WO2020208541A1 - Composition comprising glp-1 analogue - Google Patents
Composition comprising glp-1 analogue Download PDFInfo
- Publication number
- WO2020208541A1 WO2020208541A1 PCT/IB2020/053345 IB2020053345W WO2020208541A1 WO 2020208541 A1 WO2020208541 A1 WO 2020208541A1 IB 2020053345 W IB2020053345 W IB 2020053345W WO 2020208541 A1 WO2020208541 A1 WO 2020208541A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- glp
- analogue
- liraglutide
- present
- pharmaceutical composition
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/22—Hormones
- A61K38/26—Glucagons
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/02—Inorganic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/08—Solutions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
Definitions
- the present invention relates to a pharmaceutical composition wherein the GLP-1 analogue is the active drug substance.
- the present invention relates to method of using the composition comprising GLP-1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof.
- the pharmaceutical compositions comprising GLP- 1 analogues according to the present invention are particularly useful for the treatment of disease(s) or disorder(s) which are particularly acute in nature and which require a short term but mild to moderate treatment, or even some chronic conditions which favorably respond to or are alleviated by the pharmaceutical compositions comprising GLP-1 analogues.
- the pharmaceutical compositions comprising GLP-1 analogues are useful prophylactically or therapeutically depending upon the pathological condition intended to be prevented or treated respectively.
- a pharmaceutical formulation is found to be physically unstable when it exhibits turbidity. Physical stability of the formulations is evaluated by means of visual inspection and turbidity after storage of the formulation at different temperatures in top fdled glass cartridges for various time periods. Visual inspection of the formulations is performed in a sharp focused light with a dark background. A formulation is classified physical unstable with respect to protein aggregation, when it shows visual turbidity in daylight. [00104] In one embodiment, the pharmaceutical composition according to the present invention is physically stable for a period of Bout 10-25 days at about 40 °C.
- Formulations of semaglutide were prepared as per Examples 1 and 2 compositions and following the method as per Example 1, except that the liraglutide was replaced with semaglutide in all the compositions formulated.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Diabetes (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Endocrinology (AREA)
- Engineering & Computer Science (AREA)
- Gastroenterology & Hepatology (AREA)
- Hematology (AREA)
- Dermatology (AREA)
- Immunology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Inorganic Chemistry (AREA)
- Emergency Medicine (AREA)
- Zoology (AREA)
- Obesity (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Medicinal Preparation (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
The present invention relates to pharmaceutical composition comprising GLP-1 analogue its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof. The present invention further relates to a liquid composition comprising GLP-1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof, processes for preparing the composition and methods of using it. More particularly, the present invention relates to a liquid composition comprising Liraglutide, Semaglutide, D-Liraglutide, D-Semaglutide or other GLP-1 analogue thereof, processes for preparing the composition and methods of using it.
Description
COMPOSITION COMPRISING GLP-1 ANALOGUE
FIELD OF THE INVENTION
[0001] The present invention relates to pharmaceuticals. Specifically, the present invention relates to a pharmaceutical composition comprising GLP-1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof. The present invention further relates to a liquid stable composition comprising GLP-1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof, processes for preparing the composition and methods of using it. More particularly, the present invention relates to a liquid composition comprising Liraglutide, Semaglutide, D-Liraglutide, D-Semaglutide or other analogue thereof, processes for preparing the composition and methods of using the same.
BACKGROUND OF THE INVENTION
[0002] Background description includes information that may be useful in understanding the present invention. It is not an admission that any of the information provided herein is prior art or relevant to the presently claimed invention, or that any publication specifically or implicitly referenced is prior art.
[0003] GLP-1 is a physiological hormone that has multiple actions on glucose, mediated by the GLP-1 receptors. It stimulates b-cell activity via its Gs- coupled receptor, leading to stimulation of b-cell proliferation and differentiation, insulin gene expression and insulin secretion. Importantly, GLP-1 enhances insulin release in a glucose-dependent manner and therefore has less risk of causing hypoglycemia.
[0004] Liraglutide, Semaglutide, D-Liraglutide and D-Semaglutide are analogs of the human glucagon like peptide (GLP)-land act as a GLP-1 receptor agonist. These are indicated for the treatment of patients with type 2 diabetes to improve glycemic control. D-Liraglutide is an analog of Liraglutide wherein the
amino acid at 2nd position is replaced with D-Alanine. D-Semaglutide is an analog of Semaglutide wherein the amino acid at2nd position is replaced with D-Alanine.
[0005] US8846618 and W003002136 discloses pharmaceutical formulation comprising: a GLP-1 compound; an isotonic agent; a buffer; and a preservative. US8114833 discloses formulation comprising at least one GLP-1 agonist, a disodium phosphate dihydrate buffer and propylene glycol. WO2016038521 discloses Liraglutide composition with buffer, propylene glycol and preservative. CN103800301 discloses pulsatile controlled-release tablet composition with Liraglutide. WO2018096460 disclose pharmaceutical composition comprising: (a) Liraglutide as active drug substance; (b) Buffer selected from the group consisting of Sodium citrate, arginine, disodium hydrogen phosphate, glycine or any combination thereof; (c) Isotonic agent selected from glycerol, mannitol, propylene glycol, xylitol and trehalose and (d) preservative selected from phenol, cresol, resorcinol, parabens or any combination thereof.
[0006] While there have attempts to develop compositions and formulations for various delivery modes in recent years, many have limited application because they cannot be given orally. This is due to a number of reasons including: poor oral toleration with complications including gastric irritation and bleeding; breakdown/degradation of the drug compounds in the stomach; and poor, slow or erratic absorption of the drug. Conventional protein and or peptide formulation for drug delivery routes such as subcutaneous, intravenous and intramuscular delivery need further improvements like better stability for example at various temperatures for solving long unaddressed need of providing a composition that can be formulated into suitable dosage form that can be stored or carried by a patient at an ordinary prevalent temperatures without having need to store under controlled temperature or refrigeration
[0007] Thus, there is an unmet need to provide compositions comprising GLP-1 analogue that can be used for treating diabetes that could be easily manufactured, scalable, economical, and can overcome deficiencies associated with the known arts as mentioned above.
OBJECTS OF THE INVENTION
[0008] An object of the present invention is to provide pharmaceutical composition comprising GLP-1 analogue, its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof, that can satisfy the existing needs and/or can generally overcome one or more deficiencies found in the existing art.
[0009] Another object of the present invention is to provide process for preparation of pharmaceutical composition comprising GLP-1 analogue, its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof, that can satisfy the existing needs and/or can generally overcome one or more deficiencies found in the existing art.
[0010] Yet another object of the present invention is to provide liquid pharmaceutical composition comprising GLP-1 analogue, its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof, that is economically viable to produce and/or that can overcome one or more of deficiencies found in the existing art.
[0011] Still another object of the present invention is to provide process for preparation of liquid pharmaceutical composition comprising GLP-1 analogue, its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof, that is economically viable to produce and/or that can overcome one or more of deficiencies found in the existing art.
[0012] An object of the present invention is to provide liquid pharmaceutical composition comprising GLP-1 analogue, its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof, that is easily scalable.
[0013] An object of the present invention is to provide process for preparation of liquid pharmaceutical composition comprising GLP-1 analogue, its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof, that is easily scalable.
SUMMARY OF THE INVENTION
[0014] This summary is provided to introduce a selection of concepts in a simplified form that are further described below in Detailed Description section. This summary is not intended to identify key features or essential features of the claimed subject matter, nor is it intended to be used as an aid in determining the scope of the claimed subject matter.
[0015] The present invention relates to pharmaceutical compositions comprising GLP-1 analogues.
[0016] In one aspect, the present invention relates to pharmaceutical compositions comprising GLP-1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof.
[0017] In another aspect, the present invention relates to process for preparing the composition comprising GLP-1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof.
[0018] In one aspect, the present invention relates to a liquid composition comprising GLP-1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof.
[0019] In another aspect, the present invention relates to process for preparing the liquid composition comprising GLP-1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof.
[0020] In still another aspect, the present invention relates to a liquid composition comprising GLP- 1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof, wherein the liquid composition is stable at various temperatures.
[0021] In still another aspect, the present invention relates to a process for preparing a liquid composition comprising GLP-1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates,
enantiomers or racemates thereof, wherein the liquid composition is stable at various temperatures.
[0022] In another aspect, the present invention relates to a liquid composition comprising Liraglutide, Semaglutide, D-Liraglutide, D-Semaglutide or other GLP-1 analogue thereof.
[0023] In still another aspect, the present invention relates to a process for preparing liquid composition comprising Liraglutide, Semaglutide, D-Liraglutide, D-Semaglutide or other GLP-1 analogue thereof.
[0024] In one aspect, the present invention relates to a pharmaceutical composition comprising GLP-1 analogue or its pharmaceutically acceptable complexes, salts, polymorphs, hydrates, solvates, enantiomers or racemates thereof, and at least one pharmaceutically acceptable excipient.
[0025] In another aspect, the present invention relates to a pharmaceutical composition comprising:
a) a GLP-1 analogue;
b) a buffer;
c) an isotonic agent; and
d) a preservative.
[0026] In one aspect, the present invention relates to a pharmaceutical composition wherein the GLP-1 analogue is the active drug substance.
[0027] In one aspect, the present invention relates to a pharmaceutical composition wherein the GLP-1 analogue is selected from the group consisting of Liraglutide, Semaglutide, D-Liraglutide, D-Semaglutide and the like as active drug substance.
[0028] In another aspect, the present invention relates to a pharmaceutical composition wherein buffer is selected from the group consisting of tromethamine (Tris), arginine, sodium phosphate, glycine, sodium phosphate dibasic dehydrate, tromethamine (Tris) acetate, tromethamine (Tris) phosphate, glutamic acid, acetate or any combination thereof.
[0029] In another aspect, the present invention relates to a pharmaceutical composition wherein isotonic agent is selected from the group consisting of
glycerol, Sucrose, propylene glycol, sorbitol, PEG 400, trehalose or any combination thereof.
[0030] In another aspect, the present invention relates to a pharmaceutical composition wherein the preservative is selected from the group consisting of phenol, cresol or any combination thereof.
[0031] In another aspect, the present invention relates to a process for preparation of GLP-1 analogue injection, said process comprising:
a) dissolving a buffering agent, an isotonic agent and a preservative in water for injection;
b) dissolving a GLP-1 analogue into buffer formulation solution of step (a); and
c) adjusting pH of formulated bulk solution of step (b) with a pH- adjusting agent to a pH range from about 7.0 to about 8.5.
[0032] Various objects, features, aspects and advantages of the inventive subject matter will become more apparent from the following detailed description of preferred embodiments.
BRIEF DESCRIPTION OF DRAWINGS THE INVENTION
[0033] The following drawings form part of the present specification and are included to further illustrate aspects of the present disclosure. The disclosure may be better understood by reference to the drawings in combination with the detailed description of the specific embodiments presented herein.
[0034] Figure 1 is a RP-HPLC purity profile of liraglutide formulations F2, F3, F4, F5, F6 under stress condition (40°C) in accordance with exemplary embodiments of the present disclosure.
[0035] Figure 2 is a RP-HPLC purity profile of liraglutide formulations F2, F6, F7, F8, under stress condition (40°C) in accordance with exemplary embodiments of the present disclosure.
[0036] Figure 3 is a SE-HPLC purity profile of liraglutide formulations F2, F6, F7, F8, under stress condition (40°C) in accordance with exemplary embodiments of the present disclosure.
[0037] Figure 4 is a RP-HPLC purity profile of liraglutide formulations F9, F10, FI 1, F12 under stress condition (40°C) in accordance with exemplary embodiments of the present disclosure.
[0038] Figure 5 is a SE-HPLC purity profile of liraglutide formulations F9, F10, FI 1, F12 under stress condition (40°C) in accordance with exemplary embodiments of the present disclosure.
[0039] Figure 6 is a SE-HPLC purity profile of liraglutide formulations FI 3, F14 at 2-8 °C in accordance with exemplary embodiments of the present disclosure.
[0040] Figure 7 is a RP-HPLC purity profile of liraglutide formulations FI 3, F14 at 2-8 °C in accordance with exemplary embodiments of the present disclosure.
[0041] Figure 8 is a SE-HPLC purity profile of liraglutide formulations F13, F14 at 25 °C in accordance with exemplary embodiments of the present disclosure.
[0042] Figure 9 is a RP-HPLC purity profile of liraglutide formulations FI 3, F14 at 25 °C in accordance with exemplary embodiments of the present disclosure.
DETAILED DESCRIPTION
[0043] The following is a detailed description of embodiments of the disclosure. The embodiments are in such detail as to clearly communicate the disclosure. However, the amount of detail offered is not intended to limit the anticipated variations of embodiments; on the contrary, the intention is to cover all modifications, equivalents, and alternatives falling within the spirit and scope of the present disclosure as defined by the appended claims.
[0044] All publications herein are incorporated by reference to the same extent as if each individual publication or patent application were specifically and individually indicated to be incorporated by reference. Where a definition or use of a term in an incorporated reference is inconsistent or contrary to the definition
of that term provided herein, the definition of that term provided herein applies and the definition of that term in the reference does not apply.
[0045] Reference throughout this specification to“one embodiment” or“an embodiment” means that a particular feature, structure or characteristic described in connection with the embodiment is included in at least one embodiment. Thus, the appearances of the phrases“in one embodiment” or“in an embodiment” in various places throughout this specification are not necessarily all referring to the same embodiment. Furthermore, the particular features, structures, or characteristics may be combined in any suitable manner in one or more embodiments.
[0046] In some embodiments, the numbers expressing quantities of ingredients, properties such as concentration, reaction conditions, and so forth, used to describe and claim certain embodiments of the invention are to be understood as being modified in some instances by the term“about”. The term “about” in some embodiments is + 5%. Accordingly, in some embodiments, the numerical parameters set forth in the written description and attached claims are approximations that can vary depending upon the desired properties sought to be obtained by a particular embodiment. In some embodiments, the numerical parameters should be construed in light of the number of reported significant digits and by applying ordinary rounding techniques. Notwithstanding that the numerical ranges and parameters setting forth the broad scope of some embodiments of the invention are approximations, the numerical values set forth in the specific examples are reported as precisely as practicable. The numerical values presented in some embodiments of the invention may contain certain errors necessarily resulting from the standard deviation found in their respective testing measurements.
[0047] As used in the description herein and throughout the claims that follow, the meaning of“a,”“an,” and“the” includes plural reference unless the context clearly dictates otherwise. Also, as used in the description herein, the meaning of “in” includes “in” and “on” unless the context clearly dictates otherwise.
[0048] Unless the context requires otherwise, throughout the specification which follow, the word“comprise” and variations thereof, such as,“comprises” and “comprising” are to be construed in an open, inclusive sense that is as “including, but not limited to.”
[0049] The recitation of ranges of values herein is merely intended to serve as a shorthand method of referring individually to each separate value falling within the range. Unless otherwise indicated herein, each individual value is incorporated into the specification as if it were individually recited herein. All methods described herein can be performed in any suitable order unless otherwise indicated herein or otherwise clearly contradicted by context. The use of any and all examples, or exemplary language (e.g.“such as”) provided with respect to certain embodiments herein is intended merely to better illuminate the invention and does not pose a limitation on the scope of the invention otherwise claimed. No language in the specification should be construed as indicating any non-claimed element essential to the practice of the invention.
[0050] Groupings of alternative elements or embodiments of the invention disclosed herein are not to be construed as limitations. Each group member can be referred to and claimed individually or in any combination with other members of the group or other elements found herein. One or more members of a group can be included in, or deleted from, a group for reasons of convenience and/or patentability. When any such inclusion or deletion occurs, the specification is herein deemed to contain the group as modified thus fulfilling the written description of all Markush groups used in the appended claims.
[0051] The description that follows, and the embodiments described therein, is provided by way of illustration of an example, or examples, of particular embodiments of the principles and aspects of the present disclosure. These examples are provided for the purposes of explanation, and not of limitation, of those principles and of the disclosure.
[0052] It should also be appreciated that the present disclosure can be implemented in numerous ways, including as a system, a method or a device. In this specification, these implementations, or any other form that the invention may
take, may be referred to as processes. In general, the order of the steps of the disclosed processes may be altered within the scope of the invention.
[0053] The headings and abstract of the invention provided herein are for convenience only and do not interpret the scope or meaning of the embodiments.
[0054] The following discussion provides many example embodiments of the inventive subject matter. Although each embodiment represents a single combination of inventive elements, the inventive subject matter is considered to include all possible combinations of the disclosed elements. Thus, if one embodiment comprises elements A, B, and C, and a second embodiment comprises elements B and D, then the inventive subject matter is also considered to include other remaining combinations of A, B, C, or D, even if not explicitly disclosed.
[0055] Various terms as used herein are shown below. To the extent a term used in a claim is not defined below, it should be given the broadest definition persons in the pertinent art have given that term as reflected in printed publications and issued patents at the time of filing.
Definitions
[0056] The term "Pharmaceutical composition" refers to the combination of one or more drug substances and one or more excipients"
[0057] The term "Drug product," "pharmaceutical dosage form," "dosage form," "final dosage form" and the like, refer to a pharmaceutical composition that is administered to a subject in need of treatment and generally may be in the form of tablets, capsules, sachets containing powder or granules, pellets, liquid solutions or suspensions, patches, and the like.
[0058] The term 'pharmaceutically acceptable excipient' according to the present invention means, but not limited to, any inactive ingredient which is required for the formulation of Liraglutide in a suitable dosage form. Particularly the excipient includes, but not limited to, diluents, carriers, fillers, bulking agents, binders, disintegrants, polymer, lubricant, glidant, surface active agents, stabilizers, absorption accelerators, flavoring agents, preservatives, antioxidants,
buffering agents, and any other excipient commonly used in the pharmaceutical industry.
[0059] The term 'buffer’ according to the present invention means, but not limited to, is an aqueous solution consisting of a mixture of a weak acid and its conjugate base, or vice versa. Its pH changes very little when a small amount of strong acid or base is added to it. particularly the buffer includes, but not limited to, tromethamine (Tris), arginine, sodium phosphate and glycine, sodium phosphate dibasic dehydrate, tromethamine (Tris) acetate, tromethamine (Tris) phosphate, glutamic acid, acetate and combination thereof.
[0060] The term 'isotonic agent’ according to the present invention means, but not limited to, refers to two solutions having the same osmotic pressure across a semipermeable membrane. This state allows for the free movement of water across the membrane without changing the concentration of solutes on either side. Particularly the isotonic agent includes, but not limited to, glycerol, sucrose, propylene glycol, sorbitol, PEG 400 and trehalose or combinations thereof.
[0061] The term 'preservative’ according to the present invention means, but not limited to, a substance or a chemical that is added to prevent decomposition by microbial growth or by undesirable chemical changes. Particularly the preservative includes, but not limited to, phenol, cresol and parabens and combination thereof.
[0062] The term "pH-adjusting agent," according to the present invention means, but not limited to, an agent used to adjust the pH in the desired range. Particularly the preservative includes, but not limited to sodium hydroxide or hydrochloric acid and combination thereof.
[0063] The term“D-Liraglutide” according to the present invention means, but not limited to, a Liraglutide analog wherein D-Alanine is present at 2nd amino acid position instead of the conventional L- Alanine.
[0064] The term“D-Semaglutide” according to the present invention means, but not limited to, an analog of glucagon-like peptide- 1 (glp-1) receptor agonist wherein, the Aib (Amino isobutyric acid) is replaced with D-Alanine.
[0065] “Subject” includes humans, non-human mammals (e.g., dogs, cats, rabbits, cattle, horses, sheep and the like) or non-mammals (e.g., birds and the like).
[0066] The present invention relates to pharmaceutical compositions comprising GLP-1 analogues.
[0067] In one embodiment, the present invention relates to pharmaceutical compositions comprising GLP-1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof.
[0068] In another embodiment, the present invention relates to process for preparing the composition comprising GLP-1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof.
[0069] In another embodiment, the present invention relates to method of using the composition comprising GLP-1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof.
[0070] In one embodiment, the present invention relates to a liquid composition comprising GLP- 1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof.
[0071] In another embodiment, the present invention relates to process for preparing the liquid composition comprising GLP-1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof.
[0072] In still another embodiment, the present invention relates to method of using the liquid composition comprising GLP-1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof.
[0073] In still another embodiment, the present invention relates to a liquid composition comprising GLP- 1 analogue or its pharmaceutically acceptable salts,
complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof, wherein the liquid composition is stable at various temperatures.
[0074] In still another embodiment, the present invention relates to a process for preparing a liquid composition comprising GLP-1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof, wherein the liquid composition is stable at various temperatures.
[0075] In another embodiment, the present invention relates to a liquid composition comprising Liraglutide, Semaglutide, D-Liraglutide, D-Semaglutide or other GLP-1 analogue thereof.
[0076] In still another embodiment, the present invention relates to a process for preparing liquid composition comprising Liraglutide, Semaglutide, D- Liraglutide, D-Semaglutide or other GLP-1 analogue thereof.
[0077] In yet another embodiment, the present invention relates to methods of using the liquid composition comprising Liraglutide, Semaglutide, D-Liraglutide, D-Semaglutide or other GLP-1 analogue thereof.
[0078] In one embodiment, the present invention relates to a pharmaceutical composition comprising GLP-1 analogue or its pharmaceutically acceptable complexes, salts, polymorphs, hydrates, solvates, enantiomers or racemates thereof, and at least one pharmaceutically acceptable excipient.
[0079] In another embodiment, the present invention relates to a liquid stable pharmaceutical composition comprising:
a) a GLP-1 analogue;
b) a buffer;
c) an isotonic agent; and
d) a preservative.
[0080] In one embodiment, the present invention relates to a pharmaceutical composition wherein the GLP-1 analogue is the active drug substance.
[0081] In one embodiment, the present invention relates to a pharmaceutical composition wherein the GLP-1 analogue is selected from the group consisting of
Liraglutide, Semaglutide, D-Liraglutide, D-Semaglutide and the like as active drug substance.
[0082] In another embodiment, the present invention relates to a pharmaceutical composition wherein buffer is selected from the group consisting of tromethamine (Tris), arginine, sodium phosphate, glycine, sodium phosphate dibasic dehydrate, tromethamine (Tris) acetate, tromethamine (Tris) phosphate, glutamic acid, acetate or any combination thereof.
[0083] In another embodiment, the present invention relates to a pharmaceutical composition wherein isotonic agent is selected from the group consisting of glycerol, sucrose, propylene glycol, sorbitol, PEG 400, trehalose or any combination thereof.
[0084] In another embodiment, the present invention relates to a pharmaceutical composition wherein the preservative is selected from the group consisting of phenol, cresol or any combination thereof.
[0085] In another embodiment, the present invention relates to a process for preparation of GLP-1 analogue injection, said process comprising:
a) dissolving a buffering agent, an isotonic agent and a preservative in water for injection;
b) dissolving a GLP-1 analogue into buffer formulation solution of step
(a);
c) adjusting pH of formulated bulk solution of step (b) with a pH- adjusting agent to a pH range from about 7.0 to about 8.5.
[0086] In one embodiment, the present invention provides a pharmaceutical composition comprising GLP-1 analogue, wherein the formulation is maintained at a pH of about 7.0 to 8.5, more preferably at pH 7.8 to 8.3.
[0087] In an embodiment, the pH-adjusting agent is selected from hydrochloric acid, sodium hydroxide, o-phosphoric acid or glacial acetic acid using concentration range from 0.1N to 5N.
[0088] In another embodiment, the present invention provides a pharmaceutical composition comprising GLP-1 analogue, wherein the concentration of GLP-1 analogue is in the range from about O. lmg/ml to about
100 mg/ml. More preferably, the concentration of GLP-lanalogue is selected from about 1 mg/ml to about 10 mg/ml.
[0089] In another embodiment, the present invention provides a pharmaceutical composition comprising GLP-1 analogue, wherein the concentration of buffer is in the range from about 3 mM to aboutlOO mM. More preferably, the concentration of buffer is selected from about 3 mM to about 60 mM.
[0090] In another embodiment, the present invention provides a pharmaceutical composition comprising GLP-1 analogue, wherein the buffer is in the concentration ranging from about 0.01% to about 10%, preferably, from about 0.02 % to about 8.0 % w/w of the composition.
[0091] In another embodiment, the present invention provides a pharmaceutical composition comprising GLP-1 analogue, wherein the amount of isotonic agent is in the range from about 0.1 mg/ml to about 100 mg/ml. More preferably, the concentration of isotonic agent is selected from about 0.2 mg/ml to about 30 mg/ml.
[0092] In another embodiment, the present invention provides a pharmaceutical composition comprising GLP-1 analogue, wherein the concentration of isotonic agent is in the range from about 0.01% to about 10%. More preferably, the concentration of isotonic agent is selected from about 0.02 % to about 8 %.
[0093] In another embodiment, the present invention provides a pharmaceutical composition comprising GLP-1 analogue, wherein the concentration of preservative is in the range from about 0.01% w/v to about 10% w/v of the composition. More preferably, the concentration of GLP-1 is selected from about 0.01% w/v to about 5 % w/v.
[0094] The pharmaceutical compositions of the present invention can be manufactured by the processes well known in the art, for example, by means of conventional mixing, dissolving, dry granulation, wet granulation, dragee-making, levigating, emulsifying, encapsulating, entrapping, lyophilizing processes or spray drying. The pharmaceutical compositions comprising GLP-1 analogues may be
administered in the form of any pharmaceutical formulation. The pharmaceutical formulation will depend upon the nature of the active compound and its route of administration.
[0095] In an embodiment, the pharmaceutical compositions comprising GLP- 1 analogues according to the present invention are particularly useful for the treatment of disease(s) or disorder(s) which are particularly acute in nature and which require a short term but mild to moderate treatment, or even some chronic conditions which favorably respond to or are alleviated by the pharmaceutical compositions comprising GLP-1 analogues. The pharmaceutical compositions comprising GLP-1 analogues are useful prophylactically or therapeutically depending upon the pathological condition intended to be prevented or treated respectively.
[0096] In one embodiment pharmaceutical compositions comprising GLP-1 analogue of the present invention are useful in the prophylaxis, amelioration and/or treatment of diabetes, in a subject in need thereof.
[0097] In another embodiment pharmaceutical compositions comprising GLP-1 analogue of the present invention are useful in the prophylaxis, amelioration and/or treatment of diabetes, which comprises administrating to a subject in need thereof a pharmaceutically effective amount of GLP-1 analogues composition.
[0098] An embodiment of the present invention relates to methods of using pharmaceutical compositions comprising GLP-1 analogues of the present invention for the prophylaxis, amelioration and/or treatment of diabetes, which comprises administrating to a subject in need thereof a pharmaceutically effective amount of composition comprising GLP-1 analogue.
[0099] In yet another embodiment, the pharmaceutical compositions comprising GLP-1 analogues of the present invention are useful in the treatment of the aforementioned diseases, disorders and conditions in combination with another disease modifying drug. The compounds of the present invention may be used in combination with one or more other drugs in the treatment, prevention, suppression or amelioration of diseases or conditions for which pharmaceutical
compositions comprising GLP-1 analogues may have utility, where the combination of the drugs together is safer or more effective than either drug alone.
[00100] A further embodiment of the present invention is the use of a pharmaceutical compositions comprising GLP-1 analogues for the manufacture of a medicament for the prophylaxis, amelioration and/or treatment of diabetes, involving GLP-1 analogs in a subject in need thereof.
[00101] In still another embodiment of the present invention is the pharmaceutical compositions comprising GLP-1 analogues for use as a medicament for the prophylaxis, amelioration and/or treatment of diabetes, involving GLP-1 analogs, in a subject in need thereof preferably a mammal including a human.
[00102] Pharmaceutical compositions containing a GLP-1 analogue according to the present invention may be administered parenterally to patients in need of such a treatment. Parenteral administration may be performed by subcutaneous, intramuscular or intravenous injection by means of a syringe, optionally a pen-like syringe. Alternatively, parenteral administration can be performed by means of an infusion pump. A further option is a composition which may be a solution or suspension for the administration of the GLP-1 analogue in the form of a nasal or pulmonal spray. As a still further option, the pharmaceutical compositions containing the GLP-1 compound of the invention can also be adapted for transdermal administration, for example in the form of a patch, optionally a iontophoretic patch, or transmucosal for example for buccal administration.
[00103] A pharmaceutical formulation is found to be physically unstable when it exhibits turbidity. Physical stability of the formulations is evaluated by means of visual inspection and turbidity after storage of the formulation at different temperatures in top fdled glass cartridges for various time periods. Visual inspection of the formulations is performed in a sharp focused light with a dark background. A formulation is classified physical unstable with respect to protein aggregation, when it shows visual turbidity in daylight.
[00104] In one embodiment, the pharmaceutical composition according to the present invention is physically stable for a period of Bout 10-25 days at about 40 °C.
[00105] In one embodiment, the pharmaceutical composition according to the present invention is physically stable for a period up to about 6 months at about 2°C - about 8 °C.
[00106] In one embodiment, the pharmaceutical composition according to the present invention is physically stable for a period of up to about 6 months at about 25 °C.
[00107] In one embodiment, the pharmaceutical composition according to the present invention is found to be stable with no significant change in pH for a period of up to about 6 months at about 25 °C.
[00108] In one embodiment, the pharmaceutical composition according to the present invention is found to be stable with no significant change in purity of the active drug substance that is the GLP-1 analogue for a period of up to about 6 months at about 25 °C.
[00109] In one embodiment, the pharmaceutical composition according to the present invention is found to be stable with no significant change in the content of the active drug substance that is the GLP-1 analogue for a period of up to about 6 months at about 25 °C.
[00110] While the foregoing describes various embodiments of the disclosure, other and further embodiments of the disclosure may be devised without departing from the basic scope thereof. The scope of the invention is determined by the claims that follow. The invention is not limited to the described embodiments, versions or examples, which are included to enable a person having ordinary skill in the art to make and use the invention when combined with information and knowledge available to the person having ordinary skill in the art.
EXAMPLES
[00111] The present invention is further explained in the form of following examples. However, it is to be understood that the following examples are merely illustrative and are not to be taken as limitations upon the scope of the invention.
Example 1
Preparation of the GLP- 1 receptor agonist liraglutide Formulations
Step 1: Preparation of buffer formulation
[00112] Different sets of buffer formulations including excipients as listed in Table 1 were prepared as follows:
1) Each of the excipients in the quantities as per Tables 2, 4 and 9 were weighed.
2) Each of the excipients were dissolved one by one into water for injection.
3) pH of the solution was in range of about 7-9.5.
4) Volume was adjusted up to 100 ml using water for injection and in process testing was carried out.
5) Aseptic fdtration was carried out using 0.2-micron fdter.
Step 2: Preparation of concentrated form of bulk solution
1) Active drug substance equivalent to target concentration of about 6 mg/mL (±10%) was weighed.
2) Weighed quantity of active drug substance was dissolved in 50% -80 % volume of buffer formulation by stirring at optimum speed of about 100 rpm - about 200 rpm.
3) Adjustment of the pH was carried out using HCl/NaOH solution to target pH in the range of 6.5-8.5.
4) In process testing was carried out and final dilution was adjusted based on the results to achieve target concentration of the active drug substance to about 6 mg/mL (±10%).
5) pH was adjusted again and final volume was made up to 100 ml using formulation buffer prepared in step 1.
Step 3: Aseptic filtration and filing into containers
1) Aseptic filtration was carried out using 0.2-micron PES filter and filling into sterile containers (Cartridges and its component).
2) Labeling was carried out as per the label format, release testing and storage as per their storage condition till further use.
[00113] Different formulations of liraglutide were prepared with compositions as per below Table lby using above process.
Table 1 : Compositions of different Formulations
A. Formulations Set 1
[00114] Specific formulations denoted as Formulations Set 1, with compositions as mentioned in Table 2 were formulated comprising different isotonic agents using process as mentioned above. Purity determination by RP HPLC at different time interval was performed.
Table 2: Formulations set 1
Table 3: Purity profile observed with different Isotonic agents at different time interval at stress conditions
Observation:
[00115] Purity levels of active drug substance in terms of % of monomer peak of different formulations of Formulation Set 1, as determined by RP HPLC were found to be as per Table 3 for and as illustrated in Figure 1.
B. Formulations Set 2
[00116] Specific formulations denoted as Formulations Set 2, with compositions as mentioned in Table 4were formulated comprising different isotonic agents using process as mentioned above. Purity determination of monomer peak of active drug substance by RP HPLC and SE HPLC at different time interval under stress condition of temperature at 40 °C was performed.
Table 4: Formulations set 2
[00117] Results of purity of % main peak by RP HPLC and SE HPLC analysis of different formulations at different time intervals under stress condition at 40 °C
have been described below in Table5 and Table 6 respectively and illustrated by Figure 2 and Figure 3.
Table 5: Purity profile by RP HPLC at different time intervals and under stressed condition of 40 °C
Table 6: Purity profile by SE HPLC at different time intervals under stressed condition of 40 °C
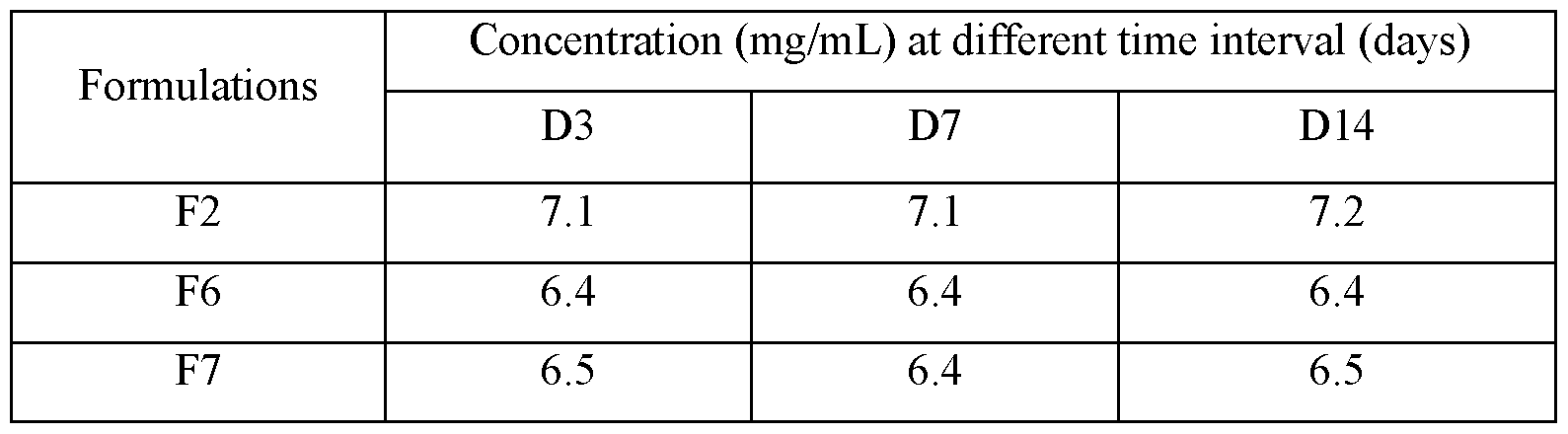
[00118] Concentration of liraglutide in different formulations was also determined to check any change in the content of liraglutide in presence of different isotonic agents under stress condition. Concentrations observed were as per Table 7.
Table 7: Concentration of liraglutide in presence of isotonic agent under stressed condition
[00119] The pH of the formulations was check at the initial and last time point. The pH at the initial time point and the last time point were found to be similar. The pH observations were found be as per Table 8.
Table 8: pH of formulation at initial and last time point under stressed condition
Observation:
[00120] Purity of liraglutide as determined by RP HPLC and SE HPLC in the presence of selected isotonic agents as per Table 5 and Table 6 respectively and Figure 2 and Figure 3 was found to be not affected by said isotonic agents. Also, there was no change in concentration of liraglutide and pH of the formulation under stress condition of 40 °C.
C. Formulations Set 3
[00121] Specific formulations denoted as Formulations Set 3, with compositions as mentioned in Table 9were formulated comprising different buffers using process as mentioned above.
Table 9: Composition of Formulations set 3
[00122] RP HPLC and SE HPLC were performed to determine % purity of liraglutide for all time points and pH was checked at the initial and last time point. Results of % purity of monomer peak of liraglutide by RP HPLC and SE HPLC are reported in Table 10 and Table 11 respectively. Plot of % purity by of monomer peak of liraglutide by RP HPLC and SE HPLC are illustrated in Figure 4 and Figure 5 showing no significant difference between any formulation whether by RP HPLC or SE HPLC.
Table 10: Purity profile by RP HPLC of formulations under stressed condition (40°C)
Table 11 : Purity profile by SE HPLC for selected buffering agents under stressed condition (40°C)
[00123] pH of the formulations was also checked at initial and at the end of 15 days of the study under stress condition of temperature at 40°C and reported in Table 12.
Table 12: pH of Formulations of set 3
Observation:
[00124] Purity of liraglutide as per different formulations as per Table 9 as determined by RP HPLC and SE HPLC as per Table 10 and Table 1 lrespectively and Figure 4 and Figure 5 was found to be not affected by the presence of selected isotonic agents under stress condition of 40 °C. Also, there was no change in the pH of the formulations under stress condition of 40 °C at the initial or the last time point as can be seen from Table 12.
Example 2
Formulations of Liraglutide
[00125] Formulations comprising liraglutide were prepared as per compositions of Table 13 and following method as described above under Example 1.
Table 13: Formulations of liraglutide
[00126] % Purity of monomer peak of liraglutide in each formulation was analyzed immediately after preparation of formulation at different time intervals under stress condition to determine stability as described hereinbelow under Example 6.
Example 3
Formulations of Semaglutide
[00127] Formulations of semaglutide were prepared as per Examples 1 and 2 compositions and following the method as per Example 1, except that the liraglutide was replaced with semaglutide in all the compositions formulated.
Example 4
Formulations of D-Liraglutide
[00128] Formulations of D-Firaglutide were prepared as per Examples 1 and 2 compositions and following the method as per Example 1, except that the liraglutide was replaced with D-Liraglutide in all the compositions formulated.
Example 5
Formulations of D-Semaglutide
[00129] Formulations of D-Semaglutide were prepared as per Examples 1 and
2 compositions and following the method as per Example 1, except that the liraglutide was replaced with D- Semaglutide in all the compositions formulated.
Example 6
Stability Studies of formulations of Liraglutide
[00130] Long term stability study at 2-8 °C and accelerated stability study at 25 °C of formulations (FI 3 and FI 4) of Examples 2 were carried out for three consistency batches of each of F13 and F14 formulations. Data obtained from the long-term study at 2-8 °C up to 6 months are presented below.
I. Long term stability study at 2-8 °C
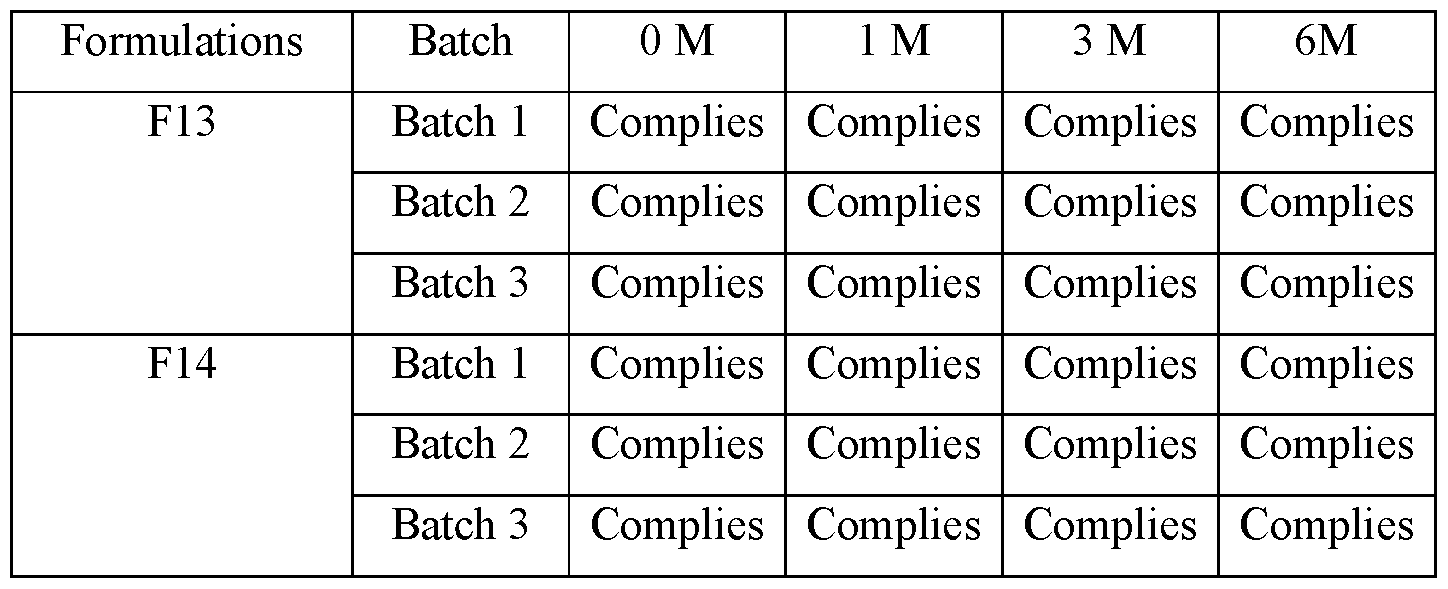
A. Physical appearance
[00131] No change in physical appearance was observed up to 6 months at 2-8 °C condition in any formulation. Observation of physical appearance are tabulated below in Table 14.
Table 1: results of Physical appearance under real time condition (2-8°C)
B. pH
[00132] No significant change in pH was observed up to 6 months at 2-8 °C condition in any formulation. pH is in range of 7.8 to 8.1 for all the formulation.
Observations of pH are tabulated below in Table 15.
Table 2: Results of pH under real time condition (2-8°C)
C. Purity by SE HPLC
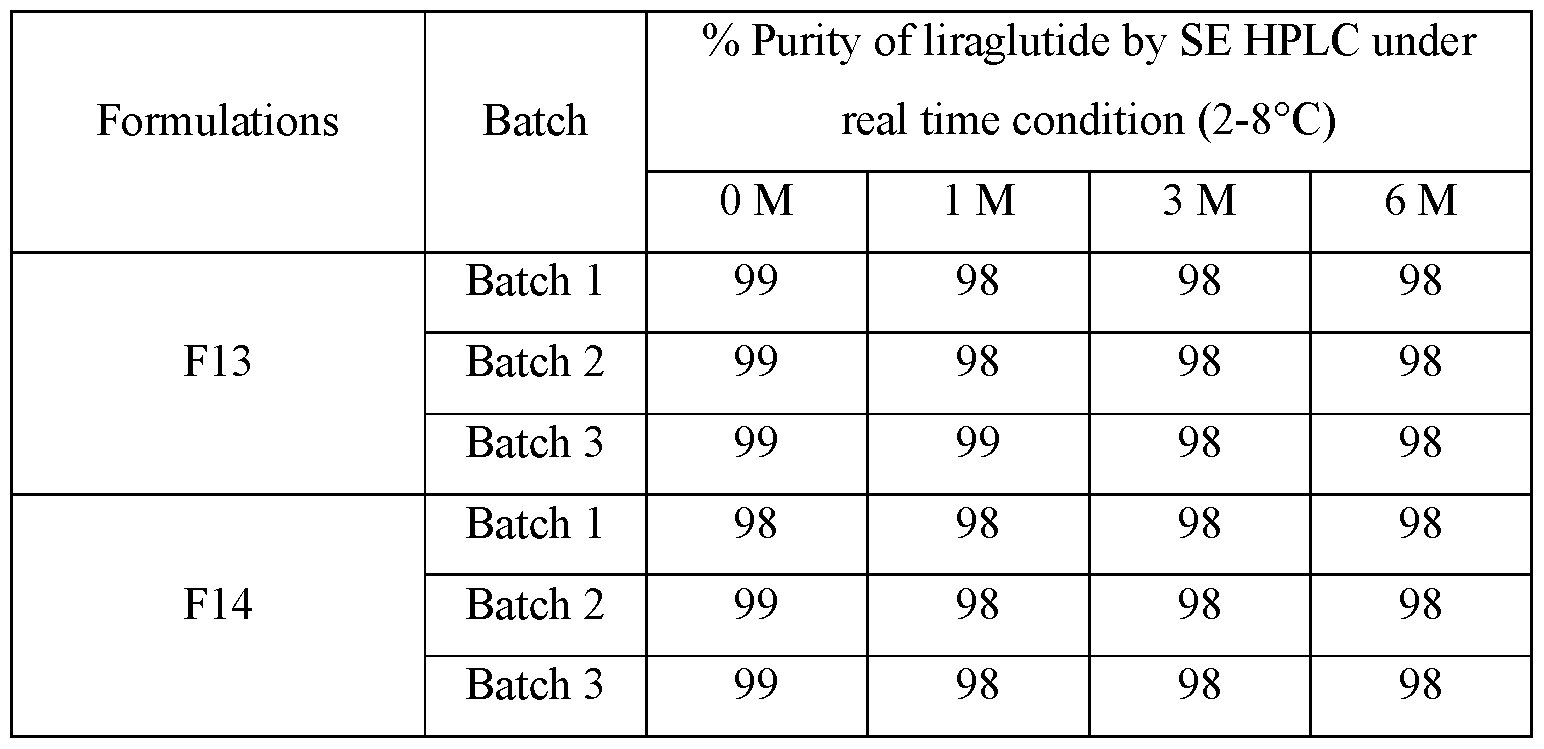
[00133] No significant change in Purity of monomer peak of liraglutide by SE
HPLC was observed up to 6 months at 2-8 °C condition in any formulation.
Observation of purity by SE HPLC of % monomer peas are tabulated below in Table 16 and same was plotted in Figure 6.
Table 3: results of Purity by SE HPLC under real time condition (2-8°C)
[00134] No change in % Purity of monomer peak of liraglutide by RP HPLC was observed up to 6 months at 2-8 °C condition in any formulation. Observation of Purity by RP HPLC % main peak are tabulated below in Table 17, the same was plotted in Figure 7.
Table 4: results of Purity by RP HPLC under real time condition (2-8°C)
[00135] No significant change in liraglutide content was observed up to 6 months at 2-8 °C condition in any formulation. Observations of liraglutide content are tabulated below in Table 18.
Table 5: results of liraglutide content under real time condition (2-8 °C)
II. Accelerated stability study (25°C)
[00136] Accelerated condition stability study of formulations F13 and F14 in comparison with the generic formulation of RMP is carried out using three consistency batches. Data obtained from the accelerated condition so far are discussed here.
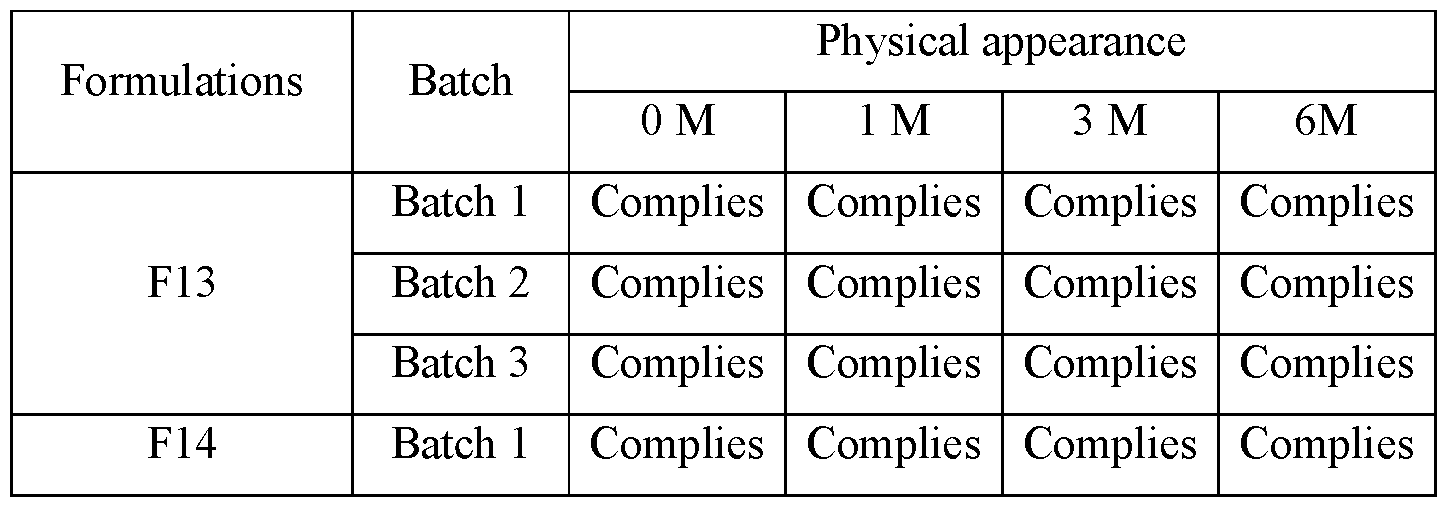
A. Physical Appearance
[00137] No change in physical appearance was observed up to 6 months at 25 °C condition in any formulation. Observation of physical appearance are tabulated below in Table 19.
Table 6: results of Physical appearance under accelerated condition (25°C)
B. pH
[00138] No significant change in pH was observed up to 6 months at 25 °C condition in any formulation. pH is in range of 7.8 to 8.1 for all the formulation. Observations of pH are tabulated below in Table 20.
Table 7: results of pH under accelerated condition (25 °C)
C. Purity by SE HPLC
[00139] No significant change in % Purity of liraglutide in terms of monomer peak by SE HPLC was observed up to 6 months at 25 °C condition in any formulation. Observation of % purity of monomer liraglutide by SE HPLC are tabulated below in Table 21, same were plotted in Figure 8.
Table 8: results of Purity by SE HPLC under accelerated condition (25 °C)
D. Purity by RP HPLC
[00140] No change in Purity of liraglutide in terms of monomer peak by RP HPLC was observed up to 6 months at 25 °C condition in any formulation. Observations of Purity of liraglutide in terms of monomer peak by RP HPLC % are tabulated below in Table 22, the same were plotted in Figure 9.
Table 9: Purity of liraglutide by RP HPLC under accelerated condition (25°C)
E. Liraglutide content
[00141] No significant change in liraglutide content was observed up to 6 months at 25 °C condition in any formulation. Observations of liraglutide content are tabulated below in Table 23.
Table 10: Liraglutide content under accelerated condition (25 °C)
[00142] The present invention provides pharmaceutical composition comprising GLP-1 analogue, its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof and process for preparing the same that can satisfy the existing needs and/or can generally overcome one or more deficiencies found in the existing art.
[00143] The present invention provides liquid pharmaceutical composition comprising GLP-1 analogue, its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof, and process for preparation of the same that is economically viable to produce and/or easily scalable.
[00144] The present invention provides a liquid composition comprising GLP- 1 analogue or its pharmaceutically acceptable salts, complexes, polymorphs, hydrates, solvates, enantiomers or racemates thereof, wherein the liquid composition is stable at various temperatures and process for preparing the said compositions.
[00145] The present invention provides a liquid composition comprising Liraglutide, Semaglutide, D-Liraglutide, D-Semaglutide or other GLP-1 analogue thereof, wherein the liquid composition is stable at various temperatures and process for preparing the said compositions.
Claims
1. A liquid stable pharmaceutical composition comprising:
a) a GLP-1 analogue;
b) a buffer selected from the group consisting of tromethamine (Tris), arginine, sodium phosphate, glycine, sodium phosphate dibasic dehydrate, tromethamine (Tris) acetate, tromethamine (Tris) phosphate, glutamic acid, acetate or combination;
c) an isotonic agent selected from the group consisting of glycerol, sucrose, propylene glycol, sorbitol, PEG 400, trehalose or combination thereof; and
d) a preservative.
2. The pharmaceutical composition of claim 1, wherein the GLP-1 analogue is selected from Liraglutide, D-Liraglutide, Semaglutide, D-Semaglutide or combination thereof.
3. The pharmaceutical composition of claim 1, wherein the preservative is selected from the group consisting of phenol, cresol or combination thereof.
4. The pharmaceutical composition of claim 1, wherein the buffer is in the range from 3 mM to 100 mM, preferably from about 3 mM to about 60 mM.
5. The pharmaceutical composition of claim 1, wherein the isotonic agent is in the concentration ranging from about 0.01% to about 10%, preferably from about 0.02 % to about 8 % w/w of the composition.
6. The pharmaceutical composition of claim 1 for use in the prophylaxis, amelioration and/or treatment of diabetes, in a subject in need thereof.
7. The pharmaceutical composition of claim 1 for parenteral administration to patients in need of such a treatment.
8. A process for preparation of GLP-1 analogue composition wherein said process comprises:
a) dissolving a buffering agent selected from the group consisting of tromethamine (Tris), arginine, sodium phosphate, glycine, sodium
phosphate dibasic dehydrate, tromethamine (Tris) acetate, tromethamine (Tris) phosphate, glutamic acid, acetate or combination; an isotonic agent selected from the group consisting of glycerol, sucrose, propylene glycol, sorbitol, PEG 400, trehalose or combination thereof; and a preservative in water for injection;
b) dissolving a GLP-1 analogue into buffer formulation solution of step (a); and
c) adjusting pH of formulated bulk solution of step (b) with a pH- adjusting agent to a pH range from about 7.0 to about 8.5.
9. The process of claim 8, wherein the GLP-1 analogue is selected from
Liraglutide, D-Liraglutide, Semaglutide, D-Semaglutide or combination thereof.
10. The process of claim 9, wherein the pH-adjusting agent is selected from hydrochloric acid, sodium hydroxide, o-phosphoric acid or glacial acetic acid.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN201921014160 | 2019-04-08 | ||
| IN201921014160 | 2019-04-08 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020208541A1 true WO2020208541A1 (en) | 2020-10-15 |
Family
ID=70476268
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2020/053345 Ceased WO2020208541A1 (en) | 2019-04-08 | 2020-04-08 | Composition comprising glp-1 analogue |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2020208541A1 (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114146163A (en) * | 2021-12-07 | 2022-03-08 | 苏州天马医药集团天吉生物制药有限公司 | Preparation method of semaglutide preparation |
| CN116763763A (en) * | 2022-12-05 | 2023-09-19 | 齐鲁制药有限公司 | Semiglutide inhalation spray and preparation method thereof |
| WO2023238017A1 (en) * | 2022-06-08 | 2023-12-14 | Zydus Lifesciences Limited | Reusable multi-dose, variable dose, single pen injector for type 2 diabetes and weight management |
| RU2855506C1 (en) * | 2020-12-16 | 2026-02-02 | Зилэнд Фарма А/С | Pharmaceutical composition of glp-1/glp-2 dual agonists |
Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2000037098A1 (en) * | 1998-12-22 | 2000-06-29 | Eli Lilly And Company | Shelf-stable formulation of glucagon-like peptide-1 |
| WO2003002136A2 (en) | 2001-06-28 | 2003-01-09 | Novo Nordisk A/S | Stable formulation of modified glp-1 |
| WO2005049061A2 (en) * | 2003-11-20 | 2005-06-02 | Novo Nordisk A/S | Propylene glycol-containing peptide formulations which are optimal for production and for use in injection devices |
| WO2006051110A2 (en) * | 2004-11-12 | 2006-05-18 | Novo Nordisk A/S | Stable formulations of insulinoptropic peptides |
| CN103800301A (en) | 2013-12-27 | 2014-05-21 | 深圳市健元医药科技有限公司 | Pulsatile delivery composition for treating diabetes mellitus and preparation method thereof |
| WO2016038521A1 (en) | 2014-09-08 | 2016-03-17 | Sun Pharmaceutical Industries Limited | Pharmaceutical compositions of liraglutide |
| EP3034090A1 (en) * | 2013-08-13 | 2016-06-22 | Shanghai Benemae Pharmaceutical Corporation | Stable insulin secretagogue peptide hydro-injection pharmaceutical composition |
| US20170326069A1 (en) * | 2009-11-13 | 2017-11-16 | Sanofi-Aventis Deutschland Gmbh | Pharmaceutical Composition Comprising a GLP-1-Agonist and Methionine |
| WO2018096460A1 (en) | 2016-11-22 | 2018-05-31 | Biocon Research Limited | Pharmaceutical compositions of glp-1 analogues |
| WO2018115901A1 (en) * | 2016-12-22 | 2018-06-28 | Arecor Limited | Glucagon-like peptide 1 (glp-1) receptor agonist compositions |
| WO2019038412A1 (en) * | 2017-08-24 | 2019-02-28 | Novo Nordisk A/S | Glp-1 compositions and uses thereof |
-
2020
- 2020-04-08 WO PCT/IB2020/053345 patent/WO2020208541A1/en not_active Ceased
Patent Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2000037098A1 (en) * | 1998-12-22 | 2000-06-29 | Eli Lilly And Company | Shelf-stable formulation of glucagon-like peptide-1 |
| US8846618B2 (en) | 2001-06-28 | 2014-09-30 | Novo Nordisk A/S | Stable formulation of modified GLP-1 |
| WO2003002136A2 (en) | 2001-06-28 | 2003-01-09 | Novo Nordisk A/S | Stable formulation of modified glp-1 |
| WO2005049061A2 (en) * | 2003-11-20 | 2005-06-02 | Novo Nordisk A/S | Propylene glycol-containing peptide formulations which are optimal for production and for use in injection devices |
| US8114833B2 (en) | 2003-11-20 | 2012-02-14 | Novo Nordisk A/S | Propylene glycol-containing peptide formulations which are optimal for production and for use in injection devices |
| WO2006051110A2 (en) * | 2004-11-12 | 2006-05-18 | Novo Nordisk A/S | Stable formulations of insulinoptropic peptides |
| US20170326069A1 (en) * | 2009-11-13 | 2017-11-16 | Sanofi-Aventis Deutschland Gmbh | Pharmaceutical Composition Comprising a GLP-1-Agonist and Methionine |
| EP3034090A1 (en) * | 2013-08-13 | 2016-06-22 | Shanghai Benemae Pharmaceutical Corporation | Stable insulin secretagogue peptide hydro-injection pharmaceutical composition |
| CN103800301A (en) | 2013-12-27 | 2014-05-21 | 深圳市健元医药科技有限公司 | Pulsatile delivery composition for treating diabetes mellitus and preparation method thereof |
| WO2016038521A1 (en) | 2014-09-08 | 2016-03-17 | Sun Pharmaceutical Industries Limited | Pharmaceutical compositions of liraglutide |
| WO2018096460A1 (en) | 2016-11-22 | 2018-05-31 | Biocon Research Limited | Pharmaceutical compositions of glp-1 analogues |
| WO2018115901A1 (en) * | 2016-12-22 | 2018-06-28 | Arecor Limited | Glucagon-like peptide 1 (glp-1) receptor agonist compositions |
| WO2019038412A1 (en) * | 2017-08-24 | 2019-02-28 | Novo Nordisk A/S | Glp-1 compositions and uses thereof |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| RU2855506C1 (en) * | 2020-12-16 | 2026-02-02 | Зилэнд Фарма А/С | Pharmaceutical composition of glp-1/glp-2 dual agonists |
| RU2855486C1 (en) * | 2020-12-16 | 2026-02-02 | Зилэнд Фарма А/С | Pharmaceutical composition of glp-1/glp-2 dual agonists |
| CN114146163A (en) * | 2021-12-07 | 2022-03-08 | 苏州天马医药集团天吉生物制药有限公司 | Preparation method of semaglutide preparation |
| CN114146163B (en) * | 2021-12-07 | 2023-09-26 | 苏州天马医药集团天吉生物制药有限公司 | Preparation method of semaglutin preparation |
| WO2023238017A1 (en) * | 2022-06-08 | 2023-12-14 | Zydus Lifesciences Limited | Reusable multi-dose, variable dose, single pen injector for type 2 diabetes and weight management |
| CN116763763A (en) * | 2022-12-05 | 2023-09-19 | 齐鲁制药有限公司 | Semiglutide inhalation spray and preparation method thereof |
| CN116763763B (en) * | 2022-12-05 | 2024-08-30 | 齐鲁制药有限公司 | A semaglutide inhalation spray and a preparation method thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3536380B1 (en) | Rapid-acting insulin compositions | |
| KR101978527B1 (en) | Stable formulations for parenteral injection of peptide drugs | |
| CA2740685C (en) | Combination of an insulin and a glp-1 agonist | |
| EP4327804B1 (en) | Methods for producing stable therapeutic formulations in aprotic polar solvents | |
| JP6525987B2 (en) | Stable preparation of insulin glulysin | |
| US20180271792A1 (en) | Oral delivery of physiologically active substances | |
| MX2012000304A (en) | Aqueous insulin preparations containing methionine. | |
| JP2012532179A (en) | Slow-acting insulin preparation | |
| WO2020208541A1 (en) | Composition comprising glp-1 analogue | |
| US11771773B2 (en) | Pharmaceutical preparation containing polyethylene gylcol loxenatide and preparation method thereof | |
| AU2024219736A1 (en) | Sublingual epinephrine tablets | |
| US11590205B2 (en) | Methods for producing stable therapeutic glucagon formulations in aprotic polar solvents | |
| US20140249077A1 (en) | Gel compositions | |
| US20170360891A1 (en) | Stable, Benzyl Alcohol-free Aqueous Solution Formulations Containing Alpha-type Interferon | |
| US12343383B2 (en) | High concentration insulin formulation | |
| HK40101667B (en) | Methods for producing stable therapeutic formulations in aprotic polar solvents | |
| HK40101667A (en) | Methods for producing stable therapeutic formulations in aprotic polar solvents | |
| WO2004096266A1 (en) | Improved physical stability of insulin formulations | |
| HK1228824A1 (en) | Rapid-acting insulin compositions | |
| HK1228824B (en) | Rapid-acting insulin compositions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 20722650 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 20722650 Country of ref document: EP Kind code of ref document: A1 |