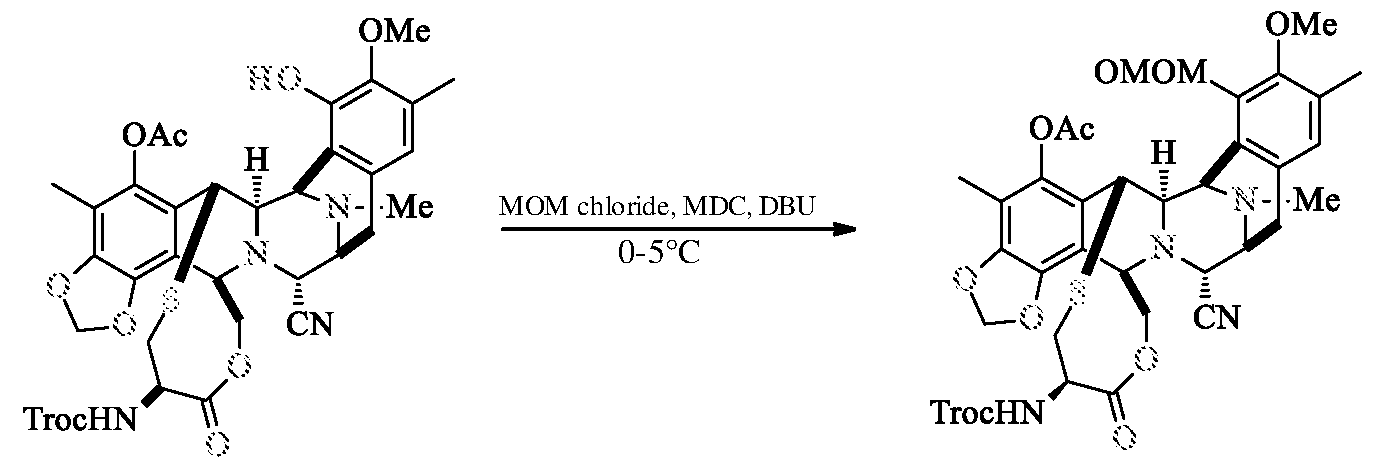WO2020105068A1 - Process for the preparation of ecteinascidin derivative and its intermediate - Google Patents
Process for the preparation of ecteinascidin derivative and its intermediateInfo
- Publication number
- WO2020105068A1 WO2020105068A1 PCT/IN2019/050862 IN2019050862W WO2020105068A1 WO 2020105068 A1 WO2020105068 A1 WO 2020105068A1 IN 2019050862 W IN2019050862 W IN 2019050862W WO 2020105068 A1 WO2020105068 A1 WO 2020105068A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- formula
- reacting
- trabectedin
- preparation
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 CC(OCC(c1c2OCOc2c(C)c(OCOC)c1)N(C(C*=C(C1)C=C(C)C(OC)=C(**)C2)C2[N-]C)[C@@]1C#N)=O Chemical compound CC(OCC(c1c2OCOc2c(C)c(OCOC)c1)N(C(C*=C(C1)C=C(C)C(OC)=C(**)C2)C2[N-]C)[C@@]1C#N)=O 0.000 description 3
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D515/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen, oxygen, and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00
- C07D515/22—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen, oxygen, and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00 in which the condensed system contains four or more hetero rings
Definitions
- the present invention relates to an improved and industrially viable process for the preparation of Trabectedin.
- the present invention also relates to an improved and industrially viable process for the preparation of Trabectedin Intermediate compound of formula-A.
- the present invention involves less expensive reagents, solvents and the process conditions can be easily adopted for commercial scale.
- Trabectedin (Ecteinascidin 743 or ET-743) has a complex tris(tetrahydroisoquinolinephenol) structure of the following formula.
- Cyanosafracin B is a pentacyclic antibiotic obtained by fermentation from the bacteria Pseudomonas fluorescence.
- US 8058435 also relates to the synthesis of Ecteinascidins-583 and 597 using intermediate compounds of formula.
- Ecteinascidin are a family of
- the HF is glass corrosive and handling it also difficult.
- the inventors of the present invention have developed an alternative improved process.
- the present process is simple, cost effective and feasible in large scale production.
- One aspect of the present invention is to prepare Trabectedin.
- One aspect of the present invention is to provide a process for the preparation of Trabectedin,
- Another aspect of the present invention is to provide a process for the preparation of Trabectedin
- Yet another aspect of the present invention is to provide a process for the preparation of a compound of formula- A comprising the step of reacting a compound of formula- B with Tetra-n-butyl ammonium fluoride and acidic reagent to get the compound of formula-A.
- One embodiment of the present invention is to provide a process for the preparation of Trabectedin, comprising the steps of:
- the catalyst is selected from DBU and DABCO.
- chlorinated solvent is selected from dichlorome thane, dichloroethane and chlorobenzene, preferably dichloromethane.
- the copper catalyst is selected from cuprous chloride, cuprous bromide, cuprous iodide, cuprous sulfate, preferably cuprous chloride.
- Yet another embodiment of the present invention is to provide a process for the preparation of a compound of formula- A comprising the step of reacting a compound of formula- B with Tetra-n-butyl ammonium fluoride and acidic reagent to get the compound of formula-A.
- acidic reagent is selected from Imidazole HC1, acetic acid, para toluene sulfonic acid.
- compound of formula-A is used in process for the preparation of compound of formula- VI accoriding to prior art process.
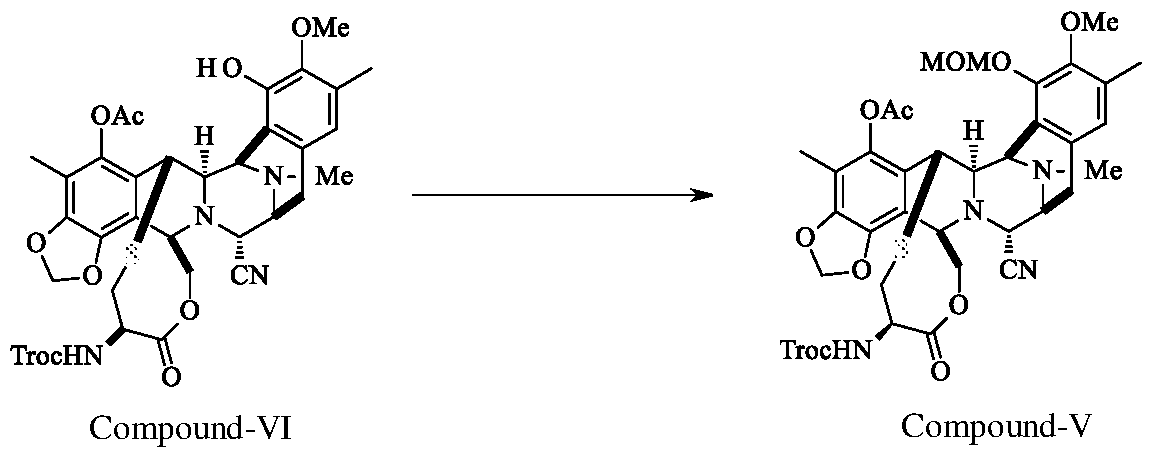
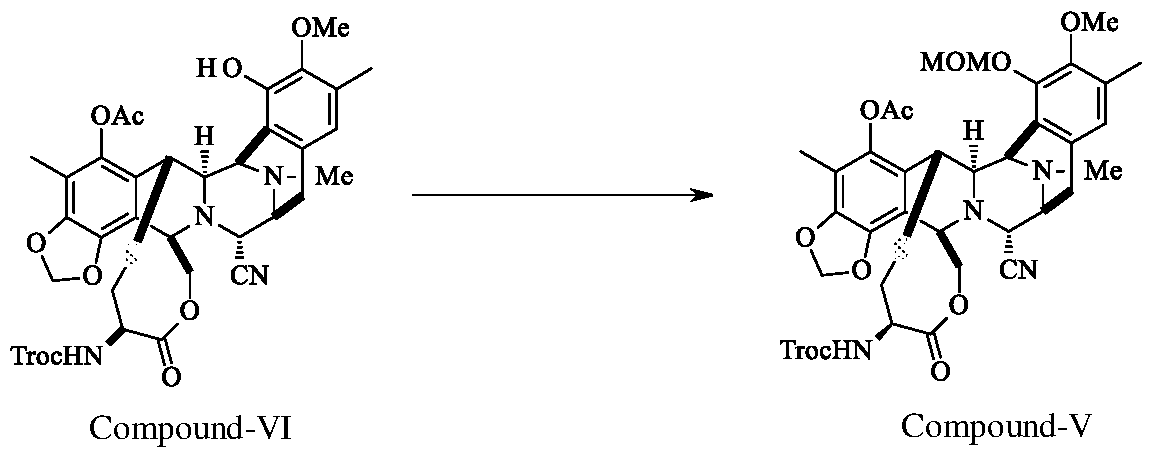
- Compound-VI Compound-V DBU (20.1 g) was added to the reaction mixture of Compound-VI (7.0 g), Chloro methyl methyl ether (19.5 g) in methylene chloride (25 ml) at 0-5°C. The reaction mass maintained at 0-5°C for 2.0 h. After completion of the reaction, washed the organic layer with citric acid solution, saturated sodium bicarbonate solution and water. The organic layer was concentrated, purified by flash column chromatography using hexane and ethyl acetate solvents (gradient from 1 :0 to 0.75:0.25) to yield Compound-V (5.5 g; 75.0%; Purity 99.2%).
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
The present invention relates to an improved and industrially viable process for the preparation of Trabectedin. The present invention also relates to an improved and industrially viable process for the preparation of Trabectedin Intermediate compound of formula-A. The present invention involves less expensive reagents, solvents and the process conditions can be easily adopted for commercial scale.
Description
“PROCESS FOR THE PREPARATION OF ECTEINASCIDIN DERIVATIVE
AND ITS INTERMEDIATE”
This patent application claims the benefit of our Indian provisional patent application No. 201841044299 filed on 24 November, 2018 and another provisional patent application No. 201841044298 filed on 24 November, 2018 which is incorporated by reference.
Field of the invention:
The present invention relates to an improved and industrially viable process for the preparation of Trabectedin. The present invention also relates to an improved and industrially viable process for the preparation of Trabectedin Intermediate compound of formula-A. The present invention involves less expensive reagents, solvents and the process conditions can be easily adopted for commercial scale.
Trabectedin Compound of formula- A
Background of the Invention:
Trabectedin (Ecteinascidin 743 or ET-743) has a complex tris(tetrahydroisoquinolinephenol) structure of the following formula.
It was approved under the brand name Y ONDELIS® and indicated for the treatment of patients with unresectable or metastatic liposarcoma or leiomyosarcoma. It was originally prepared by isolation from extracts of the marine tunicate Ecteinascidia turbinata. The yield was low, and alternative preparative processes had been sought.
The first synthetic process for producing ecteinascidin compounds was described in U.S. Pat. No. 5,721,362. This process employed sesamol as starting material and yielded ET-743 after a long and complicated sequence of 38 examples each describing one or more steps in the synthetic sequence.
An improvement in the preparation of one intermediate used in such process was disclosed in U.S. Pat. No. 6,815,544. Even with this improvement, the total synthesis was not suitable for manufacturing ET-743 at an industrial scale.
A semi synthetic process for producing ecteinascidin compounds was described in EP 1185536. This process employs cyanosafracin B as starting material to provide ET- 743. Cyanosafracin B is a pentacyclic antibiotic obtained by fermentation from the bacteria Pseudomonas fluorescence.
US 8058435 also relates to the synthesis of Ecteinascidins-583 and 597 using intermediate compounds of formula.
The inventors of the present invention have developed an alternative improved process. The present process is simple, cost effective and feasible in large scale production. Compound of formula-A is key intermediate useful in the synthesis of Ecteinascidin such as Ecteinascidin 743, Ecteinascidin 736, Ecteinascidin 729, Ecteinascidin 722, Ecteinascidin 637, and Ecteinascidin 594. Ecteinascidin are a family of
tetrahydroisoquinoline alkaloids isolated from the Caribbean tunicate Ecteinascidia turbinate, which possess potent cytotoxic activity against a variety of tumors.
Compound of formula-A
J. AM. CHEM. SOC. 2006, 128, 87-89 has reported the process for the preparation of compound formula -A. According to the process given in this reference, compound of formula - B is dissolved in acetonitrile and reacted with HF, after completion of reaction dichloromethane was added and isolate the compound of formula - A as a residue and the residue was purified by flash column chromatography. Compound of formula - A is used as a key intermediate in the synthesis of Trabectedin.
Compound of formula- B Compound of formula- A
In this process, the HF is glass corrosive and handling it also difficult.
The inventors of the present invention have developed an alternative improved process. The present process is simple, cost effective and feasible in large scale production.
Summary of the Invention:
One aspect of the present invention is to prepare Trabectedin. One aspect of the present invention is to provide a process for the preparation of Trabectedin,
comprising the steps of:
a) reacting compound of formula VI with chloro methyl methyl ether in presence of catalyst and a chlorinated solvent to get the compound of formula V,
c) reacting the compound of formula IV with 4-formyl- 1-methylpyridinium benzenesulfonate to get the compound of formula III,
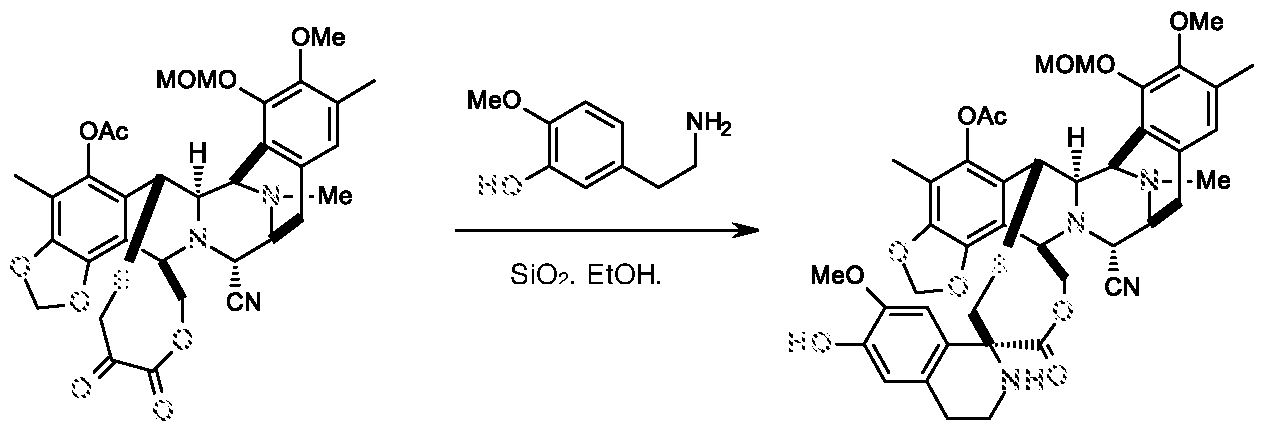
d) reacting the compound of formula III with 3-hydroxy-4-methoxyphenethyl amine to get compound of formula-II,
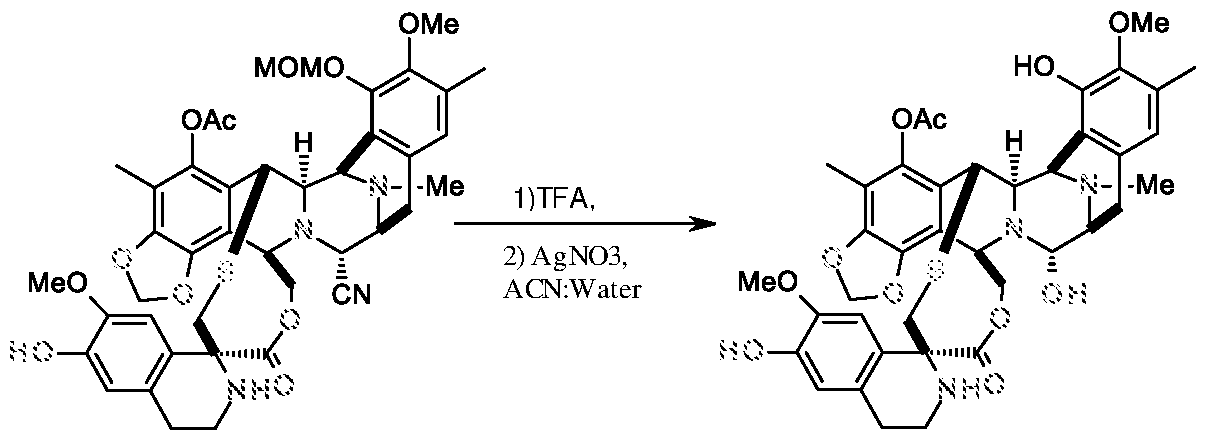
e) reacting the compound of formula II with tri fluoro acetic acid followed by silver nitrate to get the Trabectedin compound of formula-I,
Compound-II Compound-I
f) Optionally recrystallizing the Trabectedin from ethanol to get the pure Trabectedin. Another aspect of the present invention is to provide a process for the preparation of Trabectedin
comprising the steps of:
a) reacting compound of formula VI with chloro methyl methyl ether in presence of catalyst and a chlorinated solvent to get the compound of formula V,
Compound-VI Compound-V
b) reacting compound of formula-V with Zinc powder to get the compound of formula-IV,
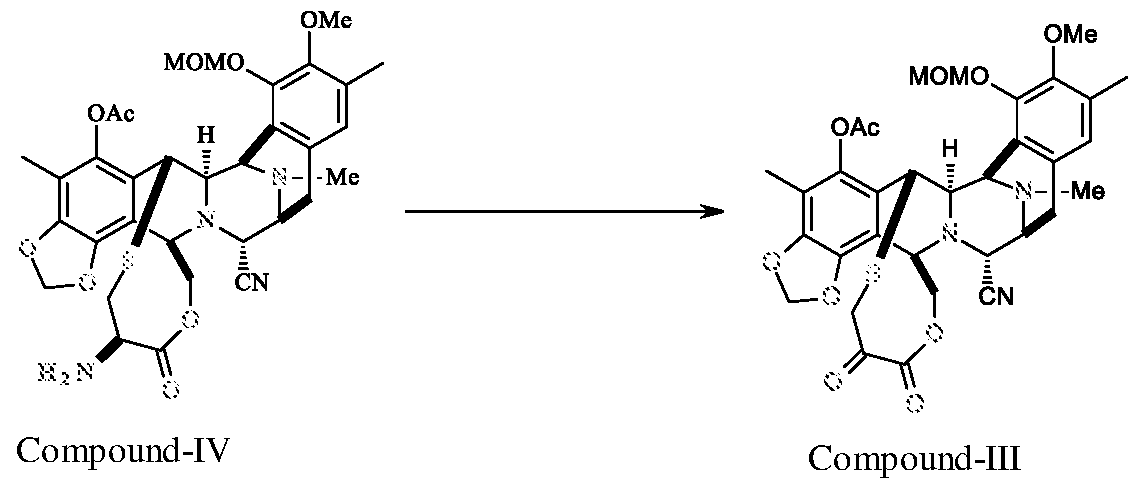
c) reacting the compound of formula IV with sodium glyoxylate or Magnesium glyoxylate to get the compound of formula III,
d) reacting the compound of formula III with 3-hydroxy-4-methoxyphenethyl amine to get compound of formula-II,
e) reacting the compound of formula II with tri fluoro acetic acid followed by copper catalyst to get the Trabectedin compound of formula-I,
Compound-II Compound-I
f) Optionally recrystallizing the Trabectedin from ethanol to get the pure Trabectedin. Yet another aspect of the present invention is to provide a process for the preparation of a compound of formula- A comprising the step of reacting a compound of formula- B with Tetra-n-butyl ammonium fluoride and acidic reagent to get the compound of formula-A.
Detailed description of the Invention:
One embodiment of the present invention is to provide a process for the preparation of Trabectedin, comprising the steps of:
a) reacting compound of formula VI with chloro methyl methyl ether in presence of catalyst and a chlorinated solvent to get the compound of formula V,
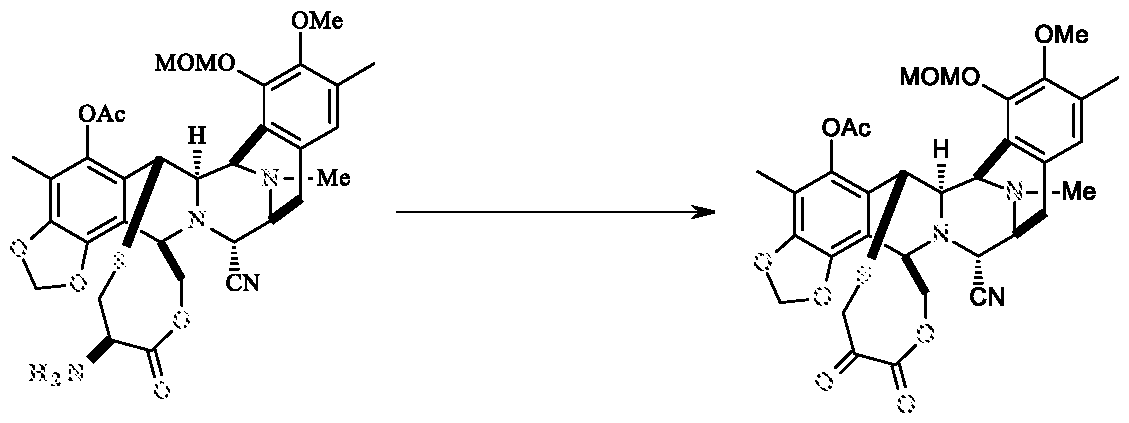
b) reacting compound of formula-V with Zn-Cu powder to get the compound of formula-IV,
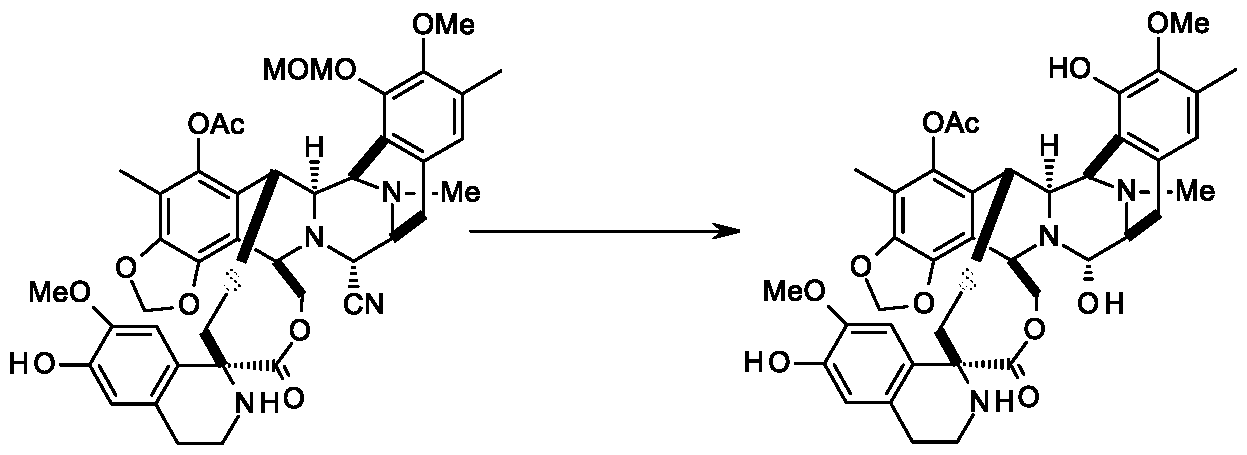
c) reacting the compound of formula IV with 4-formyl- 1-methylpyridinium benzenesulfonate to get the compound of formula III,
Compound-IV Compound-Ill d) reacting the compound of formula III with 3-hydroxy-4-methoxyphenethyl amine to get compound of formula-II,
e) reacting the compound of formula II with tri fluoro acetic acid followed by silver nitrate to get the Trabectedin compound of formula-I,
Compound-II Compound-I
f) optionally recrystallizing the Trabectedin from ethanol to get the pure Trabectedin.
Another embodiment of the present invention is to provide a process for the preparation of Trabectedin comprising the steps of:
a) reacting compound of formula VI with chloro methyl methyl ether in presence of catalyst and a chlorinated solvent to get the compound of formula V,
c) reacting the compound of formula IV with sodium glyoxylate or Magnesium glyoxylate to get the compound of formula III,
Compound-IV Compound-Ill d) reacting the compound of formula III with 3-hydroxy-4-methoxyphenethyl amine to get compound of formula-II,
e) reacting the compound of formula II with tri fluoro acetic acid followed by copper catalyst to get the Trabectedin compound of formula-I,
Compound-II Compound-I
f) optionally recrystallizing the Trabectedin from ethanol to get the pure Trabectedin. According to the present invention, wherein the catalyst is selected from DBU and DABCO.
According to the present invention, wherein the chlorinated solvent is selected from dichlorome thane, dichloroethane and chlorobenzene, preferably dichloromethane.
According to the present invention, wherein the copper catalyst is selected from cuprous chloride, cuprous bromide, cuprous iodide, cuprous sulfate, preferably cuprous chloride. Yet another embodiment of the present invention is to provide a process for the preparation of a compound of formula- A comprising the step of reacting a compound of formula- B with Tetra-n-butyl ammonium fluoride and acidic reagent to get the compound of formula-A.
According to the present embodiment, wherein acidic reagent is selected from Imidazole HC1, acetic acid, para toluene sulfonic acid.
According to the Present invention, compound of formula-A is used in process for the preparation of compound of formula- VI accoriding to prior art process.
Advantages of the present Invention:
• High Pure Trabectedin was achieved with the present process.
• Yields were high compare to prior art process.
• HF is glass corrosive and handling also difficult, the present process avoids the use of HF.
The Present invention is further illustrated in detail with reference to following examples. It is desired that the examples be considered in all respect as illustrative and are not intended to limit the scope of the invention in any way.
Examples:
Example-1: Preparation of compound of formula-V
Compound-VI Compound-V DBU (20.1 g) was added to the reaction mixture of Compound-VI (7.0 g), Chloro methyl methyl ether (19.5 g) in methylene chloride (25 ml) at 0-5°C. The reaction mass maintained at 0-5°C for 2.0 h. After completion of the reaction, washed the organic layer with citric acid solution, saturated sodium bicarbonate solution and water. The organic layer was concentrated, purified by flash column chromatography using hexane
and ethyl acetate solvents (gradient from 1 :0 to 0.75:0.25) to yield Compound-V (5.5 g; 75.0%; Purity 99.2%).
Example-2: Preparation of compound of formula-IV
Compound- V Compound-IV
To a solution of Compound-V (1.2 g) in a mixture of acetic acid/diethyl ether (1:2, 72 ml) was added Zn-Cu powder (7.8 g) at 25-30°C. The reaction mass was stirred for 2.5 h at 25-30°C, diluted with ethyl acetate (600 ml), filtered and organic layer was washed with saturated sodium bicarbonate solution and brine solution. The organic layer was concentrated, purified by flash column chromatography using hexane and ethyl acetate solvents (gradient from 1:0 to 0.2:0.8) to yield compound -IV (0.58 g; 61.0%).
Example-3: Preparation of compound of formula-III
To a solution of Compound-IV (0.58 g) in a mixture of Dimethyl formamide/dichloro methane (1: 1, 77 ml) was added 4-formyl- 1-methylpyridinium benzenesulfonate (2.43 g) at 25-30°C for 20 min. To the solution, DBU (1.19 g) was added, and the black suspension was stirred at 20-25 °C for 15 min before saturated oxalic acid solution (23
ml) was added. The mixture was stirred at 20-25 °C for 30 min, diluted with ethyl acetate (750 ml), washed with saturated sodium bicarbonate solution and water. The organic layer was concentrated, purified by flash column chromatography using hexane and ethyl acetate solvents (gradient from 1 :0 to 0.75:0.25) to yield Compound-Ill (0.21 g; 36.0%).
Example-4: Preparation of compound of formula-II
Compound-Ill Compound-II
To a solution of Compound-Ill (0.21 g), 3-hydroxy-4-methoxyphenethyl amine (0.186 g) and silicagel (0.46 g) in anhydrous ethanol was added and stirred at 20-25 °C for 3.0h.The reaction mixture was diluted with methylene chloride (70 ml) and filtered. The filtrate was concentrated and the residue was purified by flash column chromatography using hexane and ethyl acetate solvents (gradient from 1:0 to 0.5:0.5) to yield Compound-II (0.145 g; 56.0%).
Example-5: Preparation of Trabectedin
Compound-II Compound-I
Compound-II (0.05 g) was dissolved in a mixture of tri fluoro acetic acid/THF/water (4: 1 : 1, 50 ml), and the solution was stirred at 20-25 °C for 16 h and concentrated. The
residue was dissolved in a mixture of acetonitrile and water (3:2, 15 ml) and to this solution silver nitrate (0.214 g) was added. The reaction mass was stirred at 20-25 °C for 18 h, diluted with methylene chloride (30 ml), saturated sodium bicarbonate solution (20 ml) and brine solution (20 ml). The organic layer was separated, concentrated to yield Trabectedin. (0.039 g; with 86.4% purity).
Example-6: Preparation of compound of formula-IV
Compound-V (5.0 g) was dissolved in mixture of THF and methanol solution (250.0 ml) and a freshly prepared buffer solution 40 ml (prepared by dissolving 7.35 g of H3PO4 and 13.6 g of KH2PO4 in 100 ml of water) was added 310 g of Zn-dust to the reaction system at 25-30°C. The reaction mass was stirred for 10-15 min at 20-25°C, diluted with MDC (1000 ml) and 8% sodium bicarbonate solution, organic layer was separated and washed with brine solution. The organic layer was concentrated, purified by flash column chromatography using MDC and methanol solvents to yield compound -IV (3.0 g; 75.0%; Purity: - 99.5%).
Alternative Preparation of compound of formula-IV
Compound-V (50 mg) was dissolved in methanol (2.0 ml) and a freshly prepared buffer solution (prepared by dissolving 36.0 mg of H3PO4 and 54.0 mg of KH2PO4 in 0.4 ml of water) was added 320.0 mg of Zn-dust to the reaction system at 25-30°C. The reaction mass was stirred for 30 min at 20-25 °C, diluted with ethyl acetate (25 ml), filtered and organic layer was washed with saturated sodium bicarbonate solution and brine solution. The organic layer was concentrated to yield compound -IV (40.0 mg).
Example-7: Preparation of compound of formula-III
Compound-IV Compound-in
Compound-IV (0.15 g) was dissolved in acetonitrile (2.5 ml) and a freshly prepared buffer solution (prepared by dissolving 15.6 mg of NaOAc in 2.5 ml of AcOH) and anhydrous zinc sulfate (18.0 mg) was added 0.45 g of sodium glyoxylate to the reaction system at 25-30°C. The reaction mass was stirred for 40 min at 20-25°C, diluted with MDC (15.0 ml), organic layer was washed with water. The organic layer was concentrated, and the crude compound was isolated from hexane to yield compound - III (0.12 g; 80.0%).
Alternative Preparation of compound of formula-III
Compound-IV (0.7 g) was dissolved in acetonitrile (11.5 ml) and a freshly prepared buffer solution (prepared by dissolving 71.9 mg of NaOAc in 11.5 ml of AcOH) and anhydrous zinc sulfate (84.0 mg) was added 2.15 g of magnesium glyoxylate to the reaction system at 20-25°C. The reaction mass was stirred for 60 min at 20-25°C, diluted with MDC (70.0 ml), organic layer was washed with water. The organic layer was concentrated, and the crude compound was isolated from hexane to yield compound -III (0.6 g; 85.8%).
Compound-Ill Compoimd-II
To a solution of Compound-Ill (30.0 mg), 3-hydroxy-4-methoxyphenethyl amine (30.0 mg) and NaOAc (36.0 mg) in anhydrous ethanol was added and stirred at 20-25°C for 3.0h.The reaction mixture was diluted with methylene chloride (70 ml) and filtered. The filtrate was concentrated and the residue was purified by flash column chromatography using hexane and ethyl acetate solvents to yield Compound-II (20 mg; 55%).
Alternation Preparation of compound of formula-II
Compound-Ill Compound-II
To a solution of Compound-Ill (2.5 g), 3-hydroxy-4-methoxyphenethyl amine (0.94 g) and AcOH (0.385 g) in ethanol was added and stirred at 20-25°C for 5.0h. The reaction mixture was added to pre cooled water (300 ml) and stirred for 45 min at 0-5°C. Solid was filtered, wet compound was dissolved in MDC and distilled. Crude compound was purified by flash column chromatography using hexane and ethyl acetate solvents to yield Compound-II (2.5 g; 81.6%; Purity: - 99.0%).
Example-9: Preparation of Trabectedin
Compound-II Compound-I
Compound-II (0.08 g) was dissolved in a mixture of tri fluoro acetic acid/THF/water (4: 1 : 1, 80 ml), and the solution was stirred at 20-25 °C for 16 h and concentrated. The residue was dissolved in a mixture of THF and water (3:2, 4 ml) and to this CuCl (0.1 g) was added. The reaction mass was stirred at 20-25 °C for 22 h, diluted with methylene chloride (30 ml), saturated sodium bicarbonate solution (20 ml) and brine solution (20 ml). The organic layer was separated, concentrated to yield Trabectedin. (0.09 g).
Example-10: Purification of Trabectedin
A solution of Trabectedin (1.1 g), in anhydrous ethanol (19.0 ml) was added to distilled water (330.0 ml) at 20-25 °C and stirred for 30 min at 15-20°C. The product was filtered, washed and dried to yield pure Trabectedin (1.0 g with 99.8% purity).
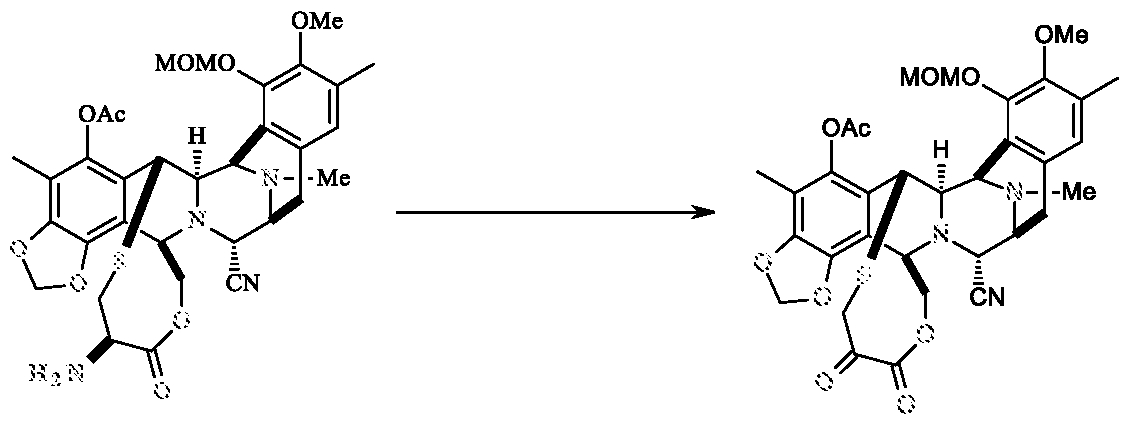
Example-11: Preparation of compound of formula-A
Compound of formula- B ^ , A
Compound of formula-A
To a solution of compound of formula-B (20.0 g in 100.0 ml THF) in a mixture of 13.0 gm of TBAF, Imidazole HC1 (2.6 g) in THF (100 ml) was added at 25-30°C. The reaction mass was stirred for 24-26 h at 25-30°C.The reaction mixture was diluted with methylene chloride (400 ml) and washed with 5% sodium bicarbonate solution and brine solution. The organic layer was concentrated, purified by flash column chromatography using hexane and ethyl acetate solvents (gradient from 1:0 to 0.25:0.75) to yield compound of formula-A (15.0 g; 87.0%).
Example-12: Preparation of compound of formula-A
Compound of formula- B , A
^ Compound ot formula- A
TBAF (2.44 ml) was added to the reaction mixture of compound of formula-B (1.0 g) and acetic acid (0.074 g) in THF (10 ml) at 10°C. The reaction mass was allowed to ambient temperature and stirred for 24h.The reaction mixture was diluted with methylene chloride (20 ml) and washed with 10% ammonium chloride solution, water and brine solution. The organic layer was concentrated, purified by flash column chromatography using hexane and ethyl acetate solvents (gradient from 1:0 to 0.25:0.75) to yield compound of formula-A (0.58 g; 67.0%).
Example-13: Preparation of compound of formula-A
Compound of formula- B ^ , .
1 Compound or formula- A
TBAF (1.25 ml) was added to the reaction mixture of compound of formula-B (0.5 g) and para toluene sulfonic acid (0.106 g) in THF (5 ml) at 20-25 °C. The reaction mass was allowed to ambient temperature and stirred for 21h.The reaction mixture was diluted with methylene chloride (20 ml) and washed with 5% ammonium chloride solution and brine solution. The organic layer was concentrated, to yield compound of formula-A (0.8 g with 84% purity).
Claims
1. A process for the preparation of Trabectedin comprising the steps of:
a) reacting compound of formula VI with chloro methyl methyl ether in presence of catalyst and a chlorinated solvent to get the compound of formula V,
b) reacting compound of formula-V with Zn-Cu powder to get the compound of formula-IV,
c) reacting the compound of formula IV with 4-formyl- 1-methylpyridinium benzenesulfonate to get the compound of formula III,
Compound-IV Compound-Ill d) reacting the compound of formula III with 3-hydroxy-4-methoxyphenethyl amine to get compound of formula-II,
e) reacting the compound of formula II with tri fluoro acetic acid followed by silver nitrate to get the Trabectedin compound of formula-I,
Compound-II Compound-I
f) optionally recrystallizing the Trabectedin from ethanol to get the pure
Trabectedin.
2. A process for the preparation of Trabectedin comprising the steps of:
a) reacting compound of formula VI with chloro methyl methyl ether in presence of catalyst and a chlorinated solvent to get the compound of formula V,
c) reacting the compound of formula IV with sodium glyoxylate or Magnesium glyoxylate to get the compound of formula III,
Compound-IV Compound-Ill d) reacting the compound of formula III with 3-hydroxy-4-methoxyphenethyl amine to get compound of formula-II,
e) reacting the compound of formula II with tri fluoro acetic acid followed by copper catalyst to get the Trabectedin compound of formula-I,
Compound-II Compound-I
f) optionally recrystallizing the Trabectedin from ethanol to get the pure Trabectedin.
3. The process as claimed in Claim 1 and 2, wherein the catalyst is selected from DBU and DABCO.
4. The process as claimed in Claim 1 and 2, wherein the chlorinated solvent is selected from dichloromethane, dichloroethane and chlorobenzene.
5. The process as claimed in Claim 2, wherein the copper catalyst is selected from cuprous chloride, cuprous bromide, cuprous iodide, cuprous sulfate.
6. A process for the preparation of a compound of formula- A comprising the step of reacting a compound of formula-B with Tetra-n-butyl ammonium fluoride and acidic reagent to get the compound of formula-A.
Compound of formula-B
Compound of formula- A
7. The process as claimed in Claim 6, wherein the acidic reagent is selected from Imidazole HC1, acetic acid, para toluene sulfonic acid.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN201841044298 | 2018-11-24 | ||
| IN201841044299 | 2018-11-24 | ||
| IN201841044299 | 2018-11-24 | ||
| IN201841044298 | 2018-11-24 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020105068A1 true WO2020105068A1 (en) | 2020-05-28 |
Family
ID=70773366
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IN2019/050862 Ceased WO2020105068A1 (en) | 2018-11-24 | 2019-11-22 | Process for the preparation of ecteinascidin derivative and its intermediate |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2020105068A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023031958A1 (en) * | 2021-08-31 | 2023-03-09 | Natco Pharma Limited | An improved process for the preparation of trabectedin |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1289999B1 (en) * | 2000-05-15 | 2004-11-17 | Pharma Mar, S.A. | Antitumoral analogs of et-743 |
| WO2017219917A1 (en) * | 2016-06-20 | 2017-12-28 | 浙江海正药业股份有限公司 | Method for preparing trabectedin and intermediates thereof |
-
2019
- 2019-11-22 WO PCT/IN2019/050862 patent/WO2020105068A1/en not_active Ceased
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1289999B1 (en) * | 2000-05-15 | 2004-11-17 | Pharma Mar, S.A. | Antitumoral analogs of et-743 |
| WO2017219917A1 (en) * | 2016-06-20 | 2017-12-28 | 浙江海正药业股份有限公司 | Method for preparing trabectedin and intermediates thereof |
Non-Patent Citations (2)
| Title |
|---|
| "SK , Silyl protective groups", 8 March 2014 (2014-03-08), Retrieved from the Internet <URL:https://en.chem-station.com/reactions-2/2014/03/silyl-protective-groups.html> [retrieved on 20200217] * |
| MELANIE REICH, DEPROTECTION OF A TERT-BUTYLDIMETHYLSILYL ETHER ; ALCOHOL, SYNTHETIC, 24 August 2001 (2001-08-24), pages 132, Retrieved from the Internet <URL:https://cssp.chemspider.com/Article.aspx?id=132> [retrieved on 20200217] * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023031958A1 (en) * | 2021-08-31 | 2023-03-09 | Natco Pharma Limited | An improved process for the preparation of trabectedin |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3882236B1 (en) | Production method for 2,5-dicyano-3,6-dihalogenopyrazine | |
| CN103517911B (en) | Regioselective acylation of rapamycin at C-42 | |
| DK162647B (en) | PROCEDURE FOR PREPARING PYRIDO-IMIDAZORIFAMYCINES | |
| LU86461A1 (en) | PROCESS FOR THE DEPROTECTION OF ALLYL ESTERS AND ETHERS | |
| US20250002506A1 (en) | Improved process for the preparation of lurbinectedin and its morphs thereof | |
| HU204064B (en) | New process for producing 6-methyleneandrosta-1,4-diene-3,17-dione derivatives | |
| WO2020105068A1 (en) | Process for the preparation of ecteinascidin derivative and its intermediate | |
| CA2960473A1 (en) | Processes for the preparation of tadalafil and intermediates thereof | |
| EP3911660A1 (en) | Process for preparation of 2-amino-5-hydroxy propiophenone | |
| CN110551123A (en) | Preparation method of 5- (tert-butyloxycarbonyl) -2-methyl-4, 5,6, 7-tetrahydro-2H-pyrazolo [4,3-C ] pyridine-7-carboxylic acid | |
| KR20090063256A (en) | Fluorine-containing catalantine derivatives, preparation method thereof and use as vinca dimeric alkaloid precursor | |
| WO2012111025A4 (en) | 1,2,3-triazole containing artemisinin compounds and process for preparation thereof | |
| WO2023031958A1 (en) | An improved process for the preparation of trabectedin | |
| CN108129357B (en) | Preparation method of anamorelin intermediate | |
| KR101590106B1 (en) | A method for preparing 1-Oxacephalosporin derivatives | |
| CN113816955B (en) | RET kinase inhibitor intermediate and preparation method thereof | |
| EP2331549A1 (en) | Method for preparing 1,6:2,3-dianhydro-beta-d-mannopyranose | |
| KR100299044B1 (en) | Methods for the preparation of 7-aminodesacetoxycephalosporanic acid derivatives | |
| CN110922355A (en) | Preparation method of nicorandil | |
| KR100543172B1 (en) | Process for preparing terane compound | |
| CN117659032A (en) | Intermediates of ascidin derivatives and their synthesis methods | |
| Sheehan et al. | Synthesis and reactions of 7-hydrazonocephalosporanates | |
| KR100998536B1 (en) | Method for preparing oxazolidin-2-one compound using isocyanide dichloride | |
| WO2025059336A1 (en) | An improved process for preparation of lurbinectedin | |
| CN104876872A (en) | Method for preparing 1-tert-butoxy carbonyl-3-hydroxymethyl indazole and application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19888099 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 19888099 Country of ref document: EP Kind code of ref document: A1 |