl boronic acid of formula (B), in the presence of a suitable palladium catalyst, a base and in a suitable solvent.
Scheme 2
Suitable palladium catalysts are generally palladium(ll) or palladium(O) complexes, for example palladium(ll) dihalides, palladium(ll) acetate, palladium(ll) sulfate, bis(triphenylphosphine)-palladium(ll) dichloride, bis(tricyclopentylphosphine)- palladium(ll) dichloride, bis(tricyclohexyl-phosphine)palladium(ll) dichloride, bis(dibenzylideneacetone)palladium(0) or tetrakis-(triphenylphosphine)palladium(0). The palladium catalyst can also be prepared "in situ" from palladium(ll) or palladium(O) compounds by complexing with the desired ligands, by, for example, combining the palladium(ll) salt to be complexed, for example palladium(ll) dichloride (PdCh) or palladium(ll) acetate (Pd(OAc)2), together with the desired ligand, for example triphenylphosphine (PPhi3), tricyclopentylphosphine, tricyclohexylphosphine, 2-dicyclohexylphosphino-2’,6’-dimethoxybiphenyl or 2-dicyclohexylphosphino-2’,4’,6’- triisopropylbiphenyl and the selected solvent, with a compound of formula (N), the arylboronic acid of formula (O), and a base. Also suitable are bidendate ligands, for example 1 ,T-bis(diphenylphosphino)ferrocene or 1 ,2-bis(diphenylphosphino)ethane. By heating the reaction medium, the palladium(ll) complex or palladium(O) complex desired for the C-C coupling reaction is thus formed "in situ", and then initiates the C- C coupling reaction.
The palladium catalysts are used in an amount of from 0.001 to 50 mol %, preferably in an amount of from 0.1 to 15 mol %, based on the compound of formula (N). The reaction may also be carried out in the presence of other additives, such as tetralkylammonium salts, for example, tetrabutylammonium bromide. Preferably the palladium catalyst is palladium acetate, the base is lithium hydroxide and the solvent is aqueous 1 ,2-dimethoxyethane.
A compound of formula (A) may be prepared from a 1 ,3 dione compound of formula (C) by treatment with a hypervalent iodine reagent such as a (diacetoxy)iodobenzene or an iodosylbenzene and a base such as aqueous sodium carbonate, lithium hydroxide or sodium hydroxide in a solvent such as water or an aqueous alcohol such as aqueous ethanol using known procedures.
Scheme 3
Alternatively, the R1 group may be added later in the synthetic sequence by decarboxylative propynylation (where R1 is propyne, or alternative Suzuki/Stille or similar cross coupling where R1 is phenyl or heteroaryl) such as in step 2 below.
Scheme 4
Boronic acids can be prepared by methods such as below in Scheme 5. For example, a compound of formula (B) or (D) may be prepared from an aryl halide of formula (F) or (H) by known methods. For example, an aryl halide of formula (F) or (H) may be treated with an alkyl lithium or alkyl magnesium halide in a suitable solvent, preferably diethyl ether or tetrahydrofuran, at a temperature of between -80 °C and 30 °C, and the aryl magnesium or aryl lithium reagent obtained may then be reacted with a trialkyl borate (preferably tri methyl borate) to give an aryl dialkylboronate which may be hydrolysed to provide a boronic acid of formula (B) or (D) under acidic conditions.
Scheme 5
Compounds of formula (I) can also be prepared via Pb coupling as shown in the scheme below by reacting a compound of formula (D), to form an organolead reagent of formula (J) and subsequent reaction with 1 ,3 dione (C) under conditions described, for example, by J. Pinhey, Pure and Appl. Chem., (1996), 68 (4), 819 and by M. Moloney et al., Tetrahedron Lett., (2002), 43, 3407. A suitable triarylbismuth compound under conditions described, for example, by A. Yu. Fedorov et al., Russ. Chem. Bull. Int. Ed., (2005), 54 (11), 2602, and by P. Koech and M. Krische, J. Am. Chem. Soc., (2004), 126 (17), 5350 and references therein may be used as a related procedure.
Scheme 6
The compounds of type (I) can also be prepared via palladium coupling as shown in the scheme below, where boronic acid of type (B) is coupled to the suitably protected halo-alkene of type (K) in a Suzuki type coupling.
Scheme 7
With suitable conditions, a suitable 1 ,3 dione may also be directly coupled to a Halo- compound (for example of formula (L)) with palladium catalysis. Propynylation or arylation/Heteroarylation of intermediate (M) as described earlier gives compounds of type (I).
Scheme 8
A compound of formula (I, G= H) may be prepared by the cyclisation of a compound of formula (N), wherein R is hydrogen or an alkyl group, preferably in the presence of an acid or base, and optionally in the presence of a suitable solvent, by analogous methods to those described by T. Wheeler, US 4,209,532. The compounds of formula (N) have been particularly designed as intermediates in the synthesis of the compounds of the Formula (I). A compound of formula (N) wherein R is hydrogen may be cyclised under acidic conditions, preferably in the presence of a strong acid such as sulfuric acid, polyphosphoric acid or Eaton’s reagent, optionally in the presence of a suitable solvent such as acetic acid, toluene or dichloromethane.
Scheme 9
The following non-limiting examples provide specific synthesis methods for representative compounds of the present invention, as referred to in the Tables below.
Example 1 : 4-(2,6-dimethyl-4-prop-1-ynyl-phenyl)-N,N-dimethyl-3,5-dioxo- cyclohexanecarboxamide (Compound 1.001)
Step 1 : Synthesis of tert-butyl 9-(4-bromo-2,6-dimethyl-phenyl)-8,10-dioxo-3- azaspiro[5.5]undecane-3-carboxylate
To a stirred solution of compound ethyl 3,5-dioxocyclohexanecarboxylate (0.558 g, 3.03 mmol) in Chloroform (25 ml_) under nitrogen was added 4- Dimethylaminopyridine (1.61 g, 13.2 mmol). The reaction mixture was stirred for 15 min at RT, then anhydrous toluene (7.5 ml_) was added followed by the addition of [diacetoxy-(4-bromo-2,6-dimethyl-phenyl)plumbyl] acetate (1.50 g, 2.64 mmol). The reaction mixture was stirred at 80°C for 3h and then allowed stand at RT overnight. The reaction mixture was then acidified with 10% citric acid solution. The white ppt was filtered through a celite bed and washed with fresh chloroform. The organic layers were separated and aqueous layer extracted with Chloroform (x2). The combined organics were washed with brine solution, dried over Na2SC>4, filtered and evaporated to obtain crude material, which was purified by flash chromatography to give product as a white foam, (730 mg). 1HNMR (MeOD, 400MHz): d 7.17 (s, 2H), 4.17 (q, 2H), 3.29-3.22 (m, 1 H), 2.92-2.79 (m, 4H), 2.01-1.95 (6H), 1.27 (t, 3H).
Step 2: Synthesis of ethyl 4-(2,6-dimethyl-4-prop-1-ynyl-phenyl)-3,5-dioxo- cyclohexanecarboxylate
Ethyl 4-(4-bromo-2,6-dimethyl-phenyl)-3,5-dioxo-cyclohexanecarboxylate (730 mg, 1.99 mmol), 4-diphenylphosphanylbutyl(diphenyl)phosphane (0.169 g, 0.397 mmol), Bis(triphenylphosphine)palladium(ll) chloride (0.140 g, 0.199 mmol) and 2-Butynoic acid (0.501 g, 5.96 mmol) were added to a round bottomed flask and a vacuum/argon back fill cycle was repeated twice, then added anhydrous Dimethyl Sulfoxide (12 ml_) was added and again a vacuum/argon back fill cycle was repeated for three times. Then 1 ,8-Diazabicyclo[5.4.0]undec-7-ene (1.82 g, 11.9 mmol) was added to. The reaction mixture was stirred at 120°C overnight. The reaction mixture was acidified with 10% citric acid solution and extracted with EtOAc (x3). The combined organics were washed with brine solution, dried over Na2SC>4, filtered and evaporated to obtain crude material. The crude was purified by flash chromatography to give product as an off white solid, (375 mg). 1 HNMR (MeOD, 400MHz): 7.01-7.00 (2H), 4.17 (q, 2H), 3.26-3.23 (m, 1 H), 2.92-2.79 (m, 4H), 1.99-1.93 (9H), 1.27 (t, 3H).
Ethyl 4-(2,6-dimethyl-4-prop-1-ynyl-phenyl)-3,5-dioxo-cyclohexanecarboxylate (5.93 g, 18.2 mmol) was taken up into THF (86 ml_) and a solution of UOH.H2O (3.81 g, 90.8 mmol) in water (173.0 ml_) was added. The reaction mixture was stirred at room temperature for 2h and was then concentrated and then diluted with water, washed with EtOAc (x2). The aqueous layer was acidified with 2N HCI and extracted with EtOAc (x4). The combined organics were washed with brine solution and dried over Na2SC>4, filtered and concentrated to obtain crude material, which was triturated with pentane to give product. 1H NMR (MeOD, 400MHz): d 7.01 (s, 2H), 3.22-3.18 (1 H), 2.91-2.78 (4H), 1.99-1.95(9H).
Step 4: 4-(2,6-dimethyl-4-prop-1-ynyl-phenyl)-N,N-dimethyl-3,5-dioxo- cyclohexanecarboxamide (Compound 1.001)
4-(2,6-dimethyl-4-prop-1-ynyl-phenyl)-3,5-dioxo-cyclohexanecarboxylic acid (340 g, 1.14 mmol) was taken up in dichloromethane (30.0 ml_) and triethylamine (0.477 ml_, 3.42 mmol) was added followed by dimethylamine (2M , 1.71 ml_, 3.42 mmol) and T3P (50.0 %, 0.873 ml_, 1.48 mmol) at RT. The reaction mixture was stirred at RT for 2 h, then quenched with 2N HCI. The organic layer was separated and the aqueous layer extracted with DCM (x2). The combined organics were dried over Na2SC>4, filtered and concentrated to obtain crude, which was purified by flash chromatography to give product obtain as a white solid, (120 mg).
1H NMR (400MHz, Methanol-d4) d = 7.05 - 6.94 (m, 2H), 3.72 - 3.60 (m, 1 H), 3.20 - 3.13 (m, 3H), 3.01 - 2.93 (m, 3H), 2.85 - 2.74 (m, 2H), 2.65 - 2.52 (m, 2H), 2.07 - 2.04 (m, 3H), 2.03 - 1.95 (m, 6H)
Example 2: 4-(2,6-dimethyl-4-prop-1-ynyl-phenyl)-N,N-dimethyl-3,5-dioxo- cyclohexanecarboxamide (Compound 3.008)
Step 1 : Te/f-butyl 4-[(E)-3-oxobut-1-enyl]piperidine-1-carboxylate
Te/f-butyl 4-formylpiperidine-1-carboxylate (25.0 g, 117 mmol and 1- (triphenylphosphoranylidene)-2-propanone (37.3 g, 117 mmol) were dissolved in DCM (300ml_) and refluxed overnight. The reaction mixture was cooled and evaporated. The crude was purified by flash chromatography to give product as a yellow liquid (26.8 g). 1 H NMR (D-6 DMSO) d 6.83-6.77 (m, 1 H), 6.02-5.98 (m, 1 H), 3.95-3.92 (m, 2H), 2.76 (ill.res.m, 2H), 2.35-2.33 (m, 1 H), 2.18 (s, 3H), 1.69-1.66 (m, 2H), 1.36 (s, 9H), 1.26-1.17 (m, 2H)
Step 2: Tert- butyl 4-(2-ethoxycarbonyl-3,5-dioxo-cyclohexyl)piperidine-1-carboxylate
To a stirred solution of tert- butyl 4-[(E)-3-oxobut-1-enyl]piperidine-1-carboxylate (28.6 g, 113 mmol) in dry ethanol (120 ml_) under nitrogen was added diethyl propanedioate (17.2 ml_, 113 mmol) at RT followed by drop wise addition of sodium ethoxide (21.0 %, 47.6 g, 261 ml_, 147 mmol). The reaction mixture was stirred at RT for 3 h and refluxed for 2 h and then kept at RT overnight. The ethanol was evaporated off under reduced pressure, and then the resulting mixture was diluted with water, then acidified with aqueous citric acid and extracted with ethyl acetate. The combined organics were washed with water, brine and dried over Na2SC>4, filtered and then concentrated to give crude product (36 g) as a gum. This material was used in the next step without further purification
Te/f-butyl 4-(2-ethoxycarbonyl-3,5-dioxo-cyclohexyl)piperidine-1-carboxylate (36.0 g, 98.0 mmol) was taken in ethanol (88 ml_). Sodium hydroxide (12.0 M, 173 ml_, 2078 mmol) was added at RT and the reaction mixture was stirred over the weekend. The reaction mixture was concentrated and cooled in an ice bath then acidified with 10% citric acid solution. The mixture was saturated with solid NaCI then extracted with EtOAc (1 L x 3). The combined organics were washed with brine and dried over Na2SC>4. It was then filtered, concentrated and the crude was triturated with hexane- ether mixture to obtain product a light yellow solid (26 .5 g). 1 H NMR (400 MHz, MeOD) d = 4.12-4.09 (2H), 2.71 (brs. 2H), 2.40(dd, 2H), 2.22-2.15(m, 2H), 1.98- 1.91 (m, 1 H), 1.75-1 72(2H), 1.47-1.41(10H), 1.16-1.09 (m, 2H).
Step 4: Tert- butyl 4-[4-(4-bromo-2,6-dimethyl-phenyl)-3,5-dioxo- cyclohexyl]piperidine-1-carboxylate
To a stirred solution of tert- butyl 4-(3,5-dioxocyclohexyl)piperidine-1-carboxylate (3.35 g, 11.3 mmol) in chloroform (150 ml_) under nitrogen was added 4- dimethylaminopyridine (9.67 g, 79.2 mmol). The reaction mixture was stirred for 15 minutes at RT, then anhydrous toluene (45.0 ml_) was added followed by the addition of [diacetoxy-(4-bromo-2,6-dimethyl-phenyl)plumbyl] acetate (9.00 g, 15.8 mmol). The reaction mixture was stirred at 80°C for 3h and then allowed stand at RT overnight. The reaction mixture was acidified with 10% citric acid and the mixuture was filtered through a celite-bed. The celite-bed was washed with chloroform. The aqueous layer was separated and extracted with chloroform (x2). The combined organics were washed with brine, dried over Na2SC>4, filtered and evaporated to give a crude material , which was purified by flash chromatography to give product as a light brown foam. 1 HNMR (MeOD, 400MHz): 7.18 (s, 2H), 4.15-4.12 (m, 2H), 2.73 (m,
2H), 2.61-2.58 (m, 2H), 2.41 (m, 2H), 2.02-2.0 (6H), 1.81-1.78 (m, 2H), 1.60 (1 H), 1.45 (s, 9H), 1.35-1.09 (m, 3H).
Step 5: Te/f-butyl 4-[4-(2,6-dimethyl-4-prop-1-ynyl-phenyl)-3,5-dioxo- cyclohexyl]piperidine-1-carboxylate
Te/f-butyl 4-[4-(4-bromo-2,6-dimethyl-phenyl)-3,5-dioxo-cyclohexyl]piperidine-1- carboxylate (500 mg, 1.05 mmol), 4-diphenylphosphanylbutyl(diphenyl)phosphane (0.0891 g, 0.209 mmol), Bis(triphenylphosphine)palladium(ll) chloride (0.0734 g, 0.105 mmol, and 2-Butynoic acid (0.264 g, 3.14 mmol) were taken up in a round bottomed flask. A vacuum/argon back fill cycle was repeated twice, then anhydrous dimethyl sulfoxide (5.3 ml_) was added and again a vacuum/argon back fill cycle was repeated three times then 1 ,8-diazabicyclo[5.4.0]undec-7-ene (0.955 g, 6.27 mmol) was added to the reaction mixture. This was stirred at 120°C for 40h and then allowed stand at RT. The reaction mixture was diluted with water (10 ml) and extracted with ethyl acetate (20 ml x 1). The aqueous layer was then acidified with 10% citric acid to pH4, then saturated with NaCI and extracted with ethyl acetate (25 ml_ x 3). The combined organic layers were washed with brine and dried over Na2SC>4. The organics were then filtered, concentrated under reduced pressure to obtained crude which was purified by flash chromatography to give product (300 mg). 1 HNMR (DMSO-D6, 400MHz): d 10.48 (bs, 1 H), 6.99 (s, 2H), 4.03-4.00 (m, 3H), 2.66 (2H), 2.32 (1 H), 2.16 (1 H), 2.01-1.98 (4H), 1.93-1.91 (6H), 1.68 (2H), 1.39 (11 H), 1.06 (2H).
Step 6: 2-(2,6-dimethyl-4-prop-1-ynyl-phenyl)-5-(4-piperidyl)cyclohexane-1 ,3-dione hydrochloride
Tert- butyl 4-[4-(2,6-dimethyl-4-prop-1-ynyl-phenyl)-3,5-dioxo-cyclohexyl]piperidine-1- carboxylate (0.300 g, 0.686 mmol) was stirred for 1 hour at rt in HCI (4M) in Dioxane
(5.68 ml_, 22.7 mmol). The reaction mixture was evaporated to dryness to leave a light brown solid (212 mg). This crude solid was used for the next step without further purification.
Step 7 : [5-(1 -acetyl-4-piperidyl)-2-(2,6-dimethyl-4-prop-1 -ynyl-phenyl)-3-oxo- cyclohexen-1-yl] acetate
2-(2,6-dimethyl-4-prop-1-ynyl-phenyl)-3-hydroxy-5-(4-piperidyl)cyclohex-2-en-1-one (0.212 g, 0.628 mmol) was taken up in DCM (4ml_), cooled to 0°C and acetyl chloride (0.247 g, 3.14 mmol) was added followed by triethylamine (0.438 ml_, 3.14 mmol) upon which all solids went immediately into solution. After stirring at room temperature for 2 hours, the reaction mixture was then diluted with water and extracted with DCM (2 x 50 ml_). The organics were washed with brine and dried over Na2SC>4. They were then filtered, concentrated and the crude was was purified by flash chromatography to give product (0.153 g) as a white solid. 1 H NMR (400 MHz, CDC ) d = 7.05(s, 2H), 4.70(d, 1 H), 3.87(d, 1 H), 3.03(t, 1 H), 2.65-2.63(3H), 2.49(t, 1 H), 2.36(t, 1 H), 2.09-1 97(14H), 1.84(5H), 1.24(m, 2H).
Step 8: 5-(1-acetyl-4-piperidyl)-2-(2,6-dimethyl-4-prop-1-ynyl-phenyl)cyclohexane- 1 ,3-dione (Compound 3.008)
[5-(1 -acetyl-4-piperidyl)-2-(2,6-dimethyl-4-prop-1 -ynyl-phenyl)-3-oxo-cyclohexen-1 -yl] acetate (0.153 g, 0.363 mmol) was dissolved in methanol (3ml_) and to this was added K2CO3 (0.100 g, 0.726 mmol). The reaction mixture was stirred at room temperature for 2h and allowed stand overnight at RT. The methanol was evaporated off and the crude was acidified with 10% citric acid solution to pH3. It was then extracted with ethyl acetate (3 x 10 ml_). The combined organics were washed with
brine and dried over Na2SC>4. This was filtered and concentrated and the solid thus obtained was triturated with pentane and ether to get a white solid which was freeze- dried in lyophilizer for 2 days to give the desired product. 1 H NMR (400MHz, CDC ) d = 1.13 - 1.38 (m, 2 H) 1.48 - 1.58 (m, 2 H) 1.83 (br d, J=14.43 Hz, 2 H) 2.03 (d, J=3.18 Hz, 6 H) 2.06 (s, 3 H) 2.09 (s, 3 H) 2.25 (dd, J=15.89, 12.72 Hz, 1 H) 2.37 -
2.57 (m, 2 H) 2.57 - 2.74 (m, 2 H) 2.97 - 3.12 (m, 1 H) 3.82 - 3.96 (m, 1 H) 4.70 (dt, J= 13.33, 1.83 Hz, 1 H) 5.72 - 5.95 (m, 1 H) 7.15 (s, 2 H).
Examples of herbicidal compounds of the present invention.
Table 1
Biological Examples
Seeds of a variety of test species are sown in standard soil in pots ( Lolium perenne (LOLPE), Setaria faberi (SETFA), Alopecurus myosuroides (ALOMY), Echinochloa crus-galli (ECHCG), Avena fatua (AVEFA)). After cultivation for one day (pre emergence) or after 8 days cultivation (post-emergence) under controlled conditions in a glasshouse (at 24/16°C, day/night; 14 hours light; 65 % humidity), the plants are sprayed with an aqueous spray solution derived from the formulation of the technical active ingredient in acetone / water (50:50) solution containing 0.5% Tween 20 (polyoxyethelyene sorbitan monolaurate, CAS RN 9005-64-5). Compounds are applied at 250 g/h. The test plants are then grown in a glasshouse under controlled conditions in a glasshouse (at 24/16°C, day/night; 14 hours light; 65 % humidity) and watered twice daily. After 13 days for pre and post-emergence, the test is evaluated
for the percentage damage caused to the plant. The biological activities are shown in the following table on a five-point scale (5 = 80-100%; 4 = 60-79%; 3=40-59%; 2=20-39%; 1=0-19%). TABLE B1 Application Pre-Emergence
TABLE B2 Application Post-Emergence
NT= Not Tested
Claims
1 . A compound of Formula (I)
wherein R1 is selected from methyl, ethynyl, 1-propynyl, phenyl and a 5 or 6 membered heteroaryl which comprises one or two nitrogen heteroatoms, said phenyl and heteroaryl optionally substituted by one or two R15 substituents;
R2 is selected from the group consisting of methyl, ethyl, methoxy and chloro;
R3 is selected from the group consisting of methyl, ethyl, methoxy and chloro; k is 0, 1 or 2; R4x is selected from the group consisting of hydrogen, C1-C6 alkyl, Ci-
Cehaloalkyl, CrCealkoxyCrCsalkyl- and Ci-CehaloalkoxyCrCsalkyl-;
R4y is selected from the group consisting of R4a, R4b, R4C, R4d and R4e;
R4aa is selected from the group consisting of hydrogen, C1-C6 alkyl and Ci- Cealkoxy-;
R4ab is selected from the group consisting of hydrogen, CrCealkyl, C2- Cealkenyl, C2-C6alkynyl, CrCehaloalkyl, hydroxy-, hydroxyCi-Cealkyl-, Ci- Cealkoxy, C3-C6cycloalkyl, -Ci-C4alkoxyCi-Cealkyl, -Ci-CsalkoxyCrCehaloalkyl, cyanoCrCealkyl-, C(0)R27, S(0)nR27, phenyl, -pyridyl and pyrimidinyl, wherein the phenyl, pyridyl or pyrimidinyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, C2-C3 alkenyl, C2-C3 alkynyl, halogen, cyano and nitro; or
R4aa and R4ab together form— (CH2)q-, -OCH2CH2CH2CH2-, -CH2OCH2C(CH3)2-, -CH2CH2X1CH2CH2- or -C(0)CH2X1CH2CH2- wherein X1 is selected from the group consisting of O, S(0)n and N-R28; and
R4ba is selected from the group consisting of hydrogen, C1-C4 alkyl and Ci- C4alkoxy-;
R4bb is selected from the group consisting of CrC4alkyl, Ci-C4alkoxy-, Ci- C4haloalkyl, -C(0)CrC4alkyl, -C(0)CrC4haloalkyl, hydroxyCi-C6alkyl-C(0)-, - S(0)nCi-Cealkyl, -S(0)nCi-C6haloalkyl, -S(0)n-(CH2)n-C3-C6cycloalkyl, S(0)nC(R11)R12R13, -C(0)H, -C(0)-(CH2)n-C3-C6cycloalkyl, -C(0)C(R11)R12R13, -C(0)C2-C4alkenyl, -C(O)(CR9R10)CN, -C(O)(CR9R10)(CR9R10)CN,
C(0)CH2C(0)-Ci-C6alkyl, -C(0)CH20C(0)-Ci-C6alkyl, -C(0)OCi-C6alkyl, - C(0)OCi-C6haloalkyl, -C(0)(CH2)nS(0)nC1-C6alkyl -C(0)Ci-C3alkoxyCi-
Cealkyl, -C(0)Ci-C3alkoxyC2-C6alkenyl, -C(0)Ci-C3alkoxyC2-C6alkynyl, - C(0)Ci-C3alkoxyCi-C6haloalkyl, -C(0)Ci-C3alkoxyC3-C6cycloalkyl, -C(0)OCi-
C(0)C(R23)=N-NR25R26, -(CH2)n-phenyl, -C(0)-(CH2)n-phenyl, -S(0)n-(CH2)n- phenyl, -heterocyclyl, -C(0)-(CH2)n-heterocyclyl, -C(0)(CH2)n0-(CH2)n- heterocyclyl, -S(0)n-(CH2)n-heterocyclyl, wherein each heterocyclyl is a 5- or 6- membered heterocyclyl which may be aromatic, saturated or partially saturated and can contain from 1 to 4 heteroatoms each independently selected from the group consisting of oxygen, nitrogen and sulphur, and wherein said heterocyclyl or phenyl groups are optionally substituted by one, two or three substituents independently selected from the group consisting of CrC3alkyl, CrCshaloalkyl, CrCsalkoxy, C2-C3alkenyl, C2-C3alkynyl, halogen, cyano and nitro; p is 0, 1 or 2; r is 0, 1 or 2; wherein p+r = 0, 1 or 2;
R5 is independently selected from the group consisting of hydrogen and C1-C6 alkyl;
R6 is selected from the group consisting of hydrogen, CrCealkyl, C2-C6alkenyl, C2-C6alkynyl, CrCehaloalkyl, hydroxyl-, CrCealkoxy, C3-C6 cycloalkyl, , -Ci- C4alkoxyCrC6alkyl, -Ci-CsalkoxyCrCehaloalkyl, -(CR9R10)Ci-C6haloalkyl, - (CR9R10)C(O)NR5R5, phenyl, -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, C2-C3 alkenyl, C2-C3 alkynyl, halogen, cyano and nitro; or
R5 and R6 together form -CH2CH2OCH2CH2-; and
R7 is selected from the group consisting of hydrogen and C1-C6 alkyl;
R8 is selected from the group consisting of hydrogen, C1-C6 alkyl, C1-C6 alkoxy, C3-C6 cycloalkyl, phenyl, -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, C2-C3 alkenyl, C2-C3 alkynyl, halogen, cyano and nitro;
R9 is hydrogen or methyl;
R1 0 is hydrogen or methyl; or
R9 and R1 0 together form -CH2CH2-; and
R1 1 is hydrogen or methyl;
R1 2 is selected from the group consisting of hydrogen, C1-C6 alkyl, hydroxyl and C1-C6 alkoxy-;
R1 3 is selected from the group consisting of hydrogen, C1-C6 alkyl, hydroxyl and C1-C6 alkoxy; or
R1 2 and R1 3 together form -CH2-X2-CH2- wherein X2 is selected from the group consisting of O, S and N-R14; and
R1 4 is selected from the group consisting of hydrogen, C1-C3 alkyl and C1-C3 alkoxy-;
R1 5 is independently selected from the group consisting of C1-C4 alkyl, C1-C4 haloalkyl, cyano and halogen;
R16 is hydrogen or CrCealkyl; and
R17 is selected from the group consisting of hydrogen, CrCealkyl, C3- Cecycloalkyl, CrCe alkoxy-CrC3alkyl-,-C(0)CrCealkyl, -C(0)OCrCealkyl and CH2CN; or
R1 6 and R1 7 together form -CH2CH2OCH2CH2-, -CH2CH2S(0)2CH2CH2-; and
R18 is hydrogen or CrCealkyl;
R1 9 is selected from the group consisting of hydrogen, C1-C6 alkyl, C1-C6 alkoxy, C3-C6cycloalkyl, phenyl, -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, C2-C3 alkenyl, C2-C3 alkynyl, halogen, cyano and nitro;
R20 is selected from the group consisting of CrCealkyl, CrCehaloalkyl, Cr Cealkoxy-, CrCehaloalkoxy, -NR21R22, phenyl and -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, C2-C3 alkenyl, C2-C3 alkynyl, halogen, cyano and nitro;
R21 is selected from the group consisting of hydrogen, C1-C6 alkyl, C1-C6 alkoxy, CrCealkoxyCrCsalkyl-, C3-C6 cycloalkyl, CrCehaloalkyl- and Cr Cehaloalkoxy-, -C(0)CrCealkyl, phenyl, -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, C2-C3 alkenyl, C2-C3 alkynyl, halogen, cyano and nitro; R22 is hydrogen or CrCealkyl; or
R21 and R22 together form -CH2CH2OCH2CH2-; and
R23 is selected from the group consisting of hydrogen, CrCealkyl, Cr Cehaloalkyl, CrCealkoxy- and CrCehaloalkoxy-;
R24 is selected from the group consisting of hydrogen, CrCealkyl, Cr C6alkoxyCrC3alkyl-, C3-Cecycloalkyl, -CH2CN, tetrahydropyranyl-, phenyl and -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of Cr Cealkyl, CrCehaloalkyl, CrCealkoxy, C2-C3alkenyl, C2-C3alkynyl, halogen, cyano and nitro;
R25 is hydrogen or CrCe alkyl;
R26 is hydrogen or C1-C6 alkyl; and
R4ca is selected from hydrogen, CrCealkyl, C2-C6alkenyl, C2-C6alkynyl, Ci- Cehaloalkyl, hydroxyl-, CrCealkoxy, C3-C6cycloalkyl, -Ci-C4alkoxyCrC6alkyl, - Ci-C3alkoxyCrC6haloalkyl, cyanoC1-C6alkyl-. C(0)R27, S(0)nR27, phenyl, - pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of Ci- C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, C2-C3 alkenyl, C2-C3 alkynyl, halogen, cyano and nitro;
R4cb is selected from the group consisting of hydrogen, CrCe alkyl and Ci- Cealkoxy-; or
R4ca and R4cb together form — (CH2)q-, -CH2CH2X1CH2CH2- or - C(0)CH2X1CH2CH2- wherein X1 is selected from the group consisting of O, S(0)n and N-R28;
R27 is selected from the group consisting of hydrogen, CrCe alkyl, CrCe haloalkyl, C2-Cealkenyl, C2-Cealkynyl, CrCe alkoxy, C3-C6 cycloalkyl, phenyl, -pyridyl, wherein the phenyl and pyridyl are optionally substituted by one, two or three substituents independently selected from the group consisting of Ci- C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, C2-C3 alkenyl, C2-C3 alkynyl, halogen, cyano and nitro;
R28 is selected from the group consisting of hydrogen, C1-C3 alkyl, -C(0)Ci- C3alkyl and C1-C3 alkoxy-;
R4ea is R4ea1 or R4ea2
p4ea1 p4ea2 n is independently 0, 1 or 2; q is 3, 4 or 5; and
G is selected from the group consisting of hydrogen, -(Chb R3, -C(0)-Ra, - C(0)-(CRcRd)n-0-Rb, -C(0)-(CRcRd)n-S-Rb, -C(0)NRaRa, -S(0)2-Ra and Ci- Csalkoxy-Ci-Csalkyl-;
Ra is independently selected from the group consisting of hydrogen, Ci- Csalkyl, CrCshaloalkyl, C2-Csalkenyl, C2-Csalkynyl, C3-C6cycloalkyl, heterocyclyl and phenyl wherein said heterocyclyl and phenyl groups are optionally substituted by one, two or three substituents independently selected from the group consisting of CrCsalkyl, CrCshaloalkyl, CrCsalkoxy, C2-Csalkenyl, C2-Csalkynyl, halogen, cyano and nitro;
Rb is selected from the group consisting of CrCsalkyl, CrCshaloalkyl, C2- Csalkenyl, C2-Csalkynyl, C3-C6 cycloalkyl, heterocyclyl and phenyl wherein said heterocyclyl and phenyl groups are optionally substituted by one, two or three substituents independently selected from the group consisting of Cr Csalkyl, CrCshaloalkyl, CrCsalkoxy, C2-C3alkenyl, C2-C3alkynyl, halogen, cyano and nitro;
Rc is hydrogen or C1-C3 alkyl; and
Rd is hydrogen or C1-C3 alkyl; or an agriculturally acceptable salt thereof.
2. A compound according to claim 1 , wherein R1 is 1-propynyl.
3. A compound according to claim 1 , wherein R1 is a 5 or 6 membered heteroaryl which comprises one or two nitrogen heteroatoms, said phenyl and heteroaryl optionally substituted by one or two R15 substituents.
4. A compound according to any one of the previous claims, wherein R2 is methyl.
5. A compound according to claim any one of the previous claims, wherein R3 is methyl.
6. A compound according to any one of claims 1 to 4, wherein R4 is selected from the group consisting of R4a, R4b R4d and R4e
7. A compound according to any one of claims 1 to 6, wherein R4 is R4a.
8. A compound according to any one of the previous claims, wherein G is hydrogen.
9. A compound according to any one of claims 1 to 7, wherein G is -C(0)Ci- Cealkyl.
10. A compound according to any one of claims 1 to 7, wherein G is -C(0)-0-Ci- Cealkyl.
11. A herbicidal composition comprising a compound of Formula (I) according to any one of the previous claims and an agriculturally acceptable formulation adjuvant.
12. A herbicidal composition according to claim 11 , further comprising at least one additional pesticide.
13. A herbicidal composition according to claim 12, wherein the additional pesticide is a herbicide or herbicide safener.
14. A method of controlling weeds at a locus comprising application to the locus of a weed controlling amount of a composition according to any one of claims 11 to 13.
15. Use of a compound of Formula (I) as defined in claim 1 as a herbicide.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB1910930.5 | 2019-07-31 | ||
| GBGB1910930.5A GB201910930D0 (en) | 2019-07-31 | 2019-07-31 | Improvements in or relating to organic compounds |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2021018833A1 true WO2021018833A1 (en) | 2021-02-04 |
Family
ID=67990355
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2020/071130 Ceased WO2021018833A1 (en) | 2019-07-31 | 2020-07-27 | Cyclohexanediones as herbicidal compounds |
Country Status (4)
| Country | Link |
|---|---|
| AR (1) | AR119498A1 (en) |
| GB (1) | GB201910930D0 (en) |
| UY (1) | UY38811A (en) |
| WO (1) | WO2021018833A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023181030A1 (en) * | 2022-03-21 | 2023-09-28 | Ag Plenus Ltd | Pesticidal and herbicidal compounds and methods of use thereof |
| WO2025040301A1 (en) * | 2023-08-21 | 2025-02-27 | Bayer Aktiengesellschaft | Herbicide/safener combinations based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy] acetic acids and herbicides from the class of substituted cyclic diones and the salts thereof |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4209532A (en) | 1977-03-28 | 1980-06-24 | Union Carbide Corporation | Biocidal 2-aryl-1,3-cyclohexane dione compounds and alkali metal and ammonium salts thereof |
| US4659372A (en) * | 1977-03-28 | 1987-04-21 | Union Carbide Corporation | Biocidal 2-aryl-1,3-cyclohexanedione enol ester compounds |
| EP0374753A2 (en) | 1988-12-19 | 1990-06-27 | American Cyanamid Company | Insecticidal toxines, genes coding therefor, antibodies binding them, transgenic plant cells and plants expressing these toxines |
| EP0427529A1 (en) | 1989-11-07 | 1991-05-15 | Pioneer Hi-Bred International, Inc. | Larvicidal lectins and plant insect resistance based thereon |
| EP0451878A1 (en) | 1985-01-18 | 1991-10-16 | Plant Genetic Systems, N.V. | Modifying plants by genetic engineering to combat or control insects |
| WO1993007278A1 (en) | 1991-10-04 | 1993-04-15 | Ciba-Geigy Ag | Synthetic dna sequence having enhanced insecticidal activity in maize |
| WO1995034656A1 (en) | 1994-06-10 | 1995-12-21 | Ciba-Geigy Ag | Novel bacillus thuringiensis genes coding toxins active against lepidopteran pests |
| WO2002034048A1 (en) | 2000-10-23 | 2002-05-02 | Syngenta Participations Ag | Agrochemical compositions with quinoline safeners |
| WO2003052073A2 (en) | 2001-12-17 | 2003-06-26 | Syngenta Participations Ag | Novel corn event |
| WO2008110308A2 (en) | 2007-03-09 | 2008-09-18 | Syngenta Participations Ag | Novel herbicides |
| WO2012165648A1 (en) * | 2011-05-30 | 2012-12-06 | Sumitomo Chemical Company, Limited | Cyclohexanone compounds and herbicides comprising the same |
| EP2923571A1 (en) * | 2012-11-08 | 2015-09-30 | Sumitomo Chemical Company Limited | Herbicide composition |
-
2019
- 2019-07-31 GB GBGB1910930.5A patent/GB201910930D0/en not_active Ceased
-
2020
- 2020-07-27 AR ARP200102100A patent/AR119498A1/en unknown
- 2020-07-27 WO PCT/EP2020/071130 patent/WO2021018833A1/en not_active Ceased
- 2020-07-29 UY UY0001038811A patent/UY38811A/en not_active Application Discontinuation
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4209532A (en) | 1977-03-28 | 1980-06-24 | Union Carbide Corporation | Biocidal 2-aryl-1,3-cyclohexane dione compounds and alkali metal and ammonium salts thereof |
| US4659372A (en) * | 1977-03-28 | 1987-04-21 | Union Carbide Corporation | Biocidal 2-aryl-1,3-cyclohexanedione enol ester compounds |
| EP0451878A1 (en) | 1985-01-18 | 1991-10-16 | Plant Genetic Systems, N.V. | Modifying plants by genetic engineering to combat or control insects |
| EP0374753A2 (en) | 1988-12-19 | 1990-06-27 | American Cyanamid Company | Insecticidal toxines, genes coding therefor, antibodies binding them, transgenic plant cells and plants expressing these toxines |
| EP0427529A1 (en) | 1989-11-07 | 1991-05-15 | Pioneer Hi-Bred International, Inc. | Larvicidal lectins and plant insect resistance based thereon |
| WO1993007278A1 (en) | 1991-10-04 | 1993-04-15 | Ciba-Geigy Ag | Synthetic dna sequence having enhanced insecticidal activity in maize |
| WO1995034656A1 (en) | 1994-06-10 | 1995-12-21 | Ciba-Geigy Ag | Novel bacillus thuringiensis genes coding toxins active against lepidopteran pests |
| WO2002034048A1 (en) | 2000-10-23 | 2002-05-02 | Syngenta Participations Ag | Agrochemical compositions with quinoline safeners |
| WO2003052073A2 (en) | 2001-12-17 | 2003-06-26 | Syngenta Participations Ag | Novel corn event |
| WO2008110308A2 (en) | 2007-03-09 | 2008-09-18 | Syngenta Participations Ag | Novel herbicides |
| WO2012165648A1 (en) * | 2011-05-30 | 2012-12-06 | Sumitomo Chemical Company, Limited | Cyclohexanone compounds and herbicides comprising the same |
| EP2923571A1 (en) * | 2012-11-08 | 2015-09-30 | Sumitomo Chemical Company Limited | Herbicide composition |
Non-Patent Citations (6)
| Title |
|---|
| "Manual on Development and Use of FAO Specifications for Plant Protection Products", 1999 |
| "The Pesticide Manual", 2012, BRITISH CROP PROTECTION COUNCIL |
| A. YU. FEDOROV ET AL., RUSS. CHEM. BULL. INT. ED., vol. 54, no. 11, 2005, pages 2602 |
| J. PINHEY, PURE AND APPL. CHEM., vol. 68, no. 4, 1996, pages 819 |
| M. MOLONEY ET AL., TETRAHEDRON LETT., vol. 43, 2002, pages 3407 |
| P. KOECHM. KRISCHE, J. AM. CHEM. SOC., vol. 126, no. 17, 2004, pages 5350 |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023181030A1 (en) * | 2022-03-21 | 2023-09-28 | Ag Plenus Ltd | Pesticidal and herbicidal compounds and methods of use thereof |
| WO2025040301A1 (en) * | 2023-08-21 | 2025-02-27 | Bayer Aktiengesellschaft | Herbicide/safener combinations based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy] acetic acids and herbicides from the class of substituted cyclic diones and the salts thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| AR119498A1 (en) | 2021-12-22 |
| GB201910930D0 (en) | 2019-09-11 |
| UY38811A (en) | 2021-02-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12281081B2 (en) | Herbicidal compounds | |
| AU2020223480B2 (en) | Substituted azaspiro-cycles as herbicides | |
| EP3927691B1 (en) | Herbicidal compounds | |
| EP3864006A1 (en) | Herbicidal 2-azaspiro[3-5]nonane compounds | |
| WO2021009229A1 (en) | Herbicidal compounds | |
| EP3997072B1 (en) | Herbicidal cyclohexanedione derivatives | |
| EP3999492B1 (en) | Herbicidal compounds | |
| WO2021018833A1 (en) | Cyclohexanediones as herbicidal compounds | |
| EP4003956B1 (en) | Cyclic diones as herbicidal compounds | |
| WO2021018826A1 (en) | Cyclic diones as herbicides | |
| RU2830286C2 (en) | Herbicidal cyclohexanedione derivatives |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 20749829 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 20749829 Country of ref document: EP Kind code of ref document: A1 |