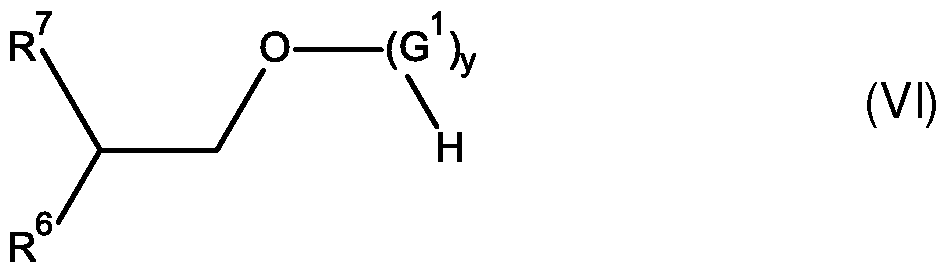WO2020030623A1 - Packaging unit comprising a detergent composition containing an enzyme and at least one chelating agent - Google Patents
Packaging unit comprising a detergent composition containing an enzyme and at least one chelating agent Download PDFInfo
- Publication number
- WO2020030623A1 WO2020030623A1 PCT/EP2019/071083 EP2019071083W WO2020030623A1 WO 2020030623 A1 WO2020030623 A1 WO 2020030623A1 EP 2019071083 W EP2019071083 W EP 2019071083W WO 2020030623 A1 WO2020030623 A1 WO 2020030623A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- enzyme
- compartment
- acid
- packaging unit
- range
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/26—Organic compounds containing nitrogen
- C11D3/33—Amino carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D17/00—Detergent materials or soaps characterised by their shape or physical properties
- C11D17/04—Detergent materials or soaps characterised by their shape or physical properties combined with or containing other objects
- C11D17/041—Compositions releasably affixed on a substrate or incorporated into a dispensing means
- C11D17/042—Water soluble or water disintegrable containers or substrates containing cleaning compositions or additives for cleaning compositions
- C11D17/045—Multi-compartment
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/38—Products with no well-defined composition, e.g. natural products
- C11D3/386—Preparations containing enzymes, e.g. protease or amylase
- C11D3/38618—Protease or amylase in liquid compositions only
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/38—Products with no well-defined composition, e.g. natural products
- C11D3/386—Preparations containing enzymes, e.g. protease or amylase
- C11D3/38627—Preparations containing enzymes, e.g. protease or amylase containing lipase
Definitions
- Packaging unit comprising a detergent composition containing an enzyme and at least one che- lating agent
- the present invention is directed towards a packaging unit containing a detergent single unit dose wherein said packaging unit comprises a compartment 1 comprising an aqueous solution of enzyme (A) and at least one chelating agent (B) dissolved in an aqueous medium, said chelating agent (B) being
- (B1 ) at least one alkali metal salt of glutamic acid diacetic acid (GLDA), and
- (B2) at least one alkali metal salt of methyl glycine diacetic acid (MGDA), and one more compartment comprising at least one ingredient of a detergent other than en- zyme (A) and chelating agent(s) (B), wherein the overall concentration of chelating agent(s) (B) in compartment 1 is in the range of from 35 to 75 % by weight.
- MGDA methyl glycine diacetic acid
- MGDA methyl glycine diacetic acid
- STPP sodium tripoly- phosphate
- MGDA and related compounds are recommended and used for various pur- poses such as laundry detergents and for automatic dishwashing (ADW) formulations, in partic- ular for so-called phosphate-free laundry detergents and phosphate-free ADW formulations.
- unit doses are of increased com-tapal importance. They are of great convenience for the end-user because such unit doses contain the right amounts of the ingredients for the washing and rinsing steps and because they can be easily placed into the automatic dishwasher or washing machine by the end-user, see, e.g., WO 2002/042400 and WO 201 1/072017.
- unit doses are tablets and pellets and in particular pouches. Pouches in the form of multi-compartment pouches have been dis- closed as well, see WO 2009/1 12994.
- Such detergent composi- tions can preferably be used in laundry or automatic dishwashing.
- packaging units as defined at the outset have been found, hereinafter also re- ferred to as inventive units or units according to the (present) invention.
- packaging units comprise more than one compartments. Such compartments are separated from each other, for example by a polymer layer that may be mechanically flexible or stiff.
- a polymer layer that may be mechanically flexible or stiff.
- the distinction between mechanically flexible and mechanically stiff may be made by manual determination of the degree of deformability by an average end user with two fingers. If such an average end user can deform the shape of said unit by at least 5% into one dimension the respective unit is deemed mechanically flexible, otherwise it is deemed stiff.
- Inventive units comprise more than one compartment.
- one of such compartments will be named“compartment 1” to distinguish it from the other com- partment(s).
- Such compartments may have the same or different appearance, and they may have the same or different sizes.
- Such compartments are separated from each other, for exam- pie by a polymer film.
- inventive units are tablets that have at least one cavity per tablet. Per cavity there is at least one pouch, preferably there is at least one pouch placed into the cavity and attached to the tablet.
- the volume of the pouch including the solu- tion of enzyme (A) and complexing agent(s) (B) corresponds to the volume of the cavity, for example they may have the same volume ⁇ 10%, preferably ⁇ 5%. The better the shape and the size of cavity on one side and the shape of the pouch including the solution of enzyme (A) and complexing agent(s) (B) on the other side correspond to each other the less breakage during transport can be observed.
- Such tablets may be packaged in a film of, e.g., polyvinyl alcohol.
- the tablet comprises components of the respective detergent composition such as surfactants, builder(s), enzymes, and/or bleaching agent.
- inventive units are shaped as a box that has at least one cavity per box. Per cavity there is at least one pouch, preferably there is at least one pouch placed into the cavity and attached to the box.
- the volume of the pouch including the solution of enzyme (A) and complexing agent(s) (B) corresponds to the volume of the cavity, for example they may have the same volume ⁇ 10%, preferably ⁇ 5%. The better the shape and the size of cavity on one side and the shape of the box including the solution of enzyme (A) and complexing agent(s) (B) on the other side correspond to each other the less breakage during transport can be observed.
- the box comprises components of the respective detergent compo- sition such as surfactants, builder(s), enzymes, and/or bleaching agent.
- inventive units are pouches that encompass at least two com- partments, for example two, three or four compartments.
- One of the compartments contains the solution of enzyme (A) and complexing agent(s) (B).
- the other components of the respective detergent compositions are in the one or more other compartment(s).
- all complexing agent(s) (B) that are comprised in inventive units is in the very compartment as defined above.
- a share of complexing agent(s) (B) is comprised in one compartment in dis- solved form, as stated above, and more of complexing agent(s) (B) is comprised in the other compartment or one other compartment, as applicable, of the inventive unit.
- Solid detergent compositions may contain residual moisture.
- Residual moisture refers to water other than the water that is part of the aqueous medium in which complexing agent(s) (B) are dissolved in.
- the residual moisture content is in the range of from 0.1 to 10 % by weight, referring to the total respective detergent composition.
- the residual moisture content can be determined, e. g., by Karl-Fischer-Titration or by measuring the weight loss upon drying.
- the term“pouch” refers to a unit made from a flexible film. Said unit is essentially closed upon storage of the respective dishwashing detergent corn- position. During the dishwashing process the contents of the pouch are removed from the pouch itself, preferably by dissolving the pouch.
- Units and especially pouches in the context of the present invention may have various shapes.
- units may be in the form of a ball, an ellipsoid, a cube, a cuboid, or they may be of geometrically irregular shape.
- pouches may have the shape of an enve- lope, of a pillow, of a flexible sleeve or flexible tube that is closed at both ends, of a ball or a cube.
- units according to the present invention and espe- cially pouches have a diameter in the range of from 0.5 to 7 cm.
- units according to the present invention and espe- cially pouches have a volume - in the closed state - in the range of from 15 to 70 ml, preferably 18 ml to 50 ml and in particular 20 to 30 ml.
- inventive units are particularly useful for au- tomatic dishwash in home care application.
- Inventive units particularly useful for fabric care in home care applications may have a volume in the range of from 15 to 40 ml, preferably 25 to 30 ml.
- each compartment has a content in the range of from 0.5 to 50 ml, preferably 5 to 25 ml.
- inventive units encompass two or more compartments, such compartments may have equal size or different size.
- inventive units en- compass one major compartment and one or two or three smaller compartments.
- Inventive units are made from a polymer, preferably from a water-soluble polymer.
- Pouches in the context of the present invention are made from a polymer film.
- Said polymer may be selected from natural polymers, modified natural polymers, and synthetic polymers.
- suitable natural polymers are alginates, especially sodium alginate, fur- thermore xanthum, carragum, dextrin, maltodextrin, gelatine, starch, and pectin.
- suitable modified natural polymers are methylcellulose, ethylcellulose, carboxymethyl cellulose, hydroxypropylcellulose, hydroxypropyl methyl cellulose (HPMC), and hydroxymethyl cellulose.
- suitable synthetic polymers are polyvinyl pyrrolidone, polyacrylamide, polyalkylene glycols, preferably polypropylene glycol and polyethylene glycol, especially polyethylene glycol with a molecular weight M w in the range of at least 2,000 g/mol, preferably of from 3,000 to 100,000 g/mol, and in particular polyvinyl alcohol.
- polyvinyl alcohol does not only include homopolymers of polyvinyl alcohol that can be made by free-radical polymerization of vinyl acetate followed by subsequent hydrolysis (saponification) of all or the vast majority of the ester groups.
- Polyvinyl alcohol also includes copolymers obtainable by free-radical copolymerization of vinyl acetate and at least one comonomer selected from maleic acid, maleic anhydride, itaconic anhydride, methyl (meth)acrylate and 2-acrylamido-2-methyl propanesulfonic acid (“AMPS”).
- AMPS 2-acrylamido-2-methyl propanesulfonic acid
- polyvinyl alcohol as used for making units and especially for pouches may have an average degree of polymerization (weight average) in the range of from 500 to 3,000 g/mol.
- the molecular weight M w of such polyvinyl alcohol is pref- erably, in the range of from 6,000 to 250,000 g/mol, preferably up to 75,000 g/mol.
- the molecu- lar weight is preferably determined by gel permeation chromatography of the respective polyvi- nyl acetate or respective copolymer before saponification.
- polyvinyl alcohol used for making units and especially pouches is atactic as deter- mined by 1 H NMR spectroscopy.
- Polyvinyl alcohols used for making units - especially pouches - essentially have repeating units of (CH2-CHOH).
- the hydroxyl groups in polyvinyl alcohol are mostly in 1 ,3-position, thus form- ing structural units of the type -CH2-CH(OH)-CH2-CH(OH)-.
- One or more modified polyvinyl alcohols may be employed as polymers instead of polyvinyl al- cohol or in combination with polyethylene glycol or with polyvinyl alcohol.
- graft copolymers such as polyalkylene glycol grafted with polyvinyl acetate followed by subsequent hydrolysis/saponification of the ester groups.
- Polymer may be used without or with one or more additives.
- Suitable additives are especially plasticizers such as C4-Cio-dicarboxylic acids, for example adipic acid, and glycols such as eth- ylene glycol and diethylene glycol.
- polyvinyl alcohols Due to their production, commercially available polyvinyl alcohols usually have residual non- saponified ester groups, especially acetate groups. Polyvinyl alcohols used for making units and especially pouches for embodiments of the present invention essentially have a degree of sa- ponification in the range of from 87 to 89 mole-%. The degree of saponification can be deter- mined in accordance with the determination of the ester value, for example according to DIN EN ISO 3681 (2007-10).
- polyvinyl alcohols used for making units and espe- cially for making pouches for embodiments of the present invention have a glass transition tem- perature in the range of from 55 to 60°C, preferably 58°C, determinable according to, e.g., DIN 53765: 1994-03, or ISO 1 1357-2: 1999-03.
- polyvinyl alcohols used for making inventive units and especially for making pouches for embodiments of the present invention have a melting point in the range of from 185 to 187°C.
- polyvinyl alcohols used for making for embodi- ments of the present invention and especially for making pouches comprising a single unit dose are partially acetalized or ketalized with sugars such as, glucose, fructose, or with starch.
- polyvinyl alcohols used for making units and es- pecially pouches are partially esterified with, e. g., maleic acid or itaconic acid.
- polyvinyl alcohol films may contain a plasticizer. Plasticizers may be used for reducing the stiffness of such polyvinyl alcohol films.
- Suitable compounds usable as plasticizers for polyvinyl alcohol are ethylene glycol, diethylene glycol, triethylene glycol, polyethylene glycol, for example with an average molecular weight M w up to 400 g/mol, glycerol, trimethylol propane, triethanolamine, and neo-pentyl glycol. Up to 25 % by weight of the respective polyvinyl alcohol may be plasticizer.
- said pouches are made from a polymer film, said polymer being water-soluble at a temperature of at least 40°C, for example in the range of from 40 to 95°C, but insoluble in water at a temperature in the range of from 5 to 30°C.
- said pouches are made from polymer films that are soluble in water even at 1 °C.
- the terms water-soluble and soluble in water are used in- terchangeably. They both refer to polymers that dissolve in water at 20°C. However, such poly- mers dissolve much slower or not detectably at all in the aqueous medium containing complex- ing agent(s) (B).
- a polymer is deemed water-soluble if the percentage of solubility is at least 90%. A suitable method of determination of the percentage is disclosed below.
- polymer films that are soluble at 1 °C or more and of polymer films that are soluble at 40°C are polyvinyl alcohol films available from Syntana E. Harke GmbH & Co under the trademark of Solublon ®.
- polymer films and preferably polyvinyl alcohol films used for making pouches that can be used in the present invention have a thickness (strength) in the range of from 10 to 100 pm, preferably 20 to 90 pm, even more preferably 25 to 35 pm. If the strength of polymer films and especially of polyvinyl alcohol films exceeds 100 pm it takes too long to dissolve them during the washing cycle. If the strength of polymer films and especial- ly of polyvinyl alcohol films is below 10 pm they are too sensitive to mechanical stress.
- the aqueous solution in compartment 1 also contains enzyme (A), hereinafter also referred to as enzyme (A), for example hydrolase (A).
- enzyme (A) are selected from proteases, amylases, and lipases, hereinafter also referred to as proteases (A), amylases (A), or lipases (A), respectively.
- enzymes (A) in inventive compositions exhibit a particularly high life-time.
- Enzymes (A) that are particularly beneficial in inventive compositions are selected from hydro- lases (EC 3).
- Preferred enzymes (A) are selected from the group of enzymes acting on ester bond (E.C. 3.1 ), glycosylases (E.C. 3.2), and peptidases (E.C. 3.4).
- Enzymes acting on ester bond (E.C. 3.1 ), hereinafter also refer to lipases (component (b)), respectively.
- Glycosylases (E.C. 3.2) hereinafter also refer to either amylases (A) and cellulases (A).
- Peptidases hereinaf- ter also refer to proteases (A).
- Hydrolases (A) in the context of the present invention are identified by polypeptide sequences, also called amino acid sequences herein.
- the polypeptide sequence specifies the three- dimensional structure including the“active site” of an enzyme which in turn determines the cata- lytic activity of the same.
- Polypeptide sequences may be identified by a SEQ ID NO. According to the World Intellectual Property Office (WIPO) Standard ST.25 (1998) the amino acids herein are represented using three-letter code with the first letter as a capital or the corresponding one letter.
- Enzymes (A) in the context of the present invention may relate to parent enzymes and/or variant enzymes, both having enzymatic activity. Enzymes having enzymatic activity are enzymatically active or exert enzymatic conversion, meaning that enzymes act on substrates and convert these into products.
- A“parent” sequence of a parent protein or enzyme also called“parent enzyme” is the starting sequence for introduction of changes, e.g., by introducing one or more amino acid substitutions, insertions, deletions, or a combination thereof to the sequence, resulting in“variants” of the par- ent sequences.
- the term parent enzyme (or parent sequence) includes wild-type enzymes (se- quences) and synthetically generated sequences (enzymes) which are used as starting se- quences for introduction of (further) changes.
- variant enzyme variant or“sequence variant” or“variant enzyme” refers to an enzyme that differs from its parent enzyme in its amino acid sequence to a certain extent. If not indicated otherwise, variant enzyme“having enzymatic activity” means that this variant enzyme has the same type of enzymatic activity as the respective parent enzyme.
- Amino acid substitutions are described by providing the original amino acid of the parent en- zyme followed by the number of the position within the amino acid sequence, followed by the substituted amino acid.
- Amino acid deletions are described by providing the original amino acid of the parent enzyme followed by the number of the position within the amino acid sequence, followed by * .
- Amino acid insertions are described by providing the original amino acid of the parent enzyme followed by the number of the position within the amino acid sequence, followed by the original amino acid and the additional amino acid. For example, an insertion at position 180 of lysine next to glycine is designated as“Gly180Glyl_ys” or“G180GK”.
- alterations can be introduced at a position
- the different alterations are separat- ed by a comma, e.g.“Arg170Tyr, Glu” represents a substitution of arginine at position 170 with tyrosine or glutamic acid.
- different alterations or optional substitutions may be indi- cated in brackets e.g. Arg170[Tyr, Gly] or Arg170 ⁇ Tyr, Gly ⁇ ; or in short R170 [Y,G] or R170 ⁇ Y, G ⁇ ; or in long R170Y, R170G.
- Enzyme variants may be defined by their sequence identity when compared to a parent en- zyme. Sequence identity usually is provided as“% sequence identity” or“% identity”. For calcu- lation of sequence identities, in a first step a sequence alignment has to be produced. According to this invention, a pairwise global alignment has to be produced, meaning that two sequences have to be aligned over their complete length, which is usually produced by using a mathemati- cal approach, called alignment algorithm.
- the alignment is generated by using the algorithm of Needleman and Wunsch (J. Mol. Biol. 1979 48 p. 443-453).
- the program“NEEDLE” The European Molecular Biology Open Software Suite (EMBOSS)
- EMBOSS European Molecular Biology Open Software Suite
- %-identity (identical residues / length of the alignment region which is show- ing the respective sequence of this invention over its complete length)-100.
- enzyme variants may be described as an amino acid sequence which is at least n% identical to the amino acid sequence of the respective parent enzyme with“n” being an integer between 10 and 100.
- variant enzymes are at least 70%, at least 75%, at least 80%, at least 81 %, at least 82%, at least 83%, at least 84%, at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical when compared to the full length amino acid sequence of the parent enzyme, wherein the enzyme variant has enzymatic activity.
- Enzyme variants may be defined by their sequence similarity when compared to a parent en- zyme. Sequence similarity usually is provided as“% sequence similarity” or“%-similarity”. % sequence similarity takes into account that defined sets of amino acids share similar properties, e.g by their size, by their hydrophobicity, by their charge, or by other characteristics. Herein, the exchange of one amino acid with a similar amino acid may be called“conservative mutation”.
- amino acid A is similar to amino acids S; amino acid D is similar to amino acids E and N; amino acid E is simi- lar to amino acids D and K and Q; amino acid F is similar to amino acids W and Y; amino acid H is similar to amino acids N and Y; amino acid I is similar to amino acids L and M and V; amino acid K is similar to amino acids E and Q and R; amino acid L is similar to amino acids I and M and V; amino acid M is similar to amino acids I and L and V; amino acid N is similar to amino acids D and H and S; amino acid Q is similar to amino acids E and K and R; amino acid R is similar to amino acids K and Q; amino acid S is similar to amino acids A and N and T; amino acid T is similar to amino acids S; amino acid V is similar to amino acids I and L and M; amino acid W is similar to amino acids F and Y; amino acid Y is similar to amino acids F and H and W.
- Conservative amino acid substitutions may occur over the full length of the sequence of a poly- peptide sequence of a functional protein such as an enzyme.
- such muta- tions are not pertaining the functional domains of an enzyme.
- conservative mutations are not pertaining the catalytic centers of an enzyme.
- %-similarity [(iden- tical residues + similar residues) / length of the alignment region which is showing the respec- tive sequence(s) of this invention over its complete length]-100.
- enzyme variants may be described as an amino acid sequence which is at least m% similar to the respective parent sequences with“m” being an integer between 10 and 100.
- variant enzymes are at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% similar when corn- pared to the full length polypeptide sequence of the parent enzyme, wherein the variant enzyme has enzymatic activity.
- Enzymatic activity means the catalytic effect exerted by an enzyme, which usually is ex- pressed as units per milligram of enzyme (specific activity) which relates to molecules of sub- strate transformed per minute per molecule of enzyme (molecular activity).
- Variant enzymes may have enzymatic activity according to the present invention when said en- zyme variants exhibit at least 20%, at least 25%, at least 30%, at least 35%, at least 40%, at least 45%, at 10 least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or 100% of the enzymatic activity of the respective parent enzyme.
- inventive compositions comprise at least one protease (A).
- Enzymes (A) having proteolytic activity are called“proteases” or peptidases in the context of the present in- vention.
- Such enzymes are members of class EC 3.4.
- Proteases (A) are further classified as aminopeptidases (EC 3.4.11 ), dipeptidases (EC 3.4.13), dipeptidyl-peptidases and tripeptidyl-peptidases (EC 3.4.14), peptidyl-dipeptidases (EC 3.4.15), serine-type carboxypeptidases (EC 3.4.16), metallocarboxypeptidases (EC 3.4.17), cysteine- type carboxypeptidases (EC 3.4.18), omega peptidases (EC 3.4.19), serine endopeptidases (EC 3.4.21 ), cysteine endopeptidases (EC 3.4.22), aspartic endopeptidases (EC 3.4.23), metal- lo-endopeptidases (EC 3.4.24), threonine endopeptidases (EC 3.4.25), or endopeptidases of unknown catalytic mechanism (EC 3.4.99).
- aminopeptidases EC 3.4.11
- dipeptidases EC 3.4.
- Protease (A) may be an endopeptidase of any kind or a mixture of endopeptidases of any kind.
- protease according to the invention is selected from serine protease (EC 3.4.21 ).
- Serine proteases or serine peptidases are characterized by having a serine in the catalytically active site, which forms a covalent adduct with the substrate during the catalytic reaction.
- a ser- ine protease in the context of the present invention is selected from the group consisting of chymotrypsin (e.g., EC 3.4.21.1 ), elastase (e.g., EC 3.4.21.36), elastase (e.g., EC 3.4.21.37 or EC 3.4.21.71 ), granzyme (e.g., EC 3.4.21.78 or EC 3.4.21.79), kallikrein (e.g., EC 3.4.21.34, EC 3.4.21.35, EC 3.4.21.118, or EC 3.4.21.119,) plasmin (e.g., EC 3.4.21.7), trypsin (e.g., EC 3.4.21.4), thrombin (e.g.
- Crystallographic structures of proteases (A) show that the active site is commonly located in a groove on the surface of the molecule between adjacent structural domains, and the substrate specificity is governed by the properties of binding sites arranged along the groove on one or both sides of the catalytic site that is responsible for hydrolysis of the scissile bond. Accordingly, the specificity of protease (A) can be described by use of a conceptual model in which each specificity subsite is able to accommodate the sidechain of a single amino acid residue.
- the sites are numbered from the catalytic site, S1 , S2...Sn towards the N-terminus of the substrate, and S1 ', S2'...Sn' towards the C-terminus.
- the residues they accommodate are numbered P1 , P2...Pn, and PT, P2'...Pn', respectively:
- protease activity In general, the three main types of protease activity (proteolytic activity) are: trypsin-like, where there is cleavage of amide substrates following Arg (N) or Lys (K) at P1 , chymotrypsin-like where cleavage occurs following one of the hydrophobic amino acids at P1 , and elastase-like with cleavage following an Ala (A) at P1.
- subtilases A sub-group of the serine proteases tentatively designated as subtilases has been proposed by Siezen et al. (1991 ), Protein Eng. 4:719-737 and Siezen et al. (1997), Protein Science 6:501 - 523. They are defined by homology analysis of more than 170 amino acid sequences of serine proteases previously referred to as subtilisin-like proteases. A subtilisin was previously often defined as a serine protease produced by Gram-positive bacteria or fungi, and according to Siezen et al. now is a subgroup of the subtilases. A wide variety of subtilases have been identi fied, and the amino acid sequence of a number of subtilases has been determined.
- subtilases may be divided into 6 sub-divisions, i.e. the subtilisin family, the thermitase family, the proteinase K family, the lantibiotic peptidase family, the kexin family and the pyrolysin family.
- subtilases A subgroup of the subtilases are the subtilisins which are serine proteases from the family S8 as defined by the MEROPS database (http://merops.sanger.ac.uk).
- Peptidase family S8 con- tains the serine endopeptidase subtilisin and its homologues.
- subfamily S8A the active site residues frequently occur in the motifs Asp-Thr/Ser-Gly similarly to the sequence motif in fami- lies of aspartic endopeptidases in clan AA, His-Gly-Thr-His and Gly-Thr-Ser-Met-Ala-Xaa-Pro.
- Prominent members of family S8, subfamily A are:
- subtilisin related class of serine proteases shares a common amino acid sequence defining a catalytic triad which distinguishes them from the chymotrypsin related class of serine proteas- es.
- Subtilisins and chymotrypsin related serine proteases both have a catalytic triad comprising aspartate, histidine and serine.
- subtilisin related proteases In subtilisin related proteases (A) the relative order of these amino acids, reading from the ami- no to carboxy-terminus is aspartate-histidine-serine. In the chymotrypsin related proteases the relative order, however is histidine-aspartate-serine.
- subtilisin herein refers to a serine protease having the catalytic triad of subtilisin related proteases. Examples include the subtil isins as described in WO 89/06276 and EP 0283075, WO 89/06279, WO 89/09830, WO 89/09819, WO 91/06637 and WO 91/02792.
- Parent proteases of the subtilisin type (EC 3.4.21.62) and variants may be bacterial proteases.
- Said bacterial protease may be a Gram-positive bacterial polypeptide such as a Bacillus, Clos tridium, Enterococcus, Geobacillus, Lactobacillus, Lactococcus, Oceanobacillus, Staphylococ cus, Streptococcus, or Streptomyces protease, or a Gram-negative bacterial polypeptide such as a Campylobacter, E. cdi, Flavobacterium, Fusobacterium, Helicobacter, Hyobacter, Neis seria, Pseudomonas, Salmonella, or Ureaplasma protease. They act as unspecific endopepti- dases, i.e. they hydrolyze any peptide bonds.
- protease enzymes include those sold under the trade names Alcalase®, Blaze®, DuralaseTM, DurazymTM, Relase®, Relase® Ultra, Savinase®, Savinase® Ultra, Pri- mase®, Polarzyme®, Kannase®, Liquanase®, Liquanase® Ultra, Ovozyme®, Coronase®, Co- ronase® Ultra, Neutrase®, Everlase® and Esperase® (Novozymes A/S), those sold under the tradename Maxatase®, Maxacal®, Maxapem®, Purafect®, Purafect® Prime, Purafect MA®, Purafect Ox®, Purafect OxP®, Puramax®, Properase®, FN2®, FN3®, FN4®, Excellase®, Eraser®, Ultimase®, Opticlean®, Effectenz®, Preferenz® and Optimase®
- the parent enzymes and variants may be a Bacillus alcalophilus, Bacillus amyloliquefaciens, Bacillus brevis, Bacillus circulans, Bacillus dausii, Bacillus coagu- lans, Bacillus firmus, Bacillus gibsonii, Bacillus lautus, Bacillus lentus, Bacillus Hcheniformis, Bacillus megaterium, Bacillus pumiius, Bacillus sphaericus, Bacillus stearothermophiius, Bacil lus subti/is, or Bacillus thuringiensis protease.
- subtilase is selected from the following:
- Bacillus Hcheniformis subtilisin from Bacillus Hcheniformis (subtilisin Carlsberg; disclosed in EL Smith et al.
- Bacillus lentus as disclosed in WO 91/02792, such as from Bacillus lentus DSM 5483 or the variants of Bacillus lentus DSM 5483 as described in WO 95/23221 ,
- subtilisin having SEQ ID NO: 4 as described in WO 2005/063974 or a subtilisin which is at least 40% identical thereto and having proteolytic activity
- subtilisin having SEQ ID NO: 4 as described in WO 2005/103244 or subtilisin which is at least 80% identical thereto and having proteolytic activity
- - subtilisin having SEQ ID NO: 7 as described in WO 2005/103244 or subtilisin which is at least 80% identical thereto and having proteolytic activity
- subtilisin having SEQ ID NO: 2 as described in application DE 102005028295.4 or subtil- isin which is this at least 66% identical thereto and having proteolytic activity.
- Examples of useful proteases (A) in accordance with the present invention comprise the vari- ants described in: WO 92/19729, WO 95/23221 , WO 96/34946, WO 98/201 15, WO 98/201 16, WO 99/11768, WO 01/44452, WO 02/088340, WO 03/006602, WO 2004/03186, WO
- Suitable examples comprise especially protease variants of subtilisin protease derived from SEQ ID NO:22 as described in EP 1921147 (which is the sequence of mature alkaline protease from Bacillus lentus DSM 5483) with amino acid substitutions in one or more of the following positions: 3, 4, 9, 15, 24, 27, 33, 36, 57, 68, 76, 77, 87, 95, 96, 97, 98, 99, 100, 101 , 102, 103, 104, 106, 1 18, 120, 123, 128, 129, 130, 131 , 154, 160, 167, 170, 194, 195, 199, 205, 206, 217, 218, 222, 224, 232, 235, 236, 245, 248, 252 and 274 (according to the BPN' numbering), which have proteo
- subtilisin has SEQ ID NO:22 as described in EP 1921147, or a subtilisin which is at least 80% identical thereto and has proteolytic activity.
- a subtil- isin is at least 80% identical to SEQ ID NO:22 as described in EP 1921147 and is characterized by having amino acid glutamic acid (E), or aspartic acid (D), or asparagine (N), or glutamine (Q), or alanine (A), or glycine (G), or serine (S) at position 101 (according to BPN’ numbering) and has proteolytic activity.
- subtilisin is at least 80% identical to SEQ ID NO:22 as described in EP 1921147 and is characterized by having amino acid glutamic acid (E), or aspartic acid (D), at position 101 (according to BPN’ numbering) and has proteolytic activity.
- Such a subtilisin variant may comprise an amino acid substitution at position 101 , such as R101 E or R101 D, alone or in combination with one or more substitutions at positions 3, 4, 9, 15, 24, 27, 33, 36, 57, 68, 76, 77, 87, 95, 96, 97, 98, 99, 100, 101 , 102, 103, 104, 106, 118, 120, 123, 128, 129, 130, 131 , 154, 160, 167, 170, 194, 195, 199, 205, 206, 217, 218, 222, 224, 232, 235, 236, 245, 248, 252 and/or 274 (according to BPN’ numbering) and has proteolytic activity.
- a subtilisin is at least 80% identical to SEQ ID NO:22 as described in EP 1921147 and is characterized by comprising at least the following amino acids (according to BPN’ numbering) and has proteolytic activity:
- a subtilisin is at least 80% identical to SEQ ID NO:22 as described in EP 1921147 and is characterized by comprising one amino acid (according to (a)-(h)) or combi- nations according to (i) together with the amino acid 101 E, 101 D, 101 N, 101 Q, 101A, 101 G, or 101 S (according to BPN’ numbering) and has proteolytic activity.
- a subtilisin is at least 80% identical to SEQ ID NO:22 as described in EP 1921147 and is characterized by comprising the mutation (according to BPN’ numbering)
- R101 E or S3T + V4I + V205I, or S3T + V4I + V199M + V205I + L217D and has proteolytic ac- tivity.
- the subtilisin comprises an amino acid sequence having at least 80% identity to SEQ ID NO:22 as described in EP 1921147 and being further characterized by corn- prising R101 E and S3T, V4I, and V205I (according to the BPN’ numbering) and has proteolytic activity.
- a subtilisin comprises an amino acid sequence having at least 80% identical to SEQ ID NO:22 as described in EP 1921 147 and being further characterized by comprising R101 E, and one or more substitutions selected from the group consisting of S156D, L262E, Q137H, S3T, R45E,D,Q, P55N, T58W,Y,L, Q59D,M,N,T, G61 D,R, S87E, G97S, A98D,E,R, S106A,W, N117E, H120V,D,K,N, S125M, P129D, E136Q, S144W, S161T,
- subtilisin variant enzymes as disclosed above which are at least n% identical to the respective parent sequences include variants with n being at least 40 to 100.
- subtilisin variants in one embodiment have proteolytic activity and are at least 40%, at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical when compared to the full length polypeptide sequence of the parent enzyme.
- subtilisin variants comprising conservative mu- tations not pertaining the functional domain of the respective subtilisin protease.
- subtilisin variants of this embodiment have proteolytic activity and are at least 40%, at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% similar when compared to the full length polypeptide sequence of the parent enzyme.
- protease proteolysis, which means hydrolysis of peptide bonds linking amino acids together in a polypeptide chain
- protease proteolysis, which means hydrolysis of peptide bonds linking amino acids together in a polypeptide chain
- proteolytic activity is related to the rate of degradation of protein by a protease or pro- teolytic enzyme in a defined course of time.
- the methods for analyzing proteolytic activity are well-known in the literature (see e.g. Gupta et al. (2002), Appl.
- Proteolytic activity as such can be determined by using Succinyl-Ala-Ala-Pro-Phe-p- nitroanilide (Suc-AAPF-pNA, short AAPF; see e.g. DelMar et al. (1979), Analytical Biochem 99, 316-320) as substrate.
- pNA is cleaved from the substrate molecule by proteolytic cleavage, re- sulting in release of yellow color of free pNA which can be quantified by measuring OD 405 .
- Protease variants may have proteolytic activity when said protease variants exhibit at least 20%, at least 25%, at least 30%, at least 35%, at least 40%, at least 45%, at 10 least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or 100% of the proteolytic activity of the respective parent protease.
- the pi value (isoelectric point) of subtilisin protease used in the present invention is in the range of from pH 7.0 to pH 10.0, preferably from pH 8.0 to pH 9.5.
- inventive compositions comprise at least one lipase (A).“Lipases”,“lipolytic enzyme”,“lipid esterase”, all refer to an enzyme of EC class 3.1.1 (“carboxylic ester hydrolase”).
- Such an enzyme (A) may have lipase activity (or lipolytic activity; triacylglycerol lipase, EC 3.1.1.3), cutinase activity (EC 3.1.1.74; enzymes having cutinase activity may be called cutinase herein), sterol esterase activity (EC 3.1.1.13) and/or wax-ester hydrolase activity (EC 3.1.1.50).
- Lipases include those of bacterial or fungal origin.
- lipase (A) include but are not limited to those sold under the trade names LipolaseTM, LipexTM, LipolexTM and LipocleanTM (Novozymes A/S), Lumafast (originally from Genencor) and Lipomax (Gist-Brocades/ now DSM).
- a suitable lipase is selected from the following:
- Humicoia (synonym Thermomyces), e.g. from H. lanuginosa ( T. lanuginosus) as described in EP 258068, EP 305216, WO 92/05249 and WO 2009/109500 or from H. insolens as described in WO 96/13580,
- Suitable lipases (A) also include those referred to as acyltransferases or perhydrolases, e.g. acyltransferases with homology to Candida antarctica lipase A (WO 2010/11 1143), acyltrans- ferase from Mycobacterium smegmatis (WO 2005/056782), perhydrolases from the CE7 family (WO 2009/67279), and variants of the M. smegmatis perhydrolase in particular the S54V variant (WO 2010/100028).
- acyltransferases or perhydrolases e.g. acyltransferases with homology to Candida antarctica lipase A (WO 2010/11 1143), acyltrans- ferase from Mycobacterium smegmatis (WO 2005/056782), perhydrolases from the CE7 family (WO 2009/67279), and variants of the M. smegmatis perhydrolase in particular the S54V variant (
- Suitable lipases include also those which are variants of the above described lipases and/or cutinases which have lipolytic activity.
- Such suitable lipase variants are e.g. those which are developed by methods as disclosed in WO 95/22615, WO 97/04079, WO 97/07202, WO 00/60063, WO 2007/087508, EP 407225 and EP 260105.
- Suitable lipases/cutinases include also those that are variants of the above described lipas- es/cutinases which have lipolytic activity.
- Suitable lipase/cutinase variants include variants with at least 40 to 100% identity when compared to the full length polypeptide sequence of the par- ent enzyme as disclosed above.
- lipase/cutinase variants having lipolytic activity may be at least 40%, at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98% or at least 99% identical when compared to the full length polypeptide sequence of the parent en- zyme as disclosed above.
- inventive compositions comprise at least one lipase/cutinase variant comprising conservative mutations not pertaining the functional domain of the respective li- pase/cutinase.
- Lipase/cutinase variants of such embodiments having lipolytic activity may be at least 40%, at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98% or at least 99% similar when compared to the full length polypeptide sequence of the parent enzyme.
- Lipases have“lipolytic activity”.
- the methods for determining lipolytic activity are well-known in the literature (see e.g. Gupta et al. (2003), Biotechnol. Appl. Biochem. 37, p. 63-71 ).
- the lipase activity may be measured by ester bond hydrolysis in the substrate para-nitrophenyl pal- mitate (pNP-Palmitate, C:16) and releases pNP which is yellow and can be detected at 405 nm.
- Lipase variants may have lipolytic activity according to the present invention when said lipase variants exhibit at least 20%, at least 25%, at least 30%, at least 35%, at least 40%, at least 45%, at 10 least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or 100% of the lipolytic activity of the re- spective parent lipase.
- a combination of at least two of the foregoing li- pases (A) may be used.
- Lipase (A) may be used in its non-purified form or in a purified form, e.g. purified with the aid of well-known adsorption methods, such as phenyl sepharose adsorption techniques.
- lipases (A) are included in inventive composition in such an amount that a finished inventive composition has a lipolytic enzyme activity in the range of from 100 to 0.005 LU/mg, preferably 25 to 0.05 LU/mg of the composition.
- proteases (A) are included in inventive composition in such an amount that a finished inventive composition has a proteolytic enzyme activity in the range of from 0.1 to 50 GU.
- lipase (A) and protease (A) in compositions, for example 1 to 2% by weight of protease (A) and 0.1 to 0.5% by weight of lipase (A).
- an enzyme (A) is called stable when its enzymatic activi- ty“available in application” equals 100% when compared to the initial enzymatic activity before storage.
- An enzyme may be called stable within this invention if its enzymatic activity available in application is at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or at least 99.5% when compared to the initial enzymatic activity before storage.
- lipolytic activity available after storage at 37°C for 30 days is at least 60% when compared to the initial lipolytic activity before storage.
- an enzyme is stable accord- ing to the invention when essentially no loss of enzymatic activity occurs during storage, i.e. loss in enzymatic activity equals 0% when compared to the initial enzymatic activity before stor- age.
- no loss of enzymatic activity within this invention may mean that the loss of enzymatic activity is less than 30%, less than 25%, less than 20%, less than 15%, less than 10%, less than 9%, less than 8%, less than 7%, less than 6%, less than 5%, less than 4%, less than 3%, less than 2%, or less than 1 % when compared to the initial enzymatic activity before storage.
- the loss of lipolytic activity after storage at 37°C for 30 days is less than 40% when compared to the initial lipolytic activity before storage.
- Reduced loss of enzymatic activity within this invention may mean that the loss of enzymatic activity is reduced by at least 5%, by at least 10%, by at least 15%, by at least 20%, by at least 25%, by at least 30%, by at least 40%, by at least 50%, by least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or at least 99.5% when compared to the initial enzymatic activity before storage.
- Compartment 1 further comprises chelating agent (B) dissolved in said aqueous medium, said chelating agent (B) being
- (B1 ) at least one alkali metal salt of glutamic acid diacetic acid (GLDA), hereinafter also re- ferred to as complexing agent (B1 ) or simply as (B1 ), and
- (B2) at least one alkali metal salt of methyl glycine diacetic acid (MGDA), hereinafter altogether also referred to as complexing agent (B2) or simply as (B2).
- MGDA methyl glycine diacetic acid
- alkali refers to alkali metal cations, same or different, for example cations of lithium, sodium, potassium, rubidium, cesium, and combinations of at least two of the foregoing.
- Pre- ferred examples of alkali metal cations are sodium and potassium and combinations of sodium and potassium.
- Most preferred alkali metal cation is sodium.
- Alkali metal salts may refer to fully or partially neutralized with alkali but not to ammonium ions in relation to the carboxyl groups of MGDA and GLDA.
- Alkali metal salts may refer to fully or partially neutralized with alkali but not to ammonium ions in relation to the carboxyl groups of MGDA and GLDA.
- (B1 ) is at least one alkali metal salt of L- and D-enantiomers of glutamic acid diacetic acid (GLDA) or of enantiomerically pure L-GLDA, with an average of more than three of the carboxyl groups being neutralized with alkali, preferably an average of more than three and less than four of the carboxyl groups are neutralized with alkali metal, and
- (B2) being at least one alkali metal salt of methyl glycine diacetic acid (MGDA), with an average of more than two and less than three of the carboxyl groups being neutralized with alkali.
- MGDA methyl glycine diacetic acid
- carboxyl groups in the above definition refers to carboxyl groups per molecule.
- Alkali metal salts of GLDA are selected from compounds according to general formula (I)
- M is selected from alkali metal cations, same or different, as defined above,
- variable x1 in formula (I) may be in the range of from zero to 2.0, preferred are 0.02 to 0.5. In particularly preferred embodiments, x1 is from 0.1 to 0.3.
- variable z1 in formula (I) may be in the range of from zero to 1.0, preferred are 0.0005 to 0.5.
- the sum of x1 +z1 may be zero.
- the sum of x1 +z1 in formula (I) is prefera- bly greater than zero, for example 0.05 to 0.6.
- Examples of M 4 -c ⁇ - z ⁇ (NH 4 ) z 2Hc ⁇ are Na 4-xi H xi , [Nao .7 (NH 4 ) 0.3 ] 4-xi H xi , [(NH 4 ) 0.7 Nao .3 ] 4-xi H xi ,
- M 4-xi H xi are selected from Na 4-xi H xi , (Ko esNao -isK x -iH x -i, and (Nao . ssKo . -isK xi H x -i.
- M is the same for (B1 ) and (B2).
- the ratio of such different alkali metal cations is preferably the same for both MGDA and GLDA.
- the aqueous medium containing enzyme (A) and complexing agent(s) (B) have a pH value in the range of from 8 to 1 1 , preferably 9 to 10.8.
- the pH value is determined at ambient temperature at a dilution of 1 % by weight solids content.
- complexing agent (B1 ) is selected from at least one alkali metal salt of a mixture of L- and D- enantiomers according to formula (I), said mixture containing the racemic mixture or preferably predominantly the respective L-isomer, for example with an enantiomeric excess (ee) in the range of from 5 to 99%, preferably 5 to 95%.
- Alkali metal salts of MGDA are selected from compounds according to general formula (II)
- M is selected from alkali metal cations, same or different, for example cations of lithium, sodium, potassium, rubidium, cesium, and combinations of at least two of the foregoing.
- Preferred ex- amples of alkali metal cations are sodium and potassium and combinations of sodium and po- tassium.
- variable x2 in formula (II) may be in the range of from zero to 1 .0, preferred are 0.01 to 0.5. In particularly preferred embodiments, x2 is from 0.1 to 0.2.
- variable z2 in formula (II) may be in the range of from zero to 1 .0, preferred are 0.0005 to 0.5.
- M3-x2-z2(NH 4 ) Z2 H X2 are Na3-x2H X2 , [Nao.7(NH 4 )o.3]3-x2H X2 , [(NH 4 )o.7Nao.3]3-x2H X2 ,
- M 3 -x2(NH ) Z 2H X 2 are selected from Na3-x2H x2, (K 0 .85Nao.i5)3-x2H X 2, and (Nao.ssKo.-is
- complexing agent (B2) is selected from at least one alkali metal salt of racemic MGDA and from alkali metal salts of mixtures of L- and D- enantiomers according to formula (II), said mixture containing predominantly the respective L- isomer with an enantiomeric excess (ee) in the range of from 5 to 99%, preferably 15 to 80 %, more preferably from 10 to 75% and even more preferably from 10 to 66%.
- the total degree of alkali neutralization of (B2) and (B1 ) is in the range of from 0.80 to 0.98 mol-%, preferred are 0.90 to 0.97%.
- the total degree of alkali neutralization does not take into account any neutralization with ammonium.
- the enantiomeric excess of MGDA and of its salts may be determined by measuring the polari- zation (polarimetry) or preferably by chromatography, for example by HPLC with a chiral col- umn, for example with one or more cyclodextrins as immobilized phase or with a ligand ex- change (Pirkle-brush) concept chiral stationary phase. Preferred is determination of the ee by HPLC with an immobilized optically active amine such as D-penicillamine in the presence of copper(ll) salt.
- the enantiomeric excess of GLDA and of its salts may be determined by meas- uring the polarization (polarimetry).
- alkali metal salts of complexing agent(s) (B) may contain one or more impurities that may result from the synthesis of the respective complexing agent(s) (B).
- impurities may be propionic acid, lactic acid, glutamate, alanine, nitrilotriacetic acid (NTA) or the like and their respective alkali metal salts.
- Such impurities are usually present in minor amounts.“Minor amounts” in this context refer to a total of 0.1 to 5% by weight, referring to alkali metal salt of complexing agent (B), preferably up to 2.5% by weight.
- compartment 1 contains complexing agent (B1) as sole complexing agent.
- the weight ratio of (B1 ) to (B1 ) or their respective salts in the mixture is in the range of from 1 :9 to 9:1 , preferably 1 : 3 to 3 : 1 , more preferably 2 : 3 to 3 : 2.
- the weight ratio may be determined, for example, by chromatographic methods such as HPLC.
- the ee refers to the enantiomeric excess of all L-isomers present in the respective mixture compared to all D- isomers.
- the ee refers to the sum of the disodium salt and trisodium salt of L-MGDA with respect to the sum of the disodium salt and the trisodium salt of D-MGDA.
- complexing agent(s) (B) may contain in the range of from 0.1 to 10 % by weight of one or more optically inactive impurities, at least one of the impurities being selected from iminodiacetic acid, formic acid, glycolic acid, propionic acid, ace- tic acid and their respective alkali metal or mono-, di- or triammonium salts.
- inventive mixtures may contain less than 0.2 % by weight of nitrilotri- acetic acid (NTA), preferably 0.01 to 0.1 % by weight. The percentages refer to total complexing agent(s) (B).
- complexing agent(s) (B) may contain in the range of from 0.1 to 10 % by weight of one or more optically active impurities, at least one of the impu- rities being selected from L-carboxymethylalanine and its respective mono- or dialkali metal salts, L-carboxymethylglutamic acid and its respective di- or trialkali metal salts, and optically active mono- or diamides that result from an incomplete saponification during the synthesis of complexing agent(s) (B).
- the amount of optically active impurities is in the range of from 0.2 to 10 % by weight, referring to the sum of complexing agent(s) (B).
- complexing agent(s) (B) may contain minor amounts of cations other than alkali metal or ammonium. It is thus possible that minor amounts, such as 0.01 to 5 mol-% of total complexing agent, based on anion, bear alkali earth metal cati ons such as Mg 2+ or Ca 2+ , or transition metal ions such as Fe 2+ or Fe 3+ cations.
- the overall concentration of chelating agent(s) (B) in compartment 1 is in the range of from 35 to 75 % by weight, preferably 40 to 70% by weight, more preferably 42 to 70 % by weight and even more preferably 46 to 65 % by weight.
- Aqueous medium refers to media in which the solvent is essentially water. In one embodiment, in such aqueous medium water is the sole solvent. In other embodiments, mixtures of water with one or more water-miscible solvents are used as aqueous medium.
- water- miscible solvent refers to organic solvents that are miscible with water at ambient temperature without phase-separation. Examples are ethylene glycol, 1 ,2-propylene glycol, isopropanol, and diethylene glycol. Preferably, at least 50 % by volume of the respective aqueous medium is wa- ter, referring to the solvent.
- the aqueous medium containing complexing agent(s) (B) contains at least one inorganic basic salt selected from alkali metal carbonates.
- alkali metal carbonates Preferred examples are sodium carbonate and potassium carbonate, for example 0.1 to 1.5 % by weight.
- compartment 1 contains 0.5 to 20 g aqueous me- dium containing enzyme (A) and complexing agent(s) (B), preferably 1 to 8 g.
- the solution containing enzyme (A) and complexing agent(s) (B) contains at least one dyestuff.
- dyestuffs are Acid Red 1 , Acid Red 52, Acid Blue 9, Acid Yellow 3, Acid Yellow 23, Acid Yellow 73, Acid Green 1 , Solvent Green 7, and Acid Green 25.
- the solutions containing complexing agent(s) (B) addi- tionally contain a pigment formulation, for example a formulation of Cl Pigment Yellow 101 or Cl Pigment Blue 15:1.
- the solution containing enzyme (A) and complexing agent(s) (B) may contain a viscosity modi- fying agent, for example a thickener.
- a viscosity modi- fying agent for example a thickener.
- the solution containing enzyme (A) and complexing agent(s) (B) does not contain any thickener.
- the aqueous medium in compartment 1 may have an opaque or translucent or transparent ap- pearance. Preferably, neither haze nor precipitations may be detected on eyesight.
- Inventive units contain one more compartment - hereinafter also referred to as additional com- partment - comprising at least one ingredient of a detergent other than enzyme (A) and chelat- ing agent(s) (B).
- a detergent other than enzyme A
- chelat- ing agent(s) B
- examples of such ingredients are surfactants or a combination of surfactants, a bleaching agent, a bleach catalyst, or a builder other than complexing agents (B).
- inventive units may contain two or up to 10 further compartments with identical or different compositions of detergents.
- in- ventive units comprise compartment 1 and a maximum of one or two additional compartments.
- Detergent compositions comprised in said additional compartment(s) may contain one or more complexing agent other than MGDA and GLDA.
- complexing agent other than MGDA and GLDA are citrate, phosphonic acid derivatives, for example the disodium salt of hy- droxyethane-1 ,1-diphosphonic acid (“HEDP”), for example trisodium citrate, and phosphates such as STPP (sodium tripolyphosphate). Due to the fact that phosphates raise environmental concerns, it is preferred that detergent compositions comprised in inventive containers are free from phosphate.
- Free from phosphate is to be understood in the context of the present inven- tion as meaning that the content of phosphate and polyphosphate is in sum in the range from 10 ppm to 0.2% by weight, determined by gravimetric analysis and referring to the total detergent composition.
- Detergent compositions comprised in said additional compartment(s) may contain one or more surfactant, preferably one or more non-ionic surfactant.
- Preferred non-ionic surfactants are alkoxylated alcohols, di- and multiblock copolymers of eth- ylene oxide and propylene oxide and reaction products of sorbitan with ethylene oxide or pro- pylene oxide, alkyl polyglycosides (APG), hydroxyalkyl mixed ethers and amine oxides.
- APG alkyl polyglycosides
- alkoxylated alcohols and alkoxylated fatty alcohols are, for example, compounds of the general formula (III)
- R 2 is identical or different and selected from hydrogen and linear Ci-Cio-alkyl, preferably in each case identical and ethyl and particularly preferably hydrogen or methyl,
- R 3 is selected from C8-C22-alkyl, branched or linear, for example n-CsH , n-C-ioFh-i, n-C- ⁇ Fhs, n-Ci 4 H29, n-Ci6H33 or n-CieH37,
- R 4 is selected from Ci-Cio-alkyl, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, n-pentyl, isopentyl, sec-pentyl, neopentyl, 1 ,2-dimethylpropyl, isoamyl, n-hexyl, isohexyl, sec-hexyl, n-heptyl, n-octyl, 2-ethylhexyl, n-nonyl, n-decyl or isodecyl.
- e and f are in the range from zero to 300, where the sum of e and f is at least one, preferably in the range of from 3 to 50.
- e is in the range from 1 to 100 and f is in the range from 0 to 30.
- compounds of the general formula (II I) may be block copolymers or random copolymers, preference being given to block copolymers.
- alkoxylated alcohols are, for example, compounds of the general formula (IV)
- R 2 is identical or different and selected from hydrogen and linear Ci-Co-alkyl, preferably iden- tical in each case and ethyl and particularly preferably hydrogen or methyl,
- R 5 is selected from C6-C2o-alkyl, branched or linear, in particular n-CsH ⁇ , n-C-ioHh-i, n-Ci2H25, n-Ci3H27, h-O ⁇ dHb ⁇ , n-Ci 4 H29, h-O ⁇ qH33, n-CieH37, a is a number in the range from zero to 10, preferably from 1 to 6, b is a number in the range from 1 to 80, preferably from 4 to 20, d is a number in the range from zero to 50, preferably 4 to 25.
- the sum a + b + d is preferably in the range of from 5 to 100, even more preferably in the range of from 9 to 50.
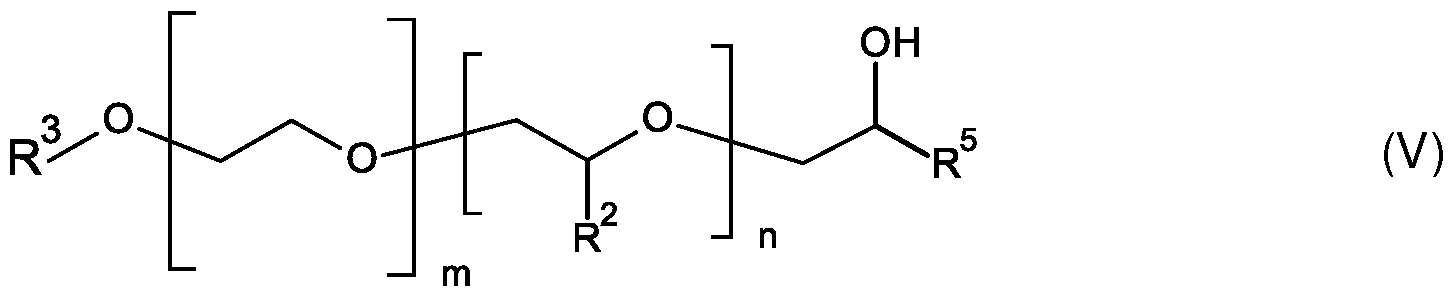
- Preferred examples for hydroxyalkyl mixed ethers are compounds of the general formula (V)
- R 2 is identical or different and selected from hydrogen and linear Ci-Cio-alkyl, preferably in each case identical and ethyl and particularly preferably hydrogen or methyl,
- R 3 is selected from C 8 -C 22 -alkyl, branched or linear, for example iso-CnH23, iso-Ci3H27, n- CeHi7, n-CioH2i, n-Ci2H25, n-Ci 4 H29, n-Ci6H33 or n-CieH37,
- R 4 is selected from Ci-Cie-alkyl, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, n-pentyl, isopentyl, sec-pentyl, neopentyl, 1 ,2-dimethylpropyl, isoamyl, n-hexyl, isohexyl, sec-hexyl, n-heptyl, n-octyl, 2-ethylhexyl, n-nonyl, n-decyl, isodecyl, n-dodecyl, n-tetradecyl, n-hexadecyl, and n-octadecyl.
- n and n are in the range from zero to 300, where the sum of n and m is at least one, preferably in the range of from 5 to 50.
- m is in the range from 1 to 100 and n is in the range from 0 to 30.
- Compounds of the general formula (IV) and (V) may be block copolymers or random copoly- mers, preference being given to block copolymers.
- nonionic surfactants are selected from di- and multiblock copolymers, corn- posed of ethylene oxide and propylene oxide. Further suitable nonionic surfactants are selected from ethoxylated or propoxylated sorbitan esters. Amine oxides or alkyl polyglycosides, espe- cially linear C 4 -C-i 6 -alkyl polyglucosides and branched Cs-C-u-alkyl polyglycosides such as corn- pounds of general average formula (VI) are likewise suitable.
- R 6 is Ci-C 4 -alkyl, in particular ethyl, n-propyl or isopropyl,
- R 7 is -(CH 2 ) 2 -R 6 ,
- G 1 is selected from monosaccharides with 4 to 6 carbon atoms, especially from glucose and xylose, y in the range of from 1.1 to 4, y being an average number,
- non-ionic surfactants are compounds of general formula (VII) and (VIII)
- AO is selected from ethylene oxide, propylene oxide and butylene oxide,
- EO is ethylene oxide, CH2CH2-O,
- R 8 selected from Cs-C-is-alkyl, branched or linear, and R 5 is defined as above.
- a 3 0 is selected from propylene oxide and butylene oxide,
- w is a number in the range of from 15 to 70, preferably 30 to 50,
- w1 and w3 are numbers in the range of from 1 to 5, and
- w2 is a number in the range of from 13 to 35.
- An overview of suitable further nonionic surfactants can be found in EP-A 0 851 023 and in DE- A 198 19 187.
- Nonionic surfactants may also be present.
- Other surfactants that may be present are selected from amphoteric (zwitterionic) surfactants and anionic surfactants and mixtures thereof.
- amphoteric surfactants are those that bear a positive and a negative charge in the same molecule under use conditions.
- Preferred examples of amphoteric surfactants are so- called betaine-surfactants.
- Many examples of betaine-surfactants bear one quaternized nitrogen atom and one carboxylic acid group per molecule.
- a particularly preferred example of amphoter- ic surfactants is cocamidopropyl betaine (lauramidopropyl betaine).
- amine oxide surfactants are compounds of the general formula (IX)
- R 9 is selected from C8-C20- alkyl or C2-C 4 -alkylene Cio-C2o-alkylamido and R 10 and R 11 are both methyl.
- a particularly preferred example is lauryl dimethyl aminoxide, sometimes also called lauramine oxide.
- a further particularly preferred example is cocamidylpropyl dimethylaminoxide, some- times also called cocamidopropylamine oxide.
- Suitable anionic surfactants are alkali metal and ammonium salts of Cs-C-is-alkyl sulfates, of Cs-C-is-fatty alcohol polyether sulfates, of sulfuric acid half-esters of ethoxylated C 4 - Ci2-alkylphenols (ethoxylation: 1 to 50 mol of ethylene oxide/mol), C12-C18 sulfo fatty acid alkyl esters, for example of C12-C18 sulfo fatty acid methyl esters, furthermore of Ci2-Ci8-alkylsulfonic acids and of Cio-Ci 8 -alkylarylsulfonic acids.
- Suitable anionic surfactants are soaps, for example the sodium or potassi- um salts of stearic acid, oleic acid, palmitic acid, ether carboxylates, and alkylether phosphates.
- detergent compositions comprised in inventive containers may contain 0.1 to 60 % by weight of at least one surfactant, selected from anionic surfactants, amphoteric surfactants and amine oxide surfactants.
- detergent compositions comprised in said additional compart- ment(s) do not contain any anionic surfactant.
- Detergent compositions comprised in said additional compartment(s) may contain at least one bleaching agent, also referred to as bleach.
- Bleaching agents may be selected from chlorine bleach and peroxide bleach, and peroxide bleach may be selected from inorganic peroxide bleach and organic peroxide bleach.
- peroxide bleach may be selected from inorganic peroxide bleach and organic peroxide bleach.
- Preferred are inorganic peroxide bleaches, selected from alkali metal percarbonate, alkali metal perborate and alkali metal persulfate.
- organic peroxide bleaches are organic percarboxylic acids, especially organic per- carboxylic acids.
- alkali metal percarbonates especially sodium percarbonates
- Such coatings may be of organic or inorganic nature.
- glycerol sodium sulfate, silicate, sodium carbonate, and combinations of at least two of the foregoing, for example combinations of sodium carbonate and sodium sulfate.
- Suitable chlorine-containing bleaches are, for example, 1 ,3-dichloro-5,5-dimethylhydantoin, N-chlorosulfamide, chloramine T, chloramine B, sodium hypochlorite, calcium hypochlorite, magnesium hypochlorite, potassium hypochlorite, potassium dichloroisocyanurate and sodium dichloroisocyanurate.
- Detergent compositions comprised in said additional compartment(s) compositions may corn- prise, for example, in the range from 3 to 10% by weight of chlorine-containing bleach.
- Detergent compositions comprised in said additional compartment(s) may comprise one or more bleach catalysts.
- Bleach catalysts can be selected from bleach-boosting transition metal salts or transition metal complexes such as, for example, manganese-, iron-, cobalt-, ruthenium- or molybdenum-salen complexes or carbonyl complexes.
- Manganese, iron, cobalt, ruthenium, molybdenum, titanium, vanadium and copper complexes with nitrogen-containing tripod ligands and also cobalt-, iron-, copper- and ruthenium-amine complexes can also be used as bleach catalysts.
- Detergent compositions comprised in said additional compartment(s) may comprise one or more bleach activators, for example N-methylmorpholinium-acetonitrile salts (“MMA salts”), tri- methylammonium acetonitrile salts, N-acylimides such as, for example, N-nonanoylsuccinimide, 1 ,5-diacetyl-2,2-dioxohexahydro-1 ,3,5-triazine (“DADHT”) or nitrile quats (trimethylammonium acetonitrile salts).
- MMA salts N-methylmorpholinium-acetonitrile salts
- DADHT dioxohexahydro-1 ,3,5-triazine
- nitrile quats trimethylammonium acetonitrile salts
- TAED tetraacetylethylenediamine
- TAED tetraacetylhexylenediamine
- Detergent compositions comprised in said additional compartment(s) may comprise one or more corrosion inhibitors.
- corrosion inhibitors include tria zoles, in particular benzotriazoles, bisbenzotriazoles, aminotriazoles, alkylaminotriazoles, also phenol derivatives such as, for example, hydroquinone, pyrocatechol, hydroxyhydroquinone, gallic acid, phloroglucinol or pyrogallol.
- detergent compositions comprised in said addi- tional compartment(s) comprise in total in the range from 0.1 to 1.5% by weight of corrosion inhibitor.
- Detergent compositions comprised in said additional compartment(s) may comprise one or more builders, selected from organic and inorganic builders.
- suitable inorganic builders are sodium sulfate or sodium carbonate or silicates, in particular sodium disilicate and sodium metasilicate, zeolites, sheet silicates, in particular those of the formula a-I ⁇ ShOs, b- I ⁇ ShOs, and b-I ⁇ ShOs, also fatty acid sulfonates, a-hydroxypropionic acid, alkali metal ma- lonates, fatty acid sulfonates, alkyl and alkenyl disuccinates, tartaric acid diacetate, tartaric acid monoacetate, oxidized starch, and polymeric builders, for example polycarboxylates and poly- aspartic acid.
- organic builders are especially polymers and copolymers.
- organic builders are selected from polycarboxylates, for example alkali metal salts of (meth)acrylic acid homopolymers or (meth)acrylic acid copolymers.
- Suitable comonomers are monoethylenically unsaturated dicarboxylic acids such as maleic ac- id, fumaric acid, maleic anhydride, itaconic acid and citraconic acid.
- a suitable polymer is in par- ticular polyacrylic acid, which preferably has an average molecular weight M w in the range from 2000 to 40 000 g/mol, preferably 2000 to 10 000 g/mol, in particular 3000 to 8000 g/mol.
- copolymeric polycarboxylates in particular those of acrylic acid with methacrylic acid and of acrylic acid or methacrylic acid with maleic acid and/or fumaric acid, and in the same range of molecular weight.
- Suitable hydrophobic monomers are, for example, isobutene, diisobutene, butene, pentene, hexene and styrene, olefins with 10 or more carbon atoms or mixtures thereof, such as, for ex- ample, 1-decene, 1-dodecene, 1-tetradecene, 1 -hexadecene, 1 -octadecene, 1 -eicosene, 1 - docosene, 1 -tetracosene and 1-hexacosene, C22-oolefin, a mixture of C2o-C24-a-olefins and polyisobutene having on average 12 to 100 carbon atoms per molecule.
- Suitable hydrophilic monomers are monomers with sulfonate or phosphonate groups, and also nonionic monomers with hydroxyl function or alkylene oxide groups.
- men- tion may be made of: allyl alcohol, isoprenol, methoxypolyethylene glycol (meth)acrylate, meth- oxypolypropylene glycol (meth)acrylate, methoxypolybutylene glycol (meth)acrylate, methoxy- poly(propylene oxide-co-ethylene oxide) (meth)acrylate, ethoxypolyethylene glycol
- Polyalkylene glycols here may comprise 3 to 50, in particular 5 to 40 and especially 10 to 30 alkylene oxide units per molecule.
- Particularly preferred sulfonic-acid-group-containing monomers are 1 -acrylamido-
- 3-methacrylamido-2-hydroxypropanesulfonic acid allylsulfonic acid, methallylsulfonic acid, al- lyloxybenzenesulfonic acid, methallyloxybenzenesulfonic acid, 2-hydroxy- 3-(2-propenyloxy)propanesulfonic acid, 2-methyl-2-propene-1 -sulfonic acid, styrenesulfonic ac- id, vinylsulfonic acid, 3-sulfopropyl acrylate, 2-sulfoethyl methacrylate, 3-sulfopropyl methacry- late, sulfomethacrylamide, sulfomethylmethacrylamide, and salts of said acids, such as sodium, potassium or ammonium salts thereof.
- Particularly preferred phosphonate-group-containing monomers are vinylphosphonic acid and its salts.
- a further example of builders is carboxymethyl inulin.
- amphoteric polymers can also be used as builders.
- Detergent compositions comprised in said additional compartment(s) may comprise, for exam- pie, in the range from in total 10 to 70% by weight, preferably up to 50% by weight, of builder.
- builder In the context of the present invention, neither MGDA nor GLDA are counted as builder.
- such detergent compositions comprised in said additional compartment(s) may comprise one or more co-builders.
- Detergent compositions comprised in said additional compartment(s) may comprise one or more antifoams, selected for example from silicone oils and paraffin oils.
- detergent compositions comprised in inventive containers compositions comprise in total in the range from 0.05 to 0.5% by weight of antifoam.
- detergent compositions comprised in said addi- tional compartment(s) may comprise, for example, up to 5% by weight of enzyme, preference being given to 0.1 to 3% by weight.
- Said enzyme may be stabilized, for example with the sodi- um salt of at least one Ci-C3-carboxylic acid or C4-Cio-dicarboxylic acid. Preferred are formates, acetates, adipates, and succinates.
- detergent compositions comprised in said addi- tional compartment(s) comprise at least one zinc salt.
- Zinc salts can be selected from water- soluble and water-insoluble zinc salts.
- water-insoluble is used to refer to those zinc salts which, in distilled water at 25°C, have a solubility of 0.1 g/l or less.
- Zinc salts which have a higher solubility in water are accordingly referred to within the context of the present invention as water-soluble zinc salts.
- zinc salt is selected from zinc benzoate, zinc glu conate, zinc lactate, zinc formate, ZnCh, ZnS0 4 , zinc acetate, zinc citrate, Zn(NOs)2,
- Zn(CH3S03)2 and zinc gallate preferably ZnCh, ZnS0 4 , zinc acetate, zinc citrate, Zn(NOs)2, Zn(CH3S03)2 and zinc gallate.
- zinc salt is selected from ZnO, ZnO-aq,
- zinc salt is selected from zinc oxides with an aver- age particle diameter (weight-average) in the range from 10 nm to 100 pm.
- the cation in zinc salt can be present in complexed form, for example complexed with ammonia ligands or water ligands, and in particular be present in hydrated form.
- ligands are generally omitted if they are water lig- ands.
- detergent compositions comprised in said addi- tional compartment(s) may comprise additional enzyme (A). In other embodiments, no enzyme (A) is comprised in said additional compartment(s).
- detergent compositions comprised in said addi- tional compartment(s) may comprise complexing agent (B).
- B complexing agent
- neither MGDA nor GLDA is comprised in said additional compartment(s).
- Detergent compositions comprised in inventive units fulfil numerous functions. They exhibit good cleaning properties in automatic dishwashing applications. They show a good storage and shelf-life behavior and a low tendency to colorization and especially yellowing.
- Dishwashing and laundry cleaning may refer to home care or to industrial and institutional applications, home care applications being preferred. Particularly preferred is automatic dishwash in home care applications.
- Another aspect of the present invention is directed towards a process for making inventive con- tainers comprising a single unit dose, said process also being referred to as inventive process.
- Another aspect of the present invention is directed towards a process for making a compart- ment of a container according to the present invention, hereinafter also being referred to as in- ventive process.
- the inventive process comprises several steps, hereinafter also being referred to as steps (a) to (e), and said steps briefly being summarized as follows:
- step (d) placing said aqueous medium containing enzyme (A) and complexing agent(s) (B) ac- cording to step (c) into the formed recess according to step (b),
- the container is a pouch made from a polymer film.
- the polymer is polyvinyl alcohol. Steps (a) to (e) are described hereinafter in more detail.
- Step (a) refers to providing a polymer, preferably a polymer film and even more preferably a film from polyvinyl alcohol.
- such polymer may have a different thickness compared to films, preferably a greater thickness.
- step (b) - shaping the polymer - may be performed, for example, by injection molding.
- polymer films and preferably polyvinyl alco- hol films used for making pouches for inventive compositions have a thickness (strength) in the range of from 10 to 100 pm, preferably 20 to 90 pm, even more preferably 25 to 40 pm. If the strength of polymer films and especially of polyvinyl alcohol films exceeds 100 pm it takes too long to dissolve them during the washing cycle. If the strength of polymer films and especially of polyvinyl alcohol films is below 10 pm they are too sensitive to mechanical stress.
- the polymer - preferably, the polymer film - is shaped in a way that it has at least one recess so it can contain a liquid.
- thermoforming processes especially at a temperature of 5 to 20°C below the melting point of the respective polymer.
- the shaping may be performed through shaping in shaping a hose and cutting the hose into shorter pieces and closing one side each, thereby shaping sachets.
- step (b) is performed with the aid of a forming die having at least one cavity, preferably with a plurality of cavities.
- Such cavities may have ap- ertures (holes) through which reduced pressure (“vacuum”) may be applied.
- a polymer film is placed of the die. The polymer is then heated through a heating device. The polymer filmed is simultaneously shaped by the application of a vacuum for exam- pie through apertures of the cavity/the cavities.
- step (c) enzyme (A) and complexing agent(s) (B) dissolved in an aqueous medium is provid- ed.
- Methods to make such solutions of complexing agent(s) (B) are know from the art.
- step (d) of the inventive process aqueous medium containing complexing agent(s) (B) is then placed into the recesses obtained in step (b).
- Step (d) can be performed by applying pressure or simply using gravity. Applying pressure is preferred. In embodiments wherein a die with a plurality of cavities has been used, it is preferred to simultaneously place aqueous medium con- taining complexing agent(s) (B) into more than one recess.
- the recesses are filled completely. In other embodiments, the recesses are only filled partially, for example 50 to 90 % by volume, the latter embodiment being preferred in order to prevent spilling of aqueous medium in step (e) to follow.
- step (e) of the inventive process the filled but still open containers are closed. It is preferred to perform such closing step by sealing, for example heat-sealing.
- Other embodiments refer to gluing a closing device on the open container, for example a polymer film, preferably a film made from water-soluble polymer.
- sachets of polymer film may be closed by simply applying heat to the upper rim of the sachets, for example through a heated metal device.
- containers made from polymer film may be closed by performing a chemical reaction of a sealing substance. Said chemical reaction may be promoted by applying a vacu- um.
- steps (b), (d) and (e) are performed as a vertical form-fill-seal route yield ing envelope-shaped pouches that contain aqueous medium containing complexing agent(s)
- the present invention is further illustrated by working examples.
- Percentages refer to % by weight unless expressly specified otherwise.
- the contents of MGDA- Na3 and of GLDA-Na 4 were determined by measuring the iron binding capacity (FeCl3, potenti- ometric detection).
- AS.2 From a 100 g solution of (B1.1 ) 3.2 g water were removed. To the solution were then added 2.2 g formic acid (100%) and 1 g protease. The final pH of the solution is 9.8. The activity of the enzyme in the chelating agent solution was measured over time using a Gallery Plus (Thermo Scientific).
- the solutions were stored at 37°C.
- the activity of the enzyme was measured over time over a period of 16 weeks.
- Polyvinylalcohol films with a degree of saponification: 88 mole-%, thickness 38 pm, commercial- ly available from MonoSol were used.
- a polyvinylalcohol film was cut into rectangular pieces of 12-5 cm each. Envelope-like pouches were made by folding the rectangular pieces accordingly. The brinks were connected by heat- sealing. With a pipette, 10 ml of 53% by weight solution of protease and complexing agents (B) as obtained in example I. were placed into the envelope-like pouch. The pouch was closed again by heat-sealing. After 4 weeks of storage at 23°C, neither deterioration of the pouch with AS.2 or AS.3 nor disadvantageous change of the contents could be observed.
- Example detergent compositions according to Table 2 are prepared by mixing the respective ingredients in dry state.
- Tablets may be formed of any of the above mixture, weight: 18 g, and one pouch from (II) may be placed on each tablet.
- the tablets are packed - together with the pouch - into a film of poly- vinyl alcohol, degree of saponification: 88 mole-%, thickness: 35 pm. They may be used as unit doses in an automatic dishwasher and yield excellent dishwashing results. Their use is conven- ient.
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Wood Science & Technology (AREA)
- Organic Chemistry (AREA)
- Detergent Compositions (AREA)
Abstract
Packaging unit containing a detergent single unit dose wherein said packaging unit comprises a compartment 1 comprising an aqueous solution of enzyme (A) and at least one chelating agent (B) dissolved in an aqueous medium, said chelating agent (B) being (B1) at least one alkali metal salt of glutamic acid diacetic acid (GLDA), and (B2) at least one alkali metal salt of methyl glycine diacetic acid (MGDA), and one more compartment comprising at least one ingredient of a detergent other than en- zyme (A) and chelating agent(s) (B), wherein the overall concentration of chelating agent(s) (B) in compartment 1 is in the range of from 35 to 75 % by weight.
Description
Packaging unit comprising a detergent composition containing an enzyme and at least one che- lating agent
The present invention is directed towards a packaging unit containing a detergent single unit dose wherein said packaging unit comprises a compartment 1 comprising an aqueous solution of enzyme (A) and at least one chelating agent (B) dissolved in an aqueous medium, said chelating agent (B) being
(B1 ) at least one alkali metal salt of glutamic acid diacetic acid (GLDA), and
(B2) at least one alkali metal salt of methyl glycine diacetic acid (MGDA), and one more compartment comprising at least one ingredient of a detergent other than en- zyme (A) and chelating agent(s) (B), wherein the overall concentration of chelating agent(s) (B) in compartment 1 is in the range of from 35 to 75 % by weight.
Complexing agents such as methyl glycine diacetic acid (MGDA) and their respective alkali metal salts are useful and environmentally friendly sequestrants for alkaline earth metal ions such as Ca2+ and Mg2+. They can replace phosphate-type sequestrants such as sodium tripoly- phosphate (“STPP”), the latter being replaced now in many countries for environmental rea- sons. Therefore, MGDA and related compounds are recommended and used for various pur- poses such as laundry detergents and for automatic dishwashing (ADW) formulations, in partic- ular for so-called phosphate-free laundry detergents and phosphate-free ADW formulations.
For automatic dishwashing and laundry care, so-called single unit doses are of increased com- mercial importance. They are of great convenience for the end-user because such unit doses contain the right amounts of the ingredients for the washing and rinsing steps and because they can be easily placed into the automatic dishwasher or washing machine by the end-user, see, e.g., WO 2002/042400 and WO 201 1/072017. Examples of unit doses are tablets and pellets and in particular pouches. Pouches in the form of multi-compartment pouches have been dis- closed as well, see WO 2009/1 12994.
It can be observed, though, that many laundry detergents lose their efficacy concerning the re- moval of organic materials such as milk, blood, and especially of egg residues after some time of storage. Especially liquid laundry detergent compositions, exhibit only minor activity after a few weeks of storage at 30°C or even higher temperatures, for example 35 or 37°C. Such tem- peratures are not only quite common in Southern European countries, Southern American countries and South-east Asia but also in laundering facilities.
Dishwashing compositions have to fulfil many requirements. Thus, they have to thoroughly clean the crockery, they should not put any harmful or potentially harmful substances into the waste water, they should allow the draining and drying of water from the crockery, and they should not cause any problems in the operation of the dishwasher. Finally, they should not cause any undesired esthetic effects on the item to be cleaned.
Without wishing to be bound to any theory it is believed that strong complexing agents may ex- tract the central Ca2+ metal ion(s) of the active site(s) of detergent proteases and amylases, thus, reduce the activity of said enzymes.
It was therefore an objective of the present invention to provide a detergent composition that is environmentally friendly and also exhibits good performance with respect to the removal of or- ganic materials such as milk, blood, and egg residues, even after a few weeks and more of storage at 30°C or even higher temperatures, for example 35 or 37°C. Such detergent composi- tions can preferably be used in laundry or automatic dishwashing. It was also an objective to provide a process for manufacturing a detergent composition that is environmentally friendly and also exhibits good activity with respect to the removal of organic materials such as milk, blood, and egg residues. It was further an objective of the present invention to provide a method of use, and a use, of inventive detergent compositions.
Accordingly, the packaging units as defined at the outset have been found, hereinafter also re- ferred to as inventive units or units according to the (present) invention.
In the context of the present invention, packaging units comprise more than one compartments. Such compartments are separated from each other, for example by a polymer layer that may be mechanically flexible or stiff. The distinction between mechanically flexible and mechanically stiff may be made by manual determination of the degree of deformability by an average end user with two fingers. If such an average end user can deform the shape of said unit by at least 5% into one dimension the respective unit is deemed mechanically flexible, otherwise it is deemed stiff.
Inventive units comprise more than one compartment. In the context of the present invention, one of such compartments will be named“compartment 1” to distinguish it from the other com- partment(s). Such compartments may have the same or different appearance, and they may have the same or different sizes. Such compartments are separated from each other, for exam- pie by a polymer film.
In specific embodiments, inventive units are tablets that have at least one cavity per tablet. Per cavity there is at least one pouch, preferably there is at least one pouch placed into the cavity and attached to the tablet. In special embodiment, the volume of the pouch including the solu- tion of enzyme (A) and complexing agent(s) (B) corresponds to the volume of the cavity, for example they may have the same volume ±10%, preferably ±5%. The better the shape and the
size of cavity on one side and the shape of the pouch including the solution of enzyme (A) and complexing agent(s) (B) on the other side correspond to each other the less breakage during transport can be observed. Such tablets may be packaged in a film of, e.g., polyvinyl alcohol. The tablet comprises components of the respective detergent composition such as surfactants, builder(s), enzymes, and/or bleaching agent.
In another specific embodiment, inventive units are shaped as a box that has at least one cavity per box. Per cavity there is at least one pouch, preferably there is at least one pouch placed into the cavity and attached to the box. In special embodiment, the volume of the pouch including the solution of enzyme (A) and complexing agent(s) (B) corresponds to the volume of the cavity, for example they may have the same volume ±10%, preferably ±5%. The better the shape and the size of cavity on one side and the shape of the box including the solution of enzyme (A) and complexing agent(s) (B) on the other side correspond to each other the less breakage during transport can be observed. The box comprises components of the respective detergent compo- sition such as surfactants, builder(s), enzymes, and/or bleaching agent.
In another specific embodiment, inventive units are pouches that encompass at least two com- partments, for example two, three or four compartments. One of the compartments contains the solution of enzyme (A) and complexing agent(s) (B). The other components of the respective detergent compositions are in the one or more other compartment(s).
In one embodiment of the present invention, all complexing agent(s) (B) that are comprised in inventive units is in the very compartment as defined above. In another embodiment of the pre- sent invention, a share of complexing agent(s) (B) is comprised in one compartment in dis- solved form, as stated above, and more of complexing agent(s) (B) is comprised in the other compartment or one other compartment, as applicable, of the inventive unit.
Solid detergent compositions may contain residual moisture. Residual moisture refers to water other than the water that is part of the aqueous medium in which complexing agent(s) (B) are dissolved in. In one embodiment of the present invention, the residual moisture content is in the range of from 0.1 to 10 % by weight, referring to the total respective detergent composition. The residual moisture content can be determined, e. g., by Karl-Fischer-Titration or by measuring the weight loss upon drying.
In the context with the present invention, the term“pouch” refers to a unit made from a flexible film. Said unit is essentially closed upon storage of the respective dishwashing detergent corn- position. During the dishwashing process the contents of the pouch are removed from the pouch itself, preferably by dissolving the pouch.
Units and especially pouches in the context of the present invention may have various shapes. For examples, units may be in the form of a ball, an ellipsoid, a cube, a cuboid, or they may be of geometrically irregular shape. In special examples, pouches may have the shape of an enve-
lope, of a pillow, of a flexible sleeve or flexible tube that is closed at both ends, of a ball or a cube.
In one embodiment of the present invention, units according to the present invention and espe- cially pouches have a diameter in the range of from 0.5 to 7 cm.
In one embodiment of the present invention, units according to the present invention and espe- cially pouches have a volume - in the closed state - in the range of from 15 to 70 ml, preferably 18 ml to 50 ml and in particular 20 to 30 ml. Such inventive units are particularly useful for au- tomatic dishwash in home care application. Inventive units particularly useful for fabric care in home care applications may have a volume in the range of from 15 to 40 ml, preferably 25 to 30 ml.
In one embodiment of the present invention, each compartment has a content in the range of from 0.5 to 50 ml, preferably 5 to 25 ml. In embodiments wherein inventive units encompass two or more compartments, such compartments may have equal size or different size. Preferably, in embodiments wherein inventive units encompass two or more compartments, such units en- compass one major compartment and one or two or three smaller compartments.
Inventive units are made from a polymer, preferably from a water-soluble polymer. Pouches in the context of the present invention are made from a polymer film.
Said polymer may be selected from natural polymers, modified natural polymers, and synthetic polymers. Examples of suitable natural polymers are alginates, especially sodium alginate, fur- thermore xanthum, carragum, dextrin, maltodextrin, gelatine, starch, and pectin. Examples of suitable modified natural polymers are methylcellulose, ethylcellulose, carboxymethyl cellulose, hydroxypropylcellulose, hydroxypropyl methyl cellulose (HPMC), and hydroxymethyl cellulose. Examples of suitable synthetic polymers are polyvinyl pyrrolidone, polyacrylamide, polyalkylene glycols, preferably polypropylene glycol and polyethylene glycol, especially polyethylene glycol with a molecular weight Mw in the range of at least 2,000 g/mol, preferably of from 3,000 to 100,000 g/mol, and in particular polyvinyl alcohol.
The term“polyvinyl alcohol” as used herein does not only include homopolymers of polyvinyl alcohol that can be made by free-radical polymerization of vinyl acetate followed by subsequent hydrolysis (saponification) of all or the vast majority of the ester groups. Polyvinyl alcohol also includes copolymers obtainable by free-radical copolymerization of vinyl acetate and at least one comonomer selected from maleic acid, maleic anhydride, itaconic anhydride, methyl (meth)acrylate and 2-acrylamido-2-methyl propanesulfonic acid (“AMPS”).
In a preferred embodiment of the present invention, polyvinyl alcohol as used for making units and especially for pouches may have an average degree of polymerization (weight average) in the range of from 500 to 3,000 g/mol. The molecular weight Mw of such polyvinyl alcohol is pref-
erably, in the range of from 6,000 to 250,000 g/mol, preferably up to 75,000 g/mol. The molecu- lar weight is preferably determined by gel permeation chromatography of the respective polyvi- nyl acetate or respective copolymer before saponification.
Preferably, polyvinyl alcohol used for making units and especially pouches is atactic as deter- mined by 1H NMR spectroscopy.
Polyvinyl alcohols used for making units - especially pouches - essentially have repeating units of (CH2-CHOH). The hydroxyl groups in polyvinyl alcohol are mostly in 1 ,3-position, thus form- ing structural units of the type -CH2-CH(OH)-CH2-CH(OH)-. In minor amounts (1 to 2 mole-%) there are geminal hydroxyl groups, thus forming structural units of -CH2-CH(OH)-CH(OH)-CH2-.
One or more modified polyvinyl alcohols may be employed as polymers instead of polyvinyl al- cohol or in combination with polyethylene glycol or with polyvinyl alcohol. Examples are graft copolymers such as polyalkylene glycol grafted with polyvinyl acetate followed by subsequent hydrolysis/saponification of the ester groups.
Polymer may be used without or with one or more additives. Suitable additives are especially plasticizers such as C4-Cio-dicarboxylic acids, for example adipic acid, and glycols such as eth- ylene glycol and diethylene glycol.
Due to their production, commercially available polyvinyl alcohols usually have residual non- saponified ester groups, especially acetate groups. Polyvinyl alcohols used for making units and especially pouches for embodiments of the present invention essentially have a degree of sa- ponification in the range of from 87 to 89 mole-%. The degree of saponification can be deter- mined in accordance with the determination of the ester value, for example according to DIN EN ISO 3681 (2007-10).
In one embodiment of the present invention, polyvinyl alcohols used for making units and espe- cially for making pouches for embodiments of the present invention have a glass transition tem- perature in the range of from 55 to 60°C, preferably 58°C, determinable according to, e.g., DIN 53765: 1994-03, or ISO 1 1357-2: 1999-03.
In one embodiment of the present invention, polyvinyl alcohols used for making inventive units and especially for making pouches for embodiments of the present invention have a melting point in the range of from 185 to 187°C.
In one embodiment of the present invention, polyvinyl alcohols used for making for embodi- ments of the present invention and especially for making pouches comprising a single unit dose are partially acetalized or ketalized with sugars such as, glucose, fructose, or with starch. In another embodiment of the present invention polyvinyl alcohols used for making units and es- pecially pouches are partially esterified with, e. g., maleic acid or itaconic acid.
In one embodiment of the present invention, polyvinyl alcohol films may contain a plasticizer. Plasticizers may be used for reducing the stiffness of such polyvinyl alcohol films. Suitable compounds usable as plasticizers for polyvinyl alcohol are ethylene glycol, diethylene glycol, triethylene glycol, polyethylene glycol, for example with an average molecular weight Mw up to 400 g/mol, glycerol, trimethylol propane, triethanolamine, and neo-pentyl glycol. Up to 25 % by weight of the respective polyvinyl alcohol may be plasticizer.
In one embodiment of the present invention, said pouches are made from a polymer film, said polymer being water-soluble at a temperature of at least 40°C, for example in the range of from 40 to 95°C, but insoluble in water at a temperature in the range of from 5 to 30°C. In other em- bodiments, said pouches are made from polymer films that are soluble in water even at 1 °C. In the context of the present invention, the terms water-soluble and soluble in water are used in- terchangeably. They both refer to polymers that dissolve in water at 20°C. However, such poly- mers dissolve much slower or not detectably at all in the aqueous medium containing complex- ing agent(s) (B). A polymer is deemed water-soluble if the percentage of solubility is at least 90%. A suitable method of determination of the percentage is disclosed below.
Examples of polymer films that are soluble at 1 °C or more and of polymer films that are soluble at 40°C are polyvinyl alcohol films available from Syntana E. Harke GmbH & Co under the trademark of Solublon ®.
In one embodiment of the present invention, polymer films and preferably polyvinyl alcohol films used for making pouches that can be used in the present invention have a thickness (strength) in the range of from 10 to 100 pm, preferably 20 to 90 pm, even more preferably 25 to 35 pm. If the strength of polymer films and especially of polyvinyl alcohol films exceeds 100 pm it takes too long to dissolve them during the washing cycle. If the strength of polymer films and especial- ly of polyvinyl alcohol films is below 10 pm they are too sensitive to mechanical stress.
The aqueous solution in compartment 1 also contains enzyme (A), hereinafter also referred to as enzyme (A), for example hydrolase (A). Preferred enzymes (A) are selected from proteases, amylases, and lipases, hereinafter also referred to as proteases (A), amylases (A), or lipases (A), respectively.
It has been found that enzymes (A) in inventive compositions exhibit a particularly high life-time. Enzymes (A) that are particularly beneficial in inventive compositions are selected from hydro- lases (EC 3). Preferred enzymes (A) are selected from the group of enzymes acting on ester bond (E.C. 3.1 ), glycosylases (E.C. 3.2), and peptidases (E.C. 3.4). Enzymes acting on ester bond (E.C. 3.1 ), hereinafter also refer to lipases (component (b)), respectively. Glycosylases (E.C. 3.2) hereinafter also refer to either amylases (A) and cellulases (A). Peptidases hereinaf- ter also refer to proteases (A).
Hydrolases (A) in the context of the present invention are identified by polypeptide sequences, also called amino acid sequences herein. The polypeptide sequence specifies the three- dimensional structure including the“active site” of an enzyme which in turn determines the cata- lytic activity of the same. Polypeptide sequences may be identified by a SEQ ID NO. According to the World Intellectual Property Office (WIPO) Standard ST.25 (1998) the amino acids herein are represented using three-letter code with the first letter as a capital or the corresponding one letter.
Enzymes (A) in the context of the present invention may relate to parent enzymes and/or variant enzymes, both having enzymatic activity. Enzymes having enzymatic activity are enzymatically active or exert enzymatic conversion, meaning that enzymes act on substrates and convert these into products. The term“enzyme” herein excludes inactive variants of an enzyme.
A“parent” sequence of a parent protein or enzyme, also called“parent enzyme” is the starting sequence for introduction of changes, e.g., by introducing one or more amino acid substitutions, insertions, deletions, or a combination thereof to the sequence, resulting in“variants” of the par- ent sequences. The term parent enzyme (or parent sequence) includes wild-type enzymes (se- quences) and synthetically generated sequences (enzymes) which are used as starting se- quences for introduction of (further) changes.
The term“enzyme variant” or“sequence variant” or“variant enzyme” refers to an enzyme that differs from its parent enzyme in its amino acid sequence to a certain extent. If not indicated otherwise, variant enzyme“having enzymatic activity” means that this variant enzyme has the same type of enzymatic activity as the respective parent enzyme.
In describing the variants of the present invention, the nomenclature described as follows is used:
Amino acid substitutions are described by providing the original amino acid of the parent en- zyme followed by the number of the position within the amino acid sequence, followed by the substituted amino acid.
Amino acid deletions are described by providing the original amino acid of the parent enzyme followed by the number of the position within the amino acid sequence, followed by *.
Amino acid insertions are described by providing the original amino acid of the parent enzyme followed by the number of the position within the amino acid sequence, followed by the original amino acid and the additional amino acid. For example, an insertion at position 180 of lysine next to glycine is designated as“Gly180Glyl_ys” or“G180GK”.
In cases where a substitution and an insertion occur at the same position, this may be indicated as S99SD+S99A or in short S99AD. In cases where an amino acid residue identical to the exist- ing amino acid residue is inserted, it is clear that degeneracy in the nomenclature arises. If for
example a glycine is inserted after the glycine in the above example this would be indicated by G180GG.
Where different alterations can be introduced at a position, the different alterations are separat- ed by a comma, e.g.“Arg170Tyr, Glu” represents a substitution of arginine at position 170 with tyrosine or glutamic acid. Alternatively different alterations or optional substitutions may be indi- cated in brackets e.g. Arg170[Tyr, Gly] or Arg170{Tyr, Gly}; or in short R170 [Y,G] or R170 {Y, G}; or in long R170Y, R170G.
Enzyme variants may be defined by their sequence identity when compared to a parent en- zyme. Sequence identity usually is provided as“% sequence identity” or“% identity”. For calcu- lation of sequence identities, in a first step a sequence alignment has to be produced. According to this invention, a pairwise global alignment has to be produced, meaning that two sequences have to be aligned over their complete length, which is usually produced by using a mathemati- cal approach, called alignment algorithm.
In the context of the present invention, the alignment is generated by using the algorithm of Needleman and Wunsch (J. Mol. Biol. 1979 48 p. 443-453). Preferably, the program“NEEDLE” (The European Molecular Biology Open Software Suite (EMBOSS)) is used for the purposes of the current invention, with using the programs default parameter (gap open=10.0, gap ex- tend=0.5 and matrix=EBLOSUM62). According to this invention, the following calculation of %- identity applies: %-identity = (identical residues / length of the alignment region which is show- ing the respective sequence of this invention over its complete length)-100.
In the context of the present invention, enzyme variants may be described as an amino acid sequence which is at least n% identical to the amino acid sequence of the respective parent enzyme with“n” being an integer between 10 and 100. In one embodiment, variant enzymes are at least 70%, at least 75%, at least 80%, at least 81 %, at least 82%, at least 83%, at least 84%, at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical when compared to the full length amino acid sequence of the parent enzyme, wherein the enzyme variant has enzymatic activity.
Enzyme variants may be defined by their sequence similarity when compared to a parent en- zyme. Sequence similarity usually is provided as“% sequence similarity” or“%-similarity”. % sequence similarity takes into account that defined sets of amino acids share similar properties, e.g by their size, by their hydrophobicity, by their charge, or by other characteristics. Herein, the exchange of one amino acid with a similar amino acid may be called“conservative mutation”.
For determination of %-similarity according to this invention the following applies: amino acid A is similar to amino acids S; amino acid D is similar to amino acids E and N; amino acid E is simi- lar to amino acids D and K and Q; amino acid F is similar to amino acids W and Y; amino acid H is similar to amino acids N and Y; amino acid I is similar to amino acids L and M and V; amino
acid K is similar to amino acids E and Q and R; amino acid L is similar to amino acids I and M and V; amino acid M is similar to amino acids I and L and V; amino acid N is similar to amino acids D and H and S; amino acid Q is similar to amino acids E and K and R; amino acid R is similar to amino acids K and Q; amino acid S is similar to amino acids A and N and T; amino acid T is similar to amino acids S; amino acid V is similar to amino acids I and L and M; amino acid W is similar to amino acids F and Y; amino acid Y is similar to amino acids F and H and W. Conservative amino acid substitutions may occur over the full length of the sequence of a poly- peptide sequence of a functional protein such as an enzyme. In one embodiment, such muta- tions are not pertaining the functional domains of an enzyme. In one embodiment, conservative mutations are not pertaining the catalytic centers of an enzyme.
To take conservative mutations into account, a value for sequence similarity of two amino acid sequences may be calculated from the same alignment, which is used to calculate %-identity. According to this invention, the following calculation of %-similarity applies: %-similarity = [(iden- tical residues + similar residues) / length of the alignment region which is showing the respec- tive sequence(s) of this invention over its complete length]-100.
In the context of the present invention, enzyme variants may be described as an amino acid sequence which is at least m% similar to the respective parent sequences with“m” being an integer between 10 and 100. In one embodiment, variant enzymes are at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% similar when corn- pared to the full length polypeptide sequence of the parent enzyme, wherein the variant enzyme has enzymatic activity.
“Enzymatic activity” means the catalytic effect exerted by an enzyme, which usually is ex- pressed as units per milligram of enzyme (specific activity) which relates to molecules of sub- strate transformed per minute per molecule of enzyme (molecular activity).
Variant enzymes may have enzymatic activity according to the present invention when said en- zyme variants exhibit at least 20%, at least 25%, at least 30%, at least 35%, at least 40%, at least 45%, at 10 least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or 100% of the enzymatic activity of the respective parent enzyme.
In one embodiment, inventive compositions comprise at least one protease (A). Enzymes (A) having proteolytic activity are called“proteases” or peptidases in the context of the present in- vention. Such enzymes are members of class EC 3.4.
Proteases (A) are further classified as aminopeptidases (EC 3.4.11 ), dipeptidases (EC 3.4.13), dipeptidyl-peptidases and tripeptidyl-peptidases (EC 3.4.14), peptidyl-dipeptidases (EC 3.4.15), serine-type carboxypeptidases (EC 3.4.16), metallocarboxypeptidases (EC 3.4.17), cysteine-
type carboxypeptidases (EC 3.4.18), omega peptidases (EC 3.4.19), serine endopeptidases (EC 3.4.21 ), cysteine endopeptidases (EC 3.4.22), aspartic endopeptidases (EC 3.4.23), metal- lo-endopeptidases (EC 3.4.24), threonine endopeptidases (EC 3.4.25), or endopeptidases of unknown catalytic mechanism (EC 3.4.99).
Protease (A) may be an endopeptidase of any kind or a mixture of endopeptidases of any kind. In one embodiment, protease according to the invention is selected from serine protease (EC 3.4.21 ).
Serine proteases or serine peptidases are characterized by having a serine in the catalytically active site, which forms a covalent adduct with the substrate during the catalytic reaction. A ser- ine protease in the context of the present invention is selected from the group consisting of chymotrypsin (e.g., EC 3.4.21.1 ), elastase (e.g., EC 3.4.21.36), elastase (e.g., EC 3.4.21.37 or EC 3.4.21.71 ), granzyme (e.g., EC 3.4.21.78 or EC 3.4.21.79), kallikrein (e.g., EC 3.4.21.34, EC 3.4.21.35, EC 3.4.21.118, or EC 3.4.21.119,) plasmin (e.g., EC 3.4.21.7), trypsin (e.g., EC 3.4.21.4), thrombin (e.g., EC 3.4.21.5), and subtilisin. Subtilisin is also known as subtilopepti- dase, e.g., EC 3.4.21.62, the latter hereinafter also being referred to as“subtilisin”.
Crystallographic structures of proteases (A) show that the active site is commonly located in a groove on the surface of the molecule between adjacent structural domains, and the substrate specificity is governed by the properties of binding sites arranged along the groove on one or both sides of the catalytic site that is responsible for hydrolysis of the scissile bond. Accordingly, the specificity of protease (A) can be described by use of a conceptual model in which each specificity subsite is able to accommodate the sidechain of a single amino acid residue. The sites are numbered from the catalytic site, S1 , S2...Sn towards the N-terminus of the substrate, and S1 ', S2'...Sn' towards the C-terminus. The residues they accommodate are numbered P1 , P2...Pn, and PT, P2'...Pn', respectively:
In this representation the catalytic site of the enzyme is marked“*” and the peptide bond cleaved (the scissile bond) is indicated by the symbol“+”.
In general, the three main types of protease activity (proteolytic activity) are: trypsin-like, where there is cleavage of amide substrates following Arg (N) or Lys (K) at P1 , chymotrypsin-like where cleavage occurs following one of the hydrophobic amino acids at P1 , and elastase-like with cleavage following an Ala (A) at P1.
A sub-group of the serine proteases tentatively designated as subtilases has been proposed by Siezen et al. (1991 ), Protein Eng. 4:719-737 and Siezen et al. (1997), Protein Science 6:501 - 523. They are defined by homology analysis of more than 170 amino acid sequences of serine proteases previously referred to as subtilisin-like proteases. A subtilisin was previously often
defined as a serine protease produced by Gram-positive bacteria or fungi, and according to Siezen et al. now is a subgroup of the subtilases. A wide variety of subtilases have been identi fied, and the amino acid sequence of a number of subtilases has been determined. For a more detailed description of such subtilases and their amino acid sequences reference is made to Siezen et al. (1997), Protein Science 6:501-523. Subtilases may be divided into 6 sub-divisions, i.e. the subtilisin family, the thermitase family, the proteinase K family, the lantibiotic peptidase family, the kexin family and the pyrolysin family.
A subgroup of the subtilases are the subtilisins which are serine proteases from the family S8 as defined by the MEROPS database (http://merops.sanger.ac.uk). Peptidase family S8 con- tains the serine endopeptidase subtilisin and its homologues. In subfamily S8A, the active site residues frequently occur in the motifs Asp-Thr/Ser-Gly similarly to the sequence motif in fami- lies of aspartic endopeptidases in clan AA, His-Gly-Thr-His and Gly-Thr-Ser-Met-Ala-Xaa-Pro.
Prominent members of family S8, subfamily A are:
The subtilisin related class of serine proteases shares a common amino acid sequence defining a catalytic triad which distinguishes them from the chymotrypsin related class of serine proteas- es. Subtilisins and chymotrypsin related serine proteases both have a catalytic triad comprising aspartate, histidine and serine.
In subtilisin related proteases (A) the relative order of these amino acids, reading from the ami- no to carboxy-terminus is aspartate-histidine-serine. In the chymotrypsin related proteases the relative order, however is histidine-aspartate-serine. Thus, subtilisin herein refers to a serine protease having the catalytic triad of subtilisin related proteases. Examples include the subtil isins as described in WO 89/06276 and EP 0283075, WO 89/06279, WO 89/09830, WO 89/09819, WO 91/06637 and WO 91/02792.
Parent proteases of the subtilisin type (EC 3.4.21.62) and variants may be bacterial proteases. Said bacterial protease may be a Gram-positive bacterial polypeptide such as a Bacillus, Clos tridium, Enterococcus, Geobacillus, Lactobacillus, Lactococcus, Oceanobacillus, Staphylococ cus, Streptococcus, or Streptomyces protease, or a Gram-negative bacterial polypeptide such
as a Campylobacter, E. cdi, Flavobacterium, Fusobacterium, Helicobacter, Hyobacter, Neis seria, Pseudomonas, Salmonella, or Ureaplasma protease. They act as unspecific endopepti- dases, i.e. they hydrolyze any peptide bonds.
Commercially available protease enzymes include those sold under the trade names Alcalase®, Blaze®, Duralase™, Durazym™, Relase®, Relase® Ultra, Savinase®, Savinase® Ultra, Pri- mase®, Polarzyme®, Kannase®, Liquanase®, Liquanase® Ultra, Ovozyme®, Coronase®, Co- ronase® Ultra, Neutrase®, Everlase® and Esperase® (Novozymes A/S), those sold under the tradename Maxatase®, Maxacal®, Maxapem®, Purafect®, Purafect® Prime, Purafect MA®, Purafect Ox®, Purafect OxP®, Puramax®, Properase®, FN2®, FN3®, FN4®, Excellase®, Eraser®, Ultimase®, Opticlean®, Effectenz®, Preferenz® and Optimase® (Danisco/DuPont), Axapem™ (Gist-Brocases N.V.), Bacillus lentus Alkaline Protease (BLAP; sequence shown in Figure 29 of US 5,352,604) and variants thereof and KAP ( Bacillus alkalophilus subi\\\s\n) from Kao.
In one aspect of the invention, the parent enzymes and variants may be a Bacillus alcalophilus, Bacillus amyloliquefaciens, Bacillus brevis, Bacillus circulans, Bacillus dausii, Bacillus coagu- lans, Bacillus firmus, Bacillus gibsonii, Bacillus lautus, Bacillus lentus, Bacillus Hcheniformis, Bacillus megaterium, Bacillus pumiius, Bacillus sphaericus, Bacillus stearothermophiius, Bacil lus subti/is, or Bacillus thuringiensis protease.
In one embodiment of the present invention, the subtilase is selected from the following:
- subtilisin from Bacillus amyloliquefaciens BPN' (described by Vasantha et al. (1984) J. Bacteriol. Volume 159, p. 811 -819 and JA Wells et al. (1983) in Nucleic Acids Research, Volume 11 , p. 7911-7925),
- subtilisin from Bacillus Hcheniformis (subtilisin Carlsberg; disclosed in EL Smith et al.
(1968) in J. Biol Chem, Volume 243, pp. 2184-2191 , and Jacobs et al. (1985) in Nucl. Ac- ids Res, Vol 13, p. 8913-8926),
- subtilisin PB92 (original sequence of the alkaline protease PB92 is described in EP
283075 A2),
- subtilisin 147 and/or 309 (Esperase®, Savinase®) as disclosed in GB 1243784,
- subtilisin from Bacillus lentus as disclosed in WO 91/02792, such as from Bacillus lentus DSM 5483 or the variants of Bacillus lentus DSM 5483 as described in WO 95/23221 ,
- subtilisin from Bacillus alcalophilus (DSM 11233) disclosed in DE 10064983,
- subtilisin from Bacillus gibsonii (DSM 14391 ) as disclosed in WO 2003/054184,
- subtilisin from Bacillus sp. (DSM 14390) disclosed in WO 2003/056017,
- subtilisin from Bacillus sp. (DSM 14392) disclosed in WO 2003/055974,
- subtilisin from Bacillus gibsonii( DSM 14393) disclosed in WO 2003/054184,
- subtilisin having SEQ ID NO: 4 as described in WO 2005/063974 or a subtilisin which is at least 40% identical thereto and having proteolytic activity,
- subtilisin having SEQ ID NO: 4 as described in WO 2005/103244 or subtilisin which is at least 80% identical thereto and having proteolytic activity,
- subtilisin having SEQ ID NO: 7 as described in WO 2005/103244 or subtilisin which is at least 80% identical thereto and having proteolytic activity, and
- subtilisin having SEQ ID NO: 2 as described in application DE 102005028295.4 or subtil- isin which is this at least 66% identical thereto and having proteolytic activity.
Examples of useful proteases (A) in accordance with the present invention comprise the vari- ants described in: WO 92/19729, WO 95/23221 , WO 96/34946, WO 98/201 15, WO 98/201 16, WO 99/11768, WO 01/44452, WO 02/088340, WO 03/006602, WO 2004/03186, WO
2004/041979, WO 2007/006305, WO 2011/036263, WO 201 1/036264, and WO 2011/072099. Suitable examples comprise especially protease variants of subtilisin protease derived from SEQ ID NO:22 as described in EP 1921147 (which is the sequence of mature alkaline protease from Bacillus lentus DSM 5483) with amino acid substitutions in one or more of the following positions: 3, 4, 9, 15, 24, 27, 33, 36, 57, 68, 76, 77, 87, 95, 96, 97, 98, 99, 100, 101 , 102, 103, 104, 106, 1 18, 120, 123, 128, 129, 130, 131 , 154, 160, 167, 170, 194, 195, 199, 205, 206, 217, 218, 222, 224, 232, 235, 236, 245, 248, 252 and 274 (according to the BPN' numbering), which have proteolytic activity. In one embodiment, such a subtilisin protease is not mutated at posi- tions Asp32, His64 and Ser221 (according to BPN’ numbering).
In one embodiment, subtilisin has SEQ ID NO:22 as described in EP 1921147, or a subtilisin which is at least 80% identical thereto and has proteolytic activity. In one embodiment, a subtil- isin is at least 80% identical to SEQ ID NO:22 as described in EP 1921147 and is characterized by having amino acid glutamic acid (E), or aspartic acid (D), or asparagine (N), or glutamine (Q), or alanine (A), or glycine (G), or serine (S) at position 101 (according to BPN’ numbering) and has proteolytic activity. In one embodiment, subtilisin is at least 80% identical to SEQ ID NO:22 as described in EP 1921147 and is characterized by having amino acid glutamic acid (E), or aspartic acid (D), at position 101 (according to BPN’ numbering) and has proteolytic activity. Such a subtilisin variant may comprise an amino acid substitution at position 101 , such as R101 E or R101 D, alone or in combination with one or more substitutions at positions 3, 4, 9, 15, 24, 27, 33, 36, 57, 68, 76, 77, 87, 95, 96, 97, 98, 99, 100, 101 , 102, 103, 104, 106, 118, 120, 123, 128, 129, 130, 131 , 154, 160, 167, 170, 194, 195, 199, 205, 206, 217, 218, 222, 224, 232, 235, 236, 245, 248, 252 and/or 274 (according to BPN’ numbering) and has proteolytic activity.
In another embodiment, a subtilisin is at least 80% identical to SEQ ID NO:22 as described in EP 1921147 and is characterized by comprising at least the following amino acids (according to BPN’ numbering) and has proteolytic activity:
(a) threonine at position 3 (3T)
(b) isoleucine at position 4 (4I)
(c) alanine, threonine or arginine at position 63 (63A, 63T, or 63R)
(d) aspartic acid or glutamic acid at position 156 (156D or 156E)
(e) proline at position 194 (194P)
(f) methionine at position 199 (199M)
(g) isoleucine at position 205 (205I)
(h) aspartic acid, glutamic acid or glycine at position 217 (217D, 217E or 217G),
(i) combinations of two or more amino acids according to (a) to (h).
In another embodiment, a subtilisin is at least 80% identical to SEQ ID NO:22 as described in EP 1921147 and is characterized by comprising one amino acid (according to (a)-(h)) or combi- nations according to (i) together with the amino acid 101 E, 101 D, 101 N, 101 Q, 101A, 101 G, or 101 S (according to BPN’ numbering) and has proteolytic activity.
In one embodiment, a subtilisin is at least 80% identical to SEQ ID NO:22 as described in EP 1921147 and is characterized by comprising the mutation (according to BPN’ numbering)
R101 E, or S3T + V4I + V205I, or S3T + V4I + V199M + V205I + L217D and has proteolytic ac- tivity.
In another embodiment, the subtilisin comprises an amino acid sequence having at least 80% identity to SEQ ID NO:22 as described in EP 1921147 and being further characterized by corn- prising R101 E and S3T, V4I, and V205I (according to the BPN’ numbering) and has proteolytic activity.
In another embodiment, a subtilisin comprises an amino acid sequence having at least 80% identical to SEQ ID NO:22 as described in EP 1921 147 and being further characterized by comprising R101 E, and one or more substitutions selected from the group consisting of S156D, L262E, Q137H, S3T, R45E,D,Q, P55N, T58W,Y,L, Q59D,M,N,T, G61 D,R, S87E, G97S, A98D,E,R, S106A,W, N117E, H120V,D,K,N, S125M, P129D, E136Q, S144W, S161T,
S163A,G, Y171 L, A172S, N185Q, V199M, Y209W, M222Q, N238H, V244T, N261T,D and L262N,Q,D (as described in WO 2016/096711 and according to the BPN’ numbering), and has proteolytic activity.
Percentage-identity for subtilisin variants is calculated as disclosed above. Subtilisin variant enzymes as disclosed above which are at least n% identical to the respective parent sequences include variants with n being at least 40 to 100. Depending on the %-identity values applicable as provided above, subtilisin variants in one embodiment have proteolytic activity and are at least 40%, at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical when compared to the full length polypeptide sequence of the parent enzyme.
In another embodiment, the invention relates to subtilisin variants comprising conservative mu- tations not pertaining the functional domain of the respective subtilisin protease. Depending on the %-identity values applicable as provided above, subtilisin variants of this embodiment have proteolytic activity and are at least 40%, at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% similar when compared to the full length polypeptide sequence of the parent enzyme.
Proteases (A), including serine proteases, according to the invention have“proteolytic activity” or“protease activity” or“proteolytic activity”. This property is related to hydrolytic activity of a protease (proteolysis, which means hydrolysis of peptide bonds linking amino acids together in a polypeptide chain) on protein containing substrates, e.g. casein, hemoglobin and BSA. Quan- titatively, proteolytic activity is related to the rate of degradation of protein by a protease or pro- teolytic enzyme in a defined course of time. The methods for analyzing proteolytic activity are well-known in the literature (see e.g. Gupta et al. (2002), Appl. Microbiol. Biotechnol. 60: 381 - 395). Proteolytic activity as such can be determined by using Succinyl-Ala-Ala-Pro-Phe-p- nitroanilide (Suc-AAPF-pNA, short AAPF; see e.g. DelMar et al. (1979), Analytical Biochem 99, 316-320) as substrate. pNA is cleaved from the substrate molecule by proteolytic cleavage, re- sulting in release of yellow color of free pNA which can be quantified by measuring OD405.
Protease variants may have proteolytic activity when said protease variants exhibit at least 20%, at least 25%, at least 30%, at least 35%, at least 40%, at least 45%, at 10 least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or 100% of the proteolytic activity of the respective parent protease.
Preferably, the pi value (isoelectric point) of subtilisin protease used in the present invention is in the range of from pH 7.0 to pH 10.0, preferably from pH 8.0 to pH 9.5.
Lipases and cutinases
In one embodiment, inventive compositions comprise at least one lipase (A).“Lipases”,“lipolytic enzyme”,“lipid esterase”, all refer to an enzyme of EC class 3.1.1 (“carboxylic ester hydrolase”). Such an enzyme (A) may have lipase activity (or lipolytic activity; triacylglycerol lipase, EC 3.1.1.3), cutinase activity (EC 3.1.1.74; enzymes having cutinase activity may be called cutinase herein), sterol esterase activity (EC 3.1.1.13) and/or wax-ester hydrolase activity (EC 3.1.1.50). Lipases include those of bacterial or fungal origin.
Commercially available lipase (A) include but are not limited to those sold under the trade names Lipolase™, Lipex™, Lipolex™ and Lipoclean™ (Novozymes A/S), Lumafast (originally from Genencor) and Lipomax (Gist-Brocades/ now DSM).
In one aspect of the invention, a suitable lipase is selected from the following:
- lipases from Humicoia (synonym Thermomyces), e.g. from H. lanuginosa ( T. lanuginosus) as described in EP 258068, EP 305216, WO 92/05249 and WO 2009/109500 or from H. insolens as described in WO 96/13580,
- lipases derived from Rhizomucor miehei as described in WO 92/05249,
- lipase from strains of Pseudomonas (some of these now renamed to Burkhoideria ), e.g. from P. alcaligenes or P. pseudoalcaligenes (EP 218272, WO 94/25578, WO 95/30744, WO 95/35381 , WO 96/00292), P. cepacia (EP 331376), P. stutzeri GB 1372034), P. fiuo- rescens, Pseudomonas sp. strain SD705 (WO 95/06720 and WO 96/27002), P. wiscon-
sinensis (WO 96/12012), Pseudomonas mendocina (WO 95/14783), P. giumae (WO 95/35381 , WO 96/00292),
- lipase from Streptomyces griseus (WO 2011 150157) and S. pristinaespiraiis (WO
2012/137147), GDSL-type Streptomyces lipases (WO 2010/065455),
- lipase from Thermobifida fusca as disclosed in WO 2011/084412,
- lipase from Geobaciiius stearothermophiius as disclosed in WO 2011/084417,
- Bacillus lipases, e.g. as disclosed in WO 00/60063, lipases from B. subti/is as disclosed in Dartois et al. (1992), Biochemica et Biophysica Acta, 1 131 , 253-360 or WO 2011/084599, B. stearothermophiius ( J P S64-074992) or B. pumiius (WO 91/16422),
- lipase from Candida antarctica as disclosed in WO 94/01541 ,
- cutinase from Pseudomonas mendocina (US 5389536, WO 88/09367),
- cutinase from Magnaporthe grisea (WO 2010/107560),
- cutinase from Fusarum soiani pisi as disclosed in WO 90/09446, WO 00/34450 and WO 01/92502,
- cutinase from Humicoia lanuginosa as disclosed in WO 00/34450 and WO 01/92502.
Suitable lipases (A) also include those referred to as acyltransferases or perhydrolases, e.g. acyltransferases with homology to Candida antarctica lipase A (WO 2010/11 1143), acyltrans- ferase from Mycobacterium smegmatis (WO 2005/056782), perhydrolases from the CE7 family (WO 2009/67279), and variants of the M. smegmatis perhydrolase in particular the S54V variant (WO 2010/100028).
Suitable lipases include also those which are variants of the above described lipases and/or cutinases which have lipolytic activity. Such suitable lipase variants are e.g. those which are developed by methods as disclosed in WO 95/22615, WO 97/04079, WO 97/07202, WO 00/60063, WO 2007/087508, EP 407225 and EP 260105.
Suitable lipases/cutinases (A) include also those that are variants of the above described lipas- es/cutinases which have lipolytic activity. Suitable lipase/cutinase variants include variants with at least 40 to 100% identity when compared to the full length polypeptide sequence of the par- ent enzyme as disclosed above. In one embodiment lipase/cutinase variants having lipolytic activity may be at least 40%, at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98% or at least 99% identical when compared to the full length polypeptide sequence of the parent en- zyme as disclosed above.
In another embodiment, inventive compositions comprise at least one lipase/cutinase variant comprising conservative mutations not pertaining the functional domain of the respective li- pase/cutinase. Lipase/cutinase variants of such embodiments having lipolytic activity may be at least 40%, at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91 %, at least 92%, at least 93%, at
least 94%, at least 95%, at least 96%, at least 97%, at least 98% or at least 99% similar when compared to the full length polypeptide sequence of the parent enzyme.
Lipases (A) have“lipolytic activity”. The methods for determining lipolytic activity are well-known in the literature (see e.g. Gupta et al. (2003), Biotechnol. Appl. Biochem. 37, p. 63-71 ). E.g. the lipase activity may be measured by ester bond hydrolysis in the substrate para-nitrophenyl pal- mitate (pNP-Palmitate, C:16) and releases pNP which is yellow and can be detected at 405 nm.
Lipase variants may have lipolytic activity according to the present invention when said lipase variants exhibit at least 20%, at least 25%, at least 30%, at least 35%, at least 40%, at least 45%, at 10 least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or 100% of the lipolytic activity of the re- spective parent lipase.
In one embodiment of the present invention, a combination of at least two of the foregoing li- pases (A) may be used.
Lipase (A) may be used in its non-purified form or in a purified form, e.g. purified with the aid of well-known adsorption methods, such as phenyl sepharose adsorption techniques.
In one embodiment of the present invention, lipases (A) are included in inventive composition in such an amount that a finished inventive composition has a lipolytic enzyme activity in the range of from 100 to 0.005 LU/mg, preferably 25 to 0.05 LU/mg of the composition. A Lipase Unit (LU) is that amount of lipase which produces 1 pmol of titratable fatty acid per minute in a pH stat. under the following conditions: temperature 30° C.; pH=9.0; substrate is an emulsion of 3.3 wt.
% of olive oil and 3.3% gum arabic, in the presence of 13 mmol/l Ca2+ and 20 mmol/l NaCI in 5 mmol/l Tris-buffer.
In one embodiment of the present invention, proteases (A) are included in inventive composition in such an amount that a finished inventive composition has a proteolytic enzyme activity in the range of from 0.1 to 50 GU.
It is preferred to use a combination of lipase (A) and protease (A) in compositions, for example 1 to 2% by weight of protease (A) and 0.1 to 0.5% by weight of lipase (A).
In the context of the present invention, an enzyme (A) is called stable when its enzymatic activi- ty“available in application” equals 100% when compared to the initial enzymatic activity before storage. An enzyme may be called stable within this invention if its enzymatic activity available in application is at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or at least 99.5% when compared to the initial enzymatic activity before storage.
In one embodiment, lipolytic activity available after storage at 37°C for 30 days is at least 60% when compared to the initial lipolytic activity before storage.
Subtracting a% from 100% gives the“loss of enzymatic activity during storage” when compared to the initial enzymatic activity before storage. In one embodiment, an enzyme is stable accord- ing to the invention when essentially no loss of enzymatic activity occurs during storage, i.e. loss in enzymatic activity equals 0% when compared to the initial enzymatic activity before stor- age. Essentially no loss of enzymatic activity within this invention may mean that the loss of enzymatic activity is less than 30%, less than 25%, less than 20%, less than 15%, less than 10%, less than 9%, less than 8%, less than 7%, less than 6%, less than 5%, less than 4%, less than 3%, less than 2%, or less than 1 % when compared to the initial enzymatic activity before storage.
In one embodiment, the loss of lipolytic activity after storage at 37°C for 30 days is less than 40% when compared to the initial lipolytic activity before storage.
Reduced loss of enzymatic activity within this invention may mean that the loss of enzymatic activity is reduced by at least 5%, by at least 10%, by at least 15%, by at least 20%, by at least 25%, by at least 30%, by at least 40%, by at least 50%, by least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or at least 99.5% when compared to the initial enzymatic activity before storage.
Compartment 1 further comprises chelating agent (B) dissolved in said aqueous medium, said chelating agent (B) being
(B1 ) at least one alkali metal salt of glutamic acid diacetic acid (GLDA), hereinafter also re- ferred to as complexing agent (B1 ) or simply as (B1 ), and
(B2) at least one alkali metal salt of methyl glycine diacetic acid (MGDA), hereinafter altogether also referred to as complexing agent (B2) or simply as (B2).
The term alkali refers to alkali metal cations, same or different, for example cations of lithium, sodium, potassium, rubidium, cesium, and combinations of at least two of the foregoing. Pre- ferred examples of alkali metal cations are sodium and potassium and combinations of sodium and potassium. Most preferred alkali metal cation is sodium.
Alkali metal salts may refer to fully or partially neutralized with alkali but not to ammonium ions in relation to the carboxyl groups of MGDA and GLDA.
In a preferred embodiment,
(B1 ) is at least one alkali metal salt of L- and D-enantiomers of glutamic acid diacetic acid (GLDA) or of enantiomerically pure L-GLDA, with an average of more than three of the carboxyl groups being neutralized with alkali, preferably an average of more than three and less than four of the carboxyl groups are neutralized with alkali metal, and
(B2) being at least one alkali metal salt of methyl glycine diacetic acid (MGDA), with an average of more than two and less than three of the carboxyl groups being neutralized with alkali.
The term carboxyl groups in the above definition refers to carboxyl groups per molecule.
Alkali metal salts of GLDA are selected from compounds according to general formula (I)
[OOC-CH2CH2-CH(COO)-N(CH2-COO)2]M4-xi-zi(NH4)zi Hxi (I) wherein
M is selected from alkali metal cations, same or different, as defined above,
The variable x1 in formula (I) may be in the range of from zero to 2.0, preferred are 0.02 to 0.5. In particularly preferred embodiments, x1 is from 0.1 to 0.3.
The variable z1 in formula (I) may be in the range of from zero to 1.0, preferred are 0.0005 to 0.5.
The sum of x1 +z1 may be zero. Preferably, however, the sum of x1 +z1 in formula (I) is prefera- bly greater than zero, for example 0.05 to 0.6.
Examples of M4-cΐ-zΐ(NH4)z2Hcΐ are Na4-xiHxi, [Nao.7(NH4)0.3]4-xiHxi, [(NH4)0.7Nao.3]4-xiHxi,
(Ko.7Nao.3)4-xiHxi, (Na0.7Ko.3)4-xiHxi, (Ko.22Nao.78)4-xi Hxi , (Nao.22Ko.78)4-xiHxi, and K4-XI HXI . Preferred examples of M4-xiHxi are selected from Na4-xi Hxi , (Ko esNao -isKx-iHx-i, and (Nao.ssKo.-isKxiHx-i.
In a preferred embodiment of the present invention, M is the same for (B1 ) and (B2). In embod- iments wherein combinations of different alkali metals are employed the ratio of such different alkali metal cations is preferably the same for both MGDA and GLDA.
In one embodiment of the present invention, the aqueous medium containing enzyme (A) and complexing agent(s) (B) have a pH value in the range of from 8 to 1 1 , preferably 9 to 10.8. The pH value is determined at ambient temperature at a dilution of 1 % by weight solids content.
Specifically, complexing agent (B1 ) is selected from at least one alkali metal salt of a mixture of L- and D- enantiomers according to formula (I), said mixture containing the racemic mixture or
preferably predominantly the respective L-isomer, for example with an enantiomeric excess (ee) in the range of from 5 to 99%, preferably 5 to 95%.
Alkali metal salts of MGDA are selected from compounds according to general formula (II)
[CH3-CH(COO)-N(CH2-COO)2]M3-X2-Z2(N H4)Z2HX2 (II) wherein
M is selected from alkali metal cations, same or different, for example cations of lithium, sodium, potassium, rubidium, cesium, and combinations of at least two of the foregoing. Preferred ex- amples of alkali metal cations are sodium and potassium and combinations of sodium and po- tassium.
The variable x2 in formula (II) may be in the range of from zero to 1 .0, preferred are 0.01 to 0.5. In particularly preferred embodiments, x2 is from 0.1 to 0.2.
The variable z2 in formula (II) may be in the range of from zero to 1 .0, preferred are 0.0005 to 0.5.
However, the sum of x2+z2 in formula (II) is greater than zero, for example 0.01 to 2.
Examples of M3-x2-z2(NH4)Z2HX2 are Na3-x2HX2, [Nao.7(NH4)o.3]3-x2HX2, [(NH4)o.7Nao.3]3-x2HX2,
(Ko.7Nao.3)3-x2Hx2, (Nao.7Ko.3)3-xHx, (Ko.22Nao.78)3-x2Hx2, (Nao.22Ko.78)3-x2HX2, and K3-c2Hc2. Preferred examples of M3-x2(NH )Z2HX2 are selected from Na3-x2Hx2, (K0.85Nao.i5)3-x2HX2, and (Nao.ssKo.-is
In one embodiment of the present invention, complexing agent (B2) is selected from at least one alkali metal salt of racemic MGDA and from alkali metal salts of mixtures of L- and D- enantiomers according to formula (II), said mixture containing predominantly the respective L- isomer with an enantiomeric excess (ee) in the range of from 5 to 99%, preferably 15 to 80 %, more preferably from 10 to 75% and even more preferably from 10 to 66%.
In one embodiment of the present invention, the total degree of alkali neutralization of (B2) and (B1 ) is in the range of from 0.80 to 0.98 mol-%, preferred are 0.90 to 0.97%. The total degree of alkali neutralization does not take into account any neutralization with ammonium.
The enantiomeric excess of MGDA and of its salts may be determined by measuring the polari- zation (polarimetry) or preferably by chromatography, for example by HPLC with a chiral col- umn, for example with one or more cyclodextrins as immobilized phase or with a ligand ex- change (Pirkle-brush) concept chiral stationary phase. Preferred is determination of the ee by HPLC with an immobilized optically active amine such as D-penicillamine in the presence of
copper(ll) salt. The enantiomeric excess of GLDA and of its salts may be determined by meas- uring the polarization (polarimetry).
In one embodiment of the present invention, alkali metal salts of complexing agent(s) (B) may contain one or more impurities that may result from the synthesis of the respective complexing agent(s) (B). In the cases of (B2) and (B1 ), such impurities may be propionic acid, lactic acid, glutamate, alanine, nitrilotriacetic acid (NTA) or the like and their respective alkali metal salts. Such impurities are usually present in minor amounts.“Minor amounts” in this context refer to a total of 0.1 to 5% by weight, referring to alkali metal salt of complexing agent (B), preferably up to 2.5% by weight. in one embodiment of the present invention, compartment 1 contains complexing agent (B1) as sole complexing agent.
In one embodiment of the present invention, the weight ratio of (B1 ) to (B1 ) or their respective salts in the mixture is in the range of from 1 :9 to 9:1 , preferably 1 : 3 to 3 : 1 , more preferably 2 : 3 to 3 : 2. The weight ratio may be determined, for example, by chromatographic methods such as HPLC.
In embodiments where two or more compounds of general formula (II) are present, the ee refers to the enantiomeric excess of all L-isomers present in the respective mixture compared to all D- isomers. For example, in cases wherein a mixture of the di- and trisodium salt of MGDA is pre- sent, the ee refers to the sum of the disodium salt and trisodium salt of L-MGDA with respect to the sum of the disodium salt and the trisodium salt of D-MGDA.
In one embodiment of the present invention, complexing agent(s) (B) may contain in the range of from 0.1 to 10 % by weight of one or more optically inactive impurities, at least one of the impurities being selected from iminodiacetic acid, formic acid, glycolic acid, propionic acid, ace- tic acid and their respective alkali metal or mono-, di- or triammonium salts. In one embodiment of the present invention, inventive mixtures may contain less than 0.2 % by weight of nitrilotri- acetic acid (NTA), preferably 0.01 to 0.1 % by weight. The percentages refer to total complexing agent(s) (B).
In one embodiment of the present invention, complexing agent(s) (B) may contain in the range of from 0.1 to 10 % by weight of one or more optically active impurities, at least one of the impu- rities being selected from L-carboxymethylalanine and its respective mono- or dialkali metal salts, L-carboxymethylglutamic acid and its respective di- or trialkali metal salts, and optically active mono- or diamides that result from an incomplete saponification during the synthesis of complexing agent(s) (B). Preferably, the amount of optically active impurities is in the range of from 0.2 to 10 % by weight, referring to the sum of complexing agent(s) (B). Even more prefera- bly, the amount of optically active impurities is in the range of from 1 to 7 % by weight.
In one embodiment of the present invention, complexing agent(s) (B) may contain minor amounts of cations other than alkali metal or ammonium. It is thus possible that minor amounts, such as 0.01 to 5 mol-% of total complexing agent, based on anion, bear alkali earth metal cati ons such as Mg2+ or Ca2+, or transition metal ions such as Fe2+ or Fe3+ cations.
The overall concentration of chelating agent(s) (B) in compartment 1 is in the range of from 35 to 75 % by weight, preferably 40 to 70% by weight, more preferably 42 to 70 % by weight and even more preferably 46 to 65 % by weight.
Aqueous medium refers to media in which the solvent is essentially water. In one embodiment, in such aqueous medium water is the sole solvent. In other embodiments, mixtures of water with one or more water-miscible solvents are used as aqueous medium. The term water- miscible solvent refers to organic solvents that are miscible with water at ambient temperature without phase-separation. Examples are ethylene glycol, 1 ,2-propylene glycol, isopropanol, and diethylene glycol. Preferably, at least 50 % by volume of the respective aqueous medium is wa- ter, referring to the solvent.
In one embodiment of the present invention, the aqueous medium containing complexing agent(s) (B) contains at least one inorganic basic salt selected from alkali metal carbonates. Preferred examples are sodium carbonate and potassium carbonate, for example 0.1 to 1.5 % by weight.
In one embodiment of the present invention, compartment 1 contains 0.5 to 20 g aqueous me- dium containing enzyme (A) and complexing agent(s) (B), preferably 1 to 8 g.
In one embodiment of the present invention, the solution containing enzyme (A) and complexing agent(s) (B) contains at least one dyestuff. Examples of dyestuffs are Acid Red 1 , Acid Red 52, Acid Blue 9, Acid Yellow 3, Acid Yellow 23, Acid Yellow 73, Acid Green 1 , Solvent Green 7, and Acid Green 25. In other embodiments, the solutions containing complexing agent(s) (B) addi- tionally contain a pigment formulation, for example a formulation of Cl Pigment Yellow 101 or Cl Pigment Blue 15:1.
The solution containing enzyme (A) and complexing agent(s) (B) may contain a viscosity modi- fying agent, for example a thickener. Preferably, though, the solution containing enzyme (A) and complexing agent(s) (B) does not contain any thickener.
The aqueous medium in compartment 1 may have an opaque or translucent or transparent ap- pearance. Preferably, neither haze nor precipitations may be detected on eyesight.
Inventive units contain one more compartment - hereinafter also referred to as additional com- partment - comprising at least one ingredient of a detergent other than enzyme (A) and chelat- ing agent(s) (B). Examples of such ingredients are surfactants or a combination of surfactants, a
bleaching agent, a bleach catalyst, or a builder other than complexing agents (B). In one em- bodiment, such inventive units may contain two or up to 10 further compartments with identical or different compositions of detergents. In a preferred embodiment of the present invention, in- ventive units comprise compartment 1 and a maximum of one or two additional compartments.
Detergent compositions comprised in said additional compartment(s) may contain one or more complexing agent other than MGDA and GLDA. Examples for complexing agent other than MGDA and GLDA are citrate, phosphonic acid derivatives, for example the disodium salt of hy- droxyethane-1 ,1-diphosphonic acid (“HEDP”), for example trisodium citrate, and phosphates such as STPP (sodium tripolyphosphate). Due to the fact that phosphates raise environmental concerns, it is preferred that detergent compositions comprised in inventive containers are free from phosphate. "Free from phosphate" is to be understood in the context of the present inven- tion as meaning that the content of phosphate and polyphosphate is in sum in the range from 10 ppm to 0.2% by weight, determined by gravimetric analysis and referring to the total detergent composition.
Detergent compositions comprised in said additional compartment(s) may contain one or more surfactant, preferably one or more non-ionic surfactant.
Preferred non-ionic surfactants are alkoxylated alcohols, di- and multiblock copolymers of eth- ylene oxide and propylene oxide and reaction products of sorbitan with ethylene oxide or pro- pylene oxide, alkyl polyglycosides (APG), hydroxyalkyl mixed ethers and amine oxides.
Preferred examples of alkoxylated alcohols and alkoxylated fatty alcohols are, for example, compounds of the general formula (III)
R2 is identical or different and selected from hydrogen and linear Ci-Cio-alkyl, preferably in each case identical and ethyl and particularly preferably hydrogen or methyl,
R3 is selected from C8-C22-alkyl, branched or linear, for example n-CsH , n-C-ioFh-i, n-C-^Fhs, n-Ci4H29, n-Ci6H33 or n-CieH37,
R4 is selected from Ci-Cio-alkyl, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, n-pentyl, isopentyl, sec-pentyl, neopentyl, 1 ,2-dimethylpropyl, isoamyl, n-hexyl, isohexyl, sec-hexyl, n-heptyl, n-octyl, 2-ethylhexyl, n-nonyl, n-decyl or isodecyl.
The variables e and f are in the range from zero to 300, where the sum of e and f is at least one, preferably in the range of from 3 to 50. Preferably, e is in the range from 1 to 100 and f is in the range from 0 to 30.
In one embodiment, compounds of the general formula (II I) may be block copolymers or random copolymers, preference being given to block copolymers.
Other preferred examples of alkoxylated alcohols are, for example, compounds of the general formula (IV)
R2 is identical or different and selected from hydrogen and linear Ci-Co-alkyl, preferably iden- tical in each case and ethyl and particularly preferably hydrogen or methyl,
R5 is selected from C6-C2o-alkyl, branched or linear, in particular n-CsH^, n-C-ioHh-i, n-Ci2H25, n-Ci3H27, h-OΐdHbΐ , n-Ci4H29, h-OΐqH33, n-CieH37, a is a number in the range from zero to 10, preferably from 1 to 6, b is a number in the range from 1 to 80, preferably from 4 to 20, d is a number in the range from zero to 50, preferably 4 to 25.
The sum a + b + d is preferably in the range of from 5 to 100, even more preferably in the range of from 9 to 50.
Preferred examples for hydroxyalkyl mixed ethers are compounds of the general formula (V)
R2 is identical or different and selected from hydrogen and linear Ci-Cio-alkyl, preferably in each case identical and ethyl and particularly preferably hydrogen or methyl,
R3 is selected from C8-C22-alkyl, branched or linear, for example iso-CnH23, iso-Ci3H27, n- CeHi7, n-CioH2i, n-Ci2H25, n-Ci4H29, n-Ci6H33 or n-CieH37,
R4 is selected from Ci-Cie-alkyl, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, n-pentyl, isopentyl, sec-pentyl, neopentyl, 1 ,2-dimethylpropyl, isoamyl, n-hexyl, isohexyl, sec-hexyl, n-heptyl, n-octyl, 2-ethylhexyl, n-nonyl, n-decyl, isodecyl, n-dodecyl, n-tetradecyl, n-hexadecyl, and n-octadecyl.
The variables m and n are in the range from zero to 300, where the sum of n and m is at least one, preferably in the range of from 5 to 50. Preferably, m is in the range from 1 to 100 and n is in the range from 0 to 30.
Compounds of the general formula (IV) and (V) may be block copolymers or random copoly- mers, preference being given to block copolymers.
Further suitable nonionic surfactants are selected from di- and multiblock copolymers, corn- posed of ethylene oxide and propylene oxide. Further suitable nonionic surfactants are selected from ethoxylated or propoxylated sorbitan esters. Amine oxides or alkyl polyglycosides, espe- cially linear C4-C-i6-alkyl polyglucosides and branched Cs-C-u-alkyl polyglycosides such as corn- pounds of general average formula (VI) are likewise suitable.
R6 is Ci-C4-alkyl, in particular ethyl, n-propyl or isopropyl,
R7 is -(CH2)2-R6,
G1 is selected from monosaccharides with 4 to 6 carbon atoms, especially from glucose and xylose, y in the range of from 1.1 to 4, y being an average number,
Further examples of non-ionic surfactants are compounds of general formula (VII) and (VIII)
I)
EO is ethylene oxide, CH2CH2-O,
R8 selected from Cs-C-is-alkyl, branched or linear, and R5 is defined as above. A30 is selected from propylene oxide and butylene oxide,
w is a number in the range of from 15 to 70, preferably 30 to 50,
w1 and w3 are numbers in the range of from 1 to 5, and
w2 is a number in the range of from 13 to 35. An overview of suitable further nonionic surfactants can be found in EP-A 0 851 023 and in DE- A 198 19 187.
Mixtures of two or more different nonionic surfactants may also be present. Other surfactants that may be present are selected from amphoteric (zwitterionic) surfactants and anionic surfactants and mixtures thereof.
Examples of amphoteric surfactants are those that bear a positive and a negative charge in the same molecule under use conditions. Preferred examples of amphoteric surfactants are so- called betaine-surfactants. Many examples of betaine-surfactants bear one quaternized nitrogen atom and one carboxylic acid group per molecule. A particularly preferred example of amphoter- ic surfactants is cocamidopropyl betaine (lauramidopropyl betaine).
Examples of amine oxide surfactants are compounds of the general formula (IX)
R9R1°R11N 0 (IX) wherein R9, R10, and R11 are selected independently from each other from aliphatic, cycloali- phatic or C2-C4-alkylene Cio-C2o-alkylamido moieties. Preferably, R9 is selected from C8-C20- alkyl or C2-C4-alkylene Cio-C2o-alkylamido and R10 and R11 are both methyl.
A particularly preferred example is lauryl dimethyl aminoxide, sometimes also called lauramine oxide. A further particularly preferred example is cocamidylpropyl dimethylaminoxide, some- times also called cocamidopropylamine oxide.
Examples of suitable anionic surfactants are alkali metal and ammonium salts of Cs-C-is-alkyl sulfates, of Cs-C-is-fatty alcohol polyether sulfates, of sulfuric acid half-esters of ethoxylated C4- Ci2-alkylphenols (ethoxylation: 1 to 50 mol of ethylene oxide/mol), C12-C18 sulfo fatty acid alkyl esters, for example of C12-C18 sulfo fatty acid methyl esters, furthermore of Ci2-Ci8-alkylsulfonic acids and of Cio-Ci8-alkylarylsulfonic acids. Preference is given to the alkali metal salts of the aforementioned compounds, particularly preferably the sodium salts.
Further examples for suitable anionic surfactants are soaps, for example the sodium or potassi- um salts of stearic acid, oleic acid, palmitic acid, ether carboxylates, and alkylether phosphates.
In one embodiment of the present invention, detergent compositions comprised in inventive containers may contain 0.1 to 60 % by weight of at least one surfactant, selected from anionic surfactants, amphoteric surfactants and amine oxide surfactants.
In a preferred embodiment, detergent compositions comprised in said additional compart- ment(s) do not contain any anionic surfactant.
Detergent compositions comprised in said additional compartment(s) may contain at least one bleaching agent, also referred to as bleach. Bleaching agents may be selected from chlorine bleach and peroxide bleach, and peroxide bleach may be selected from inorganic peroxide bleach and organic peroxide bleach. Preferred are inorganic peroxide bleaches, selected from alkali metal percarbonate, alkali metal perborate and alkali metal persulfate.
Examples of organic peroxide bleaches are organic percarboxylic acids, especially organic per- carboxylic acids.
In solid detergent compositions, alkali metal percarbonates, especially sodium percarbonates, are preferably used in coated form. Such coatings may be of organic or inorganic nature. Ex- amples are glycerol, sodium sulfate, silicate, sodium carbonate, and combinations of at least two of the foregoing, for example combinations of sodium carbonate and sodium sulfate.
Suitable chlorine-containing bleaches are, for example, 1 ,3-dichloro-5,5-dimethylhydantoin, N-chlorosulfamide, chloramine T, chloramine B, sodium hypochlorite, calcium hypochlorite, magnesium hypochlorite, potassium hypochlorite, potassium dichloroisocyanurate and sodium dichloroisocyanurate.
Detergent compositions comprised in said additional compartment(s) compositions may corn- prise, for example, in the range from 3 to 10% by weight of chlorine-containing bleach.
Detergent compositions comprised in said additional compartment(s) may comprise one or more bleach catalysts. Bleach catalysts can be selected from bleach-boosting transition metal salts or transition metal complexes such as, for example, manganese-, iron-, cobalt-, ruthenium- or molybdenum-salen complexes or carbonyl complexes. Manganese, iron, cobalt, ruthenium, molybdenum, titanium, vanadium and copper complexes with nitrogen-containing tripod ligands and also cobalt-, iron-, copper- and ruthenium-amine complexes can also be used as bleach catalysts.
Detergent compositions comprised in said additional compartment(s) may comprise one or more bleach activators, for example N-methylmorpholinium-acetonitrile salts (“MMA salts”), tri- methylammonium acetonitrile salts, N-acylimides such as, for example, N-nonanoylsuccinimide, 1 ,5-diacetyl-2,2-dioxohexahydro-1 ,3,5-triazine (“DADHT”) or nitrile quats (trimethylammonium acetonitrile salts).
Further examples of suitable bleach activators are tetraacetylethylenediamine (TAED) and tetraacetylhexylenediamine.
Detergent compositions comprised in said additional compartment(s) may comprise one or more corrosion inhibitors. In the present case, this is to be understood as including those corn- pounds which inhibit the corrosion of metal. Examples of suitable corrosion inhibitors are tria zoles, in particular benzotriazoles, bisbenzotriazoles, aminotriazoles, alkylaminotriazoles, also phenol derivatives such as, for example, hydroquinone, pyrocatechol, hydroxyhydroquinone, gallic acid, phloroglucinol or pyrogallol.
In one embodiment of the present invention, detergent compositions comprised in said addi- tional compartment(s) comprise in total in the range from 0.1 to 1.5% by weight of corrosion inhibitor.
Detergent compositions comprised in said additional compartment(s) may comprise one or more builders, selected from organic and inorganic builders. Examples of suitable inorganic builders are sodium sulfate or sodium carbonate or silicates, in particular sodium disilicate and sodium metasilicate, zeolites, sheet silicates, in particular those of the formula a-I ^ShOs, b- I ^ShOs, and b-I ^ShOs, also fatty acid sulfonates, a-hydroxypropionic acid, alkali metal ma-
lonates, fatty acid sulfonates, alkyl and alkenyl disuccinates, tartaric acid diacetate, tartaric acid monoacetate, oxidized starch, and polymeric builders, for example polycarboxylates and poly- aspartic acid.
Examples of organic builders are especially polymers and copolymers. In one embodiment of the present invention, organic builders are selected from polycarboxylates, for example alkali metal salts of (meth)acrylic acid homopolymers or (meth)acrylic acid copolymers.
Suitable comonomers are monoethylenically unsaturated dicarboxylic acids such as maleic ac- id, fumaric acid, maleic anhydride, itaconic acid and citraconic acid. A suitable polymer is in par- ticular polyacrylic acid, which preferably has an average molecular weight Mw in the range from 2000 to 40 000 g/mol, preferably 2000 to 10 000 g/mol, in particular 3000 to 8000 g/mol. Also of suitability are copolymeric polycarboxylates, in particular those of acrylic acid with methacrylic acid and of acrylic acid or methacrylic acid with maleic acid and/or fumaric acid, and in the same range of molecular weight.
It is also possible to use copolymers of at least one monomer from the group consisting of mo- noethylenically unsaturated C3-Cio-mono- or C4-Cio-dicarboxylic acids or anhydrides thereof, such as maleic acid, maleic anhydride, acrylic acid, methacrylic acid, fumaric acid, itaconic acid and citraconic acid, with at least one hydrophilic or hydrophobic monomer as listed below.
Suitable hydrophobic monomers are, for example, isobutene, diisobutene, butene, pentene, hexene and styrene, olefins with 10 or more carbon atoms or mixtures thereof, such as, for ex- ample, 1-decene, 1-dodecene, 1-tetradecene, 1 -hexadecene, 1 -octadecene, 1 -eicosene, 1 - docosene, 1 -tetracosene and 1-hexacosene, C22-oolefin, a mixture of C2o-C24-a-olefins and polyisobutene having on average 12 to 100 carbon atoms per molecule.
Suitable hydrophilic monomers are monomers with sulfonate or phosphonate groups, and also nonionic monomers with hydroxyl function or alkylene oxide groups. By way of example, men- tion may be made of: allyl alcohol, isoprenol, methoxypolyethylene glycol (meth)acrylate, meth- oxypolypropylene glycol (meth)acrylate, methoxypolybutylene glycol (meth)acrylate, methoxy- poly(propylene oxide-co-ethylene oxide) (meth)acrylate, ethoxypolyethylene glycol
(meth)acrylate, ethoxypolypropylene glycol (meth)acrylate, ethoxypolybutylene glycol
(meth)acrylate and ethoxypoly(propylene oxide-co-ethylene oxide) (meth)acrylate. Polyalkylene glycols here may comprise 3 to 50, in particular 5 to 40 and especially 10 to 30 alkylene oxide units per molecule.
Particularly preferred sulfonic-acid-group-containing monomers here are 1 -acrylamido-
1-propanesulfonic acid, 2-acrylamido-2-propanesulfonic acid, 2-acrylamido-
2-methylpropanesulfonic acid, 2-methacrylamido-2-methylpropanesulfonic acid,
3-methacrylamido-2-hydroxypropanesulfonic acid, allylsulfonic acid, methallylsulfonic acid, al- lyloxybenzenesulfonic acid, methallyloxybenzenesulfonic acid, 2-hydroxy-
3-(2-propenyloxy)propanesulfonic acid, 2-methyl-2-propene-1 -sulfonic acid, styrenesulfonic ac- id, vinylsulfonic acid, 3-sulfopropyl acrylate, 2-sulfoethyl methacrylate, 3-sulfopropyl methacry- late, sulfomethacrylamide, sulfomethylmethacrylamide, and salts of said acids, such as sodium, potassium or ammonium salts thereof.
Particularly preferred phosphonate-group-containing monomers are vinylphosphonic acid and its salts.
A further example of builders is carboxymethyl inulin.
Moreover, amphoteric polymers can also be used as builders.
Detergent compositions comprised in said additional compartment(s) may comprise, for exam- pie, in the range from in total 10 to 70% by weight, preferably up to 50% by weight, of builder. In the context of the present invention, neither MGDA nor GLDA are counted as builder.
In one embodiment of the present invention, such detergent compositions comprised in said additional compartment(s) may comprise one or more co-builders.
Detergent compositions comprised in said additional compartment(s) may comprise one or more antifoams, selected for example from silicone oils and paraffin oils. In one embodiment of the present invention, detergent compositions comprised in inventive containers compositions comprise in total in the range from 0.05 to 0.5% by weight of antifoam.
In one embodiment of the present invention, detergent compositions comprised in said addi- tional compartment(s) may comprise, for example, up to 5% by weight of enzyme, preference being given to 0.1 to 3% by weight. Said enzyme may be stabilized, for example with the sodi- um salt of at least one Ci-C3-carboxylic acid or C4-Cio-dicarboxylic acid. Preferred are formates, acetates, adipates, and succinates.
In one embodiment of the present invention, detergent compositions comprised in said addi- tional compartment(s) comprise at least one zinc salt. Zinc salts can be selected from water- soluble and water-insoluble zinc salts. In this connection, within the context of the present in- vention, water-insoluble is used to refer to those zinc salts which, in distilled water at 25°C, have a solubility of 0.1 g/l or less. Zinc salts which have a higher solubility in water are accordingly referred to within the context of the present invention as water-soluble zinc salts.
In one embodiment of the present invention, zinc salt is selected from zinc benzoate, zinc glu conate, zinc lactate, zinc formate, ZnCh, ZnS04, zinc acetate, zinc citrate, Zn(NOs)2,
Zn(CH3S03)2 and zinc gallate, preferably ZnCh, ZnS04, zinc acetate, zinc citrate, Zn(NOs)2, Zn(CH3S03)2 and zinc gallate.
In another embodiment of the present invention, zinc salt is selected from ZnO, ZnO-aq,
Zn(OH)2 and ZnC03. Preference is given to ZnO-aq.
In one embodiment of the present invention, zinc salt is selected from zinc oxides with an aver- age particle diameter (weight-average) in the range from 10 nm to 100 pm.
The cation in zinc salt can be present in complexed form, for example complexed with ammonia ligands or water ligands, and in particular be present in hydrated form. To simplify the notation, within the context of the present invention, ligands are generally omitted if they are water lig- ands.
In one embodiment of the present invention, detergent compositions comprised in said addi- tional compartment(s) may comprise additional enzyme (A). In other embodiments, no enzyme (A) is comprised in said additional compartment(s).
In one embodiment of the present invention, detergent compositions comprised in said addi- tional compartment(s) may comprise complexing agent (B). In other embodiments, neither MGDA nor GLDA is comprised in said additional compartment(s).
Detergent compositions comprised in inventive units fulfil numerous functions. They exhibit good cleaning properties in automatic dishwashing applications. They show a good storage and shelf-life behavior and a low tendency to colorization and especially yellowing.
Another aspect of the present invention is directed towards the use of inventive packaging units for dishwashing or laundry cleaning. Dishwashing and laundry cleaning may refer to home care or to industrial and institutional applications, home care applications being preferred. Particularly preferred is automatic dishwash in home care applications.
Another aspect of the present invention is directed towards a process for making inventive con- tainers comprising a single unit dose, said process also being referred to as inventive process. Another aspect of the present invention is directed towards a process for making a compart- ment of a container according to the present invention, hereinafter also being referred to as in- ventive process. The inventive process comprises several steps, hereinafter also being referred to as steps (a) to (e), and said steps briefly being summarized as follows:
(a) providing a polymer,
(b) shaping the polymer in a way that it has at least one recess so it can contain a liquid,
(c) providing enzyme (A) and complexing agent (B1 ) and (B2) dissolved in an aqueous me- dium,
(d) placing said aqueous medium containing enzyme (A) and complexing agent(s) (B) ac- cording to step (c) into the formed recess according to step (b),
(e) closing the open container or a compartment, respectively.
In a preferred embodiment, the container is a pouch made from a polymer film. Preferably, the polymer is polyvinyl alcohol. Steps (a) to (e) are described hereinafter in more detail.
Step (a) refers to providing a polymer, preferably a polymer film and even more preferably a film from polyvinyl alcohol.
In embodiments wherein the container or its respective compartment is different from a pouch, such polymer may have a different thickness compared to films, preferably a greater thickness.
It may be in the form of granules, and step (b) - shaping the polymer - may be performed, for example, by injection molding.
In a preferred embodiment of the present invention, polymer films and preferably polyvinyl alco- hol films used for making pouches for inventive compositions have a thickness (strength) in the range of from 10 to 100 pm, preferably 20 to 90 pm, even more preferably 25 to 40 pm. If the strength of polymer films and especially of polyvinyl alcohol films exceeds 100 pm it takes too long to dissolve them during the washing cycle. If the strength of polymer films and especially of polyvinyl alcohol films is below 10 pm they are too sensitive to mechanical stress.
In step (b), the polymer - preferably, the polymer film - is shaped in a way that it has at least one recess so it can contain a liquid. Examples are thermoforming processes, especially at a temperature of 5 to 20°C below the melting point of the respective polymer.
In embodiments wherein said container is a pouch the shaping may be performed through shaping in shaping a hose and cutting the hose into shorter pieces and closing one side each, thereby shaping sachets.
In special embodiments of the inventive process, step (b) is performed with the aid of a forming die having at least one cavity, preferably with a plurality of cavities. Such cavities may have ap- ertures (holes) through which reduced pressure (“vacuum”) may be applied. In such special embodiments, a polymer film is placed of the die. The polymer is then heated through a heating device. The polymer filmed is simultaneously shaped by the application of a vacuum for exam- pie through apertures of the cavity/the cavities.
In addition to applying the vacuum, it is possible to blow air or an inert gas against the polymer film in order to force it into intimate contact with the die.
In step (c), enzyme (A) and complexing agent(s) (B) dissolved in an aqueous medium is provid- ed. Methods to make such solutions of complexing agent(s) (B) are know from the art.
In step (d) of the inventive process, aqueous medium containing complexing agent(s) (B) is then placed into the recesses obtained in step (b). Step (d) can be performed by applying pressure or simply using gravity. Applying pressure is preferred. In embodiments wherein a die with a
plurality of cavities has been used, it is preferred to simultaneously place aqueous medium con- taining complexing agent(s) (B) into more than one recess.
In one embodiment, the recesses are filled completely. In other embodiments, the recesses are only filled partially, for example 50 to 90 % by volume, the latter embodiment being preferred in order to prevent spilling of aqueous medium in step (e) to follow.
In step (e) of the inventive process, the filled but still open containers are closed. It is preferred to perform such closing step by sealing, for example heat-sealing. Other embodiments refer to gluing a closing device on the open container, for example a polymer film, preferably a film made from water-soluble polymer.
In order to achieve sealing or heat-sealing of pouches, it is preferred to provide another polymer film and place it on the die containing the shaped film containing aqueous medium containing complexing agent(s) (B).
In other embodiments wherein sachets of polymer film have been formed and at least partially filled with aqueous medium containing complexing agent(s) (B) they may be closed by simply applying heat to the upper rim of the sachets, for example through a heated metal device. In other embodiments containers made from polymer film may be closed by performing a chemical reaction of a sealing substance. Said chemical reaction may be promoted by applying a vacu- um.
In other embodiments, steps (b), (d) and (e) are performed as a vertical form-fill-seal route yield ing envelope-shaped pouches that contain aqueous medium containing complexing agent(s)
(B).
The present invention is further illustrated by working examples.
Percentages refer to % by weight unless expressly specified otherwise. The contents of MGDA- Na3 and of GLDA-Na4 were determined by measuring the iron binding capacity (FeCl3, potenti- ometric detection).
I. Manufacture of Mixtures of enzyme (A) and chelating agents (B1 ) and (B2)
(B1 .1 ): Aqueous solution of GLDA-Na4: 47.6% by weight
(B2.1 ): Aqueous solution of MGDA-Na3: 40% by weight
(B1 .1 ) + (B2.1 ): Aqueous solution of MGDA-Na3/GLDA-Na4: 1 :1 by weight mixture: 53% by weight solids content
C-AS.1 : From a 100 g 40% By weight solution of (B2.1 ) 3.5 g water were removed. To the solu- tion were then added 2.5 g formic acid (100%) and 1 g protease. The pH value of the solution was 9.8. The activity of the enzyme in the chelating agent solution was measured over time us- ing a Gallery Plus (Thermo Scientific).
AS.2: From a 100 g solution of (B1.1 ) 3.2 g water were removed. To the solution were then added 2.2 g formic acid (100%) and 1 g protease. The final pH of the solution is 9.8. The activity of the enzyme in the chelating agent solution was measured over time using a Gallery Plus (Thermo Scientific).
AS.3 From a 100 g solution of a mixture from (B1.1 ) + (B2.1 ) (1 :1) 3.5 g water were removed. To the solution were then added 2.5 g formic acid (100%) and 1 g protease. The final pH of the solution is 9.8. The activity of the enzyme in the chelating agent solution was measured over time using a Gallery Plus (Thermo Scientific).
C-AS.4 To 100 g distillated water 0.001 g NaOH (1 M) and 1 g protease were added. The final pH of the solution is 9.7.
The solutions were stored at 37°C. The activity of the enzyme was measured over time over a period of 16 weeks.
Table 1 : Enzyme activity
Polyvinylalcohol films with a degree of saponification: 88 mole-%, thickness 38 pm, commercial- ly available from MonoSol were used.
A polyvinylalcohol film was cut into rectangular pieces of 12-5 cm each. Envelope-like pouches were made by folding the rectangular pieces accordingly. The brinks were connected by heat- sealing. With a pipette, 10 ml of 53% by weight solution of protease and complexing agents (B) as obtained in example I. were placed into the envelope-like pouch. The pouch was closed again by heat-sealing. After 4 weeks of storage at 23°C, neither deterioration of the pouch with AS.2 or AS.3 nor disadvantageous change of the contents could be observed.
III. Manufacture of detergent compositions Example detergent compositions according to Table 2 are prepared by mixing the respective ingredients in dry state.
Table 2: Example detergent compositions for automatic dishwashing
Tablets may be formed of any of the above mixture, weight: 18 g, and one pouch from (II) may be placed on each tablet. The tablets are packed - together with the pouch - into a film of poly- vinyl alcohol, degree of saponification: 88 mole-%, thickness: 35 pm. They may be used as unit doses in an automatic dishwasher and yield excellent dishwashing results. Their use is conven- ient.
Claims
1. Packaging unit containing a detergent single unit dose wherein said packaging unit com- prises a compartment 1 comprising an aqueous solution of enzyme (A) and at least one chelat- ing agent (B) dissolved in said aqueous solution, said chelating agent (B) being
(B1 ) at least one alkali metal salt of glutamic acid diacetic acid (GLDA), and
(B2) at least one alkali metal salt of methyl glycine diacetic acid (MGDA), and one more compartment comprising at least one ingredient of a detergent other than enzyme (A) and chelating agent(s) (B), wherein the overall concentration of chelating agent(s) (B) in compartment 1 is in the range of from 35 to 75 % by weight.
2. Packaging unit according to claim 1 wherein compartment 1 is in the form of a pouch.
3. Packaging unit according to claim 1 or 2 wherein enzyme (A) is selected from proteases, lipases ad amylases.
4. Packaging unit according to any of the preceding claims wherein (B1 ) is selected from compounds according to general formula (I)
[OOC-CH2CH2-CH(COO)-N(CH2-COO)2]M4-xi-zi(NH4)ziHxi (I) wherein M is selected from sodium and potassium and combinations thereof, and wherein each x1 and z1 are in the range from 0.0 to 1.0 and the sum of x1 +z1 is greater than zero but less than 1.0.
5. Packaging unit according to any of the preceding claims wherein the alkali metal salt (B1 ) is a mixture of L- and D- enantiomers of glutamic acid diacetic acid (GLDA), said mixture containing the racemic mixture or preferably predominantly the respective L-isomer, for example with an enantiomeric excess (ee) in the range of from 5 to 99%.
6. Packaging unit according to any of the preceding claims wherein the alkali metal salt (B2) is a mixture of L- and D- enantiomers of methyl glycine diacetic acid (MGDA), said mixture containing predominantly the respective L-isomer with an enantiomeric excess (ee) in the range of from 15 to 80 %.
7. Packaging unit according to any of the preceding claims wherein said aqueous medium in compartment 1 has a pH value in the range of from 8 to 11 .
8. Packaging unit according to any of the preceding claims wherein said unit encompasses at least two compartments, wherein said detergent additionally comprises an inorganic peroxide, said inorganic peroxide not being in the compartment containing the aqueous medium containing chelating agents (B).
9. Packaging unit according to any of the preceding claims wherein the aqueous solution of chelating agent(s) (B) has a pH value in the range of from 9 to 10.8.
10. Packaging unit according to any of the preceding claims wherein the solution in compart- ment 1 additionally comprises a dyestuff.
1 1. Use of a packing unit according to any of the preceding claims for dishwashing or laundry cleaning.
12. Process for making a unit or a compartment of a unit in accordance with any of claims 1 to 10, said process comprising the steps of
(a) providing a polymer,
(b) shaping the polymer in a way that it has at least one recess so it can contain a liquid,
(c) providing enzyme (A) and chelating agents (B1 ) and (B2) dissolved in an aqueous medium,
(d) placing said aqueous medium according to step (c) into the formed recess according to step (b),
(e) closing the open unit or compartment, respectively.
13. Process according to claim 12 wherein step (e) is performed by sealing.
14. Process according to claim 12 or 13 wherein step (b) is performed with the aid of a form- ing die having at least one cavity.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP18188434.7 | 2018-08-10 | ||
| EP18188434 | 2018-08-10 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020030623A1 true WO2020030623A1 (en) | 2020-02-13 |
Family
ID=63209252
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2019/071083 Ceased WO2020030623A1 (en) | 2018-08-10 | 2019-08-06 | Packaging unit comprising a detergent composition containing an enzyme and at least one chelating agent |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2020030623A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20240015655A (en) | 2021-05-31 | 2024-02-05 | 다우 도레이 캄파니 리미티드 | Display device, especially haptic display device and method for designing the same |
| WO2024151575A1 (en) * | 2023-01-09 | 2024-07-18 | The Procter & Gamble Company | Superposed multi-sectioned water-soluble unit dose automatic dishwashing detergent pouch |
| US12275919B2 (en) | 2019-04-01 | 2025-04-15 | Basf Se | Process for making a granule or powder |
Citations (80)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1243784A (en) | 1967-10-03 | 1971-08-25 | Novo Terapeutisk Labor As | Proteolytic enzymes, their production and use |
| EP0218272A1 (en) | 1985-08-09 | 1987-04-15 | Gist-Brocades N.V. | Novel lipolytic enzymes and their use in detergent compositions |
| EP0258068A2 (en) | 1986-08-29 | 1988-03-02 | Novo Nordisk A/S | Enzymatic detergent additive |
| EP0260105A2 (en) | 1986-09-09 | 1988-03-16 | Genencor, Inc. | Preparation of enzymes having altered activity |
| EP0283075A2 (en) | 1987-02-27 | 1988-09-21 | Gist-Brocades N.V. | Molecular cloning and expression of genes encoding proteolytic enzymes |
| WO1988009367A1 (en) | 1987-05-29 | 1988-12-01 | Genencor, Inc. | Cutinase cleaning composition |
| EP0305216A1 (en) | 1987-08-28 | 1989-03-01 | Novo Nordisk A/S | Recombinant Humicola lipase and process for the production of recombinant humicola lipases |
| WO1989006279A1 (en) | 1988-01-07 | 1989-07-13 | Novo-Nordisk A/S | Mutated subtilisin genes |
| WO1989006276A2 (en) | 1988-01-08 | 1989-07-13 | Dpz Deutsches Primatenzentrum Gesellschaft Mbh | Hiv-2-type retroviruses of primates, vaccines, diagnostic and pharmaceutical compositions |
| EP0331376A2 (en) | 1988-02-28 | 1989-09-06 | Amano Pharmaceutical Co., Ltd. | Recombinant DNA, bacterium of the genus pseudomonas containing it, and process for preparing lipase by using it |
| WO1989009830A1 (en) | 1988-04-12 | 1989-10-19 | Genex Corporation | Subtilisin mutations |
| WO1989009819A1 (en) | 1988-04-12 | 1989-10-19 | Genex Corporation | Combining mutations for stabilization of subtilisin |
| WO1990009446A1 (en) | 1989-02-17 | 1990-08-23 | Plant Genetic Systems N.V. | Cutinase |
| EP0407225A1 (en) | 1989-07-07 | 1991-01-09 | Unilever Plc | Enzymes and enzymatic detergent compositions |
| WO1991002792A1 (en) | 1989-08-25 | 1991-03-07 | Henkel Research Corporation | Alkaline proteolytic enzyme and method of production |
| WO1991006637A1 (en) | 1989-10-31 | 1991-05-16 | Genencor International, Inc. | Subtilisin mutants |
| WO1991016422A1 (en) | 1990-04-14 | 1991-10-31 | Kali-Chemie Aktiengesellschaft | Alkaline bacillus lipases, coding dna sequences therefor and bacilli which produce these lipases |
| WO1992005249A1 (en) | 1990-09-13 | 1992-04-02 | Novo Nordisk A/S | Lipase variants |
| WO1992019729A1 (en) | 1991-05-01 | 1992-11-12 | Novo Nordisk A/S | Stabilized enzymes and detergent compositions |
| WO1994001541A1 (en) | 1992-07-06 | 1994-01-20 | Novo Nordisk A/S | C. antarctica lipase and lipase variants |
| WO1994025578A1 (en) | 1993-04-27 | 1994-11-10 | Gist-Brocades N.V. | New lipase variants for use in detergent applications |
| US5389536A (en) | 1986-11-19 | 1995-02-14 | Genencor, Inc. | Lipase from Pseudomonas mendocina having cutinase activity |
| WO1995006720A1 (en) | 1993-08-30 | 1995-03-09 | Showa Denko K.K. | Novel lipase, microorganism producing the lipase, process for producing the lipase, and use of the lipase |
| WO1995014783A1 (en) | 1993-11-24 | 1995-06-01 | Showa Denko K.K. | Lipase gene and variant lipase |
| WO1995022615A1 (en) | 1994-02-22 | 1995-08-24 | Novo Nordisk A/S | A method of preparing a variant of a lipolytic enzyme |
| WO1995023221A1 (en) | 1994-02-24 | 1995-08-31 | Cognis, Inc. | Improved enzymes and detergents containing them |
| WO1995030744A2 (en) | 1994-05-04 | 1995-11-16 | Genencor International Inc. | Lipases with improved surfactant resistance |
| WO1995035381A1 (en) | 1994-06-20 | 1995-12-28 | Unilever N.V. | Modified pseudomonas lipases and their use |
| WO1996000292A1 (en) | 1994-06-23 | 1996-01-04 | Unilever N.V. | Modified pseudomonas lipases and their use |
| WO1996012012A1 (en) | 1994-10-14 | 1996-04-25 | Solvay S.A. | Lipase, microorganism producing same, method for preparing said lipase and uses thereof |
| WO1996013580A1 (en) | 1994-10-26 | 1996-05-09 | Novo Nordisk A/S | An enzyme with lipolytic activity |
| WO1996027002A1 (en) | 1995-02-27 | 1996-09-06 | Novo Nordisk A/S | Novel lipase gene and process for the production of lipase with the use of the same |
| WO1996034946A1 (en) | 1995-05-05 | 1996-11-07 | Novo Nordisk A/S | Protease variants and compositions |
| WO1997004079A1 (en) | 1995-07-14 | 1997-02-06 | Novo Nordisk A/S | A modified enzyme with lipolytic activity |
| WO1997007202A1 (en) | 1995-08-11 | 1997-02-27 | Novo Nordisk A/S | Novel lipolytic enzymes |
| WO1998020115A1 (en) | 1996-11-04 | 1998-05-14 | Novo Nordisk A/S | Subtilase variants and compositions |
| WO1998020116A1 (en) | 1996-11-04 | 1998-05-14 | Novo Nordisk A/S | Subtilase variants and compositions |
| EP0851023A2 (en) | 1996-12-23 | 1998-07-01 | Unilever N.V. | Machine dishwashing tablets containing a peracid |
| WO1999011768A1 (en) | 1997-08-29 | 1999-03-11 | Novo Nordisk A/S | Protease variants and compositions |
| DE19819187A1 (en) | 1998-04-30 | 1999-11-11 | Henkel Kgaa | Solid dishwasher detergent with phosphate and crystalline layered silicates |
| WO2000034450A1 (en) | 1998-12-04 | 2000-06-15 | Novozymes A/S | Cutinase variants |
| WO2000060063A1 (en) | 1999-03-31 | 2000-10-12 | Novozymes A/S | Lipase variant |
| WO2001044452A1 (en) | 1999-12-15 | 2001-06-21 | Novozymes A/S | Subtilase variants having an improved wash performance on egg stains |
| WO2001092502A1 (en) | 2000-06-02 | 2001-12-06 | Novozymes A/S | Cutinase variants |
| WO2002042400A2 (en) | 2000-11-27 | 2002-05-30 | The Procter & Gamble Company | Dishwashing method |
| DE10064983A1 (en) | 2000-12-23 | 2002-07-18 | Henkel Kgaa | New subtilisin protease from Bacillus alcalophilus, useful e.g. in washing and cleaning compositions, comprises at positions 230, 256 and 259 amino acids Val, Gly and Asn |
| WO2002088340A2 (en) | 2001-05-02 | 2002-11-07 | Henkel Kommanditgesellschaft Auf Aktien | Alkaline protease variants and detergents and cleaning agents containing said alkaline protease variants |
| WO2003006602A2 (en) | 2001-07-12 | 2003-01-23 | Novozymes A/S | Subtilase variants |
| WO2003054184A1 (en) | 2001-12-20 | 2003-07-03 | Henkel Kommanditgesellschaft Auf Aktien | Novel alkali protease formed by bacillus gibsonii (dsm 14393) and washing and cleaning agents containing said novel alkali protease |
| WO2003055974A2 (en) | 2001-12-22 | 2003-07-10 | Henkel Kommanditgesellschaft Auf Aktien | Novel alkaline protease from bacillus sp. (dsm 14392) and washing and cleaning products comprising said novel alkaline protease |
| WO2003056017A2 (en) | 2001-12-22 | 2003-07-10 | Henkel Kommanditgesellschaft Auf Aktien | Alkaline protease from bacillus sp. (dsm 14390) and washing and cleaning products comprising said alkaline protease |
| WO2004003186A2 (en) | 2002-06-26 | 2004-01-08 | Novozymes A/S | Subtilases and subtilase variants having altered immunogenicity |
| WO2004041979A2 (en) | 2002-11-06 | 2004-05-21 | Novozymes A/S | Subtilase variants |
| WO2005056782A2 (en) | 2003-12-03 | 2005-06-23 | Genencor International, Inc. | Perhydrolase |
| WO2005063974A1 (en) | 2003-12-23 | 2005-07-14 | Henkel Kommanditgesellschaft Auf Aktien | Novel alkaline protease and washing and cleaning products containing said novel alkaline protease |
| WO2005103244A1 (en) | 2004-04-23 | 2005-11-03 | Henkel Kommanditgesellschaft Auf Aktien | Novel alkaline proteases, and detergents and cleaners containing the same |
| DE102005028295A1 (en) | 2005-06-18 | 2006-11-16 | Henkel Kgaa | New polynucleotide encoding a protease active at low temperature, for use e.g. in cleaning, pharmaceutical and cosmetic compositions and for eliminating biofilms |
| WO2007006305A1 (en) | 2005-07-08 | 2007-01-18 | Novozymes A/S | Subtilase variants |
| WO2007087508A2 (en) | 2006-01-23 | 2007-08-02 | Novozymes A/S | Lipase variants |
| WO2009067279A1 (en) | 2007-11-21 | 2009-05-28 | E.I. Du Pont De Nemours And Company | Production of peracids using an enzyme having perhydrolysis activity |
| US20090209447A1 (en) * | 2008-02-15 | 2009-08-20 | Michelle Meek | Cleaning compositions |
| WO2009109500A1 (en) | 2008-02-29 | 2009-09-11 | Novozymes A/S | Polypeptides having lipase activity and polynucleotides encoding same |
| WO2009112994A1 (en) | 2008-03-14 | 2009-09-17 | The Procter & Gamble Company | Automatic dishwashing detergent composition |
| WO2010065455A2 (en) | 2008-12-01 | 2010-06-10 | Danisco Us Inc. | Enzymes with lipase activity |
| WO2010100028A2 (en) | 2009-03-06 | 2010-09-10 | Huntsman Advanced Materials (Switzerland) Gmbh | Enzymatic textile bleach-whitening methods |
| WO2010107560A2 (en) | 2009-03-18 | 2010-09-23 | Danisco Us Inc. | Fungal cutinase from magnaporthe grisea |
| WO2010111143A2 (en) | 2009-03-23 | 2010-09-30 | Danisco Us Inc. | Cal a-related acyltransferases and methods of use, thereof |
| WO2011036263A1 (en) | 2009-09-25 | 2011-03-31 | Novozymes A/S | Subtilase variants |
| WO2011036264A1 (en) | 2009-09-25 | 2011-03-31 | Novozymes A/S | Use of protease variants |
| WO2011072017A2 (en) | 2009-12-10 | 2011-06-16 | The Procter & Gamble Company | Detergent composition |
| WO2011072099A2 (en) | 2009-12-09 | 2011-06-16 | Danisco Us Inc. | Compositions and methods comprising protease variants |
| WO2011084599A1 (en) | 2009-12-21 | 2011-07-14 | Danisco Us Inc. | Detergent compositions containing bacillus subtilis lipase and methods of use thereof |
| WO2011084417A1 (en) | 2009-12-21 | 2011-07-14 | Danisco Us Inc. | Detergent compositions containing geobacillus stearothermophilus lipase and methods of use thereof |
| WO2011084412A1 (en) | 2009-12-21 | 2011-07-14 | Danisco Us Inc. | Detergent compositions containing thermobifida fusca lipase and methods of use thereof |
| WO2011150157A2 (en) | 2010-05-28 | 2011-12-01 | Danisco Us Inc. | Detergent compositions containing streptomyces griseus lipase and methods of use thereof |
| WO2012137147A1 (en) | 2011-04-08 | 2012-10-11 | Danisco Us, Inc. | Compositions |
| WO2016058872A1 (en) * | 2014-10-17 | 2016-04-21 | Basf Se | Container comprising a detergent composition containing glda |
| EP2794836B1 (en) * | 2011-12-22 | 2016-06-01 | Unilever N.V. | Detergent composition comprising glutamic-n,n-diacetate, water and bleaching agent |
| WO2016096711A2 (en) | 2014-12-15 | 2016-06-23 | Novozymes A/S | Subtilase variants |
| JP6474992B2 (en) | 2014-10-21 | 2019-02-27 | 浜松ホトニクス株式会社 | Photodetection unit |
-
2019
- 2019-08-06 WO PCT/EP2019/071083 patent/WO2020030623A1/en not_active Ceased
Patent Citations (82)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1243784A (en) | 1967-10-03 | 1971-08-25 | Novo Terapeutisk Labor As | Proteolytic enzymes, their production and use |
| EP0218272A1 (en) | 1985-08-09 | 1987-04-15 | Gist-Brocades N.V. | Novel lipolytic enzymes and their use in detergent compositions |
| EP0258068A2 (en) | 1986-08-29 | 1988-03-02 | Novo Nordisk A/S | Enzymatic detergent additive |
| EP0260105A2 (en) | 1986-09-09 | 1988-03-16 | Genencor, Inc. | Preparation of enzymes having altered activity |
| US5389536A (en) | 1986-11-19 | 1995-02-14 | Genencor, Inc. | Lipase from Pseudomonas mendocina having cutinase activity |
| EP0283075A2 (en) | 1987-02-27 | 1988-09-21 | Gist-Brocades N.V. | Molecular cloning and expression of genes encoding proteolytic enzymes |
| WO1988009367A1 (en) | 1987-05-29 | 1988-12-01 | Genencor, Inc. | Cutinase cleaning composition |
| EP0305216A1 (en) | 1987-08-28 | 1989-03-01 | Novo Nordisk A/S | Recombinant Humicola lipase and process for the production of recombinant humicola lipases |
| WO1989006279A1 (en) | 1988-01-07 | 1989-07-13 | Novo-Nordisk A/S | Mutated subtilisin genes |
| WO1989006276A2 (en) | 1988-01-08 | 1989-07-13 | Dpz Deutsches Primatenzentrum Gesellschaft Mbh | Hiv-2-type retroviruses of primates, vaccines, diagnostic and pharmaceutical compositions |
| EP0331376A2 (en) | 1988-02-28 | 1989-09-06 | Amano Pharmaceutical Co., Ltd. | Recombinant DNA, bacterium of the genus pseudomonas containing it, and process for preparing lipase by using it |
| WO1989009830A1 (en) | 1988-04-12 | 1989-10-19 | Genex Corporation | Subtilisin mutations |
| WO1989009819A1 (en) | 1988-04-12 | 1989-10-19 | Genex Corporation | Combining mutations for stabilization of subtilisin |
| WO1990009446A1 (en) | 1989-02-17 | 1990-08-23 | Plant Genetic Systems N.V. | Cutinase |
| EP0407225A1 (en) | 1989-07-07 | 1991-01-09 | Unilever Plc | Enzymes and enzymatic detergent compositions |
| US5352604A (en) | 1989-08-25 | 1994-10-04 | Henkel Research Corporation | Alkaline proteolytic enzyme and method of production |
| WO1991002792A1 (en) | 1989-08-25 | 1991-03-07 | Henkel Research Corporation | Alkaline proteolytic enzyme and method of production |
| WO1991006637A1 (en) | 1989-10-31 | 1991-05-16 | Genencor International, Inc. | Subtilisin mutants |
| WO1991016422A1 (en) | 1990-04-14 | 1991-10-31 | Kali-Chemie Aktiengesellschaft | Alkaline bacillus lipases, coding dna sequences therefor and bacilli which produce these lipases |
| WO1992005249A1 (en) | 1990-09-13 | 1992-04-02 | Novo Nordisk A/S | Lipase variants |
| WO1992019729A1 (en) | 1991-05-01 | 1992-11-12 | Novo Nordisk A/S | Stabilized enzymes and detergent compositions |
| WO1994001541A1 (en) | 1992-07-06 | 1994-01-20 | Novo Nordisk A/S | C. antarctica lipase and lipase variants |
| WO1994025578A1 (en) | 1993-04-27 | 1994-11-10 | Gist-Brocades N.V. | New lipase variants for use in detergent applications |
| WO1995006720A1 (en) | 1993-08-30 | 1995-03-09 | Showa Denko K.K. | Novel lipase, microorganism producing the lipase, process for producing the lipase, and use of the lipase |
| WO1995014783A1 (en) | 1993-11-24 | 1995-06-01 | Showa Denko K.K. | Lipase gene and variant lipase |
| WO1995022615A1 (en) | 1994-02-22 | 1995-08-24 | Novo Nordisk A/S | A method of preparing a variant of a lipolytic enzyme |
| WO1995023221A1 (en) | 1994-02-24 | 1995-08-31 | Cognis, Inc. | Improved enzymes and detergents containing them |
| EP1921147A2 (en) | 1994-02-24 | 2008-05-14 | Henkel Kommanditgesellschaft auf Aktien | Improved enzymes and detergents containing them |
| WO1995030744A2 (en) | 1994-05-04 | 1995-11-16 | Genencor International Inc. | Lipases with improved surfactant resistance |
| WO1995035381A1 (en) | 1994-06-20 | 1995-12-28 | Unilever N.V. | Modified pseudomonas lipases and their use |
| WO1996000292A1 (en) | 1994-06-23 | 1996-01-04 | Unilever N.V. | Modified pseudomonas lipases and their use |
| WO1996012012A1 (en) | 1994-10-14 | 1996-04-25 | Solvay S.A. | Lipase, microorganism producing same, method for preparing said lipase and uses thereof |
| WO1996013580A1 (en) | 1994-10-26 | 1996-05-09 | Novo Nordisk A/S | An enzyme with lipolytic activity |
| WO1996027002A1 (en) | 1995-02-27 | 1996-09-06 | Novo Nordisk A/S | Novel lipase gene and process for the production of lipase with the use of the same |
| WO1996034946A1 (en) | 1995-05-05 | 1996-11-07 | Novo Nordisk A/S | Protease variants and compositions |
| WO1997004079A1 (en) | 1995-07-14 | 1997-02-06 | Novo Nordisk A/S | A modified enzyme with lipolytic activity |
| WO1997007202A1 (en) | 1995-08-11 | 1997-02-27 | Novo Nordisk A/S | Novel lipolytic enzymes |
| WO1998020115A1 (en) | 1996-11-04 | 1998-05-14 | Novo Nordisk A/S | Subtilase variants and compositions |
| WO1998020116A1 (en) | 1996-11-04 | 1998-05-14 | Novo Nordisk A/S | Subtilase variants and compositions |
| EP0851023A2 (en) | 1996-12-23 | 1998-07-01 | Unilever N.V. | Machine dishwashing tablets containing a peracid |
| WO1999011768A1 (en) | 1997-08-29 | 1999-03-11 | Novo Nordisk A/S | Protease variants and compositions |
| DE19819187A1 (en) | 1998-04-30 | 1999-11-11 | Henkel Kgaa | Solid dishwasher detergent with phosphate and crystalline layered silicates |
| WO2000034450A1 (en) | 1998-12-04 | 2000-06-15 | Novozymes A/S | Cutinase variants |
| WO2000060063A1 (en) | 1999-03-31 | 2000-10-12 | Novozymes A/S | Lipase variant |
| WO2001044452A1 (en) | 1999-12-15 | 2001-06-21 | Novozymes A/S | Subtilase variants having an improved wash performance on egg stains |
| WO2001092502A1 (en) | 2000-06-02 | 2001-12-06 | Novozymes A/S | Cutinase variants |
| WO2002042400A2 (en) | 2000-11-27 | 2002-05-30 | The Procter & Gamble Company | Dishwashing method |
| DE10064983A1 (en) | 2000-12-23 | 2002-07-18 | Henkel Kgaa | New subtilisin protease from Bacillus alcalophilus, useful e.g. in washing and cleaning compositions, comprises at positions 230, 256 and 259 amino acids Val, Gly and Asn |
| WO2002088340A2 (en) | 2001-05-02 | 2002-11-07 | Henkel Kommanditgesellschaft Auf Aktien | Alkaline protease variants and detergents and cleaning agents containing said alkaline protease variants |
| WO2003006602A2 (en) | 2001-07-12 | 2003-01-23 | Novozymes A/S | Subtilase variants |
| WO2003054184A1 (en) | 2001-12-20 | 2003-07-03 | Henkel Kommanditgesellschaft Auf Aktien | Novel alkali protease formed by bacillus gibsonii (dsm 14393) and washing and cleaning agents containing said novel alkali protease |
| WO2003055974A2 (en) | 2001-12-22 | 2003-07-10 | Henkel Kommanditgesellschaft Auf Aktien | Novel alkaline protease from bacillus sp. (dsm 14392) and washing and cleaning products comprising said novel alkaline protease |
| WO2003056017A2 (en) | 2001-12-22 | 2003-07-10 | Henkel Kommanditgesellschaft Auf Aktien | Alkaline protease from bacillus sp. (dsm 14390) and washing and cleaning products comprising said alkaline protease |
| WO2004003186A2 (en) | 2002-06-26 | 2004-01-08 | Novozymes A/S | Subtilases and subtilase variants having altered immunogenicity |
| WO2004041979A2 (en) | 2002-11-06 | 2004-05-21 | Novozymes A/S | Subtilase variants |
| WO2005056782A2 (en) | 2003-12-03 | 2005-06-23 | Genencor International, Inc. | Perhydrolase |
| WO2005063974A1 (en) | 2003-12-23 | 2005-07-14 | Henkel Kommanditgesellschaft Auf Aktien | Novel alkaline protease and washing and cleaning products containing said novel alkaline protease |
| WO2005103244A1 (en) | 2004-04-23 | 2005-11-03 | Henkel Kommanditgesellschaft Auf Aktien | Novel alkaline proteases, and detergents and cleaners containing the same |
| DE102005028295A1 (en) | 2005-06-18 | 2006-11-16 | Henkel Kgaa | New polynucleotide encoding a protease active at low temperature, for use e.g. in cleaning, pharmaceutical and cosmetic compositions and for eliminating biofilms |
| WO2007006305A1 (en) | 2005-07-08 | 2007-01-18 | Novozymes A/S | Subtilase variants |
| WO2007087508A2 (en) | 2006-01-23 | 2007-08-02 | Novozymes A/S | Lipase variants |
| WO2009067279A1 (en) | 2007-11-21 | 2009-05-28 | E.I. Du Pont De Nemours And Company | Production of peracids using an enzyme having perhydrolysis activity |
| US20090209447A1 (en) * | 2008-02-15 | 2009-08-20 | Michelle Meek | Cleaning compositions |
| WO2009109500A1 (en) | 2008-02-29 | 2009-09-11 | Novozymes A/S | Polypeptides having lipase activity and polynucleotides encoding same |
| WO2009112994A1 (en) | 2008-03-14 | 2009-09-17 | The Procter & Gamble Company | Automatic dishwashing detergent composition |
| WO2010065455A2 (en) | 2008-12-01 | 2010-06-10 | Danisco Us Inc. | Enzymes with lipase activity |
| WO2010100028A2 (en) | 2009-03-06 | 2010-09-10 | Huntsman Advanced Materials (Switzerland) Gmbh | Enzymatic textile bleach-whitening methods |
| WO2010107560A2 (en) | 2009-03-18 | 2010-09-23 | Danisco Us Inc. | Fungal cutinase from magnaporthe grisea |
| WO2010111143A2 (en) | 2009-03-23 | 2010-09-30 | Danisco Us Inc. | Cal a-related acyltransferases and methods of use, thereof |
| WO2011036263A1 (en) | 2009-09-25 | 2011-03-31 | Novozymes A/S | Subtilase variants |
| WO2011036264A1 (en) | 2009-09-25 | 2011-03-31 | Novozymes A/S | Use of protease variants |
| WO2011072099A2 (en) | 2009-12-09 | 2011-06-16 | Danisco Us Inc. | Compositions and methods comprising protease variants |
| WO2011072017A2 (en) | 2009-12-10 | 2011-06-16 | The Procter & Gamble Company | Detergent composition |
| WO2011084599A1 (en) | 2009-12-21 | 2011-07-14 | Danisco Us Inc. | Detergent compositions containing bacillus subtilis lipase and methods of use thereof |
| WO2011084417A1 (en) | 2009-12-21 | 2011-07-14 | Danisco Us Inc. | Detergent compositions containing geobacillus stearothermophilus lipase and methods of use thereof |
| WO2011084412A1 (en) | 2009-12-21 | 2011-07-14 | Danisco Us Inc. | Detergent compositions containing thermobifida fusca lipase and methods of use thereof |
| WO2011150157A2 (en) | 2010-05-28 | 2011-12-01 | Danisco Us Inc. | Detergent compositions containing streptomyces griseus lipase and methods of use thereof |
| WO2012137147A1 (en) | 2011-04-08 | 2012-10-11 | Danisco Us, Inc. | Compositions |
| EP2794836B1 (en) * | 2011-12-22 | 2016-06-01 | Unilever N.V. | Detergent composition comprising glutamic-n,n-diacetate, water and bleaching agent |
| WO2016058872A1 (en) * | 2014-10-17 | 2016-04-21 | Basf Se | Container comprising a detergent composition containing glda |
| JP6474992B2 (en) | 2014-10-21 | 2019-02-27 | 浜松ホトニクス株式会社 | Photodetection unit |
| WO2016096711A2 (en) | 2014-12-15 | 2016-06-23 | Novozymes A/S | Subtilase variants |
Non-Patent Citations (12)
| Title |
|---|
| DARTOIS ET AL., BIOCHEMICA ET BIOPHYSICA ACTA, vol. 1131, 1992, pages 253 - 360 |
| DELMAR ET AL., ANALYTICAL BIOCHEM, vol. 99, 1979, pages 316 - 320 |
| EL SMITH ET AL., J. BIOL CHEM, vol. 243, 1968, pages 2184 - 2191 |
| GUPTA ET AL., APPL. MICROBIOL. BIOTECHNOL., vol. 60, 2002, pages 381 - 395 |
| GUPTA ET AL., BIOTECHNOL. APPL. BIOCHEM., vol. 37, 2003, pages 63 - 71 |
| JA WELLS ET AL., NUCLEIC ACIDS RESEARCH, vol. 11, 1983, pages 7911 - 7925 |
| JACOBS ET AL., NUCL. ACIDS RES, vol. 13, 1985, pages 8913 - 8926 |
| NEEDLEMANWUNSCH, J. MOL. BIOL., vol. 48, 1979, pages 443 - 453 |
| SIEZEN ET AL., PROTEIN ENG., vol. 4, 1991, pages 719 - 737 |
| SIEZEN ET AL., PROTEIN SCIENCE, vol. 6, 1997, pages 501 - 523 |
| SIEZEN ET AL., PROTEIN SCIENCE, vol. 6, September 1997 (1997-09-01), pages 501 - 523 |
| VASANTHA ET AL., J. BACTERIOL., vol. 159, 1984, pages 811 - 819 |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12275919B2 (en) | 2019-04-01 | 2025-04-15 | Basf Se | Process for making a granule or powder |
| KR20240015655A (en) | 2021-05-31 | 2024-02-05 | 다우 도레이 캄파니 리미티드 | Display device, especially haptic display device and method for designing the same |
| WO2024151575A1 (en) * | 2023-01-09 | 2024-07-18 | The Procter & Gamble Company | Superposed multi-sectioned water-soluble unit dose automatic dishwashing detergent pouch |
| WO2024151572A1 (en) * | 2023-01-09 | 2024-07-18 | The Procter & Gamble Company | Superposed multi-sectioned water-soluble unit dose automatic dishwashing detergent pouch |
| WO2024151574A1 (en) * | 2023-01-09 | 2024-07-18 | The Procter & Gamble Company | Superposed multi-sectioned water-soluble unit dose automatic dishwashing detergent pouch |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR20170061687A (en) | Detergent composition | |
| US10093888B2 (en) | Solid dishwashing detergent with improved protease performance | |
| CA3043443A1 (en) | Stabilization of enzymes in compositions | |
| US11104867B2 (en) | Dishwashing liquid having bleaching catalyst and protease | |
| CA3033062A1 (en) | Liquid laundry formulation | |
| EP3167036B1 (en) | Co-granulate of enzyme and bleach catalyst | |
| CN115605568B (en) | High alkaline textile detergent containing protease | |
| WO2020104231A1 (en) | Powders and granules containing a chelating agent and an enzyme | |
| WO2016100320A1 (en) | Detergent composition | |
| WO2020030623A1 (en) | Packaging unit comprising a detergent composition containing an enzyme and at least one chelating agent | |
| US11220660B2 (en) | Portion bag having bleach activator/complexing agent compound | |
| US20170073619A1 (en) | Dishwashing Composition | |
| CA2969465C (en) | Low ph automatic dishwashing detergent composition | |
| WO2017105857A1 (en) | Automatic dishwashing detergent composition | |
| CN107683326B (en) | Manganese bleach catalyst/enzyme granules for dishwashing detergents | |
| CA3085345C (en) | Automatic dishwashing detergent composition | |
| US20220251475A1 (en) | Dishwashing agent with bleaching catalyst and bacillus gibsonii protease | |
| CA3085344A1 (en) | Phosphate-free automatic dishwashing cleaning composition, and its use | |
| CA3084144A1 (en) | Automatic dishwashing detergent composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| DPE2 | Request for preliminary examination filed before expiration of 19th month from priority date (pct application filed from 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19748547 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 19748547 Country of ref document: EP Kind code of ref document: A1 |