WO2020012435A1 - Hiv binding agents - Google Patents
Hiv binding agents Download PDFInfo
- Publication number
- WO2020012435A1 WO2020012435A1 PCT/IB2019/055966 IB2019055966W WO2020012435A1 WO 2020012435 A1 WO2020012435 A1 WO 2020012435A1 IB 2019055966 W IB2019055966 W IB 2019055966W WO 2020012435 A1 WO2020012435 A1 WO 2020012435A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- antibody
- binding agent
- hiv
- seq
- human
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C07K16/1145—
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
- A61P31/18—Antivirals for RNA viruses for HIV
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/21—Immunoglobulins specific features characterized by taxonomic origin from primates, e.g. man
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
Definitions
- This disclosure relates to binding agents with specificity for human immunodeficiency virus (HIV), methods for making the same, and to methods for using the same to treat and / or prevent HIV infection.
- HIV human immunodeficiency virus
- Figure 1 shows the results of neutralization of a panel of nine (9) HIV-1 pseudoviruses from the Global Panel of HIV-1 reference strains by 107 plasma samples from chronically infected patients naive to antiretroviral therapy. MLV pseudovirus is used as a negative control. Boxed are the eight donors selected for the collection of lymph nodes to isolate potent broadly neutralizing antibodies. Highlighted with the arrow is donor SA090 who was selected for the isolation of the broadly neutralizing antibody described herein. Inhibitory dose values indicate the dilution of plasma capable of neutralizing 50% of viral infection (ID50).
- Figure 2 shows the gating and sorting strategy used to purify memory and germinal center IgG B cells from lymph node samples.
- B cells were selected for the expression of the surface marker CD19 and IgG B cells were negatively selected for the lack of IgA and IgM B cell receptor (BCR) expression.
- Germinal center B cells were further selected for the expression of the CD38 marker (that is absent on memory B cells).
- Figure 3 shows the results of neutralization of a panel of nine (9) HIV-1 pseudoviruses (and MLV as negative control) from the Global Panel of HIV-1 reference strains by different concentration (in pg/ml) of the monoclonal antibody LN02.
- Inhibitory concentration values indicate the concentration of monoclonal antibody capable of neutralizing 50% of viral infection (IC50). Error bars indicate the standard deviation of duplicates.
- Figure 4 shows the results of neutralization of a multi-clade panel of 57 HIV-1 pseudoviruses by the monoclonal antibody LN02. IC50 values indicate the concentration of monoclonal antibody capable of neutralizing 50% of viral infection
- Figure 5 shows the distribution of IC50 values on the whole panel of 57 viruses described in Figure 4 and on individual clades or circulating recombinant forms.
- Figure 6 shows that the neutralization activity of LN02 is enhanced by cleavage site mutation E500K and a combination of all cleavage site mutations together (CS). IC50 values and fold change in neutralization activity relative to the WT CAP45 pseudo-virus is shown in
- Figure 7 shows the binding, as assessed by surface plasmon resonance, of the LN02 monoclonal antibody to the soluble HIV-1 Env trimer 426c WT SOSIP gp140 that expresses multiple epitopes for broadly neutralizing antibodies (panel A, solid line). HIV-1 Env trimer that has been pre-incubated with unlabeled LN02 completely blocks the binding of a biotinylated LN02 that is detected with a streptavidin probe (panel A, dashed line).
- the PGT151 monoclonal antibody (binding to a site at the interface between gp120 and gp41) bound with high affinity to 426c WT SOSIP gp140 protein (panel B, solid line) but PGT151 binding is significantly reduced to the LN02 + 426c WT SOSIPgp140 complex.
- Figure 8 shows the binding, as measured by ELISA, of LN02 antibody to a set of HIV-1 Env antigens and a negative control antigen (soluble CD4 protein, sCD4).
- LN02 only binds tightly to the gp140 ConsB, which is the only protein construct that is predominantly trimeric in tertiary structure.
- Figure 9 is a comparison of the amino acid sequences of the HXB2 (SEQ ID NO.: 18) and CAP45 (SEQ ID NO. 17) Env polypeptides.
- This disclosure relates to binding agents with specificity for human immunodeficiency virus (HIV), methods for producing such binding agents, as well as methods for using such binding agents to treat, prevent and / or ameliorate HIV infection.
- HIV human immunodeficiency virus
- binding agents having binding affinity for human immunodeficiency virus (HIV).
- the binding agent can bind HIV antigens on viral particles per se or on the surface of cells in vitro and / or in vivo.
- the binding agents may also bind isolated HIV antigens and / or fragments and / or derivatives thereof, typically in vitro.
- methods for using such binding agents to diagnose, treat, prevent and / or ameliorate one or more diseases associated with HIV.
- the binding agents may be antibodies (e.g., monoclonal antibodies) that may react with and / or bind to the epitopes of HIV or polypeptides thereof.
- the binding agents may be useful for treating disease caused by HIV, such as Acquired Immune Deficiency Syndrome (AIDS).
- the binding agents described herein may selectively target and/or eliminate HIV and/or HIV-infected cells containing HIV (e.g., replication competent HIV) and/or expressing proteins thereof. In some embodiments, such cells may be reservoirs for replication competent HIV.
- binding agents having, for instance, different specificities (e.g., recognizing different epitopes) may be combined to HIV activity such as infection, replication and/or spread to other cells.
- the binding agents described herein may also provide for the selective elimination and / or suppression of HIV or HIV-expressing cells.
- the binding agents described herein may be used to suppress and / or eliminate HIV and/or HIV-expressing cells to treat, for instance, HIV infection and/or AIDS. Other embodiments, uses and the like are described below.
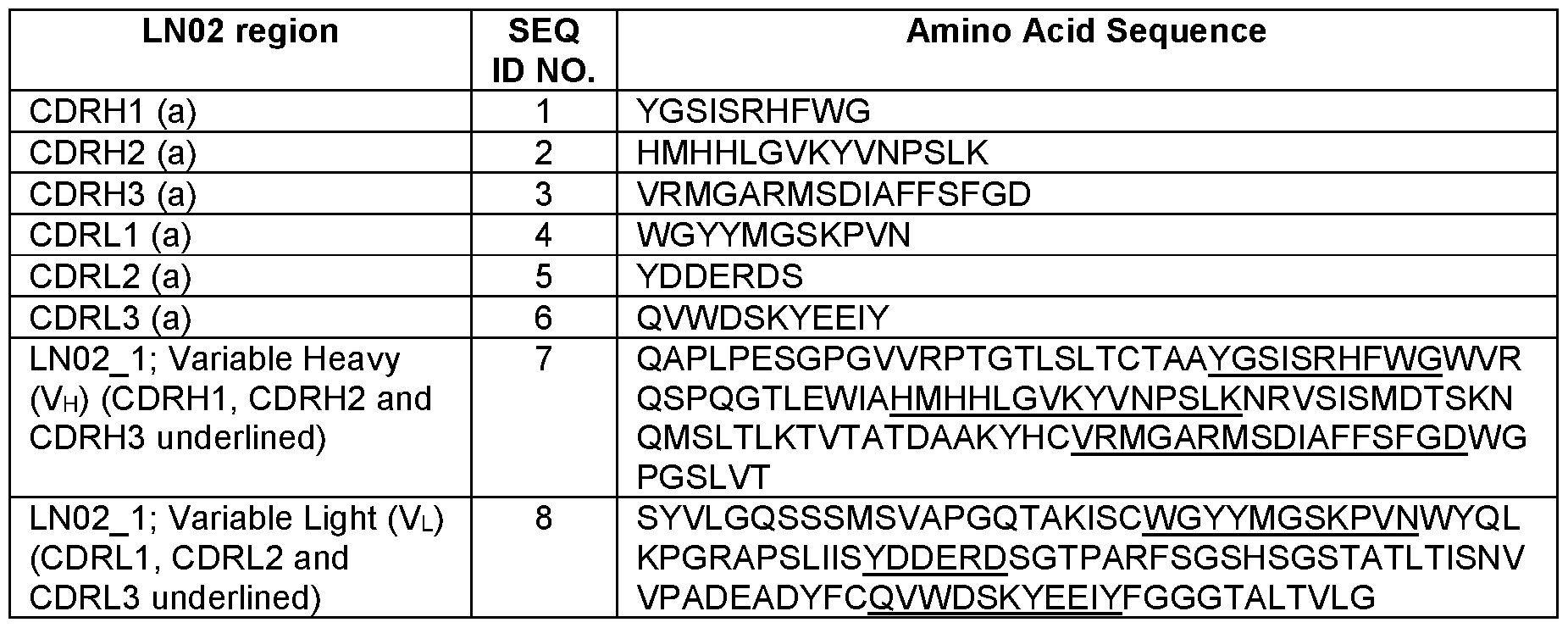
- the binding agents may be antibodies such as monoclonal antibodies. As shown in the examples herein, the techniques discussed below have been used to identify a fully human mAb termed“LN02”, having particular characteristics that are described herein and shown in the examples. The LN02 antibody was isolated and the amino acid sequences of variable heavy (V H ) and light (V L ) chain domains of said antibody determined. A binding agent such as LN02 may identified by referencing the amino acid and/or nucleic acid sequences corresponding to the variability and / or complementarity determining regions (“CDRs”) thereof.
- CDRs complementarity determining regions
- a CDR comprises amino acid residues within the variable region identified in accordance with the definitions of the Kabat, Chothia, the accumulation of both Kabat and Chothia, AbM, contact, and/or conformational definitions or any method of CDR determination well known in the art.
- antibody modeling software now Accelrys®
- the“contact definition” of CDRs based on observed antigen contacts described by MacCallum et al. , 1996, J. Mol. Biol., 262:732-745.
- the positions of the CDRs may be identified as the residues that make enthalpic contributions to antigen binding (Makabe et al., 2008, Journal of Biological Chemistry, 283:1 156-1166).
- CDR boundary definitions may not strictly follow one of the above approaches, but may nonetheless overlap with at least a portion of the Kabat CDRs, although they may be shortened or lengthened in light of prediction or experimental findings that particular residues or groups of residues or even entire CDRs do not significantly impact antigen binding.
- a CDR may refer to CDRs defined by any approach known in the art, including combinations of approaches. The methods used herein may utilize CDRs defined according to any of these approaches. For any given embodiment containing more than one CDR, the CDRs may be defined in accordance with any of Kabat, Chothia, extended, AbM, contact, and/or conformational definitions.
- a binding agent of this disclosure may comprise, for example, any one or more of the amino acid sequences shown in Table 1 (i.e. , any one or more of SEQ ID NOS. 1-8), and/or or a conservatively substituted variant thereof. Fragments and/or derivatives (e.g., comprising substituted amino acids, such as conservative substitutions) thereof are also disclosed.
- a binding agent of this disclosure may comprise one or more (i.e., one, two, three, four, five, six or seven) of SEQ ID NOS. 1-8, provided the binding agent exhibits the functional characteristics described herein.
- the binding agent comprises at least one of SEQ ID NO. 1 (CDRH1 region); at least one of SEQ ID NO.
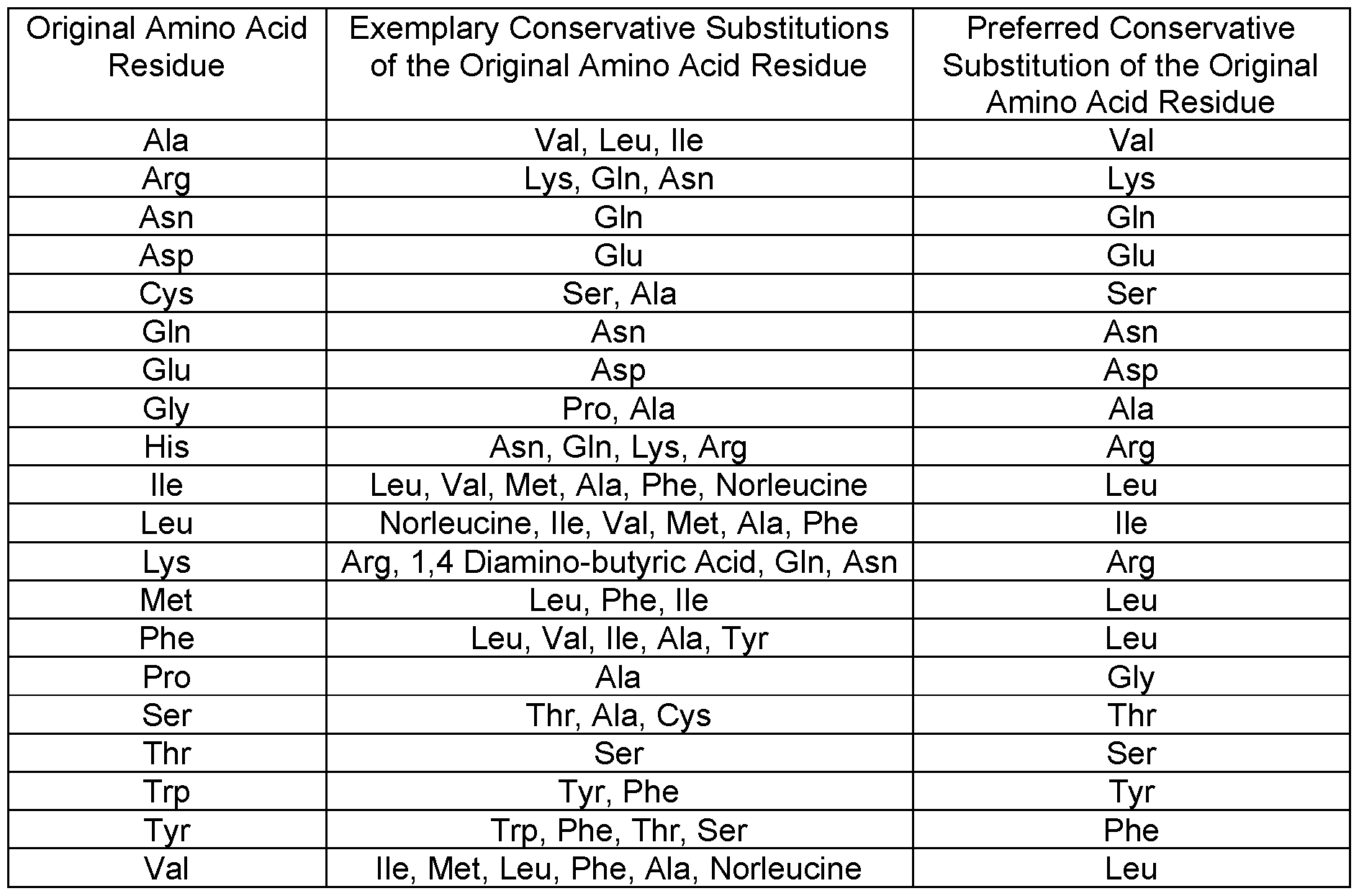
- the binding agent may comprise any of SEQ ID NOS. 1-8 comprising one or more amino acid substitutions, in particular or preferably conservative substitutions (see, e.g., the substitutions listed in Table 2).
- the binding agent may be a monoclonal antibody (mAb) or a fragment or derivative thereof.
- the binding agent may be an HIV-binding fragment of such a monoclonal antibody (mAb).
- the mAb, fragment or derivative thereof includes at least one or more of SEQ ID NOS. 1 (CDRH1 region), SEQ ID NO. 2 (CDRH2 region); SEQ ID NO. 3 (CDRH3 region); SEQ ID NO. 4 (CDRL1 region); SEQ ID NO. 5 (CDRL2 region); and/or, SEQ ID NO. 6 (CDRL3 region); and/or any one or more of the same comprising one or more amino acid substitutions, in particular or preferably conservatively substituted derivatives thereof (see, e.g., the substitutions listed in Table 2).
- LN02 mAb is termed“lgG1 LN02” in which the LN02 variable regions (e.g., SEQ ID NOS. 7 and 8) were cloned into an lgG1 or lgG3 backbone using standard techniques.
- the binding agent comprises SEQ ID NO. 7 and/or 8, preferably SEQ ID NOS. 7 and 8; as well as any of the same comprising one or more amino acid substitutions, in particular or preferably conservative substitutions (see, e.g., the substitutions listed in Table 2).
- SEQ ID NO. 7 and/or 8 preferably SEQ ID NOS. 7 and 8
- any of the same comprising one or more amino acid substitutions, in particular or preferably conservative substitutions (see, e.g., the substitutions listed in Table 2).
- Other suitable embodiments may be derived by those of ordinary skill in the art from this disclosure.
- the binding agent e.g., antibody, or the antigen binding fragment thereof
- identities of less than 100% may result from the natural or synthetic substitution of one or more amino acids with another amino acid(s), as in a conservative substitution (see, e.g., Table 2).
- the binding agent binds HIV and/or cells infected by HIV and/or expressing HIV proteins. In some especially preferred embodiments, the binding agent neutralizes HIV as described herein. In preferred embodiments, the binding agent both binds HIV and/or cells infected by HIV and/or expressing HIV proteins and neutralizes HIV.
- variable region and/or CDR sequences may be used in combination with one or more other variable region / CDR amino acid sequences available to those of ordinary skill in the art.
- Such variable region / CDR amino acid sequences may alternatively and / or also be adjoined to one or more types of constant region polypeptides of an antibody molecule.
- the CDR amino acid sequences shown in Table 1 may be adjoined to or associated with the constant regions of any antibody molecule of the same or a different species (e.g., human, goat, rat, sheep, chicken) and / or antibody subtype of that from which the CDR amino acid sequence was derived.
- an exemplary binding agent may be, or may be derived from, one having about the same neutralizing activity and/or binding the same or similar epitopes and/or exhibiting about the same affinity as another binding agent comprising one or more of the amino acid sequences shown in Table 1.
- the binding agent may comprise an antibody heavy and / or a light chain that each comprises one or more constant and / or variable regions.
- any of the amino acid sequences described herein may also be combined with any other variable region and / or CDR in any order and / or combination to form new binding agents, e.g., hybrid and / or fusion binding agents, and / or inserted into other heavy and / or light chain variable regions using standard techniques.
- binding agents to isolate, identify, and / or target HIV and / or cells harboring and/or infected by HIV and / or expressing HIV antigens.
- binding agents may be reactive against HIV antigens such as proteins expressed on the surface of cells.
- the binding agent(s) is an antibody (antibodies).
- antibody or“antibodies” may refer to whole or fragmented antibodies in unpurified or partially purified form (e.g., hybridoma supernatant, ascites, polyclonal antisera) or in purified form.
- the antibodies may be of any suitable origin or form including, for example, murine (e.g., produced by murine hybridoma cells), or expressed as humanized antibodies, chimeric antibodies, human antibodies, and the like.
- antibodies may be wholly or partially dervied from human (e.g., IgG (lgG1 , lgG2, lgG2a, Ig2b, lgG3, lgG4), IgM, IgA (lgA1 and lgA2), IgD, and IgE), canine (e.g., IgGA, IgGB, IgGC, IgGD), chicken (e.g., IgA, IgD, IgE, IgG, IgM, IgY), goat (e.g., IgG), mouse (e.g., IgG, IgD, IgE, IgG, IgM), pig (e.g., IgG, IgD, IgE, IgG
- the antibodies may be contained within hybridoma supernatant or ascites and utilized either directly as such or following concentration using standard techniques.
- the antibodies may be further purified using, for example, salt fractionation and ion exchange chromatography, or affinity chromatography using Protein A, Protein G, Protein A/G, and / or Protein L ligands covalently coupled to a solid support such as agarose beads, or combinations of these techniques.
- the antibodies may be stored in any suitable format, including as a frozen preparation (e.g., -20°C or -70°C), in lyophilized form, or under normal refrigeration conditions (e.g., 4°C).
- the binding agent When stored in liquid form, for instance, it is preferred that a suitable buffer such as Tris-buffered saline (TBS) or phosphate buffered saline (PBS) is utilized.
- TBS Tris-buffered saline
- PBS phosphate buffered saline
- the binding agent may be prepared as an injectable preparation, such as in suspension in a non-toxic parenterally acceptable diluent or solvent.
- Suitable vehicles and solvents that may be utilized include water, Ringer's solution, and isotonic sodium chloride solution, TBS and / or PBS, among others.
- Such preparations may be suitable for use in vitro or in vivo may be prepared as is known in the art and the exact preparation may depend on the particular application.
- the binding agents described herein are not, however, in any way limited to antibodies (i.e., whole antibodies).
- the binding agent may be any compound exhibiting similar binding properties as another (e.g., a mimetic).
- an exemplary binding agent may be one that binds HIV and/or can compete with another binding agent having specificity therefor (e.g., a monoclonal antibody such as LN02).
- the mimetic may exhibit substantially the same affinity in binding assays as the binding agent (e.g., monoclonal antibody) to which it is being compared.
- the affinity of a particular binding agent may be measured by any suitable assay including but not limited to FACS staining of cell surface HIV antigens (e.g., polypeptides).
- One binding agent may be said to have “substantially the same affinity” as another where the measurements (e.g., nm) are within about any of 1-20, 1-5, 5-10, 10-15, or 15-20 percent of one another.
- Exemplary mimetics may include, for example, organic compounds that specifically bind HIV, or an affibody (Nygren, et al. FEBS J. 275 (11): 2668-76 (2008)), affilin (Ebersbach, et al. J. Mol. Biol.
- Other mimetics may include, for example, a derivative of an antibody such as, for example, an F a b, F a b2, Fab’ single chain antibody, F v , single domain antibody, mono-specific antibody, bi-specific antibody, tri-specific antibody, multi-valent antibody, chimeric antibody, canine-human chimeric antibody, canine-mouse chimeric antibody, antibody comprising a canine Fc, humanized antibody, human antibody, caninized, CDR-grafted antibody (i.e., comprising any of SEQ ID NOS. 1-8 shown in Table 1), shark antibody, nanobody, camelid antibody, microbody, and / or intrabody; and/or derivative thereof.
- Other binding agents are also provided herein as would be understood by one of ordinary skill in the art.
- binding agents having specificity for (e.g., binding to) HIV Any method known to those of ordinary skill in the art may be used to generate binding agents having specificity for (e.g., binding to) HIV.
- an animal such as a mouse may be administered (e.g., immunized) with one or more HIV proteins. Animals exhibiting serum reactivity to HIV expressed on activated human T lymphocytes (as determined by, for instance, flow cytometry and / or microscopy) may then be selected for generation of anti-HIV hybridoma cell lines. This may be repeated for multiple rounds. Screening may also include, for instance, affinity binding and / or functional characterization to identify the binding agent as being specific for HIV.
- human beings may be screened for the expression of antibodies against HIV.
- plasma samples of human beings infected by HIV may be screened to identify persons expressing anti-HIV antibodies, and in particular, neutralizing antibodies.
- Neutralizing antibody-producing cells of such persons may then be isolated, followed by the isolation and characterization of the antibodies produced thereby (e.g., as in the examples herein).
- a neutralizing antibody may be one that exhibits the ability to neutralize, or inhibit, infection of cells by HIV.
- a neutralization assay typically measures the loss of infectivity of the virus through reaction of the virus with specific antibodies.
- a loss of infectivity is caused by interference by the bound antibody with any of the virus replication steps including but not limited to binding to target cells, entry, and/or viral release.
- the presence of un-neutralized virus is detected after a predetermined amount of time, e.g., one, two, three, four, five, six, seven, eight, nine, 10, 12 or 14 days, by measuring the infection of target cells using any of the systems available to those of ordinary skill in the art (e.g., a luciferase-based system).
- a non-limiting example of a neutralization assay may include combining a given amount of a virus or pseudovirus (see below) and different concentrations of the test or control (typically positive and negative controls assayed separately) antibody or antibodies are mixed under appropriate conditions (e.g., one (1) hour at room temperature) and then inoculated into an appropriate target cell culture (e.g., TZM-bl cells).
- a target cell culture e.g., TZM-bl cells
- binding agent-producing cells may be assayed for the production of HIV-1 neutralizing antibodies by seeding such cells in separate plates as single cell micro-cultures on human feeder cells in the presence of Epstein-Barr Virus (EBV) (which also stimulate polyclonally memory B cells), a cocktail of growth factors (e.g., TLR9 agonist CpG-2006, IL-2 (1000 lU/ml), IL-6 (10 ng/ml), IL-21 (10 ng/ml), and anti-B cell receptor (BCR) goat antibodies (which trigger BCRs).
- ESV Epstein-Barr Virus
- supernatants of such cultures may tested in a primary luciferase-based screening system using two or more representative HIV-1 viruses or pseudoviruses that productively infect such cells.
- the pseudoviruses may be incubated with B cell culture supernatants for an appropriate time and temperature (e.g., one (1) h at 37% (5% C0 2 )) before the addition of host cells (e.g., 3000 TZM-bl cells). Incubation for an appropriate time (e.g., 72 hours) may then follow, after which the supernatant may be removed and Steadylite reagent (Perkin Elmer) added (e.g., 15 pi).
- Steadylite reagent Perkin Elmer
- Luciferase activity may then deteremined (e.g., five minutes later) on a Synergy microplate luminometer (BioTek). Decreased luciferase activity relative to a negative control typically indicates virus neutralization.
- Neutralization assays such as these, suitable for analyzing binding agents of this disclosure, are known in the art (see, e.g., Montefiori, D.C. Curr. Protocol. Immunol. Chapter 12, Unit 12.1 1 (2005); Edmonds, et al. Virology, 408(1): 1-13 (2010); Seaman, et al. J. Virol. 84(3): 1439-1452 (2010); Pace, et al. PNAS USA, 1 10(33): 13540-13545 (2013)).
- test samples may be screened for the presence of antibodies able to neutralize a panel of HIV pseudoviruses (e.g., nine (9) HIV-1 pseudoviruses from the Global Panel of HIV-1 reference strains as conducted in the examples herein (those pseudoviruses being TRO.1 1 (B), 246F3 (AC), BJOX2000 (CRF007_BC), CE1176 (C), CE0217 (C), CH1 19 (CRF07_BC), CNE55 (CRF01_AE), 25710 (C), and X1632 (see, e.g., Figs. 1 (showing the inhibitory dose (ID)) and 3); DeCamp, A. et al.
- HIV pseudoviruses e.g., nine (9) HIV-1 pseudoviruses from the Global Panel of HIV-1 reference strains as conducted in the examples herein (those pseudoviruses being TRO.1 1 (B), 246F3 (AC), BJOX2000 (CRF007_BC), CE1176 (C), CE
- a panel of ten HIV isolates may be tested and a bNab may be identified as one that neutralizes six, seven, eight, nine members of a panel of nine pseudoviruses; or six, seven, eight, nine, 10, 1 1 or 12 members of a panel of 12 pseudoviruses. Screening of larger panels of such pseudoviruses (e.g., a panel of 57 pseudoviruses as in the examples herein) may also be carried out.
- an exemplary panel of 57 pseudoviruses used in the examples against which test samples may be tested for neutralizing antibodies may include, for instance, those shown in Fig.
- neutralization may be determined as a measure of the concentration (e.g., pg/ml) of monoclonal antibody capable of neutralizing any of about 50%, 60%, 70%, 80%, 90%, 95%, or 99% of viral infection (as may be measured by percent neutralization and/or by determining an“IC 5 o” and/or“IC 8 o” value).
- a binding agent may be considered neutralizing if it is able to neutralize 50% of viral infection at a concentration of, for instance, about any of 10 5 , 10 4 , 10 3 , 10 2 , 10 1 , 10°, 10 1 , 10 2 , or 10 3 pg/ml (e.g., an IC 5 o value as shown in in Figs. 3-5).
- the ability of a neutralizing antibody to neutralize viral infection may be expressed as a percent neutralization (e.g., 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 99% (e.g., as in Fig. 3)).
- the ability of a neutralizing antibody to neutralize viral infection may be expressed as, and, in preferred embodiments, the IC 5 o and/or ICso value is below 25 pg/ml, and is even more preferably below about any of 15, 10, 5, 2, 1 , 0.5, 0.25, 0.1 , 0.05, or 0.01 pg/ml (see, e.g., Fig. 4).
- Other measures of neutralization may also be suitable as may be determined by those of ordinary skill in the art.
- the binding agents described herein may be broadly neutralizing antibodies (bNabs) identified in biological samples (e.g., plasma) obtained from HIV-infected persons.
- biological samples e.g., plasma
- bNabs may be identified by testing plasma samples of patients chronically infected by HIV (preferably those naive to antiretroviral therapy) for the ability to neutralize multi-clade HIV isolates (e.g., initially using a nine or 12-member panel and then a larger panel (e.g., 57 members) of pseudoviruses)).
- the samples may be derived from patients known to be“Elite Controllers” with viremia ⁇ 50 HIV RNA copies per ml of plasma.
- Screening procedures such as these may result in the identification of patients that may serve as lymph node donors for the subsequent isolation and characterization of B cells producing bNabs.
- neutralizing activity is typically compared to a negative control such as murine leukemia virus (MLV) pseudovirus.
- MLV murine leukemia virus
- germinal center and memory IgG B cells of patients expressing neutralizing binding agents may be isolated and further studied.
- the cells may be sorted separately according to IgG (e.g., IgA and IgM negative cells), CD19, and CD38 expression (germinal center B cells are CD38 positive) (see, e.g., Figure 2) and interrogated for the production of HIV-1 neutralizing antibodies.
- IgG memory B cells and IgG germinal cells may be seeded in separate plates as single cell micro-cultures on human feeder cells in the presence of Epstein-Barr Virus (EBV) (which also stimulate polyclonally memory B cells) and a cocktail of growth factors and the like (e.g., composed TLR9 agonist CpG-2006, IL-2 (1000 lU/ml), IL-6 (10 ng/ml), IL-21 (10 ng/ml), and anti-BCR goat antibodies (B cell receptor (BCR) triggering)).
- EBV Epstein-Barr Virus
- Supernatants of such cultures may then be tested in a primary screening (e.g., using a 384-well based HIV-1 pseudoviruses neutralization assay using in parallel two strains, CE1 176 and BJOX2000, representative of clade C and CRF07, as shown in the examples herein).
- Neutralization assays may be carried out using any suitable host cells (e.g., TZM-bl cells (Seaman, et al. J. Virol. 84(3): 1439-52 (2010); NIH AIDS Reagent Program Catalog Number 8129)).
- HIV-1 pseudoviruses resulting in a significant output relative light units may then incubated with B cell culture supernatants for an appropriate time and temperature (e.g., one (1) h at 37% (5% C0 2 )) before the addition of host cells (e.g., 3000 TZM-bl cells).
- RLU relative light units
- B cell culture supernatants for an appropriate time and temperature (e.g., one (1) h at 37% (5% C0 2 )) before the addition of host cells (e.g., 3000 TZM-bl cells).
- Incubation for an appropriate time typically follows, after which the supernatant may be removed and Steadylite reagent (Perkin Elmer) added (e.g., 15 pi).
- Luciferase activity may then be detected (e.g., five minutes later) on a Synergy microplate luminometer (BioTek). Decreased luciferase activity indicates a lesser amount of virus being released by the cells and virus neutralization. For instance, if the base RLU for a particular pseudovirus is 50-100 x 10 4 RLU, a neutralizing antibody may be determined to decrease the RLU for that pseudovirus to 25-50 x 10 4 RLU (i.e., a 50% decrease), or less. Using such systems, supernatants capable of cross-neutralizing strains may be identified, further harvested, and tested for their ability to neutralize other pseudoviruses.
- the antibodies derived from such neutralizing antibody-containing cultures may then be further characterized by determining the amino acid and nucleotide sequences of the antibody variable and complementarity determining regions (CDRs) regions.
- CDRs complementarity determining regions
- variable heavy chain (V H ) and variable light chain (V L ) genes of a binding agent may then be cloned into an IgG expression vector of the same or a different isotype.
- nucleic acids encoding LN02 CDRs (Table 1) were cloned into lgG1 backbone, and the recombinant lgG1-based antibody (lgG1 LN02) was produced by transfecting appropriate host cells (e.g., Expi293F cells).
- the antibody full-length lgG1-based antibody may then be purified using standard techniques (e.g., a full-length lgG1-based antibody may be purified using a recombinant protein-A column (GE-Healthcare)).
- the recombinantly-produced lgG1 antibody may then be tested against any of a panel of pseudoviruses such as any of those described herein (e.g., the Global Panel of nine (9) HIV-1 reference pseudoviruses used in the examples) on an appropriate host cell (e.g., TZM-bl cells).
- the binding agent will exhibit the ability to neutralize a majority (i.e., at least about 50% or greater) of the pseudovirus panel members (e.g., comprising nine, 12 or 1 18 members) without neutralizing a negative control virus (e.g., MLV pseudovirus). It is preferred that the binding agent exhibit the ability to neutralize a majority of such viruses (e.g., neutralization of greater than about 50%, such as any of about 60%, 70%, 80%, 90%, 95%, 99%, or 100%) with IC 5 o and/or IC 8 o values considered neutralizing (see below).
- a majority of such viruses e.g., neutralization of greater than about 50%, such as any of about 60%, 70%, 80%, 90%, 95%, 99%, or 100%
- IC 5 o and/or IC 8 o values considered neutralizing (see below).
- a binding agent of this disclosure may exhibit neutralization of HIV-1 pseudoviruses BJOX (CRF07_BC), CE1 176, TRO.11 (B), X1632 (G), CH1 19 (CRF07_BC), CNE55 (CRF01_AE), 25710 (C), CD0217(C), but not of the control virus SVA-MLV at about 10° pg/ml or less (e.g., Figure 3).
- neutralization of the HIV-1 pseudoviruses viruses may be observed where the antibody concentration is from 10 2 -10° pg/ml, or between 10°-10 1 pg/ml ( Figure 3).
- the percent neutralization by the binding agent is at least about 50% ( Figure 3).
- infection of one HIV-1 isolate is considered neutralized by a binding agent (e.g., antibody) at an IC50 and/or ICso of less than 25 mg/ml, if infection of at least one isolate of this isolate is neutralized with an IC50 of less than 25 mg/ml.
- a binding agent e.g., antibody
- the binding agent may be considered neutralizing where a majority of the 57 HIV-1 pseudoviruses listed in Figure 4 are considered neutralized at an IC50 of less than 25 pg/ml, such as about 10 pg/ml, 9 pg/ml, 8 pg/ml, 7 pg/ml, 6 pg/ml, 5 pg/ml, 4 pg/ml, 3 pg/ml, 2 pg/ml, 1 pg/ml, 0.9 pg/ml, 0.8 pg/ml, 0.7 pg/ml, 0.6 pg/ml, 0.5 pg/ml, 0.4 pg/ml, 0.3 pg/ml, 0.2 pg/ml, 0.1 pg/ml, 0.09 pg/ml, 0.08 pg/ml, 0.07 pg/ml, 0.06 pg/ml, 0.05 p
- the binding agent may neutralize HIV-1 pseudovirus strains of ID MS208.A1 , Q23.17, Q769.d22, Q842.d12, Q259.d2.17, 0260.v5.c36, 191955_A11 , 191084 B7-19, TR0.11 , 6535.3, REJ04541.67, SC422661.8, QH0692.42, TRJ04551.58, RHPA4259.7, PVO.4, SC05 8C1 1 2344, CNE17, CNE19, CNE20, CNE21 , Du422.1 , CAP210.2.00.E8, ZM249M.PB6, HIV-001428-2.42, ZM214M.PL15, CAP45.2.00.G3, Ce704809221_1 B3, Ce1 176_A3, ZM247v1 (Rev-), Ce0682_E4, 249M B10, 246F C1G, and/or BF1266.431a at an IC50 of less than or about 1
- the binding agent may neutralize HIV-1 pseudovirus strains of ID MS208.A1 , Q23.17, Q769.d22, Q842.d12, Q259.d2.17, 0260.v5.c36, 191084 B7-19, TR0.11 , REJ04541.67, SC422661.8, QH0692.42, TRJ04551.58, RHPA4259.7, SC05 8C1 1 2344, CNE19, CNE20, CNE21 , CAP210.2.00.E8, HIV-001428-2.42, ZM214M.PL15, CAP45.2.00.G3,
- the binding agent may neutralize HIV-1 pseudovirus strains of ID MS208.A1 , Q23.17, Q769.d22, Q842.d12, Q259.d2.17, 0260.v5.c36, 191084 B7-19, TR0.1 1 , 6535.3, REJ04541.67, SC422661.8, QH0692.42, TRJ04551.58, RHPA4259.7, PVO.4, SC05 8C1 1 2344, CNE19, CNE20, CNE21 , Du422.1 , HIV-001428-2.42, ZM214M.PL15, CAP45.2.00.G3, Ce704809221_1 B3, Ce1 176_A3, Ce0682_E4, and/or 246F C1 G, at an IC 8 o
- the binding agent may exhibit the ability to neutralize pseudoviruses of HIV-1 Clades A, A(T/F), B, B (T/F), BC, C, and C (T/F) (Fig. 4). In some preferred embodiments, the binding agent may neutralize at least one pseudovirus in each of clades A, A(T/F), B, B (T/F), BC, C, and C (T/F) at at an IC50 of less than or about 1 pg/ml (Fig. 4).
- the binding agent may neutralize at least one pseudovirus in each of clades A, A(T/F), B, B (T/F), BC, C, and C (T/F) at an IC50 of less than or about 0.5 pg/ml (Fig. 4). In some preferred embodiments, the binding agent may neutralize at least one pseudovirus in each of clades A, A(TVF), B, B (T/F), BC, C, and C (T/F) at an IC 8 o of less than or about 1 pg/ml (Fig. 4). In some embodiments, the binding agent comprises any one or more of these properties and one or more of SEQ ID NOS. 1-8, preferably at least one of SEQ ID NO.
- LN02 variable region SEQ ID NO. 1 (CDRH1 region); SEQ ID NO. 2 (CDRH2 region); SEQ ID NO. 3 (CDRH3 region); SEQ ID NO. 4 (CDRL1 region); SEQ ID NO. 5 (CDRL2 region); and, at least one of SEQ ID NOS. 6 (CDRL3 region); and/or, at least one of SEQ ID NO. 7, and/or SEQ ID NO. 8 (LN02 variable region); and/or fragments and/or derivatives thereof; as well as any of the same comprising one or more amino acid substitutions, in particular or preferably conservative substitutions (see, e.g., the substitutions listed in Table 2).
- LN02-type binding agents such as but not limited to lgG1 LN02 (“LN02”), as shown in Fig. 4.
- the binding agents may be tested for neutralization capacity against HIV reference pseudoviruses (e.g., the above-described Global Panel of nine (9) HIV- 1 reference pseudoviruses) using cells expressing or not expressing one or more types of Fc receptors (e.g., parental TZM-bl cells and TZM-bl cells expressing Fc-gamma receptor I (CD64) as in the examples; see e.g. Perez, et al. Utilization of immunoglobulin G Fc receptors by human immunodeficiency virus type 1 : a specific role for antibodies against the membrane- proximal external region of gp41.
- HIV reference pseudoviruses e.g., the above-described Global Panel of nine (9) HIV- 1 reference pseudoviruses
- Fc receptors e.g., parental TZM-bl cells and TZM-bl cells expressing Fc-gamma receptor I (CD64) as in the examples; see e.g. Perez, et al
- Enhanced neutralizing activity in cells expressing Fc receptors may provide antibodies a kinetic advantage for virus inhibition. This kinetic advantage could be unique to antibodies, whose epitopes are thought to be difficult to access or exposed for only a short time on intermediate conformations of the Env protein during an early stage of fusion. Fc-gamma receptors could also potentially facilitate HIV-1 neutralization is phagocytosis, thereby increasing neutralization capacity of the antibodies. To this point, HeLa cells, from which the TZM-bl cell line was constructed, are known to exhibit properties of nonprofessional phagocytes.
- TZM-bl cells were converted to professional phagocytic cells by introducing Fc-gamma receptor on their surface.
- Fc-gamma receptors are rarely expressed on CD4+ lymphocytes, but several other HIV-1 -susceptible cell types express multiple Fc-gamma receptors and are involved in sexual transmission and the early establishment of long-lived viral reservoirs.
- macrophages are among the first infection-susceptible cells that the virus encounters after mucosal exposure, and are thought to serve as a long-lived virus reservoir in chronic infection.
- Macrophages, as well as certain subsets of monocytes and dendritic cells, are known to express multiple Fc-gamma receptors. It is also important to mention that Fc-gamma receptors play a role in regulating adaptive immunity and peripheral tolerance, by facilitating antigen uptake, antigen presentation, cell activation and B cell tolerance.
- the binding agents described herein may be used in conjunction with agents that induce and/or enhance Fc receptor expression, including the introduction of nucleic acids encoding one or more Fc receptors with or in conjunction with treatment by the binding agents described herein.
- the specificity of the binding agents described herein may be determined using any of the many techniques available to those of ordinary skill in the art. For instance, as shown in the examples herein, the specificity of a binding agent (e.g., lgG1 LN02 antibody), with respect to particular epitopes, may be ascertained using a panel of pseudoviruses (e.g., CAP45) that encode mutations in the HIV envelope gene. Mutations summarized in Table 5 show that neutralization activity for LN02 is lost (>10 ug/ml) when the Q619Y substitution is introduced into the gp120/gp41 interface region of HIV-1 Env (Table 5).
- a binding agent e.g., lgG1 LN02 antibody
- pseudoviruses e.g., CAP45
- the cleavage site mutation E500K results in a 5-fold improvement in neutralization activity of LN02 (Fig. 6).
- Mutation of the N-glycan at position 88 in Env with an N88A substitution resulted in an 8.4- to 22-fold improvement potency for different viruses (Table 6). This observation is consistent with LN02 binding at the interface region of Env since the glycan is able to shield the interface from antibody binding.
- the HIV-1 envelope amino acid sequence for CAP45 is shown below (GenBank Accession No EF203962; NCBI GenPept Accession No. ABQ02701.1 ; SEQ ID NO.: 67) with the mutated resides indicated in bold and underlined (Fig. 9).
- a binding agent of this disclosure may comprise these binding specificities along with the neutralization characteristics described above (i.e., neutralization of HIV-1 pseudoviruses BJOX (CRF07_BC), CE1 176, TR0.1 1 (B), X1632 (G), CH1 19 (CRF07_BC), CNE55 (CRF01_AE), 25710 (C), CD0217(C) but not of the control virus SVA- MLV at a concentration is from 10 2 -10° ug/ml, or between 10°-10 1 ug/ml, to at least about 50% (Figs. 3 and 4), as well as the neutralization of a majority of the 57 HIV-1 pseudoviruses listed in Figure 4 at an IC50 of less than 25 pg/ml.
- a binding agent may also be tested for binding to soluble trimers representing HIV proteins (e.g., soluble, cleaved SOSIP.664 gp140 trimers based on the subtype A transmitted/founder strain, BG505 as used in the examples herein).
- Preferred trimers are those being highly stable, homogenous and closely resembling native virus spikes when visualized by negative stain electron microscopy (EM) (Sanders, R. W. et al.
- a next-generation cleaved, soluble HIV-1 Env trimer BG505 SOSIP.664 gp140, expresses multiple epitopes for broadly neutralizing but not nonneutralizing antibodies.
- BG505 SOSIP.664 gp140 expresses multiple epitopes for broadly neutralizing but not nonneutralizing antibodies.
- non-neutralizing antibodies to the CD4-binding site, CD4-induced epitopes or gp41 ectodomain would not (and did not in the example) react with the trimers, even when their epitopes were present on simpler forms of Env (e.g., gp120 monomers or dissociated gp41 subunits).
- the examples also included a test, which may be used in testing any of binding agents described herein, in which the MPER was also deleted to improve trimer solubility and reduce aggregate formation. The binding agents may also be tested for binding to such trimers in the presence or absence of soluble CD4 (sCD4).
- binding affinity and/or K D refers to the dissociation rate of a particular antibody-antigen interaction.
- the K D is the ratio of the rate of dissociation (“off-rate (K d )”) to the association rate (“on-rate” (K a )).
- K D therefore equals K d /K a and is expressed as a molar concentration (M).
- M molar concentration
- the binding agents described herein may be compared with another binding agent with reference to the respective K D values of each. These properties may be combined with other characteristics such as neutralization capacity and/or epitope specificity in order to compare binding agents to one another. Accordingly, binding agents having a similar K D to those described herein, perhaps also sharing the neutralization capacity and epitope specificity described herein (e.g., as exhibited by LN02), are also contemplated as part of this disclosure.
- any of the amino acid sequences of Table 1 may be also substituted by any other amino acid as desired by one of ordinary skill in the art. Preferably such substitutions do not interfere with a beneficial or desired property of the binding agent (e.g., having a similar specificities and/or K D to LN02).
- a beneficial or desired property of the binding agent e.g., having a similar specificities and/or K D to LN02.
- one of skill in the art may make conservative substitutions by replacing particular amino acids with others as shown in Table 2 below.
- the specific amino acid substitution selected may depend on the location of the site selected.
- An amino acid substitution may be said to“correspond to” where one of ordinary skill in the art could ascertain a significant amount of similarity between the amino acid sequences surrounding the amino acid being substituted.
- a particular amino acid sequence may correspond to another where two, three, four or more N-terminal and C-terminal amino acids surrounding the amino acid being substituted are the same or similar (e.g., as described in Table 2) in the polypeptides being compared.
- Conservative amino acid substitutions may involve a substitution of a native amino acid residue with a non-native residue such that there is little or no effect on the size, polarity, charge, hydrophobicity, or hydrophilicity of the amino acid residue at that position and, in particular, does not result in, e.g., decreased HIV neutralization capacity and/or different epitope specificity.
- a nucleic acid molecule encoding one or more binding agents described herein may be inserted into one or more expression vectors, as discussed below in greater detail.
- the binding agent may be encoded by nucleotides corresponding to the amino acid sequence.
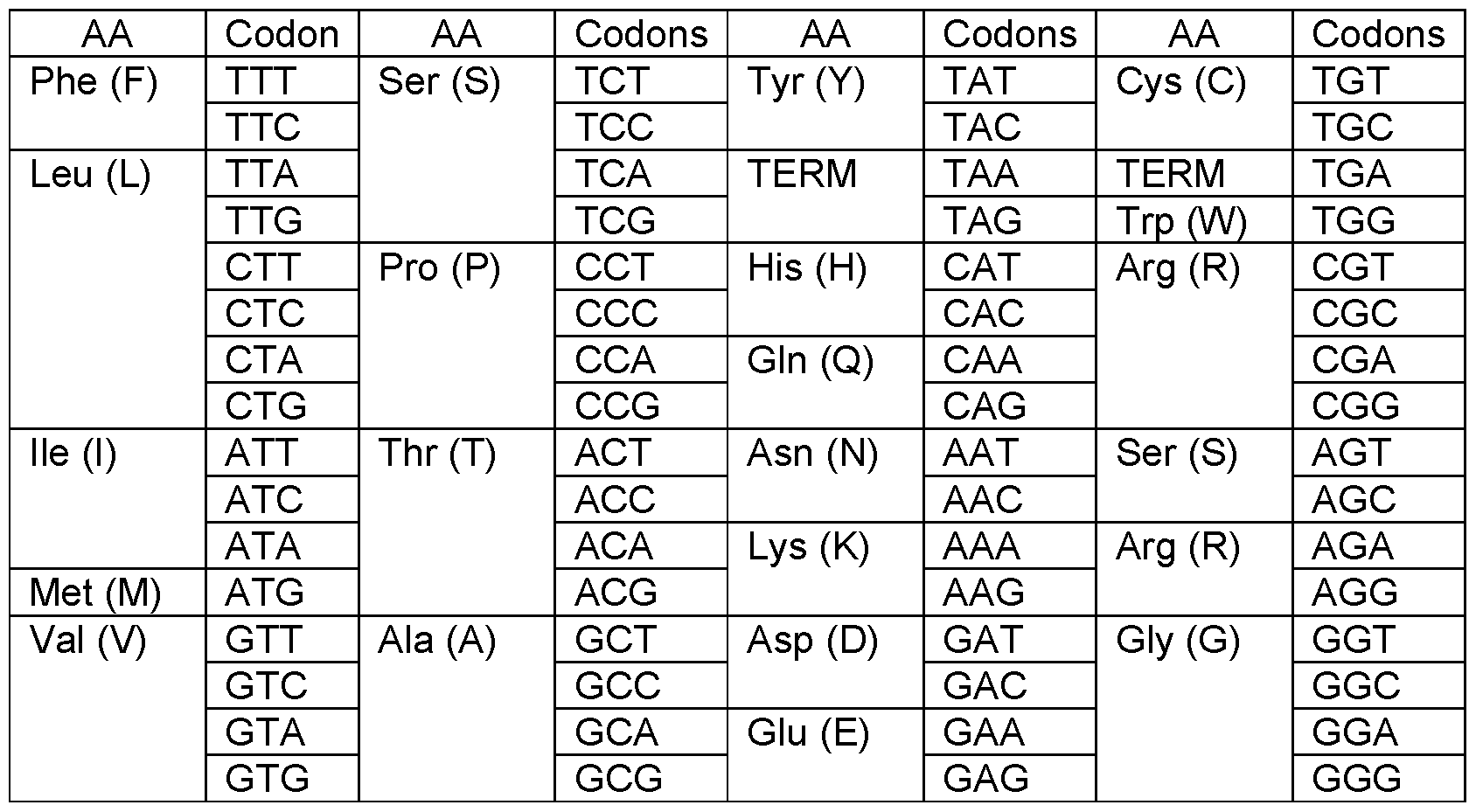
- the particular combinations of nucleotides (codons) that encode the various amino acids (AA) are well known in the art, as described in various references used by those skilled in the art (e.g., Lewin, B. Genes V, Oxford University Press, 1994).
- the nucleotide sequences encoding the amino acids of said binding agents may be ascertained with reference to Table 3, for example.
- Nucleic acid variants may use any combination of nucleotides that encode the binding agent.
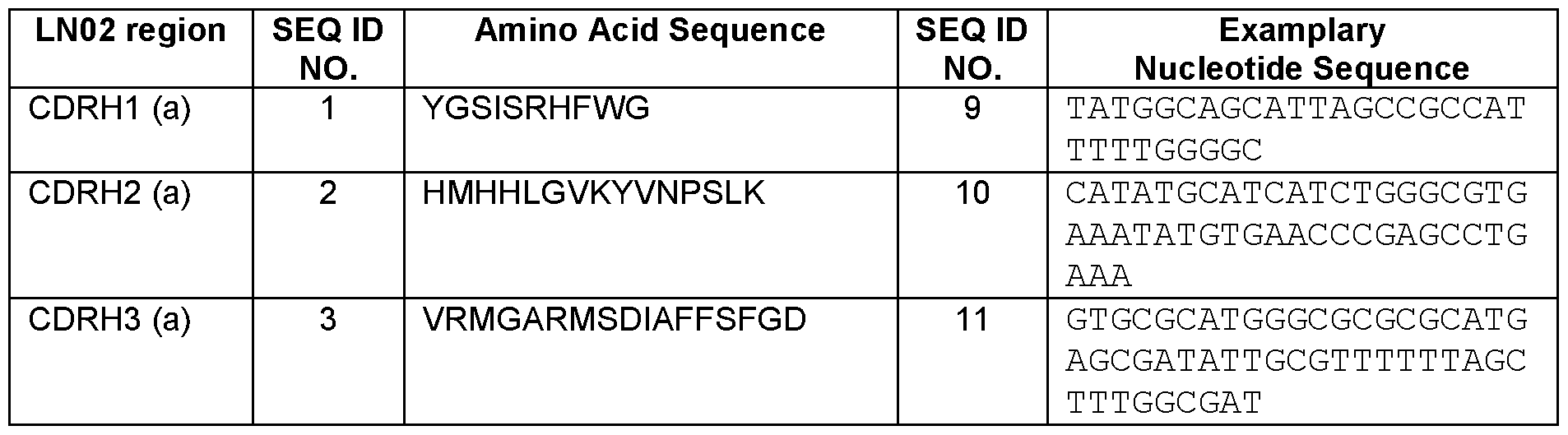
- nucleotide sequence encoding a particular amino acid sequence may be easily derived from the amino acid sequence of any of SEQ ID NOS. 1-8 and the information presented in Table 4. For instance, it may be deduced from the amino acid sequence YGSISRHFWG (SEQ ID NO.: 19) and the information presented in Table 4 that the amino acid sequence may be encoded by the nucleotide sequence TATG G CAG CATT AG COG CCATTTTT G G GG C (SEQ ID NO.: 20). Those of ordinary skill in the art would understand that nucleotide sequences encoding SEQ ID NOS. 1-8 may be deduced in the same way, and such nucleotide sequences are contemplated herein. Table 4 provides exemplary nucleic acid sequences encoding the amino acid sequences shown in Table 1 (SEQ ID NOS. 1-8):
- a nucleic acid encoding a binding agent of this disclosure may comprise one or more SEQ ID NOS. 9-16 and/or a fragment and/or derivative thereof (e.g., the binding agent comprising any of SEQ ID NOS. 1-8 (that can be conservatively substituted, e.g., as described in Table 2), encoded by a nucleotide sequence determined using standard techniques, e.g., as described in Table 3).
- Exemplary coding sequences included in such nucleic acids include but are not limited to those described in Table 4 (SEQ ID NOS. 9-16), or derivatives (e.g.,“wobble” derivatives in which the third member of codon is changed without causing an amino acid sequence substitution), and/or fragments thereof.
- nucleic acid sequences comprising such nucleic acid sequences are also contemplated by this disclosure.
- binding agents are antibodies
- nucleotide sequences encoding the variable regions thereof may also be isolated from the phage and / or hybridoma cells expressing the same cloned into expression vectors. Methods for producing such preparations are well-known in the art.
- Nucleic acid molecules encoding one or more HIV binding agents may be contained within a viral and / or a non-viral vector.
- a DNA vector is utilized to deliver nucleic acids encoding one or more HIV binding agents to the patient.
- various strategies may be utilized to improve the efficiency of such mechanisms including, for example, the use of self-replicating viral replicons (Caley, et al. 1999. Vaccine , 17: 3124-2135; Dubensky, et al. 2000. Mol. Med. 6: 723-732; Leitner, et al. 2000. Cancer Res. 60: 51-55), codon optimization (Liu, et al. 2000. Mol.
- Vaccine, 19: 2647- 2656 incorporation of stimulatory motifs such as CpG (Gurunathan, supra ; Leitner, supra), sequences for targeting of the endocytic or ubiquitin-processing pathways (Thomson, et al. 1998. J. Virol. 72: 2246-2252; Velders, et al. 2001. J. Immunol. 166: 5366-5373), prime-boost regimens (Gurunathan, supra ; Sullivan, et al. 2000. Nature, 408: 605-609; Hanke, et al. 1998. Vaccine, 16: 439-445; Amara, et al. 2001.
- stimulatory motifs such as CpG (Gurunathan, supra ; Leitner, supra), sequences for targeting of the endocytic or ubiquitin-processing pathways (Thomson, et al. 1998. J. Virol. 72: 2246-2252; Velders, et al.
- Various viral vectors that have been successfully utilized for introducing a nucleic acid to a host include retrovirus, adenovirus, adeno-associated virus (AAV), herpes virus, and poxvirus, among others.
- the vectors may be constructed using standard recombinant techniques widely available to one skilled in the art.
- Non-viral plasmid vectors may also be suitable in certain embodiments. Preferred plasmid vectors are compatible with bacterial, insect, and / or mammalian host cells.
- Such vectors include, for example, PCR-ii, PCR3, and pcDNA3.1 (Invitrogen, San Diego, CA), pBSii (Stratagene, La Jolla, CA), pet15 (Novagen, Madison, Wl), pGEX (Pharmacia Biotech, Piscataway, NJ), pEGFp-n2 (Clontech, Palo Alto, CA), pETI (Bluebacii, Invitrogen), pDSR-alpha (PCT pub. No. WO 90/14363) and pFASTBACdual (Gibco-
- Bluescript plasmid derivatives a high copy number COLel- based phagemid, Stratagene Cloning Systems, La Jolla, CA
- PCR cloning plasmids designed for cloning TAQ-amplified PCR products e.g. , TOPOTM TA cloning ® kit, PCR2.1 ® plasmid derivatives, Invitrogen, Carlsbad, CA.
- Bacterial vectors may also be used.
- vectors include, for example, Shigella, Salmonella, Vibrio cholerae, Lactobacillus, Bacille Calmette Guerin (BCG), and Streptococcus (see for example, WO 88/6626; WO 90/0594; WO 91/13157; WO 92/1796; and WO 92/21376).
- BCG Bacille Calmette Guerin
- Streptococcus see for example, WO 88/6626; WO 90/0594; WO 91/13157; WO 92/1796; and WO 92/21376).
- Other delivery techniques may also suffice including, for example, DNA-ligand complexes, adenovirus-ligand-DNA complexes, direct injection of DNA, CaP0 4 precipitation, gene gun techniques, electroporation, and colloidal dispersion systems.
- Colloidal dispersion systems include macromolecule complexes, nanocapsules, microspheres, beads, and lipid-based systems including oil-in-water emulsions, micelles, mixed micelles, and liposomes.
- the preferred colloidal system is a liposome, which are artificial membrane vesicles useful as delivery vehicles in vitro and in vivo.
- RNA, DNA and intact virions can be encapsulated within the aqueous interior and be delivered to cells in a biologically active form (Fraley, R., et al., 1981 , Trends Biochem. Sci., 6: 77).
- the composition of the liposome is usually a combination of phospholipids, particularly high-phase-transition- temperature phospholipids, usually in combination with steroids, especially cholesterol. Other phospholipids or other lipids may also be used.
- the physical characteristics of liposomes depend on pH, ionic strength, and the presence of divalent cations. Examples of lipids useful in liposome production include phosphatidyl compounds, such as phosphatidylglycerol, phosphatidylcholine, phosphatidylserine, phosphatidylethanolamine, sphingolipids, cerebrosides, and gangliosides.
- diacylphosphatidylglycerols where the lipid moiety contains from 14-18 carbon atoms, particularly from 16-18 carbon atoms, and is saturated.
- Illustrative phospholipids include egg phosphatidylcholine, dipalmitoylphosphatidylcholine and distearoylphosphatidylcholine.
- a cultured cell comprising the vector is also provided.
- the cultured cell may be a cultured cell transfected with the vector or a progeny of the cell, wherein the cell expresses the immunogenic polypeptide.
- Suitable cell lines are known to those of skill in the art and are commercially available, for example, through the American Type Culture Collection (ATCC).
- ATCC American Type Culture Collection
- the transfected cells can be used in a method of producing an immunogenic polypeptide.
- the method comprises culturing a cell comprising the vector under conditions that allow expression of the immunogenic polypeptide, optionally under the control of an expression sequence.
- the immunogenic polypeptide can be isolated from the cell or the culture medium using standard protein purification methods.
- the binding agents described herein may be conjugated to active agents to target and inhibit the function of and /or eliminate cell populations expressing HIV polypeptides and/or harboring HIV (and / or another antigen in the case of binding agents with multiple specificities).
- CD4 + T-cell populations containing replication competent HIV may be targeted and eliminated using binding agent / drug conjugates (e.g., antibody-drug conjugates (ADC)).
- ADC antibody-drug conjugates
- Mono- and/or bi-specific candidate binding agents may be conjugated with one or more types of drugs (e.g., drugs damaging DNA, targeting microtubules).
- the binding agents described herein and/ or derivatives thereof may also be adjoined to and / or conjugated to functional agents for in vitro and / or in vivo use.
- the binding agent may be adjoined to and / or conjugated to functional moieties such as cytotoxic drugs or toxins, and / or active fragments thereof such as diphtheria A chain, exotoxin A chain, ricin A chain, abrin A chain, curcin, crotin, phenomycin, enomycin, among others.
- Suitable functional moieties may also include radiochemicals.
- Binding agents, such as antibodies, may be adjoined to and / or conjugated to the one or more functional agents using standard techniques in the art.
- this disclosure provides binding agents with multiple specificities such that epitopes bound by an LN02 binding agent (e.g., antibody) and at least one other secondary antigen (e.g., a cell surface protein) may be bound by a single binding agent.
- the secondary antigen may be one expressed by cells infected by an infectious agent.
- an exemplary secondary antigen may be HIV Env antigen other than gp41.
- binding agents may bind the secondary antigen and / or may serve to neutralize the infectious agent as may be determined using the assays described herein. Combinations of binding agents, such as one or more described herein with another available to those of ordinary skill in the art, are also contemplated herein.
- the combinations may be identified to provide statistically significant differences from results (e.g., neutralization assays) obtained using only one or more of the binding agents and not others.
- combinations exhibit additive and/or, preferably synergistic, neutralization of HIV, for example.
- the combination may comprise a first binding agent having the characteristics of an LN02 binding agent (e.g., antibody such as lgG1 LN02) and/or comprising any one or more of SEQ ID NOS. 1-8 (and/or as described in Table 1), and/or derivatives thereof, and any one or more of the antibodies described in any one or more of U.S. Pat. No. 5,087,557; U.S. Pat. No.
- any of the binding agents described herein may be combined with (i.e. , as a single composition, and/or used in conjunction with) one or more the antibodies commonly known as 2F5, 4E10 and/or Z13e1 , and/or derivatives thereof, among others.
- the binding agents of such compositions may be different entities such as two or more different monoclonal antibodies or derivatives thereof, or may be found on the same entity such as a bi-functional antibody (a single antibody or derivative thereof comprising multiple binding specificities).
- Such combinations as described herein may also be combined with one or more other agents that may affect immune cell function such as antibodies against CTLA-4, and the like.
- One of ordinary skill in the art would recognize that many such combinations may be suitable for use as described herein.
- HIV binding agents described herein may be used to treat and / or prevent and / or ameliorate the symptoms of infection by HIV.
- HIV isolates are now classified into discrete genetic subtypes.
- HIV-1 is known to comprise at least ten subtypes (A1 , A2, A3, A4, B, C, D, E, F1 , F2, G, H, J and K) (Taylor et al, NEJM, 359(18): 1965-1966 (2008)).
- HIV-2 is known to include at least five subtypes (A, B, C, D, and E).
- Subtype B has been associated with the HIV epidemic in homosexual men and intravenous drug users worldwide.
- HIV-1 immunogens laboratory adapted isolates, reagents and mapped epitopes belong to subtype B.
- subtype B In sub-Saharan Africa, India and China, areas where the incidence of new HIV infections is high, HIV-1 subtype B accounts for only a small minority of infections, and subtype HIV-1 C appears to be the most common infecting subtype. Any of these types of isolates may be addressed using the binding agents described herein.
- One or more binding agents may also be administered with or in conjunction with one or more agents used to prevent, treat and / or ameliorate HIV such as for example, a protease inhibitor, an HIV entry inhibitor, a reverse transcriptase inhibitor, and / or an anti- retroviral nucleoside analog.
- Suitable compounds include, for example, Agenerase (amprenavir), Combivir (Retrovir / Epivir), Crixivan (indinavir), Emtriva (emtricitabine), Epivir (3tc / lamivudine), Epzicom, Fortovase / Invirase (saquinavir), Fuzeon (enfuvirtide), Hivid (ddc / zalcitabine), Kaletra (lopinavir), Lexiva (Fosamprenavir), Norvir (ritonavir), Rescriptor (delavirdine), Retrovir / AZT (zidovudine), Reyatax (atazanavir, BMS-232632), Sustiva (efavirenz), Trizivir (abacavir/ zidovudine / lamivudine), Truvada (Emtricitabine / Tenofovir DF), Videx (ddl / didanosine), Videx EC (d
- binding agents e.g., antibodies
- antibodies may be utilized to isolate HIV or cells containing HIV and / or expressing HIV antigens using, for example, immunoprecipitation or other capture-type assay.
- This well-known technique is performed by attaching the antibody to a solid support or chromatographic material (e.g., a bead coated with Protein A, Protein G and / or Protein L).
- the bound antibody is then introduced into a solution either containing or believed to contain HIV antigens (e.g., an HIV-infected cell).
- the HIV antigen(s) may then bind to the antibody and non-binding materials are washed away under conditions in which the HIV antigen(s) remains bound to the antibody.
- the bound protein may then be separated from the antibody and analyzed as desired. Similar methods for isolating a protein using an antibody are well- known in the art.
- the binding agents e.g., antibodies
- the antibodies may also be utilized to detect HIV or HIV antigens within a biological sample.
- the antibodies may be used in assays such as, for example, flow cytometric analysis, ELISA, immunoblotting (e.g., western blot), in situ detection, immunocytochemistry, and / or immunhistochemistry. Methods of carrying out such assays are well-known in the art.
- the binding agents may be adjoined to and / or conjugated to one or more detectable labels.
- suitable detectable labels may include, for instance, fluorosceins (e.g., DyLight, Cy3, Cy5, FITC, HiLyte Fluor 555, HiLyte Fluor 647; 5-carboxy-2,7-dichlorofluorescein; 5-Carboxyfluorescein (5-FAM); 5-HAT (Hydroxy Tryptamine); 5-Hydroxy Tryptamine (HAT); 6-JOE; 6-carboxyfluorescein (6- FAM); FITC; 6-carboxy-1 ,4-dichloro-2’,7’-dichlorofluorescein (TET); 6-carboxy-1 ,4-dichloro- 2’, 4’, 5’, 7’-tetrachlorofluorescein (HEX); 6-carboxy-4’,5’-dichloro-2’, 7’-
- the binding agents described herein may be also be used to determine the presence of a disease state in a patient, to predict prognosis, or to determine the effectiveness of a chemotherapeutic or other treatment regimen.
- Expression profile assays performed as described herein or as is otherwise known in the art, may be used to determine the relative level of expression of HIV in a cell, for instance. The level of expression may then be correlated with base (e.g., control) levels to determine whether a particular disease is present within the patient, the patient’s prognosis, or whether a particular treatment regimen is effective.
- an increased or decreased level of expression of HIV in the patient’s tissues may indicate the regimen is worsening or improving the load of HIV in that host.
- the increase or decrease in expression may indicate the regimen is having or not having the desired effect and another therapeutic modality may therefore be selected.
- the binding agents described herein as reagents in drug screening assays to test, for example, new drug candidates.
- the reagents may be used to ascertain the effect of a drug candidate on the expression of the immunogenic target in a cell line, or a cell or tissue of a patient.
- the expression profiling technique may be combined with high throughput screening techniques to allow rapid identification of useful compounds and monitor the effectiveness of treatment with a drug candidate (see, for example, Zlokarnik, et al. , Science 279, 84-8 (1998)).
- Drug candidates may be chemical compounds, nucleic acids, proteins, antibodies, or derivatives therefrom, whether naturally occurring or synthetically derived. Drug candidates thus identified may be utilized, among other uses, as pharmaceutical compositions for administration to patients or for use in further screening assays.
- the binding agents are in purified form.
- A“purified” binding agent e.g., antibody
- a purified binding agent may be one that is separated from at least about 50% of the proteins and / or other components with which it is initially found (e.g., as part of a hybridoma supernatant or ascites preparation in the case of a monoclonal antibody).
- a purified binding agent e.g., antibody
- binding agents e.g., polypeptides
- nucleic acids described herein may also be combined with one or more pharmaceutically acceptable carriers prior to administration to a host.
- a pharmaceutically acceptable carrier is a material that is not biologically or otherwise undesirable, e.g., the material may be administered to a subject, without causing any undesirable biological effects or interacting in a deleterious manner with any of the other components of the pharmaceutical composition in which it is contained.
- the carrier would naturally be selected to minimize any degradation of the active ingredient and to minimize any adverse side effects in the subject, as would be well known to one of skill in the art. Suitable pharmaceutical carriers and their formulations are described in, for example, Remington’s: The Science and Practice of Pharmacy, 21 st Edition, David B.
- a pharmaceutically-acceptable salt is used in the formulation to render the formulation isotonic.
- the pharmaceutically-acceptable carriers include, but are not limited to, sterile water, saline, buffered solutions like Ringer's solution, and dextrose solution. The pH of the solution is generally from about 5 to about 8 or from about 7 to about 7.5.
- Other carriers include sustained-release preparations such as semipermeable matrices of solid hydrophobic polymers containing polypeptides or fragments thereof. Matrices may be in the form of shaped articles, e.g., films, liposomes or microparticles.
- Carriers are those suitable for administration of polypeptides and / or fragments thereof to humans or other subjects.
- Pharmaceutical compositions may also include carriers, thickeners, diluents, buffers, preservatives, surface active agents, adjuvants, immunostimulants, in addition to the immunogenic polypeptide.
- Pharmaceutical compositions may also include one or more active ingredients such as antimicrobial agents, anti inflammatory agents and anesthetics.
- the pharmaceutical composition may be administered orally, parentally, by inhalation spray, rectally, intranodally, or topically in dosage unit formulations containing conventional pharmaceutically acceptable carriers, adjuvants, and vehicles.
- pharmaceutically acceptable carrier or“physiologically acceptable carrier” as used herein refers to one or more formulation materials suitable for accomplishing or enhancing the delivery of a nucleic acid, polypeptide, or peptide as a pharmaceutical composition.
- A“pharmaceutical composition” is a composition comprising a therapeutically effective amount of a nucleic acid or polypeptide.
- effective amount and “therapeutically effective amount” each refer to the amount of a binding agent, nucleic acid or the like used to observe the desired therapeutic effect (e.g., eliminating HIV).
- binding agent is a monoclonal antibody or fragment or derivative thereof comprising one or more of SEQ ID NOS. 1-8, including all of SEQ ID NOS. 1-6; or SEQ ID NOS.: 7 and 8; the amino acid sequences shown in Table 1 ; and/or substituted derivatives and/or fragments thereof; as well as in some embodiments conservatively substituted variants thereof.
- the one or more binding agents may be administered in a dosage amount of about 1 to about 50 mg / kg, about 1 to about 30 mg / kg, or about 5 to about 30 mg / kg (e.g., about any of 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 21 , 22, 23, 24, 25, 26, 27, 28, 29, 30, 35, or 40 mg / kg).
- the one or more binding agents may be administered to the mammal (e.g., intradermally, intravenously, orally, rectally) at about 10 mg / kg one or more times. When multiple doses are administered, the doses may comprise about the same or different amount of binding agent in each dose.
- the doses may also be separated in time from one another by the same or different intervals.
- the doses may be separated by about any of 6, 12, 24, 36, 48, 60, 72, 84, or 96 hours, one week, two weeks, three weeks, one month, two months, three months, four months, five months, six months, seven months, eight months, nine months, 10 months, 1 1 months, 12 months, 1.5 years, 2 years, 3 years, 4 years, 5 years, or any time period before, after, and / or between any of these time periods.
- the binding agents may be administered in conjunction with other agents (e.g., anti-infective agents and/or chemotherapeutic agent). Such other agents may be administered about simultaneously with the binding agents, or at a different time and / or frequency. Other embodiments of such methods may also be appropriate as could be readily determined by one of ordinary skill in the art.
- kit format A kit including one or more of such binding agents and optionally other components necessary for using the same to detect cells expressing HIV is also provided.
- the binding agents of the kit may be provided in any suitable form, including frozen, lyophilized, or in a pharmaceutically acceptable buffer such as TBS or PBS.
- the kit may also include other reagents required for utilization of the binding agents in vitro or in vivo such as buffers (e.g., TBS, PBS), blocking agents (solutions including nonfat dry milk, normal sera, Tween-20 Detergent, BSA, or casein), and / or detection reagents (e.g., goat anti mouse IgG biotin, streptavidin-HRP conjugates, allophycocyanin, B-phycoerythrin, R- phycoerythrin, peroxidase, detectable labels, and other labels and / or staining kits (e.g., ABC Staining Kit, Pierce)).
- buffers e.g., TBS, PBS
- blocking agents solutions including nonfat dry milk, normal sera, Tween-20 Detergent, BSA, or casein
- detection reagents e.g., goat anti mouse IgG biotin, streptavidin-HRP conjugates, allophycocyanin, B-
- kits may also include other reagents and / or instructions for using the antibodies in commonly utilized assays described above such as, for example, flow cytometric analysis, ELISA, immunoblotting (e.g., western blot), in situ detection, immunocytochemistry, and/or immunhistochemistry.
- the kit provides a binding agent in purified form.
- the binding agent may be provided in biotinylated form either alone or along with an avidin-conjugated detection reagent (e.g., antibody).
- the kit includes a binding agents comprising one or more detectable labels that may be used to directly detect HIV.
- kits and the like required for using any of these systems are well-known in the art and / or may be prepared by the end-user or provided as a component of the kit.
- the kit may also include a solid support containing positive- and negative-control protein and / or tissue samples.
- kits for performing spotting or western blot-type assays may include control cell or tissue lysates for use in SDS-PAGE or nylon or other membranes containing pre-fixed control samples with additional space for experimental samples.
- Kits for visualization of HIV in cells on slides may include pre-formatted slides containing control cell or tissue samples with additional space for experimental samples.
- Other embodiments of kits are also contemplated herein as would be understood by those of ordinary skill in the art.
- binding agents such as the LN02 antibody with specificity for HIV (e.g., and/or an antigen thereof).
- the binding agent is a polypeptide comprising at least one amino acid sequence selected from the group consisting of SEQ ID NOS. 1-8 and / or shown in Table 1 , and/or substituted derivatives and/or fragments thereof.
- the binding agent is a polypeptide comprising one or more combinations of SEQ ID NOS. 1-8 and/or substituted derivatives and/or fragments thereof.
- the binding agent is an antibody.
- the binding agent is a polypeptide such as an antibody comprising a heavy chain CDR amino acid sequence selected from one of SEQ ID NO.
- the binding agent is a polypeptide such as an antibody comprising a light chain CDR amino acid sequence selected from one of SEQ ID NO. 4 or a conservatively substituted variant thereof; one of SEQ ID NO. 5 or a conservatively substituted variant thereof; and, one of SEQ ID NO. 6 or a conservatively substituted variant thereof.
- the binding agent is a polypeptide such as an antibody comprising a V H amino acid sequence of any of SEQ ID NO. 7 or a conservatively substituted variant thereof.
- the binding agent is a polypeptide such as an antibody comprising a V L amino acid sequence of any of SEQ ID NO. 8 or a conservatively substituted variant thereof.
- this disclosure provides binding agents comprising a combination of a heavy chain variable region (VH) and a light chain variable region (VL) selected from the group consisting of: a VH comprising complementarity determining regions (CDRs) 1 , 2, and 3 as set forth in SEQ ID NOS: 1 , 2, and 3, respectively; and a VL comprising CDRs 1 , 2 and 3 as set forth in SEQ ID NOS: 4, 5, and 6, respectively; and/or a conservatively substituted variant thereof.
- VH heavy chain variable region
- VL light chain variable region
- this disclosure provides binding agents comprising a combination of a heavy chain variable region (VH) and a light chain variable region (VL) selected from the group consisting of: a VH comprising SEQ ID NO. 7 or a conservatively substituted variant thereof; and a VL comprising SEQ ID NO, 8, or a conservatively substituted variant thereof.
- the binding agents are antibodies such as monoclonal antibodies.
- the binding agents are human antibodies.
- this disclosure also provides isolated polynucleotides encoding such binding agent(s), and/or an expression vector, and/or host cell, comprising the polynucleotide.
- this disclosure also provides methods for expressing such a polynucleotide in order to express and produce said binding agent(s). In some embodiments, this disclosure provides methods for isolating said binding agent(s) and preparing compositions comprising the same. In some embodiments, this disclosure provides compositions comprising such binding agent(s), such as pharmaceutical compositions comprising at least one pharmaceutically acceptable carrier. In some embodiments, this disclosure provides methods for neutralizing HIV in vitro and/or in vivo. In some embodiments, this disclosure provides methods for administering the binding agents and/or polynucleotides to a subject such as a human being in order to, in some embodiments, treat and/or prevent infection by HIV or AIDS. Other embodiments are also contemplated by this disclosure as may be ascertained by those of ordinary skill in the art.
- the binding agents have specificity for an epitope comprising amino acid residues in the proximity of Q619, N677 and E500K at the gp120/gp41 interface of HIV-1 Env (corresponding residues underlined in SEQ ID NO. 17).
- a binding agent of this disclosure may comprise any one or more of these binding specificities along with the neutralization characteristics described above but not of a control virus at a concentration is from 10 2 -10° ug/ml, or between 10°-10 1 ug/ml, to at least about 50%, and/or the ability to the neutralize a majority of the 57 HIV-1 pseudoviruses listed in Figure 4 at an IC 5 o of less than 25.
- nucleic acids encoding such binding agents are also provided as in Table 4.
- the binding agent is derived from or related to (e.g., by sequence or derivation) a human antibody, human IgG, human lgG1 , human lgG2, human lgG2a, human lgG2b, human lgG3, human lgG4, human IgM, human IgA, human lgA1 , human lgA2, human IgD, human IgE, canine antibody, canine IgGA, canine IgGB, canine IgGC, canine IgGD, chicken antibody, chicken IgA, chicken IgD, chicken IgE, chicken IgG, chicken IgM, chicken IgY, goat antibody, goat IgG, mouse antibody, mouse IgG, pig antibody, and / or rat antibody, and / or a derivative thereof.
- a human antibody human IgG, human lgG1 , human lgG2, human lgG2a, human lgG2b, human lgG3,
- the derivative may be selected from the group consisting of an F ab , F ab2 , Fab’ single chain antibody, F v , single chain, mono-specific antibody, bispecific antibody, trimeric antibody, multi-specific antibody, multivalent antibody, chimeric antibody, canine-human chimeric antibody, canine-mouse chimeric antibody, antibody comprising a canine Fc, humanized antibody, human antibody, caninized antibody, CDR-grafted antibody, shark antibody, nanobody, and / or canelid antibody.
- the binding agent comprises at least a least a first and second specificity, the first being against HIV gp41 and the second being against a different antigen (e.g., an antigen of an infectious agent such as HIV (e.g., env) and / or a tumor antigen).
- a different antigen e.g., an antigen of an infectious agent such as HIV (e.g., env) and / or a tumor antigen.
- the binding agent and / or derivative thereof may comprise a detectable label fixably attached thereto.
- the binding agent of any one and / or derivative thereof comprises an effector moiety (e.g., a cytotoxic drug, toxin, diphtheria A chain, exotoxin A chain, ricin A chain, abrin A chain, curcin, crotin, phenomycin, enomycin, and radiochemical) fixably attached thereto.
- an effector moiety e.g., a cytotoxic drug, toxin, diphtheria A chain, exotoxin A chain, ricin A chain, abrin A chain, curcin, crotin, phenomycin, enomycin, and radiochemical fixably attached thereto.
- polynucleotides encoding one or more binding agents are also provided (e.g., as an expression vector). Host cells comprising and / or expressing the polypeptide products of such polynucleotides are also provided.
- compositions comprising at least one binding agent or derivative; at least one isolated polynucleotide; at least one expression vector; and / or, at least one host cell; or a combination thereof; and, a pharmaceutically acceptable carrier are also provided.
- This disclosure also provides methods for detecting HIV on a cell, the method comprising contacting a test biological sample with a binding agent or derivative described herein and detecting the binding agent bound to the biological sample or components thereof.
- Such methods may be an in vivo method or an in vitro method.
- the method may comprise comparing the amount of binding to the test biological sample or components thereof to the amount of binding to a control biological sample or components thereof, wherein increased binding to the test biological sample or components thereof relative to the control biological sample or components thereof indicates the presence of a cell expressing HIV polypeptides in the test biological sample (e.g., mammalian blood).
- a kit for detecting the expression of HIV in or on a cell comprising a binding agent or derivative thereof and instructions for use.
- the binding agent and / or derivative thereof is in lyophilized form.
- this disclosure provides methods for treating, preventing and / or ameliorating an infectious disease, cancer and / or autoimmunity in a mammal comprising administering to the mammal at least one effective dose of a pharmaceutical composition comprising a binding agent or derivative thereof.
- the infectious disease is human immunodeficiency virus (HIV).
- multiple doses are administered to the animal.
- the binding agent and / or derivative thereof may be administered in a dosage amount of about 1 to 50 mg / kg.
- a subject or a host is meant to be an individual.
- the subject can include domesticated animals, such as cats and dogs, livestock (e.g., cattle, horses, pigs, sheep, and goats), laboratory animals (e.g., mice, rabbits, rats, guinea pigs) and birds.
- livestock e.g., cattle, horses, pigs, sheep, and goats
- laboratory animals e.g., mice, rabbits, rats, guinea pigs
- the subject is a mammal such as a primate or a human.
- Optional or optionally means that the subsequently described event or circumstance can or cannot occur, and that the description includes instances where the event or circumstance occurs and instances where it does not.
- the phrase optionally the composition can comprise a combination means that the composition may comprise a combination of different molecules or may not include a combination such that the description includes both the combination and the absence of the combination (i.e., individual members of the combination).
- Ranges may be expressed herein as from about one particular value, and/or to about another particular value. When such a range is expressed, another aspect includes from the one particular value and/or to the other particular value. Similarly, when values are expressed as approximations, by use of the antecedent about or approximately, it will be understood that the particular value forms another aspect. It will be further understood that the endpoints of each of the ranges are significant both in relation to the other endpoint, and independently of the other endpoint. Ranges (e.g., 90-100%) are meant to include the range per se as well as each independent value within the range as if each value was individually listed.
- the term“combined” or“in combination” or“in conjunction” may refer to a physical combination of agents that are administered together or the use of two or more agents in a regimen (e.g., administered separately, physically and / or in time) for treating, preventing and / or ameliorating a particular disease.
- a given treatment for a given condition e.g., preventing cancer infection by HIV
- a treatment will be said to have prevented the condition if it is given during exposure of a patient to a stimulus that would have been expected to produce a given manifestation of the condition, and results in the patient's experiencing fewer and/or milder symptoms of the condition than otherwise expected.
- a treatment can“prevent” infection by resulting in the patient's displaying only mild overt symptoms of the infection; it does not imply that there must have been no penetration of any cell by the infecting microorganism.
- reduce, reducing, and reduction as used herein in connection with prevention, treatment and / or amelioration of a given condition by a particular treatment typically refers to a subject developing an infection more slowly or to a lesser degree as compared to a control or basal level of developing an infection in the absence of a treatment (e.g., administration of one or more HIV binding agents).
- a reduction in the risk of infection may result in the patient's displaying only mild overt symptoms of the infection or delayed symptoms of infection; it does not imply that there must have been no penetration of any cell by the infecting microorganism.
- Germinal center and Memory IgG B cells from donor SA090 were sorted separately according to IgG (i.e. IgA and IgM negative cells), CD19 and CD38 expression (germinal center B cells are CD38 positive, which is not present on memory B cells) ( Figure 2) and interrogated for the production of HIV-1 neutralizing antibodies.
- IgG memory B cells and IgG germinal cells were seeded in separate plates as single cell micro-cultures on human feeder cells in the presence of Epstein-Barr Virus (EBV) (which also stimulate polyclonally memory B cells) and a cocktail composed TLR9 agonist CpG-2006, IL-2 (1000 lU/ml), IL-6 (10 ng/ml), IL-21 (10 ng/ml), and anti-BCR goat antibodies (triggers BCR).
- EBV Epstein-Barr Virus
- TLR9 agonist CpG-2006 IL-2 (1000 lU/ml), IL-6 (10 ng/ml), IL-21 (10 ng/ml), and anti-BCR goat antibodies (triggers BCR).
- Supernatants from day 14 cultures were then tested in a primary screening using a 384-well based HIV-1 pseudoviruses neutralization assay (using in parallel two strains, CE1 176 and BJOX2000, representative of clade C and CRF07
- TZM-bl cells Neutralization assays were undertaken on TZM-bl cells.
- HIV-1 pseudoviruses that resulted in an output of 50-100 x 10 4 relative light units (RLU) were incubated with B cell culture supernatants for 1 h at 37% (5% C0 2 ) before the addition of 3000 TZM-bl cells. These were incubated for a further 72 h, after which supernatant was removed and 15 pi Steadylite reagent (Perkin Elmer) was added. Luciferase activity was detected 5 min later by reading the plates on a Synergy microplate luminometer (BioTek).
- the supernatants derived from germinal center B cells found to produce antibodies that cross-neutralize one or more of the HIV strains.
- the supernatants from these two cultures were further harvested and tested for their ability to neutralize pseudoviruses.
- the LN02 antibody was characterized by determining the amino acid and nucleotide sequences of its variable regions and the complementarity determining regions (CDRs) ascertained. Accordingly, the binding agent termed “LN02” is an lgG1-type fully human monoclonal antibody having the CDR, VH and VL sequences as shown above in Table 1. The LN02 antibody was also determined to be derived from the IGHV4-4*02 and IGLV3-21*01 germline genes and highly somatically mutated in variable genes of both heavy chain (31.2%) and kappa light chain (31.6%) compared to germ line.
- Recombinant LN02 antibody was produced and tested against the Global Panel of nine (9) HIV-1 reference pseudoviruses on TZM-bl cells. Surprisingly, lgG1 LN02 antibody neutralized seven out of nine HIV-1 pseudoviruses with IC 5 o values ranging from 1 mg/ml to less than 0.1 mg/ml ( Figure 3) and did not neutralize the negative control MLV pseudovirus. lgG1 LN02 antibody was subsequently tested on an extended panel of 57 HIV-1 pseudoviruses including clade A, clade A (T/F), clade B, clade B (T/F), clade BC, clade C, and clade C (T/F). As shown in Fig.
- the LN02 antibody broadly neutralized 43 viruses out of 57 with IC 5 o below 25 pg/ml, i.e., about 75% of the tested viruses, with a median IC 5 o of 0.046 pg/ml.
- the LN02 antibody broadly neutralized 36 of out of 57 with IC 8 o below 25 pg/ml, i.e., about 63% of the tested viruses, with a median IC 8 o of 0.13 pg/ml.
- LN02 binds selectively to the gp140 ConsB, which is the only protein construct that is predominantly trimeric in tertiary structure.
- LN02 has a unique footprint among interface mAbs
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Virology (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Tropical Medicine & Parasitology (AREA)
- Communicable Diseases (AREA)
- Oncology (AREA)
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- AIDS & HIV (AREA)
- Medicinal Chemistry (AREA)
- Molecular Biology (AREA)
- Organic Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Peptides Or Proteins (AREA)
Abstract
This disclosure relates to binding agents that, in some embodiments, comprise one or more of SEQ ID NOS. 1-8, or conservatively substituted variants thereof, with binding specificity for HIV, preferably neutralizing activity, and to methods for using the same to treat, prevent and / or ameliorate HIV infection and/or AIDS.
Description
HIV BINDING AGENTS
Related Applications
[001] This application claims priority to U.S. Ser. No. 62/697,462 filed on July 13, 2018, which is hereby incorporated into this application in its entirety.
Field of the Disclosure
[002] This disclosure relates to binding agents with specificity for human immunodeficiency virus (HIV), methods for making the same, and to methods for using the same to treat and / or prevent HIV infection.
Background of the Disclosure
[003] As we enter the fourth decade of the HIV epidemic, significant advances have been made in the understanding of HIV pathogenesis and in the development of potent and safe antiviral drugs. More than 30 antiviral drugs have been registered and the impact of combination antiretroviral therapy (ART) on both morbidity and mortality has been remarkable. However, despite the long-term suppression of HIV replication achieved in patients with optimal adherence to ART, HIV invariably rebounds after interruption of therapy. Furthermore, successful therapy does not induce or allow restoration/development of virus-specific immune responses capable of controlling HIV replication in the absence of ART. Thus, life-long ART is needed to control HIV replication and associated disease in the large majority of HIV infected subjects.
[004] A number of immunological interventions have been investigated in the past and currently being further developed with the goal to achieve HIV functional cure, wherein viral replication is suppressed without sustained antiviral therapy. Therapeutic vaccine strategies have been the primary intervention strategy investigated but the results have shown modest efficacy in experimental animal models and patients with the exception of a CMV-based vector HIV vaccine (50% efficacy in the NHP model). Recent studies have generated interesting results on the possibility of using anti-envelope broad neutralizing antibodies (bNabs) as therapeutic agents in HIV infection.
[005] There is a need in the art for additional reagents for targeting HIV, especially neutralizing antibodies, and methods for using the same. This disclosure addresses those needs by
providing reagents and methods that may be used to target HIV and cells and / or tissues infected by and /or harboring the same.
Brief Description of the Drawings
[006] In the following a brief description of the appended figures will be given. The figures are intended to illustrate the present invention in more detail. However, they are not intended to limit the subject matter of the invention in any way.
[007] Figure 1 shows the results of neutralization of a panel of nine (9) HIV-1 pseudoviruses from the Global Panel of HIV-1 reference strains by 107 plasma samples from chronically infected patients naive to antiretroviral therapy. MLV pseudovirus is used as a negative control. Boxed are the eight donors selected for the collection of lymph nodes to isolate potent broadly neutralizing antibodies. Highlighted with the arrow is donor SA090 who was selected for the isolation of the broadly neutralizing antibody described herein. Inhibitory dose values indicate the dilution of plasma capable of neutralizing 50% of viral infection (ID50).
[008] Figure 2 shows the gating and sorting strategy used to purify memory and germinal center IgG B cells from lymph node samples. B cells were selected for the expression of the surface marker CD19 and IgG B cells were negatively selected for the lack of IgA and IgM B cell receptor (BCR) expression. Germinal center B cells were further selected for the expression of the CD38 marker (that is absent on memory B cells).
[009] Figure 3 shows the results of neutralization of a panel of nine (9) HIV-1 pseudoviruses (and MLV as negative control) from the Global Panel of HIV-1 reference strains by different concentration (in pg/ml) of the monoclonal antibody LN02. Inhibitory concentration values indicate the concentration of monoclonal antibody capable of neutralizing 50% of viral infection (IC50). Error bars indicate the standard deviation of duplicates.
[0010] Figure 4 shows the results of neutralization of a multi-clade panel of 57 HIV-1 pseudoviruses by the monoclonal antibody LN02. IC50 values indicate the concentration of monoclonal antibody capable of neutralizing 50% of viral infection
[0011] Figure 5 shows the distribution of IC50 values on the whole panel of 57 viruses described in Figure 4 and on individual clades or circulating recombinant forms.
[0012] Figure 6 shows that the neutralization activity of LN02 is enhanced by cleavage site mutation E500K and a combination of all cleavage site mutations together (CS). IC50 values and fold change in neutralization activity relative to the WT CAP45 pseudo-virus is shown in
Table 5.
[0013] Figure 7 shows the binding, as assessed by surface plasmon resonance, of the LN02 monoclonal antibody to the soluble HIV-1 Env trimer 426c WT SOSIP gp140 that expresses multiple epitopes for broadly neutralizing antibodies (panel A, solid line). HIV-1 Env trimer that has been pre-incubated with unlabeled LN02 completely blocks the binding of a biotinylated LN02 that is detected with a streptavidin probe (panel A, dashed line). The PGT151 monoclonal antibody (binding to a site at the interface between gp120 and gp41) bound with high affinity to 426c WT SOSIP gp140 protein (panel B, solid line) but PGT151 binding is significantly reduced to the LN02 + 426c WT SOSIPgp140 complex.
[0014] Figure 8 shows the binding, as measured by ELISA, of LN02 antibody to a set of HIV-1 Env antigens and a negative control antigen (soluble CD4 protein, sCD4). LN02 only binds tightly to the gp140 ConsB, which is the only protein construct that is predominantly trimeric in tertiary structure.
[0015] Figure 9 is a comparison of the amino acid sequences of the HXB2 (SEQ ID NO.: 18) and CAP45 (SEQ ID NO. 17) Env polypeptides.
Summary of the Disclosure
[0016] This disclosure relates to binding agents with specificity for human immunodeficiency virus (HIV), methods for producing such binding agents, as well as methods for using such binding agents to treat, prevent and / or ameliorate HIV infection.
Detailed Description
[0017] This disclosure relates to binding agents having binding affinity for human immunodeficiency virus (HIV). In some embodiments, the binding agent can bind HIV antigens on viral particles per se or on the surface of cells in vitro and / or in vivo. The binding agents may also bind isolated HIV antigens and / or fragments and / or derivatives thereof, typically in vitro. Also provided are methods for using such binding agents to diagnose, treat, prevent and
/ or ameliorate one or more diseases associated with HIV. For instance, the binding agents may be antibodies (e.g., monoclonal antibodies) that may react with and / or bind to the epitopes of HIV or polypeptides thereof. The binding agents may be useful for treating disease caused by HIV, such as Acquired Immune Deficiency Syndrome (AIDS). In some embodiments, the binding agents described herein may selectively target and/or eliminate HIV and/or HIV-infected cells containing HIV (e.g., replication competent HIV) and/or expressing proteins thereof. In some embodiments, such cells may be reservoirs for replication competent HIV. In some embodiments, binding agents having, for instance, different specificities (e.g., recognizing different epitopes) may be combined to HIV activity such as infection, replication and/or spread to other cells. In some embodiments, the binding agents described herein may also provide for the selective elimination and / or suppression of HIV or HIV-expressing cells. In some embodiments, the binding agents described herein may be used to suppress and / or eliminate HIV and/or HIV-expressing cells to treat, for instance, HIV infection and/or AIDS. Other embodiments, uses and the like are described below.
[0018] The binding agents may be antibodies such as monoclonal antibodies. As shown in the examples herein, the techniques discussed below have been used to identify a fully human mAb termed“LN02”, having particular characteristics that are described herein and shown in the examples. The LN02 antibody was isolated and the amino acid sequences of variable heavy (VH) and light (VL) chain domains of said antibody determined. A binding agent such as LN02 may identified by referencing the amino acid and/or nucleic acid sequences corresponding to the variability and / or complementarity determining regions (“CDRs”) thereof. A CDR comprises amino acid residues within the variable region identified in accordance with the definitions of the Kabat, Chothia, the accumulation of both Kabat and Chothia, AbM, contact, and/or conformational definitions or any method of CDR determination well known in the art. antibody modeling software (now Accelrys®), or the“contact definition” of CDRs based on observed antigen contacts described by MacCallum et al. , 1996, J. Mol. Biol., 262:732-745. In the“conformational definition” of CDRs, the positions of the CDRs may be identified as the residues that make enthalpic contributions to antigen binding (Makabe et al., 2008, Journal of Biological Chemistry, 283:1 156-1166). Still other CDR boundary definitions may not strictly follow one of the above approaches, but may nonetheless overlap with at least a portion of the Kabat CDRs, although they may be shortened or lengthened in light of prediction or experimental findings that particular residues or groups of residues or even entire CDRs do not significantly impact antigen binding. As used herein, a CDR may refer to CDRs defined by any
approach known in the art, including combinations of approaches. The methods used herein may utilize CDRs defined according to any of these approaches. For any given embodiment containing more than one CDR, the CDRs may be defined in accordance with any of Kabat, Chothia, extended, AbM, contact, and/or conformational definitions.
[0019] The amino acid sequences of the three heavy chain CDRs (CDRH1 , CDRH2, CDRH3), three light chain CDRs (CDRL1 , CDRL2, CDRL3), VH and VL domains of LN02 (LN02_1), and certain exemplary variants thereof are shown in Table 1 below.
Table 1
A binding agent of this disclosure may comprise, for example, any one or more of the amino acid sequences shown in Table 1 (i.e. , any one or more of SEQ ID NOS. 1-8), and/or or a conservatively substituted variant thereof. Fragments and/or derivatives (e.g., comprising substituted amino acids, such as conservative substitutions) thereof are also disclosed. In some embodiments, a binding agent of this disclosure may comprise one or more (i.e., one, two, three, four, five, six or seven) of SEQ ID NOS. 1-8, provided the binding agent exhibits the functional characteristics described herein. In some embodiments, it is preferred that the binding agent comprises at least one of SEQ ID NO. 1 (CDRH1 region); at least one of SEQ ID NO. 2 (CDRH2 region); at least one of SEQ ID NO. 3 (CDRH3 region); at least one of SEQ I D NO. 4 (CDRL1 region); at least one of SEQ ID NO. 5 (CDRL2 region); and, at least one of SEQ ID NO. 6 (CDRL3 region).
[0020] In some embodiments, the binding agent may comprise any of SEQ ID NOS. 1-8 comprising one or more amino acid substitutions, in particular or preferably conservative substitutions (see, e.g., the substitutions listed in Table 2). In some embodiments, the binding agent may be a monoclonal antibody (mAb) or a fragment or derivative thereof. In some embodiments, the binding agent may be an HIV-binding fragment of such a monoclonal antibody (mAb). In some embodiments, the mAb, fragment or derivative thereof includes at least one or more of SEQ ID NOS. 1 (CDRH1 region), SEQ ID NO. 2 (CDRH2 region); SEQ ID NO. 3 (CDRH3 region); SEQ ID NO. 4 (CDRL1 region); SEQ ID NO. 5 (CDRL2 region); and/or, SEQ ID NO. 6 (CDRL3 region); and/or any one or more of the same comprising one or more amino acid substitutions, in particular or preferably conservatively substituted derivatives thereof (see, e.g., the substitutions listed in Table 2). An exemplary LN02 mAb is termed“lgG1 LN02” in which the LN02 variable regions (e.g., SEQ ID NOS. 7 and 8) were cloned into an lgG1 or lgG3 backbone using standard techniques. In some embodiments, then, the binding agent comprises SEQ ID NO. 7 and/or 8, preferably SEQ ID NOS. 7 and 8; as well as any of the same comprising one or more amino acid substitutions, in particular or preferably conservative substitutions (see, e.g., the substitutions listed in Table 2). Other suitable embodiments may be derived by those of ordinary skill in the art from this disclosure.
[0021] It is preferred that the binding agent (e.g., antibody, or the antigen binding fragment thereof), comprises one or more amino acid sequences having at least 70%, at least 75%, at least 80%, at least 85%, at least 88%, at least 90%, at least 92%, at least 95%, at least 96%, at least 97%, at least 98% or at least 99% identity to at least one of SEQ ID NOS. 1-8 (i.e. , the CDR sequences, the VH sequence and/or the VL sequence shown in Table 1). As discussed below, identities of less than 100% may result from the natural or synthetic substitution of one or more amino acids with another amino acid(s), as in a conservative substitution (see, e.g., Table 2). Various combinations of SEQ ID NOS. 1-8 may be useful as may ascertained by one of ordinary skill in the art using the techniques described herein or as may be otherwise available to those of ordinary skill in the art. In preferred embodiments, the binding agent binds HIV and/or cells infected by HIV and/or expressing HIV proteins. In some especially preferred embodiments, the binding agent neutralizes HIV as described herein. In preferred embodiments, the binding agent both binds HIV and/or cells infected by HIV and/or expressing HIV proteins and neutralizes HIV.
[0022] The variable region and/or CDR sequences (e.g., of Table 1) may be used in
combination with one or more other variable region / CDR amino acid sequences available to those of ordinary skill in the art. Such variable region / CDR amino acid sequences may alternatively and / or also be adjoined to one or more types of constant region polypeptides of an antibody molecule. For instance, the CDR amino acid sequences shown in Table 1 may be adjoined to or associated with the constant regions of any antibody molecule of the same or a different species (e.g., human, goat, rat, sheep, chicken) and / or antibody subtype of that from which the CDR amino acid sequence was derived. For instance, an exemplary binding agent may be, or may be derived from, one having about the same neutralizing activity and/or binding the same or similar epitopes and/or exhibiting about the same affinity as another binding agent comprising one or more of the amino acid sequences shown in Table 1. The binding agent may comprise an antibody heavy and / or a light chain that each comprises one or more constant and / or variable regions. Any of the amino acid sequences described herein (e.g., the sequences shown in Table 1), and / or any fragments and / or derivatives thereof, may also be combined with any other variable region and / or CDR in any order and / or combination to form new binding agents, e.g., hybrid and / or fusion binding agents, and / or inserted into other heavy and / or light chain variable regions using standard techniques.
[0023] This disclosure also provides for the use of such binding agents to isolate, identify, and / or target HIV and / or cells harboring and/or infected by HIV and / or expressing HIV antigens. In certain embodiments, such binding agents may be reactive against HIV antigens such as proteins expressed on the surface of cells. In some embodiments, the binding agent(s) is an antibody (antibodies). The term "antibody" or“antibodies” may refer to whole or fragmented antibodies in unpurified or partially purified form (e.g., hybridoma supernatant, ascites, polyclonal antisera) or in purified form. The antibodies may be of any suitable origin or form including, for example, murine (e.g., produced by murine hybridoma cells), or expressed as humanized antibodies, chimeric antibodies, human antibodies, and the like. For instance, antibodies may be wholly or partially dervied from human (e.g., IgG (lgG1 , lgG2, lgG2a, Ig2b, lgG3, lgG4), IgM, IgA (lgA1 and lgA2), IgD, and IgE), canine (e.g., IgGA, IgGB, IgGC, IgGD), chicken (e.g., IgA, IgD, IgE, IgG, IgM, IgY), goat (e.g., IgG), mouse (e.g., IgG, IgD, IgE, IgG, IgM), pig (e.g., IgG, IgD, IgE, IgG, IgM), and / or rat (e.g., IgG, IgD, IgE, IgG, IgM) antibodies, for instance. Methods of preparing, utilizing and storing various types of antibodies are well- known to those of skill in the art and would be suitable in practicing the present invention (see, for example, Harlow, et al. Antibodies: A Laboratory Manual, Cold Spring Harbor Laboratory, 1988; Harlow, et al. Using Antibodies: A Laboratory Manual, Portable Protocol No. 1, 1998;
Kohler and Milstein, Nature, 256:495 (1975)); Jones et al. Nature, 321 :522-525 (1986); Riechmann et al. Nature, 332:323-329 (1988); Presta (Curr. Op. Struct. Biol., 2:593-596 (1992); Verhoeyen et al. (Science, 239:1534-1536 (1988); Hoogenboom et al., J. Mol. Biol., 227:381 (1991); Marks et al., J. Mol. Biol., 222:581 (1991); Cole et al., Monoclonal Antibodies and Cancer Therapy, Alan R. Liss, p. 77 (1985); Boerner et al., J. Immunol., 147(1):86-95 (1991); Marks et al., Bio/Technology 10, 779-783 (1992); Lonberg et al., Nature 368 856-859 (1994); Morrison, Nature 368 812-13 (1994); Fishwild et al., Nature Biotechnology 14, 845-51 (1996); Neuberger, Nature Biotechnology 14, 826 (1996); Lonberg and Huszar, Intern. Rev. Immunol. 13 65-93 (1995); as well as U.S. Pat. Nos. 4,816,567; 5,545,807; 5,545,806; 5,569,825; 5,625,126; 5,633,425; and, 5,661 ,016). In certain applications, the antibodies may be contained within hybridoma supernatant or ascites and utilized either directly as such or following concentration using standard techniques. In other applications, the antibodies may be further purified using, for example, salt fractionation and ion exchange chromatography, or affinity chromatography using Protein A, Protein G, Protein A/G, and / or Protein L ligands covalently coupled to a solid support such as agarose beads, or combinations of these techniques. The antibodies may be stored in any suitable format, including as a frozen preparation (e.g., -20°C or -70°C), in lyophilized form, or under normal refrigeration conditions (e.g., 4°C). When stored in liquid form, for instance, it is preferred that a suitable buffer such as Tris-buffered saline (TBS) or phosphate buffered saline (PBS) is utilized. In some embodiments, the binding agent may be prepared as an injectable preparation, such as in suspension in a non-toxic parenterally acceptable diluent or solvent. Suitable vehicles and solvents that may be utilized include water, Ringer's solution, and isotonic sodium chloride solution, TBS and / or PBS, among others. Such preparations may be suitable for use in vitro or in vivo may be prepared as is known in the art and the exact preparation may depend on the particular application.
[0024] The binding agents described herein are not, however, in any way limited to antibodies (i.e., whole antibodies). For example, the binding agent may be any compound exhibiting similar binding properties as another (e.g., a mimetic). For example, an exemplary binding agent may be one that binds HIV and/or can compete with another binding agent having specificity therefor (e.g., a monoclonal antibody such as LN02). In some embodiments, the mimetic may exhibit substantially the same affinity in binding assays as the binding agent (e.g., monoclonal antibody) to which it is being compared. The affinity of a particular binding agent may be measured by any suitable assay including but not limited to FACS staining of cell
surface HIV antigens (e.g., polypeptides). One binding agent may be said to have “substantially the same affinity” as another where the measurements (e.g., nm) are within about any of 1-20, 1-5, 5-10, 10-15, or 15-20 percent of one another. Exemplary mimetics may include, for example, organic compounds that specifically bind HIV, or an affibody (Nygren, et al. FEBS J. 275 (11): 2668-76 (2008)), affilin (Ebersbach, et al. J. Mol. Biol. 372 (1): 172-85 (2007)), affitin (Krehenbrink, et al. J. Mol. Biol. 383 (5): 1058-68 (2008)), anticalin (Skerra, A. FEBS J. 275 (1 1): 2677-83 (2008)), avimer (Silverman, et al. Nat. Biotechnol. 23 (12): 1556- 61 (2005)), DARPin (Stumpp, et al. Drug Discov. Today 13 (15-16): 695-701 (2008)), Fynomer (Grabulovski, et al. J. Biol. Chem. 282 (5): 3196-3204 (2007)), Kunitz domain peptide (Nixon, et al. Curr. Opin. Drug Discov. Devel. 9 (2): 261-8 (2006)), and / or a monobody (Koide, et al. Methods Mol. Biol. 352: 95-109 (2007)). Other mimetics may include, for example, a derivative of an antibody such as, for example, an Fab, Fab2, Fab’ single chain antibody, Fv, single domain antibody, mono-specific antibody, bi-specific antibody, tri-specific antibody, multi-valent antibody, chimeric antibody, canine-human chimeric antibody, canine-mouse chimeric antibody, antibody comprising a canine Fc, humanized antibody, human antibody, caninized, CDR-grafted antibody (i.e., comprising any of SEQ ID NOS. 1-8 shown in Table 1), shark antibody, nanobody, camelid antibody, microbody, and / or intrabody; and/or derivative thereof. Other binding agents are also provided herein as would be understood by one of ordinary skill in the art.
[0025] Any method known to those of ordinary skill in the art may be used to generate binding agents having specificity for (e.g., binding to) HIV. For instance, to generate and isolate monoclonal antibodies an animal such as a mouse may be administered (e.g., immunized) with one or more HIV proteins. Animals exhibiting serum reactivity to HIV expressed on activated human T lymphocytes (as determined by, for instance, flow cytometry and / or microscopy) may then be selected for generation of anti-HIV hybridoma cell lines. This may be repeated for multiple rounds. Screening may also include, for instance, affinity binding and / or functional characterization to identify the binding agent as being specific for HIV. In some embodiments, such as in the Examples herein, human beings may be screened for the expression of antibodies against HIV. In some embodiments, plasma samples of human beings infected by HIV may be screened to identify persons expressing anti-HIV antibodies, and in particular, neutralizing antibodies. Neutralizing antibody-producing cells of such persons may then be isolated, followed by the isolation and characterization of the antibodies produced thereby (e.g., as in the examples herein). A neutralizing antibody may be one that exhibits the ability to
neutralize, or inhibit, infection of cells by HIV. In general, a neutralization assay typically measures the loss of infectivity of the virus through reaction of the virus with specific antibodies. Typically, a loss of infectivity is caused by interference by the bound antibody with any of the virus replication steps including but not limited to binding to target cells, entry, and/or viral release. The presence of un-neutralized virus is detected after a predetermined amount of time, e.g., one, two, three, four, five, six, seven, eight, nine, 10, 12 or 14 days, by measuring the infection of target cells using any of the systems available to those of ordinary skill in the art (e.g., a luciferase-based system). A non-limiting example of a neutralization assay may include combining a given amount of a virus or pseudovirus (see below) and different concentrations of the test or control (typically positive and negative controls assayed separately) antibody or antibodies are mixed under appropriate conditions (e.g., one (1) hour at room temperature) and then inoculated into an appropriate target cell culture (e.g., TZM-bl cells). For instance, binding agent-producing cells (e.g., B cells producing antibodies) may be assayed for the production of HIV-1 neutralizing antibodies by seeding such cells in separate plates as single cell micro-cultures on human feeder cells in the presence of Epstein-Barr Virus (EBV) (which also stimulate polyclonally memory B cells), a cocktail of growth factors (e.g., TLR9 agonist CpG-2006, IL-2 (1000 lU/ml), IL-6 (10 ng/ml), IL-21 (10 ng/ml), and anti-B cell receptor (BCR) goat antibodies (which trigger BCRs). After an appropriate time (e.g., 14 days), supernatants of such cultures may tested in a primary luciferase-based screening system using two or more representative HIV-1 viruses or pseudoviruses that productively infect such cells. The pseudoviruses may be incubated with B cell culture supernatants for an appropriate time and temperature (e.g., one (1) h at 37% (5% C02)) before the addition of host cells (e.g., 3000 TZM-bl cells). Incubation for an appropriate time (e.g., 72 hours) may then follow, after which the supernatant may be removed and Steadylite reagent (Perkin Elmer) added (e.g., 15 pi). Luciferase activity may then deteremined (e.g., five minutes later) on a Synergy microplate luminometer (BioTek). Decreased luciferase activity relative to a negative control typically indicates virus neutralization. Neutralization assays such as these, suitable for analyzing binding agents of this disclosure, are known in the art (see, e.g., Montefiori, D.C. Curr. Protocol. Immunol. Chapter 12, Unit 12.1 1 (2005); Edmonds, et al. Virology, 408(1): 1-13 (2010); Seaman, et al. J. Virol. 84(3): 1439-1452 (2010); Pace, et al. PNAS USA, 1 10(33): 13540-13545 (2013)). In some embodiments, test samples may be screened for the presence of antibodies able to neutralize a panel of HIV pseudoviruses (e.g., nine (9) HIV-1 pseudoviruses from the Global Panel of HIV-1 reference strains as conducted in the examples herein (those pseudoviruses being TRO.1 1 (B), 246F3 (AC), BJOX2000 (CRF007_BC),
CE1176 (C), CE0217 (C), CH1 19 (CRF07_BC), CNE55 (CRF01_AE), 25710 (C), and X1632 (see, e.g., Figs. 1 (showing the inhibitory dose (ID)) and 3); DeCamp, A. et al. Global panel of HIV-1 Env reference strains for standardized assessments of vaccine-elicited neutralizing antibodies. J Virol 88, 2489-2507 (2014)). Neutralization of a larger panel of psuedoviruses may also be tested; for instance, de Camp et al. describe a group of 12 pseudoviruses (also known as HIV-1 Env Reference Strains): 398F1 , 25710, CNE8, TR01 1 , X2278, BJOX2000, X1632, CE1176, 246F3, CH1 19, CE0217, and CNE55. In some embodiments, a panel of ten HIV isolates may be tested and a bNab may be identified as one that neutralizes six, seven, eight, nine members of a panel of nine pseudoviruses; or six, seven, eight, nine, 10, 1 1 or 12 members of a panel of 12 pseudoviruses. Screening of larger panels of such pseudoviruses (e.g., a panel of 57 pseudoviruses as in the examples herein) may also be carried out. In one embodiment, then, an exemplary panel of 57 pseudoviruses used in the examples against which test samples may be tested for neutralizing antibodies may include, for instance, those shown in Fig. 4 (including clade A, clade A (T/F), clade B, clade B (T/F), clade BC, clade C, and/or clade C (T/F)). In some embodiments, neutralization may be determined as a measure of the concentration (e.g., pg/ml) of monoclonal antibody capable of neutralizing any of about 50%, 60%, 70%, 80%, 90%, 95%, or 99% of viral infection (as may be measured by percent neutralization and/or by determining an“IC5o” and/or“IC8o” value). In some embodiments, a binding agent may be considered neutralizing if it is able to neutralize 50% of viral infection at a concentration of, for instance, about any of 105, 104, 103, 102, 10 1 , 10°, 101 , 102, or 103 pg/ml (e.g., an IC5o value as shown in in Figs. 3-5). In some embodiments, the ability of a neutralizing antibody to neutralize viral infection may be expressed as a percent neutralization (e.g., 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 99% (e.g., as in Fig. 3)). And in some embodiments, as in the Examples herein, the ability of a neutralizing antibody to neutralize viral infection may be expressed as, and, in preferred embodiments, the IC5o and/or ICso value is below 25 pg/ml, and is even more preferably below about any of 15, 10, 5, 2, 1 , 0.5, 0.25, 0.1 , 0.05, or 0.01 pg/ml (see, e.g., Fig. 4). Other measures of neutralization may also be suitable as may be determined by those of ordinary skill in the art.
[0026] In some embodiments, the binding agents described herein may be broadly neutralizing antibodies (bNabs) identified in biological samples (e.g., plasma) obtained from HIV-infected persons. As mentioned above and shown in the examples herein, such bNabs may be identified by testing plasma samples of patients chronically infected by HIV (preferably those naive to antiretroviral therapy) for the ability to neutralize multi-clade HIV isolates (e.g., initially
using a nine or 12-member panel and then a larger panel (e.g., 57 members) of pseudoviruses)). In some embodiments, the samples may be derived from patients known to be“Elite Controllers” with viremia <50 HIV RNA copies per ml of plasma. Screening procedures such as these may result in the identification of patients that may serve as lymph node donors for the subsequent isolation and characterization of B cells producing bNabs. In carrying out such screening assays, neutralizing activity is typically compared to a negative control such as murine leukemia virus (MLV) pseudovirus.
[0027] In some embodiments, germinal center and memory IgG B cells of patients expressing neutralizing binding agents (e.g., antibodies) may be isolated and further studied. In some embodiments, the cells may be sorted separately according to IgG (e.g., IgA and IgM negative cells), CD19, and CD38 expression (germinal center B cells are CD38 positive) (see, e.g., Figure 2) and interrogated for the production of HIV-1 neutralizing antibodies. For instance, highly pure IgG memory B cells and IgG germinal cells may be seeded in separate plates as single cell micro-cultures on human feeder cells in the presence of Epstein-Barr Virus (EBV) (which also stimulate polyclonally memory B cells) and a cocktail of growth factors and the like (e.g., composed TLR9 agonist CpG-2006, IL-2 (1000 lU/ml), IL-6 (10 ng/ml), IL-21 (10 ng/ml), and anti-BCR goat antibodies (B cell receptor (BCR) triggering)). Supernatants of such cultures (e.g., from day 14 cultures) may then be tested in a primary screening (e.g., using a 384-well based HIV-1 pseudoviruses neutralization assay using in parallel two strains, CE1 176 and BJOX2000, representative of clade C and CRF07, as shown in the examples herein). Neutralization assays may be carried out using any suitable host cells (e.g., TZM-bl cells (Seaman, et al. J. Virol. 84(3): 1439-52 (2010); NIH AIDS Reagent Program Catalog Number 8129)). HIV-1 pseudoviruses resulting in a significant output relative light units (RLU) (e.g., of 50-100 x 104 RLU) (i.e. , indicating productive infection of cells) may then incubated with B cell culture supernatants for an appropriate time and temperature (e.g., one (1) h at 37% (5% C02)) before the addition of host cells (e.g., 3000 TZM-bl cells). Incubation for an appropriate time (e.g., 72 hours) typically follows, after which the supernatant may be removed and Steadylite reagent (Perkin Elmer) added (e.g., 15 pi). Luciferase activity may then be detected (e.g., five minutes later) on a Synergy microplate luminometer (BioTek). Decreased luciferase activity indicates a lesser amount of virus being released by the cells and virus neutralization. For instance, if the base RLU for a particular pseudovirus is 50-100 x 104 RLU, a neutralizing antibody may be determined to decrease the RLU for that pseudovirus to 25-50 x 104 RLU (i.e., a 50% decrease), or less. Using such systems, supernatants capable of cross-neutralizing
strains may be identified, further harvested, and tested for their ability to neutralize other pseudoviruses.
[0028] The antibodies derived from such neutralizing antibody-containing cultures may then be further characterized by determining the amino acid and nucleotide sequences of the antibody variable and complementarity determining regions (CDRs) regions. Using these techniques, the HIV-neutralizing binding agent termed“LN02” was identified as an lgG3-type fully human monoclonal antibody having the CDR, VH and VL sequences shown in Table 1 (SEQ ID NOS. 1-8). LN02 was determined to be derived from the IGHV4-4*02 and IGLV3- 21*01 germline genes and highly somatically mutated in variable genes of both heavy chain (31.2%) and kappa light chain (31.6%) compared to germ line. In some embodiments, the variable heavy chain (VH) and variable light chain (VL) genes of a binding agent may then be cloned into an IgG expression vector of the same or a different isotype. As shown in the examples, for instance, nucleic acids encoding LN02 CDRs (Table 1) were cloned into lgG1 backbone, and the recombinant lgG1-based antibody (lgG1 LN02) was produced by transfecting appropriate host cells (e.g., Expi293F cells). The antibody full-length lgG1-based antibody may then be purified using standard techniques (e.g., a full-length lgG1-based antibody may be purified using a recombinant protein-A column (GE-Healthcare)). The recombinantly-produced lgG1 antibody may then be tested against any of a panel of pseudoviruses such as any of those described herein (e.g., the Global Panel of nine (9) HIV-1 reference pseudoviruses used in the examples) on an appropriate host cell (e.g., TZM-bl cells). In preferred embodiments, the binding agent will exhibit the ability to neutralize a majority (i.e., at least about 50% or greater) of the pseudovirus panel members (e.g., comprising nine, 12 or 1 18 members) without neutralizing a negative control virus (e.g., MLV pseudovirus). It is preferred that the binding agent exhibit the ability to neutralize a majority of such viruses (e.g., neutralization of greater than about 50%, such as any of about 60%, 70%, 80%, 90%, 95%, 99%, or 100%) with IC5o and/or IC8o values considered neutralizing (see below). For example, in some embodiments, a binding agent of this disclosure may exhibit neutralization of HIV-1 pseudoviruses BJOX (CRF07_BC), CE1 176, TRO.11 (B), X1632 (G), CH1 19 (CRF07_BC), CNE55 (CRF01_AE), 25710 (C), CD0217(C), but not of the control virus SVA-MLV at about 10° pg/ml or less (e.g., Figure 3). In some embodiments, neutralization of the HIV-1 pseudoviruses viruses may be observed where the antibody concentration is from 102-10° pg/ml, or between 10°-101 pg/ml (Figure 3). In some such embodiments, the percent neutralization by the binding agent is at least about 50% (Figure 3). In some embodiments,
infection of one HIV-1 isolate is considered neutralized by a binding agent (e.g., antibody) at an IC50 and/or ICso of less than 25 mg/ml, if infection of at least one isolate of this isolate is neutralized with an IC50 of less than 25 mg/ml. In some embodiments, the binding agent may be considered neutralizing where a majority of the 57 HIV-1 pseudoviruses listed in Figure 4 are considered neutralized at an IC50 of less than 25 pg/ml, such as about 10 pg/ml, 9 pg/ml, 8 pg/ml, 7 pg/ml, 6 pg/ml, 5 pg/ml, 4 pg/ml, 3 pg/ml, 2 pg/ml, 1 pg/ml, 0.9 pg/ml, 0.8 pg/ml, 0.7 pg/ml, 0.6 pg/ml, 0.5 pg/ml, 0.4 pg/ml, 0.3 pg/ml, 0.2 pg/ml, 0.1 pg/ml, 0.09 pg/ml, 0.08 pg/ml, 0.07 pg/ml, 0.06 pg/ml, 0.05 pg/ml, 0.04 pg/ml, 0.03 pg/ml, 0.02 pg/ml, or 0.01 pg/ml. In preferred embodiments, the binding agent may neutralize HIV-1 pseudovirus strains of ID MS208.A1 , Q23.17, Q769.d22, Q842.d12, Q259.d2.17, 0260.v5.c36, 191955_A11 , 191084 B7-19, TR0.11 , 6535.3, REJ04541.67, SC422661.8, QH0692.42, TRJ04551.58, RHPA4259.7, PVO.4, SC05 8C1 1 2344, CNE17, CNE19, CNE20, CNE21 , Du422.1 , CAP210.2.00.E8, ZM249M.PB6, HIV-001428-2.42, ZM214M.PL15, CAP45.2.00.G3, Ce704809221_1 B3, Ce1 176_A3, ZM247v1 (Rev-), Ce0682_E4, 249M B10, 246F C1G, and/or BF1266.431a at an IC50 of less than or about 1 pg/ml (Fig. 4). In some preferred embodiments, the binding agent may neutralize HIV-1 pseudovirus strains of ID MS208.A1 , Q23.17, Q769.d22, Q842.d12, Q259.d2.17, 0260.v5.c36, 191084 B7-19, TR0.11 , REJ04541.67, SC422661.8, QH0692.42, TRJ04551.58, RHPA4259.7, SC05 8C1 1 2344, CNE19, CNE20, CNE21 , CAP210.2.00.E8, HIV-001428-2.42, ZM214M.PL15, CAP45.2.00.G3,
Ce704809221_1 B3, Ce1 176_A3, Ce0682_E4, and/or 246F C1 G at an IC50 of less than or about 0.05 pg/ml (Fig. 4). In preferred embodiments, the binding agent may neutralize HIV-1 pseudovirus strains of ID MS208.A1 , Q23.17, Q769.d22, Q842.d12, Q259.d2.17, 0260.v5.c36, 191084 B7-19, TR0.1 1 , 6535.3, REJ04541.67, SC422661.8, QH0692.42, TRJ04551.58, RHPA4259.7, PVO.4, SC05 8C1 1 2344, CNE19, CNE20, CNE21 , Du422.1 , HIV-001428-2.42, ZM214M.PL15, CAP45.2.00.G3, Ce704809221_1 B3, Ce1 176_A3, Ce0682_E4, and/or 246F C1 G, at an IC8o of less than or about 1 pg/ml (Fig. 4). It is further preferred that the binding agent not exhibit clade-dependency. For instance, in some embodiments, the binding agent may exhibit the ability to neutralize pseudoviruses of HIV-1 Clades A, A(T/F), B, B (T/F), BC, C, and C (T/F) (Fig. 4). In some preferred embodiments, the binding agent may neutralize at least one pseudovirus in each of clades A, A(T/F), B, B (T/F), BC, C, and C (T/F) at at an IC50 of less than or about 1 pg/ml (Fig. 4). In some preferred embodiments, the binding agent may neutralize at least one pseudovirus in each of clades A, A(T/F), B, B (T/F), BC, C, and C (T/F) at an IC50 of less than or about 0.5 pg/ml (Fig. 4). In some preferred embodiments, the binding
agent may neutralize at least one pseudovirus in each of clades A, A(TVF), B, B (T/F), BC, C, and C (T/F) at an IC8o of less than or about 1 pg/ml (Fig. 4). In some embodiments, the binding agent comprises any one or more of these properties and one or more of SEQ ID NOS. 1-8, preferably at least one of SEQ ID NO. 1 (CDRH1 region); SEQ ID NO. 2 (CDRH2 region); SEQ ID NO. 3 (CDRH3 region); SEQ ID NO. 4 (CDRL1 region); SEQ ID NO. 5 (CDRL2 region); and, at least one of SEQ ID NOS. 6 (CDRL3 region); and/or, at least one of SEQ ID NO. 7, and/or SEQ ID NO. 8 (LN02 variable region); and/or fragments and/or derivatives thereof; as well as any of the same comprising one or more amino acid substitutions, in particular or preferably conservative substitutions (see, e.g., the substitutions listed in Table 2). These are characteristics of LN02-type binding agents, such as but not limited to lgG1 LN02 (“LN02”), as shown in Fig. 4.
[0029] In some embodiments, the binding agents may be tested for neutralization capacity against HIV reference pseudoviruses (e.g., the above-described Global Panel of nine (9) HIV- 1 reference pseudoviruses) using cells expressing or not expressing one or more types of Fc receptors (e.g., parental TZM-bl cells and TZM-bl cells expressing Fc-gamma receptor I (CD64) as in the examples; see e.g. Perez, et al. Utilization of immunoglobulin G Fc receptors by human immunodeficiency virus type 1 : a specific role for antibodies against the membrane- proximal external region of gp41. J Virol 83, 7397-7410 (2009); NIH AIDS Reagent Program Catalog No. 1 1798). Enhanced neutralizing activity in cells expressing Fc receptors may provide antibodies a kinetic advantage for virus inhibition. This kinetic advantage could be unique to antibodies, whose epitopes are thought to be difficult to access or exposed for only a short time on intermediate conformations of the Env protein during an early stage of fusion. Fc-gamma receptors could also potentially facilitate HIV-1 neutralization is phagocytosis, thereby increasing neutralization capacity of the antibodies. To this point, HeLa cells, from which the TZM-bl cell line was constructed, are known to exhibit properties of nonprofessional phagocytes. Thus, it is possible that TZM-bl cells were converted to professional phagocytic cells by introducing Fc-gamma receptor on their surface. Any Fc-gamma-receptor-mediated antiviral effects on HIV-1 neutralizing antibodies, whether by entry inhibition or phagocytosis, might be beneficial in HIV treatment and vaccine regimens. Fc-gamma receptors are rarely expressed on CD4+ lymphocytes, but several other HIV-1 -susceptible cell types express multiple Fc-gamma receptors and are involved in sexual transmission and the early establishment of long-lived viral reservoirs. In particular, macrophages are among the first infection-susceptible cells that the virus encounters after mucosal exposure, and are thought
to serve as a long-lived virus reservoir in chronic infection. Macrophages, as well as certain subsets of monocytes and dendritic cells, are known to express multiple Fc-gamma receptors. It is also important to mention that Fc-gamma receptors play a role in regulating adaptive immunity and peripheral tolerance, by facilitating antigen uptake, antigen presentation, cell activation and B cell tolerance. Thus, in some embodiments, the binding agents described herein may be used in conjunction with agents that induce and/or enhance Fc receptor expression, including the introduction of nucleic acids encoding one or more Fc receptors with or in conjunction with treatment by the binding agents described herein.
[0030] The specificity of the binding agents described herein may be determined using any of the many techniques available to those of ordinary skill in the art. For instance, as shown in the examples herein, the specificity of a binding agent (e.g., lgG1 LN02 antibody), with respect to particular epitopes, may be ascertained using a panel of pseudoviruses (e.g., CAP45) that encode mutations in the HIV envelope gene. Mutations summarized in Table 5 show that neutralization activity for LN02 is lost (>10 ug/ml) when the Q619Y substitution is introduced into the gp120/gp41 interface region of HIV-1 Env (Table 5). In the same region of Env, the cleavage site mutation E500K results in a 5-fold improvement in neutralization activity of LN02 (Fig. 6). Mutation of the N-glycan at position 88 in Env with an N88A substitution resulted in an 8.4- to 22-fold improvement potency for different viruses (Table 6). This observation is consistent with LN02 binding at the interface region of Env since the glycan is able to shield the interface from antibody binding. The HIV-1 envelope amino acid sequence for CAP45 is shown below (GenBank Accession No EF203962; NCBI GenPept Accession No. ABQ02701.1 ; SEQ ID NO.: 67) with the mutated resides indicated in bold and underlined (Fig. 9). The amino acid substitutions described above and in Table 5 refer to the HXB2 Env sequence (SEQ ID NO.: 68 (GenBank Accession No. MF944225.1 , protein_id=ATG88205.1)) numbering as a reference (Fig. 9). Mutated residues in CAP45 (SEQ ID NO. 17) described in Table 5 are indicated in bold and underlined, as are with the corresponding residues in the HXB2 Env sequence (SEQ ID NO. 18) below:
1 MGVRGILGNW PQWWIWSILG FWMLIICRVM GNLWVTVYYG VPVWKEAKAT LFCASDARAY
61 EKEVHNVWAT HACVPTDPNP QEIYLGNVTE NFNMWKNDMV DQMHEDI I SL WDQSLKPCVK
121 LTPLCVTLRC TNATINGSLT EEVKNCS FNI TTELRDKKQK AYALFYRPDV VPLNKNSPSG
181 NSSEYILINC NTSTITQACP KVSFDPIPIH YCAPAGYAIL KCNNKTFNGT GPCNNVSTVQ
241 CTHGIKPWS TQLLLNGSLA EEDIIIKSEN LTNNIKTIIV HLNKSVEIVC RRPNNNTRKS
301 IRIGPGQAFY ATNDIIGDIR QAHCNINNST WNRTLEQIKK KLREHFLNRT IEFESPSGGD
361 LEVTTHSFNC GGEFFYCNTT RLFKWSSNVT NDTITIPCRI KQFINMWQGV GRAMYAPPI E
421 GNITCNSSIT GLLLTRDGGK TDRNDTEI FR PGGGNMKDNW RNELYKYKW El KPLGVAPT
481 EARRRWERE KRAVGIGAVL LGFLGAAGST MGAAS ITLTV QARQLLSGIV QQQSNLLRAI
541 EAQQHMLQLT VWGIKQLQTR VLAIERYLKD QQLLGLWGCS GKLICTTNVP WNSSWSNKSQ
601 TDIWDNMTWI QWDREI SNYS NTI YKLLEDS QNQQEQNEKD LLALDSWNNL WNWFNITNWL
661 WYIKIFIMII GGLIGLRIIL GVLSIVKRVR QGYSPLSFQT LTPNPRGLDR LGRIEEEGGE
721 QDKDRSIRLV NGFLALAWED LRSLCLFSYH RLRDFI LIAV RAVELLGSSS LRGLQRGWEA
781 LKYLGSLLQY WGLELKKSAI NLLDTVAIAV AEGTDRIIEL IQRICRAIRN IPRRIRQGFE
841 AALL (CAP4; ; SEQ ID N< ) . 17 )
1 MRVKEKYQHL WRWGWRWGTM LLGMLMI CSA TEKLWVTVYY GVPVWKEATT TLFCASDAKA
61 YDTEVHNVWA THACVPTDPN PQEWLVNVT ENFNMWKNDM VEQMHEDI I S LWDQSLKPCV
121 KLTPLCVSLK CTDLKNDTNT NSSSGRMIME KGEIKNCSFN ISTSIRGKVQ KEYAFFYKLD
181 11 PI DNDTTS YTLTSCNTSV ITQACPKVSF EPI PIHYCAP AGFAILKCNN KTFNGTGPCT
241 NVSTVQCTHG IRPWSTQLL LNGSLAEEEV VIRSVNFTDN AKTI IVQLNT SVEINCTRPN
301 NNTRKKI RIQ RGPGRAFVTI GKI GNMRQAH CNI SRAKWNA TLKQIASKLR EQFGNNKTI I
361 FKQS SGGDPE IVTHSFNCGG EFFYCNSTQL FNSTWFNSTW STEGSNNTEG SDTITLPCRI
421 KQFINMWQEV GKAMYAPPIS GQIRCSSNIT GLLLTRDGGN NNNGSEI FRP GGGDMRDNWR
481 SELYKYKWK IEPLGVAPTK AKRRWQREK RAVGIGALFL GFLGAAGSTM GAASMTLTVQ
541 ARQLLSGIVQ QQNNLLRAIE AQQHLLQLTV WGIKQLQARI LAVERYLKDQ QLLGIWGCSG
601 KLICTTAVPW NASWSNKSLE QIWNHTTWME WDREINNYTS LIHSLIEESQ NQQEKNEQEL
661 LELDKWASLW NWFNITNWLW YIKLFIMIVG GLVGLRIVFA VLSWNRVRQ GYSPLSFQTH
721 LPI PRGPDRP EGI EEEGGER DRDRSIRLVN GSLALIWDDL RSLCLFSYHR LRDLLLIVTR
781 IVELLGRRGW EALKYWWNLL QYWSQELKNS AVSLLNATAI AVAEGTDRVI EWQGACRAI
841 RHIPRRIRQG LERILL (HX: S2 ; SEQ ID ] iO . 18 )
In some embodiments, a binding agent of this disclosure may comprise these binding specificities along with the neutralization characteristics described above (i.e., neutralization of HIV-1 pseudoviruses BJOX (CRF07_BC), CE1 176, TR0.1 1 (B), X1632 (G), CH1 19 (CRF07_BC), CNE55 (CRF01_AE), 25710 (C), CD0217(C) but not of the control virus SVA- MLV at a concentration is from 102-10° ug/ml, or between 10°-101 ug/ml, to at least about 50% (Figs. 3 and 4), as well as the neutralization of a majority of the 57 HIV-1 pseudoviruses listed in Figure 4 at an IC50 of less than 25 pg/ml.
[0031] The specificity of a binding agent may also be tested for binding to soluble trimers representing HIV proteins (e.g., soluble, cleaved SOSIP.664 gp140 trimers based on the subtype A transmitted/founder strain, BG505 as used in the examples herein). Preferred trimers (such as those used in the examples herein) are those being highly stable, homogenous and closely resembling native virus spikes when visualized by negative stain electron microscopy (EM) (Sanders, R. W. et al. A next-generation cleaved, soluble HIV-1 Env trimer, BG505 SOSIP.664 gp140, expresses multiple epitopes for broadly neutralizing but not nonneutralizing antibodies. PLoS Pathog. 9, e1003618 (2013)). Typically, broadly neutralizing antibodies against multiple neutralizing epitopes on HIV-1 Env will be highly reactive with such
trimers. Conversely, non-neutralizing antibodies (NAbs) to the CD4-binding site, CD4-induced epitopes or gp41 ectodomain would not (and did not in the example) react with the trimers, even when their epitopes were present on simpler forms of Env (e.g., gp120 monomers or dissociated gp41 subunits). The examples also included a test, which may be used in testing any of binding agents described herein, in which the MPER was also deleted to improve trimer solubility and reduce aggregate formation. The binding agents may also be tested for binding to such trimers in the presence or absence of soluble CD4 (sCD4). The examples herein describe the testing of the LN02 and PGT151 (binding to a site at the interface between gp120 and gp41 (Dingens et al. Cell Host Microbe. 2017 Jun 14;21 (6):777-787.e4. doi: 10.1016/j.chom.2017.05.003. Epub 2017 Jun 1)) antibodies for binding to the 426c WT SOSIP Env protein complex, measured by surface plasmon resonance (SPR)). As shown in the examples, the biotinylated lgG1 LN02 antibody was shown to bind the 426c WT SOSIP Env protein but was completely blocked from binding the Env protein that had been pre-incubated with an unlabeled LN02 antibody. In a similar experiment, the biotinylated interface binding bNab PGT151 bound tightly to 426c WT SOSIP Env protein but PGT151 bound more weakly to the LN02 +426c WT SOSIP Env protein complex. The results presented in the examples therefore indicate that lgG1 LN02 antibody might recognize an epitope at the gp120/gp41 interface of HIV-1 Env. Given that PGT151 binding to 426c WT SOSIP was not completely blocked by LN02, it is possible that LN02 binds in the same region but not identical epitope compared to PGT151. Other assay systems including surface plasmon resonance may be used to test the binding agents contemplated herein. And similar tests may also be performed on any of the binding agents contemplated herein.
[0032] The term “binding affinity” and/or KD refers to the dissociation rate of a particular antibody-antigen interaction. The KD is the ratio of the rate of dissociation (“off-rate (Kd)”) to the association rate (“on-rate” (Ka)). KD therefore equals Kd/Ka and is expressed as a molar concentration (M). Thus, the smaller the KD, the stronger the affinity of binding. For example, a KD of 1 mM indicates weak binding as compared to a KD of 1 nM. KD values for antibodies can be determined using methods well established in the art such as by using a Biacore® system. In some embodiments, the binding agents described herein may be compared with another binding agent with reference to the respective KD values of each. These properties may be combined with other characteristics such as neutralization capacity and/or epitope specificity in order to compare binding agents to one another. Accordingly, binding agents having a similar KD to those described herein, perhaps also sharing the neutralization capacity
and epitope specificity described herein (e.g., as exhibited by LN02), are also contemplated as part of this disclosure.
[0033] Any of the amino acid sequences of Table 1 (and / or any one or more fragments and / or derivatives thereof) may be also substituted by any other amino acid as desired by one of ordinary skill in the art. Preferably such substitutions do not interfere with a beneficial or desired property of the binding agent (e.g., having a similar specificities and/or KD to LN02). For example, one of skill in the art may make conservative substitutions by replacing particular amino acids with others as shown in Table 2 below. The specific amino acid substitution selected may depend on the location of the site selected. An amino acid substitution may be said to“correspond to” where one of ordinary skill in the art could ascertain a significant amount of similarity between the amino acid sequences surrounding the amino acid being substituted. For instance, a particular amino acid sequence may correspond to another where two, three, four or more N-terminal and C-terminal amino acids surrounding the amino acid being substituted are the same or similar (e.g., as described in Table 2) in the polypeptides being compared. Conservative amino acid substitutions may involve a substitution of a native amino acid residue with a non-native residue such that there is little or no effect on the size, polarity, charge, hydrophobicity, or hydrophilicity of the amino acid residue at that position and, in particular, does not result in, e.g., decreased HIV neutralization capacity and/or different epitope specificity.
Table 2
[0034] In certain embodiments, a nucleic acid molecule encoding one or more binding agents described herein may be inserted into one or more expression vectors, as discussed below in greater detail. In such embodiments, the binding agent may be encoded by nucleotides corresponding to the amino acid sequence. The particular combinations of nucleotides (codons) that encode the various amino acids (AA) are well known in the art, as described in various references used by those skilled in the art (e.g., Lewin, B. Genes V, Oxford University Press, 1994). The nucleotide sequences encoding the amino acids of said binding agents may be ascertained with reference to Table 3, for example. Nucleic acid variants may use any combination of nucleotides that encode the binding agent.
Table 3
Codons Encoding Amino Acids (AA)
Those of ordinary skill in the art understand that the nucleotide sequence encoding a particular amino acid sequence may be easily derived from the amino acid sequence of any of SEQ ID NOS. 1-8 and the information presented in Table 4. For instance, it may be deduced from the amino acid sequence YGSISRHFWG (SEQ ID NO.: 19) and the information presented in Table 4 that the amino acid sequence may be encoded by the nucleotide sequence TATG G CAG CATT AG COG CCATTTTT G G GG C (SEQ ID NO.: 20). Those of ordinary skill in the art would understand that nucleotide sequences encoding SEQ ID NOS. 1-8 may be deduced in the same way, and such nucleotide sequences are contemplated herein. Table 4 provides exemplary nucleic acid sequences encoding the amino acid sequences shown in Table 1 (SEQ ID NOS. 1-8):
TABLE 4
Thus, a nucleic acid encoding a binding agent of this disclosure may comprise one or more SEQ ID NOS. 9-16 and/or a fragment and/or derivative thereof (e.g., the binding agent comprising any of SEQ ID NOS. 1-8 (that can be conservatively substituted, e.g., as described in Table 2), encoded by a nucleotide sequence determined using standard techniques, e.g., as described in Table 3). Exemplary coding sequences included in such nucleic acids include but are not limited to those described in Table 4 (SEQ ID NOS. 9-16), or derivatives (e.g.,“wobble” derivatives in which the third member of codon is changed without causing an amino acid sequence
substitution), and/or fragments thereof. Expression vectors comprising such nucleic acid sequences are also contemplated by this disclosure. Where the binding agents are antibodies, nucleotide sequences encoding the variable regions thereof may also be isolated from the phage and / or hybridoma cells expressing the same cloned into expression vectors. Methods for producing such preparations are well-known in the art.
[0035] Nucleic acid molecules encoding one or more HIV binding agents may be contained within a viral and / or a non-viral vector. In one embodiment, a DNA vector is utilized to deliver nucleic acids encoding one or more HIV binding agents to the patient. In doing so, various strategies may be utilized to improve the efficiency of such mechanisms including, for example, the use of self-replicating viral replicons (Caley, et al. 1999. Vaccine , 17: 3124-2135; Dubensky, et al. 2000. Mol. Med. 6: 723-732; Leitner, et al. 2000. Cancer Res. 60: 51-55), codon optimization (Liu, et al. 2000. Mol. Then, 1 : 497-500; Dubensky, supra] Huang, et al. 2001. J. Virol. 75: 4947-4951), in vivo electroporation (Widera, et al. 2000. J. Immunol. 164: 4635-3640), incorporation of nucleic acids encoding co-stimulatory molecules, cytokines and / or chemokines (Xiang, et al. 1995. Immunity, 2: 129-135; Kim, et al. 1998. Eur. J. Immunol. , 28: 1089-1103; Iwasaki, et al. 1997. J. Immunol. 158: 4591-3301 ; Sheerlinck, et al. 2001. Vaccine, 19: 2647- 2656), incorporation of stimulatory motifs such as CpG (Gurunathan, supra ; Leitner, supra), sequences for targeting of the endocytic or ubiquitin-processing pathways (Thomson, et al. 1998. J. Virol. 72: 2246-2252; Velders, et al. 2001. J. Immunol. 166: 5366-5373), prime-boost regimens (Gurunathan, supra ; Sullivan, et al. 2000. Nature, 408: 605-609; Hanke, et al. 1998. Vaccine, 16: 439-445; Amara, et al. 2001. Science, 292: 69-74), proteasome-sensitive cleavage sites, and the use of mucosal delivery vectors such as Salmonella (Darji, et al. 1997. Cell, 91 : 765-775; Woo, et al. 2001. Vaccine, 19: 2945-2954). Other methods are known in the art, some of which are described below. Various viral vectors that have been successfully utilized for introducing a nucleic acid to a host include retrovirus, adenovirus, adeno-associated virus (AAV), herpes virus, and poxvirus, among others. The vectors may be constructed using standard recombinant techniques widely available to one skilled in the art. Such techniques may be found in common molecular biology references such as Molecular Cloning: A Laboratory Manual (Sambrook, et al., 1989, Cold Spring Harbor Laboratory Press), Gene Expression Technology (Methods in Enzymology, Vol. 185, edited by D. Goeddel, 1991. Academic Press, San Diego, CA), and PCR Protocols: A Guide to Methods and Applications (Innis, et al. 1990. Academic Press, San Diego, ca). “Non-viral” plasmid vectors may also be suitable in certain embodiments. Preferred plasmid vectors are compatible with bacterial, insect, and / or
mammalian host cells. Such vectors include, for example, PCR-ii, PCR3, and pcDNA3.1 (Invitrogen, San Diego, CA), pBSii (Stratagene, La Jolla, CA), pet15 (Novagen, Madison, Wl), pGEX (Pharmacia Biotech, Piscataway, NJ), pEGFp-n2 (Clontech, Palo Alto, CA), pETI (Bluebacii, Invitrogen), pDSR-alpha (PCT pub. No. WO 90/14363) and pFASTBACdual (Gibco-
BRL, Grand island, NY) as well as Bluescript plasmid derivatives (a high copy number COLel- based phagemid, Stratagene Cloning Systems, La Jolla, CA), PCR cloning plasmids designed for cloning TAQ-amplified PCR products (e.g. , TOPO™ TA cloning® kit, PCR2.1® plasmid derivatives, Invitrogen, Carlsbad, CA). Bacterial vectors may also be used. These vectors include, for example, Shigella, Salmonella, Vibrio cholerae, Lactobacillus, Bacille Calmette Guerin (BCG), and Streptococcus (see for example, WO 88/6626; WO 90/0594; WO 91/13157; WO 92/1796; and WO 92/21376). Many other non-viral plasmid expression vectors and systems are known in the art and may be use. Other delivery techniques may also suffice including, for example, DNA-ligand complexes, adenovirus-ligand-DNA complexes, direct injection of DNA, CaP04 precipitation, gene gun techniques, electroporation, and colloidal dispersion systems. Colloidal dispersion systems include macromolecule complexes, nanocapsules, microspheres, beads, and lipid-based systems including oil-in-water emulsions, micelles, mixed micelles, and liposomes. The preferred colloidal system is a liposome, which are artificial membrane vesicles useful as delivery vehicles in vitro and in vivo. RNA, DNA and intact virions can be encapsulated within the aqueous interior and be delivered to cells in a biologically active form (Fraley, R., et al., 1981 , Trends Biochem. Sci., 6: 77). The composition of the liposome is usually a combination of phospholipids, particularly high-phase-transition- temperature phospholipids, usually in combination with steroids, especially cholesterol. Other phospholipids or other lipids may also be used. The physical characteristics of liposomes depend on pH, ionic strength, and the presence of divalent cations. Examples of lipids useful in liposome production include phosphatidyl compounds, such as phosphatidylglycerol, phosphatidylcholine, phosphatidylserine, phosphatidylethanolamine, sphingolipids, cerebrosides, and gangliosides. Particularly useful are diacylphosphatidylglycerols, where the lipid moiety contains from 14-18 carbon atoms, particularly from 16-18 carbon atoms, and is saturated. Illustrative phospholipids include egg phosphatidylcholine, dipalmitoylphosphatidylcholine and distearoylphosphatidylcholine.
[0036] A cultured cell comprising the vector is also provided. The cultured cell may be a cultured cell transfected with the vector or a progeny of the cell, wherein the cell expresses the immunogenic polypeptide. Suitable cell lines are known to those of skill in the art and are
commercially available, for example, through the American Type Culture Collection (ATCC). The transfected cells can be used in a method of producing an immunogenic polypeptide. The method comprises culturing a cell comprising the vector under conditions that allow expression of the immunogenic polypeptide, optionally under the control of an expression sequence. The immunogenic polypeptide can be isolated from the cell or the culture medium using standard protein purification methods. In some embodiments, the binding agents described herein may be conjugated to active agents to target and inhibit the function of and /or eliminate cell populations expressing HIV polypeptides and/or harboring HIV (and / or another antigen in the case of binding agents with multiple specificities). For instance, CD4+ T-cell populations containing replication competent HIV may be targeted and eliminated using binding agent / drug conjugates (e.g., antibody-drug conjugates (ADC)). Mono- and/or bi-specific candidate binding agents may be conjugated with one or more types of drugs (e.g., drugs damaging DNA, targeting microtubules). The binding agents described herein and/ or derivatives thereof may also be adjoined to and / or conjugated to functional agents for in vitro and / or in vivo use. For instance, the binding agent may be adjoined to and / or conjugated to functional moieties such as cytotoxic drugs or toxins, and / or active fragments thereof such as diphtheria A chain, exotoxin A chain, ricin A chain, abrin A chain, curcin, crotin, phenomycin, enomycin, among others. Suitable functional moieties may also include radiochemicals. Binding agents, such as antibodies, may be adjoined to and / or conjugated to the one or more functional agents using standard techniques in the art.
[0037] In some embodiments, this disclosure provides binding agents with multiple specificities such that epitopes bound by an LN02 binding agent (e.g., antibody) and at least one other secondary antigen (e.g., a cell surface protein) may be bound by a single binding agent. In some embodiments, the secondary antigen may be one expressed by cells infected by an infectious agent. For instance, an exemplary secondary antigen may be HIV Env antigen other than gp41. Such binding agents may bind the secondary antigen and / or may serve to neutralize the infectious agent as may be determined using the assays described herein. Combinations of binding agents, such as one or more described herein with another available to those of ordinary skill in the art, are also contemplated herein. For instance, in some embodiments, the combinations may be identified to provide statistically significant differences from results (e.g., neutralization assays) obtained using only one or more of the binding agents and not others. In some embodiments, combinations exhibit additive and/or, preferably synergistic, neutralization of HIV, for example. In some embodiments, the combination may
comprise a first binding agent having the characteristics of an LN02 binding agent (e.g., antibody such as lgG1 LN02) and/or comprising any one or more of SEQ ID NOS. 1-8 (and/or as described in Table 1), and/or derivatives thereof, and any one or more of the antibodies described in any one or more of U.S. Pat. No. 5,087,557; U.S. Pat. No. 5,298,419; U.S. Pat. No. 5,459,060; U.S. Pat. No. 5,693,752; U.S. Pat. No. 5,731 ,189; U.S. Pat. No. 5,753,503; U.S. Pat. No. 5,756,674; U.S. Pat. No. 5,777,074; U.S. Pat. No. 5,804,440; U.S. Pat. No. 5,831 ,034; U.S. Pat. No. 6,008,044; U.S. Pat. No. 7,774,887B2; U.S. Pat. Publications 2003/01 18985A1 , 2007/0292390A1 , or 2014/0205612A1 ; WO 2002/032452A1 (e.g., binding the gp41 epitopes ELDKWA, ELEKWA, ELNKWA, ELDEWA); EP0335134B1 US 176077 (e.g., a humanized version of the mouse mAbs described therein); DE3932461 A1 (mAb against the epitope Arg-lle-Leu-Ala-Val-Glu-Arg-Leu-Lys-Try-Asp-Gln-Gln-Leu-Leu-Gly-lle-Trp-Gly-Cys- Ser); Evans, et al. J. Immunol. 140(3): 941-3 (1988); Gorney, et al. Proc. Natl. Acad. Sci. USA, 86: 1624-28 (1989); Teeuwsen, et al. (1990) AIDS Res. Hum. Retroviruses 6, 381-392; Earl, et al. J. Virol. 71 (4): 2674-2684 (1997); Jiang, et al. J. Virol. 72(12): 10213-17 (1998); Zwick, et al. J. Virol. 75(22): 10892-10905 (2001); Eckert et al. PNAS USA, 98(20): 1 1 187- 1 1192 (2001); Louis, et al. J. Biol. Chem. 278(22): 20278-20285 (2003); and/or Pietzsch, et al. J. Virol. 84(10): 5032-42 (2010); all of which are incorporated herein in their entirety. For instance, any of the binding agents described herein may be combined with (i.e. , as a single composition, and/or used in conjunction with) one or more the antibodies commonly known as 2F5, 4E10 and/or Z13e1 , and/or derivatives thereof, among others. The binding agents of such compositions may be different entities such as two or more different monoclonal antibodies or derivatives thereof, or may be found on the same entity such as a bi-functional antibody (a single antibody or derivative thereof comprising multiple binding specificities). Such combinations as described herein may also be combined with one or more other agents that may affect immune cell function such as antibodies against CTLA-4, and the like. One of ordinary skill in the art would recognize that many such combinations may be suitable for use as described herein.
[0038] As mentioned above, the HIV binding agents described herein may be used to treat and / or prevent and / or ameliorate the symptoms of infection by HIV. As is well-known in the art, HIV isolates are now classified into discrete genetic subtypes. HIV-1 is known to comprise at least ten subtypes (A1 , A2, A3, A4, B, C, D, E, F1 , F2, G, H, J and K) (Taylor et al, NEJM, 359(18): 1965-1966 (2008)). HIV-2 is known to include at least five subtypes (A, B, C, D, and E). Subtype B has been associated with the HIV epidemic in homosexual men and
intravenous drug users worldwide. Most HIV-1 immunogens, laboratory adapted isolates, reagents and mapped epitopes belong to subtype B. In sub-Saharan Africa, India and China, areas where the incidence of new HIV infections is high, HIV-1 subtype B accounts for only a small minority of infections, and subtype HIV-1 C appears to be the most common infecting subtype. Any of these types of isolates may be addressed using the binding agents described herein. One or more binding agents may also be administered with or in conjunction with one or more agents used to prevent, treat and / or ameliorate HIV such as for example, a protease inhibitor, an HIV entry inhibitor, a reverse transcriptase inhibitor, and / or an anti- retroviral nucleoside analog. Suitable compounds include, for example, Agenerase (amprenavir), Combivir (Retrovir / Epivir), Crixivan (indinavir), Emtriva (emtricitabine), Epivir (3tc / lamivudine), Epzicom, Fortovase / Invirase (saquinavir), Fuzeon (enfuvirtide), Hivid (ddc / zalcitabine), Kaletra (lopinavir), Lexiva (Fosamprenavir), Norvir (ritonavir), Rescriptor (delavirdine), Retrovir / AZT (zidovudine), Reyatax (atazanavir, BMS-232632), Sustiva (efavirenz), Trizivir (abacavir/ zidovudine / lamivudine), Truvada (Emtricitabine / Tenofovir DF), Videx (ddl / didanosine), Videx EC (ddl, didanosine), Viracept (nevirapine), Viread (tenofovir disoproxil fumarate), Zerit (d4T / stavudine), and Ziagen (abacavir) may be utilized. Other suitable agents are known to those of skill in the art and may be suitable for use as described herein. Such agents may either be used prior to, during, or after administration of the binding agents and / or use of the methods described herein.
[0039] The skilled artisan has many suitable techniques for using the binding agents (e.g., antibodies) described herein to identify biological samples containing proteins that bind thereto. For instance, antibodies may be utilized to isolate HIV or cells containing HIV and / or expressing HIV antigens using, for example, immunoprecipitation or other capture-type assay. This well-known technique is performed by attaching the antibody to a solid support or chromatographic material (e.g., a bead coated with Protein A, Protein G and / or Protein L). The bound antibody is then introduced into a solution either containing or believed to contain HIV antigens (e.g., an HIV-infected cell). The HIV antigen(s) may then bind to the antibody and non-binding materials are washed away under conditions in which the HIV antigen(s) remains bound to the antibody. The bound protein may then be separated from the antibody and analyzed as desired. Similar methods for isolating a protein using an antibody are well- known in the art. The binding agents (e.g., antibodies) may also be utilized to detect HIV or HIV antigens within a biological sample. For instance, the antibodies may be used in assays such as, for example, flow cytometric analysis, ELISA, immunoblotting (e.g., western blot), in
situ detection, immunocytochemistry, and / or immunhistochemistry. Methods of carrying out such assays are well-known in the art. In some embodiments, the binding agents may be adjoined to and / or conjugated to one or more detectable labels. For instance, suitable detectable labels may include, for instance, fluorosceins (e.g., DyLight, Cy3, Cy5, FITC, HiLyte Fluor 555, HiLyte Fluor 647; 5-carboxy-2,7-dichlorofluorescein; 5-Carboxyfluorescein (5-FAM); 5-HAT (Hydroxy Tryptamine); 5-Hydroxy Tryptamine (HAT); 6-JOE; 6-carboxyfluorescein (6- FAM); FITC; 6-carboxy-1 ,4-dichloro-2’,7’-dichlorofluorescein (TET); 6-carboxy-1 ,4-dichloro- 2’, 4’, 5’, 7’-tetrachlorofluorescein (HEX); 6-carboxy-4’,5’-dichloro-2’, 7’-dimethoxyfluorescein (JOE); Alexa fluors (e.g., 350, 405, 430, 488, 500, 514, 532, 546, 555, 568, 594, 610, 633, 635, 647, 660, 680, 700, 750); BODIPY fluorophores (e.g., 492/515, 493/503, 500/510, 505/515, 530/550, 542/563, 558/568, 564/570, 576/589, 581/591 , 630/650-X, 650/665-X, 665/676, FL, FL ATP, Fl-Ceramide, R6G SE, TMR, TMR-X conjugate, TMR-X, SE, TR, TR ATP, TR-X SE)), rhodamines (e.g., 1 10, 123, B, B 200, BB, BG, B extra, 5-carboxytetramethylrhodamine (5- TAMRA), 5 GLD, 6-Carboxyrhodamine 6G, Lissamine, Lissamine Rhodamine B, Phallicidine, Phalloidine, Red, Rhod-2, ROX (6-carboxy-X-rhodamine), 5-ROX (carboxy-X-rhodamine), Sulphorhodamine B can C, Sulphorhodamine G Extra, TAMRA (6-carboxytetramethyl- rhodamine), Tetramethylrhodamine (TRITC), WT), Texas Red, and / or Texas Red-X. Other detectable labels known in the art may also be suitable for use. Binding agents, such as antibodies, may be adjoined to and / or conjugated to the one or more detectable labels using standard techniques in the art.
[0040] The binding agents described herein may be also be used to determine the presence of a disease state in a patient, to predict prognosis, or to determine the effectiveness of a chemotherapeutic or other treatment regimen. Expression profile assays, performed as described herein or as is otherwise known in the art, may be used to determine the relative level of expression of HIV in a cell, for instance. The level of expression may then be correlated with base (e.g., control) levels to determine whether a particular disease is present within the patient, the patient’s prognosis, or whether a particular treatment regimen is effective. For example, if the patient is being treated with a particular anti-infective regimen, an increased or decreased level of expression of HIV in the patient’s tissues (e.g., in plasma) may indicate the regimen is worsening or improving the load of HIV in that host. The increase or decrease in expression may indicate the regimen is having or not having the desired effect and another therapeutic modality may therefore be selected.
[0041] It is also possible to use the binding agents described herein as reagents in drug screening assays to test, for example, new drug candidates. The reagents may be used to ascertain the effect of a drug candidate on the expression of the immunogenic target in a cell line, or a cell or tissue of a patient. The expression profiling technique may be combined with high throughput screening techniques to allow rapid identification of useful compounds and monitor the effectiveness of treatment with a drug candidate (see, for example, Zlokarnik, et al. , Science 279, 84-8 (1998)). Drug candidates may be chemical compounds, nucleic acids, proteins, antibodies, or derivatives therefrom, whether naturally occurring or synthetically derived. Drug candidates thus identified may be utilized, among other uses, as pharmaceutical compositions for administration to patients or for use in further screening assays.
[0042] In some embodiments, the binding agents are in purified form. A“purified” binding agent (e.g., antibody) may be one that is separated from at least about 50% of the proteins and / or other components with which it is initially found (e.g., as part of a hybridoma supernatant or ascites preparation in the case of a monoclonal antibody). A purified binding agent (e.g., antibody) may be one that is separated from at least about 50%, 60%, 75%, 90%, or 95% of the proteins and / or other components with which it is initially found.
[0043] The binding agents (e.g., polypeptides) and nucleic acids described herein may also be combined with one or more pharmaceutically acceptable carriers prior to administration to a host. A pharmaceutically acceptable carrier is a material that is not biologically or otherwise undesirable, e.g., the material may be administered to a subject, without causing any undesirable biological effects or interacting in a deleterious manner with any of the other components of the pharmaceutical composition in which it is contained. The carrier would naturally be selected to minimize any degradation of the active ingredient and to minimize any adverse side effects in the subject, as would be well known to one of skill in the art. Suitable pharmaceutical carriers and their formulations are described in, for example, Remington’s: The Science and Practice of Pharmacy, 21st Edition, David B. Troy, ed., Lippicott Williams & Wilkins (2005). Typically, an appropriate amount of a pharmaceutically-acceptable salt is used in the formulation to render the formulation isotonic. Examples of the pharmaceutically-acceptable carriers include, but are not limited to, sterile water, saline, buffered solutions like Ringer's solution, and dextrose solution. The pH of the solution is generally from about 5 to about 8 or from about 7 to about 7.5. Other carriers include sustained-release preparations such as semipermeable matrices of solid hydrophobic polymers containing polypeptides or fragments
thereof. Matrices may be in the form of shaped articles, e.g., films, liposomes or microparticles. It will be apparent to those persons skilled in the art that certain carriers may be more preferable depending upon, for instance, the route of administration and concentration of composition being administered. Carriers are those suitable for administration of polypeptides and / or fragments thereof to humans or other subjects. Pharmaceutical compositions may also include carriers, thickeners, diluents, buffers, preservatives, surface active agents, adjuvants, immunostimulants, in addition to the immunogenic polypeptide. Pharmaceutical compositions may also include one or more active ingredients such as antimicrobial agents, anti inflammatory agents and anesthetics. The pharmaceutical composition may be administered orally, parentally, by inhalation spray, rectally, intranodally, or topically in dosage unit formulations containing conventional pharmaceutically acceptable carriers, adjuvants, and vehicles. The term“pharmaceutically acceptable carrier” or“physiologically acceptable carrier” as used herein refers to one or more formulation materials suitable for accomplishing or enhancing the delivery of a nucleic acid, polypeptide, or peptide as a pharmaceutical composition. A“pharmaceutical composition” is a composition comprising a therapeutically effective amount of a nucleic acid or polypeptide. The terms “effective amount” and “therapeutically effective amount” each refer to the amount of a binding agent, nucleic acid or the like used to observe the desired therapeutic effect (e.g., eliminating HIV).
[0044] Methods for treating one or more disease conditions (e.g., HIV or cancer) in a mammalian host comprising administering to the mammal at least one or more effective doses of one or more binding agents (and / or derivative(s) thereof) described herein are also provided. In some embodiments, the binding agent is a monoclonal antibody or fragment or derivative thereof comprising one or more of SEQ ID NOS. 1-8, including all of SEQ ID NOS. 1-6; or SEQ ID NOS.: 7 and 8; the amino acid sequences shown in Table 1 ; and/or substituted derivatives and/or fragments thereof; as well as in some embodiments conservatively substituted variants thereof. The one or more binding agents may be administered in a dosage amount of about 1 to about 50 mg / kg, about 1 to about 30 mg / kg, or about 5 to about 30 mg / kg (e.g., about any of 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 21 , 22, 23, 24, 25, 26, 27, 28, 29, 30, 35, or 40 mg / kg). In certain embodiments, the one or more binding agents may be administered to the mammal (e.g., intradermally, intravenously, orally, rectally) at about 10 mg / kg one or more times. When multiple doses are administered, the doses may comprise about the same or different amount of binding agent in each dose. The doses may also be separated in time from one another by the same or different intervals. For
instance, the doses may be separated by about any of 6, 12, 24, 36, 48, 60, 72, 84, or 96 hours, one week, two weeks, three weeks, one month, two months, three months, four months, five months, six months, seven months, eight months, nine months, 10 months, 1 1 months, 12 months, 1.5 years, 2 years, 3 years, 4 years, 5 years, or any time period before, after, and / or between any of these time periods. In some embodiments, the binding agents may be administered in conjunction with other agents (e.g., anti-infective agents and/or chemotherapeutic agent). Such other agents may be administered about simultaneously with the binding agents, or at a different time and / or frequency. Other embodiments of such methods may also be appropriate as could be readily determined by one of ordinary skill in the art.
[0045] To assist the skilled artisan in using the binding agents such as antibodies described herein, the same may be provided in kit format. A kit including one or more of such binding agents and optionally other components necessary for using the same to detect cells expressing HIV is also provided. The binding agents of the kit may be provided in any suitable form, including frozen, lyophilized, or in a pharmaceutically acceptable buffer such as TBS or PBS. The kit may also include other reagents required for utilization of the binding agents in vitro or in vivo such as buffers (e.g., TBS, PBS), blocking agents (solutions including nonfat dry milk, normal sera, Tween-20 Detergent, BSA, or casein), and / or detection reagents (e.g., goat anti mouse IgG biotin, streptavidin-HRP conjugates, allophycocyanin, B-phycoerythrin, R- phycoerythrin, peroxidase, detectable labels, and other labels and / or staining kits (e.g., ABC Staining Kit, Pierce)). The kits may also include other reagents and / or instructions for using the antibodies in commonly utilized assays described above such as, for example, flow cytometric analysis, ELISA, immunoblotting (e.g., western blot), in situ detection, immunocytochemistry, and/or immunhistochemistry. In one embodiment, the kit provides a binding agent in purified form. In another embodiment, the binding agent may be provided in biotinylated form either alone or along with an avidin-conjugated detection reagent (e.g., antibody). In another embodiment, the kit includes a binding agents comprising one or more detectable labels that may be used to directly detect HIV. Buffers and the like required for using any of these systems are well-known in the art and / or may be prepared by the end-user or provided as a component of the kit. The kit may also include a solid support containing positive- and negative-control protein and / or tissue samples. For example, kits for performing spotting or western blot-type assays may include control cell or tissue lysates for use in SDS-PAGE or nylon or other membranes containing pre-fixed control samples with additional space for
experimental samples. Kits for visualization of HIV in cells on slides may include pre-formatted slides containing control cell or tissue samples with additional space for experimental samples. Other embodiments of kits are also contemplated herein as would be understood by those of ordinary skill in the art.
[0046] Thus, this disclosure provides binding agents such as the LN02 antibody with specificity for HIV (e.g., and/or an antigen thereof). In some embodiments, the binding agent is a polypeptide comprising at least one amino acid sequence selected from the group consisting of SEQ ID NOS. 1-8 and / or shown in Table 1 , and/or substituted derivatives and/or fragments thereof. In some embodiments, the binding agent is a polypeptide comprising one or more combinations of SEQ ID NOS. 1-8 and/or substituted derivatives and/or fragments thereof. In some embodiments, the binding agent is an antibody. In some embodiments, the binding agent is a polypeptide such as an antibody comprising a heavy chain CDR amino acid sequence selected from one of SEQ ID NO. 1 or a conservatively substituted variant thereof; one of SEQ ID NOS. 2 or a conservatively substituted variant thereof; and, one of SEQ ID NO. 3 or a conservatively substituted variant thereof. In some embodiments, the binding agent is a polypeptide such as an antibody comprising a light chain CDR amino acid sequence selected from one of SEQ ID NO. 4 or a conservatively substituted variant thereof; one of SEQ ID NO. 5 or a conservatively substituted variant thereof; and, one of SEQ ID NO. 6 or a conservatively substituted variant thereof. In some embodiments, the binding agent is a polypeptide such as an antibody comprising a VH amino acid sequence of any of SEQ ID NO. 7 or a conservatively substituted variant thereof. In some embodiments, the binding agent is a polypeptide such as an antibody comprising a VL amino acid sequence of any of SEQ ID NO. 8 or a conservatively substituted variant thereof. In some embodiments, this disclosure provides binding agents comprising a combination of a heavy chain variable region (VH) and a light chain variable region (VL) selected from the group consisting of: a VH comprising complementarity determining regions (CDRs) 1 , 2, and 3 as set forth in SEQ ID NOS: 1 , 2, and 3, respectively; and a VL comprising CDRs 1 , 2 and 3 as set forth in SEQ ID NOS: 4, 5, and 6, respectively; and/or a conservatively substituted variant thereof. In some embodiments, this disclosure provides binding agents comprising a combination of a heavy chain variable region (VH) and a light chain variable region (VL) selected from the group consisting of: a VH comprising SEQ ID NO. 7 or a conservatively substituted variant thereof; and a VL comprising SEQ ID NO, 8, or a conservatively substituted variant thereof. In some embodiments, the binding agents are antibodies such as monoclonal antibodies. In some embodiments, the binding agents are
human antibodies. In some embodiments, this disclosure also provides isolated polynucleotides encoding such binding agent(s), and/or an expression vector, and/or host cell, comprising the polynucleotide. In some embodiments, this disclosure also provides methods for expressing such a polynucleotide in order to express and produce said binding agent(s). In some embodiments, this disclosure provides methods for isolating said binding agent(s) and preparing compositions comprising the same. In some embodiments, this disclosure provides compositions comprising such binding agent(s), such as pharmaceutical compositions comprising at least one pharmaceutically acceptable carrier. In some embodiments, this disclosure provides methods for neutralizing HIV in vitro and/or in vivo. In some embodiments, this disclosure provides methods for administering the binding agents and/or polynucleotides to a subject such as a human being in order to, in some embodiments, treat and/or prevent infection by HIV or AIDS. Other embodiments are also contemplated by this disclosure as may be ascertained by those of ordinary skill in the art.
[0047] In some embodiments, the binding agents have specificity for an epitope comprising amino acid residues in the proximity of Q619, N677 and E500K at the gp120/gp41 interface of HIV-1 Env (corresponding residues underlined in SEQ ID NO. 17). In some embodiments, a binding agent of this disclosure may comprise any one or more of these binding specificities along with the neutralization characteristics described above but not of a control virus at a concentration is from 102-10° ug/ml, or between 10°-101 ug/ml, to at least about 50%, and/or the ability to the neutralize a majority of the 57 HIV-1 pseudoviruses listed in Figure 4 at an IC5o of less than 25. In some embodiments, nucleic acids encoding such binding agents are also provided as in Table 4.
[0048] In some embodiments, the binding agent is derived from or related to (e.g., by sequence or derivation) a human antibody, human IgG, human lgG1 , human lgG2, human lgG2a, human lgG2b, human lgG3, human lgG4, human IgM, human IgA, human lgA1 , human lgA2, human IgD, human IgE, canine antibody, canine IgGA, canine IgGB, canine IgGC, canine IgGD, chicken antibody, chicken IgA, chicken IgD, chicken IgE, chicken IgG, chicken IgM, chicken IgY, goat antibody, goat IgG, mouse antibody, mouse IgG, pig antibody, and / or rat antibody, and / or a derivative thereof. In some embodiments, the derivative may be selected from the group consisting of an Fab, Fab2, Fab’ single chain antibody, Fv, single chain, mono-specific antibody, bispecific antibody, trimeric antibody, multi-specific antibody, multivalent antibody, chimeric antibody, canine-human chimeric antibody, canine-mouse chimeric antibody, antibody
comprising a canine Fc, humanized antibody, human antibody, caninized antibody, CDR-grafted antibody, shark antibody, nanobody, and / or canelid antibody. In some embodiments, the binding agent comprises at least a least a first and second specificity, the first being against HIV gp41 and the second being against a different antigen (e.g., an antigen of an infectious agent such as HIV (e.g., env) and / or a tumor antigen). In some embodiments, the binding agent and / or derivative thereof may comprise a detectable label fixably attached thereto. In some embodiments, the binding agent of any one and / or derivative thereof comprises an effector moiety (e.g., a cytotoxic drug, toxin, diphtheria A chain, exotoxin A chain, ricin A chain, abrin A chain, curcin, crotin, phenomycin, enomycin, and radiochemical) fixably attached thereto. In some embodiments, polynucleotides encoding one or more binding agents are also provided (e.g., as an expression vector). Host cells comprising and / or expressing the polypeptide products of such polynucleotides are also provided. In some embodiments, compositions comprising at least one binding agent or derivative; at least one isolated polynucleotide; at least one expression vector; and / or, at least one host cell; or a combination thereof; and, a pharmaceutically acceptable carrier are also provided.
[0049] This disclosure also provides methods for detecting HIV on a cell, the method comprising contacting a test biological sample with a binding agent or derivative described herein and detecting the binding agent bound to the biological sample or components thereof. Such methods may be an in vivo method or an in vitro method. In some embodiments, the method may comprise comparing the amount of binding to the test biological sample or components thereof to the amount of binding to a control biological sample or components thereof, wherein increased binding to the test biological sample or components thereof relative to the control biological sample or components thereof indicates the presence of a cell expressing HIV polypeptides in the test biological sample (e.g., mammalian blood). In some embodiments, a kit for detecting the expression of HIV in or on a cell, the kit comprising a binding agent or derivative thereof and instructions for use. In some embodiments, the binding agent and / or derivative thereof is in lyophilized form. In some embodiments, this disclosure provides methods for treating, preventing and / or ameliorating an infectious disease, cancer and / or autoimmunity in a mammal comprising administering to the mammal at least one effective dose of a pharmaceutical composition comprising a binding agent or derivative thereof. In some embodiments, the infectious disease is human immunodeficiency virus (HIV). In some embodiments, multiple doses are administered to the animal. In some embodiments, the
binding agent and / or derivative thereof may be administered in a dosage amount of about 1 to 50 mg / kg.
[0050] The terms“about”,“approximately”, and the like, when preceding a list of numerical values or range, refer to each individual value in the list or range independently as if each individual value in the list or range was immediately preceded by that term. The terms mean that the values to which the same refer are exactly, close to, or similar thereto.
[0051] As used herein, a subject or a host is meant to be an individual. The subject can include domesticated animals, such as cats and dogs, livestock (e.g., cattle, horses, pigs, sheep, and goats), laboratory animals (e.g., mice, rabbits, rats, guinea pigs) and birds. In one aspect, the subject is a mammal such as a primate or a human.
[0052] Optional or optionally means that the subsequently described event or circumstance can or cannot occur, and that the description includes instances where the event or circumstance occurs and instances where it does not. For example, the phrase optionally the composition can comprise a combination means that the composition may comprise a combination of different molecules or may not include a combination such that the description includes both the combination and the absence of the combination (i.e., individual members of the combination).
[0053] Ranges may be expressed herein as from about one particular value, and/or to about another particular value. When such a range is expressed, another aspect includes from the one particular value and/or to the other particular value. Similarly, when values are expressed as approximations, by use of the antecedent about or approximately, it will be understood that the particular value forms another aspect. It will be further understood that the endpoints of each of the ranges are significant both in relation to the other endpoint, and independently of the other endpoint. Ranges (e.g., 90-100%) are meant to include the range per se as well as each independent value within the range as if each value was individually listed.
[0054] The term“combined” or“in combination” or“in conjunction” may refer to a physical combination of agents that are administered together or the use of two or more agents in a regimen (e.g., administered separately, physically and / or in time) for treating, preventing and / or ameliorating a particular disease.
[0055] When the terms treat, prevent, and / or ameliorate or derivatives thereof are used herein in connection with a given treatment for a given condition (e.g., preventing cancer infection by HIV), it is meant to convey that the treated patient either does not develop a clinically observable level of the condition at all, or develops it more slowly and/or to a lesser degree than he/she would have absent the treatment. These terms are not limited solely to a situation in which the patient experiences no aspect of the condition whatsoever. For example, a treatment will be said to have prevented the condition if it is given during exposure of a patient to a stimulus that would have been expected to produce a given manifestation of the condition, and results in the patient's experiencing fewer and/or milder symptoms of the condition than otherwise expected. For instance, a treatment can“prevent” infection by resulting in the patient's displaying only mild overt symptoms of the infection; it does not imply that there must have been no penetration of any cell by the infecting microorganism.
[0056] Similarly, reduce, reducing, and reduction as used herein in connection with prevention, treatment and / or amelioration of a given condition by a particular treatment typically refers to a subject developing an infection more slowly or to a lesser degree as compared to a control or basal level of developing an infection in the absence of a treatment (e.g., administration of one or more HIV binding agents). A reduction in the risk of infection may result in the patient's displaying only mild overt symptoms of the infection or delayed symptoms of infection; it does not imply that there must have been no penetration of any cell by the infecting microorganism.
[0057] All references cited within this disclosure are hereby incorporated by reference in their entirety. Certain embodiments are further described in the following examples. These embodiments are provided as examples only and are not intended to limit the scope of the claims in any way /
EXAMPLES
Example 1
Lymph Node Donors
[0058] Selection of HIV-1 lymph node donors for the isolation of broadly neutralizing antibodies. In order to isolate broadly neutralizing antibodies capable to broadly neutralize multi-clade HIV-1 isolates in 107 plasma samples from chronically infected patients naive to antiretroviral therapy were screened for the presence of high titers of antibodies able to neutralize a panel of nine (9) HIV-1 pseudoviruses from the Global Panel of HIV-1 reference
strains (DeCamp, A. et al. Global panel of HIV-1 Env reference strains for standardized assessments of vaccine-elicited neutralizing antibodies. J Virol 88, 2489-2507 (2014)). This analysis resulted in the identification of eight (8) patients (Figures 1 A-F) as lymph node donors for the subsequent isolation and characterization of potent broadly neutralizing antibodies. In particular, donor SA090 (highlighted with an arrow in Fig. 1 F) as having high virus neutralizing activity (and for the lack of background activity against the negative control MLV pseudovirus).
Example 2
Isolation and characterization of a potent HIV-1 broadly neutralizing antibody
[0059] Germinal center and Memory IgG B cells from donor SA090 were sorted separately according to IgG (i.e. IgA and IgM negative cells), CD19 and CD38 expression (germinal center B cells are CD38 positive, which is not present on memory B cells) (Figure 2) and interrogated for the production of HIV-1 neutralizing antibodies. In particular, highly pure IgG memory B cells and IgG germinal cells were seeded in separate plates as single cell micro-cultures on human feeder cells in the presence of Epstein-Barr Virus (EBV) (which also stimulate polyclonally memory B cells) and a cocktail composed TLR9 agonist CpG-2006, IL-2 (1000 lU/ml), IL-6 (10 ng/ml), IL-21 (10 ng/ml), and anti-BCR goat antibodies (triggers BCR). Supernatants from day 14 cultures were then tested in a primary screening using a 384-well based HIV-1 pseudoviruses neutralization assay (using in parallel two strains, CE1 176 and BJOX2000, representative of clade C and CRF07). Neutralization assays were undertaken on TZM-bl cells. In a 384-well plate, HIV-1 pseudoviruses that resulted in an output of 50-100 x 104 relative light units (RLU) were incubated with B cell culture supernatants for 1 h at 37% (5% C02) before the addition of 3000 TZM-bl cells. These were incubated for a further 72 h, after which supernatant was removed and 15 pi Steadylite reagent (Perkin Elmer) was added. Luciferase activity was detected 5 min later by reading the plates on a Synergy microplate luminometer (BioTek). The supernatants derived from germinal center B cells found to produce antibodies that cross-neutralize one or more of the HIV strains. The supernatants from these two cultures were further harvested and tested for their ability to neutralize pseudoviruses. One of these produced an antibody designated“LN02” and was found to neutralize HIV.
[0060] The LN02 antibody was characterized by determining the amino acid and nucleotide sequences of its variable regions and the complementarity determining regions (CDRs) ascertained. Accordingly, the binding agent termed “LN02” is an lgG1-type fully human monoclonal antibody having the CDR, VH and VL sequences as shown above in Table 1. The
LN02 antibody was also determined to be derived from the IGHV4-4*02 and IGLV3-21*01 germline genes and highly somatically mutated in variable genes of both heavy chain (31.2%) and kappa light chain (31.6%) compared to germ line.
[0061] Recombinant LN02 antibody was produced and tested against the Global Panel of nine (9) HIV-1 reference pseudoviruses on TZM-bl cells. Surprisingly, lgG1 LN02 antibody neutralized seven out of nine HIV-1 pseudoviruses with IC5o values ranging from 1 mg/ml to less than 0.1 mg/ml (Figure 3) and did not neutralize the negative control MLV pseudovirus. lgG1 LN02 antibody was subsequently tested on an extended panel of 57 HIV-1 pseudoviruses including clade A, clade A (T/F), clade B, clade B (T/F), clade BC, clade C, and clade C (T/F). As shown in Fig. 4, the LN02 antibody broadly neutralized 43 viruses out of 57 with IC5o below 25 pg/ml, i.e., about 75% of the tested viruses, with a median IC5o of 0.046 pg/ml. As also shown in Fig. 4, the LN02 antibody broadly neutralized 36 of out of 57 with IC8o below 25 pg/ml, i.e., about 63% of the tested viruses, with a median IC8o of 0.13 pg/ml. The analysis of the viruses neutralized as shown in Figs. 5A-G indicated that LN02 neutralizing activity is not clade-dependent for the pseudoviruses tested.
Example 4
LN02 Specificity
[0062] In order to better define the specificity of LN02 antibody, experiments were performed to determine whether the neutralization activity of LN02 was enhanced by a series of cleavage site mutations in the CAP45 HIV-1 virus encoding amino acid substitutions in Env. The neutralization activity and IC5o values relative to the WT CAP45 pseudo-virus are shown in Fig. 6 and Table 5 and show that the cleavage site mutation E500K and a combination of all cleavage site mutations together (CS) enhance the neutralization of LN02 by approximately 5- fold. These results are similar to those reported for the 35022 gp120/gp41 interface bNab (Huang and Connors. Nature. 2014 Nov 6;515(7525): 138-42. doi: 10.1038/nature13601. Epub 2014 Sep 3).
Table 5
impact of Cleavage site mutations and Q619Y substitution in Env of the
CAP45 HIV-1 virus
*Amino acid numbering corresponds to HXB2 Env sequence (SEQ ID NO.: 18)
[0063] Experiments were also performed to determine whether removal of the giycan at position 88 of three HIV-1 viruses enhanced neutralization by LN02. The results are presented in Table 6 show that the LN02 neutralization is enhanced by removal of the giycan at position 88 in gp120 suggesting this conserved giycan occludes the epitope for LN02. Similar results were reported for the 3BC315/3BC176 gp120/gp41 interface bNabs (Lee, et al. Nat Commun. 2015 Sep 25;6:8167. doi: 10.1038/ncomms9167). Additionally, LN02 had no detectable neutralization activity against the CAP45 HIV-1 virus with the Q619Y mutation. This substitution is located at the gp120/gp41 interface region of Env providing further evidence that LN02 is a gp120/gp41 interface bNab.
Table 6
Enhancement after removing the giycan at position 88 in three HIV-1 viruses
*Amino acid numbering corresponds to HXB2 Env sequence (SEQ ID NO.: 18)
[0064] Experiments were also performed to determine the binding of the LN02 mAb to the soluble HIV-1 Env trimer 426c WT SOSIP gp140 that expresses multiple epitopes for broadly
neutralizing antibodies, determined by surface plasmon resonance (Fig. 7A, solid line). HIV-1 Env trimer that has been pre-incubated with unlabeled LN02 completely blocks the binding of a biotinylated LN02 that is detected with a streptavidin probe (Fig. 7A, dashed line). The PGT151 monoclonal antibody (binding to a site at the interface between gp120 and gp41) bound with high affinity to 426c WT SOSIP gp140 protein (Fig. 7B, solid line), but PGT151 binding is significantly reduced to the LN02 + 426c WT SOSIPgp140 complex (Fig. 7B, dashed line). These results provide evidence that PGT151 and LN02 bind to an overlapping epitope on the 426c WT SOSIP gp140 protein.
[0065] The binding of the LN02 mAb, as measured by ELISA, to a set of HIV-1 Env antigens and a negative control antigen (soluble CD4 protein, sCD4) was also determined (Fig. 8). LN02 binds selectively to the gp140 ConsB, which is the only protein construct that is predominantly trimeric in tertiary structure.
[0066] The neutralization of LN02 mAb was also tested against a panel of CAP45 viruses that encode substitutions that confer resistance to gp120/gp41 interface bNabs. Profiling performed in parallel with CAP248-2B, PGT151 , VRC34, 3BC315 and 35022 interface bNabs (reported in the literature) showed that LN02 effectively neutralized all pseudoviruses that conferenced resistance to these bNabs. Additionally, all interface bNabs tested retained neutralization activity against the Q619Y substitution that conferred resistance to LN02. These results show that LN02 has a unique neutralization footprint compared to the other interface antibodies, as illustrated in Table 7.
Table 7
LN02 has a unique footprint among interface mAbs
*Amino acid numbering corresponds to HXB2 Env sequence (SEQ ID NO.: 18)
[0067] While certain embodiments have been described in terms of the preferred embodiments, it is understood that variations and modifications will occur to those skilled in the art. Therefore, it is intended that the appended claims cover all such equivalent variations that come within the scope of the following claims.
Claims
1. A binding agent that neutralizes HIV in an in vitro HIV neutralization assay and/or in vivo, the binding agent comprising at least one amino acid sequence selected from the group consisting of SEQ ID NOS. 1-8 and / or shown in Table 1 ; and/or a conservatively substituted variant thereof.
2. The binding agent of claim 1 comprising at least one heavy chain CDR amino acid sequence selected from the group consisting of SEQ ID NOS. 1-3; or comprises each of SEQ ID NOS. 1-3; and/or a conservatively substituted variant thereof.
3. The binding agent of claim 1 comprising at least one light chain CDR amino acid sequence selected from the group consisting of SEQ ID NOS. 4-6; or comprises each of SEQ ID NOS. 4-6; and/or a conservatively substituted variant thereof.
4. The binding agent of claim 1 comprising SEQ ID NO. 1 ; SEQ ID NO. 2; SEQ ID NO. 3; SEQ ID NO. 4; SEQ ID NO. 5; and, SEQ ID NO. 6; and/or a conservatively substituted derivative thereof.
5. The binding agent of claim 1 comprising at least one of SEQ ID NOS. 7 or SEQ ID NO. 8; and/or a conservatively substituted derivative thereof.
6. The binding agent of claim 1 comprising SEQ ID NO. 7 and SEQ ID NO. 8; and/or a conservatively substituted derivative thereof.
7. The binding agent of any one of claims 1-6 wherein the binding agent exhibits neutralization of HIV-1 pseudoviruses BJOX (CRF07_BC), CE1 176, TRO.1 1 (B), X1632 (G), CH1 19 (CRF07_BC), CNE55 (CRF01_AE), 25710 (C), CD0217(C) but not of the control virus SVA- MLV at a concentration is from 102-10° ug/ml, or between 10°-101 pg/ml.
8. The binding agent of claim 7 wherein the percent neutralization is at least about 50%.
9. The binding agent of any one of claims 1-8 wherein the binding agent neutralizes a majority of the 57 HIV-1 pseudoviruses listed in Figure 4 at an IC5o of less than 25 pg/ml.
10. The binding agent of any one of claims 1-9 that is an antibody.
1 1. The binding agent of claim 10 that is an isolated monoclonal antibody.
12. The binding agent of claim 11 wherein the monoclonal antibody is a human monoclonal antibody.
13. The binding agent of any one of claims -10-12 wherein the antibody isotype is lgG1 or lgG3.
14. The binding agent of any one of claims 1-13 derived from a human antibody, human IgG, human lgG1 , human lgG2, human lgG2a, human lgG2b, human lgG3, human lgG4, human IgM, human IgA, human lgA1 , human lgA2, human IgD, human IgE, canine antibody, canine
IgGA, canine IgGB, canine IgGC, canine IgGD, chicken antibody, chicken IgA, chicken IgD, chicken IgE, chicken IgG, chicken IgM, chicken IgY, goat antibody, goat IgG, mouse antibody, mouse IgG, pig antibody, and rat antibody.
15. A derivative of a binding agent of any one of claims 1-14.
16. The derivative of claim 15 selected from the group consisting of an Fab, Fab2, Fab’ single chain antibody, Fv, single chain, mono-specific antibody, bispecific antibody, trimeric antibody, multi-specific antibody, multivalent antibody, chimeric antibody, canine-human chimeric antibody, canine-mouse chimeric antibody, antibody comprising a canine Fc, humanized antibody, human antibody, caninized antibody, CDR-grafted antibody, shark antibody, nanobody, and canelid antibody.
17. The binding agent or derivative of any one of claims 1-16 comprising at least a least a first and second specificity, the first being against gp41 and the second being against a different antigen.
18. The binding agent or derivative of any one of claims 1-17 comprising a detectable label fixably attached thereto.
19. The binding agent of claim 18 wherein the detectable label is selected from the group consisting of fluorescein, DyLight, Cy3, Cy5, FITC, HiLyte Fluor 555, HiLyte Fluor 647, 5- carboxy-2,7-dichlorofluorescein, 5-carboxyfluorescein, 5-FAM, hydroxy tryptamine, 5- hydroxy tryptamine (5-HAT), 6-carboxyfluorescein (6-FAM), FITC, 6-carboxy-1 ,4-dichloro- 2’,7’-dichlorofluorescein (TET), 6-carboxy-1 ,4-dichloro-2’,4’,5’,7’-tetrachlorofluorescein (HEX), 6-carboxy-4’,5’-dichloro-2’,7’-dimethoxyfluorescein (6-JOE), an Alexa fluor, Alexa fluor 350, Alexa fluor 405, Alexa fluor 430, Alexa fluor 488, Alexa fluor 500, Alexa fluor 514, Alexa fluor 532, Alexa fluor 546, Alexa fluor 555, Alexa fluor 568, Alexa fluor 594, Alexa fluor 610, Alexa fluor 633, Alexa fluor 635, Alexa fluor 647, Alexa fluor 660, Alexa fluor 680, Alexa fluor 700, Alexa fluor 750, a BODIPY fluorophores, BODIPY 492/515, BODIPY 493/503, BODIPY 500/510, BODIPY 505/515, BODIPY 530/550, BODIPY 542/563, BODIPY 558/568, BODIPY 564/570, BODIPY 576/589, BODIPY 581/591 , BODIPY 630/650-X, BODIPY 650/665-X, BODIPY 665/676, FL, FL ATP, Fl-Ceramide, R6G SE, TMR, TMR-X conjugate, TMR-X, SE, TR, TR ATP, TR-X SE, a rhodamine, rhodamine 1 10, rhodamine 123, rhodamine B, rhodamine B 200, rhodamine BB, rhodamine BG, rhodamine B extra, 5- carboxytetramethylrhodamine (5-TAMRA), 5 GLD, 6-carboxyrhodamine 6G, Lissamine, Lissamine Rhodamine B, Phallicidine, Phalloidine, rhodamine red, Rhod-2, 6-carboxy-X- rhodamine (ROX), carboxy-X-rhodamine (5-ROX), Sulphorhodamine B can C,
Sulphorhodamine G Extra, 6-carboxytetramethylrhodamine (TAMRA), tetramethylrhodamine (TRITC), rhodamine WT, Texas Red, and Texas Red-X.
20. The binding agent or derivative of any one of claims 1-19 or derivative thereof comprising an effector moiety fixably attached thereto.
21. The binding agent or derivative of claim 20 wherein the effector moiety is selected from the group consisting of a cytotoxic drug, toxin, diphtheria A chain, exotoxin A chain, ricin A chain, abrin A chain, curcin, crotin, phenomycin, enomycin, and radiochemical.
22. An isolated polynucleotide encoding a binding agent of any one of claims 1-17.
23. The isolate polynucleotide of claim 22 comprising a nucleic acid sequence of at least one of SEQ ID NOS. 9-16.
24. An expression vector comprising one or more polynucleotides of claim 22 or 23.
25. A host cell comprising the isolated polynucleotide of claim 22 or 23 and / or the expression vector of claim 25.
26. A composition comprising at least one binding agent or derivative of any one of claims 1-21 ; at least one isolated polynucleotide of claim 22 or 23; or at least one expression vector of claim 24; and / or, at least one host cell of claim 25; or a combination thereof; and, a pharmaceutically acceptable carrier.
27. A method for detecting HIV on a cell, the method comprising contacting a test biological sample with a binding agent or derivative of any one of claims 1-21 and detecting the binding agent bound to the biological sample or components thereof.
28. The method of claim 27, further comprising comparing the amount of binding to the test biological sample or components thereof to the amount of binding to a control biological sample or components thereof, wherein increased binding to the test biological sample or components thereof relative to the control biological sample or components thereof indicates the presence of a cell expressing HIV in the test biological sample.
29. The method of claim 27 or 28 wherein the test biological sample is mammalian blood.
30. The method of any one of claims 27-29 wherein the method is an in vivo method.
31. The method of any one of claims 27-29 wherein the method is an in vitro method.
32. A method for treating, preventing and / or ameliorating HIV infection and/or AIDS in a mammal comprising administering to the mammal at least one effective dose of a pharmaceutical composition comprising a binding agent or derivative of any one of claims 1 21
33. The method of claim 32 wherein multiple doses are administered to the animal.
34. The method of claim 32 or 33 wherein the binding agent is administered in a dosage amount of about 1 to 50 mg / kg.
35. A kit for detecting the expression of HIV in or on a cell, the kit comprising a binding agent or derivative of any one of claims 1-21 and instructions for use.
36. The kit of claim 35 wherein the binding agent, antibody, or derivative is in lyophilized form.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201862697462P | 2018-07-13 | 2018-07-13 | |
| US62/697,462 | 2018-07-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020012435A1 true WO2020012435A1 (en) | 2020-01-16 |
Family
ID=67909436
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2019/055966 Ceased WO2020012435A1 (en) | 2018-07-13 | 2019-07-12 | Hiv binding agents |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2020012435A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114675024A (en) * | 2022-03-03 | 2022-06-28 | 中国农业科学院兰州兽医研究所 | Porcine Seneca virus A neutralizing antibody, competitive ELISA detection kit and detection method |
| CN117535316A (en) * | 2024-01-04 | 2024-02-09 | 湖南工程学院 | A ginseng PgJOX4 gene and its application in regulating ginsenoside biosynthesis |
Citations (31)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US176077A (en) | 1876-04-11 | Improvement in horse-collars | ||
| US4816567A (en) | 1983-04-08 | 1989-03-28 | Genentech, Inc. | Recombinant immunoglobin preparations |
| EP0335134A2 (en) | 1988-03-30 | 1989-10-04 | Abbott Laboratories | Mouse monoclonal antibody to human immunodeficiency virus gp41 protein |
| DE3932461A1 (en) | 1988-09-30 | 1990-04-05 | Olympus Optical Co | MONOCLONAL ANTIBODY AGAINST A SYNTHETIC PEPTIDE AND METHOD FOR THE PRODUCTION THEREOF |
| WO1990010594A1 (en) | 1989-03-08 | 1990-09-20 | August Bilstein Gmbh & Co. Kg | Vehicle jack |
| WO1990014363A1 (en) | 1989-05-19 | 1990-11-29 | Amgen Inc. | Metalloproteinase inhibitor |
| WO1991013157A1 (en) | 1990-02-26 | 1991-09-05 | Commonwealth Scientific And Industrial Research Organisation | Shuttle plasmid for escherichia coli and mycobacteria |
| US5087557A (en) | 1986-06-23 | 1992-02-11 | Genetic Systems Corporation | Human monoclonal antibody to lymphadenopathy-associated virus |
| WO1992011796A1 (en) | 1991-01-05 | 1992-07-23 | Deutsche Lufthansa Aktiengesellschaft | Process for washing crockery in large plants |
| WO1992021376A1 (en) | 1991-06-06 | 1992-12-10 | Med Immune, Inc. | Induction of ctl responses to foreign antigens expressed in mycobacteria |
| US5298419A (en) | 1988-03-31 | 1994-03-29 | The Arizona Board Of Regents On Behalf Of The University Of Arizona | Human hybridomas and monoclonal antibodies which bind both gp41 and gp120 envelope proteins of human immunodeficiency virus |
| US5459060A (en) | 1989-08-24 | 1995-10-17 | Bioclonetics Incorporated | Human monoclonal antibodies directed against the transmembrane glycoprotein (gp41) of human immunodeficiency virus-1 (HIV-1) |
| US5545806A (en) | 1990-08-29 | 1996-08-13 | Genpharm International, Inc. | Ransgenic non-human animals for producing heterologous antibodies |
| US5545807A (en) | 1988-10-12 | 1996-08-13 | The Babraham Institute | Production of antibodies from transgenic animals |
| US5569825A (en) | 1990-08-29 | 1996-10-29 | Genpharm International | Transgenic non-human animals capable of producing heterologous antibodies of various isotypes |
| US5625126A (en) | 1990-08-29 | 1997-04-29 | Genpharm International, Inc. | Transgenic non-human animals for producing heterologous antibodies |
| US5633425A (en) | 1990-08-29 | 1997-05-27 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5661016A (en) | 1990-08-29 | 1997-08-26 | Genpharm International Inc. | Transgenic non-human animals capable of producing heterologous antibodies of various isotypes |
| US5693752A (en) | 1992-05-14 | 1997-12-02 | Hermann Katinger | Peptides that induce antibodies which neutralize genetically divergent HIV-1 isolates |
| US5731189A (en) | 1989-02-28 | 1998-03-24 | New York University | Human monoclonal antibodies to human immunodeficiency virus |
| US5753503A (en) | 1987-11-13 | 1998-05-19 | Hermann W. D. Katinger | Human monoclonal anti-HIV-I-antibodies |
| US5804440A (en) | 1992-09-30 | 1998-09-08 | The Scripps Research Institute | Human neutralizing monoclonal antibodies to human immunodeficiency virus |
| US5831034A (en) | 1987-11-13 | 1998-11-03 | Hermann Katinger | Human monoclonal anti-HIV-I-antibodies |
| US6008044A (en) | 1989-08-24 | 1999-12-28 | Bioclonetics | Human monoclonal antibodies directed against the transmembrane glycoprotein (gp41) of human immunodeficiency virus-1 (HIV-1) and detection of antibodies against epitope (GCSGKLIC) |
| WO2002032452A1 (en) | 2000-08-18 | 2002-04-25 | Tsinghua University | Composition for aids and method producing it |
| US20030118985A1 (en) | 1997-05-14 | 2003-06-26 | Hunt Jeffrey C. | Mouse monoclonal antibody (5-21-3) to human immunodeficiency virus gp41 protein |
| US20070292390A1 (en) | 2004-10-29 | 2007-12-20 | Dimitrov Dimiter S | Broadly Cross-Reactive Hiv-1 Neutralizing Human Monoclonal Antibodies |
| US7774887B2 (en) | 2003-10-28 | 2010-08-17 | Applied Materials, Inc. | Scrubber box and methods for using the same |
| US20140205612A1 (en) | 2010-08-31 | 2014-07-24 | Theraclone Sciences, Inc. | Human immunodeficiency virus (hiv)-neutralizing antibodies |
| WO2015103549A1 (en) * | 2014-01-03 | 2015-07-09 | The United States Of America, As Represented By The Secretary Department Of Health And Human Services | Neutralizing antibodies to hiv-1 env and their use |
| WO2017093985A1 (en) * | 2015-12-05 | 2017-06-08 | Centre Hospitalier Universitaire Vaudois | Hiv binding agents |
-
2019
- 2019-07-12 WO PCT/IB2019/055966 patent/WO2020012435A1/en not_active Ceased
Patent Citations (33)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US176077A (en) | 1876-04-11 | Improvement in horse-collars | ||
| US4816567A (en) | 1983-04-08 | 1989-03-28 | Genentech, Inc. | Recombinant immunoglobin preparations |
| US5087557A (en) | 1986-06-23 | 1992-02-11 | Genetic Systems Corporation | Human monoclonal antibody to lymphadenopathy-associated virus |
| US5753503A (en) | 1987-11-13 | 1998-05-19 | Hermann W. D. Katinger | Human monoclonal anti-HIV-I-antibodies |
| US5831034A (en) | 1987-11-13 | 1998-11-03 | Hermann Katinger | Human monoclonal anti-HIV-I-antibodies |
| EP0335134A2 (en) | 1988-03-30 | 1989-10-04 | Abbott Laboratories | Mouse monoclonal antibody to human immunodeficiency virus gp41 protein |
| US5298419A (en) | 1988-03-31 | 1994-03-29 | The Arizona Board Of Regents On Behalf Of The University Of Arizona | Human hybridomas and monoclonal antibodies which bind both gp41 and gp120 envelope proteins of human immunodeficiency virus |
| DE3932461A1 (en) | 1988-09-30 | 1990-04-05 | Olympus Optical Co | MONOCLONAL ANTIBODY AGAINST A SYNTHETIC PEPTIDE AND METHOD FOR THE PRODUCTION THEREOF |
| US5545807A (en) | 1988-10-12 | 1996-08-13 | The Babraham Institute | Production of antibodies from transgenic animals |
| US5731189A (en) | 1989-02-28 | 1998-03-24 | New York University | Human monoclonal antibodies to human immunodeficiency virus |
| WO1990010594A1 (en) | 1989-03-08 | 1990-09-20 | August Bilstein Gmbh & Co. Kg | Vehicle jack |
| WO1990014363A1 (en) | 1989-05-19 | 1990-11-29 | Amgen Inc. | Metalloproteinase inhibitor |
| US6008044A (en) | 1989-08-24 | 1999-12-28 | Bioclonetics | Human monoclonal antibodies directed against the transmembrane glycoprotein (gp41) of human immunodeficiency virus-1 (HIV-1) and detection of antibodies against epitope (GCSGKLIC) |
| US5459060A (en) | 1989-08-24 | 1995-10-17 | Bioclonetics Incorporated | Human monoclonal antibodies directed against the transmembrane glycoprotein (gp41) of human immunodeficiency virus-1 (HIV-1) |
| WO1991013157A1 (en) | 1990-02-26 | 1991-09-05 | Commonwealth Scientific And Industrial Research Organisation | Shuttle plasmid for escherichia coli and mycobacteria |
| US5633425A (en) | 1990-08-29 | 1997-05-27 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5661016A (en) | 1990-08-29 | 1997-08-26 | Genpharm International Inc. | Transgenic non-human animals capable of producing heterologous antibodies of various isotypes |
| US5569825A (en) | 1990-08-29 | 1996-10-29 | Genpharm International | Transgenic non-human animals capable of producing heterologous antibodies of various isotypes |
| US5545806A (en) | 1990-08-29 | 1996-08-13 | Genpharm International, Inc. | Ransgenic non-human animals for producing heterologous antibodies |
| US5625126A (en) | 1990-08-29 | 1997-04-29 | Genpharm International, Inc. | Transgenic non-human animals for producing heterologous antibodies |
| US5777074A (en) | 1990-12-26 | 1998-07-07 | Bioclonetics Incorporated | Human monoclonal antibodies directed against the transmembrane glycoprotein (GP41) of human immunodeficiency virus-1 (HIV-1) |
| WO1992011796A1 (en) | 1991-01-05 | 1992-07-23 | Deutsche Lufthansa Aktiengesellschaft | Process for washing crockery in large plants |
| WO1992021376A1 (en) | 1991-06-06 | 1992-12-10 | Med Immune, Inc. | Induction of ctl responses to foreign antigens expressed in mycobacteria |
| US5756674A (en) | 1992-05-14 | 1998-05-26 | Herman Katinger | Peptides that induce antibodies which neutralize genetically divergent HIV-1 isolates |
| US5693752A (en) | 1992-05-14 | 1997-12-02 | Hermann Katinger | Peptides that induce antibodies which neutralize genetically divergent HIV-1 isolates |
| US5804440A (en) | 1992-09-30 | 1998-09-08 | The Scripps Research Institute | Human neutralizing monoclonal antibodies to human immunodeficiency virus |
| US20030118985A1 (en) | 1997-05-14 | 2003-06-26 | Hunt Jeffrey C. | Mouse monoclonal antibody (5-21-3) to human immunodeficiency virus gp41 protein |
| WO2002032452A1 (en) | 2000-08-18 | 2002-04-25 | Tsinghua University | Composition for aids and method producing it |
| US7774887B2 (en) | 2003-10-28 | 2010-08-17 | Applied Materials, Inc. | Scrubber box and methods for using the same |
| US20070292390A1 (en) | 2004-10-29 | 2007-12-20 | Dimitrov Dimiter S | Broadly Cross-Reactive Hiv-1 Neutralizing Human Monoclonal Antibodies |
| US20140205612A1 (en) | 2010-08-31 | 2014-07-24 | Theraclone Sciences, Inc. | Human immunodeficiency virus (hiv)-neutralizing antibodies |
| WO2015103549A1 (en) * | 2014-01-03 | 2015-07-09 | The United States Of America, As Represented By The Secretary Department Of Health And Human Services | Neutralizing antibodies to hiv-1 env and their use |
| WO2017093985A1 (en) * | 2015-12-05 | 2017-06-08 | Centre Hospitalier Universitaire Vaudois | Hiv binding agents |
Non-Patent Citations (69)
| Title |
|---|
| "Gene Expression Technology (Methods in Enzymology", vol. 185, 1991, ACADEMIC PRESS |
| A. DECAMP ET AL: "Global Panel of HIV-1 Env Reference Strains for Standardized Assessments of Vaccine-Elicited Neutralizing Antibodies", JOURNAL OF VIROLOGY., vol. 88, no. 5, 18 December 2013 (2013-12-18), US, pages 2489 - 2507, XP055643800, ISSN: 0022-538X, DOI: 10.1128/JVI.02853-13 * |
| AMARA ET AL., SCIENCE, vol. 292, 2001, pages 69 - 74 |
| BOERNER ET AL., J. IMMUNOL., vol. 147, no. 1, 1991, pages 86 - 95 |
| CALEY ET AL., VACCINE, vol. 17, 1999, pages 3124 - 2135 |
| COLE ET AL.: "Monoclonal Antibodies and Cancer Therapy", 1985, ALAN R. LISS, pages: 77 |
| CONSTANTINOS KURT WIBMER ET AL: "Structure and Recognition of a Novel HIV-1 gp120-gp41 Interface Antibody that Caused MPER Exposure through Viral Escape", PLOS PATHOGENS, vol. 13, no. 1, 1 November 2017 (2017-11-01), pages e1006074, XP055643850, DOI: doi.org/10.1371/journal.ppat.1006074 * |
| DARJI ET AL., CELL, vol. 91, 1997, pages 765 - 775 |
| DECAMP, A. ET AL.: "Global panel of HIV-1 Env reference strains for standardized assessments of vaccine-elicited neutralizing antibodies", J VIROL, vol. 88, 2014, pages 2489 - 2507 |
| DINGENS ET AL., CELL HOST MICROBE, vol. 21, no. 6, 14 June 2017 (2017-06-14), pages 777 - 787 |
| DUBENSKY ET AL., MOL. MED., vol. 6, 2000, pages 723 - 732 |
| EARL ET AL., J. VIROL., vol. 71, no. 4, 1997, pages 2674 - 2684 |
| EBERSBACH ET AL., J. MOL. BIOL., vol. 372, no. 1, 2007, pages 172 - 85 |
| ECKERT ET AL., PNAS USA, vol. 98, no. 20, 2001, pages 11187 - 11192 |
| EDMONDS ET AL., VIROLOGY, vol. 408, no. 1, 2010, pages 1 - 13 |
| EMILIA FALKOWSKA ET AL: "Broadly Neutralizing HIV Antibodies Define a Glycan-Dependent Epitope on the Prefusion Conformation of gp41 on Cleaved Envelope Trimers", IMMUNITY, vol. 40, no. 5, 1 May 2014 (2014-05-01), pages 657 - 668, XP055178550, ISSN: 1074-7613, DOI: 10.1016/j.immuni.2014.04.009 * |
| EVANS ET AL., J. IMMUNOL., vol. 140, no. 3, 1988, pages 941 - 3 |
| FISHWILD ET AL., NATURE BIOTECHNOLOGY, vol. 14, 1996, pages 826 - 51 |
| FRALEY, R. ET AL., TRENDS BIOCHEM. SCI., vol. 6, 1981, pages 77 |
| GORNEY ET AL., PROC. NATL. ACAD. SCI. USA, vol. 86, 1989, pages 1624 - 28 |
| GRABULOVSKI ET AL., J. BIOL. CHEM., vol. 282, no. 5, 2007, pages 3196 - 3204 |
| HANKE ET AL., VACCINE, vol. 16, 1998, pages 439 - 445 |
| HOOGENBOOM ET AL., J. MOL. BIOL., vol. 222, 1991, pages 581 |
| HUANGCONNORS, NATURE, vol. 515, no. 7525, 6 November 2014 (2014-11-06), pages 138 - 42 |
| IWASAKI ET AL., J. IMMUNOL., vol. 158, 1997, pages 4591 - 3301 |
| JEONG HYUN LEE ET AL: "Antibodies to a conformational epitope on gp41 neutralize HIV-1 by destabilizing the Env spike", NATURE COMMUNICATIONS, vol. 6, 25 September 2015 (2015-09-25), GB, pages 8167, XP055234636, ISSN: 2041-1723, DOI: 10.1038/ncomms9167 * |
| JINGHE HUANG ET AL: "Broad and potent HIV-1 neutralization by a human antibody that binds the gp41-gp120 interface", NATURE, vol. 515, no. 7525, 3 September 2014 (2014-09-03), pages 138 - 142, XP055178495, ISSN: 0028-0836, DOI: 10.1038/nature13601 * |
| JOHANNES F. SCHEID ET AL: "Broad diversity of neutralizing antibodies isolated from memory B cells in HIV-infected individuals", NATURE, vol. 458, no. 7238, 2 April 2009 (2009-04-02), pages 636 - 640, XP055003289, ISSN: 0028-0836, DOI: 10.1038/nature07930 * |
| JONES ET AL., NATURE, vol. 321, 1986, pages 522 - 525 |
| KIM ET AL., EUR. J. IMMUNOL., vol. 28, 1998, pages 1089 - 1103 |
| KOHLERMILSTEIN, NATURE, vol. 256, 1975, pages 495 |
| KOIDE ET AL., METHODS MOL. BIOL., vol. 352, 2007, pages 95 - 109 |
| KREHENBRINK ET AL., J. MOL. BIOL., vol. 383, no. 5, 2008, pages 1058 - 68 |
| LEE ET AL., NAT COMMUN., vol. 6, 25 September 2015 (2015-09-25), pages 8167 |
| LEITNER ET AL., CANCER RES., vol. 60, 2000, pages 51 - 55 |
| LEWIN, B.: "Genes V", 1994, OXFORD UNIVERSITY PRESS |
| LIU ET AL., MOL. THER., vol. 1, 2000, pages 497 - 500 |
| LONBERG ET AL., NATURE, vol. 368, 1994, pages 812 - 859 |
| LONBERGHUSZAR, INTERN. REV. IMMUNOL., vol. 13, 1995, pages 65 - 93 |
| LOUIS ET AL., J. BIOL. CHEM., vol. 278, no. 22, 2003, pages 20278 - 20285 |
| LOUISE SCHARF ET AL: "Antibody 8ANC195 Reveals a Site of Broad Vulnerability on the HIV-1 Envelope Spike", CELL REPORTS, vol. 7, no. 3, 1 May 2014 (2014-05-01), pages 785 - 795, XP055178551, ISSN: 2211-1247, DOI: 10.1016/j.celrep.2014.04.001 * |
| LOUISE SCHARF ET AL: "Broadly Neutralizing Antibody 8ANC195 Recognizes Closed and Open States of HIV-1 Env", CELL, vol. 162, no. 6, 10 September 2015 (2015-09-10), AMSTERDAM, NL, pages 1379 - 1390, XP055644015, ISSN: 0092-8674, DOI: 10.1016/j.cell.2015.08.035 * |
| MACCALLUM ET AL., J. MOL. BIOL., vol. 262, 1996, pages 732 - 745 |
| MAKABE ET AL., JOURNAL OF BIOLOGICAL CHEMISTRY, vol. 283, 2008, pages 1156 - 1166 |
| MARKS ET AL., BIO/TECHNOLOGY, vol. 10, 1992, pages 779 - 783 |
| MEI-YUN ZHANG ET AL: "Identification and Characterization of a Broadly Cross-Reactive HIV-1 Human Monoclonal Antibody That Binds to Both gp120 and gp41", PLOS ONE, vol. 7, no. 9, 10 September 2012 (2012-09-10), pages e44241, XP055178544, DOI: 10.1371/journal.pone.0044241 * |
| MONTEFIORI, D.C.: "Remington's: The Science and Practice of Pharmacy", 2005, LIPPICOTT WILLIAMS & WILKINS |
| NIXON ET AL., CURR. OPIN. DRUG DISCOV. DEVEL., vol. 9, no. 2, 2006, pages 261 - 8 |
| NYGREN ET AL., FEBS J., vol. 275, no. 11, 2008, pages 2677 - 83 |
| PACE ET AL., PNAS USA, vol. 110, no. 33, 2013, pages 13540 - 13545 |
| PEREZ ET AL.: "Utilization of immunoglobulin G Fc receptors by human immunodeficiency virus type 1: a specific role for antibodies against the membrane-proximal external region of gp41", J VIROL, vol. 83, 2009, pages 7397 - 7410 |
| PIETZSCH ET AL., J. VIROL., vol. 84, no. 10, 2010, pages 5032 - 1452 |
| PRESTA, CURR. OP. STRUCT. BIOL., vol. 2, 1992, pages 593 - 596 |
| RIECHMANN ET AL., NATURE, vol. 332, 1988, pages 323 - 329 |
| RUDIKOFF S ET AL: "Single amino acid substitution altering antigen-binding specificity", PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES, NATIONAL ACADEMY OF SCIENCES, vol. 79, no. 6, 1 March 1982 (1982-03-01), pages 1979 - 1983, XP002683593, ISSN: 0027-8424, DOI: 10.1073/PNAS.79.6.1979 * |
| SANDERS, R. W. ET AL.: "A next-generation cleaved, soluble HIV-1 Env trimer, BG505 SOSIP.664 gp140, expresses multiple epitopes for broadly neutralizing but not non-neutralizing antibodies", PLOS PATHOG., vol. 9, 2013, pages e1003618, XP055131150, doi:10.1371/journal.ppat.1003618 |
| SHEERLINCK ET AL., VACCINE, vol. 19, 2001, pages 2945 - 2954 |
| SILVERMAN ET AL., NAT. BIOTECHNOL., vol. 23, no. 12, 2005, pages 1556 - 61 |
| STUMPP ET AL., DRUG DISCOV. TODAY, vol. 13, no. 15-16, 2008, pages 695 - 701 |
| SULLIVAN ET AL., NATURE, vol. 408, 2000, pages 605 - 609 |
| TAYLOR ET AL., NEJM, vol. 359, no. 18, 2008, pages 1965 - 1966 |
| TEEUWSEN ET AL., AIDS RES. HUM. RETROVIRUSES, vol. 6, 1990, pages 381 - 392 |
| THOMSON ET AL., J. VIROL., vol. 72, no. 12, 1998, pages 10213 - 2252 |
| VELDERS ET AL., J. IMMUNOL., vol. 166, 2001, pages 5366 - 5373 |
| VERHOEYEN ET AL., SCIENCE, vol. 239, 1988, pages 1534 - 1536 |
| WIDERA ET AL., J. IMMUNOL., vol. 164, 2000, pages 4635 - 3640 |
| XIANG ET AL., IMMUNITY, vol. 2, 1995, pages 129 - 135 |
| ZLOKARNIK ET AL., SCIENCE, vol. 279, 1998, pages 84 - 8 |
| ZWICK ET AL., J. VIROL., vol. 75, no. 22, 2001, pages 10892 - 10905 |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114675024A (en) * | 2022-03-03 | 2022-06-28 | 中国农业科学院兰州兽医研究所 | Porcine Seneca virus A neutralizing antibody, competitive ELISA detection kit and detection method |
| CN114675024B (en) * | 2022-03-03 | 2023-08-15 | 中国农业科学院兰州兽医研究所 | Pig type-A sai virus neutralizing antibody, competitive ELISA detection kit and detection method |
| CN117535316A (en) * | 2024-01-04 | 2024-02-09 | 湖南工程学院 | A ginseng PgJOX4 gene and its application in regulating ginsenoside biosynthesis |
| CN117535316B (en) * | 2024-01-04 | 2024-03-29 | 湖南工程学院 | A ginseng PgJOX4 gene and its application in regulating ginsenoside biosynthesis |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20220169733A1 (en) | Immunological Reagents | |
| US20210009661A1 (en) | HIV Binding Agents | |
| EP3405497A2 (en) | Immunological reagents | |
| US20220411492A1 (en) | Antibody against claudin 18a2 and use thereof | |
| KR20230127305A (en) | Neutralizing monoclonal antibody to COVID-19 | |
| US9982052B2 (en) | Immunological reagents | |
| WO2020012435A1 (en) | Hiv binding agents | |
| US20250312438A1 (en) | Humanized constructs, vaccines, and methods | |
| US11214617B2 (en) | Immunological reagents | |
| JP7678790B2 (en) | HIV binding agent | |
| CN117062624A (en) | Neutralizing monoclonal antibodies against COVID-19 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19766092 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 19766092 Country of ref document: EP Kind code of ref document: A1 |