WO2021016682A1 - Anhydrous cosmetic composition with high concentration of hydrophobic fillers and use thereof for reducing skin lesions and oiliness - Google Patents
Anhydrous cosmetic composition with high concentration of hydrophobic fillers and use thereof for reducing skin lesions and oiliness Download PDFInfo
- Publication number
- WO2021016682A1 WO2021016682A1 PCT/BR2019/050309 BR2019050309W WO2021016682A1 WO 2021016682 A1 WO2021016682 A1 WO 2021016682A1 BR 2019050309 W BR2019050309 W BR 2019050309W WO 2021016682 A1 WO2021016682 A1 WO 2021016682A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- weight
- compounds
- cosmetic composition
- composition
- anhydrous cosmetic
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/008—Preparations for oily skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/19—Cosmetics or similar toiletry preparations characterised by the composition containing inorganic ingredients
- A61K8/25—Silicon; Compounds thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/81—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions involving only carbon-to-carbon unsaturated bonds
- A61K8/8105—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/81—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions involving only carbon-to-carbon unsaturated bonds
- A61K8/8141—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides or nitriles thereof; Compositions of derivatives of such polymers
- A61K8/8152—Homopolymers or copolymers of esters, e.g. (meth)acrylic acid esters; Compositions of derivatives of such polymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/92—Oils, fats or waxes; Derivatives thereof, e.g. hydrogenation products thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q17/00—Barrier preparations; Preparations brought into direct contact with the skin for affording protection against external influences, e.g. sunlight, X-rays or other harmful rays, corrosive materials, bacteria or insect stings
- A61Q17/04—Topical preparations for affording protection against sunlight or other radiation; Topical sun tanning preparations
Definitions
- the present disclosure is directed to high SPF anhydrous cosmetic compositions. More specifically, the present disclosure is directed to high SPF anhydrous cosmetic compositions having enhanced anti-oiliness and anti-acne effect, as well as good hardness properties, water-resistance, and dry touch to the skin after application combined with matt effect, comprising high concentration of silica aerogel, ozokerite wax, at least one polymer and an UV filter system.
- the present invention also discloses a process of manufacturing the anhydrous cosmetic compositions and uses of the cosmetic compositions.
- UVA/UVB sunscreen compositions used to accomplish photoprotection, namely, inorganic UV filters and organic UV filters.
- the degree of UV protection afforded by a sunscreen composition is directly related to the amount and type of UV filters contained therein. The higher the amount of UV filters, the greater the degree of UV protection (UVA/UVB). Particularly, sunscreen compositions must provide good protection against the sun, a measure of which is the Sun Protection Factor (SPF) value, yet have satisfactory sensory perception, such as a smooth but not greasy feel upon application.
- SPF Sun Protection Factor
- sunscreen products may be in the form of lotions, milks, creams, gels, gel creams, foams, sprays and sticks.
- Such products can be anhydrous or in the form of emulsions, generally containing sunscreen actives that are solubilized, emulsified, or dispersed in a vehicle, which is topically applied onto the skin.
- the sunscreen actives typically through the aid of polymers and other ingredients included in the vehicle, form a thin, protective, and often water-resistant layer on the skin.
- Hydrophobic silica is commonly known in the cosmetic industry for providing anti-oiliness and anti-acne effects, including in sunscreen compositions.
- the maximum concentration of silica in a composition is limited, due to its high hydrophobic properties, thereby rendering the composition instable when high concentrations of silica are applied.
- sunscreen compositions are usually limited to have about 0.5% by weight of silica, relative to the total weight of the composition.
- sunscreen compositions having higher concentrations of silica, in order to enhance the anti-oiliness and anti-acne effects thereof.
- the challenge of incorporating high concentrations of silica in the sunscreen composition is not only limited due to the hydrophobic nature of the silica, but there is also the challenge on formulating stable compositions while preserving satisfactory properties of the product, such as hardness, high SPF and dry touch to the skin after application combined with matt effect.
- anhydrous cosmetic composition that enables the high concentration of silica aerogel by combining ozokerite wax, at least one polymer and a UV filter system.
- the anhydrous cosmetic composition of the present invention having high concentration of silica showed enhanced anti-oiliness and anti-acne effects, good hardness properties, water-resistance, high SPF values, dry touch to the skin after application combined with matt effect and no white film formation.
- the present disclosure relates to anhydrous cosmetic compositions having enhanced anti-oiliness and anti-acne effects, good hardness properties, high SPF values, water-resistance and dry touch to the skin after application combined with matt effect, comprising high concentration of silica aerogel, ozokerite wax, at least one polymer and UV filter system.
- the present disclosure is also directed to a process of manufacturing the anhydrous cosmetic compositions and uses of the anhydrous cosmetic compositions.
- FIG. 1. shows the evolution of the mean count of papules across time of volunteers under daily use of an anhydrous cosmetic composition according to the present invention.
- FIG. 2. shows the evolution of the mean count of pustules across time of volunteers under daily use of an anhydrous cosmetic composition according to the present invention.
- FIG. 3. shows the evolution of the mean count of total inflammatory lesions across time of volunteers under daily use of an anhydrous cosmetic composition according to the present invention.
- FIG. 4. shows the evolution of the mean count of comedones across time of volunteers under daily use of an anhydrous cosmetic composition according to the present invention.
- FIG. 5. shows the evolution of the mean count of microcysts across time of volunteers under daily use of an anhydrous cosmetic composition according to the present invention.
- FIG. 6. shows the evolution of the mean count of total non-inflammatory lesions across time of volunteers under daily use of an anhydrous cosmetic composition according to the present invention.
- anhydrous cosmetic compositions of the present invention can comprise, consist of, or consist essentially of the essential elements and limitations of the present invention described herein, as well as any of the additional or optional ingredients, components, or limitations described herein.
- the expression“at least one” means one or more and thus includes individual components as well as mixtures/combinations.
- anhydrous means that the cosmetic composition of the present invention, and the essential or optional components thereof, are substantially free of added or free water. From a formulation standpoint, this means that the cosmetic composition of the present invention contains less than about 5%, preferably less than about 3%, more preferably less than about 2% by weight, even more preferably less than about 1 % by weight of free or added water.
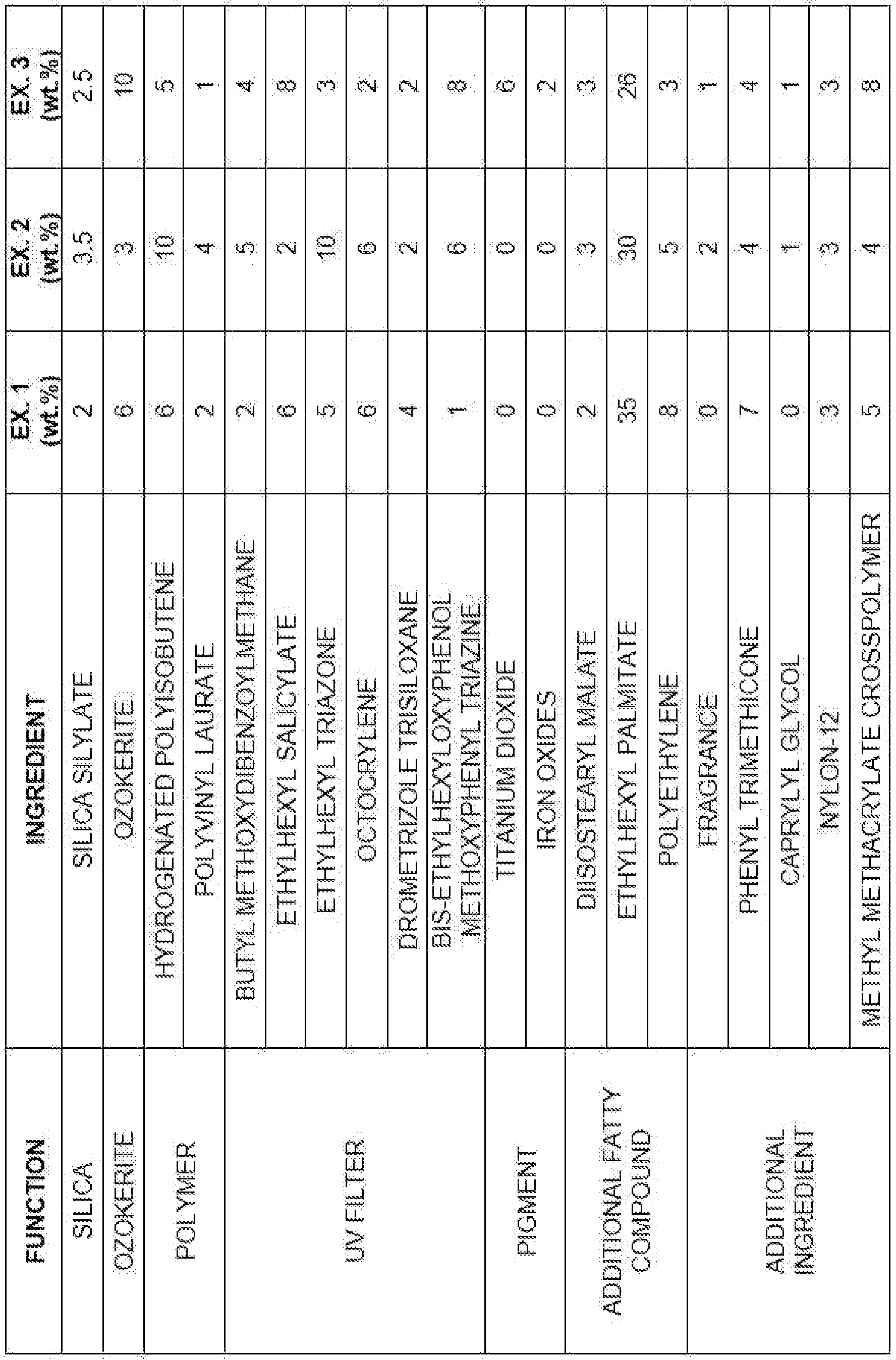
- the anhydrous cosmetic composition of the present invention comprises:
- composition according to the invention presents enhanced anti oiliness and anti-acne properties, high SPF values, water-resistance, good hardness, dry touch to the skin after application combined with matt effect and no white film formation.
- the amount of silica aerogel in the anhydrous cosmetic composition of the present invention is from about 2% by weight to about 10% by weight, more preferably from about 2% by weight to about 8% by weight, relative to the total weight of the composition.
- the silica aerogel is preferably hydrophobic silica aerogel, more preferably silica silylate aerogel.
- the amount of ozokerite wax in the anhydrous cosmetic composition of the present invention is preferably at least about 3% by weight, relative to the total weight of the composition, more preferably from about 3% by weight to about 15% by weight, even more preferably from about 3% by weight to about 10% by weight, most preferably from about 3% by weight to about 8% by weight, relative to the total weight of the composition.
- the amount of the at least one polymer in the anhydrous cosmetic composition of the present invention is preferably at least about 5% by weight, relative to the total weight of the composition, more preferably from about 5% by weight to about 25% by weight, even more preferably from about 5% by weight to about 20% by weight, most preferably from about 5% by weight to about 15% by weight, relative to the total weight of the composition.
- the anhydrous cosmetic composition of the present invention has an amount of UV filter system ranging from about 0.1 % by weight to about 50% by weight, preferably from about 1 % by weight to about 40% by weight, more preferably from about 1 % by weight to about 30% by weight, even more preferably from about 1 % by weight to about 25% by weight, in relation to the total weight of the composition.
- the UV filter system of the present invention may comprise at least one UV filter selected from the group of inorganic UV filters and organic UV filters, and mixtures thereof.
- the UV filter system comprise butyl methoxydibenzoylmethane, ethylhexyl salicylate, ethylhexyl triazone, octocrylene, drometrizole trisiloxane, bis-ethylhexyloxyphenol methoxyphenyl triazine, or mixtures thereof.
- the anhydrous cosmetic composition of the present invention comprises additional fatty compounds selected from the group of oils, waxes, fatty acids, fatty alcohols, and mixtures thereof.
- Said additional fatty compounds are preferably in an amount of at least about 20% by weight, relative to the total weight of the composition, preferably ranging from 20% by weight to 70% by weight, relative to the total weight of composition, more preferably from about 25% by weight to about 65% by weight, even more preferably from about 30% by weight to about 60% by weight, relative to the total weight of the composition.
- the additional fatty compounds are selected from the group consisting of synthetic esters, waxes, and mixtures thereof, preferably being selected from the group consisting of ethylhexyl palmitate, diisostearyl malate, polyethylene, or mixtures thereof.
- the anhydrous cosmetic composition of the present invention comprises pigments in an amount ranging from about 0.1 % to about 30% by weight, more preferably from about 1 % to about 25% by weight, even more preferably from about 1 % by weight to about 20% by weight, relative to the total weight of the composition.
- composition of the present invention presents a SPF of at least 30, preferably 30, 50, 60, 70, 80, 90 or 100.
- the anhydrous cosmetic composition of the invention can be used as a product for the skin and may be in the form of lotions, milks, creams, gels, gel creams, foams, sprays and sticks.
- the anhydrous cosmetic composition of the present invention is in the form of a stick.
- the present invention is related to the use of a composition for manufacturing a product for preventing sunburn, which can be used as cosmetic daily product, preferably in the form of a stick.
- the anhydrous cosmetic composition of the present invention can be used for the manufacture of a cosmetic product for treating inflammatory and/or non inflammatory lesions of the skin, preferably human skin, more preferably human face skin.
- the cosmetic product is preferably in the form of a stick.
- the inflammatory and/or non-inflammatory lesions to be treated by the cosmetic product according to the present invention include, but are not limited to papules, pustules, comedones and microcysts.
- the anhydrous cosmetic composition of the present invention can also be used for the manufacture of a cosmetic product for reducing the oiliness of the skin, preferably human skin, more preferably human face skin.
- the composition of the present invention is in the form of a stick having a hardness of 120g to 200g, measured by means of a Stable Micro SystemsTM Needle probe, following a penetrometry protocol.
- the present invention is also related to a process of manufacturing an anhydrous cosmetic composition that provides for the consumer the properties described above.
- the process according to the present invention comprises the steps of:
- step (b) optionally, incorporating the additional ingredients and the pigments to the mixture of step (a) at a temperature ranging from 60 to 100 °C;
- steps (a), (b) and (c) are carried out at a temperature ranging from about 70°C to about 90°C, more preferably from about 80°C to 90°C.
- the at least one additional fatty compound is melted and mixed in step (a).
- the “silica aerogel” according to the present invention is a porous material obtained by replacing (by drying) the liquid component of a silica gel with air.
- Silica aerogels are generally synthesized via a sol-gel process in a liquid medium and then dried, usually by extraction with a supercritical fluid, such as, but not limited to, supercritical carbon dioxide (CO2). This type of drying makes it possible to avoid shrinkage of the pores and of the material.
- a supercritical fluid such as, but not limited to, supercritical carbon dioxide (CO2).
- the amount of silica aerogel in the anhydrous cosmetic composition of the present invention is at least about 2% by weight, relative to the total weight of the composition, preferably from about 2% by weight to about 10% by weight, more preferably from about 2% by weight to about 8% by weight, relative to the total weight of the composition.
- the hydrophobic silica aerogel particles used in the present invention have a specific surface area per unit of mass (SM) ranging from about 500 to about 1500 m 2 /g, or alternatively from about 600 to about 1200 m 2 /g, or alternatively from about 600 to about 800 m 2 /g, and a size expressed as the mean volume diameter (D[0.5]), ranging from about 1 to about 30 pm, or alternatively from about 5 to about 25 pm, or alternatively from about 5 to about 20 pm, or alternatively from about 5 to about 15 pm.
- the specific surface area per unit of mass may be determined via the BET (Brunauer-Emmett-Teller) nitrogen absorption method described in the Journal of the American Chemical Society, vol. 60, page 309, February 1938, corresponding to the international standard ISO 5794/1.
- the BET specific surface area corresponds to the total specific surface area of the particles under consideration.
- the size of the silica aerogel particles may be measured by static light scattering using a commercial granulometer such as the MasterSizer 2000 machine from Malvern.
- the data are processed on the basis of the Mie scattering theory.
- This theory which is exact for isotropic particles, makes it possible to determine, in the case of non-spherical particles, an “effective” particle diameter. This theory is especially described in the publication by Van de Hulst, H.C.,“Light Scattering by Small Particles,” Chapters 9 and 10, Wiley, New York, 1957.
- the silica aerogel particles used in the present invention may advantageously have a tamped (or tapped) density ranging from about 0.04 g/cm 3 to about 0.10 g/cm 3 ’ or alternatively from about 0.05 g/cm 3 to about 0.08 g/cm 3 .
- this density may be assessed according to the following protocol: 40 g of powder are poured into a measuring cylinder; the measuring cylinder is then placed on a Stav 2003 machine from Stampf Volumeter; the measuring cylinder is then subjected to a series of 2500 packing motions (this operation is repeated until the difference in volume between two consecutive tests is less than 2%); the final volume Vf of packed powder is then measured directly on the measuring cylinder.
- the tamped density is determined by the ratio m/Vf, in this instance 40/Vf (Vf being expressed in cm 3 and m in g).
- the hydrophobic silica aerogel particles used in the present invention have a specific surface area per unit of volume Sv ranging from about 5 to about 60 m 2 /cm 3 , or alternatively from about 10 to about 50 m 2 /cm 3 , or alternatively from about 15 to about 40 m 2 /cm 3 .
- the silica aerogel particles have an oil-absorbing capacity, measured at the wet point, ranging from about 5 to about 18 ml/g, or alternatively from about 6 to about 15 ml/g, or alternatively from about 8 to about 12 ml/g.
- the oil-absorbing capacity measured at the wet point corresponds to the amount of water that needs to be added to 100 g of particle in order to obtain a homogeneous paste. Wp is measured according to the wet point method or the method for determining the oil uptake of a powder described in standard NF T 30-022.
- Wp corresponds to the amount of oil adsorbed onto the available surface of the powder and/or absorbed by the powder by measuring the wet point, described below:
- An amount 2 g of powder is placed on a glass plate, and the oil (isononyl isononanoate) is then added dropwise.
- mixing is performed using a spatula, and addition of oil is continued until a conglomerate of oil and powder has formed.
- the oil is added one drop at a time and the mixture is then triturated with the spatula.
- the addition of oil is stopped when a firm, smooth paste is obtained. This paste must be able to be spread on the glass plate without cracking or forming lumps.
- the volume Vs (expressed in ml) of oil used is then noted.
- the oil uptake corresponds to the ratio Vs/m.
- the aerogels used, according to the present invention are hydrophobic silica aerogels, preferably of silylated silica (INCI name: silica silylate).
- hydrophobic silica means any silica whose surface is treated with silylating agents, for example, halogenated silanes, such as alkylchlorosilanes, siloxanes, in particular dimethylsiloxanes, such as hexamethyldisiloxane, or silazanes, so as to functionalize the OH groups with silyl groups Si-Rn, for example, trimethylsilyl groups.
- silylating agents for example, halogenated silanes, such as alkylchlorosilanes, siloxanes, in particular dimethylsiloxanes, such as hexamethyldisiloxane, or silazanes, so as to functionalize the OH groups with silyl groups Si-Rn, for example, trimethylsilyl groups.
- silylating agents for example, halogenated silanes, such as alkylchlorosilanes, siloxanes, in particular dimethylsiloxanes, such as
- Suitable examples of hydrophobic silica aerogels may include, but are not limited to, the aerogels sold under the tradenames of VM-2260 (INCI name: Silica silylate) and VM-2270 (INCI name: Silica silylate), both available from Dow Corning Corporation (Midland, Michigan).
- the particles of VM-2260 have a mean size of about 1000 microns and a specific surface area per unit of mass ranging from 600 to 800 m 2 /g.
- the particles of VM-2270 have a mean size ranging from 5 to 15 microns and a specific surface area per unit of mass ranging from 600 to 800 m 2 /g.
- hydrophobic silica aerogel may include, but is not limited to, the aerogels commercially available from Cabot Corporation (Billerica, Massachusetts) under the tradename of Aerogel TLD 201 , Aerogel OGD 201 and Aerogel TLD 203, Enova Aerogel MT 1 100 and Enova Aerogel MT 1200.
- the silica aerogel is preferably hydrophobic silica aerogel, more preferably silica silylate.
- the ozokerite wax according to the present invention is a wax with high meting point. More particularly, the ozokerite is a fossil hydrocarbon having a complex composition corresponding to the solid residue from the evaporation of paraffin-rich petroleum.
- a commercial ozokerite is, for example, the product CEROZO BLANCHE E 626®, which is a mixture of C20-50 hydrocarbons which is marketed by the company BARLOCHER.
- Another commercial ozokerite is Ozokerite Wax SP 1020P, available from Strahl U Pitsch.
- the high melting point of the ozokerite wax is between about 70° C. and about 80° C.
- the composition according to the present invention may comprise ozokerite wax in an amount of preferably at least about 3% by weight, relative to the total weight of the composition, more preferably from about 3% by weight to about 15% by weight, even more preferably from about 3% by weight to about 10% by weight, most preferably from about 3% by weight to about 8% by weight, relative to the total weight of the composition.
- the composition according to the present invention comprises a UV filter system.
- the UV filter system may comprise at least one UV filter selected from the group of inorganic UV filters, organic UV filters and mixtures thereof.
- composition may comprise the UV filter system in an amount of from about 0.1 % by weight to about 50% by weight, preferably from about 1% by weight to about 40% by weight, more preferably from about 1 % by weight to about 30% by weight, even more preferably from about 1 % by weight to about 25% by weight, in relation to the total weight of the composition.
- composition according to the present invention, comprise a UV filter system comprising at least one inorganic UV filter. If two or more inorganic UV filters are used, they may be the same or different.
- the inorganic UV filter used for the present invention may be active in the UV-A and/or UV-B region.
- the inorganic UV filter may be hydrophilic and/or lipophilic.
- the inorganic UV filter is in some embodiments insoluble in solvents, such as water, and ethanol commonly used in cosmetics.
- the inorganic UV filter be in the form of a fine particle such that the mean (primary) particle diameter thereof ranges from about 1 nm to about 50 nm, and in some embodiments from about 5 nm to about 40 nm, and in some embodiments from about 10 nm to about 30 nm.
- the mean (primary) particle size or mean (primary) particle diameter here is an arithmetic mean diameter.
- the inorganic UV filter can be selected from the group consisting of silicon carbide, metal oxides which may or may not be coated, and mixtures thereof.
- the inorganic UV filters are selected from pigments (mean size of the primary particles: generally from about 5 nm to about 50 nm, and in some embodiments from about 10 nm to about 50 nm) formed of metal oxides, such as, for example, pigments formed of titanium oxide (amorphous or crystalline in the rutile and/or anatase form), iron oxide, zinc oxide, zirconium oxide, or cerium oxide, which are all UV photoprotective agents that are well known per se.
- the inorganic UV filters are selected from titanium oxide, zinc oxide, and, in some embodiments, titanium oxide.
- the inorganic UV filter may or may not be coated.
- the inorganic UV filter may have at least one coating.
- the coating may comprise at least one compound selected from the group consisting of alumina, silica, aluminum hydroxide, silicones, silanes, fatty acids or salts thereof (such as sodium, potassium, zinc, iron, or aluminum salts), fatty alcohols, lecithin, amino acids, polysaccharides, proteins, alkanolamines, waxes, such as beeswax, (meth)acrylic polymers, organic UV filters, and (per)fluoro compounds. It is in some embodiments desirable for the coating to include at least one organic UV filter.
- a dibenzoylmethane derivative such as butyl methoxydibenzoylmethane (Avobenzone) and 2,2'-Methylenebis[6-(2H-Benzotriazol-2-yl)-4-(l,l,3,3-Tetramethyl-Butyl) Phenol] (Methylene Bis-Benzotriazolyl Tetramethylbutylphenol), such as marketed as “TINOSORB M” by BASF, may be desirable.
- the silicones in the coating(s) may be organosilicon polymers or oligomers comprising a linear or cyclic and branched or cross-linked structure, of variable molecular weight, obtained by polymerization and/or polycondensation of suitable functional silanes and essentially composed of repeated main units in which the silicon atoms are connected to one another via oxygen atoms (siloxane bond), optionally substituted hydrocarbon radicals being connected directly to said silicon atoms via a carbon atom.
- silanes also encompasses silanes necessary for their preparation, in particular alkylsilanes.
- the silicones used for the coating(s) can be and in some embodiments are selected from the group consisting of alkylsilanes, polydialkylsiloxanes, and polyalkylhydrosiloxanes. And in some embodiments still, the silicones are selected from the group consisting of octyltrimethylsilane, polydimethylsiloxanes, and polymethylhydrosiloxanes.
- the inorganic UV filters made of metal oxides may, before their treatment with silicones, have been treated with other surfacing agents, in particular with cerium oxide, alumina, silica, aluminum compounds, silicon compounds, or their mixtures.
- the coated inorganic UV filter may have been prepared by subjecting the inorganic UV filter to one or more surface treatments of a chemical, electronic, mechano-chemical, and/or mechanical nature with any of the compounds as described above, as well as polyethylene waxes, metal alkoxides (titanium or aluminum alkoxides), metal oxides, sodium hexametaphosphate, and those shown, for example, in Cosmetics & Toiletries, February 1990, Vol. 105, pp. 53-64.
- the coated inorganic UV filters may be titanium oxides coated: with silica, such as the product“Sun veil” from Ikeda, and“Sunsil TIN 50” from Sunjin Chemical; with silica and with iron oxide, such as the product“Sunveil F” from Ikeda; with silica and with alumina, such as the products“Microtitanium Dioxide MT 500 SA” from Tayca,“Tioveil” from Tioxide, and“Mirasun TiW 60” from Rhodia; with alumina, such as the products“Tipaque TTO-55 (B)” and“Tipaque TTO-55 (A)” from Ishihara, and“UVT 14/4” from Kemira; with alumina and with aluminum stearate, such as the product“Microtitanium Dioxide MT 100 T, MT 100 TX, MT 100 Z or MT-01” from Tayca, the products“Solaveil CT-10 W”
- Tipaque TTO-55 (S) from Ishihara or “UV Titan M 262” from Kemira; with triethanolamine, such as the product“STT-65-S” from Titan Kogyo; with stearic acid, such as the product “Tipaque TTO-55 (C)” from Ishihara; or with sodium hexametaphosphate, such as the product“Microtitanium Dioxide MT 150 W” from
- TiO2 treated with octyltrimethylsilane and for which the mean size of the individual particles is from 25 and 40 nm such as that marketed under the trademark“T 805” by Degussa Silices
- T1O2 treated with a polydimethylsiloxane and for which the mean size of the individual particles is 21 nm such as that marketed under the trademark“70250 Cardre UF TiC Sb” by Cardre
- the following coated T1O2 can be used as the coated inorganic UV filter: Stearic acid (and) Aluminum Hydroxide (and) T1O2, such as the product“MT-100 TV” from Tayca, with a mean primary particle diameter of 15 nm; Dimethicone (and) Stearic Acid (and) Aluminum Hydroxide (and) T1O2, such as the product“S A-TTO-S4” from Miyoshi Kasei, with a mean primary particle diameter of 15 nm; Silica (and) T1O2, such as the product“MT-100 WP” from Tayca, with a mean primary particle diameter of 15 nm; Dimethicone (and) Silica (and) Aluminum Hydroxide (and) T1O2, such as the product“MT-Y02” and“MT-Y-1 10 M3S” from Tayca, with a mean primary particle diameter of 10 nm; Dimethicone (and) Aluminum Hydroxide (and) T1O2, such as the product“MT-
- T1O2 coated with at least one organic UV filter is more desirable.
- Avobenzone (and) Stearic Acid (and) Aluminum Hydroxide (and) T1O2, such as the product“HXMT-100ZA” from Tayca, with a mean primary particle diameter of 15 nm, can be used.
- the uncoated titanium oxide pigments are, for example, marketed by Tayca under the trademarks “Microtitanium Dioxide MT500B” or “Microtitanium Dioxide MT600B”, by Degussa under the trademark“P 25”, by Wacker under the trademark“Oxyde de titane transparent PW”, by Miyoshi Kasei under the trademark “UFTR”, by Tomen under the trademark“ITS” and by Tioxide under the trademark “Tioveil AQ”.
- the uncoated zinc oxide pigments are, for example, those marketed under the trademark“Z-cote” by Sunsmart; those marketed under the trademark “Nanox” by Elementis; and those marketed under the trademark“Nanogard WCD 2025” by Nanophase Technologies.
- the coated zinc oxide pigments are, for example, those marketed under the trademark“Oxide Zinc CS-5” by Toshiba (ZnO coated with polymethylhydrosiloxane); those marketed under the trademark “Nanogard Zinc Oxide FN” by Nanophase Technologies (as a 40% dispersion in Finsolv TN, C12-C15 alkyl benzoate); those marketed under the trademark “Daitopersion Zn-30” and “Daitopersion Zn-50” by Daito (dispersions in oxyethylenated polydimethylsiloxane/cyclopolymethylsiloxane comprising 30% or 50% of zinc nano-oxides coated with silica and polymethylhydrosiloxane); those marketed under the trademark“NFD Ultrafine ZnO” by Daikin (ZnO coated with phosphate of perfiuoroalkyl and a copolymer based on perfluoroalkylethyl as a dispersion in
- the uncoated iron oxide pigments are, for example, marketed by Arnaud under the trademarks“Nanogard WCD 2002 (FE 45B)”,“Nanogard Iron FE 45 BL AQ”,“Nanogard FE 45R AQ”, and“Nanogard WCD 2006 (FE 45R)”, or by
- the coated iron oxide pigments are, for example, marketed by Arnaud under the trademarks“Nanogard WCD 2008 (FE 45B FN)”,“Nanogard WCD 2009 (FE 45B 556)”,“Nanogard FE 45 BL 345”, and“Nanogard FE 45 BL”, or by BASF under the trademark“Oxyde de fer transparent”.
- Coated inorganic UV filters are desirable, because the UV filtering effects of the inorganic UV filters can be enhanced.
- the coating(s) may help uniformly or homogeneously disperse the UV filters in the composition, according to the present invention.
- composition comprises a UV filter system comprising at least one organic UV filter. If two or more organic UV filters are used, they may be the same or different.
- the organic UV filter used for the present invention may be active in the UV- A and/or UV-B region.
- the organic UV filter may be hydrophilic and/or lipophilic.
- the organic UV filter may be solid or liquid.
- solid and liquid mean solid and liquid, respectively, at 25°C under 1 atm.
- the organic UV filter can be selected from the group consisting of anthranilic compounds; dibenzoylmethane compounds; cinnamic compounds; salicylic compounds; camphor compounds; benzophenone compounds; b,b- diphenylacrylate compounds; triazine compounds; benzotriazole compounds; benzalmalonate compounds; benzimidazole compounds; imidazoline compounds; bis-benzoazolyl compounds; p-aminobenzoic acid (PABA) compounds; methylenebis(hydroxyphenylbenzotriazole) compounds; benzoxazole compounds; screening polymers and screening silicones; dimers derived from a-alkylstyrene; 4,4- diarylbutadienes compounds; guaiazulene and derivatives thereof; rutin and derivatives thereof; flavonoids; bioflavonoids; oryzanol and derivatives thereof; quinic acid and derivatives thereof; phenols; retinol; cysteine; aromatic amino acids; peptid
- Anthranilic compounds menthyl anthranilates, such as marketed under the trademark“Neo Heliopan MA” by Haarmann and Reimer.
- the dibenzoylmethane compounds Butyl methoxydibenzoylmethane, such as marketed in particular under the trademark “Parsol 1789” by Hoffmann-La Roche; and isopropyl dibenzoylmethane.
- Cinnamic compounds Ethylhexyl methoxycinnamate, such as marketed in particular under the trademark “Parsol MCX” by Hoffmann-La Roche; isopropyl methoxycinnamate; isopropoxy methoxycinnamate; isoamyl methoxycinnamate, such as marketed under the trademark “Neo Heliopan E 1000” by Haarmann and Reimer; cinoxate (2- ethoxyethyl-4-methoxy cinnamate); DEA methoxycinnamate; diisopropyl methylcinnamate; and glyceryl ethylhexanoate dimethoxycinnamate.
- Salicylic compounds Homosalate (homomentyl salicylate), such as marketed under the trademark“Eusolex HMS” by Rona/EM Industries; ethylhexyl salicylate, such as marketed under the trademark“Neo Heliopan OS” by Haarmann and Reimer; glycol salicylate; butyloctyl salicylate; phenyl salicylate; dipropyleneglycol salicylate, such as marketed under the trademark“Dipsal” by Scher; and TEA salicylate, such as marketed under the trademark “Neo Heliopan TS” by Haarmann and Reimer.
- Homosalate homomentyl salicylate
- ethylhexyl salicylate such as marketed under the trademark“Neo Heliopan OS” by Haarmann and Reimer
- glycol salicylate butyloctyl salicylate
- phenyl salicylate dipropyleneglycol salicylate
- TEA salicylate
- Camphor compounds in particular, benzylidenecamphor derivatives: 3-benzylidene camphor, such as manufactured under the trademark“Mexoryl SD” by Chimex; 4- methylbenzylidene camphor, such as marketed under the trademark“Eusolex 6300” by Merck; benzylidene camphor sulfonic acid, such as manufactured under the trademark“Mexoryl SL” by Chimex; camphor benzalkonium methosulfate, such as manufactured under the trademark “Mexoryl SO” by Chimex; terephthalylidene dicamphor sulfonic acid, such as manufactured under the trademark“Mexoryl SX” by Chimex; and polyacrylamidomethyl benzylidene camphor, such as manufactured under the trademark “Mexoryl SW” by Chimex.
- 3-benzylidene camphor such as manufactured under the trademark“Mexoryl SD” by Chimex
- 4- methylbenzylidene camphor such as
- Benzophenone compounds Benzophenone-1 (2,4-dihydroxybenzophenone), such as marketed under the trademark“Uvinul 400” by BASF; benzophenone-2 (Tetrahydroxybenzophenone), such as marketed under the trademark“Uvinul D50” by BASF; Benzophenone-3 (2- hydroxy-4-methoxybenzophenone) or oxybenzone, such as marketed under the trademark“Uvinul M40” by BASF; benzophenone-4 (hydroxymethoxy benzophonene sulfonic acid), such as marketed under the trademark “Uvinul MS40” by BASF; benzophenone-5 (Sodium hydroxymethoxy benzophenone Sulfonate); benzophenone-6 (dihydroxy dimethoxy benzophenone); such as marketed under the trademark“Helisorb 1 1” by Norquay; benzophenone-8, such as marketed under the trademark “Spectra-Sorb UV-24” by American Cyanamid; benzophenone-9 (D
- Triazine compounds Diethylhexyl butamido triazone, such as marketed under the trademark“Uvasorb HEB” by Sigma 3V; 2,4,6-tris(dineopentyl 4'-aminobenzalmalonate)-s-triazine, bis- ethylhexyloxyphenol methoxyphenyl triazine, such as marketed under the trademark «TINOSORB S » by CIBA GEIGY, and ethylhexyl triazone, such as marketed under the trademark «UVTNUL T150 » by BASF.
- Benzotriazole compounds in particular, phenylbenzotriazole derivatives: 2-(2H-benzotriazole-2-yl)-6-dodecyl-4-methylpheno, branched and linear; and those described in USP 5240975.
- Benzalmalonate compounds Dineopentyl 4'-methoxybenzalmalonate, and polyorganosiloxane comprising benzalmalonate functional groups, such as polysilicone-15, such as marketed under the trademark“Parsol SLX” by Hoffmann-LaRoche.
- Benzimidazole compounds in particular, phenylbenzimidazole derivatives: Phenylbenzimidazole sulfonic acid, such as marketed in particular under the trademark“Eusolex 232” by Merck, and disodium phenyl dibenzimidazole tetrasulfonate, such as marketed under the trademark “Neo Heliopan AP” by Haarmann and Reimer.
- Imidazoline compounds Ethylhexyl dimethoxybenzylidene dioxoimidazoline propionate.
- Bis- benzoazolyl compounds The derivatives as described in EP-669,323 and U.S. Pat. No. 2,463,264.
- Para-aminobenzoic acid compounds PABA (p-aminobenzoic acid), ethyl PABA, Ethyl dihydroxypropyl PABA, pentyl dimethyl PABA, ethylhexyl dimethyl PABA, such as marketed in particular under the trademark“Escalol 507” by ISP, glyceryl PABA, and PEG-25 PABA, such as marketed under the trademark“Uvinul P25” by BASF.
- PABA p-aminobenzoic acid
- ethyl PABA Ethyl dihydroxypropyl PABA
- pentyl dimethyl PABA ethylhexyl dimethyl PABA
- PEG-25 PABA such as marketed under the trademark“Uvinul P25” by BASF.
- Methylene bis-(hydroxyphenylbenzotriazol) compounds such as 2,2'- methylenebis[6-(2H-benzotriazol-2-yl)-4-methyl-phenol], such as marketed in the solid form under the trademark “Mixxim BB/200” by Fairmount Chemical, 2,2'- methylenebis[6-(2H-benzotriazol-2-yl)-4-(l,l,3,3-tetramethylbutyl)phenol], such as marketed in the micronized form in aqueous dispersion under the trademark “Tinosorb M” by BASF, or under the trademark “Mixxim BB/100” by Fairmount Chemical, and the derivatives as described in U.S. Pat. Nos.
- Drometrizole trisiloxane such as marketed under the trademark“Silatrizole” by Rhodia Chimie or- “Mexoryl XL” by L’Oreal.
- Benzoxazole compounds 2,4-bis[5- l(dimethylpropyl)benzoxazol-2-yl-(4-phenyl)imino]- 6-(2-ethylhexyl)imino-l,3,5-triazine, such as marketed under the trademark of Uvasorb K2A by Sigma 3V.
- the organic UV filter(s) be selected from the group consisting of: butyl methoxydibenzoylmethane, ethylhexyl methoxycinnamate, homosalate, ethylhexyl salicylate, octocrylene, phenylbenzimidazole sulfonic acid, benzophenone-3, benzophenone-4, benzophenone-5, n-hexyl 2-(4-diethylamino-2-hydroxybenzoyl)benzoate, l,r-(l,4- piperazinediyl)bis[l-[2-[4-(diethylamino)-2-hydroxybenzoyl]phenyl]-methanone 4- methylbenzylidene camphor, terephthalylidene dicamphor sulfonic acid, disodium phenyl dibenzimidazole tetrasulfonate, ethyl
- composition of the present invention can also comprise additional fatty compounds selected from oils, waxes, fatty acids, fatty alcohols, and mixtures thereof.
- additional fatty compounds selected from oils, waxes, fatty acids, fatty alcohols, and mixtures thereof.
- additional fatty compounds does not include ozokerite wax.
- the proportion of additional fatty compounds in the anhydrous cosmetic composition according to the invention is generally of at least about 20% by weight, relative to the total weight of the composition, preferably ranging from 20% by weight to 70% by weight, more preferably from about 25% by weight to about 65% by weight, even more preferably from about 30% by weight to about 60% by weight, relative to the total weight of the composition
- the waxes useful for the present invention may be of mineral, fossil, animal, or vegetable origin, hydrogenated oils, or mixtures thereof.
- waxes include hydrocarbon-based waxes such as polyethylene wax, beeswax, ouricury wax, paraffin, microcrystalline wax, synthetic wax, japan wax, cork fiber waxes or sugar cane waxes, euphorbia cerifera (candelilla) wax, copernicia cerifera (carnauba) wax, lignite waxes, lanolin wax, montan wax, ethylenediamine/stearyl dimer dilinoleate copolymer, hydrogenated oils and glycerides that are solid at 25°C. It is also possible to use silicone waxes, among which mention may be made of alkyl, alkoxy and/or esters of polymethylsiloxane.
- oils which can be used in the invention mention may be made to polar or slightly polar oils, i.e. oils including an alkyl chain, preferably a C3-C40 alkyl chain.
- oils to be used in the present invention include:
- linear or branched hydrocarbons such as liquid paraffin, isohexadecane, liquid petroleum jelly and light naphthalene oils, and lanolin, hydrocarbon-based oils of plant origin, such as glyceride triesters, which are generally triesters of fatty acids and of glycerol, the fatty acids of which can have varied chain lengths from C4 to C24, it being possible for these chains to be saturated or unsaturated and linear or branched; these oils are in particular wheat germ oil, sunflower oil, grape seed oil, sesame oil, corn oil, apricot oil, castor oil, shea oil, avocado oil, olive oil, soybean oil, sweet almond oil, palm oil, rapeseed oil, cottonseed oil, hazelnut oil, macadamia oil, jojoba oil, alfalfa oil, poppy oil, pumpkin seed oil, marrow oil, blackcurrant oil, evening primrose oil, millet oil, barley oil, quinoa oil, rye oil, sa
- esters for instance oils of formula RCOOR' in which R represents a linear or branched fatty acid residue containing from 1 to 40 carbon atoms and R' represents a hydrocarbon-based chain that is especially branched, containing from 1 to 40 carbon atoms, on condition that R + R’ is >10, for instance, cetearyl octanoate, isopropyl palmitate, C12-C15 alkyl benzoate, 2-ethylphenyl benzoate, isopropyl lanolate, hexyl laurate, ethylhexyl palmitate, isononyl isononanoate, oleyl erucate, 2-ethylhexyl palmitate, isostearyl isostearate, octanoates, decanoates or ricinoleates of alcohols or polyalcohols, such as propylene glycol dioctanoate; hydroxylated esters, such as isostea
- silicone oils such as polydimethylsiloxanes (PDMS's), optionally including a C3-C40 alkyl or alkoxy chain or a phenyl chain, such as phenyltrimethicones, optionally fluorinated polyalkylmethylsiloxanes, such as polymethyltrifluoropropyldimethylsiloxanes, or with functional groups such as hydroxyl, thiol and/or amine groups; polysiloxanes modified with fatty acids, fatty alcohols or polyoxyalkylenes, fluorosilicones and perfluoro oils; mixtures thereof.
- PDMS's polydimethylsiloxanes
- PDMS's polydimethylsiloxanes
- a C3-C40 alkyl or alkoxy chain or a phenyl chain such as phenyltrimethicones
- fluorinated polyalkylmethylsiloxanes such as polymethyltrifluoropropyld
- Non-liming examples of fatty alcohols useful for the present invention are those liquid at room temperature, containing a branched and/or unsaturated carbon-based chain containing from 12 to 26 carbon atoms, for instance octyldodecanol, isostearyl alcohol, behenyl alcohol, oleyl alcohol, 2-hexyldecanol, 2- butyloctanol or 2-undecylpentadecanol.
- Non-limiting examples of fatty acids useful for the present invention are higher fatty C12-C22 acids, such as oleic acid, linoleic acid or linolenic acid.
- the additional fatty compounds are selected from the group consisting of synthetic esters, waxes, and mixtures thereof, preferably being selected from the group consisting of ethylhexyl palmitate, diisostearyl malate, polyethylene, and mixtures thereof.
- the anhydrous cosmetic composition according to the present invention may comprise pigments.
- the suitable pigments used in the anhydrous cosmetic composition of the present invention may be coated or uncoated.
- the coated pigments are pigments which have undergone one or more surface treatments of a chemical, electronic, mechanochemical and/or mechanical nature with compounds such as those described for example in Cosmetics & Toiletries, February 1990, vol. 105, p.53-64, such as amino acids, beeswax, fatty acids, fatty alcohols, anionic surface-active agents, lecithin, sodium, potassium, zinc, iron or aluminum salts of fatty acids, (titanium or aluminum) metal alkoxides, polyethylene wax, silicones, proteins (collagen, elastin), alkanolamines, silicon oxides, metal oxides or sodium hexametaphosphate.
- a chemical, electronic, mechanochemical and/or mechanical nature such as those described for example in Cosmetics & Toiletries, February 1990, vol. 105, p.53-64, such as amino acids, beeswax, fatty acids, fatty alcohols, anionic surface-active agents, lecithin, sodium, potassium, zinc,
- Coated pigments are more particularly titanium oxides coated with silica, silica and iron oxide, silica and alumina, alumina and aluminum stearate, alumina and aluminum laurate, iron oxide and iron stearate, zinc oxide and zinc stearate, silica, alumina and silicone, alumina, aluminum stearate and silicone, alumina and silicone, etc.
- metal oxides may also be mentioned, especially titanium dioxide and cerium dioxide, including the silica-coated equiponderous mixture of titanium dioxide and cerium dioxide, as well as the alumina-silica- and silicone- coated mixture of titanium oxide and zinc dioxide, or the alumina-, silica- and glycerin-coated mixture of titanium dioxide and zinc dioxide.
- uncoated iron oxides, titanium oxides, particularly titanium dioxide, zinc oxides and cerium oxide may be used in the cosmetic composition of the present invention.
- Particular embodiments include iron oxide, triiron tetraoxide, diiron trioxide and titanium dioxide.
- the amount of the pigments in the anhydrous cosmetic composition of the invention preferably ranges from about 0.1 % to about 30% by weight, more preferably from about 0.1 % to about 25% by weight, even more preferably from about 0.1 % by weight to about 20% by weight, relative to the total weight of the composition.
- composition of the invention may further comprise any usual cosmetically acceptable ingredient, which may be chosen especially from perfume/fragrance, preserving agents, antioxidants, solvents, actives, vitamins, fillers, silicones, polymers, and mixtures thereof.
- Suitable polymers other than hydrogenated polyisobutene, polyvinyl laurate and mixtures thereof, include, but are not limited to xanthan gum, poly C10-30 alkyl acrylate, acrylates/Cio-30 alkyl acrylate crosspolymer, styrene/acrylates copolymer, methyl methacrylate crosspolymer and mixtures thereof.
- Non-limiting example of preserving agent which can be used in accordance with the invention include phenoxyethanol.
- An example of antioxidant is pentaerythrityl tetra-di-t-butyl hydroxyhydrocinnamate.
- Suitable fillers of the invention could be as examples of oil-absorbing fillers: mica, silica, zea may (corn) starch, magnesium oxide, nylon-12, nylon-66, cellulose, talc, talc (and) methicone, talc (and) dimethicone, perlite, sodium silicate, pumice, PTFE, polymethyl methacrylate, oryza sativa (rice) starch, aluminum starch octenylsuccinate, potato starch modified, alumina, calcium sodium borosilicate, magnesium carbonate, dimethicone/vinyl dimethicone crosspolymer, sodium carboxylmethyl starch.
- mica, silica, zea may (corn) starch, magnesium oxide, nylon-12, nylon-66, cellulose, talc, talc (and) methicone, talc (and) dimethicone, perlite, sodium silicate, pumice, PTFE, polymethyl methacrylate, oryza
- Suitable silicones include dimethicone and phenyl trimethicone.
- Suitable solvents include, but are not limited to alcohols, glycols and polyols such as glycerin, caprylyl glycol, pentylene glycol, propylene glycol, butylene glycol, and mixtures thereof.
- Suitable additional actives include, but are not limited to, disodium EDTA, triethanolamine, and mixtures thereof.
- Non-limiting example of vitamins suitable for the composition of the present invention includes tocopherol.
- the additional ingredients may represent from 0.1 % to 30%, such as from 0.1 % to 25% or such as from 0.1 to 20% by weight of the total weight of the composition of the invention.
- Anhydrous cosmetic compositions according to the present invention were prepared according to examples 1 to 3 below.
- An anti-acne clinical study was started to evaluate the anti-acne and comedogenic potential of the anhydrous cosmetic composition according to Example 2 by counting lesions (papules, pustules, comedones and microcysts) dermatologists and self-assessment. The test was performed with 60 female volunteers from 18 to 40 years old having skin phototype II to IV according to Fitzpatrick scale.
- DO baseline
- D28 28 days after of application
- D56 56 days after application
- D70 14 days after stopping using the product.
- Figures 1 to 6 show, respectively the mean count results of papules, pustules, total inflammatory lesions (sum of papules and pustules), comedones, microcysts and total non-inflammatory lesions (sum of comedones and microcysts) of the group of volunteers.
- anhydrous cosmetic composition according to the present invention has proved to be was proved to be capable of cause significative reduction in the number of lesions up to D56 in the normal use conditions.
- the maintenance of the face skin condition 14 days after interruption of the investigational product was maintained only in relation to the pustules and microcysts.
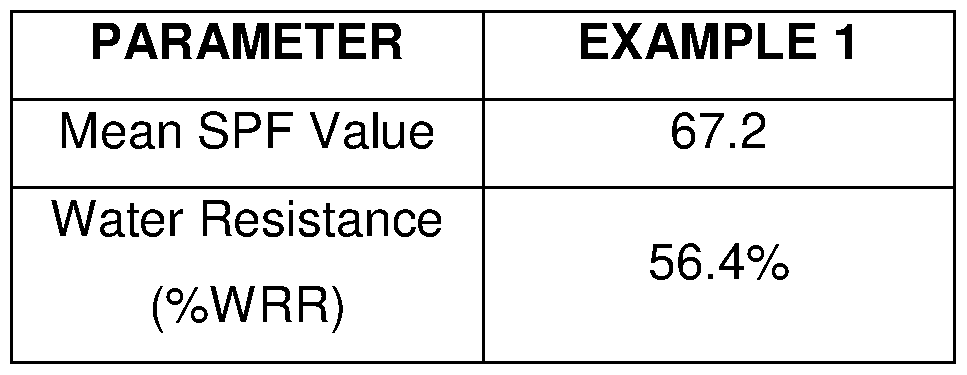
- compositions according to Example 1 was submitted to a SPF and a Water Resistance evaluation.
- the tests were performed following the ISO/EN 24444 Cosmetics Sun protection test-in vivo determination of the Sun Protection Factor (SPF) (2010).
- the Water resistance evaluation was performed according to the Colipa Guidelines (December 2005). The results are summarized in the table below.
- example 1 according to the present invention shown high mean SPF value and good water-resistance property.
- compositions were evaluated as to their fragrance, consistency, texture, color, amount of product applied, application, distribution, absorption, residues, package, sensation after application and during the day, sensation of protection and durability and interaction with water and sweat.
- composition of the prior art consists of caprylic/capric triglyceride, titanium dioxide, nylon-12, candelilla cera, ethylhexyl methoxycinnamate, oleyl alcohol, zinc oxide, octocrylene, microcrystalline wax, diethylamino hydroxybenzoyl hexyl benzoate, phenoxyethanol, talc, tocopheryl acetate, BHT, iron oxides, perfume, benzyl salicylate, D-limonene, alpha-isomethyl ionone and hydroxyisohexyl 3- cyclohexene carboxaldehyde, thereby being absent of silica aerogel, ozokerite wax, and hydrogenated polyisobutene and/or polyvinyl laurate.
- composition according to the present invention has the advantage of having dry touch after application and during the day, matte effect, pleasant fragrance and sensation of protection because of its adherence/fast absorption to the skin.
- composition according to the prior art was deemed to have, according to most of the volunteers, greasy and oily consistency and texture, especially during the day. Moreover, many volunteers stated that the product has an unpleasant fragrance and criticized the high transfer of the product to clothing and accessories. Most of the volunteers preferred the composition according to the present invention over the prior art, especially because of the dry touch after application and during the day, natural look, fast absorption, matte effect and due to the fact that it does not come out of the skin easily.
- the product according to the present invention has a better sensorial perception when compared to the prior art.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Birds (AREA)
- Dermatology (AREA)
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Inorganic Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Cosmetics (AREA)
Abstract
Description
Claims
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BR112021023920-0A BR112021023920B1 (en) | 2019-07-31 | ANHYDROUS COSMETIC COMPOSITION, PROCESS FOR MANUFACTURING AN ANHYDROUS COSMETIC COMPOSITION AND USE OF THE ANHYDROUS COSMETIC COMPOSITION | |
| PCT/BR2019/050309 WO2021016682A1 (en) | 2019-07-31 | 2019-07-31 | Anhydrous cosmetic composition with high concentration of hydrophobic fillers and use thereof for reducing skin lesions and oiliness |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/BR2019/050309 WO2021016682A1 (en) | 2019-07-31 | 2019-07-31 | Anhydrous cosmetic composition with high concentration of hydrophobic fillers and use thereof for reducing skin lesions and oiliness |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2021016682A1 true WO2021016682A1 (en) | 2021-02-04 |
Family
ID=67614400
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/BR2019/050309 Ceased WO2021016682A1 (en) | 2019-07-31 | 2019-07-31 | Anhydrous cosmetic composition with high concentration of hydrophobic fillers and use thereof for reducing skin lesions and oiliness |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2021016682A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023247794A1 (en) * | 2022-06-24 | 2023-12-28 | Nouryon Chemicals International B.V. | Polyvinyl alcohol film-forming polymers for alcohol-based sunscreen formulations and methods of using same |
Citations (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2463264A (en) | 1942-12-23 | 1949-03-01 | Ciba Ltd | Derivatives of cyclic amidines and process of making same |
| US5166355A (en) | 1991-02-04 | 1992-11-24 | Fairmount Chemical Co., Inc. | Process for preparing substituted 2,2'-methylene-bis-[6-(2H-benzotriazol-2-yl)-4-hydrocarbyl-phenols] |
| WO1993004665A1 (en) | 1991-08-29 | 1993-03-18 | L'oreal | Filtering cosmetic composition containing a liposoluble filter polymer with hydrocarbonated structure and a filter silicone |
| US5237071A (en) | 1991-01-22 | 1993-08-17 | Fairmount Chemical Company, Inc. | Process for preparing 2,2'-methylene-bis(6-(2H-benzotriazol-2-yl)-4-hydrocarbyl phenols) |
| US5240975A (en) | 1988-04-11 | 1993-08-31 | Ciba-Geigy Corporation | Liquid substituted 2H-benzotriazole mixtures, stabilized compositions |
| EP0669323A1 (en) | 1994-02-24 | 1995-08-30 | Haarmann & Reimer Gmbh | Utilization of benzazols as UV-absorbers, new benzazoles and process for their preparation |
| GB2303549A (en) | 1995-07-22 | 1997-02-26 | Ciba Geigy Ag | Micronising organic UV absorbers with alkyl polyglucosides |
| WO1997025022A1 (en) * | 1996-01-08 | 1997-07-17 | Stiefel Laboratories (Ireland) Limited | Skin care composition |
| DE19726184A1 (en) | 1997-06-20 | 1998-12-24 | Beiersdorf Ag | Oil-in-water or multiple emulsion with high concentration of suspended UVB filter |
| EP0893119A1 (en) | 1997-07-26 | 1999-01-27 | Ciba SC Holding AG | UV-protection formulation |
| DE19855649A1 (en) | 1998-12-03 | 2000-06-08 | Basf Ag | Dimeric alpha-alkyl-styrene derivatives as photostable UV filters in cosmetic and pharmaceutical preparations |
| US7470725B2 (en) | 1996-11-26 | 2008-12-30 | Cabot Corporation | Organically modified aerogels, processes for their preparation by surface modification of the aqueous gel, without prior solvent exchange, and subsequent drying, and their use |
| WO2009120602A1 (en) * | 2008-03-25 | 2009-10-01 | Mary Kay Inc. | Sebum absorption compositions |
| EP2863872A1 (en) * | 2012-06-21 | 2015-04-29 | L'Oréal | Cosmetic composition comprising a hydrocarbonated-based resin, a hydrocarbon-based block copolymer, a non volatile phenyl dimethicone oil and a non volatile hydrocarbonated oil |
| EP2923736A1 (en) * | 2014-03-28 | 2015-09-30 | Johnson & Johnson Consumer Companies, Inc. | Method of making an anhydrous, pigmented composition |
| WO2017112982A1 (en) * | 2015-12-29 | 2017-07-06 | L'oreal | Cosmetic compositions with silica aerogel sun protection factor boosters |
-
2019
- 2019-07-31 WO PCT/BR2019/050309 patent/WO2021016682A1/en not_active Ceased
Patent Citations (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2463264A (en) | 1942-12-23 | 1949-03-01 | Ciba Ltd | Derivatives of cyclic amidines and process of making same |
| US5240975A (en) | 1988-04-11 | 1993-08-31 | Ciba-Geigy Corporation | Liquid substituted 2H-benzotriazole mixtures, stabilized compositions |
| US5237071A (en) | 1991-01-22 | 1993-08-17 | Fairmount Chemical Company, Inc. | Process for preparing 2,2'-methylene-bis(6-(2H-benzotriazol-2-yl)-4-hydrocarbyl phenols) |
| US5166355A (en) | 1991-02-04 | 1992-11-24 | Fairmount Chemical Co., Inc. | Process for preparing substituted 2,2'-methylene-bis-[6-(2H-benzotriazol-2-yl)-4-hydrocarbyl-phenols] |
| WO1993004665A1 (en) | 1991-08-29 | 1993-03-18 | L'oreal | Filtering cosmetic composition containing a liposoluble filter polymer with hydrocarbonated structure and a filter silicone |
| EP0669323A1 (en) | 1994-02-24 | 1995-08-30 | Haarmann & Reimer Gmbh | Utilization of benzazols as UV-absorbers, new benzazoles and process for their preparation |
| GB2303549A (en) | 1995-07-22 | 1997-02-26 | Ciba Geigy Ag | Micronising organic UV absorbers with alkyl polyglucosides |
| WO1997025022A1 (en) * | 1996-01-08 | 1997-07-17 | Stiefel Laboratories (Ireland) Limited | Skin care composition |
| US7470725B2 (en) | 1996-11-26 | 2008-12-30 | Cabot Corporation | Organically modified aerogels, processes for their preparation by surface modification of the aqueous gel, without prior solvent exchange, and subsequent drying, and their use |
| DE19726184A1 (en) | 1997-06-20 | 1998-12-24 | Beiersdorf Ag | Oil-in-water or multiple emulsion with high concentration of suspended UVB filter |
| EP0893119A1 (en) | 1997-07-26 | 1999-01-27 | Ciba SC Holding AG | UV-protection formulation |
| DE19855649A1 (en) | 1998-12-03 | 2000-06-08 | Basf Ag | Dimeric alpha-alkyl-styrene derivatives as photostable UV filters in cosmetic and pharmaceutical preparations |
| WO2009120602A1 (en) * | 2008-03-25 | 2009-10-01 | Mary Kay Inc. | Sebum absorption compositions |
| EP2863872A1 (en) * | 2012-06-21 | 2015-04-29 | L'Oréal | Cosmetic composition comprising a hydrocarbonated-based resin, a hydrocarbon-based block copolymer, a non volatile phenyl dimethicone oil and a non volatile hydrocarbonated oil |
| EP2923736A1 (en) * | 2014-03-28 | 2015-09-30 | Johnson & Johnson Consumer Companies, Inc. | Method of making an anhydrous, pigmented composition |
| WO2017112982A1 (en) * | 2015-12-29 | 2017-07-06 | L'oreal | Cosmetic compositions with silica aerogel sun protection factor boosters |
Non-Patent Citations (4)
| Title |
|---|
| BRINKER, C.J.SCHERER, G.W.: "Sol-Gel Science", 1990, ACADEMIC PRESS |
| COSMETICS & TOILETRIES, vol. 105, February 1990 (1990-02-01), pages 53 - 64 |
| JOURNAL OF THE AMERICAN CHEMICAL SOCIETY, vol. 60, February 1938 (1938-02-01), pages 309 |
| VAN DE HULST, H.C.: "Light Scattering by Small Particles", 1957, WILEY |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023247794A1 (en) * | 2022-06-24 | 2023-12-28 | Nouryon Chemicals International B.V. | Polyvinyl alcohol film-forming polymers for alcohol-based sunscreen formulations and methods of using same |
Also Published As
| Publication number | Publication date |
|---|---|
| BR112021023920A2 (en) | 2022-02-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2775994B1 (en) | Composition with a continuous oil phase containing at least one lipophilic organic uv-screening agent and hydrophobic silica aerogel particles | |
| US20240197607A1 (en) | Non-pulverulent antisun composition comprising a polar oil phase and hydrophobic silica aerogel particles | |
| EP2981244B1 (en) | Composition containing composite particles for screening out uv radiation, with a mean size of greater than 0.1 micron, and hydrophobic silica aerogel particles | |
| EP2775993B1 (en) | Solid anti-sun composition based on lipophilic organic uv screening agent and aerogel particles of hydrophobic silica | |
| US12121602B2 (en) | Anhydrous sunscreen composition, process of manufacturing the anhydrous sunscreen composition and use of the anhydrous sunscreen composition | |
| EP3197563B1 (en) | Composition based on a lipophilic organic uv-screening agent and a filler | |
| WO2020154779A1 (en) | Sunscreen compositions, process of manufacturing the sunscreen composition, use of the sunscreen compositions, and use of emulsified carnauba wax | |
| WO2021119770A1 (en) | Cosmetic composition for a keratin substance, process for manufacturing a cosmetic composition and use of the cosmetic composition | |
| WO2022020913A1 (en) | Anhydrous cosmetic sunscreen composition, process of manufacturing the anhydrous cosmetic sunscreen composition and use of the anhydrous cosmetic sunscreen composition | |
| US20220241159A1 (en) | Cosmetic compositions in a soft-solid format comprising a hydrophobic powder selected from silica aerogel and polylactic acid, starch and an uv-filter system | |
| FR2986423A1 (en) | Composition comprising UV filter system, used to care and make up human keratin materials, comprises oil phase comprising polar oil, 4-carboxy-2-pyrrolidinone derivative and lipophilic UV filter, and hydrophobic silica aerogel particles | |
| WO2021016682A1 (en) | Anhydrous cosmetic composition with high concentration of hydrophobic fillers and use thereof for reducing skin lesions and oiliness | |
| WO2021016680A1 (en) | Anhydrous sunscreen composition comprising silica aerogel | |
| WO2022000052A1 (en) | Anhydrous cosmetic compositions, process of manufacturing a cosmetic composition and use of the anhydrous cosmetic compositions | |
| WO2023159282A1 (en) | Cosmetic composition for reducing oilness and shine of the skin and use of the cosmetic composition | |
| EP4076347A1 (en) | Cosmetic composition, process of manufacturing the cosmetic composition and use of the cosmetic composition | |
| WO2025199597A1 (en) | Cosmetic sunscreen composition and use of the cosmetic sunscreen composition | |
| WO2021016679A1 (en) | Anhydrous sunscreen composition comprising silica aerogel | |
| BR112021023920B1 (en) | ANHYDROUS COSMETIC COMPOSITION, PROCESS FOR MANUFACTURING AN ANHYDROUS COSMETIC COMPOSITION AND USE OF THE ANHYDROUS COSMETIC COMPOSITION | |
| US20160120791A1 (en) | Anhydrous alcohol-free sunscreen composition for application onto wet or dry skin | |
| WO2022067403A1 (en) | Anhydrous cosmetic sunscreen composition, process for manufacturing an anhydrous cosmetic sunscreen composition and use of an anhydrous cosmetic sunscreen composition | |
| BR112022009276B1 (en) | Sunscreen composition, cosmetic composition, manufacturing process of the cosmetic composition, and use of the cosmetic composition. |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19752634 Country of ref document: EP Kind code of ref document: A1 |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112021023920 Country of ref document: BR |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 112021023920 Country of ref document: BR Kind code of ref document: A2 Effective date: 20211126 |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 19752634 Country of ref document: EP Kind code of ref document: A1 |