WO2020115261A1 - Methods and compositions for treating melanoma - Google Patents
Methods and compositions for treating melanoma Download PDFInfo
- Publication number
- WO2020115261A1 WO2020115261A1 PCT/EP2019/083926 EP2019083926W WO2020115261A1 WO 2020115261 A1 WO2020115261 A1 WO 2020115261A1 EP 2019083926 W EP2019083926 W EP 2019083926W WO 2020115261 A1 WO2020115261 A1 WO 2020115261A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- melanoma
- lyn
- resistant
- subject
- treatment
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6876—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes
- C12Q1/6883—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material
- C12Q1/6886—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material for cancer
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/513—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim having oxo groups directly attached to the heterocyclic ring, e.g. cytosine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- G01N33/5751—
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/158—Expression markers
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2333/00—Assays involving biological materials from specific organisms or of a specific nature
- G01N2333/90—Enzymes; Proenzymes
- G01N2333/91—Transferases (2.)
- G01N2333/912—Transferases (2.) transferring phosphorus containing groups, e.g. kinases (2.7)
- G01N2333/91205—Phosphotransferases in general
- G01N2333/9121—Phosphotransferases in general with an alcohol group as acceptor (2.7.1), e.g. general tyrosine, serine or threonine kinases
- G01N2333/91215—Phosphotransferases in general with an alcohol group as acceptor (2.7.1), e.g. general tyrosine, serine or threonine kinases with a definite EC number (2.7.1.-)
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2500/00—Screening for compounds of potential therapeutic value
- G01N2500/10—Screening for compounds of potential therapeutic value involving cells
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2800/00—Detection or diagnosis of diseases
- G01N2800/56—Staging of a disease; Further complications associated with the disease
Definitions
- the invention is in the field of oncology, more particularly the invention relates to methods and compositions for treating melanoma.
- Cutaneous melanoma represents 3% of all cancers, with 8,000 new cases in France and 12,000 new cases in the United Kingdom in 2010. Melanoma has a favorable prognosis when it is diagnosed early, but at the metastatic stage, the prognosis is still very pejorative.
- BRAF inhibitors Zelboraf, Tafmlar
- MEK inhibitor Tafmlar plus Mekinist
- the kinases of the SRC family comprise 8 members and have long been known to be oncogenes [4] Recent reports showed that resistance to BRAF inhibitors can be bypassed by a combination of BRAF inhibitor and SRC inhibitors [5-7] With the aims of deciphering the molecular mechanisms involved in the implementation of resistances and to identify pharmacologically targetable players, the inventors compared the expression of kinases of the SRC family members in melanoma cells isolated from patients before treatment with targeted therapy and after acquisition of resistance to BRAF inhibitors.
- the invention relates to methods for treating melanoma and therapy-resistant melanoma in a subject in need thereof comprising the step of administering said subject with a therapeutically effective amount of an activator of LYN.
- the present invention is defined by the claims.
- inventors have shown that the loss of expression of LYN is correlated with BRAF inhibitors resistance. Secondly, inventors have shown that overexpression of LYN in resistant cells that have lost LYN causes inhibition of their proliferation and migratory capacity.
- the invention relates to a method for predicting whether a subject suffering from a melanoma is or is at risk of having resistant melanoma comprising the steps of i) quantifying the expression level of LYN in a biological sample obtained from the subject; ii) comparing the expression level quantified at step i) with a predetermined reference value and iii) concluding that the subject has or is at risk of suffering from resistant melanoma when the level determined at step i) is lower than the predetermined reference value.
- melanoma also known as malignant melanoma
- malignant melanoma refers to a type of cancer that develops from the pigment-containing cells, called melanocytes.
- melanoma There are three general categories of melanoma: 1) cutaneous melanoma which corresponds to melanoma of the skin; it is the most common type of melanoma; 2) mucosal melanoma which can occur in any mucous membrane of the body, including the nasal passages, the throat, the vagina, the anus, or in the mouth; and 3) ocular melanoma also known as uveal melanoma or choroidal melanoma, is a rare form of melanoma that occurs in the eye.
- the melanoma is cutaneous melanoma.
- the term“subject” denotes a mammal, such as a rodent, a feline, a canine, and a primate.
- the subject according to the invention is a human. More particularly, the subject according to the invention has or is susceptible to have melanoma. In particular embodiment, the subject has or is susceptible to have cutaneous melanoma. In a particular embodiment, the subject has or is susceptible to have metastatic melanoma.
- the subject has or is susceptible to have resistant melanoma.
- resistant melanoma refers to melanoma which does not respond to a treatment.
- the cancer may be resistant at the beginning of treatment or it may become resistant during treatment.
- the resistance to drug leads to rapid progression of metastatic of melanoma.
- resistant melanoma cell refers to cell which does not respond to a treatment.
- the term“sensitive melanoma cell” refers to cell which does respond to a treatment.
- the melanoma is resistant to BRAF inhibitors.
- BRAF is a member of the Raf kinase family of serine/threonine-specific protein kinases. This protein plays a role in regulating the MAP kinase / ERKs signaling pathway, which affects cell division, differentiation, and secretion.
- a number of mutations in BRAF are known. In particular, the V600E mutation is prominent.
- mutations which have been found are R461I, I462S, G463E, G463V, G465A, G465E, G465V, G468A, G468E, N580S, E585K, D593V, F594L, G595R, L596V, T598I, V599D, V599E, V599K, V599R, K600E, A727V, and most of these mutations are clustered to two regions: the glycine-rich P loop of the N lobe and the activation segment and flanking regions.
- the BRAF mutation is V600E.
- the melanoma is resistant to MEK inhibitors.
- MEK refers to Mitogen-activated protein kinase kinase, also known as MAP2K, MEK, MAPKK. It is a kinase enzyme which phosphorylates mitogen-activated protein kinase (MAPK). MEK is activated in melanoma.
- the melanoma is resistant to NRAS inhibitors.
- the NRAS gene is in the Ras family of oncogene and involved in regulating cell division. NRAS mutations in codons 12, 13, and 61 arise in 15-20 % of all melanomas.
- the melanoma is resistant to immune checkpoint inhibitors.
- immune checkpoint inhibitor refers to molecules that totally or partially reduce, inhibit, interfere with or modulate one or more immune checkpoint proteins.
- immuno checkpoint protein has its general meaning in the art and refers to a molecule that is expressed by T cells in that either turn up a signal (stimulatory checkpoint molecules) or turn down a signal (inhibitory checkpoint molecules). Immune checkpoint molecules are recognized in the art to constitute immune checkpoint pathways similar to the CTLA-4 and PD-1 dependent pathways (see e.g. Pardoll, 2012. Nature Rev Cancer 12:252-264; Mellman et al. 2011. Nature 480:480- 489).
- stimulatory checkpoint examples include CD27 CD28 CD40, CD122, CD137, 0X40, GITR, and ICOS.
- inhibitory checkpoint molecules examples include A2AR, B7-H3, B7-H4, BTLA, CTLA-4, CD277, IDO, KIR, PD-1, LAG-3, TIM-3 and VISTA.
- the Adenosine A2A receptor (A2AR) is regarded as an important checkpoint in cancer therapy because adenosine in the immune microenvironment, leading to the activation of the A2a receptor, is negative immune feedback loop and the tumor microenvironment has relatively high concentrations of adenosine.
- B7-H3 also called CD276, was originally understood to be a co-stimulatory molecule but is now regarded as co-inhibitory.
- B7-H4 also called VTCN1
- B and T Lymphocyte Attenuator (BTLA) and also called CD272 has HVEM (Herpesvirus Entry Mediator) as its ligand.
- HVEM Herpesvirus Entry Mediator
- Surface expression of BTLA is gradually downregulated during differentiation of human CD8+ T cells from the naive to effector cell phenotype, however tumor-specific human CD8+ T cells express high levels of BTLA.
- CTLA-4 Cytotoxic T -Lymphocyte- Associated protein 4 and also called CD152.
- IDO Indoleamine 2,3-dioxygenase
- TDO tryptophan catabolic enzyme
- TDO tryptophan 2,3 -dioxygenase
- KIR Killer-cell Immunoglobulin-like Receptor
- LAG3, Lymphocyte Activation Gene-3 works to suppress an immune response by action to Tregs as well as direct effects on CD8+ T cells.
- PD- 1 Programmed Death 1 (PD-1) receptor
- PD-L1 and PD-L2 This checkpoint is the target of Merck & Co.'s melanoma drug Keytruda, which gained FDA approval in September 2014.
- An advantage of targeting PD-1 is that it can restore immune function in the tumor microenvironment.
- TIM-3 short for T-cell Immunoglobulin domain and Mucin domain 3, expresses on activated human CD4+ T cells and regulates Thl and Thl7 cytokines.
- TIM-3 acts as a negative regulator of Thl/Tcl function by triggering cell death upon interaction with its ligand, galectin-9.
- VISTA Short for V-domain Ig suppressor of T cell activation, VISTA is primarily expressed on hematopoietic cells so that consistent expression of VISTA on leukocytes within tumors may allow VISTA blockade to be effective across a broad range of solid tumors. Tumor cells often take advantage of these checkpoints to escape detection by the immune system. Thus, inhibiting a checkpoint protein on the immune system may enhance the anti-tumor T-cell response.
- an immune checkpoint inhibitor refers to any compound inhibiting the function of an immune checkpoint protein. Inhibition includes reduction of function and full blockade.
- the immune checkpoint inhibitor could be an antibody, synthetic or native sequence peptides, small molecules or aptamers which bind to the immune checkpoint proteins and their ligands.
- the immune checkpoint inhibitor is an antibody.
- antibodies are directed against A2AR, B7-H3, B7-H4, BTLA, CTLA-4, CD277, IDO, KIR, PD-1, LAG-3, TIM-3 or VISTA.
- the immune checkpoint inhibitor is an anti-PD-1 antibody such as described in WO2011082400, W02006121168, W02015035606, W02004056875, W02010036959, W02009114335, W02010089411, WO2008156712, WO2011110621, WO2014055648 and WO2014194302.
- anti-PD-1 antibodies which are commercialized: Nivolumab (Opdivo®, BMS), Pembrolizumab (also called Lambrolizumab, KEYTRUDA® or MK-3475, MERCK).
- the immune checkpoint inhibitor is an anti-PD-Ll antibody such as described in WO2013079174, W02010077634, W02004004771, WO2014195852, W02010036959, WO2011066389, W02007005874, W02015048520, US8617546 and WO2014055897.
- anti-PD-Ll antibodies which are on clinical trial: Atezolizumab (MPDL3280A, Genentech/Roche), Durvalumab (AZD9291, AstraZeneca), Avelumab (also known as MSB0010718C, Merck) and BMS-936559 (BMS).
- the immune checkpoint inhibitor is an anti-PD-L2 antibody such as described in US7709214, US7432059 and US8552154.
- the immune checkpoint inhibitor inhibits Tim-3 or its ligand.
- the immune checkpoint inhibitor is an anti-Tim-3 antibody such as described in WO03063792, WO2011155607, WO2015117002, WO2010117057 and W02013006490.
- the immune checkpoint inhibitor is a small organic molecule.
- small organic molecule refers to a molecule of a size comparable to those organic molecules generally used in pharmaceuticals.
- small organic molecules range in size up to about 5000 Da, more preferably up to 2000 Da, and most preferably up to about 1000 Da.
- the small organic molecules interfere with transduction pathway of A2AR, B7-H3, B7-H4, BTLA, CTLA-4, CD277, IDO, KIR, PD-1, LAG-3, TIM-3 or VISTA.
- small organic molecules interfere with transduction pathway of PD-1 and Tim-3.
- they can interfere with molecules, receptors or enzymes involved in PD-1 and Tim-3 pathway.
- the small organic molecules interfere with Indoleamine- pyrrole 2,3-dioxygenase (IDO) inhibitor.
- IDO is involved in the tryptophan catabolism (Liu et al 2010, Vacchelli et al 2014, Zhai et al 2015). Examples of IDO inhibitors are described in WO 2014150677.
- IDO inhibitors include without limitation 1 -methyl-tryptophan (IMT), b- (3-benzofuranyl)-alanine, P-(3-benzo(b)thienyl)-alanine), 6-nitro-tryptophan, 6- fluoro-tryptophan, 4-methyl-tryptophan, 5 -methyl tryptophan, 6-methyl-tryptophan, 5- m ethoxy-tryptophan, 5 -hydroxy-tryptophan, indole 3-carbinol, 3,3'- diindolylmethane, epigallocatechin gallate, 5-Br-4-Cl-indoxyl 1,3-diacetate, 9- vinylcarbazole, acemetacin, 5- bromo-tryptophan, 5-bromoindoxyl diacetate, 3- Amino-naphtoic acid, pyrrolidine dithiocarbamate, 4-phenylimidazole a brassinin derivative, a thioh
- the IDO inhibitor is selected from 1 -methyl-tryptophan, b-(3- benzofuranyl)-alanine, 6-nitro-L-tryptophan, 3- Amino-naphtoic acid and b-[3- benzo(b)thienyl] -alanine or a derivative or prodrug thereof.
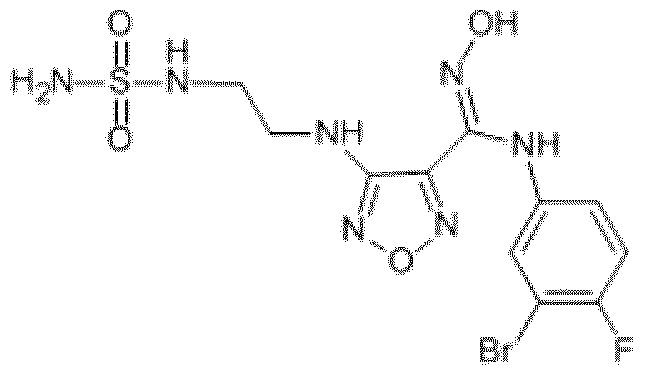
- the inhibitor of IDO is Epacadostat, (INCB24360, INCB024360) has the following chemical formula in the art and refers to -N-(3-bromo-4- fluorophenyl)-N'-hydroxy-4- ⁇ [2-(sulfamoylamino)-ethyl]amino ⁇ -l,2,5-oxadiazole-3 carboximidamide :
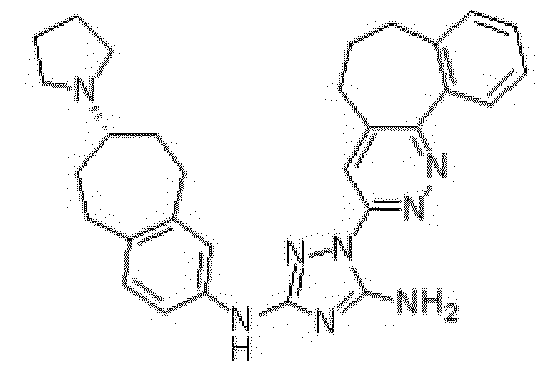
- the inhibitor is BGB324, also called R428, such as described in W02009054864, refers to lH-1, 2, 4-Triazole-3, 5-diamine, l-(6,7-dihydro-5H- benzo[6,7]cyclohepta[l,2-c]pyridazin-3-yl)-N3-[(7S)-6,7,8,9-tetrahydro-7-(l-pyrrolidinyl)- 5H-benzocyclohepten-2-yl]- and has the following formula in the art:
- the inhibitor is CA-170 (or AUPM-170): an oral, small molecule immune checkpoint antagonist targeting programmed death ligand-1 (PD-L1) and V- domain Ig suppressor of T cell activation (VISTA) (Liu et al 2015).
- PD-170 or AUPM-170
- VISTA V- domain Ig suppressor of T cell activation
- the immune checkpoint inhibitor is an aptamer.
- the aptamers are directed against A2AR, B7-H3, B7-H4, BTLA, CTLA-4, CD277, IDO, KIR, PD-1, LAG-3, TIM-3 or VISTA.
- aptamers are DNA aptamers such as described in Prodeus et al 2015.
- a major disadvantage of aptamers as therapeutic entities is their poor pharmacokinetic profiles, as these short DNA strands are rapidly removed from circulation due to renal filtration.
- aptamers according to the invention are conjugated to with high molecular weight polymers such as polyethylene glycol (PEG).
- PEG polyethylene glycol
- the aptamer is an anti-PD-1 aptamer.
- the anti-PD-1 aptamer is MP7 pegylated as described in Prodeus et al 2015.
- the term "predicting the risk” refers to assessing the probability according to which the subject as referred to herein will develop resistant melanoma. As will be understood by those skilled in the art, such an assessment is usually not intended to be correct for 100% of the subjects to be investigated.
- the term“SRC kinase family” refers to family of non-receptor tyrosine kinases implicate in cancer and immune system dysfunction.
- SRC kinase family include the following nine kinases in mammals: Src, Yes, Fyn, and Fgr, forming the SrcA subfamily, Lck, Hck, Blk, and Lyn in the SrcB subfamily, and Frk in its own subfamily. These are non-receptor protein kinases that range in molecular mass from 52 to 62 kD.
- the SRC kinase is LYN.
- LYN refers to Tyrosine-protein kinase Lyn, a protein that is encoded in human by the LYN gene. Lyn is mainly expressed in hematopoietic cells, in neural tissues, liver and adipose tissue.
- the naturally occurring human LYN gene has a nucleotide sequence as shown in Genbank Accession number NM_002350.3 and the naturally occurring human LYN protein has an aminoacid sequence as shown in Genbank Accession numbers NP_002341.1 for LYN isoform A and NP_001104567.1 for LYN isoform B.
- the term“expression level” refers to the expression level of LYN.
- the expression level of the LYN gene may be determined by any technology known by a person skilled in the art.
- each gene expression level may be measured at the genomic and/or nucleic and/or protein level.
- the expression level of gene is determined by measuring the amount of nucleic acid transcripts of each gene.
- the expression level is determined by measuring the amount of each gene corresponding protein. The amount of nucleic acid transcripts can be measured by any technology known by a man skilled in the art.
- the measure may be carried out directly on an extracted messenger RNA (mRNA) sample, or on retrotranscribed complementary DNA (cDNA) prepared from extracted mRNA by technologies well-known in the art.
- mRNA messenger RNA
- cDNA retrotranscribed complementary DNA
- the amount of nucleic acid transcripts may be measured using any technology known by a man skilled in the art, including nucleic microarrays, quantitative PCR, microfluidic cards, and hybridization with a labelled probe.
- the expression level is determined by using quantitative PCR. Quantitative, or real-time, PCR is a well-known and easily available technology for those skilled in the art and does not need a precise description. Methods for determining the quantity of mRNA are well known in the art.
- the nucleic acid contained in the biological sample is first extracted according to standard methods, for example using lytic enzymes or chemical solutions or extracted by nucleic-acid-binding resins following the manufacturer's instructions.
- the extracted mRNA is then detected by hybridization (e. g., Northern blot analysis) and/or amplification (e.g., RT-PCR).
- hybridization e. g., Northern blot analysis
- amplification e.g., RT-PCR
- RT-PCR e.g., RT-PCR
- RT-PCR e.g., RT-PCR
- RT-PCR e.g., RT-PCR
- RT-PCR e.g., RT-PCR
- RT-PCR e.g., RT-PCR
- RT-PCR e.g., RT-PCR
- RT-PCR e.g., RT-PCR
- RT-PCR e.g., RT-PCR
- Nucleic acids having at least 10 nucleotides and exhibiting sequence complementarity or homology to the mRNA of interest herein find utility as hybridization probes or amplification primers. It is understood that such nucleic acids do not need to be identical, but are typically at least about 80% identical to the homologous region of comparable size, more preferably 85% identical and even more preferably 90-95% identical. In certain embodiments, it will be advantageous to use nucleic acids in combination with appropriate means, such as a detectable label, for detecting hybridization. A wide variety of appropriate indicators are known in the art including, fluorescent, radioactive, enzymatic or other ligands (e. g. avidin/biotin).
- Probes typically comprise single-stranded nucleic acids of between 10 to 1000 nucleotides in length, for instance of between 10 and 800, more preferably of between 15 and 700, typically of between 20 and 500.
- Primers typically are shorter single-stranded nucleic acids, of between 10 to 25 nucleotides in length, designed to perfectly or almost perfectly match a nucleic acid of interest, to be amplified.
- the probes and primers are“specific” to the nucleic acids they hybridize to, i.e. they preferably hybridize under high stringency hybridization conditions (corresponding to the highest melting temperature Tm, e.g., 50 % formamide, 5x or 6x SCC.
- SCC is a 0.15 M NaCl, 0.015 M Na-citrate).
- the nucleic acid primers or probes used in the above amplification and detection method may be assembled as a kit.
- a kit includes consensus primers and molecular probes.
- a kit also includes the components necessary to determine if amplification has occurred.
- the kit may also include, for example, PCR buffers and enzymes; positive control sequences, reaction control primers; and instructions for amplifying and detecting the specific sequences.
- the method of the invention comprises the steps of providing total RNAs extracted from a biological sample and subjecting the RNAs to amplification and hybridization to specific probes, more particularly by means of a quantitative or semi-quantitative RT-PCR.
- the expression level is determined by DNA chip analysis.
- Such DNA chip or nucleic acid microarray consists of different nucleic acid probes that are chemically attached to a substrate, which can be a microchip, a glass slide or a microsphere-sized bead.
- a microchip may be constituted of polymers, plastics, resins, polysaccharides, silica or silica-based materials, carbon, metals, inorganic glasses, or nitrocellulose.
- Probes comprise nucleic acids such as cDNAs or oligonucleotides that may be about 10 to about 60 base pairs.
- a biological sample from a test subject optionally first subjected to a reverse transcription, is labelled and contacted with the microarray in hybridization conditions, leading to the formation of complexes between target nucleic acids that are complementary to probe sequences attached to the microarray surface.
- the labelled hybridized complexes are then detected and can be quantified or semi- quantified. Labelling may be achieved by various methods, e.g. by using radioactive or fluorescent labelling.
- Many variants of the microarray hybridization technology are available to the man skilled in the art (see e.g. the review by Hoheisel, Nature Reviews, Genetics, 2006, 7:200-210).
- biological sample refers to any sample obtained from a subject, such as a serum sample, a plasma sample, a urine sample, a blood sample, a lymph sample, or a tissue biopsy.
- biological sample for the determination of an expression level include samples such as a blood sample, a lymph sample, or a biopsy.
- the biological sample is a blood sample, more particularly, circulating tumor cells (CTCs). Detection of CTCs are available to the man skilled in the art (see e.g. the article by Long et al, Cancer Medecine, 2016, 5(6): 1022-1030 [9]).
- the predetermined reference value is a threshold value or a cut-off value.
- a “threshold value” or “cut-off value” can be determined experimentally, empirically, or theoretically.
- a threshold value can also be arbitrarily selected based upon the existing experimental and/or clinical conditions, as would be recognized by a person of ordinary skilled in the art. For example, retrospective measurement of cell densities in properly banked historical subject samples may be used in establishing the predetermined reference value. The threshold value has to be determined in order to obtain the optimal sensitivity and specificity according to the function of the test and the benefit/risk balance (clinical consequences of false positive and false negative).
- the optimal sensitivity and specificity can be determined using a Receiver Operating Characteristic (ROC) curve based on experimental data.
- ROC Receiver Operating Characteristic
- the full name of ROC curve is receiver operator characteristic curve, which is also known as receiver operation characteristic curve. It is mainly used for clinical biochemical diagnostic tests.
- ROC curve is a comprehensive indicator that reflects the continuous variables of true positive rate (sensitivity) and false positive rate (1 -specificity). It reveals the relationship between sensitivity and specificity with the image composition method.
- a series of different cut-off values are set as continuous variables to calculate a series of sensitivity and specificity values. Then sensitivity is used as the vertical coordinate and specificity is used as the horizontal coordinate to draw a curve. The higher the area under the curve (AUC), the higher the accuracy of diagnosis.
- AUC area under the curve
- the point closest to the far upper left of the coordinate diagram is a critical point having both high sensitivity and high specificity values.
- the AUC value of the ROC curve is between 1.0 and 0.5. When AUC>0.5, the diagnostic result gets better and better as AUC approaches 1. When AUC is between 0.5 and 0.7, the accuracy is low. When AUC is between 0.7 and 0.9, the accuracy is moderate.
- This algorithmic method is preferably done with a computer.
- Existing software or systems in the art may be used for the drawing of the ROC curve, such as: MedCalc 9.2.0.1 medical statistical software, SPSS 9.0, ROCPOWER.SAS, DESIGNROC.FOR, MULTIREADER POWER S AS, CREATE-ROC.SAS, GB STAT VIO.O (Dynamic Microsystems, Inc. Silver Spring, Md., USA), etc.
- the predetermined reference value is determined by carrying out a method comprising the steps of
- step c) classifying said tumor tissue samples in two groups for one specific arbitrary quantification value provided at step c), respectively: (i) a first group comprising tumor tissue samples that exhibit a quantification value for level that is lower than the said arbitrary quantification value contained in the said serial of quantification values; (ii) a second group comprising tumor tissue samples that exhibit a quantification value for said level that is higher than the said arbitrary quantification value contained in the said serial of quantification values; whereby two groups of tumor tissue samples are obtained for the said specific quantification value, wherein the tumor tissue samples of each group are separately enumerated;
- step g) reiterating steps f) and g) until every arbitrary quantification value provided at step d) is tested; h) setting the said predetermined reference value as consisting of the arbitrary quantification value for which the highest statistical significance (most significant P-value obtained with a log-rank test, significance when P ⁇ 0.05) has been calculated at step g).
- the cell density has been assessed for 100 tumor tissue samples of 100 subjects.
- the 100 samples are ranked according to the cell density.
- Sample 1 has the highest density and sample 100 has the lowest density.
- a first grouping provides two subsets: on one side sample Nr 1 and on the other side the 99 other samples.
- the next grouping provides on one side samples 1 and 2 and on the other side the 98 remaining samples etc., until the last grouping: on one side samples 1 to 99 and on the other side sample Nr 100.
- Kaplan-Meier curves are prepared for each of the 99 groups of two subsets. Also for each of the 99 groups, the p value between both subsets was calculated (log-rank test).
- the predetermined reference value is then selected such as the discrimination based on the criterion of the minimum P-value is the strongest.
- the cell density corresponding to the boundary between both subsets for which the P-value is minimum is considered as the predetermined reference value.
- the predetermined reference value is not necessarily the median value of cell densities.
- the predetermined reference value thus allows discrimination between a poor and a good prognosis with respect to DFS and OS for a subject. Practically, high statistical significance values (e.g. low P values) are generally obtained for a range of successive arbitrary quantification values, and not only for a single arbitrary quantification value.
- a range of values is provided instead of using a definite predetermined reference value. Therefore, a minimal statistical significance value (minimal threshold of significance, e.g. maximal threshold P value) is arbitrarily set and a range of a plurality of arbitrary quantification values for which the statistical significance value calculated at step g) is higher (more significant, e.g. lower P-value) are retained, so that a range of quantification values is provided.
- This range of quantification values includes a "cut-off value as described above. For example, according to this specific embodiment of a "cut-off value, the outcome can be determined by comparing the cell density with the range of values which are identified.
- a cut-off value thus consists of a range of quantification values, e.g. centered on the quantification value for which the highest statistical significance value is found (e.g. generally the minimum P-value which is found).
- the invention relates to a method for treating melanoma in a subject in need thereof comprising a step of administering said subject with a therapeutically effective amount of an activator of LYN.
- the subject is identified as having melanoma resistance by performing the method as described above.
- the invention relates to a method for treating resistant melanoma in a subject in need thereof comprising a step of administering said subject with a therapeutically effective amount of an activator of LYN.
- the subject is identified as having melanoma resistant to a classical treatment.
- the subject is identified as having BRAF inhibitors resistance.
- the subject is identified as having MEK inhibitors resistance.
- the subject is identified as having NRAS inhibitors resistance.
- the subject is identified as having immune checkpoint inhibitors resistance.
- the activators of LYN may be combined with an immune checkpoint inhibitor.
- the present invention relates to a method for treating melanoma and/or resistant melanoma in a subject in need thereof comprising a step of administering said subject with a therapeutically effective amount of an activator of LYN and of an immune checkpoint inhibitor.
- the terms“treating” or“treatment” refer to both prophylactic or preventive treatment as well as curative or disease modifying treatment, including treatment of subject at risk of contracting the disease or suspected to have contracted the disease as well as subject who are ill or have been diagnosed as suffering from a disease or medical condition, and includes suppression of clinical relapse.
- the treatment may be administered to a subject having a medical disorder or who ultimately may acquire the disorder, in order to prevent, cure, delay the onset of, reduce the severity of, or ameliorate one or more symptoms of a disorder or recurring disorder, or in order to prolong the survival of a subject beyond that expected in the absence of such treatment.
- therapeutic regimen is meant the pattern of treatment of an illness, e.g., the pattern of dosing used during therapy.
- a therapeutic regimen may include an induction regimen and a maintenance regimen.
- the phrase “induction regimen” or “induction period” refers to a therapeutic regimen (or the portion of a therapeutic regimen) that is used for the initial treatment of a disease.
- the general goal of an induction regimen is to provide a high level of drug to a subject during the initial period of a treatment regimen.
- An induction regimen may employ (in part or in whole) a "loading regimen", which may include administering a greater dose of the drug than a physician would employ during a maintenance regimen, administering a drug more frequently than a physician would administer the drug during a maintenance regimen, or both.
- maintenance regimen refers to a therapeutic regimen (or the portion of a therapeutic regimen) that is used for the maintenance of a subject during treatment of an illness, e.g., to keep the subject in remission for long periods of time (months or years).
- a maintenance regimen may employ continuous therapy (e.g., administering a drug at a regular intervals, e.g., weekly, monthly, yearly, etc.) or intermittent therapy (e.g., interrupted treatment, intermittent treatment, treatment at relapse, or treatment upon achievement of a particular predetermined criteria [e.g., pain, disease manifestation, etc.]).
- the term“activator of LYN” refers to a natural or synthetic compound that has a biological effect to increase the activity of LYN. More particularly, such compound induces phosphorylation of Tyr396 in the activation loop of LYN that generates a highly active LYNand/or increases the kinase activity of LYN toward its natural (Cortactin, HS1) or synthetic substrates (Poly (Glu4-Tyr) or SRCtide (GAELPPEF A AQLRKIGDK V Y C (SEQ ID NO: 1))
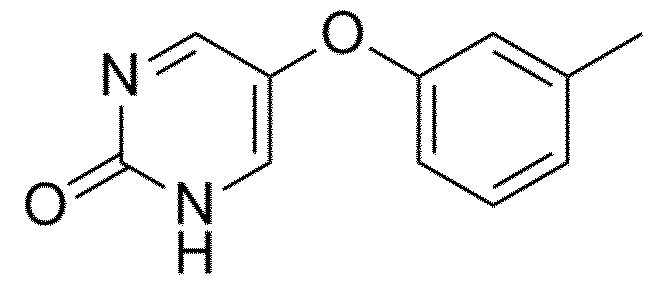
- the activator of LYN is a LYN allosteric activator. In a particular embodiment, the activator of LYN is tolimidone.
- tolimidone also known as MLR-1023 or 2(1H)- pyrimidinone,5-(3-methylphenoxy) has the formula Cl 1H10N2O2 and the following structure in the art:
- the activator of LYN is a derivative of MLR-1023 as describes in W02010/003055.
- the activator of LYN is a peptide, petptidomimetic, small organic molecule, antibody, aptamers, siRNA or antisense oligonucleotide.
- peptido mimetic refers to a small protein-like chain designed to mimic a peptide.
- the activator of LYN is an aptamer.
- Aptamers are a class of molecule that represents an alternative to antibodies in term of molecular recognition. Aptamers are oligonucleotide or oligopeptide sequences with the capacity to recognize virtually any class of target molecules with high affinity and specificity.
- the activator of LYN is a small organic molecule.
- small organic molecule refers to a molecule of a size comparable to those organic molecules generally used in pharmaceuticals. The term excludes biological macromolecules (e.g., proteins, nucleic acids, etc.). Preferred small organic molecules range in size up to about 5000 Da, more preferably up to 2000 Da, and most preferably up to about 1000 Da.
- the activator of LYN is an antibody.
- antibody is used in the broadest sense and specifically covers monoclonal antibodies, polyclonal antibodies, multispecific antibodies (e.g. bispecific antibodies) formed from at least two intact antibodies, and antibody fragments so long as they exhibit the desired biological activity.
- the term includes antibody fragments that comprise an antigen binding domain such as Fab', Fab, F(ab')2, single domain antibodies (DABs), TandAbs dimer, Fv, scFv (single chain Fv), dsFv, ds-scFv, Fd, linear antibodies, minibodies, diabodies, bispecific antibody fragments, bibody, tribody (scFv-Fab fusions, bispecific or trispecific, respectively); sc-diabody; kappa(lamda) bodies (scFv-CL fusions); BiTE (Bispecific T-cell Engager, scFv-scFv tandems to attract T cells); DVD-Ig (dual variable domain antibody, bispecific format); SIP (small immunoprotein, a kind of minibody); SMIP ("small modular immunopharmaceutical” scFv-Fc dimer; DART (ds-stabilized diabody "Dual Affinity ReTargeting"
- Antibodies can be fragmented using conventional techniques. For example, F(ab')2 fragments can be generated by treating the antibody with pepsin. The resulting F(ab')2 fragment can be treated to reduce disulfide bridges to produce Fab' fragments. Papain digestion can lead to the formation of Fab fragments.
- Fab, Fab' and F(ab')2, scFv, Fv, dsFv, Fd, dAbs, TandAbs, ds-scFv, dimers, minibodies, diabodies, bispecific antibody fragments and other fragments can also be synthesized by recombinant techniques or can be chemically synthesized. Techniques for producing antibody fragments are well known and described in the art. For example, each of Beckman et al, 2006; Holliger & Hudson, 2005; Le Gall et al, 2004; Reff & Heard, 2001 ; Reiter et al., 1996; and Young et al., 1995 further describe and enable the production of effective antibody fragments.
- the antibody is a“chimeric” antibody as described in U.S. Pat. No. 4,816,567.
- the antibody is a humanized antibody, such as described U.S. Pat. Nos. 6,982,321 and 7,087,409.
- the antibody is a human antibody.
- A“human antibody” such as described in US 6,075, 181 and 6, 150,584.
- the antibody is a single domain antibody such as described in EP 0 368 684, WO 06/030220 and WO 06/003388.
- the activator of LYN is a monoclonal antibody.
- Monoclonal antibodies can be prepared and isolated using any technique that provides for the production of antibody molecules by continuous cell lines in culture. Techniques for production and isolation include but are not limited to the hybridoma technique, the human B-cell hybridoma technique and the EBV-hybridoma technique.
- the activator is an intrabody having specificity for LYN.
- the term "intrabody” generally refer to an intracellular antibody or antibody fragment.
- Antibodies in particular single chain variable antibody fragments (scFv), can be modified for intracellular localization. Such modification may entail for example, the fusion to a stable intracellular protein, such as, e.g., maltose binding protein, or the addition of intracellular trafficking/localization peptide sequences, such as, e.g., the endoplasmic reticulum retention.
- the intrabody is a single domain antibody.
- the antibody according to the invention is a single domain antibody.
- sdAb single domain antibody
- VHH single domain antibody
- sdAb single domain antibody
- VHH single domain antibody
- LYN activator In order to test the functionality of a putative LYN activator a test is necessary. For that purpose, to identify LYN activator an in vitro kinase assay will be used. In brief, recombinant LYN kinase is incubated with a synthetic substrate (SRCtides), ATP as phosphate donor, and increasing concentration of the potential activators. MLR- 1023 is used as reference agent, and Staurosporine, (a known nonspecific LYN inhibitor) as inhibition control.
- SRCtides synthetic substrate
- MLR- 1023 is used as reference agent
- Staurosporine (a known nonspecific LYN inhibitor) as inhibition control.
- melanoma treatment with pharmacological LYN activator can overcome resistance to drugs targeting oncogenic BRAF.
- the resistance of cancer for the medication is caused by mutations in the genes which are involved in the proliferation, divisions or differentiation of cells or by phenotypic switch with a transcriptional profile favoring the resistance to treatment.
- the resistance of melanoma is caused by the mutations (single or double) in the following genes: BRAF, MEK or NRAS.
- the resistance can be also caused by a double-negative BRAF and NRAS mutation or phenotypic switch.
- the melanoma is resistant to a treatment with the inhibitors of BRAF mutations.
- the melanoma is resistant to a treatment.
- the melanoma is resistant to a treatment with the inhibitors of BRAF.
- the melanoma is resistant to a treatment with dabrafenib also known as tafmlar which is commercialized by Novartis.
- the melanoma is resistant to a treatment with vemurafenib.
- Vemurafenib also known as PLX4032, RG7204 ou R05185426 and commercialized by Roche as Zelboraf.
- the melanoma is resistant to a treatment with dacarbazine.
- dacarbazine also known as imidazole carboxamide is commercialized as DTIC-Dome by Bayer.
- the melanoma is resistant to a treatment with the inhibitors of MEK.
- the inhibitors of MEK are well known in the art.
- the melanoma is resistant to a treatment with trametinib also known as mekinist which is commercialized by GSK.
- the melanoma is resistant to a treatment with cobimetinib also known as cotellic commercialized by Genentech.
- the melanoma is resistant to a treatment with Binimetinib also knowns as MEK162, ARRY-162 is developed by Array Biopharma.
- the melanoma is resistant to a treatment with the inhibitors of NRAS. In a particular embodiment, the melanoma is resistant to a treatment with salirasib commercialized by Concordia Pharmaceuticals.
- the inhibitors of BRAF mutation or MEK are used to treat the melanoma with NRAS mutations. In a particular embodiment, the melanoma is resistant in which double-negative BRAF and NRAS mutant melanoma.
- the melanoma is resistant to a combined treatment.
- the terms“combined treatment”,“combined therapy” or“therapy combination” refer to a treatment that uses more than one medication.
- the combined therapy may be dual therapy or bi-therapy.
- the melanoma is resistant to a combined treatment characterized by using an inhibitor of BRAF mutation and an inhibitor of MEK or an inhibitor of BRAF mutation and an inhibitor of NRAS as described above.
- the combined treatment may be a combination of Vemurafenib (BRAFi) and Cobimetinib (MEKi), or a combination of Dabrafenib (BRAFi) and Trametinib (MEKi), or a combination of Cobimetinib Encorafenib (BRAFi) and Binimetinib(MEKi).
- the melanoma is resistant to a treatment with an immune checkpoint inhibitor.
- immune checkpoint inhibitor as used herein, is defined above.
- the melanoma is also resistant to a combined treatment characterized by using an inhibitor of BRAF mutation and an inhibitor of immune checkpoint.
- the melanoma is also resistant to a combined treatment characterized by using an inhibitor of BRAF mutation, an inhibitor of MEK and an inhibitor of immune checkpoint.
- the subject has or susceptible to have melanoma resistant to at least one of the treatments as described above.
- the subject having a melanoma resistant is identified by standard criteria.
- the standard criteria for resistance for example, are Response Evaluation Criteria In Solid Tumors (RECIST) criteria, published by an international consortium including NCI.
- administering refers to the act of injecting or otherwise physically delivering a substance as it exists outside the body (e.g., an activator of LYN) into the subject, such as by mucosal, intradermal, intravenous, subcutaneous, intramuscular delivery and/or any other method of physical delivery described herein or known in the art.
- a disease, or a symptom thereof is being treated, administration of the substance typically occurs after the onset of the disease or symptoms thereof.
- administration of the substance typically occurs before the onset of the disease or symptoms thereof.
- a “therapeutically effective amount” is meant a sufficient amount of activator of LYN for use in a method for the treatment of melanoma at a reasonable benefit/risk ratio applicable to any medical treatment. It will be understood that the total daily usage of the compounds and compositions of the present invention will be decided by the attending physician within the scope of sound medical judgment.
- the specific therapeutically effective dose level for any particular subject will depend upon a variety of factors including the age, body weight, general health, sex and diet of the subject; the time of administration, route of administration, and rate of excretion of the specific compound employed; the duration of the treatment; drugs used in combination or coincidental with the specific polypeptide employed; and like factors well known in the medical arts.
- the daily dosage of the products may be varied over a wide range from 0.01 to 1,000 mg per adult per day.
- the compositions contain 0.01, 0.05, 0.1, 0.5, 1.0, 2.5, 5.0, 10.0, 15.0, 25.0, 50.0, 100, 250 and 500 mg of the active ingredient for the symptomatic adjustment of the dosage to the subject to be treated.
- a medicament typically contains from about 0.01 mg to about 500 mg of the active ingredient, typically from 1 mg to about 100 mg of the active ingredient.
- An effective amount of the drug is ordinarily supplied at a dosage level from 0.0002 mg/kg to about 20 mg/kg of body weight per day, especially from about 0.001 mg/kg to 7 mg/kg of body weight per day.
- the activators of LYN as described above may be combined with pharmaceutically acceptable excipients, and optionally sustained-release matrices, such as biodegradable polymers, to form pharmaceutical compositions.
- pharmaceutically acceptable excipients such as a carboxylate, ethylene glycol, ethylene glycol, ethylene glycol, ethylene glycol, ethylene glycol, ethylene glycol, ethylene glycol, ethylene glycol, ethylene glycol dimethoxysulfate, ethylene glycol, glycerol, glycerol, glycerol, sorbitol, sorbitol, arate, arate, arate, arate, arate, arate, arate, arate, arate, arate, arate, arate, ethylene glycol, ethylene glycol, ethylene glycol, ethylene glycol, ethylene glycol, ethylene glycol, ethylene glycol, glycerol, ethylene glycol, glycerol,
- compositions of the present invention for oral, sublingual, subcutaneous, intramuscular, intravenous, transdermal, local or rectal administration can be administered in a unit administration form, as a mixture with conventional pharmaceutical supports, to animals and human beings.
- Suitable unit administration forms comprise oral-route forms such as tablets, gel capsules, powders, granules and oral suspensions or solutions, sublingual and buccal administration forms, aerosols, implants, subcutaneous, transdermal, topical, intraperitoneal, intramuscular, intravenous, subdermal, transdermal, intrathecal and intranasal administration forms and rectal administration forms.
- the pharmaceutical compositions contain vehicles which are pharmaceutically acceptable for a formulation capable of being injected.
- vehicles which are pharmaceutically acceptable for a formulation capable of being injected.
- These may be in particular isotonic, sterile, saline solutions (monosodium or disodium phosphate, sodium, potassium, calcium or magnesium chloride and the like or mixtures of such salts), or dry, especially freeze-dried compositions which upon addition, depending on the case, of sterilized water or physiological saline, permit the constitution of injectable solutions.
- the pharmaceutical forms suitable for injectable use include sterile aqueous solutions or dispersions; formulations including sesame oil, peanut oil or aqueous propylene glycol; and sterile powders for the extemporaneous preparation of sterile injectable solutions or dispersions.
- the form In all cases, the form must be sterile and must be fluid to the extent that easy syringability exists. It must be stable under the conditions of manufacture and storage and must be preserved against the contaminating action of microorganisms, such as bacteria and fungi.
- Solutions comprising compounds of the invention as free base or pharmacologically acceptable salts can be prepared in water suitably mixed with a surfactant, such as hydroxypropylcellulose. Dispersions can also be prepared in glycerol, liquid polyethylene glycols, and mixtures thereof and in oils. Under ordinary conditions of storage and use, these preparations contain a preservative to prevent the growth of microorganisms.
- the polypeptide (or nucleic acid encoding thereof) can be formulated into a composition in a neutral or salt form.
- Pharmaceutically acceptable salts include the acid addition salts (formed with the free amino groups of the protein) and which are formed with inorganic acids such as, for example, hydrochloric or phosphoric acids, or such organic acids as acetic, oxalic, tartaric, mandelic, and the like. Salts formed with the free carboxyl groups can also be derived from inorganic bases such as, for example, sodium, potassium, ammonium, calcium, or ferric hydroxides, and such organic bases as isopropylamine, trimethylamine, histidine, procaine and the like.
- the carrier can also be a solvent or dispersion medium containing, for example, water, ethanol, polyol (for example, glycerol, propylene glycol, and liquid polyethylene glycol, and the like), suitable mixtures thereof, and vegetables oils.
- the proper fluidity can be maintained, for example, by the use of a coating, such as lecithin, by the maintenance of the required particle size in the case of dispersion and by the use of surfactants.
- the prevention of the action of microorganisms can be brought about by various antibacterial and antifungal agents, for example, parabens, chlorobutanol, phenol, sorbic acid, thimerosal, and the like.
- isotonic agents for example, sugars or sodium chloride.
- Prolonged absorption of the injectable compositions can be brought about by the use in the compositions of agents delaying absorption, for example, aluminium monostearate and gelatin.
- Sterile injectable solutions are prepared by incorporating the active polypeptides in the required amount in the appropriate solvent with several of the other ingredients enumerated above, as required, followed by filtered sterilization.
- dispersions are prepared by incorporating the various sterilized active ingredients into a sterile vehicle which contains the basic dispersion medium and the required other ingredients from those enumerated above.
- sterile powders for the preparation of sterile injectable solutions
- the preferred methods of preparation are vacuum drying and freeze-drying techniques which yield a powder of the active ingredient plus any additional desired ingredient from a previously sterile-filtered solution thereof.
- solutions will be administered in a manner compatible with the dosage formulation and in such amount as is therapeutically effective.
- the formulations are easily administered in a variety of dosage forms, such as the type of injectable solutions described above, but drug release capsules and the like can also be employed.
- the solution should be suitably buffered if necessary and the liquid diluent first rendered isotonic with sufficient saline or glucose.
- aqueous solutions are especially suitable for intravenous, intramuscular, subcutaneous and intrap eritoneal administration.
- sterile aqueous media which can be employed will be known to those of skill in the art in light of the present disclosure.
- one dosage could be dissolved in 1 ml of isotonic NaCl solution and either added to 1000 ml of hypodermoclysis fluid or injected at the proposed site of infusion. Some variation in dosage will necessarily occur depending on the condition of the subject being treated. The person responsible for administration will, in any event, determine the appropriate dose for the individual subject.
- a further object of the present invention relates to a method of screening a drug suitable for the treatment of melanoma comprising i) providing a test compound and ii) determining the ability of said test compound to activate the activity of LYN.
- Any biological assay well known in the art could be suitable for determining the ability of the test compound to activate the activity of LYN. Such assay is briefly described above.
- control substance refers a molecule that is inert or has no activity relating to an ability to modulate a biological activity or expression. It is to be understood that test compounds capable of activating the activity of LYN, as determined using in vitro methods described herein, are likely to exhibit similar modulatory capacity in applications in vivo.
- the test compound is selected from the group consisting of peptides, petptidomimetics, small organic molecules (such as MLR- 1023), aptamers or nucleic acids.
- the test compound according to the invention may be selected from a library of compounds previously synthesised, or a library of compounds for which the structure is determined in a database, or from a library of compounds that have been synthesised de novo.
- the test compound may be selected form small organic molecules.
- FIGURES are a diagrammatic representation of FIGURES.
- Figure 1 LYN expression in BRAFi sensitive and resistant melanoma cells.
- Figure 2 Patients with melanoma expressing high level of LYN display a favorable prognosis. Kaplan-Meier analysis of the TCGA cohort. Patients with a low LYN expression in the tumors have a statistically significant reduced overall survival time (p ⁇ 0.0001, log rank test (Mantel-Cox).
- FIG. 4 LYN allosteric activator, MLR1023 inhibits the invasive properties of melanoma cells. Quantification of the effect of MLR1023 on migration of different melanoma cells. Note that the cells that do not express LYN (patient#2R and patient#5) were not responsive to MLR1023.
- the A375 S (Sensitive), A375 R (Resistant), WM9 S (Susceptible) and WM9 R (Resistant) human cell lines are grown under 5% C02 at 37 ° C in DMEM (Dulbecco Minimum Eagle Essential Medium) medium), Glutamax (Gibco) + 10% FCS + 50U / ml Penicillin + 50pg / ml Streptomycin.
- the cells of patients are isolated from biopsies after digestion with DNase, Dispase and Collagenase and cultured under 5% C02 at 37 ° C in RPMI medium (glucose, GlutaMAX, pyruvate, Invitrogen) 1640 Glutamax + 1% FCS + 50U / ml Penicillin + 50 pg / ml streptomycin.
- Western Blot Cells exposed to the different experimental conditions are lysed for 20 minutes at 4 ° C in Fisher buffer (50mM Hepes pH 7.4, 150mM NaCl, ImM Orthovanadate, 5mM NaF, 20mM NaPPi, Complete 7X cocktail and 1% TritonX-100).
- the lysates are then centrifuged (13000 rpm for 10 min at 4 ° C.) and the protein concentration is determined using micro BCA reagents (Thermo scientific), according to the supplier's recommendations by spectrophotometry at 561 nm.
- the membrane is saturated (lOmM Tris-HCl pH 7.4, 150mM NaCl, ImM EDTA, 0.1% Tween-20, 3% BSA and 0.5% gelatin) for lh at room temperature and incubated 18h at 4 ° C, in the presence of the primary antibody directed against the protein of interest in the solution of saturation Then, after 3 washes of 10 min in washing buffer (10 mM Tris-HCl pH 7.6, 150 mM NaCl, 1% NP40), the membrane is incubated with a secondary antibody coupled to peroxidase (in saturation solution) for lh at room temperature. The membrane is washed again three times for 10 min and then the proteins of interest are revealed by chemoluminescence using an ECL reagent (Amersham Biosciences).
- the cells are seeded at a rate of 150,000 to 200,000 per ml in 60 mm diameter culture dishes and then transfected 24h later with a siRNA targeting the members of the SRC family and siRNA control (Flexitube, Qiagen) at a final concentration of 50nM with the HiPerfect transfection reagent (Qiagen) for 48 or 72h.
- Control or LYN encoding adenoviruses were from Vector Biolabs. We seed the melanoma cells in 6-well culture plates (400,000 cells per well in 2 ml of RPMI 7% FCS medium) and then infect them the next day with the adenoviral particles (1 and 3 pi / well of particles at 4.9 * 1010 PFU / ml). After 48 hours of infection and three washes with PBS, the cells are taken up in fresh medium, and used either for gene/protein expression, or migration assays
- Transwell® inserts consists of an upper chamber and a lower chamber, separated by a porous membrane (8m) (Coming).
- the upper chamber is inoculated with a suspension of 300,000 cells in 200 m ⁇ of RPMI medium without FCS and treated according to each condition.
- the lower chamber contains only 600pL of RMPI medium 7% FCS.
- the cells migrate for 24 hours according to chemotaxis from the upper chamber to the lower chamber through the porous membrane.
- the Transwell® inserts are then deposited in a 4% PFA solution for 10 min at room temperature in order to fix the cells having migrated. These are then stained with crystal violet at 0.2% for lOmin. After rinsing with PBS, drying and cleaning the upper part of the Transwell to remove cells that have not migrated, the cells are visualized under a microscope and counted using a software IMAGE J.
- RNAs are extracted using the RNeasy kit (Qiagen).
- the cell lysates are deposited on micro-columns, washed by several short centrifugation steps and eluted in 30 pi of H20 RNase.
- a spectrophotometer Nanodrop
- 1 pg of RNA is retro-transcribed into cDNA with oligo dT as primer, according to the supplier's protocol (Reverse Transcription System, Promega).
- a 1 / 50th dilution of cDNA is incubated with 100pL of SYBR Green PCR master reagent mix (Applied biosystems) and the primers specific to the genes of interest diluted to 1/20.
- the PCR reaction is analyzed in real time using a StepOnePlus real time PCR system thermocycler (Applied Biosystems).
- the relative quantization of the amplicons was performed by the 2 (- AACT) method. Detection of the housekeeping GAPDH gene is used to normalize the results.
- the primer sequences used for each cDNA are analyzed in real time using a StepOnePlus real time PCR system thermocycler (Applied Biosystems).
- the relative quantization of the amplicons was performed by the 2 (- AACT) method. Detection of the housekeeping GAPDH gene is used to normalize the results.
- the primer sequences used for each cDNA is analyzed in real time using a StepOnePlus real time PCR system thermocycler (Applied Biosystems).
- LYN expression is decreased in BRAFi-resistant melanoma cells.
- LYN impedes the metastatic properties of melanoma cells.
- LYN it is clearly involved in the control of metastatic properties of melanoma cells. According to our data, LYN might be a valuable target to prevent or impair melanoma spreading. However, metastasis inhibition would be achieved through an increase in LYN expression or an activation of LYN activity.
- MLR1023 a LYN kinase activator, MLR1023 is currently in phase 2b clinical trial for type 2 diabetes indication [8] Therefore, this drug, if efficient in preclinical model setting can rapidly be repositioned in the treatment of melanomas.
- LYN acted negatively on the metastatic properties of melanoma cells.
- the Lyn kinase activator MLR-1023 is a novel insulin receptor potentiator that elicits a rapid-onset and durable improvement in glucose homeostasis in animal models of type 2 diabetes.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Wood Science & Technology (AREA)
- Engineering & Computer Science (AREA)
- Pathology (AREA)
- Genetics & Genomics (AREA)
- Analytical Chemistry (AREA)
- Immunology (AREA)
- Zoology (AREA)
- Epidemiology (AREA)
- Biotechnology (AREA)
- Biochemistry (AREA)
- Microbiology (AREA)
- Molecular Biology (AREA)
- Physics & Mathematics (AREA)
- Oncology (AREA)
- Hospice & Palliative Care (AREA)
- Biophysics (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The present invention relates to method and composition for treating melanoma. The inventors have shown that a decrease in LYN expression, in melanoma cells and tumors is related to the acquisition of a resistance to targeted therapies. The have also shown the inhibition of metastatic propensity of melanoma cell by an allosteric activator of LYN. In particular, the present invention relates to a method for predicting whether a subject suffering from a melanoma is or is at risk of having resistant melanoma. The present invention also relates to methods for treating melanoma and resistant melanoma in a subject in need thereof comprising a step of administering said subject with a therapeutically effective amount of an activator of LYN.
Description
METHODS AND COMPOSITIONS FOR TREATING MELANOMA
FIELD OF THE INVENTION:
The invention is in the field of oncology, more particularly the invention relates to methods and compositions for treating melanoma.
BACKGROUND OF THE INVENTION:
Cutaneous melanoma represents 3% of all cancers, with 8,000 new cases in France and 12,000 new cases in the United Kingdom in 2010. Melanoma has a favorable prognosis when it is diagnosed early, but at the metastatic stage, the prognosis is still very pejorative. Recently, targeted therapies using BRAF inhibitors (Zelboraf, Tafmlar) alone or in combination with a MEK inhibitor (Tafmlar plus Mekinist) have shown dramatic results in terms of response rates. However, the initial enthusiasm was tempered by the almost systematic observation of recurrences, due to the acquisition of secondary resistances. Immunotherapeutic approaches targeting the negative control points of the immune response (CTLA4 and PD1) have also shown spectacular results, especially anti-PDl (Nivolumab, Pembrolizumab) with which nearly 30% of durable responses have been observed [1]
Despite the undeniable progress made by these new therapies, more than half of patients are resistant or develop resistance. Resistance to targeted- and to immune-therapies frequently involves a de-differentiation process, associated with increased mesenchymal and pro- inflammatory phenotypes. This phenotypic switch frequently involves a loss of MITF and an increased AXL expression [2, 3] However, not all the resistant cells implement this molecular program and in the context of personalized medicine it is paramount to disclose all the molecular mechanisms underlying resistances and associated increased metastatic properties. This is a prerequisite to new therapeutic approaches to overcome these resistances and prevent metastatic dissemination.
The kinases of the SRC family comprise 8 members and have long been known to be oncogenes [4] Recent reports showed that resistance to BRAF inhibitors can be bypassed by a combination of BRAF inhibitor and SRC inhibitors [5-7] With the aims of deciphering the molecular mechanisms involved in the implementation of resistances and to identify pharmacologically targetable players, the inventors compared the expression of kinases of the SRC family members in melanoma cells isolated from patients before treatment with targeted therapy and after acquisition of resistance to BRAF inhibitors.
SUMMARY OF THE INVENTION:
The invention relates to methods for treating melanoma and therapy-resistant melanoma in a subject in need thereof comprising the step of administering said subject with a therapeutically effective amount of an activator of LYN. In particular, the present invention is defined by the claims.
DETAILED DESCRIPTION OF THE INVENTION:
Firstly, inventors have shown that the loss of expression of LYN is correlated with BRAF inhibitors resistance. Secondly, inventors have shown that overexpression of LYN in resistant cells that have lost LYN causes inhibition of their proliferation and migratory capacity.
Following the acquisition of the Ion Torent PGM Sequencer, they initiated the melanoma sequencing program. They decided to sequence 146 genes. Fifty genes are on the commercial panel "Ion AmpliSeq™ Cancer Hotspot Panel v2". A meta-analysis of exome sequencing data allowed them to construct a specific panel of melanoma, which includes 96 commonly mutated genes in melanoma. In a first step, they verified that the mutations found in cultured cells were identical to those found in the initial tumor. Then they performed the complete analysis of the 146 genes on 22 melanomas. Among these melanomas, 3 pairs of cells before treatment and after acquisition of Zelboraf or Tafmlar resistance. They did not find new mutations that would have appeared in resistant cells. Thus, a view of the mutational landscape allows to consider innovative therapeutic approaches in case of escape from current treatments.
Method for predicting the risk of sufferins from resistant melanoma
In a first aspect, the invention relates to a method for predicting whether a subject suffering from a melanoma is or is at risk of having resistant melanoma comprising the steps of i) quantifying the expression level of LYN in a biological sample obtained from the subject; ii) comparing the expression level quantified at step i) with a predetermined reference value and iii) concluding that the subject has or is at risk of suffering from resistant melanoma when the level determined at step i) is lower than the predetermined reference value.
As used herein, the term“melanoma” also known as malignant melanoma, refers to a type of cancer that develops from the pigment-containing cells, called melanocytes. There are three general categories of melanoma: 1) cutaneous melanoma which corresponds to melanoma of the skin; it is the most common type of melanoma; 2) mucosal melanoma which can occur
in any mucous membrane of the body, including the nasal passages, the throat, the vagina, the anus, or in the mouth; and 3) ocular melanoma also known as uveal melanoma or choroidal melanoma, is a rare form of melanoma that occurs in the eye. In a particular embodiment, the melanoma is cutaneous melanoma.
As used herein, the term“subject” denotes a mammal, such as a rodent, a feline, a canine, and a primate. Particularly, the subject according to the invention is a human. More particularly, the subject according to the invention has or is susceptible to have melanoma. In particular embodiment, the subject has or is susceptible to have cutaneous melanoma. In a particular embodiment, the subject has or is susceptible to have metastatic melanoma.
In a further embodiment, the subject has or is susceptible to have resistant melanoma.
As used herein, the term“resistant melanoma” refers to melanoma which does not respond to a treatment. The cancer may be resistant at the beginning of treatment or it may become resistant during treatment. The resistance to drug leads to rapid progression of metastatic of melanoma.
As used herein, the term“resistant melanoma cell” refers to cell which does not respond to a treatment.
As used herein, the term“sensitive melanoma cell” refers to cell which does respond to a treatment.
In some embodiments, the melanoma is resistant to BRAF inhibitors. BRAF is a member of the Raf kinase family of serine/threonine-specific protein kinases. This protein plays a role in regulating the MAP kinase / ERKs signaling pathway, which affects cell division, differentiation, and secretion. A number of mutations in BRAF are known. In particular, the V600E mutation is prominent. Other mutations which have been found are R461I, I462S, G463E, G463V, G465A, G465E, G465V, G468A, G468E, N580S, E585K, D593V, F594L, G595R, L596V, T598I, V599D, V599E, V599K, V599R, K600E, A727V, and most of these mutations are clustered to two regions: the glycine-rich P loop of the N lobe and the activation segment and flanking regions. In a particular embodiment, the BRAF mutation is V600E.
The inhibitors of BRAF mutations are well known in the art.
In some embodiments, the melanoma is resistant to MEK inhibitors. MEK refers to Mitogen-activated protein kinase kinase, also known as MAP2K, MEK, MAPKK. It is a kinase enzyme which phosphorylates mitogen-activated protein kinase (MAPK). MEK is activated in melanoma.
In some embodiments, the melanoma is resistant to NRAS inhibitors. The NRAS gene is in the Ras family of oncogene and involved in regulating cell division. NRAS mutations in codons 12, 13, and 61 arise in 15-20 % of all melanomas.
In some embodiments, the melanoma is resistant to immune checkpoint inhibitors.
As used herein, the term "immune checkpoint inhibitor" refers to molecules that totally or partially reduce, inhibit, interfere with or modulate one or more immune checkpoint proteins. As used herein, the term "immune checkpoint protein" has its general meaning in the art and refers to a molecule that is expressed by T cells in that either turn up a signal (stimulatory checkpoint molecules) or turn down a signal (inhibitory checkpoint molecules). Immune checkpoint molecules are recognized in the art to constitute immune checkpoint pathways similar to the CTLA-4 and PD-1 dependent pathways (see e.g. Pardoll, 2012. Nature Rev Cancer 12:252-264; Mellman et al. 2011. Nature 480:480- 489). Examples of stimulatory checkpoint include CD27 CD28 CD40, CD122, CD137, 0X40, GITR, and ICOS. Examples of inhibitory checkpoint molecules include A2AR, B7-H3, B7-H4, BTLA, CTLA-4, CD277, IDO, KIR, PD-1, LAG-3, TIM-3 and VISTA. The Adenosine A2A receptor (A2AR) is regarded as an important checkpoint in cancer therapy because adenosine in the immune microenvironment, leading to the activation of the A2a receptor, is negative immune feedback loop and the tumor microenvironment has relatively high concentrations of adenosine. B7-H3, also called CD276, was originally understood to be a co-stimulatory molecule but is now regarded as co-inhibitory. B7-H4, also called VTCN1, is expressed by tumor cells and tumor-associated macrophages and plays a role in tumour escape. B and T Lymphocyte Attenuator (BTLA) and also called CD272, has HVEM (Herpesvirus Entry Mediator) as its ligand. Surface expression of BTLA is gradually downregulated during differentiation of human CD8+ T cells from the naive to effector cell phenotype, however tumor-specific human CD8+ T cells express high levels of BTLA. CTLA-4, Cytotoxic T -Lymphocyte- Associated protein 4 and also called CD152. Expression of CTLA-4 on Treg cells serves to control T cell proliferation. IDO, Indoleamine 2,3-dioxygenase, is a tryptophan catabolic enzyme. A related immune-inhibitory enzymes. Another important molecule is TDO, tryptophan 2,3 -dioxygenase. IDO is known to suppress T and NK cells, generate and activate Tregs and myeloid-derived suppressor cells, and promote tumour angiogenesis. KIR, Killer-cell Immunoglobulin-like Receptor, is a receptor for MHC Class I molecules on Natural Killer cells. LAG3, Lymphocyte Activation Gene-3, works to suppress an immune response by action to Tregs as well as direct effects on CD8+ T cells. PD- 1, Programmed Death 1 (PD-1) receptor, has two ligands, PD-L1 and PD-L2. This checkpoint is the target of Merck & Co.'s melanoma drug Keytruda, which gained FDA approval in
September 2014. An advantage of targeting PD-1 is that it can restore immune function in the tumor microenvironment. TIM-3, short for T-cell Immunoglobulin domain and Mucin domain 3, expresses on activated human CD4+ T cells and regulates Thl and Thl7 cytokines. TIM-3 acts as a negative regulator of Thl/Tcl function by triggering cell death upon interaction with its ligand, galectin-9. VISTA, Short for V-domain Ig suppressor of T cell activation, VISTA is primarily expressed on hematopoietic cells so that consistent expression of VISTA on leukocytes within tumors may allow VISTA blockade to be effective across a broad range of solid tumors. Tumor cells often take advantage of these checkpoints to escape detection by the immune system. Thus, inhibiting a checkpoint protein on the immune system may enhance the anti-tumor T-cell response.
In some embodiments, an immune checkpoint inhibitor refers to any compound inhibiting the function of an immune checkpoint protein. Inhibition includes reduction of function and full blockade. In some embodiments, the immune checkpoint inhibitor could be an antibody, synthetic or native sequence peptides, small molecules or aptamers which bind to the immune checkpoint proteins and their ligands.
In a particular embodiment, the immune checkpoint inhibitor is an antibody.
Typically, antibodies are directed against A2AR, B7-H3, B7-H4, BTLA, CTLA-4, CD277, IDO, KIR, PD-1, LAG-3, TIM-3 or VISTA.
In a particular embodiment, the immune checkpoint inhibitor is an anti-PD-1 antibody such as described in WO2011082400, W02006121168, W02015035606, W02004056875, W02010036959, W02009114335, W02010089411, WO2008156712, WO2011110621, WO2014055648 and WO2014194302. Examples of anti-PD-1 antibodies which are commercialized: Nivolumab (Opdivo®, BMS), Pembrolizumab (also called Lambrolizumab, KEYTRUDA® or MK-3475, MERCK).
In some embodiments, the immune checkpoint inhibitor is an anti-PD-Ll antibody such as described in WO2013079174, W02010077634, W02004004771, WO2014195852, W02010036959, WO2011066389, W02007005874, W02015048520, US8617546 and WO2014055897. Examples of anti-PD-Ll antibodies which are on clinical trial: Atezolizumab (MPDL3280A, Genentech/Roche), Durvalumab (AZD9291, AstraZeneca), Avelumab (also known as MSB0010718C, Merck) and BMS-936559 (BMS).
In some embodiments, the immune checkpoint inhibitor is an anti-PD-L2 antibody such as described in US7709214, US7432059 and US8552154.
In the context of the invention, the immune checkpoint inhibitor inhibits Tim-3 or its ligand.
In a particular embodiment, the immune checkpoint inhibitor is an anti-Tim-3 antibody such as described in WO03063792, WO2011155607, WO2015117002, WO2010117057 and W02013006490.
In some embodiments, the immune checkpoint inhibitor is a small organic molecule.
The term "small organic molecule" as used herein, refers to a molecule of a size comparable to those organic molecules generally used in pharmaceuticals. The term excludes biological macro molecules (e. g. proteins, nucleic acids, etc.). Typically, small organic molecules range in size up to about 5000 Da, more preferably up to 2000 Da, and most preferably up to about 1000 Da.
Typically, the small organic molecules interfere with transduction pathway of A2AR, B7-H3, B7-H4, BTLA, CTLA-4, CD277, IDO, KIR, PD-1, LAG-3, TIM-3 or VISTA.
In a particular embodiment, small organic molecules interfere with transduction pathway of PD-1 and Tim-3. For example, they can interfere with molecules, receptors or enzymes involved in PD-1 and Tim-3 pathway.
In a particular embodiment, the small organic molecules interfere with Indoleamine- pyrrole 2,3-dioxygenase (IDO) inhibitor. IDO is involved in the tryptophan catabolism (Liu et al 2010, Vacchelli et al 2014, Zhai et al 2015). Examples of IDO inhibitors are described in WO 2014150677. Examples of IDO inhibitors include without limitation 1 -methyl-tryptophan (IMT), b- (3-benzofuranyl)-alanine, P-(3-benzo(b)thienyl)-alanine), 6-nitro-tryptophan, 6- fluoro-tryptophan, 4-methyl-tryptophan, 5 -methyl tryptophan, 6-methyl-tryptophan, 5- m ethoxy-tryptophan, 5 -hydroxy-tryptophan, indole 3-carbinol, 3,3'- diindolylmethane, epigallocatechin gallate, 5-Br-4-Cl-indoxyl 1,3-diacetate, 9- vinylcarbazole, acemetacin, 5- bromo-tryptophan, 5-bromoindoxyl diacetate, 3- Amino-naphtoic acid, pyrrolidine dithiocarbamate, 4-phenylimidazole a brassinin derivative, a thiohydantoin derivative, a b- carboline derivative or a brassilexin derivative. In a particular embodiment, the IDO inhibitor is selected from 1 -methyl-tryptophan, b-(3- benzofuranyl)-alanine, 6-nitro-L-tryptophan, 3- Amino-naphtoic acid and b-[3- benzo(b)thienyl] -alanine or a derivative or prodrug thereof.
In a particular embodiment, the inhibitor of IDO is Epacadostat, (INCB24360, INCB024360) has the following chemical formula in the art and refers to -N-(3-bromo-4- fluorophenyl)-N'-hydroxy-4-{[2-(sulfamoylamino)-ethyl]amino}-l,2,5-oxadiazole-3 carboximidamide :
In a particular embodiment, the inhibitor is BGB324, also called R428, such as described in W02009054864, refers to lH-1, 2, 4-Triazole-3, 5-diamine, l-(6,7-dihydro-5H- benzo[6,7]cyclohepta[l,2-c]pyridazin-3-yl)-N3-[(7S)-6,7,8,9-tetrahydro-7-(l-pyrrolidinyl)- 5H-benzocyclohepten-2-yl]- and has the following formula in the art:
In a particular embodiment, the inhibitor is CA-170 (or AUPM-170): an oral, small molecule immune checkpoint antagonist targeting programmed death ligand-1 (PD-L1) and V- domain Ig suppressor of T cell activation (VISTA) (Liu et al 2015). Preclinical data of CA-170 are presented by Curis Collaborator and Aurigene on November at ACR-NCI-EORTC International Conference on Molecular Targets and Cancer Therapeutics.
In some embodiments, the immune checkpoint inhibitor is an aptamer.
Typically, the aptamers are directed against A2AR, B7-H3, B7-H4, BTLA, CTLA-4, CD277, IDO, KIR, PD-1, LAG-3, TIM-3 or VISTA.
In a particular embodiment, aptamers are DNA aptamers such as described in Prodeus et al 2015. A major disadvantage of aptamers as therapeutic entities is their poor pharmacokinetic profiles, as these short DNA strands are rapidly removed from circulation due to renal filtration. Thus, aptamers according to the invention are conjugated to with high molecular weight polymers such as polyethylene glycol (PEG). In a particular embodiment, the aptamer is an anti-PD-1 aptamer. Particularly, the anti-PD-1 aptamer is MP7 pegylated as described in Prodeus et al 2015.
As used herein, the term "predicting the risk" refers to assessing the probability according to which the subject as referred to herein will develop resistant melanoma. As will be understood by those skilled in the art, such an assessment is usually not intended to be correct for 100% of the subjects to be investigated.
As used herein, the term“SRC kinase family” refers to family of non-receptor tyrosine kinases implicate in cancer and immune system dysfunction. Members of the SRC kinase family include the following nine kinases in mammals: Src, Yes, Fyn, and Fgr, forming the SrcA subfamily, Lck, Hck, Blk, and Lyn in the SrcB subfamily, and Frk in its own subfamily. These are non-receptor protein kinases that range in molecular mass from 52 to 62 kD.
In a further embodiment, the SRC kinase is LYN.
As used herein, the term“LYN” refers to Tyrosine-protein kinase Lyn, a protein that is encoded in human by the LYN gene. Lyn is mainly expressed in hematopoietic cells, in neural tissues, liver and adipose tissue. The naturally occurring human LYN gene has a nucleotide sequence as shown in Genbank Accession number NM_002350.3 and the naturally occurring human LYN protein has an aminoacid sequence as shown in Genbank Accession numbers NP_002341.1 for LYN isoform A and NP_001104567.1 for LYN isoform B.
As used herein, the term“expression level” refers to the expression level of LYN. Typically, the expression level of the LYN gene may be determined by any technology known by a person skilled in the art. In particular, each gene expression level may be measured at the genomic and/or nucleic and/or protein level. In a particular embodiment, the expression level of gene is determined by measuring the amount of nucleic acid transcripts of each gene. In another embodiment, the expression level is determined by measuring the amount of each gene corresponding protein. The amount of nucleic acid transcripts can be measured by any technology known by a man skilled in the art. In particular, the measure may be carried out directly on an extracted messenger RNA (mRNA) sample, or on retrotranscribed complementary DNA (cDNA) prepared from extracted mRNA by technologies well-known in the art. From the mRNA or cDNA sample, the amount of nucleic acid transcripts may be measured using any technology known by a man skilled in the art, including nucleic microarrays, quantitative PCR, microfluidic cards, and hybridization with a labelled probe. In a particular embodiment, the expression level is determined by using quantitative PCR. Quantitative, or real-time, PCR is a well-known and easily available technology for those skilled in the art and does not need a precise description. Methods for determining the quantity of mRNA are well known in the art. For example the nucleic acid contained in the biological sample is first extracted according to standard methods, for example using lytic enzymes or chemical solutions or extracted by nucleic-acid-binding resins following the manufacturer's instructions. The extracted mRNA is then detected by hybridization (e. g., Northern blot analysis) and/or amplification (e.g., RT-PCR). Preferably quantitative or semi-quantitative RT- PCR is preferred. Real-time quantitative or semi-quantitative RT-PCR is particularly
advantageous. Other methods of amplification include ligase chain reaction (LCR), transcription-mediated amplification (TMA), strand displacement amplification (SDA) and nucleic acid sequence based amplification (NASBA). Nucleic acids having at least 10 nucleotides and exhibiting sequence complementarity or homology to the mRNA of interest herein find utility as hybridization probes or amplification primers. It is understood that such nucleic acids do not need to be identical, but are typically at least about 80% identical to the homologous region of comparable size, more preferably 85% identical and even more preferably 90-95% identical. In certain embodiments, it will be advantageous to use nucleic acids in combination with appropriate means, such as a detectable label, for detecting hybridization. A wide variety of appropriate indicators are known in the art including, fluorescent, radioactive, enzymatic or other ligands (e. g. avidin/biotin). Probes typically comprise single-stranded nucleic acids of between 10 to 1000 nucleotides in length, for instance of between 10 and 800, more preferably of between 15 and 700, typically of between 20 and 500. Primers typically are shorter single-stranded nucleic acids, of between 10 to 25 nucleotides in length, designed to perfectly or almost perfectly match a nucleic acid of interest, to be amplified. The probes and primers are“specific” to the nucleic acids they hybridize to, i.e. they preferably hybridize under high stringency hybridization conditions (corresponding to the highest melting temperature Tm, e.g., 50 % formamide, 5x or 6x SCC. SCC is a 0.15 M NaCl, 0.015 M Na-citrate). The nucleic acid primers or probes used in the above amplification and detection method may be assembled as a kit. Such a kit includes consensus primers and molecular probes. A kit also includes the components necessary to determine if amplification has occurred. The kit may also include, for example, PCR buffers and enzymes; positive control sequences, reaction control primers; and instructions for amplifying and detecting the specific sequences. In a particular embodiment, the method of the invention comprises the steps of providing total RNAs extracted from a biological sample and subjecting the RNAs to amplification and hybridization to specific probes, more particularly by means of a quantitative or semi-quantitative RT-PCR. In another embodiment, the expression level is determined by DNA chip analysis. Such DNA chip or nucleic acid microarray consists of different nucleic acid probes that are chemically attached to a substrate, which can be a microchip, a glass slide or a microsphere-sized bead. A microchip may be constituted of polymers, plastics, resins, polysaccharides, silica or silica-based materials, carbon, metals, inorganic glasses, or nitrocellulose. Probes comprise nucleic acids such as cDNAs or oligonucleotides that may be about 10 to about 60 base pairs. To determine the expression level, a biological sample from a test subject, optionally first subjected to a reverse transcription, is labelled and contacted with
the microarray in hybridization conditions, leading to the formation of complexes between target nucleic acids that are complementary to probe sequences attached to the microarray surface. The labelled hybridized complexes are then detected and can be quantified or semi- quantified. Labelling may be achieved by various methods, e.g. by using radioactive or fluorescent labelling. Many variants of the microarray hybridization technology are available to the man skilled in the art (see e.g. the review by Hoheisel, Nature Reviews, Genetics, 2006, 7:200-210).
As used herein, the term“biological sample” refers to any sample obtained from a subject, such as a serum sample, a plasma sample, a urine sample, a blood sample, a lymph sample, or a tissue biopsy. In a particular embodiment, biological sample for the determination of an expression level include samples such as a blood sample, a lymph sample, or a biopsy. In a particular embodiment, the biological sample is a blood sample, more particularly, circulating tumor cells (CTCs). Detection of CTCs are available to the man skilled in the art (see e.g. the article by Long et al, Cancer Medecine, 2016, 5(6): 1022-1030 [9]).
Typically, the predetermined reference value is a threshold value or a cut-off value. Typically, a "threshold value" or "cut-off value" can be determined experimentally, empirically, or theoretically. A threshold value can also be arbitrarily selected based upon the existing experimental and/or clinical conditions, as would be recognized by a person of ordinary skilled in the art. For example, retrospective measurement of cell densities in properly banked historical subject samples may be used in establishing the predetermined reference value. The threshold value has to be determined in order to obtain the optimal sensitivity and specificity according to the function of the test and the benefit/risk balance (clinical consequences of false positive and false negative). Typically, the optimal sensitivity and specificity (and so the threshold value) can be determined using a Receiver Operating Characteristic (ROC) curve based on experimental data. For example, after quantifying the cell density in a group of reference, one can use algorithmic analysis for the statistic treatment of the measured densities in samples to be tested, and thus obtain a classification standard having significance for sample classification. The full name of ROC curve is receiver operator characteristic curve, which is also known as receiver operation characteristic curve. It is mainly used for clinical biochemical diagnostic tests. ROC curve is a comprehensive indicator that reflects the continuous variables of true positive rate (sensitivity) and false positive rate (1 -specificity). It reveals the relationship between sensitivity and specificity with the image composition method. A series of different cut-off values (thresholds or critical values, boundary values between normal and abnormal results of diagnostic test) are set as continuous variables to calculate a series of sensitivity and
specificity values. Then sensitivity is used as the vertical coordinate and specificity is used as the horizontal coordinate to draw a curve. The higher the area under the curve (AUC), the higher the accuracy of diagnosis. On the ROC curve, the point closest to the far upper left of the coordinate diagram is a critical point having both high sensitivity and high specificity values. The AUC value of the ROC curve is between 1.0 and 0.5. When AUC>0.5, the diagnostic result gets better and better as AUC approaches 1. When AUC is between 0.5 and 0.7, the accuracy is low. When AUC is between 0.7 and 0.9, the accuracy is moderate. When AUC is higher than 0.9, the accuracy is quite high. This algorithmic method is preferably done with a computer. Existing software or systems in the art may be used for the drawing of the ROC curve, such as: MedCalc 9.2.0.1 medical statistical software, SPSS 9.0, ROCPOWER.SAS, DESIGNROC.FOR, MULTIREADER POWER S AS, CREATE-ROC.SAS, GB STAT VIO.O (Dynamic Microsystems, Inc. Silver Spring, Md., USA), etc.
In some embodiments, the predetermined reference value is determined by carrying out a method comprising the steps of
a) providing a collection of tumor tissue samples from subject suffering from melanoma; b) providing, for each tumor tissue sample provided at step a), information relating to the actual clinical outcome for the corresponding subject
c) providing a serial of arbitrary quantification values;
d) quantifying the cell density for each tumor tissue sample contained in the collection provided at step a);
e) classifying said tumor tissue samples in two groups for one specific arbitrary quantification value provided at step c), respectively: (i) a first group comprising tumor tissue samples that exhibit a quantification value for level that is lower than the said arbitrary quantification value contained in the said serial of quantification values; (ii) a second group comprising tumor tissue samples that exhibit a quantification value for said level that is higher than the said arbitrary quantification value contained in the said serial of quantification values; whereby two groups of tumor tissue samples are obtained for the said specific quantification value, wherein the tumor tissue samples of each group are separately enumerated;
f) calculating the statistical significance between (i) the quantification value obtained at step e) and (ii) the actual clinical outcome of the subjects from which tumor tissue samples contained in the first and second groups defined at step f) derive;
g) reiterating steps f) and g) until every arbitrary quantification value provided at step d) is tested;
h) setting the said predetermined reference value as consisting of the arbitrary quantification value for which the highest statistical significance (most significant P-value obtained with a log-rank test, significance when P<0.05) has been calculated at step g).
For example the cell density has been assessed for 100 tumor tissue samples of 100 subjects. The 100 samples are ranked according to the cell density. Sample 1 has the highest density and sample 100 has the lowest density. A first grouping provides two subsets: on one side sample Nr 1 and on the other side the 99 other samples. The next grouping provides on one side samples 1 and 2 and on the other side the 98 remaining samples etc., until the last grouping: on one side samples 1 to 99 and on the other side sample Nr 100. According to the information relating to the actual clinical outcome for the corresponding cancer subject, Kaplan-Meier curves are prepared for each of the 99 groups of two subsets. Also for each of the 99 groups, the p value between both subsets was calculated (log-rank test). The predetermined reference value is then selected such as the discrimination based on the criterion of the minimum P-value is the strongest. In other terms, the cell density corresponding to the boundary between both subsets for which the P-value is minimum is considered as the predetermined reference value. It should be noted that the predetermined reference value is not necessarily the median value of cell densities. Thus in some embodiments, the predetermined reference value thus allows discrimination between a poor and a good prognosis with respect to DFS and OS for a subject. Practically, high statistical significance values (e.g. low P values) are generally obtained for a range of successive arbitrary quantification values, and not only for a single arbitrary quantification value. Thus, in one alternative embodiment of the invention, instead of using a definite predetermined reference value, a range of values is provided. Therefore, a minimal statistical significance value (minimal threshold of significance, e.g. maximal threshold P value) is arbitrarily set and a range of a plurality of arbitrary quantification values for which the statistical significance value calculated at step g) is higher (more significant, e.g. lower P-value) are retained, so that a range of quantification values is provided. This range of quantification values includes a "cut-off value as described above. For example, according to this specific embodiment of a "cut-off value, the outcome can be determined by comparing the cell density with the range of values which are identified. In some embodiments, a cut-off value thus consists of a range of quantification values, e.g. centered on the quantification value for which the highest statistical significance value is found (e.g. generally the minimum P-value which is found).
Method for treating melanoma and resistant melanoma
Inventors have shown that an activation of LYN in resistant cells that display a reduced level of LYN causes inhibition of proliferation and migratory capacity of melanoma cells.
Accordingly, in a second aspect, the invention relates to a method for treating melanoma in a subject in need thereof comprising a step of administering said subject with a therapeutically effective amount of an activator of LYN.
In a particular embodiment, the subject is identified as having melanoma resistance by performing the method as described above.
Accordingly, in a third aspect, the invention relates to a method for treating resistant melanoma in a subject in need thereof comprising a step of administering said subject with a therapeutically effective amount of an activator of LYN.
In a particular embodiment, the subject is identified as having melanoma resistant to a classical treatment.
In some embodiments, the subject is identified as having BRAF inhibitors resistance.
In some embodiments, the subject is identified as having MEK inhibitors resistance.
In some embodiments, the subject is identified as having NRAS inhibitors resistance.
In some embodiments, the subject is identified as having immune checkpoint inhibitors resistance.
In some embodiments, the activators of LYN may be combined with an immune checkpoint inhibitor. In some embodiment, the present invention relates to a method for treating melanoma and/or resistant melanoma in a subject in need thereof comprising a step of administering said subject with a therapeutically effective amount of an activator of LYN and of an immune checkpoint inhibitor.
As used herein, the terms“treating” or“treatment” refer to both prophylactic or preventive treatment as well as curative or disease modifying treatment, including treatment of subject at risk of contracting the disease or suspected to have contracted the disease as well as subject who are ill or have been diagnosed as suffering from a disease or medical condition, and includes suppression of clinical relapse. The treatment may be administered to a subject having a medical disorder or who ultimately may acquire the disorder, in order to prevent, cure, delay the onset of, reduce the severity of, or ameliorate one or more symptoms of a disorder or recurring disorder, or in order to prolong the survival of a subject beyond that expected in the absence of such treatment. By "therapeutic regimen" is meant the pattern of treatment of an illness, e.g., the pattern of dosing used during therapy. A therapeutic regimen may include an induction regimen and a maintenance regimen. The phrase "induction regimen" or "induction
period" refers to a therapeutic regimen (or the portion of a therapeutic regimen) that is used for the initial treatment of a disease. The general goal of an induction regimen is to provide a high level of drug to a subject during the initial period of a treatment regimen. An induction regimen may employ (in part or in whole) a "loading regimen", which may include administering a greater dose of the drug than a physician would employ during a maintenance regimen, administering a drug more frequently than a physician would administer the drug during a maintenance regimen, or both. The phrase "maintenance regimen" or "maintenance period" refers to a therapeutic regimen (or the portion of a therapeutic regimen) that is used for the maintenance of a subject during treatment of an illness, e.g., to keep the subject in remission for long periods of time (months or years). A maintenance regimen may employ continuous therapy (e.g., administering a drug at a regular intervals, e.g., weekly, monthly, yearly, etc.) or intermittent therapy (e.g., interrupted treatment, intermittent treatment, treatment at relapse, or treatment upon achievement of a particular predetermined criteria [e.g., pain, disease manifestation, etc.]).
As used herein, the term“activator of LYN” refers to a natural or synthetic compound that has a biological effect to increase the activity of LYN. More particularly, such compound induces phosphorylation of Tyr396 in the activation loop of LYN that generates a highly active LYNand/or increases the kinase activity of LYN toward its natural (Cortactin, HS1) or synthetic substrates (Poly (Glu4-Tyr) or SRCtide (GAELPPEF A AQLRKIGDK V Y C (SEQ ID NO: 1))
In one embodiment, the activator of LYN is a LYN allosteric activator. In a particular embodiment, the activator of LYN is tolimidone.
As used herein, the term “tolimidone” also known as MLR-1023 or 2(1H)- pyrimidinone,5-(3-methylphenoxy) has the formula Cl 1H10N2O2 and the following structure in the art:
In a particular embodiment, the activator of LYN is a derivative of MLR-1023 as describes in W02010/003055.
In a particular embodiment, the activator of LYN is a peptide, petptidomimetic, small organic molecule, antibody, aptamers, siRNA or antisense oligonucleotide. The term “peptido mimetic” refers to a small protein-like chain designed to mimic a peptide. In a
particular embodiment, the activator of LYN is an aptamer. Aptamers are a class of molecule that represents an alternative to antibodies in term of molecular recognition. Aptamers are oligonucleotide or oligopeptide sequences with the capacity to recognize virtually any class of target molecules with high affinity and specificity.
In a particular embodiment, the activator of LYN is a small organic molecule. The term “small organic molecule” refers to a molecule of a size comparable to those organic molecules generally used in pharmaceuticals. The term excludes biological macromolecules (e.g., proteins, nucleic acids, etc.). Preferred small organic molecules range in size up to about 5000 Da, more preferably up to 2000 Da, and most preferably up to about 1000 Da.
In some embodiments, the activator of LYN is an antibody. As used herein, the term “antibody” is used in the broadest sense and specifically covers monoclonal antibodies, polyclonal antibodies, multispecific antibodies (e.g. bispecific antibodies) formed from at least two intact antibodies, and antibody fragments so long as they exhibit the desired biological activity. The term includes antibody fragments that comprise an antigen binding domain such as Fab', Fab, F(ab')2, single domain antibodies (DABs), TandAbs dimer, Fv, scFv (single chain Fv), dsFv, ds-scFv, Fd, linear antibodies, minibodies, diabodies, bispecific antibody fragments, bibody, tribody (scFv-Fab fusions, bispecific or trispecific, respectively); sc-diabody; kappa(lamda) bodies (scFv-CL fusions); BiTE (Bispecific T-cell Engager, scFv-scFv tandems to attract T cells); DVD-Ig (dual variable domain antibody, bispecific format); SIP (small immunoprotein, a kind of minibody); SMIP ("small modular immunopharmaceutical" scFv-Fc dimer; DART (ds-stabilized diabody "Dual Affinity ReTargeting"); small antibody mimetics comprising one or more CDRs and the like. The techniques for preparing and using various antibody-based constructs and fragments are well known in the art (see Rabat et ak, 1991, specifically incorporated herein by reference). Diabodies, in particular, are further described in EP 404, 097 and WO 93/1 1 161; whereas linear antibodies are further described in Zapata et al. (1995). Antibodies can be fragmented using conventional techniques. For example, F(ab')2 fragments can be generated by treating the antibody with pepsin. The resulting F(ab')2 fragment can be treated to reduce disulfide bridges to produce Fab' fragments. Papain digestion can lead to the formation of Fab fragments. Fab, Fab' and F(ab')2, scFv, Fv, dsFv, Fd, dAbs, TandAbs, ds-scFv, dimers, minibodies, diabodies, bispecific antibody fragments and other fragments can also be synthesized by recombinant techniques or can be chemically synthesized. Techniques for producing antibody fragments are well known and described in the art. For example, each of Beckman et al, 2006; Holliger & Hudson, 2005; Le Gall et al, 2004; Reff & Heard, 2001 ; Reiter et al., 1996; and Young et al., 1995 further describe and enable the production of effective
antibody fragments. In some embodiments, the antibody is a“chimeric” antibody as described in U.S. Pat. No. 4,816,567. In some embodiments, the antibody is a humanized antibody, such as described U.S. Pat. Nos. 6,982,321 and 7,087,409. In some embodiments, the antibody is a human antibody. A“human antibody” such as described in US 6,075, 181 and 6, 150,584. In some embodiments, the antibody is a single domain antibody such as described in EP 0 368 684, WO 06/030220 and WO 06/003388.
In a particular embodiment, the activator of LYN is a monoclonal antibody. Monoclonal antibodies can be prepared and isolated using any technique that provides for the production of antibody molecules by continuous cell lines in culture. Techniques for production and isolation include but are not limited to the hybridoma technique, the human B-cell hybridoma technique and the EBV-hybridoma technique.
In a particular, the activator is an intrabody having specificity for LYN. As used herein, the term "intrabody" generally refer to an intracellular antibody or antibody fragment. Antibodies, in particular single chain variable antibody fragments (scFv), can be modified for intracellular localization. Such modification may entail for example, the fusion to a stable intracellular protein, such as, e.g., maltose binding protein, or the addition of intracellular trafficking/localization peptide sequences, such as, e.g., the endoplasmic reticulum retention. In some embodiments, the intrabody is a single domain antibody. In some embodiments, the antibody according to the invention is a single domain antibody. The term“single domain antibody” (sdAb) or "VHH" refers to the single heavy chain variable domain of antibodies of the type that can be found in Camelid mammals which are naturally devoid of light chains. Such VHH are also called“nanobody®”. According to the invention, sdAb can particularly be llama sdAb.
In order to test the functionality of a putative LYN activator a test is necessary. For that purpose, to identify LYN activator an in vitro kinase assay will be used. In brief, recombinant LYN kinase is incubated with a synthetic substrate (SRCtides), ATP as phosphate donor, and increasing concentration of the potential activators. MLR- 1023 is used as reference agent, and Staurosporine, (a known nonspecific LYN inhibitor) as inhibition control.
Acquired resistance to targeted therapies is currently a clinical challenge in the treatment of advanced metastatic melanoma. Therefore, inventors examined the impact of targeting LYN in melanoma cells resistant to BRAF inhibitors (BRAFi). They have shown that melanoma treatment with pharmacological LYN activator can overcome resistance to drugs targeting oncogenic BRAF.
The resistance of cancer for the medication is caused by mutations in the genes which are involved in the proliferation, divisions or differentiation of cells or by phenotypic switch with a transcriptional profile favoring the resistance to treatment. In the context of the invention, the resistance of melanoma is caused by the mutations (single or double) in the following genes: BRAF, MEK or NRAS. The resistance can be also caused by a double-negative BRAF and NRAS mutation or phenotypic switch.
In a particular embodiment, the melanoma is resistant to a treatment with the inhibitors of BRAF mutations.
In some embodiments, the melanoma is resistant to a treatment.
In a further embodiment, the melanoma is resistant to a treatment with the inhibitors of BRAF. In a particular embodiment, the melanoma is resistant to a treatment with dabrafenib also known as tafmlar which is commercialized by Novartis. In a particular embodiment, the melanoma is resistant to a treatment with vemurafenib. Vemurafenib also known as PLX4032, RG7204 ou R05185426 and commercialized by Roche as Zelboraf. In a particular embodiment, the melanoma is resistant to a treatment with dacarbazine. Dacarbazine also known as imidazole carboxamide is commercialized as DTIC-Dome by Bayer.
In a further embodiment, the melanoma is resistant to a treatment with the inhibitors of MEK. The inhibitors of MEK are well known in the art. In a particular embodiment, the melanoma is resistant to a treatment with trametinib also known as mekinist which is commercialized by GSK. In a particular embodiment, the melanoma is resistant to a treatment with cobimetinib also known as cotellic commercialized by Genentech. In a particular embodiment, the melanoma is resistant to a treatment with Binimetinib also knowns as MEK162, ARRY-162 is developed by Array Biopharma.
In a particular embodiment, the melanoma is resistant to a treatment with the inhibitors of NRAS. In a particular embodiment, the melanoma is resistant to a treatment with salirasib commercialized by Concordia Pharmaceuticals. The inhibitors of BRAF mutation or MEK are used to treat the melanoma with NRAS mutations. In a particular embodiment, the melanoma is resistant in which double-negative BRAF and NRAS mutant melanoma.
In a particular embodiment, the melanoma is resistant to a combined treatment. As used herein, the terms“combined treatment”,“combined therapy” or“therapy combination” refer to a treatment that uses more than one medication. The combined therapy may be dual therapy or bi-therapy. In the context of the invention, the melanoma is resistant to a combined treatment characterized by using an inhibitor of BRAF mutation and an inhibitor of MEK or an inhibitor of BRAF mutation and an inhibitor of NRAS as described above. For example, the combined
treatment may be a combination of Vemurafenib (BRAFi) and Cobimetinib (MEKi), or a combination of Dabrafenib (BRAFi) and Trametinib (MEKi), or a combination of Cobimetinib Encorafenib (BRAFi) and Binimetinib(MEKi).
In a further embodiment, the melanoma is resistant to a treatment with an immune checkpoint inhibitor. The term“immune checkpoint inhibitor”, as used herein, is defined above.
In a particular embodiment, the melanoma is also resistant to a combined treatment characterized by using an inhibitor of BRAF mutation and an inhibitor of immune checkpoint.
In a particular embodiment, the melanoma is also resistant to a combined treatment characterized by using an inhibitor of BRAF mutation, an inhibitor of MEK and an inhibitor of immune checkpoint.
In a particular embodiment, the subject has or susceptible to have melanoma resistant to at least one of the treatments as described above. The subject having a melanoma resistant is identified by standard criteria. The standard criteria for resistance, for example, are Response Evaluation Criteria In Solid Tumors (RECIST) criteria, published by an international consortium including NCI.
As used herein the terms "administering" or "administration" refer to the act of injecting or otherwise physically delivering a substance as it exists outside the body (e.g., an activator of LYN) into the subject, such as by mucosal, intradermal, intravenous, subcutaneous, intramuscular delivery and/or any other method of physical delivery described herein or known in the art. When a disease, or a symptom thereof, is being treated, administration of the substance typically occurs after the onset of the disease or symptoms thereof. When a disease or symptoms thereof, are being prevented, administration of the substance typically occurs before the onset of the disease or symptoms thereof.
By a "therapeutically effective amount" is meant a sufficient amount of activator of LYN for use in a method for the treatment of melanoma at a reasonable benefit/risk ratio applicable to any medical treatment. It will be understood that the total daily usage of the compounds and compositions of the present invention will be decided by the attending physician within the scope of sound medical judgment. The specific therapeutically effective dose level for any particular subject will depend upon a variety of factors including the age, body weight, general health, sex and diet of the subject; the time of administration, route of administration, and rate of excretion of the specific compound employed; the duration of the treatment; drugs used in combination or coincidental with the specific polypeptide employed; and like factors well known in the medical arts. For example, it is well known within the skill of the art to start doses of the compound at levels lower than those required to achieve the
desired therapeutic effect and to gradually increase the dosage until the desired effect is achieved. However, the daily dosage of the products may be varied over a wide range from 0.01 to 1,000 mg per adult per day. Typically, the compositions contain 0.01, 0.05, 0.1, 0.5, 1.0, 2.5, 5.0, 10.0, 15.0, 25.0, 50.0, 100, 250 and 500 mg of the active ingredient for the symptomatic adjustment of the dosage to the subject to be treated. A medicament typically contains from about 0.01 mg to about 500 mg of the active ingredient, typically from 1 mg to about 100 mg of the active ingredient. An effective amount of the drug is ordinarily supplied at a dosage level from 0.0002 mg/kg to about 20 mg/kg of body weight per day, especially from about 0.001 mg/kg to 7 mg/kg of body weight per day.
The activators of LYN as described above may be combined with pharmaceutically acceptable excipients, and optionally sustained-release matrices, such as biodegradable polymers, to form pharmaceutical compositions. "Pharmaceutically" or "pharmaceutically acceptable" refer to molecular entities and compositions that do not produce an adverse, allergic or other untoward reaction when administered to a mammal, especially a human, as appropriate. A pharmaceutically acceptable carrier or excipient refers to a non-toxic solid, semi-solid or liquid filler, diluent, encapsulating material or formulation auxiliary of any type. The pharmaceutical compositions of the present invention for oral, sublingual, subcutaneous, intramuscular, intravenous, transdermal, local or rectal administration, the active principle, alone or in combination with another active principle, can be administered in a unit administration form, as a mixture with conventional pharmaceutical supports, to animals and human beings. Suitable unit administration forms comprise oral-route forms such as tablets, gel capsules, powders, granules and oral suspensions or solutions, sublingual and buccal administration forms, aerosols, implants, subcutaneous, transdermal, topical, intraperitoneal, intramuscular, intravenous, subdermal, transdermal, intrathecal and intranasal administration forms and rectal administration forms. Typically, the pharmaceutical compositions contain vehicles which are pharmaceutically acceptable for a formulation capable of being injected. These may be in particular isotonic, sterile, saline solutions (monosodium or disodium phosphate, sodium, potassium, calcium or magnesium chloride and the like or mixtures of such salts), or dry, especially freeze-dried compositions which upon addition, depending on the case, of sterilized water or physiological saline, permit the constitution of injectable solutions. The pharmaceutical forms suitable for injectable use include sterile aqueous solutions or dispersions; formulations including sesame oil, peanut oil or aqueous propylene glycol; and sterile powders for the extemporaneous preparation of sterile injectable solutions or dispersions. In all cases, the form must be sterile and must be fluid to the extent that easy syringability exists.
It must be stable under the conditions of manufacture and storage and must be preserved against the contaminating action of microorganisms, such as bacteria and fungi. Solutions comprising compounds of the invention as free base or pharmacologically acceptable salts can be prepared in water suitably mixed with a surfactant, such as hydroxypropylcellulose. Dispersions can also be prepared in glycerol, liquid polyethylene glycols, and mixtures thereof and in oils. Under ordinary conditions of storage and use, these preparations contain a preservative to prevent the growth of microorganisms. The polypeptide (or nucleic acid encoding thereof) can be formulated into a composition in a neutral or salt form. Pharmaceutically acceptable salts include the acid addition salts (formed with the free amino groups of the protein) and which are formed with inorganic acids such as, for example, hydrochloric or phosphoric acids, or such organic acids as acetic, oxalic, tartaric, mandelic, and the like. Salts formed with the free carboxyl groups can also be derived from inorganic bases such as, for example, sodium, potassium, ammonium, calcium, or ferric hydroxides, and such organic bases as isopropylamine, trimethylamine, histidine, procaine and the like. The carrier can also be a solvent or dispersion medium containing, for example, water, ethanol, polyol (for example, glycerol, propylene glycol, and liquid polyethylene glycol, and the like), suitable mixtures thereof, and vegetables oils. The proper fluidity can be maintained, for example, by the use of a coating, such as lecithin, by the maintenance of the required particle size in the case of dispersion and by the use of surfactants. The prevention of the action of microorganisms can be brought about by various antibacterial and antifungal agents, for example, parabens, chlorobutanol, phenol, sorbic acid, thimerosal, and the like. In many cases, it will be preferable to include isotonic agents, for example, sugars or sodium chloride. Prolonged absorption of the injectable compositions can be brought about by the use in the compositions of agents delaying absorption, for example, aluminium monostearate and gelatin. Sterile injectable solutions are prepared by incorporating the active polypeptides in the required amount in the appropriate solvent with several of the other ingredients enumerated above, as required, followed by filtered sterilization. Generally, dispersions are prepared by incorporating the various sterilized active ingredients into a sterile vehicle which contains the basic dispersion medium and the required other ingredients from those enumerated above. In the case of sterile powders for the preparation of sterile injectable solutions, the preferred methods of preparation are vacuum drying and freeze-drying techniques which yield a powder of the active ingredient plus any additional desired ingredient from a previously sterile-filtered solution thereof. Upon formulation, solutions will be administered in a manner compatible with the dosage formulation and in such amount as is therapeutically effective. The formulations are easily administered in
a variety of dosage forms, such as the type of injectable solutions described above, but drug release capsules and the like can also be employed. For parenteral administration in an aqueous solution, for example, the solution should be suitably buffered if necessary and the liquid diluent first rendered isotonic with sufficient saline or glucose. These particular aqueous solutions are especially suitable for intravenous, intramuscular, subcutaneous and intrap eritoneal administration. In this connection, sterile aqueous media which can be employed will be known to those of skill in the art in light of the present disclosure. For example, one dosage could be dissolved in 1 ml of isotonic NaCl solution and either added to 1000 ml of hypodermoclysis fluid or injected at the proposed site of infusion. Some variation in dosage will necessarily occur depending on the condition of the subject being treated. The person responsible for administration will, in any event, determine the appropriate dose for the individual subject.
Method of screening
A further object of the present invention relates to a method of screening a drug suitable for the treatment of melanoma comprising i) providing a test compound and ii) determining the ability of said test compound to activate the activity of LYN.
Any biological assay well known in the art could be suitable for determining the ability of the test compound to activate the activity of LYN. Such assay is briefly described above.
In particular, the effect triggered by the test compound is determined relative to that of the reference compound (MLR- 1023), in the absence of the test compound or in the presence of a control agent either of which is analogous to a negative control condition. The term "control substance", "control agent", or "control compound" as used herein refers a molecule that is inert or has no activity relating to an ability to modulate a biological activity or expression. It is to be understood that test compounds capable of activating the activity of LYN, as determined using in vitro methods described herein, are likely to exhibit similar modulatory capacity in applications in vivo. Typically, the test compound is selected from the group consisting of peptides, petptidomimetics, small organic molecules (such as MLR- 1023), aptamers or nucleic acids. For example, the test compound according to the invention may be selected from a library of compounds previously synthesised, or a library of compounds for which the structure is determined in a database, or from a library of compounds that have been synthesised de novo. In some embodiments, the test compound may be selected form small organic molecules.
The invention will be further illustrated by the following figures and examples. However, these examples and figures should not be interpreted in any way as limiting the scope of the present invention.
FIGURES:
Figure 1: LYN expression in BRAFi sensitive and resistant melanoma cells.
Analysis of LYN expression in publicly available data set (GSE50509) containing 31 pairs of melanoma tumors, before BRAFi treatment and after the acquisition of the resistance. Waterfall plot showed that LYN expression is decreased (Log Fold Change <-0.75) in about 22% of the resistant tumors.
Figure 2: Patients with melanoma expressing high level of LYN display a favorable prognosis. Kaplan-Meier analysis of the TCGA cohort. Patients with a low LYN expression in the tumors have a statistically significant reduced overall survival time (p<0.0001, log rank test (Mantel-Cox).
Figure 3. LYN inhibition increases the metastatic ability of melanoma cells.
Melanoma cells expressing high level of LYN (#2) were transfected with siLYN and assessed for motile ability using Boyden chamber experiments. Lyn down-regulation increases melanoma cells migration.
Figure 4: LYN allosteric activator, MLR1023 inhibits the invasive properties of melanoma cells. Quantification of the effect of MLR1023 on migration of different melanoma cells. Note that the cells that do not express LYN (patient#2R and patient#5) were not responsive to MLR1023.
EXAMPLE:
Material & Methods
Cell culture
The A375 S (Sensitive), A375 R (Resistant), WM9 S (Susceptible) and WM9 R (Resistant) human cell lines are grown under 5% C02 at 37 ° C in DMEM (Dulbecco Minimum Eagle Essential Medium) medium), Glutamax (Gibco) + 10% FCS + 50U / ml Penicillin + 50pg / ml Streptomycin. The cells of patients are isolated from biopsies after digestion with DNase, Dispase and Collagenase and cultured under 5% C02 at 37 ° C in RPMI medium (glucose, GlutaMAX, pyruvate, Invitrogen) 1640 Glutamax + 1% FCS + 50U / ml Penicillin + 50 pg / ml streptomycin.
Western Blot
Cells exposed to the different experimental conditions are lysed for 20 minutes at 4 ° C in Fisher buffer (50mM Hepes pH 7.4, 150mM NaCl, ImM Orthovanadate, 5mM NaF, 20mM NaPPi, Complete 7X cocktail and 1% TritonX-100). The lysates are then centrifuged (13000 rpm for 10 min at 4 ° C.) and the protein concentration is determined using micro BCA reagents (Thermo scientific), according to the supplier's recommendations by spectrophotometry at 561 nm. Fifteen micrograms of proteins are taken up in Laemmli loading buffer (40mM Na2HP04, 3% SDS, 10% glycerol, 0.01% bromophenol blue, 10% b-mercaptoethanol), denatured for 5 minutes at 95 ° C. and then deposited on a gel of Polyacrylamide (SDS-PAGE 10; 12,5 and 15). After migration (20mA, 2h:30), the proteins are transferred to a PVDF membrane (Immobilon, Millipore) using a semi-dry transfer system (TransBlot). Turbo, Biorad) The membrane is saturated (lOmM Tris-HCl pH 7.4, 150mM NaCl, ImM EDTA, 0.1% Tween-20, 3% BSA and 0.5% gelatin) for lh at room temperature and incubated 18h at 4 ° C, in the presence of the primary antibody directed against the protein of interest in the solution of saturation Then, after 3 washes of 10 min in washing buffer (10 mM Tris-HCl pH 7.6, 150 mM NaCl, 1% NP40), the membrane is incubated with a secondary antibody coupled to peroxidase (in saturation solution) for lh at room temperature. The membrane is washed again three times for 10 min and then the proteins of interest are revealed by chemoluminescence using an ECL reagent (Amersham Biosciences).
Transient transfection with small interfering RNA
The cells are seeded at a rate of 150,000 to 200,000 per ml in 60 mm diameter culture dishes and then transfected 24h later with a siRNA targeting the members of the SRC family and siRNA control (Flexitube, Qiagen) at a final concentration of 50nM with the HiPerfect transfection reagent (Qiagen) for 48 or 72h.
Infection with adenoviruses
Control or LYN encoding adenoviruses were from Vector Biolabs. We seed the melanoma cells in 6-well culture plates (400,000 cells per well in 2 ml of RPMI 7% FCS medium) and then infect them the next day with the adenoviral particles (1 and 3 pi / well of particles at 4.9 * 1010 PFU / ml). After 48 hours of infection and three washes with PBS, the cells are taken up in fresh medium, and used either for gene/protein expression, or migration assays
Migration assays
Cell migration analysis using Transwell® inserts, consists of an upper chamber and a lower chamber, separated by a porous membrane (8m) (Coming). The upper chamber is inoculated with a suspension of 300,000 cells in 200 mΐ of RPMI medium without FCS and
treated according to each condition. The lower chamber contains only 600pL of RMPI medium 7% FCS. The cells migrate for 24 hours according to chemotaxis from the upper chamber to the lower chamber through the porous membrane. The Transwell® inserts are then deposited in a 4% PFA solution for 10 min at room temperature in order to fix the cells having migrated. These are then stained with crystal violet at 0.2% for lOmin. After rinsing with PBS, drying and cleaning the upper part of the Transwell to remove cells that have not migrated, the cells are visualized under a microscope and counted using a software IMAGE J.
RNA preparation and quantitative real time PCR
The cells are washed with PBS and the RNAs are extracted using the RNeasy kit (Qiagen). The cell lysates are deposited on micro-columns, washed by several short centrifugation steps and eluted in 30 pi of H20 RNase. After quantification with a spectrophotometer (Nanodrop), 1 pg of RNA is retro-transcribed into cDNA with oligo dT as primer, according to the supplier's protocol (Reverse Transcription System, Promega). A 1 / 50th dilution of cDNA is incubated with 100pL of SYBR Green PCR master reagent mix (Applied biosystems) and the primers specific to the genes of interest diluted to 1/20. The PCR reaction is analyzed in real time using a StepOnePlus real time PCR system thermocycler (Applied Biosystems). The relative quantization of the amplicons was performed by the 2 (- AACT) method. Detection of the housekeeping GAPDH gene is used to normalize the results. The primer sequences used for each cDNA.
Statistical analyses
The statistical significance was determined by Student's t-test and the results are considered significant from p <0.05 p <0.01 p <0.001 "***".
Results
LYN expression is decreased in BRAFi-resistant melanoma cells.
First, the analysis of Cancer Cell Line Encyclopedia (CCLE) melanoma cell collection showed that melanoma cells lines expressed high level of FYN, LYN and YES1 (Data not shown). Unexpectedly, SRC was expressed at a very low level. However, numerous studies reported the expression of SRC in melanoma. Therefore, we focused our attention on these 4 kinases, FYN, LYN, SRC and YESl .
The analysis of 4 pairs of sensitive and resistant melanoma cells (resistant to BRAFi) by QPCR and western blot showed a loss of LYN in 3 out of 4 resistant cells. The other SRC kinases (YESl, FYN, SRC) did not show consistent changes between sensitive and resistant cells (Data not shown)
Next, the analysis of publicly available database (GSE50509) showed that among the 31 pairs of sensitive and resistant tumors, 7 (22%) have a decrease in LYN expression (Fig. 1), and in the TCGA cohort, low level of LYN is associated with decreased survival (Fig2.).
LYN impedes the metastatic properties of melanoma cells.
As the resistance is frequently associated with a phenotypic switch that lead to an increase in metastatic properties of melanoma cells, we studied the effect of LYN on cell migration in vitro and extravasation (a process required for tumor spreading) in vivo. Using Boyden chamber approach, we observed that inhibition of LYN expression by specific siRNA, increased migration of melanoma cells (Fig 3), while forced expression of LYN with an adenovirus inhibited their migrative property (Data not shown) demonstrating that LYN impaired the motility of melanoma cells that is one of the prerequisite for metastasis development.
In vivo, using lung extravasation model, we observed that melanoma cells over expressing LYN are not able to remain in the lung 24 hours after tail vein injection of the cells (Data not shown), thereby indicating that the cells were not able to cross the blood vessel wall to nest and develop in the lung parenchyma. Therefore, high LYN expression impedes the metastatic properties of melanoma cells.
Further, signaling analysis showed that LYN silencing increased Phospho-FAK and phospho-STAT3 (Data not shown), while forced expression of LYN inhibited phospho-FAK, consistently with an inhibition of the metastatic properties.
Pharmacological LYN activation inhibited the invasive properties of melanoma cells.
LYN it is clearly involved in the control of metastatic properties of melanoma cells. According to our data, LYN might be a valuable target to prevent or impair melanoma spreading. However, metastasis inhibition would be achieved through an increase in LYN expression or an activation of LYN activity.
Interestingly, a LYN kinase activator, MLR1023 is currently in phase 2b clinical trial for type 2 diabetes indication [8] Therefore, this drug, if efficient in preclinical model setting can rapidly be repositioned in the treatment of melanomas.
In vitro experiments demonstrated that MLR1023, induced phosphorylation of Tyr396 in the activation loop of LYN that generates a highly active LYN (Data not shown). Further, MLR1023, inhibited melanoma cell migration (Data not shown). Quantification of the effect of MLR1023 in several melanoma cells lines demonstrated a statistically significant effect of LYN
activation on 3 cell cultures out of 5. The two cell cultures (#2R and #5) that do not respond to MLR1023 do not express LYN, strengthening the specificity of the observed effect (Fig 4).
Conclusion
Counterintuitively to the pro-oncogenic role ascribed to LYN and SRC kinases, we demonstrated here that LYN acted negatively on the metastatic properties of melanoma cells. An increased LYN expression or LYN activation by an allosteric activator, currently in clinical trial for type 2 diabetes, decreases the metastatic properties of melanoma cells. These observations prompted us to propose a repositioning of the MLR1023 (i) in the treatment of metastatic melanomas to block further metastatic spreading (ii) as adjuvant therapy to prevent metastasis development after resection of primary or lymph node metastasis melanoma, and (iii) for the treatment of resistant melanomas that frequently acquire increased invasive properties caused by phenotypic switch.
REFERENCES:
Throughout this application, various references describe the state of the art to which this invention pertains. The disclosures of these references are hereby incorporated by reference into the present disclosure.
1. Luke, J.J. et al. Targeted agents and immunotherapies: optimizing outcomes in melanoma. Nat Rev Clin Oncol 14, 463-482 (2017).
2. Ahmed, F. et al. Microenvironment-Driven Dynamic Heterogeneity and
Phenotypic Plasticity as a Mechanism of Melanoma Therapy Resistance. Frontiers in oncology 8, 173 (2018).
3. Hugo, W. et al. Genomic and Transcriptomic Features of Response to Anti-PD- 1 Therapy in Metastatic Melanoma. Cell 168, 542 (2017).
4. Brickell, P.M. The p60c-src family of protein-tyrosine kinases: structure, regulation, and function. Critical reviews in oncogenesis 3, 401-446 (1992).
5. Girotti, M.R. et al. Paradox-breaking RAF inhibitors that also target SRC are effective in drug-resistant BRAF mutant melanoma. Cancer Cell 27, 85-96 (2015).
6. Girotti, M.R. et al. Inhibiting EGF receptor or SRC family kinase signaling overcomes BRAF inhibitor resistance in melanoma. Cancer Discov 3, 158-167 (2013).
7. Vergani, E. et al. Identification of MET and SRC activation in melanoma cell lines showing primary resistance to PLX4032. Neoplasia (New York, N.Y.) 13, 1132-1142
(2011).
8. Ochman, A.R. et al. The Lyn kinase activator MLR-1023 is a novel insulin receptor potentiator that elicits a rapid-onset and durable improvement in glucose homeostasis in animal models of type 2 diabetes. The Journal of pharmacology and experimental therapeutics 342, 23-32 (2012).
9. Long E., et al. High expression of TRF2, SOXIO, and CD10 in circulating tumor microemboli detected in metastatic melanoma patients. A potential impact for the assessment of disease aggressiveness. Cancer medicine, 5(6): 1022-1030 (2016)
Claims
1. A method for predicting whether a subject suffering from a melanoma is or is at risk of having resistant melanoma comprising the steps of i) quantifying the expression level of LYN in a biological sample obtained from the subject; ii) comparing the expression level quantified at step i) with a predetermined reference value and iii) concluding that the subject has or is at risk of suffering from resistant melanoma when the level determined at step i) is lower than the predetermined reference value.
2. A method for treating melanoma in a subject in need thereof comprising a step of administering said subject with a therapeutically effective amount of an activator of LYN.
3. A method for treating resistant melanoma in a subject in need thereof comprising a step of administering said subject with a therapeutically effective amount of an activator of LYN.
4. The method according to claims 2 to 3 comprising a step of administering said subject with a therapeutically effective amount of an activator of LYN and of an immune checkpoint inhibitor.
5. The method according to claims 2 to 4 wherein the activator of LYN is an LYN allosteric activator.
6. The method according to claims 2 to 5 wherein the activator of LYN is MLR1023.
7. The method according to claims 2 to 4, wherein the melanoma is resistant to a classical treatment.
8. The method according to claims 2 to 7, wherein the melanoma is resistant to a treatment with the inhibitors of BRAF mutations.
9. The method according to claims 2 to 7, wherein the melanoma is resistant to a treatment with the inhibitors of MEK.
10. The method according to claims 2 to 7, wherein the melanoma is resistant to a treatment with the inhibitors of NRAS.
11. The method according to claims 2 to 7, wherein the melanoma is resistant to a treatment with the immune checkpoint inhibitor.
12. A method of screening a drug suitable for the treatment of melanoma comprising i) providing a test compound and ii) determining the ability of said test compound to activate the activity of LYN.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP18306642 | 2018-12-07 | ||
| EP18306642.2 | 2018-12-07 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020115261A1 true WO2020115261A1 (en) | 2020-06-11 |
Family
ID=64665808
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2019/083926 Ceased WO2020115261A1 (en) | 2018-12-07 | 2019-12-06 | Methods and compositions for treating melanoma |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2020115261A1 (en) |
Citations (42)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4816567A (en) | 1983-04-08 | 1989-03-28 | Genentech, Inc. | Recombinant immunoglobin preparations |
| EP0368684A1 (en) | 1988-11-11 | 1990-05-16 | Medical Research Council | Cloning immunoglobulin variable domain sequences. |
| EP0404097A2 (en) | 1989-06-22 | 1990-12-27 | BEHRINGWERKE Aktiengesellschaft | Bispecific and oligospecific, mono- and oligovalent receptors, production and applications thereof |
| WO1993011161A1 (en) | 1991-11-25 | 1993-06-10 | Enzon, Inc. | Multivalent antigen-binding proteins |
| US6075181A (en) | 1990-01-12 | 2000-06-13 | Abgenix, Inc. | Human antibodies derived from immunized xenomice |
| US6150584A (en) | 1990-01-12 | 2000-11-21 | Abgenix, Inc. | Human antibodies derived from immunized xenomice |
| WO2003063792A2 (en) | 2002-01-30 | 2003-08-07 | The Brigham And Women's Hospital, Inc. | Compositions and methods related to tim-3, a th1-specific cell surface molecule |
| WO2004004771A1 (en) | 2002-07-03 | 2004-01-15 | Ono Pharmaceutical Co., Ltd. | Immunopotentiating compositions |
| WO2004056875A1 (en) | 2002-12-23 | 2004-07-08 | Wyeth | Antibodies against pd-1 and uses therefor |
| US6982321B2 (en) | 1986-03-27 | 2006-01-03 | Medical Research Council | Altered antibodies |
| WO2006003388A2 (en) | 2004-06-30 | 2006-01-12 | Domantis Limited | Compositions and methods for treating inflammatory disorders |
| WO2006030220A1 (en) | 2004-09-17 | 2006-03-23 | Domantis Limited | Compositions monovalent for cd40l binding and methods of use |
| US7087409B2 (en) | 1997-12-05 | 2006-08-08 | The Scripps Research Institute | Humanization of murine antibody |
| WO2006121168A1 (en) | 2005-05-09 | 2006-11-16 | Ono Pharmaceutical Co., Ltd. | Human monoclonal antibodies to programmed death 1(pd-1) and methods for treating cancer using anti-pd-1 antibodies alone or in combination with other immunotherapeutics |
| WO2007005874A2 (en) | 2005-07-01 | 2007-01-11 | Medarex, Inc. | Human monoclonal antibodies to programmed death ligand 1 (pd-l1) |
| US7432059B2 (en) | 2000-06-28 | 2008-10-07 | Dana-Farber Cancer Institute, Inc. | Methods of identifying compounds that upmodulate T cell activation in the presence of a PD-1 mediated signal |
| WO2008156712A1 (en) | 2007-06-18 | 2008-12-24 | N. V. Organon | Antibodies to human programmed death receptor pd-1 |
| WO2009054864A1 (en) | 2007-10-26 | 2009-04-30 | Rigel Pharmaceuticals, Inc. | Polycyclic aryl substituted triazoles and polycyclic heteroaryl substituted triazoles useful as axl inhibitors |
| WO2009114335A2 (en) | 2008-03-12 | 2009-09-17 | Merck & Co., Inc. | Pd-1 binding proteins |
| WO2010003055A1 (en) | 2008-07-03 | 2010-01-07 | Melior Pharmaceuticals I, Inc. | Compounds and methods for treating disorders related to glucose metabolism |
| WO2010036959A2 (en) | 2008-09-26 | 2010-04-01 | Dana-Farber Cancer Institute | Human anti-pd-1, pd-l1, and pd-l2 antibodies and uses therefor |
| WO2010077634A1 (en) | 2008-12-09 | 2010-07-08 | Genentech, Inc. | Anti-pd-l1 antibodies and their use to enhance t-cell function |
| WO2010089411A2 (en) | 2009-02-09 | 2010-08-12 | Universite De La Mediterranee | Pd-1 antibodies and pd-l1 antibodies and uses thereof |
| WO2010117057A1 (en) | 2009-04-10 | 2010-10-14 | 協和発酵キリン株式会社 | Method for treatment of blood tumor using anti-tim-3 antibody |
| WO2011066389A1 (en) | 2009-11-24 | 2011-06-03 | Medimmmune, Limited | Targeted binding agents against b7-h1 |
| WO2011082400A2 (en) | 2010-01-04 | 2011-07-07 | President And Fellows Of Harvard College | Modulators of immunoinhibitory receptor pd-1, and methods of use thereof |
| WO2011110621A1 (en) | 2010-03-11 | 2011-09-15 | Ucb Pharma, S.A. | Biological products: humanised agonistic anti-pd-1 antibodies |
| WO2011155607A1 (en) | 2010-06-11 | 2011-12-15 | 協和発酵キリン株式会社 | Anti-tim-3 antibody |
| WO2012018857A2 (en) * | 2010-08-02 | 2012-02-09 | The Broad Institute Of Mit And Harvard | Prediction of and monitoring cancer therapy response based on gene expression profiling |
| WO2013006490A2 (en) | 2011-07-01 | 2013-01-10 | Cellerant Therapeutics, Inc. | Antibodies that specifically bind to tim3 |
| WO2013079174A1 (en) | 2011-11-28 | 2013-06-06 | Merck Patent Gmbh | Anti-pd-l1 antibodies and uses thereof |
| US8617546B2 (en) | 2008-10-02 | 2013-12-31 | Seoul National University Industry Foundation | Anticancer agent comprising anti-PD-1 antibody or anti-PD-L1 antibody |
| WO2014055897A2 (en) | 2012-10-04 | 2014-04-10 | Dana-Farber Cancer Institute, Inc. | Human monoclonal anti-pd-l1 antibodies and methods of use |
| WO2014053650A1 (en) * | 2012-10-04 | 2014-04-10 | Ab Science | Use of masitinib for treatment of cancer in patient subpopulations identified using predictor factors |
| WO2014055648A1 (en) | 2012-10-02 | 2014-04-10 | Bristol-Myers Squibb Company | Combination of anti-kir antibodies and anti-pd-1 antibodies to treat cancer |
| WO2014150677A1 (en) | 2013-03-15 | 2014-09-25 | Bristol-Myers Squibb Company | Inhibitors of indoleamine 2,3-dioxygenase (ido) |
| WO2014194302A2 (en) | 2013-05-31 | 2014-12-04 | Sorrento Therapeutics, Inc. | Antigen binding proteins that bind pd-1 |
| WO2014195852A1 (en) | 2013-06-03 | 2014-12-11 | Glaxosmithkline Intellectual Property (No.2) Limited | Combinations of an anti-pd-l1 antibody and a mek inhibitor and/or a braf inhibitor |
| WO2015035606A1 (en) | 2013-09-13 | 2015-03-19 | Beigene, Ltd. | Anti-pd1 antibodies and their use as therapeutics and diagnostics |
| WO2015048520A1 (en) | 2013-09-27 | 2015-04-02 | Genentech, Inc. | Anti-pdl1 antibody formulations |
| WO2015117002A1 (en) | 2014-01-31 | 2015-08-06 | Novartis Ag | Antibody molecules to tim-3 and uses thereof |
| US20150366867A1 (en) * | 2012-12-20 | 2015-12-24 | Alan R. Tall | Methods of treating atherosclerosis or myeloproliferative neoplasms by administering a lyn kinase activator |
-
2019
- 2019-12-06 WO PCT/EP2019/083926 patent/WO2020115261A1/en not_active Ceased
Patent Citations (44)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4816567A (en) | 1983-04-08 | 1989-03-28 | Genentech, Inc. | Recombinant immunoglobin preparations |
| US6982321B2 (en) | 1986-03-27 | 2006-01-03 | Medical Research Council | Altered antibodies |
| EP0368684A1 (en) | 1988-11-11 | 1990-05-16 | Medical Research Council | Cloning immunoglobulin variable domain sequences. |
| EP0404097A2 (en) | 1989-06-22 | 1990-12-27 | BEHRINGWERKE Aktiengesellschaft | Bispecific and oligospecific, mono- and oligovalent receptors, production and applications thereof |
| US6075181A (en) | 1990-01-12 | 2000-06-13 | Abgenix, Inc. | Human antibodies derived from immunized xenomice |
| US6150584A (en) | 1990-01-12 | 2000-11-21 | Abgenix, Inc. | Human antibodies derived from immunized xenomice |
| WO1993011161A1 (en) | 1991-11-25 | 1993-06-10 | Enzon, Inc. | Multivalent antigen-binding proteins |
| US7087409B2 (en) | 1997-12-05 | 2006-08-08 | The Scripps Research Institute | Humanization of murine antibody |
| US7432059B2 (en) | 2000-06-28 | 2008-10-07 | Dana-Farber Cancer Institute, Inc. | Methods of identifying compounds that upmodulate T cell activation in the presence of a PD-1 mediated signal |
| US7709214B2 (en) | 2000-06-28 | 2010-05-04 | Dana-Farber Cancer Institute, Inc. | Methods for upregulating an immune response with agents that inhibit the intereaction between PD-L2 and PD-1 |
| WO2003063792A2 (en) | 2002-01-30 | 2003-08-07 | The Brigham And Women's Hospital, Inc. | Compositions and methods related to tim-3, a th1-specific cell surface molecule |
| WO2004004771A1 (en) | 2002-07-03 | 2004-01-15 | Ono Pharmaceutical Co., Ltd. | Immunopotentiating compositions |
| WO2004056875A1 (en) | 2002-12-23 | 2004-07-08 | Wyeth | Antibodies against pd-1 and uses therefor |
| WO2006003388A2 (en) | 2004-06-30 | 2006-01-12 | Domantis Limited | Compositions and methods for treating inflammatory disorders |
| WO2006030220A1 (en) | 2004-09-17 | 2006-03-23 | Domantis Limited | Compositions monovalent for cd40l binding and methods of use |
| WO2006121168A1 (en) | 2005-05-09 | 2006-11-16 | Ono Pharmaceutical Co., Ltd. | Human monoclonal antibodies to programmed death 1(pd-1) and methods for treating cancer using anti-pd-1 antibodies alone or in combination with other immunotherapeutics |
| WO2007005874A2 (en) | 2005-07-01 | 2007-01-11 | Medarex, Inc. | Human monoclonal antibodies to programmed death ligand 1 (pd-l1) |
| WO2008156712A1 (en) | 2007-06-18 | 2008-12-24 | N. V. Organon | Antibodies to human programmed death receptor pd-1 |
| WO2009054864A1 (en) | 2007-10-26 | 2009-04-30 | Rigel Pharmaceuticals, Inc. | Polycyclic aryl substituted triazoles and polycyclic heteroaryl substituted triazoles useful as axl inhibitors |
| WO2009114335A2 (en) | 2008-03-12 | 2009-09-17 | Merck & Co., Inc. | Pd-1 binding proteins |
| WO2010003055A1 (en) | 2008-07-03 | 2010-01-07 | Melior Pharmaceuticals I, Inc. | Compounds and methods for treating disorders related to glucose metabolism |
| WO2010036959A2 (en) | 2008-09-26 | 2010-04-01 | Dana-Farber Cancer Institute | Human anti-pd-1, pd-l1, and pd-l2 antibodies and uses therefor |
| US8552154B2 (en) | 2008-09-26 | 2013-10-08 | Emory University | Anti-PD-L1 antibodies and uses therefor |
| US8617546B2 (en) | 2008-10-02 | 2013-12-31 | Seoul National University Industry Foundation | Anticancer agent comprising anti-PD-1 antibody or anti-PD-L1 antibody |
| WO2010077634A1 (en) | 2008-12-09 | 2010-07-08 | Genentech, Inc. | Anti-pd-l1 antibodies and their use to enhance t-cell function |
| WO2010089411A2 (en) | 2009-02-09 | 2010-08-12 | Universite De La Mediterranee | Pd-1 antibodies and pd-l1 antibodies and uses thereof |
| WO2010117057A1 (en) | 2009-04-10 | 2010-10-14 | 協和発酵キリン株式会社 | Method for treatment of blood tumor using anti-tim-3 antibody |
| WO2011066389A1 (en) | 2009-11-24 | 2011-06-03 | Medimmmune, Limited | Targeted binding agents against b7-h1 |
| WO2011082400A2 (en) | 2010-01-04 | 2011-07-07 | President And Fellows Of Harvard College | Modulators of immunoinhibitory receptor pd-1, and methods of use thereof |
| WO2011110621A1 (en) | 2010-03-11 | 2011-09-15 | Ucb Pharma, S.A. | Biological products: humanised agonistic anti-pd-1 antibodies |
| WO2011155607A1 (en) | 2010-06-11 | 2011-12-15 | 協和発酵キリン株式会社 | Anti-tim-3 antibody |
| WO2012018857A2 (en) * | 2010-08-02 | 2012-02-09 | The Broad Institute Of Mit And Harvard | Prediction of and monitoring cancer therapy response based on gene expression profiling |
| WO2013006490A2 (en) | 2011-07-01 | 2013-01-10 | Cellerant Therapeutics, Inc. | Antibodies that specifically bind to tim3 |
| WO2013079174A1 (en) | 2011-11-28 | 2013-06-06 | Merck Patent Gmbh | Anti-pd-l1 antibodies and uses thereof |
| WO2014055648A1 (en) | 2012-10-02 | 2014-04-10 | Bristol-Myers Squibb Company | Combination of anti-kir antibodies and anti-pd-1 antibodies to treat cancer |
| WO2014055897A2 (en) | 2012-10-04 | 2014-04-10 | Dana-Farber Cancer Institute, Inc. | Human monoclonal anti-pd-l1 antibodies and methods of use |
| WO2014053650A1 (en) * | 2012-10-04 | 2014-04-10 | Ab Science | Use of masitinib for treatment of cancer in patient subpopulations identified using predictor factors |
| US20150366867A1 (en) * | 2012-12-20 | 2015-12-24 | Alan R. Tall | Methods of treating atherosclerosis or myeloproliferative neoplasms by administering a lyn kinase activator |
| WO2014150677A1 (en) | 2013-03-15 | 2014-09-25 | Bristol-Myers Squibb Company | Inhibitors of indoleamine 2,3-dioxygenase (ido) |
| WO2014194302A2 (en) | 2013-05-31 | 2014-12-04 | Sorrento Therapeutics, Inc. | Antigen binding proteins that bind pd-1 |
| WO2014195852A1 (en) | 2013-06-03 | 2014-12-11 | Glaxosmithkline Intellectual Property (No.2) Limited | Combinations of an anti-pd-l1 antibody and a mek inhibitor and/or a braf inhibitor |
| WO2015035606A1 (en) | 2013-09-13 | 2015-03-19 | Beigene, Ltd. | Anti-pd1 antibodies and their use as therapeutics and diagnostics |
| WO2015048520A1 (en) | 2013-09-27 | 2015-04-02 | Genentech, Inc. | Anti-pdl1 antibody formulations |
| WO2015117002A1 (en) | 2014-01-31 | 2015-08-06 | Novartis Ag | Antibody molecules to tim-3 and uses thereof |
Non-Patent Citations (18)
| Title |
|---|
| "Genbank", Database accession no. NP_002341.1 |
| AHMED, F. ET AL.: "Microenvironment-Driven Dynamic Heterogeneity and Phenotypic Plasticity as a Mechanism of Melanoma Therapy Resistance", FRONTIERS IN ONCOLOGY, vol. 8, 2018, pages 173 |
| BRICKELL, P.M.: "The p60c-src family of protein-tyrosine kinases: structure, regulation, and function", CRITICAL REVIEWS IN ONCOGENESIS, vol. 3, 1992, pages 401 - 446 |
| EVAN INGLEY: "Functions of the Lyn tyrosine kinase in health and disease", CELL COMMUNICATION AND SIGNALING, vol. 10, no. 1, 17 July 2012 (2012-07-17), BIOMED CENTRAL, LONDON, GB, pages 21, XP021133103, ISSN: 1478-811X, DOI: 10.1186/1478-811X-10-21 * |
| GIROTTI, M.R. ET AL.: "Inhibiting EGF receptor or SRC family kinase signaling overcomes BRAF inhibitor resistance in melanoma", CANCER DISCOV, vol. 3, 2013, pages 158 - 167, XP055145699, DOI: 10.1158/2159-8290.CD-12-0386 |
| GIROTTI, M.R. ET AL.: "Paradox-breaking RAF inhibitors that also target SRC are effective in drug-resistant BRAF mutant melanoma", CANCER CELL, vol. 27, 2015, pages 85 - 96, XP029131331, DOI: 10.1016/j.ccell.2014.11.006 |
| HOHEISEL, NATURE REVIEWS, GENETICS, vol. 7, 2006, pages 200 - 210 |
| HUGO, W. ET AL.: "Genomic and Transcriptomic Features of Response to Anti-PD-1 Therapy in Metastatic Melanoma", CELL, vol. 168, 2017, pages 542 |
| KIM, Y. J. ET AL.,: "Abstract 2785: LYN is a new prognostic and therapeutic target in non-small cell lung cancer", CANCER RESEARCH, vol. 74, no. 19 Suppl., 1 October 2014 (2014-10-01), XP002789294, DOI: 10.1158/1538-7445.AM2014-2785 * |
| LONG E. ET AL.: "High expression of TRF2, SOX10, and CD10 in circulating tumor microemboli detected in metastatic melanoma patients", A POTENTIAL IMPACT FOR THE ASSESSMENT OF DISEASE AGGRESSIVENESS. CANCER MEDICINE, vol. 5, no. 6, 2016, pages 1022 - 1030 |
| LONG ET AL., CANCER MEDECINE, vol. 5, no. 6, 2016, pages 1022 - 1030 |
| LUKE, J.J. ET AL.: "Targeted agents and immunotherapies: optimizing outcomes in melanoma", NAT REV CLIN ONCOL, vol. 14, 2017, pages 463 - 482 |
| M. S. SAPORITO ET AL: "MLR-1023 Is a Potent and Selective Allosteric Activator of Lyn Kinase In Vitro That Improves Glucose Tolerance In Vivo", JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS, vol. 342, no. 1, 1 July 2012 (2012-07-01), pages 15 - 22, XP055562815, DOI: 10.1124/jpet.112.192096 * |
| MELLMAN ET AL., NATURE, vol. 480, 2011, pages 480 - 489 |
| OCHMAN, A.R. ET AL.: "The Lyn kinase activator MLR-1023 is a novel insulin receptor potentiator that elicits a rapid-onset and durable improvement in glucose homeostasis in animal models of type 2 diabetes", THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS, vol. 342, 2012, pages 23 - 32, XP055543757, DOI: 10.1124/jpet.112.192187 |
| PARDOLL, NATURE REV CANCER, vol. 12, 2012, pages 252 - 264 |
| QIANQIAN ZHANG ET AL: "Lyn Kinase Promotes the Proliferation of Malignant Melanoma Cells through Inhibition of Apoptosis and Autophagy via the PI3K/Akt Signaling Pathway", JOURNAL OF CANCER, vol. 10, no. 5, 29 January 2019 (2019-01-29), AU, pages 1197 - 1208, XP055562828, ISSN: 1837-9664, DOI: 10.7150/jca.28908 * |
| VERGANI, E. ET AL.: "Identification of MET and SRC activation in melanoma cell lines showing primary resistance to PLX4032", NEOPLASIA, vol. 13, 2011, pages 1132 - 1142, XP055081482, DOI: 10.1593/neo.111102 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN105324491B (en) | Methods for diagnosis and treatment of cancer metastasis | |
| JP6212107B2 (en) | Methods for treating hair loss disorders | |
| KR102226219B1 (en) | Method for the diagnosis, prognosis and treatment of lung cancer metastasis | |
| US20230212679A1 (en) | Method for Predicting Effectiveness of Angiogenesis Inhibitor | |
| CN108192972B (en) | Methods for the diagnosis, prognosis and treatment of breast cancer metastasis | |
| CN105980576B (en) | Methods for prognosis and treatment of bone metastases derived from breast cancer | |
| CN104797935A (en) | Method for the prognosis and treatment of cancer metastasis | |
| US11674182B2 (en) | Biomarker for HER2-positive cancer and anti-HER2 therapy and applications thereof | |
| JP2016518815A (en) | Methods for diagnosis, prognosis, and treatment of metastatic cancer | |
| KR20220031069A (en) | Methods for diagnosing the effectiveness of anti-tumor treatment | |
| WO2016176617A2 (en) | Method for treating high-grade gliomas | |
| US11525008B2 (en) | Methods and pharmaceutical compositions for the treatment of lung cancer | |
| US20210186982A1 (en) | Methods and compositions for treating melanoma | |
| US20220364184A1 (en) | Compositions and methods for treatment of a poor prognosis subtype of colorectal cancer | |
| WO2020040245A1 (en) | Sensitivity marker for antibody-drug conjugate | |
| EP3591050B1 (en) | Use of rhoa in cancer diagnosis and inhibitor screening | |
| KR102074559B1 (en) | Biomarker for diagnosis of anticancer drug resistance of gastric cancer and use thereof | |
| KR101514877B1 (en) | Use of APE/Ref-1 and JAG1/Notch as a diagnostic marker of colon cancer | |
| WO2021048292A1 (en) | Methods and compositions for treating melanoma | |
| WO2022244807A1 (en) | Ltk fusion gene | |
| WO2015048367A1 (en) | Fusion proteins and methods of use thereof | |
| EP3067369A1 (en) | Methods and compositions for the treatment of anti-angiogenic resistant cancer | |
| US20210072244A1 (en) | Methods and compositions for treating melanoma resistant | |
| WO2020115261A1 (en) | Methods and compositions for treating melanoma | |
| WO2012034076A2 (en) | Etv1 as a diagnostic, prognostic and therapeutic target for gastrointestinal stromal tumors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19813032 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 19813032 Country of ref document: EP Kind code of ref document: A1 |