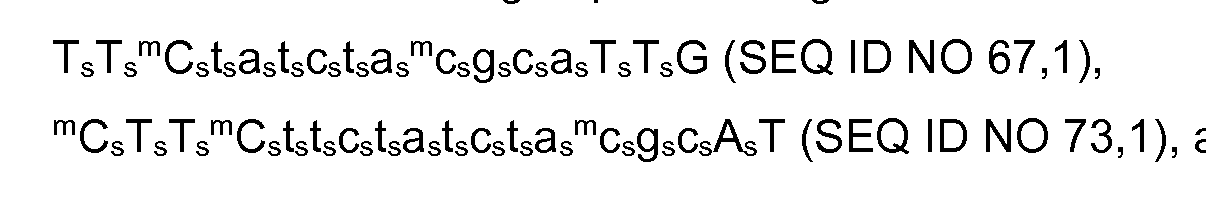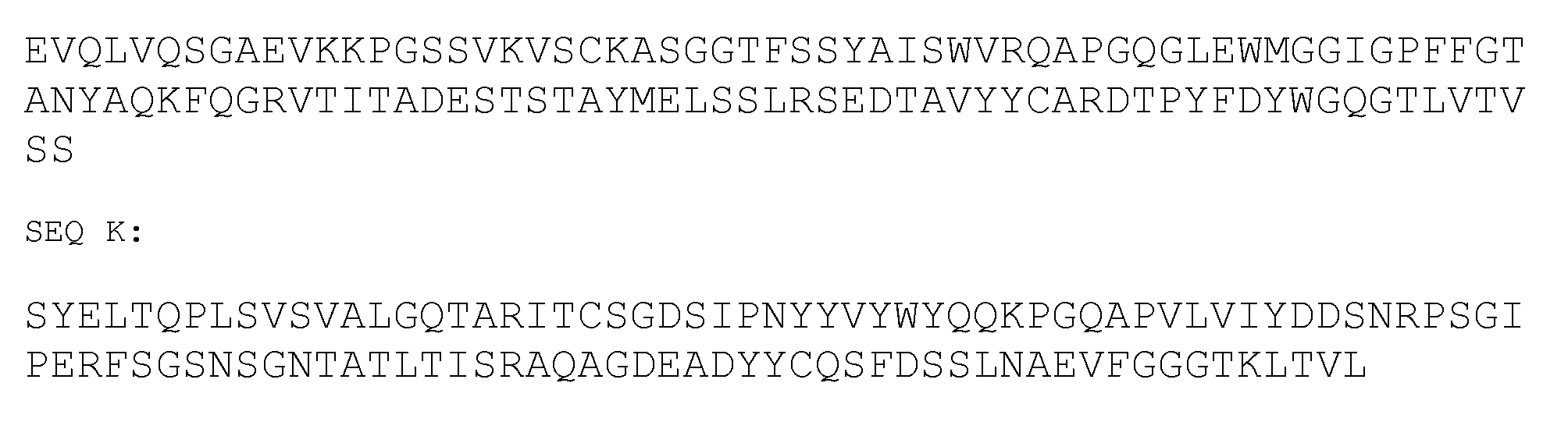WO2020109343A1 - Combination therapy for treatment of macular degeneration - Google Patents
Combination therapy for treatment of macular degeneration Download PDFInfo
- Publication number
- WO2020109343A1 WO2020109343A1 PCT/EP2019/082644 EP2019082644W WO2020109343A1 WO 2020109343 A1 WO2020109343 A1 WO 2020109343A1 EP 2019082644 W EP2019082644 W EP 2019082644W WO 2020109343 A1 WO2020109343 A1 WO 2020109343A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- antagonist
- antisense oligonucleotide
- oligonucleotide
- seq
- nucleosides
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 CC*=CC(N)=N Chemical compound CC*=CC(N)=N 0.000 description 1
- IIHPQZLOWODZRZ-ZUOJCQKJSA-N CCC[C@H](CCN)C(CC)C(/C=C\C(I)=C)C#C Chemical compound CCC[C@H](CCN)C(CC)C(/C=C\C(I)=C)C#C IIHPQZLOWODZRZ-ZUOJCQKJSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
- C12N15/1137—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing against enzymes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Y—ENZYMES
- C12Y304/00—Hydrolases acting on peptide bonds, i.e. peptidases (3.4)
- C12Y304/21—Serine endopeptidases (3.4.21)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/11—Antisense
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/31—Chemical structure of the backbone
- C12N2310/315—Phosphorothioates
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/32—Chemical structure of the sugar
- C12N2310/323—Chemical structure of the sugar modified ring structure
- C12N2310/3231—Chemical structure of the sugar modified ring structure having an additional ring, e.g. LNA, ENA
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/33—Chemical structure of the base
- C12N2310/334—Modified C
- C12N2310/3341—5-Methylcytosine
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/34—Spatial arrangement of the modifications
- C12N2310/341—Gapmers, i.e. of the type ===---===
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/34—Spatial arrangement of the modifications
- C12N2310/346—Spatial arrangement of the modifications having a combination of backbone and sugar modifications
Definitions
- wAMD wet age-related macular degeneration
- anti-angiogenic drugs such as anti-VEGF antibody therapeutics such as LUCENTISTM(ranibizumab).
- Other treatments of wAMD include laser therapy and photodynamic laser therapy.
- TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering a further therapeutic agent to the subject.
- the further therapeutic agent is an anti-platelet derived growth factor.
- the invention provides for the use of an antisense oligonucleotide of formula
- the invention provides for antisense oligonucleotide selected from the group selected from TTCtatctacgcaTTG (SEQ ID NO 67), CTTCttctatctacgcAT (SEQ ID NO 73), and
- the invention provides for antisense oligonucleotide selected from the group selected from TTCtatctacgcaTTG (SEQ ID NO 67), CTTCttctatctacgcAT (SEQ ID NO 73), and
- the invention provides for antisense oligonucleotide selected from the group selected from TTCtatctacgcaTTG (SEQ ID NO 67), CTTCttctatctacgcAT (SEQ ID NO 73), and
- the invention provides for antisense oligonucleotide of formula TTCtatctacgcaTTG (SEQ ID NO 67), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist complement factor C5 to the subject.
- the invention provides for antisense oligonucleotide of formula CTTCttctatctacgcAT (SEQ ID NO 73), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an anti-angiogenic agent to the subject.
- CTTCttctatctacgcAT SEQ ID NO 73
- the invention provides for antisense oligonucleotide of formula CTTCttctatctacgcAT (SEQ ID NO 73), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering a VEGF antagonist to the subject.
- CTTCttctatctacgcAT SEQ ID NO 73
- the invention provides for antisense oligonucleotide of formula CTTCttctatctacgcAT (SEQ ID NO 73), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist complement factor C5 to the subject.
- CTTCttctatctacgcAT SEQ ID NO 73
- the invention provides for antisense oligonucleotide of formula TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an anti-angiogenic agent to the subject.
- TACTttaatagcTCAA SEQ ID NO 86
- the invention provides for antisense oligonucleotide antagonist of HTRA1 for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an anti-angiogenic agent to the subject.
- the invention provides for antisense oligonucleotide antagonist of HTRA1 for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist of VEGF to the subject.
- the invention provides for antisense oligonucleotide antagonist of HTRA1 for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist of the complement system to the subject.
- the invention provides for antisense oligonucleotide antagonist of HTRA1 for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist of complement factor D to the subject.
- the invention provides for antisense oligonucleotide antagonist of HTRA1 for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist of complement factor B to the subject.
- the invention provides for antisense oligonucleotide antagonist of HTRA1 for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist of complement factor C5 to the subject.
- TTCtatctacgcaTTG (SEQ ID NO 67), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages, in the manufacture of a medicament for the treatment of dry age-related macular degeneration such as geographic atrophy, wherein the medicament is for use in combination treatment with a further therapeutic.
- the invention provides for a method for the treatment of dry age-related macular degeneration such as geographic atrophy in a subject in need of treatment, said method comprising administering an effective amount of the antisense oligonucleotide TTCtatctacgcaTTG (SEQ ID NO 67), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages, to said subject, and further administering an effective amount of a further therapeutic agent, to the subject.
- TTCtatctacgcaTTG SEQ ID NO 67
- the invention provides for a method for the treatment of dry age-related macular degeneration such as geographic atrophy in a subject in need of treatment, said method comprising administering an effective amount of the antisense oligonucleotide
- a Compound of the invention (Compound ID NO 67,1 ).
- the compound may be in the form of a pharmaceutical salt, such as a sodium salt or a potassium salt.
- FIG. 8 A Compound of the invention (Compound ID N073,1 ).
- the compound may be in the form of a pharmaceutical salt, such as a sodium salt or a potassium salt.
- FIG. 9 An example of a pharmaceutical salt of compound 67,1 : M+ is a suitable cation, typically a positive metal ion, such as a sodium or potassium ion.
- a suitable cation typically a positive metal ion, such as a sodium or potassium ion.
- the stoichiometric ratio of the cation to the oligonucleotide anion will depend on the charge of the cation used.
- FIG. 12A Compounds #15,3 and #17 were administered intravitreally in cynomolgus monkeys, and aqueous humor samples were collected at days 3, 8, 15, and 22 post injection. Proteins from undiluted samples were analyzed by capillary electrophoresis using a Peggy Sue device (Protein Simple). HTRA1 was detected using a custom-made polycolonal rabbit antiserum. Data from animals #J60154 (Vehicle), J60158 (C. Id#15,3), J60162 (C. Id#17) are presented.
- FIG 12B Signal intensities were quantified by comparison to purified recombinant (S328A mutant) HTRA1 protein (Origene, #TP700208). The calibration curve is shown here.
- Figure 12C Top panel: Calculated HTRA1 aqueous humor concentration from individual animal was plotted against time post injection. Bottom panel: average HTRA1 concentration for the vehicle group at each time point was determined and corresponding relative concentration in treated animals calculated. Open circle: individual value, closed circle: group average. % HTRA1 reduction for day 22 is indicated.
- FIG. 14 Correlation of HTRA1 protein in aqueous humor with (A) HTRA1 protein in retina and (B) HTRA1 mRNA in retina in cynomolgus monkeys treated with various LNA molecules targeting the HTRA1 transcript. Values are expressed as percentage normalized to PBS controls.
- the nucleotides of the oligonucleotide constitute the contiguous nucleotide sequence.
- the oligonucleotide comprises the contiguous nucleotide sequence, such as a F-G-F’ gapmer region, and may optionally comprise further nucleotide(s), for example a nucleotide linker region which may be used to attach a functional group to the contiguous nucleotide sequence.
- the nucleotide linker region may or may not be complementary to the target nucleic acid.
- the contiguous nucleotide sequence is 100% complementary to the target nucleic acid.
- Nucleotides are the building blocks of oligonucleotides and polynucleotides, and for the purposes of the present invention include both naturally occurring and non-naturally occurring nucleotides.
- nucleotides such as DNA and RNA nucleotides comprise a ribose sugar moiety, a nucleobase moiety and one or more phosphate groups (which is absent in nucleosides).
- Nucleosides and nucleotides may also interchangeably be referred to as“units” or“monomers”.
- modified nucleoside or“nucleoside modification” as used herein refers to nucleosides modified as compared to the equivalent DNA or RNA nucleoside by the introduction of one or more modifications of the sugar moiety or the (nucleo)base moiety.
- the modified nucleoside comprise a modified sugar moiety.
- modified nucleoside may also be used herein interchangeably with the term“nucleoside analogue” or modified“units” or modified“monomers”.
- Nucleosides with an unmodified DNA or RNA sugar moiety are termed DNA or RNA nucleosides herein. Nucleosides with modifications in the base region of the DNA or RNA nucleoside are still generally termed DNA or RNA if they allow Watson Crick base pairing.
- Modified internucieoside linkages are particularly useful in stabilizing oligonucleotides for in vivo use, and may serve to protect against nuclease cleavage at regions of DNA or RNA nucleosides in the oligonucleotide of the invention, for example within the gap region of a gapmer oligonucleotide, as well as in regions of modified nucleosides, such as region F and F’.
- the oligonucleotide comprises one or more internucleoside linkages modified from the natural phosphodiester, such one or more modified internucleoside linkages that is for example more resistant to nuclease attack.
- Nuclease resistance may be determined by incubating the oligonucleotide in blood serum or by using a nuclease resistance assay (e.g. snake venom phosphodiesterase (SVPD)), both are well known in the art.
- SVPD snake venom phosphodiesterase
- Internucleoside linkages which are capable of enhancing the nuclease resistance of an oligonucleotide are referred to as nuclease resistant internucleoside linkages.
- a preferred modified internucleoside linkage is phosphorothioate.
- Phosphorothioate internucleoside linkages are particularly useful due to nuclease resistance, beneficial pharmacokinetics and ease of manufacture.
- at least 50% of the internucleoside linkages in the oligonucleotide, or contiguous nucleotide sequence thereof are phosphorothioate, such as at least 60%, such as at least 70%, such as at least 80% or such as at least 90% of the internucleoside linkages in the oligonucleotide, or contiguous nucleotide sequence thereof, are phosphorothioate.
- all of the internucleoside linkages of the oligonucleotide, or contiguous nucleotide sequence thereof are phosphorothioate.
- Nuclease resistant linkages such as phosphorothioate linkages, are particularly useful in oligonucleotide regions capable of recruiting nuclease when forming a duplex with the target nucleic acid, such as region G for gapmers.
- Phosphorothioate linkages may, however, also be useful in non-nuclease recruiting regions and/or affinity enhancing regions such as regions F and F’ for gapmers.
- Gapmer oligonucleotides may, in some embodiments comprise one or more phosphodiester linkages in region F or F’, or both region F and F’, which the internucleoside linkage in region G may be fully phosphorothioate.
- nucleobase refers to both naturally occurring nucleobases such as adenine, guanine, cytosine, thymidine, uracil, xanthine and hypoxanthine, as well as non-naturally occurring variants. Such variants are for example described in Hirao et al (2012) Accounts of Chemical Research vol 45 page 2055 and Bergstrom (2009) Current Protocols in Nucleic Acid
- Watson-Crick base pairs are guanine (G)-cytosine (C) and adenine (A) - thymine (T)/uracil (U).
- G guanine
- A adenine
- T thymine
- U uracil
- oligonucleotides may comprise nucleosides with modified nucleobases, for example 5-methyl cytosine is often used in place of cytosine, and as such the term complementarity encompasses Watson Crick base-paring between non-modified and modified nucleobases (see for example Hirao et al (2012) Accounts of Chemical Research vol 45 page 2055 and Bergstrom (2009)
- % complementary refers to the number of nucleotides in percent of a contiguous nucleotide sequence in a nucleic acid molecule (e.g. oligonucleotide) which, at a given position, are complementary to ( i.e . form Watson Crick base pairs with) a contiguous sequence of nucleotides, at a given position of a separate nucleic acid molecule (e.g. the target nucleic acid or target sequence).
- a nucleic acid molecule e.g. oligonucleotide
- insertions and deletions are not allowed in the calculation of % complementarity of a contiguous nucleotide sequence.
- AG° can also be estimated numerically by using the nearest neighbor model as described by SantaLucia, 1998, Proc Natl Acad Sci USA. 95: 1460-1465 using appropriately derived thermodynamic parameters described by Sugimoto et al., 1995, Biochemistry 34:11211-1 1216 and McTigue et al., 2004, Biochemistry 43:5388-5405.
- oligonucleotides of the present invention hybridize to a target nucleic acid with estimated AG° values below -10 kcal for oligonucleotides that are 10-30 nucleotides in length.
- the degree or strength of hybridization is measured by the standard state Gibbs free energy AG°.
- the oligonucleotides may hybridize to a target nucleic acid with estimated AG° values below the range of -10 kcal, such as below -15 kcal, such as below - 20 kcal and such as below -25 kcal for oligonucleotides that are 8-30 nucleotides in length.
- the oligonucleotides hybridize to a target nucleic acid with an estimated AG° value of -10 to -60 kcal, such as -12 to -40, such as from -15 to -30 kcal or- 16 to -27 kcal such as -18 to -25 kcal.
- the target sequence consists of a region on the target nucleic acid which is complementary to the contiguous nucleotide region or sequence of the oligonucleotide of the invention.
- the target sequence is longer than the complementary sequence of a single oligonucleotide, and may, for example represent a preferred region of the target nucleic acid which may be targeted by several oligonucleotides of the invention.
- the oligonucleotide of the invention comprises a contiguous nucleotide region which is complementary to the target nucleic acid, such as a target sequence.
- the oligonucleotide comprises a contiguous nucleotide region of at least 10 nucleotides which is complementary to or hybridizes to a target sequence present in the target nucleic acid molecule.
- the contiguous nucleotide region (and therefore the target sequence) comprises of at least 10 contiguous nucleotides, such as 1 1 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 21 , 22, contiguous nucleotides, such as from 12-22, such as from 14-18 contiguous nucleotides.
- the target nucleic acid is a nucleic acid which encodes mammalian HTRA1 and may for example be a gene, a RNA, a mRNA, and pre-mRNA, a mature mRNA or a cDNA sequence.
- the target may therefore be referred to as an HTRA1 target nucleic acid.
- the target nucleic acid encodes an HTRA1 protein, in particular mammalian HTRA1 , such as human HTRA1 (See for example tables 1 & 2 which provides the mRNA and pre-mRNA sequences for human HTRA1 ).
- the target nucleic acid is selected from the group consisting of SEQ ID NO: 1 or 2 or naturally occurring variants thereof (e.g. sequences encoding a mammalian HTRA1 protein.
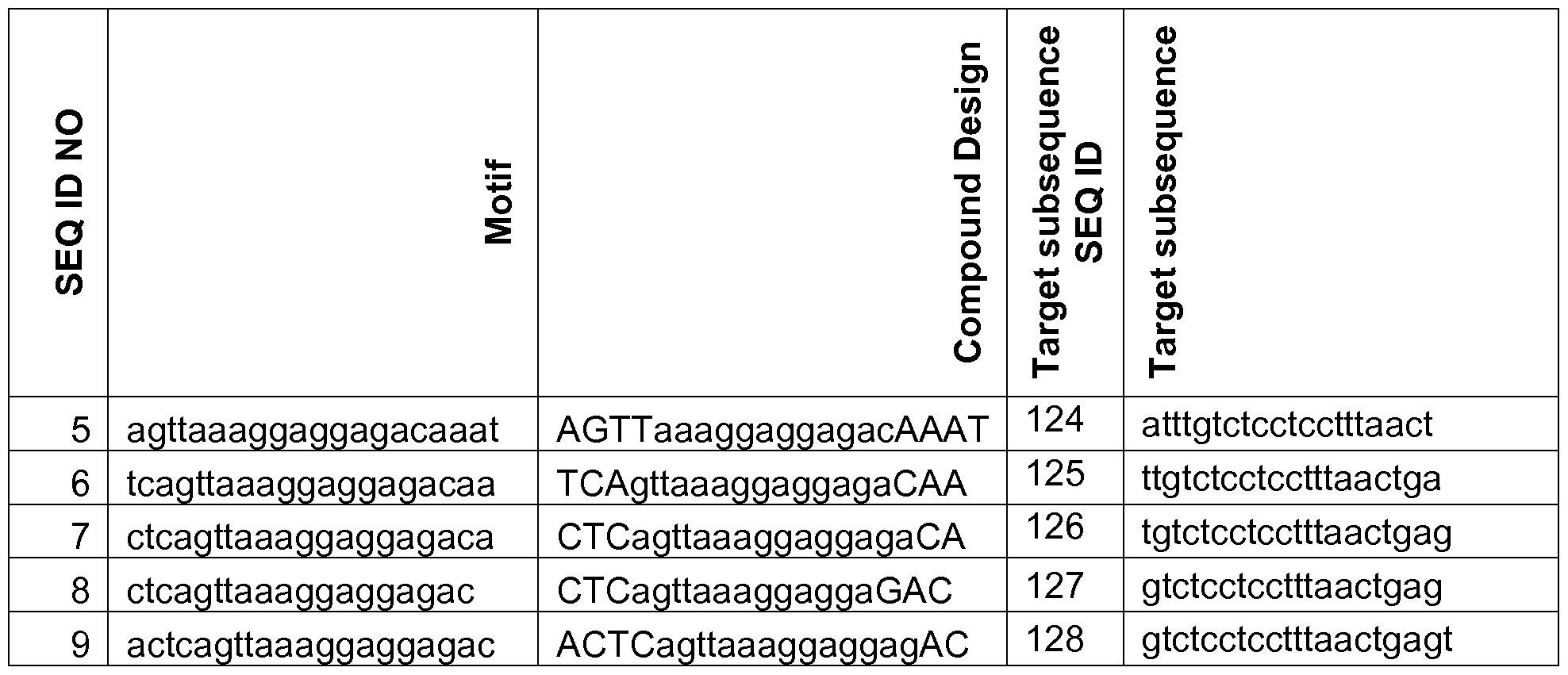
- the oligonucleotide or contiguous nucleotide region is fully complementary to, or only comprises one or two mismatches to an HTRA1 sub-sequence, such as a sequence selected from the group consisting of SEQ ID NO 113, 114, 115, 116, 117 or 231.
- the target sequence may be a sub-sequence of the target nucleic acid.
- the oligonucleotide or contiguous nucleotide region is fully complementary to, or only comprises one or two mismatches to an HTRA1 sub-sequence, such as a sequence selected from the group consisting of SEQ ID NO 124 - 230. In some embodiments the oligonucleotide or contiguous nucleotide region is fully complementary to, or only comprises one or two mismatches to an HTRA1 sub-sequence SEQ ID NO 231.
- Complementarity to the target or sub-sequence thereof is measured over the length of the oligonucleotide, or contiguous nucleotide region thereof.
- the target nucleic acid may, in some embodiments, be a RNA or DNA, such as a messenger RNA, such as a mature mRNA or a pre-mRNA.
- the target nucleic acid is a RNA or DNA which encodes mammalian HTRA1 protein, such as human HTRA1 , e.g. the human HTRA1 mRNA sequence, such as that disclosed as SEQ ID NO 1 (NM_002775.4, Gl:190014575). Further information on exemplary target nucleic acids is provided in tables 1 & 2.
- Fwd forward strand.
- the genome coordinates provide the pre-mRNA sequence (genomic sequence).
- the NCBI reference provides the mRNA sequence (cDNA sequence).
- naturally occurring variant refers to variants of HTRA1 gene or transcripts which originate from the same genetic loci as the target nucleic acid, but may differ for example, by virtue of degeneracy of the genetic code causing a multiplicity of codons encoding the same amino acid, or due to alternative splicing of pre-mRNA, or the presence of polymorphisms, such as single nucleotide polymorphisms, and allelic variants. Based on the presence of the sufficient complementary sequence to the oligonucleotide, the oligonucleotide of the invention may therefore target the target nucleic acid and naturally occurring variants thereof.
- the naturally occurring variants have at least 95% such as at least 98% or at least 99% homology to a mammalian HTRA1 target nucleic acid, such as a target nucleic acid selected form the group consisting of SEQ ID NO 1 ,2, 3 & 4.
- modulation of expression is to be understood as an overall term for an oligonucleotide’s ability to alter the amount of the target nucleic acid or target protein, such as e.g. the level of HTRA1 when compared to the amount of the target HTRA1 before administration of the oligonucleotide.
- modulation of expression may be determined by reference to a control experiment where the oligonucleotide of the invention is not administered.
- One type of modulation is an oligonucleotide’s ability to inhibit, down- regulate, reduce, suppress, remove, stop, block, prevent, lessen, lower, avoid or terminate expression of HTRA1 , e.g. by degradation of mRNA or blockage of transcription.
- the antisense oligonucleotide of the invention are capable of inhibiting, down-regulating, reduce, suppress, remove, stop, block, prevent, lessen, lower, avoid or terminate expression of HTRA1.
- a high affinity modified nucleoside is a modified nucleotide which, when incorporated into the oligonucleotide enhances the affinity of the oligonucleotide for its complementary target, for example as measured by the melting temperature (T m ).
- a high affinity modified nucleoside of the present invention preferably result in an increase in melting temperature between +0.5 to +12°C, more preferably between +1.5 to +10°C and most preferably between+3 to +8°C per modified nucleoside.
- Numerous high affinity modified nucleosides are known in the art and include for example, many 2’ substituted nucleosides as well as locked nucleic acids (LNA) (see e.g. Freier & Altmann; Nucl. Acid Res., 1997, 25, 4429-4443 and Uhlmann; Curr. Opinion in Drug Development, 2000, 3(2), 293-213).
- the oligomer of the invention may comprise one or more nucleosides which have a modified sugar moiety, i.e. a modification of the sugar moiety when compared to the ribose sugar moiety found in DNA and RNA.
- nucleosides with modification of the ribose sugar moiety have been made, primarily with the aim of improving certain properties of oligonucleotides, such as affinity and/or nuclease resistance.
- a 2’ sugar modified nucleoside is a nucleoside which has a substituent other than H or -OH at the 2’ position (2’ substituted nucleoside) or comprises a 2’ linked biradicle capable of forming a bridge between the 2’ carbon and a second carbon in the ribose ring, such as LNA (2’ - 4’ biradicle bridged) nucleosides.
- the 2’ modified sugar may provide enhanced binding affinity and/or increased nuclease resistance to the oligonucleotide.
- 2’ substituted modified nucleosides are 2’-0-alkyl-RNA, 2’-0-methyl-RNA, 2’- alkoxy-RNA, 2’-0-methoxyethyl-RNA (MOE), 2’-amino-DNA, 2’-Fluoro-RNA, and 2’-F-ANA nucleoside.
- 2’ substituted does not include 2’ bridged molecules like LNA.
- LNA Locked Nucleic Acids
- Non limiting, exemplary LNA nucleosides are disclosed in WO 99/014226, WO
- LNA nucleosides are beta-D-oxy-LNA, 6’-methyl-beta-D-oxy LNA such as (S)-6’-methyl-beta-D-oxy-LNA (ScET) and ENA.
- a particularly advantageous LNA is beta-D-oxy-LNA.
- the RNase H activity of an antisense oligonucleotide refers to its ability to recruit RNase H when in a duplex with a complementary RNA molecule.
- WO01/23613 provides in vitro methods for determining RNaseH activity, which may be used to determine the ability to recruit RNaseH.
- an oligonucleotide is deemed capable of recruiting RNase H if it, when provided with a complementary target nucleic acid sequence, has an initial rate, as measured in pmol/l/min, of at least 5%, such as at least 10% or more than 20% of the of the initial rate determined when using a oligonucleotide having the same base sequence as the modified oligonucleotide being tested, but containing only DNA monomers with
- the gap region is flanked by a 5’ flanking region (F) comprising one or more sugar modified nucleosides, advantageously high affinity sugar modified nucleosides, and by a 3’ flanking region (F’) comprising one or more sugar modified nucleosides, advantageously high affinity sugar modified nucleosides.
- the one or more sugar modified nucleosides in region F and F’ enhance the affinity of the oligonucleotide for the target nucleic acid ( i.e . are affinity enhancing sugar modified nucleosides).
- the one or more sugar modified nucleosides in region F and F’ are 2’ sugar modified nucleosides, such as high affinity 2’ sugar modifications, such as independently selected from LNA and 2’-MOE.
- the 5’ and 3’ most nucleosides of the gap region are DNA nucleosides, and are positioned adjacent to a sugar modified nucleoside of the 5’ (F) or 3’ (F’) region respectively.
- the flanks may further defined by having at least one sugar modified nucleoside at the end most distant from the gap region, i.e. at the 5’ end of the 5’ flank and at the 3’ end of the 3’ flank.
- Regions F-G-F’ form a contiguous nucleotide sequence.
- Antisense oligonucleotides of the invention, or the contiguous nucleotide sequence thereof, may comprise a gapmer region of formula F-G-F’.
- the overall length of the gapmer design F-G-F’ may be, for example 12 to 32 nucleosides, such as 13 to 24, such as 14 to 22 nucleosides, Such as from 14 to17, such as 16 to18 nucleosides.
- the gapmer oligonucleotide of the present invention can be represented by the following formulae:
- the overall length of the gapmer regions F-G-F’ is at least 12, such as at least 14 nucleotides in length.
- Regions F, G and F’ are further defined below and can be incorporated into the F-G-F’ formula.
- Region G is a region of nucleosides which enables the oligonucleotide to recruit RNaseH, such as human RNase H1 , typically DNA nucleosides.
- RNaseH is a cellular enzyme which recognizes the duplex between DNA and RNA, and enzymatically cleaves the RNA molecule.
- gapmers may have a gap region (G) of at least 5 or 6 contiguous DNA nucleosides, such as 5 - 16 contiguous DNA nucleosides, such as 6 - 15 contiguous DNA nucleosides, such as 7-14 contiguous DNA nucleosides, such as 8 - 12 contiguous DNA nucleotides, such as 8 - 12 contiguous DNA nucleotides in length.
- G gap region
- traditional gapmers have a DNA gap region, there are numerous examples of modified nucleosides which allow for RNaseH recruitment when they are used within the gap region.
- Modified nucleosides which have been reported as being capable of recruiting RNaseH when included within a gap region include, for example, alpha-L-LNA, C4’ alkylated DNA (as described in PCT/EP2009/050349 and Vester et a!., Bioorg. Med. Chem. Lett. 18 (2008) 2296 - 2300, both incorporated herein by reference), arabinose derived nucleosides like ANA and 2'F-ANA (Mangos et al. 2003 J. AM. CHEM. SOC. 125, 654-661 ), UNA
- UNA unlocked nucleic acid
- the modified nucleosides used in such gapmers may be nucleosides which adopt a 2’ endo (DNA like) structure when introduced into the gap region, i.e. modifications which allow for RNaseH recruitment).
- the DNA Gap region (G) described herein may optionally contain 1 to 3 sugar modified nucleosides which adopt a 2’ endo (DNA like) structure when introduced into the gap region.
- gapmers with a gap region comprising one or more 3’endo modified nucleosides are referred to as“gap-breaker” or“gap-disrupted” gapmers, see for example WO2013/022984.
- Gap-breaker oligonucleotides retain sufficient region of DNA nucleosides within the gap region to allow for RNaseH recruitment. The ability of gapbreaker
- Modified nucleosides used within the gap region of gap- breaker oligonucleotides may for example be modified nucleosides which confer a 3’endo confirmation, such 2’ -O-methyl (OMe) or 2’-0-MOE (MOE) nucleosides, or beta-D LNA nucleosides (the bridge between C2’ and C4’ of the ribose sugar ring of a nucleoside is in the beta conformation), such as beta-D-oxy LNA or ScET nucleosides.
- OMe 2’-O-methyl
- MOE 2-’-0-MOE
- beta-D LNA nucleosides the bridge between C2’ and C4’ of the ribose sugar ring of a nucleoside is in the beta conformation
- beta-D-oxy LNA or ScET nucleosides Gapmer - flanking regions, F and F’
- Region F’ is positioned immediately adjacent to the 3’ DNA nucleoside of region G.
- the 5’ most nucleoside of region F’ is a sugar modified nucleoside, such as a high affinity sugar modified nucleoside, for example a 2’ substituted nucleoside, such as a MOE nucleoside, or an LNA nucleoside.
- Region F is 1 - 8 contiguous nucleotides in length, such as 2-6, such as 3-4 contiguous nucleotides in length.
- the 5’ most nucleoside of region F is a sugar modified nucleoside.
- the two 5’ most nucleoside of region F are sugar modified nucleoside.
- the 5’ most nucleoside of region F is an LNA nucleoside.
- the two 5’ most nucleoside of region F are LNA nucleosides.
- the two 5’ most nucleoside of region F are 2’ substituted nucleoside nucleosides, such as two 3’ MOE nucleosides.
- the 5’ most nucleoside of region F is a 2’ substituted nucleoside, such as a MOE nucleoside.
- Region F’ is 2 - 8 contiguous nucleotides in length, such as 3-6, such as 4-5 contiguous nucleotides in length.
- the 3’ most nucleoside of region F’ is a sugar modified nucleoside.
- the two 3’ most nucleoside of region F’ are sugar modified nucleoside.
- the two 3’ most nucleoside of region F’ are LNA nucleosides.
- the 3’ most nucleoside of region F’ is an LNA nucleoside.
- the two 3’ most nucleoside of region F’ are 2’ substituted nucleoside nucleosides, such as two 3’ MOE nucleosides.
- the 3’ most nucleoside of region F’ is a 2’ substituted nucleoside, such as a MOE nucleoside. It should be noted that when the length of region F or F’ is one, it is advantageously an LNA nucleoside.
- region F and F’ independently comprises both LNA and a 2’ substituted modified nucleosides (mixed wing design).
- region F and F’ consists of only one type of sugar modified nucleosides, such as only MOE or only beta-D-oxy LNA or only ScET. Such designs are also termed uniform flanks or uniform gapmer design.
- all the nucleosides of region F or F’, or F and F’ are LNA nucleosides, such as independently selected from beta-D-oxy LNA, ENA or ScET nucleosides.
- region F consists of 1-5, such as 2-4, such as 3-4 such as 1 , 2, 3, 4 or 5 contiguous LNA nucleosides.
- all the nucleosides of region F and F’ are beta-D-oxy LNA nucleosides.
- all the nucleosides of region F or F’, or F and F’ are 2’ substituted nucleosides, such as OMe or MOE nucleosides.
- region F consists of 1 , 2, 3, 4, 5, 6, 7, or 8 contiguous OMe or MOE nucleosides.
- only one of the flanking regions can consist of 2’ substituted nucleosides, such as OMe or MOE nucleosides.
- the 5’ (F) flanking region that consists 2’ substituted nucleosides, such as OMe or MOE nucleosides whereas the 3’ (F’) flanking region comprises at least one LNA nucleoside, such as beta-D-oxy LNA nucleosides or cET nucleosides.
- the 3’ (F’) flanking region that consists 2’ substituted nucleosides, such as OMe or MOE nucleosides
- the 5’ (F) flanking region comprises at least one LNA nucleoside, such as beta-D-oxy LNA nucleosides or cET nucleosides.
- all the modified nucleosides of region F and F’ are LNA nucleosides, such as independently selected from beta-D-oxy LNA, ENA or ScET nucleosides, wherein region F or F’, or F and F’ may optionally comprise DNA nucleosides (an alternating flank, see definition of these for more details).
- all the modified nucleosides of region F and F’ are beta-D-oxy LNA nucleosides, wherein region F or F’, or F and F’ may optionally comprise DNA nucleosides (an alternating flank, see definition of these for more details).
- the 5’ most and the 3’ most nucleosides of region F and F’ are LNA nucleosides, such as beta-D-oxy LNA nucleosides or ScET nucleosides.
- the internucleoside linkage between region F and region G is a phosphorothioate internucleoside linkage. In some embodiments, the internucleoside linkage between region F’ and region G is a phosphorothioate internucleoside linkage. In some embodiments, the internucleoside linkages between the nucleosides of region F or F’, F and F’ are phosphorothioate internucleoside linkages.
- An LNA gapmer is a gapmer wherein either one or both of region F and F’ comprises or consists of LNA nucleosides.
- a beta-D-oxy gapmer is a gapmer wherein either one or both of region F and F’ comprises or consists of beta-D-oxy LNA nucleosides.
- treatment refers to both treatment of an existing disease (e.g. a disease or disorder as herein referred to), or prevention of a disease, i.e. prophylaxis. It will therefore be recognized that treatment as referred to herein may, in some embodiments, be prophylactic.
- the Htral oligonucleotide antagonist is an antisense oligonucleotide which targets HTRA1 , such as a mammalian HTRA1 , preferably human HTRA1.
- HTRA1 such as a mammalian HTRA1 , preferably human HTRA1.
- oligonucleotides targeting Htral which may be used in the present invention include those listed in the examples as Compound ID NO #5,1 - #112,1 and compound A and compound B.
- Preferred antisense oligonucleotide antagonists of HTRA1 for use in the present invention may be selected from the group of compounds of formula:
- compounds of the invention may be capable of inhibiting expression levels of HTRA1 protein by at least 50% in vitro using ARPE-19 cells.
- the examples provide assays which may be used to measure HTRA1 RNA or protein inhibition.
- the target modulation is triggered by the hybridization between a contiguous nucleotide sequence of the oligonucleotide and the target nucleic acid.
- the oligonucleotide of the invention comprises mismatches between the oligonucleotide and the target nucleic acid. Despite mismatches hybridization to the target nucleic acid may still be sufficient to show a desired modulation of HTRA1 expression.
- Reduced binding affinity resulting from mismatches may advantageously be compensated by increased number of nucleotides in the oligonucleotide and/or an increased number of modified nucleosides capable of increasing the binding affinity to the target, such as 2’ modified nucleosides, including LNA, present within the oligonucleotide sequence.
- An aspect of the present invention relates to antisense oligonucleotide which comprises a contiguous nucleotide region of 10 to 30 nucleotides in length with at least 90%
- HTRA1 target sequence such as fully complementary to an HTRA1 target sequence, e.g. a nucleic acid selected from the group consisting SEQ ID NO 1 , 2, 3 & 4.
- the oligonucleotide comprises a contiguous sequence which is at least 90% complementary, such as at least 91 %, such as at least 92%, such as at least 93%, such as at least 94%, such as at least 95%, such as at least 96%, such as at least 97%, such as at least 98%, or 100% complementary with a region of the target nucleic acid.
- the oligonucleotide of the invention, or a contiguous nucleotide sequence thereof is fully complementary (100% complementary) to a region of the target nucleic acid, or in some embodiments may comprise one or two mismatches between the oligonucleotide and the target nucleic acid.
- the Htral oligonucleotide antagonist of the present invention is an oligonucleotide targeting a mammalian HTRA1 nucleic acid, i.e. are capable of inhibiting the expression of HTRA1 and may be used to treat or prevent diseases related to the functioning of the HTRA1.
- the oligonucleotides targeting HTRA1 are antisense oligonucleotides, i.e. are complementary to their HTRA1 nucleic acid target.
- the oligonucleotide may be in the form of a
- the antisense oligonucleotides may comprise a contiguous nucleotide sequence of 10 - 30 nucleotides in length with at least 90% complementarity, such as fully complementary to a mammalian HTRA1 nucleic acid, such as SEQ ID NO 1 , SEQ ID NO 2, SEQ ID NO 3 or SEQ ID NO 4.
- the antisense oligonucleotide may be an LNA antisense oligonucleotides, such as LNA gapmer oligonucleotides, which comprise a contiguous nucleotide sequence of 10 - 30 nucleotides in length with at least 90% complementarity, such as fully complementary to a HTRA1 nucleic acid, such as a sequence selected from the group consisting of SEQ ID NO 1 , SEQ ID NO 2, SEQ ID NO 3 or SEQ ID NO 4.
- LNA antisense oligonucleotides such as LNA gapmer oligonucleotides, which comprise a contiguous nucleotide sequence of 10 - 30 nucleotides in length with at least 90% complementarity, such as fully complementary to a HTRA1 nucleic acid, such as a sequence selected from the group consisting of SEQ ID NO 1 , SEQ ID NO 2, SEQ ID NO 3 or SEQ ID NO 4.
- the antisense oligonucleotide may comprise a contiguous nucleotide region of at 10 - 30, such as 12 - 22, nucleotides, wherein the contiguous nucleotide region is at least 90% such as 100% complementary to SEQ ID NO 113.
- the antisense oligonucleotide may be of 10 - 30 nucleotides in length, wherein said antisense oligonucleotide comprises a contiguous nucleotide region of 10 - 30, such as 12 - 22, nucleotides which are at least 90% such as 100% complementarity to SEQ ID NO 1 13.
- the reverse complement of SEQ ID NO 1 13 is shown in SEQ ID NO 119.
- the antisense oligonucleotide may comprise a contiguous nucleotide region of at 10 - 30, such as 12 - 22, nucleotides, wherein the contiguous nucleotide region is at least 90% such as 100% complementary to SEQ ID NO 114.
- the antisense oligonucleotide of 10 - 30 nucleotides in length may comprise a contiguous nucleotide region of 10 - 30, such as 12 - 22 nucleotides which are at least 90% such as 100% complementarity to SEQ ID NO 114.
- the reverse complement of SEQ ID NO 114 is SEQ ID NO 120.
- the antisense oligonucleotide may comprise a contiguous nucleotide region of at 10 - 30, such as 12 - 22, nucleotides, wherein the contiguous nucleotide region is at least 90% such as 100% complementary to SEQ ID NO 115.
- the antisense oligonucleotide of 10 - 30 nucleotides in length may comrpise a contiguous nucleotide region of 10 - 30, such as 12 - 22 nucleotides which are at least 90% such as 100% complementarity to SEQ ID NO 115.
- the reverse complement of SEQ ID NO 115 is shown in SEQ ID NO 121.
- the antisense oligonucleotide may comprise a contiguous nucleotide region of at 10 - 30, such as 12 - 22, nucleotides, wherein the contiguous nucleotide region is at least 90% such as 100% complementary to SEQ ID NO 116.
- the antisense oligonucleotide of 10 - 30 nucleotides in length may comprise a contiguous nucleotide region of 10 - 30, such as 12 - 22 nucleotides which are at least 90% such as 100% complementarity to SEQ ID NO 116.
- the reverse complement of SEQ ID NO 116 is SEQ ID NO 122.
- the antisense oligonucleotide may comprise a contiguous nucleotide region of at 10 - 30, such as 12 - 22, nucleotides, wherein the contiguous nucleotide region is at least 90% such as 100% complementary to SEQ ID NO 1 17.
- the antisense oligonucleotide of 10 - 30 nucleotides in length may comprise a contiguous nucleotide region of 10 - 30, such as 12 - 22 nucleotides which are at least 90% such as 100% complementarity to SEQ ID NO 1 17.
- the reverse complement of SEQ ID NO 1 17 is SEQ ID NO 123.
- the antisense oligonucleotide comprises a contiguous nucleotide region of at least 10, or at least 12, at least 13, or at least 14 or at least 15 or at least 16 contiguous nucleotides present SEQ ID NO 1 18: 5’ CTTCTTCTATCTACGCATTG 3’.
- the reverse complement of SEQ ID NO 1 18 is SEQ ID NO 231 : C AAT G C GT AG AT AG AAG AAG .
- the invention provides an antisense oligonucleotide which comprises a contiguous nucleotide region of at least 10, or at least 12, at least 13, or at least 14 or at least 15 or at least 16 contiguous nucleotides complementary to SEQ ID NO 231 .
- the invention refers to an antisense oligonucleotide which comprises a contiguous nucleotide region of at least 10, or at least 12, or at least 13, or at least 14 or at least 15 or 16 contiguous nucleotides present SEQ ID NO 67.
- the invention provides an antisense oligonucleotide which comprises a contiguous nucleotide region of at least 10, or at least 12, or at least 13, or at least 14 or at least 15 or 16 contiguous nucleotides present SEQ ID NO 86.
- the invention provides an antisense oligonucleotide which comprises a contiguous nucleotide region of at least 10, or at least 12, or at least 13, or at least 14 or at least 15 or at least 16 or at least 17 or 18 contiguous nucleotides present SEQ ID NO 73.
- the invention provides an antisense oligonucleotide which comprises a contiguous nucleotide region of at least 10, or at least 12, or at least 13, or at least 14 or at least 15 or 16 contiguous nucleotides complementary to SEQ ID NO 186.
- the invention provides an antisense oligonucleotide which comprises a contiguous nucleotide region of at least 10, or at least 12, or at least 13, or at least 14 or at least 15 or 16 contiguous nucleotides complementary to SEQ ID NO 205.
- the invention provides an antisense oligonucleotide which comprises a contiguous nucleotide region of at least 10, or at least 12, or at least 13, or at least 14 or at least 15 or at least 16 or at least 17 or 18 contiguous nucleotides complementary to SEQ ID NO 192.
- the invention refers to an oligonucleotide comprising or consisting of an oligonucleotide selected from the group consisting of: and
- the antisense oligonucleotide of the invention is capable of modulating the expression of the target by inhibiting or down-regulating it. Preferably, such modulation produces an inhibition of expression of at least 20% compared to the normal expression level of the target, such as at least 30%, 40%, 50%, 60%, 70%, 80%, or 90% inhibition compared to the normal expression level of the target.
- compounds of the invention may be capable of inhibiting expression levels of HTRA1 mRNA by at least 60% or 70% in vitro using ARPE-19 cells. In some embodiments compounds of the invention may be capable of inhibiting expression levels of HTRA1 mRNA by at least 60% or 70% in vitro using ARPE-19 cells.
- compounds of the invention may be capable of inhibiting expression levels of HTRA1 protein by at least 50% in vitro using ARPE-19 cells.
- the examples provide assays which may be used to measure HTRA1 RNA or protein inhibition.
- the target modulation is triggered by the hybridization between a contiguous nucleotide sequence of the oligonucleotide and the target nucleic acid.
- the oligonucleotide of the invention comprises mismatches between the oligonucleotide and the target nucleic acid. Despite mismatches hybridization to the target nucleic acid may still be sufficient to show a desired modulation of HTRA1 expression.
- Reduced binding affinity resulting from mismatches may advantageously be compensated by increased number of nucleotides in the oligonucleotide and/or an increased number of modified nucleosides capable of increasing the binding affinity to the target, such as 2’ modified nucleosides, including LNA, present within the oligonucleotide sequence.
- An aspect of the present invention relates to an antisense oligonucleotide which comprises a contiguous nucleotide region of 10 to 30 nucleotides in length with at least 90%
- HTRA1 target sequence such as fully complementary to an HTRA1 target sequence, e.g. a nucleic acid selected from the group consisting SEQ ID NO 1 , 2, 3 & 4.
- the oligonucleotide comprises a contiguous sequence which is at least 90% complementary, such as at least 91 %, such as at least 92%, such as at least 93%, such as at least 94%, such as at least 95%, such as at least 96%, such as at least 97%, such as at least 98%, or 100% complementary with a region of the target nucleic acid.
- the oligonucleotide of the invention, or a contiguous nucleotide sequence thereof is fully complementary (100% complementary) to a region of the target nucleic acid, or in some embodiments may comprise one or two mismatches between the oligonucleotide and the target nucleic acid.
- the oligonucleotide, or a contiguous nucleotide sequence of at least 12 nucleotides thereof is at least 90% complementary, such as fully (or 100%)
- the oligonucleotide, or a contiguous nucleotide sequence of at least 12 nucleotides thereof is at least 90% complementary, such as fully (or 100%)
- the oligonucleotide, or a contiguous nucleotide sequence of at least 12 nucleotides thereof is at least 90% complementary, such as fully (or 100%)
- the oligonucleotide, or a contiguous nucleotide sequence of at least 13 nucleotides thereof is at least 90% complementary, such as fully (or 100%)
- an ocular disease such as macular degeneration, such as wet or dry age related macular degeneration (e.g.
- the invention relates to the use of an HTRA1 oligonucleotide antagonist for the treatment or prophylaxis of dry age-related macular degeneration, such as geographic atrophy, in
- HTRA1 oligonucleotide antagonist for the combined use of the HTRA1 oligonucleotide antagonist and a further therapeutic agent which is a protein therapeutic, and antibody therapeutic (such as a monoclonal antibody, antibody fragment or antibody fusion protein) or a peptide therapeutic.
- a further therapeutic agent which is a protein therapeutic, and antibody therapeutic (such as a monoclonal antibody, antibody fragment or antibody fusion protein) or a peptide therapeutic.
- VEGF is a well-characterised signal protein which stimulates angiogenesis.
- the VEGF antagonist is an anti-VEGF antibody therapeutic such as ranibizumab (Lucentis®) or bevacizumab (Avastin®).
- anti-VEGF antibody inhibitors of VEGF
- intravitreal injection see US9220631 for example.
- anti-VEGF antobodies are disclosed in W096/30046, W02006/047325 & W02008/063932.
- Bevacizumab comprises a light chain immunoglobulin of sequence SEQ C (SEQ ID NO 236), and a heavy chain immunoglobulin of SEQ D (SEQ ID NO 237).
- the further therapeutic agent is a small molecule inhibitor of VEGF, such as a molecule selected from the group consisting of cediranib, lapatinib, sunitinib, sorafenib, axitinib, and pazopanib (these are anti-angiogenic drugs, which are used in cancer treatment).
- VEGF small molecule inhibitor of VEGF
- the further therapeutic agent is an antagonist of complement factor D (CFD).
- anti-CFD antibody antibody antagonist of complement factor D
- lampalizumab lampalizumab
- Anti-CFD antibodies may be administered via e.g. monthly of bimonthly to the eye via intravitreal injection.
- lampalizumab comprises a light chain immunoglobulin of sequence SEQ G SEQ ID NO 240), and a heavy chain immunoglobulin of SEQ H (SEQ ID NO 241 ).
- the further therapeutic agent is a small molecule inhibitor of complement factor D, such as a small molecule selected from the group consisting of ACH-4471 , ACH-5228 or ACH-5548 (as disclosed in US10106563B2, US10092584B2, US1001 1612B2 &
- W015038939 and W015168635 disclose antisense oligonucleotides targeting the human complement factor B pre-mRNA, and GalNAc conjuagtes thereof.
- Antisense oligonucleotides targeting the complement factor pre-mRNA are in clinical devlopement:
- the further therapeutic agent is lONIS-FB-Lrx, also known as ISIS 696844.
- lONIS-FB-Lrx is disclosed in W015168635 as ISIS 696844 and comprises an antisense oligonucleotide of sequence: ATCCCACGCCCCTGTCCAGC (SEQ ID NO 243).
- the IONIS- FB-Lrx antisense oligonucleotide has a gapmer 5-10-5:
- O-methoxyethyl (MOE) RNA nucleoside each lower case letter is a DNA nucleoside, all cytosines are 5-methyl cytosine (designated by a preceding superscript m ), and the
- internucleoside linkages are phosphorothioate (designated a subscript s ).
- the 5’ nucleoside (A) is conjugated to a trivalent conjugate moiety via a phosphate bond.
- the chemical formula of lONIS-FB-Lrx is presented below:
- lONIS-FB-Lrx may be administered to the subject via for example subcutaneous, intravenous, or intramuscular injection or infusion.
- the presence of the GalNAc conjugate enhances uptake of the antisense oligonucleotide into hepatocytes.
- the HTRA1 antagonist is an antisense oligonucleotide of formula wherein capital letters represent beta-D- oxy LNA nucleosides, lower case letters are DNA nucleosides, subscript s represents a phosphorothioate internucleoside linkage, and m C represent 5 methyl cytosine beta-D-oxy LNA nucleosides, and the further therapeutic agent is lONIS-FB-Lrx.
- the HTRA1 antagonist is an antisense oligonucleotide of formula
- LNA nucleosides lower case letters are DNA nucleosides, subscript s represents a phosphorothioate internucleoside linkage, and m C represent 5 methyl cytosine beta-D-oxy LNA nucleosides, and the further therapeutic agent is lONIS-FB-Lrx.
- the HTRA1 antagonist is an antisense oligonucleotide of formula
- the HTRA1 antagonist is an antisense oligonucleotide of formula wherein capital letters represent beta-D-oxy LNA nucleosides, lower case letters are DNA nucleosides, subscript s represents a phosphorothioate internucleoside linkage, and the further therapeutic agent is lONIS-FB-Lrx.
- the further therapeutic agent is an antagonist of complement factor P, such as an anti-CFP antibody, such as CLG-561 or NM9401 .
- the further therapeutic agent is CLG-561 (also written as CLG561 ), which is a monoclonal antibody in clinical development for use in combination with LFG316 (Tesidolumab, an anti-complement factor C5 antibody). CLG-561 may be administered via intravitreal injection monthly or bimonthly.
- the further therapeutic agent is an antibody targeting CFP, such as NM9401 (US8664362): which comprises a heavy chain shown in SEQ 244 and a light chain shown in SEQ 245:
- the further therapeutic agent is an antagonist or inhibitor (used interchangeable) of complement factor C3, such as APL-2 or CB2782.
- APL-2 is a pegulated peptide conjugate that binds specifically to C3 and C3b to inhibit C3 cleavage and activation into C3a and C3b of formula (See WO2014078731 &
- CB2782 is a novel protease derived from human membrane type serine protease 1 (MTSP-1 ) that selectively cleaves C3 and has been shown to completely inhibit C3 (US9290757 and US9359598).
- the further therapeutic agent is a modified MTSP-1 protease selected from SEQ ID Nos 252, 253, 254 and 255 (as disclosed in EP2402437).
- an anti-complement factor C5 aptamer such as avacincaptad pegol (Zimura®, also known as ARC1905).
- the further therapeutic agent is Tesidolumab, which is in clinical development (NCT02515942) via administration via intravitreal injection.
- Tesidolumab comprises a heavy and light chain immunoglobulin of SEQ J (SEQ ID NO 246) and SEQ K (SEQ ID NO 247):
- the further therapeutic agent is Eculizumab, which is a humanised monoclonal (lgG2/4K) antibody (WO2017/12375) comprising a light chain of SEQ L (SEQ ID NO 248), and a heavy chain of SEQ M (SEQ ID NO 249).
- Eculizumab is a humanised monoclonal (lgG2/4K) antibody (WO2017/12375) comprising a light chain of SEQ L (SEQ ID NO 248), and a heavy chain of SEQ M (SEQ ID NO 249).
- the further therapeutic agent is avacincaptad pegol (Zimura®, also known as ARC1905).
- Avacincaptad pegol (CAS-1491 144-00-3) is as Anti-C5 aptamer being developed by Ophthotech (WO 2007103549) (NCT02686658):
- Avacincaptad pegol comprises the aptamer sequence SEQ N (SEQ ID NO 250): According to Toxnet, Avacincaptad pegol chemical name is Poly(oxy-1 ,2-ethanediyl), alpha- hydro-omega-methoxy-, 5'-ether with RNA ((2'-deoxy-2'-fluoro)C-Gm-(2'-deoxy-2'-fluoro)C-(2'- deoxy-2'-fluoro)C-G-(2'-deoxy-2'-fluoro)C-Gm-Gm-(2'-deoxy-2'-fluoro)U-(2'
- PGDF Anti-platelet derived growth factor
- Pegpleranib has the formula (sequence):
- an antibody that binds to has a dissociation constant (KD) of or i 3 ⁇ 4 0.001 nM (e.g., 10-8 M or less, e.g., from 10-8 M to 10-13 M, e.g., from 10-9 M to 10-13 M).
- KD dissociation constant
- An antibody is said to“specifically bind” to when the antibody has a KD of 1 mM or less.
- the antibody targets the human target.
- an anti antibody binds to an epitope of that is conserved among from different species.
- antibody herein is used in the broadest sense and encompasses various antibody structures, including but not limited to monoclonal antibodies, polyclonal antibodies,
- multispecific antibodies e.g., bispecific antibodies
- antibody fragments so long as they exhibit the desired antigen-binding activity.
- an“antibody fragment” refers to a molecule other than an intact antibody that comprises a portion of an intact antibody that binds the antigen to which the intact antibody binds.
- antibody fragments include but are not limited to Fv, Fab, Fab', Fab’-SH, F(ab')2; diabodies; linear antibodies; single-chain antibody molecules (e.g., scFv, and scFab); single domain antibodies (dAbs); and multispecific antibodies formed from antibody fragments.
- the invention relates to the use of oligonucleotides, such as the HTRA1 antagonist or antisense oligonucleotide therapeutic, in the treatment of diseases or disorders selected from eye disorders, such as macular degeneration, including age related macular degeneration (AMD), such as dry AMD or wet AMD, and diabetic retinopathy.
- AMD age related macular degeneration
- the oligonucleotide conjugates or pharmaceutical compositions of the invention may be for use in the treatment of geographic atrophy or intermediate dAMD or dAMD.
- the HTRA1 antagonist/antisense oligonucleotide therapeutic according to the invention is typically administered in an effective amount.
- Administration is typically via intravitreal injection, such as in an ophthalmic solution comprising a dose of (unit dose), for example 5pg - 200pg per eye, such 10pg - 100pg, such as 20pg - 50pg per eye.
- the volume of ophthalmic solution administered to each eye is, for example about 25mI - about 100mI, such as about 50mI.
- An ophthalmic solution is a solution which comprises the Htral antagonist or antisense oligonucleotide therapeutic, such as the HTRA1 oligonucleotide antagonist dissolved in a solvent which is compatible with administration via injection into the eye, such as an intravitreal injection.
- the solvent is suitably a buffered aqueous solution, such as phosphate buffered saline, e.g. of pH about 7 - about 7.5, such as about pH 7.4.
- Other ophthalmic acceptable buffered solutions may be used.
- the ophthalmic solution is sterile.
- the solvent is suitably a buffered aqueous solution, such as phosphate buffered saline, e.g. of pH about 7 - about 7.5, such as about 7.4.
- the dosage interval i.e. the period of time between consecutive dosings is at least monthy, such as at least bi monthly or at least once every three months.
- An antisense oligonucleotide selected from the group selected from TTCtatctacgcaTTG (SEQ ID NO 67), CTTCttctatctacgcAT (SEQ ID NO 73), and TACTttaatagcTCAA (SEQ ID NO 86), TATttacctggtTGTT (SEQ ID NO 232 ) AtATttacctgGTTgTT (SEQ ID NO 233), wherein capital letters represent a LNA nucleosides, LNA C are optionally 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are
- phosphorothioate internucleoside linkages for use in the treatment of geographic atrophy in a subject, wherein the treatment further comprises administering a further therapeutic agent to the subject.
- oligonucleotide is , wherein capital letters represent beta-D-oxy LNA nucleosides, lower case letters are DNA nucleosides, subscript s represents a phosphorothioate internucleoside linkage, and m C represent 5 methyl cytosine beta-D-oxy LNA nucleosides, and m c represents 5 methyl cytosine DNA nucleosides.
- oligonucleotide is , wherein capital letters represent beta-D-oxy LNA nucleosides, lower case letters are DNA nucleosides, subscript s represents a phosphorothioate internucleoside linkage, and m C represent 5 methyl cytosine beta-D-oxy LNA nucleosides.
- oligonucleotide is wherein capital letters
- a method for the treatment of geographic atrophy in a subject in need of treatment comprising administering an effective amount of the antisense oligonucleotide according to any one of embodiments 1 - 6 to said subject, and further administering an effective amount of a further therapeutic agent, to the subject.
- VEGF vascular endothelial growth factor
- VEGF antagonist therapeutic is a small molecule inhibitor of VEGF such as a molecule selected from the group consisting of cediranib, lapatinib, sunitinib, sorafenib, axitinib, and pazopanib.
- VEGF antagonist therapeutic is a recombinant fusion protein VEGF inhibitor such as aflibercept.
- antisense oligonucleotide use or method according to claim 15, wherein the further therapeutic agent is an anti-CFD antibody therapeutic such as lampalizumab.
- a small molecule inhibitor of complement factor D such as a molecule selected from the group consisting of ACH-4447, ACH-5228 & ACH-5548.
- Vitreous sample were added to a 0.5 Precellyses tubes (CK14_0.5ml, Bertin Technologies) were lysed and homogenized in 1/4x RIPA lysis buffer (20-188, Milipore) with protease inhibitors (Complete EDTA-free Proteases-lnhibitor Mini, 1 1 836 170 001 , Roche).
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Genetics & Genomics (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- General Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biomedical Technology (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Biotechnology (AREA)
- Molecular Biology (AREA)
- Biochemistry (AREA)
- Epidemiology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Virology (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Physics & Mathematics (AREA)
- Biophysics (AREA)
- Medicinal Chemistry (AREA)
- Plant Pathology (AREA)
- Microbiology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
The present invention relates to the use of highly effective Htra1 oligonucleotide antagonists for treatment of macular degeneration, in combination with a further therapeutic agent, such as an antiangiogenic agent or an antagonist of the complement pathway.
Description
COMBINATION THERAPY FOR TREATMENT OF MACULAR DEGENERATION
FIELD OF INVENTION
The present invention relates to the use of highly effective Htral oligonucleotide antagonists for treatment of macular degeneration, in combination with a further therapeutic agent, such as an antiangiogenic agent or an antagonist of the complement pathway.
BACKGROUND
The standard of care for wet age-related macular degeneration (wAMD) are anti-angiogenic drugs, such as anti-VEGF antibody therapeutics such as LUCENTIS™(ranibizumab). Other treatments of wAMD include laser therapy and photodynamic laser therapy.
There are no approved treatments for dry age-related macular degeneration (dAMD), such as geographic atrophy (GA). Numerous antagonists of the complement system are being developed for the treatment of macular degeneration (reviewed in Ricklin and Lambris, Semin Immunol. Author manuscript; available in PMC 2017 June 16.). Combination treatment for treatment of geographic atrophy include the combination of CLG561 (anti factor B antibody) with LFG316 (Complement factor 5 antagonist). W02013/055998 relates to anti-Htra1 antibodies and their use in combination therapy for example with anti-VEGF agents.
WO2018/00205 and PCT/2018/064221 describe antisense oligonucleotides targeting HTRA1 , and their use in treatment of macular degeneration via an intraocular injection.
OBJECT OF THE INVENTION
The present invention relates to the therapeutic treatment of macular degeneration, such as age-related macular degeneration, such as dryAMD and/or geographic atrophy, by the combined use of certain antisense oligonucleotide antagonists of HTRA1 with a further therapeutic agent, such as an antiangiogenic agent or an antagonist of the complement pathway.
STATEMENT OF THE INVENTION
The invention provides for antisense oligonucleotide selected from the group selected from TTCtatctacgcaTTG (SEQ ID NO 67), CTTCttctatctacgcAT (SEQ ID NO 73), and
TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all
internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering a further therapeutic agent to the subject.
In some embodiments, the further therapeutic agent is an anti-angiogenic agent, such as a VEGF antagonist, or is an inhibitor or the complement system, such as an antagonist of a complement component, or an anti-platelet derived growth factor.
In some embodiments, the further therapeutic agent is a complement system antagonist, such as an antagonist of the classical complement pathway or an antagonist of the alternative complement pathway. In some embodiments, the further therapeutic agent is selected from the group consisting of complement factor D antagonist, a complement factor B antagonist, antagonist complement factor C5, antagonist complement factor C3, antagonist complement factor P (properdin), an antagonist of complement factor I, and an antagonist of complement factor H.
In some embodiments, the further therapeutic agent is an anti-platelet derived growth factor.
The invention provides for antisense oligonucleotide of formula TTCtatctacgcaTTG (SEQ ID NO 67), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering a further therapeutic agent to the subject.
The invention provides for antisense oligonucleotide of formula CTTCttctatctacgcAT (SEQ ID NO 73) wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are
phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering a further therapeutic agent to the subject.
The invention provides for antisense of formula TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are
phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering a further therapeutic agent to the subject.
The invention provides for the use of an antisense oligonucleotide of formula TTCtatctacgcaTTG (SEQ ID NO 67), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages, in the manufacture of a medicament for the treatment of dry age-related macular degeneration such as geographic atrophy, wherein the medicament is for use in combination treatment with a further therapeutic.
The invention provides for the use of an antisense oligonucleotide of formula
CTTCttctatctacgcAT (SEQ ID NO 73), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages, in the manufacture of a medicament for the treatment dry age-related macular degeneration such as geographic atrophy, wherein the medicament is for use in combination treatment with a further therapeutic.
The invention provides for the use of an antisense oligonucleotide of formula
TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages, in the manufacture of a medicament for the treatment of dry age-related macular degeneration such as geographic atrophy, wherein the medicament is for use in combination treatment with a further therapeutic.
The invention provides for antisense oligonucleotide selected from the group selected from TTCtatctacgcaTTG (SEQ ID NO 67), CTTCttctatctacgcAT (SEQ ID NO 73), and
TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering a anti-angiogenic therapeutic agent to the subject.
The invention provides for antisense oligonucleotide selected from the group selected from TTCtatctacgcaTTG (SEQ ID NO 67), CTTCttctatctacgcAT (SEQ ID NO 73), and
TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all
internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering a VEGF antagonist, to the subject.
The invention provides for antisense oligonucleotide selected from the group selected from TTCtatctacgcaTTG (SEQ ID NO 67), CTTCttctatctacgcAT (SEQ ID NO 73), and
TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist of the complement system to the subject.
The invention provides for antisense oligonucleotide selected from the group selected from TTCtatctacgcaTTG (SEQ ID NO 67), CTTCttctatctacgcAT (SEQ ID NO 73), and
TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist complement factor D to the subject.
The invention provides for antisense oligonucleotide selected from the group selected from TTCtatctacgcaTTG (SEQ ID NO 67), CTTCttctatctacgcAT (SEQ ID NO 73), and
TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist complement factor B to the subject.
The invention provides for antisense oligonucleotide selected from the group selected from TTCtatctacgcaTTG (SEQ ID NO 67), CTTCttctatctacgcAT (SEQ ID NO 73), and
TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject,
wherein the treatment further comprises administering an antagonist complement factor C5 to the subject.
The invention provides for antisense oligonucleotide of formula TTCtatctacgcaTTG (SEQ ID NO 67), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an anti-angiogenic agent to the subject.
The invention provides for antisense oligonucleotide of formula TTCtatctacgcaTTG (SEQ ID NO 67), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering a VEGF antagonist to the subject.
The invention provides for antisense oligonucleotide of formula TTCtatctacgcaTTG (SEQ ID NO 67), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist of the complement system to the subject.
The invention provides for antisense oligonucleotide of formula TTCtatctacgcaTTG (SEQ ID NO 67, wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are
phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist complement factor D to the subject.
The invention provides for antisense oligonucleotide of formula TTCtatctacgcaTTG (SEQ ID NO 67), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist complement factor B to the subject.
The invention provides for antisense oligonucleotide of formula TTCtatctacgcaTTG (SEQ ID NO 67), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist complement factor C5 to the subject.
The invention provides for antisense oligonucleotide of formula CTTCttctatctacgcAT (SEQ ID NO 73), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an anti-angiogenic agent to the subject.
The invention provides for antisense oligonucleotide of formula CTTCttctatctacgcAT (SEQ ID NO 73), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering a VEGF antagonist to the subject.
The invention provides for antisense oligonucleotide of formula CTTCttctatctacgcAT (SEQ ID NO 73), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist of the complement system to the subject.
The invention provides for antisense oligonucleotide of formula CTTCttctatctacgcAT (SEQ ID NO 73), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are
phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist complement factor D to the subject.
The invention provides for antisense oligonucleotide of formula CTTCttctatctacgcAT (SEQ ID NO 73), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist complement factor B to the subject.
The invention provides for antisense oligonucleotide of formula CTTCttctatctacgcAT (SEQ ID NO 73), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist complement factor C5 to the subject.
The invention provides for antisense oligonucleotide of formula TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an anti-angiogenic agent to the subject.
The invention provides for antisense oligonucleotide of formula TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering a VEGF antagonist to the subject.
The invention provides for antisense oligonucleotide of formula TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist of the complement system to the subject.
The invention provides for antisense oligonucleotide of formula TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-
methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist complement factor D to the subject.
The invention provides for antisense oligonucleotide of formula TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist complement factor B to the subject.
The invention provides for antisense oligonucleotide of formula TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5- methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist complement factor C5 to the subject.
The invention provides for antisense oligonucleotide antagonist of HTRA1 for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an anti-angiogenic agent to the subject.
The invention provides for antisense oligonucleotide antagonist of HTRA1 for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist of VEGF to the subject.
The invention provides for antisense oligonucleotide antagonist of HTRA1 for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist of the complement system to the subject.
The invention provides for antisense oligonucleotide antagonist of HTRA1 for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist of complement factor D to the subject.
The invention provides for antisense oligonucleotide antagonist of HTRA1 for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist of complement factor B to the subject.
The invention provides for antisense oligonucleotide antagonist of HTRA1 for use in the treatment of dry age-related macular degeneration such as geographic atrophy in a subject, wherein the treatment further comprises administering an antagonist of complement factor C5 to the subject.
The invention provides for the use of an antisense oligonucleotide of formula
TTCtatctacgcaTTG (SEQ ID NO 67), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages, in the manufacture of a medicament for the treatment of dry age-related macular degeneration such as geographic atrophy, wherein the medicament is for use in combination treatment with a further therapeutic.
The invention provides for the use of an antisense oligonucleotide of formula
CTTCttctatctacgcAT (SEQ ID NO 73), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages, in the manufacture of a medicament for the treatment dry age-related macular degeneration such as geographic atrophy, wherein the medicament is for use in combination treatment with a further therapeutic.
The invention provides for the use of an antisense oligonucleotide of formula
TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages, in the manufacture of a medicament for the treatment of dry age-related macular degeneration such as geographic atrophy, wherein the medicament is for use in combination treatment with a further therapeutic.
The invention provides for a method for the treatment of dry age-related macular degeneration such as geographic atrophy in a subject in need of treatment, said method
comprising administering an effective amount of the antisense oligonucleotide TTCtatctacgcaTTG (SEQ ID NO 67), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages, to said subject, and further administering an effective amount of a further therapeutic agent, to the subject.
The invention provides for a method for the treatment of dry age-related macular degeneration such as geographic atrophy in a subject in need of treatment, said method comprising administering an effective amount of the antisense oligonucleotide
CTTCttctatctacgcAT (SEQ ID NO 73) wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages, to said subject, and further administering an effective amount of a further therapeutic agent, to the subject.
The invention provides for a method for the treatment of dry age-related macular degeneration such as geographic atrophy in a subject in need of treatment, said method comprising administering an effective amount of the antisense oligonucleotide
TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages, to said subject, and further administering an effective amount of a further therapeutic agent, to the subject.
BRIEF DESCRIPTION OF FIGURES
Figure 1. A library of n=231 HTRA1 LNA oligonucleotides were screened in U251 cell lines at 5 mM. The residual HTRA1 mRNA expression level was measured by qPCR and is shown as % of control (PBS-treated cells). n=10 oligos located between position 531 13 - 53384 were relatively active.
Figure 2. A library of n=210 HTRA1 LNA oligonucleotides were screened in U251 cell lines at 5 mM. The residual HTRA1 mRNA expression level was measured by qPCR and is shown as % of control (PBS-treated cells). n=33 oligos located between position 531 13 - 53384 were relatively active.
Figure 3. A library of n=305 HTRA1 LNA oligonucleotides were screened in U251 and ARPE19 cell lines at 5 and 25 mM, respectively. The residual HTRA1 mRNA expression level was measured by qPCR and is shown as % of control (PBS-treated cells). n=95 oligos located between position 531 13 - 53384 were relatively active in comparison to the rest.
Figure 4. Dose response of HTRA1 mRNA level upon treatment of human primary RPE cells with LNA oligonucleotides, , 10 days of treatment. Scrambled is a control oligo with a scrambled sequence not related to the Htral target sequence.
Figure 5. NHP PK/PD study, IVT administration, 25pg/eye. A) HTRA1 mRNA level measured in the retina by qPCR. B) oligo content in the retina measured by oligo ELISA. C) HTRA1 mRNA level illustrated by ISH. D-E) Quantification of HTRA1 protein level in retina and vitreous, respectively, by IP-MS. Dots show data for individual animals. Error bars show standard errors for technical replicates (n=3). F-G) Reduction in HTRA1 protein level in retina and vitreous, respectively illustrated by western blot.
Figure 6. A Compound of the invention (Compound ID NO 67,1 ). The compound may be in the form of a pharmaceutical salt, such as a sodium salt or a potassium salt.
Figure 7. A Compound of the invention (Compound ID NO 86,1 ). The compound may be in the form of a pharmaceutical salt, such as a sodium salt or a potassium salt.
Figure 8. A Compound of the invention (Compound ID N073,1 ). The compound may be in the form of a pharmaceutical salt, such as a sodium salt or a potassium salt.
Figure 9. An example of a pharmaceutical salt of compound 67,1 : M+ is a suitable cation, typically a positive metal ion, such as a sodium or potassium ion. The stoichiometric ratio of the cation to the oligonucleotide anion will depend on the charge of the cation used.
Suitably, cations with one, two or three positive charge (M+, M++, or M+++, may be used). For illustrative purpose, twice as many single + charged cations (monovalent), such as Na+ or K+ are needed as compared to a divalent cation such as Ca2+
Figure 10. An example of a pharmaceutical salt of compound 86,1 : See the figure legend for figure 9 for the description of the cation M+.
Figure 11. An example of a pharmaceutical salt of compound73,1 : See the figure legend for figure 9 for the description of the cation M+.
Figure 12A. Compounds #15,3 and #17 were administered intravitreally in cynomolgus monkeys, and aqueous humor samples were collected at days 3, 8, 15, and 22 post injection. Proteins from undiluted samples were analyzed by capillary electrophoresis using a Peggy Sue device (Protein Simple). HTRA1 was detected using a custom-made polycolonal rabbit antiserum. Data from animals #J60154 (Vehicle), J60158 (C. Id#15,3), J60162 (C. Id#17) are presented.
Figure 12B. Signal intensities were quantified by comparison to purified recombinant (S328A mutant) HTRA1 protein (Origene, #TP700208). The calibration curve is shown here. Figure 12C. Top panel: Calculated HTRA1 aqueous humor concentration from individual animal was plotted against time post injection. Bottom panel: average HTRA1 concentration for the vehicle group at each time point was determined and corresponding relative
concentration in treated animals calculated. Open circle: individual value, closed circle: group average. % HTRA1 reduction for day 22 is indicated.
Figure 13. HTRA1 mRNA plotted against HTRA1 protein levels in aqueous humor (blue diamonds) or in retina (red squares) in cynomolgus monkeys treated with various LNA molecules targeting the HTRA1 transcript. Values are expressed as percentage normalized to PBS controls.
Figure 14. Correlation of HTRA1 protein in aqueous humor with (A) HTRA1 protein in retina and (B) HTRA1 mRNA in retina in cynomolgus monkeys treated with various LNA molecules targeting the HTRA1 transcript. Values are expressed as percentage normalized to PBS controls.
Figure 15. Schematic of the Complement System (From Morgan et al., Nat. Rev. Drug Discovery 2015)
DEFINITIONS
Oligonucleotide
The term“oligonucleotide” as used herein is defined as it is generally understood by the skilled person as a molecule comprising two or more covalently linked nucleosides. Such covalently bound nucleosides may also be referred to as nucleic acid molecules or oligomers. Oligonucleotides are commonly made in the laboratory by solid-phase chemical synthesis followed by purification. When referring to a sequence of the oligonucleotide, reference is made to the sequence or order of nucleobase moieties, or modifications thereof, of the covalently linked nucleotides or nucleosides. The oligonucleotide of the invention is man-made, and is chemically synthesized, and is typically purified or isolated. The oligonucleotide of the invention may comprise one or more modified nucleosides or nucleotides.
Antisense oligonucleotides
The term“Antisense oligonucleotide” as used herein is defined as oligonucleotides capable of modulating expression of a target gene by hybridizing to a target nucleic acid, in particular to a contiguous sequence on a target nucleic acid. The antisense oligonucleotides are not essentially double stranded and are therefore not siRNAs or shRNAs. Preferably, the antisense oligonucleotides of the present invention are single stranded. It is understood that single stranded oligonucleotides of the present invention can form hairpins or intermolecular duplex structures (duplex between two molecules of the same oligonucleotide), as long as the degree of intra or inter self-complementarity is less than 50% across of the full length of the oligonucleotide
Contiguous Nucleotide Sequence
The term“contiguous nucleotide sequence” refers to the region of the oligonucleotide which is complementary to the target nucleic acid. The term is used interchangeably herein with the term“contiguous nucleobase sequence” and the term“oligonucleotide motif sequence”.
In some embodiments all the nucleotides of the oligonucleotide constitute the contiguous nucleotide sequence. In some embodiments the oligonucleotide comprises the contiguous nucleotide sequence, such as a F-G-F’ gapmer region, and may optionally comprise further nucleotide(s), for example a nucleotide linker region which may be used to attach a functional group to the contiguous nucleotide sequence. The nucleotide linker region may or may not be complementary to the target nucleic acid. Adventurously, the contiguous nucleotide sequence is 100% complementary to the target nucleic acid.
Nucleotides
Nucleotides are the building blocks of oligonucleotides and polynucleotides, and for the purposes of the present invention include both naturally occurring and non-naturally occurring nucleotides. In nature, nucleotides, such as DNA and RNA nucleotides comprise a ribose sugar moiety, a nucleobase moiety and one or more phosphate groups (which is absent in nucleosides). Nucleosides and nucleotides may also interchangeably be referred to as“units” or“monomers”.
Modified nucleoside
The term“modified nucleoside” or“nucleoside modification” as used herein refers to nucleosides modified as compared to the equivalent DNA or RNA nucleoside by the introduction of one or more modifications of the sugar moiety or the (nucleo)base moiety. In a preferred embodiment the modified nucleoside comprise a modified sugar moiety. The term modified nucleoside may also be used herein interchangeably with the term“nucleoside analogue” or modified“units” or modified“monomers”. Nucleosides with an unmodified DNA or RNA sugar moiety are termed DNA or RNA nucleosides herein. Nucleosides with modifications in the base region of the DNA or RNA nucleoside are still generally termed DNA or RNA if they allow Watson Crick base pairing.
Modified internucieoside linkages
The term“modified internucieoside linkage” is defined as generally understood by the skilled person as linkages other than phosphodiester (PO) linkages, that covalently couples two nucleosides together. The oligonucleotides of the invention may therefore comprise modified internucieoside linkages. In some embodiments, the modified internucieoside linkage increases the nuclease resistance of the oligonucleotide compared to a phosphodiester linkage. For naturally occurring oligonucleotides, the internucieoside linkage includes phosphate groups creating a phosphodiester bond between adjacent nucleosides. Modified internucieoside linkages are particularly useful in stabilizing oligonucleotides for in vivo use,
and may serve to protect against nuclease cleavage at regions of DNA or RNA nucleosides in the oligonucleotide of the invention, for example within the gap region of a gapmer oligonucleotide, as well as in regions of modified nucleosides, such as region F and F’.
In an embodiment, the oligonucleotide comprises one or more internucleoside linkages modified from the natural phosphodiester, such one or more modified internucleoside linkages that is for example more resistant to nuclease attack. Nuclease resistance may be determined by incubating the oligonucleotide in blood serum or by using a nuclease resistance assay (e.g. snake venom phosphodiesterase (SVPD)), both are well known in the art. Internucleoside linkages which are capable of enhancing the nuclease resistance of an oligonucleotide are referred to as nuclease resistant internucleoside linkages. In some embodiments at least 50% of the internucleoside linkages in the oligonucleotide, or contiguous nucleotide sequence thereof, are modified, such as at least 60%, such as at least 70%, such as at least 80 or such as at least 90% of the internucleoside linkages in the oligonucleotide, or contiguous nucleotide sequence thereof, are nuclease resistant internucleoside linkages. In some embodiments all of the internucleoside linkages of the oligonucleotide, or contiguous nucleotide sequence thereof, are nuclease resistant internucleoside linkages. It will be recognized that, in some embodiments the nucleosides which link the oligonucleotide of the invention to a non-nucleotide functional group, such as a conjugate, may be phosphodiester.
A preferred modified internucleoside linkage is phosphorothioate.
Phosphorothioate internucleoside linkages are particularly useful due to nuclease resistance, beneficial pharmacokinetics and ease of manufacture. In some embodiments at least 50% of the internucleoside linkages in the oligonucleotide, or contiguous nucleotide sequence thereof, are phosphorothioate, such as at least 60%, such as at least 70%, such as at least 80% or such as at least 90% of the internucleoside linkages in the oligonucleotide, or contiguous nucleotide sequence thereof, are phosphorothioate. In some embodiments all of the internucleoside linkages of the oligonucleotide, or contiguous nucleotide sequence thereof, are phosphorothioate.
Nuclease resistant linkages, such as phosphorothioate linkages, are particularly useful in oligonucleotide regions capable of recruiting nuclease when forming a duplex with the target nucleic acid, such as region G for gapmers. Phosphorothioate linkages may, however, also be useful in non-nuclease recruiting regions and/or affinity enhancing regions such as regions F and F’ for gapmers. Gapmer oligonucleotides may, in some embodiments comprise one or more phosphodiester linkages in region F or F’, or both region F and F’, which the internucleoside linkage in region G may be fully phosphorothioate.
Advantageously, all the internucleoside linkages in the contiguous nucleotide sequence of the oligonucleotide are phosphorothioate linkages.
It is recognized that, as disclosed in EP2 742 135, antisense oligonucleotide may comprise other internucleoside linkages (other than phosphodiester and phosphorothioate), for example alkyl phosphonate / methyl phosphonate internucleosides, which according to EP2 742 135 may for example be tolerated in an otherwise DNA phosphorothioate gap region.
Nucleobase
The term nucleobase includes the purine (e.g. adenine and guanine) and pyrimidine (e.g. uracil, thymine and cytosine) moiety present in nucleosides and nucleotides which form hydrogen bonds in nucleic acid hybridization. In the context of the present invention the term nucleobase also encompasses modified nucleobases which may differ from naturally occurring nucleobases, but are functional during nucleic acid hybridization. In this context “nucleobase” refers to both naturally occurring nucleobases such as adenine, guanine, cytosine, thymidine, uracil, xanthine and hypoxanthine, as well as non-naturally occurring variants. Such variants are for example described in Hirao et al (2012) Accounts of Chemical Research vol 45 page 2055 and Bergstrom (2009) Current Protocols in Nucleic Acid
Chemistry Suppl. 37 1.4.1.
In a some embodiments the nucleobase moiety is modified by changing the purine or pyrimidine into a modified purine or pyrimidine, such as substituted purine or substituted pyrimidine, such as a nucleobased selected from isocytosine, pseudoisocytosine, 5-methyl cytosine, 5-thiozolo-cytosine, 5-propynyl-cytosine, 5-propynyl-uracil, 5-bromouracil 5- thiazolo-uracil, 2-thio-uracil, 2’thio-thymine, inosine, diaminopurine, 6-aminopurine, 2- aminopurine, 2,6-diaminopurine and 2-chloro-6-aminopurine.
The nucleobase moieties may be indicated by the letter code for each corresponding nucleobase, e.g. A, T, G, C or U, wherein each letter may optionally include modified nucleobases of equivalent function. For example, in the exemplified oligonucleotides, the nucleobase moieties are selected from A, T, G, C, and 5-methyl cytosine. Optionally, for LNA gapmers, 5-methyl cytosine LNA nucleosides may be used.
Modified oligonucleotide
The term modified oligonucleotide describes an oligonucleotide comprising one or more sugar-modified nucleosides and/or modified internucleoside linkages. The term chimeric” oligonucleotide is a term that has been used in the literature to describe oligonucleotides with modified nucleosides.
Complemen tarity
The term“complementarity” describes the capacity for Watson-Crick base-pairing of nucleosides/nucleotides. Watson-Crick base pairs are guanine (G)-cytosine (C) and adenine (A) - thymine (T)/uracil (U). It will be understood that oligonucleotides may comprise nucleosides with modified nucleobases, for example 5-methyl cytosine is often used in place of cytosine, and as such the term complementarity encompasses Watson
Crick base-paring between non-modified and modified nucleobases (see for example Hirao et al (2012) Accounts of Chemical Research vol 45 page 2055 and Bergstrom (2009)
Current Protocols in Nucleic Acid Chemistry Suppl. 37 1.4.1 ).
The term“% complementary” as used herein, refers to the number of nucleotides in percent of a contiguous nucleotide sequence in a nucleic acid molecule (e.g. oligonucleotide) which, at a given position, are complementary to ( i.e . form Watson Crick base pairs with) a contiguous sequence of nucleotides, at a given position of a separate nucleic acid molecule (e.g. the target nucleic acid or target sequence). The percentage is calculated by counting the number of aligned bases that form pairs between the two sequences (when aligned with the target sequence 5’-3’ and the oligonucleotide sequence from 3’-5’), dividing by the total number of nucleotides in the oligonucleotide and multiplying by 100. In such a comparison a nucleobase/nucleotide which does not align (form a base pair) is termed a mismatch.
Preferably, insertions and deletions are not allowed in the calculation of % complementarity of a contiguous nucleotide sequence.
The term“fully complementary”, refers to 100% complementarity.
Identity
The term“Identity” as used herein, refers to the proportion of nucleotides (expressed in percent) of a contiguous nucleotide sequence in a nucleic acid molecule (e.g.
oligonucleotide) which across the contiguous nucleotide sequence, are identical to a reference sequence (e.g. a sequence motif). The percentage of identity is thus calculated by counting the number of aligned bases that are identical (a match) between two sequences (e.g. in the contiguous nucleotide sequence of the compound of the invention and in the reference sequence), dividing that number by the total number of nucleotides in the aligned region and multiplying by 100. Therefore, Percentage of Identity = (Matches x 100)/Length of aligned region (e.g. the contiguous nucleotide sequence). Insertions and deletions are not allowed in the calculation the percentage of identity of a contiguous nucleotide sequence. It will be understood that in determining identity, chemical modifications of the nucleobases are disregarded as long as the functional capacity of the nucleobase to form Watson Crick base pairing is retained (e.g. 5-methyl cytosine is considered identical to a cytosine for the purpose of calculating % identity).
Hybridization
The term“hybridizing” or“hybridizes” as used herein is to be understood as two nucleic acid strands (e.g. an oligonucleotide and a target nucleic acid) forming hydrogen bonds between base pairs on opposite strands thereby forming a duplex. The affinity of the binding between two nucleic acid strands is the strength of the hybridization. It is often described in terms of the melting temperature (Tm) defined as the temperature at which half of the oligonucleotides are duplexed with the target nucleic acid. At physiological conditions Tm is not strictly
proportional to the affinity (Mergny and Lacroix, 2003, Oligonucleotides 13:515-537). The standard state Gibbs free energy DQ° is a more accurate representation of binding affinity and is related to the dissociation constant (Kd) of the reaction by AG°=-RTIn(Kd), where R is the gas constant and T is the absolute temperature. Therefore, a very low AG° of the reaction between an oligonucleotide and the target nucleic acid reflects a strong
hybridization between the oligonucleotide and target nucleic acid. AG° is the energy associated with a reaction where aqueous concentrations are 1 M, the pH is 7, and the temperature is 37°C. The hybridization of oligonucleotides to a target nucleic acid is a spontaneous reaction and for spontaneous reactions AG° is less than zero. AG° can be measured experimentally, for example, by use of the isothermal titration calorimetry (ITC) method as described in Hansen et al., 1965, Chem. Comm. 36-38 and Holdgate et a!., 2005, Drug Discov Today. The skilled person will know that commercial equipment is available for AG° measurements. AG° can also be estimated numerically by using the nearest neighbor model as described by SantaLucia, 1998, Proc Natl Acad Sci USA. 95: 1460-1465 using appropriately derived thermodynamic parameters described by Sugimoto et al., 1995, Biochemistry 34:11211-1 1216 and McTigue et al., 2004, Biochemistry 43:5388-5405. In order to have the possibility of modulating its intended nucleic acid target by hybridization, oligonucleotides of the present invention hybridize to a target nucleic acid with estimated AG° values below -10 kcal for oligonucleotides that are 10-30 nucleotides in length. In some embodiments the degree or strength of hybridization is measured by the standard state Gibbs free energy AG°. The oligonucleotides may hybridize to a target nucleic acid with estimated AG° values below the range of -10 kcal, such as below -15 kcal, such as below - 20 kcal and such as below -25 kcal for oligonucleotides that are 8-30 nucleotides in length.
In some embodiments the oligonucleotides hybridize to a target nucleic acid with an estimated AG° value of -10 to -60 kcal, such as -12 to -40, such as from -15 to -30 kcal or- 16 to -27 kcal such as -18 to -25 kcal.
Target Sequence
The oligonucleotide comprises a contiguous nucleotide region which is complementary to or hybridizes to a sub-sequence of the target nucleic acid molecule. The term“target sequence” as used herein refers to a sequence of nucleotides present in the target nucleic acid which comprises the nucleobase sequence which is complementary to the contiguous nucleotide region or sequence of the oligonucleotide of the invention. In some
embodiments, the target sequence consists of a region on the target nucleic acid which is complementary to the contiguous nucleotide region or sequence of the oligonucleotide of the invention. In some embodiments the target sequence is longer than the complementary
sequence of a single oligonucleotide, and may, for example represent a preferred region of the target nucleic acid which may be targeted by several oligonucleotides of the invention. The oligonucleotide of the invention comprises a contiguous nucleotide region which is complementary to the target nucleic acid, such as a target sequence.
The oligonucleotide comprises a contiguous nucleotide region of at least 10 nucleotides which is complementary to or hybridizes to a target sequence present in the target nucleic acid molecule. The contiguous nucleotide region (and therefore the target sequence) comprises of at least 10 contiguous nucleotides, such as 1 1 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 21 , 22, contiguous nucleotides, such as from 12-22, such as from 14-18 contiguous nucleotides.
In some embodiments the target sequence is present within a sequence selected from the group consisting of SEQ ID NO 113, 114, 1 15, 1 16, 1 17 and 118.
Target nucleic acid
According to the present invention, the target nucleic acid is a nucleic acid which encodes mammalian HTRA1 and may for example be a gene, a RNA, a mRNA, and pre-mRNA, a mature mRNA or a cDNA sequence. The target may therefore be referred to as an HTRA1 target nucleic acid.
Suitably, the target nucleic acid encodes an HTRA1 protein, in particular mammalian HTRA1 , such as human HTRA1 (See for example tables 1 & 2 which provides the mRNA and pre-mRNA sequences for human HTRA1 ).
In some embodiments, the target nucleic acid is selected from the group consisting of SEQ ID NO: 1 or 2 or naturally occurring variants thereof (e.g. sequences encoding a mammalian HTRA1 protein.
A target cell is a cell which is expressing the HTRA1 target nucleic acid. In preferred embodiments the target nucleic acid is the HTRA1 mRNA, such as the HTRA1 pre-mRNA or HTRA1 mature mRNA. The poly A tail of HTRA1 mRNA is typically disregarded for antisense oligonucleotide targeting.
If employing the oligonucleotide of the invention in research or diagnostics the target nucleic acid may be a cDNA or a synthetic nucleic acid derived from DNA or RNA.
The target sequence may be a sub-sequence of the target nucleic acid. In some
embodiments the oligonucleotide or contiguous nucleotide region is fully complementary to, or only comprises one or two mismatches to an HTRA1 sub-sequence, such as a sequence selected from the group consisting of SEQ ID NO 113, 114, 115, 116, 117 or 231.
The target sequence may be a sub-sequence of the target nucleic acid. In some
embodiments the oligonucleotide or contiguous nucleotide region is fully complementary to, or only comprises one or two mismatches to an HTRA1 sub-sequence, such as a sequence
selected from the group consisting of SEQ ID NO 124 - 230. In some embodiments the oligonucleotide or contiguous nucleotide region is fully complementary to, or only comprises one or two mismatches to an HTRA1 sub-sequence SEQ ID NO 231.
Complementarity to the target or sub-sequence thereof is measured over the length of the oligonucleotide, or contiguous nucleotide region thereof.
For in vivo or in vitro application, the oligonucleotide of the invention is typically capable of inhibiting the expression of the HTRA1 target nucleic acid in a cell which is expressing the HTRA1 target nucleic acid. The contiguous sequence of nucleobases of the oligonucleotide of the invention is typically complementary to the HTRA1 target nucleic acid, as measured across the length of the oligonucleotide, optionally with the exception of one or two mismatches, and optionally excluding nucleotide based linker regions which may link the oligonucleotide to an optional functional group such as a conjugate, or other non
complementary terminal nucleotides (e.g. region D). The target nucleic acid may, in some embodiments, be a RNA or DNA, such as a messenger RNA, such as a mature mRNA or a pre-mRNA. In some embodiments the target nucleic acid is a RNA or DNA which encodes mammalian HTRA1 protein, such as human HTRA1 , e.g. the human HTRA1 mRNA sequence, such as that disclosed as SEQ ID NO 1 (NM_002775.4, Gl:190014575). Further information on exemplary target nucleic acids is provided in tables 1 & 2.
Table 1. Genome and assembly information for human and Cyno HTRA1.
Fwd = forward strand. The genome coordinates provide the pre-mRNA sequence (genomic sequence). The NCBI reference provides the mRNA sequence (cDNA sequence).
*The National Center for Biotechnology Information reference sequence database is a comprehensive, integrated, non-redundant, well-annotated set of reference sequences including genomic, transcript, and protein. It is hosted at www.ncbi.nlm.nih.gov/refseq.
**ln the NCBI reference sequence there is a stretch of 100 nucleotides from position 126 to position 227 whose identity is not known. In SEQ ID NO 3 & 4, this stretch has been replaced by the nucleotides appearing in both human and Macaca mulatta HTRA1 premRNA sequences in this region.
Naturally occurring variant
The term“naturally occurring variant” refers to variants of HTRA1 gene or transcripts which originate from the same genetic loci as the target nucleic acid, but may differ for example, by virtue of degeneracy of the genetic code causing a multiplicity of codons encoding the same amino acid, or due to alternative splicing of pre-mRNA, or the presence of polymorphisms, such as single nucleotide polymorphisms, and allelic variants. Based on the presence of the sufficient complementary sequence to the oligonucleotide, the oligonucleotide of the invention may therefore target the target nucleic acid and naturally occurring variants thereof. In some embodiments, the naturally occurring variants have at least 95% such as at least 98% or at least 99% homology to a mammalian HTRA1 target nucleic acid, such as a target nucleic acid selected form the group consisting of SEQ ID NO 1 ,2, 3 & 4.
Modulation of expression
The term“modulation of expression” as used herein is to be understood as an overall term for an oligonucleotide’s ability to alter the amount of the target nucleic acid or target protein, such as e.g. the level of HTRA1 when compared to the amount of the target HTRA1 before administration of the oligonucleotide. Alternatively, modulation of expression may be determined by reference to a control experiment where the oligonucleotide of the invention is not administered. One type of modulation is an oligonucleotide’s ability to inhibit, down- regulate, reduce, suppress, remove, stop, block, prevent, lessen, lower, avoid or terminate expression of HTRA1 , e.g. by degradation of mRNA or blockage of transcription. The antisense oligonucleotide of the invention are capable of inhibiting, down-regulating, reduce, suppress, remove, stop, block, prevent, lessen, lower, avoid or terminate expression of HTRA1.
High affinity modified nucleosides
A high affinity modified nucleoside is a modified nucleotide which, when incorporated into the oligonucleotide enhances the affinity of the oligonucleotide for its complementary target, for example as measured by the melting temperature (Tm). A high affinity modified nucleoside of the present invention preferably result in an increase in melting temperature between +0.5 to +12°C, more preferably between +1.5 to +10°C and most preferably between+3 to +8°C per modified nucleoside. Numerous high affinity modified nucleosides are known in the art and include for example, many 2’ substituted nucleosides as well as locked nucleic acids (LNA)
(see e.g. Freier & Altmann; Nucl. Acid Res., 1997, 25, 4429-4443 and Uhlmann; Curr. Opinion in Drug Development, 2000, 3(2), 293-213).
Sugar modifications
The oligomer of the invention may comprise one or more nucleosides which have a modified sugar moiety, i.e. a modification of the sugar moiety when compared to the ribose sugar moiety found in DNA and RNA.
Numerous nucleosides with modification of the ribose sugar moiety have been made, primarily with the aim of improving certain properties of oligonucleotides, such as affinity and/or nuclease resistance.
Such modifications include those where the ribose ring structure is modified, e.g. by replacement with a hexose ring (HNA), or a bicyclic ring, which typically have a biradicle bridge between the C2 and C4 carbons on the ribose ring (LNA), or an unlinked ribose ring which typically lacks a bond between the C2 and C3 carbons (e.g. UNA). Other sugar modified nucleosides include, for example, bicyclohexose nucleic acids (WO201 1/017521 ) or tricyclic nucleic acids (WO2013/154798). Modified nucleosides also include nucleosides where the sugar moiety is replaced with a non-sugar moiety, for example in the case of peptide nucleic acids (PNA), or morpholino nucleic acids.
Sugar modifications also include modifications made via altering the substituent groups on the ribose ring to groups other than hydrogen, or the 2’-OH group naturally found in DNA and RNA nucleosides. Substituents may, for example be introduced at the 2’, 3’, 4’ or 5’ positions.
2’ sugar modified nucleosides.
A 2’ sugar modified nucleoside is a nucleoside which has a substituent other than H or -OH at the 2’ position (2’ substituted nucleoside) or comprises a 2’ linked biradicle capable of forming a bridge between the 2’ carbon and a second carbon in the ribose ring, such as LNA (2’ - 4’ biradicle bridged) nucleosides.
Indeed, much focus has been spent on developing 2’ substituted nucleosides, and numerous 2’ substituted nucleosides have been found to have beneficial properties when incorporated into oligonucleotides. For example, the 2’ modified sugar may provide enhanced binding affinity and/or increased nuclease resistance to the oligonucleotide. Examples of 2’ substituted modified nucleosides are 2’-0-alkyl-RNA, 2’-0-methyl-RNA, 2’- alkoxy-RNA, 2’-0-methoxyethyl-RNA (MOE), 2’-amino-DNA, 2’-Fluoro-RNA, and 2’-F-ANA nucleoside. For further examples, please see e.g. Freier & Altmann; Nucl. Acid Res., 1997, 25, 4429-4443 and Uhlmann; Curr. Opinion in Drug Development, 2000, 3(2), 293-213, and Deleavey and Damha, Chemistry and Biology 2012, 19, 937. Below are illustrations of some 2’ substituted modified nucleosides.
In relation to the present invention 2’ substituted does not include 2’ bridged molecules like LNA.
Locked Nucleic Acids (LNA)
A“LNA nucleoside” is a 2’- modified nucleoside which comprises a biradical linking the C2’ and C4’ of the ribose sugar ring of said nucleoside (also referred to as a“2’- 4’ bridge”), which restricts or locks the conformation of the ribose ring. These nucleosides are also termed bridged nucleic acid or bicyclic nucleic acid (BNA) in the literature. The locking of the conformation of the ribose is associated with an enhanced affinity of hybridization (duplex stabilization) when the LNA is incorporated into an oligonucleotide for a complementary RNA or DNA molecule. This can be routinely determined by measuring the melting temperature of the oligonucleotide/complement duplex.
Non limiting, exemplary LNA nucleosides are disclosed in WO 99/014226, WO
00/66604, WO 98/039352 , WO 2004/046160, WO 00/047599, WO 2007/134181 , WO 2010/077578, WO 2010/036698, WO 2007/090071 , WO 2009/006478, WO 201 1/156202, WO 2008/154401 , WO 2009/067647, WO 2008/150729, Morita et al., Bioorganic &
Med.Chem. Lett. 12, 73-76, Seth et al. J. Org. Chem. 2010, Vol 75(5) pp. 1569-81 , and Mitsuoka et al., Nucleic Acids Research 2009, 37(4), 1225-1238, and Wan and Seth, J. Medical Chemistry 2016, 59, 9645-9667.
Further non limiting, exemplary LNA nucleosides are disclosed in Scheme 1 .
Scheme 1 :
Particular LNA nucleosides are beta-D-oxy-LNA, 6’-methyl-beta-D-oxy LNA such as (S)-6’-methyl-beta-D-oxy-LNA (ScET) and ENA.
A particularly advantageous LNA is beta-D-oxy-LNA.
RNase H Activity and Recruitment
The RNase H activity of an antisense oligonucleotide refers to its ability to recruit RNase H when in a duplex with a complementary RNA molecule. WO01/23613 provides in vitro methods for determining RNaseH activity, which may be used to determine the ability to recruit RNaseH. Typically an oligonucleotide is deemed capable of recruiting RNase H if it, when provided with a complementary target nucleic acid sequence, has an initial rate, as measured in pmol/l/min, of at least 5%, such as at least 10% or more than 20% of the of the initial rate determined when using a oligonucleotide having the same base sequence as the modified oligonucleotide being tested, but containing only DNA monomers with
phosphorothioate linkages between all monomers in the oligonucleotide, and using the methodology provided by Example 91 - 95 of WO01/23613 (hereby incorporated by reference). For use in determining RHase H activity, recombinant human RNase H 1 is available from Lubio Science GmbH, Lucerne, Switzerland.
Gapmer
The antisense oligonucleotide of the invention, or contiguous nucleotide sequence thereof may be a gapmer. The antisense gapmers are commonly used to inhibit a target nucleic acid via RNase H mediated degradation. A gapmer oligonucleotide comprises at least three distinct structural regions a 5’-flank, a gap and a 3’-flank, F-G-F’ in the‘5 -> 3’ orientation. The“gap” region (G) comprises a stretch of contiguous DNA nucleotides which enable the oligonucleotide to recruit RNase H. The gap region is flanked by a 5’ flanking region (F) comprising one or more sugar modified nucleosides, advantageously high affinity sugar modified nucleosides, and by a 3’ flanking region (F’) comprising one or more sugar modified nucleosides, advantageously high affinity sugar modified nucleosides. The one or more sugar modified nucleosides in region F and F’ enhance the affinity of the oligonucleotide for the target nucleic acid ( i.e . are affinity enhancing sugar modified nucleosides). In some embodiments, the one or more sugar modified nucleosides in region F and F’ are 2’ sugar modified nucleosides, such as high affinity 2’ sugar modifications, such as independently selected from LNA and 2’-MOE.
In a gapmer design, the 5’ and 3’ most nucleosides of the gap region are DNA nucleosides, and are positioned adjacent to a sugar modified nucleoside of the 5’ (F) or 3’ (F’) region respectively. The flanks may further defined by having at least one sugar modified nucleoside at the end most distant from the gap region, i.e. at the 5’ end of the 5’ flank and at the 3’ end of the 3’ flank.
Regions F-G-F’ form a contiguous nucleotide sequence. Antisense oligonucleotides of the invention, or the contiguous nucleotide sequence thereof, may comprise a gapmer region of formula F-G-F’.
The overall length of the gapmer design F-G-F’ may be, for example 12 to 32 nucleosides, such as 13 to 24, such as 14 to 22 nucleosides, Such as from 14 to17, such as 16 to18 nucleosides.
By way of example, the gapmer oligonucleotide of the present invention can be represented by the following formulae:
Fi-8-G5-i6-F’i-8, such as
F1-8-G7-16-F 2-8
with the proviso that the overall length of the gapmer regions F-G-F’ is at least 12, such as at least 14 nucleotides in length.
Regions F, G and F’ are further defined below and can be incorporated into the F-G-F’ formula.
Gapmer - Region G
Region G (gap region) of the gapmer is a region of nucleosides which enables the oligonucleotide to recruit RNaseH, such as human RNase H1 , typically DNA nucleosides.
RNaseH is a cellular enzyme which recognizes the duplex between DNA and RNA, and enzymatically cleaves the RNA molecule. Suitably gapmers may have a gap region (G) of at least 5 or 6 contiguous DNA nucleosides, such as 5 - 16 contiguous DNA nucleosides, such as 6 - 15 contiguous DNA nucleosides, such as 7-14 contiguous DNA nucleosides, such as 8 - 12 contiguous DNA nucleotides, such as 8 - 12 contiguous DNA nucleotides in length. Whilst traditional gapmers have a DNA gap region, there are numerous examples of modified nucleosides which allow for RNaseH recruitment when they are used within the gap region. Modified nucleosides which have been reported as being capable of recruiting RNaseH when included within a gap region include, for example, alpha-L-LNA, C4’ alkylated DNA (as described in PCT/EP2009/050349 and Vester et a!., Bioorg. Med. Chem. Lett. 18 (2008) 2296 - 2300, both incorporated herein by reference), arabinose derived nucleosides like ANA and 2'F-ANA (Mangos et al. 2003 J. AM. CHEM. SOC. 125, 654-661 ), UNA
(unlocked nucleic acid) (as described in Fluiter et al., Mol. Biosyst, 2009, 10, 1039 incorporated herein by reference). UNA is unlocked nucleic acid, typically where the bond between C2 and C3 of the ribose has been removed, forming an unlocked“sugar” residue. The modified nucleosides used in such gapmers may be nucleosides which adopt a 2’ endo (DNA like) structure when introduced into the gap region, i.e. modifications which allow for RNaseH recruitment). In some embodiments the DNA Gap region (G) described herein may optionally contain 1 to 3 sugar modified nucleosides which adopt a 2’ endo (DNA like) structure when introduced into the gap region.
Region G -“Gap-breaker”
Alternatively, there are numerous reports of the insertion of a modified nucleoside which confers a 3’ endo conformation into the gap region of gapmers, whilst retaining some RNaseH activity. Such gapmers with a gap region comprising one or more 3’endo modified nucleosides are referred to as“gap-breaker” or“gap-disrupted” gapmers, see for example WO2013/022984. Gap-breaker oligonucleotides retain sufficient region of DNA nucleosides within the gap region to allow for RNaseH recruitment. The ability of gapbreaker
oligonucleotide design to recruit RNaseH is typically sequence or even compound specific - see Rukov et al. 2015 Nucl. Acids Res. Vol. 43 pp. 8476-8487, which discloses“gapbreaker” oligonucleotides which recruit RNaseH which in some instances provide a more specific cleavage of the target RNA. Modified nucleosides used within the gap region of gap- breaker oligonucleotides may for example be modified nucleosides which confer a 3’endo confirmation, such 2’ -O-methyl (OMe) or 2’-0-MOE (MOE) nucleosides, or beta-D LNA nucleosides (the bridge between C2’ and C4’ of the ribose sugar ring of a nucleoside is in the beta conformation), such as beta-D-oxy LNA or ScET nucleosides.
Gapmer - flanking regions, F and F’
Region F is positioned immediately adjacent to the 5’ DNA nucleoside of region G. The 3’ most nucleoside of region F is a sugar modified nucleoside, such as a high affinity sugar modified nucleoside, for example a 2’ substituted nucleoside, such as a MOE nucleoside, or an LNA nucleoside.
Region F’ is positioned immediately adjacent to the 3’ DNA nucleoside of region G. The 5’ most nucleoside of region F’ is a sugar modified nucleoside, such as a high affinity sugar modified nucleoside, for example a 2’ substituted nucleoside, such as a MOE nucleoside, or an LNA nucleoside.
Region F is 1 - 8 contiguous nucleotides in length, such as 2-6, such as 3-4 contiguous nucleotides in length. Advantageously the 5’ most nucleoside of region F is a sugar modified nucleoside. In some embodiments the two 5’ most nucleoside of region F are sugar modified nucleoside. In some embodiments the 5’ most nucleoside of region F is an LNA nucleoside. In some embodiments the two 5’ most nucleoside of region F are LNA nucleosides. In some embodiments the two 5’ most nucleoside of region F are 2’ substituted nucleoside nucleosides, such as two 3’ MOE nucleosides. In some embodiments the 5’ most nucleoside of region F is a 2’ substituted nucleoside, such as a MOE nucleoside.
Region F’ is 2 - 8 contiguous nucleotides in length, such as 3-6, such as 4-5 contiguous nucleotides in length. Advantageously, embodiments the 3’ most nucleoside of region F’ is a sugar modified nucleoside. In some embodiments the two 3’ most nucleoside of region F’ are sugar modified nucleoside. In some embodiments the two 3’ most nucleoside of region F’ are LNA nucleosides. In some embodiments the 3’ most nucleoside of region F’ is an LNA nucleoside. In some embodiments the two 3’ most nucleoside of region F’ are 2’ substituted nucleoside nucleosides, such as two 3’ MOE nucleosides. In some embodiments the 3’ most nucleoside of region F’ is a 2’ substituted nucleoside, such as a MOE nucleoside. It should be noted that when the length of region F or F’ is one, it is advantageously an LNA nucleoside.
In some embodiments, region F and F’ independently consists of or comprises a contiguous sequence of sugar modified nucleosides. In some embodiments, the sugar modified nucleosides of region F may be independently selected from 2’-0-alkyl-RNA units, 2’-0- methyl-RNA, 2’-amino-DNA units, 2’-fluoro-DNA units, 2’-alkoxy-RNA, MOE units, LNA units, arabino nucleic acid (ANA) units and 2’-fluoro-ANA units.
In some embodiments, region F and F’ independently comprises both LNA and a 2’ substituted modified nucleosides (mixed wing design).
In some embodiments, region F and F’ consists of only one type of sugar modified nucleosides, such as only MOE or only beta-D-oxy LNA or only ScET. Such designs are also termed uniform flanks or uniform gapmer design.
In some embodiments, all the nucleosides of region F or F’, or F and F’ are LNA nucleosides, such as independently selected from beta-D-oxy LNA, ENA or ScET nucleosides. In some embodiments region F consists of 1-5, such as 2-4, such as 3-4 such as 1 , 2, 3, 4 or 5 contiguous LNA nucleosides. In some embodiments, all the nucleosides of region F and F’ are beta-D-oxy LNA nucleosides.
In some embodiments, all the nucleosides of region F or F’, or F and F’ are 2’ substituted nucleosides, such as OMe or MOE nucleosides. In some embodiments region F consists of 1 , 2, 3, 4, 5, 6, 7, or 8 contiguous OMe or MOE nucleosides. In some embodiments only one of the flanking regions can consist of 2’ substituted nucleosides, such as OMe or MOE nucleosides. In some embodiments it is the 5’ (F) flanking region that consists 2’ substituted nucleosides, such as OMe or MOE nucleosides whereas the 3’ (F’) flanking region comprises at least one LNA nucleoside, such as beta-D-oxy LNA nucleosides or cET nucleosides. In some embodiments it is the 3’ (F’) flanking region that consists 2’ substituted nucleosides, such as OMe or MOE nucleosides whereas the 5’ (F) flanking region comprises at least one LNA nucleoside, such as beta-D-oxy LNA nucleosides or cET nucleosides.
In some embodiments, all the modified nucleosides of region F and F’ are LNA nucleosides, such as independently selected from beta-D-oxy LNA, ENA or ScET nucleosides, wherein region F or F’, or F and F’ may optionally comprise DNA nucleosides (an alternating flank, see definition of these for more details). In some embodiments, all the modified nucleosides of region F and F’ are beta-D-oxy LNA nucleosides, wherein region F or F’, or F and F’ may optionally comprise DNA nucleosides (an alternating flank, see definition of these for more details).
In some embodiments the 5’ most and the 3’ most nucleosides of region F and F’ are LNA nucleosides, such as beta-D-oxy LNA nucleosides or ScET nucleosides.
In some embodiments, the internucleoside linkage between region F and region G is a phosphorothioate internucleoside linkage. In some embodiments, the internucleoside linkage between region F’ and region G is a phosphorothioate internucleoside linkage. In some embodiments, the internucleoside linkages between the nucleosides of region F or F’, F and F’ are phosphorothioate internucleoside linkages.
LNA Gapmer
An LNA gapmer is a gapmer wherein either one or both of region F and F’ comprises or consists of LNA nucleosides. A beta-D-oxy gapmer is a gapmer wherein either one or both of region F and F’ comprises or consists of beta-D-oxy LNA nucleosides.
Treatment
The term’treatment’ as used herein refers to both treatment of an existing disease (e.g. a disease or disorder as herein referred to), or prevention of a disease, i.e. prophylaxis. It will
therefore be recognized that treatment as referred to herein may, in some embodiments, be prophylactic.
DETAILED DESCRIPTION OF THE INVENTION
Htral oligonucleotide antagonists
The Htral oligonucleotide antagonist is an antisense oligonucleotide which targets HTRA1 , such as a mammalian HTRA1 , preferably human HTRA1. Exemplary antisense
oligonucleotides targeting Htral which may be used in the present invention include those listed in the examples as Compound ID NO #5,1 - #112,1 and compound A and compound B. Preferred antisense oligonucleotide antagonists of HTRA1 for use in the present invention may be selected from the group of compounds of formula:
wherein capital letters represent beta-D-oxy LNA nucleosides, lower case letters are DNA nucleosides, subscript s represents a phosphorothioate internucleoside linkage, and mC represent 5 methyl cytosine beta-D-oxy LNA nucleosides, and mc represents 5 methyl cytosine DNA nucleosides.
The invention relates to oligonucleotides capable of inhibiting the expression of HTRA1. The modulation is may achieved by hybridizing to a target nucleic acid encoding HTRA1 or which is involved in the regulation of HTRA1. The target nucleic acid may be a mammalian HTRA 1 sequence, such as a sequence selected from the group consisting of SEQ ID 1 , 2, 3 or 4.
In some embodiments the antisense oligonucleotide of the invention is capable of modulating the expression of the target by inhibiting or down-regulating it. Preferably, such modulation produces an inhibition of expression of at least 20% compared to the normal expression level of the target, such as at least 30%, 40%, 50%, 60%, 70%, 80%, or 90% inhibition compared to the normal expression level of the target. In some embodiments compounds of the invention may be capable of inhibiting expression levels of HTRA1 mRNA by at least 60% or 70% in vitro using ARPE-19 cells. In some embodiments compounds of the invention may be capable of inhibiting expression levels of HTRA1 mRNA by at least 60% or 70% in vitro using ARPE-19 cells. In some embodiments compounds of the invention may be capable of inhibiting expression levels of HTRA1 protein by at least 50% in
vitro using ARPE-19 cells. Suitably, the examples provide assays which may be used to measure HTRA1 RNA or protein inhibition. The target modulation is triggered by the hybridization between a contiguous nucleotide sequence of the oligonucleotide and the target nucleic acid. In some embodiments the oligonucleotide of the invention comprises mismatches between the oligonucleotide and the target nucleic acid. Despite mismatches hybridization to the target nucleic acid may still be sufficient to show a desired modulation of HTRA1 expression. Reduced binding affinity resulting from mismatches may advantageously be compensated by increased number of nucleotides in the oligonucleotide and/or an increased number of modified nucleosides capable of increasing the binding affinity to the target, such as 2’ modified nucleosides, including LNA, present within the oligonucleotide sequence.
An aspect of the present invention relates to antisense oligonucleotide which comprises a contiguous nucleotide region of 10 to 30 nucleotides in length with at least 90%
complementarity to HTRA1 target sequence, such as fully complementary to an HTRA1 target sequence, e.g. a nucleic acid selected from the group consisting SEQ ID NO 1 , 2, 3 & 4.
In some embodiments, the oligonucleotide comprises a contiguous sequence which is at least 90% complementary, such as at least 91 %, such as at least 92%, such as at least 93%, such as at least 94%, such as at least 95%, such as at least 96%, such as at least 97%, such as at least 98%, or 100% complementary with a region of the target nucleic acid. In some embodiments, the oligonucleotide of the invention, or a contiguous nucleotide sequence thereof is fully complementary (100% complementary) to a region of the target nucleic acid, or in some embodiments may comprise one or two mismatches between the oligonucleotide and the target nucleic acid.
The Htral oligonucleotide antagonist of the present invention is an oligonucleotide targeting a mammalian HTRA1 nucleic acid, i.e. are capable of inhibiting the expression of HTRA1 and may be used to treat or prevent diseases related to the functioning of the HTRA1. The oligonucleotides targeting HTRA1 are antisense oligonucleotides, i.e. are complementary to their HTRA1 nucleic acid target. The oligonucleotide may be in the form of a
pharmaceutically acceptable salt, such as a sodium salt or a potassium salt or ammonium salt, such as a sodium salt. In some embodiments the oligonucleotide is dissolved in a pharmaceutically acceptable solvent, such as a ophthalmically acceptable solution, such as phosphate buffered saline.
The antisense oligonucleotides may comprise a contiguous nucleotide sequence of 10 - 30 nucleotides in length with at least 90% complementarity, such as fully complementary to a
mammalian HTRA1 nucleic acid, such as SEQ ID NO 1 , SEQ ID NO 2, SEQ ID NO 3 or SEQ ID NO 4.
The antisense oligonucleotide may be an LNA antisense oligonucleotides, such as LNA gapmer oligonucleotides, which comprise a contiguous nucleotide sequence of 10 - 30 nucleotides in length with at least 90% complementarity, such as fully complementary to a HTRA1 nucleic acid, such as a sequence selected from the group consisting of SEQ ID NO 1 , SEQ ID NO 2, SEQ ID NO 3 or SEQ ID NO 4.
The antisense oligonucleotide may comprise a contiguous nucleotide region of at 10 - 30, such as 12 - 22, nucleotides, wherein the contiguous nucleotide region is at least 90% such as 100% complementary to SEQ ID NO 113.
The antisense oligonucleotide may be of 10 - 30 nucleotides in length, wherein said antisense oligonucleotide comprises a contiguous nucleotide region of 10 - 30, such as 12 - 22, nucleotides which are at least 90% such as 100% complementarity to SEQ ID NO 1 13. The reverse complement of SEQ ID NO 1 13 is shown in SEQ ID NO 119.
The antisense oligonucleotide may comprise a contiguous nucleotide region of at 10 - 30, such as 12 - 22, nucleotides, wherein the contiguous nucleotide region is at least 90% such as 100% complementary to SEQ ID NO 114.
The antisense oligonucleotide of 10 - 30 nucleotides in length, may comprise a contiguous nucleotide region of 10 - 30, such as 12 - 22 nucleotides which are at least 90% such as 100% complementarity to SEQ ID NO 114. The reverse complement of SEQ ID NO 114 is SEQ ID NO 120.
The antisense oligonucleotide may comprise a contiguous nucleotide region of at 10 - 30, such as 12 - 22, nucleotides, wherein the contiguous nucleotide region is at least 90% such as 100% complementary to SEQ ID NO 115.
The antisense oligonucleotide of 10 - 30 nucleotides in length, may comrpise a contiguous nucleotide region of 10 - 30, such as 12 - 22 nucleotides which are at least 90% such as 100% complementarity to SEQ ID NO 115. The reverse complement of SEQ ID NO 115 is shown in SEQ ID NO 121.
The antisense oligonucleotide may comprise a contiguous nucleotide region of at 10 - 30, such as 12 - 22, nucleotides, wherein the contiguous nucleotide region is at least 90% such as 100% complementary to SEQ ID NO 116.
The antisense oligonucleotide of 10 - 30 nucleotides in length, may comprise a contiguous nucleotide region of 10 - 30, such as 12 - 22 nucleotides which are at least 90% such as 100% complementarity to SEQ ID NO 116. The reverse complement of SEQ ID NO 116 is SEQ ID NO 122.
The antisense oligonucleotide may comprise a contiguous nucleotide region of at 10 - 30, such as 12 - 22, nucleotides, wherein the contiguous nucleotide region is at least 90% such as 100% complementary to SEQ ID NO 1 17.
The antisense oligonucleotide of 10 - 30 nucleotides in length, may comprise a contiguous nucleotide region of 10 - 30, such as 12 - 22 nucleotides which are at least 90% such as 100% complementarity to SEQ ID NO 1 17. The reverse complement of SEQ ID NO 1 17 is SEQ ID NO 123.
In some embodiments, the antisense oligonucleotide comprises a contiguous nucleotide region of at least 10, or at least 12, at least 13, or at least 14 or at least 15 or at least 16 contiguous nucleotides present SEQ ID NO 1 18: 5’ CTTCTTCTATCTACGCATTG 3’. The reverse complement of SEQ ID NO 1 18 is SEQ ID NO 231 : C AAT G C GT AG AT AG AAG AAG . The invention provides an antisense oligonucleotide which comprises a contiguous nucleotide region of at least 10, or at least 12, at least 13, or at least 14 or at least 15 or at least 16 contiguous nucleotides complementary to SEQ ID NO 231 .
The invention refers to an antisense oligonucleotide which comprises a contiguous nucleotide region of at least 10, or at least 12, or at least 13, or at least 14 or at least 15 or 16 contiguous nucleotides present SEQ ID NO 67.
The invention provides an antisense oligonucleotide which comprises a contiguous nucleotide region of at least 10, or at least 12, or at least 13, or at least 14 or at least 15 or 16 contiguous nucleotides present SEQ ID NO 86.
The invention provides an antisense oligonucleotide which comprises a contiguous nucleotide region of at least 10, or at least 12, or at least 13, or at least 14 or at least 15 or at least 16 or at least 17 or 18 contiguous nucleotides present SEQ ID NO 73.
The invention provides an antisense oligonucleotide which comprises a contiguous nucleotide region of at least 10, or at least 12, or at least 13, or at least 14 or at least 15 or 16 contiguous nucleotides complementary to SEQ ID NO 186.
The invention provides an antisense oligonucleotide which comprises a contiguous nucleotide region of at least 10, or at least 12, or at least 13, or at least 14 or at least 15 or 16 contiguous nucleotides complementary to SEQ ID NO 205.
The invention provides an antisense oligonucleotide which comprises a contiguous nucleotide region of at least 10, or at least 12, or at least 13, or at least 14 or at least 15 or at least 16 or at least 17 or 18 contiguous nucleotides complementary to SEQ ID NO 192.
In some embodiments, the invention refers to an oligonucleotide comprising or consisting of an oligonucleotide selected from the group consisting of: and
wherein capital letters represent beta-D-oxy LNA nucleosides, lower case letters are DNA nucleosides, subscript s represents a phosphorothioate internucleoside linkage, and mC represent 5 methyl cytosine beta-D-oxy LNA nucleosides, and mc represents 5 methyl cytosine DNA nucleosides.
In some embodiments the antisense oligonucleotide of the invention is capable of modulating the expression of the target by inhibiting or down-regulating it. Preferably, such modulation produces an inhibition of expression of at least 20% compared to the normal expression level of the target, such as at least 30%, 40%, 50%, 60%, 70%, 80%, or 90% inhibition compared to the normal expression level of the target. In some embodiments compounds of the invention may be capable of inhibiting expression levels of HTRA1 mRNA by at least 60% or 70% in vitro using ARPE-19 cells. In some embodiments compounds of the invention may be capable of inhibiting expression levels of HTRA1 mRNA by at least 60% or 70% in vitro using ARPE-19 cells. In some embodiments compounds of the invention may be capable of inhibiting expression levels of HTRA1 protein by at least 50% in vitro using ARPE-19 cells. Suitably, the examples provide assays which may be used to measure HTRA1 RNA or protein inhibition. The target modulation is triggered by the hybridization between a contiguous nucleotide sequence of the oligonucleotide and the target nucleic acid. In some embodiments the oligonucleotide of the invention comprises mismatches between the oligonucleotide and the target nucleic acid. Despite mismatches hybridization to the target nucleic acid may still be sufficient to show a desired modulation of HTRA1 expression. Reduced binding affinity resulting from mismatches may advantageously be compensated by increased number of nucleotides in the oligonucleotide and/or an increased number of modified nucleosides capable of increasing the binding affinity to the target, such as 2’ modified nucleosides, including LNA, present within the oligonucleotide sequence.
An aspect of the present invention relates to an antisense oligonucleotide which comprises a contiguous nucleotide region of 10 to 30 nucleotides in length with at least 90%
complementarity to HTRA1 target sequence, such as fully complementary to an HTRA1 target sequence, e.g. a nucleic acid selected from the group consisting SEQ ID NO 1 , 2, 3 & 4.
In some embodiments, the oligonucleotide comprises a contiguous sequence which is at least 90% complementary, such as at least 91 %, such as at least 92%, such as at least 93%, such as at least 94%, such as at least 95%, such as at least 96%, such as at least 97%, such as at least 98%, or 100% complementary with a region of the target nucleic acid.
In some embodiments, the oligonucleotide of the invention, or a contiguous nucleotide sequence thereof is fully complementary (100% complementary) to a region of the target nucleic acid, or in some embodiments may comprise one or two mismatches between the oligonucleotide and the target nucleic acid.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least 12 nucleotides thereof, is at least 90% complementary, such as fully (or 100%)
complementary to a region of a sequence selected from the group consisting of SEQ ID NO 1 19, 120, 121 , 122 or 123.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least 12 nucleotides thereof, is at least 90% complementary, such as fully (or 100%)
complementary to a region of a sequence selected from the group consisting of SEQ ID NOs 124- 230.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least 12 nucleotides thereof, is at least 90% complementary, such as fully (or 100%)
complementary to a region of SEQ ID NO 186.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least 12 nucleotides thereof, is at least 90% complementary, such as fully (or 100%)
complementary to a region of SEQ ID NO 192.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least
12 nucleotides thereof, is at least 90% complementary, such as fully (or 100%)
complementary to a region of SEQ ID NO 205.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least
13 nucleotides thereof, is at least 90% complementary, such as fully (or 100%)
complementary to a region of SEQ ID NO 186.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least 13 nucleotides thereof, is at least 90% complementary, such as fully (or 100%)
complementary to a region of SEQ ID NO 192.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least
13 nucleotides thereof, is at least 90% complementary, such as fully (or 100%)
complementary to a region of SEQ ID NO 205.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least
14 nucleotides thereof, is fully (or 100%) complementary to a sequence selected from the group consisting of SEQ ID NO 113, 114, 115, 116, 117 and 231.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least 14 nucleotides thereof, is at least 90% complementary, such as fully (or 100%)
complementary to a region of SEQ ID NO 186.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least 14 nucleotides thereof, is at least 90% complementary, such as fully (or 100%)
complementary to a region of SEQ ID NO 192.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least
14 nucleotides thereof, is at least 90% complementary, such as fully (or 100%)
complementary to a region of SEQ ID NO 205.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least
15 nucleotides thereof, is at least 90% complementary, such as fully (or 100%)
complementary to a region of SEQ ID NO 186.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least 15 nucleotides thereof, is at least 90% complementary, such as fully (or 100%)
complementary to a region of SEQ ID NO 192.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least
15 nucleotides thereof, is at least 90% complementary, such as fully (or 100%)
complementary to a region of SEQ ID NO 205.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least
16 nucleotides thereof, is fully (or 100%) complementary to a sequence selected from the group consisting of SEQ ID NO SEQ ID NO 1 13, 1 14, 1 15, 1 16, 1 17 and 231. .
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least 16 nucleotides thereof, is at least 90% complementary, such as fully (or 100%)
complementary to a region of SEQ ID NO 186.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least 16, such as 16, 17 or 18 nucleotides thereof, is at least 90% complementary, such as fully (or 100%) complementary to a region of SEQ ID NO 192.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least 16 nucleotides thereof, is at least 90% complementary, such as fully (or 100%)
complementary to a region of SEQ ID NO 205.
In some embodiments the oligonucleotide, or contiguous nucleotide region thereof is fully (or 100%) complementary to a sequence selected from the group consisting of a sequence selected from the group consisting of SEQ ID NO SEQ ID NO 113, 1 14, 1 15, 1 16, 1 17 and 231.
In some embodiments the oligonucleotide, or contiguous nucleotide region thereof is fully (or 100%) complementary to a sequence selected from the group consisting of a sequence selected from the group consisting of SEQ ID NO 124 - 230.
In some embodiments the oligonucleotide, or contiguous nucleotide region thereof is fully (or 100%) complementary to SEQ ID NO 186.
In some embodiments the oligonucleotide, or contiguous nucleotide region thereof is fully (or 100%) complementary to SEQ ID NO 192.
In some embodiments the oligonucleotide, or contiguous nucleotide region thereof is fully (or 100%) complementary to SEQ ID NO 205.
It is understood that the oligonucleotide motif sequences can be modified to for example increase nuclease resistance and/or binding affinity to the target nucleic acid. Modifications are described in the definitions and in the Oligonucleotide design” section.
In some embodiments, the oligonucleotide of the invention, or contiguous nucleotide region thereof is fully complementary (100% complementary) to a region of the target nucleic acid, or in some embodiments may comprise one or two mismatches between the oligonucleotide and the target nucleic acid. In some embodiments the oligonucleotide, or contiguous nucleotide sequence of at least 12 nucleotides thereof, is at least 90% complementary, such as fully (or 100%) complementary to the target nucleic acid sequence.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least 12 nucleotides thereof, has 100% identity to a sequence selected from the group consisting of SEQ ID NOs 5 - 1 11.
In some embodiments the oligonucleotide, or a contiguous nucleotide sequence of at least 14 nucleotides thereof, has 100% identity to a sequence selected from the group consisting of SEQ ID NOs 5 - 11 1
In some embodiments the oligonucleotide, or contiguous nucleotide sequence of at least 16 nucleotides thereof, has 100% identity to a sequence selected from the group consisting of SEQ ID NOs 5 - 11 1
In some embodiments the oligonucleotide, or contiguous nucleotide region thereof, comprises or consists of a sequence selected from SEQ ID NOs 5 - 1 11.
In some embodiments the oligonucleotide of the invention is selected from the following group (Note the target subsequence is the reverse complement of the oligonucleotide motif):
or conjugate thereof; wherein for the column entitled compound design, capital letters are LNA nucleosides, lower case letters are DNA nucleosides, cytosine nucleosides are optionally 5 methyl cytosine, and internucleoside linkages are at least 80%, such as at least 90% or 100% modified internucleoside linkages, such as phosphorothioate internucleoside linkages. In some embodiments all internucleoside linkages of the compounds in the compound design column in the above table are phosphorothioate internucleoside linkages. The motif and target subsequence sequences are nucleobase sequences.
The invention provides the following oligonucleotides:
wherein in the compounds of the above table, capital letters represent beta-D-oxy LNA nucleosides, all LNA cytosines are 5-methyl cytosine (as indicated by the superscript m), lower case letters represent DNA nucleosides, superscript m before a lower case c represents a 5 methyl cytosine DNA nucleoside. All internucleoside linkages are phosphorothioate internucleoside linkages.
Pharmaceutical Composition
In a further aspect, the invention provides pharmaceutical compositions comprising any of the aforementioned oligonucleotides and/or oligonucleotide conjugates and a pharmaceutically acceptable diluent, carrier, salt and/or adjuvant. A pharmaceutically acceptable diluent includes phosphate-buffered saline (PBS) and pharmaceutically acceptable salts include, but are not limited to, sodium and potassium salts. In some embodiments the pharmaceutically acceptable
diluent is sterile phosphate buffered saline. In some embodiments the oligonucleotide is used in the pharmaceutically acceptable diluent at a concentration of 50 - 300mM solution. In some embodiments, the oligonucleotide of the invention is administered at a dose of 10 - 1000pg.
WO 2007/031091 provides suitable and preferred examples of pharmaceutically acceptable diluents, carriers and adjuvants (hereby incorporated by reference). Suitable dosages, formulations, administration routes, compositions, dosage forms, combinations with other therapeutic agents, pro-drug formulations are also provided in W02007/031091.
Oligonucleotides or oligonucleotide conjugates of the invention may be mixed with
pharmaceutically acceptable active or inert substances for the preparation of pharmaceutical compositions or formulations. Compositions and methods for the formulation of pharmaceutical compositions are dependent upon a number of criteria, including, but not limited to, route of administration, extent of disease, or dose to be administered.
In some embodiments, the oligonucleotide or oligonucleotide conjugate of the invention is a prodrug. In particular, with respect to oligonucleotide conjugates the conjugate moiety is cleaved of the oligonucleotide once the prodrug is delivered to the site of action, e.g. the target cell.
The invention provides for a method of treatment of a subject suffering from an ocular disease, such as macular degeneration, such as wet or dry age related macular degeneration (e.g.
wAMD, dAMD, geographic atrophy, intermediate dAMD) or diabetic retinopathy, said method comprising the step of administering an effective amount of the oligonucleotide, conjugate or composition of the invention to the subject, wherein said method further comprises
administering a furtehr therapeutic agent to the subject, such as an antiangiogenic agent (e.g. a VEFG antagonist) or an inhibitor of the complement system.
In some embodiments of the method of treatment of a subject suffering from an ocular disease, such as macular degeneration, such as wet or dry age related macular degeneration (e.g.
wAMD, dAMD, geographic atrophy, intermediate AMD) of the invention, the method comprises administering at least two dosages of the HTRA1 oligonucleotide antagonist, such as antisense oligonucleotide, or pharmaceutically acceptable salt thereof, in an intraocular injection in a dosage of from about 10pg - 200 pg, wherein the dosage interval between administration consecutive is at least 4 weeks ( i.e . a dosage interval is > 4 weeks), or at least monthly ( i.e . a dosage interval is > 1 month).
The Complement System
The term“complement component",“complement system", "complement proteins" or“ complement component proteins” refers to the molecules that are involved inactivation of the complement system (see figure 15). The classical pathway components include, e.g., C1q, C1 r,
complex (membrane attack complex: MAC).
The alternative pathway components include, e.g., Factor B, Factor D, Properdin, Factor H and Factor I.
The complement system is a powerful effector of innate immunity. In many clinical situations, including age related macular degeneration, the defensive function of the complement system can be detrimental due to inducing or exacerbating immune, inflammatory and degenerative processes and conditions. At present, numerous complement system antagonists are in clinical development (see for example Ricklin & Lambris, Semin. Immunol. 2017) including, but not limited to complement factor D antagonists, complement factor B antagonists, complement factor C5 antagonists, complement factor C3 antagonists, complement factor P (also known as properdin) antagonists.
Further Therapeutics / Further Therapeutic Agents
The invention relates to the use of an HTRA1 oligonucleotide antagonist for the treatment or prophylaxis of dry age-related macular degeneration, such as geographic atrophy, in
combination with a further therapeutic agent, such as a further therapeutic agent selected from the group consisting of an anti-angiogenic agent, a VEGF antagonist, or a complement system antagonist.
In some embodiments, the further therapeutic agent is an anti-angiogenic agent, such as a VEGF antagonist, or is an inhibitor or the complement system, such as an antagonist of a complement component.
In some embodiments, the further therapeutic agent is a complement system antagonist, such as an antagonist of the classical complement pathway or an antagonist of the alternative complement pathway. In some embodiments, the further therapeutic agent is selected from the group consisting of complement factor D antagonist, a complement factor B antagonist, antagonist complement factor C5, antagonist complement factor C3, antagonist complement factor P (properdin), an antagonist of complement factor I, and an antagonist of complement factor H.
The further therapeutic may for example, be a small molecule therapeutic, a peptide
therapeutic, a protein therapeutic, an antibody therapeutic or an oligonucleotide therapeutic.
For the combined use of the HTRA1 oligonucleotide antagonist and a further therapeutic agent which is a protein therapeutic, and antibody therapeutic (such as a monoclonal antibody, antibody fragment or antibody fusion protein) or a peptide therapeutic.
In some embodiments, due to the protein binding capability of phosphorothioate
oligonucleotides, it may be advantageous to administer the HTRA1 oligonucleotide antagonist and a further therapeutic agent separately.
In some embodiments, the further therapeutic agent is an oligonucleotide therapeutic, such as an antisense oligonucleotide, a siRNA or an aptamer.
An anti-angiogenic agent, also known as angiogenesis inhibitor, is a therapeutic agent which inhibits blood vessel growth. Angiogenesis inhibitors interfere in several ways with various steps in blood vessel growth. Some are monoclonal antibodies that specifically recognize and bind to VEGF. When VEGF is attached to these drugs, it is unable to activate the VEGF receptor. Other angiogenesis inhibitors bind to VEGF and/or its receptor as well as to other receptors on the surface of endothelial cells or to other proteins in the downstream signaling pathways, blocking their activities. Some angiogenesis inhibitors are immunomodulatory drugs— agents that stimulate or suppress the immune system— that also have antiangiogenic properties. The most common angiogenesis inhibitors are antagonists of VEGF.
VEGF Antagonists
VEGF is a well-characterised signal protein which stimulates angiogenesis. In some
embodiments, the VEGF antagonist is an anti-VEGF antibody therapeutic such as ranibizumab (Lucentis®) or bevacizumab (Avastin®).
In some embodiments, the VEGF antagonist is administered via intraocular injection, such as via vitreal-injection. Administration of VEGF antagonists, such as anti-VEFG antibodies, is often advantageous to avoid systemic side effects, such as bleeding. In some embodiments, the VEGF antagonist is an anti-VEGF antibody such as ranibizumab (Lucentis®) or bevacizumab (Avastin®).
In some embodiments, the further therapeutic agent is a VEGF antagonist therapeutic which is an anti-VEGF antibody such as ranibizumab, or bevacizumab.
In some embodiments, the further therapeutic agent is a VEGF antagonist therapeutic which is an aptamer, such as pegaptanib (Macugen®).
In some embodiments, the further therapeutic agent is a VEGF antagonist therapeutic is a small molecule inhibitor of VEGF such as cediranib, lapatinib, sunitinib, sorafenib, axitinib, and pazopanib. The structure and formula of these are provided below:
Cediranib:
In some embodiments, the further therapeutic agent is a VEGF antagonist is a recombinant fusion protein VEGF inhibitor such as aflibercept (Eylea) - aflibercept is composed of vascular endothelial growth factor (VEGF)-binding portions from the extracellular domains of human VEGF receptors 1 and 2, that are fused to the Fc portion of the human lgG1 immunoglobulin.
Typically, antibody inhibitors of VEGF (anti-VEGF) are administered monthly of bimonthly to the eye via intravitreal injection (see US9220631 for example). Further examples of anti-VEGF antobodies are disclosed in W096/30046, W02006/047325 & W02008/063932.
Ranibizumab, CAS Number = 347396-82-1 , is disclosed in W098/45331 & W09845332 and comprises a light chain immunoglobulin of sequence SEQ A (SEQ ID NO 234, and a heavy chain immunoglobulin of SEQ B (SEQ ID NO 235).
Bevacizumab, CAS Number = 216974-75-3, comprises a light chain immunoglobulin of sequence SEQ C (SEQ ID NO 236), and a heavy chain immunoglobulin of SEQ D (SEQ ID NO 237).
In some embodiments, the further therapeutic agent is the anti-VEGF antibody, brolucizumab (CAS Number = 1531589-13-5) which is in clinical development for treatment of wAMD.
In some embodiments, the further therapeutic agent is aflibercept (Eylea®) (Holash et al. (2002) PNAS USA 99:11393-98; Riely and Miller (2007) Clin Cancer Res 13:4623-7s).
Aflibercept is a recombinant fusion protein antagonist of VEGF, composed of vascular endothelial growth factor (VEGF)-binding portions from the extracellular domains of human VEGF receptors 1 and 2, that are fused to the Fc portion of the human lgG1 immunoglobulin, for intravitreal administration (see US7306799, US7531 173, US7374758, US7608261 , US7070959 & US7374757)
In some embodiments the further therapeutic agent is a small molecule inhibitor of VEGF, such as a molecule selected from the group consisting of cediranib, lapatinib, sunitinib, sorafenib, axitinib, and pazopanib (these are anti-angiogenic drugs, which are used in cancer treatment). Complement Factor D inhibitors
In some embodiments, the further therapeutic agent is an antagonist of complement factor D (CFD). In some embodiments, the further therapeutic agent is an antibody antagonist of complement factor D (anti-CFD antibody), such as lampalizumab (CAS Number = 1278466-20- 8). Other examples of antibody inhibitors of complement factor D are disclosed in
WO2017/075173 and WO2017/075252. Anti-CFD antibodies may be administered via e.g. monthly of bimonthly to the eye via intravitreal injection.
Lampalizumab is an antigen-binding fragment (Fab) of a humanized, monoclonal antibody directed against complement factor D developed by Genentech (see e.g. Yaspan et al., Sci Transl Med. 2017 Jun 21 ;9(395)) (NCT02247479).
lampalizumab comprises a light chain immunoglobulin of sequence SEQ G SEQ ID NO 240), and a heavy chain immunoglobulin of SEQ H (SEQ ID NO 241 ).
In some embodiments, the further therapeutic agent is a small molecule inhibitor of complement factor D, such as a small molecule selected from the group consisting of ACH-4471 , ACH-5228 or ACH-5548 (as disclosed in US10106563B2, US10092584B2, US1001 1612B2 &
US10000516B2).
ACH-4471 (CAS No.: 1903768-17-1 )
These small molecule inhibitors of CFD, targets, binds to and blocks the activity of CFD, and thereby inhibits cleavage of complement factor B into Ba and Bb in the alternative pathway of the complement cascade. This inhibits CFD-mediated signaling and activation of the alternative complement pathway (ACP), blocks complement-mediated hemolysis in paroxysmal nocturnal hemoglobinuria (PNH) and prevents ACP-induced tissue damage. They may be administered orally.
Complement Factor B (CFB) Antagonists
In some embodiments the further therapeutic agent is an antagonist (inhibitor) of complement factor B.
The human complement factor B gene is found on human Chromosome
CHR_HSCHR6_MHC_MANN_CTG1 : 31 ,985,034-31 ,990,148 forward strand
(GRCh38:GL000253.2)).
Shaded bold text regions are the mature mRNA sequence.
Therapeutic oligonucleotides, such as antisense oligonucleotides targeting complement factor B may comprise a contiguous nucleotide sequence of at least 12, or at least 14 nucleotides which are complementary to, such as fully complementary to the human complement factor B pre- mRNA or mRNA. Therapeutic siRNAs or shRNAs typically comprise a region of at least 17 - 25 contiguous nucleotides which are complementary to, such as fully complementary to the human complement factor B mRNA.
W015038939 and W015168635 disclose antisense oligonucleotides targeting the human complement factor B pre-mRNA, and GalNAc conjuagtes thereof. Antisense oligonucleotides targeting the complement factor pre-mRNA are in clinical devlopement: In some advantageous embodiments, the further therapeutic agent is lONIS-FB-Lrx, also known as ISIS 696844. lONIS-FB-Lrx is disclosed in W015168635 as ISIS 696844 and comprises an antisense oligonucleotide of sequence: ATCCCACGCCCCTGTCCAGC (SEQ ID NO 243). The IONIS- FB-Lrx antisense oligonucleotide has a gapmer 5-10-5:
O-methoxyethyl (MOE) RNA nucleoside, each lower case letter is a DNA nucleoside, all cytosines are 5-methyl cytosine (designated by a preceding superscript m), and the
internucleoside linkages are phosphorothioate (designated a subscript s). The 5’ nucleoside (A) is conjugated to a trivalent conjugate moiety via a phosphate bond. The chemical formula of lONIS-FB-Lrx is presented below:
lONIS-FB-Lrx may be administered to the subject via for example subcutaneous, intravenous, or intramuscular injection or infusion. The presence of the GalNAc conjugate enhances uptake of the antisense oligonucleotide into hepatocytes.
In certain embodiments, the complement factor B antisense oligonucleotide is administered parenterally. For example, in certain embodiments the complement factor B antisense oligonucleotide is administered through injection or infusion, such as subcutaneous
administration, intravenous administration, intramuscular administration, intraarterial administration, or intraperitoneal administration. In some advantageous embodiments, IONIS- FB-Lrx is administered sub-cutaneous injection, such as a monthly or bimonthly sub-cutaneous injection.
In some embodiment the HTRA1 antagonist is an antisense oligonucleotide of formula
wherein capital letters represent beta-D-oxy LNA nucleosides, lower case letters are DNA nucleosides, subscript s represents a phosphorothioate internucleoside linkage, and mC represent 5 methyl cytosine beta-D-oxy LNA nucleosides, and mc represents 5 methyl cytosine DNA nucleosides, and the further therapeutic agent is lONIS-FB-Lrx.
In some embodiment the HTRA1 antagonist is an antisense oligonucleotide of formula
wherein capital letters represent beta-D- oxy LNA nucleosides, lower case letters are DNA nucleosides, subscript s represents a phosphorothioate internucleoside linkage, and mC represent 5 methyl cytosine beta-D-oxy LNA nucleosides, and the further therapeutic agent is lONIS-FB-Lrx.
In some embodiment the HTRA1 antagonist is an antisense oligonucleotide of formula
LNA nucleosides, lower case letters are DNA nucleosides, subscript s represents a phosphorothioate internucleoside linkage, and mC represent 5 methyl cytosine beta-D-oxy LNA nucleosides, and the further therapeutic agent is lONIS-FB-Lrx.
In some embodiment the HTRA1 antagonist is an antisense oligonucleotide of formula
nucleosides, lower case letters are DNA nucleosides, subscript s represents a phosphorothioate internucleoside linkage, and the further therapeutic agent is lONIS-FB-Lrx.
In some embodiment the HTRA1 antagonist is an antisense oligonucleotide of formula
wherein capital letters represent beta-D-oxy LNA nucleosides, lower case letters are DNA nucleosides, subscript s represents a phosphorothioate internucleoside linkage, and the further therapeutic agent is lONIS-FB-Lrx.
Complement Factor P (Properdin, CFP) Antagonists
In some embodiments, the further therapeutic agent is an antagonist of complement factor P, such as an anti-CFP antibody, such as CLG-561 or NM9401 .
In some embodiments, the further therapeutic agent is CLG-561 (also written as CLG561 ), which is a monoclonal antibody in clinical development for use in combination with LFG316 (Tesidolumab, an anti-complement factor C5 antibody). CLG-561 may be administered via intravitreal injection monthly or bimonthly.
In some embodiments, the further therapeutic agent is an antibody targeting CFP, such as NM9401 (US8664362): which comprises a heavy chain shown in SEQ 244 and a light chain shown in SEQ 245:
Heavy chain SEQ ID NO 244,
Complement Factor C3 Antagonist
In some embodiments the further therapeutic agent is an antagonist or inhibitor (used interchangeable) of complement factor C3, such as APL-2 or CB2782.
APL-2 is a pegulated peptide conjugate that binds specifically to C3 and C3b to inhibit C3 cleavage and activation into C3a and C3b of formula (See WO2014078731 &
CB2782 is a novel protease derived from human membrane type serine protease 1 (MTSP-1 ) that selectively cleaves C3 and has been shown to completely inhibit C3 (US9290757 and US9359598). In some embodiments the further therapeutic agent is a modified MTSP-1 protease selected from SEQ ID Nos 252, 253, 254 and 255 (as disclosed in EP2402437).
Complement Factor C5 Antagonist
In some embodiments the further therapeutic agent is an antagonist or inhibitor (used interchangeable) of complement factor C5, such as an anti-complement factor C5 antibody, such as LFG316 (Tesidolumab, CAS number = 1531594-08-7, disclosed in WO2017/12375) or Eculizumab (solris, CAS Number = 219685-50-4), and an anti-complement factor C5 aptamer, such as avacincaptad pegol (Zimura®, also known as ARC1905).
In some embodiments, the further therapeutic agent is Tesidolumab, which is in clinical development (NCT02515942) via administration via intravitreal injection. Tesidolumab comprises a heavy and light chain immunoglobulin of SEQ J (SEQ ID NO 246) and SEQ K (SEQ ID NO 247):
SEQ J:
In some embodiments, the further therapeutic agent is Eculizumab, which is a humanised monoclonal (lgG2/4K) antibody (WO2017/12375) comprising a light chain of SEQ L (SEQ ID NO 248), and a heavy chain of SEQ M (SEQ ID NO 249).
In some embodiments, the further therapeutic agent is avacincaptad pegol (Zimura®, also known as ARC1905). Avacincaptad pegol (CAS-1491 144-00-3) is as Anti-C5 aptamer being developed by Ophthotech (WO 2007103549) (NCT02686658): Avacincaptad pegol comprises the aptamer sequence SEQ N (SEQ ID NO 250):
According to Toxnet, Avacincaptad pegol chemical name is Poly(oxy-1 ,2-ethanediyl), alpha- hydro-omega-methoxy-, 5'-ether with RNA ((2'-deoxy-2'-fluoro)C-Gm-(2'-deoxy-2'-fluoro)C-(2'- deoxy-2'-fluoro)C-G-(2'-deoxy-2'-fluoro)C-Gm-Gm-(2'-deoxy-2'-fluoro)U-(2'-deoxy-2'-fluoro)C- (2'-deoxy-2'-fluoro)U-(2'-deoxy-2'-fluoro)C-Am-Gm-Gm-(2'-deoxy-2'-fluoro)C-G-(2'-deoxy-2'- fluoro)C-(2'-deoxy-2'-fluoro)U-Gm-Am-Gm-(2'-deoxy-2'-fluoro)U-(2'-deoxy-2'-fluoro)C-(2'-deoxy- 2'-fluoro)U-Gm-Am-Gm-(2'-deoxy-2'-fluoro)U-(2'-deoxy-2'-fluoro)U-(2'-deoxy-2'-fluoro)U-A-(2'- deoxy-2'-fluoro)C-(2'-deoxy-2'-fluoro)C-(2'-deoxy-2'-fluoro)U-Gm-(2'-deoxy-2'-fluoro)C-Gm-(3'- >3')-dT) 5'-(6-(((2,3-dihydroxypropoxy)carbonyl)amino)hexyl hydrogen phosphate), sodium salt (2:1 :39)
Anti-platelet derived growth factor (PGDF) therapeutic.
In some embodiment the further therapeutic agent is a PGDF antagonist (e.g. a PGFD-B antagonist), for example an antagonist of an anti-PGDF aptamer, such as Fovista (pegpleranib). Pegpleranib is in clinical trials for treatment of dAMD in combination with Lucentis®.
Pegpleranib has the formula (sequence):
Antibody Therapeutics
In some embodiments, the further therapeutic agent is an antibody therapeutic. For treatment of macular degeneration antibodies may be administered directly to the eye via intravitreal injection, such as via a monthly or bimonthly intravitreal injection.
The terms antibody is used herein refer to an antibody that is capable of binding with sufficient affinity to the target (anti-“Target”) such that the antibody is useful as a therapeutic agent in targeting the target (e.g. anti-VEGF or anti-complement component, such as anti-CFD, anti- CFC3, anti-CFC5, anti-CFP etc). In one aspect, the extent of binding of an anti- antibody to an unrelated, non- protein is less than about 10% of the binding of the antibody to as measured, e.g., by surface plasmon resonance (SPR). In certain aspects, an antibody that binds to has a
dissociation constant (KD) of
or i¾ 0.001 nM (e.g., 10-8 M or less, e.g., from 10-8 M to 10-13 M, e.g., from 10-9 M to 10-13 M). An antibody is said to“specifically bind” to when the antibody has a KD of 1 mM or less. For use in human therapy the antibody targets the human target. However, in certain aspects, an anti antibody binds to an epitope of that is conserved among from different species.
The term“antibody” herein is used in the broadest sense and encompasses various antibody structures, including but not limited to monoclonal antibodies, polyclonal antibodies,
multispecific antibodies (e.g., bispecific antibodies), and antibody fragments so long as they exhibit the desired antigen-binding activity.
An“antibody fragment” refers to a molecule other than an intact antibody that comprises a portion of an intact antibody that binds the antigen to which the intact antibody binds. Examples of antibody fragments include but are not limited to Fv, Fab, Fab', Fab’-SH, F(ab')2; diabodies; linear antibodies; single-chain antibody molecules (e.g., scFv, and scFab); single domain antibodies (dAbs); and multispecific antibodies formed from antibody fragments. For a review of certain antibody fragments, see Holliger and Hudson, Nature Biotechnology 23:1 126-1136 (2005).
Administration of the HTRA1 antagonist/antisense oligonucleotide therapeutic
In some embodiments the invention relates to the use of oligonucleotides, such as the HTRA1 antagonist or antisense oligonucleotide therapeutic, in the treatment of diseases or disorders selected from eye disorders, such as macular degeneration, including age related macular degeneration (AMD), such as dry AMD or wet AMD, and diabetic retinopathy. In some embodiments the oligonucleotide conjugates or pharmaceutical compositions of the invention may be for use in the treatment of geographic atrophy or intermediate dAMD or dAMD.
The HTRA1 antagonist/antisense oligonucleotide therapeutic according to the invention is typically administered in an effective amount. Administration is typically via intravitreal injection, such as in an ophthalmic solution comprising a dose of (unit dose), for example 5pg - 200pg per eye, such 10pg - 100pg, such as 20pg - 50pg per eye. In some embodiments, the volume of ophthalmic solution administered to each eye is, for example about 25mI - about 100mI, such as about 50mI.
Ophthalmic solution
An ophthalmic solution is a solution which comprises the Htral antagonist or antisense oligonucleotide therapeutic, such as the HTRA1 oligonucleotide antagonist dissolved in a solvent which is compatible with administration via injection into the eye, such as an intravitreal injection. The solvent is suitably a buffered aqueous solution, such as phosphate buffered saline, e.g. of pH about 7 - about 7.5, such as about pH 7.4. Other ophthalmic acceptable
buffered solutions may be used. The ophthalmic solution is sterile. The solvent is suitably a buffered aqueous solution, such as phosphate buffered saline, e.g. of pH about 7 - about 7.5, such as about 7.4.
In some embodiments, the dosage interval, i.e. the period of time between consecutive dosings is at least monthy, such as at least bi monthly or at least once every three months.
EMBODIMENTS
1. An antisense oligonucleotide selected from the group selected from TTCtatctacgcaTTG (SEQ ID NO 67), CTTCttctatctacgcAT (SEQ ID NO 73), and TACTttaatagcTCAA (SEQ ID NO 86), TATttacctggtTGTT (SEQ ID NO 232 ) AtATttacctgGTTgTT (SEQ ID NO 233), wherein capital letters represent a LNA nucleosides, LNA C are optionally 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are
phosphorothioate internucleoside linkages; for use in the treatment of geographic atrophy in a subject, wherein the treatment further comprises administering a further therapeutic agent to the subject.
2. The antisense oligonucleotide according to embodiment 1 wherein the antisense oligonucleotide is
wherein capital letters represent beta-D-oxy LNA nucleosides, lower case letters are DNA nucleosides, subscript s represents a phosphorothioate internucleoside linkage, and mC represent 5 methyl cytosine beta-D-oxy LNA nucleosides, and mc represents 5 methyl cytosine DNA nucleosides.
3. The antisense oligonucleotide according to embodiment 1 wherein the antisense
oligonucleotide is
, wherein capital letters represent beta-D-oxy LNA nucleosides, lower case letters are DNA nucleosides, subscript s represents a phosphorothioate internucleoside linkage, and mC represent 5 methyl cytosine beta-D-oxy LNA nucleosides, and mc represents 5 methyl cytosine DNA nucleosides.
4. The antisense oligonucleotide according to embodiment 1 wherein the antisense
oligonucleotide is
, wherein capital letters represent beta-D-oxy LNA nucleosides, lower case letters are DNA nucleosides, subscript s represents a phosphorothioate internucleoside linkage, and mC represent 5 methyl cytosine beta-D-oxy LNA nucleosides.
5. The antisense oligonucleotide according to embodiment 1 wherein the antisense
represent beta-D-oxy LNA nucleosides, lower case letters are DNA nucleosides, and subscript s represents a phosphorothioate internucleoside linkage.
6. The antisense oligonucleotide according to embodiment 1 wherein the antisense oligonucleotide is
wherein capital letters represent beta-D-oxy LNA nucleosides, lower case letters are DNA nucleosides, and subscript s represents a phosphorothioate internucleoside linkage.
7. The use of an antisense oligonucleotide according to any one of embodiments 1 - 6, in the manufacture of a medicament for the treatment of geographic atrophy, wherein the medicament is for use in combination treatment with a further therapeutic.
8. A method for the treatment of geographic atrophy in a subject in need of treatment, said method comprising administering an effective amount of the antisense oligonucleotide according to any one of embodiments 1 - 6 to said subject, and further administering an effective amount of a further therapeutic agent, to the subject.
9. The antisense oligonucleotide, use or method according to any one of embodiments 1 - 8, wherein the further therapeutic agent is a vascular endothelial growth factor (VEGF) antagonist therapeutic, such as an anti-VEGF antibody therapeutic.
10. The antisense oligonucleotide, use or method according to embodiment 9, wherein the VEGF antagonist therapeutic is ranibizumab.
1 1. The antisense oligonucleotide, use or method according to embodiment 9, wherein the VEGF antagonist therapeutic is bevacizumab.
12. The antisense oligonucleotide, use or method according to embodiment 9, wherein the VEGF antagonist therapeutic is pegaptanib.
13. The antisense oligonucleotide, use or method according to embodiment 9, wherein the VEGF antagonist therapeutic is a small molecule inhibitor of VEGF such as a molecule selected from the group consisting of cediranib, lapatinib, sunitinib, sorafenib, axitinib, and pazopanib.
14. The antisense oligonucleotide, use or method according to embodiment 9, wherein the VEGF antagonist therapeutic is a recombinant fusion protein VEGF inhibitor such as aflibercept.
15. The antisense oligonucleotide, use or method according to any one of embodiments 1 - 8, wherein the further therapeutic agent is an antagonist of complement factor D (CFD).
16. The antisense oligonucleotide, use or method according to claim 15, wherein the further therapeutic agent is an anti-CFD antibody therapeutic such as lampalizumab.
17. The antisense oligonucleotide, use or method according to claim 15, wherein the further therapeutic agent is a small molecule inhibitor of complement factor D, such as a molecule selected from the group consisting of ACH-4447, ACH-5228 & ACH-5548.
18. The antisense oligonucleotide, use or method according to any one of embodiments 1 - 8, wherein the further therapeutic agent is an antagonist of complement factor B.
19. The antisense oligonucleotide, use or method according to any one of embodiments 1 - 8, or 18 wherein the further therapeutic agent is an oligonucleotide antagonist of complement factor B, such as an antisense oligonucleotide or siRNA targeting complement factor B encoding RNA (e.g. a nucleic acid inhibitor of CFB which comprises a sequence of at least 12 or at least 14 contiguous nucleotides which are fully complementary to SEQ ID NO 242).
20. The antisense oligonucleotide, use or method according to embodiment 18 or 19,
wherein the further therapeutic agent is lONIS-FB-Lrx.
21. The antisense oligonucleotide, use or method according to any one of embodiments 1 - 8, wherein the further therapeutic agent is an antagonist of complement factor C3.
22. The antisense oligonucleotide, use or method according to embodiment 21 , wherein the further therapeutic agent is APL-2.
23. The antisense oligonucleotide, use or method according to embodiment 21 , wherein the further therapeutic agent is a modified MTSP-1 protease, such as CB2782.
24. The antisense oligonucleotide, use or method according to any one of embodiments 1 - 8, wherein the further therapeutic agent is an antagonist of complement factor C5 (CFC5), such as an anti-CFC5 antibody therapeutic.
25. The antisense oligonucleotide, use or method according to embodiment 24, wherein the further therapeutic agent is tesidolumab.
26. The antisense oligonucleotide, use or method according to embodiment 24, wherein the further therapeutic agent is avacincaptad pegol.
27. The antisense oligonucleotide, use or method according to embodiment 24, wherein the further therapeutic agent is eculizumab.
28. The antisense oligonucleotide, use or method according to any one of embodiments 1- 8, wherein the further therapeutic agent is an anti-platelet derived growth factor (PGDF), such as an anti-PGDF antibody, an aptamer inhibitor of PGDF.
EXAMPLES
Example 1. Testing in vitro efficacy of LNA oligonucleotides in U251 cell line at a single concentration.
Identification of promising“hot spot” region for HTRA1. A library of n=231 HTRA1 LNA oligonucleotides were screened in U251 cell line at 5mM, 6 days of treatment. From this library, we identified a series of active oligonucleotides targeting human HTRA1 pre-mRNA between position 531 13 - 53384 as shown in figure 1 (SEQ ID NO 1 16 or 1 17).
Human glioblastoma U251 cell line was purchased from ECACC and maintained as
recommended by the supplier in a humidified incubator at 37°C with 5% CO2. For assays, 15000 U251 cells/well were seeded in a 96 multi well plate in starvation media (media
recommended by the supplier with the exception of 1 % FBS instead of 10%). Cells were incubated for 24 hours before addition of oligonucleotides dissolved in PBS. Concentration of oligonucleotides: 5 mM. 3-4 days after addition of oligonucleotides, media was removed and new media (without oligonucleotide) was added. 6 days after addition of oligonucleotides, the cells were harvested. RNA was extracted using the PureLink Pro 96 RNA Purification kit (Ambion, according to the manufacturer’s instructions). cDNA was then synthesized using M- MLT Reverse Transcriptase, random decamers RETROscript, RNase inhibitor (Ambion, according the manufacturer’s instruction) with 100mM dNTP set PCR Grade (Invitrogen) and DNase/RNase free Water (Gibco). For gene expressions analysis, qPCR was performed using TagMan Fast Advanced Master Mix (2X) (Ambion) in a doublex set up. Following TaqMan primer assays were used for qPCR: HTRA1 , Hs01016151_m1 (FAM-MGB) and house keeping gene, TBP, Hs4326322E (VIC-MGB) from Life Technologies. n= 2 independent biological replicates. The residual HTRA1 mRNA expression level in the table is shown as % of control (PBS-treated cells).
Example 2.Testing in vitro efficacy of LNA oligonucleotides in U251 cell line at a single concentration.
The“hot spot” region 53113 - 53384 described in Example 1 was further validated in a new library of n=210 HTRA1 LNA oligonucleotides that were screened in U251 cell line at 5mM. n=33 LNA oligonucleotides were targeting human HTRA1 pre-mRNA between position 531 13 - 53384 and these oligos were relatively active in comparison to the rest as shown in figure 2.
The assay was performed as described in example 1. n= 2 independent biological replicates. The residual HTRA1 mRNA expression level is shown in the table as % of control (PBS-treated cells).
Example 3. Testing in vitro efficacy of LNA oligonucleotides in U251 and ARPE19 cell lines at a single concentration.
The“hot spot” region 531 13 - 53384 described in Example 1 and 2 was further validated in a new library of n=305 HTRA1 LNA oligonucleotides that were screened in U251 and ARPE19
cell lines at 5mM and 25mM, respectively. n=95 LNA oligonucleotides were targeting human HTRA1 pre-mRNA between position 531 13 - 53384 and these oligos were relatively active in comparison to the rest as shown in figure 3.
Human retinal pigmented epithelium ARPE19 cell line was purchased by from ATCC and maintained in DMEM-F12 (Sigma, D8437), 10% FBS, 1% pen/strep in a humidified incubator at 37°C with 5% CO2. The U251 cell line was described in example 1. For assays, 2000 U251 or ARPE19 cells/well were seeded in a 96 multi well plate in culture media recommended by the supplier. Cells were incubated for 2 hours before addition of oligonucleotides dissolved in PBS. Concentration of oligo was 5 and 25mM in U251 and ARPE19 cells, respectively. 4 days after addition of oligonucleotides, the cells were harvested. RNA extraction was performed as described in example 1 , cDNA synthesis and qPCR were performed using qScript XLT one-step RT-qPCR ToughMix Low ROX, 95134-100 (Quanta Biosciences). Following TaqMan primer assays were used for U251 and ARPE19 cells in a douplex set up: HTRA1 , Hs01016151_m1 (FAM-MGB) and house keeping gene, GAPDH, Hs4310884E (VIC-MGB). All primer sets were purchased from Life Technologies. n=1 biological replicate. The relative HTRA1 mRNA expression level in the table is shown as % of control (PBS-treated cells).
Compound A = SEQ ID NO 232
Example 4. Testing in vitro potency and efficacy of selected compounds in U251 and ARPE19 cell lines in a dose response curve.
The U251 and ARPE19 cell lines were described in example 1 and 3, respectively. The U251 assay was performed as described in Example 1. The ARPE19 assay was performed as
follows: 5000 ARPE19 cells/well were seeded in a 96 multi well plate in culture media recommended by the supplier (with the exception of 5% FBS instead of 10%). Cells were incubated for 2 hour before addition of oligonucleotides dissolved in PBS. Concentration of oligonucleotides: from 50mM, half-log dilution, 8 points. 4 days after addition of oligonucleotides, the cells were harvested. RNA extraction, cDNA synthesis and qPCR were performed as described in Example 1. n=2 independent biological replicates. The EC50 value and the residual HTRA1 mRNA level at 50mM are shown in the table as % of control (PBS).
Example 5, Testing in vitro potency and efficacy of selected compounds in U251 and ARPE19 cell lines in a dose response curve.
The assays were performed as described in Example 3. Concentration of oligonucleotides: from 50mM, half-log dilution, 8 points. n=2 and n=1 independent biological replicates for U251 and ARPE19, respectively. The EC50 value and the residual HTRA1 mRNA level at 50mM are shown in the table as % of control (PBS).
Compound A = SEQ ID NO 232
Compound B = SEQ ID NO 233 Example 6. Testing in vitro potency and efficacy of selected compounds in U251 cell line in a dose response curve.
The assay was performed as described in Example 3. Concentration of oligonucleotides: from 50mM, half-log dilution, 8 points. n=2 independent biological replicates. The EC50 value and the residual HTRA1 mRNA level at 50mM are shown in the table as % of control (PBS).
Example 7. Testing in vitro potency and efficacy of selected compounds in U251 cell line in a dose response curve.
The ARPE19 cell line was described in example 3. For assays, ARPE19 cells, 24000 cells/well were seeded in 100mI_ in a 96 multi well plate in starvation media (culture media as
recommended by the supplier with the exception of 1 % FBS instead of 10%). Cells were incubated for 2 hour before addition of oligonucleotides dissolved in PBS. Concentration of oligonucleotides: from 50mM, half-log dilution, 8 points. At day 4 and 7 after addition of oligonucleotide compounds 75mI_ fresh starvation media without oligonucleotides was added to the cells (without removing the old media). RNA extraction, cDNA synthesis and qPCR were performed as described in Example 3. n=2 independent biological replicates. The EC50 value and the residual HTRA1 mRNA level at 50mM are shown in the table as % of control (PBS).
Example 8.
Testing in vitro efficacy in human primary RPE cells.
Human primary Retinal Pigmented Epithelium (hpRPE) cells were purchased from Sciencell (Cat# 6540). For assays, 5000 hpRPE cells/well were seeded in a Laminin (Laminin 521 , BioLamina Cat# LN521-03) coated 96 multi well plate in culture media (EpiCM, Sciencell Cat# 4101 ). They were expanded with this media for one week and differentiated using the following media for 2 weeks : MEM Alpha media (Sigma Cat# M-4526) supplemented with N1
supplement (Sigma Cat# N-6530), Glutamine-Penicillin-Streptomycin (Sigma Cat# G-1 146),
Non Essential Amino Acid (NEAA, Sigma Cat# M-7145), Taurine (Sigma Cat# T-0625), Hydrocortisone (Sigma Cat# H-03966), Triiodo-thyronin (Sigma Cat# T-5516) and Bovine Serum Albumin (BSA, Sigma Cat# A-9647). Cells were cultured in a humidified incubator at 37°C with 5% C02.
On the day of the experiment, cells were incubated for 1 hour with fresh differentiation media before addition of oligonucleotides. These were dissolved in PBS and applied on cells at day 0 and day 4. On day 7, the media was changed, and on day 10 cells were harvested with 50mI of RLT buffer with b-mercapto-ethanol (Qiagen Cat# 79216). The extraction of the RNA was performed according to the user’s manual of the Qiagen RNeasy Mini Kit (Cat# 74104; Lot 151048073) including DNase I treatment (Cat# 79254; Lot 151042674). RNA quality control was performed with the Agilent Bioanalyzer Nano Kit (Agilent; Cat# 5067-1511 ; Lot 1446). Reverse transcription of total RNA into cDNA (cDNA synthesis) was performed using the High Capacity cDNA Reverse Transcription Kit (based on random hexamer oligonucleotides), according to the manufacturer’s instructions (Thermo Fisher Scientific, Cat# 4368814; Lot 00314158). The measurement of the cDNA samples was carried out in triplicates, in a 384-well plate format on the 7900HT real-time PCR instrument (Thermo Fisher Scientific). The following TaqMan primer assays were used for qPCR: HTRA1 , Hs01016151_m1 and Hs00170197_m1 , housekeeping genes, GAPDH, Hs99999905_m1 and PPIA, Hs99999904_m1 , from Life Technologies. n=3 biological replicates. The residual HTRA1 mRNA expression level is shown in figure 4 and the following table as % of control (PBS).
Example 9. Cynomolgus monkey in vivo pharmacokinetics and pharmacodynamics study, 21 days of treatment, intravitreal (IVT) injection, single dose.
Knock down was observed for 3 HTRA1 LNA oligonucleotides targeting the“hotspot” in human HTRA1 pre-mRNA between position 531 13 - 53384 both at mRNA in the retina and at protein level in the retina and in the vitreous (see figure 5)
Animals
All experiments were performed on Cynomolgus monkeys ( Macaca fascicularis).
Four animals were included in each group of the study, 20 in total.
Compounds and dosing procedures
Buprenorphine analgesia was administered prior to, and two days after test compound injection. The animals were anesthetized with an intramuscular injection of ketamine and xylazine. The test item and negative control (PBS) were administered intravitreally in both eyes of
anesthetized animals (50 pl_ per administration) on study day 1 after local application of tetracaine anesthetic.
Euthanasia
At the end of the in-life phase (Day 22) all monkeys were euthanized by intraperitoneal an overdose injection of pentobarbital.
Oligo content measurement and quantification of Htral RNA expression by qPCR
Immediately after euthanasia, eye tissues were quickly and carefully dissected out on ice and stored at -80°C until shipment. Retina sample was lysed in 700 pl_ MagNa Pure 96 LC RNA Isolation Tissue buffer and homogenized by adding 1 stainless steel bead per 2 ml tube 2 x 1 ,5 min using a precellys evolution homogenizer followed by 30 min incubation at RT. The samples were centrifuged, 13000 rpm, 5 min. Half was set aside for bioanalysis and for the other half, RNA extraction was continued directly.
For bioanalysis, the samples were diluted 10-50 fold for oligo content measurements with a hybridization ELISA method. A biotinylated LNA-capture probe and a digoxigenin-conjugated LNA-detection probe (both 35nM in 5xSSCT, each complementary to one end of the LNA oligonucleotide to be detected) was mixed with the diluted homogenates or relevant standards, incubated for 30 minutes at RT and then added to a streptavidine-coated ELISA plates (Nunc cat. no. 436014).
The plates were incubated for 1 hour at RT, washed in 2xSSCT (300mM sodium chloride,
30mM sodium citrate and 0,05% v/v Tween-20, pH 7.0) The captured LNA duplexes were detected using an anti-DIG antibodies conjugated with alkaline phosphatase (Roche Applied Science cat. No. 1 1093274910) and an alkaline phosphatase substrate system (Blue Phos substrate, KPL product code 50-88-00). The amount of oligo complexes was measured as absorbance at 615 nm on a Biotek reader.
For RNA extraction, cellular RNA large volume kit (05467535001 , Roche) was used in the MagNA Pure 96 system with the program: Tissue FF standard LV3.1 according to the instructions of the manufacturer, including DNAse treatment. RNA quality control and concentration were measured with an Eon reader (Biotek). The RNA concentration was normalized across samples, and subsequent cDNA synthesis and qPCR was performed in a one-step reaction using qScript XLT one-step RT-qPCR ToughMix Low ROX, 95134-100 (Quanta Biosciences). The following TaqMan primer assays were used in singplex reactions: Htral , Mf01016150_, Mf01016152_m1 and Rh02799527_m1 and housekeeping genes, ARFGAP2, Mf01058488_g1 and Rh01058485_m1 , and ARL1 , Mf02795431_m1 , from Life Technologies. The qPCR analyses were run on a ViiA7 machine (Life Technologies).
Eyes/group: n=3 eyes. Each eye was treated as an individual sample. The relative Htral mRNA expression level is shown as % of control (PBS).
Histology
Eyeballs were removed and fixed in 10% neutral buffered formalin for 24 hours, trimmed and embedded in paraffin.
For ISH analysis, sections of formalin-fixed, paraffin-embedded cyno retina tissue 4pm thick were processed using the fully automated Ventana Dicovery ULTRA Staining Module
(Procedure: mRNA Discovery Ultra Red 4.0 - vO.00.0152) using the RNAscope 2.5 VS Probe- Mmu-HTRA1 , REF 486979, Advanced Cell Diagnostics, Inc.. Chromogen used is Fastred, Hematoxylin II counterstain.
HTRA1 protein quantification using a plate-based immunoprecipitation mass
spectrometry (IP-MS) approach
Sample preparation, Retina
Retinas were homogenized in 4 volumes (w/v) of RIPA buffer (50 mM Tris-HCI, pH 7.4, 150 mM NaCI, 0.25% deoxycholic acid, 1% NP-40, 1 mM EDTA, Millipore) with protease inhibitors (Complete EDTA-free, Roche) using a Precellys 24 (5500, 15 s, 2 cycles). Homogenates were centrifuged (13,000 rpm, 3 min) and the protein contents of the supernatants determined (Pierce BCA protein assay)
Sample preparation, Vitreous
Vitreous humors (300 pi) were diluted with 5x RIPA buffer (final concentration: 50 mM Tris-HCI, pH 7.4, 150 mM NaCI, 0.25% deoxycholic acid, 1% NP-40, 1 mM EDTA) with protease inhibitors (Complete EDTA-free, Roche) and homogenized using a Precellys 24 (5500, 15 s, 2 cycles). Homogenates were centrifuged (13,000 rpm, 3 min) and the protein contents of the
supernatants determined (Pierce BCA protein assay)
Plate-based HTRA 1 immunoprecipitation and tryptic digest
A 96 well plate (Nunc MaxiSorp) was coated with anti-HTRA1 mouse monoclonal antibody (R&D MAB2916, 500 ng/well in 50 pi PBS) and incubated overnight at 4°C. The plate was washed twice with PBS (200 mI) and blocked with 3% (w/v) BSA in PBS for 30 min at 20 °C followed by two PBS washes. Samples (75 pg retina, 100 pg vitreous in 50 mI PBS) were randomized and added to the plate followed by overnight incubation at 4 °C on a shaker (150 rpm). The plate was then washed twice with PBS and once with water. 10 mM DTT in 50 mM TEAB (30 mI) were then added to each well followed by incubation for 1 h at 20 °C to reduce cysteine sulfhydryls. 150 mM iodoacetamide in 50 mM TEAB (5 mI) were then added to each well followed by incubation for 30 min at 20 °C in the dark in order to block cysteine sulfhydryls. 10 mI Digestion solution were added to each well (final concentrations: 1.24 ng/pl trypsin, 20 fmol/mI BSA peptides, 26 fmol/mI isotope-labeled HTRA1 peptides, 1 fmol/mI iRT peptides, Biognosys) followed by incubation overnight at 20 °C.
HTRA 1 peptide quantification by targeted mass spectrometry (selected reaction monitoring, SRM)
Mass spectrometry analysis was performed on an Ultimate RSLCnano LC coupled to a TSQ Quantiva triple quadrupole mass spectrometer (Thermo Scientific). Samples (20 pL) were injected directly from the 96 well plate used for IP and loaded at 5 pL/min for 6 min onto a
Acclaim Pepmap 100 trap column (100 pm x 2 cm, C18, 5 pm, 100 A, Thermo Scientific) in loading buffer (0.5% v/v formic acid, 2% v/v ACN). Peptides were then resolved on a PepMap Easy-SPRAY analytical column (75 pm x 15 cm, 3 pm, 100 A, Thermo Scientific) with integrated electrospray emitter heated to 40°C using the following gradient at a flow rate of 250 nL/min: 6 min, 98% buffer A (2% ACN, 0.1% formic acid), 2% buffer B (ACN + 0.1% formic acid); 36 min, 30% buffer B; 41 min, 60% buffer B; 43 min, 80% buffer B; 49 min, 80% buffer B; 50 min, 2% buffer B. The TSQ Quantiva was operated in SRM mode with the following parameters: cycle time, 1.5 s; spray voltage, 1800 V; collision gas pressure, 2 mTorr; Q1 and Q3 resolution, 0.7 FWHM; ion transfer tube temperature 300 °C. SRM transitions were acquired for the HTRA1 peptide“LHRPPVIVLQR” and an isotope labelled (L-[U-13C, U-15N]R) synthetic version, which was used an internal standard.
Data analysis was performed using Skyline version 3.6.
Western blot
Dissected retina sample in 0.5 Precellyses tubes (CK14_0.5ml, Bertin Technologies) were lysed and homogenized in RIPA lysis buffer (20-188, Milipore) with protease inhibitors (Complete EDTA-free Proteases-lnhibitor Mini, 1 1 836 170 001 , Roche).
Vitreous sample were added to a 0.5 Precellyses tubes (CK14_0.5ml, Bertin Technologies) were lysed and homogenized in 1/4x RIPA lysis buffer (20-188, Milipore) with protease inhibitors (Complete EDTA-free Proteases-lnhibitor Mini, 1 1 836 170 001 , Roche).
Samples (retina 20 pg protein, vitreous 40 pg protein) were analyzed on 4-15% gradient gel (#567-8084 Bio-Rad) under reducing conditions and transferred on Nitrocellulose (#170-4159 Bio-Rad) using a Trans-Blot Turbo Device from Bio-Rad.
Primary antibodies: Rabbit anti human HTRA1 (SF1 ) was a kind gift of Sascha Fauser
(University of Cologne), mouse anti human Gapdh (#98795 Sigma-Aldrich). Secondary antibody: goat anti rabbit 800CW and goat anti mouse 680RD were from Li-Cor
Blot was imaged and analyzed on an Odyssee CLX from Li-Cor.
Example 10 - Cynomolgus monkey in vivo Assessment: HTRA1 protein determination in aqueous humor and comparison to HTRA1 mRNA and protein inhibition in retina.
Experimental Methodology: See above example. Aqueous humor samples were taken and samples were prepared as according to example 9 vitreous humor samples. Cynomolgus Monkey Aqueous humor samples (AH) were analyzed with a size-based assay on a Analytical Methodology: Capillary Electrophoresis System (Peggy Sue™, Proteinsimple)
Samples were thawed on ice and used undiluted. For quantification, recombinant HTRA1- S328A mutant (Origene #TP700208). Preparation was as described by the provider.
Primary rabbit anti- human HTRA Antibody SF1 was provided by Prof. Dr. Sascha Fauser and used diluted 1 :300. All other reagents were from Proteinsimple.
Samples were processed in technical triplicate, calibration curve in duplicate using a 12 -230 kDa Separation module. Area under the peak was computed and analyzed using Xlfit (IDBS software).
Note - the compound IDs shown in figures 12 - 14 utilize a different numbering system as the rest of the examples. The above table provides the key to the numbering used figures 12 -14 as compared to that used in the previous examples and elsewhere herein. Figure 12A shows a visualization of the HTRA1 protein levels in the aqueous humor of monkeys administered with compounds B and #73,1 , with samples taken at days 3, 8, 15, and 22 post injection. Figure 12B provides the calibration curve used in calculating HTRA1 protein levels. Figure 12C provides the calculated HTRA1 levels from aqueous humor from individual animal was plotted against time post injection.
Figure 13 illustrates a direct correlation between the level of HTRA1 protein in the aqueous humor and the level of HTRA1 mRNA in the retina. Aqueous humor HTRA1 protein levels may therefore be used as a biomarker for HTRA1 retina mRNA levels or HTRA1 retinal mRNA inhibition.
Figure 14 illustrates that there is also a correlation between HTRA1 protein levels in retina and the HTRA1 protein levels in aqueous humor, although the correlation was not, in this experiment, as strong as the correlation between HTRA1 mRNA inhibition in the retina and HTRA1 protein levels in the aqueous humor, indicating that aqueous humor HTRA1 protein levels are particularly suited as biomarker for HTRA1 mRNA antagonists.
Claims
1 . An antisense oligonucleotide selected from the group selected from TTCtatctacgcaTTG (SEQ ID NO 67), CTTCttctatctacgcAT (SEQ ID NO 73), and TACTttaatagcTCAA (SEQ ID NO 86), wherein capital letters represent a beta-D-oxy LNA nucleosides, LNA C are 5-methyl C, lower case letters are DNA nucleosides, and all internucleoside linkages are phosphorothioate internucleoside linkages; for use in the treatment of geographic atrophy in a subject, wherein the treatment further comprises administering a further therapeutic agent to the subject.
2. The antisense oligonucleotide according to claim 1 wherein the antisense
oligonucleotide is TTCtatctacgcaTTG (SEQ ID NO 67)
3. The antisense oligonucleotide according to claim 1 wherein the antisense
oligonucleotide is CTTCttctatctacgcAT (SEQ ID NO 73),
4. The antisense oligonucleotide according to claim 1 wherein the antisense
oligonucleotide is TACTttaatagcTCAA (SEQ ID NO 86)
5. The use of an antisense oligonucleotide according to any one of claims 1 - 4, in the manufacture of a medicament for the treatment of geographic atrophy, wherein the medicament is for use in combination treatment with a further therapeutic.
6. A method for the treatment of geographic atrophy in a subject in need of treatment, said method comprising administering an effective amount of the antisense oligonucleotide according to any one of claims 1 - 4 to said subject, and further administering an effective amount of a further therapeutic agent, to the subject.
7. The antisense oligonucleotide, use or method according to any one of claims 1 - 6, wherein the further therapeutic agent is a vascular endothelial growth factor (VEGF) antagonist therapeutic.
8. The antisense oligonucleotide, use or method according to claim 7, wherein VEGF antagonist therapeutic is an anti-VEGF antibody or an anti-VEGF aptamer.
9. The antisense oligonucleotide, use or method according to claim 7, wherein the VEGF antagonist therapeutic is a small molecule inhibitor of VEGF.
10. The antisense oligonucleotide, use or method according to any one of claims 1 - 6, wherein the further therapeutic agent is an antagonist of the complement system (a complement targeted therapy), such as a complement factor D antagonist, a
complement factor B antagonist, complement factor C5 antagonist, complement factor C3 antagonist, complement factor P (properdin) antagonist, a complement factor I antagonist, and a complement factor H antagonist.
1 1. The antisense oligonucleotide, use or method according to claim 10, wherein antagonist of the complement system is an antagonist of complement factor D, such as an anti complement factor D antibody, a small-molecule inhibitor of complement factor D.
12. The antisense oligonucleotide, use or method according to claim 10, wherein antagonist of the complement system is an antagonist of complement factor C, such as
oligonucleotide inhibitor of complement factor B.
13. The antisense oligonucleotide, use or method according to claim 12, wherein the further therapeutic agent is an antisense oligonucleotide which comprises at least 14 contiguous nucleotides which are fully complementary to SEQ ID NO 242, wherein the contiguous nucleotide sequence comprises a modified nucleoside.
14. The antisense oligonucleotide, use or method according to claim 12, wherein the further therapeutic agent is an antisense oligonucleotide which comprises the sequence:
15. The antisense oligonucleotide, use or method according to claim 10, wherein the
antagonist of the complement system is an antagonist of properdin, such as an anti properdin antibody.
16. The antisense oligonucleotide, use or method according to claim 10, wherein antagonist of the complement system is an antagonist of complement factor C3, such as a cyclic peptide inhibitor of C3.
17. The antisense oligonucleotide, use or method according to claim 10, wherein antagonist of the complement system is an antagonist of complement factor C5, such as an anti-C5 antibody.
18. The antisense oligonucleotide, use or method according to claim 10, wherein antagonist of the complement system is an oligonucleotide inhibitor of complement factor C5, complement factor B or complement factor D.
19. The antisense oligonucleotide, use or method according to any one of claims 1- 6, wherein the further therapeutic agent is an anti-platelet derived growth factor (PGDF), such as an anti-PGDF antibody or an aptamer inhibitor of PGDF.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP18209218.9 | 2018-11-29 | ||
| EP18209218 | 2018-11-29 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020109343A1 true WO2020109343A1 (en) | 2020-06-04 |
Family
ID=64559571
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2019/082644 Ceased WO2020109343A1 (en) | 2018-11-29 | 2019-11-27 | Combination therapy for treatment of macular degeneration |
Country Status (2)
| Country | Link |
|---|---|
| TW (1) | TW202039842A (en) |
| WO (1) | WO2020109343A1 (en) |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3898617A4 (en) * | 2018-12-17 | 2022-08-17 | Achillion Pharmaceuticals, Inc. | TARGETED DOSING FOR THE TREATMENT OF COMPLEMENT-MEDIED DISORDERS |
| US11649223B2 (en) | 2015-08-26 | 2023-05-16 | Achillion Pharmaceuticals, Inc. | Amino compounds for treatment of immune and inflammatory disorders |
| US11649229B2 (en) | 2015-08-26 | 2023-05-16 | Achillion Pharmaceuticals, Inc. | Amide compounds for treatment of immune and inflammatory disorders |
| US11708351B2 (en) | 2017-03-01 | 2023-07-25 | Achillion Pharmaceuticals, Inc. | Aryl, heteroaryl, and heterocyclic pharmaceutical compounds for treatment of medical disorders |
| US11718626B2 (en) | 2017-03-01 | 2023-08-08 | Achillion Pharmaceuticals, Inc. | Macrocyclic compounds for treatment of medical disorders |
| US11807627B2 (en) | 2018-09-25 | 2023-11-07 | Achillon Pharmaceuticals, Inc. | Morphic forms of complement factor D inhibitors |
| US11814363B2 (en) | 2018-09-06 | 2023-11-14 | Achillion Pharmaceuticals, Inc. | Morphic forms of danicopan |
| US11926617B2 (en) | 2015-08-26 | 2024-03-12 | Achillion Pharmaceuticals, Inc. | Aryl, heteroaryl, and heterocyclic compounds for treatment of immune and inflammatory disorders |
| US12006307B2 (en) | 2017-03-01 | 2024-06-11 | Achillion Pharmaceuticals, Inc. | Pharmaceutical compounds for treatment of medical disorders |
| US12076319B2 (en) | 2017-08-02 | 2024-09-03 | Achillion Pharmaceuticals, Inc. | Therapeutic regimens for treatment of paroxysmal nocturnal hemoglobinuria |
| US12139497B2 (en) | 2015-08-26 | 2024-11-12 | Achillion Pharmaceuticals, Inc. | Aryl, heteroaryl, and heterocyclic compounds for treatment of medical disorders |
Citations (49)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1996030046A1 (en) | 1995-03-30 | 1996-10-03 | Genentech, Inc. | Vascular endothelial cell growth factor antagonists |
| WO1998039352A1 (en) | 1997-03-07 | 1998-09-11 | Takeshi Imanishi | Novel bicyclonucleoside and oligonucleotide analogues |
| WO1998045332A2 (en) | 1997-04-07 | 1998-10-15 | Genentech, Inc. | Humanized antibodies and methods for forming humanized antibodies |
| WO1998045331A2 (en) | 1997-04-07 | 1998-10-15 | Genentech, Inc. | Anti-vegf antibodies |
| WO1999014226A2 (en) | 1997-09-12 | 1999-03-25 | Exiqon A/S | Bi- and tri-cyclic nucleoside, nucleotide and oligonucleotide analogues |
| WO2000047599A1 (en) | 1999-02-12 | 2000-08-17 | Sankyo Company, Limited | Novel nucleosides and oligonucleotide analogues |
| WO2000066604A2 (en) | 1999-05-04 | 2000-11-09 | Exiqon A/S | L-ribo-lna analogues |
| WO2001023613A1 (en) | 1999-09-30 | 2001-04-05 | Isis Pharmaceuticals, Inc. | Human rnase h and oligonucleotide compositions thereof |
| WO2004046160A2 (en) | 2002-11-18 | 2004-06-03 | Santaris Pharma A/S | Amino-lna, thio-lna and alpha-l-oxy-ln |
| WO2006047325A1 (en) | 2004-10-21 | 2006-05-04 | Genentech, Inc. | Method for treating intraocular neovascular diseases |
| US7070959B1 (en) | 1999-06-08 | 2006-07-04 | Regeneron Pharmaceuticals, Inc. | Modified chimeric polypeptides with improved pharmacokinetic properties |
| WO2007031091A2 (en) | 2005-09-15 | 2007-03-22 | Santaris Pharma A/S | Rna antagonist compounds for the modulation of p21 ras expression |
| WO2007090071A2 (en) | 2006-01-27 | 2007-08-09 | Isis Pharmaceuticals, Inc. | 6-modified bicyclic nucleic acid analogs |
| WO2007103549A2 (en) | 2006-03-08 | 2007-09-13 | Archemix Corp. | Complement binding aptamers and anti-c5 agents useful in the treatment of ocular disorders |
| WO2007134181A2 (en) | 2006-05-11 | 2007-11-22 | Isis Pharmaceuticals, Inc. | 5'-modified bicyclic nucleic acid analogs |
| US7306799B2 (en) | 1999-06-08 | 2007-12-11 | Regeneron Pharmaceuticals, Inc. | Use of VEGF inhibitors for treatment of eye disorders |
| WO2008063932A2 (en) | 2006-11-10 | 2008-05-29 | Genentech, Inc. | Method for treating age-related macular degeneration |
| WO2008150729A2 (en) | 2007-05-30 | 2008-12-11 | Isis Pharmaceuticals, Inc. | N-substituted-aminomethylene bridged bicyclic nucleic acid analogs |
| WO2008154401A2 (en) | 2007-06-08 | 2008-12-18 | Isis Pharmaceuticals, Inc. | Carbocyclic bicyclic nucleic acid analogs |
| WO2009006478A2 (en) | 2007-07-05 | 2009-01-08 | Isis Pharmaceuticals, Inc. | 6-disubstituted bicyclic nucleic acid analogs |
| US7531173B2 (en) | 2005-02-02 | 2009-05-12 | Regeneron Pharmaceuticals, Inc. | Ophthalmic composition of a VEGF antagonist |
| WO2009067647A1 (en) | 2007-11-21 | 2009-05-28 | Isis Pharmaceuticals, Inc. | Carbocyclic alpha-l-bicyclic nucleic acid analogs |
| US7608261B2 (en) | 2006-06-16 | 2009-10-27 | Regeneron Pharmacuticals, Inc. | VEGF antagonist formulations suitable for intravitreal administration |
| WO2010036698A1 (en) | 2008-09-24 | 2010-04-01 | Isis Pharmaceuticals, Inc. | Substituted alpha-l-bicyclic nucleosides |
| WO2010077578A1 (en) | 2008-12-09 | 2010-07-08 | Isis Pharmaceuticals, Inc. | Bis-modified bicyclic nucleic acid analogs |
| WO2011017521A2 (en) | 2009-08-06 | 2011-02-10 | Isis Pharmaceuticals, Inc. | Bicyclic cyclohexose nucleic acid analogs |
| WO2011156202A1 (en) | 2010-06-08 | 2011-12-15 | Isis Pharmaceuticals, Inc. | Substituted 2 '-amino and 2 '-thio-bicyclic nucleosides and oligomeric compounds prepared therefrom |
| EP2402437A2 (en) | 2006-07-05 | 2012-01-04 | Catalyst Biosciences, Inc. | Protease screening methods and proteases identified thereby |
| WO2013022984A1 (en) | 2011-08-11 | 2013-02-14 | Isis Pharmaceuticals, Inc. | Selective antisense compounds and uses thereof |
| WO2013055998A1 (en) | 2011-10-14 | 2013-04-18 | Genentech, Inc. | ANTI-HtrA1 ANTIBODIES AND METHODS OF USE |
| WO2013154798A1 (en) | 2012-04-09 | 2013-10-17 | Isis Pharmaceuticals, Inc. | Tricyclic nucleic acid analogs |
| US8664362B2 (en) | 2008-03-03 | 2014-03-04 | NovelMed Therapeutics | Humanized and chimeric anti-properdin antibodies |
| WO2014078731A2 (en) | 2012-11-15 | 2014-05-22 | Apellis Pharmaceuticals, Inc. | Cell-reactive, long-acting, or targeted compstatin analogs and related compositions and methods |
| WO2015038939A2 (en) | 2013-09-13 | 2015-03-19 | Isis Pharmaceuticals, Inc. | Modulators of complement factor b |
| WO2015168635A2 (en) | 2014-05-01 | 2015-11-05 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulating complement factor b expression |
| US9220631B2 (en) | 2012-07-03 | 2015-12-29 | Novartis Ag | Syringe |
| US9359598B2 (en) | 2004-04-12 | 2016-06-07 | Catalyst Biosciences, Inc. | Mutant MT-SP1 proteases with altered substrate specificity or activity |
| WO2017012375A1 (en) | 2015-07-22 | 2017-01-26 | 惠州市神龙松材线虫病综合治理有限公司 | Pine needle organic fertilizer, preparation method therefor and use thereof |
| US9598446B2 (en) | 2014-02-25 | 2017-03-21 | Achillion Pharmaceuticals, Inc. | Amino compounds for treatment of complement mediated disorders |
| WO2017075173A2 (en) | 2015-10-30 | 2017-05-04 | Genentech, Inc. | Anti-factor d antibodies and conjugates |
| WO2017075252A1 (en) | 2015-10-30 | 2017-05-04 | Genentech, Inc. | Anti-factor d antibody variant conjugates and uses thereof |
| WO2018000205A1 (en) | 2016-06-28 | 2018-01-04 | 深圳狗尾草智能科技有限公司 | Question answering method and system based on multiple intents and multiple skill packets, and robot |
| WO2018002105A1 (en) * | 2016-07-01 | 2018-01-04 | F. Hoffmann-La Roche Ag | Antisense oligonucleotides for modulating htra1 expression |
| WO2018064221A1 (en) | 2016-09-27 | 2018-04-05 | Novelis Inc. | System for and method of threading a metal substrate on a rolling mill |
| WO2018075357A1 (en) | 2016-10-17 | 2018-04-26 | Duke University | PRODUCTION OF IMMUNOTOXIN D2C7-(scdsFv)-PE38KDEL |
| US10000516B2 (en) | 2015-08-26 | 2018-06-19 | Achillion Pharmaceuticals, Inc. | Phosphonate compounds for treatment of medical disorders |
| US10011612B2 (en) | 2015-08-26 | 2018-07-03 | Achillion Pharmaceuticals, Inc. | Aryl, heteroaryl, and heterocyclic compounds for treatment of medical disorders |
| US10092584B2 (en) | 2015-08-26 | 2018-10-09 | Achillion Pharmaceuticals, Inc. | Compounds for the treatment of medical disorders |
| WO2019121838A1 (en) * | 2017-12-21 | 2019-06-27 | F. Hoffmann-La Roche Ag | Companion diagnostic for htra1 rna antagonists |
-
2019
- 2019-11-27 WO PCT/EP2019/082644 patent/WO2020109343A1/en not_active Ceased
- 2019-11-28 TW TW108143360A patent/TW202039842A/en unknown
Patent Citations (54)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1996030046A1 (en) | 1995-03-30 | 1996-10-03 | Genentech, Inc. | Vascular endothelial cell growth factor antagonists |
| WO1998039352A1 (en) | 1997-03-07 | 1998-09-11 | Takeshi Imanishi | Novel bicyclonucleoside and oligonucleotide analogues |
| WO1998045332A2 (en) | 1997-04-07 | 1998-10-15 | Genentech, Inc. | Humanized antibodies and methods for forming humanized antibodies |
| WO1998045331A2 (en) | 1997-04-07 | 1998-10-15 | Genentech, Inc. | Anti-vegf antibodies |
| WO1999014226A2 (en) | 1997-09-12 | 1999-03-25 | Exiqon A/S | Bi- and tri-cyclic nucleoside, nucleotide and oligonucleotide analogues |
| WO2000047599A1 (en) | 1999-02-12 | 2000-08-17 | Sankyo Company, Limited | Novel nucleosides and oligonucleotide analogues |
| WO2000066604A2 (en) | 1999-05-04 | 2000-11-09 | Exiqon A/S | L-ribo-lna analogues |
| US7070959B1 (en) | 1999-06-08 | 2006-07-04 | Regeneron Pharmaceuticals, Inc. | Modified chimeric polypeptides with improved pharmacokinetic properties |
| US7374758B2 (en) | 1999-06-08 | 2008-05-20 | Regeneron Pharmaceuticals, Inc. | Modified chimeric polypeptides with improved pharmacokinetic properties and methods of using thereof |
| US7374757B2 (en) | 1999-06-08 | 2008-05-20 | Regeneron Pharmaceuticals, Inc. | Modified chimeric polypeptides with improved pharmacokinetic properties |
| US7306799B2 (en) | 1999-06-08 | 2007-12-11 | Regeneron Pharmaceuticals, Inc. | Use of VEGF inhibitors for treatment of eye disorders |
| WO2001023613A1 (en) | 1999-09-30 | 2001-04-05 | Isis Pharmaceuticals, Inc. | Human rnase h and oligonucleotide compositions thereof |
| WO2004046160A2 (en) | 2002-11-18 | 2004-06-03 | Santaris Pharma A/S | Amino-lna, thio-lna and alpha-l-oxy-ln |
| US9359598B2 (en) | 2004-04-12 | 2016-06-07 | Catalyst Biosciences, Inc. | Mutant MT-SP1 proteases with altered substrate specificity or activity |
| WO2006047325A1 (en) | 2004-10-21 | 2006-05-04 | Genentech, Inc. | Method for treating intraocular neovascular diseases |
| US7531173B2 (en) | 2005-02-02 | 2009-05-12 | Regeneron Pharmaceuticals, Inc. | Ophthalmic composition of a VEGF antagonist |
| WO2007031091A2 (en) | 2005-09-15 | 2007-03-22 | Santaris Pharma A/S | Rna antagonist compounds for the modulation of p21 ras expression |
| WO2007090071A2 (en) | 2006-01-27 | 2007-08-09 | Isis Pharmaceuticals, Inc. | 6-modified bicyclic nucleic acid analogs |
| WO2007103549A2 (en) | 2006-03-08 | 2007-09-13 | Archemix Corp. | Complement binding aptamers and anti-c5 agents useful in the treatment of ocular disorders |
| WO2007134181A2 (en) | 2006-05-11 | 2007-11-22 | Isis Pharmaceuticals, Inc. | 5'-modified bicyclic nucleic acid analogs |
| US7608261B2 (en) | 2006-06-16 | 2009-10-27 | Regeneron Pharmacuticals, Inc. | VEGF antagonist formulations suitable for intravitreal administration |
| US9290757B2 (en) | 2006-07-05 | 2016-03-22 | Catalyst Biosciences, Inc. | Protease screening methods and proteases identified thereby |
| EP2402437A2 (en) | 2006-07-05 | 2012-01-04 | Catalyst Biosciences, Inc. | Protease screening methods and proteases identified thereby |
| WO2008063932A2 (en) | 2006-11-10 | 2008-05-29 | Genentech, Inc. | Method for treating age-related macular degeneration |
| WO2008150729A2 (en) | 2007-05-30 | 2008-12-11 | Isis Pharmaceuticals, Inc. | N-substituted-aminomethylene bridged bicyclic nucleic acid analogs |
| WO2008154401A2 (en) | 2007-06-08 | 2008-12-18 | Isis Pharmaceuticals, Inc. | Carbocyclic bicyclic nucleic acid analogs |
| WO2009006478A2 (en) | 2007-07-05 | 2009-01-08 | Isis Pharmaceuticals, Inc. | 6-disubstituted bicyclic nucleic acid analogs |
| WO2009067647A1 (en) | 2007-11-21 | 2009-05-28 | Isis Pharmaceuticals, Inc. | Carbocyclic alpha-l-bicyclic nucleic acid analogs |
| US8664362B2 (en) | 2008-03-03 | 2014-03-04 | NovelMed Therapeutics | Humanized and chimeric anti-properdin antibodies |
| WO2010036698A1 (en) | 2008-09-24 | 2010-04-01 | Isis Pharmaceuticals, Inc. | Substituted alpha-l-bicyclic nucleosides |
| WO2010077578A1 (en) | 2008-12-09 | 2010-07-08 | Isis Pharmaceuticals, Inc. | Bis-modified bicyclic nucleic acid analogs |
| WO2011017521A2 (en) | 2009-08-06 | 2011-02-10 | Isis Pharmaceuticals, Inc. | Bicyclic cyclohexose nucleic acid analogs |
| WO2011156202A1 (en) | 2010-06-08 | 2011-12-15 | Isis Pharmaceuticals, Inc. | Substituted 2 '-amino and 2 '-thio-bicyclic nucleosides and oligomeric compounds prepared therefrom |
| EP2742135A1 (en) | 2011-08-11 | 2014-06-18 | Isis Pharmaceuticals, Inc. | Linkage modified gapped oligomeric compounds and uses thereof |
| WO2013022984A1 (en) | 2011-08-11 | 2013-02-14 | Isis Pharmaceuticals, Inc. | Selective antisense compounds and uses thereof |
| WO2013055998A1 (en) | 2011-10-14 | 2013-04-18 | Genentech, Inc. | ANTI-HtrA1 ANTIBODIES AND METHODS OF USE |
| WO2013154798A1 (en) | 2012-04-09 | 2013-10-17 | Isis Pharmaceuticals, Inc. | Tricyclic nucleic acid analogs |
| US9220631B2 (en) | 2012-07-03 | 2015-12-29 | Novartis Ag | Syringe |
| WO2014078731A2 (en) | 2012-11-15 | 2014-05-22 | Apellis Pharmaceuticals, Inc. | Cell-reactive, long-acting, or targeted compstatin analogs and related compositions and methods |
| WO2015038939A2 (en) | 2013-09-13 | 2015-03-19 | Isis Pharmaceuticals, Inc. | Modulators of complement factor b |
| US9598446B2 (en) | 2014-02-25 | 2017-03-21 | Achillion Pharmaceuticals, Inc. | Amino compounds for treatment of complement mediated disorders |
| US10106563B2 (en) | 2014-02-25 | 2018-10-23 | Achillion Pharmaecuticals, Inc. | Ether compounds for treatment of complement mediated disorders |
| WO2015168635A2 (en) | 2014-05-01 | 2015-11-05 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulating complement factor b expression |
| WO2017012375A1 (en) | 2015-07-22 | 2017-01-26 | 惠州市神龙松材线虫病综合治理有限公司 | Pine needle organic fertilizer, preparation method therefor and use thereof |
| US10011612B2 (en) | 2015-08-26 | 2018-07-03 | Achillion Pharmaceuticals, Inc. | Aryl, heteroaryl, and heterocyclic compounds for treatment of medical disorders |
| US10000516B2 (en) | 2015-08-26 | 2018-06-19 | Achillion Pharmaceuticals, Inc. | Phosphonate compounds for treatment of medical disorders |
| US10092584B2 (en) | 2015-08-26 | 2018-10-09 | Achillion Pharmaceuticals, Inc. | Compounds for the treatment of medical disorders |
| WO2017075252A1 (en) | 2015-10-30 | 2017-05-04 | Genentech, Inc. | Anti-factor d antibody variant conjugates and uses thereof |
| WO2017075173A2 (en) | 2015-10-30 | 2017-05-04 | Genentech, Inc. | Anti-factor d antibodies and conjugates |
| WO2018000205A1 (en) | 2016-06-28 | 2018-01-04 | 深圳狗尾草智能科技有限公司 | Question answering method and system based on multiple intents and multiple skill packets, and robot |
| WO2018002105A1 (en) * | 2016-07-01 | 2018-01-04 | F. Hoffmann-La Roche Ag | Antisense oligonucleotides for modulating htra1 expression |
| WO2018064221A1 (en) | 2016-09-27 | 2018-04-05 | Novelis Inc. | System for and method of threading a metal substrate on a rolling mill |
| WO2018075357A1 (en) | 2016-10-17 | 2018-04-26 | Duke University | PRODUCTION OF IMMUNOTOXIN D2C7-(scdsFv)-PE38KDEL |
| WO2019121838A1 (en) * | 2017-12-21 | 2019-06-27 | F. Hoffmann-La Roche Ag | Companion diagnostic for htra1 rna antagonists |
Non-Patent Citations (26)
| Title |
|---|
| BERGSTROM, CURRENT PROTOCOLS IN NUCLEIC ACID CHEMISTRY, 2009 |
| CHEMICAL ABSTRACTS, Columbus, Ohio, US; abstract no. 1491144-00-3 |
| DELEAVEYDAMHA, CHEMISTRY AND BIOLOGY, vol. 19, 2012, pages 937 |
| FLUITER ET AL., MOL. BIOSYST., vol. 10, 2009, pages 1039 |
| FREIERALTMANN, NUCL. ACID RES., vol. 25, 1997, pages 4429 - 4443 |
| FROM MORGAN ET AL., NAT. REV. DRUG DISCOVERY, 2015 |
| HANSEN ET AL., CHEM. COMM., 1965, pages 36 - 38 |
| HIRAO ET AL., ACCOUNTS OF CHEMICAL RESEARCH, vol. 45, 2012, pages 2055 |
| HOLASH ET AL., PNAS USA, vol. 99, 2002, pages 11393 - 98 |
| HOLDGATE ET AL., DRUG DISCOV TODAY, 2005 |
| HOLLIGERHUDSON, NATURE BIOTECHNOLOGY, vol. 23, 2005, pages 1126 - 1136 |
| MANGOS ET AL., J. AM. CHEM. SOC., vol. 125, 2003, pages 654 - 661 |
| MCTIGUE ET AL., BIOCHEMISTRY, vol. 43, pages 5388 - 5405 |
| MERGNYLACROIX, OLIGONUCLEOTIDES, vol. 13, 2003, pages 515 - 537 |
| MITSUOKA ET AL., NUCLEIC ACIDS RESEARCH, vol. 37, no. 4, 2009, pages 1225 - 1238 |
| MORITABIOORGANIC ET AL., MED.CHEM. LETT., vol. 12, pages 73 - 76 |
| RICKLINLAMBRIS, SEMIN. IMMUNOL., 2017 |
| RIELYMILLER, CLIN CANCER RES, vol. 13, 2007, pages 4623 - 7s |
| RUKOV ET AL., NUCL. ACIDS RES., vol. 43, 2015, pages 8476 - 8487 |
| SANTALUCIA, PROC NATL ACAD SCI USA., vol. 95, 1998, pages 1460 - 1465 |
| SETH ET AL., J. ORG. CHEM., vol. 75, no. 5, 2010, pages 1569 - 81 |
| SUGIMOTO ET AL., BIOCHEMISTRY, vol. 34, 1995, pages 11211 - 11216 |
| UHLMANN, CURR. OPINION IN DRUG DEVELOPMENT, vol. 3, no. 2, 2000, pages 293 - 213 |
| VESTER ET AL., BIOORG. MED. CHEM. LETT., vol. 18, 2008, pages 2296 - 2300 |
| WANSETH, J. MEDICAL CHEMISTRY, vol. 59, 2016, pages 9645 - 9667 |
| YASPAN ET AL., SCI TRANSL MED., vol. 9, no. 395, 21 June 2017 (2017-06-21), pages NCT02247479 |
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12139497B2 (en) | 2015-08-26 | 2024-11-12 | Achillion Pharmaceuticals, Inc. | Aryl, heteroaryl, and heterocyclic compounds for treatment of medical disorders |
| US11649223B2 (en) | 2015-08-26 | 2023-05-16 | Achillion Pharmaceuticals, Inc. | Amino compounds for treatment of immune and inflammatory disorders |
| US11649229B2 (en) | 2015-08-26 | 2023-05-16 | Achillion Pharmaceuticals, Inc. | Amide compounds for treatment of immune and inflammatory disorders |
| US12338230B2 (en) | 2015-08-26 | 2025-06-24 | Achillion Pharmaceuticals, Inc. | Aryl, heteroaryl, and heterocyclic compounds for treatment of immune and inflammatory disorders |
| US11926617B2 (en) | 2015-08-26 | 2024-03-12 | Achillion Pharmaceuticals, Inc. | Aryl, heteroaryl, and heterocyclic compounds for treatment of immune and inflammatory disorders |
| US11708351B2 (en) | 2017-03-01 | 2023-07-25 | Achillion Pharmaceuticals, Inc. | Aryl, heteroaryl, and heterocyclic pharmaceutical compounds for treatment of medical disorders |
| US11718626B2 (en) | 2017-03-01 | 2023-08-08 | Achillion Pharmaceuticals, Inc. | Macrocyclic compounds for treatment of medical disorders |
| US12297205B2 (en) | 2017-03-01 | 2025-05-13 | Achillion Pharmaceuticals, Inc. | Macrocyclic compounds for treatment of medical disorders |
| US12006307B2 (en) | 2017-03-01 | 2024-06-11 | Achillion Pharmaceuticals, Inc. | Pharmaceutical compounds for treatment of medical disorders |
| US12162860B2 (en) | 2017-03-01 | 2024-12-10 | Achillion Pharmaceuticals, Inc. | Aryl, heteroaryl, and heterocyclic pharmaceutical compounds for treatment of medical disorders |
| US12076319B2 (en) | 2017-08-02 | 2024-09-03 | Achillion Pharmaceuticals, Inc. | Therapeutic regimens for treatment of paroxysmal nocturnal hemoglobinuria |
| US11814363B2 (en) | 2018-09-06 | 2023-11-14 | Achillion Pharmaceuticals, Inc. | Morphic forms of danicopan |
| US11807627B2 (en) | 2018-09-25 | 2023-11-07 | Achillon Pharmaceuticals, Inc. | Morphic forms of complement factor D inhibitors |
| EP3898617A4 (en) * | 2018-12-17 | 2022-08-17 | Achillion Pharmaceuticals, Inc. | TARGETED DOSING FOR THE TREATMENT OF COMPLEMENT-MEDIED DISORDERS |
| US12239645B2 (en) | 2018-12-17 | 2025-03-04 | Achillion Pharmaceuticals, Inc. | Targeted dosing for the treatment of complement mediated disorders |
Also Published As
| Publication number | Publication date |
|---|---|
| TW202039842A (en) | 2020-11-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2020109343A1 (en) | Combination therapy for treatment of macular degeneration | |
| JP7416852B2 (en) | Antisense oligonucleotides for regulating HTRA1 expression | |
| KR102473431B1 (en) | Antisense nucleic acids | |
| KR101838308B1 (en) | Treatment of pyrroline-5-carboxylate reductase 1 (pycr1) related diseases by inhibition of natural antisense transcript to pycr1 | |
| KR101807324B1 (en) | Treatment of down syndrome gene related diseases by inhibition of natural antisense transcript to a down syndrome gene | |
| WO2019215175A1 (en) | Oligonucleotides for modulating myh7 expression | |
| US20220042022A1 (en) | Antisense oligonucleotides for modulating htra1 expression | |
| CN111512160B (en) | Companion diagnostics for HTRA1 RNA antagonists | |
| JP2024513131A (en) | oligonucleotide | |
| EP4149486A1 (en) | Complement component c1s inhibitors for treating a neurological disease, and related compositions, systems and methods of using same | |
| WO2020109344A1 (en) | Occular administration device for antisense oligonucleotides | |
| EP4137156A1 (en) | Prophylactic or therapeutic agent for at least one type of cancer selected from group consisting of pancreatic cancer, lung cancer, colorectal cancer, biliary tract cancer and liver cancer, prophylactic or therapeutic agent for said cancer which is used in combination drug in combination with said agent, combination drug comprising said agents, and method for screening for prophylactic or therapeutic agent for cancer | |
| JP2025522625A (en) | Oligonucleotides | |
| HK40021168A (en) | Antisense oligonucleotides for modulating htra1 expression | |
| TW202532088A (en) | Compositions and methods for inhibiting expression of huntingtin (htt) | |
| HK40084771A (en) | Rnai agents for inhibiting expression of pnpla3, pharmaceutical compositions thereof, and methods of use | |
| NZ749395A (en) | Antisense oligonucleotides for modulating htra1 expression | |
| HK40031713A (en) | Companion diagnostic for htra1 rna antagonists |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19808599 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 19808599 Country of ref document: EP Kind code of ref document: A1 |