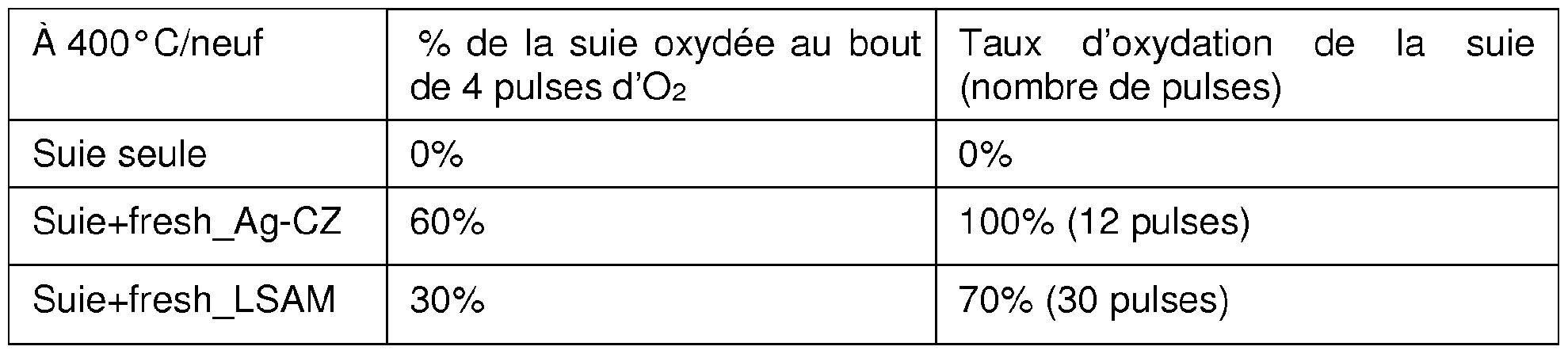WO2020094935A1 - Vehicle equipped with a spark ignition engine and a self-regenerating particulate filter, and associated control method - Google Patents
Vehicle equipped with a spark ignition engine and a self-regenerating particulate filter, and associated control method Download PDFInfo
- Publication number
- WO2020094935A1 WO2020094935A1 PCT/FR2019/052445 FR2019052445W WO2020094935A1 WO 2020094935 A1 WO2020094935 A1 WO 2020094935A1 FR 2019052445 W FR2019052445 W FR 2019052445W WO 2020094935 A1 WO2020094935 A1 WO 2020094935A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- filter
- engine
- vehicle
- vehicle according
- phase
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F01—MACHINES OR ENGINES IN GENERAL; ENGINE PLANTS IN GENERAL; STEAM ENGINES
- F01N—GAS-FLOW SILENCERS OR EXHAUST APPARATUS FOR MACHINES OR ENGINES IN GENERAL; GAS-FLOW SILENCERS OR EXHAUST APPARATUS FOR INTERNAL-COMBUSTION ENGINES
- F01N3/00—Exhaust or silencing apparatus having means for purifying, rendering innocuous, or otherwise treating exhaust
- F01N3/02—Exhaust or silencing apparatus having means for purifying, rendering innocuous, or otherwise treating exhaust for cooling, or for removing solid constituents of, exhaust
- F01N3/021—Exhaust or silencing apparatus having means for purifying, rendering innocuous, or otherwise treating exhaust for cooling, or for removing solid constituents of, exhaust by means of filters
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F01—MACHINES OR ENGINES IN GENERAL; ENGINE PLANTS IN GENERAL; STEAM ENGINES
- F01N—GAS-FLOW SILENCERS OR EXHAUST APPARATUS FOR MACHINES OR ENGINES IN GENERAL; GAS-FLOW SILENCERS OR EXHAUST APPARATUS FOR INTERNAL-COMBUSTION ENGINES
- F01N13/00—Exhaust or silencing apparatus characterised by constructional features
- F01N13/009—Exhaust or silencing apparatus characterised by constructional features having two or more separate purifying devices arranged in series
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F01—MACHINES OR ENGINES IN GENERAL; ENGINE PLANTS IN GENERAL; STEAM ENGINES
- F01N—GAS-FLOW SILENCERS OR EXHAUST APPARATUS FOR MACHINES OR ENGINES IN GENERAL; GAS-FLOW SILENCERS OR EXHAUST APPARATUS FOR INTERNAL-COMBUSTION ENGINES
- F01N3/00—Exhaust or silencing apparatus having means for purifying, rendering innocuous, or otherwise treating exhaust
- F01N3/02—Exhaust or silencing apparatus having means for purifying, rendering innocuous, or otherwise treating exhaust for cooling, or for removing solid constituents of, exhaust
- F01N3/021—Exhaust or silencing apparatus having means for purifying, rendering innocuous, or otherwise treating exhaust for cooling, or for removing solid constituents of, exhaust by means of filters
- F01N3/023—Exhaust or silencing apparatus having means for purifying, rendering innocuous, or otherwise treating exhaust for cooling, or for removing solid constituents of, exhaust by means of filters using means for regenerating the filters, e.g. by burning trapped particles
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F01—MACHINES OR ENGINES IN GENERAL; ENGINE PLANTS IN GENERAL; STEAM ENGINES
- F01N—GAS-FLOW SILENCERS OR EXHAUST APPARATUS FOR MACHINES OR ENGINES IN GENERAL; GAS-FLOW SILENCERS OR EXHAUST APPARATUS FOR INTERNAL-COMBUSTION ENGINES
- F01N3/00—Exhaust or silencing apparatus having means for purifying, rendering innocuous, or otherwise treating exhaust
- F01N3/02—Exhaust or silencing apparatus having means for purifying, rendering innocuous, or otherwise treating exhaust for cooling, or for removing solid constituents of, exhaust
- F01N3/021—Exhaust or silencing apparatus having means for purifying, rendering innocuous, or otherwise treating exhaust for cooling, or for removing solid constituents of, exhaust by means of filters
- F01N3/033—Exhaust or silencing apparatus having means for purifying, rendering innocuous, or otherwise treating exhaust for cooling, or for removing solid constituents of, exhaust by means of filters in combination with other devices
- F01N3/035—Exhaust or silencing apparatus having means for purifying, rendering innocuous, or otherwise treating exhaust for cooling, or for removing solid constituents of, exhaust by means of filters in combination with other devices with catalytic reactors
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F01—MACHINES OR ENGINES IN GENERAL; ENGINE PLANTS IN GENERAL; STEAM ENGINES
- F01N—GAS-FLOW SILENCERS OR EXHAUST APPARATUS FOR MACHINES OR ENGINES IN GENERAL; GAS-FLOW SILENCERS OR EXHAUST APPARATUS FOR INTERNAL-COMBUSTION ENGINES
- F01N2510/00—Surface coverings
- F01N2510/06—Surface coverings for exhaust purification, e.g. catalytic reaction
Definitions
- the invention relates to the field of depollution of internal combustion engines, more particularly to a vehicle provided with a spark-ignition engine connected to a particulate filter and to a method for controlling the latter.
- the published patent document CN103495418 discloses a diesel engine connected to an exhaust line equipped with a particulate filter comprising a catalytic phase allowing self-regeneration of soot at low temperature.
- the diesel engine has the defect of having a high level of pollution due to the NOx-soot compromise.
- the object of the invention is to overcome at least one of the drawbacks of the above-mentioned state of the art. More particularly, the invention aims to significantly reduce the soot emissions of a vehicle fitted with an internal combustion engine.
- the invention relates to a vehicle comprising an internal combustion engine and an exhaust line connected to the internal combustion engine, the line being provided with a particle filter; the filter comprising a filtering medium and a catalytic phase, the catalytic phase having a remarkable oxygen storage capacity in that the engine is of the spark-ignition type operating at least under stoichiometric conditions and the engine is configured to cut the injecting fuel or operating in a lean mixture in order to charge the oxygen filter, the catalytic phase comprising a pure or doped cerine oxide with at least one element from the following list: La, Zr, Y, Gd, Pr, Nd and Sm , and / or another element from the following list: Ag and Pd.
- the catalytic phase is at least based on silver doped zirconium cerine oxide.
- the silver-doped zirconium cerine oxide is of the Ceo, 49 Zro.si O2 type.
- the catalytic phase comprises perovskite of the Lai-x- y Ag x Sr y B O3 type, B being one of the elements on the list: Fe, Mn, Cu Ti, Co and Ni, the element Sr can be replaced by one of the elements in the list: Ca, Ba and K, x is between 0 and 0.5, and y is between 0 and 0.5.
- the perovskite is of the Lao.s Ago, 25 Sro, 25 Mn O3 type.
- the catalytic phase is impregnated directly on the surface of walls of incoming channels of the filter, deposited on a filtration membrane of the particle filter and / or constituting the membrane.
- the invention also relates to a method of controlling the vehicle according to the invention, and the method triggers a phase of suspension of the injection or of lean operation if each of the following conditions is met: there is at least one release of the foot driver, zero engine torque demand and / or vehicle drive by an engine / generator; the engine speed is greater than a minimum threshold.
- the method comprises an additional condition: the temperature of the filter is lower than a single predefined,
- the predefined threshold being between 300 and 500 ° C.
- the phase of suspension or reduction of the fuel injection ends when at least one of the following conditions is met: the release of the foot is no longer established, the torque demand n is no longer zero and / or the duration of the release of the foot, the demand for zero torque or the drive by a motor / generator exceeds a predetermined threshold.
- the filtering medium is based on cordierite, aluminum titanate or silicon carbide.
- the internal combustion engine is combined with an engine / generator forming a hybrid powertrain.
- the measures of the invention are advantageous in that the self-regeneration process of the particulate filter starts at low temperature, around 300 C instead of 600 ° C for known solutions.
- the materials used in the catalytic phase of the particulate filter to ensure the oxidation of the soot are much more economical than those of previous solutions, such as the metals of the platinum group, namely platinum, palladium or rhodium. .
- FIG. 1 illustrates an engine connected to an exhaust line.
- an engine 2 of the spark-ignition type operating at least under stoichiometric conditions is connected to an exhaust line 4.
- the engine 2 and the line 4 are integrated into a vehicle which is not shown.
- the exhaust gases from engine 2 are successively purified by passing through a first three-way catalyst mounted upstream of a particulate filter 6. Downstream of the exhaust line 4 is a second catalyst, for example.
- the particle filter 6 comprises a filter medium and a catalytic phase.
- the filter medium can be based on cordierite, aluminum titanate or silicon carbide.
- the catalytic phase can be impregnated directly on the surface of walls of incoming channels of the particle filter 6, deposited on a filtration membrane of the particle filter 6 and / or constituting the membrane.
- Sr can be replaced by Ca, Ba, K (alkali and alkaline earth), B being included in the list: Fe, Mn, Cu, Ti, Co and Ni. More particularly, tests have been carried out on two kinds of oxides.
- the first type is silver-doped zirconium cerine oxide (2% by mass), named Ag-CZ (Ceo.49Zro.51O2).
- Ag-CZ Ceo.49Zro.51O2
- the silver is deposited on a commercial cerine-zirconia oxide by wet impregnation with Ag nitrate.
- the catalyst is then dried overnight at study at 100 ° C. and then calcined at 700 ° C. in air for 2 hours.
- Sr can be replaced by Ca, Ba, K (alkali and alkaline earth)
- B is an element from the list Fe, Mn, Cu, Ti, Co, Ni.
- the perovskite of composition Lao, 5Ago, 25Sro, 25Mn03 (LSAM) can be synthesized in another by the so-called complexation method.
- the protocol consists in dissolving the metallic nitrates of lanthanum, silver, manganese and strontium in the desired proportions in the minimum of deionized water.
- a chelating agent, maleic acid 10%, of formula C 4 H 4 0 4 is then added to this solution.
- the pH is maintained at 9 by the addition of ammonia.
- the solution is then evaporated on a hot plate until a gel is obtained and then steamed at 130 ° C for ten hours.
- the powder thus obtained is calcined at 500 ° C for 2 hours, in order to remove all the carbonaceous residues linked to the synthesis. Calcination at higher temperature (800 ° C) for 4 hours is then necessary to form the perovskite phase and stabilize the microstructure of the powder.
- the two kinds of oxide are evaluated on test means reproducing the filling strategy of the material is the supply of air (containing 21% oxygen) via the foot raisers throughout the running of the vehicle.
- the soot used is Printex® U commercial carbon black.
- Printex® U soot is used as reference soot in numerous studies on pollution control.
- the measurements of catalytic activities of the two catalysts synthesized for the oxidation of soot were carried out under the following conditions: the soot and the catalyst are mixed in a 1/60 ratio (by mass) and co-ground in a mortar for 15 minutes to obtain intimate contact between the soot and the catalyst. A mass of 200 mg of this mixture is taken and placed in a conventional U-shaped quartz reactor.
- the experimental conditions for programmed temperature oxidation are: a flow of 250 ppm of Ü2 and 10% of hBO in the He at 10 l / h and an introduction for example of wells (term “Anglo-Saxon", puffs of air) of 2 seconds every 7 minutes containing 20% of oxygen in the He, wells representative of the foot raisers .
- the reactor is heated from 100 ° C to 680 ° C at ut® speed of 10 ° C / min with an intermediate plate at 400 ° C and 350 ° C according to the requirements of the experiment.
- the concentration of products resulting (CO and CO2) from the combustion of soot is measured using a gas phase micro-chromatograph (SRA 3000, SRA INSTRUMENTS®) and a mass spectrometer (QMS aspec: Quadrupole Mass Spectrometer, Aspec Technology Inc).
- the soot conversion is calculated from the CO and CO2 concentrations and is expressed as a function of the temperature.
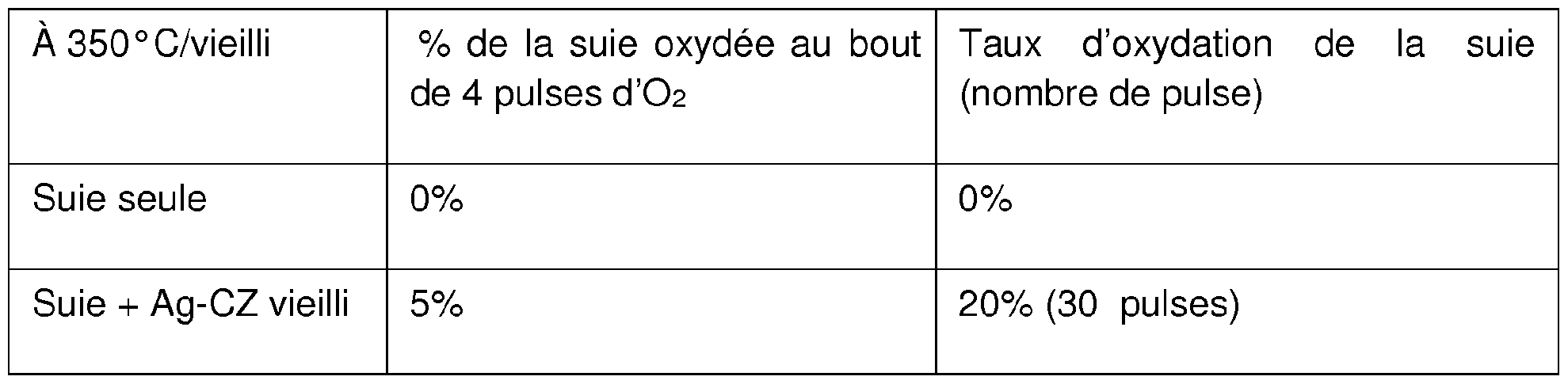
- Oxidation performance at 400 ° C is provided for the material: - in new condition:
- fresh_Ag-CZ fresh term “Anglo-Saxon”; in the context of the tests, it means new
- the results on the catalytic performance of soot oxidation show an oxidation of the soot which still exists at 350 ° C.
- the injection of oxygen via the introduction of air at 400 ° C or even 350 ° C makes it possible to charge the catalyst with oxide ions resulting from the dissociation of oxygen and at the same time to initiate the phase d oxidation of soot from highly reactive oxide ions.
- the engine 2 is configured to produce air wells by cutting the fuel injection on at least one of the engine cylinders 2.
- the engine control can be configured to recharge the particle filter 6 by triggering a injection suspension phase or lean operation if each of the following conditions is met: there is at least one release of the driver's foot, a zero torque request from engine 2 and / or a vehicle drive by an engine / generator ; the speed of rotation of the motor 2 is greater than a minimum threshold; the temperature of the filter 6 is less than a single predefined, the predefined threshold being between 300 and 500 ° C, for example 400 ⁇ , 375 ° C, 350 ° C, or 300 ° C.
- the fuel injection suspension or reduction phase ends when at least one of the following conditions is met: the release of the foot is no longer established, the torque demand is no longer zero and / or the duration of the letting go of the zero torque demand or the drive by a motor / generator exceeds a predetermined threshold.
Landscapes
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Combustion & Propulsion (AREA)
- Mechanical Engineering (AREA)
- General Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Catalysts (AREA)
Abstract
Description
DESCRIPTION DESCRIPTION
VÉHICULE ÉQUIPÉ D’UN MOTEUR À ALLUMAGE COMMANDÉ ET D’UN FILTRE À PARTICULES AUTORÉGÉNÉRANT ET PROCÉDÉ DE CONTRÔLEVEHICLE EQUIPPED WITH A CONTROLLED IGNITION ENGINE AND A SELF-GENERATING PARTICLE FILTER AND METHOD OF MONITORING
ASSOCIÉ ASSOCIATED
Domaine technique Technical area
L’invention a trait au domaine de la dépollution des moteurs à combustion interne plus particulièrement à un véhicule pourvu d’un moteur à allumage commandé relié à un filtre à particules et un procédé de contrôle de ce dernier. The invention relates to the field of depollution of internal combustion engines, more particularly to a vehicle provided with a spark-ignition engine connected to a particulate filter and to a method for controlling the latter.
Technique antérieure Prior art
Le document de brevet publié CN103495418 divulgue un moteur diesel relié à une ligne d’échappement équipée d’un filtre à particules comprenant une phase catalytique permettant une autorégénération des suies à basse température. Cependant, le moteur diesel présente le défaut d’avoir un niveau de pollution élevé à cause du compromis NOx-suies. The published patent document CN103495418 discloses a diesel engine connected to an exhaust line equipped with a particulate filter comprising a catalytic phase allowing self-regeneration of soot at low temperature. However, the diesel engine has the defect of having a high level of pollution due to the NOx-soot compromise.
Exposé de l'invention Statement of the invention
L’invention a pour objectif de pallier au moins un des inconvénients de l’état de la technique susmentionné. Plus particulièrement, l’invention a pour objectif de réduire significativement les émissions de suies d’un véhicule pourvu d’un moteur à combustion interne. The object of the invention is to overcome at least one of the drawbacks of the above-mentioned state of the art. More particularly, the invention aims to significantly reduce the soot emissions of a vehicle fitted with an internal combustion engine.
L’invention a pour objet un véhicule comprenant un moteur à combustion interne et une ligne d’échappement reliée au moteur à combustion interne, la ligne étant dotée d’un filtre à particules ; le filtre comprenant un média filtrant et une phase catalytique, la phase catalytique ayant une capacité de stockage d’oxygène remarquable en ce que le moteur est du type à allumage commandé fonctionnant au moins dans des conditions stoechiométriques et le moteur est configuré pour couper l’injection de carburant ou fonctionner en mélange pauvre afin de charger le filtre en oxygène, la phase catalytique comprenant un oxyde de cérine pure ou dopée avec au moins un élément de la liste suivante : La, Zr, Y, Gd, Pr, Nd et Sm, et/ou un autre élément de la liste suivante : Ag et Pd. Selon un mode avantageux de l’invention, la phase catalytique est au moins à base d’oxyde de cérine zirconium dopé à l’argent. The invention relates to a vehicle comprising an internal combustion engine and an exhaust line connected to the internal combustion engine, the line being provided with a particle filter; the filter comprising a filtering medium and a catalytic phase, the catalytic phase having a remarkable oxygen storage capacity in that the engine is of the spark-ignition type operating at least under stoichiometric conditions and the engine is configured to cut the injecting fuel or operating in a lean mixture in order to charge the oxygen filter, the catalytic phase comprising a pure or doped cerine oxide with at least one element from the following list: La, Zr, Y, Gd, Pr, Nd and Sm , and / or another element from the following list: Ag and Pd. According to an advantageous embodiment of the invention, the catalytic phase is at least based on silver doped zirconium cerine oxide.
Selon un mode avantageux de l’invention, l’oxyde de cérine zirconium dopé à l’argent est du type Ceo,49 Zro.si O2. According to an advantageous embodiment of the invention, the silver-doped zirconium cerine oxide is of the Ceo, 49 Zro.si O2 type.
Selon un mode avantageux de l’invention, la phase catalytique comprend de la pérovskite du type Lai-x-y Agx Sry B O3, B étant un des éléments de la liste : Fe, Mn, Cu Ti, Co et Ni, l’élément Sr pouvant être remplacé par un des éléments de la liste : Ca, Ba et K, x est compris entre 0 et 0,5, et y étant compris entre 0 et 0,5. According to an advantageous embodiment of the invention, the catalytic phase comprises perovskite of the Lai-x- y Ag x Sr y B O3 type, B being one of the elements on the list: Fe, Mn, Cu Ti, Co and Ni, the element Sr can be replaced by one of the elements in the list: Ca, Ba and K, x is between 0 and 0.5, and y is between 0 and 0.5.
Selon un mode avantageux de l’invention, la pérovskite est du type Lao.s Ago,25 Sro,25 Mn O3. According to an advantageous embodiment of the invention, the perovskite is of the Lao.s Ago, 25 Sro, 25 Mn O3 type.
Selon un mode avantageux de l’invention, la phase catalytique est imprégnée directement à la surface de parois de canaux entrants du filtre, déposée sur une membrane de filtration du filtre à particules et/ou constitutive de la membrane. According to an advantageous embodiment of the invention, the catalytic phase is impregnated directly on the surface of walls of incoming channels of the filter, deposited on a filtration membrane of the particle filter and / or constituting the membrane.
L’invention concerne aussi un procédé de contrôle du véhicule selon l’invention, et le procédé déclenche une phase de suspension de l'injection ou de fonctionnement en pauvre si chacune des conditions suivantes est respectée : il y a au moins un lâcher de pied conducteur, une demande en couple du moteur nulle et/ou un entraînement du véhicule par un moteur/générateur; la vitesse de rotation du moteur est supérieure à un seuil minimal. The invention also relates to a method of controlling the vehicle according to the invention, and the method triggers a phase of suspension of the injection or of lean operation if each of the following conditions is met: there is at least one release of the foot driver, zero engine torque demand and / or vehicle drive by an engine / generator; the engine speed is greater than a minimum threshold.
Selon un mode avantageux de l’invention, le procédé comprend une condition supplémentaire : la température du filtre est inférieure à un seul prédéfini, According to an advantageous embodiment of the invention, the method comprises an additional condition: the temperature of the filter is lower than a single predefined,
notamment le seuil prédéfini étant compris entre 300 et 500°C. in particular the predefined threshold being between 300 and 500 ° C.
Selon un mode avantageux de l’invention, la phase de suspension ou de réduction de l’injection de carburant se termine lorsqu’au moins une des conditions suivantes est respectée : le lâcher de pied n’est plus établi, la demande en couple n’est plus nulle et/ou la durée du lâcher de pied, de la demande en couple nulle ou de l’entraînement par un moteur/générateur dépasse un seuil prédéterminé. According to an advantageous embodiment of the invention, the phase of suspension or reduction of the fuel injection ends when at least one of the following conditions is met: the release of the foot is no longer established, the torque demand n is no longer zero and / or the duration of the release of the foot, the demand for zero torque or the drive by a motor / generator exceeds a predetermined threshold.
Préférentiellement, le média filtrant est à base de cordiérite, de titanate d’aluminium ou de carbure de silicium. Préférentiellement, le moteur à combustion interne est combiné à un moteur/générateur formant un groupe motopropulseur hybride. Preferably, the filtering medium is based on cordierite, aluminum titanate or silicon carbide. Preferably, the internal combustion engine is combined with an engine / generator forming a hybrid powertrain.
Les mesures de l’invention sont intéressantes en ce que le processus d’autorégénération du filtre à particules s’amorce à basse température, environ 300 C au lieu de 600°C pour les solutions connues. En oute, les matériaux utilisés dans la phase du catalytique du filtre à particules pour assurer l’oxydation de la suie sont bien plus économiques que ceux des solutions antérieures, comme les métaux du groupe platine, à savoir le platine, le palladium ou le rhodium. The measures of the invention are advantageous in that the self-regeneration process of the particulate filter starts at low temperature, around 300 C instead of 600 ° C for known solutions. In addition, the materials used in the catalytic phase of the particulate filter to ensure the oxidation of the soot are much more economical than those of previous solutions, such as the metals of the platinum group, namely platinum, palladium or rhodium. .
Brève description des dessins Brief description of the drawings
D’autres caractéristiques et avantages de la présente invention seront mieux compris à l’aide de la description et du dessin : Other characteristics and advantages of the present invention will be better understood with the aid of the description and the drawing:
[Fig. 1 ] illustre un moteur relié à une ligne d’échappement. [Fig. 1] illustrates an engine connected to an exhaust line.
Description détaillée detailed description
Dans la figure 1 , un moteur 2 du type à allumage commandé fonctionnant au moins dans des conditions stoechiométriques est relié à une ligne d’échappement 4. Le moteur 2 et la ligne 4 sont intégrés à un véhicule qui n’est pas représenté. Les gaz d’échappement du moteur 2 sont purifiés successivement en passant par un premier catalyseur trois voies monté en amont d'un filtre à particules 6. En aval de la ligne d’échappement 4 se trouve un deuxième catalyseur, par exemple. In FIG. 1, an engine 2 of the spark-ignition type operating at least under stoichiometric conditions is connected to an exhaust line 4. The engine 2 and the line 4 are integrated into a vehicle which is not shown. The exhaust gases from engine 2 are successively purified by passing through a first three-way catalyst mounted upstream of a particulate filter 6. Downstream of the exhaust line 4 is a second catalyst, for example.
Le filtre à particules 6 comprend un média filtrant et une phase catalytique. Le média filtrant peut être à base de cordiérite, de titanate d'aluminium ou de carbure de silicium. La phase catalytique peut être imprégnée directement à la surface de parois de canaux entrants du filtre à particules 6, déposée sur une membrane de filtration du filtre à particules 6 et/ou constitutive de la membrane. The particle filter 6 comprises a filter medium and a catalytic phase. The filter medium can be based on cordierite, aluminum titanate or silicon carbide. The catalytic phase can be impregnated directly on the surface of walls of incoming channels of the particle filter 6, deposited on a filtration membrane of the particle filter 6 and / or constituting the membrane.
Les propriétés de stockage d’oxygène et d’oxydation catalytique sont bien connues de l’homme du métier pour l’oxyde de cérine pure, l’oxyde de cérine dopée La, Zr, Y, Gd, Pr, Nd, Sm, en présence ou non de métaux précieux de type Ag, Pd, et leur combinaison ainsi que les pérovskites du type La-i-*-y Agx Sry B O3 avec x = 0 - 0,5 et y = 0 - 0,5. Le Sr peut être remplacé par Ca, Ba, K (alcalins et alcalinoterreux), B étant compris dans la liste : Fe, Mn, Cu, Ti, Co et Ni. Plus particulièrement, des essais ont été menés sur deux sortes d’oxydes. The oxygen storage and catalytic oxidation properties are well known to those skilled in the art for pure cerine oxide, doped cerine oxide La, Zr, Y, Gd, Pr, Nd, Sm, en presence or absence of precious metals of type Ag, Pd, and their combination as well as perovskites of type La-i - * - y Agx Sr y B O3 with x = 0 - 0.5 and y = 0 - 0.5. Sr can be replaced by Ca, Ba, K (alkali and alkaline earth), B being included in the list: Fe, Mn, Cu, Ti, Co and Ni. More particularly, tests have been carried out on two kinds of oxides.
La première sorte est l’oxyde de cérine zirconium dopé à l’argent (2% en masse), nommé Ag-CZ (Ceo.49Zro.51O2). L’argent est déposé sur un oxyde commercial de cérine-zircone par imprégnation en voie humide à partir de nitrate d’Ag. Le catalyseur est ensuite séché une nuit à l’étude à 100°C puis calciné à 700°C sous air pendant 2 heures. The first type is silver-doped zirconium cerine oxide (2% by mass), named Ag-CZ (Ceo.49Zro.51O2). The silver is deposited on a commercial cerine-zirconia oxide by wet impregnation with Ag nitrate. The catalyst is then dried overnight at study at 100 ° C. and then calcined at 700 ° C. in air for 2 hours.
La deuxième sorte d’oxyde est une pérovskite du type Lai-x-y Agx Sry B O3 avec x = 0 - 0,5 et y = 0 - 0,5. Le Sr peut être remplacé par Ca, Ba, K (alcalins et alcalinoterreux), B est un élément de la liste Fe, Mn, Cu, Ti, Co, Ni. La pérovskite de composition Lao,5Ago,25Sro,25Mn03 (LSAM) peut être synthétisée en autre par la méthode dite de complexation. Le protocole consiste à dissoudre les nitrates métalliques de lanthane, argent, manganèse et strontium dans les proportions souhaitées dans le minimum d’eau déionisée. Un agent chélatant, l’acide maléique 10%, de formule C4H404 est ensuite ajouté à cette solution. Pour favoriser la complexation des précurseurs métalliques avec l’acide maléique, le pH est maintenu à 9 par l'ajout d’ammoniac. La solution est ensuite évaporée sur une plaque chauffante jusqu’à l’obtention d’un gel puis étuvé à 130°C une dizaine d’heures. La poudre ainsi obtenue est calcinée à 500 °C pendant 2 heures, afin d’éliminer tous les résidus carbonés liés à la synthèse. Une calcination à plus haute température (800° C) pendant 4 heures est ensuite nécessaire pour former la phase pérovskite et stabiliser la microstructure de la poudre. The second kind of oxide is a perovskite of the Lai-x- y Ag x Sr y B O3 type with x = 0 - 0.5 and y = 0 - 0.5. Sr can be replaced by Ca, Ba, K (alkali and alkaline earth), B is an element from the list Fe, Mn, Cu, Ti, Co, Ni. The perovskite of composition Lao, 5Ago, 25Sro, 25Mn03 (LSAM) can be synthesized in another by the so-called complexation method. The protocol consists in dissolving the metallic nitrates of lanthanum, silver, manganese and strontium in the desired proportions in the minimum of deionized water. A chelating agent, maleic acid 10%, of formula C 4 H 4 0 4 is then added to this solution. To promote the complexation of metal precursors with maleic acid, the pH is maintained at 9 by the addition of ammonia. The solution is then evaporated on a hot plate until a gel is obtained and then steamed at 130 ° C for ten hours. The powder thus obtained is calcined at 500 ° C for 2 hours, in order to remove all the carbonaceous residues linked to the synthesis. Calcination at higher temperature (800 ° C) for 4 hours is then necessary to form the perovskite phase and stabilize the microstructure of the powder.
Les deux sortes d’oxyde sont évaluées sur des moyens d’essais reproduisant la stratégie de remplissage du matériau est l’apport d’air (contenant 21 % d’oxygène) via les levers de pied tout au long du roulage du véhicule. The two kinds of oxide are evaluated on test means reproducing the filling strategy of the material is the supply of air (containing 21% oxygen) via the foot raisers throughout the running of the vehicle.
Pour reproduire la situation, deux mélanges mécaniques entre, d’une part, la poudre d’Ag-CZ et la suie, et d’autre part, la poudre LSAM et la suie, ont été réalisés. La suie utilisée est du noir de carbone commercial Printex® U. La suie Printex® U est utilisée comme suie de référence dans de nombreuses études portant sur la dépollution. Les mesures d’activités catalytiques des deux catalyseurs synthétisés pour l’oxydation des suies ont été réalisées dans les conditions suivantes : la suie et le catalyseur sont mélangés dans un rapport 1/60 (en masse) et co-broyés dans un mortier pendant 15 minutes afin d’obtenir un contact intime entre la suie et le catalyseur. Une masse de 200 mg, de ce mélange, est prélevée et placée dans un réacteur en quartz classique en U. Les conditions expérimentales des oxydations en température programmée sont : un flux de 250 ppm d’Ü2 et 10% d’hbO dans l’He à 10 l/h et une introduction par exemple de puises (terme « anglo-saxon », bouffées d’air) de 2 secondes toutes les 7 minutes contenant 20% d’oxygène dans l’He, puises représentatifs des levers de pied. Le réacteur est chauffé de 100°C à 680 °C à ut® vitesse de 10°C/ min avec un plateau intermédiaire à 400°C et 350°C selon les bsoins de l’expérience. La concentration des produits issus (CO et CO2) de la combustion des suies est mesurée à l’aide d’un micro-chromatographe en phase gaz (SRA 3000, SRA INSTRUMENTS®) et d’un spectromètre de masse (QMS aspec : Quadrupole Mass Spectrometer, Aspec Technology Inc). La conversion de la suie est calculée à partir des concentrations en CO et CO2 et est exprimée en fonction de la température. To reproduce the situation, two mechanical mixtures between, on the one hand, the Ag-CZ powder and the soot, and on the other hand, the LSAM powder and the soot, were produced. The soot used is Printex® U commercial carbon black. Printex® U soot is used as reference soot in numerous studies on pollution control. The measurements of catalytic activities of the two catalysts synthesized for the oxidation of soot were carried out under the following conditions: the soot and the catalyst are mixed in a 1/60 ratio (by mass) and co-ground in a mortar for 15 minutes to obtain intimate contact between the soot and the catalyst. A mass of 200 mg of this mixture is taken and placed in a conventional U-shaped quartz reactor. The experimental conditions for programmed temperature oxidation are: a flow of 250 ppm of Ü2 and 10% of hBO in the He at 10 l / h and an introduction for example of wells (term "Anglo-Saxon", puffs of air) of 2 seconds every 7 minutes containing 20% of oxygen in the He, wells representative of the foot raisers . The reactor is heated from 100 ° C to 680 ° C at ut® speed of 10 ° C / min with an intermediate plate at 400 ° C and 350 ° C according to the requirements of the experiment. The concentration of products resulting (CO and CO2) from the combustion of soot is measured using a gas phase micro-chromatograph (SRA 3000, SRA INSTRUMENTS®) and a mass spectrometer (QMS aspec: Quadrupole Mass Spectrometer, Aspec Technology Inc). The soot conversion is calculated from the CO and CO2 concentrations and is expressed as a function of the temperature.
Les performances d’oxydation à 400°C sont fourniespour le matériau : - à l’état neuf : Oxidation performance at 400 ° C is provided for the material: - in new condition:
• calciné à 700°C pendant 2 heures, nommé « fresh_Ag-CZ» (« fresh » terme « anglo-saxon » ; dans le contexte des essais, il signifie neuf) • calcined at 700 ° C for 2 hours, called "fresh_Ag-CZ" ("fresh" term "Anglo-Saxon"; in the context of the tests, it means new)
• calciné à 800 °C pendant 4 heures, nommé « fresh_LS¾M » • calcined at 800 ° C for 4 hours, called "fresh_LS¾M"
- à l’état vieilli : - in the aged state:
· 4 heures à 980 °C sous ISh et 10% d’eau, nommé « Ag-CZ vieilli » et · 4 hours at 980 ° C under ISh and 10% water, called "Ag-CZ aged" and
« LSAM vielli» "Old LSAM"
[Tableau 1] [Table 1]
[Tableau 2] [Table 2]
[Tableau 3] [Table 3]
[Tableau 4] [Table 4]
Les résultats sur les performances catalytiques d’oxydation de la suie montrent une oxydation de la suie qui existe toujours à 350 °C. L’injection d’oxygène via l’introduction d’air à 400°C voire même 350°C, permet de charger le catalyseur en ions oxydes issus de la dissociation de l’oxygène et dans le même temps d’initier la phase d’oxydation de la suie à partir des ions oxydes très réactifs. En phase de roulage, le moteur 2 est configuré pour produire des puises d’air en coupant l’injection de carburant sur au moins un des cylindres du moteur 2. Le contrôle moteur peut être configuré pour recharger le filtre à particules 6 en déclenchant une phase de suspension de l’injection ou de fonctionnement en pauvre si chacune des conditions suivantes est respectée : il y a au moins un lâcher de pied conducteur, une demande en couple du moteur 2 nulle et/ou un entraînement du véhicule par un moteur/générateur ; la vitesse de rotation du moteur 2 est supérieure à un seuil minimal ; la température du filtre 6 est inférieure à un seul prédéfini, le seuil prédéfini étant compris entre 300 et 500°C, par exemple 400Ό, 375° C, 350°C, ou 300°C. The results on the catalytic performance of soot oxidation show an oxidation of the soot which still exists at 350 ° C. The injection of oxygen via the introduction of air at 400 ° C or even 350 ° C, makes it possible to charge the catalyst with oxide ions resulting from the dissociation of oxygen and at the same time to initiate the phase d oxidation of soot from highly reactive oxide ions. In the driving phase, the engine 2 is configured to produce air wells by cutting the fuel injection on at least one of the engine cylinders 2. The engine control can be configured to recharge the particle filter 6 by triggering a injection suspension phase or lean operation if each of the following conditions is met: there is at least one release of the driver's foot, a zero torque request from engine 2 and / or a vehicle drive by an engine / generator ; the speed of rotation of the motor 2 is greater than a minimum threshold; the temperature of the filter 6 is less than a single predefined, the predefined threshold being between 300 and 500 ° C, for example 400Ό, 375 ° C, 350 ° C, or 300 ° C.
La phase de suspension ou réduction de l’injection de carburant se termine lorsqu’au moins une des conditions suivantes est respectée : le lâcher de pied n’est plus établi, la demande en couple n’est plus nulle et/ou la durée du lâcher de pied de la demande en couple nulle ou de l’entraînement par un moteur/générateur dépasse un seuil prédéterminé. The fuel injection suspension or reduction phase ends when at least one of the following conditions is met: the release of the foot is no longer established, the torque demand is no longer zero and / or the duration of the letting go of the zero torque demand or the drive by a motor / generator exceeds a predetermined threshold.
Claims
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR1871414 | 2018-11-08 | ||
| FR1871414A FR3088369A1 (en) | 2018-11-08 | 2018-11-08 | VEHICLE HAVING A CONTROLLED IGNITION ENGINE AND A SELF-REGENERATING PARTICLE FILTER AND ASSOCIATED CONTROL METHOD |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020094935A1 true WO2020094935A1 (en) | 2020-05-14 |
Family
ID=66041562
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/FR2019/052445 Ceased WO2020094935A1 (en) | 2018-11-08 | 2019-10-16 | Vehicle equipped with a spark ignition engine and a self-regenerating particulate filter, and associated control method |
Country Status (2)
| Country | Link |
|---|---|
| FR (1) | FR3088369A1 (en) |
| WO (1) | WO2020094935A1 (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20110030346A1 (en) * | 2009-08-05 | 2011-02-10 | Basf Se | Treatment System for Gasoline Engine Exhaust Gas |
| CN103495418A (en) | 2013-09-26 | 2014-01-08 | 中国石油大学(北京) | Macroporous-mesoporous cerium-zirconium solid solution silver-loaded catalyst and preparation method and application thereof |
| US20180202336A1 (en) * | 2017-01-13 | 2018-07-19 | Toyota Jidosha Kabushiki Kaisha | Exhaust gas control apparatus for internal combustion engine and exhaust gas control method for internal combustion engine |
-
2018
- 2018-11-08 FR FR1871414A patent/FR3088369A1/en active Pending
-
2019
- 2019-10-16 WO PCT/FR2019/052445 patent/WO2020094935A1/en not_active Ceased
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20110030346A1 (en) * | 2009-08-05 | 2011-02-10 | Basf Se | Treatment System for Gasoline Engine Exhaust Gas |
| CN103495418A (en) | 2013-09-26 | 2014-01-08 | 中国石油大学(北京) | Macroporous-mesoporous cerium-zirconium solid solution silver-loaded catalyst and preparation method and application thereof |
| US20180202336A1 (en) * | 2017-01-13 | 2018-07-19 | Toyota Jidosha Kabushiki Kaisha | Exhaust gas control apparatus for internal combustion engine and exhaust gas control method for internal combustion engine |
Also Published As
| Publication number | Publication date |
|---|---|
| FR3088369A1 (en) | 2020-05-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12345193B2 (en) | Exhaust gas purification system for a gasoline engine | |
| US11614015B2 (en) | Exhaust gas purification system for a gasoline engine | |
| WO2000035564A1 (en) | Exhaust emission control system for internal combustion engines, exhaust emission control method and exhaust emission control catalyst | |
| US11649753B2 (en) | Exhaust gas purification system for a gasoline engine | |
| EP3639922B1 (en) | Exhaust gas purification system for a gasoline engine | |
| US11547969B2 (en) | Exhaust gas purification system for a gasoline engine | |
| WO2009053589A2 (en) | Purification structure incorporating a biased electrochemical catalyst system | |
| EP2134452B1 (en) | Method of operating a diesel engine with a view to making it easier to regenerate a particle filter in the exhaust system | |
| EP1034026B1 (en) | PROCESS FOR TREATING EXHAUST GASES WITH A COMPOSITION BASED ON MANGANESE FOR TRAPPING NOx | |
| WO1999067509A1 (en) | Method for treating by combustion carbon-containing particles in an internal combustion engine exhaust circuit | |
| WO2010076509A1 (en) | Purification structure including a catalysis system supported by a zircon in reduced state | |
| WO2011157963A2 (en) | Electrochemical catalysis system | |
| WO2020094935A1 (en) | Vehicle equipped with a spark ignition engine and a self-regenerating particulate filter, and associated control method | |
| EP1681096B1 (en) | Catalyst for exhaust gas cleaning | |
| FR2961407A1 (en) | Catalyzed particle filter for e.g. stratified direct injection type petrol engine of automobile, has multi-metallic perovskite type oxide based catalytic composition comprising lanthanum-manganese-silver oxide having specific index | |
| WO2020084207A1 (en) | Self-regenerating particulate filter producing reducing agent for reduction of nitrogen oxide | |
| EP2439385B1 (en) | Device for post-treatment of the exhaust gases from an internal combustion engine | |
| JP4196573B2 (en) | Exhaust gas purification method and purification device for internal combustion engine | |
| EP2990097A1 (en) | Catalytic particle filter | |
| FR3088368A1 (en) | VEHICLE EQUIPPED WITH A SELF-REGENERATING PARTICLE FILTER AND A MOTOR AND METHOD FOR CONTROLLING THE SAME | |
| FR2956329A1 (en) | PARTICULATE FILTER COMPRISING A CATALYTIC PHASE | |
| EP1474220A2 (en) | Device for use in treatment of exhaust gases particularly from engines operating with a weak mixture | |
| EP2976148A1 (en) | Particle filter | |
| FR3103117A1 (en) | Process for treating NOx with ammonia produced in-situ in a motor vehicle particulate filter and device for implementing the process | |
| FR3039079A1 (en) | CATALYSIS PARTICULATE FILTER |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19806023 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 19806023 Country of ref document: EP Kind code of ref document: A1 |